Branched Polyimide Compositions, Method Of Manufacture, And Uses Thereof
ODLE; Roy Ray ; et al.
U.S. patent application number 16/472131 was filed with the patent office on 2020-04-16 for branched polyimide compositions, method of manufacture, and uses thereof. The applicant listed for this patent is SABIC GLOBAL TECHNOLOGIES B.V.. Invention is credited to Joseph Michael DENNIS, Thomas Link GUGGENHEIM, Lioba Maria KLOPPENBURG, Timothy Edward LONG, Roy Ray ODLE.
| Application Number | 20200115501 16/472131 |
| Document ID | / |
| Family ID | 61054500 |
| Filed Date | 2020-04-16 |
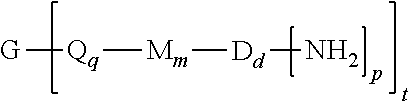
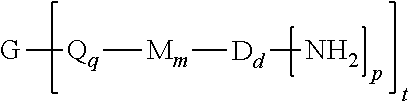
View All Diagrams
| United States Patent Application | 20200115501 |
| Kind Code | A1 |
| ODLE; Roy Ray ; et al. | April 16, 2020 |
BRANCHED POLYIMIDE COMPOSITIONS, METHOD OF MANUFACTURE, AND USES THEREOF
Abstract
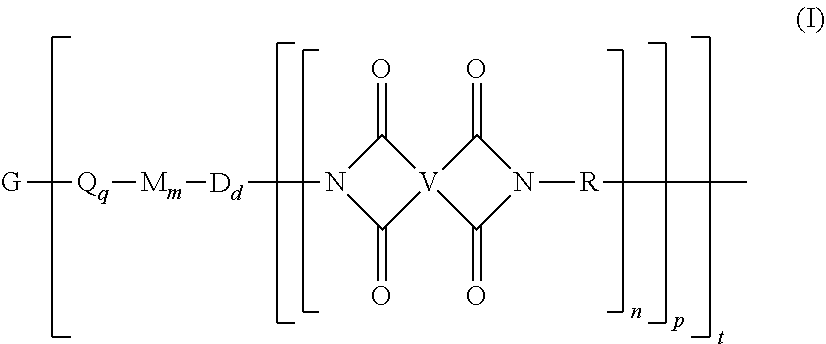
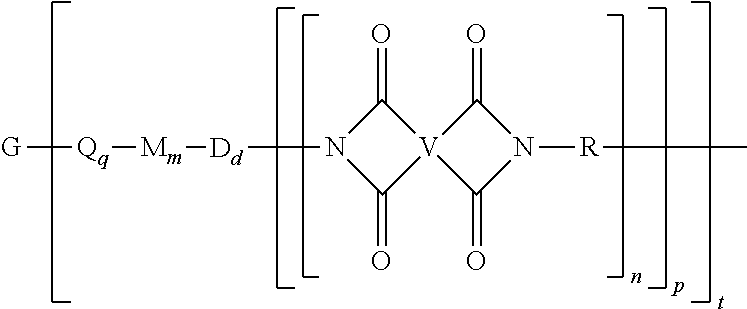
A branched polyimide of the formula (I) wherein G is a group having a valence of t present in an amount 0.01 to 20 mol %, each Q is independently the same or different, and is a divalent C.sub.1-60 hydrocarbon group, each M is independently the same or different, and is --O--, --C(O)--, --OC(O)--, --OC(O)O--, --NHC(O), --(O)CNH--, --S--, --S(O)--, or --S(O).sub.2--, D is a phenylene, each V is independently the same or different, and is a tetravalent C.sub.4-40 hydrocarbon group, each R is independently the same or different, and is a C.sub.1-20 divalent hydrocarbon group, q is 0 or 1, m is 0 or 1, d is 0 or 1, p is 1 or 2, t is 2 to 6, and each n is independently the same or different, and is 1 to 1,000, provided that the total of all values of n is greater than 4. ##STR00001##
| Inventors: | ODLE; Roy Ray; (Mt. Vernon, IN) ; GUGGENHEIM; Thomas Link; (Mt. Vernon, IN) ; KLOPPENBURG; Lioba Maria; (Mt. Vernon, IN) ; LONG; Timothy Edward; (Blacksburg, VA) ; DENNIS; Joseph Michael; (San Jose, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 61054500 | ||||||||||
| Appl. No.: | 16/472131 | ||||||||||
| Filed: | December 29, 2017 | ||||||||||
| PCT Filed: | December 29, 2017 | ||||||||||
| PCT NO: | PCT/US2017/068966 | ||||||||||
| 371 Date: | June 20, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62441226 | Dec 31, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C08L 2203/12 20130101; C08G 73/1071 20130101; C08G 73/1007 20130101; C08L 79/08 20130101; C08G 73/101 20130101; C08L 2205/03 20130101; C08L 2205/025 20130101; C08G 73/122 20130101; C08L 2203/14 20130101; C08G 73/1046 20130101; C08L 79/08 20130101; C08L 79/08 20130101 |
| International Class: | C08G 73/10 20060101 C08G073/10 |
Claims
1. A branched polyimide of the formula ##STR00036## wherein G is a group having a valence of t, present in an amount of 0.01 to 20 mole percent; each Q is independently the same or different, and is a divalent C.sub.1-60 hydrocarbon group, each M is independently the same or different, and is --O--, --C(O)--, --OC(O)--, --OC(O)O--, --NHC(O), --(O)CNH--, --S--, --S(O)--, or --S(O).sub.2--, D is a phenylene, each V is independently the same or different, and is a tetravalent C.sub.4-40 hydrocarbon group, each R is independently the same or different, and is a C.sub.1-20 divalent hydrocarbon group, q is 0 or 1, m is 0 or 1, d is 0 or 1, p is 1 or 2, t is 2 to 6, when t is 2, G is --O--, --C(O)--, --OC(O)--, --(O)CO--, --NHC(O), --(O)CNH--, --S--, --S(O)--, --S(O).sub.2--, or --P(R.sup.a)(O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, and each n is independently the same or different, and is 1 to 1,000, to 500, r provided that the total of all values of n is greater than 4.
2. The branched polyimide of claim 1, wherein when t is 3, G is a nitrogen, phosphorus, or pentavalent P(O); or G is a C.sub.1-60 hydrocarbon group having a valence oft.
3. The branched polyimide of claim 1, wherein G is --O-- when m is 0, or G is pentavalent P(O), a C.sub.6-50 hydrocarbon having at least one aromatic group, a C.sub.2-20 aliphatic group, a C.sub.4-8 cycloaliphatic group, a C.sub.3-12 heteroarylene, or a polymer moiety.
4. The branched polyimide of claim 1, wherein q is 1, Q is a C.sub.6-20 arylene, m is 1, and M is --O--.
5. The branched polyimide of claim 1, wherein V is a group of the formula ##STR00037## wherein W is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, --P(R.sup.a)(.dbd.O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof, or a group of the formula --O--Z--O-- wherein Z is an aromatic C.sub.6-24 monocyclic or polycyclic moiety optionally substituted with 1 to 6 C.sub.1-8 alkyl groups, 1 to 8 halogen atoms, or a combination thereof, provided that the valence of Z is not exceeded.
6. The branched polyimide of claim 1, wherein the branched polyimide is a branched polyetherimide of the formula ##STR00038## wherein each Z is independently an aromatic C.sub.6-24 monocyclic or polycyclic moiety optionally substituted with 1 to 6 C.sub.1-8 alkyl groups, 1 to 8 halogen atoms, or a combination thereof, provided that the valence of Z is not exceeded.
7. The branched polyimide of claim 6, wherein Z is a divalent group of the formula ##STR00039## wherein J is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, or --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof, and R is m-phenylene, p-phenylene, bis(4,4'-phenylene)sulfone, bis(3,4'-phenylene)sulfone, or bis(3,3'-phenylene)sulfone.
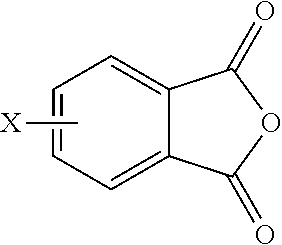
8. A method for the manufacture of the branched polyimide of claim 1, the method comprising reacting a polyamine of the formula ##STR00040## and a diamine of the formula H.sub.2N--R--NH.sub.2 with either a dianhydride of the formula ##STR00041## or an anhydride of the formula ##STR00042## in a solvent and under conditions effective to provide the branched polyimide, wherein G, Q, M, D, R, V, q, m, d, p, and t are as defined in claim 1, and wherein X is a nitro group or halogen.
9. The method of claim 8, further comprising pre-dissolving the polyamine and the diamine in the solvent before adding the dianhydride.
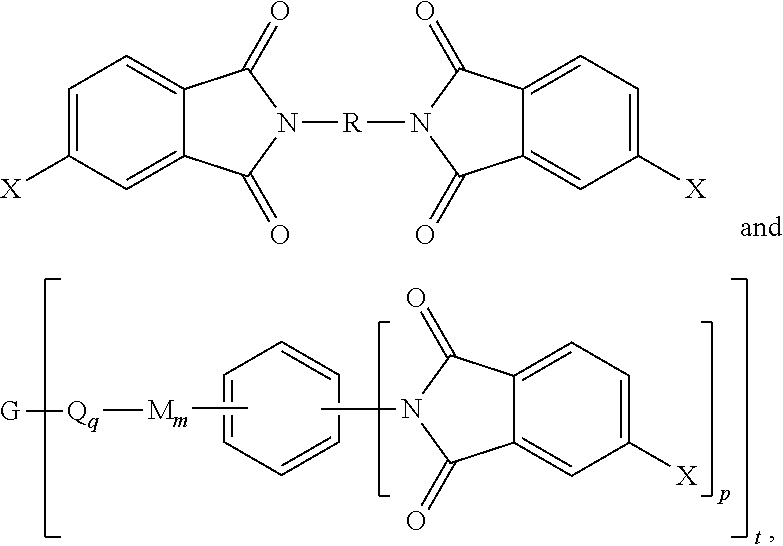
10. The method of claim 8, wherein the branched polyimide is a branched polyetherimide, the method comprising: reacting the polyamine of the formula ##STR00043## and the diamine of the formula H.sub.2N--R--NH.sub.2 with the anhydride of the formula ##STR00044## wherein X is defined in claim 8, to provide intermediate bis(phthalimide)s of the formulas ##STR00045## and reacting the bis(phthalimide)s with an alkali metal salt of a dihydroxy aromatic compound having the formula AMO--Z-OAM wherein AM is an alkali metal, to provide the branched polyetherimide, wherein G, Q, M, D, R, V, Z, q, m, d, p, and t are as defined in claim 1.
11. The method of claim 8, wherein the polyamine is of the formulas ##STR00046##
12. The method of claim 8, wherein the dianhydride is 2,2-bis[4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride, and the diamine is bis-(4-aminophenyl) sulfone or m-phenylenediamine.
13. The branched polyimide of claim 1, having one or more of the following properties: a T.sub.g greater than 100.degree. C.; or an average branch molecular weight of 12,000 to 50,000 grams per mole; or less than 5 weight percent of a gel, based on the total weight of the branched polyimide; a polydispersity of 1.5 to 3.0, as determined by size exclusion chromatography multi-angle light scattering; or a UL94 rating that is the better than or equal to a UL94 rating of the same polyimide manufactured without the polyamine.
14. (canceled)
15. A polymer composition, comprising: 1 to 99 weight percent of the branched polyimide of claim 1.
16. (canceled)
17. The polymer composition of claim 15, further comprising: a polymer different from the branched polyimide.
18. The polymer composition of claim 17, wherein the polymer is a polyacetal, poly(C.sub.1-6 alkyl)acrylate, polyacrylamide, polyacrylonitrile, polyamide, polyamideimide, polyanhydride, polyarylene ether, polyarylene ether ketone, polyarylene ketone, polyarylene sulfide, polyarylene sulfone, polybenzothiazole, polybenzoxazole, polybenzimidazole, polycarbonate, polyester, polyetherimide, polyimide, poly(C.sub.1-6 alkyl)methacrylate, polymethacrylamide, cyclic olefin polymer, polyolefin, polyoxadiazole, polyoxymethylene, polyphthalide, polysilazane, polysiloxane, polystyrene, polysulfide, polysulfonamide, polysulfonate, polythioester, polytriazine, polyurea, polyurethane, vinyl polymer, or a combination thereof.
19. (canceled)
20. (canceled)
21. The polymer composition of claim 15, further comprising 99 to 1 weight percent of a second polyimide that is not the same as the branched polyimide, wherein each amount is based on a total weight of the branched polyimide and the second polyimide.
22. The branched polyimide of claim 1, wherein group G is derived from a polyamine of the formula ##STR00047## wherein the amount of the polyamine present during the preparation of the branched polyimide is substantially the same as the amount of group G present in the branched polyimide.
23. The method of claim 8, wherein the polyamine is at least one compound of the formulas (8a) to (8t) ##STR00048## ##STR00049## ##STR00050## ##STR00051## ##STR00052## wherein, in formula (8f), Z is a divalent C.sub.1-60 hydrocarbon group.
24. The method of claim 8, wherein the polyamine is present in the reacting step in an amount of 0.01 to 20 mole percent, based on the total number of moles of amine functionality of the polyamine and the diamine.
Description
BACKGROUND
[0001] Polyimides (PIs), and in particular polyetherimides (PEIs), are amorphous, transparent, high performance polymers having a high glass transition temperature. Polyetherimides further have high strength, heat resistance, and modulus, and broad chemical resistance, and thus are widely used in applications as diverse as automotive, telecommunication, aerospace, electrical/electronics, transportation, and healthcare. Moreover, PEIs can be recycled, whereas some PIs are thermosets that cannot be recycled.
[0002] An ongoing challenge associated with polyimides and polyetherimides is synthesizing desirable polyimides and polyetherimides having long-chain branches (LCBs). The inclusion of long-chain branches can influence the melt strength of the polymers, and can reduce the melt viscosity of higher molecular weight polymers for a given processing temperature. Furthermore, long-chain branches can improve shear-thinning and extensional flow processing over linear analogues. For instance, the introduction of long-chain branches into polyesters (e.g., poly(ethylene terephthalate)) can improve melt strength and reduce the rate of crystallinity.
[0003] Accordingly, there remains a continuing need in the art for polyimides and polyetherimides that have long-chain branches.
SUMMARY
[0004] A branched polyimide has the formula
##STR00002##
wherein G is a group having a valence of t, present in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol %, each Q is independently the same or different, and is a divalent C.sub.1-60 hydrocarbon group, each M is independently the same or different, and is --O--, --C(O)--, --OC(O)--, --OC(O)O--, --NHC(O), --(O)CNH--, --S--, --S(O)--, or --S(O).sub.2--, D is a phenylene, each V is independently the same or different, and is a tetravalent C.sub.4-40 hydrocarbon group, each R is independently the same or different, and is a C.sub.1-20 divalent hydrocarbon group, q is 0 or 1, m is 0 or 1, d is 0 or 1, p is 1 or 2, t is 2 to 6, preferably 2 to 4, and each n is independently the same or different, and is 1 to 1,000, preferably 2 to 500, or 3 to 100, the total of all values of n is greater than 4, preferably greater than 10, or greater than 20, or greater than 50, or greater than 100, or greater than 250.
[0005] A method for the manufacture of the branched polyimide includes reacting a polyamine of the formula
##STR00003##
and a diamine of the formula
H.sub.2N--R--NH.sub.2
with a dianhydride of the formula
##STR00004##
in a solvent and under conditions effective to provide the branched polyimide, wherein G, Q, M, D, R, V, q, m, d, p, and t are as defined above.
[0006] Another method for the manufacture of a branched polyetherimide includes reacting a polyamine of the formula
##STR00005##
and a diamine of the formula
H.sub.2N--R--NH.sub.2
with an anhydride of the formula
##STR00006##
wherein X is a nitro group or halogen, to provide intermediate bis(phthalimide)s of the formulas
##STR00007##
and reacting the bis(phthalimide)s with an alkali metal salt of a dihydroxy aromatic compound having the formula
AMO--Z--OAM
wherein AM is an alkali metal, to provide the branched polyetherimide, wherein G, Q, M, D, R, V, Z, q, m, d, p, and t are as defined above.
[0007] A polyimide composition includes 1 to 99 wt %, or 10 to 90 wt %, or 0.1 to 20 wt %, or 0.5 to 10 wt %, or 1 to 5 wt % of a branched polyimide; and 91 to 1 wt %, or 90 to 10 wt %, or 99.9 to 80 wt %, or 99.5 to 90 wt %, or 99 to 95 wt % of the above branched polyimide and a second polyimide that is not the same as the branched polyimide, wherein each amount is based on the total weight of the branched polyimide and the polyimide.
