System And Methods For Multiplexed Analysis Of Cellular And Other Immunotherapeutics
MACKAY; Sean ; et al.
U.S. patent application number 16/332627 was filed with the patent office on 2020-05-28 for system and methods for multiplexed analysis of cellular and other immunotherapeutics. The applicant listed for this patent is IsoPlexis Corporation. Invention is credited to Emily BETTINI, Sean MACKAY, Colin NG.
| Application Number | 20200166518 16/332627 |
| Document ID | / |
| Family ID | 60083408 |
| Filed Date | 2020-05-28 |






View All Diagrams
| United States Patent Application | 20200166518 |
| Kind Code | A1 |
| MACKAY; Sean ; et al. | May 28, 2020 |
SYSTEM AND METHODS FOR MULTIPLEXED ANALYSIS OF CELLULAR AND OTHER IMMUNOTHERAPEUTICS
Abstract
Disclosed are methods of identifying a secretome from a subject cell within a heterogeneous cell population when the subject cell contacts a target cell (e.g. a subject immune cell contacts a target cancer cell) or a stimulatory agent and methods of using the identified secretome to identify cells that are safe and efficacious for cellular therapies, including adoptive
| Inventors: | MACKAY; Sean; (New Haven, CT) ; NG; Colin; (Branford, CT) ; BETTINI; Emily; (Waterbury, CT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 60083408 | ||||||||||
| Appl. No.: | 16/332627 | ||||||||||
| Filed: | September 12, 2017 | ||||||||||
| PCT Filed: | September 12, 2017 | ||||||||||
| PCT NO: | PCT/US2017/051223 | ||||||||||
| 371 Date: | March 12, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62393612 | Sep 12, 2016 | |||
| 62431318 | Dec 7, 2016 | |||
| 62480147 | Mar 31, 2017 | |||
| 62480752 | Apr 3, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G01N 33/574 20130101; G01N 33/5091 20130101; G01N 33/5014 20130101; G01N 2800/7095 20130101; G01N 2800/52 20130101; G01N 33/68 20130101; G01N 33/505 20130101 |
| International Class: | G01N 33/68 20060101 G01N033/68; G01N 33/50 20060101 G01N033/50 |
Claims
1. A method of identifying a secretome from a subject cell within a heterogeneous cell population comprising: (a) contacting the subject cell and a target cell or a stimulatory agent in at least one chamber of a plurality of chambers, wherein the chamber is in fluid communication with an antibody panel and wherein the antibody panel is removably attached to the chamber; (b) maintaining the subject cell and the target cell or the stimulatory agent in the chamber under conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex; (c) removing the antibody panel from the chamber; wherein the at least one chamber comprises cell media that maintains the viability of the subject cell from step (a) through (c); and (d) imaging the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, thereby identifying the secretome of the subject cell when the subject cell contacts the target cell or the stimulatory agent.
2. A method of identifying a secretome from a subject cell within a heterogeneous cell population comprising: (a) contacting the subject cell and a target cell or a stimulatory agent under conditions sufficient to permit stimulation of the subject cell; (b) introducing the subject cell to at least one chamber of a plurality of chambers, wherein the chamber is in fluid communication with an antibody panel and wherein the antibody panel is removably attached to the chamber; (c) maintaining the subject cell in the chamber under conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex; wherein the at least one chamber comprises cell media that maintains the viability of the subject cell from step (b) through (c); and (d) removing the antibody panel from the chamber; and (e) imaging the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, thereby identifying the secretome of the subject cell following contact with the target cell or the stimulatory agent.
3. The method of claim 2, further comprising the step of disrupting contact between the subject cell and the target cell or the stimulatory agent.
4. The method of claim 2, wherein the subject cell and the target cell are comprised by a composition and wherein the subject cell and the target cell or the stimulatory agent are in fluid communication.
5. The method of claim 4, further comprising the step of depleting the target cell or the stimulatory agent from the composition.
6. The method of claim 1, wherein the heterogeneous cell population is a functionally heterogeneous cell population.
7. The method of claim 1, wherein the functionally heterogeneous cell population comprises at least two cells that produce a secretome in response to a stimulus, wherein the first cell produces a first secretome, wherein the second cell produces a second secretome, and wherein the first secretome and the second secretome are not identical.
8. The method of claim 1, wherein the functionally heterogeneous cell population comprises one or more immune cells.
9. The method of claim 8, wherein the one or more immune cells comprise a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil.
10. (canceled)
11. The method of claim 9, wherein the T-lymphocyte expresses a non-naturally occurring antigen receptor.
12. The method of claim 7, wherein the T-lymphocyte expresses a Chimeric Antigen Receptor (CAR).
13. (canceled)
14. The method of claim 1, wherein the subject cell is an immune cell.
15. The method of claim 14, wherein the immune cell is a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil.
16. (canceled)
17. The method of claim 15, wherein the T-lymphocyte expresses a non-naturally occurring antigen receptor.
18. The method of claim 17, wherein the T-lymphocyte expresses a Chimeric Antigen Receptor (CAR).
19. The method of claim 1, wherein the functionally heterogeneous cell population comprises one or more neuronal cells.
20. (canceled)
21. The method of claim 1, wherein the subject cell is a neuronal cell.
22. (canceled)
23. The method of claim 1, wherein the functionally heterogeneous cell population comprises one or more endocrine cells.
24. (canceled)
25. The method of claim 1, wherein the subject cell is an endocrine cell.
26. (canceled)
27. The method of claim 1, wherein the functionally heterogeneous cell population comprises one or more exocrine cells.
28.-29. (canceled).
30. The method of claim 1, wherein the subject cell is an exocrine cell.
31.-32. (canceled)
33. The method of claim 1, wherein the step of contacting the subject cell and the target cell or the stimulatory agent in a chamber comprises direct contact of the subject cell and the target cell.
34. The method of claim 1, wherein the step of contacting the subject cell and the target cell or the stimulatory agent in a chamber comprises indirect contact of the subject cell and the target cell.
35. The method of claim 34, wherein the indirect contact comprises fluid communication between the subject cell and the target cell or the stimulatory agent.
36. The method of claim 34, wherein the indirect contact comprises communication between the subject cell and the target cell or the stimulatory agent through a natural or artificial extracellular matrix.
37. The method of claim 34, wherein the indirect contact comprises communication between the subject cell and the target cell or the stimulatory agent through an intermediate cell.
38. The method of claim 1, wherein the target cell is a cancer cell.
39. The method of claim 38, wherein the cancer cell is a primary cancer cell or a cultured cancer cell.
40. The method of claim 39, wherein the primary cancer cell is metastatic.
41. The method of claim 1, wherein the target cell is a B-lymphocyte.
42. The method of claim 1, wherein the target cell is a bacteria, yeast, or microbe.
43. The method of claim 1, wherein the target cell is an infected cell.
44. (canceled)
45. The method of claim 1, wherein the target cell is a host cell.
46. The method of claim 47, wherein the host cell comprises any cell isolated or derived from the same individual as the subject cell.
47. The method of claim 45, wherein the host cell perpetuates or is a target of an autoimmune response.
48. (canceled)
49. The method of claim 45, wherein the host cell is a functional cell and wherein the host cell does not stimulate an autoimmune response.
50. The method of claim 1, wherein the stimulatory agent comprises a stimulatory antibody.
51.-54. (canceled)
55. The method of claim 50, wherein the stimulatory antibody specifically binds an epitope of a T cell regulator protein.
56. The method of claim 55, wherein the T cell regulator protein comprises programmed cell death protein 1 (PD-1).
57. (canceled)
58. The method of claim 1, wherein the stimulatory agent comprises a stimulatory ligand.
59. The method of claim 58, wherein the stimulatory ligand comprises programmed death ligand 1 (PD-L1).
60. The method of claim 1, wherein each antibody of the antibody panel is attached to a surface that is removably attached to the chamber.
61. The method of claim 60, wherein each antibody of the antibody panel is attached to the surface to form a repeating pattern and wherein each chamber of the plurality of chambers comprises a repeat of the pattern.
62. The method of claim 1, wherein the conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one and an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, comprise 5% CO.sub.2 and 37.degree. C. for a period of at least 2 hours.
63. The method of claim 62, wherein the period is at least 4 hours, at least 8 hours, at least 12 hours, at least 16 hours, or at least 24 hours.
64. (canceled)
65. The method of claim 1, wherein the at least one chamber comprises a cell media that maintains the viability of the subject cell from step (a) through (c).
66. The method of claim 1, wherein the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate diminished or decreasing cell function or cell viability.
67. The method of claim 1, wherein the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate(s) augmented or increasing inflammation.
68. The method of claim 1, wherein the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate(s) increased cell activity or cellular stimulation.
69. The method of claim 1, further comprising determining a Polyfunctional Strength Index (PSI).
70. The method of claim 69, wherein the Polyfunctional Strength Index is the product of a percentage of polyfunctional subject cells within the heterogeneous cell population and an average signal intensity of two or more cytokines.
71. The method of claim 70, wherein the polyfunctional subject cells, at a single cell level, secrete at least two cytokines.
72. The method of claim 71, wherein the at least two cytokines produced by each of the polyfunctional subject cells and the two or more cytokines of the average signal intensity comprise the same cytokines.
73. The method of claim 71, wherein the at least two cytokines produced by each of the polyfunctional subject cells and the two or more cytokines of the average signal intensity consist of the same cytokines.
74. The method of claim 69, wherein an increase in the PSI indicates an increase in the potency of the polyfunctional subject cells.
75.-136. (canceled)
Description
RELATED APPLICATIONS
[0001] This application claims priority to U.S. Patent Application No. 62/393,612, filed Sep. 12, 2016, U.S. Patent Application No. 62/431,318, filed Dec. 7, 2016, U.S. Patent Application No. 62/480,147, filed Mar. 31, 2017, and U.S. Patent Application No. 62/480,752, filed Apr. 3, 2017, the contents of each of which are herein incorporated by reference in their entirety.
FIELD OF THE DISCLOSURE
[0002] The disclosure is directed to molecular biology, and more, specifically, to methods of identifying the contents of the secretome of a subject cell and the use of the secretome for determining the safety and efficacy of the subject cell as a cell-based therapy.
BACKGROUND
[0003] There have been long-felt but unmet needs in the art for a system and methods of identifying cells suitable for use in a cellular therapy as well as methods of evaluating the safety and efficacy of these cells ex vivo or in vitro in a highly multiplexed reaction that can simultaneously analyze thousands of single cells from a heterogeneous cell population. The disclosure provides a system and methods to solve these long-felt but unmet needs.
SUMMARY
[0004] The systems of the disclosure provide a highly multiplexed method of evaluating the secretome of individual cells in a functionally heterogeneous population following contact with a target cell or a stimulatory agent. Analysis of the composition of the secretome of the subject cell may be used to determine the subject cell's identity, viability, safety and efficacy when used in a cell-based therapy. Cellular therapies may include autologous or allogeneic cells. Cellular therapies may include modified cells, including, but not limited to T-cells that express at least one artificial or chimeric antigen receptor.
[0005] The disclosure provides a method of identifying a secretome from a subject cell within a heterogeneous cell population comprising: (a) contacting the subject cell and a target cell or a stimulatory agent in at least one chamber of a plurality of chambers, wherein the chamber is in fluid communication with an antibody panel and wherein the antibody panel is removably attached to the chamber; (b) maintaining the subject cell and the target cell or the stimulatory agent in the chamber under conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex; (c) removing the antibody panel from the chamber; and (d) imaging the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, thereby identifying the secretome of the subject cell when the subject cell contacts the target cell or the stimulatory agent.
[0006] The disclosure provides a method of identifying a secretome from a subject cell within a heterogeneous cell population comprising: (a) contacting the subject cell and a target cell or a stimulatory agent under conditions sufficient to permit stimulation of the subject cell; (b) introducing the subject cell to at least one chamber of a plurality of chambers, wherein the chamber is in fluid communication with an antibody panel and wherein the antibody panel is removably attached to the chamber; (c) maintaining the subject cell in the chamber under conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at 1least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex; (d) removing the antibody panel from the chamber; and (e) imaging the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, thereby identifying the secretome of the subject cell following contact with the target cell or the stimulatory agent. In certain embodiments, this method further comprises the step of disrupting contact between the subject cell and the target cell or the stimulatory agent. In certain embodiments of this method, the subject cell and the target cell or the stimulatory agent are comprised by a composition and wherein the subject cell and the target cell or the stimulatory agent are in fluid communication. In certain embodiments, including those wherein the subject cell and the target cell or the stimulatory agent are comprised by a composition and wherein the subject cell and the target cell or the stimulatory agent are in fluid communication, the method further comprises the step of depleting the target cell or the stimulatory agent from the composition.
[0007] In certain embodiments of the systems and methods of the disclosure, the heterogeneous cell population is a functionally heterogeneous cell population. In certain embodiments, the functionally heterogeneous cell population may comprise at least two cells that produce a secretome in response to a stimulus, wherein the first cell produces a first secretome, wherein the second cell produces a second secretome, and wherein the first secretome and the second secretome are not identical.
[0008] Secretomes of the disclosure may comprise one or more peptides, polypeptides, proteins, small molecules, and ions. In certain embodiments, the secretome comprises one or more peptides, polypeptides or proteins. In certain embodiments, the secretome comprises one or more small molecules. In certain embodiments, the secretome comprises one or more ions. When the secretome comprises a small molecule or an ion, detectable labels may be used in addition or in place of antibodies to identify, quantify, or otherwise analyze the small molecule and ions of the secretome.
[0009] Secretomes of the disclosure may be released from a subject cell actively or passively. For example, secretomes of the disclosure may be released from a subject cell via a vesicle, an intercellular gap junction and/or a transmembrane channel or pump.
[0010] In certain embodiments of the systems and methods of the disclosure, the functionally heterogeneous cell population comprises one or more immune cells. In certain embodiments, the one or more immune cells comprise a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil. In certain embodiments, the T-lymphocyte comprises a naive T-lymphocyte, an activated T-lymphocyte, an effector T-lymphocyte, a helper T-lymphocyte, a cytotoxic T-lymphocyte, a gamma-delta T-lymphocyte, a regulatory T-lymphocyte, a memory T-lymphocyte, or a memory stem T-lymphocyte. In certain embodiments, the T-lymphocyte expresses a non-naturally occurring antigen receptor. In certain embodiments, the T-lymphocyte expresses a Chimeric Antigen Receptor (CAR).
[0011] In certain embodiments of the systems and methods of the disclosure, the functionally heterogeneous cell population comprises one or more immune cells. In certain embodiments, the one or more immune cells comprise a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil. In certain embodiments, the B-lymphocyte comprises a plasmablast, a plasma cell, a memory B-lymphocyte, a regulatory B cell, a follicular B cell, or a marginal zone B cell.
[0012] In certain embodiments of the systems and methods of the disclosure, the subject cell is an immune cell. In certain embodiments, the immune cell comprises a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil. In certain embodiments, the T-lymphocyte comprises a naive T-lymphocyte, an activated T-lymphocyte, an effector T-lymphocyte, a helper T-lymphocyte, a cytotoxic T-lymphocyte, a gamma-delta T-lymphocyte, a regulatory T-lymphocyte, a memory T-lymphocyte, or a memory stem T-lymphocyte. In certain embodiments, the T-lymphocyte expresses a non-naturally occurring antigen receptor. In certain embodiments, the T-lymphocyte expresses a Chimeric Antigen Receptor (CAR).
[0013] In certain embodiments of the systems and methods of the disclosure, the subject cell is an immune cell. In certain embodiments, the immune cell comprises a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, or a basophil. In certain embodiments, the B-lymphocyte comprises a plasmablast, a plasma cell, a memory B-lymphocyte, a regulatory B cell, a follicular B cell, or a marginal zone B cell.
[0014] In certain embodiments of the systems and methods of the disclosure, the functionally heterogeneous cell population comprises one or more neuronal cells. In certain embodiments, the one or more neuronal cells comprise a neuron, a glial cell, an astrocyte, a satellite cell, or an enteric glial cell.
[0015] In certain embodiments of the systems and methods of the disclosure, the subject cell is a neuronal cell. In certain embodiments, the neuronal cell comprises a neuron, a glial cell, an astrocyte, a satellite cell, or an enteric glial cell.
[0016] In certain embodiments of the systems and methods of the disclosure, the functionally heterogeneous cell population comprises one or more endocrine cells. In certain embodiments, the one or more endocrine cells are isolated or derived from a pineal gland, a pituitary gland, a pancreas, an ovary, a testicle, a thyroid gland, a parathyroid gland, a hypothalamus, or an adrenal gland.
[0017] In certain embodiments of the systems and methods of the disclosure, the subject cell is an endocrine cell. In certain embodiments, the endocrine cell is isolated or derived from a pineal gland, a pituitary gland, a pancreas, an ovary, a testicle, a thyroid gland, a parathyroid gland, a hypothalamus, or an adrenal gland.
[0018] In certain embodiments of the systems and methods of the disclosure, the functionally heterogeneous cell population comprises exocrine cells. In certain embodiments, the exocrine cell is isolated or derived from a salivary gland, a sweat gland or a component of the gastrointestinal tract. Components of the gastrointestinal tract may comprise a mouth, a stomach, a small intestine, and a large intestine.
[0019] In certain embodiments of the systems and methods of the disclosure, the subject cell is an exocrine cell. In certain embodiments, the exocrine cell is isolated or derived from a salivary gland, a sweat gland or a component of the gastrointestinal tract. Components of the gastrointestinal tract may comprise a mouth, a stomach, a small intestine, and a large intestine.
[0020] In certain embodiments of the systems and methods of the disclosure, the step of contacting the subject cell and the target cell or the stimulatory agent in a chamber comprises direct contact of the subject cell and the target cell or the stimulatory agent.
[0021] In certain embodiments of the systems and methods of the disclosure, the step of contacting the subject cell and the target cell or the stimulatory agent in a chamber comprises indirect contact of the subject cell and the target cell or the stimulatory agent. In certain embodiments, the indirect contact comprises fluid communication between the subject cell and the target cell or the stimulatory agent. In certain embodiments, the indirect contact comprises communication between the subject cell and the target cell or the stimulatory agent through a natural or artificial extracellular matrix. In certain embodiments, the indirect contact comprises communication between the subject cell and the target cell or the stimulatory agent through an intermediate cell.
[0022] In certain embodiments of the systems and methods of the disclosure, the target cell is a cancer cell. In certain embodiments, the cancer cell is a primary cancer cell or a cultured cancer cell. In certain embodiments, the primary cancer cell is metastatic.
[0023] In certain embodiments of the systems and methods of the disclosure, the target cell is a B-lymphocyte.
[0024] In certain embodiments of the systems and methods of the disclosure, the target cell is a bacteria, yeast, or microbe.
[0025] In certain embodiments of the systems and methods of the disclosure, the target cell is an infected cell. Exemplary infected cells may have contacted or may have been exposed to a virus, bacteria, yeast, or microbe.
[0026] In certain embodiments of the systems and methods of the disclosure, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell perpetuates an autoimmune response.
[0027] In certain embodiments of the systems and methods of the disclosure, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell is a target of an autoimmune response.
[0028] In certain embodiments of the systems and methods of the disclosure, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell is a functional cell and wherein the host cell does not stimulate an autoimmune response. The term "functional" cell is meant to describe a viable cell that does not contribute to a disease or disorder in the host. Alternatively, or in addition, the term "functional" cell may describe a cell without any known mutations that cause a disease or disorder in the host. For example, a functional cell may be non-cancerous and/or non-autoimmune.
[0029] In certain embodiments of the systems and methods of the disclosure, the stimulatory agent comprises a stimulatory antibody. In certain embodiments, the stimulatory antibody is a monoclonal antibody. In certain embodiments, the monoclonal antibody is a fully human antibody. In certain embodiments, the monoclonal antibody is a humanized antibody, a chimeric antibody, a recombinant antibody or a modified antibody. In certain embodiments, the modified antibody comprises one or more sequence variations when compared to a fully human version of an antibody having the same epitope specificity, one or more modified or synthetic amino acids, or a chemical moiety to enhance a stimulatory function. In certain embodiments, the stimulatory antibody specifically binds an epitope of a T cell regulator protein. In certain embodiments, the T cell regulator protein comprises programmed cell death protein 1 (PD-1). In certain embodiments, the stimulatory antibody comprises Nivolumab or a biosimilar thereof.
[0030] In certain embodiments of the systems and methods of the disclosure, the stimulatory agent comprises a stimulatory ligand. In certain embodiments, the stimulatory ligand comprises programmed death ligand 1 (PD-L1).
[0031] In certain embodiments of the systems and methods of the disclosure, each antibody of the antibody panel is attached to a surface that is removably attached to the chamber.
[0032] In certain embodiments of the systems and methods of the disclosure, each antibody of the antibody panel is attached to the surface to form a repeating pattern and wherein each chamber of the plurality of chambers comprises a repeat of the pattern. Antibody panels of the disclosure form patterns in which each repeat comprises the full panel of antibodies. For examples, if the antibody panel comprises antibodies "a", "b" and "c", then each repeat of the pattern also comprises at least one of antibody "a", "b" and "c". The pattern need only have a size scale such that each chamber aligns with at least one repeat of the pattern. In preferred embodiments, the pattern need only have a size scale such that each chamber aligns with one repeat of the pattern. When additional detectable labels are added to the panel to identify, capture or quantify secreted small molecules and/or ions, the detectable labels also repeat by the same rules set out for the antibody pattern.
[0033] In certain embodiments of the systems and methods of the disclosure, the conditions sufficient to permit (1) the subject cell to secrete at least one of a peptide, polypeptide, and protein and (2) at least one antibody of the antibody panel specific for the at least one protein to bind the at least one peptide, polypeptide, or protein, forming at least one of an antibody:secreted peptide, antibody:secreted polypeptide, or an antibody:secreted protein complex, may comprise 5% CO.sub.2 and 37.degree. C. for a period of 2 hours, about 2 hours or at least 2 hours. Alternatively, the period may be 4 hours, about 4 hours or at least 4 hours; 8 hours, about 8 hours or at least 8 hours; 12 hours, about 12 hours or at least 12 hours; 16 hours, about 16 hours or at least 16 hours; or 24 hours, about 24 hours or at least 24 hours. In certain embodiments, the period is 16 hours, about or at least 16 hours.
[0034] In certain embodiments of the systems and methods of the disclosure, at least one chamber comprises a cell media that maintains the viability of the subject cell from steps (a) through (c) (e.g., from contacting the subject cell and target cell or the stimulatory agent through removal of the antibody panel comprising antibody complexes with one or more of a peptide, polypeptide or protein secreted from the subject cell).
[0035] In certain embodiments of the systems and methods of the disclosure, the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate diminished or decreasing cell function or cell viability.
[0036] In certain embodiments of the systems and methods of the disclosure, the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate(s) augmented or increasing inflammation.
[0037] In certain embodiments of the systems and methods of the disclosure, the secretome comprises one or more distinct peptide(s), polypeptide(s), or protein(s) that indicate(s) increased cell activity or cellular stimulation.
[0038] In certain embodiments of the systems and methods of the disclosure, the methods further comprise determining a Polyfunctional Strength Index (PSI). In certain embodiments, the Polyfunctional Strength Index is the product of a percentage of polyfunctional subject cells within the heterogeneous cell population and an average signal intensity of two or more cytokines. In certain embodiments, the average signal intensity of two or more cytokines is the average signal intensity of two or more distinct cytokines (i.e. AB versus AA). In certain embodiments, the polyfunctional subject cells, at a single cell level, secrete at least two cytokines. In certain embodiments, the polyfunctional subject cells, at a single cell level, secrete at least two distinct cytokines (i.e. AB versus AA). In certain embodiments, the at least two cytokines produced by each of the polyfunctional subject cells and the two or more cytokines of the average signal intensity comprise the same cytokines (e.g. AB and AB). In certain embodiments, the at least two cytokines produced by each of the polyfunctional subject cells and the two or more cytokines of the average signal intensity consist of the same cytokines. In certain embodiments, an increase in the PSI indicates an increase in the potency of the polyfunctional subject cells.
[0039] The disclosure provides a use of a secretome produced by a method of the disclosure for the identification of a T-Lymphocyte expressing a CAR that specifically binds an antigen presented on a target cell, a CAR that specifically binds a stimulatory agent, or a CAR that specifically binds an antigen presented on a target cell and specifically binds a stimulatory agent.
[0040] The disclosure provides a use of a secretome produced by a method of the disclosure for the evaluation of the safety of a cellular therapy, wherein the cellular therapy comprises the subject cell, the cellular therapy is intended to respond to the target cell or the stimulatory agent, and wherein the cellular therapy is considered safe when the secretome lacks one or more peptide(s), polypeptide(s), or protein(s) that stimulate the immune system.
[0041] The disclosure provides a use of a secretome produced by a method of the disclosure for the evaluation of the safety of a cellular therapy, wherein the cellular therapy comprises the subject cell, the cellular therapy is intended to respond to the target cell or the stimulatory agent, and wherein the cellular therapy is considered safe when the secretome lacks one or more peptide(s), polypeptide(s), or protein(s) that indicate decreased cell viability.
[0042] The disclosure provides a use of a secretome produced by a method of the disclosure for the evaluation of the efficacy of a cellular therapy, wherein the cellular therapy comprises the subject cell, the cellular therapy is intended to respond to the target cell or the stimulatory agent, and wherein the cellular therapy is considered safe when the secretome contains one or more peptide(s), polypeptide(s), or protein(s) that indicate a selective response to the target cell.
[0043] The disclosure provides a use of the secretome produced by a method of the disclosure for the evaluation of a cellular therapy, wherein the cellular therapy comprises the subject cell and wherein the subject cell is a chimeric antigen receptor (CAR)-expressing T cell; wherein the cellular therapy is intended to respond to the target cell or the stimulatory agent, wherein the target cell is a cancer cell that expresses an antigen to which the chimeric antigen receptor (CAR) specifically binds, and wherein, upon binding the antigen, the chimeric antigen receptor (CAR) stimulates the T cell; wherein the cellular therapy is considered efficacious when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that stimulate the immune system above a first threshold, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of effector, stimulatory or chemoattractive cytokines; and wherein the cellular therapy is considered safe when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that mediate a deleterious process, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of regulatory and inflammatory cytokines. In certain embodiments of this use, the effector cytokines are selected from the group consisting of Granzyme B, IFN-.gamma., M1P-1.alpha., Performin, TNF-.alpha., and TNF-.beta.. In certain embodiments of this use, the use of claim 59, wherein the stimulatory cytokines are selected from the group consisting of GM-CSF, IL-12, IL-15, IL-2, IL-21, IL-5, IL-7, IL-8 and IL-9. In certain embodiments of this use, the chemoattractive cytokines are selected from the group consisting of CCL-11, IP-10, MIP-1.beta. and RANTES. In certain embodiments of this use, the regulatory cytokines are selected from the group consisting of IL-10, IL-13, IL-22, IL-4, TGF-.beta.1, sCD137 and sCD40L. In certain embodiments of this use, the inflammatory cytokines are selected from the group consisting of IL-17A, IL-17F, IL-1.beta., IL-6, MCP-1 and MCP-4. In certain embodiments of this use, the deleterious process comprises inflammation. In certain embodiments of this use, the deleterious process comprises an autoimmune response. In certain embodiments of this use, the deleterious process comprises a non-selective response to the target cell. In certain embodiments of this use, the subject cell is isolated or derived from an adoptive cell therapy for use in the treatment of a cancer.
