Compositions And Methods For Treating Hair Loss
COTSARELIS; George ; et al.
U.S. patent application number 16/260034 was filed with the patent office on 2020-06-25 for compositions and methods for treating hair loss. This patent application is currently assigned to The Trustees of the University of Pennsylvania. The applicant listed for this patent is The Trustees of the University of Pennsylvania. Invention is credited to George COTSARELIS, Oh Sang Kwon.
| Application Number | 20200197488 16/260034 |
| Document ID | / |
| Family ID | 42170304 |
| Filed Date | 2020-06-25 |










View All Diagrams
| United States Patent Application | 20200197488 |
| Kind Code | A1 |
| COTSARELIS; George ; et al. | June 25, 2020 |
COMPOSITIONS AND METHODS FOR TREATING HAIR LOSS
Abstract
The present invention provides methods for treating hair loss, treating, inhibiting, or suppressing a degenerative skin disorder, treating androgenetic alopecia (AGA), generating new hair follicles (HF), and increasing the size of existing HF. The methods comprise epidermal disruption or administration of wnt, a fibroblast growth factor-9 polypeptide or another compound that upregulates sonic hedgehog gene signaling.
| Inventors: | COTSARELIS; George; (Berwyn, PA) ; Kwon; Oh Sang; (Seoul, KR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | The Trustees of the University of
Pennsylvania Philadelphia PA |
||||||||||
| Family ID: | 42170304 | ||||||||||
| Appl. No.: | 16/260034 | ||||||||||
| Filed: | January 28, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16004277 | Jun 8, 2018 | |||
| 16260034 | ||||
| 14505970 | Oct 3, 2014 | |||
| 16004277 | ||||
| 13129100 | Aug 2, 2011 | 8871711 | ||
| PCT/US09/64049 | Nov 11, 2009 | |||
| 14505970 | ||||
| 16222705 | Dec 17, 2018 | |||
| 13129100 | ||||
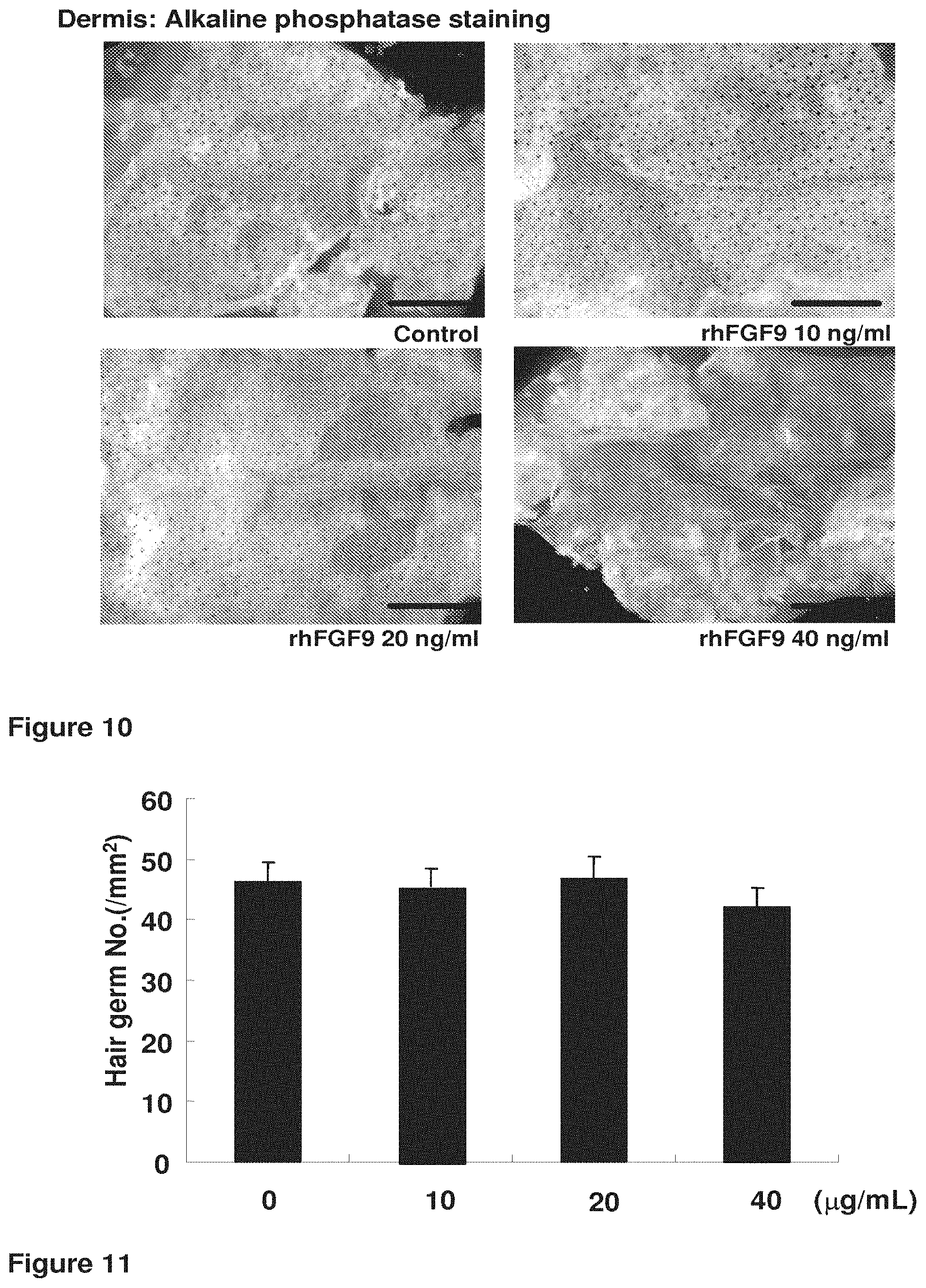
| 14946512 | Nov 19, 2015 | |||
| 16222705 | ||||
| 13327611 | Dec 15, 2011 | 9220926 | ||
| 14946512 | ||||
| 11887104 | Sep 25, 2007 | |||
| PCT/US2006/011319 | Mar 28, 2006 | |||
| 13327611 | ||||
| 61114028 | Nov 12, 2008 | |||
| 60665857 | Mar 29, 2005 | |||
| 60683293 | May 23, 2005 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61N 2005/1098 20130101; A61K 38/1825 20130101; A61M 37/00 20130101; A61P 43/00 20180101; A61P 17/00 20180101; A61P 35/00 20180101; A61K 8/64 20130101; Y10S 514/88 20130101; A61K 45/06 20130101; A61N 5/10 20130101; A61Q 7/00 20130101; A61M 2037/0007 20130101; A61P 17/14 20180101 |
| International Class: | A61K 38/18 20060101 A61K038/18; A61N 5/10 20060101 A61N005/10; A61M 37/00 20060101 A61M037/00; A61K 45/06 20060101 A61K045/06; A61Q 7/00 20060101 A61Q007/00; A61K 8/64 20060101 A61K008/64 |
Goverment Interests
GOVERNMENT INTEREST STATEMENT
[0002] This invention was made with government support under grant number AR046837 awarded by the National Institutes of Health. The United States government has certain rights in the invention.
Claims
1. A method of treating hair loss in a subject, the method comprising: administering an effective amount of a sonic hedgehog (Shh) agonist to said subject, thereby treating said hair loss in said subject.
2. The method of claim 1, wherein the method comprising the steps of: wounding a region of said hair loss in said subject; and administering said effective amount of said sonic hedgehog (Shh) agonist to the wounded area of said subject.
3. The method of claim 2, wherein the step wounding is performed by disrupting a dermis or an epidermis in the region of said hair loss in said subject.
4. The method of claim 1, wherein said sonic hedgehog (Shh) agonist is Hh-Ag.
5. The method of claim 1, wherein the subject is a human.
6. The method of claim 1, wherein the subject has hair loss.
7. The method of claim 1, wherein said hair loss is due to androgenetic alopecia (AGA).
8. The method of claim 7, wherein the AGA is male pattern baldness or female pattern baldness.
9. The method of claim 1, wherein said hair loss is due to skin injury.
10. The method of claim 1, wherein said hair loss is in the scalp or eyebrow of said subject.
11. The method of claim 7, wherein said AGA is in the scalp or eyebrow.
12. The method of claim 1, wherein said hair loss is in scarred skin tissue of said subject.
13. The method of claim 3, wherein said disrupting is performed by exposing the region of said hair loss to a mechanical or chemical stimulus.
14. The method of claim 3, wherein said disrupting is performed by exposing the region of said hair loss to radiation.
15. A method of increasing the number of hair follicles in a subject, the method comprising: administering an effective amount of a sonic hedgehog (Shh) agonist to said subject, thereby treating increasing the number of hair follicles in said subject.
16. The method of claim 15, wherein the method comprising the steps of: wounding a region of said hair loss in said subject; and administering said effective amount of said sonic hedgehog (Shh) agonist to the wounded area of said subject.
17. The method of claim 16, wherein the step wounding is performed by disrupting a dermis or an epidermis in the region of said hair loss in said subject.
18. The method of claim 15, wherein said sonic hedgehog (Shh) agonist is Hh-Ag.
19. The method of claim 15, wherein the subject is a human.
20. The method of claim 15, wherein the subject has hair loss.
21. The method of claim 20, wherein said hair loss is due to androgenetic alopecia (AGA).
22. The method of claim 21, wherein the AGA is male pattern baldness or female pattern baldness.
23. The method of claim 20, wherein said hair loss is due to skin injury.
24. The method of claim 20, wherein said hair loss is in the scalp or eyebrow of said subject.
25. The method of claim 21, wherein said AGA is in the scalp or eyebrow.
26. The method of claim 20, wherein said hair loss is in scarred skin tissue of said subject.
27. The method of claim 17, wherein said disrupting is performed by exposing the region of said hair loss to a mechanical or chemical stimulus.
28. The method of claim 17, wherein said disrupting is performed by exposing the region of said hair loss to radiation.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation application of U.S. patent application Ser. No. 16/004,277, filed Jun. 8, 2018, which is a continuation application of U.S. patent application Ser. No. 14/505,970, filed Oct. 3, 2014, which is a continuation application of U.S. patent application Ser. No. 13/129,100, filed Aug. 2, 2011, now issued as U.S. Pat. No. 8,871,711, which is a National Phase Application of PCT International Application PCT/US09/64049, filed Nov. 11, 2009, which claims priority to and the benefit of U.S. Provisional Patent Application 61/114,028, filed Nov. 12, 2008. This application is also a continuation application of U.S. patent application Ser. No. 16/222,705, filed Dec. 17, 2018, which is a continuation of U.S. patent application Ser. No. 14/946,512, filed on Nov. 19, 2015, which is a continuation of U.S. patent application Ser. No. 13/327,611, filed on Dec. 15, 2011, which is a continuation of U.S. patent application Ser. No. 11/887,104, filed Sep. 25, 2007, which is a National Phase Application of PCT International Application PCT/US06/11319, filed Mar. 28, 2006, claiming priority to U.S. Provisional Patent Applications 60/665,857 and 60/683,293, filed 29 Mar. 2005, and 23 May 2005, respectively. Additionally, this application relates to U.S. patent application Ser. Nos. 12/904,822 and 12/904,981, both filed on Oct. 14, 2010. All of the above-identified applications are incorporated by reference herein in their entirety.
FIELD OF THE INVENTION
[0003] The invention relates to pharmaceutical compositions and methods for treating hair loss or regenerating hair follicles.
BACKGROUND OF THE INVENTION
[0004] Follicular neogenesis is defined as the generation of new hair follicles (HF) after birth. Humans are born with a full complement of HF, which can change in size and growth characteristics as in early baldness or can ultimately degenerate and disappear as in late stages of baldness or in permanent scarring (cicatricial) alopecias. Therefore, the generation of new HF is desirable in the treatment of common baldness as well as less common hair loss conditions, such as discoid lupus erythematosis, congenital hypotrichosis, lichen planopilaris and other scarring alopecias.
SUMMARY OF THE INVENTION
[0005] The present invention provides methods of treating hair loss, treating, inhibiting, or suppressing a degenerative skin disorder, and treating androgenetic alopecia (AGA) in a subject and generating new hair follicles (HF) and increasing the size of existing HF, comprising epidermal disruption or administration of wnt, and administration of a fibroblast growth factor-9 polypeptide or another compound that upregulates sonic hedgehog gene signaling.
[0006] Thus, in one embodiment, the present invention provides a method of treating hair loss in a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0007] In one embodiment, the hair loss is due to androgenetic alopecia (AGA). In one embodiment, the AGA is male pattern baldness. In another embodiment, the AGA is female pattern baldness. In one embodiment, the hair loss is the result of a skin injury. In one embodiment, the hair loss is in the scalp or eyebrow of said subject. In one embodiment, the hair loss is in scarred skin tissue of said subject. In one embodiment, the step of administering is performed 3-12 days after said step of disrupting. In one embodiment, the step of disrupting is performed by exposing the region of said hair loss to a mechanical, chemical, or optical stimulus. In one embodiment, the optical stimulus is radiation. In one embodiment, the administering step is via topical administration. In another embodiment, the administering step is via subepidermal administration.
[0008] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0009] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0010] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0011] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the steps of (a) disrupting the epidermis in the region of said degenerative skin disorder in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0012] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the steps of (a) disrupting the epidermis in the region of said AGA in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0013] In another embodiment, the present invention provides a method of treating hair loss in a subject comprising the step administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0014] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0015] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0016] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0017] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0018] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0019] In another embodiment, the invention provides a method of treating hair loss in a subject, the method comprising: administering an effective amount of a compound or factor that upregulates sonic hedgehog (SHH) (e.g., SHH agonist) to said subject, thereby treating said hair loss in said subject. In some embodiments, the method comprises the steps of: wounding a region of said hair loss in said subject; and administering said effective amount of said SHH agonist to the wounded area of said subject.
[0020] In another embodiment, the invention provides a method of increasing the number of hair follicles in a subject, the method comprising: administering an effective amount of a compound or factor that upregulates sonic hedgehog (SHH) (e.g., SHH agonist) to said subject, thereby treating increasing the number of hair follicles in said subject. In some embodiments, the method comprises the steps of: wounding a region of said hair loss in said subject; and administering said effective amount of said SHH agonist to the wounded area of said subject.
[0021] In another embodiment, the present invention provides a method of treating hair loss in a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0022] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0023] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0024] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0025] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0026] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0027] Other features and advantages of the present invention will become apparent from the following detailed description examples and figures. It should be understood, however, that the detailed description and the specific examples while indicating preferred embodiments of the invention are given by way of illustration only, since various changes and modifications within the spirit and scope of the invention will become apparent to those skilled in the art from this detailed description.
BRIEF DESCRIPTION OF THE DRAWINGS
[0028] FIG. 1. FGF9 is expressed during inductive period of hair follicle regeneration at Day 1 after scab detachment (SD). The ratio of FGF9 mRNA compared to control mRNA expression q-PCR of FGF9 mRNA expression in regenerated epidermis is presented.
[0029] FIG. 2. .gamma..delta.TCR immunostaining of regenerated epidermis (SD7, wholemount) (.times.200) and FGF9 immunostaining of SD1 sample (frozen section) (.times.400).
[0030] FIG. 3. .gamma..delta.TCR & FGF9 immunostaining of regenerated epidermis for SD1 sample.
[0031] FIG. 4. .gamma..delta.TCR & FGF9 immunostaining of E14 embryonic skin.
[0032] FIG. 5. Specificity of anti-FGF9 neutralization antibody for E14.5 mouse embryonal whole lysate (lanes 1 and 2) and for recombinant hFGF9 (+).
[0033] FIGS. 6A-6C. Anti-FGF9 neutralization experiment in 3 week-old C57BL/6 mice. (FIG. 6A) Treatment schedule in which 50 .mu.l of 10 .mu.g/ml anti-FGF9 or IgG2a isotype control were injected subepidermally on scab detachment day (SD)1-SD4, and tissue was sampled at SD5. (FIG. 6B) Hair follicle numbers after anti-FGF9 or IgG2a control injections in mice using the treatment protocol described in (FIG. 6A). (FIG. 6C) Diagram showing injection site.
[0034] FIG. 7. Hair follicle number in anti-FGF9-treated mice vs controls at various stages of hair follicle development, as described in Paus R, et al. J Invest Dermatol 1999).
[0035] FIG. 8. Model showing how hair germ counting was conducted per mm.sup.2 at 3 different fields per each sample.
[0036] FIG. 9. rhFGF9 treatment for three days in embryonic skin explant culture (E13.5). Cultures were treated with 10, 20, or 40 ng/mL of rhFGF9 or control buffer for three days, and hair germ number/mm.sup.2 was evaluated as described in FIG. 8. Mean.+-.SD. *: P<0.05, **: P<0.01, compared to control. EDA-A1 (50 ng/ml) was used as a positive control for hair germ number.
[0037] FIG. 10. Immunohistochemical staining showing alkaline phosphatase staining of the dermis in control and rhFGF9 (10, 20, 40 ng/ml)-treated embryonic skin explants.
[0038] FIG. 11. Anti-FGF9 neutralizing antibody treatment for three days in embryonic skin explant culture (E13.5). Cultures were treated with 10, 20, or 40 .mu.g/mL of anti-FGF9 neutralizing antibody or control for three days, and hair germ number/mm.sup.2 was evaluated as described in FIG. 8.
[0039] FIG. 12. Immunohistochemical staining showing K17 staining of the epidermis in control and anti-FGF9 (10, 20, 40 .mu.g/ml)-treated skin explants.
[0040] FIG. 13. Immunohistochemical staining showing alkaline phosphatase staining of the dermis in control and anti-FGF9 (10, 20, 40 .mu.g/ml)-treated embryonic skin explants.
[0041] FIG. 14. Effect of 24 h treatment using rhFGF9 (10, 20, 40 ng/ml) on markers of embryonic hair follicle development sonic hedgehog (Shh), Ptch1, and Ptch2 by qPCR.
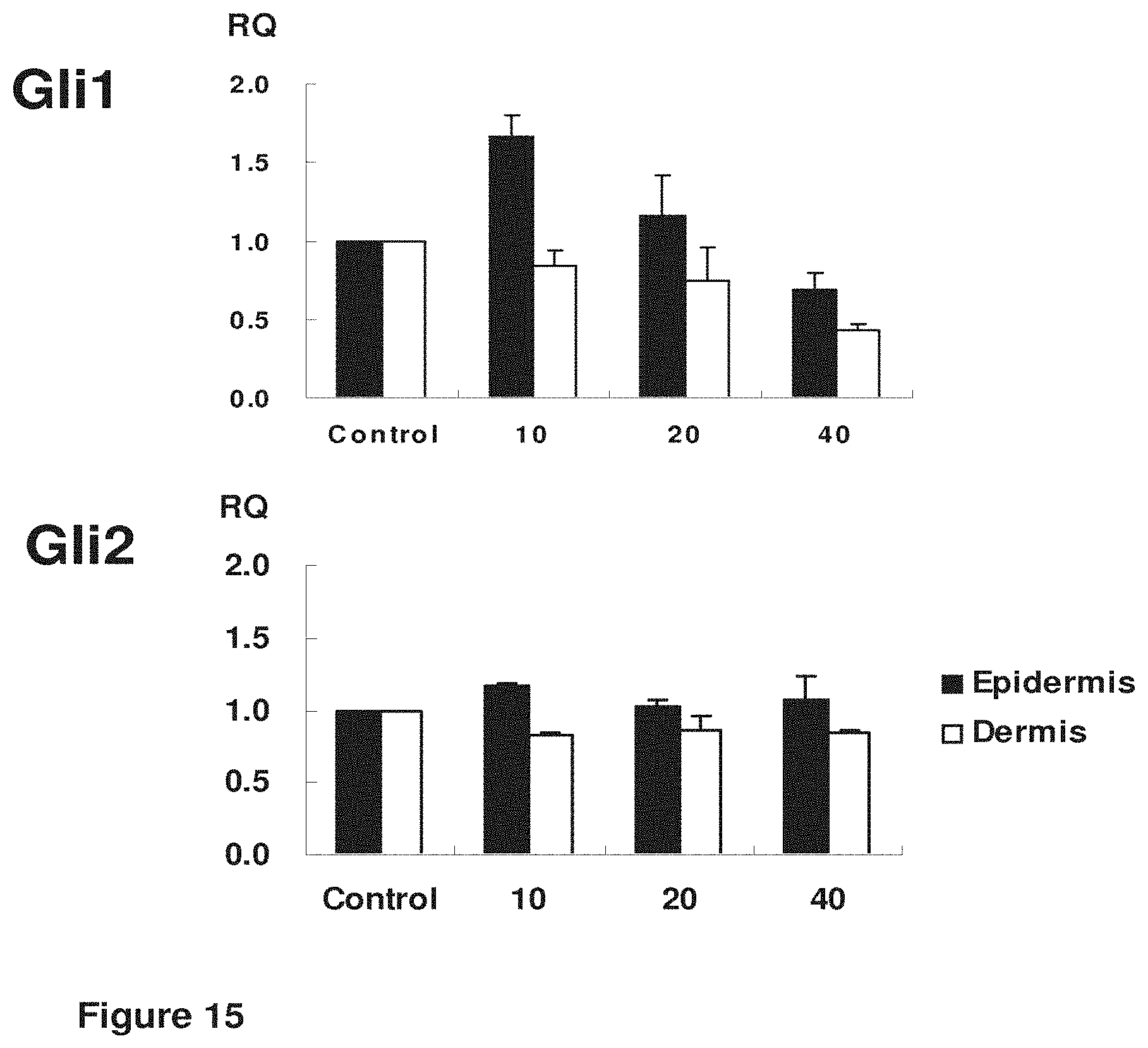
[0042] FIG. 15. Effect of 24 h treatment using rhFGF9 (10, 20, 40 ng/ml) on markers of embryonic hair follicle development Gli1 and Gli2 by qPCR.
[0043] FIGS. 16A-16H. Fgf9 is expressed during HFN initiation and is important to HFN. (FIG. 16A) Fgf9 is highly expressed in regenerated epidermis prior to hair follicle formation at Day 1 and 3 after reepithelization with scab detachment (SD) and decreased to basal level at Day 5. The ratio of Fgf9 mRNA expression in regenerated epidermis was compared to the level of unwounded epidermis, Day 0. *: P<0.05, **: P<0.01, mean.+-.standard deviation. (FIG. 16B) Effect of FGF9 neutralization on HFN after wounding in 3-week old mice. The number of regenerated hair follicles was significantly decreased in the mice treated with anti-FGF9 neutralizing antibody compared to controls. **: P<0.05 (FIG. 16C) Determination of developmental stages of hair follicles. Hair follicles in the anti-FGF9-treated mice showed delay in hair follicle maturation. (C-E) Wholemount hair follicle neogenesis assay stained for KRT17 protein and (F-H) alkaline phosphatase activity in separated epidermis and dermis at Day 5 after reepithelization, respectively. Overexpression of Fgf9 in K14rtTA; TRE-Fgf9-IRES-eGfp mice resulted in increased numbers of hair follicles at Day 17 after wounding. Scale bar, 1 mm.
[0044] FIG. 17. FGF9 is expressed by activated DETC (A) Double immunostaining for FGF9 and .gamma..delta. TCR. Fgf9expression in repopulated .gamma..delta. TCR-positive DETCs after reepithelization. Dot line, basement membrane. Scale bar, 50 .mu.m. (B) Fgf9 gene expression is highly upregulated in the isolated DETCs after activationwith mIL-2 and anti-CD3.
[0045] FIGS. 18E-18H. Hair follicle neogenesis in TCRd-/- mice is severely impaired. (A-D) Wholemount epidermal and dermal samples treated to detect KRT17 protein and (E-H) alkaline phosphatase activity at Day 5 after reepithelization. Hair follicle formation was significantly impeded in 8 week and 40 week old mice. Scale bar, 1 mm. *: P<0.05, mean.+-.standard error.
[0046] FIG. 19. Developmental stages of HFs in control and anti-fgf9 antibody-treated wounds.
[0047] FIG. 20. FGF9 expression in K14rtTA; TRE-fgf9-IRES-eGfp mice compared to control mice during 2 days of doxycycline treatment.
[0048] FIGS. 21A-21C. FGF9 expression in DETCs. (A) FGF9 is highly expressed in suprabasal dendritic cell. Wholemount double immunostaining of FGF9 and .gamma..delta. TCR of ear epidermis from wild-type mouse without (B) or with IL-2 incubation (C).
[0049] FIG. 22. FGF9 expression in FGF9.sup.flox/flox;lck-cre mice compared to FGF9.sup.flox/flox control.
[0050] FIGS. 23A-23J. Shh signaling is necessary for HFN following injury. a X-gal staining of large wound (LW) and small wound (SW) from of Gli1-LacZ mice at indicated time (n=2-4 W (2-5M) per condition). b qRT-PCR for Shh expression in SW and LW of wild-type mice at 7 days after complete re-epithelialization (n=2-6 W(2 M) per condition). c Immunohistochemistry of Shh on SW and LW of wild-type mice at 7 days after complete re-epithelialization. *Indicates nonspecific signals. d-f K14-CreER; Shh fl/fl (K14-Shh fl/fl) and littermate control mice were subjected to large wound (LW) and treated with TAM from PW3d until tissue harvest at PW21d (n=11-12 W (11-12 M) per condition). Whole-mount HFN assay (d) and quantifications (e, f). g-j Pdgfra-CreER; Smo fl/fl (Pdgfra-Smo fl/fl) and littermate controls were subjected to large wound (LW) and were treated with TAM from 3 or 4 weeks before wounding until tissue harvest at PW21d (n=3-5 W (3-5M) per condition). Whole-mount HFN assay (g, i) and quantifications (h, j). n: number of wounds (W) or mice (M), Data are represented as mean.+-.s.d., *p<0.05; **p<0.01; ***p<0.001; Student's t-test, Dashed white circle: wound boundary, dashed line: epidermis-dermis border, DP dermal papilla, AP alkaline phosphatase, FE follicular epithelium, PW post-wound, Scale bars represent 500 .mu.m (a (whole mount), d, g, i), 50 .mu.m (a (section), c)
[0051] FIGS. 24A-24K. Epidermal Shh is capable of regenerating HF in wounds without alteration of collagen. a, b K14-CreER; LSL-Shh; Gli1-LacZ (K14-Shh-Gli1-LacZ) and littermate controls with Gli1-LacZ were subjected to SW and treated with TAM from PW1d until tissue harvest at indicated time (n=12 W (4M) per condition). X-gal staining was analyzed at PW16d (a), and PW35d (b). Arrowheads show regenerated HFs (b). c-e Distribution of regenerated DP was analyzed from three representative LWs of K14-CreER; LSL-Shh (K14-Shh) and littermate controls treated with TAM from PW3d until tissue harvesting at PW35d (n=9 W(9 M) per condition). AP+DP in each picture were converted into dots with three different colors (red, green, and cyan) and merged into one picture (c, right). The original images and the corresponding colors represented in three columns on the left (c). Quantifications of AP+DP per wound (d) and area occupied by DP (e). "Area occupied by DP" was defined by drawing a line that connects the outermost regenerated DP in wound. f-k K14-CreER; LSL-Shh (K14-Shh) and littermate controls were subjected to SW and treated with TAM from PW1d until tissue harvest at indicated time. RNA-seq analyses at PW11d showing heatmap of differentially expressed genes for K14-Shh and control mice (f, g). Red and green correspond to high and low expression levels, respectively (n=12 W (4M) per condition). Hydroxyproline assay to measure collagen contents at PW11d (n=12 W (4M) per condition) (h). Diameter of collagen fiber at PW30d (TEM images on the left, n=2 W (2 M) per condition) (i). Detection of collagen fiber by Masson's trichrome staining (j) and Picrosirius red staining (k) at PW1 d and PW35d (insets). n: number of wounds (W) or mice (M), Data are represented as mean.+-.s.d., **p<0.01; ***p<0.001; ns: non-significant; Student's t-test, Zigzag line and dashed white circle: wound boundary, Dashed line: epidermis-dermis border, SW small wound, LW large wound, PW post-wound, DP dermal papilla, AP alkaline phosphatase, FE follicular epithelium. Scale bars represent 500 .mu.m (b (whole mount), c), 50 .mu.m (b (section), j, k), 10 .mu.m (a), 100 nm (i).
[0052] FIGS. 25A-25N. Dermal Hh activation is sufficient to promote HFN in non-regenerative wounds. A Detection of tomato reporter on SW of SM22-rtTA; tetO-Cre; R26-Tomato mice at indicated time. b-n SM22-rtTA; tetO-Cre: R26-SmoM2 (SM22-SmoM2) and littermate controls were subjected to SW and treated with doxycycline from PW1d until tissue harvest at PW30d (n=18 W (4-5M) per condition). Whole-mount HFN assay (b, d) and quantifications (c, e). Percentage of AP+DP with K17+FE (f). Percentage of K17+HF with hair shaft (HS) (g). Immunohistochemistry with indicated markers (h-m) and H&E (n). n: number of wounds (W) or mice (M), Data are represented as mean.+-.s.d., *p<0.05; Student's t-test, Dashed white circle: wound boundary, Dashed line: epidermis-dermis border, SW small wound, PW post-wound, DP dermal papilla, AP alkaline phosphatase, FE follicular epithelium, HF hair follicle, HS hair shaft, Bu bulge stem cell area, SG sebaceous gland. Scale bars represent 500 m (b, d), 100 .mu.m (n (Control)), 10 .mu.m (a, h-m, n (SM22-SmoM2)
[0053] FIGS. 26A-26J. Dermal Hh activation induces DP fate in wound fibroblasts. scRNA-seq was performed with Tomato+ cells isolated from wound dermis of both SM22-rtTA; tetO-Cre; R26 SmoM2/Tomato (SM22-SmoM2) and SM22-rtTA; tetO-Cre; R26-Tomato (control) mice 3 days after complete re-epithelialization. The mice were subject to SW and treated with doxycycline from PW1d until PW12d (n=12-25 W(3-5M) per condition). a-i tSNE plots of 4680 SM22+ dermal cells split between control and Hh activated conditions. tSNE plot of SM22+ dermal cells colored by assigned lineages (a). tSNE plot of SM22+ dermal cells according to lineage-specific markers (b-f). tSNE plot of SM22+ dermal cells according to Hh pathway components (g). tSNE plot of SM22+ myofibroblasts according to Hh pathway components (h). tSNE plot of SM22+ myofibroblasts according to cell origin (i). j Heatmap showing the expression of DP signature genes. Yellow and black/purple correspond to high and low expression levels, respectively.
[0054] FIGS. 27A-27L. Dermal Wnt activation alone is not sufficient for HFN. a Violin plots of Wnt pathway related genes in SM22+ myofibroblasts. b X-gal staining in epidermis and dermis of SW of Axin2-LacZ mice at PW10d and PW32d. c-h SM22-rtTA; tetO-Cre; .beta.-catenin fl(ex3)/+(SM22-ex3) and littermate controls were subjected to SW and treated with doxycycline from PW1d until tissue harvest at PW30d (n=3-4 W (3-4M) per condition). Whole-mount HFN assay (c) and quantifications (d, e). H&E (f, g) and AP/Lef1 staining (h) show lack of hair germ (HG) formation by .beta.-catenin activation in dermis. i-l SM22-rtTA; tetO-Cre; Wls fl/fl (SM22-Wls fl/fl) and littermate controls were subjected to LW and treated with doxycycline from PW3d until tissue harvest at PW21d (n=4-5 W (4-5M) per condition). Whole-mount HFN assay (i, k) and quantifications (j, 1). n: number of wounds (W) or mice (M), Dashed white circle and zigzag line: wound boundary, Dashed line: epidermis-dermis border, SW small wound, LW large wound, AP alkaline phosphatase, PW postwound, Scale bars represent 500 .mu.m (b, c, i, k), 100 .mu.m (f), 50 .mu.m (g, h).
[0055] FIGS. 28A-28F. Hh activation can convert fibrotic Wnt-active dermal cells into DP in wounds. a-d Axin2 CreER; R26-SmoM2 (Axin2-SmoM2) and littermate controls were subjected to SW and treated with TAM from PW1d until tissue harvest at PW30d (n=9-15 W (3-5M) per condition). Whole-mount HFN assay (a) and quantifications (b, c). H&E and immunohistochemical analyses with indicated markers (d). e Tracing of tomato-labeled Axin2+ cells in Axin2-CreER; R26-Tomato (Axin2-Tom). Axin2-Tom mice were subjected to LW and treated with TAM from PW3d until PW12d (before complete re-epithelialization). Tissue was harvested at PW23d and stained with anti-RFP antibody. f Model: Conversion of wound repair to regeneration in adult skin. Wound healing in mammalian skin typically results in scarring and lack of appendage regeneration. Dermal Hh activation can install de novo dermal papilla into wounds, resulting in regenerative HF neogenesis, despite collagen deposition in adult wounds. n: number of wounds (W) or mice (M), Data are represented as mean.+-.s.d., **p<0.01, ***p<0.001; Student's t-test, Dashed white circle: wound boundary, Dashed line: epidermis-dermis border, SW small wound, LW large wound, PW post-wound, DP dermal papilla, AP alkaline phosphatase, FE follicular epithelium, HF hair follicle, Scale bars represent 500 .mu.m (a), 10 .mu.m (d, e).
[0056] FIG. 29. C57B6, wounded at P21, followed by subcutaneous injection of Hh-Ag from PWD5 to PWD8 increases hair follicle formation.
[0057] FIG. 30. Cultured human and mouse dermal cells treated with Shh agonist (i.e., Hh-Ag) showed increased hair follicle (HF) number, relative to control.
[0058] FIG. 31. Mouse dermal cells infected with active Smo virus at PO result in more HF in recon assay. Cultured mouse dermal cells transduced with Smo plus mouse epi.
[0059] FIG. 32. Mouse dermal cells infected with active Smo virus at P2 result in more HF in recon assay. P2 cells typically lose activity as seen in control. Smo transduction maintains inductivity.
[0060] FIG. 33. DP and DS cells in HFs are made from cultured dermal cells (RFP+) infected with activated Smo.
[0061] FIG. 34. Quantitation of recon assay of cultured mouse dermal cells treated with Shh Ag or infected with active Smo virus. Number of hair formed per assay was significantly higher in Shh agonist, Hh-Ag, treated cells, relative to control and Smo virus infected cells.
[0062] FIG. 35. Foreskin dermal cells were infected with active Smo. Human foreskin dermal cells have no hair inducing activity but activated smo induces them to promote HF formation.
[0063] FIG. 36. Dose dependent response. High concentration of Hh-Ag inhibited HF formation.
[0064] FIG. 37. Mouse neonatal dermal cells were cultured from PO to P2.
[0065] FIG. 38. Hair follicles in recon assay using cultured mouse neonatal dermal cells.
[0066] FIG. 39. Cultured dermal cells from Gli1-Lacz mouse.
[0067] FIG. 40. The percentage of Gli1-Lacz positive cells decreased in culture even in the presence of Hh-Ag (See Figure X). Culturing dermal cells in the presence of Hh-Ag does not maintain the Shh responding population. 24 hr Shh treatment showed fewer Gli1 positive cells but had similar number of HFs in patch assay compared to 7 day treatment.
[0068] FIG. 41. Blank-blank best images
[0069] FIG. 42. Blank-blank histology.
[0070] FIG. 43. Blank-SHH best images
[0071] FIG. 44. Blank-SHH histology.
DETAILED DESCRIPTION OF THE PRESENT INVENTION
[0072] The invention relates to pharmaceutical compositions and methods for treating hair loss and regenerating hair follicles. Specifically, the invention relates to fibroblast growth factor-9 polypeptides and administering a fibroblast growth factor-9 polypeptide for treating hair loss or regenerating hair follicles.
[0073] The present invention provides methods of treating hair loss, treating, inhibiting, or suppressing a degenerative skin disorder, and treating androgenetic alopecia (AGA) in a subject and generating new hair follicles (HF) and increasing the size of existing HF, comprising epidermal disruption or administration of wnt, and administration of a fibroblast growth factor-9 polypeptide or another compound that upregulates sonic hedgehog gene signaling.
[0074] In one embodiment, the present invention provides methods of treating hair loss, methods for generating a hair follicle, methods for increasing the size of a hair follicle, methods for treating an androgenetic alopecia (AGA), methods for arresting alopecia, methods of reversing alopecia, and methods of depilation comprising administering a composition comprising a neutralizing fibroblast growth factor-9 antibody to a subject.
[0075] In another embodiment, a composition or method of the present invention is utilized on human skin. In another embodiment, the composition or method is utilized on an area of unwanted hair growth. In another embodiment, the area is the face. In another embodiment, the area is the bikini area. In another embodiment, the area is the legs. In another embodiment, the area is the arms. In another embodiment, the area is the chest.
[0076] In one embodiment, the methods of the present invention include contacting a subject with an inhibitor of FGF9, SHH, WNT, or other compositions for use in the present invention. An "inhibitor" utilized in methods and compositions of the present invention is, in another embodiment, an antibody that binds the protein or biological factor that is the target of the inhibitor. In another embodiment, the inhibitor is a pharmacologic inhibitor. In another embodiment, the inhibitor is any other type of inhibitor known in the art. Each possibility represents a separate embodiment of the present invention.
[0077] In one embodiment, the present invention provides a method of treating hair loss comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide to a subject.
[0078] In one embodiment, the present invention provides a method of treating hair loss in a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0079] In another embodiment, the present invention provides a method of treating hair loss in a subject comprising the step administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0080] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the steps of (a) disrupting the epidermis in the region of said AGA in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0081] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0082] In another embodiment, the present invention provides a method of treating hair loss in a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0083] In one aspect, the invention relates to a method of treating a hair loss in a subject. In another aspect, the invention relates to a method of increasing the number of hair follicles in a subject. The method comprises the step of administering an effective amount of a sonic hedgehog (SHH) agonist to the subject. In some embodiments, the method comprises the steps of: wounding a region of the hair loss and administering the SHH agonist to the wounded region. The wounding step can be performed, for example, by disrupting a dermis or an epidermis in the region of the hair loss.
[0084] Any SHH agonist known to one of skilled in the art can be used. Examples of Shh agonists include, but not limited to, Hh-Ag, Purmorphamine, SAG, and 20(S)-Hydroxycholesterol.
[0085] In a particular embodiment, the SHH agonist is Hh-Ag. Hh-Ag (also known as Hh-Ag1.5) is a small-molecule chemical agonist of Smoothened (Smo) receptor and is an activator of sonic hedgehog (Shh) signaling. It is derived from an initial synthetic hit compound "Hh-Ag1.1", and optimized to achieve the activity IC50=1 nM for agonist activity. This molecule is commercially available for research use and being sold by Cellagentech, Inc. Its molecular formula is C.sub.28H.sub.26N.sub.3OS, CAS number is 612542-14-0, and chemical name is 3-chloro-4,7-difluoro-N-(4-(methylamino)cyclohexyl)-N-(3-(pyridin-4-yl)be- nzyl)benzo[b]thiophene-2-carboxamide.
[0086] The region of the hair loss includes, for example, but not limited to, scalp, eyebrow, and scar. The hair loss may occur due to various reasons, including, for example, but not limited to, androgenetic alopecia (AGA) and skin injury. In one embodiment, the AGA is a male pattern baldness. In another embodiment, the AGA is a female pattern baldness. In some embodiments, the AGA is in the scalp or eyebrow.
