Stable IGG4 Based Binding Agent Formulations
Schnieders; Julia ; et al.
U.S. patent application number 16/707889 was filed with the patent office on 2020-07-16 for stable igg4 based binding agent formulations. The applicant listed for this patent is Sanofi. Invention is credited to Annika Hagendorf, Martina Kirsch, Sabrina Ruggeberg, Julia Schnieders, Dirk Usener, Ahmed Youssef.
| Application Number | 20200222534 16/707889 |
| Document ID | / |
| Family ID | 48048304 |
| Filed Date | 2020-07-16 |





View All Diagrams
| United States Patent Application | 20200222534 |
| Kind Code | A1 |
| Schnieders; Julia ; et al. | July 16, 2020 |
Stable IGG4 Based Binding Agent Formulations
Abstract
The present invention provides stable pharmaceutical antibody formulations, including liquid drug product formulations and lyophilized drug product formulations, comprising an IgG4 binding agent and a citrate buffer, wherein the pH of the formulation is at or below both pH 6 and the pI of the binding agent. The formulations can be used in the treatment of chronic bowel diseases or rheumatoid arthritis.
| Inventors: | Schnieders; Julia; (Frankfurt, DE) ; Usener; Dirk; (Frankfurt, DE) ; Ruggeberg; Sabrina; (Frankfurt, DE) ; Youssef; Ahmed; (Frankfurt, DE) ; Kirsch; Martina; (Frankfurt, DE) ; Hagendorf; Annika; (Hattersheim, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 48048304 | ||||||||||
| Appl. No.: | 16/707889 | ||||||||||
| Filed: | December 9, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15416289 | Jan 26, 2017 | 10525130 | ||
| 16707889 | ||||
| 13850849 | Mar 26, 2013 | 9592289 | ||
| 15416289 | ||||
| 61615539 | Mar 26, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 39/39558 20130101; C07K 2317/21 20130101; A61K 39/39591 20130101; A61K 47/12 20130101; C07K 16/2866 20130101; A61K 47/26 20130101; C07K 16/2875 20130101; C07K 2317/515 20130101; C07K 2317/51 20130101; A61K 39/3955 20130101; A61K 9/19 20130101; C07K 2317/565 20130101; A61K 47/22 20130101; C07K 2317/52 20130101 |
| International Class: | A61K 39/395 20060101 A61K039/395; C07K 16/28 20060101 C07K016/28; A61K 9/19 20060101 A61K009/19; A61K 47/12 20060101 A61K047/12; A61K 47/22 20060101 A61K047/22; A61K 47/26 20060101 A61K047/26 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 6, 2013 | FR | 1351013 |
Claims
1: A pharmaceutical formulation for intravenous or subcutaneous administration, comprising: a humanized or a fully human IgG4 antibody; about 5 to about 50 mM citrate buffer; about 0.005% to about 0.1% polysorbate 20; about 0.1% to about 10% of a tonicity agent; wherein the pH of the formulation is at or below both about pH 6 and the pI of the antibody.
2: The formulation of claim 1, wherein the antibody concentration is from about 5 to about 280 mg/mL.
3: The formulation of claim 1, wherein the citrate concentration is from about 5 to about 15 mM.
4: The formulation of claim 1, wherein the formulation further comprises about 0.1% to about 5% of an amino acid.
5: The formulation of claim 1, wherein the tonicity agent is at least one of a saccharide and sodium chloride.
6: The formulation of claim 5, wherein the saccharide is mannitol or sucrose.
7: The formulation of claim 6, wherein the formulation comprises about 4% mannitol or about 4% to about 6% sucrose.
8: The formulation of claim 5, wherein the formulation comprises about 0.01% to about 1% sodium chloride.
9: The formulation of claim 4, wherein the amino acid is proline or arginine.
10: The formulation of claim 1, wherein the pI of the antibody is from about 6.8 to about 7.2.
11: The formulation of claim 1, wherein the pI of the antibody is from about 7.6 to about 8.4.
12: The formulation of claim 1, wherein the pH is about pH 5 to about pH 6.
13: The formulation of claim 12, wherein the pH is selected from the group consisting of about pH 5.0, about pH 5.5, and about pH 6.0.
14: The formulation of claim 1, wherein the formulation exhibits a reduced amount of at least one byproduct selected from the group consisting of aggregates, half-molecules, degradation products, low molecular weight proteins, high molecular weight proteins, and rearrangement of acidic/basic/neutral isoforms of the humanized or fully human IgG4 antibody as compared to a reference formulation comprising the humanized or fully human IgG4 antibody in phosphate buffered saline at pH 7.3.
15: The formulation of claim 1, wherein the formulation is a liquid formulation.
16: The formulation of claim 1, wherein the formulation is a lyophilized formulation.
17: A kit comprising a container comprising: 1) the formulation of claim 1, and 2) a label or instructions for the administration and use of the formulation.
18: A pre-filled container comprising the formulation of claim 1.
19: The formulation of claim 1, wherein the formulation is stable for at least 6 months at +5.degree. C.
20: The formulation of claim 1, wherein the formulation is stable for at least 9 months at +5.degree. C.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 15/416,289, filed Jan. 26, 2017, which is a divisional of U.S. patent application Ser. No. 13/850,849, filed Mar. 26, 2013, now U.S. Pat. No. 9,592,289, which claims the benefit of U.S. Provisional Patent Application No. 61/615,539, filed Mar. 26, 2012, all of which are incorporated by reference herein in their entireties. This application also claims the benefit of French Patent Application No. 1351013, filed Feb. 6, 2013, which is incorporated by reference herein in its entirety.
BACKGROUND OF THE INVENTION
[0002] The human LIGHT antigen is one potential cytokine target that has been implicated in the processes of chronic inflammatory autoimmune disease. As a member of the TNF superfamily (TNFSF) of ligands, LIGHT is also known as TNFSF14 or CD258. LIGHT is expressed on the surface of T cells upon activation in a tightly regulated manner. However, LIGHT is also present at detectable levels constitutively on the surface of immature dendritic cells and on T cells and natural killer (NK) cells of the gut. LIGHT mediates its biologic effects by binding three TNF superfamily receptors, including the lymphotoxin .beta. receptor (LT.beta.R), the herpes virus entry mediator (HVEM), and decoy receptor 3 (DcR3). LIGHT-expressing lymphocytes can induce IBD-like symptoms in humans, and increases of LIGHT expression have been observed in patients with active Crohn's disease and other inflammatory disorders such as Graft-vs.-Host Disease.
[0003] CXCR5, also known as Burkitt lymphoma receptor (BLR1), CD185, MDR15, and MGC117347, is a G protein-coupled receptor that is a member of the CXC chemokine receptor family. The unprocessed CXCR5 precursor is 372 amino acids in length with a molecular weight of 42 KD. CXCR5 has a role in B cell migration and localization within particular anatomic compartments. Knockout mice lack peripheral lymph nodes, have fewer Peyer's patches and have decreased B cell levels. CXCL13, also known as BLC, is a ligand for CXCR5. CXCL13 is a B cell chemoattractant.
[0004] Anti-LIGHT binding agents and anti-CXCR5 binding agents are each therapeutically relevant, and a need exists to formulate each of these binding agents into drug products that may be administered to subjects, particularly human subjects, for the treatment of inflammatory diseases.
[0005] In order to develop a pharmaceutical formulation containing an anti-LIGHT binding agent or an anti-CXCR5 binding agent suitable for intravenous or subcutaneous administration, the binding agent must be concentrated to about 20 mg/mL or greater, usually about 100-150 mg/mL, and even up to 250 mg/mL. Many complications can arise at such high concentrations, including an increase in viscosity, a shift of pH, a change of the color of the solution, and the formation of visible and sub-visible particles.
[0006] The formulation of these binding agents is further complicated by the fact that these agents are highly prone to aggregation at such high concentrations.
[0007] The formulation of IgG4 antibodies is even further complicated by the fact that IgG4 antibodies tend to form half-molecules at high concentrations in solution. However, IgG4 antibodies are of therapeutic interest because they have reduced effector function.
SUMMARY OF THE INVENTION
[0008] To meet these and other needs, provided herein are highly stable IgG4 binding agent formulations. Highly stable IgG4 binding agent formulations have surprisingly been found in the form of liquids and lyophilized powders that comprise an IgG4 binding agent and a citrate buffer, wherein the pH of the formulation is at or below both about pH 6 and the isoelectric point (pI) of the binding agent. These formulations improve upon conventional formulations, which often lead to dimerization of the binding agent, such as an antibody, upon increasing the concentration of the binding agent, such as an antibody, in the formulation. In particular, the formulations of the invention reduce the amount of unwanted byproducts, including aggregates, half-molecules, degradation products, low molecular weight proteins (LMWPs), high molecular weight proteins (HMWPs), and rearrangements of acid, basic, and neutral isoforms of the binding agent, such as an antibody, component in the formulation.
[0009] In certain aspects, the invention provides a stable formulation comprising: a binding agent comprising at least a portion of a Fc region of an IgG4 antibody; and about 5 to about 50 mM citrate as a buffering agent; wherein the pH of the formulation is at or below both about pH 6 and the pI of the binding agent. In certain embodiments of the invention, the binding agent is an antibody.
[0010] In certain embodiments of the invention, the binding agent or antibody binds to lymphotoxin-like, exhibits inducible expression and competes with herpes virus glycoprotein D for herpes virus entry mediator, a receptor expressed on lymphocytes (LIGHT). In specific embodiments of the invention, the anti-LIGHT binding agent or antibody comprises a heavy chain variable region and a light chain variable region, the heavy chain variable region comprising complementary determining regions (CDRs) comprising the amino acid sequences of SEQ ID NOS: 1, 2, and 3, and the light chain variable region comprising CDRs comprising the amino acid sequences of SEQ ID NOS: 4, 5, and 6. In other specific embodiments of the invention, the antibody is a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8.
[0011] In certain embodiments of the invention, the binding agent or antibody binds to C-X-C chemokine receptor type 5 (CXCR5). In specific embodiments of the invention, the anti-CXCR5 binding agent or antibody comprises a heavy chain variable region and a light chain variable region, the heavy chain variable region comprising complementary determining regions (CDRs) comprising the amino acid sequences of SEQ ID NOS: 15, 16, and 17, and the light chain variable region comprising CDRs comprising the amino acid sequences of SEQ ID NOS: 18, 19, and 20. In other specific embodiments of the invention, the antibody is a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26.
[0012] In certain embodiments of the invention, the antibody concentration is from about 5 to about 280 mg/mL. In certain specific embodiments of the invention, the antibody concentration is about 150 mg/mL. In other specific embodiments of the invention, the antibody concentration is about 50 mg/mL. In further specific embodiments of the invention, the antibody concentration is about 20 mg/mL. In yet further specific embodiments of the invention, the antibody concentration is about 100 mg/mL.
[0013] In certain embodiments of the invention, the citrate concentration is from about 5 to about 15 mM. In some embodiments of the invention, the citrate concentration is about 10 mM. In some embodiments of the invention, the citrate buffer is sodium citrate dihydrate.
[0014] In certain embodiments of the invention, the pH of the formulation is from about pH 5 and about pH 6. In specific embodiments of the invention, the pH of the formulation is selected from the group consisting of about pH 5.0, about pH 5.5, and about pH 6.0.
[0015] In certain specific embodiment of the invention, the pI of the binding agent or antibody is from about 6.8 and about 7.2. In alternative specific embodiments of the invention, the pI of the binding agent or antibody is from about 7.6 and about 8.4.
[0016] In certain specific embodiments of the invention, the formulation further comprises a surfactant. In certain specific embodiments of the invention, the concentration of surfactant is between about 0.001% and about 0.1% w/v. In certain embodiments of the invention, the surfactant is a polysorbate. In certain specific embodiments of the invention, the polysorbate is polysorbate 20. In some specific embodiments of the invention, the concentration of polysorbate 20 is about 0.005% w/v. In alternative specific embodiments of the invention, the concentration of polysorbate 20 is about 0.01% w/v. In further alternative specific embodiments of the invention, the concentration of polysorbate 20 is about 0.02% w/v.
[0017] In certain embodiments of the invention, the formulation further comprises a tonicity agent. In certain specific embodiments of the invention, the concentration of tonicity agent is between about 0.1% and about 10% w/v. In certain specific embodiments of the invention, the tonicity agent is a saccharide. In some specific embodiments of the invention, the saccharide is mannitol. In other specific embodiments of the invention, the concentration of mannitol is between about 1% and about 10% w/v. In yet other specific embodiments of the invention, the concentration of mannitol is about 4%. In alternative specific embodiments of the invention, the saccharide is sucrose. In some specific embodiments of the invention, the concentration of sucrose is between about 1% and about 10% w/v. In some specific embodiments of the invention, the concentration of sucrose is about 5% w/v. In alternative specific embodiments of the invention, the concentration of sucrose is about 6% w/v. In yet other specific embodiments of the invention, the concentration of sucrose is about 4.5% w/v. In further specific alternative embodiments of the invention, the tonicity agent is sodium chloride. In some specific embodiments of the invention, the concentration of sodium chloride is between about 0.01% and about 1%. In some specific embodiments of the invention, the concentration of sodium chloride is about 0.2%. In other specific embodiments of the invention, the tonicity agent is a combination of sucrose and sodium chloride. In specific embodiments of the invention, the concentration of sucrose is between about 1% and about 10% w/v. In other specific embodiments of the invention, the concentration of sodium chloride is between about 0.01% and about 1%. In alternative specific embodiments of the invention, the concentration of sucrose is about 6% w/v and the concentration of sodium chloride is about 0.2%. In yet further alternative specific embodiments of the invention, the concentration of sucrose is about 4.5% w/v and the concentration of sodium chloride is about 0.2%.
[0018] In certain embodiments of the invention, the formulation further comprises an amino acid. In certain specific embodiments of the invention, the amino acid concentration is between about 0.1% and about 5% w/v. In certain specific embodiments of the invention, the amino acid is proline or arginine. In specific embodiments of the invention, the proline or arginine concentration is between about 1% and about 2% w/v. In other specific embodiments of the invention, the proline concentration is about 1.5% w/v. In alternative specific embodiments of the invention, the arginine concentration is about 1% w/v.
[0019] In certain embodiments of the invention, the formulation is a liquid formulation. In other specific embodiments of the invention, the formulation is a lyophilized formulation.
[0020] In certain embodiments of the invention, the formulation is stable for at least 6 months at +5.degree. C. In alternative embodiments of the invention, the formulation is stable for at least 9 months at +5.degree. C.
[0021] In certain embodiments of the invention, the formulation exhibits a reduced amount of at least one byproduct selected from the group consisting of aggregates, half-molecules, degradation products, low molecular weight proteins, high molecular weight proteins, and rearrangements of acidic/basic/neutral isoforms of the antibody as compared to either a reference anti-LIGHT formulation comprising an anti-LIGHT antibody in phosphate buffered saline at pH 7.3 or a reference anti-CXCR5 formulation comprising an anti-LIGHT antibody in phosphate buffered saline at pH 7.3.
[0022] In certain specific embodiments of the invention, the invention provides a stable liquid antibody formulation suitable for subcutaneous administration, the formulation comprising:
[0023] a) about 150 mg/mL of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0024] b) about 10 mM citrate buffer;
[0025] c) about 0.005% polysorbate 20; and
[0026] d) about 4% mannitol;
wherein the pH of the formulation is about pH 5.5.
[0027] In other specific embodiments of the invention, the invention provides a stable liquid antibody formulation suitable for intravenous administration, the formulation comprising:
[0028] a) about 50 mg/mL of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0029] b) about 10 mM citrate buffer; and
[0030] c) about 0.01% polysorbate 20;
wherein the pH of the formulation is about pH 5.5.
[0031] In yet other specific embodiments of the invention, the invention provides a stable lyophilized antibody formulation suitable for intravenous administration, the formulation comprising:
[0032] a) about 50 mg/mL of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0033] b) about 10 mM citrate buffer;
[0034] c) about 0.01% polysorbate 20;
[0035] d) about 5% sucrose; and
[0036] e) about 1.5% proline;
wherein the pH of the formulation is about pH 5.5.
[0037] In alternative specific embodiments of the invention, the invention provides a stable antibody formulation comprising:
[0038] a) about 20 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26;
[0039] b) about 10 mM citrate buffer;
[0040] c) about 0.02% polysorbate 20;
[0041] d) about 6% sucrose; and
[0042] e) about 0.2% sodium chloride;
wherein the pH of the formulation is about pH 6.0.
[0043] In further alternative specific embodiments of the invention, the invention provides a stable antibody formulation comprising:
[0044] a) about 100 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26;
[0045] b) about 10 mM citrate buffer;
[0046] c) about 0.01% polysorbate 20;
[0047] d) about 4.5% sucrose;
[0048] e) about 0.2% sodium chloride; and
[0049] f) about 1% arginine;
wherein the pH of the formulation is about pH 6.0.
[0050] In certain embodiments of the invention, the invention provides a kit comprising a container comprising: 1) the formulation of any one of the previous claims, and 2) a label or instructions for the administration and use of the formulation. In certain embodiments of the invention, the label comprises one or more of the following: instructions for the administration of the formulation, instructions for use of the formulation, instructions concerning the storage conditions of the formulation, information concerning lot and batch number of the formulation and/or kit, information concerning the composition of the formulation, safety information, information concerning possible adverse reactions, secondary effects, and/or side effects in connection with the administration of the formulation, or information concerning possible indications and/or contra-indications of the formulation.
[0051] In certain embodiments of the invention, the invention provides a pre-filled device or pre-filled container, such as a syringe, cartridge, vial, ampoule, or autoinjector comprising the formulation of the invention. In certain other embodiments, the invention provides a kit comprising such pre-filled syringe, cartridge, vial, ampoule, or autoinjector.
[0052] In certain embodiments, the invention provides a method for treating an inflammatory bowel disease comprising administering to a subject in need thereof a formulation of the invention.
[0053] In other certain embodiments, the invention provides a method for treating rheumatoid arthritis comprising administering to a subject in need thereof a formulation of the invention.
[0054] In certain embodiments, the invention provides a formulation for use in a method of diagnosis or treatment of the human or animal body. In specific embodiments, the formulation is used in the treatment of inflammatory bowel disease. In alternative embodiments, the formulation is used in the treatment of rheumatoid arthritis.
[0055] In certain embodiments of the invention, the invention provides a method for preparing a formulation of the invention comprising mixing the components of the formulation and adjusting the pH, wherein the preparation is performed under sterile conditions or the formulation is sterilized after the mixing of the components and the pH adjustment or both.
[0056] In certain specific embodiments of the invention, the invention provides a method for preparing a stable antibody formulation comprising: a) providing an anti-LIGHT binding agent; b) resuspending the anti-LIGHT binding agent in about 5 to about 50 mM citrate buffer; and c) adjusting the pH of the formulation to about pH 5.0 to about pH 6.0.
BRIEF DESCRIPTION OF THE FIGURES
[0057] FIG. 1 is a picture of a gel showing the results of denatured isoelectric focusing experiments that were used to determine the isoelectric point (pI) of the fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8 formulated in phosphate buffered saline at pH 7.3 at a concentration of 5.5 mg/mL (the "Original Formulation", "PBS Formulation", or "Reference Lot"). Lanes 1 & 5: IEF Calibration Kit High Range pI 5-10.5; lanes 2 & 4: a first batch of Reference Lot; lanes 3 & 4: a second batch of Reference Lot. The pI values are indicated by numbers.
[0058] FIG. 2 is a picture of an SDS-PAGE gel that compared different Reference Lot batches under reducing and non-reducing conditions. Lanes 1 & 10: Biorad Precision Plus Protein Standard; lane 5: empty; lane 2: a first batch of Reference Lot under non-reduced conditions; lanes 3 & 4: a second batch of Reference Lot under non-reduced conditions; lane 6: a first batch of Reference Lot under reduced conditions; lanes 7 & 8: a second batch of Reference Lot under reduced conditions; and lane 9: system control. The sizes are indicated by numbers within the rows.
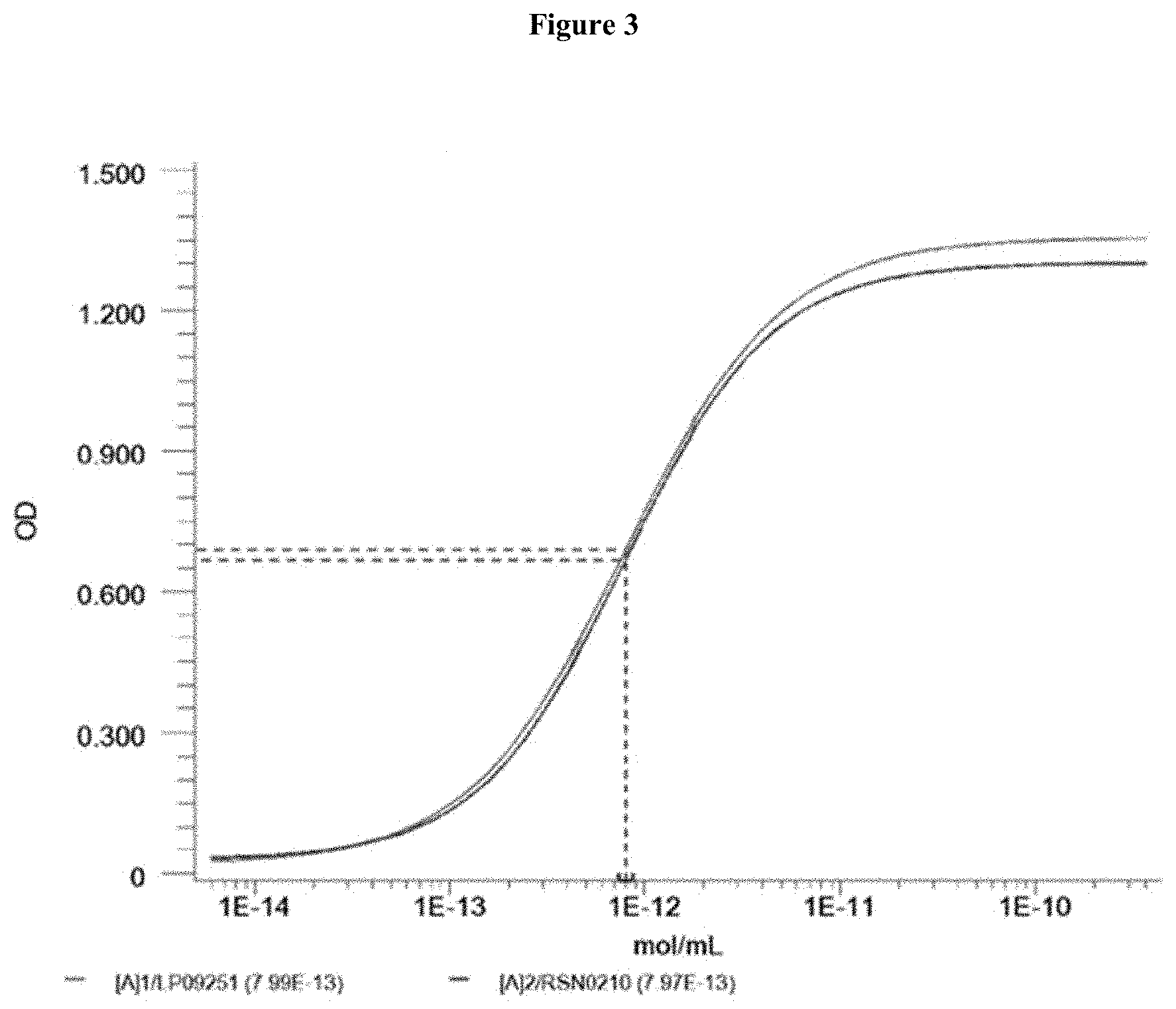
[0059] FIG. 3 shows an ELISA graph that was used to determine the antigen binding activity of the first and second batches of Reference Lot.
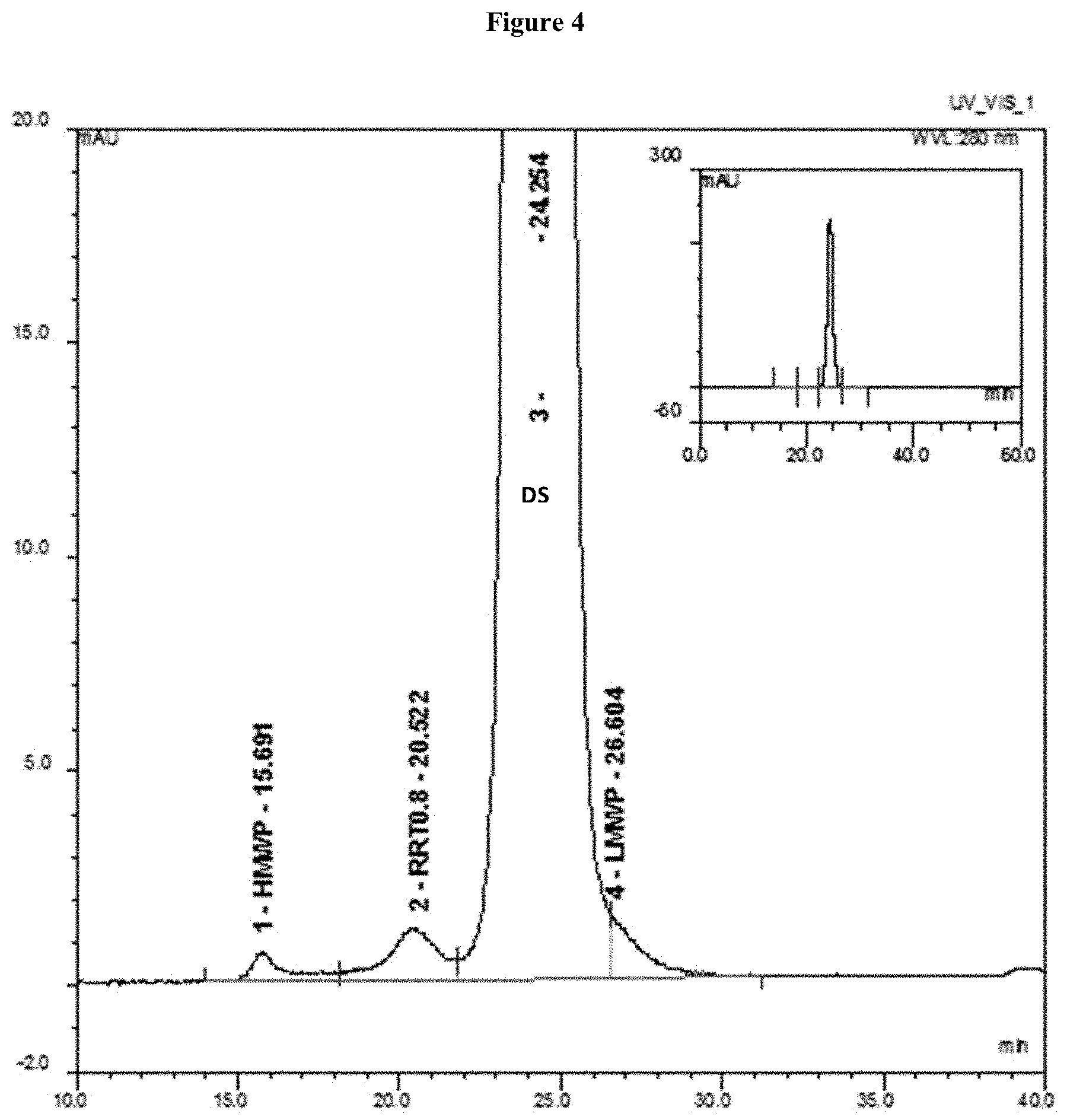
[0060] FIG. 4 shows a size exclusion chromatography (SEC) chromatogram of the first batch of Reference Lot. As shown in FIG. 4, SEC detected high molecular weight proteins (HMWP), e.g., di-/oligomers (RRT0.8) or aggregates, and low molecular weight proteins (LMWPs) or degradation products. The first batch of Reference Lot batch had a purity of 97% monomer content.
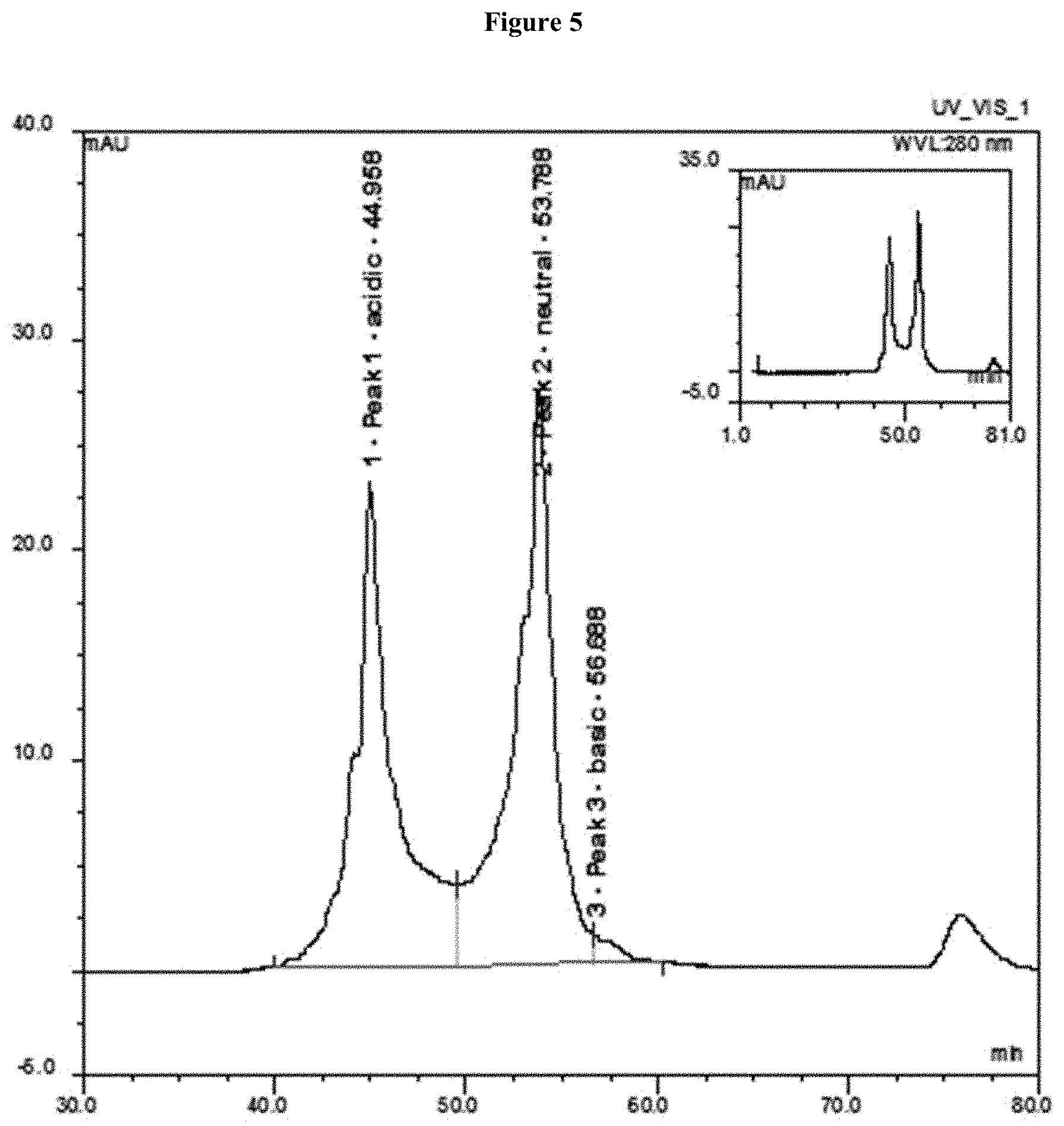
[0061] FIG. 5 shows a weak cation exchange chromatogram for the first batch of Reference Lot. As shown in FIG. 5, rearrangements of acidic, neutral, and basic isoforms occurred during stability studies. The first batch of Reference Lot had a distribution of acidic/neutral/basic isoforms of 42.3/55.6/1.9%.
[0062] FIG. 6 shows a differential scanning calorimetry thermogram of the first batch of Reference Lot. As shown in FIG. 6, the three domains of the antibody unfold at 68.degree. C., 75.degree. C., and 78.degree. C.
[0063] FIG. 7 shows a dynamic light scattering pattern of the first batch of Reference Lot, which was unfiltered. DLS was used to determine the hydrodynamic diameter of the first batch of Reference Lot antibody monomer and potential soluble aggregates.
[0064] FIG. 8 shows a dynamic light scattering pattern of the first batch of Reference Lot, which was filtered. DLS was used to determine the hydrodynamic diameter of the first batch of Reference Lot antibody monomer and potential soluble aggregates.
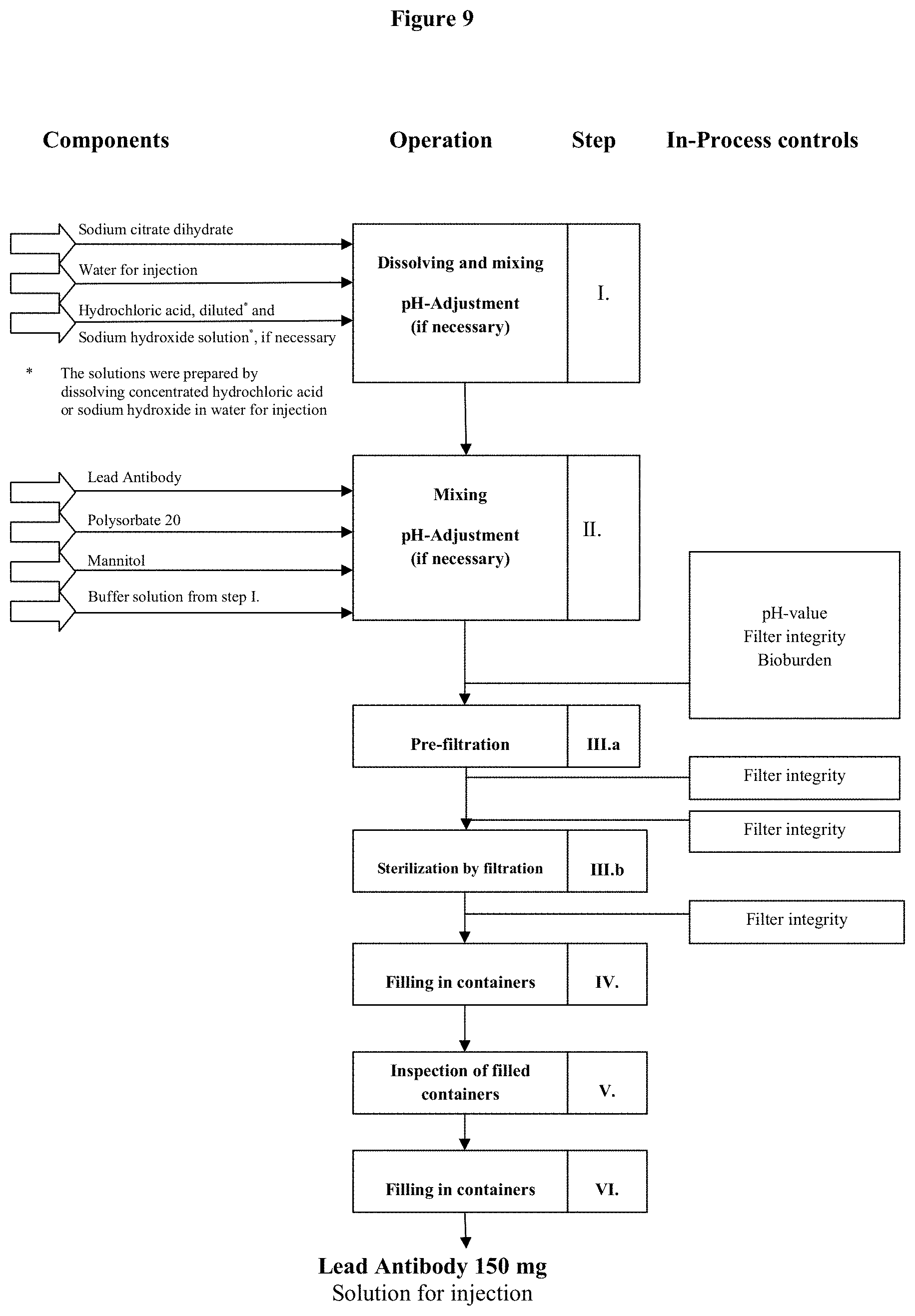
[0065] FIG. 9 is a flow diagram of the drug product manufacturing process for the high antibody concentration formulation.
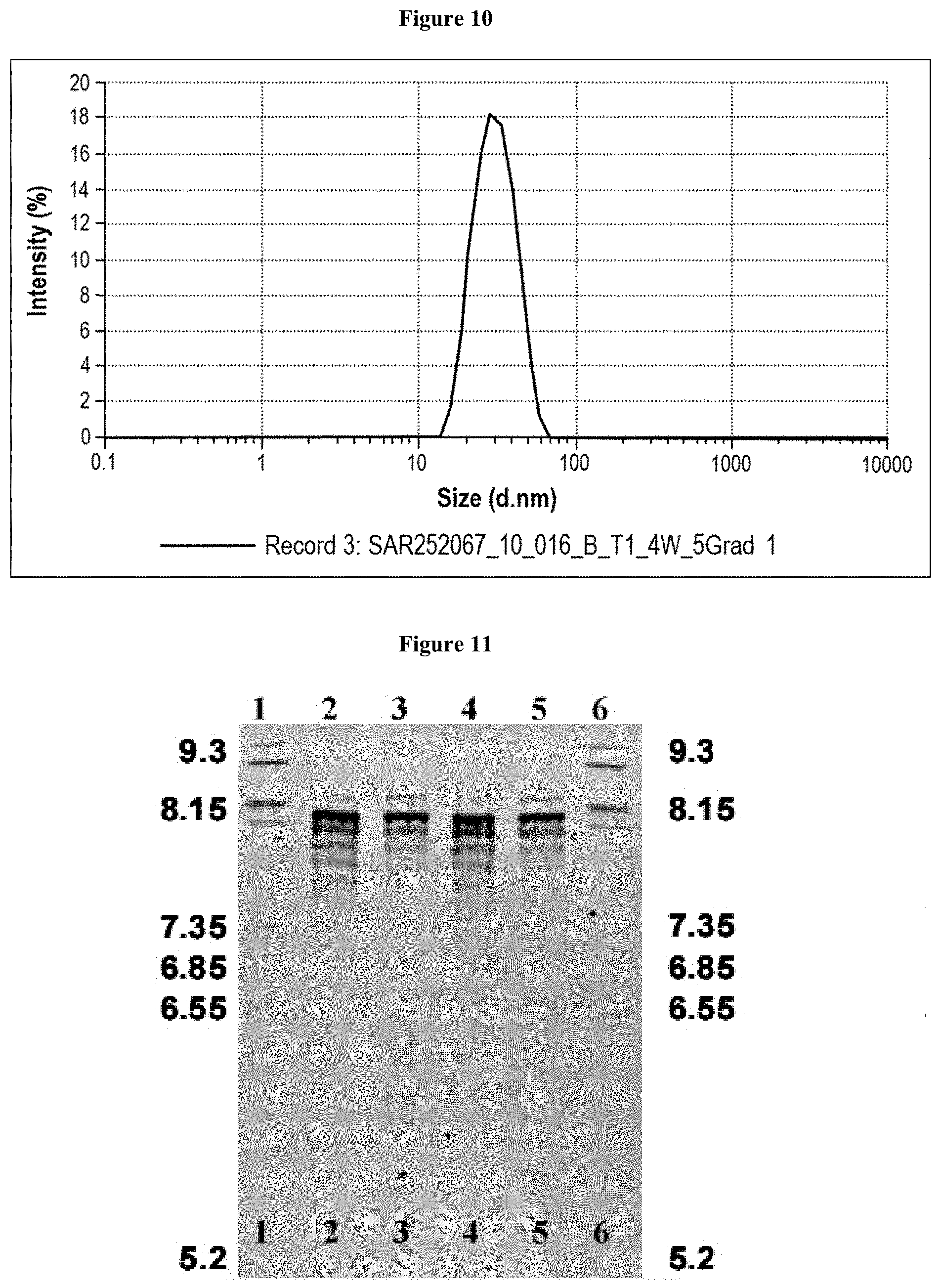
[0066] FIG. 10 shows a dynamic light scattering pattern of Formulation 14. DLS was used to determine the hydrodynamic diameter of the antibody monomer and potential soluble aggregates.
[0067] FIG. 11 is a picture of a gel showing the results of isoelectric focusing to determine the pI (isoelectric point) of the Lead CXCR5 Antibody. Lanes 1,6: IEF Calibration High Range pI Kit; Lanes 2,4: Reference Standard Lead Antibody LP08031; and Lanes 3,5: Lead Antibody Drug Substance, RSN0151.
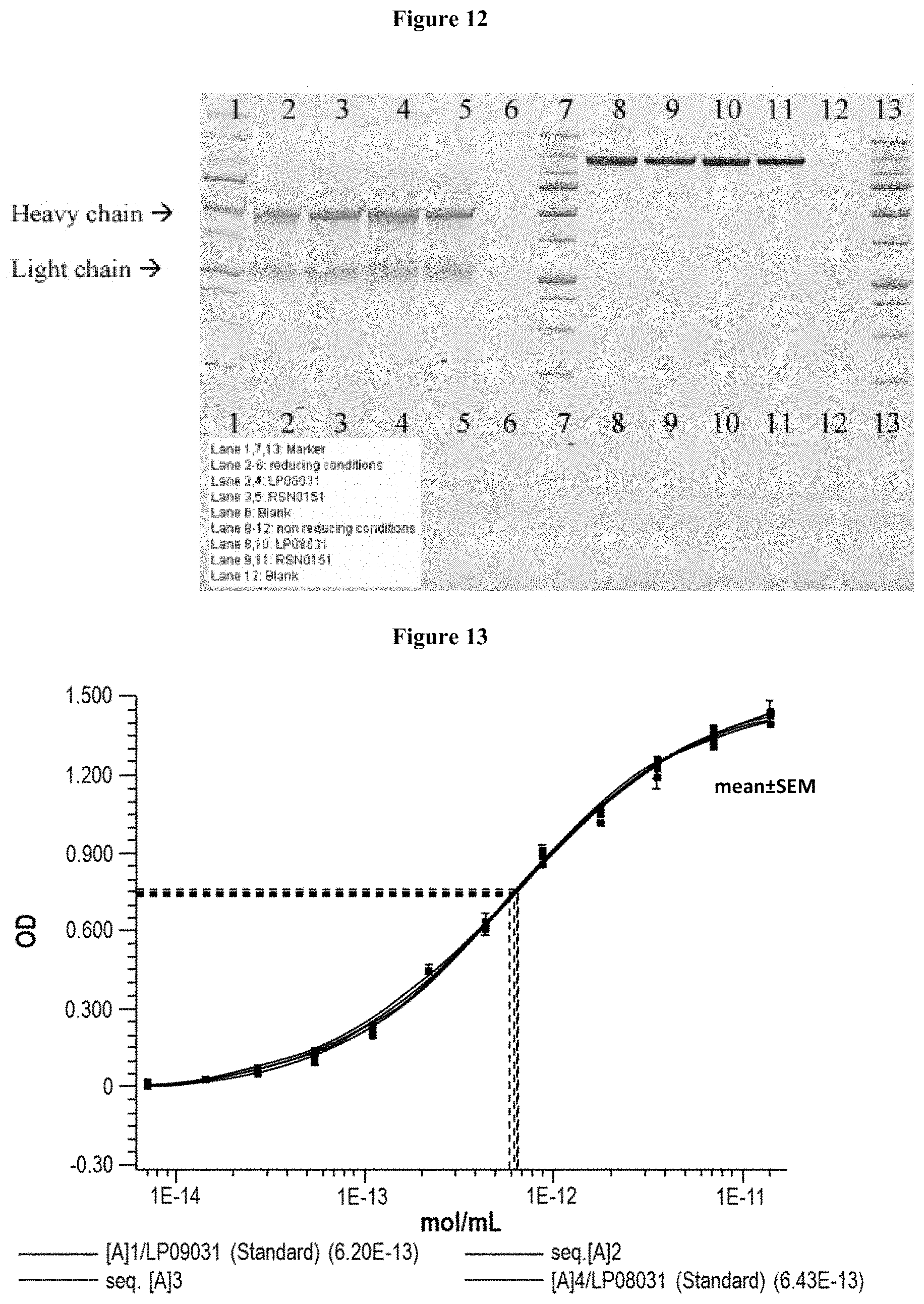
[0068] FIG. 12 is a picture of an SDS-PAGE gel that compared different drug substance batches under reducing and non-reducing conditions. The gel was also used to determine the molecular weight of the Lead CXCR5 Antibody, and the presence of any aggregates.
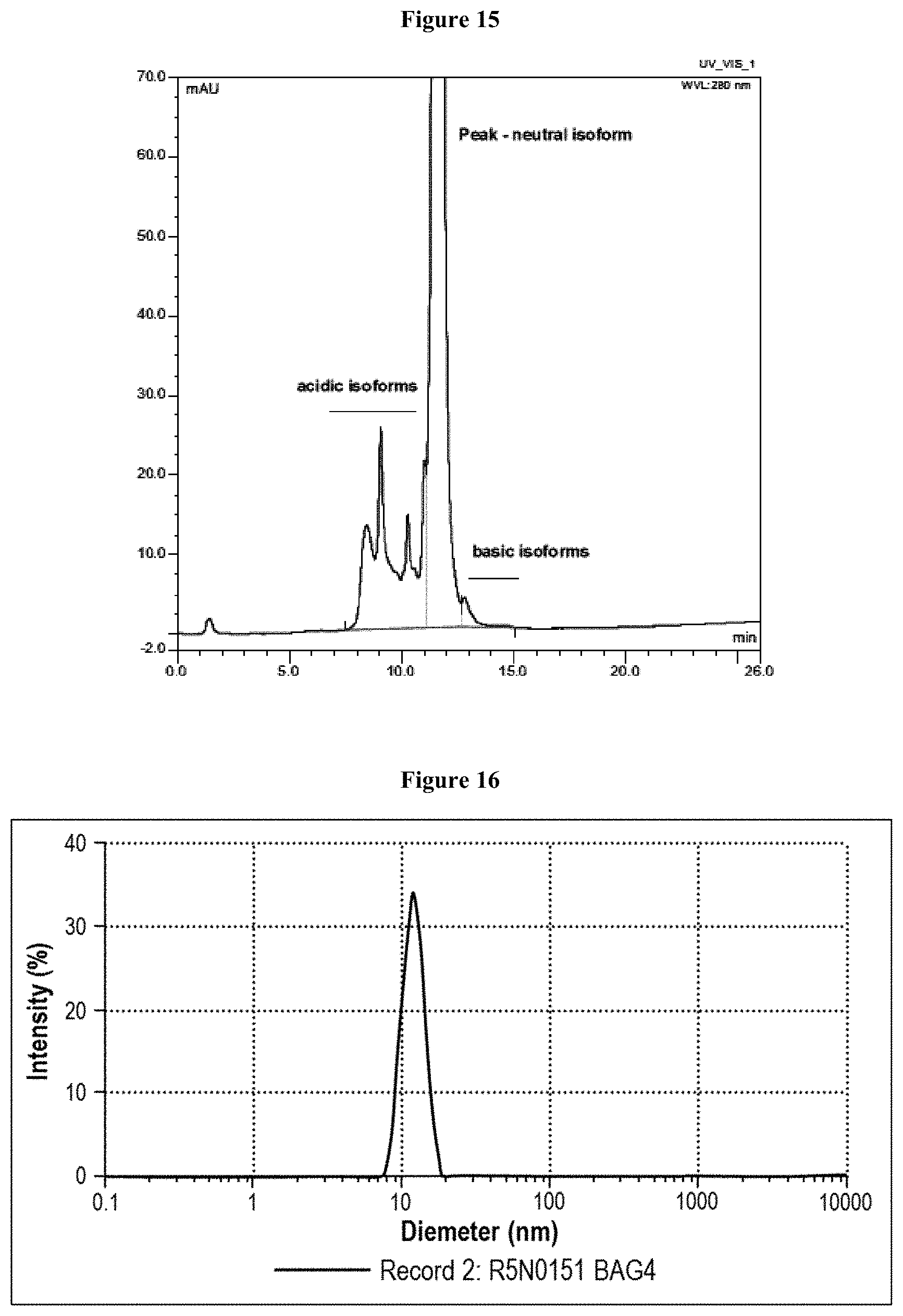
[0069] FIG. 13 is an ELISA graph that was used to determine antigen binding activity of the Lead CXCR5 Antibody to a 28mer peptide of the CXCR5 antigen.
[0070] FIG. 14 is a SEC chromatogram of stressed Lead CXCR5 Antibody. SEC could detect high molecular weight proteins (HMWP), e.g., di-/oligomers or aggregates and low molecular weight proteins (LMWP) or degradation products. The Lead CXCR5 Antibody had a purity of 99% monomer content.
[0071] FIG. 15 is a WCX chromatogram that was used to determine acidic, neutral, and basic isoforms of the Lead CXCR5 Antibody. The Lead CXCR5 Antibody had a distribution of acidic/neutral/basic isoforms of 14/85/1%.
[0072] FIG. 16 is a DLS measurement that was used to determine the hydrodynamic diameter of the antibody monomer and potential soluble aggregates.
[0073] FIG. 17 is a picture of the Lead CXCR5 Antibody in acetate buffer pH 5.0 (left) and pH 5.5 (right); each v. WFI (water for injection) and after thermal stress. This figure shows that acetate is a suitable buffer system.
[0074] FIG. 18 is a picture of the Lead CXCR5 Antibody in histidine buffer pH 6.0 (left), pH 5.5 (middle), and pH 5.0 (right); each v. WFI (water for injection) and after thermal stress. This figure shows that histidine is a suitable buffer.
[0075] FIG. 19 is a picture of the Lead CXCR5 Antibody in TRIS buffer pH 7.5 after UF/DF (left) and after filtration (right); each v. WFI (water for injection) and after thermal stress. This figure shows that TRIS is an incompatible buffer system.
[0076] FIG. 20 is a picture of the Lead CXCR5 Antibody in citrate buffer pH 6.0 after UF/DF and filtration.
[0077] FIG. 21 is a picture of the Lead CXCR5 Antibody in acetate buffer pH 5.5 after UF/DF and filtration.
[0078] FIG. 22 is a picture of the Lead CXCR5 Antibody in succinate buffer pH 5.0 after UF/DF and filtration.
[0079] FIG. 23 is a picture of the Lead CXCR5 Antibody in histidine buffer pH 5.0 after UF/DF and filtration.
[0080] FIG. 24 is a picture of the Lead CXCR5 Antibody in arginine buffer pH 6.0 after UF/DF and filtration.
[0081] FIG. 25 is a picture of the appearance of Lead CXCR5 Antibody LA_09_016 solutions with different surfactants (without surfactant, polysorbate 20, polysorbate 80, Lutrol F68, Cremophor RH40, Solutol HS15, and SDS) after mechanical stress (350 rpm, 2.5 h, RT).
[0082] FIG. 26 is a graph that shows an increase of dimers under accelerated conditions, as analyzed by SEC. An increase of dimer formation up to 10% after three months of storage in all four histidine formulation can be seen. Acetate formulations showed an increase of dimer content up to 6%. In all four citrate formulations, the dimer concentration was below 2%, even after three months at +40.degree. C.
[0083] FIG. 27 is a graph showing an increase of basic isoforms under accelerated conditions, as analyzed by WCX. Histidine is worse for Lead CXCR5 Antibody stability under accelerated conditions. A slight increase of basic isoforms can be noticed for all four acetate formulations. Interestingly, it was not possible to discriminate between the four citrate formulations.
[0084] FIG. 28 is a graph showing a decrease of neutral isoforms under accelerated conditions, as analyzed by WCX. This figure shows a strong decrease in neutral isoforms for the histidine formulations. A slight decrease was seen in acetate. Citrate was affected the least.
[0085] FIG. 29 shows the delta pH of all four formulations (A-D) in citrate buffer at accelerated conditions. The most pH stabilizing formulations are the citrate buffered, and especially formulation B and D.
[0086] FIG. 30 shows the delta pH of all four formulations (A-D) in acetate buffer at accelerated conditions. In acetate buffered solutions of the Lead CXCR5 Antibody, the pH was shifted towards higher value.
[0087] FIG. 31 shows the delta pH of all four formulations (A-D) in histidine buffer at accelerated conditions. In histidine buffered solutions of the Lead CXCR5 Antibody, the pH was slightly decreasing.
[0088] FIG. 32 is a graph showing the hydrodynamic diameter of CXCR5 LA_09_027 A-D after 3 months storage at 40.degree. C. Citrate buffered formulations showed only slight aggregates after three weeks in formulation C, and after six weeks of storage in formulation A. Some aggregates could be detected after three months in formulation B as well. But, compared to acetate buffered formulations, the amount was very little.
[0089] FIG. 33 is a graph showing the hydrodynamic diameter of CXCR5 LA_09_028 A-D after 3 months storage at 40.degree. C. The acetate buffered formulation C showed some aggregates <200 nm after three weeks. Formulation A showed some aggregates after three months.
[0090] FIG. 34 is a chart showing the effect of increasing Lead CXCR5 Antibody concentration on the Z-average. The Lead CXCR5 Antibody showed a significant increase in the hydrodynamic diameter (Z-Average) by increasing the concentration of the antibody.
[0091] FIG. 35 is a chart showing the effect of different stabilizers (excipients) on the Z-Average at 100 mg/mL of Lead CXCR5 Antibody after thermal stress. Z-Average was measured before and after thermal stress. The stabilizing effect was similar to all tested excipients, but the increase in Z-average was generally reduced by using amino acids as stabilizers (arginine, lysine, or glycine). Lysine was excluded due to a higher content of aggregates after stress. Arginine showed a better effect than glycine.
[0092] FIG. 36 is a chart showing the effect of different stabilizers on the Z-Average at 100 mg/mL Lead CXCR5 Antibody after mechanical stress. Z-Average was measured before and after mechanical stress. The same reduction in Z-average was noticed in the presence of amino acids. Sucrose had a better protective effect than trehalose against mechanical stress. Arginine and glycine performed better in combination with NaCl.
[0093] FIG. 37 is a set of graphs showing particle size distribution, as measured by DLS, of Lead CXCR5 Antibody formulated in 10 mM citrate buffer at pH 6 before mechanical stress (A) and after mechanical stress (B). A higher molecular weight species was measured by DLS after mechanical stress of DS.
[0094] FIG. 38 is a set of graphs showing particle size distribution, as measured by DLS, of Lead CXCR5 Antibody drug product prototype formulations (A-D; Table 110) before (A) and after (B) mechanical stress.
DETAILED DESCRIPTION
A. Definitions
[0095] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as is commonly understood by one of ordinary skill in the art.
[0096] It is noted here that as used in this specification and the appended claims, the singular forms "a", "an", and "the" also include plural reference, unless the context clearly dictates otherwise.
[0097] The term "about" or "approximately" means within 10%, such as within 5% (or 1% or less) of a given value or range.
[0098] The terms "administer" or "administration" refers to the act of injecting or otherwise physically delivering a substance as it exists outside the body (e.g., a formulation of the invention) into a patient, such as by mucosal, intradermal, intravenous, subcutaneous, intramuscular delivery and/or any other method of physical delivery described herein or known in the art. When a disease, or a symptom thereof, is being treated, administration of the substance typically occurs after the onset of the disease or symptoms thereof. When a disease or its symptoms are being prevented, administration of the substance typically occurs before the onset of the disease or symptoms thereof.
