Therapeutic Trem-1 Peptides
Kind Code
U.S. patent application number 16/794037 was filed with the patent office on 2020-08-13 for therapeutic trem-1 peptides. The applicant listed for this patent is Bristol-Myers Squibb Company Universite de Lorraine. Invention is credited to Gilbert FAURE, Sebastien GIBOT, Paola PANINA, Nadia PASSINI.
| Application Number | 20200254058 16/794037 |
| Document ID | 20200254058 / US20200254058 |
| Family ID | 1000004784519 |
| Filed Date | 2020-08-13 |
| Patent Application | download [pdf] |











View All Diagrams
| United States Patent Application | 20200254058 |
| Kind Code | A1 |
| FAURE; Gilbert ; et al. | August 13, 2020 |
THERAPEUTIC TREM-1 PEPTIDES
Abstract
A polypeptide comprising one or more sequences derived from CDR2 or CDR3 of a TREM-1 protein, characterised by the ability to treat, ameliorate, or lessen the symptoms of conditions including sepsis, septic shock or sepsis-like conditions and IBD.
| Inventors: | FAURE; Gilbert; (Vandoeuvre Les Nancy, FR) ; GIBOT; Sebastien; (Vandoeuvre Les Nancy, FR) ; PANINA; Paola; (Milan, IT) ; PASSINI; Nadia; (Milan, IT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004784519 | ||||||||||
| Appl. No.: | 16/794037 | ||||||||||
| Filed: | February 18, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14968479 | Dec 14, 2015 | 10603357 | ||
| 16794037 | ||||
| 13181323 | Jul 12, 2011 | 9273111 | ||
| 14968479 | ||||
| 12320707 | Feb 2, 2009 | 8013116 | ||
| 13181323 | ||||
| 11284086 | Nov 22, 2005 | |||
| 12320707 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/00 20130101; C07K 2319/70 20130101; C07K 2319/20 20130101; G01N 2800/065 20130101; G01N 33/5088 20130101; C07K 14/705 20130101; G01N 2800/26 20130101; A61K 47/6813 20170801; A61K 38/177 20130101 |
| International Class: | A61K 38/17 20060101 A61K038/17; G01N 33/50 20060101 G01N033/50; C07K 14/705 20060101 C07K014/705; A61K 47/68 20060101 A61K047/68 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| May 19, 2005 | JP | 2005-146848 |
Claims
1. An isolated peptide or a derivative thereof, which is capable of acting as antagonist of the TREM-1 protein as defined by SEQ ID NO.2, comprising SEQ ID NO. 22 or at least 3 amino acids from SEQ ID NO. 23, wherein: (a) the polypeptide consists of: (i) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 2; or (ii) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 2 in which one amino acid is substituted conservatively with another amino acid; or (b) the polypeptide consists of an amino acid sequence having at least 80% sequence identity to SEQ ID NOS: 3, 4, 6 or 7; wherein the derivatives of the polypeptides of (a) or (b) are modified by glycosylation, acetylation, pegylation, phosphorylation, amidation, or derivatization by protecting or blocking groups.
2. An isolated polynucleotide encoding the peptide or derivative thereof of claim 1.
3. A vector comprising the polynucleotide of claim 2.
4. A method of treating sepsis, septic shock, or an inflammatory disorder in a subject in need thereof comprising administering to the subject the peptide or derivative thereof of claim 1.
5. An isolated polypeptide consisting of SEQ ID NO: 19 or SEQ ID NO: 7.
6. An isolated polynucleotide encoding the polypeptide of claim 5.
7. A vector comprising the polynucleotide of claim 6.
8. A method of treating sepsis, septic shock, or an inflammatory disorder in a subject in need thereof comprising administering to the subject the polypeptide of claim 5.
9. An isolated peptide or derivative thereof, which is capable of acting as an antagonist of the TREM-1 protein as defined by SEQ ID NO. 1, comprising the amino acid sequence of SEQ ID NO. 20 or at least 3 amino acids of SEQ ID NO. 21, wherein: (a) the peptide consists of: (i) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 1; or (ii) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 1 in which one amino acid is substituted conservatively with another amino acid; or (b) the peptide consists of an amino acid sequence having at least 80% sequence identity to SEQ ID NOs: 16, 17, 18 or 19; and wherein the derivatives of the peptides of (a) or (b) are modified by glycosylation, acetylation, pegylation, phosphorylation, amidation, or derivatization by protecting or blocking groups.
10. An isolated polynucleotide encoding the peptide or derivative thereof of claim 9.
11. A vector comprising the polynucleotide of claim 10.
12. A method of treating sepsis, septic shock, or an inflammatory disorder in a subject in need thereof comprising administering to the subject the peptide or derivative thereof of claim 9.
13. The method of claim 12, wherein the disorder comprises inflammatory bowel disease (IBD).
14. The method of claim 12, wherein the peptide or derivative thereof consists of: (a) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 1; or (b) a contiguous sequence of 5 to 29 amino acids from SEQ ID NO: 1 in which one amino acid is substituted conservatively with another amino acid; and wherein the derivatives of the peptides of (a) or (b) are modified by glycosylation, acetylation, pegylation, phosphorylation, amidation, or derivatization by protecting or blocking groups.
15. The method of claim 12, wherein the peptide or derivative thereof consists of an amino acid sequence having at least 80% sequence identity to SEQ ID NOs: 16, 17, 18 or 19; and wherein the derivatives of the peptide are modified by glycosylation, acetylation, pegylation, phosphorylation, amidation, or derivatization by protecting or blocking groups.
16. The method of claim 15, wherein the peptide consists of an amino acid sequence having at least 80% sequence identity to SEQ ID NO: 19.
17. The method of claim 15, wherein the peptide consists of an amino acid sequence having the sequence of SEQ ID NOs: 16, 17, 18 or 19, or which differs from said sequence by one or more conservative amino acid modifications.
18. The method of claim 17, wherein the peptide consists of an amino acid having the sequence of SEQ ID NO: 19, or which differs from said sequence by one or more conservative modifications.
19. The method of claim 12, wherein the peptide consists of a contiguous sequence of 5 to 29 amino acids from SEQ ID NO. 1.
20. The method of claim 12, wherein the peptide or derivative comprises at least 3 amino acids from SEQ ID NO. 21, wherein the at least 3 amino acids from SEQ ID NO. 21 are selected from the group consisting of QPP, QPPK (SEQ ID NO: 24), and QPPKE (SEQ ID NO. 21).
Description
[0001] This application is a divisional of U.S. patent application Ser. No. 14/968,479, filed Jul. 12, 2011, which is a divisional of U.S. patent application Ser. No. 13/181,323, filed Jul. 12, 2011, which is a continuation of U.S. patent application Ser. No. 12/320,707, filed Feb. 2, 2009, which is a continuation-in-part of U.S. Patent Application No. 11/284,086, filed on Nov. 22, 2005, which claims priority from United Kingdom Patent Application No. 0426146.7, filed on Nov. 29, 2004, and Japanese Patent Application No. 2005-146848, filed on May 19, 2005. The contents of these applications are incorporated herein by reference in their entirety.
[0002] The content of the electronically submitted sequence listing in ASCII text file (Name: 3338_0800007_Seqlisting_ST25; Size: 13,792 bytes; and Date of Creation: Feb. 18, 2020) is incorporated herein by reference in its entirety.
[0003] The present invention relates to the field of immunology. More particularly, the present invention relates to inflammation and the use of proteins and peptides containing certain sequences of the TREM-1 protein and their functional equivalents (referred to herein as TREM1-peptides) in the treatment of disease, for example, sepsis, septic shock and inflammatory bowel disease (IBD).
[0004] Sepsis constitutes a significant consumption of intensive care resources and remains an ever-present problem in the intensive care unit. It has been estimated that between 400 000 and 500 000 patients are so affected each year in both the USA and Europe. Morbidity and mortality have remained high despite improvements in both supportive and antimicrobial therapies. Mortality rates vary from 40% for uncomplicated sepsis to 80% in those suffering from septic shock and multi-organ dysfunction. The pathogenesis of the conditions is now becoming better understood. Greater understanding of the complex network of immune, inflammatory and haematological mediators may allow the development of rational and novel therapies.
[0005] Following an infection, innate and cognitive immune responses develop in sequential phases that build-up in specificity and complexity, resulting ultimately in the clearance of infectious agents and restoration of homeostasis. The innate immune response serves as the first line of defence and is initiated upon activation of pattern recognition receptors, such as Toll-like receptors (TLRs) (1, 2), by various pathogen-associated microbial patterns (PAMPs) (3). Activation of the TLRs triggers the release of large quantities of such cytokines as TNF-.alpha. and IL-1.beta., which in case of such massive infections as sepsis, can precipitate tissue injury and lethal shock (4, 5). Although antagonists of TNF-.alpha. and IL-1.beta. appeared in this context as possibly interesting therapeutic agents of sepsis, they have unfortunately shown limited efficacy in clinical trials (6-8). This could be due to the fact that these cytokines are necessary for the clearance of infections, and that their removal would allow for fatal bacterial growth (9-11).
[0006] Another receptor involved in, inter alia, response to infection, triggering receptor expressed on myeloid cells-1 (TREM-1) is a member of a recently discovered family of receptors, the TREM family, expressed on the surface of neutrophils and a subset of monocytes. TREM receptors activate myeloid cells via association with the adaptor molecule DAP12. Engagement of TREM-1 has been reported to trigger the synthesis of pro-inflammatory cytokines in the presence of microbial products.
[0007] The triggering receptor expressed on myeloid cells (TREM)-1 is a recently discovered cell-surface molecule that has been identified both on human and murine polymorphonuclear neutrophils and mature monocytes (12). It belongs to the immunoglobulin superfamily and activates downstream signalling pathways with the help of an adapter protein called DAP12 (12-15). Bouchon and co-workers have shown that the expression of TREM-1 was greatly up-regulated on neutrophils and monocytes in the presence of such bacteria as Pseudomonas aeruginosa or Staphylococcus aureus, both in cell culture and in tissue samples from patients with infection (16). In striking contrast, TREM-1 was not up-regulated in samples from patients with non-infectious inflammatory diseases such as psoriasis, ulcerative colitis or vasculitis caused by immune complexes (16). Moreover, when TREM-1 is bound to its ligand, there is a synergistic effect of LPS and an amplified synthesis of the pro-inflammatory cytokines TNF-.alpha. and GM-CSF, together with an inhibition of IL-10 production (17). In a murine model of LPS-induced septic shock, blockade of TREM-1 signalling protected the animals from death, further highlighting the crucial role of this molecule (13, 16).
[0008] Recent studies demonstrate that TREM-1 plays a critical role in the inflammatory response to infection (see BOUCHON et al. (2000) J. Immunol. 164:4991-4995). Expression of TREM-1 is increased on myeloid cells in response to both bacterial and fungal infections in humans. Similarly, in mice the induction of shock by lipopolysaccharide (LPS) is associated with increased expression of TREM-1. Further, treatment of mice with a soluble TREM-1/Ig fusion protein, as a `decoy` receptor, protects mice from death due to LPS or E.coli.
[0009] Triggering via TREM-1 results in the production of pro-inflammatory cytokines, chemokines and reactive oxygen species, and leads to rapid degranulation of neutrophilic granules, and phagocytosis. Since interfering with TREM-1 engagement leads to the simultaneous reduction in production and secretion of a variety of proinflammatory mediators, TREM-1 represents an attractive target for treating chronic inflammatory disorders. Indeed, a role for TREM-1 has been demonstrated in a variety of inflammatory disorders, including (but not limited to) acute and chronic inflammatory disorders, sepsis, acute endotoxemia, encephalitis, Chronic Obstructive Pulmonary Disease (COPD), allergic inflammatory disorders, asthma, pulmonary fibrosis, pneumonia, Community acquired pneumonia (CAP), Ventilator associated pneumonia (VAP), Acute respiratory infection, Acute respiratory distress syndrome (ARDS), Infectious lung diseases, Pleural effusion, Peptic ulcer, Helicobacter pylori infection, hepatic granulomatosis, arthritis, rheumatoid arthritis, osteoarthritis, inflammatory osteolysis, ulcerative colitis, psoriasis, vasculitis, autoimmune disorders, thyroiditis, Meliodosis, (mesenteric) Ischemia reperfusion, Filovirus infection, Infection of the urinary tract, Bacterial meningitis, Salmonella enterica infection, Marburg and Ebola viruses infections, and in particular Inflammatory Bowel Disease (IBD).
[0010] Inflammatory bowel disease (IBD) covers a group of disorders in which the intestines become inflamed (red and swollen), probably as a result of an immune reaction of the body against its own intestinal tissue. Two major types of IBD are described: ulcerative colitis (UC) and Crohn disease (CD). Ulcerative colitis is limited to the colon (large intestine). Crohn disease can involve any part of the gastrointestinal tract from the mouth to the anus, but it most commonly affects the small intestine and/or the colon. Both ulcerative colitis and Crohn disease vary in the intensity and severity during the course of the disease. When there is severe inflammation, the disease is considered to be in an active stage, and the person experiences a flare-up of the condition. When the degree of inflammation is less (or absent), the person usually is without symptoms, and the disease is considered to be in remission. In IBD factor or factors trigger the body's immune system to produce an inflammatory reaction in the intestinal tract that continues without control. As a result of the inflammatory reaction, the intestinal wall is damaged leading to bloody diarrhea and abdominal pain.
[0011] U.S. Pat. No. 6,420,526 entitled "186 Secreted Proteins" claims unspecified and unexemplified isolated fragments of TREM-1 containing at least 30 contiguous amino acids of human TREM-1. No biological data relating to such fragments are provided.
[0012] As described in US2003165875A, fusion proteins between human IgG1 constant region and the extracellular domain of mouse TREM-1 or that of human TREM-1 show an effect against endotoxemia in mice.
[0013] The inventors have surprisingly found that certain peptides derived from the TREM-1 protein are capable of acting as antagonists of the TREM-1 protein and therefore have applications in the treatment of sepsis, septic shock and inflammatory bowel disease (IBD). The Inventors further demonstrate that the same peptides also modulate in vivo the pro-inflammatory cascade triggered by infection, thus inhibiting hyper-responsiveness and death in an animal model of sepsis, and that blocking TREM-1 attenuates the symptoms of IBD in mice.
[0014] Previously, the Inventors have identified a soluble form of TREM-1 (sTREM-1) and observed significant levels in serum samples from septic shock patients but not controls. As also described herein the Inventors have investigated its putative role in the modulation of inflammation during sepsis (see Gibot et al. (2004) Ann. Intern. Med. 141(1):9-15 and Gibot et al. (2004) N. Engl. J. Med. 350(5):451-8).
[0015] As described herein the Inventors show that a soluble form of TREM-1 (sTREM-1) is released in the peripheral blood during infectious aggression in mouse. The Inventors also confirm monocytes as a major source of sTREM, and show that synthetic peptides mimicking a part of the extra-cellular domain of TREM-1 can modulate cytokine production by activated monocytes in vitro.
[0016] The Inventors have observed that sTREM-1 is secreted by monocytes activated in vitro by LPS, as well as in the serum of animals involved in an experimental model of septic shock. Both in vitro and in vivo, synthetic peptides mimicking a short highly conserved domain of sTREM-1 attenuate cytokine production by human monocytes and protect septic animals from hyper-responsiveness and death. These peptides are efficient not only in preventing but also in down-regulating the deleterious effects of pro-inflammatory cytokines. These data demonstrate that in vivo modulation of TREM-1 by TREM-1 peptides is a valuable therapeutic tool for the treatment of infection, for example sepsis or septic shock or for the treatment of sepsis-like conditions.
[0017] Accordingly, the present invention provides methods and compositions for the treatment of infectious disease, in particular, sepsis and septic shock or for the treatment of sepsis-like conditions.
[0018] Other diseases or disorders that may also be treated by the methods and compositions of the present invention include any inflammatory disorder (or other disorder) that is mediated by the binding of the TREM-1 ligand to a TREM-1 receptor. Examples of inflammatory disorders include (but are not limited to) acute and chronic inflammatory disorders, sepsis, acute endotoxemia, encephalitis, Chronic Obstructive Pulmonary Disease (COPD), allergic inflammatory disorders, asthma, pulmonary fibrosis, pneumonia, Community acquired pneumonia (CAP), Ventilator associated pneumonia (VAP), Acute respiratory infection, Acute respiratory distress syndrome (ARDS), Infectious lung diseases, Pleural effusion, Peptic ulcer, Helicobacter pylori infection, hepatic granulomatosis, arthritis, rheumatoid arthritis, osteoarthritis, inflammatory osteolysis, ulcerative colitis, psoriasis, vasculitis, autoimmune disorders, thyroiditis, Meliodosis, (mesenteric) Ischemia reperfusion, Filovirus infection, Infection of the urinary tract, Bacterial meningitis, Salmonella enterica infection, Marburg and Ebola viruses infections, and in particular Inflammatory Bowel Disease (IBD).
[0019] As described herein, the Inventors have determined that several peptides of the extracellular portion of the TREM-1 protein (see Table 1), which incorporate sequences from "CDR2" and "CDR3" surprisingly have activity similar to previously described fusion proteins of IgG1 constant region and the extracellular domain of TREM-1 in models of sepsis. These peptides also have advantages over the protein particularly in terms of cost of manufacture.
[0020] Thus, the invention provides polypeptides comprising one or more sequences derived from CDR2 or CDR3 of a TREM-1 protein. Preferably, said polypeptides comprise less than 30 contiguous amino acids of said TREM-1 protein.
[0021] As shown in Table 1, examples of such peptides or polypeptides, contain or comprise for example 15-25 amino acid ("AA") peptides from the TREM-1 protein and contain or comprise all or part of a CDR domain (3-6 AAs) of the receptor flanked by natural sequences from the protein that can vary in length so long as function of the CDR-like domain is not lost. Such peptides are derived from the TREM-1 receptor protein amino acid sequence for example, as shown in Table 2 (human) and Table 3 (mouse).
[0022] Table 1 shows peptides derived from mouse TREM-1 "mPX" (NCBI Reference Sequences (RefSeq) NP_067381) or human TREM-1 "hPX''" (NCBI Reference Sequences (RefSeq) NP_061113). Underlined amino acids span the human TREM-1 Complementarity Determining Regions (CDR), as described by Radaev et al. 2003 Structure (Camb.) 11 (12), 1527-1535 (2003).
[0023] Table 2 shows the human TREM-1 amino acid sequence NP_061113. Underlined amino acids span the human TREM-1 Complementarity Determining Regions (CDR) 2 (RPSKNS; [SEQ ID NO:20]) and 3 (QPPKE [SEQ ID NO:21]), as described by Radaev et al. 2003 Structure (Camb.) 11 (12), 1527-1535 (2003).
[0024] Table 3 shows the mouse TREM-1 amino acid sequence NP_067381. Underlined amino acids span the mouse TREM-1 Complementarity Determining Regions (CDR) 2 (RPFTRP; [SEQ ID NO:22]) and 3 (HPPND; [SEQ ID NO:23]).
TABLE-US-00001 TABLE 1 Peptides including sequences from human and mouse TREM-1 CDR 2 and CDR 3 hCDR 2 mP1(67-89): LVVTQRPFTRPSEVHMGKFTLKH [SEQ ID NO: 3] hP1(67-89): LACTERPSKNSHPVQVGRIILED [SEQ ID NO: 16] hCDR 3 mP2(114-136): VIYHPPNDPVVLFHPVRLVVTKG [SEQ ID NO: 4] mP4(103-123): LQVTDSGLYRCVIYHPPNDPV [SEQ ID NO: 6] mP5(103-119): LQVTDSGLYRCVIYHPP [SEQ ID NO: 7] hP2(114-136): VIYQPPKEPHMLFDRIRLVVTKG [SEQ ID NO: 17] hP4(103-123): LQVEDSGLYQCVIYQPPKEPH [SEQ ID NO: 18] hP5(103-119): LQVEDSGLYQCVIYQPP [SEQ ID NO: 19]
TABLE-US-00002 TABLE 2 Human TREM-1 amino acid sequence NP_061113 1 MRKTRLWGLL WMLFVSELRA ATKLTEEKYE LKEGQTLDVK CDYTLEKFAS SQKAWQIIRD 61 GEMPKTLACT ERPSKNSHPV QVGRIILEDY HDHGLLRVRM VNLQVEDSGL YQCVIYQPPK 121 EPHMLFDRIR LVVTKGFSGT PGSNENSTQN VYKIPPTTTK ALCPLYTSPR TVTQAPPKST 181 ADVSTPDSEI NLTNVTDIIR VPVFNIVILL AGGFLSKSLV FSVLFAVTLR SFVP [SEQ ID NO: 1]
TABLE-US-00003 TABLE 3 Mouse TREM-1 amino acid sequence NP_067381 1 MRKAGLWGLL CVFFVSEVKA AIVLEEERYD LVEGQTLTVK CPFNIMKYAN SQKAWQRLPD 61 GKEPLTLVVT QRPFTRPSEV HMGKFTLKHD PSEAMLQVQM TDLQVTDSGL YRCVIYHPPN 121 DPVVLFHPVR LVVTKGSSDV FTPVIIPITR LTERPILITT KYSPSDTTTT RSLPKPTAVV 181 SSPGLGVTII NGTDADSVST SSVTISVICG LLSKSLVFII LFIVTKRTFG [SEQ ID NO: 2]
[0025] Accordingly, the invention provides isolated or recombinantly prepared polypeptides or peptides comprising or consisting essentially of one or more sequences derived from CDR2 or CDR3 of a TREM-1 protein, or fragments, homologues, derivatives, fusion proteins or variants of such polypeptides, as defined herein, which are herein collectively referred to as "polypeptides or peptides of the invention" or "TREM-1 peptides or TREM-1 polypeptides", preferably such entities comprise less than 30 contiguous amino acids of a TREM-1 protein, for example as shown in Table 2 or Table 3. Generally where polypeptides or proteins of the invention or fragments, homologues, derivatives, or variants thereof are intended for use (for example treatment) in a particular species, the sequences of CDR2 or CDR3 of a TREM-1 protein are chosen from the TREM-1 protein amino acid sequence of that species, or if the sequence is not known, an analogous species. For example, polypeptides or proteins of the invention for the treatment of human disease, in particular sepsis, septic shock or sepsis-like conditions, will comprise one or more sequences comprising all or part of CDR2 or CDR3 from the human TREM-1 protein.
