Method Of Enhancing Delivery Of Therapeutic Compounds To The Eye
PAN; Zhuo-Hua ; et al.
U.S. patent application number 16/593302 was filed with the patent office on 2020-08-27 for method of enhancing delivery of therapeutic compounds to the eye. The applicant listed for this patent is Wayne State University. Invention is credited to Elena IVANOVA, Zhuo-Hua PAN.
| Application Number | 20200268647 16/593302 |
| Document ID | / |
| Family ID | 1000004816199 |
| Filed Date | 2020-08-27 |




| United States Patent Application | 20200268647 |
| Kind Code | A1 |
| PAN; Zhuo-Hua ; et al. | August 27, 2020 |
METHOD OF ENHANCING DELIVERY OF THERAPEUTIC COMPOUNDS TO THE EYE
Abstract
The invention provides methods for enhancing the delivery of therapeutic compounds to the eye of a subject by administering plasmin or derivatives thereof and the therapeutic compounds to the eye.
| Inventors: | PAN; Zhuo-Hua; (Troy, MI) ; IVANOVA; Elena; (White Plains, NY) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004816199 | ||||||||||
| Appl. No.: | 16/593302 | ||||||||||
| Filed: | October 4, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15676268 | Aug 14, 2017 | |||
| 16593302 | ||||
| 14777420 | Sep 15, 2015 | 9730888 | ||
| PCT/US2014/026224 | Mar 13, 2014 | |||
| 15676268 | ||||
| 61785015 | Mar 14, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 31/7088 20130101; A61K 36/05 20130101; A61K 38/484 20130101; A61K 9/0048 20130101; A61K 48/0075 20130101; C12Y 304/21007 20130101; A61K 38/36 20130101; A61K 38/16 20130101; A61K 31/713 20130101; A61K 48/0008 20130101 |
| International Class: | A61K 9/00 20060101 A61K009/00; A61K 31/713 20060101 A61K031/713; A61K 38/36 20060101 A61K038/36; A61K 31/7088 20060101 A61K031/7088; A61K 36/05 20060101 A61K036/05; A61K 38/16 20060101 A61K038/16; A61K 38/48 20060101 A61K038/48; A61K 48/00 20060101 A61K048/00 |
Goverment Interests
GOVERNMENT SUPPORT
[0002] This invention was made with government support under EY017130 awarded by the National Institutes of Health. The government has certain rights in the invention.
Claims
1. A method of enhancing the delivery of a therapeutic agent to an eye of a subject comprising administering a plasmin or derivative thereof and the therapeutic agent to the eye.
2. The method of claim 1, wherein the plasmin or derivative thereof is a miniplasmin or a microplasmin (ocriplasmin).
3. The method of claim 1, wherein the therapeutic agent is selected from a nucleic acid, a small molecule, an antibody, or a peptide.
4. The method of claim 3, wherein the nucleic acid is a nucleic acid expression vector, a plasmid, or an siRNA.
5. The method of claim 4, wherein nucleic acid expression vector is a viral vector comprising a transgene.
6. The method of claim 5, wherein the transgene is an opsin.
7. The method of claim 6, wherein the opsin is selected from the group consisting of channelrhodopsin, halorhodopsin, melanopsin, pineal opsin, bacteriorhodopisin, and proteorhodopsin, or a functional variant thereof.
8. The method of claim 7, wherein said transgene is operably linked to a cell-specific promoter.
9. The method of claim 8, wherein the therapeutic agent is encapsulated in a nanoparticle, a polymer, or a liposome.
10. The method of claim 9, wherein the therapeutic agent is selected from the group consisting of ranibizumab antibody FAB (Lucentis), VEGF Trap fusion molecule (VEGF Trap-Eye), macugen pegylated polypeptide (Pegaptanib), and bevacimzumab (Avastin).
11. The method of claim 1, wherein the subject is suffering from an ocular disease or disorder.
12. The method of claim 1, wherein the plasmin or derivative thereof and the therapeutic agent are delivered concurrently or sequentially.
13. The method of claim 1, wherein the therapeutic agent is delivered to a retinal cell.
14. The method of claim 13, wherein the retinal cell is a retinal ganglion cell, a retinal bipolar cell, a retinal horizontal cell, an amacrine cell, a photoreceptor cell, Muller glial cell, or a retinal pigment epithelial cell.
15. The method of claim 1, wherein the administration is to the vitreous of the eye.
16. A method of increasing light sensitivity or improving or restoring vision in a subject comprising administering a plasmin or derivative thereof and a viral vector that encodes an opsin to the vitreous of the eye.
17. The method of claim 16, wherein said opsin is selected from the group consisting of channelrhodopsin, halorhodopsin, melanopsin, pineal opsin, bacteriorhodopisin, and proteorhodopsin, or a functional variant thereof.
18. The method of claim 16, wherein the subject has an ocular disease or disorder.
Description
RELATED APPLICATIONS
[0001] This application is a continuation application of U.S. application Ser. No. 14/777,420 filed on Sep. 15, 2015, which is a 371 of International Application No. PCT/US2014/026224, filed Mar. 13, 2014, which claims priority to, and the benefit of, U.S. Provisional Application No. 61/785,015, filed on Mar. 14, 2013; the contents of each of which are hereby incorporated by reference in its entirety.
INCORPORATION OF SEQUENCE LISTING
[0003] The contents of the text file named "RTRO-704/C01US_SeqList.txt," which was created on Aug. 14, 2017 and is 25.7 KB in size, are hereby incorporated by reference in their entirety.
FIELD OF THE INVENTION
[0004] This invention relates generally to methods of enhancing the delivery of therapeutic compounds to the eye.
BACKGROUND OF THE INVENTION
[0005] The eye is a complex optical system that detects light, converts the light to a set of electrical signals, and transmits the signals to the brain, ultimately generating a representation of our world. Ocular diseases and disorders can cause diminished visual acuity, diminished light sensitivity, and blindness.
[0006] Delivery of therapeutic compounds to specific ocular tissues affected by an ocular disease or disorder, such as the retina, is a challenge. Current methods, such as intravitreal injection or implanted drug delivery devices, are still limited in the efficacy of delivery. Specifically, the therapeutic agents are often localized only to the immediate areas surrounding the delivery site, and fail to permeate or diffuse beyond intervening ocular structures or throughout the targeted ocular tissue, thereby severely limiting the efficacy of such therapeutics. Thus, there exists a long-felt need for methods to enhance the delivery of therapeutic compounds to the eye.
SUMMARY OF THE INVENTION
[0007] The invention provides a solution for the long-felt need for methods to enhance or improve the delivery of therapeutic compounds to the eye.
[0008] The present invention features a method of enhancing the delivery of a therapeutic agent to an eye of a subject by administering a plasmin or derivative thereof and the therapeutic agent to the eye. The present invention also features the use of a composition comprising a plasmin or derivative thereof for delivery to the eye of a subject for enhancing the delivery of a therapeutic agent.
[0009] In one aspect, the plasmin or derivative thereof is a miniplasmin or a microplasmin (Ocriplasmin). The plasmin or derivative thereof encompassed in the present invention includes amino acid sequences SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 4, SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7, SEQ ID NO: 8, SEQ ID NO: 9 or functional variants or fragments thereof.
[0010] In one aspect, the therapeutic agent is selected from a small molecule, a nucleic acid, an antibody, or a peptide. The nucleic acid is a nucleic acid expression vector (i.e., a viral vector), a plasmid, or an siRNA. For example, the viral vector is a AAV viral vector (i.e., recombinant AAV or rAAV) that encodes a transgene. Preferably, the transgene encodes a gene product that increases or restores light sensitivity, increases light detection, increases photosensitivity, increases visual evoked potential, or restores vision to the blind. More preferably, the transgene is an opsin gene. Examples of opsin genes include, but are not limited to, channelrhodopsins (i.e., channelrhodopsin-1, channelrhodopsin-2, Volvox carteri channelrhodopsins 1 or 2), melanopsin, pineal opsin, photopsins, halorhodopsin, bacteriorhodopisin, proteorhodopsin, or any functional variants or fragments thereof.
[0011] Other examples of therapeutic agents include, but are not limited to ranibizumab antibody FAB (Lucentis), VEGF Trap fusion molecule (VEGF Trap-Eye), macugen pegylated polypeptide (Pegaptanib), and bevacimzumab (Avastin). Any of the therapeutic agents used in the present invention may be encapsulated in a nanoparticle, a polymer, or a liposome.
[0012] In one aspect, the plasmin or derivative thereof and the therapeutic agent are delivered concurrently or sequentially.
[0013] The present invention provides a method in which the therapeutic agent is delivered to a retinal cell. The retinal cell is a retinal ganglion cell, a retinal horizontal cell, a retinal bipolar cell, an amacrine cell, a photoreceptor cell, a Muller glial cell, or a retinal pigment epithelial cell.
[0014] In one aspect, the plasmin or derivative thereof and the therapeutic agent is administered to the vitreous of the eye.
[0015] The present invention further provides a method of increasing or restoring light sensitivity in a subject comprising administering a plasmin or derivative thereof and a viral vector that encodes an opsin to the vitreous of the eye. The present invention also provides a method of improving or restoring vision in a subject comprising administering a plasmin or derivative thereof and a viral vector that encodes an opsin to the vitreous of the eye.
[0016] Uses of a composition comprising a plasmin or derivative thereof for treating an ocular disease or disorder in a subject are also provided herein.
[0017] The subject is suffering from an ocular disease or disorder. Preferably, the ocular disease or disorder is associated with photoreceptor degeneration.
[0018] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention pertains. Although methods and materials similar or equivalent to those described herein can be used in the practice of the present invention, suitable methods and materials are described below. All publications, patent applications, patents, and other references mentioned herein are expressly incorporated by reference in their entirety. In cases of conflict, the present specification, including definitions, will control. In addition, the materials, methods, and examples described herein are illustrative only and are not intended to be limiting.
[0019] Other features and advantages of the invention will be apparent from and are encompassed by the following detailed description and claims.
BRIEF DESCRIPTION OF THE FIGURES
[0020] FIG. 1 is a series of representative GFP fluorescence images in retinal vertical sections after intravitreal injection of AAV2 vectors (6.times.10.sup.12 vg/ml), AAV2/2-ChR2-GFP-WPRE-hGHpA, in control (A, B) or co-injection with plasmin (0.025 IU/eye) (C, D). The vectors were co-injected along with plasmin into the vitreous space of adult C56BL/6J mice at age of approximately one month. Transduction efficiency was evaluated one month after virus injection by immunostaining and cell counting.
[0021] FIG. 2 is a series of representative GFP fluorescence and DAPI staining images demonstrating the effects of plasmin on AAV-mediated transduction efficiency and the potential neurotoxicity in retinal ganglion cells. AAV2 vectors (2.times.10.sup.12 vg/ml), AAV2/2-ChR2-GFP-WPRE-hGHpA, was injected in control (A, E), or was co-injected at (B, F) low (L: 0.005 IU), (C, G) middle (M: 0.025 IU), and (D, H) high (H: 0.100 IU plasmin/eye) concentrations. Retinal ganglion cells were stained by DAPI (A-D).
[0022] FIG. 3 is two graphs showing the quantitative assessment of the effects of plasmin on (A) AAV-mediated transduction efficiency and (B) potential neurotoxicity of plasmin in retinal ganglion cells. Co-injection of plasmin at low (L: 0.005 IU), middle (M: 0.025 IU), and high (H: 0.100 IU plasmin/eye) concentrations significantly increased the AAV-mediated transduction efficiency in retinal ganglion cells (A). Co-injection of plasmin did not show any significant neurotoxicity to retinal ganglion cells. The ganglion cell counts were assessed from multiple unit areas of 223 .mu.m.times.167 .mu.m. * p<0.05; ** p<0.005.
[0023] FIG. 4 is a series of representative fluorescence images of mCherry-expressing retinal bipolar cells in retinal whole-mounts. AAV2 vectors (2.times.10.sup.12 vg/ml) with Y444F capsid mutation carrying mCherry under control of a mGluR6 promoter were co-injected along with plasmin of three doses (L: 0.005 IU, M: 0.025 IU, and H: 0.100 IU/eye) into the vitreous space of adult C56BL/6J mice at age of approximately one month. Transduction efficiency was evaluated one month after virus injection by immunostaining and cell counting.
