Compositions Containing A Hollow Glucan Particle Or A Cell Wall Particle Encapsulating A Terpene Component, Methods Of Making An
FRANKLIN; Lanny ; et al.
U.S. patent application number 16/828530 was filed with the patent office on 2020-07-30 for compositions containing a hollow glucan particle or a cell wall particle encapsulating a terpene component, methods of making an. The applicant listed for this patent is Eden Research PLC. Invention is credited to Lanny FRANKLIN, Gary OSTROFF.
| Application Number | 20200236927 16/828530 |
| Document ID | 20200236927 / US20200236927 |
| Family ID | 1000004753957 |
| Filed Date | 2020-07-30 |
| Patent Application | download [pdf] |




| United States Patent Application | 20200236927 |
| Kind Code | A1 |
| FRANKLIN; Lanny ; et al. | July 30, 2020 |
COMPOSITIONS CONTAINING A HOLLOW GLUCAN PARTICLE OR A CELL WALL PARTICLE ENCAPSULATING A TERPENE COMPONENT, METHODS OF MAKING AND USING THEM
Abstract
The present invention relates to compositions comprising a hollow glucan particle or cell wall particle encapsulating a terpene component, methods of their manufacture and their use. The compositions are suitable for preventing and treating infections in plants and animals, including humans.
| Inventors: | FRANKLIN; Lanny; (Atlanta, GA) ; OSTROFF; Gary; (Worcester, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004753957 | ||||||||||
| Appl. No.: | 16/828530 | ||||||||||
| Filed: | March 24, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 11597116 | Oct 27, 2008 | 10638750 | ||
| PCT/GB2005/002011 | May 20, 2005 | |||
| 16828530 | ||||
| 60572892 | May 20, 2004 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A01N 35/06 20130101; A01N 31/08 20130101; A01N 25/28 20130101; A01N 31/16 20130101; B01J 13/203 20130101; A01N 49/00 20130101; A61K 9/5068 20130101; A01N 25/26 20130101; A61K 9/5036 20130101; B01J 13/206 20130101; A01N 35/02 20130101; A01N 31/02 20130101 |
| International Class: | A01N 25/26 20060101 A01N025/26; A01N 31/08 20060101 A01N031/08; A01N 31/16 20060101 A01N031/16; A01N 31/02 20060101 A01N031/02; A61K 9/50 20060101 A61K009/50; A01N 35/02 20060101 A01N035/02; A01N 25/28 20060101 A01N025/28; B01J 13/20 20060101 B01J013/20; A01N 35/06 20060101 A01N035/06; A01N 49/00 20060101 A01N049/00 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jan 24, 2005 | GB | PCT/GB2005/000240 |
Claims
1. A composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component.
2. A composition according to claim 1 wherein the hollow glucan particle or cell wall particle is a fungal cell wall.
3. A composition according to claim 2 wherein the hollow glucan particle or cell wall particle is a yeast cell wall.
4. A composition according to claim 3 wherein the yeast cell wall is derived from a Baker's yeast cell.
5. A composition according to any preceding claim wherein the hollow glucan particle or cell wall particle is an insoluble waste product from a yeast extract manufacturing process.
6. A composition according to any one of claims any preceding claim wherein the hollow glucan particle or cell wall particle has been alkali extracted.
7. A composition according to any preceding claim wherein the hollow glucan particle or cell wall particle has been acid extracted.
8. A composition according to any preceding claim wherein the hollow glucan particle or cell wall particle has been organic solvent extracted.
9. A composition according to any preceding claim wherein the hollow glucan particle or cell wall particle has a lipid content of 1% or greater.
10. A composition according to claim 9 wherein the lipid content of the hollow glucan particle or cell wall particle is 5% w/w or greater.
11. A composition according to claim 10 wherein the lipid content is 10% w/w or greater.
12. A composition according to any preceding claim wherein the terpene component comprises one or more of the terpenes selected from the group consisting of citral, pinene, nerol, b-ionone, geraniol, carvacrol, eugenol, carvone (for example L-carvone), terpeniol, anethole, camphor, menthol, thymol, limonene, nerolidol, farnesol, phytol, carotene (vitamin A.sub.1), squalene, thymol, tocotrienol, perillyl alcohol, borneol, myrcene, simene, carene, terpenene, linalool or a mixture thereof.
13. A composition according to any preceding claim wherein the terpene component comprises a terpene having have the general structure C.sub.10H.sub.16.
14. A composition according to any preceding claim wherein the terpene component comprises one or more terpenes selected from the group consisting of geraniol, thymol, citral, carvone (for example L-carvone), eugenol, b-ionone or a mixture thereof.
15. A composition according to any preceding claim wherein the terpene component comprises a mixture of geraniol, thymol and eugenol.
16. A composition according to claim 14 wherein the terpene component comprises 100% thymol.
17. A composition according to claim 14 wherein the terpene component comprises 50% geraniol and 50% thymol w/w.
18. A composition according to claim 14 wherein the terpene component comprises 50% eugenol and 50% thymol w/w.
19. A composition according to claim 14 wherein the terpene component comprises 33% geraniol, 33% eugenol and 33% thymol w/w.
20. A composition according to claim 14 wherein the terpene component comprises 33% eugenol, 33% thymol and 33% citral w/w.
21. A composition according to claim 14 wherein the terpene component comprises 25% geraniol, 25% eugenol, 25% thymol and 25% citral w/w.
22. A composition according to claim 14 wherein the terpene component comprises 20% geraniol, 20% eugenol, 20% citral, 20% thymol and 20% L-carvone w/w.
23. A composition according to any preceding claim wherein the terpene component is associated with a surfactant.
24. A composition according to any preceding claim wherein the surfactant is selected from the group consisting of sodium lauryl sulphate, polysorbate 20, polysorbate 80, polysorbate 40, polysorbate 60, polyglyceryl ester, polyglyceryl monooleate, decaglyceryl monocaprylate, propylene glycol dicaprilate, triglycerol monostearate, polyoxyethylenesorbitan monooleate, Tween.RTM., Span.RTM. 20, Span.RTM. 40, Span.RTM. 60, Span.RTM. 80, Brig 30 or a mixture of two or more thereof.
25. A composition according to any preceding claim comprising 1 to 99% by volume terpenes, 0 to 99% by volume surfactant and 1 to 99% hollow glucan particles or cell wall particles.
26. A composition according to claim 25 comprising from about 10 to about 67% w/w terpenes, from about 0.1 to about 10% w/w surfactant and from about 40 to about 90% w/w hollow glucan particles or cell wall particles.
27. A composition according to any preceding claim suitable for killing bacteria or fungi.
28. A composition according to any preceding claim suitable for killing mold.
29. A composition according to any preceding claim, suitable for killing mycoplasma.
30. A composition according to any preceding claim wherein the terpenes used are food grade.
31. A composition according to any preceding claim comprising an additional food grade active compound.
32. A composition according to claim 31 wherein the additional food grade active compound is an antimicrobial agent or enzyme.
33. A composition according to any preceding claim comprising an antimicrobial agent, an anti-fungal agent, an insecticidal agent, an anti-inflammatory agent or an anaesthetic.
34. A composition according to any preceding claim further comprising an antioxidant.
35. A composition according to claim 34 wherein the antioxidant is rosemary oil, vitamin C or vitamin E.
36. A composition according to any preceding claim in the form of a dry powder.
37. A composition according to any one of claims 1 to 35 in a pellet, tablet or other solid form.
38. A composition according to any preceding claim comprising a dispersal agent which promotes dispersal of the composition when placed into a liquid.
39. A composition according to any preceding claim in combination with an agriculturally, food or pharmaceutically acceptable carrier or excipient in a liquid, solid or gel-like form.
40. A composition according to any one of claims 1 to 35 suspended or dissolved in a liquid.
41. A composition according to claim 40 wherein the liquid is water.
42. A composition according to either claim 40 or 41 comprising from about 500 to about 10,000 ppm hollow glucan particles or cell wall particles, where the particles contain from about 1 to about 67% terpene component.
43. A composition according to claim 42 comprising from about 1000 to about 2000 ppm hollow glucan particles or cell wall particles, where the particles contain from about 10 to about 50% terpene component w/w.
44. A composition according to any one of claims 40 to 43 comprising between about 1 ppm and about 25 ppt of the terpene component.
45. A composition according to claim 44 comprising between about 100 to 1000 ppm of the terpene component.
46. A composition according to any one of claims 1 to 39 which is dispersed in water, saline, aqueous dextrose, glycerol or ethanol to form a solution or suspension.
47. A composition according to claim any preceding claim which includes a wetting agent, an emulsifying agent or a pH buffering agent.
48. A composition according to any preceding claim dispersed in a liquid human or animal food or drink material.
49. A composition according to any preceding claim in a form suitable for oral administration.
50. A composition according to any one of claims 1 to 46 in a form suitable for parental administration.
51. A composition according to any one of claims 1 to 46 in a form suitable for topical administration.
52. A method of preparing a hollow glucan particle or cell wall particle encapsulating a terpene component, said method comprising the steps of; a) providing a terpene component; b) providing a hollow glucan particle or cell wall particle; c) incubating the terpene component with the glucan particle or cell wall particle under suitable conditions for terpene encapsulation; and d) recovering the glucan particle or cell wall particle encapsulating the terpene component.
53. A method according to claim 52 further comprising the step of drying the glucan particle or cell wall particle encapsulating the terpene component.
54. A method according to claim 53 wherein drying is achieved by freeze drying, fluidised bed drying, drum drying or spray drying.
55. A method according to any one of claims 52 to 54 wherein in step a) the terpene component is provided as a suspension in an aqueous solvent.
56. A method according to claim 55 wherein the terpene component is provided in association with a surfactant.
57. A method according to claim 56 wherein the surfactant is polyoxyethylenesorbitan monooleate at a concentration of about 0.1 to 10% by volume of the total reaction mixture.
58. A method according to any one of claims 52 to 54 wherein in step a) the terpene component is provided as a true solution in the aqueous solvent.
59. A method according to any one of claims 52 to 58 wherein in step b) the hollow glucan particle or cell wall particle is provided as a suspension in water or other suitable liquid.
60. A method according to claim 59 wherein the suspension comprises approximately 1 to 1000 mg glucan particle or cell wall particles per ml.
61. A method according to claim 59 wherein the particles are dispersed in a volume of from the hydrodynamic volume (HV) to 1.5 HV of liquid.
62. A method according to any one of claims 52 to 58 wherein in step b) the hollow glucan particle or cell wall particle is provided as a dry powder.
63. A method according to any one of claims 52 to 62 wherein in step c) the reaction is carried out at atmospheric pressure at a temperature of about 20 to 37.degree. C.
64. A method of killing a microorganism, said method comprising the step of; contacting said microorganism with a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component.
65. A method of treating or preventing infection of a plant, said method comprising the step of; administering, in a therapeutically effective dose, a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component to the plant or to soil in proximity to the plant.
66. A method according to claim 65 wherein the infection of the plant is caused by a nematode.
67. A method according to claim 65 wherein the infection of a plant is caused by a fungus.
68. A method according to claim 67 wherein the fungus is downy mildew, powdery mildew or botrytis bunch rot.
69. A method according to any one of claims 65 to 68 wherein the plant is a grape vine.
70. A method according to any one of claims 65 to 69 wherein the composition is administered 21 days or less prior to harvest of a crop from the plant.
71. A method according to claim 70 wherein the composition is administered 14 days or less prior to harvest.
72. A method according to claim 71 wherein the composition is administered 7 days or less prior to harvest.
73. A method according to claim 72 wherein the composition is administered 3 days or less prior to harvest.
74. A method according any one of claims 65 to 73 wherein the composition is administered by spraying.
75. A method according to claim 74 wherein the composition is sprayed at a rate of 500 L/Ha or greater.
76. A method according to claim 75 wherein the composition is sprayed at a rate of 900 L/Ha or greater.
77. A method according to claim 76 wherein the composition is sprayed at a rate of 1200 L/Ha or greater.
78. A method according to any one of claims 65 to 73 wherein the composition is administered via irrigation.
79. The present invention further provides a method of preventing or treating an infection in a patient, said method comprising the step of; administering to said patient in a therapeutically effective dose, a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component.
80. A method according to claim 79 wherein the infection of the patient is caused by Staphylococcus aureus, Aspergillius fumigatus, Mycoplasma iowae, Penicillium sp. or Mycoplasma pneumoniae.
81. A method according to claim 80 wherein the composition is administered orally, vaginally, rectally, by inhalation, topically or by parenteral routes.
82. A composition comprising a hollow glucan particle encapsulating a terpene component for use in the prevention or treatment of an infection in a patient or a plant.
83. Use of a hollow glucan particle encapsulating a terpene component in the manufacture of a medicament for the treatment of an infection in patient.
84. The use of claim 83 wherein the infection is caused by Aspergillius fumigatus, Scierotinta homeocarpa, Rhizoctonia solani, Colletotrichum graminicola or Penicillium sp.
Description
[0001] The present invention relates to compositions comprising terpenes and hollow glucan particles or cell wall particles and methods for preparing such compositions. The compositions increase terpene stability and activity and provide a suitable carrier for the terpenes. The invention also relates to methods of using such compositions in the medical, veterinary and agricultural fields.
[0002] Terpenes are chemical compounds that are widespread in nature, mainly in plants as constituents of essential oils. Their building block is the hydrocarbon isoprene (C.sub.5H.sub.8).sub.n. Examples of terpenes include citral, pinene, nerol, b-ionone, geraniol, carvacrol, eugenol, carvone, terpeniol, anethole, camphor, menthol, limonene, nerolidol, farnesol, phytol, carotene (vitamin A.sub.1), squalene, thymol, tocotrienol, perillyl alcohol, borneol, myrcene, simene, carene, terpenene, and linalool.
[0003] Terpenes are classified as Generally Recognized as Safe (GRAS) and have been used for many years in the flavouring and aroma industries. The LD.sub.50 in rats of citral is approximately 5 g/kg, which is a further indication of the relative safety of these compounds. Furthermore, terpenes have a relatively short life span of approximately 28 days once exposed to oxygen (e.g. air). Terpenes will decompose to CO.sub.2 and water. This decomposition or break down of terpenes demonstrates the safety and environmental friendliness of the compositions and methods of the invention.
[0004] Terpenes have been found to inhibit the growth of cancerous cells, decrease tumour size, decrease cholesterol levels, and have a biocidal effect on micro-organisms in vitro. Owawunmi, (Letters in Applied Microbiology, 1993, 9(3): 105-108), showed that growth media with more than 0.01% citral reduced the concentration of E. coli, and at 0.08% there was a bactericidal effect. U.S. Pat. No. 5,673,468 describes a terpene formulation, based on pine oil, used as a disinfectant or antiseptic cleaner. U.S. Pat. No. 5,849,956 teaches that a terpene found in rice has antifungal activity. U.S. Pat. No. 5,939,050 describes an oral hygiene antimicrobial product with a combination of 2 or 3 terpenes that showed a synergistic effect. Several (U.S. Pat. Nos. 5,547,677, 5,549,901, 5,618,840, 5,629,021, 5,662,957, 5,700,679, 5,730,989) teach that certain types of oil-in-water emulsions have antimicrobial, adjuvant, and delivery properties. Terpenes have been found to be effective and nontoxic dietary anti-tumor agents, which act through a variety of mechanisms of action (Crowell et al. Crit. Rev. Oncog., 1994, 5(1): 1-22; Crowell et al. Adv. Exp. Med. Biol., 1996, 401: 131-136). The terpenes geraniol, tocotrienol, perillyl alcohol, b-ionone, and d-limonene, suppress hepatic HMG-CoA reductase activity, a rate limiting step in cholesterol synthesis, and modestly lower cholesterol levels in animals (Elson et al, J. Nutr., 1994, 124: 607-614). D-limonene and geraniol reduced mammary tumors (Elegbede et al. Carcinogenesis, 1984, 5(5): 661-664; Elegbede et al., J. Natl. Cancer Inst., 1986, 76(2): 323-325; Karlson et al. Anticancer Drugs, 1996, 7(4): 422-429) and suppressed the growth of transplanted tumors (Yu et al., J. Agri. Food Chem., 1995, 43: 2144-2147).
