Bis-primary Amines As Cross-linkers For The Curing Of Acetoacetylated Resin Compositions
Collins; Nick Allen ; et al.
U.S. patent application number 16/705394 was filed with the patent office on 2020-06-11 for bis-primary amines as cross-linkers for the curing of acetoacetylated resin compositions. This patent application is currently assigned to Eastman Chemical Company. The applicant listed for this patent is Eastman Chemical Company. Invention is credited to Nick Allen Collins, Leslie Shane Moody, Robert Jacks Sharpe.
| Application Number | 20200181338 16/705394 |
| Document ID | / |
| Family ID | 69006074 |
| Filed Date | 2020-06-11 |
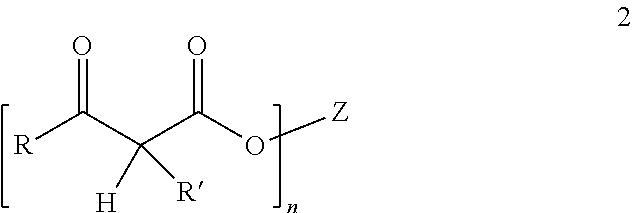
| United States Patent Application | 20200181338 |
| Kind Code | A1 |
| Collins; Nick Allen ; et al. | June 11, 2020 |
BIS-PRIMARY AMINES AS CROSS-LINKERS FOR THE CURING OF ACETOACETYLATED RESIN COMPOSITIONS
Abstract
This invention provides a composition comprising I. a first component comprising at least one resin having two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and II. a second component having at least one or at least two primary amine functional groups.
| Inventors: | Collins; Nick Allen; (Fall Branch, TN) ; Sharpe; Robert Jacks; (Johnson City, TN) ; Moody; Leslie Shane; (Johnson City, TN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Eastman Chemical Company Kingsport TN |
||||||||||
| Family ID: | 69006074 | ||||||||||
| Appl. No.: | 16/705394 | ||||||||||
| Filed: | December 6, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62777876 | Dec 11, 2018 | |||
| 62777865 | Dec 11, 2018 | |||
| 62777871 | Dec 11, 2018 | |||
| 62900939 | Sep 16, 2019 | |||
| 62916503 | Oct 17, 2019 | |||
| 62777880 | Dec 11, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C08J 2323/06 20130101; C09D 175/08 20130101; C08G 63/005 20130101; C08K 5/18 20130101; C08J 5/18 20130101; C09J 167/03 20130101; C08G 63/87 20130101; C09J 167/02 20130101; C09D 167/02 20130101; C08J 7/043 20200101; C08K 5/0066 20130101; C09D 161/20 20130101; C08J 2367/02 20130101; C08K 5/07 20130101; C09J 2461/00 20130101; C08G 63/12 20130101; C08K 3/013 20180101; C09D 7/65 20180101; C09D 167/00 20130101; C08J 2323/12 20130101; C09J 171/00 20130101; C09D 7/20 20180101; C08G 12/00 20130101; C08K 5/3462 20130101; C08J 3/24 20130101; C08K 5/0016 20130101; C08K 5/0025 20130101; C09J 11/08 20130101; C08G 63/914 20130101; C08G 63/916 20130101; C09J 5/00 20130101; C08L 67/03 20130101; C08K 5/005 20130101; C09J 161/20 20130101; C09J 2467/00 20130101 |
| International Class: | C08J 3/24 20060101 C08J003/24; C08G 63/91 20060101 C08G063/91; C09J 167/02 20060101 C09J167/02; C09D 167/02 20060101 C09D167/02; C09J 5/00 20060101 C09J005/00 |
Claims
1. A composition comprising: I. a first component comprising at least one amorphous or semi-crystalline polyester or polyether having two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and II. a second component having at least one or at least two primary amine functional groups.
2. The composition of claim 1, wherein said polyester is an acetoacetate functional polyester comprising the residues of a. a hydroxyl component comprising: i. a diol in an amount ranging from 60 to 100 mole %, based on the total moles of (i) and (ii) equaling 100 mole %; and ii. a polyol in an amount ranging from 0 to 40 mole %, based on the total moles of (i) and (ii) equaling 100 mole %; b. a carboxyl component comprising a polycarboxylic acid compound, a derivative of polycarboxylic acid compound, or a combination thereof; and c. an alkyl acetoacetate, a diketene, or a combination thereof in an amount ranging from about 5 to about 65 weight %, based on the total weight of (a), (b), and (c) equaling 100 weight %.
3. The composition of claim 2, wherein the diol component (a)(i) is selected from the group consisting of 2,2-dimethyl-1,3-propanediol, 2-methyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 1,4-butanediol, 1,6-hexanediol, 1,2-cyclohexanedimethanol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 2,2,4-trimethyl-1,3-pentanediol, hydroxypivalyl hydroxypivalate, tricyclodecanedimethanol, 2,2,4,4-tetramethylcyclobutane-1,3-diol, and polyethylene glycol, or mixtures thereof.
4. The composition of claim 2, wherein the polyol component (a)(ii) is selected from 1,1,1-trimethylol propane, 1,1,1-trimethylolethane, glycerin, sorbitol, and pentaerythritol or mixtures thereof.
5. The composition of claim 2, wherein said carboxyl component (b) is selected from the group consisting of adipic acid, isophthalic acid, dimethyl isophthalate, terephthalic acid, dimethyl terephthalate, 1,4-cyclohexanedicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, tetrahydrophthalic acid or tetrahydrophthalic anhydride, hexahydrophthalic acid or hexahydrophthalic anhydride, maleic anhydride, succinic anhydride, phthalic acid, and phthalic anhydride, or mixtures thereof.
6. The composition of claim 1 wherein said alkyl acetoacetate is selected from t-butyl acetoacetate, methyl acetoacetate, ethyl acetoacetate, n-propyl acetoacetate, isopropyl acetoacetate, and n-butyl acetoacetate or mixtures thereof.
7. The composition of claim 1 wherein said polyester has a glass transition temperature (Tg) of about -75.degree. C. to about 50.degree. C.
8. The composition of claim 1 containing essentially no volatile solvents.
9. The composition of claim 1 comprising one or more organic solvents.
10. The composition of claim 1 wherein at least one said amine is selected from aliphatic, cycloaliphatic, aromatic-aliphatic, or aromatic primary amines.
11. The composition of claim 1 wherein said at least one primary amine is selected from C1-C20 alkyl or aryl amines or a mixture thereof.
12. The composition of claim 1 wherein at least one primary amine is selected from at least one of: ethylenediamine; diethylenetriamine; triethylenetetramine; tetraethylenepentamine; 2,2,4-trimethylhexamethylenediamine; 2,4,4-trimethylhexamethylenediamine; 1,2-diaminopropane; 1,3-diaminopropane; 2,2-dimethylpropylenediamine; 1,4-diaminobutane; 2-methylpentamethylenediamine; 1,6-hexanediamine; 1,7-diaminoheptane; 1,8-diaminooctane; 1,9-diaminononane; 1,12-diaminododecane; 4-azaheptamethylenediamine; N,N-bis(3-aminopropyl)butane-1,4-diamine; N,N-bis(3-aminopropyl)ethylenediamine; 2,4-toluenediamine; 2,6-toluenediamine; 3,3'-iminobispropylamine; 1,2-diaminocyclohexane; 1,3-diaminocyclohexane; 1,4-diamino-2,5-diethylcyclohexane; 1,4-diamino-3,6-diethylcyclohexane; 1,2-diamino-4-ethylcyclohexane; 1,2-diamino-4-cyclohexylcyclohexane; isophorone diamine; norbornanediamine; 4,4'-diaminodicyclohexylmethane; 4,4'-diaminodicyclohexylethane; 4,4'-diaminodicyclohexylpropane; 2,2-bis(4-aminocyclohexyl)propane; 3,3'-dimethyl-4,4'-diaminodicyclohexylmethane; 3-amino-1-(4-aminocyclohexyl)propane; 1,3-bis(aminomethyl)cyclohexane;1,4-bis(aminomethyl)cyclohexane; 1-cyclohexyl-3,4-diamino-cyclohexane; m-xylylenediamine and its hydrogenation products; p-xylylenediamine and its hydrogenation products; 4,4'-methylenedianiline; 2,4-bis(p-aminobenzyl)aniline; diethyltoluenediamine; m-phenylenediamine, diphenylethylenediamine, 2,2,4,4-tetramethylcyclobutane-1,3-diamine, 2,2-dimethylpropane-1,3-diamine, 2,3-dimethylbutane-2,3-diamine, 1,2-diaminocyclopentane, 1,2,2-trimethylcyclopentane-1,3-diamine, 1,7,7-trimethylbicyclo[2.2.1]heptane-2,3-diamine, 4,4'-methylenebis[2-methylcyclohexanamine], and polyetheramines.
13. The composition of claim 1 wherein said amine is selected from the group consisting of triethylenetetramine, 1,6-hexanediamine, 2-methylpentamethylenediamine, isophorone diamine, 4,4'-methylenebis[2-methylcyclohexanamine], and polyetheramines.
14. The composition of claim 1 wherein the equivalent ratio of the acetoacetate (AcAc) functionality in the resin to the primary amine functionality in the composition is from about 0.25 about 4.
15. The composition of claim 1 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more .beta.-ketoester functional groups, and at least one component having two or more primary amine functional groups, and (b) after optional activation, curing the composition at 20.degree. C. or higher in six days or less, and (c) optionally post-curing the composition at or above 20.degree. C.
16. A composition according to claim 1 comprising at least one additive selected from the group consisting of tackifiers, plasticizers, fillers, pigments, stabilizers, antioxidants, waxes, adhesion promoters, flame retardants, conductive agents, rheology modifiers or mixtures thereof.
17. An adhesive, coating, or article of manufacture comprising the composition of claim 1.
18. An article comprising a cured adhesive composition of claim 1 where the bond is maintained after cure and/or post-cure at 60.degree. C.
19. An article comprising said composition of claim 1 selected from at least one of the following: an adhesive, a laminate, a tape, a label, a tag, a radio frequency identification (RFID) tag, a coating, a sealant, a flexible or non-flexible film, a foam, a potting compound, a disposable hygiene article, a fiberglass reinforced plastic, a composite, a compound, an assembly part, a motor vehicle molded part, a motor vehicle extruded part, a motor vehicle laminated part, a furniture part, fabric, a woven textile, a nonwoven, a flexible packaging multilayer; wherein the adhesive is selected from at least one of the following: automotive interior adhesive, flexible laminating adhesive, rigid laminating adhesive, assembly adhesive, labelling adhesive, nonwoven adhesive, tape adhesive, structural adhesive, hygiene nonwoven construction adhesive, hygiene elastic attachment adhesive, home repair adhesive, industrial adhesive, construction adhesive, furniture adhesive, medical adhesive, contact adhesive, hot melt adhesive, solvent-based adhesive, packaging adhesive, product assembly adhesive, woodworking adhesive, flooring adhesive, automotive assembly adhesive, assembly part adhesive, structural adhesive, pressure sensitive adhesive, pressure sensitive adhesive tape, pressure sensitive adhesive label, pressure sensitive adhesive protective film, laminating adhesive, packaging adhesive, flexible film adhesive, flexible packaging adhesive, hygiene core integrity adhesive, hygiene core integrity adhesive, and solvent-based adhesives; wherein the composite is selected from at least one of a polyester composite, a glass composite, or a wood-plastic composite; and wherein said compound is selected from at least one of an extruded compound, a polyacrylic blended compound, a potting compound, a rubber compound, a sheet molding compound or a dough molding compound.
20. The process of applying the adhesive of claim 1 to a substrate and subsequently laminating to another substrate.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application Nos. 62/777,876, 62/777,865, 62/777,871, 62/777,880 all filed Dec. 11, 2018; 62/900,939 filed Sep. 16, 2019; and 62/916,503 filed Oct. 17, 2019 under 35 U.S.C. .sctn. 119(e)(1); the entire contents of the provisional applications are hereby incorporated by reference.
FIELD OF THE INVENTION
[0002] The present invention relates to polyester compositions having beta-ketoester group(s) arid primary amines, having improved properties.
BACKGROUND OF THE INVENTION
[0003] Reactive compositions based on isocyanate crosslinkers are widely used for coating and adhesive applications. Such systems are curable at room temperature or low temperatures (e.g. <100.degree. C.) and are capable of providing the desirable properties for a variety of applications. However, there have been increasing concerns associated with the production and the use of isocyanate compounds and formulations based on isocyanates.
[0004] Compositions based on isocyanate crosslinkers frequently require reaction with moisture to form the crosslinked composition. If a solvent-based system is used due to application equipment or other reasons, it is desirable that trace moisture in the solvent will not initiate curing or crosslinking of the curable composition and thus will not reduce the pot life of the curable composition.
[0005] Commercially, it is desirable that the system does not: generate by-products during curing that can be detrimental to film formation or quality, have volatility, cause defects, have adhesion failure, and/or have unpleasant odor.
[0006] In addition, there is a need in commerce for a system that has a combination of two or more of the following properties: isocyanate free, curable at low temperatures, and good adhesion to substrates typically bonded or coated with isocyanate-containing systems. It is also desirable to have a curable system that is applied and cured below about 100.degree. C. while providing suitable adhesion at service temperatures up to 130.degree. C.
[0007] Further, there is a need in the art for a commercial adhesive or coating composition that has the desired level of adhesion to the substrate to which it is applied and that also adheres to many different substrates.
SUMMARY OF THE INVENTION
[0008] In view of the above commercial shortcomings in the art, the present disclosure addresses the need for properties in curable compositions including one or more of the following properties: (1) a system that cures and that is isocyanate free, (2) curable at low temperatures (e.g. <100.degree. C.), (3) suitable adhesion at service temperatures up to 130.degree. C., (4) good adhesion to substrates typically bonded or coated with isocyanate-containing systems, (5) no Volatile Components, e.g., Volatile Organic Components (VOCs), or has low Volatile Components, e.g., VOCs, released before, during, and/or after curing, (6) does not generate by-products during curing that can be detrimental to film formation or bond quality, (7) reduces defects, (8) has more pleasant odor, less odor, or is odorless, (9) can contain low or no moisture, (10) can be used with moisture-impermeable substrates such as artificial leather, (11) under certain conditions, can be a crosslinking system, (12) can provide more controlled curing, (13) good thermal stability and/or UV stability, (14) good chemical resistance, (15) good mechanical properties, for example, tensile strength, elongation at break, impact strength, and/or Young's modulus, (16) good aging resistance or stability, (17) can provide improved green strength, (18) can provide a usable working life for many applications and/or (19) can provide longer pot life [in some instances, the pot life can be extended by controlling the reaction of the components (controlled cure)].
[0009] The compositions of this invention can provide desirable properties for a variety of applications. In certain embodiments, the compositions of this invention are suitable for applications in the adhesives area, for example, automotive adhesives, structural adhesives, wood adhesives, and laminating adhesives, and applications in the coatings area, for example, automotive, industrial maintenance, marine craft, field-applied coatings, and furniture.
[0010] For the ease of reference but not intending to be limiting in any way, certain aspects of this disclosure are numbered consecutively, as follows:
[0011] In aspect 1 of this invention, there is provided a composition comprising: Component (I) having at least one or at least two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups; and Component (II) having at least one or at least two primary amine functional groups.
[0012] In aspect 2 of the invention, there is provided the composition of aspect 1 comprising: (I) a first component [Component (I)] comprising at least one amorphous or semi-crystalline polyester or at least one polyether having at least one or at least two functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and (II) a second component [Component (II)] comprising at least one or at least two primary amine functional groups.
[0013] In aspect 3 of this invention, there is provided a composition of aspects 1 or 2, wherein the polyester is an acetoacetate functional polyester comprising the residues of
[0014] a. a hydroxyl component comprising: [0015] i. at least one diol in an amount ranging from 50 to 100 mole %, based on the total moles of (i) and (ii) equaling 100 mole %; and [0016] ii. at least one polyol in an amount ranging from 0 to 50 mole %, based on the total moles of (i) and (ii) equaling 100 mole %;
[0017] b. at least one carboxyl component comprising a polycarboxylic acid compound, a derivative of a polycarboxylic acid compound, or a combination thereof; and
[0018] c. at least one compound selected from an alkyl acetoacetate, a diketene, or a combination thereof.
[0019] In aspect 4 of this invention, there is provided the composition of aspect 2, wherein the residues of alkyl acetoacetate or diketene are present in an amount ranging from about 5 to about 50 weight %, or from about 5 to about 40 weight %, or from about 10 to about 30 weight %, based on the total weight percentages of (a), (b), and (c) equaling 100 weight %.
[0020] In aspect 5 of this invention, there is provided the composition of aspect 4, wherein the residues of alkyl acetoacetate or diketene is present in an amount ranging from about 15 to about 30 weight %.
