Methods And Compositions For Protein Sequencing
Reed; Brian ; et al.
U.S. patent application number 16/686028 was filed with the patent office on 2020-07-09 for methods and compositions for protein sequencing. This patent application is currently assigned to Quantum-Si Incorporated. The applicant listed for this patent is Quantum-Si Incorporated. Invention is credited to Thomas Christian, Kathren Croce, David Dodd, Alexander Goryaynov, Jeremy Lackey, Roger Nani, Brian Reed, Jonathan M. Rothberg.
| Application Number | 20200219590 16/686028 |
| Document ID | / |
| Family ID | 68841224 |
| Filed Date | 2020-07-09 |








View All Diagrams
| United States Patent Application | 20200219590 |
| Kind Code | A1 |
| Reed; Brian ; et al. | July 9, 2020 |
METHODS AND COMPOSITIONS FOR PROTEIN SEQUENCING
Abstract
Aspects of the application provide methods of identifying and sequencing proteins, polypeptides, and amino acids, and compositions useful for the same. In some aspects, the application provides methods of obtaining data during a degradation process of a polypeptide, and outputting a sequence representative of the polypeptide. In some aspects, the application provides amino acid recognition molecules comprising a shielding element that enhances photostability in polypeptide sequencing reactions.
| Inventors: | Reed; Brian; (Madison, CT) ; Lackey; Jeremy; (Foster City, CA) ; Christian; Thomas; (Killingworth, CT) ; Nani; Roger; (Madison, CT) ; Dodd; David; (Guilford, CT) ; Croce; Kathren; (West Haven, CT) ; Goryaynov; Alexander; (New Haven, CT) ; Rothberg; Jonathan M.; (Guilford, CT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Quantum-Si Incorporated Guilford CT |
||||||||||
| Family ID: | 68841224 | ||||||||||
| Appl. No.: | 16/686028 | ||||||||||
| Filed: | November 15, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62907507 | Sep 27, 2019 | |||
| 62768076 | Nov 15, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G16B 50/00 20190201; G16B 50/30 20190201; G01N 33/6824 20130101; G01N 33/581 20130101; G01N 21/6428 20130101; G16B 25/10 20190201; C07K 19/00 20130101; G01N 33/58 20130101; G01N 2021/6439 20130101; G16B 30/00 20190201; C07K 14/47 20130101; G01N 33/6821 20130101; G16B 40/00 20190201; C12Q 1/6806 20130101; G01N 2458/00 20130101; G01N 1/28 20130101; G01N 33/582 20130101 |
| International Class: | G16B 50/30 20060101 G16B050/30; G16B 25/10 20060101 G16B025/10; G16B 40/00 20060101 G16B040/00; C12Q 1/6806 20060101 C12Q001/6806 |
Claims
1. A method comprising: obtaining data during a degradation process of a polypeptide; analyzing the data to determine portions of the data corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process; and outputting an amino acid sequence representative of the polypeptide.
2. The method of claim 1, wherein the data is indicative of amino acid identity at the terminus of the polypeptide during the degradation process.
3. The method of claim 2, wherein the data is indicative of a signal produced by one or more amino acid recognition molecules binding to different types of terminal amino acids at the terminus during the degradation process.
4. The method of claim 1, wherein the data is indicative of a luminescent signal generated during the degradation process.
5. The method of claim 1, wherein the data is indicative of an electrical signal generated during the degradation process.
6. The method of claim 1, wherein analyzing the data further comprises detecting a series of cleavage events and determining the portions of the data between successive cleavage events.
7. The method of claim 1, wherein analyzing the data further comprises determining a type of amino acid for each of the individual portions.
8. The method of claim 1, wherein each of the individual portions comprises a pulse pattern, and analyzing the data further comprises determining a type of amino acid for one or more of the portions based on its respective pulse pattern.
9. The method of claim 8, wherein determining the type of amino acid further comprises identifying an amount of time within a portion when the data is above a threshold value and comparing the amount of time to a duration of time for the portion.
10. The method of claim 8, wherein determining the type of amino acid further comprises identifying at least one pulse duration for each of the one or more portions.
11. The method of claim 8, wherein determining the type of amino acid further comprises identifying at least one interpulse duration for each of the one or more portions.
12. The method of claim 1, wherein the amino acid sequence includes a series of amino acids corresponding to the portions.
13. A system comprising: at least one hardware processor; and at least one non-transitory computer-readable storage medium storing processor-executable instructions that, when executed by the at least one hardware processor, cause the at least one hardware processor to perform the method of claim 1.
14. At least one non-transitory computer-readable storage medium storing processor-executable instructions that, when executed by at least one hardware processor, cause the at least one hardware processor to perform the method of claim 1.
15-283. (canceled)
284. The method of claim 10, wherein the pulse pattern of each of the one or more portions comprises a mean pulse duration of between about 1 millisecond and about 10 seconds.
285. The method of claim 284, wherein the mean pulse duration is between about 10 milliseconds and about 100 milliseconds or between about 100 milliseconds and about 500 milliseconds.
286. The method of claim 10, wherein the pulse pattern of one type of amino acid is different from the pulse pattern of another type of amino acid by a mean pulse duration of at least 10 milliseconds.
287. The method of claim 286, wherein the pulse pattern of one type of amino acid is different from the pulse pattern of another type of amino acid by a mean pulse duration of between about 10 milliseconds and about 100 milliseconds or between about 100 milliseconds and about 10 seconds.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority under 35 U.S.C. .sctn. 119(e) to U.S. Provisional Patent Application No. 62/907,507, filed Sep. 27, 2019, and U.S. Provisional Patent Application No. 62/768,076, filed Nov. 15, 2018, each of which is hereby incorporated by reference in its entirety.
BACKGROUND
[0002] Proteomics has emerged as an important and necessary complement to genomics and transcriptomics in the study of biological systems. The proteomic analysis of an individual organism can provide insights into cellular processes and response patterns, which lead to improved diagnostic and therapeutic strategies. The complexity surrounding protein structure, composition, and modification present challenges in determining large-scale protein sequencing information for a biological sample.
SUMMARY
[0003] In some aspects, the application provides methods and compositions for determining amino acid sequence information from polypeptides (e.g., for sequencing one or more polypeptides). In some embodiments, amino acid sequence information can be determined for single polypeptide molecules. In some embodiments, the relative position of two or more amino acids in a polypeptide is determined, for example for a single polypeptide molecule. In some embodiments, one or more amino acids of a polypeptide are labeled (e.g., directly or indirectly) and the relative positions of the labeled amino acids in the polypeptide is determined.
[0004] In some aspects, the application provides methods comprising obtaining data during a degradation process of a polypeptide. In some embodiments, the methods further comprise analyzing the data to determine portions of the data corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process. In some embodiments, the methods further comprise outputting an amino acid sequence representative of the polypeptide. In some embodiments, the data is indicative of amino acid identity at the terminus of the polypeptide during the degradation process. In some embodiments, the data is indicative of a signal produced by one or more amino acid recognition molecules binding to different types of terminal amino acids at the terminus during the degradation process. In some embodiments, the data is indicative of a luminescent signal generated during the degradation process. In some embodiments, the data is indicative of an electrical signal generated during the degradation process.
[0005] In some embodiments, analyzing the data further comprises detecting a series of cleavage events and determining the portions of the data between successive cleavage events. In some embodiments, analyzing the data further comprises determining a type of amino acid for each of the individual portions. In some embodiments, each of the individual portions comprises a pulse pattern (e.g., a characteristic pattern), and analyzing the data further comprises determining a type of amino acid for one or more of the portions based on its respective pulse pattern. In some embodiments, determining the type of amino acid further comprises identifying an amount of time within a portion when the data is above a threshold value and comparing the amount of time to a duration of time for the portion. In some embodiments, determining the type of amino acid further comprises identifying at least one pulse duration for each of the one or more portions. In some embodiments, determining the type of amino acid further comprises identifying at least one interpulse duration for each of the one or more portions. In some embodiments, the amino acid sequence includes a series of amino acids corresponding to the portions.
[0006] In some aspects, the application provides systems comprising at least one hardware processor, and at least one non-transitory computer-readable storage medium storing processor-executable instructions that, when executed by the at least one hardware processor, cause the at least one hardware processor to perform a method in accordance with the application. In some aspects, the application provides at least one non-transitory computer-readable storage medium storing processor-executable instructions that, when executed by at least one hardware processor, cause the at least one hardware processor to perform a method in accordance with the application.
[0007] In some aspects, the application provides methods of polypeptide sequencing. In some embodiments, the methods comprise contacting a single polypeptide molecule with one or more terminal amino acid recognition molecules. In some embodiments, the methods further comprise detecting a series of signal pulses indicative of association of the one or more terminal amino acid recognition molecules with successive amino acids exposed at a terminus of the single polypeptide molecule while it is being degraded, thereby obtaining sequence information about the single polypeptide molecule. In some embodiments, the amino acid sequence of most or all of the single polypeptide molecule is determined. In some embodiments, the series of signal pulses is a series of real-time signal pulses.
[0008] In some embodiments, association of the one or more terminal amino acid recognition molecules with each type of amino acid exposed at the terminus produces a characteristic pattern in the series of signal pulses that is different from other types of amino acids exposed at the terminus. In some embodiments, a signal pulse of the characteristic pattern corresponds to an individual association event between a terminal amino acid recognition molecule and an amino acid exposed at the terminus. In some embodiments, the characteristic pattern corresponds to a series of reversible terminal amino acid recognition molecule binding interactions with the amino acid exposed at the terminus of the single polypeptide molecule. In some embodiments, the characteristic pattern is indicative of the amino acid exposed at the terminus of the single polypeptide molecule and an amino acid at a contiguous position (e.g., amino acids of the same type or different types).
[0009] In some embodiments, the single polypeptide molecule is degraded by a cleaving reagent that removes one or more amino acids from the terminus of the single polypeptide molecule. In some embodiments, the methods further comprise detecting a signal indicative of association of the cleaving reagent with the terminus. In some embodiments, the cleaving reagent comprises a detectable label (e.g., a luminescent label, a conductivity label). In some embodiments, the single polypeptide molecule is immobilized to a surface. In some embodiments, the single polypeptide molecule is immobilized to the surface through a terminal end distal to the terminus to which the one or more terminal amino acid recognition molecules associate. In some embodiments, the single polypeptide molecule is immobilized to the surface through a linker (e.g., a solubilizing linker comprising a biomolecule).
[0010] In some aspects, the application provides methods of sequencing a polypeptide comprising contacting a single polypeptide molecule in a reaction mixture with a composition comprising one or more terminal amino acid recognition molecules and a cleaving reagent. In some embodiments, the methods further comprise detecting a series of signal pulses indicative of association of the one or more terminal amino acid recognition molecules with a terminus of the single polypeptide molecule in the presence of the cleaving reagent. In some embodiments, the series of signal pulses is indicative of a series of amino acids exposed at the terminus over time as a result of terminal amino acid cleavage by the cleaving reagent.
[0011] In some aspects, the application provides methods of sequencing a polypeptide comprising (a) identifying a first amino acid at a terminus of a single polypeptide molecule, (b) removing the first amino acid to expose a second amino acid at the terminus of the single polypeptide molecule, and (c) identifying the second amino acid at the terminus of the single polypeptide molecule. In some embodiments, (a)-(c) are performed in a single reaction mixture. In some embodiments, (a)-(c) occur sequentially. In some embodiments, (c) occurs before (a) and (b). In some embodiments, the single reaction mixture comprises one or more terminal amino acid recognition molecules. In some embodiments, the single reaction mixture comprises a cleaving reagent. In some embodiments, the first amino acid is removed by the cleaving reagent. In some embodiments, the methods further comprise repeating the steps of removing and identifying one or more amino acids at the terminus of the single polypeptide molecule, thereby determining a sequence (e.g., a partial sequence or a complete sequence) of the single polypeptide molecule.
[0012] In some aspects, the application provides methods of identifying an amino acid of a polypeptide comprising contacting a single polypeptide molecule with one or more amino acid recognition molecules that bind to the single polypeptide molecule. In some embodiments, the methods further comprise detecting a series of signal pulses indicative of association of the one or more amino acid recognition molecules with the single polypeptide molecule under polypeptide degradation conditions. In some embodiments, the methods further comprise identifying a first type of amino acid in the single polypeptide molecule based on a first characteristic pattern in the series of signal pulses.
[0013] In some aspects, the application provides methods of identifying a terminal amino acid (e.g., the N-terminal or the C-terminal amino acid) of a polypeptide. In some embodiments, the methods comprise contacting a polypeptide with one or more labeled affinity reagents (e.g., one or more amino acid recognition molecules) that selectively bind one or more types of terminal amino acids at a terminus of the polypeptide. In some embodiments, the methods further comprise identifying a terminal amino acid at the terminus of the polypeptide by detecting an interaction of the polypeptide with the one or more labeled affinity reagents.
[0014] In yet other aspects, the application provides methods of polypeptide sequencing by Edman-type degradation reactions. In some embodiments, Edman-type degradation reactions may be performed by contacting a polypeptide with different reaction mixtures for purposes of either detection or cleavage (e.g., as compared to a dynamic sequencing reaction, which can involve detection and cleavage using a single reaction mixture).
[0015] Accordingly, in some aspects, the application provides methods of determining an amino acid sequence of a polypeptide comprising (i) contacting a polypeptide with one or more labeled affinity reagents that selectively bind one or more types of terminal amino acids at a terminus of the polypeptide. In some embodiments, the methods further comprise (ii) identifying a terminal amino acid (e.g., the N-terminal or the C-terminal amino acid) at the terminus of the polypeptide by detecting an interaction of the polypeptide with the one or more labeled affinity reagents. In some embodiments, the methods further comprise (iii) removing the terminal amino acid. In some embodiments, the methods further comprise (iv) repeating (i)-(iii) one or more times at the terminus of the polypeptide to determine an amino acid sequence of the polypeptide.
[0016] In some embodiments, the methods further comprise, after (i) and before (ii), removing any of the one or more labeled affinity reagents that do not selectively bind the terminal amino acid. In some embodiments, the methods further comprise, after (ii) and before (iii), removing any of the one or more labeled affinity reagents that selectively bind the terminal amino acid.
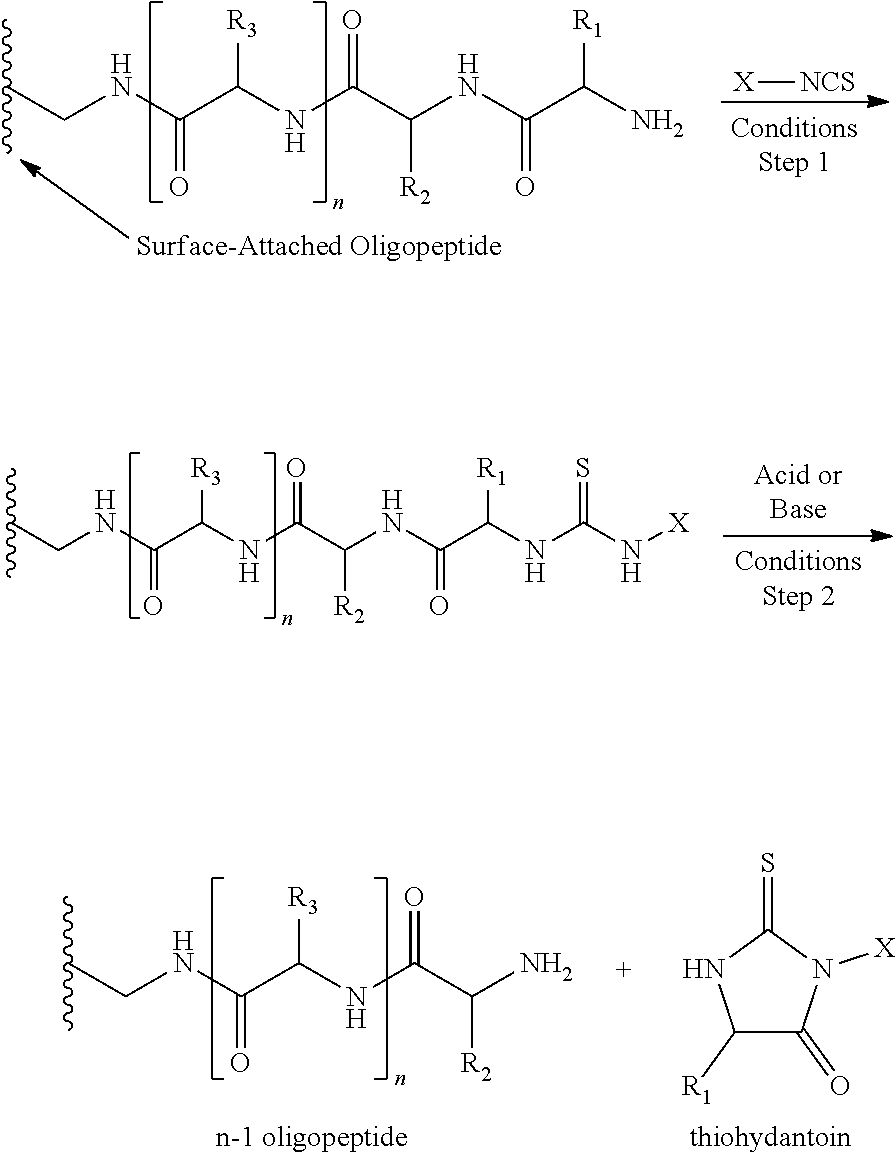
[0017] In some embodiments, removing a terminal amino acid (e.g., (iii)) comprises modifying the terminal amino acid by contacting the terminal amino acid with an isothiocyanate (e.g., phenyl isothiocyanate), and contacting the modified terminal amino acid with a protease that specifically binds and removes the modified terminal amino acid. In some embodiments cleaving a terminal amino acid (e.g., (iii)) comprises modifying the terminal amino acid by contacting the terminal amino acid with an isothiocyanate, and subjecting the modified terminal amino acid to acidic or basic conditions sufficient to remove the modified terminal amino acid.
[0018] In some embodiments, identifying a terminal amino acid comprises identifying the terminal amino acid as being one type of the one or more types of terminal amino acids to which the one or more labeled affinity reagents bind. In some embodiments, identifying a terminal amino acid comprises identifying the terminal amino acid as being a type other than the one or more types of terminal amino acids to which the one or more labeled affinity reagents bind.
[0019] In some aspects, the application provides amino acid recognition molecules comprising a shielding element, e.g., for enhanced photostability in polypeptide sequencing reactions. In some aspects, the application provides an amino acid recognition molecule of Formula (I):
A-(Y).sub.n-D (I),
wherein: A is an amino acid binding component comprising at least one amino acid recognition molecule; each instance of Y is a polymer that forms a covalent or non-covalent linkage group; n is an integer from 1 to 10, inclusive; and D is a label component comprising at least one detectable label. In some embodiments, D is less than 200 .ANG. in diameter. In some embodiments, --(Y).sub.n-- is at least 2 nm in length (e.g., at least 5 nm, at least 10 nm, at least 20 nm, at least 30 nm, at least 50 nm, or more, in length). In some embodiments, --(Y).sub.n-- is between about 2 nm and about 200 nm in length (e.g., between about 2 nm and about 100 nm, between about 5 nm and about 50 nm, or between about 10 nm and about 100 nm in length). In some embodiments, each instance of Y is independently a biomolecule or a dendritic polymer (e.g., a polyol, a dendrimer). In some embodiments, the application provides a composition comprising the amino acid recognition molecule of Formula (I). In some embodiments, the amino acid recognition molecule is soluble in the composition.
[0020] In some aspects, the application provides an amino acid recognition molecule of Formula (II):
A-Y.sup.1-D (II),
wherein: A is an amino acid binding component comprising at least one amino acid recognition molecule; Y.sup.1 is a nucleic acid or a polypeptide; D is a label component comprising at least one detectable label. In some embodiments, when Y.sup.1 is a nucleic acid, the nucleic acid forms a covalent or non-covalent linkage group. In some embodiments, provided that when Y.sup.1 is a polypeptide, the polypeptide forms a non-covalent linkage group characterized by a dissociation constant (K.sub.D) of less than 50.times.10.sup.-9 M. In some embodiments, the K.sub.D is less than 1.times.10.sup.-9 M, less than 1.times.10.sup.-10 M, less than 1.times.10.sup.-11 M, or less than 1.times.10.sup.-12 M.
[0021] In some aspects, the application provides an amino acid recognition molecule comprising: a nucleic acid; at least one amino acid recognition molecule attached to a first attachment site on the nucleic acid; and at least one detectable label attached to a second attachment site on the nucleic acid, where the nucleic acid forms a covalent or non-covalent linkage group between the at least one amino acid recognition molecule and the at least one detectable label. In some embodiments, the nucleic acid comprises a first oligonucleotide strand. In some embodiments, the nucleic acid further comprises a second oligonucleotide strand hybridized with the first oligonucleotide strand.
[0022] In some aspects, the application provides an amino acid recognition molecule comprising: a multivalent protein comprising at least two ligand-binding sites; at least one amino acid recognition molecule attached to the protein through a first ligand moiety bound to a first ligand-binding site on the protein; and at least one detectable label attached to the protein through a second ligand moiety bound to a second ligand-binding site on the protein. In some embodiments, the multivalent protein is an avidin protein.
[0023] In some embodiments, a shielded amino acid recognition molecule may be used in polypeptide sequencing methods in accordance with the application, or any method known in the art. Accordingly, in some aspects, the application provides methods of polypeptide sequencing (e.g., in an Edman-type degradation reaction, in a dynamic sequencing reaction, or other method known in the art) comprising contacting a polypeptide molecule with one or more shielded amino acid recognition molecules of the application. For example, in some embodiments, the methods comprise contacting a polypeptide molecule with at least one amino acid recognition molecule that comprises a shield or shielding element in accordance with the application, and detecting association of the at least one amino acid recognition molecule with the polypeptide molecule.
[0024] In some aspects, the application provides methods of identifying a protein of interest in a mixed sample. In some embodiments, the methods comprise cleaving a mixed protein sample to produce a plurality of polypeptide fragments. In some embodiments, the methods further comprise determining an amino acid sequence of at least one polypeptide fragment of the plurality in a method in accordance with the methods of the application. In some embodiments, the methods further comprise identifying a protein of interest in the mixed sample if the amino acid sequence is uniquely identifiable to the protein of interest.
[0025] In some embodiments, methods of identifying a protein of interest in a mixed sample comprise cleaving a mixed protein sample to produce a plurality of polypeptide fragments. In some embodiments, the methods further comprise labeling one or more types of amino acids in the plurality of polypeptide fragments with one or more different luminescent labels. In some embodiments, the methods further comprise measuring luminescence over time for at least one labeled polypeptide of the plurality. In some embodiments, the methods further comprise determining an amino acid sequence of the at least one labeled polypeptide based on the luminescence detected. In some embodiments, the methods further comprise identifying a protein of interest in the mixed sample if the amino acid sequence is uniquely identifiable to the protein of interest.
[0026] Accordingly, in some embodiments, a polypeptide molecule or protein of interest to be analyzed in accordance with the application can be of a mixed or purified sample. In some embodiments, the polypeptide molecule or protein of interest is obtained from a biological sample (e.g., blood, tissue, saliva, urine, or other biological source). In some embodiments, the polypeptide molecule or protein of interest is obtained from a patient sample (e.g., a human sample).
[0027] The details of certain embodiments of the invention are set forth in the Detailed Description of Certain Embodiments, as described below. Other features, objects, and advantages of the invention will be apparent from the Definitions, Examples, Figures, and Claims.
BRIEF DESCRIPTION OF THE DRAWINGS
[0028] The skilled artisan will understand that the figures, described herein, are for illustration purposes only. It is to be understood that, in some instances, various aspects of the invention may be shown exaggerated or enlarged to facilitate an understanding of the invention. In the drawings, like reference characters generally refer to like features, functionally similar and/or structurally similar elements throughout the various figures. The drawings are not necessarily to scale, emphasis instead being placed upon illustrating the principles of the teachings. The drawings are not intended to limit the scope of the present teachings in any way.
[0029] The features and advantages of the present invention will become more apparent from the detailed description set forth below when taken in conjunction with the drawings.
[0030] When describing embodiments in reference to the drawings, direction references ("above," "below," "top," "bottom," "left," "right," "horizontal," "vertical," etc.) may be used. Such references are intended merely as an aid to the reader viewing the drawings in a normal orientation. These directional references are not intended to describe a preferred or only orientation of an embodied device. A device may be embodied in other orientations.
[0031] As is apparent from the detailed description, the examples depicted in the figures and further described for the purpose of illustration throughout the application describe non-limiting embodiments, and in some cases may simplify certain processes or omit features or steps for the purpose of clearer illustration.
[0032] FIGS. 1A-1B show an example of polypeptide sequencing by detection (FIG. 1A) and analysis (FIG. 1B) of single molecule binding interactions.
[0033] FIGS. 1C-1E show various examples of labeled affinity reagents and methods of use in accordance with the application. FIG. 1C depicts example configurations of labeled affinity reagents, including labeled enzymes and labeled aptamers which selectively bind one or more types of terminal amino acids. FIG. 1D generically depicts a degradation-based process of polypeptide sequencing using labeled affinity reagents. FIG. 1E shows an example of polypeptide sequencing using labeled aptamers by repeated cycles of terminal amino acid detection, modification, and cleavage.
[0034] FIG. 2 shows an example of polypeptide sequencing in real-time using labeled exopeptidases that each selectively binds and cleaves a different type of terminal amino acid.
[0035] FIGS. 3A-3B show examples of polypeptide sequencing in real-time by evaluating binding interactions of terminal and/or internal amino acids with labeled affinity reagents and a labeled cleaving reagent (e.g., a labeled non-specific exopeptidase). FIG. 3A shows an example of real-time sequencing by detecting a series of pulses in a signal output. FIG. 3B schematically depicts a temperature-dependent sequencing process.
[0036] FIG. 4 shows an example of polypeptide sequencing in real-time by evaluating binding interactions of terminal and internal amino acids with labeled affinity reagents and a labeled non-specific exopeptidase.
[0037] FIGS. 5A-5E show non-limiting examples of affinity reagents labeled through a shielding element. FIG. 5A illustrates single-molecule peptide sequencing with an affinity reagent labeled through a conventional covalent linkage. FIG. 5B illustrates single-molecule peptide sequencing with an affinity reagent comprising a shielding element. FIGS. 5C-5E illustrate various examples of shielding elements in accordance with the application.
[0038] FIG. 6 shows an example of identifying polypeptides based on a unique combination of amino acids detected in a labeled polypeptide.
[0039] FIG. 7 shows an example of polypeptide sequencing by detecting luminescence of a labeled polypeptide which is subjected to repeated cycles of terminal amino acid modification and cleavage.
[0040] FIGS. 8A-8C show an example of polypeptide sequencing by processive enzymatic cleavage of a labeled polypeptide. FIG. 8A shows an example of sequencing by processive enzymatic cleavage of a labeled polypeptide by an immobilized terminal peptidase. FIG. 8B shows an example of sequencing by processive enzymatic cleavage of an immobilized labeled polypeptide by a terminal peptidase. FIG. 8C schematically illustrates an example of a real-time sequencing process performed in accordance with FIG. 8B.
[0041] FIG. 9 schematically illustrates an example of sequencing by cofactor-based FRET using an immobilized ATP-dependent protease, donor-labeled ATP, and acceptor-labeled amino acids of a polypeptide substrate.
[0042] FIGS. 10A-10C show various examples of preparing samples and sample well surfaces for analysis of polypeptides and proteins in accordance with the application. FIG. 10A generically depicts an example process of preparing terminally modified polypeptides from a protein sample. FIG. 10B generically depicts an example process of conjugating a solubilizing linker to a polypeptide. FIG. 10C shows an example schematic of a sample well having modified surfaces which may be used to promote single molecule immobilization to a bottom surface.
[0043] FIG. 11 is a diagram of an illustrative sequence data processing pipeline for analyzing data obtained during a polypeptide degradation process, in accordance with some embodiments of the technology described herein.
[0044] FIG. 12 is a flow chart of an illustrative process for determining an amino acid sequence of a polypeptide molecule, in accordance with some embodiments of the technology described herein.
[0045] FIG. 13 is a flow chart of an illustrative process for determining an amino acid sequence representative of a polypeptide, in accordance with some embodiments of the technology described herein.
[0046] FIG. 14 is a block diagram of an illustrative computer system that may be used in implementing some embodiments of the technology described herein.
[0047] FIGS. 15A-15C show experimental data for select peptide-linker conjugates prepared and evaluated for enhanced solubility provided by different solubilizing linkers. FIG. 15A shows example structures of peptide-linker conjugates that were synthesized and evaluated. FIG. 15B shows results from LCMS which demonstrate peptide cleavage at the N-terminus. FIG. 15C shows results from a loading experiment.
[0048] FIG. 16 shows a summary of amino acid cleavage activities for select exopeptidases based on experimental results.
[0049] FIGS. 17A-17C show experimental data for a dye/peptide conjugate assay for detecting and cleaving terminal amino acids. FIG. 17A shows example schemes and structures used for performing a dye/peptide conjugate assay. FIG. 17B shows imaging results for peptide-linker conjugate loading into sample wells in an on-chip assay. FIG. 17C shows example signal traces which detected peptide-conjugate loading and terminal amino acid cleavage.
[0050] FIGS. 18A-18F show experimental data for a FRET dye/peptide conjugate assay for detecting and cleaving terminal amino acids. FIG. 18A shows example schemes and structures used for performing a FRET dye/peptide conjugate assay. FIG. 18B shows FRET imaging results for different time points. FIG. 18C shows cutting efficiency at the different time points. FIG. 18D shows cutting displayed at each of the different time points. FIG. 18E shows additional FRET imaging results for different time points with a proline iminopeptidase from Yersinia pestis (yPIP). FIG. 18F shows FRET imaging results for different time points with an aminopeptidase from Vibrio proteolyticus (VPr).
[0051] FIGS. 19A-19H show experimental data for terminal amino acid discrimination by a labeled affinity reagent. FIG. 19A shows a crystal structure of a ClpS2 protein that was labeled for these experiments. FIG. 19B shows single molecule intensity traces which illustrate N-terminal amino acid discrimination by the labeled ClpS2 protein. FIG. 19C is a plot showing mean pulse duration for different terminal amino acids. FIG. 19D is a plot showing mean interpulse duration for different terminal amino acids. FIG. 19E shows plots further illustrating discriminant pulse durations among the different terminal amino acids.
[0052] FIGS. 19F, 19G, and 19H show example results from dwell time analysis demonstrating leucine recognition by a ClpS protein from Thermosynochoccus elongatus (teClpS). FIG. 19I shows example results from dwell time analysis demonstrating differentiable recognition of phenylalanine, leucine, tryptophan, and tyrosine by A. tumefaciens ClpS1. FIG. 19J shows example results from dwell time analysis demonstrating leucine recognition by S. elongatus ClpS2. FIGS. 19K-19L show example results from dwell time analysis demonstrating proline recognition by GID4.
[0053] FIGS. 20A-20D show example results from polypeptide sequencing reactions conducted in real-time using a labeled ClpS2 recognition protein and an aminopeptidase cleaving reagent in the same reaction mixture. FIG. 20A shows signal trace data for a first sequencing reaction. FIG. 20B shows pulse duration statistics for the signal trace data shown in FIG. 20A. FIG. 20C shows signal trace data for a second sequencing reaction. FIG. 20D shows pulse duration statistics for the signal trace data shown in FIG. 20C.
[0054] FIGS. 21A-21F show experimental data for terminal amino acid identification and cleavage by a labeled exopeptidase. FIG. 21A shows a crystal structure of a proline iminopeptidase (yPIP) that was site-specifically labeled for these experiments. FIG. 21B shows the degree of labeling for the purified protein product. FIG. 21C is an image of SDS page confirming site-specific labeling of yPIP. FIG. 21D is an overexposed image of the SDS page gel confirming site-specific labeling. FIG. 21E is an image of a Coomassie stained gel confirming purity of labeled protein product. FIG. 21F is an HPLC trace demonstrating cleavage activity of the labeled exopeptidase. The sequence YPYPYPK corresponds to SEQ ID NO: 82. The sequence PYPYPK corresponds to SEQ ID NO: 83.
[0055] FIGS. 22A-22F show data from experiments evaluating recognition of amino acids containing specific post-translational modifications. FIG. 22A shows representative traces which demonstrated phospho-tyrosine recognition by an SH2 domain-containing protein; FIG. 22B shows pulse duration data corresponding to the traces of FIG. 22A; and FIG. 22C shows statistics determined for the traces. FIGS. 22D-22F show representative traces from negative control experiments.
[0056] FIG. 23 is a plot showing median pulse duration from experiments evaluating the effects of penultimate amino acids on pulse duration.
[0057] FIGS. 24A-24C show data from experiments evaluating simultaneous amino acid recognition by differentially labeled recognition molecules. FIG. 24A shows a representative trace. FIG. 24B is a plot comparing pulse duration data obtained during these experiments for each recognition molecule. FIG. 24C shows pulse duration statistics for these experiments.
[0058] FIGS. 25A-25C show data from experiments evaluating the photostability of peptides during single-molecule recognition. FIG. 25A shows a representative trace from recognition using atClpS2-V1 labeled with a dye .about.2 nm from the amino acid binding site. FIG. 25B shows a visualization of the structure of the ClpS2 protein used in these experiments. FIG. 25C shows a representative trace from recognition using ClpS2 labeled with a dye >10 nm from the amino acid binding site through a DNA/protein linker.
[0059] FIGS. 26A-26D show representative traces from polypeptide sequencing reactions conducted in real-time on a complementary metal-oxide-semiconductor (CMOS) chip using a ClpS2 recognition protein labeled through a DNA/streptavidin linker in the presence of an aminopeptidase cleaving reagent.
[0060] FIG. 27 shows representative traces from polypeptide sequencing reactions conducted in real-time using atClpS2-V1 recognition protein labeled through a DNA/streptavidin linker in the presence of Pyrococcus horikoshii TET aminopeptidase cleaving reagent.
[0061] FIGS. 28A-28J show representative trace data from polypeptide sequencing reactions conducted in real-time using multiple types of exopeptidases with differential cleavage specificities. FIG. 28A shows a representative trace from a reaction performed with hTET exopeptidase, with expanded pulse pattern regions shown in FIG. 28B. The sequence YAAWAAFADDDWK in FIG. 28A corresponds to SEQ ID NO: 78. FIG. 28C shows a representative trace from a reaction performed with both hTET and yPIP exopeptidases, with expanded pulse pattern regions shown in FIG. 28D, and additional representative traces shown in FIG. 28E. The sequence FYPLPWPDDDYK in FIG. 28C corresponds to SEQ ID NO: 80. FIG. 28F shows a representative trace from a further reaction performed with both hTET and yPIP exopeptidases, with expanded pulse pattern regions shown in FIG. 28G, and additional representative traces shown in FIG. 28H. FIG. 28I shows a representative trace from a reaction performed with both PfuTET and yPIP exopeptidases, with expanded pulse pattern regions shown in FIG. 28J. The sequence YPLPWPDDDYK in FIGS. 28F and 28I corresponds to SEQ ID NO: 81.
DETAILED DESCRIPTION
[0062] Aspects of the application relate to methods of protein sequencing and identification, methods of polypeptide sequencing and identification, methods of amino acid identification, and compositions for performing such methods.
[0063] In some aspects, the application relates to the discovery of polypeptide sequencing techniques which may be implemented using existing analytic instruments with few or no device modifications. For example, previous polypeptide sequencing strategies have involved iterative cycling of different reagent mixtures through a reaction vessel containing a polypeptide being analyzed. Such strategies may require modification of an existing analytic instrument, such as a nucleic acid sequencing instrument, which may not be equipped with a flow cell or similar apparatus capable of reagent cycling. The inventors have recognized and appreciated that certain polypeptide sequencing techniques of the application do not require iterative reagent cycling, thereby permitting the use of existing instruments without significant modifications which might increase instrument size. Accordingly, in some aspects, the application provides methods of polypeptide sequencing that permit the use of smaller sequencing instruments. In some aspects, the application relates to the discovery of polypeptide sequencing techniques that allow both genomic and proteomic analyses to be performed using the same sequencing instrument.
[0064] The inventors have further recognized and appreciated that differential binding interactions can provide an additional or alternative approach to conventional labeling strategies in polypeptide sequencing. Conventional polypeptide sequencing can involve labeling each type of amino acid with a uniquely identifiable label. This process can be laborious and prone to error, as there are at least twenty different types of naturally occurring amino acids in addition to numerous post-translational variations thereof. In some aspects, the application relates to the discovery of techniques involving the use of amino acid recognition molecules which differentially associate with different types of amino acids to produce detectable characteristic signatures indicative of an amino acid sequence of a polypeptide. Accordingly, aspects of the application provide techniques that do not require polypeptide labeling and/or harsh chemical reagents used in certain conventional polypeptide sequencing approaches, thereby increasing throughput and/or accuracy of sequence information obtained from a sample.
[0065] In some aspects, the application relates to the discovery that a polypeptide sequencing reaction can be monitored in real-time using only a single reaction mixture (e.g., without requiring iterative reagent cycling through a reaction vessel). As detailed above, conventional polypeptide sequencing reactions can involve exposing a polypeptide to different reagent mixtures to cycle between steps of amino acid detection and amino acid cleavage. Accordingly, in some aspects, the application relates to an advancement in next generation sequencing that allows for the analysis of polypeptides by amino acid detection throughout an ongoing degradation reaction in real-time. Approaches for such polypeptide analysis by dynamic sequencing are described below.
[0066] As described herein, in some aspects, the application provides methods of sequencing a polypeptide by obtaining data during a polypeptide degradation process, and analyzing the data to determine portions of the data corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process. In some embodiments, the portions of the data comprise a series of signal pulses indicative of association of one or more amino acid recognition molecules with successive amino acids exposed at the terminus of the polypeptide (e.g., during a degradation). In some embodiments, the series of signal pulses corresponds to a series of reversible single molecule binding interactions at the terminus of the polypeptide during the degradation process.
[0067] A non-limiting example of polypeptide sequencing by detecting single molecule binding interactions during a polypeptide degradation process is schematically illustrated in FIG. 1A. An example signal trace (I) is shown with a series of panels (II) that depict different association events at times corresponding to changes in the signal. As shown, an association event between an amino acid recognition molecule (stippled shape) and an amino acid at the terminus of a polypeptide (shown as beads-on-a-string) produces a change in magnitude of the signal that persists for a duration of time.
[0068] Panels (A) and (B) depict different association events between an amino acid recognition molecule and a first amino acid exposed at the terminus of the polypeptide (e.g., a first terminal amino acid). Each association event produces a change in the signal trace (I) characterized by a change in magnitude of the signal that persists for the duration of the association event. Accordingly, the time duration between the association events of panels (A) and (B) may correspond to a duration of time within which the polypeptide is not detectably associated with an amino acid recognition molecule.
[0069] Panels (C) and (D) depict different association events between an amino acid recognition molecule and a second amino acid exposed at the terminus of the polypeptide (e.g., a second terminal amino acid). As described herein, an amino acid that is "exposed" at the terminus of a polypeptide is an amino acid that is still attached to the polypeptide and that becomes the terminal amino acid upon removal of the prior terminal amino acid during degradation (e.g., either alone or along with one or more additional amino acids). Accordingly, the first and second amino acids of the series of panels (II) provide an illustrative example of successive amino acids exposed at the terminus of the polypeptide, where the second amino acid became the terminal amino acid upon removal of the first amino acid.
[0070] As generically depicted, the association events of panels (C) and (D) produce changes in the signal trace (I) characterized by changes in magnitude that persist for time durations that are relatively shorter than that of panels (A) and (B), and the time duration between the association events of panels (C) and (D) is relatively shorter than that of panels (A) and (B). As described herein, in some embodiments, either one or both of these distinctive changes in signal may be used to determine characteristic patterns in the signal trace (I) which can discriminate between different types of amino acids. In some embodiments, a transition from one characteristic pattern to another is indicative of amino acid cleavage. As used herein, in some embodiments, amino acid cleavage refers to the removal of at least one amino acid from a terminus of a polypeptide (e.g., the removal of at least one terminal amino acid from the polypeptide). In some embodiments, amino acid cleavage is determined by inference based on a time duration between characteristic patterns. In some embodiments, amino acid cleavage is determined by detecting a change in signal produced by association of a labeled cleaving reagent with an amino acid at the terminus of the polypeptide. As amino acids are sequentially cleaved from the terminus of the polypeptide during degradation, a series of changes in magnitude, or a series of signal pulses, is detected. In some embodiments, signal pulse data can be analyzed as illustrated in FIG. 1B.
[0071] In some embodiments, signal data can be analyzed to extract signal pulse information by applying threshold levels to one or more parameters of the signal data. For example, panel (III) depicts a threshold magnitude level ("M.sub.L") applied to the signal data of the example signal trace (I). In some embodiments, M.sub.L is a minimum difference between a signal detected at a point in time and a baseline determined for a given set of data. In some embodiments, a signal pulse ("sp") is assigned to each portion of the data that is indicative of a change in magnitude exceeding M.sub.L and persisting for a duration of time. In some embodiments, a threshold time duration may be applied to a portion of the data that satisfies M.sub.L to determine whether a signal pulse is assigned to that portion. For example, experimental artifacts may give rise to a change in magnitude exceeding M.sub.L that does not persist for a duration of time sufficient to assign a signal pulse with a desired confidence (e.g., transient association events which could be non-discriminatory for amino acid type, non-specific detection events such as diffusion into an observation region or reagent sticking within an observation region). Accordingly, in some embodiments, a signal pulse is extracted from signal data based on a threshold magnitude level and a threshold time duration.
[0072] Extracted signal pulse information is shown in panel (III) with the example signal trace (I) superimposed for illustrative purposes. In some embodiments, a peak in magnitude of a signal pulse is determined by averaging the magnitude detected over a duration of time that persists above M.sub.L. It should be appreciated that, in some embodiments, a "signal pulse" as used herein can refer to a change in signal data that persists for a duration of time above a baseline (e.g., raw signal data, as illustrated by the example signal trace (I)), or to signal pulse information extracted therefrom (e.g., processed signal data, as illustrated in panel (IV)).
[0073] Panel (IV) shows the signal pulse information extracted from the example signal trace (I). In some embodiments, signal pulse information can be analyzed to identify different types of amino acids in a sequence based on different characteristic patterns in a series of signal pulses. For example, as shown in panel (IV), the signal pulse information is indicative of a first type of amino acid based on a first characteristic pattern ("CP.sub.1") and a second type of amino acid based on a second characteristic pattern ("CP.sub.2"). By way of example, the two signal pulses detected at earlier time points provide information indicative of the first amino acid at the terminus of the polypeptide based on CP.sub.1, and the two signal pulses detected at later time points provide information indicative of the second amino acid at the terminus of the polypeptide based on CP.sub.2.
[0074] Also as shown in panel (IV), each signal pulse comprises a pulse duration ("pd") corresponding to an association event between the amino acid recognition molecule and the amino acid of the characteristic pattern. In some embodiments, the pulse duration is characteristic of a dissociation rate of binding. Also as shown, each signal pulse of a characteristic pattern is separated from another signal pulse of the characteristic pattern by an interpulse duration ("ipd"). In some embodiments, the interpulse duration is characteristic of an association rate of binding. In some embodiments, a change in magnitude (".DELTA.M") can be determined for a signal pulse based on a difference between baseline and the peak of a signal pulse. In some embodiments, a characteristic pattern is determined based on pulse duration. In some embodiments, a characteristic pattern is determined based on pulse duration and interpulse duration. In some embodiments, a characteristic pattern is determined based on any one or more of pulse duration, interpulse duration, and change in magnitude.
[0075] Accordingly, as illustrated by FIGS. 1A-1B, in some embodiments, polypeptide sequencing is performed by detecting a series of signal pulses indicative of association of one or more amino acid recognition molecules with successive amino acids exposed at the terminus of a polypeptide in an ongoing degradation reaction. The series of signal pulses can be analyzed to determine characteristic patterns in the series of signal pulses, and the time course of characteristic patterns can be used to determine an amino acid sequence of the polypeptide.
[0076] In some embodiments, the series of signal pulses comprises a series of changes in magnitude of an optical signal over time. In some embodiments, the series of changes in the optical signal comprises a series of changes in luminescence produced during association events. In some embodiments, luminescence is produced by a detectable label associated with one or more reagents of a sequencing reaction. For example, in some embodiments, each of the one or more amino acid recognition molecules comprises a luminescent label. In some embodiments, a cleaving reagent comprises a luminescent label. Examples of luminescent labels and their use in accordance with the application are provided elsewhere herein.
[0077] In some embodiments, the series of signal pulses comprises a series of changes in magnitude of an electrical signal over time. In some embodiments, the series of changes in the electrical signal comprises a series of changes in conductance produced during association events. In some embodiments, conductivity is produced by a detectable label associated with one or more reagents of a sequencing reaction. For example, in some embodiments, each of the one or more amino acid recognition molecules comprises a conductivity label. Examples of conductivity labels and their use in accordance with the application are provided elsewhere herein. Methods for identifying single molecules using conductivity labels have been described (see, e.g., U.S. Patent Publication No. 2017/0037462).
[0078] In some embodiments, the series of changes in conductance comprises a series of changes in conductance through a nanopore. For example, methods of evaluating receptor-ligand interactions using nanopores have been described (see, e.g., Thakur, A. K. & Movileanu, L. (2019) Nature Biotechnology 37(1)). The inventors have recognized and appreciated that such nanopores may be used to monitor polypeptide sequencing reactions in accordance with the application. Accordingly, in some embodiments, the application provides methods of polypeptide sequencing comprising contacting a single polypeptide molecule with one or more amino acid recognition molecules, where the single polypeptide molecule is immobilized to a nanopore. In some embodiments, the methods further comprise detecting a series of changes in conductance through the nanopore indicative of association of the one or more terminal amino acid recognition molecules with successive amino acids exposed at a terminus of the single polypeptide while the single polypeptide is being degraded, thereby sequencing the single polypeptide molecule.
[0079] In some aspects, the application provides methods of sequencing and/or identifying an individual protein in a complex mixture of proteins by identifying one or more types of amino acids of a polypeptide from the mixture. In some embodiments, one or more amino acids (e.g., terminal amino acids and/or internal amino acids) of the polypeptide are labeled (e.g., directly or indirectly, for example using a binding agent such as an amino acid recognition molecule) and the relative positions of the labeled amino acids in the polypeptide are determined. In some embodiments, the relative positions of amino acids in a polypeptide are determined using a series of amino acid labeling and cleavage steps. However, in some embodiments, the relative position of labeled amino acids in a polypeptide can be determined without removing amino acids from the polypeptide but by translocating a labeled polypeptide through a pore (e.g., a protein channel) and detecting a signal (e.g., a FRET signal) from the labeled amino acid(s) during translocation through the pore in order to determine the relative position of the labeled amino acids in the polypeptide molecule.
[0080] In some embodiments, the identity of a terminal amino acid (e.g., an N-terminal or a C-terminal amino acid) is assessed after which the terminal amino acid is removed and the identity of the next amino acid at the terminus is assessed, and this process is repeated until a plurality of successive amino acids in the polypeptide are assessed. In some embodiments, assessing the identity of an amino acid comprises determining the type of amino acid that is present. In some embodiments, determining the type of amino acid comprises determining the actual amino acid identity, for example by determining which of the naturally-occurring 20 amino acids is the terminal amino acid is (e.g., using a binding agent that is specific for an individual terminal amino acid). In some embodiments, the type of amino acid is selected from alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, selenocysteine, serine, threonine, tryptophan, tyrosine, and valine.
[0081] However, in some embodiments assessing the identity of a terminal amino acid type can comprise determining a subset of potential amino acids that can be present at the terminus of the polypeptide. In some embodiments, this can be accomplished by determining that an amino acid is not one or more specific amino acids (and therefore could be any of the other amino acids). In some embodiments, this can be accomplished by determining which of a specified subset of amino acids (e.g., based on size, charge, hydrophobicity, post-translational modification, binding properties) could be at the terminus of the polypeptide (e.g., using a binding agent that binds to a specified subset of two or more terminal amino acids).
[0082] In some embodiments, assessing the identity of a terminal amino acid type comprises determining that an amino acid comprises a post-translational modification. Non-limiting examples of post-translational modifications include acetylation, ADP-ribosylation, caspase cleavage, citrullination, formylation, N-linked glycosylation, O-linked glycosylation, hydroxylation, methylation, myristoylation, neddylation, nitration, oxidation, palmitoylation, phosphorylation, prenylation, S-nitrosylation, sulfation, sumoylation, and ubiquitination.
[0083] In some embodiments, assessing the identity of a terminal amino acid type comprises determining that an amino acid comprises a side chain characterized by one or more biochemical properties. For example, an amino acid may comprise a nonpolar aliphatic side chain, a positively charged side chain, a negatively charged side chain, a nonpolar aromatic side chain, or a polar uncharged side chain. Non-limiting examples of an amino acid comprising a nonpolar aliphatic side chain include alanine, glycine, valine, leucine, methionine, and isoleucine. Non-limiting examples of an amino acid comprising a positively charged side chain includes lysine, arginine, and histidine. Non-limiting examples of an amino acid comprising a negatively charged side chain include aspartate and glutamate. Non-limiting examples of an amino acid comprising a nonpolar, aromatic side chain include phenylalanine, tyrosine, and tryptophan. Non-limiting examples of an amino acid comprising a polar uncharged side chain include serine, threonine, cysteine, proline, asparagine, and glutamine.
[0084] In some embodiments, a protein or polypeptide can be digested into a plurality of smaller polypeptides and sequence information can be obtained from one or more of these smaller polypeptides (e.g., using a method that involves sequentially assessing a terminal amino acid of a polypeptide and removing that amino acid to expose the next amino acid at the terminus).
[0085] In some embodiments, a polypeptide is sequenced from its amino (N) terminus. In some embodiments, a polypeptide is sequenced from its carboxy (C) terminus. In some embodiments, a first terminus (e.g., N or C terminus) of a polypeptide is immobilized and the other terminus (e.g., the C or N terminus) is sequenced as described herein.
[0086] As used herein, sequencing a polypeptide refers to determining sequence information for a polypeptide. In some embodiments, this can involve determining the identity of each sequential amino acid for a portion (or all) of the polypeptide. However, in some embodiments, this can involve assessing the identity of a subset of amino acids within the polypeptide (e.g., and determining the relative position of one or more amino acid types without determining the identity of each amino acid in the polypeptide). However, in some embodiments amino acid content information can be obtained from a polypeptide without directly determining the relative position of different types of amino acids in the polypeptide. The amino acid content alone may be used to infer the identity of the polypeptide that is present (e.g., by comparing the amino acid content to a database of polypeptide information and determining which polypeptide(s) have the same amino acid content).
[0087] In some embodiments, sequence information for a plurality of polypeptide products obtained from a longer polypeptide or protein (e.g., via enzymatic and/or chemical cleavage) can be analyzed to reconstruct or infer the sequence of the longer polypeptide or protein.
[0088] Accordingly, in some embodiments, the one or more types of amino acids are identified by detecting luminescence of one or more labeled affinity reagents that selectively bind the one or more types of amino acids. In some embodiments, the one or more types of amino acids are identified by detecting luminescence of a labeled polypeptide.
[0089] The inventors have further recognized and appreciated that the polypeptide sequencing techniques described herein may involve generating novel polypeptide sequencing data, particularly in contrast with conventional polypeptide sequencing techniques. Thus, conventional techniques for analyzing polypeptide sequencing data may not be sufficient when applied to the data generated using the polypeptide sequencing techniques described herein.
[0090] For example, conventional polypeptide sequencing techniques that involve iterative reagent cycling may generate data associated with individual amino acids of a polypeptide being sequenced. In such instances, analyzing the data generated may simply involve determining which amino acid is being detected at a particular time because the data being detected corresponds to only one amino acid. In contrast, the polypeptide sequencing techniques described herein may generate data during a polypeptide degradation process while multiple amino acids of the polypeptide molecule are being detected, resulting in data where it may be difficult to discern between sections of the data corresponding to different amino acids of the polypeptide. Accordingly, the inventors have developed new computational techniques for analyzing such data generated by the polypeptide sequencing techniques described herein that involve determining sections of the data that correspond to individual amino acids, such as by segmenting the data into portions that correspond to respective amino acid association events. Those sections may be then further analyzed to identify the amino acid being detected during those individual sections.
[0091] As another example, conventional sequencing techniques that involve using uniquely identifiable labels for each type of amino acid may involve simply analyzing which label is being detected at a particular time without taking into consideration any dynamics in how individual amino acids interact with other molecules. In contrast, the polypeptide sequencing techniques described herein generate data indicating how amino acids interact with recognition molecules. As discussed above, the data may include a series of characteristic patterns corresponding to association events between amino acids and their respective recognition molecules. Accordingly, the inventors have developed new computational techniques for analyzing the characteristic patterns to determine a type of amino acid corresponding to that portion of the data, allowing for an amino acid sequence of a polypeptide to be determined by analyzing a series of different characteristic patterns.
Labeled Affinity Reagents and Methods of Use
[0092] In some embodiments, methods provided herein comprise contacting a polypeptide with a labeled affinity reagent (also referred to herein as an amino acid recognition molecule, which may or may not comprise a label) that selectively binds one type of terminal amino acid. As used herein, in some embodiments, a terminal amino acid may refer to an amino-terminal amino acid of a polypeptide or a carboxy-terminal amino acid of a polypeptide. In some embodiments, a labeled affinity reagent selectively binds one type of terminal amino acid over other types of terminal amino acids. In some embodiments, a labeled affinity reagent selectively binds one type of terminal amino acid over an internal amino acid of the same type. In yet other embodiments, a labeled affinity reagent selectively binds one type of amino acid at any position of a polypeptide, e.g., the same type of amino acid as a terminal amino acid and an internal amino acid.
[0093] As used herein, in some embodiments, a type of amino acid refers to one of the twenty naturally occurring amino acids or a subset of types thereof. In some embodiments, a type of amino acid refers to a modified variant of one of the twenty naturally occurring amino acids or a subset of unmodified and/or modified variants thereof. Examples of modified amino acid variants include, without limitation, post-translationally-modified variants (e.g., acetylation, ADP-ribosylation, caspase cleavage, citrullination, formylation, N-linked glycosylation, O-linked glycosylation, hydroxylation, methylation, myristoylation, neddylation, nitration, oxidation, palmitoylation, phosphorylation, prenylation, S-nitrosylation, sulfation, sumoylation, and ubiquitination), chemically modified variants, unnatural amino acids, and proteinogenic amino acids such as selenocysteine and pyrrolysine. In some embodiments, a subset of types of amino acids includes more than one and fewer than twenty amino acids having one or more similar biochemical properties. For example, in some embodiments, a type of amino acid refers to one type selected from amino acids with charged side chains (e.g., positively and/or negatively charged side chains), amino acids with polar side chains (e.g., polar uncharged side chains), amino acids with nonpolar side chains (e.g., nonpolar aliphatic and/or aromatic side chains), and amino acids with hydrophobic side chains.
[0094] In some embodiments, methods provided herein comprise contacting a polypeptide with one or more labeled affinity reagents that selectively bind one or more types of terminal amino acids. As an illustrative and non-limiting example, where four labeled affinity reagents are used in a method of the application, any one reagent selectively binds one type of terminal amino acid that is different from another type of amino acid to which any of the other three selectively binds (e.g., a first reagent binds a first type, a second reagent binds a second type, a third reagent binds a third type, and a fourth reagent binds a fourth type of terminal amino acid). For the purposes of this discussion, one or more labeled affinity reagents in the context of a method described herein may be alternatively referred to as a set of labeled affinity reagents.
[0095] In some embodiments, a set of labeled affinity reagents comprises at least one and up to six labeled affinity reagents. For example, in some embodiments, a set of labeled affinity reagents comprises one, two, three, four, five, or six labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises ten or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises eight or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises six or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises four or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises three or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises two or fewer labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises four labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises at least two and up to twenty (e.g., at least two and up to ten, at least two and up to eight, at least four and up to twenty, at least four and up to ten) labeled affinity reagents. In some embodiments, a set of labeled affinity reagents comprises more than twenty (e.g., 20 to 25, 20 to 30) affinity reagents. It should be appreciated, however, that any number of affinity reagents may be used in accordance with a method of the application to accommodate a desired use.
[0096] In accordance with the application, in some embodiments, one or more types of amino acids are identified by detecting luminescence of a labeled affinity reagent (e.g., an amino acid recognition molecule comprising a luminescent label). In some embodiments, a labeled affinity reagent comprises an affinity reagent that selectively binds one type of amino acid and a luminescent label having a luminescence that is associated with the affinity reagent. In this way, the luminescence (e.g., luminescence lifetime, luminescence intensity, and other luminescence properties described elsewhere herein) may be associated with the selective binding of the affinity reagent to identify an amino acid of a polypeptide. In some embodiments, a plurality of types of labeled affinity reagents may be used in a method according to the application, wherein each type comprises a luminescent label having a luminescence that is uniquely identifiable from among the plurality. Suitable luminescent labels may include luminescent molecules, such as fluorophore dyes, and are described elsewhere herein.
[0097] In some embodiments, one or more types of amino acids are identified by detecting one or more electrical characteristics of a labeled affinity reagent. In some embodiments, a labeled affinity reagent comprises an affinity reagent that selectively binds one type of amino acid and a conductivity label that is associated with the affinity reagent. In this way, the one or more electrical characteristics (e.g., charge, current oscillation color, and other electrical characteristics) may be associated with the selective binding of the affinity reagent to identify an amino acid of a polypeptide. In some embodiments, a plurality of types of labeled affinity reagents may be used in a method according to the application, wherein each type comprises a conductivity label that produces a change in an electrical signal (e.g., a change in conductance, such as a change in amplitude of conductivity and conductivity transitions of a characteristic pattern) that is uniquely identifiable from among the plurality. In some embodiments, the plurality of types of labeled affinity reagents each comprises a conductivity label having a different number of charged groups (e.g., a different number of negatively and/or positively charged groups). Accordingly, in some embodiments, a conductivity label is a charge label. Examples of charge labels include dendrimers, nanoparticles, nucleic acids and other polymers having multiple charged groups. In some embodiments, a conductivity label is uniquely identifiable by its net charge (e.g., a net positive charge or a net negative charge), by its charge density, and/or by its number of charged groups.
[0098] In some embodiments, an affinity reagent (e.g., an amino acid recognition molecule) may be engineered by one skilled in the art using conventionally known techniques. In some embodiments, desirable properties may include an ability to bind selectively and with high affinity to one type of amino acid only when it is located at a terminus (e.g., an N-terminus or a C-terminus) of a polypeptide. In yet other embodiments, desirable properties may include an ability to bind selectively and with high affinity to one type of amino acid when it is located at a terminus (e.g., an N-terminus or a C-terminus) of a polypeptide and when it is located at an internal position of the polypeptide. In some embodiments, desirable properties include an ability to bind selectively and with low affinity (e.g., with a K.sub.D of about 50 nM or higher, for example, between about 50 nM and about 50 .mu.M, between about 100 nM and about 10 .mu.M, between about 500 nM and about 50 .mu.M) to more than one type of amino acid. For example, in some aspects, the application provides methods of sequencing by detecting reversible binding interactions during a polypeptide degradation process. Advantageously, such methods may be performed using an affinity reagent that reversibly binds with low affinity to more than one type of amino acid (e.g., a subset of amino acid types).
[0099] As used herein, in some embodiments, the terms "selective" and "specific" (and variations thereof, e.g., selectively, specifically, selectivity, specificity) refer to a preferential binding interaction. For example, in some embodiments, a labeled affinity reagent that selectively binds one type of amino acid preferentially binds the one type over another type of amino acid. A selective binding interaction will discriminate between one type of amino acid (e.g., one type of terminal amino acid) and other types of amino acids (e.g., other types of terminal amino acids), typically more than about 10- to 100-fold or more (e.g., more than about 1,000- or 10,000-fold). Accordingly, it should be appreciated that a selective binding interaction can refer to any binding interaction that is uniquely identifiable to one type of amino acid over other types of amino acids. For example, in some aspects, the application provides methods of polypeptide sequencing by obtaining data indicative of association of one or more amino acid recognition molecules with a polypeptide molecule. In some embodiments, the data comprises a series of signal pulses corresponding to a series of reversible amino acid recognition molecule binding interactions with an amino acid of the polypeptide molecule, and the data may be used to determine the identity of the amino acid. As such, in some embodiments, a "selective" or "specific" binding interaction refers to a detected binding interaction that discriminates between one type of amino acid and other types of amino acids.
[0100] In some embodiments, a labeled affinity reagent (e.g., an amino acid recognition molecule) selectively binds one type of amino acid with a dissociation constant (K.sub.D) of less than about 10.sup.-6 M (e.g., less than about 10.sup.-7M, less than about 10.sup.-8 M, less than about 10.sup.-9 M, less than about 10.sup.-10 M, less than about 10.sup.-11M, less than about 10.sup.-12 M, to as low as 10.sup.-16M) without significantly binding to other types of amino acids. In some embodiments, a labeled affinity reagent selectively binds one type of amino acid (e.g., one type of terminal amino acid) with a K.sub.D of less than about 100 nM, less than about 50 nM, less than about 25 nM, less than about 10 nM, or less than about 1 nM. In some embodiments, a labeled affinity reagent selectively binds one type of amino acid with a K.sub.D of between about 50 nM and about 50 .mu.M (e.g., between about 50 nM and about 500 nM, between about 50 nM and about 5 .mu.M, between about 500 nM and about 50 .mu.M, between about 5 .mu.M and about 50 .mu.M, or between about 10 .mu.M and about 50 .mu.M). In some embodiments, a labeled affinity reagent selectively binds one type of amino acid with a K.sub.D of about 50 nM.
[0101] In some embodiments, a labeled affinity reagent (e.g., an amino acid recognition molecule) selectively binds two or more types of amino acids with a dissociation constant (K.sub.D) of less than about 10.sup.-6 M (e.g., less than about 10.sup.-7 M, less than about 10.sup.-8 M, less than about 10.sup.-9M, less than about 10.sup.-10 M, less than about 10.sup.-11M, less than about 10.sup.-12 M, to as low as 10.sup.-16 M). In some embodiments, a labeled affinity reagent selectively binds two or more types of amino acids with a K.sub.D of less than about 100 nM, less than about 50 nM, less than about 25 nM, less than about 10 nM, or less than about 1 nM. In some embodiments, a labeled affinity reagent selectively binds two or more types of amino acids with a K.sub.D of between about 50 nM and about 50 .mu.M (e.g., between about 50 nM and about 500 nM, between about 50 nM and about 5 .mu.M, between about 500 nM and about 50 .mu.M, between about 5 .mu.M and about 50 .mu.M, or between about 10 .mu.M and about 50 .mu.M). In some embodiments, a labeled affinity reagent selectively binds two or more types of amino acids with a K.sub.D of about 50 nM.
[0102] In accordance with the methods and compositions provided herein, FIG. 1C shows various example configurations and uses of labeled affinity reagents. In some embodiments, a labeled affinity reagent 100 comprises a luminescent label 110 (e.g., a label) and an affinity reagent (shown as stippled shapes) that selectively binds one or more types of terminal amino acids of a polypeptide 120. In some embodiments, an affinity reagent is selective for one type of amino acid or a subset (e.g., fewer than the twenty common types of amino acids) of types of amino acids at a terminal position or at both terminal and internal positions.