[0026] Furthermore, the invention provides isolated polypeptides or proteins comprising an amino acid sequence that is at least about 60%, 70%, 75%, 80%, 85%, 90%, 95%, or 98% identical to the amino acid sequence of SEQ ID NO:20, 21, 22, 23 or fragments, homologues, derivatives, or variants thereof. The invention also provides isolated peptides, polypeptides or proteins comprising an amino acid sequence that comprises or consists of at least about 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, or 29 or more contiguous amino acids of a TREM-1 protein of which 3 or more contiguous amino acids are derived from the sequences of SEQ ID NO:20, 21, 22 or 23 (in other words a sequence representing all, or part of CDR2 or CDR3 of a TREM-1 protein is present in the peptide, polypeptide or protein), or fragments, homologues, derivatives, or variants thereof. In preferred embodiments, such peptides, polypeptides or proteins, or fragments, homologues, derivatives or variants thereof have a biological activity of a TREM-1 full-length protein, such as antigenicity, immunogenicity, triggering of proinflammatory chemokines and cytokines, mobilization of cytosolic Ca.sup.2+, protein tyrosine-phosphorylation, mediator release, and other activities readily assayable. Generally, such peptides, polypeptides or proteins or fragments, homologues, derivatives or variants thereof are capable of treating sepsis, septic shock or sepsis-like conditions, or are active in experimental models of sepsis, septic shock or sepsis-like conditions, for example by acting as antagonists of the activity of the TREM-1 receptor. Such peptides, polypeptides or proteins or fragments, homologues, derivatives or variants thereof are characterised by the ability to treat, ameliorate, or lessen the symptoms of sepsis, septic shock or sepsis-like conditions.
[0027] In particular, the invention provides, a TREM-1 polypeptide having activity against sepsis, septic shock or sepsis-like conditions which consists of (i) a contiguous sequence of 5 to 29, for example 15-25, amino acids corresponding to the native TREM-1 protein sequence which includes at least 3 amino acids from the CDR2 or CDR3 sequences; or (ii) such a sequence in which one or more (e.g. one, two or three) amino acids are substituted conservatively with another amino acid provided, however that at least 3 amino acids from the CDR2 or CDR3 sequences are not substituted; or (iii) a sequence of (i) or (ii) linked at one or both of its N and C termini to a heterologous polypeptide. For example, in a polypeptide wherein the native TREM-1 protein sequence is the human sequence identified as (SEQ ID NO: 1), the CDR2 and CDR3 sequences are RPSKNS (SEQ ID NO:20) and QPPKE (SEQ ID NO:21) respectively. In such polypeptides, the at least 3 amino acids from the CDR2 or CDR3 sequences can be QPP, PPK, PKE, RPS, PSK, SKN or KNS. Such polypeptides may comprise the sequence QPPK([SEQ ID NO:24), QPPKE (SEQ ID NO:21) or RPSKNS (SEQ ID NO:20). For example, in a polypeptide wherein the native TREM-1 protein sequence is the mouse sequence identified as (SEQ ID NO: 2) the CDR2 and CDR3 sequences are RPFTRP (SEQ ID NO:22) and HPPND (SEQ ID NO:23) respectively. In such polypeptides, the at least 3 amino acids from the CDR2 or CDR3 sequences can be HPP, PPN, PND, RPF, PFT, FTR or TRP. Such polypeptides may comprise the sequences HPP, HPPN (SEQ ID NO:25), HPPND (SEQ ID NO:23) or RPFTRP (SEQ ID NO:22).
[0028] In certain embodiments, the polypeptide of the invention is or comprises SEQ ID No. 7 which is disclosed in Gibot et al (2004) J Exp Med 200, 1419-1426.
[0029] In certain embodiments the polypeptide of the invention neither is nor comprises SEQ ID No. 7.
[0030] In certain embodiments the polypeptide of the invention is or comprises a sequence selected from SEQ ID Nos. 3, 4 and 6.
[0031] In certain embodiments the polypeptide of the invention is or comprises a sequence selected from SEQ ID Nos. 16, 17, 18 and 19.
[0032] In certain embodiments the polypeptide of the invention is or comprises a sequence derived from CDR2.
[0033] In certain embodiments the polypeptide of the invention is or comprises a sequence derived from CDR3.
[0034] The polypeptides or peptides of the invention are provided for use in therapy, in particular in the treatment of sepsis, septic shock and sepsis-like conditions, and for use in the manufacture of a medicament for the treatment of sepsis, septic shock and sepsis-like conditions. Further provided are compositions and pharmaceutical compositions containing polypeptides or peptides of the invention and methods of treatment of sepsis, septic shock and sepsis-like conditions using polypeptides or peptides of the invention. In addition the polypeptides or peptides of the invention are provided for use in therapy to restore haemodynamic parameters in sepsis, septic shock and sepsis-like conditions and for use in the manufacture of a medicament for the treatment of aberrant haemodynamic parameters in sepsis, septic shock and sepsis-like conditions.
[0035] The term "triggering receptor expressed on myeloid cells" or "TREM" refers to a group of activating receptors which are selectively expressed on different types of myeloid cells, such as mast cells, monocytes, macrophages, dendritic cells (DCs), and neutrophils, and may have a predominant role in immune and inflammatory responses. TREMs are primarily transmembrane glycoproteins with a Ig-type fold in their extracellular domain and, hence, belong to the Ig-SF. These receptors contain a short intracellular domain, but lack docking motifs for signaling mediators and require adapter proteins, such as DAP12, for cell activation.
[0036] The term "myeloid cells" as used herein refers to a series of bone marrow-derived cell lineages including granulocytes (neutrophils, eosinophils, and basophils), monocytes, macrophages, and mast cells. Furthermore, peripheral blood dendritic cells of myeloid origin, and dendritic cells and macrophages derived in vitro from monocytes in the presence of appropriate culture conditions, are also included.
[0037] The term "sepsis, septic shock" or "sepsis or septic shock" as defined herein, refers to sub-groups of systemic inflammatory response syndrome (SIRS). The term "sepsis" is generally reserved for SIRS when infection is suspected or proven. A pattern of physiological variables have been shown in critically ill patients in response to a range of insults including; trauma, burns, pancreatitis and infection. These include inflammatory responses, leucocytosis or severe leucopaenia, hyperthermia or hypothermia, tachycardia and tachypnoea and have been collectively termed the systemic inflammatory response syndrome (SIRS). This definition emphasises the importance of the inflammatory process in these conditions regardless of the presence of infection. Sepsis is further stratified into severe sepsis when there is evidence of organ hypoperfusion, made evident by signs of organ dysfunction such as hypoxaemia, oliguria, lactic acidosis or altered cerebral function. "Septic shock" is severe sepsis usually complicated by hypotension, defined in humans as systolic blood pressure less than 90 mmHg despite adequate fluid resuscitation. Sepsis and SIRS may be complicated by the failure of two or more organs, termed multiple organ failure (MOF), due to disordered organ perfusion and oxygenation. In addition to systemic effects of infection, a systemic inflammatory response may occur in severe inflammatory conditions such as pancreatitis and burns. The appearance of signs of an inflammatory response is less well defined following traumatic insults. In the intensive care unit, gram-negative bacteria are implicated in 50 to 60% of sepsis cases with gram-positive bacteria accounting for a further 35 to 40% of cases. The remainder of cases are due to the less common causes of fungi, viruses and protozoa.
[0038] The term "sepsis-like conditions" as used herein refers to those states in which a patient presents with symptoms similar to sepsis or septic shock but where an infectious agent is not the primary or initial cause of a similar cascade of inflammatory mediators and/or change in haemodynamic parameters as seen in cases of sepsis, for example in patients with acute or chronic liver failure (see Wasmuth H E, et al. J Hepatol. 2005 February; 42(2): 195-201), in cases of post-resuscitation disease after cardiac arrest (see Adrie C et al. Curr Opin Crit Care. 2004 June; 10(3):208-12) in the treatment of sepsis-like symptoms after cancer chemotherapy (seeTsuji E et al. Int J Cancer. 2003 Nov. 1; 107(2):303-8) in patients undergoing hyperthermic isolated limb perfusion with recombinant TNF-alpha or similar treatments (see Zwaveling J H et al. Crit Care Med. 1996 May; 24(5):765-70) or sepsis-like illness in neonates (see Griffin MP et al. Pediatr Res. 2003 June; 53(6):920-6).
[0039] The term "activity against sepsis, septic shock or sepsis-like conditions" as used herein refers to the capability of a molecule, for example a peptide, polypeptide or engineered antibody, to treat sepsis, septic shock or sepsis-like conditions, or be active in experimental models of sepsis, septic shock or sepsis-like conditions, for example by acting as an antagonist of the activity of the TREM-1 receptor.
[0040] Typically the indication for polypeptides of the invention is sepsis or septic-shock or Inflammatory Bowel Disease (IBD). Other indications may include any inflammatory disorder (or other disorder) that is mediated by the binding of the TREM-1 ligand to a TREM-1 receptor. Examples of inflammatory disorders include (but are not limited to) acute and chronic inflammatory disorders, sepsis, acute endotoxemia, encephalitis, Chronic Obstructive Pulmonary Disease (COPD), allergic inflammatory disorders, asthma, pulmonary fibrosis, pneumonia, Community acquired pneumonia (CAP), Ventilator associated pneumonia (VAP), Acute respiratory infection, Acute respiratory distress syndrome (ARDS), Infectious lung diseases, Pleural effusion, Peptic ulcer, Helicobacter pylori infection, hepatic granulomatosis, arthritis, rheumatoid arthritis, osteoarthritis, inflammatory osteolysis, ulcerative colitis, psoriasis, vasculitis, autoimmune disorders, thyroiditis, Meliodosis, (mesenteric) Ischemia reperfusion, Filovirus infection, Infection of the urinary tract, Bacterial meningitis, Salmonella enterica infection, Marburg and Ebola viruses infections.
[0041] The term "substantial sequence identity", when used in connection with peptides/amino acid sequences, refers to peptides/amino acid sequences which are substantially identical to or similar in sequence, giving rise to a homology in conformation and thus to similar biological activity. The term is not intended to imply a common evolution of the sequences.
[0042] Typically, peptides/amino acid sequences having "substantial sequence identity" are sequences that are at least 50%, more preferably at least 80%, identical in sequence, at least over any regions known to be involved in the desired activity. Most preferably, no more than five residues, other than at the termini, are different. Preferably, the divergence in sequence, at least in the aforementioned regions, is in the form of "conservative modifications".
[0043] To determine the percent sequence identity of two peptides/amino acid sequences or of two nucleic acid sequences, the sequences are aligned for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second amino acid or nucleic acid sequence for optimal alignment and non-homologous sequences can be disregarded for comparison purposes). For example, the length of a reference sequence aligned for comparison purposes is at least 30%, preferably at least 40%, more preferably at least 50%, even more preferably at least 60%, and even more preferably at least 70%, 80%, or 90% of the length of the reference sequence (e.g., when aligning a second sequence to the first amino acid sequence which has for example 100 amino acid residues, at least 30, preferably at least 40, more preferably at least 50, even more preferably at least 60, and even more preferably at least 70, 80 or 90 amino acid residues are aligned). The amino acid residues or nucleotides at corresponding amino acid positions or nucleotide positions are then compared. When a position in the first sequence is occupied by the same amino acid residue or nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position (as used herein amino acid or nucleic acid "identity" is equivalent to amino acid or nucleic acid "homology"). The percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which need to be introduced for optimal alignment of the two sequences. The comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm. In one embodiment, the percent identity between two amino acid sequences is determined using the Needleman and Wunsch (J. Mol. Biol. (48):444-453 (1970)) algorithm which has been incorporated into the GAP program in the GCG software package (available online at the GCG website), using either a Blossom 62 matrix or a PAM250 matrix, and a gap weight of 16, 14, 12, 10, 8, 6, or 4, and a length weight of 1, 2, 3, 4, 5, or 6. In another embodiment, the percent identity between two nucleotide sequences is determined using the GAP program in the GCG software package (available online at the GCG website), using a NWSgapdna.CMP matrix and a gap weight of 40, 50, 60, 70, or 80, and a length weight of 1, 2, 3, 4, 5, or 6. In another embodiment, the percent identity between two amino acid or nucleotide sequences is determined using the algorithm of E. Meyers and W. Miller (CABIOS, 4:11-17 (1989)) which has been incorporated into the ALIGN program (version 2.0), using a PAM120 weight residue table, a gap length penalty of 12, and a gap penalty of 4. The nucleic acid and protein sequences of the present invention can further be used as a "query sequence" to perform a search against public databases to identify, for example, other family members or related sequences. Such searches can be performed using the NBLAST and XBLAST programs (version 2.0) of Altschul, et al. (1990) J. Mol. Biol. 215:403-10. BLAST nucleotide searches can be performed with the NBLAST program, score=100, wordlength=12 to obtain nucleotide sequences homologous to NIP2b, NIP2cL, and NIP2cS nucleic acid molecules of the invention. BLAST protein searches can be performed with the XBLAST program, score=50, wordlength=3 to obtain amino acid sequences homologous to NIP2b, NIP2cL, and NIP2cS protein molecules of the invention. To obtain gapped alignments for comparison purposes, Gapped BLAST can be utilized as described in Altschul et al., (1997) Nucleic Acids Res. 25(17):3389-3402. When utilizing BLAST and Gapped BLAST programs, the default parameters of the respective programs (e.g., XBLAST and NBLAST) can be used. These programs are publically available on the National Center for Biotechnology Information ("NCBI") website.
[0044] The terms "protein" and "polypeptide" are used interchangeably herein. The term "peptide" is used herein to refer to a chain of two or more amino acids or amino acid analogues (including non-naturally occurring amino acids), with adjacent amino acids joined by peptide (--NHCO--) bonds. Thus, the peptides of the invention include oligopeptides, polypeptides, proteins, mimetopes and peptidomimetics. Methods for preparing mimetopes and peptidomimetics are known in the art.
[0045] The terms "mimetope" and "peptidomimetic" are used interchangeably herein. A "mimetope" of a compound X refers to a compound in which chemical structures of X necessary for functional activity of X have been replaced with other chemical structures which mimic the conformation of X. Examples of peptidomimetics include peptidic compounds in which the peptide backbone is substituted with one or more benzodiazepine molecules (see e.g., James, G. L. et al. (1993) Science 260:1937-1942) and "retro-inverso" peptides (see U.S. Pat. No. 4,522,752 to Sisto). The terms "mimetope" and "peptidomimetic" also refer to a moiety, other than a naturally occurring amino acid, that conformationally and functionally serves as a substitute for a particular amino acid in a peptide-containing compound without adversely interfering to a significant extent with the function of the peptide. Examples of amino acid mimetics include D-amino acids. Peptides substituted with one or more D-amino acids may be made using well known peptide synthesis procedures. Additional substitutions include amino acid analogues having variant side chains with functional groups, for example, b-cyanoalanine, canavanine, djenkolic acid, norleucine, 3-phosphoserine, homoserine, dihydroxyphenylalanine, 5-hydroxytryptophan, 1-methylhistidine, or 3-methylhistidine.
[0046] As used herein an "analogue" of a compound X refers to a compound which retains chemical structures of X necessary for functional activity of X, yet which also contains certain chemical structures which differ from X. An example of an analogue of a naturally-occurring peptide is a peptide which includes one or more non-naturally-occurring amino acids. The term "analogue" is also intended to include modified mimetopes and/or peptidomimetics, modified peptides and polypeptides, and allelic variants of peptides and polypeptides. Analogues of a peptide will therefore produce a peptide analogue that is substantially homologous or, in other words, has substantial sequence identity to the original peptide. The term "amino acid" includes its art recognized meaning and broadly encompasses compounds of formula I:
##STR00001##
[0047] Preferred amino acids include the naturally occurring amino acids, as well as synthetic derivatives, and amino acids derived from proteins, e.g., proteins such as casein, i.e., casamino acids, or enzymatic or chemical digests of, e.g., yeast, an animal product, e.g., a meat digest, or a plant product, e.g., soy protein, cottonseed protein, or a corn steep liquor (see, e.g., Traders' Guide to Fermentation Media, Traders Protein, Memphis, Tenn. (1988), Biotechnology: A Textbook of Industrial Microbiology, Sinauer Associates, Sunderland, Mass. (1989), and Product Data Sheet for Corn Steep Liquor, Grain Processing Corp., IO).
[0048] The term "naturally occurring amino acid" includes any of the 20 amino acid residues which commonly comprise most polypeptides in living systems, rarer amino acids found in fibrous proteins (e.g., 4-hydorxyproline, 5-hydroxylysine, --N-methyllysine, 3-methylhistidine, desmosine, isodesmosine), and naturally occurring amino acids not found in proteins (e.g., -aminobutyric acid, homocysteine, homoserine, citrulline, ornithine, canavanine, djenkolic acid, and -cyanoalanine).
[0049] The term "side chain of a naturally occurring amino acid" is intended to include the side chain of any of the naturally occurring amino acids, as represented by R in formula I. One skilled in the art will understand that the structure of formula I is intended to encompass amino acids such as proline where the side chain is a cyclic or heterocyclic structure (e.g., in proline R group and the amino group form a five-membered heterocyclic ring.
[0050] The term "homologue," as used herein refers to any member of a series of peptides or polypeptides having a common biological activity, including antigenicity/immunogenicity and inflammation regulatory activity, and/or structural domain and having sufficient amino acid as defined herein. Such homologues can be from either the same or different species of animals.
[0051] The term "variant" as used herein refers either to a naturally occurring allelic variation of a given peptide or a recombinantly prepared variation of a given peptide or protein in which one or more (e.g. one, two or three) amino acid residues have been modified by amino acid substitution, addition, or deletion.
[0052] The term "derivative" as used herein refers to a variation of given peptide or protein that are otherwise modified, i.e., by covalent attachment of any type of molecule, preferably having bioactivity, to the peptide or protein, including non-naturally occurring amino acids.
[0053] Preferably, such homologues, variants and derivatives are capable of treating sepsis, septic shock or sepsis-like conditions, or are active in experimental models of sepsis, septic shock or sepsis-like conditions, or are capable of treating IBD or other inflammatory disorder, for example by acting as antagonists of the activity of the TREM-1 receptor.
[0054] An "isolated" or "purified" peptide or protein is substantially free of cellular material or other contaminating proteins from the cell or tissue source from which the protein is derived, or substantially free of chemical precursors or other chemicals when chemically synthesized.
[0055] The language "substantially free of cellular material" includes preparations of a polypeptide/protein in which the polypeptide/protein is separated from cellular components of the cells from which it is isolated or recombinantly produced. Thus, a polypeptide/protein that is substantially free of cellular material includes preparations of the polypeptide/protein having less than about 30%, 20%, 10%, 5%, 2.5%, or 1%, (by dry weight) of contaminating protein. When the polypeptide/protein is recombinantly produced, it is also preferably substantially free of culture medium, i.e., culture medium represents less than about 20%, 10%, or 5% of the volume of the protein preparation. When polypeptide/protein is produced by chemical synthesis, it is preferably substantially free of chemical precursors or other chemicals, i.e., it is separated from chemical precursors or other chemicals which are involved in the synthesis of the protein. Accordingly, such preparations of the polypeptide/protein have less than about 30%, 20%, 10%, 5% (by dry weight) of chemical precursors or compounds other than polypeptide/protein fragment of interest. In a preferred embodiment of the present invention, polypeptides/proteins are isolated or purified.
[0056] In addition to the polypeptides described above, polypeptides of the invention also encompass those polypeptides having a common biological activity and/or structural domain and having sufficient amino acid identity (homologues) as defined herein. These homologues can be from either the same or different species of animal, preferably from mammals, more preferably from rodents, such as mouse and rat, and most preferably from human. Preferably, they exhibit at least one structural and/or functional feature of TREM-1, and are preferably, capable of treating sepsis, septic shock or sepsis-like conditions, for example by acting as antagonists of the activity of the TREM-1 receptor. Such modifications include amino acid substitution, deletion, and/or insertion. Amino acid modifications can be made by any method known in the art and various methods are available to and routine for those skilled in the art.