[0005] Terpenes have also been found to inhibit the in vitro growth of bacteria and fungi (Chaumont et al.), Ann. Pharm. Fr., 1992, 50(3): 156-166; Moleyar et al., Int. J. Food Microbiol, 1992, 16(4): 337-342; and Pattnaik et al. Microbios, 1997, 89(358): 39-46) and some internal and external parasites (Hooser et al., J. Am. Vet. Med. Assoc., 1986, 189(8): 905-908). Geraniol was found to inhibit growth of Candida albicans and Saccharomyces cerevisiae strains by enhancing the rate of potassium leakage and disrupting membrane fluidity (Bard et al., Lipids, 1998, 23(6): 534-538). B-ionone has antifungal activity which was determined by inhibition of spore germination, and growth inhibition in agar (Mikhlin et al., A. Prikl. Biokhim. Mikrobiol, 1983, 19: 795-803; Salt et al., Adam. Physiol. Molec. Plant Path, 1986, 28: 287-297). Teprenone (geranylgeranylacetone) has an antibacterial effect on H. pylori (Ishii, Int. J. Med. Microbiol. Virol. Parasitol. Infect. Dis., 1993, 280(1-2): 239-243). Rosanol, a commercial product with 1% rose oil, has been shown to inhibit the growth of several bacteria (Pseudomonas, Staphylococus, E. coli, and H. pylori). Geraniol is the active component (75%) of rose oil. Rose oil and geraniol at a concentration of 2 mg/L inhibited the growth of H. pylori in vitro. Some extracts from herbal medicines have been shown to have an inhibitory effect in H. pylori, the most effective being decursinol angelate, decursin, magnolol, berberine, cinnamic acid, decursinol, and gallic acid (Bae et al., Biol. Pharm. Bull., 1998, 21(9) 990-992). Extracts from cashew apple, anacardic acid, and (E)-2-hexenal have shown bactericidal effect against H. pylori.
[0006] Diterpenes, i.e., trichorabdal A (from R. Trichocarpa), have shown a very strong antibacterial effect against H. pylori (Kadota et al., Zentralbl. Bakteriol, 1997, 287(1): 63-67).
[0007] Solutions of 11 different terpenes were effective in inhibiting the growth of pathogenic bacteria in in vitro tests; levels ranging between 100 ppm and 1000 ppm were effective. The terpenes were diluted in water with 1% polysorbate 20 (Kim et al., J. Agric. Food Chem., 1995, 43: 2839-2845).
[0008] There may be different modes of action of terpenes against microorganisms; they could (1) interfere with the phospholipid bilayer of the cell membrane, (2) impair a variety of enzyme systems (HMG-reductase), and (3) destroy or inactivate genetic material. It is believed that due to the modes of action of terpenes being so basic, e.g., blocking of cholesterol, that infective agents will not be able to build a resistance to terpenes.
[0009] There are, however, a number of drawbacks to the use of terpenes. These include: [0010] Terpenes are liquids which can make them difficult to handle and unsuitable for certain purposes. [0011] Terpenes are not very miscible with water, and it generally requires the use of detergents, surfactants or other emulsifiers to prepare aqueous emulsions. A stable solution can, however, be obtained by mixing the terpenes under high shear. [0012] Dry powder terpene formulations generally only contain a low percentage w/w of terpenes. [0013] Terpenes are prone to oxidation in aqueous emulsion systems, which make long term storage a problem.
[0014] There are limitations to the current techniques of spray coating, extrusion, coacervation, molecular encapsulation, and spray drying/cooling to provide ingredient delivery systems.
[0015] Baker's yeast cell walls are derived from baker's yeast cells and are composed of the insoluble biopolymers .beta.-1,3-glucan, .beta.-1,6-glucan, mannan and chitin. They are typically 2-4 micron in diameter microspheres with a shell wall that is only 0.2-0.3 micron thick surrounding an open cavity. This material has considerable liquid holding capacity, typically absorbing 5-25 times its weight in liquid. The shell is sufficiently porous that payloads up to 150,000 Daltons in size can pass through the outer shell and be absorbed into the hollow cavity of the spherical particle. Baker's yeast cell walls have several unique properties, including heat stability (e.g. to 121.degree. C.), shear stability, pH stability (e.g. pH 2-12), and at high concentrations they do not build significant viscosity. In addition to its physical properties this composition contains natural and healthy dietary fibres that deliver cardiovascular and immunopotentiation health benefits.
[0016] Yeast cell walls are prepared from yeast cells by the extraction and purification of the insoluble particulate fraction from the soluble components of the yeast cell. The fungal cell walls can be produced from the insoluble byproduct of yeast extract manufacture. Further, the yeast cells can be treated with an aqueous hydroxide solution, without disrupting the yeast cell walls, which digests the protein and intracellular portion of the cell, leaving the yeast cell wall component devoid of significant protein contamination, and having substantially the unaltered cell wall structure of .beta.(1-6) and .beta.(1-3) linked glucans. A more detailed description of whole glucan particles and the process of preparing them is described by Jamas et al. in U.S. Pat. No. 4,810,646 and in co-pending patent applications U.S. Ser. No. 166,929, U.S. Ser. No. 297,752 and U.S. Ser. No. 297,982. U.S. Pat. No. 6,242,594, assigned to Novogen Research Pty Ltd., describes a method of preparing yeast glucan particles by alkali extraction, acid extraction and then extraction with an organic solvent and finally drying. U.S. Pat. No. 5,401,727, assigned to AS Biotech-Mackzymal, discloses the methods of obtaining yeast glucan particles and methods of using them to promote resistance in aquatic animals and as an adjuvant for vaccinations. U.S. Pat. No. 5,607,677, assigned to Alpha-Beta Technology Inc., discloses the use of hollow whole glucan particles as a delivery package and adjuvant for the delivery of a variety of pharmaceutical agents. The teachings of the abovementioned patents and applications are incorporated herein by reference.
[0017] Other types of yeast and fungi cells have cell walls that do not contain glucan. The cell walls of such yeast and fungi can be isolated by similar techniques to those mentioned above to obtain cell wall particles.
[0018] Additionally, the cells of many plants, algae, bacteria and other micro-organisms also comprise a cell wall. The structure and composition of the cell wall varies between micro-organism, but in general it is a robust and relatively inert structure. It is possible to obtain cell wall particles derived from such cells through conventional techniques, such as those mentioned above in relation to yeast.
[0019] We have now found that terpenes can be taken up and stably encapsulated within hollow glucan particles or cell wall particles. Encapsulation of terpenes into such particles can be achieved by incubation of the particles with the terpene.
[0020] According to the present invention there is provided a composition comprising a hollow glucan particle or a cell wall particle encapsulating a terpene component.
[0021] The term "hollow glucan particle" as used herein includes any hollow particle comprising glucan as a structural component. Thus, in particular, the term includes yeast cell walls (in purified or crude forms) or hollow whole glucan particles. The term "cell wall particle" refers to a particle comprising the wall of a cell (in a purified or crude form), wherein glucan is not a structural component. Suitable particles include the cell walls of plant, algal, fungal or bacterial cells. Cell wall particles generally retain the shape of the cell from which they are derived, and thus, like a hollow glucan particle, provide a hollow central cavity suitable for encapsulating the terpene component.
[0022] For the present invention it is necessary that the hollow glucan particle or cell wall particle is able to stably encapsulate the terpene component. In general this means the hollow glucan particle or cell wall particle must be able to maintain its structure during incubation with the terpene component (generally the terpene component is at a relatively high concentration), and that terpene component must be able to migrate into the particle. Hollow glucan particles and cell wall particles are generally formed from relatively inert materials and are porous, and thus it can be assumed that, in general, hollow glucan particles and cell wall particles will be able to encapsulate a terpene component.
[0023] Compositions according to the present invention are effective against various infective agents including bacteria, viruses, mycoplasmas, fungi and/or nematodes.
[0024] The compositions according to the present invention can provide the following advantages: [0025] maximise terpene payload; [0026] minimise unencapsulated payload; [0027] control payload stability; [0028] control payload release kinetics; [0029] creation of a solid form of a liquid terpene to increase the mass and uniformity; [0030] simplify handling and application of terpenes; and [0031] mask the smell and taste of the terpene.
[0032] Particularly suitable hollow glucan particles or cell wall particles are fungal cell walls, preferably yeast cell walls. Yeast cell walls are preparations of yeast cells that retain the three-dimensional structure of the yeast cell from which they are derived. Thus they have a hollow structure which allows the terpene component to be encapsulated within the yeast cell walls. The yeast walls may suitably be derived from Baker's yeast cells (available from Sigma Chemical Corp., St. Louis, Mo.). Yeast cell wall particles with desirable properties can also be obtained from Biorigin (Sao Paolo, Brazil) under the trade name Nutricell MOS 55. These particles are a spray dried extract of S. cerevisiae.
[0033] Alternative particles are those known by the trade names SAF-Mannan (SAF Agri, Minneapolis, Minn.) and Nutrex (Sensient Technologies, Milwaukee, Wis.). These are hollow glucan particles that are the insoluble waste stream from the yeast extract manufacturing process. During the production of yeast extracts the soluble components of partially autolyzed yeast cells are removed and the insoluble residue is a suitable material for terpene loading. These hollow glucan particles comprise approximately 25-35% beta 1,3-glucan w/w. A key attribute of these materials are that they contain more than 10% lipid w/w and are very effective at absorbing terpenes. In addition, as a waste stream product they are a relatively cheap source of hollow glucan particles.
[0034] Alternative hollow glucan particles which have higher purity are those produced by Nutricepts (Nutricepts Inc., Burnsville, Minn.) and ASA Biotech. These particles have been alkali extracted, which removes additional intracellular components as well as removes the outer mannoprotein layer of the cell wall yielding a particle of 50-65% glucan w/w.
[0035] Higher purity hollow glucan particles are the WGP particles from Biopolymer Engineering. These particles are acid extracted removing additional yeast components yielding a product 75-85% glucan w/w.
[0036] Very high purity hollow glucan particles are Adjuvax.TM. from Alpha-beta Technology, Inc. (Worcester, Mass.) and microparticulate glucan from Novogen (Stamford, Conn.). These particles are organic solvent extracted which removes residual lipids and sd: the particles comprise more than 90% glucan w/w.
[0037] In some embodiments a high purlity glucan particle or cell wall particle may be required, for example where strict control over possible contaminants is required. In these instances the higher purity particles would be preferred aver other less pure products. For other embodiments, the less pure particles would be preferred for economic reasons; those particles have also been found to be more effective at absorbing terpenes.
[0038] Preferably the hollow glucan particle or cell wall particle has a slight lipid content, such as 1 or 2% w/w lipid. A slight lipid content can increase the ability of the particle to encapsulate the terpene component. Preferably the lipid content of the hollow glucan particle or cell wall particle is 5% w/w or greater, more preferably 10% w/w or greater.
[0039] Optionally the terpene component of the present invention can be associated with a surfactant. The surfactant can be non-ionic, cationic, or anionic. Examples of suitable surfactants include sodium lauryl sulphate, polysorbate 20, polysorbate 80, polysorbate 40, polysorbate 60 polyglyceryl ester, polyglyceryl monooleate, decagilyceryl monocaprylate, propylene glycol dicaprilate, triglycerol monostearate, polyoxyethylenesorbitan, monooleate, Tween.RTM., Span.RTM. 20, Span.RTM. 40, Span.RTM. 60, Span.RTM. 80, Brig 30 or mixtures thereof. The surfactant acts to hold the terpene component in an emulsion and also assists encapsulation of the terpene component into the hollow glucan particle or cell wall particle.
[0040] The terpene component of the present invention can comprise a single terpene or a mixture of terpenes. Mixtures of terpenes can result in synergistic effects.
[0041] The term "terpene" as used herein refers not only to terpenes of formula (C.sub.5H.sub.8)--, but also encompasses terpene derivatives, such as terpene aldehydes or terpene polymers. Natural and synthetic terpenes are included, for example monoterpenes, sesquiterpenes, diterpenes, triterpenes, and tetraterpenes. In addition, reference to a single name of a compound will encompass the various isomers of that compound. For example, the term citral includes the cis-isomer citral-a (or geranial) and the trans-isomer citral-b (or neral).
[0042] It should be noted that terpenes are also known by the names of the extract or essential oil which contain them, e.g. lemongrass oil (contains citral).
[0043] The terpenes which are exempted from US regulations and which are listed in EPA regulation 40 C.F.R. Part 152 (incorporated herein by reference in its entirety) are suitable for use in this invention.
[0044] Particularly suitable terpenes for use in the present invention include those selected from the group consisting of citral, pinene, nerol, b-ionone, geraniol, carvacrol, eugenol, carvone (for example L-carvone), terpeniol, anethole, camphor, menthol, thymol, limonene, nerolidol, farnesol, phytol, carotene (vitamin A.sub.1), squalene, thymol, tocotrienol, perillyl alcohol, borneol, myrcene, simene, carene, terpenene, linalool and mixtures thereof.
[0045] Preferably the terpenes used in the present invention have the general structure C.sub.10H.sub.16 as this sub-group is generally more effective against infective agents.
[0046] More preferably the terpene component comprises a terpene selected from the group consisting of geraniol, thymol, citral, carvone (for example L-carvone), eugenol and b-ionone.
[0047] The terpene component can suitably comprise thymol, as this terpene has been shown to be particularly effective in treating or preventing fungal plant infections.
[0048] Another particularly suitable terpene is citral which has demonstrated particular efficacy against a number of micro-organisms.
[0049] A combination of geraniol, thymol and eugenol has demonstrated particular efficacy in combating plant infections, and is thus a particularly suitable terpene component.
[0050] Other terpene formulations which have shown high efficacy in treating plant infections include (percentages are w/w): [0051] 100% thymol; [0052] 50% geraniol and 50% thymol; [0053] 50% eugenol and 50% thymol; [0054] 33% geraniol, 33% eugenol and 33% thymol; [0055] 33% eugenol, 33% thymol and 33% citral; [0056] 25% geraniol, 25% eugenol, 25% thymol and 25% citral; [0057] 20% geraniol, 20% eugenol, 20% citral, 20% thymol and 20% L-carvone.
[0058] Accordingly a terpene component comprising any of the above formulations is particularly suitable for use in the present invention.
[0059] In one embodiment the terpene component includes one or more terpenes which contain oxygen. Citral, for example citral 95, is an oxygenated C.sub.10H.sub.16 terpene, C.sub.10H.sub.16O CAS No. 5392-40-5 (3,7-dimethyl-2,6-octadien-1-al). A stable suspension of citral can be formed up to about 2500 ppm. Citral can be made into a solution at up to about 500 ppm. A stable suspension of hollow glucan particles incorporating citral of 25 ppt citral can be made.
[0060] The composition of the invention can comprise 1 to 99% by volume terpenes, 0 to 99% by volume surfactant and 1 to 99% hollow glucan particles or cell wall particles. More specifically the composition can comprise about 10% to about 67% w/w terpenes, about 0.1-10% surfactant and about 40-90% hollow glucan particles or cell wall particles.
