Low Temperature Colouring Method
BURKINSHAW; Stephen Martin
U.S. patent application number 16/618588 was filed with the patent office on 2020-05-21 for low temperature colouring method. The applicant listed for this patent is UNIVERSITY OF LEEDS. Invention is credited to Stephen Martin BURKINSHAW.
| Application Number | 20200157737 16/618588 |
| Document ID | / |
| Family ID | 62563190 |
| Filed Date | 2020-05-21 |











View All Diagrams
| United States Patent Application | 20200157737 |
| Kind Code | A1 |
| BURKINSHAW; Stephen Martin | May 21, 2020 |
LOW TEMPERATURE COLOURING METHOD
Abstract
The present invention relates to a method of colouring polymer substrates at low temperatures. The method comprises subjecting the polymer substrate that is to be coloured to a colouring liquor comprising a solvent in which the colourant has a high solubility and then adding a solvent in which the colourant has a low solubility, typically water.
| Inventors: | BURKINSHAW; Stephen Martin; (Leeds, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 62563190 | ||||||||||
| Appl. No.: | 16/618588 | ||||||||||
| Filed: | May 31, 2018 | ||||||||||
| PCT Filed: | May 31, 2018 | ||||||||||
| PCT NO: | PCT/GB2018/051480 | ||||||||||
| 371 Date: | December 2, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | D06P 3/60 20130101; D06P 3/143 20130101; D06P 3/54 20130101; D06P 3/8238 20130101; D06P 1/0032 20130101; D06P 1/928 20130101; D06P 2001/906 20130101; D06P 3/8233 20130101; D06P 3/045 20130101 |
| International Class: | D06P 1/92 20060101 D06P001/92; D06P 1/00 20060101 D06P001/00; D06P 3/54 20060101 D06P003/54; D06P 3/82 20060101 D06P003/82; D06P 3/14 20060101 D06P003/14; D06P 3/04 20060101 D06P003/04; D06P 3/60 20060101 D06P003/60 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jun 2, 2017 | GB | 1708816.2 |
| Apr 26, 2018 | GB | 1806803.1 |
Claims
1. A method of colouring a polymer substrate, the method comprising: a) subjecting the polymer substrate to a colouring liquor at a temperature T1, T1 being below 100.degree. C., said colouring liquor comprising at least one colourant dissolved in a first solvent system to provide the polymer substrate wetted with the colouring liquor; b) adding a second solvent system to the polymer substrate wetted with the colouring liquor, without raising the temperature above a temperature T2, T2 being below 100.degree. C., to provide the coloured polymer substrate wetted with a mixture of the first solvent system and the second solvent system; and c) separating the coloured polymer substrate from the mixture of the first and second solvent systems and any remaining colourant; wherein the colourant or colourant dye is more soluble in the first solvent system than in the second solvent system.
2. (canceled)
3. (canceled)
4. A method of claim 1, wherein the second solvent system is water.
5. A method of claim 1, wherein the first solvent system and the second solvent system are miscible.
6. (canceled)
7. A method of claim 1, wherein the first solvent system is a polar organic solvent or a mixture of polar organic solvents.
8. A method of claim 7, wherein the first solvent system comprises acetone.
9. A method of claim 7, wherein the first solvent system comprises a solvent selected from glycerol, ethylene glycol diacetate (EGD), triethylene glycol monomethyl (TGM), dipropylene glycol methyl ether (DME) and 1-methoxy-2-propanol.
10. A method of claim 1, wherein the polymer substrate is a fibre substrate.
11. A method of claim 10, wherein the fibre substrate comprises fibres selected from: a polyester, nylon, a polyurethane, wool, silk, cotton or a mixture thereof.
12. A method of claim 11, wherein the fibre substrate comprises polyester fibres.
13. A method of claim 12, wherein the fibre substrate comprises polyester fibres and at least one fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane.
14. A method of claim 13, wherein the fibre substrate comprises a material that is a blend of polyester fibres and at least one fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane.
15. A method of claim 14, wherein the fibre substrate comprises a first material that comprises polyester fibres and a second material that comprises at least one fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane.
16. A method of claim 15, wherein the fibre substrate is a whole garment.
17. A method of claim 1, wherein the ratio of the total volume of the first solvent system to the total volume of the second solvent system is in the range 1:2 to 1:20.
18. A method of claim 16, wherein the ratio of the total volume of the first solvent system to the total volume of the second solvent system is in the range 30:70 to 10:90.
19. A method of claim 1, wherein the weight ratio of the polymer substrate to the first solvent system is in the range from 3:1 to 1:3.
20. A method of claim 1, wherein the at least one colourant is at least one dye.
21. A method of claim 20, wherein the at least one dye is a disperse dye.
22. A method of claim 1, wherein the method comprises dissolving the at least one colourant in the first solvent system to form the colouring liquor
23. A method of claim 1, wherein the colouring liquor does not comprise a dispersing agent.
24. (canceled)
Description
[0001] The present invention relates to a method of colouring polymer substrates at low temperatures. The method comprises subjecting the polymer substrate that is to be coloured to a colouring liquor comprising a solvent in which the colourant has a high solubility and then adding a solvent in which the colourant has a low solubility, typically water. The method is particularly useful for dyeing polyester fibres and polyester fibre blends with disperse dyes.
BACKGROUND
[0002] A large proportion of the textile products produced in the world today comprise polyesters. In particular poly(ethylene terephthalate) (PES) accounted for .about.58.5% (53.1.times.10.sup.6 T) of the 90.6.times.10.sup.6 T world textile fibre demand in 2015. The outstanding success and enduring popularity of PES fibres can be attributed to their generally excellent textile characteristics and high chemical resistance, coupled with the ability of polyester fibre to be manufactured in virtually any physical form as required for different applications, which include blending with other types of fibre. In the latter context, a chief use of polyester fibres is in combination with cotton fibres. In such polycotton blend materials, the cotton component provides comfort, absorbency, etc. whilst the polyester constituent imparts strength and resilience as well as stain-resistance.
[0003] Polyester fibres are dyed exclusively using disperse dyes, which furnish a wide shade gamut and display generally very good fastness properties on polyester. Disperse dyes belong to several chemical classes, predominantly anthraquininoid, (AQ), and azo, as exemplified by C.I. Disperse Red 60 and C.I. Disperse Blue 165.
##STR00001##
[0004] Disperse dyes typically have low aqueous solubility. Solubility increases, however, markedly with increasing temperature, as exemplified by C.I. Disperse Red 121: 3.3 0.00033 gL.sup.-1 at 25.degree. C.; 0.0088 gL.sup.-1 at 130.degree. C. This is of significance for their application to polyester fibre under High Temperature (HT) dyeing conditions.
[0005] The solubility of disperse dyes is greatly increased in the presence of commercial dispersing agents, a feature that forms the basis of both the finishing process that is used to prepare commercial forms of the dye and the application method that is employed in their application to polyester fibres under aqueous immersion processes. Commercial disperse dyes typically comprise up to 60% by mass dispersing agent, adding considerably to the cost of the dyes. Dispersing agents (examples include lignin sulfonates or formaldehyde polycondensates or arylsulfonic acids) are not particularly environmentally friendly. The removal and disposal of such materials from the dyed textile fibre at the end of dyeing adds considerably to the cost, energy efficiency and environmental risk of commercial dyeing.
[0006] Owing to the very low rate of diffusion of the dyes within PES fibres at temperatures up to the commercial boil (i.e. 98.degree. C.), commercially acceptable rates of dyeing with disperse dyes are most commonly achieved by the use of elevated temperatures in the region of 130-140.degree. C. in a process commonly referred to as High Temperature (HT) dyeing. Currently, the vast majority of polyester fibre is dyed using such immersion processes at 130-140.degree. C. Because such high temperatures are required for dyeing, the dyeing process is energy-intensive and the machines that are utilised for dyeing must be able to operate at pressures >1 atmosphere; consequently, the machines are typically very expensive.
[0007] PES fibres contain small amounts of oligomer, principally the cyclic trimer tris(ethylene terephthalate), as well as smaller amounts of other oligomeric compounds. Such compounds migrate to the fibre surface during HT dyeing and can deposit on the surfaces of both the fibre and machine during cooling, which can reduce the visual depth of shade brilliance of dyeings; the removal of the compounds from the dyeing machine constitutes an additional problem in immersion dyeing. The removal of the oligomers, in addition to the excess dye and the dispersing agents, is routinely achieved using a reduction clearing process in which the dyed material is treated with Na.sub.2S.sub.2O.sub.4 and a non-ionic surfactant, typically with heating. This process adds additional time to the process as well as additional materials and energy. It also generates environmentally unacceptable efluents, including Na.sub.2S.sub.2O.sub.4 and, in the case of azo-disperse dyes, aromatic amines biproducts.
[0008] A further limitation of HT dyeing methods is that many fabrics, and particularly natural fabrics (e.g. wool, silk) and more sensitive manmade fabrics (e.g. polyurethanes, for example, Lycra.RTM.) are not stable under the high temperature conditions. Where polyesters are combined (either woven together as a blend or joined together as parts of a garment) with these materials, other less effective dyeing methods must be used (which can lead to discernible differential colour strengths for the individual fabrics in the blend) or pre-dyed fabric must be blended, which is a complex and costly process. This means that for blends or garments that are formed of more than one different material, the fibres have to be dyed separately before being woven together or joined together.