[0057] Additionally, in making amino acid substitutions, generally the amino acid residue to be substituted can be a conservative amino acid substitution (i.e. "substituted conservatively"), for example, a polar residue is substituted with a polar residue, a hydrophilic residue with a hydrophilic residue, hydrophobic residue with a hydrophobic residue, a positively charged residue with a positively charged residue, or a negatively charged residue with a negatively charged residue. Moreover, generally, the amino acid residue to be modified is not highly or completely conserved across species and/or is critical to maintain the biological activities of the peptide and/or the protein it derives from.
[0058] Peptides of the invention may be directly synthesised in any convenient way. Generally the reactive groups present (for example amino, thiol and/or carboxyl) will be protected during overall synthesis. A proportion of the peptides of the invention, i. e. those wherein the comprised amino acids are genetically coded amino acids, will be capable of being expressed in prokaryotic and eukaryotic hosts by expression systems well known to the man skilled in the art. Methods for the isolation and purification of e. g. microbially expressed peptides are also well known. Polynucleotides which encode these peptides of the invention constitute further aspects of the present invention. As used herein, "polynucleotide" refers to a polymer of deoxyribonucleotides or ribonucleotides, in the form of a separate fragment or as a component of a larger construct, e. g. an expression vector such as a plasmid. Polynucleotide sequences of the invention include DNA, RNA and cDNA sequences. Due to the degeneracy of the genetic code, of course more than one polynucleotide is capable of encoding a particular peptide according to the invention. When a bacterial host is chosen for expression of a peptide, it may be necessary to take steps to protect the host from the expressed anti-bacterial peptide. Such techniques are known in the art and include the use of a bacterial strain which is resistant to the particular peptide being expressed or the expression of a fusion peptide with sections at one or both ends which disable the antibiotic activity of the peptide according to the invention. In the latter case, the peptide can be cleaved after harvesting to produce the active peptide. If the peptide incorporates a chemical modification then the activity/stability of the expressed peptide may be low, and is only modulated by post-synthetic chemical modification.
[0059] Furthermore, the invention also encompasses derivatives of the polypeptides of the invention. For example, but not by way of limitation, derivatives may include peptides or proteins that have been modified, e.g., by glycosylation, acetylation, pegylation, phosphorylation, amidation, derivatization by known protecting/blocking groups, proteolytic cleavage, linkage to a cellular ligand or other protein, etc. Any of numerous chemical modifications may be carried out by known techniques, including, but not limited to, specific chemical cleavage, acetylation, formylation, etc. Additionally, the derivative may contain one or more non-classical amino acids. Those skilled in the art will be aware of various methods for modifying peptides to increase potency, prolong activity and/or increase half-life. In one example (WO0210195) the modification is made via coupling through an amide bond with at least one conformationally rigid substituent, either at the N-terminal of the peptide, the C-terminal of the peptide, or on a free amino or carboxyl group along the peptide chain. Other examples of peptide modifications with similar effects are described, for example, in WO2004029081, WO03086444, WO03049684, WO0145746, WO0103723 and WO9101743.
[0060] The invention further provides antibodies that comprise a peptide or polypeptide of the invention or that mimic the activity of peptides or polypeptides of the invention. Such antibodies include, but are not limited to: polyclonal, monoclonal, bi-specific, multi-specific, human, humanized, chimeric antibodies, single chain antibodies, Fab fragments, F(ab')2 fragments, disulfide-linked Fvs, and fragments containing either a VL or VH domain or even a complementary determining region (CDR) that specifically binds to a polypeptide of the invention. In another embodiment, antibodies can also be generated using various phage display methods known in the art. Techniques to recombinantly produce Fab, Fab' and F(ab')2 fragments can also be employed using methods known in the art such as those disclosed in PCT publication WO 92/22324; Mullinax, et al., BioTechniques, 12(6):864-869, 1992; and Sawai, et al., 1995, AJRI 34:26-34; and Better, et al., 1988, Science 240:10411043 (each of which is incorporated by reference in its entirety). Examples of techniques that can be used to produce single-chain Fvs and antibodies include those described in U.S. Pat. Nos. 4,946,778 and 5,258,498; Huston, et al., 1991, Methods in Enzymology 203:4688; Shu, et al., 1993, Proc. Natl. Acad. Sci. USA 90:7995-7999; and Skerra, et al., 1988, Science 240:1038-1040. For some uses, including in vivo use of antibodies in humans and in vitro detection assays, it may be preferable to use chimeric, humanized, or human antibodies. A chimeric antibody is a molecule in which different portions of the antibody are derived from different animal species, such as antibodies having a variable region derived from a murine monoclonal antibody and a constant region derived from a human immunoglobulin. Methods for producing chimeric antibodies are known in the art. See, e.g., Morrison, 1985, Science 229:1202; Oi, et al., 1986, BioTechniques 4:214; Gillies, et al., 1989, J. Immunol. Methods 125:191-202; U.S. Pat. Nos. 5,807,715; 4,816,567; and 4,816,397; which are incorporated herein by reference in their entireties. Humanized antibodies are antibody molecules from non-human species that bind the desired antigen having one or more complementarity determining regions (CDRs) from the non-human species and framework regions from a human immunoglobulin molecule or in the case of the present invention, one or more CDRs derived from a TREM-1 protein. As known in the art, framework residues in the human framework regions can be substituted with the corresponding residue from the CDR donor antibody to alter, preferably improve, antigen binding. These framework substitutions are identified by methods well known in the art, e.g., by modelling of the interactions of the CDR and framework residues to identify framework residues important for antigen binding and sequence comparison to identify unusual framework residues at particular positions. See, e.g., Queen, et al., U.S. Pat. No. 5,585,089; Riechmann, et al., 1988, Nature 332:323, 1988, which are incorporated herein by reference in their entireties. Antibodies can be humanized using a variety of techniques known in the art including, for example, CDR-grafting (EP 239,400; PCT publication WO 91/09967; U.S. Pat. Nos. 5,225,539; 5,530,101 and 5,585,089), veneering or resurfacing (EP 592,106; EP 519,596; Padlan, 1991, Molecular Immunology, 28(4/5):489-498; Studnicka, et al., 1994, Protein Engineering, 7(6):805-814; Roguska, et al., 1994, Proc Natl. Acad. Sci. USA 91:969-973, and chain shuffling (U.S. Pat. No. 5,565,332), all of which are hereby incorporated by reference in their entireties.
[0061] Completely human antibodies are particularly desirable for therapeutic treatment of human patients. Human antibodies can be made by a variety of methods known in the art including phage display methods described above using antibody libraries derived from human immunoglobulin sequences. See U.S. Pat. Nos. 4,444,887 and 4,716,111; and PCT publications WO 98/46645; WO 98/50433; WO 98/24893; WO 98/16654; WO 96/34096; WO 96/33735; and WO 91/10741, each of which is incorporated herein by reference in its entirety. Human antibodies can also be produced using transgenic mice (see Lonberg and Huszar (1995), Int. Rev. Immunol. 13:65-93). For a detailed discussion of this technology for producing human antibodies and human monoclonal antibodies and protocols for producing such antibodies, see, e.g., PCT publications WO 98/24893; WO 92/01047; WO 96/34096; WO 96/33735; European Patent No. 0 598 877; U.S. Pat. Nos. 5,413,923; 5,625,126; 5,633,425; 5,569,825; 5,661,016; 5,545,806; 5,814,318; 5,885,793; 5,916,771; and 5,939,598; which are incorporated by reference herein in their entireties. In addition, companies such as Abgenix, Inc. (Freemont, Calif.), Medarex (N.J.) and Genpharm (San Jose, Calif.) can be engaged to provide human antibodies directed against a selected antigen using technology similar to that described above. Completely human antibodies which recognize a selected epitope can be generated using a technique referred to as "guided selection." In this approach a selected non-human monoclonal antibody, e.g., a mouse antibody, is used to guide the selection of a completely human antibody recognizing the same epitope. (Jespers et al., 1988, Bio/technology 12:899-903). Antibodies fused or conjugated to heterologous polypeptides may be used in in vitro immunoassays and in purification methods (e.g., affinity chromatography) well known in the art. See, e.g., PCT publication Number WO 93/21232; EP 439,095; Naramura, et al., 1994, Immunol. Lett. 39:91-99; U.S. Pat. No. 5,474,981; Gillies, et al., 1992 Proc. Natl. Acad. Sci. USA 89:1428-1432; and Fell, et al., 1991, J. Immunol. 146:2446-2452, which are incorporated herein by reference in their entireties.
[0062] In another aspect, the present invention provides methods for identifying a compound or ligand that binds to or modulates the activity of a polypeptide of the invention. Such a method comprises measuring a biological activity of the polypeptide in the presence or absence of a test compound and identifying test compounds that alter (increase or decrease) the biological activity of the polypeptide.
[0063] In one embodiment, the invention provides a fusion protein comprising a bioactive molecule and one or more domains of a polypeptide of the invention or fragment thereof. In particular, the present invention provides fusion proteins comprising a bioactive molecule recombinantly fused or chemically conjugated (including both covalent and non-covalent conjugations) to one or more domains of a polypeptide of the invention or fragments thereof.
[0064] The present invention further encompasses fusion proteins in which the polypeptides of the invention or fragments thereof, are recombinantly fused or chemically conjugated (including both covalent and non-covalent conjugations) to heterologous polypeptides (i.e., an unrelated polypeptide or portion thereof, preferably at least 10, at least 20, at least 30, at least 40, at least 50, at least 60, at least 70, at least 80, at least 90 or at least 100 amino acids of the polypeptide) to generate fusion proteins. The fusion does not necessarily need to be direct, but may occur through linker sequences.
[0065] In one example, a fusion protein in which a polypeptide of the invention or a fragment thereof can be fused to sequences derived from various types of immunoglobulins. For example, a polypeptide of the invention can be fused to a constant region (e.g., hinge, CH2, and CH3 domains) of human IgG1 or IgM molecule, (for example, as described by Hudson & Souriauso (2003) Nature Medicine 9(1):129-134) so as to make the fused polypeptides or fragments thereof more soluble and stable in vivo. The short half-life of antibody fragments can also be extended by `pegylation`, that is, a fusion to polyethylene glycol (see Leong, S. R. et al. (2001) Cytokine 16:106-119). In one example of such fusions, described in WO0183525, Fc domains are fused with biologically active peptides. A pharmacologically active compound is produced by covalently linking an Fc domain to at least one amino acid of a selected peptide. Linkage to the vehicle increases the half-life of the peptide, which otherwise could be quickly degraded in vivo.
[0066] Alternatively, non-classical alternative protein scaffolds (for example see Nygren & Skerra (2004) J Immunol Methods 290(1-2):3-28 or WO03049684) can be used to incorporate, and replicate the properties of, the peptides of the invention, for example by inserting peptide sequences derived from TREM-1 CDR2 or CDR3 into a protein framework to support conformationally variable loops having structural/functional similarities to CDR2 or CDR3 in a fixed spatial arrangement.
[0067] Such fusion proteins or scaffold based proteins can be used as an immunogen for the production of specific antibodies which recognize the polypeptides of the invention or fragments thereof. In another preferred embodiment, such fusion proteins or scaffold based proteins can be administered to a subject so as to inhibit interactions between a ligand and its receptors in vivo. Such inhibition of the interaction will block or suppress certain cellular responses involved in sepsis and septic shock.
[0068] In one aspect, the fusion protein comprises a polypeptide of the invention which is fused to a heterologous signal sequence at its N-terminus. Various signal sequences are commercially available. For example, the secretory sequences of melittin and human placental alkaline phosphatase (Stratagene; La Jolla, Calif.) are available as eukaryotic heterologous signal sequences. As examples of prokaryotic heterologous signal sequences, the phoA secretory signal (Sambrook, et al., supra; and Current Protocols in Molecular Biology, 1992, Ausubel, et al., eds., John Wiley & Sons) and the protein A secretory signal (Pharmacia Biotech; Piscataway, N.J.) can be listed. Another example is the gp67 secretory sequence of the baculovirus envelope protein (Current Protocols in Molecular Biology, 1992, Ausubel, et al., eds., John Wiley & Sons).
[0069] In another embodiment, a polypeptide of the invention can be fused to tag sequences, e.g., a hexa-histidine peptide, such as the tag provided in a pQE vector (QIAGEN, Inc., 9259 Eton Avenue, Chatsworth, Calif., 91311), among others, many of which are commercially available. As described in Gentz, et al., 1989, Proc. Natl. Acad. Sci. USA 86:821-824, for instance, hexa-histidine provides for convenient purification of the fusion protein. Other examples of peptide tags are the hemagglutinin "HA" tag, which corresponds to an epitope derived from the influenza hemagglutinin protein (Wilson, et al., 1984, Cell 37:767) and the "flag" tag (Knappik, et al., 1994, Biotechniques 17(4):754-761). These tags are especially useful for purification of recombinantly produced polypeptides of the invention.
[0070] Fusion proteins can be produced by standard recombinant DNA techniques or by protein synthetic techniques, e.g., by use of a peptide synthesizer. For example, a nucleic acid molecule encoding a fusion protein can be synthesized by conventional techniques including automated DNA synthesizers. Alternatively, PCR amplification of gene fragments can be carried out using anchor primers which give rise to complementary overhangs between two consecutive gene fragments which can subsequently be annealed and reamplified to generate a chimeric gene sequence (see, e.g., Current Protocols in Molecular Biology, 1992, Ausubel, et al., eds., John Wiley & Sons). The nucleotide sequence coding for a fusion protein can be inserted into an appropriate expression vector, i.e., a vector which contains the necessary elements for the transcription and translation of the inserted protein-coding sequence. Various host-vector systems and selection systems are known. In a specific embodiment, the expression of a fusion protein is regulated by a constitutive promoter. In another embodiment, the expression of a fusion protein is regulated by an inducible promoter. In accordance with these embodiments, the promoter may be a tissue-specific promoter. Expression vectors containing inserts of a gene encoding a fusion protein can be identified by three general approaches: (a) nucleic acid hybridization, (b) presence or absence of "marker" gene functions, and (c) expression of inserted sequences. In the first approach, the presence of a gene encoding a fusion protein in an expression vector can be detected by nucleic acid hybridization using probes comprising sequences that are homologous to an inserted gene encoding the fusion protein. In the second approach, the recombinant vector/host system can be identified and selected based upon the presence or absence of certain "marker" gene functions (e.g., thymidine kinase activity, resistance to antibiotics, transformation phenotype, occlusion body formation in baculovirus, etc.) caused by the insertion of a nucleotide sequence encoding a fusion protein in the vector. For example, if the nucleotide sequence encoding the fusion protein is inserted within the marker gene sequence of the vector, recombinants containing the gene encoding the fusion protein insert can be identified by the absence of the marker gene function. In the third approach, recombinant expression vectors can be identified by assaying the gene product (i.e., fusion protein) expressed by the recombinant. Such assays can be based, for example, on the physical or functional properties of the fusion protein in in vitro assay systems, e.g., binding with anti-fusion protein antibody. For long-term, high-yield production of recombinant proteins, stable expression is preferred. For example, cell lines which stably express the fusion protein may be engineered. Rather than using expression vectors which contain viral origins of replication, host cells can be transformed with DNA controlled by appropriate expression control elements (e.g., promoter, enhancer, sequences, transcription terminators, polyadenylation sites, etc.), and a selectable marker. Following the introduction of the foreign DNA, engineered cells may be allowed to grow for 1-2 days in an enriched medium, and then are switched to a selective medium. The selectable marker in the recombinant plasmid confers resistance to the selection and allows cells to stably integrate the plasmid into their chromosomes and grow to form foci which in turn can be cloned and expanded into cell lines. This method may advantageously be used to engineer cell lines that express the differentially expressed or pathway gene protein. Such engineered cell lines may be particularly useful in screening and evaluation of compounds that affect the endogenous activity of the differentially expressed or pathway gene protein. Once a fusion protein of the invention has been produced by recombinant expression, it may be purified by any method known in the art for purification of a protein, for example, by chromatography (e.g., ion exchange, affinity, particularly by affinity for the specific antibody, and sizing column chromatography), centrifugation, differential solubility, or by any other standard technique for the purification of proteins.
[0071] The present invention also provides methods for treating a subject suffering from sepsis, septic shock or a sepsis-like condition by administering a peptide or polypeptide of the invention. In another embodiment, the modulator may be an antibody which mimics the activity of a polypeptide of the invention. In particular, the invention provides a method of treating or ameliorating sepsis, septic shock or a sepsis-like condition in a subject, comprising: administering a therapeutically effective amount of a peptide or polypeptide of any one of the preceding claims to a subject. In such methods, the peptide or polypeptide administered can have substantial sequence identity to sequence SEQ ID NOS: 3, 4, 6, 7, 16, 17, 18 or 19, is SEQ ID NOS: 3, 4, 6, 7, 16, 17, 18 or 19, or an active fragment, analogue or derivative of SEQ ID NOS: 3, 4, 6, 7, 16, 17, 18 or 19 or has at least about 80% sequence identity to SEQ ID NOS: 3, 4, 6, 7, 16, 17, 18 or 19.
[0072] In one aspect, the invention provides a method for preventing sepsis, septic shock or sepsis-like conditions, by administering to the subject a peptide or polypeptide of the invention. Subjects at risk of sepsis or septic shock can be identified by, for example, any diagnostic or prognostic assays as known in the art (for particularly suitable methods of diagnosis, see WO2004081233, Gibot et al. (2004) Ann Intern Med. 141 (1):9-15 and Gibot et al. (2004) N Engl J Med. 350(5):451-8. The prophylactic agents described herein, for example, can be used to treat a subject at risk of developing disorders such as those previously discussed. The methods of the invention are applicable to mammals, for example humans, non human primates, sheep, pigs, cows, horses, goats, dogs, cats and rodents, such as mouse and rat. Generally, the methods of the invention are to be used with human subjects.
[0073] Furthermore, the invention provides a pharmaceutical composition comprising a polypeptide of the present invention or an antibody or fragments thereof that mimics a polypeptide of the invention. The peptides, polypeptides and antibodies (also referred to herein as "active compounds") of the invention can be incorporated into pharmaceutical compositions suitable for administration. Such compositions typically comprise the peptide, protein, or antibody and a pharmaceutically acceptable carrier.
[0074] As used herein the language "pharmaceutically acceptable diluent, carrier or excipient" is intended to include any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like, compatible with pharmaceutical administration. The use of such media and agents for pharmaceutically active substances is well known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the compositions is contemplated. Supplementary active compounds can also be incorporated into the compositions.
[0075] The invention includes methods for preparing pharmaceutical compositions containing a peptide or polypeptide of the invention. Such compositions can further include additional active agents. Thus, the invention further includes methods for preparing a pharmaceutical composition by formulating a pharmaceutically acceptable carrier with a peptide or polypeptide of the invention and one or more additional active compounds.
[0076] A pharmaceutical composition of the invention is formulated to be compatible with its intended route of administration. Examples of routes of administration include parenteral, e.g., intravenous, intradermal, subcutaneous, transdermal (topical), transmucosal, intraarticular, intraperitoneal, and intrapleural, as well as oral, inhalation, and rectal administration. Solutions or suspensions used for parenteral, intradermal, or subcutaneous application can include the following components: a sterile diluent such as water for injection, saline solution, fixed oils, polyethylene glycols, glycerine, propylene glycol or other synthetic solvents; antibacterial agents such as benzyl alcohol or methyl parabens; antioxidants such as ascorbic acid or sodium bisulfite; chelating agents such as ethylenediaminetetraacetic acid; buffers such as acetates, citrates or phosphates and agents for the adjustment of tonicity such as sodium chloride or dextrose. pH can be adjusted with acids or bases, such as hydrochloric acid or sodium hydroxide. The parenteral preparation can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
[0077] Pharmaceutical compositions suitable for injectable use include sterile aqueous solutions (where water soluble) or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersions. For intravenous administration, suitable carriers include physiological saline, bacteriostatic water, Cremophor EL.TM. (BASF; Parsippany, N.J.) or phosphate buffered saline (PBS). In all cases, the composition must be sterile and should be fluid to the extent that easy injectability with a syringe exists. It must be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms such as bacteria and fungi. The carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyetheylene glycol, and the like), and suitable mixtures thereof. The proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. Prevention of the action of microorganisms can be achieved by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, ascorbic acid, thimerosal, and the like. In many cases, it will be preferable to include isotonic agents, for example, sugars, polyalcohols such as mannitol, sorbitol, sodium chloride in the composition. Prolonged absorption of the injectable compositions can be brought about by including in the composition an agent which delays absorption, for example, aluminum monostearate and gelatin.
[0078] Sterile injectable solutions can be prepared by incorporating the active compound (e.g., a polypeptide or antibody) in the required amount in an appropriate solvent with one or a combination of ingredients enumerated above, as required, followed by filtered sterilization. Generally, dispersions are prepared by incorporating the active compound into a sterile vehicle which contains a basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum drying and freeze-drying which yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
[0079] Oral compositions generally include an inert diluent or an edible carrier. They can be enclosed in gelatin capsules or compressed into tablets. For the purpose of oral therapeutic administration, the active compound can be incorporated with excipients and used in the form of tablets, troches, or capsules. Pharmaceutically compatible binding agents, and/or adjuvant materials can be included as part of the composition. The tablets, pills, capsules, troches and the like can contain any of the following ingredients, or compounds of a similar nature: a binder such as microcrystalline cellulose, gum tragacanth or gelatin; an excipient, such as starch or lactose; a disintegrating agent, such as alginic acid, Primogel, or corn starch; a lubricant, such as magnesium stearate or Sterotes; a glidant, such as colloidal silicon dioxide; a sweetening agent, such as sucrose or saccharin; or a flavoring agent, such as peppermint, methyl salicylate, or orange flavoring.