[0008] A polymer composition includes the polyimide composition; and a second polymer that is not the same as the branched polyimide or the second polyimide.
[0009] An article includes the branched polyimide, the polyimide composition, or the polymer composition.
[0010] The above described and other features are exemplified by the following figures and detailed description.
BRIEF DESCRIPTION OF THE DRAWINGS
[0011] The following figures are exemplary embodiments wherein the like elements are numbered alike.
[0012] FIG. 1 is a chart showing dianhydrides according to an embodiment.
[0013] FIG. 2 is a chart showing diamines according to an embodiment.
[0014] FIG. 3 is a graph of average branch molecular weight (grams per mole (g/mol), Mb) versus mole percent (mol %) of tris((p-aminophenoxy)phenyl) ethane (TAPE) as measured by size exclusion chromatography-multiple angle light scattering (SEC-MALS) and .sup.1H NMR spectroscopy.
[0015] FIG. 4 is a graph of viscosity (pascal seconds (Pa s), .eta.) versus shear rate (radians per second (rad/s), .omega.) and shows a correlation between viscosity and shear rate.
[0016] FIG. 5 is a graph of molecular weight (g/mol) versus mole percent (% BA, TAPE) versus torque (Newtonmeters, Nm).
[0017] FIG. 6 is a graph of molecular weight (g/mol) versus mole percent (% BA, TAPE) versus torque (Nm).
[0018] FIG. 7 shows photographs of a homogeneous polyetherimide with 1 wt % TAPE via a pre-dissolved amines method (left) and a homogenous polyetherimide with dark gel spots at 1.5 wt % TAPE prepared by without pre-dissolving in amines (right).
DETAILED DESCRIPTION
[0019] The inventors hereof have discovered that the synthesis of poly-functional aryl amines (polyamines), and subsequent polymerization with a dianhydride and a diamine, provides long chain branched poly(imides) (LCB-PIs) and polyetherimides (LCB-PEIs). Careful consideration of the reaction conditions and molar ratios permits the use of higher molar ratios of the polyamines without forming an insoluble network during the synthetic steps. In contrast to LCB polyesters, LCB-PEIs prepared with 0.5 mole percent (mol %) of polyamine had comparable melt viscosities and processibility as compared to PEIs without the long-chain branching. Higher incorporation (2 mol %) of the polyamines into PEI resulted in LCB-PEIs having a reduced melt viscosity. Furthermore, the chain dispersity increased at the higher incorporation of the polyamines in the LCB-PEI. The increased chain distribution includes concentrations of both higher and lower molecular weight species.
[0020] Such properties are especially useful in the manufacture of thin-wall parts, where high-flow properties, especially low melt viscosity under the high shear conditions are important in injection molding. The LCB-PI and LCB-PEI can satisfy this criterion and fare better than the linear chained counterpart of the same molecular weight. Without being bound by theory, under melt conditions, polymers display shear-thinning property being a non-Newtonian fluid. An LCB-PI and LCB-PEI can shear thin faster (akin to any branched polymer over its linear counterpart), giving lower viscosity and subsequently higher flow rates with less processing demands. While the viscosity under shear can be lowered for linear PI or PEI by either increasing the temperature or using lower molecular weight polymers, such solutions can lead to degradation at high heat or lower impact properties of the molded materials, respectively.
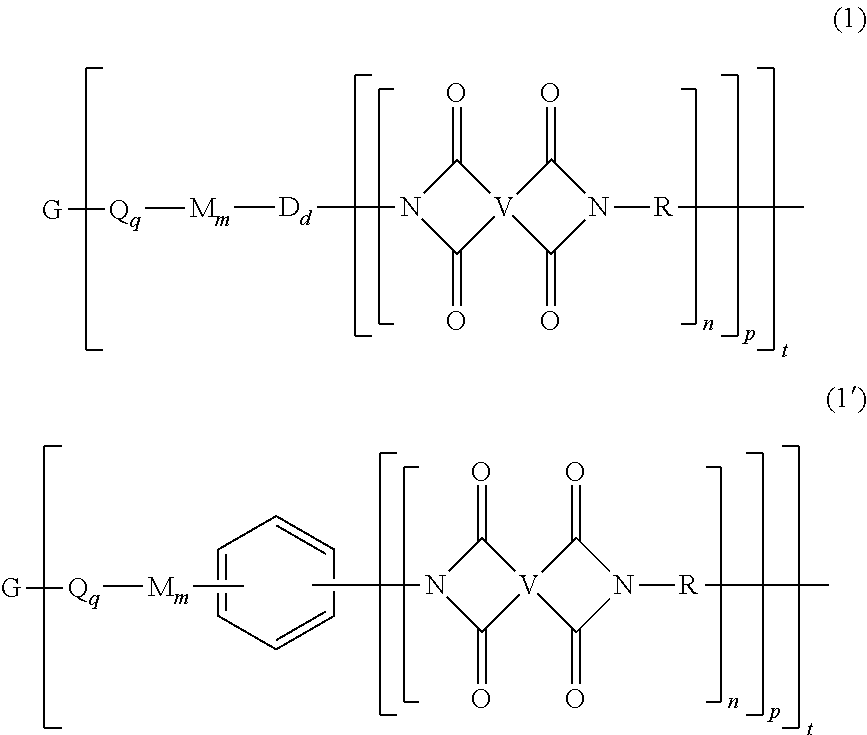
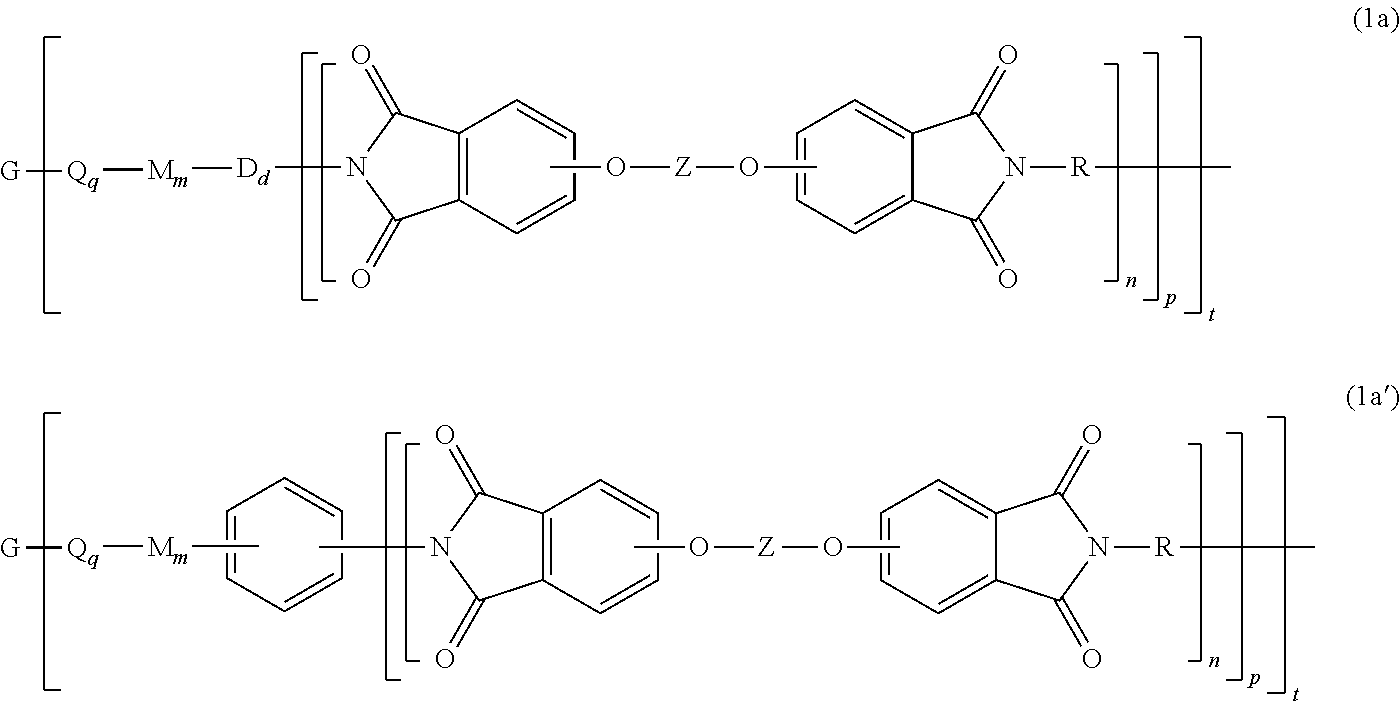
[0021] The LCB-PI is a branched polyimide of formula (1) or (1').
##STR00008##
[0022] In formula (1) and (1'), G is a group having a valence of t, present in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol %, and q is 0 or 1, m is 0 or 1, d is 0 or 1, p is 1 or 2, t is 2 to 6, preferably 2 to 4. In an embodiment, t is 2, and G is --O--, --C(O)--, --OC(O)--, --(O)CO--, --NHC(O), --(O)CNH--, --S--, --S(O)--, --S(O).sub.2--, or --P(R.sup.a)(O)-- (wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl). In another embodiment, t is 3, and G is nitrogen, phosphorus, or P(O). In still another embodiment, G is a C.sub.1-60 hydrocarbon group having a valence of t. In a preferred embodiment, G is --O-- when m is 0, pentavalent P(O), a C.sub.6-60 hydrocarbon having at least one aromatic group, for example a C.sub.6-40 aromatic hydrocarbon group, a C.sub.2-20 aliphatic group, a C.sub.4-8 cycloaliphatic group, a C.sub.3-12 heteroarylene, or a polymer moiety; or G is --O--, --S(O).sub.2--, pentavalent P(O), a C.sub.6-20 aromatic hydrocarbon group, a C.sub.2-20 aliphatic group, or a C.sub.4-8 cycloaliphatic group. In a specific embodiment, G is --O--, pentavalent P(O), or a C.sub.6-50 hydrocarbon having at least one aromatic group. When q, m, and d are 0, G can be a saturated C.sub.2-20 aliphatic group, C.sub.3-12 heteroarylene or a polymeric moiety, for example an amino resin such as a urea-formaldehyde, a melamine-formaldehyde, or other resin having active amine groups.
[0023] In formula (1) and (1'), each Q is independently the same or different, and is a divalent C.sub.1-60 hydrocarbon group. In a preferred embodiment, Q is a C.sub.6-20 arylene, a C.sub.1-20 alkylene, or a C.sub.3-8 cycloalkylene. In a more preferred embodiment, Q is a C.sub.6-20 arylene.
[0024] In formula (1) and (1'), each M is independently the same or different, and is --O--, --C(O)--, --OC(O)--, --OC(O)O--, --NHC(O), --(O)CNH--, --S--, --S(O)--, --S(O).sub.2--. In another embodiment, M is --O--, --C(O)--, --OC(O)--, --P(R.sup.a)--, or --P(O)R.sup.a--. In an embodiment, M is --O--, --C(O)--, --OC(O)--, --P(R.sup.a)--, or --P(O)R.sup.a-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl.
[0025] In formula (1) and (1'), each D is phenylene. In an embodiment, each D is the same or different, and is m-phenylene or p-phenylene.
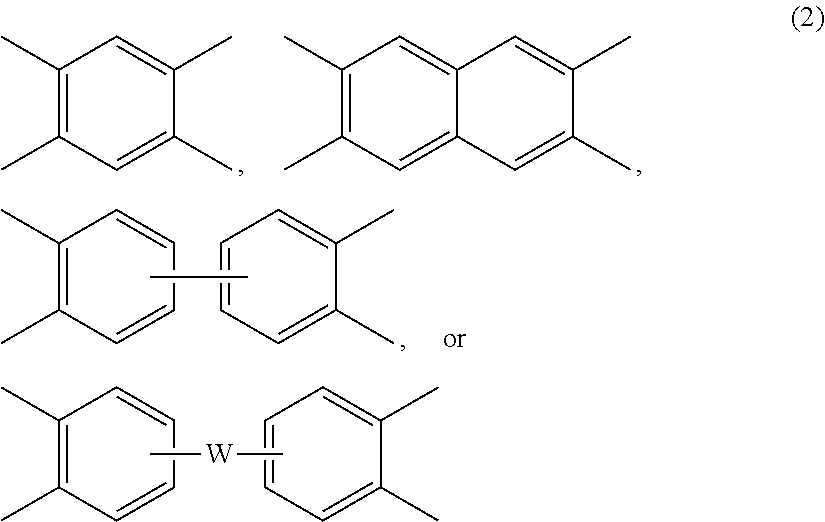
[0026] Further in formula (1) and (1'), each V is independently the same or different, and is a tetravalent C.sub.4-40 hydrocarbon group. In an embodiment, V is a C.sub.6-20 aromatic hydrocarbon group. Exemplary aromatic hydrocarbon groups include any of those of the formulas (2)
##STR00009##
wherein W is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, --P(R.sup.a)(.dbd.O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof (which includes perfluoroalkylene groups), or a group of the formula --O--Z--O-- as described in formula (1a) and (1a') below.
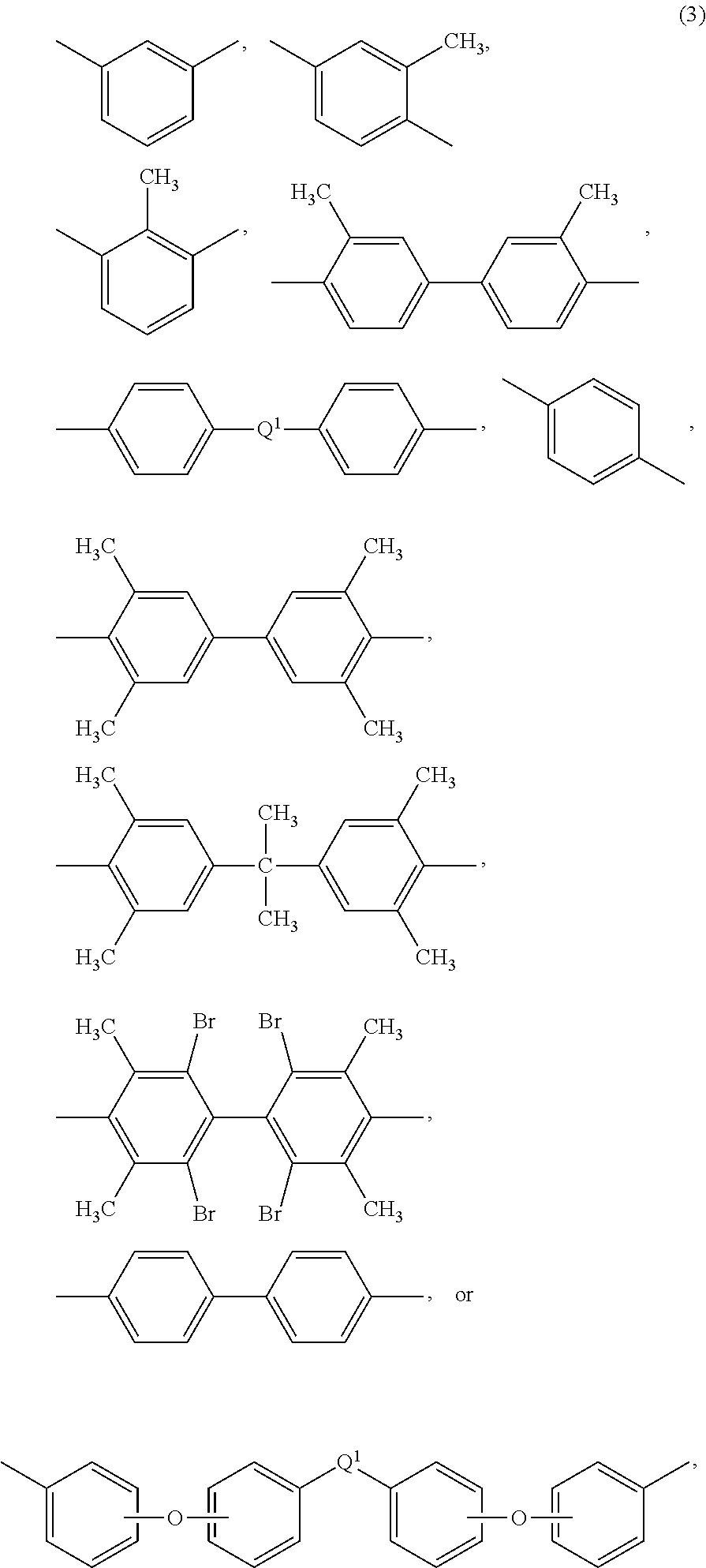
[0027] Also in formula (1) and (1'), each R is independently the same or different, and is a C.sub.1-20 divalent hydrocarbon group. Specifically, each R can be the same or different, and is a divalent organic group, such as a C.sub.6-20 aromatic hydrocarbon group or a halogenated derivative thereof, a straight or branched chain C.sub.2-20 alkylene group or a halogenated derivative thereof, a C.sub.3-8 cycloalkylene group or halogenated derivative thereof, in particular a divalent group of any of formulas (3)
##STR00010##
wherein Q.sup.1 is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, --P(R.sup.a)(.dbd.O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof (which includes perfluoroalkylene groups), or --(C.sub.6H.sub.10).sub.z-- wherein z is an integer from 1 to 4. In an embodiment R is m-phenylene, p-phenylene, or a diarylene sulfone.