[0009] When PES was first introduced in the 1950s, the preferred dyeing method used was carrier dyeing, in which a carrier, typically a low molar mass, solid organic compound, such as o-phenyl phenol, is included in the aqueous dyeing liquor. This carrier facilitates the dyeing of the fabric and allows the use of lower temperatures than HT dyeing, typically 98.degree. C. However, the carriers often have detectable smells, they often impair the light fastness of the dyed material and they also pose environmental concerns and, as a result, HT dyeing processes predominate commercially nowadays and the use of carrier dyeing has steadily declined.
[0010] Disperse dyes are much more soluble in organic solvents than they are in water. However, organic solvents are ineffective for disperse dyeing because the dye has a greater affinity for the solvent than it does for the fibres. Consequently, when disperse dyes are applied from organic solvents low dye uptake onto the fibres is achieved so that the depth of shade that can be accessed using high temperature aqueous dyeing techniques cannot be replicated using organic solvent based dyeing liquors.
[0011] It is not just in the dyeing of polyesters where improvements in dyeing can be made. Methodologies for dyeing wool, silk and polyamide fibres are often carried out at high temperatures and usually require specific values of pH for their successful application, leading to the use of additional chemicals to control the pH. The methods can also be time consuming. For example, dyeing with vat dyes is a complicated, time-consuming, multi-stage process that involves several pH changes and requires the use of environmentally questionable strong reducing agents.
[0012] In addition, dyeing auxiliaries are commonly used to assist aqueous immersion dyeing processes. The assistance provided by a given dyeing auxiliary will typically relate to a specific aspect of dyeing, such as wetting, dye levelling, fibre protection, etc. Thus, many different types of dyeing auxiliary are commonly used in aqueous dye application, such as dispersants, sequestrants, lubricants, etc.). For example, dyeing processes for polyester fibres typically utilise dispersing agents and surfactants which are added to the dyebath to aid dye dispersion and levelling, as discussed above. Also, the pH at which dyeing is performed is commonly adjusted so as to be within a defined range, such as slightly acidic (pH.about.4.5-6.0), although selected disperse dyes are suitable for application at high pH (.about.pH 9.5). Because aqueous dyebaths routinely contain many auxiliaries often in large amounts, the wastewater generated during immersion dyeing is likely to contain a wide variety of auxiliary chemicals; indeed, many of the auxiliaries that are utilised in immersion dyeing (such as s dispersing agents that are used in polyester dyeing) are intended to be removed from the dyed material at the end of dyeing, meaning that such chemicals will be present in the wastewater that ensues from dyeing processes. Although many strategies have been explored for treating dyeing effluent that contains residual dyeing auxiliaries, no single treatment method is effective for all auxiliaries or types of dye/fibre system. An additional advantage of certain embodiments of the current invention is that it is possible to reduce the number and amount of dyeing auxiliaries that are used in dyeing processes, thus offering savings in chemical costs as well as environmental advantages.
[0013] Although, the methods of the invention were developed for the dyeing of fibre substrates, the inventors have found that they can be applied more generally to other polymer substrates.
BRIEF SUMMARY OF THE DISCLOSURE
[0014] In a first aspect of the invention is provided a method of colouring a polymer substrate, the method comprising: [0015] a) subjecting the polymer substrate to a colouring liquor at a temperature T1, T1 being below 100.degree. C., said colouring liquor comprising at least one colourant dissolved in a first solvent system to provide the polymer substrate wetted with the colouring liquor; [0016] b) adding a second solvent system to the polymer substrate wetted with the colouring liquor, without raising the temperature above a temperature T2, T2 being below 100.degree. C., to provide the dyed polymer substrate wetted with a mixture of the first solvent system and the second solvent system; and [0017] c) separating the dyed polymer substrate from the mixture of the first and second solvent systems and any remaining colourant; wherein the colourant or each colourant is more soluble in the first solvent system than in the second solvent system.
[0018] The polymer substrate may be a fibre substrate. The colourant may be a dye. The inventors have found that greater depth of colour can be achieved using the same quantity of colourant when using the above method than can be achieved using HT disperse dyeing methods. This greater depth of colour is achieved at a lower temperature than used in conventional HT methods and it is not necessary to use dispersing agents. Thus, the method of the first aspect is likely to be less energy intensive, less costly and pose lower risk to the environment than known HT methods. Also being a low temperature process it can be performed on or in the presence of sensitive natural and man-made fibres. Without wishing to be bound by theory, it is believed that the addition of the second solvent system to the solution of the colourant in the first solvent system lowers the solubility of the colourant in the solvents. This in turn generates a molecular dispersion from which uptake of the colourant into the fibre is possible and indeed that is more favoured than with the dispersions formed in HT dyeing methods.
[0019] The inventors also have found that when applied to the acid dyeing of certain substrates, the methods of the invention provide effective dyeing without the use of added dyeing auxiliaries, at lower temperatures and more quickly than traditional methods.
[0020] The lower temperatures used allow for the simultaneous colouring of more than one polymer type at a time. The methods apply not just to disperse dyes but also to other types of colourant, including acid dyes, direct dyes, reactive dyes and vat dyes. The inventors have shown that it is possible to dye more than one polymer (e.g. fabric) type at a time using the methods of the invention, with different colourant being simultaneously used to colour different polymers (e.g. fabrics).
[0021] In a second aspect of the invention is provided a polymer (e.g. fibre) substrate obtainable (e.g. obtained) according to the method of the first aspect.
The Method
[0022] It may be that the total amount of the second solvent system is added as a single portion to the polymer (e.g. fibre) substrate wetted with the colouring liquor. It may be that it is added continuously over a predetermined period of time. It may be that it is added portionwise. Thus, it may be that the total amount of the second solvent system is added in a predetermined number of equal sized portions, a predetermined period of time apart. The inventors have found that the portionwise addition of the second solvent system provides the most effective colouring. Without wishing to be bound by theory, it is believed that the portionwise addition provides a more controlled formation of the molecular dispersion of the colourant (e.g. dye). In certain embodiments, the inventors have observed 100% uptake of the colourant into the substrate, leaving no colourant left in the resultant mixture of the first solvent system and the second solvent system.
[0023] The second solvent system is added to cause the colourant to precipitate out of the colouring liquor. Typically, where the polymer substrate is a fibre substrate, this precipitation will occur within the fibre substrate
[0024] The number of equal sized portions of the second solvent system may be in the range from 2 to 10, e.g. in the range from 3 to 6.
[0025] The dyed polymer (e.g. fibre) substrate wetted with a mixture of the first solvent system and the second solvent system may be held at a temperature T3, T3 being below 100.degree. C., for a predetermined period of time after the addition of the second solvent system
[0026] Typically, the coloured (e.g. dyed) polymer (e.g. fibre) substrate wetted with a mixture of the first solvent system and the second solvent system will be cooled or allowed to cool to a temperature T4 before the coloured (e.g. dyed) polymer (e.g. fibre) substrate is separated from the mixture of the first and second solvent systems and any remaining dye.
[0027] It may be that the total colouring (e.g. dyeing) time is less than 3 hours. It may be that the total colouring (e.g. dyeing) time is less than 90 minutes. It may be that the total colouring (e.g. dyeing) time is less than 45 minutes. It may be that the total colouring (e.g. dyeing) time is greater than 20 minutes. The total colouring (e.g. dyeing) time is the period of time from the first addition of the second solvent system to the cooling of the dyed polymer (e.g. fibre) substrate wetted with a mixture of the first solvent system and the second solvent system.
[0028] It may be that the step of subjecting the polymer (e.g. fibre) substrate to a colouring liquor involves spraying the colouring liquor onto the substrate (e.g. fibre) substrate. It may be that the step of subjecting the polymer (e.g. fibre) substrate to a colouring liquor involves placing the substrate into the colouring liquor.
[0029] It may be that the step of adding the second solvent system to the polymer (e.g. fibre) substrate wetted with the colouring liquor involves spraying the second solvent system onto the polymer (e.g. fibre) substrate wetted with the colouring liquor. It may be that the step of adding the second solvent system to the polymer (e.g. fibre) substrate wetted with the colouring liquor involves adding the second solvent system into a colouring liquor in which the substrate is located.
[0030] The method may comprise the step of dissolving the colourant (e.g. dye) in the first solvent system to form the colouring liquor.
[0031] It may be that the colouring liquor does not comprise a dispersing agent. Typical dispersing agents include anionic, polyelectrolyte, compounds (and mixtures thereof), such as lignin sulfonates or formaldehyde polycondensates of arylsulfonic acids (e.g. disodium methylenebisnaphthalene sulfonate, sodium oleyl-p-anisidinesulfonate). It may be that the dyeing liquor does not comprise a carrier. Typical carriers include, for example, o-dichlorobenzene, 1,2,4-trichlorobenzene, dimethyl phthalate, diallyl phthalate, o-phenyl phenol, p-phenyl phenol, diphenyl, 1-methylnaphthalene, ethylene carbonate and propylene carbonate. Thus, the colouring liquor may consist essentially of the at least one colourant (e.g. a dye, for example a disperse dye) dissolved in the first solvent system (e.g. a polar organic solvent). Thus, where the invention comprises the step of dissolving the colourant (e.g. dye) in the first solvent system to form the colouring liquor, the colourant (e.g. dye) that is dissolved may be substantially pure (e.g. greater than 90% or greater than 95% pure). The inventors have found that excellent colour strength can be obtained using the method of the invention in the absence of a dispersing agent or a carrier.