[0080] For administration by inhalation, the compounds are delivered in the form of an aerosol spray from a pressurized container or dispenser which contains a suitable propellant, e.g., a gas such as carbon dioxide, or a nebulizer.
[0081] Systemic administration can also be by transmucosal or transdermal means. For transmucosal or transdermal administration, penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art, and include, for example, for transmucosal administration, detergents, bile salts, and fusidic acid derivatives. Transmucosal administration can be accomplished through the use of nasal sprays or suppositories. For transdermal administration, the active compounds are formulated into ointments, salves, gels, or creams as generally known in the art. The compounds can also be prepared in the form of suppositories (e.g., with conventional suppository bases such as cocoa butter and other glycerides) or retention enemas for rectal delivery.
[0082] In one embodiment, the active compounds are prepared with carriers that will protect the compound against rapid elimination from the body, such as a controlled release formulation, including implants and microencapsulated delivery systems. Biodegradable, biocompatible polymers can be used, such as ethylene vinyl acetate, polyanhydrides, polyglycolic acid, collagen, polyorthoesters, and polylactic acid. Methods for preparation of such formulations will be apparent to those skilled in the art. The materials can also be obtained commercially from Alza Corporation and Nova Pharmaceuticals, Inc. Liposomal suspensions (including liposomes targeted to infected cells with monoclonal antibodies to viral antigens) can also be used as pharmaceutically acceptable carriers. These can be prepared according to methods known to those skilled in the art, for example, as described in U.S. Pat. No. 4,522,811.
[0083] It is especially advantageous to formulate oral or parenteral compositions in dosage unit form for ease of administration and uniformity of dosage. Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the subject to be treated; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier. The specification for the dosage unit forms of the invention are dictated by and directly dependent on the unique characteristics of the active compound and the particular therapeutic effect to be achieved, and the limitations inherent in the art of compounding such an active compound for the treatment of individuals.
[0084] As defined herein, a therapeutically effective amount of protein or polypeptide (i.e., an effective dosage) ranges from about 0.001 to 30 mg/kg body weight, preferably about 0.01 to 25 mg/kg body weight, more preferably about 0.1 to 20 mg/kg body weight, and even more preferably about 1 to 10 mg/kg, 2 to 9 mg/kg, 3 to 8 mg/kg, 4 to 7 mg/kg, or 5 to 6 mg/kg body weight.
[0085] For antibodies, the preferred dosage is 0.1 mg/kg to 100 mg/kg of body weight (generally 10 mg/kg to 20 mg/kg). If the antibody is to act in the brain, a dosage of 50 mg/kg to 100 mg/kg is usually appropriate. Generally, partially human antibodies and fully human antibodies have a longer half-life within the human body than other antibodies. Accordingly, lower dosages and less frequent administration is often possible. Modifications such as lipidation can be used to stabilize antibodies and to enhance uptake and tissue penetration (e.g., into the brain). A method for lipidation of antibodies is described by Cruikshank, et al., 1997, J. Acquired Immune Deficiency Syndromes and Human Retrovirology 14:193).
[0086] The pharmaceutical compositions can be included in a container, pack, or dispenser together with instructions for administration.
[0087] The invention further provides a kit containing a peptide or polypeptide of the invention of the present invention, or an antibody or fragments thereof mimicking a polypeptide of the invention, preferably with instructions for use, for example in the treatment of sepsis, septic shock or sepsis-like conditions.
[0088] The invention provides a method for identifying (or screening) modulators, i.e., candidate or test compounds or agents (e.g., peptides, peptidomimetics, small molecules or other drugs) which mimic a polypeptide of the invention or have a stimulatory or inhibitory effect on, for example, activity of a polypeptide of the invention. In particular, the invention provides a method of screening compounds or compositions to treat sepsis, septic shock or sepsis-like conditions, comprising: providing a TREM-1 peptide; contacting an animal in a cecal ligation and puncture model (or using other assay or model as described herein or known in the art) with the TREM-1 peptide; determining if there was a modulation in the sepsis, for example wherein an increase in survival indicates that the TREM-1 peptide may be useful for treating sepsis, septic shock or sepsis-like conditions.
[0089] The invention further pertains to novel agents identified by the above-described screening assays and uses thereof for treatments as described herein.
[0090] All publications, including but not limited to patents and patent applications, cited in this specification are herein incorporated by reference as if each individual publication were specifically and individually indicated to be incorporated by reference herein as though fully set forth.
[0091] Preferred features of each aspect of the invention are applicable to each other aspect, mutatis mutandis.
[0092] The present invention will now be described with reference to the following non-limiting examples, with reference to the figures, in which:
[0093] FIG. 1A. shows a sequence alignment of TREM-1 and TREM-2 family members. Human TREM-1 (SEQ ID NO:1) was aligned with mouse TREM-1 (SEQ ID NO:2) and human and mouse TREM-2 (SEQ ID NO:26 and 27, respectively in order of appearance) using version 1.74 of CLUSTAL W. Secondary structure assignments correspond to the published human TREM-1 structure (arrows for .beta.-strands and cylinder for a helices) (Radaev et al. (2003) Structure (Camb). December; 11(12):1527-35). Residues involved in homo-heterodimer formation are shown in white on black background. Cysteine making disulfide bonds conserved for V-type Ig fold are in bold. Gaps are indicated with (-), identical residues with (*), similar with (: or.). An extended region of similarities between human and mouse TREM1 sequences is shown in boxes on grey background. TREM-1 peptide sequences used in the Examples herein are indicated underlined.
[0094] FIG. 1B. shows a ribbon diagram of the published TREM-1 homodimeric structure (Kelker, et al. (2004) J Mol Biol. September 24; 342(4): 1237-48). Postulated binding sites that comprise the antibody equivalent Complementarity Determining Regions (CDRs) are in red.
[0095] FIG. 2. shows that administration of TREM-1 peptides, 1 hour before LPS, reduces death induced by endotoxaemia. BALB/c mice (10 per group) were injected intraperitoneally with 200 .mu.g LPS. The TREM-1 peptides P1, P2, P3, or P5 (200 .mu.l of a 300 .mu.M solution per mouse) were injected intraperitoneally 1 hour before LPS. Viability of mice was monitored twice a day for 7 days. Statistical analysis was performed by Logrank test. Data from control mice represent cumulative survival curves from two independent experiments performed under identical conditions.
[0096] FIG. 3. shows that TREM-1 peptide P1 is able to effectively reduce death induced by endotoxaemia when injected at 4 hours after LPS. BALB/c mice (10 per group) were injected intraperitoneally with 200 .mu.g LPS. TREM-1 peptide P1, 200 .mu.l of a 300 .mu.M solution per mouse was injected intraperitoneally 1 hour before or 4 hours after LPS. Viability of mice was monitored twice a day for 7 days. Statistical analysis was performed with the Logrank test. Data from control mice represent cumulative survival curves from two independent experiments performed under identical conditions.
[0097] FIG. 4. shows that administration of TREM-1 peptides, 4 hours after LPS, reduces death induced by endotoxaemia. BALB/c mice (10 per group) were injected intraperitoneally with 200 .mu.g LPS. P1 peptide, 200 .mu.l of a 150, 300 and 600 .mu.M solution per mouse (dots) or P3, 200 .mu.l of a 600 .mu.M solution per mouse (filled squares) were injected intraperitoneally 4 hours after LPS. Viability of mice was monitored twice a day for 7 days. Statistical analysis was performed with the Logrank test.
[0098] FIG. 5. shows that TREM-1 peptide P1 protects against cecal ligation and puncture (CLP). CLP was induced in C57BL/6 mice (15 per group) as described in Materials and Methods. P1 peptide (empty dots) or P3 peptide (filled squares) (200 .mu.l of a 600 .mu.M solution per mouse) were injected intraperitoneally 5 and 24 hours after CLP induction. Viability of mice was monitored twice a day for 10 days. Statistical analysis was performed with the Logrank test.
[0099] FIG. 6. shows that P1, P2 and P5 peptides, but not P3 peptide, inhibit the binding of soluble TREM-1/IgG1 to TREM-1 Ligand positive peritoneal exudate cells. Cytofluorimetric analysis of peritoneal exudate cells with 2 .mu.g/ml of mouse TREM-1/hIgG1 in the presence of a 500 .mu.M solution per mouse (thin line), 100 .mu.M solution per mouse (dotted line) or absence (thick line) of the peptides is shown. The grey histogram represents immunostaining with human IgG1 as a control.
[0100] FIG. 7A. shows the release of sTREM-1 from cultured monocytes after stimulation with LPS with and without proteases inhibitor. LPS stimulation induced the appearance of a 27-kD protein that was specifically recognized by an anti-TREM-1 mAb (inset). sTREM-1 levels in the conditioned culture medium were measured by reflectance of immunodots. Data are shown as mean .+-.SD (n=3).
[0101] FIG. 7B. shows expression of TREM-1 mRNA in monocytes. Cultured monocytes were stimulated with LPS (1 .mu.g/mL) for 0, 1 and 16 hours as indicated. LPS induced TREM-1 mRNA production within 1 hour.
[0102] FIG. 8A. shows the release of cytokines and sTREM-1 from cultured monocytes. For cell activation, primary monocytes were cultured in 24-well flat-bottom tissue culture plates in the presence of LPS (1 .mu.g/mL). In some experiments this stimulus was provided in combination with P5 (10 to 100 ng/mL), control peptide (10 to 100 ng/mL) or rIL-10 (500 U/mL). To activate monocytes through TREM-1, an agonist anti-TREM-1 mAb (10 .mu.g/mL) was added as indicated. Cell-free supernatants were analysed for production of TNF-.alpha., IL-1.beta. and sTREM-1 by ELISA or immunodot. All experiments were performed in triplicate and data are expressed as means (SEM). [0103] a: Media [0104] b: P5 10 ng/mL [0105] c: Anti-TREM-1 [0106] d: LPS [0107] e: LPS+Anti-TREM-1 [0108] f: LPS+P5 10 ng/mL [0109] g: LPS+P5 50 ng/mL [0110] h: LPS+P5 100 ng/mL [0111] is LPS+IL10
[0112] FIG. 8B. shows the effect of P5 on NF.kappa.B activation. Monocytes were cultured for 24 hours in the presence of E. coli LPS (O111:B4, 1 .mu.g/mL), anti-TREM-1 mAb (10 .mu.g/mL) and/or P5 (100 ng/mL) as indicated and the levels of NF.kappa.B p50 and p65 were determined using an ELISA-based assay. Experiments were performed in triplicate and data are expressed as means of optical densities (SEM).
[0113] FIG. 9. shows accumulation of sTREM-1 in serum of LPS-treated mice. Male Balb/C mice (20 to 23 g) were treated with LPS (LD.sub.50, intraperitoneally). Serum was assayed for sTREM-1 by immunodot. Serum sTREM-1 was readily detectable 1 hour after LPS administration and was maintained at a plateau level from 4 to 6 hours.
[0114] FIG. 10A. shows that P5 pre-treatment protects against LPS lethality in mice. Male Balb/C mice (20 to 23 g) were randomly grouped (10 mice per group) and treated with an LD.sub.100 of LPS. P5 (50 .mu.g or 100 .mu.g ) or control vector was administered 60 min before LPS.
[0115] FIG. 10B. shows that delayed administration of P5 protects LPS lethality in mice. Male Balb/C mice (20 to 23 g) were randomly grouped (8 mice per group) and treated with an LD.sub.100 of LPS. P5 (75 .mu.g) or control vector was administered 4 or 6 hours after LPS as indicated.
[0116] FIG. 10C. shows that administration of agonist TREM-1 mAb is lethal to mice. Male Balb/C mice (20 to 23 g) were randomly grouped (8 mice per group) and treated with a combination of an LD.sub.50 of LPS+control vector, LD.sub.50 of LPS+anti-TREM-1 mAb (5 .mu.g) or LD.sub.100 of LPS+control vector as indicated. Control vector and anti-TREM-1 mAb were administered 1 hour after LPS injection.
[0117] FIG. 11A. shows that P5 partially protects mice from CLP-induced lethality. Male Balb/C mice (20 to 23 g) were randomly grouped and treated with normal saline (n=14) or the control peptide (n=14, 100 .mu.g) or with P5 (100 .mu.g) in a single infection at HO (n=18), H+4 (n=18) or H+24 (n=18). The last group of mice (n=18) was treated with repeated injections of P5 (100 .mu.g) at H+4, H+8 and H+24.
[0118] FIG. 11B. shows the does effect of P5 on survival. Mice (n=15 per group) were treated with a single injection of normal saline or 10 .mu.g, 20 .mu.g, 50 .mu.g, 100 .mu.g or 200 .mu.g of P5 at HO after the CLP and monitored for survival.
[0119] FIG. 12. shows that P5 has no effect on bacterial counts during CLP. Mice (5 per group) were killed under anaesthesia at 24 hours after CLP. Bacterial counts in peritoneal lavage fluid and blood were determined and results are expressed as CFU per mL of blood and CFU per mouse for the peritoneal lavage.
[0120] FIG. 13A and FIG. 13B show TNF-.alpha. and IL-1.beta. plasma concentration evolution, respectively, after LPS (15 mb/kg) administration in rats.
[0121] *p<0.05 P5-treated vs Control animals
[0122] .sctn.p<0.05 P5-treated vs P1-treated animals
[0123] FIG. 14 shows Nitrite/Nitrate concentrations evolution after LPS (15 mg/kg) administration in rats. *p<0.05 P5-treated vs Control and P1-treated animals.
[0124] FIG. 15 shows mean arterial pressure evolution during caecal ligation and puncture-induced peritonitis in rats.
[0125] *p<0.05 vs Control animals
[0126] FIG. 16 shows TNF-.alpha. plasma concentration evolution during caecal ligation and puncture-induced peritonitis in rats.
[0127] *p<0.05 P5-treated vs Control animals
[0128] .sup..sctn.p<0.05 P1-treated vs Control animals
[0129] .sup.$p<0.05 P5 vs P1-treated animals
[0130] FIG. 17 shows Nitrite/Nitrate concentration evolution during caecal ligation and puncture-induced peritonitis in rats.
[0131] *p<0.05 P5 and P1-treated vs Control animals
[0132] FIG. 18 shows that hP5 efficiently protects mice from LPS induced septic shock (see Example 5). Septic shock was induced in male Balb/c mice (n=15/group) with 200 mg of LPS as described in Material and Methods. hP5 peptide or control were administered at the following time points: -1 h, 0 h, +1 h +4 h. Mortality was followed twice a day for 7 days. Data were analysed using GraphPad Prism Survival curve analysis. Log-rank (Mantel-Cox) test showed a statistically significant difference between the two curves (p=0.0003).
[0133] FIG. 19. shows the results for male C57BL/6 mice (n=15/group) that were subject to Cecal Ligation and Puncture as described in Example 6 and treated with hP5 peptide or scrambled peptide control at the following time points: -1 h, 0 h, +4 h and +24 h. Mortality was followed twice a day for 10 days. Data were analyzed using GraphPad Prism Survival curve analysis.
[0134] FIG. 20. Disease Activity Index (DAI). Colitis was induced by oral administration of DSS (see Example 7). Upon colitis induction, mice (n=8) were daily treated either with hP5 or its scrambled control peptide. Animal weight, haemoccult or presence of gross blood and stool consistency were used to determine the DAI score as indicated in Example 7. *(grey)=DSS+hP5 versus DSS+scrambled peptide; *(black)=DSS+hP5 vs DSS+vehicle; ***p.ltoreq.0.001, **p.ltoreq.0.01, *p.ltoreq.0.05.
[0135] FIG. 21. Body weight loss. Colitis was induced by oral administration of DSS (see Example 7). Upon colitis induction, mice were daily treated either with hP5 or its scrambled control peptide. Weight was daily measured and percentage of weight loss versus day 0 was calculated. *(grey)=DSS+hP5 versus DSS+scrambled peptide; *(black)=DSS+hP5 vs DSS+vehicle; ***p.ltoreq.0.001, **p.ltoreq.0.01.
[0136] FIG. 22. Haemoccult or presence of gross blood in the feces. Colitis was induced by oral administration of DSS (see Example 7). Upon colitis induction, mice were daily treated either with hP5 or its scrambled control peptide. Haemoccult or presence of gross blood was detected and score assigned as measured and score assigned at the indicated time points as described in. Example 7. *(grey)=DSS+hP5 versus DSS+scrambled peptide; *(black)=DSS+hP5 vs DSS+vehicle; ***p.ltoreq.0.001, **p.ltoreq.0.01, *p.ltoreq.0.05.
[0137] FIG. 23. Extent of diarrhea. Colitis was induced by oral administration of DSS (see Example 7). Upon colitis induction, mice were daily treated either with hP5 or its scrambled control peptide. Stool consistency was measured and score assigned at the indicated time points as described in Example 7. *(grey)=DSS+hP5 versus DSS+scrambled peptide; *(black)=DSS+hP5 vs DSS+vehicle; ***p.ltoreq.0.001, **p.ltoreq.0.01, *p.ltoreq.0.05.
[0138] FIG. 24. Shortening of the colon. Colitis was induced by oral administration of DSS (see Example 7). Upon colitis induction, mice were daily treated either with hP5 or its scrambled control peptide. At day 11 mice were euthanized and the colon length of each mouse measured from the anus to the end of the cecum. ***p.ltoreq.0.001, **p.ltoreq.0.01, *p.ltoreq.0.05.
EXAMPLE 1
TREM-1 Peptides Protect Mice From Death by Septic Shock
[0139] TREM-1 peptides matching the following criteria were synthesized: i) highest homology between human and mouse TREM-1 and lowest homology with TREM-2. ii) peptides spanning the Complementarity Determining Regions (CDRs) of TREM-1. According to the published crystal structure of TREM-1, and in analogy with antibodies, these residues are likely to be involved in cognate ligand recognition (Radaev et al. (2003) Structure (Camb). December; 11(12):1527-35 & Kelker, et al. (2004) J Mol Biol. September 24; 342(4):1237-48) (see FIG. 1A and FIG. 1B). One peptide (P1) was designed in the CDR2 region and three peptides (P2, P4 and P5) in the CDR3 region. A fourth peptide (P3) was designed in the neck region connecting the V-type immunoglobulin (Ig)-like domain (Ig-V) to the transmembrane domain. No peptide was designed in the CDR1 region due to high sequence homology between TREM-1 and TREM-2.
[0140] Thus, the following peptides of the TREM-1 protein were ordered from and were synthesized and purified by the Protein and Peptide Chemistry Facility, Institute of Biochemistry, University of Lausanne:
TABLE-US-00004 P1 (CDR2 67-89) LVVTQRPFTRPSEVHMG [SEQ ID KFTLKH NO: 3] P2 (CDR3 114-136) VIYHPPNDPVVLFHPVR [SEQ ID LVVTKG NO: 4] P3 (neck region TTTRSLPKPTAVVSSPG [SEQ ID 168-184) NO: 5] P4 (CDR3 103-123) LQVTDSGLYRCVIYHPP [SEQ ID NDPV NO: 6] P5 (CDR3 103-119) LQVTDSGLYRCVIYHPP [SEQ ID NO: 7] P1sc* (P1 scrambled LTPKHGQRSTHVTKFRV [SEQ ID seq.) FEPVML NO: 8] P5sc* (P5 scrambled TDSRCVIGLYHPPLQVY [SEQ ID seq.) NO: 9] *This is a control peptide and indeed does not protect
[0141] In the experiments of this example, the peptides were administered in a volume of 200 .mu.l of the solution molarity indicated. To assess the ability of TREM1-peptides to protect mice from LPS-induced endotoxaemia, the Inventors administered peptides P1, P2, P3 and P5 (300 .mu.M) 1 hour before a lethal dose of lipopolysaccharide (LPS) (FIG. 2). Lethality was monitored over time and compared with animals that had received control injections of vehicle alone. P5 injection confers maximal protection, with 90% of the animals still alive 7 days after LPS injection, as compared with 10% of control mice (p<0.001). 60% of the P I-treated mice and 50% of the P2 treated mice survived endotoxaemia as compared with 10% of control mice (p<0.01 and p<0.05 respectively). Interestingly, all P3-treated mice died within 4 days after LPS injection. These results indicate that peptides containing sequences of the extracellular portion of TREM-1 corresponding to the putative ligand binding site (CDR2 and CDR3) can protect mice from lethal shock.