[0087] The step of disrupting can be performed by any suitable method known to one of skilled in the art. In one embodiment, the step of disrupting can be performed by exposing the region of the hair loss to a suitable mechanical stimulus, known to one of skilled in the art. In another embodiment, the step of disrupting can be performed by exposing the region of the hair loss to a suitable chemical stimulus, known to one of skilled in the art. In yet another embodiment, the step of disrupting can be performed by exposing the region of the hair loss to a suitable radiation, known to one of skilled in the art.
[0088] In another embodiment, the present invention provides a method for treating an androgenetic alopecia (AGA) in a scalp of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0089] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0090] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0091] In another embodiment, the present invention provides a method for generating a hair follicle in the dermis of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0092] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0093] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0094] In another embodiment, the present invention provides a method for increasing the size of a hair follicle in the dermis of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0095] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject with hair loss comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0096] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0097] In another embodiment, the present invention provides a method for increasing hair follicle formation in the skin of a subject comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0098] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the steps of (a) disrupting the epidermis in the region of said degenerative skin disorder in said subject and (b) administering a composition comprising a fibroblast growth factor-9 polypeptide to said subject.
[0099] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide and a wnt polypeptide to said subject.
[0100] In another embodiment, the present invention provides a method for treating, inhibiting, or suppressing a degenerative skin disorder comprising the steps of (a) disrupting the epidermis in the region of said hair loss in said subject and (b) administering a composition comprising a compound or factor that upregulates sonic hedgehog (SHH) to said subject.
[0101] In one embodiment, the methods of the present invention treat, inhibit or suppress a degenerative skin disorder. In one embodiment, a degenerative skin disorder is Hyperkeratosis, hyperpigmentation, depigmentation, atrophy, or a combination thereof. In one embodiment, a degenerative skin disorder is calcinosis; circumscripta; cutis; Colloid milium; skin degeneration; Senile dermatosis NOS; or Subcutaneous calcification.
[0102] In another embodiment, a degenerative skin disorder is granuloma annulare. In one embodiment, the degenerative skin disorder is localized granuloma annulare, which in one embodiment, is the most common form of granuloma annulare and in another embodiment, is characterized by the presence of small, firm red or yellow colored bumps (nodules or papules) that appear arranged in a ring on the skin. In one embodiment, the sizes of the lesions range from one to five centimeters. In one embodiment, the most commonly affected sites include the feet, hands, and fingers. In other embodiments, the degenerative skin disorder is generalized or disseminated, linear, perforating, or subcutaneous granuloma annulare. In one embodiment, the lesions associated with granuloma annulare may disappear without treatment (spontaneous remission) and reappear.
[0103] In another embodiment, the methods of the present invention are suitable for the prophylaxis and treatment of dryness, roughness of the skin, the formation of dry lines, reduced rehydration by sebaceous glands and an increased susceptibility to mechanical stress (tendency to crack), for the treatment of photodermatoses, the symptoms of senile xerosis, photoaging and other degenerative conditions which are associated with a decomposition of the connective tissue (collagen and elastin fibres and also glucosaminoglycans/hyaluronane) of the skin. "Photoaging" denotes the wrinkling, dryness and decreasing elasticity of the skin brought about by light and in particular UV radiation.
[0104] Further fields of application of the compositions according to the invention are the treatment and prevention of age- and/or UV-induced collagen degeneration and also the decomposition of elastin and glycosaminoglycans; of degenerative skin conditions such as loss of elasticity and also atrophy of the epidermal and dermal cell layers, of constituents of the connective tissue, of rete pegs and capillary vessels) and/or the skin adnexa; of environmentally-triggered negative changes in the skin and the skin adnexa, e.g. caused by ultraviolet radiation, smoking, smog, reactive oxygen species, free radicals and similar; of deficitary, sensitive or hypoactive skin conditions or deficitary, sensitive or hypoactive skin adnexa conditions; the reduction in skin thickness; of skin slackness and/or skin tiredness; of changes in the transepidermal water loss and normal moisture content of the skin; of a change in the energy metabolism of healthy skin; of deviations from the normal cell-cell communication in the skin which can manifest themselves e.g. in wrinkling; of changes in the normal fibroblast and keratinocyte proliferation; of changes in the normal fibroblast and keratinocyte differentiation; of polymorphic actinodermatosis, vitiligo; of wound healing disorders; disturbances to the normal collagen, hyaluronic acid, elastin and glycosaminoglycan homeostasis; of increased activation of proteolytic enzymes in the skin, such as e.g. metalloproteinases.
[0105] In another embodiment, the present invention provides a method of treating hair loss, generating a hair follicle, in creasing the size of a hair follicle, increasing hair follicle formation, treating, inhibiting or suppressing a degenerative skin disorder, treating androgenetic alopecia (AGA), comprising any combination of the following steps: (a) disrupting the epidermis in the region of said hair loss in said subject; (b) administering a fibroblast growth factor-9 polypeptide; (c) administering a wnt polypeptide; and (d) administering a compound or factor that upregulates Sonic Hedgehog (SHH), Patched-1 (Ptch1), Patched-2 (Ptch2), Gli1, Gli2, or a combination thereof to said subject.
[0106] In another embodiment, the present invention provides a method of depilation comprising the step of administering a composition comprising a neutralizing fibroblast growth factor-9 antibody to a subject. In one embodiment, the antibody is administered at a concentration of 10 .mu.g/mL. In one embodiment, the depilation is in the legs, arms, underarms, pubic area, back, face, nose, or ears of said subject. In one embodiment, the method further comprises the step of disrupting the epidermis in the region of said depilation prior to said administering step. In one embodiment, the step of contacting is performed 3-12 days after said step of disrupting. In one embodiment, the step of disrupting is performed by exposing the region of said hair loss to a mechanical, chemical, or optical stimulus. In one embodiment, the optical stimulus is radiation. In one embodiment, the administering step is via topical administration. In another embodiment, the administering step is via subcutaneous administration.
[0107] In another embodiment, the present invention provides a method of reversing alopecia comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide to a bald or balding subject. In another embodiment, the present invention provides a method of arresting alopecia comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide to a bald or balding subject.
[0108] In another embodiment, the present invention provides a method of treating a wound in a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide to a bald or balding subject. In another embodiment, the present invention provides a method of treating an injury in a subject comprising the step of administering a composition comprising a fibroblast growth factor-9 polypeptide to a bald or balding subject.
FGF9
[0109] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a fibroblast growth factor-9 polypeptide, alone or in composition with one or more additional compounds. In one embodiment, FGF9 refers to Fgf-9, FGF-9, Fibroblast growth factor 9, GAF, glia activating factor, Glia-activating factor precursor, or HBGF-9. In one embodiment, the FGF9 protein of the methods of the present invention has the sequence:
IFPNGTIQGTRKDHSRFGILEFISIAVGLVSIRGVDSGLYLGMNEKGELYGSEKLTQEC VFREQFEENWYNTYSSNLYKHVDTGRRYYVALNKDGTPREGTRTKRHQKFTHFLPR PVDPDKVPELYKDILSQS (GenBank Accession No: BAA03572; SEQ ID No: 1). In another embodiment, the FGF9 protein has a sequence as set forth in GenBank Accession No. P31371, BAF83481, NP_002001, CAC17692, EAX08316, AAI03980, AAI03979, AAT74624 or AAH69692. In another embodiment, the FGF9 protein is encoded by a genomic nucleic acid molecule having a sequence as set forth in GenBank Accession No. AL139378.15, AY682094.1, or CH471075.1 or encoded by an mRNA molecule having a sequence as set forth in GenBank Accession No. AK290792.1, BC069692.1, BC103978.1, BC103979.1, CR746503.1, or D14838.1. In another embodiment, a biologically active fragment of an FGF9 protein is utilized in a method of the present invention. In another embodiment, a homolog of an FGF9 protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0110] In one embodiment, administration of recombinant human FGF9 increased levels of sonic hedgehog (SHH) gene expression (FIG. 14).
Shh
[0111] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a sonic hedgehog (SHH) polypeptide, alone or in composition with one or more additional compounds. In another embodiment, the methods of the present invention comprise the step of administering a compound or factor that increases SHH expression. In one embodiment, SHH refers to TPT; HHG1; HLP3; HPE3; SMMCI; TPTPS; or MCOPCB5. In one embodiment, the SHH protein of the methods of the present invention has the sequence:
[0112] MLLLARCLLLVLVSSLLVCSGLACGPGRGFGKRRHPKKLTPLAYKQFIPNVA EKTLGAS GRYEGKISRNSERFKELTPNYNPDIIFKDEENTGADRLMTQRCKDKLNALAI SVMNQWPGVKLRVTEGWDEDGHHSEESLHYEGRAVDITTSDRDRSKYGMLARLAVE AGFDWVYYESKAHIHCSVKAENSVAAKSGGCFPGSATVHLEQGGTKLVKDLSPGDRV LAADDQGRLLYSDFLTFLDRDDGAKKVFYVIETREPRERLLLTAAHLLFVAPHNDSAT GEPEASSGSGPPSGGALGPRALFASRVRPGQRVYVVAERDGDRRLLPAAVHSVTLSEE AAGAYAPLTAQGTILINRVLASCYAVIEEHSWAHRAFAPFRLAHALLAALAPARTDRG GDSGGGDRGGGGGRVALTAPGAADAPGAGATAGIHWYSQLLYQIGTWLLDSEALHPL GMAVKSS (GenBank Accession No: Q15465.1; SEQ ID No: 2). In another embodiment, the SHH protein has a sequence as set forth in GenBank Accession No. BAA34689.1; AAB67604.1; AAS01990.1; AAQ87879.1; EAL23913.1; EAX04543.1; AAA62179.1; or AAI11926.1. In another embodiment, the SHH protein is encoded by a nucleic acid having a sequence as set forth in GenBank Accession No. AB020410.1; AC002484.1; AC078834.5; AY422195.1; CH236954.1; CH471149.1; AY927450.1; L38518.1; or BC111925.1. In another embodiment, a biologically active fragment of a SHH protein is utilized in a method of the present invention. In another embodiment, a homolog of a SHH protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0113] In one embodiment, SHH binds to the patched (PTC) receptor, which functions in association with smoothened (SMO), to activate the transcription of target genes. In the absence of SHH, PTC represses the constitutive signaling activity of SMO. In another embodiment, SHH also regulates the gli oncogene. In another embodiment, SHH is an intercellular signal essential for a variety of patterning events during development: signal produced by the notochord that induces ventral cell fate in the neural tube and somites, and the polarizing signal for patterning of the anterior-posterior axis of the developing limb bud. In another embodiment, SHH displays both floor plate- and motor neuron-inducing activity.
[0114] In another embodiment, administration of recombinant human FGF9 increased levels of Patched homolog 1 (Drosophila), (PTCH1; FIG. 14), which in one embodiment, is a human gene. In one embodiment, Ptch1 encodes a member of the patched gene family. In one embodiment, Ptch1 is the receptor for sonic hedgehog (SHH), which in one embodiment, is a secreted molecule implicated in the formation of embryonic structures and in tumorigenesis. In one embodiment, Ptch1 functions as a tumor suppressor. In one embodiment, mutations of Ptch1 have been associated with nevoid basal cell carcinoma syndrome, esophageal squamous cell carcinoma, trichoepitheliomas, transitional cell carcinomas of the bladder, as well as holoprosencephaly. In one embodiment, alternative splicing of Ptch1 results in multiple transcript variants encoding different isoforms.
PTCH1
[0115] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a Patched-1 (PTCH1) polypeptide, alone or in composition with one or more additional compounds. In another embodiment, the methods of the present invention comprise the step of administering a compound or factor that increases PTCH1 expression. In one embodiment, PTCHlrefers to PTC; BCNS; HPE7; PTC1; PTCH; NBCCS; PTCH11; FLJ26746; or FLJ42602. In one embodiment, the PTCH1 protein of the methods of the present invention has the sequence:
[0116] MASAGNAAEPQDRGGGGSGCIGAPGRPAGGGRRRRTGGLRRAAAPDRDYL HRPSYCDAAFALEQISKGKATGRKAPLWLRAKFQRLLFKLGCYIQKNCGKFLVVGLLI FGAFAVGLKAANLETNVEELWVEVGGRVSRELNYTRQKIGEEAMFNPQLMIQTPKEE GANVLTTEALLQHLDSALQASRVHVYMYNRQWKLEHLCYKSGELITETGYMDQIIEY LYPCLIITPLDCFWEGAKLQSGTAYLLGKPPLRWTNFDPLEFLEELKKINYQVDSWEEM LNKAEVGHGYMDRPCLNPADPDCPATAPNKNSTKPLDMALVLNGGCHGLSRKYMH WQEELIVGGTVKNSTGKLVSAHALQTMFQLMTPKQMYEHFKGYEYVSHINWNEDKA AAILEAWQRTYVEVVHQSVAQNSTQKVLSFTTTTLDDILKSFSDVSVIRVASGYLLML AYACLTMLRWDCSKSQGAVGLAGVLLVALSVAAGLGLCSLIGISFNAATTQVLPFLAL GVGVDDVFLLAHAFSETGQNKRIPFEDRTGECLKRTGASVALTSISNVTAFFMAALIPIP ALRAFSLQAAVVVVFNFAMVLLIFPAILSMDLYRREDRRLDIFCCFTSPCVSRVIQVEPQ AYTDTHDNTRYSPPPPYSSHSFAHETQITMQSTVQLRTEYDPHTHVYYTTAEPRSEISV QPVTVTQDTLSCQSPESTSSTRDLLSQFSDSSLHCLEPPCTKWTLSSFAEKHYAPFLLKP KAKVVVIFLFLGLLGVSLYGTTRVRDGLDLTDIVPRETREYDFIAAQFKYFSFYNMYIV TQKADYPNIQHLLYDLHRSFSNVKYVMLEENKQLPKMWLHYFRDWLQGLQDAFDSD WETGKIMPNNYKNGSDDGVLAYKLLVQTGSRDKPIDISQLTKQRLVDADGIINPSAFYI YLTAWVSNDPVAYAASQANIRPHRPEWVHDKADYMPETRLRIPAAEPIEYAQFPFYLN GLRDTSDFVEAIEKVRTICSNYTSLGLSSYPNGYPFLFWEQYIGLRHWLLLFISVVLACT FLVCAVFLLNPWTAGIIVMVLALMTVELFGMMGLIGIKLSAVPVVILIASVGIGVEFTV HVALAFLTAIGDKNRRAVLALEHMFAPVLDGAVSTLLGVLMLAGSEFDFIVRYFFAVL AILTILGVLNGLVLLPVLLSFFGPYPEVSPANGLNRLPTPSPEPPPSVVRFAMPPGHTHSG SDSSDSEYSSQTTVSGLSEELRHYEAQQGAGGPAHQVIVEATENPVFAHSTVVHPESRH HPPSNPRQQPHLDSGSLPPGRQGQQPRRDPPREGLWPPPYRPRRDAFEISTEGHSGPSNR ARWGPRGARSHNPRNPASTAMGSSVPGYCQPITTVTASASVTVAVHPPPVPGPGRNPR GGLCPGYPETDHGLFEDPHVPFHVRCERRDSKVEVIELQDVECEERPRGSSSN (GenBank Accession No: Q13635.2; SEQ ID No: 3). In another embodiment, the PTCH1 protein has a sequence as set forth in GenBank Accession No. CAH73817.1; CAH73818.1; CAH73819.1; AAR21238.1; AAR21239.1; AAR21240.1; EAW92631.1; EAW92632.1; BAD74184.1; BAD74185.1; BAD74186.1; BAD74187.1; BAD74188.1; BAD92732.1; BAF47711.1; BAE45300.1; BAE45302.1; BAE45304.1; BAF47712.1; BAC85893.1; AAH43542.1; AAC50496.1; AAC50550.1; or AAI52920.1. In another embodiment, the PTCH1 protein is encoded by a nucleic acid having a sequence as set forth in GenBank Accession No. AL161729.27; AY395758.1; AY395768.1; AY395772.1; CH471174.1; AB189436.1; AB189437.1; AB189438.1; AB189439.1; AB189440.1; AB209495.1; AB212827.1; AB212828.1; AB214500.1; AB233422.1; AB233424.1; AB239329.1; AI358880.1; AI494442.1; AK124593.1; AK130256.1; BC043542.1; BF195352.1; BM974119.1; BX117041.1; CR744004.1; DB093644.1; U43148.1; U59464.1; or BC152919.1. In another embodiment, a biologically active fragment of an PTCH1 protein is utilized in a method of the present invention. In another embodiment, a homolog of an PTCH1 protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0117] In another embodiment, administration of recombinant human FGF9 increased levels of Patched homolog 2 (Drosophila), (PTCH; FIG. 14)
PTCH2
[0118] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a Patched-2 (PTCH2) polypeptide, alone or in composition with one or more additional compounds. In another embodiment, the methods of the present invention comprise the step of administering a compound or factor that increases PTCH2 expression. In one embodiment, ptch2 encodes a member of the patched gene family. In one embodiment, the patched protein is the receptor for sonic hedgehog, a secreted molecule implicated in the formation of embryonic structures and in tumorigenesis. In one embodiment, ptch2 is mutated in a medulloblastoma and in a basal cell carcinoma, suggesting that it plays a role in the development of some tumors. Alternative transcript variants have been described, but their biological function has not been determined. In one embodiment, the PTCH2 polypeptide for use in the methods of the present invention has the sequence:
[0119] FDFIVRYFFAALTVLTLLGLLHGLVLLPVLLSILGPPPEVIQMYKESPEILSPPA PQGGGLRVGSLQVNISYWKELLWCQDLRPEEI (GenBank Accession No: Q5JR97; SEQ ID No: 4). In another embodiment, the PTCH2 protein has a sequence as set forth in GenBank Accession No. CAI23127.1; CAI13000.1; AAR05447.1; EAX07017.1; AAD25953.1; AAC79847.1; AAD17260.1; AAQ88919.1; AAQ89375.1; or AAI52912.1. In another embodiment, the PTCH2 protein is encoded by a nucleic acid having a sequence as set forth in GenBank Accession No. AL136380.22; AL592166.16; AY438664.1; CH471059.2; AF087651.1; AF091501.1; AF119569.1; AK307168.1; AY358555.1; AY359016.1; or BC152911.1. In another embodiment, a biologically active fragment of a PTCH2 protein is utilized in a method of the present invention. In another embodiment, a homolog of a PTCH2 protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
GLI1
[0120] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a glioma-associated oncogene homolog 1 (zinc finger protein) (GLI1) polypeptide, alone or in a composition with one or more additional compounds. In one embodiment, gli1 encodes a protein which is a member of the Kruppel family of zinc finger proteins. In another embodiment, the methods of the present invention comprise the step of administering a compound or factor that increases GLI1 expression. In one embodiment, the GLI1 polypeptide for use in the methods of the present invention has the sequence:
[0121] MFNSMTPPPISSYGEPCCLRPLPSQGAPSVGTEGLSGPPFCHQANLMSGPHSY GPARETNSCTEGPLFSSPRSAVKLTKKRALSISPLSDASLDLQTVIRTSPSSLVAFINSRCT SPGGSYGHLSIGTMSPSLGFPAQMNHQKGPSPSFGVQPCGPHDSARGGMIPHPQSRGPF PTCQLKSELDMLVGKCREEPLEGDMSSPNSTGIQDPLLGMLDGREDLEREEKREPESVY ETDCRWDGCSQEFDSQEQLVHHINSEHIHGERKEFVCHWGGCSRELRPFKAQYMLVV HMRRHTGEKPHKCTFEGCRKSYSRLENLKTHLRSHTGEKPYMCEHEGCSKAFSNASD RAKHQNRTHSNEKPYVCKLPGCTKRYTDPSSLRKHVKTVHGPDAHVTKRHRGDGPLP RAPSISTVEPKREREGGPIREESRLTVPEGAMKPQPSPGAQSSCSSDHSPAGSAANTDSG VEMTGNAGGSTEDLSSLDEGPCIAGTGLSTLRRLENLRLDQLHQLRPIGTRGLKLPSLS HTGTTVSRRVGPPVSLERRSSSSSSISSAYTVSRRSSLASPFPPGSPPENGASSLPGLMPA QHYLLRARYASARGGGTSPTAASSLDRIGGLPMPPWRSRAEYPGYNPNAGVTRRASDP AQAADRPAPARVQRFKSLGCVHTPPTVAGGGQNFDPYLPTSVYSPQPPSITENAAMDA RGLQEEPEVGTSMVGSGLNPYMDFPPTDTLGYGGPEGAAAEPYGARGPGSLPLGPGPP TNYGPNPCPQQASYPDPTQETWGEFPSHSGLYPGPKALGGTYSQCPRLEHYGQVQVKP EQGCPVGSDSTGLAPCLNAHPSEGPPHPQPLFSHYPQPSPPQYLQSGPYTQPPPDYLPSE PRPCLDFDSPTHSTGQLKAQLVCNYVQSQQELLWEGGGREDAPAQEPSYQSPKFLGGS QVSPSRAKAPVNTYGPGFGPNLPNHKSGSYPTPSPCHENFVVGANRASHRAAAPPRLLP PLPTCYGPLKVGGTNPSCGHPEVGRLGGGPALYPPPEGQVCNPLDSLDLDNTQLDFVAI LDEPQGLSPPPSHDQRGSSGHTPPPSGPPNMAVGNMSVLLRSLPGETEFLNSSA (GenBank Accession No: P08151; SEQ ID No: 5). In another embodiment, the GLI1 protein has a sequence as set forth in GenBank Accession No. AAM13391.1; EAW97013.1; BAG60219.1; AAH13000.1; or CAA30297.1. In another embodiment, the GLI1 protein is encoded by a nucleic acid having a sequence as set forth in GenBank Accession No. AC022506.38; AF316573.1; CH471054.1; AK297899.1; BC013000.2; or X07384.1. In another embodiment, a biologically active fragment of a GLI1 protein is utilized in a method of the present invention. In another embodiment, a homolog of a GLI1 protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
GLI2
[0122] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a glioma-associated oncogene homolog 2 (zinc finger protein) (GLI2) polypeptide, alone or in a composition with one or more additional compounds. In another embodiment, the methods of the present invention comprise the step of administering a compound or factor that increases GLI2 expression. In one embodiment, GLI2 may be referred to as HPE9; THP1; or THP2. In one embodiment, gli2 encodes a protein which belongs to the C2H2-type zinc finger protein subclass of the Gli family. Members of this subclass are characterized as transcription factors which bind DNA through zinc finger motifs. These motifs contain conserved H--C links. Gli family zinc finger proteins are mediators of Sonic hedgehog (Shh) signaling and they are implicated as potent oncogenes in the embryonal carcinoma cell. The protein encoded by this gene localizes to the cytoplasm and activates patched Drosophila homolog (PTCH) gene expression. It is also thought to play a role during embryogenesis. The encoded protein is associated with several phenotypes-Greig cephalopolysyndactyly syndrome, Pallister-Hall syndrome, preaxial polydactyly type IV, postaxial polydactyly types A1 and B. In one embodiment, the GLI2 polypeptide for use in the methods of the present invention has the sequence: MALTSINATPTQLSSSSNCLSDTNQNKQSSESAVSSTVNPVAIHKRSKVKTEPEGLRPAS PLALTQGQVLDTAHVGVPFPSPQEQLADLKEDLDRDDCKQEAEVVIYETNCHWEDCT KEYDTQEQLVHHINNEHIHGEKKEFVCRWQACTREQKPFKAQYMLVVHMRRHTGEK PHKCTFEGCSKAYSRLENLKTHLRSHTGEKPYVCEHEGCNKAFSNASDRAKHQNRTHS NEKPYICKIPGCTKRYTDPSSLRKHVKTVHGPDAHVTKKQRNDVHLRTPLLKENGDSE AGTEPGGPESTEASSTSQAVEDCLHVRAIKTESSGLCQSSPGAQSSCSSEPSPLGSAPNN DSGVEMPGTGPGSLGDLTALDDTPPGADTSALAAPSAGGLQLRKHMTTMHRFEQLKK EKLKSLKDSCSWAGPTPHTRNTKLPPLPGSGSILENFSGSGGGGPAGLLPNPRLSELSAS EVTMLSQLQERRDSSTSTVSSAYTVSRRSSGISPYFSSRRSSEASPLGAGRPHNASSADS YDPISTDASRRSSEASQCSGGSGLLNLTPAQQYSLRAKYAAATGGPPPTPLPGLERMSL RTRLALLDAAEGTLPAGCPRPLGPRRGSDGPTYGHGHAGAAPAFPHEAPGGGTRRASD PVRRPDALSLPRVQRFHSTHNVNPGPLPPCADRRGLRLQSHPSTDGGLARGAYSPRPPSI SENVAMEAVAAGVDGAGPEADLGLPEDDLVLPDDVVQYIKAHASGALDEGTGQVYP TESTGFSDNPRLPSPGLHGQRRMVAADSNVGPSAPMLGGCQLGFGAPSSLNKNNMPV QWNEVSSGTVDSLASQVKPPPFPQGNLAVVQQKPAFGQYPGYSPQGLQASPGGLDSTQ PHLQPRSGAPSQGIPRVNYMQQLRQPVAGSQCPGMTTTMSPHACYGQVHPQLSPSTIS GALNQFPQSCSNMPAKPGHLGHPQQTEVAPDPTTMGNRHRELGVPNSALAGVPPPHP VQSYPQQSHHLAASMSQEGYHQVPSLLPARQPGFMEPQTGPMGVATAGFGLVQPRPP LEPSPTGRHRGVRAVQQQLAYARATGHAMAAMPSSQETAEAVPKGAMGNMGSVPPQ PPPQDAGGAPDHSMLYYYGQIHMYEQDGGLENLGSCQVMRSQPPQPQACQDSIQPQP LPSPGVNQVSSTVDSQLLEAPQIDFDAIMDDGDHSSLFSGALSPSLLHSLSQNSSRLTTP RNSLTLPSIPAGISNMAVGDMSSMLTSLAEESKFLNMMT (GenBank Accession No: P10070; SEQ ID No: 6). In another embodiment, the GLI2 protein has a sequence as set forth in GenBank Accession No. AAA35898.1; BAA25665.1; BAA25666.1; BAA25667.1; BAA25668.1; BAD92591.1; BAG61875.1; AAS72889.1; AAS72890.1; AAS72891.1; AAI1l411.1; BAA03568.1; BAA03569.1; AAY58315.1; AAY58316.1; AAY58317.1; or AAY87165.1. In another embodiment, the GLI2 protein is encoded by a nucleic acid having a sequence as set forth in GenBank Accession No. AC016764.8; AC017033.5 (60664 . . . 181887); M20672.1; M20673.1; AB007295.1; AB007296.1; AB007297.1; AB007298.1; AB209354.1; AJ707583.1; AK300071.1; AY493737.1; AY493738.1; AY493739.1; BC111410.1; D14827.1; D14828.1; DQ004396.1; DQ004397.1; DQ004398.1; or DQ086814.1. In another embodiment, a biologically active fragment of a GLI2 protein is utilized in a method of the present invention. In another embodiment, a homolog of a GLI2 protein is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
WNT
[0123] In one embodiment, the methods of the present invention comprise the step of administering a composition comprising a Wnt polypeptide. The Wnt polypeptide of methods and compositions of the present invention has, in another embodiment, the sequence: MNRKARRCLGHLFLSLGMVYLRIGGFSSVVALGASIICNKIPGLAPRQRAICQSRPDAI IVIGEGSQMGLDECQFQFRNGRWNCSALGERTVFGKELKVGSREAAFTYAIIAAGVA HAITAACTQGNLSDCGCDKEKQGQYHRDEGWKWGGCSADIRYGIGFAKVFVDAREI KQNARTLMNLHNNEAGRKILEENMKLECKCHGVSGSCTTKTCWTTLPQFRELGYVL KDKYNEAVHVEPVRASRNKRPTFLKIKKPLSYRKPMDTDLVYIEKSPNYCEEDPVTG SVGTQGRACNKTAPQASGCDLMCCGRGYNTHQYARVWQCNCKFHWCCYVKCNTC SERTEMYTCK (GenBank Accession No: BC008811; SEQ ID No: 7). In another embodiment, the Wnt polypeptide has a sequence selected from the sequences set forth in GenBank entries NM_004625, D83175, U53476, and NP_004616. In another embodiment, the Wnt polypeptide is a Wnt7 protein. In another embodiment, the Wnt polypeptide is a Wnt7a polypeptide. In another embodiment, the Wnt polypeptide is Wnt1 protein. In another embodiment, the Wnt polypeptide is a Wnt3 polypeptide. In another embodiment, the Wnt polypeptide is a Wnt3a polypeptide. In another embodiment, the Wnt polypeptide is a Wnt10 polypeptide. In another embodiment, the Wnt polypeptide is a Wnt10a protein. In another embodiment, the Wnt polypeptide is a Wnt10b polypeptide. In another embodiment, the Wnt polypeptide is encoded by a nucleic acid molecule having a sequence set forth in the one of the above GenBank entries. In another embodiment, a biologically active fragment of a Wnt polypeptide is utilized in a method of the present invention. In another embodiment, a biologically active fragment of a Wnt7 protein is utilized in a method of the present invention. In another embodiment, a biologically active fragment of a Wnt polypeptide is utilized in a method of the present invention. In another embodiment, a biologically active fragment of a Wnt7a polypeptide is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0124] In another embodiment, methods of the present invention stimulate one or more members of the SHH signaling pathway, which in one embodiment is N-Shh (cleavage product), N-Shh-Chol, which in one embodiment, inhibits Patched-1 and Patched-2, which in one embodiment, inhibit Smoothened, which in one embodiment, stimulates GLI-1, which in one embodiment, stimulates transcription of other genes (in one embodiment, GLI-1, PTC1, HNF3 3) and GLI-2, and GLI-3, which in one embodiment inhibit transcription of other genes. Thus, in one embodiment, FGF9 stimulation and the resulting increase in SHH will relieve the tonic inhibition of Patched proteins on the Smoothened protein and increase levels of GLI-1, leading to enhancement of gene transcription.
[0125] In another embodiment, methods of the present invention stimulate one or more members of the WNT signaling pathway, which in one embodiment is Frizzled, SFRP, Dishevelled (Dsh), TCF, LRP, APC, 0-catenin, Axin, Dickkopf, GSK3, Naked, Porcupine, or FRAT/GBP.
[0126] In another embodiment, the wnt pathway is stimulated before the hedgehog pathway. In another embodiment, the two pathways are stimulated in an overlapping fashion. In another embodiment, the two pathways are stimulated simultaneously. Each possibility represents a separate embodiment of the present invention.
[0127] In another embodiment, homologues and variants of transcripts and proteins of the present invention are administered in methods of the present invention. In another embodiment, homologues and variants of transcripts and proteins of the present invention are targeted in methods of the present invention. Each possibility represents a separate embodiment of the present invention.
[0128] The terms "homology," "homologous," etc, when in reference to any protein or peptide, refer in one embodiment, to a percentage of amino acid residues in the candidate sequence that are identical with the residues of a corresponding native polypeptide, after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent homology, and not considering any conservative substitutions as part of the sequence identity. Methods and computer programs for the alignment are well known in the art.
[0129] In another embodiment, the term "homology," when in reference to any nucleic acid sequence similarly indicates a percentage of nucleotides in a candidate sequence that are identical with the nucleotides of a corresponding native nucleic acid sequence.
[0130] In another embodiment, "homology" refers to identity of greater than 70%. In another embodiment, "homology" refers to identity of greater than 75%. In another embodiment, "homology" refers to identity of greater than 80%. In another embodiment, "homology" refers to identity of greater than 82%. In another embodiment, "homology" refers to identity of greater than 85%. In another embodiment, "homology" refers to identity of greater than 87%. In another embodiment, "homology" refers to identity of greater than 90%. In another embodiment, "homology" refers to identity of greater than 92%. In another embodiment, "homology" refers to identity of greater than 95%. In another embodiment, "homology" refers to identity of greater than 97%. In another embodiment, "homology" refers to identity of greater than 98%. In another embodiment, "homology" refers to identity of greater than 99%. In another embodiment, "homology" refers to identity of 100%.
[0131] Protein and/or peptide homology for any amino acid sequence listed herein is determined, in one embodiment, by methods well described in the art, including immunoblot analysis, or via computer algorithm analysis of amino acid sequences, utilizing any of a number of software packages available, via established methods. Some of these packages may include the FASTA, BLAST, MPsrch or Scanps packages, and may employ the use of the Smith and Waterman algorithms, and/or global/local or BLOCKS alignments for analysis, for example. Each method of determining homology represents a separate embodiment of the present invention.
[0132] In one embodiment, the term "peptide" includes native peptides (either degradation products, synthetically synthesized peptides or recombinant peptides) and peptidomimetics (typically, synthetically synthesized peptides), such as peptoids and semipeptoids which are peptide analogs, which may have, for example, modifications rendering the peptides more stable while in a body or more capable of penetrating into bacterial cells. Such modifications include, but are not limited to N terminus modification, C terminus modification, peptide bond modification, including, but not limited to, CH2-NH, CH2-S, CH2-S.dbd.O, O.dbd.C--NH, CH2-O, CH2-CH2, S.dbd.C--NH, CH.dbd.CH or CF.dbd.CH, backbone modifications, and residue modification. Methods for preparing peptidomimetic compounds are well known in the art and are specified, for example, in Quantitative Drug Design, C. A. Ramsden Gd., Chapter 17.2, F. Choplin Pergamon Press (1992), which is incorporated by reference as if fully set forth herein.
[0133] Peptide bonds (--CO--NH--) within the peptide may be substituted, for example, by N-methylated bonds (--N(CH3)-CO--), ester bonds (--C(R)H--C--O--O--C(R)--N--), ketomethylen bonds (--CO--CH2-), ca-aza bonds (--NH--N(R)--CO--), wherein R is any alkyl, e.g., methyl, carba bonds (--CH2-NH--), hydroxyethylene bonds (--CH(OH)--CH2-), thioamide bonds (--CS--NH--), olefinic double bonds (--CH.dbd.CH--), retro amide bonds (--NH--CO--), peptide derivatives (--N(R)--CH2-CO--), wherein R is the "normal" side chain, naturally presented on the carbon atom.
[0134] These modifications can occur at any of the bonds along the peptide chain and even at several (2-3) at the same time.
[0135] Natural aromatic amino acids, Trp, Tyr and Phe, may be substituted for synthetic non-natural acid such as TIC, naphthylelanine (Nol), ring-methylated derivatives of Phe, halogenated derivatives of Phe or o-methyl-Tyr.
[0136] In addition to the above, the peptides of the present invention may also include one or more modified amino acids or one or more non-amino acid monomers (e.g. fatty acids, complex carbohydrates etc).
[0137] As used herein in the specification and in the claims section below the term "amino acid" or "amino acids" is understood to include the 20 naturally occurring amino acids; those amino acids often modified post-translationally in vivo, including, for example, hydroxyproline, phosphoserine and phosphothreonine; and other unusual amino acids including, but not limited to, 2-aminoadipic acid, hydroxylysine, isodesmosine, nor-valine, nor-leucine and ornithine. Furthermore, the term "amino acid" includes both D- and L-amino acids.
[0138] In another embodiment, naturally occurring amino acids and non-conventional or modified amino acids as are known in the art can be used with the present invention.
[0139] In another embodiment, the present invention provides a kit, comprising a tools and/or a compound suitable for performing a method of the present invention.
[0140] In another embodiment, the present invention provides a device, comprising a tool suitable for epidermal disruption and a means of delivering a compound or factor that upregulates expression of SHH.
[0141] It is to be understood that included in the present invention are methods comprising the step of administering an isolated nucleic acid, in one embodiment, a vector or plasmid, encoding a polypeptide of the present invention, which in one embodiment, is a fibroblast growth factor-9 polypeptide, shh, wnt, ptch1, ptch, gli1, or gli2, or a composition comprising such a vector.
