Composite Fiber And Textile
KUO; Yi-Chun ; et al.
U.S. patent application number 16/531677 was filed with the patent office on 2020-07-02 for composite fiber and textile. This patent application is currently assigned to INDUSTRIAL TECHNOLOGY RESEARCH INSTITUTE. The applicant listed for this patent is INDUSTRIAL TECHNOLOGY RESEARCH INSTITUTE. Invention is credited to Yung-Pin HUANG, Yi-Chun KUO, Kuo-Hsing LEE.
| Application Number | 20200208304 16/531677 |
| Document ID | / |
| Family ID | 71122657 |
| Filed Date | 2020-07-02 |


| United States Patent Application | 20200208304 |
| Kind Code | A1 |
| KUO; Yi-Chun ; et al. | July 2, 2020 |
COMPOSITE FIBER AND TEXTILE
Abstract
A composite fiber is provided, which includes a polymer fiber, doped zinc oxide particles dispersed in the polymer fiber or combined with the polymer fiber, and a fluorescent dye combined with the polymer fiber. The light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap. The above composite fiber can be manufactured as a yarn and woven into a textile.
| Inventors: | KUO; Yi-Chun; (Zhubei City, TW) ; HUANG; Yung-Pin; (New Taipei City, TW) ; LEE; Kuo-Hsing; (Guanxi Township, TW) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | INDUSTRIAL TECHNOLOGY RESEARCH
INSTITUTE Hsinchu TW |
||||||||||
| Family ID: | 71122657 | ||||||||||
| Appl. No.: | 16/531677 | ||||||||||
| Filed: | August 5, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62784937 | Dec 26, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | D10B 2211/01 20130101; D10B 2201/28 20130101; D01F 1/106 20130101; D06M 11/45 20130101; D01F 1/04 20130101; D01F 6/70 20130101; D06M 11/46 20130101; D01F 2/28 20130101; D01F 1/06 20130101; D01F 6/60 20130101; D10B 2321/021 20130101; D01F 6/06 20130101; D01F 6/62 20130101; D06M 11/83 20130101; C08K 2003/2296 20130101; D10B 2321/022 20130101; D10B 2321/08 20130101; D10B 2331/10 20130101; D10B 2331/02 20130101 |
| International Class: | D01F 1/10 20060101 D01F001/10; D01F 6/06 20060101 D01F006/06; D01F 6/70 20060101 D01F006/70; D01F 6/62 20060101 D01F006/62; D01F 6/60 20060101 D01F006/60; D01F 2/28 20060101 D01F002/28; D01F 1/06 20060101 D01F001/06; D06M 11/46 20060101 D06M011/46; D06M 11/45 20060101 D06M011/45; D06M 11/83 20060101 D06M011/83 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jun 11, 2019 | TW | 108120085 |
Claims
1. A composite fiber, comprising: a polymer fiber, a plurality of doped zinc oxide particles dispersed in the polymer fiber or combined with the polymer fiber; and a fluorescent dye combined with the polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
2. The composite fiber as claimed in claim 1, wherein the polymer fiber comprises polyester fiber, polyacrylonitrile fiber, polyacrylate fiber, cellulose fiber, polyethylene fiber, polypropylene fiber, polyamide fiber, polyurethane fiber, cellulose acetate fiber, animal fiber, or a combination thereof.
3. The composite fiber as claimed in claim 1, wherein the doped zinc oxide particles are doped with aluminum, gallium, tin, or a combination thereof, and (a) weight of aluminum, gallium, tin, or a combination thereof and (b) total weight of zinc and aluminum, gallium, tin, or a combination thereof have a ratio of 0.1:100 to 20:100.
4. The composite fiber as claimed in claim 1, wherein the doped zinc oxide particles and the composite fiber have a weight ratio of 0.1:99.9 to 20:80.
5. The composite fiber as claimed in claim 1, further comprising a plurality of titanium oxide particles dispersed in the polymer fiber or combined with the polymer fiber, and the total weight of the titanium oxide particles and the doped zinc oxide particles and the weight of the composite fiber have a ratio of 0.1:99.9 to 20:80.
6. The composite fiber as claimed in claim 1, wherein the fluorescent dye has a light absorption wavelength of 180 nm to 600 nm, and the doped zinc oxide particles have a light emission wavelength of 400 nm to 780 nm.
7. A textile, comprising: a composite fiber, including: a first polymer fiber, a plurality of doped zinc oxide particles dispersed in the first polymer fiber or combined with the first polymer fiber; and a fluorescent dye combined with the first polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
8. The textile as claimed in claim 7, further comprising a second polymer fiber.
9. The textile as claimed in claim 8, wherein the first polymer fiber is different from the second polymer fiber.
10. A textile, comprising: a first polymer fiber and a second polymer fiber; a plurality of doped zinc oxide particles dispersed in the first polymer fiber or combined with the first polymer fiber; and a fluorescent dye combined with the second polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
11. The textile as claimed in claim 10, wherein the first polymer fiber is different from the second polymer fiber.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application No. 62/784,937, filed on Dec. 26, 2018, the entirety of which is incorporated by reference herein. The application is based on, and claims priority from, Taiwan Application Serial Number 108120085, filed on Jun. 11, 2019, the disclosure of which is hereby incorporated by reference herein in its entirety.
TECHNICAL FIELD
[0002] The technical field relates to a textile, and in particular it relates to a fiber composition thereof.
