Chemical Treatment Steel Sheet And Method For Manufacturing Chemical Treatment Steel Sheet
TANI; Yoshiaki ; et al.
U.S. patent application number 16/086152 was filed with the patent office on 2020-04-23 for chemical treatment steel sheet and method for manufacturing chemical treatment steel sheet. This patent application is currently assigned to NIPPON STEEL & SUMITOMO METAL CORPORATION. The applicant listed for this patent is NIPPON STEEL & SUMITOMO METAL CORPORATION. Invention is credited to Shigeru HIRANO, Makoto KAWABATA, Akira TACHIKI, Yoshiaki TANI, Morio YANAGIHARA, Hirokazu YOKOYA.
| Application Number | 20200123661 16/086152 |
| Document ID | / |
| Family ID | 59900119 |
| Filed Date | 2020-04-23 |


| United States Patent Application | 20200123661 |
| Kind Code | A1 |
| TANI; Yoshiaki ; et al. | April 23, 2020 |
CHEMICAL TREATMENT STEEL SHEET AND METHOD FOR MANUFACTURING CHEMICAL TREATMENT STEEL SHEET
Abstract
A chemical treatment steel sheet includes a steel sheet, an Fe--Sn alloy layer which is formed on at least one surface of the steel sheet, a Sn layer which is formed on the Fe--Sn alloy layer and contains Sn in which a total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer is 0.10 to 30.0 g/m.sup.2, and a chemical treatment layer which is formed on the Sn layer and contains a Zr compound in which an amount of Zr contained therein is 1.0 to 150 mg/m.sup.2, a phosphate compound in an amount of P contained therein is 1.0 to 100 mg/m.sup.2 and an Al compound in an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2.
| Inventors: | TANI; Yoshiaki; (Tokyo, JP) ; HIRANO; Shigeru; (Tokyo, JP) ; TACHIKI; Akira; (Tokyo, JP) ; YANAGIHARA; Morio; (Tokyo, JP) ; KAWABATA; Makoto; (Tokyo, JP) ; YOKOYA; Hirokazu; (Tokyo, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | NIPPON STEEL & SUMITOMO METAL
CORPORATION Tokyo JP |
||||||||||
| Family ID: | 59900119 | ||||||||||
| Appl. No.: | 16/086152 | ||||||||||
| Filed: | March 22, 2016 | ||||||||||
| PCT Filed: | March 22, 2016 | ||||||||||
| PCT NO: | PCT/JP2016/058905 | ||||||||||
| 371 Date: | September 18, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C25D 7/0614 20130101; C25D 5/48 20130101; C25D 3/30 20130101; C25D 11/36 20130101; C25D 11/00 20130101; C25D 9/08 20130101; C21D 9/46 20130101; C23C 2/08 20130101; C25D 9/10 20130101; C23C 28/00 20130101; C23C 28/021 20130101; C25D 5/12 20130101; C23C 22/361 20130101; C23C 22/20 20130101; C25B 9/08 20130101 |
| International Class: | C23C 22/36 20060101 C23C022/36; C23C 2/08 20060101 C23C002/08; C25D 3/30 20060101 C25D003/30; C23C 28/02 20060101 C23C028/02; C25D 5/48 20060101 C25D005/48; C21D 9/46 20060101 C21D009/46 |
Claims
1. A chemical treatment steel sheet comprising: a steel sheet; an Fe--Sn alloy layer which is formed on at least one surface of the steel sheet; a Sn layer which is formed on the Fe--Sn alloy layer and contains Sn in which a total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer is 0.10 to 30.0 g/m.sup.2; and a chemical treatment layer which is formed on the Sn layer and contains a Zr compound in which an amount of Zr contained therein is 1.0 to 150 mg/m.sup.2, a phosphate compound in which an amount of P contained therein is 1.0 to 100 mg/m.sup.2, and an Al compound in which an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2.
2. The chemical treatment steel sheet according to claim 1, wherein the chemical treatment layer contains Al.sub.2O.sub.3 in which an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2.
3. The chemical treatment steel sheet according to claim 1, wherein the chemical treatment layer contains the Zr compound in which an amount of Zr contained therein is 1.0 to 120 mg/m.sup.2; the phosphate compound in which an amount of P contained therein is 2.0 to 70.0 mg/m.sup.2; and the Al compound in which an amount of Al contained therein is 0.20 to 20.0 mg/m.sup.2.
4. The chemical treatment steel sheet according to claim 1, wherein the total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer is 0.30 to 20.0 g/m.sup.2.
5. The chemical treatment steel sheet according to claim 1, wherein a surface of the chemical treatment layer is not covered with a film or a coating material.
6. A method for manufacturing a chemical treatment steel sheet, the method comprising: a plating process of forming a Sn-plated layer containing Sn in which an amount of Sn contained therein is 0.10 to 30.0 g/m.sup.2 on a surface of a steel sheet; a reflow treatment process of forming an Fe--Sn alloy layer and a Sn layer by performing reflow treatment on the Sn-plated layer; and an electrolytic treatment process of forming a chemical treatment layer on the Sn layer by performing an electrolytic treatment under conditions of a current density of 1.0 to 100 A/dm.sup.2 and an electrolytic treatment time of 0.20 to 150 seconds using a chemical treatment solution having a temperature of 5.degree. C. or higher and lower than 90.degree. C., wherein the chemical treatment solution contains 10 to 20,000 ppm of Zr ions, 10 to 20,000 ppm of F ions, 10 to 3,000 ppm of phosphate ions, 100 to 30,000 ppm of nitrate ions and sulfate ions in total, and 500 to 5,000 ppm of Al ions, and (NH.sub.4).sub.3AlF.sub.6 is used as a supply source of Al ions.
7. The method for manufacturing a chemical treatment steel sheet according to claim 6, wherein the chemical treatment solution contains: 200 to 17,000 ppm of the Zr ions; 200 to 17,000 ppm of the F ions; 100 to 2,000 ppm of the phosphate ions; 1000 to 23000 ppm of the nitrate ions and the sulfate ions in total; and 500 to 3000 ppm of the Al ions.
Description
TECHNICAL FIELD OF THE INVENTION
[0001] The present invention relates to a chemical treatment steel sheet and a method for manufacturing a chemical treatment steel sheet.
BACKGROUND ART
[0002] Corrosion is occurred when metals are continuously used in some cases. Various techniques have been proposed to prevent corrosion of metals. Examples of the proposed techniques include a technique of applying plating to a metal plate or a technique of performing various surface treatments on the surface of a metal plate of a plated surface.
[0003] For example, Patent Document 1 describes a technique of forming an organic resin film including a vanadium compound or at least one of a phosphate and phosphate-based compound, a silane compound having at least one of an epoxy group and an amino group, and an organic resin including at least one of a water-soluble organic resin and a water-dispersible organic resin as main components on a surface of an Al--Zn-based alloy plated steel sheet used for building materials and home appliances.
[0004] On the other hand, when metal containers for the purpose of preserving beverages or foods are manufactured, Ni-plated steel sheets, Sn-plated steel sheets, Sn-based alloy plated steel sheets, or the like have been used. The Al--Zn-based alloy plated steel sheet described in Patent Document 1 is a so-called sacrificial protection steel sheet, whereas a Ni-plated steel sheet, a Sn-plated steel sheet, or a Sn-based alloy plated steel sheet is a so-called barrier plated steel sheet.
[0005] When a Ni-plated steel sheet, a Sn-plated steel sheet, or a Sn-based alloy plated steel sheet is used as a steel sheet for a metal container for the purpose of preserving beverages or foods (hereinafter referred to as a "steel sheet for containers"), the surface of the plated steel sheet is subjected to a chemical treatment using hexavalent chromium to secure adhesiveness and corrosion resistance between the steel sheet and a coating or a film in many cases. A chemical treatment using a solution containing a hexavalent chromium is referred to as a chromate treatment.
[0006] However, since hexavalent chromium used in a chromate treatment is harmful to the environment, a chemical treatment film such as a Zr-phosphate film has been developed as a replacement for the chromate treatment applied to a steel sheet for containers in the related art. For example, Patent Document 2 describes a steel sheet for containers having a chemical treatment film including Zr, a phosphate, a phenolic resin, and the like.
[0007] Examples of foods preserved in a metal container using a steel sheet for containers include meat, vegetables, and the like. Meat and vegetables contain various proteins, but these proteins contain amino acids containing sulfur (sulfur-containing amino acids represented by L-cysteine, L-methionine, and L-(-)-cystine) in some cases. When foods containing sulfur-containing amino acids is heated during sterilization, S in the sulfur-containing amino acids binds to Sn, Fe, or the like in a steel sheet for containers, resulting in black discoloration. This phenomenon is referred to as "sulfide stain." Since the appearance of the inner surface of a metal container deteriorates when sulfide stain occurs, countermeasures have been sought to prevent the occurrence of sulfide stain.