[0142] In one embodiment, an isolated nucleic acid that encodes a polypeptide of the present invention for use in the methods of the present invention is provided.
[0143] In one embodiment, an "isolated nucleic acid" refers to a nucleic acid segment or fragment which has been separated from sequences which flank it in a naturally occurring state, e.g., a DNA fragment which has been removed from the sequences which are normally adjacent to the fragment, e.g., the sequences adjacent to the fragment in a genome in which it naturally occurs. The term also applies to nucleic acids which have been substantially purified from other components which naturally accompany the nucleic acid, e.g., RNA or DNA or proteins, which naturally accompany it in the cell. The term therefore includes, for example, a recombinant DNA which is incorporated into a vector, into an autonomously replicating plasmid or virus, or into the genomic DNA of a prokaryote or eukaryote, or which exists as a separate molecule (e.g., as a cDNA or a genomic or cDNA fragment produced by PCR or restriction enzyme digestion) independent of other sequences. It also includes a recombinant DNA which is part of a hybrid gene encoding additional polypeptide sequence.
[0144] In one embodiment, the present invention provides a cell comprising an isolated nucleic acid or vector of the present invention.
[0145] In one embodiment, two polynucleotides of the present invention are operably linked. For example, in one embodiment, polynucleotides encoding FGF9 and WNT may be operably linked. In one embodiment, "operably linked" indicates that a single-stranded or double-stranded nucleic acid moiety comprises the two polynucleotides arranged within the nucleic acid moiety in such a manner that they are expressed together. By way of example, a promoter operably linked to the coding region of a gene is able to promote transcription of the coding region.
[0146] In one embodiment, a polynucleotide of the present invention comprises a promoter/regulatory sequence, which in one embodiment, the promoter/regulatory is positioned at the 5' end of the desired protein coding sequence such that it drives expression of the desired protein in a cell. Together, the nucleic acid encoding the desired protein and its promoter/regulatory sequence comprise a "transgene."
[0147] In one embodiment, the term "promoter/regulatory sequence" refers to a nucleic acid sequence which is required for expression of a gene product operably linked to the promoter/regulatory sequence. In some instances, this sequence may be the core promoter sequence and in other instances, this sequence may also include an enhancer sequence and other regulatory elements which are required for expression of the gene product. The promoter/regulatory sequence may, for example, be one which expresses the gene product in a tissue specific manner. In one embodiment, a promoter used in the present invention may be constitutive or inducible. In another embodiment, a promoter for use in the methods of the present invention may be tissue-specific. Such promoters are well known in the art.
[0148] In another embodiment, the present invention provides a delivery vehicle for administration of a polypeptide of the present invention. Examples of such delivery vehicles are known in the art and may include recombinant viruses or bacteria engineered to express said polypeptide. In one embodiment, said viruses or bacteria are attenuated. In one embodiment, viruses for use in the methods of the present invention may include retroviruses, adenoviruses, adeno-associated viruses, etc. In one embodiment, the virus may be of any known serotype or subgroup.
[0149] In one embodiment, any one of a number of different vectors can be used in the methods of the present invention, such as viral vectors, plasmid vectors, linear DNA, etc., as known in the art, to introduce an exogenous nucleic acid fragment encoding a therapeutic agent into target cells and/or tissue. These vectors can be inserted, for example, using infection, transduction, transfection, calcium-phosphate mediated transfection, DEAE-dextran mediated transfection, electroporation, liposome-mediated transfection, biolistic gene delivery, liposomal gene delivery using fusogenic and anionic liposomes (which are an alternative to the use of cationic liposomes), direct injection, receptor-mediated uptake, magnetoporation, ultrasound, or any combination thereof, as well as other techniques known in the art (for further detail see, for example, "Methods in Enzymology" Vol. 1-317, Academic Press, Current Protocols in Molecular Biology, Ausubel F. M. et al. (eds.) Greene Publishing Associates, (1989) and in Molecular Cloning: A Laboratory Manual, 2nd Edition, Sambrook et al. Cold Spring Harbor Laboratory Press, (1989), or other standard laboratory manuals). The polynucleotide segments encoding sequences of interest can be ligated into an expression vector system suitable for transducing mammalian cells and for directing the expression of recombinant products within the transduced cells. The introduction of the exogenous nucleic acid fragment is accomplished by introducing the vector into the vicinity of the micro-organ. Once the exogenous nucleic acid fragment has been incorporated into the cells using any of the techniques described above or known in the art, the production and/or the secretion rate of the therapeutic agent encoded by the nucleic acid fragment can be quantified. In one embodiment, the term "exogenous" refers to a substance that originated outside, for example a nucleic acid that originated outside of a cell or tissue.
[0150] In one embodiment, a vector for use in the methods of the present invention is a non-immunogenic gene transfer agent such as a nonviral vector (e.g. DNA plasmids or minicircle DNA), a "gutless" viral vector i.e. without endogenous genes (which in one embodiment, is due to a deletion, while in another embodiment, due to an insertion, substitution or deletion in a gene that prevents gene expression), a helper-dependent adenovirus (HDAd) vector, or adeno associated virus AAV (which in one embodiment is single stranded and in another embodiment, double stranded). In another embodiment, said formulation is so chosen such that recombinant gene expression results in lack of toxicity or immune-mediated rejection of the gene product by the tissue. In one embodiment, the vector is virally derived, and in another embodiment, the vector is a plasmid. In one embodiment, the virally-derived vector is derived from adenovirus, which in one embodiment, is helper-dependent adenovirus, while in another embodiment, the virally-derived vector is derived from adenovirus-associated vector.
[0151] In one embodiment, the term "vector" or "expression vector" refers to a carrier molecule into which a nucleic acid sequence can be inserted for introduction into a cell where it can be replicated. In one embodiment, the nucleic acid molecules are transcribed into RNA, which in some cases are then translated into a protein, polypeptide, or peptide. In other cases, RNA sequences are not translated, for example, in the production of antisense molecules or ribozymes. In one embodiment, expression vectors can contain a variety of "control sequences" which refer to nucleic acid sequences necessary for the transcription and possibly translation of an operably linked coding sequence in a particular host cell. In another embodiment, a vector further includes an origin of replication. In one embodiment the vector may be a shuttle vector, which in one embodiment can propagate both in prokaryotic and eukaryotic cells, or in another embodiment, the vector may be constructed to facilitate its integration within the genome of an organism of choice. The vector, in other embodiments may be, for example, a plasmid, a bacmid, a phagemid, a cosmid, a phage, a virus or an artificial chromosome. In one embodiment, the vector is a viral vector, which in one embodiment may be a bacteriophage, mammalian virus, or plant virus.
[0152] In other embodiments, the viral vector is derived from a virus such as vaccinia virus, lentivirus, polio virus, hepatitis virus, papilloma virus, cytomegalovirus, simian virus, or herpes simplex virus.
[0153] In certain embodiments of the invention, the vector comprising a nucleic acid sequence may comprise naked recombinant DNA or plasmids. Transfer of the construct may be performed by any method which physically or chemically permeabilizes the cell membrane. In one embodiment, the vector is a mini-circle DNA, which in one embodiment, is a supercoiled DNA molecule for non-viral gene transfer, which has neither a bacterial origin of replication nor an antibiotic resistance marker.
[0154] Construction of vectors using standard recombinant techniques is well known in the art (see, for example, Maniatis, et al., Molecular Cloning, A Laboratory Manual (Cold Spring Harbor, 1990) and Ausubel, et al., 1994, Current Protocols in Molecular Biology (John Wiley & Sons, 1996), both incorporated herein by reference).
[0155] In one embodiment, compositions of the present invention comprise the indicated agent, while in another embodiment, compositions of the present invention consist essentially of the indicated agent, while in another embodiment, compositions of the present invention consist of the indicated agent. In some embodiments, the term "comprise" refers to the inclusion of the indicated active agent, such as human fibroblast growth factor-9 polypeptide, shh, wnt, etc, as well as inclusion of other active agents, and pharmaceutically acceptable carriers, excipients, emollients, stabilizers, etc., as are known in the pharmaceutical industry. In some embodiments, the term "consisting essentially of" refers to a composition, whose only active ingredient is the indicated active ingredient, however, other compounds may be included which are for stabilizing, preserving, etc. the formulation, but are not involved directly in the therapeutic effect of the indicated active ingredient. In some embodiments, the term "consisting essentially of" may refer to components which facilitate the release of the active ingredient. In some embodiments, the term "consisting" refers to a composition, which contains the active ingredient and a pharmaceutically acceptable carrier or excipient.
[0156] In one embodiment, methods of the present invention treat, inhibit or suppress hair loss. In one embodiment, methods of the present invention generate hair follicles or increase hair follicle size, in one embodiment, in a subject with hair loss. In one embodiment, hair loss is due to androgenetic alopecia (AGA). In another embodiment, hair loss is due to male pattern baldness. In another embodiment, hair loss is due to female pattern baldness. In another embodiment, hair loss is the result of a skin injury.
[0157] In another embodiment, the methods of the present invention treat, inhibit, or suppress a disease or disorder in a subject. In one embodiment, the subject has a disease or disorder comprising balding. In another embodiment, the subject does not have a disease or disorder comprising balding. In another embodiment, the disease or disorder is androgenetic alopecia (AGA). In another embodiment, the disease or disorder is male pattern baldness. In another embodiment, the disease or disorder is female pattern baldness. In another embodiment, the disease or disorder is a discoid lupus erythematosis. In another embodiment, the disease or disorder is a congenital hypotrichosis. In another embodiment, the disease or disorder is a lichen planopilaris.
[0158] In another embodiment, the disease or disorder is a scarring (or, in another embodiment, cicatricial) alopecia, which in one embodiment, is hair loss due to scarring of the scalp area. In one embodiment, scarring alopecia typically involves the top of the scalp and occurs predominantly in women. The condition frequently occurs in African-American women and is believed to be associated with persistent tight braiding or "corn-rowing" of scalp hair. A form of scarring alopecia also may occur in post-menopausal women, associated with inflammation of hair follicles and subsequent scarring. In another embodiment, the disease or disorder is any other disease or disorder comprising balding known in the art.
[0159] In another embodiment, the present invention provides methods for treating Alopecia areata, which in one embodiment, is an autoimmune disorder that causes patchy hair loss that can range from diffuse thinning to extensive areas of baldness with "islands" of retained hair.
[0160] In another embodiment, the present invention provides methods for treating Trichotillomania, which in one embodiment, compulsive hair pulling. Hair loss due to trichotillomania is typically patchy, as compulsive hair pullers tend to concentrate the pulling in selected areas.
[0161] In another embodiment, the present invention provides methods for treating Triangular alopecia, which in one embodiment, is a loss of hair in the temporal areas that sometimes begins in childhood. Hair loss may be complete, or a few fine, thin-diameter hairs may remain.
[0162] In another embodiment, the present invention provides methods for treating Telogen effluvium, which in one embodiment, is a common type of hair loss caused when a large percentage of scalp hairs are shifted into "shedding" phase. The causes of telogen effluvium may be hormonal, nutritional, drug-associated, or stress-associated.
[0163] In another embodiment, the present invention provides methods for treating Loose-anagen syndrome, which in one embodiment, is a condition occurring primarily in fair-haired persons in which scalp hair sits loosely in hair follicles and is easily extracted by combing or pulling. In one embodiment, the condition may appear in childhood.
[0164] In another embodiment, the present invention provides methods for treating Tinea Capitis (Scalp Ringworm), which in one embodiment, is caused by a fungal infection, and in one embodiment, is characterized by patches of scaling that can spread and result in broken hair, redness, swelling, and oozing on the scalp.
[0165] In another embodiment, the present invention provides methods for treating hair loss associated with particular conditions, which in one embodiment, is cancer, thyroid disease, inadequate protein in diet, low serum iron levels, or associated with particular environmental stimuli, which in one embodiment, is chemotherapy, or, in another embodiment, radiotherapy.
[0166] In other embodiments, the present invention provides a method of treating any disease, disorder, or symptom associated with balding. In other embodiments, the present invention provides a method of treating any disease, disorder, or symptom associated with degenerative skin disorder. In other embodiments, the present invention provides a method of treating any disease, disorder, or symptom associated with alopecia. Each disease, disorder, or symptom represents a separate embodiment of the present invention.
[0167] In one embodiment, "treating" refers to either therapeutic treatment or prophylactic or preventative measures, wherein the object is to prevent or lessen the targeted pathologic condition or disorder as described hereinabove. Thus, in one embodiment, treating may include directly affecting or curing, suppressing, inhibiting, preventing, reducing the severity of, delaying the onset of, reducing symptoms associated with the disease, disorder or condition, or a combination thereof. Thus, in one embodiment, "treating" refers inter alia to delaying progression, expediting remission, inducing remission, augmenting remission, speeding recovery, increasing efficacy of or decreasing resistance to alternative therapeutics, or a combination thereof. In one embodiment, "preventing" refers, inter alia, to delaying the onset of symptoms, preventing relapse, decreasing the number or frequency of relapse episodes, increasing latency between symptomatic episodes, or a combination thereof. In one embodiment, "suppressing" or "inhibiting", refers inter alia to reducing the severity of symptoms, reducing the severity of the current episode, reducing the number of symptoms, reducing the incidence of symptoms, reducing the latency of symptoms, ameliorating symptoms, reducing secondary symptoms, reducing secondary infections, or a combination thereof.
[0168] In one embodiment, symptoms are primary, while in another embodiment, symptoms are secondary. In one embodiment, "primary" refers to a symptom that is a direct result of the alopecia, while in one embodiment, "secondary" refers to a symptom that is derived from or consequent to a primary cause. In one embodiment, the compositions and strains for use in the present invention treat primary or secondary symptoms or secondary complications related to alopecia, in one embodiment, seborrheic dermatitis.
[0169] In another embodiment, "symptoms" may be any manifestation of alopecia, comprising hair loss, balding, temporary hair loss, patchy hair loss, degenerative skin disorders or a combination thereof.
[0170] Methods of determining the presence and severity of alopecia and/or degenerative skin disorders such as those described herein are well known in the art. Each method represents a separate embodiment of the present invention.
[0171] In one embodiment, the methods of the present invention are for treating a subject with hair loss. In one embodiment, the hair loss is in the scalp of the subject. In another embodiment, the hair loss is in the eyebrow of the subject. In another embodiment, the hair loss is in scarred skin tissue of the subject, which in one embodiment, may be scalp, eyebrow, arm, or leg of a subject. In another embodiment, any other hair-bearing area or region of the skin is treated by a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0172] In one embodiment, methods of the present invention comprise the step of disrupting the epidermis in the region of said hair loss prior to said administering step. In another embodiment, the epithelium is disrupted.
[0173] In another embodiment of methods and compositions of the present invention, the first step (e.g. epidermal disruption) is performed 3-12 days prior to the second step (e.g. addition of an active compound, factor, cell, etc). In another embodiment, the interval is 4-12 days. In another embodiment, the interval is 5-12 days. In another embodiment, the interval is 4-11 days. In another embodiment, the interval is 6-11 days. In another embodiment, the interval is 6-10 days. In another embodiment, the interval is 6-9 days. In another embodiment, the interval is 6-8 days. In another embodiment, the interval is 7-8 days. In another embodiment, the interval is 5-11 days. In another embodiment, the interval is 5-10 days. In another embodiment, the interval is 7-10 days. In another embodiment, the interval is about 1 week. In another embodiment, the compositions for use in the methods of the present invention are applied as the scabbing starts to heal, which in one embodiment is 3-12 days after epidermal disruption. In one embodiment, the compositions for use in the methods of the present invention are applied one day after scab detachment, in another embodiment, two days after scab detachment, in another embodiment, three days after scab detachment, in another embodiment, four days after scab detachment, in another embodiment, five days after scab detachment, in another embodiment, six days after scab detachment, in another embodiment, seven days or more after scab detachment. In another embodiment, the compositions for use in the present invention are administered on days 1-4 after scab detachment. Each possibility represents a separate embodiment of the present invention.
[0174] In one embodiment, the step of disrupting is performed by exposing the region of said hair loss to a mechanical, chemical, or optical stimulus. In one embodiment, the optical stimulus is radiation.
[0175] The step of disrupting the epidermis in methods of the present invention is performed, in another embodiment, by abrading the skin region of interest. In another embodiment, the term "abrading" refers to an act of creating an abrasion. In another embodiment, "abrading" refers to rubbing. In another embodiment, "abrading" refers to wearing away by friction. In one embodiment, epidermal abrasion causes, under the conditions utilized herein, de novo HF neo-genesis. In another embodiment, the epidermal layer is disrupted.
[0176] In one embodiment, "abrasion" refers to a wound consisting of superficial damage to the skin. In another embodiment, "abrasion" refers to an area of the scalp or skin from which the epidermis is removed. In another embodiment, "abrasion" refers to an area of the scalp or skin from which the epidermis and dermis are removed. Each definition of "abrading" and "abrasion" represents a separate embodiment of the present invention.
[0177] In one embodiment, epidermal disruption by a method of the present invention converts the skin region of interest back to an embryonic-like state, in which the follicle regenerates. In another embodiment, a subsequent window of opportunity is created, during which the number and size of new HF in the skin region of interest can be manipulated. In another embodiment, the administration of a compound or factor that promotes a differentiation of an uncommitted epidermal cell into a HF cell during this window causes regeneration of larger and more numerous HF. In one embodiment, the morphology of HF in abraded skin is similar to that of embryonic HF, and the markers expressed are similar as well.
[0178] In another embodiment, the excisional wounds of methods of the present invention are created using a surgical tool. In one embodiment, the surgical tool is a dermal biopsy punch. In another embodiment, the excisional wounds are induced by freezing or cryoinjury. The use of freezing or cryoinjury is well known in the art, and is used, for example by dermatologists to injure skin. In one embodiment, the freezing or cryoinjury results in a blister. In another embodiment, the blister is used as a "chamber" to introduce drugs and or cells into the reepithelialized area. Each possibility represents a separate embodiment of the present invention.
[0179] In another embodiment, the epidermal disruption in methods of the present invention further removes dermal tissue from the skin region of interest. In another embodiment, the epidermal disruption does not remove dermal tissue from the skin region of interest. Each possibility represents a separate embodiment of the present invention.
[0180] "Disrupting" an epidermis or epidermal layer refers, in another embodiment, to removing part of the epidermis or epidermal layer. In another embodiment, the term refers to disturbing the intactness of the epidermis or epidermal layer. In another embodiment, the term refers to perforating the epidermis or epidermal layer. In another embodiment, only part of the epidermal layer need be removed. In another embodiment, the entire epidermal layer is removed. In another embodiment, the term refers to abrading the epidermis or epidermal layer. In another embodiment, the term refers to wounding the epidermis or epidermal layer. Each possibility represents a separate embodiment of the present invention.
[0181] In another embodiment, the epidermal disruption is performed with a tool that comprises sandpaper. In another embodiment, the epidermal disruption is performed with a laser. In another embodiment, the laser is a Fraxel laser. In another embodiment, the laser is a CO.sub.2 laser. In another embodiment, the laser is an excimer laser. In another embodiment, the laser is any other type of laser capable of inducing trans-epithelial injury. In another embodiment, the epidermal disruption is performed with a felt wheel. In another embodiment, the epidermal disruption is performed with a surgical tool. In another embodiment, the epidermal disruption is performed with any other tool known in the art that is capable of epidermal disruption. In another embodiment, the epidermal disruption comprises use of a micro-dermabrasion device. In another embodiment, the epidermal disruption comprises a burn treatment.
[0182] In another embodiment, the epidermal disruption comprises a disruption of a follicle of said epidermis and a disruption of an interfollicular region of said epidermis. In another embodiment, the epidermal disruption comprises a disruption of a follicle of said epidermis and does not comprise a disruption of an interfollicular region of said epidermis. Each possibility represents a separate embodiment of the present invention.
[0183] In another embodiment, the epidermal disruption comprises a light-based method. In another embodiment, the epidermal disruption comprises irradiation with visible light. In another embodiment, the epidermal disruption comprises irradiation with infrared light. In another embodiment, the epidermal disruption comprises irradiation with ultraviolet radiation. In another embodiment, the epidermal disruption comprises orthovoltage irradiation. In another embodiment, the epidermal disruption comprises X-ray irradiation. In another embodiment, the epidermal disruption comprises any other type of irradiation known in the art.
[0184] In another embodiment, the epidermal disruption is performed by mechanical means. In another embodiment, "mechanical means" refers to abrading. In another embodiment, the term refers to wounding. In another embodiment, the term refers to ultrasound. In another embodiment, the term refers to radio-frequency. In another embodiment, the term refers to an electrical process or the use of an electrical current. In another embodiment, the term refers to electoporation. In another embodiment, the term refers to excision. In another embodiment, the term refers to tape-stripping. In another embodiment, the term refers to microdermabrasion. In another embodiment, the term refers to the use of peels. In another embodiment, the term refers to any other type of mechanical means known in the art. Each possibility represents a separate embodiment of the present invention.
[0185] In another embodiment, the epidermal disruption comprises chemical treatment. In another embodiment, the chemical is phenol. In another embodiment, the chemical is trichloracetic acid. In another embodiment, the chemical is ascorbic acid. In another embodiment, the chemical is any other chemical capable of epidermal disruption that is known in the art.
[0186] In another embodiment, epidermal trauma is utilized in a method of the present invention.
[0187] Each method or type of epidermal disruption, abrasion, and trauma represents a separate embodiment of the present invention.
[0188] In one embodiment, "WIHN" refers to HF neogenesis induced by disruption of the epithelial layer. In another embodiment, the term refers to HF neogenesis induced by abrasion. In another embodiment, the term refers to HF neogenesis induced by wounding. In another embodiment, the term refers to HF neogenesis induced by disruption of the epithelial layer, followed by administration of a compound or factor that promotes a differentiation of an uncommitted epidermal cell into a HF cell. Each possibility represents a separate embodiment of the present invention.
[0189] In another embodiment, the epidermal disruption of methods of the present invention creates an abrasion at least about 1-1.5 centimeters (cm) in width. In another embodiment, the abrasion is at least about 1 cm in width. In another embodiment, the abrasion is at least about 1.5 cm in width. In another embodiment, the abrasion is at least about 2 cm in width. Each type of abrasion represents a separate embodiment of the present invention.
[0190] In another embodiment, the excisional wounds of methods of the present invention are not surgically closed. In another embodiment, the excisional wounds are allowed to heal by secondary intention. In another embodiment, the skin region of interest is not contacted with a bandage or dressing following the epidermal disruption. In another embodiment, the skin region of interest is not contacted with an ointment following the epidermal disruption. In another embodiment, the skin region of interest is allowed to heal for a period of time without being contacted by any substance, device, ointment, etc., that is ordinarily administered to an abrasion or wound to facilitate healing. In another embodiment, the skin region of interest is allowed to heal for a period of time without being contacted by any substance, device, ointment, etc., that is ordinarily administered to an abrasion or wound to prevent infection. In another embodiment, the "period of time" is the time it takes the epidermal disruption to heal. In another embodiment, the period of time is any time or range of times between 2 days and 3 weeks. Each possibility represents a separate embodiment of the present invention.
[0191] In one embodiment, "following" refers to a period of time of about 2 days. In another embodiment, "following" refers to a period of time of about 3 days. In another embodiment, "following" refers to a period of time of about 4 days. In another embodiment, "following" refers to a period of time of about 5 days. In another embodiment, "following" refers to a period of time of about 7 days. In another embodiment, "following" refers to a period of time of about 10 days. In another embodiment, "following" refers to a period of time of about 2 weeks. In another embodiment, "following" refers to a period of time of about 3 weeks. Each possibility represents a separate embodiment of the present invention.
[0192] In another embodiment, a method of the present invention further comprises the step of depilating the skin in the region in which hair growth or follicle formation is desired. In one embodiment, said step of depilating is performed prior to said step of epidermal disruption.
[0193] In another embodiment, the depilation is epilation. In another embodiment, the depilation comprises the step of waxing. In another embodiment, the depilation comprises the step of plucking. In another embodiment, the depilation comprises the use of an abrasive material. In another embodiment, the depilation comprises the use of a laser. In another embodiment, the depilation comprises the use of electrolysis. In another embodiment, the depilation comprises the use of a mechanical device. In another embodiment, the depilation comprises the use of thioglycolic acid. In another embodiment, the depilation comprises the use of any other method of depilation or epilation known in the art. Each possibility represents a separate embodiment of the present invention.
[0194] In another embodiment, the additional step (depilation or administration of a retinoid) is performed prior to the step of disrupting the epidermis. In another embodiment, the additional step is performed following the step of disrupting the epidermis, but prior to the addition of the compound or factor that of the present invention. In another embodiment, the additional step is performed concurrently with the addition of the differentiation-promoting compound or factor. In another embodiment, the additional step is performed following the addition of the differentiation-promoting compound or factor. Each possibility represents a separate embodiment of the present invention.
[0195] In another embodiment, a method of the present invention further comprises the step of administering a topical retinoid to the skin region of interest. In one embodiment, the topical retinoid induces resting (telogen) HF in the skin region of interest to enter anagen. Each possibility represents a separate embodiment of the present invention.
[0196] In another embodiment, the additional step is performed between about two days and about three weeks before the step of abrading. In another embodiment, the additional step is performed about two days before the step of abrading. In another embodiment, the additional step is performed about three days before the step of abrading. In another embodiment, the additional step is performed about four days before the step of abrading. In another embodiment, the additional step is performed about one week before the step of abrading. In another embodiment, the additional step is performed about ten days before the step of abrading. In another embodiment, the additional step is performed about two weeks before the step of abrading. In another embodiment, the additional step is performed about three weeks before the step of abrading. Each possibility represents a separate embodiment of the present invention.
[0197] In one embodiment, the methods of the present invention further comprise the step of administering an antagonist of an androgen or an antagonist of an androgen receptor. In another embodiment, the methods of the present invention further comprise the step of administering a 5 alpha-reductase type 2 inhibitor.
[0198] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an anti-androgen compound. In one embodiment, the anti-androgen compound is finasteride. In another embodiment, the anti-androgen compound is Fluridil.RTM.. In another embodiment, the anti-androgen compound is dutasteride. In another embodiment, the anti-androgen compound is spironolactone. In another embodiment, the anti-androgen compound is cyproterone acetate. In another embodiment, the anti-androgen compound is bicalutamide. In another embodiment, the anti-androgen compound is flutamide. In another embodiment, the anti-androgen compound is nilutamide. In another embodiment, the anti-androgen compound is an inhibitor of an androgen receptor. In another embodiment, the anti-androgen compound is any other anti-androgen compound known in the art. Each possibility represents a separate embodiment of the present invention.
