Cellulose Nanofiber, Sheet-like Material Obtained Therefrom, And Method For Producing Cellulose Nanofiber And Sheet-like Materia
KINUMOTO; Taro
U.S. patent application number 16/649108 was filed with the patent office on 2020-07-16 for cellulose nanofiber, sheet-like material obtained therefrom, and method for producing cellulose nanofiber and sheet-like materia. This patent application is currently assigned to NATIONAL UNIVERSITY CORPORATION OITA UNIVERSITY. The applicant listed for this patent is NATIONAL UNIVERSITY CORPORATION OITA UNIVERSITY. Invention is credited to Taro KINUMOTO.
| Application Number | 20200224365 16/649108 |
| Document ID | / |
| Family ID | 67218533 |
| Filed Date | 2020-07-16 |










View All Diagrams
| United States Patent Application | 20200224365 |
| Kind Code | A1 |
| KINUMOTO; Taro | July 16, 2020 |
CELLULOSE NANOFIBER, SHEET-LIKE MATERIAL OBTAINED THEREFROM, AND METHOD FOR PRODUCING CELLULOSE NANOFIBER AND SHEET-LIKE MATERIAL
Abstract
The present invention provides: a cellulose nanofiber enabling the provision of a high-performance sheet-like material; a method for producing the cellulose nanofiber; and a sheet-like material obtained from the cellulose nanofiber. A bamboo-derived cellulose nanofiber having a cellulose purity of at least 90%, a fiber diameter of 10-20 nm, and a crystallinity of at least 70% can be obtained by a method comprising: (1) a step for subjecting a bamboo material to an alkali treatment and a mechanical treatment to prepare bamboo fibers; (2) a step for delignificating the obtained bamboo fibers; (3) a step for mechanically spreading the delignificated bamboo fibers; (4) a step for removing hemicellulose from the spread bamboo fibers; and (5) a step for removing metal components from the bamboo fibers from which hemicellulose has been removed. A high strength sheet material having a tensile strength of 7-200 N for a basis weight of 10-210 g/m.sup.2 or a high strength sheet material having a tensile strength of 7-200 N for a density of 0.3-1.1 g/cm.sup.3 can be obtained by making this cellulose nanofiber into a sheet.
| Inventors: | KINUMOTO; Taro; (Oita-shi, Oita, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | NATIONAL UNIVERSITY CORPORATION
OITA UNIVERSITY Oita-shi, Oita JP |
||||||||||
| Family ID: | 67218533 | ||||||||||
| Appl. No.: | 16/649108 | ||||||||||
| Filed: | March 12, 2018 | ||||||||||
| PCT Filed: | March 12, 2018 | ||||||||||
| PCT NO: | PCT/JP2018/009537 | ||||||||||
| 371 Date: | March 19, 2020 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | D21B 1/021 20130101; D21B 1/06 20130101; D21H 11/12 20130101; D21H 11/18 20130101; D21H 15/02 20130101 |
| International Class: | D21B 1/02 20060101 D21B001/02; D21H 11/12 20060101 D21H011/12; D21B 1/06 20060101 D21B001/06 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jan 10, 2018 | JP | 2018-002149 |
Claims
1. A bamboo-derived cellulose nanofiber, wherein the bamboo-derived cellulose nanofiber has a cellulose purity of greater than or equal to 90%, a fiber diameter of 10 to 20 nm, and a crystallinity of greater than or equal to 70%.
2. A suspension wherein the bamboo-derived cellulose nanofibers according to claim 1 are dispersed in water.
3. A suspension wherein the bamboo-derived cellulose nanofibers according to claim 1 are dispersed in an organic solvent.
4. A method for manufacturing bamboo-derived cellulose nanofibers, the method comprising the following steps (1) to (5): (1) applying alkali treatment and mechanical treatment to a bamboo material to create bamboo fibers, (2) applying delignification treatment to the obtained bamboo fibers, (3) mechanically defibrating the bamboo fibers having undergone the delignification treatment, (4) removing hemicellulose from the defibrated bamboo fibers, and (5) removing metal components from the bamboo fibers after the removal of hemicellulose.
5. The method according to claim 4, wherein a chipped bamboo material with the inner and outer skins removed is used.
6. The method according to claim 5, wherein the chipped bamboo material has a length of 1 to 10 cm.
7. The method according to claim 4, wherein the alkali treatment of the bamboo material is performed using sodium hydroxide.
8. The method according to claim 4, wherein the mechanical treatment of the bamboo material is performed using a mixer.
9. The method according to claim 4, wherein the delignification treatment of the bamboo fibers is performed using at least one solvent of peracetic acid, chlorous acid, sodium sulfite, sulfuric acid, ozone, an enzyme, and a microorganism (bacteria).
10. The method according to claim 4, wherein the removal of hemicellulose is performed using an aqueous solution of potassium hydroxide.
11. The method according to claim 4, wherein the removal of the metal components is performed using an acidic solution.
12. The method of claim 11, wherein a hydrochloric acid solution is used as the acidic solution.
13. A sheet material comprising bamboo-derived cellulose nanofibers, wherein the sheet material exhibits a tensile strength of 7 to 200 N per basis weight of 10 to 210 g/m.sup.2.
14. A sheet material comprising bamboo-derived cellulose nanofibers, wherein the sheet material exhibits a tensile strength of 7 to 200 N per density of 0.3 to 1.1 g/cm.sup.3.
15. A method for manufacturing a sheet material comprising bamboo-derived cellulose nanofibers, wherein bamboo-derived cellulose nanofibers obtained by the method according to claim 4 are formed into a sheet.
16. The method according to claim 15 wherein the sheet is formed by: (a) creating a suspension comprising bamboo-derived cellulose nanofibers dispersed in water, (b) removing the water from the suspension and collecting the residue thereof, 18 and (c) applying hot press treatment to the collected residue to obtain the sheet material.
17. The method according to claim 16, wherein the sheet is formed by collecting cellulose nanofibers from the suspension of (a), dispersing the collected cellulose nanofibers in an alcohol to make a separate suspension, and applying hot press treatment thereto.
18. The method according to claim 15, wherein the sheet is formed by: (a) creating a suspension comprising cellulose nanofibers dispersed in an alcohol, (b) spreading the suspension on a substrate to form a film, and (c) applying freeze-drying treatment to the film of the suspension to obtain the sheet material.
19. A bamboo-derived lignocellulose nanofiber, which is manufactured by the method according to claim 4, and has a lignin content of 1 to 2 wt %.
Description
FIELD
[0001] The present invention relates to a cellulose nanofiber, or more specifically, a cellulose nanofiber obtained from bamboo as a raw material, a sheet-like material comprising the same, and manufacturing methods thereof. Furthermore, the present invention relates to a nanofiber known as "lignocellulose nanofiber", which includes a small amount of lignin and is obtained by the above manufacturing method.
BACKGROUND
[0002] In recent years, cellulose nanofibers using bamboo as a raw material have been focused on in a wide range of fields, including reinforcement materials for plastics, solar cells and medicine, and sheet-like materials manufactured using these cellulose nanofibers as a raw material are receiving more attention.
[0003] Conventionally, conifer pulp was primarily used as the raw material for cellulose nanofibers. In recent years, in addition to conifers, bamboo has been used as a raw material to manufacture cellulose nanofibers.
[0004] For example, PTL 1 discloses a composite material obtained from bamboo-derived cellulose nanofibers and having excellent conductivity, high tensile strength and high tensile modulus, and a manufacturing method thereof.
[0005] PTL 2 describes cellulose nanofibers with a diameter of about 50 nm, and a manufacturing method thereof, and indicates bamboo as well as various other types of plant material as a cellulose raw material.
[0006] PTL 3 describes a method for manufacturing fine fibrous cellulose, and indicates bamboo as well as various other types of plant material as a cellulose raw material.
[0007] Methods for manufacturing cellulose nanofibers include mechanical defibration methods and chemical defibration methods. Manufacturing cellulose nanofibers by a mechanical defibration method using conifers or bamboo as a raw material tends to result in low crystallinity. Industrial products which use bamboo as a raw material are provided by, for example, Chuetsu Pulp & Paper Co., Ltd. and the manufacture thereof is performed via a mechanical defibration method.