[0142] In order to investigate whether TREM-1 peptide treatment could be delayed until after the administration of LPS, the Inventors injected the peptides at 4 hours after LPS injection. Only in the case of P1, this delayed treatment conferred significant protection against a lethal dose of LPS (FIG. 3). 80% of the mice injected with P1 4 hours after LPS survived endotoxaemia compared to 60% of mice treated 1 h before LPS and 10% of mice treated with vehicle alone (p<0.001 and p<0.01 respectively). Thus, P1 is effective even when injected after the outbreak of endotoxaemia. No late death occurred over one week, indicating that P1 did not merely delay the onset of LPS lethality, but provided lasting protection. P1 administration conferred maximal protection (80%) when administered at 600 .mu.M (p<0.01) and the level of protection dropped to 50% at 300 .mu.M (p<0.05) and further down to 30% at 150 .mu.M as compared to 20% of control mice, indicating a dose dependent effect of P1 (FIG. 4). The Inventors then investigated whether P1 protects against septic shock in the "CLP" model (Cecal Ligation and Puncture is a widely used experimental model of sepsis). Mice treated with two doses of P1 at 5 and 24 hours after CLP were protected from death as compared to control treated mice (p=0.0791) although the difference was not statistically significant. 40% of the mice injected with P1 at 5 days after CLP survived compared to 5% of mice treated with P3 peptide. At 10 days after CLP, the treated mice were still alive, indicating that P1 did not merely delay mortality, but provided lasting protection (FIG. 5).
EXAMPLE 2
TREM-1 Peptide P1 Inhibits Binding of Soluble Mouse TREM-1/IgG to TREM-1 Ligand Positive Cells
[0143] Among TREM-1 derived peptides tested in CLP, peptides P1, P2 and P5 demonstrate a protective activity. A possible mechanism of action could be the ability of TREM-1 derived peptide to interfere with TREM-1/TREM-1 ligand interaction. To address this question the Inventors performed competition experiments on TREM-1 ligand positive cells: PEC (Peritoneal Exudate Cells) from CLP treated mice.
[0144] Peritoneal exudate cells (PEC) from mice suffering from a caecal ligation and puncture (CLP)-induced peritonitis were subjected to flow cytometry analysis after incubation with a PE-conjugated anti-human IgG1 (Jackson Immunoresearch, Bar Harbor, USA). Competition with TREM-1 peptides was performed by pre-incubating cells with the indicated concentrations of peptides for 45 minutes on ice before adding mTREM-1-IgG1.
[0145] As shown in FIG. 6, the P1 peptide, derived from the CDR2 region of mTREM-1, and the P2 and P5 peptides spanning the CDR3 region inhibit TREM-1 interaction with its ligand in a dose dependent manner. Conversely the P3 peptide, derived from the neck region of TREM-1 connecting the IgG like portion to the transmembrane domain was ineffective.
EXAMPLE 3
[0146] Additional Studies on the Modulation of the Inflammatory Response in Murine Sepsis by TREM-1 Peptide P5
[0147] Methods
Preparation of Monocytes from Peripheral Blood
[0148] Ten mL of peripheral blood samples were collected on EDTA-K from 5 healthy volunteer donors originating from laboratory staff. After dilution in RPMI (Life Technologies, Grand Island, N.Y.) v/v, blood was centrifuged for 30 min at room temperature over a Ficoll gradient (Amersham Pharmacia, Uppsala, Sweden) to isolate PBMC. The cells recovered above the gradient were washed and counted. In order to deplete the suspensions of lymphocytes, cells were then plated in 24-well flat-bottom tissue culture plates (Corning, Corning, N.Y.) at a concentration of 5.times.10.sup.6/mL and allowed to adhere during 2 hours at 37.degree. C. The resulting lymphocyte suspension was discarded and the adhering monocytic cells were maintained in a 5% CO2 incubator at 37.degree. C. in complete medium (RPMI 1640, 0.1 mM sodium pyruvate, 2 mM Penicillin, 50 .mu.g/mL Streptomycin; Life Technologies) supplemented with 10% FCS (Invitrogen, Cergy, France).
TREM-1 Peptide
[0149] Using the human TREM-1 sequence in Gen-Bank, accession # AF287008 and the mouse TREM-1 sequence # AF241219, a peptide "P5" (LQVTDSGLYRCVIYHPP; [SEQ ID NO:7]; was chemically synthesized as a C-terminally amidated peptide (Pepscan Systems, Lelystad, The Netherlands). The correct peptide was obtained in greater than 99% yield and with measured mass of 1961 Da versus a calculated mass of 1962 Da and was homogeneous after preparative purification, as confirmed by mass spectrometry and analytic reversed phase-high performance liquid chromatography. A peptide "P5sc" containing the same amino-acids than P5 but in a different sequence order (TDSRCVIGLYHPPLQVY; [SEQ ID NO:9]) was similarly synthesized and served as `control peptide`.
In Vitro Stimulation of Monocytes
[0150] For activation, monocytes were cultured in the presence of E. coli LPS (O111:64, 1 .mu.g/mL, Sigma-Aldrich, La Verpilliere, France). Cell viability was assessed by trypan blue exclusion and by measuring lactate dehydrogenase release. In some experiments, this stimulus was given in combination with TNF-.alpha. (5 to 100 ng/mL, R&D Systems, Lille, France), IL-1.beta. (5 to 100 ng/mL, R&D Systems), rIFN-.gamma. (up to 100 U/mL, R&D Systems), rIL-10 (500 U/mI, R&D Systems) or up to 100 ng/mL of P5 or control peptide.
[0151] In order to activate monocytes through TREM-1, an anti-TREM-1 agonist monoclonal antibody (R&D Systems) was added as follows: flat-bottom plates were precoated with 10 .mu.g/mL anti-TREM-1 per well. After thorough washing in phosphate buffered saline (PBS), the monocyte suspensions were added at a similar concentration as above. Some experiments were performed in the presence of protease inhibitors (PMSF and Protease Cocktail Inhibitor; Invitrogen). Cell-free supernatants were assayed for the production of TNF-.alpha. and IL-1.beta. by ELISA according to the recommendations of the manufacturer (BD Biosciences, San Diego, USA). To address the effect of P5 on NF-.kappa.B activity in monocytes, an ELISA-based assay was performed (BD Mercury.TM. Transfactor Kit, BD Biosciences). Monocytes were cultured for 24 hours in the presence of E. coli LPS (O111:64, 1 .mu.g/mL), and/or an agonist anti-TREM-1 monoclonal antibody (10 .mu.g/mL), and/or P5 (100 ng/mL). Whole-cell extracts were then prepared and levels of NF-.kappa.B p50 and p65 were determined according to the recommendations of the manufacturer. All experiments were performed in triplicate and data are expressed as means (SEM).
Identification and Quantitation of sTREM-1 Release
[0152] Primary monocytes suspensions were cultured as described above. The cells were treated with E. coli LPS (O111:64, 1 .mu.g/mL) for 24 hours at 37.degree. C. Cell-conditioned medium was submitted to Western-blotting using an anti-TREM-1 monoclonal antibody (R&D Systems) in order to confirm the presence of 27 kDa material recognized by anti-TREM-1. Soluble TREM-1 levels were measured by assessing the optical intensity of bands on immunodots by means of a reflectance scanner and the Quantity One Quantitation Software (Bio-Rad, Cergy, France) as reported elsewhere (18). Soluble TREM-1 concentration from each sample was determined by comparing the optical densities of the samples with reference to standard curves generated with purified TREM-1. All measurements were performed in triplicate. The sensitivity of this technique allows the detection of sTREM-1 levels as low as 5 pg/mL.
TREM-1 RT-PCR
[0153] Total mRNA was extracted from primary monocytes cultured in the presence of LPS using a TRIzol reagent (Invitrogen), and reverse transcribed using Superscript RT II (Invitrogen) to generate cDNA. RT-PCR conditions then used for all reactions were 94.degree. C., 30 s/65.degree. C., 30 s/68.degree. C., 1 min for 30 cycles. Amplification was performed with 2.5 mM MgCl2, 0.2 mM dNTP, 2.0 U Taq polymerase, and 20 pM 5' and 3' oligonucleotide primers (Proligos, Paris, France).
[0154] The sequences of the 5' and 3' primer pairs used were the following:
TABLE-US-00005 for TREM-1 (17) [SEQ ID NO: 10] TTGTCTCAGAACTCCGAGCTGC; and [SEQ ID NO: 11] GAGACATCGGCAGTTGACTTGG; for TREM-1sv (19) [SEQ ID NO: 12] GGACGGAGAGATGCCCAAGACC; and [SEQ ID NO: 13] ACCAGCCAGGAGAATGACAATG; for .beta.-actin (used as housekeeping amplicon) [SEQ ID NO: 14] GGACGACATGGAGAAGATCTGG; and [SEQ ID NO: 15] ATAGTAATGTCACGCACGATTTCC;
[0155] PCR products were run on agarose gels and visualized by ethidium bromide staining.
LPS-Induced Endotoxinemia in Mice
[0156] After approval by the local ethical committee, male Balb/C mice (20 to 23 g) were randomly grouped and treated with E. coli LPS intraperitoneally (i.p.) in combination with P5 (in 500 .mu.l normal saline) or control vector before or after LPS challenge. In some experiments, 5 .mu.g of an anti TREM-1 monoclonal antibody was administered i.p. one hour after LPS injection. The viability of mice was examined every hour, or animals were sacrificed at regular intervals. Serum samples were collected by cardiac puncture and assayed for TNF-.alpha. and IL-1.beta. by ELISA (BD Biosciences), and for sTREM-1 levels by immunodot.
CLP Polymicrobial Sepsis Model
[0157] Male Balb/C mice (7 to 9 weeks, 20 to 23 g) were anaesthetized by i.p. administration of ketamine and xylazine in 0.2 mL sterile pyrogen-free saline. The caecum was exposed through a 1.0 cm abdominal midline incision and subjected to a ligation of the distal half followed by two punctures with a G21 needle. A small amount of stool was expelled from the punctures to ensure patency. The caecum was replaced into the peritoneal cavity and the abdominal incision closed in two layers. After surgery all mice were injected s.c. with 0.5 ml of physiologic saline solution for fluid resuscitation and s.c. every 12 h with 1.25 mg (i.e. 50 .mu.g/g) of imipenem. The animals were randomly grouped and treated with normal saline (n=14), the control peptide (n=14, 100 .mu.g) or P5 (100 .mu.g) in a single injection at HO (n=18), H+4 (n=18) or H+24 (n=18). The last group of mice (n=18) was treated with repeated injections of P5 (100 .mu.g) at H+4, H+8 and H+24. All treatments were diluted into 500 .mu.l of normal saline and administered i.p. The Inventors next sought to determine the effect of various doses of P5. For this purpose, mice (n=15 per group) were treated with a single injection of normal saline or 10 .mu.g, 20 .mu.g, 50 .mu.g, 100 .mu.g or 200 .mu.g of P5 at HO after the CLP and monitored for survival. Five additional animals per group were killed under anaesthesia 24 hours after CLP for the determination of bacterial count and cytokines levels. Peritoneal lavage fluid was obtained using 2 mL RPMI 1640 (Life Technologies) and blood was collected by cardiac puncture. Concentrations of TNF-.alpha. and IL-1.beta. in the serum were determined by ELISA (BD Biosciences). For the assessment of bacterial counts, blood and peritoneal lavage fluid were plated in serial log dilutions on tryptic soy supplemented with 5% sheep blood agar plates. After plating, tryptic soy agar plates were incubated at 37.degree. C. aerobically for 24 hours, and anaerobically for 48 hours. Results are expressed as CFU per mL of blood and CFU per mouse for the peritoneal lavage.
Statistical Analyses
[0158] Serum sTREM-1 and cytokines levels were expressed as mean (.+-.SD).The protection against LPS lethality by P5 was assessed by comparison of survival curves using the Log-Rank test. All statistical analyses were completed with Statview software (Abacus Concepts, Berkeley Calif.) and a two-tailed P<0.05 was considered significant.
[0159] Results
[0160] A Soluble Form of TREM-1 is Released from Cultured Human Monocytes After Stimulation with E. coli LPS
[0161] To identify the potential release of sTREM-1 in vitro, the Inventors stimulated human monocytes with LPS and analyzed the conditioned culture medium by SDS-PAGE. LPS stimulation induced the appearance of a 27-kDa protein in a time-dependent manner (FIG. 7A). Western blotting analysis revealed that this protein was specifically recognized by a monoclonal antibody directed against the extra-cellular domain of TREM-1 (FIG. 7A). Cell viability was unaffected at LPS concentrations that induced the presence of sTREM-1 in conditioned medium, indicating that TREM-1 release was not due to cell death. Similarly, treatment of monocytes with protease inhibitors did not affect TREM-1 release (FIG. 7A). TREM-1 mRNA levels were increased upon LPS treatment (FIG. 7B) whereas TREM-1sv mRNA levels remained undetectable. This suggests that TREM-1 release is likely to be linked to an increased transcription of the gene and unrelated to TREM-1sv expression. Stimulation of monocytes for 16 hours with TNF-.alpha. (5 to 100 ng/mL) or IL-1.beta. (5 to 100 ng/mL) induced very small TREM-1 release in a cytokine dose-dependent manner. Stimulation with IFN-.gamma. did not induce TREM-1 release, even at concentrations of up to 100 U/mL.
[0162] LPS Associated Release of Pro-Inflammatory Cytokines is Attenuated by P5
[0163] Significant TNF-.alpha. and IL-1.beta. production was observed in the supernatant of monocytes cultured with LPS. TNF-.alpha. and IL-13 production was even higher for cells cultured with both TREM-1 mAb and LPS as compared with those cultured with mAb or LPS alone (FIG. 8A).
[0164] The inducible release of pro-inflammatory cytokines was significantly lower after LPS stimulation when the medium was supplemented with P5 or IL-10. P5 reduced, in a concentration-dependent manner, the TNF-.alpha. and IL-1.beta. production from cells cultured with LPS or with LPS and mAb and simultaneously increased the release of sTREM-1 from cells cultured with LPS. The control peptide displayed no action on cytokines or sTREM-1 release (data not shown). In striking contrast, IL-10 totally inhibited the release of both TREM-1 and inflammatory cytokines (FIG. 8A). Both LPS and TREM-1 mAb induced a strong activation of monocytic NF-.kappa.B p50 and p65 and combined administration of LPS and TREM-1 mAb lead to a synergistic effect. P5 inhibited the NF-.kappa.B activation induced by the engagement of TREM-1 but did not alter the effect of LPS (FIG. 8B).
[0165] Serum sTREM-1 Levels of LPS-Treated Mice are Increased
[0166] In order to determine whether sTREM-1 was released systemically during endotoxemia in mice, the Inventors measured serum sTREM-1 levels after LPS administration. Serum sTREM-1 was readily detectable 1 hour after administration of an LD.sub.50 dose of LPS and was maintained at peak plateau levels from 4 to 6 hours after LPS treatment (FIG. 9).
[0167] TREM-1 Peptide "P5" Protects Endotoxemic Mice from Lethality
[0168] Mice treated by a single dose of P5 60 min before a lethal dose (LD.sub.100) of LPS were prevented from death in a dose-dependent manner (FIG. 10A). In order to investigate whether P5 treatment could be delayed until after the administration of LPS, the Inventors injected P5 beginning 4 or 6 hours after LPS injection. This delayed treatment up to 4 hours conferred significant protection against a LD.sub.100 dose of LPS (FIG. 10B). No late death occurred over one week, indicating that P5 did not merely delay the onset of LPS lethality, but provided lasting protection. Control mice all developed lethargy, piloerection, and diarrhoea before death. By contrast, P5-treated mice remained well groomed and active, had no diarrhoea, and were lively. To clarify the mechanism by which P5 protected mice from LPS lethality, the Inventors determined the serum levels of TNF-.alpha., IL-1.beta. and sTREM-1 of endotoxemic mice at 2 and 4 hours. As compared to controls, pre-treatment by 100 .mu.g of P5 reduced cytokines levels by 30% and increased sTREM-1 levels by 2 fold as shown in Table 4:
TABLE-US-00006 TABLE 4 Serum concentrations of TNF-.alpha., IL-1.beta. and sTREM-1 in endotoxemic mice. TNF-.alpha. (ng/mL) IL-1.beta. (ng/mL) sTREM-1 (ng/mL) H2 H4 H2 H4 H2 H4 Control 3.3 .+-. 1.0 0.4 .+-. 0.1 0.3 .+-. 0.1 1.5 .+-. 0.2 249 .+-. 48 139 .+-. 8 P5 (100 .mu.g) 2.4 .+-. 0.5 0.1 .+-. 0.1 0.2 .+-. 0.1 0.9 .+-. 0.2 475 .+-. 37 243 .+-. 28
[0169] Engagement of TREM-1 is Lethal to Mice
[0170] To further highlight the role of TREM-1 engagement in LPS-mediated mortality, mice were treated with agonist anti-TREM-1 mAb in combination with the administration of an LD.sub.50 dose of LPS. This induced a significant increase in mortality rate from 50% to 100% (FIG. 10C).
[0171] P5 Protects Mice from CLP-Induced Lethality
[0172] To investigate the role of P5 in a more relevant model of septic shock, the Inventors performed CLP experiments (FIG. 11A). The control groups comprised mice injected with normal saline or with the control peptide. In this model of polymicrobial sepsis, P5 still conferred a significant protection against lethality even when administered as late as 24 hours after the onset of sepsis. Interestingly, repeated injections of P5 had the more favourable effect on survival (P<0.01). There was a dose response effect of P5 on survival (FIG. 11B) and cytokine production (Table 5). P5 had no effect on bacterial clearance (FIG. 12).
TABLE-US-00007 TABLE 5 Serum concentrations of TNF-.alpha., IL-1.beta. and sTREM-1 at 24 hours after CLP. TNF-.alpha. (pg/mL) IL-1.beta. (pg/mL) sTREM-1 (ng/mL) Control peptide 105 .+-. 12 841 .+-. 204 52 .+-. 3 Control saline 118 .+-. 8 792 .+-. 198 35 .+-. 5 P5 10 .mu.g 110 .+-. 11 356 .+-. 62 43 .+-. 8 P5 20 .mu.g 89 .+-. 10 324 .+-. 58 58 .+-. 8 P5 50 .mu.g 24 .+-. 6 57 .+-. 11 93 .+-. 10 P5 100 .mu.g 20 .+-. 3 31 .+-. 3 118 .+-. 12 P5 200 .mu.g 21 .+-. 7 37 .+-. 8 158 .+-. 13
[0173] Sepsis exemplifies a complex clinical syndrome that results from a harmful or damaging host response to severe infection. Sepsis develops when the initial, appropriate host response to systemic infection becomes amplified, and then dysregulated (4, 5). Neutrophils and monocyte/macrophages exposed to LPS, for instance, are activated and release such pro-inflammatory cytokines as TNF-.alpha. and IL-1.beta.. Excessive production of these cytokines is widely believed to contribute to the multi-organ failure that is seen in septic patients (20-23).
[0174] TREM-1 is a recently identified molecule involved in monocytic activation and inflammatory response (12, 14). It belongs to a family related to NK cell receptors that activate downstream signalling events. The expression of TREM-1 on PNNs and monocytes/macrophages has been shown to be inducible by LPS (16, 17).
[0175] As described herein, the Inventors demonstrate that a soluble form of TREM-1 was released from cultured human monocytes after stimulation with E. coli LPS. Such a soluble form was also detectable in the serum of endotoxemic mice as early as 1 hour after LPS challenge. This is consistent with the implication of TREM-1 in the very early phases of the innate immune response to infection (14, 15, 24). The mechanism by which sTREM-1 is released is not clearly elucidated but seems to be related to an increased transcription of the TREM-1 gene. Nevertheless, although incubation with a protease inhibitor cocktail does not alter the sTREM-1 release, cleavage of the surface TREM-1 from the membrane cannot be totally excluded. Interestingly, stimulation of human monocytes with such pro-inflammatory cytokines as TNF-.alpha., IL-1.beta. or IFN-.gamma. induced very small sTREM-1 release unless LPS was added as a co-stimulus. The expression of an alternative mRNA TREM-1 splice variant (TREM-1sv) has been detected in monocytes that might translate into a soluble receptor (18) upon stimulation with cell wall fraction of Mycobacterium bovis BCG but not LPS (25). This was confirmed in this study as i) LPS did not increase the level of mRNA TREM-1sv in monocytes and ii) only a 27-kDa protein was released by monocytes upon LPS stimulation and not the 17.5-kDa variant.
[0176] Although its natural ligand has not been identified (13, 14), engagement of TREM-1 on monocytes with an agonist monoclonal antibody resulted in a further enhancement of pro-inflammatory cytokines production, while P5 induced a decrease of these syntheses in a concentration-dependent manner, and IL-10 completely suppressed it.
[0177] Inflammatory cytokines, and especially TNF-.alpha., are considered to be deleterious, yet they also possess beneficial effects in sepsis (5) as shown by the fatal issue of peritonitis in animals with impaired TNF-.alpha. responses (9-11). Moreover, in clinical trials, the inhibition of TNF-.alpha. increased mortality (8). Finally, the role of TNF-.alpha. in the clearance of infection has been highlighted by the finding that sepsis is a frequent complication in rheumatoid arthritis patients treated with TNF-.alpha. antagonists (26).
[0178] The mechanism by which P5 modulates cytokine production is not yet clear. P5 comprises the complementary determining region (CDR)-3 and the `F` .beta. strand of the extracellular domain of TREM-1. The latter contains a tyrosine residue mediating dimerization. Radaev et al postulated that TREM-1 captures its ligand with its CDR-equivalent loop regions (27). P5 could thus impair TREM-1 dimerization and/or compete with the natural ligand of TREM-1. Moreover, the increase of sTREM-1 release from monocytes mediated by P5 could prevent the engagement of membrane TREM-1, sTREM-1 acting as a decoy receptor, as in the TNF-.alpha. system (28, 29).
