Systems, Methods And Devices For Delivery Systems, Methods And Devices For Implanting Prosthetic Heart Valves
Chambers; Jeffrey W. ; et al.
U.S. patent application number 16/877053 was filed with the patent office on 2020-09-03 for systems, methods and devices for delivery systems, methods and devices for implanting prosthetic heart valves. The applicant listed for this patent is 4C Medical Technologies, Inc.. Invention is credited to Gregory G. Brucker, Jeffrey W. Chambers, Jason S. Diedering, James E. Flaherty, Joseph P. Higgins, Karl A. Kabarowski, Saravana B. Kumar, Jeffrey R. Stone, Robert J. Thatcher.
| Application Number | 20200276013 16/877053 |
| Document ID | / |
| Family ID | 1000004838403 |
| Filed Date | 2020-09-03 |









| United States Patent Application | 20200276013 |
| Kind Code | A1 |
| Chambers; Jeffrey W. ; et al. | September 3, 2020 |
SYSTEMS, METHODS AND DEVICES FOR DELIVERY SYSTEMS, METHODS AND DEVICES FOR IMPLANTING PROSTHETIC HEART VALVES
Abstract
Embodiments of delivery systems, devices and methods for delivering a prosthetic heart valve device to a heart chamber for expanded implementation are disclosed. More specifically, methods, systems and devices are disclosed for delivering a self-expanding prosthetic mitral valve device to the left atrium, with no engagement of the left ventricle, the native mitral valve leaflets or the annular tissue downstream of the upper annular surface during delivery, and in some embodiments with no engagement of the ventricle, mitral valve leaflets and/or annular tissue located downstream of the upper annular surface by the delivered, positioned and expanded prosthetic mitral valve device.
| Inventors: | Chambers; Jeffrey W.; (Maple Grove, MN) ; Brucker; Gregory G.; (Maple Grove, MN) ; Higgins; Joseph P.; (Minnetonka, MN) ; Kumar; Saravana B.; (Minnetonka, MN) ; Diedering; Jason S.; (Minneapolis, MN) ; Kabarowski; Karl A.; (Maple Grove, MN) ; Thatcher; Robert J.; (Blaine, MN) ; Flaherty; James E.; (Maple Grove, MN) ; Stone; Jeffrey R.; (Minnetonka, MN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004838403 | ||||||||||
| Appl. No.: | 16/877053 | ||||||||||
| Filed: | May 18, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15874376 | Jan 18, 2018 | 10653523 | ||
| 16877053 | ||||
| 62448036 | Jan 19, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61F 2/2439 20130101; A61F 2/2436 20130101; A61F 2/2418 20130101; A61F 2/243 20130101 |
| International Class: | A61F 2/24 20060101 A61F002/24 |
Claims
1. A method of delivering a self-expanding prosthetic mitral valve device comprising prosthetic mitral valve leaflets to an implantation site within a left atrium of a patient's heart, comprising: accessing the left atrium with a delivery catheter having a proximal end, a distal end and a lumen therethrough, wherein the delivery catheter comprises a curved distal portion adapted to facilitate positioning of the self-expanding prosthetic mitral valve device within the left atrium; loading the self-expanding prosthetic mitral valve device in a collapsed configuration into the lumen of the delivery catheter at the proximal end thereof, wherein the collapsed configuration of the prosthetic mitral valve device comprises a prosthetic mitral valve attached thereto; delivering the collapsed prosthetic valve out of the distal end of the delivery catheter and into the left atrium at a location proximate the implantation site; and allowing the delivered self-expanding prosthetic mitral valve device to expand within the left atrium at the implantation site, wherein at least a portion of the expanded device engages at least a portion of the upper annular surface within the left atrium.
2. The method of claim 1, further comprising: providing a pull wire that is attached to the distal end of the delivery catheter and extends proximally out of the proximal end of the delivery catheter, wherein the curved distal portion of the delivery catheter is adapted to be curved by pulling the pull wire proximally during at least the delivery of the prosthetic mitral valve device; and pulling the pull wire proximally during at least the delivery of the prosthetic mitral valve device to curve the distal portion of the delivery catheter.
3. The method of claim 2, further comprising providing a weakened section around at least a portion of the distal portion of the delivery catheter and proximate to the attachment of the pull wire to the distal end of the delivery catheter.
4. The method of claim 3, wherein the weakened section comprises a series of cuts or serrations that extend partially through the catheter wall of the distal portion of the delivery catheter.
5. The method of claim 3, wherein the weakened section comprises a series of cuts or serrations that extend fully through the catheter wall of the distal portion of the delivery catheter.
6. The method of claim 1, wherein the curved distal portion of the delivery catheter is pre-curved.
7. The method of claim 1, further comprising: providing a plug attached to a side of the distal portion of the collapsed self-expanding prosthetic mitral valve device; aligning the plug with the left atrial appendage; and engaging the left atrial appendage with the plug when the self-expanding mitral valve device is expanded.
8. The method of claim 7, wherein the plug further comprises a flange that engages and seals the atrial wall around the left atrial appendage.
9. The method of claim 1, further comprising: providing a push rod that is connected to the collapsed prosthetic mitral valve device; and pushing the collapsed prosthetic mitral valve device with the push rod through the lumen of the delivery catheter for delivery and expansion within the left atrium.
10. The method of claim 9, further comprising at least partially collapsing the expanded prosthetic mitral valve within the lumen of the delivery catheter by pulling proximally on the push rod.
11. The method of claim 9, further comprising: translating a locating element through the lumen of the catheter; engaging an open frame portion of the prosthetic mitral valve device with the locating element; and engaging the lumen of the left upper pulmonary vein with a distal end of the locating element before delivering the prosthetic mitral valve device into the left atrium.
12. The method of claim 11, wherein the locating element extends through an open frame section of the prosthetic mitral valve device.
13. The method of claim 1, wherein the collapsed self-expanding prosthetic mitral valve device further comprises: a proximal portion; a distal portion, wherein the prosthetic mitral valve leaflets are disposed within the distal portion; and a central portion disposed between the proximal portion and the distal portion, the central portion having a maximum diameter that is smaller than a maximum diameter of the proximal portion and a maximum diameter of the distal portion.
14. The method of claim 13, further comprising: providing a hinge on the central portion; delivering the central portion out of the distal end of the delivery catheter lumen to expose the hinge to the left atrium; and rotating the distal portion toward the upper annular surface of the left atrium; and delivering the proximal portion out of the distal end of the delivery catheter lumen and into the left atrium.