[0103] As described herein, an affinity reagent (also known as a "recognition molecule") may be any biomolecule capable of selectively or specifically binding one molecule over another molecule (e.g., one type of amino acid over another type of amino acid, as with an "amino acid recognition molecule" referred to herein). In some embodiments, an affinity reagent is not a peptidase or does not have peptidase activity. For example, in some embodiments, methods of polypeptide sequencing of the application involve contacting a polypeptide molecule with one or more affinity reagents and a cleaving reagent. In such embodiments, the one or more affinity reagents do not have peptidase activity, and removal of one or more amino acids from the polypeptide molecule (e.g., amino acid removal from a terminus of the polypeptide molecule) is performed by the cleaving reagent.
[0104] Affinity reagents (e.g., recognition molecules) include, for example, proteins and nucleic acids, which may be synthetic or recombinant. In some embodiments, an affinity reagent or recognition molecule may be an antibody or an antigen-binding portion of an antibody, an SH2 domain-containing protein or fragment thereof, or an enzymatic biomolecule, such as a peptidase, an aminotransferase, a ribozyme, an aptazyme, or a tRNA synthetase, including aminoacyl-tRNA synthetases and related molecules described in U.S. patent application Ser. No. 15/255,433, filed Sep. 2, 2016, titled "MOLECULES AND METHODS FOR ITERATIVE POLYPEPTIDE ANALYSIS AND PROCESSING."
[0105] In some embodiments, an affinity reagent or recognition molecule of the application is a degradation pathway protein. Examples of degradation pathway proteins suitable for use as recognition molecules include, without limitation, N-end rule pathway proteins, such as Arg/N-end rule pathway proteins, Ac/N-end rule pathway proteins, and Pro/N-end rule pathway proteins. In some embodiments, a recognition molecule is an N-end rule pathway protein selected from a Gid protein (e.g., Gid4 or Gid10 protein), a UBR box protein (e.g., UBR1, UBR2) or UBR box domain-containing protein fragment thereof, a p62 protein or ZZ domain-containing fragment thereof, and a ClpS protein (e.g., ClpS1, ClpS2).
[0106] In some embodiments, an affinity reagent or recognition molecule of the application is a ClpS protein, such as Agrobacterium tumifaciens ClpS1, Agrobacterium tumifaciens ClpS2, Synechococcus elongatus ClpS1, Synechococcus elongatus ClpS2, Thermosynechococcus elongatus ClpS, Escherichia coli ClpS, or Plasmodium falciparum ClpS. In some embodiments, the recognition molecule is an L/F transferase, such as Escherichia coli leucyl/phenylalanyl-tRNA-protein transferase. In some embodiments, the recognition molecule is a D/E leucyltransferase, such as Vibrio vulnificus Aspartate/glutamate leucyltransferase Bpt. In some embodiments, the recognition molecule is a UBR protein or UBR-box domain, such as the UBR protein or UBR-box domain of human UBR1 and UBR2 or Saccharomyces cerevisiae UBR1. In some embodiments, the recognition molecule is a p62 protein, such as H. sapiens p62 protein or Rattus norvegicus p62 protein, or truncation variants thereof that minimally include a ZZ domain. In some embodiments, the recognition molecule is a Gid4 protein, such as H. sapiens GID4 or Saccharomyces cerevisiae GID4. In some embodiments, the recognition molecule is a Gid10 protein, such as Saccharomyces cerevisiae GID10. In some embodiments, the recognition molecule is an N-meristoyltransferase, such as Leishmania major N-meristoyltransferase or H. sapiens N-meristoyltransferase NMT1. In some embodiments, the recognition molecule is a BIR2 protein, such as Drosophila melanogaster BIR2. In some embodiments, the recognition molecule is a tyrosine kinase or SH2 domain of a tyrosine kinase, such as H. sapiens Fyn SH2 domain, H. sapiens Src tyrosine kinase SH2 domain, or variants thereof, such as H. sapiens Fyn SH2 domain triple mutant superbinder. In some embodiments, the recognition molecule is an antibody or antibody fragment, such as a single-chain antibody variable fragment (scFv) against phosphotyrosine or another post-translationally modified amino acid variant described herein.
[0107] Table 1 provides a list of example sequences of amino acid recognition molecules. Also shown are the amino acid binding preferences of each molecule with respect to amino acid identity at a terminal position of a polypeptide unless otherwise specified in Table 1. It should be appreciated that these sequences and other examples described herein are meant to be non-limiting, and recognition molecules in accordance with the application can include any homologs, variants thereof, or fragments thereof minimally containing domains or subdomains responsible for peptide recognition.
TABLE-US-00001 TABLE 1 Non-limiting examples of amino acid recognition proteins. SEQ Binding ID Name Pref.* NO: Sequence Agrobacterium F, W, Y 1 MSDSPVDLKPKPKVKPKLERPKLYKVMLLNDDYTPMSFV tumifaciens ClpS2 TVVLKAVFRMSEDTGRRVMMTAHRFGSAVVVVCERDIAE variant 1 TKAKEATDLGKEAGFPLMFTTEPEE Agrobacterium F, W, Y 2 MSDSPVDLKPKPKVKPKLERPKLYKVMLLNDDYTPREFV tumifaciens ClpS2 TVVLKAVFRMSEDTGRRVMMTAHRFGSAVVVVCERDIAE TKAKEATDLGKEAGFPLMFTTEPEE Agrobacterium F, W, Y 3 MSDSPVDLKPKPKVKPKLERPKLYKVMLLNDDYTPREFV tumifaciens ClpS2 TVVLKAVFRMSEDTGRRVMMTAHRFGSAVVVVSERDIAE C71S TKAKEATDLGKEAGFPLMFTTEPEE Agrobacterium F, W, Y, L 4 MIAEPICMQGEGDGEDGGTNRGTSVITRVKPKTKRPNLY tumifaciens ClpS1 RVLLLNDDYTPMEFVIHILERFFQKDREAATRIMLHVHQ HGVGECGVFTYEVAETKVSQVMDFARQHQHPLQCVMEKK Agrobacterium F, W, Y 5 MSDSPVDLKPKPKVKPKLERPKLYKVMLLNDDYTPMSFV tumifaciens ClpS2 TVVLKAVFRMSEDTGRRVMMTAHRFGSAVVVVSERDIAE variant 1 C72S TKAKEATDLGKEAGFPLMFTTEPEE Agrobacterium F, W, Y, L 6 MIAEPISMQGEGDGEDGGTNRGTSVITRVKPKTKRPNLY tumifaciens ClpS1 RVLLLNDDYTPMEFVIHILERFFQKDREAATRIMLHVHQ C7S HGVGECGVFTYEVAETKVSQVMDFARQHQHPLQCVMEKK Agrobacterium F, W, Y, L 7 MIAEPISMQGEGDGEDGGTNRGTSVITRVKPKTKRPNLY tumifaciens ClpS1 RVLLLNDDYTPMEFVIHILERFFQKDREAATRIMLHVHQ C7SC84SC112S HGVGESGVFTYEVAETKVSQVMDFARQHQHPLQSVMEKK Agrobacterium F, W, Y 8 MSDSPVDLKPKPKVKPKLERPKLYKVILLNDDYTPMEFV tumifaciens ClpS2 VEVLKRVFNMSEEQARRVMMTAHKKGKAVVGVCPRDIAE thermostable TKAKQATDLAREAGFPLMFTTEPEE variant Agrobacterium F, W, Y 9 MSDSPVDLKPKPKVKPKLERPKLYKVILLNDDYTPMEFV tumifaciens ClpS2 VEVLKRVFNMSEEQARRVMMTAHKKGKAVVGVSPRDIAE thermostable TKAKQATDLAREAGFPLMFTTEPEE variant C72S Synechococcus F, W, Y 10 MAVETIQKPETTTKRKIAPRYRVLLHNDDFNPMEYVVMV elongatus ClpS1 LMQTVPSLTQPQAVDIMMEAHTNGTGLVITCDIEPAEFY CEQLKSHGLSSSIEPDD Synechococcus F, W, Y, L, 11 MSPQPDESVLSILGVPRPCVKKRSRNDAFVLTVLTCSLQ elongatus ClpS2 V, I AIAAPATAPGTTTTRVRQPYPHFRVIVLDDDVNTFQHVA ECLLKYIPGMTGDRAWDLTNQVHYEGAATVWSGPQEQAE LYHEQLRREGLTMAPLEAA Thermosynechococcus F, W, Y, L 12 MPQERQQVTRKHYPNYKVIVLNDDFNTFQHVAACLMKYI elongatus ClpS PNMTSDRAWELTNQVHYEGQAIVWVGPQEQAELYHEQLL RAGLTMAPLEPE Escherichia coli F, W, Y, L 13 MGKTNDWLDFDQLAEEKVRDALKPPSMYKVILVNDDYTP ClpS MEFVIDVLQKFFSYDVERATQLMLAVHYQGKAICGVFTA EVAETKVAMVNKYARENEHPLLCTLEKA Escherichia coli F, W, Y, L 14 MGKTNDWLDFDQLAEEKVRDALKPPSMYKVILVNDDYTP ClpSM40A AEFVIDVLQKFFSYDVERATQLMLAVHYQGKAICGVFTA EVAETKVAMVNKYARENEHPLLCTLEKA Plasmodium F, W, Y, L, 15 MFKDLKPFFLCIILLLLLIYKCTHSYNIKNKNCPLNFMN falciparum ClpS I SCVRINNVNKNTNISFPKELQKRPSLVYSQKNFNLEKIK KLRNVIKEIKKDNIKEADEHEKKEREKETSAWKVILYND DIHNFTYVTDVIVKVVGQISKAKAHTITVEAHSTGQALI LSTWKSKAEKYCQELQQNGLTVSIIHESQLKDKQKK Escherichia coli K, R 16 MRLVQLSRHSIAFPSPEGALREPNGLLALGGDLSPARLL leucyl/phenylalanyl MAYQRGIFPWFSPGDPILWWSPDPRAVLWPESLHISRSM -tRNA-protein KRFHKRSPYRVTMNYAFGQVIEGCASDREEGTWITRGVV transferase EAYHRLHELGHAHSIEVWREDELVGGMYGVAQGTLFCGE SMFSRMENASKTALLVFCEEFIGHGGKLIDCQVLNDHTA SLGACEIPRRDYLNYLNQMRLGRLPNNFWVPRCLFSPQE LE Vibrio vulnificus D, E 17 MSSDIHQIKIGLTDNHPCSYLPERKERVAVALEADMHTA Aspartate/glutamate DNYEVLLANGFRRSGNTIYKPHCDSCHSCQPIRISVPDI leucyltransferase ELSRSQKRLLAKARSLSWSMKRNMDENWFDLYSRYIVAR Bpt HRNGTMYPPKKDDFAHFSRNQWLTTQFLHIYEGQRLIAV AVTDIMDHCASAFYTFFEPEHELSLGTLAVLFQLEFCQE EKKQWLYLGYQIDECPAMNYKVRFHRHQKLVNQRWQ Saccharomyces K, R, H 18 MGSVHKHTGRNCGRKFKIGEPLYRCHECGCDDTCVLCIH cerevisiae UBR1 CFNPKDHVNHHVCTDICTEFTSGICDCGDEEAWNSPLHC KAEEQ H. sapiens GID4 P 19 MSGSKFRGHQKSKGNSYDVEVVLQHVDTGNSYLCGYLKI KGLTEEYPTLTTFFEGEIISKKHPFLTRKWDADEDVDRK HWGKFLAFYQYAKSFNSDDFDYEELKNGDYVFMRWKEQF LVPDHTIKDISGASFAGFYYICFQKSAASIEGYYYHRSS EWYQSLNLTHV Saccharomyces P 20 MINNPKVDSVAEKPKAVTSKQSEQAASPEPTPAPPVSRN cerevisiae GID4 QYPITFNLTSTAPFHLHDRHRYLQEQDLYKCASRDSLSS LQQLAHTPNGSTRKKYIVEDQSPYSSENPVIVTSSYNHT VCTNYLRPRMQFTGYQISGYKRYQVTVNLKTVDLPKKDC TSLSPHLSGFLSIRGLTNQHPEISTYFEAYAVNHKELGF LSSSWKDEPVLNEFKATDQTDLEHWINFPSFRQLFLMSQ KNGLNSTDDNGTTNAAKKLPPQQLPTTPSADAGNISRIF SQEKQFDNYLNERFIFMKWKEKFLVPDALLMEGVDGASY DGFYYIVHDQVTGNIQGFYYHQDAEKFQQLELVPSLKNK VESSDCSFEFA Single-chain phospho-Y 21 MMEVQLQQSGPELVKPGASVMISCRTSAYTFTENTVHWV antibody variable KQSHGESLEWIGGINPYYGGSIFSPKFKGKATLTVDKSS fragment (scFv) STAYMELRSLTSEDSAVYYCARRAGAYYFDYWGQGTTLT against VSSGGGSGGGSGGGSENVLTQSPAIMSASPGEKVTMTCR phosphotyrosine** ASSSVSSSYLHWYRQKSGASPKLWIYSTSNLASGVPARF SGSGSGTSYSLTISSVEAEDAATYYCQQYSGYRTFGGGT KLEIKR H. sapiens Fyn phospho-Y 22 MGAMDSIQAEEWYFGKLGRKDAERQLLSFGNPRGTFLIR SH2 domain** ESETTKGAYSLSIRDWDDMKGDHVKHYKIRKLDNGGYYI TTRAQFETLQQLVQHYSERAAGLSSRLVVPSHK H. sapiens Fyn phospho-Y 23 MGAMDSIQAEEWYFGKLGRKDAERQLLSFGNPRGTFLIR SH2 domain triple ESETVKGAYALSIRDWDDMKGDHVKHYLIRKLDNGGYYI mutant TTRAQFETLQQLVQHYSERAAGLSSRLVVPSHK superbinder** H. sapiens Src phospho-Y 24 MGAMDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVR tyrosine kinase ESETTKGAYSLSVSDFDNAKGLNVKHYKIRKLDSGGFYI SH2 domain** TSRTQFNSLQQLVAYYSKHADGLCHRLTTVCPTSK H. sapiens Src phospho-Y 25 MGAMDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVR tyrosine kinase ESEVTKGAYALSVSDFDNAKGLNVKHYLIRKLDSGGFYI SH2 domain triple TSRTQFNSLQQLVAYYSKHADGLCHRLTTVCPTSK mutant** H. sapiens p62 K, R, H, 26 MASLTVKAYLLGKEDAAREIRRFSFCCSPEPEAEAEAAA fragment 1-310 W, F, Y GPGPCERLLSRVAALFPALRPGGFQAHYRDEDGDLVAFS SDEELTMAMSYVKDDIFRIYIKEKKECRRDHRPPCAQEA PRNMVHPNVICDGCNGPVVGTRYKCSVCPDYDLCSVCEG KGLHRGHTKLAFPSPFGHLSEGFSHSRWLRKVKHGHFGW PGWEMGPPGNWSPRPPRAGEARPGPTAESASGPSEDPSV NFLKNVGESVAAALSPLGIEVDIDVEHGGKRSRLTPVSP ESSSTEEKSSSQPSSCCSDPSKPGGNVEGATQSLAEQ H. sapiens p62 K, R, H, 27 MASLTVKAYLLGKEDAAREIRRFSFCCSPEPEAEAEAAA fragment 1-180 W, F, Y GPGPCERLLSRVAALFPALRPGGFQAHYRDEDGDLVAFS SDEELTMAMSYVKDDIFRIYIKEKKECRRDHRPPCAQEA PRNMVHPNVICDGCNGPVVGTRYKCSVCPDYDLCSVCEG KGLHRGHTKLAFPSPFGHLSEGFSHSRWLRKVKHGHFGW PGWEMGPPGNWSPRPPRAGEARPGPTAESASGPSEDPSV NFLKNVGESVAAALSPLGIEVDIDVEHGGKRSRLTPVSP ESSSTEEKSSSQPSSCCSDPSKPGGNVEGATQSLAEQ H. sapiens p62 K, R, H, 28 MASLTVKAYLLGKEDAAREIRRFSFCCSPEPEAEAEAAA fragment 126-180 W, F, Y GPGPCERLLSRVAALFPALRPGGFQAHYRDEDGDLVAFS SDEELTMAMSYVKDDIFRIYIKEKKECRRDHRPPCAQEA PRNMVHPNVICDGCNGPVVGTRYKCSVCPDYDLCSVCEG KGLHRGHTKLAFPSPFGHLSEGFSHSRWLRKVKHGHFGW PGWEMGPPGNWSPRPPRAGEARPGPTAESASGPSEDPSV NFLKNVGESVAAALSPLGIEVDIDVEHGGKRSRLTPVSP ESSSTEEKSSSQPSSCCSDPSKPGGNVEGATQSLAEQ H. sapiens p62 K, R, H, 29 MASLTVKAYLLGKEDAAREIRRFSFCCSPEPEAEAEAAA protein W, F, Y GPGPCERLLSRVAALFPALRPGGFQAHYRDEDGDLVAFS SDEELTMAMSYVKDDIFRIYIKEKKECRRDHRPPCAQEA PRNMVHPNVICDGCNGPVVGTRYKCSVCPDYDLCSVCEG KGLHRGHTKLAFPSPFGHLSEGFSHSRWLRKVKHGHFGW PGWEMGPPGNWSPRPPRAGEARPGPTAESASGPSEDPSV NFLKNVGESVAAALSPLGIEVDIDVEHGGKRSRLTPVSP ESSSTEEKSSSQPSSCCSDPSKPGGNVEGATQSLAEQMR KIALESEGRPEEQMESDNCSGGDDDWTHLSSKEVDPSTG ELQSLQMPESEGPSSLDPSQEGPTGLKEAALYPHLPPEA DPRLIESLSQMLSMGFSDEGGWLTRLLQTKNYDIGAALD TIQYSKHPPPL Rattus norvegicus K, R, H, 30 MASLTVKAYLLGKEEAAREIRRFSFCFSPEPEAEAAAGP p62 protein W, F, Y GPCERLLSRVAVLFPALRPGGFQAHYRDEDGDLVAFSSD EELTMAMSYVKDDIFRIYIKEKKECRREHRPPCAQEARS MVHPNVICDGCNGPVVGTRYKCSVCPDYDLCSVCEGKGL HREHSKLIFPNPFGHLSDSFSHSRWLRKLKHGHFGWPGW EMGPPGNWSPRPPRAGDGRPCPTAESASAPSEDPNVNFL KNVGESVAAALSPLGIEVDIDVEHGGKRSRLTPTSAESS STGTEDKSGTQPSSCSSEVSKPDGAGEGPAQSLTEQMKK IALESVGQPEELMESDNCSGGDDDWTHLSSKEVDPSTGE LQSLQMPESEGPSSLDPSQEGPTGLKEAALYPHLPPEAD PRLIESLSQMLSMGFSDEGGWLTRLLQTKNYDIGAALDT IQYSKHPPPL Saccharomyces P, M, V 31 MTSLNIMGRKFILERAKRNDNIEEIYTSAYVSLPSSTDT cerevisiae GID10 RLPHFKAKEEDCDVYEEGTNLVGKNAKYTYRSLGRHLDF LRPGLRFGGSQSSKYTYYTVEVKIDTVNLPLYKDSRSLD PHVTGTFTIKNLTPVLDKVVTLFEGYVINYNQFPLCSLH WPAEETLDPYMAQRESDCSHWKRFGHFGSDNWSLTERNF GQYNHESAEFMNQRYTYLKWKERFLLDDEEQENQMLDDN HHLEGASFEGFYYVCLDQLTGSVEGYYYHPACELFQKLE LVPTNCDALNTYSSGFEIA UBR-box domain K, R, H 32 MGPLGSLCGRVFKSGETTYSCRDCAIDPTCVLCMDCFQD from Homo sapiens SVHKNHRYKMHTSTGGGFCDCGDTEAWKTGPFCVNHEP UBR1 UBR-box domain K, R, H 33 MGPLGSLCGRVFKVGEPTYSCRDCAVDPTCVLCMECFLG from Homo sapiens SIHRDHRYRMTTSGGGGFCDCGDTEAWKEGPYCQKHE UBR2 Leishmania major G 34 MSRNPSNSDAAHAFWSTQPVPQTEDETEKIVFAGPMDEP N- KTVADIPEEPYPIASTFEWWTPNMEAADDIHAIYELLRD meristoyltransferase NYVEDDDSMFRFNYSEEFLQWALCPPNYIPDWHVAVRRK ADKKLLAFIAGVPVTLRMGTPKYMKVKAQEKGEGEEAAK YDEPRHICEINFLCVHKQLREKRLAPILIKEATRRVNRT NVWQAVYTAGVLLPTPYASGQYFHRSLNPEKLVEIRFSG IPAQYQKFQNPMAMLKRNYQLPSAPKNSGLREMKPSDVP QVRRILMNYLDSFDVGPVFSDAEISHYLLPRDGVVFTYV VENDKKVTDFFSFYRIPSTVIGNSNYNLLNAAYVHYYAA TSIPLHQLILDLLIVAHSRGFDVCNMVEILDNRSFVEQL KFGAGDGHLRYYFYNWAYPKIKPSQVALVML H. sapiens N- G 35 MADESETAVKPPAPPLPQMMEGNGNGHEHCSDCENEEDN meristoyltransferase SYNRGGLSPANDTGAKKKKKKQKKKKEKGSETDSAQDQP NMT1 VKMNSLPAERIQEIQKAIELFSVGQGPAKTMEEASKRSY QFWDTQPVPKLGEVVNTHGPVEPDKDNIRQEPYTLPQGF TWDALDLGDRGVLKELYTLLNENYVEDDDNMFRFDYSPE FLLWALRPPGWLPQWHCGVRVVSSRKLVGFISAIPANTH IYDTEKKMVEINFLCVHKKLRSKRVAPVLIREITRRVHL EGIFQAVYTAGVVLPKPVGTCRYWHRSLNPRKLIEVKFS HLSRNMTMQRTMKLYRLPETPKTAGLRPMETKDIPVVHQ LLTRYLKQFHLTPVMSQEEVEHWFYPQENIIDTFVVENA NGEVTDFLSFYTLPSTIMNHPTHKSLKAAYSFYNVHTQT PLLDLMSDALVLAKMKGFDVFNALDLMENKTFLEKLKFG IGDGNLQYYLYNWKCPSMGAEKVGLVLQ Drosophila A 36 MGDVQPETCRPSAASGNYFPQYPEYAIETARLRTFEAWP melanogaster BIR2 RNLKQKPHQLAEAGFFYTGVGDRVRCFSCGGGLMDWNDN DEPWEQHALWLSQCRFVKLMKGQLYIDTVAAKPVLAEEK EESTSIGGDT *Binding preferences are inferred from published scientific literature and/or further demonstrated by the inventors in single-molecule experiments, as described herein. **Binding to phosphotyrosine may occur at a peptide terminus or at an internal position.
[0108] Accordingly, in some embodiments, the application provides an amino acid recognition molecule having an amino acid sequence selected from Table 1 (or having an amino acid sequence that has at least 50%, at least 60%, at least 70%, at least 80%, 80-90%, 90-95%, 95-99%, or higher, amino acid sequence identity to an amino acid sequence selected from Table 1). In some embodiments, an amino acid recognition molecule has 25-50%, 50-60%, 60-70%, 70-80%, 80-90%, 90-95%, or 95-99%, or higher, amino acid sequence identity to an amino acid recognition molecule listed in Table 1. In some embodiments, an amino acid recognition molecule is a modified amino acid recognition molecule and includes one or more amino acid mutations relative to a sequence set forth in Table 1.
[0109] In some embodiments, an amino acid recognition molecule comprises a tag sequence that provides one or more functions other than amino acid binding. For example, in some embodiments, a tag sequence comprises a biotin ligase recognition sequence that permits biotinylation of the recognition molecule (e.g., incorporation of one or more biotin molecules, including biotin and bis-biotin moieties). Additional examples of functional sequences in a tag sequence include purification tags, cleavage sites, and other moieties useful for purification and/or modification of recognition molecules. Table 2 provides a list of non-limiting sequences of terminal tag sequences, any one or more of which may be used in combination with any one of the amino acid recognition molecules of the application (e.g., in combination with a sequence set forth in Table 1). It should be appreciated that the tag sequences shown in Table 2 are meant to be non-limiting, and recognition molecules in accordance with the application can include any one or more of the tag sequences (e.g., His-tags and/or biotinylation tags) at the N- or C-terminus of a recognition molecule polypeptide, split between the N- and C-terminus, or otherwise rearranged as practiced in the art.
TABLE-US-00002 TABLE 2 Non-limiting examples of terminal tag sequences. SEQ Name ID NO: Sequence Biotinylation tag 37 GGGSGGGSGGGSGLNDFFEAQKIEWHE Bis-biotinylation tag 38 GGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGGGSGGGSGLNDFFE AQKIEWHE Bis-biotinylation tag 39 GSGGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGGGSGGGSGLNDF FEAQKIEWHE His/biotinylation tag 40 GHHHHHHHHHHGGGSGGGSGGGSGLNDFFEAQKIEWHE His/bis-biotinylation 41 GHHHHHHHHHHGGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGGGS tag GGGSGLNDFFEAQKIEWHE His/bis-biotinylation 42 GGSHHHHHHHHHHGGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGG tag GSGGGSGLNDFFEAQKIEWHE His/bis-biotinylation 43 GSHHHHHHHHHHGGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGGG tag SGGGSGLNDFFEAQKIEWHE Bis-biotinylation/His 44 GGGSGGGSGGGSGLNDFFEAQKIEWHEGGGSGGGSGGGSGLNDFFE tag AQKIEWHEGHHHHHH
[0110] In some embodiments, a recognition molecule or affinity reagent of the application is a peptidase. A peptidase, also referred to as a protease or proteinase, is an enzyme that catalyzes the hydrolysis of a peptide bond. Peptidases digest polypeptides into shorter fragments and may be generally classified into endopeptidases and exopeptidases, which cleave a polypeptide chain internally and terminally, respectively. In some embodiments, labeled affinity reagent 100 comprises a peptidase that has been modified to inactivate exopeptidase or endopeptidase activity. In this way, labeled affinity reagent 100 selectively binds without also cleaving the amino acid from a polypeptide. In yet other embodiments, a peptidase that has not been modified to inactivate exopeptidase or endopeptidase activity may be used. For example, in some embodiments, a labeled affinity reagent comprises a labeled exopeptidase 102.
[0111] In accordance with certain embodiments of the application, protein sequencing methods may comprise iterative detection and cleavage at a terminal end of a polypeptide. In some embodiments, labeled exopeptidase 102 may be used as a single reagent that performs both steps of detection and cleavage of an amino acid. As generically depicted, in some embodiments, labeled exopeptidase 102 has aminopeptidase or carboxypeptidase activity such that it selectively binds and cleaves an N-terminal or C-terminal amino acid, respectively, from a polypeptide. It should be appreciated that, in certain embodiments, labeled exopeptidase 102 may be catalytically inactivated by one skilled in the art such that labeled exopeptidase 102 retains selective binding properties for use as a non-cleaving labeled affinity reagent 100, as described herein.
[0112] An exopeptidase generally requires a polypeptide substrate to comprise at least one of a free amino group at its amino-terminus or a free carboxyl group at its carboxy-terminus. In some embodiments, an exopeptidase in accordance with the application hydrolyses a bond at or near a terminus of a polypeptide. In some embodiments, an exopeptidase hydrolyses a bond not more than three residues from a polypeptide terminus. For example, in some embodiments, a single hydrolysis reaction catalyzed by an exopeptidase cleaves a single amino acid, a dipeptide, or a tripeptide from a polypeptide terminal end.
[0113] In some embodiments, an exopeptidase in accordance with the application is an aminopeptidase or a carboxypeptidase, which cleaves a single amino acid from an amino- or a carboxy-terminus, respectively. In some embodiments, an exopeptidase in accordance with the application is a dipeptidyl-peptidase or a peptidyl-dipeptidase, which cleave a dipeptide from an amino- or a carboxy-terminus, respectively. In yet other embodiments, an exopeptidase in accordance with the application is a tripeptidyl-peptidase, which cleaves a tripeptide from an amino-terminus. Peptidase classification and activities of each class or subclass thereof is well known and described in the literature (see, e.g., Gurupriya, V. S. & Roy, S. C. Proteases and Protease Inhibitors in Male Reproduction. Proteases in Physiology and Pathology 195-216 (2017); and Brix, K. & Stocker, W. Proteases: Structure and Function. Chapter 1). In some embodiments, a peptidase in accordance with the application removes more than three amino acids from a polypeptide terminus. Accordingly, in some embodiments, the peptidase is an endopeptidase, e.g., that cleaves preferentially at particular positions (e.g., before or after a particular amino acid). In some embodiments, the size of a polypeptide cleavage product of endopeptidase activity will depend on the distribution of cleavage sites (e.g., amino acids) within the polypeptide being analyzed.
[0114] An exopeptidase in accordance with the application may be selected or engineered based on the directionality of a sequencing reaction. For example, in embodiments of sequencing from an amino-terminus to a carboxy-terminus of a polypeptide, an exopeptidase comprises aminopeptidase activity. Conversely, in embodiments of sequencing from a carboxy-terminus to an amino-terminus of a polypeptide, an exopeptidase comprises carboxypeptidase activity. Examples of carboxypeptidases that recognize specific carboxy-terminal amino acids, which may be used as labeled exopeptidases or inactivated to be used as non-cleaving labeled affinity reagents described herein, have been described in the literature (see, e.g., Garcia-Guerrero, M. C., et al. (2018) PNAS 115(17)).
[0115] Suitable peptidases for use as cleaving reagents and/or affinity reagents (e.g., recognition molecules) include aminopeptidases that selectively bind one or more types of amino acids. In some embodiments, an aminopeptidase recognition molecule is modified to inactivate aminopeptidase activity. In some embodiments, an aminopeptidase cleaving reagent is non-specific such that it cleaves most or all types of amino acids from a terminal end of a polypeptide. In some embodiments, an aminopeptidase cleaving reagent is more efficient at cleaving one or more types of amino acids from a terminal end of a polypeptide as compared to other types of amino acids at the terminal end of the polypeptide. For example, an aminopeptidase in accordance with the application specifically cleaves alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, selenocysteine, serine, threonine, tryptophan, tyrosine, and/or valine. In some embodiments, an aminopeptidase is a proline aminopeptidase. In some embodiments, an aminopeptidase is a proline iminopeptidase. In some embodiments, an aminopeptidase is a glutamate/aspartate-specific aminopeptidase. In some embodiments, an aminopeptidase is a methionine-specific aminopeptidase. In some embodiments, an aminopeptidase is an aminopeptidase set forth in Table 3. In some embodiments, an aminopeptidase cleaving reagent cleaves a peptide substrate as set forth in Table 3.
[0116] In some embodiments, an aminopeptidase is a non-specific aminopeptidase. In some embodiments, a non-specific aminopeptidase is a zinc metalloprotease. In some embodiments, a non-specific aminopeptidase is an aminopeptidase set forth in Table 4. In some embodiments, a non-specific aminopeptidase cleaves a peptide substrate as set forth in Table 4.
[0117] Accordingly, in some embodiments, the application provides an aminopeptidase (e.g., an aminopeptidase recognition molecule, an aminopeptidase cleaving reagent) having an amino acid sequence selected from Table 3 or Table 4 (or having an amino acid sequence that has at least 50%, at least 60%, at least 70%, at least 80%, 80-90%, 90-95%, 95-99%, or higher, amino acid sequence identity to an amino acid sequence selected from Table 3 or Table 4). In some embodiments, an aminopeptidase has 25-50%, 50-60%, 60-70%, 70-80%, 80-90%, 90-95%, or 95-99%, or higher, amino acid sequence identity to an aminopeptidase listed in Table 3 or Table 4. In some embodiments, an aminopeptidase is a modified aminopeptidase and includes one or more amino acid mutations relative to a sequence set forth in Table 3 or Table 4.
TABLE-US-00003 TABLE 3 Non-limiting examples of aminopeptidases. SEQ Name ID NO: Sequence L. pneumophila M1 45 MMVKQGVFMKTDQSKVKKLSDYKSLDYFVIHVDLQIDLSKKPVESK Aminopeptidase ARLTVVPNLNVDSHSNDLVLDGENMTLVSLQMNDNLLKENEYELTK (Glu/Asp Specific) DSLIIKNIPQNTPFTIEMTSLLGENTDLFGLYETEGVALVKAESEG LRRVFYLPDRPDNLATYKTTIIANQEDYPVLLSNGVLIEKKELPLG LHSVTWLDDVPKPSYLFALVAGNLQRSVTYYQTKSGRELPIEFYVP PSATSKCDFAKEVLKEAMAWDERTFNLECALRQHMVAGVDKYASGA SEPTGLNLFNTENLFASPETKTDLGILRVLEVVAHEFFHYWSGDRV TIRDWENLPLKEGLITFRAAMFREELFGTDLIRLLDGKNLDERAPR QSAYTAVRSLYTAAAYEKSADIFRMMMLFIGKEPFIEAVAKFFKDN DGGAVTLEDFIESISNSSGKDLRSFLSWFTESGIPELIVTDELNPD TKQYFLKIKTVNGRNRPIPILMGLLDSSGAEIVADKLLIVDQEEIE FQFENIQTRPIPSLLRSFSAPVHMKYEYSYQDLLLLMQFDTNLYNR CEAAKQLISALINDFCIGKKIELSPQFFAVYKALLSDNSLNEWMLA ELITLPSLEELIENQDKPDFEKLNEGRQLIQNALANELKTDFYNLL FRIQISGDDDKQKLKGFDLKQAGLRRLKSVCFSYLLNVDFEKTKEK LILQFEDALGKNMTETALALSMLCEINCEEADVALEDYYHYWKNDP GAVNNWFSIQALAHSPDVIERVKKLMRHGDFDLSNPNKVYALLGSF IKNPFGFHSVTGEGYQLVADAIFDLDKINPTLAANLTEKFTYWDKY DVNRQAMMISTLKIIYSNATSSDVRTMAKKGLDKVKEDLPLPIHLT FHGGSTMQDRTAQLIADGNKENAYQLH E. coli methionine 46 MGTAISIKTPEDIEKMRVAGRLAAEVLEMIEPYVKPGVSTGELDRI aminopeptidase CNDYIVNEQHAVSACLGYHGYPKSVCISINEVVCHGIPDDAKLLKD (Met specific) GDIVNIDVIVIKDGFHGDTSKMFIVGKPTIMGERLCRITQESLYLA LRMVKPGINLREIGAAIQKFVEAEGFSVVREYCGHGIGRGFHEEPQ VLHYDSRETNVVLKPGMTFTIEPMVNAGKKEIRTMKDGWTVKTKDR SLSAQYEHTIVVTDNGCEILTLRKDDTIPAIISHD M. smegmatis Proline 47 MGTLEANTNGPGSMLSRMPVSSRTVPFGDHETWVQVTTPENAQPHA iminopeptidase LPLIVLHGGPGMAHNYVANIAALADETGRTVIHYDQVGCGNSTHLP (Pro specific) DAPADFWTPQLFVDEFHAVCTALGIERYHVLGQSWGGMLGAEIAVR QPSGLVSLAICNSPASMRLWSEAAGDLRAQLPAETRAALDRHEAAG TITHPDYLQAAAEFYRRHVCRVVPTPQDFADSVAQMEAEPTVYHTM NGPNEFHVVGTLGDWSVIDRLPDVTAPVLVIAGEHDEATPKTWQPF VDHIPDVRSHVFPGTSHCTHLEKPEEFRAVVAQFLHQHDLAADARV Y. pestis Proline 48 MTQQEYQNRRQALLAKMAPGSAAIIFAAPEATRSADSEYPYRQNSD iminopeptidase FSYLTGFNEPEAVLILVKSDETHNHSVLFNRIRDLTAEIWFGRRLG (Pro Specific) QEAAPTKLAVDRALPFDEINEQLYLLLNRLDVIYHAQGQYAYADNI VFAALEKLRHGFRKNLRAPATLTDWRPWLHEMRLFKSAEEIAVLRR AGEISALAHTRAMEKCRPGMFEYQLEGEILHEFTRHGARYPAYNTI VGGGENGCILHYTENECELRDGDLVLIDAGCEYRGYAGDITRTFPV NGKFTPAQRAVYDIVLAAINKSLTLFRPGTSIREVTEEVVRIMVVG LVELGILKGDIEQLIAEQAHRPFFMHGLSHWLGMDVHDVGDYGSSD RGRILEPGMVLTVEPGLYIAPDADVPPQYRGIGIRIEDDIVITATG NENLTASVVKDPDDIEALMALNHAGENLYFQE P. furiosus 49 MDTEKLMKAGEIAKKVREKAIKLARPGMLLLELAESIEKMIMELGG methionine KPAFPVNLSINEIAAHYTPYKGDTTVLKEGDYLKIDVGVHIDGFIA aminopeptidase DTAVTVRVGMEEDELMEAAKEALNAAISVARAGVEIKELGKAIENE IRKRGFKPIVNLSGHKIERYKLHAGISIPNIYRPHDNYVLKEGDVF AIEPFATIGAGQVIEVPPTLIYMYVRDVPVRVAQARFLLAKIKREY GTLPFAYRWLQNDMPEGQLKLALKTLEKAGAIYGYPVLKEIRNGIV AQFEHTIIVEKDSVIVTQDMINKSTLE Aeromonas sobria 50 HMSSPLHYVLDGIHCEPHFFTVPLDHQQPDDEETITLFGRTLCRKD Proline RLDDELPWLLYLQGGPGFGAPRPSANGGWIKRALQEFRVLLLDQRG aminopeptidase TGHSTPIHAELLAHLNPRQQADYLSHFRADSIVRDAELIREQLSPD HPWSLLGQSFGGFCSLTYLSLFPDSLHEVYLTGGVAPIGRSADEVY RATYQRVADKNRAFFARFPHAQAIANRLATHLQRHDVRLPNGQRLT VEQLQQQGLDLGASGAFEELYYLLEDAFIGEKLNPAFLYQVQAMQP FNTNPVFAILHELIYCEGAASHWAAERVRGEFPALAWAQGKDFAFT GEMIFPWMFEQFRELIPLKEAAHLLAEKADWGPLYDPVQLARNKVP VACAVYAEDMYVEFDYSRETLKGLSNSRAWITNEYEHNGLRVDGEQ ILDRLIRLNRDCLE Pyrococcus furiosus 51 MKERLEKLVKFMDENSIDRVFIAKPVNVYYFSGTSPLGGGYIIVDG Proline DEATLYVPELEYEMAKEESKLPVVKFKKEDEIYEILKNTETLGIEG Aminopeptidase (X-/- TLSYSMVENFKEKSNVKEFKKIDDVIKDLRIIKTKEEIEIIEKACE Pro) IADKAVMAAIEEITEGKREREVAAKVEYLMKMNGAEKPAFDTIIAS GHRSALPHGVASDKRIERGDLVVIDLGALYNHYNSDITRTIVVGSP NEKQREIYEIVLEAQKRAVEAAKPGMTAKELDSIAREIIKEYGYGD YFIHSLGHGVGLEIHEWPRISQYDETVLKEGMVITIEPGIYIPKLG GVRIEDTVLITENGAKRLTKTERELL Elizabethkingia 52 MIPITTPVGNFKVWTKRFGINPKIKVLLLHGGPAMTHEYMECFETF meningoseptica FQREGFEEYEYDQLGSYYSDQPTDEKLWNIDRFVDEVEQVRKAIHA Proline DKENFYVLGNSWGGILAMEYALKYQQNLKGLIVANMMASAPEYVKY aminopeptidase AEVLSKQMKPEVLAEVRAIEAKKDYANPRYTELLFPNYYAQHICRL KEWPDALNRSLKHVNSTVYTLMQGPSELGMSSDARLAKWDIKNRLH EIATPTLMIGARYDTMDPKAMEEQSKLVQKGRYLYCPNGSHLAMWD DQKVFMDGVIKFIKDVDTKSFN N. gonorrhoeae 53 MYEIKQPFHSGYLQVSEIHQIYWEESGNPDGVPVIFLHGGPGAGAS Proline PECRGFFNPDVFRIVIIDQRGCGRSHPYACAEDNTTWDLVADIEKV Iminopeptidase REMLGIGKWLVFGGSWGSTLSLAYAQTHPERVKGLVLRGIFLCRPS ETAWLNEAGGVSRIYPEQWQKFVAPIAENRRNRLIEAYHGLLFHQD EEVCLSAAKAWADWESYLIRFEPEGVDEDAYASLAIARLENHYFVN GGWLQGDKAILNNIGKIRHIPTVIVQGRYDLCTPMQSAWELSKAFP EAELRVVQAGHCAFDPPLADALVQAVEDILPRLL
TABLE-US-00004 TABLE 4 Non-limiting examples of non-specific aminopeptidases. SEQ Name ID NO: Sequence E. coli 54 MTQQPQAKYRHDYRAPDYQITDIDLTFDLDAQKTVVTAVSQAVRHG Aminopeptidase N* ASDAPLRLNGEDLKLVSVHINDEPWTAWKEEEGALVISNLPERFTL (Zinc KIINEISPAANTALEGLYQSGDALCTQCEAEGFRHITYYLDRPDVL Metalloprotease) ARFTTKIIADKIKYPFLLSNGNRVAQGELENGRHWVQWQDPFPKPC YLFALVAGDFDVLRDTFTTRSGREVALELYVDRGNLDRAPWAMTSL KNSMKWDEERFGLEYDLDIYMIVAVDFFNMGAMENKGLNIFNSKYV LARTDTATDKDYLDIERVIGHEYFHNWTGNRVTCRDWFQLSLKEGL TVFRDQEFSSDLGSRAVNRINNVRTMRGLQFAEDASPMAHPIRPDM VIEMNNFYTLTVYEKGAEVIRMIHTLLGEENFQKGMQLYFERHDGS AATCDDFVQAMEDASNVDLSHFRRWYSQSGTPIVTVKDDYNPETEQ YTLTISQRTPATPDQAEKQPLHIPFAIELYDNEGKVIPLQKGGHPV NSVLNVTQAEQTFVFDNVYFQPVPALLCEFSAPVKLEYKWSDQQLT FLMRHARNDFSRWDAAQSLLATYIKLNVARHQQGQPLSLPVHVADA FRAVLLDEKIDPALAAEILTLPSVNEMAELFDIIDPIAIAEVREAL TRTLATELADELLAIYNANYQSEYRVEHEDIAKRTLRNACLRFLAF GETHLADVLVSKQFHEANNMTDALAALSAAVAAQLPCRDALMQEYD DKWHQNGLVMDKWFILQATSPAANVLETVRGLLQHRSFTMSNPNRI RSLIGAFAGSNPAAFHAEDGSGYLFLVEMLTDLNSRNPQVASRLIE PLIRLKRYDAKRQEKMRAALEQLKGLENLSGDLYEKITKALA P. falciparum M1 55 PKIHYRKDYKPSGFIINQVTLNINIHDQETIVRSVLDMDISKHNVG aminopeptidase** EDLVFDGVGLKINEISINNKKLVEGEEYTYDNEFLTIFSKFVPKSK FAFSSEVIIHPETNYALTGLYKSKNIIVSQCEATGFRRITFFIDRP DMMAKYDVTVTADKEKYPVLLSNGDKVNEFEIPGGRHGARFNDPPL KPCYLFAVVAGDLKHLSATYITKYTKKKVELYVFSEEKYVSKLQWA LECLKKSMAFDEDYFGLEYDLSRLNLVAVSDFNVGAMENKGLNIFN ANSLLASKKNSIDFSYARILTVVGHEYFHQYTGNRVTLRDWFQLTL KEGLTVHRENLFSEEMTKTVTTRLSHVDLLRSVQFLEDSSPLSHPI RPESYVSMENFYTTTVYDKGSEVMRMYLTILGEEYYKKGFDIYIKK NDGNTATCEDFNYAMEQAYKMKKADNSANLNQYLLWFSQSGTPHVS FKYNYDAEKKQYSIHVNQYTKPDENQKEKKPLFIPISVGLINPENG KEMISQTTLELTKESDTFVFNNIAVKPIPSLFRGFSAPVYIEDQLT DEERILLLKYDSDAFVRYNSCTNIYMKQILMNYNEFLKAKNEKLES FQLTPVNAQFIDAIKYLLEDPHADAGFKSYIVSLPQDRYIINFVSN LDTDVLADTKEYIYKQIGDKLNDVYYKMFKSLEAKADDLTYFNDES HVDFDQMNMRTLRNTLLSLLSKAQYPNILNEIIEHSKSPYPSNWLT SLSVSAYFDKYFELYDKTYKLSKDDELLLQEWLKTVSRSDRKDIYE ILKKLENEVLKDSKNPNDIRAVYLPFTNNLRRFHDISGKGYKLIAE VITKTDKFNPMVATQLCEPFKLWNKLDTKRQELMLNEMNTMLQEPQ ISNNLKEYLLRLTNK Puromycin-sensitive 56 MWLAAAAPSLARRLLFLGPPPPPLLLLVFSRSSRRRLHSLGLAAMP aminopeptidase EKRPFERLPADVSPINYSLCLKPDLLDFTFEGKLEAAAQVRQATNQ ("NPEPPS") IVMNCADIDIITASYAPEGDEEIHATGFNYQNEDEKVTLSFPSTLQ TGTGTLKIDFVGELNDKMKGFYRSKYTTPSGEVRYAAVTQFEATDA RRAFPCWDEPAIKATFDISLVVPKDRVALSNMNVIDRKPYPDDENL VEVKFARTPVMSTYLVAFVVGEYDFVETRSKDGVCVRVYTPVGKAE QGKFALEVAAKTLPFYKDYFNVPYPLPKIDLIAIADFAAGAMENWG LVTYRETALLIDPKNSCSSSRQWVALVVGHELAHQWFGNLVTMEWW THLWLNEGFASWIEYLCVDHCFPEYDIWTQFVSADYTRAQELDALD NSHPIEVSVGHPSEVDEIFDAISYSKGASVIRMLHDYIGDKDFKKG MNMYLTKFQQKNAATEDLWESLENASGKPIAAVMNTWTKQMGFPLI YVEAEQVEDDRLLRLSQKKFCAGGSYVGEDCPQWMVPITISTSEDP NQAKLKILMDKPEMNVVLKNVKPDQWVKLNLGTVGFYRTQYSSAML ESLLPGIRDLSLPPVDRLGLQNDLFSLARAGIISTVEVLKVMEAFV NEPNYTVWSDLSCNLGILSTLLSHTDFYEEIQEFVKDVFSPIGERL GWDPKPGEGHLDALLRGLVLGKLGKAGHKATLEEARRRFKDHVEGK QILSADLRSPVYLTVLKHGDGTTLDIMLKLHKQADMQEEKNRIERV LGATLLPDLIQKVLTFALSEEVRPQDTVSVIGGVAGGSKHGRKAAW KFIKDNWEELYNRYQGGFLISRLIKLSVEGFAVDKMAGEVKAFFES HPAPSAERTIQQCCENILLNAAWLKRDAESIHQYLLQRKASPPTV NPEPPS E366V 57 MWLAAAAPSLARRLLFLGPPPPPLLLLVFSRSSRRRLHSLGLAAMP EKRPFERLPADVSPINYSLCLKPDLLDFTFEGKLEAAAQVRQATNQ IVMNCADIDIITASYAPEGDEEIHATGFNYQNEDEKVTLSFPSTLQ TGTGTLKIDFVGELNDKMKGFYRSKYTTPSGEVRYAAVTQFEATDA RRAFPCWDEPAIKATFDISLVVPKDRVALSNMNVIDRKPYPDDENL VEVKFARTPVMSTYLVAFVVGEYDFVETRSKDGVCVRVYTPVGKAE QGKFALEVAAKTLPFYKDYFNVPYPLPKIDLIAIADFAAGAMENWG LVTYRETALLIDPKNSCSSSRQWVALVVGHVLAHQWFGNLVTMEWW THLWLNEGFASWIEYLCVDHCFPEYDIWTQFVSADYTRAQELDALD NSHPIEVSVGHPSEVDEIFDAISYSKGASVIRMLHDYIGDKDFKKG MNMYLTKFQQKNAATEDLWESLENASGKPIAAVMNTWTKQMGFPLI YVEAEQVEDDRLLRLSQKKFCAGGSYVGEDCPQWMVPITISTSEDP NQAKLKILMDKPEMNVVLKNVKPDQWVKLNLGTVGFYRTQYSSAML ESLLPGIRDLSLPPVDRLGLQNDLFSLARAGIISTVEVLKVMEAFV NEPNYTVWSDLSCNLGILSTLLSHTDFYEEIQEFVKDVESPIGERL GWDPKPGEGHLDALLRGLVLGKLGKAGHKATLEEARRRFKDHVEGK QILSADLRSPVYLTVLKHGDGTTLDIMLKLHKQADMQEEKNRIERV LGATLLPDLIQKVLTFALSEEVRPQDTVSVIGGVAGGSKHGRKAAW KFIKDNWEELYNRYQGGFLISRLIKLSVEGFAVDKMAGEVKAFFES HPAPSAERTIQQCCENILLNAAWLKRDAESIHQYLLQRKASPPTV Francisella tularensis 58 MIYEFVMTDPKIKYLKDYKPSNYLIDEIHLIFELDESKTRVTANLY Aminopeptidase N IVANRENRENNTLVLDGVELKLLSIKLNNKHLSPAEFAVNENQLII NNVPEKFVLQTVVEINPSANTSLEGLYKSGDVFSTQCEATGFRKIT YYLDRPDVMAAFTVKIIADKKKYPIILSNGDKIDSGDISDNQHFAV WKDPFKKPCYLFALVAGDLASIKDTYITKSQRKVSLEIYAFKQDID KCHYAMQAVKDSMKWDEDRFGLEYDLDTFMIVAVPDFNAGAMENKG LNIFNTKYIMASNKTATDKDFELVQSVVGHEYFHNWTGDRVTCRDW FQLSLKEGLTVFRDQEFTSDLNSRDVKRIDDVRIIRSAQFAEDASP MSHPIRPESYIEMNNFYTVTVYNKGAEIIRMIHTLLGEEGFQKGMK LYFERHDGQAVTCDDFVNAMADANNRDFSLFKRWYAQSGTPNIKVS ENYDASSQTYSLTLEQTTLPTADQKEKQALHIPVKMGLINPEGKNI AEQVIELKEQKQTYTFENIAAKPVASLFRDFSAPVKVEHKRSEKDL LHIVKYDNNAFNRWDSLQQIATNIILNNADLNDEFLNAFKSILHDK DLDKALISNALLIPIESTIAEAMRVIMVDDIVLSRKNVVNQLADKL KDDWLAVYQQCNDNKPYSLSAEQIAKRKLKGVCLSYLMNASDQKVG TDLAQQLFDNADNMTDQQTAFTELLKSNDKQVRDNAINEFYNRWRH EDLVVNKWLLSQAQISHESALDIVKGLVNHPAYNPKNPNKVYSLIG GFGANFLQYHCKDGLGYAFMADTVLALDKFNHQVAARMARNLMSWK RYDSDRQAMMKNALEKIKASNPSKNVFEIVSKSLES Pyrococcus 59 MEVRNMVDYELLKKVVEAPGVSGYEFLGIRDVVIEEIKDYVDEVKV horikoshii TET DKLGNVIAHKKGEGPKVMIAAHMDQIGLMVTHIEKNGFLRVAPIGG Aminopeptidase VDPKTLIAQRFKVWIDKGKFIYGVGASVPPHIQKPEDRKKAPDWDQ IFIDIGAESKEEAEDMGVKIGTVITWDGRLERLGKHRFVSIAFDDR IAVYTILEVAKQLKDAKADVYFVATVQEEVGLRGARTSAFGIEPDY GFAIDVTIAADIPGTPEHKQVTHLGKGTAIKIMDRSVICHPTIVRW LEELAKKHEIPYQLEILLGGGTDAGAIHLTKAGVPTGALSVPARYI HSNTEVVDERDVDATVELMTKALENIHELKI T. aquaticus 60 MDAFTENLNKLAELAIRVGLNLEEGQEIVATAPIEAVDFVRLLAEK Aminopeptidase T AYENGASLFTVLYGDNLIARKRLALVPEAHLDRAPAWLYEGMAKAF HEGAARLAVSGNDPKALEGLPPERVGRAQQAQSRAYRPTLSAITEF VTNWTIVPFAHPGWAKAVFPGLPEEEAVQRLWQAIFQATRVDQEDP VAAWEAHNRVLHAKVAFLNEKRFHALHFQGPGTDLTVGLAEGHLWQ GGATPTKKGRLCNPNLPTEEVFTAPHRERVEGVVRASRPLALSGQL VEGLWARFEGGVAVEVGAEKGEEVLKKLLDTDEGARRLGEVALVPA DNPIAKTGLVFFDTLFDENAASHIAFGQAYAENLEGRPSGEEFRRR GGNESMVHVDWMIGSEEVDVDGLLEDGTRVPLMRRGRWVI Bacillus 61 MAKLDETLTMLKALTDAKGVPGNEREARDVMKTYIAPYADEVTTDG stearothermophilus LGSLIAKKEGKSGGPKVMIAGHLDEVGFMVTQIDDKGFIRFQTLGG Peptidase M28 WWSQVMLAQRVTIVTKKGDITGVIGSKPPHILPSEARKKPVEIKDM FIDIGATSREEAMEWGVRPGDMIVPYFEFTVLNNEKMLLAKAWDNR IGCAVAIDVLKQLKGVDHPNTVYGVGTVQEEVGLRGARTAAQFIQP DIAFAVDVGIAGDTPGVSEKEAMGKLGAGPHIVLYDATMVSHRGLR EFVIEVAEELNIPHHFDAMPGVGTDAGAIHLTGIGVPSLTIAIPTR YIHSHAAILHRDDYENTVKLLVEVIKRLDADKVKQLTFDE Vibrio cholera 62 MEDKVWISMGADAVGSLNPALSESLLPHSFASGSQVWIGEVAIDEL Aminopeptidase AELSHTMHEQHNRCGGYMVHTSAQGAMAALMMPESIANFTIPAPSQ QDLVNAWLPQVSADQITNTIRALSSFNNRFYTTTSGAQASDWLANE WRSLISSLPGSRIEQIKHSGYNQKSVVLTIQGSEKPDEWVIVGGHL DSTLGSHTNEQSIAPGADDDASGIASLSEIIRVLRDNNFRPKRSVA LMAYAAEEVGLRGSQDLANQYKAQGKKVVSVLQLDMTNYRGSAEDI VFITDYTDSNLTQFLTTLIDEYLPELTYGYDRCGYACSDHASWHKA GFSAAMPFESKFKDYNPKIHTSQDTLANSDPIGNHAVKFTKLGLAY VIEMANAGSSQVPDDSVLQDGTAKINLSGARGTQKRFTFELSQSKP LTIQTYGGSGDVDLYVKYGSAPSKSNWDCRPYQNGNRETCSFNNAQ PGIYHVMLDGYTNYNDVALKASTQ Photobacterium 63 MEDKVWISIGSDASQTVKSVMQSNARSLLPESLASNGPVWVGQVDY halotolerans SQLAELSHHMHEDHQRCGGYMVHSSPESAIAASNMPQSLVAFSIPE Aminopeptidase ISQQDTVNAWLPQVNSQAITGTITSLTSFINRFYTTTSGAQASDWL ANEWRSLSASLPNASVRQVSHFGYNQKSVVLTITGSEKPDEWIVLG GHLDSTIGSHTNEQSVAPGADDDASGIASVTEIIRVLSENNFQPKR SIAFMAYAAEEVGLRGSQDLANQYKAEGKQVISALQLDMTNYKGSV EDIVFITDYTDSNLTTFLSQLVDEYLPSLTYGFDTCGYACSDHASW HKAGFSAAMPFEAKFNDYNPMIHTPNDTLQNSDPTASHAVKFTKLG LAYAIEMASTTGGTPPPTGNVLKDGVPVNGLSGATGSQVHYSFELP AQKNLQISTAGGSGDVDLYVSFGSEATKQNWDCRPYRNGNNEVCTF AGATPGTYSIMLDGYRQFSGVTLKASTQ Yersinia pestis 64 MTQQPQAKYRHDYRAPDYTITDIDLDFALDAQKTTVTAVSKVKRQG AminopeptidaseN TDVTPLILNGEDLTLISVSVDGQAWPHYRQQDNTLVIEQLPADFTL TIVNDIHPATNSALEGLYLSGEALCTQCEAEGFRHITYYLDRPDVL ARFTTRIVADKSRYPYLLSNGNRVGQGELDDGRHWVKWEDPFPKPS YLFALVAGDFDVLQDKFITRSGREVALEIFVDRGNLDRADWAMTSL KNSMKWDETRFGLEYDLDIYMIVAVDFFNMGAMENKGLNVFNSKYV LAKAETATDKDYLNIEAVIGHEYFHNWTGNRVTCRDWFQLSLKEGL TVFRDQEFSSDLGSRSVNRIENVRVMRAAQFAEDASPMAHAIRPDK VIEMNNFYTLTVYEKGSEVIRMMHTLLGEQQFQAGMRLYFERHDGS AATCDDFVQAMEDVSNVDLSLFRRWYSQSGTPLLTVHDDYDVEKQQ YHLFVSQKTLPTADQPEKLPLHIPLDIELYDSKGNVIPLQHNGLPV HHVLNVTEAEQTFTFDNVAQKPIPSLLREFSAPVKLDYPYSDQQLT FLMQHARNEFSRWDAAQSLLATYIKLNVAKYQQQQPLSLPAHVADA FRAILLDEHLDPALAAQILTLPSENEMAELFTTIDPQAISTVHEAI TRCLAQELSDELLAVYVANMTPVYRIEHGDIAKRALRNTCLNYLAF GDEEFANKLVSLQYHQADNMTDSLAALAAAVAAQLPCRDELLAAFD VRWNHDGLVMDKWFALQATSPAANVLVQVRTLLKHPAFSLSNPNRT RSLIGSFASGNPAAFHAADGSGYQFLVEILSDLNTRNPQVAARLIE PLIRLKRYDAGRQALMRKALEQLKTLDNLSGDLYEKITKALAA Vibrio anguillarum 65 MEEKVWISIGGDATQTALRSGAQSLLPENLINQTSVWVGQVPVSEL Aminopeptidase ATLSHEMHENHQRCGGYMVHPSAQSAMSVSAMPLNLNAFSAPEITQ QTTVNAWLPSVSAQQITSTITTLTQFKNRFYTTSTGAQASNWIADH WRSLSASLPASKVEQITHSGYNQKSVMLTITGSEKPDEWVVIGGHL DSTLGSRTNESSIAPGADDDASGIAGVTEIIRLLSEQNFRPKRSIA FMAYAAEEVGLRGSQDLANRFKAEGKKVMSVMQLDMTNYQGSREDI VFITDYTDSNFTQYLTQLLDEYLPSLTYGFDTCGYACSDHASWHAV GYPAAMPFESKFNDYNPNIHSPQDTLQNSDPTGFHAVKFTKLGLAY VVEMGNASTPPTPSNQLKNGVPVNGLSASRNSKTWYQFELQEAGNL SIVLSGGSGDADLYVKYQTDADLQQYDCRPYRSGNNETCQFSNAQP GRYSILLHGYNNYSNASLVANAQ Salinivibrio spYCSC6 66 MEDKKVWISIGADAQQTALSSGAQPLLAQSVAHNGQAWIGEVSESE Aminopeptidase LAALSHEMHENHHRCGGYIVHSSAQSAMAASNMPLSRASFIAPAIS QQALVTPWISQIDSALIVNTIDRLTDFPNRFYTTTSGAQASDWIKQ RWQSLSAGLAGASVTQISHSGYNQASVMLTIEGSESPDEWVVVGGH LDSTIGSRTNEQSIAPGADDDASGIAAVTEVIRVLAQNNFQPKRSI AFVAYAAEEVGLRGSQDVANQFKQAGKDVRGVLQLDMTNYQGSAED IVFITDYTDNQLTQYLTQLLDEYLPTLNYGFDTCGYACSDHASWHQ VGYPAAMPFEAKFNDYNPNIHTPQDTLANSDSEGAHAAKFTKLGLA YTVELANADSSPNPGNELKLGEPINGLSGARGNEKYFNYRLDQSGE LVIRTYGGSGDVDLYVKANGDVSTGNWDCRPYRSGNDEVCRFDNAT PGNYAVMLRGYRTYDNVSLIVE Vibrio proteolyticus 67 MPPITQQATVTAWLPQVDASQITGTISSLESFTNRFYTTTSGAQAS Aminopeptidase I DWIASEWQALSASLPNASVKQVSHSGYNQKSVVMTITGSEAPDEWI VIGGHLDSTIGSHTNEQSVAPGADDDASGIAAVTEVIRVLSENNFQ PKRSIAFMAYAAEEVGLRGSQDLANQYKSEGKNVVSALQLDMTNYK GSAQDVVFITDYTDSNFTQYLTQLMDEYLPSLTYGFDTCGYACSDH ASWHNAGYPAAMPFESKFNDYNPRIHTTQDTLANSDPTGSHAKKFT QLGLAYAIEMGSATGDTPTPGNQLE Vibrio proteolyticus 68 MPPITQQATVTAWLPQVDASQITGTISSLESFTNRFYTTTSGAQAS Aminopeptidase I DWIASEWQFLSASLPNASVKQVSHSGYNQKSVVMTITGSEAPDEWI (A55F) VIGGHLDSTIGSHTNEQSVAPGADDDASGIAAVTEVIRVLSENNFQ PKRSIAFMAYAAEEVGLRGSQDLANQYKSEGKNVVSALQLDMTNYK GSAQDVVFITDYTDSNFTQYLTQLMDEYLPSLTYGFDTCGYACSDH ASWHNAGYPAAMPFESKFNDYNPRIHTTQDTLANSDPTGSHAKKFT QLGLAYAIEMGSATGDTPTPGNQLE P. furiosus 69 MVDWELMKKIIESPGVSGYEHLGIRDLVVDILKDVADEVKIDKLGN Aminopeptidase I VIAHFKGSAPKVMVAAHMDKIGLMVNHIDKDGYLRVVPIGGVLPET LIAQKIRFFTEKGERYGVVGVLPPHLRREAKDQGGKIDWDSIIVDV GASSREEAEEMGFRIGTIGEFAPNFTRLSEHRFATPYLDDRICLYA MIEAARQLGEHEADIYIVASVQEEIGLRGARVASFAIDPEVGIAMD VTFAKQPNDKGKIVPELGKGPVMDVGPNINPKLRQFADEVAKKYEI PLQVEPSPRPTGTDANVMQINREGVATAVLSIPIRYMHSQVELADA RDVDNTIKLAKALLEELKPMDFTPLE *Cleavage efficiency (from most to least): arginine > lysine > hydrophobic residues (including alanine, leucine, methionine, and phenylalanine) > proline (see, e.g., Matthews Biochemisty 47, 2008, 5303-5311). **Cleavage efficiency (from most to least): leucine > alanine > arginine > phenylalanine > proline; does not cleave after glutamate and aspartate.
[0118] For the purposes of comparing two or more amino acid sequences, the percentage of "sequence identity" between a first amino acid sequence and a second amino acid sequence (also referred to herein as "amino acid identity") may be calculated by dividing [the number of amino acid residues in the first amino acid sequence that are identical to the amino acid residues at the corresponding positions in the second amino acid sequence] by [the total number of amino acid residues in the first amino acid sequence] and multiplying by [100], in which each deletion, insertion, substitution or addition of an amino acid residue in the second amino acid sequence compared to the first amino acid sequence is considered as a difference at a single amino acid residue (position). Alternatively, the degree of sequence identity between two amino acid sequences may be calculated using a known computer algorithm (e.g., by the local homology algorithm of Smith and Waterman (1970) Adv. Appl. Math. 2:482c, by the homology alignment algorithm of Needleman and Wunsch, J. Mol. Biol. (1970) 48:443, by the search for similarity method of Pearson and Lipman. Proc. Natl. Acad. Sci. USA (1998) 85:2444, or by computerized implementations of algorithms available as Blast, Clustal Omega, or other sequence alignment algorithms) and, for example, using standard settings. Usually, for the purpose of determining the percentage of "sequence identity" between two amino acid sequences in accordance with the calculation method outlined hereinabove, the amino acid sequence with the greatest number of amino acid residues will be taken as the "first" amino acid sequence, and the other amino acid sequence will be taken as the "second" amino acid sequence.
[0119] Additionally, or alternatively, two or more sequences may be assessed for the identity between the sequences. The terms "identical" or percent "identity" in the context of two or more nucleic acids or amino acid sequences, refer to two or more sequences or subsequences that are the same. Two sequences are "substantially identical" if two sequences have a specified percentage of amino acid residues or nucleotides that are the same (e.g., at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, 99.5%, 99.6%, 99.7%, 99.8%, or 99.9% identical) over a specified region or over the entire sequence, when compared and aligned for maximum correspondence over a comparison window, or designated region as measured using one of the above sequence comparison algorithms or by manual alignment and visual inspection. Optionally, the identity exists over a region that is at least about 25, 50, 75, or 100 amino acids in length, or over a region that is 100 to 150, 150 to 200, 100 to 200, or 200 or more, amino acids in length.
[0120] Additionally, or alternatively, two or more sequences may be assessed for the alignment between the sequences. The terms "alignment" or percent "alignment" in the context of two or more nucleic acids or amino acid sequences, refer to two or more sequences or subsequences that are the same. Two sequences are "substantially aligned" if two sequences have a specified percentage of amino acid residues or nucleotides that are the same (e.g., at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, 99.5%, 99.6%, 99.7%, 99.8% or 99.9% identical) over a specified region or over the entire sequence, when compared and aligned for maximum correspondence over a comparison window, or designated region as measured using one of the above sequence comparison algorithms or by manual alignment and visual inspection. Optionally, the alignment exists over a region that is at least about 25, 50, 75, or 100 amino acids in length, or over a region that is 100 to 150, 150 to 200, 100 to 200, or 200 or more amino acids in length.
[0121] In addition to protein molecules, nucleic acid molecules possess a variety of advantageous properties for use as affinity reagents (e.g., amino acid recognition molecules) in accordance with the application.
[0122] Nucleic acid aptamers are nucleic acid molecules that have been engineered to bind desired targets with high affinity and selectivity. Accordingly, nucleic acid aptamers may be engineered to selectively bind a desired type of amino acid using selection and/or enrichment techniques known in the art. Thus, in some embodiments, an affinity reagent comprises a nucleic acid aptamer (e.g., a DNA aptamer, an RNA aptamer). As shown in FIG. 1C, in some embodiments, a labeled affinity reagent is a labeled aptamer 104 that selectively binds one type of terminal amino acid. For example, in some embodiments, labeled aptamer 104 selectively binds one type of amino acid (e.g., a single type of amino acid or a subset of types of amino acids) at a terminus of a polypeptide, as described herein. Although not shown, it should be appreciated that labeled aptamer 104 may be engineered to selectively bind one type of amino acid at any position of a polypeptide (e.g., at a terminal position or at terminal and internal positions of a polypeptide) in accordance with a method of the application.
[0123] In some embodiments, a labeled affinity reagent comprises a label having binding-induced luminescence. For example, in some embodiments, a labeled aptamer 106 comprises a donor label 112 and an acceptor label 114 and functions as illustrated in panels (I) and (II) of FIG. 1C. As depicted in panel (I), labeled aptamer 106 as a free molecule adopts a conformation in which donor label 112 and acceptor label 114 are separated by a distance that limits detectable FRET between the labels (e.g., about 10 nm or more). As depicted in panel (II), labeled aptamer 106 as a selectively bound molecule adopts a conformation in which donor label 112 and acceptor label 114 are within a distance that promotes detectable FRET between the labels (e.g., about 10 nm or less). In yet other embodiments, labeled aptamer 106 comprises a quenching moiety and functions analogously to a molecular beacon, wherein luminescence of labeled aptamer 106 is internally quenched as a free molecule and restored as a selectively bound molecule (see, e.g., Hamaguchi, et al. (2001) Analytical Biochemistry 294, 126-131). Without wishing to be bound by theory, it is thought that these and other types of mechanisms for binding-induced luminescence may advantageously reduce or eliminate background luminescence to increase overall sensitivity and accuracy of the methods described herein.
[0124] In addition to methods of identifying a terminal amino acid of a polypeptide, the application provides methods of sequencing polypeptides using labeled affinity reagents. In some embodiments, methods of sequencing may involve subjecting a polypeptide terminus to repeated cycles of terminal amino acid detection and terminal amino acid cleavage. For example, in some embodiments, the application provides a method of determining an amino acid sequence of a polypeptide comprising contacting a polypeptide with one or more labeled affinity reagents described herein and subjecting the polypeptide to Edman degradation.
[0125] Conventional Edman degradation involves repeated cycles of modifying and cleaving the terminal amino acid of a polypeptide, wherein each successively cleaved amino acid is identified to determine an amino acid sequence of the polypeptide. As an illustrative example of a conventional Edman degradation, the N-terminal amino acid of a polypeptide is modified using phenyl isothiocyanate (PITC) to form a PITC-derivatized N-terminal amino acid. The PITC-derivatized N-terminal amino acid is then cleaved using acidic conditions, basic conditions, and/or elevated temperatures. It has also been shown that the step of cleaving the PITC-derivatized N-terminal amino acid may be accomplished enzymatically using a modified cysteine protease from the protozoa Trypanosoma cruzi, which involves relatively milder cleavage conditions at a neutral or near-neutral pH. Non-limiting examples of useful enzymes are described in U.S. patent application Ser. No. 15/255,433, filed Sep. 2, 2016, titled "MOLECULES AND METHODS FOR ITERATIVE POLYPEPTIDE ANALYSIS AND PROCESSING."
[0126] An example of sequencing by Edman degradation using labeled affinity reagents in accordance with the application is depicted in FIG. 1D. In some embodiments, sequencing by Edman degradation comprises providing a polypeptide 122 that is immobilized to a surface 130 of a solid support (e.g., immobilized to a bottom or sidewall surface of a sample well) through a linker 124. In some embodiments, as described herein, polypeptide 122 is immobilized at one terminus (e.g., an amino-terminal amino acid or a carboxy-terminal amino acid) such that the other terminus is free for detecting and cleaving of a terminal amino acid. Accordingly, in some embodiments, the reagents used in Edman degradation methods described herein preferentially interact with terminal amino acids at the non-immobilized (e.g., free) terminus of polypeptide 122. In this way, polypeptide 122 remains immobilized over repeated cycles of detecting and cleaving. To this end, in some embodiments, linker 124 may be designed according to a desired set of conditions used for detecting and cleaving, e.g., to limit detachment of polypeptide 122 from surface 130 under chemical cleavage conditions. Suitable linker compositions and techniques for immobilizing a polypeptide to a surface are described in detail elsewhere herein.
[0127] In accordance with the application, in some embodiments, a method of sequencing by Edman degradation comprises a step (1) of contacting polypeptide 122 with one or more labeled affinity reagents that selectively bind one or more types of terminal amino acids. As shown, in some embodiments, a labeled affinity reagent 108 interacts with polypeptide 122 by selectively binding the terminal amino acid. In some embodiments, step (1) further comprises removing any of the one or more labeled affinity reagents that do not selectively bind the terminal amino acid (e.g., the free terminal amino acid) of polypeptide 122.
[0128] In some embodiments, the method further comprises identifying the terminal amino acid of polypeptide 122 by detecting labeled affinity reagent 108. In some embodiments, detecting comprises detecting a luminescence from labeled affinity reagent 108. As described herein, in some embodiments, the luminescence is uniquely associated with labeled affinity reagent 108, and the luminescence is thereby associated with the type of amino acid to which labeled affinity reagent 108 selectively binds. As such, in some embodiments, the type of amino acid is identified by determining one or more luminescence properties of labeled affinity reagent 108.
[0129] In some embodiments, a method of sequencing by Edman degradation comprises a step (2) of removing the terminal amino acid of polypeptide 122. In some embodiments, step (2) comprises removing labeled affinity reagent 108 (e.g., any of the one or more labeled affinity reagents that selectively bind the terminal amino acid) from polypeptide 122. In some embodiments, step (2) comprises modifying the terminal amino acid (e.g., the free terminal amino acid) of polypeptide 122 by contacting the terminal amino acid with an isothiocyanate (e.g., PITC) to form an isothiocyanate-modified terminal amino acid. In some embodiments, an isothiocyanate-modified terminal amino acid is more susceptible to removal by a cleaving reagent (e.g., a chemical or enzymatic cleaving reagent) than an unmodified terminal amino acid.
[0130] In some embodiments, step (2) comprises removing the terminal amino acid by contacting polypeptide 122 with a protease 140 that specifically binds and cleaves the isothiocyanate-modified terminal amino acid. In some embodiments, protease 140 comprises a modified cysteine protease. In some embodiments, protease 140 comprises a modified cysteine protease, such as a cysteine protease from Trypanosoma cruzi (see, e.g., Borgo, et al. (2015) Protein Science 24:571-579). In yet other embodiments, step (2) comprises removing the terminal amino acid by subjecting polypeptide 122 to chemical (e.g., acidic, basic) conditions sufficient to cleave the isothiocyanate-modified terminal amino acid.
[0131] In some embodiments, a method of sequencing by Edman degradation comprises a step (3) of washing polypeptide 122 following terminal amino acid cleavage. In some embodiments, washing comprises removing protease 140. In some embodiments, washing comprises restoring polypeptide 122 to neutral pH conditions (e.g., following chemical cleavage by acidic or basic conditions). In some embodiments, a method of sequencing by Edman degradation comprises repeating steps (1) through (3) for a plurality of cycles.
[0132] An example method of sequencing by Edman degradation is shown in FIG. 1E. In some embodiments, a sample containing a complex mixture of polypeptides (e.g., a mixture of proteins) can be degraded using common enzymes into short polypeptide fragments of approximately 6 to 40 amino acids. In some embodiments, sequencing of this polypeptide library in accordance with methods of the application would reveal the identity and abundance of each of the polypeptides present in the original complex mixture. As described herein and in the literature, most polypeptides in the size range of 6 to 40 amino acids can be uniquely identified by determining the number and location of just four amino acids within a polypeptide chain.
[0133] Accordingly, in some embodiments, a method of sequencing by Edman degradation may be performed using a set of labeled aptamers 150 comprising four DNA aptamer types, each type recognizing a different N-terminal amino acid. Each aptamer type may be labeled with a different luminescent label, such that the different aptamer types can be distinguished based on one or more luminescence properties. For illustrative purposes, the example set of labeled aptamers 150 includes: a cysteine-specific aptamer labeled with a first luminescent label ("dye 1"); a lysine-specific aptamer labeled with a second luminescent label ("dye 2"); a tryptophan-specific aptamer labeled with a third luminescent label ("dye 3"); and a glutamate-specific aptamer labeled with a fourth luminescent label ("dye 4").
[0134] In some embodiments, a method of sequencing by Edman degradation in accordance with the application proceeds according to a process 152 shown in FIG. 1E. In some embodiments, prior to step (1), single polypeptide molecules from a polypeptide library are immobilized to a surface of a solid support, e.g., at a bottom or sidewall surface of a sample well of an array of sample wells. In some embodiments, as described elsewhere herein, moieties that enable surface immobilization (e.g., biotin) or improve solubility (e.g., oligonucleotides) may be chemically or enzymatically attached to the C-terminus of the polypeptides. To determine the sequence of each polypeptide, in some embodiments, immobilized polypeptides are subjected to repeated cycles of N-terminal amino acid detection and N-terminal amino acid cleavage, as illustrated by process 152. In some embodiments, process 152 comprises reagent addition and wash steps which are performed by injection into a flowcell above the detection surface using an automated fluidic system. In some embodiments, steps (1) through (4) illustrate one cycle of detection and cleavage using labeled aptamers 150.
[0135] In some embodiments, a method of sequencing by Edman degradation according to process 152 comprises a step (1) of flowing in a mixture of four orthogonally labeled DNA aptamers and incubating to allow the aptamers to bind to any immobilized polypeptides (e.g., polypeptides immobilized within a sample well of an array) that contain one of the four correct amino acids at the N-terminus. In some embodiments, the method further comprises washing the immobilized polypeptides to remove unbound aptamers. In some embodiments, the method further comprises imaging the immobilized polypeptides ("Imaging step 1"). In some embodiments, the acquired images contain enough information to determine the location of aptamer-bound polypeptides (e.g., location within an array of sample wells) and which of the four aptamers is bound at each location. In some embodiments, the method further comprises washing the immobilized polypeptides using an appropriate buffer to remove the aptamers from the immobilized polypeptides.
[0136] In some embodiments, a method of sequencing according to process 152 comprises a step (2) of flowing in a solution containing a reactive molecule (e.g., PITC, as shown) that specifically modifies the N-terminal amine group. An isothiocyanate molecule such as PITC, in some embodiments, modifies the N-terminal amino acid into a substrate for cleavage by a modified protease such as the cysteine protease cruzain from Trypanosoma Cruzi.
[0137] In some embodiments, a method of sequencing according to process 152 comprises a step (3) of washing the immobilized polypeptides before flowing in a suitable modified protease that recognizes and cleaves the modified N-terminal amino acid from the immobilized polypeptide. In some embodiments, the method comprises a step (4) of washing the immobilized polypeptides after enzymatic cleavage. In some embodiments, steps (1) through (4) depict one cycle of Edman degradation. Accordingly, step (1') as shown is the start of the next reaction cycle which proceeds as steps (1') through (4') performed as described above for steps (1) through (4). In some embodiments, steps (1) through (4) are repeated for approximately 20-40 cycles.
[0138] In some embodiments, a labeled isothiocyanate (e.g., a dye-labeled PITC) may be used to monitor sample loading. For example, in some embodiments, prior to subjecting a polypeptide sample to a method of sequencing as shown in process 152, the polypeptide sample is pre-conjugated with a luminescent label at a terminal end by modification of the terminal end using a dye-labeled PITC. In this way, loading of the polypeptide sample into an array of sample wells may be monitored by detecting luminescence from the labels prior to initiating process 152. In some embodiments, the luminescence is used to determine single occupancy of sample wells in the array (e.g., a fraction of sample wells containing a single polypeptide molecule), which may advantageously increase the amount of information reliably obtained for a given sample. Once a desired sample loading status is determined by luminescence, process 152 may be initiated by chemical or enzymatic cleavage, as described, before proceeding with step (1).
[0139] In some embodiments, a labeled isothiocyanate (e.g., a dye-labeled PITC) may be used to monitor reaction progress for a polypeptide sample in an array. For example, in some embodiments, step (2) comprises flowing in a solution containing a dye-labeled PITC that specifically modifies and labels N-terminal amine groups of polypeptides in the sample. In some embodiments, luminescence from the labels may be detected during or after step (2) to evaluate N-terminal PITC modification of polypeptides in the sample. Accordingly, in some embodiments, luminescence is used to determine whether or when to proceed from step (2) to step (3). In some embodiments, luminescence from the labels may be detected during or after step (3) to evaluate N-terminal amino acid cleavage of polypeptides in the sample--e.g., to determine whether or when to proceed from step (3) to step (4).
[0140] A method of sequencing according to process 152 utilizes separate reagents for detecting and cleaving a terminal amino acid of a polypeptide. Nonetheless, in some aspects, the application provides a method of sequencing in which a single reagent comprising a peptidase may be used for detecting and cleaving a terminal amino acid of a polypeptide. FIG. 2 shows an example of polypeptide sequencing using a set of labeled exopeptidases 200, wherein each labeled exopeptidase selectively binds and cleaves a different type of terminal amino acid.
[0141] As generically illustrated in the example of FIG. 2, labeled exopeptidases 200 include a lysine-specific exopeptidase comprising a first luminescent label, a glycine-specific exopeptidase comprising a second luminescent label, an aspartate-specific exopeptidase comprising a third luminescent label, and a leucine-specific exopeptidase comprising a fourth luminescent label. In accordance with certain embodiments described herein, each of labeled exopeptidases 200 selectively binds and cleaves its respective amino acid only when that amino acid is at an amino- or carboxy-terminus of a polypeptide. Accordingly, as sequencing by this approach proceeds from one terminus of a peptide toward the other, labeled exopeptidases 200 are engineered or selected such that all reagents of the set will possess either aminopeptidase or carboxypeptidase activity.
[0142] As further shown in FIG. 2, process 202 schematically illustrates a real-time sequencing reaction using labeled exopeptidases 200. Panels (I) through (IX) illustrate a progression of events involving iterative detection and cleavage at a terminal end of a polypeptide in relation to a signal output shown below, and corresponding to, the event depicted in each panel. For illustrative purposes, a polypeptide is shown having an arbitrarily selected amino acid sequence of "KLDG . . . " (proceeding from one terminus toward the other).
[0143] Panel (I) depicts the start of a sequencing reaction, wherein a polypeptide is immobilized to a surface of a solid support, such as a bottom or sidewall surface of a sample well. In some embodiments, sequencing methods in accordance with the application comprise single molecule sequencing in real-time. In some embodiments, a plurality of single molecule sequencing reactions are performed simultaneously in an array of sample wells. In such embodiments, polypeptide immobilization prevents diffusion of a polypeptide out of a sample well by anchoring the polypeptide within the sample well for single molecule analysis.
[0144] Panel (II) depicts a detection event, wherein the lysine-specific exopeptidase from the set of labeled affinity reagents 200 selectively binds the terminal lysine residue of the polypeptide. As shown in the signal trace below panels (I) and (II), signal output reports on this binding event by displaying an increase in signal intensity, which may be used to identify the luminescent label of the lysine-specific exopeptidase to thereby identify the terminal amino acid. Panel (III) illustrates that, after selectively binding a terminal amino acid, a labeled peptidase cleaves the terminal amino acid. As a result, these components are free to diffuse away from an observation region for luminescence detection, which is reported in the signal output by a drop in signal intensity, as shown in the trace below panel (III). Panels (IV) through (IX) proceed analogously to the process as described for panels (I) through (III). That is, a labeled exopeptidase binds and cleaves a corresponding terminal amino acid to produce a corresponding increase and decrease, respectively, in signal output.
[0145] In some aspects, the application provides methods of polypeptide sequencing in real-time by evaluating binding interactions of terminal amino acids with labeled amino acid recognition molecules (e.g., labeled affinity reagents) and a labeled cleaving reagent (e.g., a labeled non-specific exopeptidase). FIG. 3A shows an example of a method of sequencing in which discrete binding events give rise to signal pulses of a signal output 300. The inset panel of FIG. 3A illustrates a general scheme of real-time sequencing by this approach. As shown, a labeled affinity reagent 310 selectively associates with (e.g., binds to) and dissociates from a terminal amino acid (shown here as lysine), which gives rise to a series of pulses in signal output 300 which may be used to identify the terminal amino acid. In some embodiments, the series of pulses provide a pulsing pattern (e.g., a characteristic pattern) which may be diagnostic of the identity of the corresponding terminal amino acid.
[0146] Without wishing to be bound by theory, labeled affinity reagent 310 selectively binds according to a binding affinity (K.sub.D) defined by an association rate, or an "on" rate, of binding (k.sub.on) and a dissociation rate, or an "off" rate, of binding (k.sub.off). The rate constants k.sub.off and k.sub.on are the critical determinants of pulse duration (e.g., the time corresponding to a detectable binding event) and interpulse duration (e.g., the time between detectable binding events), respectively. In some embodiments, these rates can be engineered to achieve pulse durations and pulse rates (e.g., the frequency of signal pulses) that give the best sequencing accuracy.
[0147] As shown in the inset panel, a sequencing reaction mixture further comprises a labeled non-specific exopeptidase 320 comprising a luminescent label that is different than that of labeled affinity reagent 310. In some embodiments, labeled non-specific exopeptidase 320 is present in the mixture at a concentration that is less than that of labeled affinity reagent 310. In some embodiments, labeled non-specific exopeptidase 320 displays broad specificity such that it cleaves most or all types of terminal amino acids. Accordingly, a dynamic sequencing approach can involve monitoring affinity reagent binding at a terminus of a polypeptide over the course of a degradation reaction catalyzed by exopeptidase cleavage activity.
[0148] As illustrated by the progress of signal output 300, in some embodiments, terminal amino acid cleavage by labeled non-specific exopeptidase 320 gives rise to a signal pulse, and these events occur with lower frequency than the binding pulses of a labeled affinity reagent 310. In this way, amino acids of a polypeptide may be counted and/or identified in a real-time sequencing process. As further illustrated in signal output 300, in some embodiments, a plurality of labeled affinity reagents may be used, each with a diagnostic pulsing pattern (e.g., characteristic pattern) which may be used to identify a corresponding terminal amino acid. For example, in some embodiments, different characteristic patterns (as illustrated by each of lysine, phenylalanine, and glutamine in signal output 300) correspond to the association of more than one labeled affinity reagent with different types of terminal amino acids. As described herein, it should be appreciated that a single affinity reagent that associates with more than one type of amino acid may be used in accordance with the application. Accordingly, in some embodiments, different characteristic patterns correspond to the association of one labeled affinity reagent with different types of terminal amino acids.
[0149] As described herein, signal pulse information may be used to identify an amino acid based on a characteristic pattern in a series of signal pulses. In some embodiments, a characteristic pattern comprises a plurality of signal pulses, each signal pulse comprising a pulse duration. In some embodiments, the plurality of signal pulses may be characterized by a summary statistic (e.g., mean, median, time decay constant) of the distribution of pulse durations in a characteristic pattern. In some embodiments, the mean pulse duration of a characteristic pattern is between about 1 millisecond and about 10 seconds (e.g., between about 1 ms and about 1 s, between about 1 ms and about 100 ms, between about 1 ms and about 10 ms, between about 10 ms and about 10 s, between about 100 ms and about 10 s, between about 1 s and about 10 s, between about 10 ms and about 100 ms, or between about 100 ms and about 500 ms). In some embodiments, different characteristic patterns corresponding to different types of amino acids in a single polypeptide may be distinguished from one another based on a statistically significant difference in the summary statistic. For example, in some embodiments, one characteristic pattern may be distinguishable from another characteristic pattern based on a difference in mean pulse duration of at least 10 milliseconds (e.g., between about 10 ms and about 10 s, between about 10 ms and about 1 s, between about 10 ms and about 100 ms, between about 100 ms and about 10 s, between about 1 s and about 10 s, or between about 100 ms and about 1 s). It should be appreciated that, in some embodiments, smaller differences in mean pulse duration between different characteristic patterns may require a greater number of pulse durations within each characteristic pattern to distinguish one from another with statistical confidence.
[0150] As detailed above, a real-time sequencing process as illustrated by FIG. 3A can generally involve cycles of terminal amino acid recognition and terminal amino acid cleavage, where the relative occurrence of recognition and cleavage can be controlled by a concentration differential between a labeled affinity reagent 310 and a labeled non-specific exopeptidase 320. In some embodiments, the concentration differential can be optimized such that the number of signal pulses detected during recognition of an individual amino acid provides a desired confidence interval for identification. For example, if an initial sequencing reaction provides signal data with too few signal pulses between cleavage events to permit determination of characteristic patterns with a desired confidence interval, the sequencing reaction can be repeated using a decreased concentration of non-specific exopeptidase relative to affinity reagent.
[0151] In some embodiments, polypeptide sequencing in accordance with the application may be carried out by contacting a polypeptide with a sequencing reaction mixture comprising one or more amino acid recognition molecules (e.g., affinity reagents) and/or one or more cleaving reagents (e.g., exopeptidases). In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of between about 10 nM and about 10 .mu.M. In some embodiments, a sequencing reaction mixture comprises a cleaving reagent at a concentration of between about 500 nM and about 500 .mu.M.
[0152] In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of between about 100 nM and about 10 .mu.M, between about 250 nM and about 10 .mu.M, between about 100 nM and about 1 .mu.M, between about 250 nM and about 1 .mu.M, between about 250 nM and about 750 nM, or between about 500 nM and about 1 .mu.M. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of about 100 nM, about 250 nM, about 500 nM, about 750 nM, or about 1 .mu.M.
[0153] In some embodiments, a sequencing reaction mixture comprises a cleaving reagent at a concentration of between about 500 nM and about 250 .mu.M, between about 500 nM and about 100 .mu.M, between about 1 .mu.M and about 100 .mu.M, between about 500 nM and about 50 .mu.M, between about 1 .mu.M and about 100 .mu.M, between about 10 .mu.M and about 200 .mu.M, or between about 10 .mu.M and about 100 .mu.M. In some embodiments, a sequencing reaction mixture comprises a cleaving reagent at a concentration of about 1 .mu.M, about 5 .mu.M, about 10 .mu.M, about 30 .mu.M, about 50 .mu.M, about 70 .mu.M, or about 100 .mu.M.
[0154] In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of between about 10 nM and about 10 .mu.M, and a cleaving reagent at a concentration of between about 500 nM and about 500 .mu.M. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of between about 100 nM and about 1 .mu.M, and a cleaving reagent at a concentration of between about 1 .mu.M and about 100 .mu.M. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of between about 250 nM and about 1 .mu.M, and a cleaving reagent at a concentration of between about 10 .mu.M and about 100 .mu.M. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule at a concentration of about 500 nM, and a cleaving reagent at a concentration of between about 25 .mu.M and about 75 .mu.M.
[0155] In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule and a cleaving reagent in a ratio of about 500:1, about 400:1, about 300:1, about 200:1, about 100:1, about 75:1, about 50:1, about 25:1, about 10:1, about 5:1, about 2:1, or about 1:1. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule and a cleaving reagent in a ratio of between about 10:1 and about 200:1. In some embodiments, a sequencing reaction mixture comprises an amino acid recognition molecule and a cleaving reagent in a ratio of between about 50:1 and about 150:1.
[0156] While the example illustrated by FIG. 3A relates to a sequencing process using a labeled cleaving reagent, the sequencing process is not intended to be limited in this respect. As described elsewhere herein, the inventors have demonstrated single-molecule sequencing using an unlabeled cleaving reagent. In some embodiments, the approximate frequency with which a cleaving reagent removes successive terminal amino acids is known, e.g., based on a known activity and/or concentration of the enzyme being used. In some embodiments, terminal amino acid cleavage by the reagent is inferred, e.g., based on signal detected for amino acid recognition or a lack of signal detected. The inventors have recognized further techniques for controlling real-time sequencing reactions, which may be used in combination with, or alternatively to, the concentration differential approach as described.
[0157] An example of a temperature-dependent real-time sequencing process is shown in FIG. 3B. Panels (I) through (III) illustrate a sequencing reaction involving cycles of temperature-dependent terminal amino acid recognition and terminal amino acid cleavage. Each cycle of the sequencing reaction is carried out over two temperature ranges: a first temperature range ("T.sub.1") that is optimal for affinity reagent activity over exopeptidase activity (e.g., to promote terminal amino acid recognition), and a second temperature range ("T.sub.2") that is optimal for exopeptidase activity over affinity reagent activity (e.g., to promote terminal amino acid cleavage). The sequencing reaction progresses by alternating the reaction mixture temperature between the first temperature range T.sub.1 (to initiate amino acid recognition) and the second temperature range T.sub.2 (to initiate amino acid cleavage). Accordingly, progression of a temperature-dependent sequencing process is controllable by temperature, and alternating between different temperature ranges (e.g., between T.sub.1 and T.sub.2) may be carried through manual or automated processes. In some embodiments, affinity reagent activity (e.g., binding affinity (K.sub.D) for an amino acid) within the first temperature range T.sub.1 as compared to the second temperature range T.sub.2 is increased by at least 10-fold, at least 100-fold, at least 1,000-fold, at least 10,000-fold, at least 100,000-fold, or more. In some embodiments, exopeptidase activity (e.g., rate of substrate conversion to cleavage product) within the second temperature range T.sub.2 as compared to the first temperature range T.sub.1 is increased by at least 2-fold, 10-fold, at least 25-fold, at least 50-fold, at least 100-fold, at least 1,000-fold, or more.
[0158] In some embodiments, the first temperature range T.sub.1 is lower than the second temperature range T.sub.2. In some embodiments, the first temperature range T.sub.1 is between about 15.degree. C. and about 40.degree. C. (e.g., between about 25.degree. C. and about 35.degree. C., between about 15.degree. C. and about 30.degree. C., between about 20.degree. C. and about 30.degree. C.). In some embodiments, the second temperature range T.sub.2 is between about 40.degree. C. and about 100.degree. C. (e.g., between about 50.degree. C. and about 90.degree. C., between about 60.degree. C. and about 90.degree. C., between about 70.degree. C. and about 90.degree. C.). In some embodiments, the first temperature range T.sub.1 is between about 20.degree. C. and about 40.degree. C. (e.g., approximately 30.degree. C.), and the second temperature range T.sub.2 is between about 60.degree. C. and about 100.degree. C. (e.g., approximately 80.degree. C.).
[0159] In some embodiments, the first temperature range T.sub.1 is higher than the second temperature range T.sub.2. In some embodiments, the first temperature range T.sub.1 is between about 40.degree. C. and about 100.degree. C. (e.g., between about 50.degree. C. and about 90.degree. C., between about 60.degree. C. and about 90.degree. C., between about 70.degree. C. and about 90.degree. C.). In some embodiments, the second temperature range T.sub.2 is between about 15.degree. C. and about 40.degree. C. (e.g., between about 25.degree. C. and about 35.degree. C., between about 15.degree. C. and about 30.degree. C., between about 20.degree. C. and about 30.degree. C.). In some embodiments, the first temperature range T.sub.1 is between about 60.degree. C. and about 100.degree. C. (e.g., approximately 80.degree. C.), and the second temperature range T.sub.2 is between about 20.degree. C. and about 40.degree. C. (e.g., approximately 30.degree. C.).
[0160] Panel (I) depicts a sequencing reaction mixture at a temperature that is within a first temperature range T.sub.1 which is optimal for affinity reagent activity over exopeptidase activity. For illustrative purposes, a polypeptide of amino acid sequence "KFVAG . . . " is shown. When the reaction mixture temperature is within the first temperature range T.sub.1, labeled affinity reagents in the mixture are activated (e.g., renatured) to initiate amino acid recognition by associating with the polypeptide terminus. Also within the first temperature range T.sub.1, labeled exopeptidases in the mixture are inactivated (e.g., denatured) to prevent amino acid cleavage during recognition. In panel (I), a first affinity reagent is shown reversibly associating with lysine at the polypeptide terminus, while a labeled exopeptidase (e.g., Pfu aminopeptidase I (Pfu API)) is shown denatured. In some embodiments, amino acid recognition occurs for a predetermined duration of time before initiating cleavage of the amino acid. In some embodiments, amino acid recognition occurs for a duration of time required to reach a desired confidence interval for identification before initiating cleavage of the amino acid. Following amino acid recognition, the reaction proceeds by changing the temperature of the mixture to within a second temperature range T.sub.2.
