Process For The Removal Of Nitrogen-Containing Compounds From A Hydrocarbon Feed
Bridier; Blaise H. ; et al.
U.S. patent application number 16/621052 was filed with the patent office on 2020-06-25 for process for the removal of nitrogen-containing compounds from a hydrocarbon feed. The applicant listed for this patent is ExxonMobil Chmical Patents Inc.. Invention is credited to Blaise H. Bridier, Paul Hamilton, John J. Houben, Mechilium J.G. Janssen, Luc R. M. Martens, Pierre J. Osterrieth, Christopher J. Taylor, Gerandine Tosin, Bee N. Yap.
| Application Number | 20200199460 16/621052 |
| Document ID | / |
| Family ID | 59409157 |
| Filed Date | 2020-06-25 |


| United States Patent Application | 20200199460 |
| Kind Code | A1 |
| Bridier; Blaise H. ; et al. | June 25, 2020 |
Process For The Removal Of Nitrogen-Containing Compounds From A Hydrocarbon Feed
Abstract
Process for the removal of nitrogen-containing compounds from a hydrocarbon feed comprising at least one olefin selected from the C3, C4, C5 and optionally C6 olefins and mixtures thereof comprising solvent extraction comprising monitoring the degradation of the solvent.
| Inventors: | Bridier; Blaise H.; (Uccle, BE) ; Hamilton; Paul; (Spring, TX) ; Janssen; Mechilium J.G.; (Kessel-Lo, Leuven, BE) ; Osterrieth; Pierre J.; (Bruxelles, BE) ; Taylor; Christopher J.; (Southampton, GB) ; Martens; Luc R. M.; (Vlaams Brabant, BE) ; Yap; Bee N.; (Brussels, BR) ; Tosin; Gerandine; (Brussels, BE) ; Houben; John J.; (Rotterdam, NL) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59409157 | ||||||||||
| Appl. No.: | 16/621052 | ||||||||||
| Filed: | June 15, 2018 | ||||||||||
| PCT Filed: | June 15, 2018 | ||||||||||
| PCT NO: | PCT/EP2018/065955 | ||||||||||
| 371 Date: | December 10, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 2/30 20130101; C10G 50/00 20130101; C10G 2300/202 20130101; C10G 21/28 20130101; C10G 2300/1088 20130101; C10G 2300/4081 20130101; C10G 45/68 20130101; C10G 29/205 20130101; C10G 21/16 20130101; C10G 21/30 20130101; C10G 2400/20 20130101; C10G 45/58 20130101; C10G 2300/1092 20130101 |
| International Class: | C10G 21/16 20060101 C10G021/16; C10G 21/28 20060101 C10G021/28 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 13, 2017 | EP | 17181097.1 |
Claims
1. A Process for the removal of nitrogen-containing compounds from a hydrocarbon feed comprising at least one olefin selected from the C3, C4, C5 and optionally C6 olefins and mixtures thereof comprising the steps of: a) contacting the hydrocarbon feed with an extracting solvent containing propylene carbonate to obtain a hydrocarbon phase of reduced nitrogen-containing compounds content and a solvent phase containing dissolved nitrogen-containing compounds; b) separating the hydrocarbon phase of reduced nitrogen-containing compounds content from a solvent phase containing dissolved nitrogen-containing compounds; c) subjecting the hydrocarbon phase from step (b) to a distillation step to separate a heavy phase containing the solvent and optionally heavy hydrocarbon compounds from a light hydrocarbon fraction containing the C3, C4 and C5 olefins; d) optionally subjecting the light fraction to further processing; and e) subjecting the solvent phase from step (b), optionally with the heavy phase from step (c), to a regeneration step; and f) recycling the regenerated solvent back to step (a), said process further comprising monitoring the degradation of the solvent by keeping the content of the propylene carbonate present in recycled solvent above 94% by weight relative to the solvent as measured by gas chromatography; keeping the Total Acid Number (TAN) of the recycled solvent measured according to ISO 1843/2 below 0.1 mg KOH/g keeping the distribution coefficient of the nitrogen-containing compounds between the recycled solvent and the hydrocarbon feed above 50% of the value measured with fresh solvent; keeping the settling time of the recycled solvent in the hydrocarbon feed below 200% of the settling time of the raw propylene carbonate; or maintaining the surface tension of the recycled solvent as measured by the Ring Tear Off method above 25 mN/m or the interfacial tension measured by the Du Nouy Ring method above 6 mN/m.
2. The process according to claim 1, wherein the accumulation of solvent degradation products and heavy hydrocarbons in the recycling loop can be avoided or at least minimize by one or more of the following steps filtration of the solvent before feeding it back to step (a); depending on the monitoring results purging an appropriate amount of solvent and replacing by fresh solvent; operating the regeneration step in such a way that the heavy hydrocarbon compounds and the solvent degradation products are removed at said regeneration step, for example by decreasing the reflux ratio in the stripper or adapting the regeneration temperature; and adding a distillation step between the regeneration and the extraction steps.
3. The process according to claim 1, wherein the regeneration step (e) is operated in a stripping column by using an inert gas or a light hydrocarbon having from 1 to 7 carbon atoms.
4. The process according to claim 1, wherein the solvent comprise propylene carbonate.
5. The process according to claim 1, wherein the hydrocarbon feed is a C5 olefin feeds including FCC Light Naphtha streams, steam cracker C5 rich streams that have been treated for diene removal, C5 olefin containing streams from Gas to Olefin (GTO) Units, or Fisher-Tropsch Units.
6. The process according to claim 1, wherein the stripping column is equipped with an overhead reflux resulting in a column configuration comprising a stripping portion in the bottom and a distillation portion at the top.
7. The process according to claim 1, wherein, depending on the monitoring results, an appropriate amount of solvent is purged at step (e) and replaced by fresh solvent.