[0008] In addition, Patent Document 3 describes a method for manufacturing a steel sheet for containers in which a Zr-containing film is formed on a surface of a steel sheet by immersing the steel sheet or performing an electrolytic treatment on the steel sheet in a solution containing Zr ions, F ions, and at least one reaction accelerating component selected from the group consisting of Al ions, boric acid ions, Cu ions, Ca ions, Al metal, and Cu metal.
CITATION LIST
Patent Documents
[0009] [Patent Document 1]
[0010] Japanese Unexamined Patent Application, First Publication No. 2005-290535
[0011] [Patent Document 2]
[0012] Japanese Unexamined Patent Application, First Publication No. 2007-284789
[0013] [Patent Document 3]
[0014] Japanese Unexamined Patent Application, First Publication No. 2012-62521
SUMMARY OF INVENTION
Problems to be Solved by the Invention
[0015] Since a film formed through a chromate treatment (hereinafter referred to as a "chromate film") is dense even when an adhered amount of film is small, a steel sheet for containers having a chromate film formed on its surface has excellent corrosion resistance and sulfide stain resistance. However, since hexavalent chromium is harmful to the environment as described above, a steel sheet for containers should preferably not contain hexavalent chromium as far as possible.
[0016] On the other hand, the organic resin film described in Patent Document 1 and the chemical treatment film described in Patent Document 2 do not contain hexavalent chromium and thus are appropriate for the environment. However, in the organic resin film described in Patent Document 1 and the chemical treatment film described in Patent Document 2, it is necessary to increase an adhered amount of film to form a dense film which can obtain appropriate sulfide stain resistance. An increase in adhered amount of film is not preferable because, when an adhered amount of film is increased, the adhesion between the film and a plated layer under the film decreases and the weldability of the chemical treatment steel sheet decreases, which is not preferable. Furthermore, an increase in adhered amount of film is not economically preferable.
[0017] In the method for manufacturing a steel sheet for containers described in Patent Document 3, the Al content in the chemical treatment film is small. Thus, it is difficult to obtain an appropriate sulfide stain resistance in some cases.
[0018] The present invention was made in view of the above-described circumstances and an objective of the present invention is to provide a chemical treatment steel sheet which has excellent corrosion resistance and sulfide stain resistance even when an amount of chemical treatment layer adhered is small and a method for manufacturing the same.
Means for Solving the Problem
[0019] The present invention employs the following means to solve the above-described problems and achieve the above objective.
[0020] (1) A chemical treatment steel sheet according to an aspect of the present invention includes a steel sheet, an Fe--Sn alloy layer which is formed on at least one surface of the steel sheet, a Sn layer which is formed on the Fe--Sn alloy layer and contains Sn in which an total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer is 0.10 to 30.0 g/m.sup.2, and a chemical treatment layer which is formed on the Sn layer and contains a Zr compound in which an amount of Zr contained therein is 1.0 to 150 mg/m.sup.2, a phosphate compound in which an amount of P contained therein is 1.0 to 100 mg/m.sup.2 and an Al compound in which an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2.
[0021] (2) In the chemical treatment steel sheet according to (1) above, the chemical treatment layer may contain Al.sub.2O.sub.3 in which an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2.
[0022] (3) In the chemical treatment steel sheet according to (1) or (2) above, the chemical treatment layer may contain the Zr compound in which an amount of Zr contained therein is 1.0 to 120 mg/m.sup.2, the phosphate compound in which an amount of P contained therein is 2.0 to 70.0 mg/m.sup.2, and the Al compound in which an amount of Al contained therein may be 0.20 to 20.0 mg/m.sup.2.
[0023] (4) In the chemical treatment steel sheet described according to any aspect of (1) to (3), the total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer may be 0.30 to 20.0 g/m.sup.2.
[0024] (5) In the chemical treatment steel sheet described according to any aspect of (1) to (4), a surface of the chemical treatment layer may not be covered with a film or a coating material.
[0025] (6) A method for manufacturing a chemical treatment steel sheet according to an aspect of the present invention includes a plating process of forming a Sn-plated layer containing Sn in which an amount of Sn contained therein is 0.10 to 30.0 g/m.sup.2 on a surface of a steel sheet, a reflow treatment process of forming an Fe--Sn alloy layer and a Sn layer by performing reflow treatment on the Sn-plated layer, and an electrolytic treatment process of forming a chemical treatment layer on the Sn layer by performing electrolytic treatment under conditions of a current density of 1.0 to 100 A/dm.sup.2 and an electrolytic treatment time of 0.20 to 150 seconds using a chemical treatment solution having a temperature of 5.degree. C. or higher and lower than 90.degree. C. The chemical treatment solution contains 10 to 20,000 ppm of Zr ions, 10 to 20,000 ppm of F ions, 10 to 3000 ppm of phosphate ions, 100 to 30,000 ppm of nitrate ions and sulfate ions in total, and 500 to 5,000 ppm of Al ions, and may be used (NH.sub.4).sub.3AlF.sub.6 as a supply source of Al ions.
[0026] (7) In the method for manufacturing a chemical treatment steel sheet according to (6) above, the chemical treatment solution may contain 200 to 17,000 ppm of the Zr ions, 200 to 17,000 ppm of the F ions, 100 to 2.000 ppm of the phosphate ions, 1000 to 23,000 ppm of the nitrate ions and the sulfate ions in total, and 500 to 3,000 ppm of the Al ions.
Effects of the Invention
[0027] According to each aspect described above, a chemical treatment steel sheet having an excellent corrosion resistance and sulfide stain resistance even in a case in which an adhered amount of a chemical treatment layer is small and a method for manufacturing the chemical treatment steel sheet can be provided.
BRIEF DESCRIPTION OF THE DRAWING(S)
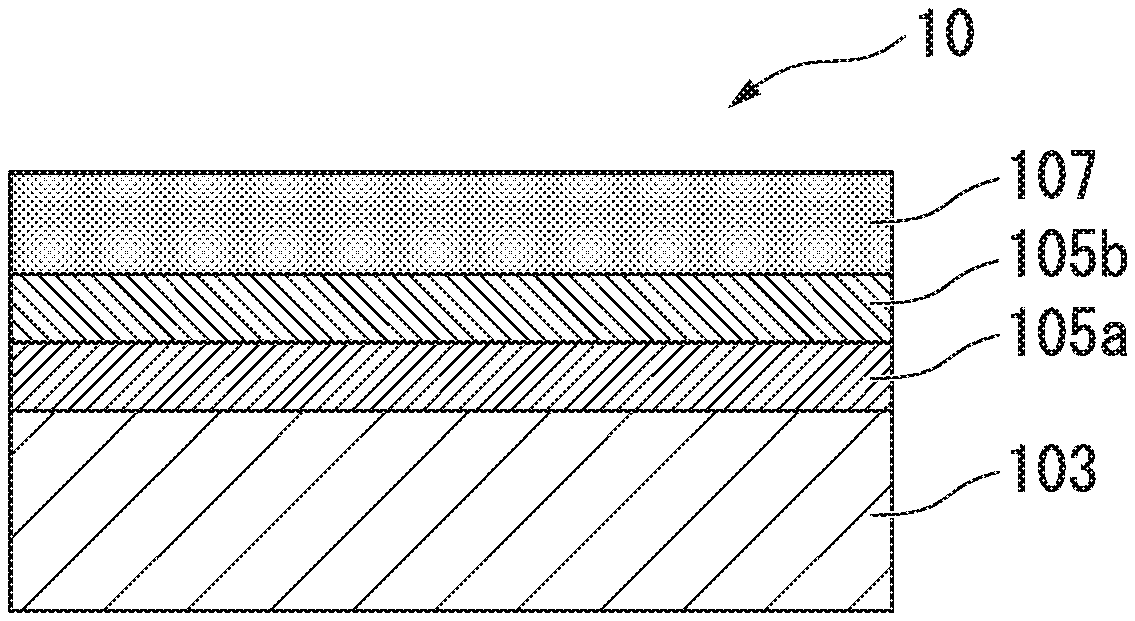
[0028] FIG. 1A is a schematic diagram showing a chemical treatment steel sheet in which a Fe--Sn alloy layer, a Sn layer, and a chemical treatment layer are formed on one surface of a steel sheet.
[0029] FIG. 1B is a schematic diagram showing a chemical treatment steel sheet in which Fe--Sn alloy layers, Sn layers, and chemical treatment layers are formed on both surfaces of a steel sheet.
[0030] FIG. 2 is a flowchart showing an example of a flow of a method for manufacturing a chemical treatment steel sheet.
[0031] FIG. 3 is a graph showing results of Example 1.