[0161] Panel (II) depicts the sequencing reaction mixture at a temperature that is within a second temperature range T.sub.2 which is optimal for exopeptidase activity over affinity reagent activity. For illustrative purposes of this example, the second temperature range T.sub.2 is higher than the first temperature range T.sub.1, although it should be appreciated that reagent activity may be optimized for any desired temperature range. Accordingly, progression from panel (I) to panel (II) is carried out by raising the reaction mixture temperature using a suitable source of heat. When the reaction mixture reaches a temperature that is within the second temperature range T.sub.2, labeled exopeptidases in the mixture are activated (e.g., renatured) to initiate terminal amino acid cleavage by exopeptidase activity. Also within the second temperature range T.sub.2, labeled affinity reagents in the mixture are inactivated (e.g., denatured) to prevent amino acid recognition during cleavage. In panel (II), a labeled exopeptidase is shown cleaving the terminal lysine residue, while labeled affinity reagents are denatured. In some embodiments, amino acid cleavage occurs for a predetermined duration of time before initiating recognition of a successive amino acid at the polypeptide terminus. In some embodiments, amino acid cleavage occurs for a duration of time required to detect cleavage before initiating recognition of a successive amino acid. Following amino acid cleavage, the reaction proceeds by changing the temperature of the mixture to within the first temperature range T.sub.1.
[0162] Panel (III) depicts the beginning of the next cycle in the sequencing reaction, wherein the reaction mixture temperature has been reduced back to within the first temperature range T.sub.1. Accordingly, in this example, progression from panel (II) to panel (III) can be carried out by removing the reaction mixture from the source of heat or otherwise cooling the reaction mixture (e.g., actively or passively) to within the first temperature range T.sub.1. As shown, labeled affinity reagents are renatured, including a second affinity reagent that reversibly associates with phenylalanine at the polypeptide terminus, while the labeled exopeptidase is shown denatured. The sequencing reaction continues by further cycling between amino acid recognition and amino acid cleavage in a temperature-dependent fashion as illustrated by this example.
[0163] Accordingly, a dynamic sequencing approach can involve reaction cycling that is controlled at the level of protein activity or function of one or more proteins within a reaction mixture. It should be appreciated that the temperature-dependent polypeptide sequencing process depicted in FIG. 3B and described above may be illustrative of a general approach to polypeptide sequencing by controllable cycling of condition-dependent recognition and cleavage. For example, in some embodiments, the application provides a luminescence-dependent sequencing process using luminescence-activated reagents. In some embodiments, a luminescence-dependent sequencing process involves cycles of luminescence-dependent amino acid recognition and cleavage. Each cycle of the sequencing reaction may be carried out by exposing a sequencing reaction mixture to two different luminescent conditions: a first luminescent condition that is optimal for affinity reagent activity over exopeptidase activity (e.g., to promote amino acid recognition), and a second luminescent condition that is optimal for exopeptidase activity over affinity reagent activity (e.g., to promote amino acid cleavage). The sequencing reaction progresses by alternating between exposing the reaction mixture to the first luminescent condition (to initiate amino acid recognition) and exposing the reaction mixture to the second luminescent condition (to initiate amino acid cleavage). By way of example and not limitation, in some embodiments, the two different luminescent conditions comprise a first wavelength and a second wavelength.
[0164] In some aspects, the application provides methods of polypeptide sequencing in real-time by evaluating binding interactions of one or more labeled affinity reagents with terminal and internal amino acids and binding interactions of a labeled non-specific exopeptidase with terminal amino acids. FIG. 4 shows an example of a method of sequencing in which the method described and illustrated for the approach in FIGS. 3A-3B is modified by using a labeled affinity reagent 410 that selectively binds to and dissociates from one type of amino acid (shown here as lysine) at both terminal and internal positions (FIG. 4, inset panel). As described in the previous approach, the selective binding gives rise to a series of pulses in signal output 400. In this approach, however, the series of pulses occur at a rate that is determined by the number of the type of amino acid throughout the polypeptide. Accordingly, in some embodiments, the rate of pulsing corresponding to binding events would be diagnostic of the number of cognate amino acids currently present in the polypeptide.
[0165] As in the previous approach, a labeled non-specific peptidase 420 would be present at a relatively lower concentration than labeled affinity reagent 410, e.g., to give optimal time windows in between cleavage events (FIG. 4, inset panel). Additionally, in certain embodiments, uniquely identifiable luminescent label of labeled non-specific peptidase 420 would indicate when cleavage events have occurred. As the polypeptide undergoes iterative cleavage, the rate of pulsing corresponding to binding by labeled affinity reagent 410 would drop in a step-wise manner whenever a terminal amino acid is cleaved by labeled non-specific peptidase 420. This concept is illustrated by plot 402, which generally depicts pulse rate as a function of time, with cleavage events in time denoted by arrows. Thus, in some embodiments, amino acids may be identified--and polypeptides thereby sequenced--in this approach based on a pulsing pattern and/or on the rate of pulsing that occurs within a pattern detected between cleavage events.
[0166] In some embodiments, terminal polypeptide sequence information (e.g., determined as described herein) can be combined with polypeptide sequence information obtained from one or more other sources. For example, terminal polypeptide sequence information could be combined with internal polypeptide sequence information. In some embodiments, internal polypeptide sequence information can be obtained using one or more amino acid recognition molecules that associate with internal amino acids, as described herein. Internal or other polypeptide sequence information can be obtained before or during a polypeptide degradation process. In some embodiments, sequence information obtained from these methods can be combined with polypeptide sequence information using other techniques, e.g., sequence information obtained using one or more internal amino acid recognition molecules.
Shielded Recognition Molecules
[0167] In accordance with embodiments described herein, single-molecule polypeptide sequencing methods can be carried out by illuminating a surface-immobilized polypeptide with excitation light, and detecting luminescence produced by a label attached to an amino acid recognition molecule (e.g., a labeled affinity reagent). In some cases, radiative and/or non-radiative decay produced by the label can result in photodamage to the polypeptide. For example, FIG. 5A illustrates an example sequencing reaction in which a recognition molecule is shown associated with a polypeptide immobilized to a surface.
[0168] In the presence of excitation illumination, the label can produce fluorescence through radiative decay which results in a detectable association event. However, in some cases, the label produces non-radiative decay which can result in the formation of reactive oxygen species 500. The reactive oxygen species 500 can eventually damage the immobilized peptide, such that the reaction ends before obtaining complete sequence information for the polypeptide. This photodamage can occur, for example, at the exposed polypeptide terminus (top open arrow), at an internal position (middle open arrow), or at the surface linker attaching the polypeptide to the surface (bottom open arrow).
[0169] The inventors have found that photodamage can be mitigated and recognition times extended by incorporation of a shielding element into an amino acid recognition molecule. FIG. 5B illustrates an example sequencing reaction using a shielded recognition molecule that includes a shield 502. Shield 502 forms a covalent or non-covalent linkage group that provides increased distance between the label and polypeptide, such that damaging effects from reactive oxygen species 500 can be reduced due to free radical decay over the label-polypeptide separation distance. Shield 502 can also provide a steric barrier that shields the polypeptide from the label by absorbing damage from reactive oxygen species 500 and radiative and/or non-radiative decay.
[0170] Without wishing to be bound by theory, it is thought that a shield, positioned between a recognition component and a label component, can absorb, deflect, or otherwise block radiative and/or non-radiative decay emitted by the label component. In some embodiments, the shield prevents or limits the extent to which one or more labels (e.g., luminescent labels) interact with one or more amino acid recognition molecules. In some embodiments, the shield prevents or limits the extent to which one or more labels interact with one or more molecules associated with an amino acid recognition molecule (e.g., a polypeptide associated with the recognition molecule). Accordingly, in some embodiments, the term shield can generally refer to a protective or shielding effect that is provided by some portion of a linkage group formed between a recognition component and a label component.
[0171] In some embodiments, a shield is attached to one or more amino acid recognition molecules (e.g., a recognition component) and to one or more labels (e.g., a label component). In some embodiments, the recognition and label components are attached at non-adjacent sites on the shield. For example, one or more amino acid recognition molecules can be attached to a first side of the shield, and one or more labels can be attached to a second side of the shield, where the first and second sides of the shield are distant from each other. In some embodiments, the attachment sites are on approximately opposite sides of the shield.
[0172] The distance between the site at which a shield is attached to a recognition molecule and the site at which the shield is attached to a label can be a linear measurement through space or a non-linear measurement across the surface of the shield. The distance between the recognition molecule and label attachment sites on a shield can be measured by modeling the three-dimensional structure of the shield. In some embodiments, this distance can be at least 2 nm, at least 4 nm, at least 6 nm, at least 8 nm, at least 10 nm, at least 12 nm, at least 15 nm, at least 20 nm, at least 30 nm, at least 40 nm, or more. Alternatively, the relative positions of the recognition molecule and label on a shield can be described by treating the structure of the shield as a quadratic surface (e.g., ellipsoid, elliptic cylinder). In some embodiments, the recognition molecule and label attachment sites are separated by a distance that is at least one eighth of the distance around an ellipsoidal shape representing the shield. In some embodiments, the recognition molecule and label are separated by a distance that is at least one quarter of the distance around an ellipsoidal shape representing the shield. In some embodiments, the recognition molecule and label are separated by a distance that is at least one third of the distance around an ellipsoidal shape representing the shield. In some embodiments, the recognition molecule and label are separated by a distance that is one half of the distance around an ellipsoidal shape representing the shield.
[0173] The size of a shield should be such that a label is unable or unlikely to directly contact the polypeptide when the amino acid recognition molecule is associated with the polypeptide. The size of a shield should also be such that an attached label is detectable when the amino acid recognition molecule is associated with the polypeptide. For example, the size should be such that an attached luminescent label is within an illumination volume to be excited.
[0174] It should be appreciated that there are a variety of parameters by which a practitioner could evaluate shielding effects. Generally, the effects of a shielding element can be evaluated by conducting a comparative assessment between a composition having the shielding element and a composition lacking the shielding element. For example, a shielding element can increase recognition time of an amino acid recognition molecule. In some embodiments, recognition time refers to the length of time in which association events between the recognition molecule and a polypeptide are observable in a polypeptide sequencing reaction as described herein. In some embodiments, recognition time is increased by about 10-25%, 25-50%, 50-75%, 75-100%, or more than 100%, for example by about 2-fold, 3-fold, 4-fold, 5-fold, or more, relative to a polypeptide sequencing reaction performed under the same conditions, with the exception that the amino acid recognition molecule lacks the shielding element but is otherwise similar or identical. In some embodiments, a shielding element can increase sequencing accuracy and/or sequence read length (e.g., by at least 5%, at least 10%, at least 15%, at least 25% or more, relative to a sequencing reaction performed under comparative conditions as described above).
[0175] Accordingly, in some aspects, the application provides shielded recognition molecules comprising at least one amino acid recognition molecule, at least one detectable label, and a shielding element (e.g., a "shield") that forms a covalent or non-covalent linkage group between the recognition molecule and label. In some embodiments, a shielding element is at least 2 nm, at least 5 nm, at least 10 nm, at least 12 nm, at least 15 nm, at least 20 nm, or more, in length (e.g., in an aqueous solution). In some embodiments, a shielding element is between about 2 nm and about 100 nm in length (e.g., between about 2 nm and about 50 nm, between about 10 nm and about 50 nm, between about 20 nm and about 100 nm).
[0176] In some embodiments, a shield (e.g., shielding element) forms a covalent or non-covalent linkage group between one or more amino acid recognition molecules (e.g., a recognition component) and one or more labels (e.g., a label component). As used herein, in some embodiments, covalent and non-covalent linkages or linkage groups refer to the nature of the attachments of the recognition and label components to the shield.
[0177] In some embodiments, a covalent linkage, or a covalent linkage group, refers to a shield that is attached to each of the recognition and label components through a covalent bond or a series of contiguous covalent bonds. Covalent attachment one or both components can be achieved by covalent conjugation methods known in the art. For example, in some embodiments, click chemistry techniques (e.g., copper-catalyzed, strain-promoted, copper-free click chemistry, etc.) can be used to attach one or both components to the shield. Such methods generally involve conjugating one reactive moiety to another reactive moiety to form one or more covalent bonds between the reactive moieties. Accordingly, in some embodiments, a first reactive moiety of a shield can be contacted with a second reactive moiety of a recognition or label component to form a covalent attachment. Examples of reactive moieties include, without limitation, reactive amines, azides, alkynes, nitrones, alkenes (e.g., cycloalkenes), tetrazines, tetrazoles, and other reactive moieties suitable for click reactions and similar coupling techniques.
[0178] In some embodiments, a non-covalent linkage, or a non-covalent linkage group, refers to a shield that is attached to one or both of the recognition and label components through one or more non-covalent coupling means, including but not limited to receptor-ligand interactions and oligonucleotide strand hybridization. Examples of receptor-ligand interactions are provided herein and include, without limitation, protein-protein complexes, protein-ligand complexes, protein-aptamer complexes, and aptamer-nucleic acid complexes. Various configurations and strategies for oligonucleotide strand hybridization are described herein and are known in the art (see, e.g., U.S. Patent Publication No. 2019/0024168).
[0179] In some embodiments, shield 502 comprises a polymer, such as a biomolecule or a dendritic polymer. FIG. 5C depicts examples of polymer shields and configurations of shielded recognition molecules of the application. A first shielded construct 504 shows an example of a protein shield 530. In some embodiments, protein shield 530 forms a covalent linkage group between a recognition molecule and a label. For example, in some embodiments, protein shield 530 is attached to each of the recognition molecule and label through one or more covalent bonds, e.g., by covalent attachment through a side-chain of a natural or unnatural amino acid of protein shield 530. In some embodiments, protein shield 530 forms a non-covalent linkage group between a recognition molecule and a label. For example, in some embodiments, protein shield 530 is a monomeric or multimeric protein comprising one or more ligand-binding sites. In some embodiments, a non-covalent linkage group is formed through one or more ligand moieties bound to the one or more ligand-binding sites. Additional examples of non-covalent linkages formed by protein shields are described elsewhere herein.
[0180] A second shielded construct 506 shows an example of a double-stranded nucleic acid shield comprising a first oligonucleotide strand 532 hybridized with a second oligonucleotide strand 534. As shown, in some embodiments, the double-stranded nucleic acid shield can comprise a recognition molecule attached to first oligonucleotide strand 532, and a label attached to second oligonucleotide strand 534. In this way, the double-stranded nucleic acid shield forms a non-covalent linkage group between the recognition molecule and the label through oligonucleotide strand hybridization. In some embodiments, a recognition molecule and a label can be attached to the same oligonucleotide strand, which can provide a single-stranded nucleic acid shield or a double-stranded nucleic acid shield through hybridization with another oligonucleotide strand. In some embodiments, strand hybridization can provide increased rigidity within a linkage group to further enhance separation between the recognition molecule and the label.
[0181] Where shielding element 502 comprises a nucleic acid, the separation distance between a label and a recognition molecule can be measured by the distance between attachment sites on the nucleic acid (e.g., direct attachment or indirect attachment, such as through one or more additional shield polymers). In some embodiments, the distance between attachment sites on a nucleic acid can be measured by the number of nucleotides within the nucleic acid that occur between the label and the recognition molecule. It should be understood that the number of nucleotides can refer to either the number of nucleotide bases in a single-stranded nucleic acid or the number of nucleotide base pairs in a double-stranded nucleic acid.
[0182] Accordingly, in some embodiments, the attachment site of a recognition molecule and the attachment site of a label can be separated by between 5 and 200 nucleotides (e.g., between 5 and 150 nucleotides, between 5 and 100 nucleotides, between 5 and 50 nucleotides, between 10 and 100 nucleotides). It should be appreciated that any position in a nucleic acid can serve as an attachment site for a recognition molecule, a label, or one or more additional polymer shields. In some embodiments, an attachment site can be at or approximately at the 5' or 3' end, or at an internal position along a strand of the nucleic acid.
[0183] The non-limiting configuration of second shielded construct 506 illustrates an example of a shield that forms a non-covalent linkage through strand hybridization. A further example of non-covalent linkage is illustrated by a third shielded construct 508 comprising an oligonucleotide shield 536. In some embodiments, oligonucleotide shield 536 is a nucleic acid aptamer that binds a recognition molecule to form a non-covalent linkage. In some embodiments, the recognition molecule is a nucleic acid aptamer, and oligonucleotide shield 536 comprises an oligonucleotide strand that hybridizes with the aptamer to form a non-covalent linkage.
[0184] A fourth shielded construct 510 shows an example of a dendritic polymer shield 538. As used herein, in some embodiments, a dendritic polymer refers generally to a polyol or a dendrimer. Polyols and dendrimers have been described in the art, and may include branched dendritic structures optimized for a particular configuration. In some embodiments, dendritic polymer shield 538 comprises polyethylene glycol, tetraethylene glycol, poly(amidoamine), poly(propyleneimine), poly(propyleneamine), carbosilane, poly(L-lysine), or a combination of one or more thereof.
[0185] A dendrimer, or dendron, is a repetitively branched molecule that is typically symmetric around the core and that may adopt a spherical three-dimensional morphology. See, e.g., Astruc et al. (2010) Chem. Rev. 110:1857. Incorporation of such structures into a shield of the application can provide for a protective effect through the steric inhibition of contacts between a label and one or more biomolecules associated therewith (e.g., a recognition molecule and/or a polypeptide associated with the recognition molecule). Refinement of the chemical and physical properties of the dendrimer through variation in primary structure of the molecule, including potential functionalization of the dendrimer surface, allows the shielding effects to be adjusted as desired. Dendrimers may be synthesized by a variety of techniques using a wide range of materials and branching reactions, as is known in the art. Such synthetic variation allows the properties of the dendrimer to be customized as necessary. Examples of polyol and dendrimer compounds which can be used in accordance with shields of the application include, without limitation, compounds described in U.S. Patent Publication No. 20180346507.
[0186] FIG. 5D depicts further example configurations of shielded recognition molecules of the application. A protein-nucleic acid construct 512 shows an example of a shield comprising more than one polymer in the form of a protein and a double-stranded nucleic acid. In some embodiments, the protein portion of the shield is attached to the nucleic acid portion of the shield through a covalent linkage. In some embodiments, the attachment is through a non-covalent linkage. For example, in some embodiments, the protein portion of the shield is a monovalent or multivalent protein that forms at least one non-covalent linkage through a ligand moiety attached to a ligand-binding site of the monovalent or multivalent protein. In some embodiments, the protein portion of the shield comprises an avidin protein.
[0187] In some embodiments, a shielded recognition molecule of the application is an avidin-nucleic acid construct 514. In some embodiments, avidin-nucleic acid construct 514 includes a shield comprising an avidin protein 540 and a double-stranded nucleic acid. As described herein, avidin protein 540 may be used to form a non-covalent linkage between one or more amino acid recognition molecules and one or more labels, either directly or indirectly, such as through one or more additional shield polymers described herein.
[0188] Avidin proteins are biotin-binding proteins, generally having a biotin binding site at each of four subunits of the avidin protein. Avidin proteins include, for example, avidin, streptavidin, traptavidin, tamavidin, bradavidin, xenavidin, and homologs and variants thereof. In some cases, the monomeric, dimeric, or tetrameric form of the avidin protein can be used. In some embodiments, the avidin protein of an avidin protein complex is streptavidin in a tetrameric form (e.g., a homotetramer). In some embodiments, the biotin binding sites of an avidin protein provide attachment sites for one or more amino acid recognition molecules, one or more labels, and/or one or more additional shield polymers described herein.
[0189] An illustrative diagram of an avidin protein complex is shown in the inset panel of FIG. 5D. As shown in the inset panel, avidin protein 540 can include a binding site 542 at each of four subunits of the protein which can be bound to a biotin moiety (shown as white circles). The multivalency of avidin protein 540 can allow for various linkage configurations, which are generally shown for illustrative purposes. For example, in some embodiments, a biotin linkage moiety 544 can be used to provide a single point of attachment to avidin protein 540. In some embodiments, a bis-biotin linkage moiety 546 can be used to provide two points of attachment to avidin protein 540. As illustrated by avidin-nucleic acid construct 514, an avidin protein complex may be formed by two bis-biotin linkage moieties, which form a trans-configuration to provide an increased separation distance between a recognition molecule and a label.
[0190] Various further examples of avidin protein shield configurations are shown. A first avidin construct 516 shows an example of an avidin shield attached to a recognition molecule through a bis-biotin linkage moiety and to two labels through separate biotin linkage moieties. A second avidin construct 518 shows an example of an avidin shield attached to two recognition molecules through separate biotin linkage moieties and to a label through a bis-biotin linkage moiety. A third avidin construct 520 shows an example of an avidin shield attached to two recognition molecules through separate biotin linkage moieties and to a labeled nucleic acid through a biotin linkage moiety of each strand of the nucleic acid. A fourth avidin construct 522 shows an example of an avidin shield attached to a recognition molecule and to a labeled nucleic acid through separate bis-biotin linkage moieties. As shown, the label is further shielded from the recognition molecule by a dendritic polymer between the label and nucleic acid.
[0191] It should be appreciated that the example configurations of shielded recognition molecules shown in FIGS. 5A-5D are provided for illustrative purposes. The inventors have conceived of various other shield configurations using one or more different polymers that form a covalent or non-covalent linkage between recognition and label components of a shielded recognition molecule. By way of example, FIG. 5E illustrates the modularity of shield configuration in accordance with the application.
[0192] As shown at the top of FIG. 5E, a shielded recognition molecule generally comprises a recognition component 550, a shielding element 552, and a label component 554. For ease of illustration, recognition component 550 is depicted as one amino acid recognition molecule, and label component 554 is depicted as one label. It should be appreciated that shielded recognition molecules of the application can comprise shielding element 552 attached to one or more amino acid recognition molecules and one or more labels. Where recognition component 550 comprises more than one recognition molecule, each recognition molecule can be attached to shielding element 552 at one or more attachment sites on shielding element 552. Where label component 554 comprises more than one label, each label can be attached to shielding element 552 at one or more attachment sites on shielding element 552.
[0193] In some embodiments, shielding element 552 comprises a protein 560. In some embodiments, protein 560 is a monovalent or multivalent protein. In some embodiments, protein 560 is a monomeric or multimeric protein, such as a protein homodimer, protein heterodimer, protein oligomer, or other proteinaceous molecule. In some embodiments, shielding element 552 comprises a protein complex formed by a protein non-covalently bound to at least one other molecule. For example, in some embodiments, shielding element 552 comprises a protein-protein complex 562. In some embodiments, protein-protein complex 562 comprises one proteinaceous molecule specifically bound to another proteinaceous molecule. In some embodiments, protein-protein complex 562 comprises an antibody or antibody fragment (e.g., scFv) bound to an antigen. In some embodiments, protein-protein complex 562 comprises a receptor bound to a protein ligand. Additional examples of protein-protein complexes include, without limitation, trypsin-aprotinin, barnase-barstar, and colicin E9-Im9 immunity protein.
[0194] In some embodiments, shielding element 552 comprises a protein-ligand complex 564. In some embodiments, protein-ligand complex 564 comprises a monovalent protein and a non-proteinaceous ligand moiety. For example, in some embodiments, protein-ligand complex 564 comprises an enzyme bound to a small-molecule inhibitor moiety. In some embodiments, protein-ligand complex 564 comprises a receptor bound to a non-proteinaceous ligand moiety.
[0195] In some embodiments, shielding element 552 comprises a multivalent protein complex formed by a multivalent protein non-covalently bound to one or more ligand moieties. In some embodiments, shielding element 552 comprises an avidin protein complex formed by an avidin protein non-covalently bound to one or more biotin linkage moieties. Constructs 566, 568, 570, and 572 provide illustrative examples of avidin protein complexes, any one or more of which may be incorporated into shielding element 552.
[0196] In some embodiments, shielding element 552 comprises a two-way avidin complex 566 comprising an avidin protein bound to two bis-biotin linkage moieties. In some embodiments, shielding element 552 comprises a three-way avidin complex 568 comprising an avidin protein bound to two biotin linkage moieties and a bis-biotin linkage moiety. In some embodiments, shielding element 552 comprises a four-way avidin complex 570 comprising an avidin protein bound to four biotin linkage moieties.
[0197] In some embodiments, shielding element 552 comprises an avidin protein comprising one or two non-functional binding sites engineered into the avidin protein. For example, in some embodiments, shielding element 552 comprises a divalent avidin complex 572 comprising an avidin protein bound to a biotin linkage moiety at each of two subunits, where the avidin protein comprises a non-functional ligand-binding site 548 at each of two other subunits. As shown, in some embodiments, divalent avidin complex 572 comprises a trans-divalent avidin protein, although a cis-divalent avidin protein may be used depending on a desired implementation. In some embodiments, the avidin protein is a trivalent avidin protein. In some embodiments, the trivalent avidin protein comprises non-functional ligand-binding site 548 at one subunit and is bound to three biotin linkage moieties, or one biotin linkage moiety and one bis-biotin linkage moiety, at the other subunits.
[0198] In some embodiments, shielding element 552 comprises a dendritic polymer 574. In some embodiments, dendritic polymer 574 is a polyol or a dendrimer, as described elsewhere herein. In some embodiments, dendritic polymer 574 is a branched polyol or a branched dendrimer. In some embodiments, dendritic polymer 574 comprises a monosaccharide-TEG, a disaccharide, an N-acetyl monosaccharide, a TEMPO-TEG, a trolox-TEG, or a glycerol dendrimer. Examples of polyols useful in accordance with shielded recognition molecules of the application include polyether polyols and polyester polyols, e.g., polyethylene glycol, polypropylene glycol, and similar such polymers well known in the art. In some embodiments, dendritic polymer 574 comprises a compound of the following formula: --(CH.sub.2CH.sub.2O).sub.n--, where n is an integer from 1 to 500, inclusive. In some embodiments, dendritic polymer 574 comprises a compound of the following formula: --(CH.sub.2CH.sub.2O).sub.n--, wherein n is an integer from 1 to 100, inclusive.
[0199] In some embodiments, shielding element 552 comprises a nucleic acid. In some embodiments, the nucleic acid is single-stranded. In some embodiments, label component 554 is attached directly or indirectly to one end of the single-stranded nucleic acid (e.g., the 5' end or the 3' end) and recognition component 550 is attached directly or indirectly to the other end of the single-stranded nucleic acid (e.g., the 3' end or the 5' end). For example, the single-stranded nucleic acid can comprise a label attached to the 5' end of the nucleic acid and an amino acid recognition molecule attached to the 3' end of the nucleic acid.
[0200] In some embodiments, shielding element 552 comprises a double-stranded nucleic acid 576. As shown, in some embodiments, double-stranded nucleic acid 576 can form a non-covalent linkage between recognition component 550 and label component 554 through strand hybridization. However, in some embodiments, double-stranded nucleic acid 576 can form a covalent linkage between recognition component 550 and label component 554 through attachment to the same oligonucleotide strand. In some embodiments, label component 554 is attached directly or indirectly to one end of the double-stranded nucleic acid and recognition component 550 is attached directly or indirectly to the other end of the double-stranded nucleic acid. For example, the double-stranded nucleic acid can comprise a label attached to the 5' end of one strand and an amino acid recognition molecule attached to the 5' end of the other strand.
[0201] In some embodiments, shielding element 552 comprises a nucleic acid that forms one or more structural motifs which can be useful for increasing steric bulk of the shield. Examples of nucleic acid structural motifs include, without limitation, stem-loops, three-way junctions (e.g., formed by two or more stem-loop motifs), four-way junctions (e.g., Holliday junctions), and bulge loops.
[0202] In some embodiments, shielding element 552 comprises a nucleic acid that forms a stem-loop 578. A stem-loop, or hairpin loop, is an unpaired loop of nucleotides on an oligonucleotide strand that is formed when the oligonucleotide strand folds and forms base pairs with another section of the same strand. In some embodiments, the unpaired loop of stem-loop 578 comprises three to ten nucleotides. Accordingly, stem-loop 578 can be formed by two regions of an oligonucleotide strand having inverted complementary sequences that hybridize to form a stem, where the two regions are separated by the three to ten nucleotides that form the unpaired loop. In some embodiments, the stem of stem-loop 578 can be designed to have one or more G/C nucleotides, which can provide added stability with the addition hydrogen bonding interaction that forms compared to A/T nucleotides. In some embodiments, the stem of stem-loop 578 comprises G/C nucleotides immediately adjacent to an unpaired loop sequence. In some embodiments, the stem of stem-loop 578 comprises G/C nucleotides within the first 2, 3, 4, or 5 nucleotides adjacent to an unpaired loop sequence. In some embodiments, an unpaired loop of stem-loop 578 comprises one or more attachment sites. In some embodiments, an attachment site occurs at an abasic site in the unpaired loop. In some embodiments, an attachment site occurs at a base of the unpaired loop.
[0203] In some embodiments, stem-loop 578 is formed by a double-stranded nucleic acid. As described herein, in some embodiments, the double-stranded nucleic acid can form a non-covalent linkage group through strand hybridization of first and second oligonucleotide strands. However, in some embodiments, shielding element 552 comprises a single-stranded nucleic acid that forms a stem-loop motif, e.g., to provide a covalent linkage group. In some embodiments, shielding element 552 comprises a nucleic acid that forms two or more stem-loop motifs. For example, in some embodiments, the nucleic acid comprises two stem-loop motifs. In some embodiments, a stem of one stem-loop motif is adjacent to the stem of the other such that the motifs together form a three-way junction. In some embodiments, shielding element 552 comprises a nucleic acid that forms a four-way junction 578. In some embodiments, four-way junction 578 is formed through hybridization of two or more oligonucleotide strands (e.g., 2, 3, or 4 oligonucleotide strands).
[0204] In some embodiments, shielding element 552 comprises one or more polymers selected from 560, 562, 564, 566, 568, 570, 572, 574, 576, 578, and 580 of FIG. 5E. It should be appreciated that the linkage moieties and attachment sites shown on each of 560, 562, 564, 566, 568, 570, 572, 574, 576, 578, and 580 are shown for illustrative purposes and are not intended to depict a preferred linkage or attachment site configuration.
[0205] In some aspects, the application provides an amino acid recognition molecule of Formula (I):
A-(Y).sub.n-D (I),
wherein: A is an amino acid binding component comprising at least one amino acid recognition molecule; each instance of Y is a polymer that forms a covalent or non-covalent linkage group; n is an integer from 1 to 10, inclusive; and D is a label component comprising at least one detectable label. In some embodiments, the application provides a composition comprising a soluble amino acid recognition molecule of Formula (I).
[0206] In some embodiments, A comprises a plurality of amino acid recognition molecules. In some embodiments, each amino acid recognition molecule of the plurality is attached to a different attachment site on Y. In some embodiments, at least two amino acid recognition molecules of the plurality are attached to a single attachment site on Y. In some embodiments, the amino acid recognition molecule is a recognition protein or a nucleic acid aptamer, e.g., as described elsewhere herein.
[0207] In some embodiments, the detectable label is a luminescent label or a conductivity label. In some embodiments, the luminescent label comprises at least one fluorophore dye molecule. In some embodiments, D comprises 20 or fewer fluorophore dye molecules. In some embodiments, the ratio of the number of fluorophore dye molecules to the number of amino acid recognition molecules is between 1:1 and 20:1. In some embodiments, the luminescent label comprises at least one FRET pair comprising a donor label and an acceptor label. In some embodiments, the ratio of the donor label to the acceptor label is 1:1, 2:1, 3:1, 4:1, or 5:1. In some embodiments, the ratio of the acceptor label to the donor label is 1:1, 2:1, 3:1, 4:1, or 5:1.
[0208] In some embodiments, D is less than 200 .ANG. in diameter. In some embodiments, --(Y).sub.n-- is at least 2 nm in length. In some embodiments, --(Y).sub.n-- is at least 5 nm in length. In some embodiments, --(Y).sub.n-- is at least 10 nm in length. In some embodiments, each instance of Y is independently a biomolecule, a polyol, or a dendrimer. In some embodiments, the biomolecule is a nucleic acid, a polypeptide, or a polysaccharide.
[0209] In some embodiments, the amino acid recognition molecule is of one of the following formulae:
A-Y.sup.1--(Y).sub.m-D or A-(Y).sub.m--Y.sup.1-D,
wherein: Y.sup.1 is a nucleic acid or a polypeptide; and m is an integer from 0 to 10, inclusive.
[0210] In some embodiments, the nucleic acid comprises a first oligonucleotide strand. In some embodiments, the nucleic acid comprises a second oligonucleotide strand hybridized with the first oligonucleotide strand. In some embodiments, the nucleic acid forms a covalent linkage through the first oligonucleotide strand. In some embodiments, the nucleic acid forms a non-covalent linkage through the hybridized first and second oligonucleotide strands.
[0211] In some embodiments, the polypeptide is a monovalent or multivalent protein. In some embodiments, the monovalent or multivalent protein forms at least one non-covalent linkage through a ligand moiety attached to a ligand-binding site of the monovalent or multivalent protein. In some embodiments, A, Y, or D comprises the ligand moiety.
[0212] In some embodiments, the amino acid recognition molecule is of one of the following formulae:
A-(Y).sub.n--Y.sup.2-D or A-Y.sup.2--(Y).sub.n-D,
wherein: Y.sup.2 is a polyol or dendrimer; and m is an integer from 0 to 10, inclusive. In some embodiments, the polyol or dendrimer comprises polyethylene glycol, tetraethylene glycol, poly(amidoamine), poly(propyleneimine), poly(propyleneamine), carbosilane, poly(L-lysine), or a combination of one or more thereof.
[0213] In some aspects, the application provides an amino acid recognition molecule of Formula (II):
A-Y.sup.1-D (II),
wherein: A is an amino acid binding component comprising at least one amino acid recognition molecule; Y.sup.1 is a nucleic acid or a polypeptide; D is a label component comprising at least one detectable label. In some embodiments, when Y.sup.1 is a nucleic acid, the nucleic acid forms a covalent or non-covalent linkage group. In some embodiments, when Y.sup.1 is a polypeptide, the polypeptide forms a non-covalent linkage group characterized by a dissociation constant (K.sub.D) of less than 50.times.10.sup.-9M.
[0214] In some embodiments, Y.sup.1 is a nucleic acid comprising a first oligonucleotide strand. In some embodiments, the nucleic acid comprises a second oligonucleotide strand hybridized with the first oligonucleotide strand. In some embodiments, A is attached to the first oligonucleotide strand, and wherein D is attached to the second oligonucleotide strand. In some embodiments, A is attached to a first attachment site on the first oligonucleotide strand, and wherein D is attached to a second attachment site on the first oligonucleotide strand. In some embodiments, each oligonucleotide strand of the nucleic acid comprises fewer than 150, fewer than 100, or fewer than 50 nucleotides.
[0215] In some embodiments, Y.sup.1 is a monovalent or multivalent protein. In some embodiments, the monovalent or multivalent protein forms at least one non-covalent linkage through a ligand moiety attached to a ligand-binding site of the monovalent or multivalent protein. In some embodiments, at least one of A and D comprises the ligand moiety. In some embodiments, the polypeptide is an avidin protein (e.g., avidin, streptavidin, traptavidin, tamavidin, bradavidin, xenavidin, or a homolog or variant thereof). In some embodiments, the ligand moiety is a biotin moiety.
[0216] In some embodiments, the amino acid recognition molecule is of one of the following formulae:
A-Y.sup.1--(Y).sub.n-D or A-(Y).sub.n--Y.sup.1-D,
wherein: each instance of Y is a polymer that forms a covalent or non-covalent linkage group; and n is an integer from 1 to 10, inclusive. In some embodiments, each instance of Y is independently a biomolecule, a polyol, or a dendrimer.
[0217] In other aspects, the application provides an amino acid recognition molecule comprising: a nucleic acid; at least one amino acid recognition molecule attached to a first attachment site on the nucleic acid; and at least one detectable label attached to a second attachment site on the nucleic acid. In some embodiments, the nucleic acid forms a covalent or non-covalent linkage group between the at least one amino acid recognition molecule and the at least one detectable label.
[0218] In some embodiments, the nucleic acid is a double-stranded nucleic acid comprising a first oligonucleotide strand hybridized with a second oligonucleotide strand. In some embodiments, the first attachment site is on the first oligonucleotide strand, and wherein the second attachment site is on the second oligonucleotide strand. In some embodiments, the at least one amino acid recognition molecule is attached to the first attachment site through a protein that forms a covalent or non-covalent linkage group between the at least one amino acid recognition molecule and the nucleic acid. In some embodiments, the at least one detectable label is attached to the second attachment site through a protein that forms a covalent or non-covalent linkage group between the at least one detectable label and the nucleic acid. In some embodiments, the first and second attachment sites are separated by between 5 and 100 nucleotide bases or nucleotide base pairs on the nucleic acid.
[0219] In yet other aspects, the application provides an amino acid recognition molecule comprising: a multivalent protein comprising at least two ligand-binding sites; at least one amino acid recognition molecule attached to the protein through a first ligand moiety bound to a first ligand-binding site on the protein; and at least one detectable label attached to the protein through a second ligand moiety bound to a second ligand-binding site on the protein.
[0220] In some embodiments, the multivalent protein is an avidin protein comprising four ligand-binding sites. In some embodiments, the ligand-binding sites are biotin binding sites, and wherein the ligand moieties are biotin moieties. In some embodiments, at least one of the biotin moieties is a bis-biotin moiety, and wherein the bis-biotin moiety is bound to two biotin binding sites on the avidin protein. In some embodiments, the at least one amino acid recognition molecule is attached to the protein through a nucleic acid comprising the first ligand moiety. In some embodiments, the at least one detectable label is attached to the protein through a nucleic acid comprising the second ligand moiety.
[0221] As described elsewhere herein, shielded recognition molecules of the application may be used in a polypeptide sequencing method in accordance with the application, or any method known in the art. For example, in some embodiments, a shielded recognition molecule provided herein may be used in an Edman-type degradation reaction provided herein, or conventionally known in the art, which can involve iterative cycling of multiple reaction mixtures in a polypeptide sequencing reaction. In some embodiments, a shielded recognition molecule provided herein may be used in a dynamic sequencing reaction of the application, which involves amino acid recognition and degradation in a single reaction mixture.
Sequencing by Degradation of Labeled Polypeptides
[0222] In some aspects, the application provides a method of sequencing a polypeptide by identifying a unique combination of amino acids corresponding to a known polypeptide sequence. For example, FIG. 6 shows a method of sequencing by detecting selectively labeled amino acids of a labeled polypeptide 600. In some embodiments, labeled polypeptide 600 comprises selectively modified amino acids such that different amino acid types comprise different luminescent labels. As used herein, unless otherwise indicated, a labeled polypeptide refers to a polypeptide comprising one or more selectively labeled amino acid sidechains. Methods of selective labeling and details relating to the preparation and analysis of labeled polypeptides are known in the art (see, e.g., Swaminathan, et al. PLoS Comput Biol. 2015, 11(2):e1004080).
[0223] As shown, in some embodiments, labeled polypeptide 600 is immobilized and exposed to an excitation source. An aggregate luminescence from labeled polypeptide 600 is detected and, in some embodiments, exposure to luminescence over time results in a loss in detected signal due to luminescent label degradation (e.g., degradation due to photobleaching). In some embodiments, labeled polypeptide 600 comprises a unique combination of selectively labeled amino acids that give rise to an initial detected signal. As generically illustrated, degradation of luminescent labels over time results in a corresponding decrease in a detected signal for the photobleached labeled polypeptide 602. In some embodiments, the signal can be deconvoluted by analysis of one or more luminescence properties (e.g., signal deconvolution by luminescence lifetime analysis). In some embodiments, the unique combination of selectively labeled amino acids of labeled polypeptide 600 have been computationally precomputed and empirically verified--e.g., based on known polypeptide sequences of a proteome. In some embodiments, the combination of detected amino acid labels are compared against a database of known sequences of a proteome of an organism to identify a particular polypeptide of the database corresponding to labeled polypeptide 600.
[0224] In some embodiments, the approach illustrated in FIG. 6 may be modified by determining an optimal sample concentration for performing a sequencing reaction that maximizes sampling in massively parallel analysis. In some embodiments, the concentration is selected so that a desired fraction of the sample wells of an array (e.g., 30%) are occupied at any given time. Without wishing to be bound by theory, it is thought that while a polypeptide is bleached over a period of time, the same well continues to be available for further analysis. Through diffusion, approximately 30% of the sample wells of an array can be used for analysis every 3 minutes. As an illustrative example, in a million sample well chip, 6,000,000 polypeptides per hour may be sampled, or 24,000,000 over a 4 hour period.
[0225] In some aspects, the application provides a method of sequencing a polypeptide by detecting luminescence of a labeled polypeptide which is subjected to repeated cycles of terminal amino acid modification and cleavage. For example, FIG. 7 shows a method of sequencing a labeled polypeptide by Edman degradation in accordance with the application. In some embodiments, the method generally proceeds as described herein for other methods of sequencing by Edman degradation. For example, in some embodiments, steps (1) and (2) shown in FIG. 7 may be performed as described elsewhere herein for terminal amino acid modification and terminal amino acid cleavage, respectively, in an Edman degradation reaction.
[0226] As shown in the example depicted in FIG. 7, in some embodiments, the method comprises a step of (1) modifying the terminal amino acid of a labeled polypeptide. As described elsewhere herein, in some embodiments, modifying comprises contacting the terminal amino acid with an isothiocyanate (e.g., PITC) to form an isothiocyanate-modified terminal amino acid. In some embodiments, an isothiocyanate modification 710 converts the terminal amino acid to a form that is more susceptible to removal by a cleaving reagent (e.g., a chemical or enzymatic cleaving reagent, as described herein). Accordingly, in some embodiments, the method comprises a step of (2) removing the modified terminal amino acid using chemical or enzymatic means detailed elsewhere herein for Edman degradation.
[0227] In some embodiments, the method comprises repeating steps (1) through (2) for a plurality of cycles, during which luminescence of the labeled polypeptide is detected, and cleavage events corresponding to the removal of a labeled amino acid from the terminus may be detected as a decrease in detected signal. In some embodiments, no change in signal following step (2) as shown in FIG. 7 identifies an amino acid of unknown type. Accordingly, in some embodiments, partial sequence information may be determined by evaluating a signal detected following step (2) during each sequential round by assigning an amino acid type by a determined identity based on a change in detected signal or identifying an amino acid type as unknown based on no change in a detected signal.
[0228] In some aspects, a method of sequencing a polypeptide in accordance with the application comprises sequencing by processive enzymatic cleavage of a labeled polypeptide, as generally illustrated in FIGS. 8A-8C. As shown, in some embodiments, a labeled polypeptide is subjected to degradation using a modified processive exopeptidase that continuously cleaves a terminal amino acid from one terminus to another terminus. Exopeptidases are described in detail elsewhere herein. FIG. 8A depicts an example in which a labeled polypeptide 800 is subjected to degradation by an immobilized processive exopeptidase 810. FIG. 8B depicts an example in which an immobilized labeled polypeptide 820 is subjected to degradation by a processive exopeptidase 830.
[0229] FIG. 8C schematically illustrates an example of a real-time sequencing process performed in accordance with the method depicted in FIG. 8B. As shown, panels (I) through (IV) show a progression of labeled polypeptide degradation, with a corresponding signal trace over time shown below each panel. As shown, each cleavage event corresponding to a labeled amino acid gives rise to a concomitant drop in signal. In some embodiments, the rate of processivity of processive exopeptidase 830 is known, such that the timing between a detected decrease in signal may be used to calculate the number of unlabeled amino acids between each detection event. For example, if a polypeptide of 40 amino acids was cleaved in such a way that an amino acid was removed every second, a labeled polypeptide having 3 signals would show all 3 initially (panel (I)), then 2 (panel (II)), then 1 (panel (III)), and finally no signal. In this way, the order of the labeled amino acids can be determined. Accordingly, these methods may be used to determine partial sequence information, e.g., for proteomic analysis based on polypeptide fragment sequencing.
[0230] In some embodiments, single molecule protein sequencing can be achieved using an ATP-based Forster resonance energy transfer (FRET) scheme (e.g., with one or more labeled cofactors), for example as illustrated in FIG. 9. In some embodiments, sequencing by cofactor-based FRET can be performed using an immobilized ATP-dependent protease, donor-labeled ATP, and acceptor-labeled amino acids of a polypeptide substrate. In some embodiments, amino acids can be labeled with acceptors, and the one or more cofactors can be labeled with donors.
[0231] For example, in some embodiments, extracted proteins are denatured, and cysteines and lysines are labeled with fluorescent dyes. In some embodiments, an engineered version of a protein translocase (e.g., bacterial ClpX) is used to bind to individual substrate proteins, unfold them, and translocate them through its nano-channel. In some embodiments, the translocase is labeled with a donor dye, and FRET occurs between the donor on the translocase and two or more distinct acceptor dyes on a substrate when the substrate passes through the nano-channel. The order of the labeled amino acids can then be determined from the FRET signal. In some embodiments, one or more of the following non-limiting labeled ATP analogues shown in Table 5 can be used.
TABLE-US-00005 TABLE 5 Non-limiting examples of labeled ATP analogues. Phosphate-labeled ATP: ##STR00001## ##STR00002## ##STR00003## ##STR00004## ##STR00005## Ribose-labeled ATP: ##STR00006## ##STR00007## ##STR00008## Base-labeled ATP: ##STR00009## ##STR00010## ##STR00011##
Preparation of Samples for Sequencing
[0232] A polypeptide sample can be modified prior to sequencing. In some embodiments, the N-terminal amino acid or the C-terminal amino acid of a polypeptide is modified. FIG. 10A illustrates a non-limiting example of terminal end modification for preparing terminally modified polypeptides from a protein sample. In step (1), protein sample 1000 is fragmented to produce polypeptide fragments 1002. A polypeptide can be fragmented by cleaving (e.g., chemically) and/or digesting (e.g., enzymatically, for example using a peptidase, for example trypsin) a polypeptide of interest. Fragmentation can be performed before or after labeling. In some embodiments, fragmentation is performed after labeling of whole proteins. One or more amino acids can be labeled before or after cleavage to produce labeled polypeptides. In some embodiments, polypeptides are size selected after chemical or enzymatic fragmentation. In some embodiments, smaller polypeptides (e.g., <2 kDa) are removed and larger polypeptides are retained for sequence analysis. Size selection can be achieved using a technique such as gel filtration, SEC, dialysis, PAGE gel extraction, microfluidic tension flow, or any other suitable technique. In step (2), the N-termini or C-termini of polypeptide fragments 1002 are modified to produce terminally modified polypeptides 1004. In some embodiments, modification comprises adding an immobilization moiety. In some embodiments, modification comprises adding a coupling moiety.
[0233] Accordingly, provided herein are methods of modifying terminal ends of proteins and polypeptides with moieties that enable immobilization to a surface (e.g., a surface of a sample well on a chip used for protein analysis). In some embodiments, such methods comprise modifying a terminal end of a labeled polypeptide to be analyzed in accordance with the application. In yet other embodiments, such methods comprise modifying a terminal end of a protein or enzyme that degrades or translocates a protein or polypeptide substrate in accordance with the application.
[0234] In some embodiments, a carboxy-terminus of a protein or polypeptide is modified in a method comprising: (i) blocking free carboxylate groups of the protein or polypeptide; (ii) denaturing the protein or polypeptide (e.g., by heat and/or chemical means); (iii) blocking free thiol groups of the protein or polypeptide; (iv) digesting the protein or polypeptide to produce at least one polypeptide fragment comprising a free C-terminal carboxylate group; and (v) conjugating (e.g., chemically) a functional moiety to the free C-terminal carboxylate group. In some embodiments, the method further comprises, after (i) and before (ii), dialyzing a sample comprising the protein or polypeptide.
[0235] In some embodiments, a carboxy-terminus of a protein or polypeptide is modified in a method comprising: (i) denaturing the protein or polypeptide (e.g., by heat and/or chemical means); (ii) blocking free thiol groups of the protein or polypeptide; (iii) digesting the protein or polypeptide to produce at least one polypeptide fragment comprising a free C-terminal carboxylate group; (iv) blocking the free C-terminal carboxylate group to produce at least one polypeptide fragment comprising a blocked C-terminal carboxylate group; and (v) conjugating (e.g., enzymatically) a functional moiety to the blocked C-terminal carboxylate group. In some embodiments, the method further comprises, after (iv) and before (v), dialyzing a sample comprising the protein or polypeptide.
[0236] In some embodiments, blocking free carboxylate groups refers to a chemical modification of these groups which alters chemical reactivity relative to an unmodified carboxylate. Suitable carboxylate blocking methods are known in the art and should modify side-chain carboxylate groups to be chemically different from a carboxy-terminal carboxylate group of a polypeptide to be functionalized. In some embodiments, blocking free carboxylate groups comprises esterification or amidation of free carboxylate groups of a polypeptide. In some embodiments, blocking free carboxylate groups comprises methyl esterification of free carboxylate groups of a polypeptide, e.g., by reacting the polypeptide with methanolic HCl. Additional examples of reagents and techniques useful for blocking free carboxylate groups include, without limitation, 4-sulfo-2,3,5,6-tetrafluorophenol (STP) and/or a carbodiimide such as N-(3-Dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (EDAC), uronium reagents, diazomethane, alcohols and acid for Fischer esterification, the use of N-hydroxylsuccinimide (NHS) to form NHS esters (potentially as an intermediate to subsequent ester or amine formation), or reaction with carbonyldiimidazole (CDI) or the formation of mixed anhydrides, or any other method of modifying or blocking carboxylic acids, potentially through the formation of either esters or amides.
[0237] In some embodiments, blocking free thiol groups refers to a chemical modification of these groups which alters chemical reactivity relative to an unmodified thiol. In some embodiments, blocking free thiol groups comprises reducing and alkylating free thiol groups of a protein or polypeptide. In some embodiments, reduction and alkylation is carried out by contacting a polypeptide with dithiothreitol (DTT) and one or both of iodoacetamide and iodoacetic acid. Examples of additional and alternative cysteine-reducing reagents which may be used are well known and include, without limitation, 2-mercaptoethanol, Tris (2-carboxyehtyl) phosphine hydrochloride (TCEP), tributylphosphine, dithiobutylamine (DTBA), or any reagent capable of reducing a thiol group. Examples of additional and alternative cysteine-blocking (e.g., cysteine-alkylating) reagents which may be used are well known and include, without limitation, acrylamide, 4-vinylpyridine, N-Ethylmalemide (NEM), N-.epsilon.-maleimidocaproic acid (EMCA), or any reagent that modifies cysteines so as to prevent disulfide bond formation.
[0238] In some embodiments, digestion comprises enzymatic digestion. In some embodiments, digestion is carried out by contacting a protein or polypeptide with an endopeptidase (e.g., trypsin) under digestion conditions. In some embodiments, digestion comprises chemical digestion. Examples of suitable reagents for chemical and enzymatic digestion are known in the art and include, without limitation, trypsin, chemotrypsin, Lys-C, Arg-C, Asp-N, Lys-N, BNPS-Skatole, CNBr, caspase, formic acid, glutamyl endopeptidase, hydroxylamine, iodosobenzoic acid, neutrophil elastase, pepsin, proline-endopeptidase, proteinase K, staphylococcal peptidase I, thermolysin, and thrombin.
[0239] In some embodiments, the functional moiety comprises a biotin molecule. In some embodiments, the functional moiety comprises a reactive chemical moiety, such as an alkynyl. In some embodiments, conjugating a functional moiety comprises biotinylation of carboxy-terminal carboxy-methyl ester groups by carboxypeptidase Y, as known in the art.
[0240] In some embodiments, a solubilizing moiety is added to a polypeptide. FIG. 10B illustrates a non-limiting example of a solubilizing moiety added to a terminal amino acid of a polypeptide, for example using a process of conjugating a solubilizing linker to the polypeptide.
[0241] In some embodiments, a terminally modified polypeptide 1010 comprising a linker conjugating moiety 1012 is conjugated to a solubilizing linker 1020 comprising a polypeptide conjugating moiety 1022. In some embodiments, the solubilizing linker comprises a solubilizing polymer, such as a biomolecule (e.g., shown as stippled shape). In some embodiments, a resulting linker-conjugated polypeptide 1030 comprising a linkage 1032 formed between 1012 and 1022 further comprises a surface conjugating moiety 1034. Accordingly, in some embodiments methods and compositions provided herein are useful for modifying terminal ends of polypeptides with moieties that increase their solubility. In some embodiments, a solubilizing moiety is useful for small polypeptides that result from fragmentation (e.g., enzymatic fragmentation, for example using trypsin) and that are relatively insoluble. For example, in some embodiments, short polypeptides in a polypeptide pool can be solubilized by conjugating a polymer (e.g., a short oligo, a sugar, or other charged polymer) to the polypeptides.
[0242] In some embodiments, one or more surfaces of a sample well (e.g., sidewalls of a sample well) can be modified. A non-limiting example of passivation and/or antifouling of a sample well sidewall is shown in FIG. 10C where an example schematic of a sample well is illustrated with modified surfaces which may be used to promote single molecule immobilization to a bottom surface. In some embodiments, 1040 is SiO.sub.2. In some embodiments, 1042 is a polypeptide conjugating moiety (e.g., TCO, tetrazine, N3, alkyne, aldehyde, NCO, NHS, thiol, alkene, DBCO, BCN, TPP, biotin, or other suitable conjugating moiety). In some embodiments, 1050 is TiO.sub.2 or Al.sub.2O.sub.3. In some embodiments, 1052 is a hydrophobic C.sub.4-18 molecule, a polytetrafluoroethylene compound (e.g., (CF.sub.2).sub.4-12), a polyol, such as a polyethylene glycol (e.g., PEG.sub.3-100), polypropylene glycol, polyoxyethylene glycol, or combinations or variations thereof, or a zwitterion, such as sulfobetaine. In some embodiments, 1060 is Si. In some embodiments, 1070 is Al. In some embodiments, 1080 is TiN.
Luminescent Labels
[0243] As used herein, a luminescent label is a molecule that absorbs one or more photons and may subsequently emit one or more photons after one or more time durations. In some embodiments, the term is used interchangeably with "label" or "luminescent molecule" depending on context. A luminescent label in accordance with certain embodiments described herein may refer to a luminescent label of a labeled affinity reagent, a luminescent label of a labeled peptidase (e.g., a labeled exopeptidase, a labeled non-specific exopeptidase), a luminescent label of a labeled peptide, a luminescent label of a labeled cofactor, or another labeled composition described herein. In some embodiments, a luminescent label in accordance with the application refers to a labeled amino acid of a labeled polypeptide comprising one or more labeled amino acids.
[0244] In some embodiments, a luminescent label may comprise a first and second chromophore. In some embodiments, an excited state of the first chromophore is capable of relaxation via an energy transfer to the second chromophore. In some embodiments, the energy transfer is a Forster resonance energy transfer (FRET). Such a FRET pair may be useful for providing a luminescent label with properties that make the label easier to differentiate from amongst a plurality of luminescent labels in a mixture--e.g., as illustrated and described herein for labeled aptamer 106 of FIG. 1C. In yet other embodiments, a FRET pair comprises a first chromophore of a first luminescent label and a second chromophore of a second luminescent label--e.g., as illustrated and described herein for sequencing of labeled peptides using a labeled cofactor (see, e.g., FIG. 9). In certain embodiments, the FRET pair may absorb excitation energy in a first spectral range and emit luminescence in a second spectral range.
[0245] In some embodiments, a luminescent label refers to a fluorophore or a dye. Typically, a luminescent label comprises an aromatic or heteroaromatic compound and can be a pyrene, anthracene, naphthalene, naphthylamine, acridine, stilbene, indole, benzindole, oxazole, carbazole, thiazole, benzothiazole, benzoxazole, phenanthridine, phenoxazine, porphyrin, quinoline, ethidium, benzamide, cyanine, carbocyanine, salicylate, anthranilate, coumarin, fluoroscein, rhodamine, xanthene, or other like compound.
[0246] In some embodiments, a luminescent label comprises a dye selected from one or more of the following: 5/6-Carboxyrhodamine 6G, 5-Carboxyrhodamine 6G, 6-Carboxyrhodamine 6G, 6-TAMRA, Abberior.RTM. STAR 440SXP, Abberior.RTM. STAR 470SXP, Abberior.RTM. STAR 488, Abberior.RTM. STAR 512, Abberior.RTM. STAR 520SXP, Abberior.RTM. STAR 580, Abberior.RTM. STAR 600, Abberior.RTM. STAR 635, Abberior.RTM. STAR 635P, Abberior.RTM. STAR RED, Alexa Fluor.RTM. 350, Alexa Fluor.RTM. 405, Alexa Fluor.RTM. 430, Alexa Fluor.RTM. 480, Alexa Fluor.RTM. 488, Alexa Fluor.RTM. 514, Alexa Fluor.RTM. 532, Alexa Fluor.RTM. 546, Alexa Fluor.RTM. 555, Alexa Fluor.RTM. 568, Alexa Fluor.RTM. 594, Alexa Fluor.RTM. 610-X, Alexa Fluor.RTM. 633, Alexa Fluor.RTM. 647, Alexa Fluor.RTM. 660, Alexa Fluor.RTM. 680, Alexa Fluor.RTM. 700, Alexa Fluor.RTM. 750, Alexa Fluor.RTM. 790, AMCA, ATTO 390, ATTO 425, ATTO 465, ATTO 488, ATTO 495, ATTO 514, ATTO 520, ATTO 532, ATTO 542, ATTO 550, ATTO 565, ATTO 590, ATTO 610, ATTO 620, ATTO 633, ATTO 647, ATTO 647N, ATTO 655, ATTO 665, ATTO 680, ATTO 700, ATTO 725, ATTO 740, ATTO Oxa12, ATTO Rho101, ATTO Rho11, ATTO Rho12, ATTO Rho13, ATTO Rho14, ATTO Rho3B, ATTO Rho6G, ATTO Thio12, BD Horizon.TM. V450, BODIPY.RTM. 493/501, BODIPY.RTM. 530/550, BODIPY.RTM. 558/568, BODIPY.RTM. 564/570, BODIPY.RTM. 576/589, BODIPY.RTM. 581/591, BODIPY.RTM. 630/650, BODIPY.RTM. 650/665, BODIPY.RTM. FL, BODIPY.RTM. FL-X, BODIPY.RTM. R6G, BODIPY.RTM. TMR, BODIPY.RTM. TR, CAL Fluor.RTM. Gold 540, CAL Fluor.RTM. Green 510, CAL Fluor.RTM. Orange 560, CAL Fluor.RTM. Red 590, CAL Fluor.RTM. Red 610, CAL Fluor.RTM. Red 615, CAL Fluor.RTM. Red 635, Cascade.RTM. Blue, CF.TM.350, CF.TM.405M, CF.TM.405S, CF.TM.488A, CF.TM.514, CF.TM.532, CF.TM.543, CF.TM.546, CF.TM.555, CF.TM.568, CF.TM.594, CF.TM.620R, CF.TM.633, CF.TM.633-V1, CF.TM.640R, CF.TM.640R-V1, CF.TM.640R-V2, CF.TM.660C, CF.TM.660R, CF.TM.680, CF.TM.680R, CF.TM.680R-V1, CF.TM.750, CF.TM.770, CF.TM.790, Chromeo.TM. 642, Chromis 425N, Chromis 500N, Chromis 515N, Chromis 530N, Chromis 550A, Chromis 550C, Chromis 550Z, Chromis 560N, Chromis 570N, Chromis 577N, Chromis 600N, Chromis 630N, Chromis 645A, Chromis 645C, Chromis 645Z, Chromis 678A, Chromis 678C, Chromis 678Z, Chromis 770A, Chromis 770C, Chromis 800A, Chromis 800C, Chromis 830A, Chromis 830C, Cy.RTM.3, Cy.RTM.3.5, Cy.RTM.3B, Cy.RTM.5, Cy.RTM.5.5, Cy.RTM.7, DyLight.RTM. 350, DyLight.RTM. 405, DyLight.RTM. 415-Col, DyLight.RTM. 425Q, DyLight.RTM. 485-LS, DyLight.RTM. 488, DyLight.RTM. 504Q, DyLight.RTM. 510-LS, DyLight.RTM. 515-LS, DyLight.RTM. 521-LS, DyLight.RTM. 530-R2, DyLight.RTM. 543Q, DyLight.RTM. 550, DyLight.RTM. 554-RO, DyLight.RTM. 554-R1, DyLight.RTM. 590-R2, DyLight.RTM. 594, DyLight.RTM. 610-B1, DyLight.RTM. 615-B2, DyLight.RTM. 633, DyLight.RTM. 633-B1, DyLight.RTM. 633-B2, DyLight.RTM. 650, DyLight.RTM. 655-B1, DyLight.RTM. 655-B2, DyLight.RTM. 655-B3, DyLight.RTM. 655-B4, DyLight.RTM. 662Q, DyLight.RTM. 675-B1, DyLight.RTM. 675-B2, DyLight.RTM. 675-B3, DyLight.RTM. 675-B4, DyLight.RTM. 679-05, DyLight.RTM. 680, DyLight.RTM. 683Q, DyLight.RTM. 690-B1, DyLight.RTM. 690-B2, DyLight.RTM. 696Q, DyLight.RTM. 700-B1, DyLight.RTM. 700-B1, DyLight.RTM. 730-B1, DyLight.RTM. 730-B2, DyLight.RTM. 730-B3, DyLight.RTM. 730-B4, DyLight.RTM. 747, DyLight.RTM. 747-B1, DyLight.RTM. 747-B2, DyLight.RTM. 747-B3, DyLight.RTM. 747-B4, DyLight.RTM. 755, DyLight.RTM. 766Q, DyLight.RTM. 775-B2, DyLight.RTM. 775-B3, DyLight.RTM. 775-B4, DyLight.RTM. 780-B1, DyLight.RTM. 780-B2, DyLight.RTM. 780-B3, DyLight.RTM. 800, DyLight.RTM. 830-B2, Dyomics-350, Dyomics-350XL, Dyomics-360XL, Dyomics-370XL, Dyomics-375XL, Dyomics-380XL, Dyomics-390XL, Dyomics-405, Dyomics-415, Dyomics-430, Dyomics-431, Dyomics-478, Dyomics-480XL, Dyomics-481XL, Dyomics-485XL, Dyomics-490, Dyomics-495, Dyomics-505, Dyomics-510XL, Dyomics-511XL, Dyomics-520XL, Dyomics-521XL, Dyomics-530, Dyomics-547, Dyomics-547P1, Dyomics-548, Dyomics-549, Dyomics-549P1, Dyomics-550, Dyomics-554, Dyomics-555, Dyomics-556, Dyomics-560, Dyomics-590, Dyomics-591, Dyomics-594, Dyomics-601XL, Dyomics-605, Dyomics-610, Dyomics-615, Dyomics-630, Dyomics-631, Dyomics-632, Dyomics-633, Dyomics-634, Dyomics-635, Dyomics-636, Dyomics-647, Dyomics-647P1, Dyomics-648, Dyomics-648P1, Dyomics-649, Dyomics-649P1, Dyomics-650, Dyomics-651, Dyomics-652, Dyomics-654, Dyomics-675, Dyomics-676, Dyomics-677, Dyomics-678, Dyomics-679P1, Dyomics-680, Dyomics-681, Dyomics-682, Dyomics-700, Dyomics-701, Dyomics-703, Dyomics-704, Dyomics-730, Dyomics-731, Dyomics-732, Dyomics-734, Dyomics-749, Dyomics-749P1, Dyomics-750, Dyomics-751, Dyomics-752, Dyomics-754, Dyomics-776, Dyomics-777, Dyomics-778, Dyomics-780, Dyomics-781, Dyomics-782, Dyomics-800, Dyomics-831, eFluor.RTM. 450, Eosin, FITC, Fluorescein, HiLyte.TM. Fluor 405, HiLyte.TM. Fluor 488, HiLyte.TM. Fluor 532, HiLyte.TM. Fluor 555, HiLyte.TM. Fluor 594, HiLyte.TM. Fluor 647, HiLyte.TM. Fluor 680, HiLyte.TM. Fluor 750, IRDye.RTM. 680LT, IRDye.RTM. 750, IRDye.RTM. 800CW, JOE, LightCycler.RTM. 640R, LightCycler.RTM. Red 610, LightCycler.RTM. Red 640, LightCycler.RTM. Red 670, LightCycler.RTM. Red 705, Lissamine Rhodamine B, Napthofluorescein, Oregon Green.RTM. 488, Oregon Green.RTM. 514, Pacific Blue.TM. Pacific Green.TM., Pacific Orange.TM., PET, PF350, PF405, PF415, PF488, PF505, PF532, PF546, PF555P, PF568, PF594, PF610, PF633P, PF647P, Quasar.RTM. 570, Quasar.RTM. 670, Quasar.RTM. 705, Rhodamine 123, Rhodamine 6G, Rhodamine B, Rhodamine Green, Rhodamine Green-X, Rhodamine Red, ROX, Seta.TM. 375, Seta.TM. 470, Seta.TM. 555, Seta.TM. 632, Seta.TM. 633, Seta.TM. 650, Seta.TM. 660, Seta.TM. 670, Seta.TM. 680, Seta.TM. 700, Seta.TM. 750, Seta.TM. 780, Seta.TM. APC-780, Seta.TM. PerCP-680, Seta.TM. R-PE-670, Seta.TM. 646, SeTau 380, SeTau 425, SeTau 647, SeTau 405, Square 635, Square 650, Square 660, Square 672, Square 680, Sulforhodamine 101, TAMRA, TET, Texas Red.RTM., TMR, TRITC, Yakima Yellow.TM. Zenon.RTM., Zy3, ZyS, Zy5.5, and Zy7.
Luminescence
[0247] In some aspects, the application relates to polypeptide sequencing and/or identification based on one or more luminescence properties of a luminescent label. In some embodiments, a luminescent label is identified based on luminescence lifetime, luminescence intensity, brightness, absorption spectra, emission spectra, luminescence quantum yield, or a combination of two or more thereof. In some embodiments, a plurality of types of luminescent labels can be distinguished from each other based on different luminescence lifetimes, luminescence intensities, brightnesses, absorption spectra, emission spectra, luminescence quantum yields, or combinations of two or more thereof. Identifying may mean assigning the exact identity and/or quantity of one type of amino acid (e.g., a single type or a subset of types) associated with a luminescent label, and may also mean assigning an amino acid location in a polypeptide relative to other types of amino acids.
[0248] In some embodiments, luminescence is detected by exposing a luminescent label to a series of separate light pulses and evaluating the timing or other properties of each photon that is emitted from the label. In some embodiments, information for a plurality of photons emitted sequentially from a label is aggregated and evaluated to identify the label and thereby identify an associated type of amino acid. In some embodiments, a luminescence lifetime of a label is determined from a plurality of photons that are emitted sequentially from the label, and the luminescence lifetime can be used to identify the label. In some embodiments, a luminescence intensity of a label is determined from a plurality of photons that are emitted sequentially from the label, and the luminescence intensity can be used to identify the label. In some embodiments, a luminescence lifetime and luminescence intensity of a label is determined from a plurality of photons that are emitted sequentially from the label, and the luminescence lifetime and luminescence intensity can be used to identify the label.