8. A process for converting a hydrocarbon feed comprising at least one olefin selected from the C3, C4, C5 and optionally C6 olefins and mixtures thereof contaminated with at least one nitrogen-containing compound into a hydrocarbon product; said process comprising the steps of: i) providing a hydrocarbon feed contaminated with at least one nitrogen-containing compound; ii) removing the nitrogen-containing compounds from the feed to produce a hydrocarbon feed stream having a reduced level of nitrogen-containing compounds; and contacting said hydrocarbon feed stream having a reduced level of nitrogen-containing compounds with a catalyst in order to convert the feed stream into a hydrocarbon product, wherein nitrogen-containing compounds are removed from the feed by using the process as defined in any one of the preceding claims.
9. The process according to claim 8 wherein step (iii) is an isomerization, an alkylation, a hydrogenation, an aromatization or an oligomerization step.
10. The process according to claim 9, wherein step (ii) is an oligomerisation step using an oligomerization catalyst comprising a material selected from the group consisting of zeolites, phosphoric acids, supported metal oxides and combinations thereof.
11. The process according to claim 10, wherein the oligomerization catalyst comprises a zeolite, in particular a zeolite selected from the group consisting of ZSM-5, ZSM-11, ZSM-1, ZSM-18, ZSM-22, ZSM-23, ZSM-35, ZSM-38, ZSM-48, ZSM-50, ZSM-57, ITQ-39 and mixtures thereof.
Description
[0001] This invention relates to a process for removing nitrogen-containing compounds from a hydrocarbon feed, in particular from a hydrocarbon feed containing C3 to C6 olefins. The present invention also relates to a process for converting a hydrocarbon feed contaminated with at least one nitrogen-containing compound into a hydrocarbon product, said process comprising a step of removal of nitrogen-containing compounds from the hydrocarbon feed.
BACKGROUND
[0002] Hydrocarbon feed streams containing light olefins, typically C3 to C6 olefins, are used in catalytic oligomerisation processes to obtain oligomers and/or polymers, typically heptenes, octenes, nonenes and dodecenes. These products may be converted to further products such as alcohols, plasticisers, adipates, mercaptans and solvents.
[0003] The hydrocarbon feed steams derive from various sources including refinery operations such as catalytic or steam cracking and are known to contain certain amounts of impurities including, but not limited to, nitrogen-containing compounds such as nitriles. These impurities may have an adverse effect on the catalysts used in the oligomerisation process such as phosphoric acid-based catalyst, zeolite-based catalyst and supported metal catalysts. In particular, nitrogen-containing compounds may act as catalyst poisons (contaminants) reducing the activity and life of the catalyst and/or inducing unsustainable operation conditions and should be removed from these hydrocarbon feed streams.
[0004] Prior art approaches to remove nitrogen-containing compounds include removal via liquid-liquid extraction techniques or adsorption techniques using a so-called guard bed.
[0005] Liquid-liquid extraction is disclosed for example in WO 2009/058229 and WO 2012/078218. The latter document discloses removal of nitriles and/or pyrroles from a feed stream comprising olefins and paraffins by contacting the feed stream with a solvent and removing at least a portion of the nitriles and the pyrroles from the feed stream. The process conditions are based on the distribution coefficient of the nitrogen-containing compounds in the feed stream and the solvent.
[0006] Solvents such as sulfone compounds or alkyl/alkenyl/aryl carbonates, typically propylene carbonate, are generally used. Such solvents may be recycled or reused in the extraction process but generally after a regeneration process such as vacuum and steam distillation, back extraction, adsorption and anion-cation exchange resin column. Regeneration processes include contacting the solvent after use in an extraction process with an inert gas or light hydrocarbons.
[0007] Continuous solvent recycling will also generate accumulation in the solvent recycle loop of solvent degradation products as well as of the heavy hydrocarbon compounds that might be present in the hydrocarbon feed stream. These components collected in the solvent recycling loop can also strongly affect the effectiveness of the removal process and need to be timely removed. These compounds are not taken into consideration in WO 2012/078218.
[0008] Thus there remains a need for further processes which allow for an efficient removal of nitrogen-based impurities that can be run for long periods of time while maintaining good operating conditions.
SUMMARY OF THE INVENTION
[0009] According to a first aspect, the present invention solves the above problem(s) by providing a process for the removal of nitrogen-containing compounds from a hydrocarbon feed, preferably from a hydrocarbon feed comprising at least one olefin selected from C3, C4, C5 and optionally C6 olefins and mixtures thereof, the process comprising the steps of: [0010] a) contacting the hydrocarbon feed with an extracting solvent containing propylene carbonate, to obtain a hydrocarbon phase with a reduced nitrogen-containing compounds content and a solvent phase containing nitrogen-containing compounds dissolved therein; [0011] b) separating the hydrocarbon phase with a reduced nitrogen-containing compounds content from the solvent phase containing nitrogen-containing compounds dissolved therein; [0012] c) subjecting the hydrocarbon phase obtained in step (b) to a distillation step to separate a heavy phase containing the solvent and optionally heavy hydrocarbon compounds, from a light hydrocarbon fraction containing the at least one olefin selected from C3, C4 and C5 olefins and mixtures thereof; [0013] d) optionally subjecting the light fraction to further processing; and [0014] e) subjecting the solvent phase obtained in step (b), optionally with the heavy phase from step (c), to a regeneration step; and [0015] f) recycling the regenerated solvent back to step (a), said process further comprising monitoring the degradation of the solvent by [0016] keeping the content of the propylene carbonate present in recycled solvent above 94% by weight relative to the total amount of recycled solvent, as measured by gas chromatography; or [0017] keeping the Total Acid Number (TAN) of the recycled solvent measured according to ISO 1843/2 below 0.1 mg KOH/g; or [0018] keeping the distribution coefficient of the nitrogen-containing compounds between the recycled solvent and the hydrocarbon feed above 50% of the value measured for fresh solvent.
[0019] According to this invention, the degradation of the solvent may also be monitored by [0020] maintaining the surface tension of the recycled solvent as measured by the Ring Tear Off method above 25 mN/m; or [0021] maintaining the interfacial tension of the recycled solvent measured by the Du Nouy Ring method above 6 mN/m; or [0022] keeping the settling time of the recycled solvent in the hydrocarbon feed below 200% of the settling time of the raw propylene carbonate.
[0023] The regeneration step (e) is preferably operated in a stripping column by using an inert gas such as nitrogen or one or more of the Group 18 inert gases of the Periodic Table of the Elements (Wikipedia--2017) or a light hydrocarbon as defined below.