[0021] In aspect 6 of this invention, there is provided a composition of any one of aspects 3-5, wherein the diol component (a)(i) is present in an amount ranging from about 35 to about 100, or from about 30 to about 95, or from about 50 to about 95, or from about 70 to 90, or from about 85 to about 95 mole % and the polyol component (a)(ii) is present in an amount ranging from about 0 to about 65, or from about 5 to about 70, or from about 5 to about 50, or from about 10 to about 30, or from about 5 to about 15 mole %, wherein the total mole % of (a)(i) and (a)(ii) equals 100 mole %.
[0022] In aspect 7 of this invention, there is provided a composition of any one of aspects 3-6, wherein the polyester is an acetoacetate functional polyester comprising the residues of
[0023] a. a hydroxyl component comprising: [0024] i. at least one diol in an amount ranging from 85 to 95 mole %, based on the total moles of (i) and (ii); and [0025] ii. a polyol in an amount ranging from 5 to 15 mole %, based on the total moles of (i) and (ii);
[0026] b. at least one carboxyl component comprising a polycarboxylic acid compound, a derivative of polycarboxylic acid compound, or a combination thereof; and
[0027] c. at least one compound selected from an alkyl acetoacetate, a diketene, or a combination thereof in an amount ranging from about 5 to about 50 weight %, based on the total weight of (a), (b), and (c).
[0028] In aspect 8 of this invention, there is provided a composition of any one of aspects 3-7, wherein the diol component (a)(i) is selected from the group consisting of 2,2-dimethyl-1,3-propanediol (neopentyl glycol), 2-methyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 1,4-butanediol, 1,6-hexanediol, 1,2-cyclohexanedimethanol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 2,2,4-trimethyl-1,3-pentanediol, hydroxypivalyl hydroxypivalate, 2,2,4,4-tetramethylcyclobutane-1,3-diol, tricyclodecanedimethanol, and polyethylene glycol, or mixtures thereof.
[0029] In aspect 9 of this invention, there is provided a composition of any of aspects 3-8 wherein diol component (a)(i) contains only oxygen, carbon, and hydrogen.
[0030] In aspect 10 of this invention, there is provided a composition of any of aspects 3-9 wherein diol component (a)(i) does not have any carbonyl groups (--CO--).
[0031] In aspect 11 of this invention, there is provided a composition of any of aspects 3-10 wherein diol component (a)(i) does not contain ether groups.
[0032] In aspect 12 of this invention, there is provided a composition of any one of aspects 3-11, wherein at least one polyol component (a)(ii) is selected from 1,1,1-trimethylol propane, 1,1,1-trimethylolethane, glycerin, sorbitol, and pentaerythritol or mixtures thereof.
[0033] In aspect 13 of this invention, there is provided a composition of any one of aspects 3-12, wherein at least one carboxyl component (b) or all of carboxyl component (b) is selected from the group consisting of adipic acid, isophthalic acid (or dimethyl isophthalate), terephthalic acid, 1,4-cyclohexanedicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, tetrahydrophthalic acid or anhydride, hexahydrophthalic acid or anhydride, maleic anhydride, succinic anhydride, phthalic acid, and phthalic anhydride, or mixtures thereof.
[0034] In aspect 14 of this invention, there is provided a composition of any one of aspects 3-13, wherein said alkyl acetoacetate is selected from t-butyl acetoacetate, methyl acetoacetate, ethyl acetoacetate, n-propyl acetoacetate, isopropyl acetoacetate, n-butyl acetoacetate, isobutyl acetoacetate, t-amyl acetoacetate, diisopropyl carbinol acetoacetate or mixtures thereof.
[0035] In aspect 15 of this invention, there is provided a composition of any one of aspects 1-14, wherein said polyester has a glass transition temperature (Tg) of about -85.degree. C. to about 60.degree. C.; or about -85.degree. C. to about 50.degree. C.; or about to about 40.degree. C.; or about -85.degree. C. to about 30.degree. C.; or about -85.degree. C. to about 20.degree. C.; or about -85.degree. C. to about 10.degree. C.; or about -85.degree. C. to about 0.degree. C.; or about -85.degree. C. to about -10.degree. C.; or about -85.degree. C. to about -20.degree. C.; or about -85.degree. C. to about -25.degree. C.; or about -85.degree. C. to about 30.degree. C.; or about -85.degree. C. to about 60.degree. C.; or about -85.degree. C. to about 50.degree. C.; or about -85.degree. C. to about 40.degree. C.; or about -85.degree. C. to about 30.degree. C.; or about -85.degree. C. to about 20.degree. C.; or about -85.degree. C. to about 10.degree. C.; or about -85.degree. C. to about 0.degree. C.; or about -85.degree. C. to about -10.degree. C.; or about -85.degree. C. to about -20.degree. C.; or about -85.degree. C. to about -25.degree. C.; or about -85.degree. C. to about -30.degree. C.; or about to about 60.degree. C.; or about -70.degree. C. to about 50.degree. C.; or about -70.degree. C. to about 40.degree. C.; or about -70.degree. C. to about 30.degree. C.; or about -70.degree. C. to about 20.degree. C.; or about -70.degree. C. to about 10.degree. C.; or about -70.degree. C. to about 0.degree. C.; or about -70.degree. C. to about -10.degree. C.; or about -70.degree. C. to about -20.degree. C.; or about -70.degree. C. to about -25.degree. C.; or about -70.degree. C. to about -30.degree. C.; or about -65.degree. C. to about 60.degree. C.; or about -65.degree. C. to about 50.degree. C.; or about -65.degree. C. to about 40.degree. C.; or about -65.degree. C. to about 30.degree. C.; or about -65.degree. C. to about 25.degree. C.; or about -65.degree. C. to about 20.degree. C. or about -65.degree. C. to about 10.degree. C.; or about -65.degree. C. to about 0.degree. C.; or about -65.degree. C. to about -10.degree. C.; or about -65.degree. C. to about -20.degree. C.; or about -65.degree. C. to about -25.degree. C.; or about -65.degree. C. to about -30.degree. C.; or about -45.degree. C. to about 60.degree. C.; or about -45.degree. C. to about 50.degree. C.; or about -45.degree. C. to about 40.degree. C.; or about -45.degree. C. to about 30.degree. C.; or about -45.degree. C. to about 25.degree. C.; or about -45.degree. C. to about 20.degree. C. or about -45.degree. C. to about 10.degree. C.; or about -45.degree. C. to about 0.degree. C.; or about -45.degree. C. to about -10.degree. C.; or about -45.degree. C. to about -20.degree. C.; or about -45.degree. C. to about -25.degree. C.; or about -45.degree. C. to about -30.degree. C. As used herein, Tg and/or melting point is determined using a Q2000 differential scanning calorimeter (DSC) from TA Instruments, New Castle, Del., US, at a scan rate of 20.degree. C./min.
[0036] In aspect 16 of this invention, there is provided a composition of any one of aspects 1-15 wherein said polyester has a Tg of from about -85.degree. C. to about -10.degree. C.; or about -70.degree. C. to about 50.degree. C.; or from about -65 to about 40.degree. C.; or from about -65.degree. C. to about 25.degree. C.; or from about -10.degree. C. to about 50.degree. C.; or from about -45.degree. C. to about -10.degree. C.; or from about -45.degree. C. to about -25.degree. C.
[0037] In aspect 17 of this invention, there is provided a composition of any one of aspects 1-16 which upon curing releases essentially no volatile organic compounds; or no VOCs; or less than 5 weight %, less than 4 weight %, less than 3 weight %, less than 2 weight %, or less than 1 weight % VOCs; based on the total weight of the composition.
[0038] In aspect 18 of this invention, there is provided a composition of any one of aspects 1-17 where the curing agent comprises an aliphatic, cycloaliphatic, or aromatic, di-, or poly-amine or mixtures thereof.
[0039] In aspect 19 of this invention, there is provided a composition of any one of aspects 1-18 wherein the primary amine can be selected from at least one of: ethylenediamine; diethylenetriamine; triethylenetetramine (TETA) or isomers thereof; tetraethylenepentamine or isomers thereof; 2,2,4-trimethylhexamethylenediamine; 2,4,4-trimethylhexamethylenediamine; 1,2-diaminopropane; 1,3-diaminopropane; 2,2-dimethylpropylenediamine; 1,4-diaminobutane; 2-methylpentamethylenediamine (MPMDA); 1,6-hexanediamine; 1,7-diaminoheptane; 1,8-diaminooctane; 1,9-diaminononane; 1,12-diaminododecane; 4-azaheptamethylenediamine; N,N-bis(3-aminopropyl)butane-1,4-diamine; N,N-bis(3-aminopropyl)ethylenediamine; 2,4-toluenediarnine; 2,6-toluenediarnine; 3,3'-irninobispropylamine; 1,2-diaminocyclohexane; 1,3-diaminocyclohexane; 1,4-diamino-2,5-diethylcyclohexane; 1,4-diamino-3,6-diethylcyclohexane; 1,2-diamino-4-ethylcyclohexane; 1,2-diamino-4-cyclohexylcyclohexane; isophorone diamine (IPDA); norbornanediamine; 4,4'-diaminodicyclohexylmethane; 4,4'-diaminodicyclohexylethane; 4,4'-diaminodicyclohexylpropane; 2,2-bis(4-aminocyclohexyl)propane; 3,3'-dimethyl-4,4'-diaminodicyclohexylmethane; 3-amino-1-(4-aminocyclohexyl)propane; 1,3-bis(aminomethyl)cyclohexane; 1,4-bis(aminomethyl)cyclohexane; 1-cyclohexyl-3,4-diamino-cyclohexane; xylylenediamine and its hydrogenation products; p-xylylenediamine and its hydrogenation products; 4,4'-methylenedianiline; 2,4-bis(p-aminobenzyl)aniline; diethyltoluenediamine; m-phenylenediamine; diphenylethylenediamine; 2,2,4,4-tetramethylcyclobutane-1,3-diamine; 2,2-dimethylpropane-1,3-diamine; 2,3-dimethylbutane-2,3-diamine; 1,2-diaminocyclopentane; 1,2,2-trimethylcyclopentane-1,3-diamine; 1,7,7-trimethylbicyclo[2.2.1]heptane-2,3-diamine; 4,4'-methylenebis[2-methylcyclohexanamine] (MACM); and polyetheramines such as JEFFAMINE.RTM. D-230, D-400, D-2000, and T-403 amines.
[0040] In aspect 20 of this invention, there is provided a composition of any of aspects 1-19 wherein the amines can be employed as their salts, including but not limited to their carboxylate, phosphate, sulfate, and/or hydrochloride salts. The salt of the amine can be prepared in situ by addition of the corresponding acid and amine and/or it can be prepared separately by prereacting amine and acid.
[0041] In aspect 21 of this invention, there is provided a composition of any one of aspects 1-20 further comprising an adduct having two or more .beta.-ketoacetate functional groups selected from 2-methyl-1,3-propanediol diacetoacetate; neopentyl glycol diacetoacetate; 2,2,4,4-tetramethylcyclobutane-1,3-diol diacetoacetate; or trimethylolpropane triacetoacetate.
[0042] In aspect 22 of this invention, there is provided a composition of any one of aspects 1-21 wherein the equivalent ratio of the acetoacetate (AcAc) functional group of Component (I) to the primary amine functional group in the composition [Component (II)] is from about 0.25 about 4, or from about 0.5 to about 2, or from about 0.7 to about 1.3, or from about 0.8 to about 1.2.
[0043] In aspect 23 of this invention, there is provided a curable composition of aspects 1-22 wherein at least one wax or at least one tackifies is pre-combined with at least one curing agent in order to provide controlled cure profile (where the curing process is controlled).
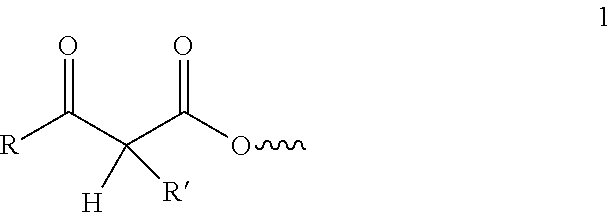
[0044] In aspect 24 of this invention, there is provided a composition of any one of aspects 1-23 comprising at least one additive selected from the group consisting of tackifiers, plasticizers, fillers, pigments, stabilizers, antioxidants, adhesion promoters, flame retardants, conductive agents, rheology modifiers and mixtures thereof.
[0045] In aspect 25 of this invention, there is provided a composition of any one of aspects 1-24 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more .beta.-ketoester functional groups, and at least one component having two or more primary amine functional groups, and (b) after optional activation, curing the composition at 20.degree. C. or higher in six days or less, and (c) optionally, post-curing the composition at or above 20.degree. C.
[0046] In aspect 26 of this invention, there is provided a composition of any one of aspect 1-25, wherein the composition is solventless or solvent free.
[0047] In aspect 27 of this invention, there is provided the composition of any of aspects 1-26 wherein the composition is substantially free of solvent or comprises essentially no solvent.
[0048] In aspect 28 of this invention, there is provided a composition of any one of aspects 1-25 comprising at least one solvent.
[0049] In aspect 29 of this invention, there is provided the composition of aspect 28 wherein the solvent borne composition comprises organic solvents.
[0050] In aspect 30 of this invention, there is provided a composition of aspect 29 comprising one or more organic solvents selected from the group comprising acetone, ethanol, ethyl acetate, butyl acetate, xylene, methyl amyl ketone, methyl ethyl ketone, arid toluene.
[0051] In aspect 31 of this invention, there is provided the composition of any of aspects 28-30 which does not contain solvents or residues of solvents such as ethylene glycol and/or ether-containing compounds, for example, dimethyl ether, ethylene glycol diethyl ether, diethylene glycol dimethyl ether, or diethylene glycol diethyl ether.
[0052] In aspect 32 of this invention, there is provided the composition of any of aspects 1-31 wherein the composition can be water-free, and/or moisture-free, and/or can comprise substantially no water, and/or can contain essentially no water, or can contain one of the following weight percentages of water: 0 to 20 weight %, or 0 to 19 weight %, or 0 to 18 weight %, or 0 to 17 weight %, or 0 to 16 weight %, or 0 to 15 weight %, or 0 to 14 weight %, or 0 to 13 weight %, or 0 to 12 weight %, or 0 to 11 weight %, or 0 to 10 weight %, or 0 to 9 weight %, or 0 to 8 weight %, or 0 to 7 weight %, or 0 to 6 weight %, or 0 to 5 weight %, or 0 to 4 weight %, or 0 to 3 weight %, or 0 to 2 weight %, or 0 to 1 weight %, or 0.01 to 20 weight %, or 0.01 to 19 weight %, or 0.01 to 18 weight %, or 0.01 to 17 weight %, or 0.01 to 16 weight %, or 0.01 to 15 weight %, or 0.01 to 14 weight %, or 0.01 to 13 weight %, or 0.01 to 12 weight %, or 0.01 to 11 weight %, or 0.01 to 10 weight %, or 0.01 to 9 weight %, or 0.01 to 8 weight %, or 0.01 to 7 weight %, or 0.01 to 6 weight %, or 0.01 to 5 weight %, or 0.01 to 4 weight %, or 0.01 to 3 weight %, or 0.01 to 2 weight %, or 0.01 to 1 weight %, based on the total weight of the composition.
[0053] In aspect 33 of this invention, there is provided the composition of any of aspects 1-32 wherein the composition is not a water-borne or a water-dispersible composition.
[0054] In aspect 34 of this invention, there is provided the composition of any of aspects 1-33 wherein the composition does not contain: a polyvinyl alcohol, or a polyvinyl polymer, or a poly vinyl acetate polymer.
[0055] In aspect 35 of this invention, there is provided the composition of any of aspects 1-34 wherein the composition does not contain neutralizing agents.
[0056] In aspect 36 of this invention, there is provided the composition of any of aspects 1-35 wherein the composition of the invention or the polyester resin of the invention has an acid number (AN) of 0 to 15, 0 to 10, less than 10, 0 to 5, or less than 5 mg KOH/g with and/or without neutralizing agents.
[0057] In aspect 37, there is provided the composition of any of aspects 1-36 wherein the acid number of the compositions of the invention or of the polyester resin of the invention can be from 0 to about 14, or from 0 to about 13, or from 0 to about 12, or from 0 to about 11, or from 0 to about 10, or from 0 to about 9, or from 0 to about 8, or from 0 to about 7, or from 0 to about 6, or from 0 to 5 or from 0 to about 4, or from 0 to about 3, or from 0 to about 2, or from 0 to about 1, or from 0.01 to about 15, or from 0.01 to about 14, or from 0.01 to about 13, or from 0.01 to about 12, or from 0.01 to about 11, or from 0.01 to about 10, or from 0.01 to about 9, or from 0.01 to about 8, or from 0.01 to about 7, or from 0.01 to about 6, or from 0.01 to 5 or from 0.01 to about 4, or from 0.01 to about 3, or from 0.01 to about 2, or from 0.01 to about 1, or from 0.50 to about 15, or from 0.50 to about 14, or from 0.50 to about 13, or from 0.50 to about 12, or from 0.50 to about 11, or from 0.50 to about 10, or from 0.50 to about 9, or from 0.50 to about 8, or from 0.50 to about 7, or from 0.50 to about 6, or from 0.50 to about 5, or from 0.50 to about 4, or from 0.50 to about 3, or from 0.50 to about 2, or from 0.50 to about 1.5 mg KOH/g, with or without addition of neutralizing agents.