[0249] In some aspects of the application, a single polypeptide molecule is exposed to a plurality of separate light pulses and a series of emitted photons are detected and analyzed. In some embodiments, the series of emitted photons provides information about the single polypeptide molecule that is present and that does not change in the reaction sample over the time of the experiment. However, in some embodiments, the series of emitted photons provides information about a series of different molecules that are present at different times in the reaction sample (e.g., as a reaction or process progresses). By way of example and not limitation, such information may be used to sequence and/or identify a polypeptide subjected to chemical or enzymatic degradation in accordance with the application.
[0250] In certain embodiments, a luminescent label absorbs one photon and emits one photon after a time duration. In some embodiments, the luminescence lifetime of a label can be determined or estimated by measuring the time duration. In some embodiments, the luminescence lifetime of a label can be determined or estimated by measuring a plurality of time durations for multiple pulse events and emission events. In some embodiments, the luminescence lifetime of a label can be differentiated amongst the luminescence lifetimes of a plurality of types of labels by measuring the time duration. In some embodiments, the luminescence lifetime of a label can be differentiated amongst the luminescence lifetimes of a plurality of types of labels by measuring a plurality of time durations for multiple pulse events and emission events. In certain embodiments, a label is identified or differentiated amongst a plurality of types of labels by determining or estimating the luminescence lifetime of the label. In certain embodiments, a label is identified or differentiated amongst a plurality of types of labels by differentiating the luminescence lifetime of the label amongst a plurality of the luminescence lifetimes of a plurality of types of labels.
[0251] Determination of a luminescence lifetime of a luminescent label can be performed using any suitable method (e.g., by measuring the lifetime using a suitable technique or by determining time-dependent characteristics of emission). In some embodiments, determining the luminescence lifetime of one label comprises determining the lifetime relative to another label. In some embodiments, determining the luminescence lifetime of a label comprises determining the lifetime relative to a reference. In some embodiments, determining the luminescence lifetime of a label comprises measuring the lifetime (e.g., fluorescence lifetime). In some embodiments, determining the luminescence lifetime of a label comprises determining one or more temporal characteristics that are indicative of lifetime. In some embodiments, the luminescence lifetime of a label can be determined based on a distribution of a plurality of emission events (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, or more emission events) occurring across one or more time-gated windows relative to an excitation pulse. For example, a luminescence lifetime of a label can be distinguished from a plurality of labels having different luminescence lifetimes based on the distribution of photon arrival times measured with respect to an excitation pulse.
[0252] It should be appreciated that a luminescence lifetime of a luminescent label is indicative of the timing of photons emitted after the label reaches an excited state and the label can be distinguished by information indicative of the timing of the photons. Some embodiments may include distinguishing a label from a plurality of labels based on the luminescence lifetime of the label by measuring times associated with photons emitted by the label. The distribution of times may provide an indication of the luminescence lifetime which may be determined from the distribution. In some embodiments, the label is distinguishable from the plurality of labels based on the distribution of times, such as by comparing the distribution of times to a reference distribution corresponding to a known label. In some embodiments, a value for the luminescence lifetime is determined from the distribution of times.
[0253] As used herein, in some embodiments, luminescence intensity refers to the number of emitted photons per unit time that are emitted by a luminescent label which is being excited by delivery of a pulsed excitation energy. In some embodiments, the luminescence intensity refers to the detected number of emitted photons per unit time that are emitted by a label which is being excited by delivery of a pulsed excitation energy, and are detected by a particular sensor or set of sensors.
[0254] As used herein, in some embodiments, brightness refers to a parameter that reports on the average emission intensity per luminescent label. Thus, in some embodiments, "emission intensity" may be used to generally refer to brightness of a composition comprising one or more labels. In some embodiments, brightness of a label is equal to the product of its quantum yield and extinction coefficient.
[0255] As used herein, in some embodiments, luminescence quantum yield refers to the fraction of excitation events at a given wavelength or within a given spectral range that lead to an emission event, and is typically less than 1. In some embodiments, the luminescence quantum yield of a luminescent label described herein is between 0 and about 0.001, between about 0.001 and about 0.01, between about 0.01 and about 0.1, between about 0.1 and about 0.5, between about 0.5 and 0.9, or between about 0.9 and 1. In some embodiments, a label is identified by determining or estimating the luminescence quantum yield.
[0256] As used herein, in some embodiments, an excitation energy is a pulse of light from a light source. In some embodiments, an excitation energy is in the visible spectrum. In some embodiments, an excitation energy is in the ultraviolet spectrum. In some embodiments, an excitation energy is in the infrared spectrum. In some embodiments, an excitation energy is at or near the absorption maximum of a luminescent label from which a plurality of emitted photons are to be detected. In certain embodiments, the excitation energy is between about 500 nm and about 700 nm (e.g., between about 500 nm and about 600 nm, between about 600 nm and about 700 nm, between about 500 nm and about 550 nm, between about 550 nm and about 600 nm, between about 600 nm and about 650 nm, or between about 650 nm and about 700 nm). In certain embodiments, an excitation energy may be monochromatic or confined to a spectral range. In some embodiments, a spectral range has a range of between about 0.1 nm and about 1 nm, between about 1 nm and about 2 nm, or between about 2 nm and about 5 nm. In some embodiments, a spectral range has a range of between about 5 nm and about 10 nm, between about 10 nm and about 50 nm, or between about 50 nm and about 100 nm.
Sequencing
[0257] Aspects of the application relate to sequencing biological polymers, such as polypeptides and proteins. As used herein, "sequencing," "sequence determination," "determining a sequence," and like terms, in reference to a polypeptide or protein includes determination of partial sequence information as well as full sequence information of the polypeptide or protein. That is, the terminology includes sequence comparisons, fingerprinting, probabalistic fingerprinting, and like levels of information about a target molecule, as well as the express identification and ordering of each amino acid of the target molecule within a region of interest. In some embodiments, the terminology includes identifying a single amino acid of a polypeptide. In yet other embodiments, more than one amino acid of a polypeptide is identified. As used herein, in some embodiments, "identifying," "determining the identity," and like terms, in reference to an amino acid includes determination of an express identity of an amino acid as well as determination of a probability of an express identity of an amino acid. For example, in some embodiments, an amino acid is identified by determining a probability (e.g., from 0% to 100%) that the amino acid is of a specific type, or by determining a probability for each of a plurality of specific types. Accordingly, in some embodiments, the terms "amino acid sequence," "polypeptide sequence," and "protein sequence" as used herein may refer to the polypeptide or protein material itself and is not restricted to the specific sequence information (e.g., the succession of letters representing the order of amino acids from one terminus to another terminus) that biochemically characterizes a specific polypeptide or protein.
[0258] In some embodiments, sequencing of a polypeptide molecule comprises identifying at least two (e.g., at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 60, at least 70, at least 80, at least 90, at least 100, or more) amino acids in the polypeptide molecule. In some embodiments, the at least two amino acids are contiguous amino acids. In some embodiments, the at least two amino acids are non-contiguous amino acids.
[0259] In some embodiments, sequencing of a polypeptide molecule comprises identification of less than 100% (e.g., less than 99%, less than 95%, less than 90%, less than 85%, less than 80%, less than 75%, less than 70%, less than 65%, less than 60%, less than 55%, less than 50%, less than 45%, less than 40%, less than 35%, less than 30%, less than 25%, less than 20%, less than 15%, less than 10%, less than 5%, less than 1% or less) of all amino acids in the polypeptide molecule. For example, in some embodiments, sequencing of a polypeptide molecule comprises identification of less than 100% of one type of amino acid in the polypeptide molecule (e.g., identification of a portion of all amino acids of one type in the polypeptide molecule). In some embodiments, sequencing of a polypeptide molecule comprises identification of less than 100% of each type of amino acid in the polypeptide molecule.
[0260] In some embodiments, sequencing of a polypeptide molecule comprises identification of at least 1, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100 or more types of amino acids in the polypeptide.
[0261] In some embodiments, the application provides compositions and methods for sequencing a polypeptide by identifying a series of amino acids that are present at a terminus of a polypeptide over time (e.g., by iterative detection and cleavage of amino acids at the terminus). In yet other embodiments, the application provides compositions and methods for sequencing a polypeptide by identifying labeled amino content of the polypeptide and comparing to a reference sequence database.
[0262] In some embodiments, the application provides compositions and methods for sequencing a polypeptide by sequencing a plurality of fragments of the polypeptide. In some embodiments, sequencing a polypeptide comprises combining sequence information for a plurality of polypeptide fragments to identify and/or determine a sequence for the polypeptide. In some embodiments, combining sequence information may be performed by computer hardware and software. The methods described herein may allow for a set of related polypeptides, such as an entire proteome of an organism, to be sequenced. In some embodiments, a plurality of single molecule sequencing reactions are performed in parallel (e.g., on a single chip) according to aspects of the present application. For example, in some embodiments, a plurality of single molecule sequencing reactions are each performed in separate sample wells on a single chip.
[0263] In some embodiments, methods provided herein may be used for the sequencing and identification of an individual protein in a sample comprising a complex mixture of proteins. In some embodiments, the application provides methods of uniquely identifying an individual protein in a complex mixture of proteins. In some embodiments, an individual protein is detected in a mixed sample by determining a partial amino acid sequence of the protein. In some embodiments, the partial amino acid sequence of the protein is within a contiguous stretch of approximately 5 to 50 amino acids.
[0264] Without wishing to be bound by any particular theory, it is believed that most human proteins can be identified using incomplete sequence information with reference to proteomic databases. For example, simple modeling of the human proteome has shown that approximately 98% of proteins can be uniquely identified by detecting just four types of amino acids within a stretch of 6 to 40 amino acids (see, e.g., Swaminathan, et al. PLoS Comput Biol. 2015, 11(2):e1004080; and Yao, et al. Phys. Biol. 2015, 12(5):055003). Therefore, a complex mixture of proteins can be degraded (e.g., chemically degraded, enzymatically degraded) into short polypeptide fragments of approximately 6 to 40 amino acids, and sequencing of this polypeptide library would reveal the identity and abundance of each of the proteins present in the original complex mixture. Compositions and methods for selective amino acid labeling and identifying polypeptides by determining partial sequence information are described in detail in U.S. patent application Ser. No. 15/510,962, filed Sep. 15, 2015, titled "SINGLE MOLECULE PEPTIDE SEQUENCING," which is incorporated by reference in its entirety.
[0265] Embodiments are capable of sequencing single polypeptide molecules with high accuracy, such as an accuracy of at least about 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, 99.99%, 99.999%, or 99.9999%. In some embodiments, the target molecule used in single molecule sequencing is a polypeptide that is immobilized to a surface of a solid support such as a bottom surface or a sidewall surface of a sample well. The sample well also can contain any other reagents needed for a sequencing reaction in accordance with the application, such as one or more suitable buffers, co-factors, labeled affinity reagents, and enzymes (e.g., catalytically active or inactive exopeptidase enzymes, which may be luminescently labeled or unlabeled).
[0266] As described above, in some embodiments, sequencing in accordance with the application comprises identifying an amino acid by determining a probability that the amino acid is of a specific type. Conventional protein identification systems require identification of each amino acid in a polypeptide to identify the polypeptide. However, it is difficult to accurately identify each amino acid in a polypeptide. For example, data collected from an interaction in which a first recognition molecule associates with a first amino acid may not be sufficiently different from data collected from an interaction in which a second recognition molecule associates with a second amino acid to differentiate between the two amino acids. In some embodiments, sequencing in accordance with the application avoids this problem by using a protein identification system that, unlike conventional protein identification systems, does not require (but does not preclude) identification of each amino acid in the protein.
[0267] Accordingly, in some embodiments, sequencing in accordance with the application may be carried out using a protein identification system that uses machine learning techniques to identify proteins. In some embodiments, the system operates by: (1) collecting data about a polypeptide of a protein using a real-time protein sequencing device; (2) using a machine learning model and the collected data to identify probabilities that certain amino acids are part of the polypeptide at respective locations; and (3) using the identified probabilities, as a "probabilistic fingerprint" to identify the protein. In some embodiments, data about the polypeptide of the protein may be obtained using reagents that selectively bind amino acids. As an example, the reagents and/or amino acids may be labeled with luminescent labels that emit light in response to application of excitation energy. In this example, a protein sequencing device may apply excitation energy to a sample of a protein (e.g., a polypeptide) during binding interactions of reagents with amino acids in the sample. In some embodiments, one or more sensors in the sequencing device (e.g., a photodetector, an electrical sensor, and/or any other suitable type of sensor) may detect binding interactions. In turn, the data collected and/or derived from the detected light emissions may be provided to the machine learning model. Machine learning models and associated systems and methods are described in detail in U.S. Provisional Patent Appl. No. 62/860,750, filed Jun. 12, 2019, titled "MACHINE LEARNING ENABLED PROTEIN IDENTIFICATION," which is incorporated by reference in its entirety.
[0268] Sequencing in accordance with the application, in some aspects, may involve immobilizing a polypeptide on a surface of a substrate (e.g., of a solid support, for example a chip, for example an integrated device as described herein). In some embodiments, a polypeptide may be immobilized on a surface of a sample well (e.g., on a bottom surface of a sample well) on a substrate. In some embodiments, the N-terminal amino acid of the polypeptide is immobilized (e.g., attached to the surface). In some embodiments, the C-terminal amino acid of the polypeptide is immobilized (e.g., attached to the surface). In some embodiments, one or more non-terminal amino acids are immobilized (e.g., attached to the surface). The immobilized amino acid(s) can be attached using any suitable covalent or non-covalent linkage, for example as described in this application. In some embodiments, a plurality of polypeptides are attached to a plurality of sample wells (e.g., with one polypeptide attached to a surface, for example a bottom surface, of each sample well), for example in an array of sample wells on a substrate.
[0269] Sequencing in accordance with the application, in some aspects, may be performed using a system that permits single molecule analysis. The system may include an integrated device and an instrument configured to interface with the integrated device. The integrated device may include an array of pixels, where individual pixels include a sample well and at least one photodetector. The sample wells of the integrated device may be formed on or through a surface of the integrated device and be configured to receive a sample placed on the surface of the integrated device. Collectively, the sample wells may be considered as an array of sample wells. The plurality of sample wells may have a suitable size and shape such that at least a portion of the sample wells receive a single sample (e.g., a single molecule, such as a polypeptide). In some embodiments, the number of samples within a sample well may be distributed among the sample wells of the integrated device such that some sample wells contain one sample while others contain zero, two or more samples.
[0270] Excitation light is provided to the integrated device from one or more light source external to the integrated device. Optical components of the integrated device may receive the excitation light from the light source and direct the light towards the array of sample wells of the integrated device and illuminate an illumination region within the sample well. In some embodiments, a sample well may have a configuration that allows for the sample to be retained in proximity to a surface of the sample well, which may ease delivery of excitation light to the sample and detection of emission light from the sample. A sample positioned within the illumination region may emit emission light in response to being illuminated by the excitation light. For example, the sample may be labeled with a fluorescent marker, which emits light in response to achieving an excited state through the illumination of excitation light. Emission light emitted by a sample may then be detected by one or more photodetectors within a pixel corresponding to the sample well with the sample being analyzed. When performed across the array of sample wells, which may range in number between approximately 10,000 pixels to 1,000,000 pixels according to some embodiments, multiple samples can be analyzed in parallel.
[0271] The integrated device may include an optical system for receiving excitation light and directing the excitation light among the sample well array. The optical system may include one or more grating couplers configured to couple excitation light to the integrated device and direct the excitation light to other optical components. The optical system may include optical components that direct the excitation light from a grating coupler towards the sample well array. Such optical components may include optical splitters, optical combiners, and waveguides. In some embodiments, one or more optical splitters may couple excitation light from a grating coupler and deliver excitation light to at least one of the waveguides. According to some embodiments, the optical splitter may have a configuration that allows for delivery of excitation light to be substantially uniform across all the waveguides such that each of the waveguides receives a substantially similar amount of excitation light. Such embodiments may improve performance of the integrated device by improving the uniformity of excitation light received by sample wells of the integrated device. Examples of suitable components, e.g., for coupling excitation light to a sample well and/or directing emission light to a photodetector, to include in an integrated device are described in U.S. patent application Ser. No. 14/821,688, filed Aug. 7, 2015, titled "INTEGRATED DEVICE FOR PROBING, DETECTING AND ANALYZING MOLECULES," and U.S. patent application Ser. No. 14/543,865, filed Nov. 17, 2014, titled "INTEGRATED DEVICE WITH EXTERNAL LIGHT SOURCE FOR PROBING, DETECTING, AND ANALYZING MOLECULES," both of which are incorporated by reference in their entirety. Examples of suitable grating couplers and waveguides that may be implemented in the integrated device are described in U.S. patent application Ser. No. 15/844,403, filed Dec. 15, 2017, titled "OPTICAL COUPLER AND WAVEGUIDE SYSTEM," which is incorporated by reference in its entirety.
[0272] Additional photonic structures may be positioned between the sample wells and the photodetectors and configured to reduce or prevent excitation light from reaching the photodetectors, which may otherwise contribute to signal noise in detecting emission light. In some embodiments, metal layers which may act as a circuitry for the integrated device, may also act as a spatial filter. Examples of suitable photonic structures may include spectral filters, a polarization filters, and spatial filters and are described in U.S. patent application Ser. No. 16/042,968, filed Jul. 23, 2018, titled "OPTICAL REJECTION PHOTONIC STRUCTURES," which is incorporated by reference in its entirety.
[0273] Components located off of the integrated device may be used to position and align an excitation source to the integrated device. Such components may include optical components including lenses, mirrors, prisms, windows, apertures, attenuators, and/or optical fibers. Additional mechanical components may be included in the instrument to allow for control of one or more alignment components. Such mechanical components may include actuators, stepper motors, and/or knobs. Examples of suitable excitation sources and alignment mechanisms are described in U.S. patent application Ser. No. 15/161,088, filed May 20, 2016, titled "PULSED LASER AND SYSTEM," which is incorporated by reference in its entirety. Another example of a beam-steering module is described in U.S. patent application Ser. No. 15/842,720, filed Dec. 14, 2017, titled "COMPACT BEAM SHAPING AND STEERING ASSEMBLY," which is incorporated herein by reference. Additional examples of suitable excitation sources are described in U.S. patent application Ser. No. 14/821,688, filed Aug. 7, 2015, titled "INTEGRATED DEVICE FOR PROBING, DETECTING AND ANALYZING MOLECULES," which is incorporated by reference in its entirety.
[0274] The photodetector(s) positioned with individual pixels of the integrated device may be configured and positioned to detect emission light from the pixel's corresponding sample well. Examples of suitable photodetectors are described in U.S. patent application Ser. No. 14/821,656, filed Aug. 7, 2015, titled "INTEGRATED DEVICE FOR TEMPORAL BINNING OF RECEIVED PHOTONS," which is incorporated by reference in its entirety. In some embodiments, a sample well and its respective photodetector(s) may be aligned along a common axis. In this manner, the photodetector(s) may overlap with the sample well within the pixel.
[0275] Characteristics of the detected emission light may provide an indication for identifying the marker associated with the emission light. Such characteristics may include any suitable type of characteristic, including an arrival time of photons detected by a photodetector, an amount of photons accumulated over time by a photodetector, and/or a distribution of photons across two or more photodetectors. In some embodiments, a photodetector may have a configuration that allows for the detection of one or more timing characteristics associated with a sample's emission light (e.g., luminescence lifetime). The photodetector may detect a distribution of photon arrival times after a pulse of excitation light propagates through the integrated device, and the distribution of arrival times may provide an indication of a timing characteristic of the sample's emission light (e.g., a proxy for luminescence lifetime). In some embodiments, the one or more photodetectors provide an indication of the probability of emission light emitted by the marker (e.g., luminescence intensity). In some embodiments, a plurality of photodetectors may be sized and arranged to capture a spatial distribution of the emission light. Output signals from the one or more photodetectors may then be used to distinguish a marker from among a plurality of markers, where the plurality of markers may be used to identify a sample within the sample. In some embodiments, a sample may be excited by multiple excitation energies, and emission light and/or timing characteristics of the emission light emitted by the sample in response to the multiple excitation energies may distinguish a marker from a plurality of markers.
[0276] In operation, parallel analyses of samples within the sample wells are carried out by exciting some or all of the samples within the wells using excitation light and detecting signals from sample emission with the photodetectors. Emission light from a sample may be detected by a corresponding photodetector and converted to at least one electrical signal. The electrical signals may be transmitted along conducting lines in the circuitry of the integrated device, which may be connected to an instrument interfaced with the integrated device. The electrical signals may be subsequently processed and/or analyzed. Processing or analyzing of electrical signals may occur on a suitable computing device either located on or off the instrument.
[0277] The instrument may include a user interface for controlling operation of the instrument and/or the integrated device. The user interface may be configured to allow a user to input information into the instrument, such as commands and/or settings used to control the functioning of the instrument. In some embodiments, the user interface may include buttons, switches, dials, and a microphone for voice commands. The user interface may allow a user to receive feedback on the performance of the instrument and/or integrated device, such as proper alignment and/or information obtained by readout signals from the photodetectors on the integrated device. In some embodiments, the user interface may provide feedback using a speaker to provide audible feedback. In some embodiments, the user interface may include indicator lights and/or a display screen for providing visual feedback to a user.
[0278] In some embodiments, the instrument may include a computer interface configured to connect with a computing device. The computer interface may be a USB interface, a FireWire interface, or any other suitable computer interface. A computing device may be any general purpose computer, such as a laptop or desktop computer. In some embodiments, a computing device may be a server (e.g., cloud-based server) accessible over a wireless network via a suitable computer interface. The computer interface may facilitate communication of information between the instrument and the computing device. Input information for controlling and/or configuring the instrument may be provided to the computing device and transmitted to the instrument via the computer interface. Output information generated by the instrument may be received by the computing device via the computer interface. Output information may include feedback about performance of the instrument, performance of the integrated device, and/or data generated from the readout signals of the photodetector.
[0279] In some embodiments, the instrument may include a processing device configured to analyze data received from one or more photodetectors of the integrated device and/or transmit control signals to the excitation source(s). In some embodiments, the processing device may comprise a general purpose processor, a specially-adapted processor (e.g., a central processing unit (CPU) such as one or more microprocessor or microcontroller cores, a field-programmable gate array (FPGA), an application-specific integrated circuit (ASIC), a custom integrated circuit, a digital signal processor (DSP), or a combination thereof). In some embodiments, the processing of data from one or more photodetectors may be performed by both a processing device of the instrument and an external computing device. In other embodiments, an external computing device may be omitted and processing of data from one or more photodetectors may be performed solely by a processing device of the integrated device.
[0280] According to some embodiments, the instrument that is configured to analyze samples based on luminescence emission characteristics may detect differences in luminescence lifetimes and/or intensities between different luminescent molecules, and/or differences between lifetimes and/or intensities of the same luminescent molecules in different environments. The inventors have recognized and appreciated that differences in luminescence emission lifetimes can be used to discern between the presence or absence of different luminescent molecules and/or to discern between different environments or conditions to which a luminescent molecule is subjected. In some cases, discerning luminescent molecules based on lifetime (rather than emission wavelength, for example) can simplify aspects of the system. As an example, wavelength-discriminating optics (such as wavelength filters, dedicated detectors for each wavelength, dedicated pulsed optical sources at different wavelengths, and/or diffractive optics) may be reduced in number or eliminated when discerning luminescent molecules based on lifetime. In some cases, a single pulsed optical source operating at a single characteristic wavelength may be used to excite different luminescent molecules that emit within a same wavelength region of the optical spectrum but have measurably different lifetimes. An analytic system that uses a single pulsed optical source, rather than multiple sources operating at different wavelengths, to excite and discern different luminescent molecules emitting in a same wavelength region can be less complex to operate and maintain, more compact, and may be manufactured at lower cost.
[0281] Although analytic systems based on luminescence lifetime analysis may have certain benefits, the amount of information obtained by an analytic system and/or detection accuracy may be increased by allowing for additional detection techniques. For example, some embodiments of the systems may additionally be configured to discern one or more properties of a sample based on luminescence wavelength and/or luminescence intensity. In some implementations, luminescence intensity may be used additionally or alternatively to distinguish between different luminescent labels. For example, some luminescent labels may emit at significantly different intensities or have a significant difference in their probabilities of excitation (e.g., at least a difference of about 35%) even though their decay rates may be similar. By referencing binned signals to measured excitation light, it may be possible to distinguish different luminescent labels based on intensity levels.
[0282] According to some embodiments, different luminescence lifetimes may be distinguished with a photodetector that is configured to time-bin luminescence emission events following excitation of a luminescent label. The time binning may occur during a single charge-accumulation cycle for the photodetector. A charge-accumulation cycle is an interval between read-out events during which photo-generated carriers are accumulated in bins of the time-binning photodetector. Examples of a time-binning photodetector are described in U.S. patent application Ser. No. 14/821,656, filed Aug. 7, 2015, titled "INTEGRATED DEVICE FOR TEMPORAL BINNING OF RECEIVED PHOTONS," which is incorporated herein by reference. In some embodiments, a time-binning photodetector may generate charge carriers in a photon absorption/carrier generation region and directly transfer charge carriers to a charge carrier storage bin in a charge carrier storage region. In such embodiments, the time-binning photodetector may not include a carrier travel/capture region. Such a time-binning photodetector may be referred to as a "direct binning pixel." Examples of time-binning photodetectors, including direct binning pixels, are described in U.S. patent application Ser. No. 15/852,571, filed Dec. 22, 2017, titled "INTEGRATED PHOTODETECTOR WITH DIRECT BINNING PIXEL," which is incorporated herein by reference.
[0283] In some embodiments, different numbers of fluorophores of the same type may be linked to different reagents in a sample, so that each reagent may be identified based on luminescence intensity. For example, two fluorophores may be linked to a first labeled affinity reagent and four or more fluorophores may be linked to a second labeled affinity reagent. Because of the different numbers of fluorophores, there may be different excitation and fluorophore emission probabilities associated with the different affinity reagents. For example, there may be more emission events for the second labeled affinity reagent during a signal accumulation interval, so that the apparent intensity of the bins is significantly higher than for the first labeled affinity reagent.
[0284] The inventors have recognized and appreciated that distinguishing nucleotides or any other biological or chemical samples based on fluorophore decay rates and/or fluorophore intensities may enable a simplification of the optical excitation and detection systems. For example, optical excitation may be performed with a single-wavelength source (e.g., a source producing one characteristic wavelength rather than multiple sources or a source operating at multiple different characteristic wavelengths). Additionally, wavelength discriminating optics and filters may not be needed in the detection system. Also, a single photodetector may be used for each sample well to detect emission from different fluorophores. The phrase "characteristic wavelength" or "wavelength" is used to refer to a central or predominant wavelength within a limited bandwidth of radiation (e.g., a central or peak wavelength within a 20 nm bandwidth output by a pulsed optical source). In some cases, "characteristic wavelength" or "wavelength" may be used to refer to a peak wavelength within a total bandwidth of radiation output by a source.
Computational Techniques
[0285] Aspects of the present application relate to computational techniques for analyzing the data generated by the polypeptide sequencing techniques described herein. As discussed above, for example in connection with FIGS. 1A and 1B, the data generated by using these sequencing techniques may include a series of signal pulses indicative of instances where an amino acid recognition molecule is associated with an amino acid exposed at the terminus of the polypeptide being sequenced. The series of signal pulses may have varying one or more features (e.g., pulse duration, interpulse duration, change in magnitude), depending on the type of amino acid presently at the terminus, over time as the degradation process proceeds in removing successive amino acids. The resulting signal trace may include characteristic patterns, which arise from the varying one or more features, associated with respective amino acids. The computational techniques described herein may be implemented as part of analyzing such data obtained using these sequencing techniques to identify an amino acid sequence.
[0286] Some embodiments may involve obtaining data during a degradation process of a polypeptide, analyzing the data to determine portions of the data corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process, and outputting an amino acid sequence representative of the polypeptide. FIG. 11 is a diagram of an illustrative processing pipeline 1100 for identifying an amino acid sequence by analyzing data obtained using the polypeptide sequencing techniques described herein. As shown in FIG. 11, analyzing sequencing data 1102 may involve using association event identification technique 1104 and amino acid identification technique 1106 to output amino acid sequence(s) 1108.
[0287] As discussed herein, sequencing data 1102 may be obtained during a degradation process of a polypeptide. In some embodiments, the sequencing data 1102 is indicative of amino acid identity at the terminus of the polypeptide during the degradation process. In some embodiments, the sequencing data 1102 is indicative of a signal produced by one or more amino acid recognition molecules binding to different types of terminal amino acids at the terminus during the degradation process. Exemplary sequencing data is shown in FIGS. 1A and 1B, which are discussed above.
[0288] Depending on how signals are generated during the degradation process, sequencing data 1102 may be indicative of one or more different types of signals. In some embodiments, sequencing data 1102 is indicative of a luminescent signal generated during the degradation process. For example, a luminescent label may be used to label an amino acid recognition molecule, and luminescence emitted by the luminescent label may be detected as the amino acid recognition molecule associates with a particular amino acid, resulting in a luminescent signal. In some embodiments, sequencing data 1102 is indicative of an electrical signal generated during the degradation process. For example, a polypeptide molecule being sequenced may be immobilized to a nanopore, and an electrical signal (e.g., changes in conductance) may be detected as an amino acid recognition molecule associates with a particular amino acid.
[0289] Some embodiments involve analyzing sequencing data 1102 to determine portions of sequencing data 1102 corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process. As shown in FIG. 11, association event identification technique 1104 may access sequencing data 1102 and analyze sequencing data to identify portions of sequencing data 1102 that correspond to association events. The association events may correspond to characteristic patterns, such as CP.sub.1 and CP.sub.2 shown in FIG. 1B, in the data. In some embodiments, association event identification technique 1104 may involve detecting a series of cleavage events and determining portions of sequencing data 1102 between successive cleavage events. As an example, a cleavage event between CP.sub.1 and CP.sub.2 shown in FIG. 1B may be detected such that a first portion of the data corresponding to CP.sub.1 may be identified as a first association event and a second portion of the data corresponding CP.sub.2 may be identified as a second association event.
[0290] Some embodiments involve identifying a type of amino acid for one or more of the determined portions of sequencing data 1102. As shown in FIG. 11, amino acid identification technique 1106 may be used to determine a type of amino acid for one or more of the association events identified by association event identification technique 1104. In some embodiments, the individual portions of data identified by association event identification technique 1104 may include a pulse pattern, and amino acid identification technique 1106 may determine a type of amino acid for one or more of the portions based on its respective pulse pattern. Referring to FIG. 1B, amino acid identification technique 1106 may identify a first type of amino acid for CP.sub.1 and a second type of amino acid for CP.sub.2. In some embodiments, determining the type of amino acid may include identifying an amount of time within a portion of data, such as a portion identified using association event identification technique 1104, when the data is above a threshold value and comparing the amount of time to a duration of time for the portion of data. For example, identifying a type of amino acid for CP.sub.1 may include determining an amount of time within CP.sub.1 where the signal is above a threshold value, such as time period, pd, where the signal is above M.sub.L, and comparing it to a total duration of time for CP.sub.1. In some embodiments, determining the type of amino acid may involve identifying one or more pulse durations for one or more portions of data identified by association event identification technique 1102. For example, identifying a type of amino acid for CP.sub.1 may include determining a pulse duration for CP.sub.1, such as time period, pd. In some embodiments, determining the type of amino acid may involve identifying one or more interpulse durations for one or more portions of the data identified using association event identification technique 1104. For example, identifying a type of amino acid for CP.sub.1 may include identifying an interpulse duration, such as ipd.
[0291] By identifying a type of amino acid for successive portions of sequencing data 1102, amino acid identification technique 1106 may output amino acid sequence(s) 1108 representative of the polypeptide. In some embodiments, the amino acid sequence includes a series of amino acids corresponding to the portions of data identified using association event identification technique 1104.
[0292] FIG. 12 is a flow chart of an illustrative process 1200 for determining an amino acid sequence of a polypeptide molecule, in accordance with some embodiments of the technology described herein. Process 1200 may be performed on any suitable computing device(s) (e.g., a single computing device, multiple computing devices co-located in a single physical location or located in multiple physical locations remote from one another, one or more computing devices part of a cloud computing system, etc.), as aspects of the technology described herein are not limited in this respect. In some embodiments, association event identification technique 1104 and amino acid identification technique 1106 may perform some or all of process 1200 to determine amino acid sequence(s).
[0293] Process 1200 begins at act 1202, which involves contacting a single polypeptide molecule with one or more terminal amino acid recognition molecules. Next, process 1200 proceeds to act 1104, which involves detecting a series of signal pulses indicative of association of the one or more terminal amino acid recognition molecules with successive amino acids exposed at a terminus of the single polypeptide while the single polypeptide is being degraded. The series of pulses may allow for sequencing of the single polypeptide molecule, such as by using association event identification technique 1104 and amino acid identification technique 1106.
[0294] In some embodiments, process 1200 may include act 1206, which involves identifying a first type of amino acid in the single polypeptide molecule based on a first characteristic pattern in the series of signal pulses, such as by using amino acid identification technique 1106.
[0295] FIG. 13 is a flow chart of an illustrative process 1300 for determining an amino acid sequence representative of a polypeptide, in accordance with some embodiments of the technology described herein. Process 1300 may be performed on any suitable computing device(s) (e.g., a single computing device, multiple computing devices co-located in a single physical location or located in multiple physical locations remote from one another, one or more computing devices part of a cloud computing system, etc.), as aspects of the technology described herein are not limited in this respect. In some embodiments, association event identification technique 1104 and amino acid identification technique 1106 may perform some or all of process 1300 to determine amino acid sequence(s).
[0296] Process 1300 begins at act 1302, where data during a degradation process of a polypeptide is obtained. In some embodiments, the data is indicative of amino acid identity at the terminus of the polypeptide during the degradation process. In some embodiments, the data is indicative of a signal produced by one or more amino acid recognition molecules binding to different types of terminal amino acids at the terminus during the degradation process. In some embodiments, the data is indicative of a luminescent signal generated during the degradation process. In some embodiments, the data is indicative of an electrical signal generated during the degradation process.
[0297] Next, process 1300 proceeds to act 1304, where the data is analyzed to determine portions of the data corresponding to amino acids that are sequentially exposed at a terminus of the polypeptide during the degradation process, such as by using association event identification technique 1104 and amino acid identification technique 1106. In some embodiments, analyzing the data further comprises detecting a series of cleavage events and determining the portions of the data between successive cleavage events, such as by using association event identification technique 1104.
[0298] In some embodiments, analyzing the data further comprises determining a type of amino acid for each of the individual portions, such as by using amino acid identification technique 1106. In some embodiments, each of the individual portions comprises a pulse pattern, and analyzing the data further comprises determining a type of amino acid for one or more of the portions based on its respective pulse pattern. In some embodiments, determining the type of amino acid further comprises identifying an amount of time within a portion when the data is above a threshold value and comparing the amount of time to a duration of time for the portion. In some embodiments, determining the type of amino acid further comprises identifying at least one pulse duration for each of the one or more portions. In some embodiments, determining the type of amino acid further comprises identifying at least one interpulse duration for each of the one or more portions.
[0299] Next, process 1300 proceeds to act 1306, where an amino acid sequence representative of the polypeptide is outputted, such as via a user interface. In some embodiments, the amino acid sequence includes a series of amino acids corresponding to the portions.
[0300] An illustrative implementation of a computer system 1400 that may be used in connection with any of the embodiments of the technology described herein is shown in FIG. 14. The computer system 1400 includes one or more processors 1410 and one or more articles of manufacture that comprise non-transitory computer-readable storage media (e.g., memory 1420 and one or more non-volatile storage media 1430). The processor 1410 may control writing data to and reading data from the memory 1420 and the non-volatile storage device 1430 in any suitable manner, as the aspects of the technology described herein are not limited in this respect. To perform any of the functionality described herein, the processor 1410 may execute one or more processor-executable instructions stored in one or more non-transitory computer-readable storage media (e.g., the memory 1420), which may serve as non-transitory computer-readable storage media storing processor-executable instructions for execution by the processor 1410.
[0301] Computing device 1400 may also include a network input/output (I/O) interface 1440 via which the computing device may communicate with other computing devices (e.g., over a network), and may also include one or more user I/O interfaces 1450, via which the computing device may provide output to and receive input from a user. The user I/O interfaces may include devices such as a keyboard, a mouse, a microphone, a display device (e.g., a monitor or touch screen), speakers, a camera, and/or various other types of I/O devices.
[0302] The above-described embodiments can be implemented in any of numerous ways. For example, the embodiments may be implemented using hardware, software or a combination thereof. When implemented in software, the software code can be executed on any suitable processor (e.g., a microprocessor) or collection of processors, whether provided in a single computing device or distributed among multiple computing devices. It should be appreciated that any component or collection of components that perform the functions described above can be generically considered as one or more controllers that control the above-discussed functions. The one or more controllers can be implemented in numerous ways, such as with dedicated hardware, or with general purpose hardware (e.g., one or more processors) that is programmed using microcode or software to perform the functions recited above.
[0303] In this respect, it should be appreciated that one implementation of the embodiments described herein comprises at least one computer-readable storage medium (e.g., RAM, ROM, EEPROM, flash memory or other memory technology, CD-ROM, digital versatile disks (DVD) or other optical disk storage, magnetic cassettes, magnetic tape, magnetic disk storage or other magnetic storage devices, or other tangible, non-transitory computer-readable storage medium) encoded with a computer program (i.e., a plurality of executable instructions) that, when executed on one or more processors, performs the above-discussed functions of one or more embodiments. The computer-readable medium may be transportable such that the program stored thereon can be loaded onto any computing device to implement aspects of the techniques discussed herein. In addition, it should be appreciated that the reference to a computer program which, when executed, performs any of the above-discussed functions, is not limited to an application program running on a host computer. Rather, the terms computer program and software are used herein in a generic sense to reference any type of computer code (e.g., application software, firmware, microcode, or any other form of computer instruction) that can be employed to program one or more processors to implement aspects of the techniques discussed herein.
EXAMPLES
Example 1. Edman Degradation by Chemical Cleavage
##STR00012##
[0305] A surface-attached oligopeptide of approximately 3 to approximately 30 amino acids (n=3-30) is provided, where amino acid residues R.sub.1-R.sub.3 can be any of the common 20 amino acids or an endogenously modified amino acid (e.g., modified by a post-translational modification). In the isothiocyanate N-terminal reaction, Step 1, an isothiocyanate X-NCS is added to a vessel containing the surface-attached oligopeptide, where X is phenyl (Ph), 4-NO.sub.2Ph, 4-SO.sub.3Ph, napthyl, benzyl, alkyl, or a derivative thereof. Step 1 is carried out under the following Conditions to afford the X-NCS derivatized N-terminal amino acid: aqueous buffer pH 4-10, MeOH or EtOH or IPA alcoholic co-solvents, trialkylamines in organic solvents (DCM, THF, MeCN, DMF, and the like), 20.degree. C. to 50.degree. C. In the thiourea cleavage reaction, Step 2, an Acid or a Base is added to a vessel containing the X-NCS derivatized N-terminal amino acid, where the Acid is acetic acid, formic acid, trichloroacetic acid, trifluoroacetic acid, phosphoric acid, or hydrochloric acid, as neat or aqueous solutions, or where the Base is a trialkylamine or a buffered trialkylamine (e.g., Et.sub.3NH.sup.+AcO.sup.-). Step 2 is carried out under the following Conditions to afford the n-1 oligopeptide and thiohydantoin byproduct: neat acid or with aqueous/organic co-solvents of any ratio, 20.degree. C. to 50.degree. C.
Example 2. Solubilizing Linkers for Peptide Surface Immobilization
[0306] Seeking to improve oligopeptide solubility in aqueous buffer, it was determined that peptide fragments could be conjugated with oligonucleotide linkers to both improve aqueous solubility and provide a functional moiety for surface immobilization of peptides at the single molecule level. Different peptide-linker conjugates were synthesized, with example structures depicted in FIG. 15A for a peptide-DNA conjugate and a peptide-PEG conjugate. Linker conjugation was observed to greatly enhance peptide solubility in aqueous solution for each of the different peptide-linker conjugates evaluated.
[0307] The peptide-linker conjugates were evaluated for amino acid cleavage at peptide N-termini by N-terminal aminopeptidases (Table 6, below).
TABLE-US-00006 TABLE 6 Terminal amino acid cleavage of peptide-linker conjugates. SEQ ID Cleaved by Rat Cleaved by Entry Peptide NO. Class Linker APN PIP 1 KF 70 positive oligo No 2 KKMKKM{LYS(N3)} 71 positive oligo No 3 KKMKKM{LYS(N3)} 71 positive oligo-PEG No 4 KKMKKM{LYS(N3)} 71 positive PEG4 Yes 5 DDMDDMILYS(N3)} 72 negative oligo Yes 6 FFMFFM{LYS(N3)} 73 aromatic oligo Yes 7 AAMAAM{LYS(N3)} 74 hydrophobic oligo Yes 8 FPFPFP{LYS(N3)} 75 aromatic oligo Yes 9 DPDPDP{LYS(N3)} 76 negative oligo Yes 10 KPKPKP{LYS(N3)} 77 positive oligo No 11 KPKPKP{LYS(N3)} 77 positive PEG4 Yes
[0308] The peptide-linker conjugates shown in Table 6 were incubated with either proline iminopeptidase ("PIP") or rat aminopeptidase N ("Rat APN"), and peptide cleavage was monitored by LCMS. An example of an LCMS demonstrating cleavage of Entry 5 from Table 6 is shown in FIG. 15B. All other cleavage reactions were measured in a similar manner. As shown in Table 6, while positively charged peptide-DNA conjugates ("oligo" and "oligo-PEG" linkers) were not cleaved by the aminopeptidases tested, all other conjugate classes (negatively charged, aromatic, hydrophobic) with DNA oligonucleotide linkers were cleaved. By comparison, the positively charged peptide-PEG conjugates were shown to be cleaved by at least one of the aminopeptidases.
[0309] Using labeled peptide-linker conjugates, it was shown that peptides of different compositions could be immobilized to individual sample well surfaces for single molecule analysis. For these experiments, the DNA linker was labeled with a dye (e.g., as depicted in FIG. 15A for the peptide-DNA conjugate), and loading of different peptide-DNA conjugates into individual sample wells was measured by dye fluorescence. An example loading experiment is shown in FIG. 15C. By measuring fluorescence emission of a labeled peptide-DNA conjugate (50 pM), it was determined that at least 18% of sample wells on a chip were loaded at single occupancy per sample well with a surface-immobilized conjugate. These experiments demonstrated that peptide-linker conjugates display enhanced aqueous solubility compared to non-conjugated peptide counterpart, that conjugated linkers do not prevent terminal amino acid cleavage of peptides by different aminopeptidases, and that peptide-linker conjugates of different compositions can be immobilized to chip surfaces at the single molecule level.
Example 3. Exopeptidase Cleavage of Polypeptide Substrates
[0310] The cleavage capabilities of various aminopeptidases were tested. The conditions and results for a set of cleavage assay experiments are shown in Table 7, including concentration of peptide substrate, concentration of enzyme, buffer conditions, temperature, and incubation time. Cleavage of peptide substrates by the indicated enzymes was assayed using High Performance Liquid Chromatography (HPLC). The "HPLC assay cony" value in Table 7 indicates the percentage of the peptide substrate that was converted to cleavage product. To determine the "HPLC assay cony" value, two solutions were prepared containing the same starting concentration of peptide. One solution was subjected to enzymatic digestion, while the other solution did not contain any enzyme, but was diluted with an equivalent amount of buffer used to store the enzyme. The reactions were quenched at the time indicated. The amount of reactant converted to product was determined by dividing the area of the peak obtained by HPLC of the starting material remaining after enzymatic digestion by the peak area of the control solution of undigested peptide, and then multiplying this ratio by 100. In Table 7, "NH2" indicates an amine group, "yPIP" refers to Y. pestis proline iminopeptidase, "NPEPPS" refers to puromycin-sensitive aminopeptidase, "VPr" refers to Vibrio proteolyticus aminopeptidase, and "EDAPN" refers to L. pneumophila M1 aminopeptidase.
TABLE-US-00007 TABLE 7 Cleavage of peptide substrates by aminopeptidases. HPLC Assay Enzyme Peptide Substrate Conditions Temp/Time Conv Pdt yPIP GlyProArgPro 5 mM peptide, 50 nM 30.degree. C./1 hr 100% ProArgPro (SEQ ID NO: 84) enzyme, 10 mM MgCl.sub.2, 10 mM Tris, 0.02% Tween-20 pH 8.0 yPIP DDPDDP{LYSN3}NH2 1 mM peptide, 700 nM 30.degree. C./6 hrs 0% n/a (SEQ ID NO: 85) enzyme, 10 mM MgCl.sub.2, 10 mM Tris, 0.02% Tween-20 pH 8.0 yPIP AAMAAM{LYSN3 1 mM peptide, 700 nM 30.degree. C./6 hrs 0% n/a H2 (SEQ ID NO: 74) enzyme, 10 mM MgCl.sub.2, 10 mM Tris, 0.02% Tween-20 pH 8.0 yPIP YPYPYP{LYSN3}NH2 600 mM peptide, 7 mM 30.degree. C./1 hr 100% PYPYP{LYSN3} (SEQ ID NO: 86) enzyme, 10 mM MgCl.sub.2, 10 NH2 mM Tris, 0.02% Tween-20 (SEQ ID NO: 87) pH 8.0 yPIP FPFPFP{LYSN3}NH2 600 mM peptide, 7 mM 30.degree. C./1 hr 100% PFPFP{LYSN3}N (SEQ ID NO: 75) enzyme, 10 mM MgCl.sub.2, 10 H2 mM Tris, 0.02% Tween-20 (SEQ ID NO: 88) pH 8.0 NPEPPS LeuTyr 5 mM 700 nM enzyme, 25 mM 37.degree. C./1 hr 5% Tyr HEPES, 1 mM Mg(OAc).sub.2, 1 mM DTT, pH7.5 (Cy3B)n- FPFPFP{LYSN3}NH2 1 mM peptide, 14 mM 30.degree. C./15 100% PFPFP{LYSN3}N yPIP (SEQ ID NO: 75) enzyme, 10 mM MgCl.sub.2, 10 min H2 mM Tris, 0.02% Tween-20 (SEQ ID NO: 88) pH 8.0 (Cy3B)n- FPFPFP{LYSN3}NH2 1 mM peptide, 14 mM 30.degree. C./15 100% PFPFP{LYSN3}N yPIP (SEQ ID NO: 75) enzyme, 200 mM Bis-tris min H2 Propane, 30 mM KOAc, 25 (SEQ ID NO: 88) mM Mg(OAc).sub.2, 32 mM 3,4 Dihydroxy-benzoic acid and 12 mM Nitrobenzoic acid P. MetTyr 1 mM peptide, 1 mM 30.degree. C./1 hr 7% Tyr falciparum enzyme, 2.5 mM ZnCl.sub.2 25 M1 mM Tris pH 8.0 (Atto647 FPFPFP{LYSN3}NH2 1 mM peptide, 14 mM 30.degree. C./1 hr 100% PFPFP{LYSN3}N N)n-yPIP (SEQ ID NO: 75) enzyme, 10 mM MgCl.sub.2, 10 H2 mM Tris, 0.02% Tween-20 (SEQ ID NO: 88) pH 8.0 Rat APN KKMKKMLys-Triazole- 1 mM peptide, 150 nM 30.degree. C./1 hr >90% PEG4 Biotin enzyme, 2.5 mM ZnCl.sub.2 25 (SEQ ID NO: 89) mM Tris pH 8.0 yPIP FPFPFP{LYSN3}NH2 1 mM peptide, 7 mM 30.degree. C./1 hr 100% PFPFP{LYSN3}N (SEQ ID NO: 75) enzyme, 10 mM MgCl.sub.2, 10 H2 mM Tris, 0.02% Tween-20 (SEQ ID NO: 88) pH 8.0 yPIP BCN PEG23 Biotin 1 mM peptide, 7 mM 30.degree. C./1 hr 100% PFPFP{LYS}BCN conjugate enzyme, 10 mM MgCl.sub.2, 10 etc mM Tris, 0.02% Tween-20 (SEQ ID NO: 88) pH 8.0 P. DDMDDM{LYSN3}N 1 mM peptide, 1 mM 70.degree. C./1 hr 60% horikoshii H2 (SEQ ID NO: 72) enzyme, 2.5 mM ZnCl.sub.2, 25 Tet mM Tris pH 8.0 GluAsp- DDMDDM{LYSN3}N 1 mM peptide, 1 mM 30.degree. C./1 hr 100% APN H2 (SEQ ID NO: 72) enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 P. AAPAAP{LYSN3}NH2 1 mM peptide, 1 mM 70.degree. C./1 hr 100% APAAP{LYSN3} horikoshii (SEQ ID NO: 90) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 Tet mM Tris pH 8.0 (SEQ ID NO: 91) P. YYPYYP{LYSN3}NH2 1 mM peptide, 1 mM 70.degree. C./1 hr 100% YPYYP{LYSN3} horikoshii (SEQ ID NO: 92) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 Tet mM Tris pH 8.0 (SEQ ID NO: 93) P. FFPFFP{LYSN3}NH2 1 mM peptide, 1 mM 70.degree. C./1 hr 100% FPFFP{LYSN3}N horikoshii (SEQ ID NO: 94) enzyme, 2.5 mM ZnCl.sub.2, 25 H2 Tet mM Tris pH 8.0 (SEQ ID NO: 95) Rat APN RRPRRP{LYSN3}NH2 1 mM peptide, 50 nM 30.degree. C./1 hr 55% RPRRP{LYSN3} (SEQ ID NO: 96) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 97) Rat APN AAPAAP{LYSN3}NH2 1 mM peptide, 50 nM 30.degree. C./1 hr 100% APAAP{LYSN3} (SEQ ID NO: 98) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 99) Rat APN KKPKKP{LYSN3}NH2 1 mM peptide, 50 nM 30.degree. C./1 hr 85% KPKKP{LYSN3} (SEQ ID NO: 100) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 101) Rat APN KKMKKM{LYSN3}N 1 mM peptide, 50 nM 30.degree. C./1 hr 50% KMKKM{LYSN3 H2 (SEQ ID NO: 71) enzyme, 2.5 mM ZnCl.sub.2, 25 }NH2 mM Tris pH 8.0 (SEQ ID NO: 102) VPr RRPRRP{LYSN3}NH2 1 mM peptide, 2 mM 30.degree. C./1 hr 100% (SEQ ID NO: 96) enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 VPr AAPAAP{LYSN3}NH2 1 mM peptide, 2 mM 30.degree. C./1 hr 100% APAAP{LYSN3} (SEQ ID NO: 98) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 99) VPr KKPKKP{LYSN3}NH2 1 mM peptide, 2 mM 30.degree. C./1 hr 100% KPKKP{LYSN3} (SEQ ID NO: 100) enzyme, 2.5 mM ZnCl.sub.2 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 101) VPr YYPYYP{LYSN3}NH2 1 mM peptide, 2 mM 30.degree. C./1 hr 50% YPYYP{LYSN3} (SEQ ID NO: 92) enzyme, 2.5 mM ZnCl.sub.2, 25 NH2 mM Tris pH 8.0 (SEQ ID NO: 93) VPr FFPFFP{LYSN3}NH2 1 mM peptide, 2 mM 30.degree. C./1 hr 100% FPFFP{LYSN3}N (SEQ ID NO: 94) enzyme, 2.5 mM ZnCl.sub.2, 25 H2 mM Tris pH 8.0 (SEQ ID NO: 95) VPr AAMAAM{LYSN3}N 1 mM peptide, 2 mM 30.degree. C./1 hr 100% multiple pdts H2 (SEQ ID NO: 74) enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 VPr KKMKKM{LYSN3}N 1 mM peptide, 2 mM 30.degree. C./1 hr 100% multiple pdts H2 (SEQ ID NO: 89) enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 VPr YYMYYM{LYSN3}N 1 mM peptide, 2 mM 30.degree. C./1 hr >90% multiple pdts H2 (SEQ ID NO: 103) enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 yPIP Attor6g-1kPEG- 44 mM Peptide, 7 mM 30.degree. C./0.5 <10% PAAAFK-1kPEG- DPAAAFK{LysN3}- enzyme, 200 mM Bis-tris hr Biotin 1kPEG-Biotin Propane, 30 mM KOAc, 25 (SEQ ID NO: 105) (SEQ ID NO: 104) mM Mg(OAc).sub.2, 32 mM 3,4 Dihydroxy-benzoic acid and 12 mM Nitrobenzoic acid + PCD + TXV PfuPIP FPFPFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M enzyme, 80.degree. C./0.5 100% PFPFP{LYSN3}N (SEQ ID NO: 75) 1 mM CoCl.sub.2, 50 mM hr H2 HEPES, 50 mM KC1 (SEQ ID NO: 88) PfuPIP Attor6g-1kPEG- 1 mM Peptide, 1 .mu.M enzyme, 80.degree. C./0.5 40% PAAAFK-1kPEG- DPAAAFK{LysN3}- 1 mM CoCl.sub.2, 50 mM hr Biotin 1kPEG-Biotin HEPES, 50 mM KC1 (SEQ ID NO: 105) (SEQ ID NO: 104) yPIP Attor6g-1kPEG-ODN- 20 .mu.M Q24 conjugate, 30 .mu.M 30.degree. C./0.3 100% PAAAFK-1kPEG- DD60-Biotin ypip, 1x Mg buffer hr Biotin (SEQ ID NO: 105) yPIP FPFPFP{LYSN3}NH2 1 mM Peptide, 7 .mu.M 37.degree. C./0.5 100% PFPFP{LYSN3}N (SEQ ID NO: 75) Enzyme, 50 mM MOPS, 10 hr H2 mM Mg(OAc).sub.2 pH 8.0 (SEQ ID NO: 88) yPIP Attor6g-1kPEG-ODN- 10 .mu.M Peptide, 7 .mu.M 37.degree. C./0.5 100% PAAAFK-1kPEG- DD60-Biotin Enzyme, 50 mM MOPS, 10 hr Biotin mM Mg(OAc).sub.2 pH 8.0 (SEQ ID NO: 105) PfuPIP FPFPFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 80.degree. C./0.5 40% PFPFP{LYSN3}N (SEQ ID NO: 75) Enzyme, 50 mM MOPS, 10 hr H2 mM Mg(OAc).sub.2 pH 8.0 (SEQ ID NO: 88) yPIP- FPFPFP{LYSN3}NH2 1 mM Peptide, 2.1 .mu.M 37.degree. C./0.5 100% PFPFP{LYSN3}N Q24- (SEQ ID NO: 75) Enzyme, 50 mM MOPS, 10 hr H2 Cy3B mM Mg(OAc).sub.2 pH 8.0 (SEQ ID NO: 88) yPIP- YPYPYP{LYSN3}NH2 1 mM Peptide, 100 nM 37.degree. C./0.5 100% YPYYP{LYSN3} Q24- (SEQ ID NO: 86) Enzyme, 1X CB2 hr NH2 Rho6G (SEQ ID NO: 93) yPIP- YPYPYP{LYSN3}NH2 1 mM Peptide, ~5 .mu.M 37.degree. C./0.5 <15% YPYYP{LYSN3} Q24Dark (SEQ ID NO: 86) Enzyme, 1X CB2 hr NH2 (SEQ ID NO: 93) Rat APN QP5-Atto649N 37 .mu.M Peptide, 100 nM 37.degree. C./0.5 >95% (KAAAAAAFK{LYSN Enzyme, 2.5 mM ZnCl.sub.2, 25 hr. 3}NH2) mM Tris pH 8.0 (SEQ ID NO: 106) VPr QP5-Atto649N 37 .mu.M Peptide, 8 .mu.M 37.degree. C./0.5 100% (KAAAAAAFK{LYSN Enzyme, 2.5 mM ZnCl.sub.2, 25 hr 3}NH2) mM Tris pH 8.0 (SEQ ID NO: 106) K287pA YPYPYP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 100% PYPYPK zF-Cy3 (SEQ ID NO: 86) Enzyme, 50 mM MOPS, 10 (SEQ ID NO: 83) yPIP mM Mg(OAc).sub.2 pH 8.0 V. AAPAAP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr ~5% cholera (SEQ ID NO: 98) Enzyme, 2.5 mM ZnCl.sub.2, 25 APT mM Tris pH 8.0 Bst M28 AAPAAP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 100% (SEQ ID NO: 98) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 Taq APT AAPAAP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr >90% (SEQ ID NO: 98) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 V. YYPYYP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 5% cholera (SEQ ID NO: 92) Enzyme, 2.5 mM ZnCl.sub.2, 25 APT mM Tris pH 8.0 Bst M28 YYPYYP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 10% (SEQ ID NO: 92) Enzyme, 2.5 mM ZnCl.sub.2, 25
mM Tris pH 8.0 Taq APT YYPYYP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 30% (SEQ ID NO: 92) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 V. FFPFFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr >95% cholera (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 APT mM Tris pH 8.0 Bst M28 FFPFFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 30% (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 Taq APT FFPFFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 60% (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 V. YYMYYM{LYSN3}N 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 30% cholera H2 (SEQ ID NO: 103) Enzyme, 2.5 mM ZnCl.sub.2, 25 APT mM Tris pH 8.0 Bst M28 YYMYYM{LYSN3}N 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr >50% multiple (N-1, H2 (SEQ ID NO: 103) Enzyme, 2.5 mM ZnCl.sub.2, 25 -2, -3, etc.) mM Tris pH 8.0 Taq APT YYMYYM{LYSN3}N 1 mM Peptide, 1 .mu.M 37.degree. C./1 hr 85% multiple (N-1, H2 (SEQ ID NO: 103) Enzyme, 2.5 mM ZnCl.sub.2, 25 -2, -3, etc.) mM Tris pH 8.0 Cy3B- YPYPYP{LYSN3}NH2 1 mM Peptide, 7 .mu.M 37.degree. C./0.5 hr 100% Q24- (SEQ ID NO: 86) Enzyme, 10 mM MgCl.sub.2, 50 pAzF- mM MOPS yPIP Cy3B- FFPFFP{LYSN3}NH2 1 mM Peptide, 10 .mu.M 37.degree. C./0.5 hr 50% BstTET (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 Cy3B- FFPFFP{LYSN3}NH2 1 mM Peptide, 20 .mu.M 37.degree. C./0.5 hr 100% taqAPT (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 2nd peak mM Tris pH 8.0 Cy3B- FFPFFP{LYSN3}NH2 1 mM Peptide, 20 .mu.M 37.degree. C./0.5 hr 100% taqAPT (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 4th peak mM Tris pH 8.0 PhaloM2 RRPRRP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr 30% multiple, even 8 (SEQ ID NO: 96) Enzyme, 2.5 mM ZnCl.sub.2, 25 higher mass mM Tris pH 8.0 yPAP RRPRRP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr 100% (SEQ ID NO: 96) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 yPAP AAPAAP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr 100% (SEQ ID NO: 98) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 yPAP KKPKKP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr 100% (SEQ ID NO: 100) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 yPAP YYPYYP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr 100% (SEQ ID NO: 92) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 PhaioM2 FFPFFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr >80% 8 (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 yPAP FFPFFP{LYSN3}NH2 1 mM Peptide, 1 .mu.M 37.degree. C./0.5 hr >80% (SEQ ID NO: 94) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 V. QP5-Atto649N 1 mM Peptide, 5 .mu.M 37.degree. C./0.5 hr 100% cholera (KAAAAAAFK{LYSN Enzyme, 2.5 mM ZnCl.sub.2, 25 APT 3}NH2) mM Tris pH 8.0 (SEQ ID NO: 106) yPAP YYPYYP{LYSN3}NH2 1 mM Peptide, 2 .mu.M 37.degree. C./0.5 hr 10% (SEQ ID NO: 92) Enzyme, 2.5 mM ZnCl.sub.2, 25 mM Tris pH 8.0 V. RRPRRP{LYSN3}NH2 1 mM peptide, 2 .mu.M enzyme, 30.degree. C./1 hr >90% anguillarum (SEQ ID NO: 96) 2.5 mM ZnCl.sub.2, 25 mM Tris APN pH 8.0 V. AAPAAP{LYSN3}NH2 1 mM peptide, 2 .mu.M enzyme, 30.degree. C./1 hr 50% anguillarum (SEQ ID NO: 98) 2.5 mM ZnCl.sub.2, 25 mM Tris APN pH 8.0 V. KKPKKP{LYSN3}NH2 1 mM peptide, 2 .mu.M enzyme, 30.degree. C./1 hr <5% anguillarum (SEQ ID NO: 100) 2.5 mM ZnCl.sub.2, 25 mM Tris APN pH 8.0 VPr FYPLPWPDDDY{LYS 300 .mu.M Peptide, 4 .mu.M 37.degree. C./0.5 100% N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 hr (SEQ ID NO: 107) mM MOPS pH 8.0 yPIP YPLPWPDDDY{LYSN 300 .mu.M Peptide, 7 .mu.M 37.degree. C./0.5 100% 3}NH2 enzyme, 10 mM MgCl.sub.2, 50 hr (SEQ ID NO: 108) mM MOPS pH 8.0 VPr PLPWPDDDY{LYSN3 300 .mu.M Peptide, 4 .mu.M 37.degree. C./0.5 100% }NH2 enzyme, 10 mM MgCl.sub.2, 50 hr (SEQ ID NO: 109) mM MOPS pH 8.0 yPIP LPWPDDDY{LYSN3} 300 .mu.M Peptide, 7 .mu.M 37.degree. C./0.5 100% NH2 (SEQ ID NO: 110) enzyme, 10 mM MgCl.sub.2, 50 hr mM MOPS pH 8.0 hTET FYPLPWPDDDY{LYS 200 .mu.M Peptide, 2 .mu.M 37.degree. C./1 hr 55% YPLPWPDDDY{ N3}NH2 enzyme, 2.5 mM ZnCl.sub.2, 25 LYSN3}NH2 (SEQ ID NO: 107) mM Tris pH 8.0 (SEQ ID NO: 108) hTET FYPLPWPDDDY{LYS 200 .mu.M Peptide, 2 .mu.M 37.degree. C./1 hr 55% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) Pro VPrA FYPLPWPDDDY{LYS 200 .mu.M Peptide, 2.2 .mu.M 37.degree. C./1 hr 6% YPLPWPDDDY{ mbr N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) ThrCut- FYPLPWPDDDY{LYS 200 .mu.M Peptide, 2.1 .mu.M 37.degree. C./1 hr 40% YPLPWPDDDY{ ProVPrA N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 mbr (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 200 .mu.M Peptide, 4 .mu.M 37.degree. C./0.5 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 hr LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) ProVPrA FYPLPWPDDDY{LYS 200 .mu.M Peptide, 4 .mu.M 37.degree. C./0.5 50% YPLPWPDDDY{ mbr N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 hr LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) ThrCut- FYPLPWPDDDY{LYS 200 .mu.M Peptide, 4 .mu.M 37.degree. C./0.5 40% YPLPWPDDDY{ ProVPrA N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 hr LYSN3}NH2 mbr (SEQ ID NO: 107) mM MOPS pH 8.0 (SEQ ID NO: 108) VPr FWPLPWPDDDY{LYS 200 .mu.M Peptide, 4 .mu.M 37.degree. C./1 hr 100% N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 (SEQ ID NO: 107) mM MOPS pH 8.0 VPr FWFPPWPDDDY{LYS 200 .mu.M Peptide, 4 .mu.M 37.degree. C./1 hr 96% pAzF N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 (SEQ ID NO: 107) mM MOPS pH 8.0 ThrCut- FWPLPWPDDDY{LYS 200 .mu.M Peptide, 6.4 .mu.M 37.degree. C./1 hr 100% Pro VPrA N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 mbr Cy3 (SEQ ID NO: 107) mM MOPS pH 8.0 clicked hTET PLPWPDDDY{LYSN3 200 .mu.M Peptide, 4 .mu.M 37.degree. C./1 hr 100% }NH2 enzyme, 10 mM MgCl.sub.2, 50 (SEQ ID NO: 109) mM MOPS pH 8.0 VPr FWPLPWPDDDY{LYS 400 .mu.M Peptide, 8 .mu.M 37.degree. C./1 hr 100% N3}NH2 enzyme, 50 mM HEPES pH (SEQ ID NO: 107) 8.0, 300 mM NaCl, 1 mM DTT, 5% Glycerol, 32 mM PCA VPr FYPLPWPDDDY{LYS 200 .mu.M Peptide, 8 .mu.M RT/1 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 5.0 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 200 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 5.0 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 200 .mu.M Peptide, 8 .mu.M RT/1 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 5.0 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 200 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM MgCl.sub.2, 50 LYSN3}NH2 (SEQ ID NO: 107) mM MOPS pH 5.0 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 600 .mu.M Peptide, 0.8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH5.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 600 .mu.M Peptide, 0.8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH6.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 600 .mu.M Peptide, 0.8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH7.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 600 .mu.M Peptide, 0.8 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH8.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.08 .mu.M RT/0.5 hr 50% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH5.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.08 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH6.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.08 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH7.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.08 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: X) 50 mM MOPS pH8.5 (SEQ ID NO: 108) VPr QP15 1200 .mu.M Peptide, 0.008 .mu.M RT/0.5 hr 1.40% YPLPWPDDDY{ FYPLPWPDDDY{LYS enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 N3}NH2 50 mM MOPS pH5.5 (SEQ ID NO: 108) (SEQ ID NO: 107) VPr QP15 1200 .mu.M Peptide, 0.008 .mu.M RT/0.5 hr 56% YPLPWPDDDY{ FYPLPWPDDDY{LYS enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 N3}NH2 50 mM MOPS pH6.5 (SEQ ID NO: 108) (SEQ ID NO: 107) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.008 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH7.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 0.008 .mu.M RT/0.5 hr 100% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2
