Nanofiber Structure Constituted Of Polyhydroxyalkanoic Acid, And Non-woven Fabric
KUMAR; Sudesh K. ; et al.
U.S. patent application number 16/315985 was filed with the patent office on 2020-06-11 for nanofiber structure constituted of polyhydroxyalkanoic acid, and non-woven fabric. This patent application is currently assigned to FUENCE CO., LTD.. The applicant listed for this patent is FUENCE CO., LTD.. Invention is credited to Kozo INOUE, Sudesh K. KUMAR, Kazuya NITTA.
| Application Number | 20200181818 16/315985 |
| Document ID | / |
| Family ID | 60577689 |
| Filed Date | 2020-06-11 |

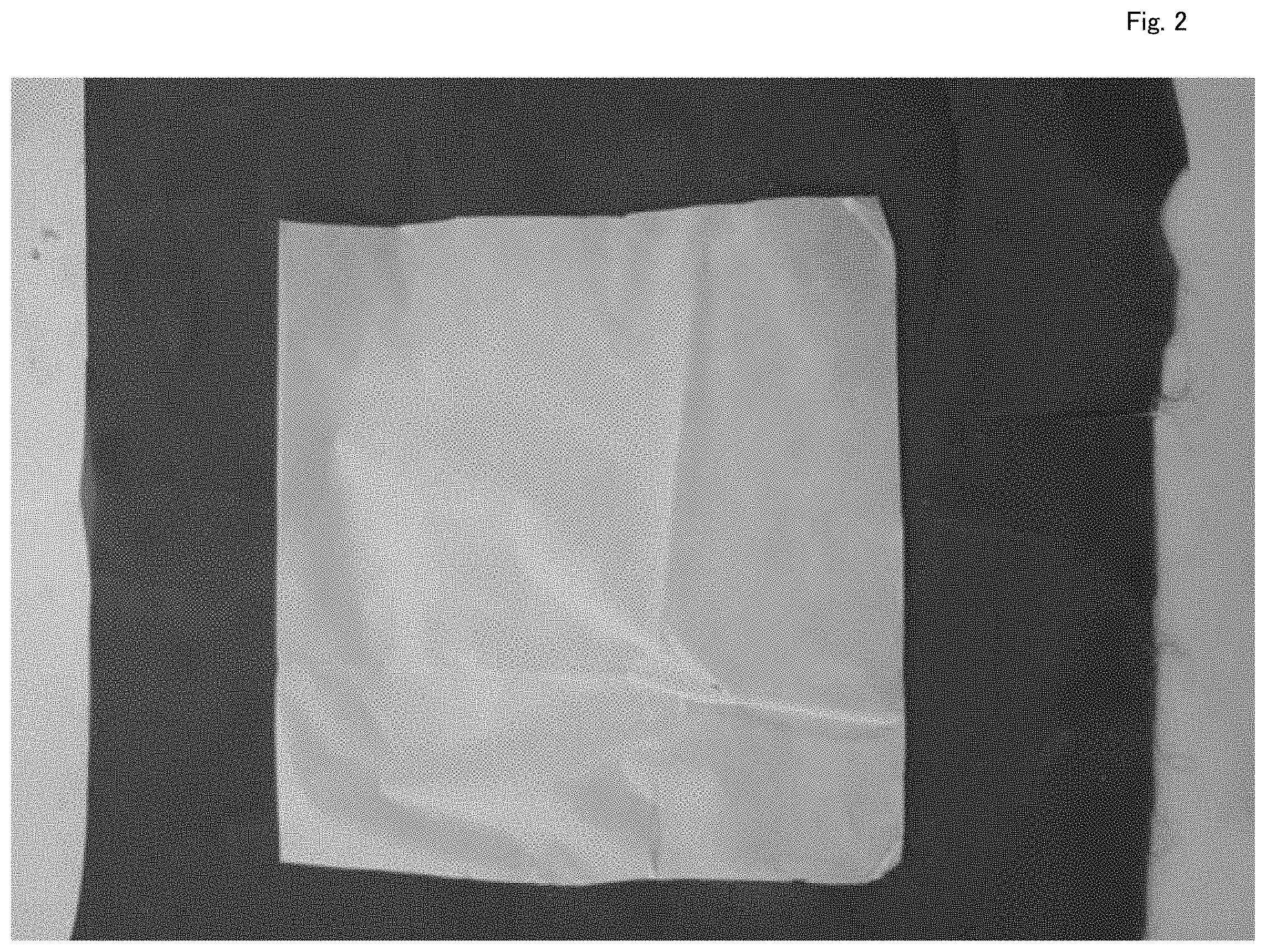


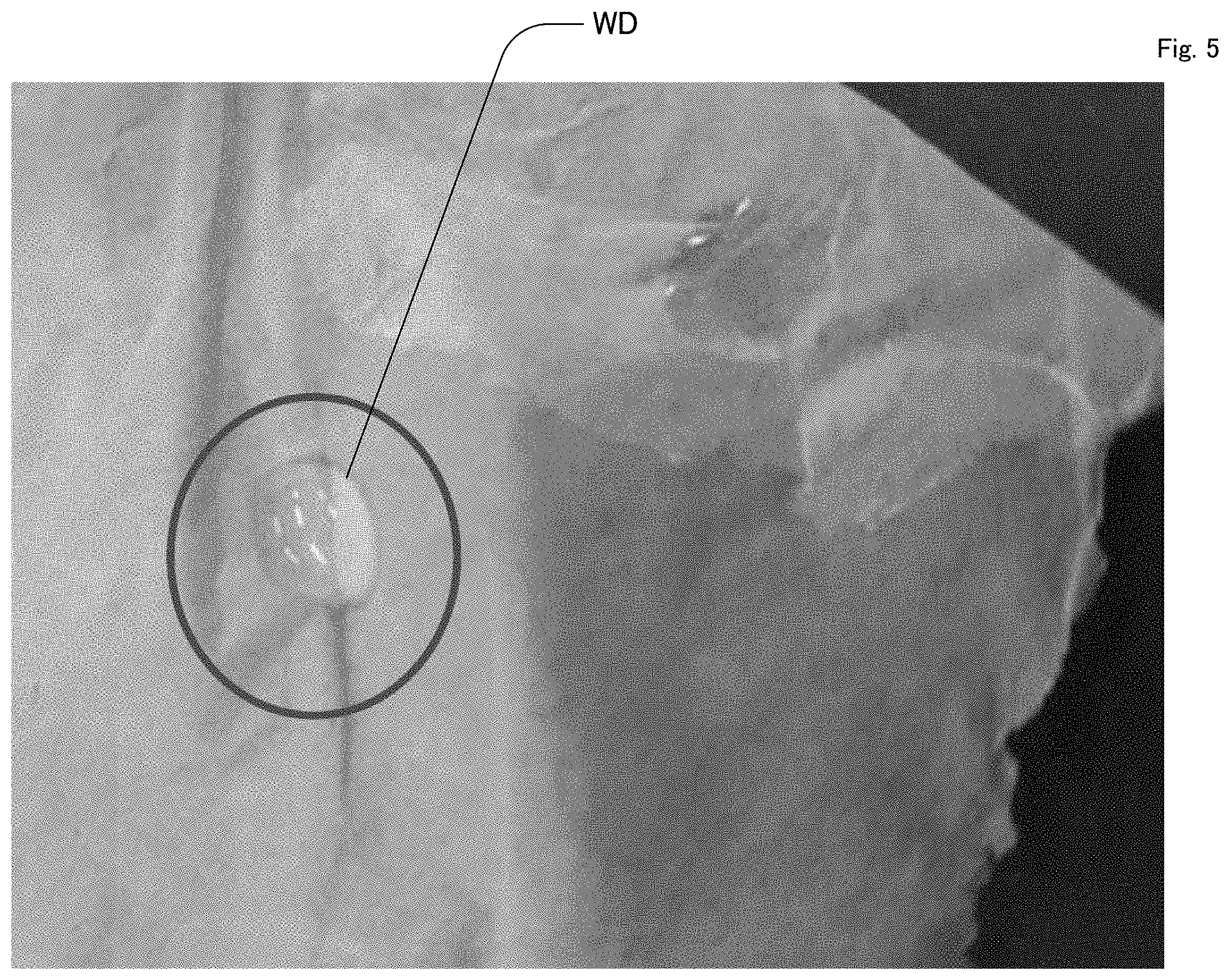




| United States Patent Application | 20200181818 |
| Kind Code | A1 |
| KUMAR; Sudesh K. ; et al. | June 11, 2020 |
NANOFIBER STRUCTURE CONSTITUTED OF POLYHYDROXYALKANOIC ACID, AND NON-WOVEN FABRIC
Abstract
The biodegradability of a nanofiber film (a nanofiber structure) produced in example 1 by microorganisms or the like when the nanofiber film is allowed to leave in soil is examined. FIG. 4(a) shows a photograph of the nanofiber film immediately after the nanofiber film is placed in soil. FIG. 4(b) shows a photograph of the nanofiber film (a) that is allowed to leave as it for 12 days. As is obvious from the comparison between these photographs, a polyhydroxyalkanoic acid nanofiber film