[0058] In aspect 38 of this invention, there is provided the composition of any of aspects 1-37 wherein the composition can have any one of the acid numbers of aspects 36 or 35, where neutralizing agents are not added to the composition after the polymerization reaction. In aspects 35-36, neutralizing agents can be any neutralizing agent known in the art. In the aspect of the invention where neutralizing agents are excluded from the scope of this invention, particularly where added after polymerization, neutralizing agents can include but are not limited to primary amines, secondary amines, tertiary amines, and ammonia as well as any other neutralizing agent known in the art, for example, for water dispersible or water dispersed microgel compositions.
[0059] In aspect 39 of this invention, there is provided the composition of any of aspects 1-38 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more primary amine functional groups, and at least one ketone, and (b) converting some or all of the amine functional groups to ketimine groups through reaction with the ketone, and (c) mixing this product with at least one component having two or more .beta.-ketoacetate functional groups.
[0060] In aspect 40 of this invention, there is provided the composition of aspect 39 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more primary amine functional groups, and at least one ketone, and (b) converting some or all of the amine functional groups to ketimine groups through reaction with the ketone, and (c) mixing this product with at least one component having two or more .beta.-ketoacetate functional groups, and (d) after optional activation with heat and/or moisture, curing the composition at 20.degree. C. or higher in six days or less, and (e) optionally, post-curing the composition at or above 20.degree. C.
[0061] In aspect 41 of this invention, there is provided the composition of any of aspects 1-38 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more primary amine functional groups, and at least one aldehyde, and (b) converting some or all of the amine functional groups to aldimine groups through reaction with the aldehyde, and (c) mixing this product with at least one component having two or more .beta.-ketoester functional groups.
[0062] In aspect 42 of this invention, there is provided the composition of aspect 41 wherein the composition is prepared by a process comprising: (a) mixing at least one component having two or more primary amine functional groups, and at least one aldehyde, and (b) converting some or all of the amine functional groups to aldimine groups through reaction with the aldehyde, and (c) mixing this product with at least one component having two or more .beta.-ketoester functional groups, and (d) after optional activation with heat and/or moisture, curing the composition at 20.degree. C. or higher in six days or less, and (e) optionally, post-curing the composition at or above 20.degree. C.
[0063] In aspect 43, there is provided the composition of any of aspects 41-42 wherein the composition can contain residues of aldimine that may be incorporated into the composition during curing.
[0064] In aspect 44 of this invention, there is provided the composition of any of aspects 1-43 wherein the polyester resin of the composition of the invention is not an alkyd polyester resin and/or is not a polyester resin containing alkyd residues and/or fatty acid residues.
[0065] In aspect 45 of this invention, there is provided the composition of any of aspects 1-44 wherein the composition of the invention is not a thermosetting composition.
[0066] In aspect 46 of this invention, there is provided the composition of any of aspects 1-45 wherein the composition of this invention does not contain polysaccharides and/or oxidized polysaccharides having aldehyde groups.
[0067] In aspect 47 of this invention, acrylates are not blended with the acetoacetylated polyester resins of any of aspects 1-46 of the invention.
[0068] In aspect 48 of this invention, acrylates are not included within the compositions and/or adhesives and/or articles of any of aspects 1-47 of the invention.
[0069] In aspect 49 of this invention, there is provided the composition of any of aspects 1-48 wherein the composition is curable (capable of curing).
[0070] In aspect 50 of this invention, there is provided the composition of any of aspects 1-49 wherein curing of the composition occurs.
[0071] In aspect 51 of this invention, there is provided the composition of any of aspects 1-50 that can be cured at low temperatures (e.g. <80.degree. C.).
[0072] In aspect 52 of this invention, there is provided the composition of any of aspects 1-51 that can be cured at room temperature, or 10.degree. C. to 30.degree. C. or to 130.degree. C., or 20.degree. C. to 120.degree. C. or 20.degree. C. to 115.degree. C., or 20.degree. C. to 110.degree. C., or 20.degree. C. to 105.degree. C., or 20.degree. C. to 100.degree. C., or 20.degree. C. to 90.degree. C., or 20.degree. C. to 80.degree. C., or 20.degree. C. to 70.degree. C., or 30.degree. C. to 130.degree. C., or 30.degree. C. to 120.degree. C., or 30.degree. C. to 115.degree. C., or 30.degree. C. to 110.degree. C., or 30.degree. C. to 105.degree. C., or 30.degree. C. to 100.degree. C., or 30.degree. C. to 90.degree. C., or 30.degree. C. to 80.degree. C., or 30.degree. C. to 70.degree. C., or 40.degree. C. to 130.degree. C., or 40.degree. C. to 120.degree. C., or 40.degree. C. to 115 .degree. C., or 40.degree. C. to 110.degree. C., or 40.degree. C. to 105.degree. C., or 40.degree. C. to 100.degree. C., or 40.degree. C. to 90.degree. C., or 40.degree. C. to 80.degree. C., or 40.degree. C. to 70.degree. C., 80.degree. C. to 130.degree. C. or 50.degree. C. to 130.degree. C. or 50.degree. C. to 120.degree. C. or 50.degree. C. to 115.degree. C., or 50.degree. C. to 110.degree. C., or 50.degree. C. to 105.degree. C., or 50.degree. C. to 100.degree. C., or 50.degree. C. to 90.degree. C., or 50.degree. C. to 80.degree. C., or 60.degree. C. to 130.degree. C., 60.degree. C. to 120.degree. C., or 60.degree. C. to 115.degree. C., or 60.degree. C. to 110.degree. C., or 60.degree. C. to 105.degree. C., or 60.degree. C. to 100.degree. C., or 60.degree. C. to 90.degree. C., or 60.degree. C. to 80.degree. C., or 70.degree. C. to 130 70.degree. C. to 120.degree. C., or 70.degree. C. to 115.degree. C., or 70.degree. C. to 110.degree. C., or 70.degree. C. to 105.degree. C., or 70.degree. C. to 100.degree. C. or 60.degree. C. to 90.degree. C., or 70.degree. C. to 80.degree. C. or 80.degree. C. to 130.degree. C., 80.degree. C. to 120.degree. C., or 80.degree. C. to 115.degree. C., or 80.degree. C. to 110.degree. C., or 80.degree. C. to 105.degree. C., or 80.degree. C. to 100.degree. C. or 80.degree. C. to 90.degree. C. or 90.degree. C. to 130.degree. C., 90.degree. C. to 120.degree. C., or 90.degree. C. to 115.degree. C., or 90.degree. C. to 110.degree. C. or 90.degree. C. to 105.degree. C., or 90.degree. C. to 100.degree. C., or 100.degree. C. to 130.degree. C., 100.degree. C. to 120.degree. C., or 100.degree. C. to 115.degree. C.
[0073] In aspect 53 of this invention, there is provided the composition of any of aspects 1-52, wherein, after curing, the compositions of the invention can have service temperatures of up to 130.degree. C., or up to 120.degree. C., or up to 115.degree. C., or up to 110.degree. C., or up to 105, or up to 100.degree. C., or up to 95.degree. C., or up to 90.degree. C., or up to 85.degree. C., or up to 80, or up to 75.degree. C.
[0074] In aspect 54 of this invention, there is provided adhesive compositions of any one of aspects 1-53 that can have a 180 degree peel strength of at least 2 lb/in, or at least 3 lb/in, or at least 4 lb/in, or at least 5 lb/in as measured according to ISO 8510-2-2006 Part 2 at 5 mm/sec.
[0075] In aspect 55 of this invention, there is provided the composition of any of aspects 1-54 that can be cured at room temperature, or 10.degree. C. to 30.degree. C., or 10.degree. C. to 130.degree. C., or 20.degree. C. to 120.degree. C., or 20.degree. C. to 115.degree. C., or 20.degree. C. to 110.degree. C., or 20.degree. C. to 105.degree. C., or 20.degree. C. to 100.degree. C., or 20.degree. C. to 90.degree. C., or 20.degree. C. to 80.degree. C., or 20.degree. C. to 70.degree. C., or 30.degree. C. to 130.degree. C., or 30.degree. C. to 120.degree. C., or 30.degree. C. to 115.degree. C., or 30.degree. C. to 110.degree. C., or 30.degree. C. to 105.degree. C., or 30.degree. C. to 100.degree. C., or 30.degree. C. to 90.degree. C., or 30.degree. C. to 80.degree. C., or 30.degree. C. to 70.degree. C., or 40.degree. C. to 130.degree. C., or 40.degree. C. to 120.degree. C., or 40.degree. C. to 115.degree. C., or to 110.degree. C. or 40.degree. C. to 105.degree. C., or 40.degree. C. to 100.degree. C., or 40.degree. C. to 90.degree. C., or 40.degree. C. to 80.degree. C., or 40.degree. C. to 70.degree. C., 80.degree. C. to 130.degree. C., or 50.degree. C. to 130.degree. C., 50.degree. C. to 120.degree. C., or 50.degree. C. to 115.degree. C., or 50.degree. C. to 110.degree. C., or 50.degree. C. to 105.degree. C., or 50.degree. C. to 100.degree. C., or 50.degree. C. to 90.degree. C., or 50.degree. C. to 80.degree. C., or 60.degree. C. to 130.degree. C., 60.degree. C. to 120.degree. C., or 60.degree. C. to 115.degree. C., or 60.degree. C. to 110.degree. C., or 60.degree. C. to 105.degree. C., or 60.degree. C. to 100.degree. C., or 60.degree. C. to 90.degree. C., or 60.degree. C. to 80.degree. C., or 70.degree. C. to 130.degree. C., 70.degree. C. to 120.degree. C., or 70.degree. C. to 115.degree. C., or 70.degree. C. to 110.degree. C., or 70.degree. C. to 105.degree. C., or 70.degree. C. to 100.degree. C., or 60.degree. C. to 90.degree. C., or 70.degree. C. to 80.degree. C. or 80.degree. C. to 130.degree. C., 80.degree. C. to 120.degree. C., or 80.degree. C. to 115.degree. C., or 80.degree. C. to 110.degree. C., or 80.degree. C. to 105.degree. C., or 80.degree. C. to 100.degree. C., or 80.degree. C. to 90.degree. C., or 90.degree. C. to 130.degree. C., 90.degree. C. to 120.degree. C., or 90.degree. C. to 115.degree. C., or 90.degree. C. to 110.degree. C., or 90.degree. C. to 105.degree. C., or 90.degree. C. to 100.degree. C., or 100.degree. C. to 130.degree. C., 100.degree. C. to 120.degree. C., or 100.degree. C. to 115.degree. C.; and wherein, after curing, the compositions of the invention can have service temperatures up to 130.degree. C., or up to 120.degree. C., or up to 115.degree. C., or up to 110.degree. C., or up to 105.degree. C., or up to 100.degree. C., or up to 95.degree. C., or up to 90.degree. C., or up to 85.degree. C., or up to 80.degree. C., or up to 75.degree. C.; and optionally, after service at these temperatures, can maintain a 180 degree peel strength of at least 2 lb/in, or at least 3 lb/in, or at least 4 lb/in, or at least 5 lb/in as measured according to ISO 8510-2-2006 Part 2 at 5 mm/sec.
[0076] In aspect 56 of this invention, there is provided a composition of any of aspects 1-55 which is not a water-dispersible polyester microgel composition.
[0077] In aspect 57 of this invention, there is provided an adhesive comprising the composition of any one of aspects 1-56.
[0078] In aspect 58 of this invention, there is provided an adhesive comprising any of the compositions of aspects 1-57 which can be used in at least one of the following: automotive interior adhesive, flexible laminating adhesive, rigid laminating adhesive, assembly adhesive, labelling adhesive, nonwoven adhesive, tape adhesive, structural adhesive, hygiene nonwoven construction adhesive, hygiene elastic attachment adhesive, home repair adhesive, industrial adhesive, construction adhesive, medical adhesive, contact adhesive, hot melt adhesive, solvent-based adhesive, packaging adhesive, product assembly adhesive, woodworking adhesive, flooring adhesive, automotive assembly adhesive, structural adhesive, pressure sensitive adhesive (PSA), PSA tape, PSA label, PSA protective film, laminating adhesive, flexible packaging adhesive, and hygiene core integrity adhesive.
[0079] In aspect 59 of this invention, there is provided a coating comprising the composition of any one of aspects 1-58.
[0080] In aspect 60 of this invention, there is provided an article comprising the composition of any of aspects 1-58.
[0081] In aspect 61 of this invention, there is provided the article of aspect 60 which is at least one of the following: an adhesive, a laminate, a tape, a label, a tag, a radio frequency identification (RFID) tag, a coating, a sealant, a film, a foam, a disposable hygiene article, a fiberglass reinforced plastic, a compound, a motor vehicle molded part, a motor vehicle extruded part, a motor vehicle laminated part, a woven textile, a nonwoven, a flexible packaging multilayer.
[0082] In aspect 62 of this invention, there is provided an article of aspects 60 or 61 comprising the cured composition of aspects 1-58 where the bond is maintained after cure and/or post-cure at or above about 60.degree. C.
[0083] In aspect 63 of this invention, there is provide an article of aspects any one of 1-59 comprising an additional step of blending in at least one additive selected from the group consisting of tackifiers, plasticizers, fillers, pigments, stabilizers, antioxidants, adhesion promoters, waxes, flame retardants, conductive agents, rheology modifiers or mixtures thereof.
[0084] In aspect 64 of this invention, there is provided a composite comprising the composition of any one of aspects 1-58 where the composite is selected from a polyester composite, a glass composite, or a wood-plastic composite.
[0085] In aspect 65 of this invention, there is provided at least one compound comprising any one of aspects 1-58 where the compound is selected from a polyacrylic blended compound, a potting compound, a rubber compound, a sheet molding compound (SMC) or a dough molding compound (DMC).
[0086] Formulations based on such polyesters and amine compounds are especially suitable for low-temperature curing adhesive applications without the use of Isocyanates. Chemical and/or physical protection of the primary amine curing agents can provide additional control options to influence the cure profile. Additionally, the novel system does not require moisture, so it can be used with moisture-impermeable substrates such as artificial leather.
[0087] Unpredictably, the compositions useful in this invention have a good combination of properties including but not limited to good thermal stability, the ability to cure and/or crosslink, good cure rate, good green strength, good peel strength, and ability to adhere to various substrates.
DETAILED DESCRIPTION
[0088] The present invention may be understood more readily by reference to the following detailed description of certain embodiments of the invention and the working examples. In accordance with the purpose(s) of this invention, certain embodiments of the invention are described in the Summary of the Invention and are further described herein below. Also, other embodiments of the invention are described herein.
[0089] Unless otherwise indicated, all numbers expressing quantities of ingredients, properties such as molecular weight, reaction conditions, and so forth used in the specification and claims are to be understood as being modified in all instances by the term "about." Unless indicated to the contrary, the numerical parameters set forth in the following specification and attached claims are approximations that may vary depending upon the desired properties sought to be obtained by the present invention. At the very least, each numerical parameter should be construed in light of the number of reported significant digits and by applying ordinary rounding techniques. Further, the ranges stated in this disclosure and the claims are intended to include the entire range specifications and not just the endpoint(s). For example, a range stated to be 0 to 10 is intended to disclose all whole numbers between 0 and 10 such as, for example 1, 2, 3, 4, etc., all fractional numbers between 0 and 10, for example 1.5, 2.3, 4.57, 6.1113, etc., and the endpoints 0 and 10.
[0090] Notwithstanding that the numerical ranges and parameters setting forth the broad scope of the invention are approximations, the numerical values set forth in the specific examples are reported as precisely as possible. Any numerical value, however, inherently contains certain errors necessarily resulting from the standard deviation found in its respective testing measurements.
[0091] As used in the specification and the appended claims, the singular forms "a," "an" and "the" include their plural referents unless the context clearly dictates otherwise. For example, a reference to "a polyester," "a dicarboxylic acid", "a residue" is synonymous with "at least one" or "one or more" polyesters, dicarboxylic acids, or residues and is thus intended to refer to both a single or plurality of polyesters, dicarboxylic acids, or residues. In addition, references to a composition containing or including "an" ingredient or "a" polyester is intended to include other ingredients or other polyesters, respectively, in addition to the one named. The terms "containing" or "including" are intended to be synonymous with the term "comprising", meaning that at least the named compound, element, particle, or method step, etc., is present in the composition or article or method, but does not exclude the presence of other compounds, catalysts, materials, particles, method steps, etc., even if the other such compounds, material, particles, method steps, etc., have the same function as what is named, unless expressly excluded in the claims.