[0028] Still further in formula (1) and (1'), each n is independently the same or different, and is 1 to 1,000, preferably 2 to 500, or 3 to 100, provided that the total of all values of n is greater than 4, preferably greater than 10, more preferably greater than 20, or greater than 50, or greater than 100, or greater than 250, or 4 to 50, or 10 to 50, or 20 to 50, or 4 to 100, or 10 to 100, or 20 to 100.
[0029] In another specific embodiment, the branched polyimide of formula (1) or (1') can be a branched polyetherimide of formula (1a), preferably (1a')
##STR00011##
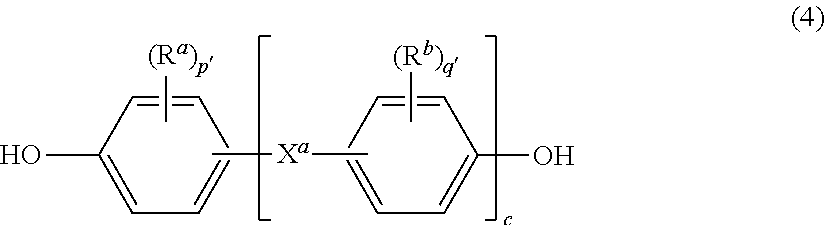
wherein G, Q, M, D, R, q, m, d, n, p, and t are as defined in formula (1) and (1'), and wherein the divalent bonds of the --O--Z--O-- group are in the 3,3', 3,4', 4,3', or the 4,4' positions. The group Z in --O--Z--O-- of formula (1a) and (1a') is a divalent organic group, and can be an aromatic C.sub.6-24 monocyclic or polycyclic moiety optionally substituted with 1 to 6 C.sub.1-8 alkyl groups, 1 to 8 halogen atoms, or a combination thereof, provided that the valence of Z is not exceeded. Exemplary groups Z include groups derived from a dihydroxy compound of formula (4)
##STR00012##
wherein R.sup.a and R.sup.b can be the same or different and are a halogen atom or a monovalent C.sub.1-6 alkyl group, for example; p' and q' are each independently integers of 0 to 4; c is 0 to 4; and X.sup.a is a bridging group connecting the hydroxy-substituted aromatic groups, where the bridging group and the hydroxy substituent of each C.sub.6 arylene group are disposed ortho, meta, or para (preferably para) to each other on the C.sub.6 arylene group. The bridging group X.sup.a can be a single bond, --O--, --S--, --S(O)--, --S(O).sub.2--, --C(O)--, or a C.sub.1-18 organic bridging group. The C.sub.1-18 organic bridging group can be cyclic or acyclic, aromatic or non-aromatic, and can further comprise heteroatoms such as halogens, oxygen, nitrogen, sulfur, silicon, or phosphorous. The C.sub.1-18 organic group can be disposed such that the C.sub.6 arylene groups connected thereto are each connected to a common alkylidene carbon or to different carbons of the C.sub.1-18 organic bridging group. A specific example of a group Z is a divalent group of formula (4a)
##STR00013##
wherein J is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, or --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof (including a perfluoroalkylene group). In a specific embodiment Z is a derived from bisphenol A, such that J in formula (4a) is 2,2-isopropylidene.
[0030] In an embodiment in formulas (1), (1'), (1a), and (1a') (hereinafter collectively "branched polyimide" for convenience) R is m-phenylene or p-phenylene, bis(4,4'-phenylene)sulfone, bis(3,4'-phenylene)sulfone, or bis(3,3'-phenylene)sulfone. In this embodiment, Z can be a divalent group of formula (4a). In an alternative embodiment, R is m-phenylene or p-phenylene and Z is a divalent group of formula (4a) and J is 2,2-isopropylidene.
[0031] In some embodiments, the branched polyimide can be a copolymer, for example a polyetherimide sulfone copolymer comprising structural units of formulas (1), (1'), (1a), or (1a') wherein at least 50 mol % of the R groups are of formula (3) wherein Q.sup.1 is --SO.sub.2-- and the remaining R groups are independently p-phenylene or m-phenylene or a combination thereof; and Z is 2,2'-(4-phenylene)isopropylidene. Alternatively, the branched polyetherimide copolymer optionally comprises additional structural imide units, for example imide units wherein V is of formula (2a) wherein R and V are as described in formula (2a), for example V is
##STR00014##
wherein W is a single bond, --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, --P(R.sup.a)(.dbd.O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, or --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof (which includes perfluoroalkylene groups). These additional structural imide units can comprise less than 20 mol % of the total number of units, or 0 to 10 mol % of the total number of units, or 0 to 5 mol % of the total number of units, or 0 to 2 mol % of the total number of units. In some embodiments, no additional imide units are present in the branched polyimides other than polyetherimide units.
[0032] The branched polyimides (which as indicated above include polyimides (1) and (1') and the branched polyetherimides (1a) and (1a')), can be prepared by methods known in the art, including a polycondensation or ether-forming polymerization. In any process, the appropriate amount of a polyamine of formula (8), preferably of formula (8')
##STR00015##
is introduced during manufacture of the branched polyimides as described in further detail below. In formula (8) and (8'), G, Q, M, D, q, m, d, p, and t are defined as described in formulas (1) (1'), (1a), and (1a').
[0033] Exemplary polyamines (8) and (8') can include any of formulas (8a)-(8t).
##STR00016## ##STR00017## ##STR00018## ##STR00019##
wherein, in formula (8f), Z is a divalent C.sub.1-600 hydrocarbon group, or a C.sub.6-40 aromatic hydrocarbon group, a C.sub.2-20 aliphatic group, or a C.sub.4-8 cycloaliphatic group.
[0034] Methods for the synthesis of the polyamines are known in the art. An exemplary method for the synthesis of the polyamine of formulas (8) and (8') uses a two-step sequence as exemplified in Examples 1 and 2. The first step, is a nucleophilic aromatic substitution of a halogenated aromatic nitro compound (e.g., 1-chloro-4-nitrobenzene) with a polyphenol (e.g., 1,1,1-tris(4-hydroxyphenyl) ethane) that is converted to a polyphenoxide in-situ, providing a sufficiently nucleophilic oxygen to displace the activated halide. A polar aprotic solvent (e.g., dimethylacetamide) can promote the substitution reaction to afford a poly(nitrophenyl) compound (e.g., 1,1,1-tris((p-nitrophenoxy)phenyl) ethane). The second step is a reduction of the poly(nitrophenyl) compound to the polyamine of formula (10) using, for example, a palladium catalyst with a reducing agent, an iron-based catalyst, vasicine, zinc, samarium, and hydrazine.
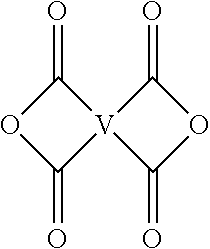
[0035] The branched polyimide can be prepared by polycondensation, which includes an imidization of a dianhydride of formula (9) or formula (9a)
##STR00020##
or a chemical equivalent thereof, with a combination of an organic diamine of formula (10)
H.sub.2N--R--NH.sub.2 (10)
and a polyamine of formula (8), preferably of formula (8')
##STR00021##
wherein V, Z, R, G, Q, M, D, q, m, d, p, and t are defined as described in formulas (1), (1'), (1a), and (1a'). The polyamine (8), preferably (8') can be present in the reaction in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol % to achieve increased branching and increased PDI.
[0036] Exemplary dianhydrides include 3,3-bis[4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride; 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl ether dianhydride; 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl sulfide dianhydride; 4,4'-bis(3,4-dicarboxyphenoxy)benzophenone dianhydride; 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl sulfone dianhydride; 2,2-bis[4-(2,3-dicarboxyphenoxy)phenyl]propane dianhydride; 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl ether dianhydride; 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl sulfide dianhydride; 4,4'-bis(2,3-dicarboxyphenoxy)benzophenone dianhydride; 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl sulfone dianhydride; 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl-2,2-propane dianhydride; 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl ether dianhydride; 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl sulfide dianhydride; 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)benzophenone dianhydride; and, 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl sulfone dianhydride, as well as various combinations thereof.
[0037] Still other specific dianhydrides include any of those as shown in FIG. 1, wherein Y' is --C(O)--, --C(CF.sub.3).sub.2--, --C(CH.sub.3).sub.2--, --SO.sub.2--, or --C.ident.C--.
[0038] Specific examples of organic diamines include hexamethylenediamine, polymethylated 1,6-n-hexanediamine, heptamethylenediamine, octamethylenediamine, nonamethylenediamine, decamethylenediamine, 1,12-dodecanediamine, 1,18-octadecanediamine, 3-methylheptamethylenediamine, 4,4-dimethylheptamethylenediamine, 4-methylnonamethylenediamine, 5-methylnonamethylenediamine, 2,5-dimethylhexamethylenediamine, 2,5-dimethylheptamethylenediamine, 2, 2-dimethylpropylenediamine, N-methyl-bis (3-aminopropyl) amine, 3-methoxyhexamethylenediamine, 1,2-bis(3-aminopropoxy) ethane, bis(3-aminopropyl) sulfide, 1,4-cyclohexanediamine, bis-(4-aminocyclohexyl) methane, m-phenylenediamine, p-phenylenediamine, 2,4-diaminotoluene, 2,6-diaminotoluene, m-xylylenediamine, p-xylylenediamine, 2-methyl-4,6-diethyl-1,3-phenylene-diamine, 5-methyl-4,6-diethyl-1,3-phenylene-diamine, benzidine, 3,3'-dimethylbenzidine, 3,3'-dimethoxybenzidine, 1,5-diaminonaphthalene, bis(4-aminophenyl) methane, bis(2-chloro-4-amino-3,5-diethylphenyl) methane, bis(4-aminophenyl) propane, 2,4-bis(p-amino-t-butyl) toluene, bis(p-amino-t-butylphenyl) ether, bis(p-methyl-o-aminophenyl) benzene, bis(p-methyl-o-aminopentyl) benzene, 1, 3-diamino-4-isopropylbenzene, bis(4-aminophenyl) sulfide, bis-(4-aminophenyl) sulfone (also known as 4,4'-diaminodiphenyl sulfone (DDS)), and bis(4-aminophenyl) ether. Any regioisomer of the foregoing compounds can be used. Combinations of these compounds can also be used. In some embodiments the organic diamine is m-phenylenediamine, p-phenylenediamine, 4,4'-diaminodiphenyl sulfone, or a combination thereof.
[0039] Still other organic diamines can include any of those as shown in FIG. 2, wherein A is --O--, --CH.sub.2--, --CH.sub.2CH.sub.2--, --SO.sub.2, --C(CF.sub.3).sub.2--, --S--, --S--S--, --CH.dbd.CH--, --C(O)--, --NH--, or --C(CH.sub.3).sub.2--; A.sub.1 is --Cl, --OH, --OCH.sub.3, --CH.sub.3, or --CH.sub.2CH.sub.3; A.sub.2 is --CH.sub.3, --CF.sub.3, or --SO.sub.3H; A.sub.3 is --CH.sub.3 or --OH; and n' is 1, 2, or 3.
[0040] An endcapping agent can be present during imidization, in particular a monofunctional compound that can react with an amine or anhydride. Exemplary compounds include monofunctional aromatic anhydrides such as phthalic anhydride, an aliphatic monoanhydride such as maleic anhydride, or monofunctional aldehydes, ketones, esters isocyanates, aromatic monoamines such as aniline, or C.sub.1-C.sub.18 linear or cyclic aliphatic monoamines. A monofunctional bisphthalimide can also be added before or during imidization. The amount of endcapping agent that can be added depends on the desired amount of chain terminating agent, and can be, for example, more than 0 to 10 mole percent (mol %), or 0.1 to 10 mol %, or 0.1 to 6 mol %, based on the moles of endcapping agent and amine or anhydride reactant.
[0041] A catalyst can be present during imidization. Exemplary catalysts include sodium aryl phosphinates, guanidinium salts, pyridinium salts, imidazolium salts, tetra(C.sub.7-24 arylalkylene) ammonium salts, dialkyl heterocycloaliphatic ammonium salts, bis-alkyl quaternary ammonium salts, (C.sub.7-24 arylalkylene)(C.sub.1-16 alkyl) phosphonium salts, (C.sub.6-24 aryl)(C.sub.1-16 alkyl) phosphonium salts, phosphazenium salts, and combinations thereof. The anionic component of the salt is not particularly limited, and can be, for example, chloride, bromide, iodide, sulfate, phosphate, acetate, maculate, tosylate, and the like. A combination of different anions can be used. A catalytically active amount of the catalyst can be determined by one of skill in the art without undue experimentation, and can be, for example, more than 0 to 5 mol % percent, or 0.01 to 2 mol %, or 0.1 to 1.5 mol %, or 0.2 to 1.0 mol % based on the moles of polyamine (8) or (8') and organic diamine (10).
[0042] A general synthesis of the branched polyetherimides via this embodiment is shown in Scheme 1.
##STR00022##
In Scheme 1, T can be a group of the formula O--Z--O as described in formula (1a) and (1a'), and G is a C.sub.1-60 hydrocarbon group having a valence of 2, or a C.sub.6-40 aromatic hydrocarbon group, a C.sub.2-20 aliphatic group, or a C.sub.4-8 cycloaliphatic group, and x'', y'', z'', and p'' each have a value of n as described in formulas (1), (1'), (1a), and (1a').
[0043] Conditions effective to provide the polyimides are generally known. Polymerization is generally carried out in a solvent, for example relatively non-polar solvents with a boiling point above 100.degree. C., or above 150.degree. C., for example o-dichlorobenzene, dichlorotoluene, 1,2,4-trichlorobenzene, diphenyl sulfone, or a monoalkoxybenzene such as anisole, veratrole, diphenylether, or phenetole. Ortho-dichlorobenzene and anisole can be particularly mentioned. The polymerization is generally at least 110.degree. C., or 150 to 275.degree. C., or 175 to 225.degree. C. for solution polymerization. At temperatures below 110.degree. C., reaction rates may be too slow for economical operation. Atmospheric or super-atmospheric pressures can be used, for example up to 5 atmospheres, to facilitate the use of high temperatures without causing solvent to be lost by evaporation. Effective times depend on the particular reactants and reaction conditions, and can be 0.5 hours to 3 days, for example, generally for 0.5 to 72 hours, preferably 1 to 30 or 1 to 20 hours. Advantageously, the reaction is complete 20 hours or less, preferably 10 hours or less, more preferably 3 hours or less.
[0044] It has been found that improved compositions can be obtained by pre-dissolving the polyamine (8), preferably (8'), and the diamine (10) before adding the dianhydride (9) or (9a), or before adding the diamine/polyamine to the dianhydride. The catalyst can be added any time during the reaction between the polyamine (8), preferably (8'), and organic diamine (10), and the dianhydride (9) or (9a) continuously or in portions during the course of the reaction. In some embodiments, the catalyst is added after pre-dissolution the polyamine (8), preferably (8'), and organic diamine (10), with the dianhydride (9) or (9a).
[0045] The solvent, polyamine (8), preferably (8'), diamine (10), dianhydride (9) or (9a), and optional components (e.g., catalyst and endcapping agent) (i.e., the reaction mixture) can be combined in amounts such that the total solids content the during the reaction to form the branched polyimide are 5 to 70 weight percent (wt %), preferably 10 to 70 wt %, more preferably 20 to 70 wt %. "Total solids content" expresses the proportion of the reactants as a percentage of the total weight including liquids present in the reaction at any given time. It can be desirable to have low water content in the reaction mixture, for example the reaction mixture can comprise 200 parts per million by weight (ppm) or less of water, 100 ppm or less of water, or 50 ppm or less of water, or to 25 ppm or less of water, based on parts by weight of the reaction mixture.
[0046] A molar ratio of dianhydride (9) or (9a) to a combination of polyamine (8), preferably (8'), and diamine (10) of 0.9:1 to 1.1:1, or 1:1 can be used. While other ratios can be used, a slight excess of dianhydride or diamine may be desirable. A proper stoichiometric balance between the dianhydride and combination of polyamine (8), preferably (8'), and diamine (10) is maintained to allow for the production of the desired molecular weight of the polymer, or prevent the formation of polymer with significant amounts of amine end groups. Accordingly, in an embodiment, imidization proceeds via forming an initial reaction mixture having a targeted initial molar ratio of dianhydride (9) or (9a) to a combination of polyamine (8), preferably (8'), and diamine (10); heating the reaction mixture to a temperature of at least 100.degree. C. to initiate polymerization; analyzing the molar ratio of the heated reaction mixture to determine the actual initial molar ratio of dianhydride (9) or (9a) to polyamine (8), preferably (8'), and diamine (10), using, e.g., an infrared spectroscopy technique, a titration technique, or a NMR technique; and, if necessary, adding dianhydride (9) or (9a), or polyamine (8), preferably (8'), or diamine (10) to the analyzed reaction mixture to adjust the molar ratio of dianhydride (9) or (9a) to polyamine (8), preferably (8'), and diamine (10) to 0.9:1 to 1.1:1.