[0032] It may be that the second solvent system does not comprise a dispersing agent. It may be that the second solvent system does not comprise a carrier. Thus, the second solvent system may consist essentially of the solvent or solvents that form the second solvent system.
[0033] Alternatively, the colouring liquor and/or the second solvent system may comprise at least one additive selected from: a dispersing agent, a carrier, a stabiliser, a surfactant, an antioxidant, a pH modifier/buffer, lubricant, softener, hydrotope, wetting agent and migrating agent. The colouring liquor and/or the second solvent system may comprise at least one additive selected from: a stabiliser, a surfactant, an antioxidant, a pH modifier/buffer, lubricant, softener, hydrotope, wetting agent and migrating agent.
[0034] T1 and T2 may be the same. T1, T2 and T3 may be the same.
[0035] T1 may be greater than 70.degree. C. T1 may be greater than 80.degree. C. T1 may be greater than 90.degree. C.
[0036] T2 may be greater than 70.degree. C. T2 may be greater than 80.degree. C. T2 may be greater than 90.degree. C.
[0037] T3 may be greater than 70.degree. C. T3 may be greater than 80.degree. C. T3 may be greater than 90.degree. C.
[0038] T4 may be less than 70.degree. C. T4 may be less than 60.degree. C.T1 may be in the range 25.degree. C. to 70.degree. C. T2 may be in the range 25.degree. C. to 70.degree. C. T2 may be in the range 25.degree. C. to 70.degree. C. Certain types of colourant, and particularly dyes used to colour natural fibres, such as, wool, silk and cotton, can be used effectively at temperatures below 70.degree. C. using the methods of the invention.
[0039] The method may be conducted at a pressure of about 1 atm. The method may be conducted at a pressure in the range from 0.9 atm to 1.5 atm. The method may be conducted at an elevated pressure, e.g. a pressure greater than 1 atm and up to 5 atm.
[0040] It may be that the weight ratio of the polymer (e.g. fibre) substrate to the first solvent system is in the range from 4:1 to 1:4. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the first solvent system is in the range from 3:1 to 1:3. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the first solvent system is in the range from 2:1 to 1:2.5. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the first solvent system is in the range from 1:1 to 1:2. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the first solvent system is in the range from 1:0.1 to 1:15.
[0041] It may be that the ratio of the polymer (e.g. fibre) substrate to the total amount of the second solvent system is in the range from 3:1 to 1:15. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the total amount of the second solvent system is in the range from 2:1 to 1:10. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the total amount of the second solvent system is in the range from 1:1 to 1:4.
[0042] It may be that the weight ratio of the polymer (e.g. fibre) substrate to the total amount of the first and second solvent systems is in the range from 1:1 to 1:20. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the total amount of the first and second solvent systems is in the range from 1:1 to 1:10. It may be that the weight ratio of the polymer (e.g. fibre) substrate to the total amount of the first and second solvent systems is in the range from 1:2 to 1:5.
[0043] It may be that the amount of colourant (e.g. dye) used is in the range from 0.5 to 10% of the mass of the polymer substrate (e.g. 0.5 to 10% on mass fibre). It may be that the amount of colourant (e.g. dye) used is in the range from 1 to 5% of the mass of the polymer substrate (e.g. 1 to 5% on mass fibre).
[0044] Typically, the total volume of the first solvent system to which the polymer (e.g. fibre) substrate is subjected is less than the total volume of the second solvent system to which the polymer (e.g. fibre) substrate is subjected. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:2 to 1:20. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:3 to 1:15. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 30:70 to 10:90.
[0045] The colouring process may be followed by reduction clearing, rinsing and optionally further treatments depending on the nature of the substrate (e.g. whether the substrate is a single or multicomponent blend of fibres), and end-use requirement. Exemplary further treatments include softening, heat setting, etc.
The Solvent Systems
[0046] The methods of the invention involve the use of two solvent systems. The colourant (e.g. dye) is more soluble in the first solvent system than it is in the second solvent system.
[0047] The first solvent system should be capable of dissolving at least one colourant.
[0048] The first solvent system may be supercritical CO.sub.2. The first solvent system may be a surfactant (e.g. a polyethoxylated fatty acid or and fatty acid ester or a mixture thereof) or a solution of a surfactant in water.
[0049] Preferably, however, the first solvent system may comprise an organic solvent or a mixture of two or more organic solvents. The first solvent system may be an organic solvent or a mixture of two or more organic solvents. The first solvent system may be an organic solvent. The first solvent system may comprise a mixture of two or more organic solvents. Where the first solvent system comprises an organic solvent or a mixture of two or more organic solvents, the inventors have shown that the presence of water is tolerated in the first solvent system but, typically, the first solvent system may comprise water but the water will typically represent less than 50% (e.g. less than 10%) by total weight of the first solvent system. Thus, the first solvent system may comprise less than 5% (e.g. less than 1%) water. The first solvent system may comprise a mixture of water and an organic solvent.
[0050] The first solvent system may be an organic solvent having a molecular weight below 200 or a mixture of two or more organic solvents each having a molecular weight below 200. The first solvent system may be an organic solvent having a molecular weight below 175 or a mixture of two or more organic solvents each having a molecular weight below 175. The first solvent system may be an organic solvent having a molecular weight below 150 or a mixture of two or more organic solvents each having a molecular weight below 150. The first solvent system may be an organic solvent having a molecular weight below 120 or a mixture of two or more organic solvents each having a molecular weight below 120. The first solvent system may be an organic solvent having a molecular weight below 100 or a mixture of two or more organic solvents each having a molecular weight below 100. The first solvent system may be an organic solvent having a molecular weight below 80 or a mixture of two or more organic solvents each having a molecular weight below 80. The first solvent system may be an organic solvent having a molecular weight above 80 or a mixture of two or more organic solvents each having a molecular weight above 80.
[0051] The first solvent may be an organic solvent that is a liquid at 25.degree. C. and 1 atm or a mixture of two or more organic solvents each of which is a liquid at 25.degree. C. and 1 atm. The first solvent may be an organic solvent that is a liquid at 0.degree. C. and 1 atm or a mixture of two or more organic solvents each of which is a liquid at 0.degree. C. and 1 atm.
[0052] The first solvent system may be selected from: a non-polar organic solvent (examples include pentane, hexane, benzene, toluene, dichloromethane, cyclohexane, heptane, CCl.sub.4 etc), a polar aprotic solvent (e.g. acetone, methyl-t-butylketone, N-methylpyrollidinone, N,N-dimethylformamide, N,N-dimethylacetamide, tetrahydrofuran, diethylether, ethylacetate, dimethylsulfoxide, diethylene glycol diethyl ether, ethylene glycol diacetate etc.) and a protic polar solvent (e.g. ethanol, methanol, propanol, isopropanol, ethylene glycol, glycerol, triethylene glycol monomethyl ether, dipropylene glycol methyl ether, 1-methoxy-2-propanoletc) or a mixture thereof. The first solvent system may comprise a polar organic solvent or a mixture of two or more polar organic solvents. The first solvent system may be a polar organic solvent or a mixture of two or more polar organic solvents. The first solvent system may comprise a polar aprotic organic solvent or a mixture of two or more polar aprotic organic solvents. The first solvent system may be an organic solvent comprising an ether group or a mixture of two or more organic solvents comprising an ether group. The first solvent system may be an organic solvent comprising both an ether group and a hydroxy group or a mixture of two or more organic solvents comprising both an ether group and a hydroxy group. The first solvent system may be a polar aprotic organic solvent or a mixture of two or more polar aprotic organic solvents. The first solvent system may comprise an organic solvent that comprises carbon, hydrogen, oxygen, nitrogen and sulphur or a mixture of two or more organic solvents that comprise carbon, hydrogen, oxygen, nitrogen and sulphur. The first solvent system may comprise an organic solvent that comprises carbon, hydrogen and oxygen or a mixture of two or more organic solvents that comprise carbon, hydrogen and oxygen.
[0053] The first solvent system may comprise acetone. The first solvent system may be acetone. The first solvent system may be a mixture of acetone and one or more other polar organic solvent, e.g. a mixture of acetone and ethanol.
[0054] The first solvent system may comprise DMSO. The first solvent system may be DMSO. The first solvent system may be a mixture of DMSO and one or more other polar organic solvent, e.g. a mixture of DMSO and ethanol or a mixture of DMSO and acetone.
[0055] The first solvent system may comprise glycerol. The first solvent system may be glycerol. The first solvent system may be a mixture of glycerol and one or more other polar organic solvent, e.g. a mixture of acetone and glycerol.
[0056] The first solvent system may comprise a solvent selected from ethylene glycol diacetate, triethylene glycol monomethyl ether, dipropylene glycol methyl ether and 1-methoxy-2-propanol. The first solvent system may be a solvent selected from ethylene glycol diacetate, triethylene glycol monomethyl ether, dipropylene glycol methyl ether and 1-methoxy-2-propanol or a mixture thereof.