[0179] Activation of the transcription factor NF-.kappa.B is a critical step in monocyte inflammatory cytokine production after exposure to bacterial stimuli such as LPS (30, 31). Among the various NF-.kappa.B/Rel dimers, the p65/p50 heterodimer is the prototypical form of LPS-inducible NF-.kappa.B in monocytes (32). P5 abolishes the p65/p50 NF-.kappa.B over-activation induced by the engagement of TREM-1. This might at least partially explain the effects of P5 on cytokine production and the protection from lethality shown here to occur when the peptide was injected one hour before LPS-induced septic shock, or even up to 4 hours after.
[0180] Endotoxemia is simple to achieve experimentally, but imperfectly suited to reproduce human sepsis, while polymicrobial sepsis induced by CLP is a more complex but better model, including the use of fluid resuscitation and antibiotics. The latter was thus also used in this study, and confirmed the dose-dependent protection provided by P5, even when administered as late as 24 hours after the onset of sepsis. The favourable effect of P5 was however unrelated to an enhanced bacterial clearance.
[0181] One difficulty in the use of immunomodulatory therapies is that it is not possible to predict the development of sepsis, and, thus, patients receiving those treatments frequently already have well-established sepsis (6). Since P5 appeared to be effective even when injected after the outbreak of sepsis, it could thus constitute a realistic treatment (24, 33).
[0182] By contrast, engagement of TREM-1 by an agonist anti-TREM-1 monoclonal antibody mediated a dramatic increase of mortality rate in LPS-challenged mice: this further underscores the detrimental effect of TREM-1 engagement during septic shock.
[0183] Experimental septic shock reproduces human sepsis only in part. Indeed, our group recently showed that significant levels of sTREM-1 were released in the serum of critically ill patients with sepsis patients (34), the highest levels being observed in patients who survived. This is consistent with our experimental findings indicating that the more important sTREM-1 release, the more favourable is the outcome, and thus sustains, at least theoretically, the potential value of soluble TREM peptides as post-onset sepsis therapy.
[0184] TREM-1 appears to be a crucial player in the immediate immune response triggered by infection. In the early phase of infection, neutrophils and monocytes initiate the inflammatory response owing to the engagement of pattern recognition receptors by microbial products (3, 4). At the same time, bacterial products induce the up-regulation and the release of sTREM-1. Upon recognition of an unknown ligand, TREM-1 activates signalling pathways which amplify these inflammatory responses, notably in monocytes/macrophages. The modulation of TREM-1 signalling reduces, although without complete inhibition, cytokine production and protects septic animals from hyper-responsiveness and death. Modulation of TREM-1 engagement with such a peptide as P5 might be a suitable therapeutic tool for the treatment of sepsis, particularly because it seems to be active even after the onset of sepsis following infectious aggression.
EXAMPLE 4
Haemodynamic Studies in LPS Treated and Septic Rats Treated with P1 and P5
[0185] The role of TREM-1 peptides in further models of septic shock, was investigated by performing LPS and CLP (caecal ligation and puncture) experiments in rats.
[0186] Materials and Methods
[0187] LPS-Induced Endotoxinemia
[0188] Animals were randomly grouped (n=10-20) and treated with Escherichia coli LPS (O111:64, Sigma-Aldrich, Lyon, France) i.p. in combination with the TREM-1 or scrambled peptides.
[0189] CLP Polymicrobial Sepsis Model
[0190] The procedure has been described in details elsewhere (see Mansart, A. et al. Shock 19:3844 (2003)). Briefly, rats (n=6-10 per group) were anesthetized by i.p. administration of ketamine (150 mg/kg). The caecum was exposed through a 3.0-cm abdominal midline incision and subjected to a ligation of the distal half followed by two punctures with a G21 needle. A small amount of stool was expelled from the punctures to ensure potency. The caecum was replaced into the peritoneal cavity and the abdominal incision closed in two layers. After surgery, all rats were injected s.c. with 50 mL/kg of normal saline solution for fluid resuscitation. TREM-1 or scrambled peptides were then administered as above.
[0191] Haemodynamic Measurements in Rats
[0192] Immediately after LPS administration as well as 16 hours after CLP, arterial BP (systolic, diastolic, and mean), heart rate, abdominal aortic blood flow, and mesenteric blood flow were recorded using a procedure described elsewhere (see Mansart, A. et al. Shock 19:3844 (2003)). Briefly, the left carotid artery and the left jugular vein were cannulated with PE-50 tubing. Arterial BP was continuously monitored by a pressure transducer and an amplifier-recorder system (IOX EMKA Technologies, Paris, France). Perivascular probes (Transonic Systems, Ithaca, N.Y.) wrapped up the upper abdominal aorta and mesenteric artery, allowed to monitor their respective flows by means of a flowmeter (Transonic Systems). After the last measurement (4.sup.th hour during LPS experiments and 24.sup.th hour after CLP), animals were sacrificed by an overdose of sodium thiopental i.v.
[0193] Biological Measurements
[0194] Blood was sequentially withdrawn from the left carotid artery. Arterial lactate concentrations and blood gases analyses were performed on an automatic blood gas analyser (ABL 735, Radiometer, Copenhagen, Denmark). Concentrations of TNF-.alpha. and IL-1.beta. in the plasma were determined by an ELISA test (Biosource, Nivelles, Belgium) according to the recommendations of the manufacturer. Plasmatic concentrations of nitrates/nitrites were measured using the Griess reaction (R&D Systems, Abingdon, UK).
[0195] Statistical Analyses
[0196] Results are expressed as mean.+-.SD. Between-group comparisons were performed using Student' t tests. All statistical analyses were completed with Statview software (Abacus Concepts, CA) and a two-tailed P<0.05 was considered significant.
[0197] Results
[0198] Endotoxinemia Model
[0199] Following LPS administration, arterial pressures, aortic and mesenteric blood flows dropped rapidly in control animals (scrambled peptides treated rats) while the heart rate remained unchanged (Table 6). The decrease of arterial pressures and aortic blood flow was delayed until the second hour in TREM-1 peptide treated animals with significantly higher values by that time than in control animals. There was no difference between P1 and P5 treated groups. By contrast, none of these two peptides had any effect on the decrease of the mesenteric blood flow (Table 6).
[0200] Arterial pH remained constant over time until the fourth hour after LPS injection where it severely dropped in the control group only (Table 6). The significant arterial lactate level elevation present in control animals after the third hour was abolished by the TREM-1 peptides (Table 6). There was no difference between P1 and P5 with regard to pH, arterial bicarbonate and lactate concentrations.
[0201] As expected, a peak of TNF-.alpha. plasmatic concentration was induced by LPS between 30 minutes and 1 hour after injection followed by a progressive decline thereafter (FIG. 13A). P1 peptide injection had no effect on this production, while P5 attenuated TNF-.alpha. production by -30%.
[0202] P1 delayed the IL-1.beta. peak until the third hour after LPS injection, but without attenuation. By contrast, P5 strongly reduced IL-1.beta. release (FIG. 13B).
[0203] Nitrite/nitrate concentrations increased rapidly after LPS administration in control and P1 treated animals but remained stable upon P5 treatment (FIG. 14).
TABLE-US-00008 TABLE 6 Hemodynamic parameters during LPS-induced endotoxinemia Heart Aortic Mesenteric Rate MAP blood flow blood flow Lactate (bpm) (mmHg) (mL/min) (mL/min) pH (mmol/L) Control H0 486 .+-. 13 123 .+-. 21 45 .+-. 7 13.6 .+-. 3.4 7.31 .+-. 0.03 3.3 .+-. 0.8 H1 522 .+-. 16 103 .+-. 25 25 .+-. 8 .sup.a 9.6 .+-. 3.3 7.28 .+-. 0.03 4.2 .+-. 0.3 H2 516 .+-. 13 98 .+-. 23 12 .+-. 5 .sup.a,b 8.0 .+-. 3.7 7.29 .+-. 0.03 5.9 .+-. 0.6 H3 490 .+-. 20 78 .+-. 8 .sup.a,b 8 .+-. 3 .sup.a,b 5.8 .+-. 1.1 7.26 .+-. 0.01 7.9 .+-. 1.8 .sup.a,b H4 510 .+-. 18 67 .+-. 9 .sup.a,b 6 .+-. 1 .sup.a,b 4.1 .+-. 0.8 7.03 .+-. 0.10 .sup.a,b 11.5 .+-. 0.7 .sup.a,b P1 H0 464 .+-. 25 116 .+-. 10 49 .+-. 11 12.0 .+-. 3.7 7.32 .+-. 0.04 2.7 .+-. 0.1 H1 492 .+-. 26 119 .+-. 14 39 .+-. 12 .sup.a 10.5 .+-. 1.7 7.29 .+-. 0.04 4.9 .+-. 1.1 H2 492 .+-. 26 113 .+-. 21 26 .+-. 14 .sup.a 7.7 .+-. 2.7 7.30 .+-. 0.01 5.0 .+-. 0.9 H3 480 .+-. 30 97 .+-. 29 .sup.a 22 .+-. 8 .sup.a 5.0 .+-. 1.0 7.26 .+-. 0.06 5.7 .+-. 0.7 .sup.a H4 480 .+-. 20 92 .+-. 7 .sup.a 16 .+-. 6 .sup.a 4.8 .+-. 0.9 7.26 .+-. 0.08 .sup.a 7.9 .+-. 1.7 .sup.a P5 H0 474 .+-. 49 115 .+-. 16 48 .+-. 9 12.8 .+-. 6.4 7.33 .+-. 0.04 3.4 .+-. 1.5 H1 498 .+-. 26 99 .+-. 22 32 .+-. 8 11.4 .+-. 2.7 7.28 .+-. 0.06 5.4 .+-. 1.4 H2 510 .+-. 42 101 .+-. 18 23 .+-. 4 .sup.b 9.2 .+-. 1.9 7.32 .+-. 0.07 5.5 .+-. 1.6 H3 517 .+-. 62 93 .+-. 21 .sup.b 20 .+-. 7 .sup.b 6.0 .+-. 0.8 7.29 .+-. 0.11 5.9 .+-. 1.7 .sup.b H4 510 .+-. 26 89 .+-. 10 .sup.b 15 .+-. 6 .sup.b 5.0 .+-. 1 .0 7.28 .+-. 0.12 .sup.b 7.4 .+-. 1.8 .sup.b .sup.a p < 0.05 P1 vs Controls .sup.b p < 0.05 P5 vs Controls
CLP Model
[0204] As the severity of the Inventors' model was at its highest 16 to 20 hours after the completion of the CLP, the Inventors chose to investigate animals by the 16.sup.th hour. Importantly, there were no deaths before this time point. Although all animals were fluid resuscitated, none received antibiotics in order to strictly consider the role of the peptides.
[0205] There was a dramatic decline in arterial pressure in the control animals over time, and by H24 systolic, diastolic and mean arterial pressures were 58.+-.7 mmHg, 25.+-.4 mmHg and 38.+-.2 mmHg respectively. This decrease was almost totally abolished with P1 or P5 treatment with no significant difference between H16 and H24 (FIG. 15). There was no difference between P1 and P5 treated rats.
[0206] TREM-1 peptides also prevented the aortic and mesenteric blood flows decrease observed in control animals (Table 7). The protective effect on mesenteric blood flow alterations was even higher under P5 treatment. The relative preservation of blood flows was not related to an increased heart rate, since the latter was rather slower than in control animals (Table 7).
[0207] The progressive metabolic acidosis that developed in control rats was attenuated by the P1 peptide, and almost abrogated by P5. The same protective trend was observed for arterial lactate elevation with a more pronounced effect of P5 (Table 7).
TABLE-US-00009 TABLE 7 Hemodynamic and selected biochemical parameters during CLP polymicrobial sepsis Heart Aortic Mesenteric Rate blood flow blood flow Bicarbonate Lactate (bpm) (mL/min) (mL/min) pH (mmol/L) (mmol/L) Control H16 516 .+-. 44 .sup.a,b 38 .+-. 10 10.6 .+-. 3.0 .sup.b 7.31 .+-. 0.07 .sup.b 6.9 .+-. 2.7 4.7 .+-. 1.5 .sup.b H20 543 .+-. 35 .sup.a,b 9 .+-. 11 .sup.a,b 4.3 .+-. 1.5 .sup.b 7.23 .+-. 0.05 .sup.a,b 12.0 .+-. 5.6 .sup.a,b 8.5 .+-. 1.4 .sup.a,b H24 480 .+-. 20 14 .+-. 9 .sup.a,b 2.5 .+-. 0.7 .sup.b 7.17 .+-. 0.01 .sup.a,b 10.3 .+-. 3.3 .sup.a 10.8 .+-. 1.9 .sup.a,b P1 H16 462 .+-. 16 .sup.a 41 .+-. 12 13.5 .+-. 7.2 7.32 .+-. 0.04 16.8 .+-. 4.4 4.9 .+-. 0.4 H20 480 .+-. 30 .sup.a 28 .+-. 17 .sup.a 5.3 .+-. 3.0 .sup.c 7.31 .+-. 0.18 .sup.a 16.0 .+-. 5.4 .sup.b 5.3 .+-. 1.1 .sup.a,c H24 420 .+-. 30 22 .+-. 16 .sup.a 4.5 .+-. 2.1 .sup.c 7.24 .+-. 0.06 .sup.a,c 11.2 .+-. 0.8 .sup.c 6.8 .+-. 0.9 .sup.a,c P5 H16 460 .+-. 17 .sup.b 41 .+-. 14 15.3 .+-. 3.5 .sup.b 7.35 .+-. 0.01 .sup.b 18.6 .+-. 2.0 3.3 .+-. 0.4 .sup.b H20 500 .+-. 17 .sup.b 31 .+-. 5 .sup.b 11.0 .+-. 6.9 .sup.b,c 7.34 .+-. 0.01 .sup.b 18.0 .+-. 0.9 .sup.a 3.6 .+-. 0.9 .sup.b,c H24 510 .+-. 20 28 .+-. 8 .sup.b 8.5 .+-. 3.5 .sup.b,c 7.36 .+-. 0.01 .sup.b,c 17.1 .+-. 0.9 .sup.a,c 4.9 .+-. 1.1 .sup.b,c .sup.a p < 0.05 P1 vs Controls .sup.b p < 0.05 P5 vs Controls .sup.c p < 0.05 P5 vs P1
[0208] Both P1 and P5 induced a decrease in TNF-.alpha. production, again with a stronger effect of P5. By H20, plasmatic TNF-.alpha. was almost undetectable under P5 treatment whereas it remained elevated in the other groups of animals (FIG. 16).
[0209] Nitrite/nitrate concentrations were increased in control animals but remained at a low level in both TREM-1 peptides treated groups (FIG. 17).
[0210] A protective action of both P5 and P1 on hemodynamics was thus observed in septic rats. Both arterial pressure and blood flows were preserved, independently of heart rate. Moreover, modulation of TREM-1 signalling reduced, although not completely, cytokine production and protected septic animals from hyper-responsiveness. The fact that the cytokine production was not totally inhibited is a crucial point. Indeed, although inflammatory cytokines such as TNF-.alpha. are considered deleterious, they also display beneficial effects in sepsis as underlined by the fatal issue of peritonitis models in animals with impaired TNF-.alpha. responses.
[0211] The activation of iNOS observed during septic shock leads to the production of large amount of NO that partly explains some of the peripheral vascular disorders (notably vasodilation and hypotension). On the myocardium itself, most of the action of NO is mediated by an activation of the soluble guanylate-cyclase responsible for the production of cGMP which impairs the effect of cytosolic calcium on contraction. Cyclic GMP is also able to stimulate the activity of some phosphodiesterases. The subsequent decrease of intracellular cAMP levels could explain the ability of NO to attenuate the effects of beta adrenergic stimulation. The preservation of arterial pressure could therefore be partly explained by a lessened production of NO, as reflected by the lower concentrations of plasma nitrite/nitrate in TREM-1 peptides treated animals.
[0212] The decrease in inflammatory cytokine production could partly explain the effect noted on blood flows. Indeed, although the list of potential cytokine mediators of myocardial depression is long, TNF-.alpha. and IL-1.beta. have been shown to be good candidates Both these latter cytokines depressed myocardial contractility in vitro or ex vivo. Moreover, the neutralization or removal of TNF-.alpha. or IL-1.beta. from human septic serum partly abrogates the myocardial depressant effect in vitro and in vivo. Although P1 and P5 had an identical action on blood flows and arterial pressure during endotoxinemia, their action on cytokine production differed with only a slight effect of P1 on plasma TNF-.alpha. and IL-1.beta. concentrations. The protective role of the TREM-1 peptides could therefore be only partly related to their action on cytokine release, or involve redundant pathways.
[0213] The modulation of the TREM-1 pathway by the use of small synthetic peptides had beneficial effects on haemodynamic parameters during experimental septic shock in rats, along with an attenuation of inflammatory cytokine production.
[0214] In summary, these data show that the TREM-1 peptides of the invention 1) efficiently protect subject animals from sepsis-related hemodynamic deterioration; 2) attenuate the development of lactic acidosis; 3) modulate the production of such pro-inflammatory cytokines as TNF-.alpha. and IL-1.beta. and 4) decrease the generation of nitric oxide. Thus TREM-1 peptides are potentially useful in the restoration of haemodynamic parameters in patients with sepsis, septic shock or sepsis-like conditions and therefore constitute a potential treatment for the aforesaid conditions.
Example 5: hP5 Activity in a Murine Model of Sepsis: Endotoxin-Induced Septic Shock.
[0215] The activity of human P5 (hP5) was investigated in a murine model. hP5 differs from mP5 according to ClustalW comparison as set our below:
TABLE-US-00010 Length Length SeqA Name (aa) SeqB Name (aa) Score 1 hP5 17 2 mP5 17 82 hP5 LQVEDSGLYQCVIYQPP 17 mP5 LQVTDSGLYRCVIYHPP 17 *** *****:****:**
[0216] For the experiment, male BALB/c mice (19-21 g) were randomly grouped (15 mice per group) and injected intraperitoneally (i.p.) with 200 microg of LPS from E. coli 0111:64 (Sigma). A blinded investigator performed all injections. 200 microliters of TREM-1 peptides dissolved in water 10% DMSO, 9% Solutol were administered intraperitoneally at -1 h, 0 h, +1 h, +4 h prior and after LPS injection. Viability of treated mice was monitored twice a day for 7 days.
[0217] To then assess the ability of hP5 peptide to protect mice from LPS-induced endotoxaemia, the inventors administered at -1 h, 0 h, +1 h, +4 h prior and after LPS injection a lethal dose of lipopolysaccharide (LPS) (FIG. 18). Lethality was monitored overtime and compared with animals that had received vehicle alone. hP5 injection confers high protection, with 80% of the animals still alive 7 days after LPS injection, as compared with no survivors in the control group (p<0.0003). The human P5 peptide shows >80% identity with mP5.
[0218] The results summarised in FIG. 18 clearly demonstrate that the human P5 protects mice from lethal shock.
Example 6 hP5 Activity in a Murine Model of Sepsis: Cecal Ligation and Puncture Model
[0219] For the experiment the mice underwent a standardized preparation for laparotomy (anaesthesia with 2% inhaled isoflurane in oxygen, shaving with animal clippers, alcohol scrub). A 1 cm incision was made on the midline. The cecum was exposed and will be tightly ligated at 50-80% over its base with a 4-0 silk suture avoiding bowel obstruction. The cecum was then punctured once with a 23 G needle. The cecum was gently squeezed until feces be just visible through the puncture, and placed again in the abdominal cavity. The incision was thereafter be closed with a 4-0 silk. 200 .mu.l of hP5 or its scrambled peptide control were freshly dissolved in water 10% DMSO, 9% solutol and injected intraperitoneally with a 22 G needle at the following time points: -1, 0, +4, +24. No fluid resuscitation was administered. Survival and Moribundity were observed twice a day for 10 days.
[0220] The Inventors then analysed whether hP5 protects against septic shock in the "CLP" model. Mice treated with four doses of hP5 at -1 h, 0 h, +4 h and +24 hours after CLP were protected from death as compared to control treated mice. At 72 hours after CLP, 73.3% of the mice injected with hP5 survived compared to 60% of mice treated with scrambled peptide. At 10 days after CLP, 66.6% of the hP5 treated mice were still alive compared to 60% of the control group, suggesting that hP5 could provide lasting protection (FIG. 19).
Example 7 The Human TREM-1 Peptide P5 (hP5) Attenuates Established Intestinal Inflammation in the DSS Colitis Model
[0221] The purpose of this example is to identify, characterize and document the therapeutic potential of the TREM-1 derived peptide hP5, in an experimental model of colitis in mice.
[0222] On day 0 of the study, the water supply was removed and replaced with 3% Dextran Sulfate Sodium (DSS) From day 0 to day 6, this solution was the only source of fluids. Water was administered for the rest of the experiment (day 7-11). Healthy controls received water only.
[0223] From day 3 to day 10 mice were treated with either hP5, or a sequence-scrambled control peptide. Peptides were dissolved in DMSO at 10 mg/ml and stored at +4.degree. C. Before administration, the stock solution was diluted 1:10 in water 10% Solutol.RTM. HS 15 (BASF). Final vehicle concentrations: 1& DMSO, 10% solutol in water. 200 .mu.l of these freshly prepared solutions of hP5, its scrambled control or vehicle were injected intraperitoneally with a 25 G needle.
