Preparation Of Substituted 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles
GRAMMENOS; Wassilios ; et al.
U.S. patent application number 16/634337 was filed with the patent office on 2020-07-23 for preparation of substituted 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles. The applicant listed for this patent is BASF SE. Invention is credited to Wassilios GRAMMENOS, Christopher KORADIN, Michael RACK, Violeta TERTERYAN-SEISER.
| Application Number | 20200231556 16/634337 |
| Document ID | / |
| Family ID | 59485232 |
| Filed Date | 2020-07-23 |








View All Diagrams
| United States Patent Application | 20200231556 |
| Kind Code | A1 |
| GRAMMENOS; Wassilios ; et al. | July 23, 2020 |
PREPARATION OF SUBSTITUTED 3-ARYL-5-TRIFLUOROMETHYL-1,2,4-OXADIAZOLES
Abstract
The present invention relates to a process for the preparation of 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles, which are useful as fungicidal compounds or as intermediates for the synthesis of fungicidal oxaciazole compounds, using trifluoroacetyl halides. ##STR00001##
| Inventors: | GRAMMENOS; Wassilios; (Ludwigshafen, DE) ; RACK; Michael; (Ludwigshafen, DE) ; TERTERYAN-SEISER; Violeta; (Ludwigshafen, DE) ; KORADIN; Christopher; (Ludwigshafen, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59485232 | ||||||||||
| Appl. No.: | 16/634337 | ||||||||||
| Filed: | July 20, 2018 | ||||||||||
| PCT Filed: | July 20, 2018 | ||||||||||
| PCT NO: | PCT/EP2018/069712 | ||||||||||
| 371 Date: | January 27, 2020 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 271/06 20130101 |
| International Class: | C07D 271/06 20060101 C07D271/06 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 28, 2017 | EP | 17183655.4 |
Claims
1.-15. (canceled)
16. A process for preparing compounds of formula I, ##STR00045## wherein A.sup.1 is CH; A.sup.2 is N CH; R.sup.A is hydrogen or fluorine; R is --COOH, --COOR.sup.1 or --C(.dbd.W)NR.sup.1R.sup.2; W is O or S; R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.3-C.sub.11-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from the group consisting of N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; the process comprising: reacting a compound of formula II, ##STR00046## wherein A.sup.1, A.sup.2, R.sup.A and R is as defined above, with a trifluoroacetyl halide of formula IIa, ##STR00047## wherein Hal is chlorine or fluorine.
17. The process of claim 16, wherein the process is conducted in the presence of an inert organic solvent.
18. The process of claim 16, wherein Hal in compound IIa is chlorine.
19. The process of claim 16, wherein the process is conducted in the presence of a base selected from trimethylamine, triethylamine, tributylamine, diisopropylethylamine, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine, sodium acetate, potassium acetate, sodium carbonate, potassium carbonate and mixtures thereof.
20. The process of claim 16, wherein the process is conducted at a pressure from 1 atm to 20 atm.
21. The process of claim 16, wherein the process is conducted at a temperature from 0.degree. C. to 100.degree. C.
22. The process of claim 16, the process comprising two steps: i) reacting a compound of formula V, ##STR00048## wherein R is as defined in claim 16, with hydroxylamine or a salt thereof; ii) reacting the product of step i), a compound of formula II, ##STR00049## wherein R is as defined above, with a trifluoroacetyl halide according to the process as defined in claim 16.
23. The process of claim 16, further comprising the step of reacting the compound of formula I to obtain a compound of formula III ##STR00050##
24. The process of 23, further comprising the step of reacting the compound of formula III with a compound of formula IV R.sup.1--NH--R.sup.2 IV, wherein R.sup.1 and R.sup.2 in the compound of formula IV is as defined in claim 16 to obtain a compound of formula Ic ##STR00051##
25. The process of claim 24, further comprising the step of reacting the compound of formula Ic to obtain a compound of formula Id ##STR00052##
26. The process of claim 24, wherein R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, cyclopropyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy and cyclopropyl; R.sup.2 is hydrogen, methyl or ethyl.
27. The process of claim 24, wherein R.sup.1 is methyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, 2-fluoro-phenyl, 4-fluoro-phenyl or 2-difluoromethoxy-phenyl; R.sup.2 is hydrogen.
Description
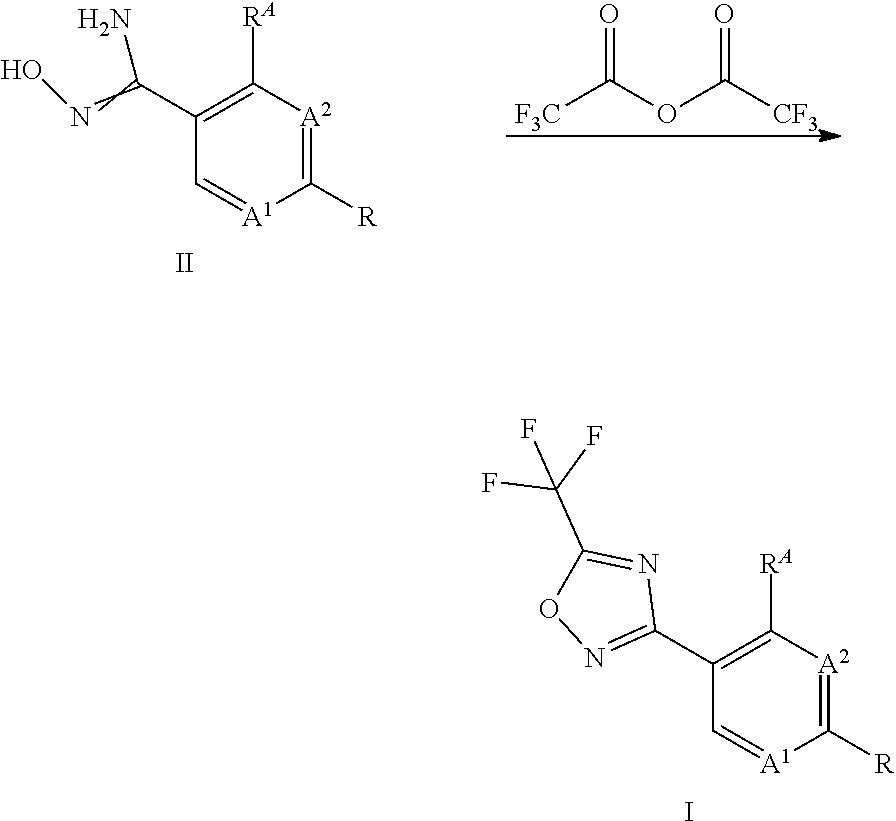
[0001] The present invention relates to a process for the preparation of substituted 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles (compounds I), which can be obtained through reaction of hydroxyamidine type compounds of formula II with trifluoroacetyl halides.
[0002] Certain 3-substituted 5-trifluoromethyl-1,2,4-oxadiazoles are known as fungicides i.a. from EP 276432 A2, EP 3165093 A1, EP 3165094 A1, EP 3167716 A1, WO 2015/185485, WO 2017/055469, WO 2017/055473, WO 2017/076739, WO 2017/076740, WO 2017/076742, WO 2017/076757, WO 2017/076935, WO 2017/081309, WO 2017/081310, WO 2017081311, WO 2017081312, WO 2017/085098, WO 2017/085100, WO2017/093019, WO2017/093348, WO 2017/085100, WO2017/093348, WO2017/102006, WO2017/103219, WO2017/103223, WO2017/109044 and WO2017/118689.
[0003] The formation of the 1,2,4-oxadiazole ring system in 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles, which are useful as fungicidally active compounds or as intermediates in the synthesis of fungicides, is described in the references cited above or in WO 2013/008162 by reacting amidoximes of type II with trifluoroacetic anhydride (TFAA) in an organic solvent.
##STR00002##
[0004] One disadvantage of this reaction lies in the formation of significant amounts of free trifluoroacetic acid (TFA) as a side product. Regulatory restrictions oftentimes require very low residue levels of TFA impurities in commercial agrochemical pesticides. For the sake of receiving a market authorization it is therefore crucial to control and minimize the amount of residual TFA during the process of producing a pesticide.
[0005] In contrast to other acids it is particularly difficult to remove free TFA from reaction mixtures, be it in solution or in mixtures that are in a solid state. TFA has a tendency to form cocrystals or clathrates with other solid reaction components. The removal of TFA is oftentimes cumbersome and requires elaborate additional purification steps (e. g. chromatography, distillation, crystallization, etc.). Furthermore, TFA is very corrosive and there is an interest to reduce the excess amounts of TFA furnished during or after the ring closing reaction, for example during workup. The reaction of compound II with TFAA produces one equivalent TFA. In the course of the oxadiazole ring closing/aromatisation reaction one equivalent of water is formed per equivalent of compound II. The water immediately reacts with another equivalent of TFAA, which in turn results in the formation of two additional equivalents TFA. As a result, at least two equivalents TFAA are necessary for a complete conversion of compounds II and in total at least three equivalents TFA are formed per equivalent of compounds II.
[0006] Reducing the number of equivalents of TFAA to the minimum required by theory, i.e. to two equivalents based on the amounts of compound II, does not always provide sufficient yield of the desired product. Typically, an excess of TFAA is used to achieve complete conversion of compounds II to compounds I. Hydrolyzation of excess TFAA under aqueous workup conditions produces even more TFA.
[0007] Thus, it was an object of the present invention to overcome the disadvantages of the known processes and to provide an improved and more economical and production plant friendly process.
[0008] It was now found that 3-aryl-5-trifluoromethyl-1,2,4-oxadiazoles can be obtained by the use of trifluoroacetyl halides, in particular trifluoroacetyl chloride (TFAC) or trifluoroacetyl fluoride (TFAF). The use of TFAC or TFAF instead of TFAA has several surprising advantages in relation to the preparation of the above-mentioned compounds.
[0009] The process of this invention does not require a large excess of trifluoroacetyl halides in order to achieve high yields of the desired oxadiazoles, which is desirable from both, an economical and an ecological point of view. As a consequence, the total organic carbon content in the production waste water is reduced compared to the prior art processes, which makes the process eco-friendly. Unlike the use of TFAA, the use of trifluoroacetic halides results in the formation of a much smaller amount of TFA, which makes such process more production plant friendly and simplifies the workup procedure. As a result the production costs are significantly lower. Furthermore, the process of this invention provides the desired oxadiazoles in high yields and with reduced amounts of undesired side products. The in-situ preparation of compounds of type II enables a process, which proceeds in a smooth and controlled manner, which is very safe, simple, economical, user-friendly and commercially viable.
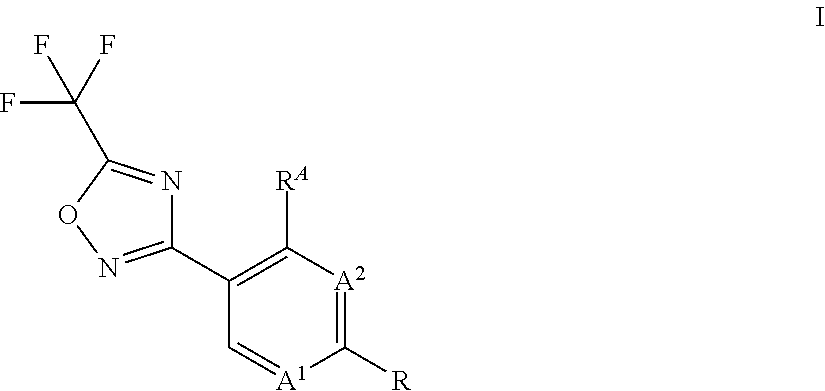
[0010] Accordingly, the present invention relates to a process for preparing compounds of formula I,
##STR00003##
wherein [0011] A.sup.1 is N or CH; [0012] A.sup.2 is N or CH; [0013] R.sup.A is hydrogen or halogen; [0014] R is methyl, trichloromethyl, ethyl, iso-propyl, OH, SH, CN, halogen, CH.sub.2F, CHF.sub.2, 2,2,2-trifluoroethyl, cyclopropyl, --COOH, --COOR.sup.1, --C(.dbd.W)NR.sup.1R.sup.2, --CR.sup.3R.sup.4NR.sup.1R.sup.2, --CR.sup.3R.sup.4OR.sup.1, --CR.sup.3(.dbd.NR.sup.1), --CR.sup.3(.dbd.O), --CR.sup.3R.sup.4COOH, --CR.sup.3R.sup.4COR.sup.1, --CR.sup.3R.sup.4C(.dbd.W)NR.sup.1R.sup.2, --OCR.sup.3R.sup.4COOH, --OCR.sup.3R.sup.4COR.sup.1, --OCR.sup.3R.sup.4C(.dbd.W)NR.sup.1R.sup.2, --CR.sup.3R.sup.4NR.sup.2C(.dbd.W)R.sup.1, --CR.sup.3R.sup.4S(.dbd.O).sub.2R.sup.1, or --CR.sup.3R.sup.4NR.sup.2S(.dbd.O).sub.2R.sup.1; [0015] W is O or S; [0016] R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxy, C.sub.3-C.sub.11-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.11-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl-C.sub.1-C.sub.4-alkyl, phenyl, pyridinyl, --C(.dbd.O)--(C.sub.1-C.sub.6-alkyl), --C(.dbd.O)--(C.sub.3-C.sub.11-cycloalkyl), --C(.dbd.O)--(C.sub.1-C.sub.6-alkoxy) and --N(R.sup.2a).sub.2; [0017] R.sup.2a is independently selected from the group consisting of hydrogen, OH, C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.3-C.sub.11-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.4-alkoxy-C.sub.1-C.sub.4-alkyl and C.sub.1-C.sub.6-alkylthio; and wherein any of the aliphatic or cyclic groups in R.sup.2 are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, hydroxy, oxo, cyano, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy and C.sub.3-C.sub.11-cycloalkyl; [0018] R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.3-C.sub.11-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.2-C.sub.6-alkenyloxyimino-C.sub.1-C.sub.4-alkyl, C.sub.2-C.sub.6-alkynyloxyimino-C.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.6-alkylamino, diC.sub.1-C.sub.6-alkylamino, C(.dbd.O)--(C.sub.1-C.sub.6-alkyl), C(.dbd.O)--(C.sub.1-C.sub.6-alkoxy), phenyl-C.sub.1-C.sub.4-alkyl, phenyl-C.sub.1-C.sub.4-alkenyl, phenyl-C.sub.1-C.sub.4-alkynyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl, naphthyl or a 3- to 10-membered saturated, partially unsaturated or aromatic mono- or bicyclic heterocycle, wherein the ring member atoms of said mono- or bicyclic heterocycle include besides carbon atoms further 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms with the provision that the heterocycle cannot contain 2 contiguous atoms selected from O and S; and wherein the heteroaryl group in the group heteroaryl-C.sub.1-C.sub.4-alkyl is a 5- or 6-membered aromatic heterocycle, wherein the ring member atoms of the heterocyclic ring include besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms with the provision that the heterocycle cannot contain 2 contiguous atoms selected from O and S; and wherein any of the above-mentioned aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different groups R.sup.1a; or [0019] R.sup.1 and R.sup.2, together with the nitrogen atom to which R.sup.2 is attached, and together with interjacent group --C(.dbd.W)--, if present, which is located between said nitrogen atom and the group R.sup.1, form a saturated or partially unsaturated mono- or bicyclic 3- to 10-membered heterocycle, wherein the heterocycle includes beside one nitrogen atom and one or more carbon atoms no further heteroatoms or 1, 2 or 3 further heteroatoms independently selected from N, O and S as ring member atoms with the provision that the heterocycle cannot contain 2 contiguous atoms selected from O and S; and wherein the heterocycle is unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different groups R.sup.1a; or [0020] if R.sup.2 is --N(R.sup.2a).sub.2, R.sup.1 and one of the two groups R.sup.2a, together with the nitrogen atom to which R.sup.2a is attached, and together with interjacent groups, which are located between said nitrogen atom and the group R.sup.1, form a saturated or partially unsaturated mono- or bicyclic 3- to 10-membered heterocycle, wherein the heterocycle includes beside two nitrogen atoms and one or more carbon atoms no further heteroatoms or 1, 2 or 3 further heteroatoms independently selected from N, O and S as ring member atoms with the provision that the heterocycle cannot contain 2 contiguous atoms selected from O and S; and wherein the heterocycle is unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different groups R.sup.1a; [0021] R.sup.1a is halogen, oxo, cyano, NO.sub.2, OH, SH, NH.sub.2, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-haloalkyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.6-haloalkoxy, C.sub.1-C.sub.6-alkylthio, C.sub.1-C.sub.6-haloalkylthio, C.sub.3-C.sub.8-cycloalkyl, --NHSO.sub.2--C.sub.1-C.sub.4-alkyl, (C.dbd.O)--C.sub.1-C.sub.4-alkyl, C(.dbd.O)--C.sub.1-C.sub.4-alkoxy, C.sub.1-C.sub.6-alkylsulfonyl, hydroxyC.sub.1-C.sub.4-alkyl, C(.dbd.O)--NH.sub.2, C(.dbd.O)--NH(C.sub.1-C.sub.4-alkyl), C.sub.1-C.sub.4-alkylthio-C.sub.1-C.sub.4-alkyl, aminoC.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.4-alkylamino-C.sub.1-C.sub.4-alkyl, diC.sub.1-C.sub.4-alkylamino-C.sub.1-C.sub.4-alkyl, aminocarbonyl-C.sub.1-C.sub.4-alkyl or C.sub.1-C.sub.4-alkoxy-C.sub.1-C.sub.4-alkyl; [0022] R.sup.3, R.sup.4 independently of each other are selected from the group consisting of hydrogen, halogen, cyano, C.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.4-alkenyl, C.sub.1-C.sub.4-alkynyl, C.sub.1-C.sub.4-haloalkyl and C.sub.1-C.sub.4-alkoxy; or [0023] R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a saturated 3- to 7-membered carbocycle or a saturated 3- to 6-membered heterocycle; wherein the saturated heterocycle includes beside carbon atoms 1, 2 or 3 heteroatoms independently selected from the group consisting of N, O and S as ring member atoms with the provision that the heterocycle cannot contain 2 contiguous atoms selected from O and S; and wherein said N ring member atom is substituted with the group R.sup.N; wherein [0024] R.sup.N is hydrogen, C.sub.1-C.sub.6-alkyl or halogen; [0025] and wherein said S ring member atom is unsubstituted or substituted with 1 or 2 oxo radicals; and wherein one or two of the ring member --CH.sub.2-- groups of said saturated carbocycle or of said saturated heterocycle may be replaced by one or two groups independently selected from --C(.dbd.O)-- and --C(.dbd.S)--; and wherein the carbon ring member atoms of the saturated carbocycle or of the saturated heterocycle are unsubstituted or substituted with a total number of 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy and C.sub.3-C.sub.8-cycloalkyl; the process comprising: reacting a compound of formula II,
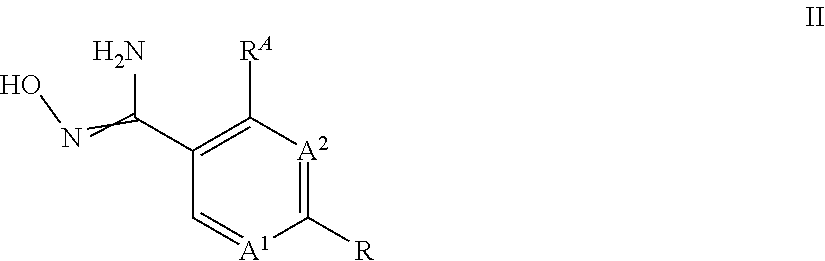
##STR00004##
[0025] wherein A.sup.1, A.sup.2, R.sup.A and R is as defined above, with a compound of formula IIa,
##STR00005##
wherein Hal is chlorine or fluorine.