15. The method of claim 1, wherein the collapsed self-expanding prosthetic mitral valve device further comprises a proximal portion and a distal portion, wherein the prosthetic mitral valve leaflets are disposed within the distal portion, the method further comprising; loading an inner sheath into the catheter lumen, the inner sheath having a distal end that engages the collapsed self-expanding prosthetic mitral valve, wherein translation of the inner sheath through the catheter lumen results in translation of the collapsed self-expanding stented valve through the catheter lumen; and wherein the inner sheath comprises a lumen therethrough sized to receive the proximal portion of the collapsed self-expanding prosthetic mitral valve device.
16. The method of claim 14, wherein the distal end of the inner sheath engages the distal portion of the collapsed self-expanding prosthetic mitral valve device, wherein the distal portion is not received within the lumen of the inner sheath.
17. The method of claim 14, wherein the distal portion is delivered, positioned proximate the implantation site and expanded before the proximal portion of the collapsed self-expanding prosthetic mitral valve device is delivered and expanded.
18. The method of claim 1, further comprising: translating an alignment wire disposed through the lumen of the delivery catheter; engaging an open frame portion of the prosthetic mitral valve device with the alignment wire; and engaging the left upper pulmonary vein with the alignment wire before delivering the prosthetic mitral valve device into the left atrium along the alignment wire.
19. The method of claim 1, further comprising at least one capture wire extending through the delivery catheter lumen and engaging the self-expanding prosthetic mitral valve device adapted to restrain the expansion of the device to a partially expanded configuration.
20. The method of claim 19, further comprising the at least one capture wire further adapted to positionally orient the partially expanded device by manipulating the at least one capture wire at the proximal end of the delivery catheter.
21. The method of claim 20, further comprising: positionally orienting the partially expanded device within the left atrium; confirming proper orientation of the partially expanded device; removing the at least one capture wire; and allowing the partially expanded device to fully expand.
22. The method of claim 21, further comprising providing a push/pull rod through the lumen of the delivery catheter adapted to assist in positionally orienting the partially expanded device.
23. The method of claim 1, further comprising a lasso element attached to the prosthetic mitral valve device adapted to be manipulated at the proximal end of the delivery catheter; manipulating the lasso element to positionally orient, prevent full expansion of the prosthetic mitral device and/or allow full expansion of the prosthetic mitral valve device within the left atrium.
24. The method of claim 1, wherein the self-expanding prosthetic mitral valve device further comprises an upper portion formed from a stent comprising at least two sets of opposing subsections, at least one set of the opposing subsections comprising a fabric covering.
25. The method of claim 24, further comprising using the fabric covering to capture blood flow to position the self-expanding prosthetic mitral valve device and expanding the positioned self-expanding prosthetic mitral valve device.
26. The method of claim 1, further comprising ensuring that the left ventricle is not touched at any point by the expanded prosthetic mitral valve device.
27. The method of claim 1, further comprising ensuring that the native mitral valve leaflets are not touched at any point by the expanded prosthetic mitral valve device.
28. The method of claim 1, wherein the left atrium is accessed through the septum between the right and left atria.
29. The method of claim 1, further comprising: translating an alignment wire through the lumen of the catheter and engaging an open frame portion of the prosthetic mitral valve device; and engaging the lumen of the left upper pulmonary vein with a distal end of the alignment wire before delivering the prosthetic mitral valve device into the left atrium along the alignment wire.
30. The method of claim 1, further comprising an alignment guide wire disposed through the lumen of the catheter and engaging the left atrial appendage.
31. A system suitable for use in a method of delivering a self-expanding prosthetic mitral valve device to an implantation site within a left atrium of a patient's heart, the system comprising: the self-expanding prosthetic mitral valve device, comprising prosthetic mitral valve leaflets; and a delivery catheter having a proximal end, a distal end and a lumen therethrough, wherein the delivery catheter comprises a curved distal portion adapted to facilitate positioning of the self-expanding prosthetic mitral valve device within the left atrium, wherein the method comprises: accessing the left atrium with the delivery catheter; loading the self-expanding prosthetic mitral valve device in a collapsed configuration into the lumen of the delivery catheter at the proximal end thereof, wherein the collapsed configuration of the prosthetic mitral valve device comprises a prosthetic mitral valve attached thereto; delivering the collapsed prosthetic valve out of the distal end of the delivery catheter and into the left atrium at a location proximate the implantation site; and allowing the delivered self-expanding prosthetic mitral valve device to expand within the left atrium at the implantation site, wherein at least a portion of the expanded device engages at least a portion of the upper annular surface within the left atrium.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 15/874,376, filed Jan. 18, 2018 and entitled SYSTEMS, METHODS AND DEVICES FOR DELIVERY SYSTEMS, METHODS AND DEVICES FOR IMPLANTING PROSTHETIC HEART VALVES and also claims the benefit of U.S. Provisional Ser. No. 62/448,036, filed Jan. 19, 2017 and entitled SYSTEMS, METHODS AND DEVICES FOR DELIVERY SYSTEMS, METHODS AND DEVICES FOR IMPLANTING PROSTHETIC HEART VALVES, the entirety of which is hereby incorporated by reference.
STATEMENT REGARDING FEDERALLY SPONSORED RESEARCH OR DEVELOPMENT
[0002] Not Applicable
INCORPORATION BY REFERENCE
[0003] All references, including but not limited to publications, patent applications and patents mentioned in this specification are hereby incorporated by reference to the same extent and with the same effect as if each reference was specifically and individually indicated to be incorporated by reference.
FIELD OF THE INVENTION
[0004] The inventions described herein relate to delivery systems, devices and methods for delivering and/or positioning a cardiac valve.
BACKGROUND OF THE INVENTION
[0005] The human heart comprises four chambers and four heart valves that assist in the forward (antegrade) flow of blood through the heart. The chambers include the left atrium, left ventricle, right atrium and left ventricle. The four heart valves include the mitral valve, the tricuspid valve, the aortic valve and the pulmonary valve.
[0006] The mitral valve is located between the left atrium and left ventricle and helps control the flow of blood from the left atrium to the left ventricle by acting as a one-way valve to prevent backflow into the left atrium. Similarly, the tricuspid valve is located between the right atrium and the right ventricle, while the aortic valve and the pulmonary valve are semilunar valves located in arteries flowing blood away from the heart. The valves are all one-way valves, with leaflets that open to allow forward (antegrade) blood flow. The normally functioning valve leaflets close under the pressure exerted by reverse blood to prevent backflow (retrograde) of the blood into the chamber it just flowed out of.