[0099] In the context of a polypeptide, the term "analog" refers to a polypeptide that possesses a similar or identical function as a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody, but does not necessarily comprise a similar or identical amino acid sequence of a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody, or possess a similar or identical structure of a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody. A polypeptide that has a similar amino acid sequence refers to a polypeptide that satisfies at least one of the following: (a) a polypeptide having an amino acid sequence that is at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% identical to the amino acid sequence of a LIGHT or CXCR5 polypeptide (e.g., SEQ ID NO: 9 or SEQ ID NO: 14, respectively), a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody described herein; (b) a polypeptide encoded by a nucleotide sequence that hybridizes under stringent conditions to a nucleotide sequence encoding a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody (or VH or VL region thereof) described herein of at least 5 amino acid residues, at least 10 amino acid residues, at least 15 amino acid residues, at least 20 amino acid residues, at least 25 amino acid residues, at least 40 amino acid residues, at least 50 amino acid residues, at least 60 amino residues, at least 70 amino acid residues, at least 80 amino acid residues, at least 90 amino acid residues, at least 100 amino acid residues, at least 125 amino acid residues, or at least 150 amino acid residues (see, e.g., Sambrook et al. (2001) Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.; Maniatis et al. (1982) Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, N.Y.); and (c) a polypeptide encoded by a nucleotide sequence that is at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% identical to the nucleotide sequence encoding a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody (or VH or VL region thereof) described herein. A polypeptide with similar structure to a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or an anti-LIGHT or anti-CXCR5 antibody refers to a polypeptide that has a similar secondary, tertiary or quaternary structure of a LIGHT or CXCR5 polypeptide, a fragment of a LIGHT or CXCR5 polypeptide, a LIGHT or CXCR5 epitope, or a LIGHT or CXCR5 antibody. The structure of a polypeptide can determined by methods known to those skilled in the art, including but not limited to, X-ray crystallography, nuclear magnetic resonance, and crystallographic electron microscopy.
[0100] To determine the percent identity of two amino acid sequences or of two nucleic acid sequences, the sequences are aligned for optimal comparison purposes (e.g., gaps can be introduced in the sequence of a first amino acid or nucleic acid sequence for optimal alignment with a second amino acid or nucleic acid sequence). The amino acid residues or nucleotides at corresponding amino acid positions or nucleotide positions are then compared. When a position in the first sequence is occupied by the same amino acid residue or nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position. The percent identity between the two sequences is a function of the number of identical positions shared by the sequences (i.e., % identity=number of identical overlapping positions/total number of positions X 100%). In one embodiment, the two sequences are the same length.
[0101] The determination of percent identity between two sequences (e.g., amino acid sequences or nucleic acid sequences) can also be accomplished using a mathematical algorithm. A non-limiting example of a mathematical algorithm utilized for the comparison of two sequences is the algorithm of Karlin and Altschul, 1990, Proc. Natl. Acad. Sci. U.S.A. 87:2264 2268, modified as in Karlin and Altschul, 1993, Proc. Natl. Acad. Sci. U.S.A. 90:5873 5877. Such an algorithm is incorporated into the NBLAST and XBLAST programs of Altschul et al., 1990, J. Mol. Biol. 215:403. BLAST nucleotide searches can be performed with the NBLAST nucleotide program parameters set, e.g., for score=100, wordlength=12 to obtain nucleotide sequences homologous to nucleic acid molecules of interest. BLAST protein searches can be performed with the XBLAST program parameters set, e.g., to score 50, wordlength=3 to obtain amino acid sequences homologous to a protein molecule of interest. To obtain gapped alignments for comparison purposes, Gapped BLAST can be utilized as described in Altschul et al., 1997, Nucleic Acids Res. 25:3389 3402. Alternatively, PSI BLAST can be used to perform an iterated search which detects distant relationships between molecules (Id.). When utilizing BLAST, Gapped BLAST, and PSI Blast programs, the default parameters of the respective programs (e.g., of XBLAST and NBLAST) can be used (see, e.g., National Center for Biotechnology Information (NCBI) on the worldwide web at ncbi dot nlm dot nih dot gov). Another non limiting example of a mathematical algorithm utilized for the comparison of sequences is the algorithm of Myers and Miller, 1988, CABIOS 4:11 17. Such an algorithm is incorporated in the ALIGN program (version 2.0), which is part of the GCG sequence alignment software package. When utilizing the ALIGN program for comparing amino acid sequences, a PAM120 weight residue table, a gap length penalty of 12, and a gap penalty of 4 can be used.
[0102] The percent identity between two sequences can be determined using techniques similar to those described above, with or without allowing gaps. In calculating percent identity, typically only exact matches are counted.
[0103] An "antagonist" or "inhibitor" refers to a molecule capable of inhibiting one or more biological activities of a target molecule. Antagonists may interfere with the binding of a receptor to a ligand and vice versa, by incapacitating or killing cells activated by a ligand, and/or by interfering with receptor or ligand activation (e.g., tyrosine kinase activation) or signal transduction after ligand binding to a receptor. The antagonist may completely block receptor-ligand interactions or may substantially reduce such interactions. All such points of intervention by an antagonist shall be considered equivalent for purposes of the instant invention.
[0104] For example, an "antagonist" or "inhibitor" of LIGHT refers to a molecule that is capable of inhibiting or otherwise decreasing one or more of the biological activities of LIGHT, such as in a cell expressing LIGHT or in a cell expressing a LIGHT ligand, such as a LIGHT receptor. For example, in certain embodiments, antibodies of the invention are antagonist antibodies that inhibit or otherwise decrease secretion of CCL20, IL-8, and/or RANTES from a cell having a cell surface-expressed LIGHT receptor (e.g., HVEM, LT.beta.R and/or DcR3) when said antibody is contacted with said cell. In some embodiments, an antagonist of LIGHT (e.g., an antagonistic antibody of the invention) may, for example, act by inhibiting or otherwise decreasing the activation and/or cell signaling pathways of the cell expressing a LIGHT receptor, thereby inhibiting a LIGHT-mediated biological activity of the cell relative to the LIGHT-mediated biological activity in the absence of antagonist. In certain embodiments of the invention, the anti-LIGHT antibodies are fully human, antagonistic anti-LIGHT antibodies, such as fully human, monoclonal, antagonistic anti-LIGHT antibodies.
[0105] For example, an "antagonist" or "inhibitor" of CXCR5 refers to a molecule capable of inhibiting one or more biological activities, such as signaling, by CXCR5. Thus, included within the scope of the invention are antagonists (e.g., neutralizing antibodies) that bind to CXCR5, CXCL13 or other ligands of CXCR5, or a complex of CXCR5 and a ligand thereof, such as CXCL13; amino acid sequence variants or derivatives of CXCR5 or CXCL13 which antagonize the interaction between CXCR5 and a ligand, such as CXCL13; soluble CXCR5, optionally fused to a heterologous molecule such as an immunoglobulin region (e.g., an immunoadhesin); a complex comprising CXCR5 in association with another receptor or biological molecule; synthetic or native sequence peptides which bind to CXCR5; and so on.
[0106] The terms "antibody", "immunoglobulin", or "Ig" may be used interchangeably herein. The term antibody includes, but is not limited to, synthetic antibodies, monoclonal antibodies, recombinantly produced antibodies, multispecific antibodies (including bi-specific antibodies), human antibodies, humanized antibodies, chimeric antibodies, intrabodies, single-chain Fvs (scFv) (e.g., including monospecific, bispecific, etc.), camelized antibodies, Fab fragments, F(ab') fragments, disulfide-linked Fvs (sdFv), anti-idiotypic (anti-Id) antibodies, and epitope-binding fragments of any of the above. In particular, antibodies include immunoglobulin molecules and immunologically active portions of immunoglobulin molecules, i.e., antigen binding domains or molecules that contain an antigen-binding site that specifically binds to a LIGHT antigen (e.g., one or more complementarity determining regions (CDRs) of an anti-LIGHT antibody) or CXCR5 antigen (e.g., one or more complementarity determining regions (CDRs) of an anti-CXCR5 antibody). The anti-LIGHT or anti-CXCR5 antibodies can be of any type (e.g., IgG, IgE, IgM, IgD, IgA and IgY), any class (e.g., IgG1, IgG2, IgG3, IgG4, IgA1 and IgA2), or any subclass (e.g., IgG2a and IgG2b) of immunoglobulin molecule. In some embodiments, the anti-LIGHT antibodies are fully human, such as fully human monoclonal anti-LIGHT antibodies. In certain embodiments, the anti-LIGHT antibodies are IgG antibodies, human IgG4 antibodies. Alternatively, in some embodiments, the anti-CXCR5 antibodies are humanized, such as humanized monoclonal anti-CXCR5 antibodies. In certain embodiments, the anti-CXCR5 antibodies are IgG antibodies, humanized IgG4 antibodies.
[0107] As used herein, the term "anti-LIGHT antibody" means an antibody or polypeptide derived therefrom (a derivative) that binds specifically to human LIGHT as defined herein, including, but not limited to, molecules that inhibit or substantially reduce the binding of LIGHT to its ligands or inhibit LIGHT activity.
[0108] As used herein, the term "anti-CXCR5 antibody" means an antibody or polypeptide derived therefrom (a derivative) that binds specifically to human CXCR5 as defined herein, including, but not limited to, molecules that inhibit or substantially reduce the binding of CXCR5 to its ligands or inhibit CXCR5 activity.
[0109] The term "B cell activity" means higher than normal B cell levels, which can be local, or evidence of a biological manifestation or function of a B cell, such as antibody expression, Bruton's tyrosine kinase presence or activity, expression or presence of CD19, expression or presence of B cell activating factor and so on.
[0110] The term "binding agent" means any molecule, such as an antibody, a siRNA, a nucleic acid, an aptamer, a protein, or a small molecule organic compound, that binds or specifically binds to LIGHT or CXCR5, or a variant or a fragment thereof.
[0111] The term "by-product" includes undesired products, which detract or diminish the proportion of therapeutic/prophylactic binding agent, such as an antibody, in a given formulation. For example, typical by-products include aggregates of the antibody, fragments of the antibody, e.g. produced by degradation of the antibody by deamidation or hydrolysis, or mixtures thereof. Typically, aggregates are complexes that have a molecular weight greater than the monomer antibody. Antibody degradation products may include, for example, fragments of the antibody, for example, brought about by deamidation or hydrolysis. Typically, degradation products are complexes that have a molecular weight less than the monomer antibody. In the case of an IgG antibody, such degradation products are less than about 150 kD.
[0112] The terms "composition" and "formulation" are intended to encompass a product containing the specified ingredients (e.g., an anti-LIGHT antibody or an anti-CXCR5 antibody) in, optionally, the specified amounts, as well as any product that results, directly or indirectly, from the combination of the specified ingredients in, optionally, the specified amounts.
[0113] The terms "constant region" or "constant domain" refer to a carboxy terminal portion of the light and heavy chain which is not directly involved in binding of the antibody to antigen but exhibits various effector functions, such as interaction with the Fc receptor. The terms refer to the portion of an immunoglobulin molecule having a more conserved amino acid sequence relative to the other portion of the immunoglobulin, the variable domain, which contains the antigen binding site. The constant domain contains the CH1, CH2 and CH3 domains of the heavy chain and the CHL domain of the light chain.
[0114] The term "CXCR5" relates to the naturally occurring, known molecule found on lymphocytes, particularly B cells, and particularly naive B cells; to such a molecule isolated from such cells; to such a molecule manufactured recombinantly using known materials and means, and using a nucleic acid encoding a CXCR5; as well as to portions of CXCR5, such as the extracellular (EC) domain, that retain the characteristics and properties relevant to the practice of the instant invention, such as CXCL13 binding. A soluble CXCR5 molecule can consist essentially of the EC domain of CXCR5, which includes, generally, about the first sixty amino acids of the molecule, that is, the amino terminal portion of CXCR5.
[0115] CXCR5 is a non-promiscuous receptor. CXCL13 is a ligand of CXCR5 and is expressed constitutively on stromal cells, such as follicular dendritic cells, and in lymphoid tissues. CXCL13 specifically attracts B cells and a small subset of T cells called B helper follicular T cells, TFH. This may not be unexpected given the many interactions between T cell and B cell populations in the immune system. Moreover, activated T cells induce or upregulate CXCR5 expression. Infiltration of lymphocytes into tertiary, ectopic germinal centers (GCs) has been found to correlate well with increased disease severity and tolerance breakdown in certain disorders that present with such atypical lymph node-like structures. Using in vivo murine models, such as CXCR5-/- and CXCL13-/- mice, the absence of either the receptor or the ligand results in an altered GC fine architecture due to altered T and B cell localization, and possibly interaction. These mice are also protected against developing severe collagen-induced arthritis (CIA). As CXCR5 is selectively expressed on mature B cells, which are linked to the pathogenesis of RA, blocking this receptor will modulate the arthritogenic response in affected individuals. Rheumatoid arthritis treatment with biologics (i.e., anti-TNF.alpha. and anti-CD20 antibodies, Rituximab) has shown to be clinically effective; in particular, patients on B cell-directed therapy have shown long-lasting improvements in clinical signs and symptoms. Selective targeting of CXCR5, which is only expressed on mature B cells and B helper T cells, will not affect B cell development or immunocompromise the patient. Unlike Rituximab, the instant anti-CXCR5 antibody is a neutralizing antibody that does not mediate cell cytotoxicity.
[0116] A "CXCR5 disease" is a malady, disorder, disease, condition, abnormality and so on, that is characterized by or caused by overexpression or increased levels of CXCL13 or other CXCR5 ligand, increased levels of B cells, increased levels of B cell activity, increased levels of CXCR5, or improper metabolism and activity of CXCR5.
[0117] The term "epitope" refers to a localized region on the surface of an antigen, such as a LIGHT or CXCR5 polypeptide, or LIGHT or CXCR5 polypeptide fragment, that is capable of being bound to one or more antigen binding regions of a binding agent, such as an antibody, and that has antigenic or immunogenic activity in an animal, such a mammal, such as in a human, that is capable of eliciting an immune response. An epitope having immunogenic activity is a portion of a polypeptide that elicits an antibody response in an animal. An epitope having antigenic activity is a portion of a polypeptide to which an antibody specifically binds, as determined by any method well known in the art, for example, such as an immunoassay. Antigenic epitopes need not necessarily be immunogenic. Epitopes usually consist of chemically active surface groupings of molecules, such as amino acids or sugar side chains, and have specific three dimensional structural characteristics, as well as specific charge characteristics. A region of a polypeptide contributing to an epitope may be contiguous amino acids of the polypeptide or the epitope may come together from two or more non-contiguous regions of the polypeptide. The epitope may or may not be a three-dimensional surface feature of the antigen. In certain embodiments, a LIGHT or CXCR5 epitope is a three-dimensional surface feature of a LIGHT or CXCR5 polypeptide (e.g., in a trimeric form of a LIGHT polypeptide). In other embodiments, a LIGHT epitope is a linear feature of a LIGHT or CXCR5 polypeptide (e.g., in a trimeric form or monomeric form of the LIGHT polypeptide). Anti-LIGHT or anti-CXCR5 antibodies may specifically bind to an epitope of the monomeric (denatured) form of LIGHT or CXCR5, an epitope of the trimeric (native) form of LIGHT or CXCR5, or both the monomeric (denatured) form and the trimeric (native) form of LIGHT or CXCR5. In specific embodiments, the anti-LIGHT antibodies specifically bind to an epitope of the trimeric form of LIGHT but do not specifically bind the monomeric form of LIGHT.
[0118] The term "excipients" refers to inert substances that are commonly used as a diluent, vehicle, preservative, binder, stabilizing agent, etc. for drugs and includes, but is not limited to, proteins (e.g., serum albumin, etc.), amino acids (e.g., aspartic acid, glutamic acid, lysine, arginine, glycine, histidine, etc.), fatty acids and phospholipids (e.g., alkyl sulfonates, caprylate, etc.), surfactants (e.g., SDS, polysorbate, nonionic surfactant, etc.), saccharides (e.g., sucrose, maltose, trehalose, etc.) and polyols (e.g., mannitol, sorbitol, etc.). See, also, Remington's Pharmaceutical Sciences (1990) Mack Publishing Co., Easton, Pa., which is hereby incorporated by reference in its entirety.
[0119] In the context of a peptide or polypeptide, the term "fragment" refers to a peptide or polypeptide that comprises less than the full length amino acid sequence. Such a fragment may arise, for example, from a truncation at the amino terminus, a truncation at the carboxy terminus, and/or an internal deletion of a residue(s) from the amino acid sequence. Fragments may, for example, result from alternative RNA splicing or from in vivo protease activity. In certain embodiments, hLIGHT or hCXCR5 fragments include polypeptides comprising an amino acid sequence of at least 5 contiguous amino acid residues, at least 10 contiguous amino acid residues, at least 15 contiguous amino acid residues, at least 20 contiguous amino acid residues, at least 25 contiguous amino acid residues, at least 40 contiguous amino acid residues, at least 50 contiguous amino acid residues, at least 60 contiguous amino residues, at least 70 contiguous amino acid residues, at least 80 contiguous amino acid residues, at least 90 contiguous amino acid residues, at least contiguous 100 amino acid residues, at least 125 contiguous amino acid residues, at least 150 contiguous amino acid residues, at least 175 contiguous amino acid residues, at least 200 contiguous amino acid residues, or at least 250 contiguous amino acid residues of the amino acid sequence of a LIGHT or CXCR5 polypeptide or an antibody that specifically binds to a LIGHT or CXCR5 polypeptide. In a specific embodiment, a fragment of a LIGHT or CXCR5 polypeptide or an antibody that specifically binds to a LIGHT or CXCR5 antigen retains at least 1, at least 2, or at least 3 functions of the polypeptide or antibody.
[0120] The terms "fully human antibody" or "human antibody" are used interchangeably herein and refer to an antibody that comprises a human variable region and, possibly a human constant region. In specific embodiments, the terms refer to an antibody that comprises a variable region and constant region of human origin. "Fully human" anti-LIGHT antibodies, in certain embodiments, can also encompass antibodies that bind LIGHT polypeptides and are encoded by nucleic acid sequences that are naturally occurring somatic variants of a human germline immunoglobulin nucleic acid sequence. In a specific embodiment, the anti-LIGHT antibodies are fully human antibodies. The term "fully human antibody" includes antibodies having variable and constant regions corresponding to human germline immunoglobulin sequences as described by Kabat et al. (See Kabat et al. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242). Methods of producing fully human antibodies are known in the art.
[0121] The phrase "recombinant human antibody" includes human antibodies that are prepared, expressed, created, or isolated by recombinant means, such as antibodies expressed using a recombinant expression vector transfected into a host cell, antibodies isolated from a recombinant, combinatorial human antibody library, antibodies isolated from an animal (e.g., a mouse or cow) that is transgenic and/or transchromosomal for human immunoglobulin genes (see, e.g., Taylor, L. D. et al. (1992) Nucl. Acids Res. 20:6287-6295) or antibodies prepared, expressed, created, or isolated by any other means that involves splicing of human immunoglobulin gene sequences to other DNA sequences. Such recombinant human antibodies can have variable and constant regions derived from human germline immunoglobulin sequences (See Kabat, E. A. et al. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242). In certain embodiments, however, such recombinant human antibodies are subjected to in vitro mutagenesis (or, when an animal transgenic for human Ig sequences is used, in vivo somatic mutagenesis) and thus the amino acid sequences of the VH and VL regions of the recombinant antibodies are sequences that, while derived from and related to human germline VH and VL sequences, may not naturally exist within the human antibody germline repertoire in vivo.
[0122] An "IgG4 binding agent" or a "binding agent comprising at least a portion of an IgG4 Fc region" both refer to binding agents described herein that include at least a fragment of IgG4 Fc. In certain embodiments, the fragment comprises 10, 20, 30, 40, 50, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 210 or 220 amino acids of the IgG4 Fc region. In other embodiments, the fragment includes 10-50, 50-100, 100-150, or 150-200 amino acids of the IgG4 Fc region. In other embodiments, the portion of the IgG4 Fc region can have a certain homology to the IgG4 Fc region. For example, the IgG4 binding agent may include a portion of a protein with greater than 50, 60, 70, 80, 90, 93, 95, 96, 97, 98, 99, or 100% homology to the IgG4 Fc region. Exemplary Fc regions of IgG4 are described throughout the specification.
[0123] The term "heavy chain", when used in reference to an antibody, refers to five distinct types, called alpha (.alpha.), delta (.DELTA.), epsilon (.epsilon.), gamma (.gamma.), and mu (.mu.), based on the amino acid sequence of the heavy chain constant domain. These distinct types of heavy chains are well known in the art and give rise to five classes of antibodies, IgA, IgD, IgE, IgG, and IgM, respectively, including four subclasses of IgG, namely IgG1, IgG1, IgG3, and IgG4. In some embodiments, the heavy chain is a human heavy chain.
[0124] "Humanized" forms of non-human (e.g., murine) antibodies are chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as F.sub.v, F.sub.ab, F.sub.ab', F.sub.(ab')2 or other target-binding subsequences of antibodies) that contain sequences derived from non-human immunoglobulin, as compared to a human antibody. In general, the humanized antibody will comprise substantially all of one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or substantially all of the FR regions are those of a human immunoglobulin template sequence. The humanized antibody may also comprise at least a portion of an immunoglobulin constant region (Fc), typically that of the human immunoglobulin template chosen. In general, the goal is to have an antibody molecule that is minimally immunogenic in a human. Thus, it is possible that one or more amino acids in one or more CDRs also can be changed to one that is less immunogenic to a human host, without substantially minimizing the specific binding function of the one or more CDRs to CXCR5 or to CXCL13. Alternatively, the FR can be non-human but those amino acids most immunogenic are replaced with ones less immunogenic. Nevertheless, CDR grafting, as discussed above, is not the only way to obtain a humanized antibody. For example, modifying just the CDR regions may be insufficient as it is not uncommon for framework residues to have a role in determining the three-dimensional structure of the CDR loops and the overall affinity of the antibody for its ligand. Hence, any means can be practiced so that the non-human parent antibody molecule is modified to be one that is less immunogenic to a human, and global sequence identity with a human antibody is not always a necessity. So, humanization also can be achieved, for example, by the mere substitution of just a few residues, particularly those which are exposed on the antibody molecule and not buried within the molecule, and hence, not readily accessible to the host immune system. Such a method is taught herein with respect to substituting "mobile" or "flexible" residues on the antibody molecule, the goal being to reduce or dampen the immunogenicity of the resultant molecule without comprising the specificity of the antibody for its epitope or determinant. See, for example, Studnicka et al., Prot Eng 7(6)805-814, 1994; Mol Imm 44:1986-1988, 2007; Sims et al., J Immunol 151:2296 (1993); Chothia et al., J Mol Biol 196:901 (1987); Carter et al., Proc Natl Acad Sci USA 89:4285 (1992); Presta et al., J Immunol 151:2623 (1993), WO 2006/042333 and U.S. Pat. No. 5,869,619.
[0125] An "isolated" or "purified" binding agent, such as an antibody, is substantially free of cellular material or other contaminating proteins from the cell or tissue source from which the binding agent is derived, or substantially free of chemical precursors or other chemicals when chemically synthesized. For example, the language "substantially free of cellular material" includes preparations of an antibody in which the antibody is separated from cellular components of the cells from which it is isolated or recombinantly produced. Thus, an antibody that is substantially free of cellular material includes preparations of antibody having less than about 30%, 20%, 10%, or 5% (by dry weight) of heterologous protein (also referred to herein as a "contaminating protein"). When the antibody is recombinantly produced, it is also desirable to be substantially free of culture medium, i.e., culture medium represents less than about 20%, 10%, or 5% of the volume of the protein preparation. When the antibody is produced by chemical synthesis, in some embodiments it is substantially free of chemical precursors or other chemicals, i.e., it is separated from chemical precursors or other chemicals that are involved in the synthesis of the protein. Accordingly, such preparations of the antibody have less than about 30%, 20%, 10%, 5% (by dry weight) of chemical precursors or compounds other than the antibody of interest. In some embodiments, anti-LIGHT or anti-CXCR5 antibodies are isolated or purified.
[0126] The term "human LIGHT," "hLIGHT" or "hLIGHT polypeptide" and similar terms refer to the polypeptides ("polypeptides," "peptides" and "proteins" are used interchangeably herein) comprising the amino acid sequence of SEQ ID NO: 9 and related polypeptides, including SNP variants thereof. Related polypeptides include allelic variants (e.g., SNP variants); splice variants; fragments; derivatives; substitution, deletion, and insertion variants; fusion polypeptides; and interspecies homologs, in some embodiments, which retain LIGHT activity and/or are sufficient to generate an anti-LIGHT immune response. Also encompassed are soluble forms of LIGHT that are sufficient to generate an anti-LIGHT immunological response. As those skilled in the art will appreciate, an anti-LIGHT binding agent, such as an antibody, can bind to a LIGHT polypeptide, polypeptide fragment, antigen, and/or epitope, as an epitope is part of the larger antigen, which is part of the larger polypeptide fragment, which, in turn, is part of the larger polypeptide. hLIGHT can exist in a trimeric (native) or monomeric (denatured) form.
[0127] The term "human CXCR5," "hCXCR5" or "hCXCR5 polypeptide" and similar terms refer to the polypeptides ("polypeptides," "peptides" and "proteins" are used interchangeably herein) comprising the amino acid sequence of SEQ ID NO: 14 and related polypeptides, including SNP variants thereof. Related polypeptides include allelic variants (e.g., SNP variants); splice variants; fragments; derivatives; substitution, deletion, and insertion variants; fusion polypeptides; and interspecies homologs, in some embodiments, which retain CXCR5 activity and/or are sufficient to generate an anti-CXCR5 immune response. Also encompassed are soluble forms of CXCR5 that are sufficient to generate an anti-CXCR5 immunological response. As those skilled in the art will appreciate, an anti-CXCR5 binding agent, such as an antibody, can bind to a CXCR5 polypeptide, polypeptide fragment, antigen, and/or epitope, as an epitope is part of the larger antigen, which is part of the larger polypeptide fragment, which, in turn, is part of the larger polypeptide.
[0128] The term "Kabat numbering," and like terms are recognized in the art and refer to a system of numbering amino acid residues that are more variable (i.e. hypervariable) than other amino acid residues in the heavy and light chain variable regions of an antibody, or an antigen binding portion thereof (Kabat et al. (1971) Ann. NY Acad. Sci. 190:382-391 and, Kabat et al. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242). For the heavy chain variable region, the hypervariable region typically ranges from amino acid positions 31 to 35 for CDR1, amino acid positions 50 to 65 for CDR2, and amino acid positions 95 to 102 for CDR3. For the light chain variable region, the hypervariable region typically ranges from amino acid positions 24 to 34 for CDR1, amino acid positions 50 to 56 for CDR2, and amino acid positions 89 to 97 for CDR3.
[0129] The term "light chain" when used in reference to an antibody refers to two distinct types, called kappa (.kappa.) of lambda (.lamda.) based on the amino acid sequence of the constant domains. Light chain amino acid sequences are well known in the art. In some embodiments, the light chain is a human light chain.
[0130] The terms "manage", "managing", and "management" refer to the beneficial effects that a subject derives from a therapy (e.g., a prophylactic or therapeutic agent), which does not result in a cure of the infection. In certain embodiments, a subject is administered one or more therapies (e.g., prophylactic or therapeutic agents, such as a formulation of the invention) to "manage" a LIGHT-mediated disease (e.g., chronic bowel disease, IBD, Crohn's disease, ulcerative colitis, or GVHD) or CXCR5-mediated disease (e.g., rheumatoid arthritis), one or more symptoms thereof, so as to prevent the progression or worsening of the disease.
[0131] The term "monoclonal antibody" refers to an antibody obtained from a population of homogenous or substantially homogeneous antibodies, and each monoclonal antibody will typically recognize a single epitope on the antigen. In some embodiments, a "monoclonal antibody" is an antibody produced by a single hybridoma or other cell. The term "monoclonal" is not limited to any particular method for making the antibody. For example, monoclonal antibodies may be made by the hybridoma method as described in Kohler et al.; Nature, 256:495 (1975) or may be isolated from phage libraries. Other methods for the preparation of clonal cell lines and of monoclonal antibodies expressed thereby are well known in the art (see, for example, Chapter 11 in: Short Protocols in Molecular Biology, (2002) 5th Ed.; Ausubel et al., eds., John Wiley and Sons, New York).
[0132] The term "pharmaceutically acceptable" means being approved by a regulatory agency of the Federal or a state government, or listed in the U.S. Pharmacopeia, European Pharmacopeia or other generally recognized Pharmacopeia for use in animals, and more particularly in humans.
[0133] By "pharmaceutically acceptable excipient" is meant any inert substance that is combined with an active molecule, such as a monoclonal antibody, for preparing an agreeable or convenient dosage form. The "pharmaceutically acceptable excipient" is an excipient that is non-toxic to recipients at the dosages and concentrations employed, and is compatible with other ingredients of the formulation comprising the monoclonal antibody.
[0134] The terms "prevent", "preventing", and "prevention" refer to the total or partial inhibition of the development, recurrence, onset or spread of a LIGHT-mediated or CXCR5-mediated disease and/or symptom related thereto, resulting from the administration of a therapy or combination of therapies provided herein (e.g., a combination of prophylactic or therapeutic agents, such as a formulation of the invention).
[0135] The term "prophylactic agent" refers to any agent that can totally or partially inhibit the development, recurrence, onset or spread of a LIGHT-mediated or CXCR5-mediated disease and/or symptom related thereto in a subject. In certain embodiments, the term "prophylactic agent" refers to a formulation of the invention. In certain other embodiments, the term "prophylactic agent" refers to an agent other than a formulation of the invention. In some embodiments, a prophylactic agent is an agent that is known to be useful to or has been or is currently being used to prevent a LIGHT-mediated or CXCR5-mediated disease and/or a symptom related thereto, or impede the onset, development, progression and/or severity of a LIGHT-mediated or CXCR5-mediated disease and/or a symptom related thereto. In specific embodiments, the prophylactic agent is a fully human anti-LIGHT antibody, such as a fully human anti-LIGHT monoclonal antibody, or a humanized anti-CXCR5 antibody, such as a humanized anti-CXCR5 monoclonal antibody.
[0136] The term "LIGHT antigen" refers to that portion of a LIGHT polypeptide to which a binding agent, such as an antibody, specifically binds. A LIGHT antigen also refers to an analog or derivative of a LIGHT polypeptide or fragment thereof to which a binding agent, such as an antibody, specifically binds. In some embodiments, a LIGHT antigen is a monomeric LIGHT antigen or a trimeric LIGHT antigen. A region of a LIGHT polypeptide contributing to an epitope may be contiguous amino acids of the polypeptide, or the epitope may come together from two or more non-contiguous regions of the polypeptide. The epitope may or may not be a three-dimensional surface feature of the antigen. A localized region on the surface of a LIGHT antigen that is capable of eliciting an immune response is a LIGHT epitope. The epitope may or may not be a three-dimensional surface feature of the antigen.
[0137] The term "CXCR5 antigen" refers to that portion of a CXCR5 polypeptide to which a binding agent, such as an antibody, specifically binds. A CXCR5 antigen also refers to an analog or derivative of a CXCR5 polypeptide or fragment thereof to which a binding agent, such as an antibody, specifically binds. A region of a CXCR5 polypeptide contributing to an epitope may be contiguous amino acids of the polypeptide, or the epitope may come together from two or more non-contiguous regions of the polypeptide. The epitope may or may not be a three-dimensional surface feature of the antigen. A localized region on the surface of a CXCR5 antigen that is capable of eliciting an immune response is a CXCR5 epitope. The epitope may or may not be a three-dimensional surface feature of the antigen.
[0138] The terms "LIGHT-mediated disease" and "LIGHT-mediated disorder" are used interchangeably and refer to any disease that is completely or partially caused by or is the result of LIGHT. In certain embodiments, LIGHT is aberrantly (e.g., highly) expressed on the surface of a cell. In some embodiments, LIGHT may be aberrantly upregulated on a particular cell type. In other embodiments, normal, aberrant, or excessive cell signaling is caused by binding of LIGHT to a LIGHT ligand. In certain embodiments, the LIGHT ligand is a LIGHT receptor (e.g., HVEM, LT.beta.R, or DCR3), for example, that is expressed on the surface of a cell, such as a colonic epithelial cell. In certain embodiments, the LIGHT-mediated disease is a chronic bowel disease, an inflammatory bowel disease (IBD), such as Crohn's disease (CD) or ulcerative colitis (UC). In other embodiments, the LIGHT-mediated disease is graft-versus-host disease (GVHD).
[0139] The terms "CXCR5-mediated disease" and "CXCR5-mediated disorder" are used interchangeably and refer to any disease that is completely or partially caused by or is the result of CXCR5. In certain embodiments, CXCR5 is aberrantly (e.g., highly) expressed on the surface of a cell. In some embodiments, CXCR5 may be aberrantly upregulated on a particular cell type. In other embodiments, normal, aberrant, or excessive cell signaling is caused by binding of CXCR5 to a CXCR5 ligand. In certain embodiments, the CXCR5 ligand is CXCL13. In certain embodiments, the CXCR5-mediated disease is rheumatoid arthritis (RA).
[0140] The term "saccharide" refers to a class of molecules that are derivatives of polyhydric alcohols. Saccharides are commonly referred to as carbohydrates and may contain different amounts of sugar (saccharide) units, e.g., monosaccharides, disaccharides, and polysaccharides.
[0141] The terms "specifically binds" or "specifically binding" mean specifically binding to an antigen or a fragment thereof and not specifically binding to other antigens. For example, an antibody that specifically binds to an antigen may bind to other peptides or polypeptides with lower affinity, as determined by, e.g., radioimmunoassays (RIA), enzyme-linked immunosorbent assays (ELISA), BIACORE, or other assays known in the art. Antibodies or variants or fragments thereof that specifically bind to an antigen may be cross-reactive with related antigens. In some embodiments, antibodies or variants or fragments thereof that specifically bind to an antigen do not cross-react with other antigens. An antibody or a variant or a fragment thereof that specifically binds to a LIGHT or CXCR5 antigen can be identified, for example, by immunoassays, BIAcore, or other techniques known to those of skill in the art. Typically a specific or selective reaction will be at least twice background signal or noise, and more typically more than 10 times background. See, e.g., Paul, ed., 1989, Fundamental Immunology Second Edition, Raven Press, New York at pages 332-336 for a discussion regarding antibody specificity.
[0142] A "stable" or "stabilized" formulation is one in which the binding agent, such as an antibody, therein essentially retains its physical stability, identity, integrity, and/or chemical stability, identity, integrity, and/or biological activity upon storage. Various analytical techniques for measuring protein stability are available in the art and are reviewed in Peptide and Protein Drug Delivery, 247-301, Vincent Lee Ed., Marcel Dekker, Inc., New York, N.Y., Pubs. (1991) and Jones, A. Adv. Drug Delivery Rev. 10:29-90 (1993), for example. Stability can be measured at a selected temperature and other storage conditions for a selected time period. The stability may be determined by at least one of the methods selected from the group consisting of visual inspection, SDS-PAGE, IEF, HPSEC, RFFIT, and kappa/lambda ELISA. For example, an antibody "retains its physical stability" in a pharmaceutical formulation, if it shows no signs of aggregation, precipitation, and/or denaturation upon visual examination of color and/or clarity, or as measured by UV light scattering, SDS-PAGE, or by (high pressure) size exclusion chromatography (HPSEC). In some embodiments, when using the formulations of the invention, 5% or less, typically 4% or less, typically 3% or less, more typically 2% or less, and particularly 1% or less of the antibodies forms aggregates, as measured by HPSEC or any other suitable method for measuring aggregation formation. For example, an antibody is considered stable in a particular formulation if the antibody monomer has a purity of about 90%, typically about 95%, in particular about 98% after a certain predetermined period of time under certain storage conditions in a particular formulation. Chemical stability can be assessed by detecting and quantifying chemically altered forms of the protein. Chemical alteration may involve size modification (e.g., clipping), which can be evaluated using (HP)SEC, SDS-PAGE, and/or matrix-assisted laser desorption ionization/time-of-flight mass spectrometry (MALDI/TOF MS), for example. Other types of chemical alteration include charge alteration (e.g., occurring as a result of deamidation), which can be evaluated by ion-exchange chromatography, for example. An antibody "retains its biological activity" in a pharmaceutical formulation at a given time, if the biological activity of the antibody at a given time is at least about 90% (within the errors of the assay) of the biological activity exhibited at the time the pharmaceutical formulation was prepared, as determined in an antigen binding assay or virus neutralizing assay, for example.
[0143] The terms "subject" and "patient" are used interchangeably. As used herein, a subject is typically a mammal, such as a non-primate (e.g., cows, pigs, horses, cats, dogs, rats, etc.) or a primate (e.g., monkey and human), and in some embodiments a human. In one embodiment, the subject is a mammal, such as a human, having a LIGHT-mediated or CXCR5-mediated disease. In another embodiment, the subject is a mammal, such as a human, at risk of developing a LIGHT-mediated or CXCR5-mediated disease.
[0144] The term "therapeutically effective amount" refers to the amount of a therapy (e.g., a formulation of the invention) that is sufficient to reduce and/or ameliorate the severity and/or duration of a given disease and/or a symptom related thereto. This term also encompasses an amount necessary for the reduction or amelioration of the advancement or progression of a given disease, reduction or amelioration of the recurrence, development or onset of a given disease, and/or to improve or enhance the prophylactic or therapeutic effect(s) of another therapy (e.g., a therapy other than a formulation of the invention). In some embodiments, the therapeutically effective amount of an antibody of the invention is from about 0.1 mg/kg (mg of antibody per kg weight of the subject) to about 100 mg/kg. In certain embodiments, a therapeutically effective amount of an antibody provided therein is about 0.1 mg/kg, about 0.5 mg/kg, about 1 mg/kg, 3 mg/kg, 5 mg/kg, about 10 mg/kg, about 15 mg/kg, about 20 mg/kg, about 25 mg/kg, about 30 mg/kg, about 35 mg/kg, about 40 mg/kg, about 45 mg/kg, about 50 mg/kg, about 60 mg/kg, about 70 mg/kg, about 80 mg/kg about 90 mg/kg or about 100 mg/kg (or a range therein). In some embodiments, "therapeutically effective amount" as used herein also refers to the amount of an antibody of the invention to achieve a specified result (e.g., inhibition of a LIGHT biological activity of a cell, such as inhibition of secretion of CCL20, IL-8, or RANTES from the cell; or inhibition of a CXCR5 biological activity of a cell, such as binding to CXCL13).
[0145] The term "therapeutic agent" refers to any agent that can be used in the treatment, management or amelioration of a LIGHT-mediated or CXCR5-mediated disease and/or a symptom related thereto. In certain embodiments, the term "therapeutic agent" refers to a formulation of the invention. In certain other embodiments, the term "therapeutic agent" refers to an agent other than a formulation of the invention. In some embodiments, a therapeutic agent is an agent that is known to be useful for, or has been or is currently being used for the treatment, management or amelioration of a LIGHT-mediated or CXCR5-mediated disease or one or more symptoms related thereto.
[0146] The term "therapy" refers to any protocol, method, and/or agent that can be used in the prevention, management, treatment, and/or amelioration of a LIGHT-mediated disease (e.g., IBD or GVHD) or CXCR5-mediated disease (e.g., rheumatoid arthritis). In certain embodiments, the terms "therapies" and "therapy" refer to a biological therapy, supportive therapy, and/or other therapies useful in the prevention, management, treatment, and/or amelioration of a LIGHT-mediated or CXCR5-mediated disease known to one of skill in the art, such as medical personnel.
[0147] The terms "treat", "treatment", and "treating" refer to the reduction or amelioration of the progression, severity, and/or duration of a LIGHT-mediated disease (e.g., chronic bowel disease, IBD, or GVHD) or CXCR5-mediated disease (e.g., rheumatoid arthritis) resulting from the administration of one or more therapies (including, but not limited to, the administration of one or more prophylactic or therapeutic agents, such as a formulation of the invention). In specific embodiments for LIGHT, such terms refer to the reduction or inhibition of the binding of LIGHT to HVEM, the reduction or inhibition of the binding of LIGHT to LT.beta.R, the reduction or inhibition of the binding of LIGHT to DcR3, the reduction or inhibition of the production or secretion of CCL20 from a cell expressing a LIGHT receptor of a subject, the reduction or inhibition of the production or secretion of IL-8 from a cell expressing a LIGHT receptor of a subject, the reduction or inhibition of the production or secretion of RANTES from a cell expressing a LIGHT receptor of a subject, and/or the inhibition or reduction of one or more symptoms associated with a LIGHT-mediated disease, such as a chronic bowel disease, IBD, or GVHD. In specific embodiments for CXCR5, such terms refer to the reduction or inhibition of the binding of CXCR5 to CXCL13, and/or the inhibition or reduction of one or more symptoms associated with a CXCR5-mediated disease, such as rheumatoid arthritis.
[0148] The terms "variable region" or "variable domain" refer to a portion of the light and heavy chains, typically about the amino-terminal 120 to 130 amino acids in the heavy chain and about 100 to 110 amino acids in the light chain, which differ extensively in sequence among antibodies and are used in the binding and specificity of each particular antibody for its particular antigen. The variability in sequence is concentrated in those regions called complementarity determining regions (CDRs), while the more highly conserved regions in the variable domain are called framework regions (FR). The CDRs of the light and heavy chains are primarily responsible for the interaction of the antibody with antigen. Numbering of amino acid positions is according to the EU Index, as in Kabat et al. (1991) Sequences of proteins of immunological interest. (U.S. Department of Health and Human Services, Washington, D.C.) 5.sup.th ed. ("Kabat et al."). In some embodiments, the variable region is a human variable region.
B. Formulations and Formulation Components
[0149] As stated previously, the formulations of the invention have surprisingly been found in the form of liquids and lyophilized powders that comprise an IgG4 binding agent and a citrate buffer, wherein the pH of the formulation is at or below both about pH 6 and the isoelectric point (pI) of the binding agent. The formulations of the invention provide significant improvements over conventional IgG4 binding agent formulations (e.g., phosphate buffered saline (PBS) formulations), which form unwanted byproducts upon increasing the concentration of the binding agent in the formulation. In particular, the formulations of the invention have a reduced amount of aggregates, half-molecules, degradation products, low molecular weight proteins (LMWPs), high molecular weight proteins (HMWPs), and rearrangements of acid, basic, and neutral isoforms of the binding agent in the formulations.
i. Anti-LIGHT Binding Agents, and Variants and Fragments Thereof.
[0150] In certain embodiments, the formulations of the invention include an anti-LIGHT binding agent. The binding agents may be any molecule, such as an antibody, a siRNA, a nucleic acid, an aptamer, a protein, or a small molecule organic compound, that binds or specifically binds to LIGHT, or a variant or a fragment thereof. In some embodiments, the binding agent is an anti-LIGHT antibody, or a variant thereof, or an antigen binding fragment thereof. Anti-LIGHT antibodies specifically bind to a LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) protein, polypeptide fragment, or epitope. The LIGHT molecule may be from any species. In some embodiments, the LIGHT molecule is from a human, known herein as "hLIGHT". hLIGHT has the following amino acid sequence, which is identified as SEQ ID NO: 9:
TABLE-US-00001 (SEQ ID NO: 9) 1 MEESVVRPSV FVVDGQTDIP FTRLGRSHRR QSCSVARVGL GLLLLLMGAG 51 LAVQGWFLLQ LHWRLGEMVT RLPDGPAGSW EQLTQERRSH EVNPAAHLTG 101 ANSSLTGSGG PLLWETQLGL AFLRGLSYHD GALVVTKAGY YYIYSKVQLG 150 GVGCPLGLAS TITHGLYKRT PRYPEELELL VSQQSPCGRA TSSSRVWWDS 200 SFLGGVVHLE AGEEVVVRVL DERLVRLRDG TRSYFGAFMV
[0151] In certain exemplary embodiments, the anti-LIGHT antibody is a humanized antibody, a fully human antibody, or a variant thereof or an antigen-binding fragment thereof. In some embodiments, anti-LIGHT antibodies prevent binding of LIGHT with its receptors and inhibit LIGHT biological activity (e.g., the LIGHT-mediated production or secretion of CCL20, IL-8, or RANTES from cells expressing a LIGHT ligand, such as a LIGHT receptor (e.g., HVEM, LT.beta.R, and/or DcR3).
[0152] In certain specific embodiments, the anti-LIGHT antibody comprises a heavy chain variable region (VH) comprising the amino acid sequence of any one or more of the following complementary determining regions (CDRs):
TABLE-US-00002 (SEQ ID NO: 1) HCDR1 - GYNWH; (SEQ ID NO: 2) HCDR2 - EITHSGSTNYNPSLKS; or (SEQ ID NO: 3) HCDR3 - EIAVAGTGYYGMDV.
[0153] In other specific embodiments, the anti-LIGHT antibody comprises a light chain variable region (VL) comprising the amino acid sequence of any one or more of the following complementary determining regions (CDRs):
TABLE-US-00003 (SEQ ID NO: 4) LCDR1 - RASQGINSAFA; (SEQ ID NO: 5) LCDR2 - DASSLES; or (SEQ ID NO: 6) LCDR3 - QQFNSYPLT.
[0154] In one specific embodiment, the anti-LIGHT antibody comprises a heavy chain variable region (VH) comprising the amino acid sequences of SEQ ID NOs: 1, 2, and 3.
[0155] In another specific embodiment, the anti-LIGHT antibody comprises a light chain variable region (VL) comprising the amino acid sequences of SEQ ID NOs: 4, 5, and 6.
[0156] In more specific embodiments, the anti-LIGHT antibody comprises a heavy chain variable region comprising the amino acid sequences of SEQ ID NOs: 1, 2, and 3; and a light chain variable region comprising the amino acid sequences of SEQ ID NOs: 4, 5, and 6.
[0157] In specific embodiments, the anti-LIGHT antibody comprises a heavy chain comprising the amino acid sequence of SEQ ID NO: 7:
TABLE-US-00004 (SEQ ID NO: 7) 1 QVQLQQWGAG LLKPSETLSL TCAVYGGSFS GYNWHWIRQP PGKGLEWIGE 51 ITHSGSTNYN PSLKSRVTIS VDTSKNQFSL KLSSVTAADT AVYYCVREIA 101 VAGTGYYGMD VWGQGTTVTV SSASTKGPSV FPLAPCSRST SESTAALGCL 151 VKDYFPEPVT VSWNSGALTS GVHTFPAVLQ SSGLYSLSSV VTVPSSSLGT 201 KTYTCNVDHK PSNTKVDKRV ESKYGPPCPP CPAPEFEGGP SVFLFPPKPK 251 DTLMISRTPE VTCVVVDVSQ EDPEVQFNWY VDGVEVHNAK TKPREEQFNS 301 TYRVVSVLTV LHQDWLNGKE YKCKVSNKGL PSSIEKTISK AKGQPREPQV 351 YTLPPSQEEM TKNQVSLTCL VKGFYPSDIA VEWESNGQPE NNYKTTPPVL 401 DSDGSFFLYS RLTVDKSRWQ EGNVFSCSVM HEALHNHYTQ KSLSLSLG
[0158] Positions 1-122: variable region of the heavy chain (VH). The CDRs (complementary determining regions, according to Kabat definition) are underlined. [0159] Positions 123-448: constant region of human IgG4 (SwissProt IGHG4_HUMAN with the two mutations S241P and L248E, according to Kabat numbering).
[0160] In other specific embodiments, the anti-LIGHT antibody comprises a light chain comprising the amino acid sequence of SEQ ID NO: 8:
TABLE-US-00005 (SEQ ID NO: 8) 1 AIQLTQSPSS LSASVGDRVT ITCRASQGIN SAFAWYQQKP GKAPKLLIYD 51 ASSLESGVPS RFSGSGSGTD FTLTISSLQP EDFATYYCQQ FNSYPLTFGG 101 GTKVEIKRTV AAPSVFIFPP SDEQLKSGTA SVVCLLNNFY PREAKVQWKV 151 DNALQSGNSQ ESVTEQDSKD STYSLSSTLT LSKADYEKHK VYACEVTHQG 201 LSSPVTKSFN RGEC
[0161] Positions 1-107: variable region of the light chain (VL). The CDRs (complementary determining regions, according to Kabat definition) are underlined. [0162] Positions 108-214: constant region of human Cc.
[0163] In further embodiments, the anti-LIGHT antibody comprises a heavy chain comprising the amino acid sequence of SEQ ID NO: 7, and a light chain comprising the amino acid sequence of SEQ ID NO: 8.
[0164] In certain embodiments, the anti-LIGHT antibody comprises a heavy chain derived from the amino acid sequence of SEQ ID NO: 10, which may be encoded by the nucleic acid sequence of SEQ ID NO: 11.
TABLE-US-00006 M K H L W F F L L L V A A P R W V L S Q V Q L Q Q W G .cndot. 1 ATGAAGCACCTGTGGTTCTTTCTGCTGCTGGTGGCCGCTCCTAGATGGGTGCTGTCCCAGGTGCAGCTG- CAGCAGTGGGG .cndot. A G L L K P S E T L S L T C A V Y G G S F S G Y N W H .cndot. 81 CGCTGGCCTGCTGAAGCCTTCCGAGACACTGTCCCTGACCTGCGCCGTGTACGGCGGCTCCTTCTCCGG- CTACAACTGGC .cndot. W I R Q P P G K G L E W I G E I T H S G S T N Y N P 161 ACTGGATCAGGCAGCCTCCCGGCAAGGGCCTGGAATGGATCGGCGAGATCACCCACTCCGGCTCCACCA- ACTACAACCCT S L K S R V T I S V D T S K N Q F S L K L S S V T A A .cndot. 241 AGCCTGAAGTCCAGAGTGACCATCTCCGTGGACACCTCCAAGAACCAGTTCTCCCTGAAGCTGTCCTCT- GTGACCGCCGC .cndot. D T A V Y Y C V R E I A V A G T G Y Y G M D V W G Q G .cndot. 321 TGACACCGCCGTGTACTACTGTGTGCGGGAGATCGCCGTGGCTGGCACCGGCTACTACGGCATGGATGT- GTGGGGCCAGG .cndot. T T V T V S S A S T K G P S V F P L A P C S R S T S 401 GCACCACCGTGACCGTGTCCAGCGCTTCTACCAAGGGCCCTTCCGTGTTCCCTCTGGCCCCTTGCTCCC- GGTCCACCTCC E S T A A L G C L V K D Y F P E P V T V S W N S G A L .cndot. 481 GAGTCCACCGCCGCTCTGGGCTGCCTGGTGAAGGACTACTTCCCTGAGCCTGTGACCGTGTCCTGGAAC- TCTGGCGCCCT .cndot. T S G V H T F P A V L Q S S G L Y S L S S V V T V P S .cndot. 561 GACCTCCGGCGTGCACACCTTCCCTGCCGTGCTGCAGTCCTCCGGCCTGTACTCCCTGTCCTCCGTGGT- GACCGTGCCTT .cndot. S S L G T K T Y T C N V D H K P S N T K V D K R V E 641 CCTCCTCCCTGGGCACCAAGACCTACACCTGTAACGTGGACCACAAGCCTTCCAACACCAAGGTGGACA- AGCGGGTGGAG S K Y G P P C P P C P A P E F E G G P S V F L F P P K .cndot. 721 TCCAAGTACGGCCCTCCTTGCCCTCCCTGCCCTGCCCCTGAGTTCGAGGGCGGACCTAGCGTGTTCCTG- TTCCCTCCTAA .cndot. P K D T L M I S R T P E V T C V V V D V S Q E D P E V .cndot. 801 GCCTAAGGACACCCTGATGATCTCCCGGACCCCTGAGGTGACCTGTGTGGTGGTGGACGTGTCCCAGGA- GGACCCTGAGG .cndot. Q F N W Y V D G V E V H N A K T K P R E E Q F N S T 881 TCCAGTTCAACTGGTACGTGGACGGCGTGGAGGTGCACAACGCCAAGACCAAGCCTCGGGAGGAGCAGT- TCAATTCCACC Y R V V S V L T V L H Q D W L N G K E Y K C K V S N K .cndot. 961 TACCGGGTGGTGTCTGTGCTGACCGTGCTGCACCAGGACTGGCTGAACGGCAAAGAATACAAGTGTAAG- GTCTCCAACAA .cndot. G L P S S I E K T I S K A K G Q P R E P Q V Y T L P P .cndot. 1041 GGGCCTGCCCTCCTCCATCGAGAAAACCATCTCCAAGGCCAAGGGCCAGCCTAGGGAGCCTCAGGTGTA- CACCCTGCCTC .cndot. S Q E E M T K N Q V S L T C L V K G F Y P S D I A V 1121 CTAGCCAGGAAGAGATGACCAAGAACCAGGTGTCCCTGACCTGTCTGGTGAAGGGCTTCTACCCTTCCG- ACATCGCCGTG E W E S N G Q P E N N Y K T T P P V L D S D G S F F L .cndot. 1201 GAGTGGGAGTCCAACGGCCAGCCTGAGAACAACTACAAGACCACCCCTCCTGTGCTGGACTCCGACGGC- TCCTTCTTCCT .cndot. Y S R L T V D K S R W Q E G N V F S C S V M H E A L H .cndot. 1281 GTACTCCAGGCTGACCGTGGACAAGTCCCGGTGGCAGGAGGGCAACGTCTTTTCCTGCTCCGTGATGCA- CGAGGCCCTGC (SEQ ID NO: 10) .cndot. N H Y T Q K S L S L S L G * (SEQ ID NO: 11) 1361 ACAACCACTACACCCAGAAGTCCCTGTCCCTGTCTCTGGGCTGA
Amino acids 1-19: signal peptide Amino acids 20-141: 124F19k2 variable region (VH) Amino acids 142-end: hIgG4 PE constant region Nucleic acids 1-57: nucleic acids encoding the signal peptide Nucleic acids 58-423: nucleic acids encoding the 124F19k2 variable region (VH) Nucleic acids 424-end: nucleic acids encoding the hIgG4 PE constant region
[0165] In alternative specific embodiments, the anti-LIGHT antibody comprises a light chain derived from the amino acid sequence of SEQ ID NO: 12, which may be encoded by the nucleic acid sequence of SEQ ID NO: 13.