[0057] The first solvent system may comprise a solvent selected from glycerol, ethylene glycol diacetate, triethylene glycol monomethyl ether, dipropylene glycol methyl ether and 1-methoxy-2-propanol. The first solvent system may be a solvent selected from glycerol, ethylene glycol diacetate, triethylene glycol monomethyl ether, dipropylene glycol methyl ether and 1-methoxy-2-propanol or a mixture thereof.
[0058] The second solvent system should be one in which the colourant (e.g. dye) is poorly soluble. The second solvent system may be an organic solvent in which the colourant (e.g. dye) is less soluble than the colourant (e.g. dye) is in the first solvent system. Typically, the second solvent system comprises water. The second solvent system may be water or an aqueous solution. The second solvent system may be a mixture of water or an aqueous solution and an organic solvent. The second solvent system may be water. The second solvent may comprise a mixture of two or more organic solvents.
[0059] Where the second solvent system is an aqueous solution it may be a solution of an electrolyte, an acid, a base or a buffer or a mixture of an electrolyte with an acid, a base or a buffer.
[0060] Suitable electrolytes include NaCl, Na.sub.2SO.sub.4, ammonium sulfate and others that are commonly used in the application of dyes by dyeing.
[0061] Suitable bases include Na.sub.2CO.sub.3, NaHCO.sub.3, K.sub.2CO.sub.3, KOH, NaOH and others that are commonly used in the application of dyes by dyeing. Suitable acids include acetic acid, formic acid, and others that are commonly used in the application of dyes by dyeing.
[0062] Suitable buffers include those based upon citrate, phosphate, acetate and others that are commonly used in the application of dyes by dyeing.
[0063] Electrolytes are particularly useful when using a reactive dye, a vat dye or a direct dye.
[0064] Bases are particularly useful when using a reactive dye.
[0065] It may be that the first solvent system is miscible with the second solvent system. Thus, where the second solvent system is or comprises water, the first solvent system may be water miscible.
[0066] Both the first and second solvent system are typically selected such that the polymer (e.g. fibre) substrate is not soluble in either the first or second solvent system.
[0067] Typically, the total volume of the first solvent system to which the polymer (e.g. fibre) substrate is subject is less than the total volume of the second solvent system to which the polymer (e.g. fibre) substrate is subjected. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:1.1 to 1:10. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:2 to 1:20. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:3 to 1:15. The ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 30:70 to 10:90.
The Substrate
[0068] The polymer substrate may comprise natural, man-made and/or synthetic polymers of organic or inorganic derivation, including, polypeptides, polysaccharides, hydrocarbons, elastomers, thermosets and thermoplastics, as exemplified by, but not limited to, polymers such as collagen, keratin, cellulosics, alginates, polysulfide, polyamide, poly(lactic acid), polyvinyl chloride, polyacrylonitrile, polyethylene, polypropylene, polystyrene, polyurethane, aramid and polyimide.
[0069] The polymer substrate may take any solid physical form, including powder, pellet, sheet, film, fibre or any irregular shape. The polymer substrate may be a moulded plastic shape, e.g. a car bumper or a pair of spectacles. The polymer substrate may be a 3D printed object.
[0070] The polymer substrate may comprise more than one type of polymer. The polymer substrate may comprise two or more polymers present conjointly in various mixtures formed by physical blending, mixing, dissolution, precipitation, the interconnection of moulded or 3D printed parts, etc.
[0071] The inventors have found that the methods of the invention can be used to simultaneously colour different polymers with different types and classes of colourant.
[0072] The polymer substrate may be a fibre substrate, e.g. a yarn, a fabric or a garment or part of a garment.
[0073] The fibre substrate may comprise synthetic fibres or natural fibres or a mixture thereof. The fibre substrate may comprise fibres selected from: a polyester, a polyamide, a polyurethane, a polyalkylene, a polyacrylonitrile, wool, silk, natural or regenerated cellulose, cellulose ester, hair, polyvinyl chloride, carbon or a mixture thereof.
[0074] Exemplary polyesters include poly(ethylene terephthalate) (PES), poly(butylene terephthalate) (PBT), poly(trimethylene terephthalate) (PTT). Exemplary polyurethanes include Lycra.RTM.. Exemplary polyamides include nylon.
[0075] The fibre substrate may be or may comprise polyamide fibres, e.g. nylon fibres.
[0076] The fibre substrate may comprise polyester fibres or a mixture of a polyester with a fibre selected from cotton, wool, silk and polyurethane (e.g. Lycra.RTM.). The fibre substrate may comprise PES fibres or a mixture of PES with a fibre selected from cotton, wool, silk and polyurethane (e.g. Lycra.RTM.). The fibre substrate may be a polyester. The fibre substrate may be PES. The fibre substrate may comprise a mixture of a polyester with a fibre selected from cotton, wool, silk and polyurethane (e.g. Lycra.RTM.). The fibre substrate may comprise a mixture of PES with a fibre selected from cotton, wool, silk and polyurethane (e.g. Lycra.RTM.).
[0077] The fibre substrate may comprise silk or wool fibres.
[0078] It may be that the fibre substrate comprises both polyester fibres and at least one other type of fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane. The fibre substrate may comprise a material that is a blend of polyester fibres and at least one fibre selected from other type of fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane. The fibre substrate may be a material that is a blend of polyester fibres and at least one fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane. Alternatively, the fibre substrate comprises a first material that comprises polyester fibres and a second material that comprises at least one fibre selected from cotton, regenerated cellulose, wool, silk, polyamide, a different polyester, polyvinylchloride, polyacrylonitrile, mohair, cashmere and a polyurethane. The fibre substrate may be a whole garment, e.g. a trainer or pair of trainers, a shirt or blouse, a dress, a pair of trousers, a skirt, a t-shirt etc.
[0079] It may be that the fibre substrate comprises both polyester fibres and at least one other type of fibre selected from cotton, wool, silk, mohair, cashmere and a polyurethane. The fibre substrate may comprise a material that is a blend of polyester fibres and at least one fibre selected from other type of fibre selected from cotton, wool, silk, mohair, cashmere and a polyurethane. The fibre substrate may be a material that is a blend of polyester fibres and at least one fibre selected from cotton, wool, silk, mohair, cashmere and a polyurethane. Alternatively, the fibre substrate comprises a first material that comprises polyester fibres and a second material that comprises at least one fibre selected from cotton, wool, silk, mohair, cashmere and a polyurethane.
[0080] As mentioned above, the inventors have found that the methods of the invention can be used to simultaneously colour different polymers with different types and classes of colourant. In particular, the methods of the invention can be used to simultaneously colour (e.g. dye) different fibres with different types and classes of colourant (e.g. dye). This offers the possibility that, with judicious choice of fabric, a blended fabric could be generated in which a pattern is designed into the weave of the fabric and the different fibres within the weave can simultaneously and selectively be dyed different colours. Likewise, a whole garment that is formed of two or more different materials (e.g. a shoe, for example a sports shoe or pair of sports shoes) can be dyed once formed and the various materials can be dyed different colours simultaneously. This would allow for the bespoke dyeing, based on the purchaser's selected colour preferences, of a whole garment either in a distribution warehouse or in a shop.
The Colourant(s)
[0081] The at least one colourant may comprise at least one pigment.
[0082] The at least one colourant may comprise at least one dye.
[0083] The at least one colourant may be a single dye. The at least one colourant may be mixture of two or more dyes.
[0084] Suitable dyes include disperse dyes, solvent dyes, vat dyes, sulphur dyes, mordant dyes, acid dyes, direct dyes and reactive dyes. The disperse dyes, solvent dyes, vat dyes, sulphur dyes, mordant dyes, acid dyes, direct dyes and reactive dyes that can be used in the methods of the invention include all dyes classified as such in The Colour Index.TM. published by the Society of Dyers and Colourists (SDC) and American Association of Textile Chemists and Colourists (AATCC). In certain embodiments, the disperse dyes, solvent dyes, vat dyes, sulphur dyes, mordant dyes, acid dyes, direct dyes and reactive dyes that can be used in the methods of the invention may include all dyes classified as such in The Colour Index.TM. on the 1 May 2017.
[0085] The at least one dye may include a disperse dye. The single dye may be a disperse dye.
[0086] The at least one dye may include an acid dye (e.g. a non-metallised acid dye or a pre-metallised acid dye). The single dye may be an acid dye (e.g. a non-metallised acid dye or a pre-metallised acid dye).
[0087] The at least one dye may include a vat dye. The single dye may be a vat dye.
[0088] The at least one dye may include a reactive dye. The single dye may be a reactive dye.
[0089] The at least one dye may include a direct dye. The single dye may be a direct dye.
[0090] The at least one pigment may include a pigment selected from an organic pigment, an inorganic pigment and a metallic pigment.