can be degraded in soil remarkably rapidly. Therefore, PHA can be produced from a plant-derived resource occurring in nature, can be degraded by microorganisms in soil to return to nature, and can be used as a resource material which can overcome the disadvantages of the conventional PP non-woven fabrics (e.g., the generation of CO.sub.2 upon incineration) and which can be used permanently, thereby enabling the production of a novel non-woven fabric.
| Inventors: | KUMAR; Sudesh K.; (Penang, MY) ; INOUE; Kozo; (Tokyo, JP) ; NITTA; Kazuya; (Tokyo, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | FUENCE CO., LTD. Tokyo JP |
||||||||||
| Family ID: | 60577689 | ||||||||||
| Appl. No.: | 16/315985 | ||||||||||
| Filed: | June 7, 2016 | ||||||||||
| PCT Filed: | June 7, 2016 | ||||||||||
| PCT NO: | PCT/JP2016/066904 | ||||||||||
| 371 Date: | January 7, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | D10B 2401/021 20130101; D04H 1/4382 20130101; D04H 1/435 20130101; D01F 6/62 20130101; D10B 2401/022 20130101; D10B 2401/12 20130101 |
| International Class: | D04H 1/435 20060101 D04H001/435; D04H 1/4382 20060101 D04H001/4382; D01F 6/62 20060101 D01F006/62 |
Claims
1. A nanofiber structure formed of polyhydroxyalkanoic acid.
2. The nanofiber structure according to claim 1, wherein the polyhydroxyalkanoic acid includes polyhydroxybutylate as a main component.