[0061] Suitably a composition of the present invention comprises from about 500 to about 10,000 ppm hollow glucan particles or cell wall particles, where the particles contain from about 1 to about 67% terpene component. Preferably the composition comprises from about 1000 to about 2000 ppm hollow glucan particles or cell wall particles, where the particles contain from about 10 to about 50% terpene component.
[0062] Specific compositions can include e.g., for bacteria and fungi, hollow glucan particles or cell wall particles encapsulating terpenes in water or standard 0.9% saline with up to 67% L-carvone, up to 67% eugenol, up to 67% citral, up to 67% thymol and L-carvone, up to 67% geraniol, or up to 67% citral and L-carvone and eugenol, and 1% Tween.RTM. 80; for mold, hollow glucan particles or cell wall particles encapsulating terpenes in water or standard 0.9% saline with up to 67% citral and 1% Tween.RTM. 80; or for mycoplasma, hollow glucan particles or cell wall particles encapsulating terpenes in water or standard 0.9% saline with up to 67% citral, up to 67% L-carvone and eugenol, up to 67% eugenol, up to 67% geraniol, or up to 67% geraniol, thymol, and 1% Tween.RTM. 80.
[0063] Concentrations of hollow glucan particles or cell wall particles encapsulating terpenes of 1, 5, 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 110, 125, 130, 140, 150, 160, 175, 190, 200, 225, 250, 275, 300, 325, 350, 375, 400, 425, 450, 475, 500, 525, 550, 575, 600, 625, 650, 675, 700, 725, 750, 775, 800, 825, 850, 875, 900, 925, 950, 975, 1000, 1100, 1250, 1375, 1425, 1500, 1600, 1750, or 2000 ppm can be used as effective concentrations in the compositions and methods of the current invention. Even higher concentrations (up to 25 ppt, i.e. parts per thousand) can be made and may be useful in the current invention.
[0064] The composition of the present invention can comprise between about 1 ppm and about 25 ppt (25000 ppm) of the terpene component, preferably 100 to 2000 ppm of the terpene component, for example, 250, 500, 1000, 2000 ppm thereof.
[0065] The terpenes, surfactants, and other components of the invention may be readily purchased or synthesised using techniques generally known to synthetic chemists.
[0066] It is highly preferred that terpenes used in the present invention, for safety and regulatory reasons, are at least food grade terpenes (as defined by the United States FDA or equivalent national regulatory body outside the USA).
[0067] Optionally the composition can comprise other food-grade active compounds in addition to the terpene component, for example other antimicrobial agents, enzymes, or the like.
[0068] Optionally the composition can comprise a further active agents in addition to the terpene component, for example an antimicrobial agent, an anti-fungal agent, an insecticidal agent, an anti-inflammatory agent, an anaesthetic or the like. Suitable agents include: [0069] Anti-fungal: Cell wall hydrolyases (assuming they do not degrade the hollow glucan particle or cell wall particle), cell wall synthesis inhibitors, standard antifungals. [0070] Anti-bacterial: Antiseptics, cell wall hydrolases, synthesis inhibitors, antibiotics. [0071] Insecticidal: Natural insecticides, chitinase.
[0072] The composition can comprise an antioxidant to reduce oxidation of the terpene. An example of such an anti-oxidant might be rosemary oil, vitamin C or vitamin E.
[0073] The composition of the present invention can be in the form of a dry powder. The composition can be provided in combination with an agriculturally, food or pharmaceutically acceptable carrier or excipient in a liquid, solid or gel-like form.
[0074] For solid compositions, suitable carriers include pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, talc, cellulose, glucose, sucrose, magnesium carbonate, and the like. Suitably the formulation is in tablet or pellet form. As suitable carrier could also be a human or animal food material. Additionally, conventional agricultural carriers could also be used.
[0075] A pellet, tablet or other solid form of the composition can preferably also contain a dispersal agent which promotes dispersal of the composition when placed into a liquid, e.g. water. Suitable dispersal agents include xanthan gum, maltodextrin, alginates, or the like.
[0076] Liquid compositions can, for example, be prepared by dispersing the composition in water, saline, aqueous dextrose, glycerol, ethanol, or the like, to form a solution or suspension. If desired, these compositions can contain minor amounts of non-toxic auxiliary substances such as wetting or emulsifying agents, pH buffering agents (for example, sodium acetate, sorbitan monolaurate, triethanolamine sodium acetate or triethanolamine oleate). The methods of preparing such liquid compositions are known, or will be apparent, to those skilled in this art; for example see Remington: The Science and Practice of Pharmacy; Lippincott, Williams & Wilkins; (Dec. 15, 2000)--which is incorporated herein by reference. Again a liquid composition could be prepared by dispersing the composition in a liquid human or animal food or drink material. Additionally a suitable liquid agricultural excipient could be used.
[0077] For oral administration tablets and granules are generally preferred. Tablets may contain binders and lubricants. Fine powders or granules may contain diluting, dispersing and/or surface active agents and can be presented in water or in a syrup. Capsules or sachets can conveniently contain the composition in a dry state. Non-aqueous solutions or suspensions of the composition are also suitable and may contain suspending agents. Where desirable or necessary, flavouring, preserving, suspending, thickening, or emulsifying agents can be included. Of course, it would be suitable to use a food or drink material as an oral delivery method.
[0078] Parental administration is generally characterised by injection. For injectables it will be appreciated that, in general, all materials used in the composition and any excipient used must be of pharmaceutical grade. Injectables can be prepared in conventional forms, either as liquid solutions, emulsions or suspensions, solid forms suitable for dissolution, suspension in liquid prior to injection, or as emulsions. An alternative approach for parental administration involves use of a slow release or sustained release system, such that a constant level of dosage is maintained. See, for example, U.S. Pat. No. 3,710,795, which is incorporated by reference herein. Preparations for parenteral can also contain buffers, diluents and other suitable additives. Examples of non-aqueous solvents are propylene glycol, polyethylene glycol, vegetable oils (such as olive oil), and injectable organic esters (such as ethyl oleate). Aqueous carriers include water, alcoholic/aqueous solutions, emulsions, or suspensions, including saline and buffered media. Other parenteral vehicles include sodium chloride solution, Ringer's dextrose, dextrose and sodium chloride, lactated Ringer's, or fixed oils. Vehicles for intravenous use include fluid and nutrient replenishers, electrolyte replenishers (such as those based on Ringer's dextrose) and the like.
[0079] Preservatives and other additives can also be present such as, for example, antimicrobials, anti-oxidants, chelating agents, inert gases, and the like.
[0080] For topical administration liquids, suspension, lotions, creams, gels, ointments, drops, suppositories, sprays and powders may be used. Conventional pharmaceutical carriers, aqueous, powder or oily bases, thickeners, and the like can be used as necessary or desirable.
[0081] The present invention further provides a method of preparing a hollow glucan particle or cell wall particle encapsulating a terpene component, said method comprising the steps of; [0082] a) providing a terpene component; [0083] b) providing a hollow glucan particle or cell wall particle; [0084] c) incubating the terpene component with the glucan particle or cell wall particle under suitable conditions for terpene encapsulation; and [0085] d) recovering the hollow glucan particle or cell wall particle encapsulating the terpene component.
[0086] Optionally the above method can further comprise the step of drying the particles encapsulating the terpene component. Drying may be achieved in a number of ways and mention may be made of freeze drying, fluidised bed drying, drum drying or spray drying, all of which are well known processes.
[0087] In step a) of the above method, the terpene component is suitably provided as a suspension in an aqueous solvent, and optionally in the presence of a surfactant. Suitably the solvent is water. A suitable surfactant is Tween-80 (polyoxyethylenesorbitan monooleate), and preferably the surfactant is present at a concentration of about 0.1 to 10% by volume of the total reaction mixture, more preferably about 1%. Alternatively the terpene component may be provided as a true solution in a solvent, e.g. water. A true solution of terpene in water can be obtained by mixing the terpene in water at high shear until a true solution is obtained. Publication No WO 03/020024 provides further details of forming true solutions of terpenes in water.
[0088] In step b) of the above method, the hollow glucan particle or cell wall particle is suitably provided as a suspension in water or other suitable liquid. Suitably the suspension comprises approximately 1 to 1000 mg particles per ml, preferably 200 to 400 mg/ml. Alternatively the particles may be provided as a dry powder and added to the terpene-surfactant suspension.
[0089] Alternatively the particles are provided in sufficient liquid to minimally hydrate the particles, but not in significant excess. The term "hydrodynamic volume" (HV) is used to describe the volume of liquid required to minimally hydrate the particles. Thus suitably the particles: are provided with a volume ranging from the HV and a volume of 1.5 times the HV (1.5 HV). This makes the subsequent drying step more efficient. Also, where a low volume of liquid is used (i.e. around HV to 1.5 HV), it is also possible to extrude the finished product into pellet or noodle form, which is convenient for fluidised bed drying.
[0090] It has been found that the terpene component can become encapsulated by the hollow glucan particle or cell wall particle at room temperature. The rate of encapsulation is, however, increased at 37.degree. C. but the temperature should be kept below the boiling point or denaturing temperature of any component of the composition. Suitable conditions for step c) of the above method are therefore atmospheric pressure at a temperature of 20 to 37.degree. C. Optimisation of the conditions for a particular encapsulation reaction will be a matter of routine experimentation.
[0091] The present invention further provides a method of killing a microorganism, said method comprising the step of; [0092] a) contacting said microorganism with a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component.
[0093] Suitable compositions are those defined in more detail above.
[0094] The present invention further provides a method of preventing or treating an infection in a patient, said method comprising the step of; [0095] a) administering to said patient in a therapeutically effective dose, a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component.
[0096] Suitable compositions are those defined in more detail above.
[0097] The infection of the patient may be caused by any infectious agent. Examples of these infectious agents include, but are not restricted to Staphylococcus aureus, Aspergillius fumigatus, Mycoplasma iowae, Penicillium sp., and Mycoplasma pneumoniae.
[0098] For internal administration the composition may be administered orally, vaginally, rectally, by inhalation, or by parenteral routes, e.g. by intradermal, subcutaneous, intramuscular, intraperitoneal, intrarectal, intraarterial, intralymphatic, intravenous, intrathecal and intratracheal routes. Suitable formulations of the composition for these routes are discussed above.
[0099] For external treatment, the composition may be applied topically, for example as a cream or ointment or as a dry powder for treatment of a wound.
[0100] The amount of terpene administered in the above method should clearly be sufficient to achieve the desired result, i.e. prevention and/or treatment of the infection, but should not be at a level which will induce serious toxic effects in the patient.
[0101] The amount of composition administered will, of course, be dependent on the manner of administration, on the patient being treated, i.e. their weight, their age, condition, sex and extent of the disease in the subject and on the judgement of the prescribing physician. The dose, schedule of doses, and route of administration can be varied. One of skill in the art would readily be able to determine an anti-infective amount for a given application based on the general knowledge in the art and the procedures in the Examples given below. It should be noted that the term "patient" as used herein refers to any individual, either human or animal, to which the treatment is applied. Thus, the patient can be a domesticated animal (e.g., cat, dog, etc.), livestock (e.g., cattle, horse, pig, sheep, goat, etc.), laboratory animal (e.g., mouse, rabbit, rat, guinea pig, etc.), birds and fish. Suitably the subject is a mammal and especially a primate, for example a human.
[0102] In a further embodiment the present invention provides a method of treating or preventing infection of a plant, said method comprising the step of; [0103] a) administering in a therapeutically effective dose a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component to the plant or to soil in proximity to the plant.
[0104] Suitable compositions are those defined in more detail above.
[0105] Terpenes have been shown to eliminate a number of plant pathogens (see WO 03/020024) and, as described in co-pending application U.S. 60/538,627 also effectively kill nematodes which are significant plant parasites. Terpenes alone in suspension or solution, however, are somewhat unstable and degrade rapidly in the soil environment, thus losing efficacy.
[0106] Incorporation of a terpene component in a hollow glucan particle or cell wall particle can reduce the rate of terpene release and degradation, thus increasing the duration of action of the terpene in the soil.
[0107] Suitably the infection of a plant which is to be treated or prevented in the above method is infection by nematodes.
[0108] Other plant infections that may be treated or prevented include fungal plant infections, especially those affecting the surface of a plant. Such infections include downy mildew, powdery mildew or botrytis bunch rot; these infections particularly affect grape vines.
[0109] In one embodiment, the plant infection may be caused by one or more of the following: Aspergillius fumigatus, Sclerotinta homeocarpa, Rhizoctonia solani, Colletotrichum graminicola or Penicillium sp.
[0110] An advantage of a terpene based treatment of plants is that it can be applied shortly before harvest.
[0111] Many conventional treatments require an extended period before re-entry to the treated area (generally 3 weeks). This means that an outbreak of a plant disease shortly before harvest cannot be treated with conventional treatments as it would then not be possible to harvest the crop at the desired time. The compositions of the present invention can suitably be applied at any time up until harvest, for example 21 days prior to harvest, 14 days prior to harvest, 7 days prior to harvest, or even 3 days or less before harvest.
[0112] Encapsulated terpenes have shown particular efficacy in treating downy mildew, powdery mildew and botrytis bunch rot in grapes, and thus the present invention provides a method of treating or preventing these diseases.
[0113] Prevention of plant infections can be achieved by treating plants which the encapsulated terpenes regularly as a prophylactic measure.
[0114] Suitably the composition of the present invention is applied by spraying. This is particularly suitable for treating a plant disease which affects the surface of a plant. For spraying, a preparation comprising 2 g/l of the composition in water may be used. Concentrations of from 2 to 4 g/l are particularly effective, and concentrations of greater than 4 g/l can be used as required. Obviously it is important that the concentration of the composition used is sufficient to kill or inhibit the disease causing agent, but not so high as to harm the plant being treated.
[0115] When spraying plants a rate of 500 L/Ha or greater is suitable to cover the plants. Preferably a rate of 900 L/Ha or greater, more preferably 1200 L/Ha or greater is used to ensure good coverage. Where grape vines are being treated, a rate of 1200 L/Ha has proven suitably effective.
[0116] The composition of the present invention may alternatively be applied via irrigation. This is particularly suitable for treating nematodes or other soil borne pathogens or parasites.
[0117] In a further embodiment the present invention also provides a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component for use in the prevention or treatment of an infection in a patient or a plant. Suitable compositions are those defined in more detail above.
[0118] In a further embodiment the present invention provides the use of a composition comprising a hollow glucan particle or cell wall particle encapsulating a terpene component in the manufacture of a medicament for the treatment of infection caused by a micro-organism. Suitable compositions are those defined in more detail above.
[0119] The present invention will now by further described with reference to the following, non-limiting, examples and figures in which:
[0120] FIG. 1 represents a light micrograph of empty yeast cell walls;
[0121] FIG. 2 represents a light micrograph of yeast cell walls encapsulating L-carvone;
[0122] FIG. 3 represents a light micrograph of yeast cell walls encapsulating citral;
[0123] FIG. 4 represents a light micrograph of terpene emulsion;
[0124] FIG. 5 represents a light micrograph of yeast cell walls in hydrodynamic volume (HV) water;
[0125] FIG. 6 represents a light micrograph of yeast cell walls encapsulating terpene in 5 times hydrodynamic volume (HV) of water;
[0126] FIG. 7 represents a light micrograph of yeast cell walls encapsulating terpene in HV of water;
[0127] FIG. 8 represents a light micrograph of yeast cell walls encapsulating terpene in HV plus 5% of water;
[0128] FIG. 9 represents a light micrograph of yeast cell walls encapsulating terpene in HV plus 10% of water;
[0129] FIG. 10 represents a light micrograph of yeast cell walls encapsulating terpene in HV plus 20% of water;
[0130] FIG. 11 represents a light micrograph of yeast cell walls encapsulating terpene in HV plus 30% of water;
[0131] FIG. 12 represents a light micrograph of yeast cell walls encapsulating terpene in HV plus 40% of water.