(SEQ ID NO: 107) 50 mM MOPS pH8.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 800 pM RT/0.5 hr 2.70% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH7.5 (SEQ ID NO: 108) VPr FYPLPWPDDDY{LYS 1200 .mu.M Peptide, 800 pM RT/0.5 hr 6.80% YPLPWPDDDY{ N3}NH2 enzyme, 10 mM Mg(OAc).sub.2, LYSN3}NH2 (SEQ ID NO: 107) 50 mM MOPS pH8.5 (SEQ ID NO: 108) VPr FAAAWPDDDF1 600 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% WPDDF1 (SEQ ID NO: 11) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 112 50 mM MOPS pH8 VPr WAAAFPDDDF1 600 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% FPDDF1 (SEQ ID NO: 13) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 114) 50 mM MOPS pH8 VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% FPDDF1 (SEQ ID NO:13) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 114) 50 mM MOPS pH8 VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% FPDDF1 (SEQ ID NO: 13) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 114) 50 mM MOPS pH8, 0.2% Tween20 VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 30% 70% -3, 30% -4 (SEQ ID NO: 13) enzyme, 50/50 MOPS Mg buffer/RB1 VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 5% Almost all -1, -2 (SEQ ID NO: 13) enzyme, RB2 + Mg and -3 products VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% FPDDF1 (SEQ ID NO: 13) enzyme, RB4 (SEQ ID NO:114) VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/0.5 hr 70% 30% -3, 70% -4 (SEQ ID NO: 13) enzyme, 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M RT/2 hr 100% FPDDF1 (SEQ ID NO: 13) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 114) 50 mM MOPS pH8, 60 mM KOAc VPr WAAAFPDDDF1 300 .mu.M Peptide, 8 .mu.M 37.degree. C./2 hr 100% FPDDF1 (SEQ ID NO: 13) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 114) 50 mM MOPS pH8, 60 mM KOAc VPr FAAAYPDDDF1 600 .mu.M Peptide, 8 .mu.M RT/0.5 hr 100% YPDDF1 (SEQ ID NO: 11) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 115) 50 mM MOPS pH8, 60 mM KOAc VPr FAAAYPDDDF1 600 .mu.M Peptide, 0.8 .mu.M RT/0.5 hr 50% YPDDF1 (SEQ ID NO: 11) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 115) 50 mM MOPS pH8, 60 mM KOAc VPr FAAAYPDDDF1 600 .mu.M Peptide, 0.08 .mu.M RT/0.5 hr 5% YPDDF1 (SEQ ID NO: 11) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 115) 50 mM MOPS pH8, 60 mM KOAc VPr RRPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 79% RPFQQ (SEQ ID NO: 116) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 117) MOPS pH8, 60 mM KOAc VPr AAPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% APFQQ (SEQ ID NO: 118) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 119) MOPS pH8, 60 mM KOAc VPr KKPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 32% KPFQQ (SEQ ID NO: 120) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 121) MOPS pH8, 60 mM KOAc VPr YYPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 56% YPFQQ (SEQ ID NO: 122) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 123) MOPS pH8, 60 mM KOAc VPr FFPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% FPFQQ (SEQ ID NO: 124) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 125) MOPS pH8, 60 mM KOAc VPr DDPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% DPFQQ (SEQ ID NO: 126) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 127) MOPS pH8,60 mM KOAc VPr EEPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% EPFQQ (SEQ ID NO: 128) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 129) MOPS pH8, 60 mM KOAc VPr NNPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 15% NPFQQ (SEQ ID NO: 130) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 131) MOPS pH8, 60 mM KOAc VPr QQPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 66% QPFQQ (SEQ ID NO: 132) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 133) MOPS pH8, 60 mM KOAc VPr VVPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% VPFQQ (SEQ ID NO: 134) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 135) MOPS pH8, 60 mM KOAc VPr IIPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% IPFQQ (SEQ ID NO: 136) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 137) MOPS pH8, 60 mM KOAc VPr LLPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% LPFQQ (SEQ ID NO: 138) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 139) MOPS pH8, 60 mM KOAc VPr SSPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 48% SPFQQ (SEQ ID NO: 140) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 141) MOPS pH8, 60 mM KOAc VPr TTPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% TPFQQ (SEQ ID NO: 142) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 143) MOPS pH8, 60 mM KOAc VPr CCPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 70% CPFQQ (SEQ ID NO: 144) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 145) MOPS pH8, 60 mM KOAc VPr WWPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 82% WPFQQ (SEQ ID NO: 146) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 147) MOPS pH8, 60 mM KOAc VPr MMPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% MPFQQ (SEQ ID NO: 148) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 149) MOPS pH8, 60 mM KOAc VPr PPPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% PPFQQ (SEQ ID NO: 150) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 151) MOPS pH8, 60 mM KOAc VPr GGPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 8% GPFQQ (SEQ ID NO: 152) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 153) MOPS pH8, 60 mM KOAc VPr HHPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 12% HPFQQ (SEQ ID NO: 154) 10 mM Mg(OAc).sub.2, 50 mM (SEQ ID NO: 155) MOPS pH8, 60 mM KOAc yPIP RRPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 116) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP AAPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 118) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP KKPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 120) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8,60 mM KOAc yPIP YYPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 122) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP FFPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 124) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP DDPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 126) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP EEPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 128) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP NNPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 130) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP QQPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 132) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP VVPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 134) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP IIPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 136) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP LLPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 138) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP SSPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 140) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP TTPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 142) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP CCPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 144) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP WWPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 146) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP MMPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 148) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP PPPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 26% multiple pAzF (SEQ ID NO: 150) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP GGPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 152) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc yPIP HHPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% N/A pAzF (SEQ ID NO: 154) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET RRPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 116) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET AAPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 118) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET KKPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 120) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET YYPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 99% (SEQ ID NO: 122) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET FFPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 124) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET DDPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 4% (SEQ ID NO: 126) 10 mM Mg(OAc).sub.2, 50 mM
MOPS pH8, 60 mM KOAc hTET EEPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 128) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET NNPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 69% (SEQ ID NO: 130) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET QQPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 63% (SEQ ID NO: 132) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET VVPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 134) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET IIPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 136) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET LLPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 138) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET SSPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 140) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET TTPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 142) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET CCPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 32% (SEQ ID NO: 144) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET WWPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 4% (SEQ ID NO: 146) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET MMPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 148) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET PPPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 150) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET GGPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 33% (SEQ ID NO: 152) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc hTET HHPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 26% (SEQ ID NO: 154) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET RRPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 116) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET AAPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 118) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET KKPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 120) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET YYPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 122) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET FFPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 65% (SEQ ID NO: 124) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET DDPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 86% (SEQ ID NO: 126) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET EEPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 93% (SEQ ID NO: 128) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET NNPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 81% (SEQ ID NO: 130) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8,60 mM KOAc PfuTET QQPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 132) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET VVPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 134) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET IIPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 136) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET LLPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 99% (SEQ ID NO: 138) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET SSPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 90% (SEQ ID NO: 140) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET TTPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 142) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET CCPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 72% (SEQ ID NO: 144) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET WWPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 37% (SEQ ID NO: 146) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET MMPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 100% (SEQ ID NO: 148) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET PPPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 150) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET GGPFQQ 1 mM Peptide, 1 uM enzyme, RT/0.5 hr 0% (SEQ ID NO: 152) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc PfuTET HHPFQQ 1 mM Peptide, 1 .mu.M enzyme, RT/0.5 hr 19% (SEQ ID NO: 154) 10 mM Mg(OAc).sub.2, 50 mM MOPS pH8, 60 mM KOAc EDAPN RRPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 116) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN AAPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 118) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN KKPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 120) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN YYPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 122) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN FFPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 124) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN DDPFQQ 1 mM Peptide, 1.3 .mu.M RT/0.5 hr 21% (Glu/Asp (SEQ ID NO: 126) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN EEPFQQ 1 mM Peptide, 1.3 .mu.M RT/0.5 hr 31% (Glu/Asp (SEQ ID NO: 128) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN NNPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 130) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN QQPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 132) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN VVPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 134) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN IIPFQQ 1 mM Peptide, 1.3 .mu.M RT/0.5 hr 42% (Glu/Asp (SEQ ID NO: 136) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN LLPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 138) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN SSPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 140) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN TTPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 142) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN CCPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 144) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc EDAPN WWPFQQ 1 mM Peptide, 1.3 uM RT/0.5 hr 0% (Glu/Asp (SEQ ID NO: 146) enzyme, 10 mM Mg(OAc).sub.2, APN) 50 mM MOPS pH8, 60 mM KOAc PfuTET YAAFAAWADDDW1 1 mM Peptide, 1.0 .mu.M RT/0.5 hr Distri- Mainly ADDDWK (SEQ ID NO: 156) enzyme, 10 mM Mg(OAc).sub.2, bution (SEQ ID NO: 157) 50 mM MOPS pH8, 60 mM KOAc hTET YAAFAAWADDDW1 1 mM Peptide, 1.0 .mu.M RT/0.5 hr 100% WADDDWK (SEQ ID NO: 156) enzyme, 10 mM Mg(OAc).sub.2, (SEQ ID NO: 158) 50 mM MOPS pH8, 60 mM KOAc VPr YAAFAAWADDDW1 1 mM Peptide, 1.2 .mu.M RT/0.5 hr Distri- Mainly ADDDWK (SEQ ID NO: 156) enzyme, 10 mM Mg(OAc).sub.2, bution (SEQ ID NO: 157) 50 mM MOPS pH8, 60 mM KOAc
[0311] A summary of amino acid cleavage activities for select exopeptidases of Table 7 is shown in FIG. 16. Specific cleavage activities are shown for the following enzymes: "cVPr" (V. proteolyticus aminopeptidase), "yPIP" (Y. pestis proline iminopeptidase), "D/E APN" (L. pneumophila M1 Aminopeptidase), hTET (Pyrococcus horikoshii TET aminopeptidase), and Pfu API ("PfuTET" in Table 7). Specific activities with respect to terminal amino acids are classified as shown, with single-letter abbreviations used for amino acids ("XP-" represents any terminal amino acid (X) having an adjacent, or penultimate, proline (P) residue).
Example 4. Terminal Amino Acid Cleavage of Immobilized Peptides at Single Molecule Level
[0312] Assays for on-chip amino acid cleavage of immobilized peptides were developed using labeled peptide conjugates. The assays were designed to provide a method for determining enzymatic recognition and cleavage activity of exopeptidases toward immobilized peptides, which could permit measurement of kinetic binding parameters and general binding affinities.
[0313] To evaluate N-terminal amino acid cleavage of a peptide, a dye labeled peptide was designed and synthesized which contained an N-terminal aspartate that was attached to the dye by way of a PEG spacer. This peptide also contained a proline residue adjacent to the modified aspartate that is recognized specifically by the enzyme proline iminopeptidase (from Yersinia pestis, known elsewhere and referred to herein as "yPIP"). The enzyme yPIP should cleave only an N-terminal amino acid upstream from a proline residue.
[0314] After showing that this and other labeled peptides were efficiently cut by yPIP in bulk (e.g., as described in Example 2), an on-chip dye/peptide conjugate assay was developed to observe N-terminal amino acid cleavage at the single molecule level. FIG. 17A shows a general scheme for the dye/peptide conjugate assay (inset panel). As shown, a peptide having a label attached to an N-terminal amino acid via a spacer is immobilized to a surface by way of a linker. After being exposed to peptidase, N-terminal amino acid cleavage results in the removal of the labeled residue from a detectable observation volume and is measured by a concomitant loss in signal from the label. The enzyme-peptide complex to the right of the inset panel generically depicts the N-terminal cleavage site.
[0315] FIG. 17A shows a labeled peptide construct (at bottom) that was designed and synthesized for use in the dye/peptide conjugate assay. In these experiments, a rhodamine dye (ATTO Rho6G) was attached to an N-terminal aspartate residue of a peptide having a penultimate proline residue at the N-terminus. As shown, the peptide was further conjugated to a solubilizing DNA linker with a biotin moiety for surface immobilization.
[0316] The labeled peptide conjugate was loaded onto a glass chip having an array of sample wells. Images of the chip were acquired before and after loading to determine the percent loading of sample wells at single occupancy by rhodamine fluorescence. The enzyme yPIP was then introduced onto the loaded chip and allowed to incubate for two hours at 37.degree. C. An image of the chip following the introduction of yPIP was taken and the percentage of green dyes lost were calculated to evaluate N-terminal amino acid cleavage. FIG. 17B shows imaging results from an experiment which displayed 6-7% loading in the loading stage and 91% loss of signal in previously loaded wells after incubation with yPIP, which was indicative of N-terminal amino acid cleavage. FIG. 17C shows representative signal traces from these experiments, which demonstrate a detected increase in dye signal upon loading of labeled peptide and a detected loss in dye signal following exposure to yPIP.
[0317] As further confirmation of N-terminal amino acid cleavage at the single molecule level, on-chip FRET assays were developed to evaluate exopeptidase recognition and cleavage activity. FIG. 18A generically depicts a FRET peptide conjugate assay (panel A) and a FRET enzyme conjugate assay (panel B). In the FRET peptide conjugate assay (panel A), an immobilized peptide construct includes a FRET donor label attached to the linker and a FRET acceptor label attached at the N-terminus. N-terminal amino acid cleavage is detected by a loss in signal from the FRET acceptor label when exposed to peptidase. Additionally, this design permits monitoring loading of the peptide conjugate throughout an experiment by following emission from the FRET donor label.
[0318] In the FRET enzyme conjugate assay (panel B), an immobilized peptide construct includes a first label of a FRET pair attached to the linker and a peptidase is labeled with a second label of the FRET pair. N-terminal amino acid cleavage is detected by an enhancement in fluorescence attributable to FRET interactions, which would occur with sufficient proximity of peptidase to peptide and with sufficient residence time at the N-terminus. Additionally, this assay permits evaluating processive amino acid cleavage by a processive exopeptidase by detecting an increasing FRET signal over time with processive cleavage.
[0319] FIG. 18A also shows a FRET peptide construct under panel A that was designed and synthesized for use in the FRET peptide conjugate assay of panel A. As shown, the FRET peptide construct included a rhodamine dye (ATTO 647N) attached to an N-terminal aspartate residue of a peptide having a penultimate proline residue at the N-terminus. The peptide was further conjugated to a solubilizing DNA linker which was attached to a cyanine dye (Cy3B) for FRET and a biotin moiety for surface immobilization.
[0320] In this experiment, the FRET peptide construct was loaded onto a glass chip having an array of sample wells, and collected light was filtered first by a green filter and then a red filter. Loading of the FRET peptide construct was detected by measuring a signal passing through both the green and red filters. Terminal amino acid cleavage was detected when the signal was measurable only in the green filter, which indicated that the red dye conjugated N-terminal amino acid from the FRET peptide construct was cleaved by yPIP. This detection pattern is illustrated in panel C. As shown, if both dyes are detectable before the addition of yPIP, and only the green dye is visible after incubation with yPIP, it can be reasonably concluded this change in detection pattern is due to cleavage of the peptide and not photobleaching or loss of the peptide as a whole. Additionally, an increase in fluorescence from the lone green dye would be expected, as its emissions are no longer absorbed by the red dye.
[0321] Following loading of the FRET peptide construct onto the chip, which had been modified by surface passivation using phosphonic acid and silane, yPIP was introduced and images were obtained at several time points. To assess the overall cleaving trend, the ratio of (green)/(green+red) was computed for each experiment. This ratio increases with the extent of cleaving that occurs. FIG. 18B is a plot of FRET emission ratio across all apertures at different time points of incubation with yPIP. As shown, the green dye contribution to the ratio of fluorescence emissions increases over time during incubation with yPIP, indicating that more N-terminal aspartate residues have been cut, leaving behind the truncated peptide with just the green dye.
[0322] Cutting efficiency was then evaluated at different time points by determining at which time points dye fluorescence was observed. This was done with simple thresholding--e.g., if the average dye emission signal was >2.5 during excitation, the dyes were determined to be present (when each corresponding filter was applied). Apertures exhibiting cutting would then display both green and red dyes during the loading phase of the experiment, but only green dye at time points exposed to yPIP. As shown in FIG. 18C, progressively more cutting was observed as the chip was exposed to longer incubation times with yPIP. Example signal traces showing cutting displayed at each of the three yPIP-treated time points are shown in FIG. 18D.
[0323] Additional experiments were performed with yPIP and other peptidases using chips that had been modified by surface passivation using dextran, which produced similar results showing an increase in terminal amino acid cleavage over time following introduction of peptidase onto chips. FIG. 18E is a plot of FRET emission ratio across loaded apertures at different time points of incubation with yPIP. FIG. 18F is a plot of FRET emission ratio across loaded apertures at different time points of incubation with an aminopeptidase. Overall, the experiments here demonstrate that N-terminal amino acid cleavage is detectable in real-time at the single molecule level using different exopeptidases and different labeling strategies.
Example 5. Terminal Amino Acid Discrimination by Labeled Affinity Reagent
[0324] An adaptor protein involved in proteolytic pathways was identified as a potential candidate for use as a labeled affinity reagent for detecting N-terminal aromatic residues. The adaptor protein, ClpS2 from an .alpha.-proteobacterium (A. tumefaciens), was expressed and labeled at an exposed cysteine residue. FIG. 19A shows a crystal structure of the ClpS2 protein, with the exposed cysteine residue shown as sticks. The exposed cysteine residue was labeled with a rhodamine dye (ATTO 532).
[0325] Peptides having different N-terminal aromatic residues were prepared to test whether the labeled ClpS2 was capable of N-terminal amino acid discrimination at the single molecule level. Example single molecule intensity traces from these experiments are shown in FIG. 19B. As shown, the signal traces demonstrate residue-specific on-off binding patterns corresponding to the labeled affinity reagent reversibly binding the N-terminus of peptides having either: an N-terminal phenylalanine residue (F, top signal trace), an N-terminal tyrosine residue (Y, middle signal trace), or an N-terminal tryptophan residue (W, bottom signal trace).
[0326] Further analyses of the single molecule trajectories were carried out, with the results shown in FIGS. 19C-19E. FIG. 19C is a plot showing discriminant pulse durations (time duration of signal peaks) among the three N-terminal residues when reversibly bound by labeled ClpS2. FIG. 19D is a plot showing discriminant interpulse durations (time duration between signal pulses) among the three N-terminal residues. FIG. 19E shows plots which further illustrate the discriminant pulse durations among phenylalanine, tyrosine, and tryptophan at peptide N-termini. Mean pulse duration for the different N-terminal residues is visualized by histograms (A)-(B) and layered histogram (C).
[0327] Another adaptor protein, ClpS from Thermosynochoccus elongatus (teClpS) was evaluated for use as a labeled affinity reagent for leucine recognition. The data obtained from dwell time analysis, shown in FIGS. 19F-19H, demonstrated that the labeled teClpS protein produces detectable binding interactions with a terminal leucine residue of polypeptides with a mean pulse duration of 0.71 seconds. The amino acid sequence of the teClpS protein used in these experiments is shown in Table 1.
[0328] Similar experiments were carried out to evaluate A. tumefaciens ClpS1 and S. elongatus ClpS2 as potential reagents for leucine recognition, and GID4 as a potential reagent for proline recognition. FIG. 19I shows example results from dwell time analysis which showed differentiable recognition of phenylalanine, leucine, tryptophan, and tyrosine by A. tumefaciens ClpS1. FIG. 19J shows example results from dwell time analysis demonstrating leucine recognition by S. elongatus ClpS2. FIGS. 19K-19L show example results from dwell time analysis demonstrating proline recognition by GID4.
Example 6. Polypeptide Sequencing by Recognition During Degradation
[0329] Experiments were conducted to evaluate peptide sequencing by N-terminal amino acid recognition during an ongoing degradation reaction. Example results from these experiments are shown in FIGS. 20A-20D, which show single molecule intensity traces obtained over two independent polypeptide sequencing reactions conducted in real-time using a labeled ClpS2 protein and an aminopeptidase in the same reaction mixture. In each reaction, a polypeptide of sequence YAAWAAFADDDWK (SEQ ID NO: 78) was immobilized to a chip surface through the C-terminal lysine residue by loading the peptide composition (10 pM) onto chips for 20 minutes, and the immobilized peptide was monitored in the presence of a labeled affinity reagent (ATTO 542-labeled A. Tumefaciens ClpS2-V1 at 500 nM) and an aminopeptidase cleaving reagent (VPr at 8 .mu.M).
[0330] FIGS. 20A and 20C show signal trace data for two different sequencing runs, with the top panel (panel 1 in FIG. 20A, panel 2 in FIG. 20C) showing a full trace, and the bottom panels (Y, W, F) showing zoomed-in regions corresponding to each of the highlighted regions in the full trace. FIGS. 20B and 20D show pulse duration statistics in histograms for the trace data of the corresponding panels as labeled in FIGS. 20A and 20C, respectively. As shown in the full signal trace of each sequencing run (panels 1, 2), three separate time intervals of signal pulses were observed over the course of the reaction. As highlighted by the zoomed-in regions (panels Y, W, F), the three intervals are visually distinguishable from one another based on an observable difference in pattern of signal pulses.
[0331] To further analyze the signal pulse data, pulse duration statistics were determined for each time interval (FIGS. 20B and 20D). The differences in pulse duration distribution were determined to correspond to those observed for these amino acids individually in steady-state on-chip binding assays with ClpS2, and the signal pulse information was phenotypically consistent between intervals from sequencing runs and the individual amino acid binding assays.
[0332] As confirmed by the analysis of signal pulse information, the three time intervals of signal pulses observed over the progression of each sequencing run correspond to recognition patterns of Y, W, and F, respectively (panels 1, 2). The intervening time period between signal pulse patterns is due to the selectivity of ClpS2-V1, which does not bind to N-terminal alanine residues. As illustrated by the full signal trace, the first interval corresponds to Y recognition, which is followed by a pause as VPr peptidase cuts Y and two alanine residues, followed by the second interval corresponding to W recognition, which is followed by another pause as VPr peptidase cuts W and two alanine residues, and finally the third interval corresponding to F recognition before VPr peptidase cuts off the F and stops at the remaining ADDDWK peptide. These results show that pulse duration information, which was obtained by terminal amino acid recognition during an ongoing degradation reaction, can be used to determine characteristic patterns that discriminate between different types of terminal amino acids.
Example 7. Terminal Amino Acid Identification and Cleavage by Labeled Exopeptidase
[0333] Studies were performed to investigate the potential for a single reagent that is capable of both identifying a terminal amino acid of a peptide and cleaving the terminal amino acid from the peptide. As a single reagent, an exopeptidase must be able to bind to the peptide while retaining cleavage activity toward a terminal residue. Accordingly, an initial approach employing traditional labeling strategies was carried out by targeting the native surface-exposed amino acids of different exopeptidases. In these experiments, surface-exposed cysteine (--SH) or lysine (--NH.sub.2) residues were labeled with fluorescent dyes, which proved to be a robust methodology for exopeptidase labeling. In certain cases, however, this approach produced a heterogeneous population of proteins that are labeled with one or more dyes.
[0334] In order to more precisely control where labeling occurs on exopeptidases and ensure that each exopeptidase molecule is labeled with a single fluorescent dye (as well as eliminate off-target reactivity of the dye), a new labeling strategy was investigated. In these experiments, labeled exopeptidases were prepared using a site-specific labeling strategy in which an unnatural amino acid containing a reactive functional group is introduced into the exopeptidase (see, e.g., Chin, J. W., et al. J Am Chem Soc. 2002 Aug. 7; 124(31):9026-9027).
[0335] The proline iminopeptidase from Yersinia pestis (yPIP) was modified by mutation of a lysine residue at position 287 to a residue having a para-azidophenylalanine (pAzF) side chain. FIG. 21A shows a crystal structure of yPIP, with the mutation indicated by the chemical structure of pAzF shown with the K287 sidechain shown as sticks. This mutation site was selected based on the stability provided by the alpha helix at this position and to ensure that the new azido functional group is solvent exposed.
[0336] A pEVOL plasmid containing the mutant amino tRNA synthetase and the mutant tRNA necessary to incorporate pAzF into the amino acid chain was obtained. The amber stop codon (TAG), which is necessary for the specific incorporation of pAzF, was then introduced into the cDNA using the QuickChange II mutagenesis kit. The cDNA was then sequenced and the TAG codon position was confirmed. This was followed by co-transfection of both the pET21b+ plasmid containing the yPIP amber mutant and the pEVOL plasmid containing the cellular machinery to charge the tRNA for the amber codon with pAzF. The co-transfected cells were then grown to 0.8 ODU, induced with 0.02% arabinose and 1 mM IPTG in the presence of 2 mM pAzF in 2 L of LB, and harvested using chemolysis. Purification was carried out using a 5 mL affinity chromatography column, and the protein was eluted in 100 mM imidazole. The resulting protein was then dialyzed and concentrated into 50 mM HEPES pH 8.0 and 0.5 M KCl, aliquoted, and flash frozen prior to storage at -20.degree. C.
[0337] To confirm the presence of the azido group in the purified protein, DBCO-Cy3 (2 mM) was reacted with the pAzF-yPIP variant (220 .mu.M) (Reaction Conditions: 50 mM HEPES pH 8.0, 0.5 mM KCl, 20% DMSO; 10 hours at 37.degree. C., 48 hours at room temperature). The protein reaction product was purified by size-exclusion chromatography, and it was determined that the resulting protein was 100% labeled with the azide-reactive DBCO-Cy3 reagent (FIG. 21B), indicating robust incorporation of the unnatural amino acid.
[0338] Protein labeling and purity of the final product was confirmed by SDS-PAGE analysis of the unlabeled and labeled pAzF variant. FIG. 21C shows a picture of SDS-PAGE gel confirming Cy3-labeling of pAzF-yPIP (overexposed image of gel shown in FIG. 21D to show ladder). FIG. 21E shows a picture of Coomassie-stained gel confirming that both dye and protein co-migrate and are pure.
[0339] The dye-labeled pAzF-yPIP variant was used in an activity assay to confirm that the enzyme was still active after labeling and purification. As shown in FIG. 21F, Cy3-pAzF-yPIP was able to hydrolyze 100% of the peptide substrate in 1 hour using 1000-fold excess substrate, as measured by HPLC. These experiments demonstrate a methodology which allows site-specific modification and labeling of an exopeptidase with minimal perturbation of the native protein structure/function.
Example 8. Recognition of Modified Amino Acids in Polypeptide Sequencing
[0340] Experiments were performed to evaluate recognition of amino acids containing specific post-translation modifications. A triple-mutant variant (TBV, S10A, K15L) of the Src Homology 2 (SH2) domain from Fyn, a tyrosine kinase, was tested as a potential recognition molecule for phosphorylated tyrosine residues in peptide sequencing. The variant protein was immobilized to the bottom of sample wells, and single-molecule signal traces were collected upon addition of a fluorescently-labeled peptide containing N-terminal phospho-tyrosine. Peptide binding by the immobilized protein was detected during these experiments, as shown by the representative traces in FIG. 22A. Pulse duration data collected during these experiments is shown in FIG. 22B (top, middle, and bottom plots corresponding to the top, middle, and bottom traces of FIG. 22A, respectively). Pulse duration and interpulse duration statistics are shown in FIG. 22C (top and bottom panels, respectively).
[0341] Control experiments were performed to confirm that the Fyn protein was specific for the phosphorylated tyrosine. The experiments were repeated for each of three different peptides: a first peptide containing N-terminal unmodified tyrosine (Y; FIG. 22D), a second peptide containing N-terminal and penultimate unmodified tyrosines (YY; FIG. 22E), and a third peptide containing N-terminal phospho-serine (FIG. 22F). As shown, binding was not detected with any of the peptides used in the negative control experiments.
Example 9. Recognition of Penultimate Amino Acids in Polypeptide Sequencing
[0342] Experiments were performed to determine the effects of penultimate amino acids on pulse duration for A. Tumefaciens ClpS2-V1. Forty-nine different fluorescently-labeled peptides were prepared containing unique dipeptide sequences at the N-terminus, where the N-terminal amino acid was F, W, or Y, and the penultimate position was one of the 20 natural amino acids. For each experiment, ClpS2-V1 was immobilized at the bottom of sample wells, and single-molecule signal traces were collected for 10-20 minutes upon addition of one of the fluorescently-labeled peptides. Pulse duration data was collected for a minimum of 50 sample wells for each peptide.
[0343] FIG. 23 shows the median pulse duration for each of the 50 peptides, with data points grouped by penultimate amino acid (x-axis) and N-terminal amino acids represented with different symbols.
Example 10. Simultaneous Amino Acid Recognition with Multiple Recognition Molecules
[0344] Single-molecule peptide recognition experiments were performed to demonstrate terminal amino acid recognition of an immobilized peptide by more than one labeled recognition molecule. Single peptide molecules containing N-terminal phenylalanine (FYPLPWPDDDY (SEQ ID NO: 79)) were immobilized in sample wells of a chip. Buffer containing 500 nM each of atClpS1 (Agrobacterium tumifaciens ClpS1; sequence provided in Table 1) and atClpS2-V1 (Agrobacterium tumifaciens ClpS2 variant 1; sequence provided in Table 1) was added, where atClpS1 and atClpS2-V1 were labeled with Cy3 and Cy3B, respectively. Since the intensity of Cy3B is higher than Cy3, atClpS2-V1 binding events were readily distinguishable from atClpS1 binding events.
[0345] FIGS. 24A-24C shows the results of the experiments showing single-molecule peptide recognition with differentially labeled recognition molecules. A representative trace is displayed in FIG. 24A. The pulse duration distributions were distinct for each binder (FIG. 24B) and corresponded to their kinetic profiles as observed in single-binder experiments. Mean pulse duration was 1.3 seconds for atClpS1 and 1.0 seconds for atClpS2-V1 (FIG. 24C). Pulse rate was also distinct: 8.1 pulses/min for atClpS1 and 14.1 pulses/min for atClp2-V1 (FIG. 24C). Thus, when more than one recognition molecule is included for dynamic recognition of immobilized peptides, the binding characteristics of each recognition molecule (including pulse duration, interpulse duration, and pulse rate) can simultaneously provide information about peptide sequence.
Example 11. Enhancing Photostability with Recognition Molecule Linkers
[0346] Experiments were performed to evaluate the photostability of immobilized peptides during single-molecule sequencing. The dye-labeled atClpS2-V1 described in Example 5 was added to sample wells containing immobilized peptide substrates in the presence of excitation light at 532 nm to monitor recognition by emission from ATTO 532. A representative trace is shown in FIG. 25A. As shown in the top panel, recognition was observed to cease at approximately 600 seconds into the experiment. The bottom panel is a zoomed view showing signal pulses at approximately 180-430 seconds into the reaction.
[0347] FIG. 25B shows a visualization of the crystal structure of the ClpS2 protein used in these experiments. As shown, the cysteine residue that serves as the dye conjugation site is approximately 2 nm from the terminal amino acid binding site. It was hypothesized that photodamage to the peptide was caused by proximity of the dye to the N-terminus of peptide during binding. To mitigate the potential photodamaging effects of dye proximity, the ClpS2 protein was dye-labeled through a linker that increased distance between the dye and N-terminus of peptide by more than 10 nm. The linker included streptavidin and a double-stranded nucleic acid; the double-stranded nucleic acid was labeled with two Cy3B dye molecules and attached to streptavidin through a bis-biotin moiety, and a ClpS2 protein was attached to each of the remaining two binding sites on streptavidin through a biotin moiety. A representative trace using this dye-shielded ClpS2 molecule is shown in FIG. 25C. As shown in the top panel, recognition time was extended to approximately 6,000 seconds into the experiment. The bottom panel is a zoomed view showing signal pulses at approximately 750-930 seconds into the reaction.
[0348] A DNA-streptavidin recognition molecule was generated with a linker containing a double-stranded nucleic acid labeled with two Cy3B dye molecules and attached to streptavidin through a bis-biotin moiety, and a single ClpS2 protein attached to the remaining two binding sites on streptavidin through a bis-biotin moiety. This construct was used in a single-molecule peptide sequencing reaction, and representative traces from these experiments are shown in FIGS. 26A-26D.
[0349] The sequencing experiments described in example 6 were repeated, with the reaction conditions changed as follows: the DNA-streptavidin ClpS2 recognition molecule was used in combination with hTET amino acid cleaving reagent. A representative signal trace is shown in FIG. 27.
Example 12. Sequencing by Recognition During Degradation by Multiple Exopeptidases
[0350] Experiments were performed to evaluate the use of multiple types of exopeptidases with differential cleavage specificities in a single-molecule peptide sequencing reaction mixture. Single peptide molecules (YAAWAAFADDDWK (SEQ ID NO: 78)) were immobilized through a C-terminal lysine residue in sample wells of a chip. Buffer containing atClpS2-V1 for amino acid recognition and hTET for amino acid cleavage was added. A representative trace is displayed in FIG. 28A, with expanded views of pulse pattern regions shown in FIG. 28B.
[0351] An experiment was carried out to evaluate sequencing reactions in the presence of two types of exopeptidases with differential specificities. Single peptide molecules (FYPLPWPDDDYK (SEQ ID NO: 80)) were immobilized through a C-terminal lysine residue in sample wells of a chip. Buffer containing atClpS2-V1 for amino acid recognition, and both hTET and yPIP for amino acid cleavage was added. A representative trace is displayed in FIG. 28C, with expanded views of pulse pattern regions shown in FIG. 28D. Additional representative traces from these reaction conditions are shown in FIG. 28E.
[0352] Further experiments were carried out to evaluate sequencing reactions in the presence of two types of exopeptidases with differential specificities. Single peptide molecules (YPLPWPDDDYK (SEQ ID NO: 81)) were immobilized through a C-terminal lysine residue in sample wells of a chip. In one experiment, buffer containing atClpS2-V1 for amino acid recognition, and both hTET and yPIP for amino acid cleavage was added. A representative trace is displayed in FIG. 28F, with expanded views of pulse pattern regions shown in FIG. 28G. Additional representative traces from these reaction conditions are shown in FIG. 28H. In a further experiment, buffer (50 mM MOPS, 60 mM KOAc, 200 .mu.M Co(OAc).sub.2) containing atClpS2-V1 for amino acid recognition, and both PfuTET and yPIP for amino acid cleavage was added. A representative trace is displayed in FIG. 28I, with expanded views of pulse pattern regions shown in FIG. 28J.
EQUIVALENTS AND SCOPE
[0353] In the claims articles such as "a," "an," and "the" may mean one or more than one unless indicated to the contrary or otherwise evident from the context. Claims or descriptions that include "or" between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. The invention includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. The invention includes embodiments in which more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process.
[0354] Furthermore, the invention encompasses all variations, combinations, and permutations in which one or more limitations, elements, clauses, and descriptive terms from one or more of the listed claims is introduced into another claim. For example, any claim that is dependent on another claim can be modified to include one or more limitations found in any other claim that is dependent on the same base claim. Where elements are presented as lists, e.g., in Markush group format, each subgroup of the elements is also disclosed, and any element(s) can be removed from the group. It should it be understood that, in general, where the invention, or aspects of the invention, is/are referred to as comprising particular elements and/or features, certain embodiments of the invention or aspects of the invention consist, or consist essentially of, such elements and/or features. For purposes of simplicity, those embodiments have not been specifically set forth in haec verba herein.
[0355] The phrase "and/or," as used herein in the specification and in the claims, should be understood to mean "either or both" of the elements so conjoined, i.e., elements that are conjunctively present in some cases and disjunctively present in other cases. Multiple elements listed with "and/or" should be construed in the same fashion, i.e., "one or more" of the elements so conjoined. Other elements may optionally be present other than the elements specifically identified by the "and/or" clause, whether related or unrelated to those elements specifically identified. Thus, as a non-limiting example, a reference to "A and/or B", when used in conjunction with open-ended language such as "comprising" can refer, in one embodiment, to A only (optionally including elements other than B); in another embodiment, to B only (optionally including elements other than A); in yet another embodiment, to both A and B (optionally including other elements); etc.
[0356] As used herein in the specification and in the claims, "or" should be understood to have the same meaning as "and/or" as defined above. For example, when separating items in a list, "or" or "and/or" shall be interpreted as being inclusive, i.e., the inclusion of at least one, but also including more than one, of a number or list of elements, and, optionally, additional unlisted items. Only terms clearly indicated to the contrary, such as "only one of" or "exactly one of," or, when used in the claims, "consisting of," will refer to the inclusion of exactly one element of a number or list of elements. In general, the term "or" as used herein shall only be interpreted as indicating exclusive alternatives (i.e. "one or the other but not both") when preceded by terms of exclusivity, such as "either," "one of" "only one of" or "exactly one of" "Consisting essentially of," when used in the claims, shall have its ordinary meaning as used in the field of patent law.
[0357] As used herein in the specification and in the claims, the phrase "at least one," in reference to a list of one or more elements, should be understood to mean at least one element selected from any one or more of the elements in the list of elements, but not necessarily including at least one of each and every element specifically listed within the list of elements and not excluding any combinations of elements in the list of elements. This definition also allows that elements may optionally be present other than the elements specifically identified within the list of elements to which the phrase "at least one" refers, whether related or unrelated to those elements specifically identified. Thus, as a non-limiting example, "at least one of A and B" (or, equivalently, "at least one of A or B," or, equivalently "at least one of A and/or B") can refer, in one embodiment, to at least one, optionally including more than one, A, with no B present (and optionally including elements other than B); in another embodiment, to at least one, optionally including more than one, B, with no A present (and optionally including elements other than A); in yet another embodiment, to at least one, optionally including more than one, A, and at least one, optionally including more than one, B (and optionally including other elements); etc.
[0358] It should also be understood that, unless clearly indicated to the contrary, in any methods claimed herein that include more than one step or act, the order of the steps or acts of the method is not necessarily limited to the order in which the steps or acts of the method are recited.
[0359] In the claims, as well as in the specification above, all transitional phrases such as "comprising," "including," "carrying," "having," "containing," "involving," "holding," "composed of," and the like are to be understood to be open-ended, i.e., to mean including but not limited to. Only the transitional phrases "consisting of" and "consisting essentially of" shall be closed or semi-closed transitional phrases, respectively, as set forth in the United States Patent Office Manual of Patent Examining Procedures, Section 2111.03. It should be appreciated that embodiments described in this document using an open-ended transitional phrase (e.g., "comprising") are also contemplated, in alternative embodiments, as "consisting of" and "consisting essentially of" the feature described by the open-ended transitional phrase. For example, if the application describes "a composition comprising A and B," the application also contemplates the alternative embodiments "a composition consisting of A and B" and "a composition consisting essentially of A and B."
[0360] Where ranges are given, endpoints are included. Furthermore, unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or sub-range within the stated ranges in different embodiments of the invention, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise.
[0361] This application refers to various issued patents, published patent applications, journal articles, and other publications, all of which are incorporated herein by reference. If there is a conflict between any of the incorporated references and the instant specification, the specification shall control. In addition, any particular embodiment of the present invention that falls within the prior art may be explicitly excluded from any one or more of the claims. Because such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the invention can be excluded from any claim, for any reason, whether or not related to the existence of prior art.
[0362] Those skilled in the art will recognize or be able to ascertain using no more than routine experimentation many equivalents to the specific embodiments described herein. The scope of the present embodiments described herein is not intended to be limited to the above Description, but rather is as set forth in the appended claims. Those of ordinary skill in the art will appreciate that various changes and modifications to this description may be made without departing from the spirit or scope of the present invention, as defined in the following claims.
[0363] The recitation of a listing of chemical groups in any definition of a variable herein includes definitions of that variable as any single group or combination of listed groups. The recitation of an embodiment for a variable herein includes that embodiment as any single embodiment or in combination with any other embodiments or portions thereof. The recitation of an embodiment herein includes that embodiment as any single embodiment or in combination with any other embodiments or portions thereof.
Sequence CWU 1
1
1581103PRTAgrobacterium tumifaciens 1Met Ser Asp Ser Pro Val Asp
Leu Lys Pro Lys Pro Lys Val Lys Pro1 5 10 15Lys Leu Glu Arg Pro Lys
Leu Tyr Lys Val Met Leu Leu Asn Asp Asp 20 25 30Tyr Thr Pro Met Ser
Phe Val Thr Val Val Leu Lys Ala Val Phe Arg 35 40 45Met Ser Glu Asp
Thr Gly Arg Arg Val Met Met Thr Ala His Arg Phe 50 55 60Gly Ser Ala
Val Val Val Val Cys Glu Arg Asp Ile Ala Glu Thr Lys65 70 75 80Ala
Lys Glu Ala Thr Asp Leu Gly Lys Glu Ala Gly Phe Pro Leu Met 85 90
95Phe Thr Thr Glu Pro Glu Glu 1002103PRTAgrobacterium tumifaciens
2Met Ser Asp Ser Pro Val Asp Leu Lys Pro Lys Pro Lys Val Lys Pro1 5
10 15Lys Leu Glu Arg Pro Lys Leu Tyr Lys Val Met Leu Leu Asn Asp
Asp 20 25 30Tyr Thr Pro Arg Glu Phe Val Thr Val Val Leu Lys Ala Val
Phe Arg 35 40 45Met Ser Glu Asp Thr Gly Arg Arg Val Met Met Thr Ala
His Arg Phe 50 55 60Gly Ser Ala Val Val Val Val Cys Glu Arg Asp Ile
Ala Glu Thr Lys65 70 75 80Ala Lys Glu Ala Thr Asp Leu Gly Lys Glu
Ala Gly Phe Pro Leu Met 85 90 95Phe Thr Thr Glu Pro Glu Glu
1003103PRTAgrobacterium tumifaciens 3Met Ser Asp Ser Pro Val Asp
Leu Lys Pro Lys Pro Lys Val Lys Pro1 5 10 15Lys Leu Glu Arg Pro Lys
Leu Tyr Lys Val Met Leu Leu Asn Asp Asp 20 25 30Tyr Thr Pro Arg Glu
Phe Val Thr Val Val Leu Lys Ala Val Phe Arg 35 40 45Met Ser Glu Asp
Thr Gly Arg Arg Val Met Met Thr Ala His Arg Phe 50 55 60Gly Ser Ala
Val Val Val Val Ser Glu Arg Asp Ile Ala Glu Thr Lys65 70 75 80Ala
Lys Glu Ala Thr Asp Leu Gly Lys Glu Ala Gly Phe Pro Leu Met 85 90
95Phe Thr Thr Glu Pro Glu Glu 1004117PRTAgrobacterium tumifaciens
4Met Ile Ala Glu Pro Ile Cys Met Gln Gly Glu Gly Asp Gly Glu Asp1 5
10 15Gly Gly Thr Asn Arg Gly Thr Ser Val Ile Thr Arg Val Lys Pro
Lys 20 25 30Thr Lys Arg Pro Asn Leu Tyr Arg Val Leu Leu Leu Asn Asp
Asp Tyr 35 40 45Thr Pro Met Glu Phe Val Ile His Ile Leu Glu Arg Phe
Phe Gln Lys 50 55 60Asp Arg Glu Ala Ala Thr Arg Ile Met Leu His Val
His Gln His Gly65 70 75 80Val Gly Glu Cys Gly Val Phe Thr Tyr Glu
Val Ala Glu Thr Lys Val 85 90 95Ser Gln Val Met Asp Phe Ala Arg Gln
His Gln His Pro Leu Gln Cys 100 105 110Val Met Glu Lys Lys
1155103PRTAgrobacterium tumifaciens 5Met Ser Asp Ser Pro Val Asp
Leu Lys Pro Lys Pro Lys Val Lys Pro1 5 10 15Lys Leu Glu Arg Pro Lys
Leu Tyr Lys Val Met Leu Leu Asn Asp Asp 20 25 30Tyr Thr Pro Met Ser
Phe Val Thr Val Val Leu Lys Ala Val Phe Arg 35 40 45Met Ser Glu Asp
Thr Gly Arg Arg Val Met Met Thr Ala His Arg Phe 50 55 60Gly Ser Ala
Val Val Val Val Ser Glu Arg Asp Ile Ala Glu Thr Lys65 70 75 80Ala
Lys Glu Ala Thr Asp Leu Gly Lys Glu Ala Gly Phe Pro Leu Met 85 90
95Phe Thr Thr Glu Pro Glu Glu 1006117PRTAgrobacterium tumifaciens
6Met Ile Ala Glu Pro Ile Ser Met Gln Gly Glu Gly Asp Gly Glu Asp1 5
10 15Gly Gly Thr Asn Arg Gly Thr Ser Val Ile Thr Arg Val Lys Pro
Lys 20 25 30Thr Lys Arg Pro Asn Leu Tyr Arg Val Leu Leu Leu Asn Asp
Asp Tyr 35 40 45Thr Pro Met Glu Phe Val Ile His Ile Leu Glu Arg Phe
Phe Gln Lys 50 55 60Asp Arg Glu Ala Ala Thr Arg Ile Met Leu His Val
His Gln His Gly65 70 75 80Val Gly Glu Cys Gly Val Phe Thr Tyr Glu
Val Ala Glu Thr Lys Val 85 90 95Ser Gln Val Met Asp Phe Ala Arg Gln
His Gln His Pro Leu Gln Cys 100 105 110Val Met Glu Lys Lys
1157117PRTAgrobacterium tumifaciens 7Met Ile Ala Glu Pro Ile Ser
Met Gln Gly Glu Gly Asp Gly Glu Asp1 5 10 15Gly Gly Thr Asn Arg Gly
Thr Ser Val Ile Thr Arg Val Lys Pro Lys 20 25 30Thr Lys Arg Pro Asn
Leu Tyr Arg Val Leu Leu Leu Asn Asp Asp Tyr 35 40 45Thr Pro Met Glu
Phe Val Ile His Ile Leu Glu Arg Phe Phe Gln Lys 50 55 60Asp Arg Glu
Ala Ala Thr Arg Ile Met Leu His Val His Gln His Gly65 70 75 80Val
Gly Glu Ser Gly Val Phe Thr Tyr Glu Val Ala Glu Thr Lys Val 85 90
95Ser Gln Val Met Asp Phe Ala Arg Gln His Gln His Pro Leu Gln Ser
100 105 110Val Met Glu Lys Lys 1158103PRTAgrobacterium tumifaciens
8Met Ser Asp Ser Pro Val Asp Leu Lys Pro Lys Pro Lys Val Lys Pro1 5
10 15Lys Leu Glu Arg Pro Lys Leu Tyr Lys Val Ile Leu Leu Asn Asp
Asp 20 25 30Tyr Thr Pro Met Glu Phe Val Val Glu Val Leu Lys Arg Val
Phe Asn 35 40 45Met Ser Glu Glu Gln Ala Arg Arg Val Met Met Thr Ala
His Lys Lys 50 55 60Gly Lys Ala Val Val Gly Val Cys Pro Arg Asp Ile
Ala Glu Thr Lys65 70 75 80Ala Lys Gln Ala Thr Asp Leu Ala Arg Glu
Ala Gly Phe Pro Leu Met 85 90 95Phe Thr Thr Glu Pro Glu Glu
1009103PRTAgrobacterium tumifaciens 9Met Ser Asp Ser Pro Val Asp
Leu Lys Pro Lys Pro Lys Val Lys Pro1 5 10 15Lys Leu Glu Arg Pro Lys
Leu Tyr Lys Val Ile Leu Leu Asn Asp Asp 20 25 30Tyr Thr Pro Met Glu
Phe Val Val Glu Val Leu Lys Arg Val Phe Asn 35 40 45Met Ser Glu Glu
Gln Ala Arg Arg Val Met Met Thr Ala His Lys Lys 50 55 60Gly Lys Ala
Val Val Gly Val Ser Pro Arg Asp Ile Ala Glu Thr Lys65 70 75 80Ala
Lys Gln Ala Thr Asp Leu Ala Arg Glu Ala Gly Phe Pro Leu Met 85 90
95Phe Thr Thr Glu Pro Glu Glu 1001095PRTSynechococcus elongatus
10Met Ala Val Glu Thr Ile Gln Lys Pro Glu Thr Thr Thr Lys Arg Lys1
5 10 15Ile Ala Pro Arg Tyr Arg Val Leu Leu His Asn Asp Asp Phe Asn
Pro 20 25 30Met Glu Tyr Val Val Met Val Leu Met Gln Thr Val Pro Ser
Leu Thr 35 40 45Gln Pro Gln Ala Val Asp Ile Met Met Glu Ala His Thr
Asn Gly Thr 50 55 60Gly Leu Val Ile Thr Cys Asp Ile Glu Pro Ala Glu
Phe Tyr Cys Glu65 70 75 80Gln Leu Lys Ser His Gly Leu Ser Ser Ser
Ile Glu Pro Asp Asp 85 90 9511136PRTSynechococcus elongatus 11Met
Ser Pro Gln Pro Asp Glu Ser Val Leu Ser Ile Leu Gly Val Pro1 5 10
15Arg Pro Cys Val Lys Lys Arg Ser Arg Asn Asp Ala Phe Val Leu Thr
20 25 30Val Leu Thr Cys Ser Leu Gln Ala Ile Ala Ala Pro Ala Thr Ala
Pro 35 40 45Gly Thr Thr Thr Thr Arg Val Arg Gln Pro Tyr Pro His Phe
Arg Val 50 55 60Ile Val Leu Asp Asp Asp Val Asn Thr Phe Gln His Val
Ala Glu Cys65 70 75 80Leu Leu Lys Tyr Ile Pro Gly Met Thr Gly Asp
Arg Ala Trp Asp Leu 85 90 95Thr Asn Gln Val His Tyr Glu Gly Ala Ala
Thr Val Trp Ser Gly Pro 100 105 110Gln Glu Gln Ala Glu Leu Tyr His
Glu Gln Leu Arg Arg Glu Gly Leu 115 120 125Thr Met Ala Pro Leu Glu
Ala Ala 130 1351290PRTThermosynechococcus elongatus 12Met Pro Gln
Glu Arg Gln Gln Val Thr Arg Lys His Tyr Pro Asn Tyr1 5 10 15Lys Val
Ile Val Leu Asn Asp Asp Phe Asn Thr Phe Gln His Val Ala 20 25 30Ala
Cys Leu Met Lys Tyr Ile Pro Asn Met Thr Ser Asp Arg Ala Trp 35 40
45Glu Leu Thr Asn Gln Val His Tyr Glu Gly Gln Ala Ile Val Trp Val
50 55 60Gly Pro Gln Glu Gln Ala Glu Leu Tyr His Glu Gln Leu Leu Arg
Ala65 70 75 80Gly Leu Thr Met Ala Pro Leu Glu Pro Glu 85
9013106PRTEscherichia coli 13Met Gly Lys Thr Asn Asp Trp Leu Asp
Phe Asp Gln Leu Ala Glu Glu1 5 10 15Lys Val Arg Asp Ala Leu Lys Pro
Pro Ser Met Tyr Lys Val Ile Leu 20 25 30Val Asn Asp Asp Tyr Thr Pro
Met Glu Phe Val Ile Asp Val Leu Gln 35 40 45Lys Phe Phe Ser Tyr Asp
Val Glu Arg Ala Thr Gln Leu Met Leu Ala 50 55 60Val His Tyr Gln Gly
Lys Ala Ile Cys Gly Val Phe Thr Ala Glu Val65 70 75 80Ala Glu Thr
Lys Val Ala Met Val Asn Lys Tyr Ala Arg Glu Asn Glu 85 90 95His Pro
Leu Leu Cys Thr Leu Glu Lys Ala 100 10514106PRTEscherichia coli
14Met Gly Lys Thr Asn Asp Trp Leu Asp Phe Asp Gln Leu Ala Glu Glu1
5 10 15Lys Val Arg Asp Ala Leu Lys Pro Pro Ser Met Tyr Lys Val Ile
Leu 20 25 30Val Asn Asp Asp Tyr Thr Pro Ala Glu Phe Val Ile Asp Val
Leu Gln 35 40 45Lys Phe Phe Ser Tyr Asp Val Glu Arg Ala Thr Gln Leu
Met Leu Ala 50 55 60Val His Tyr Gln Gly Lys Ala Ile Cys Gly Val Phe
Thr Ala Glu Val65 70 75 80Ala Glu Thr Lys Val Ala Met Val Asn Lys
Tyr Ala Arg Glu Asn Glu 85 90 95His Pro Leu Leu Cys Thr Leu Glu Lys
Ala 100 10515192PRTPlasmodium falciparum 15Met Phe Lys Asp Leu Lys
Pro Phe Phe Leu Cys Ile Ile Leu Leu Leu1 5 10 15Leu Leu Ile Tyr Lys
Cys Thr His Ser Tyr Asn Ile Lys Asn Lys Asn 20 25 30Cys Pro Leu Asn
Phe Met Asn Ser Cys Val Arg Ile Asn Asn Val Asn 35 40 45Lys Asn Thr
Asn Ile Ser Phe Pro Lys Glu Leu Gln Lys Arg Pro Ser 50 55 60Leu Val
Tyr Ser Gln Lys Asn Phe Asn Leu Glu Lys Ile Lys Lys Leu65 70 75
80Arg Asn Val Ile Lys Glu Ile Lys Lys Asp Asn Ile Lys Glu Ala Asp
85 90 95Glu His Glu Lys Lys Glu Arg Glu Lys Glu Thr Ser Ala Trp Lys
Val 100 105 110Ile Leu Tyr Asn Asp Asp Ile His Asn Phe Thr Tyr Val
Thr Asp Val 115 120 125Ile Val Lys Val Val Gly Gln Ile Ser Lys Ala
Lys Ala His Thr Ile 130 135 140Thr Val Glu Ala His Ser Thr Gly Gln
Ala Leu Ile Leu Ser Thr Trp145 150 155 160Lys Ser Lys Ala Glu Lys
Tyr Cys Gln Glu Leu Gln Gln Asn Gly Leu 165 170 175Thr Val Ser Ile
Ile His Glu Ser Gln Leu Lys Asp Lys Gln Lys Lys 180 185
19016236PRTEscherichia coli 16Met Arg Leu Val Gln Leu Ser Arg His
Ser Ile Ala Phe Pro Ser Pro1 5 10 15Glu Gly Ala Leu Arg Glu Pro Asn
Gly Leu Leu Ala Leu Gly Gly Asp 20 25 30Leu Ser Pro Ala Arg Leu Leu
Met Ala Tyr Gln Arg Gly Ile Phe Pro 35 40 45Trp Phe Ser Pro Gly Asp
Pro Ile Leu Trp Trp Ser Pro Asp Pro Arg 50 55 60Ala Val Leu Trp Pro
Glu Ser Leu His Ile Ser Arg Ser Met Lys Arg65 70 75 80Phe His Lys
Arg Ser Pro Tyr Arg Val Thr Met Asn Tyr Ala Phe Gly 85 90 95Gln Val
Ile Glu Gly Cys Ala Ser Asp Arg Glu Glu Gly Thr Trp Ile 100 105
110Thr Arg Gly Val Val Glu Ala Tyr His Arg Leu His Glu Leu Gly His
115 120 125Ala His Ser Ile Glu Val Trp Arg Glu Asp Glu Leu Val Gly
Gly Met 130 135 140Tyr Gly Val Ala Gln Gly Thr Leu Phe Cys Gly Glu
Ser Met Phe Ser145 150 155 160Arg Met Glu Asn Ala Ser Lys Thr Ala
Leu Leu Val Phe Cys Glu Glu 165 170 175Phe Ile Gly His Gly Gly Lys
Leu Ile Asp Cys Gln Val Leu Asn Asp 180 185 190His Thr Ala Ser Leu
Gly Ala Cys Glu Ile Pro Arg Arg Asp Tyr Leu 195 200 205Asn Tyr Leu
Asn Gln Met Arg Leu Gly Arg Leu Pro Asn Asn Phe Trp 210 215 220Val
Pro Arg Cys Leu Phe Ser Pro Gln Glu Leu Glu225 230
23517231PRTVibrio vulnificus 17Met Ser Ser Asp Ile His Gln Ile Lys
Ile Gly Leu Thr Asp Asn His1 5 10 15Pro Cys Ser Tyr Leu Pro Glu Arg
Lys Glu Arg Val Ala Val Ala Leu 20 25 30Glu Ala Asp Met His Thr Ala
Asp Asn Tyr Glu Val Leu Leu Ala Asn 35 40 45Gly Phe Arg Arg Ser Gly
Asn Thr Ile Tyr Lys Pro His Cys Asp Ser 50 55 60Cys His Ser Cys Gln
Pro Ile Arg Ile Ser Val Pro Asp Ile Glu Leu65 70 75 80Ser Arg Ser
Gln Lys Arg Leu Leu Ala Lys Ala Arg Ser Leu Ser Trp 85 90 95Ser Met
Lys Arg Asn Met Asp Glu Asn Trp Phe Asp Leu Tyr Ser Arg 100 105
110Tyr Ile Val Ala Arg His Arg Asn Gly Thr Met Tyr Pro Pro Lys Lys
115 120 125Asp Asp Phe Ala His Phe Ser Arg Asn Gln Trp Leu Thr Thr
Gln Phe 130 135 140Leu His Ile Tyr Glu Gly Gln Arg Leu Ile Ala Val
Ala Val Thr Asp145 150 155 160Ile Met Asp His Cys Ala Ser Ala Phe
Tyr Thr Phe Phe Glu Pro Glu 165 170 175His Glu Leu Ser Leu Gly Thr
Leu Ala Val Leu Phe Gln Leu Glu Phe 180 185 190Cys Gln Glu Glu Lys
Lys Gln Trp Leu Tyr Leu Gly Tyr Gln Ile Asp 195 200 205Glu Cys Pro
Ala Met Asn Tyr Lys Val Arg Phe His Arg His Gln Lys 210 215 220Leu
Val Asn Gln Arg Trp Gln225 2301883PRTSaccharomyces cerevisiae 18Met
Gly Ser Val His Lys His Thr Gly Arg Asn Cys Gly Arg Lys Phe1 5 10
15Lys Ile Gly Glu Pro Leu Tyr Arg Cys His Glu Cys Gly Cys Asp Asp
20 25 30Thr Cys Val Leu Cys Ile His Cys Phe Asn Pro Lys Asp His Val
Asn 35 40 45His His Val Cys Thr Asp Ile Cys Thr Glu Phe Thr Ser Gly
Ile Cys 50 55 60Asp Cys Gly Asp Glu Glu Ala Trp Asn Ser Pro Leu His
Cys Lys Ala65 70 75 80Glu Glu Gln19167PRTHomo sapiens 19Met Ser Gly
Ser Lys Phe Arg Gly His Gln Lys Ser Lys Gly Asn Ser1 5 10 15Tyr Asp
Val Glu Val Val Leu Gln His Val Asp Thr Gly Asn Ser Tyr 20 25 30Leu
Cys Gly Tyr Leu Lys Ile Lys Gly Leu Thr Glu Glu Tyr Pro Thr 35 40
45Leu Thr Thr Phe Phe Glu Gly Glu Ile Ile Ser Lys Lys His Pro Phe
50 55 60Leu Thr Arg Lys Trp Asp Ala Asp Glu Asp Val Asp Arg Lys His
Trp65 70 75 80Gly Lys Phe Leu Ala Phe Tyr Gln Tyr Ala Lys Ser Phe
Asn Ser Asp 85 90 95Asp Phe Asp Tyr Glu Glu Leu Lys Asn Gly Asp Tyr
Val Phe Met Arg 100 105 110Trp Lys Glu Gln Phe Leu Val Pro Asp His
Thr Ile Lys Asp Ile Ser 115 120 125Gly Ala Ser Phe Ala Gly Phe Tyr
Tyr Ile Cys Phe Gln Lys Ser Ala 130 135 140Ala Ser Ile Glu Gly Tyr
Tyr Tyr His Arg Ser Ser Glu Trp Tyr Gln145 150 155 160Ser Leu
Asn Leu Thr His Val 16520362PRTSaccharomyces cerevisiae 20Met Ile
Asn Asn Pro Lys Val Asp Ser Val Ala Glu Lys Pro Lys Ala1 5 10 15Val
Thr Ser Lys Gln Ser Glu Gln Ala Ala Ser Pro Glu Pro Thr Pro 20 25
30Ala Pro Pro Val Ser Arg Asn Gln Tyr Pro Ile Thr Phe Asn Leu Thr
35 40 45Ser Thr Ala Pro Phe His Leu His Asp Arg His Arg Tyr Leu Gln
Glu 50 55 60Gln Asp Leu Tyr Lys Cys Ala Ser Arg Asp Ser Leu Ser Ser
Leu Gln65 70 75 80Gln Leu Ala His Thr Pro Asn Gly Ser Thr Arg Lys
Lys Tyr Ile Val 85 90 95Glu Asp Gln Ser Pro Tyr Ser Ser Glu Asn Pro
Val Ile Val Thr Ser 100 105 110Ser Tyr Asn His Thr Val Cys Thr Asn
Tyr Leu Arg Pro Arg Met Gln 115 120 125Phe Thr Gly Tyr Gln Ile Ser
Gly Tyr Lys Arg Tyr Gln Val Thr Val 130 135 140Asn Leu Lys Thr Val
Asp Leu Pro Lys Lys Asp Cys Thr Ser Leu Ser145 150 155 160Pro His
Leu Ser Gly Phe Leu Ser Ile Arg Gly Leu Thr Asn Gln His 165 170
175Pro Glu Ile Ser Thr Tyr Phe Glu Ala Tyr Ala Val Asn His Lys Glu
180 185 190Leu Gly Phe Leu Ser Ser Ser Trp Lys Asp Glu Pro Val Leu
Asn Glu 195 200 205Phe Lys Ala Thr Asp Gln Thr Asp Leu Glu His Trp
Ile Asn Phe Pro 210 215 220Ser Phe Arg Gln Leu Phe Leu Met Ser Gln
Lys Asn Gly Leu Asn Ser225 230 235 240Thr Asp Asp Asn Gly Thr Thr
Asn Ala Ala Lys Lys Leu Pro Pro Gln 245 250 255Gln Leu Pro Thr Thr
Pro Ser Ala Asp Ala Gly Asn Ile Ser Arg Ile 260 265 270Phe Ser Gln
Glu Lys Gln Phe Asp Asn Tyr Leu Asn Glu Arg Phe Ile 275 280 285Phe
Met Lys Trp Lys Glu Lys Phe Leu Val Pro Asp Ala Leu Leu Met 290 295
300Glu Gly Val Asp Gly Ala Ser Tyr Asp Gly Phe Tyr Tyr Ile Val
His305 310 315 320Asp Gln Val Thr Gly Asn Ile Gln Gly Phe Tyr Tyr
His Gln Asp Ala 325 330 335Glu Lys Phe Gln Gln Leu Glu Leu Val Pro
Ser Leu Lys Asn Lys Val 340 345 350Glu Ser Ser Asp Cys Ser Phe Glu
Phe Ala 355 36021240PRTUnknownSingle-chain antibody variable
fragment (scFv) against phosphotyrosine 21Met Met Glu Val Gln Leu
Gln Gln Ser Gly Pro Glu Leu Val Lys Pro1 5 10 15Gly Ala Ser Val Met
Ile Ser Cys Arg Thr Ser Ala Tyr Thr Phe Thr 20 25 30Glu Asn Thr Val
His Trp Val Lys Gln Ser His Gly Glu Ser Leu Glu 35 40 45Trp Ile Gly
Gly Ile Asn Pro Tyr Tyr Gly Gly Ser Ile Phe Ser Pro 50 55 60Lys Phe
Lys Gly Lys Ala Thr Leu Thr Val Asp Lys Ser Ser Ser Thr65 70 75
80Ala Tyr Met Glu Leu Arg Ser Leu Thr Ser Glu Asp Ser Ala Val Tyr
85 90 95Tyr Cys Ala Arg Arg Ala Gly Ala Tyr Tyr Phe Asp Tyr Trp Gly
Gln 100 105 110Gly Thr Thr Leu Thr Val Ser Ser Gly Gly Gly Ser Gly
Gly Gly Ser 115 120 125Gly Gly Gly Ser Glu Asn Val Leu Thr Gln Ser
Pro Ala Ile Met Ser 130 135 140Ala Ser Pro Gly Glu Lys Val Thr Met
Thr Cys Arg Ala Ser Ser Ser145 150 155 160Val Ser Ser Ser Tyr Leu
His Trp Tyr Arg Gln Lys Ser Gly Ala Ser 165 170 175Pro Lys Leu Trp
Ile Tyr Ser Thr Ser Asn Leu Ala Ser Gly Val Pro 180 185 190Ala Arg
Phe Ser Gly Ser Gly Ser Gly Thr Ser Tyr Ser Leu Thr Ile 195 200
205Ser Ser Val Glu Ala Glu Asp Ala Ala Thr Tyr Tyr Cys Gln Gln Tyr
210 215 220Ser Gly Tyr Arg Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile
Lys Arg225 230 235 24022111PRTHomo sapiens 22Met Gly Ala Met Asp
Ser Ile Gln Ala Glu Glu Trp Tyr Phe Gly Lys1 5 10 15Leu Gly Arg Lys
Asp Ala Glu Arg Gln Leu Leu Ser Phe Gly Asn Pro 20 25 30Arg Gly Thr
Phe Leu Ile Arg Glu Ser Glu Thr Thr Lys Gly Ala Tyr 35 40 45Ser Leu
Ser Ile Arg Asp Trp Asp Asp Met Lys Gly Asp His Val Lys 50 55 60His
Tyr Lys Ile Arg Lys Leu Asp Asn Gly Gly Tyr Tyr Ile Thr Thr65 70 75
80Arg Ala Gln Phe Glu Thr Leu Gln Gln Leu Val Gln His Tyr Ser Glu
85 90 95Arg Ala Ala Gly Leu Ser Ser Arg Leu Val Val Pro Ser His Lys
100 105 11023111PRTHomo sapiens 23Met Gly Ala Met Asp Ser Ile Gln
Ala Glu Glu Trp Tyr Phe Gly Lys1 5 10 15Leu Gly Arg Lys Asp Ala Glu
Arg Gln Leu Leu Ser Phe Gly Asn Pro 20 25 30Arg Gly Thr Phe Leu Ile
Arg Glu Ser Glu Thr Val Lys Gly Ala Tyr 35 40 45Ala Leu Ser Ile Arg
Asp Trp Asp Asp Met Lys Gly Asp His Val Lys 50 55 60His Tyr Leu Ile
Arg Lys Leu Asp Asn Gly Gly Tyr Tyr Ile Thr Thr65 70 75 80Arg Ala
Gln Phe Glu Thr Leu Gln Gln Leu Val Gln His Tyr Ser Glu 85 90 95Arg
Ala Ala Gly Leu Ser Ser Arg Leu Val Val Pro Ser His Lys 100 105
11024113PRTHomo sapiens 24Met Gly Ala Met Asp Ser Ile Gln Ala Glu
Glu Trp Tyr Phe Gly Lys1 5 10 15Ile Thr Arg Arg Glu Ser Glu Arg Leu
Leu Leu Asn Ala Glu Asn Pro 20 25 30Arg Gly Thr Phe Leu Val Arg Glu
Ser Glu Thr Thr Lys Gly Ala Tyr 35 40 45Ser Leu Ser Val Ser Asp Phe
Asp Asn Ala Lys Gly Leu Asn Val Lys 50 55 60His Tyr Lys Ile Arg Lys
Leu Asp Ser Gly Gly Phe Tyr Ile Thr Ser65 70 75 80Arg Thr Gln Phe
Asn Ser Leu Gln Gln Leu Val Ala Tyr Tyr Ser Lys 85 90 95His Ala Asp
Gly Leu Cys His Arg Leu Thr Thr Val Cys Pro Thr Ser 100 105
110Lys25113PRTHomo sapiens 25Met Gly Ala Met Asp Ser Ile Gln Ala
Glu Glu Trp Tyr Phe Gly Lys1 5 10 15Ile Thr Arg Arg Glu Ser Glu Arg
Leu Leu Leu Asn Ala Glu Asn Pro 20 25 30Arg Gly Thr Phe Leu Val Arg
Glu Ser Glu Val Thr Lys Gly Ala Tyr 35 40 45Ala Leu Ser Val Ser Asp
Phe Asp Asn Ala Lys Gly Leu Asn Val Lys 50 55 60His Tyr Leu Ile Arg
Lys Leu Asp Ser Gly Gly Phe Tyr Ile Thr Ser65 70 75 80Arg Thr Gln
Phe Asn Ser Leu Gln Gln Leu Val Ala Tyr Tyr Ser Lys 85 90 95His Ala
Asp Gly Leu Cys His Arg Leu Thr Thr Val Cys Pro Thr Ser 100 105
110Lys26310PRTHomo sapiens 26Met Ala Ser Leu Thr Val Lys Ala Tyr
Leu Leu Gly Lys Glu Asp Ala1 5 10 15Ala Arg Glu Ile Arg Arg Phe Ser
Phe Cys Cys Ser Pro Glu Pro Glu 20 25 30Ala Glu Ala Glu Ala Ala Ala
Gly Pro Gly Pro Cys Glu Arg Leu Leu 35 40 45Ser Arg Val Ala Ala Leu
Phe Pro Ala Leu Arg Pro Gly Gly Phe Gln 50 55 60Ala His Tyr Arg Asp
Glu Asp Gly Asp Leu Val Ala Phe Ser Ser Asp65 70 75 80Glu Glu Leu
Thr Met Ala Met Ser Tyr Val Lys Asp Asp Ile Phe Arg 85 90 95Ile Tyr
Ile Lys Glu Lys Lys Glu Cys Arg Arg Asp His Arg Pro Pro 100 105
110Cys Ala Gln Glu Ala Pro Arg Asn Met Val His Pro Asn Val Ile Cys
115 120 125Asp Gly Cys Asn Gly Pro Val Val Gly Thr Arg Tyr Lys Cys
Ser Val 130 135 140Cys Pro Asp Tyr Asp Leu Cys Ser Val Cys Glu Gly
Lys Gly Leu His145 150 155 160Arg Gly His Thr Lys Leu Ala Phe Pro
Ser Pro Phe Gly His Leu Ser 165 170 175Glu Gly Phe Ser His Ser Arg
Trp Leu Arg Lys Val Lys His Gly His 180 185 190Phe Gly Trp Pro Gly
Trp Glu Met Gly Pro Pro Gly Asn Trp Ser Pro 195 200 205Arg Pro Pro
Arg Ala Gly Glu Ala Arg Pro Gly Pro Thr Ala Glu Ser 210 215 220Ala
Ser Gly Pro Ser Glu Asp Pro Ser Val Asn Phe Leu Lys Asn Val225 230
235 240Gly Glu Ser Val Ala Ala Ala Leu Ser Pro Leu Gly Ile Glu Val
Asp 245 250 255Ile Asp Val Glu His Gly Gly Lys Arg Ser Arg Leu Thr
Pro Val Ser 260 265 270Pro Glu Ser Ser Ser Thr Glu Glu Lys Ser Ser
Ser Gln Pro Ser Ser 275 280 285Cys Cys Ser Asp Pro Ser Lys Pro Gly
Gly Asn Val Glu Gly Ala Thr 290 295 300Gln Ser Leu Ala Glu Gln305
31027310PRTHomo sapiens 27Met Ala Ser Leu Thr Val Lys Ala Tyr Leu
Leu Gly Lys Glu Asp Ala1 5 10 15Ala Arg Glu Ile Arg Arg Phe Ser Phe
Cys Cys Ser Pro Glu Pro Glu 20 25 30Ala Glu Ala Glu Ala Ala Ala Gly
Pro Gly Pro Cys Glu Arg Leu Leu 35 40 45Ser Arg Val Ala Ala Leu Phe
Pro Ala Leu Arg Pro Gly Gly Phe Gln 50 55 60Ala His Tyr Arg Asp Glu
Asp Gly Asp Leu Val Ala Phe Ser Ser Asp65 70 75 80Glu Glu Leu Thr
Met Ala Met Ser Tyr Val Lys Asp Asp Ile Phe Arg 85 90 95Ile Tyr Ile
Lys Glu Lys Lys Glu Cys Arg Arg Asp His Arg Pro Pro 100 105 110Cys
Ala Gln Glu Ala Pro Arg Asn Met Val His Pro Asn Val Ile Cys 115 120
125Asp Gly Cys Asn Gly Pro Val Val Gly Thr Arg Tyr Lys Cys Ser Val
130 135 140Cys Pro Asp Tyr Asp Leu Cys Ser Val Cys Glu Gly Lys Gly
Leu His145 150 155 160Arg Gly His Thr Lys Leu Ala Phe Pro Ser Pro
Phe Gly His Leu Ser 165 170 175Glu Gly Phe Ser His Ser Arg Trp Leu
Arg Lys Val Lys His Gly His 180 185 190Phe Gly Trp Pro Gly Trp Glu
Met Gly Pro Pro Gly Asn Trp Ser Pro 195 200 205Arg Pro Pro Arg Ala
Gly Glu Ala Arg Pro Gly Pro Thr Ala Glu Ser 210 215 220Ala Ser Gly
Pro Ser Glu Asp Pro Ser Val Asn Phe Leu Lys Asn Val225 230 235
240Gly Glu Ser Val Ala Ala Ala Leu Ser Pro Leu Gly Ile Glu Val Asp
245 250 255Ile Asp Val Glu His Gly Gly Lys Arg Ser Arg Leu Thr Pro
Val Ser 260 265 270Pro Glu Ser Ser Ser Thr Glu Glu Lys Ser Ser Ser
Gln Pro Ser Ser 275 280 285Cys Cys Ser Asp Pro Ser Lys Pro Gly Gly
Asn Val Glu Gly Ala Thr 290 295 300Gln Ser Leu Ala Glu Gln305
31028310PRTHomo sapiens 28Met Ala Ser Leu Thr Val Lys Ala Tyr Leu
Leu Gly Lys Glu Asp Ala1 5 10 15Ala Arg Glu Ile Arg Arg Phe Ser Phe
Cys Cys Ser Pro Glu Pro Glu 20 25 30Ala Glu Ala Glu Ala Ala Ala Gly
Pro Gly Pro Cys Glu Arg Leu Leu 35 40 45Ser Arg Val Ala Ala Leu Phe
Pro Ala Leu Arg Pro Gly Gly Phe Gln 50 55 60Ala His Tyr Arg Asp Glu
Asp Gly Asp Leu Val Ala Phe Ser Ser Asp65 70 75 80Glu Glu Leu Thr
Met Ala Met Ser Tyr Val Lys Asp Asp Ile Phe Arg 85 90 95Ile Tyr Ile
Lys Glu Lys Lys Glu Cys Arg Arg Asp His Arg Pro Pro 100 105 110Cys
Ala Gln Glu Ala Pro Arg Asn Met Val His Pro Asn Val Ile Cys 115 120
125Asp Gly Cys Asn Gly Pro Val Val Gly Thr Arg Tyr Lys Cys Ser Val
130 135 140Cys Pro Asp Tyr Asp Leu Cys Ser Val Cys Glu Gly Lys Gly
Leu His145 150 155 160Arg Gly His Thr Lys Leu Ala Phe Pro Ser Pro
Phe Gly His Leu Ser 165 170 175Glu Gly Phe Ser His Ser Arg Trp Leu
Arg Lys Val Lys His Gly His 180 185 190Phe Gly Trp Pro Gly Trp Glu
Met Gly Pro Pro Gly Asn Trp Ser Pro 195 200 205Arg Pro Pro Arg Ala
Gly Glu Ala Arg Pro Gly Pro Thr Ala Glu Ser 210 215 220Ala Ser Gly
Pro Ser Glu Asp Pro Ser Val Asn Phe Leu Lys Asn Val225 230 235
240Gly Glu Ser Val Ala Ala Ala Leu Ser Pro Leu Gly Ile Glu Val Asp
245 250 255Ile Asp Val Glu His Gly Gly Lys Arg Ser Arg Leu Thr Pro
Val Ser 260 265 270Pro Glu Ser Ser Ser Thr Glu Glu Lys Ser Ser Ser
Gln Pro Ser Ser 275 280 285Cys Cys Ser Asp Pro Ser Lys Pro Gly Gly
Asn Val Glu Gly Ala Thr 290 295 300Gln Ser Leu Ala Glu Gln305
31029440PRTHomo sapiens 29Met Ala Ser Leu Thr Val Lys Ala Tyr Leu
Leu Gly Lys Glu Asp Ala1 5 10 15Ala Arg Glu Ile Arg Arg Phe Ser Phe
Cys Cys Ser Pro Glu Pro Glu 20 25 30Ala Glu Ala Glu Ala Ala Ala Gly
Pro Gly Pro Cys Glu Arg Leu Leu 35 40 45Ser Arg Val Ala Ala Leu Phe
Pro Ala Leu Arg Pro Gly Gly Phe Gln 50 55 60Ala His Tyr Arg Asp Glu
Asp Gly Asp Leu Val Ala Phe Ser Ser Asp65 70 75 80Glu Glu Leu Thr
Met Ala Met Ser Tyr Val Lys Asp Asp Ile Phe Arg 85 90 95Ile Tyr Ile
Lys Glu Lys Lys Glu Cys Arg Arg Asp His Arg Pro Pro 100 105 110Cys
Ala Gln Glu Ala Pro Arg Asn Met Val His Pro Asn Val Ile Cys 115 120
125Asp Gly Cys Asn Gly Pro Val Val Gly Thr Arg Tyr Lys Cys Ser Val
130 135 140Cys Pro Asp Tyr Asp Leu Cys Ser Val Cys Glu Gly Lys Gly
Leu His145 150 155 160Arg Gly His Thr Lys Leu Ala Phe Pro Ser Pro
Phe Gly His Leu Ser 165 170 175Glu Gly Phe Ser His Ser Arg Trp Leu
Arg Lys Val Lys His Gly His 180 185 190Phe Gly Trp Pro Gly Trp Glu
Met Gly Pro Pro Gly Asn Trp Ser Pro 195 200 205Arg Pro Pro Arg Ala
Gly Glu Ala Arg Pro Gly Pro Thr Ala Glu Ser 210 215 220Ala Ser Gly
Pro Ser Glu Asp Pro Ser Val Asn Phe Leu Lys Asn Val225 230 235
240Gly Glu Ser Val Ala Ala Ala Leu Ser Pro Leu Gly Ile Glu Val Asp
245 250 255Ile Asp Val Glu His Gly Gly Lys Arg Ser Arg Leu Thr Pro
Val Ser 260 265 270Pro Glu Ser Ser Ser Thr Glu Glu Lys Ser Ser Ser
Gln Pro Ser Ser 275 280 285Cys Cys Ser Asp Pro Ser Lys Pro Gly Gly
Asn Val Glu Gly Ala Thr 290 295 300Gln Ser Leu Ala Glu Gln Met Arg
Lys Ile Ala Leu Glu Ser Glu Gly305 310 315 320Arg Pro Glu Glu Gln
Met Glu Ser Asp Asn Cys Ser Gly Gly Asp Asp 325 330 335Asp Trp Thr
His Leu Ser Ser Lys Glu Val Asp Pro Ser Thr Gly Glu 340 345 350Leu
Gln Ser Leu Gln Met Pro Glu Ser Glu Gly Pro Ser Ser Leu Asp 355 360
365Pro Ser Gln Glu Gly Pro Thr Gly Leu Lys Glu Ala Ala Leu Tyr Pro
370 375 380His Leu Pro Pro Glu Ala Asp Pro Arg Leu Ile Glu Ser Leu
Ser Gln385 390 395 400Met Leu Ser Met Gly Phe Ser Asp Glu Gly Gly
Trp Leu Thr Arg Leu 405 410 415Leu Gln Thr Lys Asn Tyr Asp Ile Gly
Ala Ala Leu Asp Thr Ile Gln 420 425 430Tyr Ser Lys His Pro Pro Pro
Leu 435 44030439PRTRattus norvegicus 30Met Ala Ser Leu Thr Val Lys
Ala Tyr Leu Leu Gly Lys Glu Glu Ala1 5
10 15Ala Arg Glu Ile Arg Arg Phe Ser Phe Cys Phe Ser Pro Glu Pro
Glu 20 25 30Ala Glu Ala Ala Ala Gly Pro Gly Pro Cys Glu Arg Leu Leu
Ser Arg 35 40 45Val Ala Val Leu Phe Pro Ala Leu Arg Pro Gly Gly Phe
Gln Ala His 50 55 60Tyr Arg Asp Glu Asp Gly Asp Leu Val Ala Phe Ser
Ser Asp Glu Glu65 70 75 80Leu Thr Met Ala Met Ser Tyr Val Lys Asp
Asp Ile Phe Arg Ile Tyr 85 90 95Ile Lys Glu Lys Lys Glu Cys Arg Arg
Glu His Arg Pro Pro Cys Ala 100 105 110Gln Glu Ala Arg Ser Met Val
His Pro Asn Val Ile Cys Asp Gly Cys 115 120 125Asn Gly Pro Val Val
Gly Thr Arg Tyr Lys Cys Ser Val Cys Pro Asp 130 135 140Tyr Asp Leu
Cys Ser Val Cys Glu Gly Lys Gly Leu His Arg Glu His145 150 155
160Ser Lys Leu Ile Phe Pro Asn Pro Phe Gly His Leu Ser Asp Ser Phe
165 170 175Ser His Ser Arg Trp Leu Arg Lys Leu Lys His Gly His Phe
Gly Trp 180 185 190Pro Gly Trp Glu Met Gly Pro Pro Gly Asn Trp Ser
Pro Arg Pro Pro 195 200 205Arg Ala Gly Asp Gly Arg Pro Cys Pro Thr
Ala Glu Ser Ala Ser Ala 210 215 220Pro Ser Glu Asp Pro Asn Val Asn
Phe Leu Lys Asn Val Gly Glu Ser225 230 235 240Val Ala Ala Ala Leu
Ser Pro Leu Gly Ile Glu Val Asp Ile Asp Val 245 250 255Glu His Gly
Gly Lys Arg Ser Arg Leu Thr Pro Thr Ser Ala Glu Ser 260 265 270Ser
Ser Thr Gly Thr Glu Asp Lys Ser Gly Thr Gln Pro Ser Ser Cys 275 280
285Ser Ser Glu Val Ser Lys Pro Asp Gly Ala Gly Glu Gly Pro Ala Gln
290 295 300Ser Leu Thr Glu Gln Met Lys Lys Ile Ala Leu Glu Ser Val
Gly Gln305 310 315 320Pro Glu Glu Leu Met Glu Ser Asp Asn Cys Ser
Gly Gly Asp Asp Asp 325 330 335Trp Thr His Leu Ser Ser Lys Glu Val
Asp Pro Ser Thr Gly Glu Leu 340 345 350Gln Ser Leu Gln Met Pro Glu
Ser Glu Gly Pro Ser Ser Leu Asp Pro 355 360 365Ser Gln Glu Gly Pro
Thr Gly Leu Lys Glu Ala Ala Leu Tyr Pro His 370 375 380Leu Pro Pro
Glu Ala Asp Pro Arg Leu Ile Glu Ser Leu Ser Gln Met385 390 395
400Leu Ser Met Gly Phe Ser Asp Glu Gly Gly Trp Leu Thr Arg Leu Leu
405 410 415Gln Thr Lys Asn Tyr Asp Ile Gly Ala Ala Leu Asp Thr Ile
Gln Tyr 420 425 430Ser Lys His Pro Pro Pro Leu
43531292PRTSaccharomyces cerevisiae 31Met Thr Ser Leu Asn Ile Met
Gly Arg Lys Phe Ile Leu Glu Arg Ala1 5 10 15Lys Arg Asn Asp Asn Ile
Glu Glu Ile Tyr Thr Ser Ala Tyr Val Ser 20 25 30Leu Pro Ser Ser Thr
Asp Thr Arg Leu Pro His Phe Lys Ala Lys Glu 35 40 45Glu Asp Cys Asp
Val Tyr Glu Glu Gly Thr Asn Leu Val Gly Lys Asn 50 55 60Ala Lys Tyr
Thr Tyr Arg Ser Leu Gly Arg His Leu Asp Phe Leu Arg65 70 75 80Pro
Gly Leu Arg Phe Gly Gly Ser Gln Ser Ser Lys Tyr Thr Tyr Tyr 85 90
95Thr Val Glu Val Lys Ile Asp Thr Val Asn Leu Pro Leu Tyr Lys Asp
100 105 110Ser Arg Ser Leu Asp Pro His Val Thr Gly Thr Phe Thr Ile
Lys Asn 115 120 125Leu Thr Pro Val Leu Asp Lys Val Val Thr Leu Phe
Glu Gly Tyr Val 130 135 140Ile Asn Tyr Asn Gln Phe Pro Leu Cys Ser
Leu His Trp Pro Ala Glu145 150 155 160Glu Thr Leu Asp Pro Tyr Met
Ala Gln Arg Glu Ser Asp Cys Ser His 165 170 175Trp Lys Arg Phe Gly
His Phe Gly Ser Asp Asn Trp Ser Leu Thr Glu 180 185 190Arg Asn Phe
Gly Gln Tyr Asn His Glu Ser Ala Glu Phe Met Asn Gln 195 200 205Arg
Tyr Ile Tyr Leu Lys Trp Lys Glu Arg Phe Leu Leu Asp Asp Glu 210 215
220Glu Gln Glu Asn Gln Met Leu Asp Asp Asn His His Leu Glu Gly
Ala225 230 235 240Ser Phe Glu Gly Phe Tyr Tyr Val Cys Leu Asp Gln
Leu Thr Gly Ser 245 250 255Val Glu Gly Tyr Tyr Tyr His Pro Ala Cys
Glu Leu Phe Gln Lys Leu 260 265 270Glu Leu Val Pro Thr Asn Cys Asp
Ala Leu Asn Thr Tyr Ser Ser Gly 275 280 285Phe Glu Ile Ala
2903277PRTHomo sapiens 32Met Gly Pro Leu Gly Ser Leu Cys Gly Arg
Val Phe Lys Ser Gly Glu1 5 10 15Thr Thr Tyr Ser Cys Arg Asp Cys Ala
Ile Asp Pro Thr Cys Val Leu 20 25 30Cys Met Asp Cys Phe Gln Asp Ser
Val His Lys Asn His Arg Tyr Lys 35 40 45Met His Thr Ser Thr Gly Gly
Gly Phe Cys Asp Cys Gly Asp Thr Glu 50 55 60Ala Trp Lys Thr Gly Pro
Phe Cys Val Asn His Glu Pro65 70 753376PRTHomo sapiens 33Met Gly
Pro Leu Gly Ser Leu Cys Gly Arg Val Phe Lys Val Gly Glu1 5 10 15Pro
Thr Tyr Ser Cys Arg Asp Cys Ala Val Asp Pro Thr Cys Val Leu 20 25