3. The nanofiber structure according to claim 1, wherein the nanofiber structure has a fiber diameter of 1 .mu.m or less.
4. The nanofiber structure according to claim 1, wherein the nanofiber structure is degraded by microorganisms in soil in a natural environment.
5. The nanofiber structure according to claim 1, wherein the nanofiber structure has a porosity of 50% or more.
6. The nanofiber structure according to claim 1, wherein the nanofiber structure has water repellency, and a contact angle of pure water to a surface of the nanofiber structure is 100.degree. or more.
7. The nanofiber structure according to claim 1, wherein the nanofiber structure has oil absorbency.
8. The nanofiber structure according to claim 1, wherein the nanofiber structure has organic solvent absorbency.
9. The nanofiber structure according to claim 1, wherein the surface of the nanofiber structure has hydrophilicity by surface modification by a plasma treatment, a corona discharge, electron beam irradiation, or laser irradiation, or the like.
10. The nanofiber structure according to claim 1, further comprising an adsorbent material.
11. The nanofiber structure according to claim 1, wherein the nanofiber structure is partially fused to have a film shape.
Description
TECHNICAL FIELD
[0001] The present invention relates to a nanofiber structure constituted of polyhydroxyalkanoic acid, and a non-woven fabric, and more particularly, to a nanofiber structure constituted of polyhydroxyalkanoic acid having oil or organic solvent absorbency simultaneously with a property of being rapidly degraded by microorganisms and the like in the natural environment, and a non-woven fabric.
BACKGROUND ART
[0002] Usage of a non-woven fabric manufactured from various organic polymers has recently been expanded, and is used for various uses in various industries from textile industries (interlining field) to sanitary materials, medical materials, automobile interior materials, industrial materials (filter, wiping, etc.), civil engineering materials, agricultural materials, geotextiles (fiber sheet for soil reinforcement), environmental industries, and the like. In the future also, production of the non-woven fabric is predicted to continuously expand every year. Among the non-woven fabrics, polypropylene (PP) non-woven fabric has a high growth rate, and a growth rate of nearly 10% is expected.
[0003] However, growth of the non-woven fabric industry made of the organic polymer has a big problem at the same time. Since the organic polymer which is a main component of the non-woven fabric is purified and synthesized from petroleum resources, problems of depletion of future resources and treatment of used products occur. The treatment of used products (wastes) is treating the used products by pelletizing and the like to recycle the used product as resources, incinerating the used products, burying and disposing the used products, and the like. The products to be recycled as resources are in a very small amount, and in the present situation, the used products are largely incinerated or buried. Since incineration discharges CO.sub.2 in a large amount, influence on global warming is an important problem. In addition, in the case of landfills, since resins derived from petroleum are very difficult to degrade, the resins remain in the ground almost permanently such that the global environment is polluted forever.
[0004] One of the solutions to these problems is to use a biodegradable polymer in a raw material of the non-woven fabric. Resin products using a biodegradable organic polymer such as polylactic acid or polyhydroxyalkanoic acid have been already developed. Although the polyhydroxyalkanoic acid has been developed for a long time by domestic and foreign companies, costs of production and purification by microorganisms delay practicality.
[0005] However, Kaneka Corporation recently reported that commercialization of a resin product manufactured from polyhydroxyalkanoic acid is in progress. However, since the company aims to develop a so called, resin product, development in the non-woven fabric field as described above has not been made.
[0006] As non patent literatures of the related art of the above-described biodegradable polymer, for example, there are "Conclusion of a blanket agreement for product development of "Kaneka Biopolymer AONILEX" with Biotech of Germany (http://www.kaneka.co.jp/service/news/150217)" (Non Patent Literature 1), "Certification of development of plant-derived biodegradable resin manufacture technology by Japan Science and Technology Agency (http://www.kaneka.co.jp/service/news/140710-2)" (Non Patent Literature 2), "Manufacture of biodegradable plastic by microorganisms (Microbiol. Cult. Coll. 29(1):25-29, 2013)" (Non Patent Literature 3), and "The world's first full development of 100% plant-derived biopolymer having softness and thermal resistance (http://www.kaneka.co.jp/service/news/n090206.html)" (Non patent Literature 4).
