Anti-MCAM Antibodies And Associated Methods Of Use
Flanagan; Kenneth ; et al.
U.S. patent application number 16/745178 was filed with the patent office on 2020-07-09 for anti-mcam antibodies and associated methods of use. This patent application is currently assigned to PROTHENA BIOSCIENCES LIMITED. The applicant listed for this patent is PROTHENA BIOSCIENCES LIMITED. Invention is credited to Jeanne Baker, Kenneth Flanagan, Theodore A. Yednock.
| Application Number | 20200216560 16/745178 |
| Document ID | / |
| Family ID | 50237783 |
| Filed Date | 2020-07-09 |










View All Diagrams
| United States Patent Application | 20200216560 |
| Kind Code | A1 |
| Flanagan; Kenneth ; et al. | July 9, 2020 |
Anti-MCAM Antibodies And Associated Methods Of Use
Abstract
Described herein are anti-MCAM antibodies and antigen binding fragments thereof that are capable of inhibiting the interaction between MCAM and its ligand, a protein comprising a laminin .alpha.-4 chain. These anti-MCAM antibodies and antigen binding fragments thereof may be useful for, for example, treating inflammatory conditions characterized by the infiltration of MCAM-expressing cells into a site of inflammation in the body.
| Inventors: | Flanagan; Kenneth; (Alameda, CA) ; Baker; Jeanne; (Redwood City, CA) ; Yednock; Theodore A.; (Forest Knolls, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | PROTHENA BIOSCIENCES
LIMITED Dublin 2 IE |
||||||||||
| Family ID: | 50237783 | ||||||||||
| Appl. No.: | 16/745178 | ||||||||||
| Filed: | January 16, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15726170 | Oct 5, 2017 | 10584177 | ||
| 16745178 | ||||
| 15222848 | Jul 28, 2016 | 10407507 | ||
| 15726170 | ||||
| 14021777 | Sep 9, 2013 | 9447190 | ||
| 15222848 | ||||
| 61698916 | Sep 10, 2012 | |||
| 61797179 | Nov 30, 2012 | |||
| 61797356 | Dec 5, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 19/02 20180101; C07K 2317/567 20130101; A61P 17/04 20180101; A61P 35/00 20180101; C07K 2317/92 20130101; C07K 16/2896 20130101; A61P 25/16 20180101; A61P 25/28 20180101; A61P 29/00 20180101; A61P 17/06 20180101; C07K 2317/565 20130101; C07K 2317/24 20130101; A61P 25/00 20180101; A61P 37/08 20180101; C07K 2317/56 20130101; C07K 2317/30 20130101; C07K 16/3092 20130101; A61P 37/02 20180101; A61P 37/06 20180101; A61K 2039/505 20130101; C07K 16/30 20130101; C07K 2317/76 20130101 |
| International Class: | C07K 16/30 20060101 C07K016/30; C07K 16/28 20060101 C07K016/28 |
Claims
1-61. (canceled)
62. An antibody specifically binding to human MCAM of SEQ ID NO: 11 comprising a heavy chain comprising a heavy chain variable region of SEQ ID NO: 119 and a heavy chain constant region of human IgG1 isotype, and a light chain comprising a light chain variable region of SEQ ID NO: 123 and a light chain kappa constant region.
63. The antibody of claim 62 expressed from a CHO cell.
64. A pharmaceutical composition comprising the antibody of claim 62 and a physiologically acceptable carrier.
65. A method of treating a human subject having multiple sclerosis comprising administering the antibody of claim 62 to the human subject thereby treating the multiple sclerosis.
Description
CROSS-REFERENCES TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 15/726,170 filed Oct. 5, 20176, which is a continuation of U.S. patent application Ser. No. 15/222,848 filed Jul. 28, 2016 now U.S. Pat. No. 10,407,507, which is a divisional of U.S. patent application Ser. No. 14/021,777 filed Sep. 9, 2013 now U.S. Pat. No. 9,447,190, which claims priority to U.S. Provisional Application No. 61/698,916 filed Sep. 10, 2012; U.S. Provisional Application No. 61/797,179 filed Nov. 30, 2012; and U.S. Provisional Application No. 61/797,356, filed Dec. 15, 2012, each of which applications is incorporated by reference in its entirety for all purposes.
REFERENCE TO A "SEQUENCE LISTING", A TABLE, OR A COMPUTER PROGRAM LISTING
[0002] The Sequence Listing written in file 542664SEQLST.txt was created on Jan. 15, 2020 for "ANTI-MCAM ANTIBODIES AND ASSOCIATED METHODS OF USE" is 144,373 bytes. The information contained in this file is hereby incorporated by reference.
FIELD OF THE INVENTION
[0003] The present invention is directed to antibodies that bind to melanoma cell adhesion molecule (MCAM) which are capable of blocking the interaction between MCAM and its ligand, the laminin alpha-4 chain. The present invention is also directed to methods of use of the novel anti-MCAM antibodies described herein.
BACKGROUND
[0004] A novel subset of CD4+ T cells, termed TH17 cells (T helper 17 cells), has been implicated in the pathogenesis of a number of autoimmune diseases, particularly those neuroinflammatory conditions involving CNS infiltration of T cells, such as multiple sclerosis and the animal model, experimental autoimmune encephalomyelitis (EAE). See, e.g., Cua et al., Nature 421: 744-748 (2003); see also Ivonov et al., Cell 126: 1121-1133 (2006). Much attention on the enhanced pathogenicity of TH17 cells has focused on their ability to secrete a number of select cytokines including IL-17 and IL-22. However, the role of these TH17 cytokines themselves has been called into question, as a conditional knockout of IL-17 is insufficient to affect EAE progression. See, e.g., Haak et al., J. Clin. Invest. 119: 61-69 (2009); see also Kreymborg et al., J. Immunol. 179: 8098-8104 (2007). Although IL-17 affects such vital aspects of EAE as endothelial cell permeability, TH17 cells appear to do more than just produce any one cytokine. The molecular determinants of the pathogenic function of TH17 cells remain elusive.
[0005] The pathogenicity of TH17 cells can be partially explained by their unique migration pattern as evidenced by their expression of chemokine receptors. See, e.g., Kim, Inflamm. Allergy Drug Targets 8: 221-228 (2009). It has been established that IL-17 producing cells are enriched within the CCR6+ population of CD4+ T cells, likely conferring a unique migration pattern throughout the vasculature. See, e.g., Acosta-Rodriguez et al., Nat. Immunol. 8:639-646 (2007). In fact, CCR6 expression on T cells is required for T cell migration into the CNS and the progression of EAE. Reboldi et al., Nat. Immunol. 10: 514-523 (2009). A hypothesis has arisen of two waves of T cells, the first a small population of CCR6 expressing TH17 cells that accumulates and recruits a broader second wave of T cells with a more diverse chemokine receptor repertoire. The anatomical site of this infiltration has been suggested to be the choroid plexus due to the constitutive expression of CCL20, a known ligand of CCR6. Ransohoff et al., Nat. Rev. Immunol. 3: 569-581 (2003). The implication has been made that the true pathogenic function of TH17 cells lies in their specific recruitment and infiltration of tissue.
[0006] Thus, there is still a need in the art to identify molecules that are involved in the infiltration of TH17 cells into CNS and contribute to their pathogenicity. These molecules can be targets to design therapeutic agents for neuroinflammatory conditions, such as multiple sclerosis (MS) and Parkinson's disease, as well as other TH17-mediated inflammatory conditions not associated with the central nervous system. There is also a need to identify novel antibodies that can bind to and are capable of reducing, interfering, or otherwise blocking the interaction between MCAM expressed on the surface of TH17 and its identified ligand.
SUMMARY OF THE INVENTION
[0007] TH17 cells play a significant role in the pathogenesis of various autoimmune diseases, particularly those displaying neuroinflammatory conditions involving T cells' infiltration into CNS. It has been newly discovered that (1) MCAM is selectively enriched on TH17 cells; and (2) MCAM interacts with a laminin .alpha.4 chain, such as, for example, the .alpha.4 chain of laminin 411, present in the endothelial basement membrane. An MCAM antagonist, e.g., a monoclonal antibody, capable of inhibiting MCAM's binding to a molecule containing a laminin .alpha.4 chain, such as, for example, a laminin 411 molecule, may inhibit the migration of TH17 cells into CNS, and thus can be used as a therapeutic agent to prevent or treat diseases displaying TH17-mediated neuroinflammatory conditions. MCAM antagonists, such as an MCAM monoclonal antibody or an antigen-binding fragment thereof, may also be useful to prevent or treat and TH17-mediated disease, including for example, autoimmune disease, for example, multiple sclerosis, inflammatory bowel disease, psoriasis, and rheumatoid arthritis.
[0008] The present invention is directed to novel antibodies that are capable of binding to MCAM protein on the surface of cells and, in turn, that are capable of interfering with the interaction of MCAM with its ligand, a protein comprising a laminin alpha-4 chain. Optionally, the antibody is a monoclonal antibody, antibody fragment, chimeric antibody, humanized antibody, single-chain antibody or antibody that competitively inhibits the binding of an anti-MCAM antibody to its respective antigenic epitope.
[0009] In other embodiments, the invention provides vectors comprising DNA encoding any of the herein described antibodies and host cells comprising such vectors, wherein such host cells may be CHO cells, E. coli cells, or yeast cells. A process for producing any of the herein described antibodies is further provided and comprises the steps of culturing host cells under conditions suitable for expression of the desired antibody, and recovering the desired antibody from the cell culture.
[0010] In one embodiment, the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, wherein the antibody or antigen binding fragment thereof comprises three light chain hypervariable regions (HVR-L1, HVR-L2, and HVR-L3) and three heavy chain hypervariable regions (HVR-H1, HVR-H2, and HVR-H3), and wherein:
[0011] (a) HVR-L1 comprises the amino acid sequence of SEQ ID NO:31, HVR-L2 comprises the amino acid sequence of SEQ ID NO:32, HVR-L3 comprises the amino acid sequence of SEQ ID NO:33, HVR-H1 comprises the amino acid sequence of SEQ ID NO:36, HVR-H2 comprises the amino acid sequence of SEQ ID NO:37, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:38;
[0012] (b) HVR-L1 comprises the amino acid sequence of SEQ ID NO:41, HVR-L2 comprises the amino acid sequence of SEQ ID NO:42, HVR-L3 comprises the amino acid sequence of SEQ ID NO:43, HVR-H1 comprises the amino acid sequence of SEQ ID NO:46, HVR-H2 comprises the amino acid sequence of SEQ ID NO:47, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:48;
[0013] (c) HVR-L1 comprises the amino acid sequence of SEQ ID NO:51, HVR-L2 comprises the amino acid sequence of SEQ ID NO:52, HVR-L3 comprises the amino acid sequence of SEQ ID NO:53, HVR-H1 comprises the amino acid sequence of SEQ ID NO:56, HVR-H2 comprises the amino acid sequence of SEQ ID NO:57, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:58;
[0014] (d) HVR-L1 comprises the amino acid sequence of SEQ ID NO:61, HVR-L2 comprises the amino acid sequence of SEQ ID NO:62, HVR-L3 comprises the amino acid sequence of SEQ ID NO:63, HVR-H1 comprises the amino acid sequence of SEQ ID NO:66, HVR-H2 comprises the amino acid sequence of SEQ ID NO:67, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:68;
[0015] (e) HVR-L1 comprises the amino acid sequence of SEQ ID NO:73, HVR-L2 comprises the amino acid sequence of SEQ ID NO:74, HVR-L3 comprises the amino acid sequence of SEQ ID NO:75, HVR-H1 comprises the amino acid sequence of SEQ ID NO:78, HVR-H2 comprises the amino acid sequence of SEQ ID NO:79, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:80; or
[0016] (f) HVR-L1 comprises the amino acid sequence of SEQ ID NO:85, HVR-L2 comprises the amino acid sequence of SEQ ID NO:86, HVR-L3 comprises the amino acid sequence of SEQ ID NO:87, HVR-H1 comprises the amino acid sequence of SEQ ID NO:90, HVR-H2 comprises the amino acid sequence of SEQ ID NO:91, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:92.
[0017] In certain embodiments, the anti-MCAM antibody may be a chimeric or humanized antibody. In another embodiment, the anti-MCAM antibody may be an IgG1 antibody which may optionally be produced in bacteria or CHO cells.
[0018] In yet another embodiment, the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, said antibody or antigen binding fragment thereof comprising a light chain variable region and a heavy chain variable region, wherein:
[0019] (a) the light chain variable region comprises the amino acid sequence of SEQ ID NO:30 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:35;
[0020] (b) the light chain variable region comprises the amino acid sequence of SEQ ID NO:40 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:45;
[0021] (c) the light chain variable region comprises the amino acid sequence of SEQ ID NO:50 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:55;
[0022] (d) the light chain variable region comprises the amino acid sequence of SEQ ID NO:60 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:65;
[0023] (e) the light chain variable region comprises the amino acid sequence of any one of SEQ ID NOS:70, 71, or 72 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:77; or
[0024] (f) the light chain variable region comprises the amino acid sequence of any one of SEQ ID NOS:83 or 84 and the heavy chain variable region comprises the amino acid sequence of SEQ ID NO:89.
[0025] In certain embodiments, the anti-MCAM antibody may be a chimeric or humanized antibody. In another embodiment, the anti-MCAM antibody may be an IgG1 antibody which may optionally be produced in bacteria or CHO cells.
[0026] In yet another embodiment, the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, that binds substantially the same epitope as, or competes for binding with, any of the anti-MCAM antibodies described herein.
[0027] In yet other embodiments, the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, that blocks the interaction between an MCAM protein comprising the amino acid sequence of SEQ ID NO:22 and a protein comprising a laminin .alpha.-4 chain. Another embodiment of the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, that blocks the interaction between an MCAM protein comprising the amino acid sequences of SEQ ID NOS:22 and 23 and a protein comprising a laminin .alpha.-4 chain. A further embodiment of the present invention is directed to an isolated anti-MCAM antibody or antigen binding fragment thereof which does not block the interaction between an MCAM protein consisting of the amino acid sequence of SEQ ID NO:22 and a protein comprising a laminin .alpha.-4 chain. Yet another embodiment of the present invention is directed to an isolated anti-MCAM antibody, or antigen binding fragment thereof, that blocks the interaction between an MCAM protein comprising the amino acid sequences of SEQ ID NOS:22, 23, and 24 and a protein comprising a laminin .alpha.-4 chain. Further embodiments of the present invention are directed to isolated anti-MCAM antibodies, or antigen binding fragments thereof, that bind to antigenic epitopes defined by domains 1 and 2, or domain 3 of the human MCAM protein. In a preferred embodiment, the anti-MCAM antibody or fragment thereof does not bind to a protein consisting of amino acids 19-129 of the human MCAM protein.
[0028] Yet other embodiments of the present invention are directed to pharmaceutical compositions comprising any of the herein described antibodies, or antigen binding fragment thereof, and articles of manufacture comprising the same.
[0029] Other embodiments of the present invention are directed to the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of an inflammatory disorder characterized by infiltration of MCAM-expressing cells into a site of inflammation in the body. In certain embodiments, the inflammatory disorder may be a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS.
[0030] The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of multiple sclerosis; Parkinson's disease. The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of allergic contact dermatitis. The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of, psoriasis. The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of psoriatic arthritis. The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of cancer, for example, a solid tumor, such as a melanoma. The invention also provides for the use of an anti-MCAM antibody, or antigen binding fragment thereof, in the manufacture of a medicament for the treatment of sarcoidosis.
[0031] Another embodiment of the present invention is directed to a method for the treatment of an inflammatory disorder characterized by infiltration of MCAM-expressing cells to a site of inflammation, the method comprising administering to a mammalian subject in need thereof an effective amount of an anti-MCAM antibody or antigen binding fragment thereof that inhibits the binding of MCAM to a protein comprising a laminin .alpha.-4 chain. In certain embodiments, the mammalian subject may be a human and the MCAM-expressing cells may be TH17 cells.
[0032] In another aspect, the present invention provides an isolated h1749 anti-MCAM antibody, or antigen binding fragment thereof. In one embodiment, the antibody or antigen binding fragment thereof comprises three light chain hypervariable regions (HVR-L1, HVR-L2, and HVR-L3) and three heavy chain hypervariable regions (HVR-H1, HVR-H2, and HVR-H3), wherein HVR-L1 comprises the amino acid sequence of SEQ ID NO: 61, HVR-L2 comprises the amino acid sequence of SEQ ID NO:62, HVR-L3 comprises the amino acid sequence of SEQ ID NO:63, HVR-H1 comprises the amino acid sequence of SEQ ID NO:66, HVR-H2 comprises the amino acid sequence of SEQ ID NO:67, HVR-H3 comprises the amino acid sequence of SEQ ID NO:68. In another embodiment, the isolated anti-MCAM antibody, or antibody binding fragment thereof, further comprises a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 128.
[0033] In one other aspect, the present invention provides an isolated h2107 anti-MCAM antibody, or antigen binding fragment thereof. In one embodiment, the isolated anti-MCAM antibody, or antigen binding fragment thereof, comprises three light chain hypervariable regions (HVR-L1, HVR-L2, and HVR-L3) and three heavy chain hypervariable regions (HVR-H1, HVR-H2, and HVR-H3), wherein HVR-L1 comprises the amino acid sequence of SEQ ID NO:85, HVR-L2 comprises the amino acid sequence of SEQ ID NO:86, HVR-L3 comprises the amino acid sequence of SEQ ID NO:87, HVR-H1 comprises the amino acid sequence of SEQ ID NO:90, HVR-H2 comprises the amino acid sequence of SEQ ID NO:91, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:92. In another embodiment, the isolated anti-MCAM antibody, or antibody binding fragment thereof, further comprises a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 134 and/or a heavy chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 137. In one other embodiment, the isolated anti-MCAM antibody, or antibody binding fragment thereof, further comprises a light chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 146; and/or a light chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 144.
[0034] In one additional aspect, the present invention provides an isolated h2120 anti-MCAM antibody, or antigen binding fragment thereof. In one embodiment, the antibody or antigen binding fragment thereof comprises three light chain hypervariable regions (HVR-L1, HVR-L2, and HVR-L3) and three heavy chain hypervariable regions (HVR-H1, HVR-H2, and HVR-H3), wherein HVR-L1 comprises the amino acid sequence of SEQ ID NO:73, HVR-L2 comprises the amino acid sequence of SEQ ID NO:74, HVR-L3 comprises the amino acid sequence of SEQ ID NO:75, HVR-H1 comprises the amino acid sequence of SEQ ID NO: 141, HVR-H2 comprises the amino acid sequence of SEQ ID NO:79, and HVR-H3 comprises the amino acid sequence of SEQ ID NO:80. In another embodiment, the isolated anti-MCAM antibody, or antibody binding fragment thereof, comprises a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 134 and/or a heavy chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 135. In one other embodiment, the isolated anti-MCAM antibody, or antibody binding fragment thereof, further comprises a light chain framework region 1 (FR1) comprising the amino acid sequence of SEQ ID NO: 147; a light chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 148; a light chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 149; or any combination thereof.
[0035] Additional embodiments of the present invention will be evident to those of ordinary skill in the art based upon the teachings of the present specification.
BRIEF DESCRIPTION OF THE DRAWINGS
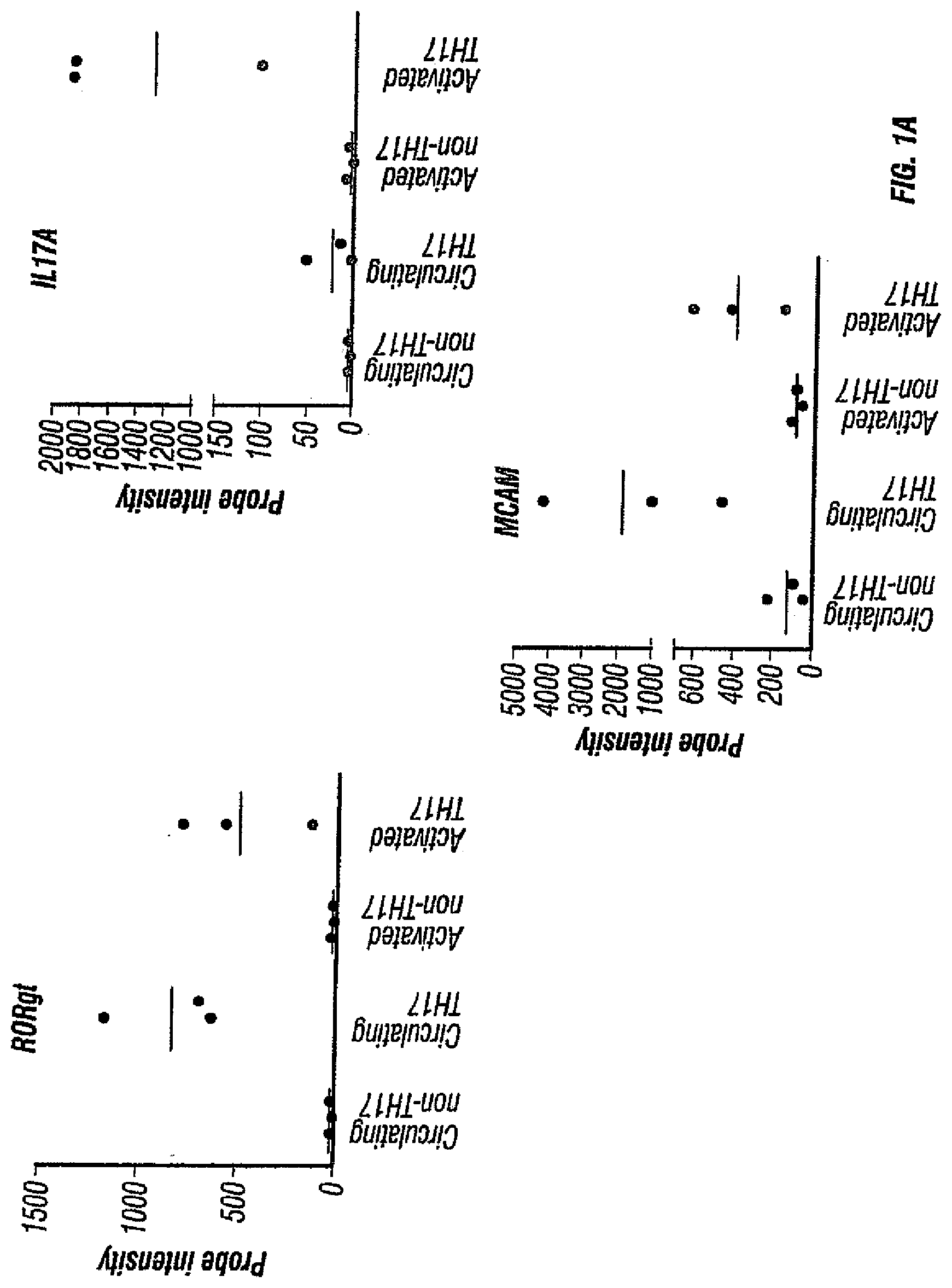
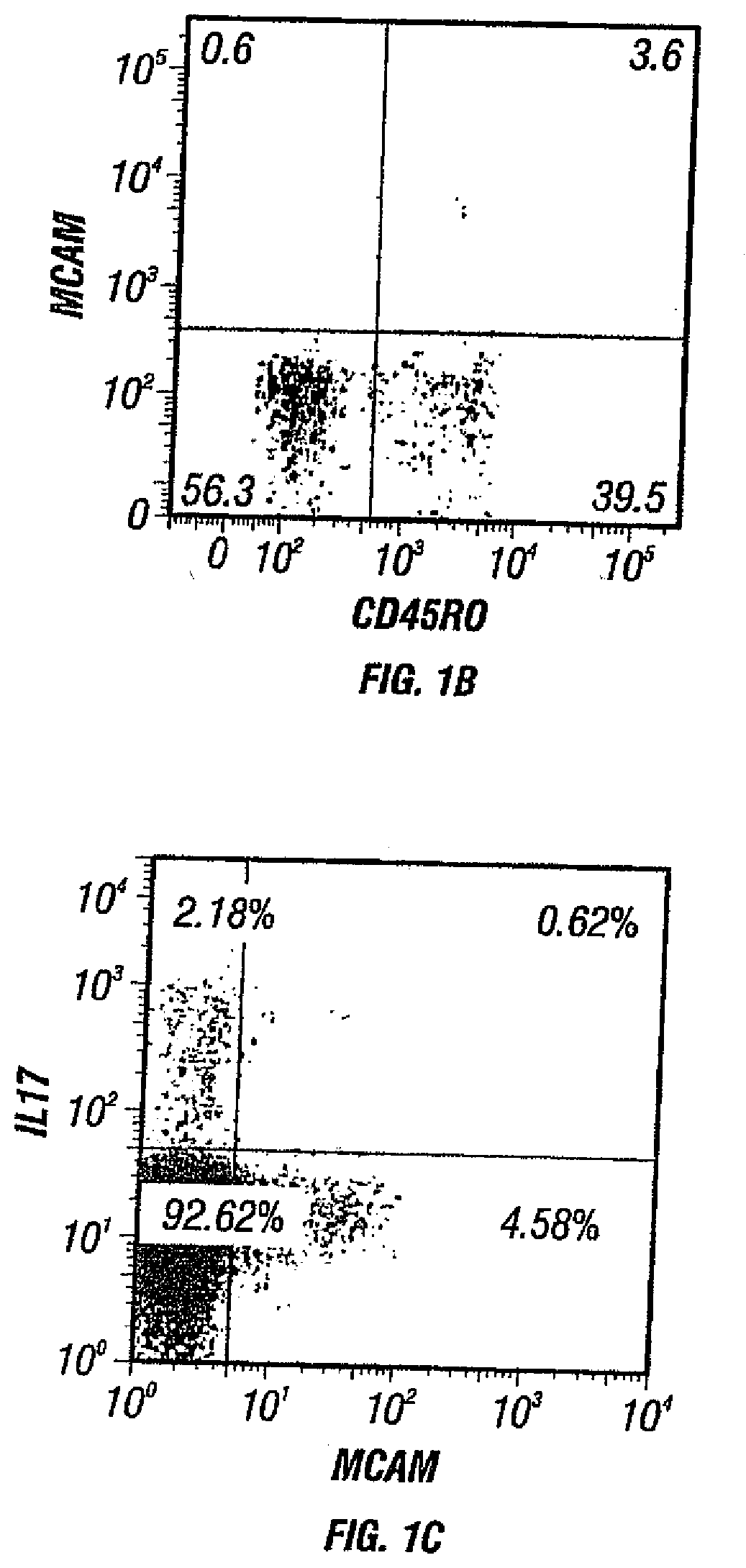
[0036] FIGS. 1A-C. FIGS. 1A and 1B depict the presence of MCAM in IL-17-producing human CD4+ cells. FIG. 1A depicts the microarray analysis showing that MCAM is an up-regulated gene in both circulating and activated TH17 cells. FIG. 1B depicts the cell sorting results showing that MCAM exist almost exclusively in a small population of memory T cells (CD45RO+ T cells). FIG. 1C depicts the cell sorting results showing that MCAM is enriched in IL-17-producing human CD4+ T cells.
[0037] FIGS. 2A, B depict the surface markers of MCAM expressing T cells. FIG. 2A depicts MCAM expressing T cells as effector memory T cells (CCR6+ while CCR7-). FIG. 2B depicts the integrin expression pattern of MCAM expressing T cells. The majority of MCAM expressing T cells are integrin .alpha.4 positive, but are largely integrin .beta.7 negative and .beta.1 positive.
[0038] FIGS. 3A-F depict the effects of various cytokines on CD4+/CD45RO+ memory T cells. FIG. 3A depicts the effects of various cytokines on IL-17 production in MCAM positive T cells. FIG. 3B depicts the percentage of cells expressing MCAM following stimulation by various cytokines. FIGS. 3C, 3D, and 3E depict the levels of IL-17 (FIG. 3C), IL-22 (FIG. 3D), and CCL20 (FIG. 3E) in both MCAM positive and MCAM negative cells after stimulations with various cytokines. FIG. 3F depicts the intracellular levels of FOXP3 in both MCAM positive and MCAM negative cells after stimulations with various cytokines.
[0039] FIGS. 4A-H depict the identification of laminin 411 as the MCAM ligand. FIG. 4A depicts co-localization of the MCAM ligand and laminin on the choroid plexus of healthy mice. FIG. 4B depicts absence of MCAM staining on the choroid plexus of healthy mice (4',6-diamidino-2-phenylindole (DAPI) was used as a counterstain). FIG. 4C depicts the presence of MCAM on vascular endothelial cells within healthy mouse brain (DAPI was used as a counterstain). FIG. 4D depicts the expression pattern of the MCAM ligand by staining healthy mouse spinal cord sections with MCAM-Fc protein. FIG. 4E depicts co-localization of the MCAM ligand and laminin on healthy mouse spinal cord. FIG. 4F depicts the extracellular matrix (ECM) localization of the MCAM ligand. CD31 staining was used to show that MCAM staining is exterior to the endothelial cell layer within the vasculature. FIG. 4G depicts the localization of the MCAM ligand within EAE lesions. MCAM-Fc is shown to colocalize with laminin within the endothelial cell basement membrane, but not within the parenchymal basement membrane. FIG. 4H depicts co-localization of the MCAM ligand and laminin 411 (or laminin alpha-4 chain).
[0040] FIGS. 5A-C. FIG. 5A depicts specific binding of MCAM antibodies to human and mouse MCAM. FIG. 5B depicts blockage of MCAM-Fc's binding to tissues by MCAM antibodies. FIG. 5C depicts inhibition of the interaction between human MCAM and its ligand laminin 411 by a monoclonal antibody.
[0041] FIGS. 6A, B. FIG. 6A depicts the light chain variable region of clone 17 monoclonal antibody. FIG. 6A discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO: 1) and the amino acid sequence of the light chain variable region (SEQ ID NO:2), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:3), CDRL2 (SEQ ID NO:4), and CDRL3 (SEQ ID NO:5). FIG. 6B depicts the heavy chain variable region clone 17 monoclonal antibody. FIG. 6B discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:6) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:7), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:8), CDRH2 (SEQ ID NO:9), and CDRH3 (SEQ ID NO: 10).
[0042] FIGS. 7A, B. FIG. 7A depicts absence of MCAM on T cells from naive mouse. FIG. 7B depicts MCAM expression levels among splenocytes in the presence of various cytokines. Splenocytes were obtained from PLP immunized SJL mice and in vitro restimulated with PLP.
[0043] FIGS. 8A, B depicts the effects of MCAM blockade on disease progression in a therapeutic model of EAE. After EAE symptoms appeared, PLP-immunized mice were treated intraperitoneally with (1) anti-MCAM antibody (clone 15) at 10 mg/kg body weight, (2) the isotype control (Bioxcell) at 10 mg/kg body weight, and (3) PBS every day thereafter. The disease progression (FIG. 8A) and body weights (FIG. 8B) were monitored every 2-3 days. Data represent the mean of 15 mice.+-.sem (standard error of the mean).
[0044] FIGS. 9A, B. FIG. 9A depicts the light chain variable region of clone 15 monoclonal antibody. FIG. 9A discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO: 12) and the amino acid sequence of the light chain variable region (SEQ ID NO: 13), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:14), CDRL2 (SEQ ID NO:15), and CDRL3 (SEQ ID NO:16). FIG. 9B depicts the heavy chain variable region clone 15 monoclonal antibody. FIG. 9B discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO: 17) and the amino acid sequence of the heavy chain variable region (SEQ ID NO: 18), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:19), CDRH2 (SEQ ID NO:20), and CDRH3 (SEQ ID NO:21).
[0045] FIGS. 10A, B depict the results of a domain binding test for MCAM antibodies.
[0046] FIGS. 11A, B depict the amino acid sequence (A) (SEQ ID NO: 11--Accession No. CAA48332) and structure (B) for human MCAM. In FIG. 11A, the amino acid residue positions corresponding to the five immunoglobulin domains of human MCAM are as follows--1: amino acid residues 19-129; 2: amino acid residues 139-242; 3: amino acid residues 244-321; 4: amino acid residues 335-424; and 5: amino acid residues 430-510) (SEQ ID NOS:22-26), which are also depicted schematically in FIG. 11B.
[0047] FIGS. 12A, B show the amino acid sequences for two .alpha.4-chain isoforms of human laminin 411. FIG. 12A shows the amino acid sequence corresponding to GenBank Accession No. NP001098676 (SEQ ID NO:27) and FIG. 12B shows the amino acid sequence corresponding to GenBank Accession No. NP001098677 (SEQ ID NO:28).
[0048] FIG. 13 depicts the light chain variable region of clone 1174.1.3 monoclonal antibody. FIG. 13 discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO:29) and the amino acid sequence of the light chain variable region (SEQ ID NO:30), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:31), CDRL2 (SEQ ID NO:32), and CDRL3 (SEQ ID NO:33).
[0049] FIG. 14 depicts the heavy chain variable region clone 1174.1.3 monoclonal antibody. FIG. 14 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:34) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:35), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:36), CDRH2 (SEQ ID NO:37), and CDRH3 (SEQ ID NO:38).
[0050] FIG. 15 depicts the light chain variable region of clone 1414.1.2 monoclonal antibody. FIG. 15 discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO:39) and the amino acid sequence of the light chain variable region (SEQ ID NO:40), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:41), CDRL2 (SEQ ID NO:42), and CDRL3 (SEQ ID NO:43).
[0051] FIG. 16 depicts the heavy chain variable region clone 1414.1.2 monoclonal antibody. FIG. 16 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:44) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:45), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:46), CDRH2 (SEQ ID NO:47), and CDRH3 (SEQ ID NO:48).
[0052] FIG. 17 depicts the light chain variable region of clone 1415.1.1 monoclonal antibody. FIG. 17 discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO:49) and the amino acid sequence of the light chain variable region (SEQ ID NO:50), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:51), CDRL2 (SEQ ID NO:52), and CDRL3 (SEQ ID NO:53).
[0053] FIG. 18 depicts the heavy chain variable region clone 1415.1.1 monoclonal antibody. FIG. 18 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:54) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:55), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:56), CDRH2 (SEQ ID NO:57), and CDRH3 (SEQ ID NO:58).
[0054] FIG. 19 depicts the light chain variable region of clone 1749.1.3 monoclonal antibody. FIG. 19 discloses the nucleic acid sequence encoding the light chain variable region (SEQ ID NO:59) and the amino acid sequence of the light chain variable region (SEQ ID NO:60), in order of appearance. The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:61), CDRL2 (SEQ ID NO:62), and CDRL3 (SEQ ID NO:63).
[0055] FIG. 20 depicts the heavy chain variable region clone 1749.1.3 monoclonal antibody. FIG. 20 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:64) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:65), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:66), CDRH2 (SEQ ID NO:67), and CDRH3 (SEQ ID NO:68).
[0056] FIGS. 21A, B depict different versions of the light chain variable region of clone 2120.4.19 monoclonal antibody. FIG. 21A-B discloses one version of the nucleic acid sequence encoding a light chain variable region (SEQ ID NO:69), the amino acid sequence of version 1 of the light chain variable region (SEQ ID NO:70), the amino acid sequence of version 2 of the light chain variable region (SEQ ID NO:71), and the amino acid sequence of version 3 of the light chain variable region (SEQ ID NO:72). The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:73), CDRL2 (SEQ ID NO:74), and CDRL3 (SEQ ID NO:75).
[0057] FIG. 22 depicts the heavy chain variable region clone 2120.4.19 monoclonal antibody. FIG. 22 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:76) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:77), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:78), CDRH2 (SEQ ID NO:79), and CDRH3 (SEQ ID NO:80).
[0058] FIGS. 23A, B depict different versions of the light chain variable region of clone 2107.4.10 monoclonal antibody. FIGS. 23A-B discloses the nucleic acid sequence encoding version 1 of a light chain variable region (SEQ ID NO:81), the nucleic acid sequence encoding version 2 of a light chain variable region (SEQ ID NO:83), the amino acid sequence of version 1 of the light chain variable region (SEQ ID NO:82), and the amino acid sequence of version 2 of the light chain variable region (SEQ ID NO:84). The three hypervariable regions are also indicated as CDRL1 (SEQ ID NO:85), CDRL2 (SEQ ID NO:86), and CDRL3 (SEQ ID NO:87).
[0059] FIG. 24 depicts the heavy chain variable region clone 2107.4.10 monoclonal antibody. FIG. 24 discloses the nucleic acid sequence encoding the heavy chain variable region (SEQ ID NO:88) and the amino acid sequence of the heavy chain variable region (SEQ ID NO:89), in order of appearance. The three hypervariable regions are also indicated as CDRH1 (SEQ ID NO:90), CDRH2 (SEQ ID NO:91), and CDRH3 (SEQ ID NO:92).
[0060] FIG. 25A shows the alignment of sequences of the variable heavy chains for the following: murine 1749.1.3 anti-MCAM antibody (1749.1.3_VH_pro; SEQ ID NO:93); 1749 VH1 humanized anti-MCAM antibody (h1749VH1; SEQ ID NO:94); 1749 VH2 humanized anti-MCAM antibody (h1749VH2; SEQ ID NO:95); and heavy chain human variable IGHV3-7*02 sequence used as the framework donor (U96282_VH; SEQ ID NO:96). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 1749.1.3 antibody to the variable heavy chain variable IGHV3-7*02 framework are boxed. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk.
[0061] FIG. 25B shows the alignment of sequences of the variable light chains for the following: murine 1749.1.3 anti-MCAM antibody (1749.1.3_VL_pro; SEQ ID NO:97); 1749 VL1 humanized anti-MCAM antibody (h1749VL1 SEQ ID NO:98); 1749 VL2 humanized anti-MCAM antibody (h1749VL2 SEQ ID NO:99); and light chain human variable X02990 IGKV4-1*01 sequence used as the framework donor (X02990_VL SEQ ID NO:100). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 1749.1.3 antibody to the variable light chain variable X02990 IGKV4-1*01 framework are boxed. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk.
[0062] FIG. 26A shows the alignment of sequences of the variable heavy chains for the following: murine 2107.4.10.18 anti-MCAM antibody (2107.4.10.18_VH_topo_pro; SEQ ID NO:101); 2107 VH1 humanized anti-MCAM antibody (h2107VH1; SEQ ID NO:102); 2107 VH2 humanized anti-MCAM antibody (h2107VH2; SEQ ID NO:103); 2107 VH3 humanized anti-MCAM antibody (h2107VH3; SEQ ID NO: 104); 2107 VH4 humanized anti-MCAM antibody (h2107VH4; SEQ ID NO: 105); 2107 VH5 humanized anti-MCAM antibody (h2107VH5; SEQ ID NO:106); 2107 VH6 humanized anti-MCAM antibody (h2107VH6; SEQ ID NO: 107); and heavy chain human variable AF062133 IGHV2-26*01 sequence used as the framework donor (AF062133_VH; SEQ ID NO:108). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 2107.4.10.18 antibody to the variable heavy chain variable AF062133 IGHV2-26*01 framework are boxed in both FIG. 26A. The S30T, I37V, L48I and K71R mutations combined with (i) mutations of the boxed N/D residues between CDR-H2 and CDR-H3 (D78N) restores murine N-glycosylation; or a mutation in an N-G sequence in CDR-H1, e.g., N32S (VH4); N32Q (VH5); or G33A (VH6)), provides an N deamidation mutant. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk.
[0063] FIG. 26B shows the alignment of sequences of the variable light chains for the following: murine 2107_L7-6 anti-MCAM antibody (2107_L7-6_pro; SEQ ID NO:109); 2107 VL1 humanized anti-MCAM antibody (h2107VL1; SEQ ID NO:110); 2107 VL2 humanized anti-MCAM antibody (h2107VL2; SEQ ID NO: 111); 2107 VL3 humanized anti-MCAM antibody (h2107VL3 SEQ ID NO: 112); and light chain human variable U86803 IGKV1-27*01 sequence used as the framework donor (U86803_VL SEQ ID NO: 113). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 2107_L7-6 antibody to the variable light chain variable U86803 IGKV1-27*01 framework are boxed. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk.
[0064] FIG. 27A shows the alignment of sequences of the variable heavy chains for the following: murine 2120.4.19.6 anti-MCAM antibody (2120.4.19.6_VH_topo_pro; SEQ ID NO:114); 2120 VH1 humanized anti-MCAM antibody (h2120VH1; SEQ ID NO:115); 2120 VH2 humanized anti-MCAM antibody (h2120VH2; SEQ ID NO: 116); 2120 VH3 humanized anti-MCAM antibody (h2120VH3; SEQ ID NO: 117); 2120 VH4 humanized anti-MCAM antibody (h2120VH4; SEQ ID NO: 118); 2120 VH5 humanized anti-MCAM antibody (h2120VH5; SEQ ID NO: 119); and heavy chain human variable AF062133 IGHV2-26*01 sequence used as the framework donor (AF062133_VH; SEQ ID NO: 108). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 2120.4.19.6 antibody to the variable heavy chain variable AF062133 IGHV2-26*01 framework are boxed. The S30T, I37V, L48I and K71R mutations combined with (i) mutations of the boxed N/D residues in CDR-H1, e.g., N32S (VH3); N32Q (VH4); or G33A (VH5)), provides an N deamidation mutant. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk. Residues where mutations were focused due to the presence of N-deamination sites or N-glycosylation sites are shown in the bracketed box.
[0065] FIG. 27B shows the alignment of sequences of the variable light chains for the following: murine 2120.4.19.6 anti-MCAM antibody (2120.4.19.6_VL_topo_ro; SEQ ID NO:120); 2120 VL1 humanized anti-MCAM antibody (h2120VL1 SEQ ID NO:121); 2120 VL2 humanized anti-MCAM antibody (h2120VL2 SEQ ID NO: 122); 2120 VL3 humanized anti-MCAM antibody (h2120VL3 SEQ ID NO: 123); and light chain human variable X84343 IGKV2-26*01 sequence used as the framework donor (X84343_VL SEQ ID NO:124). Kabat numbering is used and hypervariable regions (HVRs) grafted from the murine 2120.4.19.6 antibody to the variable light chain variable X84343 IGKV2-26*01 framework are boxed. The bolded amino acid residues in the humanized antibody sequences differ from the corresponding residues in the murine antibody sequence. The position of canonical and interface amino acid residues that may affect CDR contact or CDR structure are indicated by an asterisk.
[0066] FIGS. 28A-C compare the blocking of various 1749, 2120, and 2107 antibodies of MCAM binding to laminin 411.
[0067] FIGS. 29A, B shows the % inhibition for certain humanized anti-MCAM antibodies as compared to chimeric anti-MCAM antibodies.
[0068] FIG. 30: Treatment with an anti-MCAM antibody reduces inflammation in model of skin inflammation.
[0069] FIGS. 31A, B: Anti-MCAM antibodies inhibit melanoma growth by volume (A) and weight (B) in xenograft model.
DETAILED DESCRIPTION
1. Definitions and Abbreviations
1.1. Definitions
[0070] An "individual" or "subject" as used herein may be any of mammalian animals (e.g., domesticated animals), including human, dog, cat, cattle, horse, goat, pig, swine, sheep, monkey, guinea pig, rat, and mouse. In one embodiment, the individual or subject can be a human.
[0071] "MCAM" (melanoma cell adhesion molecule, also known as CD146 and MUC18) refers to a cell surface glycoprotein belonging to the immunoglobulin superfamily involved in cell adhesion, and in cohesion of the endothelial monolayer at intercellular junctions in vascular tissue. It also promotes tumor progression of many cancers, such as solid tumors, including melanoma and prostate cancer. It is known to interact in a homotypic/homophilic manner and may also bind to other ligands. The human MCAM has the amino acid sequence of SEQ ID NO: 11 (FIG. 11A), which includes five immunoglobulin domains (1: amino acid residues 19-129; 2: amino acid residues 139-242; 3: amino acid residues 244-321; 4: amino acid residues 335-424; and 5: amino acid residues 430-510) shown as SEQ ID NOS:22-26, which are also depicted schematically in FIG. 11B.
[0072] A "laminin .alpha.4 chain" refers to one of the polypeptide chains found in laminin molecules, which are expressed in the basal lamina (of the basement membrane), a protein network foundation for most cells and organs. Laminins are known to bind to cell membranes through plasma membrane molecules and contribute to cell attachment. The laminin .alpha.4 chain typically forms a complex with a laminin .beta.-chain, and a laminin .gamma.-chain. The laminin .alpha.4 chain is found in numerous laminin molecules including, without limitation, laminin 411 (laminin 8 or (4.beta.1.gamma.1); laminin 421 (laminin 9 or .alpha.4.beta.2.gamma.1), and laminin 423 (laminin 14 or .alpha.4.beta.2.gamma.3). There are two main isoforms of the human laminin .alpha.4-chain: GenBank Accession Nos. NP001098676 and NP001098677 as shown in FIGS. 12A-B (amino acid sequences SEQ ID NOS:27-28). "Laminin 411" refers to a trimeric polypeptide complex made up of three polypeptide subunits or chains: .alpha.4-chain, a .beta.1-chain, and a .gamma.1-chain.
[0073] The term "antagonist" is used in the broadest sense, and includes any molecule that partially or fully blocks, inhibits, or neutralizes a qualitative biological activity of an MCAM polypeptide. For the purpose of the present invention, the biological activity preferably is the ability to inhibit the ability of MCAM (i) to specifically bind its ligand: a laminin .alpha.4 chain, e.g., the .alpha.4 chain of laminin 411; and/or (ii) to facilitate an MCAM-expressing cell, e.g., a TH17 cell, to infiltrate into or migrate to a subject's tissue. Antagonists of MCAM can be identified, for example, based upon their ability to inhibit or block the specific binding of MCAM to its ligand: a laminin .alpha.4 chain, e.g., the .alpha.4 chain of laminin 411. MCAM antagonists specifically include, without limitation, antibodies (e.g., antagonist or neutralizing antibodies), including chimeric, humanized and human antibodies and their functional fragments, small molecules, ribozymes, aptamers, peptides, and nucleic acids that encode polypeptide antagonists or antagonist antibodies.
[0074] The term "MCAM antagonist antibody" refers to an antibody which inhibits or neutralizes the activity of MCAM. Such an antibody specifically binds to a polypeptide target involved in the infiltration of an MCAM-expressing cell into the CNS, e.g., MCAM or a laminin .alpha.4 chain (e.g., the .alpha.4 chain of laminin 411).
[0075] A "blocking" antibody, "neutralizing" antibody, or "antagonist" antibody is one which inhibits or reduces a biological activity of the antigen it binds. Such antibodies may substantially or completely inhibit the biological activity of the antigen.
[0076] The terms "specifically binds" or "binds specifically" as used herein means that one member of a specific binding pair will not show any statistically significant binding to molecules other than its specific binding partner. A binding partner may show at least 1000 times the affinity of binding (measured as an apparent association constant) for its specific binding pair partner than a non-specific binding partner. For example, antibodies that bind to MCAM with a binding affinity of 10.sup.7 mole/L or more, typically 10.sup.8 mole/L or more, are said to bind specifically to MCAM.