TABLE-US-00007 M D M R V P A Q L L G L L L L W L P G A R C A I Q L T .cndot. 1 ATGGACATGAGAGTGCCTGCTCAGCTGCTGGGACTGCTGCTGCTGTGGCTGCCTGGCGCTAGATGCGCCA- TCCAGCTGAC .cndot. Q S P S S L S A S V G D R V T I T C R A S Q G I N S A .cndot. 81 CCAGTCCCCCTCCTCTCTGTCCGCCTCCGTGGGCGACAGAGTGACCATCACCTGTCGGGCCTCCCAGGGC- ATCAACTCCG .cndot. F A W Y Q Q K P G K A P K L L I Y D A S S L E S G V 161 CCTTCGCCTGGTATCAGCAGAAGCCTGGCAAGGCCCCTAAGCTGCTGATCTACGACGCCTCCTCCCTGGA- ATCCGGCGTG P S R F S G S G S G T D F T L T I S S L Q P E D F A T .cndot. 241 CCCTCCAGATTTTCCGGCTCCGGCTCTGGCACCGACTTCACCCTGACCATCTCCAGCCTGCAGCCTGAGG- ACTTCGCCAC .cndot. Y Y C Q Q F N S Y P L T F G G G T K V E I K R T V A A .cndot. 321 CTACTACTGCCAGCAGTTCAACTCCTACCCTCTGACCTTCGGCGGAGGCACCAAGGTGGAGATCAAGCGT- ACGGTGGCTG .cndot. P S V F I F P P S D E Q L K S G T A S V V C L L N N 401 CACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTGTTGTGTGCCT- GCTGAATAAC F Y P R E A K V Q W K V D N A L Q S G N S Q E S V T E .cndot. 481 TTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTCCAATCGGGTAACTCCCAGGAGA- GTGTCACAGA .cndot. Q D S K D S T Y S L S S T L T L S K A D Y E K H K V Y .cndot. 561 GCAGGACAGCAAGGACAGCACCTACAGCCTCAGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAA- CACAAAGTCT (SEQ ID NO: 12) .cndot. A C E V T H Q G L S S P V T K S F N R G E C * (SEQ ID NO: 13) 641 ACGCCTGCGAAGTCACCCATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTA- G
Amino acids 1-22: signal peptide Amino acids 23-129: 124F19k2 variable region (VL) Amino acids 130-end: hKappa constant region Nucleic acids 1-66: nucleic acids encoding the signal peptide Nucleic acids 67-387: nucleic acids encoding the 124F19k2 variable region (VL) Nucleic acids 388-end: nucleic acids encoding the hKappa constant region
[0166] In an embodiment of the invention, the anti-LIGHT antibody is a fully human antibody. Examples of fully human antibody isotypes include IgA, IgD, IgE, IgG, and IgM. In some embodiments, the anti-LIGHT antibody is an IgG antibody. There are four forms of IgG. In some embodiments, the anti-LIGHT antibody is an IgG4 antibody. In some embodiments of the invention, the anti-LIGHT antibody is a fully human IgG4 antibody.
[0167] In some embodiments, the anti-LIGHT antibody further comprises a constant region, e.g., a human IgG constant region. In some embodiments, the constant region is a human IgG4 constant region. In additional embodiments, the constant region is a modified human IgG4 constant region. In other embodiments, the constant region is a human C.kappa. constant region.
[0168] In some embodiments of the invention, the anti-LIGHT antibody is a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8 (the "Lead LIGHT Antibody"). In alternative embodiments of the invention, the anti-LIGHT antibody is a fully human IgG4 anti-LIGHT antibody comprising a heavy chain variable region and a light chain variable region, the heavy chain variable region comprising 3 complementary determining regions (CDRs) comprising the amino acid sequences of SEQ ID NOs: 1, 2, and 3, and the light chain variable region comprising 3 CDRs comprising the amino acid sequences of SEQ ID NOs: 4, 5, and 6. Identification, isolation, preparation, and characterization of anti-LIGHT antibodies, including the anti-LIGHT antibody comprising a heavy chain amino acid sequence comprising SEQ ID NO: 7 and a light chain amino acid sequence comprising SEQ ID NO: 8, have been described in detail in U.S. Pat. No. 8,058,402, corresponding to PCT Publication WO 2008/027338, which are incorporated herein by reference.
[0169] Certain embodiments of formulations of the invention also include variants of anti-LIGHT binding agents, such as antibodies. Specifically, the formulations of the invention may include variants of the anti-LIGHT antibody that is a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8. Variants of anti-LIGHT antibodies may have similar physicochemical properties based on their high similarity, and therefore are also included within the scope of the invention. Variants are defined as antibodies with an amino acid sequence that is at least 95%, at least 97%, for instance at least 98% or 99% homologous to an anti-LIGHT antibody, and capable of competing for binding to a LIGHT polypeptide, a LIGHT polypeptide fragment, or a LIGHT epitope. In some embodiments, the variants will ameliorate, neutralize, or otherwise inhibit LIGHT biological activity (e.g., the LIGHT-mediated production or secretion of CCL20, IL-8, or RANTES from cells expressing a LIGHT ligand, such as a LIGHT receptor (e.g., HVEM, LT.beta.R, and/or DcR3). Determining competition for binding to the target can be done by routine methods known to the skilled person in the art. In some embodiments, the variants are human antibodies, and, in some embodiments are IgG4 molecules. In some embodiments, a variant is at least 95%, 96%, 97%, 98%, or 99% identical in amino acid sequence with the Lead Antibody. The term "variant" refers to an antibody that comprises an amino acid sequence that is altered by one or more amino acids compared to the amino acid sequences of the anti-LIGHT antibody. The variant may have conservative sequence modifications, including amino acid substitutions, modifications, additions, and deletions.
[0170] Examples of modifications include, but are not limited to, glycosylation, acetylation, pegylation, phosphorylation, amidation, derivatization by known protecting/blocking groups, proteolytic cleavage, and linkage to a cellular ligand or other protein. Amino acid modifications can be introduced by standard techniques known in the art, such as site-directed mutagenesis, molecular cloning, oligonucleotide-directed mutagenesis, and random PCR-mediated mutagenesis in the nucleic acid encoding the antibodies. Conservative amino acid substitutions include the ones in which the amino acid residue is replaced with an amino acid residue having similar structural or chemical properties. Families of amino acid residues having similar side chains have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., glycine, alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), beta-branched side chains (e.g., threonine, valine, isoleucine), and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan). It will be clear to the skilled artisan that classifications of amino acid residue families other than the one used above can also be employed. Furthermore, a variant may have non-conservative amino acid substitutions, e.g., replacement of an amino acid with an amino acid residue having different structural or chemical properties. Similar minor variations may also include amino acid deletions or insertions, or both. Guidance in determining which amino acid residues may be substituted, modified, inserted, or deleted without abolishing immunological activity may be found using computer programs well known in the art. Computer algorithms, such as, inter alia, Gap or Bestfit, which are known to a person skilled in the art, can be used to optimally align amino acid sequences to be compared and to define similar or identical amino acid residues. Variants may have the same or different, either higher or lower, binding affinities compared to an anti-LIGHT antibody, but are still capable of specifically binding to LIGHT, and may have the same, higher or lower, biological activity as the anti-LIGHT antibody.
[0171] Embodiments of the invention also include antigen binding fragments of the anti-LIGHT binding agents, such as antibodies. The term "antigen binding domain," "antigen binding region," "antigen binding fragment," and similar terms refer to that portion of an antibody which comprises the amino acid residues that interact with an antigen and confer on the binding agent its specificity and affinity for the antigen (e.g., the complementary determining regions (CDR)). The antigen binding region can be derived from any animal species, such as rodents (e.g., rabbit, rat or hamster) and humans. In some embodiments, the antigen binding region will be of human origin. Non-limiting examples of antigen binding fragments include: Fab fragments, F(ab')2 fragments, Fd fragments, Fv fragments, single chain Fv (scFv) molecules, dAb fragments, and minimal recognition units consisting of the amino acid residues that mimic the hypervariable region of the antibody.
[0172] In some embodiments of the invention, the anti-LIGHT binding agents (or a variant thereof or an antigen binding fragment thereof) will ameliorate, neutralize, or otherwise inhibit LIGHT biological activity in vivo (e.g., the LIGHT-mediated production or secretion of CCL20, IL-8, or RANTES from a cell expressing a LIGHT receptor, e.g., HVEM, LT.beta.R, and/or DcR3).
[0173] In some embodiments of the invention, the anti-LIGHT binding agents (or a variant thereof or an antigen binding fragment thereof) are antagonist binding agents that ameliorate, neutralize, or otherwise inhibit LIGHT biological activity in vivo (e.g., the LIGHT-mediated production or secretion of CCL20, IL-8, or RANTES from cells expressing a LIGHT ligand, such as a LIGHT receptor, (e.g., HVEM, LT.beta.R, and/or DcR3).
[0174] In some embodiments, the anti-LIGHT binding agent (or a variant thereof or an antigen binding fragment thereof) is present in the formulations in an amount from about 5 mg/mL to about 280 mg/mL, e.g., about 5 mg/mL to about 200 mg/mL, about 50 mg/mL to about 250 mg/mL, about 100 mg/mL to about 250 mg/mL, about 50 mg/mL to about 200 mg/mL, and about 100 mg/mL to about 200 mg/mL. For example, the anti-LIGHT binding agent may be present in the formulation in an amount of about 5 mg/mL, about 10 mg/mL, about 15 mg/mL, about 20 mg/mL, about 25 mg/mL, about 30 mg/mL, about 35 mg/mL, about 40 mg/mL, about 45 mg/mL, about 50 mg/mL, about 55 mg/mL, about 60 mg/mL, about 65 mg/mL, about 70 mg/mL, about 75 mg/mL, about 80 mg/mL, about 85 mg/mL, about 90 mg/mL, about 95 mg/mL, about 100 mg/mL, about 105 mg/mL, about 110 mg/mL, about 115 mg/mL, about 120 mg/mL, about 125 mg/mL, about 130 mg/mL, about 135 mg/mL, about 140 mg/mL, about 145 mg/mL, about 150 mg/mL, about 155 mg/mL, about 160 mg/mL, about 165 mg/mL, about 170 mg/mL, about 175 mg/mL, about 180 mg/mL, about 185 mg/mL, about 190 mg/mL, about 195 mg/mL, about 200 mg/mL, about 205 mg/mL, about 210 mg/mL, about 215 mg/mL, about 220 mg/mL, about 225 mg/mL, about 230 mg/mL, about 235 mg/mL, about 240 mg/mL, about 245 mg/mL, about 250 mg/mL, about 255 mg/mL, about 260 mg/mL, about 265 mg/mL, about 270 mg/mL, about 275 mg/mL, or about 280 mg/mL.
[0175] In alternative embodiments, the anti-LIGHT binding agent may be present in the formulation in an amount from about 5 to about 25 mg/mL, from about 26 to about 50 mg/mL, from about 51 to about 75 mg/mL, from about 76 to about 100 mg/mL, from about 101 to about 125 mg/mL, from about 126 to about 150 mg/mL, from about 151 to about 175 mg/mL, from about 176 to about 200 mg/mL, from about 201 mg/mL to about 225 mg/mL, from about 226 mg/mL to about 250 mg/mL, from about 251 to about 280 mg/mL, from about 5 to about 10 mg/mL, from about 40 to about 60 mg/mL, from about 75 to about 85 mg/mL, or from about 140 to about 160 mg/mL.
[0176] In certain exemplary embodiments, the anti-LIGHT binding agent is present in the formulation in an amount from about 50 mg/mL to about 170 mg, about 100 mg/mL to about 160 mg/mL, for example about 150 mg/mL. Alternatively, the anti-LIGHT binding agent is present in an amount of about 50 mg/mL. In another exemplary embodiment, a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8 is present in the formulation in an amount of about 150 mg/mL.
ii. Anti-CXCR5 Binding Agents, and Variants and Fragments Thereof.
[0177] In certain embodiments, the formulations of the invention include an anti-CXCR5 binding agent. The binding agents may be any molecule, such as an antibody, a siRNA, a nucleic acid, an aptamer, a protein, or a small molecule organic compound, that binds or specifically binds to CXCR5, or a variant or a fragment thereof. In some embodiments, the binding agent is an anti-CXCR5 antibody, or a variant thereof, or an antigen binding fragment thereof. Anti-CXCR5 antibodies specifically bind to a CXCL13 (also known as BLC) protein, polypeptide fragment, or epitope. The CXCR5 molecule may be from any species. In some embodiments, the CXCR5 molecule is from a human, known herein as "hCXCR5". hCXCR5 has the following amino acid sequence, which is identified as SEQ ID NO: 14:
TABLE-US-00008 (SEQ ID NO: 14) MNYPLTLEMD LENLEDLFWE LDRLDNYNDT SLVENHLCPA TEGPLMASFK AVFVPVAYSL IFLLGVIGNV LVLVILERHR QTRSSTETFL FHLAVADLLL VFILPFAVAE GSVGWVLGTF LCKTVIALHK VNFYCSSLLL ACIAVDRYLA IVHAVHAYRH RRLLSIHITC GTIWLVGFLL ALPEILFAKV SQGHHNNSLP RCTFSQENQA ETHAWFTSRF LYHVAGFLLP MLVMGWCYVG VVHRLRQAQR RPQRQKAVRV AILVTSIFFL CWSPYHIVIF LDTLARLKAV DNTCKLNGSL PVAITMCEFL GLAHCCLNPM LYTFAGVKFR SDLSRLLTKL GCTGPASLCQ LFPSWRRSSL SESENATSLT TF.
[0178] In certain exemplary embodiments, the anti-CXCR5 antibody is a humanized antibody, a fully human antibody, or a variant thereof or an antigen-binding fragment thereof. In some embodiments, anti-CXCR5 antibodies prevent binding of CXCR5 with its ligands and inhibit CXCR5 biological activity (e.g., the binding of CXCR5 to CXCL13).
[0179] In certain specific embodiments, the anti-CXCR5 antibody comprises a heavy chain variable region (VH) comprising the amino acid sequence of any one or more of the following complementary determining regions (CDRs):
TABLE-US-00009 HCDR1- (SEQ ID NO: 15) GFSLIDYGVN; HCDR2- (SEQ ID NO: 16) VIWGDGTTY; or HCDR3- (SEQ ID NO: 17) IVY.
[0180] In other specific embodiments, the anti-CXCR5 antibody comprises a light chain variable region (VL) comprising the amino acid sequence of any one or more of the following complementary determining regions (CDRs):
TABLE-US-00010 LCDR1- (SEQ ID NO: 18) RSSKSLLHSSGKTYLY; LCDR2- (SEQ ID NO: 19) RLSSLA; or LCDR3- (SEQ ID NO: 20) MQHLEYPYT.
[0181] In one specific embodiment, the anti-CXCR5 antibody comprises a heavy chain variable region (VH) comprising the amino acid sequences of SEQ ID NOs: 15, 16, and 17.
[0182] In another specific embodiment, the anti-CXCR5 antibody comprises a light chain variable region (VL) comprising the amino acid sequences of SEQ ID NOs: 18, 19, and 20.
[0183] In more specific embodiments, the anti-CXCR5 antibody comprises a heavy chain variable region comprising the amino acid sequences of SEQ ID NOs: 15, 16, and 17; and a light chain variable region comprising the amino acid sequences of SEQ ID NOs: 18, 19, and 20.
[0184] In a specific embodiment, the anti-CXCR5 antibody comprises a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 21:
TABLE-US-00011 (SEQ ID NO: 21) QVQLKESGPG LVAPSESLSI TCTVSGFSLI DYGVNWIRQP PGKGLEWLGV IWGDGTTYYN PSLKSRLSIS KDNSKSQVFL KVTSLTTDDT AMYYCARIVY WGQGTLVTVS A.
[0185] In another specific embodiment, the anti-CXCR5 antibody comprises a light chain variable region comprising the amino acid sequence of SEQ ID NO: 22:
TABLE-US-00012 (SEQ ID NO: 22) DIVMTQAAPS VAVTPGASVS ISCRSSKSLL HSSGKTYLYW FLQRPGQSPQ LLIYRLSSLA SGVPDRFSGS GSGTAFTLRI SRVEAEDVGV YYCMQHLEYP YTFGGGTKLE IK.
[0186] In more specific embodiments, the anti-CXCR5 antibody comprises a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 21; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 22.
[0187] In some embodiments, the anti-CXCR5 antibody further comprises a constant region, e.g., a human IgG constant region. In some embodiments, the constant region is a human IgG4 constant region. In additional embodiments, the constant region is a modified human IgG4 constant region. In some embodiments, the human IgG4 constant region has the following modifications: S241P (shown below in SEQ ID NO: 23 in bold), L248E (shown below in SEQ ID NO: 23 in bold), and the lack of a terminal lysine in order to avoid heterogeneity. In some embodiments, the IgG4 constant region comprises the amino acid sequence of SEQ ID NO: 23:
TABLE-US-00013 (SEQ ID NO: 23) ASTKGPSVFP LAPCSRSTSE STAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTKT YTCNVDHKPS NTKVDKRVES KYGPPCPPCP APEFEGGPSV FLFPPKPKDT LMISRTPEVT CVVVDVSQED PEVQFNWYVD GVEVHNAKTK PREEQFNSTY RVVSVLTVLH QDWLNGKEYK CKVSNKGLPS SIEKTISKAK GQPREPQVYT LPPSQEEMTK NQVSLTCLVK GFYPSDIAVE WESNGQPENN YKTTPPVLDS DGSFFLYSRL TVDKSRWQEG NVFSCSVMHE ALHNHYTQKS LSLSLG.
[0188] In other embodiments, the constant region is a human C.kappa. constant region. In some embodiments, the C.kappa. constant region comprises the amino acid sequence of SEQ ID NO: 24:
TABLE-US-00014 (SEQ ID NO: 25) QVQLKESGPG LVAPSESLSI TCTVSGFSLI DYGVNWIRQP PGKGLEWLGV IWGDGTTYYN PSLKSRLSIS KDNSKSQVFL KVTSLTTDDT AMYYCARIVY WGQGTLVTVS AASTKGPSVF PLAPCSRSTS ESTAALGCLV KDYFPEPVTV SWNSGALTSG VHTFPAVLQS SGLYSLSSVV TVPSSSLGTK TYTCNVDHKP SNTKVDKRVE SKYGPPCPPC PAPEFEGGPS VFLFPPKPKD TLMISRTPEV TCVVVDVSQE DPEVQFNWYV DGVEVHNAKT KPREEQFNST YRVVSVLTVL HQDWLNGKEY KCKVSNKGLP SSIEKTISKA KGQPREPQVY TLPPSQEEMT KNQVSLTCLV KGFYPSDIAV EWESNGQPEN NYKTTPPVLD SDGSFFLYSR LTVDKSRWQE GNVFSCSVMH EALHNHYTQK SLSLSLG.
[0189] In specific embodiments, the anti-CXCR5 antibody comprises a heavy chain comprising the amino acid sequence of SEQ ID NO: 25:
TABLE-US-00015 (SEQ ID NO: 24) RTVAAPSVFI FPPSDEQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG NSQESVTEQD SKDSTYSLSS TLTLSKADYE KHKVYACEVT HQGLSSPVTK SFNRGEC.
[0190] Positions 1-111: variable region of the heavy chain (VH). The CDRs (complementarity determining regions, according to Kabat definition) are underlined. [0191] Positions 112-432: constant region of human IgG4 (SwissProt IGHG4_HUMAN, including the following modifications: S241P, L248E, and the lack of a terminal lysine in order to avoid heterogeneity).
[0192] In other specific embodiments, the anti-CXCR5 antibody comprises a light chain comprising the amino acid sequence of SEQ ID NO: 26:
TABLE-US-00016 (SEQ ID NO: 26) DIVMTQAAPS VAVTPGASVS ISCRSSKSLL HSSGKTYLYW FLQRPGQSPQ LLIYRLSSLA SGVPDRFSGS GSGTAFTLRI SRVEAEDVGV YYCMQHLEYP YTFGGGTKLE IKRTVAAPSV FIFPPSDEQL KSGTASVVCL LNNFYPREAK VQWKVDNALQ SGNSQESVTE QDSKDSTYSL SSTLTLSKAD YEKHKVYACE VTHQGLSSPV TKSFNRGEC.
[0193] Positions 1-112: variable region of the light chain (VL). The CDRs (complementarity determining regions, according to Kabat definition) are underlined. [0194] Positions 113-182: constant region of human C.kappa..
[0195] In further embodiments, the anti-CXCR5 antibody comprises a heavy chain comprising the amino acid sequence of SEQ ID NO: 25, and a light chain comprising the amino acid sequence of SEQ ID NO: 26.
[0196] In some embodiments, the anti-CXCR5 antibody further comprises a leader sequence. The leader sequence, in some embodiments, comprises an amino acid sequence from 1-30 amino acids in length, such as 25-25 amino acids, and typically 19 amino acids. The heavy chain, light chain, or both the heavy and light chain may comprise a leader sequence. In some embodiments, the leader sequence comprises the amino acid sequence of SEQ ID NO: 16: MGWSCIILFL VATATGVHS (SEQ ID NO: 27).
[0197] In specific embodiments, the anti-CXCR5 antibody comprises a heavy chain derived from the amino acid sequence of SEQ ID NO: 28:
TABLE-US-00017 (SEQ ID NO: 28) MGWSCIILFL VATATGVHSQ VQLKESGPGL VAPSESLSIT CTVSGFSLID YGVNWIRQPP GKGLEWLGVI WGDGTTYYNP SLKSRLSISK DNSKSQVFLK VTSLTTDDTA MYYCARIVYW GQGTLVTVSA ASTKGPSVFP LAPCSRSTSE STAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTKT YTCNVDHKPS NTKVDKRVES KYGPPCPPCP APEFEGGPSV FLFPPKPKDT LMISRTPEVT CVVVDVSQED PEVQFNWYVD GVEVHNAKTK PREEQFNSTY RVVSVLTVLH QDWLNGKEYK CKVSNKGLPS SIEKTISKAK GQPREPQVYT LPPSQEEMTK NQVSLTCLVK GFYPSDIAVE WESNGQPENN YKTTPPVLDS DGSFFLYSRL TVDKSRWQEG NVFSCSVMHE ALHNHYTQKS LSLSLG.
[0198] Positions 1-19: leader sequence [0199] Positions 20-130: variable region of the heavy chain (VH). The CDRs (complementarity determining regions, according to Kabat definition) are underlined. [0200] Positions 131-456: constant region of human IgG4 (SwissProt IGHG4_HUMAN, including the following modifications: S241P, L248E, and the lack of a terminal lysine in order to avoid heterogeneity).
[0201] In other specific embodiments, the anti-CXCR5 antibody comprises a light chain derived from the amino acid sequence of SEQ ID NO: 29:
TABLE-US-00018 (SEQ ID NO: 29) MGWSCIILFL VATATGVHSD IVMTQAAPSV AVTPGASVSI SCRSSKSLLH SSGKTYLYWF LQRPGQSPQL LIYRLSSLAS GVPDRFSGSG SGTAFTLRIS RVEAEDVGVY YCMQHLEYPY TFGGGTKLEI KRTVAAPSVF IFPPSDEQLK SGTASVVCLL NNFYPREAKV QWKVDNALQS GNSQESVTEQ DSKDSTYSLS STLTLSKADY EKHKVYACEV THQGLSSPVT KSFNRGEC.
[0202] Positions 1-19: leader sequence [0203] Positions 20-131: variable region of the light chain (VL). The CDRs (complementarity determining regions, according to Kabat definition) are underlined. [0204] Positions 132-238: constant region of human C.kappa..
[0205] In further embodiments, the anti-CXCR5 antibody comprises a heavy chain comprising the amino acid sequence of SEQ ID NO: 28, and a light chain comprising the amino acid sequence of SEQ ID NO: 29.
[0206] In some embodiments of the invention, the anti-CXCR5 antibody is a humanized or a fully human antibody. Examples of humanized and fully human antibody isotypes include IgA, IgD, IgE, IgG, and IgM. In some embodiments, the anti-CXCR5 antibody is an IgG antibody. There are four forms of IgG. In some embodiments, the anti-CXCR5 antibody is an IgG4 antibody. In some embodiments of the invention, the anti-CXCR5 antibody is a humanized IgG4 antibody.
[0207] In some embodiments of the invention, the anti-CXCR5 antibody is a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26 (the "Lead CXCR5 Antibody"). In alternative embodiments of the invention, the anti-CXCR5 antibody is a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain variable region and a light chain variable region, the heavy chain variable region comprising 3 complementary determining regions (CDRs) comprising the amino acid sequences of SEQ ID NOs: 15, 16, and 17, and the light chain variable region comprising 3 CDRs comprising the amino acid sequences of SEQ ID NOs: 18, 19, and 20. Identification, isolation, preparation, and characterization of anti-CXCR5 antibodies, including the anti-CXCR5 antibody comprising a heavy chain amino acid sequence comprising SEQ ID NO: 25 and a light chain amino acid sequence comprising SEQ ID NO: 26, have been described in detail in PCT Publication WO 2009/032661, which are incorporated herein by reference.
[0208] Certain embodiments of formulations of the invention also include variants of anti-CXCR5 binding agents, such as antibodies. Specifically, the formulations of the invention may include variants of the anti-CXCR5 antibody that is a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26. Variants of anti-CXCR5 antibodies may have similar physicochemical properties based on their high similarity, and therefore are also included within the scope of the invention. Variants are defined as antibodies with an amino acid sequence that is at least 95%, at least 97%, for instance at least 98% or 99% homologous to an anti-CXCR5 antibody, and capable of competing for binding to a CXCR5 polypeptide, a CXCR5 polypeptide fragment, or a CXCR5 epitope. In some embodiments, the variants will ameliorate, neutralize, or otherwise inhibit CXCR5 biological activity (e.g., the binding of CXCL13 to CXCR5). Determining competition for binding to the target can be done by routine methods known to the skilled person in the art. In some embodiments, the variants are human antibodies, and, in some embodiments, are IgG4 molecules. In some embodiments, a variant is at least 95%, 96%, 97%, 98%, or 99% identical in amino acid sequence with the Lead Antibody. The term "variant" refers to an antibody that comprises an amino acid sequence that is altered by one or more amino acids compared to the amino acid sequences of the anti-CXCR5 antibody. The variant may have conservative sequence modifications, including amino acid substitutions, modifications, additions, and deletions.
[0209] Examples of modifications include, but are not limited to, glycosylation, acetylation, pegylation, phosphorylation, amidation, derivatization by known protecting/blocking groups, proteolytic cleavage, and linkage to a cellular ligand or other protein. Amino acid modifications can be introduced by standard techniques known in the art, such as site-directed mutagenesis, molecular cloning, oligonucleotide-directed mutagenesis, and random PCR-mediated mutagenesis in the nucleic acid encoding the antibodies. Conservative amino acid substitutions include the ones in which the amino acid residue is replaced with an amino acid residue having similar structural or chemical properties. Families of amino acid residues having similar side chains have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., glycine, alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), beta-branched side chains (e.g., threonine, valine, isoleucine), and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan). It will be clear to the skilled artisan that classifications of amino acid residue families other than the one used above can also be employed. Furthermore, a variant may have non-conservative amino acid substitutions, e.g., replacement of an amino acid with an amino acid residue having different structural or chemical properties. Similar minor variations may also include amino acid deletions or insertions, or both. Guidance in determining which amino acid residues may be substituted, modified, inserted, or deleted without abolishing immunological activity may be found using computer programs well known in the art. Computer algorithms, such as, inter alia, Gap or Bestfit, which are known to a person skilled in the art, can be used to optimally align amino acid sequences to be compared and to define similar or identical amino acid residues. Variants may have the same or different, either higher or lower, binding affinities compared to an anti-CXCR5 antibody, but are still capable of specifically binding to CXCR5, and may have the same, higher or lower, biological activity as the anti-CXCR5 antibody.
[0210] Embodiments of the invention also include antigen binding fragments of the anti-CXCR5 binding agents, such as antibodies. The term "antigen binding domain," "antigen binding region," "antigen binding fragment," and similar terms refer to that portion of an antibody which comprises the amino acid residues that interact with an antigen and confer on the binding agent its specificity and affinity for the antigen (e.g., the complementary determining regions (CDR)). The antigen binding region can be derived from any animal species, such as rodents (e.g., rabbit, rat or hamster) and humans. In some embodiments, the antigen binding region will be of human origin. Non-limiting examples of antigen binding fragments include: Fab fragments, F(ab')2 fragments, Fd fragments, Fv fragments, single chain Fv (scFv) molecules, dAb fragments, and minimal recognition units consisting of the amino acid residues that mimic the hypervariable region of the antibody.
[0211] In some embodiments of the invention, the anti-CXCR5 binding agents (or a variant thereof or an antigen binding fragment thereof) will ameliorate, neutralize, or otherwise inhibit CXCR5 biological activity in vivo (e.g., the binding of CXCL13 to CXCR5).
[0212] In some embodiments of the invention, the anti-CXCR5 binding agents (or a variant thereof or an antigen binding fragment thereof) are antagonist binding agents that ameliorate, neutralize, or otherwise inhibit CXCR5 biological activity in vivo (e.g., the binding of CXCL13 to CXCR5).
[0213] In some embodiments, the anti-CXCR5 binding agent (or a variant thereof or an antigen binding fragment thereof) is present in the formulations in an amount from about 5 mg/mL to about 280 mg/mL, e.g., about 5 mg/mL to about 200 mg/mL, about 5 mg/mL to about 125 mg/mL, about 5 mg/mL to about 75 mg/mL, about 5 mg/mL to about 50 mg/mL, and about 5 mg/mL to about 25 mg/mL. For example, the anti-CXCR5 binding agent may be present in the formulation in an amount of about 5 mg/mL, about 10 mg/mL, about 15 mg/mL, about 20 mg/mL, about 25 mg/mL, about 30 mg/mL, about 35 mg/mL, about 40 mg/mL, about 45 mg/mL, about 50 mg/mL, about 55 mg/mL, about 60 mg/mL, about 65 mg/mL, about 70 mg/mL, about 75 mg/mL, about 80 mg/mL, about 85 mg/mL, about 90 mg/mL, about 95 mg/mL, about 100 mg/mL, about 105 mg/mL, about 110 mg/mL, about 115 mg/mL, about 120 mg/mL, about 125 mg/mL, about 130 mg/mL, about 135 mg/mL, about 140 mg/mL, about 145 mg/mL, about 150 mg/mL, about 155 mg/mL, about 160 mg/mL, about 165 mg/mL, about 170 mg/mL, about 175 mg/mL, about 180 mg/mL, about 185 mg/mL, about 190 mg/mL, about 195 mg/mL, about 200 mg/mL, about 205 mg/mL, about 210 mg/mL, about 215 mg/mL, about 220 mg/mL, about 225 mg/mL, about 230 mg/mL, about 235 mg/mL, about 240 mg/mL, about 245 mg/mL, about 250 mg/mL, about 255 mg/mL, about 260 mg/mL, about 265 mg/mL, about 270 mg/mL, about 275 mg/mL, or about 280 mg/mL.
[0214] In alternative embodiments, the anti-CXCR5 binding agent may be present in the formulation in an amount from about 5 to about 25 mg/mL, from about 26 to about 50 mg/mL, from about 51 to about 75 mg/mL, from about 76 to about 100 mg/mL, from about 101 to about 125 mg/mL, from about 126 to about 150 mg/mL, from about 151 to about 175 mg/mL, from about 176 to about 200 mg/mL, from about 201 mg/mL to about 225 mg/mL, from about 226 mg/mL to about 250 mg/mL, from about 251 to about 280 mg/mL, from about 5 to about 25 mg/mL, from about 40 to about 60 mg/mL, from about 75 to about 85 mg/mL, or from about 90 to about 110 mg/mL.
[0215] In certain exemplary embodiments, the anti-CXCR5 binding agent is present in the formulation in an amount of about 20 mg/mL. Alternatively, the anti-CXCR5 binding agent is present in an amount of about 100 mg/mL. In another exemplary embodiment, a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26 is present in the formulation in an amount of about 20 mg/mL or 100 mg/mL.
iii. Buffering Agents
[0216] The formulations of the invention comprise a citrate buffer as a buffering agent. A buffering agent maintains a physiologically suitable pH. In addition, a buffering agent enhances isotonicity and chemical stability of the formulation. In some embodiments, the citrate buffer is present in the formulations at a concentration from about 0.5 mM to about 50 mM, e.g., about 5 mM to about 15 mM. For example, the citrate buffer may be present in the formulation at a concentration about 5 mM, about 6 mM, about 7 mM, about 8 mM, about 9 mM, about 10 mM, about 11 mM, about 12 mM, about 13 mM, about 14 mM, about 15 mM, about 16 mM, about 17 mM, about 18 mM, about 19 mM, about 20 mM, about 21 mM, about 22 mM, about 23 mM, about 24 mM, about 25 mM, about 26 mM, about 27 mM, about 28 mM, about 29 mM, about 30 mM, about 31 mM, about 32 mM, about 33 mM, about 34 mM, about 35 mM, about 36 mM, about 37 mM, about 38 mM, about 39 mM, about 40 mM, about 41 mM, about 42 mM, about 43 mM, about 44 mM, about 45 mM, about 46 mM, about 47 mM, about 48 mM, about 49 mM, and about 50 mM. In some embodiments, the citrate buffer is present in the formulation at a concentration from about 7 mM to about 13 mM, such as from about 9 mM to about 11 mM. In some embodiments, the citrate buffer is present at a concentration of about 10 mM.
[0217] In certain embodiments, the formulations of the invention have a pH at or below pH 6. In some embodiments, the pH of the formulations ranges from about 5.0 to about 6.0. For example, the pH of the formulations may be about 5.0, about 5.1, about 5.2, about 5.3, about 5.4, about 5.5, about 5.6, about 5.7, about 5.8, about 5.9, and about 6.0. In some embodiments, the pH of the formulations may range from about 5.5 to about 6.0. In some embodiments, the pH is either about 5.5 or about 6.0. The pH of the formulation may be measured by any means known to those of skill in the art. A means for measuring pH is using a pH meter with a micro-electrode. The pH of the formulation may be adjusted using any means known in the art. Exemplary chemicals for altering the pH of the formulations are hydrochloric acid (HCl) and sodium hydroxide (NaOH).
[0218] In certain embodiments, the formulations of the invention have a pH at or below the isoelectric point (pI) of the binding agent, such as an antibody. The isoelectric point is the pH at which a particular molecule or surface carries no net electrical charge. The pI of an anti-LIGHT or an anti-CXCR5 binding agent may be determined by any means known to those of skill in the art. In some embodiments, the pI of an anti-LIGHT or anti-CXCR5 antibody is determined by denaturated isoelectric focusing. As shown in FIG. 1, the pI of a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8 is 6.8-7.2. As shown in FIG. 11, the pI of a humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26 is 7.6-8.4.
iv. Surfactants
[0219] The formulations of the invention may, optionally, further comprise a surfactant, which is also known as a stabilizing agent. Surfactants/stabilizing agents are chemical compounds that interact and stabilize biological molecules and/or general pharmaceutical excipients in a formulation. In certain embodiments, surfactants may be used in conjunction with lower temperature storage. Surfactants generally protect the binding agent from air/solution interface induced stresses and solution/surface induced stresses, which may otherwise result in protein aggregation. Surfactants may include, but are not limited to, polysorbates, glycerin, dicarboxylic acids, oxalic acid, succinic acid, fumaric acids, phthalic acids, and combinations thereof. Those skilled in the art are aware that other surfactants, e.g. non-ionic or ionic detergents, can be used as long as they are pharmaceutically acceptable, i.e. suitable for administration to subjects. The surfactant is, in some embodiments, a polysorbate. Examples of polysorbates include polysorbate 20, polysorbate 40, polysorbate 60, polysorbate 65, and polysorbate 80.
[0220] In exemplary embodiments, the surfactant is present in the formulations in an amount from about 0.001% to about 0.1% (w/v). For example, the surfactant may be present in the formulations in an amount of about 0.001% (w/v), about 0.002% (w/v), about 0.003% (w/v), about 0.004% (w/v), about 0.005% (w/v), about 0.006% (w/v), about 0.007% (w/v), about 0.008% (w/v), about 0.009% (w/v), about 0.01% (w/v), about 0.02% (w/v), about 0.03% (w/v), about 0.04% (w/v), about 0.05% (w/v), about 0.06% (w/v), about 0.07% (w/v), about 0.08% (w/v), about 0.09% (w/v), and about 0.1% (w/v). In particular embodiments, the surfactant is present in the formulations from about 0.003% to about 0.05% (w/v), about 0.004% to about 0.025% (w/v), or about 0.005% to about 0.02% (w/v), e.g. about 0.005% (w/v). For example, polysorbate 20 may be present in an amount from about 0.001% to about 0.1% (w/v), about 0.002% to about 0.01% (w/v), about 0.003% to about 0.008% (w/v), and about 0.004% to about 0.006% (w/v), e.g., about 0.005% (w/v). In alternative embodiments, polysorbate 20 is present in an amount from about 0.001% to about 0.1% (w/v), about 0.005% to about 0.05% (w/v), and about 0.0075% to about 0.025% (w/v), e.g., about 0.01% (w/v). In further alternative embodiments, polysorbate 20 is present in an amount from about 0.001% to about 0.1% (w/v), about 0.005% to about 0.05% (w/v), and about 0.01% to about 0.03% (w/v), e.g., about 0.02% (w/v).
v. Tonicity Agents
[0221] The formulations of the invention may, optionally, further comprise a tonicity agent. Typically, tonicity agents are used to adjust or maintain the osmolality of the formulations in order to bring it closer to the osmotic pressure of body fluids, such as blood or plasma. Tonicity agents may also maintain the binding agent levels in a formulation. In part, the tonicity agent contributes to preserving the level, ratio, or proportion of the therapeutically active binding agent present in the formulation. As used herein, the term "tonicity" refers to the behavior of biologic components in a fluid environment or solution. Isotonic solutions possess the same osmotic pressure as blood plasma, and can be intravenously infused into a subject without changing the osmotic pressure of the subject's blood plasma. Indeed, in certain embodiments of the invention, the tonicity agent is present in an amount sufficient to render the formulation suitable for intravenous infusion. Often, the tonicity agent serves as a bulking agent or a stabilizing agent as well. As such, the tonicity agent may allow the binding agent to overcome various stresses, such as freezing and shear. Tonicity agents may include, but are not limited to, saccharides, sugars, glycerol, sorbitol, mannitol, sodium chloride, potassium chloride, magnesium chloride, and other inorganic salts. Those skilled in the art are aware that other tonicity agents can be used as long as they are pharmaceutically acceptable, i.e. suitable for administration to subjects.
[0222] In certain embodiments, the tonicity agent is present in the formulations in an amount from about 0.1% to 10% (w/v). For example, the tonicity agent may be present in the formulation in an amount of about 0.1% (w/v), about 0.2% (w/v), about 0.3% (w/v), about 0.4% (w/v), about 0.5% (w/v), about 0.6% (w/v), about 0.7% (w/v), about 0.8% (w/v), about 0.9% (w/v), about 1% (w/v), about 2% (w/v), about 3% (w/v), about 4% (w/v), about 4.5% (w/v), about 5% (w/v), about 5.5% (w/v), about 6% (w/v), about 7% (w/v), about 8% (w/v), about 9% (w/v), and about 10% (w/v). Alternatively, the tonicity agent may be present in the formulation in an amount from about 2% to about 8% (w/v), from about 3% to about 7% (w/v), and from about 4% to about 6% (w/v). In further alternative embodiments, the tonicity agent may be present in the formulation in an amount from about 0.1% to about 1%, from about 0.1% to about 0.5%, from about 0.1 to about 0.3%, and about 0.2%.
[0223] In certain exemplary embodiments, the tonicity agent is a saccharide. Examples of saccharides include glucose, sucrose (which is also known as saccharose), maltose, trehalose, dextrose, xylitol, fructose and mannitol. For example, mannitol may be present in an amount of about 1% to about 10% (w/v), about 2% to about 8% (w/v), or about 3% to about 5% (w/v), e.g., about 4% (w/v). Alternatively, sucrose (which is also known as saccharose) may be present in an amount of about 1% to about 10% (w/v), about 3% to about 8% (w/v), or about 4% to about 6% (w/v), e.g., about 4.5, 5, 5.5, or 6% (w/v).
[0224] In certain other exemplary embodiments, the tonicity agent is sodium chloride. For example, sodium chloride may be present in an amount of about 0.1% (w/v), about 0.2% (w/v), about 0.3% (w/v), about 0.4% (w/v), about 0.5% (w/v), about 0.6% (w/v), about 0.7% (w/v), about 0.8% (w/v), about 0.9% (w/v), and about 1% (w/v). Alternatively, sodium chloride may be present in the formulation in an amount from about 0.1% to about 1%, from about 0.1% to about 0.5%, from about 0.1 to about 0.3%, and about 0.2%.
[0225] In further exemplary embodiments, the formulations may comprise one or more tonicity agents. For example, the formulations may comprise one or more of the above tonicity agents in the above concentrations. In certain specific embodiments, the formulations may comprise sucrose and sodium chloride, wherein each of the sucrose and sodium chloride concentrations is between about 0.1% to about 10% (w/v). In some embodiments, the sucrose concentration is about 6% and the sodium chloride concentration is about 0.2%. Alternatively, the sucrose concentration is about 4.5% and the sodium chloride concentration is about 0.2%.
[0226] In certain embodiments of the invention, the osmolality of the formulations range from about 200 mOsm/kg to about 350 mOsm/kg, about 270 mOsm/kg to about 330 mOsm/kg, about 280 mOsm/kg to about 320 mOsm/kg, or about 290 mOsm/kg to about 310 mOsm/kg, e.g., about 300 mOsm/kg. In other words, the formulations of the invention are, in some embodiments, substantially isotonic, i.e. having substantially the same osmotic pressure as human blood. Osmolality can be measured by any means known to those of skill in the art, such as using vapor pressure or ice-freezing type osmometers. The osmolality of the formulations of the invention can, for instance, be regulated by the one or more tonicity agents described herein.
vi. Amino Acids
[0227] The formulations of the invention may, optionally, further comprise an amino acid. Examples of amino acids include, but are not limited to, glycine, alanine, aspartic acid, lysine, serine, tyrosine, cysteine, glutamine, methionine, arginine, and proline. In exemplary embodiments, the amino acid is present in the formulations in an amount from about 0.1% to 5% (w/v). For example, the amino acid may be present in the formulation in an amount of about 0.1% (w/v), about 0.2% (w/v), about 0.3% (w/v), about 0.4% (w/v), about 0.5% (w/v), about 0.6% (w/v), about 0.7% (w/v), about 0.8% (w/v), about 0.9% (w/v), about 1.0% (w/v), about 1.1% (w/v), about 1.2% (w/v), about 1.3% (w/v), about 1.4% (w/v), about 1.5% (w/v), about 1.6% (w/v), about 1.7% (w/v), about 1.8% (w/v), about 1.9% (w/v), about 2.0% (w/v), about 3% (w/v), about 4% (w/v), and about 5% (w/v). Alternatively, the amino acid is present in the formulation in an amount from about 1.3% to about 1.8% (w/v), or about 1.4% to about 1.6% (w/v), e.g., about 1.5% (w/v). In further alternative embodiments, the amino acid is present in the formulation in an amount from about 0.5% to about 1.5% (w/v), or about 0.8% to about 1.2% (w/v), e.g., about 1.0% (w/v). An exemplary amino acid is proline or arginine. For example, proline may be present in the formulation in an amount from about 1% to about 2%, (w/v) about 1.3% to about 1.8% (w/v), about 1.4% to about 1.6% (w/v), e.g., about 1.5% (w/v). Alternatively, arginine may be present in the formulation in an amount from about 0.5% to about 1.5% (w/v), or about 0.8% to about 1.2% (w/v), e.g., about 1.0% (w/v).
vii. Other Excipients
[0228] Furthermore, the formulations of the invention may comprise other excipients including, but not limited to, water for injection, diluents, solubilizing agents, soothing agents, additional buffers, inorganic or organic salts, antioxidants, or the like. In some embodiments, however, the formulations of the invention comprise no other excipients, except those described above. Other pharmaceutically acceptable carriers, excipients, or stabilizers, such as those described in Remington's Pharmaceutical Sciences 16.sup.th edition, Osol, A. Ed. (1980) may be included in the formulation provided that they do not adversely affect the desired characteristics of the formulation. In a particular embodiment, the formulation is substantially free of preservatives, although, in alternative embodiments, preservatives may be added as necessary. For example, cryoprotectants or lyoprotectants may be included in lyophilized formulations.
viii. Liquid or Lyophilized Formulations
[0229] The formulations of the invention may either be liquid formulations or lyophilized formulations. In some embodiments, the formulations are liquid formulations. In some embodiments, the liquid formulations are ready for injection. Alternatively, the formulations may be lyophilized powders. In some embodiments, the lyophilized powders are ready to be combined with a solvent just prior to administration.
ix. Exemplary Formulations
[0230] In one exemplary embodiment of the invention, the invention provides a stable liquid antibody formulation suitable for subcutaneous administration, the formulation comprising:
[0231] a) greater than about 80 mg/ml, e.g., about 150 mg/ml, of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0232] b) about 10 mM citrate buffer;
[0233] c) about 0.005% (w/v) polysorbate 20; and
[0234] d) about 4% (w/v) mannitol;
wherein the pH of the formulation is about pH 5.5
[0235] In certain exemplary embodiments, this formulation may be manufactured by:
[0236] a) dissolving about 10 mM sodium citrate dihydrate in water for injection and adjusting the pH of the buffered solution to about pH 5.5, e.g., using either hydrochloric acid or sodium hydroxide;
[0237] b) adding greater than about 80 mg/ml, e.g., about 150 mg/ml, of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8, about 4% (w/v) mannitol, and 0.005% (w/v) polysorbate 20 to the buffered solution from step a) while stirring in a vessel made of inert material until completely dissolved, and then adjusting the pH of the resulting formulation to about pH 5.5 using either hydrochloric acid or sodium hydroxide, and then adding buffered solution from step a) to adjust the final weight of the resulting formulation;
[0238] c) filtering the formulation from step b) under aseptic conditions using a sterilized, compatible membrane filter having a nominal pore size of 0.2 .mu.M, and then sterilizing the formulation by filtration under aseptic conditions into sterilized containers made out of inert material using a sterilized, compatible membrane filter having a nominal pore size of 0.2 .mu.M;
[0239] d) filling the formulation from step c) under aseptic conditions into sterilized vials that are closed with stoppers and flip-off caps with a flange; and, optionally,
[0240] e) inspecting the containers from step d) for coarse contaminants, intact sealing, and visible particles.
[0241] In another exemplary embodiment of the invention, the invention provides a stable liquid antibody formulation suitable for intravenous administration, the formulation comprising:
[0242] a) about 5 to about 80 mg/mL, e.g., about 50 mg/mL, of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0243] b) about 10 mM citrate buffer; and
[0244] c) about 0.01% (w/v) polysorbate 20;
wherein the pH of the formulation is about pH 5.5.
[0245] In an alternative exemplary embodiment of the invention, the invention provides a stable lyophilized antibody formulation suitable for intravenous administration, the formulation comprising:
[0246] a) about 5 to about 80 mg/mL, e.g., about 50 mg/mL, of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8;
[0247] b) about 10 mM citrate buffer;
[0248] c) about 0.01% (w/v) polysorbate 20;
[0249] d) about 5% (w/v) sucrose; and
[0250] e) about 1.5% (w/v) proline;
wherein the pH of the formulation is about pH 5.5.
[0251] In an exemplary embodiment of the invention, the invention provides a stable antibody formulation comprising:
[0252] a) about 20 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26;
[0253] b) about 10 mM citrate buffer;
[0254] c) about 0.02% polysorbate 20;
[0255] d) about 6% sucrose; and
[0256] e) about 0.2% sodium chloride;
wherein the pH of the formulation is about pH 6.0.
[0257] In an alternative exemplary embodiment of the invention, the invention provides a stable antibody formulation comprising:
[0258] a) about 100 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26;
[0259] b) about 10 mM citrate buffer;
[0260] c) about 0.01% polysorbate 20;
[0261] d) about 4.5% sucrose;
[0262] e) about 0.2% sodium chloride; and
[0263] f) about 1% arginine;
wherein the pH of the formulation is about pH 6.0. x. Stability
[0264] The formulations of the invention are stable at 50C for at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12 months or more, and typically at least about 12, 18 or 24 months or more. In exemplary embodiments, they are stable at 5.degree. C. for at least about 6 months or more. In other exemplary embodiments, they are stable at 5.degree. C. for at least about 9 months. In further exemplary embodiments, they are stable at 5.degree. C. for at least about 1 year or more, and typically about 2 years.
C. Modes of Administration
[0265] In certain embodiments of the invention, the formulations are suitable for administration parenterally, intravenously, intramuscularly, intradermally, subcutaneously, or a combination thereof. The formulations of the invention are suitable for delivery by a variety of techniques.
[0266] In some embodiments of the invention, the formulation is administered intravenously. For example, it is desirable that formulations containing 80 mg/mL of IgG4 binding agent, such as an antibody, or less are administered intravenously. Therefore, the formulations are typically sterile. Methods for making formulations sterile are well known in the art and include, for example, filtration through sterile filtration membranes or autoclaving the ingredients of the formulation, with the exception of the antibodies, at about 120.degree. C. for about 30 minutes. For example, the invention provides a stable liquid antibody formulation suitable for intravenous administration, the formulation comprising: a) about 5 to about 80 mg/mL, e.g., about 50 mg/mL, of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8; b) about 10 mM citrate buffer; and c) about 0.01% (w/v) polysorbate 20; wherein the pH of the formulation is about pH 5.5. Alternatively, the invention provides a stable antibody formulation comprising: a) about 20 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26; b) about 10 mM citrate buffer; c) about 0.02% polysorbate 20; d) about 6% sucrose; and e) about 0.2% sodium chloride; wherein the pH of the formulation is about pH 6.0.
[0267] In some embodiments of the invention, the formulation is administered subcutaneously. For example, it is desirable that formulations containing more than 80 mg/mL of IgG4 binding agent, such as an antibody, are administered subcutaneously. In a specific embodiment, it is desirable to administer subcutaneously to subjects a stable liquid antibody formulation comprising: a) about 150 mg/mL of a fully human IgG4 anti-LIGHT (lymphotoxin-like, exhibits inducible expression and competes with HSV glycoprotein D for HVEM, a receptor expressed by T lymphocytes) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8; b) about 10 mM citrate buffer; c) about 0.005% (w/v) polysorbate 20; d) about 4% (w/v) mannitol; and wherein the pH of the formulation is about pH 5.5. Alternatively, the invention provides a stable antibody formulation comprising: a) about 100 mg/mL of a humanized IgG4 anti-CXCR5 (C-X-C chemokine receptor type 5) antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26; b) about 10 mM citrate buffer; c) about 0.01% polysorbate 20; d) about 4.5% sucrose; e) about 0.2% sodium chloride; and f) about 1% arginine; wherein the pH of the formulation is about pH 6.0.
D. Dosages and Dosage Forms
[0268] Effective doses of the formulations of the invention vary depending upon many different factors, including means of administration, target site, physiological state of the subject, whether the subject is human or an animal, other medications administered, and whether treatment is prophylactic or therapeutic. Usually, the subject is a human, but non-human mammals including transgenic mammals can also be treated. Treatment dosages may need to be titrated to optimize safety and efficacy.
[0269] The formulations of the invention may be administered on multiple occasions. Intervals between single dosages can be daily, weekly, biweekly, monthly or yearly. Intervals can also be irregular. In some methods, the dosage is adjusted to achieve a certain plasma binding agent, such as an antibody, concentration. Dosage and frequency will vary depending on the half-life of the anti-LIGHT or anti-CXCR5 binding agent, such as an antibody, in the subject. In general, human antibodies show the longest half-life, followed by humanized antibodies, chimeric antibodies, and nonhuman antibodies.
[0270] In further embodiments, the invention provides a pharmaceutical unit dosage form comprising a therapeutically effective amount of a formulation of the invention for the treatment of one or more diseases in a subject through administration of the dosage form to the subject. In some embodiments, the subject is a human. The human may be an adult or may be an infant. The term "pharmaceutical unit dosage form" refers to a physically discrete unit suitable as unitary dosages for the subjects to be treated, each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic/prophylactic effect in association with the required citrate buffer and pH.
[0271] The unit dosage form may be a container comprising the formulation. Suitable containers include, but are not limited to, sealed ampoules, vials, bottles, syringes, and test tubes. The containers may be formed from a variety of materials, such as glass or plastic, and may have a sterile access port (for example, the container may be a vial having a stopper pierceable by a hypodermic injection needle). In some embodiments, the container is a vial. Generally, the container should maintain the sterility and stability of the formulation.
[0272] In specific embodiments, the formulations are packaged in 2 mL vials that are made of clear, colorless type I glass, and closed with a stopper (fluoropolymer-coated bromobutyl) sealed with flip-of caps with flange (polypropylene). The vials are, in some embodiments, filled with 1.2 mL of the formulations so that the vial has an overfill volume of about 0.2 mL per vial, and an extractable volume of 1.0 mL. For example, this means that the dosage strength of anti-LIGHT antibody (e.g., 150 mg/mL) will be contained within 1 mL of solution.
[0273] In specific embodiment, the formulations are secondarily packaged in a container, such as a cardboard box, that protects the vials from light.
E. Methods of Treatment
[0274] Further provided herein are methods for treating a LIGHT-mediated disease or disorder, the methods comprising administering a formulation of the invention to a subject. The invention further relates to a formulation of the invention for use in a herein-described method for treating a LIGHT-mediated disease or disorder. In certain embodiments, the LIGHT-mediated disease is a chronic bowel disease, or an inflammatory bowel disease (IBD), such as Crohn's disease (CD) or ulcerative colitis (UC). In other embodiments, the LIGHT mediated disease is graft-versus-host disease (GVHD).
[0275] Also provided herein are methods for treating a CXCR5-mediated disease or disorder, the methods comprising administering a formulation of the invention to a subject. The invention further relates to a formulation of the invention for use in a herein-described method for treating a CXCR-5 mediated disease or disorder. In certain embodiments, the anti-CXCR5 binding agent is used for reduction of signs and symptoms, inhibition of progression of structural damage, induction of a major clinical response, and prevention of disability in adult patients with moderately to severely active Rheumatoid Arthritis (RA) who have had inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs), such as methotrexate (MTX), or TNF.alpha. antagonists. The anti-CXCR5 binding agent may be used in combination with DMARDs or anti-TNF.alpha. agonists.
[0276] In certain embodiments, the formulations of the invention can be administered in combination with one or more therapies (e.g., therapies that are not the formulations of the invention that are currently administered to prevent, treat, manage, and/or ameliorate a LIGHT-mediated disease or a CXCR5-mediated disease. The use of the term "in combination" does not restrict the order in which therapies are administered to a subject. A first therapy can be administered before (e.g., 1 minute, 45 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks), concurrently, or after (e.g., 1 minute, 45 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks) the administration of a second therapy to a subject that had, has, or is susceptible to a LIGHT-mediated disease or a CXCR5-mediated disease. Any additional therapy can be administered in any order with the other additional therapies. Non-limiting examples of therapies that can be administered in combination with an antibody of the invention include approved anti-inflammatory agents listed in the U.S. Pharmacopoeia and/or Physician's Desk Reference.
F. Kits
[0277] Certain embodiments of the invention include a kit comprising a formulation of the invention. The kit may further comprise one or more containers comprising pharmaceutically acceptable excipients, and include other materials desirable from a commercial and user standpoint, including filters, needles and syringes. Associated with the kits can be instructions customarily included in commercial packages of therapeutic, prophylactic or diagnostic products, that contain information about, for example, the indications, usage, dosage, manufacture, administration, contra-indications, and/or warnings concerning the use of such therapeutic, prophylactic or diagnostic products. The kit can also be associated with a label that can be any kind of data carrier (e.g., a leaflet, sticker, chip, print or bar code) comprising information. In certain embodiments, the instructions etc. as listed above can be comprised in or on the label. The kit can further comprise a device for administration of the formulation, and particularly a device that contains the formulation, i.e., a pre-filled device such as, but not limited to, a pre-filled syringe or a pre-filled autoinjector. The kit can also comprise a container comprising the formulation, i.e., a pre-filled container, such as a pre-filled vial, cartouche, sachet, or ampoule.