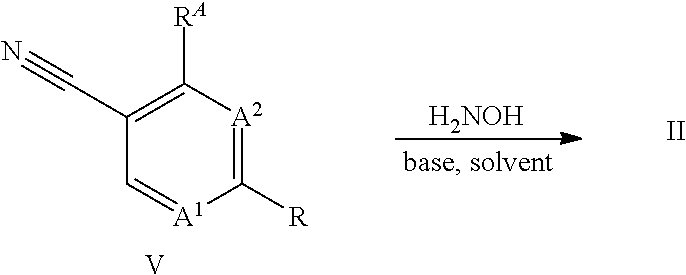
[0026] The amidoxime compounds II can be obtained from cyano compounds V by treatment with hydroxylamine or a salt thereof, for example the hydrochloride salt, in the presence of a base, preferably triethylamine, in a suitable solvent, such as methanol, at a temperature between 0.degree. C. and 100.degree. C. For related examples see Kitamura, S. et al Chemi. Pharm. Bull. (2001), 49, 268 WO 2013/066838 or WO 2017/055473:
##STR00006##
[0027] Preferably, the formation of the amidoxime II and its transformation to compounds I takes place in a two-step one-pot reaction without any workup of the amidoxime II.
[0028] Compounds of formula III are either commercially available or may be prepared using standard procedures known to a person skilled in the art from readily available starting materials. Such procedures are described in, for example, WO 2013/008162, EP 276432 A2, EP 3165093 A1, EP 3165094 A1, EP 3167716 A1, WO 2015/185485, WO 2017/055469, WO 2017/055473, WO 2017/076739, WO 2017/076740, WO 2017/076742, WO 2017/076757, WO 2017/076935, WO 2017/081309, WO 2017/081310, WO 2017081311, WO 2017081312, WO 2017/085098, WO 2017/085100, WO2017/093019, WO2017/093348, WO2017/102006, WO2017/103219, WO2017/103223, WO2017/109044 and WO2017/118689.
[0029] According to a preferred embodiment, compound IIa is trifluoacetyl chloride (TFAC).
[0030] In a preferred embodiment, the transformation of a compound of formula II with compounds of formula IIa to produce compounds of formula I is carried out in the presence of an inert organic solvent, wherein the inert organic solvent is not identical with a compound I, II, IIa, III or a base as defined herein.
[0031] In a preferred embodiment, said process is carried out in the presence of a base, wherein the base is not identical with a compound I, II, IIa, III or an inert organic solvent as defined herein.
[0032] In another preferred embodiment, said process is carried out in the presence of an inert organic solvent, wherein the inert organic solvent is not identical with a compound I, II, IIa, III or a base as defined herein; and in the absence of a base, wherein the base is not identical with a compound I, II, IIa, III or an inert organic solvent as defined herein.
[0033] In another preferred embodiment, said process is carried out in the presence of a base, wherein the base is not identical with a compound I, II, IIa, III or an inert organic solvent as defined herein; and in the absence of an inert solvent, wherein the inert organic solvent is not identical with a compound I, II, IIa, III or a base as defined herein.
[0034] In another preferred embodiment, said process is carried out in the presence of a base, wherein the base is not identical with a compound I, II, IIa, III or a solvent as defined herein; and in the presence of an inert solvent, wherein the inert organic solvent is not identical with a compound I, II, IIa, III or a base as defined herein.
[0035] By "inert organic solvent" is meant an organic solvent which, under the reaction conditions of the process of this invention, does not enter into any appreciable reaction with either the reactants or the products.
[0036] In one embodiment, the inert organic solvent is selected from non-halogenated inert organic solvents; preferably from non-halogenated aliphatic hydrocarbons, non-halogenated cycloaliphatic hydrocarbons, non-halogenated aromatic hydrocarbons, halogenated aliphatic hydrocarbons, halogenated aromatic hydrocarbons, amides, ethers, esters, ketones, nitriles and any combination thereof.
[0037] Examples of suitable non-halogenated aliphatic hydrocarbons include pentane, hexane, heptane, and the like. Preference is given to saturated aliphatic hydrocarbons having from 5 to 10 carbon atoms.
[0038] Examples of suitable non-halogenated cycloaliphatic hydrocarbons include cyclopentane, cyclohexane, cycloheptane, and the like. Preference is given to non-halogenated saturated cycloaliphatic hydrocarbons having from 5 to 10 carbon atoms. Cyclohexane is particularly preferred.
[0039] Examples of suitable a non-halogenated aromatic hydrocarbons include toluene, o-xylene, m-xylene, p-xylene, ethylbenzene, 2-propylbenzene (cumene), 2-isopropyltoluene (o-cymol), 3-isopropyltoluene (m-cymol), 4-isopropyltoluene (p-cymol), 1,3,5-trimethylbenzene (mesitylene), and the like. Preference is given to toluene, o-xylene, m-xylene, p-xylene, ethylbenzene, 1,3,5-trimethylbenzene (mesitylene), and any combination thereof. Especially preferred among the non-halogenated aromatic hydrocarbons are toluene, o-xylene, m-xylene, p-xylene, and any combination thereof, with toluene being the most preferred.
[0040] Examples of suitable halogenated aliphatic hydrocarbons include dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, 1,1,1,2-tetrachloroethane, 1,1,2,2-tetrachloroethane, 1,1-dichloroethylene, 1,2-dichloroethylene, and the like. Preference is given to dichloromethane and 1,2-dichloroethane and any combination thereof.
[0041] Examples of suitable halogenated aromatic hydrocarbons include chlorobenzene, bromobenzene, o-dichlorobenzene, m-dichlorobenzene, .alpha.,.alpha.,.alpha.-trifluorotoluene (benzotrifluoride) and the like and any combination thereof.
[0042] Examples of suitable amides include N,N-dimethylformamide, N,N-dimethylacetamide, N,N-diethylacetamide, N-methyl-pyrrolidone, and the like and any combination thereof.
[0043] Examples of suitable ethers include cyclic and acyclic ethers such as diethyl ether, diisopropyl ether, n-butyl methyl ether, isobutyl methyl ether, sec-butyl methyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, tetrahydrofuran, 1,4-dioxane, and the like and any combination thereof.
[0044] Examples of suitable esters include ethyl acetate, n-propylacetate, isopropyl acetate, tert-butyl acetate, and the like and any combination thereof.
[0045] Examples of suitable ketones include acetone, methyl ethyl ketone, methyl isopropyl ketone, methyl isobutyl ketone, cyclopropyl methyl ketone and the like, and any combination thereof. Examples of suitable nitriles include acetonitrile, benzonitrile, and the like and any combination thereof.
[0046] In a preferred embodiment, the inert organic solvent is selected from non-halogenated aliphatic hydrocarbons, non-halogenated cycloaliphatic hydrocarbons, non-halogenated aromatic hydrocarbons, halogenated aliphatic hydrocarbons and any combination thereof.
[0047] In a more preferred embodiment, the inert organic solvent is selected from heptane, cyclohexane, cycloheptane, toluene, o-xylene, m-xylene, p-xylene, ethylbenzene, 1,3,5-trimethylbenzene (mesitylene), chlorobenzene, 1,2-dichloroethane, dichloromethane, tetrahydrofuran, dioxane, ethyl acetate, methyl ethyl ketone and benzotrifluoride and any combination thereof.
[0048] In an even more preferred embodiment, the inert organic solvent is selected from heptane, cyclohexane, toluene, dichloromethane and any combination thereof.
[0049] Still more preferably, the inert organic solvent is selected from heptane, cyclohexane, toluene and any combination thereof.
[0050] Particularly preferred inert organic solvents are non-halogenated aromatic hydrocarbons, especially non-halogenated alkylbenzenes which are mono-, di-, or tri-alkylsubstituted with each alkyl group containing 1 to 3 carbon atoms, and in particular those selected from the group consisting of toluene, o-xylene, m-xylene, p-xylene and any combination thereof. Most preferably, the inert organic solvent is toluene.
[0051] The volume ratio of the inert organic solvent to the hydroxyamidine II is generally from 0.01:1 to 20:1, preferably from 0.1:1 to 15:1, more preferably from 0.5:1 to 10:1, and most preferably from 1:1 to 5:1.
[0052] In a preferred embodiment of the present invention, the transformation of a compound of formula II with compounds of formula IIa to produce compounds of formula I is carried out in the presence of an organic base.
[0053] In one embodiment the base is selected from organic bases such as, for example: tertiary amines, pyridine, substituted pyridines, bicyclic amines and any mixture thereof. Preference is given to tertiary amines, pyridine, substituted pyridines and any mixture thereof. Particular preference is given to pyridine, substituted pyridines and any mixture thereof. Pyridine is especially preferred.
[0054] Examples of suitable tertiary amines are tri-(C.sub.1-C.sub.6)-alkylamines such as trimethylamine, triethylamine, tributylamine and diisopropylethylamine; di-(C.sub.1-C.sub.6)-alkyl-phenylamines such as N,N-dimethylaniline and N,N-diethylaniline; N-methyl imidazole, N,N-dimethylaminopyridine and the like.
[0055] Examples of suitable substituted pyridines are collidine, lutidines, 2-picoline, 3-picoline, 4-picoline, N,N-dimethyl-4-aminopyridine, 5-ethyl-2-methyl-pyridine and the like.
[0056] Examples of suitable bicyclic amines are 1,8-diazabicyclo[5.4.0]undec-7-en, 1,5-diazabicyclo[4.3.0]non-5-ene, 1,4-diazabicyclo[2.2.2]octane and the like.
[0057] In one embodiment the base is an organic base, which is selected from trimethylamine, triethylamine, tributylamine, diisopropylethylamine, N,N-dimethylaniline, N,N-diethylaniline, N-methyl imidazole, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, N,N-dimethyl-4-aminopyridine, 5-ethyl-2-methyl-pyridine,1,8-diazabicyclo[5.4.0]undec-7-en, 1,5-diazabicyclo[4.3.0]non-5-ene, 1,4-diazabicyclo[2.2.2]octane and mixtures thereof.
[0058] In another embodiment the base is an organic base, which is selected from trimethylamine, triethylamine, diisopropylethylamine, N,N-dimethylaniline, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine, N,N-dimethylaminopyridine and mixtures thereof.
[0059] More preferably the base is an organic base, which is selected from trimethylamine, triethylamine, diisopropylethylamine, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine and mixtures thereof.
[0060] In another embodiment, the base is selected from alkali metal and alkaline earth metal phosphates; alkali metal and alkaline earth metal formats; alkali metal and alkaline earth metal acetates; alkali metal and alkaline earth metal carbonates; alkali metal and alkaline earth metal citrates; alkali metal and alkaline earth metal sulfates and any combination thereof, even more preferably selected from alkali metal and alkaline earth metal phosphates; alkali metal and alkaline earth metal formats; alkali metal and alkaline earth metal acetates; alkali metal and alkaline earth metal carbonates; alkali metal and alkaline earth metal citrates and any combination thereof, in particular selected from alkali metal and alkaline earth metal phosphates; alkali metal and alkaline earth metal acetates; particularly sodium acetate, potassium acetate; alkali metal and alkaline earth metal carbonates and any combination thereof, most preferably selected from alkali metal and alkaline earth metal acetates; particularly preferred are sodium carbonate and potassium carbonate.
[0061] In another embodiment the base is selected from trimethylamine, triethylamine, tributylamine, diisopropylethylamine, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine, sodium acetate, potassium acetate, sodium carbonate and potassium carbonate and mixtures thereof.
[0062] The molar ratio of the base to hydroxyamidine II is generally from 3:1 to 0.4:1 or from 1:1 to 0.2:1, preferably from 0.02:1 to 0.3:1, more preferably from 0.02:1 to 0.25:1, most preferably from 0.02:1 to 0.2:1.
[0063] The process of the present invention can be carried out under atmospheric pressure or under elevated or reduced pressure. Typically, the atmospheric and elevated pressure is employed. In a preferred embodiment the process of the present invention can be carried out at pressure ranges typically from 0.8 atmospheres (atm) to 80 atm, preferably form 1.0 atm to 20 atm, in particular from 1.0 to 7 atm.
[0064] The temperature used in the process of the present invention can vary widely and is preferably from -30 to 150.degree. C., more preferably from -10 to 120.degree. C. and even more preferably from 0 to 100.degree. C., 20 to 80.degree. C. or 40 to 70.degree. C.
[0065] Typical reaction times are in the range of from 1 to 20 hours, preferably from 2 to 15 hours and more preferably from 3 to 10 hours.
[0066] In one preferred embodiment, the transformation of a compound of formula II with compounds of formula IIa to produce compounds of formula I is carried out in the presence of an inert organic solvent, wherein the inert organic solvent is selected from trimethylamine, triethylamine, tributylamine, diisopropylethylamine, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine, sodium acetate, potassium acetate, sodium carbonate and potassium carbonate and mixtures thereof; and in the absence of a base, wherein the base is not identical with a compound I, II, IIa, III or an inert organic solvent as defined herein; and at a pressure from 1 to 20 atm; and at a temperature from 0 to 100.degree. C.
[0067] In another preferred embodiment, said reaction is carried out in the presence of an inert organic solvent, wherein the inert organic solvent is selected from trimethylamine, triethylamine, tributylamine, diisopropylethylamine, pyridine, 2,4,6-collidine, 2,6-lutidine, 2-picoline, 3-picoline, 4-picoline, 5-ethyl-2-methyl-pyridine, sodium acetate, potassium acetate, sodium carbonate and potassium carbonate and mixtures thereof; and in the absence of a base, wherein the base is not identical with a compound I, II, IIa, III or an inert organic solvent as defined herein; and at a pressure from 1 to 7 atm; and at a temperature from 20 to 80.degree. C.
[0068] The process of the present invention may optionally be carried out in the presence of at least one phase-transfer catalyst.
[0069] Phase transfer catalysts suitable for use in the process of this invention are those well known in the art such as, for example, quaternary ammonium salts. Examples of suitable phase-transfer catalysts are trimethyl(phenyl) ammonium chloride, bromide, iodide or hydroxide and tetra-n-C.sub.1-C.sub.12-alkyl-ammonium chlorides, bromides, iodides or hydroxides, preferably tetra-n-C.sub.1-C.sub.8-alkyl-ammonium chlorides, bromides, iodides or hydroxides, e.g. tetramethylammonium chloride, bromide, iodide or hydroxide, tetraethylammonium chloride, bromide, iodide or hydroxide, tetra-n-propylammonium chloride, bromide, iodide or hydroxide, tetra-n-butylammonium chloride, bromide, iodide or hydroxide, tetra-n-pentylammonium chloride, bromide, iodide or hydroxide, tetra-n-hexylammonium chloride, bromide, iodide or hydroxide, tetra-n-heptylammonium chloride, bromide, iodide or hydroxide, tetra-n-octylammonium chloride, bromide, iodide or hydroxide, methyl-tri-n-butylammonium chloride, bromide, iodide or hydroxide, ethyl-trimethylammonium chloride, bromide, iodide or hydroxide, n-propyl-trimethyl ammonium chloride, bromide, iodide or hydroxide, methyl-triethyl ammonium chloride, bromide, iodide or hydroxide and n-butyl-triethylammonium chloride, bromide, iodide or hydroxide. Of these, the use of tetra-n-C.sub.1-C.sub.4-alkyl-ammonium chlorides, bromides, iodides or hydroxides is preferred, in particular tetra-n-butylammonium chloride, bromide, iodide or hydroxide and methyl-tri-n-butylammonium chloride, bromide, iodide or hydroxide.
[0070] The phase-transfer catalysts, which are usually solid in pure form, can be used as such or, preferably, in dissolved form. An effective amount of the phase-transfer catalyst may range from 0.001 to 0.5 molar equivalents, preferably 0.001 to 0.2 molar equivalents relative to compound II.