[0024] According to a second aspect, the present invention relates to a process for converting a hydrocarbon feed comprising at least one olefin selected from C3, C4, C5, and optionally C6 olefins and mixtures thereof, contaminated with at least one nitrogen-containing compound into a hydrocarbon product, said process comprising the steps of: [0025] i) providing a hydrocarbon feed contaminated with at least one nitrogen-containing compound; [0026] ii) removing the nitrogen-containing compounds from the feed by using a process in accordance with the first aspect of the present invention to produce a hydrocarbon feed stream having a reduced level of nitrogen-containing compounds; and [0027] iii) contacting said hydrocarbon feed stream having a reduced level of nitrogen-containing compounds with a catalyst in order to convert the feed stream into a hydrocarbon product.
[0028] Further and preferred embodiments are disclosed in the dependent claims and in the following description including the examples and figures illustrating the present invention.
BRIEF DESCRIPTION OF THE FIGURES
[0029] FIG. 1 is a schematic representation of steps (a) to (e) of the present invention including monitoring of the solvent degradation.
[0030] FIG. 2 is a schematic representation of a preferred distillation column layout used in step (c).
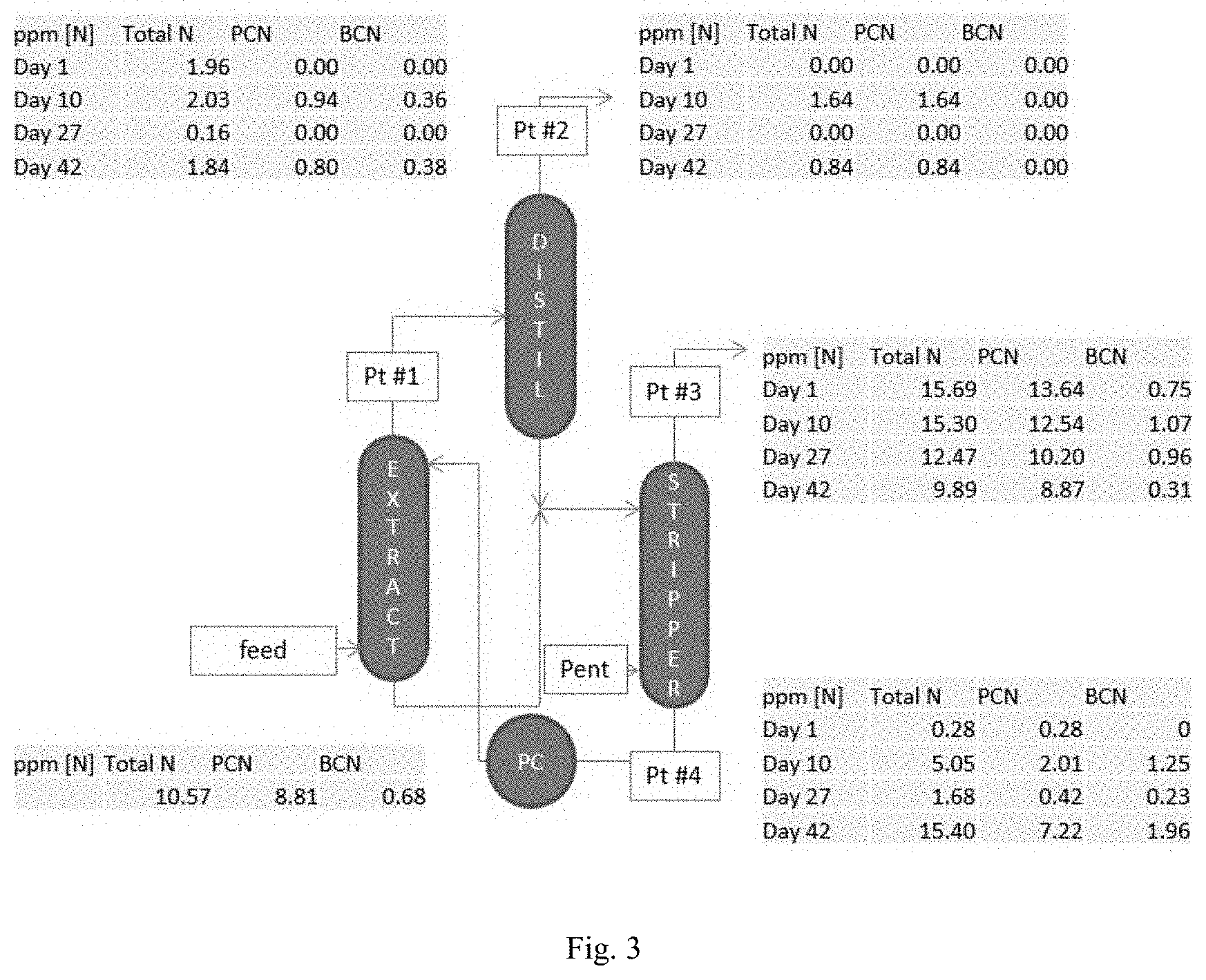
[0031] FIG. 3 shows the content of different nitrogen compounds in the different process streams in a process according to the present invention.
DETAILED DESCRIPTION OF THE INVENTION
[0032] Before the materials, compounds, components, composition and/or processes of the present invention are disclosed in more detail, it is noted that the singular forms "a", "an" and "the" include plural referents unless otherwise specified.
[0033] Furthermore, the words "comprising" (and any form thereof), having (and any form thereof), including (and any form thereof) or containing (and any form thereof) are inclusive or open ended and do not exclude additional, unrecited elements such as materials, compounds or compositions or additional process steps.
[0034] Embodiments might be combined and are not separated except otherwise specified.