[0008] The cellulose nanofibers known to date feature a purity of, at most, about 87%, a cellulose crystallinity of, at most, about 66%, and an aspect ratio of, at most, about 100. In order to obtain sheet-like materials with higher performance, it is necessary to improve the characteristics of cellulose nanofibers.
PRIOR ART DOCUMENTS
Patent Literatures
[PTL 1] JP 2017-115069 A
[PTL 2] JP 5910504 B2
[PTL 3] JP 2012-012713 A
SUMMARY
Technical Problem
[0009] Out of consideration of the above circumstances, the present invention has an object of providing a cellulose nanofiber that enables provision of a high-performing sheet-like material, a method for manufacturing the same, and a sheet-like material obtained from the cellulose nanofiber. An additional object is to provide a nanofiber known as "lignocellulose nanofiber", which is obtained from the above manufacturing method and contains a small amount of lignin.
Solution to Problem
[0010] The present inventors, through research of cellulose nanofibers using bamboo as a raw material in place of conifer pulp, were able to obtain cellulose nanofibers having a cellulose purity of greater than or equal to 90%, a fiber diameter of about 10 to 20 nm, and a crystallinity of greater than or equal to 70% by performing both a relatively mild mechanical defibration method (using a mixer) and a multi-stage chemical defibration method, thereby completing the present invention.
[0011] More specifically, the bamboo-derived cellulose nanofiber of the present invention is characterized as having a cellulose purity of not less than 90%, a fiber diameter of 10 to 20 nm, and a crystallinity of not less than 70%.
[0012] The bamboo-derived cellulose nanofiber of the present invention can be obtained according to the manufacturing method characterized by comprising the following steps (1) to (5): [0013] (1) applying alkali treatment and mechanical treatment to a bamboo material to create bamboo fibers, [0014] (2) applying delignification treatment to the obtained bamboo fibers, [0015] (3) mechanically defibrating the bamboo fibers having undergone the delignification treatment, [0016] (4) removing hemicellulose from the defibrated bamboo fibers, and [0017] (5) removing metal components from the bamboo fibers after the removal of hemicellulose.
[0018] The sheet-like material comprising the bamboo-derived cellulose nanofibers according to the present invention is characterized by exhibiting a tensile strength of 7 to 200 N per basis weight of 10 to 210 g/cm.sup.2, and a tensile strength of 7 to 200 N per density of 0.3 to 1.1 g/cm.sup.3.
[0019] The sheet-like material comprising the bamboo-derived cellulose nanofibers according to the present invention can be obtained by a manufacturing method in which the bamboo-derived cellulose nanofibers according to the present invention are formed into a sheet.
[0020] The sheet can be formed according to a method of removing the dispersion medium from a suspension of cellulose nanofibers. The method for removing the dispersion medium can be, for example, natural drying, hot press treatment, or freeze-drying. The dispersion medium can be water or an organic solvent, and the organic solvent can be an alcohol, for example.
[0021] In the case of hot press treatment, cellulose nanofibers are formed into a sheet preferably by:
(a) creating a suspension comprising bamboo-derived cellulose nanofibers dispersed in water (b) removing the water from the suspension and collecting the residue thereof, and (c) applying hot press treatment to the collected residue to obtain a sheet-like material.
[0022] The sheet-like material may be obtained by collecting the cellulose nanofibers from the suspension of (a) above, dispersing the collected cellulose nanofibers in an alcohol to make a separate suspension, and applying hot press treatment thereto.
[0023] In the case of freeze-drying, cellulose nanofibers can be formed into a sheet preferably by:
(a) creating a suspension comprising cellulose nanofibers dispersed in an alcohol, (b) spreading the suspension on a substrate to form a film, and (c) applying freeze-drying treatment to the film of the suspension to obtain a sheet-like material.
[0024] Additionally, the bamboo-derived lignocellulose nanofiber according to the present invention has a lignin content of about 1 to 2 wt %, and is obtained by stopping the delignification treatment of the step (2) of the above method for manufacturing bamboo-derived cellulose nanofibers at the time when a predetermined amount of lignin is obtained.
Advantageous Effects of Invention
[0025] The present invention enables improvement in the performance of bamboo-derived cellulose nanofibers and use of a strong sheet-like material using the bamboo-derived cellulose nanofibers as a raw material. Thus, the present invention can be applied to new uses thereof.
[0026] Additionally, the bamboo-derived lignocellulose fibers of the present invention mixed with a resin can be expected to form a composite material (for example, a composite material for automobiles or home appliances) which is more useful than a composite material using high-strength cellulose nanofibers having reduced lignin content.
BRIEF DESCRIPTION OF DRAWINGS
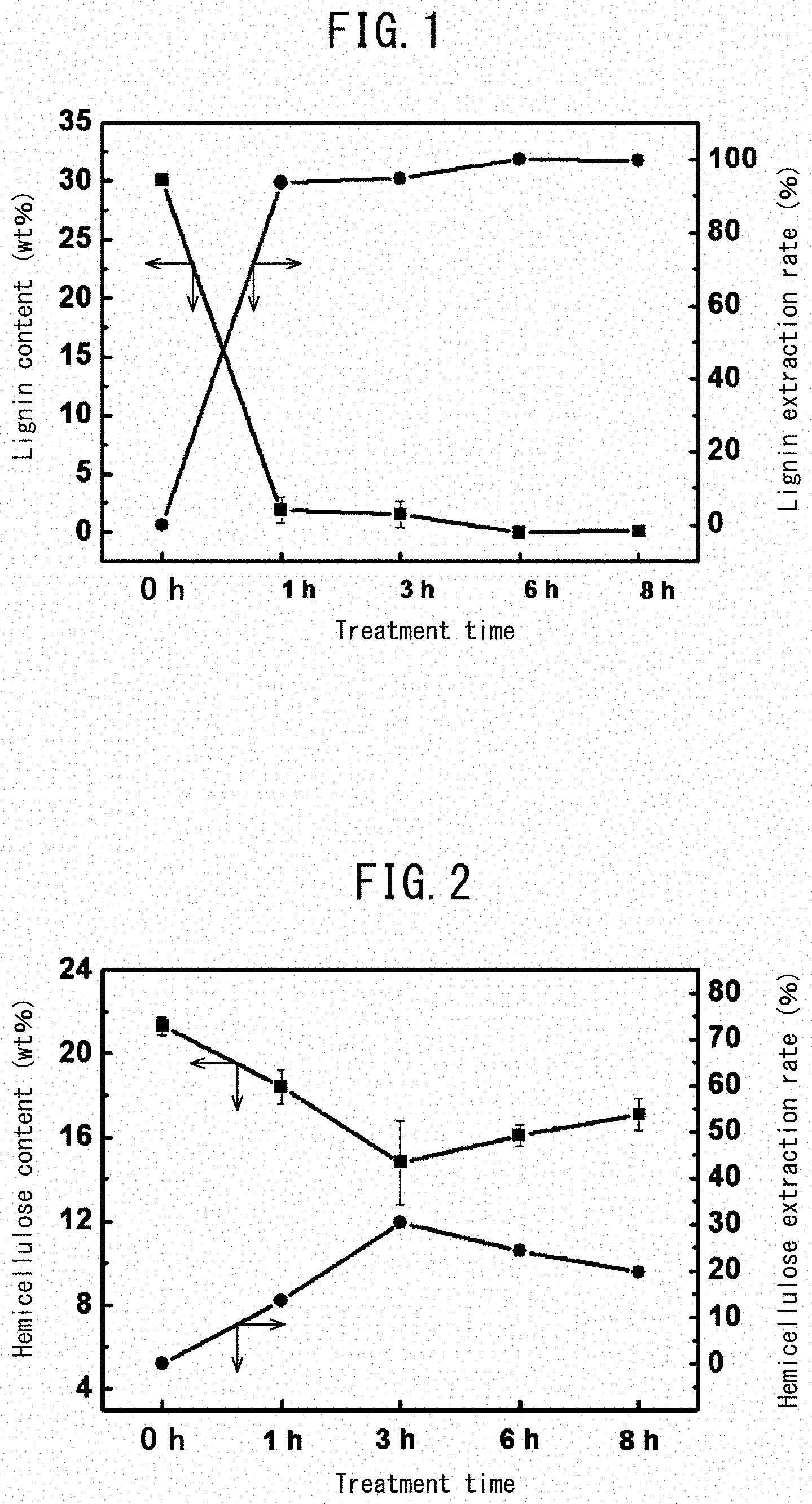
[0027] FIG. 1 is a graph showing the content and extraction rate of bamboo-fiber lignin relative to time of treatment by peracetic acid.