[0199] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an estrogen compound. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an estrogen receptor agonist. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an estrogen analogue. In one embodiment, the estrogen analogue is estradiol. In another embodiment, the estrogen analogue is 17 beta-estradiol. In another embodiment, the estrogen analogue is 17 alpha-estradiol. In another embodiment, the estrogen analogue is ZYC3. In another embodiment, the estrogen compound, estrogen receptor agonist, or estrogen analogue is any other estrogen compound, estrogen receptor agonist, or estrogen analogue known in the art. Each possibility represents a separate embodiment of the present invention.
[0200] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an inhibitor of an EGF protein. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an inhibitor of an EGFR. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with a compound that reduces an expression of an EGF protein or an EGFR. Each possibility represents a separate embodiment of the present invention.
[0201] In another embodiment, the inhibitor of an EGF or an EGF receptor is panitumumab. In another embodiment, the inhibitor is AG1478. In another embodiment, the inhibitor is nimotuzumab. In another embodiment, the inhibitor is an antibody that binds EGF or EGFR. In another embodiment, the inhibitor is HuMax-EGFR.RTM. (Genmab, Copenhagen, Denmark). In another embodiment, the inhibitor is cetuximab. In another embodiment, the inhibitor is IMC 11F8. In another embodiment, the inhibitor is matuzumab. In another embodiment, the inhibitor is SC 100. In another embodiment, the inhibitor is ALT 110. In another embodiment, the inhibitor is PX 1032. In another embodiment, the inhibitor is BMS 599626. In another embodiment, the inhibitor is MDX 214. In another embodiment, the inhibitor is PX 1041. In another embodiment, the inhibitor is any other inhibitor of an EGF or an EGF receptor known in the art. Each possibility represents a separate embodiment of the present invention.
[0202] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an inhibitor of a tyrosine kinase activity of an EGF receptor. In another embodiment, the inhibitor is gefitinib. In another embodiment, the inhibitor is erlotinib. In another embodiment, the inhibitor is canertinib. In another embodiment, the inhibitor is leflunomide. In another embodiment, the inhibitor is A77 1726. In another embodiment, the inhibitor is pelitinib. In another embodiment, the inhibitor is ZD 1839. In another embodiment, the inhibitor is CL 387785. In another embodiment, the inhibitor is EKI 785. In another embodiment, the inhibitor is vandetanib. In another embodiment, the inhibitor is any other inhibitor of a tyrosine kinase activity of an EGF receptor known in the art. Each possibility represents a separate embodiment of the present invention.
[0203] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an EGF or EGFR antagonist. In another embodiment, the EGF or EGFR antagonist is a carboxypeptidase inhibitor from potato (PCI) protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is a sprouty protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is an Argos protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is a lefty protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is an antibody that recognizes EGF or EGFR, or a fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is small molecule inhibitor that binds and reduces the activity of EGF or EGFR. In another embodiment, the EGF or EGFR antagonist is CRM197. In another embodiment, the EGF or EGFR antagonist is IMC-C225 (ImClone Systems, New York, N.Y.). In another embodiment, the EGF or EGFR antagonist is any other antagonist of EGF or EGFR known in the art. Each possibility represents a separate embodiment of the present invention.
[0204] In another embodiment, the EGF or EGFR antagonist is a carboxypeptidase inhibitor from potato (PCI) protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is a sprouty protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is an Argos protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is a lefty protein or a homologue, fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is an antibody that recognizes EGF or EGFR, or a fragment or mimetic thereof. In another embodiment, the EGF or EGFR antagonist is small molecule inhibitor that binds and reduces the activity of EGF or EGFR. In another embodiment, the EGF or EGFR antagonist is CRM197. In another embodiment, the EGF or EGFR antagonist is IMC-C225 (ImClone Systems, New York, N.Y.). In another embodiment, the EGF or EGFR antagonist is any other antagonist of EGF or EGFR known in the art. Each possibility represents a separate embodiment of the present invention.
[0205] The EGFR of methods and compositions of the present invention has, in another embodiment, the sequence:
MRPSGTAGAALLALLAALCPASRALEEKKVCQGTSNKLTQLGTFEDHFLSLQRMFN NCEVVLGNLEITYVQRNYDLSFLKTIQEVAGYVLIALNTVERIPLENLQIIRGNMYYE NSYALAVLSNYDANKTGLKELPMRNLQEILHGAVRFSNNPALCNVESIQWRDIVSSD FLSNMSMDFQNHLGSCQKCDPSCPNGSCWGAGEENCQKLTKIICAQQCSGRCRGKS PSDCCHNQCAAGCTGPRESDCLVCRKFRDEATCKDTCPPLMLYNPTTYQMDVNPEG KYSFGATCVKKCPRNYVVTDHGSCVRACGADSYEMEEDGVRKCKKCEGPCRKVCN GIGIGEFKDSLSINATNIKHFKNCTSISGDLHILPVAFRGDSFTHTPPLDPQELDILKTVK EITGFLLIQAWPENRTDLHAFENLEIIRGRTKQHGQFSLAVVSLNITSLGLRSLKEISDG DVIISGNKNLCYANTINWKKLFGTSGQKTKIISNRGENSCKATGQVCHALCSPEGCW GPEPRDCVSCRNVSRGRECVDKCNLLEGEPREFVENSECIQCHPECLPQAMNITCTGR GPDNCIQCAHYIDGPHCVKTCPAGVMGENNTLVWKYADAGHVCHLCHPNCTYGCT GPGLEGCPTNGPKIPSIATGMVGALLLLLVVALGIGLFMRRRHIVRKRTLRRLLQERE LVEPLTPSGEAPNQALLRILKETEFKKIKVLGSGAFGTVYKGLWIPEGEKVKIPVAIKE LREATSPKANKEILDEAYVMASVDNPHVCRLLGICLTSTVQLITQLMPFGCLLDYVRE HKDNIGSQYLLNWCVQIAKGMNYLEDRRLVHRDLAARNVLVKTPQHVKITDFGLA KLLGAEEKEYHAEGGKVPIKWMALESILHRIYTHQSDVWSYGVTVWELMTFGSKPY DGIPASEISSILEKGERLPQPPICTIDVYMIMVKCWMIDADSRPKFRELIIEFSKMARDP QRYLVIQGDERMHLPSPTDSNFYRALMDEEDMDDVVDADEYLIPQQGFFSSPSTSRT PLLSSLSATSNNSTVACIDRNGLQSCPIKEDSFLQRYSSDPTGALTEDSIDDTFLPVPEY INQSVPKRPAGSVQNPVYHNQPLNPAPSRDPHYQDPHSTAVGNPEYLNTVQPTCVNS TFDSPAHWAQKGSHQISLDNPDYQQDFFPKEAKPNGIFKGSTAENAEYLRVAPQSSE FIGA (GenBank Accession No: NM_005228; SEQ ID No: 8). In another embodiment, the EGFR has a sequence selected from the sequences set forth in GenBank entries NM_201282, NM_201283, NM_201284, BC094761, AF288738, AY588246, AY573061, X17054, AF125253, U48722, K03193, and AY698024. In another embodiment, the EGFR is encoded by a nucleic acid molecule having a sequence set forth in the one of the above GenBank entries. In another embodiment, a biologically active fragment of an EGFR is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0206] The EGF of methods and compositions of the present invention has, in another embodiment, the sequence: MLLTLIILLPVVSKFSFVSLSAPQHWSCPEGTLAGNGNSTCVGPAPFLIFSHGNSIFRIDT EGTNYEQLVVDAGVSVIMDFHYNEKRIYWVDLERQLLQRVFLNGSRQERVCNIEKNV SGMAINWINEEVIWSNQQEGIITVTDMKGNNSHILLSALKYPANVAVDPVERFIFWSSE VAGSLYRADLDGVGVKALLETSEKITAVSLDVLDKRLFWIQYNREGSNSLICSCDYDG GSVHISKHPTQHNLFAMSLFGDRIFYSTWKMKTIWIANKHTGKDMVRINLHSSFVPLG ELKVVHPLAQPKAEDDTWEPEQKLCKLRKGNCSSTVCGQDLQSHLCMCAEGYALSR DRKYCEDVNECAFWNHGCTLGCKNTPGSYYCTCPVGFVLLPDGKRCHQLVSCPRNV SECSHDCVLTSEGPLCFCPEGSVLERDGKTCSGCSSPDNGGCSQLCVPLSPVSWECDCF PGYDLQLDEKSCAASGPQPFLLFANSQDIRHMHFDGTDYGTLLSQQMGMVYALDHD PVENKIYFAHTALKWIERANMDGSQRERLIEEGVDVPEGLAVDWIGRRFYWTDRGKS LIGRSDLNGKRSKIITKENISQPRGIAVHPMAKRLFWTDTGINPRIESSSLQGLGRLVIAS SDLIWPSGITIDFLTDKLYWCDAKQSVIEMANLDGSKRRRLTQNDVGHPFAVAVFEDY VWFSDWAMPSVIRVNKRTGKDRVRLQGSMLKPSSLVVVHPLAKPGADPCLYQNGGC EHICKKRLGTAWCSCREGFMKASDGKTCLALDGHQLLAGGEVDLKNQVTPLDILSKT RVSEDNITESQHMLVAEIMVSDQDDCAPVGCSMYARCISEGEDATCQCLKGFAGDGK LCSDIDECEMGVPVCPPASSKCINTEGGYVCRCSEGYQGDGIHCLDIDECQLGVHSCG ENASCTNTEGGYTCMCAGRLSEPGLICPDSTPPPHLREDDHHYSVRNSDSECPLSHDG YCLHDGVCMYIEALDKYACNCVVGYIGERCQYRDLKWWELRHAGHGQQQKVIVVA VCVVVLVMLLLLSLWGAHYYRTQKLLSKNPKNPYEESSRDVRSRRPADTEDGMSSCP QPWFVVIKEHQDLKNGGQPVAGEDGQAADGSMQPTSWRQEPQLCGMGTEQGCWIP VS SDKGSCPQVMERSFHMPSYGTQTLEGGVEKPHSLLSANPLWQQRALDPPHQMELT Q (GenBank Accession No: NM_001963; SEQ ID No: 9). In another embodiment, the EGF has a sequence selected from the sequences set forth in GenBank entries BC093731, AY548762, and X04571. In another embodiment, the EGF is encoded by a nucleic acid molecule having a sequence set forth in the one of the above GenBank entries. In another embodiment, a biologically active fragment of an EGF is utilized in a method of the present invention. Each possibility represents a separate embodiment of the present invention.
[0207] In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with a Hedgehog protein. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with a nucleotide encoding a Hedgehog protein. In another embodiment, a method of the present invention further comprises the step of contacting the skin region of interest with an activator of a Hedgehog protein. Each possibility represents a separate embodiment of the present invention.
[0208] In one embodiment, the methods of the present invention further comprise the step of administering a 5 alpha-reductase type 2 inhibitor, which in one embodiment, is Finasteride or, in another embodiment, turosteride.
[0209] In another embodiment, the methods of the present invention further comprise the step of administering a reductase inhibitor., which in one embodiment is an FCE--dual inhibitor, which in one embodiment is FCE 28260; FCE 28175, or FCE 27837; in another embodiment is an MK inhibitor, which in one embodiment, is MK 0434, MK 0963, or MK 386; in another embodiment, is an FK Nonsteroidal Inhibitor, which in one embodiment is FK 143; and in another embodiment is a LY Nonsteroidal Inhibitor, which in one embodiment, is LY 191704; and in another embodiment, is a SK&F Inhibitor, which in one embodiment, is SK&F 105657.
[0210] In another embodiment, the methods of the present invention further comprise the step of administering an additional composition. In one embodiment, the composition is Dutasteride (Avodart.RTM., G1198745), Finasteride (Propecia.RTM., Proscar.RTM.), Turosteride, Azelaic acid, Zinc sulphate, CS 891, or a combination thereof.
[0211] In another embodiment, the methods of the present invention further comprise the step of administering an antiandrogen, which in one embodiment is Spironolactone (Aldactone.RTM.), Flutamide (Euflex.RTM., Eulexin.RTM.), Casodex, Inocoterone, an RU Antiandrogen, TZP-4238, Win 49596, Fluridil (Eucapil.RTM.), or a combination thereof.
[0212] In another embodiment, the methods of the present invention further comprise the step of administering a K+ Channel Opener, which in one embodiment, is minoxidil (Rogaine.RTM.), Diazoxide, Cromakalim, Pinacidil, or a combination thereof.
[0213] In another embodiment, the methods of the present invention further comprise the step of administering a vasodilator, which in one embodiment, is minoxidil (Rogaine.RTM.).
[0214] In another embodiment, the methods of the present invention further comprise the step of administering an estrogen blocker, which in one embodiment, is an ICI Estrogen Blocker.
[0215] The subject of methods of the present invention, is, in another embodiment, a human. In another embodiment, the subject is a rodent, in one embodiment, a mouse, in another embodiment, a rat. In another embodiment, the subject is a mammal. In another embodiment, the subject is a vertebrate. In another embodiment, the subject is feline, canine, ovine, or bovine. In another embodiment, the subject is a male. In another embodiment, the subject is a female. In another embodiment, the subject is any other subject known in the art. Each possibility represents a separate embodiment of the present invention.
[0216] In another embodiment, the subject is an adult. In one embodiment, "adult" refers to an age greater than about 18 years. In another embodiment, "adult" refers to an age greater than about 20 years. In another embodiment, "adult" refers to an age greater than about 25 years. In another embodiment, "adult" refers to an age greater than about 30 years. In another embodiment, "adult" refers to an age greater than about 35 years. In another embodiment, "adult" refers to an age greater than about 40 years. In another embodiment, "adult" refers to an age greater than about 45 years.
[0217] In another embodiment, the subject is elderly. In one embodiment, "elderly" refers to an age greater than about 45 years. In another embodiment, "elderly" refers to an age greater than about 50 years. In another embodiment, "elderly" refers to an age greater than about 55 years. In another embodiment, "elderly" refers to an age greater than about 60 years. In another embodiment, "elderly" refers to an age greater than about 65 years. In another embodiment, "elderly" refers to an age greater than about 70 years.
[0218] In another embodiment, the first subject, or, where applicable, both the first subject and the second subject, is a laboratory animal. In another embodiment, the subject(s) is/are mice. In another embodiment, the subject(s) is/are rats. In another embodiment, the subject(s) is/are gerbils. In another embodiment, the subject(s) is/are hamsters. In another embodiment, the subject(s) is/are guinea pigs. In another embodiment, the subject(s) is/are rabbits. In another embodiment, the subject(s) is/are pigs. In another embodiment, the subject(s) is/are dogs. In another embodiment, the subject(s) is/are cats. In another embodiment, the subject(s) is/are primates. In another embodiment, the subject(s) is/are any other laboratory animal known in the art. Each possibility represents a separate embodiment of the present invention.
[0219] In one embodiment, the subject is contacted with FGF9, or in another embodiment, with a composition comprising FGF9. In another embodiment, FGF9 or a composition comprising FGF9 is administered to a subject.
[0220] "Contacting" as used herein refers, in another embodiment, to bringing skin, in one embodiment, scalp, eyebrow, etc, into to contact with a compound, factor, cell, etc. In another embodiment, the term refers to embedding the compound, factor, cell, etc into the skin region of interest. In another embodiment, the term refers to injecting the compound, factor, cell, etc into the skin region of interest. In another embodiment, term refers to any other type of contacting known in the art. Each possibility represents a separate embodiment of the present invention.
[0221] In another embodiment, the step of contacting in methods of the present invention comprises directly contacting the skin region of interest with the compound, RNA, protein, etc. In another embodiment, the step of contacting comprises indirectly contacting the skin region of interest via contacting another site or tissue of the subject, after which the compound, RNA, or protein is transported to the skin region of interest by a biological process; e.g, diffusion, active transport, or circulation in a fluid such as the blood, lymph, interstitial fluid, etc. Each possibility represents a separate embodiment of the present invention.
[0222] In one embodiment, other fibroblast growth factors may be used in the methods of the present invention. In one embodiment, FGF1, FGF2, FGF3, FGF4, FGF5, FGF6, FGF7, FGF8, FGF9, FGF10, or a combination thereof may be used in the methods of the present invention. In one embodiment, FGF1 through FGF10 all bind fibroblast growth factor receptors (FGFRs). In one embodiment, FGF1 is known as acidic fibroblast growth factor, and FGF2 is also known as basic fibroblast growth factor.
[0223] In another embodiment, FGF11, FGF12, FGF13, or FGF14, may be used in the methods of the present invention. In one embodiment, FGF11, FGF12, FGF13, and FGF14 are known as FGF homologous factors 1-4 (FHF1-FHF4), and in another embodiment, have distinct functional differences compared to the FGFs. In one embodiment, these factors possess remarkably similar sequence homology, they do not in one embodiment, bind FGFRs and are involved in intracellular processes unrelated to the FGFs. In one embodiment, this group is also known as "iFGF".
[0224] In another embodiment, FGF16, FGF17, FGF18, FGF19, FGF20, FGF21, FGF22, or FGF23 may be used in the methods of the present invention. In one embodiment, FGF15/FGF19, FGF21 and FGF23 have systemic rather than local effects.
Pharmaceutical Compositions
[0225] In another embodiment, methods of the present invention comprise administering a pharmaceutical composition comprising FGF9 or an up-regulator of SHH and/or its analog, derivative, isomer, metabolite, pharmaceutically acceptable salt, pharmaceutical product, hydrate, N-oxide, or any combination thereof; and a pharmaceutically acceptable carrier. Each possibility represents a separate embodiment of the present invention.
[0226] The pharmaceutical compositions containing FGF9 or an up-regulator of SHH can, in another embodiment, be administered to a subject by any method known to a person skilled in the art, such as topically, parenterally, paracancerally, transmucosally, transdermally, intramuscularly, intravenously, intradermally, subcutaneously, subepidermally, intraperitonealy, intraventricularly, intra-arteriolly, intravascularly, intracranially, intravaginally, intrarectally, or intratumorally. Each possibility represents a separate embodiment of the present invention. In one embodiment, the dosage regimen will be determined by skilled clinicians, based on factors such as exact nature of the condition being treated, the severity of the condition, the age and general physical condition of the patient, body weight, and response of the individual patient, etc.
[0227] In another embodiment, the pharmaceutical compositions are administered orally, and are thus formulated in a form suitable for oral administration, i.e. as a solid or a liquid preparation. Suitable solid oral formulations include tablets, capsules, pills, granules, pellets and the like. Suitable liquid oral formulations include solutions, suspensions, dispersions, emulsions, oils and the like. In one embodiment of the present invention, the FGF9 or a molecule that upregulates SHH (e.g., SHH agonist) as provided herein composition is formulated in a capsule. In another embodiment, the compositions of the present invention comprise, in addition to FGF9 or a molecule that upregulates SHH (e.g., SHH agonist) as provided herein an inert carrier or diluent, or a hard gelating capsule.
[0228] In another embodiment, the pharmaceutical compositions are administered topically to body surfaces and are thus formulated in a form suitable for topical administration. Suitable topical formulations include gels, ointments, creams, lotions, drops, gels; pastes; powders; aerosol sprays; syrups or ointments on sponges or cotton applicators; and solutions or suspensions in an aqueous liquid, non-aqueous liquid, oil-in-water emulsion, or water-in-oil liquid emulsion, and the like. Because of its ease of administration, a cream, lotion, or ointment represents the most advantageous topical dosage unit form, in which case liquid pharmaceutical carriers may be employed in the composition. These creams, lotions, or ointments, may be prepared as rinse-off or leave-on products, as well as two stage treatment products for use with other skin cleansing or managing compositions. In a preferred embodiment, the compositions are administered as a rinse-off product in a higher concentration form, such as a gel, and then a leave-on product in a lower concentration to avoid irritation of the skin. Each of these forms is well understood by those of ordinary skill in the art, such that dosages may be easily prepared to incorporate the pharmaceutical composition of the invention. In one embodiment, a delayed release patch may be used for administration of the composition of the invention. For topical administration, the composition or its physiologically tolerated derivatives such as salts, esters, N-oxides, and the like are prepared and applied as solutions, suspensions, or emulsions in a physiologically acceptable diluent with or without a pharmaceutical carrier.
[0229] Ointment preparations may be roughly classified into fat/oil type ointments, emulsified ointments, water-soluble ointments and suspended ointments according to the type of the base (vehicle) used therefor. An ointment may comprise, for example, fats, fatty oils, lanolin, vaseline, paraffins, waxes, resins, plastics, glycols, higher alcohols, glycerol, water, emulsifiers, suspending agents or other appropriate additives as a diluent, carrier or as a vehicle. Manufacture of an ointment comprises, for example, adding the compound of the present invention to the appropriate additives, diluents, carriers or vehicles followed by mixing to make the mixture homogeneous.
[0230] For parenteral application, particularly suitable are injectable, sterile solutions, preferably oily or aqueous solutions, as well as suspensions, emulsions, or implants, including suppositories and enemas. Ampoules are convenient unit dosages. Such a suppository may comprise any agent described herein.
[0231] For application by inhalation, solutions or suspensions of the compounds mixed and aerosolized or nebulized in the presence of the appropriate carrier suitable. Such an aerosol may comprise any agent described herein.
[0232] For enteral application, particularly suitable are tablets, dragees, liquids, drops, or capsules. In one embodiment, a sweetened vehicle is employed when a syrup, elixir, or the like is used for enteral application.
[0233] For liquid formulations, pharmaceutically acceptable carriers are, in another embodiment, aqueous or non-aqueous solutions, suspensions, emulsions or oils. Examples of non-aqueous solvents are propylene glycol, polyethylene glycol, and injectable organic esters such as ethyl oleate. Aqueous carriers include, in another embodiment, water, alcoholic/aqueous solutions, emulsions or suspensions, including saline and buffered media. Examples of oils are those of petroleum, animal, vegetable, or synthetic origin, for example, peanut oil, soybean oil, mineral oil, olive oil, sunflower oil, and fish-liver oil.
[0234] In another embodiment, the pharmaceutical compositions are administered by subcutaneous implantation of a pellet. In another embodiment, the pellet provides for controlled release of the composition of the invention over a period of time.
[0235] In one embodiment, the pharmaceutical compositions are controlled-release compositions, i.e. compositions in which the composition is released over a period of time after administration. Controlled- or sustained-release compositions include, in another embodiment, formulation in lipophilic depots (e.g. fatty acids, waxes, oils). In another embodiment, the composition is an immediate-release composition, i.e. a composition in which all the composition is released immediately after administration. Sustained or directed release compositions can be formulated, e.g., liposomes or those wherein the active compound is protected with differentially degradable coatings, e.g., by microencapsulation, multiple coatings, etc. It is also possible to freeze-dry the new compounds and use the lyophilisates obtained, for example, for the preparation of products for injection.
[0236] In one embodiment, compositions of this invention are pharmaceutically acceptable. In one embodiment, the term "pharmaceutically acceptable" refers to any formulation which is safe, and provides the appropriate delivery for the desired route of administration of an effective amount of at least one compound for use in the present invention. This term refers to the use of buffered formulations as well, wherein the pH is maintained at a particular desired value, ranging from pH 4.0 to pH 9.0, in accordance with the stability of the compounds and route of administration.
[0237] In one embodiment, FGF9 or upregulators of SHH used in the methods of this invention may be administered alone or within a composition. In another embodiment, compositions comprising FGF9 or upregulators of SHH in admixture with conventional excipients, i.e. pharmaceutically acceptable organic or inorganic carrier substances suitable for parenteral, enteral (e.g., oral) or topical application which do not deleteriously react with the active compounds may be used. In one embodiment, suitable pharmaceutically acceptable carriers include but are not limited to water, salt solutions, alcohols, gum arabic, vegetable oils, benzyl alcohols, polyethylene glycols, gelatine, carbohydrates such as lactose, amylose or starch, magnesium stearate, talc, silicic acid, viscous paraffin, white paraffin, glycerol, alginates, hyaluronic acid, collagen, perfume oil, fatty acid monoglycerides and diglycerides, pentaerythritol fatty acid esters, hydroxy methylcellulose, polyvinyl pyrrolidone, etc. In another embodiment, the pharmaceutical preparations can be sterilized and if desired mixed with auxiliary agents, e.g., lubricants, preservatives, stabilizers, wetting agents, emulsifiers, salts for influencing osmotic pressure, buffers, coloring, flavoring and/or aromatic substances and the like which do not deleteriously react with the active compounds. In another embodiment, they can also be combined where desired with other active agents, e.g., vitamins.
[0238] In one embodiment, the therapeutic compositions of the present invention comprise an FGF9 composition and one or more additional compounds effective in preventing or treating dermatologic conditions such as alopecia. In one embodiment, the additional compound is a moisturizer or an emollient, which in one embodiment is petrolatum, white petrolatum, hydrogenated vegetable oil, hydrophilic petrolatum, panthenol, primrose oil, omega-3 fish oils, omega-6 fish oils, linoleic acid, flax seed oil, ceramide, borage oil (linoleic acid), tocopherol (Vitamin E), tocopherol linoleate, dimethicone, glycerine or a combination thereof. In one embodiment, moisturizers improve the ability of the skin to absorb other administered compounds, including inter alia, the compounds for use in the present invention. In another embodiment, moisturizing agents minimize or prevent the skin from drying and cracking, thereby decreasing susceptibility of skin to environmental factors that generate free radicals, thereby preventing additional damage to the skin.
[0239] In another embodiment, the additional compound is a topical steroid, which in one embodiment is hydrocortisone, in one embodment 1% hydrocortisone, triamcinolone, fluocinolone acetonide, halcinonide, halobetasol propionate, clobetasol propionate, betamethasone dipropionate, betamethasone valerate, and triamcinolone acetonide or a combination thereof; oral steroids; topical immunomodulators including, inter alia, tacrolimus, pimecrolimus, Ascomycin, cyclosporine, or a combination thereof; antihistamines, which in one embodiment is hydroxyzine or diphenhydramine hydrochloride, Ketotifen, Doxepin; biologics, which in one embodiment comprises Amevive (alefacept), Enbrel, Humira, Raptiva, Remicade, or a combination thereof; or a combination thereof. In another embodiment, the additional compound is an antibiotic, which in one embodiment comprise tetracycline, doxycline, minocycline, cloxacillin, cephalexin, penicillin, clindamycin or a combination thereof. In another embodiment, the additional compound is methotrexate, tar, coal tar, anthralin, dovonex, salicyclic acid, tazorac, moisturizers, aloe vera, soriatane, accutane, hydrea, mycophenolate mofetil, sulfasalazine, 6-thioguanine, or a combination thereof. In another embodiment, additional compounds comprise acyclovir, which in one embodiment is particularly effective in patients with eczema herpeticum. In one embodiment, additional compounds to treat seborrheic dermatitis comprise zinc pyrithione, selenium sulfide, sulfur, tar shampoo, flucinolone acetonide solution, triamcinolone acetonide lotion, ketoconazole cream, other imidazoles, or a combination thereof.
[0240] In another embodiment, the additional compound is an anti-inflammatory agent, which in one embodiment comprises aspirin, ibuprofen, ketoprofen, naproxen, or a combination thereof. In another embodiment, the additional compound is a prostaglandin or prostaglandin inhibitor, which in one embodiment is an inhibitor of PGD2.
[0241] In another embodiment, the additional compound is an exfoliant, which in one embodiment comprises an enzymatic exfoliant or a mono- or -poly-hydroxy acid. In one embodiment, the exfoliant is an alpha-hydroxy acid, beta-hydroxy acid, tannic acid, glycolic acid, lactic acid, citric acid, salicylic acid, or a combination thereof. In another embodiment, the additional compound is an analgesic, or anesthetic, while in another embodiment it is aloe vera gel, aloe vera, licorice extract, pilewort, Canadian willow root, zinc, allantoin, or a combination thereof. In another embodiment, the additional compound is an anti-oxidant.
[0242] In one embodiment, fibroblast growth factor-9 protein is administered at a concentration of 10 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 20 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 40 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 80 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 5 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 3 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of 1 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of between 1 and 50 ng/mL. In another embodiment, fibroblast growth factor-9 protein is administered at a concentration of between 1 and 15 ng/mL. Each dose represents a separate embodiment.
[0243] In general, the doses utilized for the above described purposes will vary, but will be in an effective amount to exert the desired effect. As used herein, the term "pharmaceutically effective amount" refers to an amount of a FGF9 or other composition for use in the present invention, which will produce the desired alleviation in symptoms or other desired phenotype in a patient. The doses utilized for any of the above-described purposes will generally be from 1 to about 1000 milligrams per kilogram of body weight (mg/kg), administered one to four times per day, or by continuous IV infusion. In one embodiment, a topical daily dose range, in single or divided doses, for the conditions described herein is from about 1 mg to 20,000 mg, more preferably about 2,000 mg to 16,000 mg, and most preferably about 6,000 mg to 10,000 mg of the active components (i.e., excluding excipients and carriers). When the compositions are dosed topically or intraocularly, they will generally be in a concentration range of from 0.1 to about 10% w/v, administered 1-4 times per day. In one embodiment, the compositions for use in the methods of the present invention are administered topically two times a day.
[0244] In one embodiment of the invention, the concentrations of the compounds will depend on various factors, including the nature of the condition to be treated, the condition of the patient, the route of administration and the individual tolerability of the compositions.
[0245] In one embodiment, the administering step is via topical administration. In another embodiment, the administering step is via subcutaneous administration.
[0246] In one embodiment, the compound administered as part of methods of the present invention is administered systemically. In another embodiment, the compound is administered topically. In another embodiment, the compound is administered subepidermally. In another embodiment, the compound is administered subcutaneously. In another embodiment, the compound is administered transdermally. In another embodiment, the compound is administered to the site of the abrasion. In another embodiment, the compound is administered to the site of the wound induction. In another embodiment, the compound is administered to the site of the depilation. In another embodiment, the compound is administered during wound healing. In another embodiment, the compound is administered prior to HF neo-genesis. In another embodiment, the compound is administered during HF neo-genesis. Each possibility represents a separate embodiment of the present invention.
[0247] In one embodiment, the route of administration may be directed to an organ or system that is affected by alopecia. For example, compounds may be administered topically to treat dermatologic conditions such as alopecia. In another embodiment, the route of administration may be directed to a different organ or system than the one that is affected by dermatologic conditions such as alopecia. For example, compounds may be administered parenterally to treat dermatologic conditions such as alopecia. Thus, the present invention provides for the use of FGF9 or other composition for use in the present invention in various dosage forms suitable for administration using any of the routes listed hereinabove.
[0248] In one embodiment, the methods of the present invention of testing a compound are repeated using a plurality of subjects, until a statistically significant sample has been tested.
[0249] In one embodiment, FGF9 augments hair germ formation in embryonic tissue, but is not essential for hair germ formation. In one embodiment, FGF9 is necessary for hair follicle formation and/or size in adult tissue after epidermal disruption.