[0044] The disclosure provides a use of the secretome produced by a method of the disclosure for the evaluation of a cellular therapy, wherein the cellular therapy comprises the subject cell and wherein the subject cell is a chimeric antigen receptor (CAR)-expressing T cell; wherein the cellular therapy is intended to respond to the target cell or the stimulatory agent, wherein the target cell is an autoimmune cell that expresses an antigen to which the chimeric antigen receptor (CAR) specifically binds, and wherein, upon binding the antigen, the chimeric antigen receptor (CAR) stimulates the T cell; wherein the cellular therapy is considered efficacious when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that stimulate the immune system above a first threshold, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of effector, stimulatory or chemoattractive cytokines; and wherein the cellular therapy is considered safe when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that mediate a deleterious process, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of regulatory and inflammatory cytokines. In certain embodiments of this use, the effector cytokines are selected from the group consisting of Granzyme B, IFN-.gamma., M1P-1.alpha., Performin, TNF-.alpha., and TNF-.beta.. In certain embodiments of this use, the use of claim 59, wherein the stimulatory cytokines are selected from the group consisting of GM-CSF, IL-12, IL-15, IL-2, IL-21, IL-5, IL-7, IL-8 and IL-9. In certain embodiments of this use, the chemoattractive cytokines are selected from the group consisting of CCL-11, IP-10, MIP-1.beta. and RANTES. In certain embodiments of this use, the regulatory cytokines are selected from the group consisting of IL-10, IL-13, IL-22, IL-4, TGF-.beta.1, sCD137 and sCD40L. In certain embodiments of this use, the inflammatory cytokines are selected from the group consisting of IL-17A, IL-17F, IL-1.beta., IL-6, MCP-1 and MCP-4. In certain embodiments of this use, the deleterious process comprises inflammation. In certain embodiments of this use, the deleterious process comprises an autoimmune response. In certain embodiments of this use, the deleterious process comprises a non-selective response to the target cell. In certain embodiments of this use, the autoimmune cell is an immune cell that initiates, effectuates, or enhances an autoimmune response. In certain embodiments of this use, the subject cell is isolated or derived from an adoptive cell therapy for use in the treatment of an autoimmune condition.
[0045] The disclosure provides a use of the secretome produced by a method of the disclosure for the evaluation of a cellular therapy, wherein the cellular therapy comprises the subject cell and wherein the subject cell is a chimeric antigen receptor (CAR)-expressing T cell; wherein the cellular therapy is intended to respond to the target cell or the stimulatory agent, wherein the target cell is an infected cell that expresses an antigen to which the chimeric antigen receptor (CAR) specifically binds, and wherein, upon binding the antigen, the chimeric antigen receptor (CAR) stimulates the T cell; wherein the cellular therapy is considered efficacious when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that stimulate the immune system above a first threshold, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of effector, stimulatory or chemoattractive cytokines; and wherein the cellular therapy is considered safe when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that mediate a deleterious process, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of regulatory and inflammatory cytokines. In certain embodiments of this use, the effector cytokines are selected from the group consisting of Granzyme B, IFN-.gamma., M1P-1.alpha., Performin, TNF-.alpha., and TNF-.beta.. In certain embodiments of this use, the use of claim 59, wherein the stimulatory cytokines are selected from the group consisting of GM-CSF, IL-12, IL-15, IL-2, IL-21, IL-5, IL-7, IL-8 and IL-9. In certain embodiments of this use, the chemoattractive cytokines are selected from the group consisting of CCL-11, IP-10, MIP-1.beta. and RANTES. In certain embodiments of this use, the regulatory cytokines are selected from the group consisting of IL-10, IL-13, IL-22, IL-4, TGF-.beta.1, sCD137 and sCD40L. In certain embodiments of this use, the inflammatory cytokines are selected from the group consisting of IL-17A, IL-17F, IL-1.beta., IL-6, MCP-1 and MCP-4. In certain embodiments of this use, the deleterious process comprises inflammation. In certain embodiments of this use, the deleterious process comprises an autoimmune response. In certain embodiments of this use, the deleterious process comprises a non-selective response to the target cell. In certain embodiments of this use, the autoimmune cell is an immune cell that initiates, effectuates, or enhances an autoimmune response. In certain embodiments of this use, the subject cell is isolated or derived from an adoptive cell therapy for use in the treatment of an infection or an infectious/contagious condition.
[0046] The disclosure provides a use of the secretome produced by a method of the disclosure for the evaluation of a cellular therapy, wherein the cellular therapy comprises the subject cell and wherein the subject cell is a chimeric antigen receptor (CAR)-expressing T cell; wherein the cellular therapy is intended to respond to the target cell or the stimulatory agent, wherein the target cell is a cardiovascular cell that expresses an antigen to which the chimeric antigen receptor (CAR) specifically binds, and wherein, upon binding the antigen, the chimeric antigen receptor (CAR) stimulates the T cell; wherein the cellular therapy is considered efficacious when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that stimulate the immune system above a first threshold, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of effector, stimulatory or chemoattractive cytokines; and wherein the cellular therapy is considered safe when the secretome comprises one or more peptide(s), polypeptide(s), or protein(s) that mediate a deleterious process, wherein the one or more peptide(s), polypeptide(s), or protein(s) comprise one or more cytokines, and wherein the one or more cytokines are selected from the group consisting of regulatory and inflammatory cytokines. In certain embodiments of this use, the effector cytokines are selected from the group consisting of Granzyme B, IFN-.gamma., M1P-1.alpha., Performin, TNF-.alpha., and TNF-.beta.. In certain embodiments of this use, the use of claim 59, wherein the stimulatory cytokines are selected from the group consisting of GM-CSF, IL-12, IL-15, IL-2, IL-21, IL-5, IL-7, IL-8 and IL-9. In certain embodiments of this use, the chemoattractive cytokines are selected from the group consisting of CCL-11, IP-10, MIP-1.beta. and RANTES. In certain embodiments of this use, the regulatory cytokines are selected from the group consisting of IL-10, IL-13, IL-22, IL-4, TGF-.beta.1, sCD137 and sCD40L. In certain embodiments of this use, the inflammatory cytokines are selected from the group consisting of IL-17A, IL-17F, IL-1.beta., IL-6, MCP-1 and MCP-4. In certain embodiments of this use, the deleterious process comprises inflammation. In certain embodiments of this use, the deleterious process comprises an autoimmune response. In certain embodiments of this use, the deleterious process comprises a non-selective response to the target cell. In certain embodiments of this use, the autoimmune cell is an immune cell that initiates, effectuates, or enhances an autoimmune response. In certain embodiments of this use, the cardiovascular cell is a smooth muscle cell, a cardiac muscle cell, an endothelial cell, or any other cell type integrated into a capillary, vein, or an artery. In certain embodiments of this use, the cardiovascular cell is a component of the local or circulating blood, including a blood platelet. In certain embodiments of this use, the platelet is a component of a blood clot or another form of obstruction to healthy blood flow. In certain embodiments of this use, the cardiovascular cell is a damaged cell. In certain embodiments of this use, the cardiovascular cell is an infected cell. In certain embodiments of this use, the subject cell is isolated or derived from an adoptive cell therapy for use in the treatment of a cardiovascular condition or a vascular condition.
[0047] The disclosure provides a method of identifying a subject cell population as efficacious for use in an adoptive cell therapy, comprising: detecting at least one component of a secretome of each subject cell of the subject cell population according to the method of identifying a secretome from a subject cell of the disclosure; identifying a subpopulation of polyfunctional cells of the subject cell population, wherein a polyfunctional cell of subject cells of the subject cell population secrete two or more signaling molecules; calculating a percentage of polyfunctionality of the subject cell population, wherein the percentage of polyfunctionality is the percentage of polyfunctional cells within the subject cell population; measuring a signal intensity of a first signaling molecule of the secretome of each polyfunctional cell of the subject cell population; measuring a signal intensity of a second signaling molecule of the secretome of each polyfunctional cell of the subject cell population; calculating a Polyfunctional Strength Index (PSI) for each polyfunctional cell of the subject cell population, wherein the PSI comprises (a) the product of the percentage of polyfunctionality of the subject cell population and the signal intensity of the first signaling molecule and (b) the product of the percentage of polyfunctionality of the subject cell population and the signal intensity of the second signaling molecule; identifying the subject cell population as efficacious for use in an adoptive cell therapy when the PSI indicates that at least 50% of the subject cells in the subject cell population are polyfunctional, the signal intensity of the first signaling molecule indicates that the concentration of the first signaling molecule within the chamber is between 2 pg/ml and 10,000 pg/ml, inclusive of the endpoints, and the signal intensity of the second signaling molecule indicates that the concentration of the second signaling molecule within the chamber is between 2 pg/ml and 10,000 pg/ml, inclusive of the endpoints; and identifying the subject cell population as not efficacious for use in an adoptive cell therapy when the PSI indicates that less than 50% of the subject cells in the subject cell population are polyfunctional, the signal intensity of the first signaling molecule indicates that the concentration of the first signaling molecule within the chamber is less than 2 pg/ml, and the signal intensity of the second signaling molecule indicates that the concentration of the second signaling molecule within the chamber is less than 2 pg/ml.
[0048] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises a plurality of immune cells. In certain embodiments, the plurality of immune cells comprises a T-lymphocyte, a B-lymphocyte, a natural killer (NK) cell, a macrophage, a neutrophil, a mast cell, an eosinophil, a basophil or a combination thereof. In certain embodiments, the T-lymphocyte is a naive T-lymphocyte, an activated T-lymphocyte, an effector T-lymphocyte, a helper T-lymphocyte, a cytotoxic T-lymphocyte, a gamma-delta T-lymphocyte, a regulatory T-lymphocyte, a memory T-lymphocyte, or a memory stem T-lymphocyte. In certain embodiments, the T-lymphocyte expresses a non-naturally occurring antigen receptor. In certain embodiments, the T-lymphocyte expresses a Chimeric Antigen Receptor (CAR).
[0049] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises a plurality of neuronal cells. In certain embodiments, the plurality of neuronal cells comprises a neuron, a glial cell, an astrocyte, a satellite cell, an enteric glial cell or a combination thereof.
[0050] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises a plurality of endocrine cells. In certain embodiments, the plurality of endocrine cells comprises one or more cells isolated or derived from a pineal gland, a pituitary gland, a pancreas, an ovary, a testicle, a thyroid gland, a parathyroid gland, a hypothalamus, or an adrenal gland.
[0051] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises a plurality of exocrine cells. In certain embodiments, the plurality of exocrine cells comprises one or more cells isolated or derived from a salivary gland, a sweat gland or a component of the gastrointestinal tract. In certain embodiments, the component of the gastrointestinal tract comprises a mouth, a stomach, a small intestine, and a large intestine.
[0052] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a cancer cell. In certain embodiments, the cancer cell is a primary cancer cell or a cultured cancer cell. In certain embodiments, the primary cancer cell is metastatic.
[0053] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a B-lymphocyte.
[0054] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a bacteria, yeast, or microbe.
[0055] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is an infected cell. Exemplary infected cells may have contacted or may have been exposed to a virus, bacteria, yeast, or microbe.
[0056] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell perpetuates an autoimmune response.
[0057] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell is a target of an autoimmune response.
[0058] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the target cell is a host cell. In certain embodiments, the host cell comprises any cell isolated or derived from the same individual as the subject cell. In certain embodiments, the host cell is a functional cell and wherein the host cell does not stimulate an autoimmune response. The term "functional" cell is meant to describe a viable cell that does not contribute to a disease or disorder in the host. Alternatively, or in addition, the term "functional" cell may describe a cell without any known mutations that cause a disease or disorder in the host. For example, a functional cell may be non-cancerous and/or non-autoimmune.
[0059] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the first signaling molecule is a peptide, a polypeptide, or a protein. In certain embodiments, the first signaling molecule is a cytokine.
[0060] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the second signaling molecule is a peptide, a polypeptide, or a protein. In certain embodiments, the second signaling molecule is a cytokine.
[0061] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises a plurality of T-lymphocytes; wherein the target cell is a cancer cell, an infected cell or a host cell that perpetuates an autoimmune response; the first signaling molecule comprises an effector cytokine, a stimulatory cytokine, or a chemoattractive cytokine and the second signaling molecule comprises an effector cytokine, a stimulatory cytokine, or a chemoattractive cytokine. In certain embodiments, at least one T-lymphocyte of the plurality of T-lymphocytes expresses a chimeric antigen receptor (CAR). In certain embodiments, each T-lymphocyte of the plurality of T-lymphocytes expresses a chimeric antigen receptor (CAR). In certain embodiments, the effector cytokine is Granzyme B, IFN-.gamma., M1P-1.alpha., Performin, TNF-.alpha. or TNF-.beta.. In certain embodiments, the stimulatory cytokine is GM-CSF, IL-12, IL-15, IL-2, IL-21, IL-5, IL-7, IL-8 and IL-9. In certain embodiments, the chemoattractive cytokine is CCL-11, IP-10, MIP-1.beta. or RANTES.
[0062] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the method further comprises identifying the subject cell population as safe for use in an adoptive cell therapy, when the PSI indicates that at least 50% of the subject cells in the subject cell population are polyfunctional, the signal intensity of the first signaling molecule indicates that the concentration of the first signaling molecule within the chamber is less than 2 pg/ml, the signal intensity of the second signaling molecule indicates that the concentration of the second signaling molecule within the chamber is less than 2 pg/ml, the subject cell population comprises a plurality of T-lymphocytes, the first signaling molecule comprises a regulatory cytokine or an inflammatory cytokine and the second signaling molecule comprises a regulatory cytokine or an inflammatory cytokine; and identifying the subject cell population as not safe for use in an adoptive cell therapy when the PSI indicates that at least 50% of the subject cells in the subject cell population are polyfunctional, the signal intensity of the first signaling molecule indicates that the concentration of the first signaling molecule within the chamber is greater than 2 pg/ml, the signal intensity of the second signaling molecule indicates that the concentration of the second signaling molecule within the chamber is greater than 2 pg/ml, the subject cell population comprises a plurality of T-lymphocytes, the first signaling molecule comprises a regulatory cytokine or an inflammatory cytokine and the second signaling molecule comprises a regulatory cytokine or an inflammatory cytokine. In certain embodiments, the regulatory cytokine is IL-10, IL-13, IL-22, IL-4, TGF-.beta.1, sCD137 and sCD40L. In certain embodiments, the inflammatory cytokine is IL-17A, IL-17F, IL-1.beta., IL-6, MCP-1 and MCP-4. In certain embodiments, at least one T-lymphocyte of the plurality of T-lymphocytes expresses a chimeric antigen receptor (CAR). In certain embodiments, each T-lymphocyte of the plurality of T-lymphocytes expresses a chimeric antigen receptor (CAR).
[0063] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the subject cell population comprises at least 100 cells. In certain embodiments, the subject cell population comprises at least 500 cells. In certain embodiments, the subject cell population comprises at least 1000 cells. In certain embodiments, the subject cell population comprises at least 5000 cells.
[0064] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the detecting step comprises detecting at least 2 components of a secretome from each subject cell of the subject cell population. In certain embodiments, the detecting step comprises detecting at least 10 components of a secretome from each subject cell of the subject cell population. In certain embodiments, the detecting step comprises detecting at least 20 components of a secretome from each subject cell of the subject cell population. In certain embodiments, the detecting step comprises detecting at least 30 components of a secretome from each subject cell of the subject cell population. In certain embodiments, the detecting step comprises detecting at least 50 components of a secretome from each subject cell of the subject cell population. In certain embodiments, the detecting step comprises detecting at least 100 components of a secretome from each subject cell of the subject cell population.
[0065] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the percentage of polyfunctional cells comprises a first percentage of polyfunctional cells that secrete two or more signaling molecules, a second percentage of polyfunctional cells that secrete three or more signaling molecules, a third percentage of polyfunctional cells that secrete four or more signaling molecules, a fourth percentage of polyfunctional cells that secrete five or more signaling molecules, and a subsequent percentage of polyfunctional cells that secrete increasing numbers of signaling molecules.
[0066] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the measuring of the signal intensity comprises detecting a fluorescent signal from a complex of an antibody specific for the first or second signaling molecule and the first or second signaling molecule, respectively, and normalizing each fluorescent signal against a reference signal to determine a relative fluorescent unit (RFU) value. In certain embodiments, the reference signal is a maximal signal, a minimal signal, or a signal from a component of the secretome with a constant or known concentration. In certain embodiments, the reference signal is a component of the secretome of a subject cell that is most abundant.
[0067] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the method further comprises measuring a third or subsequent signaling molecule of the secretome of each polyfunctional cell.
[0068] In certain embodiments of the methods of identifying a subject cell population as efficacious for use in an adoptive cell therapy, the method further comprises determining a relative contribution of the first signaling molecule, the second signaling molecule or the subsequent signaling molecule to a response of the subpopulation of polyfunctional cells to a target cell or to a stimulatory agent, wherein the relative contribution is the product of an average of a percentage of the PSI of each polyfunctional cell from the first signaling molecule, the second signaling molecule or the subsequent signaling molecule from each polyfunctional cell and a total PSI for the subpopulation of polyfunctional cells.
BRIEF DESCRIPTION OF THE DRAWINGS
[0069] FIG. 1 is a photograph showing a system of the disclosure for high throughput analysis of Two-Cell Interactions. Natural Killer (NK) cell and leukemia cell (K562) populations loaded into a plurality of chambers (e.g. in this example, taking the form of micro-troughs) are mixed allowing for a high throughput analysis of individual two-cell interactions within one chamber (e.g. in this example, a micro-trough).
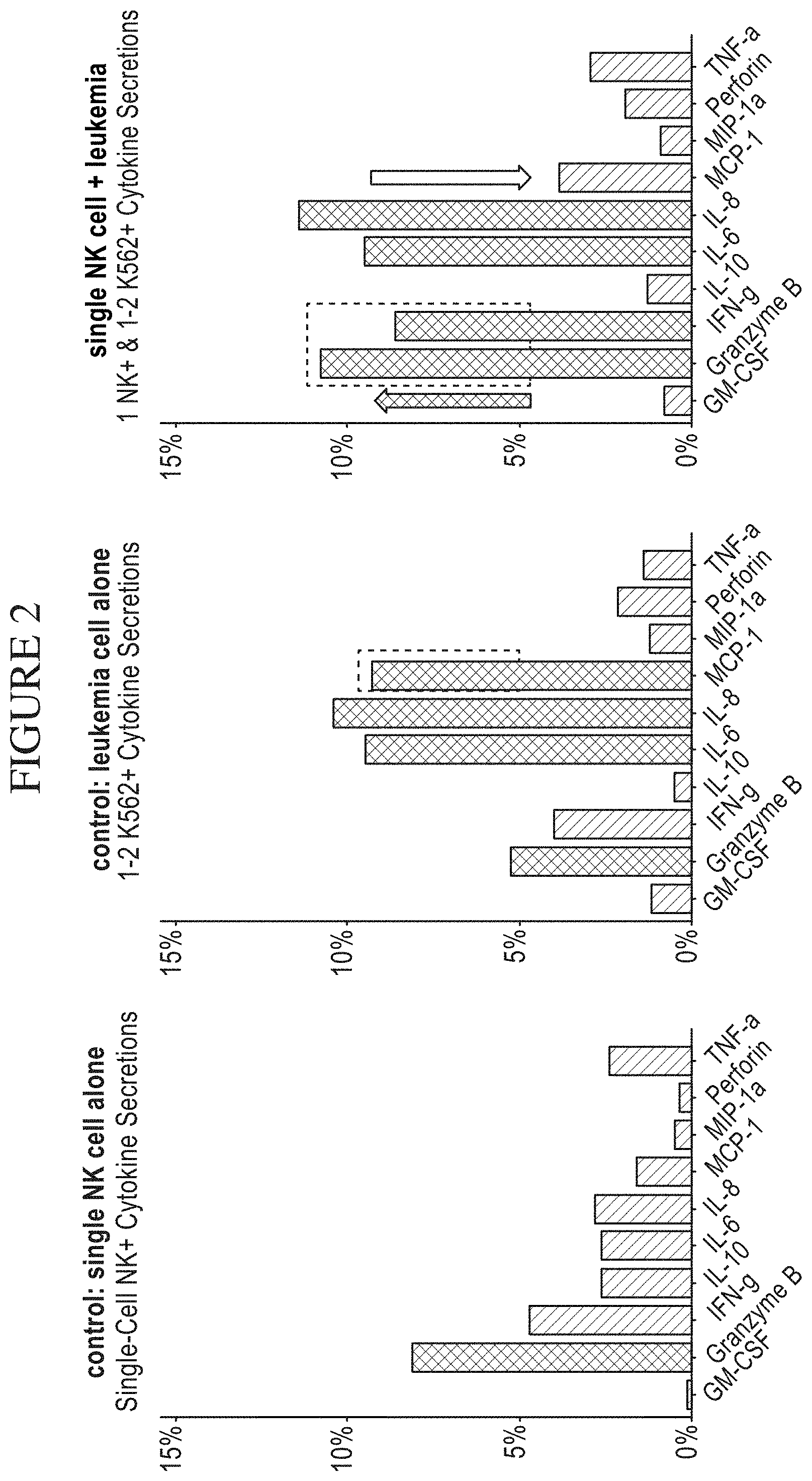
[0070] FIG. 2 is a series of graphs showing single cell secretion levels following two cell interactions with a Natural Killer (NK) cell as the subject cell and a leukemia cell (K562) as the target cell in a system of the disclosure. The percent (%) of cell subpopulation secreting is provided as a function of the type of cytokine secreted by the NK cell in each pairing. The secretomes of each NK cell were identified according to a method of the disclosure. The arrows indicate statistically significant differences between the levels of cytokine(s) identified in the secretome of single NK cells in each condition of the two-cell interaction method.
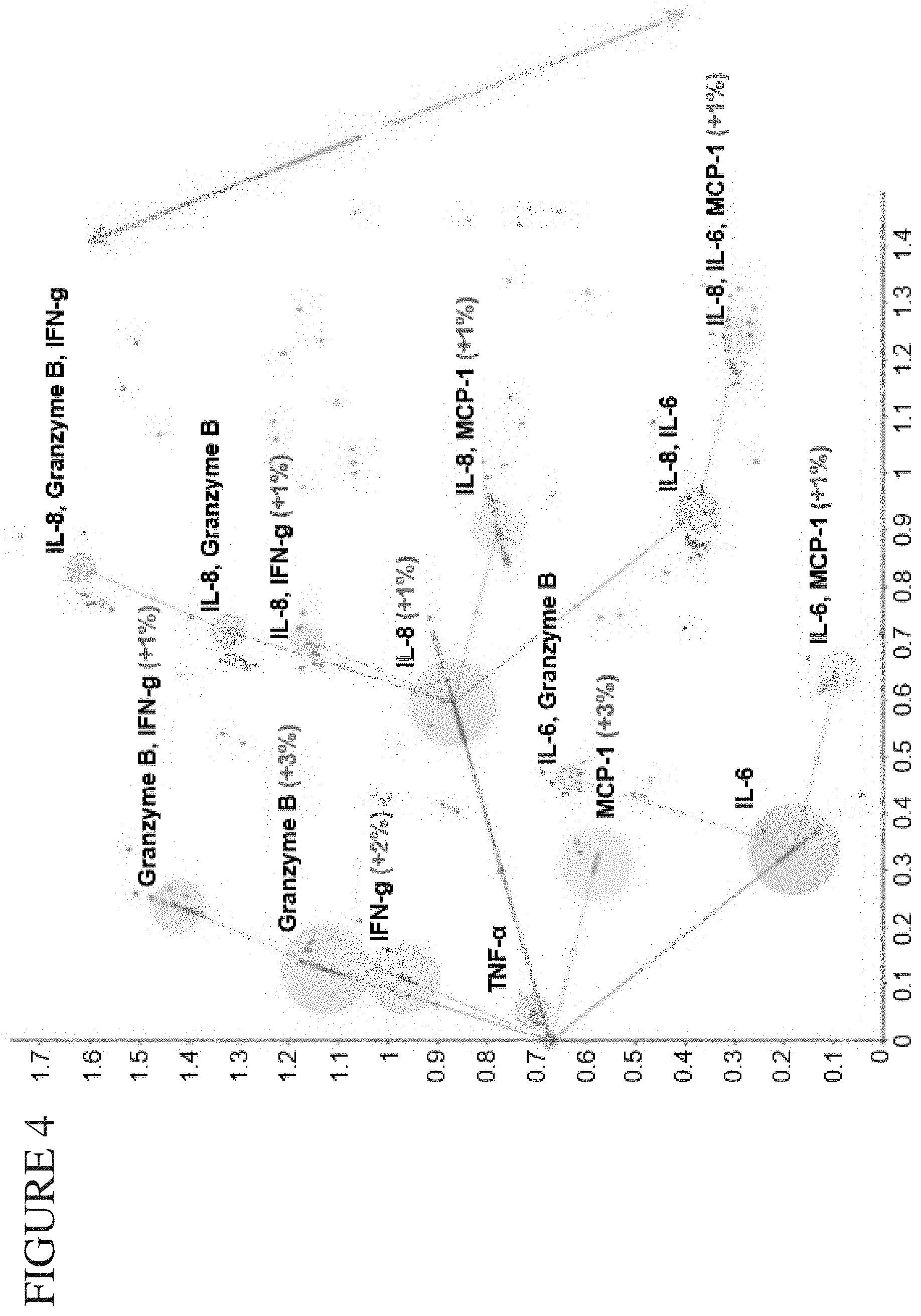
[0071] FIG. 3 is a series of graphs comparing cellular polyfunctionality following two cell interactions with a Natural Killer (NK) cell as the subject cell and a leukemia cell (K562) as the target cell in a system of the disclosure. The data demonstrate an increased percentage of poly-functional NK cells (secreting 2+cytokines) following an interaction with the leukemia cell (right-hand graph).
[0072] FIG. 4 is a Process Analytical Tools (PAT)-Principal Component Analysis (PCA) plot showing cellular polyfunctionality following two cell interactions with a Natural Killer (NK) cell as the subject cell and a leukemia cell (K562) as the target cell in a system of the disclosure. Functional groups are color-coded by population in which there is a dominant function. Leukemia-activated NK cells lead to increased effector functional cell subpopulations and reduced inflammatory cell subpopulations.