G. Combination of Different Embodiments
[0278] In the context of the present invention, any of the herein described embodiments can be combined with one or more of the other herein described embodiments unless explicitly stated to the contrary. Particularly, any of the herein described binding agents and antibodies and the herein described formulations thereof can be used in combination with any of the kits, pre-filled devices or pre-filled containers, or can be used in the methods of treatment or medical uses as described herein in connection with the respective antibody (e.g., the stable formulations comprising the anti-LIGHT antibodies or anti-CXCR5 antibodies can be combined with any of the herein described kits, containers or devices). Any of the herein described binding agents specifically binding an antigen (e.g., a binding agent specifically binding LIGHT or a binding agent specifically binding CXCR5) can also be used in any of the methods of treatment that are described herein in connection with the respective antibodies (i.e., anti-LIGHT or anti-CXCR5) and vice versa.
EXAMPLES
[0279] To help illustrate the invention, the following examples are provided. The examples are not intended to limit the scope of the invention in any way. In general, the practice of the present invention employs, unless otherwise indicated, conventional techniques of pharmaceutical formulation, chemistry, molecular biology, recombinant DNA technology, immunology such as antibody technology, and standard techniques of polypeptide preparation as described, for example, in Sambrook, Fritsch and Maniatis, Molecular Cloning: Cold Spring Harbor Laboratory Press (1989); Antibody Engineering Protocols (Methods in Molecular Biology), volume 51, Ed.: Paul S., Humana Press (1996); Antibody Engineering: A Practical Approach (Practical Approach Series, 169), Eds.: McCafferty J. et al., Humana Press (1996); Antibodies: A Laboratory Manual, Harlow and Lane, Cold Spring Harbor Laboratory Press (1999); and Current Protocols in Molecular Biology, Eds. Ausubel et al., John Wiley & Sons (1992).
Anti-LIGHT
[0280] A fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8 (the "Lead LIGHT Antibody") was used in Examples 1-9 in order to determine optimal formulation conditions.
Materials
Drug Substance Batch
[0281] The Lead Antibody, formulated in phosphate buffered saline (PBS) at a concentration of 5.5 mg/mL and at a pH of 7.3 (the "Original Formulation", "PBS Formulation", or "Reference Lot"), was used in the following examples.
Excipients
[0282] Table 1 shows the excipients that were used in the following examples, which were chosen according to their acceptability/suitability for use in parenteral products.
TABLE-US-00019 TABLE 1 Excipients used in this study Excipients Art. No./Charge Supplier Arginine 1.01587 Merck Citric acid 100241 Merck HCl 114027 Merck Sodium acetate 1.06265 Merck Sodium chloride 10158 Riedel de Haen Sodium hydroxide 114076 Merck Sodium citrate 114196 Boehringer Ingelheim KG Polysorbate 20 139850 Fluka Trehalose-dihydrate T9531 Sigma-Aldrich
Methods
[0283] The following methods were used to manufacture the experimental formulations and the formulations of the invention containing the Lead LIGHT Antibody.
Manufacturing & Composition of Buffers
[0284] All buffers were manufactured under stirring to dissolve the respective excipients. pH was adjusted using 0.1 M HCl or 0.1 M NaOH. The general concentration of all buffers was 10 mM.
Manufacturing & Composition of Excipient Stock Solutions
[0285] All stock solutions were manufactured under stirring to dissolve the excipients. Concentration was given as weight/weight (w/w).
Sterile Filtration of Samples
[0286] All samples, solutions, buffers, etc. were sterile filtered (0.22 .mu.m) using a Sartopore-2 membrane. The samples were filtered into sterilized bottles or vials and closed under aseptic conditions inside a clean-bench to prevent microbiological contamination.
Mechanical Stress Test
[0287] Mechanical stress with an agitation speed of 350/minute for 2.5 hours at room temperature was performed using a horizontal laboratory shaker with a 26 mm distance (shaker & incubation hood from Buhler Company). 2R vials were filled with 1 mL solution with a head space of about 2.5 mL. The mechanical stress test was planned and performed during the first pre-formulation studies and during relevant studies for surfactant selection.
Thermal Stress Test
[0288] Thermal stress was used as a stress test during all steps of the pre-formulation program. The samples were stored at +40.degree. C. for either 24 hours or 7 days, depending on the study.
Analytical Methods in Formulation Fill and Finish
[0289] The following analytical methods were used in the formulation fill and finish in the following examples.
Appearance
[0290] Appearance of the antibody solutions were checked visually, and additionally documented by taking a picture with a digital camera.
pH
[0291] All pH measurements were performed using a pH-meter with a micro-electrode.
Concentration Using UV
[0292] The protein concentrations of all antibody solutions were measured against buffer using a NanoDrop ND1000. Proteins concentrations near or below 5 mg/mL were diluted 1:3, while higher protein concentrations near 20 mg/mL were diluted 1:20, and the absorption was measured at 215 nm and 280 nm.
Dynamic Light Scattering (DLS)
[0293] The hydrodynamic diameter of the molecule was measured using laser light scattering. The samples were sterile filtered prior to the analytics if turbidity was observed, thus only soluble aggregates could be detected.
Differential Scanning Calorimetry (DSC)
[0294] Aliquots of most pre-formulation samples were examined by DSC using a VPCapillary DSC from Microcal and scanned in the autosampling instrument at 90.degree. C./hour with a filter time of 2 seconds. 400 .mu.l samples were placed into 96-well plates and analyzed for the unfolding temperature Tm.
Osmolarity
[0295] Osmolarity was measured using an automated Knaur Osmometer.
Density
[0296] Density of the formulations was measured using a falling sphere viscosimeter DMA4500 Anton Paar.
Analytical Methods in Bioanalytics FF
[0297] The following analytical methods were used in the bioanalytics fill and finish in the following examples.
Size Exclusion Chromatography (SEC)
[0298] Aggregates, as well as degradation products of the Lead Antibody, were quantified using size exclusion GL chromatography. The test was carried out by isocratic HPLC with a SUPERDEX 200 10/300 column.
SDS-PAGE, Reducing and Non-Reducing
[0299] Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to analyze the molecular integrity (e.g., half molecules) and the purity. This electrophoretic analysis was performed with 4-12% gradient gels under reducing and non-reducing conditions. The proteins were visualized with Coomassie staining after electrophoretic separation.
Weak Cation Exchange (WCX)
[0300] Weak cation exchange chromatography was used to monitor the charge heterogeneity of the antibody. The percentages of basic, neutral, and acidic isoforms were reported. The test was carried out by discontinuous high performance liquid chromatography (HPLC) with a ProPac WCX10 column.
Antigen-Enzyme Linked Immunosorbent Assay (Antigen-ELISA)
[0301] Antigen-ELISA was performed to determine the functionality of the antibody. The binding properties to native LIGHT protein were monitored in comparison to the current standard of the antibody. This potency was reported as the relative EC.sub.50.
Isoelectric Focusing (IEF)
[0302] IEF was performed. The isoelectric pattern was specific for the Lead Antibody and served as an identification test. Degradation could be seen by a different charge pattern.
Storage
[0303] All buffer solutions, excipient solutions, and samples were stored at 5.degree. C. (+3.degree. C.), if not otherwise mentioned.
Summary of all Formulations Prepared & Analyzed
[0304] Table 2 below shows a summary of all of the formulations that were prepared and analyzed in the following examples. Each of the formulations contained the Lead LIGHT Antibody at the concentration listed.
TABLE-US-00020 TABLE 2 Summary of all formulations prepared and analyzed Sample number Buffer pH Concentration Comment Formulation 1.1 Citrate 10 mM 5.0 5.5 mg/mL Formulation 1.2 Citrate 10 mM 5.5 5.5 mg/mL Formulation 1.3 Citrate 10 mM 6.0 5.5 mg/mL Formulation 2 PBS 7.3 <80 5 mg/mLmg/mL Very turbid Citrate 10 mM 5.0 Formulation 3.1 PS20 0.01% 5 mg/mL Citrate 10 mM 5.5 Formulation 3.2 PS20 0.01% 5 mg/mL Citrate 10 mM 5.5 Formulation 4 PS20 0.01% 80 mg/mL clear Citrate 10 mM 5.0 Formulation 5 PS20 0.01% 5 mg/mL Citrate 10 mM 5.5 PS20 0.01% Proline 1.5% Formulation 6.1 Sucrose 5% 50 mg/mL Lyo Citrate 10 mM 5.5 PS20 0.01% Formulation 6.2 Sucrose 5% 50 mg/mL Lyo Formulation 7 Histidine 10 mM 5.5 50 mg/mL Histidine 10 mM 5.5 Formulation 8 PS20 0.01% 50 mg/mL Formulation 9 Citrate 10 mM 5.5 50 mg/mL Citrate 10 mM 5.5 Formulation 10 PS20 0.01% 50 mg/mL Citrate 10 mM 5.5 Formulation 11 Sucrose 5% 50 mg/mL lyo Formulation 12 Citrate 10 mM 7.0 5 mg/mL .mu.DSC Formulation 13 PBS 5.0 5 mg/mL .mu.DSC
Example 1--Characterization of a Phosphate Buffered Saline (PBS) Formulation and Disadvantages Associated Therewith
[0305] In this example, the Reference Lot was characterized. As stated in the Materials section above, the Reference Lot contains the Lead LIGHT Antibody formulated in phosphate buffered saline (PBS) at a concentration of 5.5 mg/mL and at a pH of 7.3, and produced in research solutions Vitry (BioSCP).
[0306] Isoelectric focusing (IEF) was used to determine the isoelectric point (pI) of the Lead Antibody. The pI of the Lead LIGHT Antibody was theoretically calculated as 6.28, and then measured by denaturated isoelectric focusing using standard methods known in the art. As shown in FIG. 1, the main bands show that the pI of the Lead LIGHT Antibody was 6.8-7.2.
[0307] SDS-PAGE was used to identify the molecular weight of the antibody monomer, potential aggregates, or the presence of half-molecules. FIG. 2 shows an SDS-PAGE gel that compared different Reference Lot batches under reducing and non-reducing conditions. An ELISA was used to determine the antigen binding activity of the Lead LIGHT Antibody. FIG. 3 shows an ELISA graph that was used to determine the antigen binding activity of the first and second batches of Reference Lot.
[0308] SEC was used to determine the presence of aggregates, as well as degradation products of the first batch of Reference Lot. As shown in FIG. 4, size exclusion chromatography detected high molecular weight proteins (HMWP), e.g., di-/oligomers (RRT0.8) or aggregates, and low molecular weight proteins (LMWPs) or degradation products. The first batch of Reference Lot had a purity of 97% monomer content.
[0309] WCX was used to monitor the charge heterogeneity of the first batch of Reference Lot. As shown in FIG. 5, rearrangements of acidic, neutral, and basic isoforms occurred during stability studies. The first batch of Reference Lot had a distribution of acidic/neutral/basic isoforms of 42.3/55.6/1.9%.
[0310] DSC was used to analyze the unfolding temperature Tm of the first batch of Reference Lot. As shown in FIG. 6, the three domains of the antibody unfold at 68.degree. C., 75.degree. C., and 78.degree. C.
[0311] DLS was used to determine the hydrodynamic diameter of the antibody monomer and potential soluble aggregates. As shown in FIGS. 7 & 8, a hydrodynamic diameter of about 10 nm was detected, but aggregates were seen in PBS. However, aggregates were not seen in citrate buffer (FIG. 10).
Example 2--Development of Citrate-Buffered Formulations, and Advantages Associated Therewith
[0312] The original buffer, phosphate buffered saline (PBS) at a pH of 7.3, was, in terms of pH, very close to the isoelectric point (pI) of the Lead Antibody (see Example 1). In addition, the Original Formulation exhibited aggregates; half-molecules; degradation products; low molecular weight proteins (LMWPs); high molecular weight proteins (HMWPs); and rearrangements of acidic, basic, and neutral antibody isoforms (see Example 1). Thus, there was a need for an improved formulation that does not suffer from these disadvantages.
[0313] Formulations of the Lead LIGHT Antibody (a fully human IgG4 anti-LIGHT antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 7 and a light chain comprising the amino acid sequence of SEQ ID NO: 8) containing 10 mM citrate buffer at a pH of 5, 5.5, and 6, with and without polysorbate 20 were tested. Table 3 shows the analytical results of the first batch of Reference Lot, and the various experimental formulations of the Lead LIGHT Antibody formulated into citrate, at a pH of 5.0 and 5.5 and 6.0, with and without polysorbate 20. Aggregates were found in dynamic light scattering (DLS) measurements for the Reference Lot, but not in all other tested formulations. Tm, as measured by differential scanning calorimetry (.mu.DSC), indicated that the higher the pH, the higher the thermodynamic stability could be assumed. But for high antibody concentrated formulations, the pH had to be chosen below the pI of the antibody.
[0314] As shown in Table 4, size exclusion chromatography (SEC) data showed a significantly reduced amount of high molecular weight proteins (HMWPs) for the Lead LIGHT Antibody in citrate buffer as compared to the Reference Lot (phosphate buffer at pH 7.3). In contrast, no differences could be detected with SDS-PAGE (Table 5).
TABLE-US-00021 TABLE 3 Analytical Results of Formulations Concen- Sample Tm1 Tm2 Tm3 ZAve tration number [.degree. C.] [.degree. C.] [.degree. C.] pH [nm] [mg/mL] Buffer Reference 67.94 75.00 77.37 7.3 179.85 5.5 PBS Lot Formulation 58.39 69.98 75.75 5.0 10.97 5.0 Citrate 1.1 10 mM Formulation 62.02 72.26 76.59 5.5 10.71 5.0 Citrate 1.2 10 mM Formulation 65.46 73.74 77.02 6.0 10.81 5.0 Citrate 1.3 10 mM Formulation 58.33 69.93 75.74 5.0 13.14 5.0 Citrate 3.1 10 mM PS20 0.01% Formulation 61.42 71.97 76.45 5.5 12.79 5.0 Citrate 3.2 10 mM PS20 0.01%
TABLE-US-00022 TABLE 4 SEC data of Formulations ANTIBODY RRT0.8 LMWP HMWPs Monomer Sample Area Rel. Area Area Rel. Area Area Rel. Area Area Rel. Area Content Name mAU*min % mAU*min % mAU*min % mAU*min % [mg/mL] Ref. Lot 255.61 98.00 3.98 1.52 1.50 0.57 0.59 0.23 Formulation 223.23 98.07 3.22 1.42 1.01 0.44 0.16 0.07 45.49 3.1 Formulation 257.09 98.24 3.74 1.43 0.79 0.30 0.09 0.03 48.92 3.2
TABLE-US-00023 TABLE 5 SDS-PAGE data of Formulations Sample size Rel. QTY size Rel. QTY size Rel. QTY size Rel. QTY Name kDa % kDa % kDa % kDa % comment Ref. Lot 172.5 98.4 150.1 1.4 68.4 0.2 Additional bands <0.5% Formulation 166.1 97.7 147.8 2 71.5 0.3 Identical pattern to Ref. Lot 3.1 Formulation 166.2 96.2 147.2 3.4 71.4 0.4 Identical pattern to Ref. Lot 3.2
Example 3--Development of High-Concentration Antibody Formulations
[0315] In view of the improvements provided by the Citrate-Buffered Antibody Formulation of Example 2, the citrate buffer components were optimized for increased concentrations of Lead LIGHT Antibody. Table 6 shows the analytical results of the first batch of high concentration (about 40 mg/ml) antibody formulations: high phosphate buffered saline (PBS) at a pH of 7.3 (Formulation 2) or citrate at a pH of 5.5 with polysorbate 20 (Formulation 4).
TABLE-US-00024 TABLE 6 Analytical results of Formulations 2 & 4 Concen- Sample Tm1 Tm2 Tm3 ZAve tration number [.degree. C.] [.degree. C.] [.degree. C.] pH [nm] [mg/mL] Buffer Reference 67.94 75.00 77.37 7.3 10.05 5.5 PBS Lot Formulation 67.87 74.87 77.28 7.3 12.89 42.1 PBS 2 Formulation 61.55 72.00 76.48 5.5 16.71 39.97 Citrate 4 10 mM PS20 0.01%
[0316] Slightly reduced monomer content was observed after concentrating the protein solution in citrate buffer. Moreover, dimer concentration was reduced and high molecular weight proteins (HMWPs) could be significantly reduced as well (see Table 7). In contrast, these impurities and byproducts were increased by increasing the concentration in phosphate buffer. No differences could be detected with SDS-PAGE analysis (Table 8).
TABLE-US-00025 TABLE 7 SEC data of Formulations 2 & 4 SEC Analysis ANTIBODY RRT0.8 LMWP HMWPs Monomer Area Rel. Area Area Rel. Area Area Rel. Area Area Rel. Area Gehalt Sample Name mAU*min % mAU*min % mAU*min % mAU*min % [mg/mL] Ref. Lot 255.61 98.00 3.98 1.52 1.50 0.57 0.59 0.23 Formulation 2 121.42 97.39 2.13 1.71 0.98 0.79 0.15 0.12 44.08 Formulation 4 141.90 97.65 2.17 1.49 1.16 0.80 0.09 0.06 45.83
TABLE-US-00026 TABLE 8 SDS-PAGE data of Formulations 2 & 4 SDS-PAGE Analysis Antibody Main 2. band Half molecules Additional bands size Rel. QTY size Rel. QTY size Rel. QTY size Rel. QTY Sample Name kDa % kDa % kDa % kDa % comment Ref. Lot 172.5 98.4 150.1 1.4 68.4 0.2 Additional bands <0.5% Formulation 2 170.6 97.9 147.6 1.9 72.2 0.2 Identical pattern to Ref. Lot Formulation 4 171 97.2 149 2.5 70.5 0.3 Identical pattern to Ref. Lot
Example 4--Development of Lyophilized Antibody Formulations
[0317] To test the feasibility of lyophilization, different lyophilized experimental formulations were manufactured and subjected to stability analysis. The concentration of the Lead LIGHT Antibody was increased to 50 mg/mL.
[0318] Table 9 shows the freeze drying program that was used in this example.
TABLE-US-00027 TABLE 9 Freeze drying program Lyo program (vacuum) No 8 Chamber loading 5 min/RT/100% Freezing 2 h/-45.degree. C./100% Main drying I 30 min/-45.degree. C./30% Main drying II 5 h/-20.degree. C./30% Main drying III 8 h/+20.degree. C./30% Final drying 2 h/+20.degree. C./3%
[0319] Table 10 shows the analytical results of the first batch of Reference Lot, and the various experimental lyophilized formulations of the Lead LIGHT Antibody formulated into various combinations of citrate buffer, sucrose, polysorbate 20, and proline.
[0320] As shown in Table 11, high molecular weight proteins (HMWPs) could clearly be reduced by using citrate buffer. No differences in dimer content were seen over the time of storage at 40.degree. C. An increase of low molecular weight proteins (LMWPs) after freeze drying was observed. As before, these differences could not be detected with SDS-PAGE analysis (Table 12).
TABLE-US-00028 TABLE 10 Analytical data of Formulations 6-6.2 & 11 Sample ZAve number Tm1 Tm2 Tm3 Time/Temp. pH [nm] Concentration Buffer Reference 67.94 75.00 77.37 7.3 10.05 5.5 mg/mL PBS Lot Formulation Nd Nd Nd N/A 5.7 17.46 57.32 Citrate 6 10 mM PS20 0.01% Formulation 64.30 72.61 77.02 T0 5.7 59.66 Nd Citrate 6.1 T1/5.degree. C. 5.7 18.85 Nd 10 mM T1/40.degree. C. 5.7 19.12 Nd PS20 T2/5.degree. C. 5.7 Nd Nd 0.01% T2/40.degree. C. 5.7 Nd Nd Prolin 1.5% Sucrose 5% Formulation 65.45 75.08 79.37 T0 5.7 19.58 Nd Citrate 6.2 T1/5.degree. C. 5.7 31.34 Nd 10 mM T1/40.degree. C. 5.7 18.1 Nd PS20 T2/5.degree. C. 5.7 Nd Nd 0.01% T2/40.degree. C. 5.7 Nd Nd Sucrose 5% Formulation 68.84 75.61 77.91 T0 7.0 98.60 56.49 PBS 11 T1/5.degree. C. 5.7 20.22 Nd Sucrose T1/40.degree. C. 5.7 22.68 Nd 5% T2/5.degree. C. 5.7 Nd Nd T2/40.degree. C. 5.7 Nd Nd
TABLE-US-00029 TABLE 11 SEC data of Formulations 6.1-6.2 & 11 SEC Analysis ANTIBODY RRT0.8 LMWP HMWPs Area Rel. Area Area Rel. Area Area Rel. Area Area Rel. Area Sample Name Time [mAU*min] [%] [mAU*min] [%] [mAU*min] [%] [mAU*min] [%] Ref. Lot 255.61 98.00 3.98 1.52 1.50 0.57 0.59 0.23 Formulation 6.1 T0 222.94 97.43 3.89 1.70 1.91 0.83 0.09 0.04 Formulation 6.1 T1 5.degree. C. 369.72 97.57 6.31 1.66 2.75 0.73 0.18 0.05 Formulation 6.1 T1 40.degree. C. 405.49 97.35 7.33 1.76 3.60 0.86 0.12 0.03 Formulation 6.1 T2 5.degree. C. 422.46 97.59 7.01 1.62 2.74 0.63 0.68 0.16 Formulation 6.1 T2 40.degree. C. 289.65 97.28 5.50 1.85 2.13 0.72 0.48 0.16 Formulation 6.2 T0 230.06 97.61 3.93 1.67 1.64 0.70 0.07 0.03 Formulation 6.2 T1 5.degree. C. 407.17 97.56 6.81 1.63 3.23 0.77 0.17 0.04 Formulation 6.2 T1 40.degree. C. 468.74 97.36 8.79 1.83 3.78 0.78 0.16 0.03 Formulation 6.2 T2 5.degree. C. 552.31 97.64 9.80 1.73 2.96 0.52 0.61 0.11 Formulation 6.2 T2 40.degree. C. 249.95 96.78 5.39 2.09 2.39 0.93 0.52 0.20 Formulation 11 T0 211.45 97.49 3.64 1.68 1.47 0.68 0.35 0.16 Formulation 11 T1 5.degree. C. 339.08 97.71 5.45 1.57 2.28 0.66 0.23 0.07 Formulation 11 T1 40.degree. C. 700.91 97.30 12.69 1.76 5.19 0.72 1.60 0.22 Formulation 11 T2 5.degree. C. 325.80 97.17 5.80 1.73 2.17 0.65 1.52 0.45 Formulation 11 T2 40.degree. C. 229.29 96.96 4.33 1.83 1.78 0.75 1.09 0.46
TABLE-US-00030 TABLE 12 SDS-PAGE data of Formulations 6.1-6.2 & 11 SDS-PAGE Analysis ANTIBODY Main 2. Band HM Additional bands Sample Name Sample ID size Rel. QTY size Rel. QTY size Rel. QTY size Rel. QTY comment Ref. Lot 182.9 95.6 161.2 2.3 73.8 0.5 Formulation 6.1 T0 175.6 94.7 156.1 2.7 73.5 0.5 Formulation 6.1 T1 5.degree. C. 180.2 86.9 159.9 11.4 75.9 0.1 Formulation 6.1 T1 40.degree. C. 179.2 90.4 158.5 7.5 76.1 0.4 Formulation 6.1 T2 5.degree. C. 177.3 95.6 157.9 2.1 74.9 0.3 Formulation 6.1 T2 40.degree. C. 179.8 94.7 159.8 2.9 75.4 0.3 Formulation 6.2 T0 176.6 94.9 156.3 2.6 73.6 0.5 Formulation 6.2 T1 5.degree. C. 180.2 89.8 159.3 7.9 76.3 0.4 Formulation 6.2 T1 40.degree. C. 182.1 88.7 160.9 9.4 76.3 0.1 Formulation 6.2 T2 5.degree. C. 177.5 95.5 160.2 2.9 75.4 0.2 Formulation 6.2 T2 40.degree. C. 180.9 95.5 161.5 2.4 75.7 0.3 Formulation 11 T0 178.7 95.1 156.5 2.3 73.7 0.4 Formulation 11 T1 5.degree. C. 181.0 70.0 154.7 25.7 74.5 0.3 Formulation 11 T1 40.degree. C. 181.3 66.2 154.2 28.9 74.5 0.3 Formulation 11 T2 5.degree. C. 177.7 87.5 155.9 10.9 75.2 0.3 Formulation 11 T2 40.degree. C. 176.8 86.2 155.2 12.0 74.5 0.3
Example 5--Accelerated Stability Study
[0321] An accelerated stability study was performed with citrate and histidine buffers. Table 13 shows the analytical results of the first batch of Reference Lot, and the various experimental formulations of the Lead LIGHT Antibody formulated into various combinations of citrate buffer or histidine buffer. Notably, the citrate formulation of the invention appeared in all experiments to perform better than histidine. In particular, citrate formulations had a higher monomer content compared to the both the Reference Lot batch and the histidine (Table 13) and the content or low molecular weight proteins (LMWPs) and high molecular weight proteins (HMWPs) were also significantly lower (Table 14). As before, these differences could not be detected with SDS-PAGE analysis (Table 15).
TABLE-US-00031 TABLE 13 Analytical data of Formulations 7, 8, 9 & 10 Sample Tm1 Tm2 Tm3 ZAve Concentration number [.degree. C.] [.degree. C.] [.degree. C.] pH [nm] [mg/mL] Buffer Ref. Lot 67.94 75.00 77.37 7.3 10.05 5.5 PBS Formulation 7 58.95 68.51 76.20 5.5 12.97 53.65 Histidine 10 mM Formulation 8 58.69 68.23 76.12 5.4 13.29 58.72 Histidine 10 mM PS20 0.01% Formulation 9 61.67 72.01 76.53 5.6 59.26 55.01 Citrate 10 mM Formulation 10 62.24 72.32 76.61 5.6 17.3 55.8 Citrate 10 mM PS20 0.01%
TABLE-US-00032 TABLE 14 SEC Analysis of Formulations 7 & 8 & 9 & 10 ANTIBODY RRT0.8 LMWP HMWPs Monomer Area Rel. Area Area Rel. Area Area Rel. Area Area Rel. Area Content Sample Name Time [mAU*min] [%] [mAU*min] [%] [mAU*min] [%] [mAU*min] [%] [mg/mL] Ref. Lot 282.42 97.40 4.55 1.57 2.15 0.74 0.85 0.29 Formulation 7 T0 184.4 95.1 163.8 1.6 72.8 1 Formulation 7 T1 5.degree. C. 390.84 97.85 5.72 1.43 2.80 0.70 0.06 0.01 Formulation 7 T1 40.degree. C. 379.81 96.74 7.04 1.79 5.77 1.47 0 0 Formulation 7 T2 5.degree. C. 863.01 97.75 14.19 1.61 4.14 0.47 1.54 0.18 164.88 Formulation 7 T2 40.degree. C. 1085.91 95.22 29.59 2.60 23.25 2.04 1.69 0.15 207.47 Formulation 8 T0 184.6 94.9 165.9 2.1 72.7 0.8 Formulation 8 T1 5.degree. C. 507.64 97.74 7.52 1.45 4.19 0.81 0.05 0.01 Formulation 8 T1 40.degree. C. 461.44 96.98 8.05 1.69 6.19 1.30 0 0 Formulation 8 T2 5.degree. C. 416.54 97.46 6.59 1.54 3.49 0.82 0.79 0.18 79.58 Formulation 8 T2 40.degree. C. 422.21 93.23 11.17 2.47 18.40 4.06 1.11 0.25 80.66 Formulation 9 T0 229.01 97.63 3.75 1.60 1.63 0.70 0.19 0.08 45.28 Formulation 9 T1 5.degree. C. 307.94 97.96 4.2 1.34 2.20 0.7 0 0 Formulation 9 T1 40.degree. C. 319.10 97.54 5.24 1.60 2.59 0.79 0.23 0.07 Formulation 9 T2 5.degree. C. 337.15 97.48 5.41 1.56 2.84 0.82 0.49 0.14 64.41 Formulation 9 T2 40.degree. C. 325.54 96.26 7.78 2.30 3.66 1.08 1.20 0.36 62.20 Formulation 10 T0 233.11 97.54 3.84 1.61 1.97 0.82 0.08 0.03 46.09 Formulation 10 T1 5.degree. C. 343.38 97.77 5.21 1.48 2.58 0.73 0.04 0.01 Formulation 10 T1 40.degree. C. 329.56 97.21 5.06 1.49 4.29 1.26 0.13 0.04 Formulation 10 T2 5.degree. C. 343.33 97.43 5.47 1.55 3.06 0.87 0.53 0.15 65.59 Formulation 10 T2 40.degree. C. 257.20 94.59 5.59 2.06 8.98 3.30 0.15 0.05 49.14
TABLE-US-00033 TABLE 15 SDS-PAGE data of Formulations 7 & 8 & 9 & 10 SDS-PAGE Analysis ANTIBODY Main 2. band Half molecules Additional bands size Rel. QTY size Rel. QTY size Rel. QTY size Rel. QTY Sample Name Time/Temp [kDa] [%] [kDa] [%] [kDa] [ %] [kDa] [%] comment Ref. Lot 173.6 96.3 155.8 2.2 74 0.4 Formulation 7 T0 184.4 95.1 163.8 1.6 72.8 1 Formulation 7 T1 5.degree. C. 183.0 91.1 159.9 7.2 76.1 0.4 Formulation 7 T1 40.degree. C. 182.2 83.1 158.4 13.8 74.0 0.4 Formulation 7 T2 5.degree. C. 181.5 95.7 160.3 2.7 75.6 0.3 Formulation 7 T2 40.degree. C. 173.0 84.6 151.1 10.3 73.9 0.7 12.1 0.9 more LMWPs Formulation 8 T0 184.6 94.9 165.9 2.1 72.7 0.8 Formulation 8 T1 5.degree. C. 180.1 86.2 158.3 11.4 73.9 0.4 Formulation 8 T1 40.degree. C. 180.9 79.4 158.2 16.9 74.0 0.3 Formulation 8 T2 5.degree. C. 175.1 95.2 154.9 3.1 74.4 0.3 Formulation 8 T2 40.degree. C. 174.8 84.7 150.5 9.2 74.0 0.9 12.1 1.5 more LMWPs Formulation 9 T0 187.7 95.5 163.1 1.1 72.9 0.9 Formulation 9 T1 5.degree. C. 178.9 65.8 160.4 29.6 73.7 0.9 Formulation 9 T1 40.degree. C. 184.7 82.9 160.3 14.8 74.4 0.3 Formulation 9 T2 5.degree. C. 176.2 95.6 155.6 2.6 73.6 0.3 Formulation 9 T2 40.degree. C. 174.3 91.5 153.9 3.1 73.1 0.3 12.1 0.2 more LMWPs Formulation 10 T0 182.5 95.2 161.3 1.6 72.3 0.8 Formulation 10 T1 5.degree. C. 184.5 68.4 156.4 26.6 75.1 0.3 Formulation 10 T1 40.degree. C. 180.8 65.4 153.8 28.8 74.8 0.3 Formulation 10 T2 5.degree. C. 188.7 88.6 165.0 9.6 73.5 0.2 Formulation 10 T2 40.degree. C. 181.7 78.9 158.2 15.8 75.7 0.8 12.6 1.3 more LMWPs
Example 6--Development of High Antibody Concentration Formulation for Subcutaneous Administration
[0322] Based on the successful results of the citrate-buffered formulations of Examples 2-5, a high-concentration (150 mg/ml) antibody formulation suitable for subcutaneous administration was developed. Formulation development was performed on the Lead LIGHT Antibody with the goal of developing a liquid dosage form with an acceptable shelf life when stored at +2 to +8.degree. C. Preliminary stress studies showed the formation of subvisible and visible particles, high molecular weight species and more basic species. Therefore, these parameters were monitored during the screening of formulation candidates using visual assessment, dynamic light scattering, light obscuration, size exclusion chromatography, sodium dodecyl sulphate polyacrylamide gel electrophoresis, and weak cationic exchange chromatography. Different liquid formulations were used in the pre-formulation and formulation trials prior to selection of the clinical formulation. According to the findings, a formulation in 10 mM citrate buffer adjusted to pH 5.5 (Formulation 14) was selected for further development. The pH of the formulation is in the region of optimal physical and chemical stability of the drug substance and acceptable physiological tolerability (e.g., osmolarity).
[0323] As shown in Table 16, Formulation 14 is a solution for injection and is an aqueous, sterile, and clear solution containing the Lead LIGHT Antibody, sodium citrate dihydrate (buffering agent), polysorbate 20 (stabilizing agent), and mannitol (tonicity agent). Sodium hydroxide solution and hydrochloric acid were used to adjust the pH to 5.5.
[0324] All excipients were soluble and well tolerated pharmacopoeial standard excipients for parenterals and listed in Ph. Eur. and USP.
TABLE-US-00034 TABLE 16 Composition Composition per mL per vial (1.2 mL) Reterence to Components.sup.a (mg) (mg) Function standards.sup.b Lead Antibody 150.00 180.00 Drug substance In-house Sodium citrate dehydrate 2.94 3.53 Buffering agent Ph. Eur., USP Mannitol 40.00 48.00 Tonicity agent Ph. Eur., USP Polysorbate 20 0.05 0.06 Stabilizing agent Ph. Eur., NF, JP Hydrochloric acid, q.s pH 5.5 q.s. pH 5.5 Acidifying agent Ph. Eur., NF concentrated [Hydrochloric acid] Sodium hydroxide q.s. pH 5.5 q.s. pH 5.5 Alkalizing agent Ph. Eur., NF Water for injection q.s. 1 mL q.s. 1.2 mL Solvent Ph. Eur., USP Nitrogen Process aid for filtration Ph. Eur., NF .sup.aComponents are listed according to their pharmacopoeial names. If more than one monograph exists, other names are given in brackets, along with the compendial origin. .sup.bReference is made to the current edition of the Pharmacopoeia.
Example 7--Manufacturing Process for Subcutaneous Antibody Formulation
[0325] A GMP-compliant manufacturing process was developed for the subcutaneous, high-concentration antibody formulation (Formulation 14) of Example 6. The manufacturing procedure consisted of dissolving, pH adjustment, sterile filtration, filling, and packaging steps.
[0326] Drug substance (the Lead LIGHT Antibody) is provided in a liquid form in the formulation buffer (10 mM citrate buffer at pH 5.5). The excipients were all water-soluble and dissolved in the initial aqueous portion of the formulation buffer during manufacture. The bulk drug substance solution was further diluted with the same formulation buffer to reach the concentration of 150 mg/mL of Lead LIGHT Antibody. The bulk solution was well mixed to facilitate the dissolution process and to ensure homogeneity.
[0327] Sterilization by filtration was carried out (according to Ph. Eur. and USP) using bacteria retentive filters having a nominal pore size of 0.2 .mu.m. An additional filtration procedure before "sterilization by filtration" was performed to ensure a low bioburden. Bioburden sampling was done before the pre-filtration step as well as the sterile filtration step.
[0328] Preparation and filling of the sterilized solution into the suitable containers was performed under aseptic conditions. This was in accordance with pharmacopoeial monographs and related guidelines, which required sterilization by filtration and subsequent aseptic processing. The equipment, which comes into contact with the product after the step "sterilization by filtration", was sterilized by heat or steam using a validated method (according to Ph. Eur./USP).
[0329] Vials were filled to about 1.2 mL to ensure an extractable volume of 1.0 mL. The 2 mL vials were made of clear, colorless type I glass, and closed with a stopper (fluoropolymer-coated bromobutyl) sealed with flip-off caps with a flange (polypropylene). The primary packaging materials met the requirements of the Ph. Eur. and USP. The suitability of the primary packaging materials was substantiated by the results of the stability tests. Incompatibilities with the primary packaging material used were not observed. Secondary packaging which provides protection of the product from light.
[0330] Compatibility with application syringes was assessed using 3 different syringe sizes and needles diameters (between 25 and 28 gauges) on the drug product solution. No differences in terms of product quality were obtained. Compatibility was proven for a time period of 8 hours.
[0331] Formulation 14 was made in 5 L batches, the composition of which is shown in Table 17. However, the batch size may be adjusted according to clinical requirements.
TABLE-US-00035 TABLE 17 Batch formula Batch size 5 Liter.sup.a Components [g] Lead Antibody .sup.b 750.00 Mannitol 200.00 Poly sorbate 20 0.25 Sodium citrate dihydrate 14.71 Hydrochloric acid, concentrated .sup.c q.s. pH 5.5 Sodium hydroxide .sup.c q.s. pH 5.5 Water for injection Ad 5285.25 .sup.d Nitrogen Process aid for filtration .sup.aThe vials were filled with a volume of 1.2 mL to ensure an extractable volume of 1.0 mL. A 6.0 L batch size therefore results in a theoretical yield of 5000 vials. .sup.b For pH adjustment. .sup.c This was calculated according to the density of the final drug product solution (1.05705 mg/mL)
[0332] The manufacturing process and process controls for Formulation 14 are shown in the flow diagram in FIG. 9. Batch manufacturing included the following steps: [0333] I. Sodium citrate dihydrate was dissolved in water for injection while stirring in a vessel made of inert material (e.g., stainless steel or glass), until completely dissolved. The pH value was adjusted to 5.5 using hydrochloric acid, diluted (e.g., 0.1 M hydrochloric acid) and/or sodium hydroxide solution (e.g., 0.1 M sodium hydroxide), if necessary. [0334] II. Lead Antibody, mannitol, and polysorbate 20 were diluted in the buffer solution from step 1 while stirring in a vessel made of inert material (e.g., stainless steel or glass) until completely dissolved. If necessary, the pH value was adjusted to 5.5 using hydrochloric acid, diluted (e.g., 1 M hydrochloric acid) or sodium hydroxide solution (e.g., 1 M sodium hydroxide). Buffer solution from step 1 (remainder) was added to adjust the final weight. [0335] III. a) Pre-filtration: [0336] Solution from step II was filtered under aseptic conditions using a sterilized, compatible membrane filter (e.g., polyether sulfone or polyvinylidene difluoride) having a nominal pore size of 0.2 .mu.m. [0337] b) Sterilization by filtration: [0338] Solution from step III.a was sterilized by filtration under aseptic conditions into sterilized containers made out of inert material (e.g., stainless steel or glass) using a sterilized, compatible membrane filter (e.g., polyether sulfone or polyvinylidene difluoride) having a nominal pore size of 0.2 .mu.m. [0339] IV. Solution from step III.b was filled under aseptic conditions into sterilized vials, which were closed with stoppers and flip-off caps with a flange. [0340] V. The containers from step IV were inspected for coarse contaminants, intact sealing, and visible particles. [0341] VI. The inspected containers from step V were additionally packaged in suitable containers (e.g., cardboard boxes).
[0342] In addition, DLS was used to determine the hydrodynamic diameter of the antibody monomer and potential soluble aggregates. As shown in FIG. 10, aggregates were not seen in citrate buffer. However, as shown in FIGS. 7 & 8, aggregates were seen in PBS. Due to the higher concentration of antibody, an increase in ZAve to 28 nm was observed, compared to the sample in PBS.
Example 8--Stability Profile for Subcutaneous Antibody Formulation
[0343] The stability profile of the clinical batch (batch 2) of Example 7 was assessed for storage under long term and accelerated testing conditions according to ICH guidelines. Samples were packed and stored in 2 mL clear and colorless vials (glass type I) closed with stoppers (fluoropolymer-coated bromobutyl) and flip-off caps with a flange (polypropylene).
[0344] The following tests were performed during stability: appearance (clarity, color), assay (antigen ELISA, UV), purity (SEC, SDS-PAGE under reducing and non-reducing conditions), molecular integrity (SDS-PAGE under non-reducing conditions), charge heterogeneity (weak cation exchange chromatography, isoelectric focusing), pH, sterility, bacterial endotoxins, particulate matter (visible and subvisible particles), and closure integrity.
[0345] The samples were stored in inverted and upright positions. The results of the inverted storage were presented as the more stringent condition. Stability data at -20.degree. C., +5.degree. C. and +25.degree. C. are presented in Tables 18-20, respectively. The investigations on physical, chemical, and biological properties of storage under long term testing conditions confirmed a good stability of the drug product at 5.degree. C. (see Table 19). Under accelerated testing conditions (+25.degree. C.), only a slight decrease in the purity was detected by size exclusion chromatography (see Table 20). Therefore, it was concluded that the drug product should be stored at +2.degree. C. to +8.degree. C. protected from light.
TABLE-US-00036 TABLE 18 Long term stability at -20.degree. C. for drug product Drug product: Lead LIGHT Antibody solution for injection Batch no.: 11_021 Dosage strength: 150 mg/mL Formulation no.: 14 Container/closure: 2 mL glass vials Storage condition: -20.degree. C. .+-. 5.degree. C. Storage Upright orientation: Test item Time Initial 1 3 6 12 18 24 results month months months months months months Appearance of solution Clarity <I III II >IV IV >IV Color BY7 BY7 BY6 BY6 BY6 BY7 Identification IEF Isoelectric pattern Conforms Conforms Conforms Conforms Conforms Conforms Assay Antigen-ELISA EC50 value (in 76% 110% 76% 103% 96% 105% comparison to reference Total protein content 153 mg/mL 148 mg/mL 151 mg/mL 151 mg/mL 149 mg/mL 156 mg/mL (UV) Purity HPLC (SEC) Monomer (% area) 98.2%.sup. 97.5% 96.2%.sup. 94.5% 94.3%.sup. 94.1% HMWPs (% area) 1.3% 2.3% 3.7% 5.4% 5.5% 5.5% LMWPs (% area) 0.4% 0.2% 0.1% 0.0% 0.1% 0.5% SDS-PAGE under non reducing conditions Half molecules <1.0%.sup. <1.0% <1.0%.sup. 2.7% <1.0%.sup. <5.0% SDS-PAGE under reducing conditions Relative purity 98% 100% 100% 100% 96% 100% Molecular integrity SDS-PAGE under non- reducing conditions Gel pattern Conforms Does not Conforms Conforms Conforms Conforms conform Charge heterogeneity HPLC (WCX) acidic 40% 36% 44% 42% 43% 41% neutral 55% 60% 54% 47% 51% 56% basic isoforms 5% 4% 2% .sup. 2% 6% 3% (% area) pH (potentiometry) 5.5 5.5 5.5 5.5 5.5 5.5 Particulate matter Complies Complies Complies Complies Complies Complies (visible particles) Particulate matter 313 Not tested Not tested Not tested 33 Not tested (subvisible particles) Number of particles per 10 5 vial .gtoreq.10 .mu.m Number of particles per vial .gtoreq.25 .mu.m Closure integrity Complies Not tested Not tested Not tested Complies Not tested Microbial contamination TAMC <1 CFU/2 mL Not tested Not tested Not tested Not tested Not tested TAnMC <1 CFU/2 mL Not tested Not tested Not tested Not tested Not tested
TABLE-US-00037 TABLE 19 Long term stability at +5.degree. C. for drug product Drug product: Lead LIGHT Antibody solution for injection Batch no.: 11_021 Dosage strength: 150 mg/mL Formulation no.: 14 Container/closure: 2 mL glass vials Storage condition: +5.degree. C. .+-. 3.degree. C. Storage Upright orientation: Test item Time Initial 1 3 6 12 18 24 results month months months months months months Appearance of solution Clarity <I <I <I <I <I >IV Color BY7 BY7 BY7 BY7 BY6 BY7 Identification IEF Isoelectric pattern Conforms Conforms Conforms Conforms Conforms Conforms Assay Antigen-ELISA EC50 value (in 76% 119% 83% 107% 96% 115% comparison to reference Total protein content 153 mg/mL 150 mg/mL 150 mg/mL 151 mg/mL 148 mg/mL 155 mg/mL (UV) Purity HPLC (SEC) Monomer (% area) 98.2%.sup. 98.5% 98.5%.sup. 98.3% 98.0%.sup. 97.4% HMWPs (% area) 1.3% 1.4% 1.5% 1.7% 1.9% 2.0% LMWPs (% area) 0.4% 0.0% 0.0% 0.0% 0.1% 0.6% SDS-PAGE under non reducing conditions Half molecules <1.0%.sup. <1.0% <1.0%.sup. 1.8% <1.0%.sup. <1.0% SDS-PAGE under reducing conditions Relative purity 98% 100% 100% 100% 96% 100% Molecular integrity SDS-PAGE under non- reducing conditions Gel pattern Conforms Does not Conforms Conforms Conforms Conforms conform Charge heterogeneity HPLC (WCX) acidic 40% 36% 44% 42% 43% 39% neutral 55% 60% 54% 57% 52% 57% basic isoforms 5% 4% 2% .sup. 2% 5% 4% (%area) pH (potentiometry) 5.5 5.5 5.5 5.5 5.5 5.5 Particulate matter Complies Complies Complies Complies Complies Complies (visible particles) Particulate matter 313 Not tested Not tested Not tested 35 Not tested (subvisible particles) Number of particles per 10 5 vial .gtoreq.10 .mu.m Number of particles per vial .gtoreq.25 .mu.m Closure integrity complies Not tested Not tested Not tested Complies Complies Microbial contamination TAMC <1 CFU/2 mL Not tested Not tested Not tested Not tested Not tested TAnMC <1 CFU/2 mL Not tested Not tested Not tested Not tested Not tested
TABLE-US-00038 TABLE 20 Accelerated stability at +25.degree. C. for drug product Drug product: Lead LIGHT Antibody solution for injection Batch no.: 11_021 Dosage strength: 150 mg/mL Formulation no.: 14 Container/closure: 2 mL glass vials Storage condition: +25.degree. C. .+-. 2.degree. C./60% .+-. 5% RH Storage Upright orientation: Time Initial 1 3 6 Test item results month months months Appearance of solution Clarity <I <I <I <I Color BY7 BY7 BY7 BY7 Identification IEF Isoelectric pattern Conforms Conforms Conforms Conforms Assay Antigen-ELISA EC50 value (in 76% 108% 96% 111% comparison to reference Total protein content (UV) 153 mg/mL 149 mg/mL 150 mg/mL 151 mg/mL Purity HPLC (SEC) Monomer (% area) 98.2%.sup. 98.2% 97.7%.sup. 96.8% HMWPs (% area) 1.3% 1.7% 2.2% 3.1% LMWPs (% area) 0.4% 0.1% 0.1% 0.1% SDS-PAGE under non <1.0%.sup. <1.0% <1.0%.sup. <1.0% reducing conditions Half molecules SDS-PAGE under 98% 100% 100% 100% reducing conditions Relative purity Molecular integrity SDS-PAGE under non- Conforms Does not Conforms Conforms reducing conditions conform Gel pattern Charge heterogeneity HPLC (WCX) acidic 40% 36% 44% 41% neutral 55% 59% 53% 56% basic isoforms (% area) 5% 5% 2% 3% pH (potentiometry) 5.5 5.5 5.5 5.5 Particulate matter (visible Complies Complies Complies Complies particles) Particulate matter Not tested Not tested (subvisible particles) Number of particles per 313 22 vial .gtoreq.10 .mu.m Number of particles per 10 1 vial .gtoreq.25 .mu.m Closure integrity complies Not tested Not tested Complies Microbial contamination TAMC <1 CFU/2 mL Not tested Not tested <1 CFU/2 mL TAnMC <1 CFU/2 mL Not tested Not tested <1 CFU/2 mL
Example 9--Development of Ultra-High Antibody Concentration Formulation for Subcutaneous Administration
[0346] Based upon the successful results of the citrate-buffered formulations for antibody concentrations up to 150 mg/mL in Example 7, higher concentrated (up to 250 mg/ml) antibody formulations suitable for subcutaneous administration were developed.
[0347] Preliminary data showed that the formulation of antibody concentrations above 150 mg/mL may lead to higher viscosities affecting usage of the formulation.
TABLE-US-00039 TABLE 21 Ultra high concentrations with formulation 14 DLS Size exclusion Concentration Density Viscosity z-average chromatography Sample [mg/mL] [kg/m-3] [mPa] [nm] HMWPs Monomer LMWPs Lead LIGHT Antibody_11_30A 237 1.066 42.29 30 1.3 98.6 0.0 Lead LIGHT Antibody_11_30B 212 1.059 22.58 39 1.3 98.7 0.0 Lead LIGHT Antibody_11_30C 181 1.052 13.57 28 1.3 98.7 0.1 Lead LIGHT Antibody_11_30D 173 1.046 8.8 27 1.2 98.8 0.0 Lead LIGHT Antibody_11_30E 143 1.039 6.16 25 1.1 98.8 0.1
[0348] As can be seen in Table 21, the viscosity decreases with lower antibody concentrations, yet still being in an acceptable range at the higher concentration formulated with formulation 14. All other parameters seemed to be unaffected or just slightly affected by the ultra-high concentrations.
[0349] As shown in Table 22, the antibody concentrations did not affect the stability of the formulations, which was indicated by identical 1 month stability data at long term and stress conditions.
TABLE-US-00040 TABLE 22 1 month stability data of ultra high concentrated Lead Antibody formulations Concentration [mg/mL] HMWPs Monomer LMWPs Lead LIGHT Antibody_11_30A 40.degree. C. 237 4.7 95.2 0.2 Lead LIGHT Antibody_11_30B 40.degree. C. 212 4.4 95.4 0.2 Lead LIGHT Antibody_11_30C 40.degree. C. 181 5.8 91.7 2.6 Lead LIGHT Antibody_11_30D 40.degree. C. 173 3.9 96.0 0.2 Lead LIGHT Antibody_11_30E 40.degree. C. 143 4.2 94.7 1.1 Lead LIGHT Antibody_11_30A 5.degree. C. 237 1.4 98.6 0.0 Lead LIGHT Antibody_11_30B 5.degree. C. 212 1.3 98.7 0.0 Lead LIGHT Antibody_11_30C 5.degree. C. 181 1.3 98.7 0.0 Lead LIGHT Antibody_11_30D 5.degree. C. 173 1.2 98.8 0.0 Lead LIGHT Antibody_11_30E 5.degree. C. 143 1.1 98.9 0.0
Anti-CXCR5 (20 mg/mL) Pre-Formulation Studies
[0350] A humanized IgG4 anti-CXCR5 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 25 and a light chain comprising the amino acid sequence of SEQ ID NO: 26 (the "Lead CXCR5 Antibody") was used in Examples 10-12 in order to determine optimal formulation conditions for a 20 mg/mL formulation.
[0351] The Lead Antibody is a humanized monoclonal antibody (mAB) specific to human CXCR5, with an engineered IgG4 framework containing 2 amino acid substitutions aimed at reducing half-molecules (S241P) and effector functions (L248E). The Lead CXCR5 Antibody protein structure is comprised of two kappa light chains, each with a molecular weight of approximately 24 kDa, and two IgG4 heavy chains, each with a molecular weight of approximately 48 kDa linked through disulfide bridges. Each light chain consists of 219 amino acid residues, and each heavy chain consists of 437 amino acid residues.
[0352] The data in Examples 10-12 were collected during preformulation activities for the Lead CXCR5 Antibody and its drug product for intravenous and subcutaneous administration. The objective of the preformulation studies was to provide good stability of buffered Lead CXCR5 Antibody solutions with a target concentration of 20 mg/mL, with special emphasis on the aggregation behavior of the Lead CXCR5 Antibody and its tendency to form half-molecules, as the Lead Antibody is an IgG4 subclass antibody, which is prone to aggregation and the formation of particles.
Materials
Drug Substance (DS)
[0353] The Lead CXCR5 Antibody batch RSN0151 was formulated in PBS pH 7.2 with a concentration of 5.13 mg/mL.
Excipients
[0354] Table 23 shows excipients that were used during the preformulation studies.
TABLE-US-00041 TABLE 23 Excipients used during preformulation Excipients Art. No./Charge Supplier Acetic acid A002630 MTP/VWR International SAS Arginine-HCl A1700 AppliChem Arginine 1.01587 Merck Benzyl alcohol 113594 Industrial Affairs/Harrmann & Reimer Citric acid 100241 Merck Dextran 40 CL-AO19A Meito Sangyo Glycine 113560 Industrial Affairs.sup.1/Tessenderlo Chemie. HCl 114027 Industrial Affairs.sup.1/Merck Histidine 1.04352 Merck Potassium dihydrogen phosphate 1.04871 Merck Lysine 62840 Fluka Magnesium chloride 814733 Merck Maltose 105911 Merck Mannitol A000780 MTP/Roquette Freres Sodium acetate 1.06265 Merck Sodium chloride 10158 Industrial Affairs.sup.1/Riedel de Haen Sodium hydroxide 114076 Industrial Affairs.sup.1/Merck Sodium citrate 114196 Industrial Affairs.sup.1/Boehringer Ingelheim KG Di-sodiumhydogenphosphate anhydrous 1.06586 Merck Polysorbate 20 139850 Industrial Affairs.sup.1/Fluka Succinate/Succinic acid 14079 Fluka Sucrose S3929 Sigma-Aldrich Trehalose-dihydrate T9531 Sigma-Aldrich Trometamol 114011 Industrial Affairs/Merck
Methods
[0355] The following methods were used to manufacture the experimental formulations and the formulations of the invention containing the Lead CXCR5 Antibody.
Manufacturing & Composition of Buffers
[0356] All buffers were manufactured by stirring constantly to dissolve the respective excipients. pH was adjusted using 0.1 M HCl or 0.1 M NaOH. The general concentration of all buffers was 10 mM.
Manufacturing & Composition of Excipient Stock Solutions
[0357] All stock solutions were manufactured by stirring constantly to dissolve the excipients. Concentrations were given as weight/weight (w/w).
UF/DF--Small Scale
[0358] UF/DF experiments were performed using Vivaspin units (Sartorius Stedim, Gottingen, Germany) with a Hydrosart membrane and a 30 kDa cut-off for removing phosphate buffer and replacing it with the appropriate buffers and to increase the concentration. These units were placed in a common laboratory centrifuge (Multifuge 3S, Haereus, Germany) and centrifuged at 2000 rpm (860 G, rotor radius 192 mm) at +5.degree. C.
UF/DF--Larger Scale
[0359] UF/DF experiments were performed using Vivaflow units (Sartorius Stedim, Gottingen, Germany) with a Hydrosart membrane and a 30 kDa cut-off for removing phosphate buffer and replacing it with the appropriate buffers and to increase the concentration. The equipment was placed inside a clean-bench under aseptic conditions and the process was performed at room temperature.
Sterile Filtration of Samples
[0360] All samples, solutions, buffers, etc. were sterile filtered (0.22 .mu.m) using a Sartopore-2 membrane. The samples were filtered into sterilized bottles or vials and closed under aseptic conditions inside a clean-bench to prevent microbiological contamination.
Mechanical Stress Test
[0361] Mechanical stress with an agitation speed of 350 rpm/min for 2.5 hours at room temperature was performed using a horizontal laboratory shaker with a 26 mm distance (shaker & incubation hood from Buhler Company). 2R vials were filled with 1 mL solution with a head space of about 2.5 cm.sup.3.
[0362] A mechanical stress test was planned and performed during the first preformulation studies. As the analytical results did not show any additional information compared to the thermal stress tests, during buffer selection or pH selection, this test was only used during surfactant selection.
Thermal Stress Test
[0363] Thermal stress was used as an accelerated stress test during all steps of the preformulation program. The samples were stored at +40.degree. C. either for 24 h, 7 days, or 3 months, depending on the study.
Analytical Methods in Formulation Fill and Finish
[0364] The following analytical methods were used in the following examples.
Appearance
[0365] Appearance of the antibody solutions was checked visually and additionally documented by taking a picture with a camera.
pH
[0366] All pH measurements were performed using a pH-meter with a micro-electrode.
Concentration Using UV
[0367] The protein concentration of all antibody solutions was measured against buffer using a Nanodrop ND1000. Protein concentrations near or below 5 mg/mL were diluted 1:3, and higher protein concentrations near 20 mg/mL were diluted 1:20, before measuring the absorption at 215 nm and 280 nm.