[0132] FIG. 13 represents a light micrograph showing the dispersal of dried hollow glucan particles encapsulating a terpene component and no xanthan gum.
[0133] FIG. 14 represents a light micrograph as in FIG. 13 where 0.07 g of 1% xanthan gum is included.
[0134] FIG. 15 represents a light micrograph as in FIG. 13 where 0.14 g of 1% xanthan gum is included.
[0135] FIG. 16 represents a light micrograph as in FIG. 13 where 0.28 g of 1% xanthan gum is included.
[0136] FIG. 17 represents a light micrograph as in FIG. 13 where 0.55 g of 1% xanthan gum is included.
[0137] FIG. 18 represents a light micrograph as in FIG. 13 where 1.1 g of 1% xanthan gum is included.
[0138] FIG. 19 represents a light micrograph as in FIG. 13 where 2.2 g of 1% xanthan gum is included.
[0139] FIG. 20 represents a light micrograph as in FIG. 13 where 4.4 g of 1% xanthan gum is included.
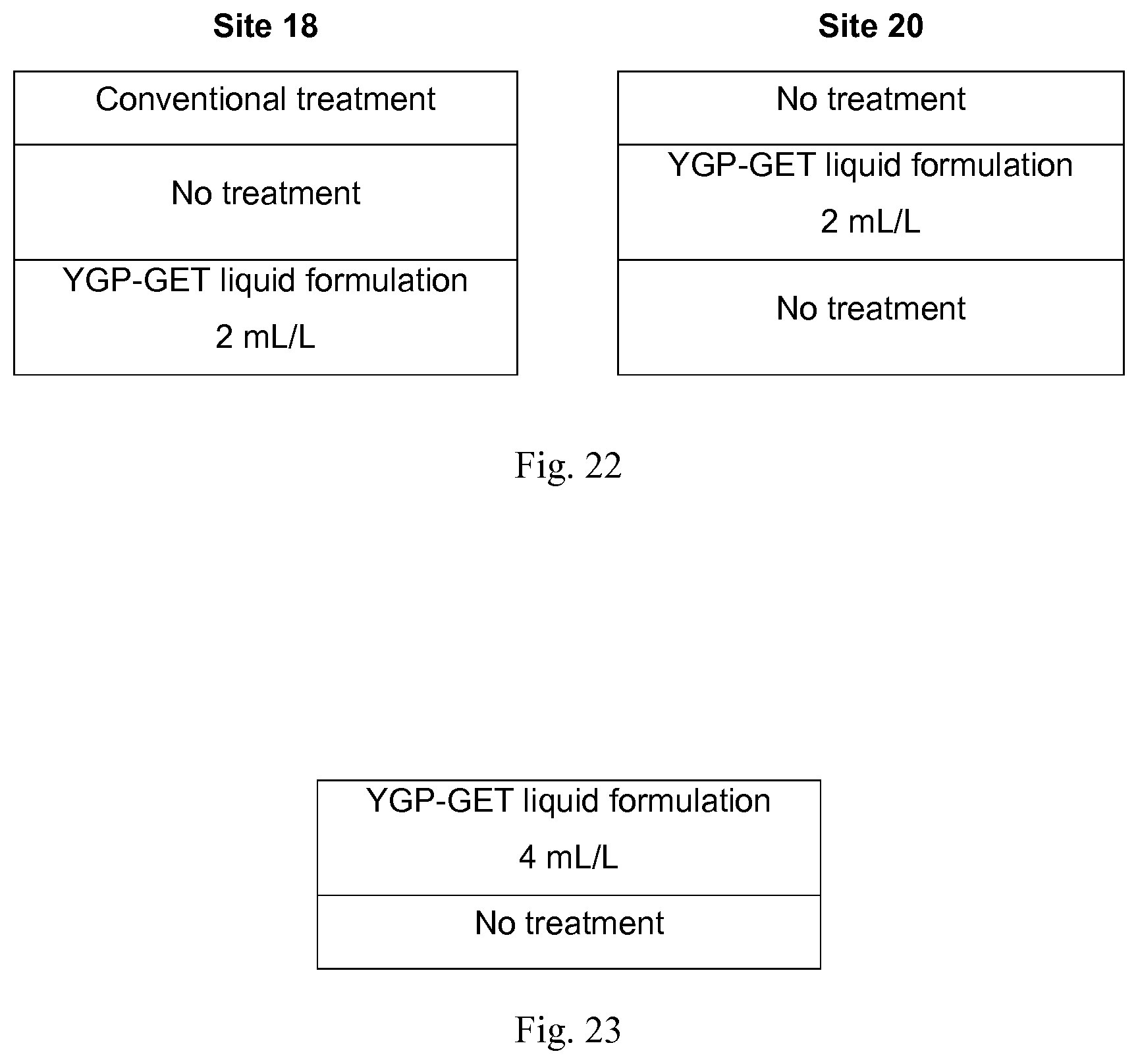
[0140] FIG. 21 shows a schematic representation of treatment areas on sites 18 and 20.
[0141] FIG. 22 shows a schematic representation of treatment areas on sites 18 and 20.
[0142] FIG. 23 shows a schematic representation of the treatment areas on site 7.
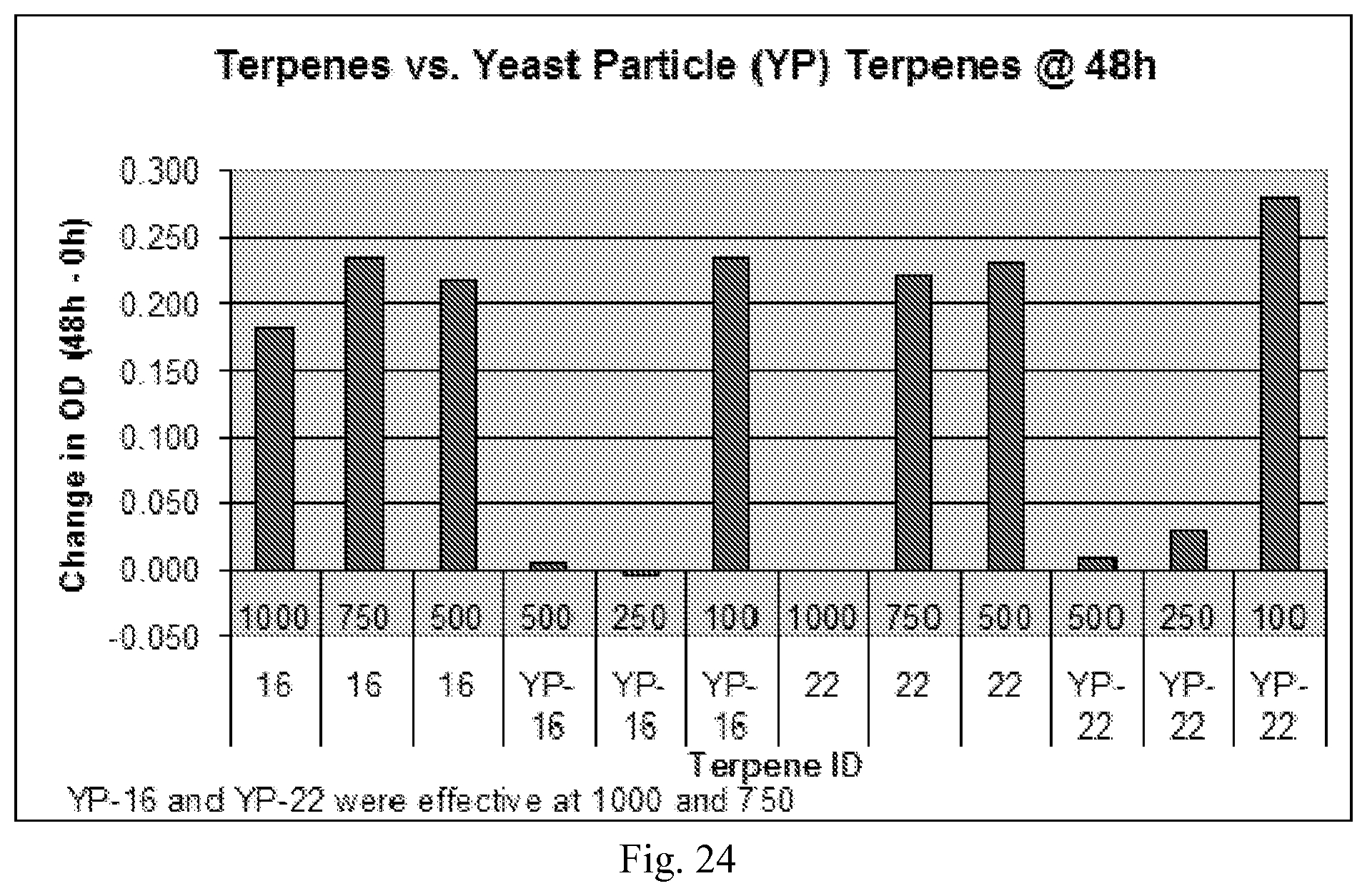
[0143] FIG. 24 shows a graph showing comparison of encapsulated vs. non-encapsulated terpene formulations.
[0144] The following examples are provided to further enable those of ordinary skill in the art to make or perform the present invention. They are purely exemplary and are not intended to limit the scope of the invention. Unless indicated otherwise, parts are parts by volume or parts by weight, as indicated, temperature is in degrees Celsius (.degree. C.) or is at ambient temperature, and pressure is at or near atmospheric. There are numerous variations and combinations of the compositions and conditions for making or using them, e.g., component concentrations, desired solvents, solvent mixtures, temperatures, pressures, and other ranges and conditions that can be used to optimise the results obtained from the described compositions and methods. Only reasonable and routine experimentation will be required to optimise these.
Example 1--Demonstration of Terpene Loading into Baker's Yeast Particles and Purified Yeast Glucan Particles
[0145] The following protocol was performed to demonstrate that terpenes would load into yeast cell walls and other hollow glucan particles.
[0146] Emulsions of citral and L-carvone were prepared by mixing 150 .mu.l of the terpene with 100 .mu.l of 10% Tween 80 in water and 250 .mu.l of water.
[0147] Baker's yeast particles (YP) or Levacan.TM. yeast glucan particles (YGP), available from Savory Systems International, Inc., Branchburg, N.J., were mixed with water to form a 250 mg/ml suspension.
[0148] 500 .mu.l of the YP or YGP suspension and 250 .mu.l of the terpene emulsion were mixed together and incubated overnight under constant agitation. 500 .mu.l YP or YGP suspension and 500 .mu.l of water were used as a control. The particles were then washed with water until free from external emulsion. The particle preparations were then frozen and lyophilised until dry.
[0149] The particles were then rehydrated and examined under light microscope. The results are shown in FIGS. 1 to 4.
[0150] FIG. 1 shows spherical structures with a dark area at their centre, these are empty hollow glucan particles. FIGS. 2 and 3 shows spherical structures with a swollen appearance with a light coloured interior, these are particles with terpene encapsulated in the central cavity--citral in FIG. 2 and L-carvone in FIG. 3. In FIGS. 2 and 3 small blobs of free terpene can also be seen, e.g. at the top of FIG. 2, just left of centre. FIG. 4 shows the terpene emulsion as small blebs of terpene suspended in water.
Example 2--Determination of Maximal Citral and L-Carvone Loading Levels in Baker's Yeast Cell Wall Particles (YP)
[0151] The following protocol was performed to determine the maximal amounts of terpenes that would load into YP. [0152] L-carvone and citral emulsions were prepared by sonicating 4.5 g of the terpene with 0.3 ml water. [0153] 10% Tween-80 solution was prepared by sonicating 4.5 g Tween-80 in 40.5 mls water. [0154] YP suspension was prepared by mixing YP with water to form 20 mg/ml suspension. [0155] Encapsulation reactions were set up as described in Table 1.
[0156] Citral or L-carvone-water emulsion was mixed with YP and Tween 80 surfactant overnight at room temperature. Samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer was scored. The results are shown in the right hand column labelled free terpene of Table 1.
[0157] The expression "free terpene" refers to the visible presence of terpene in the centrifuged reaction mixture. The absence of free terpene indicates complete absorption of the terpene by the particles. The highest volume of terpene absorbed by the particles, as evidenced by the absence of free terpene, was recorded as the maximal volume of absorbed terpene emulsion.
TABLE-US-00001 TABLE 1 20 mg/ml 10% YP Terpene Vol Tween-80 Free Tube .mu.l Emulsion .mu.l .mu.l Terpene 1 500 -- -- 500 - 2 500 L-carvone 0.5 500 - 3 500 L-carvone 1.65 500 - 4 500 L-carvone 5 495 - 5 500 L-carvone 16.5 483.5 - 6 500 L-carvone 50 450 + 7 500 L-carvone 165 335 + 8 500 L-carvone 500 -- + 9 500 Citral 0.5 500 - 10 500 Citral 1.65 500 - 11 500 Citral 5 495 - 12 500 Citral 16.5 483.5 +/- 13 500 Citral 50 450 + 14 500 Citral 165 335 + 15 500 Citral 500 -- +
[0158] As can be seen from the results, YP is capable of absorbing and encapsulating at least 16.5 .mu.l of L-carvone terpene emulsion or at least 5 .mu.l of citral emulsion per 10 mg of YP.
Example 3--Demonstration of Improved Terpene Loading with Surfactant and Determination of Optimal Tween-80:Terpene Ratio
[0159] The following protocol was performed to demonstrate that the presence of surfactant improves terpene loading and to determine the minimum level of Tween-80 surfactant required for the YP terpene loading reaction. [0160] L-carvone and citral emulsions were prepared by sonicating 4.5 g of the terpene with 0.3 ml water. [0161] 10% Tween-80 solution was prepared by sonicating 4.5 g Tween-80 in 40.5 ml water. [0162] Baker's YP suspension was prepared by mixing YP with water to form 250 mg/ml suspension.
[0163] Loading reactions were set up as shown in Table 2 below.
[0164] Citral or L-carvone-water emulsion was mixed with YP with 0-10% v/v Tween 80 surfactant overnight at room temperature. Samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer was scored. The results are shown in the right hand column labelled free terpene of Table 2.
[0165] The expression "free terpene" refers to the visible presence of terpene in the centrifuged reaction mixture. The absence of free terpene indicates complete absorption and encapsulation of the terpene by the YP. The highest volume of terpene absorbed by the YP, as evidenced by the absence of free terpene, was recorded as the maximal volume of absorbed terpene emulsion.
TABLE-US-00002 TABLE 2 250 mg/ml 10% YP Terpene Vol Tween-80 Water Free Tube ml Emulsion .mu.l .mu.l .mu.l Terpene 1 500 -- -- -- 500 - 2 500 L-carvone 150 0 350 Sl 3 500 L-carvone 150 5 345 Sl 4 500 L-carvone 150 10 340 Sl 5 500 L-carvone 150 33 317 Sl 6 500 L-carvone 150 100 250 - 7 500 L-carvone 150 200 150 - 8 500 L-carvone 150 350 -- - 9 500 L-carvone 400 0 100 ++ 10 500 L-carvone 400 5 95 ++ 11 500 L-carvone 400 10 90 ++ 12 500 L-carvone 400 33 77 ++ 13 500 L-carvone 400 100 -- + 14 500 L-carvone 400 20 .mu.l 100% 30 + 15 500 Citral 113 0 387 + 16 500 Citral 113 5 382 + 17 500 Citral 113 10 377 + 18 500 Citral 113 33 354 Sl 19 500 Citral 113 100 287 Sl 20 500 Citral 113 200 187 - 21 500 Citral 113 350 37 - 22 500 Citral 250 0 250 ++ 23 500 Citral 250 5 245 ++ 24 500 Citral 250 10 240 ++ 25 500 Citral 250 33 217 + 26 500 Citral 250 100 150 + 27 500 Citral 250 20 .mu.l 100% 230 + Sl = slight
[0166] As can be seen from the results a Tween-80 concentration of 1% (i.e. 100 .mu.l of 10% Tween-80 in 1000 .mu.l of reaction mixture) is sufficient to allow complete uptake of the terpene in the above reaction. A 2% Tween-80 causes no improvement in results, whereas with a 0.33% concentration free terpene was observed. This indicates that: [0167] a) Terpenes are absorbed into YP particles in the absence of a surfactant, but the presence of surfactant significantly increases terpene absorption. [0168] b) A Tween-80 concentration of around 1% is optimum for YP loading as it ensures proper loading whilst maximising the terpene payload of the YP particles.