CITATION LIST
Non Patent Literature
[0007] Non Patent Literature 1: A website of "Conclusion of a blanket agreement for product development of "Kaneka Biopolymer AONILEX" with Biotech of Germany": http://www.kaneka.co.jp/service/news/150217 [0008] Non Patent Literature 2: A website of "Certification of development of plant-derived biodegradable resin manufacture technology by Japan Science and Technology Agency": http://www.kaneka.co.jp/service/news/140710-2 [0009] Non Patent Literature 3: "Manufacture of biodegradable plastic by microorganisms (Microbiol. Cult. Coll. 29(1): 25-29, 2013)" written by Tetsuya Fujiki [0010] Non Patent Literature 4: A website of "The world's first full development of 100% plant-derived biopolymer having softness and thermal resistance": http://www.kaneka.co.jp/service/news/n090206.html
SUMMARY OF INVENTION
Technical Problem
[0011] According to a market report, it is predicted that a market size of a non-woven fabric, in particular a polypropylene (PP) non-woven fabric is to increase by about 8% every year in the future, and is to reach about 30 billion US dollars in 2020. The main uses thereof are sanitary goods such as diapers (for infants or the elderly), geotextiles, environmental pollutant treatments, automobile industries, furniture, and the like, and it is said that the uses are caused by growth in Asia-Pacific region which has a high population growth rate.
[0012] In addition, it is predicted that the production quantity of non-woven fabric is to increase from 5.94 million in 2013 to 9.97 million in 2020. As long as petroleum resource-derived PP is used, CO.sub.2 emissions are increased in contrast to a global agreement to prevent global warming, and thus, effective measures are needed. The inventors of the present application established a technique for inexpensively carrying out a production and purification process of biodegradable polyhydroxyalkanoic acid (PHA) by microorganisms, and conducted an applied study for various uses.
[0013] Further, the inventors of the present application repeated study and speculation for solving the problems, and as a result, found a technique of making biodegradable polyhydroxyalkanoic acid into nanofiber and using the nanofiber as a nanofiber structure (such as non-woven fabric) having various characteristics.
[0014] An object of the present invention is to provide a nanofiber structure constituted of polyhydroxyalkanoic acid. Another object of the present invention is to develop the nanofiber structure as the non-woven fabric to solve the problems of the current synthetic resin non-woven fabric.
Solution to Problem
[0015] To solve the above problems, a nanofiber structure according to a first invention is a nanofiber structure constituted of polyhydroxyalkanoic acid (one or plural types).
[0016] In addition, a nanofiber structure according to a second invention is characterized in that the polyhydroxyalkanoic acid includes polyhydroxybutylate as a main component.
[0017] The structure includes polyhydroxybutylate as a main component, and preferably, is blended with another polyhydroxyalkanoic acid (for example, a copolymer with polyhydroxyhexanoic acid).
[0018] In addition, a nanofiber structure according to a third invention is characterized by having a fiber diameter of 1 .mu.m or less.
[0019] In addition, a nanofiber structure according to a fourth invention has a characteristic of being degraded by microorganisms in the soil in the natural environment.
[0020] In addition, a nanofiber structure according to a fifth invention is characterized by having a porosity of 50% or more.
[0021] The structure has high air permeability and a light weight by having higher porosity.
[0022] In addition, a nanofiber structure according to a sixth invention is characterized by having water repellency, and a contact angle of pure water to a surface of the nanofiber structure is 100.degree. or more.
[0023] In addition, a nanofiber structure according to a seventh invention is characterized by having oil absorbency.
[0024] In addition, a nanofiber structure according to an eighth invention is characterized by having organic solvent absorbency.
[0025] In addition, a nanofiber structure according to a ninth invention is characterized in that the surface of the nanofiber structure has hydrophilicity by surface modification by a plasma treatment, a corona discharge, electron beam irradiation, or laser irradiation.
[0026] The nanofiber structure may be used in sanitary products and the like by having hydrophilicity by surface modification.
[0027] In addition, a nanofiber structure according to a tenth invention is characterized by including an adsorbent material.
[0028] An adsorbent is for example, activated carbon, zeolite, or the like, and included in a nanofiber and on a surface of the nanofiber.
[0029] In addition, a nanofiber structure according to an eleventh invention is characterized in that the nanofiber structure is partially fused to have a film shape.
[0030] As described above, the solution to the problem of the present invention is described as a nanofiber structure, however, the present invention may also be realized by a method of manufacturing a nanofiber structure which substantially corresponds to the solution, and it should be understood that the scope of the present invention also includes the method.
Advantageous Effects of Invention
[0031] According to the present invention, it is possible to provide a nanofiber structure (film) having characteristics of being flexible and having oil or organic solvent absorbency, simultaneously with being rapidly degraded by microorganisms in the natural environment so as not to cause an increase of CO.sub.2 gas. The nanofiber structure may be used as non-woven fabric in various industries.