[0028] FIG. 2 is a graph showing the content and extraction rate of bamboo-fiber hemicellulose after the peracetic acid treatment relative to the time of treatment.
[0029] FIG. 3 is a graph showing the relationship between KOH concentration in an aqueous solution and the amount and extraction rate of bamboo-fiber hemicellulose.
[0030] FIG. 4 is a graph showing the relationship between amount of an aqueous KOH solution and the content and extraction rate of bamboo-fiber hemicellulose.
[0031] FIG. 5 is a graph showing the relationship between the treatment temperature for removing hemicellulose and the content and extraction rate of hemicellulose.
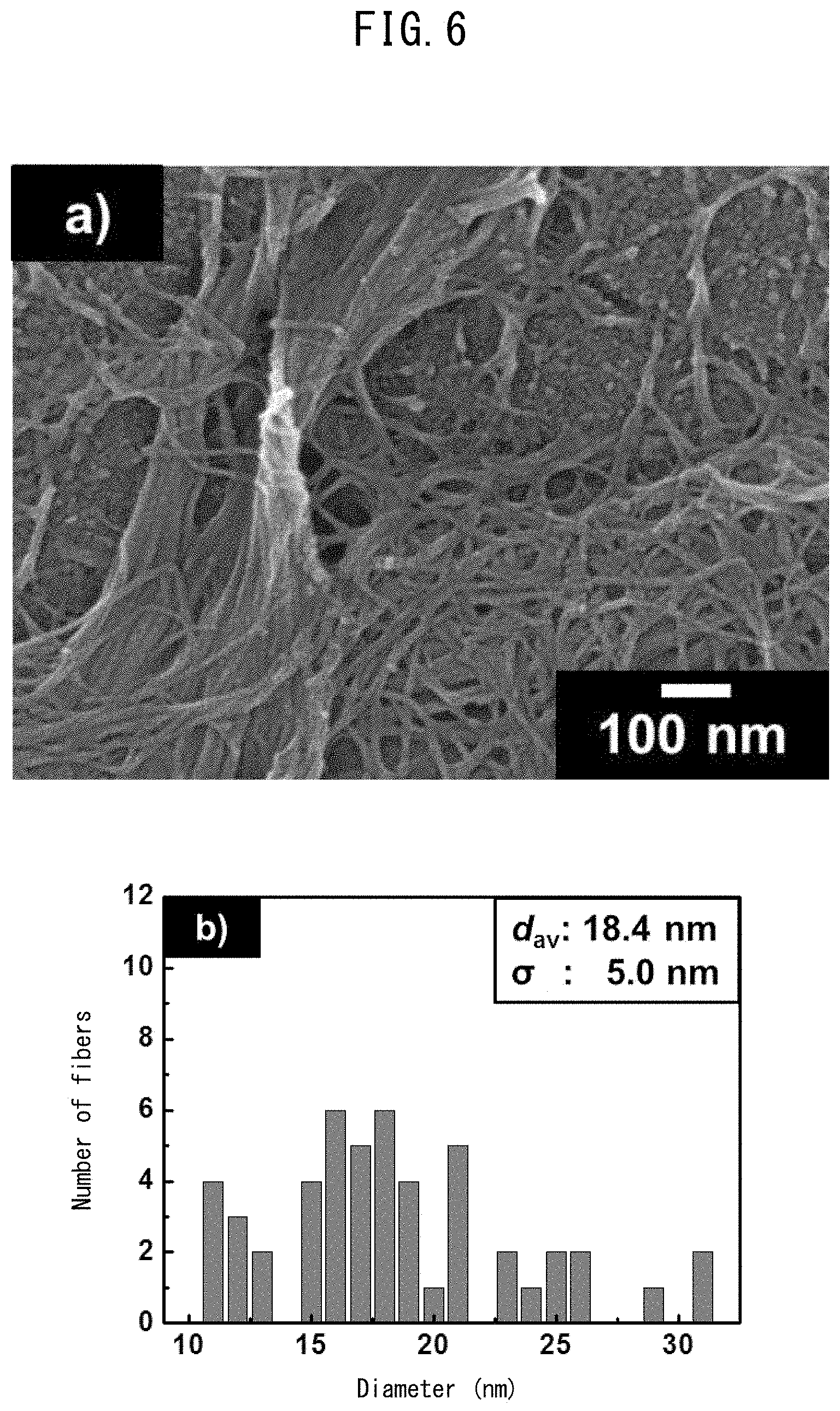
[0032] FIG. 6 shows FE-SEM results for bamboo fibers before delignification treatment with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0033] FIG. 7 shows FE-SEM results for bamboo fibers 1 hour after delignification treatment with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0034] FIG. 8 shows FE-SEM results for bamboo fibers 3 hours after delignification treatment with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0035] FIG. 9 shows FE-SEM results for bamboo fibers 6 hours after delignification treatment with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0036] FIG. 10 shows FE-SEM results for bamboo fibers 8 hours after delignification treatment with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0037] FIG. 11 shows FE-SEM results for bamboo fibers after hemicellulose removal with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0038] FIG. 12 shows FT-IR spectra of cellulose nanofiber sheet according to the present invention.
[0039] FIG. 13 shows electron microscopy results for a cellulose nanofiber sheet created by hot pressing with water as a dispersion medium with (a) a TEM image and (b) an FE-SEM image and the insert showing an external appearance of the sheet.
[0040] FIG. 14 shows electron microscopy results for a cellulose nanofiber sheet created by hot pressing with ethanol as a dispersion medium with (a) a TEM image and (b) an FE-SEM image and the insert showing an external appearance of the sheet.
[0041] FIG. 15 shows electron microscopy results for a cellulose nanofiber sheet created by freeze-drying with (a) a TEM image and (b) an FE-SEM image and the insert showing an external appearance of the sheet.
[0042] FIG. 16 is an XRD pattern of one sample of cellulose nanofiber sheet according to the present invention.
[0043] FIG. 17 is an XRD pattern of another sample of a cellulose nanofiber sheet according to the present invention.
[0044] FIG. 18 is an XRD pattern of yet another sample of a cellulose nanofiber sheet according to the present invention.
[0045] FIG. 19 is a gas desorption-adsorption isotherm of the cellulose nanofiber sheets according to the present invention.
[0046] FIG. 20 is a graph showing the pore diameter distribution for a cellulose nanofiber sheet according to the present invention created by hot pressing with water as a dispersion medium.
[0047] FIG. 21 is a graph showing the pore diameter distribution of a cellulose nanofiber sheet according to the present invention created by hot pressing with ethanol as a dispersion medium.
[0048] FIG. 22 is a graph showing the pore diameter distribution of a cellulose nanofiber sheet according to the present invention created by freeze-drying.
[0049] FIG. 23 is a graph showing the tensile strengths relative to the mass of a cellulose nanofiber sheet according to the present invention created by hot pressing with water as a dispersion medium and of comparative sheets.
[0050] FIG. 24 is a graph showing the tensile strengths relative to the thickness of a cellulose nanofiber sheet according to the present invention created by hot pressing with water as a dispersion medium and of comparative sheets.
[0051] FIG. 25 is a graph showing the tensile strengths relative to the density of a cellulose nanofiber sheet according to the present invention created by hot pressing with water as a dispersion medium and of comparative sheets.
[0052] FIG. 26 is a graph showing the tensile strengths relative to the basis weight of a cellulose nanofiber sheet according to the present invention created by hot pressing with water as a dispersion medium and of comparative sheets.
[0053] FIG. 27 shows the fibers in a cellulose nanofiber sheet according to the present invention with (a) an FE-SEM image and (b) a graph showing fiber distribution.
[0054] FIG. 28 shows the fibers in a sheet created with Celish.TM. with (a) an FE-SEM image and (b) a graph showing fiber distribution.