BACKGROUND
[0003] It is well known that the specific range of the visible light wavelength (400 nm to 780 nm) in sunlight is beneficial for human skin, exhibiting such effects as anti-aging, whitening, wrinkle reduction, collagen hyperplasia, and the like. For example, the visible light wavelength of 480 nm to 530 nm may accelerate blood circulation and activate cell, and the visible light wavelength of 600 nm to 750 nm may stimulate cytochrome activation of fibroblasts to help generate collagen. However, the other wavelength range in sunlight such as 180 nm to 400 nm (i.e. UV), which can be classified into UV-A (with wavelength of 320 nm to 400 nm), UV-B (with wavelength of 260 nm to 320 nm), and UV-C (with wavelength of 180 nm to 290 nm). UV-A may penetrate the dermis to age and relax the dermis, thereby forming wrinkles. UV-B results in erythema in skin and severe edema, blisters, and peeling. UV-C has shorter wavelength and potential carcinogenicity. Developing a composite fiber to simultaneously block UV, shield near-IR, and enhance the transmittance of visible light with a specific light wavelength is the future direction of the textile industry.
SUMMARY
[0004] One embodiment of the disclosure provides a composite fiber, including a polymer fiber, a plurality of doped zinc oxide particles dispersed in the polymer fiber or combined with the polymer fiber; and a fluorescent dye combined with the polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
[0005] One embodiment of the disclosure provides a textile, including a composite fiber which includes a first polymer fiber, a plurality of doped zinc oxide particles dispersed in the first polymer fiber or combined with the first polymer fiber; and a fluorescent dye combined with the first polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
[0006] One embodiment of the disclosure provides a textile, comprising: a first polymer fiber and a second polymer fiber; a plurality of doped zinc oxide particles dispersed in the first polymer fiber or combined with the first polymer fiber; and a fluorescent dye combined with the second polymer fiber, wherein the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap.
[0007] A detailed description is given in the following embodiments with reference to the accompanying drawings.
BRIEF DESCRIPTION OF THE DRAWINGS
[0008] The disclosure can be more fully understood by reading the subsequent detailed description and examples with references made to the accompanying drawings, wherein:
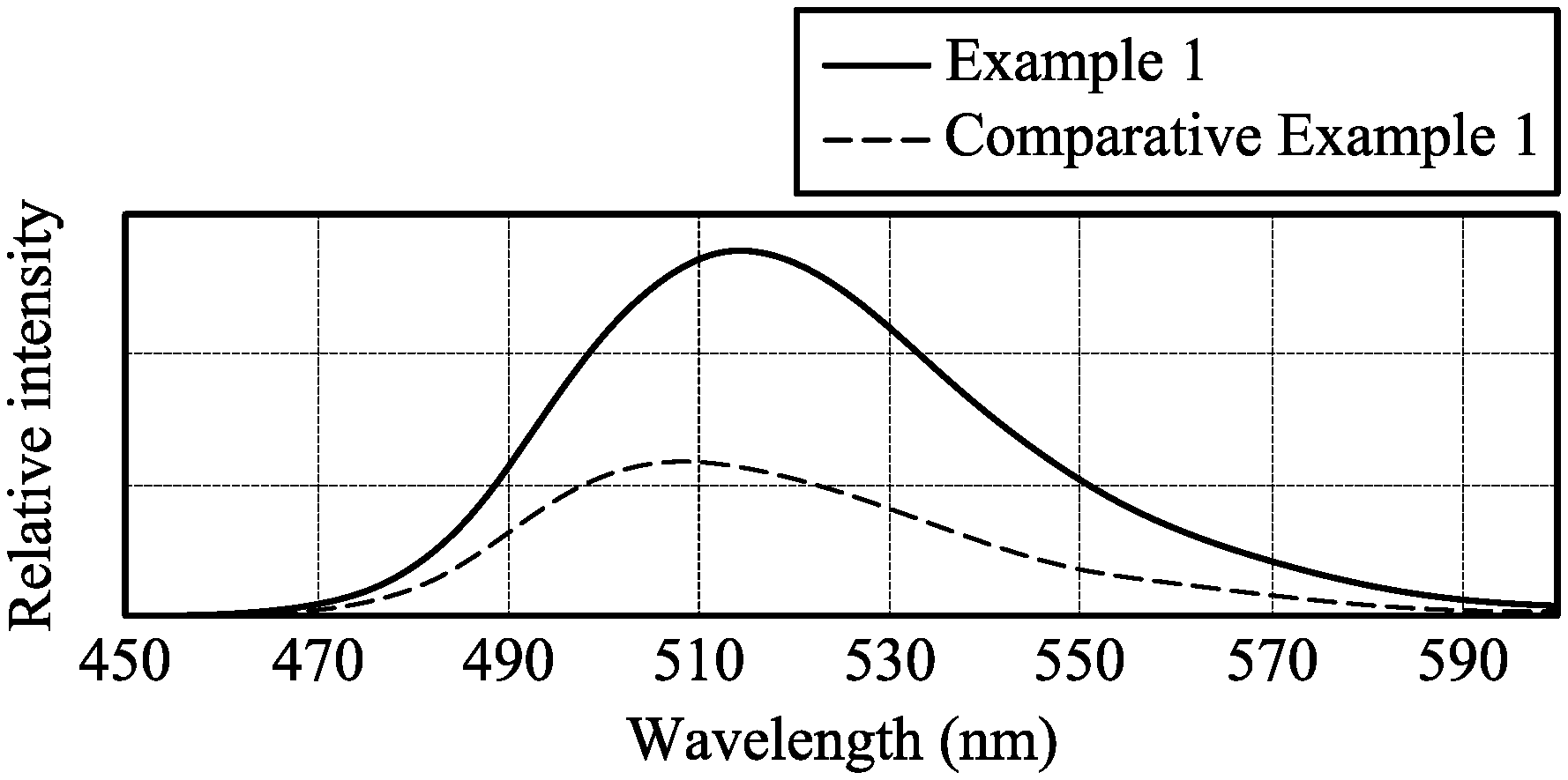
[0009] FIG. 1 shows fluorescent spectroscopic spectrum of textiles of examples.