Dynamic Light Scattering (DLS)
[0368] The hydrodynamic diameter of the molecule was measured using laser light scattering. The samples were sterile filtered prior to the analytics if turbidity was observed, thus only soluble aggregates could be detected.
Thioflavine-T Test
[0369] Fluorescence measurements of some preformulation samples were carried out using a Tecan GENios Plus, XFLOUR4. The samples were stressed mechanically (4 h at +37.degree. C., agitation speed 300 units/min and 26 mm distance in a shaker & incubator hood from Biihler company). Thioflavin-T fluorescence spectra were measured at room temperature. 10 .mu.l Thioflavin-T solution (10.1 mM in ultrapure water) was added to 1 ml of the formulations and gently mixed. The mixture was then dispensed into a black Eppendorf V-shaped cup, and then into a 96-well plate (100 .mu.L per well).
[0370] The thioflavin-T test was used in the beginning of preformulation activities to detect insoluble aggregates. But, as these aggregates can be seen visually as a turbidity of the solution, this method was not used for the whole preformulation program.
Differential Scanning Calorimetry (DSC)
[0371] Aliquots of the preformulation samples were examined by DSC using a VPCapillary DSC from Microcal and scanned in the auto sampling instrument at 90.degree. C./h with a filter time of 2 sec. 400 .mu.l samples were placed into 96-well plates and analyzed for the unfolding temperature Tm.
Osmolarity
[0372] Osmolarity was measured using an automated Knaur Osmometer.
Density
[0373] Density of the formulations was measured using a falling sphere viscosimeter DMA4500 Anton Paar.
Analytical Methods in Bioanalytics FF
Size Exclusion Chromatography (SEC)
[0374] Oligomers/dimers of the antibodies were quantified by using size exclusion chromatography. The test was carried out by isocratic HPLC with a SUPERDEX 200 10/300 column.
SDS-PAGE, Reducing and Non-Reducing
[0375] Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to analyze the molecular integrity (e.g., half molecules) and appearance of degradation products. This electrophoresis analysis was performed with 15% homogenous gels under reducing and non-reducing conditions. The proteins were visualized with silver staining after electrophoresis separation.
WCX
[0376] Weak cationic exchange chromatography (WCX) was used to monitor the charge heterogeneity of the antibody. The percentage of basic, neutral, and acidic isoforms was reported. The test was carried out by discontinuous HPLC with a ProPac WCX10 column.
Antigen-ELISA
[0377] Antigen-ELISA was performed to determine the functionality of the antibody. The binding property to a 28mer peptide of the CXCR5 antigen was monitored in comparison to the current standard of the antibody. This potency was reported as the relative EC50.
Isoelectric Focusing (IEF)
[0378] IEF was performed.
Storage
[0379] All buffer solutions, excipient solutions, and samples were stored at +5.degree. C., if not otherwise mentioned.
Summary of all Formulations Prepared & Analyzed
[0380] Table 24 below shows a summary of all of the formulations that were prepared and analyzed in Examples 10-12. Each of the formulations contained the Lead CXCR5 Antibody. PBS stands for phosphate buffered saline. PB stands for phosphate buffer. PS stands for polysorbate. LA stands for the Lead CXCR5 Antibody.
TABLE-US-00042 TABLE 24 Summary of all formulations prepared and analyzed Sample number Buffer pH LA_09_05-1 PBS 155 mM 7.5 LA_09_05-2 PBS 155 mM 7.0 LA_09_05-3 PBS 155 mM 6.5 LA_09_06-1 PB 5 mM 7.5 LA_09_06-2 PB 5 mM 7.0 LA_09_06-3 PB 5 mM 6.5 LA_09_07-1 PB 10 mM 7.5 LA_09_07-2 PB 10 mM 7.0 LA_09_07-3 PB 10 mM 6.5 LA_09_08-1 Citrate 10 mM 7.0 LA_09_08-2 Citrate 10 mM 6.5 LA_09_08-3 Citrate 10 mM 6.0 LA_09_08-4 Citrate 10 mM 5.5 LA_09_08-5 Citrate 10 mM 5.0 LA_09_09-1 Saline 150 mM 6.0 LA_09_10-1 Acetate 10 mM 5.5 LA_09_10-2 Acetate 10 mM 5.0 LA_09_11-1 Succinate 10 mM 6.0 LA_09_11-2 Succinate 10 mM 5.5 LA_09_11-3 Succinate 10 mM 5.0 LA_09_12-1 Histidine 10 mM 6.5 LA_09_12-2 Histidine 10 mM 6.0 LA_09_12-3 Histidine 10 mM 5.5 LA_09_13-1 Glycine 10 mM 8.0 LA_09_13-2 Glycine 10 mM 7.0 LA_09_14-1 Arginine 10 mM 8.0 LA_09_14-2 Arginine 10 mM 6.0 LA_09_15-1 TRIS 10 mM 8.5 LA_09_15-2 TRIS 10 mM 7.5 LA_09_16 Citrate 10 mM 6.0 LA_09_16_1 Citrate 10 mM/PS20 6.0 LA_09_16_2 Citrate 10 mM/PS80 6.0 LA_09_16_3 Citrate 10 mM/LutrolF68 6.0 LA_09_16_4 Citrate 10 mM/Cremophor 6.0 LA_09_16_5 Citrate 10 mM/SolutolHS15 6.0 LA_09_16_6 Citrate 10 mM/SDS 6.0 LA_09_17 Acetate 10 mM 5.5 LA_09_17_1 Acetate 10 mM + PS20 5.5 LA_09_17_2 Acetate 10 mM + PS80 5.5 LA_09_17_3 Acetate 10 mM + Lutrol F68 5.5 LA_09_17_4 Acetate 10 mM + Cremophor R40 5.5 LA_09_17_5 Acetate 10 mM + Solutol HS15 5.5 LA_09_17_6 Acetate 10 mM + SDS 5.5 LA_09_18 Succinate 10 mM 5.0 LA_09_18_1 Succinate 10 mM + PS20 5.0 LA_09_18_2 Succinate 10 mM + PS80 5.0 LA_09_18_3 Succinate 10 mM + Lutrol F68 5.0 LA_09_18_4 Succinate 10 mM + Cremophor 5.0 LA_09_18_5 Succinate 10 mM + Solutol HS15 5.0 LA_09_19 Histidine 10 mM 5.5 LA_09_19_1 Histidine 10 mM + PS20 5.5 LA_09_19_2 Histidine 10 mM + PS80 5.5 LA_09_19_3 Histidine 10 mM + Lutrol F68 5.5 LA_09_19_4 Histidine 10 mM + Cremophor 5.5 LA_09_19_5 Histidine 10 mM + Solutol HS15 5.5 LA_09_20 Arginine 10 mM 6.0 LA_09_20_1 Arginine 10 mM + PS20 6.0 LA_09_20_2 Arginine 10 mM + PS80 6.0 LA_09_20_3 Arginine 10 mM + Lutrol F68 6.0 LA_09_20_4 Arginine 10 mM + Cremophor 6.0 LA_09_20_5 Arginine 10 mM + Solutol HS15 6.0 LA_09_21 Histidine 10 mM + PS20 5.5 LA_09_22 PBS 155 mM 7.2 LA_09_22_1 PBS 155 mM 7.2 LA_09_22_2 PBS 155 mM + NaCl 7.2 LA_09_22_3 PBS 155 mM + MgCl.sub.2 7.2 LA_09_22_4 PBS 155 mM + CaCl.sub.2 7.2 LA_09_22_5 PBS 155 mM + Mannitol 7.2 LA_09_22_6 PBS 155 mM + Maltose 7.2 LA_09_22_7 PBS 155 mM + Trehalose 7.2 LA_09_22_8 PBS 155 mM + Sucrose 7.2 LA_09_22_9 PBS 155 mM + Dextran40 7.2 LA_09_22_10 PBS 155 mM + Benzyl alcohol 7.2 LA_09_22_11 PBS 155 mM + Arginine 7.2 LA_09_22_12 PBS 155 mM + Lysine 7.2 LA_09_23 Citrate 10 mM (=LA_09_16) 6.0 LA_09_23_1 Citrate 10 mM 6.0 LA_09_23_2 Citrate 10 mM + NaCl 6.0 LA_09_23_3 Citrate 10 mM + MgCl.sub.2 6.0 LA_09_23_4 Citrate 10 mM + Mannitol 6.0 LA_09_23_5 Citrate 10 mM + Maltose 6.0 LA_09_23_6 Citrate 10 mM + Trehalose 6.0 LA_09_23_7 Citrate 10 mM + Sucrose 6.0 LA_09_23_8 Citrate 10 mM + Benzyl alcohol 6.0 LA_09_23_9 Citrate 10 mM + Arginine 6.0 LA_09_23_10 Citrate 10 mM + Lysine 6.0 LA_09_24 Acetate 10 mM (=LA_09_17) 5.5 LA_09_24_1 Acetate 10 mM 5.5 LA_09_24_2 Acetate 10 mM + NaCl 5.5 LA_09_24_3 Acetate 10 mM + MgCl.sub.2 5.5 LA_09_24_4 Acetate 10 mM + Mannitol 5.5 LA_09_24_5 Acetate 10 mM + Maltose 5.5 LA_09_24_6 Acetate 10 mM + Trehalose 5.5 LA_09_24_7 Acetate 10 mM + Sucrose 5.5 LA_09_24_8 Acetate 10 mM + Benzyl alcohol 5.5 LA_09_24_9 Acetate 10 mM + Arginine 5.5 LA_09_24_10 Acetate 10 mM + Lysine 5.5 LA_09_25 Histidine 10 mM (=LA_09_19) 5.5 LA_09_25_1 Histidine 10 mM + NaCl 50 mM 5.5 LA_09_25_2 Histidine 10 mM + MgCl.sub.2 50 mM 5.5 LA_09_25_3 Histidine 10 mM + Mannitol 5% 5.5 LA_09_25_4 Histidine 10 mM + Maltose 10% 5.5 LA_09_26_1 Histidine 10 mM + PS20 (=LA_09_21) 5.5 LA_09_26_2 Histidine 10 mM + PS20 + NaCl 50 mM 5.5 LA_09_26_3 Histidine 10 mM + PS20 + MgC.sub.l2 50 mM 5.5 LA_09_26_4 Histidine + PS20 + 5% Mannitol 5.5 LA_09_26_5 Histidine + PS20 + 10% Maltose 5.5 LA_09_26_6 Histidine + PS20 + 6% Trehalose 5.5 LA_09_26_7 Histidine + PS20 + 5% Sucrose 5.5 LA_09_26_8 Histidine + PS20 + 9 mg Benzyl alcohol 5.5 LA_09_26_9 Histidine + PS20 + 20 mM Arginine-HCl 5.5 LA_09_26_10 Histidine + PS20 + 20 mM Lysine 5.5 LA_09_27 Citrate 10 mM + PS20 6.0 LA_09_27_A Citrate 10 mM + PS20 Prototype formulation 6.0 LA_09_27_B Citrate 10 mM + PS20 Prototype formulation 6.0 LA_09_27_C Citrate 10 mM + PS20 Prototype formulation 6.0 LA_09_27_D Citrate 10 mM + PS20 Prototype formulation 6.0 LA_09_28 Acetate 10 mM + PS20 5.5 LA_09_28_A Acetate 10 mM + PS20 Prototype formulation 5.5 LA_09_28_B Acetate 10 mM + PS20 Prototype formulation 5.5 LA_09_28_C Acetate 10 mM + PS20 Prototype formulation 5.5 LA_09_28_D Acetate 10 mM + PS20 Prototype formulation 5.5 LA_09_29 Histidine 10 mM + PS20 5.0 LA_09_29_A Histidine 10 mM + PS20 Prototype formulation 5.0 LA_09_29_B Histidine 10 mM + PS20 Prototype formulation 5.0 LA_09_29_C Histidine 10 mM + PS20 Prototype formulation 5.0 LA_09_29_D Histidine 10 mM + PS20 Prototype formulation 5.0
Example 10--Phosphate Buffer Formulation
[0381] The following will give an overview on results of the characterization of the Lead CXCR5 Antibody drug substance in phosphate buffer.
IEF
[0382] The pI (isoelectric point) of the Lead CXCR5 Antibody was theoretically calculated as 7.6, and confirmed by denaturized isoelectric focusing (pI of 7.6-8.4). See FIG. 11.
SDS-PAGE
[0383] SDS-PAGE was used to determine the molecular weight of the antibody monomer, potential aggregates, or the presence of half-molecules. FIG. 12 showed an example of an SDS-PAGE gel to compare different drug substance batches under reducing and non-reducing conditions.
ELISA
[0384] FIG. 13 shows an example of an ELISA graph to determine antigen binding activity of the Lead Antibody.
SEC
[0385] As shown in FIG. 14, size exclusion chromatography detected high molecular weight proteins (HMWP), e.g., di-/oligomers or aggregates and low molecular weight proteins (LMWPs) or degradation products. The Lead CXCR5 Antibody batch had a purity of 99% monomer content.
WCX
[0386] Weak cationic exchange chromatography for the Lead Antibody shows in FIG. 15, display charge heterogeneity. During stability studies, the arrangement of the acidic peaks changed and the percentage of basic isoforms increased. The Lead CXCR5 Antibody had a distribution of acidic/neutral/basic isoforms of 14/85/1%.
Dynamic Light Scattering
[0387] As shown in FIG. 16, DLS was used to determine the hydrodynamic diameter of the antibody monomer and potential soluble aggregates.
[0388] In conclusion, the Lead Antibody might be stable in PBS, but aggregate formation is easy to generate by shear forces or light stress.
[0389] In addition, the pH of PBS is close to the pI of the Lead CXCR5 Antibody. Therefore, the formulation should be formulated at least one pH step below the pI.
[0390] Table 25 shows 3 the results of a three month stability study for the Lead CXCR5 Antibody. The Lead Antibody was stored at different temperatures and analyzed after one and three months.
TABLE-US-00043 TABLE 25 Analytical results of a 3-month stability study of DS Temperature Spec for 5.degree. C. -20.degree. C. Method Test item release Release 1 month 3 months 1 month Appearance Color Monitoring >B9, >BY7 >B9, >BY7 >B9, >BY7 >B9, >BY7 Clarity Monitoring <I <I <I <I Identity IEF Conform 8.30-7.50* 8.31-7.60 8.31-7.51 8.30-7.63 Potency UV mg/mL Monitoring 5.13 5.26 5.18 5.14 SEC Monomer (mg/mL) Monitoring 5.34 5.04 5.13 5.02 Ag-ELISA EC50% 50-200 100 83 112 132 Purity SDS-PAGE kD values Monitoring 46.8/26.1 47.4/25.5 47.4/25.0 46.8/25.2 reduced gel pattern Monitoring does not No changes No changes No changes conform SDS-PAGE kD values Monitoring 134.6 128.0 147.5 128.4 non-reduced gel pattern Monitoring conforms No changes No changes No changes Half-molecules <5% <5% <5% <5% Western Blot gel pattern does not No changes No changes No changes reduced conform Western Blot gel pattern does not No changes No changes No changes non-reduced conform SDS-PAGE gel pattern does not No changes No changes No changes reduced silver conform SDS-PAGE gel pattern conforms No changes No changes No changes non-reduced silver SEC % Monomer .gtoreq.90 99.8 99.8 99.7 99.7 Charge WCX (acidic/neutral/ 13.9/84.9/1.2 13.2/86.2/0.6 13.1/86.1/0.8 13.1/86.1/0.9 heterogeneity basic) (%) pH pH 6.5-8.0 7.2 7.2 7.2 7.2 Temperature Spec for -20.degree. C. 25.degree. C. Method Test item release 3 months 1 month 3 months Appearance Color Monitoring >B9, >BY7 >B9, >BY7 >B9, >BY7 Clarity Monitoring <I <I <I Identity IEF Conform 8.34-7.57 8.30-7.58 8.31-7.61 Potency UV mg/mL Monitoring 5.16 5.20 5.11 SEC Monomer (mg/mL) Monitoring 5.11 5.00 5.08 Ag-ELISA EC50% 50-200 93 106 108 Purity SDS-PAGE kD values Monitoring 47.9/25.2 46.5/25.7 47.2/25.2 reduced gel pattern Monitoring No changes No changes No changes SDS-PAGE kD values Monitoring 145.9 131.3 147.1 non-reduced gel pattern Monitoring No changes No changes No changes Half-molecules <5% <5% <5% Western Blot gel pattern No changes No changes Additional bands reduced (155.0 kD, 134.4 kD) Western Blot gel pattern No changes No changes Additional bands non-reduced (117.2 kD, 33.0 kD) SDS-PAGE gel pattern No changes No changes No changes reduced silver SDS-PAGE gel pattern No changes No changes Additional band non-reduced (122.5 kD) silver SEC % Monomer .gtoreq.90 99.7 99.5 99.4 Charge WCX (acidic/neutral/ 13.3/85.5/1.2 13.0/85.8/1.3 12.6/85.2/2.2 heterogeneity basic) (%) pH pH 6.5-8.0 7.2 7.2 7.2 *Initially reported pH 8.61-7.66
[0391] The 3-month stability data with the Lead CXCR5 Antibody buffered in PBS indicated no relevant changes at +5.degree. C. and -20.degree. C. storage. After 3 months at accelerated conditions (+25.degree. C.), significant changes could be observed. Additional bands, as analyzed by SDS-PAGE and Western-Blot analysis, showed an increase of basic- and decrease of acidic-isoforms, suggesting degradation products.
Example 11--Buffer and pH Optimization
[0392] PBS pH 7.2 showed aggregation and degradation after freeze/thaw cycles and after freezing storage. Thus, it was necessary to find another buffer and a better pH range. In addition, PBS is not suitable for freezing of the solutions, as a pH shift occurs.
[0393] 30 different buffers with various pH and buffer systems were used to select the best pH range. These experiments were run in a very small scale, and analyzed intensively.
Best Buffer & pH Selection Screening--Small Scale (Yield 5 mL)
[0394] The analytical results are summarized in Table 39, Table 40, Table 41, and Table 42.
[0395] In FIG. 17 and FIG. 18, the appearance of two suitable buffer systems (acetate & histidine) after thermal stress are shown. pH 5.5 in acetate and pH 5.0 in histidine were chosen for further evaluation. By way of contrast, in FIG. 19, the appearance of an incompatible buffer system (TRIS buffer) is shown.
[0396] The following buffers were selected to test in larger UF/DF scale:
[0397] Citrate 10 mM, pH 6
[0398] Acetate 10 mM, pH 5.5
[0399] Succinate 10 mM, pH 5
[0400] Histidine 10 mM, pH 5
[0401] Arginine 10 mM, pH 6
Best Buffer & pH Selection Screening--Large Scale (Yield .about.20 g)
[0402] After the best buffers and pH could be selected, a larger quantity of Lead CXCR5 Antibody in each buffer system was prepared by using the Sartorius Vivaflow system. Each batch was analytically tested and the results are described below.
Citrate Buffer 10 mM, pH 6 (LA_09_016)
[0403] The UF/DF step worked well and only a slightly turbid solution was obtained; no difficulties during sterile filtration were encountered. No increase of hydrodynamic diameter, as analyzed by DLS, was seen.
[0404] The analytical results indicated no increase in dimers, and no changes in basic or acidic isoforms compared to the Lead CXCR5 Antibody batch material. See Table 26 and FIG. 20.
TABLE-US-00044 TABLE 26 Analytical results of Lead Antibody in citrate buffer pH 6 Sample pH Appearance Conc. UV DLS Yield Tm LA_09_016 6.0 Slightly turbid after UF/DF, 18.2 mg/mL 12.73 nm 20.8 g 79.4.degree. C. Clear after filtration
Acetate buffer 10 mM, pH 5.5 (LA_09_017)
[0405] The UF/DF step worked well, but a turbid solution was obtained; filter blockage during sterile filtration.
[0406] The analytical results indicated no increase in dimers, and no changes in basic or acidic isoforms compared to the Lead CXCR5 Antibody batch material. See Table 27 and FIG. 21.
TABLE-US-00045 TABLE 27 Analytical results of Lead Antibody in acetate buffer pH 5.5 Sample pH Appearance Conc. UV DLS Yield Tm LA_09_017 5.5 Slightly turbid after UF/DF, 17.8 mg/mL 12.22 nm 20.4 g 77.7.degree. C. Clear after filtration
Succinate Buffer 10 mM, pH 5 (LA_09_018)
[0407] The sterile filtration after UF/DF was difficult to perform because of filter blockage. The yield of 12 g was very low.
[0408] The analytical results indicated a slight decrease in dimers, and no changes in basic or acidic isoforms compared to the Lead CXCR5 Antibody batch material. After mechanical stress, the dimer concentration increased slightly, and the acidic isoforms peak in WCX decreased as the basic isoforms increased. See Table 28 and FIG. 22.
TABLE-US-00046 TABLE 28 Analytical results of Lead Antibody in succinate buffer pH 5 Sample pH Appearance Conc. UV Yield DLS Tm LA_09_018 4.9 Slightly turbid after UF/DF, 22.4 mg/mL 12 g 12.82 nm 73.3.degree. C. Clear after filtration
Histidine Buffer 10 mM, pH 5 (LA_09_019)
[0409] The sterile filtration after UF/DF was very difficult to perform because of filter blockage. The yield of 10.5 g was very low.
[0410] The analytical results indicated a slight decrease in dimers, and no changes in basic or acidic isoforms compared to the Lead CXCR5 Antibody batch material. After mechanical stress, the dimer concentration increased slightly and the acidic isoforms peak 4_7, decreased as the basic isoforms increased. See Table 29 and FIG. 23.
TABLE-US-00047 TABLE 29 Analytical results of Lead Antibody in histidine buffer pH 5 Sample pH Appearance Conc. UV Yield DLS Tm LA_09_019 5.4 Slightly turbid after UF/DF, 23.4 mg/mL 10.5 g 11.32 nm nd Clear after filtration
Arginine Buffer 10 mM, pH 6 (LA_09_020)
[0411] The sterile filtration after UF/DF was very difficult to perform. DLS showed a brought peak with a hydrodynamic diameter of 21.08 nm, which might indicate dimer formation.
[0412] The analytical results indicated a slight increase in dimers from 0.29% in the Lead CXCR5 Antibody batch to 0.49% in arginine. After mechanical stress, 0.61% dimers were found and an increase in basic isoforms in WCX was detected. See Table 30 and FIG. 24.
TABLE-US-00048 TABLE 30 Analytical results of Lead Antibody in arginine buffer pH 5 Sample pH Appearance Conc. UV Yield DLS Tm LA_09_020 6.2 Slightly turbid after UF/DF, 22.5 mg/mL 15.3 g 21.08 nm nd Clear after filtration
[0413] In conclusion, three of the five batches are compatible with Lead CXCR5 Antibody in 20 mg/ml concentration:
[0414] Citrate pH 6.0
[0415] Acetate pH 5.5
[0416] Histidine pH 5.0
These batches were characterized in terms compatibility and stability more in detail.
Example 12--Compatibility with Excipients
[0417] All the above mentioned batches were used for compatibility studies with surfactants. Compatibility studies were performed with Lead CXCR5 Antibody and four selected buffers. Succinate pH 5.0 and arginine pH 6.0 were not tested with excipients anymore, as these buffers were not compatible with the Lead CXCR5 Antibody. Excipients were classified as follows:
[0418] Surfactants
[0419] Sugars
[0420] Salts
[0421] Others (amino acids, preservative)
Mechanical stress (agitator speed 350/min, 2.5 h, room temperature) was applied to test the effect of surfactants, and thermal stress (+40.degree. C., one week) was used to test all other excipients.
Surfactants
[0422] Orientating studies on selection of type of surfactants (LA_08_001) and surfactant concentration (LA_09_003; 0.01%, 0.05%, and 0.1%) indicated that a concentration of 0.01% was sufficient to prevent visible aggregates. The following surfactants were not suitable for the Lead CXCR5 Antibody: PVP K12 and K17, as both showed turbidity before mechanical stress was applied. Additionally, it was shown that ionic surfactants such as sodium dodecyl sulfate were not compatible with Lead CXCR5 Antibody protein solutions.
[0423] As an example, FIG. 25 shows the appearance of different citrate buffered solution with various surfactants after mechanical stress, and in comparison to a solution without any surfactant. Analytical results are collected in Table 43 and Table 44.
Other Excipients
[0424] After thermal stress of +40.degree. C. for one week and analytical determination, a selection of compatible excipients with Lead CXCR5 Antibody in different buffer systems could be given.
[0425] Some excipients could not be tested in all four buffer systems, as there was only little sample volume available.
[0426] After reviewing all analytical data, the excipients in Table 31 were identified to be compatible with the Lead CXCR5 Antibody. These excipients did not show a significant increase in dimers, HMWPs or basic isoforms analyzed by SEC and WCX.
[0427] All hydrodynamic diameter measurements were indicating a sharp monomer peak and the Tm of the suitable excipients was not decreasing compared to Lead CXCR5 Antibody in the respective buffer system. All analytical data were summarized in Table 45, Table 46, Table 47 and Table 48.
TABLE-US-00049 TABLE 31 Compatibility of all tested excipients in the different buffer systems PBS Citrate Acetate Histidine pH 7.2 pH 6.0 pH 5.5 pH 5.0 NaCl X X X MgCl.sub.2 X X X X CaCl.sub.2 X Nd Nd Nd Mannitol X X X Maltose Trehalose X X Nd Sucrose X X X Nd Dextran Nd Nd Nd Benzyl alcohol Nd Arginine-HCl X X X Nd Lysine X Nd
[0428] In conclusion, compatibility studies with surfactants show clearly that polysorbate 20 is suited for all selected buffers in combination with the Lead CXCR5 Antibody at 20 mg/mL. The surfactant prevents particle formation during mechanical stress. Nearly all other surfactants led to an increase of HMWPs.
[0429] The following excipients were selected to formulate the different prototype formulations: NaCl, Trehalose (sucrose is more or less comparable to trehalose in terms of stability), and Arginine-HCl.
3-Months Prototype Stability Study
[0430] To support the formulation development of the Lead CXCR5 Antibody, twelve different prototype formulations were manufactured and put on stability at different conditions (-20.degree. C., +5.degree. C. and +40.degree. C.) for three months.
[0431] Three different buffer systems were selected based on the before described buffer, pH and excipients screening.
[0432] Citrate pH 6.0 (formulation number LA_09_027), acetate pH 5.5 (LA_09_028), and histidine pH 5.0 (LA_09_029) were used as 10 mM buffer solutions with 20 mg/mL Lead Antibody and four different excipient combinations (Table 32).
[0433] These four excipients showed promising results after the excipient screening. NaCl was selected to adjust the osmolarity, trehalose was chosen for tonicity adjustment and to have a sugar for a lyophilization option, if needed. Additionally trehalose can stabilize the antibody, and arginine-HCl was selected as a stabilizer as well. Polysorbate 20 was found to be helpful to prevent aggregation during mechanical stress.
[0434] The following paragraphs show selected data that were compiled during the stability study to select the best buffer system and the best excipients for formulation development. In Table 33 all storage conditions, time points and analytical methods were collected.
TABLE-US-00050 TABLE 32 Compositions of four different formulation options Formulation NaCl .alpha.,.alpha.-Trehalose* H.sub.2O Arginine Polysorbate 20 A 3 mg 25 mg 20 mM 10 mg B 50 mg 10 mg C 6 mg 20 mM 10 mg D 50 mg 20 mM 10 mg
TABLE-US-00051 TABLE 33 Storage conditions and time points for the analytical testing Storage conditions T0 T 21 days T 6 weeks T 3 months SEC, WCX, SDS- PAGE, ELISA, Tm, pH, DLS, UV, Appearance, HIAC, Osmolarity -80.degree. C. SEC, WCX, SDS- PAGE*, Tm, pH, DLS, UV -20.degree. C. SEC, WCX, SDS- SEC, WCX, SDS- SEC, WCX, SDS- PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, UV UV UV, Appearance +5.degree. C. SEC, WCX, SDS- SEC, WCX, SDS- SEC, WCX, SDS- PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, UV, HIAC UV UV, Appearance +40.degree. C. SEC, WCX, SDS- SEC, WCX, SDS- SEC, WCX, SDS- PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, PAGE*, Tm, pH, DLS, UV UV UV *if reasonable
Unfortunately, the 3 month stability data of the Lead CXCR5 Antibody in PBS buffer was not comparable to the prototype stability due to batch differences and due to different accelerated conditions.
Size Exclusion Chromatography (SEC)
[0435] In FIG. 26, an increase of dimer formation up to 10% after three months of storage in all four histidine formulations can be clearly seen. Acetate formulations showed an increase of dimer content up to 6%. In all four citrate formulations, the dimer concentration was below 2%, even after three months at +40.degree. C.
Weak Cationic Exchange Chromatography (WCX)
[0436] As the determination of neutral, basic, and acidic isoforms is a good indicator for the stability of different formulations, these methods were used to amend the SEC data.
[0437] In FIG. 27 it can be seen again that histidine is worse for the Lead CXCR5 Antibody stability under accelerated conditions. A slight increase of basic isoforms can be noticed for all four acetate formulations, but interestingly for citrate formulations, discrimination between the four formulations is not possible here. In addition, FIG. 28 shows a strong decrease in neutral isoforms for the histidine formulations, and a slight decrease in acetate. Again, the Lead CXCR5 Antibody in citrate is affected the least.
SDS-PAGE
[0438] The results of SDS-PAGE measurements can be found in the result tables in the appendix. See Tables 36-60.
Unfolding Temperature (Tm)
[0439] The unfolding temperature can be used to predict the stability of different formulations and was measured here with the Microcal equipment. The higher the Tm, the more promising the formulations were. Precision of the Tm measurements were +/-0.4.degree. C.
[0440] Between citrate and acetate formulations, nearly no differences between Tm at T0 were noticed. In addition, formulations A, B, and D did have a slightly higher Tm, compared to C. The formulations A, B, and D all contain trehalose.
[0441] Histidine formulations did have a significantly lower Tm in all cases.
TABLE-US-00052 TABLE 34 Unfolding temperatures at T.sub.0 Formulation LA_09_027 LA_09_028 LA_09_29 A 81.4.degree. C. 81.1.degree. C. 79.4.degree. C. B 81.5.degree. C. 81.6.degree. C. 81.0.degree. C. C 80.7.degree. C. 80.5.degree. C. 78.9.degree. C. D 81.6.degree. C. 81.7.degree. C. 80.7.degree. C.
pH
[0442] As the pH is of major interest and importance for the stability of an antibody solution, the pH was monitored. The following figures show the delta pH between T0, T1, T2, and T3 at accelerated storage conditions.
[0443] The most pH stabilizing formulations are the citrate buffered, and especially formulations B and D (FIG. 29). In acetate buffered solutions of Lead CXCR5 Antibody, the pH was shifted towards higher values (FIG. 30). In histidine buffered solutions, the pH was slightly decreasing (FIG. 31).
DLS
[0444] The hydrodynamic diameter of the monomer and potential soluble aggregates were measured using dynamic light scattering.
[0445] Only after storage under accelerated conditions (+40.degree. C.), soluble aggregates <200 nm could be seen. These aggregates mainly occurred in histidine buffered formulation LA_09_029 A, C, D after 3 weeks of storage.
[0446] Citrate buffered formulations showed only slightly aggregates (FIG. 32) after three weeks in formulation C, and after six weeks of storage in formulation A. Some aggregates could be detected after three months in formulation B as well. But, compared to acetate buffered formulations, the amount was very little.
[0447] Acetate buffered formulation LA_09_028 C showed some aggregates <200 nm after three weeks, and after three months as well in formulation A. See FIG. 33.
UV
[0448] By monitoring the protein concentration by UV measurements, no significant differences between all time points, samples, and formulations were noticeable. As the sample volume was very little, the concentration was measured with a Nanodrop equipment. The results did vary +/-5%. For detailed information, see Tables Tables 49-60.
Appearance
[0449] After the three month storage period, all samples remained clear and colorless without any turbidity, even in histidine. This observation indicates as well, that all measured aggregates in DLS were soluble. Insoluble and sub visible aggregates could be detected by light blockage measurement by HIAC.
HIAC
[0450] Sub visible particles were detected at T0 and after three weeks of storage at +5.degree. C. The formation of particles was mainly observed in acetate buffer. Interestingly, histidine showed good results for all four different formulations. In citrate formulations A, B, and C are good as well. As the level of particles >10 .mu.m and >25 .mu.m and the values in all formulations are far below the limits defined in Ph. Eur. and USP, particle formation is of no concern.
Osmolarity
[0451] The quantification of the excipients to adjust the osmolarity were done prior the manufacturing of the samples by calculation, as no samples volume was available for orientating experiments. Therefore, the osmolarity was lower than it should be (ideally between 280 and 320 mOsmol/kg) Table 35. Further studies for better adjustment will be done during formulation optimization studies.
TABLE-US-00053 TABLE 35 Osmolarity at T0 for all prototype formulations. Formulation LA_09_027 LA_09_028 LA_09_29 A 241 221 220 B 181 160 165 C 238 220 220 D 214 192 197
Conclusion
[0452] In conclusion, citrate buffer, acetate buffer, and histidine buffer showed no changes after storage at +5.degree. C. and -20.degree. C., and only a minor increase in degradation products was seen with acetate-buffer after 3 months.
[0453] The storage of Lead CXCR5 Antibody under accelerated conditions led to significant changes of the DS. While minor changes in citrate buffer were observed, acetate buffer showed a significant increase of degradation- and aggregation products and a decrease of neutral isoforms in acidic- or basic isoforms.
[0454] A tremendous effect on the Lead CXCR5 Antibody was observed under accelerated conditions (up to 29.6% high molecular weight proteins and up to 8.2% di-/oligomer and up to 1.3% low molecular weight proteins). Also, cationic exchange chromatography revealed a decrease of the neutral isoforms to 50%.
[0455] The target concentration of 20 mg/mL could be achieved with all tested buffers, e.g. citrate, acetate, and histidine.
[0456] The pH range of a stable DP could be defined as pH 5-6.5.
[0457] Two scale-up steps (4 mL 100 mL-1000 mL UF/DF) with three selected buffers were successfully performed.
[0458] The reduction of aggregate formation with 0.01% polysorbate 20 in all selected buffers after mechanical stress (agitator speed 350/min, 2.5 h, RT) was evaluated and analytically confirmed.
[0459] The absence or decrease of HMWPs could be observed, thus increasing filterability (0.22 m) by adding 0.01% polysorbate 20 could be achieved.
[0460] The amount of dimers/oligomers was highly dependent on buffer and pH and was analyzed by using SEC, SDS-PAGE and DLS.
Characterization of Drug Substance
[0461] The Lead CXCR5 Antibody molecule is very stable in terms of degradation or half molecules formation, but it turned out during preformulation activities, that Lead CXCR5 Antibody dissolved in PBS at pH 7.2 does have an aggregation tendency. Therefore, this buffer is not suitable for long term stability. The formation of visible and sub-visible particles during storage or freeze/thaw cycles should be monitored carefully during formulation development and stability studies.
Best Buffer & pH Selection
[0462] After the best buffer and pH selection, citrate buffer 10 mM at pH 6.0 was identified to be suitable for 20 mg/mL Lead CXCR5 Antibody solutions. 10 mM histidine buffer pH 5 or 10 mM acetate buffer pH 5.5 could serve as backup options.
Compatibility Study with Excipients
[0463] The following excipients are recommended for prototype formulations:
[0464] Polysorbate 20
[0465] Trehalose/sucrose
[0466] NaCl
[0467] Arginine-HCl
[0468] The following excipients are not recommended for development:
[0469] Benzyl alcohol
[0470] Maltose
[0471] Mannitol
[0472] Dextran
[0473] Lysin-HCl
Prototype Formulation 3-Months Stability Study
[0474] Excellent stability of 20 mg/mL Lead CXCR5 Antibody in citrate buffer pH 6.0, acetate buffer pH 5.5, and histidine buffer pH 5.0 was seen at +5.degree. C. and -20.degree. C. after three months of storage. A slight degradation at +40.degree. C. (<5% reduction of monomer content) was observed with citrate buffer, while acetate buffer showed low, but significant--and histidine buffer strong artefact increases.
[0475] All tested formulations showed significant reduction of particle formation during storage compared to the generic discovery formulation in PBS pH 7.2.
[0476] Thus, the recommendation of this preformulation study is to use 10 mM citrate buffer pH 6 for DS and DP of Lead CXCR5 Antibody. A sterile filtered buffered solution with 20 mg/mL Lead CXCR5 Antibody, and stability increasing excipients should be feasible with a storage recommendation at +5.degree. C. in vials.
[0477] For tonicity adjustment trehalose and NaCl could be used and polysorbate 20 should be used to prevent the formation of aggregates.
[0478] The feasibility of UF/DF experiments to either change the buffer system and/or to increase the mAB concentration from 5 mg/mL to 20 mg/mL could be shown in different scales.
TABLE-US-00054 TABLE 36 Explanation of data assessment Lead CXCR5 Antibody Preformulation Data Assessment Process Assessment Processability Duration T0 in principle small scale small scale Appearance pH Ranking DLS clearity particle personal assessment good good good clear no ok 0 no easy to handle, <10 h @ + 5.degree. C. no turbidity no visible if pH differes <0.3 not acceptable aggregates viscosity ok for 4 mL observed particles from basic value for further in row C studies medium clear-turbid 0.5 no subvisible no clear not totally clear, aggregates assessment if acceptable or observed in applicable not DLS turbid measurements bad bad bad strong yes not ok 1 aggregates highly viscous, >10 h @ + 5.degree. C. turbidity visible if pH differes >0.31 buffer can be subvisible difficult to handle for 4 mL observed particles from basic value recommended particles observed in row C for further observed in studies DSL (in 0.22 .mu.m filtered sample) Assessment T0 after 1 week at +40.degree. C. in principle UV Appearance pH Ranking DLS UV clearity particle personal assessment good basic value clear no ok 0 no ok measured at no turbidity no visible if pH differes <0.3 not acceptable aggregates if value after T0 observed particles from basic value for further thermal stress in row C studies differs from basic value in row K < 10% medium clear-turbid 0.5 no subvisible no clear not totally clear, aggregates assessment if acceptable or observed in applicable not DLS measurements bad turbid yes not ok 1 aggregates not ok strong visible if pH differes >0.31 buffer can be subvisible if value after turbidity particles from basic value recommended particles thermal stress observed observed in row C for further observed in differs from studies DSL (in basic value in 0.22 .mu.m row K < 10% filtered sample)
TABLE-US-00055 TABLE 37 Results of preliminary packaging material testing (data assessment) PSD DLS UV ASD Buffer Formulation number Packaging material Stress pH [nm] [mg/mL] Monomer [%] PBS 155 mM LA_09_004_1 Clear glas 24 h + 40.degree. C. 7.1 11.4 4.8 99.793 Standard: RSN0151 type I 99.780 LA_09_004_2 Amber glass 24 h + 40.degree. C. 7.1 10.5 4.6 99.793 Standard: RSN0151 type II 99.780 LA_09_004_3 Polyethylen-high 24 h + 40.degree. C. 7.1 11.1 5.3 99.767 Standard: RSN0151 density 99.780 LA_09_004_4 Polyethylen-Low 24 h + 40.degree. C. 7.1 11.0 4.4 99.722 Standard: RSN0151 density 99.780 LA_09_004_5 Polypropylen 24 h + 40.degree. C. 7.1 11.2 5.1 99.781 Standard: RSN0151 99.780 ASD SEC Elisa Buffer Formulation number Dimer/Oligomer [%] EC 50% EC50 slope PBS 155 mM LA_09_004_1 0.207 131.1 5.18E-13 0.98 1.12 Standard: RSN0151 0.22 100 3.95E-13 1.18 1.12 LA_09_004_2 0.207 90.1 3.56E-13 1.16 1.12 Standard: RSN0151 0.22 100 3.95E-13 1.08 1.12 LA_09_004_3 0.233 157 6.20E-13 1.06 1 Standard: RSN0151 0.22 100 3.95E-13 LA_09_004_4 0.278 104.3 4.12E-13 Standard: RSN0151 0.22 100 3.95E-13 LA_09_004_5 0.219 106.9 5.11E-13 Standard: RSN0151 0.22 100 4.78E-13
TABLE-US-00056 TABLE 38 Results of preliminary various stress tests (data assessment) PSD DLS UV Buffer Formulation number Stress Temperature Appearance pH [nm] [mg/mL] PBS 155 mM LA_09_004_6 Clear glas type I + 24 h + 40.degree. C. Clear 7.1 11.9 4.9 Standard: RSN0151 V2A piece LA_09_004_7 Clear glas type I + 24 h + 40.degree. C. Turbid 7.1 11.2 4.6 Standard: RSN0151 1 h purged with N2 LA_09_004_8 Clear glas type I + 24 h + 40.degree. C. Turbid 7.0 11.0 4.4 Standard: RSN0151 1 h purged with air LA_09_004_9 Clear glas type I + 24 h + 40.degree. C. Turbid 7.1 10.1 4.7 Standard: RSN0151 light stress LA_09_004_10 Clear glass type I as T0 24 h + 40.degree. C. Clear 7.1 11.1 4.3 Standard: RSN0151 ASD SEC Elisa Buffer Formulation number Monomer [%] Dimer/Oligomer [%] EC 50% EC50 slope PBS 155 mM LA_09_004_6 99.551 0.391 80.8 3.86E-13 1.15 Standard: RSN0151 99.780 0.22 100 4.78E-13 1 LA_09_004_7 99.727 0.273 89.3 4.27E-13 1.04 Standard: RSN0151 99.780 0.22 100 4.78E-13 1 LA 09 004 8 99.791 0.265 84.3 4.03E-13 1.05 Standard: RSN0151 99.780 0.22 100 4.78E-13 1 LA 09 004 9 99.662 0.338 72.3 3.55E-13 1.06 Standard: RSN0151 99.780 0.22 100 4.91E-13 1.15 LA_09_004_10 99.773 0.227 62.3 3.06E-13 1.19 Standard: RSN0151 99.780 0.22 100 4.91E-13 1.15
TABLE-US-00057 TABLE 39 Results of small scale buffer selection (data assessment) Lead CXCR5 Antibody Preformulation Data Assessment Process T0 Formulation Processability Duration small Apperance number Buffer pH small scale scale clearity particle pH Ranking LA_09_05-1 PBS 155 mM 7.50 good good clear-turbid yes 7.5 0 LA_09_05-2 PBS 155 mM 7.00 good good clear-turbid yes 7 0.5 LA_09_05-3 PBS 155 mM 6.50 good good clear-turbid yes 6.6 1 LA_09_06-1 PB 5 mM 7.50 bad bad turbid yes 7.4 0 LA_09_06-2 PB 5 mM 7.00 bad bad turbid yes 7 0 LA_09_06-3 PB 5 mM 6.50 bad bad turbid yes 6.6 0 LA_09_07-1 PB 10 mM 7.50 bad bad turbid yes 7.5 0 LA_09_07-2 PB 10 mM 7.00 bad bad turbid yes 7 0 LA_09_07-3 PB 10 mM 6.50 bad bad turbid yes 6.5 0 LA_09_08-1 Citrate 10 mM 7.00 good good clear no 7 1 LA_09_08-2 Citrate 10 mM 6.50 good good clear no 6.5 1 LA_09_08-3 Citrate 10 mM 6.00 good good clear no 6 1 LA_09_08-4 Citrate 10 mM 5.50 good good clear no 5.5 1 LA_09_08-5 Citrate 10 mM 5.00 good good clear no 5 1 LA_09_09-1 Saline 150 mM 6.00 good good clear-turbid yes 7 0 LA_09_10-1 Acetate 10 mM 5.50 good good clear yes 5.8 0.5 LA_09_10-2 Acetate 10 mM 5.00 good good clear-turbid yes 5.2 0 LA_09_11-1 Succinate 10 mM 6.00 bad good clear-turbid yes 6.1 1 LA_09_11-2 Succinate 10 mM 5.50 good good clear-turbid yes 5.7 0.5 LA_09_11-3 Succinate 10 mM 5.00 good good clear-turbid yes 5.1 0 LA_09_12-1 Histidine 10 mM 6.5 good good clear-turbid yes 6.6 0.5 LA_09_12-2 Histidine 10 mM 6 good good clear-turbid no 6.1 1 LA_09_12-3 Histidine 10 mM 5.5 good good clear-turbid yes 5.1 0 LA_09_13-1 Glycine 10 mM 8 bad bad turbid yes 0 0 LA_09_13-2 Glycine 10 mM 7 good good clear-turbid yes 6.91 0 LA_09_14-1 Arginine 10 mM 8 bad bad turbid yes 7.7 0 LA_09_14-2 Arginine 10 mM 6 good good clear yes 6.4 0.5 LA_09_15-1 TRIS 10 mM 8.5 bad bad turbid yes 8.4 0 LA_09_15-2 TRIS 10 mM 7.5 bad bad turbid yes 7.5 0 after 1 week at +40.degree. C. Formulation T0 Apperance number DLS UV clearity particle pH Ranking DLS UV LA_09_05-1 no aggregate 23.39 clear no 7.39 1 no aggregate 18 LA_09_05-2 no aggregate 22.63 clear yes 6.99 0 no aggregate 21.71 LA_09_05-3 no aggregate 22.73 clear yes 6.64 0.5 no aggregate 23.77 LA_09_06-1 no aggregate 13.44 turbid yes 7.45 0 no aggregate 12.72 LA_09_06-2 no aggregate 18.34 turbid no 7 0.5 no aggregate 16.94 LA_09_06-3 no aggregate 20.75 turbid no 6.5 0.5 no aggregate 20.4 LA_09_07-1 no aggregate 18.12 clear no 7.49 1 no aggregate 16.24 LA_09_07-2 no aggregate 16.97 clear-turbid no 7 0 no aggregate 17.42 LA_09_07-3 no aggregate 20.73 clear-turbid yes 6.5 0 no aggregate 18.88 LA_09_08-1 no aggregate 19.69 clear no 7.4 1 no aggregate 23 LA_09_08-2 no aggregate 23.77 clear no 6.8 1 no aggregate 24.55 LA_09_08-3 no aggregate 21.93 clear no 6.1 1 no aggregate 22.56 LA_09_08-4 no aggregate 23.67 clear no 5.6 1 no aggregate 24.6 LA_09_08-5 no aggregate 22.97 clear no 5.1 1 aggregate 23.42 LA_09_09-1 no aggregate 24.13 clear no 6.8 1 no aggregate 22.48 LA_09_10-1 no aggregate 24.86 clear no 5.8 1 no aggregate 24.32 LA_09_10-2 no aggregate 24.99 clear yes 5.2 0.5 no aggregate 22.45 LA_09_11-1 no aggregate 23.99 clear no 6.2 0.5 no aggregate 26.13 LA_09_11-2 no aggregate 24.54 clear no 5.6 0.5 no aggregate 23.33 LA_09_11-3 no aggregate 24.22 clear no 5.2 1 no aggregate 26.1 LA_09_12-1 no aggregate 19.75 clear-turbid yes 6.57 1 no aggregate 19.38 LA_09_12-2 no aggregate 20.95 clear-turbid no 6.09 0.5 no aggregate 20.74 LA_09_12-3 no aggregate 21.37 clear-turbid no 5.58 0 no aggregate 21.98 LA_09_13-1 aggregate 0 turbid yes 0 0 aggregate 0 LA_09_13-2 aggregate 21.19 clear-turbid yes 7.09 0 aggregate 20.4 LA_09_14-1 no aggregate 10.06 turbid yes 7.6 0 no aggregate 9.6 LA_09_14-2 no aggregate 21.17 turbid yes 6.4 0.5 no aggregate 21.5 LA_09_15-1 aggregate 6.37 clear no 8.5 0.5 no aggregate 6.12 LA_09_15-2 aggregate 14.28 turbid yes 7.5 0 no aggregate 13.26
TABLE-US-00058 TABLE 40 Results of small scale buffer selection, T0 (data assessment) Lead CXCR5 Antibody Preformulation Data Assessment T0 SEC SDS-PAGE SDS-PAGE Mono- WCX red. non-red. Formulation mer Dimer/Oligo- Elisa % % neu- % HC LC main number Buffer pH [%] mer [%] EC50% EC50 slope acic tral basic [kDa] [kDa] band LA 09 05-1 PBS 155 mM 7.50 99.543 0.457 81 4.00E-13 0.96 13.4 85.6 1.1 49.81 25.79 141.88 LA_09_05-2 PBS 155 mM 7.00 99.548 0.452 74 3.82E-13 1.01 13.6 85.4 1 50.81 26.37 136.83 LA_09_05-3 PBS 155 mM 6.50 99.582 0.418 139 1.43E-12 0.72 13.3 85.6 1.1 50.91 26.87 136.69 LA 09 06-1 PB 5 mM 7.50 99.437 0.563 176 1.81E-12 0.78 12 86.5 1.5 47.62 25.94 161.36 LA_09_06-2 PB 5 mM 7.00 99.194 0.806 68 9.48E-13 0.76 14 84.6 1.4 48.06 25.19 168.27 LA_09_06-3 PB 5 mM 6.50 99.286 0.714 109.63 8.54E-13 1.12 14.2 84.4 1.3 49.79 26.56 157.05 LA 09 07-1 PB 10 mM 7.50 98.876 1.124 117 1.44E-12 0.81 14.2 83.7 2.1 52.1 25.31 148.91 LA_09_07-2 PB 10 mM 7.00 98.870 1.130 144.66 1.49E-12 0.87 13.9 84.2 1.9 50.16 25.81 140.47 LA_09_07-3 PB 10 mM 6.50 99.342 0.658 105 1.29E-12 0.73 13.9 85.1 1 50.34 25.61 131.66 LA 09 08-1 Citrate 10 mM 7.00 99.394 0.606 59.85 7.90E-13 0.90 12.6 86.0 1.1 49.5 26.1 157.3 LA_09_08-2 Citrate 10 mM 6.50 99.596 0.404 59 1.26E-12 0.53 12.7 85.8 1.4 49.7 26.4 147.0 LA_09_08-3 Citrate 10 mM 6.00 99.661 0.339 52 1.10E-12 0.50 12.7 86.0 1.2 50.9 26.8 144.5 LA_09_08-4 Citrate 10 mM 5.50 99.766 0.234 112 8.83E-13 1.01 12.8 86.1 1.1 49.1 26.0 147.9 LA_09_08-5 Citrate 10 mM 5.00 99.830 0.17 79 1.46E-12 0.66 12.7 86.1 1.2 49.77 26.18 143.43 LA_09_09-1 Saline 150 mM 6.00 99.232 0.768 96.18 1.51E-12 0.71 13.4 84.9 1.6 50 26.26 128.56 LA 09 10-1 Acetate 10 mM 5.50 99.507 0.493 180.25 2.83E-12 0.55 13.5 85.2 1.3 49.46 25.31 126.83 LA_09_10-2 Acetate 10 mM 5.00 99.622 0.378 146.86 9.81E-13 0.77 13.4 85.2 1.4 50.35 26.61 127.54 LA_09_11-1 Succinate 10 mM 6.00 99.447 0.553 112.43 7.51E-13 0.81 13.0 85.7 1.3 49.45 25.17 165.19 LA_09_11-2 Succinate 10 mM 5.50 99.582 0.418 13.0 85.8 1.3 50.3 25.44 155.06 LA_09_11-3 Succinate 10 mM 5.00 99.703 0.297 124 1.81E-12 0.72 12.9 85.8 1.2 50.15 25.51 150.47 LA 09 12-1 Histidine 10 mM 6.5 99.547 0.453 110 1.60E-12 0.60 13.3 86 0.7 47.7 24.5 157.44 LA_09_12-2 Histidine 10 mM 6 99.630 0.37 184 2.68E-12 0.55 13.1 86.2 0.6 47.77 24.7 157.86 LA_09_12-3 Histidine 10 mM 5.5 99.683 0.317 57 1.71E-12 0.71 13.2 86.1 0.7 47.41 25 156.38 LA 09 13-1 Glycine 10 mM 8 n/a LA_09_13-2 Glycine 10 mM 7 99.176 0.824 127 3.81E-12 0.57 13.6 85.5 0.8 48.74 25.04 163.07 LA 09 14-1 Arginine 10 mM 8 99.705 0.295 121 2.10E-12 0.71 15.9 83.6 0.5 49 25.22 157.49 LA_09_14-2 Arginine 10 mM 6 99.562 0.438 114 1.97E-12 0.66 13.1 86.3 0.6 49.43 25.96 147.5 LA 09 15-1 TRIS 10 mM 8.5 99.226 0.774 118 2.05E-12 0.71 25.5 74.1 0.4 50.06 25.8 147.5 LA_09_15-2 TRIS 10 mM 7.5 99.294 0.706 104 1.64E-12 0.71 13 86.1 0.8 48.75 25.41 159.79 Buffers selected for larger scale testing
TABLE-US-00059 TABLE 41 Results of small scale buffer selection, T one week +40.degree. C. (ASD data assessment) Lead CXCR5 Antibody Preformulation Data Assessment after 1 week at +40.degree. C. SEC Formulation Monomer Dimer/Oligomer HMW Elisa number Buffer pH [%] [%] [%] EC50% EC50 slope LA_09_05-1 PBS 155 mM 7.50 98.897 1.103 52 2.67E-13 1.11 LA_09_05-2 PBS 155 mM 7.00 99.279 0.721 97 9.96E-13 0.85 LA_09_05-3 PBS 155 mM 6.50 99.233 0.767 115 1.18E-12 0.84 LA_09_06-1 PB 5 mM 7.50 98.770 1.23 141 1.96E-12 0.88 LA_09_06-2 PB 5 mM 7.00 98.673 1.327 68 9.49E-13 0.81 LA_09_06-3 PB 5 mM 6.50 98.644 1.356 77 1.07E-12 0.75 LA_09_07-1 PB 10 mM 7.50 98.307 1.679 0.014 68 8.40E-13 0.81 LA_09_07-2 PB 10 mM 7.00 98.612 1.388 109 1.34E-12 0.68 LA_09_07-3 PB 10 mM 6.50 98.909 1.091 78.79 1.04E-12 0.78 LA_09_08-1 Citrate 10 mM 7.00 99.214 0.786 111 1.36E-12 0.72 LA_09_08-2 Citrate 10 mM 6.50 99.596 0.404 46 9.79E-13 0.60 LA_09_08-3 Citrate 10 mM 6.00 99.661 0.339 98 7.77E-13 1.01 LA 09 08-4 Citrate 10 mM 5.50 99.766 0.234 175 1.38E-12 0.84 LA_09_08-5 Citrate 10 mM 5.00 99.772 0.228 75 1.38E-12 0.82 LA_09_09-1 Saline 150 mM 6.00 98.962 1.038 80.89 1.27E-12 0.59 LA_09_10-1 Acetate 10 mM 5.50 99.106 0.894 94.9 1.49E-12 0.55 LA_09_10-2 Acetate 10 mM 5.00 99.463 0.537 101.5 6.78E-13 0.79 LA_09_11-1 Succinate 10 mM 6.00 99.145 0.855 125.90 8.41E-13 0.74 LA_09_11-2 Succinate 10 mM 5.50 99.410 0.590 LA 09 11-3 Succinate 10 mM 5.00 99.521 0.479 157 2.29E-12 0.64 LA_09_12-1 Histidine 10 mM 6.5 99.300 0.700 175 2.55E-12 0.57 LA_09_12-2 Histidine 10 mM 6 99.508 0.492 77 2.32E-12 0.65 LA_09_12-3 Histidine 10 mM 5.5 99.667 0.333 81 2.45E-12 0.64 LA_09_13-1 Glycine 10 mM 8 LA_09_13-2 Glycine 10 mM 7 98.446 1.554 65 1.95E-12 0.70 LA_09_14-1 Arginine 10 mM 8 99.247 0.753 88 1.53E-12 0.80 LA_09_14-2 Arginine 10 mM 6 99.277 0.723 99 1.72E-12 0.76 LA_09_15-1 TRIS 10 mM 8.5 98.456 1.544 237 3.72E-12 0.84 LA_09_15-2 TRIS 10 mM 7.5 98.998 1.002 184 2.89E-12 0.69 after 1 week at +40.degree. C. SDS-PAGE SDS-PAGE WCX red. non-red. Formulation % % % HC LC main number acic neutral basic [kDa] [kDa] band comment LA_09_05-1 14.8 83.5 1.7 50.25 26.07 142.27 usatzbande in: 61.02 kD LA_09_05-2 13.4 85.2 1.4 51.14 26.27 135.83 LA_09_05-3 13.3 85.3 1.4 49.79 25.9 141 Zusatzbande in: 59.27 LA_09_06-1 15.3 82.6 2 46.92 26.73 166.69 LA_09_06-2 11.7 86.1 2.2 49 26.11 159.55 LA_09_06-3 13.9 84.1 2 49.35 26.15 164.49 LA_09_07-1 15.1 82.2 2.6 50.18 25.41 137.71 LA_09_07-2 14.8 83.2 2 49.89 25.52 131.79 LA_09_07-3 14.1 84 1.9 51.63 26.2 139.92 LA_09_08-1 10.500 88.000 1.5 50.060 26.830 153.790 LA_09_08-2 12.700 85.800 1.4 49.690 26.390 146.970 LA_09_08-3 12.700 86.000 1.2 50.860 26.780 144.500 LA 09 08-4 12.5 86.1 1.4 49.23 25.9 144.79 Zusatzbande in 152 62 kDa LA_09_08-5 12.7 86 1.3 49.05 25.84 144.336 LA_09_09-1 13 85.1 1.9 49.33 25.23 127.67 LA_09_10-1 13.5 84.7 1.9 50.17 26.14 126.06 LA_09_10-2 13.4 84.8 1.8 49.86 26.18 128.96 LA_09_11-1 12.7 85.6 1.7 49.95 49.4 163.2 LA_09_11-2 12.6 85.7 1.6 49.84 25.23 161.74 LA 09 11-3 12.8 85.6 1.6 49.72 25.01 Im Vergleich zum Standard weist die LA_09_12-1 13.4 85.7 0.9 47.73 24.7 158.71 LA_09_12-2 12.6 86.9 0.5 48.03 24.9 159.02 LA_09_12-3 13.1 86.2 0.8 47.67 24.69 153.93 LA_09_13-1 LA_09_13-2 13.4 85.1 1.3 49.02 25.27 160.97 LA_09_14-1 14.1 84.6 1.3 49.13 25.52 154.37 LA_09_14-2 12.9 86.3 0.8 49.57 25.56 149.34 LA_09_15-1 23.8 73.9 2.3 49.37 25.05 150.98 LA_09_15-2 13.7 85 1.3 49.25 25.14 158.5 Buffers selected for larger scale testing
TABLE-US-00060 TABLE 42 Results of small scale buffer selection, after mechanical stress (data assessment) Lead CXCR5 Antibody Preformulation Data Assessment after mechanical stress 350 rpm, 2.5 h SEC Formulation Monomer mer/Oligomer HMW Elisa number Buffer pH [%] [ [%] EC50% EC50 slope LA 09 05-1 PBS 155 mM 7.50 99.263 0.658 0.080 71 3.64E-13 0.9 LA_09_05-2 PBS 155 mM 7.00 98.753 0.650 0.597 50 2.58E-13 1.02 LA_09_05-3 PBS 155 mM 6.50 98.923 0.527 0.551 79.3 9.12E-13 0.77 LA_09_06-1 PB 5 mM 7.50 99.577 0.423 111 1.54E-12 0.79 LA_09_06-2 PB 5 mM 7.00 99.084 0.916 110.14 8.58E-13 0.89 LA_09_06-3 PB 5 mM 6.50 99.202 0.798 150 1.23E-12 0.94 LA 09 07-1 PB 10 mM 7.50 98.909 1.091 60.85 4.99E-13 0.87 LA_09_07-2 PB 10 mM 7.00 99.053 0.947 61.36 6.32E-13 0.92 LA_09_07-3 PB 10 mM 6.50 99.272 0.728 89.39 1.18E-12 0.94 LA 09 08-1 Citrate 10 mM 7.00 99.138 0.838 0.024 100.76 1.33E-12 0.74 LA_09_08-2 Citrate 10 mM 6.50 99.517 0.468 0.015 41 8.61E-13 0.51 LA_09_08-3 Citrate 10 mM 6.00 99.608 0.334 0.058 136 1.07E-12 0.85 LA_09_08-4 Citrate 10 mM 5.50 99.317 0.338 0.345 79 1.46E-12 0.73 LA_09_08-5 Citrate 10 mM 5.00 99.299 0.268 1.433 99 1.84E-12 0.71 LA_09_09-1 Saline 150 mM 6.00 LA 09 10-1 Acetate 10 mM 5.50 LA_09_10-2 Acetate 10 mM 5.00 LA 09 11-1 Succinate 10 mM 6.00 LA_09_11-2 Succinate 10 mM 5.50 LA_09_11-3 Succinate 10 mM 5.00 LA 09 12-1 Histidine 10 mM 6.5 LA_09_12-2 Histidine 10 mM 6 LA_09_12-3 Histidine 10 mM 5.5 LA 09 13-1 Glycine 10 mM 8 LA_09_13-2 Glycine 10 mM 7 LA 09 14-1 Arginine 10 mM 8 LA_09_14-2 Arginine 10 mM 6 LA 09 15-1 TRIS 10 mM 8.5 LA_09_15-2 TRIS 10 mM 7.5 after mechanical stress 350 rpm, 2.5 h SDS-PAGE SDS-PAGE WCX red. non-red. Formulation % % % HC LC main number acic neutral basic [kDa] [kDa] band comment LA 09 05-1 10.2 88.6 1.2 51.01 26.64 144.8 LA_09_05-2 10.1 88.6 1.3 51.38 26.94 138.72 bande in 59 LA_09_05-3 11.5 87.4 1.2 50.4 26.17 135.68 LA_09_06-1 17.1 82 0.9 47.57 25.79 163.25 LA_09_06-2 11.2 87.6 1.2 47.36 24.75 166.93 LA_09_06-3 13.5 85.2 1.3 50.68 26.88 168.84 LA 09 07-1 12.9 85.3 1.8 50.47 26.26 147.83 LA_09_07-2 14.8 83.6 1.6 52.27 26.31 136.88 LA_09_07-3 13.6 85.4 1 50.05 25.62 131.53 LA 09 08-1 10.5 88.0 1.5 50.06 26.83 153.79 LA_09_08-2 10.6 88.2 1.3 50.82 26.79 143.82 LA_09_08-3 10.3 88.4 1.3 49.57 25.38 145.79 LA_09_08-4 10.1 88.5 1.4 49.29 25.88 144.93 LA_09_08-5 10.1 88.4 1.5 49.31 26.02 143.32 LA_09_09-1 LA 09 10-1 LA_09_10-2 LA 09 11-1 LA_09_11-2 LA_09_11-3 LA 09 12-1 LA_09_12-2 N/A LA_09_12-3 LA 09 13-1 LA_09_13-2 LA 09 14-1 LA_09_14-2 LA 09 15-1 LA_09_15-2 Buffers selected for larger scale testing
[0479] As there were seen no major differences after mechanical stress, samples LA_09_09 to 15 were not stressed mechanically.