DESCRIPTION OF EMBODIMENTS
[0055] To obtain a sheet-like material comprising bamboo-derived cellulose nanofibers, it is necessary to manufacture the bamboo-derived cellulose nanofibers as the raw material thereof. According to the present invention, by using a combination of a relatively mild mechanical defibration method (using a mixer) and a multi-stage chemical defibration method, cellulose nanofibers exhibiting improved characteristics relative to conventional cellulose nanofibers can be manufactured.
[0056] More specifically, the method for manufacturing the bamboo-derived cellulose nanofiber according to the present invention includes the following steps (1) to (5): [0057] (1) applying alkali treatment and mechanical treatment to a bamboo material to create bamboo fibers, [0058] (2) applying delignification treatment to the obtained bamboo fibers, [0059] (3) mechanically defibrating the bamboo fibers having undergone the delignification treatment, [0060] (4) removing hemicellulose from the defibrated bamboo fibers, and [0061] (5) removing metal components from the bamboo fibers after the removal of hemicellulose.
[0062] In the step (1) of manufacturing bamboo fibers, the alkali treatment and mechanical treatment are used to create bamboo fibers from a bamboo material.
[0063] The bamboo material used in the present invention is not particularly limited, and can be any plant that includes so-called bamboo fibers, such as moso bamboo, madake bamboo, black bamboo, or shinotake bamboo.
[0064] In order to improve the effect of the alkali treatment and the purity of obtained fibers, it is preferable to remove the inner and outer skins of the bamboo material in advance. With the goal of manufacturing fibers with uniform diameters, it is more preferable to remove the inner and outer skins such that only a portion where the fiber bundles of the bamboo material used are uniform is left.
[0065] It is also preferable to loosen the bamboo material by pressure rolling (pressing treatment) with pinch rollers preset with a peripheral speed difference before performing the alkali solution treatment. By so doing, the infiltration rate of the alkali solution increases and infiltration becomes uniform, so that the separation removal rate of the lignin and the hemicellulose in the subsequent alkali treatment can be increased. For this purpose, it is also possible to, for example, employ a treatment that uses a hydraulic press or a treatment that uses a roller.
[0066] Additionally, since the treatment effect is reduced if the bamboo material is dry when undergoing the treatment using an alkali solution, it is preferable to store the bamboo material in a solution, or in a freezer or refrigerator, without letting the bamboo material dry by the start of the treatment. More preferably, in order to suppress the proliferation of bacteria, the bamboo material can be soaked in a liquid effective for that purpose, such as an aqueous solution of hydrogen peroxide, perchloric acid, or sulfuric acid, and stored in a refrigerator. From the perspective of safety and waste products, it is most preferable to use hydrogen peroxide.
[0067] The bamboo material to undergo alkali treatment is appropriately cut as needed for the volume of the treatment vessel. In order to increase the treatment efficiency, in the present invention, it is preferable to use a chipped bamboo material cut to, for example, approximately 1 to 10 cm in length.
[0068] The alkali treatment can be performed by soaking the bamboo chips in an alkali solution such as an aqueous solution of sodium hydroxide, sodium bicarbonate, or potassium hydroxide. If an aqueous solution of sodium hydroxide is used, from the perspective of efficiency, the concentration of the aqueous solution is preferably 0.01 to 1.00 M, more preferably 0.10 to 1.00 M, or still more preferably 0.10 to 0.50 M. The treatment temperature is preferably 30 to 200.degree. C., more preferably 50 to 150.degree. C., or still more preferably 100 to 150.degree. C. The treatment pressure is preferably 101 to 500 kPa, or more preferably 101 to 200 kPa. The treatment time is preferably 1 to 3 hours, or more preferably 3 hours. The alkali-treated bamboo chips are removed from the alkali solution and washed with water. The washing should continue until the effluent water is neutral.
[0069] Next, in order to obtain bamboo fibers, the bamboo chips are mechanically treated. This treatment can be performed using a general mixer to stir the bamboo chips with water at room temperature. The type of mixer used is not particularly limited as long as it can decompose the bamboo chips into a fibrous state. The treatment conditions can be appropriately determined such that a desired treatment effect is obtained. By drying after the treatment, bamboo fibers are obtained.
[0070] The delignification treatment step (2) can be performed by contacting the bamboo fibers obtained in step (1) with a delignification treatment solution. The delignification treatment solution can be a solution of, for example, acetic acid, chlorous acid, sodium sulfite, sulfuric acid, ozone, an enzyme, or a microorganism (bacteria). After dispersing the bamboo fibers in the delignification treatment solution and then letting the fibers sit, the fibers are separated from the treatment solution, and then washed and dried, whereby delignified bamboo fibers can be obtained. The delignification treatment solution can be used at a temperature of, for example, room temperature to 220.degree. C., or preferably approximately 60 to 100.degree. C. The sitting time is preferably 1 to 8 hours, more preferably 1 to 6 hours, or still more preferably 3 to 6 hours.
[0071] The longer the treatment time, the higher the lignin extraction rate. For example, in the case of using peracetic acid (volume ratio of acetic acid:hydrogen peroxide is 1:1) and a sitting temperature of 80.degree. C., at 6 hours of treatment, the extraction rate was 100% and white fibers were obtained. In this case, almost no hemicellulose was extracted. The longer the treatment time, the smaller the diameter of the bamboo fibers. At 6 hours of treatment, fibers with an average diameter of about 16 nm were obtained.
[0072] The bamboo-derived lignocellulose nanofibers of the present invention, in which the lignin content is 1 to 2 wt %, are obtained by stopping the sitting of bamboo fibers in the delignification treatment solution in the step of delignification treatment (2) at the time when a predetermined lignin content is obtained. In the case above, the time of sitting using peracetic acid at 80.degree. C. as mentioned above can be, for example, about 0.5 to 2 hours or about 0.5 to 1.5 hours. Except for the sitting time in step (2), the bamboo-derived lignocellulose nanofibers of the present invention can be manufactured by the same method as manufacturing the bamboo-derived cellulose nanofibers of the present invention.
[0073] In the case of using peracetic acid as the treatment solution, it is considered that the aromatic ring of lignin undergoes cleavage as shown below, and lignin is removed from bamboo fibers (see Hyoe Hatakeyama, Japan TAPPI Journal, Vol. 20, No. 11, p. 15 (1966)).
##STR00001##
[0075] The mechanical defibration step (3) of the delignified bamboo fibers can be performed by stirring the bamboo fibers with water in a mixer. As long as defibration is not hindered by the mixing, the type of mixer is not particularly limited. From the perspective treatment efficiency, the amount of water is preferably about 10 to 1000 times the mass of the bamboo fibers, more preferably about 100 to 500 times, or still more preferably about 100 to 150 times. The mixing treatment is performed preferably at a temperature of about 5 to 60.degree. C., or more preferably at a temperature of about 5 to 40.degree. C. The operating conditions of the mixer can be appropriately determined so as to achieve a predetermined defibration effect.
[0076] The step (4) of removing hemicellulose from the defibrated bamboo fibers can be performed by alkali treatment of the defibrated bamboo fibers. The alkali treatment can be performed by soaking the defibrated bamboo fibers in an aqueous alkaline solution. The aqueous alkaline solution can be an aqueous potassium hydroxide solution, or can also be an aqueous sodium hydroxide solution or the like. When using an aqueous potassium hydroxide solution, from the perspective of treatment efficiency, for every 5 g of fibers, about 50 to 500 ml or preferably about 200 to 500 ml of a 0.5 to 5.0 M or preferably 1.0 to 2.0 M aqueous KOH solution can be used. The soaking time is preferably 1 to 24 hours, more preferably 1 to 12 hours, or still more preferably 1 to 8 hours.
[0077] The step (5) of removing metal components from the bamboo fibers after hemicellulose removal can be performed by applying acid treatment to bamboo fibers from which hemicellulose has been removed. The acid treatment can be performed by contacting the bamboo fibers with an acid solution and shaking for a predetermined time. The acid solution can be an aqueous solution of hydrochloric acid, perchloric acid, sulfuric acid, or nitric acid. For example, when using a hydrochloric acid solution, the concentration of the solution is preferably about 0.001 to 1.0 M, more preferably about 0.01 to 1.0 M, or still more preferably about 0.01 to 0.1 M. The time of contacting is preferably 1 to 24 hours, more preferably 3 to 24 hours, or still more preferably 1 to 12 hours. The treatment can be performed at room temperature (about 20 to 30.degree. C.).