[0077] The terms "biological activity" and "biologically active" with regard to MCAM refer to its ability to specifically bind its ligand (a laminin .alpha.4 chain, e.g., the .alpha.4 chain of laminin 411) and/or to facilitate the infiltration of MCAM-expressing cells, e.g., TH17 cells, into the CNS.
[0078] The term an "MCAM-expressing cell" refers to a cell of the immune system that expresses MCAM. For example, MCAM expression is enriched on memory T lymphocytes, e.g., TH17 cells.
[0079] The term "binding molecule" as used herein refers to a molecule that specifically binds to a target. The term specifically includes, without limitation, antibodies and antibody fragments (e.g. those comprising one or more of the CDRs described herein), and peptide and non-peptide small molecules.
[0080] "Antibodies" (Abs) and "immunoglobulins" (Igs) are glycoproteins having some common structural characteristics. While antibodies exhibit binding specificity to a specific antigen, immunoglobulins include both antibodies and other antibody-like molecules which lack antigen specificity. Polypeptides of the latter kind can be, for example, produced at low levels by the lymph system and at increased levels by myelomas.
[0081] The term "antibody" used herein may encompass intact monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g. bispecific antibodies) formed from at least two intact antibodies, and antibody fragments, so long as they exhibit the desired biological activity. The term "antigen-binding fragment" of an antibody refers to a portion of the full-length immunoglobulin molecule that specifically binds to the antigen. An antigen-binding fragment of an antibody thus includes an antigen-binding heavy chain, light chain, heavy chain-light chain dimer, Fab fragment, F(ab').sub.2 fragment, Fv fragment, single chain Fv (scFv), diabodies, linear antibodies, and multispecific antibodies formed from antibody fragment(s).
[0082] The term "monoclonal antibody" as used herein refers to an antibody from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are substantially similar and bind the same epitope(s), except for possible variants that may arise during production of the monoclonal antibody, such variants generally being present in minor amounts. Such monoclonal antibody typically includes an antibody comprising a variable region that binds a target, wherein the antibody was obtained by a process that includes the selection of the antibody from a plurality of antibodies. For example, the selection process can be the selection of a unique clone from a plurality of clones, such as a pool of hybridoma clones, phage clones or recombinant DNA clones. It should be understood that the selected antibody can be further altered, for example, to improve affinity for the target, to humanize the antibody, to improve its production in cell culture, to reduce its immunogenicity in vivo, to create a multispecific antibody, etc., and that an antibody comprising the altered variable region sequence is also a monoclonal antibody of this invention. In addition to their specificity, the monoclonal antibody preparations are advantageous in that they are typically uncontaminated by other immunoglobulins. The modifier "monoclonal" indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method. For example, the monoclonal antibodies to be used in accordance with the present invention may be made by a variety of techniques, including the hybridoma method (e.g., Kohler et al., Nature, 256:495 (1975); Harlow et al., Antibodies: A Laboratory Manual, (Cold Spring Harbor Laboratory Press, 2nd ed. 1988); Hammerling et al., in: Monoclonal Antibodies and T-Cell Hybridomas 563-681, (Elsevier, N.Y., 1981), recombinant DNA methods (see, e.g., U.S. Pat. No. 4,816,567), phage display technologies (see, e.g., Clackson et al., Nature, 352:624-628 (1991); Marks et al., J. Mol. Biol., 222:581-597 (1991); Sidhu et al., J. Mol. Biol. 338(2):299-310 (2004); Lee et al., J. Mol. Biol. 340(5):1073-1093 (2004); Fellouse, Proc. Nat. Acad. Sci. USA 101(34):12467-12472 (2004); and Lee et al. J. Immunol. Methods 284(1-2): 119-132 (2004) and technologies for producing human or human-like antibodies from animals that have parts or all of the human immunoglobulin loci or genes encoding human immunoglobulin sequences (see, e.g., WO98/24893, WO/9634096, WO/9633735, and WO/91 10741, Jakobovits et al., Proc. Natl. Acad. Sci. USA, 90:2551 (1993); Jakobovits et al., Nature, 362:255-258 (1993); Bruggemann et al., Year in Immune, 7:33 (1993); U.S. Pat. Nos. 5,545,806, 5,569,825, 5,591,669 (all of GenPharm); U.S. Pat. No. 5,545,807; WO 97/17852, U.S. Pat. Nos. 5,545,807; 5,545,806; 5,569,825; 5,625,126; 5,633,425; and 5,661,016, and Marks et al., Bio/Technology, 10: 779-783 (1992); Lonberg et al., Nature, 368: 856-859 (1994); Morrison, Nature, 368: 812-813 (1994); Fishwild et al., Nature Biotechnology, 14: 845-851 (1996); Neuberger, Nature Biotechnology, 14: 826 (1996); and Lonberg and Huszar, Intern. Rev. Immunol., 13: 65-93 (1995).
[0083] The monoclonal antibodies herein specifically include "chimeric" antibodies in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (U.S. Pat. No. 4,816,567; and Morrison et al., Proc. Natl. Acad. Sci. USA, 81:6851-6855 (1984)). Chimeric antibodies of interest herein include "primatized" antibodies comprising variable domain antigen-binding sequences derived from a non-human primate (e.g. Old World Monkey, Ape etc) and human constant region sequences, as well as "humanized" antibodies.
[0084] "Humanized" forms of non-human (e.g., rodent) antibodies are chimeric antibodies that contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from a hypervariable region of a non-human species (donor antibody) such as mouse, rat, rabbit or nonhuman primate having the desired specificity, affinity, and capacity. In some instances, framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, humanized antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable loops correspond to those of a non-human immunoglobulin and all or substantially all of the FRs are those of a human immunoglobulin sequence. The humanized antibody optionally also will comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin. For further details, see Jones et al., Nature 321:522-525 (1986); Riechmann et al., Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol. 2:593-596 (1992).
[0085] An "intact antibody" herein is one which comprises two antigen binding regions, and an Fc region. Preferably, the intact antibody has a functional Fc region.
[0086] An "antibody (or any other binding molecule) that binds to the same epitope" as a reference antibody (or any other binding molecule) refers to an antibody (or any other binding molecule) that blocks binding of the reference antibody (or any other binding molecule) to its antigen in a competition assay by 50% or more, and conversely, the reference antibody (or any other binding molecule) blocks binding of the antibody to its antigen in a competition assay by 50% or more.
[0087] An "affinity matured" antibody is one with one or more alterations in one or more hypervariable regions thereof which result an improvement in the affinity of the antibody for antigen, compared to a parent antibody which does not possess those alteration(s). Preferred affinity matured antibodies will have nanomolar or even picomolar affinities for the target antigen. Affinity matured antibodies are produced by procedures known in the art. Marks et al. Bio/Technology 10:779-783 (1992) describes affinity maturation by VH and VL domain shuffling. Random mutagenesis of CDR and/or framework residues is described by: Barbas et al. Proc Nat. Acad. Sci, USA 91:3809-3813 (1994); Schier et al. Gene 169:147-155 (1995); Yelton et al. J. Immunol. 155:1994-2004 (1995); Jackson et al., J. Immunol. 154(7):3310-9 (1995); and Hawkins et al, J. Mol. Biol. 226:889-896 (1992).
[0088] The "light chains" of antibodies from any vertebrate species can be assigned to one of two clearly distinct types, called K and X, based on the amino acid sequences of their constant domains. Depending on the amino acid sequence of the constant domain of their heavy chains, intact antibodies can be assigned to different "classes." There are five major classes of intact antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into "subclasses" (isotypes), e.g., IgG1, IgG2, IgG3, IgG4, IgA, and IgA2. The heavy-chain constant domains that correspond to the different classes of antibodies are called .alpha., .delta., .epsilon., .gamma., and .mu., respectively. The subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known.
[0089] The term "variable" refers to the fact that certain portions of the variable domains differ extensively in sequence among antibodies and are used in the binding and specificity of each particular antibody for its particular antigen. However, the variability is not evenly distributed throughout the variable domains of antibodies. It is concentrated in three segments called complementarity-determining regions (CDRs) or hypervariable regions (HVRs) both in the light-chain and heavy-chain variable domains. The more highly conserved portions of variable domains are called the framework (FR). The variable domains of native heavy and light chains each comprise four FR regions, largely adopting a .beta.-sheet configuration, connected by three CDRs, which form loops connecting, and in some cases forming part of, the .beta.-sheet structure. The CDRs in each chain are held together in close proximity by the FR regions and, with the CDRs from the other chain, contribute to the formation of the antigen-binding site of antibodies. The constant domains are not involved directly in binding an antibody to an antigen, but exhibit various effector functions, such as participation of the antibody in antibody-dependent cellular toxicity.
[0090] "Fv" is the minimum antibody fragment which contains a complete antigen-recognition and binding site. In a two-chain Fv species, this region consists of a dimer of one heavy- and one light-chain variable domain in tight, non-covalent association. In a single-chain Fv species, one heavy- and one light-chain variable domain can be covalently linked by a flexible peptide linker such that the light and heavy chains can associate in a "dimeric" structure analogous to that in a two-chain Fv species. It is in this configuration that the three CDRs of each variable domain interact to define an antigen-binding site on the surface of the VH-VL dimer. Collectively, the six CDRs confer antigen-binding specificity to the antibody. However, even a single variable domain (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although at a lower affinity than the entire binding site.
[0091] "Hypervariable region" or "HVR" refers to the amino acid residues of an antibody that are responsible for antigen-binding. The hypervariable region generally comprises amino acid residues from a "complementarity determining region" or "CDR" (Kabat et al., Sequences of Proteins of Immunological Interest, 5.sup.th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)) and/or those residues from a "hypervariable loop" (Chothia and Lesk, J. Mol. Biol. 196: 901-917 (1987)).
[0092] The term "complementarity determining regions" or "CDRs" when used herein refers to parts of immunological receptors that make contact with a specific ligand and determine its specificity. The CDRs of immunological receptors are the most variable part of the receptor protein, giving receptors their diversity, and are carried on six loops at the distal end of the receptor's variable domains, three loops coming from each of the two variable domains of the receptor.
[0093] The term "epitope" is used to refer to binding sites for (monoclonal or polyclonal) antibodies on protein antigens. Typically, an epitope refers to a unit of structure conventionally bound by an immunoglobulin VH-VL pair. Epitopes define the minimum binding site for an antibody, and thus represent the target of specificity of an antibody. Epitopes can be linear or conformational, and can be as small as three amino acids.
[0094] A "small molecule" is defined herein to have a molecular weight below about 600, preferably below about 1000 daltons. Generally, a small molecule is a non-peptide small organic molecule.
[0095] "Isolated," when used to describe the various polypeptides, proteins and antibodies disclosed herein, means polypeptide, protein or antibody that has been identified and separated and/or recovered from a component of its natural environment. Contaminant components of its natural environment are materials that would typically interfere with diagnostic or therapeutic uses for the polypeptide, protein or antibody, and may include enzymes, hormones, and other proteinaceous or non-proteinaceous solutes. In preferred embodiments, the polypeptide, protein or antibody will be purified (1) to a degree sufficient to obtain at least 15 residues of N-terminal or internal amino acid sequence by use of a spinning cup sequenator, or (2) to homogeneity by SDS-PAGE under non-reducing or reducing conditions using Coomassie blue or, preferably, silver stain. Isolated polypeptide, protein or antibody includes polypeptide, protein or antibody in situ within recombinant cells, since at least one component of the associated natural environment will not be present. Ordinarily, however, isolated polypeptide, protein or antibody will be prepared by at least one purification step.
[0096] The terms "affinity", "binding affinity" and "K.sub.d" refer to the equilibrium dissociation constant (expressed in units of concentration) associated with each MCAM binding molecule--target complex, such as between an anti-MCAM antibody and MCAM. The binding affinity is directly related to the ratio of the off-rate constant (generally reported in units of inverse time, e.g., seconds.sup.-1) to the on-rate constant (generally reported in units of concentration per unit time, e.g., molar/second). The binding affinity may be determined by, for example, an ELISA assay, kinetic exclusion assay or surface plasmon resonance. It is noted that certain epitopes can occur repetitively (multivalent) on a cell surface and that the dissociation constant (koff) for the binding of an antibody to a repetitive epitope may be greatly diminished over the dissociation constant for the reaction of the same antibody with the corresponding ligand in univalent form. The diminished dissociation constant arises because when one antibody-ligand bond dissociates, other bonds hold the bivalent (or multivalent) antibody to the multivalent ligand, allowing the dissociated bond to form again. The dissociation constant for the reaction between bivalent (or multivalent) Ab and multivalent ligand has been termed the functional affinity to contrast it with intrinsic affinity, which is the association constant for an antibodies representative individual site.
[0097] The terms "dissociation", "dissociation rate" and "k.sub.off" as used herein, are intended to refer to the off rate constant for dissociation of a binding molecule, such as an antibody, from the binding molecule/target, e.g. antibody/antigen complex.
[0098] The terms "association", "association rate" and "k.sub.on" as used herein, are intended to refer to the on rate constant for association of a binding molecule with a target, such as an antibody with an antigen, to form a complex.
[0099] The terms "effective concentration" and "EC.sub.50" as used herein, are intended to refer to the concentration of a binding molecule (e/g/ antibody) capable of interacting with sufficient quantities of target molecules to produce an effect on approximately 50% of the treated cells.
[0100] As used herein, "treatment" (and grammatical variations thereof such as "treat" or "treating") refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis/prevention, or during the course of clinical pathology. The term refers to both therapeutic treatment and prophylactic or preventative measures, wherein the object is to prevent or slow down (lessen) an undesired physiological change or disorder. For purposes of this invention, beneficial or desired clinical results include, but are not limited to, alleviation of symptoms, diminishment of extent of disease, stabilized (i.e., not worsening) state of disease, delay or slowing of disease progression, amelioration or palliation of the disease state, and remission (whether partial or total), whether detectable or undetectable. "Treatment" can also mean prolonging survival as compared to expected survival if not receiving treatment. Those in need of treatment include those already with the condition or disorder as well as those prone to have the condition or disorder or those in which the condition or disorder is to be prevented.
[0101] "Chronic" administration refers to administration of the agent(s) in a continuous mode as opposed to an acute mode, so as to maintain the desired effect for an extended period of time.
[0102] "Intermittent" administration is treatment that is not consecutively done without interruption, but rather is cyclic in nature.
[0103] An "effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired prophylactic or therapeutic result. An effective amount refers to the amount of active compound or pharmaceutical agent that elicits the biological or medicinal response in a tissue, system, animal, individual or human that is being sought by a researcher, veterinarian, medical doctor or other clinician, which includes one or more of the following:
[0104] (A) preventing the disease; for example, preventing an inflammatory disease, such as a neuroinflammatory disease, condition or disorder in an individual that may be predisposed to the disease, condition or disorder but does not yet experience or display the pathology or symptoms of the disease,
[0105] (B) inhibiting the disease; for example, inhibiting an inflammatory disease, such as a neuroinflammatory disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptoms of the disease, condition or disorder (i.e., arresting further development of the pathology and/or symptoms), and
[0106] (C) ameliorating the disease; for example, ameliorating an inflammatory disease, such as a neuroinflammatory disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptoms of the disease, condition or disorder (i.e., reversing the pathology and/or symptoms).
[0107] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art. In some cases, terms with commonly understood meanings are defined herein for clarity and/or for ready reference, and the inclusion of such definitions herein should not necessarily be construed to represent a substantial difference over what is generally understood in the art. The techniques and procedures described or referenced herein are generally well understood and commonly employed using conventional methodology by those skilled in the art, such as, for example, the widely utilized molecular cloning methodologies described in Sambrook et al., Molecular Cloning: A Laboratory Manual 2nd. edition (1989) Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. As appropriate, procedures involving the use of commercially available kits and reagents are generally carried out in accordance with manufacturer defined protocols and/or parameters unless otherwise noted. Before the present methods, kits and uses therefore are described, it is to be understood that this invention is not limited to the particular methodology, protocols, cell lines, animal species or genera, constructs, and reagents described as such may, of course, vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only, and is not intended to limit the scope of the present invention which will be limited only by the appended claims.
[0108] It must be noted that as used herein, the singular forms "a", "and", and "the" include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to "an antibody" includes a plurality of such antibodies and reference to "the dosage" includes reference to one or more dosages and equivalents thereof known to those skilled in the art, and so forth. Throughout this specification and claims, the word "comprise," or variations such as "comprises" or "comprising," will be understood to imply the inclusion of a stated integer or group of integers but not the exclusion of any other integer or group of integers.
1.2. Abbreviations
TABLE-US-00001 [0109] Abs antibodies CDR complementarity determining region CFA complete Freund's adjuvant CFSE carboxyfluorescein succinimidyl ester CNS central nervous system DAPI 4',6-diamidino-2-phenylindole DN dopamine-containing neuron EAE experimental autoimmune encephalomyelitis ECM extracellular matrix FACS fluorescence Activated cell sorting FR Framework Region IFA incomplete Freund's adjuvant Igs immunoglobulins MCAM melanoma cell adhesion molecule MOG myelin oligodendrocyte glycoprotein (MOG) MS multiple sclerosis PD Parkinson's disease PMA phorbol myristate acetate
2. MCAM
[0110] MCAM (melanoma cell adhesion molecule) is a cell-surface glycoprotein originally identified as a melanoma antigen, whose expression is associated with tumor progression and the development of metastatic potential. MCAM is a 113 kDa cell surface integral membrane glycoprotein composed of a signal peptide, five immunoglobulin-like domains (1, 2, 3, 4, and 5; or V-V-C2-C2-C2), a transmembrane region, and a short cytoplasmic tail. See, e.g., Lehmann et al., Proc. Nat'l Acad. Sci. USA 86: 9891-9895 (1989) and FIG. 11B. MCAM is a member of the immunoglobulin superfamily and has significant sequence homology to a number of cell adhesion molecules of the Ig superfamily, including BEN (Pourquie et al., Proc. Nat'l Acad. Sci. USA 89: 5261-5265 (1992)), neural-cell adhesion molecule (N-CAM) (Owens et al., Proc. Nat'Acad. Sci. USA 84: 294-298 (1987)), myelin-associated glycoprotein (MAG) (Lai et al., Proc. Nat'l Acad. Sci. USA 84: 4337-4341 (1987)), deleted in colorectal cancer protein (DCC) (Hedrick et al., Genes Devel. 8: 1174-1183 (1994)), and gicerin (Taira et al., Neuron 12: 861-872 (1994)).
[0111] The expression of MCAM has been detected in relatively limited spectrum of normal human tissues and in a variety of malignant neoplasms. In normal adult tissues, MCAM is expressed on endothelial cells, smooth muscle cells (Shih et al., Lab. Invest. 75: 377-388 (1996); Sers et al., Cancer Res. 54: 5689-5694 (1994)), a subpopulation of activated T lymphocytes (Pickl et al., J. Immunol. 158: 2107-2115 (1997)), and intermediate trophoblasts (Shih et al., supra). MCAM is also expressed on a variety of malignant neoplasms including smooth muscle neoplasms (Leiomyomas and leiomyosarcomas), tumors of vascular origin (angiosarcomas and Kaposi's sarcomas), placental site trophoblastic tumors, choriocarcinomas, and melanomas (Shih et al., Clinical Cancer Res. 2: 569-575 (1996); Holzmann et al., Int. J. Cancer 39: 466-471 (1987)). The expression of MUC18 correlates directly with the metastatic potential of human melanoma cells (Bar-Eli, Cancer Metastasis, 18: 377-385 (1999)).
[0112] A number of studies have identified MCAM as a marker of tumor progression and metastasis in melanomas. The expression of MCAM is absent in normal melanocytes and benign nevi but prominent on many primary melanomas and in most metastatic lesions (Lehmann et al., supra; Shih et al., supra). MCAM expression correlates well with tumor vertical thickness and metastasis formation, and greater than 80% of metastatic lesions express MCAM (Lehmann et al., supra; Xie et al., Cancer Res. 57: 2295-2303 (1997); and Shih et al., supra). Modulators of MCAM have been generated to treat melanomas. See, e.g., U.S. Pat. No. 7,067,131. Recently, MCAM modulation has been suggested to identify and select inflammatory cytokine-secreting T cells or their precursors to treat various inflammatory conditions. See, e.g., U.S. Published Patent Application No. 2011/0014183.
3. Neuroinflammatory Conditions, Multiple Sclerosis, and Parkinson Disease
[0113] A neuroinflammatory condition refers to a condition associated with inflammation of the nervous system, in an embodiment the central nervous system (CNS), and which is associated with cell/tissue damage. It is typically characterized by, for example, increased glial activation, increased pro-inflammatory cytokine/chemokine levels (e.g., TNF.alpha., INF.gamma., IL-1.beta.), increased blood-brain-barrier permeability, and/or increased immune cell (e.g., leukocyte) recruitment/invasion to the CNS. It may refer to, for example, chronic neuroinflammation, such as an inflammation associated with chronic activation of cells of the immune system (i.e., autoimmune-associated neuroinflammation). Such chronic neuroinflammation can be observed in, for example, multiple sclerosis (MS). Additionally, Parkinson's disease (PD) is a neurodegenerative disease displaying neuroinflammation, for example, activated microglia and infiltrating T cells.
[0114] Multiple sclerosis, as a progressive neurological autoimmune disease, results from chronic, pathological inflammation (Yednock et al., Nature 356: 63-66 (1992); Baron et al., J. Exp. Med. 177: 57-68 (1993)). MS affects an estimated 250,000 to 350,000 people in the United States. Multiple sclerosis is thought to be the result of a specific autoimmune reaction wherein certain leukocytes attack and initiate the destruction of myelin, the insulating sheath covering nerve fibers. The onset of MS may be dramatic or so mild as to not cause a patient to seek medical attention. The most common symptoms include weakness in one or more limbs, visual blurring due to optic neuritis, sensory disturbances, diplopia, and ataxia. The course of disease may be stratified into three general categories: (1) relapsing MS, (2) chronic progressive MS, and (3) inactive MS.
[0115] Relapsing MS is generally characterized by recurrent attacks of neurologic dysfunction. MS attacks generally evolve over days to weeks and may be followed by complete, partial, or no recovery. Recovery from attacks generally occurs within weeks to several months from the peak of symptoms, although rarely some recovery may continue for 2 or more years.
[0116] Chronic progressive MS results in gradually progressive worsening without periods of stabilization or remission. This form develops in patients with a prior history of relapsing MS, although in 20% of patients, no relapses can be recalled. Acute relapses also may occur during the progressive course of MS.
[0117] A third form is inactive MS. Inactive MS is characterized by fixed neurologic deficits of variable magnitude. Most patients with inactive MS have an earlier history of relapsing MS. The course of MS is also dependent on the age of the patient. For example, favorable prognostic factors include early onset (excluding childhood), a relapsing course and little residual disability 5 years after onset. By contrast, poor prognosis is associated with a late age of onset (i.e., age 40 or older) and a progressive course. These variables are interdependent, since chronic progressive MS tends to begin at a later age that relapsing MS. Disability from chronic progressive MS is usually due to progressive paraplegia or quadriplegia in individual patients.
[0118] Parkinson's disease (PD) is a progressive neurodegenerative disease displaying primary clinical features of motor abnormalities, e.g., resting tremor, bradykinesia, and rigidity. PD is characterized by the loss of dopamine-containing neuron (DN) cells in the substantia nigra parts compacta (Forno, J. Neurophthol. Exp. Neurol. 55: 259-272 (1996)). One of the hallmarks of PD is neuroinflammation characterized by activated microglia and infiltrating T cells. Although studies have suggested various mechanisms for PD, such as mitochondrial dysfunction, oxidative stress, and impairment of protein degradation machinery, the cause of PD remains elusive (Dauer et al., Neuron 39: 889-909 (2003)). Recent findings have indicated that both innate and adaptive immunity may play important roles in the pathogenesis of PD (Stone et al., Antioxid. Redox. Signal. 11: 2151-2166 (2009)). Particularly, it has been shown in the animal model of PD that both activated microglia and T lymphocytes contribute significantly to neurodegeneration. See, e.g., Brochard et al., J. Clin. Invest. 119: 182-192 (2009). It has been hypothesized that CD4 positive T cells (e.g., proinflammatory T17 cells) mediate cytotoxicity by activating microglia in PD and/or exert a direct toxic effect on substantia nigra DNs (Appel, J. Clin. Invest. 119: 13-15 (2009)).
4. Autoimmune Diseases
[0119] An autoimmune disease herein is a disease or disorder arising from and directed against an individual's own tissues or a co-segregate or manifestation thereof or resulting condition therefrom. Examples of autoimmune diseases or disorders include, but are not limited to arthritis (rheumatoid arthritis such as acute arthritis, chronic rheumatoid arthritis, gout or gouty arthritis, acute gouty arthritis, acute immunological arthritis, chronic inflammatory arthritis, degenerative arthritis, type II collagen-induced arthritis, infectious arthritis, Lyme arthritis, proliferative arthritis, psoriatic arthritis, Still's disease, vertebral arthritis, and juvenile-onset rheumatoid arthritis, osteoarthritis, arthritis chronica progrediente, arthritis deformans, polyarthritis chronica primaria, reactive arthritis, and ankylosing spondylitis), inflammatory hyperproliferative skin diseases, psoriasis such as plaque psoriasis, gutatte psoriasis, pustular psoriasis, and psoriasis of the nails, atopy including atopic diseases such as hay fever and Job's syndrome, dermatitis including contact dermatitis, chronic contact dermatitis, exfoliative dermatitis, allergic dermatitis, allergic contact dermatitis, dermatitis herpetiformis, nummular dermatitis, seborrheic dermatitis, non-specific dermatitis, primary irritant contact dermatitis, and atopic dermatitis, x-linked hyper IgM syndrome, allergic intraocular inflammatory diseases, urticaria such as chronic allergic urticaria and chronic idiopathic urticaria, including chronic autoimmune urticaria, myositis, polymyositis/dermatomyositis, juvenile dermatomyositis, toxic epidermal necrolysis, scleroderma (including systemic scleroderma), sclerosis such as systemic sclerosis, multiple sclerosis (MS) such as spino-optical MS, primary progressive MS (PPMS), and relapsing remitting MS (RRMS), progressive systemic sclerosis, atherosclerosis, arteriosclerosis, sclerosis disseminata, ataxic sclerosis, neuromyelitis optica (NMO), inflammatory bowel disease (IBD) (for example, Crohn's disease, autoimmune-mediated gastrointestinal diseases, colitis such as ulcerative colitis, colitis ulcerosa, microscopic colitis, collagenous colitis, colitis polyposa, necrotizing enterocolitis, and transmural colitis, and autoimmune inflammatory bowel disease), bowel inflammation, pyoderma gangrenosum, erythema nodosum, primary sclerosing cholangitis, respiratory distress syndrome, including adult or acute respiratory distress syndrome (ARDS), meningitis, inflammation of all or part of the uvea, iritis, choroiditis, an autoimmune hematological disorder, rheumatoid spondylitis, rheumatoid synovitis, hereditary angioedema, cranial nerve damage as in meningitis, herpes gestationis, pemphigoid gestationis, pruritis scroti, autoimmune premature ovarian failure, sudden hearing loss due to an autoimmune condition, IgE-mediated diseases such as anaphylaxis and allergic and atopic rhinitis, encephalitis such as Rasmussen's encephalitis and limbic and/or brainstem encephalitis, uveitis, such as anterior uveitis, acute anterior uveitis, granulomatous uveitis, nongranulomatous uveitis, phacoantigenic uveitis, posterior uveitis, or autoimrnune uveitis, glomerulonephritis (GN) with and without nephrotic syndrome such as chronic or acute glomerulonephritis such as primary GN, immune-mediated GN, membranous GN (membranous nephropathy), idiopathic membranous GN or idiopathic membranous nephropathy, membrano- or membranous proliferative GN (MPGN), including Type I and Type II, and rapidly progressive GN, proliferative nephritis, autoimmune polyglandular endocrine failure, balanitis including balanitis circumscripta plasmacellularis, balanoposthitis, erythema annulare centrifugum, erythema dyschromicum perstans, eythema multiform, granuloma annulare, lichen nitidus, lichen sclerosus et atrophicus, lichen simplex chronicus, lichen spinulosus, lichen planus, lamellar ichthyosis, epidermolytic hyperkeratosis, premalignant keratosis, pyoderma gangrenosum, allergic conditions and responses, allergic reaction, eczema including allergic or atopic eczema, asteatotic eczema, dyshidrotic eczema, and vesicular palmoplantar eczema, asthma such as asthma bronchiale, bronchial asthma, and auto-immune asthma, conditions involving infiltration of T cells and chronic inflammatory responses, immune reactions against foreign antigens such as fetal A-B-O blood groups during pregnancy, chronic pulmonary inflammatory disease, autoimmune myocarditis, leukocyte adhesion deficiency, lupus, including lupus nephritis, lupus cerebritis, pediatric lupus, non-renal lupus, extra-renal lupus, discoid lupus and discoid lupus erythematosus, alopecia lupus, systemic lupus erythematosus (SLE) such as cutaneous SLE or subacute cutaneous SLE, neonatal lupus syndrome (NLE), and lupus erythematosus disseminatus, juvenile onset (Type I) diabetes mellitus, including pediatric insulin-dependent diabetes mellitus (IDDM), adult onset diabetes mellitus (Type II diabetes), autoimmune diabetes, idiopathic diabetes insipidus, diabetic retinopathy, diabetic nephropathy, diabetic large-artery disorder, immune responses associated with acute and delayed hypersensitivity mediated by cytokines and T-lymphocytes, tuberculosis, sarcoidosis, granulomatosis including lymphomatoid granulomatosis, Wegener's granulomatosis, agranulocytosis, vasculitides, including vasculitis, large-vessel vasculitis (including polymyalgia rheumatica and giant-cell (Takayasu's) arteritis), medium-vessel vasculitis (including Kawasaki's disease and polyarteritis nodosa/periarteritis nodosa), microscopic polyarteritis, immunovasculitis, CNS vasculitis, cutaneous vasculitis, hypersensitivity vasculitis, necrotizing vasculitis such as systemic necrotizing vasculitis, and ANCA-associated vasculitis, such as Churg-Strauss vasculitis or syndrome (CSS) and ANCA-associated small-vessel vasculitis, temporal arteritis, aplastic anemia, autoimmune aplastic anemia, Coombs positive anemia, Diamond Blackfan anemia, hemolytic anemia or immune hemolytic anemia including autoimmune hemolytic anemia (AIHA), pernicious anemia (anemia perniciosa), Addison's disease, pure red cell anemia or aplasia (PRCA), Factor VIII deficiency, hemophilia A, autoimmune neutropenia, pancytopenia, leukopenia, diseases involving leukocyte diapedesis, CNS inflammatory disorders, multiple organ injury syndrome such as those secondary to septicemia, trauma or hemorrhage, antigen-antibody complex-mediated diseases, anti-glomerular basement membrane disease, anti-phospholipid antibody syndrome, allergic neuritis, Behcet's disease/syndrome, Castleman's syndrome, Goodpasture's syndrome, Reynaud's syndrome, Sjogren's syndrome, Stevens-Johnson syndrome, pemphigoid such as pemphigoid bullous and skin pemphigoid, pemphigus (including pemphigus vulgaris, pemphigus foliaceus, pemphigus mucus-membrane pemphigoid, and pemphigus erythematosus), autoimmune polyendocrinopathies, Reiter's disease or syndrome, thermal injury, preeclampsia, an immune complex disorder such as immune complex nephritis, antibody-mediated nephritis, polyneuropathies, chronic neuropathy such as IgM polyneuropathies or IgM-mediated neuropathy, thrombocytopenia (as developed by myocardial infarction patients, for example), including thrombotic thrombocytopenic purpura (TTP), post-transfusion purpura (PTP), heparin-induced thrombocytopenia, and autoimmune or immune-mediated thrombocytopenia such as idiopathic thrombocytopenic purpura (ITP) including chronic or acute ITP, scleritis such as idiopathic cerato-scleritis, episcleritis, autoimmune disease of the testis and ovary including autoimmune orchitis and oophoritis, primary hypothyroidism, hypoparathyroidism, autoimmune endocrine diseases including thyroiditis such as autoimmune thyroiditis, Hashimoto's disease, chronic thyroiditis (Hashimoto's thyroiditis), or subacute thyroiditis, autoimmune thyroid disease, idiopathic hypothyroidism, Grave's disease, polyglandular syndromes such as autoimmune polyglandular syndromes (or polyglandular endocrinopathy syndromes), paraneoplastic syndromes, including neurologic paraneoplastic syndromes such as Lambert-Eaton myasthenic syndrome or Eaton-Lambert syndrome, stiff-man or stiff-person syndrome, encephalomyelitis such as allergic encephalomyelitis or encephalomyelitis allergica and experimental allergic encephalomyelitis (EAE), myasthenia gravis such as thymoma-associated myasthenia gravis, cerebellar degeneration, neuromyotonia, opsoclonus or opsoclonus myoclonus syndrome (OMS), and sensory neuropathy, multifocal motor neuropathy, Sheehan's syndrome, autoimmune hepatitis, chronic hepatitis, lupoid hepatitis, giant-cell hepatitis, chronic active hepatitis or autoimmune chronic active hepatitis, lymphoid interstitial pneumonitis (LIP), bronchiolitis obliterans (non-transplant) vs NSIP, Guillain-Barre syndrome, Berger's disease (IgA nephropathy), idiopathic IgA nephropathy, linear IgA dermatosis, acute febrile neutrophilic dermatosis, subcorneal pustular dermatosis, transient acantholytic dermatosis, cirrhosis such as primary biliary cirrhosis and pneumonocirrhosis, autoimmune enteropathy syndrome, Celiac or Coeliac disease, celiac sprue (gluten enteropathy), refractory sprue, idiopathic sprue, cryoglobulinemia, amylotrophic lateral sclerosis (ALS; Lou Gehrig's disease), coronary artery disease, autoimmune ear disease such as autoimmune inner ear disease (AIED), autoimmune hearing loss, polychondritis such as refractory or relapsed or relapsing polychondritis, pulmonary alveolar proteinosis, Cogan's syndrome/nonsyphilitic interstitial keratitis, Bell's palsy, Sweet's disease/syndrome, rosacea autoimmune, zoster-associated pain, amyloidosis, a non-cancerous lymphocytosis, a primary lymphocytosis, which includes monoclonal B cell lymphocytosis (e.g., benign monoclonal gammopathy and monoclonal gammopathy of undetermined significance, MGUS), peripheral neuropathy, paraneoplastic syndrome, channelopathies such as epilepsy, migraine, arrhythmia, muscular disorders, deafness, blindness, periodic paralysis, and channelopathies of the CNS, autism, inflammatory myopathy, focal or segmental or focal segmental glomerulosclerosis (FSGS), endocrine opthalmopathy, uveoretinitis, chorioretinitis, autoimmune hepatological disorder, fibromyalgia, multiple endocrine failure, Schmidt's syndrome, adrenalitis, gastric atrophy, presenile dementia, demyelinating diseases such as autoimmune demyelinating diseases and chronic inflammatory demyelinating polyneuropathy, Dressler's syndrome, alopecia areata, alopecia totalis, CREST syndrome (calcinosis, Raynaud's phenomenon, esophageal dysmotility, sclerodactyly, and telangiectasia), male and female autoimmune infertility, e.g., due to anti-spermatozoan antibodies, mixed connective tissue disease, Chagas' disease, rheumatic fever, recurrent abortion, farmer's lung, erythema multiforme, post-cardiotomy syndrome, Cushing's syndrome, bird-fancier's lung, allergic granulomatous angiitis, benign lymphocytic angiitis, Alport's syndrome, alveolitis such as allergic alveolitis and fibrosing alveolitis, interstitial lung disease, transfusion reaction, leprosy, malaria, parasitic diseases such as leishmaniasis, kypanosomiasis, schistosomiasis, ascariasis, aspergillosis, Sampter's syndrome, Caplan's syndrome, dengue, endocarditis, endomyocardial fibrosis, diffuse interstitial pulmonary fibrosis, interstitial lung fibrosis, pulmonary fibrosis, idiopathic pulmonary fibrosis, cystic fibrosis, endophthalmitis, erythema elevatum et diutinum, erythroblastosis fetalis, eosinophilic faciitis, Shulman's syndrome, Felty's syndrome, flariasis, cyclitis such as chronic cyclitis, heterochronic cyclitis, iridocyclitis (acute or chronic), or Fuch's cyclitis, Henoch-Schonlein purpura, human immunodeficiency virus (HIV) infection, SCID, acquired immune deficiency syndrome (AIDS), echovirus infection, sepsis, endotoxemia, pancreatitis, thyroxicosis, parvovirus infection, rubella virus infection, post-vaccination syndromes, congenital rubella infection, Epstein-Barr virus infection, mumps, Evan's syndrome, autoimmune gonadal failure, Sydenham's chorea, post-streptococcal nephritis, thromboangitis ubiterans, thyrotoxicosis, tabes dorsalis, chorioiditis, giant-cell polymyalgia, chronic hypersensitivity pneumonitis, keratoconjunctivitis sicca, epidemic keratoconjunctivitis, idiopathic nephritic syndrome, minimal change nephropathy, benign familial and ischemia-reperfusion injury, transplant organ reperfusion, retinal autoimmunity, joint inflammation, bronchitis, chronic obstructive airway/pulmonary disease, silicosis, aphthae, aphthous stomatitis, arteriosclerotic disorders, aspermiogenese, autoimmune hemolysis, Boeck's disease, cryoglobulinemia, Dupuytren's contracture, endophthalmia phacoanaphylactica, enteritis allergica, erythema nodosum leprosum, idiopathic facial paralysis, chronic fatigue syndrome, febris rheumatica, Hamman-Rich's disease, sensoneural hearing loss, haemoglobinuria paroxysmatica, hypogonadism, ileitis regionalis, leucopenia, mononucleosis infectiosa, traverse myelitis, primary idiopathic myxedema, nephrosis, ophthalmia symphatica, orchitis granulomatosa, pancreatitis, polyradiculitis acuta, pyoderma gangrenosum, Quervain's thyreoiditis, acquired spenic atrophy, non-malignant thymoma, vitiligo, toxic-shock syndrome, food poisoning, conditions involving infiltration of T cells, leukocyte-adhesion deficiency, immune responses associated with acute and delayed hypersensitivity mediated by cytokines and T-lymphocytes, diseases involving leukocyte diapedesis, multiple organ injury syndrome, antigen-antibody complex-mediated diseases, antiglomerular basement membrane disease, allergic neuritis, autoimmune polyendocrinopathies, oophoritis, primary myxedema, autoimmune atrophic gastritis, sympathetic ophthalmia, rheumatic diseases, mixed connective tissue disease, nephrotic syndrome, insulitis, polyendocrine failure, autoimmune polyglandular syndrome type I, adult-onset idiopathic hypoparathyroidism (AOIH), cardiomyopathy such as dilated cardiomyopathy, epidermolisis bullosa acquisita (EBA), hemochromatosis, myocarditis, nephrotic syndrome, primary sclerosing cholangitis, purulent or nonpurulent sinusitis, acute or chronic sinusitis, ethmoid, frontal, maxillary, or sphenoid sinusitis, an eosinophil-related disorder such as eosinophilia, pulmonary infiltration eosinophilia, eosinophilia-myalgia syndrome, Loffler's syndrome, chronic eosinophilic pneumonia, tropical pulmonary eosinophilia, bronchopneumonic aspergillosis, aspergilloma, or granulomas containing eosinophils, anaphylaxis, seronegative spondyloarthritides, polyendocrine autoimmune disease, sclerosing cholangitis, sclera, episclera, chronic mucocutaneous candidiasis, Bruton's syndrome, transient hypogammaglobulinemia of infancy, Wiskott-Aldrich syndrome, ataxia telangiectasia syndrome, angiectasis, autoimmune disorders associated with collagen disease, rheumatism, neurological disease, lymphadenitis, reduction in blood pressure response, vascular dysfunction, tissue injury, cardiovascular ischemia, hyperalgesia, renal ischemia, cerebral ischemia, and disease accompanying vascularization, allergic hypersensitivity disorders, glomerulonephritides, reperfusion injury, ischemic re-perfusion disorder, reperfusion injury of myocardial or other tissues, lymphomatous tracheobronchitis, inflammatory dermatoses, dermatoses with acute inflammatory components, multiple organ failure, bullous diseases, renal cortical necrosis, acute purulent meningitis or other central nervous system inflammatory disorders, ocular and orbital inflammatory disorders, granulocyte transfusion-associated syndromes, cytokine-induced toxicity, narcolepsy, acute serious inflammation, chronic intractable inflammation, pyelitis, endarterial hyperplasia, peptic ulcer, valvulitis, and endometriosis.