EMBODIMENTS OF THE INVENTION
[0032] Exemplary embodiments of the present invention will be described in detail below with reference to the appended drawings. Note that, in the present embodiments, repeated description will be omitted by giving the same reference symbols to constituent elements having similar configurations.
<Regarding Configuration of Chemical Treatment Steel Sheet>
[0033] First, a configuration of a chemical treatment steel sheet according to an embodiment will be described in detail with reference to FIG. 1A and FIG. 1B. FIG. 1A and FIG. 1B are explanatory diagrams schematically showing layer structures of the chemical treatment steel sheet according to the present embodiment.
[0034] The chemical treatment steel sheet 10 according to the present embodiment has steel sheets 103, Fe--Sn alloy layers 105a, Sn layers 105b, and chemical treatment layers 107 as shown in FIG. 1A and FIG. 1B.
(Regarding Steel Sheet 103)
[0035] The steel sheet 103 is used as a base material for the chemical treatment steel sheet 10 according to the present embodiment. The steel sheet 103 used in the present embodiment is not particularly limited, and a known steel sheet 103 that is used as a steel sheet for containers can be used. A method for manufacturing the steel sheet 103 and a material thereof are not particularly limited either, and a steel sheet 103 produced through, beginning from a general steel piece production process, a known process such as hot rolling, pickling, cold rolling, annealing, or temper rolling can be used.
[0036] A thickness of the steel sheet 103 is preferably 0.05 to 1 mm in consideration of practicability and economic efficiency when it is used as a steel sheet for containers.
[Regarding Fe--Sn Alloy Layer 105a and Sn Layer 105b]
[0037] The Fe--Sn alloy layer 105a and the Sn layer 105b containing Sn are formed on a surface of the steel sheet 103. The Fe--Sn alloy layer 105a and the Sn layer 105b are barrier plated layers. A barrier plated layer is a plated layer in which the corrosion of the steel sheet 103 is suppressed by preventing a cause of corrosion from acting on the base material by means of forming a metal film of Sn on the surface of the steel sheet 103 using Sn which are metals more electrochemically noble than Fe constituting the steel sheet 103 which is the base material.
[0038] On the other hand, a sacrificial protection layer has a function opposite to that of a barrier plated layer. In a sacrificial protection layer, the corrosion of the steel sheet 103 is suppressed by corroding a metal such as Zn constituting the plated layer earlier than Fe constituting the steel sheet 103 by mean of forming a metal film on the surface of the steel sheet 103 using a metal less electrochemically noble than Fe constituting the steel sheet 103 serving as the base material (for example Zn as in Patent Document 1). The interaction between the barrier plated layer and the chemical treatment layer 107 is different from that between the sacrificial protection layer and the chemical treatment layer 107.
[0039] Specific examples of the Fe--Sn alloy layer 105a and the Sn layer 105b according to the present embodiment will be described below with reference to FIG. 1A and FIG. 1B.
[0040] Note that the Fe--Sn alloy layer 105a, the Sn layer 105b, and the chemical treatment layer 107 may be formed on one surface of the steel sheet 103 as shown in FIG. 1A, and the Fe--Sn alloy layers 105a, the Sn layers 105b, and the chemical treatment layer 107 may be formed on both surfaces of the steel sheet 103 as shown in FIG. 1B.
[0041] The Fe--Sn alloy layer 105a is formed on the surface of the steel sheet 103, and the Sn layer 105b is formed on the Fe--Sn alloy layer 105a as shown in FIG. 1A and FIG. 1B. Although details will be described below, the Fe--Sn alloy layer 105a and the Sn layer 105b are formed by forming a Sn-plated layer (not shown) on the surface of the steel sheet 103 and then performing reflow treatment (reflow treatment) thereon.
[0042] The Sn-plated layer (not shown) is formed to secure corrosion resistance and weldability of the chemical treatment steel sheet 10. Regarding Sn, not only does Sn itself having high corrosion resistance, but Fe--Sn alloys contained in the Fe--Sn alloy layer 105a also have excellent corrosion resistance and weldability.
[0043] Although both the Fe--Sn alloy layer 105a and the Sn layer 105b according to the present embodiment contain Sn, the total amount of Sn contained in the Fe--Sn alloy layer 105a and the Sn layer 105b is 0.10 to 30.0 g/m.sup.2 per one surface.
[0044] Sn has excellent processability, weldability, and corrosion resistance, and by performing the reflow treatment after Sn plating, corrosion resistance of the chemical treatment steel sheet 10 can be further improved, and a surface appearance (mirror appearance) of the chemical treatment steel sheet 10 can be made more preferable. In order to obtain the above-described effects, it is necessary for the total amount of Sn contained in the Fe--Sn alloy layer 105a and the Sn layer 105b be 0.10 g/m.sup.2 per one surface.
[0045] In addition, when the amount of Sn increases further, processability, weldability, and corrosion resistance of the chemical treatment steel sheet 10 are improved, but if the total amount of Sn in the Fe--Sn alloy layer 105a and the Sn layer 105b exceeds 30.0 g/m.sup.2 per one surface, the above-described effects from Sn are saturated. In addition, if the total amount of Sn in the Fe--Sn alloy layer 105a and the Sn layer 105b exceeds 30 g/m.sup.2 per one surface, it is not economically preferable. For the above-described reasons, the total amount of Sn contained in the Fe--Sn alloy layer 105a and the Sn layer 105b is set to be 30.0 g/m.sup.2 or less per one surface.
[0046] It is preferable for the total amount of Sn in the Fe--Sn alloy layer 105a and the Sn layer 105b be 0.30 g/m.sup.2 to 20.0 g/m.sup.2 per one surface. When the total amount of Sn in the Fe--Sn alloy layer 105a and the Sn layer 105b is 0.30 g/m.sup.2 or more per one surface, the above-described effects from Sn can be obtained more reliably. In addition, when the total amount of Sn in the Fe--Sn alloy layer 105a and the Sn layer 105b is 20.0 g/m.sup.2 or less per one surface, production costs can be reduced further.
[0047] The Fe--Sn alloy layer 105a contains Fe in which an amount of Fe contained therein is 0.0010 to 100 g/m.sup.2. In addition, the Fe--Sn alloy layer 105a may contain trace elements and inevitable impurities that are incorporated thereinto during a production process or the like, in addition to Sn and Fe.
[0048] In the Fe--Sn alloy layer 105a, a total amount of Fe metal and Sn metal contained is 50 mass % or more. In the Fe--Sn alloy layer 105a, a total amount of Fe metal and Sn metal to be contained is preferably 70 mass % or more.
[0049] The Sn layer 105b may contain only Sn, or may contain Fe in which an amount of Fe contained therein is 0.0010 to 6.0 g/m.sup.2. In addition, the Sn layer 105b may contain trace elements and inevitable impurities that are incorporated thereinto during a production process or the like.
[0050] In addition, the amount of Sn contained in the Sn layer 105b is 50 mass % or higher. The amount of Sn contained in the Sn layer 105b is preferably 70 mass % or higher.
[0051] A ratio between thicknesses of the Fe--Sn alloy layer 105a and the Sn layer 105b is not particularly limited, and any ratio is possible as long as the above-described amount of Sn is secured.
[0052] However, when the steel sheet 103 having the surface on which the Fe--Sn alloy layer 105a and the Sn layer 105b are formed is used as a steel sheet for containers, it is difficult to prevent sulfide stain even if a film is laminated on a surface of the Sn layer 105b or a coating material is applied thereon. It is thought that S contained in contents such as beverages and foods binds to Sn and that black SnS, SnS.sub.2 or the like is formed.
[0053] Note that S is contained in beverages and food as a constituent element of S-containing amino acids such as L-cysteine, L-(-)-cystine, and L-methionine.
[0054] In addition, in a case where the Fe--Sn alloy layer 105a and the Sn layer 105b are formed not to be dense, part of the steel sheet 103 as a base material is exposed. In such a case, there are cases in which Fe contained in the steel sheet 103 bonds with S contained in a beverage, food, or the like, and black FeS, Fe.sub.2S.sub.3, and Fe.sub.2S are formed.
[0055] In order to reduce the blackening caused by SnS, SnS.sub.2, FeS, Fe.sub.2S.sub.3, Fe.sub.2S, or the like, a chromate film has previously mainly been formed on the surfaces of the Fe--Sn alloy layer 105a and the Sn layer 105b.
[0056] In order to improve sulfide stain resistance, the chemical treatment layer 107 containing a Zr compound, a phosphate compound, and an Al compound is formed on the surfaces of the Fe--Sn alloy layer 105a and the Sn layer 105b as a substitute for the conventional chromate film in the chemical treatment steel sheet 10 according to the present embodiment.