[0250] In one embodiment, the signaling pathway for embryonic hair germ formation and for wound-induced hair follicle neogenesis share one or more components. In another embodiment, the signaling pathway for embryonic hair germ formation and for wound-induced hair follicle neogenesis is not identical. Thus, in one embodiment, FGF9 is essential for hair follicle formation in WIHF but not during ED13.5.
[0251] In another embodiment, the signaling pathway for hair growth signals differs in different parts of the body. Thus, in one embodiment, embryonic hair germ formation and wound-induced hair follicle neogenesis differ in their dependence on FGF9 due to the differences in their respective developmental stages and differences in location in the body.
[0252] In one embodiment, the combination of FGF9 and wound healing increases its efficacy as a hair growth promoter. In one embodiment, FGF9 application alone causes epidermal thickening.
[0253] In another embodiment, the invention provides a method of treating hair loss or regenerating hair follicles in a subject comprising the step of disrupting the epidermis in the region of said hair loss in said subject. In some embodiments, the method further comprises the step of recruiting the gamma-delta T cells to the wound epidermis. In an exemplary embodiment, the method further comprises the step of recruiting the gamma-delta T cells to the wound epidermis through cytokines.
EXAMPLES
Experimental Details
Depilation and Epidermal Abrasion
[0254] Mice were anesthetized with an injection of sodium pentobarbital before the hair on the back was clipped and depilated with Nair (Carter-Wallace, New York, N.Y.), then epidermis was removed using a rotating felt wheel as described by Argyris T, J Invest Dermatol, 75: 360-362, 1980). After scrubbing with 70% ethanol and drying under an incandescent lamp, the basal and supra-basal layers in an area of (1.5 cm).sup.2 cm of the inter-follicular epidermis were removed by careful abrasion with a felt wheel mounted on a Dremel Moto-tool (Racine, Wis.). After abrasion, the skin was shiny and smooth, and there was no blood. One day later, the abraded area was covered by a fibrin crust, which fell off after 3-7 days, exposing the newly regenerated epidermis. A group of control mice was sacrificed immediately after abrasion to confirm microscopically the complete removal of the interfollicular epidermis.
Punch Wound and Excisional Wound Induction
[0255] The backs of 21-day-old mice were depilated as described for Example 1 and sterilized with alcohol, followed by 1% iodine solution. Punch wounds, 4 mm in diameter, were induced using a dermal biopsy punch, down to, but not through, the muscle fascia. Excisional wounds were full thickness and 1 cm in diameter; skin and panniculus carnosus was excised using fine surgical scissors.
Immunohistochemistry
[0256] Skin samples were fixed in PBS-buffered 10% formalin. Six-micron thick paraffin sections were cut and stained, where applicable, with antibodies.
Whole Mounting and Immunofluorescence
[0257] HF whole mounts were obtained by incubating fresh skin with EDTA (20 mM in PBS) at 37.degree. C. overnight, then separating the epidermis and dermis. Epidermis was then fixed in 10% formalin for 10 min, room temperature (RT). Dermis was fixed in acetone overnight, RT. After rinsing with PBS, whole mounts were stained with antibodies for immunohistochemistry (schematically depicted in FIG. 12) and were imaged using a Leica confocal microscope.
Statistics
[0258] Hair follicle numbers are expressed as mean.+-.s.d. The student's two-tailed t-test function in Excel was used to calculate P values.
Embryonic Mouse Skin Culture Protocol
[0259] The following materials were used: Center well dishes (Fisher 08-772-12); Metal grids (Goodfellow 688-485-21); Nitrocellulose filters (Millipore AABP04700); Media: DMEM+5% FBS+1.times. Pen/Strep.
[0260] Gestational day 13.5 timed pregnant mothers (Charles River) were ordered. Center well dishes were set up with 2 ml media/dish. Metal grid was placed in center well. Dishes were stored in incubator so the media warmed to 37.degree. C. Nitrocellulose filter were cut into rectangles and placed in a beaker of dH.sub.2O on a hot plate. Water was allowed to boil and then filters were boiled for 10 min.
[0261] Two petri dishes with sterile PBS were prepared. Mothers were euthanized and embryos were dissected out in the sac. Embryos were placed into one petri dish. The embryos were dissected out of the sac and placed in a second clean petri dish with sterile PBS. The dish of embryos were placed on ice. Dorsal skin was dissected from the embryo under a dissecting scope, in a clean petri dish containing sterile PBS.
[0262] The crown-rump length of the embryo was checked with a ruler to ensure it is E13.5 stage. (.about.10-10.3 mm). The head of the embryo was removed with micro dissecting scissors. A smaller pair of micro dissecting scissors were used to make incisions along both sides of the back, above the limbs. A third incision was made across the back, anterior to the tail. Using fine tipped Dumont tweezers, the skin was peeled from the tail towards the head. The skin was laid onto the black side of nitrocellulose filter, as flat as possible. The nitrocellulose filter was placed onto metal grid so that the skin is at the liquid-air interface. The dish was incubated at 37.degree. C. When all skins were dissected, compounds were added to culture media (if necessary), and returned to the incubator. Skins were cultured for up to 3 days. Placodes started to develop on E14.5.
Example 1
FGF9 Expressed in Early Period of Hair Germ Formation
[0263] FGF9 mRNA expression was evaluated in regenerated epidermis by quantitative real time-PCR. FGF9 was expressed at higher levels prior to the earliest stages of hair follicle regeneration at Day 1 after scab detachment (SD; which occurs at reepithelialization) compared to Day 5 after scab detachment when follicles have formed (FIG. 1). Skin .gamma..delta. T-cells (detected by immunostaining using antibodies against .gamma..delta.TCreceptor) repopulate the reepithelialized epidermis by SD7 (FIG. 2, left panel) and these cells express FGF9 protein at SD1 (red dendritic cell in epidermis, FIG. 2, right panel, and FIG. 3).
[0264] Thus, FGF9 was selectively expressed prior to hair germ formation (during the undifferentiated period) rather than during differentiation. Skin .gamma..delta. T cells appeared to be the source of FGF9, which suggests inflammatory cells may have a role in Wound-induced hair follicle neogenesis (WIHN).
Example 2
FGF9 Expressed in Embryonic Day 14 (E14) Skin
[0265] FGF9 (red staining) is expressed by .gamma..delta.TC (green staining) in embryonic day 14 (E14) skin (FIG. 4).
Example 3
FGF9 Plays a Role in Wound-Induced Hair Follicle Neogenesis (WIHN)
[0266] Anti-FGF9 neutralization experiment in adult mice
[0267] 3 week-old (adult) C57BL/6 mice were subjected to the wounding model as described hereinabove. Mice then received subepidermal injections of 50 .mu.l of 10 .mu.g/ml anti-FGF9 or IgG2a isotype control on days SD1-SD4. Tissue samples were taken and analyzed at SD5.
[0268] Immunoblots were used to verify the specificity of the anti-FGF9 neutralization antibody. Mouse FGF9 has 198 bp and greater than 99% homology with human FGF9 (with only one amino acid difference). FGF9 exists in both monomer (25-27 kd) and dimer forms. Immunoblots demonstrated the presence of both the 26 Kd monomer and the 52 Kd dimmer forms in E14.5 mouse embryonal whole cell lysates, as well as in control samples containing recombinant hFGF9 (FIG. 5).
[0269] Mice receiving anti-FGF9 antibody had significantly lower hair follicle numbers on SD5 than IgG2a controls (FIG. 6). Thus, FGF9 plays a role in wound-induced hair follicle neogenesis.
[0270] The developmental stages of the hair follicles were quantitated as described in Paus R et al., J Invest Dermatol 1999. There was a decrease in mature hair follicles and an increase in immature hair follicles in the anti-FGF9 treated group (FIG. 7).
Example 4
FGF9 Plays a Role in Embryonic Skin Development
[0271] Scheduled pregnant C57BL/6 mice were sacrificed at E13.5, and embryonic whole back skin was dissected. E13.5 skin was cultured for three days floated on filter paper with metal grid.
[0272] To determine the role of FGF-9 in hair follicle neogenesis in embryonic skin, embryonic skin explant cultures were treated for three days with recombinant human (rh)FGF9 (control, 10, 20, or 40 ng/mL) or with an anti-FGF9 neutralizing antibody (control, 10, 20, or 40 .mu.g/mL) or IgG2a isotype control (10, 20, 40 .mu.g/mL). Alkaline phosphatase (AP) for dermis immunostaining (FIG. 10) and K17 was used for epidermis immunostaining (FIG. 12).
[0273] Hair germ counting was performed at three separate fields per sample and was evaluated per mm.sup.2 (FIG. 8). q-PCR for Shh, Ptch1, Ptch2, Gli1, and Gli2 was performed after 24 h of rhFGF9 treatment.
Real-Time PCR Protocol
[0274] The following materials were used: RNeasy.RTM. fibrous tissue mini kit (Qiagen, 74704); High capacity cDNA reverse transcription kit (Applied Biosystems, P/N 4368814); Taqman.RTM. Fast universal PCR master mix (2.times.) (Applied Biosystems, P/N 4352042); Applied Biosystems StepOne.TM. real-time PCR system; (Applied Biosystems, P/N 4376373); MicroAmp.TM. 48-well optical adhesive film (Applied Biosystems, P/N 4375928); MicroAmp.TM. 48-well reaction plate (Applied Biosystems, P/N 4375816).
[0275] The following PCR primers were used (Taqman.RTM. gene expression assay, Applied Biosystems):
TABLE-US-00001 TABLE 1 Target gene Gene name Assay primer ID Reference sequence Fgf9 fibroblast growth Mm00442795_m1 NM_013518.3 factor 9 Shh Sonic hedgehog Mm00436527_m1 NM_009170.3 Ptch1 patched homolog 1 Mm00436026_m1 NM_008957.2 Ptch2 patched homolog 2 Mm00436047_m1 NM_008958.2 Gli1 GLI-Kruppel family Mm00494645_m1 NM_010296.2 member GLI1 Gli2 GLI-Kruppel family Mm01293116_m1 NM_001081125.1 member GLI2 ACTB actin, beta P/N 4352933E NM_007393.1 (endogenous control)
Protocol for q-PCR with Cultured Embryonic Skin Samples
[0276] Embryonic skin culture: E13.5 timed pregnant B57BL/6 female mice (Charles-River) were euthanized in CO.sub.2 chamber. Embryos were dissected and placed in sterile cold PBS on ice.
[0277] Preparation: Millipore nitrocellulose membrane (0.5.times.1.0 cm2); Autoclave metal grids; Culture dishes (Falcon center-well organ culture dish, 35-3037); 5% FBS-DMEM (lx penicillin/streptomycin, not necessary to inactivate FBS).
[0278] 2.5 mL of culture media was added and metal mesh and nitrocellulose membrane were set on the individual culture dishes. Embryonic back skin was dissected. Head & buttock area were cut. Dissection was through flank in a caudo-cranial direction. Dissected back skin was loaded on Millipore membrane (dermal side down). The samples were prepared in triplicate per needed for each concentration. Skin samples were cultured for 24 hr at 37.degree. C. in 5% CO.sub.2.
RNA Isolation & cDNA Preparation
[0279] Skin samples were incubated in 20 mM EDTA for 10 min. Epidermis and dermis were separated with fine-tipped tweezers under a dissecting microscope, respectively.
[0280] Samples were disrupted with a homogenizer and total RNA extracted with RNeasy.RTM. fibrous tissue mini kit (Qiagen, 74704) following manufacturer's information.
[0281] RNA concentration was measured by spectrophotometer and then converted to gg of total RNA to cDNA using High capacity cDNA reverse transcription kit (Applied Biosystems, P/N 4368814) with program in thermal cycler.
Real-time PCR
[0282] PCR running program was set up and arranged the reaction plate layout with provided StepOne software in comparative CT (.DELTA..DELTA.CT) method. The reaction mixture of target gene and .beta.-actin endogenous control were prepared together in triplicate. cDNA template was diluted from stock to final total cDNA amount of 30-50 ng in 2 .mu.l.
[0283] Reaction Mix Components
TABLE-US-00002 Component Volume (.mu.l) for 1 reaction Taqman .RTM. Fast universal 10.0 PCR master mix (2X) PCR primers 1.0 (Taqman .RTM. gene expression assay) H2O 7.0 cDNA template 2.0 Total volume 20.0
[0284] 2. Prepare the reaction plate: A reaction volume of 20 .mu.l/well is added on 48-well reaction plate. The plate is sealed tightly with optical adhesive film.
[0285] 3. Load the plate into StepOne instrument and start the programmed reaction.
[0286] 4. Analyze the results with the StepOne software and obtain relative quantitation data of gene expression.
[0287] The effect of rhFGF9 treatment for three days in the dermis of embryonic skin explant culture (E13.5) was dose-dependent, with 10 ng/mL and 20 ng/ml resulting in an increase in hair germ number/mm.sup.2, while a 40 ng/mL dose resulted in decreased hair germ number/mm.sup.2 (FIGS. 9-10). On the other hand, there was no discernable effect of anti-FGF9 neutralizing antibody treatment for three days in the epidermis or dermis of embryonic skin explant culture (E13.5; FIGS. 11-13).
[0288] 24-hr treatment of E13.5 embryonic skin explant culture with 10 ng/mL of rhFGF9 resulted in increases in markers of embryonic hair follicle development including sonic hedgehog (Shh), Ptch1, Ptch2, and Gli1, particularly in the epidermis (FIGS. 14-15).
[0289] Fibroblast growth factor 9 increases hair follicle formation when injected into the wound after healing. This is just prior to and during the time when new hair follicles are forming. FGF9 also increases hair follicle formation during hair follicle development by using embryonic mouse skin explanted in culture. These findings support the notion that wounding converts the epidermis to a "receptive" state in which it responds to exogenous factors.
Example 5
Overexpression of FGF9 in Basal Keratinocytes
[0290] The gain of function mutant TRE-fgf9-IRES-eGfp;K5-rtTA (.times.Ptch1-LacZ reporter) (White et al., Development 133, 1507-1517, 2006; Diamond, et al. (2000) J. Invest. Dermatol. 115, 788-794, both incorporated herein by reference) is used to validate that early FGF9 expression in hair neogenesis stage would enhance hair follicle development and to confirm that FGF9 is upstream of Shh signaling.
Example 6
Deletion of FGF9 Expression in .gamma..delta. T Cells
[0291] Deletion of FGF9 expression in .gamma..delta. T cells is accomplished using the loss of function mutant FGF9flox/flox;lck-cre to selectively delete FGF9 using T-cell targeting lck-cre promoter. The Lck-Cre uses the proximal promoter of the Lck (lymphocyte protein tyrosine kinase) gene, which is first expressed early in thymocyte development at the double negative stage. After T cells fully mature, the level of expression of this transgene decreases by approximately 10 fold. This particular mouse gene shows a high degree of expression of the transgene in the thymus and has been found to bring about the selective deletion of genes flanked by loxP targeting sequences in almost all early thymocytes. It thus is used to delete a specific gene in the T cell lineage starting at the double negative stage. Since the homozygous Lck-Cre mice strains are crossed to a strain containing a floxed FGF9 and offspring with deleted FGF9 in the T cell lineage are obtained. Control animals are obtained in the same litter by typing for the presence or absence of the floxed gene in genomic DNA tail samples. This system is described in more detail in Lee et al., Immunity November 2001:15(5) 763-74, which is incorporated herein by reference.
Example 7
K17-EGFP Reporter Mice
[0292] K17-eGFP reporter mice (Bianchi et al., Mol Cell Biol., 2005 August; 25(16): 7249-7259, incorporated herein by reference) are used to confirm the accumulation of FGF9-producing .gamma..delta. T-cells around newly developing hair germs.
Example 8
FGF9 Mediates Hair Follicle Neogenesis Through Epidermal .gamma..delta. T Cells
[0293] Understanding molecular mechanisms responsible for hair follicle regeneration during wound healing raises the opportunity to develop new treatments for hair loss and other skin disorders. Here, it is clearly shown that Fibroblast Growth Factor 9 (Fgf9) modulates hair follicle formation following wounding of adult mice. Forced overexpression of Fgf9 in the newly formed wound epidermis results in a 2-3-fold increase in the number of neogenic hair follicles. Remarkably, during wound healing in normal mice, .gamma..delta. T cells, which reside in the epidermis, serve as the primary source for Fgf9. Specific deletion of the Fgf9 gene in T cells using Lck-Cre;floxed fgf9 transgenic mice results in a marked reduction of hair follicle neogenesis following wounding. Similarly, mice lacking .gamma..delta. T cells demonstrate severe impairment of follicular neogenesis. Overall, these findings explain the robustness of hair follicle regeneration in mouse compared to human and highlight the important relationship between the immune system and tissue regeneration.
[0294] Materials and Methods:
[0295] Mice and Wounding.
[0296] Full thickness excision (FTE) of skin was performed on the back of C57BL/6J mice (Jackson laboratory) under ketamine/zylazine anesthesia as previously described (1). Three-week-old mice were used for all experiments with a 1.times.1 cm.sup.2 FTE, except as indicated. Timed pregnant C57BL/6 female mice of gestational day 13.5 (Charles River) were utilized for embryonic skin explant culture. K14-rtTA mice harboring the doxycycline-sensitive transactivator were mated to TRE-Fgf9-IRES-eGfpmice. Both K14-rtTA and K14rtTA/TRE-Fgf9 mice were fed Dox-containing food (Bio-SERV) for 4 days after complete reepithelization. Deletion of FGF9 expression in .gamma..delta. T cells was accomplished using Fgf9 flox/flox mated to Ick-cre mice (Jackson Laboratory) with T-cell targeting proximal promoter of the lymphocyte protein tyrosine kinase (lck). .gamma..delta. T cell null mice (Tcrd.sup.-/-) were purchased from Jackson Laboratory. All animal protocols were approved by the University of Pennsylvania IACUC.
[0297] Whole-Mount Hair Follicle Neogenesis Assay.
[0298] Healed skin was taken at day 5 after reepithelization. Whole-mount hair follicle neogenesis assays for epidermal KRT17 immunostaining (1:5000, from P. Coulombe) and dermal NBT/BCIP incubation were performed to identify new hair germs and follicular dermal papillae in wound area as previously described.
[0299] Real-Time PCR.
[0300] Dorsal skins were as day 0 samples or the wounded skin at day 1, 3 and 5 after scab detachment after reepithelization (SD), respectively. The epidermis was separated from dermis by incubation with 4.degree. C. dispase overnight or 20 mM EDTA for 30 min at 37.degree. C. RNA was isolated using RNeasy minikit (Qiagen) and then 1 .mu.g of total RNA was converted to cDNA with a High capacity cDNA kit (Applied Biosystems). All primer sets including fgf9 of Taqman gene expression assay were purchased from Applied Biosystems. Reactions were performed in triplicate and relative expression levels were standardized using f3-actin as an internal control. The results were analyzed using StepOne program.
[0301] Immunostaining.
[0302] Reepithelialized skin after wounding was placed either frozen in OCT (Tissue-Tek). Staining for FGF9 (1:200; R&D systems) and .gamma..delta.TCR (1:100; GL3, BD Bioscience) were performed on 8 .mu.m frozen section. Immunohistochemisty with antibodies against BrdU (1:500; Harlan-Seralab) was done as previously described. For pulse-chase experiments, BrdU (Sigma) was administered 2 hr before sample preparation.
[0303] Isolation of DETCs and Activation of the Cells.
[0304] Epidermal cell suspension was prepared from C57BL6 mice and was incubated overnight at 37.degree. C. in complete DMEM containing 20U/ml of recombinant mouse IL-2 (mIL-2) to allow surface receptor re-expression as described. DETCs were isolated by FACS sorting with PE-.gamma..delta.TCR (GL3, Abcam) and allophycocyanin-Thy1.2 (BD Bioscience) staining. The isolated DETCs were cultured in RPMI-1640 medium supplemented with 10% FCS, 25 mM HEPES, 100 U penicillin, 100 .mu.g streptomycin, 2 mM glutamine, 100 .mu.M nonessential amino acids, 1 mM sodium pyruvate, 50 .mu.M 2-mercaptoethanol and 20 U/ml mIL-2. For cell stimulation, the cells were harvested for 4 h in the growth factor-free media excluding FCS and mIL-2 and then incubated in the media described above supplemented with anti-CD3.epsilon. (10 .mu.g/ml, eBioscience) at 37.degree. C. for 4, 24 and 48 h. Stimulation was arrested by the addition of ice-cold PBS and samples were placed on ice. Supernatants were removed and cells were collected with lysis buffer. RNA isolation and subsequent cDNA generation were performed with Gene expression cells-to C.sub.T kit (Applied Biosystems).
[0305] Wholemount Epidermal .gamma..delta.T Cell Staining.
[0306] Ears were cut from 8-week old C57BL/6J mice and epidermal sheets were separated as previously described. Epidermal sheets were incubated overnight in the growth factor-free media described above or complete media with 20 U/ml mIL-2 at 37.degree. C. Epidermis was then washed in PBS and fixed in ice-cold acetone for 20 min at -20.degree. C. Primary antibodies of FGF9 and .gamma..delta.TCR mentioned above were incubated overnight at 4.degree. C. The following morning, sheets were incubated with secondary antibodies for 1 hr and mounted on silane-coated slides.
[0307] In Vitro Embryonic Skin Culture.
[0308] Embryos at E13.5 were dissected out of the sac and the crown-lump length was checked to ensure exact developmental age. Dorsal skin was dissected and then cultured for up to 3 days as previously described. Recombinant human FGF-9 (0-20 ng/ml, R&D systems) or EDA1 (50 ng/ml, R&D systems) as a positive control were added into culture media. In addition, FGF9 neutralization experiment was paralleled with anti-FGF9 antibody incubation (0-40 .mu.g/ml; MAB273, R&D systems). Epidermal-dermal separations were performed by incubating skin samples in 20 mM EDTA at 37.degree. C. for 5 min. Tissues were homogenized to isolated RNA or harvested for wholemount assay as described above. The number of hair follicles was counted per mm.sup.2 at 3 different fields of each sample and the mean value was calculated.
[0309] Neutralization Experiment in Adult Mice.
[0310] Reepithelization of epidermis, indicated by scab detachment, was complete 10-12 days after FTE. One day after complete reepithelization, 50 .mu.l of anti-FGF9 neutralization antibody or IgG isotype control (MAB 003) at 10 .mu.g/ml were daily injected just beneath epidermis for 4 consecutive days. After then, tissues were harvested at day 5, epidermis and dermis were separated using 20 mM EDTA solution and processed for KRT17 immunostaining and detecting alkaline phosphatase activity, respectively. The number of regenerated hair follicles was characterized with respect to their density inside the epidermis. The developmental stages of the hair follicles were quantified as previously described.
[0311] In Vivo Confocal Microscopy.
[0312] To chase dynamic process of hair follicle neogenesis, the changes of newly formed hair follicle number was quantified using in vivo confocal microscope (Vivascope 1500, Lucid). Briefly, surrounding area of healed skin was clipped and adhesive window (Lucid) and ultrasonic transmission gel (Parker laboratory) were applied under ketamine/zylazine anesthesia. New hair follicles could be visualized and counted at the level beneath epidermal-dermal junction. The number was measured at day 2 after reepithelization and then every 3 days for 2 weeks.
Results
[0313] Fgf9 Expression Significantly Increases after Wounding Prior to Hair Follicle Neogenesis:
[0314] To define molecular events responsible for hair follicle neogenesis following wounding, we compared gene expression in wounded epidermis soon after reepithelialization (1 and 3 days after scab detachment "SD") to the initiation of hair follicle neogenesis (SD5). Microarray analyses showed that Fibroblast growth factor 9 (Fgf9) was significantly upregulated (4.2 fold) prior to hair follicle germ formation. We further analyzed Fgf9 gene expression changes in reepithelialized epidermis around the time of hair follicle neogenesis by quantitative RTPCR (FIG. 16A). Fgf9 gene expression increased significantly after reepithelialization until the initial stages of hair follicle neogenesis when expression decreased dramatically. These results show that Fgf9 is upregulated in the newly formed epidermis just prior to hair follicle neogenesis presumably at a time when cells are committing to the hair follicle lineage.
[0315] Inhibition of Fgf9 Decreases Hair Follicle Neogenesis.
[0316] Fgf9 is a secreted ligand with a known role in lung, kidney and gonad development, but it has not been previously implicated in hair follicle development or regeneration. Nevertheless, the main receptor for Fgf9 in the skin, Fgr3b,is expressed in epidermis and is upregulated in regenerated skin after wounding. To address the importance of Fgf9 in hair follicle neogenesis following wounding, we injected Fgf9 neutralizing antibody into the reepithelialized skin daily for four days (FIG. 16b, Table 2). Wounds treated with anti-Fgf9 antibody showed a significant reduction of new hair follicle formation when compared with controls. The hair follicles that did form in anti-Fgf9-treated wounds were in immature stages of development (FIG. 19).
[0317] Forced Overexpression of Fgf9 in the New Epidermis Increases Hair Follicle Formation.
[0318] Since blocking Fgf9 inhibited hair follicle neogenesis, we asked whether increasing levels of Fgf9 in the wound would promote hair follicle neogenesisfollowing wounding. We used a doxycycline-inducible transgenic mouse (K14rtTAx TRE-Fgf9-IRES-eGfp) to inducibly target Fgf9 expression to the epidermis following wound re-epithelialization. Administration of doxcycline from SD1 to SD4 increased Fgf9 expression 150-fold (FIG. 20) compared to doxycycline treated control mice. This targeted overexpression of Fgf9 to the epidermis for four days after reepithelialization led to a marked increase in the number of hair follicles compared to controls (FIG. 16c, Table2).
TABLE-US-00003 TABLE 2 Hair follicle neogenesis assay. The number of new hair follicles were counted at Day 5 after reepithelization. Hair follicle No. Mice Experiment mice (mean .+-. SD) No. Range P-value Deletion of lck-cre;Fgf9.sup.flox/flox 9.1 .+-. 16.7 11 0-49 <0.05 FGF9 Fgf9.sup.flox/flox, Fgf9.sup.flox/+ (Control) 30.7 .+-. 34.0 15 1-131 in T cells FGF9 Double transgenic K14rtTA; 168.2 .+-. 117.1 12 2-189 <0.05 overexpression TRE-Fgf9-IRES-eGfp Single transgenic K14rtTA, 64.8 .+-. 50.3 21 26-431 TRE-Fgf9-IRES-eGfp (Control) Absence of NS .gamma..delta. T cells 8-week old wild-type 43.4 .+-. 31.7 8 1-87 (1.5 .times. 1.5 cm.sup.2 wounding) 24-40 week old wild-type 36.7 .+-. 24.5 6 1-76 (1.5 .times. 1.5 cm.sup.2 wounding) 8-week old .gamma..delta. T cell null mice 9.8 .+-. 10.1 13 0-27 (1.5 .times. 1.5 cm.sup.2 wounding) 24-40 week old.gamma..delta. T cell null mice 7.8 .+-. 13.7 8 0-39 {close oversize bracket} 0.01 (1.5 .times. 1.5 cm.sup.2 wounding) NS: not significant. indicates data missing or illegible when filed
[0319] Fgf9 expression localizes to .gamma..delta. T cells.
[0320] To identify the source of Fgf9 in re-epithelialized skin of normal mice, we immunostained tissue sections of healed skin prior to HFN. Surprisingly, we discovered that .gamma..delta. T cell receptor-bearing epidermal T cells (DETC), which repopulate the epidermis, express Fgf9.DETCs appear to be the primary source of Fgf9 in epidermis, with little or no contribution from keratinocytes or other epithelial residents (FIG. 17A). Previous gene expression data from basal keratinocytes (Alpha-6-integrin-positive) isolated by FACS showed an absence of Fgf9 expression (NAT BIOTech paper). To further confirm the origin of the Fgf9, we treated unwounded ear epidermis with mIL-2 and analysed wholemount preparations for Fgf9 expression by immunofluorescence. IL2-induced DETCs stained strongly with anti-FGF9 antibodies whereas adjacent keratinocytes exhibited background staining (FIG. 21).
[0321] To determine if FGF9 is constitutively expressed by DETCs in skin or upregulated following stimulation, DETCs were isolated from skin by cell sorting and were cultured in vitro with anti-CD3 and IL2 as previously described (havren ref). Fgf9 mRNA levels increased by greater than 10 fold within 4 hours, followed by diminution to baseline levels within 24 hours. (FIG. 17B). This rapid upregulation contrasts with the much longer 48 hour induction period required for expression of FGF7 and FGF10, two factors known to be secreted by DETCs during wound repair and indicates distinct transcriptional regulatory mechanisms.
[0322] DETCs are Essential for Hair Follicle Neogenesis.
[0323] Since Fgf9 mediates hair follicle neogenesis and DETCs appear to be the primary source of Fgf9 in re-epithelialized epidermis, we hypothesized that activated DETCs repopulate the wound during reepithelialization and secrete FGF9 to induce hair follicle neogenesis. To better define the role of DETCs in hair follicle neogenesis, we studied Tcrd-/- mice that fail to develop these cells.
[0324] We wounded age-matched wild-type and Tcrd-/- mice at 8 or 24-40 weeks of age and quantified hair follicle neogenesis. As previously reported, TCRd-/- mice showed slight delays in wound closure (data not shown), but hair follicle neogenesis was markedly decreased. Quantitation of Fgf9 levels indicated that Fgf9 was consistently negligible in the Tcrd-/- mice.
[0325] As shown in FIG. 18, 8 wk and 40 wk-/- mice exhibited profound defects in HFN, with reductions of >80% in HF numbers compared with wt mice (18A,B, Table 2). Thus, reduced numbers of HFs in -/- mice reflect a true defect in hair follicle neogenesis rather than delayed kinetics of response.
[0326] The above findings supported the hypothesis that activation of DETCs following wounding leads to FGF9 production and subsequent hair follicle neogenesis. Nevertheless, to address the concern that DETCs may have a role in hair follicle neogenesis other than the production of FGF9, mutant (lck-cre.times.Fgf9.sup.flox/flox) mice carrying a deletion of the FGF9 gene specifically in T cells, including DETCs, were analysed for hair follicle neogenesis following wounding. Quantitative rtPCR analyses showed that these mice express low constitutive levels of FGF9 in skin. (FIG. 22). Wounding studies showed that these mutant (lck-cre.times.Fgf9.sup.flox/flox) mice exhibited a dramatic reduction in post-wound hair follicle numbers comparable to that observed in TCRd-/- animals (FIG. 18E, Table 2).
[0327] Taken together, the above described results show that DETCs are essential immunologic contributors to HF neogenesis through the production of FGF9.
[0328] In summary, we discovered that DETCs are the source of FGF9. Further, the findings indicate that more divergent cellular & molecular events could be implicated in HFN after wounding, not exactly the recapitulation of embryonic development, and provide additional evidence that acquired immune system including DETCs would have a role in tissue regeneration.
[0329] FGF9 and DETCs are critical for HFN after wounding. Overexpression of FGF9 in reepithelized epidermis resulted in increase of hair follicle formation. These results show that manipulation of FGF9 expression during wound healing or after reepithelization could be a useful approach to develop a new treatment for hair loss.
Example 9
Hedgehog Stimulates Hair Follicle Neogenesis by Creating Inductive Dermis During Murine Skin Wound Healing
[0330] Activation of the Sonic hedgehog (Shh) pathway reinstalls a regenerative dermal niche, called dermal papilla, which is required and sufficient for HF neogenesis (HFN). Epidermal Shh overexpression or constitutive Smoothened dermal activation results in extensive HFN in wounds that otherwise end in scarring. While long-term Wnt activation is associated with fibrosis, Shh signal activation in Wnt active cells promotes the dermal papilla fate in scarring wounds. These studies demonstrate that mechanisms of scarring and regeneration are not distant from one another and that wound repair can be redirected to promote regeneration following injury by modifying a key dermal signal.
Methods
[0331] Mice.
[0332] All animal protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at NYU School of Medicine. All mice with proper genotype were used for the designed experiments regardless of sex. LSL-Shh mice in which mouse Shh is expressed under the control of .beta.-actin promoter upon Cre medicated excision of stop sequence, were previously generated as described in Wang et al. Axin2-CreER59 and .beta.-catenin fl(ex3) were obtained from indicated researchers. Gli1-LacZ (008211), R26-SmoM2 (005130), K14-CreER (005107), Shh fl/fl (004293), Pdgfra-CreER (018280), Smofl/fl (004526), SM22-rtTA (006875), tetO-Cre (006224), Wls fl/fl (012888), Axin2-LacZ (009120) and R26-Tomato reporter mouse (007908), were purchased from the Jackson laboratory.
[0333] To induce CreER activity, tamoxifen (TAM) treatment was performed by intraperitoneal injection (0.1 mg per g body weight) of a 20 mg per ml solution in corn oil. To induce rtTA activity, mice were administered doxycycline-containing chow (20 g per kg, Bio-Serv).
[0334] Wound Experiment.