[0035] As stated above, according to a first aspect, the present invention solves the problems of the accumulation of degradation products and heavy hydrocarbon compound in a solvent recycled loop by providing a process for the removal of nitrogen-containing compounds from a hydrocarbon feed, preferably a hydrocarbon feed comprising preferably at least one olefin selected from the C3, C4, C5 and optionally C6 olefins and mixtures thereof comprising the steps of: [0036] a) contacting the hydrocarbon feed with an extracting solvent containing propylene carbonate to obtain a hydrocarbon phase of reduced nitrogen-containing compounds content and a solvent phase containing dissolved nitrogen-containing compounds; [0037] b) separating the hydrocarbon phase of reduced nitrogen-containing compounds content from solvent phase containing dissolved nitrogen-containing compounds; [0038] c) subjecting the hydrocarbon phase from step (b) to a distillation step to separate a heavy phase containing the solvent and optionally heavy hydrocarbon compounds and from a light fraction containing the C3, C4 and C5 olefins; [0039] d) optionally subjecting the light fraction to further processing; and [0040] e) subjecting the solvent phase from step (b), optionally with the heavy phase from step (c), to a regeneration step; and [0041] (f) recycling the regenerated solvent back to step (a), said process further monitoring the degradation of the solvent by [0042] keeping the content of the propylene carbonate present in the recycled solvent above 94 wt % by weigh relative to the weight of recycled solvent as measured by gas chromatography; [0043] keeping the TAN of the recycled solvent measured according to ISO 1843/2 below 0.1 mg KOH/g. [0044] keeping the distribution coefficient of the nitrogen-containing compounds between the recycled solvent and the hydrocarbon feed above 50% of the value measured with fresh solvent; [0045] keeping the settling time of the recycled solvent in the hydrocarbon feed below 200% of the settling time of the raw propylene carbonate; or [0046] maintaining the surface tension of the recycled solvent as measured by the Ring Tear Off method above 25 mN/m or the interfacial tension measured by the Du Nouy Ring method above 6 mN/m.
[0047] Preferably the recycled solvent is stored in a reservoir located in the solvent recycle loop after its regeneration and before being recycled to step (a). Preferably such reservoir is equipped with skimming device to ensure that any accumulation of hydrocarbons material on the top of said reservoir can be removed.
[0048] In a preferred embodiment, the accumulation of solvent degradation products and heavy hydrocarbons in the recycling loop can be avoided or at least minimized by one or more of the following steps [0049] 1. filtration of the solvent before feeding it back to step (a); [0050] 2. depending on the monitoring results, purging an appropriate amount of solvent and replacing by fresh solvent; [0051] 3. operating the regeneration step in such a way that the heavy hydrocarbon compounds and the solvent degradation products are removed during said regeneration step, for example by decreasing the reflux ratio in the stripper or adapting the regeneration temperature; and [0052] 4. adding a distillation step between the regeneration and the extraction steps.
[0053] According to this invention, degradation of the solvent is preferably limited by at least one of preventing oxygen and/or air intake in the equipment used to operate the processes according to this invention, keeping the water content in the solvent recycle loop below 1 wt %, more preferably below 0.6 wt %, or operating the regeneration process at temperature below 140.degree. C., preferably below 135.degree. C.
[0054] These operating conditions further limit corrosion of the equipment.
[0055] The hydrocarbon feed used in this invention can be any hydrocarbon feed. Preferably the hydrocarbon feed contains at least one olefin selected from C3, C4, C5 and optionally C6 olefins and is contaminated with nitrogen-containing compounds. More preferably the hydrocarbon feed contains at least one olefin selected from C3, C4, C5 and C6 olefins and nitrogen-containing compounds.
[0056] As used herein, "olefins" refers to any unsaturated hydrocarbons having the formula C.sub.nH.sub.2n. The feed may also comprise one or more paraffins. As used herein, "paraffins" refers to any of the saturated hydrocarbons having the formula C.sub.nH.sub.2n+2. The paraffins that may be present in the olefin feed typically have from 1 to 15 carbon atoms, conveniently at least 3 and no more than 6 carbon atoms. Examples of suitable paraffins include methane, ethane, propane, butane, pentane, hexane, isomers thereof and mixtures thereof.
[0057] If present, the paraffin usually acts as a diluent. The olefin feed may comprise at least 10%, at least 25%, at least 30%, at least 35%, or at least 40% paraffin, based upon the total volume of the feed. Alternatively stated, if used, the diluent may be present in the olefin feed in the range from 10% to 40%, alternatively, from 10% to 35%, and alternatively, from 20% to 35% based upon the total volume of the feed. The diluent may also be fed to the reactor(s) separately from the olefin feed. When fed separately, the diluent may be fed in amounts equivalent to those mentioned above, where the diluent is co-fed with the feed.
[0058] The olefin containing feed comprises olefins selected from propene, butenes, pentenes, optionally hexenes, their isomers, and mixtures thereof. The process of this invention is especially useful for the oligomerization of feeds comprising propene, butenes, pentenes, their isomers, and mixtures thereof. As used herein, "isomers" refers to compounds having the same molecular formula but different structural formula.
[0059] Additionally, the feed may comprise an oligomer (higher olefin), for example, a dimer, such as one provided by recycling a part of an olefin oligomerization product stream. As used herein, "oligomer(s)" or "oligomer product" refers to an olefin (or a mixture of olefins) made from a few light olefins. For example, oligomers include dimers, trimers, and tetramers, obtained from two, three or four light olefins of the same number of carbon atoms, and mixed oligomers, obtained from 2 or more olefins having different numbers of carbon atoms and mixtures thereof. Typically oligomers are olefins or mixture of olefins having 20 carbon atoms or less, alternatively, 15 carbon atoms or less, such as 9 carbon atoms or less, and conveniently, 8 carbon atoms or less.
[0060] The hydrocarbon feed preferably comprises 30 wt % or more olefins, such as 40 wt % or more olefins, alternatively, 50 wt % or more olefins, alternatively, 60 wt % or more olefins, alternatively, 70 wt % or more olefins, and alternatively, 80 wt % or more olefins, based upon the total weight of the feed.
[0061] According to the present invention, any of the above-described feeds may further contain at least one of diolefins and/or cyclic olefins. Typical cyclic compounds are cyclopentene, methylcyclohexene, cyclohexene and cycloheptene.