[0071] According to one embodiment of said process, a mixture of the E/Z-isomers of compounds of formula II is used, more preferably said mixture comprises an E/Z-isomer ratio of from 0.1:1 to 10:1, even more preferably of from 0.5:1 to 2:1.
[0072] According to another embodiment, the E-isomer of compounds of formula II is used.
[0073] According to a further embodiment, the Z-isomer of compounds of formula II is used.
[0074] The reaction mixture obtained is worked up and the compound I can be isolated in a customary manner, e. g. by an aqueous, extractive workup, in particular extraction with a basic or neutral aqueous medium, and by removing the solvent, e. g. under reduced pressure, or by a combination of these measures. Further purification can be effected, for example, by crystallization, thin-film-evaporation, rectification, sublimation, distillation or by chromatography.
[0075] In a further embodiment, a compound of formula I, in which R is methyl, is converted into valuable chemical products or intermediates. Accordingly, compounds of formula I, wherein R is methyl, can be further chlorinated to obtain a compound of formula Ib
##STR00007##
wherein the variables A.sup.1, A.sup.2 and R.sup.A in compounds I and Ib are as defined or preferably defined herein.
[0076] The chlorination of the methyl group R of compounds of formula I can be achieved using suitable chlorinating agents, for example molecular chlorine, N-chlorosuccinimide, trichloroisocyanuric acid, sulfuryl chloride or phosphorus pentachloride. The chlorination is conducted at temperatures between 0.degree. C. and 200.degree. C., preferably between 60.degree. C. and 150.degree. C., under irradiation or in the presence of radical starters, for example azobis(isobutyronitril) or dibenzoyl peroxide. For reference, see J. Am. Chem. Soc. 1951, 73, 455.
[0077] In one embodiment, the chlorination is carried out in the presence of at least one inert organic solvent, or mixtures of such solvents. The term "inert organic solvent" means an organic solvent, which does not enter into any appreciable reaction with either the reactants or the products under the reaction conditions of the process of this invention. The inert organic solvent used in the process is preferably selected from halogenated aliphatic hydrocarbons and halogenated aromatic hydrocarbons, such as dichloromethane, tetrachloromethane, dichloroethane, chlorobenzene, homologues of dichlorobenzenes or 1,2,4-trichlorobenzene.
[0078] In a particularly preferred embodiment the chlorination step is conducted in substance, for example with the liquid reaction product, which is directly obtained after the reaction of compounds II and IIa and in the absence of a further solvent, as described above.
[0079] The chlorination can be performed in the presence or absence of TFA impurities originating from the previous reaction. TFA can be removed during the chlorination process via distillation or separately by distillation before carrying out the chlorination step. After completion of the reaction the reaction mixture is worked up in the usual manner or it can be used directly in the next step.
[0080] In a further preferred embodiment, the compound of formula Ib is hydrolyzed to obtain a compound of formula III
##STR00008##
wherein the variables A.sup.1, A.sup.2 and R.sup.A in compounds Ib and III are as defined or preferably defined herein.
[0081] In one embodiment this transformation is carried out in the presence of catalytic amounts of a lewis acid and water to obtain a compound of formula III, as described in WO 2007/063028 A2 on pages 42-43. Preferably, the lewis acid is a metal salt, for example aluminum(III) chloride or iron(III) chloride, particularly iron(III) chloride. The lewis acid is used in sub-stoichiometric or catalytic amounts, for example 0.001 to 0.5 molar equivalents, preferably 0.002 to 0.2 molar equivalents, more preferably 0.005 to 0.1 molar equivalents, based on the amount of the compound of formula Ib.
[0082] In one embodiment, the hydrolysis step is carried out in the presence of at least one inert organic solvent, or mixtures of such solvents. The inert organic solvent used in the process of this invention is preferably selected from non-halogenated aliphatic hydrocarbons, non-halogenated cycloaliphatic hydrocarbons, halogenated aliphatic hydrocarbons, halogenated aromatic hydrocarbons, amides, ethers, esters, ketones, nitriles.
[0083] In a preferred embodiment, the hydrolysis step is carried out in the absence of a solvent, i.e. in substance. Under these conditions the trichloromethyl compound Ib or the crude material comprising Ib, which was obtained from the previous reaction step, is heated to a temperature, where such material is a molten mass.
[0084] The amount of water in the hydrolysis step is between 0.8 to 1.5 molar equivalents, preferably between 0.95 to 1.05 molar equivalents, based on the amount of the compound Ib. The reaction is carried out at temperatures between 20.degree. C. and 200.degree. C., preferably between 80.degree. C. and 130.degree. C. After completion of the reaction the reaction mixture is worked up in the usual manner or it can be used directly in the next step.
[0085] In an especially preferred embodiment, the compound of formula III is reacted with an amine of formula IV to obtain a compound of formula Ic,
##STR00009##
wherein the variables A.sup.1, A.sup.2, R.sup.A, R.sup.1 and R.sup.2 in compounds of formulae III and IV are as defined or preferably defined herein.
[0086] A skilled person will recognize that oxadiazole compounds of type Ic can be accessed by treating benzoic acid chloride of formula III with an amine of formula IV. The reaction is preferably carried out in a suitable inert organic solvent, such as non-halogenated aliphatic hydrocarbons, non-halogenated cycloaliphatic hydrocarbons, halogenated aliphatic hydrocarbons, halogenated aromatic hydrocarbons, amides, ethers, esters, ketones, nitriles; for example, N,N-dimethylformamide, dichloromethane or tetrahydrofuran; preferably at a temperature between -20.degree. C. and 200.degree. C., preferably between 0.degree. C. and 80.degree. C., and optionally in the presence of a base such as pyridine, triethylamine or N,N-diisopropylethylamine, or under conditions described in the literature for an amide coupling. For examples, see Valeur, E.; Bradley, M. Chem. Soc. Rev. 2009, 38, 606 and Chinchilla, R., Najera, C. Chem. Soc. Rev. 2011, 40, 5084. After completion of the reaction the reaction mixture is worked up in the usual manner.
[0087] In another especially preferred embodiment, the compound of formula Ic is used to obtain a compound of formula Id
##STR00010##
wherein the variables A.sup.1, A.sup.2, R.sup.A, R.sup.1 and R.sup.2 in compounds of formulae Ic and Id are as defined or preferably defined herein.
[0088] Compounds of formula Ib can be prepared from compounds of formula Ic through treatment with Lawesson's reagent or phosphorus pentasulfide in an inert organic solvent, such as non-halogenated aliphatic hydrocarbons, non-halogenated cycloaliphatic hydrocarbons, halogenated aliphatic hydrocarbons, halogenated aromatic hydrocarbons, amides, ethers, esters, ketones, nitriles; for example toluene, tetrahydrofuran, dioxane or ethyl acetate; at a temperature between 0.degree. C. and 130.degree. C., preferentially between 60.degree. C. and 80.degree. C. For examples, see Eur. J. Med. Chem. 2011, 46(9), 3917-3925; Synthesis 2003, 13, 1929-1958; WO 2006/0123242; WO 2010/086820; WO 2014/0151863. After completion of the reaction the reaction mixture is worked up in the usual manner.
[0089] The terms "compounds I", "compounds II", "compounds IIa" and "compounds III" refer to compounds of formulae I, II, IIa and III, respectively. In the definitions of the variables given above, collective terms are used which are generally representative for the substituents in question.
[0090] The term "C.sub.n-C.sub.m" indicates the number of carbon atoms possible in each case in the substituent or substituent moiety in question.
[0091] The term "halogen" refers to fluorine, chlorine, bromine and iodine.
[0092] The term "oxo" refers to an oxygen atom .dbd.O, which is bound to a carbon atom or sulfur atom, thus forming, for example, a ketonyl --C(.dbd.O)-- or sulfinyl --S(.dbd.O)-- group.
[0093] The term "formyl" refers to a group C(.dbd.O)H.
[0094] The term "C.sub.1-C.sub.6-alkyl" refers to a straight-chained or branched saturated hydrocarbon group having 1 to 6 carbon atoms, for example methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2-methylpropyl, and 1,1-dimethylethyl.
[0095] The term "C.sub.2-C.sub.6-alkenyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and a double bond in any position, such as ethenyl, 1-propenyl, 2-propenyl (allyl), 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl.
[0096] The term "C.sub.2-C.sub.6-alkynyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and containing at least one triple bond, such as ethynyl, 1-propynyl, 2-propynyl (propargyl), 1-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl.
[0097] The term "C.sub.1-C.sub.6-haloalkyl" refers to a straight-chained or branched alkyl group having 1 to 6 carbon atoms (as defined above), wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above, for example chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1-fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro-2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl and pentafluoroethyl, 2-fluoropropyl, 3-fluoropropyl, 2,2-difluoropropyl, 2,3-difluoropropyl, 2-chloropropyl, 3-chloropropyl, 2,3-dichloropropyl, 2-bromopropyl, 3-bromopropyl, 3,3,3-trifluoropropyl, 3,3,3-trichloropropyl, CH.sub.2--C.sub.2F.sub.5, CF.sub.2--C.sub.2F.sub.5, CF(CF.sub.3).sub.2, 1-(fluoromethyl)-2-fluoroethyl, 1-(chloromethyl)-2-chloroethyl, 1-(bromomethyl)-2-bromoethyl, 4-fluorobutyl, 4-chlorobutyl, 4-bromobutyl or nonafluorobutyl.
[0098] The term "C.sub.1-C.sub.6-alkoxy" refers to a straight-chain or branched alkyl group having 1 to 6 carbon atoms (as defined above) which is bonded via an oxygen, at any position in the alkyl group, for example methoxy, ethoxy, n-propoxy, 1-methylethoxy, butoxy, 1-methylpropoxy, 2-methylpropoxy or 1,1-dimethylethoxy.
[0099] The term "C.sub.1-C.sub.6-haloalkoxy" refers to a C.sub.1-C.sub.6-alkoxy group as defined above, wherein some or all of the hydrogen atoms may be replaced by halogen atoms as mentioned above, for example, OCH.sub.2F, OCHF.sub.2, OCF.sub.3, OCH.sub.2Cl, OCHCl.sub.2, OCCl.sub.3, chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2-bromoethoxy, 2-iodoethoxy, 2,2-difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-chloro-2,2-difluoroethoxy, 2,2-dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, OC.sub.2F.sub.5, 2-fluoropropoxy, 3-fluoropropoxy, 2,2-difluoropropoxy, 2,3-difluoropropoxy, 2-chloropropoxy, 3-chloropropoxy, 2,3-dichloropropoxy, 2-bromopropoxy, 3-bromopropoxy, 3,3,3-trifluoropropoxy, 3,3,3-trichloropropoxy, OCH.sub.2--C.sub.2F.sub.5, OCF.sub.2--C.sub.2F.sub.5, 1-(CH.sub.2F)-2-fluoroethoxy, 1-(CH.sub.2Cl)-2-chloroethoxy, 1-(CH.sub.2Br)-2-bromoethoxy, 4-fluorobutoxy, 4-chlorobutoxy, 4-bromobutoxy or nonafluorobutoxy.
[0100] The terms "phenyl-C.sub.1-C.sub.4-alkyl or heteroaryl-C.sub.1-C.sub.4-alkyl" refer to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a phenyl or hetereoaryl radical respectively.
[0101] The term "C.sub.1-C.sub.4-alkoxy-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a C.sub.1-C.sub.4-alkoxy group (as defined above). Likewise, the term "C.sub.1-C.sub.4-alkylthio-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a C.sub.1-C.sub.4-alkylthio group.
[0102] The term "C.sub.1-C.sub.6-alkylthio" as used herein refers to straight-chain or branched alkyl groups having 1 to 6 carbon atoms (as defined above) bonded via a sulfur atom. Accordingly, the term "C.sub.1-C.sub.6-haloalkylthio" as used herein refers to straight-chain or branched haloalkyl group having 1 to 6 carbon atoms (as defined above) bonded through a sulfur atom, at any position in the haloalkyl group.
[0103] The term "C.sub.1-C.sub.4-alkoxyimino" refers to a divalent imino radical (C.sub.1-C.sub.4-alkyl-O--N.dbd.) carrying one C.sub.1-C.sub.4-alkoxy group as substituent, e.g. methylimino, ethylimino, propylimino, 1-methylethyl-imino, butylimino, 1-methylpropylimino, 2-methylpropylimino, 1,1-dimethylethylimino and the like.
[0104] The term "C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein two hydrogen atoms of one carbon atom of the alkyl radical are replaced by a divalent C.sub.1-C.sub.6-alkoxyimino radical (C.sub.1-C.sub.6-alkyl-O--N.dbd.) as defined above.
[0105] The term "C.sub.2-C.sub.6-alkenyloxyimino-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein two hydrogen atoms of one carbon atom of the alkyl radical are replaced by a divalent C.sub.2-C.sub.6-alkenyloxyimino radical (C.sub.2-C.sub.6-alkenyl-O--N.dbd.).
[0106] The term "C.sub.2-C.sub.6-alkynyloxyimino-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein two hydrogen atoms of one carbon atom of the alkyl radical are replaced by a divalent C.sub.2-C.sub.6-alkynyloxyimino radical (C.sub.2-C.sub.6-alkynyl-O--N.dbd.).
[0107] The term "hydroxyC.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein one hydrogen atom of the alkyl radical is replaced by a OH group.
[0108] The term "aminoC.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein one hydrogen atom of the alkyl radical is replaced by a NH.sub.2 group.
[0109] The term "C.sub.1-C.sub.6-alkylamino" refers to an amino group, which is substituted with one residue independently selected from the group that is defined by the term C.sub.1-C.sub.6-alkyl. Likewise the term "diC.sub.1-C.sub.6-alkylamino" refers to an amino group, which is substituted with two residues independently selected from the group that is defined by the term C.sub.1-C.sub.6-alkyl.
[0110] The term "C.sub.1-C.sub.4-alkylamino-C.sub.1-C.sub.4-alkyl" refers to refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a C.sub.1-C.sub.4-alkyl-NH-group which is bound through the nitrogen. Likewise the term "diC.sub.1-C.sub.4-alkylamino-C.sub.1-C.sub.4-alkyl" refers to refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a (C.sub.1-C.sub.4-alkyl).sub.2N-group which is bound through the nitrogen.
[0111] The term "aminocarbonyl-C.sub.1-C.sub.4-alkyl" refers to alkyl having 1 to 4 carbon atoms, wherein one hydrogen atom of the alkyl radical is replaced by a --(C.dbd.O)--NH.sub.2 group.
[0112] The term "C.sub.2-C.sub.6-alkenyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and a double bond in any position, such as ethenyl, 1-propenyl, 2-propenyl (allyl), 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl.
[0113] The term "C.sub.2-C.sub.6-alkynyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and containing at least one triple bond, such as ethynyl, 1-propynyl, 2-propynyl (propargyl), 1-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl.
[0114] The term "C.sub.3-C.sub.11-cycloalkyl" refers to a monocyclic, bicyclic or tricyclic saturated univalent hydrocarbon radical having 3 to 11 carbon ring members that is connected through one of the ring carbon atoms by substitution of one hydrogen atom, such as cyclopropyl (C.sub.3H.sub.5), cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, bicyclo[1.1.0]butyl, bicyclo[2.1.0]pentyl, bicyclo[1.1.1]pentyl, bicyclo[3.1.0]hexyl, bicyclo[2.1.1]hexyl, norcaranyl (bicyclo[4.1.0]heptyl) and norbornyl (bicyclo[2.2.1]heptyl). Further examples of bicyclic or tricyclic cycloalkyl radicals are found herein as examples R.sup.10.1 to R.sup.10.57.
[0115] The term "C.sub.3-C.sub.11-cycloalkyl" refers to a monocyclic, bicyclic or tricyclic saturated univalent hydrocarbon radical having 3 to 11 carbon ring members that is connected through one of the ring carbon atoms by substitution of one hydrogen atom, such as cyclopropyl (C.sub.3H.sub.5), cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, bicyclo[1.1.0]butyl, bicyclo[2.1.0]pentyl, bicyclo[1.1.1]pentyl, bicyclo[3.1.0]hexyl, bicyclo[2.1.1]hexyl, norcaranyl (bicyclo[4.1.0]heptyl) and norbornyl (bicyclo[2.2.1]heptyl).
[0116] The term "C.sub.3-C.sub.11-cycloalkyl-C.sub.1-C.sub.6-alkyl" refers to alkyl having 1 to 11 carbon atoms, wherein one hydrogen atom of the alkyl radical is replaced by a C.sub.3-C.sub.11-cycloalkyl group as defined above.
[0117] The term "C.sub.3-C.sub.11-cycloalkoxy" refers to a cyclic univalent hydrocarbon radical having 3 to 11 carbon ring members (as defined above) that is bonded via an oxygen, at any position in the cycloalkyl group, for example cyclopropyloxy.
[0118] The terms "--C(.dbd.O)--C.sub.1-C.sub.4-alkyl", "--C(.dbd.O)--C.sub.1-C.sub.4-alkoxy" and "--C(.dbd.O)--C.sub.3-C.sub.11-cycloalkyl" refer to radicals which are attached through the carbon atom of the --C(.dbd.O)-- group.
[0119] The term "aliphatic" refers to compounds or radicals composed of carbon and hydrogen and which are non-aromatic compounds. An "alicyclic" compound or radical is an organic compound that is both aliphatic and cyclic. They contain one or more all-carbon rings which may be either saturated or unsaturated, but do not have aromatic character.
[0120] The terms "cyclic moiety" or "cyclic group" refer to a radical which is an alicyclic ring or an aromatic ring, such as, for example, phenyl or heteroaryl.
[0121] The term "and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with . . . " refers to aliphatic groups, cyclic groups and groups, which contain an aliphatic and a cyclic moiety in one group, such as in, for example, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl; therefore a group which contains an aliphatic and a cyclic moiety both of these moieties may be substituted or unsubstituted independently of each other.