5. Cancer
[0120] Cancer or a cancerous condition is the physiological condition in mammals that is typically characterized by unregulated cell growth/proliferation. Included in this definition are benign and malignant cancers, as well metastatic cancers. Also included are solid tumors and hematopoietic malignancies. Metastatic cancer refers to a cancer that has spread from the place where it first started to another place in the body. Tumors formed by metastatic cancer cells are called a metastatic tumor or a metastasis (which is also used to refer to the process by which cancer cells spread to other parts of the body). In general, metastatic cancer has the same name and same type of cancer cells as the original, or primary, cancer. Metastatic cancer includes prostate cancer, lung cancer, and pancreas cancer. By prostate cancer or conditions related to prostate cancer is meant the malignant growth of abnormal cells in the prostate gland, capable of invading and destroying other prostate cells, and spreading (metastasizing) to other parts of the body, including bones, lungs, liver, and lymph nodes. By lung cancer or conditions related to lung cancer is meant the malignant growth of abnormal cells in the lungs, capable of invading and destroying other lung cells, and spreading (metastasizing) to other parts of the body, including the adrenal gland, and liver. By pancreatic cancer or conditions related to pancreatic cancer is meant the malignant growth of abnormal cells in the pancreas, capable of invading and destroying other pancreas cells, and spreading (metastasizing) to other parts of the body, including the liver, lungs, and peritoneum.
6. MCAM Antagonists
[0121] The present invention provides antagonists of MCAM. Such antagonists encompass those that directly act upon MCAM (e.g., an anti-MCAM antibody) and those that indirectly affect MCAM activity (e.g., an anti-laminin .alpha.4 chain antibody). Such antagonists are useful, for example, for treating a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS. In one embodiment, a composition comprising an MCAM antagonist is useful for reducing inflammation in a mammalian subject. In another embodiment, such a composition is useful for partially or fully inhibiting CNS infiltration of MCAM-expressing cells. Examples of MCAM antagonists include, without limitation, antagonist or neutralizing antibodies or antibody fragments against one or more domains, e.g., an immunoglobulin domain of a native sequence MCAM polypeptide or a domain of a native sequence laminin .alpha.4 chain polypeptide (e.g., the .alpha.4 chain of laminin 411), small molecules, ribozymes, aptamers, peptides, and nucleic acids that encode polypeptide antagonists or antagonist antibodies. Reference to "an" antagonist encompasses a single antagonist. In one embodiment, the MCAM antagonists are antibodies including, without limitation, chimeric, humanized and human antibodies and their functional fragments.
[0122] In a preferred embodiment, the laminin .alpha.4 chain is an .alpha.4 chain of laminin 411. In another preferred embodiment, the MCAM antagonist blocks the interaction of an MCAM domain comprising the amino acid sequence of SEQ ID NO:22 and/or SEQ ID NO:23 with a laminin .alpha.4 chain.
[0123] 6.1 Screening Assays to Identify MCAM Antagonists
[0124] The present invention includes screening assays to identify MCAM antagonists, which find utility in the treatment of inflammatory conditions characterized by infiltration of MCAM-expressing cells into the central nervous system (CNS).
[0125] In one aspect, the invention concerns a method for identifying an inhibitor of CNS infiltration by MCAM-expressing cells comprising the steps of: (a) incubating a population of cells expressing a laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411, with MCAM, in the presence or absence of a candidate molecule; (b) monitoring the level of binding of MCAM to the cells; and (c) identifying said candidate molecule as an inhibitor of CNS infiltration by MCAM-expressing cells if the level of MCAM binding is lower in the presence than in the absence of said candidate molecule. In one embodiment, the candidate molecule is selected from the group consisting of a small molecule, a peptide, a polypeptide, and an antibody. Those of ordinary skill in the art will appreciate that other types of candidate molecule may be suitable. In another embodiment, the level of binding of MCAM is monitored by known techniques including, without limitation, fluorescent microscopy, FACS, and ELISA. In one other embodiment, the cells expressing a laminin .alpha.4 chain are endothelial cells. In a preferred embodiment, the laminin .alpha.4 chain is an .alpha.4 chain of laminin 411.
[0126] Screening assays for antagonist drug candidates may be designed to identify compounds that bind or complex with MCAM (including a subunit or other fragment thereof) or with an MCAM ligand, such as a laminin .alpha.4 chain (e.g., an .alpha.4 chain of laminin 411), or otherwise interfere with the interaction of MCAM with other cellular proteins, thereby interfering with the interaction of MCAM with its ligand, e.g., a laminin .alpha.4 chain. The screening assays provided herein include assays amenable to high-throughput screening of chemical libraries, making them particularly suitable for identifying small molecule drug candidates. Generally, binding assays and activity assays are provided.
[0127] The assays can be performed in a variety of formats, including, without limitation, protein-protein binding assays, biochemical screening assays, immunoassays, and cell-based assays, which are well characterized in the art.
[0128] All assays for antagonists and agonists are common in that they call for contacting the drug candidate with an MCAM polypeptide, or an MCAM ligand polypeptide, e.g., a laminin .alpha.4 chain, or a fragment of such polypeptides (specifically including MCAM and laminin .alpha.4 chains) under conditions and for a time sufficient to allow these two components to interact.
[0129] For example, human MCAM is a 646 amino acid polypeptide, the sequence of which is available from the GenBank database under Accession Number AAA20922.1 (CAA48332) (SEQ ID NO: 11; FIG. 11A). Amino acid sequences for human laminin .alpha.4-chain are available from the GenBank database under Accession Nos. NP001098676 and NP001098677 (SEQ ID NOS: 27-28; FIGS. 12A-B). The making of antibodies or small molecules binding to such polypeptides is well within the skill of the ordinary artisan.
[0130] In binding assays, the interaction is binding, and the complex formed can be isolated or detected in the reaction mixture. In a particular embodiment, either the MCAM or MCAM ligand polypeptide or the drug candidate is immobilized on a solid phase, e.g., on a microtiter plate, by covalent or non-covalent attachments. Non-covalent attachment generally is accomplished by coating the solid surface with a solution of the MCAM or MCAM ligand polypeptide and drying. Alternatively, an immobilized antibody, e.g., a monoclonal antibody, specific for the MCAM or MCAM ligand polypeptide to be immobilized can be used to anchor it to a solid surface. The assay is performed by adding the non-immobilized component, which may be labeled by a detectable label, to the immobilized component, e.g., the coated surface containing the anchored component. When the reaction is complete, the non-reacted components are removed, e.g., by washing, and complexes anchored on the solid surface are detected. When the originally non-immobilized component carries a detectable label, the detection of label immobilized on the surface indicates that complexing occurred. Where the originally non-immobilized component does not carry a label, complexing can be detected, for example, by using a labeled antibody specifically binding the immobilized complex.
[0131] If the candidate compound is a polypeptide which interacts with but does not bind to MCAM or the MCAM ligand polypeptide, its interaction with the respective polypeptide can be assayed by methods well known for detecting protein-protein interactions. Such assays include traditional approaches, such as, e.g., cross-linking, co-immunoprecipitation, and co-purification through gradients or chromatographic columns. In addition, protein-protein interactions can be monitored by using a yeast-based genetic system described by Fields and co-workers (Fields and Song, Nature (London), 340:245-246 (1989); Chien et al., Proc. Natl. Acad. Sci. USA, 88:9578-9582 (1991)) as disclosed by Chevray and Nathans, Proc. Natl. Acad. Sci. USA, 89: 5789-5793 (1991).
[0132] Compounds that interfere with the interaction of MCAM and other extracellular components, in particular an MCAM ligand polypeptide, can be tested as follows. Usually a reaction mixture is prepared containing MCAM and the extracellular component (e.g., MCAM ligand such as a laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411) under conditions and for a time allowing for the interaction of the two products. To test the ability of a candidate compound to inhibit the interaction of MCAM and its ligand, the reaction is run in the absence and in the presence of the test compound. In addition, a placebo may be added to a third reaction mixture, to serve as positive control. Since MCAM has been shown to specifically bind its ligand, e.g., a laminin .alpha.4 chain, the ability of the test compound to inhibit the MCAM/MCAM ligand interaction can, for example, be tested by measuring the degree of binding between MCAM and its ligand in the absence and presence of the test compound. If the degree of MCAM binding to its ligand is lower in the absence of the candidate compound than in its presence, the candidate compound is an MCAM antagonist by the definition of the present invention.
[0133] An alternate screening protocol involves the use of a population of cells expressing a laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411, which can be incubated with MCAM, in the presence and absence of a test compound, and binding of MCAM to the cell population monitored, e.g. by fluorescent microscopy (exemplified in Example 5). Other methods of monitoring will be appreciated by those skilled in the art, including fluorescence-activated cell sorting (FACS) and enzyme-linked immunosorbent assay (ELISA). If the binding of MCAM to the cell population in the presence of the test compound is lower than in its absence, the test compound is an MCAM antagonist.
[0134] The MCAM antagonists identified based upon their ability to inhibit the binding of MCAM to its ligand, e.g., a laminin .alpha.4 chain, are drug candidates for the treatment of neuroinflammatory conditions characterized by infiltration of MCAM-expressing cells into the CNS.
[0135] It is emphasized that the screening assays specifically discussed herein are for illustration only. A variety of other assays, which can be selected depending on the type of the antagonist candidates screened (e.g. polypeptides, peptides, non-peptide small organic molecules, aptamers, ribozymes, nucleic acid, etc.) are well known to those skilled in the art and are equally suitable for the purposes of the present invention.
[0136] 6.2 Antibodies
[0137] In one aspect, an MCAM antagonist is an anti-MCAM antibody or an anti-laminin .alpha.4 chain, e.g., .alpha.4 chain of laminin 411, antibody, or an antigen-binding fragment thereof. In some embodiments, an anti-MCAM antibody is a blocking antibody that fully or partially blocks the interaction of MCAM with its ligand, a laminin .alpha.4 chain. In other embodiments, an anti-laminin .alpha.4 chain antibody is a blocking antibody that fully or partially blocks the interaction of a laminin .alpha.4 chain with MCAM. In certain embodiments, the anti-MCAM antibody binds to the extracellular domain of MCAM which interacts with its ligand, a laminin .alpha.4 chain. In a preferred embodiment, the laminin .alpha.4 chain is an .alpha.4 chain of laminin 411.
[0138] In one embodiment, an anti-MCAM antibody specifically or selectively binds to an MCAM fragment comprising or having the amino acid sequence of position 19 to position 129 of SEQ ID NO: 11 (SEQ ID NO:22). In another embodiment, an anti-MCAM antibody specifically or selectively binds to an MCAM fragment comprising or having the amino acid sequence of position 139 to position 242 of SEQ ID NO: 11 (SEQ ID NO:23). In one other embodiment, an anti-MCAM antibody specifically or selectively binds to an MCAM fragment comprising the amino acid sequences of SEQ ID NOS:22 and 23.
[0139] In a preferred embodiment, the antagonist antibody blocks the interaction of an MCAM domain comprising the amino acid sequence of SEQ ID NO:22 and/or SEQ ID NO:23 with a laminin .alpha.4 chain.
[0140] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0141] a) HVR-L1 shown as SEQ ID NO:3;
[0142] b) HVR-L2 shown as SEQ ID NO:4;
[0143] c) HVR-L3 shown as SEQ ID NO:5;
[0144] d) HVR-H1 shown as SEQ ID NO:8;
[0145] e) HVR-H2 shown as SEQ ID NO:9; and/or
[0146] f) HVR-H3 shown as SEQ ID NO:10.
[0147] In another embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO:2 and/or a heavy chain variable domain shown as SEQ ID NO:7.
[0148] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0149] a) HVR-L1 shown as SEQ ID NO: 14;
[0150] b) HVR-L2 shown as SEQ ID NO: 15;
[0151] c) HVR-L3 shown as SEQ ID NO: 16;
[0152] d) HVR-H1 shown as SEQ ID NO:19;
[0153] e) HVR-H2 shown as SEQ ID NO:20; and/or
[0154] f) HVR-H3 shown as SEQ ID NO:21.
[0155] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO: 13 and/or a heavy chain variable domain shown as SEQ ID NO:18.
[0156] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0157] a) HVR-L1 shown as SEQ ID NO:31;
[0158] b) HVR-L2 shown as SEQ ID NO:32;
[0159] c) HVR-L3 shown as SEQ ID NO:33;
[0160] d) HVR-H1 shown as SEQ ID NO:36;
[0161] e) HVR-H2 shown as SEQ ID NO:37; and/or
[0162] f) HVR-H3 shown as SEQ ID NO:38.
[0163] In another embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO:30 and/or a heavy chain variable domain shown as SEQ ID NO:35.
[0164] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0165] a) HVR-L1 shown as SEQ ID NO:41;
[0166] b) HVR-L2 shown as SEQ ID NO:42;
[0167] c) HVR-L3 shown as SEQ ID NO:43;
[0168] d) HVR-H1 shown as SEQ ID NO:46;
[0169] e) HVR-H2 shown as SEQ ID NO:47; and/or
[0170] f) HVR-H3 shown as SEQ ID NO:48.
[0171] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO:40 and/or a heavy chain variable domain shown as SEQ ID NO:45.
[0172] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0173] a) HVR-L1 shown as SEQ ID NO:51;
[0174] b) HVR-L2 shown as SEQ ID NO:52;
[0175] c) HVR-L3 shown as SEQ ID NO:53;
[0176] d) HVR-H1 shown as SEQ ID NO:56;
[0177] e) HVR-H2 shown as SEQ ID NO:57; and/or
[0178] f) HVR-H3 shown as SEQ ID NO:58.
[0179] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO:50 and/or a heavy chain variable domain shown as SEQ ID NO:55.
[0180] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0181] a) HVR-L1 shown as SEQ ID NO:61;
[0182] b) HVR-L2 shown as SEQ ID NO:62;
[0183] c) HVR-L3 shown as SEQ ID NO:63;
[0184] d) HVR-H1 shown as SEQ ID NO:66;
[0185] e) HVR-H2 shown as SEQ ID NO:67; and/or
[0186] f) HVR-H3 shown as SEQ ID NO:68.
[0187] In another embodiment, the anti-MCAM antibody or antibody fragment further comprises a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 128.
[0188] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as SEQ ID NO:60 and/or a heavy chain variable domain shown as SEQ ID NO:65.
[0189] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0190] a) HVR-L1 shown as SEQ ID NO:73;
[0191] b) HVR-L2 shown as SEQ ID NO:74;
[0192] c) HVR-L3 shown as SEQ ID NO:75;
[0193] d) HVR-H1 shown as SEQ ID NO:78;
[0194] e) HVR-H2 shown as SEQ ID NO:79; and/or
[0195] f) HVR-H3 shown as SEQ ID NO:80.
[0196] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as any one of SEQ ID NOS:70, 71, or 72 and/or a heavy chain variable domain shown as SEQ ID NO:77.
[0197] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0198] a) HVR-L1 shown as SEQ ID NO:85;
[0199] b) HVR-L2 shown as SEQ ID NO:86;
[0200] c) HVR-L3 shown as SEQ ID NO:87;
[0201] d) HVR-H1 shown as SEQ ID NO:90;
[0202] e) HVR-H2 shown as SEQ ID NO:91; and/or
[0203] f) HVR-H3 shown as SEQ ID NO:92.
[0204] In another embodiment, the anti-MCAM antibody or antibody fragment further comprises (a) a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 134; (b) a heavy chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 137; (c) a light chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 146; and/or (d) a light chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 144.
[0205] In one other embodiment, the anti-MCAM antibody or antibody fragment comprises a light chain variable domain shown as any one of SEQ ID NOS:83 or 84 and/or a heavy chain variable domain shown as SEQ ID NO:89.
[0206] In other embodiments, the anti-MCAM antibody or antibody fragment comprises the following hypervariable regions (HVRs):
[0207] a) HVR-L1 shown as SEQ ID NO:73;
[0208] b) HVR-L2 shown as SEQ ID NO:74;
[0209] c) HVR-L3 shown as SEQ ID NO:75;
[0210] d) HVR-H1 shown as SEQ ID NO:141;
[0211] e) HVR-H2 shown as SEQ ID NO:79; and/or
[0212] f) HVR-H3 shown as SEQ ID NO:80.
[0213] In another embodiment, the anti-MCAM antibody or antibody fragment further comprises (a) a heavy chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 134; (b) a heavy chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 135; (c) a light chain framework region 1 (FR1) comprising the amino acid sequence of SEQ ID NO: 147; (d) a light chain framework region 2 (FR2) comprising the amino acid sequence of SEQ ID NO: 148; and/or (e) a light chain framework region 3 (FR3) comprising the amino acid sequence of SEQ ID NO: 149.
[0214] In another aspect, the present invention provides MCAM antagonists that bind to substantially the same epitope as, or compete for binding with, an anti-MCAM antibody described herein. In one embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0215] a) HVR-L1 shown as SEQ ID NO:3;
[0216] b) HVR-L2 shown as SEQ ID NO:4;
[0217] c) HVR-L3 shown as SEQ ID NO:5;
[0218] d) HVR-H1 shown as SEQ ID NO:8;
[0219] e) HVR-H2 shown as SEQ ID NO:9; and/or
[0220] f) HVR-H3 shown as SEQ ID NO:10.
[0221] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO:2 and/or a heavy chain variable domain shown as SEQ ID NO:7.
[0222] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0223] a) HVR-L1 shown as SEQ ID NO: 14;
[0224] b) HVR-L2 shown as SEQ ID NO: 15;
[0225] c) HVR-L3 shown as SEQ ID NO: 16;
[0226] d) HVR-H1 shown as SEQ ID NO:19;
[0227] e) HVR-H2 shown as SEQ ID NO:20; and/or
[0228] f) HVR-H3 shown as SEQ ID NO:21.
[0229] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO: 13 and/or a heavy chain variable domain shown as SEQ ID NO:18.
[0230] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0231] a) HVR-L1 shown as SEQ ID NO:31;
[0232] b) HVR-L2 shown as SEQ ID NO:32;
[0233] c) HVR-L3 shown as SEQ ID NO:33;
[0234] d) HVR-H1 shown as SEQ ID NO:36;
[0235] e) HVR-H2 shown as SEQ ID NO:37; and/or
[0236] f) HVR-H3 shown as SEQ ID NO:38.
[0237] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO:30 and/or a heavy chain variable domain shown as SEQ ID NO:35.
[0238] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0239] a) HVR-L1 shown as SEQ ID NO:41;
[0240] b) HVR-L2 shown as SEQ ID NO:42;
[0241] c) HVR-L3 shown as SEQ ID NO:43;
[0242] d) HVR-H1 shown as SEQ ID NO:46;
[0243] e) HVR-H2 shown as SEQ ID NO:47; and/or
[0244] f) HVR-H3 shown as SEQ ID NO:48.
[0245] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO:40 and/or a heavy chain variable domain shown as SEQ ID NO:45.
[0246] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0247] a) HVR-L1 shown as SEQ ID NO:51;
[0248] b) HVR-L2 shown as SEQ ID NO:52;
[0249] c) HVR-L3 shown as SEQ ID NO:53;
[0250] d) HVR-H1 shown as SEQ ID NO:56;
[0251] e) HVR-H2 shown as SEQ ID NO:57; and/or
[0252] f) HVR-H3 shown as SEQ ID NO:58.
[0253] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO:50 and/or a heavy chain variable domain shown as SEQ ID NO:55.
[0254] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0255] a) HVR-L1 shown as SEQ ID NO:61;
[0256] b) HVR-L2 shown as SEQ ID NO:62;
[0257] c) HVR-L3 shown as SEQ ID NO:63;
[0258] d) HVR-H1 shown as SEQ ID NO:66;
[0259] e) HVR-H2 shown as SEQ ID NO:67; and/or
[0260] f) HVR-H3 shown as SEQ ID NO:68.
[0261] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as SEQ ID NO:60 and/or a heavy chain variable domain shown as SEQ ID NO:65.
[0262] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0263] a) HVR-L1 shown as SEQ ID NO:73;
[0264] b) HVR-L2 shown as SEQ ID NO:74;
[0265] c) HVR-L3 shown as SEQ ID NO:75;
[0266] d) HVR-H1 shown as SEQ ID NO:78;
[0267] e) HVR-H2 shown as SEQ ID NO:79; and/or
[0268] f) HVR-H3 shown as SEQ ID NO:80.
[0269] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as any one of SEQ ID NOS:70, 71, or 72 and/or a heavy chain variable domain shown as SEQ ID NO:77.
[0270] In one other embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising the following HVRs:
[0271] a) HVR-L1 shown as SEQ ID NO:85;
[0272] b) HVR-L2 shown as SEQ ID NO:86;
[0273] c) HVR-L3 shown as SEQ ID NO:87;
[0274] d) HVR-H1 shown as SEQ ID NO:90;
[0275] e) HVR-H2 shown as SEQ ID NO:91; and/or
[0276] f) HVR-H3 shown as SEQ ID NO:92.
[0277] In another embodiment, the MCAM antagonist binds to substantially the same epitope as, or competes for binding with, an anti-MCAM antibody comprising a light chain variable domain shown as any one of SEQ ID NOS:83 or 84 and/or a heavy chain variable domain shown as SEQ ID NO:89.
[0278] In an embodiment, the invention provides humanized antibodies. In some embodiments, the humanized antibody comprises a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 94 and SEQ ID NO: 95 (FIG. 25A; h1749). In some embodiments, the humanized antibody comprises a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 98 and SEQ ID NO: 99 (FIG. 25B; h1749). In another embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 94 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 98 and SEQ ID NO:99. In another embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 95 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 98 and SEQ ID NO:99.
[0279] In some embodiments, the humanized antibody comprises a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 102, SEQ ID NO: 103, SEQ ID NO: 104, SEQ ID NO: 105, SEQ ID NO: 106, and SEQ ID NO: 107 (FIG. 26A; h2107). In some embodiments, the humanized antibody comprises a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 110, SEQ ID NO: 111, and SEQ ID NO: 112 (FIG. 26B; h2107). In another embodiment, the humanized antibody comprises a light chain variable domain sequence SEQ ID NO: 111 and a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 102, SEQ ID NO: 103, SEQ ID NO: 104, and SEQ ID NO: 105. In one other embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 102 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 110 and SEQ ID NO: 112. In one embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 106 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 110, SEQ ID NO: 111, and SEQ ID NO: 112. In one embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 107 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 110, SEQ ID NO: 111, and SEQ ID NO: 112.
[0280] In some embodiments, the humanized antibody comprises a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 115, SEQ ID NO: 116, SEQ ID NO: 117, SEQ ID NO: 118, and SEQ ID NO: 119 (FIG. 27A; h2120). In some embodiments, the humanized antibody comprises a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 121, SEQ ID NO: 122, and SEQ ID NO: 123 (FIG. 27B; h2120). In another embodiment, the humanized antibody comprises a light chain variable domain sequence SEQ ID NO: 123 and a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 115, SEQ ID NO: 116, and SEQ ID NO: 117. In one other embodiment, the humanized antibody comprises a light chain variable domain sequence SEQ ID NO: 123 and a heavy chain variable domain sequence selected from the group consisting of SEQ ID NO: 108 and SEQ ID NO: 109. In one embodiment, the humanized antibody comprises a heavy chain variable domain sequence SEQ ID NO: 115 and a light chain variable domain sequence selected from the group consisting of SEQ ID NO: 121 and SEQ ID NO: 122.
[0281] In one embodiment, the invention provides an isolated humanized h1749 anti-MCAM antibody, or antigen binding fragment thereof, comprising two heavy chains (VH1 and VH2) and two light chains (VL1 and VL2). The VH1 heavy chain comprises SEQ ID NO: 94, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 66, 67, and 68, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 155, 125, 126, and 127, respectively. The VH2 heavy chain of h1749 comprises SEQ ID NO: 95, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 66, 67, and 68, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 155, 128, 126, and 127, respectively. The VL1 light chain of h1749 comprises SEQ ID NO: 98, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 61, 62, and 63, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 128, 129, 130, and 131, respectively. The VL2 light chain of h1749 comprises SEQ ID NO: 99, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 61, 62, and 63, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 128, 129, 132, and 131, respectively.
[0282] In another embodiment, the invention provides an isolated humanized h2107 anti-MCAM antibody, or antigen binding fragment thereof, comprising six heavy chains (VH1, VH2, VH3, VH4, VH5, and VH6) and three light chains (VL1, VL2, and VL3). The VH1 heavy chain comprises SEQ ID NO: 102, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 90 or 151, 91, and 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH2 heavy chain of h2107 comprises SEQ ID NO: 103, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 90 or 151, 91, and 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 137, and 136, respectively. The VH3 heavy chain of h2107 comprises SEQ ID NO: 104, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 90 or 151, 91, and 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 138, and 136, respectively. The VH4 heavy chain of h2107 comprises SEQ ID NO: 105, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 139 or 152, 91, and 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH5 heavy chain of h2107 comprises SEQ ID NO: 106, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 140, 91, 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH6 heavy chain of h2107 comprises SEQ ID NO: 107, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 141, 91, and 92, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively.
[0283] The VL1 light chain of h2107 comprises SEQ ID NO: 110, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 85, 86, and 87, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 142, 143, 144, and 145, respectively. The VL2 light chain of h2107 comprises SEQ ID NO: 111, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 85, 86, and 87, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 142, 146, 144, and 145, respectively. The VL3 light chain of h2107 comprises SEQ ID NO: 112, three light chain hypervariable regions (HVR-LC), comprising SEQ ID NOs: 85, 86, and 87, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 147, 143, 144, and 145, respectively.
[0284] In another embodiment, the invention provides an isolated humanized h2120 anti-MCAM antibody, or antigen binding fragment thereof, comprising five heavy chains (VH1, VH2, VH3, VH4, and VH5) and three light chains (VL1, VL2, and VL3). The VH1 heavy chain comprises SEQ ID NO: 115, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 78, 79, and 153, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH2 heavy chain of h2120 comprises SEQ ID NO: 116, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 78, 79, and 153, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 138, and 136, respectively. The VH3 heavy chain of h2120 comprises SEQ ID NO: 117, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 139, 79, and 153, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH4 heavy chain of h2120 comprises SEQ ID NO: 118, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 140, 79, and 153, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively. The VH5 heavy chain of h2120 comprises SEQ ID NO: 119, three heavy chain hypervariable regions (HVR-HC) comprising SEQ ID NOs: 141, 79, and 153, respectively, and four heavy chain frame work regions (FR-HC) comprising SEQ ID NOs: 133, 134, 135, and 136, respectively.
[0285] The VL1 light chain of h2120 comprises SEQ ID NO: 121, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 73, 74, and 75, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 142, 148, 149, and 150, respectively. The VL2 light chain of h2120 comprises SEQ ID NO: 122, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 73, 74, and 75, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 142, 148, 154, and 150, respectively. The VL3 light chain of h2120 comprises SEQ ID NO: 123, three light chain hypervariable regions (HVR-LC) comprising SEQ ID NOs: 73, 74, and 75, respectively, and four light chain frame work regions (FR-LC) comprising SEQ ID NOs: 147, 148, 149, and 150, respectively.
[0286] In some embodiments, the invention provides an antibody that binds to MCAM, wherein the antibody comprises a heavy chain variable domain having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to an amino acid sequence selected from SEQ ID NO: 94, SEQ ID NO: 95, SEQ ID NO: 102, SEQ ID NO: 103, SEQ ID NO: 104; SEQ ID NO: 105, SEQ ID NO: 106, SEQ ID NO: 107, SEQ ID NO: 115, SEQ ID NO: 116, SEQ ID NO: 117, SEQ ID NO: 118, and SEQ ID NO: 119. In some embodiments, the anti-MCAM antibody comprises a light chain variable domain having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to an amino acid sequence selected from the group consisting of SEQ ID NO: 98, SEQ ID NO: 99, SEQ ID NO: 110, SEQ ID NO: 111, SEQ ID NO: 112, SEQ ID NO: 121, SEQ ID NO: 122, and SEQ ID NO: 123. In some embodiment, the antibody comprises a heavy chain variable domain having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to an amino acid sequence selected from SEQ ID NO: 94, SEQ ID NO: 95, SEQ ID NO: 102, SEQ ID NO: 103, SEQ ID NO: 104; SEQ ID NO: 105, SEQ ID NO: 106, SEQ ID NO: 107, SEQ ID NO: 115, SEQ ID NO: 116, SEQ ID NO: 117, SEQ ID NO: 118, and SEQ ID NO: 119, and the antibody further comprises a light chain variable domain having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to an amino acid sequence selected from the group consisting of SEQ ID NO: 98, SEQ ID NO: 99, SEQ ID NO: 110, SEQ ID NO: 111, SEQ ID NO: 112, SEQ ID NO: 121, SEQ ID NO: 122, and SEQ ID NO: 123.
[0287] In some embodiments, the invention provides an antibody that is a variant of any of the above antibodies having one or more amino acid substitutions, deletions, insertions or modifications, and which retains a biological function of the antibody. In some embodiments, the invention provides an antibody that binds to MCAM expressed on the cell surface and inhibits the binding of MCAM to laminin 411. In some embodiments, the anti-MCAM antibody binds to MCAM expressed on the cell surface and inhibits disease progression. In some embodiments, the progression of an autoimmune disease is inhibited. In one embodiment, the progression of multiple sclerosis is inhibited. In some embodiments, the invention provides an antibody that is a variant of any one of the above antibodies having improvements in one or more of a property such as binding affinity, specificity, thermostability, expression level, effector function, glycosylation, reduced immunogenicity, or solubility as compared to the unmodified antibody.
[0288] In other embodiments, the anti-MCAM antibody binds to MCAM expressed on the cell surface and inhibits progression of a metastatic cancer. In one embodiment, the metastatic cancer is selected from the group consisting of prostate cancer, lung cancer, and pancreatic cancer.
[0289] The invention herein includes the production and use of MCAM antagonist antibodies. Exemplary methods for generating antibodies are described in more detail herein. MCAM antibodies can include, but are not limited to, polyclonal, monoclonal, multispecific, human, humanized, primatized, or chimeric antibodies, single chain antibodies (e.g., scFv), Fab fragments, F(ab') fragments, fragments produced by a Fab expression library, anti-idiotypic (anti-Id) antibodies (including, e.g., anti-Id antibodies to antibodies of the present embodiments), and epitope-binding fragments of any of the above. Human antigen-binding antibody fragments include, but are not limited to, Fab, Fab' and F(ab').sub.2, Fd, single-chain Fvs (scFv), single-chain antibodies, disulfide-linked Fvs (sdFv), and fragments comprising either a VL or VH domain. Antigen-binding antibody fragments, including single-chain antibodies, may comprise the variable region(s) alone or in combination with the entirety or a portion of the following: hinge region, CH1, CH2, and CH3 domains. Also included are antigen-binding fragments that can comprise any combination of variable region(s) with a hinge region, CH1, CH2, and CH3 domains. The antibodies may be from any animal origin including birds and mammals. Typically, the antibodies are from human or other primates, murine (e.g., mouse and rat), donkey, sheep, monkey, rabbit, goat, guinea pig, pig, camel, horse, or chicken (or other avian). As used herein, "human" antibodies include antibodies having the amino acid sequence of a human immunoglobulin and include antibodies isolated from human immunoglobulin libraries or from animals transgenic for one or more human immunoglobulins and that do not express endogenous immunoglobulins, as described, for example in, U.S. Pat. No. 5,939,598.
[0290] In another embodiment, the MCAM antibody can be a monoclonal antibody. In yet a further embodiment, the antibody may be chemically modified, e.g., by pegylation. Additionally, other antibodies can be identified using techniques available in the art. For example, antibodies capable of specifically binding to MCAM can be produced using phage display technology. Antibody fragments that selectively bind to MCAM can then be isolated. Exemplary methods for producing such antibodies via phage display are disclosed, for example, in U.S. Pat. No. 6,225,447, for example.
[0291] Monoclonal antibodies can also be produced using the conventional hybridoma methods. These methods have been widely applied to produce hybrid cell lines that secrete high levels of monoclonal antibodies against many specific antigens, and can also be used to produce monoclonal antibodies capable of specifically binding to MCAM. For example, mice (e.g., Balb/c mice) can be immunized with an antigenic MCAM epitope by intraperitoneal injection. After sufficient time has passed to allow for an immune response, the mice are sacrificed, and the spleen cells obtained and fused with myeloma cells, using techniques well known in the art. The resulting fused cells, hybridomas, are then grown in a selective medium, and the surviving cells grown in such medium using limiting dilution conditions. After cloning and recloning, hybridomas can be isolated for secreting antibodies (for example, of the IgG or IgM class or IgG1 subclass) that selectively bind to MCAM. To produce agents specific for human use, the isolated monoclonal can then be used to produce chimeric and humanized antibodies.
[0292] MCAM antagonist antibodies are selected using an antigen derived from a mammalian species. Preferably the antigen is human MCAM or a laminin .alpha.4 chain, e.g., .alpha.4 chain of laminin 411. However, polypeptides from other species such as murine MCAM or laminin .alpha.4 chain can also be used as the target antigen. The antigens from various mammalian species may be isolated from natural sources. In other embodiments, the antigen is produced recombinantly or made using other synthetic methods known in the art. The antibody selected will normally have a sufficiently strong binding affinity for the antigen. For example, the antibody may bind human MCAM or a laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411 with a K.sub.d value of no more than about 5 nM, preferably no more than about 2 nM, and more preferably no more than about 500 pM. Antibody affinities may be determined by a surface plasmon resonance based assay (such as the BIAcore assay as described in Examples); enzyme-linked immunoabsorbent assay (ELISA); and competition assays (e.g. RIA's), for example.
[0293] Also, the antibody may be subject to other biological activity assays, e.g., in order to evaluate its effectiveness as a therapeutic. Such assays are known in the art and depend on the target antigen and intended use for the antibody. Examples include the experimental autoimmune encephalomyelitis (EAE) (as described in Example 7 below), and in vitro and in vivo assays described herein for identifying MCAM antagonists.
[0294] To screen for antibodies which bind to a particular epitope on the antigen of interest, a routine cross-blocking assay such as that described in Antibodies, A Laboratory Manual, Cold Spring Harbor Laboratory, Ed Harlow and David Lane (1988), can be performed. Alternatively, epitope mapping, e.g. as described in Champe et al. (1995) J. Biol. Chem. 270:1388-1394, can be performed to determine whether the antibody binds an epitope of interest.