[0062] In any of the olefin oligomerization embodiments described herein, the feed should be totally free, or at least substantially free, of aromatic hydrocarbon compounds that consist solely of hydrogen and carbon atoms. In this context, "substantially free" means that the olefin feed contains 25 wt % or less, preferably 15 wt % or less, more preferably 10 wt % or less, such as 5 wt % or less, and most preferably 1 wt % or less aromatic hydrocarbon, based upon the total weight of the olefin feed.
[0063] Examples of suitable olefin feeds include untreated refinery streams such as Fluidized Catalytic Cracking (FCC) streams, steam cracker streams, coker streams, pyrolysis gasoline streams or reformates.
[0064] Examples of suitable C3 olefin-containing feeds include untreated C3 rich refinery streams such as "dilute" or "refinery grade" propylene from a Fluidized Catalytic Cracker (FCC), C3 rich stream from a steam cracker, from the production of "chemical grade" or "polymer grade" propylene, from refinery gas recovery units, from Propane Dehydrogenation Units, from Gas to Olefin (GTO) Units, or from Fisher-Tropsch Units, and C3 rich return streams from polypropylene producing units. These C3 streams may contain for example from 50 to 60 wt % of propylene, or 65 wt % or more, or 70 wt % or above such as 72 wt % or 75 wt % or even up to 79 wt %.
[0065] Examples of suitable C4 olefin containing feeds include refinery feeds often referred to as Raffinate-1 (RAF-1), Raffinate-2 (RAF-2) or Raffinate-3 (RAF-3). Typically, Raffinate-1, Raffinate-2 and Raffinate-3 may be regarded as streams obtainable at various stages in the processing of crude C4 streams obtained from petroleum refining processes. These streams are well known by the person skilled in the art.
[0066] Examples of suitable C5 olefin feeds include FCC Light Naphtha streams, steam cracker C5 rich streams that have been treated for diene removal, C5 olefin containing streams from Gas to Olefin (GTO) Units, or Fisher-Tropsch Units. In this context, "Light Naphtha" is understood to mean a stream having a specific gravity in the range 0.65 to 0.73, An ASTM-D86 boiling point range between 35 and 125.degree. C. and that contains a range of olefin, paraffin, diolefins and cyclic hydrocarbon compounds with carbons numbers typically in the range C5 to C8. More specifically, according to an embodiment, a so-called Light Light Catalytic Naphtha (LLCN) stream may be used. Such stream is characterized by a boiling point range of, for example, from 25 to 70.degree. C. at atmospheric pressure and a specific gravity between 0.63 and 0.68 and contains at least 60 wt % C5 hydrocarbons.
[0067] The hydrocarbon feed is characterized by comprising a certain level of nitrogen-containing compounds and, optionally, certain levels of other compounds such as oxygen-containing compounds, sulphur-containing compounds, water, diolefins, cyclic olefins and mixtures thereof. The levels (concentrations) of both the nitrogen-containing compounds and said other compounds are usually in a range referred to as impurities or at least as minor components of the stream. In particular, with regard to nitrogen- and other heteroatom-containing compounds, the concentrations of these types of compounds will usually be in the range of from 0.1 to several hundred weight ppm (wt ppm), typically in a range of from 10 to 500 wt ppm, relative to the total weight of the stream. With regard to non-heteroatom-containing compounds, such as dienes, the concentrations of these types of compounds will typically be in the range of from 0.01 to 5 or 10 weight % (wt %) relative to the total weight of the feed.
[0068] The process of this invention is capable of reducing the content of nitrogen-containing compounds in the hydrocarbon feed from an initial range of from 10 to 500 wt ppm to the range of from 20 to 500 wt ppb relative to the weight of the feed. According to the present invention, any of the above-described feeds contains nitrogen-containing compounds and other impurities acting as catalyst contaminants which must be removed to an acceptable level before the hydrocarbon feed undergoes a catalyzed reaction.
[0069] In particular, the nitrogen-containing compounds comprise nitriles, pyrroles or mixtures thereof, typically nitriles. As used herein, pyrroles are chemical compounds of formula C.sub.4H.sub.5N optionally substituted by alkyl radical(s) containing from 1 to 3 carbon atoms. As used herein, "nitrile" is any organic compound that has a nitrile group (or --C.ident.N functional group). As used herein, "acetonitrile" (ACN) is the chemical compound with formula CH.sub.3CN. This colorless liquid is the simplest organic nitrile. As used herein, "propanenitrile", "propionitrile", or "ethyl cyanide" is a nitrile with the molecular formula C.sub.2H.sub.5CN and the terms may be used interchangeably. It is also a clear liquid. Preferably the nitriles removed are a C2 to C5 nitrile. In the most preferred embodiment the nitrile to be removed is propionitrile and butyl nitrile, C.sub.3H.sub.7CN. These compounds are especially toxic to oligomerization catalysts and their removal leads to significant catalyst life improvement.
[0070] The process of the invention will be further described with reference to FIG. 1 in which a hydrocarbon feed (1) is introduced into an extraction separation unit (2) where it is contacted with an extracting solvent (3) to obtain as overhead stream a hydrocarbon phase (4) of reduced nitrogen-containing compounds content and at the bottoms a solvent phase (5) containing dissolved nitrogen-containing compounds.
[0071] According to this invention the solvent comprises, consist essentially of propylene carbonate ("PC"). The content of propylene carbonate in the solvent is of at least 95 wt %, preferably at least 98 wt %. Content of above 99% are provide good results. Typically the solvent consists of propylene carbonate comprising traces of water.
[0072] The extraction is preferably carried out in an extraction column, more preferably a counter current column. The temperature and pressure of the extraction column are not critical provided that they ensure that both the hydrocarbon feed and the solvent remain in the liquid phase. The temperature is usually between 0 and 90.degree. C., preferably between 10 and 70.degree. C. The pressure may be between 0 and 10 bara, more preferably between 1 and 5 bara.
[0073] The operating parameters are selected to produce a hydrocarbon phase containing preferably between 1000 and 15,000 wt ppm of carbonates, more preferably between 3000 and 10,000 wt ppm of carbonates.
[0074] The solvent to hydrocarbon feed weight ratio typically ranges from between 0.05 and 2; preferably this ratio is between 0.1 and 1, more preferably about 0.3.