[0122] The term "phenyl" refers to an aromatic ring systems including six carbon atoms (commonly referred to as benzene ring.
[0123] The term "heteroaryl" refers to aromatic monocyclic or polycyclic ring systems including besides carbon atoms, 1, 2, 3 or 4 heteroatoms independently selected from the group consisting of N, O and S.
[0124] The term "saturated 3- to 7-membered carbocycle" is to be understood as meaning monocyclic saturated carbocycles having 3, 4 or 5 carbon ring members. Examples include cyclopropyl, cyclopentyl, cyclohexyl, cycloheptyl, and the like.
[0125] The term "3- to 10-membered saturated, partially unsaturated or aromatic mono- or bicyclic heterocycle, wherein the ring member atoms of said mono- or bicyclic heterocycle include besides carbon atoms further 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms", is to be understood as meaning both, aromatic mono- and bicyclic heteroaromatic ring systems, and also saturated and partially unsaturated heterocycles, for example: a 3- or 4-membered saturated heterocycle which contains 1 or 2 heteroatoms from the group consisting of N, O and S as ring members such as oxirane, aziridine, thiirane, oxetane, azetidine, thiethane, [1,2]dioxetane, [1,2]dithietane, [1,2]diazetidine;
[0126] and a 5- or 6-membered saturated or partially unsaturated heterocycle which contains 1, 2 or 3 heteroatoms from the group consisting of N, O and S as ring members such as 2-tetrahydrofuranyl, 3-tetrahydrofuranyl, 2-tetrahydrothienyl, 3-tetrahydrothienyl, 2-pyrrolidinyl, 3-pyrrolidinyl, 3-isoxazolidinyl, 4-isoxazolidinyl, 5-isoxazolidinyl, 3-isothiazolidinyl, 4-isothiazolidinyl, 5-isothiazolidinyl, 3-pyrazolidinyl, 4-pyrazolidinyl, 5-pyrazolidinyl, 2-oxazolidinyl, 4-oxazolidinyl, 5-oxazolidinyl, 2-thiazolidinyl, 4-thiazolidinyl, 5-thiazolidinyl, 2-imidazolidinyl, 4-imidazolidinyl, 1,2,4-oxadiazolidin-3-yl, 1,2,4-oxadiazolidin-5-yl, 1,2,4-thiadiazolidin-3-yl, 1,2,4-thiadiazolidin-5-yl, 1,2,4-triazolidin-3-yl, 1,3,4-oxadiazolidin-2-yl, 1,3,4-thiadiazolidin-2-yl, 1,3,4-triazolidin-2-yl, 2,3-dihydrofur-2-yl, 2,3-dihydrofur-3-yl, 2,4-dihydrofur-2-yl, 2,4-dihydrofur-3-yl, 2,3-dihydrothien-2-yl, 2,3-dihydrothien-3-yl, 2,4-dihydrothien-2-yl, 2,4-dihydrothien-3-yl, 2-pyrrolin-2-yl, 2-pyrrolin-3-yl, 3-pyrrolin-2-yl, 3-pyrrolin-3-yl, 2-isoxazolin-3-yl, 3-isoxazolin-3-yl, 4-isoxazolin-3-yl, 2-isoxazolin-4-yl, 3-isoxazolin-4-yl, 4-isoxazolin-4-yl, 2-isoxazolin-5-yl, 3-isoxazolin-5-yl, 4-isoxazolin-5-yl, 2-isothiazolin-3-yl, 3-isothiazolin-3-yl, 4-isothiazolin-3-yl, 2-isothiazolin-4-yl, 3-isothiazolin-4-yl, 4-isothiazolin-4-yl, 2-isothiazolin-5-yl, 3-isothiazolin-5-yl, 4-isothiazolin-5-yl, 2,3-dihydropyrazol-1-yl, 2,3-dihydropyrazol-2-yl, 2,3-dihydropyrazol-3-yl, 2,3-dihydropyrazol-4-yl, 2,3-dihydropyrazol-5-yl, 3,4-dihydropyrazol-1-yl, 3,4-dihydropyrazol-3-yl, 3,4-dihydropyrazol-4-yl, 3,4-dihydropyrazol-5-yl, 4,5-dihydropyrazol-1-yl, 4,5-dihydropyrazol-3-yl, 4,5-dihydropyrazol-4-yl, 4,5-dihydropyrazol-5-yl, 2,3-dihydrooxazol-2-yl, 2,3-dihydrooxazol-3-yl, 2,3-dihydrooxazol-4-yl, 2,3-dihydrooxazol-5-yl, 3,4-dihydrooxazol-2-yl, 3,4-dihydrooxazol-3-yl, 3,4-dihydrooxazol-4-yl, 3,4-dihydrooxazol-5-yl, 3,4-dihydrooxazol-2-yl, 3,4-dihydrooxazol-3-yl, 3,4-dihydrooxazol-4-yl, 2-piperidinyl, 3-piperidinyl, 4-piperidinyl, 1,3-dioxan-5-yl, 2-tetrahydropyranyl, 4-tetrahydropyranyl, 2-tetrahydrothienyl, 3-hexahydropyridazinyl, 4-hexahydropyridazinyl, 2-hexahydropyrimidinyl, 4-hexahydropyrimidinyl, 5-hexahydropyrimidinyl, 2-piperazinyl, 1,3,5-hexahydrotriazin-2-yl and 1,2,4-hexahydrotriazin-3-yl and also the corresponding -ylidene radicals; and
[0127] a 7-membered saturated or partially unsaturated heterocycle such as tetra- and hexahydroazepinyl, such as 2,3,4,5-tetrahydro[1H]azepin-1-,-2-,-3-,-4-,-5-,-6- or- 7-yl, 3,4,5,6-tetrahydro[2H]azepin-2-,-3-,-4-,-5-,-6- or- 7-yl, 2,3,4,7-tetrahydro[1H]azepin-1-,-2-,-3-,-4-,-5-,-6- or- 7-yl, 2,3,6,7-tetrahydro[1H]azepin-1-,-2-,-3-,-4-,-5-,-6- or- 7-yl, hexahydroazepin-1-,-2-,-3- or- 4-yl, tetra- and hexahydrooxepinyl such as 2,3,4,5-tetrahydro[1H]oxepin-2-,-3-,-4-,-5-,-6- or- 7-yl, 2,3,4,7-tetrahydro[1H]oxepin-2-,-3-,-4-,-5-,-6- or- 7-yl, 2,3,6,7-tetrahydro[1H]oxepin-2-,-3-,-4-,-5-,-6- or- 7-yl, hexahydroazepin-1-,-2-,-3- or- 4-yl, tetra- and hexahydro-1,3-diazepinyl, tetra- and hexahydro-1,4-diazepinyl, tetra- and hexahydro-1,3-oxazepinyl, tetra- and hexahydro-1,4-oxazepinyl, tetra- and hexahydro-1,3-dioxepinyl, tetra- and hexahydro-1,4-dioxepinyl and the corresponding -ylidene radicals.
[0128] The term "5- or 6-membered heteroaryl" or the term "5- or 6-membered aromatic heterocycle" refer to aromatic ring systems including besides carbon atoms, 1, 2, 3 or 4 heteroatoms independently selected from the group consisting of N, O and S, for example, a 5-membered heteroaryl such as pyrrol-1-yl, pyrrol-2-yl, pyrrol-3-yl, thien-2-yl, thien-3-yl, furan-2-yl, furan-3-yl, pyrazol-1-yl, pyrazol-3-yl, pyrazol-4-yl, pyrazol-5-yl, imidazol-1-yl, imidazol-2-yl, imidazol-4-yl, imidazol-5-yl, oxazol-2-yl, oxazol-4-yl, oxazol-5-yl, isoxazol-3-yl, isoxazol-4-yl, isoxazol-5-yl, thiazol-2-yl, thiazol-4-yl, thiazol-5-yl, isothiazol-3-yl, isothiazol-4-yl, isothiazol-5-yl, 1,2,4-triazolyl-1-yl, 1,2,4-triazol-3-yl 1,2,4-triazol-5-yl, 1,2,4-oxadiazol-3-yl, 1,2,4-oxadiazol-5-yl and 1,2,4-thiadiazol-3-yl, 1,2,4-thiadiazol-5-yl; or a 6-membered heteroaryl, such as pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-4-yl, pyrimidin-5-yl, pyrazin-2-yl and 1,3,5-triazin-2-yl and 1,2,4-triazin-3-yl.
[0129] In respect of the variables, the embodiments of the intermediates correspond to the embodiments of the compounds I. Preference is given to those compounds I and, where applicable, also to compounds of all subformulae provided herein, e. g. formulae I.1, I.2, II, Ia, III, wherein the variables have independently of each other or more preferably in combination (any possible combination of 2 or more substituents as defined herein) the following meanings:
[0130] According to one embodiment of the invention in compounds of formulae I, II, and III, R is methyl, ethyl, iso-propyl, CH.sub.2F, CHF.sub.2, 2,2,2-trifluoroethyl or cyclopropyl.
[0131] According to a further embodiment in compounds of formulae I, II, and III, R is CR.sup.3R.sup.4NR.sup.1R.sup.2, wherein R.sup.3, R.sup.4 and R.sup.1 are as defined or preferably defined herein and R.sup.2 is hydrogen. According to a further embodiment in compounds of formulae I, II, and III, R is C(.dbd.O)NR.sup.1R.sup.2, CR.sup.3R.sup.4C(.dbd.O)NR.sup.1R.sup.2 or CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1, wherein R.sup.3, R.sup.4 and R.sup.1 are as defined or preferably defined herein and R.sup.2 is hydrogen.
[0132] Further embodiments of the present invention relate to the process for preparing compounds I, wherein group R is defined as follows:
[0133] Embodiment R.1: R is COOH, --C(.dbd.W)NR.sup.1R.sup.2, CR.sup.3R.sup.4COOH, CR.sup.3R.sup.4C(.dbd.W)NR.sup.1R.sup.2 or CR.sup.3R.sup.4NR.sup.2C(.dbd.W)R.sup.1.
[0134] Embodiment R.2: R is COOH, --C(.dbd.O)NR.sup.1R.sup.2, CR.sup.3R.sup.4COOH, CR.sup.3R.sup.4C(.dbd.O)NR.sup.1R.sup.2 or CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1.
[0135] Embodiment R.3: R is COOH or CR.sup.3R.sup.4COOH.
[0136] Embodiment R.4: R is --C(.dbd.W)NR.sup.1R.sup.2, CR.sup.3R.sup.4C(.dbd.W)NR.sup.1R.sup.2 or CR.sup.3R.sup.4NR.sup.2C(.dbd.W)R.sup.1.
[0137] Embodiment R.5: R is --C(.dbd.O)NR.sup.1R.sup.2, CR.sup.3R.sup.4C(.dbd.O)NR.sup.1R.sup.2 or CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1.
[0138] Embodiment R.6: R is CR.sup.3R.sup.4NR.sup.2R.sup.1.
[0139] Embodiment R.7: R is CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1.
[0140] Even more preferred embodiments of the present invention relate to the process for preparing compounds I, wherein group R in formulae of compounds I, II and III is defined as follows:
[0141] Embodiment R.8: R is CF.sub.2COOH or CF.sub.2C(.dbd.O)NR.sup.1R.sup.2.
[0142] Embodiment R.9: R is CH.sub.2COOH or CH.sub.2C(.dbd.O)NR.sup.1R.sup.2.
[0143] Embodiment R.10: R is --C(CH.sub.2--CH.sub.2)COOH or --C(CH.sub.2--CH.sub.2)C(.dbd.O)NR.sup.1R.sup.2.
[0144] According to a further embodiment, W in compounds of formulae I, II and III is O.
[0145] Further embodiments of the present invention relate to the process for preparing compounds I, wherein group R.sup.1 in formulae of compounds I, II and III is defined as follows: Embodiment 1.1: R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0146] Embodiment 1.2: R.sup.1 is phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0147] Embodiment 1.3: R.sup.1 is C.sub.3-C.sub.8-cycloalkyl or C.sub.3-C.sub.8-cycloalkenyl; and wherein the cyclic group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0148] Embodiment 1.4: R.sup.1 is C.sub.1-C.sub.6-alkyl; and wherein the alkyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0149] Embodiment 1.5: R.sup.1 is difluoromethyl, trifluoromethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro-2,2-difluoroethyl, 2,2,2-trichloroethyl and pentafluoroethyl, 3,3,3-trifluoropropyl, CH.sub.2CF.sub.2CF.sub.3 or CF.sub.2CF.sub.2CF.sub.5, CH(CH.sub.3)CF.sub.3, CH.sub.2CF.sub.2CH.sub.3, CH.sub.2C(CH.sub.3).sub.2F, CH.sub.2CH(CH.sub.3)CF.sub.3 or CH.sub.2C(CH.sub.3).sub.2CF.sub.3.
[0150] Embodiment 1.6: R.sup.1 is C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.2-C.sub.6-alkenyloxyimino-C.sub.1-C.sub.4-alkyl or C.sub.2-C.sub.6-alkynyloxyimino-C.sub.1-C.sub.4-alkyl.
[0151] Embodiment 1.7: R.sup.1 is a bicyclic or tricyclic C.sub.4-C.sub.11-cycloalkyl which is unsubstituted or substituted with 1, 2 or 3 radicals selected from the group consisting of oxo, hydroxy, halogen and C.sub.1-C.sub.3-alkyl.
[0152] Embodiment 1.8: R.sup.1 is a bicyclic or tricyclic carbocycle selected from the group consisting of radicals R.sup.10.1 to R.sup.10.31 below; wherein each radical may be connected to the remainder of the compounds of formula I through one of the ring carbon atoms by substitution of one hydrogen atom; and wherein R.sup.1 is unsubstituted or substituted with 1, 2 or 3 radicals selected from the group consisting of oxo, hydroxy, halogen and C.sub.1-C.sub.3-alkyl.
##STR00011## ##STR00012## ##STR00013##
[0153] Embodiment 1.9: R.sup.1 is selected from the group consisting of R.sup.10.32 to R.sup.10.57 below, particularly from R.sup.10.32 to R.sup.10.49, which are further unsubstituted, and wherein "# C" indicates the carbon atom, which is attached to the remainder of the compounds of formula I.
##STR00014## ##STR00015## ##STR00016##
[0154] Further embodiments of the present invention relate to the process for preparing compounds I, wherein group R.sup.1a is defined as follows: In one embodiment R.sup.1a is selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-haloalkyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.6-haloalkoxy and C.sub.3-C.sub.8-cycloalkyl.
[0155] In another aspect of the invention R.sup.1a is selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy or cyclopropyl.
[0156] In another embodiment R.sup.1a is selected from the group consisting of halogen, C.sub.1-C.sub.6-alkyl and C.sub.3-C.sub.8-cycloalkyl; particularly from methyl, ethyl, fluorine and chlorine; more particularly from fluorine and chlorine.
[0157] According to a further embodiment in compounds of formulae I, II, and III, R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.3-C.sub.6-cycloalkyl or a 4- to 5-membered saturated or partially unsaturated heterocycle, wherein the ring member atoms of said heterocycle include besides carbon atoms 1 or 2 heteroatoms selected from N and O as ring member atoms; and wherein any of the above-mentioned aliphatic or cyclic groups R.sup.1 are unsubstituted or substituted with 1, 2 or 3 of identical or different groups R.sup.1a; wherein R.sup.1a is halogen, oxo, cyano, NO.sub.2, OH, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-haloalkyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.6-haloalkoxy or C.sub.3-C.sub.8-cycloalkyl.
[0158] In another embodiment R.sup.2 in formulae of compounds I, II and III is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, pyridinyl or --N(R.sup.2a).sub.2; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; more preferably from halogen, in particular the radical is fluorine; and wherein R.sup.2a is independently selected from the group consisting of hydrogen, OH, C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.3-C.sub.8-cycloalkyl or C.sub.1-C.sub.6-alkoxy.
[0159] Further embodiments of the present invention relate to the process for preparing compounds I, wherein group R.sup.2 in formulae of compounds I, II and III is defined as follows:
[0160] Embodiment 2.1: R.sup.2 independently of each other are hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy.
[0161] Embodiment 2.2: R.sup.2 independently of each other are hydrogen, formyl, methyl, ethyl, n-propyl, iso-propyl, methoxy, ethyoxy, propyloxy, cyclopropyl, cyclopropyl-CH.sub.2--, allyl, phenyl, 4-F-phenyl, 2-F-phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino.
[0162] Embodiment 2.3: R.sup.2 independently of each other are hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino.
[0163] Embodiment 2.4: R.sup.2 independently of each other are hydrogen, formyl, methyl, ethyl, n-propyl, iso-propyl, methoxy, ethyoxy, propyloxy, cyclopropyl, cyclopropyl-CH.sub.2--, allyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino.
[0164] Embodiment 2.5: R.sup.2 independently of each other are hydrogen, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl or propargyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino.
[0165] Embodiment 2.6: R.sup.2 independently of each other are hydrogen, methy, ethyl, methoxy, ethyoxy, propyloxy, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino.
[0166] Embodiment 2.7: R.sup.2 is hydrogen.
[0167] According to a further embodiment in compounds of formulae I, II, and III, R.sup.2 is C.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.6-alkoxy or C.sub.3-C.sub.8-cycloalkyl, and wherein any of the aliphatic or cyclic groups in R.sup.2 are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from halogen.
[0168] In one embodiment R.sup.2 in formulae of compounds I, II and III is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, pyridinyl or --N(R.sup.2a).sub.2; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; more preferably from halogen, in particular the radical is fluorine; and wherein R.sup.2a is independently selected from the group consisting of hydrogen, OH, C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.3-C.sub.8-cycloalkyl or C.sub.1-C.sub.6-alkoxy; and R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups in R.sup.1 are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0169] In another aspect R.sup.2 in formulae of compounds I, II and III is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and R.sup.1 in formulae of compounds I, II and III is C.sub.1-C.sub.6-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups in R.sup.1 are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals R.sup.1a as defined or preferably defined herein.
