Aligned Nematic Elastomer
Gleeson; Helen Frances ; et al.
U.S. patent application number 16/756809 was filed with the patent office on 2020-07-30 for aligned nematic elastomer. The applicant listed for this patent is University of Leeds. Invention is credited to Helen Frances Gleeson, Devesh Arvind Mistry.
| Application Number | 20200239774 16/756809 |
| Document ID | 20200239774 / US20200239774 |
| Family ID | 60481580 |
| Filed Date | 2020-07-30 |
| Patent Application | download [pdf] |









| United States Patent Application | 20200239774 |
| Kind Code | A1 |
| Gleeson; Helen Frances ; et al. | July 30, 2020 |
ALIGNED NEMATIC ELASTOMER
Abstract
There is provided the use of an aligned nematic elastomer to form a material having auxetic properties wherein the aligned nematic material has a mechanical Freedericksz transition. Also provided is a method of producing an aligned nematic elastomer for said use.
| Inventors: | Gleeson; Helen Frances; (Leeds, West Yorkshire, GB) ; Mistry; Devesh Arvind; (Leeds, West Yorkshire, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 60481580 | ||||||||||
| Appl. No.: | 16/756809 | ||||||||||
| Filed: | October 18, 2018 | ||||||||||
| PCT Filed: | October 18, 2018 | ||||||||||
| PCT NO: | PCT/GB2018/053018 | ||||||||||
| 371 Date: | April 16, 2020 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C09K 2019/0448 20130101; C09K 19/12 20130101; C09K 2019/3004 20130101; C09K 19/3003 20130101; C09K 2019/548 20130101; C09K 2019/122 20130101; C09K 19/542 20130101; C09K 19/54 20130101; C09K 2019/3009 20130101 |
| International Class: | C09K 19/12 20060101 C09K019/12; C09K 19/30 20060101 C09K019/30; C09K 19/54 20060101 C09K019/54 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Oct 19, 2017 | GB | 1717174.5 |
Claims
1. A method of producing a material having auxetic properties, said method comprising the steps of: a) providing an aligned nematic elastomer; and b) forming an aligned nematic material using the aligned nematic elastomer, wherein the aligned nematic material has a mechanical Freedericksz transition, thereby producing a material having auxetic properties.
2. The method according to claim 1 wherein the auxetic properties enable the material to be used in a medical device or in a biomedical application.
3. The method according to claim 1 wherein the auxetic properties enable the material to be used in a piezoelectric sensor or actuator, or in a micro- or nano-mechanical or electromechanical device.
4. The method according to claim 1 wherein the auxetic properties enable the material to be used in composite materials as reinforcements, or in personal protection clothing.
5. The method according to claim 1 wherein the aligned nematic elastomer comprises a monodomain liquid crystal elastomer.
6. The method according to claim 5 wherein the monodomain liquid crystal elastomer comprises; a polymeric component; a liquid crystal mesogen component; and a cross-linker component, wherein the liquid crystal mesogen component is physically linked to the polymeric component.
7. The method according to claim 6 wherein the liquid crystal mesogen component comprises a liquid crystal core component selected from the group consisting of aromatic rings, aliphatic rings, poly aromatic rings, poly aliphatic rings, phenyls, biphenyls, benzenes, and combinations thereof.