[0091] Where the polymer (e.g. fibre) substrate to be coloured comprises different polymers (e.g. different fibres), a mixture of two or more classes or types of colourant (e.g. dye) may be used. For example, where a fibre substrate comprises both polyester fibres (e.g. PES) and a natural fibre (e.g. cotton, silk or wool), a mixture of a disperse dye (to dye the polyester fibres) and a reactive dye (to dye the natural fibres) might be used. Alternatively a mixture of a disperse dye (to dye the polyester) and a direct dye (to dye the natural fibre) might be used. In a further alternative, a mixture of a disperse dye (to dye the polyester fibres) and an acid dye (to dye the natural fibres) might be used. In a further example, where a fibre substrate comprises polyester fibres (e.g. PES), cotton fibres and either wool or silk fibres, a mixture of a disperse dye (to dye the polyester fibres), a reactive dye (to dye the cotton fibre) and an acid dye (to dye the wool or silk fibres) might be used.
[0092] The invention may further be described in the following numbered paragraphs: [0093] 1. A method of dyeing a fibre substrate, the method comprising: [0094] a) subjecting the fibre substrate to a dye liquor at a temperature T1, T1 being below 100.degree. C., said dye liquor comprising at least one dye dissolved in a first solvent system to provide the fibre substrate wetted with the dye liquor; [0095] b) adding a second solvent system to the fibre substrate wetted with the dye liquor, without raising the temperature above a temperature T2, T2 being below 100.degree. C., to provide the dyed fibre substrate wetted with a mixture of the first solvent system and the second solvent system; and [0096] c) separating the dyed fibre substrate from the mixture of the first and second solvent systems and any remaining dye; wherein the dye or each dye is more soluble in the first solvent system than in the second solvent system. [0097] 2. A method of paragraph 1, wherein the step of adding the second solvent system comprises adding the second solvent system portionwise to the mixture of the fibre substrate and the dye liquor. [0098] 3. A method of paragraph 1 or paragraph 2, wherein the second solvent system comprises water. [0099] 4. A method of paragraph 3, wherein the second solvent system is water. [0100] 5. A method of any preceding paragraph, wherein the first solvent system and the second solvent system are miscible. [0101] 6. A method of any preceding paragraph, wherein the first solvent system is an organic solvent or a mixture of one or more organic solvents. [0102] 7. A method of paragraph 6, wherein the first solvent system is a polar organic solvent or a mixture of polar organic solvents. [0103] 8. A method of paragraph 7, wherein the first solvent system is acetone. [0104] 9. A method of any preceding paragraph, wherein the fibre substrate comprises fibres selected from: a polyester, nylon, a polyurethane, wool, silk, cotton or a mixture thereof. [0105] 10. A method of paragraph 10, wherein the fibre substrate comprises polyester fibres or a mixture of a polyester with a fibre selected from cotton, wool, silk and a polyurethane 11. A method of any preceding paragraph, wherein the ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 1:2 to 1:20. [0106] 12. A method of paragraph 11, wherein the ratio of the total volume of the first solvent system to the total volume of the second solvent system may be in the range 30:70 to 10:90. [0107] 13. A method of any preceding paragraph, wherein the weight ratio of the fibre substrate to the first solvent system is in the range from 3:1 to 1:3. [0108] 14. A method of any preceding paragraph, wherein the at least one dye is a disperse dye. [0109] 15. A method of any preceding paragraph, wherein the method comprises dissolving the at least one dye in the first solvent system to form the dye liquor 16. A method of any preceding paragraph, wherein the dye liquor does not comprise a dispersing agent. [0110] 17. A fibre substrate obtainable according to the method of any preceding paragraph.
BRIEF DESCRIPTION OF THE DRAWINGS
[0111] Embodiments of the invention are further described hereinafter with reference to the accompanying drawings, in which:
[0112] FIG. 1 shows the high temperature (HT) PES dyeing method used as a comparator in the Examples below;
[0113] FIG. 2 shows the reduction clearing process employed for the Examples;
[0114] FIG. 3 provides a general depiction of controlled precipitation dyeing method of the invention;
[0115] FIG. 4 shows the colour strength of 2% omf dyeings on PES achieved using controlled precipitation method of the invention; Teratop Yellow HL-G 150%; 120 mins at 98.degree. C.;
[0116] FIG. 5 shows the colour strength of 2% omf dyeings on PES achieved using controlled precipitation method of the invention; Teratop Yellow HL-G 150%; 30', 60' and 90' at 98.degree. C.;
[0117] FIG. 6 shows the colour strength of 2% omf dyeings on PES achieved using controlled precipitation method of the invention for 20 mins at 98.degree. C.; Teratop Yellow HL-G 150%;
[0118] FIG. 7 shows the colour strength of 2% omf dyeings on PES achieved using controlled precipitation method of the invention for 120 mins at 85.degree. C.; Teratop Yellow HL-G 150%;
[0119] FIG. 8 shows the colour strength of 2% omf dyeings on PES achieved using controlled precipitation method of the invention for 20 mins at 98.degree. C. (Teratop Blue HL-G 150%: left and Teratop Pink HL-G 150%: right);
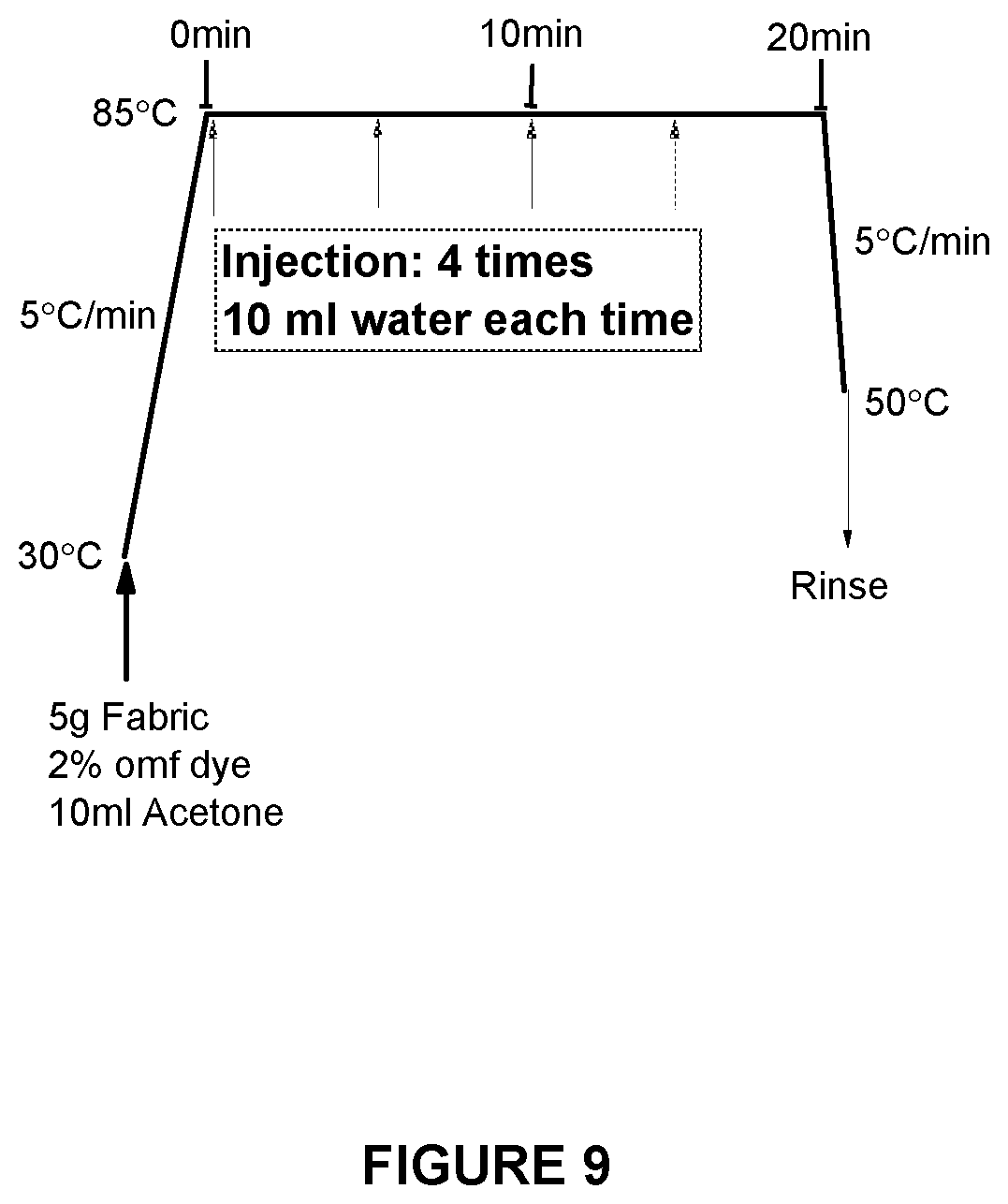
[0120] FIG. 9 shows the dyeing method used for the dyeing methods described in Examples 3, 4 and 5;
[0121] FIG. 10 shows the colour strength of 2% omf dyeing of crude grade Teratop Yellow HL-G 150% on a fabric composite comprising polyester fabric attached to scoured PA 66 fabric;
[0122] FIG. 11 shows the colour strength of 2% omf dyeings of commercial Teratop Yellow HL-G 150% on PES as a function of dyeing temperature using DMSO as solvent
DETAILED DESCRIPTION
[0123] Organic solvents are organic compounds that are liquids at room temperature and atmospheric pressure. Typically, organic solvents are compounds that comprise both carbon atoms and hydrogen atoms. One exception to this is carbon tetrachloride. Organic solvents may also comprise oxygen, nitrogen, chlorine, fluorine or sulphur.