[0010] FIG. 2 shows visible light transmittance spectrum of textiles of examples.
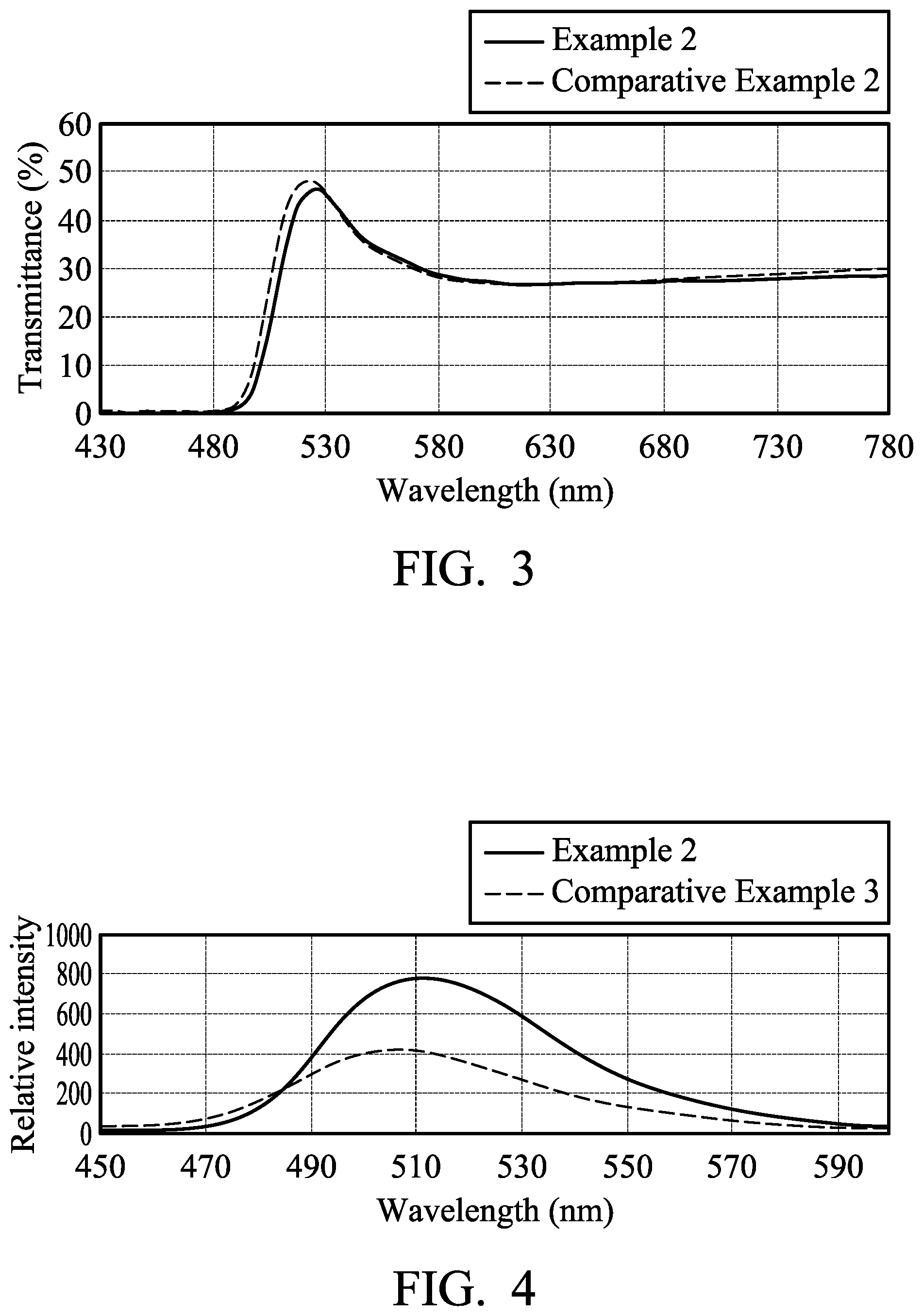
[0011] FIG. 3 shows visible light transmittance spectrum of textiles of examples.
[0012] FIG. 4 shows fluorescent spectroscopic spectrum of textiles of examples.