[0092] Also, it is to be understood that the mention of one or more process steps does not preclude the presence of additional process steps before or after the combined recited steps or intervening process steps between those steps expressly identified.
[0093] Compositions comprising polyesters having moieties containing activated methylene or methine groups, such as those of beta-ketoesters, have properties including but not limited to one or more of the following properties: (1) cures and is isocyanate free, (2) is curable at low temperatures (e.g. <100.degree. C.), (3) has suitable adhesion at service temperatures up to 130.degree. C., (4) has good adhesion to substrates typically bonded or coated with isocyanate-containing systems, (5) no Volatile Organic Components (VOCs) or has low VOCs, (6) does not generate by-products during curing that can be detrimental to film formation or quality, (7) reduces defects, (8) has more pleasant odor, less odor, or is odorless, (9) can contain low or no moisture, (10) can be used with moisture-impermeable substrates such as artificial leather, (11) under certain conditions, can be a crosslinking system, (12) can provide more controlled curing, (13) good thermal stability, (14) good chemical resistance, (15) good impact strength, (16) good tensile strength, (17) good aging resistance, and/or (18) can provide longer pot life.
[0094] In one embodiment, the invention provides a cured composition.
[0095] In one embodiment, the invention provides a cured composition that has good thermal stability.
[0096] In one embodiment, the invention provides a cured composition that has good thermal stability and good peel strength.
[0097] In one embodiment, the invention provides an adhesive composition comprising: [0098] I. a first component [Component (I)] comprising at least one polyester having two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and [0099] II. a second component [Component (II)] comprising at least one curing agent having two or more primary amine functional groups, salts thereof, or mixtures thereof.
[0100] The beta-ketoester groups are represented by Formula 1, wherein R is a branched or straight chain, saturated or unsaturated alkyl, alicyclic, or aromatic group; and R' is a hydrogen or a branched or straight chain, saturated or unsaturated alkyl, alicyclic, or aromatic group.
##STR00001##
[0101] In one embodiment, the equivalent ratio of the .beta.-ketoacetate functional groups in the resin(s) of Component (I) to the primary amine functionality in the composition (i.e. eq. of .beta.-ketoacetate/eq. of primary amine) is from about 0.10 to about 8.0; or from about 0.25 to about 8.0; or from about 0.50 to about 8.0; or from about 1.0 to about 8.0; or from about 1.5 to about 8.0; or from about 2.0 to 8.0; or from about 2.5 to about 8.0; or from about 3.0 to about 8.0; or from about 3.5 to about 8.0; or from about 0.10 to about 7.0; or from about 0.25 to about 7.0; or from about 0.50 to about 7.0; or from about 1.0 to about 7.0; or from about 1.5 to about 7.0; or from about 2.0 to about 7.0; or from about 2.5 to about 7.0; or from about 3.0 to about 7.0; or from about 3.5 to about 7.0; or from about 0.10 to about 6.0; or from about 0.25 to about 6.0; or from about 0.50 to about 6.0; or from about 1.0 to about 6.0; or from about 1.5 to about 6.0; or from about 2.0 to about 6.0; or from about 2.5 to about 6.0; or from about 3.0 to about 6.0; or from about 3.5 to about 6.0; or from about 0.10 to about 5.0; or from about 0.25 to about 5.0; or from about 0.50 to about 5.0; or from about 1.0 to about 5.0; or from about 1.5 to about 5.0; or from about 2.0 to 5.0 or from about 2.5 to about 5.0; or from about 3.0 to about 5.0; or from about 3.5 to about 5.0; or from about 0.10 to about 4.0; or from about 0.25 to about 4.0; or from about 0.50 to about 4.0; or from about 1.0 to about 4.0; or from about 1.5 to about 4.0; or from about 2.0 to 4.0 or from about 2.5 to about 4.0; or from about 3.0 to about 4.0; from about 0.10 to about 3.0; or from about 0.25 to about 3.0; or from about 0.50 to about 3.0; or from about 1.0 to about 3.0; or from about 1.5 to about 3.0; or from about 2.0 to 3.0; or from about 0.10 to about 2.0; or from about 0.25 to about 2.0; or from about 0.50 to about 2.0; or from about 0.50 to about 1.5; or from about 0.7 to about 1.3; or from about 0.8 to about 1.2; or from about 1.0 to about 2.0; or from about 1.05 to about 2.0; or from about 1.10 to about 2.0; or from about 1.2 to about 2.0; or from about 1.3 to about 2.0; or from about 1.4 to about 2.0; or from about 1.5 to about 2.0; or from about 0.10 to about 1.0; or from about 0.25 to about 1.0; or from about 0.50 to about 1.0. The equivalent ratio of the .beta.-ketoacetate functional groups in the resin(s) of Component (I) to the primary amine functional groups in the composition is defined as the ratio of the equivalents of .beta.-ketoacetate functional groups or acetoacetate (AcAc) functional groups in the resin to the equivalents of primary amine functional groups in the composition. For example, the equivalents of the AcAc functional group in a sample of 100 g AcAc polyester with AcAc number of 120 mgKOH/g can be calculated by the formula: equivalents of AcAc polyester=100.times.120/56100=0.21; whereas the equivalents of the amine functional group in a sample of 0.10 moles of a diamine is equal to 0.10.times.2=0.20. Thus, the equivalent ratio is equal to 0.21/0.20=1.05.
[0102] In one embodiment, the equivalent ratio of the .beta.-ketoacetate functionality in the polyester to the primary amine functionality in the composition (i.e. eq. of .beta.-ketoacetate/eq. of primary amine) is from about 0.25 to about 4.0; or from about 0.50 to about 4.0; or from about 0.25 to about 3.0; or from about 0.50 to about 3.0; or from about 0.25 to about 2.0; or from about 0.50 to about 2.0; or from about 0.50 to about 1.5; or from about 0.7 to about 1.3; or from about 0.8 to about 1.2.
[0103] The polyesters useful in the invention can contain two or more beta-ketoester groups, represented by Formula 2, wherein Z is a polyester residue, R is an alkyl group, R' is hydrogen or an alkyl group, and n represents the average number of beta-ketoester groups per polymer chain and is an average of at least 2. In one aspect, the beta-ketoester group is acetoacetate (AcAc), and the polyester is an acetoacetate-functional polyester.
##STR00002##
[0104] The acetoacetate-functional polyester may be prepared by reacting a polyester resin containing hydroxyl groups, for example, a polyester having a hydroxyl number of at least 5 mg KOH/g, desirably about 30 to 200 mg KOH/g, with diketene or a compound having the beta-ketoacetate moiety such as t-butylacetoacetate (tBAA). Various methods for the preparation of acetoacetylated polyester coating resins have been described by Witzeman et al. in the Journal of Coatings Technology, Vol. 62, No. 789, pp. 101-112 (1990). A reaction mixture can include from about 50 to 95, 60 to 90, 65 to 85, or 70 to 80 weight % of the polyester resin and from about 5 to 50, 10 to 40, 15 to 35, or 20 to 30 weight % of the compound having a beta-ketoacetate moiety or a diketene; these can be reacted together, wherein the weight percentages are based on the total weight of the polyester resin and the compound having the beta-ketoacetate moiety equaling 100 weight %.
[0105] In one embodiment, the curing agent [Component (II)] is a small molecule or an adduct having one or more primary amine functional groups or two or more primary amine functional groups. Examples of amines useful in this invention include but are not limited to at least one of ethylenediamine; diethylenetriamine; triethylenetetramine (TETA) or isomers thereof; tetraethylenepentamine or isomers thereof; 2,2,4-trimethylhexamethylenediamine; 2,4,4-trimethylhexamethylenediamine; 1,2-diaminopropane; 1,3-diaminopropane; 2,2-dimethylpropylenediamine; 1,4-diaminobutane; 2-methylpentamethylenediamine (MPMDA); 1,6-hexanediamine; 1,7-diaminaheptane; 1,8-diaminooctane; 1,9-diaminononane; 2-diaminododecane; 4-azaheptamethylenediamine; N,N-bis(3-aminopropyl)butane-1,4-diamine; N,N-bis(3-aminopropyl)ethylenediamine; 2,4-toluenediamine; 2,6-toluenediamine; 3,3'-iminobispropylamine; 1,2-diaminocyclohexane; 1,3-diaminocyclohexane; 1,4-diamino-2,5-diethylcyclohexane; 1,4-diamino-3,6-diethylcyclohexane; 1,2-diamino-4-ethylcyclohexane; 1,2-diamino-4-cyclohexylcyclohexane; isophorone diamine (IPDA); norbornanediamine; 4,4'-diaminodicyclohexylmethane; 4,4'-diaminodicyclohexylethane; 4,4'-diaminodicyclohexylpropane; 2,2-bis(4-aminocyclohexyl)propane; 3,3'-dimethyl-4,4'-diaminodicyclohexylmethane; 3-amino-1-(4-aminocyclohexyl)propane; 1,3-bis(aminomethyl)cyclohexane; 1,4-bis(aminomethyl)cyclohexane; 1-cyclohexyl-3,4-diamino-cyclohexane; xylylenediamine and its hydrogenation products; p-xylylenediamine and its hydrogenation products; 4,4'-methylenedianiline; 2,4-bis(p-aminobenzyl)aniline; diethyltoluenediamine; m-phenylenediamine; diphenylethylenediamine; 2,2,4,4-tetramethylcyclobutane-1,3-diamine; 2,2-dimethylpropane-1,3-diamine; 2,3-dimethylbutane-2,3-diamine; 1,2-diaminocyclopentane; 1,2,2-trimethylcyclopentane-1,3-diamine; 1,7,7-trimethylbicyclo[2.2.1]heptane-2,3-diamine; 4,4'-methylenebis[2-methylcyclohexanamine] (MACM); and polyetheramines such as JEFFAMINE.RTM. D-230, 0-400, 0-2000, and T-403 amines.
[0106] The amine compounds may be chemically blocked to improve storage stability and/or extend the open time for application of the curable composition of the invention. The amine may be reacted with a ketone such as acetone to form a ketimine. The amine may be reacted with an aldehyde such as isobutyraldehyde to form an aldimine. The resulting blocked amine may be de-blocked by changing the pH or heating or moisture.
[0107] In this invention, "curing" means to develop at least one strength property and/or increase in the average molecular weight (either Mn or Mw) of the composition(s) of the invention, for example, an adhesive composition or a coating composition, by chemical reaction. "Curing agent" means a substance or mixture of substances that reacts with resin during a curing reaction. "Curing temperature" means the temperature to which at least one composition(s) of the invention or an article of manufacture containing at least one composition(s) of the invention is subjected to cure the composition. "Curing time" means the period of time for curing of the composition(s) of the invention to occur, whether or not they cure at room temperature, and whether or not they are subjected to application of thermal energy; radiation, e.g., microwave, ultraviolet, or infrared radiation; ultrasonic waves; pressure; moisture; or other sources of activation; or any other variables. "Curable" means capable of curing.
[0108] In another embodiment, the acetoacetate functional polyester comprises the reaction product (or residues) of (a) from about 50 to about 95 weight percent of a hydroxyl functional polyester and (b) from about 5 to about 50 weight percent of an alkyl acetoacetate or diketene, wherein the weight percentages are based on the total weight of (a) and (b) equaling 100 weight %.
[0109] In one embodiment, the polyester (hydroxyl functional) can be present in the amount of from 50 to 95 weight %, or from 60 to 90 weight %, or from 65 to 85 weight %, or from 70 to 80 weight %; and the alkyl acetoacetate or diketene can be present in the amount of 5 to 50 weight %, or from 10 to 40 weight %, or from 15 to 35 weight %, or from 20 to 30 weight %, respectively, wherein the weight percentages are based on the total weight of (a) and (b) equaling 100 weight %.
[0110] The hydroxyl number of the hydroxyl functional polyester resin used to make the curable acetoacetate functional polyester resin can be within a range of from 0 to 300 mg KOH/g. Suitable ranges of hydroxyl number can include from 0 to 300, or from 10 to 300, or from 30 to 300, or from 50 to 300, or from 60 to 300, or from 80 to 300, or from 100 to 300, or from 120 to 300, or from 140 to 300, or from 160 to 300, or from 180 to 300, or greater than 0 to 250, or from 10 to 250, or from 30 to 250, or from 50 to 250, or from 60 to 250, or from 80 to 250, or from 100 to 250, or from 120 to 250, or from 140 to 250, or from 160 to 250, or from 180 to 250, or greater than 0 to 240, or from 10 to 240, or from 30 to 240, or from 50 to 240, or from 60 to 240, or from 80 to 240, or from 100 to 240, or from 120 to 240, or from 140 to 240, or from 160 to 240, or from 180 to 240, or greater than 0 to 220, or from 10 to 220, or from 30 to 220, or from 50 to 220, or from 60 to 220, or from 80 to 220, or from 100 to 220, or from 120 to 220, or from 140 to 220, or from 160 to 220, or from 180 to 220 or greater than 0 to 200, or from 10 to 200, or from 30 to 200, or from 50 to 200, or from 60 to 200, or from 80 to 200, or from 100 to 200, or from 120 to 200, or from 140 to 200, or from 160 to 200, or from 180 to 200 or greater than 0 to 180, or from 10 to 180, or from 30 to 180, or from 50 to 180, or from 60 to 180, or from 80 to 180, or from 100 to 180, or from 120 to 180, or from 140 to 180, or from 160 to 180, or from 0 to 160, or from 10 to 160, or from 30 to 160, or from 50 to 160, or from 60 to 160, or from 80 to 160, or from 100 to 160, or from 120 to 160, or from 140 to 160 or from 0 to 150, or from 10 to 150, or from 30 to 150, or from 50 to 150, or from 60 to 150, or from 70 to 150, or from 80 to 150, or from 100 to 150, or from 120 to 150, or from 130 to 150 mg KOH/g.
[0111] In one embodiment, the hydroxyl number of the hydroxyl functional polyester resin is desirably at least 5, or at least 30, or at least 50, or at least 80, or from 30 to 200; or from 50 to 150 mg KOH/g.
[0112] In one embodiment, the composition of the invention or the polyester resin of the invention has an acid number of 0 to 15, or 0 to 10, or less than 10, or 0 to 5, or less than 5 mg KOH/g, with and/or without neutralizing agents being added to the composition.
[0113] In one embodiment, the compositions of the invention or of the polyester resin of the invention can have acid numbers from 0 to about 14, or from 0 to about 13, or from 0 to about 12, or from 0 to about 11, or from 0 to about 10, or from 0 to about 9, or from 0 to about 8, or from 0 to about 7, or from 0 to about 6, or from 0 to about 5, or from 0 to about 4, or from 0 to about 3, or from 0 to about 2, or from 0 to about 1, or from 0.01 to about 15, or from 0.01 to about 14, or from 0.01 to about 13, or from 0.01 to about 12, or from 0.01 to about 11, or from 0.01 to about 10, or from 0.01 to about 9, or from 0.01 to about 8, or from 0.01 to about 7, or from 0.01 to about 6, or from 0.01 to about 5, or from 0.01 to about 4, or from 0.01 to about 3, or from 0.01 to about 2, or from 0.01 to about 1, or from 0.50 to about 15, or from 0.50 to about 14, or from 0.50 to about 13, or from 0.50 to about 12, or from 0.50 to about 11, or from 0.50 to about 10, or from 0.50 to about 9, or from 0.50 to about 8, or from 0.50 to about 7, or from 0.50 to about 6, or from 0.50 to 5 or from 0.50 to about 4, or from 0.50 to about 3, or from 0.50 to about 2, or from 0.50 to about 1.5 mg KOH/g, with or without addition of neutralizing agents.
[0114] In one embodiment of this invention, the composition can have any one of the acid numbers in the previous paragraph, where neutralizing agents are not added to the composition after the polymerization reaction. Neutralizing agents can any neutralizing agent known in the art. In the embodiment of the invention where neutralizing agents are excluded from the scope of this invention, particularly where added after polymerization, neutralizing agents can include but are not limited to primary amines, secondary amines, tertiary amines, and ammonia as well as any other neutralizing agent known in the art, for example, for water dispersible or water dispersed microgel compositions.
[0115] In one embodiment of the invention, there are provided compositions wherein the composition can contain residues of amine, formed from the use of at least one primary amine, that may be incorporated into the composition during curing. The primary amines or amine salts useful in this invention can be used as curing agents for the reaction of the p-ketoacetate (AcAc) group of Component (I) and not as a neutralizing agent.
[0116] The acid number as referred to herein was measured by using a procedure based on ASTM D7253-16 entitled "Standard Test Method for Polyurethane Raw Materials: Determination of Acidity as Acid Number for Poly ether Polyols," and hydroxyl number was measured using a procedure based on ASTM E222-17 entitled "Standard Test Methods for Hydroxyl Groups Using Acetic Anhydride."