[Regarding Chemical Treatment Layer 107]
[0057] The chemical treatment layers 107 are formed on the surfaces of the Sn layer 105b as shown in FIG. 1A and FIG. 1B. The chemical treatment layer 107 is a composite film layer having the Zr compound as a main component, containing the Zr compound in which an amount of Zr contained therein is 1.0 to 150 mg/m.sup.2 per one surface, the phosphate compound in which an amount of P contained therein is 1.0 to 100 mg/m.sup.2 per one surface, and the Al compound in which an amount of Al contained therein is 0.10 to 30.0 mg/m.sup.2 per one surface.
[0058] Note that, in the present embodiment, a composite film layer indicates a film layer in which a Zr compound, a phosphate compound, and an Al compound are in a partially mixed state without being fully mixed.
[0059] In a case where three films such as a Zr film containing the Zr compound, a phosphate film containing the phosphate compound, and an Al film containing the Al compound are formed to be stacked on the Sn layer 105b, although a certain degree of effects relating to corrosion resistance and adhesiveness is obtained, they are practically insufficient. However, if the Zr compound, the phosphate compound, and the Al compound are partially mixed in the chemical treatment layer 107 as described in the present embodiment, better corrosion resistance and adhesiveness can be obtained than in the case in which the three films are formed to be stacked as described above.
[0060] The Zr compound contained in the chemical treatment layer 107 according to the present embodiment has a function of improving corrosion resistance, adhesiveness, and process adhesiveness. As the Zr compound according to the present embodiment, a Zr oxide, a Zr phosphate, a Zr hydroxide, a Zr fluoride, and the like are exemplary examples, and the chemical treatment layer 107 may contain multiple above-described Zr compounds. A preferable combination with the Zr compound is a Zr oxide, a Zr phosphate, and a Zr fluoride.
[0061] In a case where the amount of Zr contained in the chemical treatment layer 107 is 1.0 mg/m.sup.2 or more per one surface, practically preferable corrosion resistance, adhesiveness, and process adhesiveness are secured.
[0062] Meanwhile, as the amount of Zr in the Zr compound increases, corrosion resistance, adhesiveness, and process adhesiveness are improved accordingly. However, if the amount of Zr contained in the Zr compound exceeds 150 mg/m.sup.2 per one surface, the chemical treatment layer 107 becomes excessively thick, and due to the main cause of a cohesive fracture, adhesiveness of the chemical treatment layer 107 to the Sn layer 105b deteriorates, electrical resistance increases, and weldability is degraded. In addition, if the amount of Zr contained in the Zr compound exceeds 150 mg/m.sup.2, adhesion of the chemical treatment layer 107 becomes uneven, which may cause an irregular appearance.
[0063] Thus, the amount of Zr contained in the Zr compound (i.e., the amount of Zr) of the chemical treatment layer 107 according to the present embodiment is set to 1.0 mg/m.sup.2 to 150 mg/m.sup.2 per one surface. The amount of Zr contained in the Zr compound is preferably 1.0 mg/m.sup.2 to 120 mg/m.sup.2 per one surface. By setting the amount to 120 g/m.sup.2 per one surface, the production costs of the chemical treatment layer 107 can be reduced further.
[0064] The chemical treatment layer 107 further contains one or more types of phosphate compounds in addition to the above-described Zr compounds.
[0065] A phosphate compound according to the present embodiment has a function of improving corrosion resistance, adhesiveness, and process adhesiveness. As the phosphate compound according to the present embodiment, Fe phosphate, Ni phosphate, Sn phosphate, Zr phosphate, Al phosphate, and the like which are formed when phosphate ions react with compounds contained in the steel sheet 103, the Fe--Sn alloy layer 105a, the Sn layer 105b, and the chemical treatment layer 107 are exemplary examples. The chemical treatment layer 107 may contain one or more types of the above-described phosphate compounds.
[0066] When the amount of phosphate compounds contained in the chemical treatment layer 107 is greater, corrosion resistance, adhesiveness, and process adhesiveness of the chemical treatment steel sheet 10 are improved. Specifically, in a case where the amount of P contained in the phosphate compound contained in the chemical treatment layer 107 is 1.0 mg/m.sup.2 or more, practically preferable corrosion resistance, adhesiveness, and process adhesiveness are secured.
[0067] Although corrosion resistance, adhesiveness, and process adhesiveness are improved as the amount of phosphate compound increases, if the amount of P contained in the phosphate compound exceeds 100 mg/m.sup.2 per one surface, the chemical treatment layer 107 becomes excessively thick, and thus mainly due to a cohesive fracture, adhesiveness of the chemical treatment layer 107 to the Sn layer 105b deteriorates, electrical resistance increases, and thus weldability is degraded. In addition, if the amount of P contained in the phosphate compound exceeds 100 mg/m.sup.2, adhesion of the chemical treatment layer 107 becomes uneven, which may cause an irregular appearance.
[0068] Thus, the amount of P contained in the phosphate compound in the chemical treatment layer 107 according to the present embodiment is set to 1.0 to 100 mg/m.sup.2 per one surface.
[0069] The amount of P contained in the phosphate compound in the chemical treatment layer 107 is preferably 2.0 to 70.0 mg/m.sup.2 per one surface. By setting the amount of P contained in the phosphate compound in the chemical treatment layer 107 to 2.0 mg/m.sup.2 or more per one surface, more preferable sulfide stain resistance can be obtained. In addition, by setting the amount of P contained in the phosphate compound of the chemical treatment layer 107 to 70.0 mg/m.sup.2 or less per one surface, the production costs of the chemical treatment layer 107 can be reduced further.
[0070] The chemical treatment layer 107 further contains the Al compound in addition to the above-described Zr compound and phosphate compound. The Al compound of the chemical treatment layer 107 exists mainly as Al oxide in the chemical treatment layer 107. By reinforcing a film defect of the chemical treatment layer 107 having Zr as a main component with the Al oxide, the chemical treatment steel sheet 10 can have excellent sulfide stain resistance.
[0071] Since the chemical treatment layer 107 having Zr as a main component is inherently a very uniform film, the amount of Al contained in the Al compound added to the chemical treatment layer 107 to reinforce the film defect may be 0.10 mg/m.sup.2 or more per one surface. By setting the amount of Al contained in the Al compound to 0.10 mg/m.sup.2 or more per one surface, sulfide stain resistance of the chemical treatment steel sheet 10 can be satisfactorily improved.
[0072] Meanwhile, although sulfide stain resistance is improved when the amount of Al compound in the chemical treatment layer 107 increases, if the amount of Al contained in the Al compound exceeds 30.0 mg/m.sup.2 per one surface, sulfide stain resistance is saturated, which is not economically preferable. Thus, the amount of Al contained in the Al compound contained in the chemical treatment layer 107 is set to 30.0 mg/m.sup.2 or less per one surface.
[0073] The amount of Al contained in the Al compound in the chemical treatment layer 107 is more preferably 0.20 to 20.0 mg/m.sup.2 per one surface. By setting the amount of Al contained in the Al compound to 0.20 mg/m.sup.2 or more per one surface, sulfide stain resistance can be satisfactorily improved. In addition, by setting the amount of Al contained in the Al compound to 20.0 mg/m.sup.2 or less per one surface, the production costs of the chemical treatment layer 107 can be reduced further.
[0074] The amount of Al contained in the Al oxide (Al.sub.2O.sub.3) contained in the chemical treatment layer 107 is preferably 0.10 to 30.0 mg/m.sup.2. When the amount of Al oxide contained in the chemical treatment layer 107 is within the above-described range, a film defect of the chemical treatment layer 107 can be satisfactorily reinforced and excellent sulfide stain resistance can be obtained.
[0075] In addition, by causing the Al compound to be contained in the chemical treatment layer 107, the amount of phosphate compound for improving sulfide stain resistance, like Al, can be reduced.
[0076] Phosphoric acid Zr included in the phosphate compound contained in the chemical treatment layer 107, which is generated from reaction of phosphoric acid ions with Zr ions, is precipitated when it exists in a large amount in a chemical treatment solution for forming the chemical treatment layer 107, and the chemical treatment solution becomes clouded.
[0077] Here, the Al compound contributes to improvement of sulfide stain resistance more than the phosphate compound. Thus, since the chemical treatment layer 107 contains the Al compound, the amount of phosphate compound, which is the cause of cloudiness of the chemical conversion solution, can be reduced while satisfactorily improving the sulfide stain resistance.
[0078] In addition, when the amount of phosphate compound is reduced, it is possible to reduce an amount of F ions which inhibit the binding between Zr and phosphates and the binding between Al and phosphates. As a result, since Zr is precipitated more easily, it is possible to improve the electrolysis efficiency for forming the chemical treatment layer 107.