[0295] In a preferred embodiment, the antagonist antibodies are selected using a unique phage display approach. The approach involves generation of synthetic antibody phage libraries based on single framework template, design of sufficient diversities within variable domains, display of polypeptides having the diversified variable domains, selection of candidate antibodies with high affinity to target antigen, and isolation of the selected antibodies. Details of the phage display methods can be found, for example, in WO03/102157 published Dec. 11, 2003. The antibody generated from phage libraries can be further modified to generate antibody mutants with improved physical, chemical and or biological properties over the parent antibody. Where the assay used is a biological activity assay, the antibody mutant preferably has a biological activity in the assay of choice which is at least about 10 fold better, preferably at least about 20 fold better, more preferably at least about 50 fold better, and sometimes at least about 100 fold or 200 fold better, than the biological activity of the parent antibody in that assay. For example, an anti-MCAM antibody mutant preferably has a binding affinity for MCAM which is at least about 10 fold stronger, preferably at least about 20 fold stronger, more preferably at least about 50 fold stronger, and sometimes at least about 100 fold or 200 fold stronger, than the binding affinity of the parent anti-MCAM antibodies, such as clone 15 or 17 antibodies.
[0296] Chimeric and humanized antibodies can be produced from non-human antibodies, and can have the same or similar binding affinity as the antibody from which they are produced. Exemplary techniques for producing chimeric antibodies include splicing the genes from, e.g., a mouse antibody molecule of appropriate antigen specificity together with genes from a human antibody molecule of appropriate biological activity. See, e.g., Morrison et al., 1984 Proc. Nat'l. Acad. Sci. USA 81: 6851; Neuberger et al., 1984 Nature 312: 604; and Takeda et al., 1985 Nature 314: 452. For example, a nucleic acid encoding a variable (V) region of a mouse monoclonal antibody can be joined to a nucleic acid encoding a human constant (C) region, e.g., IgG1 or IgG4. The resulting antibody is thus a species hybrid, generally with the antigen binding domain from the non-human antibody and the C or effector domain from a human or primate antibody.
[0297] Humanized antibodies are antibodies with variable regions that are primarily from a human antibody (i.e., the acceptor antibody), but which have complementarity determining regions substantially from a non-human antibody (the donor antibody). See, e.g., Queen et al., Proc. Nat'l. Acad. Sci USA 86: 10029-10033 (1989); WO 90/07861, U.S. Pat. Nos. 7,435,802, 6,054,297; 5,693,761; 5,585,089; 5,530,101; and 5,224,539. The constant region or regions of these antibodies are generally also from a human antibody. The human variable domains are typically chosen from human antibodies having sequences displaying a high homology with the desired non-human variable region binding domains. The heavy and light chain variable residues can be derived from the same antibody, or a different human antibody. In addition, the sequences can be chosen as a consensus of several human antibodies, such as described in WO 92/22653.
[0298] A "Primatized.TM. antibody" is a recombinant antibody containing primate variable sequences or antigen binding portions, and human constant domain sequences. See e.g., Newman, Bio/Technology, 1992, 10: 1455-60. Primatization of antibodies results in the generation of antibodies that contain primate (e.g., monkey) variable domains and human constant sequences. See, e.g., U.S. Pat. No. 6,113,898. This technique modifies antibodies such that they are not rejected upon administration in humans because they are antigenic. This technique relies on immunization of cynomolgus monkeys with human antigens or receptors. This technique was developed to create high affinity monoclonal antibodies directed to human cell surface antigens.
[0299] In another aspect, specific amino acids within the human variable region can be selected for substitution based on the predicted conformation and antigen binding properties. This can be determined using techniques such as computer modeling, prediction of the behavior and binding properties of amino acids at certain locations within the variable region, and observation of effects of substitution. For example, when an amino acid differs between a non-human variable region and a human variable region, the human variable region can be altered to reflect the amino acid composition of the non-human variable region. In a specific embodiment, the antibodies used in the chronic dosage regime can be humanized antibodies as disclosed in U.S. Pat. No. 5,840,299. In another embodiment, transgenic mice containing human antibody genes can be immunized with an antigenic MCAM structure and hybridoma technology can be used to generate human antibodies that selectively bind to MCAM.
[0300] Chimeric, human and/or humanized antibodies can be produced by using recombinant expression, e.g., expression in human hybridomas (Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, p. 77 (1985)), in myeloma cells, or in Chinese hamster ovary (CHO) cells. Alternatively, antibody coding sequences can be incorporated into transgenes for introduction into the genome of a transgenic animal and subsequent expression in the milk of the transgenic animal. See, e.g., U.S. Pat. No. 6,197,946. Exemplary suitable transgenes include, but are not limited to, transgenes having a promoter and/or enhancer from a mammary gland specific gene, for example casein or .beta.-lactoglobulin.
[0301] 6.3 Antibody Variants
[0302] In addition to the MCAM antagonist antibodies described herein, it is contemplated that variants of such antibodies can be prepared. Anti-MCAM antagonist antibody variants can be prepared by introducing appropriate nucleotide changes into the encoding DNA, and/or by synthesis of the desired antibody. Those skilled in the art will appreciate that amino acid changes may alter post-translational processes of the anti-MCAM antibody, such as changing the number or position of glycosylation sites.
[0303] Variations in the MCAM antagonist antibodies described herein, can be made, for example, using any of the techniques and guidelines for conservative and non-conservative mutations set forth, for instance, in U.S. Pat. No. 5,364,934. Variations may be a substitution, deletion or insertion of one or more codons encoding the antibody that results in a change in the amino acid sequence as compared with the native sequence antibody. Optionally the variation is by substitution of at least one amino acid with any other amino acid in one or more of the domains of the MCAM antagonist antibody. Guidance in determining which amino acid residue may be inserted, substituted or deleted without adversely affecting the desired activity may be found by comparing the sequence of the MCAM antagonist antibody with that of homologous known protein molecules and minimizing the number of amino acid sequence changes made in regions of high homology. Amino acid substitutions can be the result of replacing one amino acid with another amino acid having similar structural and/or chemical properties, such as the replacement of a leucine with a serine, i.e., conservative amino acid replacements. Insertions or deletions may optionally be in the range of about 1 to 5 amino acids. The variation allowed may be determined by systematically making insertions, deletions or substitutions of amino acids in the sequence and testing the resulting variants for activity exhibited by the full-length or mature native sequence.
[0304] Covalent modifications of anti-MCAM antibodies are included within the scope of this invention. Covalent modifications include reacting targeted amino acid residues of an anti-MCAM antibody with an organic derivatizing agent that is capable of reacting with selected side chains or the N- or C-terminal residues of the anti-MCAM antibody. Other modifications include deamidation of glutaminyl and asparaginyl residues to the corresponding glutamyl and aspartyl residues, respectively, hydroxylation of proline and lysine, phosphorylation of hydroxyl groups of seryl or threonyl residues, methylation of the .alpha.-amino groups of lysine, arginine, and histidine side chains (T.E. Creighton, Proteins: Structure and Molecular Properties, W.H. Freeman & Co., San Francisco, pp. 79-86 (1983)), acetylation of the N-terminal amine, and amidation of any C-terminal carboxyl group.
[0305] In one aspect, the MCAM antagonist antibodies of the present invention comprise one or more deamidation mutations in the amino acid sequence. The deamidation of amino acid residues is a common structural modification in recombinant polypeptides, which can lead to the formation of iso-aspartic acid resulting in decreased stability. Deamidation may be associated with glycine (G)-asparginine (N) sequences, including G-N and N-G sequences. In one embodiment, the antibody comprises a deamidation mutation. In another embodiment, the deamidation mutation is the substitution of an N amino acid residue or a G amino acid residue. In some embodiments, the substitution is selected from the group consisting of N.fwdarw.S, N.fwdarw.A, and G.fwdarw.Q. In one embodiment, the deamidation mutation is located at Kabat residue N32 or G33.
[0306] Other types of covalent modification of the anti-MCAM antibody included within the scope of this invention include altering the native glycosylation pattern of the antibody or polypeptide (Beck et al., Curr. Pharm. Biotechnol. 9: 482-501, 2008; Walsh, Drug Discov. Today 15: 773-780, 2010), and linking the antibody to one of a variety of nonproteinaceous polymers, e.g., polyethylene glycol (PEG), polypropylene glycol, or polyoxyalkylenes, in the manner set forth in U.S. Pat. No. 4,640,835; 4,496,689; 4,301,144; 4,670,417; 4,791,192 or 4,179,337.
[0307] In one aspect, the MCAM antagonist antibodies of the present invention comprise one or more glycosylation mutations in the amino acid sequence. The mutation may remove or restore/introduce a glycosylation site. Generally, the glycosylation mutation is associated with an asparginine (N) residue. In one embodiment, the antibody comprises a glycosylation mutation. In another embodiment, the glycosylation mutation is the introduction of an N amino acid residue. In some embodiments, the introduction is the replacement of an aspartic acid (D) residue with an N residue (D.fwdarw.N). In one embodiment, the mutation is located at Kabat residue 72.
[0308] MCAM antagonist antibody fragments are provided herein. Such fragments may be truncated at the N-terminus or C-terminus, or may lack internal residues, for example, when compared with a full length native antibody. Certain fragments lack amino acid residues that are not essential for a desired biological activity of the MCAM antagonist antibody.
[0309] MCAM antagonist antibody fragments may be prepared by any of a number of conventional techniques. Desired peptide fragments may be chemically synthesized. An alternative approach involves generating antibody or polypeptide fragments by enzymatic digestion, e.g., by treating the protein with an enzyme known to cleave proteins at sites defined by particular amino acid residues, or by digesting the DNA with suitable restriction enzymes and isolating the desired fragment. Yet another suitable technique involves isolating and amplifying a DNA fragment encoding a desired antibody or polypeptide fragment, by polymerase chain reaction (PCR). Oligonucleotides that define the desired termini of the DNA fragment are employed at the 5' and 3' primers in the PCR. Preferably, anti-MCAM antagonist antibody fragments share at least one biological and/or immunological activity with a native MCAM antagonist antibody disclosed herein.
[0310] In particular embodiments, conservative substitutions of interest are shown in Table 1 below under the heading of preferred substitutions. If such substitutions result in a change in biological activity, then more substantial changes, as further described below in reference to amino acid classes, are introduced and the products screened.
[0311] Substantial modifications in function or immunological identity of the MCAM antagonist antibody are accomplished by selecting substitutions that differ significantly in their effect on maintaining (a) the structure of the polypeptide backbone in the area of the substitution, for example, as a sheet or helical conformation, (b) the charge or hydrophobicity of the molecule at the target site, or (c) the bulk of the side chain. Naturally occurring residues are divided into groups based on common side-chain properties:
[0312] (1) hydrophobic: norleucine, met, ala, val, leu, ile;
[0313] (2) neutral hydrophilic: cys, ser, thr;
[0314] (3) acidic: asp, glu;
[0315] (4) basic: asn, gln, his, lys, arg;
[0316] (5) residues that influence chain orientation: gly, pro; and
[0317] (6) aromatic: trp, tyr, phe.
[0318] Non-conservative substitutions will entail exchanging a member of one of these classes for another class. Such substituted residues also may be introduced into the conservative substitution sites or, more preferably, into the remaining (non-conserved) sites.
TABLE-US-00002 TABLE 1 Preferred Original Residue Exemplary Substitutions Substitutions Arg (R) lys; gln; asn lys Asn (N) gln; his; lys; arg gln Asp (D) glu glu Cys (C) ser ser Gln (Q) asn asn Glu (E) asp asp Gly (G) pro; ala ala His (H) asn; gln; lys; arg arg Ile (I) leu; val; met; ala; phe; norleucine leu Leu (L) norleucine; ile; val; met; ala; phe ile Lys (K) arg; gln; asn arg Met (M) leu; phe; ile leu Phe (F) leu; val; ile; ala; tyr leu Pro (P) ala ala Ser (S) thr thr Thr (T) ser ser Trp (W) tyr; phe tyr Tyr (Y) trp; phe; thr; ser phe Val (V) ile; leu; met; phe; ala; norleucine leu
[0319] The variations can be made using methods known in the art such as oligonucleotide-mediated (site-directed) mutagenesis, alanine scanning, and PCR mutagenesis. Site-directed mutagenesis [Carter et al., Nucl. Acids Res., 13:4331 (1986); Zoller et al., Nucl. Acids Res., 10:6487 (1987)], cassette mutagenesis [Wells et al., Gene, 34:315 (1985)], restriction selection mutagenesis [Wells et al., Philos. Trans. R. Soc. London SerA, 317:415 (1986)] or other known techniques can be performed on the cloned DNA to produce the MCAM antagonist antibody variant DNA.
[0320] Scanning amino acid analysis can also be employed to identify one or more amino acids along a contiguous sequence. Among the preferred scanning amino acids are relatively small, neutral amino acids. Such amino acids include alanine, glycine, serine, and cysteine. Alanine is typically a preferred scanning amino acid among this group because it eliminates the side-chain beyond the beta-carbon and is less likely to alter the main-chain conformation of the variant [Cunningham and Wells, Science, 244:1081-1085 (1989)]. Alanine is also typically preferred because it is the most common amino acid. Further, it is frequently found in both buried and exposed positions [Creighton, The Proteins, (W.H. Freeman & Co., N.Y.); Chothia, J. Mol. Biol., 150:1 (1976)]. If alanine substitution does not yield adequate amounts of variant, an isoteric amino acid can be used.
[0321] Any cysteine residue not involved in maintaining the proper conformation of the MCAM antagonist antibody also may be substituted, generally with serine, to improve the oxidative stability of the molecule and prevent aberrant crosslinking. Conversely, cysteine bond(s) may be added to the MCAM antagonist antibody to improve its stability (particularly where the antibody is an antibody fragment such as an Fv fragment).
[0322] A particularly preferred type of substitutional variant involves substituting one or more hypervariable region residues of a parent antibody (e.g., a humanized or human antibody). Generally, the resulting variant(s) selected for further development will have improved biological properties relative to the parent antibody from which they are generated. A convenient way for generating such substitutional variants involves affinity maturation using phage display. Briefly, several hypervariable region sites (e.g., 6-7 sites) are mutated to generate all possible amino substitutions at each site. The antibody variants thus generated are displayed in a monovalent fashion from filamentous phage particles as fusions to the gene III product of M13 packaged within each particle. The phage-displayed variants are then screened for their biological activity (e.g., binding affinity) as herein disclosed. In order to identify candidate hypervariable region sites for modification, alanine scanning mutagenesis can be performed to identify hypervariable region residues contributing significantly to antigen binding. Alternatively, or additionally, it may be beneficial to analyze a crystal structure of the antigen-antibody complex to identify contact points between the antibody and human MCAM or laminin 411 polypeptide. Such contact residues and neighboring residues are candidates for substitution according to the techniques elaborated herein. Once such variants are generated, the panel of variants is subjected to screening as described herein and antibodies with superior properties in one or more relevant assays may be selected for further development.
[0323] Preferred affinity matured antibodies have an affinity which is five times, more preferably 10 times, even more preferably 20 or 30 times greater than the starting antibody (generally murine, humanized or human) from which the matured antibody is prepared.
[0324] Nucleic acid molecules encoding amino acid sequence variants of the MCAM antagonist antibody are prepared by a variety of methods known in the art. These methods include, but are not limited to, isolation from a natural source (in the case of naturally occurring amino acid sequence variants) or preparation by oligonucleotide-mediated (or site-directed) mutagenesis, PCR mutagenesis, and cassette mutagenesis of an earlier prepared variant or a non-variant version of the MCAM antagonist antibody.
[0325] Also included in the invention are antibodies that bind to the same epitope as the antibodies described herein. For example, antibodies of the invention specifically bind to an epitope that includes one or more amino acid residues on human MCAM (Accession No. AAA20922.1/CAA48332). In some embodiments, antibodies of the invention specifically bind MCAM, wherein the antibody binds to an epitope on human MCAM (e.g., Accession No. AAA20922.1/CAA48332).
[0326] Those skilled in the art will recognize that it is possible to determine, without undue experimentation, if a monoclonal antibody (e.g., fully human monoclonal antibody) has the same specificity as a monoclonal antibody of the invention by ascertaining whether the former prevents the latter from binding to MCAM. If the monoclonal antibody being tested competes with the monoclonal antibody of the invention, as shown by a decrease in binding by the monoclonal antibody of the invention, then the two monoclonal antibodies bind to the same, or a closely related, epitope.
[0327] An alternative method for determining whether a monoclonal antibody has the specificity of monoclonal antibody of the invention is to pre-incubate the monoclonal antibody of the invention with MCAM (e.g., an MCAM-Fc molecule exemplified in the Examples) and then add the monoclonal antibody being tested to determine if the monoclonal antibody being tested is inhibited in its ability to bind MCAM. If the monoclonal antibody being tested is inhibited then, in all likelihood, it has the same, or functionally equivalent, epitopic specificity as the monoclonal antibody of the invention.
[0328] Where antibody fragments are used, the smallest inhibitory fragment that specifically binds to the binding domain of the target protein is preferred. For example, based upon the variable-region sequences of an antibody, peptide molecules can be designed that retain the ability to bind the target protein sequence. Such peptides can be synthesized chemically and/or produced by recombinant DNA technology. See, e.g., Marasco et al., Proc. Natl. Acad. Sci. USA, 90: 7889-7893 (1993).
[0329] In a further embodiment, the invention comprises a binding agent that binds to essentially the same epitope as any of the antibodies disclosed herein. In one embodiment, the binding agent is capable of binding to MCAM protein on the surface of cells. In some embodiments, the binding agent inhibits the interaction of MCAM (e.g., cell surface MCAM) with its ligand, a protein comprising a laminin alpha-4 chain. In some embodiments, the binding agent is an antibody or a functional fragment thereof.
[0330] In one embodiment, the invention provides a binding agent capable of binding to MCAM, wherein any one of the antibodies disclosed above displaces the binding agent in a competitive binding assay. In some embodiments, the binding agent is an antibody, or a functional fragment thereof. In another embodiment, the invention provides a binding agent capable of binding to MCAM, wherein the binding agent displaces any one of the antibodies disclosed above in a competitive binding assay. In some embodiments, the binding agent is an antibody, or a functional fragment thereof.
[0331] In other embodiments, the binding agent is an alternative binding agent. These alternative binding agents may include, for example, any of the engineered protein scaffolds known in the art. Such scaffolds include, for example, anticalins, which are based upon the lipocalin scaffold, a protein structure characterized by a rigid beta-barrel that supports four hypervariable loops which form the ligand binding site. Novel binding specificities are engineered by targeted random mutagenesis in the loop regions, in combination with functional display and guided selection (Skerra (2008) FEBS J. 275: 2677-2683). Other suitable scaffolds may include, for example, adnectins, or monobodies, based on the tenth extracellular domain of human fibronectin III (Koide and Koide (2007) Methods Mol. Biol. 352: 95-109); affibodies, based on the Z domain of staphylococcal protein A (Nygren et al. (2008) FEBS J. 275: 2668-2676)); DARPins, based on ankyrin repeat proteins (Stumpp et al. (2008) Drug. Discov. Today 13: 695-701); fynomers, based on the SH3 domain of the human Fyn protein kinase (Grabulovski et al. (2007) J. Biol. Chem. 282: 3196-3204); affitins, based on Sac7d from Sulfolobus acidolarius (Krehenbrink et al. (2008) J. Mol. Biol. 383: 1058-1068); affilins, based on human y-B-crystallin (Ebersbach et al. (2007) J. Mol. Biol. 372: 172-185); avimers, based on the A domains of membrane receptor proteins (Silverman et al. (2005) Biotechnol. 23: 1556-1561); cysteine-rich knottin peptides (Kolmar (2008) FEBS J. 275: 2684-2690); and engineered Kunitz-type inhibitors (Nixon and Wood (2006) Curr. Opin. Drug. Discov. Dev. 9: 261-268). For review, see Gebauer and Skerra (2009) Curr. Opin. Chem. Biol. 13: 245-255. In other embodiments, the binding agent is an anticalin, an adnectin, an affibody, a DARPin, a fynomer, an affitin, an affilin, an avimer, a cysteine-rich knottin peptide, or an engineered Kunitz-type inhibitor.
7. Methods of Use
[0332] The present invention provides MCAM antagonists as therapeutic agents for neuroinflammatory conditions, cancer, and autoimmune diseases. For the prevention, treatment or reduction in the severity of a given disease or condition, the appropriate dosage of a compound of the invention will depend on the type of disease or condition to be treated, as defined above, the severity and course of the disease or condition, whether the agent is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the compound, and the discretion of the attending physician. The compound is suitably administered to the patient at one time or over a series of treatments. Preferably, it is desirable to determine the dose-response curve and the pharmaceutical composition of the invention first in vitro, and then in useful animal models prior to testing in humans.
[0333] In one aspect, the present invention provides a method for inhibiting or blocking the interaction of MCAM expressed on T cells and laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411, comprising treating the T cells with an MCAM antagonist (as described herein), thereby inhibiting the interaction of MCAM with laminin .alpha.4 chain. In one embodiment, the laminin .alpha.4 chain is expressed on the surface of a cell, e.g., an endothelial cell. In a preferred embodiment, the MCAM antagonist is an anti-MCAM antibody. In another embodiment, the T cells are TH17 cells. In one other embodiment, the treatment with an MCAM antagonist is performed in vivo. In yet another embodiment, the treatment is performed in a mammalian subject. In one embodiment, the mammalian subject is a human.
[0334] In another aspect, the present invention provides a method for inhibiting or preventing extravasation of MCAM-expressing T cells into the central nervous system (CNS) comprising treating the T cells with an MCAM antagonist (as described herein), thereby inhibiting or preventing the extravasation of MCAM-expressing T cells into the CNS. In one embodiment, the MCAM antagonist blocks the interaction of MCAM with laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411. In a preferred embodiment, the MCAM antagonist is an anti-MCAM antibody. In one other embodiment, the laminin .alpha.4 chain is expressed on the surface of a cell, e.g., an endothelial cell. In another embodiment, the T cells are TH17 cells. In one other embodiment, the treatment with an MCAM antagonist is performed in vivo. In yet another embodiment, the treatment is performed in a mammalian subject. In one embodiment, the mammalian subject is a human.
[0335] In one other aspect, the present invention provides methods of treatment for a neuroinflammatory condition, a cancerous condition, or an autoimmune disease. In one embodiment, the method comprises administering to a mammalian subject in need a therapeutically effective amount of an MCAM antagonist. In another aspect, the invention provides a method for the delaying or slowing down of the progression of a neuroinflammatory condition, a cancerous condition, or an autoimmune disease. In one embodiment, the method comprises administering to subject diagnosed with the condition or disease, an effective amount of an MCAM antagonist. In another aspect, the invention provides a method for preventing indicia of a neuroinflammatory condition, cancerous condition, or an autoimmune disease. In one embodiment, the method comprises administering an effective amount of an MCAM antagonist to a subject at risk of the condition or disease, wherein the MCAM antagonist is effective against the development of indicia of the condition or disease. In one additional aspect, the present invention provides methods of treatment for a metastatic cancer.
[0336] In one embodiment, the present invention provides an MCAM antagonist for use as a medicament for, or for the treatment of a disease, condition or disorder described herein. In another embodiment, the present invention provides the use of an MCAM antagonist for the manufacture of a medicament for treating a disease, condition or disorder described herein. In one other embodiment, the present invention provides the use of an MCAM antagonist described herein, in the manufacture of a medicament for the treatment of a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS.
[0337] 7.1 Neuroinflammatory Conditions
[0338] In one aspect, the MCAM antagonists provide a preventative or prophylactic effect against the development of, or the progression of, clinical and/or histological and/or biochemical and/or pathological indicia (including both symptoms and signs) of neuroinflammatory conditions in a subject. In one embodiment, the neuroinflammatory condition is characterized by CNS inflammation and/or cell/tissue damage. In one embodiment, the indicia include increased glial activation, increased pro-inflammatory cytokine/chemokine levels (e.g., TNF.alpha., INF.gamma., IL-1.beta.), increased blood-brain-barrier permeability, and/or increased immune cell (e.g., leukocyte) recruitment/invasion to the CNS. In another embodiment, the neuroinflammation is progressive or chronic neuroinflammation associated with chronic activation of cells of the immune system (i.e., autoimmune-associated neuroinflammation). Chronic neuroinflammation conditions include, without limitation, relapsing multiple sclerosis (MS), chronic progressive MS, inactive MS, and Parkinson's disease (PD). In another embodiment, the subject is at risk for a neuroinflammatory condition. In general, a subject at risk will previously have had a neuroinflammatory condition as described herein, or will have a genetic predisposition for neuroinflammatory condition.
[0339] The efficacy of the treatment of neuroinflammatory conditions can be measured by various assessments commonly used in evaluating neuroinflammatory condition. For example, CNS health can be evaluated by testing for MS symptoms including, but not limited to, impaired vision (e.g., blurred or double vision, red-green color distortion, or blindness); muscle weakness in the extremities; impaired coordination and balance; partial or complete paralysis, parenthesis, transitory abnormal sensory feelings (e.g., numbness, prickling, or "pins and needles" sensations); pain; speech impediments; tremors; dizziness; hearing loss; cognitive impairments (e.g., difficulties with concentration, attention, memory, and poor judgment); and depression. MS testing may also include a lumbar puncture (spinal tap) for cerebrospinal fluid (CSF) tests (e.g., CSF oligoclonal banding suggesting inflammation of the CNS); a magnetic resonance imaging (MRI) scan of the head or spine; and a nerve function test (e.g., evoked potential test).
[0340] CNS health may also be evaluated by testing for PD symptoms including, but not limited to, tremor (e.g., trembling in hands, arms, legs, jaw, and face); rigidity or stiffness of the limbs and trunk; bradykinesia or slowness of movement; postural instability or impaired balance and coordination; depression and other emotional changes; difficulty in swallowing, chewing, and speaking; urinary problems or constipation; skin problems; sleep disruptions; and brain scans or other tests to rule out other diseases.
[0341] 7.2 Autoimmune Diseases
[0342] For autoimmune diseases, the term "treatment" refers to both therapeutic treatment and prophylactic or preventative measures for an autoimmune disease, wherein the object is to prevent or slow down (lessen) the targeted pathologic condition or disorder. Those in need of treatment include those already with an autoimmune disease as well as those prone to have an autoimmune disease or those in whom the autoimmune disease is to be prevented.
[0343] In one aspect, the MCAM antagonists provide a preventative or prophylactic effect against the development of, or the progression of, clinical and/or histological and/or biochemical and/or pathological indicia (including both symptoms and signs) of autoimmune disease in a subject. In another embodiment, the subject is at risk for autoimmune disease or an autoimmune disease flare-up. In general, a subject at risk will previously have had autoimmune disease and/or one or more autoimmune disease flare-ups, or will have a genetic predisposition for an autoimmune disease.
[0344] 7.3 Metastatic Cancers
[0345] For metastatic cancer, the term "treatment" refers to both therapeutic treatment and prophylactic or preventative measures for a metastatic cancer, wherein the object is to prevent or slow down (lessen) the targeted pathologic condition or disorder. Those in need of treatment include those already with a metastatic cancer as well as those prone to have a metastatic cancer or those in whom the metastatic cancer is to be prevented.
[0346] In one aspect, the MCAM antagonists provide a preventative or prophylactic effect against the development of, or the progression of, clinical and/or histological and/or biochemical and/or pathological indicia (including both symptoms and signs) of a metastatic cancer in a subject. In one embodiment, the metastatic cancer is selected from the group consisting of prostate cancer, lung cancer, and pancreas cancer.
[0347] 7.4 Combination Therapy
[0348] Antibodies of the invention can be used either alone or in combination with other compositions in a therapy. For instance, an antibody of the invention may be co-administered with at least one additional therapeutic agent. In certain embodiments, an additional therapeutic agent is one or more of the following disease-modifying agents: teriflunomide, interferon beta-1a, interferon beta-1b, glatiramer acetate, fingolimod, and mitoxantrone. In another embodiment, an additional therapeutic agent is an agent which treats an acute exacerbation of a disease. In one embodiment, the additional therapeutic agent for acute exacerbation is one or more corticosteroids. In one other embodiment, the one or more corticosteroids are selected from the group consisting of prednisone, methylprednisolone, and dexamethasone. In another embodiment, the disease is multiple sclerosis and the acute exacerbation is a relapse or attack (e.g., inflammation of the CNS).
[0349] Such combination therapies noted above encompass combined administration (where two or more therapeutic agents are included in the same or separate formulations), and separate administration, in which case, administration of the antibody of the invention can occur prior to, simultaneously, and/or following, administration of the additional therapeutic agent.
8. Pharmaceutical Compositions
[0350] MCAM antagonist antibodies specifically binding MCAM or a laminin .alpha.4 chain, e.g., an .alpha.4 chain of laminin 411, as well as other MCAM antagonist molecules identified by the screening assays disclosed hereinbefore, can be administered for the treatment of various disorders, in particular neuroinflammatory diseases or diseases benefiting from the inhibition of the infiltration of MCAM-expressing cells into the CNS, in the form of pharmaceutical compositions.
[0351] In one aspect, the present invention concerns pharmaceutical compositions comprising an antibody, or antigen binding fragment thereof, as described herein. In one embodiment, the pharmaceutical composition comprises [0352] (i) an isolated anti-MCAM antibody, or antigen binding fragment thereof, that binds to an immunoglobulin domain of MCAM comprising the amino acid sequence shown as SEQ ID NO:22; [0353] (ii) an isolated anti-MCAM antibody, or antigen binding fragment thereof, that binds to an immunoglobulin domain of MCAM comprising the amino acid sequence shown as SEQ ID NO:23; or [0354] (iii) an isolated anti-MCAM antibody, or antigen binding fragment thereof, that binds to a domain of MCAM comprising the amino acid sequences shown as SEQ ID NOS: 22 and 23.
[0355] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0356] (i) an HVR-L1 comprising the amino acid sequence KASKNIDTYLA (SEQ ID NO:3); [0357] (ii) an HVR-L2 comprising the amino acid sequence SGSTL (SEQ ID NO:4); [0358] (iii) an HVR-L3 comprising the amino acid sequence QQHNEYPLT (SEQ ID NO:5); [0359] (iv) an HVR-H1 comprising the amino acid sequence GFTFSNYYMA (SEQ ID NO:8) [0360] (v) an HVR-H2 comprising the amino acid sequence SISFEGNRNHYGDSVK (SEQ ID NO:9); and/or [0361] (vi) an HVR-H3 comprising the amino acid sequence HRGYSTNFYHDVLDAWGQG (SEQ ID NO:10).
[0362] In one other embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0363] (i) an HVR-L1 comprising the amino acid sequence KSSQSLLYSGTQKNYLA (SEQ ID NO: 14); [0364] (ii) an HVR-L2 comprising the amino acid sequence WASTRQS (SEQ ID NO: 15); [0365] (iii) an HVR-L3 comprising the amino acid sequence QQYYDTLTDT (SEQ ID NO:16); [0366] (iv) an HVR-H1 comprising the amino acid sequence GFKFSNYYMS (SEQ ID NO:19); [0367] (v) an HVR-H2 comprising the amino acid sequence SISDGGGDTFCRDLVKG (SEQ ID NO:20); and/or [0368] (vi) an HVR-H3 comprising the amino acid sequence RGAAMGGVMDAWGQG (SEQ ID NO:21).
[0369] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0370] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:31; [0371] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:32; [0372] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:33; [0373] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:36; [0374] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:37; and/or [0375] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:38.
[0376] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0377] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:41; [0378] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:42; [0379] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:43; [0380] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:46; [0381] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:47; and/or [0382] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:48.
[0383] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0384] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:51; [0385] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:52; [0386] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:53; [0387] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:56; [0388] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:57; and/or [0389] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:58.
[0390] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0391] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:61; [0392] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:62; [0393] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:63; [0394] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:66; [0395] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:67; and/or [0396] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:68.
[0397] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0398] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:73; [0399] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:74; [0400] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:75; [0401] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:78; [0402] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:79; and/or [0403] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:80.
[0404] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising the following hypervariable regions (HVRs): [0405] (i) an HVR-L1 comprising the amino acid sequence of SEQ ID NO:85; [0406] (ii) an HVR-L2 comprising the amino acid sequence of SEQ ID NO:86; [0407] (iii) an HVR-L3 comprising the amino acid sequence of SEQ ID NO:87; [0408] (iv) an HVR-H1 comprising the amino acid sequence of SEQ ID NO:90; [0409] (v) an HVR-H2 comprising the amino acid sequence of SEQ ID NO:91; and/or [0410] (vi) an HVR-H3 comprising the amino acid sequence of SEQ ID NO:92.
[0411] In another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, comprising [0412] (a) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO:2 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:7; [0413] (b) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO: 13 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO: 18; [0414] (c) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO:30 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:35; [0415] (d) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO:40 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:45; [0416] (e) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO:50 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:55; [0417] (f) a light chain variable domain comprising the amino acid sequence shown as SEQ ID NO:60 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:65; [0418] (g) a light chain variable domain comprising the amino acid sequence shown as any one of SEQ ID NOS:70, 71, or 72 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:77; or [0419] (h) a light chain variable domain comprising the amino acid sequence shown as any one of SEQ ID NOS:83 or 84 and a heavy chain variable domain comprising the amino acid sequence shown as SEQ ID NO:89.
[0420] In yet another embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, which binds to substantially the same epitope as an antibody described herein. In one other embodiment, the pharmaceutical composition comprises an isolated anti-MCAM antibody, or antigen binding fragment thereof, that competes for binding to human MCAM with an antibody described herein. In additional embodiments, the present invention provides the use of an anti-MCAM antibody, or antigen binding fragment thereof, as described herein, in the manufacture of a medicament for the treatment of a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS.
[0421] The compounds of the invention for prevention or treatment of a neuroinflammatory condition or autoimmune disease are typically administered by intravenous injection. Other methods administration by also be used, which includes but is not limited to, topical, parenteral, subcutaneous, intraperitoneal, intrapulmonary, intranasal, ocular, intraocular, intravitreal, intralesional, intracerobrospinal, intra-articular, intrasynovial, intrathecal, oral, topical, or inhalation administration. Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, or subcutaneous administration. In addition, the compounds described herein are administered to a human subject, in accord with known methods, such as intravenous administration as a bolus or by continuous infusion over a period of time.