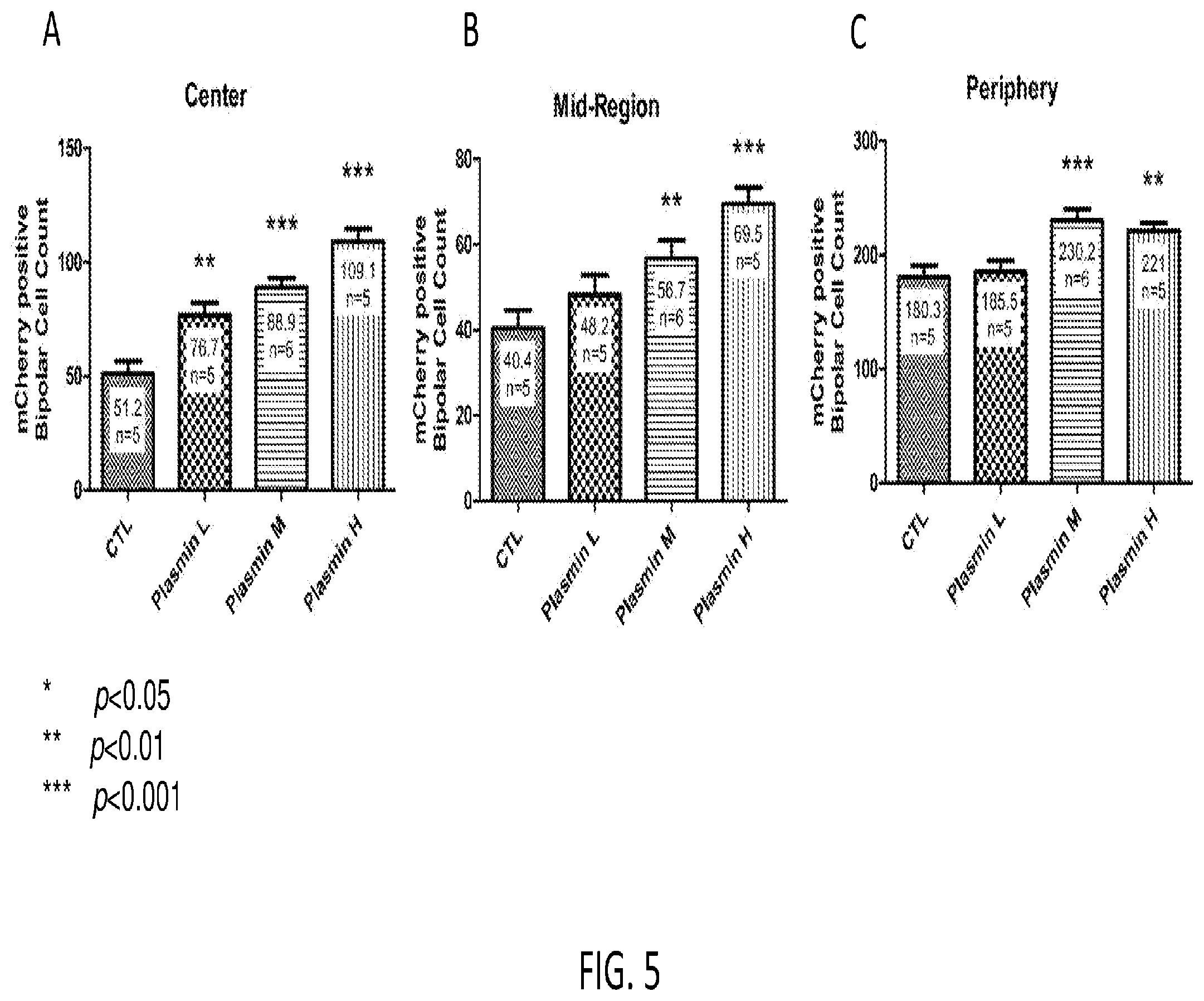
[0024] FIG. 5 is three graphs showing quantitative data for the effects of plasmin on AAV-mediated transduction efficiency in retinal bipolar cells. A) Center; B) Mid-region; and C) Periphery. The counts of mCherry expressing retinal bipolar cell were assessed from multiple unit areas of 223 .mu.m.times.167 .mu.m. * p<0.05; ** p<0.01; ***p<0.001.
DETAILED DESCRIPTION
[0025] The present invention provides methods for enhanced delivery of therapeutic compounds or agents to the eye of a subject by administering a plasmin or derivative thereof and the therapeutic agent to the eye. In some embodiments, the plasmin or derivative thereof and the therapeutic agent may be delivered to the vitreous for enhanced delivery to the retina and retinal cells. The retinal cells include, for example, photoreceptor cells (e.g., rods, cones, and photosensitive retinal ganglion cells), horizontal cells, retinal bipolar cells, amacrine cells, retinal ganglion cells, Muller glial cells, and retinal pigment epithelial cells. In other embodiments, the plasmin or derivative thereof and the therapeutic agent may be delivered to, for example, the posterior segment, the anterior segment, the sclera, the choroid, the conjunctiva, the iris, the lens, or the cornea.
[0026] The retina is a complex tissue in the back of the eye that contains specialized photoreceptor cells called rods and cones. The photoreceptors connect to a network of nerve cells for the local processing of visual information. This information is sent to the brain for decoding into a visual image. The retina is susceptible to a variety of diseases, including age-related macular degeneration (AMD), diabetic retinopathy (DR), retinitis pigmentosa (RP), glaucoma, and other inherited retinal degenerations, uveitis, retinal detachment, and eye cancers (ocular melanoma and retinoblastoma). Each of these can lead to visual loss or complete blindness.
[0027] Delivery of therapeutic compounds to the retina is a challenge, due to the complex structure of the eye. Intravitreal injection and vitreal delivery devices are frequently used to deliver therapeutic compounds to the retina, however the efficiency of delivery is impaired by the inner limiting membrane (ILM) and the multiple layers of cells of the retina.
Plasmin
[0028] Plasmin is a serine protease that is present in the blood that degrades fibrin blood clots and other blood plasma proteins. Specifically, plasmin cleaves fibrin, fibronectin, thrombospondin, laminin, proaccelerin, and Von Willebrand Factor (VWF) into soluble products. Plasmin exhibits preferential cleavage at the carboxyl side of Lysine and Arginine residues with higher selectivity than trypsin.
[0029] Specifically, plasmin originates from a zymogen, or inactive precursor protein, called plasminogen (PLG). The amino acid sequence of plasminogen is known in the art, for example, Genbank Accession Number NP_000292, and listed below:
TABLE-US-00001 (SEQ ID NO: 1) MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAA KCEEDEEFTCRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSE CKTGNGKNYRGTMSKTKNGITCQKWSSTSPHRPRFSPATHPSEGLEENYCR NPDNDPQGPWCYTTDPEKRYDYCDILECEEECMHCSGENYDGKISKTMSGL ECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRELRPWCFTTDPNKRWE LCDIPRCTTPPPSSGPTYQCLKGTGENYGNVAVTVSGHTCQHWSAQTPHTH ANRTPENFPCKNLDENYCRNPDGKRPWCHTTNSQVRWEYCKIPSCDSSPVS TEQLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRH QKTPENYPNAGLTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEAS VVAPPPVVLLPDVETPSEEDCMFGNGKGYRGKRATTVTGTPCQDWAAQEPH RHSIFTPETNPRAGLEKNYCRNPDGDVGGPWCYTTNPRKLYDYCDVPQCAA PSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRTRFGMHFCGGTLIS PEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLEPTRK DIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGL LKEAQLPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVC FEKDKYILQGVTSWGLGCARPNKPGVYVRVSRFVTWIEGVMRNN
[0030] The nucleic acid of plasminogen is known in the art, for example, Genbank Accession Number NM_000301, and as listed below:
TABLE-US-00002 (SEQ ID NO: 2) GAATCATTAACTTAATTTGACTATCTGGTTTGTGGATGCGTTTACTCTCAT GTAAGTCAACAACATCCTGGGATTGGGACCCACTTTCTGGGCACTGCTGGC CAGTCCCAAAATGGAACATAAGGAAGTGGTTCTTCTACTTCTTTTATTTCT GAAATCAGGTCAAGGAGAGCCTCTGGATGACTATGTGAATACCCAGGGGGC TTCACTGTTCAGTGTCACTAAGAAGCAGCTGGGAGCAGGAAGTATAGAAGA ATGTGCAGCAAAATGTGAGGAGGACGAAGAATTCACCTGCAGGGCATTCCA ATATCACAGTAAAGAGCAACAATGTGTGATAATGGCTGAAAACAGGAAGTC CTCCATAATCATTAGGATGAGAGATGTAGTTTTATTTGAAAAGAAAGTGTA TCTCTCAGAGTGCAAGACTGGGAATGGAAAGAACTACAGAGGGACGATGTC CAAAACAAAAAATGGCATCACCTGTCAAAAATGGAGTTCCACTTCTCCCCA CAGACCTAGATTCTCACCTGCTACACACCCCTCAGAGGGACTGGAGGAGAA CTACTGCAGGAATCCAGACAACGATCCGCAGGGGCCCTGGTGCTATACTAC TGATCCAGAAAAGAGATATGACTACTGCGACATTCTTGAGTGTGAAGAGGA ATGTATGCATTGCAGTGGAGAAAACTATGACGGCAAAATTTCCAAGACCAT GTCTGGACTGGAATGCCAGGCCTGGGACTCTCAGAGCCCACACGCTCATGG ATACATTCCTTCCAAATTTCCAAACAAGAACCTGAAGAAGAATTACTGTCG TAACCCCGATAGGGAGCTGCGGCCTTGGTGTTTCACCACCGACCCCAACAA GCGCTGGGAACTTTGTGACATCCCCCGCTGCACAACACCTCCACCATCTTC TGGTCCCACCTACCAGTGTCTGAAGGGAACAGGTGAAAACTATCGCGGGAA TGTGGCTGTTACCGTGTCCGGGCACACCTGTCAGCACTGGAGTGCACAGAC CCCTCACACACATAACAGGACACCAGAAAACTTCCCCTGCAAAAATTTGGA TGAAAACTACTGCCGCAATCCTGACGGAAAAAGGGCCCCATGGTGCCATAC AACCAACAGCCAAGTGCGGTGGGAGTACTGTAAGATACCGTCCTGTGACTC CTCCCCAGTATCCACGGAACAATTGGCTCCCACAGCACCACCTGAGCTAAC CCCTGTGGTCCAGGACTGCTACCATGGTGATGGACAGAGCTACCGAGGCAC ATCCTCCACCACCACCACAGGAAAGAAGTGTCAGTCTTGGTCATCTATGAC ACCACACCGGCACCAGAAGACCCCAGAAAACTACCCAAATGCTGGCCTGAC AATGAACTACTGCAGGAATCCAGATGCCGATAAAGGCCCCTGGTGTTTTAC CACAGACCCCAGCGTCAGGTGGGAGTACTGCAACCTGAAAAAATGCTCAGG AACAGAAGCGAGTGTTGTAGCACCTCCGCCTGTTGTCCTGCTTCCAGATGT AGAGACTCCTTCCGAAGAAGACTGTATGTTTGGGAATGGGAAAGGATACCG AGGCAAGAGGGCGACCACTGTTACTGGGACGCCATGCCAGGACTGGGCTGC CCAGGAGCCCCATAGACACAGCATTTTCACTCCAGAGACAAATCCACGGGC GGGTCTGGAAAAAAATTACTGCCGTAACCCTGATGGTGATGTAGGTGGTCC CTGGTGCTACACGACAAATCCAAGAAAACTTTACGACTACTGTGATGTCCC TCAGTGTGCGGCCCCTTCATTTGATTGTGGGAAGCCTCAAGTGGAGCCGAA GAAATGTCCTGGAAGGGTTGTAGGGGGGTGTGTGGCCCACCCACATTCCTG GCCCTGGCAAGTCAGTCTTAGAACAAGGTTTGGAATGCACTTCTGTGGAGG CACCTTGATATCCCCAGAGTGGGTGTTGACTGCTGCCCACTGCTTGGAGAA AGTCCCCAAGGCCTTCATCCTACAAGGTCATCCTGGGTGCACCCAAGAAGT GAATCTCGAACCGCATGTTCAGGAAATAGAAGTGTCTAGGCTGTTCTTGGA GCCCACACGAAAAGATATTGCCTTGCTAAAGCTAAGCAGTCCTGCCGTCAT CACTGACAAAGTAATCCCAGCTTGTCTGCCATCCCCAAATTATGTGGTCGC CTGACCGGACCGAATGTTTCATCACTGGCTGGGGAGAAACCCAAGGTATTT TGGAGCTGGCCTTCTCAAGGAAGCCCAGCTCCCTGTGATTGAGAATAAAGT GTGCAATCGCTATGAGTTTCTGAATGGAAGAGTCCAATCCACCGAACTCTG TGCTGGGCATTTGGCCGGAGGCACTGACAGTTGCCAGGGTGACAGTGGAGG TCCTCTGGTTTGCTTCGAGAAGGACAAATACATTTTACAAGGAGTCACTTC TTGGGGTCTTGGCTGTGCACGCCCCAATAAGCCTGGTGTCTATGTTCGTGT TTCAAGGTTTGTTACTTGGATTGAGGGAGTGATGAGAAATAATTAATTGGA CGGGAGACAGAGTGACGCACTGACTCACCTAGAGGCTGGAACGTGGGTAGG GATTTAGCATGCTGGAAATAACTGGCAGTAATCAAACGAAGACACTGTCCC CAGCTACCAGCTACGCCAAACCTCGGCATTTTTTGTGTTATTTTCTGACTG CTGGATTCTGTAGTAAGGTGACATAGCTATGACATTTGTTAAAAATAAACT CTGTACTTAACTTTGATTTGAGTAAATTTTGGTTTTGGTCTTCAACATTTT CATGCTCTTTGTTCACCCCACCAATTTTTAAATGGGCAGATGGGGGGATTT AGCTGCTTTTGATAAGGAACAGCTGCACAAAGGACTGAGCAGGCTGCAAGG TCACAGAGGGGAGAGCCAAGAAGTTGTCCACGCATTTACCTCATCAGCTAA GCGAGGGCTTGACATGCATTTTTACTGTCTTTATTCCTGACACTGAGATGA ATTTTTCAAAGCTGCAACATGTATGGGGAGTCATGCAAACCGATTCTGTTA TTGGGAATGAAATCTGTCACCGACTGCTTGACTTGAGCCCAGGGGACACGG AAGCAGAGAGCTGTATATGATGGAGTGAACCGGTCCATGGTGTGTAACACA AGACCAACTGAGAGTCTGAATGTTATTCTGGGGCACACGTGAGTCTAGGAT TGGTGCCAAGAGCATGTAAATGAACAACAAGCAAATATTGAAGGTGGACCA CTTATTTCCCATTGCTAATTGCCTGCCCGGTTTTGAAACAGTCTGCAGTAC AACACGGTCACAGGAGAATGACCTGTGGGAGAGATACATGTTTAGAGGAAG AGAAAGGACAAAGGCACACGTTTTACCATTTAAAATATTGTTACCAAACAA AAATATCCATTCAAAATACAATTTAACAATGCAACAGTCATCTTACAGCAG CAGAAATGCAGAGAAAAGCAAAACTGCAAGTGATGTGAATAAAGGGTGAAT GTAGTCTCAAATCCTCAAA
[0031] The signal peptide sequence of plasminogen is 19 amino acids long. Thus, the plasminogen sequence without the signal peptide encompasses amino acids from positions 20-810 of the plasminogen sequence. The signal peptide sequence is as follows:
TABLE-US-00003 (SEQ ID NO: 3) MEHKEVVLLLLLFLKSGQG
[0032] The plasmin heavy chain A is 561 amino acids, comprising the amino acid sequence provided below:
TABLE-US-00004 (SEQ ID NO: 4) EPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFTCRAFQYHSKE QQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKNG ITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKR YDYCDILECEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSK FPNKNLKKNYCRNPDRELRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQ CLKGTGENYRGNVAVTVSGHTCQHWSAQTPHTHNRTPENFPCKNLDENYCR NPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTEQLAPTAPPELTPVVQD CYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAGLTMNYCR NPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSE EDCMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKN YCRNPDGDVGGPWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGR
[0033] The short form of the plasmin heavy chain A is 483 amino acids. The amino acid sequence of the short form of the plasmin heavy chain A is as follows:
TABLE-US-00005 (SEQ ID NO: 5) VYLSECKTGNGKNYRGTMSKTKNGITCQKWSSTSPHRPRFSPATHPSEGLE ENYCRNPDNDPQGPWCYTTDPEKRYDYCDILECEEECMHCSGENYDGKISK TMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRELRPWCFTTDP NKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWSA QTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSC DSSPVSTEQLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSS MTPHRHQKTPENYPNAGLTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKC SGTEASVVAPPPVVLLPDVETPSEEDCMFGNGKGYRGKRATTVTGTPCQDW AAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGGPWCYTTNPRKLYDYCD VPQCAAPSFDCGKPQVEPKKCPGR
[0034] The amino acid sequence of the activation peptide comprises the following amino acid sequence:
TABLE-US-00006 (SEQ ID NO: 6) EPLDDYVNIQGASLFSVIKKQLGAGSIEECAAKCEEDEEFTCRAFQYHSKE QQCVIMAENRKSSIIIRMRDVVLFEKK
[0035] The plasmin light chain B is 230 amino acids. The amino acid sequence of the plasmin light chain B is as follows:
TABLE-US-00007 (SEQ ID NO: 7) VVGGCVAHPHSWPWQVSLRTRFGMHFCGGTLISPEWVLTAAHCLEKSPRPS SYKVILGAHQEVNLEPHVQEIEVSRLFLEPTRKDIALLKLSSPAVITDKVI PACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQLPVIENKVCNRYE FLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSWGLGC ARPNKPGVYVRVSRFVTWIEGVMRNN
[0036] In some preferred embodiments, the plasmin can be a miniplasmin or a microplasmin or a derivative thereof. Miniplasmin and microplasmin are produced upon the activation of miniplasminogen and microplasminogen by plasminogen activators such as, but not limited to, streptokinase, staphylokinase, tissue-type plasminogen activator or urokinase. Miniplasminogen and microplasminogen are derived from plasminogen, which is a single chain glycoprotein that is an important component of mammalian blood. Human plasminogen is a multi-domain protein of 791 residues (SEQ ID NO:1), composed of an N-terminal pre-activation domain, five homologous kringle domains each of about 80 amino acids, a serine protease catalytic domain and inter-domain connecting sequences. Plasmin or plasminogen activators cleave the peptide bonds between Arg-68 and Met-69, or Lys-77 and Lys-78 or Lys-78 and Val-79 at the N-terminal of human plasminogen, resulting in shorter proenzymes called Lys-plasminogens (for example, proteins consisting of amino acids 69-791 or 78-791 or 79-791). Additional cleavage by the enzyme elastase removes the first four kringle domains producing the proenzyme, miniplasminogen (typically amino acids 442-791). Further cleavage of the fifth kringle yields the proenzyme, microplasminogen (typically amino acids 543-791). The kringles of plasminogen contain lysine-binding sites that mediate specific binding of plasminogen to substrates such as fibrin. The proenzyme forms of plasminogen are activated to their enzymatically active form by the cleavage of the peptide bond between Arg-561 and Val-562 to yield a disulfide bonded double chain form of the corresponding protein. The product of activation of a plasminogen protein is called a plasmin Thus, the product of Lys-plasminogen activation is called Lys-plasmin, while the products of activation of miniplasminogen and microplasminogen, are referred to as miniplasmin and microplasmin, respectively. Like plasmin, miniplasmin and microplasmin possess catalytic activity. An advantage of miniplasmin and microplasmin over plasmin is their smaller size compared to plasmin Thus, both microplasmin and miniplasmin are expected to have faster diffusion rates in the vitreous than plasmin.
[0037] The plasmin of the present invention may comprise any one of the plasminogen-related sequences described herein, for example, any one of SEQ ID NOs: 1 and 3-7, or a functional fragment or variant thereof.
[0038] The plasmin may also be ocriplasmin (JETREA.RTM.) or variants or derivatives thereof. Ocriplasmin is a recombinant truncated form of human plasmin produced by recombinant DNA technology in a Pichia pastoris expression system. Ocriplasm is a protein made up of 249 amino acids with a molecular weight of 27.2 kDa, and has two peptide chains. The amino acid sequence for the truncated heavy chain is as follows:
TABLE-US-00008 (SEQ ID NO: 8) APSFDCGKPQVEPKKCPGR
The amino acid sequence for the light chain is as follows:
TABLE-US-00009 (SEQ ID NO: 9) VVGGCVAHPHSWPWQVSLRTRFGMHFCGGTLISPEWVLTAAHCLEKSPRPS SYKVILGAHQEVNLEPHVQEIEVSRLFLEPTRKDIALLKLSSPAVITDKVI PACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQLPVIENKVCNRYE FLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSWGLGC ARPNKPGVYVRVSRFVTWIEGVMRNN
The present invention further encompasses variants and derivatives of ocriplasmin.
[0039] The plasmin may be isolated from blood using known isolation techniques. Alternatively, plasminogen may be isolated from the blood using known isolation techniques, and then incubated with proteases that cleave plasminogen into active plasmin to produce purified or isolated plasmin suitable for use in the methods described herein.
[0040] The plasmin may be synthesized chemically using commercially available peptide synthesizers to produce purified or isolated plasmin suitable for use in the methods described herein. Chemical synthesis of peptides and proteins can be used for the incorporation of modified or unnatural amino acids, including D-amino acids and other small organic molecules. Replacement of one or more L-amino acids in a peptide or protein with the corresponding D-amino acid isoforms can be used to increase resistance to enzymatic hydrolysis, and to enhance one or more properties of biological activity, i.e., functional potency or duration of action. Other modifications to the plasmin can be introduced, for example, cross-linking to change the conformation to alter the potency, selectivity or stability of the plasmin.
[0041] For example, the plasmin may be purchased from a commercial vendor, such as Sigma Aldrich, Catalog Number P1867. The present invention also encompasses variants or derivatives of the plasmin supplied by Sigma Aldrich (Cat. No. P1867).
[0042] The plasmin may be a recombinant plasmin obtained by methods well known in the art for recombinant protein expression and purification. A DNA molecule encoding a plasmin or a variant or analog thereof can be generated from known DNA sequences or by deducing the nucleic acid sequences from the amino acid sequence based on known codon usage. The DNA molecule encoding a plasmin can be cloned into a suitable vector, such as a cloning or expression vector, by any of the methodologies known in the art. The cloning or expression vectors contain all the components required for additional cloning of the plasmin DNA, such as restriction enzyme sites, or for the expression of the plasmin, such as a host-specific promoter, and optionally, enhancer sequences. The expression vector can be introduced and expressed in a host cell. A host cell can be any prokaryotic or eukaryotic cell. For example, the plasmin can be expressed in bacterial cells (i.e., E. coli), yeast, insect cells (i.e., Sf9), or mammalian cells. Other suitable host cells are known to those skilled in the art. The host cell can be used to produce or overexpress the plasmin, variant or derivative thereof in culture. Then the biologically expressed plasmin, variant or derivative thereof may be purified using known purification techniques, such as affinity chromatography, to produce purified or isolated plasmin suitable for use in the methods described herein.
[0043] In some embodiments, a variant, derivative or analog of a plasmin may be preferred. Variants, derivatives and analogs of plasmin can be identified or generated by one ordinarily skilled in the art. The plasmin variants, derivatives and analogs can be generated, for example, by using the recombinant methods or methods of synthesis described herein. Plasmin derivatives known in the art include miniplasmin and microplasmin are also suitable for use in the methods disclosed herein.
[0044] As used herein, the term "derivative" and "variant" may be used interchangeably and refers to a plasmin that differs from naturally occurring plasmin, but retains the essential properties thereof. For example, the plasmin derivative may be a biologically active fragment of plasmin, for example, a truncated plasmin. The biologically active fragment contains the catalytic domain of plasmin and possesses serine protease catalytic activity. Alternatively, the plasmin derivative may be a mutated plasmin, wherein at least 1 amino acid, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 20, at least 30, at least 40, or at least 50 amino acids are mutated from the wild-type plasmin. Mutated plasmin may have altered sequences by substitutions, additions, or deletions, that still result in functionally equivalent molecules. In one embodiment, the plasmin derivative is about 99%, about 98%, about 97%, about 96%, about 95%, about 90%, about 85%, about 80%, about 75%, about 70%, about 65%, about 60%, about 55%, or about 50% identity to wild-type plasmin. In some instances, the mutation may increase the potency (protease activity), stability, or number of targets of the plasmin. In another aspect, the mutation may decrease the potency, stability, or number of targets of the plasmin. The mutation may be a conservative amino acid substitution. Alternatively, the mutation may be a non-conservative amino acid substitution. In another embodiment, the plasmin derivative may be chemically modified, for example, by the addition of a chemical moiety that alters activity or stability.