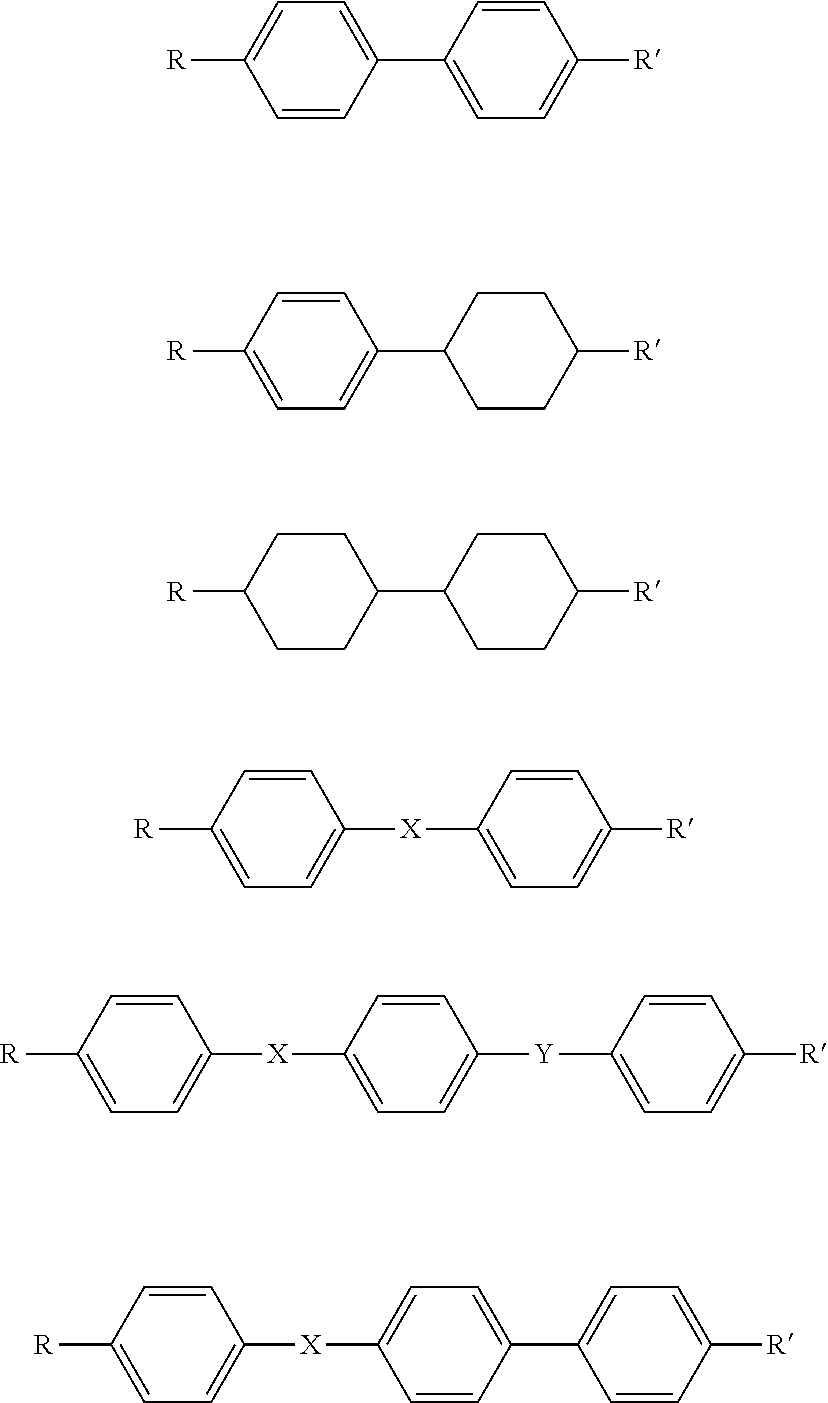
8. The method according to claim 7 wherein the liquid crystal core component is selected from one or more of the following systems: ##STR00003## wherein R and R' are independently selected from the group consisting of alkyl, alkoxy, halide, --NO.sub.2 or --CN and wherein the alkyl and alkoxy groups may be bivalent when forming part of the linking group which connects the liquid crystal core component to the polymeric component; and X and Y are independently selected from the group consisting of --CH.dbd.CH--, --C.ident.--, --CH.dbd.N--, --N.dbd.N--, or --C(O)O--.
9. The method according to claim 6 wherein the polymeric component is formed from both mesogenic and non-mesogenic components.
10. The method according to claim 9 wherein the polymeric component comprises an acrylate polymer and the non-mesogenic component comprises 2-ethylhexyl acrylate.
11. The method according to claim 6 wherein the crosslinker component comprises a mesogenic component.
12. The method according to claim 9 wherein the mesogenic component is 6-(4-cyano-biphenyl-4'-yloxy)hexyl acrylate, the crosslinker component is 1,4-bis-[4-(6-acryloyloxyhexyloxy)benzoyloxy]-2-methylbenzene, and the non-mesogenic component is 2-ethylhexyl acrylate.
13. A method of producing an aligned nematic elastomer, said method comprising the steps of: a) applying an aligning means to a substrate b) applying liquid crystal elastomer components to the substrate and allowing them to form an aligned nematic phase c) curing the liquid crystal elastomer components to form an aligned nematic elastomer.