[0224] Animals were weighted daily and monitored on days 3, 4, 5, 6, 7 and 10 for rectal bleeding and stool consistency. For each group, the disease activity index (DAI) was determined by evaluating changes in weight, stool consistency and presence of gross blood during the study, as described below:
TABLE-US-00011 Weight loss Blood in Score (%) Stool consistency stool 0 <1 Normal Negative 1 1-4.9 Soft +/- 2 5-9.9 Mixed (soft and liquid) + 3 10-15 Liquid ++ 4 >15 Diarrhea (liquid stools that Gross adhere to the anus) bleeding
[0225] Scoring System for the Disease Activity Index (DAI). Individual scores for each parameter are added and then divided by three to give a DAI score for each mouse.
[0226] To determine the presence of occult blood in stool, a pea-sized stool sample was placed on a slide. Then two drops of reagent (Hemocult Sensa, Beckman Coulter) were placed onto the stool sample on the slide and a change of colour was observed. The presence of occult blood was graded using a score of 0 for no colour; 1 for a very light blue (+/-) colour taking over 30 seconds to appear, 2 for a blue colour developing in 30 seconds or more (+); 3 for an immediate change in colour (++) and 4 for gross blood observable on the slide.
[0227] On day 11 mice were euthanized by cervical dislocation to allow colon length evaluation. An incision was done in the abdomen to expose the colon. The stool in the colon was removed flushing with saline. The entire colon from cecum to anus was removed and the length was measured and reported as colon length (cm).
[0228] Data were analyzed using GraphPad Prism. Results are given as means .+-. standard error of the mean. The BW score, stool score and blood score were analyzed using two-way ANOVA test, Bonferroni post test. Colon length was analysed using one-way ANOVA, Dunn's post test. * P.ltoreq.0.05; **P.ltoreq.0.01; *** P.ltoreq.0.001
[0229] mP5 is a mTREM-1 derived peptide whose efficacy has been proven in several models of sepsis. In this study, the investigators have tested the efficacy of the human orthologue of mP5, compared to its scrambled peptide control, in the above described DSS-induced colitis model. All peptides were administered in a therapeutic fashion, 3 days after initiation of DSS treatment.
[0230] In all animals, body weight, haemoccult or presence of gross blood and stool consistency were monitored. Bloody stools were observed from day 3 onwards, loose stools and weight loss appeared beginning from day 4-5. These comprehensive functional measures, that were somewhat analogous to clinical symptoms observed in human IBD, are summarized by the Disease Activity Index, as shown in FIG. 20. This scoring method, validated by repeated studies, showed minimum variations and correlates well with more specific measures of inflammation.
[0231] None of the control animals showed disease activity. DAI peaked at day 5-6 and regressed upon DSS removal. As soon as colitis was established (day 5), hP5 administration significantly ameliorated stool consistency (FIG. 23) and colon bleeding (FIG. 22), as shown by approximately a 50% DAI inhibition, when compared to mice treated with DSS+vehicle or DSS+scrambled peptide (1.95.+-.0.170 vs. 2.92.+-.0.054, p.ltoreq.0.001 or 3.00.+-.0.088, p.ltoreq.0.001, respectively).
[0232] Protection lasted during the whole treatment. Notably at day 10, in the hP5-treated group both stool score and hemoccult were normal (FIGS. 23 and 22), while the groups treated with DSS+vehicle and DSS+scrambled peptide still showed clinical signs of inflammation (blood score: DSS+hP5=0.43.+-.0.202 vs DSS+vehicle=1.25.+-.0.164; p.ltoreq.0.01; DSS+hP5=0.43.+-.0.202 vs DSS+scrambled peptide=1.75.+-.0.250; p.ltoreq.0.001. Stool score: DSS+hP5=0.71.+-.0.474 vs DSS+vehicle=1.75.+-.0.250 p.ltoreq.0.05; DSS+hP5=0.71.+-.0.474 vs DSS+scrambled peptide=2.38.+-.0.324; p.ltoreq.0.001).
[0233] We also monitored weight loss associated with the development of colitis. FIG. 21 represents the percentage of weight loss, expressed as a score. Mice started loosing weight at day 4 and the maximal weight loss was reached at day 8. hP5 treatment significantly reduced weight loss in comparison with DSS+scrambled, from day 8 on (day 10: hP5=2.00.+-.0.436 vs DSS+scrambled pep. 3.50.+-.0.189; p.ltoreq.0.001). At day 11 mice were euthanized and the colon length of each mouse measured from the anus to the end of the cecum (FIG. 24). DSS-induced colon inflammation caused a 30% colon shortening when compared to naive animals. In the hP5-treated group, colon shortening was significantly ameliorated when compared to control group (hP5=7.41.+-.0.237 vs DSS+vehicle=5.96.+-.0.230; p.ltoreq.0.05). In conclusion, blocking TREM-1 with a human TREM-1 derived peptide attenuates intestinal inflammation even when the peptide is administered after the appearance of the clinical signs of colitis. This finding indicates that the human TREM-1 -derived peptide efficiently blocks interaction of the mouse TREM-1 receptor with its endogenous ligand.
REFERENCES
[0234] 1. Aderem, A, and R. J. Ulevitch, R. J. 2000. Toll-like receptors in the induction of the innate immune response. Nature 406:782-786. [0235] 2. Thoma-Uszynski, S., S. Stenger, O. Takeuchi, M. T. Ochoa, P. A. Engele, P. A. Sieling, P. F. Barnes, M. Rollinghoff, P. L. Bolcskei, and M. Wagner. 2001. Induction of direct antimicrobial activity through mammalian Toll-like receptors. Science 291:1544-1549. [0236] 3. Medzhitov, R., and C Jr. Janeway. 2000. Innate immunity. N. Engl. J. Med. 343:338-344. [0237] 4. Cohen, J. 2002. The immunopathogenesis of sepsis. Nature 420:885-91. [0238] 5. Hotchkiss, R. S., and I. E. Karl. 2003. The pathophysiology and treatment of sepsis. N. Engl. J. Med. 348:138-50. [0239] 6. Vincent, J. L., Q. Sun, and M. J. Dubois. 2002. Clinical trials of immunomodulatory therapies in severe sepsis and septic shock. Clin. Infect. Dis. 34:1084-1093. [0240] 7. Riedemann, N. C., R. F. Guo, and P. Ward. 2003. Novel strategies for the treatment of sepsis. Nat. Med. 9:517-524. [0241] 8. Fisher, C. J. Jr., J. M. Agosti, S. M. Opal, S. F. Lowry, R. A. Balk, J. C. Sadoff, E. Abraham, R. M. Schein, and E. Benjamin. 1996. Treatment of septic shock with the tumor necrosis factor receptor:Fc fusion protein. The Soluble TNF Receptor Sepsis Study Group. N. Engl. J. Med 334:1697-702. [0242] 9. Echtenacher, B., W. Falk, D. N. Mannel, and P. H. Krammer. 1990. Requirement of endogenous tumor necrosis factor/cachectin for recovery from experimental peritonitis. J. Immunol. 145:3762-3766. [0243] 10. Echtenacher, B., K. Weigl, N. Lehn, and D. N. Mannel. 2001. Tumor necrosis factor-dependent adhesions as a major protective mechanism early in septic peritonitis in mice. Infect. Immun. 69:3550-5. [0244] 11. Eskandari, M. K., G. Bolgos, C. Miller, D. T. Nguyen, L. E. DeForge, and D. G. Remick. 1992. Anti-tumor necrosis factor antibody therapy fails to prevent lethality after cecal ligation and puncture or endotoxemia. J. Immunol. 148:2724-30. [0245] 12. Bouchon, A., J. Dietrich, and M. Colonna. 2000. Inflammatory responses can be triggered by TREM-1, a novel receptor expressed on neutrophils and monocytes. J. Immunol. 164:4991-4995. [0246] 13. Colonna, M., and F. Facchetti. 2003. TREM-1 (triggering receptor expressed on myeloid cells): a new player in acute inflammatory responses. J. Infect. Dis. 187 (Suppl):5297-301. [0247] 14. Colonna, M. 2003. TREMs in the immune system and beyond. Nat. Rev. Immunol. 6:445-453. [0248] 15. Nathan, C., and A. Ding. 2001. TREM-1: a new regulator of innate immunity in sepsis syndrome. Nat. Med. 7:530-2. [0249] 16. Bouchon, A., F. Facchetti, M. A. Weigand, and M. Colonna. 2001. TREM-1 amplifies inflammation and is a crucial mediator of septic shock. Nature 410:1103-1107. [0250] 17. Bleharski, J. R., V. Kiessler, C. Buonsanti, P. A. Sieling, S. Stenger, M. Colonna, and R. L. Modlin. 2003. A role for Triggering Receptor Expressed on Myeloid cells-1 in host defense during the early-induced and adaptive phases of the immune response. J. Immunol. 170:3812-3818. [0251] 18. Gibot, S., A. Cravoisy, B. Levy, M. C. Bene, G. Faure, and P. E. Bollaert. 2004. Soluble triggering receptor expressed on myeloid cells and the diagnosis of pneumonia. N. Engl. J. Med. 350:451-8. [0252] 19. Gingras, M. C., H. Lapillonne, and J. F. Margolin. 2001. TREM-1, MDL-1, and DAP12 expression is associated with a mature stage of myeloid development. Mol. Immunol. 38:817-24. [0253] 20. Dinarello, C. A. 1997. Proinflammatory and anti-inflammatory cytokines as mediators in the pathogenesis of septic shock. Chest 112 (suppl):521-9. [0254] 21. Bone, R. C., R. A Balk, F. B. Cerra, R. P. Dellinger, W. A. Knauss, R. M. Schein, and W. J. Sibbald. 1992. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 101:1644-55. [0255] 22. Warren, H. S. 1997. Strategies for the treatment of sepsis. N. Engl. J. Med. 336:952-3. [0256] 23. Stone, R. 1994. Search for sepsis drugs goes on despite past failures. Science 264:365-7. [0257] 24. Cohen, J. 2001. TREM-1 in sepsis. Lancet 358:776-8. [0258] 25. Begun, N. A., K. Ishii, M. Kurita-Taniguchi, M. Tanabe, M. Kobayashi, Y. Moriwaki, M. Matsumoto, Y. Fukumori, I. Azuma, K. Toyoshima, and T. Seya. 2004. Mycobacterium bovis BCG cell wall-specific differentially expressed genes identified by differential display and cDNA substraction in human macrophages. Infect. Immun. 