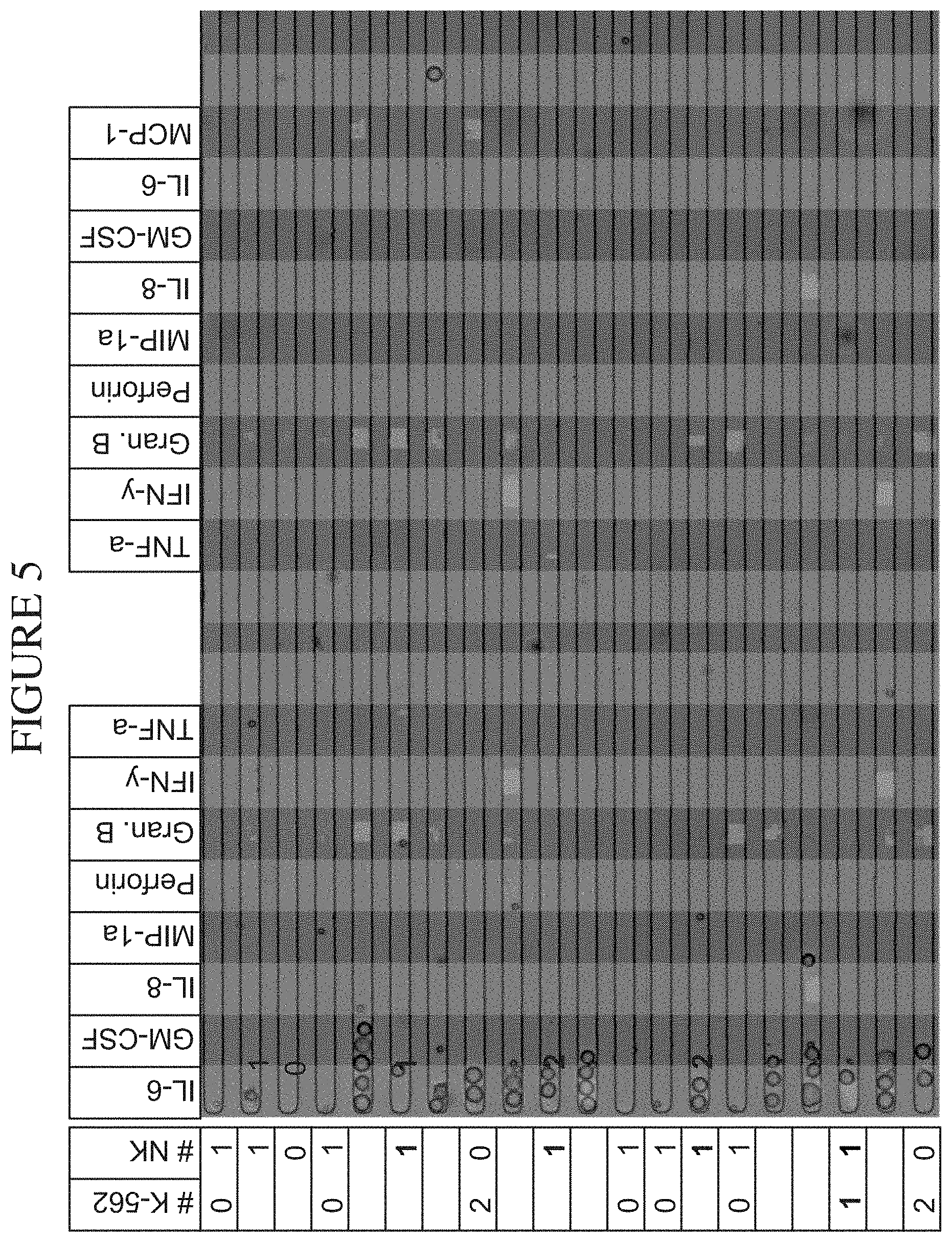
[0073] FIG. 5 is a photograph depicting the isolation and manipulation of human immune cell subsets in a system of the disclosure. NK/K562 interactions isolated at the single cell level are combined with their corresponding secretome (also referred to as a secretion profile), enabling a comprehensive analysis of a Target/ Effector relationship.
[0074] FIG. 6 is a pair of graphs depicting a cytokine and cytotoxicity profile of CD8-positive T cells. The addition of the bispecific antibody substantially increases both cytokine effector function and cytotoxicity to the target Raji cells (right-hand graph). The table above the graphs summarizes the data presented in the graphs below.
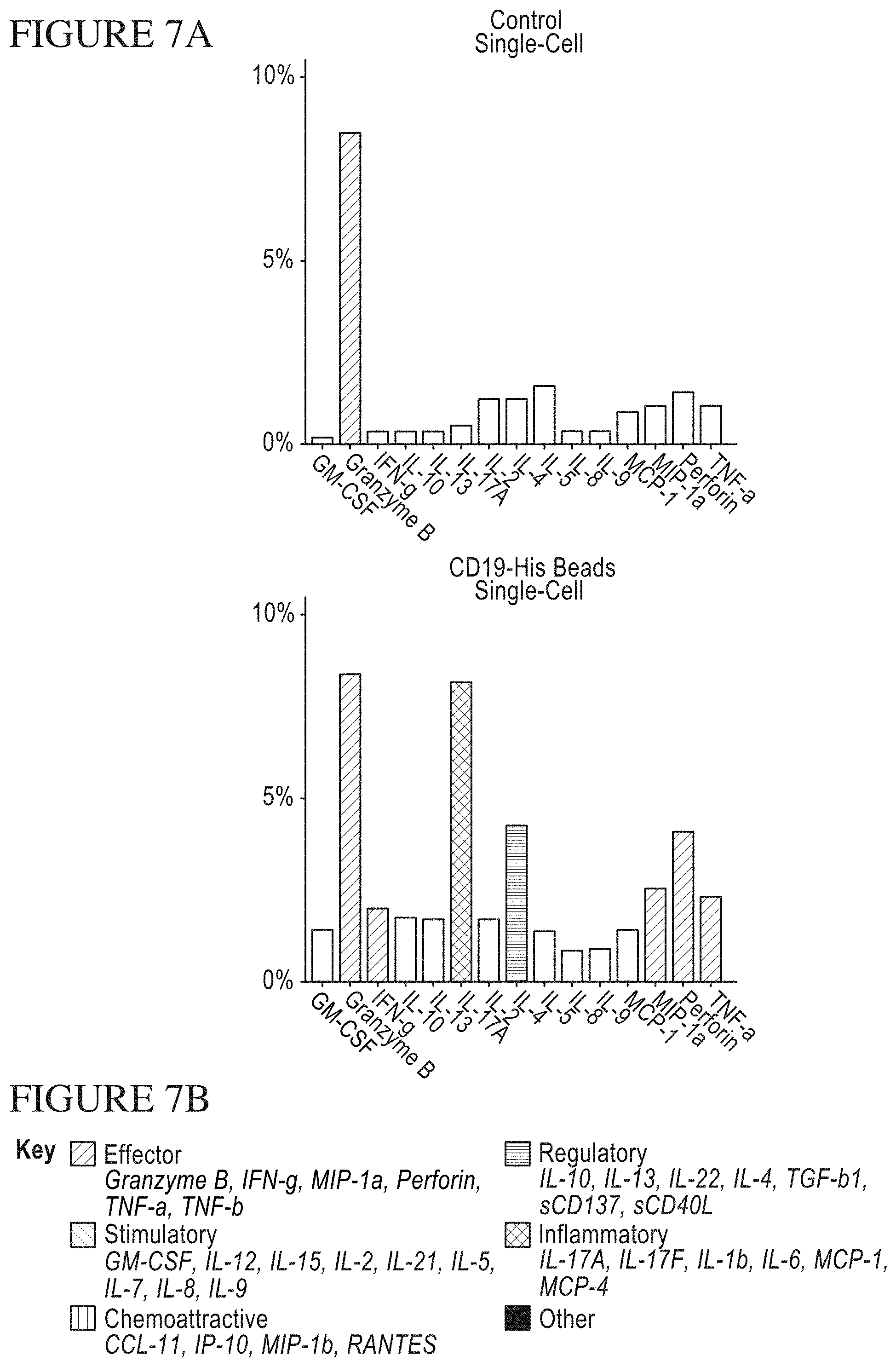
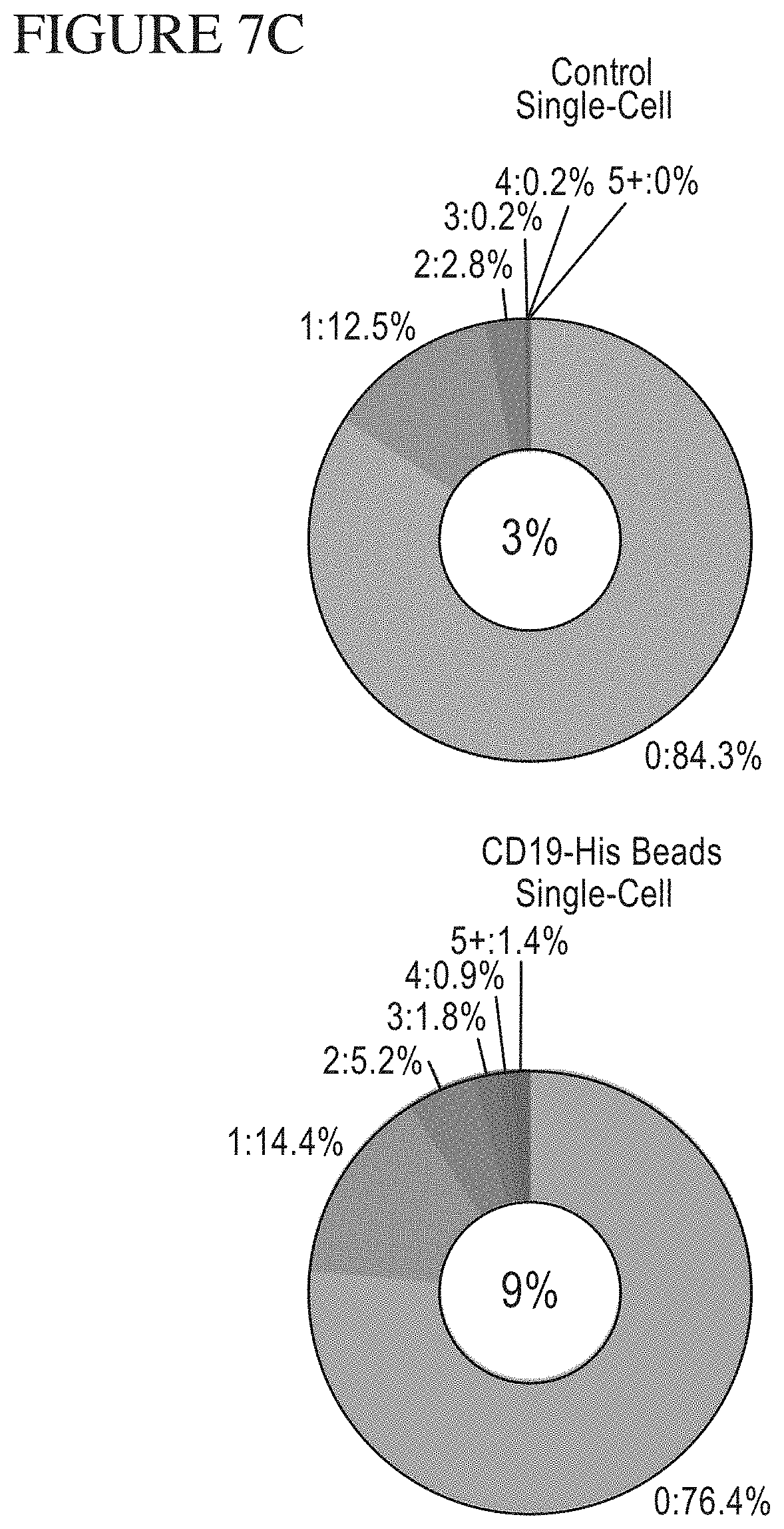
[0075] FIG. 7A-C is a series of graphs and a key identifying the components of the secretomes of CAR-expressing T-Cells following stimulation by CD19 Beads. A. Shows the upregulation in cytokine secretion by CAR T-cells stimulated with CD19 versus control beads. In the up-regulation case, certain cytokines associated with potency (TNF.alpha., Granzyme, etc.) as well as certain cytokines associated with toxicity (IL-17A) are detailed. B. Key for the bar graphs in A. Each bar color represents the inclusion of a cytokine in a specific functional group. Effector cytokines include, but are not limited to, Granzyme B, Interferon-gamma (IFN-.gamma.), MIP-1.alpha., Perforin, TNF-.alpha., and TNF-.beta.. C. Stimulatory cytokines include, but are not limited to, GM-CSF, Interleukin-12 (IL-12), Interleukin-15 (IL-15), Interleukin-2 (IL-2), Interleukin-21 (IL-21), Interleukin-5 (IL-5), Interleukin-7 (IL-7), Interleukin-8 (IL-8), and Interleukin-9 (IL-9). Chemoattractive cytokines include, but are not limited to, CCL-11, IP-10, MIP-1.beta., and RANTES. Regulatory cytokines include, but are not limited to, Interleukin-10 (IL-10), Interleukin-13 (IL-13), Interleukin-22 (IL-22), Interleukin-4 (IL-4), TGF-.beta.1, sCD137 and sCD40L. Inflammatory cytokines include, but are not limited to, Interleukin-17A (IL-17A), Interleukin-17F (IL-17F), Interleukin-1b (IL-1b), Interleukin-6 (IL-6), MCP-1, and MCP-4. Polyfunctionality of single CAR T-cells after stimulation by control or CD19 beads. Polyfunctionality is correlated with potency or success of immune mediated therapies.
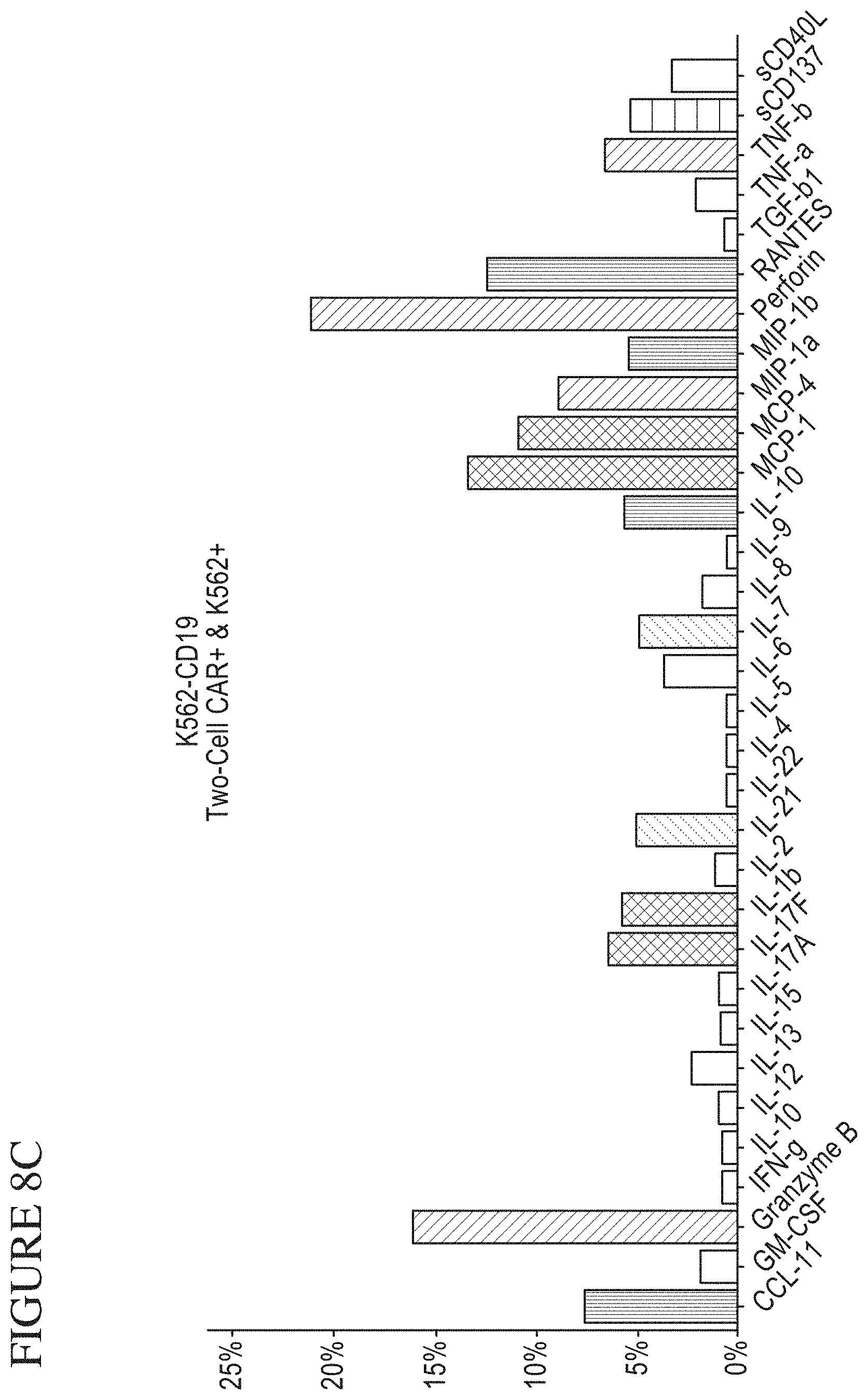
[0076] FIG. 8A-G is a series of graphs and a key identifying the components of the secretomes of CAR-expressing T-Cells following stimulation by target cells. A. Cytokine secretion levels by CAR-expressing T-cells after stimulation with K562-CD19 target cells, where CD19 is a proxy for the leukemia cells or B-cell malignancies that are the cell therapy target. B. Dampened cytokine secretion by CAR-expressing T-cells after stimulation with K562-NGFR control cells (negative control cells because they do not include the CD19 target). C. Cytokine secretion levels in chambers (taking the form of micro-troughs for this example) containing a single CAR-expressing T-cell and a single K562-CD19 target cell. D. Dampened cytokine secretion in chambers containing a single CAR-expressing T-cell and a single K562-NGFR control cell. E. Key for the bar graphs in A-D. Each bar color represents the inclusion of a cytokine in a specific functional group. Effector cytokines include, but are not limited to, Granzyme B, Interferon-gamma (IFN-.gamma.), MIP-1.alpha., Perforin, TNF-.alpha. and TNF-.beta.. C. Stimulatory cytokines include, but are not limited to, GM-CSF, Interleukin-12 (IL-12), Interleukin-15 (IL-15), Interleukin-2 (IL-2), Interleukin-21 (IL-21), Interleukin-5 (IL-5), Interleukin-7 (IL-7), Interleukin-8 (IL-8), and Interleukin-9 (IL-9). Chemoattractive cytokines include, but are not limited to, CCL-11, IP-10, MIP-1.beta., and RANTES. Regulatory cytokines include, but are not limited to, Interleukin-10 (IL-10), Interleukin-13 (IL-13), Interleukin-22 (IL-22), Interleukin-4 (IL-4), TGF-.beta.1, sCD137 and sCD40L. Inflammatory cytokines include, but are not limited to, Interleukin-17A (IL-17A), Interleukin-17F (IL-17F), Interleukin-1b (IL-1b), Interleukin-6 (IL-6), MCP-1, and MCP-4. F. Polyfunctionality of single CAR-expressing T cells in response to K562-CD19 or control stimulation. G. An increase in polyfunctionality when the K562-CD19 target cell is present in the chamber (taking the form of a micro-trough for this example) with the CAR-expressing T-cell.
[0077] FIG. 9 is a series of photographs depicting systems of the disclosure and the use of fluorescent tracking of cytotoxicity. K562 cells, labeled with the membrane dye CFSE, are mixed with non-labeled NK cells and loaded into chambers (taking the form of micro-troughs for this example) with an excess of SYTOX Orange. The chambers (taking the form of micro-troughs for this example) are imaged immediately after loading (TO) and then again after a 16 hour incubation (T1) and assessed for cell death via fluorescence.
[0078] FIG. 10A-C is a series of signal plots of control and stimulated CAR (chimeric antigen receptor expressing) cells. Panel A. PSI (poly-functional strength intensity) Overview and Individual Cytokine contributions from control CAR cells. Panel B. PSI Overview and Individual Cytokine contributions from stimulated CAR cells (stimulated cells were contacted to CD19-conjugated beads). Panel C. Individual signal plots of both control and stimulated CAR cells.
[0079] FIG. 11A-F is a series of graphs depicting signals detected from CAR cells stimulated by target cells. PSI Overview from CAR cells stimulated with K562-NFGR control cells (A) or K562-CD19 (B) target cells. Individual Cytokine contributions from CAR cells stimulated with K562-NGFR (C) and K562-CD19 (D). The final plots show signal cell secretion points from CAR cells stimulated with K562-NGFR (E) or K562-CD19 (F).
[0080] FIG. 12A-M is a series of graphs comparing results of Memorial Sloan-Kettering Cancer Center (MSKCC) assays of cytokine secretion to exemplary detection sensitivities of the compositions, systems and methods of the disclosure (referred to as "IsoPlexis" in the figure). In the study by MSKCC (Brenthens et al. Blood. 2011 Nov. 3; 118(18): 4817-4828), safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias, MSKCC stimulated EOP 19-28z transduced T cells with 3T3 fibroblasts expressing CD19 for 48 hours. MSKCC then analyzed the cytokine secretion levels from the transduced cells on a Luminex IS100 to determine pg/mL amounts of analyte. The compositions, systems and methods of the disclosure can gather the same type of data as the MSKCC assay with equal or greater sensitivity as the MSKCC assay. The above side-by-side graphs show the sensitivity, per analyte, on the compositions, systems and methods of the disclosure versus the amounts measured from the MSKCC Luminex assay. The measurements from the MSKCC assay clearly fall within the measurable range of the compositions, systems and methods of the disclosure (a concentration of cytokines between about 2 pg/ml and about 10,000 pg/ml, inclusive of the endpoints, per cytokine in a highly multiplexed reaction). Therefore the same data could have been gathered using the compositions, systems and methods of the disclosure with the additional benefit of being able to transform this data to represent the Polyfunctional Strength Index (PSI) of individual cell populations. PSI measurements for each cytokine of a plurality of cytokines for each cell in a large-scale experiment (simultaneously measuring thousands of individual cells within a population), enables a prediction of statistically powerful biomarker and cell subsets that drive patient responses.
[0081] FIG. 13A-B is a pair of tables comparing results of National Cancer Institute cytokine assays of secretions from CARs to exemplary detection sensitivities of the compositions, systems and methods of the disclosure (referred to as "IsoPlexis" in the figure). In the study by NCI (Kochenderfer et al. Blood, 22 Mar. 2012, 119(12): 2709-2720), B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells, the NCI cultured CD19 directed CAR cells with either CD19-positive K562 cells or the control NGFR-positive (CD19-negative) overnight and ran the cytokine secretions next day on a standard ELISA assay. Cytokine secretion levels for IFN-gamma, TNF, and IL-2 are listed in the top table. In the bottom table, are listed the limits of detection on the compositions, systems, and methods of the disclosure, with either a signal-to-noise ratio (SNR) of greater than 2 or greater than 5. The compositions, systems, and methods of the disclosure are able to measure cytokine secretions, with great confidence, in the same range as those measured by standard ELISA in this NCI study with the additional benefit of being able to transform this data to represent the Polyfunctional Strength Index (PSI) of individual cell populations. PSI measurements for each cytokine of a plurality of cytokines for each cell in a large-scale experiment (simultaneously measuring thousands of individual cells within a population), enables a prediction of statistically powerful biomarker and cell subsets that drive patient responses.
[0082] FIG. 14A-C is a series of graphs comparing results of National Cancer Institute cytokine assays of secretions from CARs to exemplary detection sensitivities of the compositions, systems and methods of the disclosure (referred to as "IsoPlexis" in the figure). In the study by NCI (Zhao et al. The Journal of Immunology, 2007, 179: 5845-5854), high-affinity TCRs generated by phage display provide CD4+ cells with the ability to recognize and kill tumor cell lines, the NCI measured the cytokine secretions from TCR-transfected T cells. CD8+ and CD4+ T cells were purified and transfected with the TCRs. The cells were then pulsed with NY-ESO-1, MART-1 or gp100 peptides, and the resulting secretions of IL-2 and IFN-gamma were measured using commercially available ELISA kits at a range of between 200 pg/mL and 30,000 pg/mL, inclusive of the endpoints. The compositions, systems and methods of the disclosure are able to measure cytokine secretions, with great confidence, in the same range as those measured by standard ELISA in this NCI study with the additional benefit of being able to transform this data to represent the Polyfunctional Strength Index (PSI) of individual cell populations. PSI measurements for each cytokine of a plurality of cytokines for each cell in a large-scale experiment (simultaneously measuring thousands of individual cells within a population), enables a prediction of statistically powerful biomarker and cell subsets that drive patient responses.
[0083] FIG. 15 is a series of graphs depicting a method of measuring single-cell Polyfunctional Strength Index (PSI). This metric quantifies the overall activity of a sample. The PSI is equal to the percentage of polyfunctional cells (secreting two or more cytokines) in a sample multiplied by the average signal intensity of the secreted cytokines. In order to determine which cytokines are driving the polyfunctional response, each individual cytokine's contribution to PSI can also be calculated. This is equivalent to the fraction of the total PSI coming from a specific cytokine, and is found by calculating the percentage of each cell's signal corresponding to that cytokine, averaging it across all cells, and multiplying this percentage by the total PSI.
DETAILED DESCRIPTION
[0084] The disclosure provides systems and methods capable of measuring key effector proteins at the single cell level. For example, in certain embodiments, the systems and methods of the disclosure can simultaneously measure 42 key effector proteins at the single cell level. The systems and methods of the disclosure may be used, for example, directly in pipeline drug development and CAR-T assessment by large-scale developers of cell-based therapeutics. The multiplexed parameters measured by the systems and methods of the disclosure cover, for example, the complete range of relevant immune effector functions including stimulatory, proinflammatory, regulatory (negative), chemoattractive, pro-growth and cytolytic (effective).
[0085] The systems and methods of the disclosure require smaller amounts of cell input (approximately 1000 cells) compared to the existing single-cell instruments (e.g., flow cytometer) for the analysis of low quantity patient samples, which minimally 100,000 cells per sample for analysis (and typically require millions of cells per sample).
[0086] The systems and method of the disclosure provide an analytical approach capable of evaluating single cell secretion profiles in a highly multiplex manner. In certain embodiments, this analysis involves crosstalk of a single immune cell to a diseased target cell (i.e. direct or indirect contact of a subject cell and a target cell) while avoiding or minimizing paracrine effects from the total population (because each pair of subject cell and target cell may be isolated into a chamber separated from the remaining plurality of chambers). This approach enables the study and detection of rare subject to target cell interactions that may be otherwise masked by other cells within the population or sample.
[0087] The systems and methods of the disclosure are exemplified in a nonlimiting manner through demonstrating the ability of a system of the disclosure to evaluate individual cell-to-cell interactions for the Target/Effector relationship of immune cells, including CAR cells, with various target cells or stimulatory agents according to a method of the disclosure. Highly multiplexed paired immune and cancer cell assays provide mechanisms for assessing a polyfunctional cytokine profile at the single cell level, in response to a cancer cell and its specific antigens. Such assays may further be used to evaluate the immune response quality in relation to the magnitude of an immune cell response to a cancer cell. Even more, such assays may be used to correlate the immune response quality and/or the magnitude of an immune cell response to a cancer cell with the ability of the immune cell to kill the cancer cell. Accordingly, the systems and method of the disclosure provide novel tools for addressing the critically relevant needs of identifying and evaluating cellular therapies for safety and efficacy in the field of cancer immunology, among many other fields.
[0088] The systems and methods of the disclosure provide the ability to analyze cells in a highly multiplexed fashion, down to the single-cell level and are capable of determining the polyfunctional response to a specific diseased cell. While an exemplary method includes monitoring killing of a target tumor cell, by an immune cell, within a chamber, for purposes of correlating with a highly multiplexed immune response, the systems and methods of the disclosure include many other applications.