[0045] The term "% identity," in the context of two or more nucleic acid or polypeptide sequences, refer to two or more sequences or subsequences that are the same or have a specified percentage of amino acid residues or nucleotides that are the same, when compared and aligned for maximum correspondence, as measured using one of the following sequence comparison algorithms or by visual inspection. For example, % identity is relative to the entire length of the coding regions of the sequences being compared.
[0046] For sequence comparison, typically one sequence acts as a reference sequence, to which test sequences are compared. When using a sequence comparison algorithm, test and reference sequences are input into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated. The sequence comparison algorithm then calculates the percent sequence identity for the test sequence(s) relative to the reference sequence, based on the designated program parameters. Percent identity is determined using search algorithms such as BLAST and PSI-BLAST (Altschul et al., 1990, J Mol Biol 215:3, 403-410; Altschul et al., 1997, Nucleic Acids Res 25:17, 3389-402).
[0047] The term "analog" as used herein refers to compounds or peptides that retain the essential protease activity of the plasmin. For example, the analog may bear some amino acid sequence similarity to or no sequence similarity to naturally occurring plasmin. However all plasmin analogs retain the functional capability of protease cleavage of any one of the targets of plasmin (i.e., laminin, fibrin), and/or the preferential cleavage at the carboxyl side of Lysine and Arginine residues.
[0048] Plasmin derivatives and analogs can be identified by one ordinarily skilled in the art by screening combinatorial libraries of mutants of plasmin or general therapeutic or pharmaceutical compounds.
Methods of Use
[0049] The present invention provides methods for enhancing the delivery of a therapeutic agent to the eye. The methods of the present invention include administering a plasmin and a therapeutic agent to the eye. The therapeutic agent may be delivered to the eye by any method known in the art. Routes of administration include, but are not limited to, intravitreal, intracameral, subconjunctival, subtenon, retrobulbar, posterior juxtascleral, or topical. Delivery methods include, for example, injection by a syringe and a drug delivery device, such as an implanted vitreal delivery device (i.e., VITRASERT.RTM.).
[0050] Preferably, the therapeutic agent is administered to the vitreous by intravitreal injection for delivery of therapeutic agents to the retina. In some embodiments, the methods of the present invention provide enhanced delivery to cells of the retina. Exemplary retinal cells, include, but are not limited to, photoreceptor cells (e.g., rods, cones, and photosensitive retinal ganglion cells), horizontal cells, bipolar cells, amacrine cells, retinal ganglion cells, Muller glial cell, and retinal pigment epithelial cells.
[0051] In one embodiment, the plasmin or derivative thereof is administered concurrently or sequentially with the therapeutic agent. For concurrent administration, the plasmin or derivative thereof can be formulated with the therapeutic agent in a single composition suitable for delivery, for example, injection, by methods known in the art. Alternatively, the plasmin or derivative thereof can be injected in separate compositions, simultaneously or sequentially. In a preferred embodiment, the plasmin may be administered prior to administration of the therapeutic agent.
[0052] Such formulations comprise a pharmaceutically and/or physiologically acceptable vehicle, diluent, carrier or excipient, such as buffered saline or other buffers, e.g., HEPES, to maintain physiologic pH. For a discussion of such components and their formulation, see, generally, Gennaro, A E., Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins Publishers; 2003 or latest edition). See also, WO00/15822. If the preparation is to be stored for long periods, it may be frozen, for example, in the presence of glycerol.
[0053] The dosage of plasmin or derivative thereof to be administered can be optimized by one of ordinary skill in the art. Delivery to certain target ocular tissues may require lower doses of plasmin or higher doses of plasmin, depending on the location of the target tissue, intervening ocular structures, and ability of the agent to penetrate the target tissue. Preferably, the dose of plasmin administered is about 0.001 UI (enzyme units) per eye, 0.025 UI per eye, about 0.05 UI per eye, about 0.075 UI per eye, about 0.100 UI per eye, about 0.150 UI per eye, or about 0.200 UI per eye.
[0054] In some embodiments, the methods for enhanced delivery disclosed herein may provide increased efficacy of a therapeutic agent. Increased efficacy of the therapeutic agent can be determined by measuring the therapeutic effect of the therapeutic agent. Treatment is efficacious if the treatment leads to clinical benefit such as, alleviation of a symptom in the subject. For example, in a degenerative retinal disease, such as retinitis pigmentosa, treatment is efficacious when light sensitivity or another aspect of vision is improved or restored. When treatment is applied prophylactically, "efficacious" means that the treatment retards or prevents an ocular disease or disorder or prevents or alleviates a symptom of clinical symptom of an ocular disease or disorder. Efficaciousness is determined in association with any known method for diagnosing or treating the particular ocular disease or disorder.
[0055] In some embodiments, the therapeutic agent is a nucleic acid or a nucleic acid expression vector (i.e. a viral vector) encoding a therapeutic transgene or an siRNA species (i.e., short hairpin or microRNA). With regard to such therapeutic agents, the enhanced delivery of a therapeutic agent provided by the methods disclosed herein may result in increased transduction efficiency. Increased transduction efficiency can be determined by measuring the level of expression of the transgene introduced by the viral vector.
Therapeutic Agents
[0056] The therapeutic agents to be delivered to the eye by the methods described herein are any therapeutic agents known in the art for treating, alleviating, reducing, or preventing a symptom of an ocular disease, an ocular disorder, or an ocular condition. The therapeutic agent may be a small molecule, a nucleic acid, an antibody, or a peptide.
[0057] Examples of small molecules suitable for use in the methods described herein include, but are not limited to, tyrosine kinase inhibitors, antibiotics, anti-inflammatory agents, glucocorticoids, opioid antagonists, and other enzyme inhibitors.
[0058] Examples of nucleic acids suitable for use in the methods described herein include, but are not limited to, viral vectors encoding therapeutic transgenes (i.e., channelopsins, or halorhodopsin), RNA interference molecules (i.e., short hairpins, siRNA, or microRNAs). In a particularly preferred embodiment, the therapeutic agents are viral vectors encoding transgenes for gene therapy. Particularly preferred viral vectors are rAAV vectors that encode channelopsins or halorhodopsins for expression in the retina to restore light sensitivity.
[0059] Examples of antibodies suitable for use in the methods described herein include, but are not limited to, ranibizumab (Lucentis.RTM.), VEGF antibodies (Eylea.RTM.), bevacizumab (Avastin.RTM.), infliximab, etanercept, and adalimumab.
[0060] Examples of proteins or peptides suitable for use in the methods described herein include, but are no limited to, microplasmin (Ocriplasmin, Jetrea.RTM.), macugen pagylated polypeptide (Pegaptanib), and integrin peptides. In some aspects, the peptide therapeutic is a collection of peptides, containing two or more peptides.
[0061] Any of the therapeutic agents described herein may be optionally encapsulated in a carrier, such as a nanoparticle, a polymer, or a liposome. These carrier agents may serve to further enhance the delivery of the therapeutic agent to the eye. In some aspects, the carrier agents may alter the properties of the therapeutic agents, such as increasing the stability (half-life) or providing sustained-release properties to the therapeutic agents. Alternatively, the carrier may protect the therapeutic agent from the proteolytic activities of plasmin if formulated in the same composition for delivery.
Gene Therapy
[0062] As a large number of ocular diseases and disorders result from aberrant gene expression in various ocular tissues, gene therapy possesses increasing potential as an effective therapy. However, the efficacy of gene therapy in the eye has been limited due to the challenges of effective delivery and transduction of the therapeutic viral vectors throughout any ocular tissue.
[0063] Thus, the present invention provides methods for increased efficiency of delivery of transgenes to the eye for treating an ocular disease or disorder, or for restoring or improving vision. Transgenes of particular interest for restoration of photosensitivity or vision include photosensitive proteins, such as opsin genes or rhodopsin genes. As used herein, "transgene" refers to a polynucleotide encoding a polypeptide of interest, wherein the polynucleotide is present in a nucleic acid expression vector suitable for gene therapy (e.g., a viral vector such as AAV).
[0064] Previous studies have shown that injection of a recombinant adeno-associated viral vector encoding a transgene, such as channelopsin-2, results in poor delivery of the vector and low expression of Chop2 in the inner retinal cells, especially bipolar cells. In non-human primates, AAV-mediated gene transfection was found to be more efficient in peripheral retina, fovea, and along blood vessels, suggesting that inner limiting membrane (ILM), which is the boundary between the retina and the vitreous space, is a major barrier (Ivanova et al., 2010).
[0065] The present invention provides a solution to this problem by using plasmin or derivatives thereof to dissolve the components the ILM, such as laminin and fibronectin. Accordingly, therapeutic agents will have greater accessibility to the retina, specifically the cells of the inner retina such as the retinal bipolar cells, retinal ganglion cells, Muller glial cells, and retinal pigment epithelial cells. The methods described herein provide enhanced delivery of therapeutic compounds, such as therapeutic viral vectors. The enhanced delivery of viral vectors is demonstrated by increased transduction efficiency, increased expression of the therapeutic transgene (i.e., Chop2), and increased efficacy of the therapeutic compound (i.e., increased light sensitivity or restoration of vision).
[0066] Nucleic acid expression vectors suitable for use in gene therapy are known in the art. For example, the nucleic acid expression vector is a viral vector. The viral vectors can be retroviral vectors, adenoviral vectors, adeno-associated vectors (AAV), or lentiviral vectors, or any engineered or recombinant viral vector known in the art. Particularly preferred viral vectors are adeno-associated vectors, for example, AAV-1, AAV-2, AAV-3, AAV-4, AAV-5, AAV-6, AAV-7, AAV-8, AAV-9, AAV-10, AAV-11, AAV-12 or any engineered or recombinant AAV known in the art. In a particularly preferred embodiment, the vector is recombinant AAV-2 (rAAV2).
[0067] In some embodiments, a recombinant adeno-associated viral (rAAV) vector comprises a capsid protein with a mutated tyrosine residue which enables to the vector to have improved transduction efficiency of a target cell, e.g., a retinal bipolar cell (e.g. ON or OFF retinal bipolar cells; rod and cone bipolar cells). In some cases, the rAAV further comprises a promoter (e.g., mGluR6, or fragment thereof) capable of driving the expression of a protein of interest in the target cell.
[0068] In one embodiment, a mutation may be made in any one or more of tyrosine: residues of the capsid protein of AAV 1-12 or hybrid. AAVs. In specific embodiments these are surface exposed tyrosine residues. In a related embodiment the tyrosine residues are part of the VP1, VP2, or VP3 capsid protein. In exemplary embodiments, the mutation may be made at one or more of the following amino acid residues of an AAV-VP3 capsid protein: Tyr252, Tyr272, Tyr444, Tyr500, Tyr700, Tyr704, Tyr730; Tyr275, Tyr281, Tyr508, Tyr576, Tyr612, Tyr673 or Tyr720. Exemplary mutations are tyrosine-to-phenylalanine mutations including, but not limited to, Y252F, Y272F, Y444F, Y500F, Y700F, Y704F, Y730F, Y275F, Y281F, Y508F, Y576F, Y612G, Y673F and Y720F. in a specific embodiment these mutations are made in the AAV2 serotype. In some cases, an AAV2 serotype comprises a Y444F mutation and/or an AAV8 serotype comprises a Y733F mutation, wherein 444 and 733 indicate the location of a point tyrosine mutation of the viral capsid. In further embodiments, such mutated AAV2 and AAV8 serotypes encode a light-sensitive protein and also comprise a modified mGluR6 promoter to drive expression of such light-sensitive protein. Such AAV vectors are described in, for example, Petrs-Silva et al., Mol Ther., 2011 19:293-301).