[0335] Wound experiments were carried out with 3-4-week-old or 7-8-week-old mice as described in Ito et al. All wounding experiments were performed after anesthetization of mice with isoflurane. Briefly, for full thickness large wound, 1 cm2 (1.times.1 cm) or 2.25 cm2 (1.5.times.1.5 cm) of skin were excised for 3-4-week or 7-8-week-old mice, respectively. For small wound, skin was excised by 4 mm full-thickness biopsy punch (Acuderm Inc.) as published. For loss of function study of Hh pathway, wounds were harvested at PW21d by which the number of neogenic HFs was saturated in control wounds. For gain of function study of Hh pathway, samples were harvested around 30 days after wounding (PW30d.about.) to allow continuous formation of neogenic HFs.
[0336] Whole-Mount HFN Assay.
[0337] Whole-mount HFN assay to detect K17+ hair follicles and AP+DP was performed. To analyze hair follicle (dermal papilla and follicular epithelium) regeneration after wounding, wounded skin was harvested from the mice and incubated in 5 mM or 20 mM EDTA in PBS at 37.degree. C. for 30 min .about.2 h or overnight. The epidermis was gently separated from the dermis under a dissecting microscope (Axiovision Discovery V12, Zeiss, Germany). Both epidermis and dermis were fixed in 4% paraformaldehyde for 10 min at room temperature (RT) and rinsed with PBS. For the epidermis, standard DAB immunohistochemistry (see below) was performed with anti-K17 antibody (ab) (Abcam, 1:500). For the dermis, AP staining was performed. Dermis was incubated in NTMT solution (100 mM NaCl, 100 mM Tris-Cl (pH 9.5), 50 mM MgCl2, 0.1% Tween-20) for 10 min and then incubated in NTMT containing in NBT/BCIP (Roche, 1:50) solution at RT until color was visualized.
[0338] Histochemistry.
[0339] Immunohistochemistry was performed as published. For paraffin sections, tissues were fixed in 4% PFA at 4.degree. C. overnight and rinsed with PBS. Following sequential dehydration in ethanol and xylene, tissues were infiltrated by paraffin and embedded in fresh paraffin. The paraffin-embedded tissue blocks were chilled on ice for 10-15 min and cut at 6 .mu.m thickness. The 6 .mu.m tissue slices were flattened out on warm water and placed on microscope slides and dried out at 37.degree. C. overnight. The tissue sections were rehydrated through xylene and graded series of ethanol (2.times.100%, 90%, 80%, 70%, and 50%) and rinsed with PBS. After antigen retrieval in Tris-EDTA (pH 8.0), the tissue sections were incubated with blocking solution (10% BCS in PBS containing 0.1% Tween-20) for 1 h at RT then appropriate primary antibodies at RT for 2 h or 4.degree. C. overnight. After PBS washing, the tissue sections were incubated with secondary antibodies at RT for 1 h. If necessary, the second and third primary antibodies were used on the same tissue sections with corresponding secondary antibodies. Following PBS rinsing, the stained slides were mounted with mounting medium and stored at 4.degree. C. for analysis. For frozen section, tissues were fixed in 4% PFA on ice for 10 min and embedded in OCT compound on dry ice. The frozen blocks were cut at 10 .mu.m thickness, placed on microscope slides, and dried out at RT. After PBS washing, the tissue sections were incubated in blocking, primary, and secondary ab solutions as described above. All immunohistochemical analysis was observed and photographed on an upright Nikon Eclipse Ti or Zeiss Axiophot microscopes. Following antibodies were used: rabbit anti-K17 (1:500, Abcam), rabbit anti-Lef1 (1:100, Cell signaling), rabbit anti-Noggin (1:100, Abcam), rabbit anti-RFP (1:500, Rockland), rabbit anti-Shh (1:50, Santa Cruz), rabbit anti-SMA (1:100, Thermo Scientific), rabbit anti-F4/80 (1:100, Cell signaling), chicken anti-K14 (1:500, BioLegend), mouse anti-.beta.-catenin (1:500, Sigma), mouse anti-SM22.alpha. (1:100, Abcam), mouse anti-AE13 (1:20, a gift of T. T. Sun), mouse anti-AE15 (1:20, a gift of T. T. Sun), mouse anti-K15 (1:100, NeoMarkers), and rat anti-CD34 (1:50, BD Bioscience). For histology, paraffin sections were stained with hematoxylin and eosin in accordance with a general method. To detect collagen protein, trichrome staining and picrosirius red staining were carried out using Masson's Trichrome Stain Kit and Picrosirius Red Stain Kit, respectively (Polysciences). Trichrome staining was performed at NYUMC experimental pathology core.
[0340] X-Gal Staining.
[0341] X-gal staining was performed as published. Skin wound tissues were fixed in 4% PFA at 4.degree. C. for 30 min and rinsed with PBS. The tissues were incubated in X-gal rinse buffer (2 mM MgCl2, 0.01% Sodium deoxycholate, and 0.02% NP-40 in PBS) for 10 min at RT and then in X-gal (5-bromo-4-chloro-3-indolyl-.beta.-d-galactopyranoside) staining buffer (0.7 mg/ml X-gal, 0.5M K4Fe(CN)6 and 0.5M K3Fe(CN)6 in X-gal rinse buffer) at RT or 37.degree. C. until color was visualized. The tissues were photographed in whole mount using a dissection microscope (Zeiss, Discovery V12). The tissues were then dehydrated in standard graded series of ethanol, embedded in paraffin blocks and cut into 6-.mu.m-thick sections. To visualize nucleus, the tissues were counterstained with nuclear fast red solution. Small wounds of Axin2-LacZ mice were incubated in 20 mM EDTA in PBS at 37.degree. C. for 1 h to separate dermis from epidermis before X-gal staining.
[0342] RNA-Seq Analysis.
[0343] Skin wound tissues (PW11d) of control and K14-CreER; LSLShh mice were incubated in 20 mM EDTA solution at 37.degree. C. for 30 min. After separation of epidermis from dermis, total RNA was isolated from epidermis and dermis using RNeasy Plus Micro-Kit (Qiagen) as described by the manufacturer. Total RNA was provided to Genome Technology Center at NYU Langone Medical Center for preparing RNA-seq libraries and sequencing. RNA-seq libraries were prepared using the Illumina TruSeq Stranded Total RNA library prep, after ribodepletion with Ribozero Gold kit (Illumina) starting from 200 ng of DNAse I treated total RNA, following the manufacturer's protocol (15 cycles of PCR amplification). The amplified libraries were purified using AMPure beads, quantified by Qubit and QPCR, and visualized in an Agilent Bioanalyzer. The libraries were pooled equimolarly, and sequenced on two lanes of an Illumina HiSeq 2500 flow cell, v4 chemistry as paired-end. The differentially expressed genes (DEG) were submitted to DAVID for GO term analysis. Top related enriched terms were selected and shown in the figures.
[0344] Single-Cell RNA-Seq Analysis.
[0345] Skin wounds were collected from of both SM22-rtTA; tetO-Cre; R26-SmoM2/Tomato (SM22-SmoM2) and SM22-rtTA; tetO-Cre; R26-Tomato (control) mice 3 days after complete re-epithelialization (dox treatment from PW1d to PW12d) and incubated in 20 mM EDTA solution at 37.degree. C. for 30 min to separate dermis from epidermis. The separated dermis was incubated in Dulbecco's Modified Eagle Medium (DMEM, Corning) containing 10% FBS (Corning) and 0.35% type I collagenase (Worthington) at 37.degree. C. for 1 h. After rinsing with PBS, Tomato+ cells from isolated dermal cells were sorted with a FACSAria II cell sorter (BD biosciences). scRNA-seq libraries were prepared using the following: Single-Cell 3' Reagent Kits v2: Chromium.TM. Single-Cell 3' Library & Gel Bead Kit v2 PN-120237, Single-Cell 3' Chip Kit v2 PN-120236, i7 Multiplex Kit PN-120262'' (10.times. Genomics) and the Single-Cell 3' Reagent Kits v2 User Guide (Manual Part # CG00052 Rev A). Libraries were run on an Illumina HiSeq 4000 as 2.times.150 paired-end reads, one full lane per sample, for approximately >90% sequencing saturation. Sequencing results were demultiplexed and converted to FASTQ format using Illumina bcl2fastq software. The Cell Ranger Single-Cell Software Suite was used to perform sample demultiplexing, barcode processing, and single-cell 3' gene counting. The cDNA insert was aligned to the mm10/GRCm38 reference genome. Only confidently mapped, non-PCR duplicates with valid barcodes and UMIs were used to generate the genebarcode matrix. Further analysis and visualization was performed using Seurat, an R package containing implementations of commonly used single-cell analytical techniques, including the identification of highly variable genes, dimensionality reduction, standard unsupervised clustering algorithms, and the discovery of differentially expressed genes and markers.
[0346] Quantitative Reverse Transcription PCR (qRT-PCR).
[0347] Total RNA was isolated using RNeasy Micro-Kit (Qiagen) as described by manufacturer and reversetranscribed with Superscript III First Strand Synthesis System (Invitrogen) for cDNA synthesis. cDNA was amplified using taqman probes and the ABI 7900HT SDS system. Transcripts were quantified relative to the housekeeping gene, GAPDH.
[0348] Hydroxyproline Assay.
[0349] To measure collagen content, hydroxyproline assay kit (Sigma) was used according to manufacturer's protocol. Whole wound tissues were collected and homogenized in 100 .mu.l of water per 10 mg tissue. After adding 100 .mu.l of HCl (.about.12 M) per 10 mg tissue into the homogenized tissue, the mixture was hydrolyzed at 120.degree. C. for 3 h. A total of 1-2 .mu.l of supernatant was incubated in 100 .mu.l of Chloramine T/Oxidation buffer mixture for 5 min and then, in 100 .mu.l of diluted DMAB reagent for 90 min, sequentially. The absorbance was measured at 560 nm using SpectraMax M3 (Molecular Devices).
[0350] Transmission Electron Microscopy.
[0351] Transmission electron microscopy (TEM) was carried out in Microscopy Laboratory at NYU Langone Medical Center. The harvested wounds were dissected (0.5.times.1 cm) and put the wounds on top of paper tower. The skin wounds were fixed in the fixative containing 2.5% glutaraldehyde, and 2% paraformaldehyde in 0.1M sodium cacodylate buffer (pH 7.2) with 1% tannic acid for 30 min and further dissected to 1.times.3 mm smaller pieces. Fixation process was continued in the same fixative at RT for 2 h, then 4.degree. C. overnight. The skin then post-fixed with 1% osmium tetroxide for 2 h at RT, block staining in 1% uranyl acetate overnight at 4.degree. C., then dehydration in a standard manner and embedded in EMbed 812 (Electron Microscopy Sciences, Hatfield, Pa.) for transmission electron microscopy. Semi-thin sections were cut at 1 mm and stained with 1% Toluidine Blue to evaluate the orientation of the sample. Ultrathin sections (60 nm) were cut, mounted on copper grids and stained with uranyl acetate and lead citrate. Stained grids were examined under Philips cm-12 electron microscope (FEI; Eindhoven, The Netherlands) and photographed with a Gatan (4k X2.7 k) digital camera (Gatan, Inc., Pleasanton, Calif.).
[0352] Statistical Analysis and Image Processing.
[0353] The whole-mount HFN assay was performed with at least three wound samples per genotype, and the data were representative of over three independent experiments. Data were represented as mean.+-.s.d. To calculate p-values, Student's t-test was used on Microsoft Excel, with two-tailed tests and unequal variance. All graphs were generated by Microsoft Excel and GraphPad Prism. Images were processed using Image J and Adobe Photoshop. For transforming AP signals into colored dots using Photoshop, images of AP staining of wound were first turned into black and white, reduced background, and changed into red, green or cyan colors. Three images were then merged by overlapping the center of each wound.
[0354] Data Availability.
[0355] RNA-seq data and scRNA-seq have been deposited in the Gene Expression Omnibus (GEO) database under accession codes GSE94893 and GSE112671, respectively.
Results
[0356] Shh Signaling is Essential for Wound-Induced HF Neogenesis.
[0357] In analyzing the disparate healing responses in large and small wounds in adult mice, it was found that Gli1 expression, a readout of Hh pathway activation, was localized to the center of large wounds, corresponding to regions of hair placode/germ formation. In striking contrast, it was absent from small wounds (FIG. 23a). The Gli1 signal in large wounds localized to both DP and epithelial hair germ cells, recapitulating the pattern observed in embryonic HF development.
[0358] Moreover, Shh, a major ligand of the pathway, was upregulated at the site of HFN in the epithelial compartment in large wounds but absent from either epidermal or dermal compartment in small wounds (FIG. 23b, c). Shh expression during hair follicle morphogenesis is conserved between mice and humans and vital for hair follicle development and hair cycle. The absence of Hh pathway activation in mouse small wounds can explain their failure to undergo regenerative wound healing during wound repair.
[0359] First, to understand the importance of epithelial Shh expression in large wounds, Shh was genetically deleted from epidermal cells in healed large wounds of K14-CreER; Shh fl/fl mice upon tamoxifen (TAM) induction from post wound (PW)3d to PW21d. This resulted in a loss of DP and hair germ formation compared to control mice (FIG. 23d-f). These results showed that epithelial Shh ligands are essential for DP formation and HFN. Additionally, deletion of Smo, an essential component of Shh pathway activation, in underlying wound dermal cells in TAM treated Pdgfra-CreER; Smo fl/fl mice also resulted in inhibition of DP formation and associated HFN events (FIG. 23g-j). Thus, activation of the Shh signaling pathway in the wound dermis plays a vital role in promoting DP formation.
[0360] Epithelial Shh Leads to HF Neogenesis in Wounds.
[0361] To determine if Shh activation could induce HFN in scar-forming wounds, Shh was overexpressed in epithelial cells in K14-CreER; LSL-Shh or K14-CreER; LSL-Shh; Gli1-LacZ mice and HFN was examined in small wounds. To induce Shh overexpression, TAM was injected into control and K14-CreER; LSL-Shh or K14-CreER; LSL-Shh; Gli1-LacZ mice from PW1d to indicated time points in FIG. 24. This treatment resulted in extensive HFN in wounds compared to control wounds (FIG. 24a, b). Epidermal Shh overexpression resulted in Gli1 activation in both the epidermis and dermis, corresponding to the areas of HFN. Shh-driven hair germs expressed Lef1 and K17 and exhibited normal hair follicle morphogenesis. AP+ DPs were associated with overlying K17+ epithelial buds as typically observed in HFN. Eventually, many of these hair follicles (52.+-.16%, mean.+-.s.d.) grew downward to form mature hair follicles with hair shafts, an event rarely observed in control small wounds. New DP expressed Lef1 and Noggin as well as AP, further demonstrating their DP identity. Aberrant basaloid growths resembling superficial basal-cell carcinomas (BCCs) were rarely in these wounds.
[0362] Previous studies noted that HFN in large wounds in WT mice was limited to the central area of the wounds. Given the ability of Shh to induce ectopic HFN in small wounds, exogenous Shh might overcome the inability of new hair follicles to form outside this central region in large wounds. In large wounds from TAM-treated K14-CreER; LSL-Shh mice, extensive DP formation was observed covering the entire wound area compared with TAM-treated controls. (FIG. 24c-e). These results verify the potency of Shh activation to overcome regional inhibition of HFN.
[0363] To characterize changes in gene expression following Shh overexpression, TAM was administered into control and K14-CreER; LSL-Shh mice from PW1d to PW11d and wound cells isolated for RNA-seq analyses. Epidermal and dermal cells from TAM-treated control and K14-CreER; LSL-Shh mice were compared by RNA-seq analyses (FIG. 24f, g and Table 1). Gene ontology (GO) analyses showed that processes and signatures involved in embryonic HF morphogenesis, including cell proliferation, cell adhesion, and Hh signalingl, were enriched by Shh overexpression. There was no significant difference in expression of Fgf9, which is known to promote hair follicle neogenesis in a large wound model, either in the epidermis (FDR:0.64) or dermis (FDR:0.97) of K14-CreER; LSL-Shh mice compared to controls. Most notably, there was upregulation of the Shh signaling pathway in both the epithelial cells and dermal cells. Upregulation of DP signature genes Bmp7, Enpp2, lamc3, and Trpsl in the wound dermis and hair placode signature genes, including Trp73, Vwa2, Samd5, Cxcll4, Nedd9, and Tnfaip3 were observed in the wound epidermis of Shh overexpressed mice. These data were confirmed by qPCR analyses. These results suggest that epidermal Shh overexpression can induce key embryonic signatures of HF morphogenesis.
[0364] Epithelial Shh Regenerates HFs without Altering Neighboring Collagen.
[0365] Although the increased collagen I deposition by adult dermal fibroblasts vs. fetal fibroblasts during wound healing is well-established, whether the low level of collagen I is essential for embryonic/neonatal non-scarring healing is currently unknown.
[0366] Our RNA-seq indicates that epithelial Shh overexpression did not significantly change the overall extracellular matrix composition of the wound dermis towards an embryonic state. The increased ratio of type III versus type I collagen is a key characteristic of fetal scarless wound healing, and expressions of the genes encoding these collagens were unchanged (FDR: 0.99 for Col1a1, Col1a2, and Col3a1) (Table 1). This was verified by biochemical measurement for the content of hydroxyproline, a major component of collagen (FIG. 24h). In addition, transmission electron microscopy (TEM) analysis of the wound area showed no change in collagen fiber diameter due to Shh overexpression, which is directly proportional to the tensile strength (FIG. 24i). Consistently, no significant differences were noted in the histological assessments for collagen staining (i.e., Masson trichrome and Picrosirius red staining) before and after the formation of neogenic hair follicles (FIG. 24j, k). It was found that the epithelial Shh expression and the reduction of type I collagen in the neighboring wound scar does not play a significant role in Shh-driven HF neogenesis. The data indicates that DP and hair follicles were forming within the scar of the wounds.
TABLE-US-00004 TABLE 1 Comparison of Hh pathway component and collagen expression between K14-Shh and control mice based on RNA-seq Genes Fold change FDR Dermis Hh pathway Shh 2.41 0.64 Gli1 3.20 0.16 Gli2 4.18 0.06 Ptch1 3.96 0.03 Collagen Colla1 0.95 0.99 Colla2 1.00 0.99 Col3a1 0.94 0.99 Epidermis Hh pathway Shh 51.83 2.01E-04 Gli1 4.21 1.33E-01 Gli2 7.27 8.65E-07 Ptch1 3.74 3.08E-04 Collagen Colla1 0.56 0.56 Colla2 0.82 0.87 Col3a1 0.86 0.91
[0367] Dermal Hh Activation Induces HF Neogenesis in Scarring Wounds.
[0368] To ask if direct Shh pathway activation of myofibroblasts in the small wound dermis would also promote DP formation and HFN, expression of the activated form of Smo was induced under the control of the SM22.alpha. promoter, known to be specifically active only in dermal myofibroblasts (SM22-rtTA; tetO-Cre; R26-SmoM2) (FIG. 25a). In the transgenic mice, constitutively active Smo expression is dependent on doxycycline (dox) administration.
[0369] Expression of the activated form of Smo in myofibroblasts during wounding (dox treatment from PW1d to PW30d) or following re-epithelialization (dox treatment from PW10.+-.2d to PW46d) resulted in DP formation within small wounds of SM22-SmoM2 mice compared to control mice (FIG. 25b, c). Constitutive Hh activation in wound myofibroblasts from SM22-SmoM2 mice also resulted in striking changes in wound epithelial cells. Those epithelial cells directly above Smo-active DP expressed K17 and Shh, well established hair germ markers (FIG. 25d, e, i). Additionally, these hair germ cells displayed nuclear .beta.-catenin and Lef1 expression indicating active Wnt signaling, a known signature of hair follicle development and growth (FIG. 25h, j-m). A significant number of new hair follicles were also observed with differentiated hair shafts (28.+-.7%) containing both outer root sheath (AE13+ hair cortex, AE15+ medulla), and inner root sheath (AE15+) structures, stem cell compartments (K15+CD34+), hair matrix cells (Shh+) (FIG. 25g, n). Neogenic hair follicles also maintained an adjoining dermal sheath (SMA+, SM22.alpha.+), biochemically distinct from DP (Noggin+). AP+DP structures were frequently observed without accompanying hair germs despite their close proximity to overlying epidermis (FIG. 25j). Indeed, there were almost twice as many AP+DP as K17+ hair germs, indicating that many DP (42.+-.15%) formed without establishing epithelial-mesenchymal interactions that could promote HF morphogenesis (FIG. 25f). DP formation without hair germ formation is not observed in normal endogenous hair follicle development in embryo or in adult large wounds. These regenerative events were observed following wound closure.
[0370] Intriguingly, even in the experiments where Hh activation was induced early during wound healing, the time of wound closure, proliferation, epidermal differentiation, AP distribution, angiogenesis and infiltration of immune cells including macrophages into the wound site were not altered prior to wound closure.
[0371] Hh Activation Shifts Dermal Fibroblast Fate Toward DP.
[0372] To understand whether SM22+ dermal cells universally induce the DP fate upon Hh activation, Tomato+ cells were isolated from wound dermis of SM22-rtTA; tetO-Cre; R26-SmoM2/Tomato (SM22-SmoM2) and SM22-rtTA; tetO-Cre; R26-Tomato (control) mice 3 days after complete re-epithelialization (dox treatment from PW1d to PW12d) and compared their molecular signatures by single-cell RNA sequencing (scRNA-seq) (FIG. 26). Unsupervised clustering with K means was performed based on differentially expressed genes (DEGs), using tSNE (t-distributed stochastic neighbor embedding). Based on expression of lineage markers for different cell types, cellular clusters of fibroblasts, muscle cells, schwann cells, endothelial cells and immune cells were identified (FIG. 26a-f). Examination of Hh pathway mediators Gli1, Gli2, Ptch1, and Ptch2 showed that Hh pathway components were mainly expressed in the fibroblast cluster (FIG. 26g) and were largely restricted to the SM22-SmoM2 group (FIG. 26h, i). This is consistent with the lack of Gli1-lacZ expression in the wild-type dermis of small wounds (FIG. 23a). The Hh-active fibroblast clusters showed upregulation of DP signature genes including Hey1, Sema6a, Wif1, Cxcr4, Ggta1, Hck, Snrpn and Rasd1 as previously defined by several groups (FIG. 26j), indicating that Hh activation in SM22+ myofibroblasts are sufficient to globally induce upregulation of DP signature genes. Nonetheless, within this Hh-activated fibroblast population, a divergence was identified in the number and level of DP signature genes (Hh-active I and II), indicating that Hh-independent mechanisms are also involved in the regulation of DP signature genes. For example, while the upregulation of Bmp3 and Plk2, known DP signature genes, was widely observed among Hh-active fibroblasts (Hhactive I and II), expression of Alpl (AP) and Lef1, vital markers for DP identification in histochemical analyses, was limited to a small subpopulation of Hh-active fibroblasts (Hh-active II) (FIG. 26j).
[0373] Hh Activation in Wound Epidermis Forms BCC-Like Structure.
[0374] In contrast to dermis-specific activation of the Hh pathway, forced Hh activation solely in epithelial cells (K14-CreER; R26-SmoM2, TAM administration from PW1d to PW30d) did not promote DP formation in the wound area. As previously observed, the epidermis of these wounds maintained numerous K17+ epithelial invaginations resembling BCCs without signs of hair follicle differentiation. Also, the Hh driven, BCC-like epithelial growths were not accompanied by underlying DP52.
[0375] Hh Activation Converts Wnt-Active Wound Fibroblasts into DP.
[0376] Next, given that Wnt signaling plays an essential role in hair follicle development and neogenesis, the relationship between Wnt and Shh signaling was examined. Our scRNA-seq analyses in myofibroblasts and examination of Axin2 expression in wounds from Axin2-LacZ mice both showed Wnt activity in scarring dermis of small wounds. scRNA-seq analyses demonstrated the expression of Axin2, Wls, and canonical Wnt ligands such as Wnt2 and Wnt10a (FIG. 27a, b). These findings are consistent with previous reports that long-term Wnt signaling within dermal fibroblasts correlates with injury-induced fibrosis. It was hypothesized that dermal Wnt signaling is not sufficient to induce HFN without Hh activation. Indeed, constitutive activation of Wnt signaling (SM22-rtTA; tetO-Cre; .beta.-catenin ex3/fl) in SM22+ dermal cells of small wounds did not promote new DP regeneration or HFN (FIG. 27c-h). However, dermal depletion of Wls, which is essential for Wnt ligand secretion, inhibited HFN in large wounds (SM22-rtTA; tetO-Cre; Wls fl/fl and dox treatment from PW3d to PW21d). These results indicate the requirement of dermal Wnt ligands for HFN (FIG. 27i-1). A study performed to test whether Hh activation in dermal cells was capable of converting Wnt-active dermal fibroblasts to the DP fate. Forced Hh activation was induced in Wnt responsive cells within small wounds, using the promoter of Axin2, a well established target of canonical Wnt activity (Axin2-CreER;R26-SmoM2). TAM was administered into control and Axin2-CreER; R26-SmoM2 from PW1d to PW30d. Smo overexpression in Wnt-active myofibroblasts resulted in extensive DP formation in small wounds (FIG. 28a-d). Genetic tracing of Axin2+ cells during HFN in reporter Axin2-CreER; R26-Tomato mice showed tomato expression in de novo DPs establishing that Axin2+ cells in wounds form DPs (FIG. 28e). In contrast, control wounds without Hh activation in Wnt-active dermal cells underwent scarring without HFN (FIG. 28a-d). These results show that Hh activation in Wnt-active dermal cells promotes their fate conversion into DP, the regenerative dermal niche for HF formation.
[0377] The results demonstrate that wound repair can be redirected to promote regeneration following injury by modifying a key dermal signal (FIG. 28f). This study provides definitive evidence for a longheld concept that dermal cells are key components in determining wound healing outcome. The results show that the suppression of skin appendage regeneration in wound healing is due to the absence of dermal regeneration signals rather than intrinsic lack of regenerative competence in scarring cells. Installing developmental signals in the wound dermis may be a reasonable strategy to achieve regenerative healing in mammals. Epithelial activation of Wnt and Shh signaling was previously identified as critical for hair regeneration; however, these same pathways may also induce skin epithelial cancers when experimentally or pharmacologically augmented. This basic study with preclinical mouse models shows that the capacity to create ectopic de novo DP in vivo by modulation of specific signals in the dermis may overcome this barrier and bring us closer to true skin renewal after injury.
[0378] Fibrotic scarring and epimorphic regeneration are frequently considered to be on opposite ends of the wound healing spectrum. This concept underpins attempts to promote regenerative healing by suppression of scarring mechanisms. This study shows that scarring/fibrosis in skin wounds may not affect HF morphogenesis if the appropriate regenerative ques are applied.
[0379] Although long-term activation of Wnt signaling, a hallmark of fibrotic repair, was observed in small wounds, the physiological level of Wnt signaling in scarring wounds did not negatively impact HFN in the presence of Shh activation. These studies provide evidence that fibrotic repair can be genetically subverted and offers possible tools to bypass extensive reprograming of adult skin cells into an "embryonic status" or for "stem cell transplantation" strategies to lead to regenerative healing in mammals.
[0380] HFN is largely a recapitulation of hair follicle development. However, the requirements for Shh regulation may be different in embryonic HF development and adult HFN. Shh-null mice can develop DP16 and Smo is dispensable for the initial establishment of DP in embryos. Similarly, while epithelial Wnts are sufficient for embryonic HF development, both epithelial and dermal Wnts are needed for adult HFN. The absolute requirement for Shh/Smo signaling in adult de novo DP formation may reflect a vulnerability in regeneration mechanisms compared to organogenesis in the embryo, whose development is often ensured by redundant compensatory mechanisms.
[0381] In conclusion, this study demonstrates that de novo DP can be created in adult skin by modulating a signaling pathway in the dermis.
Example 10
Effect of Shh in Multiple Hair Regeneration Assays
[0382] Shh agonist, Hh-Ag, promotes hair neogenesis in the following models: (1) wound induced hair neogenesis; (2) patch assay of cultured mouse and human dermal cells; and (3) in vitro organoid culture.
[0383] As shown in FIG. 29, C57B6, wounded at P21, followed by subcutaneous injection of Hh-Ag from PWD5 to PWD8 increases hair follicle formation.
[0384] As shown in FIG. 30, cultured human and mouse dermal cells treated with Shh agonist (i.e., Hh-Ag) showed increased hair follicle (HF) number, relative to control.
[0385] FIGS. 31 and 32 show that mouse dermal cells infected with active Smo virus at PO and P2 result in more HF in recon assay. Cultured mouse dermal cells transduced with Smo plus mouse epi. P2 cells typically lose activity as seen in control. Smo transduction maintains inductivity.
[0386] DP and DS cells in HFs are made from cultured dermal cells (RFP+) infected with activated Smo. (See FIG. 33).
[0387] FIG. 34 shows quantitation of recon assay of cultured mouse dermal cells treated with Shh Ag or infected with active Smo virus. Number of hair formed per assay was significantly higher in Shh agonist, Hh-Ag, treated cells, relative to control and Smo virus infected cells.
[0388] Foreskin dermal cells were infected with active Smo. As shown in FIG. 35, human foreskin dermal cells have no hair inducing activity but activated smo induces them to promote HF formation.
[0389] FIG. 36 shows dose dependent response. High concentration of Hh-Ag inhibited HF formation.
[0390] Mouse neonatal dermal cells were cultured from PO to P2 (See FIG. 37). Hair follicles were quantified in recon assay using cultured mouse neonatal dermal cells (See FIG. 38). FIG. 39 shows cultured dermal cells from Gli1-Lacz mouse. The percentage of Gli1-Lacz positive cells decreased in culture even in the presence of Hh-Ag (See FIG. 40). Culturing dermal cells in the presence of Hh-Ag does not maintain the Shh responding population. 24 hr Shh treatment showed fewer Gli1 positive cells but had similar number of HFs in patch assay compared to 7 day treatment.
[0391] Organoids were cultured. Dermal and epidermal cells from neonatal mice harvested. 5000:500 dermal:epidermal cells per well in 2 96 well low-attachment plates. They were cultured in GMEM with: 1.5% Serum Replacement, 1.times. Sodium Pyruvate, 1.times. MEM NEAA, lx 2-Mercaptoethanol, 1.times. Anti-Anti, 1.times. GlutaMAX, 1.times. FGF, and 2% Matrigel. Five days later, organoid development was noticed. In one plate, 10 .mu.M CHIR was added. CHIR is a GSK-3 inhibitor, preventing GSK-mediated phosphorylation and subsequent degradation of .beta.-catenin, activating the WNT-pathway. The other plate was kept under the same conditions as before.
[0392] Differentiation medium was replaced with maturation medium under 3 different conditions: nothing, SHH (10 .mu.M), CHIR (10 .mu.M)+SHH (10 .mu.M). The SHH pathway is implicated in hair follicle morphogenesis. The cells were cultured in DMEM with: lx Glut.mu.MAX, 1.times. Anti-Anti, and 1.times. N2 Supplement. The medium was changed every 2 days. The experiments included blank-blank, blank-SHH, blank-SHH--CHIR, CHIR-blank, CHIR-SHH, and CHIR-SHH-CHIR. FIGS. 41 and 42 show blank-blank best images and histology, respectively. FIGS. 43 and 44 show blank-SHH best images and histology, respectively.
[0393] Although the invention herein has been described with reference to particular embodiments, it is to be understood that these embodiments are merely illustrative of the principles and applications of the present invention. It is therefore to be understood that numerous modifications may be made to the illustrative embodiments and that other arrangements may be devised without departing from the spirit and scope of the present invention as defined by the appended claims.
Sequence CWU 1
1
91133PRTHomo sapiens 1Ile Phe Pro Asn Gly Thr Ile Gln Gly Thr Arg
Lys Asp His Ser Arg1 5 10 15Phe Gly Ile Leu Glu Phe Ile Ser Ile Ala
Val Gly Leu Val Ser Ile 20 25 30Arg Gly Val Asp Ser Gly Leu Tyr Leu
Gly Met Asn Glu Lys Gly Glu 35 40 45Leu Tyr Gly Ser Glu Lys Leu Thr
Gln Glu Cys Val Phe Arg Glu Gln 50 55 60Phe Glu Glu Asn Trp Tyr Asn
Thr Tyr Ser Ser Asn Leu Tyr Lys His65 70 75 80Val Asp Thr Gly Arg
Arg Tyr Tyr Val Ala Leu Asn Lys Asp Gly Thr 85 90 95Pro Arg Glu Gly
Thr Arg Thr Lys Arg His Gln Lys Phe Thr His Phe 100 105 110Leu Pro
Arg Pro Val Asp Pro Asp Lys Val Pro Glu Leu Tyr Lys Asp 115 120
125Ile Leu Ser Gln Ser 1302462PRTHomo sapiens 2Met Leu Leu Leu Ala
Arg Cys Leu Leu Leu Val Leu Val Ser Ser Leu1 5 10 15Leu Val Cys Ser
Gly Leu Ala Cys Gly Pro Gly Arg Gly Phe Gly Lys 20 25 30Arg Arg His
Pro Lys Lys Leu Thr Pro Leu Ala Tyr Lys Gln Phe Ile 35 40 45Pro Asn
Val Ala Glu Lys Thr Leu Gly Ala Ser Gly Arg Tyr Glu Gly 50 55 60Lys
Ile Ser Arg Asn Ser Glu Arg Phe Lys Glu Leu Thr Pro Asn Tyr65 70 75