14. The method according to claim 13 wherein the aligning means is an aligning force which is applied by brushing the substrate.
Description
[0001] The present invention relates to the use of an aligned nematic elastomer, in particular to its use in forming material having auxetic properties.
[0002] In conventional materials, when the material is stretched, the material simultaneously becomes thinner in cross-section. Similarly, if a conventional material is compressed, the material expands laterally. These conventionally behaved materials have a positive Poisson's ratio where the Poisson's ratio is described as the negative ratio of the proportional decrease in a lateral measurement to the proportional increase in length in a sample of material that is elastically stretched. Materials with auxetic properties on the other hand have a negative Poisson's ratio. On stretching, the materials become thicker in one or both of the directions perpendicular to the applied force. Auxetic materials are of particular interest because of this unusual behaviour under deformation. Auxetic materials exist in nature, for example some minerals and a large number of cubic elemental metals, but synthetic auxetic materials were only developed in the 1980s. Macroscopic auxetic behaviour has been used in multiple applications from sportswear to space travel. The design and synthesis of molecular auxetic materials is a particularly exciting prospect. However synthetic molecular auxetic materials have not yet been developed.
[0003] Liquid crystals have long range order and through varying the components that give the liquid crystals their desired properties, the physical properties of the resulting materials can be fine-tuned. It has therefore been suggested that liquid crystal polymers may be developed which display auxetic properties. However, to date no such material has been reported.
[0004] The applicant has surprisingly developed a self assembled nematic material having auxetic properties.
[0005] In a first embodiment there is provided the use of an aligned nematic elastomer to form a material having auxetic properties wherein the aligned nematic material has a mechanical Freedericksz transition (MFT).
[0006] It has surprisingly been found that by forming an aligned nematic elastomer which displays an MFT the resulting material has auxetic properties and therefore has use in a wide range of novel applications. Because the materials have auxetic properties, they demonstrate improved shock absorbance and shear performance. These properties are useful in multiple applications. For example in aerospace, automotive, defence and sports applications as well as in biomedical fields where materials having auxetic properties can be used to mimic biological systems. Use in medical devices such as stents and valves and blood vessel dilators, where controlled expansions and contraction using external stimuli are important, is envisaged, as well as in prosthetic materials and surgical implants where reaction to external pressures is of particular use. The materials may also find use in medical attachment means such as sutures and anchors or for controlled release of active pharmaceutical ingredients through controlled contraction or expansion.
[0007] Materials displaying auxetic properties may also find application in piezoelectric sensors and actuators, as well as in micro- and nano-mechanical and electromechanical devices. Other potential uses include in composite materials where the materials displaying auxetic properties could act as reinforcements, or in personal protection clothing such as crash helmets, body armour, and sports clothing where expansion in response to external forces is clearly desirable.
[0008] A mechanical Freedericksz transition is defined as a deformation mode of an aligned elastomer wherein the director within the elastomer rotates sharply at a critical strain to reorient towards the direction parallel to the stress axis at a critical extension. Materials displaying this property were first described by Mitchell et al (Mitchell, G. R., Davis, F. J. and Guo, W., Phys. Rev. Lett., 1993, 71(18), 2947) and Roberts et al (Roberts, P. M. S., Mitchell, G. R and Davis, F. J., J. Phys, II France, 1997, 7, 1337 and Roberts, P. M. S., Mitchell, G. R, Davis, F. J. and Pople, J. A., Mol. Cryst. Liq. Cryst., 1997, 299, 181). An MFT is often described in analogy to the well-known electric (or magnetic) field Freedericksz transitions (EFT) that occur in low molar mass nematic display devices. In the EFT, the director reorients sharply beyond a well-defined critical field (or voltage), becoming increasingly aligned with respect to the electric field as the amplitude of the field is increased. The EFT threshold is discontinuous in theory, but is known to be softened if an ideal LC monodomain with alignment exactly parallel or perpendicular to the substrates is not achieved in practice. The threshold is nonetheless sharp and well-defined. The sharp rotation of the director seen in an MFT is different from the director rotation response for an aligned elastomer which deforms via semi-soft elasticity (SSE), the alternative deformation mode. In the case of semi-soft elasticity, the director rotates comparatively gradually over a plateau-like region of the tensile load curve. A theoretical plot demonstrating an SSE transition is shown in FIG. 1. The director rotates gradually across region II.