[0069] In some embodiments, the expression of the therapeutic transgene is driven by a constitutive promoter, i.e., CAG promoter, CMV promoter, LTR. In other embodiments, the promoter is an inducible or a cell-specific promoter. Cell type-specific promoters that enable transgene expression in specific subpopulations of cells, i.e., retinal neuron cells or degenerating cells, may be preferred. These cells may include, but are not limited to, a retinal ganglion cell, a photoreceptor cell, a bipolar cell, a rod bipolar cell, an ON-type cone bipolar cell, a retinal ganglion cell, a photosensitive retinal ganglion cell, a horizontal cell, an amacrine cell, an AII amacrine cell, or a retinal pigment epithelial cell. Cell type-specific promoters are well known in the art. Particularly preferred cell type-specific promoters include, but are not limited to mGluR6, NK-3, and Pcp2(L7). Cell type-specific promoters modified using recombinant DNA techniques known in the art to increase efficiency of expression and selective targeting are also encompassed in the present invention. For example, a modified mGluR6 promoter contains a combination of regulatory elements from the mGluR6 gene, as described in U.S. Provisional Application No. 61/951,360, hereby incorporated by reference in its entirety.
[0070] In one embodiment of the present invention, the therapeutic transgene can be any light-sensitive opsin. The opsin family of genes includes vertebrate (animal) and invertebrate opsins. Animal opsins are G-protein coupled receptors (GPCRs) with 7-transmembrane helices which regulate the activity of ion channels. Invetertebrate rhodopsins are usually not GPCRs but are light-sensitive or light-activated ion pumps or ion channels.
[0071] As referred to herein, an opsin gene or light-sensitive protein includes, but is not limited to, channelrhodopsins, or channelopsins, (i.e., ChR1, ChR2, vChR1 from Volvox carteri, vChR2, and other variants identified from any vertebrate, invertebrate, or microbe), halorhodopsins (NpHR), melanopsins, pineal opsins, photopsins, bacteriorhodopisins, proteorhodopsins and functional variants or chimeras thereof. A light-sensitive protein of this invention can occur naturally in plant, animal, archaebacterial, algal, or bacterial cells, or can alternatively be created through laboratory techniques. Examples of opsin genes are discussed in further detail below.
[0072] Examples of channelrhodopsins, or channelopsins, as transgenes in the present invention include channelrhodopsins Chop1 (also known as ChR1) (GenBank accession number AB058890/AF385748) and Chop2 (also known as ChR2) (GenBank accession number AB058891/AF461397) are two rhodopsins from the green alga Chlamydomonas reinhardtii (Nagel, 2002; Nagel, 2003) Channelopsins are a seven transmembrane domain proteins that become photo-switchable (light sensitive) when bound to the chromophore all-trans-retinal. Channelopsins, when linked to a retinal molecule via Schiff base linkage forms a light-gated, nonspecific, inwardly rectifying, cation channel, called a channelrhodopsin. These light-sensitive channels that, when expressed and activated in neural tissue, allow for a cell to be depolarized when stimulated with light (Boyden, 2005). A Chop2 fragment (315 amino acids) has been shown to efficiently increase photosensitivity and vision in mouse models of photoreceptor degeneration (Bi et al., Neuron, 2006, and U.S. Pat. No. 8,470,790; both of which are hereby incorporated by reference). Chop2 mutants and variants as described in PCT Publication WO 2013/134295 (hereby incorporated by reference) may also be expressed using the promoters described herein. The present invention also provides for use of Volvox carteri channelrhodopsins (i.e., vChR1 and vChR2).
[0073] NpHR (Halorhodopsin) (GenBank accession number EF474018) is from the haloalkaliphilic archaeon Natronomonas pharaonic. In certain embodiments variants of NpHR can be created. In specific embodiments single or multiple point mutations to the NpHR protein can result in NpHR variants. In specific embodiments a mammalian codon optimized version of NpHR can be utilized. In one embodiment NpHR variants are utilized. In one specific embodiment eNpHR (enhanced NpHR) is utilized. Addition of the amino acids FCYENEV to the NpHR C-terminus along with the signal peptide from the (3 subunit of the nicotinic acetylcholine receptor to the NpHR N-terminus results in the construction of eNpHR.
[0074] Melanopsin (GenBank accession number 6693702) is a photopigment found in specialized photosensitive ganglion cells of the retina that are involved in the regulation of circadian rhythms, pupillary light reflex, and other non-visual responses to light. In structure, melanopsin is an opsin, a retinylidene protein variety of G-protein-coupled receptor. Melanopsin resembles invertebrate opsins in many respects, including its amino acid sequence and downstream signaling cascade. Like invertebrate opsins, melanopsin appears to be a bistable photopigment, with intrinsic photoisomerase activity. In certain embodiments variants of melanopsin can be created. In specific embodiments single or multiple point mutations to the melanopsin protein can result in melanopsin variants.
[0075] Light-sensitive proteins may also include proteins that are at least about 10%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 95%, or at least about 99% identical to any of the light-sensitive proteins described herein (i.e., ChR1, ChR2, vChR1, vChR2, NpHR and melanopsin). The light-sensitive proteins of the present invention may also include proteins that have at least one mutation. The mutation may be a point mutation.
[0076] In some embodiments, light-sensitive proteins can modulate signaling within neural circuits and bidirectionally control behavior of ionic conductance at the level of a single neuron. In some embodiments the neuron is a retinal neuron, a retinal bipolar cell (e.g. ON or OFF retinal bipolar cells; rod and cone bipolar cells), a retinal ganglion cell, a photoreceptor cell, or a retinal amacrine cell.
[0077] In some embodiments, a polyA tail can be inserted downstream of the transgene in an expression cassette or nucleic acid expression vector of the present invention. Suitable polyA tails are known in the art, and include, for example, human growth hormone poly A tail (hGHpA), bovine growth hormone polyA tail (bGHpA), bovine polyA, SV40 polyA, and AV40pA.
[0078] Upon illumination by the preferred dose of light radiation, rhodopsin proteins opens the pore of the channel, through which H.sup.+, Na.sup.+, K.sup.+, and/or Ca.sup.2+ ions flow into the cell from the extracellular space. Activation of the rhodopsin channel typically causes a depolarization of the cell expressing the channel Depolarized cells produce graded potentials and or action potentials to carry information from the rhodopsin-expressing cell to other cells of the retina or brain, to increase light sensitivity or restore vision. Methods of improving vision or light sensitivity by administration of a vector encoding a channelopsin (or variant thereof) are described in PCT/US2007/068263, the contents of which are herein incorporated in its entirety.
[0079] Accordingly, a dual rhodopsin system can be used to recapitulate the ON and OFF pathways integral to visual processing and acuity. Briefly, a Chop2 protein of the present invention can be specifically targeted to ON type retinal neurons (i.e., ON type ganglion cells and/or ON type bipolar cells), while a hypopolarizing light sensor (i.e., halorhodopsin or other chloride pump known in the art) can be targeted to OFF type retinal neurons (i.e. OFF type ganglion cells and/or OFF type bipolar cells) to create ON and OFF pathways. The specific targeting to preferred cell subpopulations can be achieved through the use of different cell type-specific promoters. For example, Chop2 expression may be driven by the mGluR6 promoter for targeted expression in ON-type retinal neurons (i.e., ON type ganglion cells and/or ON type bipolar cells) while a hypopolarizing channel, such as halorhodopsin, expression is driven by the NK-3 promoter for targeted expression in OFF-type retinal neurons (i.e., OFF type ganglion cells and/or OFF type bipolar cells).
[0080] An alternative approach to restore ON and OFF pathways in the retina is achieved by, expressing a depolarizing light sensor, such as ChR2, to rod bipolar cells or AII amacrine. In this approach, the depolarization of rod bipolar cells or AII amacrine cells can lead to the ON and OFF responses at the levels of cone bipolar cells and the downstream retinal ganglion cells. Thus, the ON and OFF pathways that are inherent in the retina are maintained.
[0081] An effective amount of rAAV virions carrying a nucleic acid sequence encoding the rhodopsin DNA under the control of the promoter of choice, preferably a constitutive CMV promoter or a cell-specific promoter such as mGluR6, is preferably in the range of between about 10.sup.10 to about 10.sup.13 rAAV infectious units in a volume of between about 25 and about 800 .mu.l per injection. The rAAV infectious units can be measured according to McLaughlin, S K et al., 1988, J Virol 62:1963. More preferably, the effective amount is between about 10.sup.10 and about 10.sup.12 rAAV infectious units and the injection volume is preferably between about 50 and about 150 .mu.l. Other dosages and volumes, preferably within these ranges but possibly outside them, may be selected by the treating professional, taking into account the physical state of the subject (preferably a human), who is being treated, including, age, weight, general health, and the nature and severity of the particular ocular disorder.
[0082] It may also be desirable to administer additional doses ("boosters") of the present nucleic acid(s) or rAAV compositions. For example, depending upon the duration of the transgene expression within the ocular target cell, a second treatment may be administered after 6 months or yearly, and may be similarly repeated. Neutralizing antibodies to AAV are not expected to be generated in view of the routes and doses used, thereby permitting repeat treatment rounds.
[0083] The need for such additional doses can be monitored by the treating professional using, for example, well-known electrophysiological and other retinal and visual function tests and visual behavior tests. The treating professional will be able to select the appropriate tests applying routine skill in the art. It may be desirable to inject larger volumes of the composition in either single or multiple doses to further improve the relevant outcome parameters.
Ocular Disorders
[0084] The ocular disorders for which the methods of the present invention are intended and may be used to improve one or more parameters of vision include, but are not limited to, developmental abnormalities that affect both anterior and posterior segments of the eye. Anterior segment disorders include glaucoma, cataracts, corneal dystrophy, keratoconus. Posterior segment disorders include blinding disorders caused by photoreceptor malfunction and/or death caused by retinal dystrophies and degenerations. Retinal disorders include congenital stationary night blindness, age-related macular degeneration, congenital cone dystrophies, and a large group of retinitis-pigmentosa (RP)-related disorders. These disorders include genetically pre-disposed death of photoreceptor cells, rods and cones in the retina, occurring at various ages. Among those are severe retinopathies, such as subtypes of RP itself that progresses with age and causes blindness in childhood and early adulthood and RP-associated diseases, such as genetic subtypes of LCA, which frequently results in loss of vision during childhood, as early as the first year of life. The latter disorders are generally characterized by severe reduction, and often complete loss of photoreceptor cells, rods and cones. Other ocular diseases that may benefit from the methods described herein include, but are not limited to, retinoblastoma, ocular melanoma, diabetic retinopathy, hypertensive retinopathy, any inflammation of the ocular tissues (i.e., chorioretinal inflammation, scleritis, keratitis, uveitis, etc.), or infection (i.e., bacterial or viral).
[0085] In particular, the viral-mediated delivery of rhodopsins using the methods of the present invention useful for the treatment and/or restoration of at least partial vision to subjects that have lost vision due to ocular disorders, such as RPE-associated retinopathies, which are characterized by a long-term preservation of ocular tissue structure despite loss of function and by the association between function loss and the defect or absence of a normal gene in the ocular cells of the subject. A variety of such ocular disorders are known, such as childhood onset blinding diseases, retinitis pigmentosa, macular degeneration, and diabetic retinopathy, as well as ocular blinding diseases known in the art. It is anticipated that these other disorders, as well as blinding disorders of presently unknown causation which later are characterized by the same description as above, may also be successfully treated by the methods described herein. Thus, the particular ocular disorder treated by the present invention may include the above-mentioned disorders and a number of diseases which have yet to be so characterized.
Restoration of Light Sensitivity
[0086] These methods described herein may be used in subjects of normal and/or impaired vision. The enhanced delivery of a therapeutic compound, as described herein, may preserve, improve, or restore vision. The term "vision" as used herein is defined as the ability of an organism to usefully detect light as a stimulus for differentiation or action. Vision is intended to encompass the following: [0087] 1. Light detection or perception--the ability to discern whether or not light is present; [0088] 2. Light projection--the ability to discern the direction from which a light stimulus is coming; [0089] 3. Resolution--the ability to detect differing brightness levels (i.e., contrast) in a grating or letter target; and [0090] 4. Recognition--the ability to recognize the shape of a visual target by reference to the differing contrast levels within the target. Thus, "vision" includes the ability to simply detect the presence of light. The methods of the present invention can be used to improve or restore vision, wherein the improvement or restoration in vision includes, for example, increases in light detection or perception, increase in light sensitivity or photosensitivity in response to a light stimulus, increase in the ability to discern the direction from which a light stimulus is coming, increase in the ability to detect differing brightness levels, increase in the ability to recognize the shape of a visual target, and increases in visual evoked potential or transmission from the retina to the cortex. As such, improvement or restoration of vision may or may not include full restoration of sight, i.e., wherein the vision of the patient treated with the present invention is restored to the degree to the vision of a non-affected individual. The visual recovery described in the animal studies described below may, in human terms, place the person on the low end of vision function by increasing one aspect of vision (i.e., light sensitivity, or visual evoked potential) without restoring full sight. Nevertheless, placement at such a level would be a significant benefit because these individuals could be trained in mobility and potentially in low order resolution tasks which would provide them with a greatly improved level of visual independence compared to total blindness. Even basic light perception can be used by visually impaired individuals, whose vision is improved using the present compositions and methods, to accomplish specific daily tasks and improve general mobility, capability, and quality of life.