[0089] As shown in the Figures of the disclosure, and in particular, FIGS. 7, 8, 10, and 11, polyfunctionality is a measure of efficacy and potency of cells intended for cellular therapy. Of particular value is the polyfunctional strength index (PSI). The polyfunctional strength index (PSI) is a metric that factors in the polyfunctionality of cells in a sample, and the signal intensity of the cytokines secreted by each cell. It is found by multiplying the percentage of polyfunctional cells of a sample (single cells secreting two or more cytokines), by the average signal intensity of these cytokines. This PSI is shown on the left of FIGS. 10A and 10B, as well as FIGS. 11A and 11B. For example, FIG. 10B demonstrates that the polyfunctional strength is roughly 2x higher in the CD19 stimulated sample, relative to the control PSI shown in FIG. 10A. FIG. 11B demonstrates that the stimulated cells have a polyfunctional strength roughly 5.times. higher than the control cells. For more detail regarding PSI, see Example 7.
Methods of Analyzing Immune Cells
[0090] Secreted proteins and in particular cytokines, are key mediators of intercellular communication within the immune system. Homeostatic immune response requires tightly regulated cytokine synthesis and secretion. Many analytical technologies have been developed to analyze protein secretion during the immune response, but the methods used are generally restricted to measuring the average secretions for an entire cellular population. Such analyses, while helpful in understanding disease pathogenesis and the immune process, are insufficient to characterize cytokine activity for individual subsets of cells within a population. Recent investigations using single-cell analysis have shown that immune cells display highly heterogeneous cytokine profile even in cells with similar phenotypes further demonstrating a significant limitation of focusing only on cellular response at the bulk population level. These heterogeneous subsets of cells within the population may dictate a complex signaling interplay between cells that represent important checks and balances for disease immunotherapy evaluation. This is particularly notable when a cellular population's response can be determined by the cell-cell interactions in a rare subset of cells. As result, understanding these interactions is crucial to developing more effective therapeutic treatments in the future.
[0091] Challenges to defining consistent and high functional quality "drug" in cell-based cancer immunotherapy: Despite the demonstrated benefit of emerging CAR-T cancer immunotherapeutics, two major concerns remain: one, manufacturing the cell therapies consistently, and two, managing the immuno-toxicity, such as cytokine release syndrome, that could be potentially life threatening. In cell-based therapies such as Chimeric Antigen Receptor T cell (CAR-T) therapy, in which the living cells are the "drug", cellular manufacturing is still relatively new, and each patient batch generated may differ substantially even if a standardized operation procedure (SOP) exists to ensure consistency in manufacturing. Giving clinicians and biotech companies an effective cellular function monitoring tool could change clinical paradigms by allowing them to remove or modify the inconsistent or unsafe cell therapies prior to injection, significantly reducing risk to the patient, and improving odds of therapeutic success.
[0092] Defining the "quality" of a cytokine mediated anti-tumor T cell: To evaluate engineered T cells for an immunotherapy or to evaluate endogenous T cells reactivated to battle cancer or infection, a T cell's functional status is largely determined by a spectrum of secreted effector function proteins (e.g., cytokines). In a protective immune response, the `quality` of an immune cell correlates to the extent of polyfunctionality (the ability of a T cell to co-secrete multiple effector proteins).While these anti-tumor cytolytic, chemoattractive cytokines produce an effective response, these poly-functional cell subsets must not also secrete immuno-toxic inflammatory or regulatory cytokines (up to 15) prevalent in NK or CAR-T cells. To detect consistent performance of this "quality" effective and safe response of CAR single-cell subsets, a need exists for a new technology to conduct highly multiplexed measurement of immune effector proteins in single T cells. And while single-cell flow cytometry technologies fix cells to detect cytokines, a more predictive platform would detect true secretions.
[0093] Current methods of cell function evaluation: Single parameter ELISpot assay remains the state-of-the-art for T cell activation assay, but does not measure polyfunctionality. Averaging bead based multiplexing platform (Luminex based) measure many cytokines, yet not at the required single-cell level. Fluorescence-based multicolor flow cytometry is a powerful single cell analysis tool and has been used to detect cytokines via retaining and staining proteins within the cytoplasm by blocking vesicle transport. The number of proteins that can be simultaneously measured is limited by fluorescence spectral overlap with ICS (intracellular cytokine staining). Time-of-flight mass spectrometer-coupled flow cytometry (CyTOF) has been used recently by CAR-T companies, though not as regularly in trials. Similar to fluorescence flow cytometry, it does not measure true secretion and so far the number of cytokines co-measured in single cells by CyTOF is 11 due in part to the high background of ICS. Other single cell technologies being developed in research laboratories (e.g., microengraving) provides advantages in sensitivity and assay speed but still limited in the multiplexing capability (typically <5).
[0094] Risks involved in adoptive cell therapies and other immune mediated therapies: Cytokine release syndrome (CRS) is a non-antigen specific, life-threatening toxicity that results from the over-activation of the immune system due to immune therapies, such as CAR T-cell therapy. Although CAR T-cells are potent on-target killers, they activate the immune system far above naturally occurring levels and due to the nature of their design, have a large degree of "on target, off tumor" toxicity. Based on the level of mortalities in recent clinical trials, it has become apparent that the cytokine profiles of individual CAR-T cells must be known before introduction to the patient. The systems and methods of the disclosure determine an abundance of up to 42 cytokines, per single cell, falling into the following groups: effector, stimulatory, inflammatory, and regulatory. This information allows the user to identify any potentially toxic subsets of cells (pro-inflammatory or regulatory) within a population that would have been missed by conventional means, providing a safer and more effective means of monitoring immunotherapies prior to patient introduction.
Systems and Arrays
[0095] The disclosure provides a system for the multiplexed detection of a plurality of compounds from single cells comprising an array comprising a plurality of chambers and a panel of capture agents. Preferred capture agents include antibodies, however, capture agents may include any detectable entity that specifically binds to a component of a secretome of the disclosure. The detectable entity may comprise a detectable label, for example. Detectable labels may include, but are not limited to fluorescent labels.
[0096] Systems of the disclosure comprise a plurality of individual chambers, preferably in uniform arrangement. In certain embodiments, at least some of the plurality of individual chambers have a length of greater than 50 .mu.m and, optionally, may be configured to contain an isolated single cell in a sub-nanoliter volume of contents.
[0097] Capture agent panels of the disclosure may comprise a plurality of immobilized capture agents, each immobilized capture agent capable of specifically binding to one of the plurality of components of a secretome of the disclosure. Preferably, the immobilized capture agents are arranged in uniform capture agent panels. Preferably, the immobilized capture agents are attached to a surface in a repeatable pattern, wherein each repeat of the pattern aligns with a chamber of the plurality of chambers.
[0098] The array and capture agent panels are coupled to form a plurality of enclosed interfaces, each enclosed interface comprising a chamber and a capture agent panel such that the contents of each chamber are accessible to each and every capture agent of the capture agent panel.
[0099] Chambers of the array may take on any shape and may have any dimension, however, in certain embodiments of the disclosure, the array comprises at least 1, 2, 5, 10, 15, 20, 25, 50, 100, 150, 500, 1000, 1500, 2000 or any integer between of chambers. Each chamber may have a depth/height of between 1 .mu.m and 2000 .mu.m, a diameter of between 1 .mu.m and 2000 .mu.m, a width of between 1 .mu.m and 2000 .mu.m and/or a length of between 1 .mu.m and 2000 .mu.m. The distance between any two chambers of the array may be between 1 .mu.m and 2000 .mu.m.
[0100] In certain embodiments, at least one chamber is a high aspect ratio rectangular well, having dimensions of about 1-2 mm in length and about 5-50 .mu.m in depth.
[0101] In certain embodiments, each chamber is rectangular with a length of about 10-2000 .mu.m, a width of about 10-100 .mu.m, and a depth of about 10-100 .mu.m.
[0102] In certain embodiments, the capture agent panel may comprise between 3 and 50 different capture agents, thereby allowing for the detection of between 3 and 50 different components of a secretome. In certain embodiments, the capture agent panel may comprise greater than 3 different capture agents, thereby allowing for the detection of greater than 3 different components of a secretome. In certain embodiments, the capture agent panel may comprise greater than 10 different capture agents, thereby allowing for the detection of greater than 10 different components of a secretome. In certain embodiments, the capture agent panel may comprise greater than 42 different capture agents, thereby allowing for the detection of greater than 42 different components of a secretome.
[0103] In certain embodiments, the array comprises a chamber density of about 200 microwells per cm.sup.2 to about 20,000 microchambers per cm.sup.2.
Definitions
[0104] Unless otherwise defined, scientific and technical terms used in connection with the disclosure shall have the meanings that are commonly understood by those of ordinary skill in the art. Further, unless otherwise required by context, singular terms shall include pluralities and plural terms shall include the singular. Generally, nomenclatures utilized in connection with, and techniques of, cell and tissue culture, molecular biology, and protein and oligo- or polynucleotide chemistry and hybridization described herein are those well-known and commonly used in the art. Standard techniques are used for recombinant DNA, oligonucleotide synthesis, and tissue culture and transformation (e.g., electroporation, lipofection). Enzymatic reactions and purification techniques are performed according to manufacturer's specifications or as commonly accomplished in the art or as described herein. The practice of the present invention will employ, unless indicated specifically to the contrary, conventional methods of virology, immunology, microbiology, molecular biology and recombinant DNA techniques within the skill of the art, many of which are described below for the purpose of illustration. Such techniques are explained fully in the literature. See, e.g., Sambrook, et al. Molecular Cloning: A Laboratory Manual (2nd Edition, 1989); Maniatis et al. Molecular Cloning: A Laboratory Manual (1982); DNA Cloning: A Practical Approach, vol. I & II (D. Glover, ed.); Oligonucleotide Synthesis (N. Gait, ed., 1984); Nucleic Acid Hybridization (B. Hames & S. Higgins, eds., 1985); Transcription and Translation (B. Hames & S. Higgins, eds., 1984); Animal Cell Culture (R. Freshney, ed., 1986); Perbal, A Practical Guide to Molecular Cloning (1984).
[0105] The following definitions are useful in understanding the present invention:
[0106] The term "antibody" (Ab) as used herein includes monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g., bispecific antibodies), and antibody fragments, as long as they exhibit the desired biological activity. The term "immunoglobulin" (Ig) is used interchangeably with "antibody" herein.
[0107] An "isolated antibody" is one that has been separated and/or recovered from a component of its natural environment. Contaminant components of its natural environment are materials that would interfere with diagnostic or therapeutic uses for the antibody, and may include enzymes, hormones, and other proteinaceous or nonproteinaceous solutes. In preferred embodiments, the antibody is purified: (1) to greater than 95% by weight of antibody as determined by the Lowry method, and most preferably more than 99% by weight; (2) to a degree sufficient to obtain at least 15 residues of N-terminal or internal amino acid sequence by use of a spinning cup sequenator; or (3) to homogeneity by SDS-PAGE under reducing or non-reducing conditions using Coomassie blue or, preferably, silver stain. Isolated antibody includes the antibody in situ within recombinant cells since at least one component of the antibody's natural environment will not be present. Ordinarily, however, isolated antibody will be prepared by at least one purification step.
[0108] Capture agents of the disclosure may comprise one or more monoclonal antibodies. The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical except for possible naturally occurring mutations that may be present in minor amounts. Monoclonal antibodies are highly specific, being directed against a single antigenic site. Furthermore, in contrast to polyclonal antibody preparations that include different antibodies directed against different determinants (epitopes), each monoclonal antibody is directed against a single determinant on the antigen. In addition to their specificity, the monoclonal antibodies are advantageous in that they may be synthesized uncontaminated by other antibodies.
[0109] Monoclonal antibodies contemplated herein include "chimeric" antibodies in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity. Chimeric antibodies of primary interest herein include antibodies having one or more human antigen binding sequences (e.g., CDRs) and containing one or more sequences derived from a non-human antibody, e.g., an FR or C region sequence. In addition, chimeric antibodies of primary interest herein include those comprising a human variable domain antigen binding sequence of one antibody class or subclass and another sequence, e.g., FR or C region sequence, derived from another antibody class or subclass. Chimeric antibodies of interest herein also include those containing variable domain antigen-binding sequences related to those described herein or derived from a different species, such as a non-human primate (e.g., Old World Monkey, Ape, etc). Chimeric antibodies also include primatized and humanized antibodies.
[0110] Capture agents of the disclosure may comprise humanized antibodies. A "humanized antibody" is generally considered to be a human antibody that has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are often referred to as "import" residues, which are typically taken from an "import" variable domain. Humanization is traditionally performed by substituting import hypervariable region sequences for the corresponding sequences of a human antibody. Accordingly, such "humanized" antibodies are chimeric antibodies wherein substantially less than an intact human variable domain has been substituted by the corresponding sequence from a non-human species.
[0111] A "human antibody" is an antibody containing only sequences present in an antibody naturally produced by a human. However, as used herein, human antibodies may comprise residues or modifications not found in a naturally occurring human antibody, including those modifications and variant sequences described herein. These are typically made to further refine or enhance antibody performance.
[0112] Capture agents of the disclosure may comprise intact antibodies. An "intact" antibody is one that comprises an antigen-binding site as well as a CL and at least heavy chain constant domains, CH 1, CH 2 and CH 3. The constant domains may be native sequence constant domains (e.g., human native sequence constant domains) or amino acid sequence variant thereof. Preferably, the intact antibody has one or more effector functions.
[0113] Capture agents of the disclosure may comprise an antibody fragment. An "antibody fragment" comprises a portion of an intact antibody, preferably the antigen binding or variable region of the intact antibody. Examples of antibody fragments include Fab, Fab', F(ab')2, and Fv fragments; diabodies; linear antibodies; single-chain antibody molecules; and multispecific antibodies formed from antibody fragments.
[0114] Capture agents of the disclosure may comprise a functional fragment or an analog of an antibody. The phrase "functional fragment or analog" of an antibody is a compound having qualitative biological activity in common with a full-length antibody. For example, a functional fragment or analog of an anti-IgE antibody is one that can bind to an IgE immunoglobulin in such a manner so as to prevent or substantially reduce the ability of such molecule from having the ability to bind to the high affinity receptor, Fc RI.
[0115] Papain digestion of antibodies produces two identical antigen-binding fragments, called "Fab" fragments, and a residual "Fc" fragment, a designation reflecting the ability to crystallize readily. The Fab fragment consists of an entire L chain along with the variable region domain of the H chain (VH), and the first constant domain of one heavy chain (CH 1). Each Fab fragment is monovalent with respect to antigen binding, i.e., it has a single antigen-binding site. Pepsin treatment of an antibody yields a single large F(ab')2 fragment that roughly corresponds to two disulfide linked Fab fragments having divalent antigen-binding activity and is still capable of cross-linking antigen. Fab' fragments differ from Fab fragments by having additional few residues at the carboxy terminus of the CH1 domain including one or more cysteines from the antibody hinge region. Fab'-SH is the designation herein for Fab' in which the cysteine residue(s) of the constant domains bear a free thiol group. F(ab')2 antibody fragments originally were produced as pairs of Fab' fragments that have hinge cysteines between them. Other chemical couplings of antibody fragments are also known.
[0116] The "Fc" fragment comprises the carboxy-terminal portions of both H chains held together by disulfides. The effector functions of antibodies are determined by sequences in the Fc region, which region is also the part recognized by Fc receptors (FcR) found on certain types of cells.
[0117] "Fv" is the minimum antibody fragment that contains a complete antigen-recognition and -binding site. This fragment consists of a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. From the folding of these two domains emanate six hypervariable loops (three loops each from the H and L chain) that contribute the amino acid residues for antigen binding and confer antigen binding specificity to the antibody. However, even a single variable domain (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although at a lower affinity than the entire binding site.
[0118] Capture agents of the disclosure may comprise single-chain antibodies (also referred to as scFv). "Single-chain Fv" also abbreviated as "sFv" or "scFv" are antibody fragments that comprise the VH and VL antibody domains connected into a single polypeptide chain. Preferably, the sFv polypeptide further comprises a polypeptide linker between the VH and VL domains that enables the sFv to form the desired structure for antigen binding. For a review of sFv, see Pluckthun in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., Springer-Verlag, N.Y., pp. 269-315 (1994); Borrebaeck 1995, infra.
[0119] Capture agents of the disclosure may comprise diabodies. The term "diabodies" refers to small antibody fragments prepared by constructing sFv fragments (see preceding paragraph) with short linkers (about 5-10 residues) between the VH and VL domains such that inter-chain but not intra-chain pairing of the V domains is achieved, resulting in a bivalent fragment, i.e., fragment having two antigen-binding sites. Bispecific diabodies are heterodimers of two "crossover" sFv fragments in which the VH and VL domains of the two antibodies are present on different polypeptide chains. Diabodies are described more fully in, for example, EP 404,097; WO 93/11161; and Hollinger et al., Proc. Natl. Acad. Sci. USA, 90:6444-6448 (1993).
[0120] Capture agents of the disclosure may comprise bispecific antibodies. In certain embodiments, antibodies of the present invention are bispecific or multi-specific. Bispecific antibodies are antibodies that have binding specificities for at least two different epitopes. Exemplary bispecific antibodies may bind to two different epitopes of a single antigen. Other such antibodies may combine a first antigen binding site with a binding site for a second antigen.
[0121] Methods for making bispecific antibodies are known in the art. Traditional production of full length bispecific antibodies is based on the co-expression of two immunoglobulin heavy chain-light chain pairs, where the two chains have different specificities. Because of the random assortment of immunoglobulin heavy and light chains, these hybridomas (quadromas) produce a potential mixture of ten different antibody molecules, of which only one has the correct bispecific structure. Purification of the correct molecule, which is usually done by affinity chromatography steps, is rather cumbersome, and the product yields are low.
[0122] According to a different approach, antibody variable domains with the desired binding specificities (antibody-antigen combining sites) are fused to immunoglobulin constant domain sequences. Preferably, the fusion is with an Ig heavy chain constant domain, comprising at least part of the hinge, C.sub.H2, and C.sub.H3 regions. It is preferred to have the first heavy-chain constant region (C.sub.H1) containing the site necessary for light chain bonding, present in at least one of the fusions. DNAs encoding the immunoglobulin heavy chain fusions and, if desired, the immunoglobulin light chain, are inserted into separate expression vectors, and are co-transfected into a suitable host cell. This provides for greater flexibility in adjusting the mutual proportions of the three polypeptide fragments in embodiments when unequal ratios of the three polypeptide chains used in the construction provide the optimum yield of the desired bispecific antibody. It is, however, possible to insert the coding sequences for two or all three polypeptide chains into a single expression vector when the expression of at least two polypeptide chains in equal ratios results in high yields or when the ratios have no significant effect on the yield of the desired chain combination.
[0123] As used herein, an antibody is said to be "immunospecific," "specific for" or to "specifically bind" an antigen if it reacts at a detectable level with the antigen, preferably with an affinity constant, K.sub.a, of greater than or equal to about 10.sup.4 M.sup.-1, or greater than or equal to about 10.sup.5 M.sup.-1, greater than or equal to about 10.sup.6 M.sup.-1, greater than or equal to about 10.sup.7 M.sup.-1, or greater than or equal to 10.sup.8 M.sup.-1. Affinity of an antibody for its cognate antigen is also commonly expressed as a dissociation constant KD, and in certain embodiments, an antibody specifically binds to a component of a secretome if it binds with a KD of less than or equal to 10.sup.-1 M, less than or equal to about 10.sup.-5 M, less than or equal to about 10.sup.-6 M, less than or equal to 10.sup.-7 M, or less than or equal to 10.sup.-8 M. Affinities of antibodies can be readily determined using conventional techniques, for example, those described by Scatchard et al. (Ann. N.Y. Acad. Sci. USA 51:660 (1949)).
[0124] Subject and target cells of the disclosure may be isolated, derived, or prepared from any species, including any mammal. A "mammal" for purposes of treating n infection, refers to any mammal, including humans, domestic and farm animals, and zoo, sports, or pet animals, such as dogs, cats, cattle, horses, sheep, pigs, goats, rabbits, etc. Preferably, the mammal is human.
[0125] Subject cells of the disclosure may be used in a cellular therapy for the treatment of a disease or disorder. "Treating" or "treatment" or "alleviation" refers to both therapeutic treatment and prophylactic or preventative measures; wherein the object is to prevent or slow down (lessen) the targeted pathologic condition or disorder. Those in need of treatment include those already with the disorder as well as those prone to have the disorder or those in whom the disorder is to be prevented. A subject or mammal may be successfully "treated" when, after receiving a cellular therapy with a subject cell of the disclosure, the patient shows observable and/or measurable reduction in or absence of one or more of the following: reduction in one or more of the symptoms associated with disease or disorder; reduced morbidity and mortality, and improvement in quality of life issues. The above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician. Methods of the disclosure may be used to determine the safety and/or efficacy of a cellular therapy before, during or after initiation of treatment of the subject.
[0126] Capture agents of the disclosure may be labeled to render them detectable using one or more means. "Label" as used herein refers to a detectable compound or composition that is conjugated directly or indirectly to the capture agent (e.g. an antibody) so as to generate a "labeled" capture agent (e.g. an antibody). The label may be detectable by itself (e.g., a fluorescent label) or, in the case of an enzymatic label, may catalyze chemical alteration of a substrate compound or composition that is detectable.
[0127] Capture agents of the disclosure may selectively or specifically identify, capture, and/or quantify one or more small molecules in a secretome. A "small molecule" is defined herein to have a molecular weight below about 500 Daltons.
[0128] Capture agents of the disclosure may include nucleic acids or labeled nucleic acids. The terms "nucleic acid" and "polynucleotide" are used interchangeably herein to refer to single- or double-stranded RNA, DNA, or mixed polymers. Polynucleotides may include genomic sequences, extra-genomic and plasmid sequences, and smaller engineered gene segments that express, or may be adapted to express polypeptides.
[0129] An "isolated nucleic acid" is a nucleic acid that is substantially separated from other genome DNA sequences as well as proteins or complexes such as ribosomes and polymerases, which naturally accompany a native sequence. The term embraces a nucleic acid sequence that has been removed from its naturally occurring environment, and includes recombinant or cloned DNA isolates and chemically synthesized analogues or analogues biologically synthesized by heterologous systems. A substantially pure nucleic acid includes isolated forms of the nucleic acid. Of course, this refers to the nucleic acid as originally isolated and does not exclude genes or sequences later added to the isolated nucleic acid by the hand of man.
[0130] The term "polypeptide" is used in its conventional meaning, i.e., as a sequence of amino acids. The polypeptides are not limited to a specific length of the product. Peptides, oligopeptides, and proteins are included within the definition of polypeptide, and such terms may be used interchangeably herein unless specifically indicated otherwise. This term also does not refer to or exclude post-expression modifications of the polypeptide, for example, glycosylations, acetylations, phosphorylations and the like, as well as other modifications known in the art, both naturally occurring and non-naturally occurring. A polypeptide may be an entire protein, or a subsequence thereof.
[0131] An "isolated polypeptide" is one that has been identified and separated and/or recovered from a component of its natural environment. In preferred embodiments, the isolated polypeptide will be purified (1) to greater than 95% by weight of polypeptide as determined by the Lowry method, and most preferably more than 99% by weight, (2) to a degree sufficient to obtain at least 15 residues of N-terminal or internal amino acid sequence by use of a spinning cup sequenator, or (3) to homogeneity by SDS-PAGE under reducing or non-reducing conditions using Coomassie blue or, preferably, silver stain. Isolated polypeptide includes the polypeptide in situ within recombinant cells since at least one component of the polypeptide's natural environment will not be present. Ordinarily, however, isolated polypeptide will be prepared by at least one purification step.
[0132] A "native sequence" polynucleotide is one that has the same nucleotide sequence as a polynucleotide derived from nature. A "native sequence" polypeptide is one that has the same amino acid sequence as a polypeptide (e.g., antibody) derived from nature (e.g., from any species). Such native sequence polynucleotides and polypeptides can be isolated from nature or can be produced by recombinant or synthetic means.
[0133] A polynucleotide "variant," as the term is used herein, is a polynucleotide that typically differs from a polynucleotide specifically disclosed herein in one or more substitutions, deletions, additions and/or insertions. Such variants may be naturally occurring or may be synthetically generated, for example, by modifying one or more of the polynucleotide sequences of the invention and evaluating one or more biological activities of the encoded polypeptide as described herein and/or using any of a number of techniques well known in the art.
[0134] A polypeptide "variant," as the term is used herein, is a polypeptide that typically differs from a polypeptide specifically disclosed herein in one or more substitutions, deletions, additions and/or insertions. Such variants may be naturally occurring or may be synthetically generated, for example, by modifying one or more of the above polypeptide sequences of the invention and evaluating one or more biological activities of the polypeptide as described herein and/or using any of a number of techniques well known in the art.
[0135] Modifications may be made in the structure of the polynucleotides and polypeptides of the disclosure and still obtain a functional molecule that encodes a variant or derivative polypeptide with desirable characteristics. When it is desired to alter the amino acid sequence of a polypeptide to create an equivalent, or even an improved, variant or portion of a polypeptide of the invention, one skilled in the art will typically change one or more of the codons of the encoding DNA sequence.
[0136] For example, certain amino acids may be substituted for other amino acids in a protein structure without appreciable loss of its ability to bind other polypeptides (e.g., antigens) or cells. Since it is the binding capacity and nature of a protein that defines that protein's biological functional activity, certain amino acid sequence substitutions can be made in a protein sequence, and, of course, its underlying DNA coding sequence, and nevertheless obtain a protein with like properties. It is thus contemplated that various changes may be made in the peptide sequences of the disclosed compositions, or corresponding DNA sequences that encode said peptides without appreciable loss of their biological utility or activity.
[0137] In many instances, a polypeptide variant will contain one or more conservative substitutions. A "conservative substitution" is one in which an amino acid is substituted for another amino acid that has similar properties, such that one skilled in the art of peptide chemistry would expect the secondary structure and hydropathic nature of the polypeptide to be substantially unchanged.
[0138] In making such changes, the hydropathic index of amino acids may be considered. The importance of the hydropathic amino acid index in conferring interactive biologic function on a protein is generally understood in the art (Kyte and Doolittle, 1982). It is accepted that the relative hydropathic character of the amino acid contributes to the secondary structure of the resultant protein, which in turn defines the interaction of the protein with other molecules, for example, enzymes, substrates, receptors, DNA, antibodies, antigens, and the like. Each amino acid has been assigned a hydropathic index on the basis of its hydrophobicity and charge characteristics (Kyte and Doolittle, 1982). These values are: isoleucine (+4.5); valine (+4.2); leucine (+3.8); phenylalanine (+2.8); cysteine/cystine (+2.5); methionine (+1.9); alanine (+1.8); glycine (-0.4); threonine (-0.7); serine (-0.8); tryptophan (-0.9); tyrosine (-1.3); proline (-1.6); histidine (-3.2); glutamate (-3.5); glutamine (-3.5); aspartate (-3.5); asparagine (-3.5); lysine (-3.9); and arginine (-4.5).
[0139] Certain amino acids may be substituted by other amino acids having a similar hydropathic index or score and still result in a protein with similar biological activity, i.e. still obtain a biological functionally equivalent protein. In making such changes, the substitution of amino acids whose hydropathic indices are within .+-.2 is preferred, those within .+-.1 are particularly preferred, and those within .+-.0.5 are even more particularly preferred. The substitution of like amino acids can be made effectively on the basis of hydrophilicity. The greatest local average hydrophilicity of a protein, as governed by the hydrophilicity of its adjacent amino acids, correlates with a biological property of the protein.