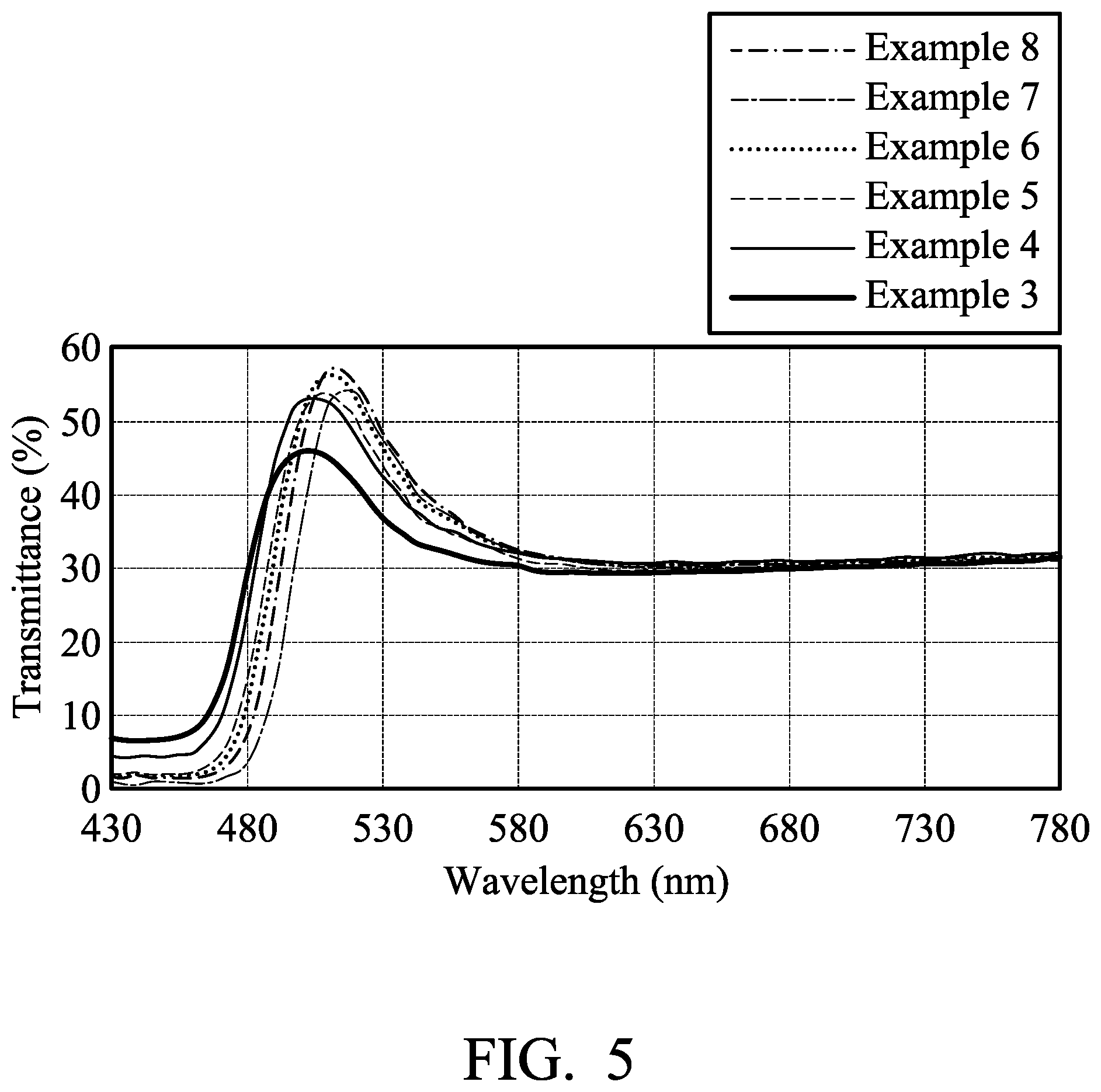
[0013] FIG. 5 shows visible light transmittance spectrum of textiles of examples.
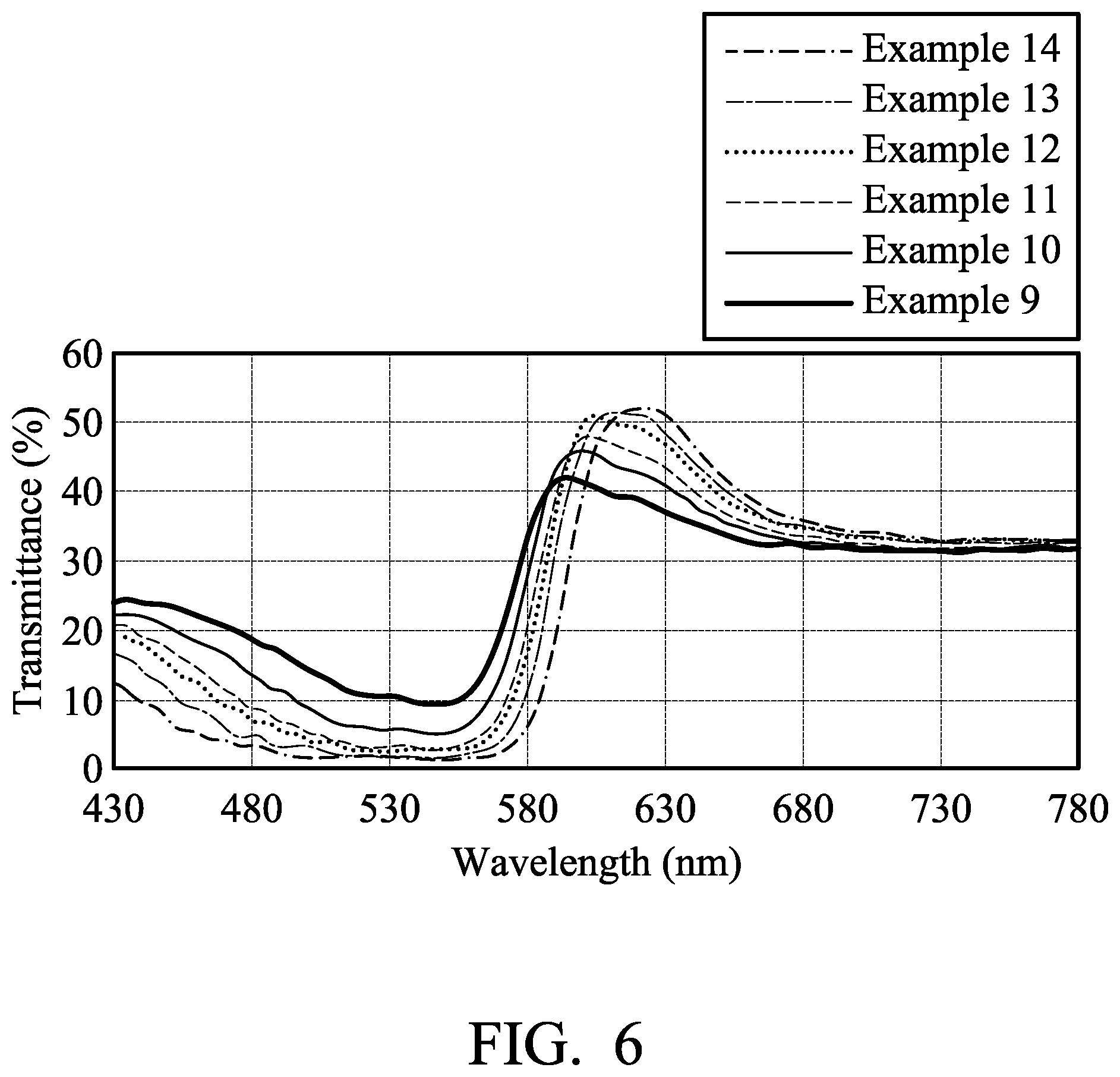
[0014] FIG. 6 shows visible light transmittance spectrum of textiles of examples.