[0009] The mechanical Freedericksz transition is measurable by applying stress in a direction perpendicular or close to perpendicular to the aligned director in a sample and tracking the orientation of the director, for example by using polarising microscopy.
[0010] Such a method comprises: [0011] loading a strip of the elastomer into clamps of opposing actuators, placing the strip between crossed polarisers; [0012] applying strain incrementally by imposing extension steps of 5% of the strip's initial length per minute, in a direction perpendicular to the initial director orientation; [0013] at each extension increment, taking a series of measurements of transmitted light intensity (intensity of light transmitted through the polariser, strip and analyser), with the polariser and analyser being rotated relative to the strip by 10 degrees between each measurement. [0014] fitting the measurements of transmitted intensity using equation
[0014] I = I 0 sin 2 ( b .pi. .times. ( .theta. - c ) 1 8 0 ) + d ##EQU00001##
to determine c, the angle of the director relative to the direction of the applied stress (where I is the measured intensity, .theta. is the angle between the polariser and the fast axis of the birefringent material projected onto the plane of the polarizer, and I.sub.0, b, c, and d are fitting parameters). [0015] from the relationship between the director angle and the extension ratio, the critical extension ratio at which the elastomer undergoes the MFT can be determined--at the critical extension ratio, the director sharply rotates.
[0016] The aligned nematic elastomer preferably comprises a monodomain liquid crystal elastomer. More preferably the aligned nematic elastomer is a monodomain liquid crystal elastomer.
[0017] By "monodomain" herein is meant that the director orientation of the elastomer is macroscopically aligned in the sample. Monodomain alignment over the sample can be determined, for example, by polarising microscopy where it is characterised by uniform birefringence when the macroscopic sample is viewed between crossed polarisers.
[0018] Preferably the aligned nematic elastomer comprises a monodomain liquid crystal elastomer comprising; a polymeric component; a liquid crystal mesogen component; and a cross-linker component, wherein the liquid crystal mesogen component is physically linked to the polymeric component.
[0019] Preferably the liquid crystal mesogen component is physically linked to the polymeric component via a flexible spacer.
[0020] Preferably the flexible spacer comprises a C.sub.2-C.sub.10 alkylene group, preferably a linear C.sub.2-C.sub.10 alkylene group, more preferably a linear C.sub.3-C.sub.7 alkylene group, most preferably a linear C.sub.3 or C.sub.6 alkylene group. For example, the flexible spacer may comprise an ethylene, propylene, butylene, pentylene, hexylene, heptylene, octylene, nonylene or decylene group.
[0021] The liquid crystal mesogen component of the liquid crystal elastomer may comprise any suitable nematic mesogen.
[0022] Preferably the liquid crystal mesogen component comprises a liquid crystal core component selected from the group consisting of aromatic rings, aliphatic rings, poly aromatic rings, poly aliphatic rings, phenyls, biphenyls, benzenes, and combinations thereof.
[0023] Preferably the liquid crystal core component comprises a plurality of aromatic and/or aliphatic rings.
[0024] Preferably the liquid crystal core component is selected from one or more of the following systems:
##STR00001##
wherein R and R' are each independently selected from the group consisting of alkyl, alkoxy, halide, --NO.sub.2 or CN and wherein the alkyl and alkoxy groups may be bivalent when forming part of the linking group which connects the liquid crystal core to the polymeric component; and X and Y are each independently selected from the group consisting of CH.dbd.CH, --C.ident.C--, --CH.dbd.N--, --N.dbd.N--, or --C(O)O--.
[0025] Preferably the liquid core component comprises at least two phenyl groups.
[0026] The phenyl groups may be optionally substituted with any suitable functional group.
[0027] Preferably at least one of X or Y is --C(O)O-- or X or Y is absent.
[0028] Preferably the liquid crystal core component is selected from a 4-cyano-biphenyl-4'-yloxy, a 4-oxyphenyl 4-methoxybenzoate or a 4-oxyphenyl 4-(trans-4-propylcyclohexyl)benzoate group.
[0029] In certain embodiments the liquid crystal mesogen component is present as part of the side chain of the polymeric component, i.e. the liquid crystal mesogen component is a pendant group extending from the backbone of the polymeric component.