[0140] The following hydrophilicity values have been assigned to amino acid residues: arginine (+3.0); lysine (+3.0); aspartate (+3.0.+-.1); glutamate (+3.0.+-.1); serine (+0.3); asparagine (+0.2); glutamine (+0.2); glycine (0); threonine (-0.4); proline (-0.5.+-.1); alanine (-0.5); histidine (-0.5); cysteine (-1.0); methionine (-1.3); valine (-1.5); leucine (-1.8); isoleucine (-1.8); tyrosine (-2.3); phenylalanine (-2.5); tryptophan (-3.4). It is understood that an amino acid can be substituted for another having a similar hydrophilicity value and still obtain a biologically equivalent, and in particular, an immunologically equivalent protein. In such changes, the substitution of amino acids whose hydrophilicity values are within .+-.2 is preferred, those within .+-.1 are particularly preferred, and those within .+-.0.5 are even more particularly preferred.
[0141] As outlined above, amino acid substitutions are generally therefore based on the relative similarity of the amino acid side-chain substituents, for example, their hydrophobicity, hydrophilicity, charge, size, and the like. Exemplary substitutions that take various of the foregoing characteristics into consideration are well known to those of skill in the art and include: arginine and lysine; glutamate and aspartate; serine and threonine; glutamine and asparagine; and valine, leucine and isoleucine.
[0142] Amino acid substitutions may further be made on the basis of similarity in polarity, charge, solubility, hydrophobicity, hydrophilicity and/or the amphipathic nature of the residues. For example, negatively charged amino acids include aspartic acid and glutamic acid; positively charged amino acids include lysine and arginine; and amino acids with uncharged polar head groups having similar hydrophilicity values include leucine, isoleucine and valine; glycine and alanine; asparagine and glutamine; and serine, threonine, phenylalanine and tyrosine. Other groups of amino acids that may represent conservative changes include: (1) ala, pro, gly, glu, asp, gln, asn, ser, thr; (2) cys, ser, tyr, thr; (3) val, ile, leu, met, ala, phe; (4) lys, arg, his; and (5) phe, tyr, trp, his. A variant may also, or alternatively, contain nonconservative changes. In a preferred embodiment, variant polypeptides differ from a native sequence by substitution, deletion or addition of five amino acids or fewer. Variants may also (or alternatively) be modified by, for example, the deletion or addition of amino acids that have minimal influence on the immunogenicity, secondary structure and hydropathic nature of the polypeptide.
[0143] When comparing polynucleotide and polypeptide sequences, two sequences are said to be "identical" if the sequence of nucleotides or amino acids in the two sequences is the same when aligned for maximum correspondence, as described below. Comparisons between two sequences are typically performed by comparing the sequences over a comparison window to identify and compare local regions of sequence similarity. A "comparison window" as used herein, refers to a segment of at least about 20 contiguous positions, usually 30 to about 75, 40 to about 50, in which a sequence may be compared to a reference sequence of the same number of contiguous positions after the two sequences are optimally aligned.
[0144] Optimal alignment of sequences for comparison may be conducted using the Megalign program in the Lasergene suite of bioinformatics software (DNASTAR, Inc., Madison, Wis.), using default parameters. This program embodies several alignment schemes described in the following references: Dayhoff, Mo. (1978) A model of evolutionary change in proteins--Matrices for detecting distant relationships. In Dayhoff, Mo. (ed.) Atlas of Protein Sequence and Structure, National Biomedical Research Foundation, Washington D.C. Vol. 5, Suppl. 3, pp. 345-358; Hein J. (1990) Unified Approach to Alignment and Phylogenes pp. 626-645 Methods in Enzymology vol. 183, Academic Press, Inc., San Diego, Calif.; Higgins, D. G. and Sharp, P. M. (1989) CABIOS 5:151-153; Myers, E. W. and Muller W. (1988) CABIOS 4:11-17; Robinson, E. D. (1971) Comb. Theor 11:105; Santou, N. Nes, M. (1987) Mol. Biol. Evol. 4:406-425; Sneath, P. H. A. and Sokal, R. R. (1973) Numerical Taxonomy--the Principles and Practice of Numerical Taxonomy, Freeman Press, San Francisco, Calif.; Wilbur, W. J. and Lipman, D. J. (1983) Proc. Natl. Acad., Sci. USA 80:726-730.
[0145] Alternatively, optimal alignment of sequences for comparison may be conducted by the local identity algorithm of Smith and Waterman (1981) Add. APL. Math 2:482, by the identity alignment algorithm of Needleman and Wunsch (1970) J. Mol. Biol. 48:443, by the search for similarity methods of Pearson and Lipman (1988) Proc. Natl. Acad. Sci. USA 85: 2444, by computerized implementations of these algorithms (GAP, BESTFIT, BLAST, FASTA, and TFASTA in the Wisconsin Genetics Software Package, Genetics Computer Group (GCG), 575 Science Dr., Madison, Wis.), or by inspection.
[0146] One preferred example of algorithms that are suitable for determining percent sequence identity and sequence similarity are the BLAST and BLAST 2.0 algorithms, which are described in Altschul et al. (1977) Nucl. Acids Res. 25:3389-3402 and Altschul et al. (1990) J. Mol. Biol. 215:403-410, respectively. BLAST and BLAST 2.0 can be used, for example with the parameters described herein, to determine percent sequence identity for the polynucleotides and polypeptides of the invention. Software for performing BLAST analyses is publicly available through the National Center for Biotechnology Information.
[0147] "Homology" refers to the percentage of residues in the polynucleotide or polypeptide sequence variant that are identical to the non-variant sequence after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent homology. In particular embodiments, polynucleotide and polypeptide variants have at least 70%, at least 75%, at least 80%, at least 90%, at least 95%, at least 98%, or at least 99% polynucleotide or polypeptide homology with a polynucleotide or polypeptide described herein.
[0148] As used in this specification and the appended claims, the singular forms "a," "an" and "the" include plural references unless the content clearly dictates otherwise.
EXAMPLES
[0149] The methods detailed below are capable of analyzing distinct single subject cell to single target cell interactions on a system of the disclosure (also referred to below as an array). Cell populations may be cultured separately and briefly mixed before being loading onto the array. The array may comprise a plurality of chambers (which, for these examples, may take the form of micro-troughs) that are enclosed by surface (e.g. a glass slide or other cover material) creating isolated microenvironments for high throughput cellular studies such as paired cell interactions. The surface enclosing each chamber of the array may have attached thereto one or more detection agents (including, but not limited to antibodies). The detection agents (e.g. antibodies in the Examples provided below) may be attached to the surface in a repeated pattern such that each chamber comprises one repeat of the pattern on the portion of the surface enclosing the chamber. In this circumstance, each repeat of the pattern comprises at least one of every individual detection agent (e.g. antibody specific for a particular cytokine). The surface may have attached thereto between 20-1000 detection agents that are each specific for a distinct component of a subject cell's secretome. The systems of the disclosure include a plurality of chambers. The plurality of chambers may include, for example, between 2 and 2000 chambers.
Example 1: Analysis of Natural Killer (NK) Cell and Target Cell
[0150] Cell Culture. Isolated peripheral blood mononuclear cells (PBMCs), stored in-house at a concentration of 2.5.times.10.sup.7 cells/vial, were thawed and cultured overnight in a T25 cell culture flask with 5 mL of X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1 mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083), 100 ng/mL recombinant IL-18 (R&D Systems, B001-5) and 10 ng/mL IL-12 (R&D Systems, 219-IL-005). After an overnight recovery, NK cells were isolated using the Miltenyi NK Cell Isolation Kit according to the manufacturer's instructions. NK cells were then cultured at 37.degree. C., 5% CO.sub.2 in supplemented X-Vivo 15 media at a concentration of 1.times.10.sup.6 cells/mL until use. A human K562 Cell line (ATCC CCL243) was purchased from the American Type Culture Collection (ATCC) and cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061).
[0151] Single-Cell Secretome Assay. Before performing the single cell assay, the system of the disclosure (in this example, a polydimethylsiloxane (PDMS) microchamber array) was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. Immediately prior to the assay, K562 cells were taken from culture, resuspended in 1 mL of PBS and stained with CellTrace Carboxyfluorescein succinimidyl ester (CFSE) (1:1000, ThermoFisher) for 20 minutes. K562 cells were then washed 3.times. with supplemented X-Vivo 15 media and resuspended at a concentration of 1.times.10.sup.6 cells/mL. K562 cells and NK cells were then combined at a 1:1 ratio to generate a cell suspension. This suspension containing K562 cells and NK cells was centrifuged at 300.times.g for 10 minutes and re-suspended in fresh media at a concentration of 2.5.times.10.sup.6 cells/mL. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was positioned onto a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegend, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0152] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flash4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0153] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
Example 2: Analysis of CD8+T-Cell and Target Cell with Bispecific Antibodies
[0154] Cell Culture. Isolated PBMCs, stored in-house at a concentration of 2.5.times.10.sup.7 cells/vial, were thawed and cultured overnight in a T25 cell culture flask with 5 mL of X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1 mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083), and 4 ng/mL recombinant IL-2 (BioLegend). After an overnight recovery, CD8+ T-cells were isolated using the Miltenyi CD8+ T-Cell Isolation Kit according to the manufacturer's instructions. CD8+ T-cells were then cultured at 37.degree. C., 5% CO.sub.2 in supplemented X-Vivo 15 media at a concentration of 1.times.10.sup.6 cells/mL until use. The human Raji B Cell line (purchased from the American Type Culture Collection (ATCC)) was cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061).
[0155] Single-Cell Secretome Assay. Before performing the single cell assay, the system of the disclosure (in this example, a polydimethylsiloxane (PDMS) microchamber array) was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. CD8+ T-cells were resuspended in 1 mL of PBS and stained with CellTrace Violet (1:1000, ThermoFisher) for 20 minutes. CD8+ T-cells were then washed 3.times. with supplemented X-Vivo 15 media and resuspended at a concentration of 2.times.10.sup.6 cells/mL. Raji cells were taken from culture and resuspended in 1 mL of PBS and stained with Cell Trace CFSE (1:1000, ThermoFisher) for 20 minutes. Raji cells were then washed 3.times. with supplemented X-Vivo 15 media and resuspended at a concentration of 2.times.10.sup.6 cells/mL. CD8+ T-cells and Raji cells were then combined at a 1:1 ratio to generate a cell suspension. Subsequently, 100 ng/mL of a bispecific antibody was added to this suspension. The cell suspension containing CD8+ T-cells and Raji cells was centrifuged for 10 minutes at 300.times.g and then resuspended before loading onto the array. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was then positioned on a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegened, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0156] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flash4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0157] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
Example 3: Analysis of Interactions between CAR T-Cells and Stimulation Beads
[0158] CD19 Bead Design. 1.times.10.sup.8 M450 Tosylactivated Dynabeads (ThermoFisher) (250 .mu.L of the M450 Tosylactivated Dynabeads) were washed 3.times. with PBS. Recombinant human CD19-Histidine (His) (Sino) was resuspeneded in dH20 at 1 .mu.g/.mu.L and incubated at room temperature for 30 minutes. Fifty microliters of CD19-His was then added to the beads and incubated at room temperature overnight, with rotation. The CD19-coated beads were then washed 3.times. with PBS. To inactivate the remaining tosyl groups on the beads, the beads were resuspended in 250 .mu.L of 0.2M Tris with 0.1% BSA (pH 8.5) and incubated with rotation overnight at room temperature. The beads were then resupended in PBS at a concentration of 4.times.10.sup.6 beads/mL and stored at 4.degree. C. until use.
[0159] CAR T-cell Stimulation. CAR-T cells were mixed with CD19-coated beads in a 1:4 ratio and plated at a concentration of 1.times.10.sup.6 cells/mL in a 96-well plate. The plate was incubated for 6 hours at 37.degree. C. and 5% CO.sub.2 before performing the assay.
[0160] Cell Culture. Target cells were cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061). CAR T-cells were thawed and cultured overnight at a concentration of 1.times.10.sup.6 cells/mL in X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 4 ng/mL recombinant human IL-2 (BioLegend, 589104). Immediately prior to use, dead cells were removed from culture using a Dead Cell Removal Kit (Miltenyi, 130-090-101) and LS Magnetic Column (Miltenyi, 130-042-401) or by using a standard Ficoll-Paque Plus (GE, 17-1440-02) dead cell removal protocol. If it is desired to separate the CD8+ from CD4+ cells in culture, CD8 microbeads (Miltenyi, 130-045-201) may be used for isolation according to the manufacturer's instructions following dead cell removal. Single-Cell Secretome Assay. Before performing the single cell assay, the array was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. Stimulated T-cells were collected, stained at 1:100 with an anti-human CD4 RPE antibody (ThermoFisher, MHCD0404) and an anti-human CD8a Alexa Fluor 647 antibody (BioLegend, 300918) and incubated at room temperature for 10 minutes. The stimulated T-cells were centrifuged at 300.times.g for 10 minutes and re-suspended in fresh media at a concentration of 2.5.times.10.sup.6 cells/mL. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was then positioned on a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegened, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0161] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flash4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0162] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
Example 4: Analysis of Interactions between CAR T-Cells and Target Cells
[0163] Target Cell Design. Target cells are generated by cloning full length cDNA (see list of target cDNAs below) into a pcDNA3.1 (+) vector (ThermoFisher, V79020) using the EcoRI and XhoI cut sites. The target plasmid is transfected into K562 cells (ATCC, CCL243) in a 24-well plate following the standard Lipofectamine 3000 (ThermoFisher) protocol. Stable pool lines are created by selecting transformants with 500 .mu.g/mL Geneticin (ThermoFisher) for 3-4 weeks. Once stable pools are created, clonal lines are developed by serially diluting cells in a 96 well plate. Wells with a single cell are cultured and selected with Geneticin for 3-4 weeks, until a stable clonal line is developed. Clonal lines were frozen at a concentration of 1.times.10.sup.7 cells/vial in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083), 1.times. GlutaMAX supplement (ThermoFisher, 35050061) and 10% DMSO. Target cDNAs include, but are not limited to, the following: Nerve Growth Factor Receptor (NGFR; Negative control cDNA), CD19, Epidermal Growth Factor Receptor (EGFR), Epidermal Growth Factor Receptor type III mutation (EGFRvIII; also referred to as de2-7 EGFR or AEGFR), Melanoma-associated antigen 3 (MAGE A3), NY-ESO-1 (also known as CTAG-1B; an immunogenic cancer antigen), Prostate Stem Cell Antigen (PSCA), Preferentially Expressed Antigen In Melanoma (PRAME), human epidermal growth factor receptor 2 (HER2), B-cell maturation antigen (BCMA; also known as CD296), carcinoembryonic antigen (CEA), Mucin 1 (MUC-1; also known as episialin, PEM, H23Ag, EMA, CA15-3, and MCA), Mucin 16 (MUC-16), and Mesothelin.
[0164] Cell Culture. Target cells were cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061). CAR T-cells were thawed and cultured overnight at a concentration of 1.times.10.sup.6 cells/mL in X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 4 ng/mL recombinant human IL-2 (BioLegend, 589104). Immediately prior to use, dead cells were removed from culture using a Dead Cell Removal Kit (Miltenyi, 130-090-101) and LS Magnetic Column (Miltenyi, 130-042-401) or by using a standard Ficoll-Paque Plus (GE, 17-1440-02) dead cell removal protocol. If it is desired to separate the CD8+ from CD4+ cells in culture, CD8 microbeads (Miltenyi, 130-045-201) may be used for isolation according to the manufacturer's instructions following dead cell removal.
[0165] CAR T-cell Stimulation. Target cells were stained for 20 minutes with CellTrace CFSE (ThermoFisher, C34554) and rinsed 3.times. with supplemented X-Vivo 15 media prior to use. Target cells (at a concentration of 1.times.10.sup.6 cells/mL) and CAR T-cells (at a concentration of 1.times.10.sup.6 cells/mL) were combined in a 1:1 ratio to generate a cell suspension. Two hundred microliters of the cell suspension containing the CAR T-cells and the target cells was plated per well into a 96-well plate and incubated at 37.degree. C., 5% CO.sub.2 for 1-2 hours.
[0166] Single-Cell Secretome Assay. Before performing the single cell assay, the array was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. Stimulated T-cells were collected, stained at 1:100 with an anti-human CD4 RPE antibody (ThermoFisher, MHCD0404) and an anti-human CD8a Alexa Fluor 647 antibody (BioLegend, 300918) and incubated at room temperature for 10 minutes. The stimulated T-cells were centrifuged at 300.times.g for 10 minutes and re-suspended in fresh media at a concentration of 2.5.times.10.sup.6 cells/mL. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was then positioned on a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegened, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0167] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flas4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0168] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
Example 5: Analysis of Interactions between CAR T-Cells and Target Cells with Target Cell Depletion
[0169] Target Cell Design. Target cells are generated by cloning full length cDNA (see list of target cDNAs below) into a pcDNA3.1 (+) vector (ThermoFisher, V79020) using the EcoRI and XhoI cut sites. The target plasmid is transfected into K562 cells (ATCC, CCL243) in a 24-well plate following the standard Lipofectamine 3000 (ThermoFisher) protocol. Stable pool lines are created by selecting transformants with 500 .mu.g/mL Geneticin (ThermoFisher) for 3-4 weeks. Once stable pools are created, clonal lines are developed by serially diluting cells in a 96 well plate. Wells with a single cell are cultured and selected with Geneticin for 3-4 weeks, until a stable clonal line is developed. Clonal lines were frozen at a concentration of 1.times.10.sup.7 cells/vial in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083), 1.times. GlutaMAX supplement (ThermoFisher, 35050061) and 10% DMSO. Target cDNAs include, but are not limited to, the following: Nerve Growth Factor Receptor (NGFR; Negative control cDNA), CD19, Epidermal Growth Factor Receptor (EGFR), Epidermal Growth Factor Receptor type III mutation (EGFRvIII; also referred to as de2-7 EGFR or AEGFR), Melanoma-associated antigen 3 (MAGE A3), NY-ESO-1 (also known as CTAG-1B; an immunogenic cancer antigen), Prostate Stem Cell Antigen (PSCA), Preferentially Expressed Antigen In Melanoma (PRAME), human epidermal growth factor receptor 2 (HER2), B-cell maturation antigen (BCMA; also known as CD296), carcinoembryonic antigen (CEA), Mucin 1 (MUC-1; also known as episialin, PEM, H23Ag, EMA, CA15-3, and MCA), Mucin 16 (MUC-16), and Mesothelin.
[0170] Cell Culture. Target cells were cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061). CAR T-cells were thawed and cultured overnight at a concentration of 1.times.10.sup.6 cells/mL in X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1 mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 4 ng/mL recombinant human IL-2 (BioLegend, 589104). Immediately prior to use, dead cells were removed from culture using a Dead Cell Removal Kit (Miltenyi, 130-090-101) and LS Magnetic Column (Miltenyi, 130-042-401) or by using a standard Ficoll-Paque Plus (GE, 17-1440-02) dead cell removal protocol. If it is desired to separate the CD8+ from CD4+ cells in culture, CD8 microbeads (Miltenyi, 130-045-201) may be used for isolation according to the manufacturer's instructions following dead cell removal.
[0171] CAR T-cell Stimulation. CAR T-cells (at a concentration of 1.times.10.sup.6 cells/mL) and target cells (at a concentration of 1.times.10.sup.6 cells/mL) are mixed in a ratio of 1:1 to generate a cell suspension. Two hundred microliters of the cell suspension is plated per well of a 96-well plate. The plate is then incubated overnight (in this example is approximately 20 hours) in an incubator at 37.degree. C. with 5% CO.sub.2.
[0172] Target Cell Depletion. After an overnight stimulation, the cell suspension containing CAR T-cells and target cells is removed from the wells and centrifuged for 10 minutes at 300.times.g. The supernatant is then removed and the cells are resuspended in a conjugated bead solution. The conjugated bead solution contains M280 Dynabeads conjugated to an antibody against the target receptor. The beads of the conjugated bead solution are designed to remove all of the target cells from the solution. This conjugated bead solution containing the CAR T-cells and the target cells is incubated, with gentle mixing, at room temperature for 10 minutes. After the incubation, the solution containing the CAR T-cells and the target cells is brought up to a total volume of 1 mL with PBS and transferred to a 5 mL round bottom polypropylene falcon tube (Corning, 352063). The tube is then placed in an EasySep Magnet (StemCell Technologies, 18000) for 2 minutes before decanting the solution of CAR T-cells into a clean tube. The beads are washed with media and decanted 2.times. before centrifuging the CAR T-cells and resuspending the target cell depleted CAR-T cells for use in the assay.
[0173] Single-Cell Secretome Assay. Before performing the single cell assay, the array was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. Stimulated T-cells were collected, stained at 1:100 with an anti-human CD4 RPE antibody (ThermoFisher, MHCD0404) and an anti-human CD8a Alexa Fluor 647 antibody (BioLegend, 300918) and incubated at room temperature for 10 minutes. The stimulated T-cells were centrifuged at 300.times.g for 10 minutes and re-suspended in fresh media at a concentration of 2.5.times.10.sup.6 cells/mL. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was then positioned on a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in a 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegened, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0174] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flash4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0175] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
Example 6: Fluorescent Cytotoxicity Analysis of Target/Effector Interaction
[0176] This method can be applied to any of the target/effector (subject) cell types of the disclosure with the addition of the following step to the Single-Cell Secretome Assay Protocol. If fluorescent tracking of cytotoxicity is desired, 1 .mu.L/mL of SYTOX orange (ThermoFisher) may be added to the cell suspension containing the target cells and effector (subject) cells immediately prior to loading the cell suspension onto the array.
Example 7: Assessing Polyfunctionality in CAR-Expressing Anti-Tumor T-Cells
[0177] To evaluate engineered T cells for an immunotherapy or to evaluate endogenous T cells reactivated to battle cancer or infection, a T cell's functional status is largely determined by a spectrum of secreted effector function proteins (e.g., cytokines). In a protective immune response, the `quality` of an immune cell correlates to the extent of polyfunctionality (the ability of a T cell to co-secrete multiple effector proteins).While these anti-tumor cytolytic, chemoattractive cytokines produce an effective response, these poly-functional cell subsets must not also secrete immuno-toxic inflammatory or regulatory cytokines (up to 15) prevalent in NK or CAR-T cells.
[0178] The following provides one method for determining whether a stimulated CAR-expressing anti-tumor T cell responds in a therapeutically effective and safe manner before administering the cell or substantially similar cell of a population of CAR-expressing anti-tumor T-cells to a patient as a therapeutic.
[0179] Target Cell Design. Target cells are generated by cloning full length cDNA (see list of target cDNAs below) into a pcDNA3.1 (+) vector (ThermoFisher, V79020) using the EcoRI and XhoI cut sites. The target plasmid is transfected into K562 cells (ATCC, CCL243) in a 24-well plate following the standard Lipofectamine 3000 (ThermoFisher) protocol. Stable pool lines are created by selecting transformants with 500 .mu.g/mL Geneticin (ThermoFisher) for 3-4 weeks. Once stable pools are created, clonal lines are developed by serially diluting cells in a 96 well plate. Wells with a single cell are cultured and selected with Geneticin for 3-4 weeks, until a stable clonal line is developed. Clonal lines were frozen at a concentration of 1.times.10.sup.7 cells/vial in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083), 1.times. GlutaMAX supplement (ThermoFisher, 35050061) and 10% DMSO. Target cDNAs include, but are not limited to, the following: Nerve Growth Factor Receptor (NGFR; Negative control cDNA), CD19, Epidermal Growth Factor Receptor (EGFR), Epidermal Growth Factor Receptor type III mutation (EGFRvIII; also referred to as de2-7 EGFR or AEGFR), Melanoma-associated antigen 3 (MAGE A3), NY-ESO-1 (also known as CTAG-1B; an immunogenic cancer antigen), Prostate Stem Cell Antigen (PSCA), Preferentially Expressed Antigen In Melanoma (PRAME), human epidermal growth factor receptor 2 (HER2), B-cell maturation antigen (BCMA; also known as CD296), carcinoembryonic antigen (CEA), Mucin 1 (MUC-1; also known as episialin, PEM, H23Ag, EMA, CA15-3, and MCA), Mucin 16 (MUC-16), and Mesothelin.
[0180] Cell Culture. Target cells were cultured in Corning.TM. cellgro.TM. RPMI 1640 Medium (Mod.) 1.times. with L-Glutamine (Corning; ThermoFisher MT10040CV) supplemented with 10% FBS (Sigma-Aldrich, F2442), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 1.times. GlutaMAX supplement (ThermoFisher, 35050061). CAR T-cells were thawed and cultured overnight at a concentration of 1.times.10.sup.6 cells/mL in X-Vivo 15 without Gentamicin or phenol red (Lonza, 04-744Q) supplemented with 1.times. GlutaMAX (ThermoFisher, 35050061), 1 mM sodium pyruvate (ThermoFisher, 11360-070), 1.times. MEM Vitamin Solution (Gibco; ThermoFisher, 11120-052), 20 mM HEPES (Gibco; ThermoFisher, 15630-080), 2% Human AB serum (Valley Biomedical, HP1022HI), 1.times. Penicillin-Streptomycin-Neomycin (Sigma-Aldrich, P4083) and 4 ng/mL recombinant human IL-2 (BioLegend, 589104). Immediately prior to use, dead cells were removed from culture using a Dead Cell Removal Kit (Miltenyi, 130-090-101) and LS Magnetic Column (Miltenyi, 130-042-401) or by using a standard Ficoll-Paque Plus (GE, 17-1440-02) dead cell removal protocol. If it is desired to separate the CD8+ from CD4+ cells in culture, CD8 microbeads (Miltenyi, 130-045-201) may be used for isolation according to the manufacturer's instructions following dead cell removal.
[0181] CAR T-cell Stimulation. Target cells were stained for 20 minutes with CellTrace CFSE (ThermoFisher, C34554) and rinsed 3.times. with supplemented X-Vivo 15 media prior to use. Target cells (at a concentration of 1.times.10.sup.6 cells/mL) and CAR T-cells (at a concentration of 1.times.10.sup.6 cells/mL) were combined in a 1:1 ratio to generate a cell suspension. Two hundred microliters of the cell suspension containing the CAR T-cells and the target cells was plated per well into a 96-well plate and incubated at 37.degree. C., 5% CO.sub.2 for 1-2 hours.