TABLE-US-00061 TABLE 43 Results - Surfactant selection data assessment Lead CXCR5 Antibody Assessment - Surfactant data ASD Mechanical PSD SEC Formulation Stress 350 Tm DLS UV Appear- Mono- Dimer/Oligo- number rpm 2.5 h Surfactant pH [.degree. C.] pH [nm] [mg/mL] ance mer [% mer [%] Citrate LA 09 16 no non 79.4 6.0 12.7 18.2 Clear 99.74 0.260 10 mM 99.74 0.260 LA_09_16 yes non nd 6.1 N/A 18.1 Turbid LA_09_16-1 yes Polysorbate 20 79.1 6.1 12.2 17.1 Clear 99.72 0.280 99.74 0.260 LA_09_16-2 yes Polysorbate 80 78.6 6.1 12.5 18.6 Clear 99.71 0.290 6.0 99.74 0.260 LA_09_16-3 yes Lutrol F68 78.6 6.1 12.7 18.5 Clear 99.73 0.270 99.74 0.260 LA_09_16-4 yes Cremophor RH40 78.6 6.1 13.0 17.7 Clear 98.52 0.440 99.74 0.260 LA_09_16-5 yes Solutol HS15 78.4 6.1 12.8 19.2 Clear 99.1 0.560 99.74 0.260 LA_09_16 6 N/A SDS nd 6.1 N/A N/A Turbid Acetate LA_09_17 no non 77.7 5.5 12.2 17.8 Clear 99.74 0.260 10 mM 99.75 0.250 LA_09_17 yes non nd 5.6 13.4 13.2 Turbid LA_09_17-1 yes Polysorbate 20 77.4 5.5 12.5 17.7 Clear 99.74 0.260 99.75 0.250 LA_09_17-2 yes Polysorbate 80 76.4 5.6 12.8 17.8 Clear 99.52 0.380 5.5 99.75 0.250 LA_09_17-3 yes Lutrol F68 76.7 5.5 12.5 18.0 Clear 99.76 0.240 99.75 0.250 LA_09_17-4 yes Cremophor RH40 76.7 5.5 13.3 17.7 Clear 97.23 0.830 99.75 0.250 LA_09_17-5 yes Solutol HS15 76.4 5.5 12.9 17.7 Clear 98.54 0.450 99.75 0.250 LA_09_17_6 yes SDS nd 5.6 N/A N/A Turbid Succinate LA 09 18 no non 73.3 4.9 12.8 20.1 Clear 99.76 0.240 10 mM 99.71 0.290 LA_09_18 yes non nd 5.0 13.0 20.8 Turbid 99.7 0.300 99.71 0.290 LA_09_18-1 yes Polysorbate 20 72.9 5.0 12.6 20.4 Clear LA_09_18-2 yes Polysorbate 80 5.0 72.6 5.0 12.7 20.1 Clear LA_09_18-3 yes Lutrol F68 72.6 5.0 12.7 21.0 Clear LA_09_18-4 yes Cremophor RH40 72.4 5.0 12.9 21.1 Clear LA_09_18-5 yes Solutol HS15 nd 5.0 12.6 20.4 Clear ASD SEC Formulation HMW WCX SDS-PAGE non- number [%] % acic % neutral % basic Comment main band comment Citrate LA 09 16 12.79 85.99 1.23 153.79 10 mM 13.43 85.41 1.16 146.19 LA_09_16 N/A LA_09_16-1 12.79 85.93 1.28 151.33 13.43 85.41 1.16 146.19 LA_09_16-2 12.71 86.04 1.25 149.35 13.43 85.41 1.16 146.19 LA_09_16-3 13.13 85.6 1.27 147.77 13.43 85.41 1.16 146.19 LA_09_16-4 1.04 11.97 86.85 1.19 1. saurer P 149.68 13.43 85.41 1.16 146.19 LA_09_16-5 0.56 12.22 86.52 1.26 1. saurer P 151.82 13.43 85.41 1.16 146.19 LA_09_16 6 N/A Acetate LA_09_17 12.92 85.98 1.11 173.63 10 mM 13.42 85.43 1.14 169.47 LA_09_17 N/A LA_09_17-1 12.86 86.02 1.12 175.92 13.42 85.43 1.14 169.47 LA_09_17-2 0.1 12.76 86.03 1.21 178.34 13.42 85.43 1.14 169.47 LA_09_17-3 13 85.86 1.14 176.33 13.42 85.43 1.14 169.47 LA_09_17-4 1.95 11.51 87.18 1.31 k im saure 174.25 13.42 85.43 1.14 169.47 LA_09_17-5 1.01 12.23 86.67 1.1 k im saure 169.78 13.42 85.43 1.14 169.47 LA_09_17_6 N/A Succinate LA 09 18 13.319 85.425 1.256 159.8 10 mM 13.8 85.06 1.13 160.51 LA_09_18 12.189 86.388 1.423 rer Peak k 155.65 13.8 85.06 1.13 160.51 LA_09_18-1 LA_09_18-2 LA_09_18-3 N/A LA_09_18-4 LA_09_18-5
TABLE-US-00062 TABLE 44 Results - Surfactant selection data assessment Histidin LA_09_19 no non nd nd N/A 11.3 23.1 Clear Turbid 99.76 0.240 10 mM 99.7 0.300 LA_09_19 yes non nd nd 5.4 21.9 23.4 Clear Clear 97.98 LA_09_19-1 yes Polysorbate 20 nd 5.3 21.0 21.1 Turbid 99.7 0.300 99.3 0.320 LA_09_19-2 yes Polysorbate 80 5.0 5.3 21.0 21.0 99.7 0.300 99.53 0.350 LA_09_19-3 yes Lutrol F68 5.3 21.0 21.3 99.7 0.300 98.37 0.400 99.7 0.300 LA_09_19-4 yes Cremophor RH40 nd 5.3 N/A 20.1 Clear 97.19 0.850 99.7 0.300 LA_09_19-5 yes Solutol HS15 nd 5.3 21.5 21.2 Clear 99.14 0.330 99.7 0.300 Arginine LA 09 20 no non nd 20.2 Clear 99.51 0.490 10 mM 99.71 0.290 LA_09_20-1 yes non nd 6.2 N/A 22.5 Turbid 99.29 0.610 99.71 0.290 LA_09_20-2 yes Polysorbate 20 71.8 6.2 11.9 22.64 Clear LA_09_20-3 yes Polysorbate 80 6.0 nd 6.2 12.22 22.16 Clear LA_09_20-4 yes Lutrol F68 nd 6.2 12.34 22.79 Turbid LA_09_20-5 yes Cremophor RH40 nd 6.2 13.2 22.36 Clear LA_09_20-1 yes Solutol HS15 nd 6.2 N/A 22.04 Turbid Histidin LA_09_19 13.967 84.557 1.457 157.26 10 mM 13.662 85.263 1.173 160.51 LA_09_19 12.237 86.506 1.257 162.08 13.662 85.263 1.173 160.51 LA_09_19-1 13.756 84.86 1.384 rer Peak k 159.2 13.662 85.263 1.173 160.51 Zusatzban de LA_09_19-2 13.516 84.949 1.535 161.06 0.13 13.662 85.263 1.173 160.51 LA_09_19-3 12.182 86.561 1.257 163.05 1.23 rer Peak k Zusatzban de 13.662 85.263 1.173 160.51 LA_09_19-4 1.96 10.437 87.657 1.906 rer Peak k 168.73 Zusatzban de 13.662 85.263 1.173 160.51 LA_09_19-5 0.52 12.561 85.891 1.548 rer Peak k 160.22 13.662 85.263 1.173 160.51 Arginine LA 09 20 13.31 85.324 1.367 157.89 10 mM 13.8 85.06 1.13 155.98 LA_09_20-1 0.1 12.33 86.015 1.655 rer Peak k 152.77 Zusatzban den: 300 13.8 85.06 1.13 155.98 LA_09_20-2 LA_09_20-3 LA_09_20-4 N/A LA_09_20-5 LA_09_20-1 N/A, samples were not tested analytically, as too little sample volume was available.
Excipients and Lead CXCR5 Antibody (LA_09_022)
TABLE-US-00063 [0480] TABLE 45 Results - compatibility of Lead Antibody with excipients Lead CXCR5 Antibody Assessment - Compatibility SEC PSD pH Mono- Dimer/Oligo- Tm DLS UV Appear- mer mer Thermal pH [.degree. C.] pH [nm] [mg/mL] ance [%] [% HMW [%]] RSNO151 LA_09_22 No Non 80.3 6.0 12.7 18.2 Clear 99.686 0.314 155 mMpBS Standard: RSN0151 99.707 0.293 LA_09_22_1 Yes Non 6.1 N/A 18.1 Turbid 99.388 0.542 Standard: RSN0151 99.707 0.293 LA_09_22_2 Yes NaCl 80.3 6.1 12.2 17.1 Clear 99.539 0.4 Standard: RSN0151 99.707 0.293 LA_09_22_3 Yes MgCl2 nd 6.1 12.5 18.6 Clear 99.697 0.303 Standard: RSN0151 99.707 0.293 LA_09_22_4 Yes CaCl2 78.2 6.1 12.7 18.5 Clear 99.868 0.132 Standard: RSN0151 99.707 0.293 LA_09_22_5 Yes Mannitol nd 6.1 13.0 17.7 Clear 99.488 0.499 0.013 Standard: RSN0151 99.707 0.293 LA_09_22_6 Yes Maltose nd 6.1 12.8 19.2 Clear 98.324 1.676 Standard: RSN0151 99.707 0.293 LA_09_22_7 Yes Trehalose 80.8 6.1 N/A N/A Turbid 99.507 0.389 Standard: RSN0151 99.366 0.343 LA_09_22_8 Yes Sucrose 80.5 5.5 12.2 17.8 Clear 99.307 0.535 Standard: RSN0151 99.366 0.343 LA_09_22_9 Yes Dextran 40 79.3 5.6 13.4 13.2 Turbid 99.821 1.545 Standard: RSN0151 99.366 0.343 LA_09_22_10 Yes Benzylalkohol 75.8 5.5 12.5 17.7 Clear 97.515 1.027 Standard: RSN0151 99.366 0.343 LA_09_22_11 Yes Arginine-HCl 80.0 5.6 12.8 17.8 Clear 99.622 0.331 99.366 0.343 LA_09_22_12 Yes Lysin 80.9 5.5 12.5 18.0 Clear 99.047 0.701 99.366 0.434 WCX SDS-PAGE non-red % basic Main % Acid % neutral Comment band comment RSNO151 LA_09_22 84.306 1.373 155.76 155 mMpBS Standard: RSN0151 13.867 85.032 1.102 155.98 LA_09_22_1 13.878 84.289 1.833 142.95 Standard: RSN0151 13.867 85.032 1.102 154.13 LA_09_22_2 13.806 84.404 1.790 147.97 Standard: RSN0151 13.867 85.032 1.102 154.13 LA_09_22_3 15.353 83.235 1.412 149.38 Standard: RSN0151 13.867 85.032 1.102 151.43 LA_09_22_4 14.887 83.908 1.205 148.81 Standard: RSN0151 13.867 85.032 1.102 154.13 LA_09_22_5 15.757 82.649 1.624 150.42 Standard: RSN0151 13.867 85.032 1.102 154.13 LA_09_22_6 34.894 62.913 2.191 152.49 Standard: RSN0151 13.867 85.032 1.102 154.13 271.47 kDa Bandesta LA_09_22_7 17.291 81.157 1.552 154.74 Standard: RSN0151 14.019 84.575 1.407 154.13 LA_09_22_8 15.184 83.508 1.308 158.76 Standard: RSN0151 14.019 84.575 1.407 154.13 LA_09_22_9 20.308 77.996 1.696 166.61 Standard: RSN0151 14.019 84.575 1.407 154.13 LA_09_22_10 14.005 83.824 2.17 168.13 Standard: RSN0151 14.019 84.575 1.407 154.13 LA_09_22_11 15.262 83.116 1.621 169.77 14.019 84.575 1.407 154.13 LA_09_22_12 19.042 63.104 17.854 169.9 Adation bands main 14.019 84.575 1.407 15.413 ca/
Excipients and Citrate Buffered (LA_09_023)
TABLE-US-00064 [0481] TABLE 46 Results - compatibility of Lead Antibody in acetate buffer with excipients Lead CXCR5 Antibody Assessment - Compatibility in citrate buffer ASD SDS- SEC PAGE PSD pH Mono- Dimer/Oligo- WCX non-red Tm DLS UV mer mer % % neu- % Main Thermal Excipient pH [.degree. C.] [nm] [mg/mL] [%] [% HMW [%]] Acid tral basic band RSNO151 LA_09_23 No Non 81.5 12.6 18.6 99.596 0.404 13.307 85.310 1.383 155.59 155 mMpBS Standard: RSN0151 14.058 84.609 1.333 146.11 LA_09_23_1 Yes Non 12.4 18.7 99.491 0.509 12.807 85.597 1.596 151.85 Standard: RSNO151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_2 Yes NaCl 81.7 12.2 21.8 99.498 0.502 12.805 85.641 1.554 145.12 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_3 Yes MgCl2 77.2 12.3 14.7 99.538 0.462 12.799 85.692 1.509 151.67 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_4 Yes Mannitol 82.1 13.5 23.3 99.533 0.467 12.703 85.802 1.495 147.96 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_5 98.899 0.677 16.363 82.106 1.631 155.02 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_6 Yes Trehalose 82.1 13.9 17.6 99.656 0.344 12.693 85.843 1.465 151.43 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_7 Yes Sucrose 81.9 13.5 17.5 99.594 0.406 12.758 85.754 1.487 152.67 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_8 Yes Benzyl- 77.1 13.9 20.5 97.744 0.507 1.322 11.591 87.216 1.192 145.93 Standard: RSN0151 alkohol 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_9 Yes Arginine- 80.7 14.7 17.6 99.327 0.412 13.047 85.843 1.500 146.01 Standard: RSN0151 HCl 99.686 0.314 14.058 84.609 1.333 146.11 LA_09_23_10 Yes Lysin nd 12.0 16.7 98.849 0.969 13.642 80.767 5.591 144.05 Standard: RSN0151 99.686 0.314 14.058 84.609 1.333 146.11
Excipients and Acetate Buffered (LA_09_024)
TABLE-US-00065 [0482] TABLE 47 Results - compatibility with excipients and Lead Antibody in acetate buffer Lead CXCR5 Antibody Assessment - Compatibility in acetate buffer - data ASD PSD pH SEC Tm DLS UV Monomer Dimer/Oligomer Thermal Excipient pH [.degree. C.] [nm] [mg/mL] [%] [% HMW [%]] RSNO151 LA_09_24 No Non 81.8 12.2 99.603 0.397 155 mMpBS Standard: RSN0151 99.628 0.309 LA_09_24_1 Yes Non nd 12.5 99.405 0.595 Standard: RSN0151 99.628 0.309 LA_09_24_2 Yes NaCl 81.3 12.3 99.479 0.521 Standard: RSN0151 99.628 0.309 LA_09_24_3 Yes MgCl2 81.0 12.3 99.517 0.483 Standard: RSN0151 99.628 0.309 LA_09_24_4 Yes Mannitol 82.5 14.2 99.546 0.431 0.023 Standard: RSN0151 99.628 0.309 LA_09_24_5 Yes 98.647 0.539 0.029 Standard: RSN0151 99.628 0.309 LA_09_24_6 Yes Trehalose 82.5 14.0 99.522 0.478 Standard: RSN0151 99.628 0.309 LA_09_24_7 Yes Sucrose 82.3 14.0 99.542 0.458 Standard: RSN0151 99.628 0.309 LA_09_24_8 Yes Benzylalkohol 78.0 13.2 98.272 0.67 0.745 Standard: RSN0151 99.628 0.309 LA_09_24_9 Yes Arginine- 81.5 12.2 99.226 0.439 Standard: RSN0151 HCl 99.628 0.309 LA_09_24_10 Yes Lysin 81.2 12.3 99.134 0.692 Standard: RSN0151 99.628 0.309 ASD SDS-PAGE WCX non-red % % % basic Main Acid neutral Comment band comment RSNO151 LA_09_24 13.408 85.225 1.368 173.63 155 mMpBS Standard: RSN0151 14.230 84.407 1.363 169.47 LA_09_24_1 13.368 85.052 1.580 149.18 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_2 13.507 84.895 1.598 156.31 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_3 13.470 84.915 1.615 142.23 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_4 13.557 84.965 1.478 155.69 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_5 16.304 82.016 1.680 156.15 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_6 13.558 84.905 1.537 146.61 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_7 13.587 84.923 1.490 149.12 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_8 12.739 85.960 1.301 152.48 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_9 13.412 84.978 1.611 152.88 Standard: RSN0151 14.230 84.407 1.363 151.63 LA_09_24_10 13.546 84.814 1.636 153.32 Standard: RSN0151 14.230 84.407 1.363 151.63
Excipients and Histidine Buffered (LA_09_025)
TABLE-US-00066 [0483] TABLE 48 Results - compatibility with excipients and Lead Antibody In histidine buffer Lead CXCR5 Antibody Assessment - Compatibility in Histidine buffer - data PSD SEC Formulation Thermal Tm DL8 UV Monomer Dimer/Oligomer Buffer Number stress Excipient pH [.degree. C.] pH [nm] [mg/mL] [%] [% HMW [%]] Histidine SAR113244_09_25 (- No non Not 11.3 23.1 22.76 0.240 10 mM SAR113244_09-019) tested 22.7 0.300 Standard: RSN0151 SAR113244_09_25_1 Yes NaCl 78.8 25.8 23.8 25.784 0.5 Standard: RSN0151 22.458 0.317 SAR113244_09_25 2 Yes MgCl2 5.5 nd 12.8 21.5 22.558 0.442 Standard: RSN0151 22.458 0.317 SAR113244_09_25 3 Yes Mannitol 82.3 13.1 22.1 22.582 0.411 Standard: RSN0151 22.458 0.317 SAR113244_09_25 4 Yes Maltose 80.8 15.5 21.5 22.257 0.475 Standard: RSN0151 22.458 0.317 SDS-PAGE non-red Formulation WCX Main Buffer Number % Acid % neutral % basic band Comment Histidine SAR113244_09_25 (- 13.287 84.557 1.457 157.28 10 mM SAR113244_09-019) 13.882 85.283 1.173 150.51 Standard: RSN0151 SAR113244_09_25_1 13.687 84.223 1.320 158.38 Standard: RSN0151 14.121 84.602 1.277 154.13 SAR113244_09_25 2 13.880 84.783 1.557 153.77 Standard: RSN0151 14.121 84.602 1.277 154.13 SAR113244_09_25 3 13.704 85.008 1.282 158.28 Standard: RSN0151 14.121 84.602 1.277 154.13 SAR113244_09_25 4 16.228 82.223 1.478 155.32 Standard: RSN0151 14.121 84.602 1.277 154.13
TABLE-US-00067 TABLE 49 Results - Prototype formulation LA_09_27A ASD PSD SEC Formulation Time Storage Tm DLS UV Mono- Dimer/Oligo- HMW Buffer number point condition .degree. C. [.degree. C.] pH [nm] [mg/mL] mer [%] mer [%] [%] Citrate 10 mM. LA_09_27A T0 N/A N/A 12.2 99.712 0.217 3 mg/mL Standard: RSN0151 81.4 20.0 99.691 0.309 NaCl. LA_09_27A T: 3 weeks {grave over ( )}+5.degree. C. T: 5.8 12.2 99.670 0.309 25 mg/mL Standard: RSN0151 81.6 5.8 19.7 99.463 0.563 Trehalose. LA_09_27A 3 weeks {grave over ( )}-20.degree. C. T: 3 81.6 12.2 20.1 99.649 0.318 20 mM Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 5.8 99.463 0.563 Arginine- LA_09_27A 81.6 13.4 19.3 97.322 0.807 1.324 HCl. 0.01% 99.463 0.563 Polysorbate Standard: RSN0151 weeks {grave over ( )}+5.degree. C. T: 6 81.6 5.9 12.3 24.8 99.673 0.327 20 LA_09_27A 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}-20.degree. C. T: 6 6.0 81.4 5.9 12.2 24.9 99.670 0.33 LA_09_27A 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 81.4 5.9 13.8 + A 25.9 96.290 1.06 1.919 LA_09_27A 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.9 12.2 24.7 99.667 0.333 LA_09_27A 99.460 0.54 Standard: RSN0151 months {grave over ( )}+5.degree. C. T: 3 81.7 5.8 12.2 19.9 99.582 0.418 LA_09_27A 99.427 0.573 Standard: RSN0151 months {grave over ( )}-20.degree. C. T: 3 81.7 5.8 12.4 20.4 99.684 0.316 LA_09_27A 99.427 0.573 Standard: RSN0151 months {grave over ( )}+40.degree. C. 81.6 5.8 14.66 + A 20.3 94.276 2.005 2.599 LA_09_27A 99.427 0.573 ASD SDS-PAGE WCX non-red. Formulation SEC % % % main Elisa Buffer number NP[%] acic neutra basic Comment band comment EC50% EC50 slope Citrate 10 mM. LA_09_27A 13.988 84.86 1.151 118.94 112 2.09E+12 0.53 3 mg/mL Standard: RSN0151 13.759 85.041 1.2 122.88 100 1.86E+12 0.56 NaCl. LA_09_27A 0.021 14.143 84.561 1.297 N/A 25 mg/mL Standard: RSN0151 14.000 84.450 1.551 Trehalose. LA_09_27A 0.033 14.382 84.423 1.195 N/A 112 2.09E+12 0.53 20 mM Standard: RSN0151 14.000 84.450 1.551 100 1.86E+12 0.56 Arginine- LA_09_27A 0.547 12.133 85.819 2.048 HCl. 0.01% 14.000 84.450 1.551 Polysorbate Standard: RSN0151 13.798 85.176 1.026 20 LA_09_27A 14.054 84.285 1.662 Standard: RSN0151 13.756 85.192 1.052 LA_09_27A 14.054 84.285 1.662 Standard: RSN0151 0.731 11.826 85.289 2.886 LA_09_27A 14.054 84.285 1.662 Standard: RSN0151 13.298 85.669 1.033 N/A LA_09_27A 14.054 84.285 1.662 Standard: RSN0151 14.528 83.929 1.543 LA_09_27A 14.413 84.08 1.507 Standard: RSN0151 14.936 83.655 1.409 LA_09_27A 14.413 84.08 1.507 Standard: RSN0151 14.871 80.287 4.843 LA_09_27A 14.413 84.08 1.507
TABLE-US-00068 TABLE 50 Results - Prototype formulation LA_09_027B ASD PSD SEC Formulation Time Storage Tm DLS UV Mono- Dimer/Oligo- HMW Buffer number point condition pH [.degree. C.] pH [nm] [mg/mL] mer [%] mer [%] [%] Citrate 10 mM, LA_09_27B T0 N/A 81.5 N/A 13.6 20.9 99.688 0.312 50 mg Standard: RSN0151 99.691 0.309 Trehalose, LA_09_27B T: 3 weeks {grave over ( )}+5.degree. C. 6.1 13.6 20.5 99.321 0.384 0.01% Standard: RSN0151 99.463 0.563 Polysorbate LA_09_27B T: 3 weeks {grave over ( )}-20.degree. C. 6.0 13.6 19.7 99.626 0.374 20 Standard: RSN0151 99.463 0.563 LA_09_27B T: 3 weeks {grave over ( )}+40.degree. C. 6.0 14.4 19.9 96.851 0.934 1.7 Standard: RSN0151 99.463 0.563 LA_09_27B T: 6 weeks {grave over ( )}+5.degree. C. 6.1 13.6 25.6 99.612 0.388 Standard: RSN0151 99.460 0.54 LA_09_27B T: 6 weeks {grave over ( )}-20.degree. C. 6.1 13.6 25.0 99.628 0.372 Standard: RSN0151 6.0 N/A 99.460 0.54 LA_09_27B T: 6 weeks {grave over ( )}+40.degree. C. 6.2 14.9 24.9 95.806 1.283 2.182 Standard: RSN0151 99.460 0.54 LA_09_27B T: 6 weeks {grave over ( )}-80.degree. C. 6.1 13.7 25.0 Standard: RSN0151 99.460 0.54 LA_09_27B T: 3 months {grave over ( )}+5.degree. C. 6.1 13.7 20.8 99.636 0.349 Standard: RSN0151 99.427 0.573 LA_09_27B T: 3 months {grave over ( )}-20.degree. C. 6.1 13.6 19.9 99.621 0.379 Standard: RSN0151 99.427 0.573 LA_09_27B T: 3 months {grave over ( )}+40.degree. C. 6.0 15.56 + A 19.4 94.631 1.783 2.567 Standard: RSN0151 99.427 0.573 ASD SDS-PAGE WCX non-red. Formulation SEC % % % main Elisa Buffer number NP[%] acic neutral basic Comment band comment EC50% EC50 slope Citrate 10 mM, LA_09_27B 13.723 85.084 1.194 117.40 104 1.93E+12 0.62 50 mg Standard: RSN0151 13.759 85.041 1.2 122.88 100 1.86E+12 0.56 Trehalose, LA_09_27B 0.295 13.911 84.772 1.317 0.01% Standard: RSN0151 14.000 84.450 1.551 Polysorbate LA_09_27B 14.14 84.541 1.318 20 Standard: RSN0151 14.000 84.450 1.551 LA_09_27B 0.515 12.455 85.392 2.153 NA Standard: RSN0151 14.000 84.450 1.551 LA_09_27B 13.747 84.98 1.273 Standard: RSN0151 14.054 84.285 1.662 LA_09_27B 13.835 84.901 1.264 Standard: RSN0151 14.054 84.285 1.662 LA_09_27B 0.729 12.685 83.832 3.483 218.32 90. 28. 14 kD N/A Standard: RSN0151 14.054 84.285 1.662 189.54 LA_09_27B 13.882 84.783 1.336 Standard: RSN0151 14.054 84.285 1.662 LA_09_27B 14.63 83.798 1.571 Standard: RSN0151 14.413 84.08 1.507 LA_09_27B 15.128 83.406 1.466 Standard: RSN0151 14.413 84.08 1.507 NA LA_09_27B 15.373 79.378 5.25 Standard: RSN0151 14.413 84.08 1.507
TABLE-US-00069 TABLE 51 Results - Prototype formulation LA_09_027C ASD PSD SEC Formulation Time Storage Tm DLS UV Mono- Dimer/Oligo- HMW Buffer number point condition pH [.degree. C.] pH [nm] [mg/mL] mer [%] mer [%] [%] Citrate 10 mM, LA_09_27C T0 N/A N/A 11.6 99.688 0.312 6 mg NaCl, Standard: RSN0151 80.7 19.921.1 99.691 0.309 0.01% LA_09_27C T: 3 weeks {grave over ( )}+5.degree. C. T: 5.8 11.6 99.321 0.384 Polysorbate Standard: RSN0151 3 weeks {grave over ( )}-20.degree. C. T: 3 80.9 20.620.4 99.463 0.563 20 5.8 11.6 99.626 0.374 LA_09_27C 80.7 25.425.4 99.463 0.563 Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 5.8 12.98 + A 96.851 0.934 1.7 LA_09_27C 80.4 5.325.7 99.463 0.563 Standard: RSN0151 weeks {grave over ( )}+5.degree. C. T: 6 5.9 11.6 99.612 0.388 LA_09_27C 80.7 21.420.9 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}-20.degree. C. T: 6 6.0 5.9 11.5 99.628 0.372 LA_09_27C 80.9 21.4 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 80.7 5.9 13.77 + A 95.806 1.283 2.182 LA_09_27C 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.8 18.7 LA_09_27C 99.460 0.54 Standard: RSN0151 months {grave over ( )}+5.degree. C. T: 3 80.9 5.8 11.6 99.636 0.349 LA_09_27C 99.427 0.573 Standard: RSN0151 months {grave over ( )}-20.degree. C. T: 3 81.2 5.8 11.7 99.621 0.379 LA_09_27C 99.427 0.573 Standard: RSN0151 months {grave over ( )}+40.degree. C. 80.9 5.8 14.28 + A 94.631 1.783 2.567 LA_09_27C 99.427 0.573 ASD SDS-PAGE WCX non-red. Formulation SEC % % % main Elisa Buffer number NP[%] acic neutral basic Comment band comment EC50% EC50 slope Citrate 10 mM, LA_09_27C 13.723 85.084 1.194 117.40 104 1.93E+12 0.62 6 mg NaCl, Standard: RSN0151 13.759 85.041 1.2 122.88 100 1.86E+12 0.56 0.01% LA_09_27C 0.295 13.911 84.772 1.317 Polysorbate Standard: RSN0151 14.000 84.450 1.551 20 14.14 84.541 1.318 LA_09_27C 14.000 84.450 1.551 Standard: RSN0151 0.515 12.455 85.392 2.153 LA_09_27C 14.000 84.450 1.551 N/A Standard: RSN0151 13.747 84.98 1.273 LA_09_27C 14.054 84.285 1.662 Standard: RSN0151 13.835 84.901 1.264 LA_09_27C 14.054 84.285 1.662 Standard: RSN0151 0.729 12.685 83.832 3.483 218.32 90. 28. 14 kD N/A LA_09_27C 14.054 84.285 1.662 189.54 Standard: RSN0151 13.882 84.783 1.336 LA_09_27C 14.054 84.285 1.662 Standard: RSN0151 14.63 83.798 1.571 LA_09_27C 14.413 84.08 1.507 N/A Standard: RSN0151 15.128 83.406 1.466 LA_09_27C 14.413 84.08 1.507 Standard: RSN0151 15.373 79.378 5.25 LA_09_27C 14.413 84.08 1.507
TABLE-US-00070 TABLE 52 Results - Prototype formulation LA_09_027D ASD PSD SEC Formulation Time Storage Tm DLS UV Mono- Dimer/Oligo- HMW Buffer number point condition pH [.degree. C.] pH [nm] [mg/mL] mer [%] mer [%] [%] Citrate LA_09_27D T0 N/A N/A 99.579 0.283 10 mM, Standard: RSN0151 81.6 13.3 19.8 99.691 0.309 50 mg LA_09_27D T: 3 weeks {grave over ( )}+5.degree. C. T: 6.0 99.676 0.313 Trehalose, Standard: RSN0151 81.4 13.1 19.5 99.463 0.563 20 mM LA_09_27D 3 weeks {grave over ( )}-20.degree. C. T: 3 5.8 13.2 20.4 99.638 0.338 Arginine- Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 81.4 99.463 0.563 HCl, LA_09_27D 81.4 6.0 13.9 19.6 97.653 0.809 0.946 0.01% Standard: RSN0151 weeks {grave over ( )}+5.degree. C. T: 6 99.463 0.563 Polysorbate LA_09_27D 80.9 6.0 13.1 24.4 99.660 0.34 20 Standard: RSN0151 weeks {grave over ( )}-20.degree. C. T: 6 99.460 0.54 6.0 13.2 24.2 99.648 0.352 LA_09_27D 6.0 80.6 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 96.544 1.056 1.637 LA_09_27D 80.9 6.0 14.5 24.8 99.460 0.54 Standard: RSN0151 weeks {grave over ( )}-80.degree. C. T: 3 N/A 6.0 13.2 24.5 99.664 0.336 LA_09_27D 99.460 0.54 Standard: RSN0151 months {grave over ( )}+5.degree. C. T: 3 82.1 5.9 13.1 20.4 99.636 0.341 LA_09_27D 99.427 0.573 Standard: RSN0151 months {grave over ( )}-20.degree. C. T: 3 82.1 5.9 13.2 19.8 99.655 0.334 LA_09_27D 99.427 0.573 Standard: RSN0151 months {grave over ( )}+40.degree. C. 81.9 5.9 15.2 20.4 94.413 1.634 2.446 LA_09_27D 99.427 0.573 ASD SDS-PAGE WCX non-red. Formulation SEC % % % main Elisa Buffer number NP[%] acic neutra basic Comment band comment EC50% EC50 slope Citrate LA_09_27D 13.974 84.912 1.114 117.29 94 1.75E+12 0.61 10 mM, Standard: RSN0151 13.759 85.041 1.2 122.88 100 1.86E+12 50 mg LA_09_27D 0.011 13.825 84.918 1.256 Trehalose, Standard: RSN0151 14.000 84.450 1.551 20 mM LA_09_27D 0.024 14.028 84.745 1.227 Arginine- Standard: RSN0151 14.000 84.450 1.551 HCl, LA_09_27D 0.592 12.315 85.553 2.131 0.01% Standard: RSN0151 14.000 84.450 1.551 N/A Polysorbate LA_09_27D 13.475 85.337 1.188 20 Standard: RSN0151 14.054 84.285 1.662 13.445 85.502 1.053 LA_09_27D 14.054 84.285 1.662 Standard: RSN0151 0.763 11.916 85.099 2.985 192.29 LA_09_27D 14.054 84.285 1.662 Standard: RSN0151 13.485 85.357 1.159 LA_09_27D 14.054 84.285 1.662 Standard: RSN0151 14.09 84.529 1.38 LA_09_27D 14.413 84.08 1.507 Standard: RSN0151 14.521 84.08 1.398 LA_09_27D 14.413 84.08 1.507 189.54 0.56 Standard: RSN0151 1.12 14.173 81.003 4.823 LA_09_27D 14.413 84.08 1.507
TABLE-US-00071 TABLE 53 Results - Prototype formulation LA_09_028A ASD PSD SEC Formulation Time Storage Tm DLS UV Mono- Dimer/Oligo- HMW Buffer number point condition pH [.degree. C.] pH [nm] [mg/mL] mer [%] mer [%] [%] Acetate LA_09_28A T0 N/A N/A 99.503 0.26 10 mM, 3 mg/mL Standard: RSN0151 81.1 12.9 17.4 99.691 0.309 NaCl, 25 mg/mL LA_09_28A T: 3 weeks {grave over ( )}+5.degree. C. T: 5.6 99.650 0.35 Trehalose, Standard: RSN0151 81.1 12.6 16.1 99.481 0.519 20 mM LA_09_28A 3 weeks {grave over ( )}-20.degree. C. T: 3 81.1 5.6 12.5 15.7 99.639 0.315 Arginine- 99.481 0.519 HCl, = .01% Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 81.1 5.6 13.9 15.6 96.870 1.168 1.484 Polysorbate LA_09_28A 99.481 0.519 20 Standard: RSN0151 weeks {grave over ( )}+5.degree. C. T: 6 81.1 5.6 12.6 16.4 99.634 0.366 LA_09_28A 99.460 0.545 Standard: RSN0151 weeks {grave over ( )}-20.degree. C. T: 6 5.5 81.1 5.6 12.7 16.6 99.650 0.35 LA_09_28A 99.460 0.545 Standard: RSN0151 weeks {grave over ( )}+40.degree. C. T: 6 N/A 5.7 14.7 16.5 94.853 1.837 2.189 LA_09_28A 99.460 0.545 Standard: RSN0151 weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.6 12.8 16.6 99.628 0.372 LA_09_28A 99.460 0.545 Standard: RSN0151 months {grave over ( )}+5.degree. C. T: 3 81.1 5.7 12.5 16.6 99.564 0.415 LA_09_28A 99.421 0.579 Standard: RSN0151 months {grave over ( )}-20.degree. C. T: 3 81.3 5.7 12.7 16.4 99.594 0.388 LA_09_28A 99.421 0.579 Standard: RSN0151 months {grave over ( )}+40.degree. C. 80.7 5.7 15.86 + A 16.5 91.596 3.693 2.839 LA_09_28A 99.421 0.579 ASD SDS-PAGE WCX non-red. Formulation SEC % % % main Elisa Buffer number NP[%] acic neutra basic Comment band comment EC50% EC50 slope Acetate LA_09_28A 14.153 84.626 1.221 118.94 112 2.09E+12 0.53 10 mM, 3 mg/mL Standard: RSN0151 13.772 85.024 1.204 122.88 100 1.86E+12 0.56 NaCl, 25 mg/mL LA_09_28A 14.036 84.717 1.247 Trehalose, Standard: RSN0151 14.278 84.346 1.377 N/A 20 mM LA_09_28A 0.046 13.99 84.778 1.231 N/A 112 2.09E+12 0.53 Arginine- 14.278 84.346 1.377 100 1.86E+12 0.56 HCl, = .01% Standard: RSN0151 0.478 13.418 83.852 2.730 Polysorbate LA_09_28A 14.278 84.346 1.377 20 Standard: RSN0151 13.803 84.981 1.216 LA_09_28A 13.864 85.048 1.088 N/A Standard: RSN0151 13.844 84.952 1.205 LA_09_28A 13.864 85.048 1.088 Standard: RSN0151 1.121 13.596 82.452 3.952 195.57 66. 28. 14 kDa N/A LA_09_28A 13.864 85.048 1.088 189.54 Standard: RSN0151 LA_09_28A 13.864 85.048 1.088 Standard: RSN0151 14.363 83.628 2.009 LA_09_28A 14.618 83.409 1.973 Standard: RSN0151 14.314 83.738 1.948 N/A LA_09_28A 14.618 83.409 1.973 Standard: RSN0151 1.872 18.117 74.736 7.147 LA_09_28A 14.618 83.409 1.973
TABLE-US-00072 TABLE 54 Results - Prototype formulation LA_09_028B ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm] mL] [%] [%] [%] [%] Acetate 10 mM, LA_09_28B T0 N/A N/A 99.590 0.41 50 mg Trehalose, Standard: RSN0151 81.6 14.4 17.1 99.691 0.309 0.01% Polysorbate 20 LA_09_28B T: 3 weeks {grave over ( )}+5.degree. C. T: 5.6 99.476 0.476 0.048 Standard: RSN0151 81.6 14.2 16.3 99.481 0.519 LA_09_28B 3 weeks {grave over ( )}-20.degree. C. T: 3 5.7 99.411 0.497 0.092 Standard: RSN0151 81.6 14.2 16.2 99.481 0.519 LA_09_28B weeks {grave over ( )}+40.degree. C. T: 6 5.6 97.322 2.039 0.639 Standard: RSN0151 81.6 14.8 16.2 99.481 0.519 LA_09_28B weeks {grave over ( )}+5.degree. C. T: 6 5.7 99.510 0.49 Standard: RSN0151 81.6 14.3 16.7 99.460 0.545 LA_09_28B weeks {grave over ( )}-20.degree. C. T: 6 5.5 5.7 99.530 0.47 Standard: RSN0151 81.6 14.6 17.4 99.460 0.545 LA_09_28B weeks {grave over ( )}+40.degree. C. T: 6 5.7 94.607 3.574 0.776 1.043 Standard: RSN0151 81.6 15.4 16.7 99.460 0.545 LA_09_28B weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.7 14.5 17.2 99.536 0.464 Standard: RSN0151 99.460 0.545 LA_09_28B months {grave over ( )}+5.degree. C. T: 3 82.1 5.7 14.04 16.9 99.446 0.554 Standard: RSN0151 99.421 0.579 LA_09_28B months {grave over ( )}-20.degree. C. T: 3 81.9 5.7 14.1 16.8 99.548 0.452 Standard: RSN0151 99.421 0.579 LA_09_28B months {grave over ( )}+40.degree. C. 81.9 5.7 16.3 16.8 90.598 6.155 1.864 1.383 99.421 0.579 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Acetate 10 mM, LA_09_28B 14.257 84.511 1.232 118.94 112 2.09E+12 0.53 50 mg Trehalose, Standard: RSN0151 13.772 85.024 1.204 122.88 100 1.86E+12 0.56 0.01% Polysorbate 20 LA_09_28B 14.56 84.112 1.328 Standard: RSN0151 14.278 84.346 1.377 N/A LA_09_28B 14.174 84.403 1.423 N/A 112 2.09E+12 0.53 Standard: RSN0151 14.278 84.346 1.377 100 1.86E+12 LA_09_28B 14.243 82.977 2.78 Standard: RSN0151 14.278 84.346 1.377 LA_09_28B 13.844 84.829 1.327 Standard: RSN0151 13.864 85.048 1.088 N/A LA_09_28B 13.755 84.943 1.302 Standard: RSN0151 13.864 85.048 1.088 LA_09_28B 14.485 80.65 4.864 203.14 66. Standard: RSN0151 13.864 85.048 1.088 LA_09_28B 13.98 84.747 1.273 Standard: RSN0151 13.864 85.048 1.088 LA_09_28B 14.511 83.408 2.081 189.54 0.56 Standard: RSN0151 14.618 83.409 1.973 LA_09_28B 15.255 82.643 2.103 Standard: RSN0151 14.618 83.409 1.973 LA_09_28B 19.813 71.010 9.177 14.618 83.409 1.973
TABLE-US-00073 TABLE 55 Results - Prototype formulation LA_09_028C ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm] mL] [%] [%] [%] [%] Acetate 10 mM, LA_09_28C T0 N/A N/A 11.9 99.561 0.439 6 mg NaCl, Standard: RSN0151 80.5 18.0 99.691 0.309 0.01% Polysorbate 20 LA_09_28C T: 3 weeks {grave over ( )}+5.degree. C. T: 5.6 11.9 99.560 0.44 Standard: RSN0151 80.5 17.1 99.481 0.519 LA_09_28C 3 weeks {grave over ( )}-20.degree. C. T: 3 5.6 11.8 99.611 0.389 Standard: RSN0151 80.7 16.3 99.481 0.519 LA_09_28C weeks {grave over ( )}+40.degree. C. T: 6 5.5 13.46 + A 95.969 1.283 1.917 0.831 Standard: RSN0151 80.7 16.4 99.481 0.519 LA_09_28C weeks {grave over ( )}+5.degree. C. T: 6 5.6 12.1 99.586 0.414 Standard: RSN0151 80.5 17.7 99.460 0.545 LA_09_28C weeks {grave over ( )}-20.degree. C. T: 6 5.6 12.1 99.584 0.416 Standard: RSN0151 5.5 80.7 17.6 99.460 0.545 LA_09_28C weeks {grave over ( )}+40.degree. C. T: 6 5.7 14.34 + A 94.811 1.975 2.38 0.834 Standard: RSN0151 80.5 5.6 12.1 17.2 99.460 0.545 LA_09_28C weeks {grave over ( )}-80.degree. C. T: 3 N/A 16.1 99.524 0.476 Standard: RSN0151 80.5 5.660 11.64 17.2 99.460 0.545 LA_09_28C months {grave over ( )}+5.degree. C. T: 3 99.522 0.478 Standard: RSN0151 80.5 5.6 11.7 17.2 99.421 0.579 LA_09_28C months {grave over ( )}-20.degree. C. T: 3 99.565 0.435 Standard: RSN0151 80.5 5.7 15.17 + A 17.2 99.421 0.579 LA_09_28C months {grave over ( )}+40.degree. C. 91.992 3.777 2.953 1.278 Standard: RSN0151 99.421 0.579 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Acetate 10 mM, LA_09_28C 14.329 84.427 1.244 130.69 142 1.52E+12 0.87 6 mg NaCl, Standard: RSN0151 13.772 85.024 1.204 122.88 100 1.07E+12 0.81 0.01% Polysorbate 20 LA_09_28C 14.581 84.001 1.418 Standard: RSN0151 14.278 84.346 1.377 LA_09_28C 14.478 84.192 1.330 Standard: RSN0151 14.278 84.346 1.377 LA_09_28C 12.989 83.971 3.04 Standard: RSN0151 14.278 84.346 1.377 N/A LA_09_28C 13.913 84.88 1.207 Standard: RSN0151 13.864 85.048 1.088 LA_09_28C 13.975 84.693 1.332 Standard: RSN0151 13.864 85.048 1.088 LA_09_28C 13.449 82.466 4.085 204.41 66. 28. 14 kDa Standard: RSN0151 13.864 85.048 1.088 189.54 N/A LA_09_28C 13.949 84.815 1.235 Standard: RSN0151 13.864 85.048 1.088 LA_09_28C 14.849 83.298 1.853 Standard: RSN0151 14.618 83.409 1.973 LA_09_28C 14.827 82.896 2.277 N/A Standard: RSN0151 14.618 83.409 1.973 LA_09_28C 18.279 74.233 7.488 Standard: RSN0151 14.618 83.409 1.973
TABLE-US-00074 TABLE 56 Results - Prototype formulation LA_09_028D ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm] mL] [%] [%] [%] [%] Acetate 10 mM, LA_09_28D T0 N/A N/A 99.231 0.336 50 mg Trehalose, Standard: RSN0151 81.7 14.3 16.2 99.691 0.309 20 mM Arginine-HCl, LA_09_28D T: 3 weeks {grave over ( )}+5.degree. C. T: 5.6 99.608 0.353 0.039 0.01% Polysorbate 20 Standard: RSN0151 81.7 14.1 15.9 99.481 0.519 LA_09_28D 3 weeks {grave over ( )}-20.degree. C. T: 3 5.6 99.558 0.395 0.047 Standard: RSN0151 81.5 14.1 15.5 99.481 0.519 LA_09_28D weeks {grave over ( )}+40.degree. C. T: 6 5.6 96.904 1.403 0.957 0.736 Standard: RSN0151 81.7 14.8 15.7 99.481 0.519 LA_09_28D weeks {grave over ( )}+ 5.degree. C. T: 6 81.5 5.6 99.600 0.4 Standard: RSN0151 14.3 16.5 99.460 0.545 LA_09_28D weeks {grave over ( )}-20.degree. C. T: 6 5.5 81.5 5.6 99.633 0.367 Standard: RSN0151 14.4 16.5 99.460 0.545 95.087 2.24 1.578 1.095 LA_09_28D weeks {grave over ( )}+40.degree. C. T: 6 81.8 5.7 16.0 16.4 99.460 0.545 Standard: RSN0151 99.616 0.384 LA_09_28D weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.6 14.4 15.5 99.460 0.545 Standard: RSN0151 99.562 0.416 LA_09_28D months {grave over ( )}+5.degree. C. T: 3 81.8 5.7 13.8 16.3 99.421 0.579 Standard: RSN0151 81.7 5.7 14.1 15.8 99.578 0.380 LA_09_28D months {grave over ( )}-20.degree. C. T: 3 99.421 0.579 Standard: RSN0151 81.5 5.7 16.9 16.4 90.910 4.506 2.69 1.894 LA_09_28D months {grave over ( )}+40.degree. C. 99.421 0.579 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Acetate 10 mM, LA_09_28D 14.336 84.478 1.186 131.16 91 9.75E+13 0.60 50 mg Trehalose, Standard: RSN0151 13.772 85.024 1.204 122.88 100 1.07E+12 20 mM Arginine-HCl, LA_09_28D 14.309 84.351 1.34 0.01% Polysorbate 20 Standard: RSN0151 14.278 84.346 1.377 LA_09_28D 14.048 84.700 1.252 Standard: RSN0151 14.278 84.346 1.377 LA_09_28D 13.872 83.307 2.821 Standard: RSN0151 14.278 84.346 1.377 N/A LA_09_28D 13.931 84.894 1.175 Standard: RSN0151 13.864 85.048 1.088 LA_09_28D 13.683 85.139 1.179 Standard: RSN0151 13.864 85.048 1.088 13.794 82.051 4.155 LA_09_28D 13.864 85.048 1.088 0.81 Standard: RSN0151 13.795 84.882 1.323 LA_09_28D 13.864 85.048 1.088 Standard: RSN0151 14.089 83.957 1.954 LA_09_28D 14.618 83.409 1.973 Standard: RSN0151 14.488 83.570 1.942 LA_09_28D 14.618 83.409 1.973 Standard: RSN0151 19.018 72.345 8.637 LA_09_28D 14.618 83.409 1.973
TABLE-US-00075 TABLE 57 Results - Prototype formulation LA_09_029A ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm[ mL] [%] [%] [%] [%] Histidine 10 mM, LA_09_29A T0 N/A N/A 12.5 99.748 0.252 3 mg/mL NaCl, Standard: RSN0151 79.4 20.3 99.691 0.309 25 mg/mL Trehalose, LA_09_29A T: 3 weeks {grave over ( )}+5.degree. C. T: 5.1 12.5 99.690 0.31 20 mM Arginine-HCl, Standard: RSN0151 79.7 18.1 99.504 0.496 0.01% Polysorbate 20 LA_09_29A 3 weeks {grave over ( )}-20.degree. C. T: 3 5.2 12.5 99.625 0.338 0.037 Standard: RSN0151 79.3 17.9 99.504 0.496 85.224 5.159 9.048 0.569 LA_09_29A weeks {grave over ( )}+40.degree. C. T: 6 5.0 19.43 + A 18.6 99.504 0.496 Standard: RSN0151 78.4 99.664 0.336 LA_09_29A weeks {grave over ( )}+5.degree. C. T: 6 78.3 5.1 12.3 18.9 99.461 0.539 Standard: RSN0151 99.683 0.317 LA_))9_29A weeks {grave over ( )}-20.degree. C. T: 6 5.0 78.9 5.1 12.6 18.7 99.461 0.539 Standard: RSN0151 67.866 8.317 22.58 1.237 LA_09_29A weeks {grave over ( )}+40.degree. C. T: 6 75.8 4.9 29.13 + A 17.9 99.461 0.539 Standard: RSN0151 99.666 0.334 LA_09_29A weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.2 12.5 19.3 99.461 0.539 Standard: RSN0151 79.2 99.642 0.358 LA_09_29A months {grave over ( )}+5.degree. C. T: 3 99.424 0.576 Standard: RSN0151 79.2 99.684 0.316 LA_09_29A months {grave over ( )}-20.degree. C. T: 3 N/A 99.424 0.576 Standard: RSN0151 77.3 46.856 10.905 39.135 3.104 LA_09_29A months {grave over ( )}+40.degree. C. 99.424 0.576 ASD SDS-PAGE WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Histidine 10 mM, LA_09_29A 13.946 84.881 1.174 123.08 174 1.71E+12 0.77 3 mg/mL NaCl, Standard: RSN0151 13.755 85.045 1.2 122.88 100 9.84E+13 0.64 25 mg/mL Trehalose, LA_09_29A 14.304 84.620 1.077 20 mM Arginine-HCl, Standard: RSN0151 0.01% Polysorbate 20 LA_09_29A 14.252 84.531 1.217 Standard: RSN0151 LA_09_29A 50.132 7.197 N/A Standard: RSN0151 13.962 84.777 1.261 LA_09_29A 13.982 84.916 1.102 Standard: RSN0151 16.596 85.342 1.061 LA_))_29A 13.982 84.916 1.102 Standard: RSN0151 42.029 39.19 18.78 LA_09_29A 13.982 84.916 1.102 205.8 90. 14 kDa Standard: RSN0151 13.621 85.201 1.177 189.54 LA_09_29A 13.982 84.916 1.102 Standard: RSN0151 14.401 83.691 1.908 LA_09_29A 14.922 83.041 2.037 Standard: RSN0151 14.189 83.583 2.228 N/A LA_09_29A 14.922 83.041 2.037 Standard: RSN0151 26.766 41.194 32.04 LA_09_29A 14.922 83.041 2.037
TABLE-US-00076 TABLE 58 Results - Prototype formulation LA_09_029B ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH ]nm] mL] [%] [%] [%] [%] Histidine 10 mM, LA_09_27B T0 N/A 81.0 N/A 10.7 20.2 99.679 0.321 50 mg Trehalose, Standard: RSN0151 99.691 0.309 0.01% Polysorbate 20 LA_09_27B T: 3 weeks {grave over ( )}+5.degree. C. 81.3 5.0 10.5 19.3 99.622 0.351 Standard: RSN0151 99.504 0.496 LA 09 27B T: 3 weeks {grave over ( )}-20.degree. C. N/A 5.1 10.4 17.6 99.623 0.342 Standard: RSN0151 99.504 0.496 LA 09 27B T: 3 weeks {grave over ( )}+40.degree. C. N/A 4.9 11.2 18.2 93.016 4.186 2.448 0.368 Standard: RSN0151 99.504 0.496 LA_09_27B T: 6 weeks {grave over ( )}+5.degree. C. 81.0 5.1 10.4 19.9 99.613 0.387 Standard: RSN0151 99.461 0.539 LA_09_27B T: 6 weeks {grave over ( )}-20.degree. C. 5.0 5.1 10.4 19.2 99.653 0.347 Standard: RSN0151 99.461 0.539 LA_09_27B T: 6 weeks {grave over ( )}+40.degree. C. 4.9 11.98 + A 19.4 85.502 7.644 6.093 0.761 Standard: RSN0151 99.461 0.539 LA_09_27B T: 6 weeks {grave over ( )}-80.degree. C. 5.1 10.3 19.0 99.654 0.346 Standard: RSN0151 N/A 99.461 0.539 LA_09_27B T: 3 months {grave over ( )}+5.degree. C. 99.603 0.397 Standard: RSN0151 99.424 0.576 LA_09_27B T: 3 months {grave over ( )}-20.degree. C. N/A 99.650 0.350 Standard: RSN0151 99.424 0.576 LA_09_27B T: 3 months {grave over ( )}+40.degree. C. 67.612 11.218 18.161 3.009 Standard: RSN0151 99.424 0.576 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Histidine 10 mM, LA_09_27B 13.891 84.907 1.202 122.26 118 1.16E+12 0.73 50 mg Trehalose, Standard: RSN0151 13.755 85.045 1.2 122.88 100 9.84E+13 0.64 0.01% Polysorbate 20 LA_09_27B 14.145 84.674 1.182 Standard: RSN0151 14.208 84.672 1.22 LA 09 27B 14.369 84.470 1.162 Standard: RSN0151 14.208 84.672 1.22 LA 09 27B 35.561 59.191 5.247 NA Standard: RSN0151 14.208 84.672 1.22 LA_09_27B 13.622 85.381 0.988 Standard: RSN0151 13.982 84.916 1.102 LA_09_27B 13.758 85.184 1.058 Standard: RSN0151 13.982 84.916 1.102 LA_09_27B 45.968 42.961 11.071 200.33 90. 14 kDa Standard: RSN0151 13.982 84.916 1.102 189.54 LA_09_27B 13.454 83.382 1.164 Standard: RSN0151 13.982 84.916 1.102 LA_09_27B 14.238 83.852 1.910 Standard: RSN0151 14.922 83.041 2.037 NA LA_09_27B 14.108 83.764 2.128 Standard: RSN0151 14.922 83.041 2.037 LA_09_27B 41.575 42.311 16.113 Standard: RSN0151 14.922 83.041 2.037
TABLE-US-00077 TABLE 59 Results - Prototype formulation LA_09_029C ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm] mL] [%] [%] [%] [%] Histidine 10 mM, LA_09_29C T0 N/A 78.9 N/A 11.9 99.741 0.259 6 mg NaCl, Standard: RSN0151 19.4 99.691 0.309 0.01% Polysorbate 20 LA_09_29C T: 3 weeks {grave over ( )}+5.degree. C. T: 79.0 5.1 11.9 19.0 99.643 0.357 Standard: RSN0151 99.504 0.496 LA_09_29C 3 weeks {grave over ( )}-20.degree. C. T: 3 78.4 5.2 12.0 18.6 99.674 0.326 Standard: RSN0151 99.504 0.496 LA_09_29C weeks {grave over ( )}+40.degree. C. T: 6 77.4 5.0 19.67 + A 18.7 84.940 5.173 9.325 0.562 Standard: RSN0151 99.504 0.496 LA_09_29C weeks {grave over ( )}+5.degree. C. T: 6 78.1 5.2 11.8 19.7 99.627 0.373 Standard: RSN0151 99.461 0.539 LA_09_29C weeks {grave over ( )}-20.degree. C. T: 6 5.0 77.8 5.2 12.0 20.8 99.661 0.339 Standard: RSN0151 99.461 0.539 LA_09_29C weeks {grave over ( )}+40.degree. C. T: 6 76.6 4.9 34.92 + A 19.2 61.267 7.789 29.686 1.359 Standard: RSN0151 99.461 0.539 LA_09_29C weeks {grave over ( )}-80.degree. C. T: 3 N/A 5.2 11.9 19.9 99.663 0.337 Standard: RSN0151 99.461 0.539 LA_09_29C months {grave over ( )}+5.degree. C. T: 3 78.4 99.594 0.406 Standard: RSN0151 99.424 0.576 LA_09_29C months {grave over ( )}-20.degree. C. T: 3 78.5 N/A 99.680 0.32 Standard: RSN0151 99.424 0.576 LA_09_29C months {grave over ( )}+40.degree. C. 76.6 36.901 9.237 50.322 3.54 99.424 0.576 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Histidine 10 mM, LA_09_29C 14.119 84.681 1.2 122.43 6 mg NaCl, Standard: RSN0151 13.755 85.045 1.2 122.88 0.01% Polysorbate 20 LA_09_29C 14.046 84.796 1.158 Standard: RSN0151 14.208 84.672 1.22 LA_09_29C 14.171 84.753 1.076 Standard: RSN0151 14.208 84.672 1.22 LA_09_29C 41.068 47.301 11.631 N/A Standard: RSN0151 14.208 84.672 1.22 LA_09_29C 13.871 84.916 1.213 Standard: RSN0151 13.982 84.916 1.102 LA_09_29C 13.764 85.153 1.083 Standard: RSN0151 13.982 84.916 1.102 LA_09_29C 43.326 35.038 21.636 199.58 90. 14 kDa N/A Standard: RSN0151 13.982 84.916 1.102 189.54 LA_09_29C 13.521 85.426 1.053 Standard: RSN0151 13.982 84.916 1.102 LA_09_29C 14.244 83.772 1.984 Standard: RSN0151 14.922 83.041 2.037 N/A LA_09_29C 14.199 83.824 1.977 Standard: RSN0151 14.922 83.041 2.037 LA_09_29C 25.609 37.193 37.198 14.922 83.041 2.037
TABLE-US-00078 TABLE 60 Results - Prototype Formulation LA_09_029D ASD PSD SEC UV Dimer/ Time point Tm DLS [mg/ Monomer Oligomer HMW NP Buffer Formulation number Storage condition pH [.degree. C.] pH [nm] mL] [%] [%] [%] [%] Histidine 10 mM, LA_09_29D T0 N/A N/A 13.1 99.789 0.211 50 mg Trehalose, Standard: RSN0151 80.7 19.4 99.691 0.309 20 mM Arginine-HCl, LA_09_29D T: 3 weeks {grave over ( )}+5.degree. C. T: 5.1 12.6 99.670 0.33 0.01% Polysorbate 20 Standard: RSN0151 80.3 17.8 99.504 0.496 LA_09_29D 3 weeks {grave over ( )}-20.degree. C. T: 3 99.663 0.337 Standard: RSN0151 99.504 0.496 LA_09_29D weeks {grave over ( )}+40.degree. C. T: 6 75.5 5.1 15.99 + A 17.1 87.165 6.561 6.721 Standard: RSN0151 99.504 0.496 LA_09_29D weeks {grave over ( )}+5.degree. C. T: 6 78.6 4.9 12.6 17.2 99.655 0.345 Standard: RSN0151 80.0 18.7 99.461 0.539 LA_09_29D weeks {grave over ( )}-20.degree. C. T: 6 5.1 12.6 99.687 0.313 1.054 Standard: RSN0151 72.1 18.9 99.461 0.539 LA_09_29D weeks {grave over ( )}+40.degree. C. T: 6 5.1 20.47 + A 76.511.000 8.167 14.268 Standard: RSN0151 72.7 17.9 99.461 0.539 LA_09_29D weeks {grave over ( )}-80.degree. C. T: 3 4.9 12.5 99.680 0.32 Standard: RSN0151 N/A 18.5 99.461 0.539 LA_09_29D months {grave over ( )}+5.degree. C. T: 3 5.1 99.632 0.368 Standard: RSN0151 80.0 99.424 0.576 LA_09_29D months {grave over ( )}-20.degree. C. T: 3 N/A 99.677 0.323 2.8 Standard: RSN0151 80.0 99.424 0.576 56.365 11.326 29.509 78.0 99.424 0.576 ASD SDS-PAGE non-red. WCX main Elisa Buffer Formulation number % acic % neutral % basic Comment band comment EC50% EC50 slope Histidine 10 mM, LA_09_29D 14.446 84.366 1.189 124.60 169 1.66E+12 0.66 50 mg Trehalose, Standard: RSN0151 13.755 85.045 1.2 122.88 100 9.84E+13 20 mM Arginine-HCl, LA_09_29D 14.168 84.691 1.141 0.01% Polysorbate 20 Standard: RSN0151 14.208 84.672 1.22 LA_09_29D 14.366 84.473 1.161 Standard: RSN0151 14.208 84.672 1.22 0.64 LA_09_29D 40.897 48.391 10.712 Standard: RSN0151 14.208 84.672 1.22 LA_09_29D 13.796 84.86 1.344 Standard: RSN0151 13.982 84.916 1.102 LA_09_29D 13.806 84.995 1.2 Standard: RSN0151 13.982 84.916 1.102 LA_09_29D 44.302 41.575 14.124 Standard: RSN0151 13.982 84.916 1.102 LA_09_29D 13.995 84.985 1.020 Standard: RSN0151 13.982 84.916 1.102 198.81 90. 14 kDa N/A N/A LA_09_29D 14.623 83.446 1.931 189.54 Standard: RSN0151 14.922 83.041 2.037 LA_09_29D 14.150 83.575 2.276 Standard: RSN0151 14.922 83.041 2.037 30.807 41.914 27.279 14.922 83.041 2.037
Anti-CXCR5 (20 mg/mL) Formulation Studies
[0484] The data in Examples 13-16 were collected during formulation studies for the Lead CXCR5 Antibody and its drug product for intravenous and subcutaneous administration. The objective of the formulation studies was to provide a stable, clear or slightly opalescent, and colorless or slightly yellow, visual particle-free Lead CXCR5 Antibody solution for injection for phase I.