[0007] Native heart valves may be, or become, dysfunctional for a variety of reasons and/or conditions including but not limited to disease, trauma, congenital malformations, and aging. These types of conditions may cause the valve structure to either fail to properly open (stenotic failure) and/or fail to close properly (regurgitant).
[0008] Mitral valve regurgitation is a specific problem resulting from a dysfunctional mitral valve. Mitral regurgitation results from the mitral valve allowing at least some retrograde blood flow back into the left atrium from the right atrium. This backflow of blood places a burden on the left ventricle with a volume load that may lead to a series of left ventricular compensatory adaptations and adjustments, including remodeling of the ventricular chamber size and shape, that vary considerably during the prolonged clinical course of mitral regurgitation.
[0009] Native heart valves generally, e.g., mitral valves, therefore, may require functional repair and/or assistance, including a partial or complete replacement. Such intervention may take several forms including open heart surgery and open heart implantation of a replacement heart valve. See e.g., U.S. Pat. No. 4,106,129 (Carpentier), for a procedure that is highly invasive, fraught with patient risks, and requiring not only an extended hospitalization but also a highly painful recovery period.
[0010] Less invasive methods and devices for replacing a dysfunctional heart valve are also known and involve percutaneous access and catheter-facilitated delivery of the replacement valve. Most of these solutions involve a replacement heart valve attached to a structural support such as a stent, commonly known in the art, or other form of wire network designed to expand upon release from a delivery catheter. See, e.g., U.S. Pat. No. 3,657,744 (Ersek); U.S. Pat. No. 5,411,552 (Andersen). The self-expansion variants of the supporting stent assist in positioning the valve, and holding the expanded device in position, within the subject heart chamber or vessel. This self-expanded form also presents problems when, as is often the case, the device is not properly positioned in the first positioning attempt and, therefore, must be recaptured and positionally adjusted. This recapturing process in the case of a fully, or even partially, expanded device requires re-collapsing the device to a point that allows the operator to retract the collapsed device back into a delivery sheath or catheter, adjust the inbound position for the device and then re-expand to the proper position by redeploying the positionally adjusted device distally out of the delivery sheath or catheter. Collapsing the already expanded device is difficult because the expanded stent or wire network is generally designed to achieve the expanded state which also resists contractive or collapsing forces.
[0011] Besides the open heart surgical approach discussed above, gaining access to the valve of interest is achieved percutaneously via one of at least the following known access routes: transapical; transfemoral; transatrial; and transseptal delivery techniques.
[0012] Generally, the art is focused on systems and methods that, using one of the above-described known access routes, allow a partial delivery of the collapsed valve device, wherein one end of the device is released from a delivery sheath or catheter and expanded for an initial positioning followed by full release and expansion when proper positioning is achieved. See, e.g., U.S. Pat. No. 8,852,271 (Murray, III); U.S. Pat. No. 8,747,459 (Nguyen); U.S. Pat. No. 8,814,931 (Wang); U.S. Pat. No. 9,402,720 (Richter); U.S. Pat. No. 8,986,372 (Murray, III); and U.S. Pat. No. 9,277,991 (Salahieh); and U.S. Pat. Pub. Nos. 2015/0272731 (Racchini); and 2016/0235531 (Ciobanu).
[0013] However, known delivery systems, devices and methods still suffer from significant flaws in delivery methodology including, inter alia, positioning and recapture capability and efficiency.
[0014] In addition, known "replacement" heart valves are intended for full replacement of the native heart valve. Therefore, these replacement heart valves physically engage the annular throat and/or valve leaflets, thereby eliminating all remaining functionality of the native valve and making the patient completely reliant on the replacement valve. Generally speaking, it is a preferred solution that maintains and/or retains the native function of a heart valve, thus supplementation of the valve is preferred rather than full replacement. Obviously, there will be cases when native valve has either lost virtually complete functionality before the interventional implantation procedure, or the native valve continues to lose functionality after the implantation procedure. The preferred solution is delivery and implantation of a valve device that will function both as a supplementary functional valve as well as be fully capable of replacing the native function of a valve that has lost most or all of its functionality. However, the inventive solutions described infra will apply generally to all types and forms of heart valve devices, unless otherwise specified.
[0015] Finally, known solutions for, e.g., the mitral valve replacement systems, devices and methods require 2-chamber solutions, i.e., there is involvement and engagement of the implanted replacement valve device in the left atrium and the left ventricle. Generally, these solutions include a radially expanding stent in the left atrium, with anchoring or tethering (disposed downward through the annular through) connected from the stent device down through the annular throat, with the sub-annular surface within the left ventricle, the left ventricular chordae tendineae and even into the left ventricle wall surface(s).
[0016] Such 2-chamber solutions are unnecessary bulky and therefore more difficult to deliver and to position/recapture/reposition from a strictly structural perspective. Further, the 2-chamber solutions present difficulties in terms of making the ventricular anchoring and/or tethering connections required to hold position. Moreover, these solutions interfere with the native valve functionality as described above because the device portions that are disposed within the left ventricle must be routed through the annulus, annular throat and native mitral valve, thereby disrupting any remaining coaptation capability of the native leaflets. In addition, the 2-chamber solutions generally require an invasive anchoring of some of the native tissue, resulting in unnecessary trauma and potential complication.
[0017] It will be further recognized that the 2-chamber mitral valve solutions require sub-annular and/or ventricular engagement with anchors, tethers and the like precisely because the atrial portion of the device fails to adequately anchor itself to the atrial chamber and/or upper portion of the annulus. Again, the inventive solutions described herein are readily applicable to single or 2-chamber solutions, unless otherwise indicated.
[0018] Various embodiments of the several inventions disclosed herein address these, inter alia, issues.
BRIEF DESCRIPTION OF THE SEVERAL VIEWS OF THE DRAWINGS
[0019] FIG. 1 illustrates a side cutaway view of one embodiment of the present invention.
[0020] FIG. 2A illustrates a side view of one embodiment of the present invention.
[0021] FIG. 2B illustrates a side cutaway view of one embodiment of the present invention.
[0022] FIG. 3A illustrates a side cutaway view of one embodiment of the present invention.
[0023] FIG. 3B illustrates a side cutaway view of one embodiment of the present invention.
[0024] FIG. 4 illustrates a side cutaway view of one embodiment of the present invention.
[0025] FIG. 5A illustrates a side cutaway view of one embodiment of the present invention.