12:937-48. [0259] 26. Keane, J., S. Gershon, R. P. Wise, E. Mirabile-Levens, J. Kasznica, W. D. Schwieterman, J. N. Siegel, and M. M. Braun. 2001. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N. Engl. J. Med. 345:1098-104. [0260] 27. Radaev, S., M. Kattah, B. Rostro, M. Colonna, and P. Sun. 2003. Crystal structure of the human myeloid cell activating receptor TREM-1. Structure 11:1527-35. [0261] 28. Lantz, M., U. Gullberg, and E. Nilsson. 1990. Characterization in vitro of a human tumor necrosis factor binding protein. A soluble form of tumor necrosis factor receptor. J. Clin. Invest. 86:1396-1401. [0262] 29. van Zee, K. J., T. Kohno, and E. Fischer. 1992. Tumor necrosis soluble receptors circulate during experimental and clinical inflammation and can protect against excessive tumor necrosis factor .alpha. in vitro and in vivo. Proc. Natl. Acad. Sci. USA 89:4845-4853. [0263] 30. Collart, M. A., P. Baeuerle, and P. Vassalli. 1990. Regulation of tumor necrosis factor alpha transcription in macrophages. Involvement of four NF.kappa.B motifs and constitutive and inducible form of NF.kappa.B. Mol. Cell Biol. 10:1498-506. [0264] 31. Hiscott, J. M., J. J. Garoufalis, A. Roulston, I. Kwan, N. Pepin, J. Lacoste, H. Nguyen, G. Bensi, and M. Fenton. 1993. Characterization of a functional NF.kappa.B site in the human IL-1P promoter: evidence for a positive autoregulatory loop. Mol. Cell Biol. 13:6231-40. [0265] 32. Urban, M. B., R. Schreck, and P.A. Baeuerle. 1991. NF.kappa.B contacts DNA by a heterodimer of the p50 and p65 subunit. EMBO J. 10:1817-25. [0266] 33. Lolis, E., and R. Bucala. 2003. Therapeutic approaches to innate immunity: severe sepsis and septic shock. Nat. Rev. Drug. Discov. 2:635-45. [0267] 34. Gibot, S., M. N. Kolopp-Sarda, M. C. Bene, A. Cravoisy, B. Levy, G. Faure, and P. E. Bollaert. 2004. Plasma level of a triggering receptor expressed on myeloid cells-1: its diagnostic accuracy in patients with suspected sepsis. Ann. Intern. Med. 141:9-15. [0268] 35. Schenk M, Bouchon A, Seibold F, and Mueller C. 2007.
Sequence CWU 1
1
271234PRTHomo sapiens 1Met Arg Lys Thr Arg Leu Trp Gly Leu Leu Trp
Met Leu Phe Val Ser1 5 10 15Glu Leu Arg Ala Ala Thr Lys Leu Thr Glu
Glu Lys Tyr Glu Leu Lys 20 25 30Glu Gly Gln Thr Leu Asp Val Lys Cys
Asp Tyr Thr Leu Glu Lys Phe 35 40 45Ala Ser Ser Gln Lys Ala Trp Gln
Ile Ile Arg Asp Gly Glu Met Pro 50 55 60Lys Thr Leu Ala Cys Thr Glu
Arg Pro Ser Lys Asn Ser His Pro Val65 70 75 80Gln Val Gly Arg Ile
Ile Leu Glu Asp Tyr His Asp His Gly Leu Leu 85 90 95Arg Val Arg Met
Val Asn Leu Gln Val Glu Asp Ser Gly Leu Tyr Gln 100 105 110Cys Val
Ile Tyr Gln Pro Pro Lys Glu Pro His Met Leu Phe Asp Arg 115 120
125Ile Arg Leu Val Val Thr Lys Gly Phe Ser Gly Thr Pro Gly Ser Asn
130 135 140Glu Asn Ser Thr Gln Asn Val Tyr Lys Ile Pro Pro Thr Thr
Thr Lys145 150 155 160Ala Leu Cys Pro Leu Tyr Thr Ser Pro Arg Thr
Val Thr Gln Ala Pro 165 170 175Pro Lys Ser Thr Ala Asp Val Ser Thr
Pro Asp Ser Glu Ile Asn Leu 180 185 190Thr Asn Val Thr Asp Ile Ile
Arg Val Pro Val Phe Asn Ile Val Ile 195 200 205Leu Leu Ala Gly Gly
Phe Leu Ser Lys Ser Leu Val Phe Ser Val Leu 210 215 220Phe Ala Val
Thr Leu Arg Ser Phe Val Pro225 2302230PRTMus musculus 2Met Arg Lys
Ala Gly Leu Trp Gly Leu Leu Cys Val Phe Phe Val Ser1 5 10 15Glu Val
Lys Ala Ala Ile Val Leu Glu Glu Glu Arg Tyr Asp Leu Val 20 25 30Glu
Gly Gln Thr Leu Thr Val Lys Cys Pro Phe Asn Ile Met Lys Tyr 35 40
45Ala Asn Ser Gln Lys Ala Trp Gln Arg Leu Pro Asp Gly Lys Glu Pro
50 55 60Leu Thr Leu Val Val Thr Gln Arg Pro Phe Thr Arg Pro Ser Glu
Val65 70 75 80His Met Gly Lys Phe Thr Leu Lys His Asp Pro Ser Glu
Ala Met Leu 85 90 95Gln Val Gln Met Thr Asp Leu Gln Val Thr Asp Ser
Gly Leu Tyr Arg 100 105 110Cys Val Ile Tyr His Pro Pro Asn Asp Pro
Val Val Leu Phe His Pro 115 120 125Val Arg Leu Val Val Thr Lys Gly
Ser Ser Asp Val Phe Thr Pro Val 130 135 140Ile Ile Pro Ile Thr Arg
Leu Thr Glu Arg Pro Ile Leu Ile Thr Thr145 150 155 160Lys Tyr Ser
Pro Ser Asp Thr Thr Thr Thr Arg Ser Leu Pro Lys Pro 165 170 175Thr
Ala Val Val Ser Ser Pro Gly Leu Gly Val Thr Ile Ile Asn Gly 180 185
190Thr Asp Ala Asp Ser Val Ser Thr Ser Ser Val Thr Ile Ser Val Ile
195 200 205Cys Gly Leu Leu Ser Lys Ser Leu Val Phe Ile Ile Leu Phe
Ile Val 210 215 220Thr Lys Arg Thr Phe Gly225 230323PRTMus musculus
3Leu Val Val Thr Gln Arg Pro Phe Thr Arg Pro Ser Glu Val His Met1 5
10 15Gly Lys Phe Thr Leu Lys His 20423PRTMus musculus 4Val Ile Tyr
His Pro Pro Asn Asp Pro Val Val Leu Phe His Pro Val1 5 10 15Arg Leu
Val Val Thr Lys Gly 20517PRTArtificial SequenceDescription of
Artificial Sequence Synthetic peptide 5Thr Thr Thr Arg Ser Leu Pro
Lys Pro Thr Ala Val Val Ser Ser Pro1 5 10 15Gly621PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 6Leu
Gln Val Thr Asp Ser Gly Leu Tyr Arg Cys Val Ile Tyr His Pro1 5 10
15Pro Asn Asp Pro Val 20717PRTArtificial SequenceDescription of
Artificial Sequence Synthetic peptide 7Leu Gln Val Thr Asp Ser Gly
Leu Tyr Arg Cys Val Ile Tyr His Pro1 5 10 15Pro823PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 8Leu
Thr Pro Lys His Gly Gln Arg Ser Thr His Val Thr Lys Phe Arg1 5 10
15Val Phe Glu Pro Val Met Leu 20917PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 9Thr
Asp Ser Arg Cys Val Ile Gly Leu Tyr His Pro Pro Leu Gln Val1 5 10
15Tyr1022DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 10ttgtctcaga actccgagct gc 221122DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
11gagacatcgg cagttgactt gg 221222DNAArtificial SequenceDescription
of Artificial Sequence Synthetic primer 12ggacggagag atgcccaaga cc
221322DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 13accagccagg agaatgacaa tg 221422DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
14ggacgacatg gagaagatct gg 221524DNAArtificial SequenceDescription
of Artificial Sequence Synthetic primer 15atagtaatgt cacgcacgat
ttcc 241623PRTHomo sapiens 16Leu Ala Cys Thr Glu Arg Pro Ser Lys
Asn Ser His Pro Val Gln Val1 5 10 15Gly Arg Ile Ile Leu Glu Asp
201723PRTHomo sapiens 17Val Ile Tyr Gln Pro Pro Lys Glu Pro His Met
Leu Phe Asp Arg Ile1 5 10 15Arg Leu Val Val Thr Lys Gly
201821PRTHomo sapiens 18Leu Gln Val Glu Asp Ser Gly Leu Tyr Gln Cys
Val Ile Tyr Gln Pro1 5 10 15Pro Lys Glu Pro His 201917PRTHomo
sapiens 19Leu Gln Val Glu Asp Ser Gly Leu Tyr Gln Cys Val Ile Tyr
Gln Pro1 5 10 15Pro206PRTHomo sapiens 20Arg Pro Ser Lys Asn Ser1
5215PRTHomo sapiens 21Gln Pro Pro Lys Glu1 5226PRTMus musculus
22Arg Pro Phe Thr Arg Pro1 5235PRTMus musculus 23His Pro Pro Asn
Asp1 5244PRTHomo sapiens 24Gln Pro Pro Lys1254PRTMus musculus 25His
Pro Pro Asn126230PRTHomo sapiens 26Met Glu Pro Leu Arg Leu Leu Ile
Leu Leu Phe Val Thr Glu Leu Ser1 5 10 15Gly Ala His Asn Thr Thr Val
Phe Gln Gly Val Ala Gly Gln Ser Leu 20 25 30Gln Val Ser Cys Pro Tyr
Asp Ser Met Lys His Trp Gly Arg Arg Lys 35 40 45Ala Trp Cys Arg Gln
Leu Gly Glu Lys Gly Pro Cys Gln Arg Val Val 50 55 60Ser Thr His Asn
Leu Trp Leu Leu Ser Phe Leu Arg Arg Trp Asn Gly65 70 75 80Ser Thr
Ala Ile Thr Asp Asp Thr Leu Gly Gly Thr Leu Thr Ile Thr 85 90 95Leu
Arg Asn Leu Gln Pro His Asp Ala Gly Leu Tyr Gln Cys Gln Ser 100 105
110Leu His Gly Ser Glu Ala Asp Thr Leu Arg Lys Val Leu Val Glu Val
115 120 125Leu Ala Asp Pro Leu Asp His Arg Asp Ala Gly Asp Leu Trp
Phe Pro 130 135 140Gly Glu Ser Glu Ser Phe Glu Asp Ala His Val Glu
His Ser Ile Ser145 150 155 160Arg Ser Leu Leu Glu Gly Glu Ile Pro
Phe Pro Pro Thr Ser Ile Leu 165 170 175Leu Leu Leu Ala Cys Ile Phe
Leu Ile Lys Ile Leu Ala Ala Ser Ala 180 185 190Leu Trp Ala Ala Ala
Trp His Gly Gln Lys Pro Gly Thr His Pro Pro 195 200 205Ser Glu Leu
Asp Cys Gly His Asp Pro Gly Tyr Gln Leu Gln Thr Leu 210 215 220Pro
Gly Leu Arg Asp Thr225 23027227PRTMus musculus 27Met Gly Pro Leu
His Gln Phe Leu Leu Leu Leu Ile Thr Ala Leu Ser1 5 10 15Gln Ala Leu
Asn Thr Thr Val Leu Gln Gly Met Ala Gly Gln Ser Leu 20 25 30Arg Val
Ser Cys Thr Tyr Asp Ala Leu Lys His Trp Gly Arg Arg Lys 35 40 45Ala
Trp Cys Arg Gln Leu Gly Glu Glu Gly Pro Cys Gln Arg Val Val 50 55
60Ser Thr His Gly Val Trp Leu Leu Ala Phe Leu Lys Lys Arg Asn Gly65
70 75 80Ser Thr Val Ile Ala Asp Asp Thr Leu Ala Gly Thr Val Thr Ile
Thr 85 90 95Leu Lys Asn Leu Gln Ala Gly Asp Ala Gly Leu Tyr Gln Cys
Gln Ser 100 105 110Leu Arg Gly Arg Glu Ala Glu Val Leu Gln Lys Val
Leu Val Glu Val 115 120 125Leu Glu Asp Pro Leu Asp Asp Gln Asp Ala
Gly Asp Leu Trp Val Pro 130 135 140Glu Glu Ser Ser Ser Phe Glu Gly
Ala Gln Val Glu His Ser Thr Ser145 150 155 160Arg Asn Gln Glu Thr
Ser Phe Pro Pro Thr Ser Ile Leu Leu Leu Leu 165 170 175Ala Cys Val
Leu Leu Ser Lys Phe Leu Ala Ala Ser Ile Leu Trp Ala 180 185 190Val
Ala Arg Gly Arg Gln Lys Pro Gly Thr Pro Val Val Arg Gly Leu 195 200
205Asp Cys Gly Gln Asp Ala Gly His Gln Leu Gln Ile Leu Thr Gly Pro
210 215 220Gly Gly Thr225

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012
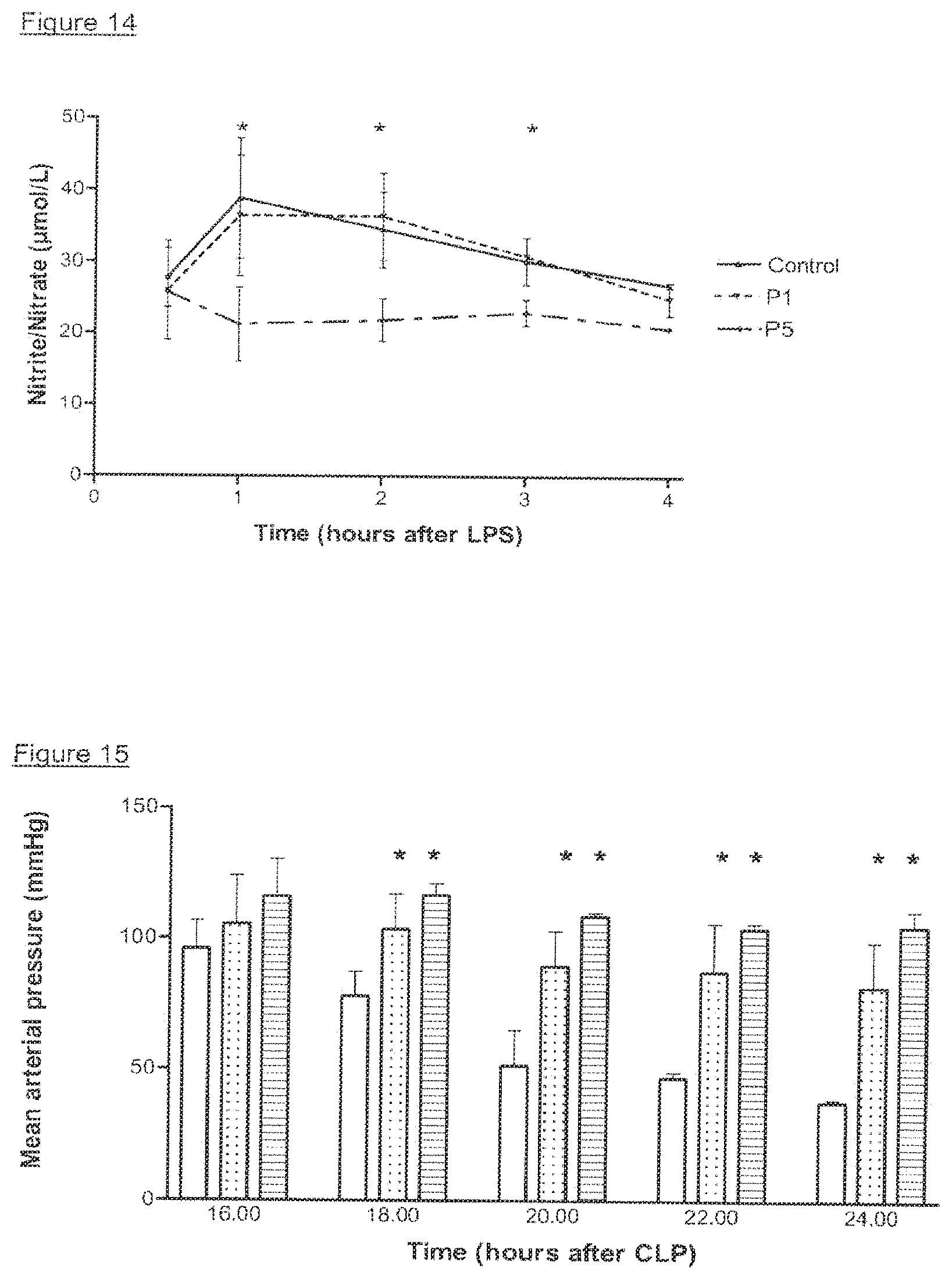
D00013
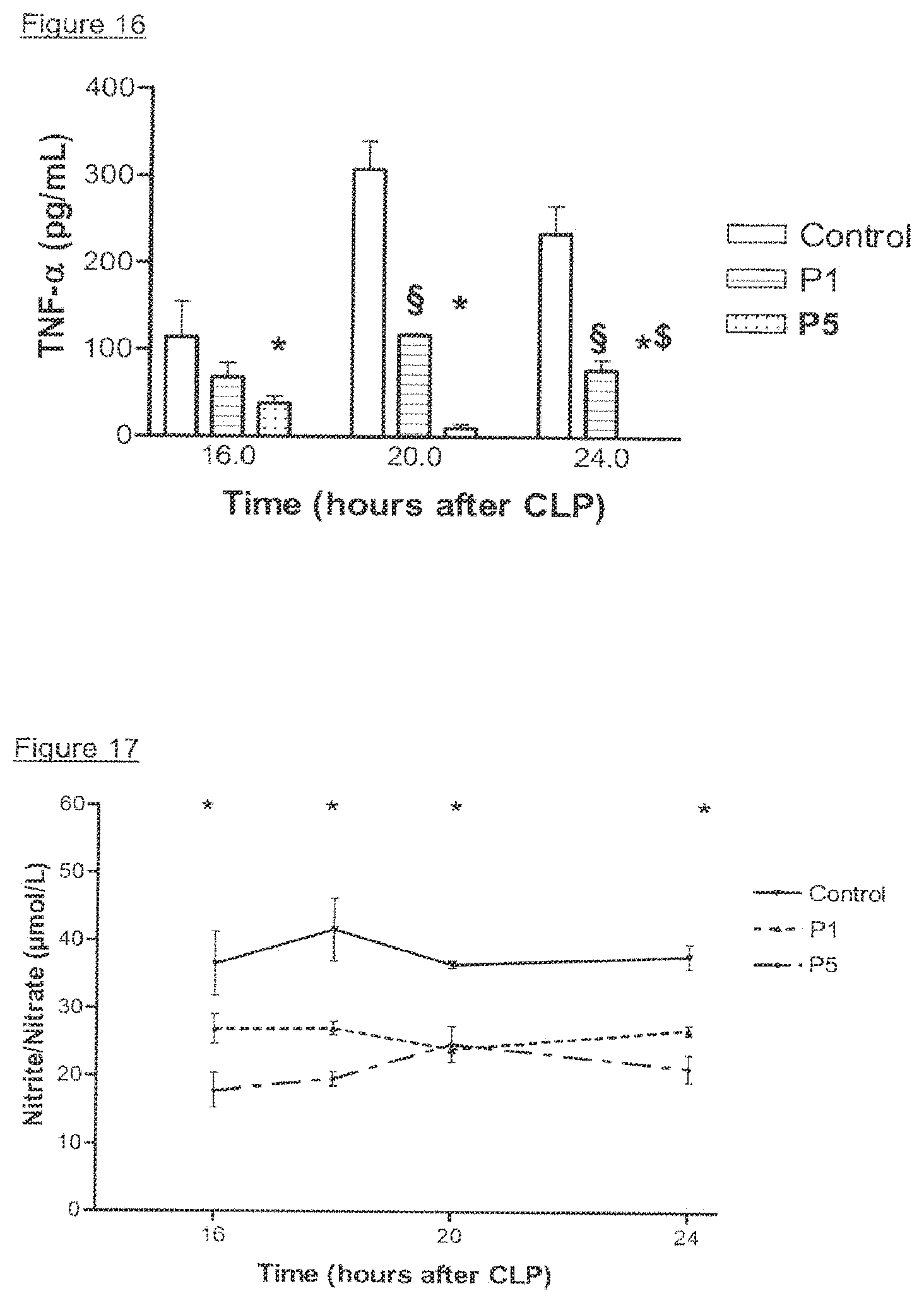
D00014
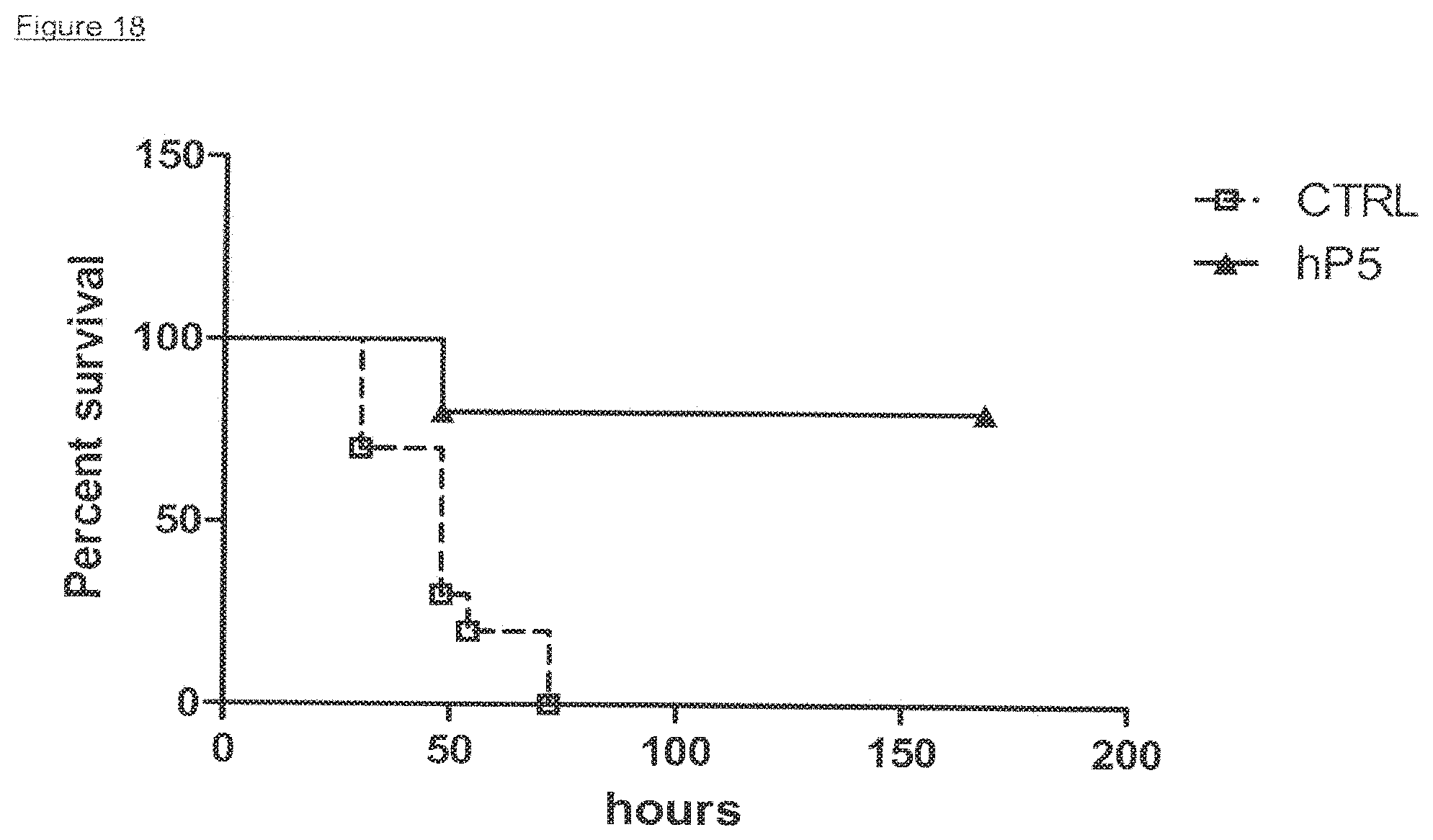
D00015

D00016

D00017
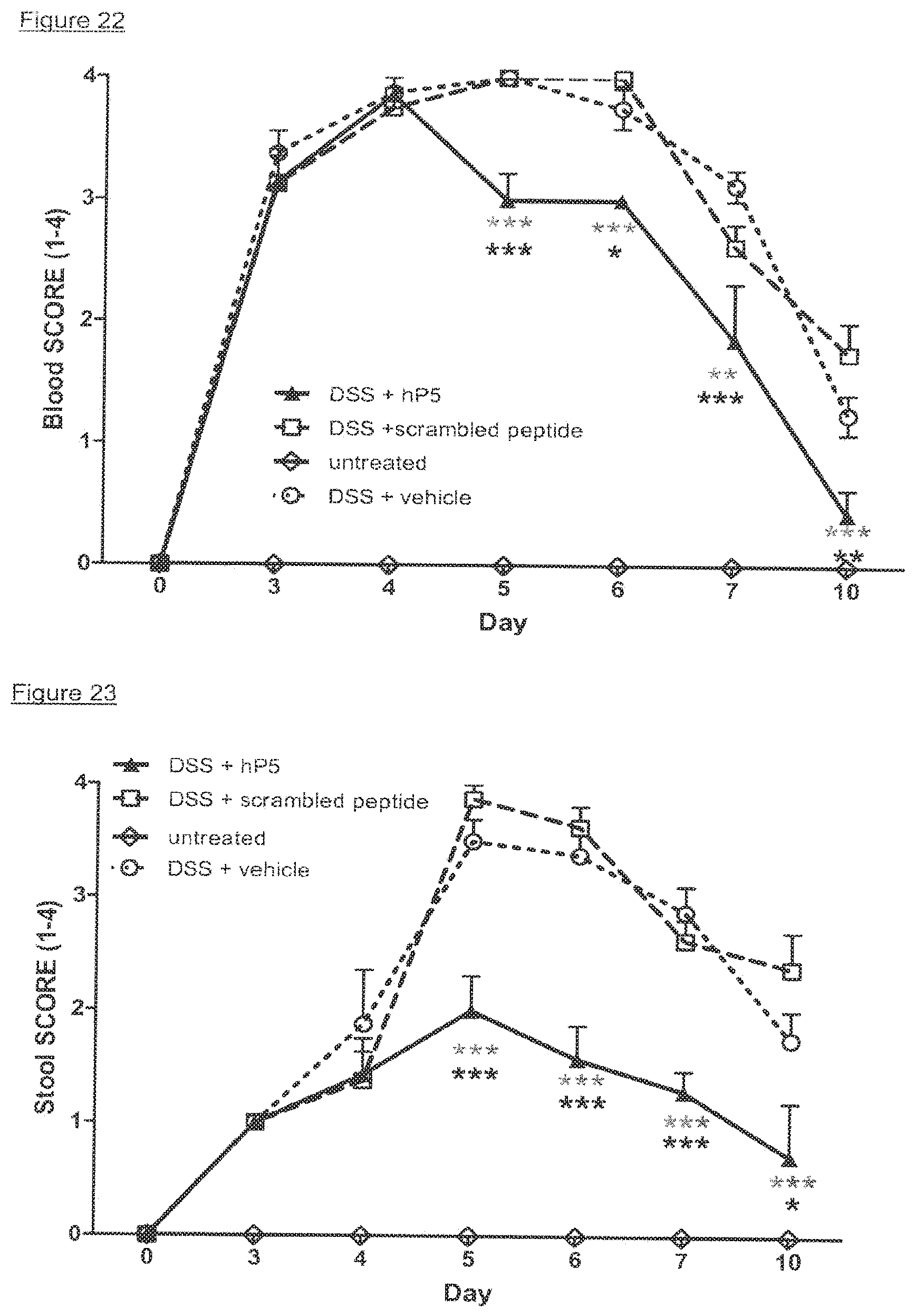
D00018
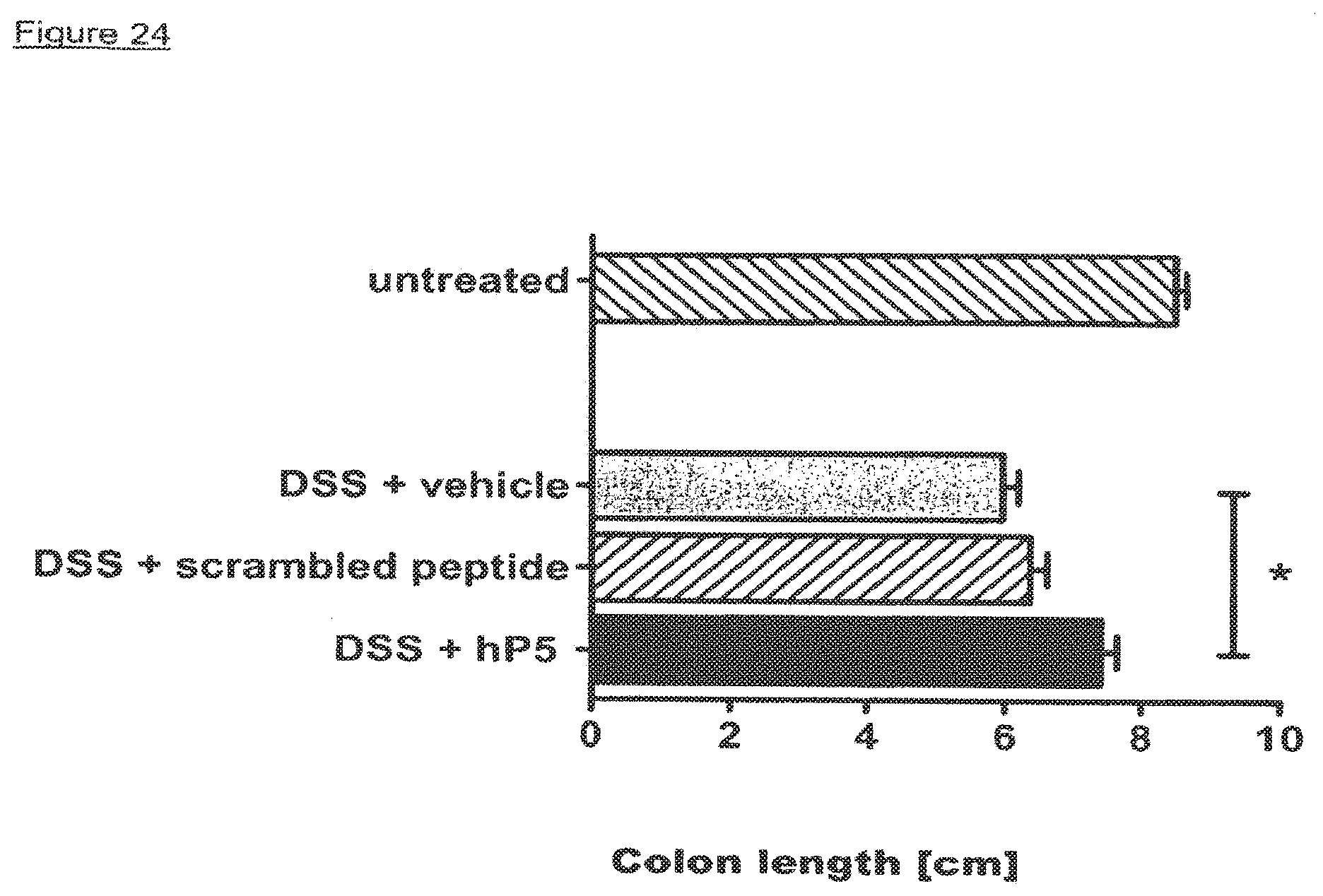
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.