[0047] If an amine-containing endcapping agent is used, the amount can be more than 0 to 10 mol % based on the total amount of dianhydride (9) or (9a). If an anhydride-containing endcapping agent is used, the amount can be in the range of more than 0 to 20 mol %, or 1 to 10 mol % based on the amount of the polyamine (8), preferably (8'), and diamine (10) combined. In general, due to the presence of the polyamines, an anhydride-containing endcapping agent is used to decrease the number of amine end groups in the branched polyimides and polyetherimides. The endcapping agent can be added at any time. In some embodiments, the endcapping agents are mixed with or dissolved into reactants having the similar functionality. For example, anhydride-containing endcapping agent can be combined with dianhydride (9) or (9a). Where an anhydride-containing endcapping agent is used, in order to achieve maximum molecular weight, the quantity of amine functionality ([2.times.diamine moles]+[t.times.polyamine, moles wherein t is the number of reactive amino groups])=moles of anhydride functionality ([2.times.dianhydride moles+moles of anhydride in the endcapping agent]). As described above, the stoichiometry condition of the polymerization reaction mixture can be analyzed, and the stoichiometry corrected if needed to provide a stoichiometry within +0.2 mol % of a stoichiometry of 1:1.
[0048] In other embodiments, the branched polyimides can be synthesized by an ether-forming polymerization, which proceeds via an imidization, i.e., reaction of the polyamine of formula (8), preferably (8'), and the diamine of formula (10) with an anhydride of formula (11)
##STR00023##
wherein X is a nitro group or halogen, to provide intermediate bis(phthalimide)s of the formulas (12a) and (12b)
##STR00024##
wherein G, Q, M, q, m, p, and t are as described in formula (1) and (1a) and X is as described in formula (11). The polyamine (8), preferably (8'), can be present in the reaction in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol % to achieve increased branching. An optional catalyst or optional monofunctional chain terminating agent as described above can be present during imidization.
[0049] The bis(phthalimide)s (12a) and (12b) are reacted with an alkali metal salt of a dihydroxy aromatic compound of formula (13)
AMO--Z--OAM (13)
wherein AM is an alkali metal and Z is as defined above, to provide the branched polyetherimide. Polymerization conditions effective to provide the branched polyimides are generally known, and can be conducted in a solvent as described above. This polymerization can also be conducted in the melt, for example at 250 to 350.degree. C., where a solvent is generally not present.
[0050] In another embodiment, an anhydride of formula (11), for example 4-chlorophthalic anhydride (Cl-PA), is reacted with meta-phenylenediamine (mPD) to produce the intermediate bis(phthalimide) (Cl-PAMI). Alternatively, a polyamine of formula (8'), for example a triamine such as 2,4,4'-triaminodiphenyl ether (TADE), can be reacted with 4Cl-PA to produce a reactive tris-imide branching agent. These reactions are illustrated in Scheme 2a.
##STR00025##
The reactive tris-imide branching agent can then be reacted with the alkali metal salt of a dihydroxy aromatic compound (13) to provide the branched polyimide. In an exemplary embodiment, the intermediate bis(phthalimide) of formula (12a) is prepared by reacting Cl-PAMI with BPA, which can then be reacted with the alkali metal salt of a dihydroxy aromatic compound of formula (13) to provide the branched polyimide. This embodiment is shown in Scheme 2b.
##STR00026##
In other embodiments, other branching phenols such as THPE can be reacted with Cl-PAMI to provide a tris(phthalimide) intermediate a shown in Scheme 2c. The tris(phthalimide) intermediate can then be reacted with the alkali metal salt of a dihydroxy aromatic compound of formula (13) to provide the branched polyimide.
##STR00027##
[0051] The branched polyimide can have one or more of the following properties. The branched polyimide can have a T.sub.g greater than 100.degree. C., preferably 100 to 395.degree. C., more preferably 180 to 280.degree. C., even more preferably 200 to 250.degree. C. The branched polyimide can have an average branch molecular weight (M.sub.b) of 12,000 to 50,000 grams per mole (g/mol), preferably 15,000 to 40,000 g/mol, more preferably 23,000 to 38,000 g/mol, as determined by size exclusion chromatography or proton nuclear magnetic resonance. In some embodiments, the branched polyimide can have a viscosity of greater than 25,000 pascal-seconds at a frequency of 0.1 radians per second. The branched polyimide can have a polydispersity (PDI) of 1.5 to 3.0, as determined by size exclusion chromatography multi-angle light scattering (SEC-MALS). The branched polyimide can have a melt index of 0.1 to 10 grams per minute (g/min), as measured by American Society for Testing Materials (ASTM) D1238 at 340 to 370.degree. C., using a 6.7 kilogram (kg) weight. The branched polyimide can have a weight average molecular weight (M.sub.w) of 1,000 to 150,000 g/mol, or 10,000 to 80,000 g/mol, or 20,000 to 60,000 g/mol, as measured by gel permeation chromatography (GPC), using polystyrene standards, light scattering, and/or triple point detector. In some embodiments, the branched polyimide can have an intrinsic viscosity greater than 0.2 deciliters per gram (dL/g), or, more preferably, 0.35 to 0.7 dL/g, as measured in m-cresol at 25.degree. C.
[0052] In a specific embodiment, the branched polyimide can have less than 5 wt %, or less than 3 wt %, or less than 1 wt %, or less than 0.5 wt % of a gel. The gel can be observed visually. In an embodiment, no gel is observable.
[0053] The branched polyimide can have a UL94 rating of V-1 or better, as measured following the procedure of Underwriter's Laboratory Bulletin 94 entitled "Tests for Flammability of Plastic Materials for Parts in Devices and Appliances" (ISBN 0-7629-0082-2), Fifth Edition, Dated Oct. 29, 1996, incorporating revisions through and including Dec. 12, 2003. In an embodiment, the branched polyimide has a UL94 rating of V-0 or V-1 at a thickness of 0.2, 0.3, 0.5, 0.6, 0.75, 0.9, 1, 1.2, 1.5, 2, 2.5, or 3 mm. In some embodiments, the branched polyimide has a UL94 rating of V-0 at a thickness of 0.3, 0.5, 0.75, 0.9, 1, 1.5, 2, or 3 mm. In a preferred embodiment, the branched polyimide has a UL94 rating of V-0 at a thickness of 0.5 mm or of 1.5 mm. In another preferred embodiment, the branched polyimide can have a flame retardance that is greater than or equal to the same polyimide manufactured without the polyamine (8) or (8').
[0054] As described above, the polyamines (8), preferably (8'), and diamines (10) are reacted in combination, wherein the polyamine is present in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol % to achieve increased branching and increased PDI. Under these conditions, it is possible that both branched and unbranched poly(imides) are formed, to provide a polyimide composition comprising the branched polyimides and a second polyimide that is not the same as the branched polyimide. This second polyimide is generally an unbranched polyimide that comprises more than 1, for example 5 to 1000, or 5 to 500, or 10 to 100, structural units of formula (15)
##STR00028##
wherein V and R are as described in formula (1) and (1'). It is also possible to combine the branched polyimide or polyimide composition with a second polyimide that is separately manufactured, and contains a different degree of branching or no branching, to obtain the polyimide composition. In an embodiment, the polyimide composition includes 1 to 99 wt %, or 10 to 90 wt %, or 0.1 to 20 wt %, or 0.5 to 10 wt %, or 1 to 5 wt % of a branched polyimide and 99 to 1 wt %, or 90 to 10 wt %, or 99.9 to 80 wt %, or 99.5 to 90 wt %, or 99 to 95 wt % of a second polyimide.
[0055] Similarly, both branched and unbranched poly(etherimides) can be formed, to provide a polyetherimide composition comprising the branched polyetherimides (1a) or (1a') and a second polyetherimide that is not the same as the branched polyetherimides. This second polyetherimide is generally an unbranched polyetherimide that comprises more than 1, for example 5 to 1000, or 5 to 500, or 10 to 100, structural units of formula (16)
##STR00029##
wherein Z and R are as described in formula (1a) and (1a'). In an embodiment, the polyetherimide composition includes 1 to 99 wt %, or 10 to 90 wt %, or 0.1 to 20 wt %, or 0.5 to 10 wt %, or 1 to 5 wt % of a branched polyetherimide (1a) or (1a') and 99 to 1 wt %, or 90 to 10 wt %, 99.9 to 80 wt %, or 99.5 to 90 wt %, or 99 to 95 wt % of a second polyetherimide.
[0056] It is also possible to combine the polyimide composition or the polyetherimide compositions with a third polymer that is not the same as the branched polyimide and second polyimide (or the branched polyetherimide and second polyetherimide). Such polymer compositions can include 1 to 99 wt % of the polyimide or polyetherimide composition and 1 to 99 wt % of the third polymer, or 10 to 90% of the polyimide or polyetherimide composition and 10 to 90 wt % of the third polymer.
[0057] Illustrative examples of third polymers include a polyacetal, poly(C.sub.1-6 alkyl)acrylate, polyacrylamide, polyacrylonitrile, polyamide, polyamideimide, polyanhydride, polyarylene ether, polyarylene ether ketone, polyarylene ketone, polyarylene sulfide, polyarylene sulfone, polybenzothiazole, polybenzoxazole, polybenzimidazole, polycarbonate, polyester, poly(C.sub.1-6 alkyl)methacrylate, polymethacrylamide, cyclic olefin polymer, polyolefin, polyoxadiazole, polyoxymethylene, polyphthalide, polysilazane, polysiloxane, polystyrene, polysulfide, polysulfonamide, polysulfonate, polythioester, polytriazine, polyurea, polyurethane, vinyl polymer, or a combination comprising at least one of the foregoing.
[0058] The branched polyimide, the polyimide composition, the polyetherimide composition, and the polymer composition can include various additives ordinarily incorporated into compositions of this type, with the proviso that any additive is selected so as to not significantly adversely affect the desired properties of the composition. Exemplary additives include antioxidants, thermal stabilizers, light stabilizers, ultraviolet light (UV) absorbing additives, quenchers, plasticizers, lubricants, mold release agents, antistatic agents, visual effect additives such as dyes, pigments, and light effect additives, flame resistances, anti-drip agents, and radiation stabilizers. Other useful additives include carbon nanotubes, exfoliated nanoclays, carbon nanowires, carbon nanospheres, carbon-metal nanospheres, carbon nanorods, carbon-metal nanorods, nanoparticles, or insoluble polymers. Combinations of additives can be used. The foregoing additives can be present individually in an amount from 0.005 to 10 wt %, or combined in an amount from 0.005 to 20 wt %, preferably 0.01 to 10 wt %, based on the total weight of the composition. Particulate fillers and reinforcing fillers can also be present.
[0059] A wide variety of articles can be manufactured using the branched polyimide, the polyimide compositions, the polyetherimide compositions, and the polymer compositions, for example articles of utility in automotive, telecommunication, aerospace, electrical/electronics, battery manufacturing, wire coatings, transportation, food industry, and healthcare applications. Such articles can include films, fibers, foams (both open- and closed-cell foams), thin sheets, small parts, coatings, fibers, preforms, matrices for polymer composites, or the like. In a particular embodiment, the article is an open- or closed-cell foam, preferably a closed-cell foam. The articles can be extruded or molded, for example injection molded. In some embodiments, the articles can be made by an additive manufacturing method, for example three dimensional printing. Components for electronic devices and components for sterilizable medical articles are preferably mentioned. Thin-wall components manufactured by injection molding are also preferred, such as a wall having a thickness from 0.1 to 10 millimeters (mm), or 0.2 to 5 mm, or 0.5 to 2 mm. In some embodiments, a film can be manufactured by solution-casting or melt processing the branched polyimide, the polyimide compositions, the polyetherimide compositions, and the polymer compositions described herein.
[0060] The polyimides and polyetherimides are further illustrated by the following non-limiting examples.
EXAMPLES
[0061] Table 1 list components that are used in the examples. Unless specifically indicated otherwise, the amount of each component is in weight percent in the following examples, based on the total weight of the composition.
TABLE-US-00001 TABLE 1 Component Description THPE 1,1,1-tris(4-hydroxyphenyl) ethane (Sigma-Aldrich) TADE 2,4,4'-triaminodiphenyl ether (TCI America) 4ClPA 4-chlorophthalic anhydride (Sigma-Aldrich) Pd/C 10 wt % palladium over carbon (Sigma-Aldrich) mPD m-phenylene diamine (Sigma-Aldrich) pPD p-phenylene diamine (Sigma-Aldrich) PA phthalic anhydride (SABIC) BPA bisphenol A (Sigma-Aldrich) CaO calcium oxide (Sigma-Aldrich) 4-AA 4-amino acetophenone (Sigma-Aldrich) TsOH p-toluenesulfonic acid (Sigma-Aldrich) oDCB o-dicholorobenzene (Sigma-Aldrich) BPA-DA 2,2-bis[4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride (4,4'-Bisphenol A dianhydride) (SABIC)
[0062] Physical testing of the compositions was conducted according to the following test methods and procedures. Unless indicated otherwise, all test standards set forth herein are the test standards in effect as of 2016.
[0063] Proton nuclear magnetic resonance (.sup.1H NMR) spectroscopy characterization was performed on a Varian Unity 400 at 399.98 MHz in deuterated chloroform.
[0064] Carbon nuclear magnetic resonance (.sup.13C NMR) spectroscopy characterization was performed on a Varian Unity 400 at 100 MHz in deuterated chloroform.
[0065] Glass transition temperature (T.sub.g) and melting temperature (T.sub.m) were determined using Differential Scanning Calorimetry (DSC) according to ASTM D3418. The test was performed using a TA Q1000 DSC instrument. In a typical procedure, a polymer sample (10-20 milligrams) was heated from 40 to 400.degree. C. at a rate of 20.degree. C./min, held at 400.degree. C. for 1 minute, cooled to 40.degree. C. at a rate of 20.degree. C./min, then held at 40.degree. C. for 1 minute, and the above heating/cooling cycle was repeated. The second heating cycle is usually used to obtain the T.sub.g and T.sub.m.
[0066] Average branch molecular weight was determined by size exclusion chromatography (SEC) and .sup.1H NMR spectroscopy. Chloroform size exclusion chromatography (SEC) provided absolute molecular weights using a Waters 1515 Isocratic HPLC Pump and Waters 717 plus Autosampler with Waters 2414 refractive index and Wyatt MiniDAWN MALS detectors (flow rate 1.0 mL min.sup.-1).
[0067] Polydispersity was determined by size exclusion chromatography-multiple angle light scattering SEC-MALS in chloroform using dn/dc=0.271 or determined by GPC using polystyrene standards (or a light scattering detector in combination with a refractive index detector, or a triple detector).
[0068] Weight average molecular weight (M.sub.w) was determined by SEC or determined by GPC using polystyrene standards (or a light scattering detector in combination with a refractive index detector, or a triple detector).
[0069] Torque was determined using a Haake.TM. torque rheometer from Thermo Scientific.
Example 1: Synthesis of tris((p-nitrophenoxy)phenyl) ethane (TNPE)
[0070] A general synthesis of (TNPE) is shown in Scheme 3, where R is 1,1,1-ethane.
##STR00030##
[0071] Specifically, 1,1,1-tris(4-hydroxyphenyl) ethane (20 g, 65.3 millimoles (mmol)), potassium carbonate (45.1 g), dimethylacetamide (115 milliliter (mL)), and toluene (58 mL) were charged to a three-necked, 500-mL, round-bottomed flask. A Dean-Stark trap with condenser, glass stir rod with Teflon blade and glass bearing, and rubber septa were attached to each of the three necks, respectively. Purging the whole setup with nitrogen for 20 minutes (min) provided an inert atmosphere. Next, the round-bottomed flask was lowered into a 180.degree. C. preheated oil bath resulting in a reflux of toluene to the Dean-Stark trap. Deprotonation of the phenol proceeded at 180.degree. C., and was monitored through collection of water in the Dean-Stark trap. Once water removal ceased, toluene removal proceeded by distillation through the Dean-Stark trap. A solution of 1-chloro-4-nitrobenzene (33.9 g, 215.5 mmol) in dimethylacetamide (115 mL) was charged drop-wise to the reaction. After addition of the 1-chloro-4-nitrobenzene, the resulting solution color changed from pale pink to dark brown. The reaction proceeded at 180.degree. C. overnight to afford a heterogeneous dark brown solution.