30Cys Met Glu Cys Phe Leu Gly Ser Ile His Arg Asp His Arg Tyr Arg
35 40 45Met Thr Thr Ser Gly Gly Gly Gly Phe Cys Asp Cys Gly Asp Thr
Glu 50 55 60Ala Trp Lys Glu Gly Pro Tyr Cys Gln Lys His Glu65 70
7534421PRTLeishmania major 34Met Ser Arg Asn Pro Ser Asn Ser Asp
Ala Ala His Ala Phe Trp Ser1 5 10 15Thr Gln Pro Val Pro Gln Thr Glu
Asp Glu Thr Glu Lys Ile Val Phe 20 25 30Ala Gly Pro Met Asp Glu Pro
Lys Thr Val Ala Asp Ile Pro Glu Glu 35 40 45Pro Tyr Pro Ile Ala Ser
Thr Phe Glu Trp Trp Thr Pro Asn Met Glu 50 55 60Ala Ala Asp Asp Ile
His Ala Ile Tyr Glu Leu Leu Arg Asp Asn Tyr65 70 75 80Val Glu Asp
Asp Asp Ser Met Phe Arg Phe Asn Tyr Ser Glu Glu Phe 85 90 95Leu Gln
Trp Ala Leu Cys Pro Pro Asn Tyr Ile Pro Asp Trp His Val 100 105
110Ala Val Arg Arg Lys Ala Asp Lys Lys Leu Leu Ala Phe Ile Ala Gly
115 120 125Val Pro Val Thr Leu Arg Met Gly Thr Pro Lys Tyr Met Lys
Val Lys 130 135 140Ala Gln Glu Lys Gly Glu Gly Glu Glu Ala Ala Lys
Tyr Asp Glu Pro145 150 155 160Arg His Ile Cys Glu Ile Asn Phe Leu
Cys Val His Lys Gln Leu Arg 165 170 175Glu Lys Arg Leu Ala Pro Ile
Leu Ile Lys Glu Ala Thr Arg Arg Val 180 185 190Asn Arg Thr Asn Val
Trp Gln Ala Val Tyr Thr Ala Gly Val Leu Leu 195 200 205Pro Thr Pro
Tyr Ala Ser Gly Gln Tyr Phe His Arg Ser Leu Asn Pro 210 215 220Glu
Lys Leu Val Glu Ile Arg Phe Ser Gly Ile Pro Ala Gln Tyr Gln225 230
235 240Lys Phe Gln Asn Pro Met Ala Met Leu Lys Arg Asn Tyr Gln Leu
Pro 245 250 255Ser Ala Pro Lys Asn Ser Gly Leu Arg Glu Met Lys Pro
Ser Asp Val 260 265 270Pro Gln Val Arg Arg Ile Leu Met Asn Tyr Leu
Asp Ser Phe Asp Val 275 280 285Gly Pro Val Phe Ser Asp Ala Glu Ile
Ser His Tyr Leu Leu Pro Arg 290 295 300Asp Gly Val Val Phe Thr Tyr
Val Val Glu Asn Asp Lys Lys Val Thr305 310 315 320Asp Phe Phe Ser
Phe Tyr Arg Ile Pro Ser Thr Val Ile Gly Asn Ser 325 330 335Asn Tyr
Asn Leu Leu Asn Ala Ala Tyr Val His Tyr Tyr Ala Ala Thr 340 345
350Ser Ile Pro Leu His Gln Leu Ile Leu Asp Leu Leu Ile Val Ala His
355 360 365Ser Arg Gly Phe Asp Val Cys Asn Met Val Glu Ile Leu Asp
Asn Arg 370 375 380Ser Phe Val Glu Gln Leu Lys Phe Gly Ala Gly Asp
Gly His Leu Arg385 390 395 400Tyr Tyr Phe Tyr Asn Trp Ala Tyr Pro
Lys Ile Lys Pro Ser Gln Val 405 410 415Ala Leu Val Met Leu
42035496PRTHomo sapiens 35Met Ala Asp Glu Ser Glu Thr Ala Val Lys
Pro Pro Ala Pro Pro Leu1 5 10 15Pro Gln Met Met Glu Gly Asn Gly Asn
Gly His Glu His Cys Ser Asp 20 25 30Cys Glu Asn Glu Glu Asp Asn Ser
Tyr Asn Arg Gly Gly Leu Ser Pro 35 40 45Ala Asn Asp Thr Gly Ala Lys
Lys Lys Lys Lys Lys Gln Lys Lys Lys 50 55 60Lys Glu Lys Gly Ser Glu
Thr Asp Ser Ala Gln Asp Gln Pro Val Lys65 70 75 80Met Asn Ser Leu
Pro Ala Glu Arg Ile Gln Glu Ile Gln Lys Ala Ile 85 90 95Glu Leu Phe
Ser Val Gly Gln Gly Pro Ala Lys Thr Met Glu Glu Ala 100 105 110Ser
Lys Arg Ser Tyr Gln Phe Trp Asp Thr Gln Pro Val Pro Lys Leu 115 120
125Gly Glu Val Val Asn Thr His Gly Pro Val Glu Pro Asp Lys Asp Asn
130 135 140Ile Arg Gln Glu Pro Tyr Thr Leu Pro Gln Gly Phe Thr Trp
Asp Ala145 150 155 160Leu Asp Leu Gly Asp Arg Gly Val Leu Lys Glu
Leu Tyr Thr Leu Leu 165 170 175Asn Glu Asn Tyr Val Glu Asp Asp Asp
Asn Met Phe Arg Phe Asp Tyr 180 185 190Ser Pro Glu Phe Leu Leu Trp
Ala Leu Arg Pro Pro Gly Trp Leu Pro 195 200 205Gln Trp His Cys Gly
Val Arg Val Val Ser Ser Arg Lys Leu Val Gly 210 215 220Phe Ile Ser
Ala Ile Pro Ala Asn Ile His Ile Tyr Asp Thr Glu Lys225 230 235
240Lys Met Val Glu Ile Asn Phe Leu Cys Val His Lys Lys Leu Arg Ser
245 250 255Lys Arg Val Ala Pro Val Leu Ile Arg Glu Ile Thr Arg Arg
Val His 260 265 270Leu Glu Gly Ile Phe Gln Ala Val Tyr Thr Ala Gly
Val Val Leu Pro 275 280 285Lys Pro Val Gly Thr Cys Arg Tyr Trp His
Arg Ser Leu Asn Pro Arg 290 295 300Lys Leu Ile Glu Val Lys Phe Ser
His Leu Ser Arg Asn Met Thr Met305 310 315 320Gln Arg Thr Met Lys
Leu Tyr Arg Leu Pro Glu Thr Pro Lys Thr Ala 325 330 335Gly Leu Arg
Pro Met Glu Thr Lys Asp Ile Pro Val Val His Gln Leu 340 345 350Leu
Thr Arg Tyr Leu Lys Gln Phe His Leu Thr Pro Val Met Ser Gln 355 360
365Glu Glu Val Glu His Trp Phe Tyr Pro Gln Glu Asn Ile Ile Asp Thr
370 375 380Phe Val Val Glu Asn Ala Asn Gly Glu Val Thr Asp Phe Leu
Ser Phe385 390 395 400Tyr Thr Leu Pro Ser Thr Ile Met Asn His Pro
Thr His Lys Ser Leu 405 410 415Lys Ala Ala Tyr Ser Phe Tyr Asn Val
His Thr Gln Thr Pro Leu Leu 420 425 430Asp Leu Met Ser Asp Ala Leu
Val Leu Ala Lys Met Lys Gly Phe Asp 435 440 445Val Phe Asn Ala Leu
Asp Leu Met Glu Asn Lys Thr Phe Leu Glu Lys 450 455 460Leu Lys Phe
Gly Ile Gly Asp Gly Asn Leu Gln Tyr Tyr Leu Tyr Asn465 470 475
480Trp Lys Cys Pro Ser Met Gly Ala Glu Lys Val Gly Leu Val Leu Gln
485 490 49536127PRTDrosophila melanogaster 36Met Gly Asp Val Gln
Pro Glu Thr Cys Arg Pro Ser Ala Ala Ser Gly1 5 10 15Asn Tyr Phe Pro
Gln Tyr Pro Glu Tyr Ala Ile Glu Thr Ala Arg Leu 20 25 30Arg Thr Phe
Glu Ala Trp Pro Arg Asn Leu Lys Gln Lys Pro His Gln 35 40 45Leu Ala
Glu Ala Gly Phe Phe Tyr Thr Gly Val Gly Asp Arg Val Arg 50 55 60Cys
Phe Ser Cys Gly Gly Gly Leu Met Asp Trp Asn Asp Asn Asp Glu65 70 75
80Pro Trp Glu Gln His Ala Leu Trp Leu Ser Gln Cys Arg Phe Val Lys
85 90 95Leu Met Lys Gly Gln Leu Tyr Ile Asp Thr Val Ala Ala Lys Pro
Val 100 105 110Leu Ala Glu Glu Lys Glu Glu Ser Thr Ser Ile Gly Gly
Asp Thr 115 120 1253727PRTArtificial SequenceSynthetic Polypeptide
37Gly Gly Gly Ser Gly Gly Gly Ser Gly Gly Gly Ser Gly Leu Asn Asp1
5 10 15Phe Phe Glu Ala Gln Lys Ile Glu Trp His Glu 20
253854PRTArtificial SequenceSynthetic Polypeptide 38Gly Gly Gly Ser
Gly Gly Gly Ser Gly Gly Gly Ser Gly Leu Asn Asp1 5 10 15Phe Phe Glu
Ala Gln Lys Ile Glu Trp His Glu Gly Gly Gly Ser Gly 20 25 30Gly Gly
Ser Gly Gly Gly Ser Gly Leu Asn Asp Phe Phe Glu Ala Gln 35 40 45Lys
Ile Glu Trp His Glu 503956PRTArtificial SequenceSynthetic
Polypeptide 39Gly Ser Gly Gly Gly Ser Gly Gly Gly Ser Gly Gly Gly
Ser Gly Leu1 5 10 15Asn Asp Phe Phe Glu Ala Gln Lys Ile Glu Trp His
Glu Gly Gly Gly 20 25 30Ser Gly Gly Gly Ser Gly Gly Gly Ser Gly Leu
Asn Asp Phe Phe Glu 35 40 45Ala Gln Lys Ile Glu Trp His Glu 50
554038PRTArtificial SequenceSynthetic Polypeptide 40Gly His His His
His His His His His His His Gly Gly Gly Ser Gly1 5 10 15Gly Gly Ser
Gly Gly Gly Ser Gly Leu Asn Asp Phe Phe Glu Ala Gln 20 25 30Lys Ile
Glu Trp His Glu 354165PRTArtificial SequenceSynthetic Polypeptide
41Gly His His His His His His His His His His Gly Gly Gly Ser Gly1
5 10 15Gly Gly Ser Gly Gly Gly Ser Gly Leu Asn Asp Phe Phe Glu Ala
Gln 20 25 30Lys Ile Glu Trp His Glu Gly Gly Gly Ser Gly Gly Gly Ser
Gly Gly 35 40 45Gly Ser Gly Leu Asn Asp Phe Phe Glu Ala Gln Lys Ile
Glu Trp His 50 55 60Glu654267PRTArtificial SequenceSynthetic
Polypeptide 42Gly Gly Ser His His His His His His His His His His
Gly Gly Gly1 5 10 15Ser Gly Gly Gly Ser Gly Gly Gly Ser Gly Leu Asn
Asp Phe Phe Glu 20 25 30Ala Gln Lys Ile Glu Trp His Glu Gly Gly Gly
Ser Gly Gly Gly Ser 35 40 45Gly Gly Gly Ser Gly Leu Asn Asp Phe Phe
Glu Ala Gln Lys Ile Glu 50 55 60Trp His Glu654366PRTArtificial
SequenceSynthetic Polypeptide 43Gly Ser His His His His His His His
His His His Gly Gly Gly Ser1 5 10 15Gly Gly Gly Ser Gly Gly Gly Ser
Gly Leu Asn Asp Phe Phe Glu Ala 20 25 30Gln Lys Ile Glu Trp His Glu
Gly Gly Gly Ser Gly Gly Gly Ser Gly 35 40 45Gly Gly Ser Gly Leu Asn
Asp Phe Phe Glu Ala Gln Lys Ile Glu Trp 50 55 60His
Glu654461PRTArtificial SequenceSynthetic Polypeptide 44Gly Gly Gly
Ser Gly Gly Gly Ser Gly Gly Gly Ser Gly Leu Asn Asp1 5 10 15Phe Phe
Glu Ala Gln Lys Ile Glu Trp His Glu Gly Gly Gly Ser Gly 20 25 30Gly
Gly Ser Gly Gly Gly Ser Gly Leu Asn Asp Phe Phe Glu Ala Gln 35 40
45Lys Ile Glu Trp His Glu Gly His His His His His His 50 55
6045901PRTL. pneumophila 45Met Met Val Lys Gln Gly Val Phe Met Lys
Thr Asp Gln Ser Lys Val1 5 10 15Lys Lys Leu Ser Asp Tyr Lys Ser Leu
Asp Tyr Phe Val Ile His Val 20 25 30Asp Leu Gln Ile Asp Leu Ser Lys
Lys Pro Val Glu Ser Lys Ala Arg 35 40 45Leu Thr Val Val Pro Asn Leu
Asn Val Asp Ser His Ser Asn Asp
Leu 50 55 60Val Leu Asp Gly Glu Asn Met Thr Leu Val Ser Leu Gln Met
Asn Asp65 70 75 80Asn Leu Leu Lys Glu Asn Glu Tyr Glu Leu Thr Lys
Asp Ser Leu Ile 85 90 95Ile Lys Asn Ile Pro Gln Asn Thr Pro Phe Thr
Ile Glu Met Thr Ser 100 105 110Leu Leu Gly Glu Asn Thr Asp Leu Phe
Gly Leu Tyr Glu Thr Glu Gly 115 120 125Val Ala Leu Val Lys Ala Glu
Ser Glu Gly Leu Arg Arg Val Phe Tyr 130 135 140Leu Pro Asp Arg Pro
Asp Asn Leu Ala Thr Tyr Lys Thr Thr Ile Ile145 150 155 160Ala Asn
Gln Glu Asp Tyr Pro Val Leu Leu Ser Asn Gly Val Leu Ile 165 170
175Glu Lys Lys Glu Leu Pro Leu Gly Leu His Ser Val Thr Trp Leu Asp
180 185 190Asp Val Pro Lys Pro Ser Tyr Leu Phe Ala Leu Val Ala Gly
Asn Leu 195 200 205Gln Arg Ser Val Thr Tyr Tyr Gln Thr Lys Ser Gly
Arg Glu Leu Pro 210 215 220Ile Glu Phe Tyr Val Pro Pro Ser Ala Thr
Ser Lys Cys Asp Phe Ala225 230 235 240Lys Glu Val Leu Lys Glu Ala
Met Ala Trp Asp Glu Arg Thr Phe Asn 245 250 255Leu Glu Cys Ala Leu
Arg Gln His Met Val Ala Gly Val Asp Lys Tyr 260 265 270Ala Ser Gly
Ala Ser Glu Pro Thr Gly Leu Asn Leu Phe Asn Thr Glu 275 280 285Asn
Leu Phe Ala Ser Pro Glu Thr Lys Thr Asp Leu Gly Ile Leu Arg 290 295
300Val Leu Glu Val Val Ala His Glu Phe Phe His Tyr Trp Ser Gly
Asp305 310 315 320Arg Val Thr Ile Arg Asp Trp Phe Asn Leu Pro Leu
Lys Glu Gly Leu 325 330 335Thr Thr Phe Arg Ala Ala Met Phe Arg Glu
Glu Leu Phe Gly Thr Asp 340 345 350Leu Ile Arg Leu Leu Asp Gly Lys
Asn Leu Asp Glu Arg Ala Pro Arg 355 360 365Gln Ser Ala Tyr Thr Ala
Val Arg Ser Leu Tyr Thr Ala Ala Ala Tyr 370 375 380Glu Lys Ser Ala
Asp Ile Phe Arg Met Met Met Leu Phe Ile Gly Lys385 390 395 400Glu
Pro Phe Ile Glu Ala Val Ala Lys Phe Phe Lys Asp Asn Asp Gly 405 410
415Gly Ala Val Thr Leu Glu Asp Phe Ile Glu Ser Ile Ser Asn Ser Ser
420 425 430Gly Lys Asp Leu Arg Ser Phe Leu Ser Trp Phe Thr Glu Ser
Gly Ile 435 440 445Pro Glu Leu Ile Val Thr Asp Glu Leu Asn Pro Asp
Thr Lys Gln Tyr 450 455 460Phe Leu Lys Ile Lys Thr Val Asn Gly Arg
Asn Arg Pro Ile Pro Ile465 470 475 480Leu Met Gly Leu Leu Asp Ser
Ser Gly Ala Glu Ile Val Ala Asp Lys 485 490 495Leu Leu Ile Val Asp
Gln Glu Glu Ile Glu Phe Gln Phe Glu Asn Ile 500 505 510Gln Thr Arg
Pro Ile Pro Ser Leu Leu Arg Ser Phe Ser Ala Pro Val 515 520 525His
Met Lys Tyr Glu Tyr Ser Tyr Gln Asp Leu Leu Leu Leu Met Gln 530 535
540Phe Asp Thr Asn Leu Tyr Asn Arg Cys Glu Ala Ala Lys Gln Leu
Ile545 550 555 560Ser Ala Leu Ile Asn Asp Phe Cys Ile Gly Lys Lys
Ile Glu Leu Ser 565 570 575Pro Gln Phe Phe Ala Val Tyr Lys Ala Leu
Leu Ser Asp Asn Ser Leu 580 585 590Asn Glu Trp Met Leu Ala Glu Leu
Ile Thr Leu Pro Ser Leu Glu Glu 595 600 605Leu Ile Glu Asn Gln Asp
Lys Pro Asp Phe Glu Lys Leu Asn Glu Gly 610 615 620Arg Gln Leu Ile
Gln Asn Ala Leu Ala Asn Glu Leu Lys Thr Asp Phe625 630 635 640Tyr
Asn Leu Leu Phe Arg Ile Gln Ile Ser Gly Asp Asp Asp Lys Gln 645 650
655Lys Leu Lys Gly Phe Asp Leu Lys Gln Ala Gly Leu Arg Arg Leu Lys
660 665 670Ser Val Cys Phe Ser Tyr Leu Leu Asn Val Asp Phe Glu Lys
Thr Lys 675 680 685Glu Lys Leu Ile Leu Gln Phe Glu Asp Ala Leu Gly
Lys Asn Met Thr 690 695 700Glu Thr Ala Leu Ala Leu Ser Met Leu Cys
Glu Ile Asn Cys Glu Glu705 710 715 720Ala Asp Val Ala Leu Glu Asp
Tyr Tyr His Tyr Trp Lys Asn Asp Pro 725 730 735Gly Ala Val Asn Asn
Trp Phe Ser Ile Gln Ala Leu Ala His Ser Pro 740 745 750Asp Val Ile
Glu Arg Val Lys Lys Leu Met Arg His Gly Asp Phe Asp 755 760 765Leu
Ser Asn Pro Asn Lys Val Tyr Ala Leu Leu Gly Ser Phe Ile Lys 770 775
780Asn Pro Phe Gly Phe His Ser Val Thr Gly Glu Gly Tyr Gln Leu
Val785 790 795 800Ala Asp Ala Ile Phe Asp Leu Asp Lys Ile Asn Pro
Thr Leu Ala Ala 805 810 815Asn Leu Thr Glu Lys Phe Thr Tyr Trp Asp
Lys Tyr Asp Val Asn Arg 820 825 830Gln Ala Met Met Ile Ser Thr Leu
Lys Ile Ile Tyr Ser Asn Ala Thr 835 840 845Ser Ser Asp Val Arg Thr
Met Ala Lys Lys Gly Leu Asp Lys Val Lys 850 855 860Glu Asp Leu Pro
Leu Pro Ile His Leu Thr Phe His Gly Gly Ser Thr865 870 875 880Met
Gln Asp Arg Thr Ala Gln Leu Ile Ala Asp Gly Asn Lys Glu Asn 885 890
895Ala Tyr Gln Leu His 90046265PRTE. coli 46Met Gly Thr Ala Ile Ser
Ile Lys Thr Pro Glu Asp Ile Glu Lys Met1 5 10 15Arg Val Ala Gly Arg
Leu Ala Ala Glu Val Leu Glu Met Ile Glu Pro 20 25 30Tyr Val Lys Pro
Gly Val Ser Thr Gly Glu Leu Asp Arg Ile Cys Asn 35 40 45Asp Tyr Ile
Val Asn Glu Gln His Ala Val Ser Ala Cys Leu Gly Tyr 50 55 60His Gly
Tyr Pro Lys Ser Val Cys Ile Ser Ile Asn Glu Val Val Cys65 70 75
80His Gly Ile Pro Asp Asp Ala Lys Leu Leu Lys Asp Gly Asp Ile Val
85 90 95Asn Ile Asp Val Thr Val Ile Lys Asp Gly Phe His Gly Asp Thr
Ser 100 105 110Lys Met Phe Ile Val Gly Lys Pro Thr Ile Met Gly Glu
Arg Leu Cys 115 120 125Arg Ile Thr Gln Glu Ser Leu Tyr Leu Ala Leu
Arg Met Val Lys Pro 130 135 140Gly Ile Asn Leu Arg Glu Ile Gly Ala
Ala Ile Gln Lys Phe Val Glu145 150 155 160Ala Glu Gly Phe Ser Val
Val Arg Glu Tyr Cys Gly His Gly Ile Gly 165 170 175Arg Gly Phe His
Glu Glu Pro Gln Val Leu His Tyr Asp Ser Arg Glu 180 185 190Thr Asn
Val Val Leu Lys Pro Gly Met Thr Phe Thr Ile Glu Pro Met 195 200
205Val Asn Ala Gly Lys Lys Glu Ile Arg Thr Met Lys Asp Gly Trp Thr
210 215 220Val Lys Thr Lys Asp Arg Ser Leu Ser Ala Gln Tyr Glu His
Thr Ile225 230 235 240Val Val Thr Asp Asn Gly Cys Glu Ile Leu Thr
Leu Arg Lys Asp Asp 245 250 255Thr Ile Pro Ala Ile Ile Ser His Asp
260 26547322PRTM. smegmatis 47Met Gly Thr Leu Glu Ala Asn Thr Asn
Gly Pro Gly Ser Met Leu Ser1 5 10 15Arg Met Pro Val Ser Ser Arg Thr
Val Pro Phe Gly Asp His Glu Thr 20 25 30Trp Val Gln Val Thr Thr Pro
Glu Asn Ala Gln Pro His Ala Leu Pro 35 40 45Leu Ile Val Leu His Gly
Gly Pro Gly Met Ala His Asn Tyr Val Ala 50 55 60Asn Ile Ala Ala Leu
Ala Asp Glu Thr Gly Arg Thr Val Ile His Tyr65 70 75 80Asp Gln Val
Gly Cys Gly Asn Ser Thr His Leu Pro Asp Ala Pro Ala 85 90 95Asp Phe
Trp Thr Pro Gln Leu Phe Val Asp Glu Phe His Ala Val Cys 100 105
110Thr Ala Leu Gly Ile Glu Arg Tyr His Val Leu Gly Gln Ser Trp Gly
115 120 125Gly Met Leu Gly Ala Glu Ile Ala Val Arg Gln Pro Ser Gly
Leu Val 130 135 140Ser Leu Ala Ile Cys Asn Ser Pro Ala Ser Met Arg
Leu Trp Ser Glu145 150 155 160Ala Ala Gly Asp Leu Arg Ala Gln Leu
Pro Ala Glu Thr Arg Ala Ala 165 170 175Leu Asp Arg His Glu Ala Ala
Gly Thr Ile Thr His Pro Asp Tyr Leu 180 185 190Gln Ala Ala Ala Glu
Phe Tyr Arg Arg His Val Cys Arg Val Val Pro 195 200 205Thr Pro Gln
Asp Phe Ala Asp Ser Val Ala Gln Met Glu Ala Glu Pro 210 215 220Thr
Val Tyr His Thr Met Asn Gly Pro Asn Glu Phe His Val Val Gly225 230
235 240Thr Leu Gly Asp Trp Ser Val Ile Asp Arg Leu Pro Asp Val Thr
Ala 245 250 255Pro Val Leu Val Ile Ala Gly Glu His Asp Glu Ala Thr
Pro Lys Thr 260 265 270Trp Gln Pro Phe Val Asp His Ile Pro Asp Val
Arg Ser His Val Phe 275 280 285Pro Gly Thr Ser His Cys Thr His Leu
Glu Lys Pro Glu Glu Phe Arg 290 295 300Ala Val Val Ala Gln Phe Leu
His Gln His Asp Leu Ala Ala Asp Ala305 310 315 320Arg Val48446PRTY.
pestis 48Met Thr Gln Gln Glu Tyr Gln Asn Arg Arg Gln Ala Leu Leu
Ala Lys1 5 10 15Met Ala Pro Gly Ser Ala Ala Ile Ile Phe Ala Ala Pro
Glu Ala Thr 20 25 30Arg Ser Ala Asp Ser Glu Tyr Pro Tyr Arg Gln Asn
Ser Asp Phe Ser 35 40 45Tyr Leu Thr Gly Phe Asn Glu Pro Glu Ala Val
Leu Ile Leu Val Lys 50 55 60Ser Asp Glu Thr His Asn His Ser Val Leu
Phe Asn Arg Ile Arg Asp65 70 75 80Leu Thr Ala Glu Ile Trp Phe Gly
Arg Arg Leu Gly Gln Glu Ala Ala 85 90 95Pro Thr Lys Leu Ala Val Asp
Arg Ala Leu Pro Phe Asp Glu Ile Asn 100 105 110Glu Gln Leu Tyr Leu
Leu Leu Asn Arg Leu Asp Val Ile Tyr His Ala 115 120 125Gln Gly Gln
Tyr Ala Tyr Ala Asp Asn Ile Val Phe Ala Ala Leu Glu 130 135 140Lys
Leu Arg His Gly Phe Arg Lys Asn Leu Arg Ala Pro Ala Thr Leu145 150
155 160Thr Asp Trp Arg Pro Trp Leu His Glu Met Arg Leu Phe Lys Ser
Ala 165 170 175Glu Glu Ile Ala Val Leu Arg Arg Ala Gly Glu Ile Ser
Ala Leu Ala 180 185 190His Thr Arg Ala Met Glu Lys Cys Arg Pro Gly
Met Phe Glu Tyr Gln 195 200 205Leu Glu Gly Glu Ile Leu His Glu Phe
Thr Arg His Gly Ala Arg Tyr 210 215 220Pro Ala Tyr Asn Thr Ile Val
Gly Gly Gly Glu Asn Gly Cys Ile Leu225 230 235 240His Tyr Thr Glu
Asn Glu Cys Glu Leu Arg Asp Gly Asp Leu Val Leu 245 250 255Ile Asp
Ala Gly Cys Glu Tyr Arg Gly Tyr Ala Gly Asp Ile Thr Arg 260 265
270Thr Phe Pro Val Asn Gly Lys Phe Thr Pro Ala Gln Arg Ala Val Tyr
275 280 285Asp Ile Val Leu Ala Ala Ile Asn Lys Ser Leu Thr Leu Phe
Arg Pro 290 295 300Gly Thr Ser Ile Arg Glu Val Thr Glu Glu Val Val
Arg Ile Met Val305 310 315 320Val Gly Leu Val Glu Leu Gly Ile Leu
Lys Gly Asp Ile Glu Gln Leu 325 330 335Ile Ala Glu Gln Ala His Arg
Pro Phe Phe Met His Gly Leu Ser His 340 345 350Trp Leu Gly Met Asp
Val His Asp Val Gly Asp Tyr Gly Ser Ser Asp 355 360 365Arg Gly Arg
Ile Leu Glu Pro Gly Met Val Leu Thr Val Glu Pro Gly 370 375 380Leu
Tyr Ile Ala Pro Asp Ala Asp Val Pro Pro Gln Tyr Arg Gly Ile385 390
395 400Gly Ile Arg Ile Glu Asp Asp Ile Val Ile Thr Ala Thr Gly Asn
Glu 405 410 415Asn Leu Thr Ala Ser Val Val Lys Asp Pro Asp Asp Ile
Glu Ala Leu 420 425 430Met Ala Leu Asn His Ala Gly Glu Asn Leu Tyr
Phe Gln Glu 435 440 44549303PRTP. furiosus 49Met Asp Thr Glu Lys
Leu Met Lys Ala Gly Glu Ile Ala Lys Lys Val1 5 10 15Arg Glu Lys Ala
Ile Lys Leu Ala Arg Pro Gly Met Leu Leu Leu Glu 20 25 30Leu Ala Glu
Ser Ile Glu Lys Met Ile Met Glu Leu Gly Gly Lys Pro 35 40 45Ala Phe
Pro Val Asn Leu Ser Ile Asn Glu Ile Ala Ala His Tyr Thr 50 55 60Pro
Tyr Lys Gly Asp Thr Thr Val Leu Lys Glu Gly Asp Tyr Leu Lys65 70 75
80Ile Asp Val Gly Val His Ile Asp Gly Phe Ile Ala Asp Thr Ala Val
85 90 95Thr Val Arg Val Gly Met Glu Glu Asp Glu Leu Met Glu Ala Ala
Lys 100 105 110Glu Ala Leu Asn Ala Ala Ile Ser Val Ala Arg Ala Gly
Val Glu Ile 115 120 125Lys Glu Leu Gly Lys Ala Ile Glu Asn Glu Ile
Arg Lys Arg Gly Phe 130 135 140Lys Pro Ile Val Asn Leu Ser Gly His
Lys Ile Glu Arg Tyr Lys Leu145 150 155 160His Ala Gly Ile Ser Ile
Pro Asn Ile Tyr Arg Pro His Asp Asn Tyr 165 170 175Val Leu Lys Glu
Gly Asp Val Phe Ala Ile Glu Pro Phe Ala Thr Ile 180 185 190Gly Ala
Gly Gln Val Ile Glu Val Pro Pro Thr Leu Ile Tyr Met Tyr 195 200
205Val Arg Asp Val Pro Val Arg Val Ala Gln Ala Arg Phe Leu Leu Ala
210 215 220Lys Ile Lys Arg Glu Tyr Gly Thr Leu Pro Phe Ala Tyr Arg
Trp Leu225 230 235 240Gln Asn Asp Met Pro Glu Gly Gln Leu Lys Leu
Ala Leu Lys Thr Leu 245 250 255Glu Lys Ala Gly Ala Ile Tyr Gly Tyr
Pro Val Leu Lys Glu Ile Arg 260 265 270Asn Gly Ile Val Ala Gln Phe
Glu His Thr Ile Ile Val Glu Lys Asp 275 280 285Ser Val Ile Val Thr
Gln Asp Met Ile Asn Lys Ser Thr Leu Glu 290 295
30050428PRTAeromonas sobria 50His Met Ser Ser Pro Leu His Tyr Val
Leu Asp Gly Ile His Cys Glu1 5 10 15Pro His Phe Phe Thr Val Pro Leu
Asp His Gln Gln Pro Asp Asp Glu 20 25 30Glu Thr Ile Thr Leu Phe Gly
Arg Thr Leu Cys Arg Lys Asp Arg Leu 35 40 45Asp Asp Glu Leu Pro Trp
Leu Leu Tyr Leu Gln Gly Gly Pro Gly Phe 50 55 60Gly Ala Pro Arg Pro
Ser Ala Asn Gly Gly Trp Ile Lys Arg Ala Leu65 70 75 80Gln Glu Phe
Arg Val Leu Leu Leu Asp Gln Arg Gly Thr Gly His Ser 85 90 95Thr Pro
Ile His Ala Glu Leu Leu Ala His Leu Asn Pro Arg Gln Gln 100 105
110Ala Asp Tyr Leu Ser His Phe Arg Ala Asp Ser Ile Val Arg Asp Ala
115 120 125Glu Leu Ile Arg Glu Gln Leu Ser Pro Asp His Pro Trp Ser
Leu Leu 130 135 140Gly Gln Ser Phe Gly Gly Phe Cys Ser Leu Thr Tyr
Leu Ser Leu Phe145 150 155 160Pro Asp Ser Leu His Glu Val Tyr Leu
Thr Gly Gly Val Ala Pro Ile 165 170 175Gly Arg Ser Ala Asp Glu Val
Tyr Arg Ala Thr Tyr Gln Arg Val Ala 180 185 190Asp Lys Asn Arg Ala
Phe Phe Ala Arg Phe Pro His Ala Gln Ala Ile 195 200 205Ala Asn Arg
Leu Ala Thr His Leu Gln Arg His Asp Val Arg Leu Pro 210 215 220Asn
Gly Gln Arg Leu Thr Val Glu Gln Leu Gln Gln Gln Gly Leu Asp225 230
235 240Leu Gly Ala Ser Gly Ala Phe Glu Glu Leu Tyr Tyr Leu Leu Glu
Asp 245 250 255Ala Phe Ile Gly Glu Lys Leu Asn Pro Ala Phe Leu Tyr
Gln Val Gln 260 265 270Ala Met Gln Pro Phe Asn Thr Asn Pro Val Phe
Ala Ile Leu His Glu
275 280 285Leu Ile Tyr Cys Glu Gly Ala Ala Ser His Trp Ala Ala Glu
Arg Val 290 295 300Arg Gly Glu Phe Pro Ala Leu Ala Trp Ala Gln Gly
Lys Asp Phe Ala305 310 315 320Phe Thr Gly Glu Met Ile Phe Pro Trp
Met Phe Glu Gln Phe Arg Glu 325 330 335Leu Ile Pro Leu Lys Glu Ala
Ala His Leu Leu Ala Glu Lys Ala Asp 340 345 350Trp Gly Pro Leu Tyr
Asp Pro Val Gln Leu Ala Arg Asn Lys Val Pro 355 360 365Val Ala Cys
Ala Val Tyr Ala Glu Asp Met Tyr Val Glu Phe Asp Tyr 370 375 380Ser
Arg Glu Thr Leu Lys Gly Leu Ser Asn Ser Arg Ala Trp Ile Thr385 390
395 400Asn Glu Tyr Glu His Asn Gly Leu Arg Val Asp Gly Glu Gln Ile
Leu 405 410 415Asp Arg Leu Ile Arg Leu Asn Arg Asp Cys Leu Glu 420
42551348PRTPyrococcus furiosus 51Met Lys Glu Arg Leu Glu Lys Leu
Val Lys Phe Met Asp Glu Asn Ser1 5 10 15Ile Asp Arg Val Phe Ile Ala
Lys Pro Val Asn Val Tyr Tyr Phe Ser 20 25 30Gly Thr Ser Pro Leu Gly
Gly Gly Tyr Ile Ile Val Asp Gly Asp Glu 35 40 45Ala Thr Leu Tyr Val
Pro Glu Leu Glu Tyr Glu Met Ala Lys Glu Glu 50 55 60Ser Lys Leu Pro
Val Val Lys Phe Lys Lys Phe Asp Glu Ile Tyr Glu65 70 75 80Ile Leu
Lys Asn Thr Glu Thr Leu Gly Ile Glu Gly Thr Leu Ser Tyr 85 90 95Ser
Met Val Glu Asn Phe Lys Glu Lys Ser Asn Val Lys Glu Phe Lys 100 105
110Lys Ile Asp Asp Val Ile Lys Asp Leu Arg Ile Ile Lys Thr Lys Glu
115 120 125Glu Ile Glu Ile Ile Glu Lys Ala Cys Glu Ile Ala Asp Lys
Ala Val 130 135 140Met Ala Ala Ile Glu Glu Ile Thr Glu Gly Lys Arg
Glu Arg Glu Val145 150 155 160Ala Ala Lys Val Glu Tyr Leu Met Lys
Met Asn Gly Ala Glu Lys Pro 165 170 175Ala Phe Asp Thr Ile Ile Ala
Ser Gly His Arg Ser Ala Leu Pro His 180 185 190Gly Val Ala Ser Asp
Lys Arg Ile Glu Arg Gly Asp Leu Val Val Ile 195 200 205Asp Leu Gly
Ala Leu Tyr Asn His Tyr Asn Ser Asp Ile Thr Arg Thr 210 215 220Ile
Val Val Gly Ser Pro Asn Glu Lys Gln Arg Glu Ile Tyr Glu Ile225 230
235 240Val Leu Glu Ala Gln Lys Arg Ala Val Glu Ala Ala Lys Pro Gly
Met 245 250 255Thr Ala Lys Glu Leu Asp Ser Ile Ala Arg Glu Ile Ile
Lys Glu Tyr 260 265 270Gly Tyr Gly Asp Tyr Phe Ile His Ser Leu Gly
His Gly Val Gly Leu 275 280 285Glu Ile His Glu Trp Pro Arg Ile Ser
Gln Tyr Asp Glu Thr Val Leu 290 295 300Lys Glu Gly Met Val Ile Thr
Ile Glu Pro Gly Ile Tyr Ile Pro Lys305 310 315 320Leu Gly Gly Val
Arg Ile Glu Asp Thr Val Leu Ile Thr Glu Asn Gly 325 330 335Ala Lys
Arg Leu Thr Lys Thr Glu Arg Glu Leu Leu 340
34552298PRTElizabethkingia meningoseptica 52Met Ile Pro Ile Thr Thr
Pro Val Gly Asn Phe Lys Val Trp Thr Lys1 5 10 15Arg Phe Gly Thr Asn
Pro Lys Ile Lys Val Leu Leu Leu His Gly Gly 20 25 30Pro Ala Met Thr
His Glu Tyr Met Glu Cys Phe Glu Thr Phe Phe Gln 35 40 45Arg Glu Gly
Phe Glu Phe Tyr Glu Tyr Asp Gln Leu Gly Ser Tyr Tyr 50 55 60Ser Asp
Gln Pro Thr Asp Glu Lys Leu Trp Asn Ile Asp Arg Phe Val65 70 75
80Asp Glu Val Glu Gln Val Arg Lys Ala Ile His Ala Asp Lys Glu Asn
85 90 95Phe Tyr Val Leu Gly Asn Ser Trp Gly Gly Ile Leu Ala Met Glu
Tyr 100 105 110Ala Leu Lys Tyr Gln Gln Asn Leu Lys Gly Leu Ile Val
Ala Asn Met 115 120 125Met Ala Ser Ala Pro Glu Tyr Val Lys Tyr Ala
Glu Val Leu Ser Lys 130 135 140Gln Met Lys Pro Glu Val Leu Ala Glu
Val Arg Ala Ile Glu Ala Lys145 150 155 160Lys Asp Tyr Ala Asn Pro
Arg Tyr Thr Glu Leu Leu Phe Pro Asn Tyr 165 170 175Tyr Ala Gln His
Ile Cys Arg Leu Lys Glu Trp Pro Asp Ala Leu Asn 180 185 190Arg Ser
Leu Lys His Val Asn Ser Thr Val Tyr Thr Leu Met Gln Gly 195 200
205Pro Ser Glu Leu Gly Met Ser Ser Asp Ala Arg Leu Ala Lys Trp Asp
210 215 220Ile Lys Asn Arg Leu His Glu Ile Ala Thr Pro Thr Leu Met
Ile Gly225 230 235 240Ala Arg Tyr Asp Thr Met Asp Pro Lys Ala Met
Glu Glu Gln Ser Lys 245 250 255Leu Val Gln Lys Gly Arg Tyr Leu Tyr
Cys Pro Asn Gly Ser His Leu 260 265 270Ala Met Trp Asp Asp Gln Lys
Val Phe Met Asp Gly Val Ile Lys Phe 275 280 285Ile Lys Asp Val Asp
Thr Lys Ser Phe Asn 290 29553310PRTN. gonorrhoeae 53Met Tyr Glu Ile
Lys Gln Pro Phe His Ser Gly Tyr Leu Gln Val Ser1 5 10 15Glu Ile His
Gln Ile Tyr Trp Glu Glu Ser Gly Asn Pro Asp Gly Val 20 25 30Pro Val
Ile Phe Leu His Gly Gly Pro Gly Ala Gly Ala Ser Pro Glu 35 40 45Cys
Arg Gly Phe Phe Asn Pro Asp Val Phe Arg Ile Val Ile Ile Asp 50 55
60Gln Arg Gly Cys Gly Arg Ser His Pro Tyr Ala Cys Ala Glu Asp Asn65
70 75 80Thr Thr Trp Asp Leu Val Ala Asp Ile Glu Lys Val Arg Glu Met
Leu 85 90 95Gly Ile Gly Lys Trp Leu Val Phe Gly Gly Ser Trp Gly Ser
Thr Leu 100 105 110Ser Leu Ala Tyr Ala Gln Thr His Pro Glu Arg Val
Lys Gly Leu Val 115 120 125Leu Arg Gly Ile Phe Leu Cys Arg Pro Ser
Glu Thr Ala Trp Leu Asn 130 135 140Glu Ala Gly Gly Val Ser Arg Ile
Tyr Pro Glu Gln Trp Gln Lys Phe145 150 155 160Val Ala Pro Ile Ala
Glu Asn Arg Arg Asn Arg Leu Ile Glu Ala Tyr 165 170 175His Gly Leu
Leu Phe His Gln Asp Glu Glu Val Cys Leu Ser Ala Ala 180 185 190Lys
Ala Trp Ala Asp Trp Glu Ser Tyr Leu Ile Arg Phe Glu Pro Glu 195 200
205Gly Val Asp Glu Asp Ala Tyr Ala Ser Leu Ala Ile Ala Arg Leu Glu
210 215 220Asn His Tyr Phe Val Asn Gly Gly Trp Leu Gln Gly Asp Lys
Ala Ile225 230 235 240Leu Asn Asn Ile Gly Lys Ile Arg His Ile Pro
Thr Val Ile Val Gln 245 250 255Gly Arg Tyr Asp Leu Cys Thr Pro Met
Gln Ser Ala Trp Glu Leu Ser 260 265 270Lys Ala Phe Pro Glu Ala Glu
Leu Arg Val Val Gln Ala Gly His Cys 275 280 285Ala Phe Asp Pro Pro
Leu Ala Asp Ala Leu Val Gln Ala Val Glu Asp 290 295 300Ile Leu Pro
Arg Leu Leu305 31054870PRTE. coli 54Met Thr Gln Gln Pro Gln Ala Lys
Tyr Arg His Asp Tyr Arg Ala Pro1 5 10 15Asp Tyr Gln Ile Thr Asp Ile
Asp Leu Thr Phe Asp Leu Asp Ala Gln 20 25 30Lys Thr Val Val Thr Ala
Val Ser Gln Ala Val Arg His Gly Ala Ser 35 40 45Asp Ala Pro Leu Arg
Leu Asn Gly Glu Asp Leu Lys Leu Val Ser Val 50 55 60His Ile Asn Asp
Glu Pro Trp Thr Ala Trp Lys Glu Glu Glu Gly Ala65 70 75 80Leu Val
Ile Ser Asn Leu Pro Glu Arg Phe Thr Leu Lys Ile Ile Asn 85 90 95Glu
Ile Ser Pro Ala Ala Asn Thr Ala Leu Glu Gly Leu Tyr Gln Ser 100 105
110Gly Asp Ala Leu Cys Thr Gln Cys Glu Ala Glu Gly Phe Arg His Ile
115 120 125Thr Tyr Tyr Leu Asp Arg Pro Asp Val Leu Ala Arg Phe Thr
Thr Lys 130 135 140Ile Ile Ala Asp Lys Ile Lys Tyr Pro Phe Leu Leu
Ser Asn Gly Asn145 150 155 160Arg Val Ala Gln Gly Glu Leu Glu Asn
Gly Arg His Trp Val Gln Trp 165 170 175Gln Asp Pro Phe Pro Lys Pro
Cys Tyr Leu Phe Ala Leu Val Ala Gly 180 185 190Asp Phe Asp Val Leu
Arg Asp Thr Phe Thr Thr Arg Ser Gly Arg Glu 195 200 205Val Ala Leu
Glu Leu Tyr Val Asp Arg Gly Asn Leu Asp Arg Ala Pro 210 215 220Trp
Ala Met Thr Ser Leu Lys Asn Ser Met Lys Trp Asp Glu Glu Arg225 230
235 240Phe Gly Leu Glu Tyr Asp Leu Asp Ile Tyr Met Ile Val Ala Val
Asp 245 250 255Phe Phe Asn Met Gly Ala Met Glu Asn Lys Gly Leu Asn
Ile Phe Asn 260 265 270Ser Lys Tyr Val Leu Ala Arg Thr Asp Thr Ala
Thr Asp Lys Asp Tyr 275 280 285Leu Asp Ile Glu Arg Val Ile Gly His
Glu Tyr Phe His Asn Trp Thr 290 295 300Gly Asn Arg Val Thr Cys Arg
Asp Trp Phe Gln Leu Ser Leu Lys Glu305 310 315 320Gly Leu Thr Val
Phe Arg Asp Gln Glu Phe Ser Ser Asp Leu Gly Ser 325 330 335Arg Ala
Val Asn Arg Ile Asn Asn Val Arg Thr Met Arg Gly Leu Gln 340 345
350Phe Ala Glu Asp Ala Ser Pro Met Ala His Pro Ile Arg Pro Asp Met
355 360 365Val Ile Glu Met Asn Asn Phe Tyr Thr Leu Thr Val Tyr Glu
Lys Gly 370 375 380Ala Glu Val Ile Arg Met Ile His Thr Leu Leu Gly
Glu Glu Asn Phe385 390 395 400Gln Lys Gly Met Gln Leu Tyr Phe Glu
Arg His Asp Gly Ser Ala Ala 405 410 415Thr Cys Asp Asp Phe Val Gln
Ala Met Glu Asp Ala Ser Asn Val Asp 420 425 430Leu Ser His Phe Arg
Arg Trp Tyr Ser Gln Ser Gly Thr Pro Ile Val 435 440 445Thr Val Lys
Asp Asp Tyr Asn Pro Glu Thr Glu Gln Tyr Thr Leu Thr 450 455 460Ile
Ser Gln Arg Thr Pro Ala Thr Pro Asp Gln Ala Glu Lys Gln Pro465 470
475 480Leu His Ile Pro Phe Ala Ile Glu Leu Tyr Asp Asn Glu Gly Lys
Val 485 490 495Ile Pro Leu Gln Lys Gly Gly His Pro Val Asn Ser Val
Leu Asn Val 500 505 510Thr Gln Ala Glu Gln Thr Phe Val Phe Asp Asn
Val Tyr Phe Gln Pro 515 520 525Val Pro Ala Leu Leu Cys Glu Phe Ser
Ala Pro Val Lys Leu Glu Tyr 530 535 540Lys Trp Ser Asp Gln Gln Leu
Thr Phe Leu Met Arg His Ala Arg Asn545 550 555 560Asp Phe Ser Arg
Trp Asp Ala Ala Gln Ser Leu Leu Ala Thr Tyr Ile 565 570 575Lys Leu
Asn Val Ala Arg His Gln Gln Gly Gln Pro Leu Ser Leu Pro 580 585
590Val His Val Ala Asp Ala Phe Arg Ala Val Leu Leu Asp Glu Lys Ile
595 600 605Asp Pro Ala Leu Ala Ala Glu Ile Leu Thr Leu Pro Ser Val
Asn Glu 610 615 620Met Ala Glu Leu Phe Asp Ile Ile Asp Pro Ile Ala
Ile Ala Glu Val625 630 635 640Arg Glu Ala Leu Thr Arg Thr Leu Ala
Thr Glu Leu Ala Asp Glu Leu 645 650 655Leu Ala Ile Tyr Asn Ala Asn
Tyr Gln Ser Glu Tyr Arg Val Glu His 660 665 670Glu Asp Ile Ala Lys
Arg Thr Leu Arg Asn Ala Cys Leu Arg Phe Leu 675 680 685Ala Phe Gly
Glu Thr His Leu Ala Asp Val Leu Val Ser Lys Gln Phe 690 695 700His
Glu Ala Asn Asn Met Thr Asp Ala Leu Ala Ala Leu Ser Ala Ala705 710
715 720Val Ala Ala Gln Leu Pro Cys Arg Asp Ala Leu Met Gln Glu Tyr
Asp 725 730 735Asp Lys Trp His Gln Asn Gly Leu Val Met Asp Lys Trp
Phe Ile Leu 740 745 750Gln Ala Thr Ser Pro Ala Ala Asn Val Leu Glu
Thr Val Arg Gly Leu 755 760 765Leu Gln His Arg Ser Phe Thr Met Ser
Asn Pro Asn Arg Ile Arg Ser 770 775 780Leu Ile Gly Ala Phe Ala Gly
Ser Asn Pro Ala Ala Phe His Ala Glu785 790 795 800Asp Gly Ser Gly
Tyr Leu Phe Leu Val Glu Met Leu Thr Asp Leu Asn 805 810 815Ser Arg
Asn Pro Gln Val Ala Ser Arg Leu Ile Glu Pro Leu Ile Arg 820 825
830Leu Lys Arg Tyr Asp Ala Lys Arg Gln Glu Lys Met Arg Ala Ala Leu
835 840 845Glu Gln Leu Lys Gly Leu Glu Asn Leu Ser Gly Asp Leu Tyr
Glu Lys 850 855 860Ile Thr Lys Ala Leu Ala865 87055889PRTP.
falciparum 55Pro Lys Ile His Tyr Arg Lys Asp Tyr Lys Pro Ser Gly
Phe Ile Ile1 5 10 15Asn Gln Val Thr Leu Asn Ile Asn Ile His Asp Gln
Glu Thr Ile Val 20 25 30Arg Ser Val Leu Asp Met Asp Ile Ser Lys His
Asn Val Gly Glu Asp 35 40 45Leu Val Phe Asp Gly Val Gly Leu Lys Ile
Asn Glu Ile Ser Ile Asn 50 55 60Asn Lys Lys Leu Val Glu Gly Glu Glu
Tyr Thr Tyr Asp Asn Glu Phe65 70 75 80Leu Thr Ile Phe Ser Lys Phe
Val Pro Lys Ser Lys Phe Ala Phe Ser 85 90 95Ser Glu Val Ile Ile His
Pro Glu Thr Asn Tyr Ala Leu Thr Gly Leu 100 105 110Tyr Lys Ser Lys
Asn Ile Ile Val Ser Gln Cys Glu Ala Thr Gly Phe 115 120 125Arg Arg
Ile Thr Phe Phe Ile Asp Arg Pro Asp Met Met Ala Lys Tyr 130 135
140Asp Val Thr Val Thr Ala Asp Lys Glu Lys Tyr Pro Val Leu Leu
Ser145 150 155 160Asn Gly Asp Lys Val Asn Glu Phe Glu Ile Pro Gly
Gly Arg His Gly 165 170 175Ala Arg Phe Asn Asp Pro Pro Leu Lys Pro
Cys Tyr Leu Phe Ala Val 180 185 190Val Ala Gly Asp Leu Lys His Leu
Ser Ala Thr Tyr Ile Thr Lys Tyr 195 200 205Thr Lys Lys Lys Val Glu
Leu Tyr Val Phe Ser Glu Glu Lys Tyr Val 210 215 220Ser Lys Leu Gln
Trp Ala Leu Glu Cys Leu Lys Lys Ser Met Ala Phe225 230 235 240Asp
Glu Asp Tyr Phe Gly Leu Glu Tyr Asp Leu Ser Arg Leu Asn Leu 245 250
255Val Ala Val Ser Asp Phe Asn Val Gly Ala Met Glu Asn Lys Gly Leu
260 265 270Asn Ile Phe Asn Ala Asn Ser Leu Leu Ala Ser Lys Lys Asn
Ser Ile 275 280 285Asp Phe Ser Tyr Ala Arg Ile Leu Thr Val Val Gly
His Glu Tyr Phe 290 295 300His Gln Tyr Thr Gly Asn Arg Val Thr Leu
Arg Asp Trp Phe Gln Leu305 310 315 320Thr Leu Lys Glu Gly Leu Thr
Val His Arg Glu Asn Leu Phe Ser Glu 325 330 335Glu Met Thr Lys Thr
Val Thr Thr Arg Leu Ser His Val Asp Leu Leu 340 345 350Arg Ser Val
Gln Phe Leu Glu Asp Ser Ser Pro Leu Ser His Pro Ile 355 360 365Arg
Pro Glu Ser Tyr Val Ser Met Glu Asn Phe Tyr Thr Thr Thr Val 370 375
380Tyr Asp Lys Gly Ser Glu Val Met Arg Met Tyr Leu Thr Ile Leu
Gly385 390 395 400Glu Glu Tyr Tyr Lys Lys Gly Phe Asp Ile Tyr Ile
Lys Lys Asn Asp 405 410 415Gly Asn Thr Ala Thr Cys Glu Asp Phe Asn
Tyr Ala Met Glu Gln Ala 420 425 430Tyr Lys Met Lys Lys Ala Asp Asn
Ser Ala Asn Leu Asn Gln Tyr Leu 435 440 445Leu Trp Phe Ser Gln Ser
Gly Thr Pro His Val Ser Phe Lys Tyr Asn 450 455 460Tyr Asp Ala Glu
Lys Lys Gln Tyr Ser Ile His Val Asn Gln Tyr Thr465 470 475 480Lys
Pro Asp Glu Asn Gln Lys Glu Lys Lys Pro Leu
Phe Ile Pro Ile 485 490 495Ser Val Gly Leu Ile Asn Pro Glu Asn Gly
Lys Glu Met Ile Ser Gln 500 505 510Thr Thr Leu Glu Leu Thr Lys Glu
Ser Asp Thr Phe Val Phe Asn Asn 515 520 525Ile Ala Val Lys Pro Ile
Pro Ser Leu Phe Arg Gly Phe Ser Ala Pro 530 535 540Val Tyr Ile Glu
Asp Gln Leu Thr Asp Glu Glu Arg Ile Leu Leu Leu545 550 555 560Lys
Tyr Asp Ser Asp Ala Phe Val Arg Tyr Asn Ser Cys Thr Asn Ile 565 570
575Tyr Met Lys Gln Ile Leu Met Asn Tyr Asn Glu Phe Leu Lys Ala Lys
580 585 590Asn Glu Lys Leu Glu Ser Phe Gln Leu Thr Pro Val Asn Ala
Gln Phe 595 600 605Ile Asp Ala Ile Lys Tyr Leu Leu Glu Asp Pro His
Ala Asp Ala Gly 610 615 620Phe Lys Ser Tyr Ile Val Ser Leu Pro Gln
Asp Arg Tyr Ile Ile Asn625 630 635 640Phe Val Ser Asn Leu Asp Thr
Asp Val Leu Ala Asp Thr Lys Glu Tyr 645 650 655Ile Tyr Lys Gln Ile
Gly Asp Lys Leu Asn Asp Val Tyr Tyr Lys Met 660 665 670Phe Lys Ser
Leu Glu Ala Lys Ala Asp Asp Leu Thr Tyr Phe Asn Asp 675 680 685Glu
Ser His Val Asp Phe Asp Gln Met Asn Met Arg Thr Leu Arg Asn 690 695
700Thr Leu Leu Ser Leu Leu Ser Lys Ala Gln Tyr Pro Asn Ile Leu
Asn705 710 715 720Glu Ile Ile Glu His Ser Lys Ser Pro Tyr Pro Ser
Asn Trp Leu Thr 725 730 735Ser Leu Ser Val Ser Ala Tyr Phe Asp Lys
Tyr Phe Glu Leu Tyr Asp 740 745 750Lys Thr Tyr Lys Leu Ser Lys Asp
Asp Glu Leu Leu Leu Gln Glu Trp 755 760 765Leu Lys Thr Val Ser Arg
Ser Asp Arg Lys Asp Ile Tyr Glu Ile Leu 770 775 780Lys Lys Leu Glu
Asn Glu Val Leu Lys Asp Ser Lys Asn Pro Asn Asp785 790 795 800Ile
Arg Ala Val Tyr Leu Pro Phe Thr Asn Asn Leu Arg Arg Phe His 805 810
815Asp Ile Ser Gly Lys Gly Tyr Lys Leu Ile Ala Glu Val Ile Thr Lys
820 825 830Thr Asp Lys Phe Asn Pro Met Val Ala Thr Gln Leu Cys Glu
Pro Phe 835 840 845Lys Leu Trp Asn Lys Leu Asp Thr Lys Arg Gln Glu
Leu Met Leu Asn 850 855 860Glu Met Asn Thr Met Leu Gln Glu Pro Gln
Ile Ser Asn Asn Leu Lys865 870 875 880Glu Tyr Leu Leu Arg Leu Thr
Asn Lys 88556919PRTUnknownPuromycin-sensitive aminopeptidase 56Met
Trp Leu Ala Ala Ala Ala Pro Ser Leu Ala Arg Arg Leu Leu Phe1 5 10
15Leu Gly Pro Pro Pro Pro Pro Leu Leu Leu Leu Val Phe Ser Arg Ser
20 25 30Ser Arg Arg Arg Leu His Ser Leu Gly Leu Ala Ala Met Pro Glu
Lys 35 40 45Arg Pro Phe Glu Arg Leu Pro Ala Asp Val Ser Pro Ile Asn
Tyr Ser 50 55 60Leu Cys Leu Lys Pro Asp Leu Leu Asp Phe Thr Phe Glu
Gly Lys Leu65 70 75 80Glu Ala Ala Ala Gln Val Arg Gln Ala Thr Asn
Gln Ile Val Met Asn 85 90 95Cys Ala Asp Ile Asp Ile Ile Thr Ala Ser
Tyr Ala Pro Glu Gly Asp 100 105 110Glu Glu Ile His Ala Thr Gly Phe
Asn Tyr Gln Asn Glu Asp Glu Lys 115 120 125Val Thr Leu Ser Phe Pro
Ser Thr Leu Gln Thr Gly Thr Gly Thr Leu 130 135 140Lys Ile Asp Phe
Val Gly Glu Leu Asn Asp Lys Met Lys Gly Phe Tyr145 150 155 160Arg
Ser Lys Tyr Thr Thr Pro Ser Gly Glu Val Arg Tyr Ala Ala Val 165 170
175Thr Gln Phe Glu Ala Thr Asp Ala Arg Arg Ala Phe Pro Cys Trp Asp
180 185 190Glu Pro Ala Ile Lys Ala Thr Phe Asp Ile Ser Leu Val Val
Pro Lys 195 200 205Asp Arg Val Ala Leu Ser Asn Met Asn Val Ile Asp
Arg Lys Pro Tyr 210 215 220Pro Asp Asp Glu Asn Leu Val Glu Val Lys
Phe Ala Arg Thr Pro Val225 230 235 240Met Ser Thr Tyr Leu Val Ala
Phe Val Val Gly Glu Tyr Asp Phe Val 245 250 255Glu Thr Arg Ser Lys
Asp Gly Val Cys Val Arg Val Tyr Thr Pro Val 260 265 270Gly Lys Ala
Glu Gln Gly Lys Phe Ala Leu Glu Val Ala Ala Lys Thr 275 280 285Leu
Pro Phe Tyr Lys Asp Tyr Phe Asn Val Pro Tyr Pro Leu Pro Lys 290 295
300Ile Asp Leu Ile Ala Ile Ala Asp Phe Ala Ala Gly Ala Met Glu
Asn305 310 315 320Trp Gly Leu Val Thr Tyr Arg Glu Thr Ala Leu Leu
Ile Asp Pro Lys 325 330 335Asn Ser Cys Ser Ser Ser Arg Gln Trp Val
Ala Leu Val Val Gly His 340 345 350Glu Leu Ala His Gln Trp Phe Gly
Asn Leu Val Thr Met Glu Trp Trp 355 360 365Thr His Leu Trp Leu Asn
Glu Gly Phe Ala Ser Trp Ile Glu Tyr Leu 370 375 380Cys Val Asp His
Cys Phe Pro Glu Tyr Asp Ile Trp Thr Gln Phe Val385 390 395 400Ser
Ala Asp Tyr Thr Arg Ala Gln Glu Leu Asp Ala Leu Asp Asn Ser 405 410
415His Pro Ile Glu Val Ser Val Gly His Pro Ser Glu Val Asp Glu Ile
420 425 430Phe Asp Ala Ile Ser Tyr Ser Lys Gly Ala Ser Val Ile Arg
Met Leu 435 440 445His Asp Tyr Ile Gly Asp Lys Asp Phe Lys Lys Gly
Met Asn Met Tyr 450 455 460Leu Thr Lys Phe Gln Gln Lys Asn Ala Ala
Thr Glu Asp Leu Trp Glu465 470 475 480Ser Leu Glu Asn Ala Ser Gly
Lys Pro Ile Ala Ala Val Met Asn Thr 485 490 495Trp Thr Lys Gln Met
Gly Phe Pro Leu Ile Tyr Val Glu Ala Glu Gln 500 505 510Val Glu Asp
Asp Arg Leu Leu Arg Leu Ser Gln Lys Lys Phe Cys Ala 515 520 525Gly
Gly Ser Tyr Val Gly Glu Asp Cys Pro Gln Trp Met Val Pro Ile 530 535
540Thr Ile Ser Thr Ser Glu Asp Pro Asn Gln Ala Lys Leu Lys Ile
Leu545 550 555 560Met Asp Lys Pro Glu Met Asn Val Val Leu Lys Asn
Val Lys Pro Asp 565 570 575Gln Trp Val Lys Leu Asn Leu Gly Thr Val
Gly Phe Tyr Arg Thr Gln 580 585 590Tyr Ser Ser Ala Met Leu Glu Ser
Leu Leu Pro Gly Ile Arg Asp Leu 595 600 605Ser Leu Pro Pro Val Asp
Arg Leu Gly Leu Gln Asn Asp Leu Phe Ser 610 615 620Leu Ala Arg Ala
Gly Ile Ile Ser Thr Val Glu Val Leu Lys Val Met625 630 635 640Glu
Ala Phe Val Asn Glu Pro Asn Tyr Thr Val Trp Ser Asp Leu Ser 645 650
655Cys Asn Leu Gly Ile Leu Ser Thr Leu Leu Ser His Thr Asp Phe Tyr
660 665 670Glu Glu Ile Gln Glu Phe Val Lys Asp Val Phe Ser Pro Ile
Gly Glu 675 680 685Arg Leu Gly Trp Asp Pro Lys Pro Gly Glu Gly His
Leu Asp Ala Leu 690 695 700Leu Arg Gly Leu Val Leu Gly Lys Leu Gly
Lys Ala Gly His Lys Ala705 710 715 720Thr Leu Glu Glu Ala Arg Arg
Arg Phe Lys Asp His Val Glu Gly Lys 725 730 735Gln Ile Leu Ser Ala
Asp Leu Arg Ser Pro Val Tyr Leu Thr Val Leu 740 745 750Lys His Gly
Asp Gly Thr Thr Leu Asp Ile Met Leu Lys Leu His Lys 755 760 765Gln
Ala Asp Met Gln Glu Glu Lys Asn Arg Ile Glu Arg Val Leu Gly 770 775
780Ala Thr Leu Leu Pro Asp Leu Ile Gln Lys Val Leu Thr Phe Ala
Leu785 790 795 800Ser Glu Glu Val Arg Pro Gln Asp Thr Val Ser Val
Ile Gly Gly Val 805 810 815Ala Gly Gly Ser Lys His Gly Arg Lys Ala
Ala Trp Lys Phe Ile Lys 820 825 830Asp Asn Trp Glu Glu Leu Tyr Asn
Arg Tyr Gln Gly Gly Phe Leu Ile 835 840 845Ser Arg Leu Ile Lys Leu
Ser Val Glu Gly Phe Ala Val Asp Lys Met 850 855 860Ala Gly Glu Val
Lys Ala Phe Phe Glu Ser His Pro Ala Pro Ser Ala865 870 875 880Glu
Arg Thr Ile Gln Gln Cys Cys Glu Asn Ile Leu Leu Asn Ala Ala 885 890
895Trp Leu Lys Arg Asp Ala Glu Ser Ile His Gln Tyr Leu Leu Gln Arg
900 905 910Lys Ala Ser Pro Pro Thr Val 91557919PRTArtificial
SequenceSynthetic Polypeptide 57Met Trp Leu Ala Ala Ala Ala Pro Ser
Leu Ala Arg Arg Leu Leu Phe1 5 10 15Leu Gly Pro Pro Pro Pro Pro Leu
Leu Leu Leu Val Phe Ser Arg Ser 20 25 30Ser Arg Arg Arg Leu His Ser
Leu Gly Leu Ala Ala Met Pro Glu Lys 35 40 45Arg Pro Phe Glu Arg Leu
Pro Ala Asp Val Ser Pro Ile Asn Tyr Ser 50 55 60Leu Cys Leu Lys Pro
Asp Leu Leu Asp Phe Thr Phe Glu Gly Lys Leu65 70 75 80Glu Ala Ala
Ala Gln Val Arg Gln Ala Thr Asn Gln Ile Val Met Asn 85 90 95Cys Ala
Asp Ile Asp Ile Ile Thr Ala Ser Tyr Ala Pro Glu Gly Asp 100 105
110Glu Glu Ile His Ala Thr Gly Phe Asn Tyr Gln Asn Glu Asp Glu Lys
115 120 125Val Thr Leu Ser Phe Pro Ser Thr Leu Gln Thr Gly Thr Gly
Thr Leu 130 135 140Lys Ile Asp Phe Val Gly Glu Leu Asn Asp Lys Met
Lys Gly Phe Tyr145 150 155 160Arg Ser Lys Tyr Thr Thr Pro Ser Gly
Glu Val Arg Tyr Ala Ala Val 165 170 175Thr Gln Phe Glu Ala Thr Asp
Ala Arg Arg Ala Phe Pro Cys Trp Asp 180 185 190Glu Pro Ala Ile Lys
Ala Thr Phe Asp Ile Ser Leu Val Val Pro Lys 195 200 205Asp Arg Val
Ala Leu Ser Asn Met Asn Val Ile Asp Arg Lys Pro Tyr 210 215 220Pro
Asp Asp Glu Asn Leu Val Glu Val Lys Phe Ala Arg Thr Pro Val225 230
235 240Met Ser Thr Tyr Leu Val Ala Phe Val Val Gly Glu Tyr Asp Phe
Val 245 250 255Glu Thr Arg Ser Lys Asp Gly Val Cys Val Arg Val Tyr
Thr Pro Val 260 265 270Gly Lys Ala Glu Gln Gly Lys Phe Ala Leu Glu
Val Ala Ala Lys Thr 275 280 285Leu Pro Phe Tyr Lys Asp Tyr Phe Asn
Val Pro Tyr Pro Leu Pro Lys 290 295 300Ile Asp Leu Ile Ala Ile Ala
Asp Phe Ala Ala Gly Ala Met Glu Asn305 310 315 320Trp Gly Leu Val
Thr Tyr Arg Glu Thr Ala Leu Leu Ile Asp Pro Lys 325 330 335Asn Ser
Cys Ser Ser Ser Arg Gln Trp Val Ala Leu Val Val Gly His 340 345
350Val Leu Ala His Gln Trp Phe Gly Asn Leu Val Thr Met Glu Trp Trp
355 360 365Thr His Leu Trp Leu Asn Glu Gly Phe Ala Ser Trp Ile Glu
Tyr Leu 370 375 380Cys Val Asp His Cys Phe Pro Glu Tyr Asp Ile Trp
Thr Gln Phe Val385 390 395 400Ser Ala Asp Tyr Thr Arg Ala Gln Glu
Leu Asp Ala Leu Asp Asn Ser 405 410 415His Pro Ile Glu Val Ser Val
Gly His Pro Ser Glu Val Asp Glu Ile 420 425 430Phe Asp Ala Ile Ser
Tyr Ser Lys Gly Ala Ser Val Ile Arg Met Leu 435 440 445His Asp Tyr
Ile Gly Asp Lys Asp Phe Lys Lys Gly Met Asn Met Tyr 450 455 460Leu
Thr Lys Phe Gln Gln Lys Asn Ala Ala Thr Glu Asp Leu Trp Glu465 470
475 480Ser Leu Glu Asn Ala Ser Gly Lys Pro Ile Ala Ala Val Met Asn
Thr 485 490 495Trp Thr Lys Gln Met Gly Phe Pro Leu Ile Tyr Val Glu
Ala Glu Gln 500 505 510Val Glu Asp Asp Arg Leu Leu Arg Leu Ser Gln
Lys Lys Phe Cys Ala 515 520 525Gly Gly Ser Tyr Val Gly Glu Asp Cys
Pro Gln Trp Met Val Pro Ile 530 535 540Thr Ile Ser Thr Ser Glu Asp
Pro Asn Gln Ala Lys Leu Lys Ile Leu545 550 555 560Met Asp Lys Pro
Glu Met Asn Val Val Leu Lys Asn Val Lys Pro Asp 565 570 575Gln Trp
Val Lys Leu Asn Leu Gly Thr Val Gly Phe Tyr Arg Thr Gln 580 585
590Tyr Ser Ser Ala Met Leu Glu Ser Leu Leu Pro Gly Ile Arg Asp Leu
595 600 605Ser Leu Pro Pro Val Asp Arg Leu Gly Leu Gln Asn Asp Leu
Phe Ser 610 615 620Leu Ala Arg Ala Gly Ile Ile Ser Thr Val Glu Val
Leu Lys Val Met625 630 635 640Glu Ala Phe Val Asn Glu Pro Asn Tyr
Thr Val Trp Ser Asp Leu Ser 645 650 655Cys Asn Leu Gly Ile Leu Ser
Thr Leu Leu Ser His Thr Asp Phe Tyr 660 665 670Glu Glu Ile Gln Glu
Phe Val Lys Asp Val Phe Ser Pro Ile Gly Glu 675 680 685Arg Leu Gly
Trp Asp Pro Lys Pro Gly Glu Gly His Leu Asp Ala Leu 690 695 700Leu
Arg Gly Leu Val Leu Gly Lys Leu Gly Lys Ala Gly His Lys Ala705 710
715 720Thr Leu Glu Glu Ala Arg Arg Arg Phe Lys Asp His Val Glu Gly
Lys 725 730 735Gln Ile Leu Ser Ala Asp Leu Arg Ser Pro Val Tyr Leu
Thr Val Leu 740 745 750Lys His Gly Asp Gly Thr Thr Leu Asp Ile Met
Leu Lys Leu His Lys 755 760 765Gln Ala Asp Met Gln Glu Glu Lys Asn
Arg Ile Glu Arg Val Leu Gly 770 775 780Ala Thr Leu Leu Pro Asp Leu
Ile Gln Lys Val Leu Thr Phe Ala Leu785 790 795 800Ser Glu Glu Val
Arg Pro Gln Asp Thr Val Ser Val Ile Gly Gly Val 805 810 815Ala Gly
Gly Ser Lys His Gly Arg Lys Ala Ala Trp Lys Phe Ile Lys 820 825
830Asp Asn Trp Glu Glu Leu Tyr Asn Arg Tyr Gln Gly Gly Phe Leu Ile
835 840 845Ser Arg Leu Ile Lys Leu Ser Val Glu Gly Phe Ala Val Asp
Lys Met 850 855 860Ala Gly Glu Val Lys Ala Phe Phe Glu Ser His Pro
Ala Pro Ser Ala865 870 875 880Glu Arg Thr Ile Gln Gln Cys Cys Glu
Asn Ile Leu Leu Asn Ala Ala 885 890 895Trp Leu Lys Arg Asp Ala Glu
Ser Ile His Gln Tyr Leu Leu Gln Arg 900 905 910Lys Ala Ser Pro Pro
Thr Val 91558864PRTFrancisella tularensis 58Met Ile Tyr Glu Phe Val
Met Thr Asp Pro Lys Ile Lys Tyr Leu Lys1 5 10 15Asp Tyr Lys Pro Ser
Asn Tyr Leu Ile Asp Glu Thr His Leu Ile Phe 20 25 30Glu Leu Asp Glu
Ser Lys Thr Arg Val Thr Ala Asn Leu Tyr Ile Val 35 40 45Ala Asn Arg
Glu Asn Arg Glu Asn Asn Thr Leu Val Leu Asp Gly Val 50 55 60Glu Leu
Lys Leu Leu Ser Ile Lys Leu Asn Asn Lys His Leu Ser Pro65 70 75
80Ala Glu Phe Ala Val Asn Glu Asn Gln Leu Ile Ile Asn Asn Val Pro
85 90 95Glu Lys Phe Val Leu Gln Thr Val Val Glu Ile Asn Pro Ser Ala
Asn 100 105 110Thr Ser Leu Glu Gly Leu Tyr Lys Ser Gly Asp Val Phe
Ser Thr Gln 115 120 125Cys Glu Ala Thr Gly Phe Arg Lys Ile Thr Tyr
Tyr Leu Asp Arg Pro 130 135 140Asp Val Met Ala Ala Phe Thr Val Lys
Ile Ile Ala Asp Lys Lys Lys145 150 155 160Tyr Pro Ile Ile Leu Ser
Asn Gly Asp Lys Ile Asp Ser Gly Asp Ile 165 170 175Ser Asp Asn Gln
His Phe Ala Val Trp Lys Asp Pro Phe Lys Lys Pro 180 185 190Cys Tyr
Leu Phe Ala Leu Val Ala Gly Asp Leu Ala Ser Ile Lys Asp 195 200
205Thr Tyr Ile Thr Lys Ser Gln Arg Lys
Val Ser Leu Glu Ile Tyr Ala 210 215 220Phe Lys Gln Asp Ile Asp Lys
Cys His Tyr Ala Met Gln Ala Val Lys225 230 235 240Asp Ser Met Lys
Trp Asp Glu Asp Arg Phe Gly Leu Glu Tyr Asp Leu 245 250 255Asp Thr
Phe Met Ile Val Ala Val Pro Asp Phe Asn Ala Gly Ala Met 260 265
270Glu Asn Lys Gly Leu Asn Ile Phe Asn Thr Lys Tyr Ile Met Ala Ser
275 280 285Asn Lys Thr Ala Thr Asp Lys Asp Phe Glu Leu Val Gln Ser
Val Val 290 295 300Gly His Glu Tyr Phe His Asn Trp Thr Gly Asp Arg
Val Thr Cys Arg305 310 315 320Asp Trp Phe Gln Leu Ser Leu Lys Glu
Gly Leu Thr Val Phe Arg Asp 325 330 335Gln Glu Phe Thr Ser Asp Leu
Asn Ser Arg Asp Val Lys Arg Ile Asp 340 345 350Asp Val Arg Ile Ile
Arg Ser Ala Gln Phe Ala Glu Asp Ala Ser Pro 355 360 365Met Ser His
Pro Ile Arg Pro Glu Ser Tyr Ile Glu Met Asn Asn Phe 370 375 380Tyr
Thr Val Thr Val Tyr Asn Lys Gly Ala Glu Ile Ile Arg Met Ile385 390
395 400His Thr Leu Leu Gly Glu Glu Gly Phe Gln Lys Gly Met Lys Leu
Tyr 405 410 415Phe Glu Arg His Asp Gly Gln Ala Val Thr Cys Asp Asp
Phe Val Asn 420 425 430Ala Met Ala Asp Ala Asn Asn Arg Asp Phe Ser
Leu Phe Lys Arg Trp 435 440 445Tyr Ala Gln Ser Gly Thr Pro Asn Ile
Lys Val Ser Glu Asn Tyr Asp 450 455 460Ala Ser Ser Gln Thr Tyr Ser
Leu Thr Leu Glu Gln Thr Thr Leu Pro465 470 475 480Thr Ala Asp Gln
Lys Glu Lys Gln Ala Leu His Ile Pro Val Lys Met 485 490 495Gly Leu
Ile Asn Pro Glu Gly Lys Asn Ile Ala Glu Gln Val Ile Glu 500 505
510Leu Lys Glu Gln Lys Gln Thr Tyr Thr Phe Glu Asn Ile Ala Ala Lys
515 520 525Pro Val Ala Ser Leu Phe Arg Asp Phe Ser Ala Pro Val Lys
Val Glu 530 535 540His Lys Arg Ser Glu Lys Asp Leu Leu His Ile Val
Lys Tyr Asp Asn545 550 555 560Asn Ala Phe Asn Arg Trp Asp Ser Leu
Gln Gln Ile Ala Thr Asn Ile 565 570 575Ile Leu Asn Asn Ala Asp Leu
Asn Asp Glu Phe Leu Asn Ala Phe Lys 580 585 590Ser Ile Leu His Asp
Lys Asp Leu Asp Lys Ala Leu Ile Ser Asn Ala 595 600 605Leu Leu Ile
Pro Ile Glu Ser Thr Ile Ala Glu Ala Met Arg Val Ile 610 615 620Met
Val Asp Asp Ile Val Leu Ser Arg Lys Asn Val Val Asn Gln Leu625 630
635 640Ala Asp Lys Leu Lys Asp Asp Trp Leu Ala Val Tyr Gln Gln Cys
Asn 645 650 655Asp Asn Lys Pro Tyr Ser Leu Ser Ala Glu Gln Ile Ala
Lys Arg Lys 660 665 670Leu Lys Gly Val Cys Leu Ser Tyr Leu Met Asn
Ala Ser Asp Gln Lys 675 680 685Val Gly Thr Asp Leu Ala Gln Gln Leu
Phe Asp Asn Ala Asp Asn Met 690 695 700Thr Asp Gln Gln Thr Ala Phe
Thr Glu Leu Leu Lys Ser Asn Asp Lys705 710 715 720Gln Val Arg Asp
Asn Ala Ile Asn Glu Phe Tyr Asn Arg Trp Arg His 725 730 735Glu Asp
Leu Val Val Asn Lys Trp Leu Leu Ser Gln Ala Gln Ile Ser 740 745
750His Glu Ser Ala Leu Asp Ile Val Lys Gly Leu Val Asn His Pro Ala
755 760 765Tyr Asn Pro Lys Asn Pro Asn Lys Val Tyr Ser Leu Ile Gly
Gly Phe 770 775 780Gly Ala Asn Phe Leu Gln Tyr His Cys Lys Asp Gly
Leu Gly Tyr Ala785 790 795 800Phe Met Ala Asp Thr Val Leu Ala Leu
Asp Lys Phe Asn His Gln Val 805 810 815Ala Ala Arg Met Ala Arg Asn
Leu Met Ser Trp Lys Arg Tyr Asp Ser 820 825 830Asp Arg Gln Ala Met
Met Lys Asn Ala Leu Glu Lys Ile Lys Ala Ser 835 840 845Asn Pro Ser
Lys Asn Val Phe Glu Ile Val Ser Lys Ser Leu Glu Ser 850 855
86059353PRTPyrococcus horikoshii 59Met Glu Val Arg Asn Met Val Asp
Tyr Glu Leu Leu Lys Lys Val Val1 5 10 15Glu Ala Pro Gly Val Ser Gly
Tyr Glu Phe Leu Gly Ile Arg Asp Val 20 25 30Val Ile Glu Glu Ile Lys
Asp Tyr Val Asp Glu Val Lys Val Asp Lys 35 40 45Leu Gly Asn Val Ile
Ala His Lys Lys Gly Glu Gly Pro Lys Val Met 50 55 60Ile Ala Ala His
Met Asp Gln Ile Gly Leu Met Val Thr His Ile Glu65 70 75 80Lys Asn
Gly Phe Leu Arg Val Ala Pro Ile Gly Gly Val Asp Pro Lys 85 90 95Thr
Leu Ile Ala Gln Arg Phe Lys Val Trp Ile Asp Lys Gly Lys Phe 100 105
110Ile Tyr Gly Val Gly Ala Ser Val Pro Pro His Ile Gln Lys Pro Glu
115 120 125Asp Arg Lys Lys Ala Pro Asp Trp Asp Gln Ile Phe Ile Asp
Ile Gly 130 135 140Ala Glu Ser Lys Glu Glu Ala Glu Asp Met Gly Val
Lys Ile Gly Thr145 150 155 160Val Ile Thr Trp Asp Gly Arg Leu Glu
Arg Leu Gly Lys His Arg Phe 165 170 175Val Ser Ile Ala Phe Asp Asp
Arg Ile Ala Val Tyr Thr Ile Leu Glu 180 185 190Val Ala Lys Gln Leu
Lys Asp Ala Lys Ala Asp Val Tyr Phe Val Ala 195 200 205Thr Val Gln
Glu Glu Val Gly Leu Arg Gly Ala Arg Thr Ser Ala Phe 210 215 220Gly
Ile Glu Pro Asp Tyr Gly Phe Ala Ile Asp Val Thr Ile Ala Ala225 230
235 240Asp Ile Pro Gly Thr Pro Glu His Lys Gln Val Thr His Leu Gly
Lys 245 250 255Gly Thr Ala Ile Lys Ile Met Asp Arg Ser Val Ile Cys
His Pro Thr 260 265 270Ile Val Arg Trp Leu Glu Glu Leu Ala Lys Lys
His Glu Ile Pro Tyr 275 280 285Gln Leu Glu Ile Leu Leu Gly Gly Gly
Thr Asp Ala Gly Ala Ile His 290 295 300Leu Thr Lys Ala Gly Val Pro
Thr Gly Ala Leu Ser Val Pro Ala Arg305 310 315 320Tyr Ile His Ser
Asn Thr Glu Val Val Asp Glu Arg Asp Val Asp Ala 325 330 335Thr Val
Glu Leu Met Thr Lys Ala Leu Glu Asn Ile His Glu Leu Lys 340 345
350Ile60408PRTT. aquaticus 60Met Asp Ala Phe Thr Glu Asn Leu Asn
Lys Leu Ala Glu Leu Ala Ile1 5 10 15Arg Val Gly Leu Asn Leu Glu Glu
Gly Gln Glu Ile Val Ala Thr Ala 20 25 30Pro Ile Glu Ala Val Asp Phe
Val Arg Leu Leu Ala Glu Lys Ala Tyr 35 40 45Glu Asn Gly Ala Ser Leu
Phe Thr Val Leu Tyr Gly Asp Asn Leu Ile 50 55 60Ala Arg Lys Arg Leu
Ala Leu Val Pro Glu Ala His Leu Asp Arg Ala65 70 75 80Pro Ala Trp
Leu Tyr Glu Gly Met Ala Lys Ala Phe His Glu Gly Ala 85 90 95Ala Arg
Leu Ala Val Ser Gly Asn Asp Pro Lys Ala Leu Glu Gly Leu 100 105
110Pro Pro Glu Arg Val Gly Arg Ala Gln Gln Ala Gln Ser Arg Ala Tyr
115 120 125Arg Pro Thr Leu Ser Ala Ile Thr Glu Phe Val Thr Asn Trp
Thr Ile 130 135 140Val Pro Phe Ala His Pro Gly Trp Ala Lys Ala Val
Phe Pro Gly Leu145 150 155 160Pro Glu Glu Glu Ala Val Gln Arg Leu
Trp Gln Ala Ile Phe Gln Ala 165 170 175Thr Arg Val Asp Gln Glu Asp
Pro Val Ala Ala Trp Glu Ala His Asn 180 185 190Arg Val Leu His Ala
Lys Val Ala Phe Leu Asn Glu Lys Arg Phe His 195 200 205Ala Leu His
Phe Gln Gly Pro Gly Thr Asp Leu Thr Val Gly Leu Ala 210 215 220Glu
Gly His Leu Trp Gln Gly Gly Ala Thr Pro Thr Lys Lys Gly Arg225 230
235 240Leu Cys Asn Pro Asn Leu Pro Thr Glu Glu Val Phe Thr Ala Pro
His 245 250 255Arg Glu Arg Val Glu Gly Val Val Arg Ala Ser Arg Pro
Leu Ala Leu 260 265 270Ser Gly Gln Leu Val Glu Gly Leu Trp Ala Arg
Phe Glu Gly Gly Val 275 280 285Ala Val Glu Val Gly Ala Glu Lys Gly
Glu Glu Val Leu Lys Lys Leu 290 295 300Leu Asp Thr Asp Glu Gly Ala
Arg Arg Leu Gly Glu Val Ala Leu Val305 310 315 320Pro Ala Asp Asn
Pro Ile Ala Lys Thr Gly Leu Val Phe Phe Asp Thr 325 330 335Leu Phe
Asp Glu Asn Ala Ala Ser His Ile Ala Phe Gly Gln Ala Tyr 340 345
350Ala Glu Asn Leu Glu Gly Arg Pro Ser Gly Glu Glu Phe Arg Arg Arg
355 360 365Gly Gly Asn Glu Ser Met Val His Val Asp Trp Met Ile Gly
Ser Glu 370 375 380Glu Val Asp Val Asp Gly Leu Leu Glu Asp Gly Thr
Arg Val Pro Leu385 390 395 400Met Arg Arg Gly Arg Trp Val Ile
40561362PRTBacillus stearothermophilus 61Met Ala Lys Leu Asp Glu
Thr Leu Thr Met Leu Lys Ala Leu Thr Asp1 5 10 15Ala Lys Gly Val Pro
Gly Asn Glu Arg Glu Ala Arg Asp Val Met Lys 20 25 30Thr Tyr Ile Ala
Pro Tyr Ala Asp Glu Val Thr Thr Asp Gly Leu Gly 35 40 45Ser Leu Ile
Ala Lys Lys Glu Gly Lys Ser Gly Gly Pro Lys Val Met 50 55 60Ile Ala
Gly His Leu Asp Glu Val Gly Phe Met Val Thr Gln Ile Asp65 70 75
80Asp Lys Gly Phe Ile Arg Phe Gln Thr Leu Gly Gly Trp Trp Ser Gln
85 90 95Val Met Leu Ala Gln Arg Val Thr Ile Val Thr Lys Lys Gly Asp
Ile 100 105 110Thr Gly Val Ile Gly Ser Lys Pro Pro His Ile Leu Pro
Ser Glu Ala 115 120 125Arg Lys Lys Pro Val Glu Ile Lys Asp Met Phe
Ile Asp Ile Gly Ala 130 135 140Thr Ser Arg Glu Glu Ala Met Glu Trp
Gly Val Arg Pro Gly Asp Met145 150 155 160Ile Val Pro Tyr Phe Glu
Phe Thr Val Leu Asn Asn Glu Lys Met Leu 165 170 175Leu Ala Lys Ala
Trp Asp Asn Arg Ile Gly Cys Ala Val Ala Ile Asp 180 185 190Val Leu
Lys Gln Leu Lys Gly Val Asp His Pro Asn Thr Val Tyr Gly 195 200
205Val Gly Thr Val Gln Glu Glu Val Gly Leu Arg Gly Ala Arg Thr Ala
210 215 220Ala Gln Phe Ile Gln Pro Asp Ile Ala Phe Ala Val Asp Val
Gly Ile225 230 235 240Ala Gly Asp Thr Pro Gly Val Ser Glu Lys Glu
Ala Met Gly Lys Leu 245 250 255Gly Ala Gly Pro His Ile Val Leu Tyr
Asp Ala Thr Met Val Ser His 260 265 270Arg Gly Leu Arg Glu Phe Val
Ile Glu Val Ala Glu Glu Leu Asn Ile 275 280 285Pro His His Phe Asp
Ala Met Pro Gly Val Gly Thr Asp Ala Gly Ala 290 295 300Ile His Leu
Thr Gly Ile Gly Val Pro Ser Leu Thr Ile Ala Ile Pro305 310 315
320Thr Arg Tyr Ile His Ser His Ala Ala Ile Leu His Arg Asp Asp Tyr
325 330 335Glu Asn Thr Val Lys Leu Leu Val Glu Val Ile Lys Arg Leu
Asp Ala 340 345 350Asp Lys Val Lys Gln Leu Thr Phe Asp Glu 355
36062484PRTVibrio cholera 62Met Glu Asp Lys Val Trp Ile Ser Met Gly
Ala Asp Ala Val Gly Ser1 5 10 15Leu Asn Pro Ala Leu Ser Glu Ser Leu
Leu Pro His Ser Phe Ala Ser 20 25 30Gly Ser Gln Val Trp Ile Gly Glu
Val Ala Ile Asp Glu Leu Ala Glu 35 40 45Leu Ser His Thr Met His Glu
Gln His Asn Arg Cys Gly Gly Tyr Met 50 55 60Val His Thr Ser Ala Gln
Gly Ala Met Ala Ala Leu Met Met Pro Glu65 70 75 80Ser Ile Ala Asn
Phe Thr Ile Pro Ala Pro Ser Gln Gln Asp Leu Val 85 90 95Asn Ala Trp
Leu Pro Gln Val Ser Ala Asp Gln Ile Thr Asn Thr Ile 100 105 110Arg
Ala Leu Ser Ser Phe Asn Asn Arg Phe Tyr Thr Thr Thr Ser Gly 115 120
125Ala Gln Ala Ser Asp Trp Leu Ala Asn Glu Trp Arg Ser Leu Ile Ser
130 135 140Ser Leu Pro Gly Ser Arg Ile Glu Gln Ile Lys His Ser Gly
Tyr Asn145 150 155 160Gln Lys Ser Val Val Leu Thr Ile Gln Gly Ser
Glu Lys Pro Asp Glu 165 170 175Trp Val Ile Val Gly Gly His Leu Asp
Ser Thr Leu Gly Ser His Thr 180 185 190Asn Glu Gln Ser Ile Ala Pro
Gly Ala Asp Asp Asp Ala Ser Gly Ile 195 200 205Ala Ser Leu Ser Glu
Ile Ile Arg Val Leu Arg Asp Asn Asn Phe Arg 210 215 220Pro Lys Arg
Ser Val Ala Leu Met Ala Tyr Ala Ala Glu Glu Val Gly225 230 235
240Leu Arg Gly Ser Gln Asp Leu Ala Asn Gln Tyr Lys Ala Gln Gly Lys
245 250 255Lys Val Val Ser Val Leu Gln Leu Asp Met Thr Asn Tyr Arg
Gly Ser 260 265 270Ala Glu Asp Ile Val Phe Ile Thr Asp Tyr Thr Asp
Ser Asn Leu Thr 275 280 285Gln Phe Leu Thr Thr Leu Ile Asp Glu Tyr
Leu Pro Glu Leu Thr Tyr 290 295 300Gly Tyr Asp Arg Cys Gly Tyr Ala
Cys Ser Asp His Ala Ser Trp His305 310 315 320Lys Ala Gly Phe Ser
Ala Ala Met Pro Phe Glu Ser Lys Phe Lys Asp 325 330 335Tyr Asn Pro
Lys Ile His Thr Ser Gln Asp Thr Leu Ala Asn Ser Asp 340 345 350Pro
Thr Gly Asn His Ala Val Lys Phe Thr Lys Leu Gly Leu Ala Tyr 355 360
365Val Ile Glu Met Ala Asn Ala Gly Ser Ser Gln Val Pro Asp Asp Ser
370 375 380Val Leu Gln Asp Gly Thr Ala Lys Ile Asn Leu Ser Gly Ala
Arg Gly385 390 395 400Thr Gln Lys Arg Phe Thr Phe Glu Leu Ser Gln
Ser Lys Pro Leu Thr 405 410 415Ile Gln Thr Tyr Gly Gly Ser Gly Asp
Val Asp Leu Tyr Val Lys Tyr 420 425 430Gly Ser Ala Pro Ser Lys Ser
Asn Trp Asp Cys Arg Pro Tyr Gln Asn 435 440 445Gly Asn Arg Glu Thr
Cys Ser Phe Asn Asn Ala Gln Pro Gly Ile Tyr 450 455 460His Val Met
Leu Asp Gly Tyr Thr Asn Tyr Asn Asp Val Ala Leu Lys465 470 475
480Ala Ser Thr Gln63488PRTPhotobacterium halotolerans 63Met Glu Asp
Lys Val Trp Ile Ser Ile Gly Ser Asp Ala Ser Gln Thr1 5 10 15Val Lys
Ser Val Met Gln Ser Asn Ala Arg Ser Leu Leu Pro Glu Ser 20 25 30Leu
Ala Ser Asn Gly Pro Val Trp Val Gly Gln Val Asp Tyr Ser Gln 35 40
45Leu Ala Glu Leu Ser His His Met His Glu Asp His Gln Arg Cys Gly
50 55 60Gly Tyr Met Val His Ser Ser Pro Glu Ser Ala Ile Ala Ala Ser
Asn65 70 75 80Met Pro Gln Ser Leu Val Ala Phe Ser Ile Pro Glu Ile
Ser Gln Gln 85 90 95Asp Thr Val Asn Ala Trp Leu Pro Gln Val Asn Ser
Gln Ala Ile Thr 100 105 110Gly Thr Ile Thr Ser Leu Thr Ser Phe Ile
Asn Arg Phe Tyr Thr Thr 115 120 125Thr Ser Gly Ala Gln Ala Ser Asp
Trp Leu Ala Asn Glu Trp Arg Ser 130 135 140Leu Ser Ala Ser Leu Pro
Asn Ala Ser Val Arg Gln Val Ser His Phe145 150 155 160Gly Tyr Asn
Gln Lys Ser Val Val Leu Thr Ile Thr Gly Ser Glu Lys 165 170 175Pro
Asp Glu Trp Ile Val Leu Gly Gly His Leu Asp Ser Thr Ile Gly 180 185
190Ser His Thr Asn Glu Gln Ser Val Ala Pro Gly Ala Asp Asp
Asp Ala 195 200 205Ser Gly Ile Ala Ser Val Thr Glu Ile Ile Arg Val
Leu Ser Glu Asn 210 215 220Asn Phe Gln Pro Lys Arg Ser Ile Ala Phe
Met Ala Tyr Ala Ala Glu225 230 235 240Glu Val Gly Leu Arg Gly Ser
Gln Asp Leu Ala Asn Gln Tyr Lys Ala 245 250 255Glu Gly Lys Gln Val
Ile Ser Ala Leu Gln Leu Asp Met Thr Asn Tyr 260 265 270Lys Gly Ser
Val Glu Asp Ile Val Phe Ile Thr Asp Tyr Thr Asp Ser 275 280 285Asn
Leu Thr Thr Phe Leu Ser Gln Leu Val Asp Glu Tyr Leu Pro Ser 290 295
300Leu Thr Tyr Gly Phe Asp Thr Cys Gly Tyr Ala Cys Ser Asp His
Ala305 310 315 320Ser Trp His Lys Ala Gly Phe Ser Ala Ala Met Pro
Phe Glu Ala Lys 325 330 335Phe Asn Asp Tyr Asn Pro Met Ile His Thr
Pro Asn Asp Thr Leu Gln 340 345 350Asn Ser Asp Pro Thr Ala Ser His
Ala Val Lys Phe Thr Lys Leu Gly 355 360 365Leu Ala Tyr Ala Ile Glu
Met Ala Ser Thr Thr Gly Gly Thr Pro Pro 370 375 380Pro Thr Gly Asn
Val Leu Lys Asp Gly Val Pro Val Asn Gly Leu Ser385 390 395 400Gly
Ala Thr Gly Ser Gln Val His Tyr Ser Phe Glu Leu Pro Ala Gln 405 410
415Lys Asn Leu Gln Ile Ser Thr Ala Gly Gly Ser Gly Asp Val Asp Leu
420 425 430Tyr Val Ser Phe Gly Ser Glu Ala Thr Lys Gln Asn Trp Asp
Cys Arg 435 440 445Pro Tyr Arg Asn Gly Asn Asn Glu Val Cys Thr Phe
Ala Gly Ala Thr 450 455 460Pro Gly Thr Tyr Ser Ile Met Leu Asp Gly
Tyr Arg Gln Phe Ser Gly465 470 475 480Val Thr Leu Lys Ala Ser Thr
Gln 48564871PRTYersinia pestis 64Met Thr Gln Gln Pro Gln Ala Lys
Tyr Arg His Asp Tyr Arg Ala Pro1 5 10 15Asp Tyr Thr Ile Thr Asp Ile
Asp Leu Asp Phe Ala Leu Asp Ala Gln 20 25 30Lys Thr Thr Val Thr Ala
Val Ser Lys Val Lys Arg Gln Gly Thr Asp 35 40 45Val Thr Pro Leu Ile
Leu Asn Gly Glu Asp Leu Thr Leu Ile Ser Val 50 55 60Ser Val Asp Gly
Gln Ala Trp Pro His Tyr Arg Gln Gln Asp Asn Thr65 70 75 80Leu Val
Ile Glu Gln Leu Pro Ala Asp Phe Thr Leu Thr Ile Val Asn 85 90 95Asp
Ile His Pro Ala Thr Asn Ser Ala Leu Glu Gly Leu Tyr Leu Ser 100 105
110Gly Glu Ala Leu Cys Thr Gln Cys Glu Ala Glu Gly Phe Arg His Ile
115 120 125Thr Tyr Tyr Leu Asp Arg Pro Asp Val Leu Ala Arg Phe Thr
Thr Arg 130 135 140Ile Val Ala Asp Lys Ser Arg Tyr Pro Tyr Leu Leu
Ser Asn Gly Asn145 150 155 160Arg Val Gly Gln Gly Glu Leu Asp Asp
Gly Arg His Trp Val Lys Trp 165 170 175Glu Asp Pro Phe Pro Lys Pro
Ser Tyr Leu Phe Ala Leu Val Ala Gly 180 185 190Asp Phe Asp Val Leu
Gln Asp Lys Phe Ile Thr Arg Ser Gly Arg Glu 195 200 205Val Ala Leu
Glu Ile Phe Val Asp Arg Gly Asn Leu Asp Arg Ala Asp 210 215 220Trp
Ala Met Thr Ser Leu Lys Asn Ser Met Lys Trp Asp Glu Thr Arg225 230
235 240Phe Gly Leu Glu Tyr Asp Leu Asp Ile Tyr Met Ile Val Ala Val
Asp 245 250 255Phe Phe Asn Met Gly Ala Met Glu Asn Lys Gly Leu Asn
Val Phe Asn 260 265 270Ser Lys Tyr Val Leu Ala Lys Ala Glu Thr Ala
Thr Asp Lys Asp Tyr 275 280 285Leu Asn Ile Glu Ala Val Ile Gly His
Glu Tyr Phe His Asn Trp Thr 290 295 300Gly Asn Arg Val Thr Cys Arg
Asp Trp Phe Gln Leu Ser Leu Lys Glu305 310 315 320Gly Leu Thr Val
Phe Arg Asp Gln Glu Phe Ser Ser Asp Leu Gly Ser 325 330 335Arg Ser
Val Asn Arg Ile Glu Asn Val Arg Val Met Arg Ala Ala Gln 340 345
350Phe Ala Glu Asp Ala Ser Pro Met Ala His Ala Ile Arg Pro Asp Lys
355 360 365Val Ile Glu Met Asn Asn Phe Tyr Thr Leu Thr Val Tyr Glu
Lys Gly 370 375 380Ser Glu Val Ile Arg Met Met His Thr Leu Leu Gly
Glu Gln Gln Phe385 390 395 400Gln Ala Gly Met Arg Leu Tyr Phe Glu
Arg His Asp Gly Ser Ala Ala 405 410 415Thr Cys Asp Asp Phe Val Gln
Ala Met Glu Asp Val Ser Asn Val Asp 420 425 430Leu Ser Leu Phe Arg
Arg Trp Tyr Ser Gln Ser Gly Thr Pro Leu Leu 435 440 445Thr Val His
Asp Asp Tyr Asp Val Glu Lys Gln Gln Tyr His Leu Phe 450 455 460Val
Ser Gln Lys Thr Leu Pro Thr Ala Asp Gln Pro Glu Lys Leu Pro465 470
475 480Leu His Ile Pro Leu Asp Ile Glu Leu Tyr Asp Ser Lys Gly Asn
Val 485 490 495Ile Pro Leu Gln His Asn Gly Leu Pro Val His His Val
Leu Asn Val 500 505 510Thr Glu Ala Glu Gln Thr Phe Thr Phe Asp Asn
Val Ala Gln Lys Pro 515 520 525Ile Pro Ser Leu Leu Arg Glu Phe Ser
Ala Pro Val Lys Leu Asp Tyr 530 535 540Pro Tyr Ser Asp Gln Gln Leu
Thr Phe Leu Met Gln His Ala Arg Asn545 550 555 560Glu Phe Ser Arg
Trp Asp Ala Ala Gln Ser Leu Leu Ala Thr Tyr Ile 565 570 575Lys Leu
Asn Val Ala Lys Tyr Gln Gln Gln Gln Pro Leu Ser Leu Pro 580 585
590Ala His Val Ala Asp Ala Phe Arg Ala Ile Leu Leu Asp Glu His Leu
595 600 605Asp Pro Ala Leu Ala Ala Gln Ile Leu Thr Leu Pro Ser Glu
Asn Glu 610 615 620Met Ala Glu Leu Phe Thr Thr Ile Asp Pro Gln Ala
Ile Ser Thr Val625 630 635 640His Glu Ala Ile Thr Arg Cys Leu Ala
Gln Glu Leu Ser Asp Glu Leu 645 650 655Leu Ala Val Tyr Val Ala Asn
Met Thr Pro Val Tyr Arg Ile Glu His 660 665 670Gly Asp Ile Ala Lys
Arg Ala Leu Arg Asn Thr Cys Leu Asn Tyr Leu 675 680 685Ala Phe Gly
Asp Glu Glu Phe Ala Asn Lys Leu Val Ser Leu Gln Tyr 690 695 700His
Gln Ala Asp Asn Met Thr Asp Ser Leu Ala Ala Leu Ala Ala Ala705 710
715 720Val Ala Ala Gln Leu Pro Cys Arg Asp Glu Leu Leu Ala Ala Phe
Asp 725 730 735Val Arg Trp Asn His Asp Gly Leu Val Met Asp Lys Trp
Phe Ala Leu 740 745 750Gln Ala Thr Ser Pro Ala Ala Asn Val Leu Val
Gln Val Arg Thr Leu 755 760 765Leu Lys His Pro Ala Phe Ser Leu Ser
Asn Pro Asn Arg Thr Arg Ser 770 775 780Leu Ile Gly Ser Phe Ala Ser
Gly Asn Pro Ala Ala Phe His Ala Ala785 790 795 800Asp Gly Ser Gly
Tyr Gln Phe Leu Val Glu Ile Leu Ser Asp Leu Asn 805 810 815Thr Arg
Asn Pro Gln Val Ala Ala Arg Leu Ile Glu Pro Leu Ile Arg 820 825
830Leu Lys Arg Tyr Asp Ala Gly Arg Gln Ala Leu Met Arg Lys Ala Leu
835 840 845Glu Gln Leu Lys Thr Leu Asp Asn Leu Ser Gly Asp Leu Tyr
Glu Lys 850 855 860Ile Thr Lys Ala Leu Ala Ala865 87065483PRTVibrio
anguillarum 65Met Glu Glu Lys Val Trp Ile Ser Ile Gly Gly Asp Ala
Thr Gln Thr1 5 10 15Ala Leu Arg Ser Gly Ala Gln Ser Leu Leu Pro Glu
Asn Leu Ile Asn 20 25 30Gln Thr Ser Val Trp Val Gly Gln Val Pro Val
Ser Glu Leu Ala Thr 35 40 45Leu Ser His Glu Met His Glu Asn His Gln
Arg Cys Gly Gly Tyr Met 50 55 60Val His Pro Ser Ala Gln Ser Ala Met
Ser Val Ser Ala Met Pro Leu65 70 75 80Asn Leu Asn Ala Phe Ser Ala
Pro Glu Ile Thr Gln Gln Thr Thr Val 85 90 95Asn Ala Trp Leu Pro Ser
Val Ser Ala Gln Gln Ile Thr Ser Thr Ile 100 105 110Thr Thr Leu Thr
Gln Phe Lys Asn Arg Phe Tyr Thr Thr Ser Thr Gly 115 120 125Ala Gln
Ala Ser Asn Trp Ile Ala Asp His Trp Arg Ser Leu Ser Ala 130 135
140Ser Leu Pro Ala Ser Lys Val Glu Gln Ile Thr His Ser Gly Tyr
Asn145 150 155 160Gln Lys Ser Val Met Leu Thr Ile Thr Gly Ser Glu
Lys Pro Asp Glu 165 170 175Trp Val Val Ile Gly Gly His Leu Asp Ser
Thr Leu Gly Ser Arg Thr 180 185 190Asn Glu Ser Ser Ile Ala Pro Gly
Ala Asp Asp Asp Ala Ser Gly Ile 195 200 205Ala Gly Val Thr Glu Ile
Ile Arg Leu Leu Ser Glu Gln Asn Phe Arg 210 215 220Pro Lys Arg Ser
Ile Ala Phe Met Ala Tyr Ala Ala Glu Glu Val Gly225 230 235 240Leu
Arg Gly Ser Gln Asp Leu Ala Asn Arg Phe Lys Ala Glu Gly Lys 245 250
255Lys Val Met Ser Val Met Gln Leu Asp Met Thr Asn Tyr Gln Gly Ser
260 265 270Arg Glu Asp Ile Val Phe Ile Thr Asp Tyr Thr Asp Ser Asn
Phe Thr 275 280 285Gln Tyr Leu Thr Gln Leu Leu Asp Glu Tyr Leu Pro
Ser Leu Thr Tyr 290 295 300Gly Phe Asp Thr Cys Gly Tyr Ala Cys Ser
Asp His Ala Ser Trp His305 310 315 320Ala Val Gly Tyr Pro Ala Ala
Met Pro Phe Glu Ser Lys Phe Asn Asp 325 330 335Tyr Asn Pro Asn Ile
His Ser Pro Gln Asp Thr Leu Gln Asn Ser Asp 340 345 350Pro Thr Gly
Phe His Ala Val Lys Phe Thr Lys Leu Gly Leu Ala Tyr 355 360 365Val
Val Glu Met Gly Asn Ala Ser Thr Pro Pro Thr Pro Ser Asn Gln 370 375
380Leu Lys Asn Gly Val Pro Val Asn Gly Leu Ser Ala Ser Arg Asn
Ser385 390 395 400Lys Thr Trp Tyr Gln Phe Glu Leu Gln Glu Ala Gly
Asn Leu Ser Ile 405 410 415Val Leu Ser Gly Gly Ser Gly Asp Ala Asp
Leu Tyr Val Lys Tyr Gln 420 425 430Thr Asp Ala Asp Leu Gln Gln Tyr
Asp Cys Arg Pro Tyr Arg Ser Gly 435 440 445Asn Asn Glu Thr Cys Gln
Phe Ser Asn Ala Gln Pro Gly Arg Tyr Ser 450 455 460Ile Leu Leu His
Gly Tyr Asn Asn Tyr Ser Asn Ala Ser Leu Val Ala465 470 475 480Asn
Ala Gln66482PRTUnknownSalinivibrio spYCSC6 66Met Glu Asp Lys Lys
Val Trp Ile Ser Ile Gly Ala Asp Ala Gln Gln1 5 10 15Thr Ala Leu Ser
Ser Gly Ala Gln Pro Leu Leu Ala Gln Ser Val Ala 20 25 30His Asn Gly
Gln Ala Trp Ile Gly Glu Val Ser Glu Ser Glu Leu Ala 35 40 45Ala Leu
Ser His Glu Met His Glu Asn His His Arg Cys Gly Gly Tyr 50 55 60Ile
Val His Ser Ser Ala Gln Ser Ala Met Ala Ala Ser Asn Met Pro65 70 75
80Leu Ser Arg Ala Ser Phe Ile Ala Pro Ala Ile Ser Gln Gln Ala Leu
85 90 95Val Thr Pro Trp Ile Ser Gln Ile Asp Ser Ala Leu Ile Val Asn
Thr 100 105 110Ile Asp Arg Leu Thr Asp Phe Pro Asn Arg Phe Tyr Thr
Thr Thr Ser 115 120 125Gly Ala Gln Ala Ser Asp Trp Ile Lys Gln Arg
Trp Gln Ser Leu Ser 130 135 140Ala Gly Leu Ala Gly Ala Ser Val Thr
Gln Ile Ser His Ser Gly Tyr145 150 155 160Asn Gln Ala Ser Val Met
Leu Thr Ile Glu Gly Ser Glu Ser Pro Asp 165 170 175Glu Trp Val Val
Val Gly Gly His Leu Asp Ser Thr Ile Gly Ser Arg 180 185 190Thr Asn
Glu Gln Ser Ile Ala Pro Gly Ala Asp Asp Asp Ala Ser Gly 195 200
205Ile Ala Ala Val Thr Glu Val Ile Arg Val Leu Ala Gln Asn Asn Phe
210 215 220Gln Pro Lys Arg Ser Ile Ala Phe Val Ala Tyr Ala Ala Glu
Glu Val225 230 235 240Gly Leu Arg Gly Ser Gln Asp Val Ala Asn Gln
Phe Lys Gln Ala Gly 245 250 255Lys Asp Val Arg Gly Val Leu Gln Leu
Asp Met Thr Asn Tyr Gln Gly 260 265 270Ser Ala Glu Asp Ile Val Phe
Ile Thr Asp Tyr Thr Asp Asn Gln Leu 275 280 285Thr Gln Tyr Leu Thr
Gln Leu Leu Asp Glu Tyr Leu Pro Thr Leu Asn 290 295 300Tyr Gly Phe
Asp Thr Cys Gly Tyr Ala Cys Ser Asp His Ala Ser Trp305 310 315
320His Gln Val Gly Tyr Pro Ala Ala Met Pro Phe Glu Ala Lys Phe Asn
325 330 335Asp Tyr Asn Pro Asn Ile His Thr Pro Gln Asp Thr Leu Ala
Asn Ser 340 345 350Asp Ser Glu Gly Ala His Ala Ala Lys Phe Thr Lys
Leu Gly Leu Ala 355 360 365Tyr Thr Val Glu Leu Ala Asn Ala Asp Ser
Ser Pro Asn Pro Gly Asn 370 375 380Glu Leu Lys Leu Gly Glu Pro Ile
Asn Gly Leu Ser Gly Ala Arg Gly385 390 395 400Asn Glu Lys Tyr Phe
Asn Tyr Arg Leu Asp Gln Ser Gly Glu Leu Val 405 410 415Ile Arg Thr
Tyr Gly Gly Ser Gly Asp Val Asp Leu Tyr Val Lys Ala 420 425 430Asn
Gly Asp Val Ser Thr Gly Asn Trp Asp Cys Arg Pro Tyr Arg Ser 435 440
445Gly Asn Asp Glu Val Cys Arg Phe Asp Asn Ala Thr Pro Gly Asn Tyr
450 455 460Ala Val Met Leu Arg Gly Tyr Arg Thr Tyr Asp Asn Val Ser
Leu Ile465 470 475 480Val Glu67301PRTVibrio proteolyticus 67Met Pro
Pro Ile Thr Gln Gln Ala Thr Val Thr Ala Trp Leu Pro Gln1 5 10 15Val
Asp Ala Ser Gln Ile Thr Gly Thr Ile Ser Ser Leu Glu Ser Phe 20 25
30Thr Asn Arg Phe Tyr Thr Thr Thr Ser Gly Ala Gln Ala Ser Asp Trp
35 40 45Ile Ala Ser Glu Trp Gln Ala Leu Ser Ala Ser Leu Pro Asn Ala
Ser 50 55 60Val Lys Gln Val Ser His Ser Gly Tyr Asn Gln Lys Ser Val
Val Met65 70 75 80Thr Ile Thr Gly Ser Glu Ala Pro Asp Glu Trp Ile
Val Ile Gly Gly 85 90 95His Leu Asp Ser Thr Ile Gly Ser His Thr Asn
Glu Gln Ser Val Ala 100 105 110Pro Gly Ala Asp Asp Asp Ala Ser Gly
Ile Ala Ala Val Thr Glu Val 115 120 125Ile Arg Val Leu Ser Glu Asn
Asn Phe Gln Pro Lys Arg Ser Ile Ala 130 135 140Phe Met Ala Tyr Ala
Ala Glu Glu Val Gly Leu Arg Gly Ser Gln Asp145 150 155 160Leu Ala
Asn Gln Tyr Lys Ser Glu Gly Lys Asn Val Val Ser Ala Leu 165 170
175Gln Leu Asp Met Thr Asn Tyr Lys Gly Ser Ala Gln Asp Val Val Phe
180 185 190Ile Thr Asp Tyr Thr Asp Ser Asn Phe Thr Gln Tyr Leu Thr
Gln Leu 195 200 205Met Asp Glu Tyr Leu Pro Ser Leu Thr Tyr Gly Phe
Asp Thr Cys Gly 210 215 220Tyr Ala Cys Ser Asp His Ala Ser Trp His
Asn Ala Gly Tyr Pro Ala225 230 235 240Ala Met Pro Phe Glu Ser Lys
Phe Asn Asp Tyr Asn Pro Arg Ile His 245 250 255Thr Thr Gln Asp Thr
Leu Ala Asn Ser Asp Pro Thr Gly Ser His Ala 260 265 270Lys Lys Phe
Thr Gln Leu Gly Leu Ala Tyr Ala Ile Glu Met Gly Ser 275 280 285Ala
Thr Gly Asp Thr Pro Thr Pro Gly Asn Gln Leu Glu 290 295
30068301PRTVibrio proteolyticus 68Met Pro Pro Ile Thr Gln Gln Ala
Thr Val Thr Ala Trp Leu Pro Gln1 5 10 15Val Asp Ala Ser Gln Ile Thr
Gly Thr Ile Ser Ser Leu Glu Ser Phe 20 25 30Thr Asn Arg
Phe Tyr Thr Thr Thr Ser Gly Ala Gln Ala Ser Asp Trp 35 40 45Ile Ala
Ser Glu Trp Gln Phe Leu Ser Ala Ser Leu Pro Asn Ala Ser 50 55 60Val
Lys Gln Val Ser His Ser Gly Tyr Asn Gln Lys Ser Val Val Met65 70 75
80Thr Ile Thr Gly Ser Glu Ala Pro Asp Glu Trp Ile Val Ile Gly Gly
85 90 95His Leu Asp Ser Thr Ile Gly Ser His Thr Asn Glu Gln Ser Val
Ala 100 105 110Pro Gly Ala Asp Asp Asp Ala Ser Gly Ile Ala Ala Val
Thr Glu Val 115 120 125Ile Arg Val Leu Ser Glu Asn Asn Phe Gln Pro
Lys Arg Ser Ile Ala 130 135 140Phe Met Ala Tyr Ala Ala Glu Glu Val
Gly Leu Arg Gly Ser Gln Asp145 150 155 160Leu Ala Asn Gln Tyr Lys
Ser Glu Gly Lys Asn Val Val Ser Ala Leu 165 170 175Gln Leu Asp Met
Thr Asn Tyr Lys Gly Ser Ala Gln Asp Val Val Phe 180 185 190Ile Thr
Asp Tyr Thr Asp Ser Asn Phe Thr Gln Tyr Leu Thr Gln Leu 195 200
205Met Asp Glu Tyr Leu Pro Ser Leu Thr Tyr Gly Phe Asp Thr Cys Gly
210 215 220Tyr Ala Cys Ser Asp His Ala Ser Trp His Asn Ala Gly Tyr
Pro Ala225 230 235 240Ala Met Pro Phe Glu Ser Lys Phe Asn Asp Tyr
Asn Pro Arg Ile His 245 250 255Thr Thr Gln Asp Thr Leu Ala Asn Ser
Asp Pro Thr Gly Ser His Ala 260 265 270Lys Lys Phe Thr Gln Leu Gly
Leu Ala Tyr Ala Ile Glu Met Gly Ser 275 280 285Ala Thr Gly Asp Thr
Pro Thr Pro Gly Asn Gln Leu Glu 290 295 30069348PRTP. furiosus
69Met Val Asp Trp Glu Leu Met Lys Lys Ile Ile Glu Ser Pro Gly Val1
5 10 15Ser Gly Tyr Glu His Leu Gly Ile Arg Asp Leu Val Val Asp Ile
Leu 20 25 30Lys Asp Val Ala Asp Glu Val Lys Ile Asp Lys Leu Gly Asn
Val Ile 35 40 45Ala His Phe Lys Gly Ser Ala Pro Lys Val Met Val Ala
Ala His Met 50 55 60Asp Lys Ile Gly Leu Met Val Asn His Ile Asp Lys
Asp Gly Tyr Leu65 70 75 80Arg Val Val Pro Ile Gly Gly Val Leu Pro
Glu Thr Leu Ile Ala Gln 85 90 95Lys Ile Arg Phe Phe Thr Glu Lys Gly
Glu Arg Tyr Gly Val Val Gly 100 105 110Val Leu Pro Pro His Leu Arg
Arg Glu Ala Lys Asp Gln Gly Gly Lys 115 120 125Ile Asp Trp Asp Ser
Ile Ile Val Asp Val Gly Ala Ser Ser Arg Glu 130 135 140Glu Ala Glu
Glu Met Gly Phe Arg Ile Gly Thr Ile Gly Glu Phe Ala145 150 155
160Pro Asn Phe Thr Arg Leu Ser Glu His Arg Phe Ala Thr Pro Tyr Leu
165 170 175Asp Asp Arg Ile Cys Leu Tyr Ala Met Ile Glu Ala Ala Arg
Gln Leu 180 185 190Gly Glu His Glu Ala Asp Ile Tyr Ile Val Ala Ser
Val Gln Glu Glu 195 200 205Ile Gly Leu Arg Gly Ala Arg Val Ala Ser
Phe Ala Ile Asp Pro Glu 210 215 220Val Gly Ile Ala Met Asp Val Thr
Phe Ala Lys Gln Pro Asn Asp Lys225 230 235 240Gly Lys Ile Val Pro
Glu Leu Gly Lys Gly Pro Val Met Asp Val Gly 245 250 255Pro Asn Ile
Asn Pro Lys Leu Arg Gln Phe Ala Asp Glu Val Ala Lys 260 265 270Lys
Tyr Glu Ile Pro Leu Gln Val Glu Pro Ser Pro Arg Pro Thr Gly 275 280
285Thr Asp Ala Asn Val Met Gln Ile Asn Arg Glu Gly Val Ala Thr Ala
290 295 300Val Leu Ser Ile Pro Ile Arg Tyr Met His Ser Gln Val Glu
Leu Ala305 310 315 320Asp Ala Arg Asp Val Asp Asn Thr Ile Lys Leu
Ala Lys Ala Leu Leu 325 330 335Glu Glu Leu Lys Pro Met Asp Phe Thr
Pro Leu Glu 340 345702PRTArtificial SequenceSynthetic Polypeptide
70Lys Phe1717PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 71Lys Lys Met Lys
Lys Met Xaa1 5727PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 72Asp Asp Met Asp
Asp Met Xaa1 5737PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 73Phe Phe Met Phe
Phe Met Xaa1 5747PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 74Ala Ala Met Ala
Ala Met Xaa1 5757PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 75Phe Pro Phe Pro
Phe Pro Xaa1 5767PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 76Asp Pro Asp Pro
Asp Pro Xaa1 5777PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 77Lys Pro Lys Pro
Lys Pro Xaa1 57813PRTArtificial SequenceSynthetic Polypeptide 78Tyr
Ala Ala Trp Ala Ala Phe Ala Asp Asp Asp Trp Lys1 5
107911PRTArtificial SequenceSynthetic Polypeptide 79Phe Tyr Pro Leu
Pro Trp Pro Asp Asp Asp Tyr1 5 108012PRTArtificial
SequenceSynthetic Polypeptide 80Phe Tyr Pro Leu Pro Trp Pro Asp Asp
Asp Tyr Lys1 5 108111PRTArtificial SequenceSynthetic Polypeptide
81Tyr Pro Leu Pro Trp Pro Asp Asp Asp Tyr Lys1 5 10827PRTArtificial
SequenceSynthetic Polypeptide 82Tyr Pro Tyr Pro Tyr Pro Lys1
5836PRTArtificial SequenceSynthetic Polypeptide 83Pro Tyr Pro Tyr
Pro Lys1 5844PRTArtificial SequenceSynthetic Polypeptide 84Gly Pro
Arg Pro1857PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 85Asp Asp Pro Asp
Asp Pro Xaa1 5867PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 86Tyr Pro Tyr Pro
Tyr Pro Xaa1 5876PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 87Pro Tyr Pro Tyr
Pro Xaa1 5886PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 88Pro Phe Pro Phe
Pro Xaa1 5897PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Lys-Triazole-PEG4 89Lys Lys
Met Lys Lys Met Xaa1 5907PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 90Ala Ala Pro Ala
Ala Pro Xaa1 5916PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 91Ala Pro Ala Ala
Pro Xaa1 5927PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 92Tyr Tyr Pro Tyr
Tyr Pro Xaa1 5936PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 93Tyr Pro Tyr Tyr
Pro Xaa1 5947PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 94Phe Phe Pro Phe
Phe Pro Xaa1 5956PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 95Phe Pro Phe Phe
Pro Xaa1 5967PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 96Arg Arg Pro Arg
Arg Pro Xaa1 5976PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 97Arg Pro Arg Arg
Pro Xaa1 5987PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 98Ala Ala Pro Ala
Ala Pro Xaa1 5996PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 99Ala Pro Ala Ala
Pro Xaa1 51007PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 100Lys Lys Pro
Lys Lys Pro Xaa1 51016PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 101Lys Pro Lys
Lys Pro Xaa1 51026PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(6)..(6)Xaa is Azidolysine 102Lys Met Lys
Lys Met Xaa1 51037PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(7)..(7)Xaa is Azidolysine 103Tyr Tyr Met
Tyr Tyr Met Xaa1 51048PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(8)..(8)Xaa is Azidolysine 104Asp Pro Ala
Ala Ala Phe Lys Xaa1 51056PRTArtificial SequenceSynthetic
Polypeptide 105Pro Ala Ala Ala Phe Lys1 510610PRTArtificial
SequenceSynthetic Polypeptidemisc_feature(10)..(10)Xaa is
Azidolysine 106Lys Ala Ala Ala Ala Ala Ala Phe Lys Xaa1 5
1010712PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(12)..(12)Xaa is Azidolysine 107Phe Tyr Pro
Leu Pro Trp Pro Asp Asp Asp Tyr Xaa1 5 1010811PRTArtificial
SequenceSynthetic Polypeptidemisc_feature(11)..(11)Xaa is
Azidolysine 108Tyr Pro Leu Pro Trp Pro Asp Asp Asp Tyr Xaa1 5
1010910PRTArtificial SequenceSynthetic
Polypeptidemisc_feature(10)..(10)Xaa is Azidolysine 109Pro Leu Pro
Trp Pro Asp Asp Asp Tyr Xaa1 5 101109PRTArtificial
SequenceSynthetic Polypeptidemisc_feature(9)..(9)Xaa is Azidolysine
110Leu Pro Trp Pro Asp Asp Asp Tyr Xaa1 511110PRTArtificial
SequenceSynthetic Polypeptide 111Phe Ala Ala Ala Trp Pro Asp Asp
Asp Phe1 5 101125PRTArtificial SequenceSynthetic Polypeptide 112Trp
Pro Asp Asp Phe1 511310PRTArtificial SequenceSynthetic Polypeptide
113Trp Ala Ala Ala Phe Pro Asp Asp Asp Phe1 5 101145PRTArtificial
SequenceSynthetic Polypeptide 114Phe Pro Asp Asp Phe1
51155PRTArtificial SequenceSynthetic Polypeptide 115Tyr Pro Asp Asp
Phe1 51166PRTArtificial SequenceSynthetic Polypeptide 116Arg Arg
Pro Phe Gln Gln1 51175PRTArtificial SequenceSynthetic Polypeptide
117Arg Pro Phe Gln Gln1 51186PRTArtificial SequenceSynthetic
Polypeptide 118Ala Ala Pro Phe Gln Gln1 51195PRTArtificial
SequenceSynthetic Polypeptide 119Ala Pro Phe Gln Gln1
51206PRTArtificial SequenceSynthetic Polypeptide 120Lys Lys Pro Phe
Gln Gln1 51215PRTArtificial SequenceSynthetic Polypeptide 121Lys
Pro Phe Gln Gln1 51226PRTArtificial SequenceSynthetic Polypeptide
122Tyr Tyr Pro Phe Gln Gln1 51235PRTArtificial SequenceSynthetic
Polypeptide 123Tyr Pro Phe Gln Gln1 51246PRTArtificial
SequenceSynthetic Polypeptide 124Phe Phe Pro Phe Gln Gln1
51255PRTArtificial SequenceSynthetic Polypeptide 125Phe Pro Phe Gln
Gln1 51266PRTArtificial SequenceSynthetic Polypeptide 126Asp Asp
Pro Phe Gln Gln1 51275PRTArtificial SequenceSynthetic Polypeptide
127Asp Pro Phe Gln Gln1 51286PRTArtificial SequenceSynthetic
Polypeptide 128Glu Glu Pro Phe Gln Gln1 51295PRTArtificial
SequenceSynthetic Polypeptide 129Glu Pro Phe Gln Gln1
51306PRTArtificial SequenceSynthetic Polypeptide 130Asn Asn Pro Phe
Gln Gln1 51315PRTArtificial SequenceSynthetic Polypeptide 131Asn
Pro Phe Gln Gln1 51326PRTArtificial SequenceSynthetic Polypeptide
132Gln Gln Pro Phe Gln Gln1 51335PRTArtificial SequenceSynthetic
Polypeptide 133Gln Pro Phe Gln Gln1 51346PRTArtificial
SequenceSynthetic Polypeptide 134Val Val Pro Phe Gln Gln1
51355PRTArtificial SequenceSynthetic Polypeptide 135Val Pro Phe Gln
Gln1 51366PRTArtificial SequenceSynthetic Polypeptide 136Ile Ile
Pro Phe Gln Gln1 51375PRTArtificial SequenceSynthetic Polypeptide
137Ile Pro Phe Gln Gln1 51386PRTArtificial SequenceSynthetic
Polypeptide 138Leu Leu Pro Phe Gln Gln1 51395PRTArtificial
SequenceSynthetic Polypeptide 139Leu Pro Phe Gln Gln1
51406PRTArtificial SequenceSynthetic Polypeptide 140Ser Ser Pro Phe
Gln Gln1 51415PRTArtificial SequenceSynthetic Polypeptide 141Ser
Pro Phe Gln Gln1 51426PRTArtificial SequenceSynthetic Polypeptide
142Thr Thr Pro Phe Gln Gln1 51435PRTArtificial SequenceSynthetic
Polypeptide 143Thr Pro Phe Gln Gln1 51446PRTArtificial
SequenceSynthetic Polypeptide 144Cys Cys Pro Phe Gln Gln1
51455PRTArtificial SequenceSynthetic Polypeptide 145Cys Pro Phe Gln
Gln1 51466PRTArtificial SequenceSynthetic Polypeptide 146Trp Trp
Pro Phe Gln Gln1 51475PRTArtificial SequenceSynthetic Polypeptide
147Trp Pro Phe Gln Gln1 51486PRTArtificial SequenceSynthetic
Polypeptide 148Met Met Pro Phe Gln Gln1 51495PRTArtificial
SequenceSynthetic Polypeptide 149Met Pro Phe Gln Gln1
51506PRTArtificial SequenceSynthetic Polypeptide 150Pro Pro Pro Phe
Gln Gln1 51515PRTArtificial SequenceSynthetic Polypeptide 151Pro
Pro Phe Gln Gln1 51526PRTArtificial SequenceSynthetic Polypeptide
152Gly Gly Pro Phe Gln Gln1 51535PRTArtificial SequenceSynthetic
Polypeptide 153Gly Pro Phe Gln Gln1 51546PRTArtificial
SequenceSynthetic Polypeptide 154His His Pro Phe Gln Gln1
51555PRTArtificial SequenceSynthetic Polypeptide 155His Pro Phe Gln
Gln1 515612PRTArtificial SequenceSynthetic Polypeptide 156Tyr Ala
Ala Phe Ala Ala Trp Ala Asp Asp Asp Trp1 5 101576PRTArtificial
SequenceSynthetic Polypeptide 157Ala Asp Asp Asp Trp Lys1
51587PRTArtificial SequenceSynthetic Polypeptide 158Trp Ala Asp Asp
Asp Trp Lys1 5