[0026] FIG. 5B illustrates a side cutaway view of one embodiment of the present invention.
[0027] FIG. 6A illustrates a side cutaway view of one embodiment of the present invention.
[0028] FIG. 6B illustrates a side cutaway view of one embodiment of the present invention.
[0029] FIG. 6C illustrates a side cutaway view of one embodiment of the present invention.
[0030] FIG. 7A illustrates a side cutaway view of one embodiment of the present invention.
[0031] FIG. 7B illustrates a side cutaway view of one embodiment of the present invention.
[0032] FIG. 8A illustrates a top view of one embodiment of the present invention.
[0033] FIG. 8B illustrates a side and partially exploded view of one embodiment of the present invention.
[0034] FIG. 9A illustrates a side cutaway view of one embodiment of the present invention.
[0035] FIG. 9B illustrates a side cutaway view of one embodiment of the present invention.
[0036] FIG. 9C illustrates a side cutaway view of one embodiment of the present invention.
[0037] FIG. 9D illustrates a side cutaway view of one embodiment of the present invention.
[0038] FIG. 10A illustrates a side cutaway view of one embodiment of the present invention.
[0039] FIG. 10B illustrates a side cutaway view of one embodiment of the present invention.
[0040] FIG. 10C illustrates a side cutaway view of one embodiment of the present invention.
[0041] FIG. 11 illustrates a side cutaway view of one embodiment of the present invention.
[0042] FIG. 12 illustrates a side view of one embodiment of the present invention.
DETAILED DESCRIPTION OF THE INVENTION
[0043] Various embodiments of the present invention are disclosed in the Figures for providing percutaneous access to the valve of interest via one of at least the following known access routes: transapical; transfemoral; transatrial; and transseptal delivery techniques. Each of these access routes may be used for the embodiments disclosed herein.
[0044] Thus, FIG. 1 illustrates one embodiment of a prosthetic valve device 100 with a 2-part frame in collapsed configuration. The distal portion 102 of the collapsed device comprises the valve with prosthetic leaflets with a portion of the supporting frame, and is longitudinally translatably and rotatably confined within the lumen of an outer sheath 104 having a first outer diameter D. The proximal portion 106 of the collapsed device 100 comprises the remaining supporting frame which is in operative connection with the distal portion 102 of collapsed device 100 and in longitudinally translatably and rotatably confined within the lumen of an inner sheath 108 that is at least longitudinally translatable relative to the outer sheath 104 and wherein the outer sheath 104 is at least longitudinally translatable relative to the inner sheath 108. The inner and/or outer sheath 108, 104 may also be rotationally translatable relative to the other sheath. The inner sheath 108 is disposed within the lumen of the outer sheath 104 and, therefore, the inner sheath 108 comprises a second outer diameter D' that is smaller than the outer sheath's outer diameter D.
[0045] The preferred configuration of the device of FIG. 1 comprises the collapsed device 100 consisting of one unit with a proximal and distal portion 106,102 as shown. The outer sheath 104 may be retracted to release expose firstly the distal portion 102 from the distal end 110 of the outer sheath 104 for initial expansion and positioning in the subject chamber of the heart. Alternatively, the distal portion 102 of the device 100 may be pushed distally to be released from the distal end 110 of the outer sheath 104 in response to distal translation of the inner sheath 108, e.g., or to a push rod pushing against the proximal portion 106 of the device 100. In the case of a push rod, the proximal portion 106 will eventually be pushed distally out of the smaller lumen of the inner sheath 106 and into the larger lumen of the outer sheath 104 where an interim secondary expansion of the proximal portion 106 occurs, followed by the secondary positioning expansion when the proximal portion 106 is eventually released from the distal end 110 of the outer sheath 104.
[0046] If expanded within the left atrium in connection with a prosthetic mitral valve, the lower portion of the distal portion 102 may be positioned against the upper surface of the annulus within the left atrium.
[0047] In this configuration, if the distal portion 102 is properly positioned and released/expanded, then the secondary release and expansion of the proximal portion 106 of the device 100 may be initiated and achieved according to the alternative methods described above in connection with the initial release and expansion of the distal portion 102. The skilled artisan will recognize that once the initial positioning expansion of the distal portion 102 is accomplished, then the secondary positioning expansion of the proximal portion 106 will also be properly located and positioned.
[0048] The configuration of FIG. 1, in its various embodiments, enables delivery of a device 100 comprising a frame that may be slightly oversized for the chamber, e.g., atrial, dimensions through the two-step frame positioning expansion method. Some frames in collapsed form may be as much as 2x in longitudinal length than any chamber, e.g., atrial, dimension. Thus, the staged positioning expansion method is necessary for delivery.
[0049] Turning now to FIGS. 2A and 2B a prosthetic valve device 200 comprising a supporting stent frame, with prosthetic valve attached and/or supported therein, is provided wherein the design comprises two portions (distal 202 and proximal 206, wherein the prosthetic valve with leaflets 205 is held/supported within the distal portion 202) with expanded diameters that are connected by a central portion 203 that has a smaller diameter than the expanded diameters of the two portions 202, 206. As illustrated, the two portions 202, 206 comprise an undeformed and fully expanded spherical shape, though other shapes may be used as the skilled artisan will readily understand. Certain embodiments may comprise at least one of the proximal and distal portions 206, 202 having an expanded sizing that is slightly larger than the subject chamber's dimensions, e.g., the left atrial dimensions to allow expansion anchoring. Moreover, the aspect ratio of each of the two portions 206, 202 may vary.
[0050] As shown, the collapsed stent with valve is held within the lumen of a delivery sheath 204, with a distal portion that holds or supports the device 200 therein being released from the end 210 of the delivery sheath 204 with subsequent positioning expansion of same within the subject chamber, e.g., the left atrium. When proper positioning is confirmed, the remaining central portion 203 (if not previously released along with the distal portion 202) and/or the proximal portion 206 may then be released and positionally expanded by methods described in connection with FIG. 1, including use of an inner sheath as described above and/or a push rod to translate the device 200 out of the distal end 210 of the outer delivery sheath 204. As with FIG. 1, this embodiment comprises a two-step or staged delivery mechanism. Both FIG. 1 and FIG. 2A/2B sets of embodiments may comprise a coating or covering on the distal portion 202 while the proximal portion 206 may comprise an open frame formed from, e.g., stent cells. In the case of FIG. 2A/2B, the central portion 202 may also comprise and open cell construction and uncovered. The dashed lines of FIG. 2B show an alternate embodiment wherein the expanded delivered portion 202 comprises a hinge point 212 to assist in orienting the prosthetic valve and leaflets within distal portion 202 downward toward the native valve.