[0078] The amount of lignin in the bamboo fibers can be measured according to, for example, a sulfuric acid method (The Japan Wood Research Society, Wood Science Experiment Manual, pp. 96-97, Bun-eido Publishing Co., Ltd. (2010)) (refer to the Examples below).
[0079] The amount of hemicellulose in the bamboo fibers can be measured based on the mass of bamboo fibers before and after hemicellulose removal (refer to the Examples below).
[0080] The bamboo-derived cellulose nanofibers manufactured according to the method of the present invention have the following characteristics: a cellulose purity of not less than 90%, a fiber diameter of 10 to 20 nm, and a crystallinity of not less than 70%. The cellulose purity and the crystallinity of the cellulose nanofibers of the present invention are significantly higher than the cellulose purity (at most 87%) and the crystallinity (at most 66%) of conventional cellulose nanofibers.
[0081] By forming the bamboo-derived cellulose nanofibers according to the present invention into a sheet, a sheet-like material according to the present invention can be obtained. The sheet can be formed by, for example, hot pressing or freeze-drying. Natural drying can also be used.
[0082] Forming a sheet by hot pressing can be preferably performed using a suspension obtained by stirring a to-be-treated solution prepared by adding, to water, cellulose nanofibers after removal of metal components. By removing the water as a dispersion medium from the suspension, and, without drying, treating the residue collected with by a hot press machine to form a sheet, a sheet-like material comprising the cellulose nanofibers according to the present invention can be obtained.
[0083] A suspension obtained by re-dispersing in an alcohol, such as ethanol, as a dispersion medium, residue obtained by removing water from a suspension can be used. When the dispersion medium is water, aggregation of fibers is observed in the obtained sheet-like material, and when the dispersion medium is alcohol, disaggregation of fibers is observed due to solvation by the alcohol between cellulose molecules.
[0084] Forming a sheet by freeze-drying can be performed preferably using a suspension in which an organic solvent (an alcohol, for example) is the dispersion medium. By spreading the suspension as a film on a predetermined substrate, freezing it, and then applying freeze-drying treatment, a sheet-like material comprising the cellulose nanofiber according to the present invention can be obtained. If the dispersion medium is an alcohol, the dispersion medium can be ethanol. butanol or the like. If the dispersion medium is an organic solvent other than an alcohol, the dispersion medium can be a ketone (for example, acetone), an aromatic compound (for example, toluene), a carboxylic acid (for example, acetic acid), an amine (for example, N,N-dimethylformamide), or acetonitrile. The dispersion medium (alcohol or the like) sublimes due to the freeze-drying, whereby agglomeration of fibers can be suppressed. The dispersion medium can be one type alone (for example, ethanol), can be a mixture of multiple types, or can be multiple types used sequentially (for example, after collecting bamboo fibers from a suspension of ethanol, re-dispersing the bamboo fibers in butanol to form a suspension and creating a sheet-like material from the resultant suspension). The latter case has the merit of suppressing aggregation of the cellulose nanofibers.
[0085] Regardless of the method of forming a sheet, the stirring to obtain the suspension can be performed, for example, using a general mixer, or using ultrasonic waves. The amount of cellulose nanofiber contained in the suspension is generally 0.1 to 10 wt %, more preferably 0.1 to 2.0 wt %, or even more preferably 0.1 to 1.0 wt %. The stirring conditions are not particularly limited, as long as a suspension in which cellulose nanofibers are sufficiently dispersed is obtained. Removal of the water or alcohol of the dispersion medium can be performed by any treatment such as filtering.
[0086] Natural drying can be performed by dispersing the bamboo-derived cellulose nanofiber, spreading the suspension into a film over a substrate, and then letting the suspension sit such that the dispersion medium is removed. The dispersion medium can be water or an organic solvent such as an alcohol. In some cases, the removal of dispersion medium can be accelerated by ventilation.
[0087] The sheet-like material created from the bamboo-derived cellulose nanofiber of the present invention demonstrates improved strength over a sheet-like material created with conventional cellulose nanofiber, when measuring the strength under the same conditions. For example, comparing tensile strengths relative to 200 g/m.sup.2 of basis weight (mass per m.sup.2 of sheet material), the tensile strength of the sheet material according to the present invention is about 200 N, whereas the tensile strengths of a sheet material created from cellulose fibers FD100G obtained from Daicel FineChem Ltd. and commercially available paper (ISO9707-certified paper) obtained from Mondi Limited are about 100 N and 145 N, respectively.
[0088] Thus, the sheet-like material comprising bamboo-derived cellulose nanofibers of the present invention, which demonstrates such a high tensile strength, is expected to be useful in fields such as reinforcement, acoustics, medical treatment, food products, packaging materials, transport and the like.
EXAMPLES
[0089] The present invention will now be further described by way of Examples. Naturally, the present invention is not limited to the following Examples.
[0090] 1. Preparation of Bamboo Fibers in Nanometer Size
[0091] The inner and outer skins were removed, and pressing treatment was applied, to obtain chipped bamboo pieces with a length of about 10 cm. 120 g of the bamboo pieces were placed in an electric pressure cooker (Panasonic, SR-P37-N), soaked in 2 L of a 0.10 M aqueous sodium hydroxide solution, and treated for 3 hours under conditions of 120.degree. C. at 200 kPa. After the treated bamboo pieces were allowed to cool, the bamboo pieces were placed in a metallic sieve and washed with ultrapure water until the effluent water became neutral, whereby the bamboo fibers were obtained. 60 g of the obtained bamboo fibers were placed in a mixer (Vitamix.TM. ABS-BU), 1 L of ultrapure water was added, and the mixture was stirred for 1 minute at 37,000 rpm. Thereafter, the ultrapure water was removed, and the fibers were dried to obtain bamboo fibers.
[0092] 2. Delignification Treatment
[0093] In a 300 ml glass Erlenmeyer flask, a 17.5 M acetic acid solution was added, and a 11.6 M hydrogen peroxide solution was slowly dripped therein using a separatory funnel to make 100 ml of a peracetic acid solution. The volume ratio of acetic acid to hydrogen peroxide was set to 1:1.
[0094] 10 g of the bamboo fibers obtained in above item 1 were added, in a batch-wise manner, about 1 g at a time with mixing by a glass rod into a container containing 100 ml of a peracetic acid solution, and thereafter the temperature of a water bath (EYELA, SB-350) was set to 80.degree. C., the cooling pipe which was connected to the container was connected to a low temperature water bath (EYELA, NCB-1200) and let sit for 1, 3, 6, or 8 hours while under reflux. Thereafter, the container was allowed to cool, and suction filtering was performed using plastic filters (ADVANTEC, KP-47H and KP-47S). The residue was washed with ultrapure water until the effluent water became neutral, and thereafter was dried for 12 hours in a dryer kept at 60.degree. C., whereby delignified bamboo fibers were obtained.
[0095] After 1 hour of treatment in the peracetic acid, the fiber changed from a brown color to a yellow color, and after 3 hours of treatment, the color was yellow-white. After 6 hours of further treatment, white-colored bamboo fibers were obtained. This is considered to correspond to the removal of the colored component lignin as the peracetic treatment progresses. No changes were observed after 6 or more hours of treatment.
[0096] 3. Defibration
[0097] Delignified bamboo fibers were added to ultrapure water to make its concentration of 0.7 wt %, and the mixture was stirred for 5 minutes at 37,000 rpm using a mixer (Vitamix.TM. ABS-BU). Thereafter, the mixture was allowed to cool, and then mixed intermittently for a total of 60 minutes, whereby a suspension of defibrated bamboo fibers was obtained.