Example 4--Determination of Maximal Terpene Loading and Encapsulation at High Baker's Yeast Cell Wall Particles (YP) Levels
[0169] The following protocol was performed to determine the maximal amounts of terpenes that would load into YP at high YP levels. [0170] L-carvone and citral emulsions were prepared by sonicating 4.5 g of the terpene with 3 ml 1% Tween. [0171] 5% Tween-80 solution was prepared by sonicating 0.5 g Tween-80 in 9.5 ml water. [0172] YP suspension was prepared by mixing YP with water to form 250 mg/ml suspension. [0173] Encapsulation reactions were set up as shown in Table 3.
[0174] Citral or L-carvone-water emulsion was mixed with YP and Tween 80 surfactant overnight at room temperature. Samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer was scored. The results are shown in the right hand column labelled free terpene of Table 3.
[0175] The expression "free terpene" refers to the visible presence of terpene in the centrifuged reaction mixture. The absence of free terpene indicates complete absorption of the terpene by the YP. The highest volume of terpene absorbed by the YP, as evidenced by the absence of free terpene, was recorded as the maximal volume of absorbed terpene emulsion.
TABLE-US-00003 TABLE 3 250 1% mg/ml YP Terpene Vol Tween-80 Free Tube .mu.l Emulsion .mu.l .mu.l Terpene 1 500 -- -- 500 - 2 500 L-carvone 15 485 - 3 500 L-carvone 37.5 462.5 - 4 500 L-carvone 75 425 - 5 500 L-carvone 112.5 387.5 - 6 500 L-carvone 150 350 Sl+ 7 500 L-carvone 225 275 + 8 500 L-carvone 450 50 + 9 500 Citral 15 485 - 10 500 Citral 37.5 462.5 - 11 500 Citral 75 425 - 12 500 Citral 112.5 387.5 Sl+ 13 500 Citral 150 350 + 14 500 Citral 225 275 + 15 500 Citral 450 50 +
[0176] As can be seen from the results in Table 3, YP is capable of absorbing and encapsulating terpenes at high YP concentration. YP absorbed and encapsulated at least 112.5 .mu.l of L-carvone terpene emulsion or at least 75 .mu.l of citral emulsion per 125 mg of YP. This demonstrates that the terpene encapsulation reaction is independent of YP concentration within the ranges tested.
Example 5--Screen Commercially Available Particles for Terpene Absorption
[0177] The following protocol was performed to analyse the loading properties of different types of particles. The particles studied were Baker's Yeast Cell Wall Particles (Sigma Chemical Corp., St. Louis, Mo.), Nutrex.TM. Walls (Sensient Technologies, Milwaukee, Wis.), SAF-Mannan.TM. (SAF Agri, Minneapolis, Minn.), Nutricept Walls.TM. (Nutricepts Inc., Burnsville, Minn.), Levacan.TM. (Savory Systems International, Inc., Branchburg, N.J.) and WGP.TM. (Alpha-beta Technology, Inc. Worcester, Mass.).
[0178] L-carvone and citral emulsions were prepared by sonicating 7 g terpene+3 ml 3.3% Tween-80.
[0179] Table 4 below compares the purity with the number of yeast particles per mg and the packed solids weight/volume ratio.
TABLE-US-00004 TABLE 4 Purity Yeast % Beta 1,3- No. Mg Particle glucan particles/mg particles/ml Bakers 11.2 4 .times. 10.sup.7 250 Nutrex 24.5 1.7 .times. 10.sup.8 58.8 SAF Mannan 33.4 2.4 .times. 10.sup.8 41.7 Nutricepts 55.7 5.2 .times. 10.sup.8 37 Levacan 74.6 1 .times. 10.sup.8 19.2 WGP 82.1 3.5 .times. 10.sup.8 10
[0180] From Table 4 it can be concluded that the number of particles per mg is inversely proportional to purity. Thus the number of particles per mg of WGP is almost 10-fold higher than Baker's YP.
[0181] The YP suspensions were prepared as follows: [0182] Baker's yeast cell wall particle suspension (YP) was prepared by mixing 250 mg YP/ml 1% Tween 80. [0183] Nutrex suspension was prepared by mixing 163 mg Nutrex YGP/ml 1% Tween 80. [0184] SAF Mannan suspension was prepared by mixing 234 mg Biospringer YGP/ml 1% Tween 80. [0185] Nutricepts suspension was prepared by mixing 99 mg Nutricepts YGP/ml 1% Tween 80. [0186] Levacan suspension was prepared by mixing 217 mg Lev YGP/ml 1% Tween 80. [0187] WGP suspension was prepared by mixing 121 mg WGP YGP/ml 1% Tween 80.
[0188] The packed volume of the above particles is identical which means that equal numbers of particles were assayed.
[0189] Loading reactions were set up as shown in Table 5 and left to incubate overnight. Samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer and the color of the encapsulated terpenes in the pellet was scored. The results are shown in the two right hand columns of Table 5. The highest volume of terpene absorbed by particles as evidenced by the absence of free terpene was recorded as the volume of absorbed terpene emulsion.
TABLE-US-00005 TABLE 5 conc Terpene Vol 1% Tween Free Tube Particle mg/ml .mu.l Emulsion .mu.l 80 .mu.l Terpene Colour 1 Baker's 250 500 L-carvone 125 375 - W 2 Nutrex 163 500 L-carvone 125 375 - W 3 SAF Mannan 234 500 L-carvone 125 375 - W 4 Nutricepts 99 500 L-carvone 125 375 + W 5 Levacan 217 500 L-carvone 125 375 + W 6 WGP 121 500 L-carvone 125 375 + W 7 Baker's 250 500 Citral 100 375 - Y 8 Nutrex 163 500 Citral 100 375 - Y 9 SAF Mannan 234 500 Citral 100 375 - W 10 Nutricepts 99 500 Citral 100 375 + Y 11 Levacan 217 500 Citral 100 375 + int 12 WGP 121 500 Citral 100 375 + int 13 -- -- -- L-carvone 125 875 + -- 14 -- -- -- Citral 100 900 + Y W = white; Y = yellow; sl = slight; int = intermediate
[0190] From the results the following conclusions were reached: [0191] Purified particles with a low lipid content were less effective at absorbing terpenes. [0192] Less pure particles were more effective at absorbing terpenes. [0193] Yellow degradation product of citral was not formed when encapsulated in SAF-Mannan.TM.. [0194] Based on qualitative loading at the single terpene level tested, SAF Mannan.TM. appears to be best, Nutrex.TM. second and Baker's third.
Example 6--Kinetics of Terpene Loading into Various Types of Particles and Different Incubation Temperatures
[0195] The following protocol was adopted to compare the loading kinetics of various types of yeast particles.
[0196] L-carvone and citral emulsions were prepared by sonicating 7 g terpene with 3 ml 3.3% Tween-80.
[0197] 1% Tween-80 solution was prepared by sonicating 1 ml 10% Tween-80 in 10 ml water. [0198] Baker's YP was prepared by mixing 5 g of bakers YP in 20 ml 1% Tween-80. [0199] Nutrex.TM. YGP suspension was prepared by mixing 2 g Nutrex.TM. YGP in 20 ml 1% Tween-80. [0200] SAF Mannan.TM. suspension was prepared by mixing 2 g SAF Mannan.TM. in 20 ml 1% Tween-80.
[0201] Loading reactions were set up as shown in Table 6.
[0202] The reactions were incubated for 1, 3, 6, 9 and 24 hours at room temperature or 37.degree. C. After incubation samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer was scored. The results are shown in the two right hand columns of Table 6. The highest volume of terpene absorbed by the particles as evidenced by the absence of free terpene was recorded as the volume of absorbed terpene emulsion. Colour of the encapsulated pellet was scored at 24 hours.
TABLE-US-00006 TABLE 6 T conc Terpene Vol 1% Free Terpene (hr) Tube .degree. C. Particle mg/ml .mu.l Emulsion .mu.l Tween-80 1 3 6 9 24 Color 1 Rt Bakers 250 3500 L-carvone 788 2712 + - - - - W 2 37 Bakers 250 3500 L-carvone 788 2712 + - - - - W 3 Rt Nutrex 100 3500 L-carvone 1050 2450 + - - - - W 4 37 Nutrex 100 3500 L-carvone 1050 2450 + - - - - W 5 Rt SAF 100 3500 L-carvone 1050 2450 <+ - - - - W 6 37 SAF 100 3500 L-carvone 1050 2450 <+ - - - - W 7 Rt Bakers 250 3500 Citral 525 2975 + - - - - Y 8 37 Bakers 250 3500 Citral 525 2975 + - - - - VY 9 Rt Nutrex 100 3500 Citral 788 2712 + - - - - Y 10 37 Nutrex 100 3500 Citral 788 2712 + - - - - VY 11 Rt SAF 100 3500 Citral 788 2712 + - - - - W 12 37 SAF 100 3500 Citral 788 2712 + - - - - W White, W; Yellow, Y; Very Yellow, VY; Room Temperature, Rt
[0203] From the results shown in Table 6 and other observations the following conclusions can be made: [0204] Terpene loading reaction takes between 1 and 3 hours. [0205] Terpene loading occurs faster at 37.degree. C. than at room temperature. [0206] SAF Mannan.TM. appears to be preferable particles for two reasons: [0207] Faster and more complete uptake of both terpenes. [0208] Citral remains stable when loaded as evidenced by the absence of yellow colour, characteristic of citral degradation, after 24 hours at 37.degree. C.
Example 7--Screen a Range of Single Terpenes and Terpene Combinations for Particle Loading
[0209] The following protocol was adopted to compare the loading efficiency of Baker's YP versus SAF Mannan.TM..
[0210] Terpene emulsions were prepared as follows: [0211] L-carvone--4.5 g L-carvone in 1.5 ml 3.3% Tween-80. [0212] Citral--4.5 g citral in 1.5 ml 3.3% Tween-80. [0213] Thymol/L-carvone mixture (T/L)--2.25 g thymol and 2.25 g L-carvone in 1.5 ml 3.3% Tween-80. [0214] Eugenol--4.5 g eugenol in 1.5 ml 3.3% Tween-80. [0215] Geraniol--4.5 g geraniol in 1.5 ml 3.3% Tween-80. [0216] Citral/L-carvone/Eugenol mixture (C/L/E)--1.5 g citral, 1.5 g L-carvone, 1.5 g eugenol in in 1.5 ml 3.3% Tween-80.
[0217] Emulsions composed of terpene:water:surfactant ratio of 0.75:0.3:0.05 were used for these experiments.
[0218] Increasing volumes of terpene emulsion were mixed with 250 mg/ml Baker's YP or 250 mg/ml SAF Mannan.TM. overnight at room temperature as shown in Tables 7 and 8. Samples were centrifuged at 14,000.times.g for 10 minutes and the appearance of free terpene floating on the aqueous layer was scored. The highest volume of terpene emulsion absorbed by Baker's YP or SAF Mannan.TM. as evidenced by the absence of free terpene was recorded as the volume of absorbed terpene emulsion. Colour of encapsulated terpenes in the pellet was recorded. The results in Tables 7 and 8 show that all single and terpene combinations were efficiently loaded into both Baker's YP or SAF Mannan particles.
TABLE-US-00007 TABLE 7 Evaluation of Baker's YP Loading of Different Terpenes and Terpene Mixtures. Baker Terpene Vol 1% Tween- Free Tube (.mu.l) Emulsion (.mu.l) 80 (.mu.l) Terpene Colour 1 500 -- -- 500 - W 2 500 L-carvone 15 485 - W 3 500 L-carvone 37.5 462.5 - W 4 500 L-carvone 7 425 +/- W 5 500 L-carvone 112.5 387.5 +/- W 6 500 L-carvone 150 350 + W 7 500 L-carvone 225 275 + W 8 500 L-carvone 450 50 ++ W 9 500 Citral 15 485 - Y 10 500 Citral 37.5 462.5 - Y 11 500 Citral 75 425 - Y 12 500 Citral 112.5 387.5 +/- Y 13 500 Citral 150 350 + Y 14 500 Citral 225 275 + Y 15 500 Citral 450 50 + Y 16 500 T/L 15 485 - W 17 500 T/L 37.5 462.5 - W 18 500 T/L 75 425 - W 19 500 T/L 112.5 387.5 +/- W 20 500 T/L 150 350 + W 21 500 T/L 225 275 + W 22 500 T/L 450 50 + W 23 500 Eugenol 15 485 - W 24 500 Eugenol 37.5 462.5 - W 25 500 Eugenol 75 425 - W 26 500 Eugenol 112.5 387.5 +/- W 27 500 Eugenol 150 350 + W 28 500 Eugenol 225 275 + W 29 500 Eugenol 450 50 + W 30 500 Geraniol 15 485 - W 31 500 Geraniol 37.5 462.5 - W 32 500 Geraniol 75 425 - W 33 500 Geraniol 112.5 387.5 + W 34 500 Geraniol 150 350 + W 35 500 Geraniol 225 275 + W 36 500 Geraniol 450 50 + W 37 500 C/L/E 15 485 - Y 38 500 C/L/E 37.5 462.5 - Y 39 500 C/L/E 75 425 - Y 40 500 C/L/E 112.5 387.5 +/- Y 41 500 C/L/E 150 350 + Y 42 500 C/L/E 225 275 + Y 43 500 C/L/E 450 50 + Y
TABLE-US-00008 TABLE 8 Evaluation of SAF Mannan Loading of Different Terpenes and Terpene Mixtures. SAF Terpene 1% Tween- Free Tube (.mu.l) Emulsion Vol 80 (.mu.l) Terpene Colour 1 500 -- -- 500 - W 2 500 L-carvone 15 485 - W 3 500 L-carvone 37.5 462.5 - W 4 500 L-carvone 75 425 - W 5 500 L-carvone 112.5 387.5 - W 6 500 L-carvone 150 350 +/- W 7 500 L-carvone 225 275 +/- W 8 500 L-carvone 450 50 + W 9 500 Citral 15 485 - W 10 500 Citral 37.5 462.5 - W 11 500 Citral 75 ul 425 - W 12 500 Citral 112.5 387.5 - W 13 500 Citral 150 350 +/- W Inverted 14 500 Citral 225 275 + W Inverted 15 500 Citral 450 50 + W Inverted 16 500 T/L 15 485 - W 17 500 T/L 37.5 462.5 - W 18 500 T/L 75 425 - W 19 500 T/L 112.5 387.5 - W 20 500 T/L 150 350 +/- W 21 500 T/L 225 275 + W 22 500 T/L 450 50 + W 23 500 Eugenol 15 485 - W 24 500 Eugenol 37.5 462.5 - W 25 500 Eugenol 75 425 - W 26 500 Eugenol 112.5 387.5 +/- W 27 500 Eugenol 150 350 + W 28 500 Eugenol 225 275 + W 29 500 Eugenol 450 50 + W 30 500 Geraniol 15 485 - W 31 500 Geraniol 37.5 462.5 - W 32 500 Geraniol 75 425 - W 33 500 Geraniol 112.5 387.5 - W 34 500 Geraniol 150 350 - W 35 500 Geraniol 225 275 - W Inverted 36 500 Geraniol 450 50 + W Inverted 37 500 C/L/E 15 485 - W 38 500 C/L/E 37.5 462.5 - W 39 500 C/L/E 75 425 - W 40 500 C/L/E 112.5 387.5 - W 41 500 C/L/E 150 350 - W 42 500 C/L/E 225 275 +/- W 43 500 C/L/E 450 50 + W Inverted = Phase Inverted - solids floating on top - no free oil; W = white; Y = yellow.