[0072] Filtration of the heterogeneous solution through a fritted funnel equipped with a Celite cake (2.5 cm.times.15 cm) resulted in a brown, transparent solution. Precipitation into 1.0 molar (M) NH.sub.4OH aqueous solution afforded a fine, yellow precipitate. The yellow precipitate was filtered and washed until a filtrate pH of 7 was obtained, and then the precipitate was dried in-vacuo at 120.degree. C. overnight. Recrystallization from ethyl acetate isolated the target compound in >90% overall yield. NMR spectroscopy and mass spectrometry confirmed the structure and purity.
Example 2: Synthesis of tris((p-aminophenoxy)phenyl) ethane (TAPE) via Pd--C Reduction
[0073] A general synthesis of tris((p-aminophenoxy)phenyl)-1,1,1-ethane (TAPE) is shown in Scheme 4 wherein R is 1,1,1-ethane.
##STR00031##
[0074] In particular, tris((p-nitrophenoxy)phenyl) ethane (60 g, 89.6 mmol) and tetrahydrofuran (270 mL) were charged to the Parr reactor, and subsequently purged with nitrogen for 20 minutes. Next, 10 wt % palladium over carbon (10 g) was added, and the reactor sealed and purged with nitrogen for 20 minutes. Three successive cycles of pressurizing the reactor to 100 psi with hydrogen ensured saturation of the solution with hydrogen, and the reaction proceeded under 100 psi of hydrogen pressure. After 24 hours, the pressure was released and the black, heterogeneous solution was recovered. Filtration of the heterogeneous solution through a fritted funnel equipped with a Celite cake (2.5 cm.times.15 cm) resulted in a brown, transparent solution. Evaporation of the solvent afforded the target compound as a light brown powder in a 98% yield.
[0075] The complete synthesis of tris((p-aminophenoxy)phenyl) 1,1,1 ethane (TAPE) is shown in Scheme 5.
##STR00032##
[0076] Further purification of TAPE was afforded by flash column chromatography on silica gel using ethyl acetate and hexanes, and further including tetrahydrofuran and triethylamine as co-eluents.
Example 3: Synthesis of tris((p-aminophenoxy)phenyl) ethane (TAPE) via Hydrazine Reduction
[0077] Tris((p-nitrophenoxy)phenyl) ethane (60 g, 89.6 mmol), 10 wt % palladium over carbon (10 g), and tetrahydrofuran (200 mL) were charged to a two-necked, round-bottomed, 500 mL flask. The flask was equipped with a condenser and addition funnel and subsequently purged with nitrogen for 20 minutes. A solution of hydrazine (537.6 mmol) in tetrahydrofuran (70 mL) was added to the addition funnel. Under a constant nitrogen purge, the round-bottomed flask was heated to 80.degree. C. Over the course of 1 h, the hydrazine solution was added dropwise to the round-bottomed flask. After addition, the reaction was maintained at 80.degree. C. and reflux for several hours until the reaction was completed (98% yield).
Example 4: Synthesis of tris(p-aminophenyl)benzene (TAPB)
[0078] The synthesis of tris(p-aminophenyl)benzene (TAPB) is shown in Scheme 6.
##STR00033##
[0079] The synthesis of 1,3,5-triphenylbenzene, and related derivatives, is by an acid catalyzed cyclotrimerization of acetophenone or a derivative thereof, which provides a one-step method for making a triamine without hydrogenation of a nitro derivative. The trimerization of 4-amino acetophenone to give 1,3,5-tris(4-aminophenyl)benzene in a one-pot reaction is low yielding (.about.5%) due to the unfavorable side reaction which yields a polyamine. This can be overcome by protecting the amine group during the reaction. Protecting the amine by preparing an ammonium salt results in an increase in product yield.
Example 5. Synthesis of tris(p-aminophenyl)benzene (TAPB)
[0080] Tris(p-aminophenyl)benzene (TAPB) is synthesized from an ammonium salt of 4-amino acetophenone (4-AA), as shown in Scheme 7.
##STR00034##
[0081] An acetone solution of 4-AA is added to an acetone solution of p-toluenesulfonic acid (TsOH), where the TsOH is provided in a 20% molar excess of the 4-AA. Immediate precipitation of the ammonium tosylate salt is observed. This is filtered, dried (1.4 g), and placed in a round bottom flask with TsOH (20%, as a catalyst) and heated at 160.degree. C. overnight. The resulting dark red material is washed with a saturated aqueous NaHCO.sub.3 solution, extracted into methylene chloride, and dried with MgSO.sub.4. The solvent was removed by rotary evaporation under vacuum to give a crude red product. A purified yellow product is obtained after flash column chromatography on silica gel with ethyl acetate and hexanes as eluents (elution gradient of 0-40% ethyl acetate over 25 minutes), to obtain the TAPB (.about.20% yield).
Example 6: Synthesis of Long-Chain Branched Polyetherimides (LCB-PEIs)
[0082] A synthesis of a 45 kg/mol polyetherimide branched with 1 mol % of TAPE follows as an example. TAPE (0.12 g, 0.34 mmol), m-phenylene diamine (5.52 g, 51.0 mmol), and o-dichlorobenzene (75 mL) were charged to a three-necked, 500-mL, round-bottomed flask. The flask was then equipped with a rubber septum, glass stir rod with Teflon blade, and Dean-Stark trap. A condenser completed the set up on the Dean-Stark trap, and the contents purged with nitrogen for 20 minutes. The round-bottomed flask was then heated to 100.degree. C. to generate a homogeneous solution. Next, BPA-DA (26.3 g, 50.6 mmol), phthalic anhydride (PA) (0.691 g, 4.6 mmol), and oDCB (25 mL) were added. The flask was then heated to 180.degree. C., where the reaction proceeded for 18 hours. Devolatilization of the solvent and thermal imidization was completed utilizing a melt kneader operating at 380.degree. C., resulting in an orange, transparent product.
Example 7: Synthesis of Long-Chain Branched Polyetherimides (LCB-PEIs)
[0083] The synthetic procedure of Example 6 was followed to prepare larger batches of long-chain branched polyetherimides. Specifically, a 500 mL, 3-neck round, bottom-flask connected to a stirrer and a Dean-Stark trap was charged with mPD (16.8 g, 155 mmol) and TAPE via an addition funnel. Degassed oDCB (120 mL) was then added and residual solids from the sides of the flask were rinsed into the solution. While stirring, the flask was lowered into an oil bath pre-heated to 80.degree. C. The mixture of amines was stirred for 30 minutes under N.sub.2, and the temperature was slowly increased to 100.degree. C. over the course of 15 minutes. A flashlight was used to ensure the complete solvation of the amines. BPA-DA (80 g, 154 mmol) and PA (2.1 g, 14 mmol) were then slowly added to the amine solution using additional oDCB (120 mL). The temperature of the oil-bath was then raised to 160.degree. C. Stirring was gradually slowed to a halt as the heterogeneous mixture began to react and clump, and water vapor begins to release at a temperature of around 140.degree. C. Once the clumped material begins to dissolve, stirring is resumed and a homogeneous amber-yellow solution is obtained after 20-30 hours. The temperature is then increased to 190.degree. C. and a stoichiometric analysis is performed after 1.5-3 hours. For example, if the polymer media is found to be rich in mPD by 0.5-1 mol %, then an appropriate additional amount of BPA-DA can be added. The temperature is then lowered to 170.degree. C. and after 24 hours of heating, a second stoichiometric analysis is performed to evaluate if stoichiometric conditions are met. The polymer is then let to heat for an additional 24 h if desired. The reaction product is subsequently isolated by precipitation with hexanes (2-3 L) in an industrial blender or in the vessel of a Haake rheometer.
Example 8: Synthesis of bis(2,4-dinitrophenoxy)bisphenol A
[0084] Synthesis of bis(2,4-dinitrophenoxy)bisphenol A follows similar procedures as Example 1. Bisphenol A (24.4 g, 107 mmol), potassium carbonate (24 g), dimethylacetamide (150 mL), and toluene (75 mL) were charged to a three-necked, 500-mL, round-bottomed flask. A dean-stark trap with condenser, glass stir rod with Teflon blade and glass bearing, and rubber septa was attached to the three necks, respectively. Purging the whole setup with nitrogen for 20 min provided an inert atmosphere. Next, the round-bottomed flask was lowered into a 180.degree. C. preheated oil bath resulting in a reflux of toluene to the Dean-Stark trap. Deprotonation of the phenol proceeded at 180.degree. C., and was monitored through collection of water in the Dean-Stark trap. Once water removal ceased, toluene removal proceeded by distillation through the Dean-Stark trap. A solution of 1-chloro-2,4-dinitrobenzene (45.5 g, 225 mmol) in dimethylacetamide (150 mL) was charged drop-wise to the reaction. After addition of the 1-chloro-2,4-dinitrobenzene, the resulting solution color changed from pale pink to dark brown. The reaction proceeded at 180.degree. C. overnight to afford a heterogeneous dark brown solution.
[0085] Filtration of the heterogeneous solution through a fritted funnel equipped with a Celite cake (2.5 cm.times.15 cm) resulted in a brown, transparent solution. Precipitation into 1.0 M NH.sub.4OH aqueous solution afforded a fine, yellow precipitate. The yellow precipitate was filtered and washed until a filtrate pH of 7 was obtained, and then the precipitate was dried in-vacuo at 120.degree. C. overnight. (92% yield).
Example 9: Synthesis of bis(2,4-diaminophenoxy)bisphenol A
[0086] Reduction of bis(2,4-dinitrophenoxy)bisphenol A to bis(2,4-diaminophenoxy)bisphenol A follows similar procedures as Example 2. Bis(2,4-dinitrophenoxy)bisphenol A (55 g, 98.1 mmol) and tetrahydrofuran (250 mL) were charged to a Parr reactor, and subsequently purged with nitrogen for 20 minutes. Next, 10 wt % palladium over carbon (10 g) was added, and the reactor sealed and purged with nitrogen for 20 minutes. Three successive cycles of pressurizing the reactor to 100 psi with hydrogen ensured saturation of the solution with hydrogen, and the reaction proceeded under 100 psi of hydrogen pressure. After 24 hours, the pressure was released and the black, heterogeneous solution was recovered. Filtration of the heterogeneous solution through a fritted funnel equipped with a Celite cake (2.5 cm.times.15 cm) resulted in a brown, transparent solution. Evaporation of the solvent afforded the target compound as a light brown powder.
Example 10: Synthesis of bis(3,5-diaminobenzylcarboxy)hydroquinone
[0087] Synthesis of multi-functional aryl-amines based on aromatic esters is illustrated by the synthesis of bis(3,5-diaminobenzylcarboxy)hydroquinone and is shown in Scheme 10.
##STR00035##
[0088] 3,5-Dinitrobenzoic acid (4.0000 g, 18.856 mmol) and hydroquinone diacetate (1.8307 g, 9.428 mmol) were charged to a one-neck, 100-mL, round-bottomed flask. The flask was then equipped with a metal stir rod with metal blade, a t-neck glass adapter with a gas inlet, stir rod adapter, and outlet, and condensing tube connected to a collection flask. The contents were purged with nitrogen for 20 minutes. Under constant nitrogen flow, the round-bottomed flask was then heated to 160.degree. C. to generate a melt. The reaction proceeded for 8 h and the removal of acetic acid helped monitor the reaction progress. The resulting materials were collected, washed, and purified. Reduction of the nitro group followed the same procedure as discussed in Example 3.
Example 11: Synthesis of Long-Chain Branched Polyetherimides
[0089] Synthesis of long-chain branched follows similar procedures as Example 6. The synthesis of a 36 kg/mol polyetherimide branched with 1 mol % of bis(2,4-diaminophenoxy)bisphenol A follows.
[0090] Bis(2,4-diaminophenoxy)bisphenol A (0.267 g, 0.26 mmol), m-phenylene diamine (5.57 g, 51.4 mmol), and o-dichlorobenzene (75 mL) were charged to a three-necked, 500-mL, round-bottomed flask. The flask was then equipped with a rubber septum, glass stir rod with Teflon blade, and Dean-Stark trap. A condenser completed the set up on the Dean-Stark trap, and the contents were purged with nitrogen for 20 minutes. The round-bottomed flask was then heated to 100.degree. C. to generate a homogeneous solution. Next, BPA-DA (26.3 g, 50.6 mmol), phthalic anhydride (0.494 g, 3.3 mmol), and o-dichlorobenzene (25 mL) were added. The flask was then heated to 180.degree. C., where the reaction proceeded for 18 hours. Devolatilization of the solvent and thermal imidization was completed utilizing a melt kneader operating at 380.degree. C., resulting in an orange, transparent product.
Example 12: Long-Chain Branched Polyetherimides
[0091] Table 2a shows the M.sub.w for a series of long-chain branched polyetherimides synthesized in the presence of different amounts of TAPE, as determined by SEC-MALS in chloroform with dn/dc=0.271. Table 2b shows the absolute M.sub.w for the same PEIs, as determined by a triple point detector in chloroform.
TABLE-US-00002 TABLE 2a TAPE (mol %) Targeted M.sub.w 0 0.11 0.26 0.56 0.92 1.1 1.3 1.5 2 2.5 3 (kg/mol) Measured M.sub.w (kg/mol).sup.a 45 40.2 42.4 39.9 39.7 33.5 40.7 40.7 44.2 36.4 35.7 39.1 40 30.3 36.8 29.8 36.1 56.2 36.2 38.6 35.4 31.2 40.0 41.3 35 31.6 36.0 39.0 33.6 34.4 37.6 28.7 32.7 N/A N/A N/A 30 37.2 N/A N/A 30.0 28.4 27.7 29.1 26.8 N/A N/A N/A .sup.aStd. dev. = 5%
TABLE-US-00003 TABLE 2b TAPE (mol %) Targeted M.sub.w 0 0.11 0.26 0.56 0.92 1.1 1.3 1.5 2 2.5 3 (kg/mol) Measured Absolute M.sub.w (kg/mol).sup.a 45 47.8 53.8 48.0 43.1 38.2 49.5 50.0 50.3 48.3 45.7 47.2 40 33.1 44.1 42.1 43.8 62.2 47.6 43.1 45.0 42.5 44.5 42.8 35 36.2 41.3 49.4 39.9 36.2 39.4 31.3 36.9 N/A N/A N/A 30 32.4 N/A N/A 33.0 34.2 32.6 32.6 33.4 N/A N/A N/A .sup.aStd. dev. = 5%
[0092] Minimal changes in polydispersity are observed at incorporations below 1.0 mol % TAPE, but at relatively higher incorporations of the triamine, the polydispersity dramatically increases. With targeted M.sub.w closely matching measured values, a direct correlation between branching and dispersity is achieved.
[0093] Coupling the SEC molecular weight information with .sup.1H NMR spectroscopy enabled determination of the average molecular weight of the branches (M.sub.b) as shown in FIG. 3, where the top solid line corresponds to predicted values for M.sub.b of 30 kg/mol and the bottom solid line corresponds to predicted values for M.sub.b of 15 kg/mol. Without being bound by theory, at higher incorporations of polyamine, greater deviations between the experimental data and the theoretical predictions are observed, which can be as a result of increased cyclization. Furthermore, the M.sub.b remains greater than the molecular weight of the entanglements (M.sub.e), which may indicate that these long-chain branches are of significant length to entangle. Overall, these results are consistent with long chain branching in the branched PEIs.
[0094] DSC was used to identify the thermal transitions of the LCB-PEIs, and the results are shown in Table 3.
TABLE-US-00004 TABLE 3 TAPE (mol %) M.sub.w 0 0.11 0.26 0.56 0.92 1.1 1.3 1.5 2 2.5 3 (kg/mol) Measured T.sub.g (.degree. C.) 45 214 215 213 214 214 213 210 216 209 209 207 40 212 214 212 220 214 210 212 210 212 207 213 35 212 212 211 214 212 210 210 210 N/A N/A N/A 30 212 N/A N/A 212 212 210 208 N/A N/A N/A N/A
[0095] The observation of a glass transition temperature (T.sub.g) near 215.degree. C. correlated well to linear PEI controls. Without being bound by theory, increasing the amount of the branching agent may not significantly affect the onset of segmental motion. The relatively low concentration of long-chain branches (M.sub.b>>M.sub.e) may not significantly promote or retard the onset of segmental motion, and may allow for the use of these branched materials in typical PEI applications. However, introduction of the long-chain branches affects the rheological properties.
Example 13: Long-Chain Branched Polyetherimides
[0096] In an exemplary procedure, TADE, m-phenylene diamine, and BPA-DA were reacted in the presence of either phthalic anhydride or aniline as an endcapping agent. The LCB-PEI was obtained following imidization according to the procedure of Example 11.