[0124] The term `wetted` is used in this specification to mean that the polymer (e.g. fibre) substrate is in contact with a liquid, e.g. the dye liquor or the mixture of the first solvent system and the second solvent system. It may mean that the substrate (e.g. fibre substrate) is soaked in the liquid, e.g. saturated with the liquid. It may mean that the substrate (e.g. fibre substrate) is coated in the liquid. It may mean that the substrate (e.g. fibre substrate) is immersed in the liquid. Other techniques that could be used include spraying, soaking, vapourisation, imbibition, etc.
[0125] The polymer (e.g. fibre) substrate is described as `coloured` where the colourant molecules have become absorbed into the substrate (e.g. into the fibres). Throughout the description and claims of this specification, the words "comprise" and "contain" and variations of them mean "including but not limited to", and they are not intended to (and do not) exclude other moieties, additives, components, integers or steps. Throughout the description and claims of this specification, the singular encompasses the plural unless the context otherwise requires. In particular, where the indefinite article is used, the specification is to be understood as contemplating plurality as well as singularity, unless the context requires otherwise.
[0126] Features, integers, characteristics, compounds, chemical moieties or groups described in conjunction with a particular aspect, embodiment or example of the invention are to be understood to be applicable to any other aspect, embodiment or example described herein unless incompatible therewith. All of the features disclosed in this specification (including any accompanying claims, abstract and drawings), and/or all of the steps of any method or process so disclosed, may be combined in any combination, except combinations where at least some of such features and/or steps are mutually exclusive. The invention is not restricted to the details of any foregoing embodiments. The invention extends to any novel one, or any novel combination, of the features disclosed in this specification (including any accompanying claims, abstract and drawings), or to any novel one, or any novel combination, of the steps of any method or process so disclosed.
[0127] The reader's attention is directed to all papers and documents which are filed concurrently with or previous to this specification in connection with this application and which are open to public inspection with this specification, and the contents of all such papers and documents are incorporated herein by reference.
EXAMPLES
General Methods
Materials
[0128] Scoured poly(ethylene terephthalate) (PES) fabric (120 gm.sup.-2), scoured, bleached and mercerised woven cotton fabric (180 gm.sup.-2) and scoured PA 66 fabric (116 gm.sup.-2) was obtained from Whaleys (Bradford, UK). Commercial grade as well as crude grade (ie as-synthesised) samples of three disperse dyes, namely Teratop Yellow HL-G 150%, Teratop Blue HL-G 150% and Teratop Pink HL-G 150% were kindly provided by Huntsman (Switzerland). Commercial dyes were used in this work so as to reflect commercial dyeing practice whilst equivalent crude grade dyes were also used to determine whether it would be possible to dye PES using dispersant-free disperse dyes. The three dyes used were chosen arbitrarily as being representative of modern commercial disperse dyes. A 2% omf depth of shade was used for each of the three dyes as this provided typical pale/medium depth dyeings.
[0129] Samples of Polysorbate 20, Polysobate 60, Polysorbate 80, Lecithin and Superclean were obtained from Sigma-Aldrich. All other chemicals were of general purpose grade.
[0130] From measured values of the relative dye contents of the respective pairs of commercial grade and crude grade dye samples, the amount of crude grade dye employed in dyeing was adjusted so that the depth of shade obtained (ie 2% omf) was the same as that secured using the commercial grade dye, was obtained.
Comparative General Method
[0131] The high temperature (HT) dyeing method used for comparison in this work is shown in FIG. 1.
[0132] The PES fabric (5 g or 10 g) was dyed using both the commercial grade and grade crude disperse dyes following the procedure shown in FIG. 1. At the end of dyeing, the dyed sample was removed from the dyebath, squeezed to remove surplus dye liquor and rinsed in water according to the procedure depicted in FIG. 1. The dyed sample was then squeezed once more and subjected to the reduction clear procedure displayed in FIG. 2. At the end of the reduction clearing process the sample was removed, squeezed to remove surplus liquor and rinsed in cold water as shown in FIG. 2. The reduction cleared dyeing was squeezed and allowed to dry in the open air.
General Method of the Invention
[0133] The controlled precipitation method of the invention is illustrated in a general sense in FIG. 3.
Example 1--Development of the Methodology Using Disperse Dyes, PES and Acetone
[0134] The method depicted in FIG. 3 was carried out with a.sub.1; a.sub.2; a.sub.3; a.sub.4=10 cm.sup.3 water; t.sub.1=30'; t.sub.2=30'; a.sub.3=30'; a.sub.4=30'; x=98.degree. C.
[0135] The disperse dye was dissolved in a given volume (in this case 10 cm.sup.3) of acetone and the ensuing solution was applied to the PES fibre.
[0136] When the temperature of the dyebath reached 98.degree. C., a volume (10 cm.sup.3) of water was added and dyeing continued for a certain period of time, after which a further 10 cm.sup.3 of water was added. This process continued until the final dyebath volume was 50 cm.sup.3, corresponding to a 1:10 liquor ratio. Thus, over the length of the dyeing process, the ratio of water:acetone was gradually increased from 0:100 at the start of dyeing to 80:20 at the end of dyeing.
[0137] Without wishing to be bound by theory, the purpose of progressively introducing water to the acetone dye solution was to gradually force the disperse dye to precipitate out of solution in a controlled manner, so as to achieve increased dye-fibre substantivity and, thus, increased dye uptake, in a controlled and measured manner.
[0138] Following the procedure just described, the total dyeing time at 98.degree. C. was 120 mins. FIG. 4 reveals that using this approach, the ensuing dyeing displayed very high colour strength, which was considerably greater than that secured using the commercial grade dye and the conventional HT dyeing method at 130.degree. C. Visual inspection revealed that the dye precipitation method of the invention not only provided a deeper depth of shade than the HT method but, significantly, the level of dyebath exhaustion secured was much greater.
[0139] Thus the higher colour strength of the 120 mins 98.degree. C. dyeing (FIG. 4) can be attributed to the greater extent of dye exhaustion achieved. The fact that the residual dyebath was essentially free of unexhausted disperse dye offers obvious cost and environmental potential. In other words, in order to achieve a colour strength that is equivalent to that furnished using a commercial grade dye employing a conventional 2% omf dyeing method at 130.degree. C. (ie an f.sub.k value of .about.50) will require less disperse dye, if the controlled precipitation dyeing method of the invention is used (ie .about.1.25% omf dye), because virtually 100% dye exhaustion is achieved. Furthermore, because crude grade disperse dye was used for the precipitation dyeing method of the invention, then the exhausted dyebath will contain essentially no dye, zero dispersing agent and zero levelling agent, compared to the residual dyebath that will ensue from the conventional HT dyeing method.
[0140] Attempts were made to reduce the length of dyeing time at 98.degree. C.; for this, in FIG. 3, a.sub.1, a.sub.2, a.sub.3 and a.sub.4=10 cm.sup.3 water and the total time at the commercial boil was 90 min, 60 min and 30 min (in all cases a.sub.1=a.sub.2=a.sub.3=a.sub.4. As FIG. 5 shows, reducing the dyeing time at 98.degree. C. did not affect the colour yield of the ensuing dyeings; a comparison of FIGS. 4 and 5 shows that the colour yields of all four dyeings were the same.
[0141] When the total time at the commercial boil was further reduced to 20 mins (ie, a.sub.1, a.sub.2, a.sub.3 and a.sub.4=10 cm.sup.3 water and a.sub.1=a.sub.2=a.sub.3=a.sub.4=5') the colour strength of the dyed fabric was similar to that achieved for dyeing times of 30, 60, 90 and 120 mins (FIG. 6). However, with further reduction of dyeing time at the commercial boil the colour strength of the dyeings reduced.
[0142] When the temperature of dyeing was lowered from 98.degree. C. to 85.degree. C., it was found (FIG. 7) that whilst the colour strength of dyeings carried out for 120 min were the same as those achieved at 98.degree. C., shorter dyeing times at 85.degree. C. of 60 min and 90 min produced dyeings that were of lower colour strength. As observed for dyeings undertaken at the commercial boil, the residual dyebath obtained after 120 min at 85.degree. C. was observed on visual inspection to be devoid of dye.
[0143] When two other disperse dyes, namely Teratop Blue HL-G 150% and Teratop Pink HL-G 150%, were applied to PES using the precipitation dyeing method of the invention for 20 min at 98.degree. C., and the colour strengths of the dyeings were compared to those secured using the HT dyeing method (ie at 130.degree. C.) are shown in FIG. 8.
[0144] It is apparent that for each of the dyes, higher colour strength dyeings were achieved using the precipitation dyeing method of the invention at 98.degree. C. (FIG. 8) and, also, that the extent of dye exhaustion observed, was much higher for the HT dyeing method.
Example 2--Fastness
[0145] Table 1 shows that 2% omf dyeings obtained using the three commercial grade dyes when applied using the HT method (i.e. 130.degree. C.) displayed very good fastness to washing at 60.degree. C., as expected; visual inspection also showed the impressive depths of shade of the dyeings after wash fastness testing. The results presented in Table 1 also reveal that the corresponding dyeings which had been produced using crude grade samples of the three dyes employing the precipitation dyeing method of the invention at 98.degree. C. for 20 min displayed essentially the same high level of wash fastness. The latter findings are impressive when it is recalled that the colour strength of the 98.degree. C. dyeings were much greater than that of their 130.degree. C. counterparts. Thus, as expected, the manner by which the disperse dyes were applied (ie differences in dyeing temperature, dyeing duration and acetone) had no effect on wash fastness.