[0098] 4. Quantitative Determination of Lignin
[0099] 15 ml of 13.4 M sulfuric acid and 1 g of the product (delignified bamboo fibers) obtained from above item 2 were added into a 100 ml glass beaker, and the mixture was stirred with a glass rod until the sulfuric acid evenly infiltrated the fibers. After letting the mixture sit for 4 hours, it was boiled for 4 hours under reflux and then allowed to cool. Thereafter, the residue was collected after suction filtering using a glass filter (Sibata Scientific Technology, 1GP16), and was washed with 500 ml of hot water and then dried for 12 hours in a dryer kept at 105.degree. C. After drying, the yield was quantitatively measured to 4 decimal places, and the content of lignin was found using the following equation (1) (see The Japan Wood Research Society, Wood Science Experiment Manual, p. 97, Bun-eido Publishing Co., Ltd. (2010)).
Lignin content (wt %)=(mass after experiment/mass before experiment).times.100 (1)
[0100] The content and extraction rate of lignin relative to treatment time in peracetic acid are shown in Table 1, and the graph thereof is shown in FIG. 1. The longer the time of peracetic acid treatment, the larger the extraction rate, such that at 6 hours, the entire amount of lignin was extracted.
TABLE-US-00001 TABLE 1 Extrac- Peracetic Lignin content (wt %) tion acid Sample Sample Sample Aver- Standard rate treatment 1 2 3 age deviation (%) Before 30.7 30.1 29.6 30.1 0.45 -- treatment 1 hour 0.94 1.17 1.21 1.11 0.12 96.3 3 hours 0 0 0 0 0 100 6 hours 0 0 0 0 0 100 8 hours 0.23 0 0 0.08 0.11 99.7
[0101] In the case of obtaining nanofibers including a small amount of residual lignin, which is known as so-called "lignocellulose nanofibers", bamboo fibers (including about 1 wt % of residual lignin) obtained after about 1 hour of treatment can be further treated in accordance with the procedures explained below.
[0102] 5. Quantitative Determination of Hemicellulose
[0103] According to a reference literature, .beta.-cellulose, .gamma.-cellulose, and hemicellulose are categorized as hemicellulose, and all others as .alpha.-cellulose (see The Japan Wood Research Society, Wood Science Experiment Manual, p. 95, Bun-eido Publishing (2010)). In the present invention, accordingly, the quantification method for .alpha.-cellulose was used to measure hemicellulose (see The Japan Wood Research Society, Wood Science Experiment Manual, pp. 96-97, Bun-eido Publishing Co., Ltd. (2010)). Therefore, the hemicellulose here includes .beta.- and .gamma.-celluloses.
[0104] 25 ml of a 5.80 M aqueous sodium hydroxide solution and 1 g of the product (delignified bamboo fibers) obtained from above item 2 were added into a 200 ml plastic beaker. Once the liquid evenly infiltrated the fibers, the mixture was let sit for 4 minutes, then stirred using a plastic stirring rod for 5 minutes, and let sit for 30 minutes. Ultrapure water was added to the beaker and stirred for 1 minute, and then let sit for 5 minutes. Thereafter, suction filtering was performed using a glass filter (Sibata Scientific Technology, 1GP250), the filtrate was collected and re-filtered, and then the residue was washed with ultrapure water until the filtrate was neutral. The residue and 40 ml of 1.75 M aqueous acetic acid solution were added to a 100 ml glass beaker, and then let sit for 5 minutes. The residue was then collected by suction filtering, and washed with 1 L of ultrapure water. Thereafter, the residue was dried for 12 hours in a dryer kept at 105.degree. C., and quantitatively measured to 4 decimal places. The content of hemicellulose was found using the following equation (2) (see The Japan Wood Research Society, Wood Science Experiment Manual, p. 96, Bun-eido Publishing Co., Ltd. (2010)).
Hemicellulose content (wt %)=((mass before experiment-(mass of .alpha.-cellulose))/mass before experiment).times.100 (2)
[0105] The content and extraction rate of the hemicellulose of the bamboo fibers after peracetic acid treatment are shown in Table 2, and the graph thereof is shown in FIG. 2. The hemicellulose content did not greatly decrease in the peracetic acid treatment. Additionally, no great changes due to treatment time were observed.
TABLE-US-00002 TABLE 2 Extrac- Peracetic Hemicellulose content (wt %) tion acid Sample Sample Sample Aver- Standard rate treatment 1 2 3 age deviation (%) Before 20.8 21.8 21.3 21.3 0.41 -- treatment 1 hour 19.2 17.3 18.7 18.4 0.80 13.6 3 hours 13.6 13.2 17.6 14.8 1.99 30.5 6 hours 15.6 15.8 16.8 16.1 0.52 24.4 8 hours 17.8 16.0 17.3 17.1 0.76 19.7
[0106] 6. Removal and Quantitative Determination of Hemicellulose
[0107] 5 g of bamboo fibers which had undergone delignification treatment for 6 hours using peracetic acid and defibration treatment were placed in a 200 ml glass Erlenmeyer flask, to which 200 ml of a 0.71 M or 1.18 M aqueous potassium hydroxide solution was added. The solution was allowed to evenly infiltrate the fibers. The flask was sealed with a stopper, and let sit at room temperature for 12 hours. Then the solution underwent suction filtering using a plastic filter (ADVANTEC, KP-47H and KP-47S), and the residue was washed using ultrapure water until the effluent water became neutral. Thereafter, the residue was dried for 12 hours in a dryer kept at 60.degree. C., and the content of hemicellulose was measured using the same procedure and formula as in above item 5. The results are shown in Table 3 and the relationships between treatment solution concentration and hemicellulose content and extraction rate are shown in FIG. 3. With a 1.18 M solution, about 7% hemicellulose was included. The content of .alpha.-cellulose was about 93%.
TABLE-US-00003 TABLE 3 Extrac- Hemicellulose content (wt %) tion KOH Sample Sample Sample Aver- Standard rate treatment 1 2 3 age deviation (%) Before 15.6 15.8 16.8 16.1 0.52 -- treatment 0.71 M 17.9 18.1 17.0 17.7 0.48 16.9 1.18 M 7.37 6.67 7.24 7.09 0.30 66.7
[0108] In order to clarify the relationship between the volume at the time of treatment with potassium hydroxide and extraction rate, while the concentration in the aqueous KOH solution was kept at 1.18 M, the volume of the solution was changed. The quantitative results for the cases of using 100 ml and 200 ml of solution are shown in Table 4. Additionally, the relationship between the solution volume and the content and extraction rate of hemicellulose is shown in FIG. 4. From the results, it is clear that the most hemicellulose was extracted when 200 ml of a 1.18 M aqueous KOH solution was used for 5 g of bamboo fibers.
TABLE-US-00004 TABLE 4 Extrac- Content (wt %) tion KOH Sample Sample Sample Aver- Standard rate treatment 1 2 3 age deviation (%) Before 15.6 15.8 16.8 16.1 0.52 -- treatment 100 ml 11.3 12.3 11.6 11.7 0.42 44.1 200 ml 7.37 6.67 7.24 7.09 0.30 66.7
[0109] Next, the relationship between treatment temperature and the content and extraction rate of hemicellulose was investigated. The measured results are shown in Table 5 and FIG. 5. When the aqueous KOH solution concentration was 1.18 M and 200 ml of a solution was used for 5 g of bamboo fibers, more hemicellulose was extracted when treated at temperatures higher than 100.degree. C. When the treatment temperature was 100.degree. C., the bamboo fibers and an aqueous KOH solution were added into a Teflon.TM. container. The container was placed in a heat-resistant stainless steel container, which was then sealed. The dryer temperature was set to 100.degree. C., and the contents were left to sit for 12 hours. After allowing the containers to be cooled, the contents were suction filtered, washed and dried as described above.
TABLE-US-00005 TABLE 5 Extrac- Content (wt %) tion Treatment Sample Sample Sample Aver- Standard rate temperature 1 2 3 age deviation (%) Before 15.6 15.8 16.8 16.1 0.52 -- treatment Room 7.37 6.67 7.24 7.09 0.30 66.7 temperature 100.degree. C. 11.3 11.2 11.3 11.3 0.05 46.9
[0110] 7. Quantitative Determination of .beta.-Cellulose
[0111] The content of .alpha.-cellulose in the product treated in the optimal conditions is about 93%.