[0219] From the results the following observations were made: [0220] All terpenes appeared to load into Baker's YP and SAF Mannan. [0221] SAF Mannan has a higher terpene loading capacity than bakers YP. [0222] The two and three way mixtures of terpenes also appear to efficiently load. [0223] The terpene Eugenol appears to have a higher density than the particles and water as it was found associated with the pellet. [0224] For the SAF Mannan, the higher load levels and lighter particles resulted in loaded particles floating on the surface of the aqueous layer for citral and geraniol. [0225] Citral was protected from oxidation by the SAF Mannan but not by the Baker's YP.
[0226] The approximate maximal loading for each particle type was determined and is shown in Tables 9 and 10 below. Percentage loaded represents a ratio of the amount of terpene loaded to the amount of particle present (weight for weight).
TABLE-US-00009 TABLE 9 Maximal terpene loading in Baker's YP. Terpene Vol. Loaded .mu.l % Loaded w/w L-carvone 37.5 33.3 Citral 75 67% Thymol/L-carvone 1:1 75 67% Eugenol 75 67% Geraniol 75 67% Citral/L-carvone/ 75 67% Eugenol (1:1:1)
TABLE-US-00010 TABLE 10 Maximal terpene loading in SAF Mannan. Terpene Vol. loaded .mu.l % Loaded w/w L-carvone 112.5 100% Citral 150 133% Thymol/L-carvone 1:1 112.5 100% Eugenol 112.5 100% Geraniol 150 133% Citral/L-carvone/ 150 133% Eugenol (1:1:1)
Example 8--Evaluation of Terpene Stability in Aqueous Emulsions and Encapsulated Terpene Formulations
[0227] Terpene stability was assessed by the observation of citral formulations for the formation of a yellow colored oxidation product. As noted in the right hand column in Tables 5-8 citral emulsions and citral encapsulated Bakers YP turned a progressively increasing yellow color over time. However, citral encapsulation in SAF Mannan.TM. increased citral stability as evidenced by a reduction or absence of yellow color over time.
Example 9--Loading of Terpenes in Minimal Water
[0228] The following protocol was carried out to evaluate the possibility that terpene loading and encapsulation into YP could be carried out at a very high Yeast Particles (YP) solids level to allow for direct extrusion of the loaded formulation into a fluidised bed drier. The minimal amount of water to completely hydrate the SAF Mannan.TM. particles was determined to be 3.53 g water per g solids. This defines the hydrodynamic volume (HV) or water absorptive capacity of the particles. At this level of water the hydrated particles have a consistency of a stiff dough which is thixotropic, i.e. shear thinning like mayonnaise. Addition of water up to 40% above the HV results in a thick flowable paste. The standard reaction that has been used in the above examples was carried out at 3.times.HV water.
[0229] A series of terpene (L-carvone) loading reactions were carried out keeping the ratio of particle:terpene:Tween (1:0.44:0.04) constant and varying the amount of water in the system from the HV (3.53 g) to HV+40% water (4.92 g). Controls were the standard loading system which uses 3.times.HV water, particles only and terpene only reactions. Following overnight incubation samples of the mixtures were evaluated microscopically for free terpene and evidence of terpene uptake into the particles and for material flow characteristics by assessing flow in inverted tubes over 15 minutes. In addition, the presence of free oil was assessed by hydrating the reaction mixture with 5.times.HV, vortexing to obtain a complete dispersion of particles and centrifugation to sediment the particle encapsulated terpene. The results are shown in Table 11 and FIGS. 7 to 12. FIGS. 7 to 12 show the loading results of the following tubes: [0230] FIG. 7--Tube 3 [0231] FIG. 8--Tube 5 [0232] FIG. 9--Tube 6 [0233] FIG. 10--Tube 8 [0234] FIG. 11--Tube 10 [0235] FIG. 12--Tube 11
TABLE-US-00011 [0235] TABLE 11 SAF Terpene Weight Water Free Tube g Emulsion (g) (g) Terpene Flow 1 -- L-carvone 4.64 4.5 + + 2 1 -- -- 8.0 - + 3 1 L-carvone 4.64 4.5 - + 4 1 L-carvone 4.64 -- - - 5 1 L-carvone 4.64 0.17 - - 6 1 L-carvone 4.64 0.35 - - 7 1 L-carvone 4.64 0.52 - Sl 8 1 L-carvone 4.64 0.7 - Mod 9 1 L-carvone 4.64 0.87 - High 10 1 L-carvone 4.64 1.05 - High 11 1 L-carvone 4.64 1.39 - High
[0236] The results shown in Table 11 and FIGS. 7 to 12 demonstrate that terpene loading and encapsulation into the particles occurred at all water ratios evaluated. Surprisingly, equivalent loading occurred even when the loading reaction was taking place in a reaction with the consistency of a stiff dough using the minimal amount of water to hydrate the particles. The absence of free terpene was observed microscopically (FIGS. 7 to 12) and in the low level of terpene in the supernatants, as evidenced by a marked reduction in the turbidity of the supernatant compared to the terpene only control.
[0237] These results extend our understanding of the conditions to load terpenes into hollow glucan particles. The flexibility to use a minimal volume of water to hydrate the particles during the loading process will allow loading of the terpenes under conditions where the reaction mixture is a malleable dough-like consistency using standard food-grade swept surface dough mixers. The consistency of the final high solids terpene loaded mixture is suitable for direct extrusion to form noodles and pellets for fluidised bed drying.
[0238] Suitable facilities to scale up production in this manner would require: [0239] Gaulin homogeniser, or equivalent to produce stable terpene emulsion. [0240] Swept surface dough mixing tank. [0241] Extruder. [0242] Fluidised bed drier.
Example 10--Evaluation of an Interstitial Hydrocolloid Agent to Aid Dispersion in Dried Hollow Glucan Particles Encapsulating a Terpene Component Dispersion when Re-Hydrated
[0243] The following protocol was adopted to evaluate the effect of an interstitial hydrocolloid to increase dried hollow glucan particle encapsulated terpene formulations to disperse when hydrated. [0244] SAF Mannan.TM. particles [0245] 0.1% Tween 80 [0246] L-carvone [0247] Xanthan Gum--1% w/v in 0.1% Tween 80
[0248] The effect of increasing xanthan gum levels on dry hollow glucan particle encapsulated L-carvone dispersion in water was assessed by loading L-carvone into SAF Mannan by incubating 1.1 g of an L-carvone emulsion (L-carvone:water:surfactant ratio of 0.75:0.3:0.05) with 1 g SAF Mannan and 4.4 g 0.1% Tween 80 containing 0-1% xanthan gum as shown in Table 12.
TABLE-US-00012 TABLE 12 L-carvone 0.1% 1% SAF Emulsion Tween-80 Xanthan Visual Tube g (g) (g) (g) Observations 1 1 1.1 4.4 0 Large non- uniform clumps 2 1 1.1 4.33 0.07 Uniform suspension 3 1 1.1 4.26 0.14 Uniform suspension 4 1 1.1 4.12 0.28 Uniform suspension 5 1 1.1 3.85 0.55 Uniform suspension 6 1 1.1 3.3 1.1 Finer Uniform suspension 7 1 1.1 2.2 2.2 Finer Uniform suspension 8 1 1.1 0 4.4 Finer Uniform suspension
[0249] The results in Table 12 and FIGS. 13 to 20 demonstrate that the inclusion of a high molecular weight hydrocolloid during the drying of the particle encapsulated terpene aids in the hydration and dispersion of the microparticles into a uniform suspension. Other examples of such hydrocolloid agents are maltodextrin, alginates, or the like.
[0250] It may also be worthwhile to include a pellet coating to increase the stability of the loaded terpenes, and to provide a sustained release of terpene.
Example 11--Evaluation of Minimum Inhibitory Concentration (MIC) of Terpene Emulsions, Fresh Baker's YP and SAF Mannan Encapsulated Terpenes and Freeze-Dried Baker's YP and SAF Mannan Encapsulated Terpenes Against S. aureus
[0251] The results of a protocol performed to compare the MIC of fresh versus freeze dried hollow glucan particle encapsulated terpene formulations are shown below in Table 13. A simple terpene emulsion was also tested and the results are shown for comparison.
TABLE-US-00013 TABLE 13 MIC .mu.g/ml terpene Bakers SAF Mannan Freeze Freeze Terpene Emulsion Fresh Dried Fresh Dried L-carvone 3.75 0.1 >0.04 0.01 >0.02 Citral 0.94 0.01 0.05 0.01 >0.03 L-carvone/ 0.23 0.01 0.03 0.01 0.05 Thymol Eugenol 0.12 0.03 0.05 0.01 0.05 Geraniol 0.47 0.03 0.06 0.02 >0.03 L-carvone/ 0.23 0.03 0.06 0.02 0.05 Citral/Eugenol
[0252] The conclusions taken from the above results were: [0253] Terpene loading into hollow glucan particles appears to enhance terpene MIC. Generally the fresh terpene emulsions are-4-375 fold less potent than the encapsulated formulations [0254] Terpenes loaded in SAF Mannan.TM. perform slightly better than Baker's YP. [0255] Freshly loaded terpene compositions perform slightly better than freeze dried compositions (there may be some volatilisation of terpenes from dry compositions during freeze drying). [0256] Terpenes in aqueous emulsions are stable for at least 3 weeks.
Example 12--Efficacy of Encapsulated Terpenes at Pilot Plant Scale Against S. aureus
[0257] Anti-microbial assays were carried out with encapsulated terpenes and mixtures produced at the pilot plant scales against S. aureus. Both the fresh and freeze dried encapsulated terpene samples containing materials demonstrated strong anti-microbial activities. The results are summarised in Table 14 below.
[0258] Terpenes were encapsulated in SAF-Nannan.TM. at a 2.5 Kg scale. A mixture of three terpenes (Geraniol, 275 g; Eugenol, 385 g; and thymol, 440 gram was dissolved and homogenized with 100 g Tween-80 and 8 L of water. SAF-Mannan.TM. (2.5 Kg) was added to form a homogenous suspension. The suspension was passed through a Gaulin homogenizer to reduce particle size and the homogenate was incubated overnight at room temperature. A sample of the encapsulated terpene was removed and stored at room temperature. The remaining encapsulated terpene was then frozen in trays and freeze dried. The freeze dried encapsulated terpene powder was ground and stored at room temperature.
TABLE-US-00014 TABLE 14 Material MIC (ppm) Staphylococcus aureus assays YGP empty shell control >2500 Pilot Plant - Fresh 100 Pilot Plant - Freeze dried 100
[0259] At the pilot plant scale both the fresh and freeze dried samples were equally potent on a w/w terpene basis.
[0260] Based on the large scale preparation results, the predicted effective dose of the freeze dried formulation against S. aureus is 200 ppm (the formulation contains .about.50% terpene w/w) or 0.2 g/L water.
Example 13--Efficacy of Encapsulated Terpenes Against Mycobacterium
[0261] Terpene emulsions were prepared as follows: [0262] Citral--4.5 g citral in 1.5 ml 3.3% Tween-80. [0263] L-carvone/eugenol--2.25 g L-carvone and 2.25 g Eugenol in 1.5 ml 3.3% Tween-80. [0264] Eugenol--4.5 g eugenol in 1.5 ml 3.3% Tween-80. [0265] Geraniol--4.5 g geraniol in 1.5 ml 3.3% Tween-80. [0266] Geraniol/thymol mixture--2.25 g geraniol and 2.25 g thymol in 1.5 ml 3.3% Tween-80. [0267] Control emulsion--6 ml 1% Tween-80.
[0268] SAF-Mannan.TM. (2.5 g) was mixed with 3 ml of each emulsion and 7 ml of 1% Tween 80 and incubated overnight to encapsulate the terpenes and terpene mixtures. The encapsulated terpene formulations were frozen and freeze dried and the powders ground to a fine powder. Suspensions of encapsulated terpenes (25 mg/ml) and unencapsulated terpene emulsions were assayed for antibacterial activity against Mycobacterium. The results are set out in Table 15
TABLE-US-00015 TABLE 15 Material MIC (ppm) Mycobacterial assays YGP Citral FD 250 YGP L-Carvone/Eugenol FD 500 YGP Eugenol FD 500 YGP Geraniol FD 125 YGP Geraniol/Thymol FD 62.5 Control Emulsion >1000 Citral Emulsion 35 L-carvone/Eugenol Emulsion 53 Eugenol Emulsion 105 Gernaniol Emulsion 70 Geraniol/Thymol Emulsion 53 FD = (Freeze Dried)
Example 14--Nematocidal Activity of Encapsulated Terpenes
[0269] Preparations of yeast cell walls encapsulating citral were prepared according to the procedures described above. The hollow glucan particles contained 17.5% citral, and the particles were present at in the test preparations at a concentration of 1000 ppm. This means that terpenes were effectively present at a concentration of 175 ppm.
[0270] 1.0 ml of the test preparations was added to 0.1 to 0.15 ml of water containing root-knot nematodes. 1.0 water was added to the nematodes as the control.
[0271] Observations were made as previously described and the kill rate assessed (i.e. percentage dead) after 24 and 48 hrs. The results shown below in Table 16 are an average of 2 sets of results.
TABLE-US-00016 TABLE 16 Nematicidal activity of encapsulated terpene solution (17.5% citral @ 1000 ppm) Kill Rate Time Test Control 24 h 45 17 48 h 56 21
[0272] The results demonstrate that hollow glucan particles encapsulating terpenes are effective at killing root-knot nematodes at a particle concentration of 1000 ppm, which corresponds to a citral concentration of only 175 ppm.
[0273] Thus hollow glucan particles encapsulating terpenes appear to be as effective as terpenes in solution or with surfactant as nematicides. The nematicidal activity is retained despite the terpene being encapsulated within the particle. It can be expected that higher concentrations of terpenes within the hollow glucan particles, or higher concentrations of the particles would result in an even higher kill rate, as is the case for terpenes in solution or with surfactant.
Example 15--Fugicidal Properties of Encapsulated and Non-Encapsulated Terpenes
[0274] The following protocols were carried out to assess the fungicidal properties of various terpene combinations, and to compare the efficacy of encapsulated and non-encapsulated compositions.
[0275] Assessment of Anti-Fungal Properties of Different Terpene Formulation
[0276] A microtitre plate assay was used to assess the minimum inhibitory concentration (MIC) of a range of terpene compounds against different pathogenic organisms. The assay used for each organism is described in detail later but important general features are as follows.
[0277] The assay uses two incubation periods to distinguish between static (growth inhibition) and cidal (killing) activities. The first incubation period allows assessment of growth inhibition, but cannot distinguish between merely prevention of growth and killing of the cells. The purpose of the second incubation period is to allow sufficient time and nutrients for any dormant or inhibited cells that survive terpene exposure to proliferate. Any cells that were inhibited by fungistatic effects should respond and grow during the second incubation period, whereas cells that were killed by exposure to terpenes will not grow in the fresh medium.