Materials
Drug Substance (DS)
[0485] Two drug substance batches were used for these formulation studies. One was formulated in phosphate buffered saline (PBS) and the other was formulated in citrate buffer. See Table 61.
TABLE-US-00079 TABLE 61 Available drug substance batches Lead Ab pH-value Batch no. Amount concentration Buffer [--] RSN0151 10 g 5.0 mg/mL 155 mM PBS 7.2 SCB0001 20 g 20.30 mg/mL 10 mM Citrate 6.0
Excipients
[0486] Table 62 shows excipients that were used during the formulation studies.
TABLE-US-00080 TABLE 62 Excipients Excipient Material no. Supplier Arginine 1.01587 Merck Citric acid 100241 Merck Histidine 1.04352 Merck Hydrochloric acid 114027 H600 Saccharose S3929 Sigma-Aldrich Sodium acetate 1.06265 Merck Sodium chloride 10158 H600 Sodium citrate 114196 H600 Sodium hydroxide 114076 H600 Polysorbate 20 139850 H600 .alpha.,.alpha.-Trehalose T9531 Sigma-Aldrich
Methods
Sample Preparation
[0487] Ultrafiltration/Diafiltration was performed on a small scale using VivaSpin devices with a Hydrosart membrane and a 30 kDa cut-off. RSN material was concentrated from 5 mg/mL to 20 mg/mL, and phosphate (PBS) buffer was exchanged to either 10 mM citrate buffer pH 6.0, acetate buffer pH 5.5, or histidine buffer pH 5.0. The VivaSpin units were placed at room temperature (RT) in a common laboratory centrifuge and centrifuged with 2000 rpm. The solution was filtered over a 0.2 .mu.m Minisart before analytical testing. All samples were stored between +2.degree. and +8.degree. C., tightly closed, and protected from light, until analytical testing at T0 and after one week thermal stress at +40.degree. C. or after mechanical stress for 2.5 hours, 300 rpm at RT (only for evaluation of polysorbate 20 concentration).
Analytical Methods
[0488] The following techniques were used for sample analysis:
TABLE-US-00081 TABLE 63 Analytical techniques used Technique (Company) Parameter to investigate Organoleptic (--) Appearance Nephelometer (Hach Lange) Turbidity pH-meter (WTW) pH-value UV (Perkin Elmer) Concentration of mAB Densimeter (Paar) Density Osmometer (Knauer) Osmolality Viscosimeter (Paar) Viscosity Dynamic Light Scattering Hydrodynamic diameter (Malvern) SEC (N/A) Mono-/Di-/Oligomer and High Molecular Weight Protein (HMWP)/ Low Molecular Weight (LMW) content WCX(N/A) Isoforms (acid/basic/neutral) ELISA*(N/A) Potency (Binding) SDS-Page (red.)** (N/A) HC/LC, mAB-fragments SDS-Page (non-red.)** (N/A) Aggregation and degradation products HIAC*(N/A) Particulate matter *Some samples will be analyzed. **SDS-Page will be performed in case SEC shows unusual results.
Example 13--Additional pH Optimization
[0489] Preformulation studies identified 10 mM citrate buffer at pH 6.0 as the best buffer with less Lead CXCR5 Antibody aggregation tendency. To obtain a pH-profile in citrate buffer, stepwise pH-dependent stability from pH 5.0 to 7.0 was evaluated. Due to limited drug substance availability, in-depth pH-screening was performed only with 10 mM citrate buffer. Samples were taken at T0 and after one week thermal stress at +40.degree. C. See Tables 64-68.
TABLE-US-00082 TABLE 64 Overview of samples Batch no. Target pH-value (--) Measured pH-value (--) LA_09_030 5.0 5.1 LA_09_031 5.3 5.4 LA_09_032 5.5 5.6 LA_09_033 5.7 5.8 LA_09_034 6.0 6.1 LA_09_035 6.3 6.4 LA_09_036 6.5 6.6 LA_09_037 6.7 6.8 LA_09_038 7.0 7.1
TABLE-US-00083 TABLE 65 results T0 Appear- Measured pH- mAB conc. Hydrodynamic Batch no. ance value (--) (mg/mL) diameter (nm) LA_09_030 Clear 5.1 23.35 15.42 + aggr. LA_09_031 Clear 5.4 21.95 12.85 LA_09_032 Clear 5.6 23.00 12.98 LA_09_033 Clear 5.8 21.21 12.99 LA_09_034 Clear 6.1 22.77 12.83 LA_09_035 Clear 6.4 23.87 13.22 LA_09_036 Clear 6.6 23.74 13.04 LA_09_037 Clear 6.8 22.85 13.00 LA_09_038 Clear 7.1 21.96 13.32
TABLE-US-00084 TABLE 66 results T1 week +40.degree. C. Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_030 Clear 5.2 15.62* 15.27 + aggr. LA_09_031 Clear 5.5 21.91 16.74 + aggr. LA_09_032 Clear 5.6 24.32 13.59 LA_09_033 Clear 5.8 24.74 13.83 LA_09_034 Clear 6.1 24.18 13.25 LA_09_035 Clear 6.5 N/A 13.41 LA_09_036 Clear 6.6 23.03 13.37 LA_09_037 Clear 6.9 22.68 13.24 LA_09_038 Clear 7.2 23.33 14.40 *Unusual result due to dilution mistake
TABLE-US-00085 TABLE 67 ASD results T0 % % Di-/ % % Batch no. Monomer Oligomer HMWP % % % basic LA_09_030 99.63 0.37 -- -- 13.60 85.18 1.22 LA_09_031 99.57 0.43 -- -- 13.52 85.24 1.24 LA_09_032 99.48 0.52 -- -- 13.71 85.04 1.25 LA_09_033 99.51 0.49 -- -- 13.99 84.61 1.40 LA_09_034 99.41 0.59 -- -- 13.62 85.17 1.21 LA_09_035 99.24 0.76 -- -- 13.72 84.64 1.64 LA_09_036 98.72 1.28 -- -- 13.72 84.45 1.83 LA_09_037 98.95 1.05 -- -- 13.60 84.73 1.67 LA_09_038 98.58 1.42 -- -- 13.84 84.13 2.03
TABLE-US-00086 TABLE 68 results T1 week +40.degree. C. Batch no. % Monomer % Di-/Oligomer % HMWP % LMW % acidic % neutral % basic LA_09_030 95.19 0.97 3.29 0.55 11.90 83.92 4.18 LA_09_031 96.47 0.89 2.14 0.50 12.19 84.70 3.11 LA_09_032 96.82 0.92 1.69 0.57 12.14 85.48 2.38 LA_09_033 97.13 0.94 1.48 0.45 12.41 85.04 2.55 LA_09_034 97.73 0.97 0.82 0.48 12.35 85.69 1.96 LA_09_035 97.58 1.12 0.89 0.41 11.93 85.74 2.33 LA_09_036 97.47 1.32 0.86 0.35 12.01 85.46 2.53 LA_09_037 97.35 1.41 0.87 0.37 12.08 85.28 2.64 LA_09_038 96.97 1.62 0.97 0.44 11.65 85.10 3.25
[0490] In conclusion, the data confirm the results already generated during preformulation studies: increasing the pH causes the monomer content to decrease and dimer rate to increase. Samples at +40.degree. C. showed with lower pH-value decrease in HMWs up to pH 6.0 and then increase up to pH 5.0.
Example 14--Additional Buffer Optimization
[0491] Next, citrate, acetate, and histidine (as back-up buffer) buffers were screened at 5/10/25/50 mM at the selected pH-values. See Tables 69-83.
TABLE-US-00087 TABLE 69 Overview on samples - Citrate buffer pH 6.0 Batch no. Citrate buffer conc. [mM] LA_09_040 5 LA_09_034 10 LA_09_041 25 LA_09_042 50
TABLE-US-00088 TABLE 70 results after T0 Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_040 Clear 6.1 20.05 13.47 LA_09_034 Clear 6.1 22.77 12.83 LA_09_041 Clear 6.2 20.48 11.91 LA_09_042 Clear 6.1 22.19 11.87
TABLE-US-00089 TABLE 71 results after T1 week +40.degree. C. Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_040 Clear 6.3 21.62 13.78 LA_09_034 Clear 6.1 24.18 13.25 LA_09_041 Clear 6.2 18.37 12.50 LA_09_042 Clear 6.2 20.59 12.07
TABLE-US-00090 TABLE 72 results T0 % Di-/Oligomer % HMWP Batch no. % Monomer (RRT 0.84) (RRT 0.68) % % % % basic LA_09_040 99.49 0.51 -- -- 13.19 85.81 1.00 LA_09_034 99.41 0.59 -- -- 13.62 85.17 1.21 LA_09_041 99.55 0.42 0.03 -- 13.24 85.67 1.09 LA_09_042 99.60 0.39 0.01 -- 13.41 85.48 1.11
TABLE-US-00091 TABLE 73 results after thermal stress 1 week/+40.degree. C. % Di-/Oligomer % HMWP Batch no. % Monomer (RRT 0.84) (RRT 0.68) % % % % basic LA_09_040 98.58 0.86 0.17 0.39 12.52 85.95 1.53 LA_09_034 97.73 0.97 0.82 0.48 12.35 85.69 1.96 LA_09_041 98.81 0.65 0.21 0.33 12.54 86.07 1.38 LA_09_042 98.87 0.59 0.14 0.40 12.45 86.10 1.45
TABLE-US-00092 TABLE 74 Overview on samples - Histidine buffer pH 5.0 Batch no. Histidine buffer conc. [mM] LA_09_043 5 LA_09_044 10 LA_09_045 25 LA_09_046 50
TABLE-US-00093 TABLE 75 results after T0 Appear- ance Hydrody- Measured pH mAB conc. diameter Batch no. namic value (--) [mg/mL] (nm) LA_09_043 Clear 5.5 21.89 8.40 + aggr. LA_09_044 Clear N/A 6.95* 11.34 LA_09_045 Clear 5.2 21.78 11.86 + aggr. LA_09_046 Clear 5.1 20.04 11.86 *Low data due to sample dilution mistake
TABLE-US-00094 TABLE 76 results after T1 week +40.degree. C. Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter LA_09_043 Clear 5.5 21.34 8.81 LA_09_044 Clear 5.5 24.18 13.25 LA_09_045 Clear 5.2 23.62 11.88 LA_09_046 Clear 5.1 21.41 12.50 + aggr.
TABLE-US-00095 TABLE 77 ASD results T0 Batch no. % % % basic % Di- HMWP % acidic % % LA_09_043 99.55 0.45 -- -- 13.69 85.16 1.15 LA_09_044* N/A N/A N/A N/A N/A N/A N/A LA_09_045 99.68 0.32 -- -- 13.73 85.00 1.27 LA_09_046 99.70 0.30 -- -- 13.43 85.49 1.08 *not analyzed due to dilution mistake
TABLE-US-00096 TABLE 78 results after thermal stress 1 week/+40.degree. C. Batch no. % % % basic % Di- HMWP % acidic % % LA_09_043 98.72 0.82 -- 0.46 13.63 84.60 1.75 LA_09_044* N/A N/A N/A N/A N/A N/A N/A LA_09_045 98.30 0.80 0.44 0.56 12.79 85.19 2.02 LA_09_046 97.79 0.68 1.07 0.46 12.61 84.75 2.64 *not analyzed due to dilution mistake
TABLE-US-00097 TABLE 79 Overview of samples - Acetate buffer pH 5.5 Batch no. Acetate buffer conc. [mM] LA113244_09_053 5 LA113244_09_054 10 LA113244_09_055 25 LA113244_09_056 50
TABLE-US-00098 TABLE 80 results after T0 Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter LA_09_053 Clear 5.88 25.41 10.44 LA_09_054 Clear 5.68 21.91 13.21 + aggr. LA_09_055 Clear 5.56 21.53 14.06 LA_09_056 Clear 5.56 22.08 13.54
TABLE-US-00099 TABLE 81 results after T0 Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter LA_09_053 Clear 5.88 26.43 11.48 LA_09_054 Clear 5.69 23.40 13.44 LA_09_055 Clear 5.61 21.53 14.46 LA_09_056 Clear 5.56 21.68 13.71
TABLE-US-00100 TABLE 82 results T0 % % % % Batch no. % Di- HMWP % acidic % basic LA_09_053 98.89 0.99 0.02 0.10 14.44 83.67 1.89 LA_09_054 98.84 1.09 0.07 -- 11.30 86.80 1.90 LA_09_055 98.91 0.99 0.07 0.03 11.30 86.77 1.93 LA_09_056 98.97 0.87 0.10 0.06 11.27 86.90 1.83
TABLE-US-00101 TABLE 83 results after thermal stress 1 week/+40.degree. C. % Di-/Oligomer % HMWP Batch no. % Monomer (RRT 0.84) (RRT 0.68) % % acidic % % basic LA_09_053 97.68 1.96 0.04 0.32 14.53 81.50 3.91 LA_09_054 97.83 1.99 0.09 0.09 11.13 85.79 3.08 LA_09_055 97.88 2.00 0.09 0.09 11.09 85.76 3.15 LA_09_056 98.10 1.22 0.59 0.09 10.92 86.24 2.74
[0492] In conclusion, the data confirm the results generated during the preformulation studies. Using citrate as the buffer agent, the monomer content is slightly higher than with acetate buffer and histidine buffer. With histidine, high aggregation behavior is observable, even at T0, leading to difficulties in analytical sample preparation. A significant difference between the tested buffer concentrations cannot be measured, so all three buffers citrate, histidine, and acetate will be used with a concentration of 10 mM.
Example 15--Additional Surfactant Optimization
[0493] Based on preformulation trials, the addition of non-ionic surfactant polysorbate 20 (0.01%) showed beneficial effects on stability, so further evaluation of its concentration was performed by adding the following polysorbate 20 concentrations to the respective buffers: 0.0025%/0.005%/0.01%/0.02%. See Tables 84-93.
TABLE-US-00102 TABLE 84 Overview of samples in acetate buffer Polysorbate 20 concentration Batch no. in [mg/mL] as percent[%] LA_09_058 0.2 0.02 LA_09_059 0.1 0.01 LA_09_060 0.05 0.005 LA_09_061 0.025 0.0025
TABLE-US-00103 TABLE 85 results after T0 Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_058 Clear 5.61 23.71 12.50 LA_09_059 Clear 5.64 22.76 12.94 LA_09_060 Clear 5.63 23.89 12.83 LA_09_061 Clear 5.64 25.79 12.88
TABLE-US-00104 TABLE 86 results after mechanical stress 300 rpm/150 min Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_058 Clear 5.61 22.82 12.48 LA_09_059 Clear 5.67 22.47 12.73 LA_09_060 Clear 5.55 22.90 12.59 LA_09_061 Clear 5.65 25.19 12.76
TABLE-US-00105 TABLE 87 results T0 Batch no. basic % % Di- % HMWP % % acidic % % LA_09_058 99.17 0.80 0.03 -- 11.25 86.94 1.81 LA_09_059 99.16 0.81 0.03 -- 11.27 86.94 1.79 LA_09_060 99.17 0.81 0.03 -- 11.44 86.80 1.76 LA_09_061 99.13 0.84 0.03 -- 11.31 86.91 1.78
TABLE-US-00106 TABLE 88 results after mechanical stress 300 rpm/150 min % Di-/Oligomer % HMWP Batch no. % Monomer (RRT 0.84) (RRT 0.68) % % acidic % % basic LA_09_058 99.16 0.81 0.03 -- 11.27 86.95 1.78 LA_09_059 99.16 0.82 0.02 -- 11.24 86.91 1.85 LA_09_060 99.17 0.81 0.03 -- 11.46 86.77 1.77 LA_09_061 99.16 0.81 0.02 11.22 86.97 1.79
TABLE-US-00107 TABLE 89 Overview of samples in citrate buffer Polysorbate 20 concentration Batch no. in (mg/mL) as percent (%) LA_09_062 0.2 0.02 LA_09_063 0.1 0.01 LA_09_064 0.05 0.005 LA_09_065 0.025 0.0025
TABLE-US-00108 TABLE 90 results after T0 Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) (mg/mL) diameter (nm) LA_09_062 Clear 6.05 23.72 12.67 LA_09_063 Clear 6.03 25.18 12.73 LA_09_064 Clear 6.04 23.85 12.47 LA_09_065 Clear 6.04 22.65 12.46
TABLE-US-00109 TABLE 91 results after T1 week +40.degree. C. Appear- Measured pH mAB conc. Hydrodynamic Batch no. ance value (--) [mg/mL] diameter (nm) LA_09_062 Clear 6.07 23.44 12.99 LA_09_063 Clear 6.03 24.39 12.59 LA_09_064 Clear 6.04 23.93 12.39 LA_09_065 Clear 6.04 22.27 12.37
TABLE-US-00110 TABLE 92 results T0 Batch no. % Monomer % Di-/Oligomer % HMWP % % acidic % % basic LA_09_062 99.25 0.70 0.05 -- 11.33 86.24 2.43 LA_09_063 99.28 0.68 0.04 -- 11.00 86.36 2.64 LA_09_064 99.23 0.74 0.03 -- 10.93 86. 45 2.62 LA_09_065 99.28 0.69 0.03 -- 10.97 86.25 2.77
TABLE-US-00111 TABLE 93 results after mechanical stress 300 rpm/150 min Batch no. % Monomer % Di-/Oligomer % HMWP % % acidic % % basic LA_09_062 99.27 0.69 0.04 -- 11.25 86.27 2.48 LA_09_063 99.32 0.65 0.03 -- 10.87 86.59 2.54 LA_09_064 99.19 0.78 0.03 -- 10.91 86.56 2.53 LA_09_065 99.16 0.80 0.04 -- 10.79 86.51 2.70
[0494] In conclusion, no significant differences in samples containing acetate or citrate buffer with various polysorbate concentration were measurable. To ensure mAb prevention against mechanical stress over a longer period of time than tested for 150 min, the polysorbate concentration was set to 0.2 mg/mL. This amount was also proposed based on preformulation studies.
Example 16--Additional Isotonicity Optimization
[0495] During preformulation studies, NaCl, Trehalose, and Arginine-HCl were identified as additives for isotonicity and stability purposes. Arginine-HCl was then dropped due to less mAb stability effects. Depending on buffer concentration and pH-value, isotonant/stabilizer amount is adapted to achieve osmolality of at least 240 mOsmol/kg according to Ph. Eur.
[0496] The use of trehalose was challenged as it is not a compendial excipient and is high priced. During preformulation studies, sucrose (saccharose) caused slightly more aggregation, but was not followed-up and verified in further studies. Therefore, a new short-term stability study over four weeks was designed, including trehalose as well as saccharose in both 10 mM citrate and acetate buffer with storage temperatures at +50, +250, and +40.degree. C. See Tables 94-102.
[0497] Fine-tuning of osmolality of at least 240 mOsmol/kg was performed with NaCl.
TABLE-US-00112 TABLE 94 Overview of samples Target pH- Batch no. Buffer value [--] Polysorbate 20 NaCl Stabilizing agent LA_09_051A 10 mM Citrate 6.0 0.2 mg/mL 2 mg/mL Sucrose 60 mg/mL LA_09_051B 10 mM Citrate 6.0 0.2 mg/mL 2 mg/mL Trehalose 60 mg/mL LA_09_052A 10 mM Citrate 5.5 0.2 mg/mL 2 mg/mL Sucrose 60 mg/mL LA_09_052B 10 mM Citrate 5.5 0.2 mg/mL 2 mg/mL Trehalose 60 mg/mL
TABLE-US-00113 TABLE 95 results T0 Appear- Measured pH mAB-conc. Osmolality Batch no. ance value (--) (mg/mL) (mOsmol/kg) LA_09_051A Clear 5.89 21.46 289 LA_09_051B Clear 5.94 21.46 268 LA_09_052A Clear 5.82 22.07 273 LA_09_052B Clear 5.80 22.07 256
TABLE-US-00114 TABLE 96 results T 4 weeks, +5.degree. C. Measured pH- mAB-conc. Hydrodynamic Batch no. value (--) (mg/mL) diameter (nm) LA_09_051A 6.02 21.81 13.58 LA_09_051B 5.95 22.10 13.35 LA_09_052A 5.86 21.70 15.12 LA_09_052B N/A N/A N/A
TABLE-US-00115 TABLE 97 results T 4 weeks, +25.degree. C. Measured pH- mAB-conc. Hydrodynamic Batch no. value (mg/mL) diameter (nm) LA_09_051A 6.06 22.30 13.57 LA_09_051B 6.02 22.09 13.41 LA_09_052A 5.91 21.98 15.14 LA_09_052B N/A N/A N/A
TABLE-US-00116 TABLE 98 results T 4 weeks, +40.degree. C. Measured pH- mAB-conc. Hydrodynamic Batch no. value (mg/mL) diameter (nm) LA_09_051A 6.04 22.21 14.73 LA_09_051B 5.95 21.86 14.11 LA_09_052A 5.91 22.23 16.37 LA_09_052B 5.89 22.84 16.02
TABLE-US-00117 TABLE 99 results T0 mAB-conc. Batch no. % % Di- % % % (mg/mL) LA_09_051A 99.53 0.47 13.78 83.85 2.37 23.45 LA_09_051B 99.54 0.46 13.73 84.83 1.94 22.91 LA_09_052A 99.44 0.56 13.83 83.99 2.18 22.54 LA_09_052B 99.44 0.56 14.39 83.38 2.23 23.19
TABLE-US-00118 TABLE 100 results after thermal stress 4 weeks/+5.degree. C. % Di- % HMWP mAB-conc. Batch no. % (RRT 0.84) (RRT 0.68) % % % % (mg/mL) LA_39_05 1A 99.21 0.38 -- 0.41 11.34 87.11 1.55 24.24 LA_09_05 1B 98.97 0.46 -- 0.57 11.26 87.17 1.58 23.71 LA_09_05 2A 98.81 0.54 -- 0.65 11.47 86.86 1.67 22.63 LA_09_05 2B 99.00 0.55 -- 0.45 11.46 86.90 1.64 23.17
TABLE-US-00119 TABLE 101 results after thermal stress 4 weeks/+25.degree. C. mAB-conc. Batch no. % % Di- % % LMW % % % (mg/mL) LA_09_051A 98.87 0.46 -- 0.67 11.01 87.31 1.69 26.16 LA_09_051B 98.72 0.53 -- 0.75 11.02 87.29 1.70 23.63 LA_09_052A 98.25 0.83 -- 0.92 11.69 86.35 1.96 24.27 LA_09_052B 98.52 0.74 -- 0.74 11.46 86.62 1.92 23.52
TABLE-US-00120 TABLE 102 results after thermal stress 4 weeks/+40.degree. C. mAB-conc. Batch no. % % Di- % % LMW % % % (mg/mL) LA_09_051A 96.84 0.95 1.06 1.15 10.16 87.05 2.79 25.04 LA_09_051B 96.96 0.97 1.01 1.06 10.01 87.05 2.83 23.60 LA_09_052A 96.00 1.63 1.17 1.20 11.32 84.89 3.79 24.11 LA_09_052B 96.23 1.49 1.21 1.07 11.01 85.30 3.69 24.29
[0498] In conclusion, no significant differences between citrate and acetate buffer were measured, and no difference at accelerated conditions between trehalose and saccharose was visible. Citrate buffer with saccharose was selected for further studies.
Determination of DP Manufacturing Process Parameters
[0499] DS batch in citrate buffer was used to determine manufacturing process parameters. Preformulation studies indicated that the DS was not that susceptible to oxidation, and that light protection or nitrogen overlay or purging during manufacturing was required. Standard glass equipment as well as silicone tubings (SaniTech65) were used.
Adding Order
[0500] Experiments evaluating the adding order of the excipients were limited due to the small dilution volume of DS.
[0501] The DS was weighed in a glass bottle, polysorbate 20 as first excipient, saccharose as the second excipient, and NaCl as third excipient were added and rinsed with citrate buffer 10 mM pH 6.0 to dilute the content of DS to 20 mg/mL.
Stirring Speed and Time
[0502] Stirring speed was set at 100 rpm to reduce mechanical stress for the DS. Due to the fact that all excipients were well water-soluble, stirring time was set to 5 minutes.
Monitoring Parameters and IPCs
[0503] Monitoring parameters such as appearance, turbidity, density, and viscosity, and IPCs such as pH-value and osmolality were routinely checked during sample manufacturing according to the following Table 103:
TABLE-US-00121 TABLE 103 Before Filtration After Filtration After Filling Appearance colorless colorless colorless to slightly to slightly to slightly yellow yellow yellow Density 1.006 mg/mL Not measured Not measured Turbidity Clear Clear Clear Viscosity Not measured Not measured <5 mPa s pH-value 6.0 .+-. 0.2 6.0 .+-. 0.2 Not measured (20-25.degree. C.) (20-25.degree. C.) Osmolality 290 .+-. 40 mOsmol/kg 290 .+-. 40 Not measured mOsmol/kg
No issues were observed during manufacturing. The limits for osmolality were set-up based on measured data.
Filtration Process
[0504] According to preformulation studies, polyethersulfone was a suitable membrane for sterile filtration (Sartorius, 0.22 .mu.m). No potential pH-shifts after filtration could be observed, as filtration rate and time showed standard values for filtration of an aqueous solution. Filter integrity testing was routinely performed without any issues.
Filling Process
[0505] Standard dosing equipment made of stainless steel, such as the filling pump and filling needle were investigated. Also, duration and filling speed was monitored. Extractable volume of filled DP was determined. An overfilling of 0.2 mL was required to ensure an extractable volume of 1.5 mL.
Material Compatibility
[0506] All preformulation and formulation studies were performed in glass as standard manufacturing equipment, which is also the recommendation for equipment to be used for GMP manufacturing.
Cleaning Agents
[0507] Cleaning of manufacturing equipment was performed according to the respective SOPs using the dishwasher with standard cleaning agent Neodisher.RTM.. A manual pre-cleaning with water for injection was routinely done before. No harmful effects of cleaning agents were observed.
Summary of Additional Formulation Studies for Lead CXCR5 Antibody (20 mg/mL)
[0508] For selection of phase I Lead CXCR5 Antibody DP formulation, citrate 10 mM at pH 6.0 was selected as the buffer over histidine and acetate. The pH-value of the solution was set at 6.0, as increasing or decreasing the pH-value means a reduction in monomer content. The buffer concentration was set at a medium concentration of 10 mM, although there was no significant difference between concentrations of 5-50 mM.
[0509] Polysorbate 20 was chosen as the surfactant with 0.2 mg/mL (0.02%), sufficient to stabilize the DS against mechanical stress.
[0510] Sucrose (saccharose) was selected as the stabilizer against thermal stress in favour of trehalose. The concentration of saccharose was set at 60 mg/mL (6%).
[0511] NaCl will be used as the isotonant agent in a concentration of 2.0 mg/mL (0.2%) in order to achieve an osmolality of DP of about 300 mOsmol/kg.
Anti-CXCR5 (100 Mg/Ml) Formulation Studies
[0512] The data in Examples 17-21 were collected during formulation studies for the Lead CXCR5 Antibody and its drug product for intravenous and subcutaneous administration. The objective of the formulation studies was to provide a stable, clear or slightly opalescent, and colorless or slightly yellow, visual particle-free Lead CXCR5 Antibody solution for injection for phase I.
Methods
Sample Preparation
[0513] UF/DF was performed on a small scale using VivaSpin devices with a Hydrosart membrane and a 30 kDa cut-off. RSN material was concentrated from ca. 20 mg/mL to 100 mg/mL. All solutions were already in the final formulation buffer (10 mM citrate buffer at pH 6.0).
[0514] The VivaSpin units were placed at RT in a common laboratory centrifuge and centrifuged at 2000 rpm. Solution was filtered over 0.2 .mu.m Minisart before analytical testing.
[0515] All samples were stored between +2.degree. and +8.degree. C., tightly closed and protected from light, until analytical testing at T0 and after one week thermal stress at +40.degree. C. or after mechanical stress.
Analytical Methods
[0516] The following techniques were used for sample analysis:
TABLE-US-00122 TABLE 104 Analytical techniques used Technique Parameter to investigate Organoleptic Appearance Nephelometer Turbidity pH-meter pH-value UV mAB-concentration Densimeter Density Osmometer Osmolality Viscosimeter Viscosity DLS Hydrodynamic diameter DSC* Unfolding temperature SEC Mono-/Di-/Oligomer/HMW content WCX Isoforms (acid/basic/neutral) ELISA* Potency (Binding) SDS-Page (red.)** HC/LC, mAB-fragments SDS-Page (non-red.)** Aggregation and degradation products HIAC* Subvisible particles *Some samples will be analyzed. **SDS-Page will be performed in case SEC shows unusual results.
Example 17--Excipient Screening
[0517] Preformulation studies identified 10 mM citrate buffer at pH 6.0 as the best buffer with less Lead CXCR5 Antibody aggregation tendency. In previous studies at 20 mg/mL, a formulation containing 10 mM citrate buffer, 60 mg/mL (6%) sucrose, 2 mg/mL (0.2%) NaCl, and 0.2 mg/mL (0.02%) Polysorbate 20 was selected. Those excipients plus some alternatives were tested to confirm the suitability of the selected formulation at a higher concentration (100 mg/mL).
[0518] Different formulations were stressed thermally at 40.degree. C. for 7 days and mechanically at 100 rpm for 5 hrs. Additionally, the unfolding temperature for the different formulations were screened at 100 mg/mL using DSC (Differential scanning calorimetry).
[0519] The following excipients were tested:
Sucrose.fwdarw.60 mg/mL Trehalose.fwdarw.60 mg/mL Arginine.fwdarw.30 mg/mL Lysine.fwdarw.30 mg/mL Glycine.fwdarw.30 mg/mL
[0520] NaCl or Mannitol was added as an isotonant. No salts were needed for viscosity reduction (around 2.1 cP).
[0521] The results of T0 and T7 days are shown in Table 105.
TABLE-US-00123 TABLE 105 Excipients screening SEC Isoforms by WCX Activity SDS-Page Formulation HMW Mono. acidic neutral basic rel. potency non-reducing T composition (mg/mL) Nr. [%] [%] [%] [%] [%] [%] conditions T zero Sucrose (60) + NaCl (2) 72_A1 2.7 97.0 11.7 84.2 4.0 165 comparable to reference Sucrose (60) + Mannitol (15) 72_A2 2.8 97.1 11.9 84.4 3.7 205 comparable to reference Trehalose (60) + NaCl (2) 72_B1 2.8 97.0 11.7 84.8 3.6 110 comparable to reference Trehalose (60) + Mannitol (15) 72_B2 2.8 97.0 11.8 84.2 4.0 156 comparable to reference Arginine (30) 72_C1 2.5 97.2 11.9 84.3 3.8 160 comparable to reference Arginine (20) + NaCl (2) 72_C2 2.6 97.2 11.8 84.6 3.6 144 comparable to reference Arginine (20) + Mannitol (15) 72_C3 2.6 97.2 12.2 83.8 3.9 117 comparable to reference Lysine (30) + NaCl (2) 72_D1 2.6 97.1 12.7 82.1 5.3 130 comparable to reference Lysine (30) + Mannitol (15) 72_D21 2.6 97.1 12.6 82.2 5.3 88 comparable to reference Glycine (20) 72_E1 2.7 97.1 12.4 83.5 4.1 170 comparable to reference Glycine (20) + NaCl (2) 72_E2 2.7 97.1 12.4 83.5 4.1 174 comparable to reference Glycine (20) + Mannitol (15) 72_E3 2.7 97.0 12.7 83.3 4.1 111 comparable to reference T 7 days Sucrose (60) + NaCl (2) 72_A1 3.5 96.3 11.4 84.4 4.2 188 comparable to reference at 40.degree. C. Sucrose (60) + Mannitol (15) 72_A2 3.5 96.3 11.3 84.6 4.2 243 comparable to reference Trehalose (60) + NaCl (2) 72_B1 3.4 96.4 11.5 84.4 4.1 191 comparable to reference Trehalose (60) + Mannitol (15) 72_B2 3.5 96.3 11.4 84.3 4.3 266 comparable to reference Arginine (30) 72_C1 3.6 96.1 11.2 84.9 4.0 143 comparable to reference Arginine (20) + NaCl (2) 72_C2 3.4 96.4 11.6 84.5 3.9 164 comparable to reference Arginine (20) + Mannitol (15) 72_C3 3.3 96.4 11.3 85.3 3.4 not tested comparable to reference Lysine (30) + NaCl (2) 72_D1 6.5 93.1 30.9 48.0 21.1 264 comparable to reference Lysine (30) + Mannitol (15) 72_D21 5.9 93.7 31.2 47.9 20.9 297 comparable to reference Glycine (20) 72_E1 3.2 96.6 11.3 84.4 4.3 180 comparable to reference Glycine (20) + NaCl (2) 72_E2 3.3 96.4 11.6 84.4 4.0 not tested comparable to reference Glycine (20) + Mannitol (15) 72_E3 3.2 96.6 11.2 84.6 4.2 not tested comparable to reference
Thermal Stress
[0522] None of the samples showed turbidity before or after stress.
[0523] Lysine showed: a pH shift to 9.8, a very high tendency to aggregate, a very high increase in acidic and basic isoforms, and high molecular weight bands in SDS-PAGE. As a result, it was excluded from further consideration.
[0524] Formulations with mannitol showed bad binding in an ELISA assay after stress. As a result, NaCl is the favored isotonant.
[0525] Sucrose showed slightly better chemical stability than trehalose, but additional bands were seen in SDS-PAGE after stress (for both).
[0526] Arginine (especially in the presence of NaCl) and glycine had a similar SEC profile, but no additional bands were seen in SDS-PAGE after stress.
Protein Associated Formation Measured by Dynamic Light Scattering (DLS)
[0527] Lead CXCR5 Antibody showed a significant increase in the hydrodynamic diameter (Z-Average) by increasing the concentration (FIG. 34). This behavior was fully reversible upon dilution. For further investigation of this effect, the different Lead CXCR5 Antibody concentrations were measured by analytical ultra centrifugation (AUC) and aggregation was excluded. The conclusion of the AUC study was that this behavior was due to the formation of protein associates.
[0528] The effect of the above listed excipient on this behavior was studied and the results are shown in FIG. 35. The Z-Average was measure before and after thermal stress. The stabilizing effect was similar to all tested excipients, but the increase in Z-average was generally reduced by using amino acids as stabilizers (Arginine, Lysine or Glycine). Lysine was excluded due to higher content of aggregates after stress. Arginine showed a better effect than Glycine. Both amino acids were considered for the final design of experiment in order to choose the best excipient combination.
Mechanical Stress
[0529] Lysine formulations were excluded as well as all formulations containing mannitol. SEC data showed no effect of the stress on the tested samples. See Table 106.
TABLE-US-00124 TABLE 106 Mechanical stress SEC before SEC after mechanical stress mechanical stress Formulation composition HMW monomer HMW monomer (mg/mL) Formulation No. [%] [%] [%] [%] Sucrose (60) + NaCl (2) 080_A 2.6 97.3 2.7 97.2 Trehalose (60) + NaCl (2) 080_B 2.7 97.2 2.6 97.3 Arginine (30) 080_C1 2.5 97.5 2.3 97.6 Arginine (20) + NaCl (2) 080_C2 2.5 97.4 2.5 97.4 Glycine (20) 080_D1 2.5 97.5 2.4 97.5 Glycine (20) + NaCl (2) 080_D2 2.5 97.5 2.4 97.5
The same reduction in Z-average was noticed in the presence of amino acids. Sucrose had a better protective effect than trehalose against mechanical stress. Arginine and glycine performed better in combination with NaCl. See FIG. 36.
Differential Scanning Calorimetry (DSC) Screening
[0530] A screening study to determine the unfolding temperature of Lead CXCR5 Antibody was performed using Differential scanning calorimetry (DSC). Sucrose, trehalose, arginine, and glycine were screened.
[0531] The Tm results are listed in Table 107.
TABLE-US-00125 TABLE 107 Effect of different excipients on the Tm values of Lead CXCR5 Antibody. All formulations were in 10 mM citrate buffer at pH 6 Excipient screened Tm1 Tm2 Tm3 Sucrose + NaCl 65.3 73.6 83.8 Trehalose + NaCl 65.5 73.9 83.9 Arginine 63.8 72.2 82.6 Arginine + NaCl 64.3 72.8 82.6 Glycine 64.8 74.1 84.2 Glycine + NaCl 64.9 73.6 83.8
Based on Tm1, sucrose and trehalose showed the highest values. Arginine performed better in combination with NaCl.
[0532] In conclusion, the data collected suggests that the final Lead CXCR5 Antibody 100 mg/ml formulation would contain a combination of a sugar (in some embodiments, sucrose) and an amino acid (in some embodiments, arginine or glycine) in the presence of NaCl as the isotonant.
Example 18--Surfactant Screening
[0533] Polysorbate as a stabilizer was evaluated for protection of Lead CXCR5 Antibody against both thermal and mechanical stresses.
[0534] Polysorbate 20 and 80 were tested in two different concentrations: 0.1 and 0.2 mg/ml.
Thermal Stress
[0535] DLS showed no effect by the addition of Polysorbate after thermal stress. The formation of HMWs and fragments after 7 days storage at 40.degree. C. was noticed in all samples, as detected by SEC. No additional bands in SDS-PAGE were detected. Slight changes were seen after thermal stress, but no differences between PS20 and PS80, as well as between the 2 concentrations, were seen (data not shown).
Mechanical Stress
[0536] DLS showed no changes after mechanical stress. Polysorbate 20 showed no aggregations after mechanical stress. Polysorbate 80 showed aggregates formation after mechanical stress. No additional bands in SDS-PAGE (data not shown) were seen.
[0537] In conclusion, Polysorbate 20 was the desired surfactant due to superiority in mechanical stabilization of the Lead CXCR5 Antibody.
Example 19--Prototype Formulation Pre-Selection Using DSC
[0538] Based on the excipient screening and the surfactant screening studies, 12 different excipient combinations were suggested (see Tables 108 and 109))
[0539] The unfolding temperature for all formulations was determined using DSC and the resulting Tms, as well as the osmolality for each formulation, are listed in Tables 108 and 109.
TABLE-US-00126 TABLE 108 Excipient combinations for prototype formulations (Arginine) pre-selection study using DSC. Tm values and osmolality are listed as well Composition mg/mL DSC Osmo. Formulation Sucrose Arginine NaCl PS 20 Tm1 Tm2 Tm3 (mosmol/kg) LA_10_087_A 60 20 2 0.1 65.2 73.3 83.2 495 LA_10_087_C 60 20 2 0.2 65.1 73.0 83.2 486 LA_10_087_E 30 10 2 0.1 64.5 72.8 83.0 304 LA_10_087_G 30 10 2 0.2 64.4 72.7 83.0 304 LA_10_087_L 45 10 2 0.1 64.7 73 83.2 349 LA_10_087_M 45 10 2 0.2 64.6 72.8 83.1 357
TABLE-US-00127 TABLE 109 Excipient combinations for prototype formulations (glycine) pre-selection study using DSC. Tm values and osmolality are listed as well Composition mg/mL DSC Osmo. Formulation Sucrose Glycine NaCl PS 20 Tm1 Tm2 Tm3 (mosmol/kg) LA_10_087_B 60 15 2 0.1 66.2 74.1 84.3 539 LA_10_087_D 60 15 2 0.2 65.8 74.1 84.3 533 LA_10_087_F 30 7.5 2 0.1 65.0 73.3 83.5 330 LA_10_087_H 30 7.5 2 0.2 64.8 73.1 83.4 320 LA_10_090_A 45 7.5 2 0.1 65.3 73.7 83.6 408 LA_10_090_B 45 7.5 2 0.2 65.3 73.7 83.9 391
The formulations didn't show great differences in Tm, but the osmolality varied a lot. The pre-selection of the prototype formulations were made based on Tm and osmolality. Accordingly, in each excipient group (arginine and glycine), the highest Tm was selected (regardless of the osmolality). In addition, the highest Tm in the isotonic region was also selected.
Example 20--Prototype Exploratory Stability Study
[0540] The above prototype selection resulted in 4 prototype formulations, which are listed in Table 110. Those formulations were tested for mechanical stress (100 rpm for 5 hours), 5 freeze/thaw cycles and isothermal stress at 5, 20, and 40.degree. C.
TABLE-US-00128 TABLE 110 Prototype formulations for the 100 mg/mL Lead CXCR5 Antibody formulation Composition Osmo. Formulation Sucrose Arginine Glycine NaCl PS 20 (mosmol/kg) LA_10_102_A 60 20 2 0.1 518 LA_10_102_B 45 10 2 0.1 374 LA_10_102_C 60 15 2 0.1 550 LA_10_102_D 30 7.5 2 0.1 325
Mechanical Stability
[0541] Lead CXCR5 Antibody in 10 mM citrate buffer at pH 6, without addition of any excipients (DS formulation), was also stressed in parallel with the prototype formulations. A higher molecular weight species was measured by DLS after mechanical stress of DS (FIG. 37), stress where no changes have been seen in all tested formulations after mechanical stress. The formation of aggregates after mechanical stress was measured using size exclusion chromatography (SEC) and the results are shown in Table 111. In general, the 4 formulations were equally stable to mechanical stress except formulation A, where more HMWs were found by SEC after mechanical stress. See FIG. 38.
TABLE-US-00129 TABLE 111 Size exclusion chromatography (SEC) results of the prototype formulations before and after mechanical stress SEC For_A For_B For_C For_D % Mon. % Mon. % Mon. % Mon. -20.degree. C. Before 99.6 99.5 99.5 99.5 After 98.1 99.5 99.3 99.4
Freeze/Thaw Stability
[0542] No significant differences were detected, either on DS or DP, after 5 freeze/thawing cycles. Therefore, there should be no instability issues by freezing and thawing (data are not shown).
Exploratory Prototype Stability Study
[0543] The prototype formulations were stored at -20, 5, 20, and 40.degree. C. They were analyzed at the start of the study, after 1 month, after 3 months, and after 6 months. The formulations were selected based on the 3 months results (Tables 112-114). The results showed that formulation B performed the best with regard to SEC, WCX, and sub-visible particles, especially at 40.degree. C.