[0112] In order to accurately find the amount of .beta.-cellulose in the product, the product was added to 10 ml of a 30% acetic acid solution, to which 200 ml of the washing liquid obtained in above item 5 was added. Then the solution was heated to 80.degree. C., kept at that temperature, and left to sit for 9 hours. The obtained precipitate was collected in pre-weighed filter paper. The increase in mass after drying was considered to be the content of .beta.-cellulose (see The Japan Wood Research Society, Wood Science Experiment Manual, p. 96, Bun-eido Publishing Co., Ltd. (2010)).
TABLE-US-00006 TABLE 6 Content (wt %) Sample 1 Sample 2 Sample 3 Average Hemicellulose 7.37 6.67 7.24 7.09 .beta.-cellulose 7.3 6.4 7.0 6.9 .alpha.-cellulose 92.2 93.6 92.8 92.9 Total of .beta.- and 99.5 100 99.8 99.8 .alpha.- celluloses
[0113] From the results in the third and fourth rows from the top of Table 6, it was found that 97% of the hemicellulose measured by the method described in above item 5 is .beta.-cellulose. Combined with the total for .alpha.-cellulose content, the cellulose content was confirmed to be 99.8%.
[0114] 8. Morphological Observation Via Field Emission Scanning Electron Microscope
[0115] 1 mL of ethanol was added to one drop of suspension of the bamboo fibers (before delignification) obtained in above item 1, and then the fibers were ultrasonically dispersed. 10 .mu.L of the suspension after ultrasonic dispersion was dripped onto glassy carbon, and dried in a dryer kept at 60.degree. C. Thereafter, platinum was vapor deposited onto the dried bamboo fibers using a vapor deposition apparatus (JEOL Ltd., JFC-1600), and then the morphology of the bamboo fibers was observed via a field emission scanning electron microscope (FE-SEM (JEOL Ltd., JSM-6701F)). The vapor deposition conditions are shown in Table 7 and the measurement conditions are shown in Table 8. Additionally, the bamboo fibers which underwent the delignification treatment described in above item 2 for 1, 3, 6, and 8 hours were likewise observed. The respective observation results are shown in FIGS. 6 (a) to 10 (a) (FE-SEM images) and FIGS. 6 (b) to 10 (b) (fiber distribution).
TABLE-US-00007 TABLE 7 Sputtering current 20 mA Vapor deposition time 15 s Number of vapor deposition events 2
TABLE-US-00008 TABLE 8 Acceleration voltage 3 kV Working distance 3 mm Irradiation current 7 .mu.A Emission current 10 .mu.A
[0116] When observing at high magnification using an FE-SEM, it was found that the short fiber (not shown) having a diameter of about 16 .mu.m as confirmed by optical microscope observation of bamboo fibers before delignification treatment was a bundle of tangled fibers having diameters within a wide range (FIGS. 6 (a) and (b)). The longer the delignification treatment time, the smaller the diameter of fibers. The particulate substance seen in the FE-SEM image is vapor deposited platinum.
[0117] After 8 hours, the fiber diameters were 15.9 nm on average. In general, the diameter of the cellulose nanofibers contained in wood material or the like is several nanometers, and therefore the above fiber diameters were somewhat thicker than that. This is because samples for FE-SEM observation must be completely dry, and there was a possibility that during the drying, hydrogen bonding between the cellulose molecules led to association of the molecules.
[0118] Next, similar observation via FE-SEM was performed for bamboo fibers (bamboo fibers treated using 1.18 M aqueous potassium hydroxide solution indicated in above item 6) from which hemicellulose as well as lignin was removed. The results are shown in FIG. 11 (a) (FE-SEM image) and FIG. 11 (b) (fiber distribution). Even for fibers from which hemicellulose was removed, a portion of bundles remained, but the average fiber diameter was small, and it is likely that hemicellulose extraction is related to the diameter of cellulose nonafibers produced.
[0119] 9. Qualitative Analysis Via Fourier Transform Infrared Spectroscopy
[0120] Respective bamboo fibers after peracetic acid treatment (delignification treatment) for 1, 3, 6, and 8 hours and bamboo fibers for which hemicellulose removal was performed after 8 hours of peracetic acid treatment were placed in a mixer (Vitamix.TM. ABS-BU), ultrapure water was added, and the mixture was stirred for 60 minutes. Thereafter, the mixture was suction filtered using a plastic filter (ADVANTEC, KP-47H and KP-47S), and dried in a dryer kept at 60.degree. C. An FT-IR spectra in the range of 4000 to 550 cm.sup.-1 was measured for the obtained bamboo fiber sheet using a Fourier transform infrared spectrometer (FT-IR (Thermo Fisher SCIENTIFIC, ART iD5)) equipped with a diffuse reflection unit. The results are shown in FIG. 12.
[0121] The peaks of the CO stretch at 1760 cm.sup.-1, aromatic C.dbd.C stretch at 1500 cm.sup.-1, CO antisymmetric stretch of a methoxy group at 1250 cm.sup.-1, and aromatic CH stretch at 840 cm.sup.-1, which are attributable to lignin confirmed in the FT-IR spectra (not shown) of the raw material bamboo fibers before peracetic acid treatment (delignification treatment), were not observed, but the peaks of the OH stretch at 3600 to 3000 cm.sup.-1 and the CH stretch at 2920 cm.sup.-1 which are attributable to cellulose and hemicellulose were confirmed.
[0122] 10. Removal of Metal Components and Qualitative and Quantitative Analysis of Metals Via ICP Emission Spectroscopy
[0123] The bamboo fibers obtained in above item 2 (bamboo fibers with the lignin removed) were contacted with an aqueous hydrochloric acid solution, to thereby remove metal components. 1 g of bamboo fibers and 50 ml of a 0.01 M aqueous hydrochloric acid solution were added into a plastic sample tube. While leaving the bamboo fibers behind, the solution was quickly extracted from the sample tube to obtain solution "before treatment". 50 ml of an aqueous hydrochloric acid solution was newly added to the sample tube, and after 24 hours of shaking the tube, the solution was quickly extracted leaving the bamboo fibers behind to obtain solution "after treatment". Each of the solutions was added into a 10 ml glass screw top test tube, treated with a centrifuge (AS ONE Corporation, C-12B) such that the solid fraction precipitated, and then the supernatant liquid was removed. The supernatant liquid was analyzed using an inductively coupled plasma optical emission spectrometer (ICP-OES (Agilent Technologies, 710 ICP-OES)), and qualitative and quantitative analysis was performed on the metals contained in the bamboo fiber. The results are shown in Table 9.
TABLE-US-00009 TABLE 9 Content (%) Element Al Ba K Mg Mn Zn Before 0 0 0.356 0 0 0.014 treatment After 0 0 0.005 0 0 0.054 treatment
[0124] Bamboo is known to contain mainly silica (silicon oxide), calcium, potassium, magnesium, and sodium in large amounts. In the bamboo fiber of the present invention analyzed here, potassium and zinc were confirmed to be present, but other metals were present in extremely minor amounts at nearly 0%. It was found that according to the present invention, metals can be removed by soaking the fibers in hydrochloric acid for 24 hours, and the content of metals can be reduced to about 0.06% relative to the mass of obtained cellulose nanofiber.
[0125] 11. Creation of Cellulose Nanofiber Sheet by Hot Pressing
[0126] 3.5 g of bamboo fibers reacted with peracetic acid solution for 6 hours in the procedure explained above in above item 2 were added to 500 ml of ultrapure water to make a fiber content of 0.7 wt %, the solution was stirred for 5 minutes at 37,000 rpm using a mixer (Vitamix.TM. ABS-BU), and after allowing it to be cooled, the solution was stirred intermittently for a total of 60 minutes to obtain a suspension. The obtained suspension was suction filtered using a glass filter (ADVANTEC, KG-47), and thereafter, without drying, was pressed at 120.degree. C. using a small heat press machine (AS ONE Corporation, AH-2003) to create a sheet. Additionally, after filtering the suspension obtained above, the residue was added to 100 ml of ethanol, ultrasonically dispersed and then suction filtered. After this was repeated two times, a sheet was created using the small heat press machine in a similar method.