[0079] Note that the chemical treatment layer 107 may contain inevitable impurities that are incorporated thereinto during the production process or the like, in addition to the above-described Zr compound, phosphate compound, and Al compound. In addition, in a case where the chemical treatment layer 107 contains Cr, the upper limit of the amount of Cr is 2 mg/m.sup.2.
[0080] The chemical treatment steel sheet 10 according to the present embodiment exhibits excellent sulfide stain resistance even if the adhered amount of the chemical treatment layer 107 is reduced.
[0081] For example, a coating material may be applied to a surface of the chemical treatment steel sheet 10, which is then baked, which results in forming a coating film. The chemical treatment steel sheet 10 having the coating film formed on the surface is placed to be fixed to the mouth of a heat resistant bottle holding 0.6 mass % of an L-cysteine solution that has been boiled for one hour as a lid, and is subjected to a heat treatment using a soaking furnace at 110.degree. C. for 30 minutes or the like. When the appearance of the portion of the above-described heat-treated chemical treatment steel sheet 10 in contact with the heat resistance bottle is observed, blackening does not occur in 50% or more of the area of the contact portion in a case where the chemical treatment steel sheet 10 according to the present embodiment is used.
[0082] As described above, the chemical treatment steel sheet 10 according to the present embodiment has excellent corrosion resistance and sulfide stain resistance. Thus, even in a case where the surface of the chemical treatment layer 107 is not covered with a film or a coating material, the chemical treatment steel sheet 10 can be used as a steel sheet for containers.
<Regarding Layer Structure of Chemical Treatment Steel Sheet 10>
[0083] The chemical treatment steel sheet 10 has the Fe--Sn alloy layer 105a, the Sn layer 105b, and the chemical treatment layer 107 on the steel sheet 103 as described above. That is, in the chemical treatment steel sheet 10, the steel sheet 103 is in contact with the Fe--Sn alloy layer 105a, and there is no other layer between the steel sheet 103 and the Fe--Sn alloy layer 105a. Likewise, the Fe--Sn alloy layer 105a is in contact with the Sn layer 105b, and there is no other layer between the Fe--Sn alloy layer 105a and the Sn layer 105b. Furthermore, the Sn layer 105b is in contact with chemical treatment layer 107, and there is no other layer between the Sn layer 105b and the chemical treatment layer 107.
<Regarding Measurement Method for Amount of Component>
[0084] The amount of Sn and the amount of Fe contained in the Fe--Sn alloy layer 105a and the Sn layer 105b can be measured using, for example, a fluorescent X-ray method. In this case, using known samples for the amount of Sn or the amount of Fe, a calibration curve for the amount of Sn or the amount of Fe is created in advance, and the amount of Sn or the amount of Fe is determined relatively using the created calibration curve.
[0085] The amount of Zr, the amount of P, and the amount of Al contained in the chemical treatment layer 107 can be measured using a quantitative analysis method, for example, fluorescent X-ray analysis, or the like. In addition, which compounds exist in the chemical treatment layer 107 can be specified by performing analysis using X-ray photoelectron spectroscopy (XPS).
[0086] In addition, with regard to the amount of Al.sub.2O.sub.3 contained in the chemical treatment layer 107, a peak intensity ratio of Al.sub.2O.sub.3, Al metal, and other Al compound is first obtained using X-ray photoelectron spectroscopy (XPS). Further, the amount of Al.sub.2O.sub.3 contained in the chemical treatment layer 107 is calculated on the basis of the total Al metal amount obtained using the quantitative analysis method such as fluorescent X-ray analysis and the peak intensity ratio obtained using XPS as described above.
[0087] Note that a measurement method for each component is not limited to the above-described methods, and a known measurement method can be applied.
<Regarding Method for Manufacturing Chemical Treatment Steel Sheet>
[0088] Next, a method for manufacturing the chemical treatment steel sheet 10 according to the present embodiment will be described in detail with reference to FIG. 2. FIG. 2 is a flowchart for describing an example of a flow of a method for manufacturing the chemical treatment steel sheet 10 according to the present embodiment.
[Pre-Treatment Process]
[0089] In the method for manufacturing the chemical treatment steel sheet 10 according to the present embodiment, first, known pre-treatment is performed on the steel sheet 103 if necessary (Step S101).
[Plating Process]
[0090] Then, a Sn-plated layer (not shown) is formed on a surface of the steel sheet 103 (Step S103). A method for forming the Sn-plated layer (not shown) is not particularly limited, and a known electroplating method, a method of immersing the steel sheet 103 in molten Sn, or the like can be used.
[Reflow Treatment (Reflow Treatment Process) Process]
[0091] After the Sn-plated layer (not shown) is formed, reflow treatment (reflow treatment) is performed (Step S104). Accordingly, the Fe--Sn alloy layer 105a and the Sn layer 105b are formed on the surface of the steel sheet 103.
[0092] The reflow treatment is performed by forming the Sn-plated layer (not shown) on the steel sheet 103, then heating the steel sheet to a temperature of 200.degree. C. or higher to melt the Sn-plated layer (not shown) temporarily, and then quenching the steel sheet. Sn in the Sn-plated layer (not shown) located on the steel sheet 103 side is alloyed with Fe contained in the steel sheet 103 through the reflow treatment, whereby the Fe--Sn alloy layer 105a is formed, and the remaining Sn forms the Sn layer 105b.
[Electrolytic Treatment Process]
[0093] Then, the chemical treatment layer 107 is formed through cathodic electrolytic treatment (Step S105).
[0094] The chemical treatment layer 107 is formed through electrolytic treatment (e.g., cathodic electrolytic treatment). A chemical treatment solution used to form the chemical treatment layer 107 through electrolytic treatment contains 10 ppm or more and 20,000 ppm or less of Zr ions, 10 ppm or more and 20,000 ppm or less of F ions, 10 ppm or more and 3,000 ppm or less of phosphate ions, 100 ppm or more and 30,000 ppm or less of nitrate ions and sulfate ions in total, and 500 ppm or more and 5,000 ppm or less of Al ions. In addition, (NH.sub.4).sub.3AlF.sub.6 is used as a supply source of Al ions in the chemical treatment solution.
[0095] Note that nitrate ions and sulfate ions may be contained at 10 ppm or more and 3,000 ppm or less in total of both ions in the chemical treatment solution, both ions of nitrate ions and sulfate ions may be contained in the chemical treatment solution, and only one of nitrate ions and sulfate ions may be contained in the chemical treatment solution.
[0096] It is preferable for the chemical treatment solution to contain 200 ppm or more and 17,000 ppm or less of Zr ions, 200 ppm or more and 17,000 ppm or less of F ions, 100 ppm or more and 2,000 ppm or less of phosphate ions, 1,000 ppm or more and 23,000 ppm or less of nitrate ions and sulfate ions in total, and 500 ppm or more and 3,000 ppm or less of Al ions.
[0097] By setting the concentration of Zr ions to 200 ppm or more, it is possible to reliably prevent a Zr adhered amount from decreasing. In addition, by setting the concentration of F ions to 200 ppm or more, it is possible to more reliably prevent cloudiness of the chemical treatment layer 107 accompanying with precipitation of phosphate.
[0098] Likewise, by setting the concentration of phosphate ions to 100 ppm or more, it is possible to more reliably prevent cloudiness of the chemical treatment layer 107 accompanied by precipitation of phosphate. In addition, by setting the concentration of nitrate ions, or sulfate ions, or combination thereof to 1000 ppm or more, it is possible to more reliably prevent adhesion efficiency of the chemical treatment layer 107 from decreasing. In addition, by setting the concentration of Al ions to 500 ppm or more, it is possible to realize the effect of improvement in sulfide stain resistance more reliably.
[0099] Note that, by setting the upper limit value of each component of the chemical treatment solution to the above-described values, the production costs of the chemical treatment layer 107 can be reduced more reliably.
[0100] The temperature of the chemical treatment solution is preferably 5.degree. C. or higher and lower than 90.degree. C. In a case where the temperature of the chemical treatment solution is lower than 5.degree. C., it is not preferable because formation efficiency of the chemical treatment layer 107 becomes poor, which is not economically efficient. In addition, in a case where the temperature of the chemical treatment solution is 90.degree. C. or higher, it is not preferable because a formed structure of the chemical treatment layer 107 becomes inhomogeneous and cracks, and defects such as micro cracks occur, and such defects become a starting point of corrosion or the like.
[0101] Note that, when the temperature of the chemical treatment solution is higher than the surface temperature of the steel sheet 103 on which the Fe--Sn alloy layer 105a and the Sn layer 105b have been formed, reactivity of the chemical treatment solution on the interface becomes higher, and thus adhesion efficiency of the chemical treatment layer 107 is improved. Thus, it is preferable for the temperature of the chemical treatment solution to be higher than the surface temperature of the steel sheet 103 on which the Fe--Sn alloy layer 105a and the Sn layer 105b have been formed.