[0182] Single-Cell Secretome Assay. Before performing the single cell assay, the array was plasma treated for 2.5 minutes with a Plasma Etch PE-25 plasma cleaner to increase hydrophilicity. The array was then blocked in 3% BSA/PBS for 30 minutes. Stimulated T-cells were collected, stained at 1:100 with an anti-human CD4 RPE antibody (ThermoFisher, MHCD0404) and an anti-human CD8a Alexa Fluor 647 antibody (BioLegend, 300918) and incubated at room temperature for 10 minutes. The stimulated T-cells were centrifuged at 300.times.g for 10 minutes and re-suspended in fresh media at a concentration of 2.5.times.10.sup.6 cells/mL. Immediately before the assay, the array was rinsed with media and dried using compressed air. The array was then positioned on a glass slide and secured into a custom clamping system. Thirty microliters of the cell suspension was pipetted, chamber-by-chamber, onto the array. A surface (a glass side in this example) with the a repeated pattern of antibodies attached thereto was contacted to the array such that the repeated pattern of antibodies was facing the array and such that each repeat of the pattern aligned with a chamber of the array. The array and the surface (with the antibody pattern) enclosing the array were clamped tightly together in our custom clamping system. Cells were trapped in microchambers and imaged immediately after loading as described below. The microchamber assembly was placed in a standard 5% CO.sub.2, incubator at 37.degree. C. for 16 hours. Following incubation, the surface with the antibody pattern was removed in a 1% BSA/PBS bath and rinsed with 1% BSA/PBS. Subsequently, 300 .mu.L of a biotin-labeled secondary antibody cocktail was introduced to the surface and incubated for 45 minutes. This cocktail contains detection antibodies at a concentration of 0.25 .mu.g/mL each in 1:200 suspension of 1% BSA/PBS. Following this step, the surface was rinsed with 1% BSA, 300 .mu.L of a 1:100 solution of APC streptavidin (BioLegened, 405207) was added and the surface was incubated for 30 minutes. Following this incubation, the surface was washed with 1% BSA/PBS then washed in coplin jars PBS, 50% PBS/DI water, DI water, DI water sequentially for 3 minutes in a centrifuge set at 125 rpm. The surface was dried for 30 seconds in a Labnet slide spinner (C1303-T). The surface was analyzed using a GenePix microarray scanner as described below.
[0183] Microchamber Array Imaging. A Zeiss Axio Observer.Z1 fluorescent microscope with an automatic stage was used to acquire both bright field and fluorescent images of the array. Images of the entire array were taken with a Hamamatsu Orca-Flash4.0 LT Digital CMOS camera (C11440-42U) of a 54183 uM.times.24866 uM area corresponding to either 253 tiles (5.times. image) or 494 tiles (10.times. image). Using Zen2 Pro software, the tiles were stitched together with 0% overlap and were exported as TIFFs for analysis by our proprietary CytoSpeak software.