BRIEF DESCRIPTION OF DRAWINGS
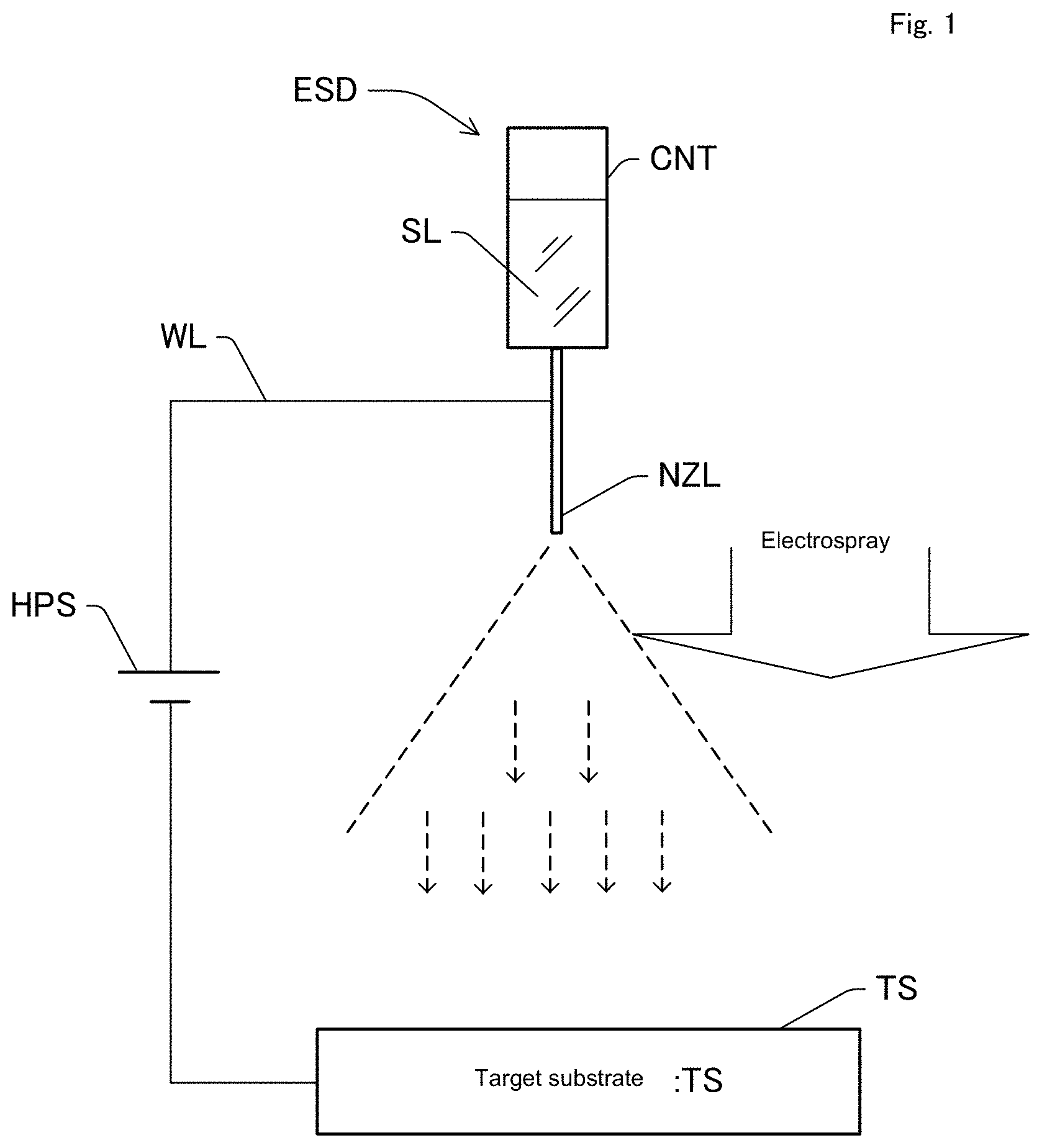
[0032] FIG. 1 is a conceptual diagram representing a basic configuration of an electrospray deposition device.
[0033] FIG. 2 is a SEM photograph of a nanofiber structure manufactured in Example 1 as a material.
[0034] FIG. 3 is an electron microscope photograph (SEM photograph) of a PHA nanofiber structure shown in FIG. 2.
[0035] FIG. 4 is a drawing representing biodegradability of a nanofiber film of Example 1.
[0036] FIG. 5 is a drawing representing water repellency of the nanofiber film of Example 1.
[0037] FIG. 6 is a drawing representing oil-water separability and oil absorbency of the nanofiber film of Example 1.
[0038] FIG. 7 is a drawing representing organic solvent absorbency of the nanofiber film of Example 1.
[0039] FIG. 8 is an electron microscope photograph (SEM photograph) of a nanofiber film (nanofiber structure) which is partially fused to have a film shape.
[0040] FIG. 9 is an electron microscope photograph (SEM photograph) of a nanofiber film including fine particles as an adsorbent material.
DESCRIPTION OF EMBODIMENTS
[0041] Hereinafter, embodiments of the present invention will be described in detail with reference to the drawings.
[0042] A polyhydroxyalkanoic acid used in an exemplary embodiment of the present invention is a sample prepared by microbial culture and purification method, the patentee of which is University of Science-Malaysia to which one of the inventors of the present application belongs. Nanofiber may be manufactured from the sample by an electrospray deposition (ESD) method, a melt blown method, or other method of manufacturing nanofiber, however, an ESD method or a melt blown method is preferred.
<Electrospray Deposition Method>
[0043] Before the embodiments of the present invention are described, the principle of an electrospray deposition method (ESD method) used in the embodiment of the present invention and an electrospray deposition device (ESD: electrospray device) allowing the electrospray deposition method to be carried out will be described.