[0278] Initial screening experiments were carried out using a total of 31 different terpene formulations (Table 17). These experiments were repeated using a subset of strongly active terpene formulations (Table 18).
[0279] A combination of the terpenes geraniol, eugenol and thymol in a ratio of 2:1:2 encapsulated within glucan particles was also tested; this sample is referred to as YP-GET. A non-encapsulated geraniol, eugenol and thymol combination in the same ratio was also tested for comparison with the encapsulated form.
[0280] MIC Assay Using Saccharomyces cerevisiae
[0281] S. cerevisiae (5.times.10.sup.5 cells/mL in YPD growth medium) were added to each well of a 96-well microtitre plate in 100 .mu.L aliquots. At least one column per plate was designated as a cell-only control and no terpene was added to these wells. Aliquots (100 .mu.L) of different terpene formulations were added to the first row of the remaining columns, and serial 2-fold dilutions were performed by transferring 100 .mu.L from one row to the next a total of 7 times. Finally, 100 .mu.L was discarded from the last row in order to ensure that all wells contained the same volume. Microtitre plates were incubated statically overnight at 30.degree. C.
[0282] Following incubation, plates were scored for inhibition of growth (evidenced by a lack of turbidity). Growth inhibition (75%) was visually confirmed by microscopy.
[0283] Once the MIC had been determined for each formulation, the microtitre plates were centrifuged and the spent medium was removed from non-turbid wells. The cells were resuspended in fresh medium (100 .mu.L) and the plates were re-incubated overnight at 30.degree. C. Assessment of growth inhibition was performed as before.
[0284] MIC Assay Using a Mixed Inoculum
[0285] The different terpene formulations were serially diluted in the 96-well microtitre plate as described for S. cerevisiae. Molten YPD agar was then added to the wells, together with 5 .mu.L mixed inoculum (prepared from mouldy grape leaves to a concentration of 5.times.10.sup.4 cells/mL). The plates were incubated statically for 24 hours at room temperature and spore growth was visually assessed by microscopy.
[0286] Due to the use of solid medium, the second incubation period in fresh media could not be performed.
[0287] MIC Assay Using Colletotrichum graminicola
[0288] The different terpene formulations were serially diluted in the 96-well microtitre plate as described for S. cerevisiae. C. graminicola (300 spores/well) were added to the diluted terpenes and the plates were incubated statically for 48 hours at room temperature. Spore germination and growth were visually assessed by microscopy.
[0289] Once the MIC had been determined for each formulation, the microtitre plates were centrifuged and the spent medium was removed from growth-inhibited wells. The spores were resuspended in fresh medium (100 .mu.L) and the plates were re-incubated overnight at room temperature. Assessment of growth inhibition was performed as before.
TABLE-US-00017 TABLE 17 MIC and fungicidal MIC values obtained from initial screening of 31 terpene formulations Saccharomyces Mixed Colletotrichum cerevisiae microbes graminicola Terpene Cidal Cidal Cidal formulation .sup.a MIC MIC MIC MIC MIC MIC 1 Geraniol (G) 500 500 250 NT 63 63 2 Eugenol (E) 500 500 125 NT 125 125 3 Thymol (T) 250 250 63 NT 63 500 4 Citral (C) 250 250 63 NT 125 63 5 L-carvone (L) 250 500 63 NT 125 125 6 GE 1000 2000 125 NT 63 250 7 GT 500 500 250 NT 125 63 8 GC 500 500 125 NT 125 250 9 GL 500 500 125 NT 125 125 10 ET 500 500 125 NT 125 125 11 EC 250 1000 31 NT 125 125 12 EL 500 1000 125 NT 125 125 13 TC 500 500 16 NT 63 63 14 TL 500 1000 63 NT 63 63 15 CL 500 500 .ltoreq.8 NT 63 63 16 GET 500 500 23 NT 94 94 17 GEC 250 500 94 NT 94 94 18 GEL 500 1000 188 NT 188 188 19 GTC 500 500 47 NT 188 188 20 GTL 500 1000 94 NT 94 94 21 GCL 250 500 94 NT 47 47 22 ETC 125 250 188 NT 94 94 23 ETL 500 1000 .ltoreq.12 NT 94 94 24 ECL 500 1000 .ltoreq.12 NT 188 188 25 TCL 500 1000 23 NT 94 375 26 GETC 500 1000 125 NT 250 500 27 ETCL 500 1000 63 NT 125 125 28 GTCL 500 1000 125 NT 250 250 29 GECL 500 1000 .ltoreq.16 NT 500 500 30 GETL 1000 1000 125 NT 500 250 31 GECTL 1000 1000 78 NT 625 625 GET (2:1:2 NT NT 98 NT 78 156 ratio, w/w/w) YP-GET 98 391 98 NT 20 20 (G:E:T ratio of 2:1:2, w/w) .sup.b NT, not tested; YP-GET, yeast-encapsulated GET formulation. .sup.a Terpene combinations were mixed in a 1:1 (w/w) ratio unless otherwise indicated. .sup.b MICs calculated by terpene content.
TABLE-US-00018 TABLE 18 Repeat assay to determine MIC and fungicidal MIC values Mixed microbes isolated from Saccharomyces mouldy grape Colletotrichum Terpene cerevisiae leaves .sup.b graminicola formulation .sup.a Cidal Cidal Cidal (by No.) MIC MIC MIC MIC MIC MIC T (3) NT NT 63 NT NT NT L (5) NT NT 250 NT NT NT GE (6) NT NT NT NT 125 500 EC (11) 125 250 NT NT NT NT TC (13) NT NT 250 NT 63 250 TL (14) NT NT 500 NT 250 500 CL (15) NT NT 500 NT 125 500 GET (16) NT NT 375 NT 188 375 GEC (17) 250 500 NT NT NT NT GCL (21) 250 500 NT NT 375 750 ETC (22) 125 250 NT NT 94 188 ETL (23) NT NT 375 NT 188 750 ECL (24) NT NT 750 NT NT NT TCL (25) NT NT 750 NT 94 375 ETCL (27) NT NT 500 NT 63 500 GECL (29) NT NT 1000 NT NT NT YP-GET 98 195 NT NT 39 156 (G:E:T ratio of 2:1:2, w/w) .sup.c NT, not tested; YP-GET, yeast-encapsulated GET formulation. NOTE: Samples were assayed in duplicate. If different values were obtained between duplicate samples, the higher value has been presented. No duplicate samples differed by more than one 2-fold dilution. .sup.a Terpene combinations were mixed in a 1:1 (w/w) ratio unless otherwise indicated. .sup.b 1 .times. 10.sup.4 cells/mL stock suspension. .sup.c MICs calculated by terpene content.
[0290] Mixed Inoculum
[0291] Using a mixed inoculum presents a number of problems. The variability in spore content between preparations results in poor interassay reproducibility, and growth of contaminating organisms impedes the scoring of spore germination. Unicellular yeast species are particularly problematic in masking spore growth. Although precise data could not be obtained from this assay, an inhibitory effect of terpenes was observed.
[0292] Identification of spores was easier during scoring of the repeat assay than during the initial screening assay as a larger number of spores were used (approximately 50/well versus approximately 10/well). Therefore, data obtained during the repeat assay may provide a more reliable estimate of MIC.
[0293] Colletotrichum graminicola
[0294] The generally higher MIC values obtained from the repeat assay compared to the initial screening assay may be due to: [0295] use of 1-week-old terpene solutions [0296] use of freshly prepared spores, which had a higher viability than those used in the initial screening assay and may therefore be more difficult to kill.
[0297] Comparison of Terpene Formulations as Free Emulsions with the Same Terpene Formulations when Encapsulated in Hollow Glucan Particles: Saccharomyces cerevisiae MIC Assays
[0298] YPD growth medium (100 .mu.L) was added to each well of a 96-well microtitre plate and aliquots of different terpene formulations were added to the first row, giving a total volume of 200 .mu.L in this row. One column was designated as a cell-only control and no terpene was added to these wells. Serial 2-fold dilutions were performed by transferring 100 .mu.L from one row to the next a total of 7 times. Finally, 100 .mu.L was discarded from the last row in order to ensure that all wells contained the same volume. S. cerevisiae (5.times.10.sup.5 cells/mL in YPD growth medium) were then added to each well in 100 .mu.L aliquots, and the absorbance at 620 nm (A.sub.620) was measured for each well using a microtitre plate reader. Microtitre plates were incubated statically overnight at 30.degree. C.
[0299] Following incubation, the A.sub.620 was measured again and plates were scored for inhibition of growth (.gtoreq.75%). Growth inhibition was visually confirmed by microscopy.
[0300] For the free terpene emulsions, once the MIC had been determined for each formulation, the microtitre plates were centrifuged and the spent medium was removed from the growth-inhibited wells. The cells were resuspended in fresh medium (100 .mu.L) and the plates were re-incubated overnight at 30.degree. C.
[0301] Assessment of growth inhibition was performed as before.
[0302] MIC and fungicidal MIC results are summarised in Table 19.
Results
TABLE-US-00019 [0303] TABLE 19 MIC and fungicidal MIC values obtained from screening of 31 terpene formulations against Saccharomyces cerevisiae Terpene formulation .sup.a Yeast-encapsulated (Reference formulations .sup.b, c Free terpene emulsions No) MIC Cidal MIC MIC Cidal MIC G (1) 111 NT 250 250 E (2) 131 NT 125 250 T (3) 115 NT 125 250 C (4) 118 NT 125 250 L (5) 254 NT 250 500 GE (6) 118 NT 250 500 GT (7) 108 NT 125 250 GC (8) 113 NT 125 250 GL (9) 117 NT 250 500 ET (10) 131 NT 125 250 EC (11) 126 NT 125 250 EL (12) 129 NT 125 250 TC (13) 59 NT 63 63 TL (14) 124 NT 63 125 CL (15) 124 NT 125 125 GET (16) 119 NT 63 125 GEC (17) 119 NT 125 250 GEL (18) 121 NT 125 125 GTC (19) 115 NT 125 125 GTL (20) 119 NT 125 125 GCL (21) 234 NT 125 125 ETC (22) 124 NT 125 125 ETL (23) 123 NT 125 125 ECL (24) 63 NT 63 125 TCL (25) 61 NT 125 500 GETC (26) 61 NT 63 250 ETCL (27) 120 NT 63 125 GTCL (28) 124 NT 125 125 GECL (29) 125 NT 125 125 GETL (30) 122 NT 125 250 GECTL (31) 120 NT 125 250 GET (2:1:2 .sup. 125 .sup.d NT 125 250 ratio, w/w/w) YP-GET 125 NT .sup. 125 .sup.c .sup. 250 .sup.c (G:E:T ratio of 2:1:2, w/w) YP-ETC 125 NT .sup. 125 .sup.c .sup. 250 .sup.c (E:T:C ratio of 1:1:1, w/w) NT, not tested; YP-GET, yeast-encapsulated GET formulation; YP-ETC, yeast-encapsulated ETC formulation. .sup.a Terpene combinations were mixed in a 1:1 (w/w) ratio unless otherwise indicated. .sup.b Yeast-encapsulated formulations unless otherwise indicated. .sup.c MIC calculated by terpene content. .sup.d Non-encapsulated emulsion formulation.
[0304] For both the terpene emulsions and yeast-encapsulated terpenes, MICs were typically .ltoreq.125 ppm, with the most active formulations inhibiting growth at .about.60 ppm. MIC values obtained for the terpene emulsions were similar to those obtained for their respective yeast-encapsulated formulations. When different values were obtained, they only differed by approximately one 2-fold dilution.
[0305] Many of the free terpene emulsions were fungicidal at the growth inhibitory MIC, with the majority showing fungicidal activity at a 2-fold higher concentration.
[0306] These results demonstrate that terpenes encapsulated in glucan particles are at least as effective at killing fungus as non-encapsulated forms.
[0307] Additionally the encapsulated compositions used may have had reduced potency due to having been stored for 45 days at 4.degree. C. and having a sub-optimal terpene content of .about.4% w/w.
[0308] The assay to determine fungicidal activity involves a centrifugation step, which attempts to separate the microbial cells from any residual terpene in the growth medium by producing a pellet of cells at the bottom of the well. This pellet is then resuspended in fresh media and incubated for a second time in the absence of terpene. However, the centrifugation step cannot discriminate between microbial cells and yeast particles, therefore when yeast-encapsulated terpenes are used, the cell pellet will also contain terpene-loaded yeast particles. As a result, both the yeast particles and the microbial cells are then resuspended in the fresh medium.
[0309] This methodology issue is not considered to affect the results obtained in the experiments described above for the following reasons. [0310] In previous experiments, terpene emulsions have been used instead of terpene-loaded yeast particles and fungicidal activity has been clearly shown. [0311] Encapsulated terpenes are released by diffusion, and an equilibrium between the concentration of encapsulated terpenes and the concentration of released terpenes in the surrounding medium is quickly reached. Thus, following centrifugation and resuspension in fresh medium, the concentration of released terpene in the growth medium is likely to be well below that required for growth inhibitory activity. [0312] There was no growth when the contents of the fungicidal MIC well were plated onto solid agar growth medium. When plated onto solid growth medium, diffusion of any residual terpene throughout the large volume of the agar plate results in a local terpene concentration that is too low to cause growth inhibition. The lack of growth from the contents of the fungicidal MIC well must therefore be due to initial fungicidal activity. In contrast, when an MIC was obtained that was lower than the fungicidal MIC and the contents of the MIC well were plated onto solid agar growth medium, growth was observed, indicating a fungistatic effect.
Example 16--Preparation of Encapsulated Terpene Compositions for Field Trials
[0313] The purpose of the following protocol was to encapsulate a terpene composition into hollow glucan particles for subsequent field trials.
[0314] Materials:
[0315] Thymol (supplied by Alpha-Gamma Corporation)
[0316] Eugenol (supplied by Alpha-Gamma Corporation)
[0317] Geraniol (supplied by Alpha-Gamma Corporation) 1% Tween-80 (supplied by Alpha-Gamma Corporation)
[0318] Yeast Cell Wall Particles
[0319] Xanthan Gum.
[0320] The yeast cell wall particles were obtained from Biorigin (Sao Paolo, Brazil) under the trade name Nutricell MOS 55, and were manufactured by Acucareira Quata S. A, Usina Quata, Quata--Sao Paolo--Brazil--Zip Code 19780 000. The particles are a spray dried cell wall extract of S. cerevisiae and are a free flowing powder of light beige to tan colour.
[0321] Protocol: The following protocol was suitable for a 1 Kg of particles, but can simply be scaled up for larger production. [0322] 1. Prepare terpene mixture--mix 375 grams of Geraniol+525 grams Eugenol+600 grams of Thymol and stir in a glass flask. [0323] 2. Prepare 6.2 L of 1% Tween 80 by mixing 62 grams Tween 80 in 6.2 L water in 2 gallon white bucket. Mix to form solution. [0324] 3. Add 6.2 grams Xanthan Gum to Tween solution and stir to dissolve. [0325] 4. Prepare terpene emulsion by mixing 1.5 Kg terpene mixture+6.2 L 1% Tween 80/0.1% Xanthan gum in white bucket using polytron mixer. [0326] 5. Add 1,000 grams of yeast cell wall particles--mix using paint mixer to form uniform suspension. [0327] 6. Add the terpene emulsion of step 4 to the yeast cell wall particles while mixing to form a thin mayonnaise-like consistency. [0328] 7. Pour terpene mixture into cans and incubate overnight.