[0051] FIGS. 3A, 3B, 3C, 5A and 5B provide further disclosure of exemplary prosthetic valve devices with support means, e.g., stented and associated exemplary delivery methods. Thus FIG. 3A shows the collapsed device 300 in the lumen of a delivery sheath 304 in operative communication with a push/pull rod 308 actuated by the device operator that is capable of distally translating the collapsed device 300 as in FIG. 3A out of the distal end 310 of the delivery sheath 304 for positioning expansion and, conversely, pulling the expanded device 300 as shown in FIG. 3B back into the distal end 310 of the delivery sheath 304 if necessary. The push/pull rod 308 may also allow in certain embodiments the rotation of the collapsed device 300 within the lumen of the delivery sheath 310 to aid in positioning prior to release and expansion. Further, the operative communication of the push/pull rod 308 with the collapsed device 300 may comprise a screw or clip release mechanism 311 connected with the most proximal portion of the collapsed device 300. The base or lower portion of the device 300 may be covered with tissue or other biocompatible material while the upper portion of the device may comprise an open cell construction.
[0052] Generally, the collapsed device 300 is loaded and positioned within the delivery sheath 304 with the valve portion 305 oriented in a downward position as shown. This allows the collapsed valve device 305 to be pushed out of the delivery sheath 304 in a sideways orientation as illustrated and enables the expanding valve device 300 upon release from the delivery sheath to be properly oriented to the native valve and subject chamber, e.g., mitral valve and left atrium.
[0053] In certain cases, an alignment wire 315 may be translated from the delivery sheath 304 into a pulmonary vein, e.g., the left upper pulmonary vein PV to assist in positioning and delivery of the device 300.
[0054] FIG. 4 illustrates a sideways delivery of device 300 during expansion and just after delivery from the outer end 310 of the delivery sheath 304, wherein the delivered device is oriented substantially vertically and aligned for positioning over the native valve.
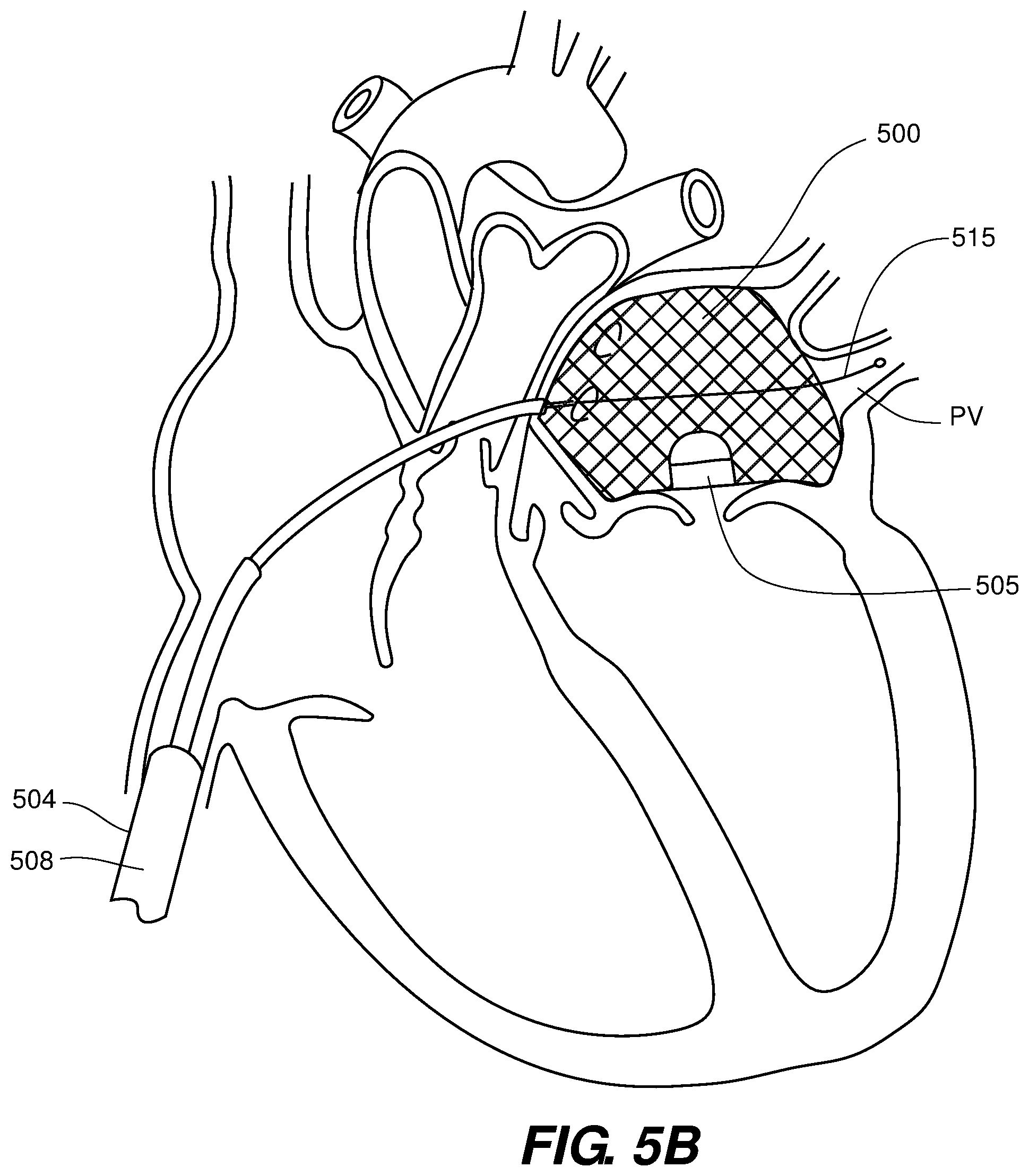
[0055] Thus, as shown best in FIGS. 5A and 5B, the prosthetic valve device 500 may be delivered sideways (with the valve portion 505 oriented on the bottom as shown), asymmetrically and may comprise a locating element 515 in operative connection with the prosthetic valve device 500 and that extends from the delivery sheath 503 with at least a distal end of the locating element 515 or push tube disposed within a pulmonary vein PV, e.g., the left upper pulmonary vein as shown. This system provides a self-centering system that may expand upon releasing/translating the collapsed prosthetic valve device 500 from the distal end 510 of the delivery sheath 504. As shown a push tube 508 and associated connector 511 may be used to assist in manipulating the orientation of the prosthetic valve device 500 once it is delivered from the distal end 510 of the delivery sheath 504.