[0422] The present invention provides dosages for the MCAM antagonist-based therapeutics. For example, depending on the type and severity of the disease, about 1 .mu.g/kg to 15 mg/kg (e.g. 0.1-20 mg/kg) of polypeptide is an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion. A typical daily dosage might range from about 1 .mu.g/kg to 100 mg/kg or more, depending on the factors mentioned above. For repeated administrations over several days or longer, depending on the condition, the treatment is sustained until a desired suppression of disease symptoms occurs. However, other dosage regimens may be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
[0423] The MCAM antagonist (including MCAM antagonist antibody) compositions herein will be formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The "therapeutically effective amount" of the antagonist to be administered will be governed by such considerations, and is the minimum amount necessary to prevent, ameliorate, or treat a given disease or condition.
[0424] In some embodiments, the composition is used to prevent the occurrence or reoccurrence of the disease or condition disease in the subject. In one embodiment, the present invention can be used for increasing the duration of survival of a human patient susceptible to or diagnosed with the disease or condition disease. Duration of survival is defined as the time from first administration of the drug to death.
[0425] Therapeutic formulations are prepared using standard methods known in the art by mixing the active ingredient having the desired degree of purity with optional physiologically acceptable carriers, excipients or stabilizers (see, e.g., Alfonso R Gennaro (ed), Remington: The Science and Practice of Pharmacy, formerly Remington's Pharmaceutical Sciences 20th ed., Lippincott, Williams & Wilkins, 2003, incorporated herein by reference in its entirety). Acceptable carriers, include saline, or buffers such as phosphate, citrate and other organic acids; antioxidants including ascorbic acid; low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone, amino acids such as glycine, glutamine, asparagines, arginine or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugar alcohols such as mannitol or sorbitol; salt-forming counterions such as sodium; and/or nonionic surfactants such as TWEEN.TM., PLURONIC S.TM., or PEG.
[0426] Optionally, but preferably, the formulation contains a pharmaceutically acceptable salt, preferably sodium chloride, and preferably at about physiological concentrations.
[0427] Optionally, the formulations of the invention can contain a pharmaceutically acceptable preservative. In some embodiments the preservative concentration ranges from 0.1 to 2.0%, typically v/v. Suitable preservatives include those known in the pharmaceutical arts. Benzyl alcohol, phenol, m-cresol, methylparaben, and propylparaben are preferred preservatives. Optionally, the formulations of the invention can include a pharmaceutically acceptable surfactant at a concentration of 0.005 to 0.02%.
[0428] The active ingredients may also be entrapped in microcapsule prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-microcapsule and poly-(methylmethacylate) microcapsule, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions. Such techniques are disclosed in Remington's Pharmaceutical Sciences, supra.
[0429] Sustained-release preparations may be prepared. Suitable examples of sustained-release preparations include semipermeable matrices of solid hydrophobic polymers containing the antibody, which matrices are in the form of shaped articles, e.g., films, or microcapsule. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2-hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides (U.S. Pat. No. 3,773,919), copolymers of L-glutamic acid and .gamma. ethyl-L-glutamate, non-degradable ethylene-vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOT.TM. (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid. While polymers such as ethylene-vinyl acetate and lactic acid-glycolic acid enable release of molecules for over 100 days, certain hydrogels release proteins for shorter time periods. When encapsulated antibodies remain in the body for a long time, they may denature or aggregate as a result of exposure to moisture at 37.degree. C., resulting in a loss of biological activity and possible changes in immunogenicity. Rational strategies can be devised for stabilization depending on the mechanism involved. For example, if the aggregation mechanism is discovered to be intermolecular S--S bond formation through thio-disulfide interchange, stabilization may be achieved by modifying sulfhydryl residues, lyophilizing from acidic solutions, controlling moisture content, using appropriate additives, and developing specific polymer matrix compositions.
9. Articles of Manufacture and Kits
[0430] The instant invention further includes kits comprising the MCAM antagonists of the invention and related materials, such as instructions for use. The instructions for use may contain, for example, instructions for administration of the MCAM antagonists and optionally one or more additional agents. The invention also provides kits for the treatment of a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS. The disorders include, without limitation, neuroinflammatory conditions, such as, for example, multiple sclerosis and Parkinson's disease, and autoimmune disease. The kits of the invention comprise one or more containers of at least one MCAM antagonist, preferably an antibody, in combination with a set of instructions, generally written instructions, relating to the use and dosage of the MCAM antagonist for the treatment of the disorder. The instructions included with the kit generally include information as to dosage, dosing schedule, and route of administration for the treatment of the target disorder, such as a neuroinflammatory condition or an autoimmune disease. The containers of MCAM antagonist(s) may be unit doses, bulk packages (e.g., multi-dose packages), or sub-unit doses.
[0431] In one aspect, the present invention provides a kit comprising an MCAM antagonist as described herein and instructions for use in the treatment of a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS. In one embodiment, the present invention provides a kit for the treatment of a central nervous system (CNS) inflammatory disorder characterized by infiltration of MCAM-expressing cells into the CNS, said kit comprising: (a) a container comprising an MCAM antagonist antibody; and (b) a label or instructions for administering said antibody to treat said CNS inflammatory disorder. Preferably, the CNS inflammatory disorder is a neuroinflammatory condition or an autoimmune disease. In one embodiment, the CNS inflammatory disorder is multiple sclerosis or Parkinson's disease.
[0432] Also provided is an article of manufacture for therapeutic use, comprising a container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, etc. The containers may be formed from a variety of materials such as glass or plastic. The container holds a composition which is effective for treating the condition and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). At least one active agent in the composition is an MCAM antagonist of the invention. The label or package insert indicates that the composition is used for treating the particular condition. The label or package insert will further comprise instructions for administering the antibody composition to the patient. Articles of manufacture and kits comprising combinatorial therapies described herein are also contemplated.
[0433] Package insert refers to instructions customarily included in commercial packages of therapeutic products that contain information about the indications, usage, dosage, administration, contraindications and/or warnings concerning the use of such therapeutic products
[0434] Additionally, the article of manufacture may further comprise a second container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
[0435] All patents, patent applications, and literature references cited in the present specification are hereby incorporated by reference in their entirety.
EXAMPLES
[0436] The following examples are not to be interpreted as limiting, but are exemplary means of using the methods disclosed.
Materials and Methods
Animals and Manipulation of Cells
[0437] SJL mice (Jackson), 8-16 week old, were immunized with PLP 139-151 peptide emulsified in CFA. The commercial kit, EK-0122 (Hooke Laboratories) was used for this immunization experiment. For some experiments, spleens were removed 11 days later and processed into a single cell suspension. For some experiments, splenocytes were processed for in vitro analysis as described below. For EAE studies, mice were injected on days 5, 9, 13, and 17 after PLP immunization with either PBS, isotype control antibody (BioXcell), or anti-MCAM clone 17. Progression of the disease was monitored daily and scored in a blinded fashion by standard techniques. Mice were sacrificed 35 days after PLP immunization, and brains and spinal cords were analyzed for infiltration of immune cells.
[0438] For analysis of MCAM-Fc binding to EAE tissues, 8-16 week old C57BL6 mice were immunized with myelin oligodendrocyte glycoprotein (MOG) 35-55 emulsified in CFA. The commercial kit, EK-0111 (Hooke laboratories) was used for this immunization experiment. The immunized animals were sacrificed at the peak of disease. Brains and spinal cords were snap frozen in OCT (optimal cutting temperature media) and analyzed by fluorescent microscopy as described below.
Flow Cytometry/Marker Staining and Detection/FACS Protocols
[0439] Buffy coats were obtained from healthy human donors (Stanford Blood Center, Palo Alto, Calif.) and CD4 T cells were negatively enriched using RosetteSep (Stem Cell Technologies). Where indicated, CD4+/CD45RO+ memory T cells were further negatively purified using magnetic beads (Miltenyi Biotec). T cells were plated (2.times.10.sup.5 cells/well) in anti-CD3 (5 .mu.g/ml, BD Pharmingen) coated 96 well U bottom plates in RPMI containing 10% heat-inactivated FCS (HyClone Laboratories), penicillin, streptomycin, L-glutamine, anti-IFN.gamma. (5 g/ml; R&D Systems), anti-IL4 (0.5 .mu.g/ml, R&D Systems), and anti-CD28 (2 .mu.g/ml; BD Pharmingen) for five days. Where indicated, TGFP.beta. (2 ng/ml, unless otherwise indicated), IL12, IL1.beta., and/or IL-23 (all at 20 ng/ml) were added. All cytokines were obtained from R&D Systems. Analysis of intracellular cytokines occurred following five hours in the presence of PMA (50 ng/ml) and Ionomycin (500 ng/ml; both from Sigma-Aldrich) and GolgiStop (BD Pharmingen). Surface staining with anti-MCAM (Pharmingen) was followed by fixation, permeabilization, and staining with anti-IL-17A (Ebioscience), IL-22 (R&D Systems), CCL20 (R&D Systems) and/or FOXP3 using a FOXP3 staining kit (Biolegend). In some experiments, unmanipulated whole blood was stained for surface expression with anti-CCR7, anti-CCR6, anti-integrin alpha 4, anti-integrin beta 7, or anti-integrin beta 1 (all from BD Pharmingen).
Antibody Generation/Characterization
[0440] For the generation of antibodies capable of binding to murine MCAM, MCAM-Fc was generated by fusing the extracellular domain of murine MCAM to human IgG and produced in CHO cells using standard techniques. Lou/M rats were immunized with 100 .mu.g of MCAM-Fc protein in CFA (1:1 volume). Rats were boosted two times at two week intervals with MCAM-Fc protein in incomplete Freund's adjuvant (IFA) (1:1 volume). Hybridomas were generated from immunized rats using standard protocols and clones were selected by Clonepix. CHO cells were transfected with the full length murine MCAM gene and selected for stable expression using neomycin and standard techniques. Parental CHO cells (MCAM negative) were fluorescently labeled with carboxyfluorescein succinimidyl ester (CFSE) using standard techniques and mixed at a 1:1 ratio with unlabeled MCAM transfected CHO cells. Hybridoma supernatants were incubated with this mixture of cells for 30 minutes and binding of potential MCAM specific antibodies was detected with a fluorescently labeled anti-rat secondary antibody (Jackson Immuno) by flow cytometry.
[0441] Supernatants from hybridomas that screened positive for MCAM specific antibodies were pre-incubated with fluorescently labeled mouse MCAM-Fc protein (5 .mu.g/mL) for 30 minutes before addition to the laminin .alpha.4 expressing cell line WM2664 and neutralization of binding of the MCAM-Fc protein to the cell line was determined by flow cytometry.
[0442] For the generation of rat antibodies capable of binding to human MCAM, hMCAM-Fc was generated by fusing the extracellular domain of human MCAM to human IgG and produced in CHO cells using standard techniques. Lou/M rats were immunized with 250 .mu.g of hMCAM-Fc protein in CFA (1:1 volume). Rats were boosted two times at two week intervals with hMCAM-Fc protein in incomplete Freund's adjuvant (IFA) (1:1 volume). Hybridomas were generated from immunized rats using standard protocols and clones were selected by Clonepix. CHO cells were transfected with the full length human MCAM gene and selected for stable expression using neomycin and standard techniques. Parental CHO cells (MCAM negative) were fluorescently labeled with carboxyfluorescein succinimidyl ester (CFSE) using standard techniques and mixed at a 1:1 ratio with unlabeled human MCAM transfected CHO cells. Hybridoma supernatants were incubated with this mixture of cells for 30 minutes and binding of potential human MCAM specific antibodies was detected with a fluorescently labeled anti-rat secondary antibody (Jackson Immuno) by flow cytometry.
[0443] For the generation of mouse antibodies capable of binding to human MCAM, hMCAM-Fc was generated by fusing the extracellular domain of human MCAM to human IgG and produced in CHO cells using standard techniques. Balb/c mice were immunized with 50 .mu.g of hMCAM-Fc protein in CFA (1:1 volume). Mice were boosted two times at two week intervals with hMCAM-Fc protein in incomplete Freund's adjuvant (IFA) (1:1 volume). Hybridomas were generated from immunized mice using standard protocols and clones were selected by Clonepix. CHO cells were transfected with the full length human MCAM gene and selected for stable expression using neomycin and standard techniques. Parental CHO cells (MCAM negative) were fluorescently labeled with carboxyfluorescein succinimidyl ester (CFSE) using standard techniques and mixed at a 1:1 ratio with unlabeled human MCAM transfected CHO cells. Hybridoma supernatants were incubated with this mixture of cells for 30 minutes and binding of potential human MCAM specific antibodies was detected with a fluorescently labeled anti-mouse secondary antibody (Jackson Immuno) by flow cytometry.
[0444] Supernatants from hybridomas that screened positive for human MCAM specific antibodies were pre-incubated with fluorescently labeled hMCAM-Fc protein (5 .mu.g/mL) for 30 minutes before addition to the laminin .alpha.4 expressing cell line WM2664 and neutralization of binding of the hMCAM-Fc protein to the cell line was determined by flow cytometry.
Nucleic Acid and Protein Manipulation
[0445] For microarray experiments, human CD4+ T cells were isolated as above, stained for CD161 and CCR6 (both from BD Pharmingen), and sorted into CD4+/CD161-/CCR6- (non-TH17) and CD4+/CD161+/CCR6+(TH17) cells from three independent healthy donors. RNA was isolated from half of the cells from each donor immediately (circulating) and the other half was stimulated with plate bound anti-CD3 and soluble anti-CD28 as above, in the absence of exogenous cytokines for four days (activated) before RNA isolation. RNA was amplified (Nugen) and hybridized on Human U133 Plus 2.0 Array (Affymetrix). All microarray experiments were performed at Expression Analysis, Inc. (Durham, N.C.).
[0446] For determination of CDRs, total RNA was isolated from hybridoma cells using RNAquous-4PCR kit (Ambion), and was used for cDNA synthesis. First and second strand cDNA was synthesized using methods modified from Marathon cDNA amplification (Clontech) with the cDNA adaptor ligated to the 5'-end of the obtained dscDNA. The reverse specific primer was designed based on the specific antibody isotype constant region sequence for both heavy and light chains, and was used along with the adaptor primer in the PCR amplification of both VL and VH fragments using Pfu Ultra DNA polymerase (Stratagene). The amplified PCR product was cloned into pCR-Blunt-TOPO (Invitrogen), and the nucleotide sequence was determined. Identical VL and VH sequences (those of clone 17) were identified from at least 3 out of 5 individual clones for both light and heavy chains.
[0447] For determination of IL-17 concentrations in the supernatant, ELISA was performed using a commercial kit (R&D Systems).
Fluorescence Microscopy/Standard Immunofluorescent Methods
[0448] Tissues from EAE induced mice were snap frozen in OCT and sectioned at 10 .mu.M.
[0449] Sections were fixed in cold acetone and stained with directly conjugated anti-pan-laminin (Novus Biologicals), MCAM-Fc, anti-CD31 (BD Pharmingen), or anti-laminin .alpha.4 (Novus biological). In some experiments, MCAM-Fc was preincubated with anti-MCAM antibodies prior to addition to tissues to ascertain neutralization of MCAM binding to its ligand on tissues.
Mouse Polarization Experiment
[0450] Splenocytes from mice immunized with PLP in CFA for 11 days were isolated and cultured in the presence of PLP (5 .mu.g/mL, Hooke Laboratories). Where indicated, human TGFP.beta. (5 ng/ml) and/or murine IL-23 (20 ng/mL), and murine IL-113 (20 ng/mL) were added for five days in RPMI containing 10% heat-inactivated FCS (HyClone Laboratories), penicillin, streptomycin, L-glutamine, anti-IFN.gamma. (5 .mu.g/ml; R&D Systems), anti-IL4 (0.5 .mu.g/ml, R&D Systems) and .beta.-ME (50 .mu.M). All cytokines were from R&D Systems. Cells were stained with anti-CD4, anti-NK1.1 (both from BD Pharmingen) and anti-MCAM generated as described above.
Example 1. MCAM is a Gene Up-Regulated in IL-17-Producing Human CD4+ T Cells
[0451] To identify novel targetable molecules associated with TH17 cell infiltration of the CNS, human CD4+ T cells from three healthy donors were enriched by magnetic negative selection as described in Materials and Methods above. After the enriched human CD4+ T cells were stained for surface expression of CD161 and CCR6, cells were FACS sorted into two populations: CCR6-/CD161- (representing circulating non-TH17 cells) and CCR6+/CD161+ (representing circulating TH17 cells) as described in Materials and Methods above. RNA was isolated immediately from half of the cells in each population as described in Materials and Methods above. The other half was put into culture with plate-bound anti-CD3 and soluble anti-CD28, without exogenous cytokines, for four days to obtain activated non-TH17 cells and activated TH17 cells, respectively. RNA was similarly isolated from these two types of activated cells. RNA was subject to microarray analysis as described in Materials and Methods above to identify genes specifically expressed in TH17 cells.
[0452] As shown in FIG. 1A, ROR.gamma.t, a known TH17 transcription factor, was up-regulated in both circulating and activated TH17 cells, while IL-17, as an activated TH17 marker, was nearly exclusively expressed in the activated TH17 population. These results indicate that the above procedures of separation and activation were successful. Microarray analysis identified MCAM as an up-regulated gene in both circulating and activated TH17 cells--a profile similar to that of ROR.gamma.t (FIG. 1A).
[0453] MCAM expressing T cells have been described previously as having enriched expression among T cell clones generated from multiple sclerosis patients, and are particularly prominent at sites of inflammation. See, e.g., Brucklacher-Waldert et al., Brian 132: 3329-3341 (2009); see also Pickl et al., J Immunol. 158: 2107-2115 (1997). Here, the MCAM protein was found to be present on the surface of a small population of CD4+ T cells (typically 3-5% of healthy donors). MCAM protein was also found to exist nearly entirely within the CD45RO+ memory population of T cells (FIG. 1B). The human CD4+ T cells were isolated as above, and stimulated for four hours with phorbol myristate acetate (PMA)/Ionomycin. The stimulated CD4+ T cells were analyzed for intracellular IL-17 and surface MCAM levels as described in Materials and Methods above. As shown in FIG. 1C, although the majority of T cells producing IL-17 under these conditions were MCAM negative, MCAM protein was enriched on IL-17- producing cells. Only 2.3% of MCAM negative cells (2.18%/(2.18%+92.62%)) stained positive for IL-17; while 11.9% of MCAM expressing cells (0.62%/(0.62%+4.58%)) were IL-17 positive. Given these data, MCAM is enriched in IL-17-producing human CD4+ T cells.
[0454] Furthermore, when CD4+/CD45RO+ memory T cells were separated into purified populations of MCAM positive and MCAM negative cells and stimulated in vitro with anti-CD3 and anti-CD28, the MCAM positive population produced nearly ten times as much IL-17 (data not shown). The majority of the potential IL-17 production was found to be from the small population of T cells expressing MCAM. In one donor, only the MCAM positive population produced detectable levels of IL-17. Thus, the majority of the potential IL-17 production is from the small population of T cells expressing MCAM.
Example 2. MCAM Expressing T Cells are Effector Memory T Cells Having a Unique Integrin Expression Profile
[0455] The CD45RO+ memory population of human CD4 T cells can be segregated into (1) effector memory cells with tissue tropism, and (2) central memory cells with lymphoid tissue homing based upon expression of CCR7. See, e.g., Sallusto et al., Nature 401: 708-712 (1999).
[0456] To determine which subpopulation includes the MCAM expressing T cells, MCAM expression in T cells was further characterized by staining peripheral human T cells with various markers (CCR6, CCR7, integrin subunits alpha 4, beta 1, and beta 7) as described in Materials and Methods above. MCAM expressing CD4+ T cells were largely CCR7 negative, indicating that most are effector memory T cells, and would be more likely to home to tissues (FIG. 2A). The TH17 enrichment protocol suggested that MCAM expressing T cells obtained would be disproportionately CCR6+. As shown in FIG. 2A, about 64% of MCAM+ cells (2.8%/(2.8%+1.6%)) express CCR6, while only 16.1% of MCAM negative cells (15.4%/(15.4%+80%)) express CCR6 (FIG. 2A). These data suggest that MCAM positive cells would be largely tropic for areas where the ligand for CCR6, CCL20, is high. See, e.g., Liao et al., J. Immunol. 162: 186-194 (1999).
[0457] The integrin expression pattern of MCAM expressing T cells was further characterized. The majority of MCAM expressing T cells are integrin .alpha.4 positive, but are largely integrin .beta.7 negative and .beta.1 positive (FIG. 2B), which is a phenotype associated with the T cells involved in the pathogenesis of EAE (experimental autoimmune encephalomyelitis). See, e.g., Bauer et al., Proc. Nat'l Acad. Sci. USA 106: 1920-1925 (2009).
Example 3. MCAM Expressing T Cells are Expanded by IL1 and Produce the Majority of Both IL-17 and IL-22 Under TH17 Conditions
[0458] MCAM expressing CD4+ T cells, at only 3-5% of cells, is a small minority of the T cell population. It is of interest to determine the conditions under which this population expands and exerts TH17 effector function. For this, human CD4+/CD45RO+ T cells were purified as described in Materials and Methods above and stimulated in vitro with anti-CD3 and anti-CD28 in the presence of a number of cytokine conditions (TGF.beta., IL-12, IL-1.beta., IL-23, and various combinations), and the percentage of MCAM expressing cells, as well as IL-17 expressing cells, was determined by flow cytometry (FIG. 3A). MCAM expression expanded upon stimulation with IL-1.beta. alone (16.4% in the absence of IL-1.beta. vs. 38.1% in the presence of IL-1.beta., FIG. 3B). Furthermore, while TGFP.beta. alone did not expand the MCAM positive population greatly, it functioned synergistically with IL-1.beta., as the combination of both cytokines resulted in more than half of the memory T cell population becoming MCAM positive. Under the same conditions that expanded the population of MCAM expressing cells, the population of IL-17 producing cells was concomitantly increased, with considerable enrichment within the MCAM+ population under all cytokine conditions tested (FIG. 3C). In fact, in the presence of TGFP.beta. and IL-1.beta., more than 80% of the IL-17 producing cells (20.2%/(20.2%+4.4%)) were MCAM positive.
[0459] Additional to IL-17, the known TH17 associated cytokine IL-22 (Liang et al., J. Exp. Med. 203: 2271-2279 (2006)) was also elevated in MCAM expressing T cells. IL-22 receptor is largely expressed on non-immune cells such as epithelial cells and functions in anti-microbial responses as well as tissue remodeling. See, e.g., Dumoutier et al., J. Immunol. 167: 3545-3549 (2001); see also Zenewicz et al., Int. Immunol. 23: 159-163 (2011). Although IL-22 has been shown to be involved in blood brain barrier function, it is not absolutely required for induction or progression of EAE. See, e.g., Kreymborg et al., J. Immunol. 179: 8098-8104 (2007); see also Kebir et al., Nat. Med. 13: 1173-1175 (2007). In a similar fashion to IL-17, a significantly higher percentage of MCAM+ cells expressed IL-22 (FIG. 3D).
[0460] TH17 cells have also been reported to express CCL20. See, e.g., Hirota et al., J. Exp. Med. 204: 2803-2812 (2007). Similar to IL-17 and IL-22, there was a considerably higher population of MCAM expressing T cells that were positive for CCL20 (FIG. 3E), suggesting a possible positive feedback loop in the migration of CCR6+ T cells.
[0461] While the above data are suggestive of a T cell population with a particularly pathogenic phenotype, it was unexpected to observe that MCAM expression was not mutually exclusive with intracellular FOXP3, and in fact, a slightly higher percentage of MCAM+ T cells were FOXP3 positive (FIG. 3F). In the presence of increasing doses of TGFP.beta., the percentage of MCAM+ cells that were FOXP3+ increased, while the percentage of FOXP3 expressing cells in the MCAM-population remained largely unchanged. These results suggest that MCAM expressing cells have the potential to function in an immunoregulatory role in the presence of TGF.beta..
Example 4. MCAM Binds to the ECM at Known Sites of T Cell Infiltration of the CNS, and the MCAM Ligand is Laminin 411
[0462] The function of MCAM has been elucidated in tumor models, showing that MCAM expression confers an adhesive, infiltrative, and ultimately metastatic phenotype to tumor cells. See, e.g., Xie et al., Cancer Res. 57: 2295-2303 (1997). However, the ligand that MCAM binds remains to be identified. Although the above data indicate that MCAM is enriched in TH17 cells, it is unknown whether MCAM is functionally involved in the T cell infiltration of the CNS. It was thus of great interest to determine (1) where MCAM binds, i.e., the identity of the MCAM ligand, (2) whether MCAM is critical to initial infiltration of TH17 cells into the uninflamed brain, and (3) whether the expression of the MCAM ligand is required at the established points of entry to the CNS.
[0463] An MCAM-Fc fusion protein was generated (as described in Materials and Methods above) to detect MCAM binding on healthy mouse tissue, particularly those regions known to be involved in T cell infiltration. As the choroid plexus has been suggested as a route of entry for TH17 cells into the uninflamed brain, healthy choroid plexus tissue was stained with MCAM-Fc and anti-laminin. As shown in FIGS. 4A and 4B, the choroid plexus widely expresses the MCAM ligand, but is negative for MCAM. These results strongly suggest that (1) MCAM unlikely mediates adhesion to the choroid plexus tissue through a homotypic MCAM/MCAM interaction; and (2) there is an additional MCAM ligand with considerably more widespread expression than MCAM, whose expression was limited to vascular endothelium within healthy tissues (FIG. 4C). It was unexpected that MCAM-Fc bound nearly ubiquitously to healthy mouse spinal cord (FIG. 4D) in a pattern that was suggestive of an extracellular matrix (ECM) protein, and specifically laminin. MCAM-Fc and anti-laminin co-localized on healthy mouse spinal cord (FIG. 4E), suggesting that the ligand for MCAM might be a form of laminin. MCAM ligand was confirmed to be in the ECM, as it was exterior to the endothelial cell layer within the vasculature, as determined by CD31 co-staining (FIG. 4F).
[0464] While MCAM co-localized with laminin within healthy mouse tissues, the identity of the MCAM ligand was further confirmed by co-staining EAE tissues with laminin and MCAM-Fc. In regions of lymphocyte infiltration, it has been found that the basement membrane separates into two distinct membranes, the endothelial basement membrane and the parenchymal basement membrane with important distinctions in laminin isoform composition. See, e.g., Sixt et al., J. Cell Biol. 153: 933-945 (2001). When MCAM-Fc was used to stain the MCAM ligand within these regions, it was found that MCAM-Fc stained only the endothelial basement membrane, while pan-laminin stained both the endothelia basement membrane and the parenchymal basement membrane (FIG. 4G). This same expression pattern has been noted for the laminin 411 (laminin 8 (.alpha.4.beta.1.gamma.1)). Co-localization of MCAM-Fc protein and laminin alpha 4 was observed by using a laminin alpha 4 specific antibody (FIG. 4H), suggesting that laminin 411 is a ligand for MCAM. Confirmation that a laminin molecule comprising an alpha 4 chain is the ligand for MCAM, and which may contribute to the unique migratory capability of TH17 cells is described by Flanagan et al., PLoS ONE 7(7): e40443. doi:10.1371/journal.pone.0040443 (2012).
Example 5. Anti-MCAM Antibodies Block Binding of MCAM to Laminin 411
[0465] Monoclonal antibodies against mouse MCAM were generated as described in Materials and Methods above. The specific binding between the monoclonal antibody and MCAM was confirmed by assessing the monoclonal antibody's ability to bind to cells transfected with either mouse or human MCAM. For this, untransfected cells were labeled with carboxyfluorescein succinimidyl ester (CFSE) and mixed with unlabeled MCAM transfected cells. Untransfected cells (in blue) could therefore be differentiated. As shown in FIG. 5A, clones 15 and 17 showed specific binding to mouse MCAM (top, orange) while only clone 17 bound to human MCAM (bottom, orange).
[0466] Next, the monoclonal antibodies were used to test their ability to block the binding of MCAM to its ligand. Murine or human MCAM-Fc protein (5 .mu.g/mL) was pre-incubated with isotype control antibody, clone 15, or clone 17 (10 .mu.g/mL) for 30 minutes in PBS. The mixture was added to healthy spinal cord tissue sections and subsequently characterized by fluorescence microscopy as described in Materials and Methods above.
[0467] As shown in FIG. 5B, both clones 15 and 17 could block binding of the murine MCAM-Fc protein to the tissue, while only clone 17 could block human MCAM-Fc protein binding to the tissue. CDRs of clone 17 have been sequenced and are presented in FIGS. 6A (SEQ ID NO:2, light chain) and 6B (SEQ ID NO:7, heavy chain). Non-denaturing Western blot analysis using clone 17 on individual Fc domains of MCAM confirmed that clone 17 binds specifically to a domain comprising amino acid residues 19 to 129 of MCAM. This binding was confirmed by ForteBio analysis.
[0468] Furthermore, the MCAM monoclonal antibodies were shown to inhibit the interaction between MCAM and its ligand, laminin 411. Parental CHO cells (CHOK1) or CHO cells transfected with mouse MCAM gene were preincubated with CHO culture media (DMEM), recombinant laminin 411 (10 .mu.g/ml), or recombinant laminin 511 (i.e., laminin 10 (.alpha.5.beta.1.gamma.1)) (10 .mu.g/ml) at 37.degree. C. for 45 minutes. Cells were washed, and specific binding of laminin 411, but not laminin 511, to MCAM was detected with a pan-laminin antibody by flow cytometry (FIG. 5C, top right panel). Preincubation of mouse MCAM transfected CHO cells with the anti-MCAM antibody (clone 15 or clone 17, each at 20 .mu.g/ml), prior to laminin incubation, abolished the binding of MCAM to laminin 411 (FIG. 5C, bottom panels).
[0469] The above-presented data suggest that clone 17, which is capable of specifically blocking the binding of human MCAM to its ligand, might be useful to prevent or treat various TH17-mediated diseases by inhibiting MCAM-mediated adhesion of TH17 cells to the vasculature and blocking the migration of TH17 cells into central nervous system.
Example 6. MCAM is not Expressed on Circulating Mouse T Cells, but is Induced Following TH17 Polarization
[0470] Using the antibodies above, peripheral mouse blood was stained to detect MCAM expressing T cells in mice as described in Materials and Methods above. As previously described, mouse T cells lack expression of MCAM, while expression is noted on a population of NK cells (FIG. 7A). The expression of MCAM solely on memory T cells in humans suggests that mice, if living in a clean environment with limited previous T cell activation, would have to be polarized in order to generate a population of MCAM expressing T cells. Considering the link between MCAM and TH17 cells in humans, experiments were conducted to determine whether it was possible to induce a population of MCAM expressing T cells in mice. Myelin proteolipid protein (PLP) specific T cells were generated by immunizing wild type mice with PLP in the presence of complete Freund's adjuvant (CFA) as described in Materials and Methods above. Splenocytes were restimulated in vitro with 5 .mu.g/mL PLP in the presence of the indicated cytokines and analyzed five days later for MCAM expression (FIG. 7B). In the absence of exogenous cytokines, the restimulation did not induce statistically significant MCAM expression on CD4+ cells (as compared to isotype control). In the presence of IL-23, a small population of MCAM expressing CD4+ T cells was detectable. While TGFP.beta. alone did not induce a sizable population of MCAM expressing T cells, the combination of TGFP.beta. and IL-23 synergistically generated MCAM expression among CD4+ T cells. Both of these cytokines have an important role in the polarization and effector function of mouse TH17 cells. Notably, MCAM was expressed on a population of CD4 high T cells which have been described to exclusively contain the pathogenic T cells in EAE. See, e.g., Li et al., J. Neuroimmunol. 192: 57-67 (2007). Thus, unlike humans, mice do not possess a population of circulating CD4+MCAM+ T cells, but polarization under TH17 conditions with TGFP.beta. and IL-23 is sufficient to generate such a population. Mice remain a viable model to study the role of MCAM in the infiltration of CNS by pathogenic T cells.
Example 7. MCAM Blockade by an Anti-MCAM Antibody Inhibits EAE Disease Progression
[0471] EAE is a disease that is generated laboratory animals to produce symptoms similar to those of multiple sclerosis (MS) in humans. EAE is generally produced by injecting animals with different proteins from the central nervous system of other animals, for example, extracts of myelin basic protein and whole spinal cord or brain tissue, or with T cells that specifically react to myelin. EAE is commonly used to follow the course the relapsing or progressive forms of MS. EAE has been served as a suitable animal model to both develop therapeutic agents for MS and study the specific disease processes of MS. See, e.g., Gold et al., Brain 129: 1953-1971 (2006); see also Steinman et al., Ann. Neurol. 60: 12-21 (2006).
[0472] The effects of MCAM blockade on disease progression were further examined in a therapeutic model of EAE, wherein the TH17 polarization occurs in vivo (see Example 6). Mice were immunized with PLP 139-151 peptide as described in Materials and Methods above. Immunized mice were randomized into groups based on clinical scores and day of onset. On the second day following disease onset (EAE symptoms appeared between 12 and 14 days post immunization), mice were treated (N=15 per group) intraperitoneally with either anti-MCAM antibody (clone 15) or isotype control (Bioxcell) at 10 mg/kg body weight, and every day thereafter. Mice were monitored daily and scored for in a blinded manner (FIG. 8A), and body weights were obtained every 2-3 days (FIG. 8B). While MCAM blockade does not appear to affect the severity or duration of the ongoing acute phase of the disease, relapse was delayed and was significantly less severe in mice treated with anti-MCAM antibody (clone 15). These results are consistent with the idea that MCAM may not be essential for infiltration of immune cells during an existing inflammatory process, but may be involved in the subsequent recruitment of antigen experienced pioneer T cells to initiate new inflammatory sites.
Example 8. Domain Binding Test for Murine Anti-MCAM Antibodies
[0473] The following protocol was used: ForteBio Domain Mapping Protocol. ForteBio anti-human IgG Fc biosensors were used to immobilize various mouse MCAMhFc domains including full length mouse MCAMhFc protein on to biosensor surface. These sensors were dipped into either clone 15 or 17 MCAM specific antibody for detection of binding to these domains or full length protein. After loading these samples into a black 96 well plate, the Octet Red was programmed as follows: 60 seconds for baseline #1; 180 seconds for loading various domains; 60 seconds for baseline #2; 180 seconds for association of antibody to domain; and 240 seconds for dissociation of antibody from domain.
Reagents and Supplies Used:
[0474] 1. Mouse MCAMhFc final concentration @ 5 ug/ml [0475] 2. Antibody clone 15 or 17 @ 5 ug/ml [0476] 3. ForteBio anti-human IgG Fc Capture (AHC) biosensors for kinetics experiments, cat#18-5060 [0477] 4. Block 96 well plate from Greiner Bio-one, cat#655209 [0478] 5. ForteBio Octet Red machine [0479] 6. Fresh tissue culture medium, DMEM with 20% FCS, was used as buffer for dilution
[0480] FIG. 10A demonstrates that clone 15 binds specifically to MCAM Fc domains 1 and 2, but not Fc domain 1 alone. FIG. 10B demonstrates that clone 17 binds specifically to either MCAM Fc domains 1 and 2, or Fc domain 1 alone. For FIGS. 10A-B, clones 15 and 17 were tested against the following protein samples (all have human IgG Fc tag): Murine MCAM; Human Fc full length protein; Murine MCAM domain 1 (Ig1); Murine MCAM domain 2 (Ig2); and Murine MCAM domain 1 and 2 (Ig1-2A).
Example 9. MCAM Domains Bind Laminin A4 (.alpha.4) Chain
[0481] The binding affinity of the human laminin-.alpha.4 to human MCAM IgG1-2A was measured by Surface Plasmon Resonance on a Biacore T200 machine. Human Fc-specific F(ab').sub.2 IgG (Jackson Laboratories) was immobilized on a CM5 chip using amine coupling. The four flow cells of the CM5 chips dextran surface are activated by a 7 min injection of freshly prepared 1:1 50 mM NHS: EDC at a flow rate of 5 .mu.l/min. 70 .mu.l IgG solution (pH 4.5) was injected for 3 min to a density of up to 3 000 RU. The coupling is then blocked by a 7 min injection of 1M ethanolamine to deactivate residual reactive sites. Recombinant human Fc-tagged MCAM IgG1-2A in degassed and filtered HBS-P buffer containing 12 mg/ml BSA and 12 mg/ml carboxy-methylated dextran sodium salt was captured by anti-Fc IgG to a capture level 1560 RU. Recombinant human Fc-tagged MCAM IgG1-2A was centrifuged at 14 000 rpm for 5 min at 4.degree. C. before injection for 20 min at a flow rate of 5 l/min over the anti-Fc IgG containing surface. Flow cell 1 was left free of IgG to serve as a control surface. One flow cell was used to capture recombinant human IgG1 Fc (R&D systems) to serve as a negative control. Recombinant human laminin-.alpha.4 (R&D systems) or recombinant human laminin 411 (Biolamina) or recombinant human laminin 511 (Biolamina) (negative control) was diluted in degassed and filtered HBS-P buffer containing 12 mg/ml BSA and 12 mg/ml carboxy-methylated dextran sodium salt to concentrations spanning 5-175 nM and injected (1 min association, 3 min dissociation) over the MCAM IgG1-2A surfaces and control surfaces at a flow rate of 10 .mu.l/min. Buffer injections served as negative control. Data evaluation: Data from the buffer injections and the control surface were subtracted to remove artifacts. The data was fitted globally to a 1:1 interaction model using the Biaevaluation software or Scrubber.
[0482] The laminin .alpha.-4 chain was found to bind specifically to the MCAM Fc domains 1 and 2, but not to Fc domain 1 alone (data not shown). The negative controls included: a lack of binding of laminin 511 to either domain, and a lack of binding of laminin 411 to hIgG1-Fc. Recombinant human laminin-.alpha.4 (R&D systems) binds to human Fc-tagged MCAM IgG1-2A (data not shown) at an affinity of 60 nM, but not to recombinant human IgG1 Fc (R&D systems) (data not shown). Recombinant human laminin 411 (Biolamina) binds to human Fc-tagged MCAM IgG1-2A at an affinity of 66 nM as measured by steady state kinetics (data not shown) but not to recombinant human IgG1 Fc (R&D systems) (data not shown). The negative control, recombinant human laminin 511 (Biolamina) does not bind to human Fc-tagged MCAM IgG1-2A (data not shown).