D00000
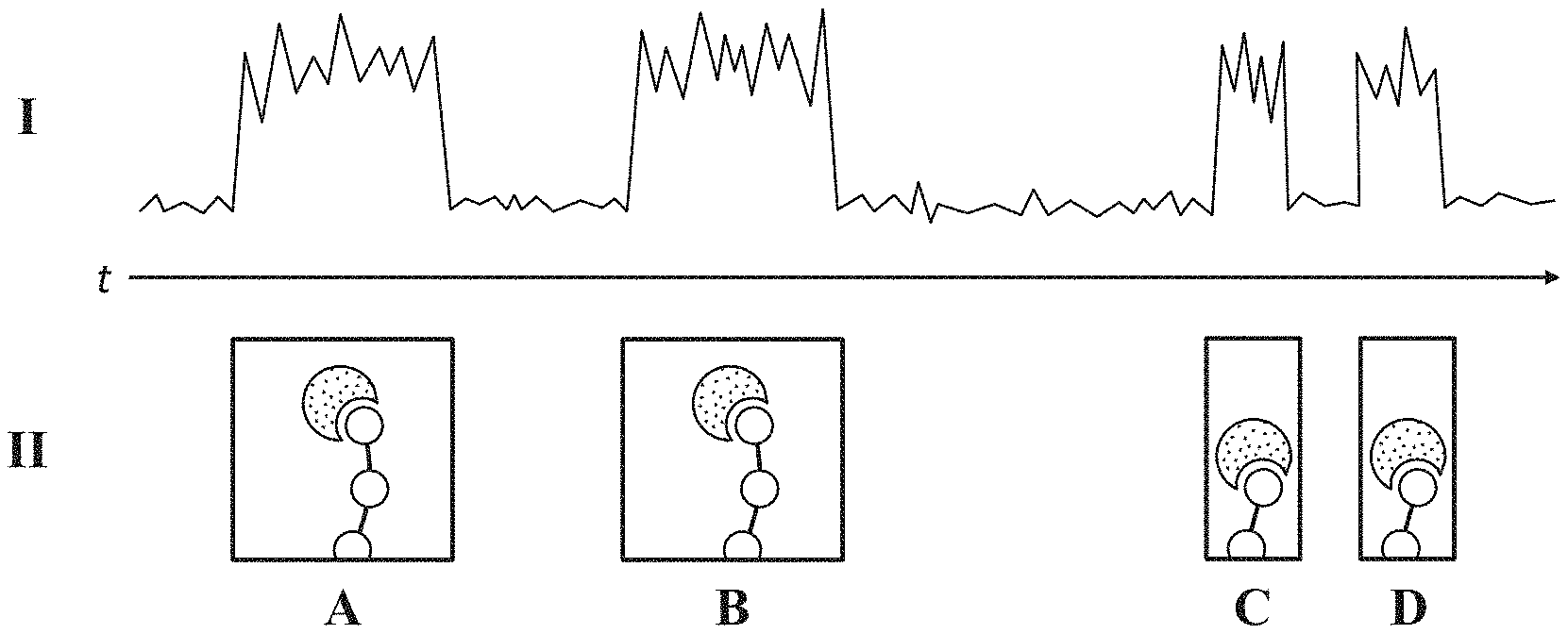
D00001

D00002

D00003

D00004
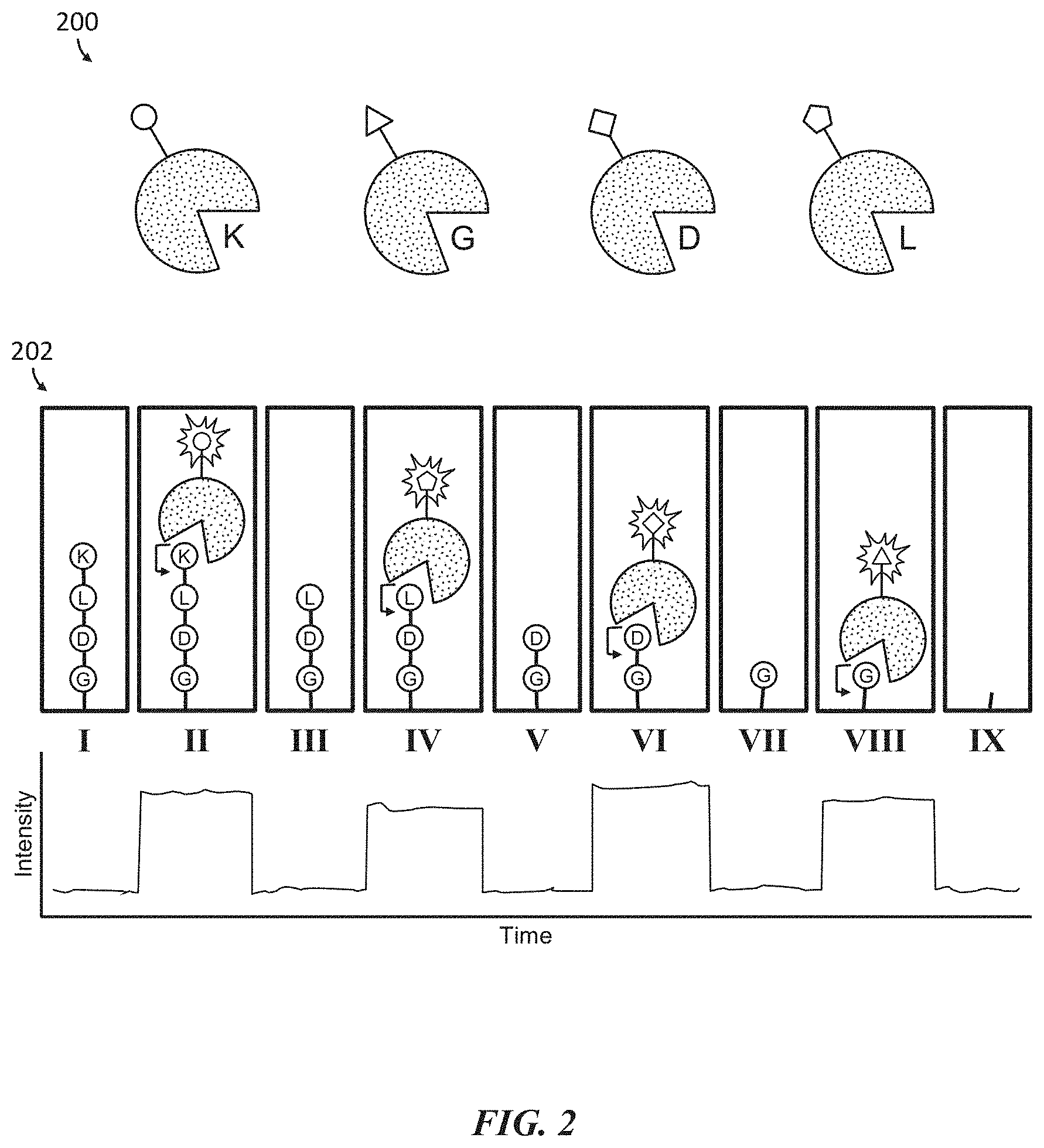
D00005

D00006

D00007
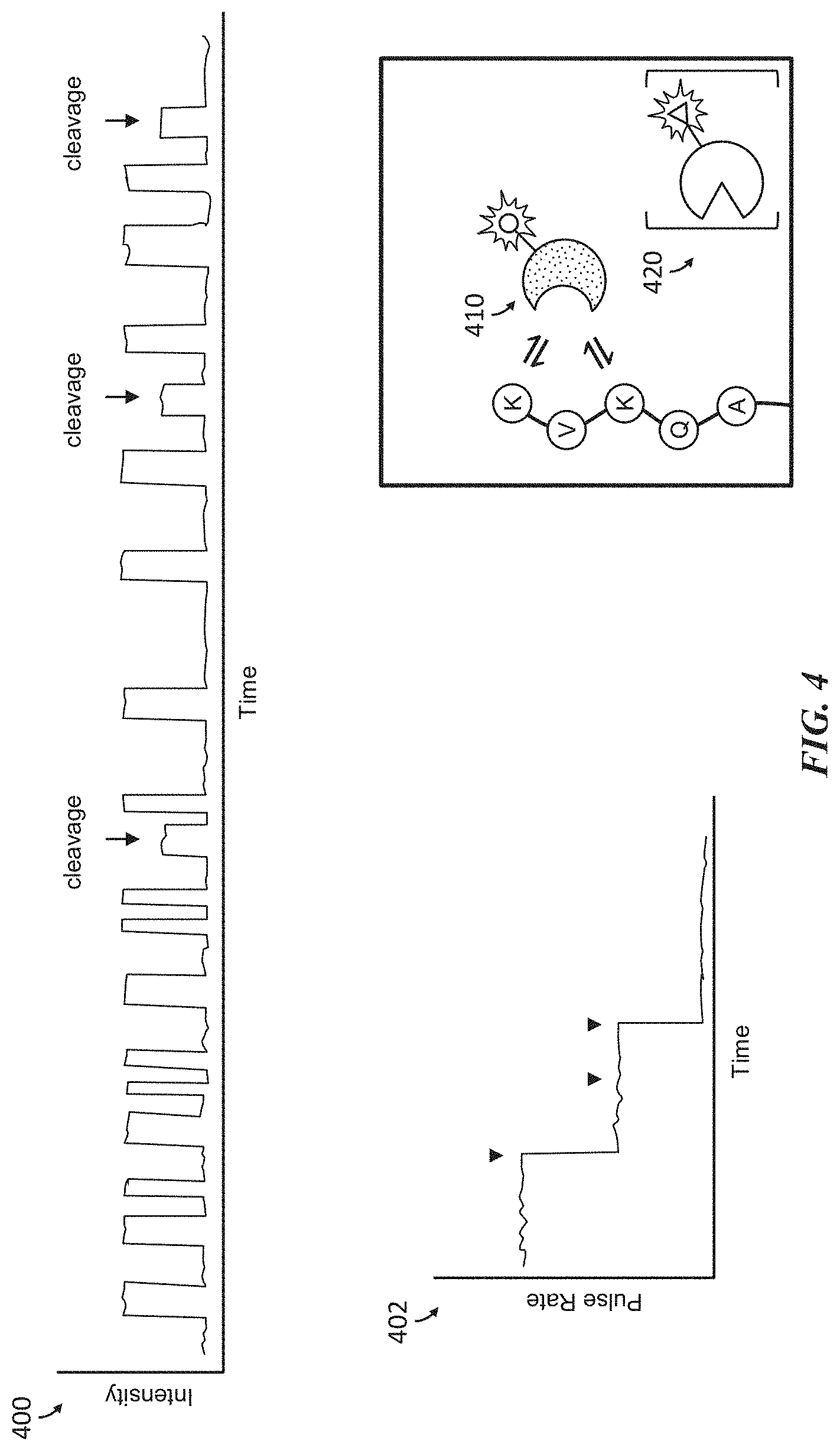
D00008

D00009

D00010
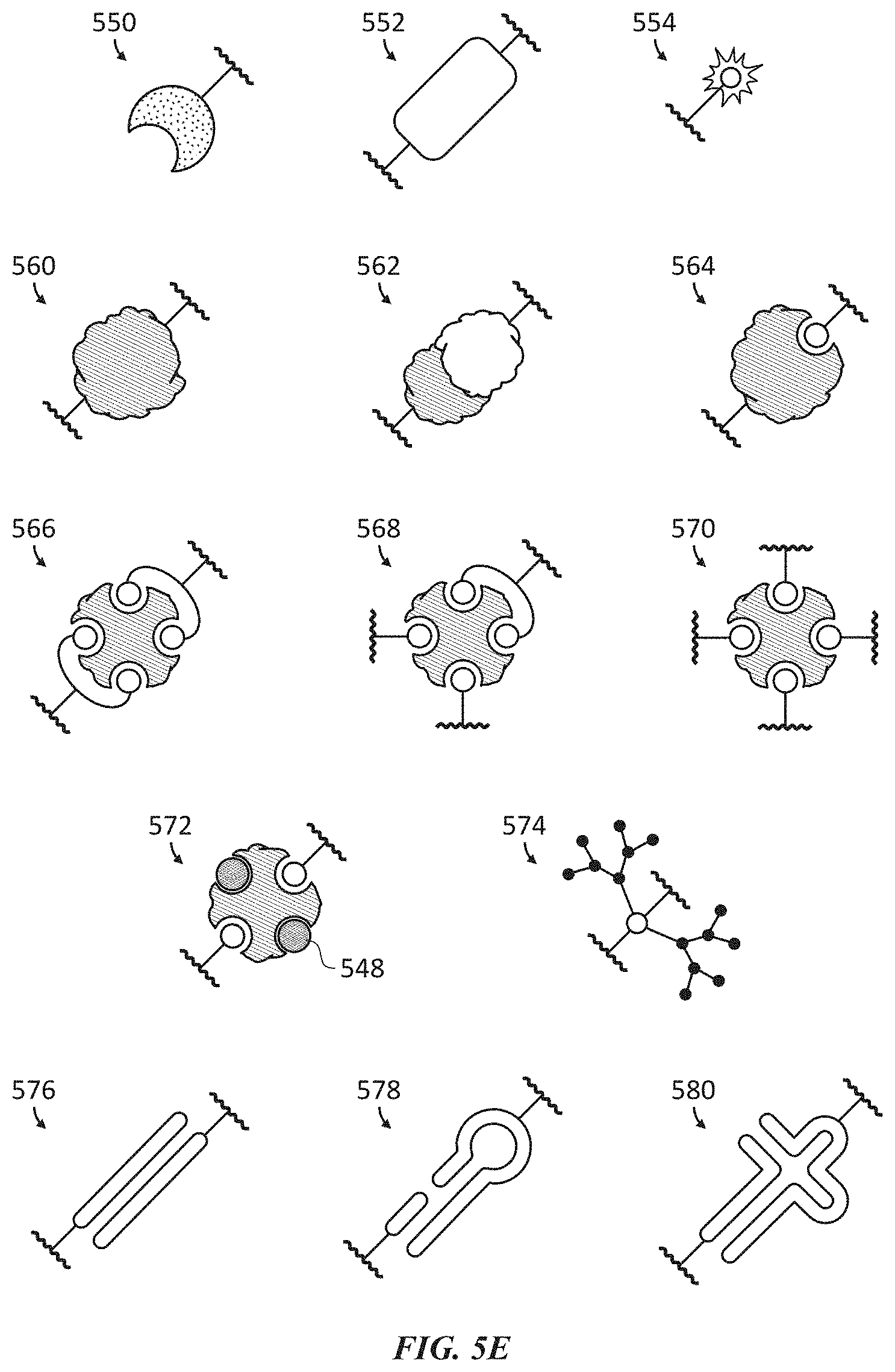
D00011

D00012
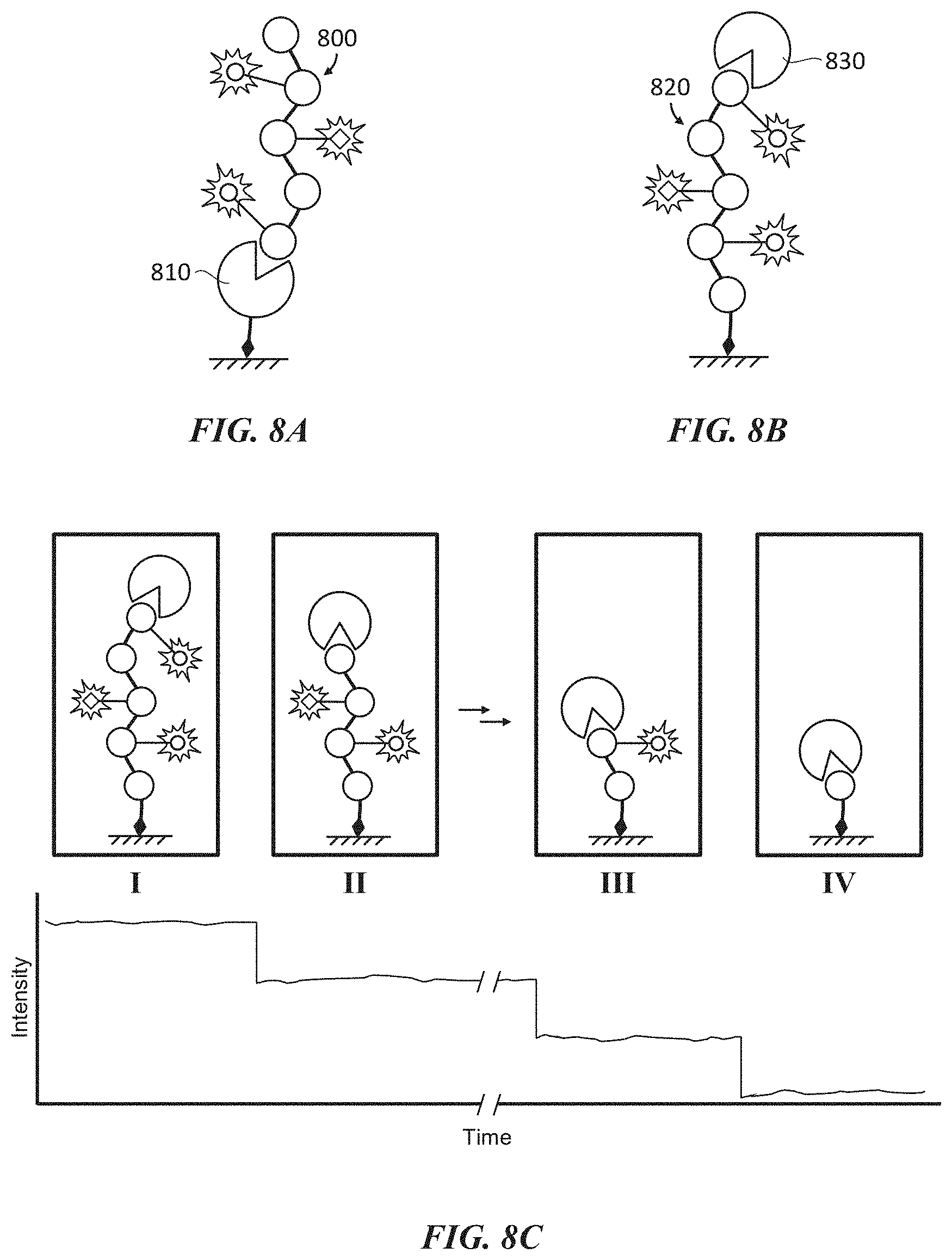
D00013
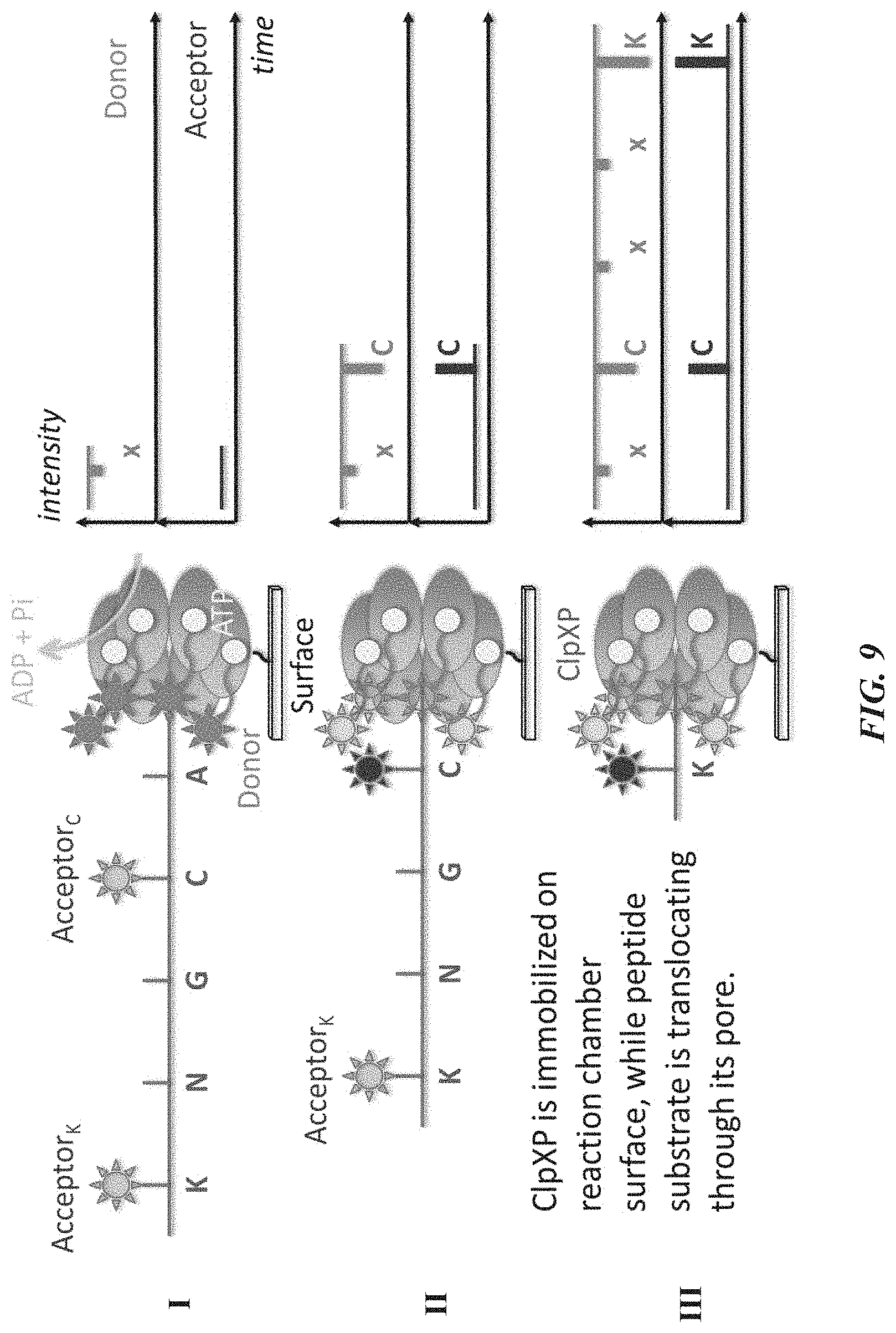
D00014
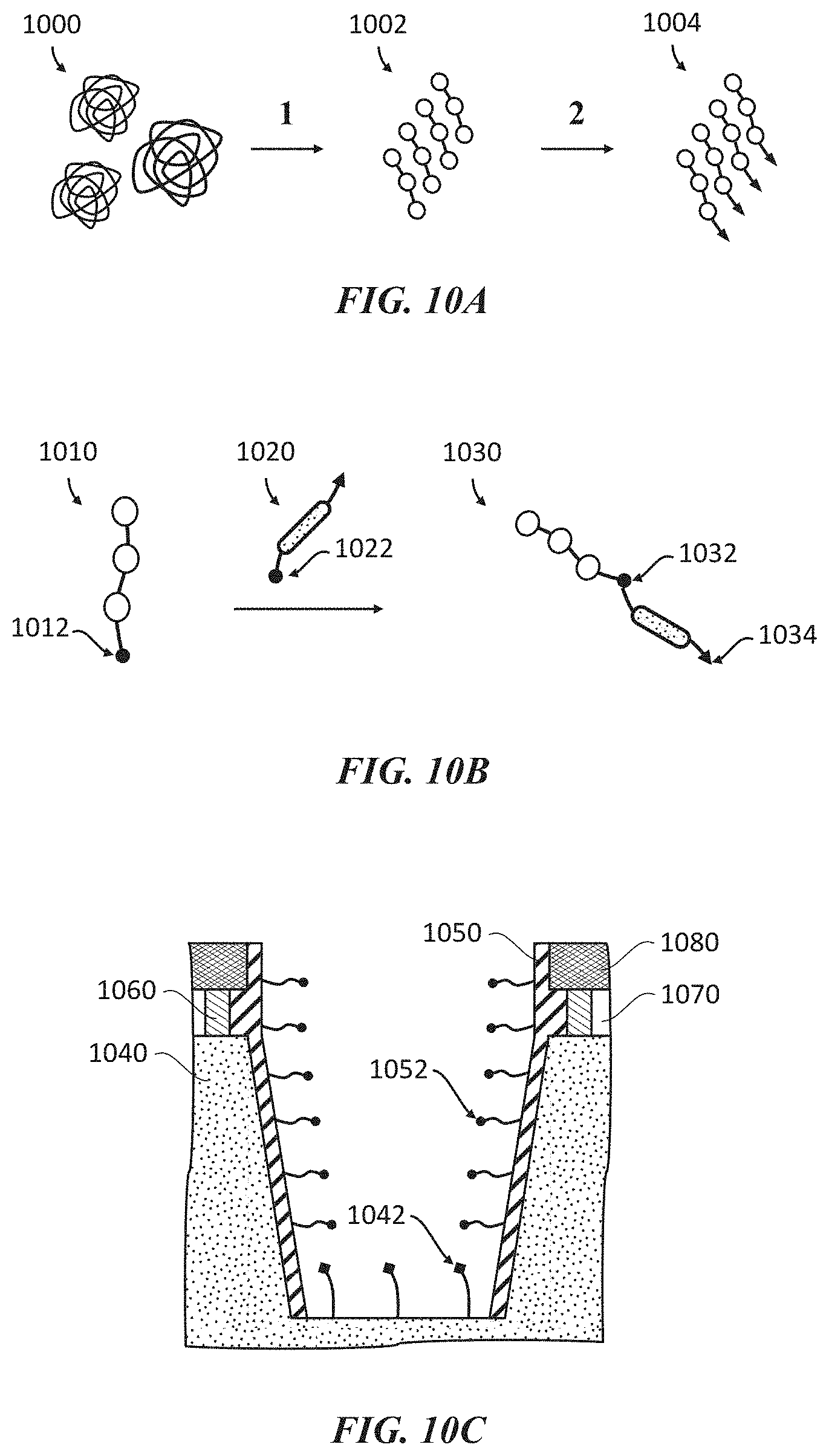
D00015