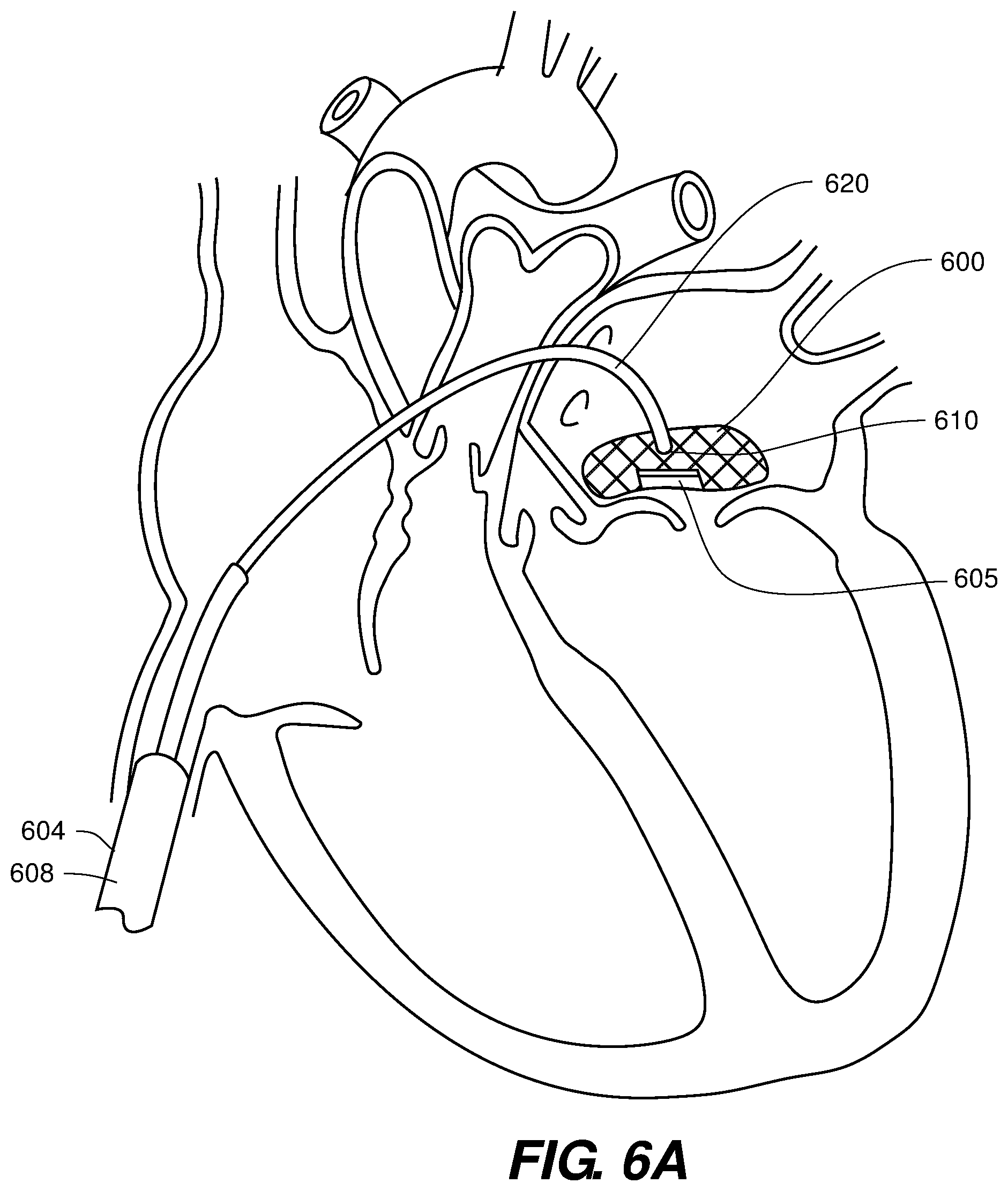
[0056] Turning now to FIGS. 6A-6C, a the prosthetic valve device 600 is delivered using a delivery catheter or sheath 604 comprises either a pre-curved distal portion 620 or a distal portion adapted to be able to be curved 620 to present a substantially straight distal section within the atrium. FIG. 6A provides a pre-curved embodiment, curved to enable loading of a prosthetic valve device 600 in a configuration that places the valved bottom portion 605 in the proper location on release from the distal end 610 of the pre-curved distal portion 620 of the delivery catheter or sheath, more specifically from the straightened distal section distal to the pre-curved distal portion 620. Thus, as shown, the valve supported portion 605 of the collapsed and expandable frame/stent is distal-most within the lumen of the delivery catheter/sheath 604. The pre-curved portion 620 enables easy orienting of the valved portion 605 with an exemplary mitral valve and/or upper surface of the annulus thereof.
[0057] Thus, the delivery system with a curved distal portion as in FIG. 6A enables the prosthetic valve device 600 to be positioned over the annulus and native valve leaflets. When positioned over the annulus and native valve leaflets, the curved delivery catheter or sheath 604 may be withdrawn proximally, alone or in combination with a push rod 608 or similar device on the proximal side of the prosthetic valve device 600, to release and deliver the prosthetic valve device 600 into the left atrium and expand the delivered device 600. It is noteworthy that the curved delivery catheter or sheath 604 comprises in some embodiments a straight distal end within the left atrium and located distal to the curved section 620 wherein the compressed prosthetic valve device 600 is translated and manipulated around the curved portion 620 of the curved delivery catheter or sheath 604. The compressed prosthetic valve device 600 may be assisted in translating around the curved portion 620 of the curved delivery catheter or sheath by including a suture attachment to the distal end of the implant, a pull wire attached to the distal end of the implant extending to the proximal end of the delivery catheter or sheath, or by taking advantage of the natural flexion point in the arrangement of FIG. 1 between the proximal and the distal portions of the prosthetic valve device and/or by a hinging point as in FIG. 2.