[0097] DSC was used to identify the thermal transitions of the LCB-PEIs prepared using TADE and different endcapping agents, and the results are shown in Table 4.
TABLE-US-00005 TABLE 4 Target M.sub.w TADE Tg Endcapping (kg/mol) (mol %) (.degree. C.) Agent 38 0.1 216 PA 38 0.5 211 Aniline 38 1 214 PA 38 1.5 211 PA 40 3 215 Aniline 38 3 213 PA 38 4.4 212 Aniline
[0098] As shown in Table 4, the use of triamine branching agents such as TADE provides LCB-PEIs having glass transition temperatures from 210 to 215.degree. C. These results are similar to those observed with TAPE as branching agent and also correlated well to linear PEI controls.
Example 14: Properties of Long-Chain Branched Polyetherimides
[0099] Measurement of viscosity as a function of angular frequency provided insight into the processibility of the LCB-PEIs as compared to the linear analogues. Comparing samples with molecular weights within the SEC-instrumental margin of error helped to isolate the long-chain branching effects from the molecular weight effects. FIG. 4 shows the viscosity profiles of the LCB-PEIs having various branching densities.
[0100] Across the relevant processing window of 0.1 to 100 rad/s, the PEIs with a greater incorporation of branching agent (>2.0 mol %) demonstrated significant reductions in the observed melt viscosity. Without being held to theory, it is believed that the length and the dispersity of polymer chains and branches play an instrumental role in this observation. As noted above, the dispersity of the polymer increased with greater incorporations of TAPE. As a result, the lower molecular weight chains and branches may help to plasticize the melt and reduce the overall viscosity. In contrast, the lower incorporations of TAPE had an increased melt viscosity as compared to the linear analogue. The M.sub.b at these incorporations were relatively long and their distribution does not involve the molecular weights that plasticize the melt.
Example 15: Long-Chain Branched Polyetherimides
[0101] A second series of LCB-PEIs with varying amounts of TAPE were prepared on a small scale (ca. 4 g). Samples were prepared with 0, 0.3, 0.6, 1.0 and 3.0 mol % of TAPE, and each sample included 3.07 mol % of an endcapping agent. The solutions obtained were homogeneous, with the exception of the LCB-PEI prepared using 3.0 mol % TAPE, which was a dark brown gel-like material a few millimeters wide.
[0102] Table 5 shows the apparent weight average molecular weight (Apparent M.sub.w), absolute weight average molecular weight (Absolute M.sub.w), polydispersity (PDI), and PDI*, respectively, obtained for the second series of LCB-PEIs prepared with different amounts of TAPE.
TABLE-US-00006 TABLE 5 .sup..dagger-dbl.Apparent M.sub.w/ TAPE (mol %) .sup..dagger.Absolute M.sub.w (g/mol) PDI* (M.sub.z/M.sub.w) PDI (M.sub.w/M.sub.n) 0 53,552.sup..dagger-dbl./38,094.sup..dagger. 1.62.sup..dagger-dbl./1.61.sup..dagger. 2.68.sup..dagger-dbl./1.54.sup..dagger. 0.3 57,860.sup..dagger-dbl./42,305.sup..dagger. 1.72.sup..dagger-dbl./1.78.sup..dagger. 2.79.sup..dagger-dbl./1.62.sup..dagger. 0.6 56,731.sup..dagger-dbl./43,125.sup..dagger. 2.05.sup..dagger-dbl./2.07.sup..dagger. 2.91.sup..dagger-dbl./1.75.sup..dagger. 1.0 74,295.sup..dagger-dbl./58,400.sup..dagger. 2.46.sup..dagger-dbl./2.33.sup..dagger. 3.77.sup..dagger-dbl./1.98.sup..dagger. 3.0 163,529.sup..dagger-dbl./280,607.sup..dagger. 3.85.sup..dagger-dbl./6.55.sup..dagger. 7.60.sup..dagger-dbl./5.57.sup..dagger. .sup..dagger-dbl.GPC data based on polystyrene standard; .sup..dagger.Data from GPC coupled with triple-detector Note: GPC integrations for apparent M.sub.w include the small overlapping lower M.sub.w peaks. Hence, higher than theoretical PDI is observed but the trends are consistent.
[0103] The data in Table 5 shows an exponential increase in the M.sub.w correlated to an increase in the amount of branching agent. This increase is also reflected in the polydispersity index (PDI) and PDI* values, which indicated a broader M.sub.w distribution and more branching, respectively, as the amount of TAPE is increased in the samples.
Example 16: Long-Chain Branched Polyetherimides
[0104] A third series of LCB-PEIs with varying amounts of TAPE were prepared on a larger scale (ca. 80 g). LCB-PEIs were prepared with 0, 0.5, and 1.5 mol % TAPE, and each sample included 3.07 mol % of an endcapping agent. After 21 hours of reaction time, the control sample (0 mol % TAPE) was a clear amber solution, while the samples prepared with 0.5 and 1.5 mol % TAPE were dark gels interspersed throughout the solution media. These gels were found to be insoluble even in a 30% hexafluoroisopropanol in methylene chloride solution. While GPC data on the soluble mixture does show an increase in M.sub.w and PDI* with higher amounts of TAPE, it is to be noted that a homogeneous branched polymer mixture is not obtained when the reagents are first combined in oDCB and then heated with stirring. This method, at least at the larger scale, appears to cause higher molecular weight PEIs to rapidly form in isolated regions and results in gel spots.
[0105] The torque was measured after 15 minutes for each of the LCB-PEIs of the third series of samples. The torque increased based on the increase in M.sub.w, which in turn was as a result of higher incorporation of TAPE loading (FIG. 5).
Example 17: Long-Chain Branched Polyetherimides
[0106] To achieve branching homogeneity in the polymer mixtures, an alternate method of preparation was explored. A fourth series of LCB-PEIs were prepared on a smaller scale (ca. 4 g). The LCB-PEIs each included 1 wt % of TAPE, with variations between the M.sub.w for each of the PEIs. Three of the samples were prepared from amines that were pre-dissolved in a heated oDCB solution, whereas the other three samples were prepared by way of a "regular addition"-all reagents were combine in oDCB and then heated with stirring. The appearance of the resulting polymer mixtures after reaching stoichiometric conditions were evaluated and physical properties were identified by GPC. These results are shown in Table 6.
TABLE-US-00007 TABLE 6 Entry Method Gel spots M.sub.w (g/mol) PDI* PDI 1 Regular yes 49,071 2.046 3.12 2 Pre-dissolved no 58,397 2.295 3.69 3 Pre-dissolved no 56,311 2.052 3.42 4 Regular yes 64,054 2.008 3.36 5 Pre-dissolved no 68,346 2.222 3.92 6 Pre-dissolved no 66,393 2.282 3.81 Note: GPC data collected at 32.degree. C. Values are underestimated by ~5 to 9 kg/mol, but trends are consistent. The PDI values are larger as the smaller traces of low M.sub.w peaks were integrated as well.
[0107] The polymer mixtures prepared via the pre-dissolved amines method appeared homogeneous and lighter in color, and gels did not form. The samples prepared by the regular addition method--where the reagents were combined and heated together with oDCB, had a darker color and gel spotting was observed. In addition, the M.sub.w of the PEIs prepared by the pre-dissolved amines method were heavier than the PEIs prepared by the regular addition method by 4 to 9 kg/mol.
[0108] An additional large scale preparation (ca. 80 g) of LCB-PEIs was used to determine the efficacy of the pre-dissolved amines method on the synthesis of LCB-PEIs with 1 mol % of TAPE. After 21 hours of reaction time and reaching stoichiometric conditions, no gels were observed. In contrast, a larger scale preparation of LCB-PEIs that were prepared by the regular addition method, wherein all reagents are added cool and heated together with oDCB, formed gels with as little as 0.5 mol % TAPE. Upon closer inspection, smaller dark specks can be observed in the polymer media, but this could very well be the charred unreacted 5% impurity present in the triamine. The GPC and torque data are displayed in FIG. 6. The M.sub.w of the LCB-PEIs obtained with 1 mol % TAPE using the pre-dissolved amines method is similar to the data obtained for the LCB-PEIs prepared with 1.5 mol % TAPE using the regular addition method. This further validates that the pre-dissolved amines method provides more efficient branching, which also translated into a decreased amount of gel formation. Pictures of the polymer media at the end of these reactions are shown in FIG. 7.
Example 18. Endcapping Agent
[0109] With the method of preparation for LCB-PEIs shown to be important in preventing the formation of gel spots, LCP-PEIs with varying degrees of branching were synthesized at a targeted M.sub.w by the incorporation of additional amounts of endcapping agent (phthalic anhydride) to cap the additional endgroups introduced by the TAPE branching agent. A series of LCB-PEIs were prepared with 0.3, 0.5, 1.0, and 1.5 mol % TAPE on an 80 g scale by pre-dissolving the amines in hot oDCB followed by the addition of anhydrides. The polymers were heated for 2 days under stoichiometric conditions and then precipitated as white flakes in hexanes in a large industrial blender. GPC measurements on the stoichiometric films after 1 day of heating shows that the M.sub.w was centered around 51 kg/mol.+-.3 kg/mol (Table 7). The formation of gels was not observed in any of the samples. Tiny black specks were observed for 1.0 and 1.5 mol % TAPE samples, but are attributed to charred triamine impurities (presumably more visible in these two batches due to higher loadings), as described herein.
TABLE-US-00008 TABLE 7 TAPE (mol %) M.sub.w (g/mol) PDI* PDI 0 54,437 1.597 2.08 0.3 52,087 1.654 2.10 0.5 48,203 1.674 2.11 1.0 55,434 1.857 2.31 1.5 48,770 1.916 2.35 M.sub.w is from integration of the major GPC trace, and excluding the smaller low M.sub.w peaks. GPC data is from stoichiometric films after 1 day of heating.
[0110] The experiments revealed that LCB-PEIs with varying degrees of branching at a targeted M.sub.w can be prepared without the formation of gels in the polymer media. This can be successfully done using the pre-dissolved amines method.
[0111] In a separate set of experiments, no differences were observed in the M.sub.w for LCB-PEIs prepared using different endcapping agents (PA versus aniline).
[0112] This disclosure is further illustrated by the following aspects.
[0113] Aspect 1. A branched polyimide of formula (1), preferably formula (1'), wherein G is a group having a valence of t, present in an amount of 0.01 to 20 mol %, or 0.1 to 20 mol %, or 0.5 to 10 mol %, or 1.0 to 5 mol %, or 1.5 to 4 mol %, or 0.01 to 2 mol %, each Q is independently the same or different, and is a divalent C.sub.1-60 hydrocarbon group, each M is independently the same or different, and is --O--, --C(O)--, --OC(O)--, --OC(O)O--, --NHC(O), --(O)CNH--, --S--, --S(O)--, or --S(O).sub.2--, D is a phenylene, each V is independently the same or different, and is a tetravalent C.sub.4-40 hydrocarbon group, each R is independently the same or different, and is a C.sub.1-20 divalent hydrocarbon group, q is 0 or 1, m is 0 or 1, d is 0 or 1, p is 1 or 2, t is 2 to 6, preferably 2 to 4, and each n is independently the same or different, and is 1 to 1,000, preferably 1 to 500, or 1 to 100, the total of all values of n is greater than 4, or greater than 10, or greater than 20, or greater than 50, or greater than 100, or greater than 250.
[0114] Aspect 2. The branched polyimide of aspect 1, wherein when t is 2, G is --O--, --C(O)--, --OC(O)--, --(O)CO--, --NHC(O), --(O)CNH--, --S--, --S(O)--, --S(O).sub.2--, or --P(R.sup.a)(O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl; or when t is 3, G is a nitrogen, phosphorus, or pentavalent P(O); or G is a C.sub.1-60 hydrocarbon group having a valence of t.
[0115] Aspect 3. The branched polyimide of aspect 1 or 2, wherein G is --O-- when m is 0, pentavalent P(O), a C.sub.6-50 hydrocarbon having at least one aromatic group, a C.sub.2-20 aliphatic group, a C.sub.4-8 cycloaliphatic group, or a C.sub.3-12 heteroarylene, or a polymer moiety.
[0116] Aspect 4. The branched polyimide of any one or more of aspects 1 to 3, wherein q is 1, Q is a C.sub.6-20 arylene, m is 1, and M is --O--.
[0117] Aspect 5. The branched polyimide of any one or more of aspects 1 to 4, wherein V is of the formula (2), wherein W is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, --P(R.sup.a)(.dbd.O)-- wherein R.sup.a is a C.sub.1-8 alkyl or C.sub.6-12 aryl, --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof, or a group of the formula --O--Z--O-- wherein Z is an aromatic C.sub.6-24 monocyclic or polycyclic moiety optionally substituted with 1 to 6 C.sub.1-8 alkyl groups, 1 to 8 halogen atoms, or a combination comprising at least one of the foregoing, provided that the valence of Z is not exceeded.
[0118] Aspect 6. The branched polyimide of any one or more of aspects 1 to 5, wherein the branched polyimide is a branched polyetherimide of formula (1a), preferably (1a'), wherein each Z is independently an aromatic C.sub.6-24 monocyclic or polycyclic moiety optionally substituted with 1 to 6 C.sub.1-8 alkyl groups, 1 to 8 halogen atoms, or a combination comprising at least one of the foregoing, provided that the valence of Z is not exceeded.
[0119] Aspect 7. The branched polyimide of aspect 6, wherein Z is a divalent group of formula (7a) wherein J is --O--, --S--, --C(O)--, --SO.sub.2--, --SO--, or --C.sub.yH.sub.2y-- wherein y is an integer from 1 to 5 or a halogenated derivative thereof and R is m-phenylene, p-phenylene, bis(4,4'-phenylene)sulfone, bis(3,4'-phenylene)sulfone, or bis(3,3'-phenylene)sulfone.
[0120] Aspect 8. A method for the manufacture of the branched polyimide of any one or more of aspects 1 to 7, the method comprising: reacting a polyamine of formula (8) and a diamine of formula (10) with a dianhydride of formula (9) or (9a) in a solvent and under conditions effective to provide the branched polyimide, wherein G, Q, M, D, R, V, q, m, d, p and t are as defined in any one or more of aspects 1 to 7.
[0121] Aspect 9. The aspect of aspect 8, further comprising pre-dissolving the polyamine and the diamine in the solvent before adding the dianhydride.
[0122] Aspect 10. A method for the manufacture of the branched polyimide of any one or more of aspects 1 to 7, wherein the branched polyimide is a branched polyetherimide, the method comprising: reacting a polyamine of formula (8) and a diamine of formula (10) with an anhydride of the formula (11) wherein X is a nitro group or halogen, to provide intermediate bis(phthalimide)s of the formulas (12) and (12a); and reacting the bis(phthalimide)s with an alkali metal salt of a dihydroxy aromatic compound of formula (13) wherein AM is an alkali metal, to provide the branched polyetherimide, wherein G, Q, M, D, R, V, q, m, d, p, and t are as defined in any one or more of aspects 1 to 7.
[0123] Aspect 11. The method of any one or more of aspects 8 to 10, wherein the polyamine is of the formulas (8b), (8k), (8r), (8s), or (8t).
[0124] Aspect 12. The method of any one or more of aspects 8, 9, or 11, wherein the dianhydride is 2,2-bis[4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride, and the diamine is bis-(4-aminophenyl) sulfone or m-phenylenediamine.
[0125] Aspect 13. The branched polyimide of any one or more of claims 1 to 7, or made by the method of any one or more of claims 8 to 12, having one or more of the following properties: a T.sub.g greater than 100.degree. C., preferably 100 to 395.degree. C., more preferably 180 to 280.degree. C., even more preferably 200 to 250.degree. C.; or an average branch molecular weight of 12,000 to 50,000 g/mol, preferably 15,000 to 40,000 g/mol, more preferably 23,000 to 38,000 g/mol; or a viscosity of greater than 25,000 pascal seconds at a frequency of 0.1 radians per second; or less than 5 wt %, or less than 3 wt %, or less than 1 wt %, or less than 0.5 wt % of a gel, based on the total weight of the branched polyimide; or a polydispersity of 1.5 to 3.0, as determined by size exclusion chromatography multi-angle light scattering; a UL94 rating that is the better than or equal to a UL94 rating of the same polyimide manufactured without the polyamine.
[0126] Aspect 14. An article comprising the branched polyimide of any one or more of aspects 1 to 7 or 13, or made by any one or more of the methods of aspects 8 to 12.
[0127] Aspect 15. A polyimide composition, comprising 1 to 99 wt %, or 10 to 90 wt %, 0.1 to 20 wt %, or 0.5 to 10 wt %, or 1 to 5 wt % of the branched polyimide of any one or more of aspects 1 to 7 or 13, or made by the method of aspects 8 to 12; and 1 to 99 wt %, or 10 to 90 wt %, or 0.9 to 80 wt %, or 99.5 to 90 wt %, or 99 to 95 wt % of a polyimide a second polyimide that is not the same as the branched polyimide; and wherein each amount is based on the total weight of the branched polyimide and the polyimide.