[0127] 12. Creation of Cellulose Nanofiber Sheet by Freeze-Drying
[0128] After filtering the suspension obtained in above item 11, the suspension was added to 50 ml of ethanol, ultrasonically dispersed, and suction filtered. After this was repeated 2 times, the residue was added to 50 ml of t-butyl alcohol and ultrasonically dispersed. This process was also repeated 2 times, and after filtering, the residue was transferred to a petri dish, frozen in a freezer (Panasonic, NR-B175W), and dried using vacuum freeze-drying equipment (Hitachi Corp., ES-2030) to create a sheet. The drying conditions are shown in Table 10. For the sake of comparing morphology, a water suspension was also frozen directly in the freezer and freeze-dried to create a sheet.
TABLE-US-00010 TABLE 10 Setting temperature -10.degree. C. Degree of vacuum at completion of drying Less than 0.1 Torr (13.3 Pa)
[0129] 13. Observation of Morphology of Sheets Via Field Emission Scanning Electron Microscope and Transmission Electron Microscope
[0130] The morphology of the obtained cellulose nanofiber sheet was observed via a field emission scanning electron microscope (FE-SEM). The measurement conditions are shown in Table 11. Platinum was vapor deposited on the sheet using a vapor deposition apparatus (JEOL Ltd., JFC-1600) before observation. The vapor deposition conditions are shown in Table 12.
TABLE-US-00011 TABLE 11 Acceleration voltage 3 kV Working distance 3 mm Irradiation current 7 Emission current 10 .mu.A
TABLE-US-00012 TABLE 12 Sputtering current 20 mA Vapor deposition time 15 s Number of vapor deposition events 2
[0131] Additionally, the created sheet was ultrasonically dispersed in 1-butanol. The dispersion was dripped onto a TEM grid (Okenshoji Co., Ltd., STEM 150 Cu grid), dried by a dryer kept at 100.degree. C., and then the sheet morphology was observed via a transmission electron microscope (TEM) (JEOL Ltd., JEM-2100).
[0132] FIG. 13 (a) shows a TEM image of a cellulose nanofiber sheet created using a hot press with water as a dispersion medium, FIG. 13 (b) shows a picture of the external appearance and an FE-SEM image thereof, and FIGS. 14 (a) and (b) show the respective images for a sheet created using a hot press with ethanol as a dispersion medium.
[0133] In the case of water as the dispersion medium, it is confirmed that the fibers aggregate, which is considered to be due to the strong hydrogen bonds between cellulose molecules. When the dispersion medium was changed to ethanol, slight dispersion of the fibers was confirmed, which is considered to be due to ethanol entering the gap between cellulose molecules and solvating them such that the cellulose molecules disaggregate.
[0134] FIG. 15 (a) shows a TEM image of the cellulose nanofiber sheet created by freeze-drying, and FIG. 15 (b) shows a picture of the external appearance and an FE-SEM image thereof. It was confirmed that by freeze-drying, the fibers do not aggregate and are disaggregated compared to sheets created by hot press (FIG. 13 (a), (b) and FIG. 14 (a), (b)). This is considered to be due the fact that fiber aggregation is suppressed because the dispersion medium t-butyl alcohol directly sublimates, without passing through a liquid state, from a solid state to a gaseous state due to freeze-drying.
[0135] 14. Evaluation of Crystallinity of Sheets Via XRD
[0136] The crystallinities of obtained cellulose nanofiber sheets were evaluated using an X-ray diffractometer (XRD (Rigaku Corporation, RINT-Ultima III)). The measurement conditions are shown in Table 13.
TABLE-US-00013 TABLE 13 X-ray tube Cu Acceleration voltage 30 kV Tube current 16 mA Divergence slit 0.5 degrees Scattering slit 0.5 degrees Receiving slit 0.15 mm Sampling range 0.05 degrees Scanning speed 5 degrees/min Measuring range 2.theta. = 10 to 80 degrees
[0137] Additionally, the crystallinity of cellulose was calculated using a strength (I.sub.A) of the 10-1 diffraction peak of cellulose at 2.theta.=15.degree. from the baseline drawn from 2.theta.=10.degree. to 80.degree. and a strength (I.sub.B) from the baseline drawn from 2.theta.=10.degree. to 20.degree., and using the following equation (3).
Crystallinity=(I.sub.A/I.sub.B).times.100 (3)
[0138] The XRD patterns for 3 samples of cellulose nanofiber sheets obtained from 3 samples are shown in FIGS. 16 to 18. In each of the samples, the 10-1 and 002 diffraction peaks of cellulose were confirmed at 2.theta.=15.degree. and 22.5.degree.. Additionally, the crystallinities of the celluloses calculated based on these peak strengths are shown in Table 14. The crystallinities were 71 to 77% regardless of dispersion medium.
TABLE-US-00014 TABLE 14 Method for manufacturing sheet Crystallinity (%) Hot press, water dispersion medium 75.0 Hot press, ethanol dispersion medium 71.4 Freeze-drying, butanol dispersion medium 77.5
[0139] 15. Measurement of Surface Area According to Gas Adsorption-Desorption Measurement
[0140] The BET surface area of the obtained cellulose nanofiber sheet was measured using a nitrogen gas adsorption/desorption device (Yuasa Ionics, AUTOSORB-3). Nitrogen (99.9% purity) was adsorbed on the sheet in a cell at 77 K, and the adsorption amount and pressure inside the cell were measured to obtain an adsorption isotherm. The obtained adsorption isotherm was analyzed using a BET method to calculate the BET surface area. The sample was deaerated by evacuation at 200.degree. C. for 24 hours before the measurement. The measured gas adsorption/desorption isotherm is shown in FIG. 19, and the BET surface area is shown in Table 15.
TABLE-US-00015 TABLE 15 Method for manufacturing sheet BET surface area (m.sup.2/g) Hot press (HP), water dispersion medium 3.18 Hot press (HP), ethanol dispersion medium 22.6 Freeze-drying, t-butanol dispersion medium 36.0
[0141] In the case of a sheet created by hot pressing using ethanol as a dispersion medium, the fibers experienced disaggregation such that the surface area was larger than the case with water as the dispersion medium. In the sheet created by freeze-drying, aggregation of fibers was prevented, and thus, an even larger surface area was obtained relative to the sheets created by hot pressing.
[0142] Additionally, FIG. 20 (sheet created by hot pressing using water as a dispersion medium), FIG. 21 (sheet created by hot pressing using ethanol as a dispersion medium), and FIG. 21 (sheet created by freeze-drying) show distributions of micropore sizes in the sheets.
[0143] 16. Measurement of Strength by Tensile Test
[0144] The tensile strength of a cellulose nanofiber sheet created by hot pressing using water as a dispersion medium was measured. The created sheet was cut into rectangular strips 1.5 cm wide by 2.5 cm long. The top and bottom of the strip were clipped to a length of about 5 mm, and a tensile strength was measured using a desktop precision universal tester (SHIMADZU, AGS-J) at a tensile speed of 1 mm/min. For comparison, tensile strengths of a sheet formed by hot pressing food product-use cellulose nanofibers (Celish.TM.) produced by Daicel FineChem Ltd. and commercially-available paper (ISO9707-certified paper, low residual lignin) from Mondi Limited were measured in the same way.
[0145] FIG. 23 shows the maximum strength relative to the mass of each sample, FIG. 24 shows the maximum strength relative to the thickness of each sample, FIG. 25 shows the maximum strength relative to the density of each sample, and FIG. 26 shows the maximum strength relative to the basis weight (mass per m.sup.2) of each sample. For each sample, it was confirmed that as mass, thickness, density or basis weight increased, the strength also increased.
[0146] Additionally, of the three types of sheets tested, the bamboo-derived cellulose nanofiber sheet of the present invention demonstrated the highest strength. This is considered to be because the bamboo-derived cellulose nanofibers according to the present invention are finer than other fibers, such that the number of fibers per amount of mass is higher, and the sites for hydrogen bonding between fibers increases, thereby increasing the strength. For example, when comparing the FE-SEM images and fiber diameter distributions obtained from the sheet of bamboo-derived cellulose nanofibers according to the present invention and the sheet created in the same manner using cellulose nanofiber Celish.TM. (respectively shown in FIGS. 27 (a) and (b) and FIGS. 28 (a) and (b)), it can be confirmed that the fibers of the former are thinner.
* * * * *

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

P00999

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.