[0091] The degree of restoration of vision can be determined through the measurement of vision before, and preferably after, administering a vector comprising, for example, DNA encoding a therapeutic transfene such as Chop2 or halorhodopsin or both. Vision can be measured using any of a number of methods well-known in the art or methods not yet established. Vision, as improved or restored by the present invention, can be measured by any of the following visual responses: [0092] 1. a light detection response by the subject after exposure to a light stimulus--in which evidence is sought for a reliable response of an indication or movement in the general direction of the light by the subject individual when the light it is turned on; [0093] 2. a light projection response by the subject after exposure to a light stimulus in which evidence is sought for a reliable response of indication or movement in the specific direction of the light by the individual when the light is turned on; [0094] 3. light resolution by the subject of a light vs. dark patterned visual stimulus, which measures the subject's capability of resolving light vs dark patterned visual stimuli as evidenced by: [0095] a. the presence of demonstrable reliable optokinetically produced nystagmoid eye movements and/or related head or body movements that demonstrate tracking of the target (see above) and/or [0096] b. the presence of a reliable ability to discriminate a pattern visual stimulus and to indicate such discrimination by verbal or non-verbal means, including, for example pointing, or pressing a bar or a button; or [0097] 4. electrical recording of a visual cortex response to a light flash stimulus or a pattern visual stimulus, which is an endpoint of electrical transmission from a restored retina to the visual cortex, also referred to as the visual evoked potential (VEP). Measurement may be by electrical recording on the scalp surface at the region of the visual cortex, on the cortical surface, and/or recording within cells of the visual cortex.
[0098] Thus, improvement or restoration of vision, according to the present invention, can include, but is not limited to: increases in amplitude or kinetics of photocurents or electrical response in response to light stimulus in the retinal cells, increases in light sensitivity (i.e., lowering the threshold light intensity required for initiating a photocurrent or electrical response in response to light stimulus, thereby requiring less or lower light to evoke a photocurrent) of the retinal cells, increases in number or amplitude of light-evoked spiking or spike firings, increases in light responses to the visual cortex, which includes increasing in visual evoked potential transmitted from the retina or retinal cells to the visual cortex or the brain.
[0099] Both in vitro and in vivo studies to assess the various parameters of the present invention may be used, including recognized animal models of blinding human ocular disorders. Large animal models of human retinopathy, e.g., childhood blindness, are useful. The examples provided herein allow one of skill in the art to readily anticipate that this method may be similarly used in treating a range of retinal diseases.
[0100] While earlier studies by others have demonstrated that retinal degeneration can be retarded by gene therapy techniques, the present invention demonstrates a definite physiological recovery of function, which is expected to generate or improve various parameters of vision, including behavioral parameters.
[0101] Behavioral measures can be obtained using known animal models and tests, for example performance in a water maze, wherein a subject in whom vision has been preserved or restored to varying extents will swim toward light (Hayes, J M et al., 1993, Behav Genet 23:395-403).
[0102] In models in which blindness is induced during adult life or congenital blindness develops slowly enough that the individual experiences vision before losing it, training of the subject in various tests may be done. In this way, when these tests are re-administered after visual loss to test the efficacy of the present compositions and methods for their vision-restorative effects, animals do not have to learn the tasks de novo while in a blind state. Other behavioral tests do not require learning and rely on the instinctiveness of certain behaviors. An example is the optokinetic nystagmus test (Balkema G W et al., 1984, Invest Ophthalmol Vis Sci. 25:795-800; Mitchiner J C et al., 1976, Vision Res. 16:1169-71).
[0103] The present invention may also be used in combination with other forms of vision therapy known in the art to improve or restore vision. For example, the use of visual prostheses, which include retinal implants, cortical implants, lateral geniculate nucleus implants, or optic nerve implants. Thus, in addition to genetic modification of surviving retinal neurons using the present methods, the subject being treated may be provided with a visual prosthesis before, at the same time as, or after the molecular method is employed. The effectiveness of visual prosthetics can be improved with training of the individual, thus enhancing the potential impact of the Chop2 transformation of patient cells as contemplated herein. Training methods, such as habituation training characterized by training the subject to recognize recognize (i) varying levels of light and/or pattern stimulation, and/or (ii) environmental stimulation from a common light source or object as would be understood by one skilled in the art; and orientation and mobility training characterized by training the subject to detect visually local objects and move among said objects more effectively than without the training. In fact, any visual stimulation techniques that are typically used in the field of low vision rehabilitation are applicable here.
[0104] As used herein, by a "subject" is meant an individual. Thus, the "subject" can include domesticated animals (e.g., cats, dogs, etc.), livestock (e.g., cattle, horses, pigs, sheep, goats, etc.), laboratory animals (e.g., mouse, rabbit, rat, guinea pig, etc.), and birds. "Subject" can also include a mammal, such as a primate or a human. Preferably, the subject is a human. A "subject in need thereof" is a subject suffering from or at risk of developing or suffering from an ocular disease or disorder. A subject at risk of developing or suffering from an ocular disease or disorder can be diagnosed by a physician or ocular specialist using routine methods in the art.
EXAMPLES
Example 1: Plasmin Increases Delivery of AAV2-Vector Encoding Chop2 to the Retina
[0105] Delivery of a therapeutic viral construct encoding a functional GFP-channelopsin-2 protein to the retina was examined Injection of 6.times.10.sup.12 vg/ml of AAV2 vector AAV2/2-ChR2-GFP-WPRE-hGHpA in control (FIG. 1A) or co-injection with plasmin (FIG. 1B) was performed into the vitreous space of the eye of one month old C56BL/6J. The concentration of plasmin injected with vector was 0.025 IU/eye. After one month, the mice retinas were isolated, and retinal vertical sections were prepared. The sections were immunostained and cells were counted Immunofluorescence analysis of the sections showed that co-injection with plasmin increased the transduction efficiency of the therapeutic AAV2-ChR2-GFP vector, as evidenced by increased fluorescence in comparison to control (FIG. 1).
Example 2: Plasmin Increases Transduction Efficiency in Retinal Ganglion Cells
[0106] Using the same experimental set-up as in Example 1, a vector (2.times.10.sup.12 vg/ml) encoding GFP was co-injected with either control or varying concentrations of plasmin: low (L=0.005 IU/eye), middle (M=0.025 IU/eye), and high (H=0.100 IU/eye). After 1 month, retinal whole mounts were prepared and immunostained. Representative images for each plasmin concentration and control are shown in FIG. 2. As shown, treatment with plasmin increases GFP expression.
[0107] To further quantify these results, GFP-expressing retinal ganglion cells were counted from multiple unit areas of 223 .mu.m.times.167 .mu.m. The results are presented in FIG. 3A. As shown, treatment with low, middle and high doses of plasmin resulted in statistically significantly increased levels of GFP expression in retinal ganglion cells.
[0108] Neurotoxicity as a result of plasmin injection was also examined. The retinal whole mounts were stained with DAPI for cell-counting. The number of cells over multiple unit areas of 223 .mu.m.times.167 .mu.m were counted and compared between control and low, middle and high doses of plasmin. As shown in FIG. 3B, the cell counts were not found to differ significantly between control and plasmin-treated retinas (p=0.74). As such, the tested concentrations of plasmin were not shown to have any neurotoxic effect to the retinal ganglion cells, thereby indicating that plasmin is safe for use in the eye, even at high doses.
Example 3: Plasmin Increases Transduction Efficiency in Retinal Bipolar Cells
[0109] Comparison of the transduction efficiency of a viral vector encoding mCherry fluorescent protein when co-injected with different concentrations of plasmin was assessed in vivo. Specifically, overall levels and the localization of mCherry expression throughout the retina were examined. An AAV2 vector with an Y444F capsid mutation carrying mCherry under control of an mGluR6 promoter were injected at a concentration of 2.times.10.sup.12 vg/ml. The mGluR6 promoter directs expression of mCherry specifically to the retinal bipolar cells.
[0110] The AAV2 mCherry vector was co-injected with three doses of plasmin, high (H=0.100 IU/eye), middle (M=0.025 IU/eye), and low (L=0.005 IU/eye). After 1 month, the retinas were isolated and retinal whole-mounts were prepared. Transduction efficiency was evaluated by immunostaining of mCherry for immunofluorescence analysis and cell counting. Cells were counted from multiple unit areas of 223 .mu.m.times.167 .mu.m.
[0111] Injection of the vectors without plasmin did not result in uniform mCherry expression in retinal bipolar cells across the entire retina (FIG. 4, control, top panels). Transduction efficiency was low in the center (A) and middle (B) retina, but high in the periphery. Co-injection of the AAV2 mCherry vector with increasing dosages of plasmin (low, middle and high, bottom panels) resulted in increased transduction efficiency at each retinal region in a dose-dependent manner.
[0112] The qualitative results from immunofluorescence images were verified by cell counting. Quantification of mCherry-expressing cells when co-injected with or without plasmin showed that plasmin significantly increased the density of mCherry-expressing retinal cells. The increase in transduction efficiency with plasmin compared to control was statistically significant with all three doses of plasmin at the center of the retina. Middle and high doses of plasmin resulted in a statistically significant increase in mCherry expression at the mid-region and periphery of the retina. These results show that plasmin enhances transduction efficiency throughout the retina, including the peripheral, middle, and center regions of the retina.
Other Embodiments
[0113] While the invention has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications are within the scope of the following claims.
[0114] The patent and scientific literature referred to herein establishes the knowledge that is available to those with skill in the art. All United States patents and published or unpublished United States patent applications cited herein are incorporated by reference. All published foreign patents and patent applications cited herein are hereby incorporated by reference. All other published references, documents, manuscripts and scientific literature cited herein are hereby incorporated by reference.
[0115] While this invention has been particularly shown and described with references to preferred embodiments thereof, it will be understood by those skilled in the art that various changes in form and details may be made therein without departing from the scope of the invention encompassed by the appended claims.