[0015] FIG. 7 shows visible light transmittance spectrum of textiles of examples.
[0016] FIG. 8 shows fluorescent spectroscopic spectrum of textiles of examples.
[0017] FIG. 9 shows fluorescent spectroscopic spectrum of textiles of examples.
DETAILED DESCRIPTION
[0018] In the following detailed description, for purposes of explanation, numerous specific details are set forth in order to provide a thorough understanding of the disclosed embodiments. It will be apparent, however, that one or more embodiments may be practiced without these specific details. In other instances, well-known structures and devices are schematically shown in order to simplify the drawing.
[0019] Because humans wear clothing in most conditions, it is beneficial to human health to wear clothing that may block harmful light wavelength range and strengthen the wholesome light wavelength range. One embodiment of the disclosure provides a textile, in which the ratio of the visible light with specific light wavelength range transmitting the textile is enhanced.
[0020] For example, one embodiment of the disclosure provides a composite fiber including a polymer fiber, doped zinc oxide particles, and fluorescent dye. The doped zinc oxide particles is dispersed in the polymer fiber or combined with the polymer fiber. For example, the doped zinc oxide particles can be dispersed in the polymer, and then melt spun and false twisted to form a yarn. The yarn is then woven to form a textile such as cloth. As such, the doped zinc oxide particles are dispersed in the polymer fiber. The polymer fiber with the doped zinc oxide particles dispersed therein can be spun to form a yarn, and the yarn can be woven to form a textile. Then, the yarn or the textile can be soaked in an aqueous solution of the fluorescent dye, such that the fluorescent dye is combined with the polymer fiber. On the other hand, the fluorescent dye and the doped zinc oxide particles are dispersed in a solution, and the yarn or the textile (spun or woven from a polymer fiber) are soaked in the dispersion, such that the doped zinc oxide particles and the fluorescent dye are combined with the polymer fiber. No matter what the method is selected to form the composite fiber, the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap. For example, the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye are partially overlapped. For example, the doped zinc oxide particles have a light emission wavelength of 400 nm to 780 nm, and the fluorescent dye has a light absorption wavelength of 180 nm to 600 nm. If the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye do not overlap, the emission light intensity of a combination of the fluorescent dye and the doped zinc oxide particles will be similar to the emission light intensity of the fluorescent dye alone. The doped zinc oxide may block most of UV and IR and emit light of specific wavelength. The fluorescent dye may absorb the light emitted by the doped zinc oxide particles, and then enhance the emission of visible light of a specific wavelength.
[0021] In one embodiment, the polymer fiber includes polyester fiber, polyacrylonitrile fiber, polyacrylate fiber, cellulose fiber, polyethylene fiber, polypropylene fiber, polyamide fiber, polyurethane fiber, cellulose acetate fiber, animal fiber, or a combination thereof. The composite fiber formed from the polymer fiber can be spun to a yarn with other polymer fiber (without the doped zinc oxide particles and the fluorescent dye), and the yarn is woven to form a textile such as cloth.
[0022] In one embodiment, the doped zinc oxide particles can be doped aluminum, gallium, tin, or a combination thereof, and (a) weight of aluminum, gallium, tin, or a combination thereof and (b) total weight of zinc and aluminum, gallium, tin, or a combination thereof have a ratio of 0.1:100 to 20:100. If (a) weight of aluminum, gallium, tin, or a combination thereof has an overly high or overly low ratio, the appearance of the composite fiber, the yarn containing the composite fiber, and the textile containing the composite fiber has an overly high haze degree due to overly low transmittance. The doped zinc oxide particles can be prepared by the following steps. The nitrate or sulfate of zinc and chloride or sulfate of doping element (containing aluminum, gallium, or tin) are formulated to a mixture solution with a concentration of 0.5 mL/L to 5.0 mL/L. The weight of the doping element and the total weight of zinc and the doping element may have a ratio of 0.1% to 20%, such as 0.1% to 10%. The mixture solution and the ammonium bicarbonate are dropwise added into water to obtain evenly doped and white alkaline zinc carbonate precipitation. During the dropwise addition, the temperature is kept at 40.degree. C., the pH value is controlled at 7.0 to 7.5, and the solution is vigorously stirred. The precipitation is washed, separated, and baking dried to obtain powders. The powders are sintered under a mixture gas of hydrogen and argon at a temperature of 400.degree. C. to 700.degree. C. for a period of 30 minutes to 60 minutes to obtain doped zinc oxide particles that are doped with aluminum, gallium, tin, or a combination thereof. The doped zinc oxide particles may have a diameter of 50 nm to 1000 nm, which can be tuned by the sintering temperature and sintering period. Overly large doped zinc oxide particles are difficult to spin to form the polymer fiber with the doped zinc oxide particles dispersed therein. On the other hand, the overly large zinc oxide particles may negatively influence the transmittance of the composite fiber and even the breathability of the textile. Overly small doped zinc oxide are easily aggregated, which are difficult to be evenly dispersed in the polymer fiber or evenly combined with the polymer fiber.