[0075] The level of nitrogen-containing compounds in the hydrocarbon phase having a reduced nitrogen-containing compound content is below 1 ppm, preferably below 0.6 ppm, more preferably below 0.3 ppm by weight.
[0076] Following the liquid liquid separation, the hydrocarbon phase (4) having a reduced nitrogen-containing compound content is transferred to a distillation column (6) in order to separate a heavy phase (7) containing the solvent and optionally heavy hydrocarbon compounds present in the feed, including any C6 olefins, if present in the feed, from a light fraction (8) containing the at least one olefin selected from C3, C4, and C5 olefins and mixtures thereof.
[0077] Heavy hydrocarbon compounds preferably refer to C6 olefins, C6+ olefins and hydrocarbon compounds with boiling point equal or above C6 olefin boiling point. Polycarbonate and cyclopentene are considered as heavy hydrocarbons.
[0078] The distillation is usually carried out through the use of columns with trays, packed columns including structured packing, random packing or a combination of both.
[0079] The final boiling point of the light fraction (8) is preferably below 60.degree. C., more preferably below 55.degree. C.
[0080] The light fraction (8) may be considered as a clean feed and may be used in any downstream process. Additional treatments may be required by said downstream process
[0081] The presence of two liquid phases in the overhead of the extraction column and in the bottom of the distillation column is preferably avoided as it potentially leads to process difficulties such as incorrect instrument readings, unexpected low patterns and/or accumulation of PC layers stalling of gravity driven heat exchangers.
[0082] To that effect, reboiler technology is preferably used at the bottom of the distillation column (6). Also at the top of the extraction column the level instrument tapping is preferably oriented to allow free draining of any PC that might be carried into the pipe work.
[0083] An example of a preferred distillation column layout is represented in FIG. 2. As shown, the hydrocarbon phase (4) is preheated before being introduced in a middle zone of the distillation column (6). The top of the distillation column is equipped with a condenser (14). A separator (15), typically a settling tank, at the bottom of the column, separates the heavier phase from the lighter phase that is sent to a reboiler (16) optionally after purging. The separator (15) may be replaced by a pump.
[0084] The solvent phase (7) removed from the distillation column (6) is subjected to regeneration to remove undesired materials described hereabove and comprising solvent degradation products and/or heavy hydrocarbon compounds from the feed before being returned back to the extraction/separation unit with the hydrocarbon feed.
[0085] Methods to remove undesired materials from the solvent phase (7) include but are not limited to, vacuum and steam distillation, back extraction, adsorption (e.g. using a solid sorbent) and anion-cation exchange resin columns.
[0086] In this invention, regeneration is preferably performed in a stripping unit (9), typically a stripping column, by stripping the solvent phase with a stream of stripping gas (12) whereby the undesired materials are taken out overhead and regenerated solvent (10) is taken as bottoms.
[0087] Optionally the solvent phase (5) separated at the bottoms of the extraction unit is routed to the stripping column as well as the solvent phase (7) but preferably via a different feed point.
[0088] According to this invention the stripping gas (12) may be selected from inert gas, typically nitrogen from light hydrocarbons having from 1 to 6 carbon atoms, preferably from 1 to 5 carbon atoms or mixture thereof. The stripping gas is typically free of nitrogen-containing compounds, i.e. contains preferably less than 0.3, more preferably less than 0.1 wt % of nitrogen-containing compounds.
[0089] According to a preferred embodiment, the stripping gas comprises at least 98, preferably at least 99, and more preferably at least 99.5 wt % of one or more hydrocarbons. The stripping gas is preferably a fraction of the light fraction (8) produced at step (c).
[0090] The contacting of the solvent phase with the stripping gas is preferably carried out at a pressure of from 1 to 5 bars. The weight ratio of the stripping gas to the crude product is from 50:1 to 125:1 determined at the temperature and pressure used in the stripping unit.
[0091] Recovery of the undesired materials is maximized by operating the stripper under high mixing conditions, for example by minimizing the residence time of the solvent phase inside the stripper and/or by operating the stripping unit at high load. It is also preferred that the stripper column includes inert solid surfaces or trays to facilitate contact between the liquid and gas phases.
[0092] Removal of undesired materials by stripping the solvent phase with the stripping gas could result in some entrainment of solvent into the stripping column overheads.
[0093] According to a preferred embodiment, the stripping column is equipped with an overhead reflux resulting in a column configuration comprising a stripping portion in the bottom and a distillation portion at the top. By using such embodiment the degradation of the solvent will be limited and the need for monitoring such degradation is reduced.
[0094] Such configuration also further allows control of the solvent lost to the stripping tower meaning that the amount in the overheads could be increased to allow for solvent purging if required by the overall process. Also such configuration allows for better separation of the heavy hydrocarbons.
[0095] The reflux rate is usually fixed relative to the net hydrocarbon feed to the stripping tower, the net hydrocarbon feed being the sum of the stripping gas with the extractor and distillation bottoms streams.
[0096] It is recommended that between 2 and 40, preferably 5 and 30% wt being routed back to the tower as reflux.
[0097] The stream (13) recovered at the top of the stripper column is suitable for Mogas blending.
[0098] After regeneration, the regenerated solvent (10) is transferred back to the extraction tower, preferably at the top of the extraction tower (2), thereby closing the solvent recycle loop.
[0099] According to this invention the accumulation of the degradation products in the solvent recycle loop is monitored to control the efficiency of the overall process as described here above.
[0100] The monitoring is usually carried out by control device (17) adapted to the analytical method step described below.
[0101] The content of one or more alkyl, alkenyl, or aryl carbonate is measured by gas chromatography using a 6890 gas chromatograph from Agilent using a HP-FFAP polyethylene glycol TPA column using 3.3 ml*min of carrier gas. The operating temperature is between room temperature and 220.degree. C. Flame Ionization Detector (FID) is used as detector.