Example 10. Generation of New Anti-MCAM Monoclonal Antibodies
[0483] Mouse and rat monoclonal antibodies directed against human MCAM protein were generated as described in Materials and Methods above. The specific binding between the monoclonal antibody and human MCAM was confirmed by assessing the monoclonal antibody's ability to bind to cells transfected with human MCAM. For this, untransfected cells were labeled with carboxyfluorescein succinimidyl ester (CFSE) and mixed with unlabeled human MCAM transfected cells. Untransfected cells could, therefore, be differentiated.
[0484] Using these techniques, 823 independent mouse fusions clones were isolated and shown to express an antibody capable of binding to human MCAM. Additionally, 152 independent rat fusions clones were isolated and shown to express an antibody capable of binding to human MCAM.
[0485] Next, the anti-human MCAM monoclonal antibodies were used to test their ability to block the binding of human MCAM to its ligand. Human MCAM-Fc protein (5 .mu.g/mL) was pre-incubated with isotype control antibody, or 10 .mu.g/mL of the test monoclonal antibody for 30 minutes in PBS. The mixture was added to healthy spinal cord tissue sections and subsequently characterized by fluorescence microscopy as described in Materials and Methods above. Furthermore, parental CHO cells (CHOK1) or CHO cells transfected with a human MCAM gene were preincubated with CHO culture media (DMEM), recombinant laminin 411 (10 .mu.g/ml), or recombinant laminin 511 (i.e., laminin 10 ((5.beta.1.gamma.1)) (10 .mu.g/ml) at 37.degree. C. for 45 minutes. Cells were washed, and specific binding of laminin 411, but not laminin 511, to MCAM was detected with a pan-laminin antibody by flow cytometry. Preincubation of human MCAM transfected CHO cells with the anti-MCAM antibody (at 20 .mu.g/ml), prior to laminin incubation, abolished the binding of human MCAM to laminin 411.
[0486] Using this technique, it was shown that 87 of the 823 independent mouse fusion clones and 26 of the 152 independent rat fusion clones described above expressed an antibody that was capable of blocking the interaction between human MCAM protein and its ligand, .alpha.-4 chain of laminin.
Example 11. Further Characterization of New Anti-MCAM Monoclonal Antibodies
[0487] The 87 independent mouse fusion clones and 26 independent rat fusion clones described in Example 10 above as being capable of (i) binding to human MCAM, and (ii) blocking the interaction between human MCAM and the .alpha.-4 chain of laminin were further characterized as follows. First, IC50 quantitation for the ability of the monoclonal antibody to block the binding of human MCAM to the .alpha.-4 chain of laminin was determined as follows. CHO cells expressing human MCAM were incubated with an anti-human MCAM antibody (at various concentrations) for 30 minutes at 4 degrees Celsius. Unbound antibody was then washed away, and the cells were incubated with recombinant human laminin 411 at 20 ug/ml for 45 minutes at 37 degrees Celsius. Unbound laminin was then washed away, and the laminin bound to the surface of the cells was detected with fluorescently labeled anti-laminin antibodies. After washing, the amount of laminin bound to the surface was detected by flow cytometry, and IC50s were calculated based on the mean fluorescent intensity.
[0488] Using the above described assay, six independent anti-human MCAM monoclonal antibody clones were identified as binding to human MCAM and having the greatest ability to block the interaction between human MCAM expressed on the surface of cells and its binding ligand, human laminin 411. These six anti-MCAM monoclonal antibody clones are herein referred to as (i) the mouse anti-human MCAM monoclonal clones 1174.1.3, 1414.1.2, 1415.1.1, and 1749.1.3, and (ii) the rat anti-human MCAM monoclonal antibody clones 2120.4.19 and 2107.4.10. Amino acid and nucleic acid sequences of the heavy and light chains of these antibodies are provided in FIGS. 13-24. More specifically, in the above assay, IC50s for the monoclonal antibody clones 1174.1.3, 1414.1.2, 1415.1.1, 1749.1.3, 2120.4.19, and 2107.4.10 were determined to be 0.469 ug/ml, 0.431 ug/ml, 0.307 ug/ml, 0.545 ug/ml, 0.888 ug/ml, and 0.290 ug/ml, respectively. Moreover, experiments performed to determine the specific binding affinity of each monoclonal antibody demonstrated that each was capable of binding to human MCAM protein with high affinity (data not shown). As such, each of these specific monoclonal antibodies was very capable of binding to human MCAM and inhibiting the interaction of cell-expressed human MCAM with its .alpha.-4 laminin binding ligand. In contrast, two control antibodies, a non-specific human IgG1 antibody and a previously described, fully human anti-MCAM antibody referred to as ABX-MA1 (e.g., see Mills et al., Cancer Res. 62:5106 (2002), and U.S. Pat. Nos. 6,924,360, 7,067,131, and 7,090,844) were both incapable of blocking the binding interaction between human MCAM and its laminin 411 counterpart. As such, the six specific monoclonal antibodies identified above possess the novel ability to both (i) bind with high affinity to human MCAM on the surface of living cells, and (ii) block the interaction of cell expressed human MCAM with a laminin protein comprising an .alpha.-4 laminin polypeptide chain.
Example 12. Domain Binding Analysis for New Anti-MCAM Monoclonal Antibodies
[0489] The technique described in Example 8 above was employed to determine the location of the antigen epitope on the human MCAM protein that is recognized and bound by monoclonal antibody clones 1174.1.3, 1414.1.2, 1415.1.1, 1749.1.3, 2120.4.19, and 2107.4.10. The results from these analyses are as follows.
[0490] Monoclonal antibody clones 1174.1.3, 1414.1.2, 1415.1.1, and 1749.1.3 were all shown to bind to an antigenic epitope found on domain 3 of the human MCAM protein, defined specifically by amino acids 244-321 (SEQ ID NO:24) of the human MCAM protein. These monoclonal antibodies were not capable of binding to human MCAM domain 1 (namely amino acids 19-129, SEQ ID NO:22), domain 2 (namely amino acids 139-242, SEQ ID NO:23), or the combination of domains 1 and 2 (namely, amino acids 19-242). Hence, monoclonal antibody clones 1174.1.3, 1414.1.2, 1415.1.1, and 1749.1.3 define a novel antigenic epitope located within domain 3 of the human MCAM protein.
[0491] Monoclonal antibody clones 2120.4.19, and 2107.4.10 were each shown to bind to an antigenic epitope defined by the combination of human MCAM domains 1 (namely amino acids 19-129, SEQ ID NO:22), and domain 2 (namely amino acids 139-242, SEQ ID NO:23). Neither of these two monoclonal antibodies bound to human MCAM domain 1 by itself. Hence, monoclonal antibody clones 2120.4.19, and 2107.4.10 define a novel antigenic epitope determined by the presence of both human MCAM protein domains 1 and 2.
[0492] In contrast to the above, the previously described fully human anti-MCAM antibody ABX-MA1 binds to a different antigenic epitope than those described above, namely an antigenic epitope that is fully defined and encompassed within human MCAM domain 1 only.
[0493] Given these results, since each of monoclonal antibody clones 1174.1.3, 1414.1.2, 1415.1.1, 1749.1.3, 2120.4.19, and 2107.4.10 are capable of both (i) binding to human MCAM, and (ii) blocking the interaction between human MCAM and an .alpha.-4 laminin containing protein, whereas the ABX-MA1 antibody is capable of only binding to human MCAM, but not blocking the interaction between human MCAM and an .alpha.-4 laminin containing protein, these results demonstrate that human MCAM domain 2, human MCAM domain 3, and the combination thereof play a role in the binding interaction with .alpha.-4 laminin chain. Given this, it is clear that antibodies which bind to human MCAM domain 2, human MCAM domain 3, and/or the combination thereof would find use as agents capable of blocking the interaction between human MCAM and .alpha.-4 laminin and, thereby, find use for inhibiting the various consequences described herein resulting from that interaction. In contrast, antibodies that bind to an antigenic epitope defined solely by human MCAM domain 1 (such as the ABX-MA1 antibody described herein) are not useful for blocking the MCAM/.alpha.-4 laminin interaction and its various downstream biological consequences.
Example 13. Generation of Humanized Anti-MCAM Antibodies
[0494] Various humanized anti-MCAM antibodies were generated according to the following protocol. First, a three-dimensional molecular model of the variable regions was constructed using JN Biosciences' proprietary algorithm. Second, the framework amino acid residues important for the formation of the CDR structure or necessary for the binding to antigen were identified using the molecular model. In parallel, cDNA-derived human VH and VL amino acid sequences with high homology to the VH and VL amino acid sequences, respectively, were selected. Lastly, CDR sequences together with framework amino acid residues important for CDR structure or antigen binding were grafted from VH and VL into the corresponding selected human framework sequences.
[0495] FIGS. 25-27 depict the alignment of various 1749, 2107, and 2120 heavy and light chain sequences. Residue numbering is according to Kabat numbering. Different mutations to the framework (FR) amino acid residues involved in CDR formation and antigen binding were identified depending upon the version of antibody. For example, exemplary mutations of the 1749 antibodies are depicted in FIG. 25A (boxed residue between CDR-H2 and CDR-H3 (A93T) affects CDR contact; the A93T mutation combined with mutations of the boxed residue between CDR-H1 and CDR-H2 (G44R) affects VH/VL interface and CDR conformation) and FIG. 25B (Mutations of the boxed residue between CDR-L2 and CDR-L3 (S63T) affects CDR2 and antigen contact). Exemplary mutations of the 2107 antibodies are depicted in FIG. 26A (boxed residues before CDR-H1 (S30T), between CDR-H1 and CDR-H2 (137V and L48I), and between CDR-H2 and CDR-H3 (K71R) affect CDR contacts; and S30T, I37V, L48I and K71R mutations combined with a mutation of the boxed residue immediately after CDR-H2 (T68S) affect antigen/antibody interactions); and FIG. 26B (boxed residues between CDR-L1 and CDR-L2 (Y36F), and between CDR-L2 and CDR-L3 (V58I) affect CDR contact; and Y36F and V58I mutations combined with (i) an additional mutation of the boxed residue between CDR-L1 and CDR-L2 (Q38L) affect CDR structure; or (ii) an additional mutation before CDR-L1 (T22N) affect antibody antigen interaction). Exemplary mutations of the 2120 antibodies are depicted in FIG. 27A (boxed residues in CDR-H1 (S30T), between CDR-H1 and CDR-H2 (I37V and L48I), and between CDR-H2 and CDR-H3 (K71R) affect CDR contact; and S30T, I37V, L48I, and K71R mutations combined with an additional mutation after CDR-H2 (T68S) affect CDR contact); and FIG. 27B (boxed residues between CDR-L1 and CDR-L2 (L46V and Y49F) and between CDR-L2 and CDR-L3 (V58I) affect CDR contact; boxed residues between CDR-L1 and CDR-L2 (L46V and Y49F) affect CDR contact; and L46V, Y49F, and V58I mutations combined with an additional mutation before CDR-L1 (T22N) affect antibody/antigen interaction).
[0496] Several versions of each chain were designed (standard vs. aggressive or conservative). For those antibodies that contained N-deamidation motifs (NG), mutations to the asparagines or glycine were introduced into the standard version. The various humanized V regions were synthesized with a heterologous signal sequence and cloned into expression vectors containing human CK (VL) or human IgG1 (VH).
[0497] The heavy and light chain plasmids were co-transfected into 293F cells with the FreeStyle.TM. MAX transfection regent (Invitrogen) according to the manufacturer's protocol. The expressed antibody was purified with protein A PhyTip columns (Phynexus) and quantified via OD280.
[0498] The apparent affinities of the humanized antibodies were compared to the parental rodent or chimeric antibody in a competitive ELISA according to the following protocol.
[0499] ELISA plates were coated with recombinant hMCAM-His, and blocked with casein buffer to prevent non-specific binding. Biotinylated rodent or chimeric antibody was added at a subsaturating concentration, in the presence or absence of 3.times. increasing concentrations of unlabeled competitor (humanized antibody, rodent, or chimeric). After washing to remove unbound antibody, streptavidin HRP was added to allow detection of the biotinylated antibody. The ELISA was developed with TMB substrate and the OD450 was measured. The IC50 of the unlabeled competitor was determined using the GraphPad Prism5 software.
[0500] Table 2 summarizes the design of humanized sequences (see also FIGS. 25-27 and 30-32).
TABLE-US-00003 TABLE 2 Donor Framework Mutations 1749 VH1 U96282 IGHV3-7*02 A93T VH2 U96282 IGHV3-7*02 VH1 + G44R VL1 X02990 IGKV4-1*01 None VL2 X02990 IGHKV-1*01 VL1 + S63T 2107 VH1 AF062133 IGHV2-26*01 S30T*, I37V, L48I and K71R VH2 AF062133 IGHV2-26*01 VH1 mutations + T68S VH3 AF062133 IGHV2-26*01 VH1 mutations + D72N VH4 AF062133 IGHV2-26*01 VH1 mutations + N32S VH5 AF062133 IGHV2-26*01 VH1 mutations + N32Q VH6 AF062133 IGHV2-26*01 VH1 mutations + G33A VL1 U86803 IGKV1-27*01 Y36F, V58I VL2 U86803 IGKV1-27*01 VL1 mutations + Q38L VL3 U86803 IGKV1-27*01 VL1 mutations + T22N 2120 VH1 AF062133 IGHV2-26*01 S30T*, I37V, L48I and K71R VH2 AF062133 IGHV2-26*01 VH1 mutations + T68S VH3 AF062133 IGHV2-26*01 VH1 mutations + N32S VH4 AF062133 IGHV2-26*01 VH1 mutations + N32Q VH5 AF062133 IGHV2-26*01 VH1 mutations + G33A VL1 X84343 IGKV1-39*01 L46V, Y49F and V58I VL2 X84343 IGKV1-39*01 L46V, Y49F VL3 X84343 IGKV1-39*01 VL1 + T22N
[0501] The heavy and light chain plasmids were co-transfected into 293F cells with the FreeStyle.TM. MAX transfection regent (Invitrogen) according to the manufacturer's protocol. The expressed antibody was purified with protein A PhyTip columns (Phynexus) and quantified via OD280.
[0502] The apparent affinities of the humanized antibodies were compared to the parental rodent or chimeric antibody in a competitive ELISA according to the following protocol:
[0503] ELISA plates were coated with recombinant hMCAM-His, and blocked with casein buffer to prevent non-specific binding. Biotinylated rodent or chimeric antibody was added at a subsaturating concentration, in the presence or absence of 3.times. increasing concentrations of unlabeled competitor (humanized antibody, rodent, or chimeric). After washing to remove unbound antibody, streptavidin HRP was added to allow detection of the biotinylated antibody. The ELISA was developed with TMB substrate and the OD450 was measured. The IC50 of the unlabeled competitor was determined using the GraphPad Prism5 software.
[0504] The affinities were measured using the ForteBio Octet Red. Anti-human Fc sensors were used to capture the humanized antibodies, and several concentrations of hMCAMHis analyte were used to determine the affinity using a 1:1 fitting model.
[0505] The potencies of the antibodies were measured in the laminin/FACS assay according to the following protocol: recombinant laminin 411 (Biolaminate) was added to hMCAM expressing CHO cells in the presence or absence of varying concentrations of the humanized, rodent, or chimeric antibodies. Following incubation for 30-45 minutes, the cells were washed and anti-laminin conjugated to AF650 (NovusBio) was added to detect the bound laminin. The cells were run on a flow cytometer to measure the laminin binding signal.
[0506] Table 3 provides the constructs used for transfection.
TABLE-US-00004 TABLE 3 Construct Description h1749VH1 Standard h1749VH2 Conservative h1749VL1 Standard h1749VL2 Conservative h2120_VH1 Standard h2120_VH2 Conservative h2120_VH3 Standard + N-S h2120_VH4 Standard + N-Q h2120_VH5 Standard + G-A h2120_VL1 Standard h2120_VL2 Aggressive h2120_VL3 Conservative h2107_VH1 Standard h2107_VH2 Conservative h2107_VH3 Standard + glyc h2107_VH4 Standard + N-S h2107_VH5 Standard + N-Q h2107_VH6 Standard + G-A h2107_VL1 Standard h2107_VL2 Conservative--rec h2107_VL3 Conservative
[0507] Table 4 describes the specific transfection experiments.
TABLE-US-00005 TABLE 4 Transfection--round 1 h1749VH1 + h1749VL1 Standard VH and VL h1749VH2 + h1749VL2 Conservative VH and VL h2120_VH1 + h2120_VL3 Standard VH + conservative VL h2120_VH2 + h2120_VL3 Conservative VH + conservative VL h2120_VH3 + h2120_VL3 N-S deamidate VH + conservative VL h2107_VH1 + h2107_VL2 Standard VH + conservative VL h2107_VH2 + h2107_VL2 Conservative VH + conservative VL h2107_VH3 + h2107_VL2 Standard VH/restore N-gly + conservative VL h2107_VH4 + h2107_VL2 N-S deamidate mut VH + conservative VL Transfection--round 2 h1749VH1 + h1749VL2 Standard VH and conservative VL h1749VH2 + h1749VL1 Conservative VH and standard VL h2120_VH4 + h2120_VL3 N-Q deamidate VH + conservative VL h2120_VH5 + h2120_VL3 G-A deamidate VH + conservative VL h2120_VH1 + h2120_VL1 Standard VH + standard VL h2120_VH1 + h2120_VL2 Standard VH + aggressive VL h2107_VH1 + h2107_VL1 Standard VH and standard VL h2107_VH1 + h2107_VL3 Standard VH and other VL h2107_VH5 + h2107_VL2 N-Q deamidate VH + conservative VL h2107_VH6 + h2107_VL2 G-A deamidate VH + conservative VL h2107_VH5 + h2107_VL3 N-Q deamidate VH + other VL h2107_VH6 + h2107_VL3 G-A deamidate VH + other VL h2107_VH5 + h2107_VL1 N-Q deamidate VH + standard VL h2107_VH6 + h2107_VL1 G-A deamidate VH + standard VL
[0508] Table 5 shows the relative affinities of the humanized antibodies compared to the rodent parent as measured by ForteBio and competitive ELISA, as well as the expression levels for the first round of transfections. FIG. 28A-C compares the blocking of various 1749, 2120, and 2107 antibodies of MCAM binding to laminin from the first round of transfections.
TABLE-US-00006 TABLE 5 Forte ELISA Expt. #1 Expt. #2 Expt. #1 Expt. #2 Fold over Fold over Fold over Fold over Transfection--round 1 rodent rodent rodent rodent Expression level rodent 1749 1.00 1.00 1.00 1.00 h1749VH1 + h1749VL1 2.50 2.41 1.26 1.35 7.2 mg/L h1749VH2 + h1749VL2 0.73 1.09 1.28 1.46 7.2 mg/L chimeric 1749 0.79 0.81 0.97 chimeric 1749 .TM. 1.07 rodent 2120 1.00 1.00 1.00 1.00 h2120_VH1 + h2120_VL3 5.64 6.21 2.23 2.42 22 mg/L h2120_VH2 + h2120_VL3 6.57 6.43 1.93 2.62 16 mg/L h2120_VH3 + h2120_VL3 16.14 3.47 22 mg/L chimeric 2120 0.97 1.72 rodent 2107 1.00 1.00 1.00 1.00 h2107_VH1 + h2107_VL2 2.37 3.40 1.29 1.32 12 mg/L h2107_VH2 + h2107_VL2 2.54 3.58 1.32 1.48 26.7 mg/L h2107_VH3 + h2107_VL2 2.54 1.62 14.6 mg/L h2107_VH4 + h2107_VL2 5.59 11.72 26.7 mg/L chimeric 2107 0.68 1.01 chimeric 2107 .TM. 0.96
[0509] Table 6 shows the measured affinity by ForteBio, competitive ELISA, and functional blocking data (laminin/FACS assay) compared to the rodent parent, as well as the expression levels, from the second round of transfections.
TABLE-US-00007 TABLE 6 Blocking ELISA Expt#1 Expt#2 Forte Forte Fold fold fold Fold over Fold over over over over Expression Transfection--round 2 rodent rodent rodent rodent rodent level h1749VH1 + h1749VL2 2.5 1.0 1.1 1.2 6.9 mg/L h1749VH2 + h1749VL1 1.2 1.0 1.5 1.8 3.2 mg/L h1749VH1 + h1749VL1 2.5 1.0 1.4 1.4 7.2 mg/L h1749VH2 + h1749VL2 0.7 1.1 1.8 1.7 7.2 mg/L chimeric 1749 0.6 1.4 1.4 rodent 1749 1.0 1.0 1.0 1.0 h2120_VH4 + h2120_VL3 17.4 5.0 3.8 5.6 15 mgL h2120_VH5 + h2120_VL3 1.1 1.2 2.4 1.2 1.5 22 mg/L h2120_VH1 + h2120_VL1 8.8 3.1 2.0 3.5 17 mg/L h2120_VH1 + h2120_VL2 10.8 3.1 4.6 12.6 2 mg/L h2120_VH1 + h2120_VL3 5.9 5.8 1.8 1.7 2.8 22 mg/L rodent 2120 1.0 1.0 1.0 1.0 1.0 h2107_VH1 + h2107_VL1 12.9 2.1 2.3 2.1 25 mg/L h2107_VH1 + h2107_VL3 16.3 2.3 2.6 2.0 22 mg/L h2107_VH5 + h2107_VL2 5.9 6.0 1.3 1.6 1.5 19 mg/L h2107_VH6 + h2107_VL2 5.2 5.5 3.5 4.8 2.3 3 mg/L h2107_VH5 + h2107_VL3 33.2 3.4 19 mg/L h2107_VH6 + h2107_VL3 22.1 9.9 3 mg/L h2107_VH5 + h2107_VL1 28.2 3.7 18 mg/L h2107_VH6 + h2107_VL1 36.8 15.3 3 mg/L h2107_VH1 + h2107_VL2 3.0 2.8 1.4 1.9 1.4 12 mg/L h2107_VH2 + h2107_VL2 2.7 2.9 1.3 1.4 1.5 26.7 mg/L rodent 2107 1.0 1.0 1.0 1.0 chimeric 2107 1.0
[0510] Overall, the data demonstrates that the various 1749 humanized antibodies have <2.times. reduction in potency and affinity compared to the rodent. The various 2120 humanized antibodies have a >5.times. reduction in affinity as measured by ForteBio, and most have a >2-3.times. reduction in apparent affinity and potency as measured by the competitive ELISA and laminin blocking assay, with the exception of VH5VL3 (G-A N-deamidation mutant VH/conservative VL), which had a <2.times. reduction in affinity and potency. Many of the 2107 humanized antibodies have a significant loss of affinity and potency, with the exception of VH1VL2, VH2VL2, and VH3VL2 (conservative light chain with standard, conservative, and the restored N-glycosylation site D72N heavy chains); these generally had <3.times. reduction and affinity and potency while retaining the N-deamidation site.
[0511] Certain candidate antibodies were re-expressed and tested for their affinity by ForteBio and their IC.sub.50. The results are provided in Table 7 below and in FIG. 29.
TABLE-US-00008 TABLE 7 Blocking Forte kD IC50 Expression h2120VH5VL3 1.3 0.7 12.7 mg/L h2107VH2VL2 1.4 0.8 20 mg/L h1749VH2VL1 0.67 0.4 3.3 mg/L
Example 14. Analysis of MCAM Blockade by a Humanized Anti-MCAM Antibody and Inhibition of EAE Disease Progression
[0512] The humanized antibodies described herein, e.g., Example 13, are tested for their effect on EAE disease progression according to the protocol provided in Example 7.
Example 15. Use of Humanized Antibodies to Analyze MCAM Expression Following TH17 Polarization
[0513] The humanized antibodies described herein, e.g., Example 13, are used to analyze MCAM expression following TH17 polarization according to the protocol provided in Example 6.
Example 16. DNFB-Induced Skin Inflammation in Mice
[0514] DNFB (2,4-dinitrofluorobezene, Sigma) was diluted in acetone and olive oil (4/1). Abdomen of BALB/c mice (6-10 weeks old) were shaved and sensitized with 25 ul of 0.5% DNFB solution on days 0 and 1 as a model of allergic contact dermatitis and psoriasis. Animals received intraperitoneal treatment of antibodies (Anti-MCAM Clone 15 and Isotype, 10 mg/kg) on days 6 and 7. Right mouse ear was challenge with 5 ul of 0.2% DNFB and the left ear received vehicle (acetone/olive oil) on day 7 (Nakae et al., Immunity. 2002 September; 17(3):375-871). Mice were euthanized on day 8 and ear thickness or swelling was monitored using a micrometer (Mitutoyo, USA). The ear swelling was calculated as [(T1+T2+T3+T4)/4 left ear]-[(T1+T2+T3+T4)/4 right ear], where T represent four different values of ear thickness. FIG. 30 shows the antibody significantly inhibited inflammation.
Example 17. Inhibition of Tumor Growth Nude SCID Mice
[0515] 5.times.10.sup.5 human MCAM expressing melanoma cells (WM2664) were injected subcutaneously into male nude severe combined immunodeficiency (SCID) mice. Mice were treated weekly with 1 mg of total antibody (0.5 mg of each antibody in the combination group) weekly beginning on day 4 post tumor implantation. The antibodies used were anti-mouse MCAM clone 15 or anti-human MCAM clone 2120.4.19 or a combination thereof. Tumors were measured in a blinded fashion with calipers 2.times. per week and tumor volume was determined by standard formula (Volume=(width)2.times.length/2) (FIG. 31A). On day 40 post tumor implantation, mice were euthanized, and tumors were removed intact, and weighed (FIG. 31B). Each antibody inhibited tumor growth and inhibition was most notable in the combination treatment. It is believed the combination treatment is more efficient in mice containing human cancer cells. The human cancer cells express human MCAM and human laminin, and the mice express mouse MCAM and mouse laminin. Because mouse and human MCAM/laminin bind to each other, antibodies with specificity for human and mouse MCAM act synergistically to inhibit tumor growth. In a human, where all MCAM and laminin are human, human anti-MCAM clone 2120.4.19 (or similar antibody) is expected to be similarly effective as the combination treatment in mice.
Sequence CWU 1
1
1551428DNAArtificial SequenceSynthetic PolynucleotideCDS(1)...(426)
1atg agg gtc cag att cag ttt ctg ggg ctc ctt ctg ctc tgg aca tca
48Met Arg Val Gln Ile Gln Phe Leu Gly Leu Leu Leu Leu Trp Thr Ser 1
5 10 15gtt gtc cag tgt gat gtc cag atg acc cag tct cca tct tat ctt
gct 96Val Val Gln Cys Asp Val Gln Met Thr Gln Ser Pro Ser Tyr Leu
Ala 20 25 30acg tct cct gga gag agt gtt tcc atc agt tgc aag gca agt
aaa aac 144Thr Ser Pro Gly Glu Ser Val Ser Ile Ser Cys Lys Ala Ser
Lys Asn 35 40 45att gac aca tac tta gcc tgg tat cag gag aaa cct ggg
aaa acg aat 192Ile Asp Thr Tyr Leu Ala Trp Tyr Gln Glu Lys Pro Gly
Lys Thr Asn 50 55 60aag ctt ctt atc tac tct ggg tca act ttg caa tct
gga act cca tcg 240Lys Leu Leu Ile Tyr Ser Gly Ser Thr Leu Gln Ser
Gly Thr Pro Ser 65 70 75 80aga ttc agt ggc agt gga tct ggt aca gat
ttc acg ctc acc atc aga 288Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp
Phe Thr Leu Thr Ile Arg 85 90 95aac ctg gag tct gaa gat ttt gca gtc
tac tac tgt caa cag cat aat 336Asn Leu Glu Ser Glu Asp Phe Ala Val
Tyr Tyr Cys Gln Gln His Asn 100 105 110gaa tac ccg ctc acg ttc ggt
tct ggg acc aag ctg gag atc aaa cgg 384Glu Tyr Pro Leu Thr Phe Gly
Ser Gly Thr Lys Leu Glu Ile Lys Arg 115 120 125gct gat gct gca cca
act gta tcc atc ttc cca cca tcc tcg 426Ala Asp Ala Ala Pro Thr Val
Ser Ile Phe Pro Pro Ser Ser 130 135 140ga 4282142PRTArtificial
SequenceSynthetic Construct 2Met Arg Val Gln Ile Gln Phe Leu Gly
Leu Leu Leu Leu Trp Thr Ser1 5 10 15Val Val Gln Cys Asp Val Gln Met
Thr Gln Ser Pro Ser Tyr Leu Ala 20 25 30Thr Ser Pro Gly Glu Ser Val
Ser Ile Ser Cys Lys Ala Ser Lys Asn 35 40 45Ile Asp Thr Tyr Leu Ala
Trp Tyr Gln Glu Lys Pro Gly Lys Thr Asn 50 55 60Lys Leu Leu Ile Tyr
Ser Gly Ser Thr Leu Gln Ser Gly Thr Pro Ser65 70 75 80Arg Phe Ser
Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Arg 85 90 95Asn Leu
Glu Ser Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln His Asn 100 105
110Glu Tyr Pro Leu Thr Phe Gly Ser Gly Thr Lys Leu Glu Ile Lys Arg
115 120 125Ala Asp Ala Ala Pro Thr Val Ser Ile Phe Pro Pro Ser Ser
130 135 140311PRTArtificial SequenceSynthetic Peptide 3Lys Ala Ser
Lys Asn Ile Asp Thr Tyr Leu Ala1 5 1045PRTArtificial
SequenceSynthetic Peptide 4Ser Gly Ser Thr Leu1 559PRTArtificial
SequenceSynthetic Peptide 5Gln Gln His Asn Glu Tyr Pro Leu Thr1
56483DNAArtificial SequenceSynthetic PolynucleotideCDS(1)...(483)
6atg gac acc agg ctc tgc ttg gtt ttc ctt gtc ctt ttc ata aaa ggt
48Met Asp Thr Arg Leu Cys Leu Val Phe Leu Val Leu Phe Ile Lys Gly 1
5 10 15gtc cag tgt gag gtg cag ctg gtg gag tct ggt gga ggc tta gtg
cag 96Val Gln Cys Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val
Gln 20 25 30cct gga agg tcc ctg aaa ctc tcc tgt gca gcc tca gga ttc
act ttc 144Pro Gly Arg Ser Leu Lys Leu Ser Cys Ala Ala Ser Gly Phe
Thr Phe 35 40 45agt aac tat tac atg gcc tgg gtc cgc cag gct cca acg
aag ggt ctg 192Ser Asn Tyr Tyr Met Ala Trp Val Arg Gln Ala Pro Thr
Lys Gly Leu 50 55 60gag tgg gtc gca tcc att agt ttt gag ggt aat aga
aat cac tat gga 240Glu Trp Val Ala Ser Ile Ser Phe Glu Gly Asn Arg
Asn His Tyr Gly 65 70 75 80gac tcc gtg aag ggc cga atc act atc tcc
aga gat aat gca aaa agc 288Asp Ser Val Lys Gly Arg Ile Thr Ile Ser
Arg Asp Asn Ala Lys Ser 85 90 95acc cta tac ctg caa atg acc agt ctg
agg cct gag gac acg gcc act 336Thr Leu Tyr Leu Gln Met Thr Ser Leu
Arg Pro Glu Asp Thr Ala Thr 100 105 110tat tat tgt gca aga cat cgg
ggg tat agt acg aat ttt tat cac gac 384Tyr Tyr Cys Ala Arg His Arg
Gly Tyr Ser Thr Asn Phe Tyr His Asp 115 120 125gtt ttg gat gcc tgg
ggt caa gga gct tta gtc act gtc tcc tca gct 432Val Leu Asp Ala Trp
Gly Gln Gly Ala Leu Val Thr Val Ser Ser Ala 130 135 140gaa aca aca
gcc cca tct gtc tat cca ctg gct cct gga act gct ctc 480Glu Thr Thr
Ala Pro Ser Val Tyr Pro Leu Ala Pro Gly Thr Ala Leu145 150 155
160aaa 483Lys7161PRTArtificial SequenceSynthetic Construct 7Met Asp
Thr Arg Leu Cys Leu Val Phe Leu Val Leu Phe Ile Lys Gly1 5 10 15Val
Gln Cys Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln 20 25
30Pro Gly Arg Ser Leu Lys Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe
35 40 45Ser Asn Tyr Tyr Met Ala Trp Val Arg Gln Ala Pro Thr Lys Gly
Leu 50 55 60Glu Trp Val Ala Ser Ile Ser Phe Glu Gly Asn Arg Asn His
Tyr Gly65 70 75 80Asp Ser Val Lys Gly Arg Ile Thr Ile Ser Arg Asp
Asn Ala Lys Ser 85 90 95Thr Leu Tyr Leu Gln Met Thr Ser Leu Arg Pro
Glu Asp Thr Ala Thr 100 105 110Tyr Tyr Cys Ala Arg His Arg Gly Tyr
Ser Thr Asn Phe Tyr His Asp 115 120 125Val Leu Asp Ala Trp Gly Gln
Gly Ala Leu Val Thr Val Ser Ser Ala 130 135 140Glu Thr Thr Ala Pro
Ser Val Tyr Pro Leu Ala Pro Gly Thr Ala Leu145 150 155
160Lys810PRTArtificial SequenceSynthetic Peptide 8Gly Phe Thr Phe
Ser Asn Tyr Tyr Met Ala1 5 10916PRTArtificial SequenceSynthetic
Peptide 9Ser Ile Ser Phe Glu Gly Asn Arg Asn His Tyr Gly Asp Ser
Val Lys1 5 10 151019PRTArtificial SequenceSynthetic Peptide 10His
Arg Gly Tyr Ser Thr Asn Phe Tyr His Asp Val Leu Asp Ala Trp1 5 10
15Gly Gln Gly11646PRTHomo sapiens 11Met Gly Leu Pro Arg Leu Val Cys
Ala Phe Leu Leu Ala Ala Cys Cys1 5 10 15Cys Cys Pro Arg Val Ala Gly
Val Pro Gly Glu Ala Glu Gln Pro Ala 20 25 30Pro Glu Leu Val Glu Val
Glu Val Gly Ser Thr Ala Leu Leu Lys Cys 35 40 45Gly Leu Ser Gln Ser
Gln Gly Asn Leu Ser His Val Asp Trp Phe Ser 50 55 60Val His Lys Glu
Lys Arg Thr Leu Ile Phe Arg Val Arg Gln Gly Gln65 70 75 80Gly Gln
Ser Glu Pro Gly Glu Tyr Glu Gln Arg Leu Ser Leu Gln Asp 85 90 95Arg
Gly Ala Thr Leu Ala Leu Thr Gln Val Thr Pro Gln Asp Glu Arg 100 105
110Ile Phe Leu Cys Gln Gly Lys Arg Pro Arg Ser Gln Glu Tyr Arg Ile
115 120 125Gln Leu Arg Val Tyr Lys Ala Pro Glu Glu Pro Asn Ile Gln
Val Asn 130 135 140Pro Leu Gly Ile Pro Val Asn Ser Lys Glu Pro Glu
Glu Val Ala Thr145 150 155 160Cys Val Gly Arg Asn Gly Tyr Pro Ile
Pro Gln Val Ile Trp Tyr Lys 165 170 175Asn Gly Arg Pro Leu Lys Glu
Glu Lys Asn Arg Val His Ile Gln Ser 180 185 190Ser Gln Thr Val Glu
Ser Ser Gly Leu Tyr Thr Leu Gln Ser Ile Leu 195 200 205Lys Ala Gln
Leu Val Lys Glu Asp Lys Asp Ala Gln Phe Tyr Cys Glu 210 215 220Leu
Asn Tyr Arg Leu Pro Ser Gly Asn His Met Lys Glu Ser Arg Glu225 230
235 240Val Thr Val Pro Val Phe Tyr Pro Thr Glu Lys Val Trp Leu Glu
Val 245 250 255Glu Pro Val Gly Met Leu Lys Glu Gly Asp Arg Val Glu
Ile Arg Cys 260 265 270Leu Ala Asp Gly Asn Pro Pro Pro His Phe Ser
Ile Ser Lys Gln Asn 275 280 285Pro Ser Thr Arg Glu Ala Glu Glu Glu
Thr Thr Asn Asp Asn Gly Val 290 295 300Leu Val Leu Glu Pro Ala Arg
Lys Glu His Ser Gly Arg Tyr Glu Cys305 310 315 320Gln Ala Trp Asn
Leu Asp Thr Met Ile Ser Leu Leu Ser Glu Pro Gln 325 330 335Glu Leu
Leu Val Asn Tyr Val Ser Asp Val Arg Val Ser Pro Ala Ala 340 345
350Pro Glu Arg Gln Glu Gly Ser Ser Leu Thr Leu Thr Cys Glu Ala Glu
355 360 365Ser Ser Gln Asp Leu Glu Phe Gln Trp Leu Arg Glu Glu Thr
Asp Gln 370 375 380Val Leu Glu Arg Gly Pro Val Leu Gln Leu His Asp
Leu Lys Arg Glu385 390 395 400Ala Gly Gly Gly Tyr Arg Cys Val Ala
Ser Val Pro Ser Ile Pro Gly 405 410 415Leu Asn Arg Thr Gln Leu Val
Lys Leu Ala Ile Phe Gly Pro Pro Trp 420 425 430Met Ala Phe Lys Glu
Arg Lys Val Trp Val Lys Glu Asn Met Val Leu 435 440 445Asn Leu Ser
Cys Glu Ala Ser Gly His Pro Arg Pro Thr Ile Ser Trp 450 455 460Asn
Val Asn Gly Thr Ala Ser Glu Gln Asp Gln Asp Pro Gln Arg Val465 470
475 480Leu Ser Thr Leu Asn Val Leu Val Thr Pro Glu Leu Leu Glu Thr
Gly 485 490 495Val Glu Cys Thr Ala Ser Asn Asp Leu Gly Lys Asn Thr
Ser Ile Leu 500 505 510Phe Leu Glu Leu Val Asn Leu Thr Thr Leu Thr
Pro Asp Ser Asn Thr 515 520 525Thr Thr Gly Leu Ser Thr Ser Thr Ala
Ser Pro His Thr Arg Ala Asn 530 535 540Ser Thr Ser Thr Glu Arg Lys
Leu Pro Glu Pro Glu Ser Arg Gly Val545 550 555 560Val Ile Val Ala
Val Ile Val Cys Ile Leu Val Leu Ala Val Leu Gly 565 570 575Ala Val
Leu Tyr Phe Leu Tyr Lys Lys Gly Lys Leu Pro Cys Arg Arg 580 585
590Ser Gly Lys Gln Glu Ile Thr Leu Pro Pro Ser Arg Lys Thr Glu Leu
595 600 605Val Val Glu Val Lys Ser Asp Lys Leu Pro Glu Glu Met Gly
Leu Leu 610 615 620Gln Gly Ser Ser Gly Asp Lys Arg Ala Pro Gly Asp
Gln Gly Glu Lys625 630 635 640Tyr Ile Asp Leu Arg His
64512474DNAArtificial SequenceSynthetic
PolynucleotideCDS(1)...(474) 12atg gaa tca cag acc cag gtc ctc atg
tcc ctg ctg ctc tgg att tct 48Met Glu Ser Gln Thr Gln Val Leu Met
Ser Leu Leu Leu Trp Ile Ser 1 5 10 15ggt acc tgt ggg gac att gtg
atg acc cag tct cca tcc tct ctg gct 96Gly Thr Cys Gly Asp Ile Val
Met Thr Gln Ser Pro Ser Ser Leu Ala 20 25 30gtg tca gct ggg gag acg
gtc tct ata cac tgc aag tcc agt cag agt 144Val Ser Ala Gly Glu Thr
Val Ser Ile His Cys Lys Ser Ser Gln Ser 35 40 45ctt tta tac agt gga
acc caa aag aac tac ttg gcc tgg ttc cag cag 192Leu Leu Tyr Ser Gly
Thr Gln Lys Asn Tyr Leu Ala Trp Phe Gln Gln 50 55 60aaa cca gga cag
tct cct aaa ctg ctg atc ttc tgg gca tct act agg 240Lys Pro Gly Gln
Ser Pro Lys Leu Leu Ile Phe Trp Ala Ser Thr Arg 65 70 75 80cag tct
ggt gtc cct gat cgc ttc ata ggc cgt gga tct ggg aca gac 288Gln Ser
Gly Val Pro Asp Arg Phe Ile Gly Arg Gly Ser Gly Thr Asp 85 90 95ttc
act ctg acc atc agc ggt gtg cag gca gaa gat ctg gca att tat 336Phe
Thr Leu Thr Ile Ser Gly Val Gln Ala Glu Asp Leu Ala Ile Tyr 100 105
110tac tgt caa caa tat tat gat act ctc acg gac acg ttt gga gcg ggg
384Tyr Cys Gln Gln Tyr Tyr Asp Thr Leu Thr Asp Thr Phe Gly Ala Gly
115 120 125acc aag ctg gaa ctg aaa cgg gct gat gct gca cca act gta
tct atc 432Thr Lys Leu Glu Leu Lys Arg Ala Asp Ala Ala Pro Thr Val
Ser Ile 130 135 140ttc cca cca tcc acg gaa cag tta gca act gga ggt
gcc tca 474Phe Pro Pro Ser Thr Glu Gln Leu Ala Thr Gly Gly Ala
Ser145 150 15513158PRTArtificial SequenceSynthetic Construct 13Met
Glu Ser Gln Thr Gln Val Leu Met Ser Leu Leu Leu Trp Ile Ser1 5 10
15Gly Thr Cys Gly Asp Ile Val Met Thr Gln Ser Pro Ser Ser Leu Ala
20 25 30Val Ser Ala Gly Glu Thr Val Ser Ile His Cys Lys Ser Ser Gln
Ser 35 40 45Leu Leu Tyr Ser Gly Thr Gln Lys Asn Tyr Leu Ala Trp Phe
Gln Gln 50 55 60Lys Pro Gly Gln Ser Pro Lys Leu Leu Ile Phe Trp Ala
Ser Thr Arg65 70 75 80Gln Ser Gly Val Pro Asp Arg Phe Ile Gly Arg
Gly Ser Gly Thr Asp 85 90 95Phe Thr Leu Thr Ile Ser Gly Val Gln Ala
Glu Asp Leu Ala Ile Tyr 100 105 110Tyr Cys Gln Gln Tyr Tyr Asp Thr
Leu Thr Asp Thr Phe Gly Ala Gly 115 120 125Thr Lys Leu Glu Leu Lys
Arg Ala Asp Ala Ala Pro Thr Val Ser Ile 130 135 140Phe Pro Pro Ser
Thr Glu Gln Leu Ala Thr Gly Gly Ala Ser145 150 1551417PRTArtificial
SequenceSynthetic Peptide 14Lys Ser Ser Gln Ser Leu Leu Tyr Ser Gly
Thr Gln Lys Asn Tyr Leu1 5 10 15Ala157PRTArtificial
SequenceSynthetic Peptide 15Trp Ala Ser Thr Arg Gln Ser1
51610PRTArtificial SequenceSynthetic Peptide 16Gln Gln Tyr Tyr Asp
Thr Leu Thr Asp Thr1 5 1017469DNAArtificial SequenceSynthetic
PolynucleotideCDS(1)...(468) 17atg gac atc agg ctc agc ttg gct ttc
ctg gtc ctt ttc ata aaa ggt 48Met Asp Ile Arg Leu Ser Leu Ala Phe
Leu Val Leu Phe Ile Lys Gly 1 5 10 15gtc cag tgt gag gtg cgg ctg
gtg gag tct ggg gga ggc tta gtg cag 96Val Gln Cys Glu Val Arg Leu
Val Glu Ser Gly Gly Gly Leu Val Gln 20 25 30cct gga aag tcc atg aaa
ctc tcc tgt gta gcc tcg gga ttc aaa ttc 144Pro Gly Lys Ser Met Lys
Leu Ser Cys Val Ala Ser Gly Phe Lys Phe 35 40 45agt aac tat tac atg
tcc tgg gtc cgc cag gct cca gcg aag ggt ctg 192Ser Asn Tyr Tyr Met
Ser Trp Val Arg Gln Ala Pro Ala Lys Gly Leu 50 55 60gag tgg gtc gca
tcc att agt gat ggt ggt ggt gac act ttc tgt cga 240Glu Trp Val Ala
Ser Ile Ser Asp Gly Gly Gly Asp Thr Phe Cys Arg 65 70 75 80gac ttg
gtg aag ggc cga ttc act atc tcc aga gat aat gca aaa agt 288Asp Leu
Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Ser 85 90 95acc
ctt tac ctg caa atg gac agt ctg agg cct gag gac acg gcc act 336Thr
Leu Tyr Leu Gln Met Asp Ser Leu Arg Pro Glu Asp Thr Ala Thr 100 105
110tat tac tgt gca aga cgg gga gca gct atg ggg ggt gtt atg gat gcc
384Tyr Tyr Cys Ala Arg Arg Gly Ala Ala Met Gly Gly Val Met Asp Ala
115 120 125tgg ggt caa gga act tca gtc act gtc tcc tca gct gaa aca
aca gcc 432Trp Gly Gln Gly Thr Ser Val Thr Val Ser Ser Ala Glu Thr
Thr Ala 130 135 140cca tct gtc tat cca ctg gct cct gga act gct ctc
a 469Pro Ser Val Tyr Pro Leu Ala Pro Gly Thr Ala Leu145 150
15518156PRTArtificial SequenceSynthetic Construct 18Met Asp Ile Arg
Leu Ser Leu Ala Phe Leu Val Leu Phe Ile Lys Gly1 5 10 15Val Gln Cys
Glu Val Arg Leu Val Glu Ser Gly Gly Gly Leu Val Gln 20 25 30Pro Gly
Lys Ser Met Lys Leu Ser Cys Val Ala Ser Gly Phe Lys Phe 35 40 45Ser
Asn Tyr Tyr Met Ser Trp Val Arg Gln Ala Pro Ala Lys Gly Leu 50 55
60Glu Trp Val Ala Ser Ile Ser Asp Gly Gly Gly Asp Thr Phe Cys Arg65
70 75 80Asp Leu Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys
Ser 85 90 95Thr Leu Tyr Leu Gln Met Asp Ser Leu Arg Pro Glu Asp Thr
Ala Thr 100 105 110Tyr Tyr Cys Ala Arg Arg Gly Ala Ala Met Gly Gly
Val Met Asp Ala 115 120 125Trp Gly Gln Gly Thr Ser Val
Thr Val Ser Ser Ala Glu Thr Thr Ala 130 135 140Pro Ser Val Tyr Pro
Leu Ala Pro Gly Thr Ala Leu145 150 1551910PRTArtificial
SequenceSynthetic Peptide 19Gly Phe Lys Phe Ser Asn Tyr Tyr Met
Ser1 5 102017PRTArtificial SequenceSynthetic Peptide 20Ser Ile Ser
Asp Gly Gly Gly Asp Thr Phe Cys Arg Asp Leu Val Lys1 5 10
15Gly2115PRTArtificial SequenceSynthetic Peptide 21Arg Gly Ala Ala
Met Gly Gly Val Met Asp Ala Trp Gly Gln Gly1 5 10 1522111PRTHomo
sapiens 22Pro Arg Val Ala Gly Val Pro Gly Glu Ala Glu Gln Pro Ala
Pro Glu1 5 10 15Leu Val Glu Val Glu Val Gly Ser Thr Ala Leu Leu Lys
Cys Gly Leu 20 25 30Ser Gln Ser Gln Gly Asn Leu Ser His Val Asp Trp
Phe Ser Val His 35 40 45Lys Glu Lys Arg Thr Leu Ile Phe Arg Val Arg
Gln Gly Gln Gly Gln 50 55 60Ser Glu Pro Gly Glu Tyr Glu Gln Arg Leu
Ser Leu Gln Asp Arg Gly65 70 75 80Ala Thr Leu Ala Leu Thr Gln Val
Thr Pro Gln Asp Glu Arg Ile Phe 85 90 95Leu Cys Gln Gly Lys Arg Pro
Arg Ser Gln Glu Tyr Arg Ile Gln 100 105 11023104PRTHomo sapiens
23Pro Asn Ile Gln Val Asn Pro Leu Gly Ile Pro Val Asn Ser Lys Glu1
5 10 15Pro Glu Glu Val Ala Thr Cys Val Gly Arg Asn Gly Tyr Pro Ile
Pro 20 25 30Gln Val Ile Trp Tyr Lys Asn Gly Arg Pro Leu Lys Glu Glu
Lys Asn 35 40 45Arg Val His Ile Gln Ser Ser Gln Thr Val Glu Ser Ser
Gly Leu Tyr 50 55 60Thr Leu Gln Ser Ile Leu Lys Ala Gln Leu Val Lys
Glu Asp Lys Asp65 70 75 80Ala Gln Phe Tyr Cys Glu Leu Asn Tyr Arg
Leu Pro Ser Gly Asn His 85 90 95Met Lys Glu Ser Arg Glu Val Thr
1002478PRTHomo sapiens 24Pro Val Phe Tyr Pro Thr Glu Lys Val Trp
Leu Glu Val Glu Pro Val1 5 10 15Gly Met Leu Lys Glu Gly Asp Arg Val
Glu Ile Arg Cys Leu Ala Asp 20 25 30Gly Asn Pro Pro Pro His Phe Ser
Ile Ser Lys Gln Asn Pro Ser Thr 35 40 45Arg Glu Ala Glu Glu Glu Thr
Thr Asn Asp Asn Gly Val Leu Val Leu 50 55 60Glu Pro Ala Arg Lys Glu
His Ser Gly Arg Tyr Glu Cys Gln65 70 752590PRTHomo sapiens 25Pro
Gln Glu Leu Leu Val Asn Tyr Val Ser Asp Val Arg Val Ser Pro1 5 10
15Ala Ala Pro Glu Arg Gln Glu Gly Ser Ser Leu Thr Leu Thr Cys Glu
20 25 30Ala Glu Ser Ser Gln Asp Leu Glu Phe Gln Trp Leu Arg Glu Glu
Thr 35 40 45Asp Gln Val Leu Glu Arg Gly Pro Val Leu Gln Leu His Asp
Leu Lys 50 55 60Arg Glu Ala Gly Gly Gly Tyr Arg Cys Val Ala Ser Val
Pro Ser Ile65 70 75 80Pro Gly Leu Asn Arg Thr Gln Leu Val Lys 85
902681PRTHomo sapiens 26Pro Pro Trp Met Ala Phe Lys Glu Arg Lys Val
Trp Val Lys Glu Asn1 5 10 15Met Val Leu Asn Leu Ser Cys Glu Ala Ser
Gly His Pro Arg Pro Thr 20 25 30Ile Ser Trp Asn Val Asn Gly Thr Ala
Ser Glu Gln Asp Gln Asp Pro 35 40 45Gln Arg Val Leu Ser Thr Leu Asn
Val Leu Val Thr Pro Glu Leu Leu 50 55 60Glu Thr Gly Val Glu Cys Thr
Ala Ser Asn Asp Leu Gly Lys Asn Thr65 70 75 80Ser271823PRTHomo
sapiens 27Met Ala Leu Ser Ser Ala Trp Arg Ser Val Leu Pro Leu Trp
Leu Leu1 5 10 15Trp Ser Ala Ala Cys Ser Arg Ala Ala Ser Gly Asp Asp
Asn Ala Phe 20 25 30Pro Phe Asp Ile Glu Gly Ser Ser Ala Val Gly Arg
Gln Asp Pro Pro 35 40 45Glu Thr Ser Glu Pro Arg Val Ala Leu Gly Arg
Leu Pro Pro Ala Ala 50 55 60Glu Lys Cys Asn Ala Gly Phe Phe His Thr
Leu Ser Gly Glu Cys Val65 70 75 80Pro Cys Asp Cys Asn Gly Asn Ser
Asn Glu Cys Leu Asp Gly Ser Gly 85 90 95Tyr Cys Val His Cys Gln Arg
Asn Thr Thr Gly Glu His Cys Glu Lys 100 105 110Cys Leu Asp Gly Tyr
Ile Gly Asp Ser Ile Arg Gly Ala Pro Gln Phe 115 120 125Cys Gln Pro
Cys Pro Cys Pro Leu Pro His Leu Ala Asn Phe Ala Glu 130 135 140Ser
Cys Tyr Arg Lys Asn Gly Ala Val Arg Cys Ile Cys Asn Glu Asn145 150
155 160Tyr Ala Gly Pro Asn Cys Glu Arg Cys Ala Pro Gly Tyr Tyr Gly
Asn 165 170 175Pro Leu Leu Ile Gly Ser Thr Cys Lys Lys Cys Asp Cys
Ser Gly Asn 180 185 190Ser Asp Pro Asn Leu Ile Phe Glu Asp Cys Asp
Glu Val Thr Gly Gln 195 200 205Cys Arg Asn Cys Leu Arg Asn Thr Thr
Gly Phe Lys Cys Glu Arg Cys 210 215 220Ala Pro Gly Tyr Tyr Gly Asp
Ala Arg Ile Ala Lys Asn Cys Ala Val225 230 235 240Cys Asn Cys Gly
Gly Gly Pro Cys Asp Ser Val Thr Gly Glu Cys Leu 245 250 255Glu Glu
Gly Phe Glu Pro Pro Thr Gly Met Asp Cys Pro Thr Ile Ser 260 265
270Cys Asp Lys Cys Val Trp Asp Leu Thr Asp Asp Leu Arg Leu Ala Ala
275 280 285Leu Ser Ile Glu Glu Gly Lys Ser Gly Val Leu Ser Val Ser
Ser Gly 290 295 300Ala Ala Ala His Arg His Val Asn Glu Ile Asn Ala
Thr Ile Tyr Leu305 310 315 320Leu Lys Thr Lys Leu Ser Glu Arg Glu
Asn Gln Tyr Ala Leu Arg Lys 325 330 335Ile Gln Ile Asn Asn Ala Glu
Asn Thr Met Lys Ser Leu Leu Ser Asp 340 345 350Val Glu Glu Leu Val
Glu Lys Glu Asn Gln Ala Ser Arg Lys Gly Gln 355 360 365Leu Val Gln
Lys Glu Ser Met Asp Thr Ile Asn His Ala Ser Gln Leu 370 375 380Val
Glu Gln Ala His Asp Met Arg Asp Lys Ile Gln Glu Ile Asn Asn385 390