[0184] Imaging of Antibody-Patterned Surface. GenePix 4400A scanners (Molecular Devices) were used to obtain scanned fluorescent images of the antibody-patterned surfaces. Two color channels, 488 (blue, PMT 350, Power 90) and 635 (red, PMT 600, Power 90) were used to collect fluorescence signals using GenePix Pro software (Molecular Devices). The image was then exported as a TIFF before analysis using CytoSpeak software.
[0185] Analysis of polyfunctionality: The absolute and relative contributions of each cytokine in the secrotome of the control and target-cell stimulated CAR T-cells were measured. FIGS. 10 and 11 demonstrate on a single-cell basis the Polyfunctional Strength Index (PSI) contributions of each detected cytokine in the secretome. As shown in FIG. 11, panels E and F, when a population of cells are detected individually, but analyzed as a population, a percentage of cells in the given population that express any one cytokine above a threshold level may be determined. The cytokines used in this plot may include those cytokines that indicate a risk of the CAR-T cell inducing a deleterious or unwanted reaction in vivo and the threshold may be raised and lowered according to what is considered safe and tolerable. Using this method, any population of cells intended for administration in vivo as a cell therapy may be tested in vitro to reveal potential harmful reactions before the cell population can negatively impact a patient.
[0186] The polyfunctional strength index (PSI) is a metric that factors in the polyfunctionality of cells in a sample, and the signal intensity of the cytokines secreted by each cell. It is found by multiplying the percentage of polyfunctional cells of a sample (single cells secreting two or more cytokines), by the average signal intensity of these cytokines. This PSI is shown on the left of FIGS. 10A and 10B, as well as FIGS. 11A and 11B. For example, FIG. 10B demonstrates that the polyfunctional strength is roughly 2.times. higher in the CD19 stimulated sample, relative to the control PSI shown in FIG. 10A. FIG. 11B demonstrates that the stimulated cells have a polyfunctional strength roughly 5.times. higher than the control cells.
[0187] As seen in the bar graphs of FIGS. 10 and 11, the polyfunctional strength can be further broken down into defined cytokine groups (see the key shown in FIG. 8E), illustrating the impact of particular groups of cytokines on the sample's polyfunctionality. In the left graph of FIG. 10B, for example, effector cytokines are major drivers of the polyfunctionality, as they account for about 75% of the total PSI. The corresponding graph on the right shows the contribution of each individual cytokine to the overall PSI. In this case, Granzyme B is the major driver of the sample's polyfunctionality.
[0188] In the right graph of FIG. 11D, we see that there are several cytokines that drive the polyfunctional strength of the sample. The PSI is primarily composed of inflammatory cytokines IL-6 and MCP-1 and effector cytokines Granzyme B and Perforin. Many other cytokines contribute to a lesser degree to the PSI of the sample.
[0189] FIGS. 10C and 11C show vertical scatterplots of all the single-cell secretions from the specified population. Each orange dot corresponds to a specific cytokine that was secreted at a particular intensity by a single cell. These are background-subtracted intensities; all intensities shown here are above 0, and thus correspond to single-cell secretions. Data points.ltoreq.0, indicating that a single cell did not secrete a specific cytokine, are not shown.