[0117] The glass transition temperature (Tg) of the acetoacetate-functional polyester of the present invention can be from about -85.degree. C. to about 150.degree. C.; or from -85.degree. C. to 130.degree. C.; or from -85 to 120.degree. C.; or from -85.degree. C. to 110.degree. C.; or from -85.degree. C. to 100.degree. C.; or from -85.degree. C. to 80.degree. C.; or from -85.degree. C. to about 60.degree. C.; or about -85.degree. C. to about 50.degree. C.; or about -85.degree. C. to about 40.degree. C.; or about -85.degree. C. to about 30.degree. C.; or about -85.degree. C. to about 20.degree. C.; or about -85.degree. C. to about 10.degree. C.; or about -85.degree. C. to about 0.degree. C.; or about -85.degree. C. to about -10.degree. C.; or about -85.degree. C. to about -20.degree. C.; or about -85.degree. C. to about -25.degree. C.; or about -85.degree. C. to about 30.degree. C. about -85.degree. C. to about 60.degree. C.; or about -85.degree. C. to about 50.degree. C.; or about -85.degree. C. to about 40.degree. C.; or about -85.degree. C. to about 30.degree. C.; or about -85.degree. C. to about 20.degree. C.; or about -85.degree. C. to about 10.degree. C.; or about -85.degree. C. to about 0.degree. C.; or about -85.degree. C. to about -10.degree. C.; or about -85.degree. C. to about -20.degree. C.; or about -85.degree. C. to about -25.degree. C.; or about -85.degree. C. to about -30.degree. C.; or about -70.degree. C. to about 60.degree. C.; or about -70.degree. C. to about 50.degree. C.; or about -70.degree. C. to about 40.degree. C.; or about -70.degree. C. to about 30.degree. C.; or about -70.degree. C. to about 20.degree. C.; or about -70.degree. C. to about 10.degree. C.; or about -70.degree. C. to about 0.degree. C.; or about -70.degree. C. to about -10.degree. C.; or about -70.degree. C. to about -20.degree. C.; or about -70.degree. C. to about -25.degree. C.; or about -70.degree. C. to about -30.degree. C.; or about -65.degree. C. to about 60.degree. C.; or about -65.degree. C. to about 50.degree. C.; or about -65.degree. C. to about 40.degree. C.; or about -65.degree. C. to about 30.degree. C.; or about -65.degree. C. to about 25.degree. C.; or about -65.degree. C. to about 20.degree. C.; or about -65.degree. C. to about 10.degree. C.; or about -65.degree. C. to about 0.degree. C.; or about -65.degree. C. to about -10.degree. C.; or about -65.degree. C. to about -20.degree. C.; or about -65.degree. C. to about -25.degree. C.; or about -65.degree. C. to about -30.degree. C.; or about -45.degree. C. to about 60.degree. C.; or about -45.degree. C. to about 50.degree. C.; or about -45.degree. C. to about 40.degree. C.; or about -45.degree. C. to about 30.degree. C.; or about -45.degree. C. to about 25.degree. C.; or about -45.degree. C. to about 20.degree. C.; or about -45.degree. C. to about 10.degree. C.; or about -45.degree. C. to about 0.degree. C.; or about -45.degree. C. to about -10.degree. C.; or about -45.degree. C. to about -20.degree. C.; or about -45.degree. C. to about -25.degree. C.; or about -45.degree. C. to about -30.degree. C.
[0118] In certain embodiments, the glass transition temperature (Tg) of the acetoacetate-functional polyester of the present invention can be from about -85.degree. C. to about -10.degree. C.; or from about -70.degree. C. to about 50.degree. C.; or from about -65.degree. C. to about 40.degree. C.; or from about -65.degree. C. to about 25.degree. C.; or from about -10.degree. C. to about 50.degree. C.; or from about -45.degree. C. to about -10.degree. C.; or from about -45.degree. C. to about -25.degree. C.
[0119] The number average molecular weight (Mn) of the acetoacetate functional polyester useful in the present invention may be from 500 to 100,000; or from 700 to 50,000; or from 700 to 40,000; or from 700 to 30,000; or from 700 to 25,000; or from 700 to 20,000; or from 700 to 15,000; or from 700 to 10,000; or from 700 to 9,000; or from 700 to 8,000; or from 700 to 7,000; or from 1000 to 6,000; or from 700 to 5,000; or from 700 to 4,000; or from 700 to 3500; or from 1,000 to 50,000; or from 1,000 to 40,000; or from 1,000 to 30,000; or from 1,000 to 25,000; or from 1,000 to 20,000; or from 1000 to 15,000; or from 1000 to 10,000; or from 1000 to 9,000; or from 1000 to 8,000; or from 1000 to 7,000; or from 1000 to 6,000; or from 1000 to 5,000; or from 1000 to 4,000; or from 1500 to 4,000; or from 1500 to 3,500; or from 1500 to 3,000; or from 1500 to 2500; or from 1200 to 3500 g/mole. The polyester may be linear or branched. The Mn is measured by gel permeation chromatography (GPO) using polystyrene equivalent molecular weight.
[0120] The hydroxyl functional polyester that is used to prepare acetoacetate polyester useful in the invention can be synthesized by reacting a hydroxyl compound, for example, a diol or triol, with a carboxyl compound, for example, a dicarboxylic acid. Examples of hydroxyl compounds include diols such as 2,2,4,4-tetraalkylcyclobutane-1,3-diol (TACO), 2,2-dimethyl-1,3-propanediol (neopentyl glycol), 1,2-cyclohexanedimethanol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 2,2,4-trimethyl-1,3-pentanediol, hydroxypivalyl hydroxypivalate, tricyclodecanedimethanol, 2-methyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 2-ethyl-2-isobutyl-1,3-propanediol, 1,3-butanediol, 1,4-butanediol, 1,5-pentanediol, 1,6-hexanediol, 2,2,4,4-tetramethyl-1,6-hexanediol, 1,10-decanediol, 1,4-benzenedimethanol, hydrogenated bisphenol A, ethylene glycol, propylene glycol, diethylene glycol, dipropylene glycol, triethylene glycol, tetraethylene glycol, polyethylene glycol, and polyols such as 1,1,1-trimethylol propane, 1,1,1-trimethylolethane, glycerin, pentaerythritol, erythritol, threitol, dipentaerythritol, sorbitol, and the like.
[0121] In one embodiment, the diol can be 2,2,4,4-tetraalkylcyclobutane-1,3-diol (TACD). Examples of TACD include 2,2,4,4-tetramethylcyclobutane-1,3-diol (TMCD), 2,2,4,4-tetraethylcyclobutane-1,3-diol, 2,2,4,4-tetra-n-propylcyclobutane-1,3-diol, and 2,2,4,4-tetra-n-butylcyclobutane-1,3-diol. In one embodiment, the TACO can be TMCD.
[0122] The carboxyl compound may be a polycarboxylic acid compound, a derivative of polycarboxylic acid compound, or a combination thereof. Suitable polycarboxylic acid compounds include compounds having at least two carboxylic acid groups. In one aspect, the polycarboxylic acid compound comprises a dicarboxylic acid compound having two carboxylic acid groups, derivatives thereof, or combinations thereof, capable of forming an ester linkage with a polyhydroxyl component. For example, a polyester can be synthesized by using a polyhydroxyl compound and a derivative of a dicarboxylic acid such as, for example, dimethyl ester or other dialkyl esters of the diacid, or diacid chloride or other diacid halides, or acid anhydride. In another aspect, the polycarboxylic acid compound comprises a tricarboxylic acid or anhydride, for example, trimellitic acid or trimellitic anhydride.
[0123] Examples of dicarboxylic acids that may be used include aliphatic dicarboxylic acids, alicyclic dicarboxylic acids, aromatic dicarboxylic acids, derivatives of each, or mixtures of two or more of these acids. Thus, suitable dicarboxylic acids include, but are not limited to, isophthalic acid (or dimethyl isophthalate), terephthalic acid (or dimethyl terephthalate), phthalic acid, phthalic anhydride, 1,4-cyclohexane-dicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, hexahydrophthalic anhydride, tetrahydrophthalic anhydride, tetrachlorophthalic anhydride, dodecanedioic acid, sebacic acid, azelaic acid, maleic acid or anhydride, fumaric acid, succinic anhydride, succinic acid, adipic acid, 2,6-naphthalenedicarboxylic acid, glutaric acid, itaconic acid, hexahydrophthalic acid or anhydride and their derivatives, diglycolic acid, 2,5-norbornanedicarboxylic acid, 1,4-naphthalenedicarboxylic acid, 2,5-naphthalenedicarboxylic acid, diphenic acid, 4,4'-oxydibenzoic acid, 4,4'-sulfonyldibenzoic acid, and mixtures thereof.
[0124] In one embodiment, where the hydroxyl component of the polyester comprises at least one diol and at least one polyol, the mole % of the diol component can be 60 to 100, 80 to 97, or 85 to 95 mole % based on the mole percentages of the diol and polyol equaling 100 mole % of the hydroxyl component, and the mole % of the polyol component of the hydroxyl component of the polyester can be 0 to 40, 3 to 20, or 5 to 15 mole %, based on the mole percentages of the diol and polyol equaling 100 mole % of the hydroxyl component.
[0125] Preferably, the diol of the hydroxyl component of the polyester comprises one or more selected from the group consisting of 2,2-dimethyl-1,3-propanediol (neopentyl glycol), 2-methyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 1,4-butanediol, 1,6-hexanediol, 1,2-cyclohexanedimethanol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 2,2,4-trimethyl-1,3-pentanediol, hydroxypivalyl hydroxypivalate, 2,2,4,4-tetramethylcyclobutane-1,3-diol, tricyclodecanedimethanol, and polyethylene glycol.
[0126] In one embodiment of this invention, there is provided a composition wherein diol component (a)(i) of the polyester contains only oxygen, carbon, and hydrogen.
[0127] In one embodiment of this invention, there is provided a composition wherein diol component (a)(i) of the polyester does not have any carbonyl groups (--CO--).
[0128] In one embodiment of this invention, there is provided a composition wherein diol component (a)(i) of the polyester does not contain ether groups.
[0129] In one embodiment, the polyol component (a)(ii) of the hydroxyl component of the polyester is selected from at least one of 1,1,1-trimethylol propane, 1,1,1-trimethylolethane, glycerin, pentaerythritol or sorbitol.
[0130] In one embodiment, the carboxyl component of the polyester comprises one or more carboxylic acids and/or anhydrides selected from the group consisting of isophthalic acid (or dimethyl isophthalate), terephthalic acid (or dimethyl terephthalate), phthalic acid, phthalic anhydride, 1,4-cyclohexane-dicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, adipic acid, 2,6-naphthalene-dicarboxylic acid, 1,4-naphthalenedicarboxylic acid' 2,5-naphthalenedicarboxylic acid' hexahydrophthalic acid or anhydride, tetrahydrophthalic acid or anhydride, trimellitic anhydride, maleic acid or anhydride, fumaric acid, succinic anhydride, and succinic acid. In one embodiment, the carboxyl compound is selected from the group consisting of adipic acid, isophthalic acid (or dimethyl isophthalate), 1,4-cyclohexanedicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, hexahydrophthalic acid or anhydride, maleic anhydride, succinic anhydride, phthalic acid, and phthalic anhydride.
[0131] In one embodiment, at least one semi-crystalline polyester resin can be used. In one embodiment, the semi-crystalline polyester resin can possess enough crystalline character to give the material a sharp melting point rather than slowly softening when heated. The semi-crystalline polyester resin can be a solid, or a waxy or a semi-solid material at room temperature with a melting point, for example, from 10 to 100.degree. C., or for example, from 15 to 75.degree. C., or for example, from 25 to 65.degree. C.
[0132] The glass transition temperature (Tg) of the semi-crystalline polyester of the invention can be from -70 to 20.degree. C., or from -60 to 0.degree. C., or from -50 to -20.degree. C.,
[0133] The polyester resin can have a number average molecular weight (Mn) from 1000-5000 viol, preferably 1500-4000 viol.
[0134] In one embodiment, the invention may comprise at least one polyether glycol functionalized with acetoacetate groups instead of or in addition to a polyester functionalized with acetoacetate groups.
[0135] Suitable polyethers include unsubstituted poly(alkylene glycol)s having alkylene chains of 1 to 3 carbon atoms, substituted or unsubstituted poly(alkylene glycol)s having alkylene chains of at least 4 carbon atoms and preferably less than 10 carbon atoms.
[0136] In one embodiment, the equivalent ratio of the acetoacetate (AcAc) functionality of the polyether glycol to the primary amine functionality in the composition is from 0.25 to 4, or from 0.5 to 2.0, or from 0.7 to 1.5.
[0137] Suitable polyethers may be branched to increase their functionality. For example, glycerol or sucrose may be included to increase branching, such as in propoxylated glycerol and propoxylated sucrose. The compositions of the invention can comprise residues of at least one polyol component selected from 1,1,1-trimethylolpropane, 1,1,1-trimethylolethane, glycerin, sorbitol, and pentaerythritol or mixtures thereof. Triisocyanates and aliphatic or aromatic glycols may be used to increase branching as well. The poly(alkylene glycol)s can be obtained by methods well known in the art. Examples of poly(alkylene glycol)s include poly(ethylene glycol), poly(propylene glycol), poly(tetramethylene glycol), poly(pentamethylene glycol), poly(hexamethylene glycol), poly(heptamethylene glycol), and poly(octamethylene glycol). Preferred poly(alkylene glycol)s include poly(ethylene glycol), poly(propylene glycol) and poly(tetramethylene glycol). Almost any number of repeating units may be used; however poly(alkylene glycol)s having number average molecular weights in the range of about 500 to 5,000 are preferred. Suitable poly(alkylene glycol)s may have a variety of suitable end groups, including, but not limited to hydroxyl, epoxy, methyl and the like. Preferred end groups are methyl and hydroxyl.
[0138] In one embodiment of the invention, there are provided compositions comprising blends of at least one acetoacetylated polyether resin and/or at least one acetoacetylated polyester resin and optionally, other polymers.
[0139] The amount of the polyether in the present invention may vary, so long as the final composition can be formed into the desired article. In one embodiment, Component (I) can contain 100% by weight acetoacetylated polyether, 0 to 100% by weight acetoacetylated polyether, or 20 to 80% by weight acetoacetylated polyether; or 35 to 65% by weight acetoacetylated polyether, based on the total weight percentage of Component (I) comprising 100 weight %. The remaining weight percentages can be any polymer, for example, polyester, acetoacetylated polyester or polycarbonate.
[0140] In one embodiment of the invention, there are provided compositions comprising from 100% or less, or 0.01 to 100%, or 0.01 to 80%, or 0.01 to 65%, or 0.01 to 25%, or 20 to 100%, or 20 to 80%, or 20 to 65%, or 5 to 25'%, or 20 to 80%, or 30 to 100%, or 30 to 80%, or 30 to 65%, or 40 to 100%, or 40 to 80%, or 40 to 65%, or 35 to 65%, or 35 to 54%, or 50 to 100%, or 50 to 80%, or 50 to 100%, or 50 to 80%, or 60 to 100%, or 60 to 80% by weight of acetoacetylated polyesters of the invention wherein the remaining weight percentages of the resin in Component (I) can be any polymer or copolymer, for example, polyethers, polyurethanes, or polycarbonates, (acetoacetylated or not), based on the total weight percentage of Component (I) equaling 100 weight %.
[0141] Polyalkylene oxides such as polypropylene oxides and ethylene oxides may also be used, such as linear polyethylene oxide), linear poly(propylene oxide), linear copolymers of polyethylene oxide) and poly(propylene oxide), linear poly(1,3-trimethylene oxide), and linear poly(1,4-tetramethylene oxide). Poly(alkylene glycol) copolymers of various glycol units may also be used in the present invention. The copolymers may be alternating, random, segmented, block, graft, or branched.
[0142] Examples of poly(alkylene glycol) copolymers include polyethylene glycol)-ran-polypropylene glycol), poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol) and polypropylene glycol)-block-poly(ethylene glycol)-block-poly(propylene glycol). A more specific example is a block copolymer in which a polypropylene glycol) initiated with a polyhydric alcohol is endcapped with ethylene oxide to convert some or the majority of the secondary hydroxyls to primary hydroxyls. The polyether condensation product may be prepared using methods known in the art. For example, a polyether condensation product may be formed by reacting polyethylene glycol with tetramethyl cyclopentane-1,2,3,4-tetracarboxylate. This is a general method that may be used to prepare other polyether condensation products useful in the invention.