[0023] In one embodiment, the doped zinc oxide particles and the composite fiber have a weight ratio of 0.1:99.9 to 20:80. Alternatively, the composite fiber further includes a plurality of titanium oxide particles dispersed in the polymer fiber or combined with the polymer fiber, and the total weight of the titanium oxide particles and the doped zinc oxide particles and the weight of the composite fiber have a weight ratio of 0.1:99.9 to 20:80. The amount of the doped zinc oxide (or the doped zinc oxide particles and the titanium oxide particles) that is too low cannot efficiently prevent UV and IR from transmitting through the textile. The amount of the doped zinc oxide ratio (or the doped zinc oxide particles and the titanium oxide particles) that is too high may increase the textile weight and reduce the breathability of the textile.
[0024] In general, the fluorescent dye has a light absorption wavelength of 180 nm to 600 nm and a light emission wavelength of 400 nm to 780 nm. For example, the fluorescent dye can be 1,8-naphthalimide (for cotton fiber, cellulose fiber, or wool fiber), coumarin (for polyester fiber), or hemicyanine (for polyacrylonitrile fiber, silk, wool, or nylon fiber). As described above, the light emission wavelength can be controlled by fluorescent dye to achieve the effects on human skin, such as anti-aging, whitening, wrinkle reduction, collagen hyperplasia, and the like. In one embodiment, the fluorescent dye and the composite fiber have a weight ratio of 0.01:100 to 20:100. The amount of the fluorescent dye that is too low cannot efficiently enhance the emission intensity of the specific visible light. The amount of the fluorescent dye that is too high cannot further increase the emission intensity of the specific visible light but increase the cost.
[0025] The composite fiber can be used to form a textile. For example, the composite fiber can be used to fabricate a yarn, and the yarn is then woven to form a textile such as cloth. In one embodiment, the composite fiber can be used to fabricate a yarn with other polymer fiber (without the doped zinc oxide particles and/or the fluorescent dye), and the yarn is then woven to form a textile such as cloth. On the other hand, the other polymer fiber can be directly used to prepare the other yarn at first, and then the yarn (containing the composite fiber) and the other yarn are woven to form a textile such as cloth. The composition of the other polymer fiber can be similar to or different from the composition of the polymer fiber of the composite fiber. For example, the melt liquid of polyester and the doped zinc oxide can be extruded from spinning nozzle to form a yarn. The yarn is then stretched and cooled, and wound as caked to fabricate synthetic fiber. On the other hand, several pieces of processing equipment or nozzle devices can be utilized to develop texture yarn such as wool-like, silk-like, cotton-like, flax-like, slub-like, variegated wool-like, cloud yarn-like, thick and thin yarn-like, or the like.
[0026] In one embodiment, the polymer fiber with the doped zinc oxide particles dispersed therein can be dyed to form a composite fiber. The composite fiber can be used to form a yarn, and the yarn is woven to form a textile such as cloth. Alternatively, the polymer fiber with the doped zinc oxide particles dispersed therein can be used to form a yarn. The yarn is dyed and then woven to form a textile such as cloth. Alternatively, the polymer fiber with the doped zinc oxide particles dispersed therein can be used to form a yarn, and the yarn is woven to form a textile such as cloth. The textile is then dyed. The above mentioned fiber, yarn, or textile can be dyed by sublimation method, water bath method, or coating method. For example, the sublimation method may include put the fluorescent dye in a crucible, put the fiber, the yarn, or the textile above the crucible, and heat the crucible to sublimate the fluorescent dye, such that the fluorescent dye vapor is combined with the polymer fiber to form the composite fiber. For example, the water bath method may include dissolve the fluorescent dye in water, and soaking the fiber, the yarn, or the textile into the aqueous solution, such that the fluorescent dye is combined with the polymer fiber to form the composite fiber. Alternatively, the fluorescent dye is dissolved in a solution, and the solution is coated to the fiber, the yarn, or the textile, such that the fluorescent dye is combined with the polymer fiber to form the composite fiber. In one embodiment, the doped zinc oxide particles and the fluorescent dye can be directly dispersed in a solution, and the dispersion is coated to the polymer fiber (free of the doped zinc oxide particles and the fluorescent dye before coating), the yarn containing the polymer fiber, or the textile containing the polymer fiber, such that the doped zinc oxide particles and the fluorescent dye are combined with the polymer fiber. No matter what method is selected for dyeing, the fluorescent dye can be combined with the polymer fiber, and the doped zinc oxide particles can be dispersed in the polymer fiber or combined with the polymer fiber. The light emission wavelength of the doped zinc oxide particles in the composite fiber, the yarn, and the textile and the light absorption wavelength of the fluorescent dye overlap to further enhance the emission intensity of the specific visible light wavelength.
[0027] In one embodiment, the doped zinc oxide particles can be dispersed in the first polymer fiber or combined with the first polymer fiber. On the other hand, the fluorescent dye can be combined with the second polymer fiber. The first polymer fiber (containing the doped zinc oxide particles) and the second polymer fiber (containing the fluorescent dye) can be spun together to form the same yarn, or respectively spun to form the different yarns. The yarns can be further woven to form a textile such as cloth. In one embodiment, each of the first polymer fiber and the second polymer fiber is independently polyester fiber, polyacrylonitrile fiber, polyacrylate fiber, cellulose fiber, polyethylene fiber, polypropylene fiber, polyamide fiber, polyurethane fiber, cellulose acetate fiber, animal fiber, or a combination thereof. The first polymer fiber can be similar to or different from the second polymer fiber. Whatever, the light emission wavelength of the doped zinc oxide particles and the light absorption wavelength of the fluorescent dye overlap to further enhance the emission intensity of specific visible light wavelength.