[0190] The units of the intensity levels (y-axis) shown in the graphs are arbitrary, but based on "on-chip" calibration curves ("on chip" may also be referred to as "in situ" with respect to the compositions of the disclosure), these values were converted to approximate pg/mL secretion amounts. The percentage label above each cytokine's vertical scatterplot corresponds to the percentage of single cells in the sample that secreted that cytokine. The larger percentage at the top of the graph indicates how many secretions (as a fraction of all secretions) fell above 1000, to highlight strong secretions (1000 is an arbitrary threshold that may be adjusted and/or made specific to each cytokine).
INCORPORATION BY REFERENCE
[0191] Every document cited herein, including any cross referenced or related patent or application is hereby incorporated herein by reference in its entirety unless expressly excluded or otherwise limited. The citation of any document is not an admission that it is prior art with respect to any invention disclosed or claimed herein or that it alone, or in any combination with any other reference or references, teaches, suggests or discloses any such invention. Further, to the extent that any meaning or definition of a term in this document conflicts with any meaning or definition of the same term in a document incorporated by reference, the meaning or definition assigned to that term in this document shall govern.
Other Embodiments
[0192] While particular embodiments of the disclosure have been illustrated and described, various other changes and modifications can be made without departing from the spirit and scope of the disclosure. The scope of the appended claims includes all such changes and modifications that are within the scope of this disclosure.
* * * * *
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015
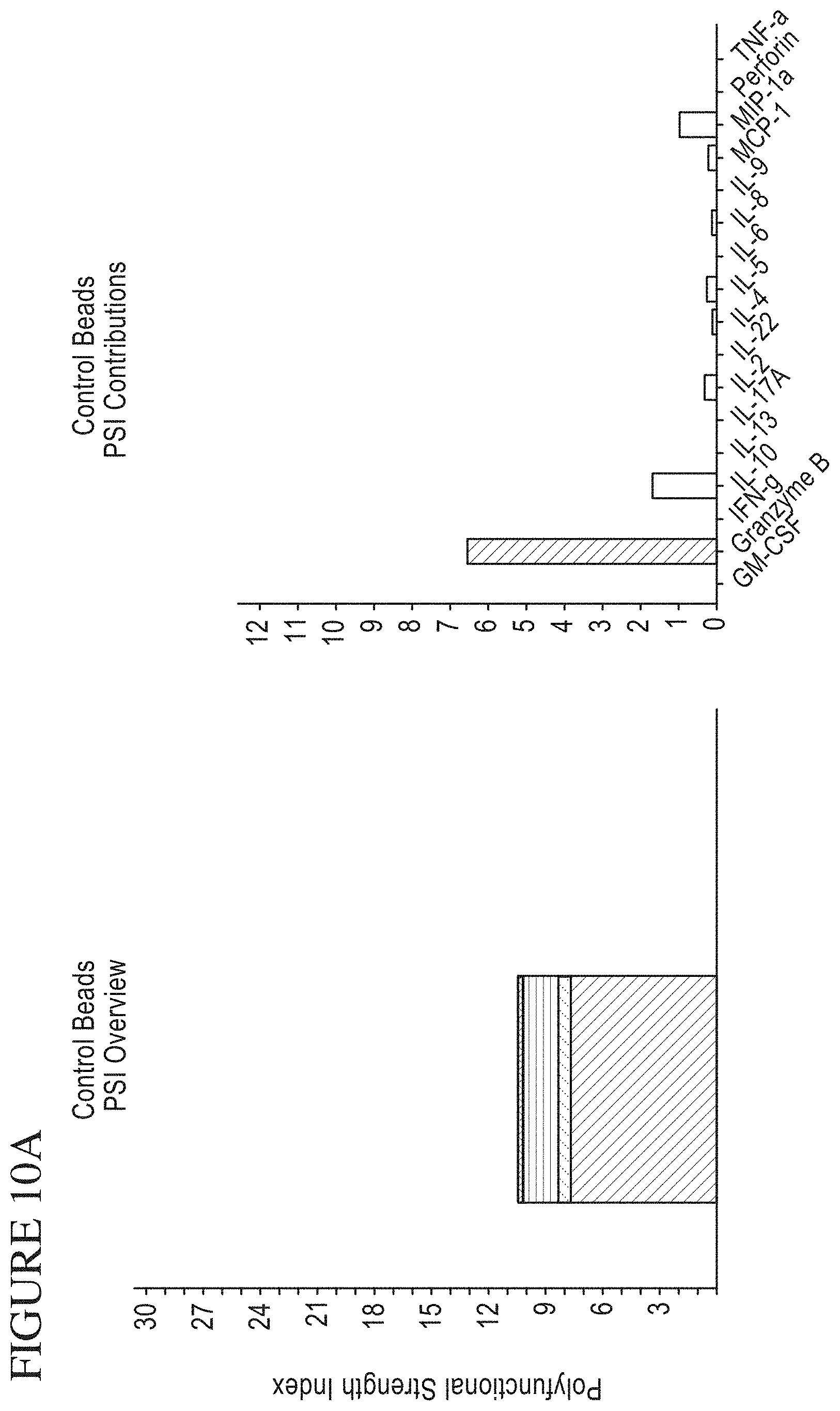
D00016

D00017

D00018
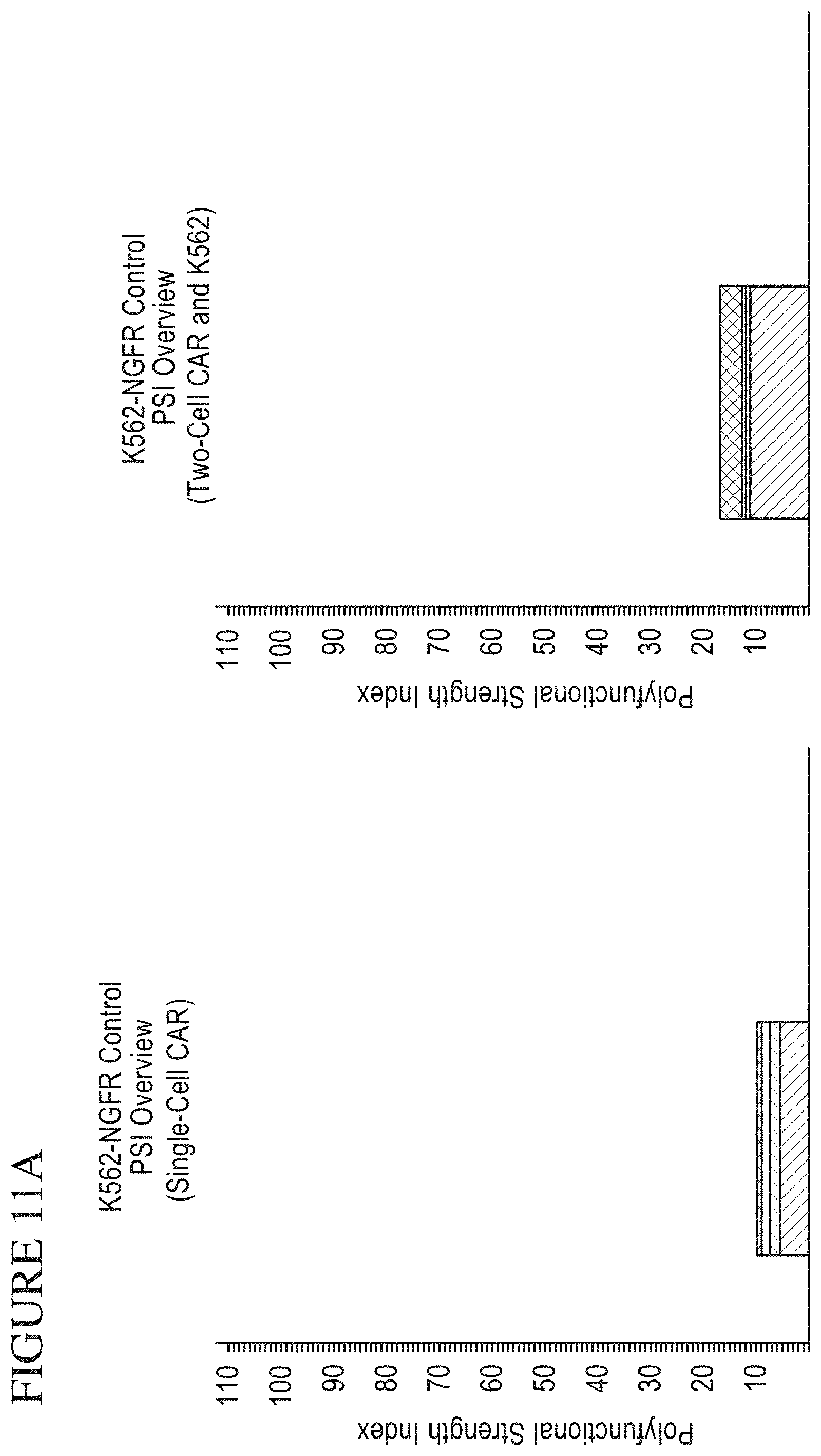
D00019

D00020
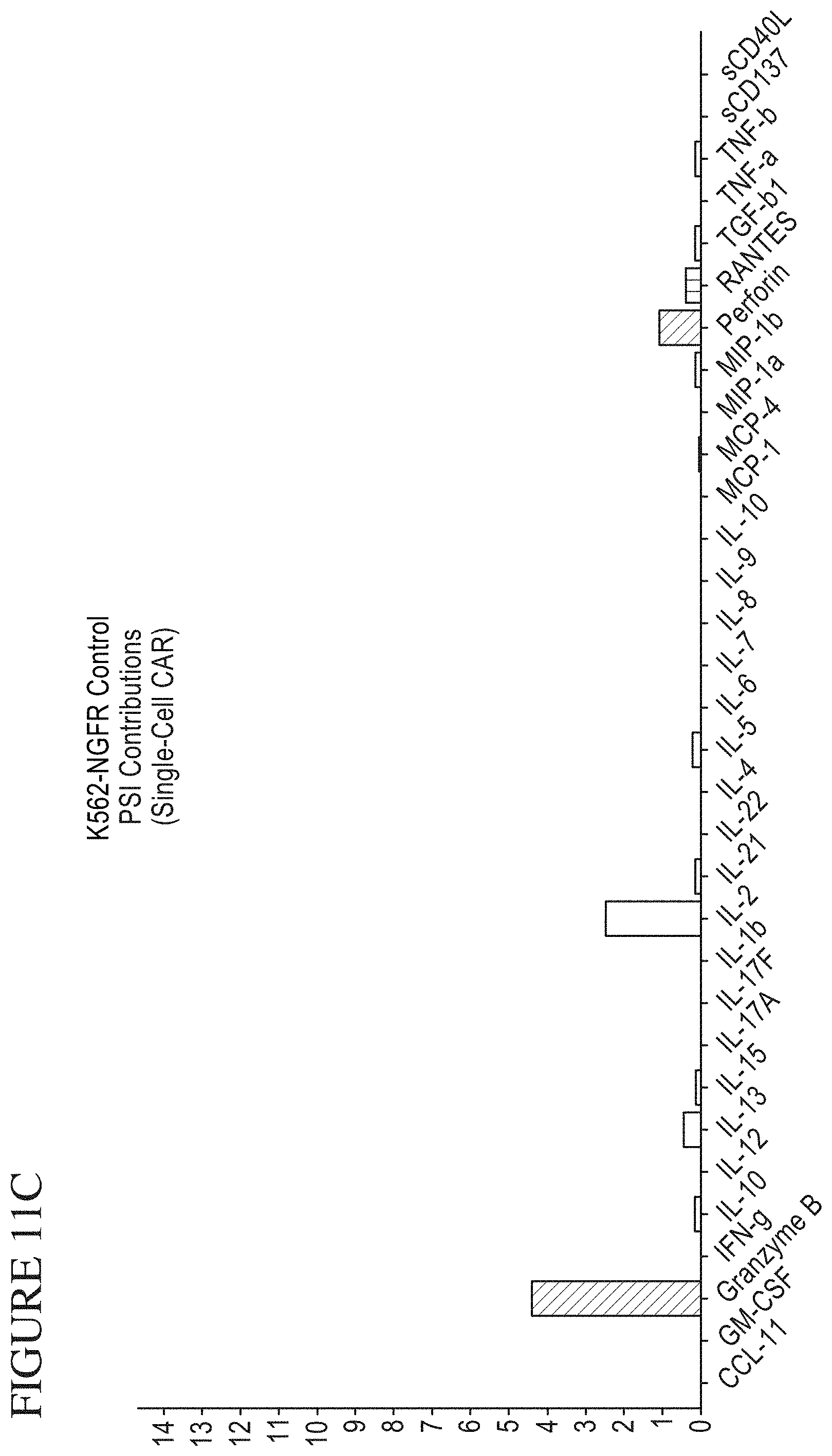
D00021
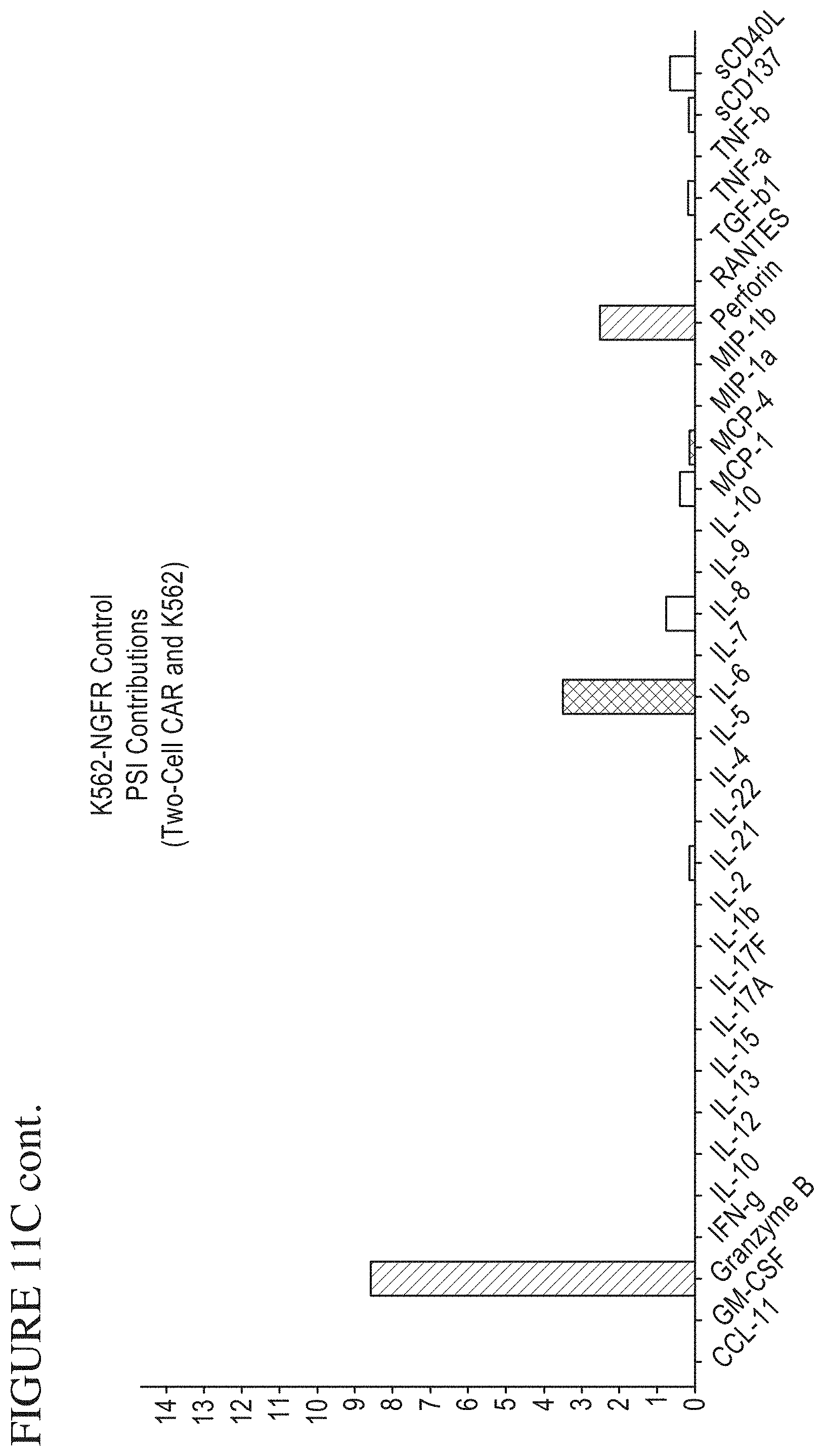
D00022
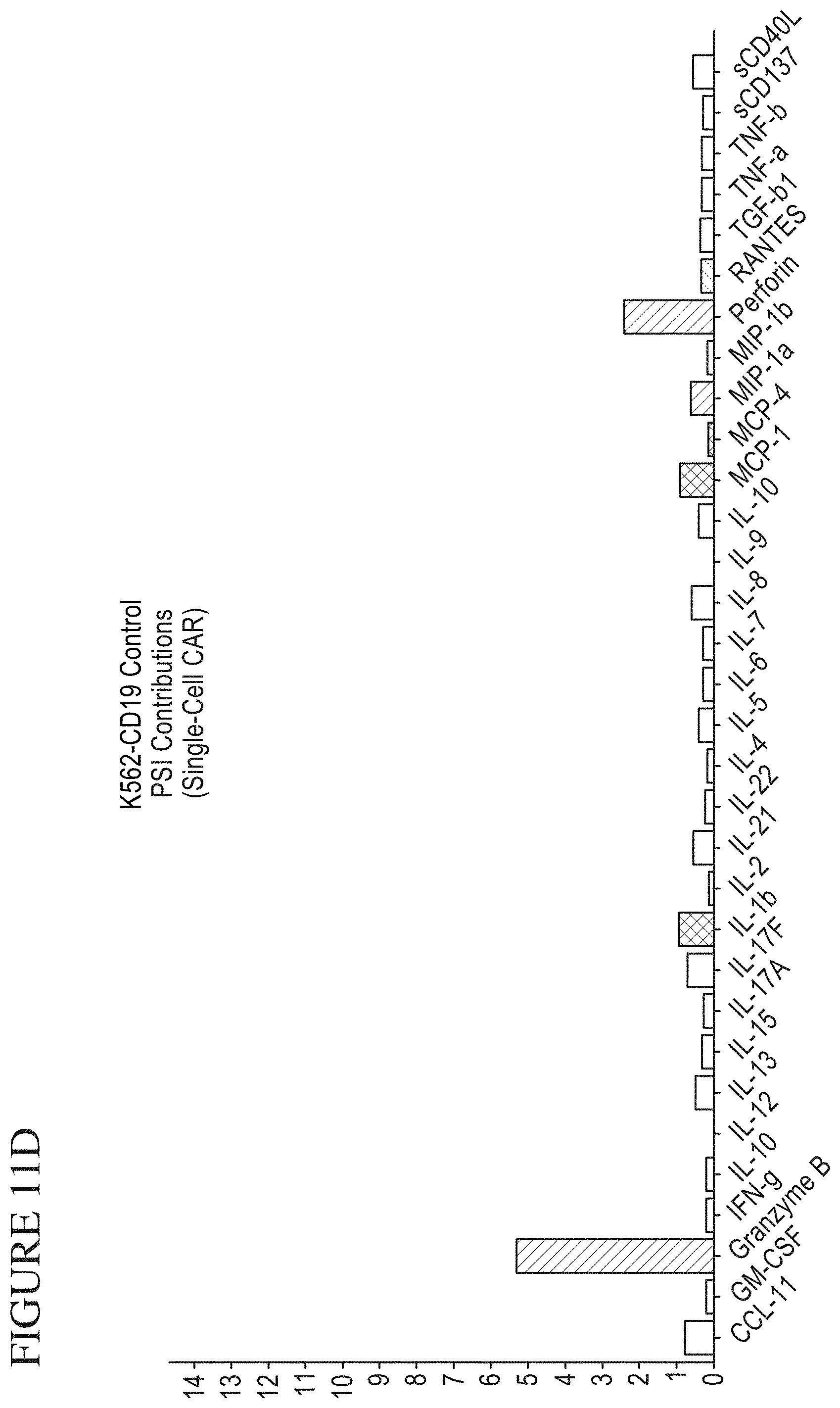
D00023
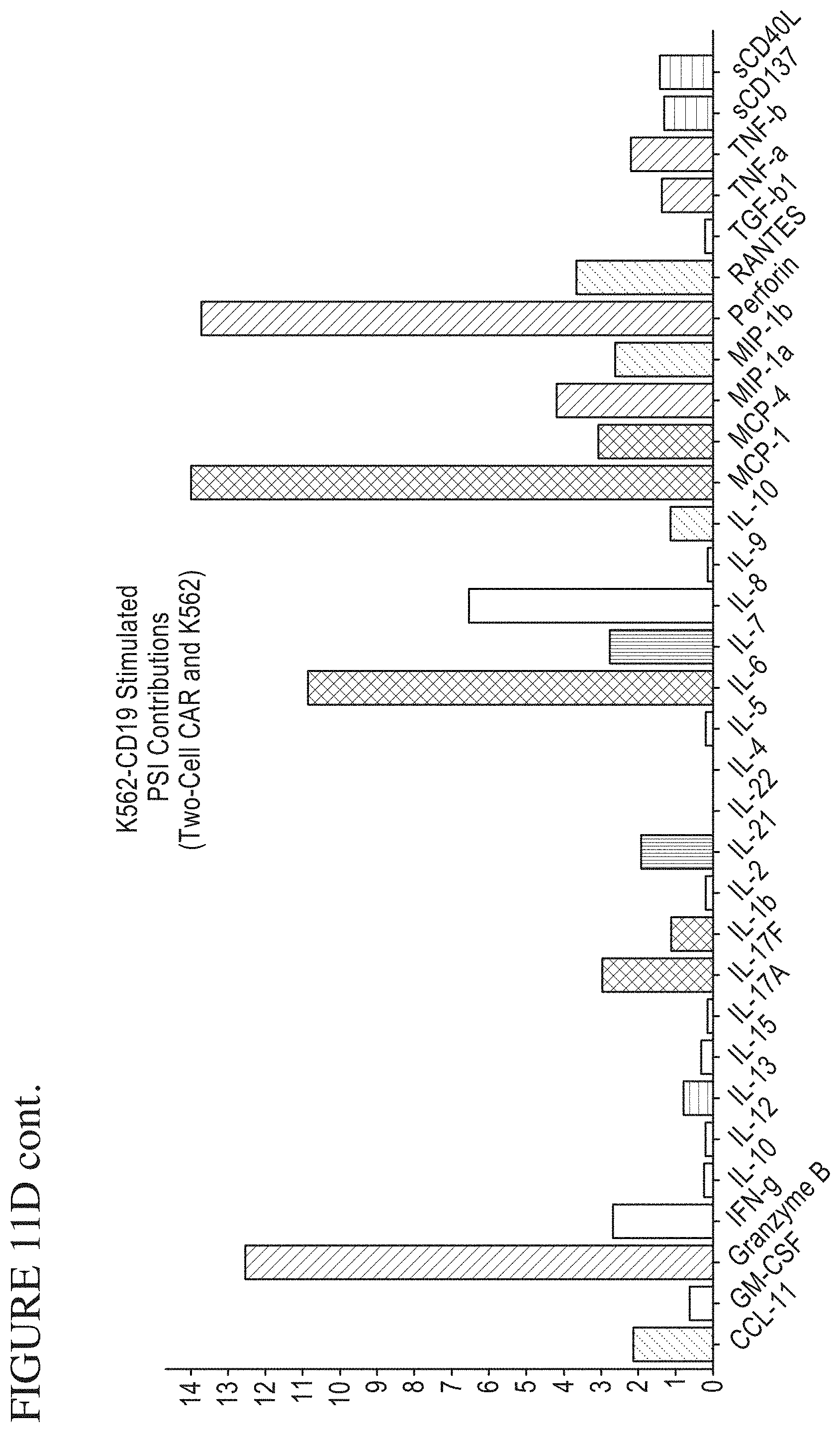
D00024

D00025
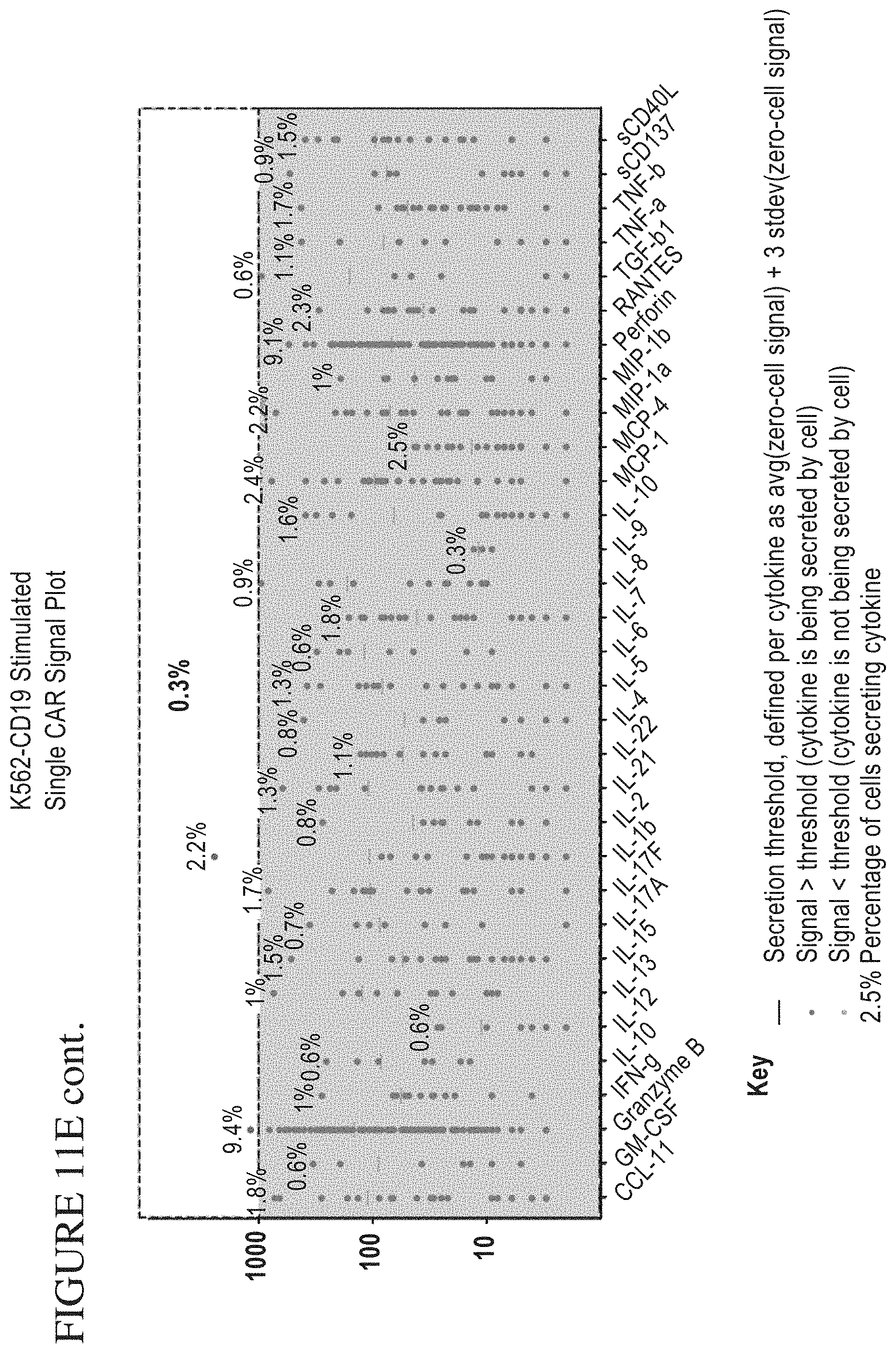
D00026

D00027
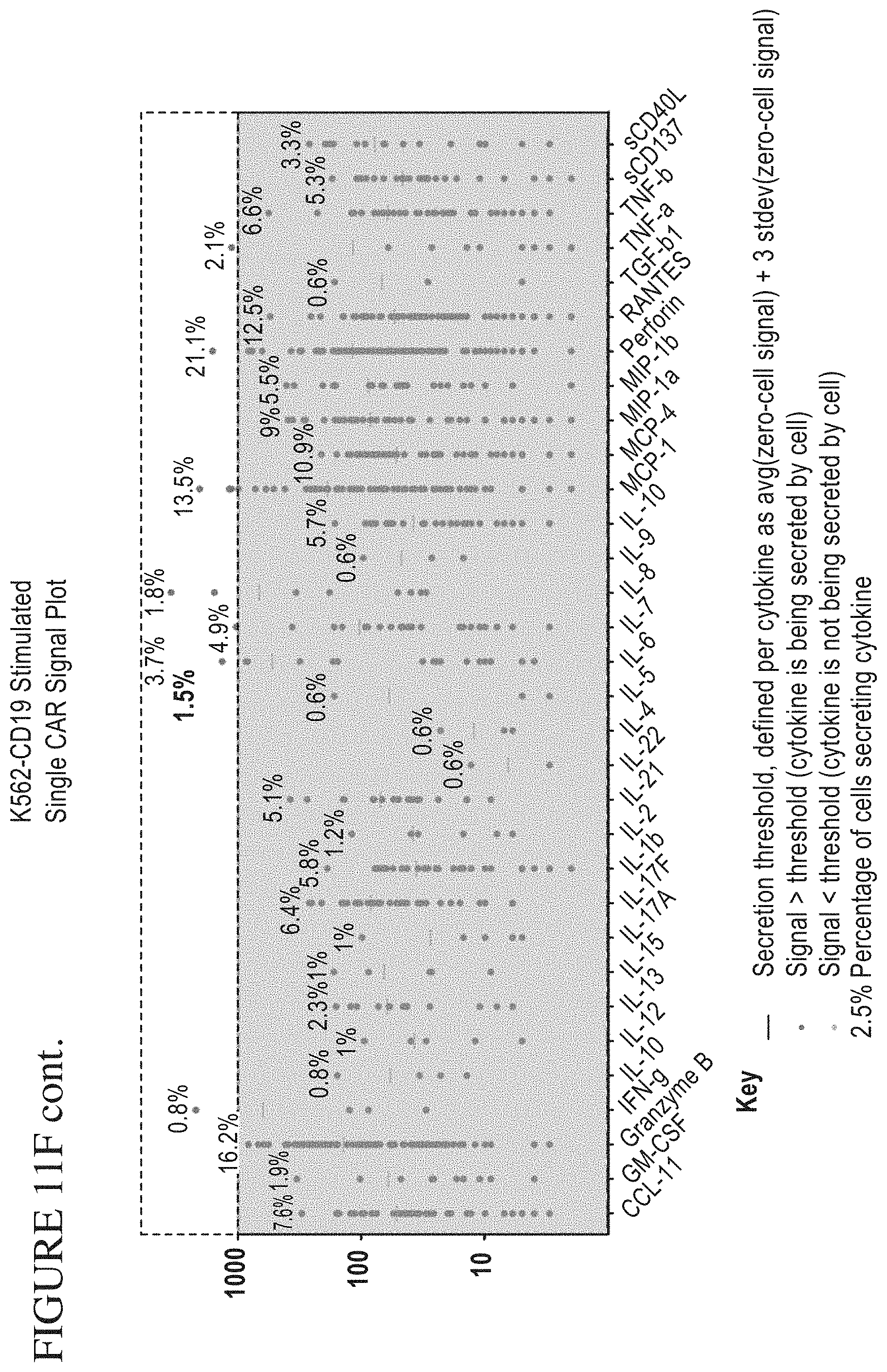
D00028

D00029
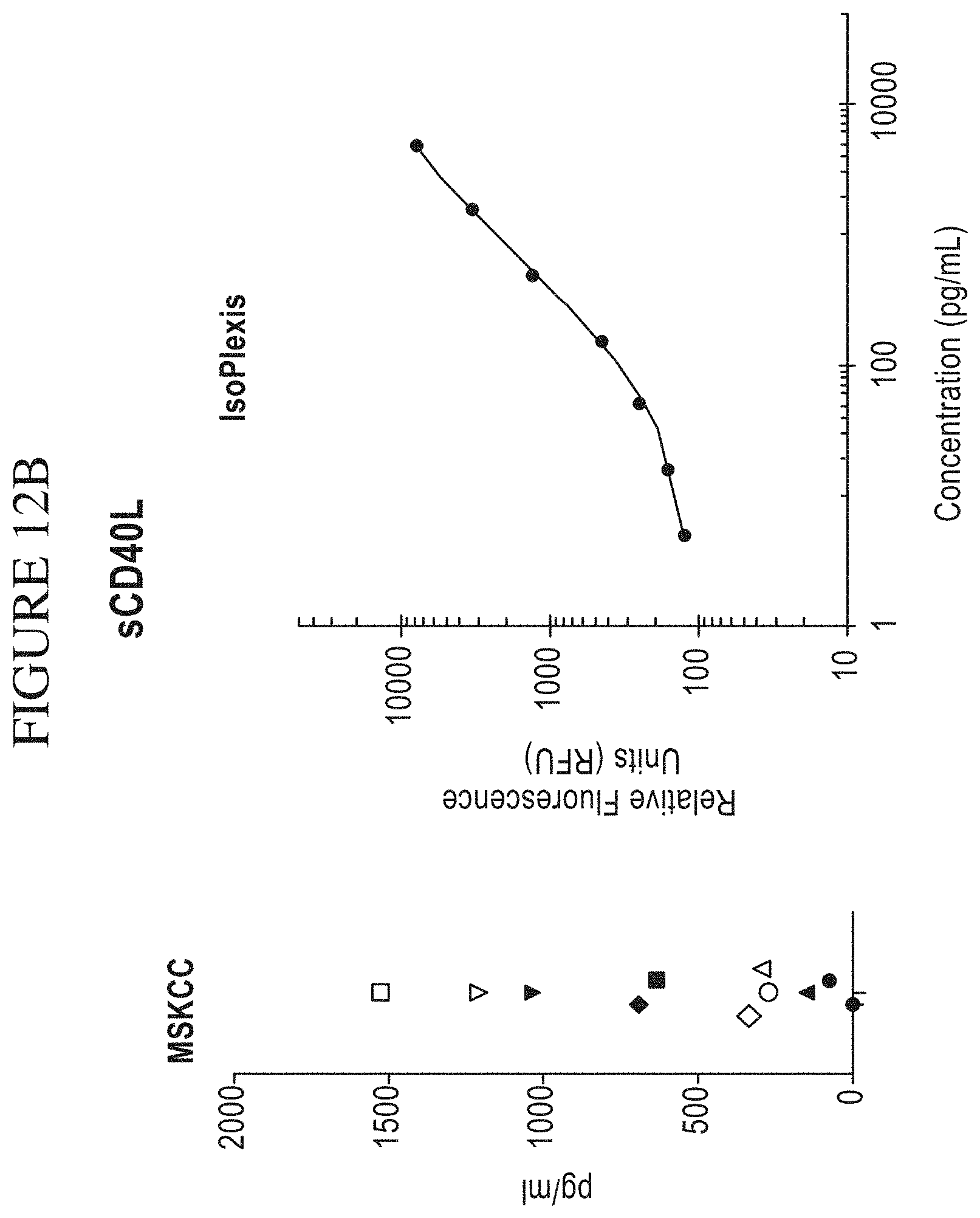
D00030
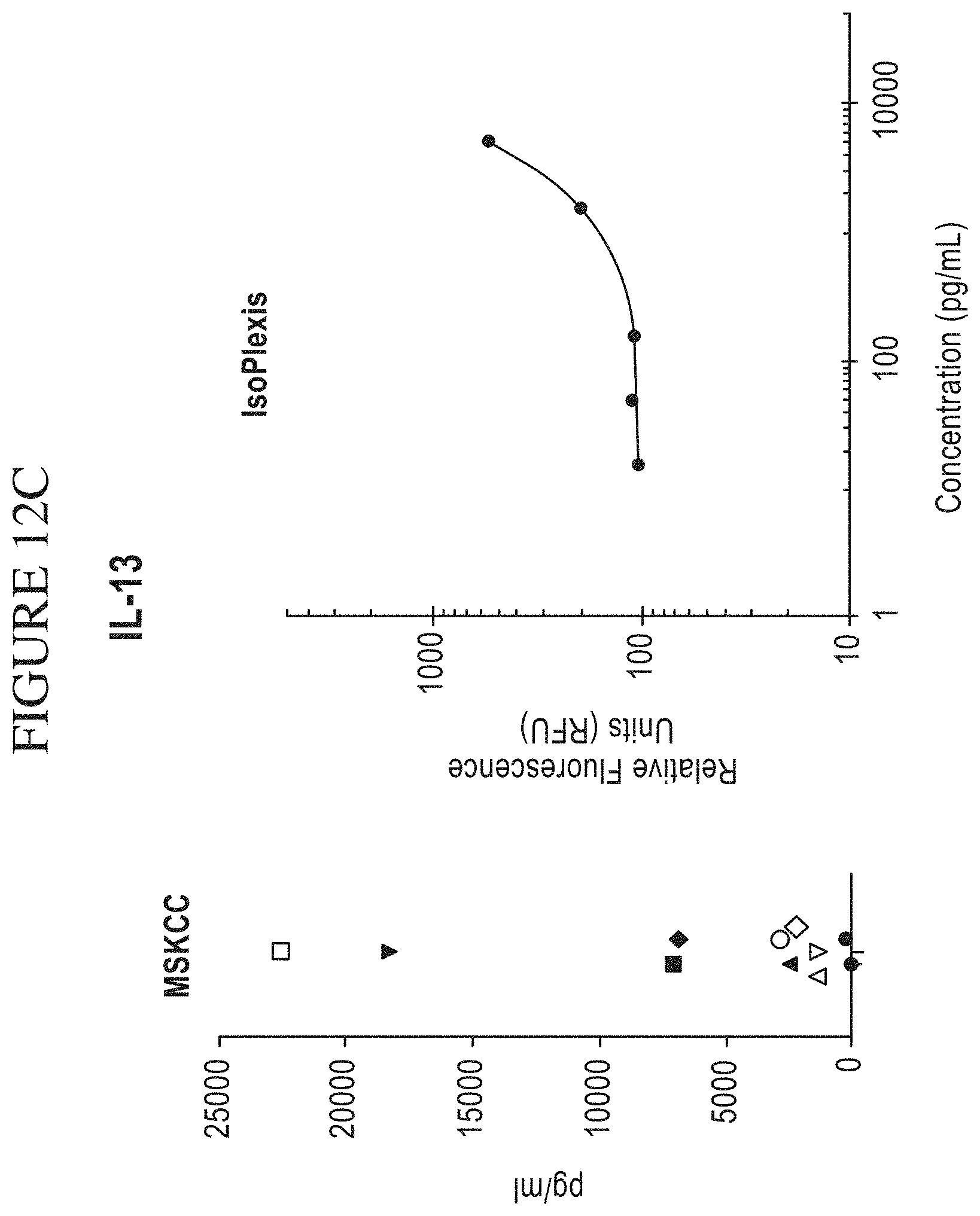
D00031
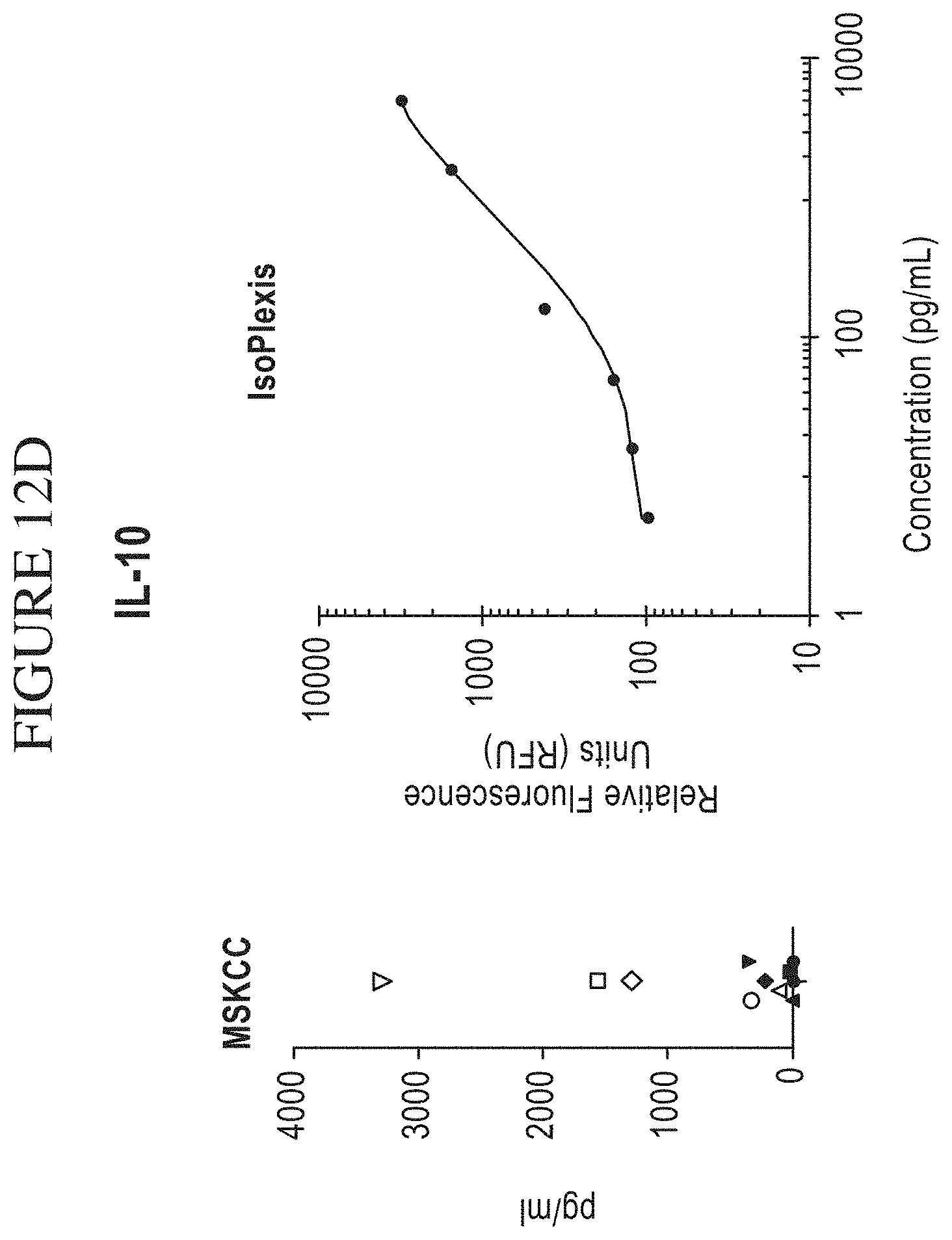
D00032

D00033
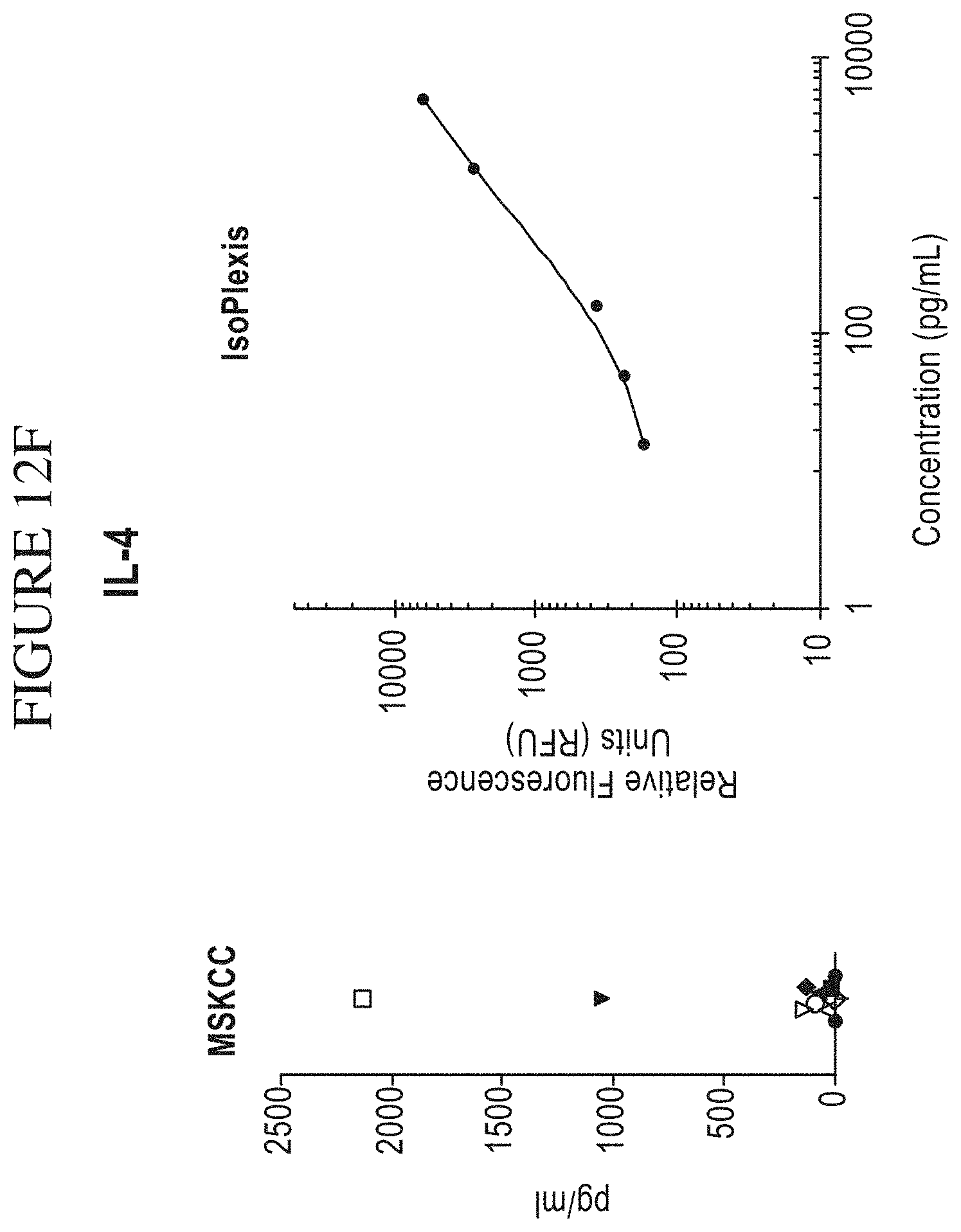
D00034
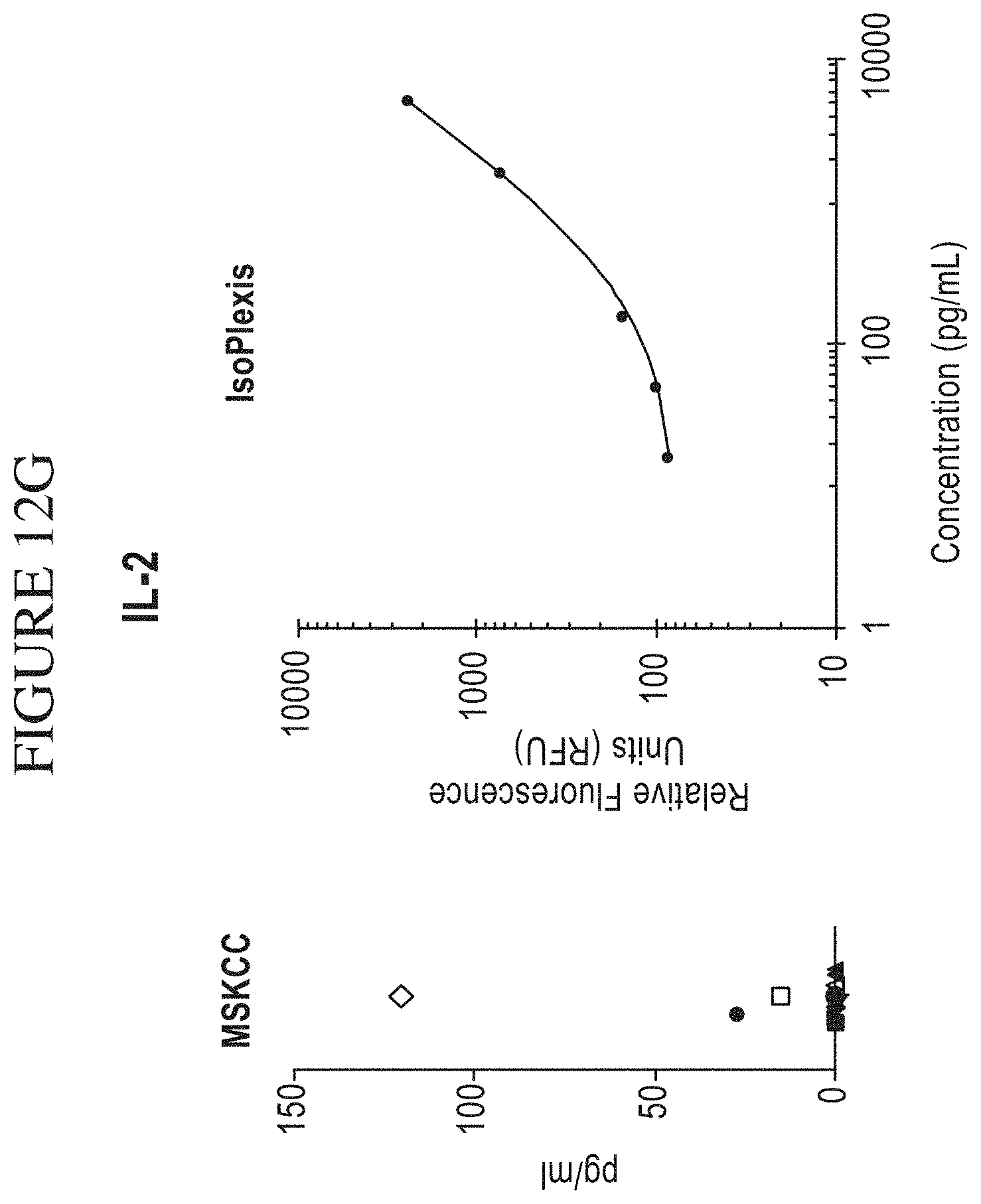
D00035
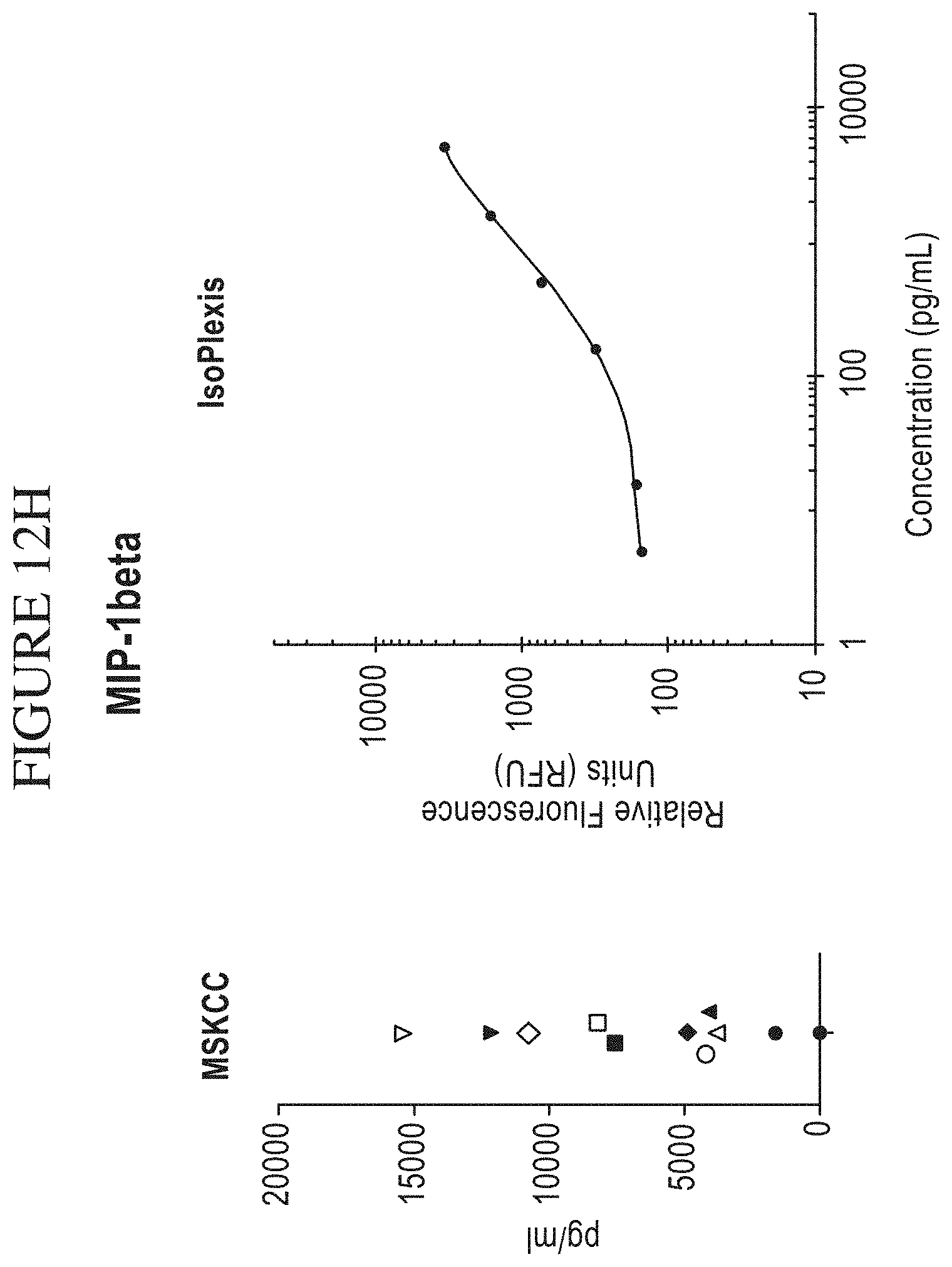
D00036

D00037
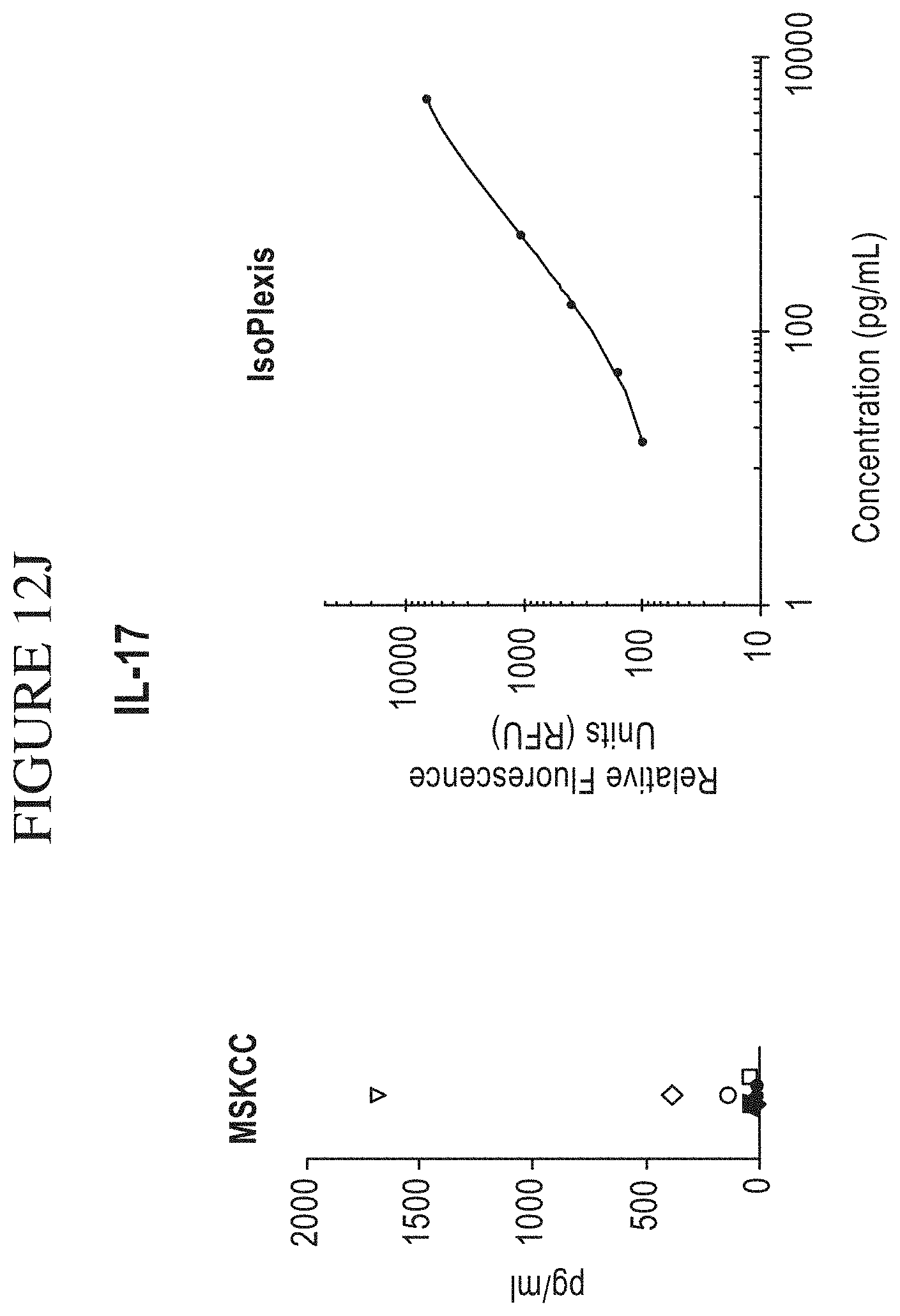
D00038
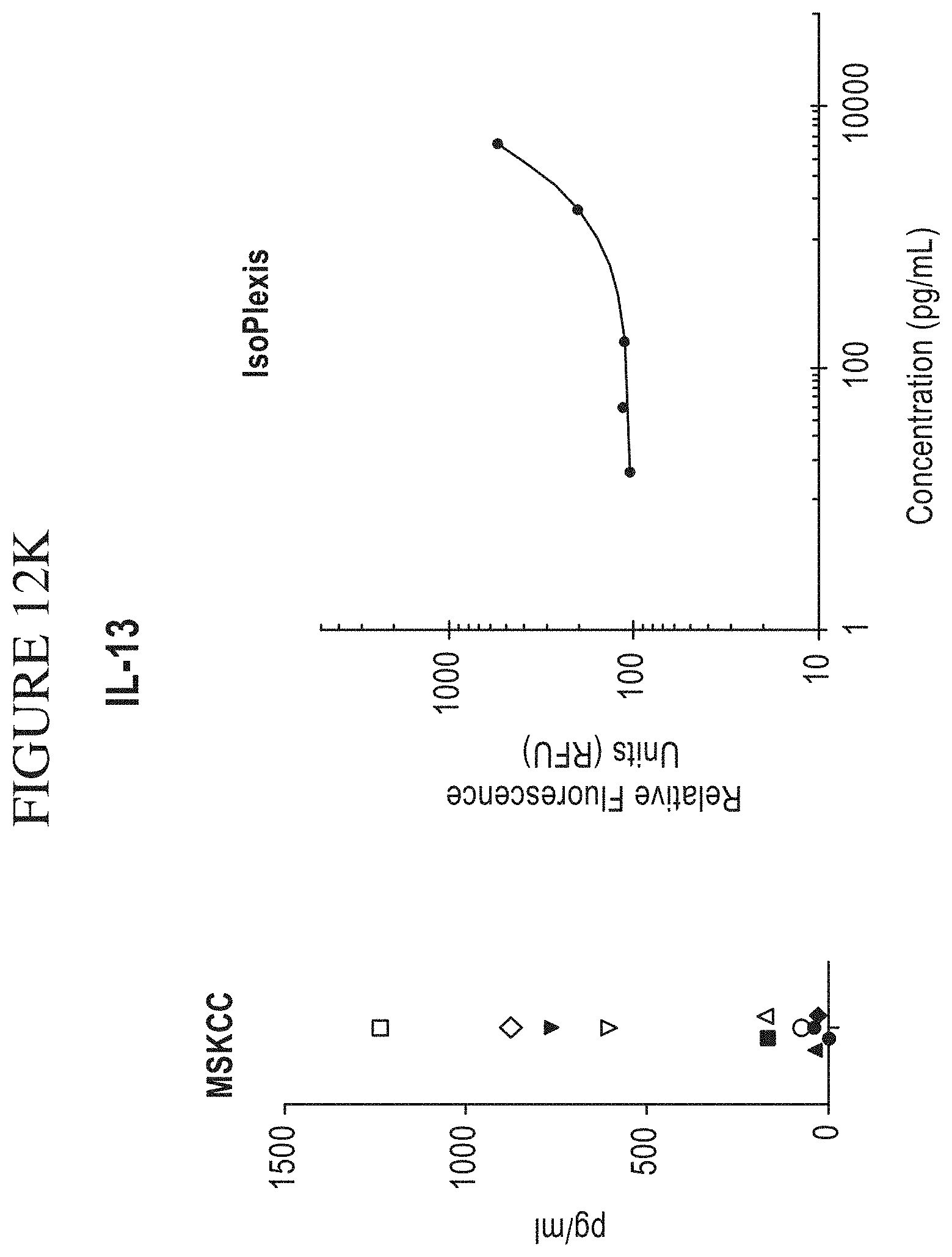
D00039

D00040

D00041
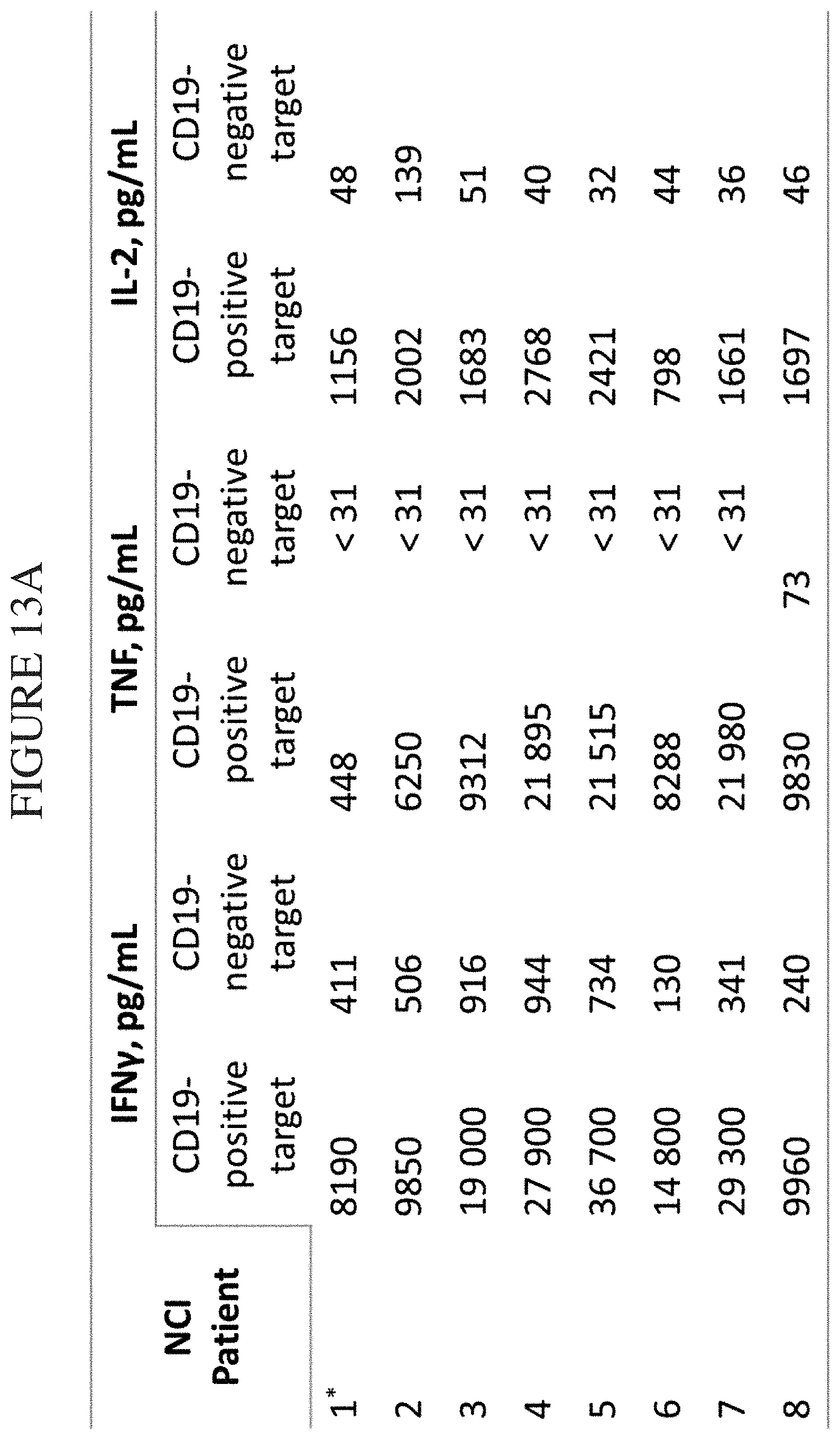
D00042

D00043

D00044
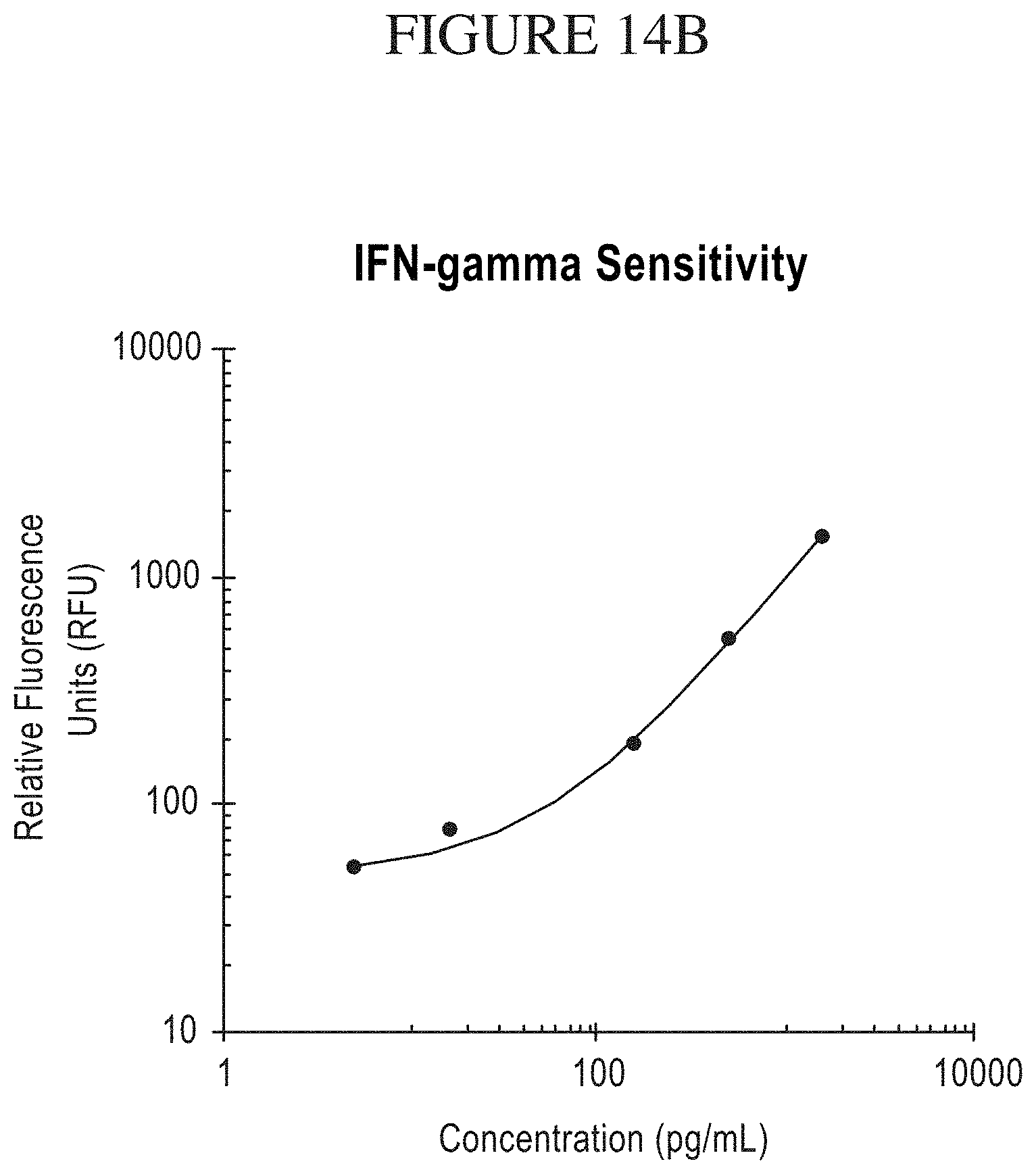
D00045
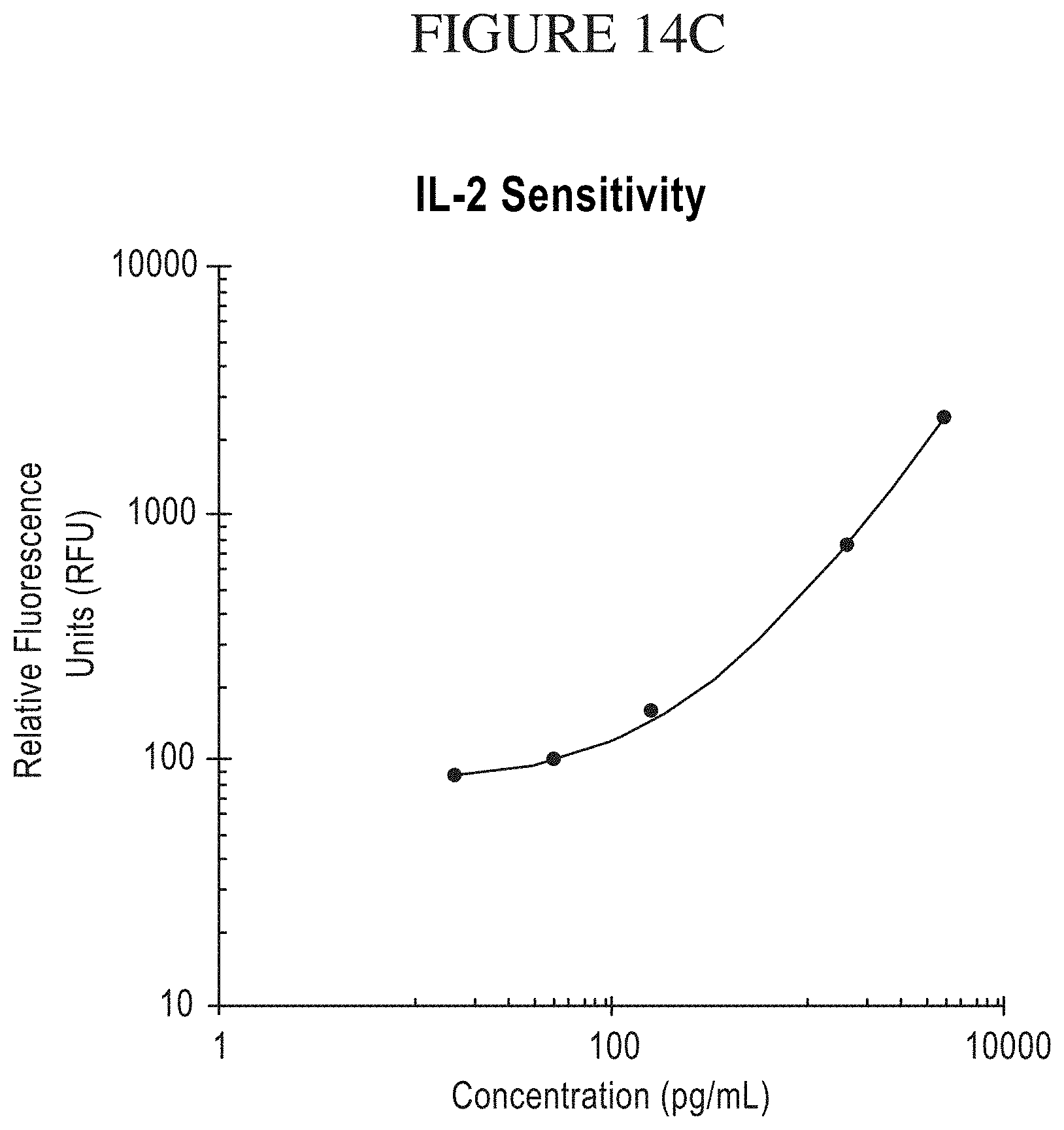
D00046
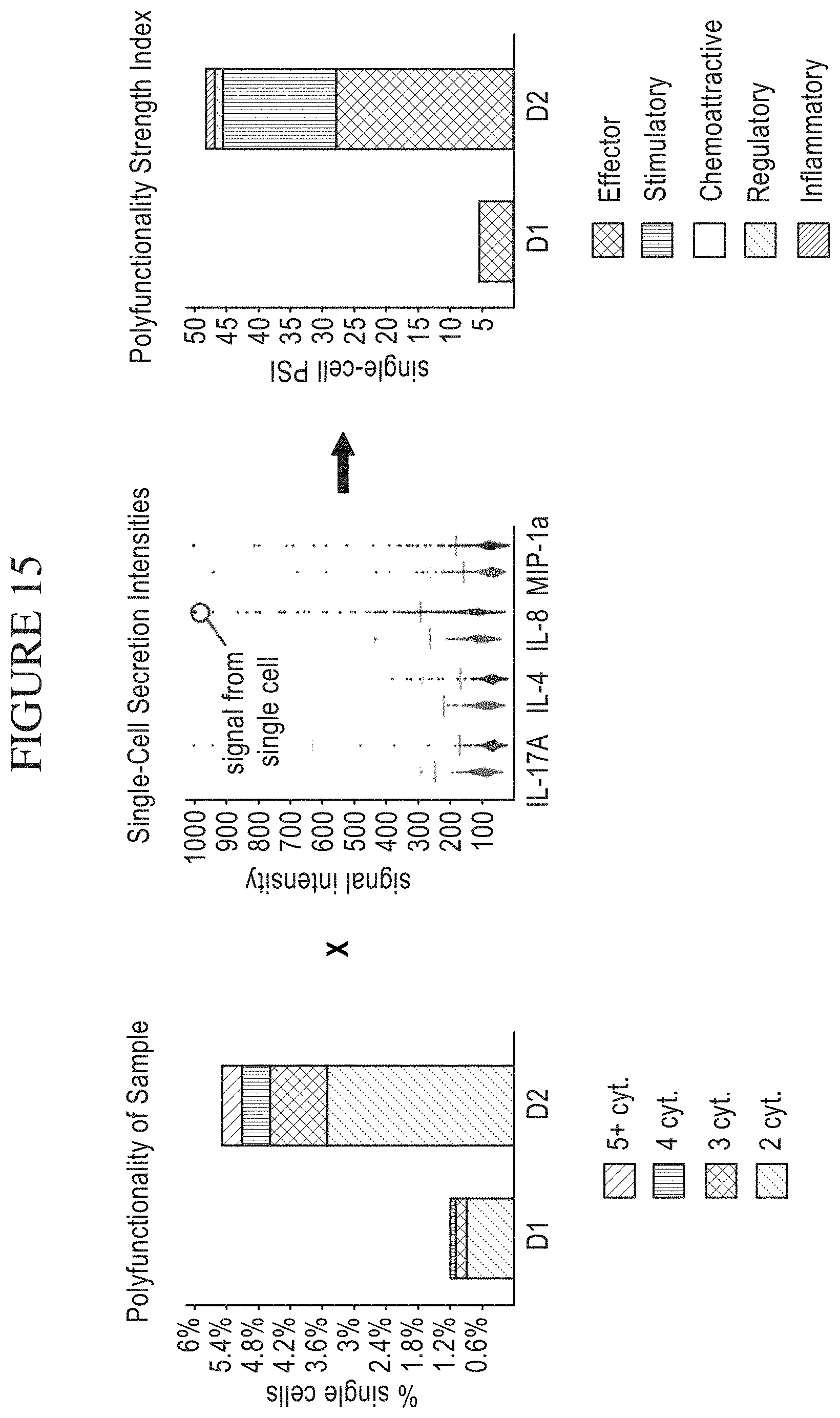
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.