[0102] The TAN of the recycled solvent is determined according to ISO 1843/2
[0103] The distribution coefficient of the nitrogen-containing compounds between the solvent and the hydrocarbon feed is obtained by contacting at room temperature equal volumes of the hydrocarbon feed and solvent and manual shaking the obtained mixture in a separation funnel at ambient temperature for 1 minute. After settling, the two phases are separated and the obtained hydrocarbon phase is further extracted with equal amount of solvent under the same shaking conditions. This manipulation is repeated at least 3 times under the same conditions and the content of nitrogen-containing compound in the different phases are measured by gas chromatography using a Free Fatty Acid Phase (FFAP) column equipped with a FID detector or with a nitrogen chemo luminescence detector. The distribution coefficient is the slope of the straight line obtained by plotting the nitrile concentration in the hydrocarbon phase versus the one in the solvent.
[0104] The interfacial tension is measured via the Ring Tear Off (RTO) method BS-EN 14370:2004
[0105] The measurement of surface tension of the solvent is performed by the Du Nouy Ring method BS-EN 14370:2004
[0106] The settling time is measured on different aged solvent recycling phase. The settling time of the recycled solvent in the hydrocarbon feed should be kept below 200% of the settling time of the raw propylene carbonate, preferably below 150% under the same settling conditions. The settling time is preferably measured by mixing 10 ml of the polycarbonate stream with 10 ml of the hydrocarbon fee during 15 sec at 20.degree. C. The settling time of recycled solvent measured under these conditions is preferably kept below 20 s.
[0107] According to a second aspect, the present invention relates to a process for converting a hydrocarbon feed comprising at least one olefin selected from the C3, C4, C5 and optionally C6 olefins and mixtures thereof contaminated with at least one nitrogen-containing compound into a hydrocarbon product, said process comprising the steps of: [0108] i) providing a hydrocarbon feed contaminated with at least one nitrogen-containing compound; [0109] ii) removing the nitrogen-containing compounds from the feed by using a process in accordance with the first aspect of the present invention to produce a hydrocarbon feed stream having a reduced level of nitrogen-containing compounds; and contacting said hydrocarbon feed stream having a reduced level of nitrogen-containing compounds with a catalyst in order to convert the feed stream into a hydrocarbon product.
[0110] The process for converting the hydrocarbon feed into a hydrocarbon product concerned by this invention may be an isomerization, an alkylation, a hydrogenation, an aromatization or an oligomerization process; preferably such process is an isomerization or an oligomerization process.
[0111] Typically the process of the present invention is an olefin oligomerization process. As used herein, "oligomerization process" refers to any process by which light olefins are linked together to form the oligomer(s) as defined herein. As used herein, the term "oligomerization conditions" refers to any and all those variations of equipment, conditions (e.g. temperatures, pressures, weight hourly space velocities etc.), materials, and reactor schemes that are suitable to conduct the oligomerization process to produce the oligomer(s) as known and applied in the art.
[0112] In a preferred embodiment, the hydrocarbon feed comprises an olefin, wherein the olefin is preferably selected from the group consisting of C3, C4, C5 and C6 olefins and mixtures thereof, in particular C3, C4 and C5 olefins.
[0113] In a preferred embodiment, the hydrocarbon product comprises an oligomerization product and the catalyst is an oligomerization catalyst comprising a material selected from the group consisting of zeolites, phosphoric acids, supported metal oxides and combinations thereof.
[0114] Preferably, the oligomerization catalyst comprises a zeolite, in particular a zeolite selected from the group consisting of ZSM-5, ZSM-11, ZSM-12, ZSM-18, ZSM-22, ZSM-23, ZSM-35, ZSM-38, ZSM-48, ZSM-50, ZSM-57, ITQ-39 and mixtures thereof.
[0115] Exemplary methods and materials utilized in the oligomerization process are provided in WO2012/033562, U.S. Pat. No. 4,973,790 and US-A-2012/0022224.
[0116] The invention is particularly, but not exclusively, concerned with processes suitable for the production of C5 to C20 olefins boiling in the range of 30.degree. to 310.degree. C., preferably 30.degree. to 300.degree. C., more preferably 30.degree. to 250.degree. C., from propylene and/or butene and/or pentene containing feedstocks or their mixtures, though ethylene may be present as well. The oligomer product may be fractionated in a series of discrete products. In particular the invention is concerned with the production of the olefins shown in the following table. Typical values are indicate in table below.
TABLE-US-00001 Distillation Range (.degree. C.) ASTM D1078 Oligomer Products Initial Boiling Point Dry Point Pentenes 30 Hexenes 63 72 Heptenes 88 97 Octenes 114 126 Nonenes 135 143 Decenes 155 160 Undecenes 167 178 Propylene Tetramers 175 225 or Dodecenes Tridecenes 204 213
[0117] The oligomer products are useful in many applications and are the starting material for further processes. For example, the oligomer product may be polymerized to produce polyolefins that have application in the plastic industry and synthetic basestocks for lubricants. The oligomer product may be used in alkylation reactions for the product of surfactants. The oligomer products may be reacted with sulphur containing compounds to produce mercaptans. The oligomer product may undergo hydroformylation and subsequently hydrogenation to produce alcohols. The alcohols may be used in industry such as, for example, solvents, or be incorporated into the production of detergents/surfactants. The alcohols may further be used in many other areas of industry such as, for example, undergoing esterification to produce esters that have application as plasticizers. Oligomer products may be hydrogenated to produce a predominately paraffin product such as ISOPAR.TM..
[0118] Products could be streams suitable for blending into fuels dispositions including Mogas, distillate, diesel, jet fuel etc. from processes like EMOGAS (ExxonMobil Olefins to Gasoline), MODG (Mobil Olefins to Diesel and Gasoline).
EXAMPLES
[0119] The examples below illustrate some aspects of the present invention
Example 1
[0120] In this example a LLCN hydrocarbon feed is first sent to an extraction tower, where it is contacted with PC as extraction solvent. Any nitriles present in the hydrocarbon feed are absorbed into the PC phase. The overheads of the extractor tower are routed to a distillation column where any PC that is present in the purified hydrocarbon stream is separated into the bottoms along with any undesired hydrocarbons. The overhead stream is the desired purified hydrocarbon stream. The bottoms of the distillation are routed to a stripping tower. The bottom of the extractor is routed to the same stripping tower as the distillation bottoms, possibly via a different feed point. In the stripping the stripping medium is heated pentane coming from higher olefin process which uses the clean LLCN.