[0028] Below, exemplary embodiments will be described in detail with reference to accompanying drawings so as to be easily realized by a person having ordinary knowledge in the art. The inventive concept may be embodied in various forms without being limited to the exemplary embodiments set forth herein. Descriptions of well-known parts are omitted for clarity, and like reference numerals refer to like elements throughout.
EXAMPLES
Preparation Example 1
Preparation of Aluminum Doped Zinc Oxide Particles
[0029] Zinc nitrate and aluminum chloride were formulated to form a mixture solution with a concentration of 1 mL/L, wherein the addition weight of aluminum and the total weight of zinc and aluminum have a ratio of 1:100. Both the mixture solution and ammonium bicarbonate were dropwise added into water to obtain evenly doped and white alkaline zinc carbonate precipitation. During the dropwise addition, the temperature was kept at 40.degree. C., the pH value was controlled at 7.0 to 7.5, and the solution was vigorously stirred. The precipitation was washed, separated, and baking dried to obtain powders. The powders were sintered under a mixture gas of hydrogen and argon at a temperature of 400.degree. C. to 700.degree. C. for a period of 30 minutes to 60 minutes to obtain aluminum doped zinc oxide particles. The aluminum doped zinc oxide particles had a diameter of 50 nm to 100 nm, which could be tuned by modifying the sintering temperature and period. For example, the particles with a diameter of 50 nm to 150 nm could be obtained by sintering at a temperature of 400.degree. C. for a period of 60 minutes. The particles with a diameter of larger than 500 nm could be obtained by sintering at a temperature of 700.degree. C. for a period of 30 minutes.
Preparation Example 2
Preparation of Gallium Doped Zinc Oxide Particles
[0030] The gallium doped zinc oxide particles were prepared by the steps as Preparation Example 1, and the difference was the aluminum chloride being replaced with gallium chloride.
Example 1
[0031] 1 part by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm) were dispersed in 99 parts by weight of polyethylene terephthalate (PET). Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0032] 0.7 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile had a UV proof of 50+, which was measured by the standard FTTS-FA-008. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 1. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 2.
Comparative Example 1
[0033] 2.4 parts by weight of titanium oxide particles (with a diameter of 50 nm to 150 nm) were dispersed in 97.6 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile. 0.7 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile had a UV proof of 50+, which was measured by the standard FTTS-FA-008. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 1. Compared to Comparative Example 1, the yellow textile in Example 1 had higher emission intensity, and its light emission wavelength was red-shift, as shown in FIG. 1. Furthermore, the yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 2. Compared to Comparative Example 1, the yellow textile in Example 1 has a higher transmittance for visible light with specific wavelength, and the transmitting visible light wavelength was red-shift, as shown in FIG. 2.
Example 2
[0034] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0035] 0.7 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile had a UV proof of 50+, which was measured by the standard FTTS-FA-008. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 3. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 4.
Comparative Example 2
[0036] 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 99.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0037] 0.7 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile had a UV proof of 10 to 20, which was measured by the standard FTTS-FA-008. Compared to Example 2, the yellow textile in Comparative Example 2 had obviously lower UV proof property. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 3. The yellow textile in Example 2 and the yellow textile in Comparative Example 2 had similar transmittance for visible light with specific wavelength. Moreover, the transmitting visible light wavelength of the yellow textile in Example 2 was red-shift, as shown in FIG. 3.
Comparative Example 3
[0038] 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 99.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0039] 0.7 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 4. Compared to Comparative Example 3, the yellow textile in Example 2 had higher emission intensity, and its light emission wavelength was red-shift, as shown in FIG. 4.
Example 3
[0040] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0041] 0.05 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5.
Example 4
[0042] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0043] 0.1 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5.
Example 5
[0044] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0045] 0.2 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5.
Example 6
[0046] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0047] 0.3 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5.
Example 7
[0048] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0049] 0.5 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5.
Example 8
[0050] 0.7 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm), 0.3 parts by weight of gallium doped zinc oxide particles (with a diameter of 50 nm to 150 nm), and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0051] 1 part by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 5. When the dye amount achieved 0.5 parts by weight, the yellow textile had the highest transmittance for specific visible light wavelength, it means that the fluorescent emission of the yellow textile was strongest. When the dye amount achieved 1 part by weight, the dye was aggregated to result in fluorescent quench.
Example 9
[0052] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0053] 0.05 parts by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6.
Example 10
[0054] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0055] 0.1 parts by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6.
Example 11
[0056] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0057] 0.2 parts by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6.
Example 12
[0058] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0059] 0.3 parts by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6.
Example 13
[0060] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0061] 0.5 parts by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6.
Example 14
[0062] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0063] 1 part by weight of pink fluorescent coumarin dye (5B, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a pink textile. The pink textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 6. When the dye amount achieved 0.3 parts by weight, the pink textile had the highest transmittance for specific visible light wavelength, it means that the fluorescent emission of the pink textile was strongest.
Example 15
[0064] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0065] 0.05 parts by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7.
Example 16
[0066] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0067] 0.1 parts by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7.
Example 17
[0068] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0069] 0.2 parts by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7.
Example 18
[0070] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0071] 0.3 parts by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7.
Example 19
[0072] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0073] 0.5 parts by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7.
Example 20
[0074] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0075] 1 part by weight of magenta fluorescent coumarin dye (G, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a magenta textile. The magenta textile was irradiated by UV-VIS light (380 nm-780 nm) to measure its transmittance spectrum, as shown in FIG. 7. When the dye amount achieved 0.3 parts by weight, the magenta textile had the highest transmittance for specific visible light wavelength, it means that the fluorescent emission of the magenta textile was strongest.