[0170] In another aspect R.sup.2 in formulae of compounds I, II and III is hydrogen, formyl, methyl, ethyl, n-propyl, iso-propyl, methoxy, ethyoxy, propyloxy, cyclopropyl, cyclopropyl-CH.sub.2--, allyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and R.sup.1 in formulae of compounds I, II and III is C.sub.1-C.sub.6-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.2-C.sub.6-alkenyl or C.sub.2-C.sub.6-alkynyl; and wherein any of the aliphatic or cyclic groups in R.sup.1 are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen or C.sub.1-C.sub.6-alkyl.
[0171] Further embodiments of the present invention relate to the process for preparing compounds I, wherein groups R.sup.3 and R.sup.4 in formulae of compounds I, II and III, if present, are defined as follows:
[0172] Embodiment 3.1: R.sup.3 and R.sup.4 independently of each other are hydrogen, halogen, C.sub.1-C.sub.6-alkyl or C.sub.1-C.sub.6-haloalkyl; or R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a cyclopropyl ring, wherein the cyclopropyl ring is unsubstituted.
[0173] Embodiment 3.2: R.sup.3 and R.sup.4 independently of each other are hydrogen or C.sub.1-C.sub.4-alkyl; Embodiment 3.3: R.sup.3 and R.sup.4 independently of each other are hydrogen, methyl or ethyl.
[0174] Embodiment 3.4: R.sup.3 and R.sup.4 are independently of each other hydrogen, fluorine, chlorine, methyl or trifluoromethyl; or R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a cyclopropyl ring, wherein the cyclopropyl ring is unsubstituted.
[0175] Embodiment 3.5: R.sup.3 and R.sup.4 are both hydrogen.
[0176] Embodiment 3.6: R.sup.3 is hydrogen and R.sup.4 is methyl.
[0177] Embodiment 3.7: R.sup.3 and R.sup.4 are both methyl.
[0178] Embodiment 3.8: R.sup.3 and R.sup.4 are both fluorine.
[0179] Embodiment 3.9: R.sup.3 and R.sup.4 are both trifluoromethyl.
[0180] Embodiment 3.10: R.sup.3 and R.sup.4 together with the carbon atom to which they are bound a saturated monocyclic 3- to 5-membered saturated heterocycle or saturated carbocycle; and wherein the saturated heterocycle includes beside one or more carbon atoms no heteroatoms or 1 or 2 heteroatoms independently selected from N, O and S as ring member atoms; and wherein the heterocycle or the carbocycle is unsubstituted or substituted 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano and C.sub.1-C.sub.2-alkyl.
[0181] Embodiment 3.11: R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a 3- or 4-membered carbocylic ring; and wherein the carbocylic ring is unsubstituted.
[0182] Embodiment 3.12: R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a cyclopropyl ring, wherein the cyclopropyl ring is unsubstituted.
[0183] Embodiment 3.13: R.sup.3 and R.sup.4 together with the carbon atom to which they are bound form a saturated 3-membered heterocycle; wherein the heterocycle includes beside two carbon atoms one heteroatom selected from N, O and S as ring member atoms; and wherein the heterocycle is unsubstituted or substituted 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano and C.sub.1-C.sub.2-alkyl.
[0184] Embodiment 3.14: R.sup.3 is methyl and R.sup.4 is fluorine.
[0185] Embodiment 3.15: R.sup.3 is hydrogen and R.sup.4 is fluorine.
[0186] According to a further embodiment in compounds of formulae I, II, and III, R is CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1, wherein R.sup.3 and R.sup.4 are independently selected from hydrogen and C.sub.1-C.sub.4-alkyl, in particular both are hydrogen; and wherein R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.3-C.sub.6-cycloalkyl or a 4- to 5-membered saturated or partially unsaturated heterocycle, wherein the ring member atoms of said heterocycle include besides carbon atoms 1 or 2 heteroatoms selected from N and O as ring member atoms; and wherein any of the above-mentioned aliphatic or cyclic groups R.sup.1 are unsubstituted or substituted with 1, 2 or 3 of identical or different groups R.sup.1a; wherein R.sup.1a is halogen, oxo, cyano, NO.sub.2, OH, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-haloalkyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.6-haloalkoxy or C.sub.3-C.sub.8-cycloalkyl; and wherein R.sup.2 is hydrogen, C.sub.1-C.sub.4-alkyl, C.sub.1-C.sub.6-alkoxy or C.sub.3-C.sub.8-cycloalkyl, and wherein any of the aliphatic or cyclic groups in R.sup.2 are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from halogen.
[0187] According to a further embodiment in compounds of formulae I, II, and III, R is CR.sup.3R.sup.4NR.sup.2C(.dbd.O)R.sup.1, wherein R.sup.3 and R.sup.4 are both hydrogen; and wherein R.sup.1 is C.sub.3-C.sub.6-cycloalkyl or a 4- to 5-membered saturated or partially unsaturated heterocycle, wherein the ring member atoms of said heterocycle include besides carbon atoms 1 or 2 heteroatoms selected from N and O as ring member atoms; and wherein any of the above-mentioned aliphatic or cyclic groups R.sup.1 are unsubstituted or substituted with 1, 2 or 3 of identical or different groups R.sup.1a; wherein R.sup.1a is halogen, oxo, cyano, NO.sub.2, OH, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-haloalkyl, C.sub.1-C.sub.6-alkoxy, C.sub.1-C.sub.6-haloalkoxy or C.sub.3-C.sub.8-cycloalkyl; and wherein R.sup.2 is cyclopropyl.
[0188] Further embodiments with regard to the meaning of the variables in compounds of formula I are as follows:
[0189] Embodiment E1: A.sup.1 and A.sup.2 are CH and R.sup.A is hydrogen.
[0190] Embodiment E2: A.sup.1 and A.sup.2 are CH and R.sup.A is fluorine.
[0191] Embodiment E3: A.sup.1 is N, A.sup.2 is CH and R.sup.A is hydrogen.
[0192] Embodiment E4: A.sup.1 and A.sup.2 are CH, R.sup.A is hydrogen, and R is methyl, trichloromethyl, --COOH, OH, SH, cyano, chlorine or bromine.
[0193] Embodiment E5: A.sup.1 and A.sup.2 are CH, R.sup.A is fluorine, and R is methyl, trichloromethyl, --COOH, OH, SH, cyano, chlorine or bromine.
[0194] Embodiment E6: A.sup.1 is N, A.sup.2 is CH, R.sup.A is hydrogen, and R is methyl, trichloromethyl, --COOH, OH, SH, cyano, chlorine or bromine.
[0195] Embodiment E7: A.sup.1 and A.sup.2 are CH, R.sup.A is hydrogen, and R is methyl, trichloromethyl, OH or SH; particularly R is methyl.
[0196] Embodiment E8: A.sup.1 and A.sup.2 are CH, R.sup.A is fluorine, and R is methyl, trichloromethyl, OH or SH; particularly R is methyl.
[0197] Embodiment E9: A.sup.1 is N, A.sup.2 is CH, R.sup.A is hydrogen, and R is methyl, trichloromethyl, OH or SH; particularly R is methyl.
[0198] Embodiment E10: the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above, and R is methyl.
[0199] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0200] R is methyl, trichloromethyl, ethyl, iso-propyl, OH, SH, cyano, halogen, CH.sub.2F, CHF.sub.2, 2,2,2-trifluoroethyl, cyclopropyl, --COOH, --COOR.sup.1 or --C(.dbd.W)NR.sup.1R.sup.2; [0201] W is O or S; [0202] R.sup.1 C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; [0203] R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy.
[0204] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0205] R is --COOH, --COOR.sup.1 or --C(.dbd.W)NR.sup.1R.sup.2; [0206] W is O or S; [0207] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy and cyclopropyl. [0208] R.sup.2 is hydrogen, methyl or ethyl.
[0209] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0210] R is --COOH, --COOR.sup.1 or --C(.dbd.W)NR.sup.1R.sup.2; [0211] W is O or S; [0212] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1 radical selected from the group consisting of fluorine and 2-difluoromethoxy-phenyl; [0213] R.sup.2 is hydrogen, methyl or ethyl.
[0214] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0215] R is --C(.dbd.O)NR.sup.1R.sup.2; [0216] R.sup.1 is methyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, 2-fluoro-phenyl, 4-fluoro-phenyl, or 2-difluoromethoxy-phenyl; [0217] R.sup.2 is hydrogen.
[0218] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0219] R is --C(.dbd.S)NR.sup.1R.sup.2; [0220] R.sup.1 is methyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, 2-fluoro-phenyl, 4-fluoro-phenyl, or 2-difluoromethoxy-phenyl; [0221] R.sup.2 is hydrogen.
[0222] In a further embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0223] A.sup.1 and A.sup.2 are CH; [0224] R.sup.A is hydrogen; [0225] R is --C(.dbd.O)NR.sup.1R.sup.2; [0226] R.sup.1 is methyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, 2-fluoro-phenyl, 4-fluoro-phenyl, or 2-difluoromethoxy-phenyl; [0227] R.sup.2 is hydrogen.
[0228] In a further embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0229] A.sup.1 and A.sup.2 are CH; [0230] R.sup.A is hydrogen; [0231] R is --C(.dbd.S)NR.sup.1R.sup.2; [0232] R.sup.1 is methyl, 2-methoxyiminoethyl, bicyclo[1.1.1]pentan-1-yl, 2-fluoro-phenyl, 4-fluoro-phenyl, or 2-difluoromethoxy-phenyl; [0233] R.sup.2 is hydrogen.
[0234] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0235] R is --CF.sub.2COOH, --CF.sub.2COR.sup.1 or --CF.sub.2C(.dbd.O)NR.sup.1R.sup.2; [0236] R.sup.1 C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; [0237] R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy.
[0238] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0239] R is --CF.sub.2COOH, --CF.sub.2COR.sup.1 or --CF.sub.2C(.dbd.O)NR.sup.1R.sup.2; [0240] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy and cyclopropyl. [0241] R.sup.2 is hydrogen, methyl or ethyl.
[0242] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0243] A.sup.1 and A.sup.2 are CH; [0244] R.sup.A is hydrogen or fluorine; [0245] R is --CF.sub.2COOH, --CF.sub.2COR.sup.1 or --CF.sub.2C(.dbd.O)NR.sup.1R.sup.2; [0246] R.sup.1 is 1-methyl-cycloprop-1-yl or cyclobutyl; [0247] R.sup.2 is hydrogen, methyl or ethyl.
[0248] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0249] A.sup.1 and A.sup.2 are CH; [0250] R.sup.A is hydrogen; [0251] R is --CF.sub.2COOH, --CF.sub.2COR.sup.1 or --CF.sub.2C(.dbd.O)NR.sup.1R.sup.2; [0252] R.sup.1 is 1-methyl-cycloprop-1-yl or cyclobutyl; [0253] R.sup.2 is hydrogen.
[0254] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0255] R is --OCF.sub.2COOH, --OCF.sub.2COR.sup.1 or --OCF.sub.2C(.dbd.W)NR.sup.1R.sup.2; [0256] R.sup.1 C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; [0257] R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy.
[0258] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0259] R is --OCF.sub.2COOH, --OCF.sub.2COR.sup.1 or --OCF.sub.2C(.dbd.W)NR.sup.1R.sup.2; [0260] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy and cyclopropyl. [0261] R.sup.2 is hydrogen, methyl or ethyl.
[0262] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0263] A.sup.1 and A.sup.2 are CH; [0264] R.sup.A is hydrogen or fluorine; [0265] R is --OCF.sub.2COOH, --OCF.sub.2COR.sup.1 or --OCF.sub.2C(.dbd.W)NR.sup.1R.sup.2; [0266] R.sup.1 is methyl or cyclopropyl; [0267] R.sup.2 is hydrogen, methyl or ethyl.
[0268] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0269] A.sup.1 and A.sup.2 are CH; [0270] R.sup.A is hydrogen; [0271] R is --OCF.sub.2COOH, --OCF.sub.2COR.sup.1 or --OCF.sub.2C(.dbd.W)NR.sup.1R.sup.2; [0272] R.sup.1 is methyl or cyclopropyl; [0273] R.sup.2 is hydrogen.
[0274] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0275] R is --CH.sub.2NR.sup.2C(.dbd.W)R.sup.1, --CH.sub.2S(.dbd.O).sub.2R.sup.1 or --CH.sub.2NR.sup.2S(.dbd.O).sub.2R.sup.1; [0276] W is O or S; [0277] R.sup.1 is C.sub.1-C.sub.6-alkyl, C.sub.2-C.sub.6-alkenyl, C.sub.2-C.sub.6-alkynyl, C.sub.1-C.sub.6-alkoxyimino-C.sub.1-C.sub.4-alkyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, phenyl-C.sub.1-C.sub.4-alkyl, heteroaryl-C.sub.1-C.sub.4-alkyl, phenyl or heteroaryl; and wherein the heteroaryl group is a 5- or 6-membered aromatic heterocycle, wherein the ring includes besides carbon atoms 1, 2, 3 or 4 heteroatoms selected from N, O and S as ring member atoms; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy; [0278] R.sup.2 is hydrogen, formyl, C.sub.1-C.sub.6-alkyl, C.sub.1-C.sub.6-alkoxy, C.sub.2-C.sub.6-alkenyl, propargyl, C.sub.3-C.sub.8-cycloalkyl, C.sub.3-C.sub.8-cycloalkenyl, C.sub.3-C.sub.8-cycloalkyl-C.sub.1-C.sub.4-alkyl, phenyl, C.sub.1-C.sub.6-alkylamino or diC.sub.1-C.sub.6-alkylamino; and wherein any of the aliphatic or cyclic groups are unsubstituted or substituted with 1, 2, 3, 4 or up to the maximum possible number of identical or different radicals selected from the group consisting of halogen, cyano, C.sub.1-C.sub.6-alkyl and C.sub.1-C.sub.6-alkoxy.
[0279] In one embodiment the present invention relates to a process for preparing compounds of formula I, wherein the combination of variables A.sup.1, A.sup.2, R.sup.A corresponds to any one of the Embodiments E1, E2 or E3 defined above; and [0280] R is --CH.sub.2NR.sup.2C(.dbd.W)R.sup.1, --CH.sub.2S(.dbd.O).sub.2R.sup.1 or --CH.sub.2NR.sup.2S(.dbd.O).sub.2R.sup.1; [0281] W is O or S; [0282] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1, 2, 3 or up to the maximum possible number of identical or different radicals selected from the group consisting of fluorine, chlorine, cyano, methyl, ethyl, methoxy, trifluoromethyl, trifluoromethoxy, difluoromethyl, difluoromethoxy and cyclopropyl. [0283] R.sup.2 is hydrogen, methyl or ethyl.
[0284] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0285] A.sup.1 and A.sup.2 are CH; [0286] R.sup.A is hydrogen or fluorine; [0287] R is --CH.sub.2NR.sup.2C(.dbd.W)R.sup.1, --CH.sub.2S(.dbd.O).sub.2R.sup.1 or --CH.sub.2NR.sup.2S(.dbd.O).sub.2R.sup.1; [0288] W is O or S; [0289] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl, bicyclo[1.1.1]pentan-1-yl, or phenyl; and wherein the phenyl group is unsubstituted or substituted with 1 radical selected from the group consisting of fluorine and chlorine; [0290] R.sup.2 is hydrogen, methyl or ethyl.
[0291] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0292] A.sup.1 and A.sup.2 are CH; [0293] R.sup.A is hydrogen or fluorine; [0294] R is --CH.sub.2NR.sup.2C(.dbd.O)R.sup.1, --CH.sub.2S(.dbd.O).sub.2R.sup.1 or --CH.sub.2NR.sup.2S(.dbd.O).sub.2R.sup.1; [0295] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl or bicyclo[1.1.1]pentan-1-yl; [0296] R.sup.2 is hydrogen.
[0297] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0298] A.sup.1 and A.sup.2 are CH; [0299] R.sup.A is hydrogen or fluorine; [0300] R is --CH.sub.2NR.sup.2C(.dbd.O)R.sup.1 or --CH.sub.2NR.sup.2S(.dbd.O).sub.2R.sup.1; [0301] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl or bicyclo[1.1.1]pentan-1-yl; [0302] R.sup.2 is hydrogen.
[0303] In another embodiment the present invention relates to a process for preparing compounds of formula I, wherein [0304] A.sup.1 and A.sup.2 are CH; [0305] R.sup.A is hydrogen or fluorine; [0306] R is --CH.sub.2S(.dbd.O).sub.2R.sup.1; [0307] R.sup.1 is methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, 2-methoxyiminoethyl, cyclopropyl or bicyclo[1.1.1]pentan-1-yl; [0308] R.sup.2 is hydrogen.
[0309] In further aspects of the present invention the embodiments E.1 to E.210 listed in Table E represent preferred combinations of the embodiments, which are defined above for each of the variables R, R.sup.1, R.sup.2, R.sup.3 and R.sup.4.