Sequence CWU 1
1
91809PRTHomo sapiens 1Met Glu His Lys Glu Val Val Leu Leu Leu Leu
Leu Phe Leu Lys Ser1 5 10 15Gly Gln Gly Glu Pro Leu Asp Asp Tyr Val
Asn Thr Gln Gly Ala Ser 20 25 30Leu Phe Ser Val Thr Lys Lys Gln Leu
Gly Ala Gly Ser Ile Glu Glu 35 40 45Cys Ala Ala Lys Cys Glu Glu Asp
Glu Glu Phe Thr Cys Arg Ala Phe 50 55 60Gln Tyr His Ser Lys Glu Gln
Gln Cys Val Ile Met Ala Glu Asn Arg65 70 75 80Lys Ser Ser Ile Ile
Ile Arg Met Arg Asp Val Val Leu Phe Glu Lys 85 90 95Lys Val Tyr Leu
Ser Glu Cys Lys Thr Gly Asn Gly Lys Asn Tyr Arg 100 105 110Gly Thr
Met Ser Lys Thr Lys Asn Gly Ile Thr Cys Gln Lys Trp Ser 115 120
125Ser Thr Ser Pro His Arg Pro Arg Phe Ser Pro Ala Thr His Pro Ser
130 135 140Glu Gly Leu Glu Glu Asn Tyr Cys Arg Asn Pro Asp Asn Asp
Pro Gln145 150 155 160Gly Pro Trp Cys Tyr Thr Thr Asp Pro Glu Lys
Arg Tyr Asp Tyr Cys 165 170 175Asp Ile Leu Glu Cys Glu Glu Glu Cys
Met His Cys Ser Gly Glu Asn 180 185 190Tyr Asp Gly Lys Ile Ser Lys
Thr Met Ser Gly Leu Glu Cys Gln Ala 195 200 205Trp Asp Ser Gln Ser
Pro His Ala His Gly Tyr Ile Pro Ser Lys Phe 210 215 220Pro Asn Lys
Asn Leu Lys Lys Asn Tyr Cys Arg Asn Pro Asp Arg Glu225 230 235
240Leu Arg Pro Trp Cys Phe Thr Thr Asp Pro Asn Lys Arg Trp Glu Leu
245 250 255Cys Asp Ile Pro Arg Cys Thr Thr Pro Pro Pro Ser Ser Gly
Pro Thr 260 265 270Tyr Gln Cys Leu Lys Gly Thr Gly Glu Asn Tyr Gly
Asn Val Ala Val 275 280 285Thr Val Ser Gly His Thr Cys Gln His Trp
Ser Ala Gln Thr Pro His 290 295 300Thr His Asn Arg Thr Pro Glu Asn
Phe Pro Cys Lys Asn Leu Asp Glu305 310 315 320Asn Tyr Cys Arg Asn
Pro Asp Gly Lys Arg Ala Pro Trp Cys His Thr 325 330 335Thr Asn Ser
Gln Val Arg Trp Glu Tyr Cys Lys Ile Pro Ser Cys Asp 340 345 350Ser
Ser Pro Val Ser Thr Glu Gln Leu Ala Pro Thr Ala Pro Pro Glu 355 360
365Leu Thr Pro Val Val Gln Asp Cys Tyr His Gly Asp Gly Gln Ser Tyr
370 375 380Arg Gly Thr Ser Ser Thr Thr Thr Thr Gly Lys Lys Cys Gln
Ser Trp385 390 395 400Ser Ser Met Thr Pro His Arg His Gln Lys Thr
Pro Glu Asn Tyr Pro 405 410 415Asn Ala Gly Leu Thr Met Asn Tyr Cys
Arg Asn Pro Asp Ala Asp Lys 420 425 430Gly Pro Trp Cys Phe Thr Thr
Asp Pro Ser Val Arg Trp Glu Tyr Cys 435 440 445Asn Leu Lys Lys Cys
Ser Gly Thr Glu Ala Ser Val Val Ala Pro Pro 450 455 460Pro Val Val
Leu Leu Pro Asp Val Glu Thr Pro Ser Glu Glu Asp Cys465 470 475
480Met Phe Gly Asn Gly Lys Gly Tyr Arg Gly Lys Arg Ala Thr Thr Val
485 490 495Thr Gly Thr Pro Cys Gln Asp Trp Ala Ala Gln Glu Pro His
Arg His 500 505 510Ser Ile Phe Thr Pro Glu Thr Asn Pro Arg Ala Gly
Leu Glu Lys Asn 515 520 525Tyr Cys Arg Asn Pro Asp Gly Asp Val Gly
Gly Pro Trp Cys Tyr Thr 530 535 540Thr Asn Pro Arg Lys Leu Tyr Asp
Tyr Cys Asp Val Pro Gln Cys Ala545 550 555 560Ala Pro Ser Phe Asp
Cys Gly Lys Pro Gln Val Glu Pro Lys Lys Cys 565 570 575Pro Gly Arg
Val Val Gly Gly Cys Val Ala His Pro His Ser Trp Pro 580 585 590Trp
Gln Val Ser Leu Arg Thr Arg Phe Gly Met His Phe Cys Gly Gly 595 600
605Thr Leu Ile Ser Pro Glu Trp Val Leu Thr Ala Ala His Cys Leu Glu
610 615 620Lys Ser Pro Arg Pro Ser Ser Tyr Lys Val Ile Leu Gly Ala
His Gln625 630 635 640Glu Val Asn Leu Glu Pro His Val Gln Glu Ile
Glu Val Ser Arg Leu 645 650 655Phe Leu Glu Pro Thr Arg Lys Asp Ile
Ala Leu Leu Lys Leu Ser Ser 660 665 670Pro Ala Val Ile Thr Asp Lys
Val Ile Pro Ala Cys Leu Pro Ser Pro 675 680 685Asn Tyr Val Val Ala
Asp Arg Thr Glu Cys Phe Ile Thr Gly Trp Gly 690 695 700Glu Thr Gln
Gly Thr Phe Gly Ala Gly Leu Leu Lys Glu Ala Gln Leu705 710 715
720Pro Val Ile Glu Asn Lys Val Cys Asn Arg Tyr Glu Phe Leu Asn Gly
725 730 735Arg Val Gln Ser Thr Glu Leu Cys Ala Gly His Leu Ala Gly
Gly Thr 740 745 750Asp Ser Cys Gln Gly Asp Ser Gly Gly Pro Leu Val
Cys Phe Glu Lys 755 760 765Asp Lys Tyr Ile Leu Gln Gly Val Thr Ser
Trp Gly Leu Gly Cys Ala 770 775 780Arg Pro Asn Lys Pro Gly Val Tyr
Val Arg Val Ser Arg Phe Val Thr785 790 795 800Trp Ile Glu Gly Val
Met Arg Asn Asn 80523538DNAHomo sapiens 2gaatcattaa cttaatttga
ctatctggtt tgtggatgcg tttactctca tgtaagtcaa 60caacatcctg ggattgggac
ccactttctg ggcactgctg gccagtccca aaatggaaca 120taaggaagtg
gttcttctac ttcttttatt tctgaaatca ggtcaaggag agcctctgga
180tgactatgtg aatacccagg gggcttcact gttcagtgtc actaagaagc
agctgggagc 240aggaagtata gaagaatgtg cagcaaaatg tgaggaggac
gaagaattca cctgcagggc 300attccaatat cacagtaaag agcaacaatg
tgtgataatg gctgaaaaca ggaagtcctc 360cataatcatt aggatgagag
atgtagtttt atttgaaaag aaagtgtatc tctcagagtg 420caagactggg
aatggaaaga actacagagg gacgatgtcc aaaacaaaaa atggcatcac
480ctgtcaaaaa tggagttcca cttctcccca cagacctaga ttctcacctg
ctacacaccc 540ctcagaggga ctggaggaga actactgcag gaatccagac
aacgatccgc aggggccctg 600gtgctatact actgatccag aaaagagata
tgactactgc gacattcttg agtgtgaaga 660ggaatgtatg cattgcagtg
gagaaaacta tgacggcaaa atttccaaga ccatgtctgg 720actggaatgc
caggcctggg actctcagag cccacacgct catggataca ttccttccaa
780atttccaaac aagaacctga agaagaatta ctgtcgtaac cccgataggg
agctgcggcc 840ttggtgtttc accaccgacc ccaacaagcg ctgggaactt
tgtgacatcc cccgctgcac 900aacacctcca ccatcttctg gtcccaccta
ccagtgtctg aagggaacag gtgaaaacta 960tcgcgggaat gtggctgtta
ccgtgtccgg gcacacctgt cagcactgga gtgcacagac 1020ccctcacaca
cataacagga caccagaaaa cttcccctgc aaaaatttgg atgaaaacta
1080ctgccgcaat cctgacggaa aaagggcccc atggtgccat acaaccaaca
gccaagtgcg 1140gtgggagtac tgtaagatac cgtcctgtga ctcctcccca
gtatccacgg aacaattggc 1200tcccacagca ccacctgagc taacccctgt
ggtccaggac tgctaccatg gtgatggaca 1260gagctaccga ggcacatcct
ccaccaccac cacaggaaag aagtgtcagt cttggtcatc 1320tatgacacca
caccggcacc agaagacccc agaaaactac ccaaatgctg gcctgacaat
1380gaactactgc aggaatccag atgccgataa aggcccctgg tgttttacca
cagaccccag 1440cgtcaggtgg gagtactgca acctgaaaaa atgctcagga
acagaagcga gtgttgtagc 1500acctccgcct gttgtcctgc ttccagatgt
agagactcct tccgaagaag actgtatgtt 1560tgggaatggg aaaggatacc
gaggcaagag ggcgaccact gttactggga cgccatgcca 1620ggactgggct
gcccaggagc cccatagaca cagcattttc actccagaga caaatccacg
1680ggcgggtctg gaaaaaaatt actgccgtaa ccctgatggt gatgtaggtg
gtccctggtg 1740ctacacgaca aatccaagaa aactttacga ctactgtgat
gtccctcagt gtgcggcccc 1800ttcatttgat tgtgggaagc ctcaagtgga
gccgaagaaa tgtcctggaa gggttgtagg 1860ggggtgtgtg gcccacccac
attcctggcc ctggcaagtc agtcttagaa caaggtttgg 1920aatgcacttc
tgtggaggca ccttgatatc cccagagtgg gtgttgactg ctgcccactg
1980cttggagaag tccccaaggc cttcatccta caaggtcatc ctgggtgcac
accaagaagt 2040gaatctcgaa ccgcatgttc aggaaataga agtgtctagg
ctgttcttgg agcccacacg 2100aaaagatatt gccttgctaa agctaagcag
tcctgccgtc atcactgaca aagtaatccc 2160agcttgtctg ccatccccaa
attatgtggt cgctgaccgg accgaatgtt tcatcactgg 2220ctggggagaa
acccaaggta cttttggagc tggccttctc aaggaagccc agctccctgt
2280gattgagaat aaagtgtgca atcgctatga gtttctgaat ggaagagtcc
aatccaccga 2340actctgtgct gggcatttgg ccggaggcac tgacagttgc
cagggtgaca gtggaggtcc 2400tctggtttgc ttcgagaagg acaaatacat
tttacaagga gtcacttctt ggggtcttgg 2460ctgtgcacgc cccaataagc
ctggtgtcta tgttcgtgtt tcaaggtttg ttacttggat 2520tgagggagtg
atgagaaata attaattgga cgggagacag agtgacgcac tgactcacct
2580agaggctgga acgtgggtag ggatttagca tgctggaaat aactggcagt
aatcaaacga 2640agacactgtc cccagctacc agctacgcca aacctcggca
ttttttgtgt tattttctga 2700ctgctggatt ctgtagtaag gtgacatagc
tatgacattt gttaaaaata aactctgtac 2760ttaactttga tttgagtaaa
ttttggtttt ggtcttcaac attttcatgc tctttgttca 2820ccccaccaat
ttttaaatgg gcagatgggg ggatttagct gcttttgata aggaacagct
2880gcacaaagga ctgagcaggc tgcaaggtca cagaggggag agccaagaag
ttgtccacgc 2940atttacctca tcagctaacg agggcttgac atgcattttt
actgtcttta ttcctgacac 3000tgagatgaat gttttcaaag ctgcaacatg
tatggggagt catgcaaacc gattctgtta 3060ttgggaatga aatctgtcac
cgactgcttg acttgagccc aggggacacg gagcagagag 3120ctgtatatga
tggagtgaac cggtccatgg atgtgtaaca caagaccaac tgagagtctg
3180aatgttattc tggggcacac gtgagtctag gattggtgcc aagagcatgt
aaatgaacaa 3240caagcaaata ttgaaggtgg accacttatt tcccattgct
aattgcctgc ccggttttga 3300aacagtctgc agtacacacg gtcacaggag
aatgacctgt gggagagata catgtttaga 3360aggaagagaa aggacaaagg
cacacgtttt accatttaaa atattgttac caaacaaaaa 3420tatccattca
aaatacaatt taacaatgca acagtcatct tacagcagag aaatgcagag