[0102] The current density when electrolytic treatment is performed is preferably 1.0 A/dm.sup.2 or higher and 100 A/dm.sup.2 or lower. In a case where the current density is lower than 1.0 A/dm.sup.2, the adhered amount of the chemical treatment layer 107 decreases, and an electrolytic treatment time becomes long, which is not preferable. In addition, in a case where the current density exceeds 100 A/dm.sup.2, the adhered amount of the chemical treatment layer 107 becomes excessive, and it is likely that a chemical treatment layer 107 with insufficient adhesion of the formed chemical treatment layer 107 will be washed away (peeled off) in a cleaning process by water-washing or the like after electrolytic treatment, which is not preferable.
[0103] A time for which electrolytic treatment is performed (electrolytic treatment time) is preferably 0.20 seconds or longer and 150 seconds or shorter. In a case where the electrolytic treatment time is shorter than 0.20 seconds, the adhered amount of the chemical treatment layer 107 decreases and thus desired performance is not obtained, which is not preferable. On the other hand, in a case where the electrolytic treatment time exceeds 150 seconds, the adhered amount of the chemical treatment layer 107 becomes excessive, and thus it is likely that a chemical treatment layer 107 with insufficient adhesion of the formed chemical treatment layer 107 is washed away (peeled off) in the cleaning process by water-washing or the like after electrolytic treatment, which is not preferable.
[0104] The pH of the chemical treatment solution is preferably in the range of 3.1 to 3.7, and more preferably about 3.5. Nitric acid, ammonia, or the like may be added to adjust the pH of the chemical treatment solution if necessary.
[0105] By performing electrolytic treatment under the above-described conditions, the chemical treatment layer 107 according to the present embodiment can be formed on a surface of the Sn layer 105b.
[0106] In forming the chemical treatment layer 107 according to the present embodiment, tannic acid may be further added to the chemical treatment solution to be used in electrolytic treatment. By adding tannic acid to the chemical treatment solution, tannic acid reacts with Fe contained in the steel sheet 103, and a film of iron tannate is formed on the surface of the steel sheet 103. The film of iron tannate is preferable because it improves corrosion resistance and adhesiveness.
[0107] As a solvent of the chemical treatment solution, for example, deionized water, distilled water, or the like can be used. A preferable electric conductivity of the solvent of the chemical treatment solution is 10 .mu.S/cm or lower, more preferably 5 .mu.S/cm or lower, and more preferably 3 .mu.S/cm or lower. However, a solvent of the chemical treatment solution is not limited thereto, and one can be appropriately selected depending on materials to be dissolved, formation methods, formation conditions of the chemical treatment layer 107, and the like. However, it is preferable to use deionized water or distilled water in light of industrial productivity, costs, and the environment based on stability of the adhered amount of each stable component.
[0108] As a supply source of Zr, for example, a Zr complex such as H.sub.2ZrF.sub.6 can be used. Zr in the above-described Zr complex exists as Zr.sup.4+ in the chemical treatment solution due to a hydrolysis reaction accompanying with an increase of the pH on a cathode electrode interface. Zr ions thereof is subjected to a dehydration condensation reaction with hydroxyl groups (--OH) existing on a metal surface in the chemical treatment solution, and thereby a compound such as ZrO.sub.2 or Zr.sub.3(PO.sub.4).sub.4 is formed.
[0109] In addition, in the chemical treatment solution, (NH.sub.4).sub.3AlF.sub.6 is used as a supply source of Al. By using (NH.sub.4).sub.3AlF.sub.6 as a supply source of Al, Al exists in the chemical treatment solution in a state in which Al forms a complex with F (which will be referred to as an AlF complex). Since Al included in the AlF complex is precipitated together with Zr in the electrolytic treatment process and is included in the chemical treatment layer 107, it contributes to sulfide stain resistance as described above.
[0110] In addition, Al exists in the chemical treatment solution as cations, like Zr. Thus, by using (NH.sub.4).sub.3AlF.sub.6 as a supply source of Al, Al can be supplied to the chemical treatment solution, without increasing the concentration of phosphate ions.
[0111] Meanwhile, in a case where Al.sub.2(SO.sub.4).sub.3 or the like is used as a supply source of Al as disclosed in Patent Document 3, no AlF complex is formed, thus Al is not satisfactorily precipitated in the electrolytic treatment process, and the amount of Al contained in the chemical treatment layer 107 becomes very small. In this case, the chemical treatment layer 107 does not have satisfactory sulfide stain resistance, which is not preferable.
[Post-Treatment Process]
[0112] Then, known post-treatment is performed on the steel sheet 103 on which the Fe--Sn alloy layer 105a, the Sn layer 105b, and the chemical treatment layer 107 have been formed, if necessary (Step S107).
[0113] By performing the treatment in the above-described flow, the chemical treatment steel sheet 10 according to the present embodiment is produced.
[0114] Note that, although the case where the chemical treatment layer 107 is formed through electrolytic treatment has been described above, in a case where a sufficient time can be allowed to form a chemical conversion coated film, the chemical treatment layer 107 may be formed through immersion treatment, rather than electrolytic treatment.
EXAMPLES
[0115] The chemical treatment steel sheet and the method for manufacturing a chemical treatment steel sheet according to an embodiment of the present invention will be described in detail below with introduction of examples. Note that the examples introduced below are merely examples of the chemical treatment steel sheet and the method for manufacturing a chemical treatment steel sheet according to an embodiment of the present invention, and the chemical treatment steel sheet and the method for manufacturing a chemical treatment steel sheet according to an embodiment of the present invention are not limited to the following examples.
Example 1
[0116] In Example 1, how sulfide stain resistance changes was examined by changing the amount of Al compound, without changing the amounts of Zr compound and phosphate compound in chemical treatment layers.
[0117] In Example 1, steel sheets that are generally used as steel sheets for containers were used as a base material. Fe--Sn alloy layers and Sn layers were formed on the steel sheets by performing reflow treatment in a state in which Sn-plated layers were formed on the steel sheets. The total amount of Sn contained in the Fe--Sn alloy layer and the Sn layer was set to 2.8 g/m.sup.2 per one surface for all samples.
[0118] Further, chemical treatment layers were formed by changing the concentration of Al compounds in the chemical treatment layers of each sample, and thereby a plurality of samples were produced. Here, the amount of Zr contained in the Zr compound in each sample was 8 mg/m.sup.2 per one surface, and the amount of P contained in the phosphate compound was 3 mg/m.sup.2 per one surface.
[0119] Evaluation of sulfide stain resistance was made as follows. First, 0.6 mass % of L-cysteine solution that has been boiled for one hour was put into heat resistant bottles, and the above-described samples (.phi.40 mm) were placed on and fixed to the mouths of the heat resistant bottles as lids. Next, the heat resistant bottles with the lids placed thereon as described above were subjected to heat treatment (retort treatment) in soaking furnace at 110.degree. C. for 15 minutes. Thereafter, for each sample, the appearance of the portion in contact with the heat-resistant bottle was observed and evaluated according to 10 levels based on the following criteria. In the following evaluation criteria, actual use could be withstood if a score was 5 points or more.
<Evaluation Criteria for Sulfide Stain Resistance>
[0120] Ratios of the areas that did not change to black to the contact areas of the samples with the 0.6 mass % of L-cysteine solution were graded by scores from 1 point to 10 points.
[0121] 10 points: 90% or higher and less than or equal to 100%
[0122] 9 points: 80% or higher and less than 90%
[0123] 8 points: 70% or higher and less than 80%
[0124] 7 points: 60% or higher and less than 70%
[0125] 6 points: 50% or higher and less than 60%
[0126] 5 points: 40% or higher and less than 50%
[0127] 4 points: 30% or higher and less than 40%
[0128] 3 points: 20% or higher and less than 30%
[0129] 2 points: 10% or higher and less than 20%
[0130] 1 point: 0% or higher and less than 10%
[0131] The obtained evaluation results are shown in FIG. 3. In FIG. 3, the horizontal axis represents the amount of Al compound (amount of Al metal) in the chemical treatment layer included in each sample, and the vertical axis represents the evaluation results of sulfide stain resistance.
[0132] As shown in FIG. 3, the score of the evaluation result of sulfide stain resistance was 1 point in the case where the amount of Al contained in the Al compound was less than 0.10 mg/m.sup.2 per one surface. On the other hand, the score of the evaluation result of sulfide stain resistance was 7 points or higher in the case where the amount of Al contained in the Al compound was 0.10 mg/m.sup.2 or more per one surface, and therefore it was clarified that the samples had extremely excellent sulfide stain resistance.