Example 21
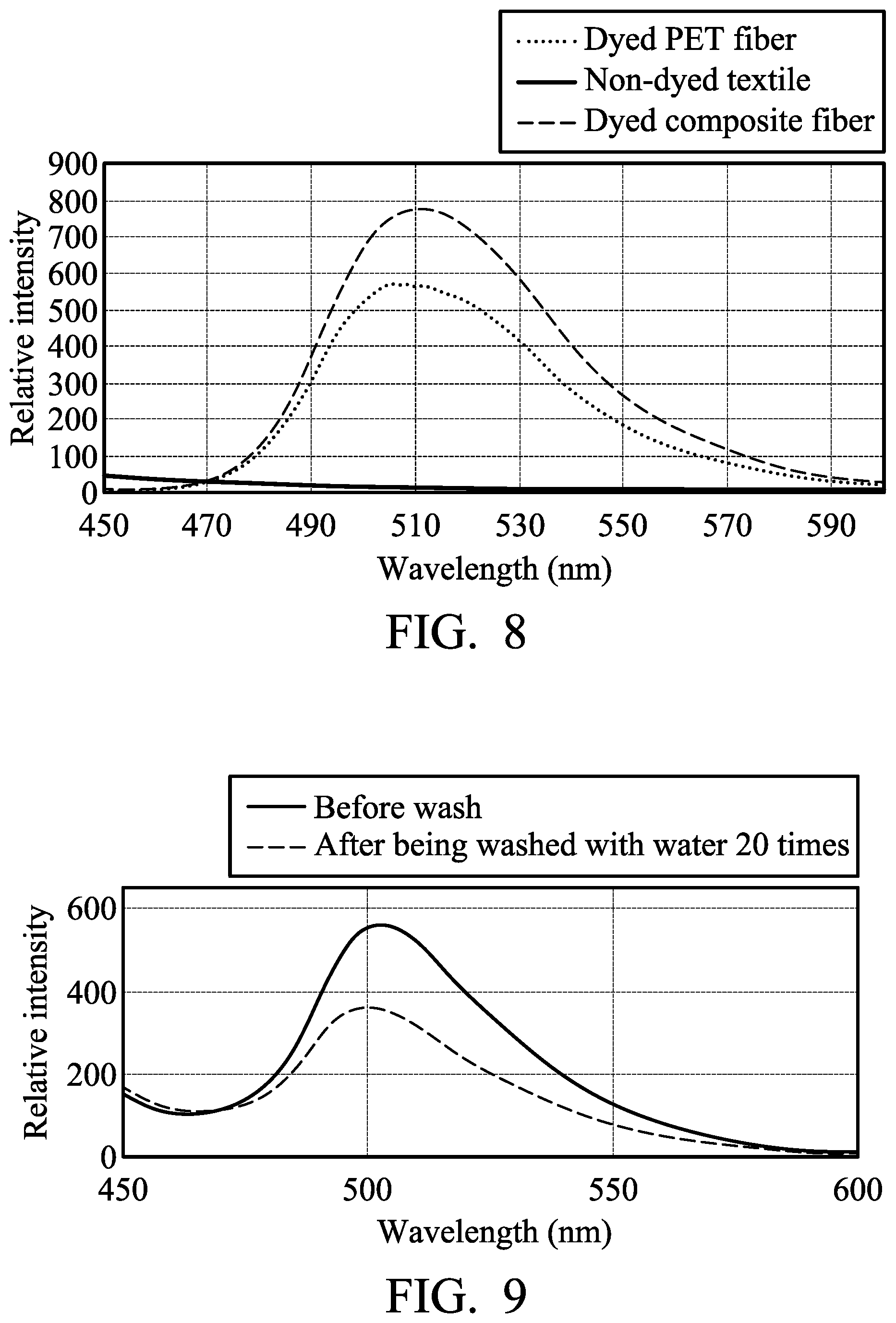
[0076] 1 part by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) and 0.3 parts by weight of titanium oxide particles (with a diameter of 100 nm to 500 nm) were dispersed in 98.7 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile. The non-dyed textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 8.
[0077] 0.5 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 8.
[0078] PET particles were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile. 0.5 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 8. The non-dyed textile did not have the emission for specific visible light wavelength. The dyed composite fiber with the doped zinc oxide particles had stronger emission intensity for specific visible light wavelength than the dyed PET fiber.
Example 22
[0079] 0.48 parts by weight of aluminum doped zinc oxide particles (with a diameter of 50 nm to 150 nm) were dispersed in 99.52 parts by weight of PET. Polyester particles of the above composition were melt spun and false twisted to form a yarn with the standard T75/72DTY. The yarn was further woven to form a textile.
[0080] 0.34 parts by weight of yellow fluorescent coumarin dye (10GN, commercially available from Gemmy Dye and Chemical, Co., Ltd, based on 100 parts by weight of the textile) was dissolved in 2000 parts by weight of water. The textile was soaked in the aqueous solution of the fluorescent dye at room temperature. The aqueous solution of the fluorescent dye was then heated to 130.degree. C. by a heating rate of 2.degree. C./min, and kept at 130.degree. C. for 40 minutes. The dyed textile was then cooled, washed with water, washed with soap, and baking dried to obtain a yellow textile. The yellow textile was irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 9. Afterward, the yellow textile was washed with water 20 times, and then irradiated by an excitation light source (325 nm) to measure its fluorescent spectroscopic spectrum, as shown in FIG. 9. Because the yellow textile after being washed with water 20 times still had a certain degree of the fluorescent emission, the dye and the textile had excellent adhesion, as shown in FIG. 9.
[0081] It will be apparent to those skilled in the art that various modifications and variations can be made to the disclosed methods and materials. It is intended that the specification and examples be considered as exemplary only, with the true scope of the disclosure being indicated by the following claims and their equivalents.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.