[0030] In certain embodiments the liquid crystal mesogen component is present as part of the backbone of the polymeric component.
[0031] The liquid crystal mesogen component may form part of both the side chain and backbone of the polymeric component.
[0032] The cross-linker component preferably comprises a bifunctional monomer having the same functionality as the polymeric component.
[0033] Preferably the cross-linker component also comprises a mesogenic component. Preferably the mesogenic component comprises a liquid crystal core component selected from one or more of the following systems:
##STR00002##
wherein R and R' are each independently selected from the group consisting of alkyl, alkoxy, halide, --NO.sub.2 or --CN and wherein the alkyl and alkoxy groups may be bivalent when forming part of the linking group which connects the liquid crystal core to the polymeric component; and X and Y are each independently selected from --CH.dbd.CH--, --C.ident.C--, --CH.dbd.N--, --N.dbd.N--, or --C(O)O--.
[0034] Preferably the liquid core component comprises at least three phenyl groups. Preferably at least one of X or Y is --C(O)O--.
[0035] The phenyl groups may be optionally substituted with any suitable functional group. Preferably the phenyl groups are optionally substituted with one or more C1-C3 alkyl groups, most preferably with one or more methyl groups.
[0036] Preferably the liquid core component comprises an optionally substituted bis-oxybenzoyloxybenzene group. Most preferably a bis-oxybenzoyloxy-2-methylbenzene group
[0037] The polymeric component may be any suitable polymeric component. Preferably the polymeric component comprises an acrylate polymer, a vinyl polymer, a siloxane polymer, a thiol based polymer, an amine based polymer or an epoxide based polymer. Most preferably the polymeric component comprises an acrylate polymer.
[0038] In certain embodiments the polymeric component is formed from both mesogenic and non-mesogenic components.
[0039] Preferably the mesogenic components are formed from mesogenic monomers which comprise a monomer unit linked to a liquid crystal core component.
[0040] The non-mesogenic component may be a Tg reducing component. In preferred embodiments the elastomer for use according to the first embodiment of the invention has a Tg at or below room temperature (25.degree. C.).
[0041] In preferred embodiments the Tg reducing components may be formed from monomers which comprise a monomer unit and a pendant medium chain (C.sub.2-C.sub.12) straight or branched alkyl group.
[0042] In particularly preferred embodiments the polymeric component comprises an acrylate polymer and the Tg reducing component comprises ethyl hexyl acrylate.
[0043] In preferred embodiments of the invention, the polymeric component comprises a polyacrylate, the liquid crystal core component is a 4-cyano-biphenyl-4'-yloxy component and the crosslinker component comprises a bis-oxybenzoyloxy-2methylbenzene comprising component.
[0044] The elastomer for use according to the first embodiment is preferably formed by polymerising a mixture comprising a mesogenic monomer, a crosslinking component and an initiator. The mixture may further comprise a non-mesogenic monomer to modify the properties of the final elastomer, for example to lower the Tg of the final elastomer. The mixture may also further comprise a non-reactive mesogenic component to broaden the nematic phase range prior to polymerisation. In preferred embodiments the crosslinking component also comprises a mesogenic component.
[0045] Preferably the mesogenic monomer comprises about 5-50% by mol. of the monomer mixture prior to polymerisation, more preferably about 10-30% by mol., most preferably approximately 15% by mol. In the final elastomer, the proportion of the material derived from the mesogenic monomer is preferably about 20-70% by mol., most preferably about 30-60% by mol.
[0046] Preferably the crosslinker component comprises about 1-20% by mol. of the monomer mixture prior to polymerisation, more preferably about 3-10% by mol, most preferably about 3-8% by mol. In the final elastomer, the proportion of the material derived from the crosslinker component is preferably about 5-20% by mol., most preferably about 8-17% by mol.
[0047] Preferably at least 10% of the crosslinker component comprises a mesogenic component, preferably at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90% or at least 95% of the crosslinker component comprises a mesogenic component.
[0048] The initiator chosen will be dependent on the polymer used and may be any suitable initiator. However, when the polymer is a polyacrylate the initiator is preferably a photoinitiator. Possible photoinitiators are well known to those skilled in the art and include benzoin ethers, benzyl ketals, alpha-dialkoxyacetophenones, alpha-hydroxyalkylphenones, acylphosphine oxides, benzophenones and thioxanthones. Preferably the photoinitiator is methyl benzoylformate. Preferably the initiator is present in an amount of approximately 1.5% by mol. of the monomer mixture.