80Asn Pro Asp Ile Ile Phe Lys Asp Glu Glu Asn Thr Gly Ala Asp Arg
85 90 95Leu Met Thr Gln Arg Cys Lys Asp Lys Leu Asn Ala Leu Ala Ile
Ser 100 105 110Val Met Asn Gln Trp Pro Gly Val Lys Leu Arg Val Thr
Glu Gly Trp 115 120 125Asp Glu Asp Gly His His Ser Glu Glu Ser Leu
His Tyr Glu Gly Arg 130 135 140Ala Val Asp Ile Thr Thr Ser Asp Arg
Asp Arg Ser Lys Tyr Gly Met145 150 155 160Leu Ala Arg Leu Ala Val
Glu Ala Gly Phe Asp Trp Val Tyr Tyr Glu 165 170 175Ser Lys Ala His
Ile His Cys Ser Val Lys Ala Glu Asn Ser Val Ala 180 185 190Ala Lys
Ser Gly Gly Cys Phe Pro Gly Ser Ala Thr Val His Leu Glu 195 200
205Gln Gly Gly Thr Lys Leu Val Lys Asp Leu Ser Pro Gly Asp Arg Val
210 215 220Leu Ala Ala Asp Asp Gln Gly Arg Leu Leu Tyr Ser Asp Phe
Leu Thr225 230 235 240Phe Leu Asp Arg Asp Asp Gly Ala Lys Lys Val
Phe Tyr Val Ile Glu 245 250 255Thr Arg Glu Pro Arg Glu Arg Leu Leu
Leu Thr Ala Ala His Leu Leu 260 265 270Phe Val Ala Pro His Asn Asp
Ser Ala Thr Gly Glu Pro Glu Ala Ser 275 280 285Ser Gly Ser Gly Pro
Pro Ser Gly Gly Ala Leu Gly Pro Arg Ala Leu 290 295 300Phe Ala Ser
Arg Val Arg Pro Gly Gln Arg Val Tyr Val Val Ala Glu305 310 315
320Arg Asp Gly Asp Arg Arg Leu Leu Pro Ala Ala Val His Ser Val Thr
325 330 335Leu Ser Glu Glu Ala Ala Gly Ala Tyr Ala Pro Leu Thr Ala
Gln Gly 340 345 350Thr Ile Leu Ile Asn Arg Val Leu Ala Ser Cys Tyr
Ala Val Ile Glu 355 360 365Glu His Ser Trp Ala His Arg Ala Phe Ala
Pro Phe Arg Leu Ala His 370 375 380Ala Leu Leu Ala Ala Leu Ala Pro
Ala Arg Thr Asp Arg Gly Gly Asp385 390 395 400Ser Gly Gly Gly Asp
Arg Gly Gly Gly Gly Gly Arg Val Ala Leu Thr 405 410 415Ala Pro Gly
Ala Ala Asp Ala Pro Gly Ala Gly Ala Thr Ala Gly Ile 420 425 430His
Trp Tyr Ser Gln Leu Leu Tyr Gln Ile Gly Thr Trp Leu Leu Asp 435 440
445Ser Glu Ala Leu His Pro Leu Gly Met Ala Val Lys Ser Ser 450 455
46031447PRTHomo sapiens 3Met Ala Ser Ala Gly Asn Ala Ala Glu Pro
Gln Asp Arg Gly Gly Gly1 5 10 15Gly Ser Gly Cys Ile Gly Ala Pro Gly
Arg Pro Ala Gly Gly Gly Arg 20 25 30Arg Arg Arg Thr Gly Gly Leu Arg
Arg Ala Ala Ala Pro Asp Arg Asp 35 40 45Tyr Leu His Arg Pro Ser Tyr
Cys Asp Ala Ala Phe Ala Leu Glu Gln 50 55 60Ile Ser Lys Gly Lys Ala
Thr Gly Arg Lys Ala Pro Leu Trp Leu Arg65 70 75 80Ala Lys Phe Gln
Arg Leu Leu Phe Lys Leu Gly Cys Tyr Ile Gln Lys 85 90 95Asn Cys Gly
Lys Phe Leu Val Val Gly Leu Leu Ile Phe Gly Ala Phe 100 105 110Ala
Val Gly Leu Lys Ala Ala Asn Leu Glu Thr Asn Val Glu Glu Leu 115 120
125Trp Val Glu Val Gly Gly Arg Val Ser Arg Glu Leu Asn Tyr Thr Arg
130 135 140Gln Lys Ile Gly Glu Glu Ala Met Phe Asn Pro Gln Leu Met
Ile Gln145 150 155 160Thr Pro Lys Glu Glu Gly Ala Asn Val Leu Thr
Thr Glu Ala Leu Leu 165 170 175Gln His Leu Asp Ser Ala Leu Gln Ala
Ser Arg Val His Val Tyr Met 180 185 190Tyr Asn Arg Gln Trp Lys Leu
Glu His Leu Cys Tyr Lys Ser Gly Glu 195 200 205Leu Ile Thr Glu Thr
Gly Tyr Met Asp Gln Ile Ile Glu Tyr Leu Tyr 210 215 220Pro Cys Leu
Ile Ile Thr Pro Leu Asp Cys Phe Trp Glu Gly Ala Lys225 230 235
240Leu Gln Ser Gly Thr Ala Tyr Leu Leu Gly Lys Pro Pro Leu Arg Trp
245 250 255Thr Asn Phe Asp Pro Leu Glu Phe Leu Glu Glu Leu Lys Lys
Ile Asn 260 265 270Tyr Gln Val Asp Ser Trp Glu Glu Met Leu Asn Lys
Ala Glu Val Gly 275 280 285His Gly Tyr Met Asp Arg Pro Cys Leu Asn
Pro Ala Asp Pro Asp Cys 290 295 300Pro Ala Thr Ala Pro Asn Lys Asn
Ser Thr Lys Pro Leu Asp Met Ala305 310 315 320Leu Val Leu Asn Gly
Gly Cys His Gly Leu Ser Arg Lys Tyr Met His 325 330 335Trp Gln Glu
Glu Leu Ile Val Gly Gly Thr Val Lys Asn Ser Thr Gly 340 345 350Lys
Leu Val Ser Ala His Ala Leu Gln Thr Met Phe Gln Leu Met Thr 355 360
365Pro Lys Gln Met Tyr Glu His Phe Lys Gly Tyr Glu Tyr Val Ser His
370 375 380Ile Asn Trp Asn Glu Asp Lys Ala Ala Ala Ile Leu Glu Ala
Trp Gln385 390 395 400Arg Thr Tyr Val Glu Val Val His Gln Ser Val
Ala Gln Asn Ser Thr 405 410 415Gln Lys Val Leu Ser Phe Thr Thr Thr
Thr Leu Asp Asp Ile Leu Lys 420 425 430Ser Phe Ser Asp Val Ser Val
Ile Arg Val Ala Ser Gly Tyr Leu Leu 435 440 445Met Leu Ala Tyr Ala
Cys Leu Thr Met Leu Arg Trp Asp Cys Ser Lys 450 455 460Ser Gln Gly
Ala Val Gly Leu Ala Gly Val Leu Leu Val Ala Leu Ser465 470 475
480Val Ala Ala Gly Leu Gly Leu Cys Ser Leu Ile Gly Ile Ser Phe Asn
485 490 495Ala Ala Thr Thr Gln Val Leu Pro Phe Leu Ala Leu Gly Val
Gly Val 500 505 510Asp Asp Val Phe Leu Leu Ala His Ala Phe Ser Glu
Thr Gly Gln Asn 515 520 525Lys Arg Ile Pro Phe Glu Asp Arg Thr Gly
Glu Cys Leu Lys Arg Thr 530 535 540Gly Ala Ser Val Ala Leu Thr Ser
Ile Ser Asn Val Thr Ala Phe Phe545 550 555 560Met Ala Ala Leu Ile
Pro Ile Pro Ala Leu Arg Ala Phe Ser Leu Gln 565 570 575Ala Ala Val
Val Val Val Phe Asn Phe Ala Met Val Leu Leu Ile Phe 580 585 590Pro
Ala Ile Leu Ser Met Asp Leu Tyr Arg Arg Glu Asp Arg Arg Leu 595 600
605Asp Ile Phe Cys Cys Phe Thr Ser Pro Cys Val Ser Arg Val Ile Gln
610 615 620Val Glu Pro Gln Ala Tyr Thr Asp Thr His Asp Asn Thr Arg
Tyr Ser625 630 635 640Pro Pro Pro Pro Tyr Ser Ser His Ser Phe Ala
His Glu Thr Gln Ile 645 650 655Thr Met Gln Ser Thr Val Gln Leu Arg
Thr Glu Tyr Asp Pro His Thr 660 665 670His Val Tyr Tyr Thr Thr Ala
Glu Pro Arg Ser Glu Ile Ser Val Gln 675 680 685Pro Val Thr Val Thr
Gln Asp Thr Leu Ser Cys Gln Ser Pro Glu Ser 690 695 700Thr Ser Ser
Thr Arg Asp Leu Leu Ser Gln Phe Ser Asp Ser Ser Leu705 710 715
720His Cys Leu Glu Pro Pro Cys Thr Lys Trp Thr Leu Ser Ser Phe Ala
725 730 735Glu Lys His Tyr Ala Pro Phe Leu Leu Lys Pro Lys Ala Lys
Val Val 740 745 750Val Ile Phe Leu Phe Leu Gly Leu Leu Gly Val Ser
Leu Tyr Gly Thr 755 760 765Thr Arg Val Arg Asp Gly Leu Asp Leu Thr
Asp Ile Val Pro Arg Glu 770 775 780Thr Arg Glu Tyr Asp Phe Ile Ala
Ala Gln Phe Lys Tyr Phe Ser Phe785 790 795 800Tyr Asn Met Tyr Ile
Val Thr Gln Lys Ala Asp Tyr Pro Asn Ile Gln 805 810 815His Leu Leu
Tyr Asp Leu His Arg Ser Phe Ser Asn Val Lys Tyr Val 820 825 830Met
Leu Glu Glu Asn Lys Gln Leu Pro Lys Met Trp Leu His Tyr Phe 835 840
845Arg Asp Trp Leu Gln Gly Leu Gln Asp Ala Phe Asp Ser Asp Trp Glu
850 855 860Thr Gly Lys Ile Met Pro Asn Asn Tyr Lys Asn Gly Ser Asp
Asp Gly865 870 875 880Val Leu Ala Tyr Lys Leu Leu Val Gln Thr Gly
Ser Arg Asp Lys Pro 885 890 895Ile Asp Ile Ser Gln Leu Thr Lys Gln
Arg Leu Val Asp Ala Asp Gly 900 905 910Ile Ile Asn Pro Ser Ala Phe
Tyr Ile Tyr Leu Thr Ala Trp Val Ser 915 920 925Asn Asp Pro Val Ala
Tyr Ala Ala Ser Gln Ala Asn Ile Arg Pro His 930 935 940Arg Pro Glu
Trp Val His Asp Lys Ala Asp Tyr Met Pro Glu Thr Arg945 950 955
960Leu Arg Ile Pro Ala Ala Glu Pro Ile Glu Tyr Ala Gln Phe Pro Phe
965 970 975Tyr Leu Asn Gly Leu Arg Asp Thr Ser Asp Phe Val Glu Ala
Ile Glu 980 985 990Lys Val Arg Thr Ile Cys Ser Asn Tyr Thr Ser Leu
Gly Leu Ser Ser 995 1000 1005Tyr Pro Asn Gly Tyr Pro Phe Leu Phe
Trp Glu Gln Tyr Ile Gly 1010 1015 1020Leu Arg His Trp Leu Leu Leu
Phe Ile Ser Val Val Leu Ala Cys 1025 1030 1035Thr Phe Leu Val Cys
Ala Val Phe Leu Leu Asn Pro Trp Thr Ala 1040 1045 1050Gly Ile Ile
Val Met Val Leu Ala Leu Met Thr Val Glu Leu Phe 1055 1060 1065Gly
Met Met Gly Leu Ile Gly Ile Lys Leu Ser Ala Val Pro Val 1070 1075
1080Val Ile Leu Ile Ala Ser Val Gly Ile Gly Val Glu Phe Thr Val
1085 1090 1095His Val Ala Leu Ala Phe Leu Thr Ala Ile Gly Asp Lys
Asn Arg 1100 1105 1110Arg Ala Val Leu Ala Leu Glu His Met Phe Ala
Pro Val Leu Asp 1115 1120 1125Gly Ala Val Ser Thr Leu Leu Gly Val
Leu Met Leu Ala Gly Ser 1130 1135 1140Glu Phe Asp Phe Ile Val Arg
Tyr Phe Phe Ala Val Leu Ala Ile 1145 1150 1155Leu Thr Ile Leu Gly
Val Leu Asn Gly Leu Val Leu Leu Pro Val 1160 1165 1170Leu Leu Ser
Phe Phe Gly Pro Tyr Pro Glu Val Ser Pro Ala Asn 1175 1180 1185Gly
Leu Asn Arg Leu Pro Thr Pro Ser Pro Glu Pro Pro Pro Ser 1190 1195
1200Val Val Arg Phe Ala Met Pro Pro Gly His Thr His Ser Gly Ser
1205 1210 1215Asp Ser Ser Asp Ser Glu Tyr Ser Ser Gln Thr Thr Val
Ser Gly 1220 1225 1230Leu Ser Glu Glu Leu Arg His Tyr Glu Ala Gln
Gln Gly Ala Gly 1235 1240 1245Gly Pro Ala His Gln Val Ile Val Glu
Ala Thr Glu Asn Pro Val 1250 1255 1260Phe Ala His Ser Thr Val Val
His Pro Glu Ser Arg His His Pro 1265 1270 1275Pro Ser Asn Pro Arg
Gln Gln Pro His Leu Asp Ser Gly Ser Leu 1280 1285 1290Pro Pro Gly
Arg Gln Gly Gln Gln Pro Arg Arg Asp Pro Pro Arg 1295 1300 1305Glu
Gly Leu Trp Pro Pro Pro Tyr Arg Pro Arg Arg Asp Ala Phe 1310 1315
1320Glu Ile Ser Thr Glu Gly His Ser Gly Pro Ser Asn Arg Ala Arg
1325 1330 1335Trp Gly Pro Arg Gly Ala Arg Ser His Asn Pro Arg Asn
Pro Ala 1340 1345 1350Ser Thr Ala Met Gly Ser Ser Val Pro Gly Tyr
Cys Gln Pro Ile 1355 1360 1365Thr Thr Val Thr Ala Ser Ala Ser Val
Thr Val Ala Val His Pro 1370 1375 1380Pro Pro Val Pro Gly Pro Gly
Arg Asn Pro Arg Gly Gly Leu Cys 1385 1390 1395Pro Gly Tyr Pro Glu
Thr Asp His Gly Leu Phe Glu Asp Pro His 1400 1405 1410Val Pro Phe
His Val Arg Cys Glu Arg Arg Asp Ser Lys Val Glu 1415 1420 1425Val
Ile Glu Leu Gln Asp Val Glu Cys Glu Glu Arg Pro Arg Gly 1430 1435
1440Ser Ser Ser Asn 1445487PRTHomo sapiens 4Phe Asp Phe Ile Val Arg
Tyr Phe Phe Ala Ala Leu Thr Val Leu Thr1 5 10 15Leu Leu Gly Leu Leu
His Gly Leu Val Leu Leu Pro Val Leu Leu Ser 20 25 30Ile Leu Gly Pro
Pro Pro Glu Val Ile Gln Met Tyr Lys Glu Ser Pro 35 40 45Glu Ile Leu
Ser Pro Pro Ala Pro Gln Gly Gly Gly Leu Arg Val Gly 50 55 60Ser Leu
Gln Val Asn Ile Ser Tyr Trp Lys Glu Leu Leu Trp Cys Gln65 70 75
80Asp Leu Arg Pro Glu Glu Ile 8551106PRTHomo sapiens 5Met Phe Asn
Ser Met Thr Pro Pro Pro Ile Ser Ser Tyr Gly Glu Pro1 5 10 15Cys Cys
Leu Arg Pro Leu Pro Ser Gln Gly Ala Pro Ser Val Gly Thr 20 25 30Glu
Gly Leu Ser Gly Pro Pro Phe Cys His Gln Ala Asn Leu Met Ser 35 40
45Gly Pro His Ser Tyr Gly Pro Ala Arg Glu Thr Asn Ser Cys Thr Glu
50 55 60Gly Pro Leu Phe Ser Ser Pro Arg Ser Ala Val Lys Leu Thr Lys
Lys65 70 75 80Arg Ala Leu Ser Ile Ser Pro Leu Ser Asp Ala Ser Leu
Asp Leu Gln 85 90 95Thr Val Ile Arg Thr Ser Pro Ser Ser Leu Val Ala
Phe Ile Asn Ser 100 105 110Arg Cys Thr Ser Pro Gly Gly Ser Tyr Gly
His Leu Ser Ile Gly Thr 115 120 125Met Ser Pro Ser Leu Gly Phe Pro
Ala Gln Met Asn His Gln Lys Gly 130 135 140Pro Ser Pro Ser Phe Gly
Val Gln Pro Cys Gly Pro His Asp Ser Ala145 150 155 160Arg Gly Gly
Met Ile Pro His Pro Gln Ser Arg Gly Pro Phe Pro Thr 165 170 175Cys
Gln Leu Lys Ser Glu Leu Asp Met Leu Val Gly Lys Cys Arg Glu 180 185
190Glu Pro Leu Glu Gly Asp Met Ser Ser Pro Asn Ser Thr Gly Ile Gln
195 200 205Asp Pro Leu Leu Gly Met Leu Asp Gly Arg Glu Asp Leu Glu
Arg Glu 210 215 220Glu Lys Arg Glu Pro Glu Ser Val Tyr Glu Thr Asp
Cys Arg Trp Asp225 230 235 240Gly Cys Ser Gln Glu Phe Asp Ser Gln
Glu Gln Leu Val His His Ile 245 250 255Asn Ser Glu His Ile His Gly
Glu Arg Lys Glu Phe Val Cys His Trp 260 265 270Gly Gly Cys Ser Arg
Glu Leu Arg Pro Phe Lys Ala Gln Tyr Met Leu 275 280 285Val Val His
Met Arg Arg His Thr Gly Glu Lys Pro His Lys Cys Thr 290 295 300Phe
Glu Gly Cys Arg Lys Ser Tyr Ser Arg Leu Glu Asn Leu Lys Thr305 310
315 320His Leu Arg Ser His Thr Gly Glu Lys Pro Tyr Met Cys Glu His
Glu
325 330 335Gly Cys Ser Lys Ala Phe Ser Asn Ala Ser Asp Arg Ala Lys
His Gln 340 345 350Asn Arg Thr His Ser Asn Glu Lys Pro Tyr Val Cys
Lys Leu Pro Gly 355 360 365Cys Thr Lys Arg Tyr Thr Asp Pro Ser Ser
Leu Arg Lys His Val Lys 370 375 380Thr Val His Gly Pro Asp Ala His
Val Thr Lys Arg His Arg Gly Asp385 390 395 400Gly Pro Leu Pro Arg
Ala Pro Ser Ile Ser Thr Val Glu Pro Lys Arg 405 410 415Glu Arg Glu
Gly Gly Pro Ile Arg Glu Glu Ser Arg Leu Thr Val Pro 420 425 430Glu
Gly Ala Met Lys Pro Gln Pro Ser Pro Gly Ala Gln Ser Ser Cys 435 440
445Ser Ser Asp His Ser Pro Ala Gly Ser Ala Ala Asn Thr Asp Ser Gly
450 455 460Val Glu Met Thr Gly Asn Ala Gly Gly Ser Thr Glu Asp Leu
Ser Ser465 470 475 480Leu Asp Glu Gly Pro Cys Ile Ala Gly Thr Gly
Leu Ser Thr Leu Arg 485 490 495Arg Leu Glu Asn Leu Arg Leu Asp Gln
Leu His Gln Leu Arg Pro Ile 500 505 510Gly Thr Arg Gly Leu Lys Leu
Pro Ser Leu Ser His Thr Gly Thr Thr 515 520 525Val Ser Arg Arg Val
Gly Pro Pro Val Ser Leu Glu Arg Arg Ser Ser 530 535 540Ser Ser Ser
Ser Ile Ser Ser Ala Tyr Thr Val Ser Arg Arg Ser Ser545 550 555
560Leu Ala Ser Pro Phe Pro Pro Gly Ser Pro Pro Glu Asn Gly Ala Ser
565 570 575Ser Leu Pro Gly Leu Met Pro Ala Gln His Tyr Leu Leu Arg
Ala Arg 580 585 590Tyr Ala Ser Ala Arg Gly Gly Gly Thr Ser Pro Thr
Ala Ala Ser Ser 595 600 605Leu Asp Arg Ile Gly Gly Leu Pro Met Pro
Pro Trp Arg Ser Arg Ala 610 615 620Glu Tyr Pro Gly Tyr Asn Pro Asn
Ala Gly Val Thr Arg Arg Ala Ser625 630 635 640Asp Pro Ala Gln Ala
Ala Asp Arg Pro Ala Pro Ala Arg Val Gln Arg 645 650 655Phe Lys Ser
Leu Gly Cys Val His Thr Pro Pro Thr Val Ala Gly Gly 660 665 670Gly
Gln Asn Phe Asp Pro Tyr Leu Pro Thr Ser Val Tyr Ser Pro Gln 675 680
685Pro Pro Ser Ile Thr Glu Asn Ala Ala Met Asp Ala Arg Gly Leu Gln
690 695 700Glu Glu Pro Glu Val Gly Thr Ser Met Val Gly Ser Gly Leu
Asn Pro705 710 715 720Tyr Met Asp Phe Pro Pro Thr Asp Thr Leu Gly
Tyr Gly Gly Pro Glu 725 730 735Gly Ala Ala Ala Glu Pro Tyr Gly Ala
Arg Gly Pro Gly Ser Leu Pro 740 745 750Leu Gly Pro Gly Pro Pro Thr
Asn Tyr Gly Pro Asn Pro Cys Pro Gln 755 760 765Gln Ala Ser Tyr Pro
Asp Pro Thr Gln Glu Thr Trp Gly Glu Phe Pro 770 775 780Ser His Ser
Gly Leu Tyr Pro Gly Pro Lys Ala Leu Gly Gly Thr Tyr785 790 795
800Ser Gln Cys Pro Arg Leu Glu His Tyr Gly Gln Val Gln Val Lys Pro
805 810 815Glu Gln Gly Cys Pro Val Gly Ser Asp Ser Thr Gly Leu Ala
Pro Cys 820 825 830Leu Asn Ala His Pro Ser Glu Gly Pro Pro His Pro
Gln Pro Leu Phe 835 840 845Ser His Tyr Pro Gln Pro Ser Pro Pro Gln
Tyr Leu Gln Ser Gly Pro 850 855 860Tyr Thr Gln Pro Pro Pro Asp Tyr
Leu Pro Ser Glu Pro Arg Pro Cys865 870 875 880Leu Asp Phe Asp Ser
Pro Thr His Ser Thr Gly Gln Leu Lys Ala Gln 885 890 895Leu Val Cys
Asn Tyr Val Gln Ser Gln Gln Glu Leu Leu Trp Glu Gly 900 905 910Gly
Gly Arg Glu Asp Ala Pro Ala Gln Glu Pro Ser Tyr Gln Ser Pro 915 920
925Lys Phe Leu Gly Gly Ser Gln Val Ser Pro Ser Arg Ala Lys Ala Pro
930 935 940Val Asn Thr Tyr Gly Pro Gly Phe Gly Pro Asn Leu Pro Asn
His Lys945 950 955 960Ser Gly Ser Tyr Pro Thr Pro Ser Pro Cys His
Glu Asn Phe Val Val 965 970 975Gly Ala Asn Arg Ala Ser His Arg Ala
Ala Ala Pro Pro Arg Leu Leu 980 985 990Pro Pro Leu Pro Thr Cys Tyr
Gly Pro Leu Lys Val Gly Gly Thr Asn 995 1000 1005Pro Ser Cys Gly
His Pro Glu Val Gly Arg Leu Gly Gly Gly Pro 1010 1015 1020Ala Leu
Tyr Pro Pro Pro Glu Gly Gln Val Cys Asn Pro Leu Asp 1025 1030
1035Ser Leu Asp Leu Asp Asn Thr Gln Leu Asp Phe Val Ala Ile Leu
1040 1045 1050Asp Glu Pro Gln Gly Leu Ser Pro Pro Pro Ser His Asp
Gln Arg 1055 1060 1065Gly Ser Ser Gly His Thr Pro Pro Pro Ser Gly
Pro Pro Asn Met 1070 1075 1080Ala Val Gly Asn Met Ser Val Leu Leu
Arg Ser Leu Pro Gly Glu 1085 1090 1095Thr Glu Phe Leu Asn Ser Ser
Ala 1100 110561258PRTHomo sapiens 6Met Ala Leu Thr Ser Ile Asn Ala
Thr Pro Thr Gln Leu Ser Ser Ser1 5 10 15Ser Asn Cys Leu Ser Asp Thr
Asn Gln Asn Lys Gln Ser Ser Glu Ser 20 25 30Ala Val Ser Ser Thr Val
Asn Pro Val Ala Ile His Lys Arg Ser Lys 35 40 45Val Lys Thr Glu Pro
Glu Gly Leu Arg Pro Ala Ser Pro Leu Ala Leu 50 55 60Thr Gln Gly Gln
Val Leu Asp Thr Ala His Val Gly Val Pro Phe Pro65 70 75 80Ser Pro
Gln Glu Gln Leu Ala Asp Leu Lys Glu Asp Leu Asp Arg Asp 85 90 95Asp
Cys Lys Gln Glu Ala Glu Val Val Ile Tyr Glu Thr Asn Cys His 100 105
110Trp Glu Asp Cys Thr Lys Glu Tyr Asp Thr Gln Glu Gln Leu Val His
115 120 125His Ile Asn Asn Glu His Ile His Gly Glu Lys Lys Glu Phe
Val Cys 130 135 140Arg Trp Gln Ala Cys Thr Arg Glu Gln Lys Pro Phe
Lys Ala Gln Tyr145 150 155 160Met Leu Val Val His Met Arg Arg His
Thr Gly Glu Lys Pro His Lys 165 170 175Cys Thr Phe Glu Gly Cys Ser
Lys Ala Tyr Ser Arg Leu Glu Asn Leu 180 185 190Lys Thr His Leu Arg
Ser His Thr Gly Glu Lys Pro Tyr Val Cys Glu 195 200 205His Glu Gly
Cys Asn Lys Ala Phe Ser Asn Ala Ser Asp Arg Ala Lys 210 215 220His
Gln Asn Arg Thr His Ser Asn Glu Lys Pro Tyr Ile Cys Lys Ile225 230
235 240Pro Gly Cys Thr Lys Arg Tyr Thr Asp Pro Ser Ser Leu Arg Lys
His 245 250 255Val Lys Thr Val His Gly Pro Asp Ala His Val Thr Lys
Lys Gln Arg 260 265 270Asn Asp Val His Leu Arg Thr Pro Leu Leu Lys
Glu Asn Gly Asp Ser 275 280 285Glu Ala Gly Thr Glu Pro Gly Gly Pro
Glu Ser Thr Glu Ala Ser Ser 290 295 300Thr Ser Gln Ala Val Glu Asp
Cys Leu His Val Arg Ala Ile Lys Thr305 310 315 320Glu Ser Ser Gly
Leu Cys Gln Ser Ser Pro Gly Ala Gln Ser Ser Cys 325 330 335Ser Ser
Glu Pro Ser Pro Leu Gly Ser Ala Pro Asn Asn Asp Ser Gly 340 345
350Val Glu Met Pro Gly Thr Gly Pro Gly Ser Leu Gly Asp Leu Thr Ala
355 360 365Leu Asp Asp Thr Pro Pro Gly Ala Asp Thr Ser Ala Leu Ala
Ala Pro 370 375 380Ser Ala Gly Gly Leu Gln Leu Arg Lys His Met Thr
Thr Met His Arg385 390 395 400Phe Glu Gln Leu Lys Lys Glu Lys Leu
Lys Ser Leu Lys Asp Ser Cys 405 410 415Ser Trp Ala Gly Pro Thr Pro
His Thr Arg Asn Thr Lys Leu Pro Pro 420 425 430Leu Pro Gly Ser Gly
Ser Ile Leu Glu Asn Phe Ser Gly Ser Gly Gly 435 440 445Gly Gly Pro
Ala Gly Leu Leu Pro Asn Pro Arg Leu Ser Glu Leu Ser 450 455 460Ala
Ser Glu Val Thr Met Leu Ser Gln Leu Gln Glu Arg Arg Asp Ser465 470
475 480Ser Thr Ser Thr Val Ser Ser Ala Tyr Thr Val Ser Arg Arg Ser
Ser 485 490 495Gly Ile Ser Pro Tyr Phe Ser Ser Arg Arg Ser Ser Glu
Ala Ser Pro 500 505 510Leu Gly Ala Gly Arg Pro His Asn Ala Ser Ser
Ala Asp Ser Tyr Asp 515 520 525Pro Ile Ser Thr Asp Ala Ser Arg Arg
Ser Ser Glu Ala Ser Gln Cys 530 535 540Ser Gly Gly Ser Gly Leu Leu
Asn Leu Thr Pro Ala Gln Gln Tyr Ser545 550 555 560Leu Arg Ala Lys
Tyr Ala Ala Ala Thr Gly Gly Pro Pro Pro Thr Pro 565 570 575Leu Pro
Gly Leu Glu Arg Met Ser Leu Arg Thr Arg Leu Ala Leu Leu 580 585
590Asp Ala Ala Glu Gly Thr Leu Pro Ala Gly Cys Pro Arg Pro Leu Gly
595 600 605Pro Arg Arg Gly Ser Asp Gly Pro Thr Tyr Gly His Gly His
Ala Gly 610 615 620Ala Ala Pro Ala Phe Pro His Glu Ala Pro Gly Gly
Gly Thr Arg Arg625 630 635 640Ala Ser Asp Pro Val Arg Arg Pro Asp
Ala Leu Ser Leu Pro Arg Val 645 650 655Gln Arg Phe His Ser Thr His
Asn Val Asn Pro Gly Pro Leu Pro Pro 660 665 670Cys Ala Asp Arg Arg
Gly Leu Arg Leu Gln Ser His Pro Ser Thr Asp 675 680 685Gly Gly Leu
Ala Arg Gly Ala Tyr Ser Pro Arg Pro Pro Ser Ile Ser 690 695 700Glu
Asn Val Ala Met Glu Ala Val Ala Ala Gly Val Asp Gly Ala Gly705 710
715 720Pro Glu Ala Asp Leu Gly Leu Pro Glu Asp Asp Leu Val Leu Pro
Asp 725 730 735Asp Val Val Gln Tyr Ile Lys Ala His Ala Ser Gly Ala
Leu Asp Glu 740 745 750Gly Thr Gly Gln Val Tyr Pro Thr Glu Ser Thr
Gly Phe Ser Asp Asn 755 760 765Pro Arg Leu Pro Ser Pro Gly Leu His
Gly Gln Arg Arg Met Val Ala 770 775 780Ala Asp Ser Asn Val Gly Pro
Ser Ala Pro Met Leu Gly Gly Cys Gln785 790 795 800Leu Gly Phe Gly
Ala Pro Ser Ser Leu Asn Lys Asn Asn Met Pro Val 805 810 815Gln Trp
Asn Glu Val Ser Ser Gly Thr Val Asp Ser Leu Ala Ser Gln 820 825
830Val Lys Pro Pro Pro Phe Pro Gln Gly Asn Leu Ala Val Val Gln Gln
835 840 845Lys Pro Ala Phe Gly Gln Tyr Pro Gly Tyr Ser Pro Gln Gly
Leu Gln 850 855 860Ala Ser Pro Gly Gly Leu Asp Ser Thr Gln Pro His
Leu Gln Pro Arg865 870 875 880Ser Gly Ala Pro Ser Gln Gly Ile Pro
Arg Val Asn Tyr Met Gln Gln 885 890 895Leu Arg Gln Pro Val Ala Gly
Ser Gln Cys Pro Gly Met Thr Thr Thr 900 905 910Met Ser Pro His Ala
Cys Tyr Gly Gln Val His Pro Gln Leu Ser Pro 915 920 925Ser Thr Ile
Ser Gly Ala Leu Asn Gln Phe Pro Gln Ser Cys Ser Asn 930 935 940Met
Pro Ala Lys Pro Gly His Leu Gly His Pro Gln Gln Thr Glu Val945 950
955 960Ala Pro Asp Pro Thr Thr Met Gly Asn Arg His Arg Glu Leu Gly
Val 965 970 975Pro Asn Ser Ala Leu Ala Gly Val Pro Pro Pro His Pro
Val Gln Ser 980 985 990Tyr Pro Gln Gln Ser His His Leu Ala Ala Ser
Met Ser Gln Glu Gly 995 1000 1005Tyr His Gln Val Pro Ser Leu Leu
Pro Ala Arg Gln Pro Gly Phe 1010 1015 1020Met Glu Pro Gln Thr Gly
Pro Met Gly Val Ala Thr Ala Gly Phe 1025 1030 1035Gly Leu Val Gln
Pro Arg Pro Pro Leu Glu Pro Ser Pro Thr Gly 1040 1045 1050Arg His
Arg Gly Val Arg Ala Val Gln Gln Gln Leu Ala Tyr Ala 1055 1060
1065Arg Ala Thr Gly His Ala Met Ala Ala Met Pro Ser Ser Gln Glu
1070 1075 1080Thr Ala Glu Ala Val Pro Lys Gly Ala Met Gly Asn Met
Gly Ser 1085 1090 1095Val Pro Pro Gln Pro Pro Pro Gln Asp Ala Gly
Gly Ala Pro Asp 1100 1105 1110His Ser Met Leu Tyr Tyr Tyr Gly Gln
Ile His Met Tyr Glu Gln 1115 1120 1125Asp Gly Gly Leu Glu Asn Leu
Gly Ser Cys Gln Val Met Arg Ser 1130 1135 1140Gln Pro Pro Gln Pro
Gln Ala Cys Gln Asp Ser Ile Gln Pro Gln 1145 1150 1155Pro Leu Pro
Ser Pro Gly Val Asn Gln Val Ser Ser Thr Val Asp 1160 1165 1170Ser
Gln Leu Leu Glu Ala Pro Gln Ile Asp Phe Asp Ala Ile Met 1175 1180
1185Asp Asp Gly Asp His Ser Ser Leu Phe Ser Gly Ala Leu Ser Pro
1190 1195 1200Ser Leu Leu His Ser Leu Ser Gln Asn Ser Ser Arg Leu
Thr Thr 1205 1210 1215Pro Arg Asn Ser Leu Thr Leu Pro Ser Ile Pro
Ala Gly Ile Ser 1220 1225 1230Asn Met Ala Val Gly Asp Met Ser Ser
Met Leu Thr Ser Leu Ala 1235 1240 1245Glu Glu Ser Lys Phe Leu Asn
Met Met Thr 1250 12557349PRTHomo sapiens 7Met Asn Arg Lys Ala Arg
Arg Cys Leu Gly His Leu Phe Leu Ser Leu1 5 10 15Gly Met Val Tyr Leu
Arg Ile Gly Gly Phe Ser Ser Val Val Ala Leu 20 25 30Gly Ala Ser Ile
Ile Cys Asn Lys Ile Pro Gly Leu Ala Pro Arg Gln 35 40 45Arg Ala Ile
Cys Gln Ser Arg Pro Asp Ala Ile Ile Val Ile Gly Glu 50 55 60Gly Ser
Gln Met Gly Leu Asp Glu Cys Gln Phe Gln Phe Arg Asn Gly65 70 75
80Arg Trp Asn Cys Ser Ala Leu Gly Glu Arg Thr Val Phe Gly Lys Glu
85 90 95Leu Lys Val Gly Ser Arg Glu Ala Ala Phe Thr Tyr Ala Ile Ile
Ala 100 105 110Ala Gly Val Ala His Ala Ile Thr Ala Ala Cys Thr Gln
Gly Asn Leu 115 120 125Ser Asp Cys Gly Cys Asp Lys Glu Lys Gln Gly
Gln Tyr His Arg Asp 130 135 140Glu Gly Trp Lys Trp Gly Gly Cys Ser
Ala Asp Ile Arg Tyr Gly Ile145 150 155 160Gly Phe Ala Lys Val Phe
Val Asp Ala Arg Glu Ile Lys Gln Asn Ala 165 170 175Arg Thr Leu Met
Asn Leu His Asn Asn Glu Ala Gly Arg Lys Ile Leu 180 185 190Glu Glu
Asn Met Lys Leu Glu Cys Lys Cys His Gly Val Ser Gly Ser 195 200
205Cys Thr Thr Lys Thr Cys Trp Thr Thr Leu Pro Gln Phe Arg Glu Leu
210 215 220Gly Tyr Val Leu Lys Asp Lys Tyr Asn Glu Ala Val His Val
Glu Pro225 230 235 240Val Arg Ala Ser Arg Asn Lys Arg Pro Thr Phe
Leu Lys Ile Lys Lys 245 250 255Pro Leu Ser Tyr Arg Lys Pro Met Asp
Thr Asp Leu Val Tyr Ile Glu 260 265 270Lys Ser Pro Asn Tyr Cys Glu
Glu Asp Pro Val Thr Gly Ser Val Gly 275 280 285Thr Gln Gly Arg Ala
Cys Asn Lys Thr Ala Pro Gln Ala Ser Gly Cys 290 295 300Asp Leu Met
Cys Cys Gly Arg Gly Tyr Asn Thr His Gln Tyr Ala Arg305 310 315
320Val Trp Gln Cys Asn Cys Lys Phe His Trp Cys Cys Tyr Val Lys Cys
325 330 335Asn Thr Cys Ser Glu Arg Thr Glu Met Tyr Thr Cys Lys 340
34581210PRTHomo sapiens 8Met Arg Pro Ser Gly Thr Ala Gly Ala Ala
Leu Leu Ala Leu Leu Ala1 5 10 15Ala Leu Cys Pro Ala Ser Arg Ala Leu
Glu Glu Lys Lys Val Cys Gln 20 25 30Gly Thr Ser Asn Lys Leu Thr Gln
Leu Gly Thr Phe Glu Asp His Phe 35 40 45Leu Ser Leu Gln Arg Met Phe
Asn Asn Cys Glu Val Val Leu Gly Asn 50 55 60Leu Glu Ile Thr Tyr Val
Gln Arg Asn Tyr Asp Leu Ser Phe Leu Lys65 70
75 80Thr Ile Gln Glu Val Ala Gly Tyr Val Leu Ile Ala Leu Asn Thr
Val 85 90 95Glu Arg Ile Pro Leu Glu Asn Leu Gln Ile Ile Arg Gly Asn
Met Tyr 100 105 110Tyr Glu Asn Ser Tyr Ala Leu Ala Val Leu Ser Asn
Tyr Asp Ala Asn 115 120 125Lys Thr Gly Leu Lys Glu Leu Pro Met Arg
Asn Leu Gln Glu Ile Leu 130 135 140His Gly Ala Val Arg Phe Ser Asn
Asn Pro Ala Leu Cys Asn Val Glu145 150 155 160Ser Ile Gln Trp Arg
Asp Ile Val Ser Ser Asp Phe Leu Ser Asn Met 165 170 175Ser Met Asp
Phe Gln Asn His Leu Gly Ser Cys Gln Lys Cys Asp Pro 180 185 190Ser
Cys Pro Asn Gly Ser Cys Trp Gly Ala Gly Glu Glu Asn Cys Gln 195 200
205Lys Leu Thr Lys Ile Ile Cys Ala Gln Gln Cys Ser Gly Arg Cys Arg
210 215 220Gly Lys Ser Pro Ser Asp Cys Cys His Asn Gln Cys Ala Ala
Gly Cys225 230 235 240Thr Gly Pro Arg Glu Ser Asp Cys Leu Val Cys
Arg Lys Phe Arg Asp 245 250 255Glu Ala Thr Cys Lys Asp Thr Cys Pro
Pro Leu Met Leu Tyr Asn Pro 260 265 270Thr Thr Tyr Gln Met Asp Val
Asn Pro Glu Gly Lys Tyr Ser Phe Gly 275 280 285Ala Thr Cys Val Lys
Lys Cys Pro Arg Asn Tyr Val Val Thr Asp His 290 295 300Gly Ser Cys
Val Arg Ala Cys Gly Ala Asp Ser Tyr Glu Met Glu Glu305 310 315
320Asp Gly Val Arg Lys Cys Lys Lys Cys Glu Gly Pro Cys Arg Lys Val
325 330 335Cys Asn Gly Ile Gly Ile Gly Glu Phe Lys Asp Ser Leu Ser
Ile Asn 340 345 350Ala Thr Asn Ile Lys His Phe Lys Asn Cys Thr Ser
Ile Ser Gly Asp 355 360 365Leu His Ile Leu Pro Val Ala Phe Arg Gly
Asp Ser Phe Thr His Thr 370 375 380Pro Pro Leu Asp Pro Gln Glu Leu
Asp Ile Leu Lys Thr Val Lys Glu385 390 395 400Ile Thr Gly Phe Leu
Leu Ile Gln Ala Trp Pro Glu Asn Arg Thr Asp 405 410 415Leu His Ala
Phe Glu Asn Leu Glu Ile Ile Arg Gly Arg Thr Lys Gln 420 425 430His
Gly Gln Phe Ser Leu Ala Val Val Ser Leu Asn Ile Thr Ser Leu 435 440
445Gly Leu Arg Ser Leu Lys Glu Ile Ser Asp Gly Asp Val Ile Ile Ser
450 455 460Gly Asn Lys Asn Leu Cys Tyr Ala Asn Thr Ile Asn Trp Lys
Lys Leu465 470 475 480Phe Gly Thr Ser Gly Gln Lys Thr Lys Ile Ile
Ser Asn Arg Gly Glu 485 490 495Asn Ser Cys Lys Ala Thr Gly Gln Val
Cys His Ala Leu Cys Ser Pro 500 505 510Glu Gly Cys Trp Gly Pro Glu
Pro Arg Asp Cys Val Ser Cys Arg Asn 515 520 525Val Ser Arg Gly Arg
Glu Cys Val Asp Lys Cys Asn Leu Leu Glu Gly 530 535 540Glu Pro Arg
Glu Phe Val Glu Asn Ser Glu Cys Ile Gln Cys His Pro545 550 555
560Glu Cys Leu Pro Gln Ala Met Asn Ile Thr Cys Thr Gly Arg Gly Pro
565 570 575Asp Asn Cys Ile Gln Cys Ala His Tyr Ile Asp Gly Pro His
Cys Val 580 585 590Lys Thr Cys Pro Ala Gly Val Met Gly Glu Asn Asn
Thr Leu Val Trp 595 600 605Lys Tyr Ala Asp Ala Gly His Val Cys His
Leu Cys His Pro Asn Cys 610 615 620Thr Tyr Gly Cys Thr Gly Pro Gly
Leu Glu Gly Cys Pro Thr Asn Gly625 630 635 640Pro Lys Ile Pro Ser
Ile Ala Thr Gly Met Val Gly Ala Leu Leu Leu 645 650 655Leu Leu Val
Val Ala Leu Gly Ile Gly Leu Phe Met Arg Arg Arg His 660 665 670Ile
Val Arg Lys Arg Thr Leu Arg Arg Leu Leu Gln Glu Arg Glu Leu 675 680
685Val Glu Pro Leu Thr Pro Ser Gly Glu Ala Pro Asn Gln Ala Leu Leu
690 695 700Arg Ile Leu Lys Glu Thr Glu Phe Lys Lys Ile Lys Val Leu
Gly Ser705 710 715 720Gly Ala Phe Gly Thr Val Tyr Lys Gly Leu Trp
Ile Pro Glu Gly Glu 725 730 735Lys Val Lys Ile Pro Val Ala Ile Lys
Glu Leu Arg Glu Ala Thr Ser 740 745 750Pro Lys Ala Asn Lys Glu Ile
Leu Asp Glu Ala Tyr Val Met Ala Ser 755 760 765Val Asp Asn Pro His
Val Cys Arg Leu Leu Gly Ile Cys Leu Thr Ser 770 775 780Thr Val Gln
Leu Ile Thr Gln Leu Met Pro Phe Gly Cys Leu Leu Asp785 790 795
800Tyr Val Arg Glu His Lys Asp Asn Ile Gly Ser Gln Tyr Leu Leu Asn
805 810 815Trp Cys Val Gln Ile Ala Lys Gly Met Asn Tyr Leu Glu Asp
Arg Arg 820 825 830Leu Val His Arg Asp Leu Ala Ala Arg Asn Val Leu
Val Lys Thr Pro 835 840 845Gln His Val Lys Ile Thr Asp Phe Gly Leu
Ala Lys Leu Leu Gly Ala 850 855 860Glu Glu Lys Glu Tyr His Ala Glu
Gly Gly Lys Val Pro Ile Lys Trp865 870 875 880Met Ala Leu Glu Ser
Ile Leu His Arg Ile Tyr Thr His Gln Ser Asp 885 890 895Val Trp Ser
Tyr Gly Val Thr Val Trp Glu Leu Met Thr Phe Gly Ser 900 905 910Lys
Pro Tyr Asp Gly Ile Pro Ala Ser Glu Ile Ser Ser Ile Leu Glu 915 920
925Lys Gly Glu Arg Leu Pro Gln Pro Pro Ile Cys Thr Ile Asp Val Tyr
930 935 940Met Ile Met Val Lys Cys Trp Met Ile Asp Ala Asp Ser Arg
Pro Lys945 950 955 960Phe Arg Glu Leu Ile Ile Glu Phe Ser Lys Met
Ala Arg Asp Pro Gln 965 970 975Arg Tyr Leu Val Ile Gln Gly Asp Glu
Arg Met His Leu Pro Ser Pro 980 985 990Thr Asp Ser Asn Phe Tyr Arg
Ala Leu Met Asp Glu Glu Asp Met Asp 995 1000 1005Asp Val Val Asp
Ala Asp Glu Tyr Leu Ile Pro Gln Gln Gly Phe 1010 1015 1020Phe Ser
Ser Pro Ser Thr Ser Arg Thr Pro Leu Leu Ser Ser Leu 1025 1030
1035Ser Ala Thr Ser Asn Asn Ser Thr Val Ala Cys Ile Asp Arg Asn
1040 1045 1050Gly Leu Gln Ser Cys Pro Ile Lys Glu Asp Ser Phe Leu
Gln Arg 1055 1060 1065Tyr Ser Ser Asp Pro Thr Gly Ala Leu Thr Glu
Asp Ser Ile Asp 1070 1075 1080Asp Thr Phe Leu Pro Val Pro Glu Tyr
Ile Asn Gln Ser Val Pro 1085 1090 1095Lys Arg Pro Ala Gly Ser Val
Gln Asn Pro Val Tyr His Asn Gln 1100 1105 1110Pro Leu Asn Pro Ala
Pro Ser Arg Asp Pro His Tyr Gln Asp Pro 1115 1120 1125His Ser Thr
Ala Val Gly Asn Pro Glu Tyr Leu Asn Thr Val Gln 1130 1135 1140Pro
Thr Cys Val Asn Ser Thr Phe Asp Ser Pro Ala His Trp Ala 1145 1150
1155Gln Lys Gly Ser His Gln Ile Ser Leu Asp Asn Pro Asp Tyr Gln
1160 1165 1170Gln Asp Phe Phe Pro Lys Glu Ala Lys Pro Asn Gly Ile
Phe Lys 1175 1180 1185Gly Ser Thr Ala Glu Asn Ala Glu Tyr Leu Arg
Val Ala Pro Gln 1190 1195 1200Ser Ser Glu Phe Ile Gly Ala 1205
121091207PRTHomo sapiens 9Met Leu Leu Thr Leu Ile Ile Leu Leu Pro
Val Val Ser Lys Phe Ser1 5 10 15Phe Val Ser Leu Ser Ala Pro Gln His
Trp Ser Cys Pro Glu Gly Thr 20 25 30Leu Ala Gly Asn Gly Asn Ser Thr
Cys Val Gly Pro Ala Pro Phe Leu 35 40 45Ile Phe Ser His Gly Asn Ser
Ile Phe Arg Ile Asp Thr Glu Gly Thr 50 55 60Asn Tyr Glu Gln Leu Val
Val Asp Ala Gly Val Ser Val Ile Met Asp65 70 75 80Phe His Tyr Asn
Glu Lys Arg Ile Tyr Trp Val Asp Leu Glu Arg Gln 85 90 95Leu Leu Gln
Arg Val Phe Leu Asn Gly Ser Arg Gln Glu Arg Val Cys 100 105 110Asn
Ile Glu Lys Asn Val Ser Gly Met Ala Ile Asn Trp Ile Asn Glu 115 120
125Glu Val Ile Trp Ser Asn Gln Gln Glu Gly Ile Ile Thr Val Thr Asp
130 135 140Met Lys Gly Asn Asn Ser His Ile Leu Leu Ser Ala Leu Lys
Tyr Pro145 150 155 160Ala Asn Val Ala Val Asp Pro Val Glu Arg Phe
Ile Phe Trp Ser Ser 165 170 175Glu Val Ala Gly Ser Leu Tyr Arg Ala
Asp Leu Asp Gly Val Gly Val 180 185 190Lys Ala Leu Leu Glu Thr Ser
Glu Lys Ile Thr Ala Val Ser Leu Asp 195 200 205Val Leu Asp Lys Arg
Leu Phe Trp Ile Gln Tyr Asn Arg Glu Gly Ser 210 215 220Asn Ser Leu
Ile Cys Ser Cys Asp Tyr Asp Gly Gly Ser Val His Ile225 230 235
240Ser Lys His Pro Thr Gln His Asn Leu Phe Ala Met Ser Leu Phe Gly
245 250 255Asp Arg Ile Phe Tyr Ser Thr Trp Lys Met Lys Thr Ile Trp
Ile Ala 260 265 270Asn Lys His Thr Gly Lys Asp Met Val Arg Ile Asn
Leu His Ser Ser 275 280 285Phe Val Pro Leu Gly Glu Leu Lys Val Val
His Pro Leu Ala Gln Pro 290 295 300Lys Ala Glu Asp Asp Thr Trp Glu
Pro Glu Gln Lys Leu Cys Lys Leu305 310 315 320Arg Lys Gly Asn Cys
Ser Ser Thr Val Cys Gly Gln Asp Leu Gln Ser 325 330 335His Leu Cys
Met Cys Ala Glu Gly Tyr Ala Leu Ser Arg Asp Arg Lys 340 345 350Tyr
Cys Glu Asp Val Asn Glu Cys Ala Phe Trp Asn His Gly Cys Thr 355 360
365Leu Gly Cys Lys Asn Thr Pro Gly Ser Tyr Tyr Cys Thr Cys Pro Val
370 375 380Gly Phe Val Leu Leu Pro Asp Gly Lys Arg Cys His Gln Leu
Val Ser385 390 395 400Cys Pro Arg Asn Val Ser Glu Cys Ser His Asp
Cys Val Leu Thr Ser 405 410 415Glu Gly Pro Leu Cys Phe Cys Pro Glu
Gly Ser Val Leu Glu Arg Asp 420 425 430Gly Lys Thr Cys Ser Gly Cys
Ser Ser Pro Asp Asn Gly Gly Cys Ser 435 440 445Gln Leu Cys Val Pro
Leu Ser Pro Val Ser Trp Glu Cys Asp Cys Phe 450 455 460Pro Gly Tyr
Asp Leu Gln Leu Asp Glu Lys Ser Cys Ala Ala Ser Gly465 470 475
480Pro Gln Pro Phe Leu Leu Phe Ala Asn Ser Gln Asp Ile Arg His Met
485 490 495His Phe Asp Gly Thr Asp Tyr Gly Thr Leu Leu Ser Gln Gln
Met Gly 500 505 510Met Val Tyr Ala Leu Asp His Asp Pro Val Glu Asn
Lys Ile Tyr Phe 515 520 525Ala His Thr Ala Leu Lys Trp Ile Glu Arg
Ala Asn Met Asp Gly Ser 530 535 540Gln Arg Glu Arg Leu Ile Glu Glu
Gly Val Asp Val Pro Glu Gly Leu545 550 555 560Ala Val Asp Trp Ile
Gly Arg Arg Phe Tyr Trp Thr Asp Arg Gly Lys 565 570 575Ser Leu Ile
Gly Arg Ser Asp Leu Asn Gly Lys Arg Ser Lys Ile Ile 580 585 590Thr
Lys Glu Asn Ile Ser Gln Pro Arg Gly Ile Ala Val His Pro Met 595 600
605Ala Lys Arg Leu Phe Trp Thr Asp Thr Gly Ile Asn Pro Arg Ile Glu
610 615 620Ser Ser Ser Leu Gln Gly Leu Gly Arg Leu Val Ile Ala Ser
Ser Asp625 630 635 640Leu Ile Trp Pro Ser Gly Ile Thr Ile Asp Phe
Leu Thr Asp Lys Leu 645 650 655Tyr Trp Cys Asp Ala Lys Gln Ser Val
Ile Glu Met Ala Asn Leu Asp 660 665 670Gly Ser Lys Arg Arg Arg Leu
Thr Gln Asn Asp Val Gly His Pro Phe 675 680 685Ala Val Ala Val Phe
Glu Asp Tyr Val Trp Phe Ser Asp Trp Ala Met 690 695 700Pro Ser Val
Ile Arg Val Asn Lys Arg Thr Gly Lys Asp Arg Val Arg705 710 715
720Leu Gln Gly Ser Met Leu Lys Pro Ser Ser Leu Val Val Val His Pro
725 730 735Leu Ala Lys Pro Gly Ala Asp Pro Cys Leu Tyr Gln Asn Gly
Gly Cys 740 745 750Glu His Ile Cys Lys Lys Arg Leu Gly Thr Ala Trp
Cys Ser Cys Arg 755 760 765Glu Gly Phe Met Lys Ala Ser Asp Gly Lys
Thr Cys Leu Ala Leu Asp 770 775 780Gly His Gln Leu Leu Ala Gly Gly
Glu Val Asp Leu Lys Asn Gln Val785 790 795 800Thr Pro Leu Asp Ile
Leu Ser Lys Thr Arg Val Ser Glu Asp Asn Ile 805 810 815Thr Glu Ser
Gln His Met Leu Val Ala Glu Ile Met Val Ser Asp Gln 820 825 830Asp
Asp Cys Ala Pro Val Gly Cys Ser Met Tyr Ala Arg Cys Ile Ser 835 840
845Glu Gly Glu Asp Ala Thr Cys Gln Cys Leu Lys Gly Phe Ala Gly Asp
850 855 860Gly Lys Leu Cys Ser Asp Ile Asp Glu Cys Glu Met Gly Val
Pro Val865 870 875 880Cys Pro Pro Ala Ser Ser Lys Cys Ile Asn Thr
Glu Gly Gly Tyr Val 885 890 895Cys Arg Cys Ser Glu Gly Tyr Gln Gly
Asp Gly Ile His Cys Leu Asp 900 905 910Ile Asp Glu Cys Gln Leu Gly
Val His Ser Cys Gly Glu Asn Ala Ser 915 920 925Cys Thr Asn Thr Glu
Gly Gly Tyr Thr Cys Met Cys Ala Gly Arg Leu 930 935 940Ser Glu Pro
Gly Leu Ile Cys Pro Asp Ser Thr Pro Pro Pro His Leu945 950 955
960Arg Glu Asp Asp His His Tyr Ser Val Arg Asn Ser Asp Ser Glu Cys
965 970 975Pro Leu Ser His Asp Gly Tyr Cys Leu His Asp Gly Val Cys
Met Tyr 980 985 990Ile Glu Ala Leu Asp Lys Tyr Ala Cys Asn Cys Val
Val Gly Tyr Ile 995 1000 1005Gly Glu Arg Cys Gln Tyr Arg Asp Leu
Lys Trp Trp Glu Leu Arg 1010 1015 1020His Ala Gly His Gly Gln Gln
Gln Lys Val Ile Val Val Ala Val 1025 1030 1035Cys Val Val Val Leu
Val Met Leu Leu Leu Leu Ser Leu Trp Gly 1040 1045 1050Ala His Tyr
Tyr Arg Thr Gln Lys Leu Leu Ser Lys Asn Pro Lys 1055 1060 1065Asn
Pro Tyr Glu Glu Ser Ser Arg Asp Val Arg Ser Arg Arg Pro 1070 1075
1080Ala Asp Thr Glu Asp Gly Met Ser Ser Cys Pro Gln Pro Trp Phe
1085 1090 1095Val Val Ile Lys Glu His Gln Asp Leu Lys Asn Gly Gly
Gln Pro 1100 1105 1110Val Ala Gly Glu Asp Gly Gln Ala Ala Asp Gly
Ser Met Gln Pro 1115 1120 1125Thr Ser Trp Arg Gln Glu Pro Gln Leu
Cys Gly Met Gly Thr Glu 1130 1135 1140Gln Gly Cys Trp Ile Pro Val
Ser Ser Asp Lys Gly Ser Cys Pro 1145 1150 1155Gln Val Met Glu Arg
Ser Phe His Met Pro Ser Tyr Gly Thr Gln 1160 1165 1170Thr Leu Glu
Gly Gly Val Glu Lys Pro His Ser Leu Leu Ser Ala 1175 1180 1185Asn
Pro Leu Trp Gln Gln Arg Ala Leu Asp Pro Pro His Gln Met 1190 1195
1200Glu Leu Thr Gln 1205
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
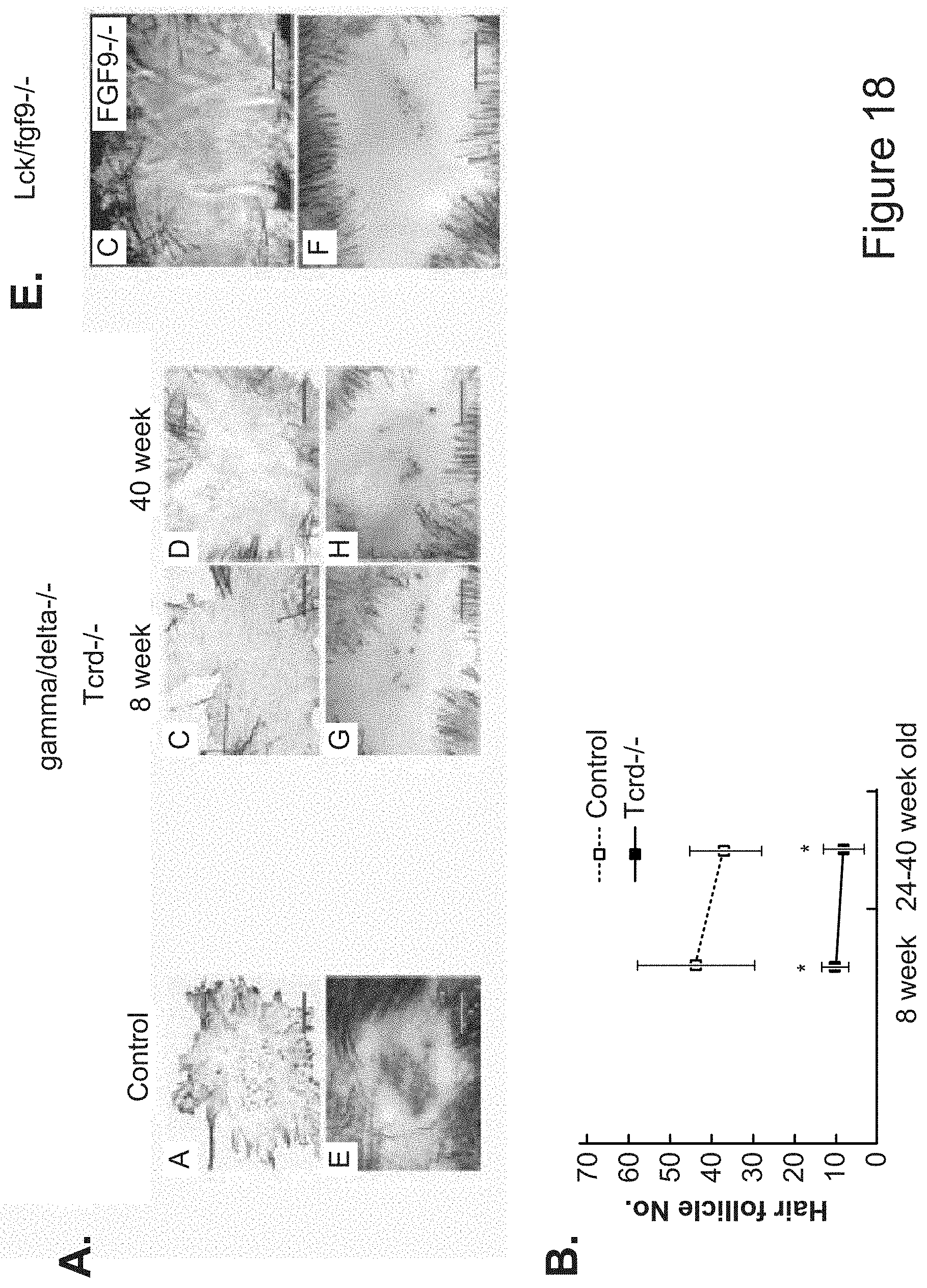
D00015
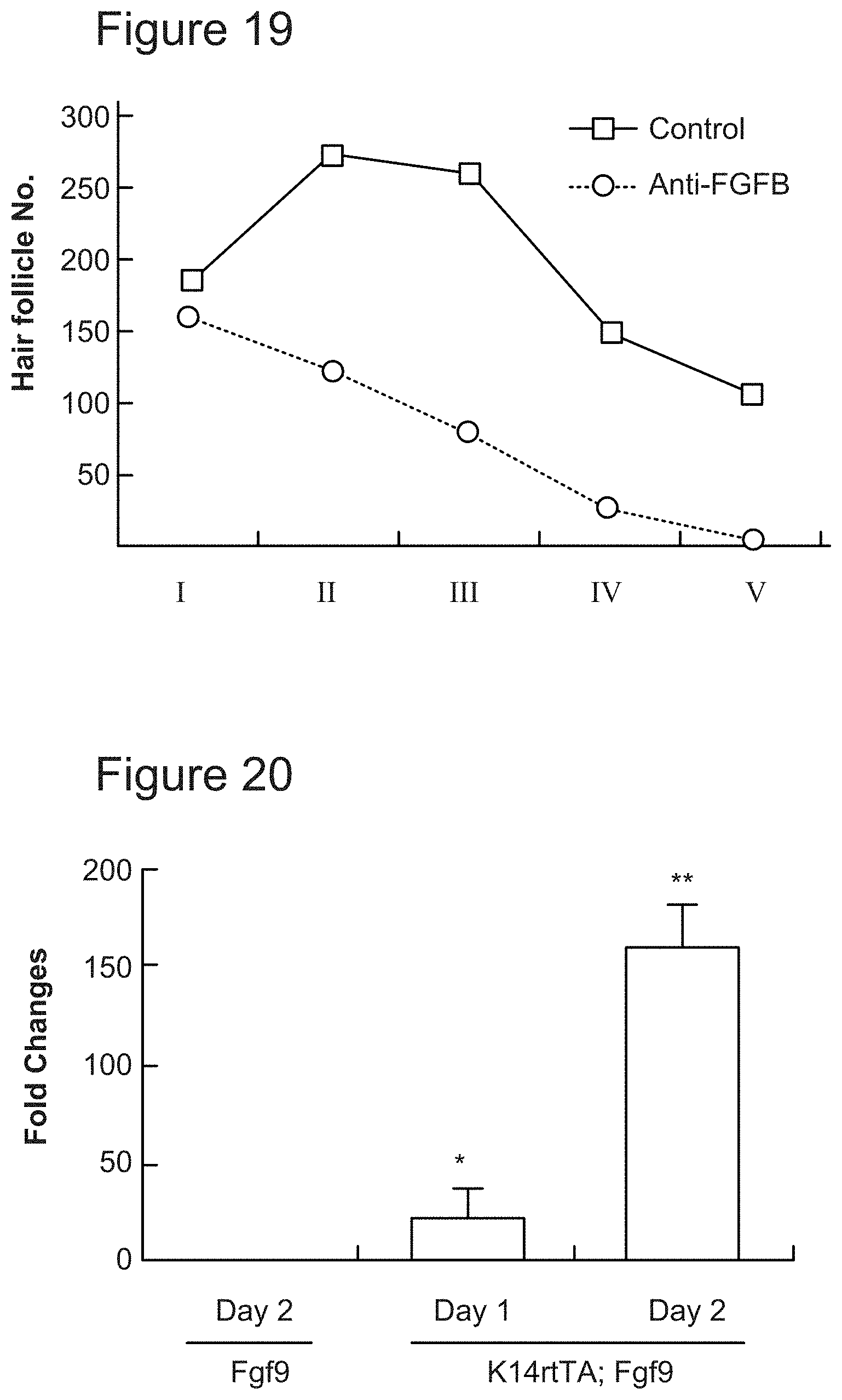
D00016