[0133] The results demonstrate that sulfide stain resistance of the chemical treatment steel sheet having the chemical treatment film is dramatically improved by causing the chemical treatment layer to contain a predetermined amount of Al compound.
Example 2
[0134] Next, how sulfide stain resistance changes while the amount of each component contained in each chemical treatment layer 107 is changed was examined.
[0135] More specifically, chemical treatment layers were formed on Sn layers using steel sheets on which Fe--Sn alloy layers and the Sn layers had been formed.
[0136] In addition, chemical treatment layers were formed using Al.sub.2(SO.sub.4).sub.3 as a supply source of Al ions in Comparative Examples a5 and a6 while (NH.sub.4).sub.3AlF.sub.6 was used as a supply source of Al ions in Invention Examples A1 to A18 and Comparative Examples a1 to a4.
[0137] With respect to the chemical treatment steel sheets of A1 to A18 and a1 to a6, the amounts of Zr, P, and Al contained in the chemical treatment layers were measured using a fluorescent X-ray adhesion meter, and then corrosion resistance and sulfide stain resistance thereof were evaluated.
[0138] In addition, for the amount of Al.sub.2O.sub.3 contained in the chemical treatment layers, peak intensity ratios of Al.sub.2O.sub.3, Al metal, and other Al compounds were first obtained using X-ray photoelectron spectroscopy (XPS). Further, the amount of Al.sub.2O.sub.3 contained in the chemical treatment layers was calculated using the total amount of Al metal obtained using a quantitative analysis method such as fluorescent X-ray analysis and the peak intensity ratios obtained through XPS as described above.
<Evaluation of Corrosion Resistance>
[0139] 3% acetic acid was used as a corrosion resistance test solution. The chemical treatment steel sheets serving as samples were cut out to .phi.35 mm, and then placed on and fixed to the mouths of heat resistant bottles each containing the corrosion resistance test solutions. After having the samples subjected to heat treatment at 121.degree. C. for 60 minutes, the portions of the samples in contact with the corrosion resistance test solutions were observed to evaluate the degrees of corrosion of the samples. Specifically, evaluation was made in 10 levels as follows for ratios of the areas with no corrosion to the contact areas of the samples with the corrosion resistance test solutions. Note that, if the score is 5 points or higher, the samples can be used as steel sheets for containers.
[0140] 10 points: 90% or higher and less than or equal to 100%
[0141] 9 points: 80% or higher and less than 90%
[0142] 8 points: 70% or higher and less than 80%
[0143] 7 points: 60% or higher and less than 70%
[0144] 6 points: 50% or higher and less than 60%
[0145] 5 points: 40% or higher and less than 50%
[0146] 4 points: 30% or higher and less than 40%
[0147] 3 points: 20% or higher and less than 30%
[0148] 2 points: 10% or higher and less than 20%
[0149] 1 point: 0% or higher and less than 10%
[0150] For the items of corrosion resistance evaluation, 10 points to 9 points are marked as "very good," 8 points to 5 points are marked as "good," and 4 points or lower are marked as "not good."
<Evaluation of Sulfide Stain Resistance>
[0151] Evaluation of sulfide stain resistance was made as follows. 0.6 mass % of L-cysteine solution that had been boiled for one hour was put into heat resistant bottles, and the above-described samples (.phi.40 mm) were placed on and fixed to the mouths of the heat resistant bottles as lids. The heat resistant bottles with the lids placed thereon as described above underwent heat treatment (retort treatment) in a soaking furnace at 110.degree. C. for 15 minutes. Then, the appearances of the portions of the respective samples in contact with the heat resistant bottles were observed, and evaluation was made in 10 levels on the basis of the same criteria as above. In Table 1 shown below, 10 points to 8 points are marked as "very good," 7 points to 5 points are marked as "good," and 4 points and lower are marked as "not good."
[0152] The obtained results are shown in the following Table 1.
TABLE-US-00001 TABLE 1 Chemical Treatment Steel Sheet Chemical treatment layer Plated layer Amount of Amount of Zr as Zr Total amount Amount of Sn as Sn metal Amount of P of Al Al in Al.sub.2O.sub.3 Corrosion Sulfur Stain Symbol metal (g/m.sup.2) (mg/m.sup.2) (mg/m.sup.2) (mg/m.sup.2) (mg/m.sup.2) Resistance Resistance Invention A1 0.12 143 77 5.2 3.0 Good Very Good Example A2 29 128 9.1 16 8.0 Very Good Very Good A3 15 1.2 29 29 13 Very Good Good A4 26 149 2.4 2.8 1.7 Very Good Very Good A5 21 23 1.1 25 22 Very Good Good A6 27 7.1 98 2.3 1.0 Very Good Very Good A7 14 66 54 0.12 0.11 Very Good Good A8 10 108 59 29 28 Very Good Very Good A9 27 1.2 59 26 14 Very Good Very Good A10 6.6 118 72 25 14 Very Good Very Good A11 7.7 112 2.3 2.5 2.1 Very Good Very Good A12 0.3 33 68 13 10 Very Good Very Good A13 23 36 79 0.2 0.13 Very Good Very Good A14 15 142 92 19 18 Very Good Very Good A15 0.3 80 90 0.6 0.3 Very Good Very Good A16 18 2.9 68 27 17 Very Good Very Good A17 2.6 14 5.3 3.2 2.3 Very Good Very Good A18 5.2 23 8.3 2.1 1.5 Very Good Very Good Comparative a1 0.05 143 142 10 4.9 Not Good Very Good Example a2 12 0.04 53 35 17 Not Good Very Good a3 35 138 0.3 5.8 3.4 Very Good Not Good a4 12 182 35 0.06 0.03 Very Good Not Good a5 23 2 3 0.03 0.02 Very Good Not Good a6 0.5 43 12 0.04 0.03 Very Good Not Good
[0153] As is obvious from Table 1, Examples A1 to A18 all had excellent corrosion resistance and excellent sulfide stain resistance. On the other hand, Comparative Examples a1 to a6 exhibited either of inferior corrosion resistance or inferior sulfide stain resistance. Note that, in Comparative Examples a5 and a6 in which Al.sub.2(SO.sub.4).sub.3 was used as a supply source of Al ions, the amounts of Al and Al.sub.2O.sub.3 were significantly small, and sulfide stain resistance was "not good."
Example 3
[0154] Next, how sulfide stain resistance changes according to the amount of Sn and the amount of each component contained in chemical treatment layers was examined.
[0155] The amount of S in each of the samples is shown in Table 2, and conditions for chemical treatment (conditions for chemical treatment solutions and conditions for electrolytic treatment) are shown in Table 3. The amounts of Zr, P, Al, and Al.sub.2O.sub.3 contained in the chemical treatment layers formed in each of the samples are shown in Table 4.
[0156] In addition, corrosion resistance and sulfide stain resistance were evaluated for each of the samples as in Example 2. The results are shown in Table 4.
[0157] Note that the chemical treatment layers were formed using Al.sub.2(SO.sub.4).sub.3 as a supply source of Al ions in Comparative Examples b9 and b10 while (NH.sub.4).sub.3AlF.sub.6 was used as a supply source of Al ions in Invention Examples B1 to B31 and Comparative Examples b1 to b8.