[0049] When a non-mesogenic monomer is present in the monomer mixture, the non-mesogenic mixture preferably comprises about 10-40% by mol. of the monomer mixture, more preferably about 15-30% by mol., most preferably about 15-20% by mol. In the final elastomer, the proportion of the material derived from the non-mesogenic monomer, if present, is preferably about 20-60% by mol, most preferably about 35-50% by mol.
[0050] When a non-reactive mesogen is present in the monomer mixture, the non-reactive mesogen preferably comprises about 10-70% by mol. of the monomer mixture, more preferably about 20-60% by mol., or 30-60% by mol., most preferably approximately 55% by mol. In preferred embodiments the non-reactive mesogen is 4-cyano-4'-hexyloxybiphenyl.
[0051] In preferred embodiments, the mesogenic monomer is 6-(4-cyano-biphenyl-4'-yloxy)hexyl acrylate, 4-methoxybenzoic acid 4-(6-acryloyloxy-hexyloxy)phenyl ester or 4-{6-(acryloyloxy)hexyloxy}phenyl 4-(trans-4-propylcyclohexyl)benzoate, the crosslinker component is 1,4-bis-[4-(6-acryloyloxyhexyloxy)benzoyloxy]-2-methylbenzene or 1,4-bis-[4-(3-acryloyloxypropyloxy)benzoyloxy]-2-methylbenzene, the non-mesogenic monomer is 2-ethylhexyl acrylate and, if present, the non-reactive mesogen is 4-cyano-4'-hexyloxybiphenyl.
[0052] In preferred embodiments, the mesogenic monomer is 6-(4-cyano-biphenyl-4'-yloxy)hexyl acrylate, the crosslinker component is 1,4-bis-[4-(6-acryloyloxyhexyloxy)benzoyloxy]-2-methylbenzene, the non-mesogenic monomer is 2-ethylhexyl acrylate and, if present, the non-reactive mesogen is 4-cyano-4'-hexyloxybiphenyl; or the mesogenic monomers are 4-methoxybenzoic acid 4-(6-acryloyloxy-hexyloxy)phenyl ester and 4-{6-(acryloyloxy)hexyloxy}phenyl 4-(trans-4-propylcyclohexyl)benzoate, the crosslinker component is 1,4-bis-[4-(6-acryloyloxyhexyloxy)benzoyloxy]-2-methylbenzene the non-mesogenic monomer is 2-ethylhexyl acrylate and, if present, the non-reactive mesogen is 4-cyano-4'-hexyloxybipheny; or the mesogenic monomers are 4-methoxybenzoic acid 4-(6-acryloyloxy-hexyloxy)phenyl ester and 4-{6-(acryloyloxy)hexyloxy}phenyl 4-(trans-4-propylcyclohexyl)benzoate, the crosslinker component is, 4-bis-[4-(3-acryloyloxypropyloxy)benzoyloxy]-2-methylbenzene, the non-mesogenic monomer is 2-ethylhexyl acrylate and, if present, the non-reactive mesogen is 4-cyano-4'-hexyloxybiphenyl.
[0053] According to a further embodiment of the invention is provided a method of producing an aligned nematic elastomer for use according to the first embodiment of the invention, said method comprising the steps of: [0054] a) applying an aligning means to a substrate [0055] b) applying the liquid crystal elastomer components to the substrate and allowing them to form an aligned nematic phase [0056] c) curing the liquid crystal elastomer components to form an aligned nematic elastomer.
[0057] Various techniques for aligning mesogenic compositions exist. For example, techniques exist to create a monodomain during synthesis, including applying a magnetic field, mechanical brushing, flow, applying an electric field, applying a thermal gradient, or providing an alignment layer or layers. The monomeric solution may also be heated, cooled or exposed to other environmental factors to influence synthesis of the monomer mixture into an aligned state.
[0058] Preferably the aligning means is an aligning force which is applied by brushing the substrate, preferably to impart a static force to the substrate.
[0059] When the crosslinking component comprises a mesogenic component and therefore may also be considered a mesogenic monomer, the ratio of mesogenic monomers to non-mesogenic monomers in the final elastomer is preferably between 2:1 and 1:1.