[0121] Any liquid in the overheads of the stripper is condensed and routed to the refinery mogas pool.
[0122] The data in FIG. 3 demonstrate that the technology achieves the required separations. Hydrocarbon feed nitrile content is typically much higher than the nitrogen measured at point 1 (extractor overheads). There are normally very low nitrogen levels at point 2 (distillation overheads), demonstrating that the cleaned hydrocarbon is suitable for the downstream acid catalyzed oligomerization process. The difference in nitrogen content between points 3 and 4 (stripper top and bottoms), shows that it is being stripped out from the circulating used PC. The consistent performance of the extractor to remove nitrogen-containing compounds from the feed demonstrates that the process works in a continuous closed loop.
[0123] Also the process according to the present invention can be operated at low corrosion rate typically less or equal than 2.6 mil/yr allowing low cost carbon steel to be used for the unit construction.
Example 2
[0124] The data in table 1 represents the model predicted compositions of the various streams in the process using PRO II simulation model based on measured physical properties of the stream components. Key points to note are the very low levels of nitrile in the clean C5 stream and the high levels of nitrile in the extractor bottoms and stripper overheads.
[0125] In the example shown in Table 1, some optimization of conditions has been completed to define allowed preferred operating ranges to be defined as shown in Table 2.
TABLE-US-00002 TABLE 1 Flow rate, temperature and compositions generated using PRO 2 for the pilot plant case. The hydrocarbon feed in this case is LLCN and the stripping gas is pentane. PCN represents propionitrile and Py is pyrrole. LLCN clean up stream Stripping C5 stream PC streams Raw Stripper Distillation LLCN Raffinate Clean C5 Pentane Overheads Solvent Extract Bottom 1 4 8 12 13 3 5 7 Flow rate kg/h 1.63 1.61 1.42 0.71 0.92 0.51 0.53 0.19 Temperature .degree. C. 40 40 33 20 40 40 40 69 Compositions C4- wt. % 1.1 1.1 1.2 0.0 0.0 0.0 0.1 0.0 C5 sats wt. % 36.6 36.6 40.3 80.4 63.1 0.5 1.6 16.1 C5 olefins wt. % 52.5 51.8 58.5 19.6 19.8 0.2 4.0 14.3 C6+ wt. % 9.9 9.9 0.0 0.0 16.6 0.0 0.5 65.6 PC wt. % 0.0 0.6 0.0 0.0 0.4 99.3 93.8 4.0 PCN wt. % 48.8 0.8 0.5 0.0 80.7 8.2 154.4 2.6 Py wt. % 0.1 0.1 0.0 0.0 0.1 1.4 1.2 0.5
TABLE-US-00003 TABLE 2 Possible operating ranges and preferred conditions. Parameter Range Preferred Range Extractor Pressure 2-10 barg 2-4 barg Extractor Tem 10-70.degree. C. 40-50.degree. C. Distillation Pressure 1-5 barg 1.5-3 barg Distillation feed Temp 30-80.degree. C. 40-50.degree. C. Distillation top temp 20-70.degree. C. 45-55.degree. C. Distillation bottom temp 50-100.degree. C. 70-85.degree. C. Distillation reflux flow 1-5 times mass 2-3 times mass feed rate feed rate Stripper feed temp 50-130.degree. C. 95-105.degree. C. Stripper top temp 30-80.degree. C. 55-65.degree. C. Stripper bottom temp 100-150.degree. C. 125-135.degree. C. Stripper reflux 2-100% of net 25-35% HC feed PC circulation rate 10-200 wt % of 10-30 wt % of hydrocarbon hydrocarbon feed rate feed rate Stripping gas rate 10-100 wt % of 70-90 wt % of C5 paraffin C5 paraffin feed feed
Example 3
[0126] This example demonstrates the impact of additional reflux at the stripper column.
[0127] In table 3 the stripper tower chemical map is shown with a reflux flow of 34% of net HC feed. There is a concentration of 890 ppmw of PC in the tower overheads. In table 4 the chemical map is shown with a reflux flow of 17% of the net HC feed and there is a concentration of 2320 ppm of PC in the tower overheads. However, the concentration of propionitrile (PCN) in the overheads in table 1 shows that the stripping is no longer effective at this higher reflux flow. These data points clearly demonstrate the PC content of the stripper overheads can be tuned based on reflux flow to the stripper, but the amount of reflux is constrained based on effective stripping.
TABLE-US-00004 TABLE 3 Stripper stream concentrations with a reflux of 8T/h- compositions generated using PRO 2 process simulation software Reflux 34 % of overhead flow wt % Stripper Overheads Stripper Bottoms PC 0.089 98.414 PCN 0.000 0.014 PYRROLE 0.000 0.000 <C4 0.065 0.000 C5 77.490 1.347 C6+ 22.355 0.225
TABLE-US-00005 TABLE 4 Stripper stream concentrations with a reflux of 4T/h- compositions generated using PRO 2 process simulation software Reflux 17 % of overhead flow wt % Stripper Overheads Stripper Bottoms PC 0.232 98.809 PCN 0.016 0.000 PYRROLE 0.000 0.000 <C4 0.065 0.000 C5 77.384 1.191 C6+ 22.302 0.000
[0128] The data shown in table 5 demonstrates that PC content in the tower overheads can be tuned using reflux. This also goes some way to showing the cutoff point beyond which additional reflux flow will lead to ineffective stripping.
TABLE-US-00006 TABLE 5 Stripper reflux flow versus tower performance- compositions generated using PRO 2 process simulation software. Reflux flow as % Stripper Overheads Stripper Bottoms of overhead flow PC (wt %) PCN (wt %) 0 1.0000 -- 13 0.3342 1.20E-07 17 0.233 2.77E-07 21 0.1628 9.59E-07 25 0.1145 1.78E-05 29 0.0898 0.0109 34 0.0886 0.0155 38 0.0894 0.0307
[0129] The amount of propionitrile in the tower bottoms indicates the effectiveness of the tower in stripping contaminants whereas the PC in the overheads demonstrates the effectiveness of the reflux in limiting PC loss.
* * * * *
D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.