D00016

D00017
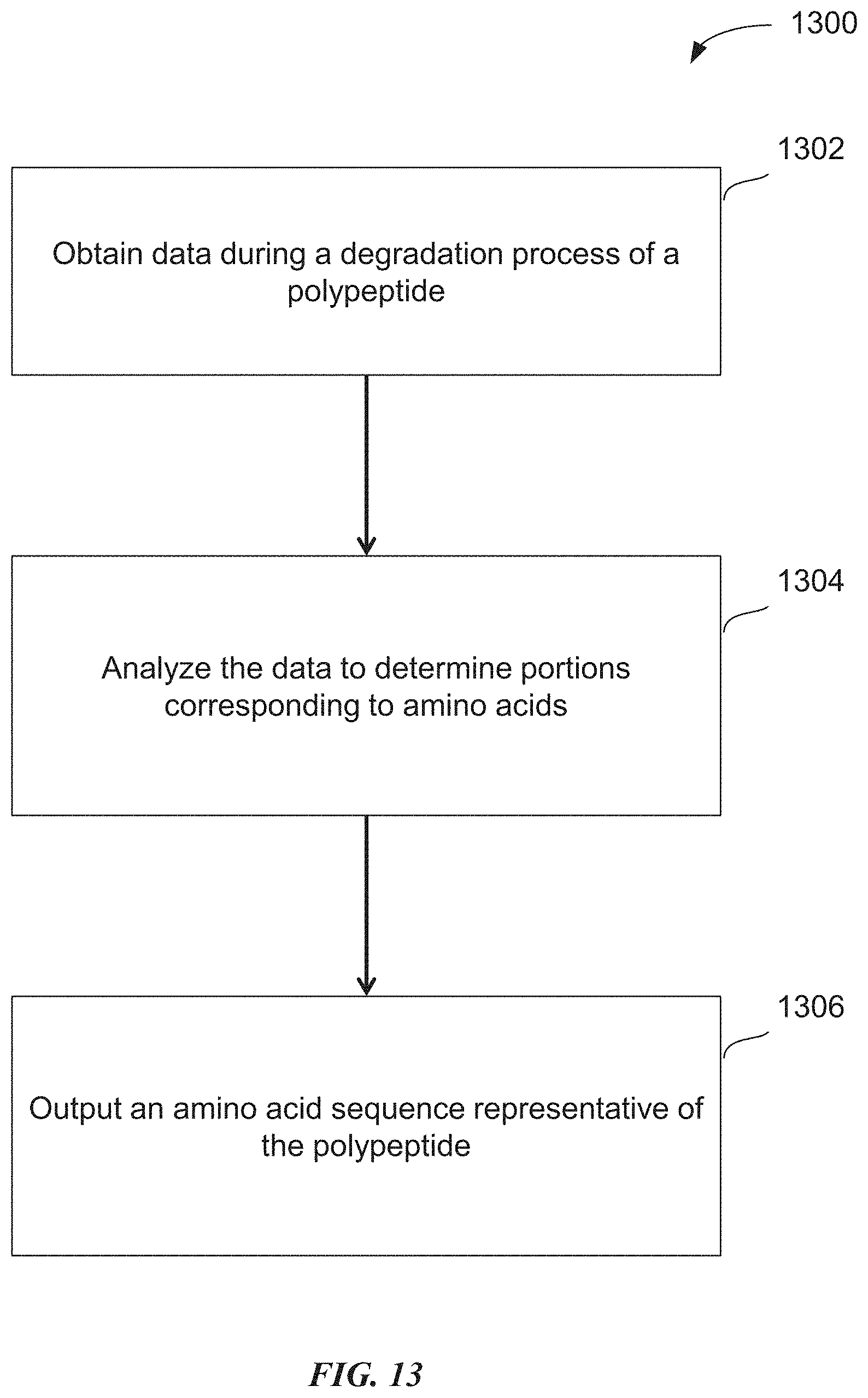
D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025
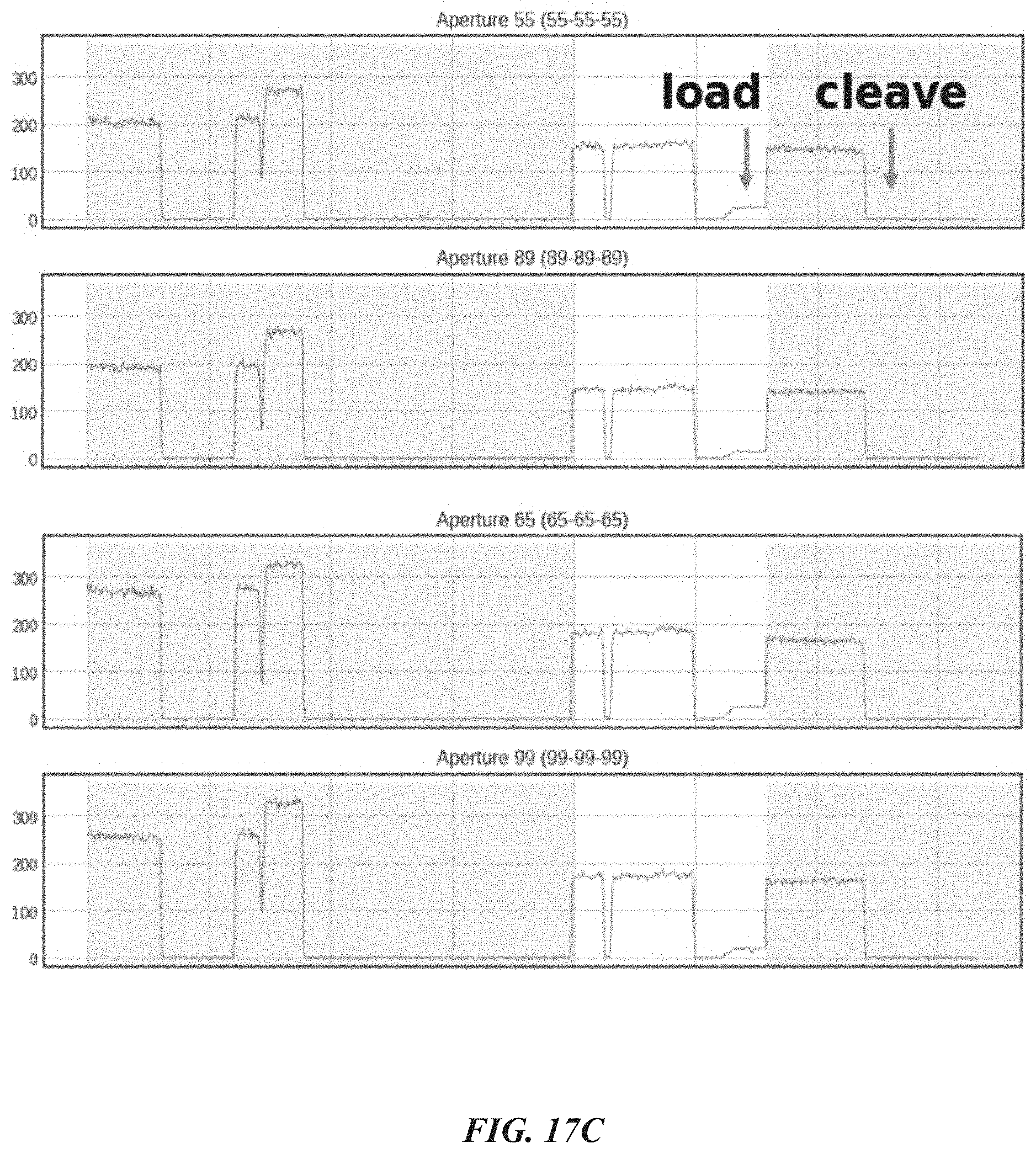
D00026
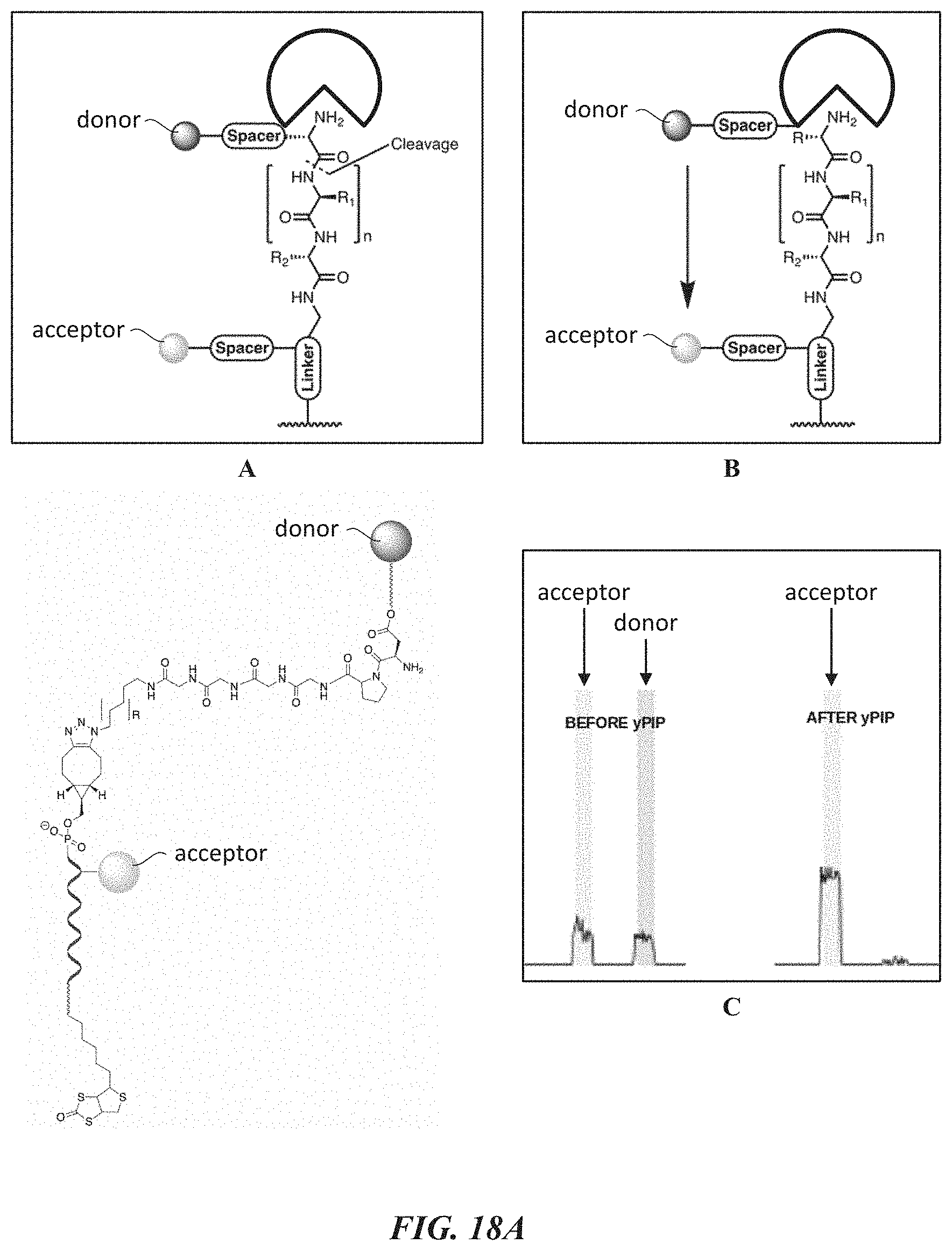
D00027
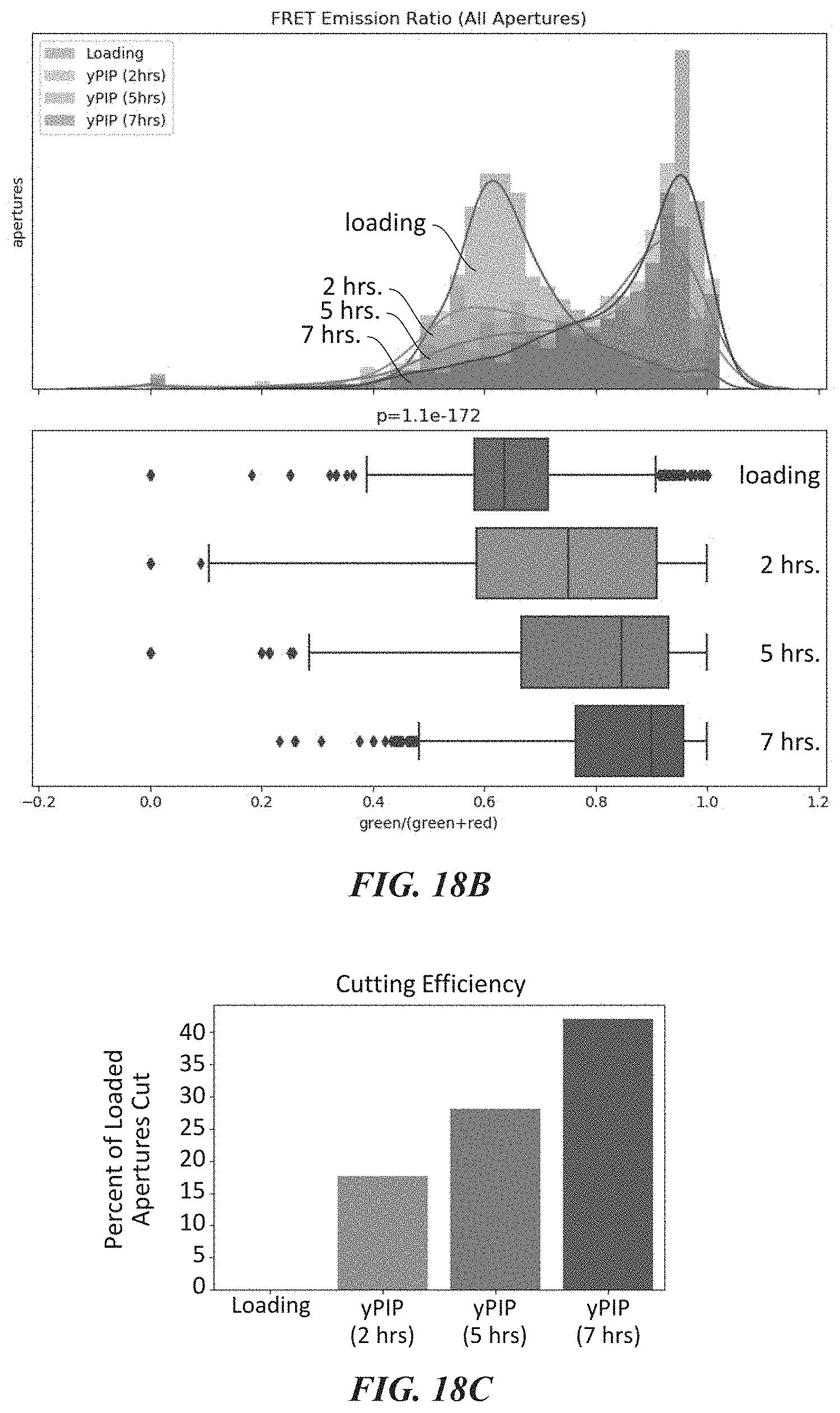
D00028
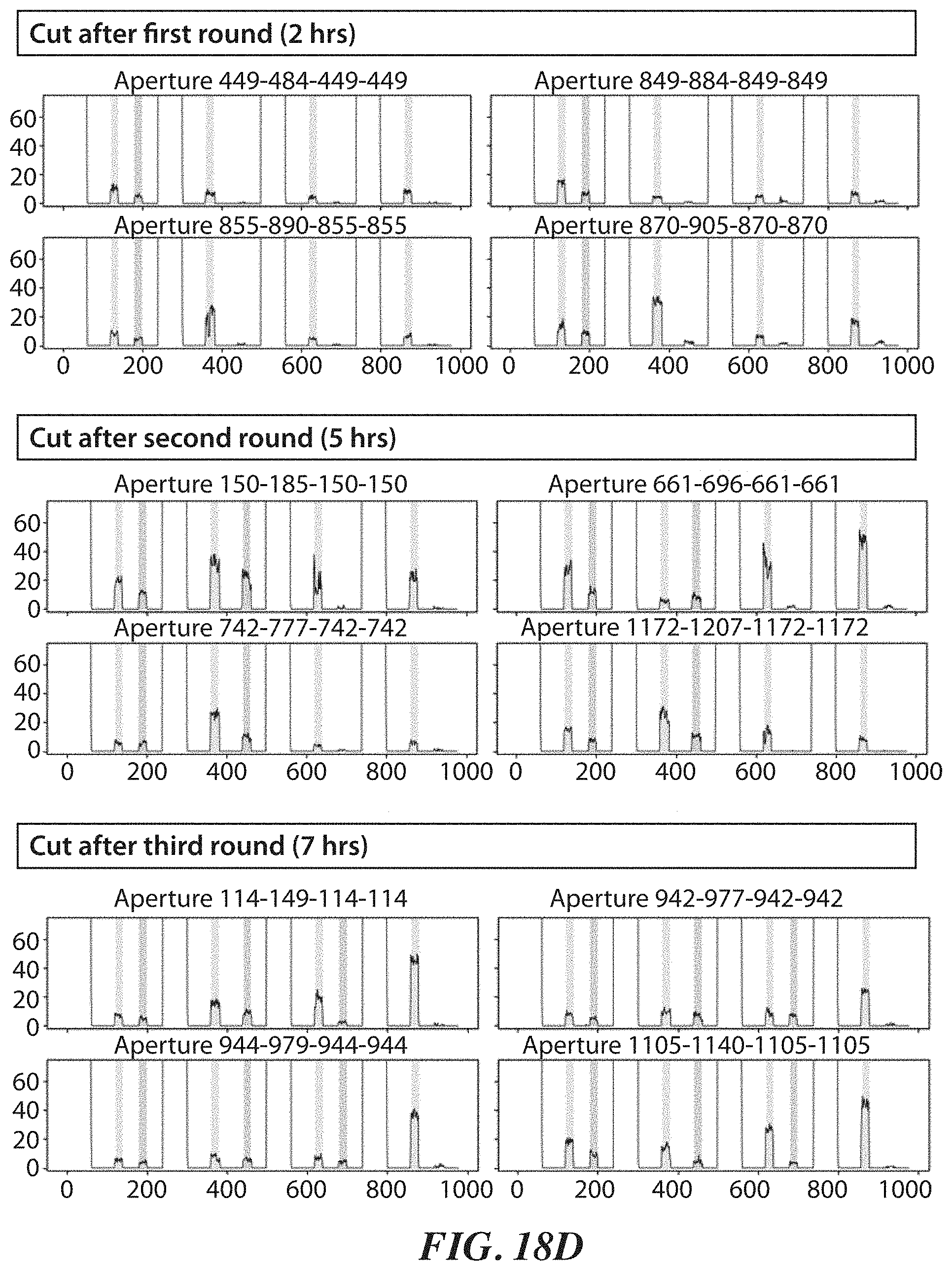
D00029

D00030

D00031
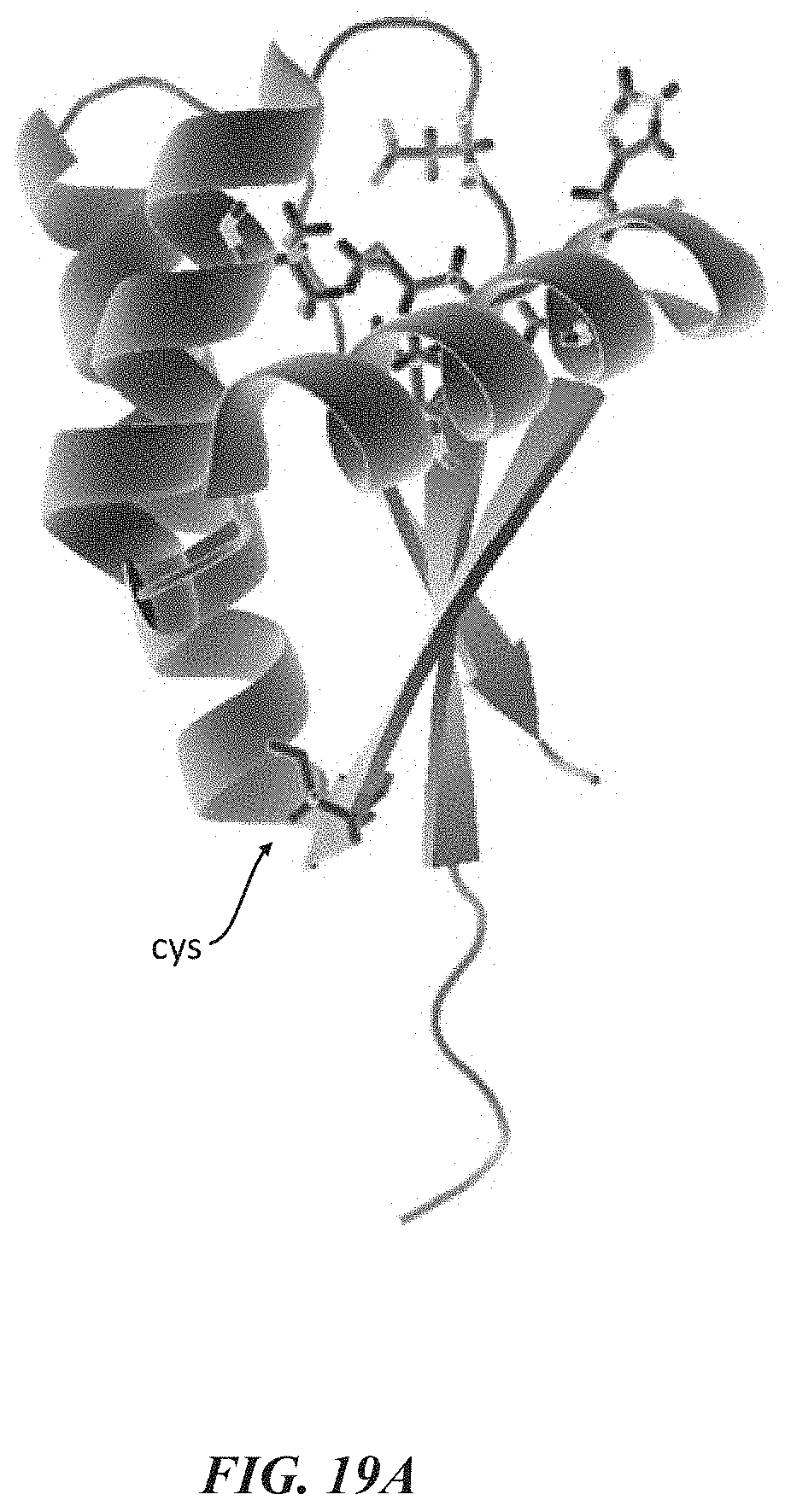
D00032

D00033
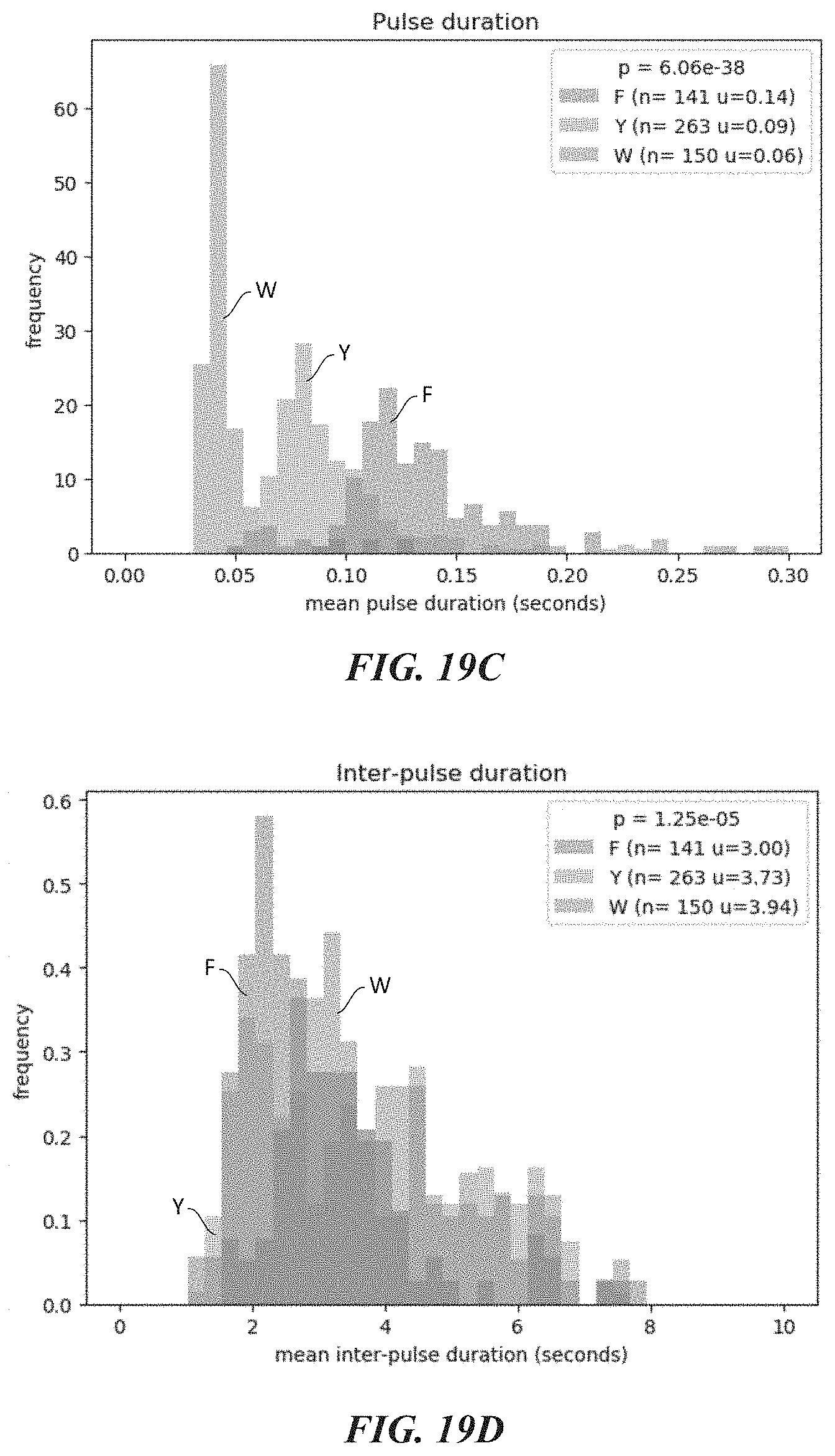
D00034
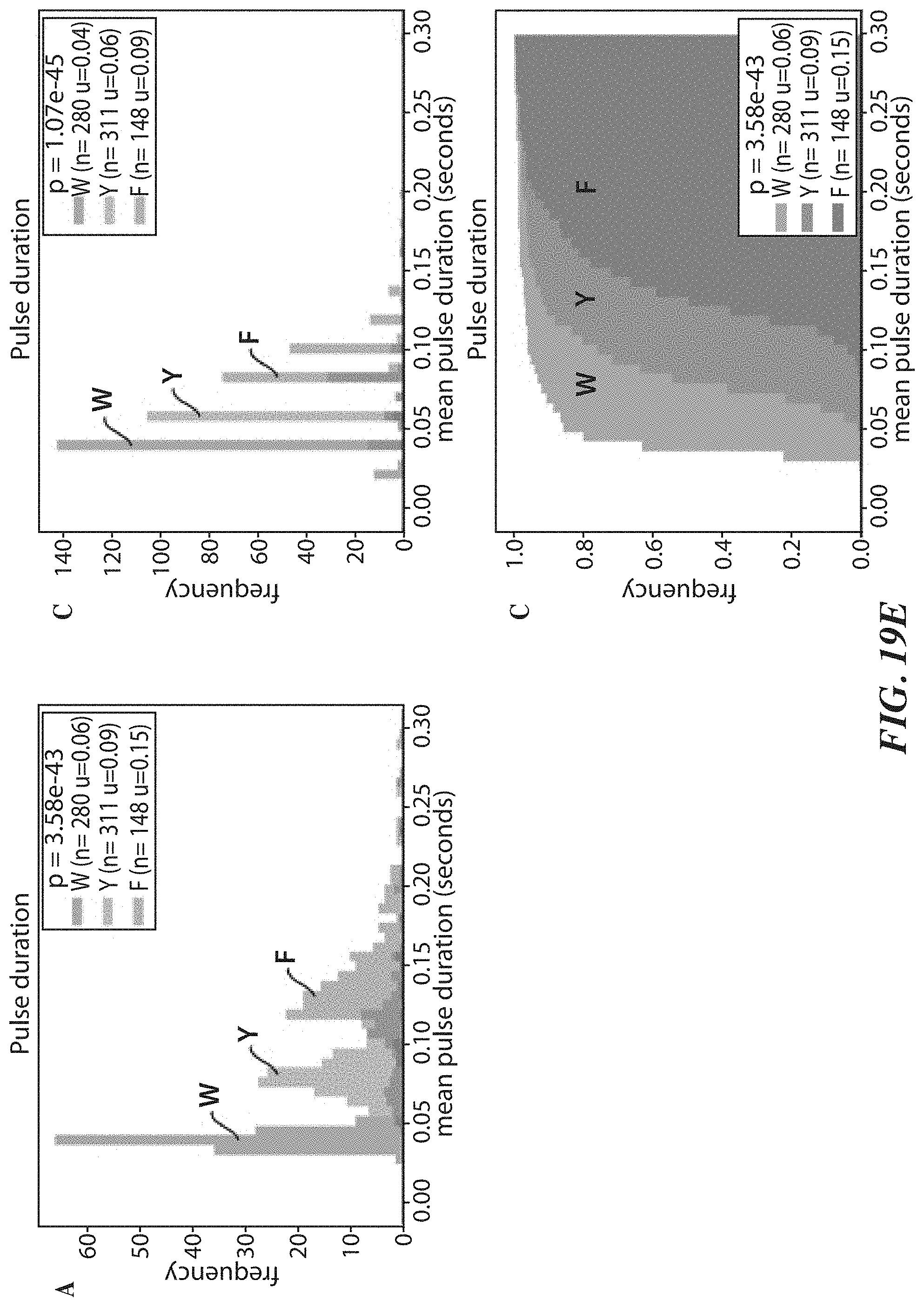
D00035
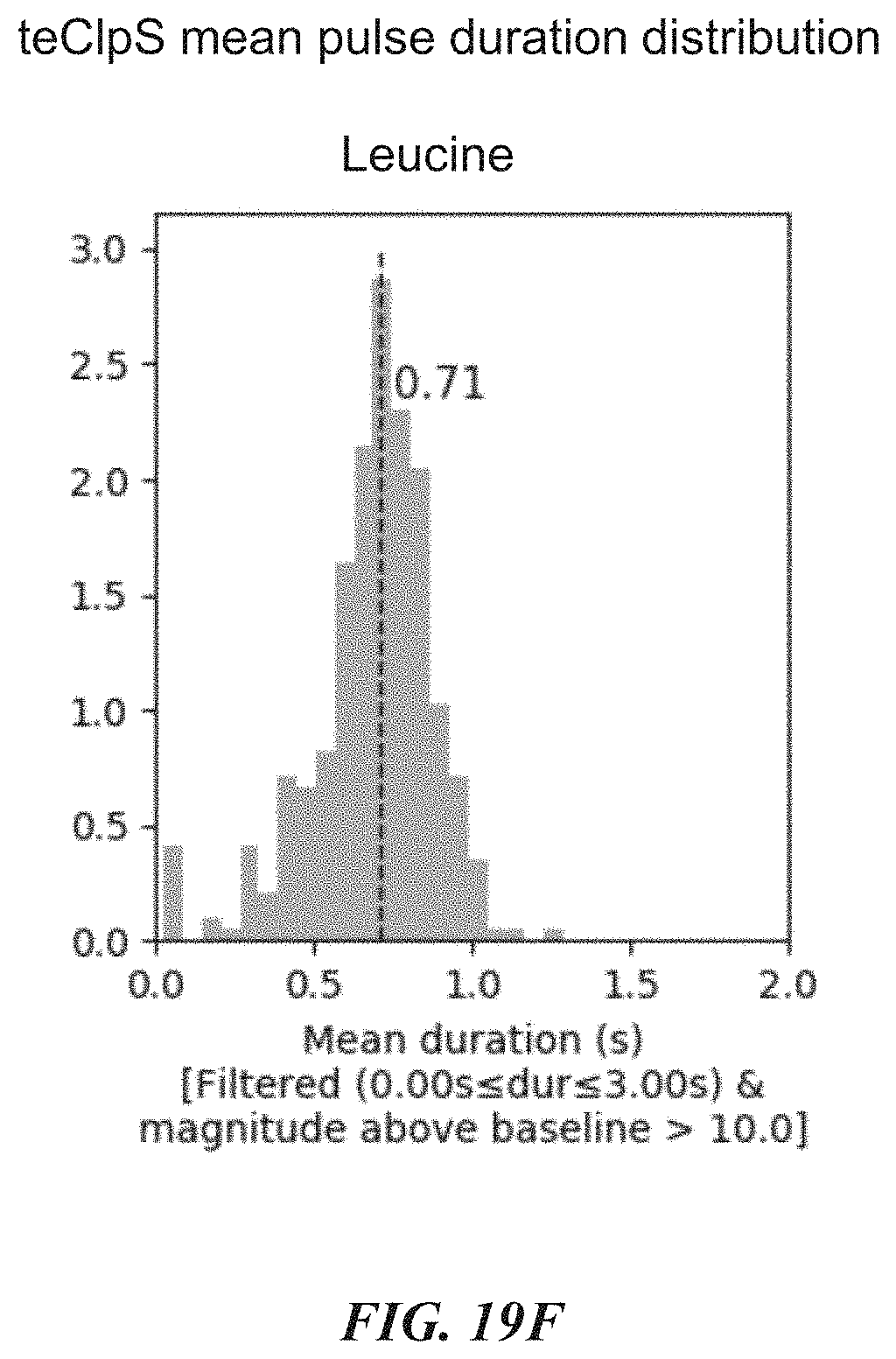
D00036

D00037
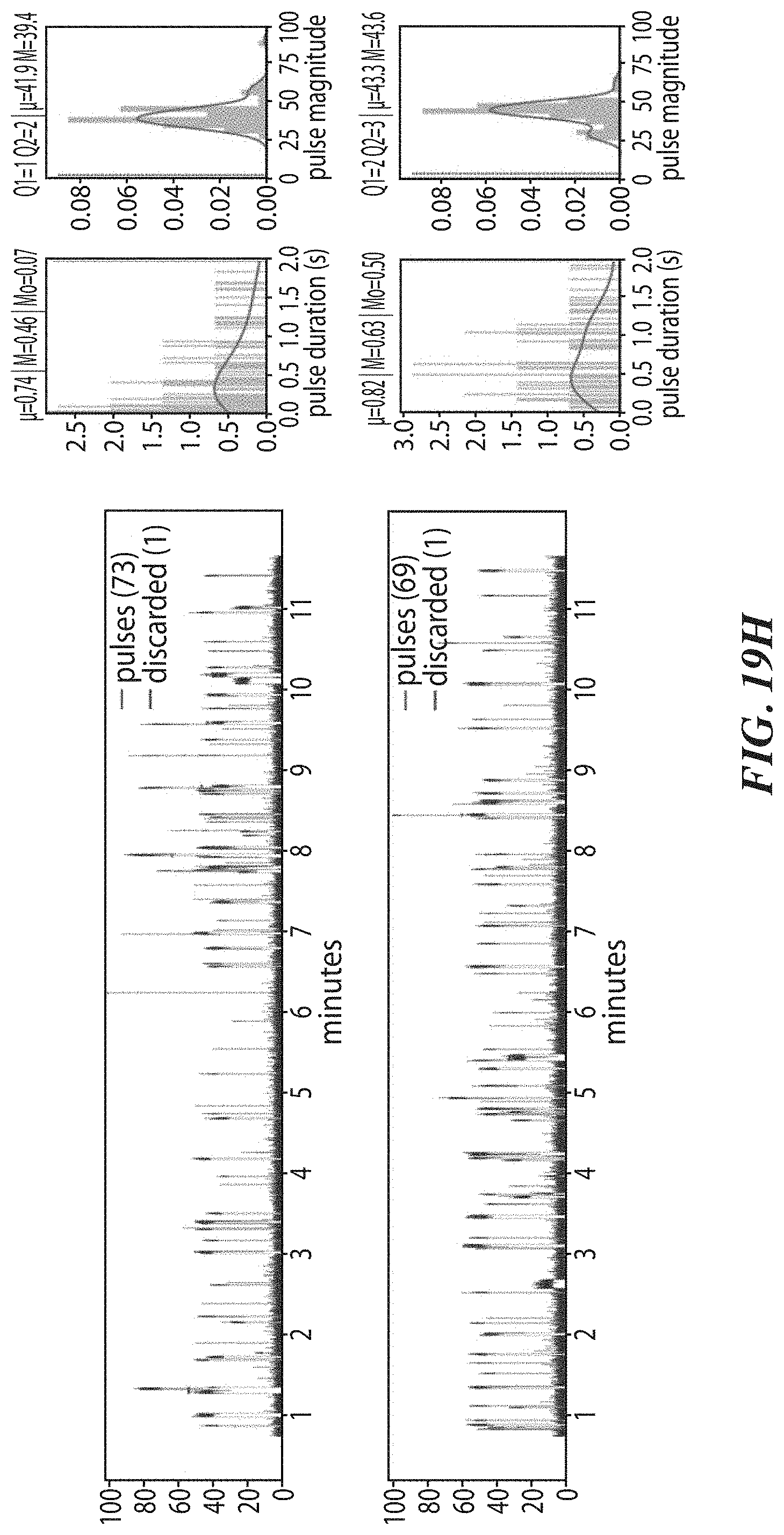
D00038

D00039

D00040
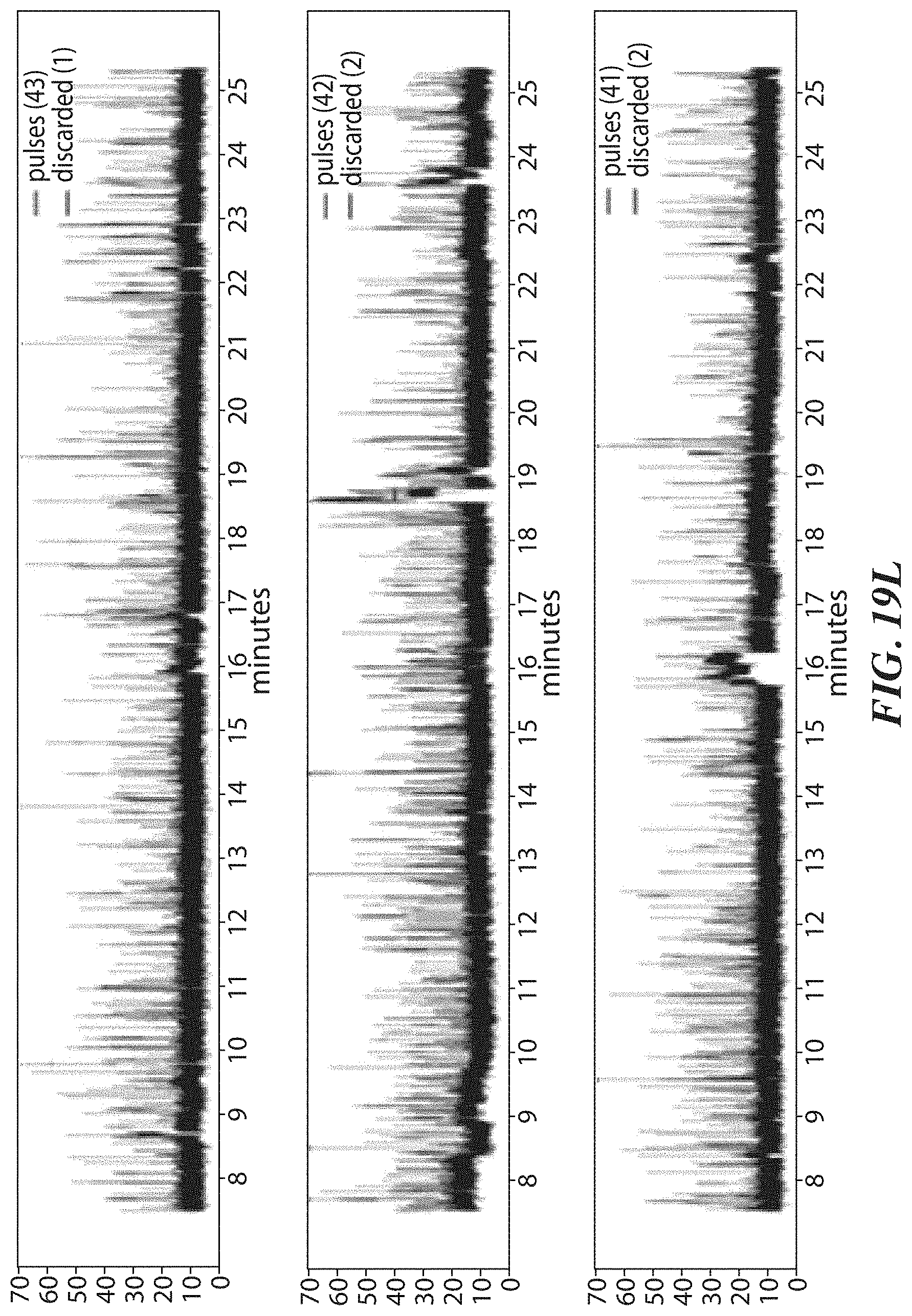
D00041

D00042

D00043

D00044
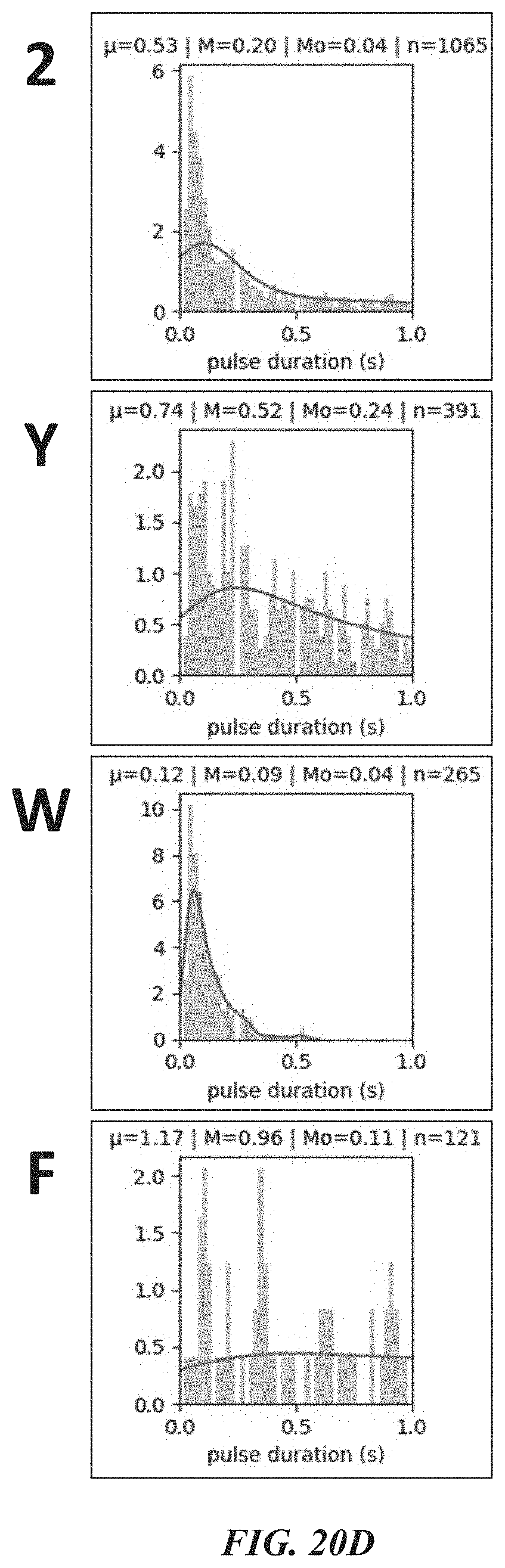
D00045

D00046

D00047

D00048

D00049

D00050
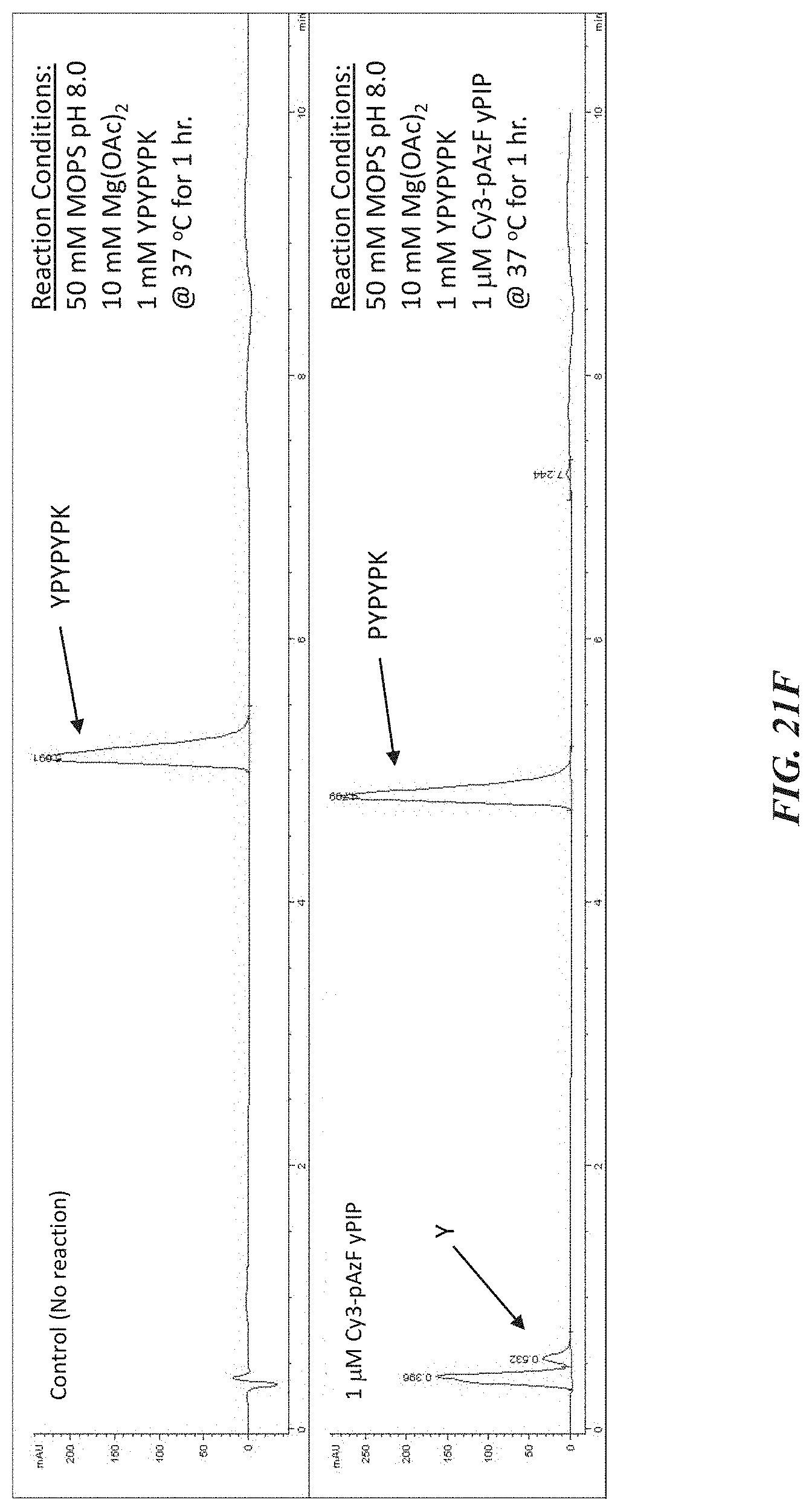
D00051

D00052

D00053
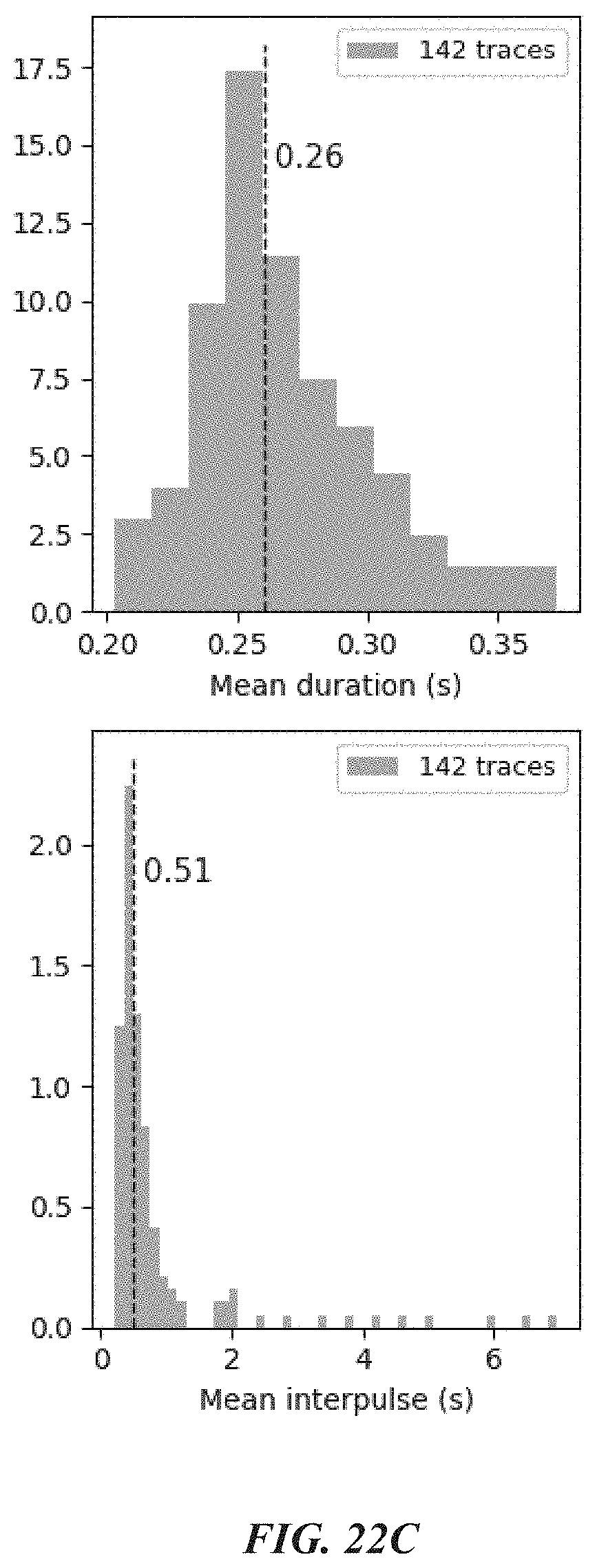
D00054

D00055

D00056

D00057
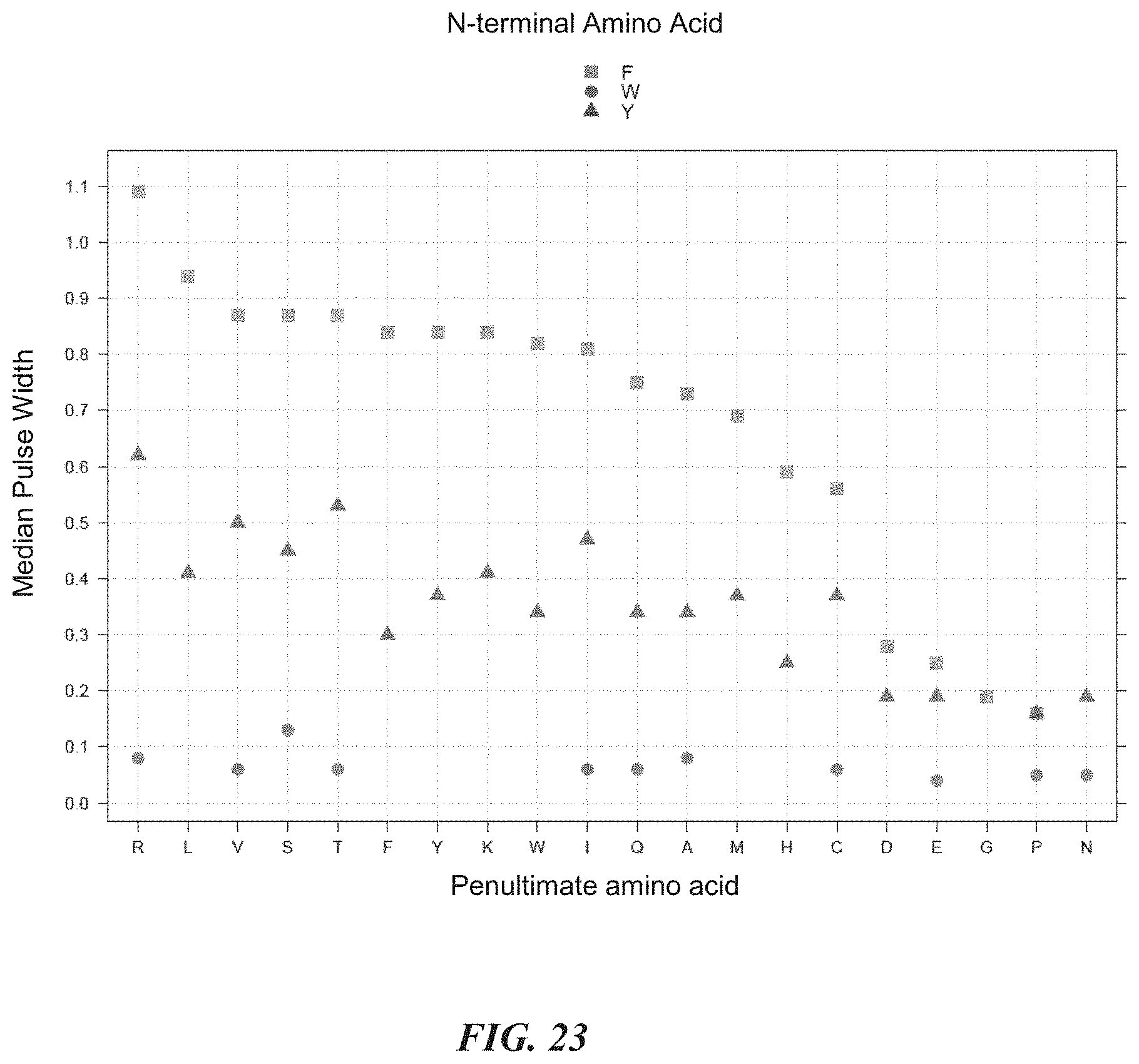
D00058

D00059
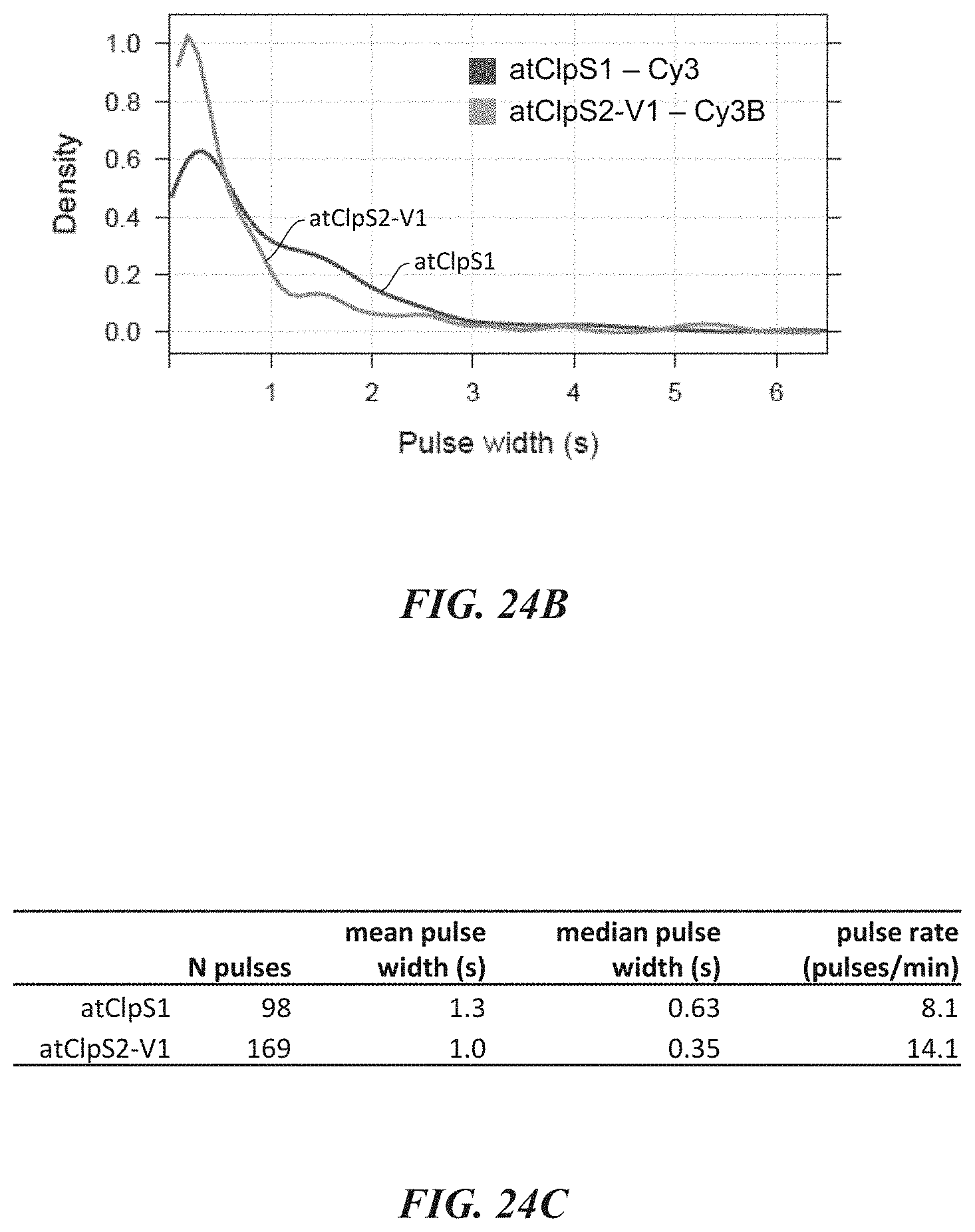
D00060
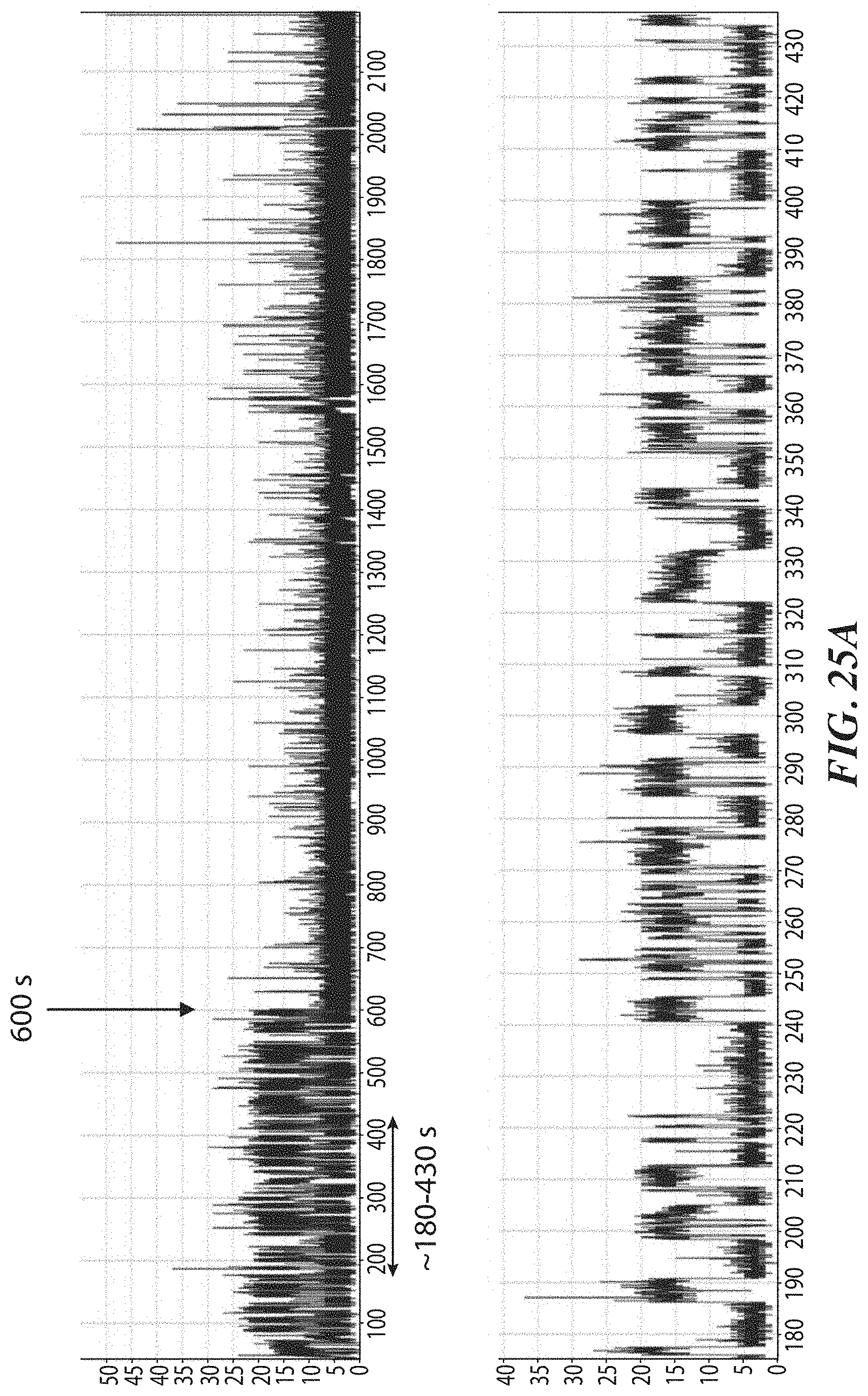
D00061
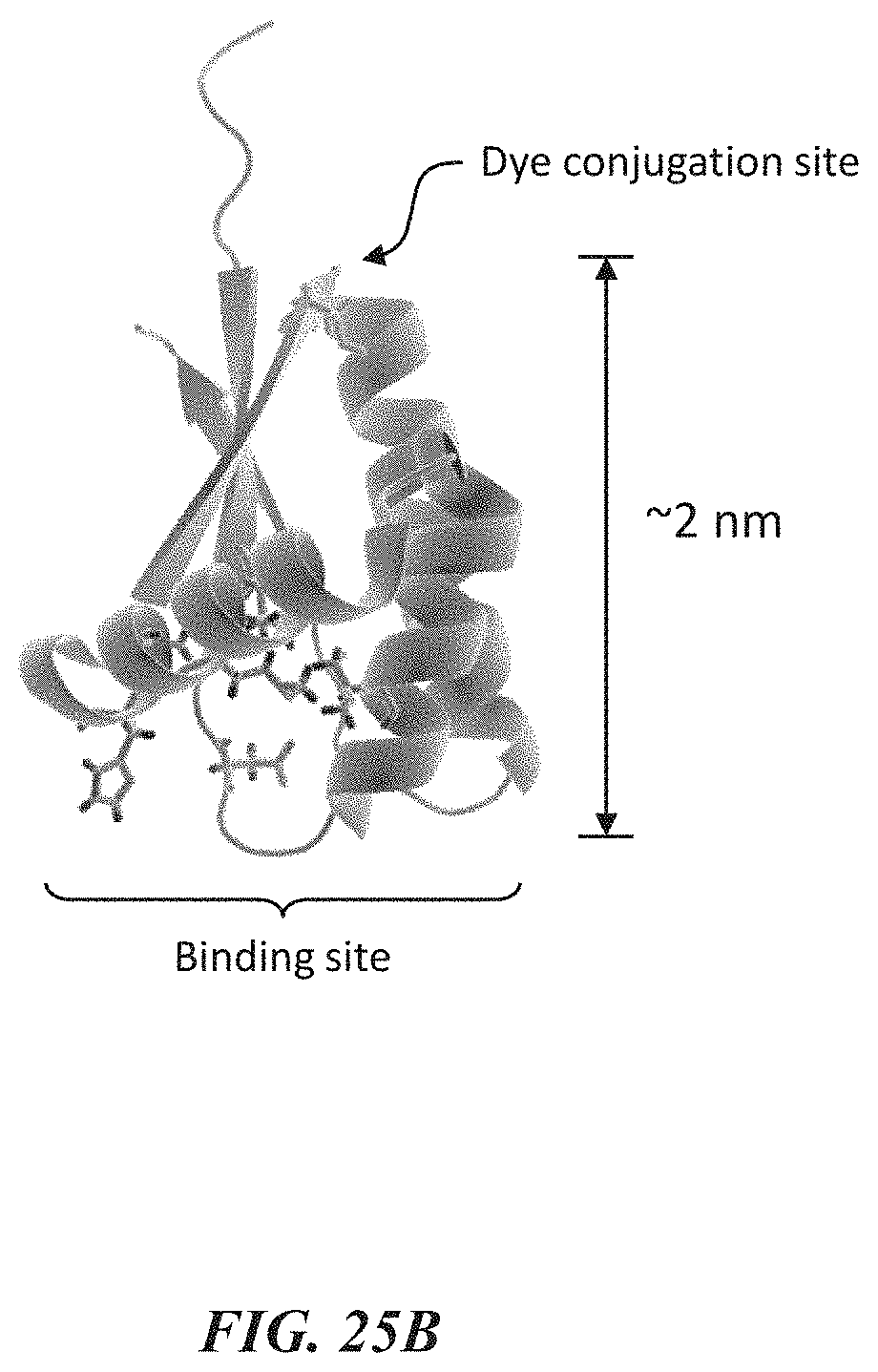
D00062

D00063
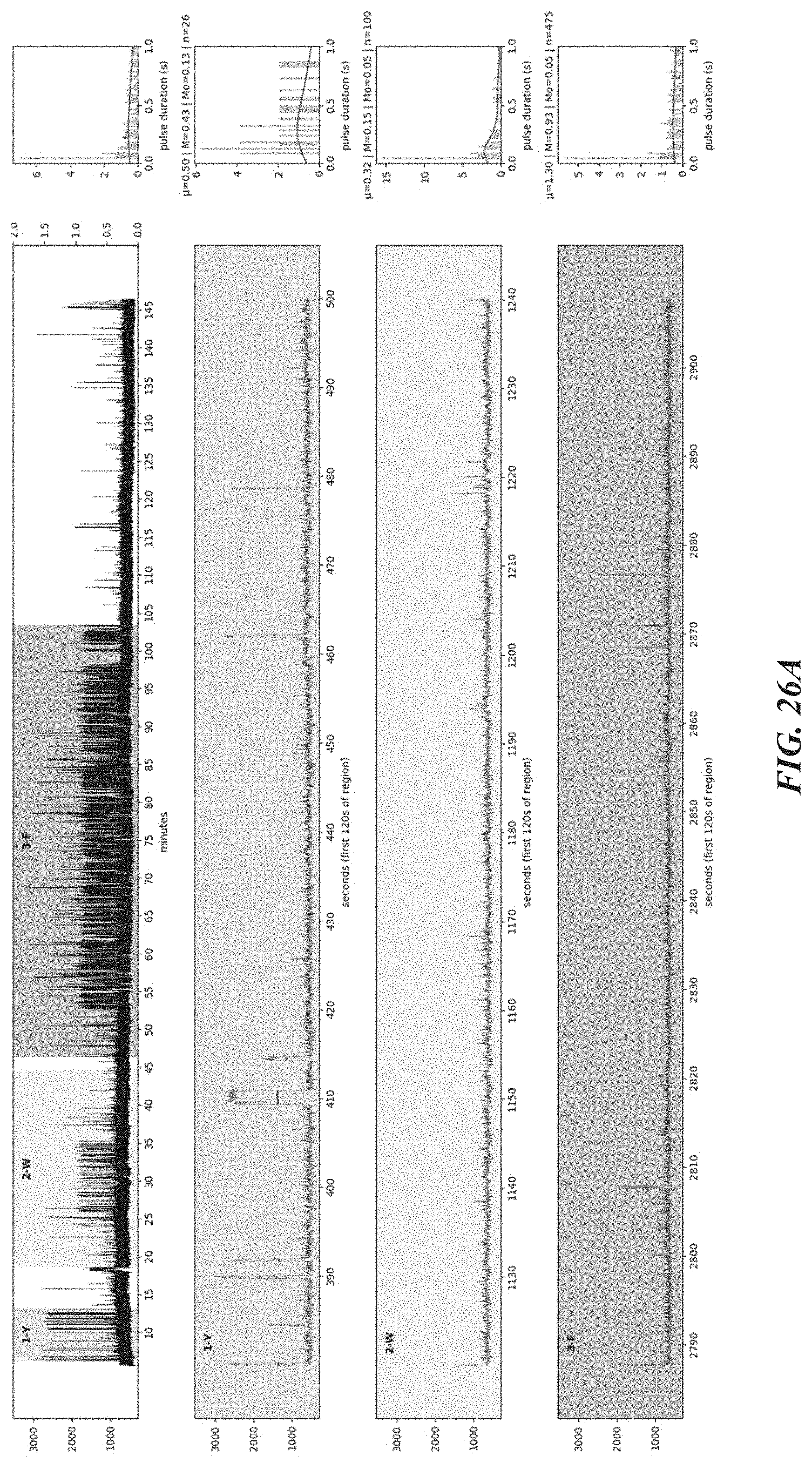
D00064

D00065

D00066

D00067

D00068

D00069
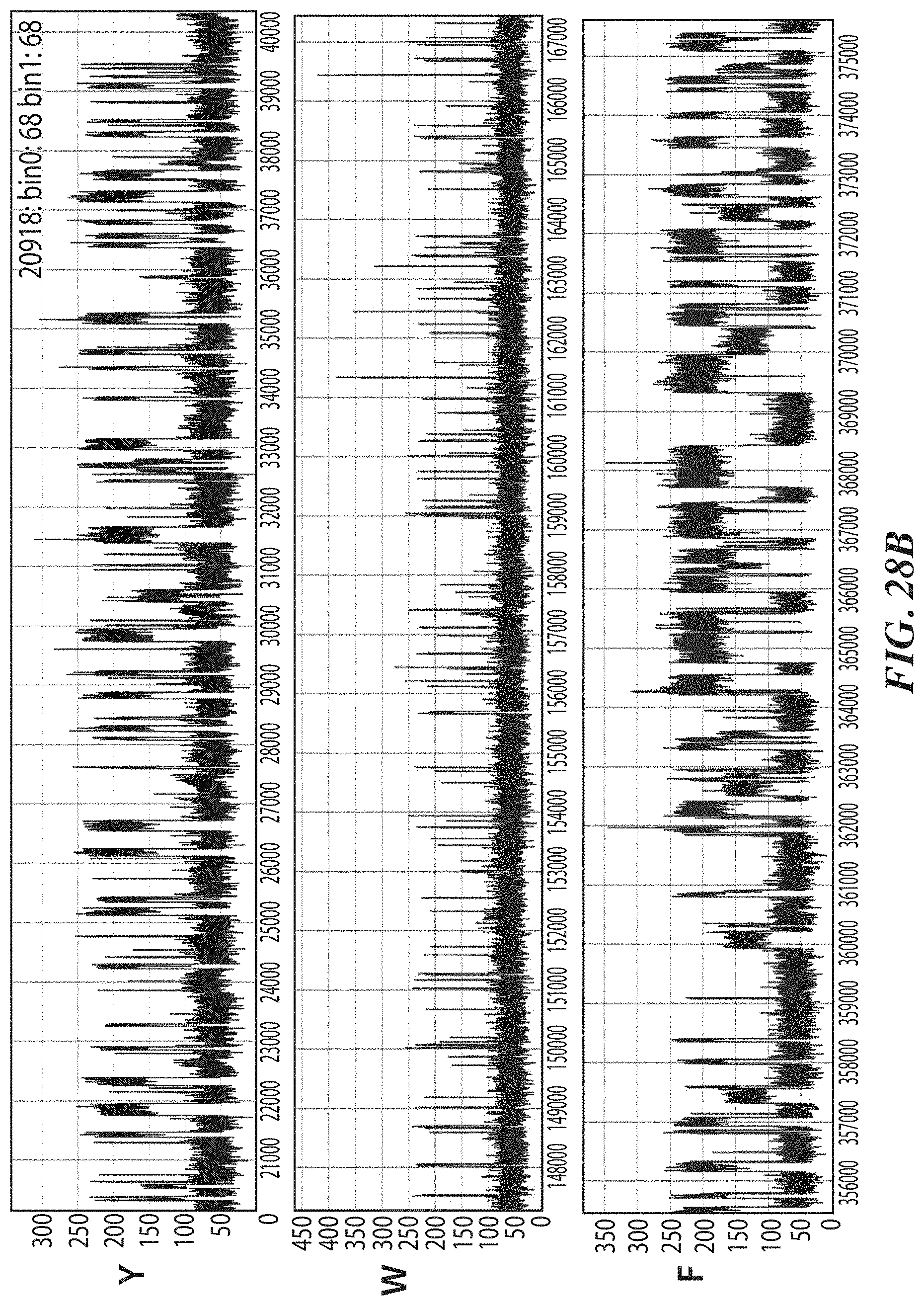
D00070

D00071

D00072

D00073
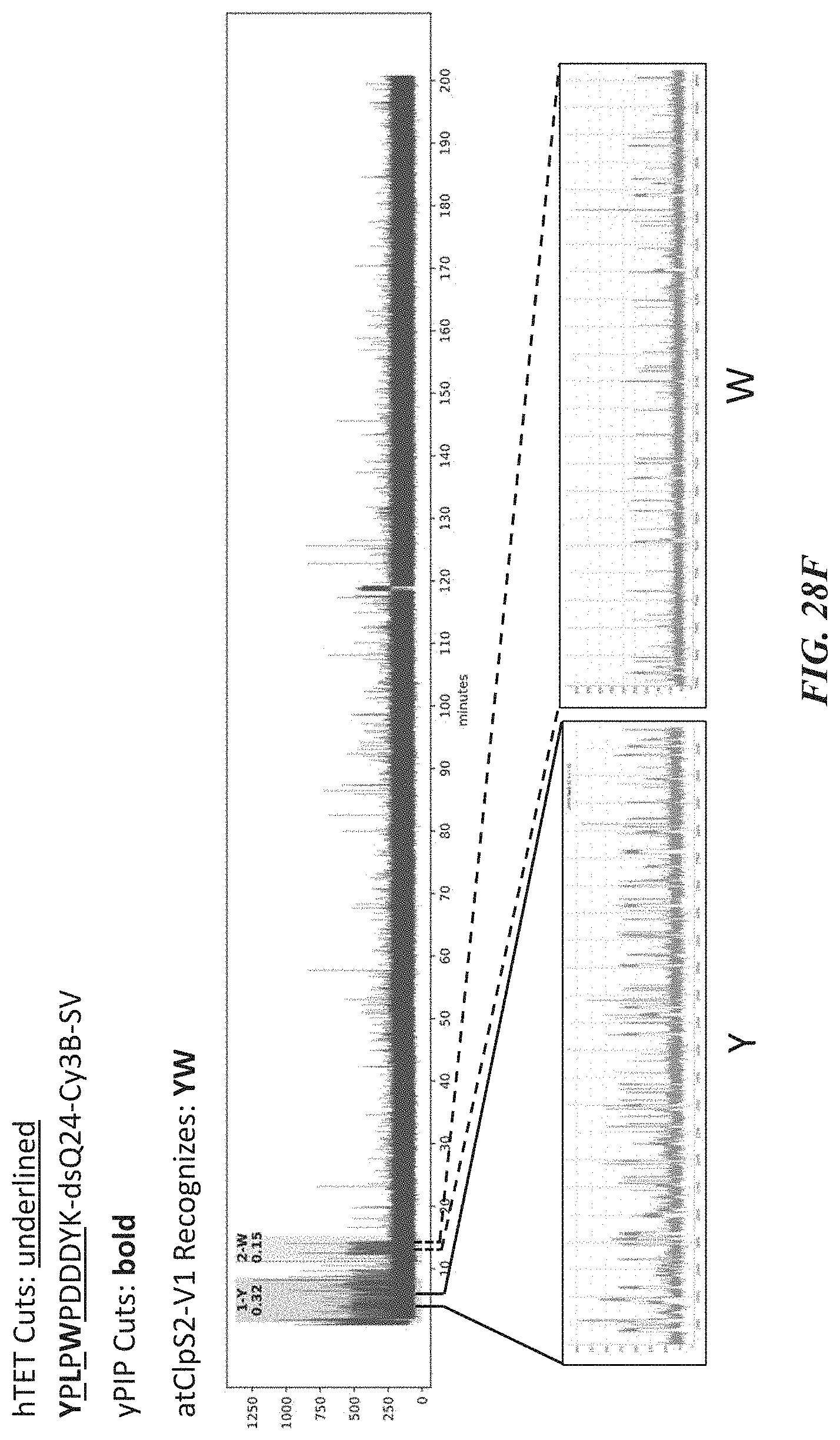
D00074

D00075
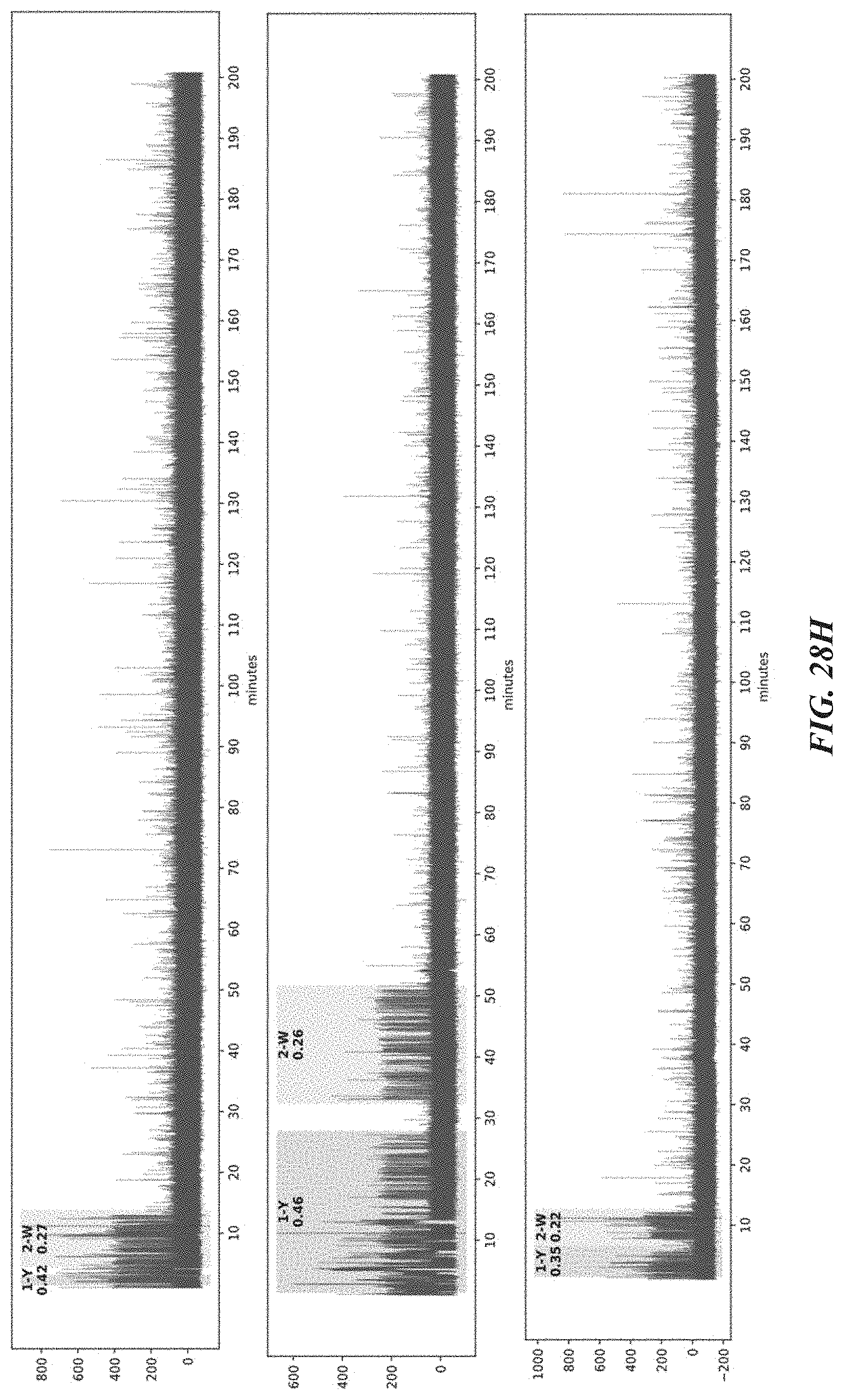
D00076

D00077

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.