TABLE-US-00001 TABLE 1 fastness of 2% omf dyeings on PES produced using the HT method (commercial grade dyes at 130.degree. C.) and 20 min precipitation method of the invention at 98.degree. C., to ISO 105-C06/C2S (60.degree. C.) Teratop HL- dyeing change bleached G 150% grade temp/.degree. C. in shade wool acrylic polyester polyamide cotton Diacetate Yellow commercial 130 5 5 5 5 4/5 5 4.5 crude 98 5 5 5 5 4.5 5 4.5 Blue commercial 130 4/5 4/5 5 4 5 5 4/5 crude 98 4/5 4/5 5 4 4/5 5 4/5 Pink commercial 130 5 4 5 4 4/5 5 4/5 crude 98 5 4 5 4 4/5 5 4/5
[0146] When the above method was followed but using polyester fabric which had been previously wetted-out using water and squeezed to remove surplus water, the colour strength of the ensuing dyeing obtained for 20 min at 98.degree. C. was comparable to that achieved using dry polyester fabric.
Example 3--Other Fabrics and Other Classes of Dyes
[0147] The methods of the invention can also be used to dye other substrates using other dye types. The following example describes the dyeing of wool, silk and polyamide substrates with acid dyes and a disperse dye.
[0148] The general method used throughout this example is shown in FIG. 9. A Roaches Pyrotec S dyeing machine) was used. 0.1 g of commercial dye was dissolved in 10 cm.sup.3 acetone and the ensuing solution was placed in a 300 cm.sup.3 capacity dye tube, followed by 5 g of fabric. The sealed dye tube was heated to 85.degree. C. and then 10 cm.sup.3 of water was injected into the dyeing tube. A further total of 40 cm.sup.3 of water was injected at time intervals, as shown in FIG. 9. The total dyeing time at 85.degree. C. was 20 min.
[0149] Both non-metallised acid (Erionyl Red A-28F (Huntsman)) and 1:2 pre-metallised acid dyes (Supralan Yellow 4GL (Dystar); Lanaset Yellow 2R (Huntsman) and Neutrilan Yellow A-3R (Yorkshire)) were applied to wool, silk and PA fabrics using the dyeing methods of the invention. In addition, both commercial grade as well as crude grade samples of the disperse dye Teratop Yellow HL-G 150% (Huntsman) was applied to wool, PA and wool fibres.
[0150] Both silk and wool were successfully dyed at 85.degree. C. in 20 min using the non-metallised dye Erionyl Red A-28F. colour measurement L*=34.3 a*=63.4 b*=58.1
[0151] In comparison to the conventional method for dyeing wool with such dye types, which is normally carried out at higher temperature (for wool: 98.degree. C.) under acidic conditions for 60-90 min, the novel dyeing method is advantageous insofar as it enables the fibres to be dyed at a lower temperature of 85.degree. C. (for wool) in a short time (ie 20 min) without recourse to pH adjustment, thereby offering savings in time, energy and chemicals.
[0152] Both silk and wool were successfully dyed at 85.degree. C. for 20 min using the 1:2 metal complex dye Supralan Yellow 4GL at 85.degree. C. for 20 min using the novel dyeing method colour measurement L*=83.8 a*=6.9 b*=116.5. Once again, compared to the conventional method for dyeing wool with such dye types, which is normally carried out at higher temperature (for wool: 98.degree. C.) under acidic conditions for 60-90 min, the novel dyeing enables the fibres to be dyed at 85.degree. C. in a short time (ie 20 min) and without using pH adjustment, thereby offering savings in time, energy and chemicals.
[0153] The novel dyeing method also enabled wool and silk to be dyed at 85.degree. C. for 20 min using the 1:2 metal complex dyes Neutrilan Yellow A-3R colour measurement L*=48.7 a*=34.5 b*=8.1 and Lanaset Yellow 2R. Once again, compared to the conventional method for dyeing wool with such dye types, which is normally carried out at higher temperature (for wool) under acidic conditions for 60-90 min, the novel dyeing enables the fibres to be dyed at 85.degree. C. in a short time (ie 20 min), thereby offering savings in time, energy and chemicals.
[0154] Both commercial (i.e. containing dispersants) and crude (i.e. not containing dispersants) samples of the disperse dye Teratop Yellow HL-G 150% were successfully applied to both scoured wool and silk and PA fibres using the novel dyeing method at 85.degree. C. and 20 min.
Example 4--Blends of Fibres
[0155] The methods of the invention can also be used to dye a combination of different types of fibre. The following example describes the dyeing of polyester/cotton blends as well as polyester/Nylon blends using disperse dye.
[0156] The general method used throughout this example is shown in FIG. 9. A Roaches Pyrotec S dyeing machine) was used. The appropriate amount of crude grade disperse dye to provide a 2% omf shade was dissolved in 10 cm.sup.3 acetone and the ensuing solution was placed in a 300 cm.sup.3 capacity dye tube, followed by a fabric composite comprising 2.5 g of polyester fabric attached to 2.5 g of scoured, bleached and mercerised woven cotton fabric. The sealed dye tube was heated to 98.degree. C. and then 10 cm.sup.3 of water was injected into the dyeing tube. A further total of 40 cm.sup.3 of water was injected at time intervals, as shown in FIG. 9. The total dyeing time at 85.degree. C. was 20 min.
[0157] 2% omf dyeings of the crude (i.e. not containing dispersants) disperse dyes Teratop Yellow HL-G 150%, Teratop Blue HL-G 150% and Teratop Pink HL-G 150%, were successfully achieved using the novel dyeing method at 98.degree. C. and 20 min. Whilst the polyester component was fully dyed, the cotton fabric was uncoloured. This was anticipated based on the relative hydrophobicity of the cotton and polyester fibres and the corresponding different substantivity for the fibres displayed by the disperse dye.
[0158] Following the method described above, a 2% omf dyeing using the crude grade disperse dye Teratop Yellow HL-G 150% was obtained using a fabric composite comprising 2.5 g of polyester fabric attached to 2.5 g of scoured PA 66 fabric.
[0159] Both the polyester and nylon 66 fabrics were dyed, as shown by FIG. 10, but the polyester was of higher depth of shade, as expected, owing to the greater hydrophobicity of the polyester material and the corresponding greater substantivity displayed by the dye towards the polyester fibre.
Example 5--Solvent Mixtures
[0160] The first solvent can comprise a mixture of two or more organic solvents or a mixture of water and an organic solvent.
[0161] The general method used throughout this example is shown in FIG. 9, with the acetone replaced with mixed solvent systems as described below. A Roaches Pyrotec S dyeing machine) was used. Crude grade disperse dye Teratop Yellow HL-G 150%, was dissolved in a mixture of 10 cm.sup.3 acetone and 2 cm.sup.3 of water. The ensuing solution was placed in a 300 cm.sup.3 capacity dye tube, followed by polyester fabric. The sealed dye tube was heated to 98.degree. C. and then 10 cm.sup.3 of water was injected into the dyeing tube. A further total of 40 cm.sup.3 of water was injected at time intervals. The total dyeing time at 98.degree. C. was 20 min.
[0162] The colour strength of the ensuing dyeing was very similar to that obtained when acetone only had been used as the first solvent.
[0163] When the above method was used but crude grade disperse dye Teratop Yellow HL-G 150% was dissolved in a mixture of 4 cm.sup.3 acetone and 6 cm.sup.3 of ethanol, successful dyeing was achieved.
Example 6--Other Solvents
[0164] Although acetone is an excellent solvent for crude grade disperse dyes, other, higher boiling solvents were examined. Being higher boiling, these solvents offer a reduced fire risk compared to acetone.
[0165] Different amounts (5, 10 and 20 cm.sup.3) of DMSO were used to dissolve 2% omf commercial Teratop Yellow HL-G and different amounts of water (45, 40 and 30 cm.sup.3) were added portionwise so as to achieve a 1:10 LR overall were used. The colour strength of the dyed polyester is shown in FIG. 11
[0166] From FIG. 11 it is apparent that lower values of colour strength are obtained in comparison to that of PES which had been dyed using acetone, which can be attributed to the lower solubility of the dye in DMSO. However, as FIG. 11 shows, by increasing the amount of DMSO employed, higher colour strength dyeings were achieved. While these dyeings did not exhibit the high colour strengths of the acetone processes, they still offer benefits in terms of lower temperature of process with lower energy use and the ability to dye PES concurrently with non-PES fibres.
[0167] Various other high boiling point solvents were used to dye PES at a 2% omf depth of shade of crude Teratop Yellow HL-G, namely ethylene glycol diacetate (EGD), triethylene glycol monomethyl ether (TGM), dipropylene glycol methyl ether (DME) and 1-methoxy-2-propanol.
[0168] Samples of PES fabric were dyed at 95.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing each of the above solvents (10 cm.sup.3) and 4 additions of water (40 cm.sup.3 water in total; 1:10 LR in total), the total dyeing time at 95.degree. C. being 20 mins.