[0143] The condensation product of the polyethers can be produced by reacting at least one core molecule having more than one carboxylic add group with a sufficient amount of at least one polyether terminated with hydroxy groups to produce an esterified polyether with an average of more than two hydroxy end groups. Suitable core molecules include, but are not limited to oxalic add, malonic add, succinic add, glutaric add, adipic add, hexahydrophthalic add or anhydride, benzenedicarboxylic add, benzenetricarboxylic add, benzenetetracarboxylic add, cyclohexanetricarboxylic add, cyclopentanetetracarboxylic add, adamantanetetracarboxylic add, biphenyltetracarboxylic add, benzophenonetetracarboxylic acid, propanetricarboxylic add, butanetetracarboxylic add, and mixtures thereof.
[0144] In addition to poly(alkylene glycol)s, polyethers comprising polymeric ethers derived from cyclic ether monomers can be used. For example, poly(2,3-dihydrofurandiyl), prepared by cationic polymerization of 2,3-dihydrofuran, can be incorporated into an oxygen scavenging composition in the same fashion as the above-mentioned poly(alkylene glycol)s.
[0145] Examples of the alkyl acetoacetate useful in the invention include t-butyl acetoacetate, methyl acetoacetate, ethyl acetoacetate, n-propyl acetoacetate, isopropyl acetoacetate, n-butyl acetoacetate, isobutyl acetoacetate, t-amyl acetoacetate, diisopropyl carbinol acetoacetate, and the like or mixtures thereof.
[0146] In one embodiment, the composition of the invention can contain blends with other polymers, including but not limited to blends of polyether resin(s) and polyester resin(s), or blends of AcAc polyether resins and AcAc polyester resin(s).
[0147] In one embodiment, any composition of the invention can be curable (capable of curing). In one embodiment, any composition useful in the invention can be cured. In one embodiment, the compositions of the invention can be cured at low temperatures (e.g. <100.degree. C.). In one embodiment, the compositions of the invention can be cured at room temperature, or 10.degree. C. to 130, or 10.degree. C. to 30.degree. C., or 20.degree. C. to 120.degree. C., or 80.degree. C. to 130.degree. C., or 50.degree. C. to 130.degree. C., or 50.degree. C. to 120.degree. C., or 50.degree. C. to 115.degree. C., or 50.degree. C. to 110.degree. C. or 50.degree. C. to 105.degree. C., or 50.degree. C. to 100.degree. C., or 50.degree. C. to 90.degree. C., or 50.degree. C. to 80.degree. C., or 60.degree. C. to 130.degree. C., or 60.degree. C. to 120 or 60.degree. C. to 115.degree. C., or 60.degree. C. to 110.degree. C., or 60.degree. C. to 105.degree. C., or 60.degree. C. to 100.degree. C., or 60.degree. C. to 90.degree. C., or 60.degree. C. to 80.degree. C., or 70.degree. C. to 130.degree. C., or 70.degree. C. to 120.degree. C., or 70.degree. C. to 115.degree. C., or 70.degree. C. to 110.degree. C., or 70.degree. C. to 105.degree. C., or 70 to 100.degree. C., or 60.degree. C. to 90.degree. C., or 70.degree. C. to 80.degree. C. or 80.degree. C. to 130.degree. C., or 80.degree. C. to 120.degree. C., or 80.degree. C. to 115.degree. C. or 80.degree. C. to 110.degree. C., or 80.degree. C. to 105.degree. C., or 80.degree. C. to 100.degree. C., or 80.degree. C. to 90.degree. C., or 90.degree. C. to 130.degree. C., or 90.degree. C. to 120.degree. C., or 90.degree. C. to 115.degree. C., or 90.degree. C. to 110.degree. C., or 90.degree. C. to 105.degree. C., or 90.degree. C. to 100.degree. C., or 100.degree. C. to 130.degree. C., or 100.degree. C. to 120.degree. C., or 100.degree. C. to 115.degree. C.
[0148] In another embodiment, after curing, the compositions of the invention can maintain suitable adhesion or bond strength at service temperatures up to 130.degree. C., or up to 120.degree. C., or up to 115.degree. C., or up to 110.degree. C., or up to 105.degree. C., or up to 100.degree. C., or up to 95.degree. C., or up to 90.degree. C., or up to 85.degree. C., or up to 80.degree. C., or up to 75.degree. C. In certain embodiments, "suitable adhesion or bond strength" can include but is not limited to the composition having a 180 degree peel strength of at least 2 lb/in, or at least 3 lb/in, or at least 4 lb/in, or at least 5 lb/in as measured according to ISO 8510-2-2006 Part 2 at 5 mm/sec.
[0149] In order to extend the pot life of the curable adhesive of the invention, the amine curing agent [Component (II)] may be temporarily blocked. For example, a carboxylic acid, such as benzoic acid, acetic acid, formic acid, or cyanoacetic acid, can also be added to the composition to chemically block the amine and subsequently deblock by heating. Such techniques for blocking and deblocking amines have been disclosed in "Delayed (Latent) Catalysis in Coatings" Werner J. Blank, Kurt Dietliker, Tunja Jung, Caroline Lordelot, Antoine Carroy (www.researchgate.net/publication/228420884). The addition of mild buffering agents such as weak acids can influence the cure rate, thermal stability and polymer performance. Solid catalysts may be particularly suited to chemical blocking, as only the outer surface of a solid particle need be blocked. "Pot life" herein means period of time during which a multi-part composition can be used after mixing the components.
[0150] In one embodiment of the invention, there are provided compositions useful in the invention that are capable of curing, being curable, having been cured, or having reached a gelation point. At the gelation point, a loose network of crosslinks are formed across the material and the gel or gelation point can be measured as either (1) the point when the storage modulus (G') and the loss modulus (G'') cross, or (2) when the tan delta is independent of frequency, each method by rotational rheometry using a Discovery Hybrid Rheometer 3 (DHR), T.A. Instruments.
[0151] As used herein, storage modulus is a measure of elastic response of a material. It measures the stored energy. Loss modulus is a measure of viscous response of a material. It measures the energy dissipated as heat. Tan delta, a measure of damping, is the ratio of loss modulus to the storage modulus, G''/G'. These properties can be used to evaluate compositions for stability at storage and spray conditions, gelation process, and thermal stability.
[0152] In one embodiment of the invention, there are provided compositions having a gelation point, or that cures, at or above 25.degree. C. as measured by a Discovery Hybrid Rheometer (DHR), T.A. Instruments.
[0153] "Pot life" herein means the period of time during which a multi-part composition can be used after mixing the components. "Working life" means the time period that a composition is available to be applied to at least one substrate and to be capable of forming a bond.
[0154] Pot life measurements can include various methods such as: (1) the time from the initial viscosity (when components have been all combined) to the time of doubling of viscosity; (2) reaching a particular viscosity value above which the composition can no longer be used in a given application or application method; (3) reaching a viscosity so high that the curable composition can no longer be molded or applied to a substrate, depending on the particular application and its requirements; or (4) for solvent-based compositions, reaching a viscosity of 24 seconds using a #2 Zahn cup following ASTM Method D4212 at room temperature.
[0155] The curing agents (amines) may be released for reaction in a controlled manner through a physical protecting mechanism or protecting layer, for example, a coated or blended component that releases the curing agent upon heating, to improve storage stability, and/or extend the pot life or open time for application of the curable composition of the invention, and/or enable rapid formation of initial "green strength" while preventing full cure until "activated" or "de-protected." This may also be called a controlled cure profile. Solid curing agents can be particularly suited for physical protection, as only the outer surface of a particle usually has to be coated. The protecting layer could include materials such as waxes, tackifier resins, or rosin ester tackifier resins, including but not limited to Permalyn.TM. 6110, Permalyn.TM. 6110M, Permalyn.TM. 5095, Kristalex.TM. 3070, or Foralyn.TM. 90 (Eastman Chemical Company). Physical protecting and chemical blocking mechanisms may be combined to improve protection and/or to control the cure profile of the inventive curable compositions by activating, or de-protecting, under varying conditions. In one embodiment, tackifier resins can be used. In one embodiment, waxes can used. In another embodiment, a combination of tackifier resins and/or waxes can be used.
[0156] In addition to adjusting the cure profile of the composition through the physical blocking of the curing agent, the wax(es) and/or tackifier resin(s) may reduce the overall viscosity of the adhesive, reduce application temperature, and improve application and uniformity.
[0157] Any conventionally known wax, which is suitable for use in formulating adhesives, can be used in the practice of the invention. Suitable waxes can include, for example, microcrystalline wax, paraffin wax, waxes produced by Fischer-Tropsch processes, functionalized waxes (maleated, fumerated, silanated, or wax with functional groups etc.), polyolefin waxes, petroleum waxes, polypropylene waxes, polyethylene waxes, ethylene vinyl acetate waxes, and vegetable waxes.
[0158] Non-limiting examples of commercially available waxes that can be suitable for this invention include A-C.TM. 9 and A-C.TM. 810, available from Honeywell International Inc. Morristown, N.J., US; POLYWAX.TM. 400, 850, 1000, and 3000 from Baker Hughes Inc., Sugar Land, Tex., US; Licocene.TM. PE4201 from Clariant International LTD, Muttenz, Switzerland; Epolene.TM. N-15, Epolene.TM. C-10, Epolene.TM. C-17 and Epolene.TM. C-18 from Westlake Polymers, LLC, Houston, Tex., US; Sasolwax.TM. 3279 microcrystalline wax from Sasol Chemical Industries Limited, South Africa; and microcrystalline wax Be Square.TM. 195 (Baker Hughes).
[0159] In various embodiments, adhesives comprising the curable compositions of the inventions can comprise at least 1, 2, 5, 8, or 10 and/or not more than 40, 30, 25, 20, or 15 weight percent of at least one wax based on the total weight of the final composition equaling 100 weight %. Moreover, said adhesives can comprise in the range of 1 to 40, 5 to 30, 8 to 25, or 10 to 20 weight percent of at least one wax.
[0160] Furthermore, a functionalized wax may react with the curable composition, reducing the amount of material that may potentially migrate from the adhesive or coating. Suitable functionalized waxes include but are not limited to Honeywell A-C.TM. 596; Clariant Licocene.TM. MA6252 and Licocene.TM. MA6452; and Westlake Epolene.TM. E-43.
[0161] In addition to physically protecting curing agents [Component (II)], tackifying resins (tackifiers) may be added to the curable compositions of the invention to modify the adhesive or coating properties, including but not limited to the viscosity, wetting behavior, adhesion, particularly to low energy surfaces, and viscoelastic behavior of the finished adhesive or coating composition. The tackifier resin selected may vary depending on the exact curable composition and the balance of properties needed in an application, such as peel strength, shear strength, and tack. These properties are important to adhesive compositions and the best advantages are achieved where the overall balance of these properties, both initially and over time in different environmental conditions, have been optimized.
[0162] Tackifier resins that may be present in the adhesive compositions include, without limitation, cycloaliphatic hydrocarbon resins, C5 hydrocarbon resins, C5/C9 hydrocarbon resins, aromatically modified C5 resins (commercially available as Piccotac.TM. resins, Eastman Chemical Company, Tenn., US), C9 hydrocarbon resins (commercially available as Picco.TM. resins, Eastman), pure monomer resins, e.g. copolymers of styrene with alpha-methyl styrene, vinyl toluene, para-methyl styrene, indene, and methyl indene (commercially available as Kristalex.TM. resins, Eastman), dicyclopentadiene (DCPD) resins, dicyclopentadiene based/containing resins, cyclopentadiene based/containing resins, terpene resins (commercially available as Sylvares.TM. resins, AZ Chem Holdings, LP, Jacksonville, Fla., US), terpene phenolic resins, terpene styrene resins, esters of rosin (commercially available as Permalyn resins, Eastman), esters of modified rosins, liquid resins of fully or partially hydrogenated rosins, fully or partially hydrogenated rosin esters (commercially available as Foral.TM. E and Foralyn.TM. resins, Eastman), fully or partially hydrogenated modified rosin resins, fully or partially hydrogenated rosin alcohols, fully or partially hydrogenated C5 resins, fully or partially hydrogenated C5/C9 resins, fully or partially hydrogenated DCPD resins (commercially available as Escorez.RTM. 5000-series resin, ExxonMobil Chemical Company, Tex., US), fully or partially hydrogenated dicyclopentadiene based/containing resins, fully or partially hydrogenated cyclopentadiene based/containing resins, fully or partially hydrogenated aromatically modified C5 resins, fully or partially hydrogenated C9 resins (commercially available as Regalite.TM. resins, Eastman), fully or partially hydrogenated pure monomer resins (e.g., copolymers or styrene with alpha-methyl styrene, vinyl toluene, para-methyl styrene, indene, and methyl indene) (commercially available as Regalrez.TM. resins, Eastman), fully or partially hydrogenated C5/cycloaliphatic resins (commercially available as Eastotac.TM. resins, Eastman), fully or partially hydrogenated C5/cycloaliphatic/styrene/C9 resins, fully or partially hydrogenated cycloaliphatic resins, and/or mixtures thereof.
[0163] The adhesive composition may be useful as a "warm" melt adhesive for those applications where hot melt adhesives are currently employed and applied by techniques known to those skilled in the art. The melting point and/or ring and ball softening point (RBSP) of one or more tackifier resins in the curable composition can be selected to suit the intended end use for the adhesive curable composition, where RBSP is measured by ASTM 06493-11 "Standard Test Methods for Softening Point of Hydrocarbon Resins and Rosin Based Resins by Automated Ring-and-Ball Apparatus." In various embodiments, the tackifier resin has a RBSP from -10.degree. C. to 160.degree. C. In some embodiments, the tackifier resin has an acid number in the range of 0 to 170, 0 to 30, 0 to 25, or 0 to 11 mg KOH/g. In some embodiments, tackifier resins with zero or low acid numbers are preferred to reduce interference with the amine crosslinker. In some embodiments, tackifier resins can have moderate or high acid numbers, for example, where the primary amine is buffered.
[0164] In other embodiments, tackifier resins with RBSP above the expected application temperature and with acid numbers of about 5 to about 10, or of about 11 to about 22, or of about 145 to about 165 mg KOH/g, can be added in order to act as a base scavenger at high temperatures and improve thermal stability of the cured composition as well as to provide improved adhesion. Non-limiting examples of tackifier resins that may be used include Poly-Pale.TM. (AN 155, RBSP 103.degree. C.), Dymerex.TM. (AN 145, RBSP 144.degree. C.), Foral.TM. AX-E (AN 165, RBSP 8.degree. C.), Permalyn.TM. 6110 (AN 13, RBSP 103.degree. C.), and Foralyn.TM. 90 (AN 8, RBSP 82.degree. C.) (Eastman).
[0165] In various embodiments, the adhesives can comprise at least 1, 2, 5, 8, or 10 and/or not more than 40, 30, 25, 20, or 15 weight percent of at least one tackifying resin based on the total weight of the composition. Moreover, the adhesives can comprise in the range of 2 to 40, 5 to 30, 8 to 25, or 10 to 20 weight percent of at least one tackifying resin based on the total weight of the composition.
[0166] In another embodiment, the curable adhesive composition of the present invention further comprises one or more adducts having two or more .beta.-ketoacetate functional groups such as, for example, 2-methyl-1,3-propanediol diacetoacetate; neopentyl glycol diacetoacetate; 2,2,4,4-tetramethylcyclobutane-1,3-diol diacetoacetate; or trimethylolpropane triacetoacetate; or any isomers thereof, or mixtures thereof. The adducts can function as reactive diluents to reduce the viscosity of the adhesive formulations, especially those of solventless formulations for extending their pot lives. The adducts can also modify the functionality of the .beta.-ketoacetate functional groups available for curing agents.
[0167] The curable adhesive composition of the invention is capable of reacting at higher temperature (above ambient temperature) with more control of shelf life in the presence of a protected curing agent. In this so-called 1K system, the present invention allows the mixing of the two components (AcAc resin and amine curing agent); the resin and the curing agent may not react when the curing agent is protected. The protected composition can be mixed and stored until the mixture is ready to be used and is activated (de-protected.)
[0168] The manner and order of protection of the curing agent(s) (amine) can be used to control the cure profile of the curable composition of the invention in both 1K and 2K systems.
[0169] The curable adhesive composition of the invention may be a 1K system with extended shelf life, and a controlled cure profile that provides desired green strength and is also capable of curing at higher temperature. For example, the curable adhesive composition of the invention may comprise a portion of a physically protected amine curing agent that is de-protected (activated) at 40.degree. C. and a portion of a physically protected amine curing agent that is de-protected at 60.degree. C. In this example 1K system, the present invention allows the mixing of the two components (resin and amine curing agent) and storage until the mixture is ready to be used. The inventive composition is activated in stages, with first de-protection at 40.degree. C. to allow a first stage of curing and formation of green strength, which is desired to hold assembled substrates in position, followed by second stage de-protection at 60.degree. C. to allow further cure of the composition.