TABLE-US-00002 TABLE 2 Plated Layer Amount of Sn as Sn metal Symbol (g/m.sup.2) Invention B1 17 Example B2 21 B3 14 B4 10 B5 13 B6 21 B7 9.2 B8 27 B9 5.0 B10 12 B11 28 B12 10 B13 24 B14 19 B15 27 B16 4.3 B17 3.8 B18 27 B19 22 B20 1.7 B21 22 B22 11 B23 18 B24 26 B25 8.1 B26 28 B27 24 B28 12 B29 3.4 B30 0.8 B31 19 Comparative b1 5.1 Example b2 0.6 b3 21 b4 7.1 b5 21 b6 8.4 b7 11 b8 17 b9 21 b10 17
TABLE-US-00003 TABLE 3 Chemical treatment Chemical treatment bath Electrolytic Phosphate iNitrate Sulfate Bath treatment Zr ions F ions ions ions ions Al ions Supply source temperature Current Time Symbol (ppm) (ppm) (ppm) (ppm) (ppm) (ppm) of Al ions (.degree. C.) (A/dm.sup.2) (sec) Invention B1 12 19683 1596 5328 11630 4447 (NH.sub.4).sub.3AlF.sub.6 25 7 115 Example B2 19857 16417 2085 2638 12852 699 (NH.sub.4).sub.3AlF.sub.6 22 67 72 B3 5092 11 535 6859 18519 4925 (NH.sub.4).sub.3AlF.sub.6 82 28 47 B4 704 19347 514 9817 4173 4451 (NH.sub.4).sub.3AlF.sub.6 40 15 24 B5 2341 5154 12 14898 5773 4354 (NH.sub.4).sub.3AlF.sub.6 77 73 96 B6 13494 16380 2945 21524 1923 4802 (NH.sub.4).sub.3AlF.sub.6 19 22 124 B7 6419 8852 2058 111 -- 2949 (NH.sub.4).sub.3AlF.sub.6 67 11 134 B8 7489 15872 1548 26374 -- 2328 (NH.sub.4).sub.3AlF.sub.6 21 24 127 B9 16214 9235 1369 -- 103 2334 (NH.sub.4).sub.3AlF.sub.6 45 82 2 B10 1348 13442 1592 -- 29834 3564 (NH.sub.4).sub.3AlF.sub.6 67 16 47 B11 4254 4545 2124 17330 8589 512 (NH.sub.4).sub.3AlF.sub.6 28 67 32 B12 8364 19302 1584 7806 21587 4983 (NH.sub.4).sub.3AlF.sub.6 44 54 114 B13 3600 17014 2229 244 22137 2573 (NH.sub.4).sub.3AlF.sub.6 5.2 82 66 B14 3567 2061 1208 9103 5683 4094 (NH.sub.4).sub.3AlF.sub.6 89 64 51 B15 5914 15314 2055 3400 8852 3626 (NH.sub.4).sub.3AlF.sub.6 19 1.2 130 B16 12026 17033 2493 18941 2356 2356 (NH.sub.4).sub.3AlF.sub.6 46 97 144 B17 12626 5608 1146 3103 23543 612 (NH.sub.4).sub.3AlF.sub.6 66 72 0.3 B18 14698 10301 1237 766 18229 4209 (NH.sub.4).sub.3AlF.sub.6 17 22 148 B19 211 6192 2522 9202 4542 2679 (NH.sub.4).sub.3AlF.sub.6 85 99 21 B20 16834 12796 1552 18544 7240 866 (NH.sub.4).sub.3AlF.sub.6 82 18 122 B21 18497 204 292 14641 7805 1490 (NH.sub.4).sub.3AlF.sub.6 10 52 96 B22 19980 16732 879 10894 18808 3949 (NH.sub.4).sub.3AlF.sub.6 50 67 15 B23 7370 14400 104 18583 5778 951 (NH.sub.4).sub.3AlF.sub.6 85 51 41 B24 14865 19570 2780 4729 9162 2954 (NH.sub.4).sub.3AlF.sub.6 49 75 31 B25 13324 5902 755 973 72 1525 (NH.sub.4).sub.3AlF.sub.6 14 16 63 B26 2470 8351 240 22873 56 3618 (NH.sub.4).sub.3AlF.sub.6 41 46 107 B27 1111 178 2985 91 983 562 (NH.sub.4).sub.3AlF.sub.6 47 96 81 B28 6655 9712 2770 115 22384 554 (NH.sub.4).sub.3AlF.sub.6 68 4 50 B29 4205 192 2849 4072 812 2984 (NH.sub.4).sub.3AlF.sub.6 78 67 133 B30 8136 17766 2984 2420 15290 4618 (NH.sub.4).sub.3AlF.sub.6 78 34 87 B31 9377 16433 136 5889 6644 4129 (NH.sub.4).sub.3AlF.sub.6 32 83 103 Comparative b1 2 4 2315 30392 30302 4646 (NH.sub.4).sub.3AlF.sub.6 7 39 31 Example b2 39584 35393 6843 2467 4417 833 (NH.sub.4).sub.3AlF.sub.6 36 100 68 b3 18608 4938 4 9843 9868 3837 (NH.sub.4).sub.3AlF.sub.6 14 70 94 b4 11174 9218 2538 69 12 2734 (NH.sub.4).sub.3AlF.sub.6 38 89 57 b5 11586 16949 716 2018 6326 120 (NH.sub.4).sub.3AlF.sub.6 98 71 103 b6 12729 227 2910 17217 276 2456 (NH.sub.4).sub.3AlF.sub.6 3 129 130 b7 15053 19771 1546 6903 20580 848 (NH.sub.4).sub.3AlF.sub.6 19 0.7 187 b8 12839 13209 2905 539 4706 4177 (NH.sub.4).sub.3AlF.sub.6 36 6 0.1 b9 19283 2004 1837 2932 523 3465 Al.sub.2(SO.sub.4).sub.3 15 12 34 b10 3829 3283 1232 23533 6545 624 Al.sub.2(SO.sub.4).sub.3 25 2 24
TABLE-US-00004 TABLE 4 Chemical treatment layer Amount of Zr as Total amount of Zr metal Amount of P Al Amount of Al in Corrosion Sulfur Stain Symbol (mg/m.sup.2) (mg/m.sup.2) (mg/m.sup.2) Al.sub.2O.sub.3 (mg/m.sup.2) Resistance Resistance Invention B1 8.1 3.5 1.5 0.9 Very Good Good Example B2 48 18 8.1 7.0 Very Good Very Good B3 13 5.6 2.4 1.2 Very Good Very Good B4 3.6 1.4 0.6 0.3 Very Good Good B5 59 27 13 10 Very Good Good B6 23 11 4.9 3.9 Very Good Very Good B7 15 6.2 2.5 1.8 Very Good Good B8 29 11 4.8 2.1 Very Good Very Good B9 1.1 1.2 0.6 0.2 Very Good Good B10 6.2 2.9 1.2 1.1 Very Good Very Good B11 19 8.4 3.5 2.8 Very Good Good B12 53 22 12 5.1 Very Good Very Good B13 49 23 9.4 7.8 Very Good Good B14 30 14 6.6 4.8 Very Good Very Good B15 1.4 1.4 0.3 0.12 Very Good Good B16 132 53 26 21 Very Good Very Good B17 1.2 3.2 0.3 0.2 Very Good Good B18 27 14 5.4 4.6 Very Good Very Good B19 20 8.1 3.8 2.8 Very Good Very Good B20 20 8.3 3.6 2.9 Very Good Very Good B21 40 21 8.3 5.1 Very Good Very Good B22 10 3.9 1.8 1.2 Very Good Very Good B23 18 7.5 4.0 2.0 Very Good Very Good B24 21 10 3.9 2.0 Very Good Very Good B25 9.1 3.7 1.8 1.3 Very Good Very Good B26 48 21 8.9 4.3 Very Good Very Good B27 76 31 15 12 Very Good Very Good B28 1.7 2.1 0.3 0.2 Very Good Very Good B29 77 33 16 11 Very Good Very Good B30 27 11 5.3 2.6 Very Good Very Good B31 74 33 16 7.8 Very Good Very Good Comparative b1 0.6 6.7 2.3 2.1 Very Good Not Good Example b2 0.3 14 13 8.7 Very Good Not Good b3 56 0.6 12 9.9 Very Good Not Good b4 0.3 33 8.1 5.9 Very Good Not Good b5 59 2.1 0.03 0.02 Very Good Not Good b6 0.4 3.2 4.7 3.1 Very Good Not Good b7 0.4 1.2 0.2 0.15 Very Good Not Good b8 0.1 1.5 0.4 0.2 Very Good Not Good b9 12 1.3 0.03 0.02 Very Good Not Good 13 1.3 0.04 0.03 Very Good Not Good
[0158] As shown in Table 4, the present invention examples B1 to B31 produced in the method for manufacturing the chemical treatment steel sheets according to the present embodiment all had excellent corrosion resistance and sulfide stain resistance. Meanwhile, while Comparative Examples b1 to b10 all exhibited excellent corrosion resistance, sulfide stain resistance was poor. Note that, in Comparative Examples b9 and b10 in which Al.sub.2(SO.sub.4).sub.3 was used as a supply source of Al ions, the amounts of Al and Al.sub.2O.sub.3 were significantly small, and sulfide stain resistance was also "not good."
[0159] Although exemplary embodiments of the present invention have been described above in detail with reference to the appended drawings, the present invention is not limited thereto. It is obvious that a person having general knowledge in the field of the technology to which the present invention belongs can conceive various modified examples or corrected examples in the scope of the technical gist described in the claims, and it is of course understood that the examples belong to the technical scope of the present invention.
FIELD OF INDUSTRIAL APPLICATION
[0160] According to an embodiment described above, even in a case where an adhered amount of a chemical treatment layer is small, a chemical treatment steel sheet having excellent corrosion resistance and sulfide stain resistance and a method for manufacturing a chemical treatment steel sheet can be provided.
BRIEF DESCRIPTION OF THE REFERENCE SYMBOLS
[0161] 10 chemical treatment steel sheet [0162] 103 Steel sheet [0163] 105a Fe--Sn alloy layer [0164] 105b Sn layer [0165] 107 chemical treatment layer
* * * * *
D00000

D00001

D00002

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.