TABLE-US-00001 TABLE E Embodiment R R.sup.1 R.sup.2 R.sup.3, R.sup.4 E.1 R.1 1.1 2.4 3.4 E.2 R.1 1.2 2.4 3.4 E.3 R.1 1.3 2.4 3.4 E.4 R.1 1.4 2.4 3.4 E.5 R.1 1.5 2.4 3.4 E.6 R.1 1.6 2.4 3.4 E.7 R.1 1.7 2.4 3.4 E.8 R.1 1.8 2.4 3.4 E.9 R.1 1.9 2.4 3.4 E.10 R.1 1.10 2.4 3.4 E.11 R.1 1.1 2.4 3.5 E.12 R.1 1.2 2.4 3.5 E.13 R.1 1.3 2.4 3.5 E.14 R.1 1.4 2.4 3.5 E.15 R.1 1.5 2.4 3.5 E.16 R.1 1.6 2.4 3.5 E.17 R.1 1.7 2.4 3.5 E.18 R.1 1.8 2.4 3.5 E.19 R.1 1.9 2.4 3.5 E.20 R.1 1.10 2.4 3.5 E.21 R.1 1.1 2.4 3.7 E.22 R.1 1.2 2.4 3.7 E.23 R.1 1.3 2.4 3.7 E.24 R.1 1.4 2.4 3.7 E.25 R.1 1.5 2.4 3.7 E.26 R.1 1.6 2.4 3.7 E.27 R.1 1.7 2.4 3.7 E.28 R.1 1.8 2.4 3.7 E.29 R.1 1.9 2.4 3.7 E.30 R.1 1.10 2.4 3.7 E.31 R.1 1.1 2.4 3.8 E.32 R.1 1.2 2.4 3.8 E.33 R.1 1.3 2.4 3.8 E.34 R.1 1.4 2.4 3.8 E.35 R.1 1.5 2.4 3.8 E.36 R.1 1.6 2.4 3.8 E.37 R.1 1.7 2.4 3.8 E.38 R.1 1.8 2.4 3.8 E.39 R.1 1.9 2.4 3.8 E.40 R.1 1.10 2.4 3.8 E.41 R.1 1.1 2.4 3.12 E.42 R.1 1.2 2.4 3.12 E.43 R.1 1.3 2.4 3.12 E.44 R.1 1.4 2.4 3.12 E.45 R.1 1.5 2.4 3.12 E.46 R.1 1.6 2.4 3.12 E.47 R.1 1.7 2.4 3.12 E.48 R.1 1.8 2.4 3.12 E.49 R.1 1.9 2.4 3.12 E.50 R.1 1.10 2.4 3.12 E.51 R.1 1.1 2.7 3.4 E.52 R.1 1.2 2.7 3.4 E.53 R.1 1.3 2.7 3.4 E.54 R.1 1.4 2.7 3.4 E.55 R.1 1.5 2.7 3.4 E.56 R.1 1.6 2.7 3.4 E.57 R.1 1.7 2.7 3.4 E.58 R.1 1.8 2.7 3.4 E.59 R.1 1.9 2.7 3.4 E.60 R.1 1.10 2.7 3.4 E.61 R.1 1.1 2.7 3.5 E.62 R.1 1.2 2.7 3.5 E.63 R.1 1.3 2.7 3.5 E.64 R.1 1.4 2.7 3.5 E.65 R.1 1.5 2.7 3.5 E.66 R.1 1.6 2.7 3.5 E.67 R.1 1.7 2.7 3.5 E.68 R.1 1.8 2.7 3.5 E.69 R.1 1.9 2.7 3.5 E.70 R.1 1.10 2.7 3.5 E.71 R.1 1.1 2.7 3.7 E.72 R.1 1.2 2.7 3.7 E.73 R.1 1.3 2.7 3.7 E.74 R.1 1.4 2.7 3.7 E.75 R.1 1.5 2.7 3.7 E.76 R.1 1.6 2.7 3.7 E.77 R.1 1.7 2.7 3.7 E.78 R.1 1.8 2.7 3.7 E.79 R.1 1.9 2.7 3.7 E.80 R.1 1.10 2.7 3.7 E.81 R.1 1.1 2.7 3.8 E.82 R.1 1.2 2.7 3.8 E.83 R.1 1.3 2.7 3.8 E.84 R.1 1.4 2.7 3.8 E.85 R.1 1.5 2.7 3.8 E.86 R.1 1.6 2.7 3.8 E.87 R.1 1.7 2.7 3.8 E.88 R.1 1.8 2.7 3.8 E.89 R.1 1.9 2.7 3.8 E.90 R.1 1.10 2.7 3.8 E.91 R.1 1.1 2.7 3.12 E.92 R.1 1.2 2.7 3.12 E.93 R.1 1.3 2.7 3.12 E.94 R.1 1.4 2.7 3.12 E.95 R.1 1.5 2.7 3.12 E.96 R.1 1.6 2.7 3.12 E.97 R.1 1.7 2.7 3.12 E.98 R.1 1.8 2.7 3.12 E.99 R.1 1.9 2.7 3.12 E.100 R.1 1.10 2.7 3.12 E.101 R.3 1.1 3.4 E.102 R.3 1.2 3.4 E.103 R.3 1.3 3.4 E.104 R.3 1.4 3.4 E.105 R.3 1.5 3.4 E.106 R.3 1.6 3.4 E.107 R.3 1.7 3.4 E.108 R.3 1.8 3.4 E.109 R.3 1.9 3.4 E.110 R.3 1.10 3.4 E.111 R.3 1.1 3.5 E.112 R.3 1.2 3.5 E.113 R.3 1.3 3.5 E.114 R.3 1.4 3.5 E.115 R.3 1.5 3.5 E.116 R.3 1.6 3.5 E.117 R.3 1.7 3.5 E.118 R.3 1.8 3.5 E.119 R.3 1.9 3.5 E.120 R.3 1.10 3.5 E.121 R.3 1.1 3.7 E.122 R.3 1.2 3.7 E.123 R.3 1.3 3.7 E.124 R.3 1.4 3.7 E.125 R.3 1.5 3.7 E.126 R.3 1.6 3.7 E.127 R.3 1.7 3.7 E.128 R.3 1.8 3.7 E.129 R.3 1.9 3.7 E.130 R.3 1.10 3.7 E.131 R.3 1.1 3.8 E.132 R.3 1.2 3.8 E.133 R.3 1.3 3.8 E.134 R.3 1.4 3.8 E.135 R.3 1.5 3.8 E.136 R.3 1.6 3.8 E.137 R.3 1.7 3.8 E.138 R.3 1.8 3.8 E.139 R.3 1.9 3.8 E.140 R.3 1.10 3.8 E.141 R.3 1.1 3.12 E.142 R.3 1.2 3.12 E.143 R.3 1.3 3.12 E.144 R.3 1.4 3.12 E.145 R.3 1.5 3.12 E.146 R.3 1.6 3.12 E.147 R.3 1.7 3.12 E.148 R.3 1.8 3.12 E.149 R.3 1.9 3.12 E.150 R.3 1.10 3.12 E.151 R.8 1.1 2.4 E.152 R.8 1.2 2.4 E.153 R.8 1.3 2.4 E.154 R.8 1.4 2.4 E.155 R.8 1.5 2.4 E.156 R.8 1.6 2.4 E.157 R.8 1.7 2.4 E.158 R.8 1.8 2.4 E.159 R.8 1.9 2.4 E.160 R.8 1.10 2.4 E.161 R.8 1.1 2.7 E.162 R.8 1.2 2.7 E.163 R.8 1.3 2.7 E.164 R.8 1.4 2.7 E.165 R.8 1.5 2.7 E.166 R.8 1.6 2.7 E.167 R.8 1.7 2.7 E.168 R.8 1.8 2.7 E.169 R.8 1.9 2.7 E.170 R.8 1.10 2.7 E.171 R.9 1.1 2.4 E.172 R.9 1.2 2.4 E.173 R.9 1.3 2.4 E.174 R.9 1.4 2.4 E.175 R.9 1.5 2.4 E.176 R.9 1.6 2.4 E.177 R.9 1.7 2.4 E.178 R.9 1.8 2.4 E.179 R.9 1.9 2.4 E.180 R.9 1.10 2.4 E.181 R.9 1.1 2.7 E.182 R.9 1.2 2.7 E.183 R.9 1.3 2.7 E.184 R.9 1.4 2.7 E.185 R.9 1.5 2.7 E.186 R.9 1.6 2.7 E.187 R.9 1.7 2.7 E.188 R.9 1.8 2.7 E.189 R.9 1.9 2.7 E.190 R.9 1.10 2.7 E.191 R.10 1.1 2.4 E.192 R.10 1.2 2.4 E.193 R.10 1.3 2.4 E.194 R.10 1.4 2.4 E.195 R.10 1.5 2.4 E.196 R.10 1.6 2.4 E.197 R.10 1.7 2.4 E.198 R.10 1.8 2.4 E.199 R.10 1.9 2.4 E.200 R.10 1.10 2.4 E.201 R.10 1.1 2.7 E.202 R.10 1.2 2.7 E.203 R.10 1.3 2.7 E.204 R.10 1.4 2.7 E.205 R.10 1.5 2.7 E.206 R.10 1.6 2.7 E.207 R.10 1.7 2.7 E.208 R.10 1.8 2.7 E.209 R.10 1.9 2.7 E.210 R.10 1.10 2.7
[0310] In one embodiment the present invention relates to compounds of the formula II, namely compounds II.A-1 to II.A-315, wherein A.sup.1 and A.sup.2 are CH, R.sup.A is hydrogen, and R in each of the compounds II.A-1 to II.A-315 is defined as in one corresponding line A-1 to A-315 in Table A below, and wherein # denotes the position, which is bound to the phenyl moiety. This means, for example, that a compound of formula II, wherein R is CO.sub.2H is named II.A-43 in correspondence to the definition in line A-43 in Table A.
TABLE-US-00002 TABLE A No R A-1 CH.sub.3 A-2 CCl.sub.3 A-3 OH A-4 SH A-5 cyano A-6 F A-7 Cl A-8 Br A-9 CH.sub.2CH.sub.3 A-10 CH(CH.sub.3).sub.2 A-11 CH.sub.2F A-12 CHF.sub.2 A-13 2,2,2-trifluoroethyl A-14 cyclopropyl A-15 CH.sub.2OH A-16 C(.dbd.O)CH.sub.3 A-17 CH.sub.2OCH.sub.3 A-18 CH.sub.2OCH.sub.2CH.sub.3 A-19 CH.sub.2OCH.sub.2CH.sub.2CH.sub.3 A-20 CH.sub.2OCH(CH.sub.3).sub.2 A-21 CH.sub.2OC(CH.sub.3).sub.3 A-22 CH.sub.2OCH.sub.2CH.dbd.CH.sub.2 A-23 CH.sub.2OC(phenyl).sub.3 A-24 CH.sub.2OSi(CH.sub.3).sub.3 A-25 CH.sub.2OSi(CH.sub.2phenyl).sub.3 A-26 CH.sub.2OC(.dbd.O)CH.sub.3 A-27 CH.sub.2OC(.dbd.O)CH.sub.2CH.sub.3 A-28 CH.sub.2OC(.dbd.O)CH.sub.2CH.sub.2CH.sub.3 A-29 CH.sub.2OC(.dbd.O)CH(CH.sub.3).sub.2 A-30 CH.sub.2OC(.dbd.O)C(CH.sub.3).sub.3 A-31 CH.sub.2OC(.dbd.O)CH.sub.2CH.dbd.CH.sub.2 A-32 CH.sub.2OC(.dbd.O)OCH.sub.3 A-33 CH.sub.2OC(.dbd.O)OCH.sub.2CH.sub.3 A-34 CH.sub.2OC(.dbd.O)OCH.sub.2CH.sub.2CH.sub.3 A-35 CH.sub.2OC(.dbd.O)OCH(CH.sub.3).sub.2 A-36 CH.sub.2OC(.dbd.O)OC(CH.sub.3).sub.3 A-37 CH.sub.2OC(.dbd.O)OCH.sub.2CH.dbd.CH.sub.2 A-38 CH.sub.2OCH.sub.2OCH.sub.3 A-39 CH.sub.2OCH.sub.2OCH.sub.2CH.sub.3 A-40 CH.sub.2OCH.sub.2OCH.sub.2CH.sub.2CH.sub.3 A-41 CH.sub.2OCH.sub.2OCH(CH.sub.3).sub.2 A-42 CH.sub.2OCH.sub.2O-tetrahydropyran-2-yl A-43 CO.sub.2H A-44 CO.sub.2CH.sub.3 A-45 CO.sub.2CH.sub.2CH.sub.3 A-46 CO.sub.2CH.sub.2CH.sub.2CH.sub.3 A-47 CO.sub.2CH(CH.sub.3).sub.2 A-48 CO.sub.2C(CH.sub.3).sub.3 A-49 CO.sub.2CH.sub.2CH.dbd.CH.sub.2 A-50 CH.sub.2CO.sub.2H A-51 CH.sub.2CO.sub.2CH.sub.3 A-52 CH.sub.2CO.sub.2CH.sub.2CH.sub.3 A-53 CH.sub.2CO.sub.2CH.sub.2CH.sub.2CH.sub.3 A-54 CH.sub.2CO.sub.2CH(CH.sub.3).sub.2 A-55 CH.sub.2CO.sub.2C(CH.sub.3).sub.3 A-56 CH.sub.2CO.sub.2CH.sub.2CH.dbd.CH.sub.2 A-57 CF.sub.2CO.sub.2H A-58 CF.sub.2CO.sub.2CH.sub.3 A-59 CF.sub.2CO.sub.2CH.sub.2CH.sub.3 A-60 CF.sub.2CO.sub.2CH.sub.2CH.sub.2CH.sub.3 A-61 CF.sub.2CO.sub.2CH(CH.sub.3).sub.2 A-62 CF.sub.2CO.sub.2C(CH.sub.3).sub.3 A-63 CF.sub.2CO.sub.2CH.sub.2CH.dbd.CH.sub.2 A-64 pyrrolidine-2-one-methyl A-65 CH.sub.2NH.sub.2 A-66 CH.sub.2NHNH.sub.2 A-67 CH.sub.2NH--N(CH.sub.3).sub.2 A-68 CH.sub.2NH--NH(CH.sub.3) A-69 CH.sub.2NH(CH.sub.3) A-70 CH.sub.2NH(CH.sub.2CH.sub.3) A-71 CH.sub.2NH(CH.sub.2CH.sub.2CH.sub.3) A-72 CH.sub.2NH(CH(CH.sub.3).sub.2) A-73 CH.sub.2NH(C(CH.sub.3).sub.3) A-74 CH.sub.2NH(CH.sub.2CH.dbd.CH.sub.2) A-75 CH.sub.2NH(OCH.sub.3) A-76 CH.sub.2NH(OCH.sub.2CH.sub.3) A-77 CH.sub.2NH(OCH.sub.2CH.sub.2CH.sub.3) A-78 CH.sub.2NH(OCH(CH.sub.3).sub.2) A-79 CH.sub.2NH(OCH.sub.2CH.dbd.CH.sub.2) A-80 CH.sub.2NH(cyclopropyl) A-81 CH.sub.2NH(cyclobutyl) A-82 CH.sub.2NH(cyclopentyl) A-83 CH.sub.2NH(cyclohexyl) A-84 CH.sub.2N(CH.sub.3).sub.2 A-85 CH.sub.2N(CH.sub.3)(CH.sub.2CH.sub.3) A-86 CH.sub.2N(CH.sub.3)(CH.sub.2CH.sub.2CH.sub.3) A-87 CH.sub.2N(CH.sub.3)(CH(CH.sub.3).sub.2) A-88 CH.sub.2N(CH.sub.3)(C(CH.sub.3).sub.3) A-89 CH.sub.2N(CH.sub.3)(CH.sub.2CH.dbd.CH.sub.2) A-90 CH.sub.2N(CH.sub.3)(OCH.sub.3) A-91 CH.sub.2N(CH.sub.3)(OCH.sub.2CH.sub.3) A-92 CH.sub.2N(CH.sub.3)(OCH.sub.2CH.sub.2CH.sub.3) A-93 CH.sub.2N(CH.sub.3)(OCH.sub.2CH.dbd.CH.sub.2) A-94 CH.sub.2N(CH.sub.3)(cyclopropyl) A-95 CH.sub.2N(CH.sub.3)(cyclobutyl) A-96 CH.sub.2N(CH.sub.3)(cyclopentyl) A-97 CH.sub.2N(CH.sub.3)(cyclohexyl) A-98 CH.sub.2NH(C(.dbd.O)H) A-99 CH.sub.2NH(C(.dbd.O)CH.sub.3) A-100 CH.sub.2NH(C(.dbd.O)CH.sub.2CH.sub.3) A-101 CH.sub.2NH(C(.dbd.O)CH.sub.2CH.sub.2CH.sub.3) A-102 CH.sub.2NH(C(.dbd.O)CH(CH.sub.3).sub.2) A-103 CH.sub.2NH(C(.dbd.O)C(CH.sub.3).sub.3) A-104 CH.sub.2NH(C(.dbd.O)CH.sub.2CH.dbd.CH.sub.2) A-105 CH.sub.2NH(C(.dbd.O)OCH.sub.3) A-106 CH.sub.2NH(C(.dbd.O)OCH.sub.2CH.sub.3) A-107 CH.sub.2NH(C(.dbd.O)OCH.sub.2CH.sub.2CH.sub.3) A-108 CH.sub.2NH(C(.dbd.O)OCH.sub.2CH.dbd.CH.sub.2) A-109 CH.sub.2NH(C(.dbd.O)cyclopropyl) A-110 CH.sub.2NH(C(.dbd.O)cyclobutyl) A-111 CH.sub.2NH(C(.dbd.O)cyclopentyl) A-112 CH.sub.2NH(C(.dbd.O)cyclohexyl) A-113 CH.sub.2N(CH.sub.3)(C(.dbd.O)H) A-114 CH.sub.2N(CH.sub.3)(C(.dbd.O)CH.sub.3) A-115 CH.sub.2N(CH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.3) A-116 CH.sub.2N(CH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.2CH.sub.3) A-117 CH.sub.2N(CH.sub.3)(C(.dbd.O)CH(CH.sub.3).sub.2) A-118 CH.sub.2N(CH.sub.3)(C(.dbd.O)C(CH.sub.3).sub.3) A-119 CH.sub.2N(CH.sub.3)(C(.dbd.O)CH.sub.2CH.dbd.CH.sub.2) A-120 CH.sub.2N(CH.sub.3)(C(.dbd.O)OCH.sub.3) A-121 CH.sub.2N(CH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.3) A-122 CH.sub.2N(CH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.2CH.sub.3) A-123 CH.sub.2N(CH.sub.3)(C(.dbd.O)OCH.sub.2CH.dbd.CH.sub.2) A-124 CH.sub.2N(CH.sub.3)(C(.dbd.O)cyclopropyl) A-125 CH.sub.2N(CH.sub.3)(C(.dbd.O)cyclobutyl) A-126 CH.sub.2N(CH.sub.3)(C(.dbd.O)cyclopentyl) A-127 CH.sub.2N(CH.sub.3)(C(.dbd.O)cyclohexyl) A-128 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)H) A-129 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.3) A-130 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.3) A-131 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.2CH.sub.3) A-132 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)CH(CH.sub.3).sub.2) A-133 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)C(CH.sub.3).sub.3) A-134 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.dbd.CH.sub.2) A-135 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.3) A-136 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.3) A-137 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.sub.2CH.sub.3) A-138 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.sub.2) A-139 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)cyclopropyl) A-140 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)cyclobutyl) A-141 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)cyclopentyl) A-142 CH.sub.2N(CH.sub.2CH.sub.3)(C(.dbd.O)cyclohexyl) A-143 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)H) A-144 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.3) A-145 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.3) A-146 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.2CH.s- ub.3) A-147 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH(CH.sub.3).sub.2) A-148 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--C(CH.sub.3).sub.3) A-149 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.dbd.CH.su- b.2) A-150 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.3) A-151 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.sub.3) A-152 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.sub.2CH.