<Electrospray Deposition Device>
[0044] FIG. 1 is a conceptual diagram representing a basic configuration of an electrospray deposition device. As shown in the drawing, a container (CNT) contains a sample solution (SL). The sample solution (SL) is, for example, an organic polymer solution, a polymer solution, or the like. In the present embodiment, the sample solution is a polyhydroxyalkanoic acid solution, that is, a polyhydroxyalkanoic acid solution.
[0045] Although the ESD method is a very complicated physical phenomenon and all of the processes are not explained, the ESD method is generally considered as being the following phenomenon. The sample solution is contained in a thin capillary shaped nozzle (NZL), and voltage of thousands to tens of thousands of volts is applied to a target substrate (TS) (counter electrode) opposing thereto. At a capillary tip, a strong electric field occurs by an electric field concentration effect, and microdroplets with charge on a liquid surface gather to form a cone (also called Taylor cone). In addition, the sample solution from the tip destroys surface tension to become a jet. The jet is strongly charged and becomes spray by a repulsion of electrostatic force (coulomb explosion). The droplets formed by spray are very small so that the solvent is evaporated and dried within a short time to become fine nanoparticles or nanofiber. Of course, the solvent may be deposited in a wet state which is not evaporated or dried. The charged fine nanoparticles or nanofiber having a small diameter is pulled to the target substrate (TS) functioning as a counter electrode by electrostatic force. A pattern to be deposited may be controlled by an insulator mask or an auxiliary electrode (not shown). The sample is not limited to a solution, and a dispersion solution is fine.
[0046] In addition, preferably, the sample solution in the container (CNT) applies extrusion pressure toward the nozzle (NZL) by an air pressure syringe pump, plunger, or the like (ejection means, not shown). The extrusion pressure is imparted by for example, a stepping motor and a screw feed mechanism (not shown). The sample solution (SL) to which the extrusion pressure is applied has increased internal pressure in the container (CNT) so as to be discharged from the tip of nozzle (NZL). As described above, by installing an adjustment mechanism (the stepping motor and the screw feed mechanism) adjusting the speed of ejecting the sample solution, it is possible to adjust the ejection speed appropriately.
[0047] The nozzle (NZL) is made of metal, and positive voltage is supplied from a high voltage power supply (HPS) through a conductor wire (WL). The negative side of the high voltage power supply (HPS) is connected to the target substrate (TS) (substrate to be a counter electrode). By applying voltage from the high voltage power supply (HPS), positive voltage is applied via the nozzle (NZL) to the sample solution (SL) so that the solution is positively charged. The polarity of the voltage applied to the sample solution (SL) may be negative.
[0048] In addition, when the nanofiber structure is manufactured, it is preferred that non-woven fabric is placed on the target substrate (TS), and the nanofiber structure is deposited on the non-woven fabric. In addition, various conditions such as voltage level, concentration of the sample solution, the kind of polyhydroxyalkanoic acid as a sample, the kind of solvent, and the like are adjusted to manufacture the nanofiber structure.
[0049] The sprayed material becomes fiber or droplets, and repeats division during scattering by repulsion due to charging to form nanofiber or nanoparticles. Since the sprayed material has a large surface area in a nano size, when the sprayed material comes into contact with the substrate, it is in an almost dried state. The shape or size may be changed depending on the spray conditions, and for example, when a polymer solution is used, thick nanofiber is formed with a high molecular weight and a high concentration, and thin nanofiber or nanoparticles are formed with a low molecular weight and a low concentration. Besides, various conditions such as voltage or a distance between the nozzle and the substrate and ambient temperature or humidity have an influence thereon. In the present embodiment, various kinds of solvent-soluble polyhydroxyalkanoic acid are used as a sample to manufacture nanofiber under various conditions, and confirmation of water repellency, air permeability, hydrophilicity, and the like were carried out by the method described in the Example. As the electrospray deposition device, another type of ESD device as well as the above-described device can be used. In particular, for mass production, a method using air current described in Japanese Patent No. 5491189, developed by the applicants, is preferred.
[0050] In addition, during mass production, a non-woven manufacturing device using a melt blown method is also preferred, in addition to the ESD device.
<Example 1> Nanofiberization by ESD Method
[0051] FIG. 2 is a photograph of a polyhydroxyalkanoic acid nanofiber film (PHA nanofiber structure) manufactured by an ESD device of FIG. 1. As a sample solution, a chloroform solution including 10% by weight of polyhydroxyalkanoic acid (PHA) was used. In the manufacturing process, an ESD device (ES-4000, manufactured by HUENS Co., LTD.) was used to spray the solution at voltage of 50 kV and a flow rate of 10 .mu.l/min. A thickness of nanofiber film shown in the drawing was 20 .mu.m. This nanofiber film is very thin, is a free-standing film in spite of the small fiber diameter, may be deposited on other non-woven fabric or film or incorporated into another member or instrument, and is very useful.