[0170] The cure profile of the curable composition may also be controlled by a combination of chemical and/or physical protection strategies and with selection of curing agent(s). For example, the curable composition may comprise two curing agents with different reactivity that are protected by the same or different physical mechanism. The protection provides extended shelf life and/or pot life, and after activation/de-protection at the indicated condition(s), for example, the curing agent that provides faster reaction speed may provide a desired level of green strength, and the curing agent that provides lower reaction speed may provide the final level of cure and bond strength. In this manner, the formulator can obtain a desirable, superior balance of composition properties for a particular application method, substrate choice, and/or end use requirement.
[0171] The curable adhesive composition of the invention can include chemical and/or physical protection of the curing agent (amine). Upon de-protection/de-activation, the protecting agent can then be released allowing the composition(s) of the invention to achieve improvement in green bond strength and/or final adhesion bond strength as measured, for example, by 180 degree peel adhesion. Non-limiting examples of such materials can include functionalized waxes, tackifier resins, rosin acid tackifier resins, and rosin ester tackifier resins.
[0172] The curable composition of the invention may be essentially solvent free, solvent-less or solvent-based. In one embodiment, the solvent-based composition of the invention comprises an organic solvent. Suitable organic solvents include acetone, ethanol, ethyl acetate, butyl acetate, xylene, ketones (for example, methyl amyl ketone and methyl ethyl ketone), 2-butoxyethanol, ethyl-3-ethoxypropionate, toluene, butanol, cyclopentanone, cyclohexanone, and other volatile inert solvents typically used in adhesives. In one embodiment, the solvents useful in the invention are acetone, ethanol, ethyl acetate, butyl acetate, xylene, methyl amyl ketone, methyl ethyl ketone, and toluene.
[0173] The amount of solvents (excluding water) can range from 0% to 80%, 5% to 70%, 10% to 70%, 15% to 70%, 20% to 70%, 25% to 70%, 20% to 70%, 0% to 60%, 5% to 60%, 10% to 60%, 15% to 60%, 20% to 60%, 25% to 60%, 20% to 60%, 0% to 50%, 5% to 55%, 10% to 55%, 15% to 55%, 20% to 55%, 25% to 55%, 20% to 55%, 25% to 55%, 30% to 55%, 5% to 50%, 10% to 50%, 15% to 50%, 20% to 50%, 25% to 50%, 20% to 50%, 25% to 50%, 30% to 50%, 0% to 40%, 5% to 40%, 10% to 40%, 15% to 40%, 20% to 40%, 25% to 40%, 0% to 30%, 5% to 35%, 10% to 35%, 15% to 35%, 20% to 35%, or 25% to 35% by weight based on the total weight of the composition equaling 100 weight %. In one embodiment, the amount of solvents (excluding water) can range from 25 to 50, or 25 to 55, or 25 to 60% by weight based on the total weight of the composition equaling 100 weight %. The percentage by weight of solids (based on the total weight of the composition equaling 100 weight %) is the remainder of the weight % of the composition that does not comprise solvent(s). Therefore, the percentage by weight of solids based on the total weight of the composition equaling 100 weight % would be, for example, 20% to 100%, 30% to 95%, 30% to 90%, 30% to 85%, 30% to 80%, 30% to 75%, 30% to 70%, 40% to 100%, 40% to 95%, 40% to 90%, 40% to 85%, 40% to 80%, 40% to 75%, 40% to 70%, 50% to 100%, 50% to 95%, 50% to 90%, 50% to 85%, 50% to 80%, 50% to 75%, 50% to 70%, 50% to 65%, 50% to 60%, 55% to 100%, 55% to 95%, 55% to 90%, 55% to 85%, 55% to 80%, 55% to 75%, 55% to 70%, 55% to 65%, 60% to 100%, 60% to 95%, 60% to 90%, 60% to 85%, 60% to 80%, 60% to 75%, 60% to 70%, etc.
[0174] In one embodiment of this invention, the curable composition of this invention does not contain solvents or residues of solvents such as ethylene glycol and/or ether-containing compounds, for example, dimethyl ether, ethylene glycol diethyl ether, diethylene glycol dimethyl ether, or diethylene glycol diethyl ether.
[0175] In one embodiment of this invention, the curable composition of the invention can be water-free, and/or moisture-free, and/or can comprise substantially no water, and/or can contain essentially no water, or can contain one of the following percentages of water: 0 to 20 weight %, 0 to 19 weight %, 0 to 18 weight %, 0 to 17 weight %, 0 to 16 weight %, 0 to 15 weight %, 0 to 14 weight %, 0 to 13 weight %, 0 to 12 weight %, 0 to 11 weight %, 0 to 10 weight %, 0 to 9 weight %, 0 to 8 weight %, 0 to 7 weight %, 0 to 6 weight %, 0 to 5 weight %, 0 to 4 weight %, 0 to 3 weight %, 0 to 2 weight %, 0 to 1 weight %, 0.01 to 20 weight %, 0.01 to 19 weight %, 0.01 to 18 weight %, 0.01 to 17 weight %, 0.01 to 16 weight %, 0.01 to 15 weight %, 0.01 to 14 weight %, 0.01 to 13 weight %, 0.01 to 12 weight %, 0.01 to 11 weight %, 0.01 to 10 weight %, 0.01 to 9 weight %, 0.01 to 8 weight %, 0.01 to 7 weight %, 0.01 to 6 weight %, 0.01 to 5 weight %, 0.01 to 4 weight %, 0.01 to 3 weight %, 0.01 to 2 weight %, or 0.01 to 1 weight %, based on the total weight of the composition.
[0176] In one embodiment of this invention, the composition of this invention is not a water-borne or a water-dispersible composition.
[0177] In one embodiment of the invention, the composition of the invention does not contain: a polyvinyl alcohol, or a polyvinyl polymer, or a poly vinyl acetate polymer.
[0178] In one embodiment of this invention, the polyester resin of the composition of the invention is not an alkyd polyester resin and/or is not a polyester resin containing alkyd and/or fatty acid residues.
[0179] In one embodiment of this invention, the composition of the invention can be or, alternatively, is not a thermosetting composition.
[0180] In one embodiment of this invention, the composition of this invention does not contain polysaccharides and/or oxidized polysaccharides having aldehyde groups.
[0181] The compositions of the invention can comprise at least one additive selected from the group consisting of tackifiers, plasticizers, fillers, pigments, stabilizers, antioxidants, adhesion promoters, flame retardants, conductive agents, rheology modifiers and mixtures thereof and the process of preparing the curable compositions of the invention may comprise an additional step of blending in one or more of these additives.
[0182] In various embodiments, the compositions of the invention can comprise at least 0.1, 0.5, 1, 2, or 3 and/or not more than 5 weight percent of at least one antioxidant, based on the total weight of the composition equaling 100 weight %.
[0183] One or more fillers, such as but not limited to, talc, carbon black, calcium carbonate, titanium oxide and zinc oxide, may be incorporated into the curable compositions of the invention. In various embodiments, the adhesives of the invention can comprise at least 1, 5, 10, 20, 30, or 40 and/or not more than 80, 70, 60, 55, 50 or 45 weight percent of at least one filler, based on the total weight of the compositions equaling 100 weight %. Moreover, the adhesives of the invention can comprise in the range of 1 to 80, 5 to 70, 10 to 60, 20 to 55, 30 to 50, or 40 to 45 weight percent of at least one filler, based on the total weight of the compositions equaling 100 weight %.
[0184] Further, in some embodiments, the composition of the invention may exclude one or more additives typically found in conventional compositions. For example, in some cases, the adhesive formed using the compositions of the invention can be used as a pressure sensitive adhesive (e.g., tapes and labels) and nonwoven adhesive applications without addition of oils or plasticizers that can possibly volatilize, migrate, or be extracted. For example, in some cases, adhesive compositions of the present invention can include not more than about 2, not more than about 1.5, not more than about 1, not more than about 0.5, not more than about 0.25, not more than about 0.1, or not more than about 0.05 weight percent of one or more oils or plasticizers, based on the total weight of the adhesive composition equaling 100 weight %.
[0185] In some cases, the adhesive composition may be a liquid composition in water or solvent, applied to a surface and dried or permitted to dry, while, in other embodiments, the composition may be at or near 100 percent solids. In some embodiments, essentially no volatile organic compounds (VOCs) or no VOCs are released during the cure process. In certain embodiments, VOCs can be present in the adhesive compositions of the present invention in the amount less than 5 weight %, less than 4 weight %, less than 3 weight %, less than 2 weight %, or less than 1 weight % VOCs, based on the total weight of the composition.
[0186] The adhesive composition of the present invention may be prepared according to any suitable method, techniques and equipment. For example, the components of the adhesive composition may be blended in a mixer, an extruder, an aluminum can, and/or at the point of application, e.g. a head mixing system. In some cases, the components of the adhesive composition may be blended, optionally with a solvent, to form a mixture, which can then be cast onto a backing or other substrate and dried or cured or partially cured to form an article comprising the curable composition.
[0187] Furthermore, the adhesive composition may be shaped into a desired form, such as a tape or sheet, by an appropriate technique including casting, extrusion, or roll coating techniques (gravure, reverse roll, etc.). Alternatively, the composition may be applied to a substrate using conventional adhesive application equipment recognized in the art, e.g. curtain coating, slot-die coating, wire-wound rod coating, gravure coating, roll coating, knife coating, hot or "warm" melt coating. The adhesive composition may be applied as either a continuous or discontinuous coating or film or layer or sprayed through different nozzle and/or head configurations at different speeds using typical application equipment. The application may be followed by drying or heat treatment.
[0188] In one embodiment, there is provided a process for preparing a flexible film laminate comprising: (1) applying at least one of Components (I)-(II) of the compositions useful in the invention to the surface of a first substrate, (2) applying the remaining component(s) of said Components on the surface of a second substrate, (3) combining said substrates and then curing said combined components, thus forming a cured composition.
[0189] In another embodiment, the curable adhesive of the present invention is a laminating adhesive for flexible packaging. The curing temperature for such an adhesive is desirably a low temperature ranging from room temperature to about 80.degree. C.
[0190] After formulation, the curable adhesive can be applied to a substrate and subsequently laminated to another substrate. Suitable substrates include but are not limited to textile, fabric, mesh, film, poly(acrylonitrile butadiene styrene) (ABS), polypropylene (PP), glass-filled PP, talc-filled PP, impact-modified PP, polycarbonate (PC), PC-ABS, biaxially oriented polypropylene (BOPP), thermoplastic polyolefin (TPO) compounds, pigmented TPO compounds, filled TPO compounds, rubber-modified TPO compounds, paper, glass, plastic, metal, PVC (polyvinyl chloride), PET(polyethylene terephthalate), modified PET such as PETG (PET modified with 1,4-cyclohexanedimethanol) and PCTG, Mylar.TM. plastic, aluminum, leather, synthetic leather, nonwoven materials, foams, painted surfaces, printed surfaces, thermosets, thermoplastics, polymer films such as polyethylene, polypropylene, oriented polyethylene, oriented polypropylene; metallized plastic films; aluminum foil; wood; metals such as aluminum, steel or galvanized sheeting; glass; urethane elastomers; primed (painted) substrates, and laminates, blends or coated substrates comprising at least one of these materials. Any of these substrates may be untreated, corona treated, chemically treated, plasma treated, flame treated, rubber-modified, impact-modified, filled with e.g. talc or glass, pigmented with e.g. carbon black, chromium oxide or titanium oxide, or otherwise modified as known by those skilled in the art to provide improved properties to the substrate.
[0191] The curable adhesive can be coated onto a substrate using techniques known in the art, for example, by spraying, draw-down, roll-coating, brushing, nozzle dispensing, printing, etc. arid subsequently laminated to another substrate manually or by a roll-to-roll laminating machine. The coating and laminating process may be done at room temperature or elevated temperatures. The laminated films may be cured at room temperature or elevated temperatures. In various embodiments, the curable mixture of the present invention can cure at 20.degree. C. in various amounts of time. In one embodiment, the curable mixture of the present invention can cure at 20.degree. C. or higher in six days or less.
[0192] In some embodiments, the pot life of the curable mixture can be 5 minutes or more; or 10 minutes or more; or 25 minutes or more. Independently, in some embodiments, pot life can be 7 days or less; or 1 day or less; or 8 hours or less; or 2 hours or less; or 30 minutes or less.
[0193] In certain embodiments of the invention, the compositions of the invention can be prepared by a process comprising: (a) mixing at least one component having two or more .beta.-ketoester functional groups, and at least one component having two or more primary amine functional groups, and (b) after optional activation, curing the composition at 20.degree. C. or higher, for example, in six days or less, and (c) optionally, post-curing the composition at or above 20.degree. C.
[0194] In certain embodiments of the invention, the compositions of the invention can be prepared by a process comprising: (a) mixing at least one component having two or more .beta.-ketoester functional groups, and at least one component having two or more primary amine functional groups, and (b) after optional activation, curing the composition at or above 20.degree. C., or above 35.degree. C., or above 50.degree. C., or above 55.degree. C., or above 70.degree. C., or above 80.degree. C., or above 100.degree. C., or from 20.degree. C. to 180.degree. C., or from 50.degree. C. to 180.degree. C., or from 50.degree. C. to 80.degree. C. in any time known to one of ordinary skill in the art, for example, in six days or less, and (c) optionally, post-curing the composition at or above 20.degree. C., or above 35.degree. C., or above 55.degree. C., or above 70.degree. C., or above 80.degree. C., or above 100.degree. C., or from 20.degree. C. to 110.degree. C., or from 50.degree. C. to 110.degree. C.
[0195] In some embodiments, the curable composition of this invention can be cured after optional activation, for example, to de-protect a component, at temperatures above 20.degree. C., or above 35.degree. C., or above 50.degree. C., or above 55.degree. C., or above 70.degree. C., or above 80.degree. C., or above 100.degree. C. and can then be post-cured at a higher temperature(s) above 20.degree. C., or above 35.degree. C., or above 55.degree. C., or above 70.degree. C., or above 80.degree. C., or above 100.degree. C.
[0196] In some embodiments, the curable composition can be partially cured before being applied to a substrate, used in a laminate, molded or otherwise incorporated into an article.
[0197] In this invention, Brookfield viscosity can be measured using a Brookfield viscometer as described in ASTM D3236 "Apparent Viscosity of Hot Melt Adhesives and Coating Materials." Viscosity measurements are reported in centipoise (cP) units. In various embodiments, the curable compositions of the invention, for example, adhesive compositions, can exhibit a Brookfield viscosity in the range of 5 to 300,000 cP, or 300 to 200,000 cP, or 300 to 150,000 cP, or 300 to 100,000 cP, or 500 to 200,000 cP, or 500 to 150,000 cP, or 500 to 100,000 cP, or 500 to 90,000 cP, or 500 to 80,000 cP, or 500 to 50,000 cP, or 500 to 45,000 cP, or 500 to 40,000 cP, or 500 to 35,000 cP, or 500 to 30,000 cP, or 500 to 25,000 cP, or 500 to 20,000 cP, or 500 to 15,000 cP, or 500 to 10,000 cP, or 500 to 8,000 cP, or 500 to 5,000 cP, or 500 to 3,000 cP, or 1000 to 100,000 cP, or 1000 to 90,000 cP, or 1000 to 80,000 cP, or 1000 to 50,000 cP, or 1000 to 45,000 cP, or 1000 to 40,000 cP, or 1000 to 35,000 cP, or 1000 to 30,000 cP, or 1000 to 25,000 cP, or 1000 to 20,000 cP, or 1000 to 15,000 cP, or 1000 to 10,000 cP, or 1000 to less than 10,000 cP, or 1000 to 8,000 cP or 1000 to 5,000 cP, or 1000 to 3,000, or 5000 to 90,000 cP, or 5000 to 80,000 cP, or 5000 to 50,000 cP, or 5000 to 45,000 cP, or 5000 to 40,000 cP, or 5000 to 35,000 cP, or 5000 to 30,000 cP, or 5000 to 25,000 cP, or 5000 to 20,000 cP, or 5000 to 15,000 cP, or 5000 to 10,000 cP, or 5000 to less than 10,000 cP, or 5000 to 8,000 cP, or 10,000 to 90,000 cP, or 10,000 to 80,000 cP, or 10,000 to 50,000 cP, or 10,000 to 45,000 cP, or 10,000 to 40,000 cP, or 10,000 to 35,000 cP, or 15,000 to 90,000 cP, or 15,000 to 80,000 cP, or 15,000 to 50,000 cP, or 15,000 to 45,000 cP, or 15,000 to 40,000 cP, or 15,000 to 35,000 cP, or 20,000 to 90,000 cP, or 20,000 to 80,000 cP, or 20,000 to 50,000 cP, or 20,000 to 45,000 cP, or 20,000 to 40,000 cP, or 20,000 to 35,000 cP, or 25,000 to 90,000 cP, or 25,000 to 80,000 cP, or 25,000 to 50,000 cP, or 25,000 to 45,000 cP, or 25,000 to 40,000 cP, or 25,000 to 35,000, or 1 to 500 cP at application temperature. In certain embodiments, application temperature can be at 45.degree. C. or less.