395 400Lys Met Leu Tyr Tyr Gly Glu Glu His Glu Leu Ser Pro Lys Glu
Ile 405 410 415Ser Glu Lys Leu Val Leu Ala Gln Lys Met Leu Glu Glu
Ile Arg Ser 420 425 430Arg Gln Pro Phe Phe Thr Gln Arg Glu Leu Val
Asp Glu Glu Ala Asp 435 440 445Glu Ala Tyr Glu Leu Leu Ser Gln Ala
Glu Ser Trp Gln Arg Leu His 450 455 460Asn Glu Thr Arg Thr Leu Phe
Pro Val Val Leu Glu Gln Leu Asp Asp465 470 475 480Tyr Asn Ala Lys
Leu Ser Asp Leu Gln Glu Ala Leu Asp Gln Ala Leu 485 490 495Asn Tyr
Val Arg Asp Ala Glu Asp Met Asn Arg Ala Thr Ala Ala Arg 500 505
510Gln Arg Asp His Glu Lys Gln Gln Glu Arg Val Arg Glu Gln Met Glu
515 520 525Val Val Asn Met Ser Leu Ser Thr Ser Ala Asp Ser Leu Thr
Thr Pro 530 535 540Arg Leu Thr Leu Ser Glu Leu Asp Asp Ile Ile Lys
Asn Ala Ser Gly545 550 555 560Ile Tyr Ala Glu Ile Asp Gly Ala Lys
Ser Glu Leu Gln Val Lys Leu 565 570 575Ser Asn Leu Ser Asn Leu Ser
His Asp Leu Val Gln Glu Ala Ile Asp 580 585 590His Ala Gln Asp Leu
Gln Gln Glu Ala Asn Glu Leu Ser Arg Lys Leu 595 600 605His Ser Ser
Asp Met Asn Gly Leu Val Gln Lys Ala Leu Asp Ala Ser 610 615 620Asn
Val Tyr Glu Asn Ile Val Asn Tyr Val Ser Glu Ala Asn Glu Thr625 630
635 640Ala Glu Phe Ala Leu Asn Thr Thr Asp Arg Ile Tyr Asp Ala Val
Ser 645 650 655Gly Ile Asp Thr Gln Ile Ile Tyr His Lys Asp Glu Ser
Glu Asn Leu 660 665 670Leu Asn Gln Ala Arg Glu Leu Gln Ala Lys Ala
Glu Ser Ser Ser Asp 675 680 685Glu Ala Val Ala Asp Thr Ser Arg Arg
Val Gly Gly Ala Leu Ala Arg 690 695 700Lys Ser Ala Leu Lys Thr Arg
Leu Ser Asp Ala Val Lys Gln Leu Gln705 710 715 720Ala Ala Glu Arg
Gly Asp Ala Gln Gln Arg Leu Gly Gln Ser Arg Leu 725 730 735Ile Thr
Glu Glu Ala Asn Arg Thr Thr Met Glu Val Gln Gln Ala Thr 740 745
750Ala Pro Met Ala Asn Asn Leu Thr Asn Trp Ser Gln Asn Leu Gln His
755 760 765Phe Asp Ser Ser Ala Tyr Asn Thr Ala Val Asn Ser Ala Arg
Asp Ala 770 775 780Val Arg Asn Leu Thr Glu Val Val Pro Gln Leu Leu
Asp Gln Leu Arg785 790 795 800Thr Val Glu Gln Lys Arg Pro Ala Ser
Asn Val Ser Ala Ser Ile Gln 805 810 815Arg Ile Arg Glu Leu Ile Ala
Gln Thr Arg Ser Val Ala Ser Lys Ile 820 825 830Gln Val Ser Met Met
Phe Asp Gly Gln Ser Ala Val Glu Val His Ser 835 840 845Arg Thr Ser
Met Asp Asp Leu Lys Ala Phe Thr Ser Leu Ser Leu Tyr 850 855 860Met
Lys Pro Pro Val Lys Arg Pro Glu Leu Thr Glu Thr Ala Asp Gln865 870
875 880Phe Ile Leu Tyr Leu Gly Ser Lys Asn Ala Lys Lys Glu Tyr Met
Gly 885 890 895Leu Ala Ile Lys Asn Asp Asn Leu Val Tyr Val Tyr Asn
Leu Gly Thr 900 905 910Lys Asp Val Glu Ile Pro Leu Asp Ser Lys Pro
Val Ser Ser Trp Pro 915 920 925Ala Tyr Phe Ser Ile Val Lys Ile Glu
Arg Val Gly Lys His Gly Lys 930 935 940Val Phe Leu Thr Val Pro Ser
Leu Ser Ser Thr Ala Glu Glu Lys Phe945 950 955 960Ile Lys Lys Gly
Glu Phe Ser Gly Asp Asp Ser Leu Leu Asp Leu Asp 965 970 975Pro Glu
Asp Thr Val Phe Tyr Val Gly Gly Val Pro Ser Asn Phe Lys 980 985
990Leu Pro Thr Ser Leu Asn Leu Pro Gly Phe Val Gly Cys Leu Glu Leu
995 1000 1005Ala Thr Leu Asn Asn Asp Val Ile Ser Leu Tyr Asn Phe
Lys His Ile 1010 1015 1020Tyr Asn Met Asp Pro Ser Thr Ser Val Pro
Cys Ala Arg Asp Lys Leu1025 1030 1035 1040Ala Phe Thr Gln Ser Arg
Ala Ala Ser Tyr Phe Phe Asp Gly Ser Gly 1045 1050 1055Tyr Ala Val
Val Arg Asp Ile Thr Arg Arg Gly Lys Phe Gly Gln Val 1060 1065
1070Thr Arg Phe Asp Ile Glu Val Arg Thr Pro Ala Asp Asn Gly Leu Ile
1075 1080 1085Leu Leu Met Val Asn Gly Ser Met Phe Phe Arg Leu Glu
Met Arg Asn 1090 1095 1100Gly Tyr Leu His Val Phe Tyr Asp Phe Gly
Phe Ser Gly Gly Pro Val1105 1110 1115 1120His Leu Glu Asp Thr Leu
Lys Lys Ala Gln Ile Asn Asp Ala Lys Tyr 1125 1130 1135His Glu Ile
Ser Ile Ile Tyr His Asn Asp Lys Lys Met Ile Leu Val 1140 1145
1150Val Asp Arg Arg His Val Lys Ser Met Asp Asn Glu Lys Met Lys Ile
1155 1160 1165Pro Phe Thr Asp Ile Tyr Ile Gly Gly Ala Pro Pro Glu
Ile Leu Gln 1170 1175 1180Ser Arg Ala Leu Arg Ala His Leu Pro Leu
Asp Ile Asn Phe Arg Gly1185 1190 1195 1200Cys Met Lys Gly Phe Gln
Phe Gln Lys Lys Asp Phe Asn Leu Leu Glu 1205 1210 1215Gln Thr Glu
Thr Leu Gly Val Gly Tyr Gly Cys Pro Glu Asp Ser Leu 1220 1225
1230Ile Ser Arg Arg Ala Tyr Phe Asn Gly Gln Ser Phe Ile Ala Ser Ile
1235 1240 1245Gln Lys Ile Ser Phe Phe Asp Gly Phe Glu Gly Gly Phe
Asn Phe Arg 1250 1255 1260Thr Leu Gln Pro Asn Gly Leu Leu Phe Tyr
Tyr Ala Ser Gly Ser Asp1265 1270 1275 1280Val Phe Ser Ile Ser Leu
Asp Asn Gly Thr Val Ile Met Asp Val Lys 1285 1290 1295Gly Ile Lys
Val Gln Ser Val Asp Lys Gln Tyr Asn Asp Gly Leu Ser 1300 1305
1310His Phe Val Ile Ser Ser Val Ser Pro Thr Arg Tyr Glu Leu Ile Val
1315 1320 1325Asp Lys Ser Arg Val Gly Ser Lys Asn Pro Thr Lys Gly
Lys Ile Glu 1330 1335 1340Gln Thr Gln Ala Ser Glu Lys Lys Phe Tyr
Phe Gly Gly Ser Pro Ile1345 1350 1355 1360Ser Ala Gln Tyr Ala Asn
Phe Thr Gly Cys Ile Ser Asn Ala Tyr Phe 1365 1370 1375Thr Arg Val
Asp Arg Asp Val Glu Val Glu Asp Phe Gln Arg Tyr Thr 1380 1385
1390Glu Lys Val His Thr Ser Leu Tyr Glu Cys Pro Ile Glu Ser Ser Pro
1395 1400 1405Leu Phe Leu Leu His Lys Lys Gly Lys Asn Leu Ser Lys
Pro Lys Ala 1410 1415 1420Ser Gln Asn Lys Lys Gly Gly Lys Ser Lys
Asp Ala Pro Ser Trp Asp1425 1430 1435 1440Pro Val Ala Leu Lys Leu
Pro Glu Arg Asn Thr Pro Arg Asn Ser His 1445 1450 1455Cys His Leu
Ser Asn Ser Pro Arg Ala Ile Glu His Ala Tyr Gln Tyr 1460 1465
1470Gly Gly Thr Ala Asn Ser Arg Gln Glu Phe Glu His Leu Lys Gly Asp
1475 1480 1485Phe Gly Ala Lys Ser Gln Phe Ser Ile Arg Leu Arg Thr
Arg Ser Ser 1490 1495 1500His Gly Met Ile Phe Tyr Val Ser Asp Gln
Glu Glu Asn Asp Phe Met1505 1510 1515 1520Thr Leu Phe Leu Ala His
Gly Arg Leu Val Tyr Met Phe Asn Val Gly 1525 1530 1535His Lys Lys
Leu Lys Ile Arg Ser Gln Glu Lys Tyr Asn Asp Gly Leu 1540 1545
1550Trp His Asp Val Ile Phe Ile Arg Glu Arg Ser Ser Gly Arg Leu Val
1555 1560 1565Ile Asp Gly Leu Arg Val Leu Glu Glu Ser Leu Pro Pro
Thr Glu Ala 1570 1575 1580Thr Trp Lys Ile Lys Gly Pro Ile Tyr Leu
Gly Gly Val Ala Pro Gly1585 1590 1595 1600Lys Ala Val Lys Asn Val
Gln Ile Asn Ser Ile Tyr Ser Phe Ser Gly 1605 1610 1615Cys Leu Ser
Asn Leu Gln Leu Asn Gly Ala Ser Ile Thr Ser Ala Ser 1620 1625
1630Gln Thr Phe Ser Val Thr Pro Cys Phe Glu Gly Pro Met Glu Thr Gly
1635 1640 1645Thr Tyr Phe Ser Thr Glu Gly Gly Tyr Val Val Leu Asp
Glu Ser Phe 1650 1655 1660Asn Ile Gly Leu Lys Phe Glu Ile Ala Phe
Glu Val Arg Pro Arg Ser1665 1670 1675 1680Ser Ser Gly Thr Leu Val
His Gly His Ser Val Asn Gly Glu Tyr Leu 1685 1690 1695Asn Val His
Met Lys Asn Gly Gln Val Ile Val Lys Val Asn Asn Gly 1700 1705
1710Ile Arg Asp Phe Ser Thr Ser Val Thr Pro Lys Gln Ser Leu Cys Asp
1715 1720 1725Gly Arg Trp His Arg Ile Thr Val Ile Arg Asp Ser Asn
Val Val Gln 1730 1735 1740Leu Asp Val Asp Ser Glu Val Asn His Val
Val Gly Pro Leu Asn Pro1745 1750 1755 1760Lys Pro Ile Asp His Arg
Glu Pro Val Phe Val Gly Gly Val Pro Glu 1765 1770 1775Ser Leu Leu
Thr Pro Arg Leu Ala Pro Ser Lys Pro Phe Thr Gly Cys 1780 1785
1790Ile Arg His Phe Val Ile Asp Gly His Pro Val Ser Phe Ser Lys Ala
1795 1800 1805Ala Leu Val Ser Gly Ala Val Ser Ile Asn Ser Cys Pro
Ala Ala 1810 1815 1820281816PRTHomo sapiens 28Met Ala Leu Ser Ser
Ala Trp Arg Ser Val Leu Pro Leu Trp Leu Leu1 5 10 15Trp Ser Ala Ala
Cys Ser Arg Ala Ala Ser Gly Asp Asp Asn Ala Phe 20 25 30Pro Phe Asp
Ile Glu Gly Ser Ser Ala Val Gly Arg Gln Asp Pro Pro 35 40 45Glu Thr
Ser Glu Pro Arg Val Ala Leu Gly Arg Leu Pro Pro Ala Ala 50 55 60Glu
Lys Cys Asn Ala Gly Phe Phe His Thr Leu Ser Gly Glu Cys Val65 70 75
80Pro Cys Asp Cys
Asn Gly Asn Ser Asn Glu Cys Leu Asp Gly Ser Gly 85 90 95Tyr Cys Val
His Cys Gln Arg Asn Thr Thr Gly Glu His Cys Glu Lys 100 105 110Cys
Leu Asp Gly Tyr Ile Gly Asp Ser Ile Arg Gly Ala Pro Gln Phe 115 120
125Cys Gln Pro Cys Pro Cys Pro Leu Pro His Leu Ala Asn Phe Ala Glu
130 135 140Ser Cys Tyr Arg Lys Asn Gly Ala Val Arg Cys Ile Cys Asn
Glu Asn145 150 155 160Tyr Ala Gly Pro Asn Cys Glu Arg Cys Ala Pro
Gly Tyr Tyr Gly Asn 165 170 175Pro Leu Leu Ile Gly Ser Thr Cys Lys
Lys Cys Asp Cys Ser Gly Asn 180 185 190Ser Asp Pro Asn Leu Ile Phe
Glu Asp Cys Asp Glu Val Thr Gly Gln 195 200 205Cys Arg Asn Cys Leu
Arg Asn Thr Thr Gly Phe Lys Cys Glu Arg Cys 210 215 220Ala Pro Gly
Tyr Tyr Gly Asp Ala Arg Ile Ala Lys Asn Cys Ala Val225 230 235
240Cys Asn Cys Gly Gly Gly Pro Cys Asp Ser Val Thr Gly Glu Cys Leu
245 250 255Glu Glu Gly Phe Glu Pro Pro Thr Gly Cys Asp Lys Cys Val
Trp Asp 260 265 270Leu Thr Asp Asp Leu Arg Leu Ala Ala Leu Ser Ile
Glu Glu Gly Lys 275 280 285Ser Gly Val Leu Ser Val Ser Ser Gly Ala
Ala Ala His Arg His Val 290 295 300Asn Glu Ile Asn Ala Thr Ile Tyr
Leu Leu Lys Thr Lys Leu Ser Glu305 310 315 320Arg Glu Asn Gln Tyr
Ala Leu Arg Lys Ile Gln Ile Asn Asn Ala Glu 325 330 335Asn Thr Met
Lys Ser Leu Leu Ser Asp Val Glu Glu Leu Val Glu Lys 340 345 350Glu
Asn Gln Ala Ser Arg Lys Gly Gln Leu Val Gln Lys Glu Ser Met 355 360
365Asp Thr Ile Asn His Ala Ser Gln Leu Val Glu Gln Ala His Asp Met
370 375 380Arg Asp Lys Ile Gln Glu Ile Asn Asn Lys Met Leu Tyr Tyr
Gly Glu385 390 395 400Glu His Glu Leu Ser Pro Lys Glu Ile Ser Glu
Lys Leu Val Leu Ala 405 410 415Gln Lys Met Leu Glu Glu Ile Arg Ser
Arg Gln Pro Phe Phe Thr Gln 420 425 430Arg Glu Leu Val Asp Glu Glu
Ala Asp Glu Ala Tyr Glu Leu Leu Ser 435 440 445Gln Ala Glu Ser Trp
Gln Arg Leu His Asn Glu Thr Arg Thr Leu Phe 450 455 460Pro Val Val
Leu Glu Gln Leu Asp Asp Tyr Asn Ala Lys Leu Ser Asp465 470 475
480Leu Gln Glu Ala Leu Asp Gln Ala Leu Asn Tyr Val Arg Asp Ala Glu
485 490 495Asp Met Asn Arg Ala Thr Ala Ala Arg Gln Arg Asp His Glu
Lys Gln 500 505 510Gln Glu Arg Val Arg Glu Gln Met Glu Val Val Asn
Met Ser Leu Ser 515 520 525Thr Ser Ala Asp Ser Leu Thr Thr Pro Arg
Leu Thr Leu Ser Glu Leu 530 535 540Asp Asp Ile Ile Lys Asn Ala Ser
Gly Ile Tyr Ala Glu Ile Asp Gly545 550 555 560Ala Lys Ser Glu Leu
Gln Val Lys Leu Ser Asn Leu Ser Asn Leu Ser 565 570 575His Asp Leu
Val Gln Glu Ala Ile Asp His Ala Gln Asp Leu Gln Gln 580 585 590Glu
Ala Asn Glu Leu Ser Arg Lys Leu His Ser Ser Asp Met Asn Gly 595 600
605Leu Val Gln Lys Ala Leu Asp Ala Ser Asn Val Tyr Glu Asn Ile Val
610 615 620Asn Tyr Val Ser Glu Ala Asn Glu Thr Ala Glu Phe Ala Leu
Asn Thr625 630 635 640Thr Asp Arg Ile Tyr Asp Ala Val Ser Gly Ile
Asp Thr Gln Ile Ile 645 650 655Tyr His Lys Asp Glu Ser Glu Asn Leu
Leu Asn Gln Ala Arg Glu Leu 660 665 670Gln Ala Lys Ala Glu Ser Ser
Ser Asp Glu Ala Val Ala Asp Thr Ser 675 680 685Arg Arg Val Gly Gly
Ala Leu Ala Arg Lys Ser Ala Leu Lys Thr Arg 690 695 700Leu Ser Asp
Ala Val Lys Gln Leu Gln Ala Ala Glu Arg Gly Asp Ala705 710 715
720Gln Gln Arg Leu Gly Gln Ser Arg Leu Ile Thr Glu Glu Ala Asn Arg
725 730 735Thr Thr Met Glu Val Gln Gln Ala Thr Ala Pro Met Ala Asn
Asn Leu 740 745 750Thr Asn Trp Ser Gln Asn Leu Gln His Phe Asp Ser
Ser Ala Tyr Asn 755 760 765Thr Ala Val Asn Ser Ala Arg Asp Ala Val
Arg Asn Leu Thr Glu Val 770 775 780Val Pro Gln Leu Leu Asp Gln Leu
Arg Thr Val Glu Gln Lys Arg Pro785 790 795 800Ala Ser Asn Val Ser
Ala Ser Ile Gln Arg Ile Arg Glu Leu Ile Ala 805 810 815Gln Thr Arg
Ser Val Ala Ser Lys Ile Gln Val Ser Met Met Phe Asp 820 825 830Gly
Gln Ser Ala Val Glu Val His Ser Arg Thr Ser Met Asp Asp Leu 835 840
845Lys Ala Phe Thr Ser Leu Ser Leu Tyr Met Lys Pro Pro Val Lys Arg
850 855 860Pro Glu Leu Thr Glu Thr Ala Asp Gln Phe Ile Leu Tyr Leu
Gly Ser865 870 875 880Lys Asn Ala Lys Lys Glu Tyr Met Gly Leu Ala
Ile Lys Asn Asp Asn 885 890 895Leu Val Tyr Val Tyr Asn Leu Gly Thr
Lys Asp Val Glu Ile Pro Leu 900 905 910Asp Ser Lys Pro Val Ser Ser
Trp Pro Ala Tyr Phe Ser Ile Val Lys 915 920 925Ile Glu Arg Val Gly
Lys His Gly Lys Val Phe Leu Thr Val Pro Ser 930 935 940Leu Ser Ser
Thr Ala Glu Glu Lys Phe Ile Lys Lys Gly Glu Phe Ser945 950 955
960Gly Asp Asp Ser Leu Leu Asp Leu Asp Pro Glu Asp Thr Val Phe Tyr
965 970 975Val Gly Gly Val Pro Ser Asn Phe Lys Leu Pro Thr Ser Leu
Asn Leu 980 985 990Pro Gly Phe Val Gly Cys Leu Glu Leu Ala Thr Leu
Asn Asn Asp Val 995 1000 1005Ile Ser Leu Tyr Asn Phe Lys His Ile
Tyr Asn Met Asp Pro Ser Thr 1010 1015 1020Ser Val Pro Cys Ala Arg
Asp Lys Leu Ala Phe Thr Gln Ser Arg Ala1025 1030 1035 1040Ala Ser
Tyr Phe Phe Asp Gly Ser Gly Tyr Ala Val Val Arg Asp Ile 1045 1050
1055Thr Arg Arg Gly Lys Phe Gly Gln Val Thr Arg Phe Asp Ile Glu Val
1060 1065 1070Arg Thr Pro Ala Asp Asn Gly Leu Ile Leu Leu Met Val
Asn Gly Ser 1075 1080 1085Met Phe Phe Arg Leu Glu Met Arg Asn Gly
Tyr Leu His Val Phe Tyr 1090 1095 1100Asp Phe Gly Phe Ser Gly Gly
Pro Val His Leu Glu Asp Thr Leu Lys1105 1110 1115 1120Lys Ala Gln
Ile Asn Asp Ala Lys Tyr His Glu Ile Ser Ile Ile Tyr 1125 1130
1135His Asn Asp Lys Lys Met Ile Leu Val Val Asp Arg Arg His Val Lys
1140 1145 1150Ser Met Asp Asn Glu Lys Met Lys Ile Pro Phe Thr Asp
Ile Tyr Ile 1155 1160 1165Gly Gly Ala Pro Pro Glu Ile Leu Gln Ser
Arg Ala Leu Arg Ala His 1170 1175 1180Leu Pro Leu Asp Ile Asn Phe
Arg Gly Cys Met Lys Gly Phe Gln Phe1185 1190 1195 1200Gln Lys Lys
Asp Phe Asn Leu Leu Glu Gln Thr Glu Thr Leu Gly Val 1205 1210
1215Gly Tyr Gly Cys Pro Glu Asp Ser Leu Ile Ser Arg Arg Ala Tyr Phe
1220 1225 1230Asn Gly Gln Ser Phe Ile Ala Ser Ile Gln Lys Ile Ser
Phe Phe Asp 1235 1240 1245Gly Phe Glu Gly Gly Phe Asn Phe Arg Thr
Leu Gln Pro Asn Gly Leu 1250 1255 1260Leu Phe Tyr Tyr Ala Ser Gly
Ser Asp Val Phe Ser Ile Ser Leu Asp1265 1270 1275 1280Asn Gly Thr
Val Ile Met Asp Val Lys Gly Ile Lys Val Gln Ser Val 1285 1290
1295Asp Lys Gln Tyr Asn Asp Gly Leu Ser His Phe Val Ile Ser Ser Val
1300 1305 1310Ser Pro Thr Arg Tyr Glu Leu Ile Val Asp Lys Ser Arg
Val Gly Ser 1315 1320 1325Lys Asn Pro Thr Lys Gly Lys Ile Glu Gln
Thr Gln Ala Ser Glu Lys 1330 1335 1340Lys Phe Tyr Phe Gly Gly Ser
Pro Ile Ser Ala Gln Tyr Ala Asn Phe1345 1350 1355 1360Thr Gly Cys
Ile Ser Asn Ala Tyr Phe Thr Arg Val Asp Arg Asp Val 1365 1370
1375Glu Val Glu Asp Phe Gln Arg Tyr Thr Glu Lys Val His Thr Ser Leu
1380 1385 1390Tyr Glu Cys Pro Ile Glu Ser Ser Pro Leu Phe Leu Leu
His Lys Lys 1395 1400 1405Gly Lys Asn Leu Ser Lys Pro Lys Ala Ser
Gln Asn Lys Lys Gly Gly 1410 1415 1420Lys Ser Lys Asp Ala Pro Ser
Trp Asp Pro Val Ala Leu Lys Leu Pro1425 1430 1435 1440Glu Arg Asn
Thr Pro Arg Asn Ser His Cys His Leu Ser Asn Ser Pro 1445 1450
1455Arg Ala Ile Glu His Ala Tyr Gln Tyr Gly Gly Thr Ala Asn Ser Arg
1460 1465 1470Gln Glu Phe Glu His Leu Lys Gly Asp Phe Gly Ala Lys
Ser Gln Phe 1475 1480 1485Ser Ile Arg Leu Arg Thr Arg Ser Ser His
Gly Met Ile Phe Tyr Val 1490 1495 1500Ser Asp Gln Glu Glu Asn Asp
Phe Met Thr Leu Phe Leu Ala His Gly1505 1510 1515 1520Arg Leu Val
Tyr Met Phe Asn Val Gly His Lys Lys Leu Lys Ile Arg 1525 1530
1535Ser Gln Glu Lys Tyr Asn Asp Gly Leu Trp His Asp Val Ile Phe Ile
1540 1545 1550Arg Glu Arg Ser Ser Gly Arg Leu Val Ile Asp Gly Leu
Arg Val Leu 1555 1560 1565Glu Glu Ser Leu Pro Pro Thr Glu Ala Thr
Trp Lys Ile Lys Gly Pro 1570 1575 1580Ile Tyr Leu Gly Gly Val Ala
Pro Gly Lys Ala Val Lys Asn Val Gln1585 1590 1595 1600Ile Asn Ser
Ile Tyr Ser Phe Ser Gly Cys Leu Ser Asn Leu Gln Leu 1605 1610
1615Asn Gly Ala Ser Ile Thr Ser Ala Ser Gln Thr Phe Ser Val Thr Pro
1620 1625 1630Cys Phe Glu Gly Pro Met Glu Thr Gly Thr Tyr Phe Ser
Thr Glu Gly 1635 1640 1645Gly Tyr Val Val Leu Asp Glu Ser Phe Asn
Ile Gly Leu Lys Phe Glu 1650 1655 1660Ile Ala Phe Glu Val Arg Pro
Arg Ser Ser Ser Gly Thr Leu Val His1665 1670 1675 1680Gly His Ser
Val Asn Gly Glu Tyr Leu Asn Val His Met Lys Asn Gly 1685 1690
1695Gln Val Ile Val Lys Val Asn Asn Gly Ile Arg Asp Phe Ser Thr Ser
1700 1705 1710Val Thr Pro Lys Gln Ser Leu Cys Asp Gly Arg Trp His
Arg Ile Thr 1715 1720 1725Val Ile Arg Asp Ser Asn Val Val Gln Leu
Asp Val Asp Ser Glu Val 1730 1735 1740Asn His Val Val Gly Pro Leu
Asn Pro Lys Pro Ile Asp His Arg Glu1745 1750 1755 1760Pro Val Phe
Val Gly Gly Val Pro Glu Ser Leu Leu Thr Pro Arg Leu 1765 1770
1775Ala Pro Ser Lys Pro Phe Thr Gly Cys Ile Arg His Phe Val Ile Asp
1780 1785 1790Gly His Pro Val Ser Phe Ser Lys Ala Ala Leu Val Ser
Gly Ala Val 1795 1800 1805Ser Ile Asn Ser Cys Pro Ala Ala 1810
181529334DNAArtificial SequenceSynthetic PolynucleotideClone
1174.1.3 - variable Light ChainCDS(1)...(333) 29gac att gtg ctg aca
cag tct cct gct tcc tta gct gta tct ctg ggg 48Asp Ile Val Leu Thr
Gln Ser Pro Ala Ser Leu Ala Val Ser Leu Gly 1 5 10 15cag agg gcc
acc atc tca tgc agg gcc agc aaa agt gtc agt aca tct 96Gln Arg Ala
Thr Ile Ser Cys Arg Ala Ser Lys Ser Val Ser Thr Ser 20 25 30ggc tat
agt tat atg tac tgg tac caa cag aaa cca gga cag cca ccc 144Gly Tyr
Ser Tyr Met Tyr Trp Tyr Gln Gln Lys Pro Gly Gln Pro Pro 35 40 45aaa
ctc ctc atc tat ctt gca tcc aac cta gaa tct ggg gtc cct gcc 192Lys
Leu Leu Ile Tyr Leu Ala Ser Asn Leu Glu Ser Gly Val Pro Ala 50 55
60agg ttc agt ggc agt ggg tct ggg aca gac ttc acc ctc aac atc cat
240Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Asn Ile His
65 70 75 80cct gtg gag gag gag gat gct gca acc tat tac tgt caa cac
agt agg 288Pro Val Glu Glu Glu Asp Ala Ala Thr Tyr Tyr Cys Gln His
Ser Arg 85 90 95gag ctt cca ttc acg ttc ggc tcg ggg aca aag ttg gaa
ata aaa 333Glu Leu Pro Phe Thr Phe Gly Ser Gly Thr Lys Leu Glu Ile
Lys 100 105 110c 33430111PRTArtificial SequenceSynthetic Construct
30Asp Ile Val Leu Thr Gln Ser Pro Ala Ser Leu Ala Val Ser Leu Gly1
5 10 15Gln Arg Ala Thr Ile Ser Cys Arg Ala Ser Lys Ser Val Ser Thr
Ser 20 25 30Gly Tyr Ser Tyr Met Tyr Trp Tyr Gln Gln Lys Pro Gly Gln
Pro Pro 35 40 45Lys Leu Leu Ile Tyr Leu Ala Ser Asn Leu Glu Ser Gly
Val Pro Ala 50 55 60Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr
Leu Asn Ile His65 70 75 80Pro Val Glu Glu Glu Asp Ala Ala Thr Tyr
Tyr Cys Gln His Ser Arg 85 90 95Glu Leu Pro Phe Thr Phe Gly Ser Gly
Thr Lys Leu Glu Ile Lys 100 105 1103115PRTArtificial
SequenceSynthetic Peptideclone 1174.1.3 - CDR-L1 31Arg Ala Ser Lys
Ser Val Ser Thr Ser Gly Tyr Ser Tyr Met Tyr1 5 10
15326PRTArtificial SequenceSynthetic PeptideClone 1174.1.3 - CDR-L2
32Ala Ser Asn Leu Glu Ser1 5339PRTArtificial SequenceSynthetic
PeptideClone 1174.1.3 - CDR-L3 33Gln His Ser Arg Glu Leu Pro Phe
Thr1 534363DNAArtificial SequenceSynthetic Polynucleotideclone
1174.1.3 - Variable Heavy chainCDS(1)...(363) 34cag att cag ttg gtg
cag tct gga cct gag ctg aag aag cct gga gag 48Gln Ile Gln Leu Val
Gln Ser Gly Pro Glu Leu Lys Lys Pro Gly Glu 1 5 10 15aca gtc aag
atc tcc tgc aag gct tct ggg tat acc ttc aca aac tat 96Thr Val Lys
Ile Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asn Tyr 20 25 30gga atg
aac tgg gtg aag cag gct cca gga aag ggt tta aag tgg atg 144Gly Met
Asn Trp Val Lys Gln Ala Pro Gly Lys Gly Leu Lys Trp Met 35 40 45ggc
tgg ata aac acc tac act gga gag cca aca tat gct gat gac ttc 192Gly
Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Asp Asp Phe 50 55
60aag gga cgg ttt gcc ttg tct ttg gaa acc tct gcc agc act gcc tat
240Lys Gly Arg Phe Ala Leu Ser Leu Glu Thr Ser Ala Ser Thr Ala Tyr
65 70 75 80ttg cag atc aac aac ctc aaa aat gag gac atg gct aca tat
ttc tgt 288Leu Gln Ile Asn Asn Leu Lys Asn Glu Asp Met Ala Thr Tyr
Phe Cys 85 90 95gca aga tat agg tat aat aaa tac gag agg gct atg gac
tac tgg ggt 336Ala Arg Tyr Arg Tyr Asn Lys Tyr Glu Arg Ala Met Asp
Tyr Trp Gly 100 105 110caa gga acc tca gtc acc gtc tcc tca 363Gln
Gly Thr Ser Val Thr Val Ser Ser 115 12035121PRTArtificial
SequenceSynthetic Construct 35Gln Ile Gln Leu Val Gln Ser Gly Pro
Glu Leu Lys Lys Pro Gly Glu1 5 10 15Thr Val Lys Ile Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asn Tyr 20 25 30Gly Met Asn Trp Val Lys Gln
Ala Pro Gly Lys Gly Leu Lys Trp Met 35 40 45Gly Trp Ile Asn Thr Tyr
Thr Gly Glu Pro Thr Tyr Ala Asp Asp Phe 50 55 60Lys Gly Arg Phe Ala
Leu Ser Leu Glu Thr Ser Ala Ser Thr Ala Tyr65 70 75 80Leu Gln Ile
Asn Asn Leu Lys Asn Glu Asp Met Ala Thr Tyr Phe Cys 85 90 95Ala Arg
Tyr Arg Tyr Asn Lys Tyr Glu Arg Ala Met Asp Tyr Trp Gly 100 105
110Gln Gly Thr Ser Val Thr Val Ser Ser 115 1203610PRTArtificial
SequenceSynthetic PeptideClone 1174.1.3 -
CDR-H1 36Gly Tyr Thr Phe Thr Asn Tyr Gly Met Asn1 5
103717PRTArtificial SequenceSynthetic PeptideClone 1174.1.3 -
CDR-H2 37Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Asp Asp
Phe Lys1 5 10 15Gly3812PRTArtificial SequenceSynthetic PeptideClone
1174.1.3 - CDR-H3 38Tyr Arg Tyr Asn Lys Tyr Glu Arg Ala Met Asp
Tyr1 5 1039338DNAArtificial SequenceSynthetic PolynucleotideClone
1414.1.2 - variable Light ChainCDS(1)...(336) 39gac att gtg atg tca
cag tct cca tcc tcc ctg gct gtg tca gca gga 48Asp Ile Val Met Ser
Gln Ser Pro Ser Ser Leu Ala Val Ser Ala Gly 1 5 10 15gag aag gtc
act atg agc tgc aaa tcc agt cag agt ctg ctc aac agt 96Glu Lys Val
Thr Met Ser Cys Lys Ser Ser Gln Ser Leu Leu Asn Ser 20 25 30agc acc
cga aag aac ttc ttg gct tgg tac cag cag aaa cca ggg cag 144Ser Thr
Arg Lys Asn Phe Leu Ala Trp Tyr Gln Gln Lys Pro Gly Gln 35 40 45tct
cct aaa ctg ctg atc tac tgg gca tcc act agg gaa tct ggg gtc 192Ser
Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr Arg Glu Ser Gly Val 50 55
60cct gat cgc ttc aca ggc agt gga tct ggg aca gat ttc act ctc acc
240Pro Asp Arg Phe Thr Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
65 70 75 80atc agc agt gtg cag gct gaa gac ctg gca gtt tat tac tgc
aag caa 288Ile Ser Ser Val Gln Ala Glu Asp Leu Ala Val Tyr Tyr Cys
Lys Gln 85 90 95tct tat aat cgg tac acg ttc gga ggg ggg acc aag ctg
gaa ata aaa 336Ser Tyr Asn Arg Tyr Thr Phe Gly Gly Gly Thr Lys Leu
Glu Ile Lys 100 105 110cg 33840112PRTArtificial SequenceSynthetic
Construct 40Asp Ile Val Met Ser Gln Ser Pro Ser Ser Leu Ala Val Ser
Ala Gly1 5 10 15Glu Lys Val Thr Met Ser Cys Lys Ser Ser Gln Ser Leu
Leu Asn Ser 20 25 30Ser Thr Arg Lys Asn Phe Leu Ala Trp Tyr Gln Gln
Lys Pro Gly Gln 35 40 45Ser Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr
Arg Glu Ser Gly Val 50 55 60Pro Asp Arg Phe Thr Gly Ser Gly Ser Gly
Thr Asp Phe Thr Leu Thr65 70 75 80Ile Ser Ser Val Gln Ala Glu Asp
Leu Ala Val Tyr Tyr Cys Lys Gln 85 90 95Ser Tyr Asn Arg Tyr Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 105 1104117PRTArtificial
SequenceSynthetic Peptideclone 1414.1.2 - CDR-L1 41Lys Ser Ser Gln
Ser Leu Leu Asn Ser Ser Thr Arg Lys Asn Phe Leu1 5 10
15Ala427PRTArtificial SequenceSynthetic Peptideclone 1414.1.2 -
CDR-L2 42Trp Ala Ser Thr Arg Glu Ser1 5438PRTArtificial
SequenceSynthetic PeptideClone 1414.1.2 - CDR-L3 43Lys Gln Ser Tyr
Asn Arg Tyr Thr1 544351DNAArtificial SequenceSynthetic
PolynucleotideClone 1414.1.2 - variable Heavy chainCDS(1)...(351)
44gag atc cag ctg cag cag act gga cct gag ctg gtg aag cct ggg gct
48Glu Ile Gln Leu Gln Gln Thr Gly Pro Glu Leu Val Lys Pro Gly Ala 1
5 10 15tca gtg aag ata tcc tgc aag gct tct ggt tat tca ttc act gac
tac 96Ser Val Lys Ile Ser Cys Lys Ala Ser Gly Tyr Ser Phe Thr Asp
Tyr 20 25 30atc atg ctc tgg gtg aag cag agc cat gga aag agc ctt gag
tgg att 144Ile Met Leu Trp Val Lys Gln Ser His Gly Lys Ser Leu Glu
Trp Ile 35 40 45gga aat att aat cct tac tct ggt agt agt ggc tac aat
ctg aag ttc 192Gly Asn Ile Asn Pro Tyr Ser Gly Ser Ser Gly Tyr Asn
Leu Lys Phe 50 55 60aag ggc aag gcc aca ttg act gta gac aaa tct tcc
agc aca gcc tac 240Lys Gly Lys Ala Thr Leu Thr Val Asp Lys Ser Ser
Ser Thr Ala Tyr 65 70 75 80atg cag ctc aac agt ctg aca tct gag gac
tct gca gtc tat tac tgt 288Met Gln Leu Asn Ser Leu Thr Ser Glu Asp
Ser Ala Val Tyr Tyr Cys 85 90 95gca aga ggg aag gac ttt gct atg gac
tac tgg ggt caa gga acc tca 336Ala Arg Gly Lys Asp Phe Ala Met Asp
Tyr Trp Gly Gln Gly Thr Ser 100 105 110gtc acc gtc tcc tca 351Val
Thr Val Ser Ser 11545117PRTArtificial SequenceSynthetic Construct
45Glu Ile Gln Leu Gln Gln Thr Gly Pro Glu Leu Val Lys Pro Gly Ala1
5 10 15Ser Val Lys Ile Ser Cys Lys Ala Ser Gly Tyr Ser Phe Thr Asp
Tyr 20 25 30Ile Met Leu Trp Val Lys Gln Ser His Gly Lys Ser Leu Glu
Trp Ile 35 40 45Gly Asn Ile Asn Pro Tyr Ser Gly Ser Ser Gly Tyr Asn
Leu Lys Phe 50 55 60Lys Gly Lys Ala Thr Leu Thr Val Asp Lys Ser Ser
Ser Thr Ala Tyr65 70 75 80Met Gln Leu Asn Ser Leu Thr Ser Glu Asp
Ser Ala Val Tyr Tyr Cys 85 90 95Ala Arg Gly Lys Asp Phe Ala Met Asp
Tyr Trp Gly Gln Gly Thr Ser 100 105 110Val Thr Val Ser Ser
1154610PRTArtificial SequenceSynthetic PeptideClone 1414.1.2 -
CDR-H1 46Gly Tyr Ser Phe Thr Asp Tyr Ile Met Leu1 5
104717PRTArtificial SequenceSynthetic PeptideClone 1414.1.2 -
CDR-H2 47Asn Ile Asn Pro Tyr Ser Gly Ser Ser Gly Tyr Asn Leu Lys
Phe Lys1 5 10 15Gly487PRTArtificial SequenceSynthetic PeptideClone
1414.1.2 - CDR-H3 48Gly Lys Asp Phe Ala Met Asp1
549322DNAArtificial SequenceSynthetic PolynucleotideClone 1415.1.1
- variable Light ChainCDS(1)...(321) 49gac att gtg atg act cag tct
cca gcc acc ctg tct gtg act cca gga 48Asp Ile Val Met Thr Gln Ser
Pro Ala Thr Leu Ser Val Thr Pro Gly 1 5 10 15gat aga gtc tct ctt
tca tgc agg gcc agc cag agt att agc gac tac 96Asp Arg Val Ser Leu
Ser Cys Arg Ala Ser Gln Ser Ile Ser Asp Tyr 20 25 30tta cac tgg tat
caa caa aaa tca cat gag tct cca agg ctt ctc atc 144Leu His Trp Tyr
Gln Gln Lys Ser His Glu Ser Pro Arg Leu Leu Ile 35 40 45aaa tat gct
tcc caa tcc atc tct ggg atc ccc tcc agg ttc agt ggc 192Lys Tyr Ala
Ser Gln Ser Ile Ser Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60agt gga
tca ggg tca gat ttc act ctc agt atc aac agt gtg gaa cct 240Ser Gly
Ser Gly Ser Asp Phe Thr Leu Ser Ile Asn Ser Val Glu Pro 65 70 75
80gaa gat gtt gga gtg tat tac tgt caa aat ggt cac aac ttt cct cgg
288Glu Asp Val Gly Val Tyr Tyr Cys Gln Asn Gly His Asn Phe Pro Arg
85 90 95acg ttc ggt gga ggc acc aag ctg gaa atc aaa c 322Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 10550107PRTArtificial
SequenceSynthetic Construct 50Asp Ile Val Met Thr Gln Ser Pro Ala
Thr Leu Ser Val Thr Pro Gly1 5 10 15Asp Arg Val Ser Leu Ser Cys Arg
Ala Ser Gln Ser Ile Ser Asp Tyr 20 25 30Leu His Trp Tyr Gln Gln Lys
Ser His Glu Ser Pro Arg Leu Leu Ile 35 40 45Lys Tyr Ala Ser Gln Ser
Ile Ser Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Ser
Asp Phe Thr Leu Ser Ile Asn Ser Val Glu Pro65 70 75 80Glu Asp Val
Gly Val Tyr Tyr Cys Gln Asn Gly His Asn Phe Pro Arg 85 90 95Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 1055111PRTArtificial
SequenceSynthetic PeptideClone 1415.1.1 - CDR-L1 51Arg Ala Ser Gln
Ser Ile Ser Asp Tyr Leu His1 5 10527PRTArtificial SequenceSynthetic
PeptideClone 1415.1.1 - CDR-L2 52Tyr Ala Ser Gln Ser Ile Ser1
5539PRTArtificial SequenceSynthetic PeptideClone 1415.1.1 - CDR-L3
53Gln Asn Gly His Asn Phe Pro Arg Thr1 554366DNAArtificial
SequenceSynthetic PolynucleotideClone 1415.1.1 - variable Heavy
chainCDS(1)...(366) 54cag gtc caa ctg cag cag cct ggg gct gag ctt
gtg cag cct ggg gct 48Gln Val Gln Leu Gln Gln Pro Gly Ala Glu Leu
Val Gln Pro Gly Ala 1 5 10 15cca gtg aag ctg tcc tgc aag gct tct
ggc tac att ttc acc agc tac 96Pro Val Lys Leu Ser Cys Lys Ala Ser
Gly Tyr Ile Phe Thr Ser Tyr 20 25 30tgg atg aac tgg gtg aag cag agg
cct gga cga ggc ctc gag tgg att 144Trp Met Asn Trp Val Lys Gln Arg
Pro Gly Arg Gly Leu Glu Trp Ile 35 40 45gga agg att gat cct tcc gat
agt aaa att cac tac aat caa aag ttc 192Gly Arg Ile Asp Pro Ser Asp
Ser Lys Ile His Tyr Asn Gln Lys Phe 50 55 60aaa gac aag gcc aca ctg
act gta gac aga tcc tcc agc aca gcc tac 240Lys Asp Lys Ala Thr Leu
Thr Val Asp Arg Ser Ser Ser Thr Ala Tyr 65 70 75 80atc caa ctc ggc
agc ctg aca tct gag gac tct gcg gtc tat tat tgt 288Ile Gln Leu Gly
Ser Leu Thr Ser Glu Asp Ser Ala Val Tyr Tyr Cys 85 90 95gca aaa gag
ggg ggt tta cga cgg ggg gac tat gct atg gac tac tgg 336Ala Lys Glu
Gly Gly Leu Arg Arg Gly Asp Tyr Ala Met Asp Tyr Trp 100 105 110ggt
caa gga acc tca gtc acc gtc tcc tca 366Gly Gln Gly Thr Ser Val Thr
Val Ser Ser 115 12055122PRTArtificial SequenceSynthetic Construct
55Gln Val Gln Leu Gln Gln Pro Gly Ala Glu Leu Val Gln Pro Gly Ala1
5 10 15Pro Val Lys Leu Ser Cys Lys Ala Ser Gly Tyr Ile Phe Thr Ser
Tyr 20 25 30Trp Met Asn Trp Val Lys Gln Arg Pro Gly Arg Gly Leu Glu
Trp Ile 35 40 45Gly Arg Ile Asp Pro Ser Asp Ser Lys Ile His Tyr Asn
Gln Lys Phe 50 55 60Lys Asp Lys Ala Thr Leu Thr Val Asp Arg Ser Ser
Ser Thr Ala Tyr65 70 75 80Ile Gln Leu Gly Ser Leu Thr Ser Glu Asp
Ser Ala Val Tyr Tyr Cys 85 90 95Ala Lys Glu Gly Gly Leu Arg Arg Gly
Asp Tyr Ala Met Asp Tyr Trp 100 105 110Gly Gln Gly Thr Ser Val Thr
Val Ser Ser 115 1205610PRTArtificial SequenceSynthetic Peptideclone
1415.1.1 - CDR-H1 56Gly Tyr Ile Phe Thr Ser Tyr Trp Met Asn1 5
105717PRTArtificial SequenceSynthetic PeptideClone 1415.1.1 -
CDR-H2 57Arg Ile Asp Pro Ser Asp Ser Lys Ile His Tyr Asn Gln Lys
Phe Lys1 5 10 15Asp5813PRTArtificial SequenceSynthetic PeptideClone
1415.1.1 - CDR-H3 58Glu Gly Gly Leu Arg Arg Gly Asp Tyr Ala Met Asp
Tyr1 5 1059337DNAArtificial SequenceSynthetic PolynucleotideClone
1749.1.3 - Variable Light ChainCDS(1)...(336) 59gac att gtg atg tca
cag tct cca tcc tcc ctg gct gtg tca gca gga 48Asp Ile Val Met Ser
Gln Ser Pro Ser Ser Leu Ala Val Ser Ala Gly 1 5 10 15gag aag gtc
act atg aac tgc aaa tcc agt cgg agt ctg ctc aac agt 96Glu Lys Val
Thr Met Asn Cys Lys Ser Ser Arg Ser Leu Leu Asn Ser 20 25 30aga atc
cga aag aac tac ttg gct tgg tac cag cag aaa cca ggg cag 144Arg Ile
Arg Lys Asn Tyr Leu Ala Trp Tyr Gln Gln Lys Pro Gly Gln 35 40 45tct
cct aaa ctg ctg atc tac tgg gca tcc act agg gaa tct ggg gtc 192Ser
Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr Arg Glu Ser Gly Val 50 55
60cct gat cgc ttc aca ggc agt gga tct ggg aca gat ttc act ctc acc
240Pro Asp Arg Phe Thr Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
65 70 75 80atc agc agt gtg cag gct gaa gac ctg gca gtt tat tac tgc
aag caa 288Ile Ser Ser Val Gln Ala Glu Asp Leu Ala Val Tyr Tyr Cys
Lys Gln 85 90 95tct tat aat ctg ctc acg ttc ggt gct ggg acc aag ctg
gag ctg aaa 336Ser Tyr Asn Leu Leu Thr Phe Gly Ala Gly Thr Lys Leu
Glu Leu Lys 100 105 110c 33760112PRTArtificial SequenceSynthetic
Construct 60Asp Ile Val Met Ser Gln Ser Pro Ser Ser Leu Ala Val Ser
Ala Gly1 5 10 15Glu Lys Val Thr Met Asn Cys Lys Ser Ser Arg Ser Leu
Leu Asn Ser 20 25 30Arg Ile Arg Lys Asn Tyr Leu Ala Trp Tyr Gln Gln
Lys Pro Gly Gln 35 40 45Ser Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr
Arg Glu Ser Gly Val 50 55 60Pro Asp Arg Phe Thr Gly Ser Gly Ser Gly
Thr Asp Phe Thr Leu Thr65 70 75 80Ile Ser Ser Val Gln Ala Glu Asp
Leu Ala Val Tyr Tyr Cys Lys Gln 85 90 95Ser Tyr Asn Leu Leu Thr Phe
Gly Ala Gly Thr Lys Leu Glu Leu Lys 100 105 1106117PRTArtificial
SequenceSynthetic PeptideClone 1749.1.3 - CDR-L1 61Lys Ser Ser Arg
Ser Leu Leu Asn Ser Arg Ile Arg Lys Asn Tyr Leu1 5 10
15Ala627PRTArtificial SequenceSynthetic Peptideclone 1749.1.3 -
CDR-L2 62Trp Ala Ser Thr Arg Glu Ser1 5638PRTArtificial
SequenceSynthetic PeptideClone 1749.1.3 - CDR-L3 63Lys Gln Ser Tyr
Asn Leu Leu Thr1 564360DNAArtificial SequenceSynthetic
PolynucleotideClone 1749.1.3 - variable Heavy chainCDS(1)...(360)
64gac gtg aag ctg gtg gag tct ggg gga gac tta gtg aag cct gga ggg
48Asp Val Lys Leu Val Glu Ser Gly Gly Asp Leu Val Lys Pro Gly Gly 1
5 10 15tcc ctg aaa ctc tcc tgt gca gcc tct gga ttc act ttc agt agc
tat 96Ser Leu Lys Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser
Tyr 20 25 30atc atg tct tgg gtt cgt cag act ccg gag aag agg ctg gag
tgg gtc 144Ile Met Ser Trp Val Arg Gln Thr Pro Glu Lys Arg Leu Glu
Trp Val 35 40 45gca acc att agt agt ggt ggt agt tcc acc tac tat cca
gac agt gtg 192Ala Thr Ile Ser Ser Gly Gly Ser Ser Thr Tyr Tyr Pro
Asp Ser Val 50 55 60aag ggc cga ttc acc atc tcc aga gac aat gcc aag
aac acc ctg tac 240Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys
Asn Thr Leu Tyr 65 70 75 80ctg caa atg agc agt ctg aag tct gag gac
aca gcc atg tat tac tgt 288Leu Gln Met Ser Ser Leu Lys Ser Glu Asp
Thr Ala Met Tyr Tyr Cys 85 90 95aca aga gat gat gat tac gac gta aag
gta ttt gct tac tgg ggc caa 336Thr Arg Asp Asp Asp Tyr Asp Val Lys
Val Phe Ala Tyr Trp Gly Gln 100 105 110ggg act ctg gtc act gtc tct
gca 360Gly Thr Leu Val Thr Val Ser Ala 115 12065120PRTArtificial
SequenceSynthetic Construct 65Asp Val Lys Leu Val Glu Ser Gly Gly
Asp Leu Val Lys Pro Gly Gly1 5 10 15Ser Leu Lys Leu Ser Cys Ala Ala
Ser Gly Phe Thr Phe Ser Ser Tyr 20 25 30Ile Met Ser Trp Val Arg Gln
Thr Pro Glu Lys Arg Leu Glu Trp Val 35 40 45Ala Thr Ile Ser Ser Gly
Gly Ser Ser Thr Tyr Tyr Pro Asp Ser Val 50 55 60Lys Gly Arg Phe Thr
Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu Tyr65 70 75 80Leu Gln Met
Ser Ser Leu Lys Ser Glu Asp Thr Ala Met Tyr Tyr Cys 85 90 95Thr Arg
Asp Asp Asp Tyr Asp Val Lys Val Phe Ala Tyr Trp Gly Gln 100 105
110Gly Thr Leu Val Thr Val Ser Ala 115 1206610PRTArtificial
SequenceSynthetic PeptideClone 1749.1.3 - CDR-H1 66Gly Phe Thr Phe
Ser Ser Tyr Ile Met Ser1 5 106717PRTArtificial SequenceSynthetic
PeptideClone 1749.1.3 - CDR-H2 67Thr Ile Ser Ser Gly Gly Ser Ser
Thr Tyr Tyr Pro Asp Ser Val Lys1 5 10 15Gly6811PRTArtificial
SequenceSynthetic PeptideClone 1749.1.3 - CDR-H3 68Asp Asp Asp Tyr
Asp Val Lys Val Phe Ala Tyr1 5 1069319DNAArtificial
SequenceSynthetic PolynucleotideClone 2120.4.19 - variable Light
ChainCDS(1)...(318) 69gat atc cgg atg act cag tct cct tca ctc ctg
tct gca tct gtg ggg 48Asp Ile Arg Met Thr Gln Ser Pro Ser Leu Leu
Ser Ala Ser Val Gly 1 5 10 15gac aga gtc act ctc aac tgc aaa gca
agt cag aat att tat aac agc 96Asp Arg Val Thr Leu Asn Cys Lys Ala
Ser Gln Asn Ile Tyr Asn Ser 20 25 30tta
gcc tgg tat cag caa aag ctt gga gaa ggt ccc aaa gtc ctg att 144Leu
Ala Trp Tyr Gln Gln Lys Leu Gly Glu Gly Pro Lys Val Leu Ile 35 40
45ttt aat gca aac agt ttg caa acg ggc atc cca tca agg ttc agt ggc
192Phe Asn Ala Asn Ser Leu Gln Thr Gly Ile Pro Ser Arg Phe Ser Gly
50 55 60agt gga tct ggt aca gat ttc aca ctc acc atc agc agc ctg cag
cct 240Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro 65 70 75 80gaa gat ttt gcc aca tat ttc tgc cag cag ttt tat agc
ggg tac acg 288Glu Asp Phe Ala Thr Tyr Phe Cys Gln Gln Phe Tyr Ser
Gly Tyr Thr 85 90 95ttt gga gct ggg acc aag ctg gaa ctg aaa c
319Phe Gly Ala Gly Thr Lys Leu Glu Leu Lys 100
10570106PRTArtificial SequenceSynthetic Construct 70Asp Ile Arg Met
Thr Gln Ser Pro Ser Leu Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Leu Asn Cys Lys Ala Ser Gln Asn Ile Tyr Asn Ser 20 25 30Leu Ala
Trp Tyr Gln Gln Lys Leu Gly Glu Gly Pro Lys Val Leu Ile 35 40 45Phe
Asn Ala Asn Ser Leu Gln Thr Gly Ile Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Phe Cys Gln Gln Phe Tyr Ser Gly Tyr
Thr 85 90 95Phe Gly Ala Gly Thr Lys Leu Glu Leu Lys 100
10571106PRTArtificial SequenceSynthetic Peptideclone 2120.4.19 -
variable Light Chain 71Asp Ile Gln Val Thr Gln Ser Pro Ser Leu Leu
Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Leu Asn Cys Lys Ala Ser
Gln Asn Ile Tyr Asn Ser 20 25 30Leu Ala Trp Tyr Gln Gln Lys Leu Gly
Glu Gly Pro Lys Val Leu Ile 35 40 45Phe Asn Ala Asn Ser Leu Gln Thr
Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe
Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr
Tyr Phe Cys Gln Gln Phe Tyr Ser Gly Tyr Thr 85 90 95Phe Gly Ala Gly
Thr Lys Leu Glu Leu Lys 100 10572106PRTArtificial SequenceSynthetic
PeptideClone 2120.4.19 - variable Light chain 72Asp Ile Val Leu Thr
Gln Ser Pro Ser Leu Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr
Leu Asn Cys Lys Ala Ser Gln Asn Ile Tyr Asn Ser 20 25 30Leu Ala Trp
Tyr Gln Gln Lys Leu Gly Glu Gly Pro Lys Val Leu Ile 35 40 45Phe Asn
Ala Asn Ser Leu Gln Thr Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60Ser
Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75
80Glu Asp Phe Ala Thr Tyr Phe Cys Gln Gln Phe Tyr Ser Gly Tyr Thr
85 90 95Phe Gly Ala Gly Thr Lys Leu Glu Leu Lys 100
1057311PRTArtificial SequenceSynthetic PeptideClone 2120.4.19 -
CDR-L1 73Lys Ala Ser Gln Asn Ile Tyr Asn Ser Leu Ala1 5
10747PRTArtificial SequenceSynthetic PeptideClone 2120.4.19 -
CDR-L2 74Asn Ala Asn Ser Leu Gln Thr1 5758PRTArtificial
SequenceSynthetic PeptideClone 2120.4.19 - CDR-L3 75Gln Gln Phe Tyr
Ser Gly Tyr Thr1 576354DNAArtificial SequenceSynthetic
PolynucleotideClone 2120.4.19 - variable Heavy ChainCDS(1)...(354)
76cag gtg cag ctg aag gag tca gga cct ggt ctg gtg cag ccc tca cag
48Gln Val Gln Leu Lys Glu Ser Gly Pro Gly Leu Val Gln Pro Ser Gln 1
5 10 15acc ctg tct ctc acc tgc act gtc tct gga ttc tca tta acc agc
aat 96Thr Leu Ser Leu Thr Cys Thr Val Ser Gly Phe Ser Leu Thr Ser
Asn 20 25 30ggt gta agc tgg gtt cgc cag cct cca gga aag ggt ctg gag
tgg att 144Gly Val Ser Trp Val Arg Gln Pro Pro Gly Lys Gly Leu Glu
Trp Ile 35 40 45gca gca ata tca tct ggt gga acc aca tat tat aat tca
gcg ttc aaa 192Ala Ala Ile Ser Ser Gly Gly Thr Thr Tyr Tyr Asn Ser
Ala Phe Lys 50 55 60tcc cga ctg agc atc agc agg aac acc tcc aag agc
caa gtt ctc tta 240Ser Arg Leu Ser Ile Ser Arg Asn Thr Ser Lys Ser
Gln Val Leu Leu 65 70 75 80aaa atg aac agt ctg caa act gaa gac aca
gcc atg tac ttc tgt gcc 288Lys Met Asn Ser Leu Gln Thr Glu Asp Thr
Ala Met Tyr Phe Cys Ala 85 90 95aga cgg tat ggg tac ggg tgg tac ttt
gac ttc tgg ggc cca gga acc 336Arg Arg Tyr Gly Tyr Gly Trp Tyr Phe
Asp Phe Trp Gly Pro Gly Thr 100 105 110atg gtc aca gtc tcc tca
354Met Val Thr Val Ser Ser 11577118PRTArtificial SequenceSynthetic
Construct 77Gln Val Gln Leu Lys Glu Ser Gly Pro Gly Leu Val Gln Pro
Ser Gln1 5 10 15Thr Leu Ser Leu Thr Cys Thr Val Ser Gly Phe Ser Leu
Thr Ser Asn 20 25 30Gly Val Ser Trp Val Arg Gln Pro Pro Gly Lys Gly
Leu Glu Trp Ile 35 40 45Ala Ala Ile Ser Ser Gly Gly Thr Thr Tyr Tyr
Asn Ser Ala Phe Lys 50 55 60Ser Arg Leu Ser Ile Ser Arg Asn Thr Ser
Lys Ser Gln Val Leu Leu65 70 75 80Lys Met Asn Ser Leu Gln Thr Glu
Asp Thr Ala Met Tyr Phe Cys Ala 85 90 95Arg Arg Tyr Gly Tyr Gly Trp
Tyr Phe Asp Phe Trp Gly Pro Gly Thr 100 105 110Met Val Thr Val Ser
Ser 1157810PRTArtificial SequenceSynthetic PeptideClone 2120.4.19 -
CDR-H1 78Gly Phe Ser Leu Thr Ser Asn Gly Val Ser1 5
107916PRTArtificial SequenceSynthetic PeptideClone 2120.4.19 -
CDR-H2 79Ala Ile Ser Ser Gly Gly Thr Thr Tyr Tyr Asn Ser Ala Phe
Lys Ser1 5 10 158010PRTArtificial SequenceSynthetic PeptideClone
2120.4.19 - CDR-H3 80Arg Tyr Gly Tyr Gly Trp Tyr Phe Asp Phe1 5
1081318DNAArtificial SequenceSynthetic PolynucleotideClone
2107.4.10 - variable Light ChainCDS(1)...(318) 81gac atc cgg gtg
act cag tct cct tca ctc ctg tct gca tct gtg gga 48Asp Ile Arg Val
Thr Gln Ser Pro Ser Leu Leu Ser Ala Ser Val Gly 1 5 10 15gac aga
gtc act ctc aac tgc aaa gga agt cag aat att tat aag agc 96Asp Arg
Val Thr Leu Asn Cys Lys Gly Ser Gln Asn Ile Tyr Lys Ser 20 25 30tta
gcc tgg ttt cgg cta aag cgt gga gaa gct ccc aag ctc ctg att 144Leu
Ala Trp Phe Arg Leu Lys Arg Gly Glu Ala Pro Lys Leu Leu Ile 35 40
45tat gat gca aac agt ttg caa acg ggc atc cca tca agg ttc agt ggc
192Tyr Asp Ala Asn Ser Leu Gln Thr Gly Ile Pro Ser Arg Phe Ser Gly
50 55 60agt gga tct ggt aca gat ttc aca ctc acc atc acc agc cta cag
cct 240Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Thr Ser Leu Gln
Pro 65 70 75 80gaa gat gtt gcc aca tat ttc tgc cag cag tat tat agc
ggt tac acg 288Glu Asp Val Ala Thr Tyr Phe Cys Gln Gln Tyr Tyr Ser
Gly Tyr Thr 85 90 95ttt gga gct ggg acc aag ctg gaa ctg aaa 318Phe
Gly Ala Gly Thr Lys Leu Glu Leu Lys 100 10582106PRTArtificial
SequenceSynthetic Construct 82Asp Ile Arg Val Thr Gln Ser Pro Ser
Leu Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Leu Asn Cys Lys
Gly Ser Gln Asn Ile Tyr Lys Ser 20 25 30Leu Ala Trp Phe Arg Leu Lys
Arg Gly Glu Ala Pro Lys Leu Leu Ile 35 40 45Tyr Asp Ala Asn Ser Leu
Gln Thr Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Thr Ser Leu Gln Pro65 70 75 80Glu Asp Val
Ala Thr Tyr Phe Cys Gln Gln Tyr Tyr Ser Gly Tyr Thr 85 90 95Phe Gly
Ala Gly Thr Lys Leu Glu Leu Lys 100 10583318DNAArtificial
SequenceSynthetic PolynucleotideClone 2107.4.10 - variable Light
ChainCDS(1)...(318) 83gac atc cag gtg act cag tct cct tca ctc ctg
tct gca tct gtg gga 48Asp Ile Gln Val Thr Gln Ser Pro Ser Leu Leu
Ser Ala Ser Val Gly 1 5 10 15gac aga gtc act ctc aac tgc aaa gga
agt cag aat att tat aag agc 96Asp Arg Val Thr Leu Asn Cys Lys Gly
Ser Gln Asn Ile Tyr Lys Ser 20 25 30tta gcc tgg ttt cgg cta aag cgt
gga gaa gct ccc aag ctc ctg att 144Leu Ala Trp Phe Arg Leu Lys Arg
Gly Glu Ala Pro Lys Leu Leu Ile 35 40 45tat gat gca aac agt ttg caa
acg ggc atc cca tca agg ttc agt ggc 192Tyr Asp Ala Asn Ser Leu Gln
Thr Gly Ile Pro Ser Arg Phe Ser Gly 50 55 60agt gga tct ggt aca gat
ttc aca ctc acc atc acc agc cta cag cct 240Ser Gly Ser Gly Thr Asp
Phe Thr Leu Thr Ile Thr Ser Leu Gln Pro 65 70 75 80gaa gat gtt gcc
aca tat ttc tgc cag cag tat tat agc ggt tac acg 288Glu Asp Val Ala
Thr Tyr Phe Cys Gln Gln Tyr Tyr Ser Gly Tyr Thr 85 90 95ttt gga gct
ggg acc aag ctg gaa ctg aaa 318Phe Gly Ala Gly Thr Lys Leu Glu Leu
Lys 100 10584106PRTArtificial SequenceSynthetic Construct 84Asp Ile
Gln Val Thr Gln Ser Pro Ser Leu Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Leu Asn Cys Lys Gly Ser Gln Asn Ile Tyr Lys Ser 20 25
30Leu Ala Trp Phe Arg Leu Lys Arg Gly Glu Ala Pro Lys Leu Leu Ile
35 40 45Tyr Asp Ala Asn Ser Leu Gln Thr Gly Ile Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Thr Ser Leu
Gln Pro65 70 75 80Glu Asp Val Ala Thr Tyr Phe Cys Gln Gln Tyr Tyr
Ser Gly Tyr Thr 85 90 95Phe Gly Ala Gly Thr Lys Leu Glu Leu Lys 100
1058511PRTArtificial SequenceSynthetic PeptideClone 2107.4.10 -
CDR-L1 85Lys Gly Ser Gln Asn Ile Tyr Lys Ser Leu Ala1 5
10867PRTArtificial SequenceSynthetic PeptideClone 2107.4.10 -
CDR-L2 86Asp Ala Asn Ser Leu Gln Thr1 5878PRTArtificial
SequenceSynthetic PeptideClone 2107.4.10 - CDR-L3 87Gln Gln Tyr Tyr
Ser Gly Tyr Thr1 588348DNAArtificial SequenceSynthetic
PolynucleotideClone 2107.4.10 - variable Heavy ChainCDS(1)...(348)
88cag gtg cag ctg aag gag tca gga cct ggt ctg gtg cag tcc tca cag
48Gln Val Gln Leu Lys Glu Ser Gly Pro Gly Leu Val Gln Ser Ser Gln 1
5 10 15acc ctg tct ctc acc tgc act gtc tct gga ttc tca tta acc agt
aat 96Thr Leu Ser Leu Thr Cys Thr Val Ser Gly Phe Ser Leu Thr Ser
Asn 20 25 30ggt gta agc tgg gtt cgc cag cct cca gga aag ggt ctg gag
tgg att 144Gly Val Ser Trp Val Arg Gln Pro Pro Gly Lys Gly Leu Glu
Trp Ile 35 40 45gca gca ata tca agt ggt gga agc aca tat tat aat tca
gcg ttc aaa 192Ala Ala Ile Ser Ser Gly Gly Ser Thr Tyr Tyr Asn Ser
Ala Phe Lys 50 55 60tcc cga ctg agc atc agc agg aac acc tcc aag agc
caa gtt ctc tta 240Ser Arg Leu Ser Ile Ser Arg Asn Thr Ser Lys Ser
Gln Val Leu Leu 65 70 75 80aaa atg aac agt ctg caa act gaa gac aca
ggc atg tac ttc tgt gcc 288Lys Met Asn Ser Le
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016
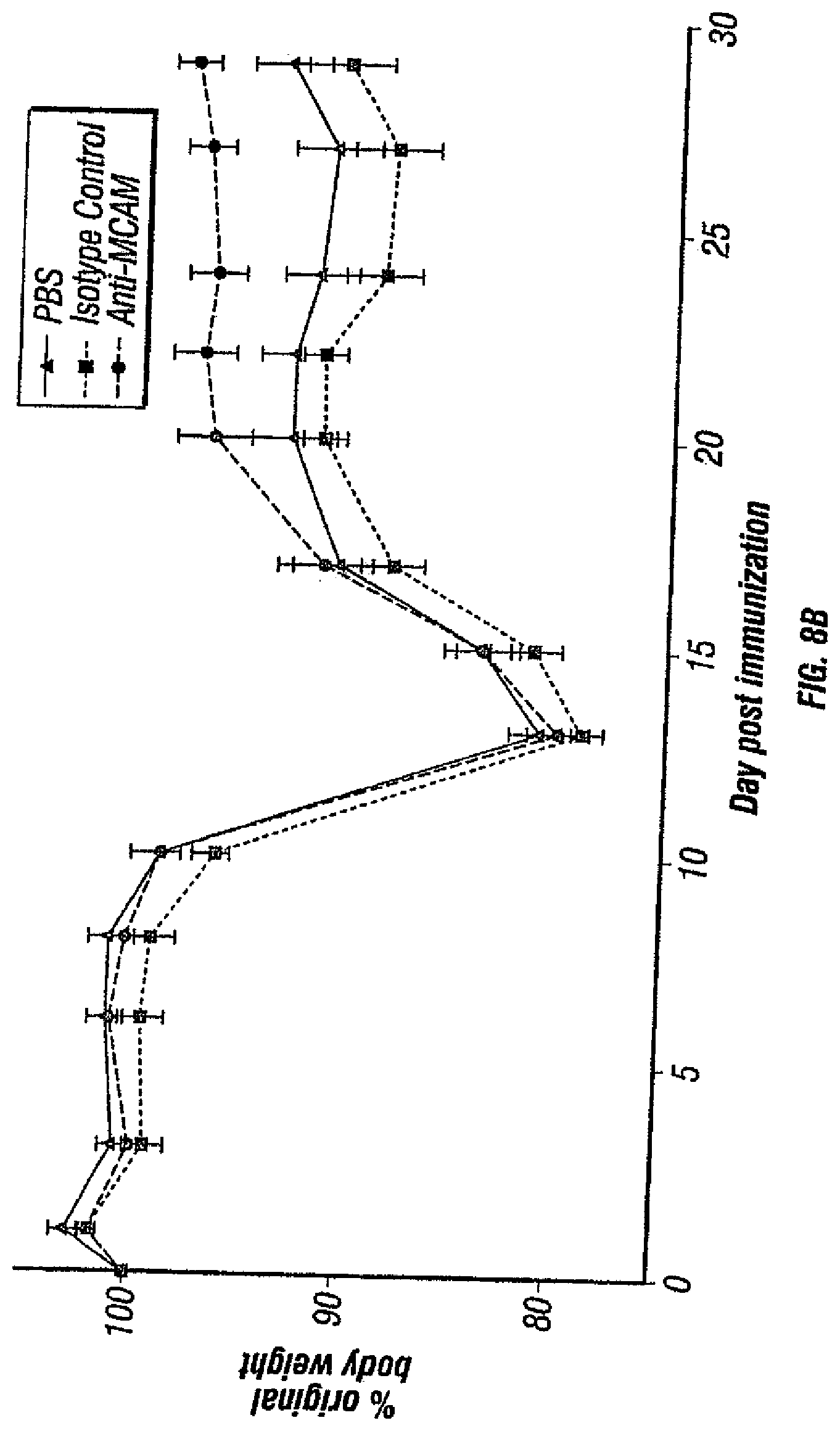
D00017