[0169] Colour measurement data (illuminant D.sub.65; specular included; UV excluded; 10.degree. standard observer)
triethylene glycol monomethyl ether (TGM) L*=88.9a*=30.5b*=95.3
ethylene glycol diacetate (EGD) L*=86.1a*=19.1b*=80.1
[0170] Each of the four solvents were able to dissolve the crude disperse dye and can be utilised in the precipitation dyeing method.
Example 7--Vat Dyes
[0171] To investigate whether the novel precipitation dyeing method could be used to apply to vat dyes, indigo was selected. Samples of PES fabric were dyed at 95.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing acetone as solvent (10 cm.sup.3) and 4 additions of water (40 cm.sup.3 water in total; 1:10 LR in total), the total dyeing time at 95.degree. C. being 20 mins.
[0172] It was found that the vat dye could be applied from acetone using the precipitation dyeing method.
[0173] No reductants or pH adjustments were needed to achieve this result.
[0174] Colour measurement data (illuminant D.sub.65; specular included; UV excluded; 10.degree. standard observer)
[0175] L*=55.9 a*=-6.3 b*=-10.9
Example 8--One Pot Dyeing of Mixtures of Fibres with Mixtures of Classes of Dyes
[0176] The novel precipitation dyeing method may offer the potential for dyeing fibre blends using different classes/types of dye simultaneously, in the same dyebath, in the absence/much reduced amounts of dyebath auxiliaries.
[0177] For this example, samples (2.5 g) of fabric were dyed at different temperatures using the controlled precipitation dyeing method shown in FIG. 3, employing acetone as primary solvent and using additions of water or solutions of inorganic electrolyte or alkali, as well as electrolyte/alkali, the total dyeing time being 20 mins. The amounts are given in the examples below
PES/Cotton Using Reactive Dye and Disperse Dye
[0178] Duractive Black B (C.I. Reactive Black 5) and crude Teratop Yellow HL-G were dissolved in acetone. Separate samples of PES fabric and cotton fabric were dyed together at 95.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing 4 additions of either a) water or b) a solution comprising 15 gl.sup.-1 Na.sub.2CO.sub.3 and 50 gl.sup.-1 NaCl (1:10 LR in total), the total dyeing time being 20 mins.
[0179] It was found that it is possible to dye both the hydrophobic PES and hydrophilic cotton substrates in the same dyebath in 20 minutes at 95.degree. C., using a mixture of non-ionic disperse dye and anionic reactive dye. As expected the colour yield secured using both electrolyte and alkali favoured reactive dye uptake.
[0180] Since it is now known that reactive dye exhaustion on cotton in the absence of added inorganic electrolyte can be increased through the use of low liquor ratio, dyeings were also made using two 10 cm.sup.3 additions (1:6 LR) of a solution comprising 15 gl.sup.-1 Na.sub.2CO.sub.3; an improvement in depth of shade of the reactive dye on the cotton component was achieved compared to that obtained at a 1:10 LR.
[0181] To samples of PES fabric and cotton fabric Commercial Novacron Red FN-2BL was applied in conjunction with crude Teratop Yellow HL-G dissolved in acetone. Four 10 cm.sup.3 additions of a solution comprising 15 gl.sup.-1 Na.sub.2CO were made (1:10 LR); the total dyeing time was 20 mins at 95.degree. C. It was found that the precipitation method enables PES and cotton substrates to be dyed in the same dyebath in 20 minutes at 95.degree. C., using a mixture of disperse dye and reactive dye.
Colour Measurement
[0182] PES L*=75.8 a*=18.9 b*=116.5
[0183] cotton L*=52.9 a*=4.3 b*=-19.8
PES/Cotton Using Direct Dye and Disperse Dye
[0184] C.I. Direct Red 81 and crude Teratop Yellow HL-G were dissolved in acetone. Samples of PES fabric and cotton fabric were dyed at 95.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing 4 additions of water or a solution containing 20 gl.sup.-1 NaCl (1:10 LR in total), the total dyeing time being 20 mins.
Colour Measurement
[0185] PES L*=73.1 a*=16.1 b*=117.6
[0186] cotton L*=41.1 a*=66.9 b*=19.4
[0187] Dyeings were also made using two 10 cm.sup.3 additions (1:6 LR) of water
[0188] Colour Measurement
[0189] PES L*=78.4 a*=35.8 b*=111.9
[0190] cotton L*=52.3 a*=61.6 b*=11.2
[0191] It was found that both PES and cotton fibres can be dyed simultaneously in the same dyebath in 20 minutes at 95.degree. C., using a mixture of disperse dye and direct dye; as expected the colour yield secured using electrolyte favoured dye uptake, and using a lower liquor ratio (ie 1:6) improved direct dye adsorption in the absence of added inorganic electrolyte.
PES/Wool Using Non-Metallised and Pre-Metallised Acid Dyes in Conjunction with Disperse Dye
[0192] Commercial samples of either a) the non-metallised acid dye Erionyl Red A-28F or b) the 1:2 pre-metallised acid dye Neutrilan Yellow A-3R were dissolved in acetone along with crude (i.e. containing no auxiliaries) Teratop Yellow HL-G. Samples of PES fabric and wool fabric were dyed at 85.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing 4 additions of water (1:10 LR in total), the total dyeing time being 20 mins.
[0193] It was found that PES and wool fibres can be dyed simultaneously in the same dyebath in 20 minutes at 85.degree. C., using a mixture of disperse dye and either non-metallised acid or pre-metallised acid dyes in the absence of all dyeing auxiliaries. The wool fabric component was dyed either a red colour or yellow colour depending on the type of acid dyes used, and the polyester fabric component was dyed a yellow colour
PES/Silk Using Non-Metallised and Pre-Metallised Acid Dyes in Conjunction with Disperse Dye
[0194] Commercial samples of either a) the non-metallised acid dye Erionyl Red A-28F or b) the 1:2 pre-metallised acid dye Neutrilan Yellow A-3R were dissolved in acetone along with crude (i.e. containing no auxiliaries) Teratop Yellow HL-G. Samples of PES fabric and silk fabric were dyed at 85.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing 4 additions of water (1:10 LR in total), the total dyeing time being 20 mins.
[0195] It was found that PES and wool fibres can be dyed simultaneously in the same dyebath in 20 minutes at 85.degree. C., using a mixture of disperse dye and either non-metallised acid or pre-metallised acid dyes in the absence of all dyeing auxiliaries. The silk fabric component was dyed either a red colour or yellow colour depending on the type of acid dyes used, and the polyester fabric component was dyed a yellow colour
PES/Cotton/Wool Using Non-Metallised Acid and Reactive Dyes in Conjunction with Disperse Dye
[0196] Commercial samples of the non-metallised acid dye Erionyl Red A-2BF and Duractive Black B were dissolved in acetone along with with crude Teratop Yellow HL-G. Samples of PES fabric, wool fabric and cotton fabric were dyed at 85.degree. C. using the controlled precipitation dyeing method shown in FIG. 3, employing 4 additions of water (1:10 LR in total), the total dyeing time being 20 mins.
[0197] The cotton fabric component was dyed a blue colour, the wool fabric was dyed a red colour and the polyester fabric component was dyed a yellow colour.
[0198] The results showed that PES, wool and cotton fibres can be dyed different colours simultaneously in the same dyebath in 20 minutes at 85.degree. C., using a mixture of disperse dye, reactive dye and non-metallised acid dye in the absence of all dyeing auxiliaries.
Example 8--One Pot Dyeing of a Solid Object
[0199] This example describes the results obtained from dyeing 3D printed nylon 12 (PA12) with a disperse dye (Dianix Blue-ACE) at the boil (98.degree. C.) under atmospheric pressure, using two different solvent systems. The dyed samples obtained were compared in terms of depth of shade and the process conditions employed (i.e. temperature, solvent system employed).
[0200] The substrate dyed were white solid, 3D printed nylon 12 parts.
[0201] The dye used in this trial was of commercial grade and was used as supplied without purification; Dianix Blue ACE manufactured by Dystar.
[0202] Photographs of all samples were recorded in a light cabinet under D.sub.65 illuminant, using a Samsung Galaxy S6+ mobile phone camera.
[0203] All dyeing was carried-out by placing the dye, substrate and solvent in a container placed on a hot plate and heated to the required processing temperature. The temperature of the dyebath was measured using a mercury-in-glass thermometer.
Solvent System 1 (Water)
[0204] A 2% omf (Dianix Blue-ACE) dyeing was produced following the procedure shown in FIG. 12A, employing a 10:1 LR. At the end of dyeing, the sample was rinsed thoroughly under running tap water and dried in the open air. A photograph of the dyed sample is shown in FIG. 12B
Solvent System 2 (Water:Glycerol; 80:20)
[0205] A 2% omf (Dianix Blue-ACE) was produced following the procedure shown in FIG. 12C, employing a 10:1 LR (liquor comprising of water:glycerol; 80:20) At the end of dyeing, the samples was rinsed thoroughly under running tap water and dried in the open air. A photograph of the dyed sample is shown in FIG. 12D
[0206] Upon visual inspection of the dyed samples it was apparent that, the sample obtained using the glycerol: water solvent system had the highest depth of shade (compare FIG. 12B and FIG. 12D).
* * * * *

D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.