[0198] In other embodiments, the curable adhesive compositions may exhibit a ring and ball softening point in the range of -10 to 180.degree. C.
[0199] In various embodiments, the curable adhesive compositions can have a peel strength in a 180 degree peel test of at least 2 lb/in or at least 5 lb/in as measured according to ISO 8510-2-2006 Part 2 at 5 mm/sec.
[0200] In various embodiments, the curable adhesive compositions can be applied in the range of about 0.5 gsm to about 200 gsm (gsm=grams per square meter). In embodiments where the curable composition will be used to bond substrates to each other, the add-on rate used will be suitable for generating laminates or composites with desired bond strength. The curable composition can be applied to one or both substrates before the substrates are brought into contact to form a composite, laminate or article. The article so formed may be optionally contacted with additional substrates, additional curable compositions, adhesives, and/or may be subjected to applied pressure and/or applied heat, in any order or combination without limitation.
[0201] The invention also provides a process for curing the curable mixture comprising the step of: contacting at least one AcAc-functionalized resin with at least one multifunctional amine, wherein said curable mixture is capable of curing after optional activation at 20.degree. C. in six days or less.
[0202] In various embodiments of the present invention, the curable composition can be utilized as an adhesive and can comprise at least one resin having two or more beta-ketoester groups, and at least one curing agent having two or more primary amine functional groups. Optionally, the curable adhesive composition can further comprise at least one tackifier resin, and/or at least one wax and/or at least one antioxidant.
[0203] The compositions of this invention can provide desirable properties for a variety of applications. In certain embodiments, the compositions of this invention are suitable for applications in the adhesives area, for example, automotive adhesives, structural adhesives, wood adhesives, and laminating adhesives, and applications in the coatings area, for example, automotive, industrial maintenance, marine craft, field-applied coatings, and furniture.
[0204] The curable compositions of the present invention can be used in a variety of adhesive compositions, including, but not limited to, automotive interior assembly adhesives, flexible laminating adhesives, rigid laminating adhesives, assembly adhesives, labelling adhesives, nonwoven adhesives, tape adhesives, pressure sensitive adhesives, structural adhesives, hygiene construction adhesives, hygiene elastic attachment adhesives, home repair adhesives, pressure sensitive adhesives, industrial adhesives, construction adhesives, medical adhesives, contact adhesives, hot melt adhesives, or solvent-based adhesives.
[0205] In one embodiment of the invention, there is provided a composition that is used at, or above, or near its glass transition temperature.
[0206] In one embodiment of the invention, there are provided cured compositions wherein AT is the difference between the temperature at use and the glass transition temperature (Tg) of the cured composition, and AT is from -30 to 60.degree. C. or from -30 to 10.degree. C.
[0207] In some embodiments, such as pressure sensitive adhesives, the curable compositions of the present invention can be characterized by adhesive strength by 180-degree peel test e.g. according to PSTC-101 or ISO 8510-2-2006 Part 2 at 5 mm/sec, cohesive strength and/or temperature resistance by static shear hold power testing (room temperature or elevated temperature, e.g., 40.degree. C. or 70.degree. C.) by PSTC-107 and/or by shear adhesion failure temperature (SAFT) by PSTC-17.
[0208] In some embodiments, the curable compositions of the present invention can be characterized by lap shear testing: ASTM D3163-01(2014) Standard Test Method for Determining Strength of Adhesively Bonded Rigid Plastic Lap-Shear Joints in Shear by Tension Loading. Impact strength can also be measured any method known in the art, for example, by pendulum or ball drop impact tests.
[0209] In some embodiments, the curable compositions of the present invention can be used in flexible packaging and characterized by tests such as DIN ISO 53357 Internal Adhesion, DIN ISO 55529 Sealed Seam Strength, DIN 53357 Bonding Adhesion, DIN 53504 Elongation at Tear and Tearing Tension, ASTM D1003 Transparency of film, ASTM D2578 Wetting Tension of Film Surface, ASTM F1249 Water Vapor Transmission Rate, ASTM F2622 or D3985 Oxygen Transmission Rate, and/or T-peel by ASTM F904-16 "Standard Test Method for Comparison of Bond Strength or Ply Adhesion of Similar Laminates Made from Flexible Materials."
[0210] The inventive compositions can exhibit improved heat resistance and/or improved adhesion over time, particularly after heat aging, as evidenced by tests such as elevated temperature aging of the adhered articles comprising the inventive compositions, followed by lap shear testing, by fiber tear testing, by peel testing, by peel adhesion failure temperature (PAFT) testing, by shear adhesion failure temperature (SAFT) testing, and/or by shear hold power testing at elevated temperatures such as 40.degree. C., 60.degree. C. 70.degree. C., 85.degree. C., 95.degree. C., 105.degree. C., or 120.degree. C. The adhered articles comprising the compositions of the invention can also exhibit improved humidity resistance as evidenced, for example, by aging at 95 to 100% relative humidity at 40.degree. C. for 24 to 144 hours followed by any of the above listed adhesion and cohesion tests at room temperature and/or at elevated temperature.
[0211] An article comprising the curable compositions of the invention may be an adhesive, a laminate, a tape, a label, a tag, a radio frequency identification (RAD) tag, a coating, a sealant, a film, a foam, a disposable hygiene article, a polyester composite, a glass composite, a fiberglass reinforced plastic, a wood-plastic composite, an extruded compound, a polyacrylic blended compound, a potting compound, a rubber compound, a motor vehicle molded part, a motor vehicle extruded part, a motor vehicle laminated part, a sheet molding compound (SMC) or dough molding compound (DMC), a woven textile, a nonwoven textile, a flexible packaging multilayer.
[0212] In one embodiment, there is provided a process wherein the composition of the invention is prepared by a process comprising: (a) mixing a first component comprising at least one resin having two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and a second component comprising at least one curing agent having at least two primary functional groups, salts thereof, or mixtures thereof and (b) curing the composition after optional activation between 20.degree. C. and 60.degree. C. in six days or less, and (c) optionally, post-curing the composition below 60.degree. C.
[0213] The curable adhesive composition may be prepared by a process comprising: (a) mixing a first component comprising at least one resin having two or more functional groups selected from the group consisting of .beta.-ketoester and malonate functional groups, and a second component comprising at least one curing agent having at least two primary amine functional groups, salts thereof, or mixtures thereof and (b) after optional activation, curing the composition at 20.degree. C. or higher in six days or less, and (c) optionally post-curing the composition at or above 20.degree. C.
[0214] In one embodiment, articles are provided comprising the above cured compositions where the bond is maintained after cure and/or post-cure at 60.degree. C.
[0215] The following examples further illustrate how the polyesters useful in the invention can be made and evaluated, and how the curable compositions useful in this invention can be made and evaluated and are intended to be purely exemplary of the invention and are not intended to limit the scope thereof. Unless indicated otherwise, parts are parts by weight, temperature is in degrees C. (Celsius) or is at room temperature, and pressure is at or near atmospheric.
EXAMPLES
[0216] In the Examples herein, analysis of the acetoacetate (AcAc) number of the resin employs a potentiometric titration method based on the titration of the proton in the AcAc group with a strong base, tetrabutylammonium hydroxide solution (TBAOH) in methanol. The titration was conducted on a titrator (904 Titrando, Metrohm AG, US) equipped with Tiamo software and a pH electrode (DG116-solvent, Mettler Toledo, US) as sensing probe. Depending on the expected AcAc number of the sample, 0.05 to 0.5 grams sample was weighted to a titration cell and stirred to dissolve in 35 mL pyridine at room temperature. The sample solution was titrated to the endpoint at pH around 16, which was determined by Tiamo software or manually. The AcAc number, reported as mg KOH/g sample, was calculated from the volume of TBAOH used at the titration endpoint, its normality, and weight of sample.
Example 1. Synthesis of Acetoacetate Functional Polyester 1 (AcAc Polyester 1 or AcAc Resin 1)
Hydroxyl Functional Polyester 1:
[0217] A 2-L kettle with a four-neck lid was equipped with a mechanical stirrer, a thermocouple, a heated partial condenser (115.degree. C.), a Dean-Stark trap, and a chilled condenser (15.degree. C.). To the flask were charged neopentyl glycol (NPG) (178.6 g), 2-methyl-1,3-propanediol (MPDiol) (154.6 g), trimethylolpropane (TMP) (131.5 g), adipic acid (AD) (511.5 g), and the acid catalyst, Fascat-4100 (Arkema Inc.) (0.98 g). The reaction was allowed to react under nitrogen at 170.degree. C. for one hour, at 200.degree. C. for one hour, and at 230.degree. C. for about 3 hours to yield a clear, viscous mixture. A total of 126 g of the distillate was collected in the Dean-Stark trap. The resulting mixture was allowed to cool to 120.degree. C. for the reaction with t-butyl acetoacetate below. Acid number: <1.0 mg KOH/g; hydroxyl number: 165.5 mg KOH/g; glass transition temperature (Tg): -49.6.degree. C.; number average molecular weight (Mn): 2050 g/mole; weight average molecular weight (Mw): 5921 g/mole.
Acetoacetate Functional Polyester 1:
[0218] The next synthesis was aimed to convert hydroxyl number of 150 mg KOH/g of the above hydroxyl functional polyester (1) to acetoacetate number of 150 mg KOH/g. To the above hydroxyl-functional polyester 1 (850g) was added t-butyl acetoacetate (359 g). The mixture was gradually heated and allowed to react at 120.degree. C. for 20 minutes and at 140.degree. C. for two hours. A total of 14 4 g of the condensate (t-butanol) was collected in the Dean-Stark adapter. The resulting viscous resin was allowed to cool and subsequently collected. Glass transition temperature (Tg): -51.degree. C.; number average molecular weight (Mn): 1804 g/mole; weight average molecular weight (Mw): 5790 g/mole; AcAc number 127 mgKOH/g.
Example 2. Synthesis of Acetoacetate Functional Polyester 2 (AcAc Polyester 2 or AcAc Resin 2)
Acetoacetate Functional Polyester 2:
[0219] To the above hydroxyl-functional polyester 1 (912g) was added t-butyl acetoacetate (321 g). The mixture was gradually heated and allowed to react at 120.degree. C. for 20 minutes and at 140.degree. C. for two hours. The condensate (t-butanol) was collected in the Dean-Stark adapter. The resulting viscous resin was allowed to cool and subsequently collected. Glass transition temperature (Tg): -51.degree. C.; number average molecular weight (Mn): 1771 g/mole; weight average molecular weight (Mw): 5848 g/mole; AcAc number 107 mgKOH/g.
TABLE-US-00001 TABLE 1 Abbreviations of amines used. Name CAS # Other names IPDA 2855-13-2 5-Amino-1,3,3- trimethylcyclohexanemethanamine; isophorone diamine MPMDA 15520-10-2 2-Methyl-1,5-pentanediamine; 2-methylpentamethylenediamine MACM 6864-37-5 4,4'-Methylenebis[2-methylcyclohexan- amine] TETA 112-24-3 Triethylenetetramine Epicure 940 25620-58-0 2,2,4(2,4,4)-Trimethyl-1,6-hexanediamine Jeffamine 39423-51-3 Amine-terminated polypropylene glycol T-403 trimethylolpropane ether 1,3-CHBMA 2579-20-6 1,3-Cyclohexanebis(methylamine); 1,3-bis(aminomethyl)cyclohexane Curing agent amount is given as equivalents (moles amine functional groups to moles AcAc functional groups in resin). E.g., 1.0 equiv means that there is one mole of amine functional groups in the composition per mole AcAc functional groups in the resin.
Examples 3-20: Demonstration of Cure Using Various Diamine Curing Agents
[0220] In Examples 3-20, the AcAc Polyester 1 or 2 was mixed in a vial with an amine curing agent, as described in Table 2. The samples were observed to see if the compositions thickened, how long they were of low enough viscosity to be workable/stirrable, and if they cured. As can be seen in Table 2, all compositions in which 1.0 equivalent of amine curing agent was added cured (Examples 3-6, 10-17). Most thickened quickly, and some were not workable after 2-4 minutes, indicating that rapid curing was taking place. Compositions with less than one equivalent of amine curing agent (Examples 7, 8, 18, 19; 0.33-0.8 equiv curing agent) also cured. However, when the amount of curing agent was reduced further to 0.25 equivalents (Examples 9, 20), compositions did not cure, even after 3-4 days.
[0221] Examples 17-19 show that for the same resin and curing agent, as the amount of curing agent is reduced from 1.0 to 0.5 to 0.33 equivalents, the time that the composition remains workable increased from less than 3.5 minutes, to less than 6 minutes, to more than 12 minutes. Thus, the amount of curing agent added can be used to modify the time that the composition is workable, while still resulting in a cured composition.
TABLE-US-00002 TABLE 2 Curable compositions comprising AcAc resins and amine curing agents. Exam- AcAc Amine ple # Resin curing agent Observation 3 1 IPDA Thickened. Still workable after 3 minutes. (1.0 equiv) Cured within 24 h. 4 1 MPMDA Not workable after 2 minutes. Cured. (1.0 equiv) 5 1 MACM Thickened. Still workable after 3 minutes. (1.0 equiv) Cured within 24 h. 6 1 TETA Not workable after 2 minutes. Cured. (1.0 equiv) 7 1 Epikure 940 Gelled fast. Not workable after 3 minutes. (0.8 equiv) Cured. 8 1 Epikure 940 Thickened. Still workable after 3 minutes. (0.65 equiv) Cured in less than 3 days. 9 1 Epikure 940 Thickened. Still workable after 3 minutes. (0.25 equiv) Was not cured after 3 days. 10 1 Jeffamine Thickened. Still workable after 3 minutes. T-403 Cured in less than 3 days. (1.0 equiv) 11 2 IPDA Thin after 4 minutes. Cured within 24 h. (1.0 equiv) 12 2 MPMDA Workable after 5 minutes. Set up after 10 (1.0 equiv) minutes. Cured. 13 2 MACM Thin after 5 minutes. Cured within 24 h. (1.0 equiv) 14 2 Epikure 940 Thin after 8 minutes. Cured within 24 h. (1.0 equiv) 15 2 Jeffamine Thin after 9 minutes. Cured after 3 hours. T-403 (1.0 equiv) 16 2 1,3-CHBMA Not workable after 6 minutes. Cured. (1.0 equiv) 17 2 TETA Not workable after 3.5 minutes. Cured. (1.0 equiv) 18 2 TETA Not workable after 6 minutes. Cured. (0.5 equiv) 19 2 TETA Still workable after 12 minutes. Cured (0.33 equiv) within 24 h. 20 2 TETA Still workable after 10 minutes. After 4 (0.25 equiv) days still gooey, not cured.
Examples 21-23: Peel Strength of Cured Compositions and Thermal Stability
[0222] Compositions were prepared by mixing AcAc Polyester 1 with an amine curing agent as described in Table 3. To prepare laminates, a 10 mil drawdown bar was used to form a film (target thickness: 4 mil) of the composition on solid substrates (Trinseo Magnum 3325 ABS, from Standard Plaque Inc., Melvindale, Mich., 1''.times.6''.times.0.125''). A mesh fabric (Teflon-coated 3-mesh spacer fabric, T5977-0370-1480-01T1, obtained from Mueller Textiles) was placed over the composition, and a handheld roller was rolled over the sample twice to laminate the sample. The samples were cured at ambient conditions for 7 days. Peel testing was done either after 7 days cure or after an additional 7 day post-cure at 80.degree. C.
[0223] The samples were tested for 180-degree peel strength following ISO 8510-2-2006 Part 2 at 5 mm/s (12 inch/minute) crosshead displacement rate using either an MTS Criterion Universal Tensile Tester model C43-104E, 500 Newton load cell (MTS Systems Corporation, Eden Prairie, Minn., US) or an Instron Model 4201. A minimum of three samples of each composition were tested and the average and standard deviation reported.
[0224] Examples 21-23 show that laminates with the curable compositions can be useful as adhesives, and some have peel strengths greater than 15 N/25 mm (Examples 21-22). Additionally, the peel strength did not change significantly after ageing at 80.degree. C. for 7 days, indicating that the compositions are thermally stable at these temperatures. The desired level of peel strength can be achieved by using different curing agents.
TABLE-US-00003 TABLE 3 180 degree peel strength of cured compositions. Peel strength Peel strength after 7 day after 7 day RT cure + Example AcAc Amine curing RT cure 7 days at 80.degree. C. # Resin agent (N/25 mm) (N/25 mm) 21 1 IPDA 17.9 (1.7) 21.7 (2.8) (1.0 equiv) 22 1 MACM 25.8 (2.5) 20.3 (3.1) (1.0 equiv) 23 1 Jeffamine 6.4 (0.4) 8.1 (1.3) T-403 (1.0 equiv)
[0225] The invention has been described in detail with reference to the embodiments described herein, but it will be understood that variations and modifications can be effected within the scope of the invention.
* * * * *
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.