[0052] FIG. 3 is an electron microscope photograph (SEM photograph) of the PHA nanofiber structure shown in FIG. 2. The magnification of the photograph is 1000 times. In addition, an average diameter of the nanofiber was about 1 .mu.m. As shown in the drawing, it is observed that a porous film in which fiber is entangled in a mesh-like pattern is formed, which has high porosity and forms a light structure. The PHA nanofiber structure may be used as a filter using the porous property. The nanofiber diameter, porosity, density, and the like are varied by changing various solution compositions or spray conditions according to the purpose, and are controllable.
<Example 2> Biodegradability
[0053] FIG. 4 is a drawing representing biodegradability of the nanofiber film of Example 1. The biodegradability of the nanofiber film (nanofiber structure) obtained in Example 1 by microorganisms and the like was studied by leaving the nanofiber film in soil. FIG. 4(a) is a photograph immediately after placing the nanofiber film in soil. FIG. 4(b) is a photograph after leaving the nanofiber film in FIG. 4(a) for 12 days as it is. As seen from the comparison of these photographs, the polyhydroxyalkanoic acid nanofiber film degrades quite rapidly in soil. As such, since PHA can be produced by microbial fermentation from a plant raw material of nature, and degraded by microorganisms in soil to be returned to nature, the nanofiber film may be used as a resource which does not increase gas causing global warming and may be permanently used.
<Example 3> Water Repellency
[0054] FIG. 5 is a drawing representing water repellency of the nanofiber film of Example 1. FIG. 5 is a photograph immediately after adding pure water dropwise by a pipette on the nanofiber film obtained in Example 1. The dropped pure water (WD) remained on the film as a droplet, as shown in the photograph. As a result of visually measuring the contact angle, a value of 87.5-130.5.degree. was obtained by measurement with 10 droplets, and the average was 113.7.degree.. The nanofiber film had water repellency.
<Example 4> Oil-Water Separability and Oil Absorbency
[0055] FIG. 6 is a drawing representing oil-water separability and oil absorbency of the nanofiber film of Example 1. The nanofiber film obtained in Example 1 was added to a container having a methylene blue solution and salad oil therein by pouring it from the above. FIG. 6(a) is a photograph before the nanofiber film was added to the container. The aqueous methylene blue solution and salad oil are mixed in a separated state. FIG. 6(b) is a photograph 1 minute after the nanofiber film was added to the container. As shown in FIG. 6(b), it is observed that the nanofiber film floated on the aqueous methylene blue solution so that only the salad oil (OL) remained in the nanofiber film. FIG. 6(c) is a photograph 10 minutes after the nanofiber film was added. As shown in the photograph of FIG. 6(c), it is observed that the nanofiber film absorbed only the salad oil within 10 minutes, and absorbed all of the salad oil in the film, at the end. In addition, the nanofiber film did not absorb the aqueous methylene blue solution at all. That is, it was found that polyhydroxyalkanoic acid nanofiber film has a function of separating water and oil simultaneously with a function of selectively absorbing only oil.
<Example 5> Organic Solvent Absorbency
[0056] FIG. 7 is a drawing representing organic solvent absorbency of the nanofiber film of Example 1. FIG. 7(a) is a photograph before the nanofiber film obtained in Example 1 was added to the container having an aqueous methylene blue solution (MB) and hexane (HX). Here, the aqueous methylene blue solution (MB) and hexane (HX) were separated into two layers. FIG. 7(b) is a photograph 10 minutes after the nanofiber film was added to the container. As shown in the photograph of the drawing, it is observed that hexane (HX) was all absorbed in the nanofiber film within 10 minutes. Since the amount of the aqueous methylene blue solution (MB) was not changed, it was found that the nanofiber film selectively absorbed only hexane of the organic solvent and did not absorb water. That is, it was found that the nanofiber film represents excellent organic solvent absorbency.
<Example 6> Nanofiber Film (Nanofiber Structure) Partially Having a Film Shape
[0057] FIG. 8 is an electron microscope photograph (SEM photograph) of the nanofiber film (nanofiber structure) in which the nanofiber film is partially fused to have a film shape. As shown in the drawing, it is observed that there is a film shape in the front side and a nanofiber film in the inside. This film shaped part is useful for improving strength of the film itself.
<Example 7> Adsorbent Material-Containing Nanofiber Film
[0058] FIG. 9 is an electron microscope photograph (SEM photograph) of the nanofiber film including fine particles as an adsorbent material. As shown in the drawing, it is observed that activated carbon fine particles AC1 and AC2 as the adsorbent material are entangled with the nanofiber FBR1 and FBR2 and maintained. In addition, the activated carbon fine particles may be in the nanofiber or on the surface of the nanofiber. The adsorbent material effectively absorbs the components dissolved in the organic solvent (impurities or components to be separated) passing between the nanofiber films. As an adsorbent, for example, activated carbon, zeolite, or the like can be selected depending on the use.
[0059] Finally, the advantages of the nanofiber film (nanofiber structure and the like) according to each Example of the present invention are indicated. Biodegradable polyhydroxyalkanoic acid (PHA) which is a raw material of the nanofiber film can be produced using a plant component of nature as a raw material. It is possible to suppress an increase in carbon dioxide gas by using the biodegradable polyhydroxyalkanoic acid to manufacture the nanofiber structure and widely use it for a non-woven fabric.