- sub.3) A-153 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.s- ub.2) A-154 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclopropyl) A-155 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclobutyl) A-156 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclopentyl) A-157 CH.sub.2N(CH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclohexyl) A-158 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)H) A-159 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)CH.sub.3) A-160 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)CH.sub.2CH.sub.3) A-161 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--CH.sub.2CH.sub.2CH.sub.3) A-162 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)CH(CH.sub.3).sub.2) A-163 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)C(CH.sub.3).sub.3) A-164 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--CH.sub.2CH.dbd.CH.sub.2) A-165 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)OCH.sub.3) A-166 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)OCH.sub.2CH.sub.3) A-167 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--OCH.sub.2CH.sub.2CH.sub.3) A-168 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.sub.2) A-169 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--cyclopropyl) A-170 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)cyclobutyl) A-171 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)--cyclopentyl) A-172 CH.sub.2N(CH(CH.sub.3).sub.2)(C(.dbd.O)cyclohexyl) A-173 CH.sub.2N(OCH.sub.3)(C(.dbd.O)H) A-174 CH.sub.2N(OCH.sub.3)(C(.dbd.O)CH.sub.3) A-175 CH.sub.2N(OCH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.3) A-176 CH.sub.2N(OCH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.2CH.sub.3) A-177 CH.sub.2N(OCH.sub.3)(C(.dbd.O)CH(CH.sub.3).sub.2) A-178 CH.sub.2N(OCH.sub.3)(C(.dbd.O)C(CH.sub.3).sub.3) A-179 CH.sub.2N(OCH.sub.3)(C(.dbd.O)CH.sub.2CH.dbd.CH.sub.2) A-180 CH.sub.2N(OCH.sub.3)(C(.dbd.O)OCH.sub.3) A-181 CH.sub.2N(OCH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.3) A-182 CH.sub.2N(OCH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.2CH.sub.3) A-183 CH.sub.2N(OCH.sub.3)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.sub.2) A-184 CH.sub.2N(OCH.sub.3)(C(.dbd.O)cyclopropyl) A-185 CH.sub.2N(OCH.sub.3)(C(.dbd.O)cyclobutyl) A-186 CH.sub.2N(OCH.sub.3)(C(.dbd.O)cyclopentyl) A-187 CH.sub.2N(OCH.sub.3)(C(.dbd.O)cyclohexyl) A-188 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)H) A-189 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.3) A-190 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.2CH.sub.3) A-191 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.2CH.sub.3) A-192 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)CH(CH.sub.3).sub.2) A-193 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)C(CH.sub.3).sub.3) A-194 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.dbd.CH.sub.2) A-195 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.3) A-196 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.3) A-197 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.sub.2CH.sub.3) A-198 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.sub.2) A-199 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--cyclopropyl) A-200 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)cyclobutyl) A-201 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--cyclopentyl) A-202 CH.sub.2N(OCH.sub.2CH.sub.3)(C(.dbd.O)--cyclohexyl) A-203 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)H) A-204 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)CH.sub.3) A-205 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.3) A-206 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.sub.2CH.- sub.3) A-207 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH(CH.sub.3).sub.2) A-208 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--C(CH.sub.3).sub.3) A-209 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--CH.sub.2CH.dbd.CH.s- ub.2) A-210 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.3) A-211 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.sub.3) A-212 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)OCH.sub.2CH.sub.2CH.s- ub.3) A-213 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--OCH.sub.2CH.dbd.CH.- sub.2) A-214 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclopropyl) A-215 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclobutyl) A-216 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclopentyl) A-217 CH.sub.2N(OCH.sub.2CH.sub.2CH.sub.3)(C(.dbd.O)--cyclohexyl) A-218 CH.sub.2NHC(.dbd.O)NH(CH.sub.3) A-219 CH.sub.2NHC(.dbd.O)NH(CH.sub.2CH.sub.3) A-220 CH.sub.2NHC(.dbd.O)NH(CH.sub.2CH.sub.2CH.sub.3) A-221 CH.sub.2NHC(.dbd.O)NH(CH(CH.sub.3).sub.2) A-222 CH.sub.2NHC(.dbd.O)NH(C(CH.sub.3).sub.3) A-223 CH.sub.2NHC(.dbd.O)NH(CH.sub.2CH.dbd.CH.sub.2) A-224 CH.sub.2NHC(.dbd.O)NH(OCH.sub.3)) A-225 CH.sub.2NHC(.dbd.O)NH(OCH.sub.2CH.sub.3) A-226 CH.sub.2NHC(.dbd.O)NH(OCH.sub.2CH.sub.2CH.sub.3) A-227 CH.sub.2NHC(.dbd.O)NH(OCH.sub.2CH.dbd.CH.sub.2) A-228 CH.sub.2NHC(.dbd.O)NHcyclopropyl A-229 CH.sub.2NHC(.dbd.O)NHcyclobutyl A-230 CH.sub.2NHC(.dbd.O)NHcyclopentyl A-231 CH.sub.2NHC(.dbd.O)NHcyclohexyl A-232 C(.dbd.O)NH.sub.2 A-233 C(.dbd.O)NH(CH.sub.3) A-234 C(.dbd.O)NH(CH.sub.2CH.sub.3) A-235 C(.dbd.O)NH(CH.sub.2CH.sub.2CH.sub.3) A-236 C(.dbd.O)NH(CH(CH.sub.3).sub.2)
A-237 C(.dbd.O)NH(C(CH.sub.3).sub.3) A-238 C(.dbd.O)NH(CH.sub.2CH.dbd.CH.sub.2) A-239 C(.dbd.O)NH(OCH.sub.3) A-240 C(.dbd.O)NH(OCH.sub.2CH.sub.3) A-241 C(.dbd.O)NH(OCH.sub.2CH.sub.2CH.sub.3) A-242 C(.dbd.O)NH(OCH.sub.2CH.dbd.CH.sub.2) A-243 C(.dbd.O)NHcyclopropyl A-244 C(.dbd.O)NHcyclobutyl A-245 C(.dbd.O)NHcyclopentyl A-246 C(.dbd.O)NHcyclohexyl A-247 C(.dbd.S)NH.sub.2 A-248 C(.dbd.S)NH(CH.sub.3) A-249 C(.dbd.S)NH(CH.sub.2CH.sub.3) A-250 C(.dbd.S)NH(CH.sub.2CH.sub.2CH.sub.3) A-251 C(.dbd.S)NH(CH(CH.sub.3).sub.2) A-252 C(.dbd.S)NH(C(CH.sub.3).sub.3) A-253 C(.dbd.S)NH(CH.sub.2CH.dbd.CH.sub.2) A-254 C(.dbd.S)NH(OCH.sub.3) A-255 C(.dbd.S)NH(OCH.sub.2CH.sub.3) A-256 C(.dbd.S)NH(OCH.sub.2CH.sub.2CH.sub.3) A-257 C(.dbd.S)NH(OCH.sub.2CH.dbd.CH.sub.2) A-258 C(.dbd.S)NH(cyclopropyl) A-259 C(.dbd.S)NH(cyclobutyl) A-260 C(.dbd.S)NH(cyclopentyl) A-261 C(.dbd.S)NH(cyclohexyl) A-262 C(.dbd.O)N(CH.sub.3).sub.2 A-263 C(.dbd.O)N(CH.sub.3)(CH.sub.2CH.sub.3) A-264 C(.dbd.O)N(CH.sub.3)(CH.sub.2CH.sub.2CH.sub.3) A-265 C(.dbd.O)N(CH.sub.3)(CH(CH.sub.3).sub.2) A-266 C(.dbd.O)N(CH.sub.3)(C(CH.sub.3).sub.3) A-267 C(.dbd.O)N(CH.sub.3)(OCH.sub.3) A-268 C(.dbd.O)N(CH.sub.3)(OCH.sub.2CH.sub.3) A-269 C(.dbd.O)N(CH.sub.3)(OCH.sub.2CH.sub.2CH.sub.3) A-270 C(.dbd.O)N(CH.sub.3)(OCH.sub.2CH.dbd.CH.sub.2) A-271 C(.dbd.O)N(CH.sub.3)(cyclopropyl) A-272 C(.dbd.O)N(CH.sub.3)(cyclobutyl) A-273 C(.dbd.O)N(CH.sub.3)(cyclopentyl) A-274 C(.dbd.O)N(CH.sub.3)(cyclohexyl) A-275 C(.dbd.S)N(CH.sub.3).sub.2 A-276 C(.dbd.S)N(CH.sub.3)(CH.sub.2CH.sub.3) A-277 C(.dbd.S)N(CH.sub.3)(CH.sub.2CH.sub.2CH.sub.3) A-278 C(.dbd.S)N(CH.sub.3)(CH(CH.sub.3).sub.2) A-279 C(.dbd.S)N(CH.sub.3)(C(CH.sub.3).sub.3) A-280 C(.dbd.S)N(CH.sub.3)(OCH.sub.3) A-281 C(.dbd.S)N(CH.sub.3)(OCH.sub.2CH.sub.3) A-282 C(.dbd.S)N(CH.sub.3)(OCH.sub.2CH.sub.2CH.sub.3) A-283 C(.dbd.S)N(CH.sub.3)(OCH.sub.2CH.dbd.CH.sub.2) A-284 C(.dbd.S)N(CH.sub.3)(cyclopropyl) A-285 C(.dbd.S)N(CH.sub.3)(cyclobutyl) A-286 C(.dbd.S)N(CH.sub.3)(cyclopentyl) A-287 C(.dbd.S)N(CH.sub.3)(cyclohexyl) A-288 ##STR00017## A-289 ##STR00018## A-290 ##STR00019## A-291 ##STR00020## A-292 ##STR00021## A-293 ##STR00022## A-294 ##STR00023## A-295 ##STR00024## A-296 ##STR00025## A-297 ##STR00026## A-298 ##STR00027## A-299 ##STR00028## A-300 ##STR00029## A-301 ##STR00030## A-302 ##STR00031## A-303 ##STR00032## A-304 ##STR00033## A-305 ##STR00034## A-306 ##STR00035## A-307 ##STR00036## A-308 ##STR00037## A-309 ##STR00038## A-310 ##STR00039## A-311 ##STR00040## A-312 ##STR00041## A-313 ##STR00042## A-314 ##STR00043## A-315 ##STR00044##
[0311] In preferred embodiments the present invention relates to the transformation of a compound of formula II with compounds of formula IIa to give compounds of formula I as described herein; and wherein the specific combination of reaction conditions is as defined in embodiments B-1 to B-32 in Table B below. All reactions defined by embodiments B1 to B-32 are carried out with 2 equivalents of trifluoroacetic acid chloride, based on the amount of compound II, with a molar ratio of the base to compound II of 1:1, at atmospheric pressure and whereas the other expressions in embodiments B-1 to B-32 have the following meaning:
[0312] Acylating agent IIa: 2 equivalents of trifluoroacetic acid chloride, based on the amount of compound II (ac1), 2 equivalents of trifluoroacetic acid fluoride, based on the amount of compound II (ac2).
[0313] Base: pyridine (b1), 2,4,6-collidine (b2), 2,6-lutidine (b3), trimethylamine (b4), triethylamin (b5), diisopropylethylamine (b6), sodium acetate (b7), potassium actetate (b8), tributylamine (b9), 2-picoline (b10), 3-picoline (b11), 4-picoline (b12), 5-ethyl-2-methyl-pyridine (b13), sodium carbonate (b14), potassium carbonate (b15).
[0314] Molar ratio of base to compound II: 3:1 (ba1), 1:1 (ba2), 1:50 (ba2).
[0315] Solvents: heptane (s1), cyclohexane (s2), toluene (s3), xylene (s4), dichloromethane (s5), 1,2-dichlorobenzene (s6), chlorobenzene (s7), 1,3,5-trimethylbenzene (s8), ethylbenzene (s9), tetrahydrofurane (s10), dioxane (s11), ethyl acetate (s12), methyl ethylketone (s13), benzotrifluoride (s14).
[0316] Volume ratio of solvent to compound II: 1:2 (sa1), 1:1 (sa2), 5:1 (sa2).
[0317] Pressure: 1 atmosphere (p1), 3 atm (p2), 8 atm (p3), 50 atm (p4).
[0318] Temperature: -10.degree. C. (t1), 0.degree. C. (t2), 50.degree. C. (t3), 100.degree. C. (t4).
[0319] This means, for example, that a reaction, in which the acylating agent IIa is 2 equivalents of TFAC based on the amount of compound II, in the presence of pyridine as a base in a molar ratio of 1:1 based on the amount of compound II, in toluene as solvent and with a volume ratio of toluene to compound II of 1:1, at atmospheric pressure and a temperature of 50.degree. C., is identified as embodiment B-2.
TABLE-US-00003 TABLE B B-1: b1, s3, t2; B-2: b1, s3, t3; B-3: b1, s5, t2; B-4: b1, s5, t3; B-5: b1, s10, t2; B-6: b1, s10, t3; B-7: b1, s12, t2; B-8: b1, s12, t3; B-9: b4, s3, t2; B-10: b4, s3, t3; B-11: b4, s5, t2; B-12: b4, s5, t3; B-13: b4, s10, t2; B-14: b4, s10, t3; B-15: b4, s12, t2; B-16: b4, s12, t3; B-17: b5, s3, t2; B-18: b5, s3, t3; B-19: b5, s5, t2; B-20: b5, s5, t3; B-21: b5, s10, t2; B-22: b5, s10, t3; B-23: b5, s12, t2; B-24: b5, s12, t3; B-25: no base, s3, t2; B-26: no base, s3, t3; B-27: no base, s5, t2; B-28: no base, s5, t3; B-29: no base, s10, t2; B-30: no base, s10, t3; B-31: no base, s12, t2; B-32: no base, s12, t3.
[0320] In further preferred embodiments D-1 to D-32, which are summarized in table D below, the present invention relates to the transformation of a compound selected from the group of compounds II.A-1 to II.A-315 with TFAC to give compounds of formula I; and wherein the specific combination of the reaction conditions in each case is as defined in embodiments B-1 to B-32 of Table B. This means, for example, that a reaction, in which any of compounds II.A-1 to II.A-315 is reacted with TFAC under the conditions as defined in Table B, embodiment B-2, is described in embodiment D-2.
TABLE-US-00004 TABLE D D-1: B-1 & II.A-1 to II-A-315; D-2: B-2 & II.A-1 to II-A-315; D-3: B-3 & II.A-1 to II-A-315; D-4: B-4 & II.A-1 to II-A-315; D-5: B-5 & II.A-1 to II-A-315; D-6: B-6 & II.A-1 to II-A-315; D-7: B-7 & II.A-1 to II-A-315; D-8: B-8 & II.A-1 to II-A-315; D-9: B-9 & II.A-1 to II-A-315; D-10: B-10 & II.A-1 to II- A-315; D-11: B-11 & II.A-1 to II-A-315; D-12: B-12 & II.A-1 to II-A-315; D-13: B-13 & II.A-1 to II- A-315; D-14: B-14 & II.A-1 to II-A-315; D-15: B-15 & II.A-1 to II-A-315; D-16: B-16 & II.A-1 to II- A-315; D-17: B-17 & II.A-1 to II-A-315; D-18: B-18 & II.A-1 to II-A-315; D-19: B-19 & II.A-1 to II- A-315; D-20: B-20 & II.A-1 to II-A-315; D-21: B-21 & II.A-1 to II-A-315; D-22: B-22 & II.A-1 to II- A-315; D-23: B-23 & II.A-1 to II-A-315; D-24: B-24 & II.A-1 to II-A-315; D-25: B-25 & II.A-1 to II- A-315; D-26: B-26 & II.A-1 to II-A-315; D-27: B-27 & II.A-1 to II-A-315; D-28: B-28 & II.A-1 to II- A-315; D-29: B-29 & II.A-1 to II-A-315; D-30: B-30 & II.A-1 to II-A-315; D-31: B-31 & II.A-1 to II- A-315; D-32: B-32 & II.A-1 to II-A-315.