D00017

D00018

D00019

D00020
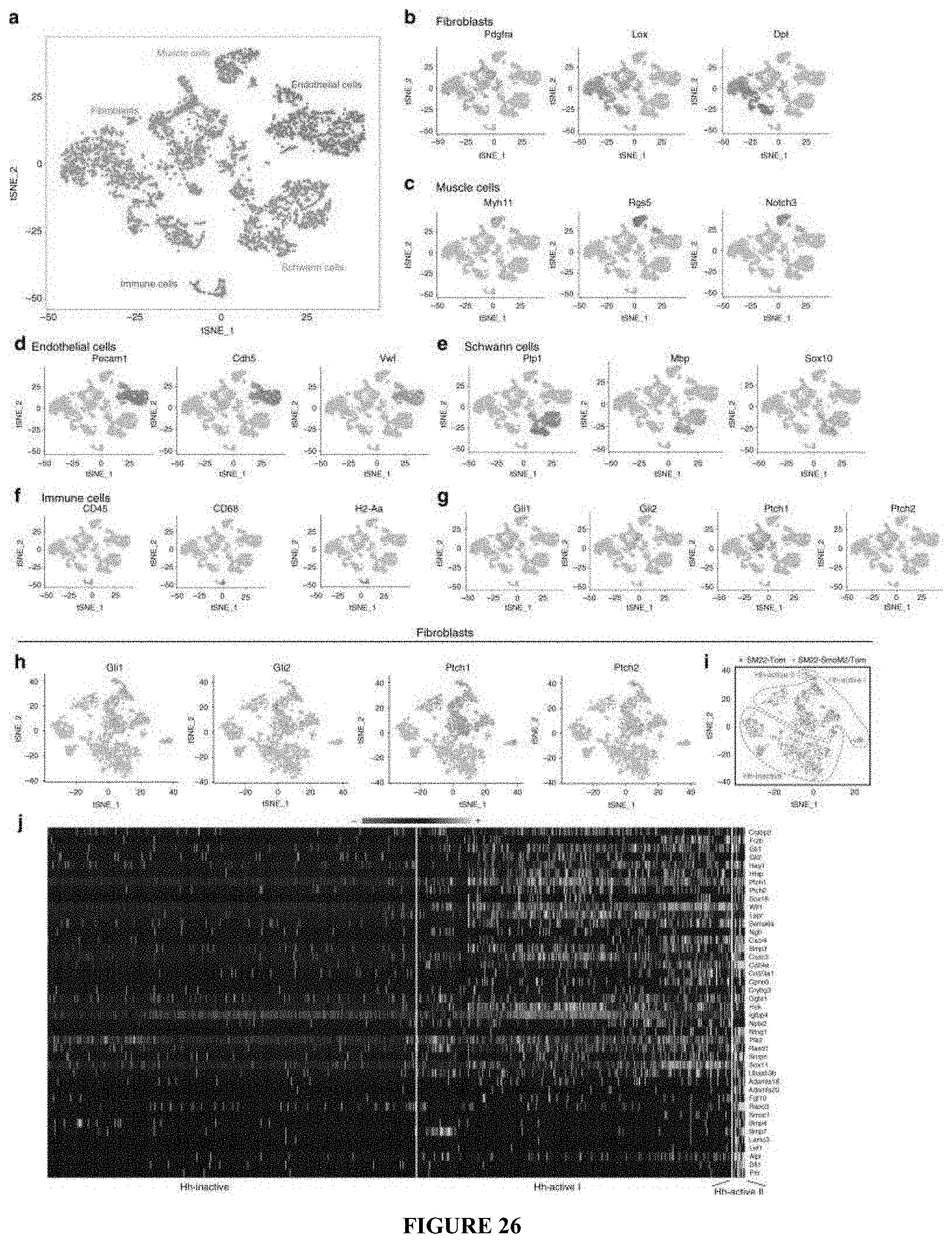
D00021

D00022

D00023

D00024
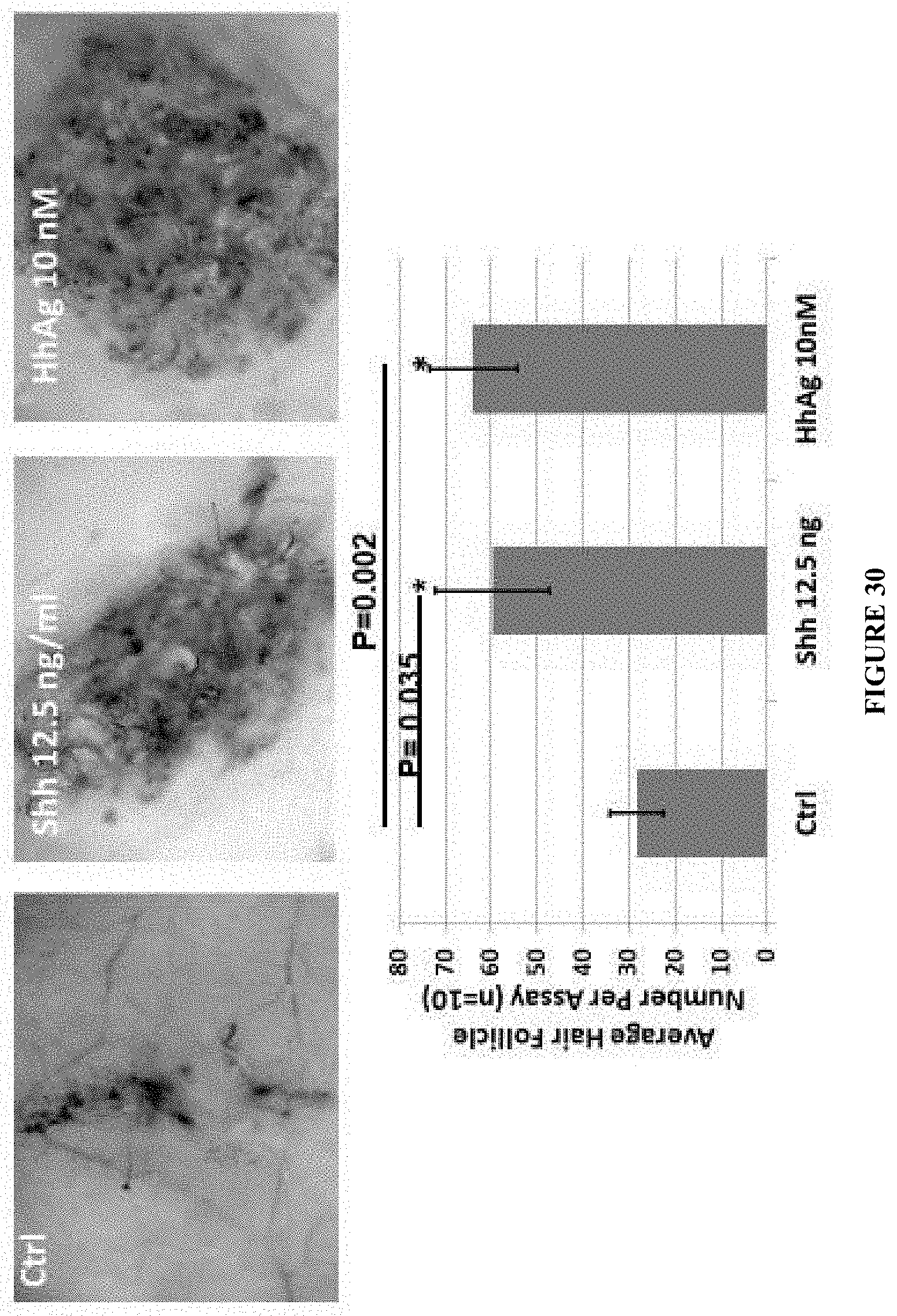
D00025
D00026
D00027

D00028

D00029

D00030

D00031

D00032

D00033

D00034
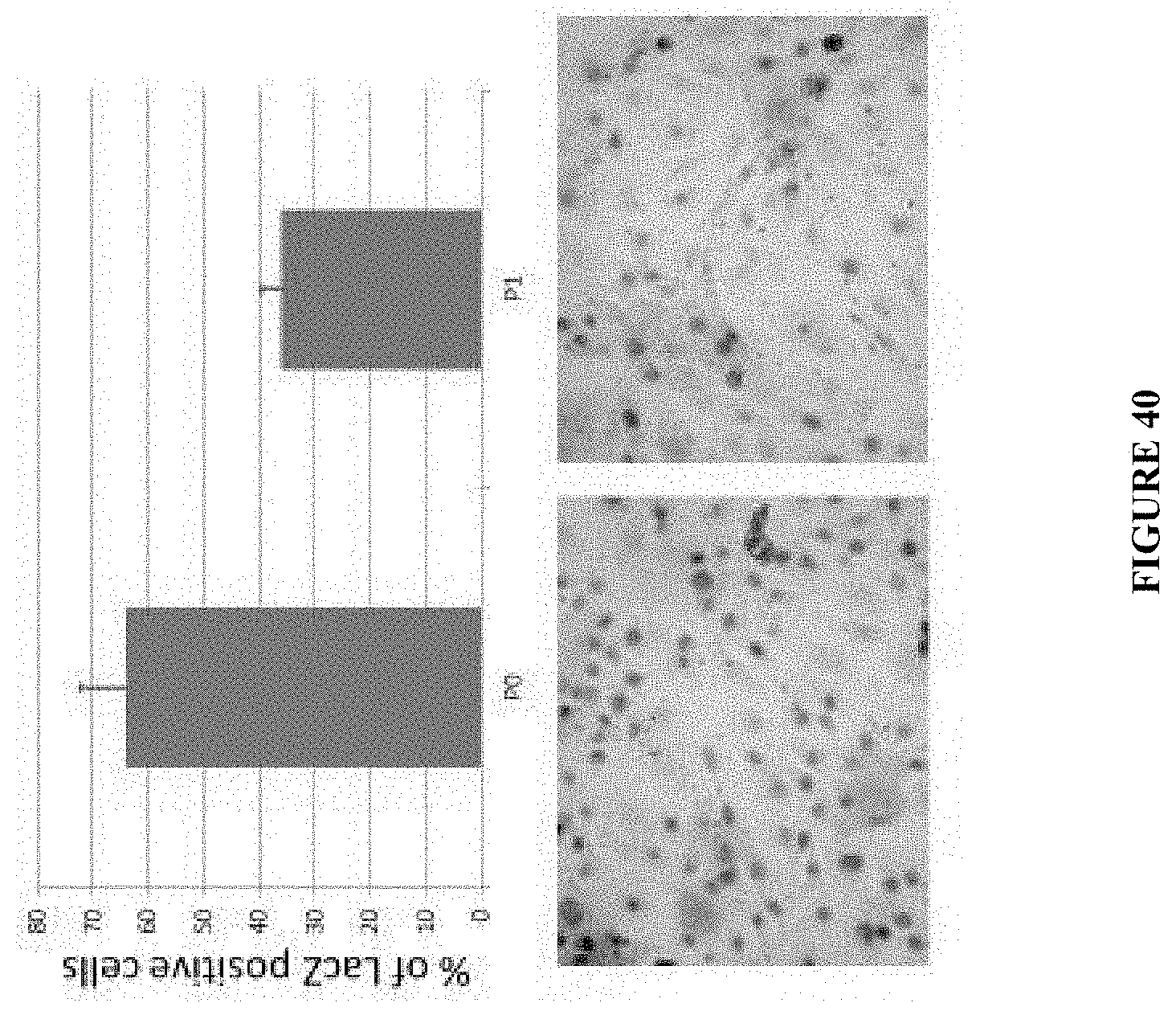
D00035

D00036

D00037

D00038

P00899

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.