[0128] Aspect 16. An article comprising the polyimide composition of aspect 15.
[0129] Aspect 17. A polymer composition comprising the polyimide composition of aspect 14; and a third polymer different from the branched polyimide and the second polyimide.
[0130] Aspect 18. The polymer composition of aspect 17, wherein the second polymer is a polyacetal, poly(C.sub.1-6 alkyl)acrylate, polyacrylamide, polyacrylonitrile, polyamide, polyamideimide, polyanhydride, polyarylene ether, polyarylene ether ketone, polyarylene ketone, polyarylene sulfide, polyarylene sulfone, polybenzothiazole, polybenzoxazole, polybenzimidazole, polycarbonate, polyester, polyetherimide, polyimide, poly(C.sub.1-6 alkyl)methacrylate, polymethacrylamide, cyclic olefin polymer, polyolefin, polyoxadiazole, polyoxymethylene, polyphthalide, polysilazane, polysiloxane, polystyrene, polysulfide, polysulfonamide, polysulfonate, polythioester, polytriazine, polyurea, polyurethane, vinyl polymer, or combination thereof.
[0131] Aspect 19: An article comprising the polymer composition of aspect 17 or 18.
[0132] The article of aspects 16 or 19, wherein the article is a foam, preferably a closed cell foam.
[0133] The compositions, methods, and articles can alternatively comprise, consist of, or consist essentially of, any appropriate components or steps herein disclosed. The compositions, methods, and articles can additionally, or alternatively, be formulated so as to be devoid, or substantially free, of any steps, components, materials, ingredients, adjuvants, or species that are otherwise not necessary to the achievement of the function or objectives of the compositions, methods, and articles.
[0134] The singular forms "a," "an," and "the" include plural referents unless the context clearly dictates otherwise. "Or" means "and/or" unless clearly indicated otherwise by context. The endpoints of all ranges directed to the same component or property are inclusive and independently combinable. Disclosure of a narrower range or more specific group in addition to a broader range is not a disclaimer of the broader range or larger group. "Combination thereof" as used herein means that a combination comprising one or more of the listed items, optionally with one or more like items not listed. A "combination" is inclusive of blends, mixtures, alloys, reaction products, and the like.
[0135] Unless defined otherwise, technical and scientific terms used herein have the same meaning as is commonly understood by one of skill in the art to which this disclosure belongs. As used herein, an unattached line, e.g., "O--" indicates a bond, not a methyl group, which is indicated by "--CH.sub.3". The terms "hydrocarbyl" and "hydrocarbon" refer broadly to a group comprising carbon and hydrogen, optionally with 1 to 3 heteroatoms, for example, oxygen, nitrogen, halogen, silicon, sulfur, or a combination thereof; the term "aliphatic" means a branched or unbranched, saturated or unsaturated group containing carbon and hydrogen, optionally with 1 to 3 heteroatoms, for example, oxygen, nitrogen, halogen, silicon, sulfur, or a combination thereof; the term "cycloaliphatic" means a saturated or unsaturated group comprising carbon and hydrogen optionally with 1 to 3 heteroatoms, for example, oxygen, nitrogen, halogen, silicon, sulfur, or a combination thereof; "alkyl" means a straight or branched chain, saturated monovalent hydrocarbon group; "alkylene" means a straight or branched chain, saturated, divalent hydrocarbon group; "alkylidene" means a straight or branched chain, saturated divalent hydrocarbon group, with both valences on a single common carbon atom; "alkenyl" means a straight or branched chain monovalent hydrocarbon group having at least two carbons joined by a carbon-carbon double bond; "cycloalkyl" means a non-aromatic monovalent monocyclic or multicyclic hydrocarbon group having at least three carbon atoms, "cycloalkenyl" means a non-aromatic cyclic divalent hydrocarbon group having at least three carbon atoms, with at least one degree of unsaturation; "aryl" and "arylene" means a monovalent group and