[0321] The invention is illustrated by the following examples:
EXAMPLE 1: PREPARATION OF N-methyl-N-[[4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]phenyl]methyl]cy- clopropanecarboxamide
[0322] 5.0 g (17.6 mmol) N-[[4-[(Z)-N'-hydroxycarbamimidoyl]phenyl]methyl]-N-methyl-cyclopropaneca- rboxamide and 25 g (271 mmol) toluene were placed in a glass reactor and cooled to -22.degree. C. After that 4.6 g (35 mmol) trifluoroacetyl chloride was dosed continuously under vigorous stirring over 10 minutes at -22.degree. C. to the reaction mixture. The reaction mixture was stirred for further 45 minutes at -15.degree. C. to -13.degree. C. Then the reaction mixture was heated up to 25.degree. C. 25 g water was added to the reaction mixture. After phase separation the organic phase was extracted with 20 g water. The solvent of the organic phase was distilled off under reduced pressure (40.degree. C.; 2 kPa). Residue: 3.1 g; HPLC-purity of 76.6%; Yield: 42%.
EXAMPLE 2: PREPARATION OF 2,2-difluoro-N-methyl-2-[4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]phen- yl]acetamide
[0323] 20.0 g (78 mmol) 2,2-difluoro-2-[4-[(Z)-N'-hydroxycarbamimidoyl]phenyl]-N-methyl-acetamide and 200 mL dichloromethane were placed in a pressure reactor and cooled to -10.degree. C. Then 21.5 g (162 mmol) trifluoroacetyl chloride was dosed continuously under vigorous stirring over 10 minutes at -10.degree. C. to the reaction mixture. The reaction mixture was stirred for 4 hours at 100.degree. C. and 990 kPa. Then, after the reactor was cooled down to 25.degree. C., the pressure of the reactor was released and 200 g water was added to the reaction mixture. After phase separation the organic phase was extracted once with 200 g of an aqueous solution of sodium hydroxide (5% by weight) and once with 100 g water. The organic phase was dried over magnesium sulfate. The solvent was the removed under reduced pressure (40.degree. C.; 2 kPa). Residue: 17.5 g; HPLC gave a purity of 76.3%; Yield: 66.3%.
EXAMPLE 3: PREPARATION OF 3-(p-tolyl)-5-(trifluoromethyl)-1,2,4-oxadiazole
[0324] 100 g (0.67 mol) N'-hydroxy-4-methyl-benzamidine were suspended in 450 g (5.22 mol) tetrahydro-2-methylfuran (Me-THF). In total, 203 g (1.53 mol) trifluoroacetyl chloride were dosed in between 0.degree. C. and 25.degree. C. in 5 hours. Then, the volatiles were removed in vacuo (60.degree. C., 250 to 5 mbar) to yield 138 g (>99% HPLC purity, 91% yield) 3-(p-tolyl)-5-(trifluoromethyl)-1,2,4-oxadiazole.
EXAMPLE 4: PREPARATION OF 3-(p-tolyl)-5-(trifluoromethyl)-1,2,4-oxadiazole
[0325] 100 g (0.67 mol) N'-hydroxy-4-methyl-benzamidine were suspended in 450 g (5.22 mol) Me-THF. In total, 203 g (1.53 mol) trifluoroacetylchloride were dosed 40.degree. C. in 3 hours. The volatiles were removed in vacuo (60.degree. C., 10 to 0.5 kPa) to yield 137 g (>98% HPLC purity, 90% yield) 3-(p-tolyl)-5-(trifluoromethyl)-1,2,4-oxadiazole.
EXAMPLE 5: PREPARATION OF N-methyl-4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzamide--Solvent Screening
[0326] 0.1 g (0.517 mmol) N'-hydroxy-4-methyl-benzamidine were suspended in 5 g solvent. Then, 1.13-1.26 g (0.0566 mol) ca. 10-12% solution of trifluoroacetyl chloride in Me-THF or ethyl acetate were added at room temperature and stirred for 1 hour. The table below shows the HPLC % of N-methyl-4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzamide following this procedure and in the presence of several different organic solvents.
TABLE-US-00005 Solvent HPLC % N,N-dimethylformamide 85 NMP 50 Tetrahydro-2-methylfuran 96 Acetonitrile 87 Tetrahydrofurane 92 Methyl tert-butylether 35 Ethyl acetate 85 Me-Imidazolin 99 Acetone 44 Dioxane 73 DME 96 Dichloromethane 41
EXAMPLE 6: PREPARATION OF N-methyl-4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzamide
[0327] 5 g (0.026 mol) N'-hydroxy-4-methyl-benzamidine were suspended in 20 g (0.232 mol) in Me-THF. 62 g (0.0566 mol) ca. 12% solution of trifluoroacetyl chloride in Me-THF were added at room temperature and stirred for 102 hours. After addition of 140 g of a 4% sodium bicarbonate-solution, phases were separated. The organic phase was washed with water and all volatiles were removed in vacuo (up to 80.degree. C. and 0.5 kPa) to yield 6.6 g (>98% HPLC purity, 93% yield) N-methyl-4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzamide.
EXAMPLE 7: PREPARATION OF N-(2-fluorophenyl)-4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzamide
[0328] N-(2-fluorophenyl)-4-[(Z)-N'-hydroxycarbamimidoyl]benzamide (1000 mg, 3.65 mmol) was suspended in Me-THF (10 mL) and trifluoroacetyl chloride (12% solution in Me-THF; 1.9 g, 14.63 mmol) was added dropwise at room temperature and stirred for 10 hours at 60.degree. C. The reaction mass was quenched with water following by adjusting pH to 8 with a 4% sodium bicarbonate solution. The organic phase was separated, washed with water and all volatiles were removed in vacuo (up to 80.degree. C. and 0.5 kPa) to yield 955 mg of the title product.
EXAMPLE 8: PREPARATION OF 4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzoic acid
[0329] 4-[(Z)-N'-hydroxycarbamimidoyl]benzoic acid (500 mg, 3 mmol) was suspended in Me-THF (2 mL) and trifluoroacetyl chloride (12% solution in Me-THF; 6.2 g, 6 mmol) was added dropwise at room temperature and stirred for 20 hours. The reaction mass was quenched with water following by adjusting pH to 8 with a 4% sodium bicarbonate solution. The organic phase was separated, washed with water and all volatiles were removed in vacuo (up to 80.degree. C. and 0.5 kPa) to yield the title product.
EXAMPLE 9: PREPARATION OF N-methyl-L[[4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]phenyl]methyl]met- hanesulfonamide
[0330] N'-hydroxy-4-[[methyl(methylsulfonyl)amino]methyl]benzamidine (500 mg, 2 mmol) was suspended Me-THF (2 mL) and trifluoroacetyl chloride (12% solution in Me-THF; 16.2 g, 14.63 mmol) was added dropwise at room temperature and stirred for 10 hours at 60.degree. C. The reaction mass was quenched with water following by adjusting pH to 8 with a 4% sodium bicarbonate solution. The organic phase was separated, washed with water and all volatiles were removed in vacuo (up to 80.degree. C. and 0.5 kPa) to yie












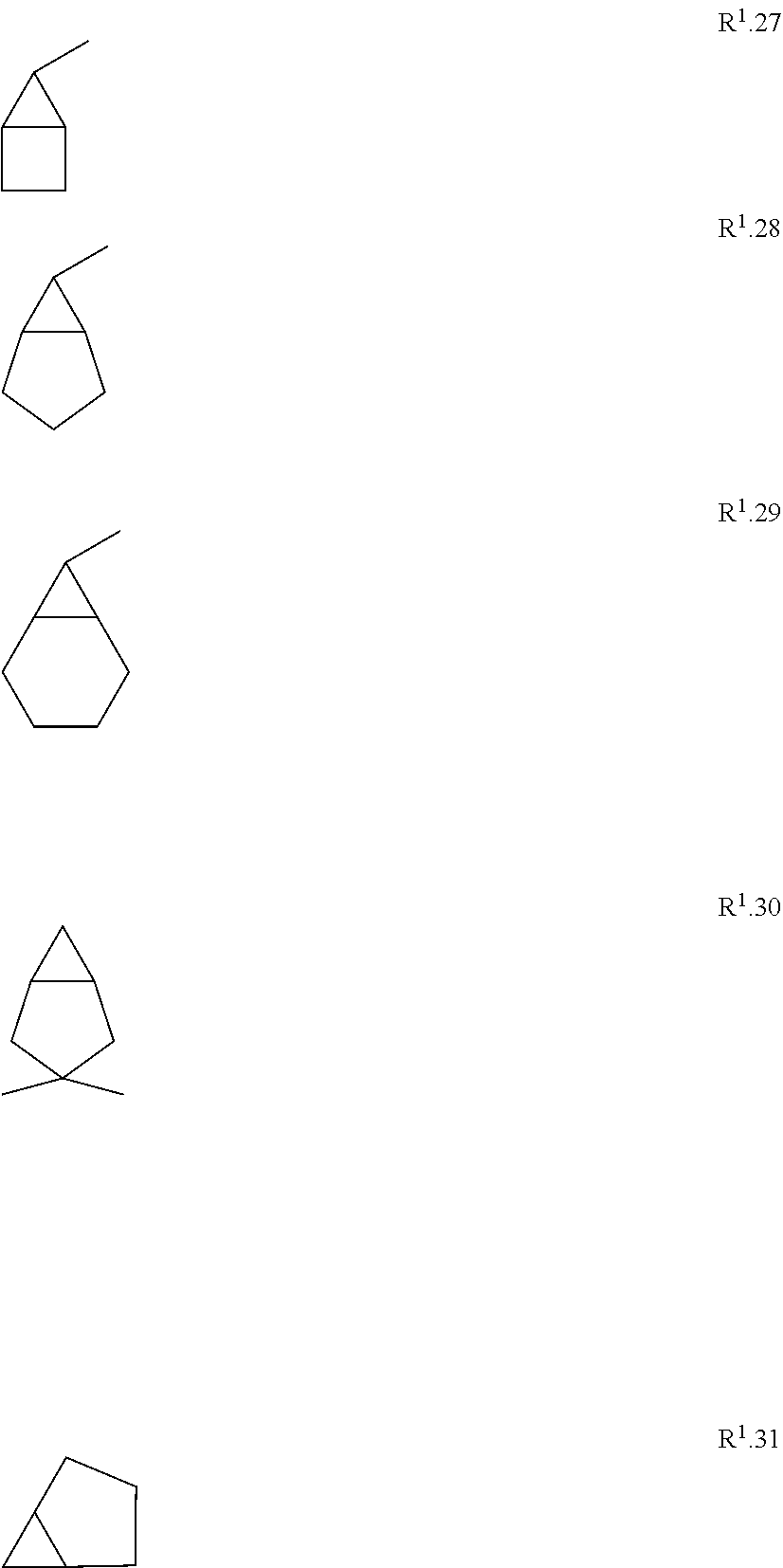
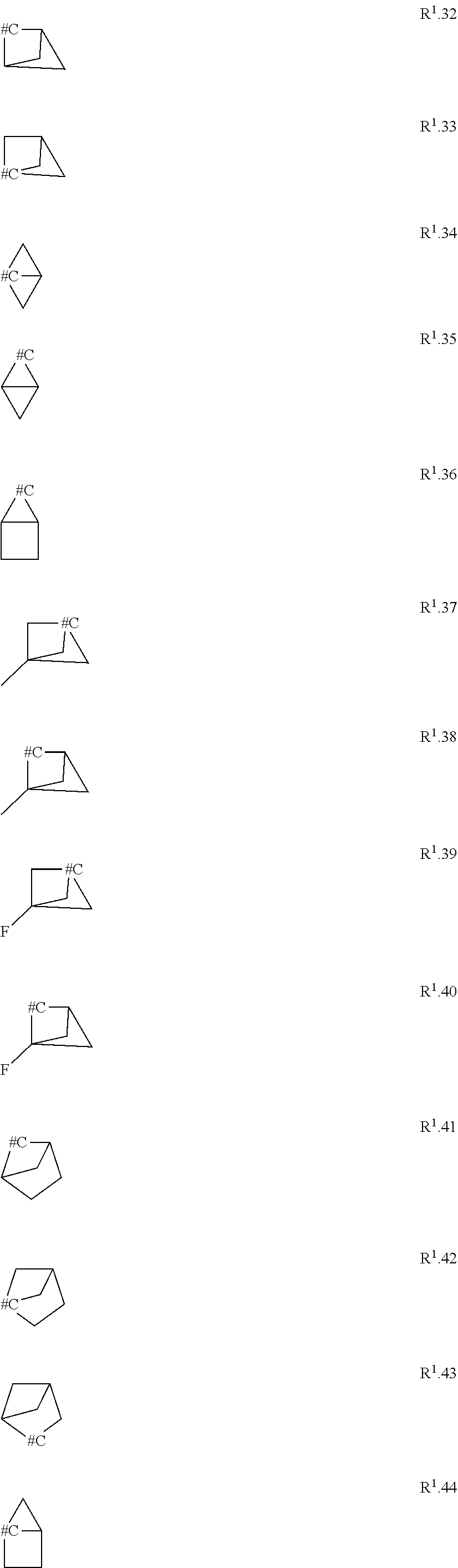

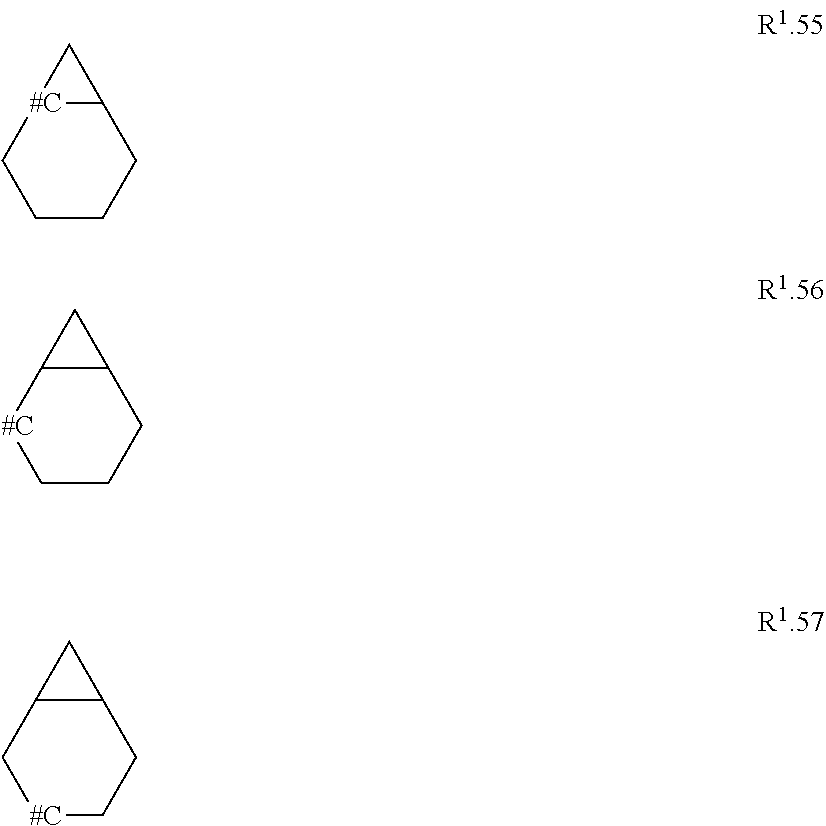

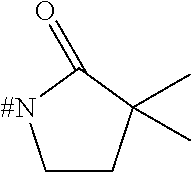
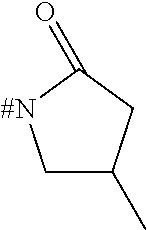
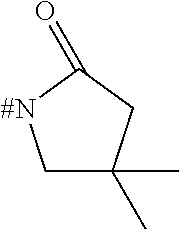




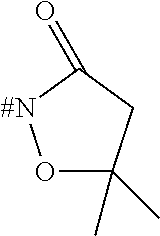

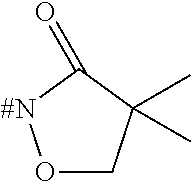







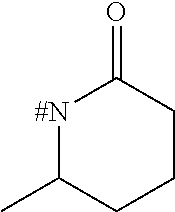
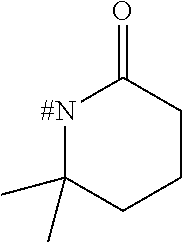






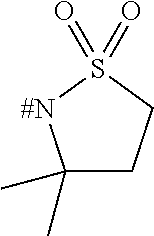
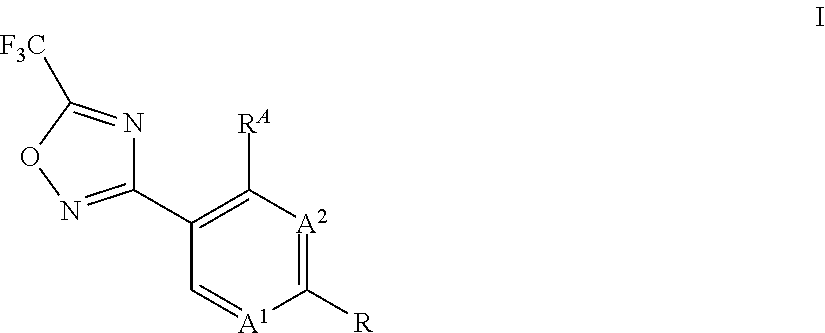



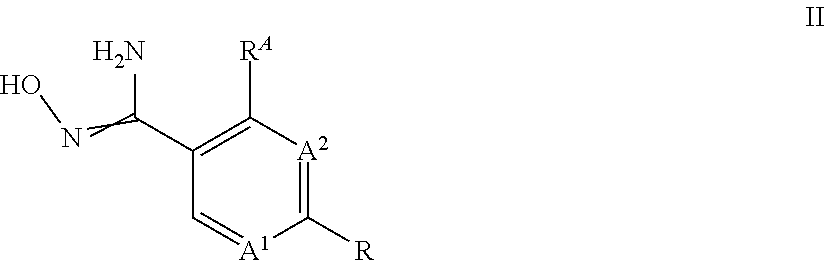



XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.