[0060] Embodiments of the invention will now be described with reference to the accompanying examples and by reference to the drawings in which:--
[0061] FIG. 1 shows a theoretical plot of extension ratio vs stress for a material showing an SSE transition;
[0062] FIGS. 2a, 2b, 2c and 2d show plots of the fractional thickness vs the extension ratio and the Poisson's ratio vs the extension ratio of the materials of examples 1 to 4 respectively. Sub-zero values of the Poisson's ratio indicate the auxetic behaviour;
[0063] FIGS. 3a, 3b, 3c and 3d show plots of the tensile load curves and director angle response vs extension ratio for materials of examples 1 to 4 respectively; and
[0064] FIG. 4 shows a plot of the fractional change vs strain for the material of example 1 at varying temperature and varying extension rate.
EXAMPLES
[0065] Elastomer Synthesis
[0066] Aligned nematic elastomers for use according to the invention were synthesised as follows using the following materials: [0067] 2-ethylhexyl acrylate (EHA), [0068] 6-(4-cyano-biphenyl-4'-yloxy)hexyl acrylate (A6OCB), [0069] 4-methoxybenzoic acid 4-(6-acryloyloxy-hexyloxy)phenyl ester (M1) [0070] 4-{6-(acryloyloxy)hexyloxy}phenyl 4-(trans-4-propylcyclohexyl)benzoate (M2) [0071] 1,4-bis-[4-(6-acryloyloxyhexyloxy)benzoyloxy]-2-methylbenzene (RM82), [0072] 1,4-bis-[4-(3-acryloyloxypropyloxy)benzoyloxy]-2-methylbenzene (RM257) [0073] 4-cyano-4'-hexyloxybiphenyl (6OCB) and [0074] methyl benzoylformate (MBF).
[0075] The elastomers for use according to the invention were prepared using the following starting compositions:
TABLE-US-00001 Material % by mol Component Example 1 Example 2 Example 3 Example 4 A60CB 14.6 24.4 0 0 M1 0 0 15.5 15.3 M2 0 0 5.6 5.5 60CB 55.9 54.6 50.0 50.7 RM82 7.1 3.5 5.6 0 RM257 0 0 0 5.3 EHA 20.9 16.0 22.4 22.3 MBF 1.6 1.5 0.8 0.8
[0076] Using a balance with an accuracy of 0.3 mg, dry materials were measured into a 4 ml sample vial. The mixture was then heated to 100.degree. C. and stirred at 60 rpm for 5 minutes. The liquid materials were added and the vial was placed on a separate stirring plate held at 40.degree. C. and stirred at 60 rpm for a further 5 minutes.
[0077] The mixtures were then filled in the isotopic phase at 40.degree. C. into the cells previously prepared via capillary action and left for approximately half an hour to cool to ambient temperature allowing the nematic phase to form via alignment with the rubbing direction. Once aligned, the cells were placed under a low intensity UV fluorescence light source (intensity of 2.5 mW cm.sup.-2) for two hours to cure. Once separated from the cells, the film was washed in dicholoromethane (DCM) by slowly adding DCM stepwise to about 30% concentration. Solvents were exchanged several times to ensure all waste materials were removed before deswelling the LCE films by adding methanol stepwise. The films were left to dry fully overnight before testing.
[0078] The auxetic properties of the four materials are demonstrated in FIGS. 2a, 2b, 2c and 2d respectively which show the materials to have a negative Poisson's ratio. Beyond an extension ratio of approximately 1.8 in FIG. 2a, approximately 1.5 in FIG. 2b, approximately 1.6 in FIG. 2c and approximately 1.6 in FIG. 2d, the fractional thickness of the materials increases with increasing extension ratio. FIGS. 3a, 3b, 3c and 3d demonstrate that the materials each possess an MFT. In FIG. 3a a sharp change in director angle is seen at a strain of approximately 2.1. In FIG. 3b a sharp change in director angle is seen at an x deformation of approximately 1.9. In FIG. 3c a sharp change in director angle is seen at an x deformation of approximately 1.9. In FIG. 3d a sharp change in director angle is seen at an x deformation of approximately 1.9.
* * * * *



D00000

D00001

D00002

D00003

D00004

D00005


XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.