D00018

D00019

D00020

D00021

D00022
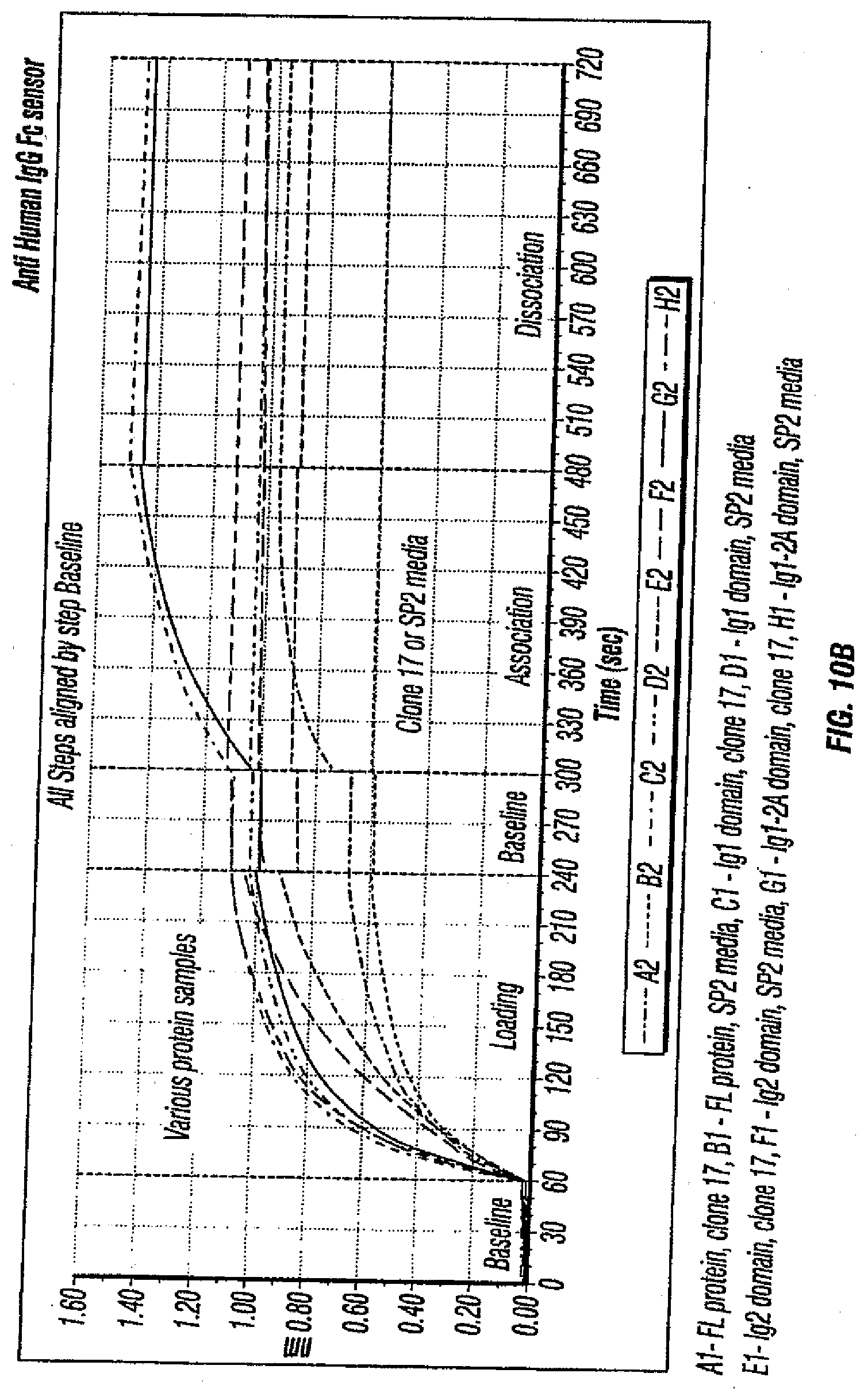
D00023

D00024

D00025

D00026

D00027

D00028

D00029

D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037

D00038

D00039

D00040

D00041

D00042

D00043

D00044

D00045
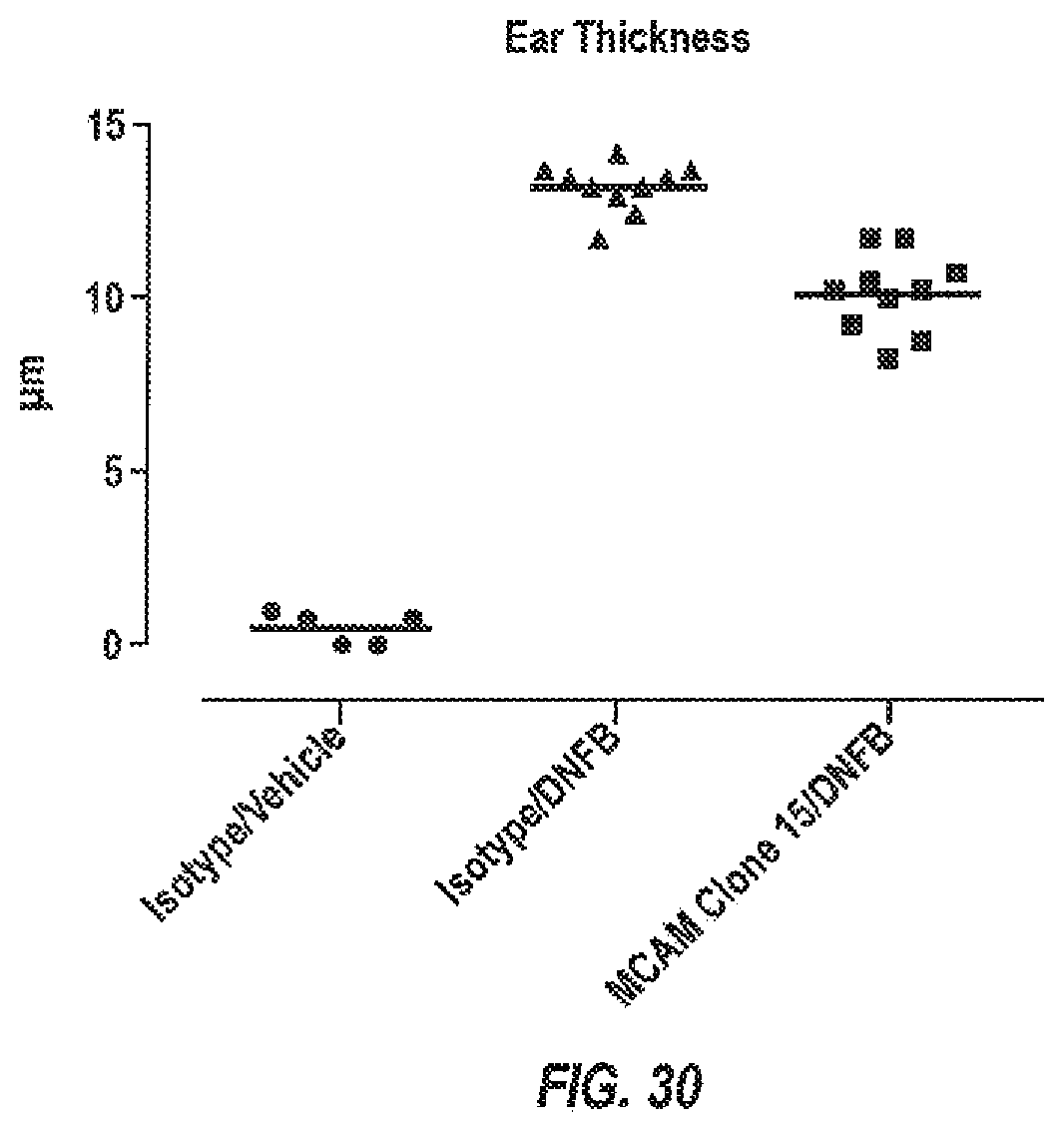
D00046

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.