[0058] FIGS. 6B and 6C comprise an alternative approach to creating the curved portion 620 by enabling curving of the distal portion of the delivery sheath or catheter 604 by providing a series of cuts or serrations 609 along a bottom portion surface of the sheath or catheter, resulting in a weak region susceptible to bending. As shown a pull wire 625 is attached to the distal end 610 of the catheter 604 along this bottom weak cut or serrated region and is disposed through the catheter/sheath lumen to an operator who may pull the wire with force F proximally to achieve the desired curvature prior to release of the collapsed prosthetic valve structure 600 which is oriented in collapsed form as in FIG. 6A and released for positioning expansion virtually directly on the subject valve or upper annular surface. The cuts 609 may extend through the catheter/sheath wall completely or may only be sections that have a catheter/sheath wall that is thinner than the rest of the catheter/sheath walls. The cuts 609 shown are uniform and generally square, though any depth, shape and uniform or non-uniform spacing of same may be used to achieve the weakened region.
[0059] FIGS. 7A and 7B illustrate delivery systems for an exemplary prosthetic valve, e.g., mitral valve replacement or supplement, to a heart chamber, e.g., the left atrium using a delivery catheter or sheath 704 as shown in FIG. 7A with transseptal access and in combination with an additional guidance tool 728 used to help guide the expanding valved device (not shown) as it is released from the distal end 710 of the catheter or sheath 704 using methods or devices described herein. The additional guidance tool 728 may be disposed within the upper pulmonary vein PV, for example. The guidance tool 728 may be hingedly or rotatingly attached to the catheter or sheath 704 enabling the tool 728 to rotate into position. A pull wire similar to that shown in FIGS. 6B and 6C may be used to connect to and manipulate the tool 728 into position.
[0060] FIG. 7B illustrates two delivery systems, a first delivery system 800 for alignment and deployment and a second delivery system 850 for recapture and repositioning if needed. One of the delivery systems, either the first 800 or the second 850, may access the subject heart chamber via a transfemoral access method while the other delivery system may access the subject heart chamber via another transvenous access method. The first delivery system 800 may thus comprise a delivery catheter or sheath 804 as described elsewhere herein while the second delivery system may comprise a recapture and repositioning catheter or sheath 854, similar in structure to the delivery catheter/sheath 804.
[0061] FIGS. 8A and 8B illustrate embodiments designed to facilitate accurate positioning of a prosthetic heart valve within a chamber, e.g., the left atrium, including but not limited to self-centering and fluoroscopy techniques. In this embodiment of a prosthetic stented valve device 900, with the prosthetic valve and leaflets 905 supported proximate the bottom portion of the valve device, the upper portion 909 of the device 900 may be divided into subsections as shown from the top in FIG. 8A. The illustrated case provides 4 subsections, though other numbers of subsections may certainly be useful and are within the scope of the present invention. As shown, opposing subsections are either open cell or open wire construction 907 or are comprised of a fabric in the form of a type of sail 908. Upon delivery of this device 900 to a subject heart chamber, the fabric sails 908 will catch and use the natural force of blood flow to maneuver the device frame 900 into proper positioning with subsequent release and expansion when positioning is confirmed.
[0062] FIG. 8B is a related concept, but also includes an annulus spacer 919 that may be delivered first via a delivery catheter/sheath as described previously herein and in certain embodiments, the spacer may be directed into position with a guidewire positioned within the lumen of the delivery catheter/sheath and further moved out of the distal end of the delivery catheter/sheath and either proximate to (on the proximal side) of the chamber upper annular surface or may be disposed at least partway within the annular throat. Once released from the lumen and distal end of the delivery catheter/sheath, the annular spacer 919 may expand from a delivered collapsed form and positioned on the upper annular surface which may space the prosthetic valve and leaflets 905 from the upper annular surface. Subsequently, the prosthetic valve device, as described herein and which may, or may not, comprise sails 908 as in FIG. 7A, is delivered from the delivery catheter/sheath and positionally expanded to connect with the previously positioned spacer 919.
[0063] We next describe positional orienting delivery structures in FIGS. 9A-9D. Generally, each of these prosthetic valved devices are designed for use in the left atrium and make use of the left atrial appendage (LAA) as an orienting mechanism. FIG. 9A therefore comprises an LAA plug 1006 disposed on a side surface of the collapsed device 100 within the delivery sheath 1004 lumen and, in FIG. 9B, the LAA plug 1006 is positioned at least partially within the LAA. Once the LAA is engaged by the LAA plug 1006, the operator has confirmation that the valved prosthetic device 1000 is in correct position. This device may be used in combination with any of the previously described devices and methods, including but not limited to the staged 2-step delivery devices and methods, whereby an initial positioning expansion would result in orienting the LAA plug into the LAA, then the secondary positioning expansion of the rest of the device initiated by release from the distal end of a delivery sheath.
[0064] An additional benefit of certain embodiments of FIGS. 9A and 9B may be to employ the LAA plug 1006 as a device to prevent clotting within the LAA, wherein the LAA plug 1006 fills the LAA entirely and/or an outer flange 1008 covers the LAA opening entirely to prevent any blood clots from forming and/or moving out of the heart to potentially cause a stroke.
[0065] FIG. 9D shows a slightly different mechanism whereby a guidewire 1020 is disposed through a delivery sheath 1004 and into the LAA to provide orienting guidance for the positioning expansion (1 step or staged) of the collapsed prosthetic valved device (not shown) within the lumen of delivery sheath 1004. The sheath 1004 may be pulled back to deploy/release the valved device (not shown) from the distal end of the sheath 1004 for positioning expansion or a push rod may be used to push the valved device out of the sheath's distal end as previously described. In these cases, the guidewire 1020 positioned within the LAA provides a key orienting guidance parameter so that the operator knows positioning will be proper on expansion. The guidewire 1020 may comprise an atraumatic tip to prevent damaging the tissue of the LAA.
[0066] FIG. 9C illustrates another alignment/orienting system wherein the delivery catheter/sheath 1004 is introduced via a pulmonary vein PV, e.g., the upper pulmonary vein, into the left atrium and a guidewire 1020 disposed through the lumen of the delivery catheter/sheath 1004 and into, or proximate, the annulus, i.e., the annular throat, as a guide for the to-be-delivered valved device (not shown but compressed and self-expanding as previously described). When the sheath 1004 is pulled back, or a push rod is used to push the collapsed valved device out of the distal end of the delivery catheter or sheath 1004, the expanding valved device may slide down over the pre-positioned guidewire 1020 to a proper position when fully expanded.