[0329] Results: Encapsulated geranoil, eugenol and thymol in hollow glucan particles was obtained as a paste. The paste was easily converted to a dry powder by conventional spray drying techniques. The paste is the "liquid" composition referred to in the following protocols, and the "powder" is the spray dried form.
Example 17--Field Trials of Encapsulated Terpene Composition on Downy Mildew
[0330] In grapes, downy mildew is caused by the fungus Plasmopara viticola, which infects vineyards worldwide and can cause devastating losses for grape-growers in terms of crop yield and wine quality. The fungus attacks the fruits and all green parts of the vine, causing the leaves to wither and the flowers and berries to rot. The disease manifests as irregular pale yellow or yellow-green spots on the upper surface of leaves, with dense, white-grey, cotton-like fungal growth covering the underside of the leaf lesions. Berries may also be covered with the downy growth and, depending on the time of infection, may turn brown and soft or may not soften at all. Downy mildew is spread through the dispersal of spores by the wind and rain, and requires wet conditions for infection. It is particularly problematic in environments with high humidity. Preventative measures are recommended for management of the disease, with early applications of fungicides followed by repeat applications at appropriate intervals. Resistance has arisen to some treatments, and although the development of resistance can be minimised by rotating the use of different fungicides, it remains a problem.
[0331] The purpose of this trial was to investigate the efficacy of the encapsulated terpene formulation of Example 16 (YGP-GET) supplied as a liquid or powder (spray dried) formulation, for the prevention of downy mildew in grapes.
[0332] Four adjacent blocks, each covering 0.1 ha, were identified on site 20 in the Kir-Yianni vineyard.
[0333] Kir-Yianni is a 35 ha vineyard at an elevation of 300 m. It is bordered by a mixed oak forest on the north and west, and overlooks orchards and vineyards to the south and east.
[0334] All four blocks had been treated with multiple products prior to application of the terpene formulation. On 26 Jun. 2004, two of the four blocks were sprayed with the terpene powder formulation at a dose of either 0.5 g/L or 2 g/L (see schematic illustration in FIG. 21). A third block was treated with conventional Bordeaux mix plus wettable sulphur, and the remaining block was left untreated. The vines in each block were monitored for signs of downy mildew over the following week.
[0335] Four further adjacent blocks, each covering 0.1 ha, were identified on site 18 in the Kir-Yianni vineyard. All four blocks had been treated with multiple products prior to application of the terpene formulation. On 26 Jun. 2004, two of the four blocks were sprayed with the terpene liquid formulation at a dose of either 1 g/L or 4 g/L (FIG. 21) (note: 1 g of the terpene liquid formulation has a volume of 1 ml). Of the remaining two blocks, one was left untreated and one was sprayed with Mikal.RTM., a conventional treatment for downy mildew, on 28 Jun. 2004. The vines in each block were monitored for signs of downy mildew over the following week.
[0336] For both sites, the terpene product was applied at a rate of 1200 L/ha.
[0337] The following growth stages of the grapes were recorded: [0338] bud break, 26 Mar. 2004 [0339] bloom, 1 Jun. 2004 [0340] veraison, 6 Aug. 2004
[0341] The study applications took place pre-veraison.
[0342] The 2004 growing season was exceptionally late and was wet throughout. Disease pressure from downy mildew was extremely high, botrytis levels were elevated, and powdery mildew pressure was moderate. Both the powder and liquid YGP-GET formulations were stored at room temperature. No special storage conditions were used.
[0343] Details of Comparator Products
[0344] Powder formulation trial: Bordeaux mix, manufactured by Manica Spa, Italy, packed in Greece by Moscholios Chemicals SA; wettable sulphur.
[0345] Liquid formulation trial: Mikal.RTM. (fosetyl-al 50%, folpet 25%), manufactured by Bayer CropScience, distributed in Greece by Bayer Hellas SA.
[0346] The comparator products were applied as follows: One application before bud-break at a dosage of 15 g/L followed by two more applications per year at a dosage of 6.5 g/L. A spraying rate of 1000 L/ha was used for all three applications.
[0347] Powder formulation trial: Bordeaux mix (2 g/L) and Wettable sulphur (2.2 g/L) were applied on 26 Jun. 2004.
[0348] Liquid formulation trial: Mikal (3.2 g/L) was applied on 28 Jun. 2004.
[0349] Vines were visually examined for symptoms of downy mildew. Onset of the disease was marked by an average of two oily spots per leaf. Treatments that prevented the appearance of further spots were considered to provide effective protection against downy mildew.
[0350] Results
[0351] YGP-GET Powder Formulation (Spray Dried)
[0352] The conventional treatment of Bordeaux mixture provided good protection against downy mildew. Mild symptoms of downy mildew were observed in the control vines. The 0.5 g/L terpene product concentration did not provide protection, and the 2 g/L terpene product concentration provided only slightly better protection than the control. Note: the disease pressure at this site was very low because of the recent pesticide treatment.
[0353] Difficulties were encountered in dissolving the powder formulation as it was very fine, resulting in dispersion in the air. This may have adversely affected the efficacy of the product.
[0354] YGP-GET Liquid Formulation
[0355] When administered at a dose of 4 g/L, the terpene product provided excellent protection against downy mildew on exposed canopy. No protection was provided by the 1 g/L dosage. Serious symptoms of downy mildew were observed in the control block.
[0356] The liquid formulation was easy to use and had a pleasant odour.
[0357] Discussion
[0358] Downy mildew can cause devastating losses for grape-growers because of its effects on crop yield and wine quality. Management of the disease focuses on prevention because, once established, the infection can quickly spread. At the site sprayed with the powder formulation, YGP-GET did not exhibit efficacy at the lower dosage (0.5 g/L), and the dose of 2 g/L was less effective than the conventional treatment. At this site, the recent pesticide applications resulted in low disease pressure, which may have limited the apparent efficacy of the terpene treatment. However, it was considered that a dosage of less than 2 g/L of the terpene product was inadequate.
[0359] At the site sprayed with the liquid formulation, excellent protection of exposed canopy was provided by the higher dose level of 4 g/L. Excessive vegetative growth at this site resulted in more effective treatment of the outer, younger branches compared with the older growth in the inner canopy. Complete foliar coverage by the terpene product is useful, as the treatment is not systemic. It is estimated that an approximately 30% increase over the volume used for conventional systemic treatments would achieve good coverage using the terpene treatment.
[0360] Conclusions:
[0361] Foliar application of YGP-GET liquid formulation was highly effective at controlling downy mildew at a concentration of 4 g/L. The lower concentrations of 0.5 g/L powder and 1 g/L liquid were not effective.
Example 18--Field Trials of Encapsulated Terpene Composition on Powdery Mildew
[0362] Powdery mildew of grapes is caused by the fungus Uncinula necator, and causes reductions in vine growth, fruit quality and winter hardiness of vines. In wine grapes, an infection level of only 3% of berries can affect wine quality. The disease is characterised by small white-grey patches of fungal growth that enlarge into a powdery, white coating on the leaves. The fungal growth can also occur on the berries, which may split. In contrast to downy mildew, which requires warm wet conditions, powdery mildew can be a problem in drier growing seasons, as it favours shaded areas with humid but not rainy weather conditions. Preventative measures are recommended for management of powdery mildew, with early applications of fungicides followed by repeat applications at appropriate intervals.
[0363] This study aimed to investigate the efficacy of application of the YGP-GET composition for the prevention of powdery mildew in grapes.
[0364] Three adjacent blocks, each covering 0.1 ha, were identified on site 18 in the Kir-Yianni vineyard. On 19 Jul. 2004, one of the three blocks was sprayed with the YGP-GET liquid formulation at a dose of 2 ml/L and one was left untreated. The remaining block was sprayed with the conventional treatment of Equesion (2.5 g/L), Alliete (0.9 g/L) and Punch (0.075 mL/L) (see FIG. 22). The vines in each block were monitored for signs of powdery mildew over the following week.
[0365] Three further adjacent blocks, each covering 0.1 ha, were identified on site 20 in the Kir-Yianni vineyard. On 20 Jul. 2004, one of the three blocks was sprayed with the YGP-GET liquid formulation at a dose of 2 mL/L and the two remaining blocks were left untreated (see FIG. 22). The vines in each block were monitored for signs of powdery mildew over the following week.
[0366] At both sites, the blocks had previously been treated with multiple products, including a prior application of terpene product.
[0367] All terpene treatments were applied at a rate of 1200 L/ha to ensure complete coverage.
[0368] The following growth stages of the grapes were recorded [0369] bud break, 26 Mar. 2004 [0370] bloom, 1 Jun. 2004 [0371] veraison, 6 Aug. 2004
[0372] The study applications took place pre-veraison.
[0373] The 2004 growing season was exceptionally late and was wet throughout. Disease pressure from downy mildew was extremely high, botrytis levels were elevated, and powdery mildew pressure was moderate.
[0374] Details of Comparator Products
[0375] No comparator product was used at site 20. The comparator treatment used at site 18 is detailed below.
[0376] Punch.RTM. (flusilazole 40%), DuPont.
[0377] On 19 Jul. 2004, Punch was applied at a dose of 0.075 ml/L as a preventative treatment for powdery mildew according to the manufacturer's instructions.
[0378] Details of Additional Products
[0379] No additional products were used at site 20. The additional products used at site 18 are detailed below.
[0380] Equesion system (famoxadone 22.5% plus cymoxanil 30%) Alliete (fosetyl-al 80%)
[0381] On 19 Jul. 2004, Equesion (2.5 g/L) and Alliete (0.9 g/L) were applied as preventative treatments for downy mildew. The dose was determined according to the manufacturer's instructions.
[0382] The comparator and additional products represent conventional treatments in the integrated pest management schedule.
[0383] Vines were visually examined for symptoms of powdery mildew.
[0384] Results:
[0385] Site 18
[0386] Approximately 20% of the peduncles and stems in the control block were black, indicating moderate infection from powdery mildew. In both the conventional treatment block and the terpene-treated block, all stems and bunches were green, indicating that adequate protection had been provided.
[0387] Site 20
[0388] No evidence of powdery mildew infection was observed in any of the blocks.
[0389] Additional Observations
[0390] At the end of the growing season, the blocks at sites 18 and 20 generally showed less stress due to disease than the rest of the vineyard.
[0391] Powdery mildew infections cause considerable losses to growers through reductions in vine growth, fruit quality and winter hardiness of vines. Furthermore, wine quality can be affected by an infection level of as little as 3% of berries. Management of the disease focuses on prevention because, once established, the infection can quickly spread. In this study, the application of terpene product YGP-GET at site 18 effectively prevented powdery mildew infection, and the level of control exhibited by the terpene product was comparable to that provided by the conventional treatment. The results from site 20 are inconclusive, however, due to the lack of powdery mildew infection. This lack of infection is likely to be due to the extensive application of pesticides prior to the study, which resulted in low disease pressure.
[0392] The lower level of stress due to disease at sites 18 and 20 suggests that the earlier terpene treatment applied at these sites may have been beneficial in control of infection in the long term.
[0393] Conclusions:
[0394] YGP-GET effectively prevented powdery mildew infection, with a comparable level of control to that provided by the conventional treatment.
Example 18--Further Field Trials of Encapsulated Terpene Composition on Powdery Mildew
[0395] The study aimed to further investigate the efficacy of YGP-GET for the treatment of powdery mildew in Grimson Seedless table grapes.
[0396] A 0.1 ha plot on the Tsigaras vineyard (approximately 80 km south of the Kir-Yianni vineyard) was inadvertently left untreated during an application of Cisteine on 1 Jul. 2004. The vines in this plot subsequently showed severe symptoms of powdery mildew on the leaves, stems and grapes. On 12 Jul. 2004, the untreated plot was sprayed with 3 ml/L liquid YGP-GET formulation at a rate of 1200 l/ha, and the rest of the vineyard was sprayed with the comparator product Rogana. The vines were assessed for symptoms of powdery mildew after 24 hours.
[0397] Vines were trained in a high lyre trellis system.
[0398] Details of Comparator Product
[0399] Rogana (fenbuconazol 5%, binocap 16%), manufactured by BASF (BASF Agro Hellas S. A., Athens, Greece) On 12 Jul. 2004, Rogana was applied to the Tsigaras vineyard as a treatment for powdery mildew. The dose was determined according to the manufacturer's instructions.
[0400] Vines were visually examined for symptoms of powdery mildew.
[0401] Results
[0402] Severe symptoms of powdery mildew were evident prior to application of YGP-GET. Only 24 hours after YGP-GET application, the white bloom of the powdery mildew turned black, indicating effective antifungal activity. As the disease was effectively halted at this time, no further treatments were applied. YGP-GET showed comparable efficacy to the conventional treatment.
[0403] Discussion
[0404] In this study, an established powdery mildew infection was treated quickly and effectively using YGP-GET. Only 24 hours after application, the previously severe powdery mildew infection was halted by application of the terpene product, with comparable efficacy to the conventional treatment. The preliminary data obtained from this study suggest that YGP-GET may be efficacious in treating established fungal infections in addition to showing preventative ability.
Example 19--Further Field Trials of Encapsulated Terpene Composition on Powdery Mildew
[0405] Background and Rationale
[0406] In the current trial, the use of YGP-GET was investigated as part of a Tasmanian vineyard's (Frogmore Creek Vineyard, Hathaway Trading Pty Ltd, Box 187, Richmond TAS 7025, Australia) experimental programme to control powdery mildew using organic products. The aim of this study was to investigate the short-term efficacy of the application of YGP-GET in the organic control of powdery mildew in Chardonnay grapevines.
[0407] In this trial grapevines (Chardonnay variety) were either treated with the terpene product YGP-GET or left untreated (control) on 7 Feb. 2005. Although suppressed by previous organic treatments, the pre-trial severity of powdery mildew was at a level considered unacceptable commercially and was equivalent in the 6 active-treatment plots and 6 control plots. The crop stage was approximately E-L 33-34 (pre-veraison).
[0408] YGP-GET (4 mL/L) (liquid formulation) was sprayed onto 6 Chardonnay plots, which had been treated previously with milk. Six Chardonnay plots served as untreated controls, but they had been treated previously with oil/whey. The number of vines per plot was typically 7.
[0409] Details of the composition of the YGP-GET used in this protocol are given in Table 20.
TABLE-US-00020 TABLE 20 Formulation of Batch Used in Present Study Raw material mix details Weight in lbs % by Weight Geraniol 323.52 6.88 Eugenol 161.76 3.44 Thymol 323.52 6.88 Yeast particles 722.13 15.35 Xanthan 3.17 0.07 Polysorbate 3.17 0.07 Water 3166.62 67.32 TOTAL 4703.89 100.00
[0410] The severity of powdery mildew was assessed 3 days before terpene treatment and again 3 days post-treatment. In each plot, 20 grape bunches were selected at random (10 bunches per panel side), and disease severity was estimated as the percentage area of the bunches covered with active mildew colonies. No further assessment was possible because the grower subsequently sprayed the entire trial area with sulphur and a vegetable oil-based spraying adjuvant (Synertrol Horti Oil).
[0411] Number/Area of Plants to be Treated
[0412] Test product: YGP-GET (4 mL/L) to be applied to 6 Chardonnay plots (total of approximately 42 vines), which had been treated previously with milk.
[0413] Control: No treatment was applied to 6 Chardonnay plots (total of approximately 42 vines) to be used as controls, but they had been treated previously with oil/whey.
[0414] Cultivation Methods
[0415] Vitis vinifera (Chardonnay) vines in Block B2: vertical shoot positioning with arched canes.
[0416] Cultivation Arrangement
[0417] Spacing: Distance of 2.5 m between rows and 1.25 m between vines (within row), with 3,200 vines per hectare. Row orientation was north to south.
[0418] Canopy Density
[0419] The point-quadrat method was use
D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.