3480aaaagcaaaa ctgcaagtga ctgtgaataa agggtgaatg tagtctcaaa tcctcaaa
3538319PRTHomo sapiens 3Met Glu His Lys Glu Val Val Leu Leu Leu Leu
Leu Phe Leu Lys Ser1 5 10 15Gly Gln Gly4561PRTHomo sapiens 4Glu Pro
Leu Asp Asp Tyr Val Asn Thr Gln Gly Ala Ser Leu Phe Ser1 5 10 15Val
Thr Lys Lys Gln Leu Gly Ala Gly Ser Ile Glu Glu Cys Ala Ala 20 25
30Lys Cys Glu Glu Asp Glu Glu Phe Thr Cys Arg Ala Phe Gln Tyr His
35 40 45Ser Lys Glu Gln Gln Cys Val Ile Met Ala Glu Asn Arg Lys Ser
Ser 50 55 60Ile Ile Ile Arg Met Arg Asp Val Val Leu Phe Glu Lys Lys
Val Tyr65 70 75 80Leu Ser Glu Cys Lys Thr Gly Asn Gly Lys Asn Tyr
Arg Gly Thr Met 85 90 95Ser Lys Thr Lys Asn Gly Ile Thr Cys Gln Lys
Trp Ser Ser Thr Ser 100 105 110Pro His Arg Pro Arg Phe Ser Pro Ala
Thr His Pro Ser Glu Gly Leu 115 120 125Glu Glu Asn Tyr Cys Arg Asn
Pro Asp Asn Asp Pro Gln Gly Pro Trp 130 135 140Cys Tyr Thr Thr Asp
Pro Glu Lys Arg Tyr Asp Tyr Cys Asp Ile Leu145 150 155 160Glu Cys
Glu Glu Glu Cys Met His Cys Ser Gly Glu Asn Tyr Asp Gly 165 170
175Lys Ile Ser Lys Thr Met Ser Gly Leu Glu Cys Gln Ala Trp Asp Ser
180 185 190Gln Ser Pro His Ala His Gly Tyr Ile Pro Ser Lys Phe Pro
Asn Lys 195 200 205Asn Leu Lys Lys Asn Tyr Cys Arg Asn Pro Asp Arg
Glu Leu Arg Pro 210 215 220Trp Cys Phe Thr Thr Asp Pro Asn Lys Arg
Trp Glu Leu Cys Asp Ile225 230 235 240Pro Arg Cys Thr Thr Pro Pro
Pro Ser Ser Gly Pro Thr Tyr Gln Cys 245 250 255Leu Lys Gly Thr Gly
Glu Asn Tyr Arg Gly Asn Val Ala Val Thr Val 260 265 270Ser Gly His
Thr Cys Gln His Trp Ser Ala Gln Thr Pro His Thr His 275 280 285Asn
Arg Thr Pro Glu Asn Phe Pro Cys Lys Asn Leu Asp Glu Asn Tyr 290 295
300Cys Arg Asn Pro Asp Gly Lys Arg Ala Pro Trp Cys His Thr Thr
Asn305 310 315 320Ser Gln Val Arg Trp Glu Tyr Cys Lys Ile Pro Ser
Cys Asp Ser Ser 325 330 335Pro Val Ser Thr Glu Gln Leu Ala Pro Thr
Ala Pro Pro Glu Leu Thr 340 345 350Pro Val Val Gln Asp Cys Tyr His
Gly Asp Gly Gln Ser Tyr Arg Gly 355 360 365Thr Ser Ser Thr Thr Thr
Thr Gly Lys Lys Cys Gln Ser Trp Ser Ser 370 375 380Met Thr Pro His
Arg His Gln Lys Thr Pro Glu Asn Tyr Pro Asn Ala385 390 395 400Gly
Leu Thr Met Asn Tyr Cys Arg Asn Pro Asp Ala Asp Lys Gly Pro 405 410
415Trp Cys Phe Thr Thr Asp Pro Ser Val Arg Trp Glu Tyr Cys Asn Leu
420 425 430Lys Lys Cys Ser Gly Thr Glu Ala Ser Val Val Ala Pro Pro
Pro Val 435 440 445Val Leu Leu Pro Asp Val Glu Thr Pro Ser Glu Glu
Asp Cys Met Phe 450 455 460Gly Asn Gly Lys Gly Tyr Arg Gly Lys Arg
Ala Thr Thr Val Thr Gly465 470 475 480Thr Pro Cys Gln Asp Trp Ala
Ala Gln Glu Pro His Arg His Ser Ile 485 490 495Phe Thr Pro Glu Thr
Asn Pro Arg Ala Gly Leu Glu Lys Asn Tyr Cys 500 505 510Arg Asn Pro
Asp Gly Asp Val Gly Gly Pro Trp Cys Tyr Thr Thr Asn 515 520 525Pro
Arg Lys Leu Tyr Asp Tyr Cys Asp Val Pro Gln Cys Ala Ala Pro 530 535
540Ser Phe Asp Cys Gly Lys Pro Gln Val Glu Pro Lys Lys Cys Pro
Gly545 550 555 560Arg5483PRTHomo sapiens 5Val Tyr Leu Ser Glu Cys
Lys Thr Gly Asn Gly Lys Asn Tyr Arg Gly1 5 10 15Thr Met Ser Lys Thr
Lys Asn Gly Ile Thr Cys Gln Lys Trp Ser Ser 20 25 30Thr Ser Pro His
Arg Pro Arg Phe Ser Pro Ala Thr His Pro Ser Glu 35 40 45Gly Leu Glu
Glu Asn Tyr Cys Arg Asn Pro Asp Asn Asp Pro Gln Gly 50 55 60Pro Trp
Cys Tyr Thr Thr Asp Pro Glu Lys Arg Tyr Asp Tyr Cys Asp65 70 75
80Ile Leu Glu Cys Glu Glu Glu Cys Met His Cys Ser Gly Glu Asn Tyr
85 90 95Asp Gly Lys Ile Ser Lys Thr Met Ser Gly Leu Glu Cys Gln Ala
Trp 100 105 110Asp Ser Gln Ser Pro His Ala His Gly Tyr Ile Pro Ser
Lys Phe Pro 115 120 125Asn Lys Asn Leu Lys Lys Asn Tyr Cys Arg Asn
Pro Asp Arg Glu Leu 130 135 140Arg Pro Trp Cys Phe Thr Thr Asp Pro
Asn Lys Arg Trp Glu Leu Cys145 150 155 160Asp Ile Pro Arg Cys Thr
Thr Pro Pro Pro Ser Ser Gly Pro Thr Tyr 165 170 175Gln Cys Leu Lys
Gly Thr Gly Glu Asn Tyr Arg Gly Asn Val Ala Val 180 185 190Thr Val
Ser Gly His Thr Cys Gln His Trp Ser Ala Gln Thr Pro His 195 200
205Thr His Asn Arg Thr Pro Glu Asn Phe Pro Cys Lys Asn Leu Asp Glu
210 215 220Asn Tyr Cys Arg Asn Pro Asp Gly Lys Arg Ala Pro Trp Cys
His Thr225 230 235 240Thr Asn Ser Gln Val Arg Trp Glu Tyr Cys Lys
Ile Pro Ser Cys Asp 245 250 255Ser Ser Pro Val Ser Thr Glu Gln Leu
Ala Pro Thr Ala Pro Pro Glu 260 265 270Leu Thr Pro Val Val Gln Asp
Cys Tyr His Gly Asp Gly Gln Ser Tyr 275 280 285Arg Gly Thr Ser Ser
Thr Thr Thr Thr Gly Lys Lys Cys Gln Ser Trp 290 295 300Ser Ser Met
Thr Pro His Arg His Gln Lys Thr Pro Glu Asn Tyr Pro305 310 315
320Asn Ala Gly Leu Thr Met Asn Tyr Cys Arg Asn Pro Asp Ala Asp Lys
325 330 335Gly Pro Trp Cys Phe Thr Thr Asp Pro Ser Val Arg Trp Glu
Tyr Cys 340 345 350Asn Leu Lys Lys Cys Ser Gly Thr Glu Ala Ser Val
Val Ala Pro Pro 355 360 365Pro Val Val Leu Leu Pro Asp Val Glu Thr
Pro Ser Glu Glu Asp Cys 370 375 380Met Phe Gly Asn Gly Lys Gly Tyr
Arg Gly Lys Arg Ala Thr Thr Val385 390 395 400Thr Gly Thr Pro Cys
Gln Asp Trp Ala Ala Gln Glu Pro His Arg His 405 410 415Ser Ile Phe
Thr Pro Glu Thr Asn Pro Arg Ala Gly Leu Glu Lys Asn 420 425 430Tyr
Cys Arg Asn Pro Asp Gly Asp Val Gly Gly Pro Trp Cys Tyr Thr 435 440
445Thr Asn Pro Arg Lys Leu Tyr Asp Tyr Cys Asp Val Pro Gln Cys Ala
450 455 460Ala Pro Ser Phe Asp Cys Gly Lys Pro Gln Val Glu Pro Lys
Lys Cys465 470 475 480Pro Gly Arg678PRTHomo sapiens 6Glu Pro Leu
Asp Asp Tyr Val Asn Thr Gln Gly Ala Ser Leu Phe Ser1 5
10 15Val Thr Lys Lys Gln Leu Gly Ala Gly Ser Ile Glu Glu Cys Ala
Ala 20 25 30Lys Cys Glu Glu Asp Glu Glu Phe Thr Cys Arg Ala Phe Gln
Tyr His 35 40 45Ser Lys Glu Gln Gln Cys Val Ile Met Ala Glu Asn Arg
Lys Ser Ser 50 55 60Ile Ile Ile Arg Met Arg Asp Val Val Leu Phe Glu
Lys Lys65 70 757230PRTHomo sapiens 7Val Val Gly Gly Cys Val Ala His
Pro His Ser Trp Pro Trp Gln Val1 5 10 15Ser Leu Arg Thr Arg Phe Gly
Met His Phe Cys Gly Gly Thr Leu Ile 20 25 30Ser Pro Glu Trp Val Leu
Thr Ala Ala His Cys Leu Glu Lys Ser Pro 35 40 45Arg Pro Ser Ser Tyr
Lys Val Ile Leu Gly Ala His Gln Glu Val Asn 50 55 60Leu Glu Pro His
Val Gln Glu Ile Glu Val Ser Arg Leu Phe Leu Glu65 70 75 80Pro Thr
Arg Lys Asp Ile Ala Leu Leu Lys Leu Ser Ser Pro Ala Val 85 90 95Ile
Thr Asp Lys Val Ile Pro Ala Cys Leu Pro Ser Pro Asn Tyr Val 100 105
110Val Ala Asp Arg Thr Glu Cys Phe Ile Thr Gly Trp Gly Glu Thr Gln
115 120 125Gly Thr Phe Gly Ala Gly Leu Leu Lys Glu Ala Gln Leu Pro
Val Ile 130 135 140Glu Asn Lys Val Cys Asn Arg Tyr Glu Phe Leu Asn
Gly Arg Val Gln145 150 155 160Ser Thr Glu Leu Cys Ala Gly His Leu
Ala Gly Gly Thr Asp Ser Cys 165 170 175Gln Gly Asp Ser Gly Gly Pro
Leu Val Cys Phe Glu Lys Asp Lys Tyr 180 185 190Ile Leu Gln Gly Val
Thr Ser Trp Gly Leu Gly Cys Ala Arg Pro Asn 195 200 205Lys Pro Gly
Val Tyr Val Arg Val Ser Arg Phe Val Thr Trp Ile Glu 210 215 220Gly
Val Met Arg Asn Asn225 230819PRTArtificial SequenceRecombinant
truncated form of human plasmin 8Ala Pro Ser Phe Asp Cys Gly Lys
Pro Gln Val Glu Pro Lys Lys Cys1 5 10 15Pro Gly
Arg9230PRTArtificial SequenceRecombinant truncated form of human
plasmin 9Val Val Gly Gly Cys Val Ala His Pro His Ser Trp Pro Trp
Gln Val1 5 10 15Ser Leu Arg Thr Arg Phe Gly Met His Phe Cys Gly Gly
Thr Leu Ile 20 25 30Ser Pro Glu Trp Val Leu Thr Ala Ala His Cys Leu
Glu Lys Ser Pro 35 40 45Arg Pro Ser Ser Tyr Lys Val Ile Leu Gly Ala
His Gln Glu Val Asn 50 55 60Leu Glu Pro His Val Gln Glu Ile Glu Val
Ser Arg Leu Phe Leu Glu65 70 75 80Pro Thr Arg Lys Asp Ile Ala Leu
Leu Lys Leu Ser Ser Pro Ala Val 85 90 95Ile Thr Asp Lys Val Ile Pro
Ala Cys Leu Pro Ser Pro Asn Tyr Val 100 105 110Val Ala Asp Arg Thr
Glu Cys Phe Ile Thr Gly Trp Gly Glu Thr Gln 115 120 125Gly Thr Phe
Gly Ala Gly Leu Leu Lys Glu Ala Gln Leu Pro Val Ile 130 135 140Glu
Asn Lys Val Cys Asn Arg Tyr Glu Phe Leu Asn Gly Arg Val Gln145 150
155 160Ser Thr Glu Leu Cys Ala Gly His Leu Ala Gly Gly Thr Asp Ser
Cys 165 170 175Gln Gly Asp Ser Gly Gly Pro Leu Val Cys Phe Glu Lys
Asp Lys Tyr 180 185 190Ile Leu Gln Gly Val Thr Ser Trp Gly Leu Gly
Cys Ala Arg Pro Asn 195 200 205Lys Pro Gly Val Tyr Val Arg Val Ser
Arg Phe Val Thr Trp Ile Glu 210 215 220Gly Val Met Arg Asn Asn225
230
D00001

D00002

D00003

D00004

D00005

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.