[0060] The polypropylene non-woven fabric which is the conventional product is flexible and strong and has good adhesion with other materials, and thus, has been used for various uses. In particular, the polypropylene non-woven fabric has been used as an oil adsorbent material since the polypropylene non-woven fabric absorbs oil. It was found by an experiment that the polyhydroxyalkanoic acid or polyhydroxybutyric acid which is a material of the nanofiber structure according to an exemplary embodiment of the present invention absorbs an organic solvent and toxic organic compounds soluble in the solvent as well as oil.
[0061] For example, when ocean, river, lake, groundwater, or the like contaminated with an organic solvent and an organic compound dissolved in the organic solvent was passed through the nanofiber structure according to an exemplary embodiment of the present invention, using these characteristics, contaminated goods can be filtered and absorbed to make clean water.
[0062] As described above, the nanofiber structure (nanofiber film) according to the present invention is expected to be used for various purposes mainly as a non-woven fabric.
REFERENCE SIGNS LIST
[0063] CNT: Container [0064] HPS: High voltage power supply [0065] NZL: Nozzle [0066] SL: Sample solution [0067] TS: Target substrate [0068] ESD: Electrospray deposition device [0069] WL: Wire
* * * * *
References
D00000

D00001

D00002
D00003

D00004

D00005
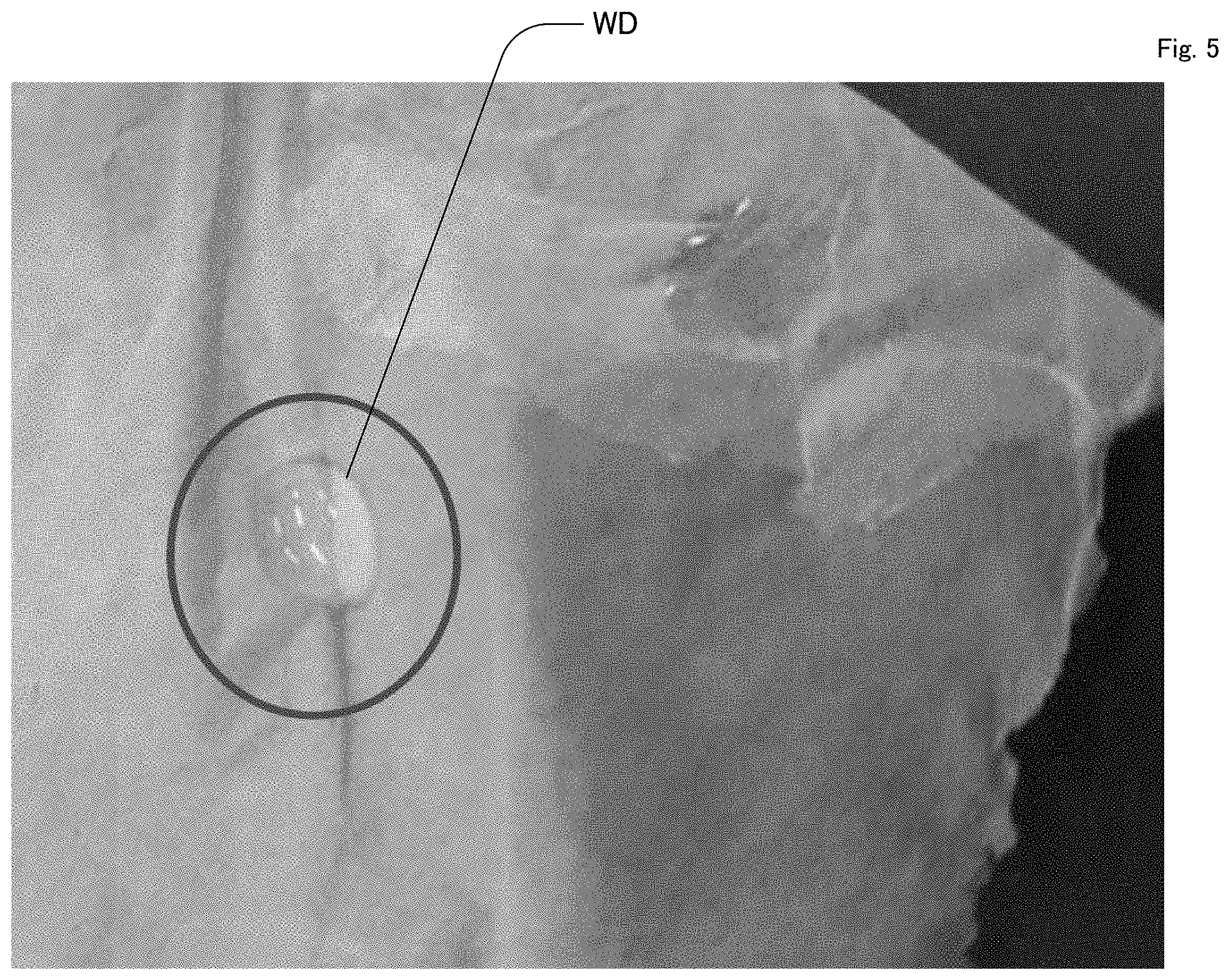
D00006

D00007

D00008

D00009

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.