[0067] FIG. 10A illustrates a partially expanded stented valved device 1100 released from the delivery catheter sheath. At least one capture wire 1030 (shown radially wrapped around the device 1100, but may take other wrapping positions) is shown and which restrains the expandable device 1100 from fully expanding until properly positioned within the subject heart chamber, e.g., the left atrium. When proper position is confirmed, the capture wire 1030 may be removed by cutting and withdrawal distally through the delivery sheath 1004 lumen or by disconnecting a connector pin or latch 1032 or equivalent to enable full expansion of the device 1100 at the proper positional location. FIG. 10B is similar with an alignment wire 1130 that assists in positional orientation as it feeds out of the distal end of the delivery catheter/sheath 1104 while in connection with the partially expanding sheath at 2 or 3 or more stabilization points 1034 until proper position is confirmed. The stabilization point 1134 connections may hold the partially expanded device in that state until proper position is confirmed, then the connections may be removed, either by cutting (as in the case of a releasable suture) or by disconnecting a connector pin or latch, to enable the full expansion at the proper positional location or by provision of a secondary over the wire cutter introduced via the delivery catheter/sheath 1104 to clip alignment wire 1130.
[0068] FIG. 10C provides an alternative prosthetic heart valve device shown in a positionally expanded position after release from the distal end of the delivery catheter/sheath 1104 and comprising at least one attachment point 1032 located within the stented heart valve device as well as two or more pull/push wires 1130 with a first end connected to the at least one attachment point 1032 and a second end attached to points 1033 around the stent frame. This arrangement may function in several different ways to facilitate recapture, repositioning and/or redeployment.
[0069] First, one embodiment may comprise the two or more pull/push wires 1130 of a length that is slightly smaller than the chamber, e.g., left atriam, dimensions to ensure proper positioning. Once position is confirmed as proper, the pull/push wires 1130 may be released by, e.g., a secondary over the wire cutter or other means to disengage the pull/push wire connection between the at least one attachment point 1032 and the two or more pull/push wires 1130, thereby enabling the full expansion of the properly positioned frame within the chamber. As with other embodiments described herein, the fully expanded frame may be slightly larger than at least one dimension to facilitate anchoring.
[0070] Another embodiment may further comprise a push rod disposed translationally within the delivery catheter/sheath 1104 lumen and that also provides a distally extending releasable connector attached to the at least one attachment point 1032 within the stented heart valve frame for disengaging the attachment between the at least one attachment point 1032 and the two or more push/pull wires 1130 once proper positioning is confirmed. This embodiment provides the further benefit of using a distally extending releasable connector tool to pull proximally on the at least one attachment point wherein the attachment point and the push/pull wires are connected to points on the stent frame that, when proximal force is applied to the attachment point, cause the stent frame to collapse slightly or fully, to enable repositioning. Once repositioned, distal force is applied to the releasable connector tool to fully expand the prosthetic valve frame.
[0071] Yet another embodiment may comprise the attachment point 1132, push/pull wires 1130, and/or the connection of the push/pull wires to the stent frame to be formed of a material that dissolves over a short time period.
[0072] FIG. 11 illustrates a prosthetic valve device 1200 comprising a ball and socket relationship between the support frame (socket or partial socket), with the prosthetic valve and leaflets 1253 disposed therein (ball or partial ball). In this embodiment, the outer frame 1250 is, as illustrated, a partial sphere having a radiusing and a central point 1252, with the central point 1252 disposed generally around the native valve and annulus. The outer frame 1250 may comprise a radially extending flange 1254 to connect and seal with the upper annular surface and may further comprise wall elements 1256 extending upward from at least portions of the radially extending flange 1252 to connect with and seal against the chamber, e.g., left atrial, walls. The radially extending flange 1252 may comprise an expandable stent-like construction to provide radial expansive force to assist in anchoring the device 1200. Alternative constructions may comprise any of the prosthetic stented valve frames described herein, e.g. and without limitation, an upper open expandable frame with a lower expandable frame covered with tissue.
[0073] The prosthetic valve further comprises an inner partial sphere 1253 with a radiusing that matches, or is complementary with, the radiusing of the outer frame's partial sphere 1250, but with a smaller radius that the outer frame 1250 as the inner partial sphere 1253 resides within the outer frame's partial sphere 1250. The prosthetic leaflets are supported within the inner partial sphere 1253. The inner partial sphere 1253 may comprise a friction fit with the outer frame's partial sphere 1250 so that some movement is possible in all dimensions, including rotational, without losing the proper valve position relative to the native valve and/or annulus. Alternatives may allow a looser friction fit so that the inner partial sphere essentially floats within the outer frame's partial sphere, thereby allowing a fuller range of motion than a tighter friction fit.
[0074] FIG. 12 illustrates an implant frame with prosthetic valve device 1300 attached thereto connected via a connector element 1302 to a lasso structure 1304 that is, in turn, operatively connected with a manipulation wire 1306, that may comprise a single wire or two wires, that extends proximally to the operator who is then able to manipulate the lasso 1304 and connector element 1302. The lasso structure 1304 may comprise two distal wires, W1, W2, or more than two distal wires, in operative connection with the connector element 1302. If the manipulation wire 1306 comprises two wires W1, W2, then a first of the two wires may be connected with wire 1 and the second of the two wires may be connected with wire 1. Wires W1, W2 may be disconnected from the connector element 1302 by the operator's pulling of one, or both, of the manipulation wire(s) W1, W2. The lasso structure 1304 may, as shown, be expandable to a diameter that is larger than the inner diameter of the catheter's 1305 lumen and is disposed through the implant frame structure with the connector element 1302 in operative connection with the lasso 1304 and the device frame 1300 generally in the middle of the implant structure. This configuration allows the operator to steer the device 1300 with the lasso structure 1304 during deployment and also allows retrieval back into the catheter's 1305 lumen if necessary. The connector element 1302 may be configured together with the device's 1300 frame structure to enable collapsing of the device's 1300 frame structure to allow pullback of the device's 1300 structure into the catheter's 1305 lumen. The connector element 1302 may also be disconnected by the operator from the device's 1300 frame, whereby one, or both, of the wires W1, W2 are disconnected and the lasso structure 1304 retracted proximally through the catheter 1305. In other embodiments, the connector element 1302 may remain attached to the device's 1300 frame structure when the operator disconnects wires W1, W2 from the connector element 1302 and pulls the lasso structure 1304 proximally through the catheter sheath 1305.
[0075] The description of the various inventions, embodiments thereof and applications as set forth herein is illustrative and is not intended to limit the scope of the invention. Features of various embodiments may be combined with other embodiments within the contemplation of these inventions. Variations and modifications of the embodiments disclosed herein are possible, and practical alternatives to and equivalents of the various elements of the embodiments would be understood to those of ordinary skill in the art upon study of this patent document. These and other variations and modifications of the embodiments disclosed herein may be made without departing from the scope and spirit of the inventions.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.