TABLE-US-00130 TABLE 112 Size exclusion chromatography (SEC) results of the prototype formulations after 3 months SEC For_A For_B For_C For_D % Mon. % Mon. % Mon. % Mon. -20.degree. C. T0 99.6 99.5 99.5 99.5 T1 month N/A N/A N/A N/A T3 months 99.1 99.1 99 99.5 5.degree. C. T0 99.6 99.5 99.5 99.5 T1 month 99.3 99.4 99.5 99.5 T3 months 99 99.4 98.8 99.4 20.degree. C. T0 99.6 99.5 99.5 99.5 T1 month 99.5 99.5 99.4 99.4 T3 months 99 98.9 98.6 99.1 40.degree. C. T0 99.6 99.5 99.5 99.5 T1 month 96.9 96.7 96.5 96.3 T3 months 91.5 91.6 89.5 90.2
TABLE-US-00131 TABLE 113 Weak Cationic exchange chromatography (WCX) results of the prototype formulations after 3 months WCX For_A For_B For_C For_D % Basic % Basic % Basic % Basic -20.degree. C. T0 2 2.2 2.1 2.2 T1 month N/A N/A N/A N/A T3 months 1.5 1.6 1.6 1.6 5.degree. C. T0 2 2.2 2.1 2.2 T1 month 1.1 1.2 1.2 1.3 T3 months 1.5 1.6 1.7 1.6 20.degree. C. T0 2 2.2 2.1 2.2 T1 month 1.3 1.3 1.4 1.3 T3 months 1.7 1.9 2 2 40.degree. C. T0 2 2.2 2.1 2.2 T1 month 2.2 2.2 2.7 2.5 T3 months 6.5 5.1 8.6 8.2
TABLE-US-00132 TABLE 114 Sub-visible particles measured by Light blockage at T zero and after 3 months (5.degree. C.) For_A For_B For_C For_D >10 .mu.m >25 .mu.m >10 .mu.m >25 .mu.m >10 .mu.m >25 .mu.m >10 .mu.m >25 .mu.m T0 4 3 4 4 5 4 4 3 T2 8 1 5 1 34 14 6 2
[0544] In conclusion, the studies showed better results for the formulation LA_10_102_B. This formulated had a concentration of 100 mg/mL Lead CXCR5 Antibody in 10 mM citrate buffer at pH 6 and contained the following excipients:
TABLE-US-00133 Sucrose 45 mg/mL (4.5%); Arginine 10 mg/mL (1%); NaCl 2 mg/mL (0.2%); and Polysorbate 20 0.1 mg/mL (0.01%).
Example 21--Supporting Stability Data for the 100 mg/mL Formulation
[0545] Additional stability studies were done on the 100 mg/mL Lead CXCR5 Antibody formulation identified in Example 20. The additional studies were performed at -20, 5, and 25.degree. C. The results are shown in Tables 115-117.
TABLE-US-00134 TABLE 115 Stability Data for 100 mg/mL Lead CXCR5 Antibody formulation at -20.degree. C. Drug product: Lead CXCR5 Antibody- Batch no.: 11_106/LST0008 solution for injection Dosage strength: 100 mg/mL Manufacturer batch no.: 11_106 Storage condition: -20.degree. C. .+-. 5.degree. C. Storage orientation: Inverted Time Initial 1 3 6 9 12 18 24 Test item results month months months months months months months Appearance of solution Clarity I <I <I <I II <IV <IV (20 NTU) (19 NTU) Color Y7 Y7 Y7 Y7 Y7 Y6 Y6 Assay Potency (Antigen ELISA) EC.sub.50 value (in 75% 107% 84% 96% 101% 96% 127% comparison to reference) Total protein 103 mg/mL 101 mg/mL 101 mg/mL 101 mg/mL 102 mg/mL 101 mg/mL 102 mg/mL content (UV) Molecular integrity SDS-PAGE under Conforms Conforms Conforms Conforms Conforms Conforms Conforms non-reducing to to to to to to to conditions (Band reference reference reference reference reference reference reference pattern) Purity HPLC (SEC) Monomer (% area) 99.1% 99.0% 99.0% 98.8% 98.9% 98.8% 98.9% High molecular 0.8% 0.8% 0.8% 0.8% 0.8% 0.8% 0.8% weight proteins (% area) SDS-PAGE under <1.0% <1.0% <1.0% <1.0% <1.0% <1.0% <1.0% non-reducing conditions Half molecules (%) SDS-PAGE under 99% 97% 99% 97% 99% 98% 97% reducing conditions Relative purity (%) Charge heterogeneity HPLC (weak cation 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/95%/2% exchange) Isoforms (acidic/ neutral/basic) (% area) IEF Conforms Conforms Conforms Conforms Conforms Conforms Conforms to to to to to to to reference reference reference reference reference reference reference pH (potentiometry) 5.9 5.8 6.0 5.9 5.9 5.9 5.9 Particulate matter Practically Complies Complies Complies Complies Complies Complies (visible particles) free from particles Particulate matter (subvisible particles) Number of 2 7 particles per vial .gtoreq. 10 .mu.m Number of 0 1 particles per vial .gtoreq. 25 .mu.m Microbial <1 cfu/2 mL contamination Closure integrity No trace No trace of of coloration coloration visible visible Dynamic light z- z- z- z- z- z- z- scattering average: average: average: average: average: average: average: 8.1 r.nm 8.0 r.nm 8.0 r.nm 8.1 r.nm 8.1 r.nm 8.1 r.nm 8.1 r.nm Pdl: 0.05 Pdl: 0.05 Pdl: 0.05 Pdl: 0.05 Pdl: 0.05 Pdl: 0.07 Pdl: 0.05
TABLE-US-00135 TABLE 116 Stability Data for 100 mg/mL Lead CXCR5 Antibody formulation at 5.degree. C. Drug product: Lead CXCR5 Antibody - Batch no.: 11_106/LST0008 solution for injection Dosage strength: 100 mg/mL Manufacturer batch no.: 11/106 Storage condition: +5.degree. C. .+-. 3.degree. C. Storage orientation: Inverted Time Initial 1 3 6 9 12 15 18 24 Test item results month months months months months months months months Appearance of solution Clarity I <I <I <I I <IV <IV <IV (22 NTU) (22 NTU) (21 NTU) Color Y7 Y7 Y7 Y7 Y7 Y6 Y6 Y6 Assay Potency (Antigen ELISA) EC.sub.50 value (in 75% 105% 95% 97% 92% 93% 114% 119% comparison to reference) Total protein 103 mg/mL 101 mg/mL 102 mg/mL 101 mg/mL 102 mg/mL 102 mg/mL 101 mg/mL 102 mg/mL content (UV) Molecular integrity SDS-PAGE under Conforms Conferms Conforms Conforms Conforms Conforms Conforms Conforms non-reducing to to to to to to to to conditions (Band reference reference reference reference reference reference reference reference pattern) Purity HPLC (SEC) Monomer (% area) 99.1% 99.0% 99.1% 98.8% 98.8% 98.7% 98.9% 98.8% High molecular 0.8% 0.7% 0.7% 0.7% 0.8% 0.8% 0.9% 0.9% weight proteins (% area) SDS-PAGE under <1.0% <1.0% <1.0% <1.0% <1.0% <1.0% <1.0% <1.0% non-reducing conditions Half molecules (%) SDS-PAGE under 99% 98% 99% 95% 99% 97% 99% 98% reducing conditions Relative purity (%) Charge heterogeneity HPLC (weak cation 4%/94%/2% 4%/94%/2% 3%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% 4%/94%/2% exchange) Isoforms (acidic/ neutral/basic) (% area) IEF Conforms Conferms Conforms Conforms Conforms Conforms Conforms Conforms to to to to to to to to reference reference reference reference reference reference reference reference pH (potentiometry) 5.9 5.9 6.0 5.9 5.9 5.9 5.9 5.9 Particulate matter Complies Complies Complies Complies Complies Complies Complies Complies (visible particles) Particulate matter (subvisible particles) Number of 2 14 2 16 particles per vial .gtoreq. 10 .mu.m Number of 0 2 0 0 particles per vial .gtoreq. 25 .mu.m Microbial <1 cfu/2 mL contamination Closure integrity No trace No trace of of coloration coloration visible visible Dynamic light z- z- z- z- z- z- z- z- scattering average: average: average: average: average: average: average: average: 8.1 r.nm 8.0 r.nm 7.9 r.nm 8.0 r.nm 8.1 r.nm 8.1 r.nm 8.0 r.nm 8.1 r.nm Pdl: 0.05 Pdl: 0.05 Pdl: 0.04 Pdl: 0.04 Pdl: 0.06 Pdl: 0.06 Pdl: 0.05 Pdl: 0.05
TABLE-US-00136 TABLE 117 Stability Data for 100 mg/mL Lead CXCR5 Antibody formulation at 25.degree. C. Drug product: Lead CXCR5 Antibody-solution for injection Batch no.: 11_106/LST0008 Dosage strength: 100 mg/mL Manufacturer batch no.: 11_106 Storage condition: +25.degree. C. .+-. 2.degree. C./60% .+-. 5% RH Storage Inverted orientation: Test item Time Initial 1 3 6 results month months months Appearance of solution Clarity I <I <I <I Color Y7 Y7 Y7 Y7 Assay Potency (Antigen ELISA) EC.sub.50 value (in comparison 75% 121% 96% 104% to reference) Total protein content (UV) 103 mg/mL 101 mg/mL 102 mg/mL 102 mg/mL Molecular integrity SDS-PAGE under non- Conforms to Conforms to Conforms to Conforms to reducing conditions (Band reference reference reference reference pattern) Purity HPLC (SEC) Monomer (% area) 99.1%.sup. 98.9% 98.8%.sup. 98.2% High molecular weight 0.8% 0.8% 1.0% 1.2% proteins (% area) SDS-PAGE under non- <1.0%.sup. <1.0% <1.0%.sup. <1.0% reducing conditions Half molecules (%) 99% 96% 99% 96% SDS-PAGE under reducing conditions Relative purity (%) Charge heterogeneity HPLC (weak cation 4%/94%/2% 4%/94%/2% 4%/94%/3% 4%/93%/3% exchange) Isoforms (acidic/neutral/basic) (% area) IEF Conforms to Conforms to Conforms to Conforms to reference reference reference reference pH (potentiometry) 5.9 5.9 6.0 6.1 Particulate matter (visible Complies Complies Complies Complies particles) Particulate matter (subvisible particles) Number of particles 2 17 per vial .gtoreq.10 .mu.m Number of particles 0 1 per vial .gtoreq.25 .mu.m Microbial contamination <1 cfu/2 mL <1 cfu/2 mL Closure integrity No trace of No trace of coloration visible coloration visible Dynamic light scattering z-average: 8.1 z-average: 8.0 z-average: 8.1 z-average: 8.1 r.nm; Pdl: 0.05 r.nm Pdl: 0.05 r.nm Pdl: 0.05 r.nm Pdl: 0.06
Sequence CWU 1
1
2915PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 1Gly Tyr Asn Trp His1 5216PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 2Glu
Ile Thr His Ser Gly Ser Thr Asn Tyr Asn Pro Ser Leu Lys Ser1 5 10
15314PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 3Glu Ile Ala Val Ala Gly Thr Gly Tyr Tyr Gly Met
Asp Val1 5 10411PRTArtificial SequenceDescription of Artificial
Sequence Synthetic peptide 4Arg Ala Ser Gln Gly Ile Asn Ser Ala Phe
Ala1 5 1057PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 5Asp Ala Ser Ser Leu Glu Ser1 569PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 6Gln
Gln Phe Asn Ser Tyr Pro Leu Thr1 57448PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
7Gln Val Gln Leu Gln Gln Trp Gly Ala Gly Leu Leu Lys Pro Ser Glu1 5
10 15Thr Leu Ser Leu Thr Cys Ala Val Tyr Gly Gly Ser Phe Ser Gly
Tyr 20 25 30Asn Trp His Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu Glu
Trp Ile 35 40 45Gly Glu Ile Thr His Ser Gly Ser Thr Asn Tyr Asn Pro
Ser Leu Lys 50 55 60Ser Arg Val Thr Ile Ser Val Asp Thr Ser Lys Asn
Gln Phe Ser Leu65 70 75 80Lys Leu Ser Ser Val Thr Ala Ala Asp Thr
Ala Val Tyr Tyr Cys Val 85 90 95Arg Glu Ile Ala Val Ala Gly Thr Gly
Tyr Tyr Gly Met Asp Val Trp 100 105 110Gly Gln Gly Thr Thr Val Thr
Val Ser Ser Ala Ser Thr Lys Gly Pro 115 120 125Ser Val Phe Pro Leu
Ala Pro Cys Ser Arg Ser Thr Ser Glu Ser Thr 130 135 140Ala Ala Leu
Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro Val Thr145 150 155
160Val Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val His Thr Phe Pro
165 170 175Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val
Val Thr 180 185 190Val Pro Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr
Cys Asn Val Asp 195 200 205His Lys Pro Ser Asn Thr Lys Val Asp Lys
Arg Val Glu Ser Lys Tyr 210 215 220Gly Pro Pro Cys Pro Pro Cys Pro
Ala Pro Glu Phe Glu Gly Gly Pro225 230 235 240Ser Val Phe Leu Phe
Pro Pro Lys Pro Lys Asp Thr Leu Met Ile Ser 245 250 255Arg Thr Pro
Glu Val Thr Cys Val Val Val Asp Val Ser Gln Glu Asp 260 265 270Pro
Glu Val Gln Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn 275 280
285Ala Lys Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr Tyr Arg Val
290 295 300Val Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly
Lys Glu305 310 315 320Tyr Lys Cys Lys Val Ser Asn Lys Gly Leu Pro
Ser Ser Ile Glu Lys 325 330 335Thr Ile Ser Lys Ala Lys Gly Gln Pro
Arg Glu Pro Gln Val Tyr Thr 340 345 350Leu Pro Pro Ser Gln Glu Glu
Met Thr Lys Asn Gln Val Ser Leu Thr 355 360 365Cys Leu Val Lys Gly
Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu 370 375 380Ser Asn Gly
Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu385 390 395
400Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser Arg Leu Thr Val Asp Lys
405 410 415Ser Arg Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val Met
His Glu 420 425 430Ala Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser
Leu Ser Leu Gly 435 440 4458214PRTArtificial SequenceDescription of
Artificial Sequence Synthetic polypeptide 8Ala Ile Gln Leu Thr Gln
Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile
Thr Cys Arg Ala Ser Gln Gly Ile Asn Ser Ala 20 25 30Phe Ala Trp Tyr
Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Asp Ala
Ser Ser Leu Glu Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly
Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75
80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Phe Asn Ser Tyr Pro Leu
85 90 95Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys Arg Thr Val Ala
Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu
Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu Ser Val Thr Glu Gln
Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr
Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr 180 185 190Ala Cys
Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser 195 200
205Phe Asn Arg Gly Glu Cys 2109240PRTHomo sapiens 9Met Glu Glu Ser
Val Val Arg Pro Ser Val Phe Val Val Asp Gly Gln1 5 10 15Thr Asp Ile
Pro Phe Thr Arg Leu Gly Arg Ser His Arg Arg Gln Ser 20 25 30Cys Ser
Val Ala Arg Val Gly Leu Gly Leu Leu Leu Leu Leu Met Gly 35 40 45Ala
Gly Leu Ala Val Gln Gly Trp Phe Leu Leu Gln Leu His Trp Arg 50 55
60Leu Gly Glu Met Val Thr Arg Leu Pro Asp Gly Pro Ala Gly Ser Trp65
70 75 80Glu Gln Leu Thr Gln Glu Arg Arg Ser His Glu Val Asn Pro Ala
Ala 85 90 95His Leu Thr Gly Ala Asn Ser Ser Leu Thr Gly Ser Gly Gly
Pro Leu 100 105 110Leu Trp Glu Thr Gln Leu Gly Leu Ala Phe Leu Arg
Gly Leu Ser Tyr 115 120 125His Asp Gly Ala Leu Val Val Thr Lys Ala
Gly Tyr Tyr Tyr Ile Tyr 130 135 140Ser Lys Val Gln Leu Gly Gly Val
Gly Cys Pro Leu Gly Leu Ala Ser145 150 155 160Thr Ile Thr His Gly
Leu Tyr Lys Arg Thr Pro Arg Tyr Pro Glu Glu 165 170 175Leu Glu Leu
Leu Val Ser Gln Gln Ser Pro Cys Gly Arg Ala Thr Ser 180 185 190Ser
Ser Arg Val Trp Trp Asp Ser Ser Phe Leu Gly Gly Val Val His 195 200
205Leu Glu Ala Gly Glu Glu Val Val Val Arg Val Leu Asp Glu Arg Leu
210 215 220Val Arg Leu Arg Asp Gly Thr Arg Ser Tyr Phe Gly Ala Phe
Met Val225 230 235 24010467PRTArtificial SequenceDescription of
Artificial Sequence Synthetic polypeptide 10Met Lys His Leu Trp Phe
Phe Leu Leu Leu Val Ala Ala Pro Arg Trp1 5 10 15Val Leu Ser Gln Val
Gln Leu Gln Gln Trp Gly Ala Gly Leu Leu Lys 20 25 30Pro Ser Glu Thr
Leu Ser Leu Thr Cys Ala Val Tyr Gly Gly Ser Phe 35 40 45Ser Gly Tyr
Asn Trp His Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu 50 55 60Glu Trp
Ile Gly Glu Ile Thr His Ser Gly Ser Thr Asn Tyr Asn Pro65 70 75
80Ser Leu Lys Ser Arg Val Thr Ile Ser Val Asp Thr Ser Lys Asn Gln
85 90 95Phe Ser Leu Lys Leu Ser Ser Val Thr Ala Ala Asp Thr Ala Val
Tyr 100 105 110Tyr Cys Val Arg Glu Ile Ala Val Ala Gly Thr Gly Tyr
Tyr Gly Met 115 120 125Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val
Ser Ser Ala Ser Thr 130 135 140Lys Gly Pro Ser Val Phe Pro Leu Ala
Pro Cys Ser Arg Ser Thr Ser145 150 155 160Glu Ser Thr Ala Ala Leu
Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu 165 170 175Pro Val Thr Val
Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val His 180 185 190Thr Phe
Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser 195 200
205Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr Cys
210 215 220Asn Val Asp His Lys Pro Ser Asn Thr Lys Val Asp Lys Arg
Val Glu225 230 235 240Ser Lys Tyr Gly Pro Pro Cys Pro Pro Cys Pro
Ala Pro Glu Phe Glu 245 250 255Gly Gly Pro Ser Val Phe Leu Phe Pro
Pro Lys Pro Lys Asp Thr Leu 260 265 270Met Ile Ser Arg Thr Pro Glu
Val Thr Cys Val Val Val Asp Val Ser 275 280 285Gln Glu Asp Pro Glu
Val Gln Phe Asn Trp Tyr Val Asp Gly Val Glu 290 295 300Val His Asn
Ala Lys Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr305 310 315
320Tyr Arg Val Val Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn
325 330 335Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys Gly Leu Pro
Ser Ser 340 345 350Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro
Arg Glu Pro Gln 355 360 365Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu
Met Thr Lys Asn Gln Val 370 375 380Ser Leu Thr Cys Leu Val Lys Gly
Phe Tyr Pro Ser Asp Ile Ala Val385 390 395 400Glu Trp Glu Ser Asn
Gly Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro 405 410 415Pro Val Leu
Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser Arg Leu Thr 420 425 430Val
Asp Lys Ser Arg Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val 435 440
445Met His Glu Ala Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu
450 455 460Ser Leu Gly465111404DNAArtificial SequenceDescription of
Artificial Sequence Synthetic polynucleotide 11atgaagcacc
tgtggttctt tctgctgctg gtggccgctc ctagatgggt gctgtcccag 60gtgcagctgc
agcagtgggg cgctggcctg ctgaagcctt ccgagacact gtccctgacc
120tgcgccgtgt acggcggctc cttctccggc tacaactggc actggatcag
gcagcctccc 180ggcaagggcc tggaatggat cggcgagatc acccactccg
gctccaccaa ctacaaccct 240agcctgaagt ccagagtgac catctccgtg
gacacctcca agaaccagtt ctccctgaag 300ctgtcctctg tgaccgccgc
tgacaccgcc gtgtactact gtgtgcggga gatcgccgtg 360gctggcaccg
gctactacgg catggatgtg tggggccagg gcaccaccgt gaccgtgtcc
420agcgcttcta ccaagggccc ttccgtgttc cctctggccc cttgctcccg
gtccacctcc 480gagtccaccg ccgctctggg ctgcctggtg aaggactact
tccctgagcc tgtgaccgtg 540tcctggaact ctggcgccct gacctccggc
gtgcacacct tccctgccgt gctgcagtcc 600tccggcctgt actccctgtc
ctccgtggtg accgtgcctt cctcctccct gggcaccaag 660acctacacct
gtaacgtgga ccacaagcct tccaacacca aggtggacaa gcgggtggag
720tccaagtacg gccctccttg ccctccctgc cctgcccctg agttcgaggg
cggacctagc 780gtgttcctgt tccctcctaa gcctaaggac accctgatga
tctcccggac ccctgaggtg 840acctgtgtgg tggtggacgt gtcccaggag
gaccctgagg tccagttcaa ctggtacgtg 900gacggcgtgg aggtgcacaa
cgccaagacc aagcctcggg aggagcagtt caattccacc 960taccgggtgg
tgtctgtgct gaccgtgctg caccaggact ggctgaacgg caaagaatac
1020aagtgtaagg tctccaacaa gggcctgccc tcctccatcg agaaaaccat
ctccaaggcc 1080aagggccagc ctagggagcc tcaggtgtac accctgcctc
ctagccagga agagatgacc 1140aagaaccagg tgtccctgac ctgtctggtg
aagggcttct acccttccga catcgccgtg 1200gagtgggagt ccaacggcca
gcctgagaac aactacaaga ccacccctcc tgtgctggac 1260tccgacggct
ccttcttcct gtactccagg ctgaccgtgg acaagtcccg gtggcaggag
1320ggcaacgtct tttcctgctc cgtgatgcac gaggccctgc acaaccacta
cacccagaag 1380tccctgtccc tgtctctggg ctga 140412236PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
12Met Asp Met Arg Val Pro Ala Gln Leu Leu Gly Leu Leu Leu Leu Trp1
5 10 15Leu Pro Gly Ala Arg Cys Ala Ile Gln Leu Thr Gln Ser Pro Ser
Ser 20 25 30Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser 35 40 45Gln Gly Ile Asn Ser Ala Phe Ala Trp Tyr Gln Gln Lys
Pro Gly Lys 50 55 60Ala Pro Lys Leu Leu Ile Tyr Asp Ala Ser Ser Leu
Glu Ser Gly Val65 70 75 80Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly
Thr Asp Phe Thr Leu Thr 85 90 95Ile Ser Ser Leu Gln Pro Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln 100 105 110Phe Asn Ser Tyr Pro Leu Thr
Phe Gly Gly Gly Thr Lys Val Glu Ile 115 120 125Lys Arg Thr Val Ala
Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp 130 135 140Glu Gln Leu
Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn145 150 155
160Phe Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu
165 170 175Gln Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser
Lys Asp 180 185 190Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser
Lys Ala Asp Tyr 195 200 205Glu Lys His Lys Val Tyr Ala Cys Glu Val
Thr His Gln Gly Leu Ser 210 215 220Ser Pro Val Thr Lys Ser Phe Asn
Arg Gly Glu Cys225 230 23513711DNAArtificial SequenceDescription of
Artificial Sequence Synthetic polynucleotide 13atggacatga
gagtgcctgc tcagctgctg ggactgctgc tgctgtggct gcctggcgct 60agatgcgcca
tccagctgac ccagtccccc tcctctctgt ccgcctccgt gggcgacaga
120gtgaccatca cctgtcgggc ctcccagggc atcaactccg ccttcgcctg
gtatcagcag 180aagcctggca aggcccctaa gctgctgatc tacgacgcct
cctccctgga atccggcgtg 240ccctccagat tttccggctc cggctctggc
accgacttca ccctgaccat ctccagcctg 300cagcctgagg acttcgccac
ctactactgc cagcagttca actcctaccc tctgaccttc 360ggcggaggca
ccaaggtgga gatcaagcgt acggtggctg caccatctgt cttcatcttc
420ccgccatctg atgagcagtt gaaatctgga actgcctctg ttgtgtgcct
gctgaataac 480ttctatccca gagaggccaa agtacagtgg aaggtggata
acgccctcca atcgggtaac 540tcccaggaga gtgtcacaga gcaggacagc
aaggacagca cctacagcct cagcagcacc 600ctgacgctga gcaaagcaga
ctacgagaaa cacaaagtct acgcctgcga agtcacccat 660cagggcctga
gctcgcccgt cacaaagagc ttcaacaggg gagagtgtta g 71114372PRTHomo
sapiens 14Met Asn Tyr Pro Leu Thr Leu Glu Met Asp Leu Glu Asn Leu
Glu Asp1 5 10 15Leu Phe Trp Glu Leu Asp Arg Leu Asp Asn Tyr Asn Asp
Thr Ser Leu 20 25 30Val Glu Asn His Leu Cys Pro Ala Thr Glu Gly Pro
Leu Met Ala Ser 35 40 45Phe Lys Ala Val Phe Val Pro Val Ala Tyr Ser
Leu Ile Phe Leu Leu 50 55 60Gly Val Ile Gly Asn Val Leu Val Leu Val
Ile Leu Glu Arg His Arg65 70 75 80Gln Thr Arg Ser Ser Thr Glu Thr
Phe Leu Phe His Leu Ala Val Ala 85 90 95Asp Leu Leu Leu Val Phe Ile
Leu Pro Phe Ala Val Ala Glu Gly Ser 100 105 110Val Gly Trp Val Leu
Gly Thr Phe Leu Cys Lys Thr Val Ile Ala Leu 115 120 125His Lys Val
Asn Phe Tyr Cys Ser Ser Leu Leu Leu Ala Cys Ile Ala 130 135 140Val
Asp Arg Tyr Leu Ala Ile Val His Ala Val His Ala Tyr Arg His145 150
155 160Arg Arg Leu Leu Ser Ile His Ile Thr Cys Gly Thr Ile Trp Leu
Val 165 170 175Gly Phe Leu Leu Ala Leu Pro Glu Ile Leu Phe Ala Lys
Val Ser Gln 180 185 190Gly His His Asn Asn Ser Leu Pro Arg Cys Thr
Phe Ser Gln Glu Asn 195 200 205Gln Ala Glu Thr His Ala Trp Phe Thr
Ser Arg Phe Leu Tyr His Val 210 215 220Ala Gly Phe Leu Leu Pro Met
Leu Val Met Gly Trp Cys Tyr Val Gly225 230 235 240Val Val His Arg
Leu Arg Gln Ala Gln Arg Arg Pro Gln Arg Gln Lys 245 250 255Ala Val
Arg Val Ala Ile Leu Val Thr Ser Ile Phe Phe Leu Cys Trp 260 265
270Ser Pro Tyr His Ile Val Ile Phe Leu Asp Thr Leu Ala Arg Leu Lys
275 280 285Ala Val Asp Asn Thr Cys Lys Leu Asn Gly Ser Leu Pro Val
Ala Ile 290 295
300Thr Met Cys Glu Phe Leu Gly Leu Ala His Cys Cys Leu Asn Pro
Met305 310 315 320Leu Tyr Thr Phe Ala Gly Val Lys Phe Arg Ser Asp
Leu Ser Arg Leu 325 330 335Leu Thr Lys Leu Gly Cys Thr Gly Pro Ala
Ser Leu Cys Gln Leu Phe 340 345 350Pro Ser Trp Arg Arg Ser Ser Leu
Ser Glu Ser Glu Asn Ala Thr Ser 355 360 365Leu Thr Thr Phe
3701510PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 15Gly Phe Ser Leu Ile Asp Tyr Gly Val Asn1 5
10169PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 16Val Ile Trp Gly Asp Gly Thr Thr Tyr1
5173PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 17Ile Val Tyr11816PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 18Arg
Ser Ser Lys Ser Leu Leu His Ser Ser Gly Lys Thr Tyr Leu Tyr1 5 10
15196PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 19Arg Leu Ser Ser Leu Ala1 5209PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 20Met
Gln His Leu Glu Tyr Pro Tyr Thr1 521111PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
21Gln Val Gln Leu Lys Glu Ser Gly Pro Gly Leu Val Ala Pro Ser Glu1
5 10 15Ser Leu Ser Ile Thr Cys Thr Val Ser Gly Phe Ser Leu Ile Asp
Tyr 20 25 30Gly Val Asn Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu Glu
Trp Leu 35 40 45Gly Val Ile Trp Gly Asp Gly Thr Thr Tyr Tyr Asn Pro
Ser Leu Lys 50 55 60Ser Arg Leu Ser Ile Ser Lys Asp Asn Ser Lys Ser
Gln Val Phe Leu65 70 75 80Lys Val Thr Ser Leu Thr Thr Asp Asp Thr
Ala Met Tyr Tyr Cys Ala 85 90 95Arg Ile Val Tyr Trp Gly Gln Gly Thr
Leu Val Thr Val Ser Ala 100 105 11022112PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
22Asp Ile Val Met Thr Gln Ala Ala Pro Ser Val Ala Val Thr Pro Gly1
5 10 15Ala Ser Val Ser Ile Ser Cys Arg Ser Ser Lys Ser Leu Leu His
Ser 20 25 30Ser Gly Lys Thr Tyr Leu Tyr Trp Phe Leu Gln Arg Pro Gly
Gln Ser 35 40 45Pro Gln Leu Leu Ile Tyr Arg Leu Ser Ser Leu Ala Ser
Gly Val Pro 50 55 60Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Ala Phe
Thr Leu Arg Ile65 70 75 80Ser Arg Val Glu Ala Glu Asp Val Gly Val
Tyr Tyr Cys Met Gln His 85 90 95Leu Glu Tyr Pro Tyr Thr Phe Gly Gly
Gly Thr Lys Leu Glu Ile Lys 100 105 11023326PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
23Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Cys Ser Arg1
5 10 15Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu Val Lys Asp
Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu
Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly
Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu
Gly Thr Lys Thr65 70 75 80Tyr Thr Cys Asn Val Asp His Lys Pro Ser
Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Ser Lys Tyr Gly Pro Pro
Cys Pro Pro Cys Pro Ala Pro 100 105 110Glu Phe Glu Gly Gly Pro Ser
Val Phe Leu Phe Pro Pro Lys Pro Lys 115 120 125Asp Thr Leu Met Ile
Ser Arg Thr Pro Glu Val Thr Cys Val Val Val 130 135 140Asp Val Ser
Gln Glu Asp Pro Glu Val Gln Phe Asn Trp Tyr Val Asp145 150 155
160Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu Glu Gln Phe
165 170 175Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu His
Gln Asp 180 185 190Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser
Asn Lys Gly Leu 195 200 205Pro Ser Ser Ile Glu Lys Thr Ile Ser Lys
Ala Lys Gly Gln Pro Arg 210 215 220Glu Pro Gln Val Tyr Thr Leu Pro
Pro Ser Gln Glu Glu Met Thr Lys225 230 235 240Asn Gln Val Ser Leu
Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp 245 250 255Ile Ala Val
Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys 260 265 270Thr
Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser 275 280
285Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu Gly Asn Val Phe Ser
290 295 300Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr Gln
Lys Ser305 310 315 320Leu Ser Leu Ser Leu Gly 32524107PRTArtificial
SequenceDescription of Artificial Sequence Synthetic polypeptide
24Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu1
5 10 15Gln Leu Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn
Phe 20 25 30Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln 35 40 45Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser
Lys Asp Ser 50 55 60Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys
Ala Asp Tyr Glu65 70 75 80Lys His Lys Val Tyr Ala Cys Glu Val Thr
His Gln Gly Leu Ser Ser 85 90 95Pro Val Thr Lys Ser Phe Asn Arg Gly
Glu Cys 100 10525437PRTArtificial SequenceDescription of Artificial
Sequence Synthetic polypeptide 25Gln Val Gln Leu Lys Glu Ser Gly
Pro Gly Leu Val Ala Pro Ser Glu1 5 10 15Ser Leu Ser Ile Thr Cys Thr
Val Ser Gly Phe Ser Leu Ile Asp Tyr 20 25 30Gly Val Asn Trp Ile Arg
Gln Pro Pro Gly Lys Gly Leu Glu Trp Leu 35 40 45Gly Val Ile Trp Gly
Asp Gly Thr Thr Tyr Tyr Asn Pro Ser Leu Lys 50 55 60Ser Arg Leu Ser
Ile Ser Lys Asp Asn Ser Lys Ser Gln Val Phe Leu65 70 75 80Lys Val
Thr Ser Leu Thr Thr Asp Asp Thr Ala Met Tyr Tyr Cys Ala 85 90 95Arg
Ile Val Tyr Trp Gly Gln Gly Thr Leu Val Thr Val Ser Ala Ala 100 105
110Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Cys Ser Arg Ser
115 120 125Thr Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu Val Lys Asp
Tyr Phe 130 135 140Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala
Leu Thr Ser Gly145 150 155 160Val His Thr Phe Pro Ala Val Leu Gln
Ser Ser Gly Leu Tyr Ser Leu 165 170 175Ser Ser Val Val Thr Val Pro
Ser Ser Ser Leu Gly Thr Lys Thr Tyr 180 185 190Thr Cys Asn Val Asp
His Lys Pro Ser Asn Thr Lys Val Asp Lys Arg 195 200 205Val Glu Ser
Lys Tyr Gly Pro Pro Cys Pro Pro Cys Pro Ala Pro Glu 210 215 220Phe
Glu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp225 230
235 240Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val
Asp 245 250 255Val Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp Tyr
Val Asp Gly 260 265 270Val Glu Val His Asn Ala Lys Thr Lys Pro Arg
Glu Glu Gln Phe Asn 275 280 285Ser Thr Tyr Arg Val Val Ser Val Leu
Thr Val Leu His Gln Asp Trp 290 295 300Leu Asn Gly Lys Glu Tyr Lys
Cys Lys Val Ser Asn Lys Gly Leu Pro305 310 315 320Ser Ser Ile Glu
Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu 325 330 335Pro Gln
Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu Met Thr Lys Asn 340 345
350Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile
355 360 365Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr
Lys Thr 370 375 380Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe
Leu Tyr Ser Arg385 390 395 400Leu Thr Val Asp Lys Ser Arg Trp Gln
Glu Gly Asn Val Phe Ser Cys 405 410 415Ser Val Met His Glu Ala Leu
His Asn His Tyr Thr Gln Lys Ser Leu 420 425 430Ser Leu Ser Leu Gly
43526219PRTArtificial SequenceDescription of Artificial Sequence
Synthetic polypeptide 26Asp Ile Val Met Thr Gln Ala Ala Pro Ser Val
Ala Val Thr Pro Gly1 5 10 15Ala Ser Val Ser Ile Ser Cys Arg Ser Ser
Lys Ser Leu Leu His Ser 20 25 30Ser Gly Lys Thr Tyr Leu Tyr Trp Phe
Leu Gln Arg Pro Gly Gln Ser 35 40 45Pro Gln Leu Leu Ile Tyr Arg Leu
Ser Ser Leu Ala Ser Gly Val Pro 50 55 60Asp Arg Phe Ser Gly Ser Gly
Ser Gly Thr Ala Phe Thr Leu Arg Ile65 70 75 80Ser Arg Val Glu Ala
Glu Asp Val Gly Val Tyr Tyr Cys Met Gln His 85 90 95Leu Glu Tyr Pro
Tyr Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 105 110Arg Thr
Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu 115 120
125Gln Leu Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
130 135 140Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln145 150 155 160Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln
Asp Ser Lys Asp Ser 165 170 175Thr Tyr Ser Leu Ser Ser Thr Leu Thr
Leu Ser Lys Ala Asp Tyr Glu 180 185 190Lys His Lys Val Tyr Ala Cys
Glu Val Thr His Gln Gly Leu Ser Ser 195 200 205Pro Val Thr Lys Ser
Phe Asn Arg Gly Glu Cys 210 2152719PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 27Met
Gly Trp Ser Cys Ile Ile Leu Phe Leu Val Ala Thr Ala Thr Gly1 5 10
15Val His Ser28456PRTArtificial SequenceDescription of Artificial
Sequence Synthetic polypeptide 28Met Gly Trp Ser Cys Ile Ile Leu
Phe Leu Val Ala Thr Ala Thr Gly1 5 10 15Val His Ser Gln Val Gln Leu
Lys Glu Ser Gly Pro Gly Leu Val Ala 20 25 30Pro Ser Glu Ser Leu Ser
Ile Thr Cys Thr Val Ser Gly Phe Ser Leu 35 40 45Ile Asp Tyr Gly Val
Asn Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu 50 55 60Glu Trp Leu Gly
Val Ile Trp Gly Asp Gly Thr Thr Tyr Tyr Asn Pro65 70 75 80Ser Leu
Lys Ser Arg Leu Ser Ile Ser Lys Asp Asn Ser Lys Ser Gln 85 90 95Val
Phe Leu Lys Val Thr Ser Leu Thr Thr Asp Asp Thr Ala Met Tyr 100 105
110Tyr Cys Ala Arg Ile Val Tyr Trp Gly Gln Gly Thr Leu Val Thr Val
115 120 125Ser Ala Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala
Pro Cys 130 135 140Ser Arg Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly
Cys Leu Val Lys145 150 155 160Asp Tyr Phe Pro Glu Pro Val Thr Val
Ser Trp Asn Ser Gly Ala Leu 165 170 175Thr Ser Gly Val His Thr Phe
Pro Ala Val Leu Gln Ser Ser Gly Leu 180 185 190Tyr Ser Leu Ser Ser
Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr 195 200 205Lys Thr Tyr
Thr Cys Asn Val Asp His Lys Pro Ser Asn Thr Lys Val 210 215 220Asp
Lys Arg Val Glu Ser Lys Tyr Gly Pro Pro Cys Pro Pro Cys Pro225 230
235 240Ala Pro Glu Phe Glu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
Lys 245 250 255Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val
Thr Cys Val 260 265 270Val Val Asp Val Ser Gln Glu Asp Pro Glu Val
Gln Phe Asn Trp Tyr 275 280 285Val Asp Gly Val Glu Val His Asn Ala
Lys Thr Lys Pro Arg Glu Glu 290 295 300Gln Phe Asn Ser Thr Tyr Arg
Val Val Ser Val Leu Thr Val Leu His305 310 315 320Gln Asp Trp Leu
Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys 325 330 335Gly Leu
Pro Ser Ser Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln 340 345
350Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu Met
355 360 365Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe
Tyr Pro 370 375 380Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln
Pro Glu Asn Asn385 390 395 400Tyr Lys Thr Thr Pro Pro Val Leu Asp
Ser Asp Gly Ser Phe Phe Leu 405 410 415Tyr Ser Arg Leu Thr Val Asp
Lys Ser Arg Trp Gln Glu Gly Asn Val 420 425 430Phe Ser Cys Ser Val
Met His Glu Ala Leu His Asn His Tyr Thr Gln 435 440 445Lys Ser Leu
Ser Leu Ser Leu Gly 450 45529238PRTArtificial SequenceDescription
of Artificial Sequence Synthetic polypeptide 29Met Gly Trp Ser Cys
Ile Ile Leu Phe Leu Val Ala Thr Ala Thr Gly1 5 10 15Val His Ser Asp
Ile Val Met Thr Gln Ala Ala Pro Ser Val Ala Val 20 25 30Thr Pro Gly
Ala Ser Val Ser Ile Ser Cys Arg Ser Ser Lys Ser Leu 35 40 45Leu His
Ser Ser Gly Lys Thr Tyr Leu Tyr Trp Phe Leu Gln Arg Pro 50 55 60Gly
Gln Ser Pro Gln Leu Leu Ile Tyr Arg Leu Ser Ser Leu Ala Ser65 70 75
80Gly Val Pro Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Ala Phe Thr
85 90 95Leu Arg Ile Ser Arg Val Glu Ala Glu Asp Val Gly Val Tyr Tyr
Cys 100 105 110Met Gln His Leu Glu Tyr Pro Tyr Thr Phe Gly Gly Gly
Thr Lys Leu 115 120 125Glu Ile Lys Arg Thr Val Ala Ala Pro Ser Val
Phe Ile Phe Pro Pro 130 135 140Ser Asp Glu Gln Leu Lys Ser Gly Thr
Ala Ser Val Val Cys Leu Leu145 150 155 160Asn Asn Phe Tyr Pro Arg
Glu Ala Lys Val Gln Trp Lys Val Asp Asn 165 170 175Ala Leu Gln Ser
Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser 180 185 190Lys Asp
Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala 195 200
205Asp Tyr Glu Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly
210 215 220Leu Ser Ser Pro Val Thr Lys Ser Phe Asn Arg Gly Glu
Cys225 230 235
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017
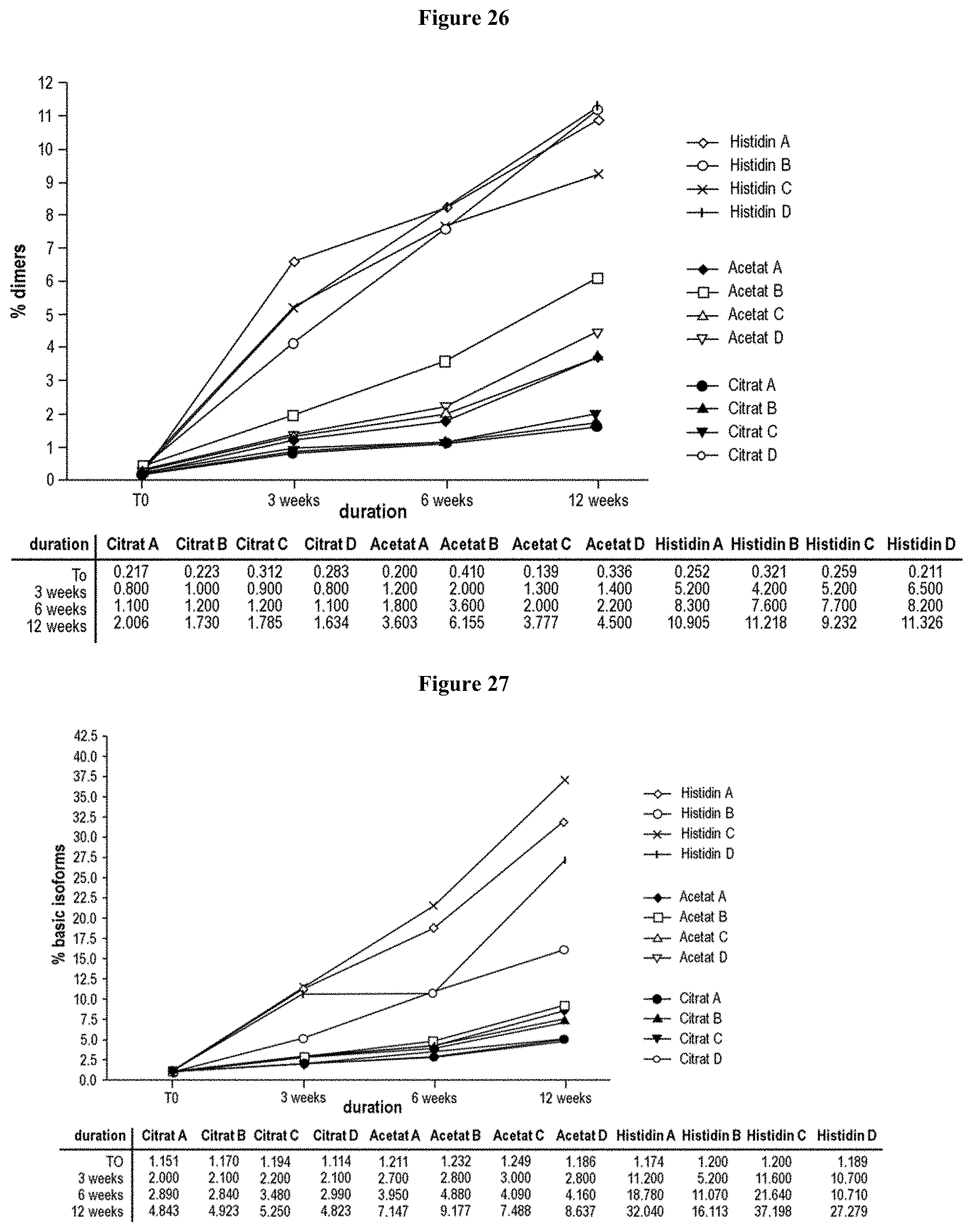
D00018

D00019
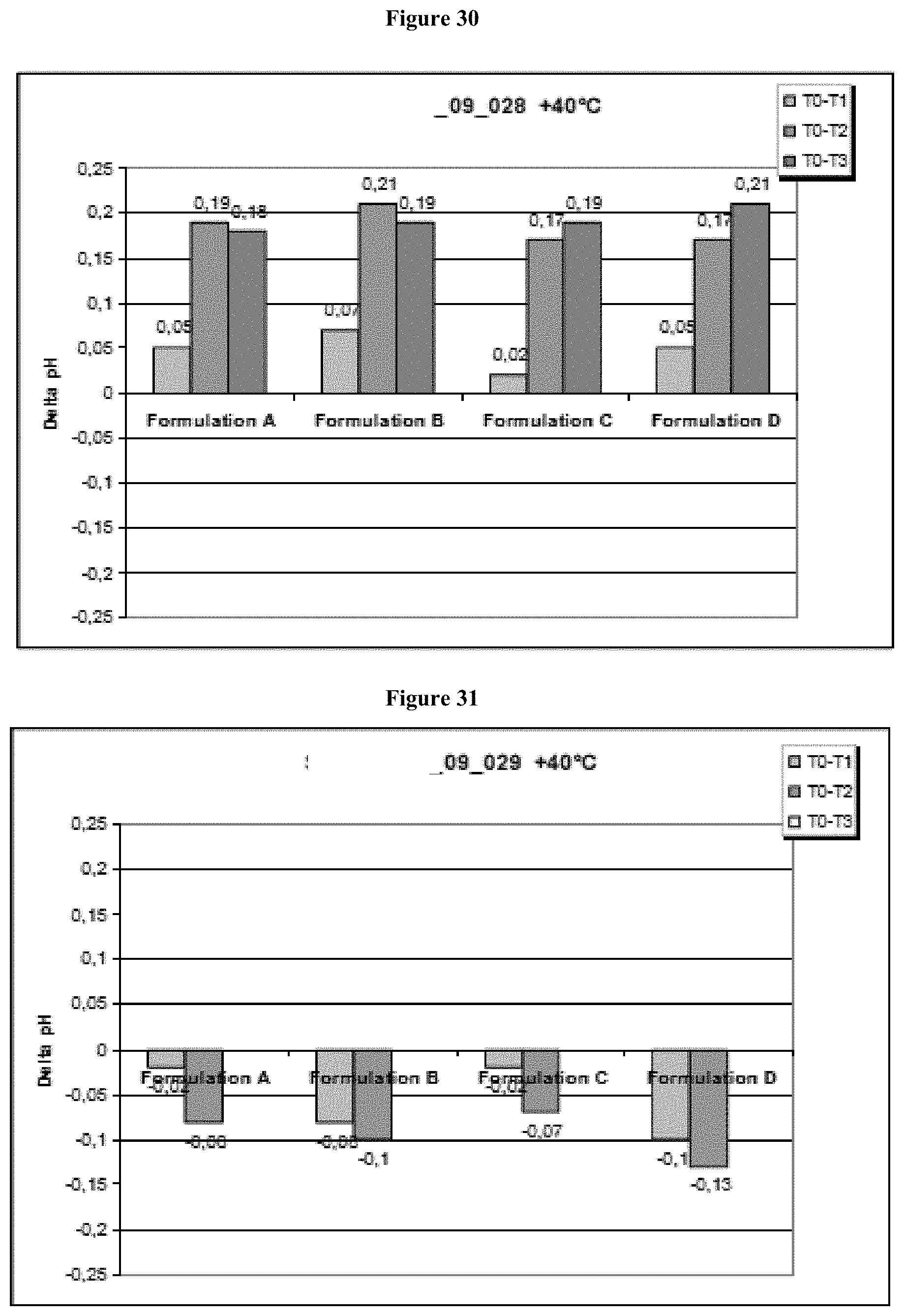
D00020
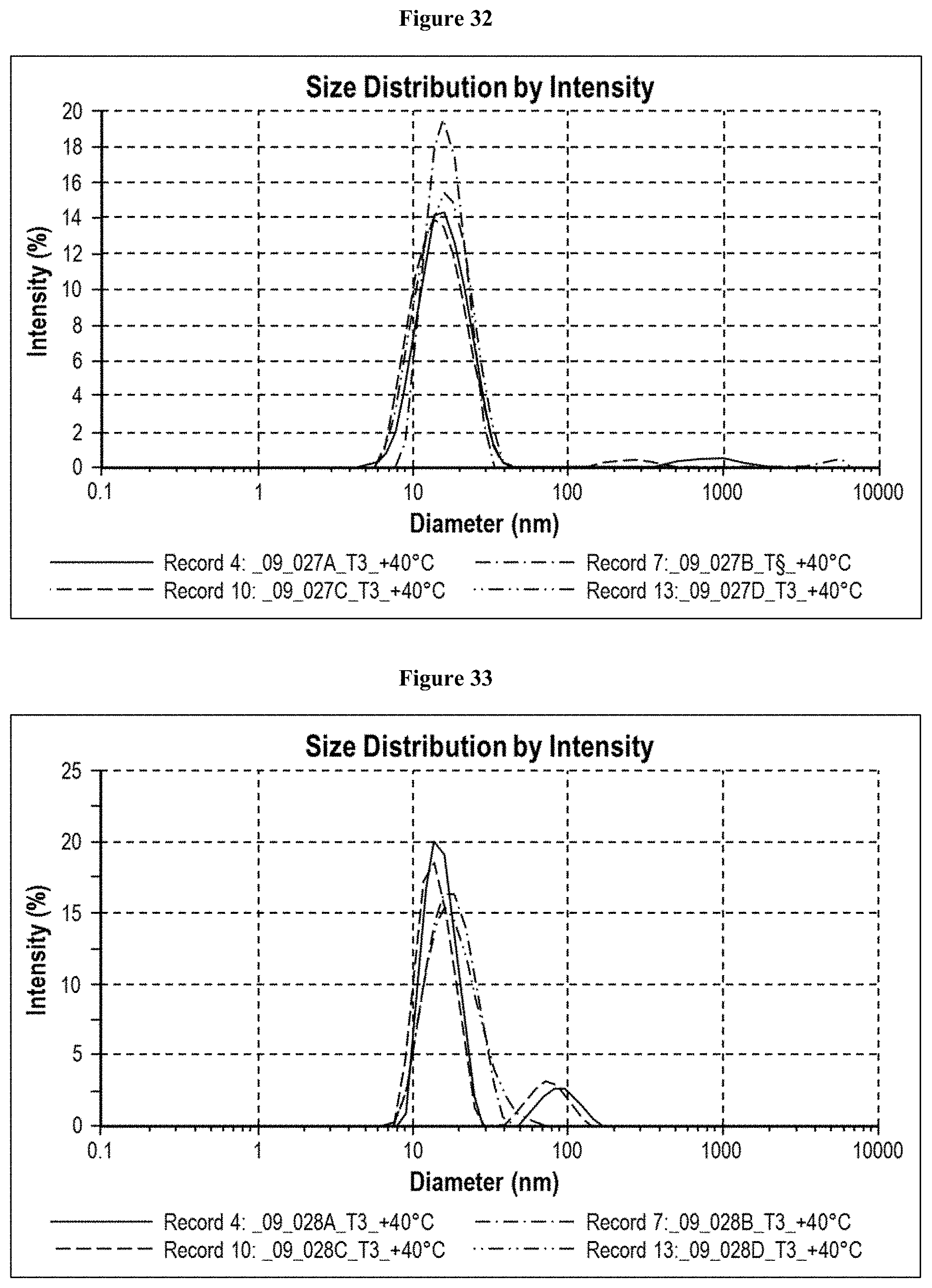
D00021

D00022

D00023
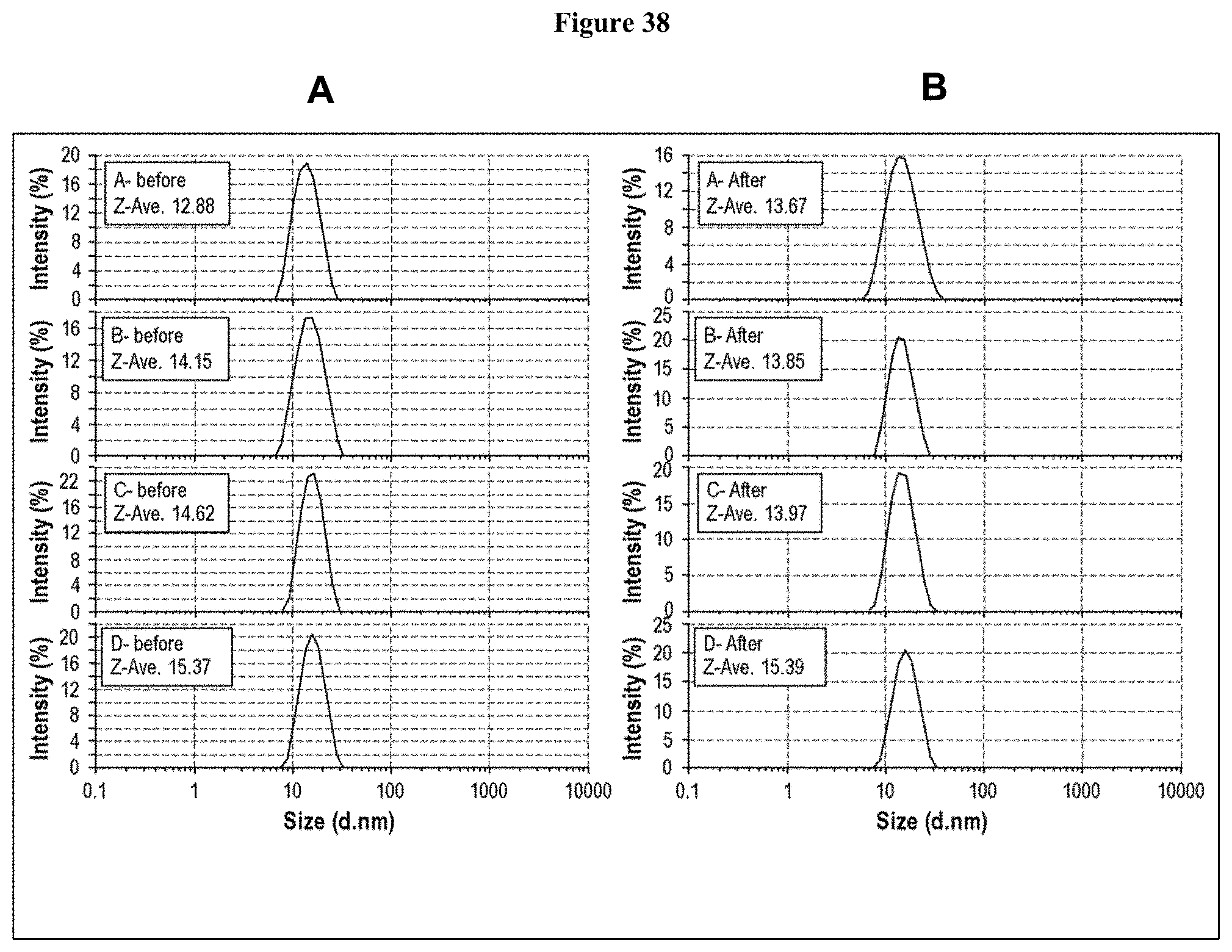
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.