divalent group respectively containing at least one aromatic ring and optionally a nonaromatic ring, and having only carbon in the ring or rings; "alkylarylene" means an aryl group that has been substituted with an alkyl group as defined above, with 4-methylphenyl being an exemplary alkylarylene group; "arylalkylene" means an alkyl group that has been substituted with an aryl group as defined above, with benzyl being an exemplary arylalkylene group; "heteroaryl" and "heteroarylene" means a monovalent group and divalent group respectively wherein at least one carbon in a ring is replaced by a heteroatom (S, O, P, or N); "acyl" means an alkyl group as defined above with the indicated number of carbon atoms attached through a carbonyl carbon bridge (--C(.dbd.O)--); "alkoxy" means an alkyl group as defined above with the indicated number of carbon atoms attached through an oxygen bridge (--O--); and "aryloxy" means an aryl group as defined above with the indicated number of carbon atoms attached through an oxygen bridge (--O--).
[0136] Unless otherwise indicated, each of the foregoing groups can be unsubstituted or substituted, provided that the substitution does not significantly adversely affect synthesis, stability, or use of the compound. The term "substituted" as used herein means that at least one hydrogen on the designated atom or group is replaced with another group, provided that the designated atom's normal valence is not exceeded. When the substituent is oxo (i.e., .dbd.O), then two hydrogens on the atom are replaced. Combinations of substituents or variables are permissible provided that the substitutions do not significantly adversely affect synthesis or use of the compound. Exemplary groups that can be present on a "substituted" position include, but are not limited to, cyano; hydroxyl; nitro; alkanoyl (such as a C.sub.2-6 alkanoyl group such as acyl); carboxamido; C.sub.1-6 or C.sub.1-3 alkyl, cycloalkyl, alkenyl, and alkynyl (including groups having at least one unsaturated linkages and from 2 to 8, or 2 to 6 carbon atoms); C.sub.1-6 or C.sub.1-3 alkoxys; C.sub.6-10 aryloxy such as phenoxy; C.sub.1-6 alkylthio; C.sub.1-6 or C.sub.1-3 alkylsulfinyl; C.sub.1-6 or C.sub.1-3 alkylsulfonyl; amino di(C.sub.1-6 or C.sub.1-3)alkyl; C.sub.6-12 aryl having at least one aromatic rings (e.g., phenyl, biphenyl, naphthyl, or the like); C.sub.7-19 arylalkylene having 1 to 3 separate or fused rings and from 6 to 18 ring carbon atoms; or C.sub.7-19 arylalkyleneoxy having 1 to 3 separate or fused rings and from 6 to 18 ring carbon atoms, with benzyloxy being an exemplary arylalkyleneoxy. When a group is substituted, the indicated number of carbon atoms includes the substituent.
[0137] All cited patents, patent applications, and other references are incorporated herein by reference in their entirety. However, if a term in the present application contradicts or conflicts with a term in the incorporated reference, the term from the present application takes precedence over the conflicting term from the incorporated reference.
[0138] While particular embodiments have been described, alternatives, modifications, variations, improvements, and substantial equivalents that are or may be presently unforeseen may arise to applicants or others skilled in the art. Accordingly, the appended claims as filed and as they may be amended are intended to embrace all such alternatives, modifications variations, improvements, and substantial equivalents.
* * * * *


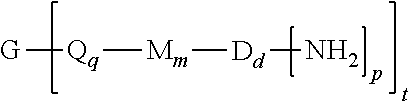

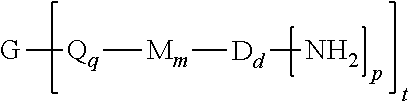







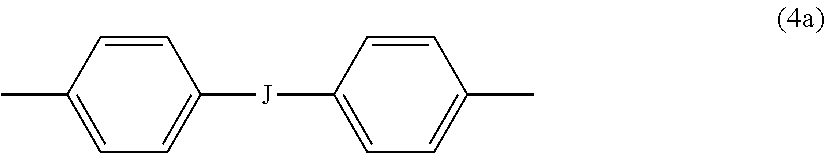
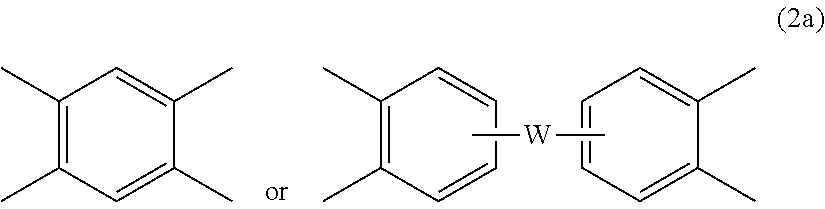
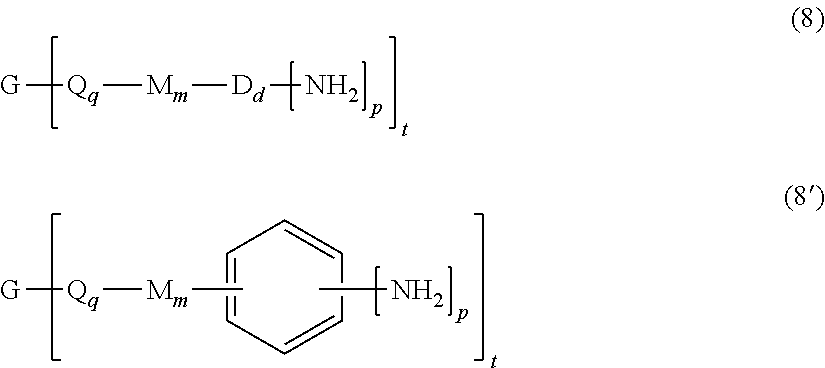
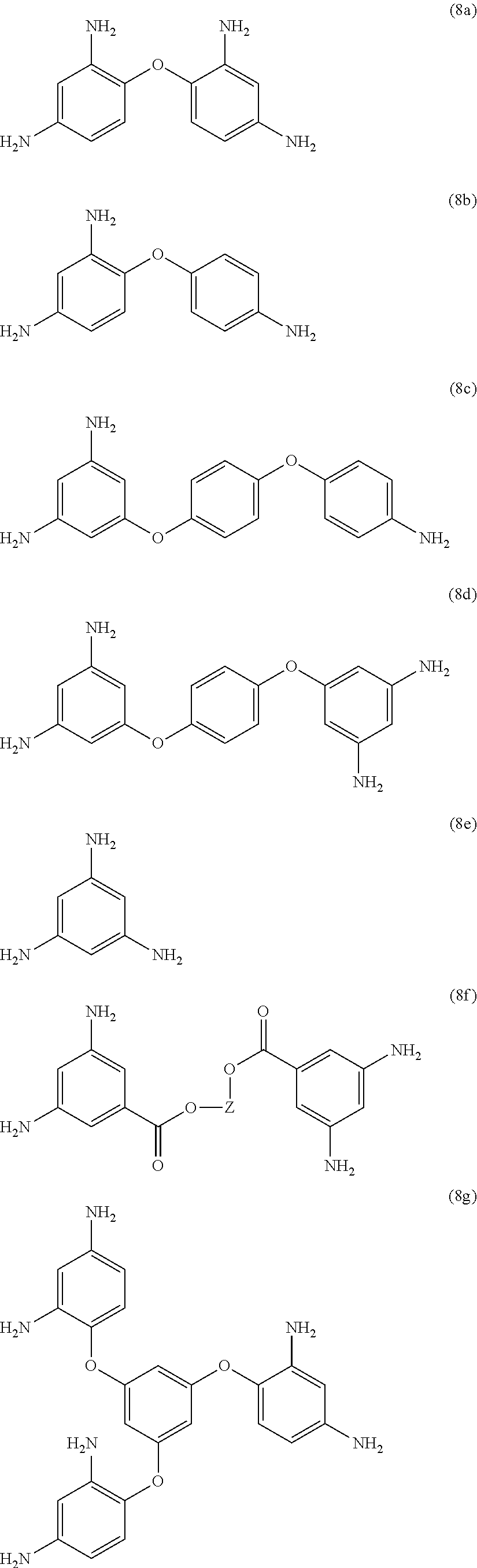
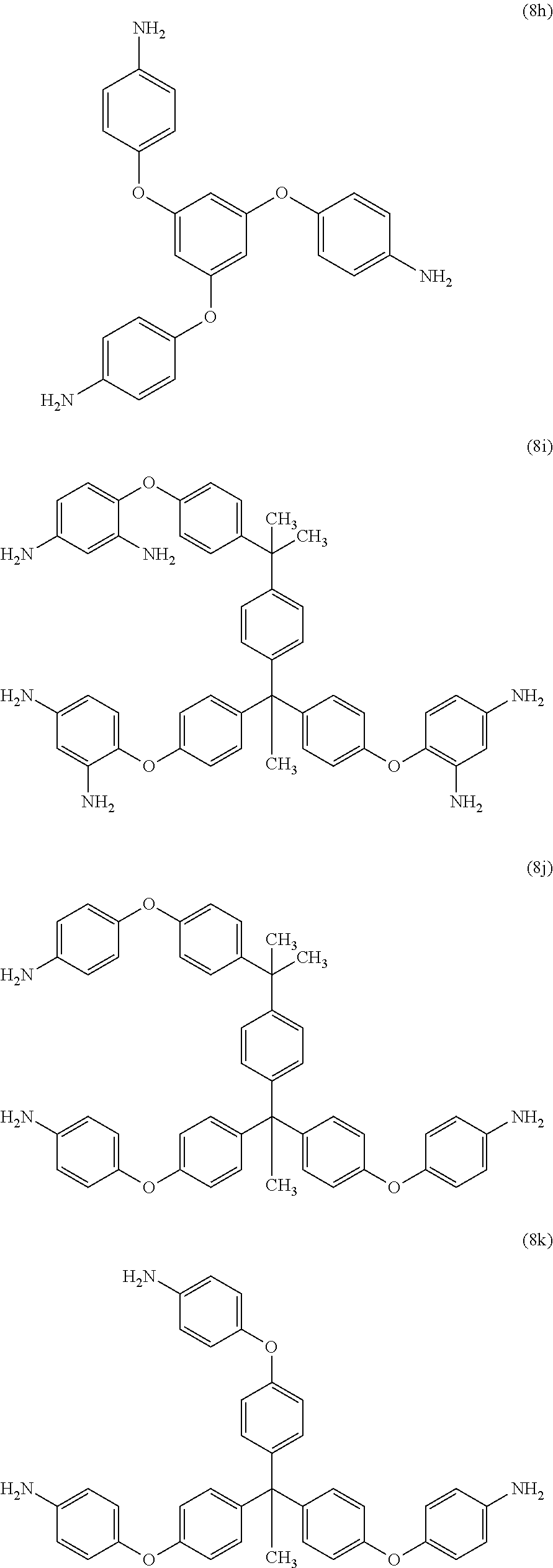
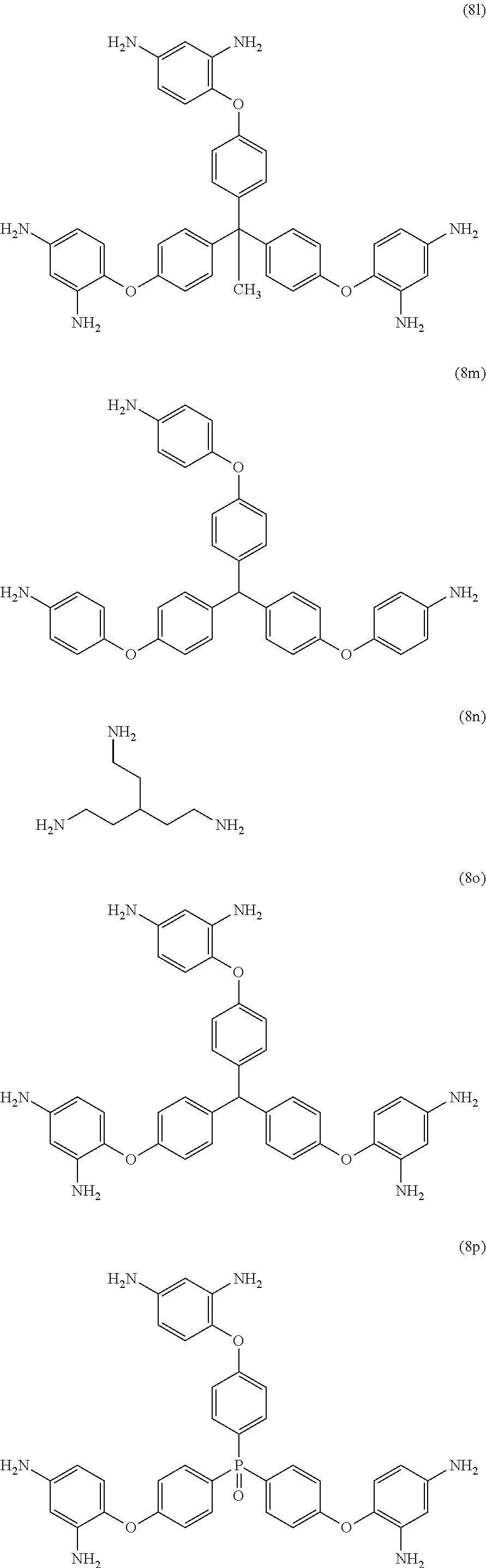
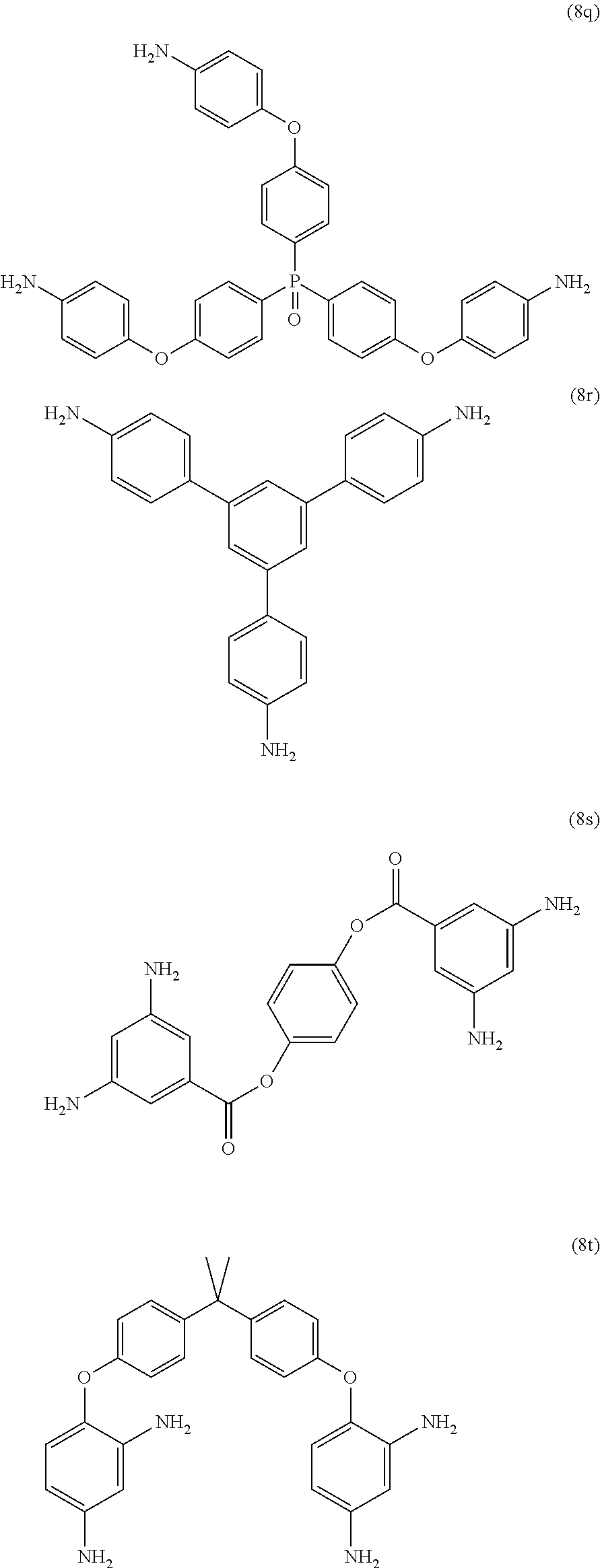
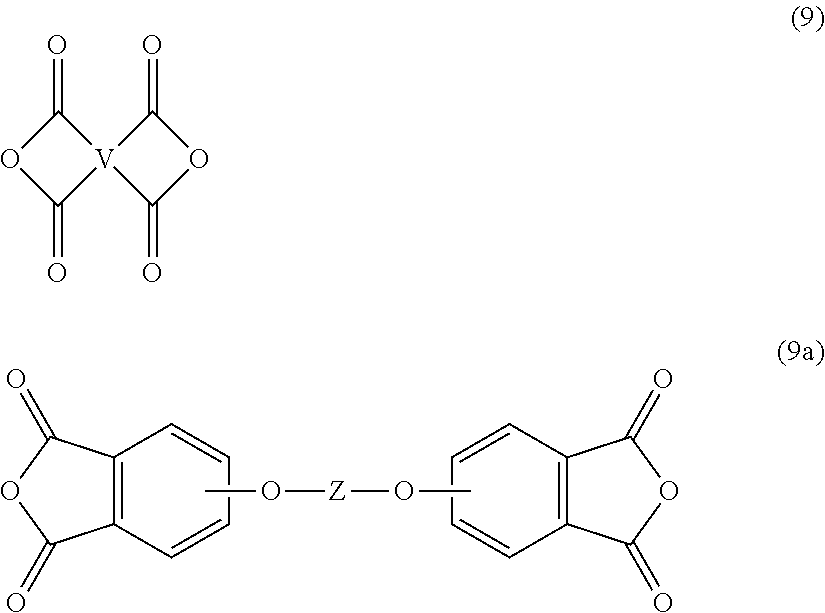
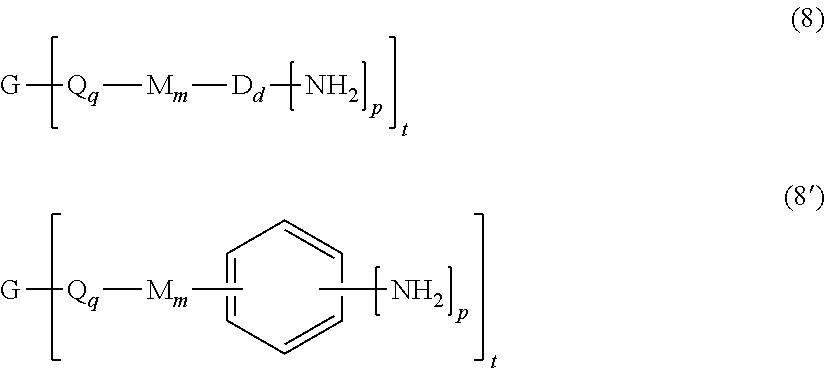
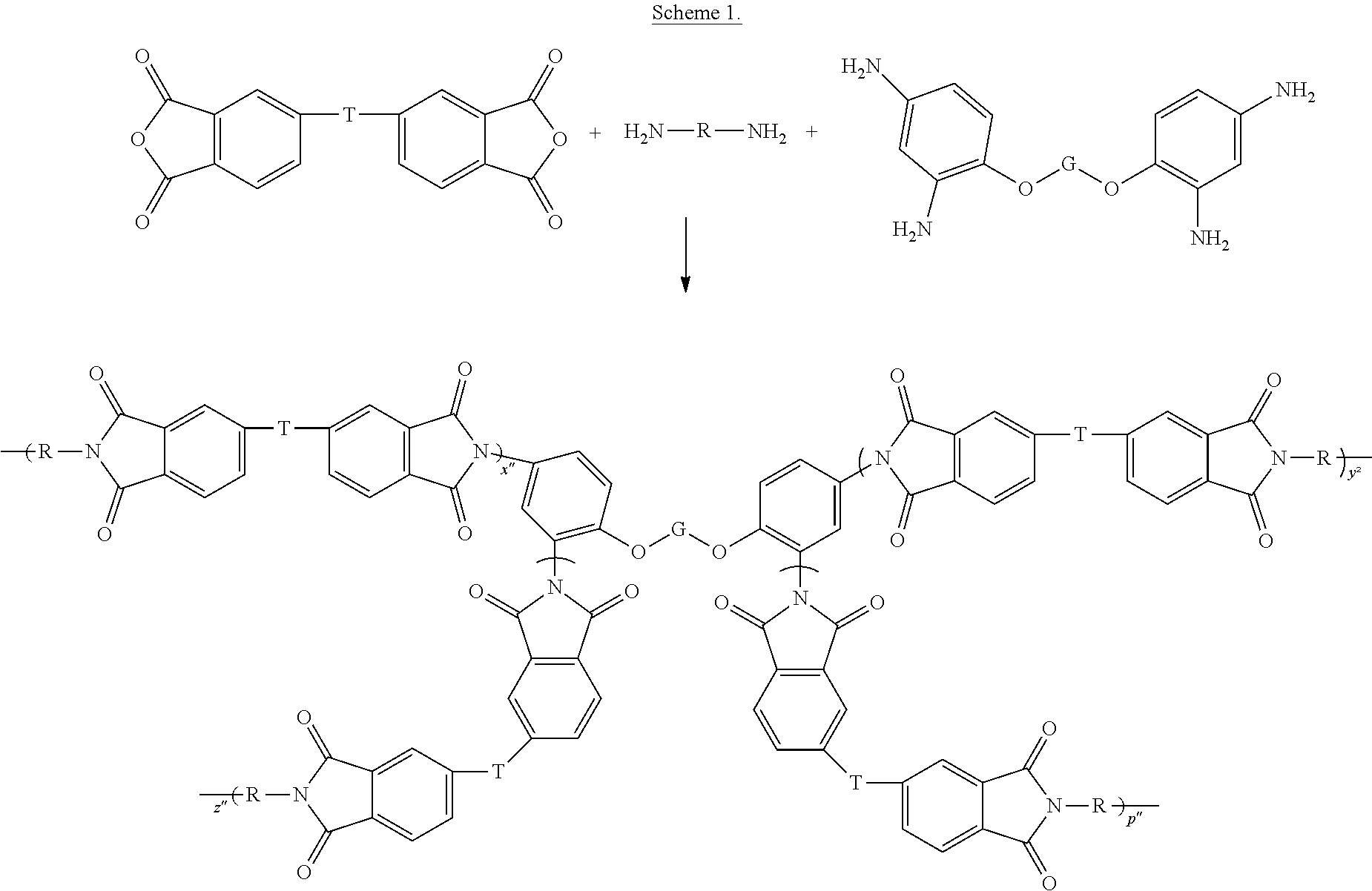
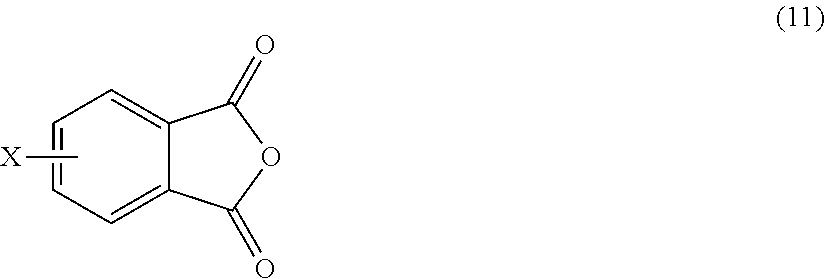
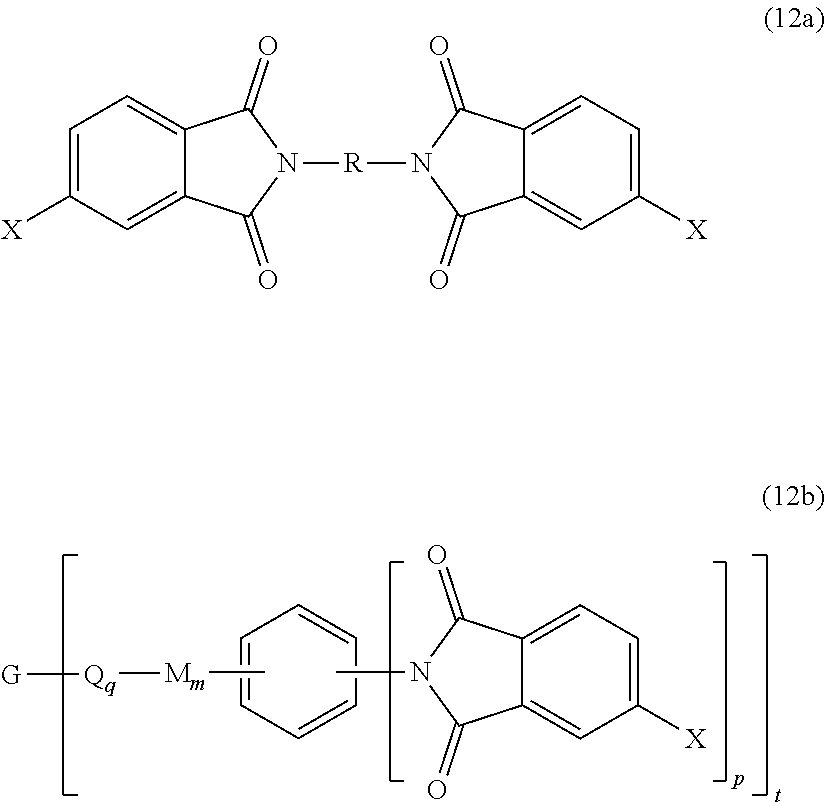
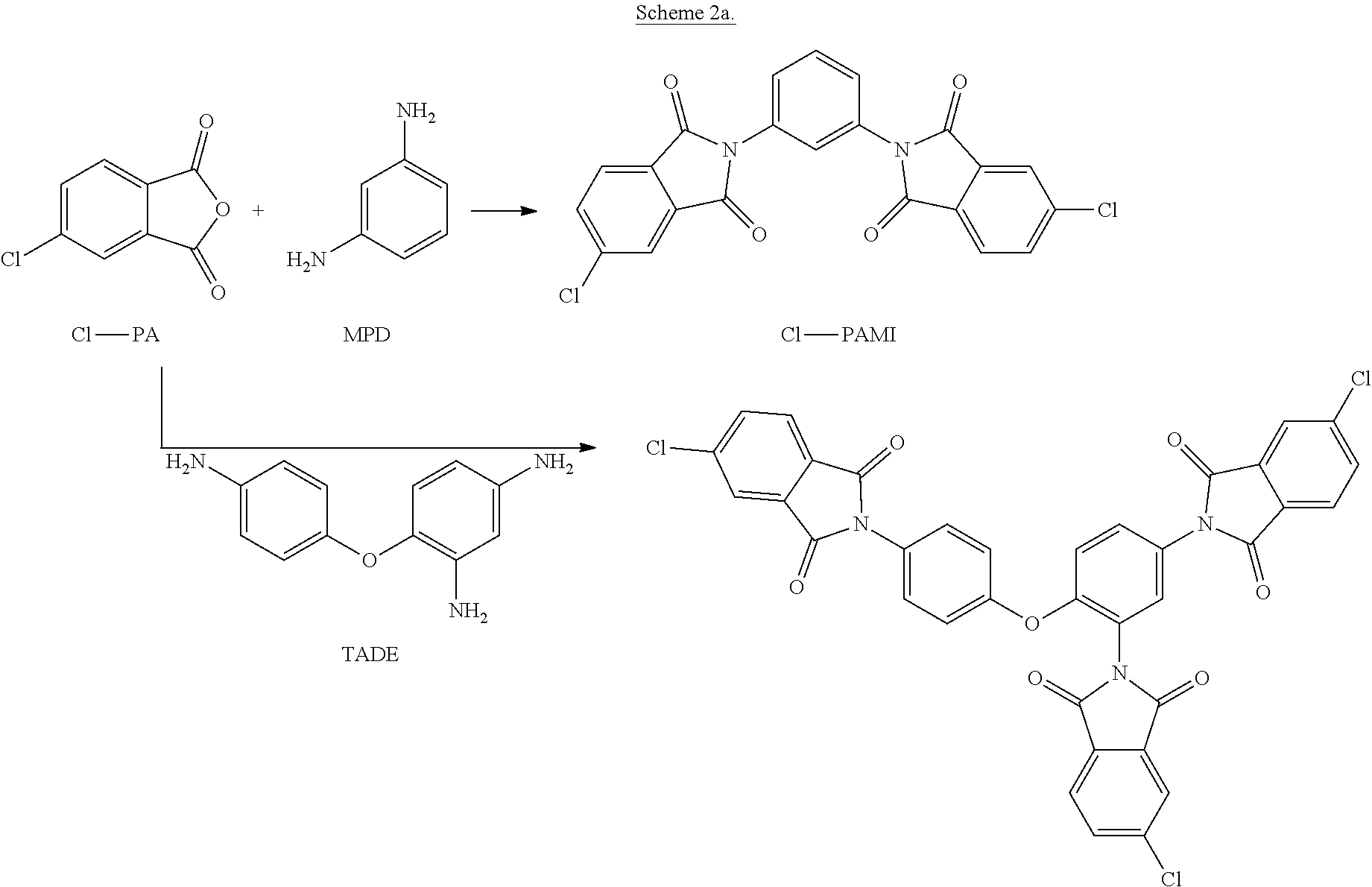
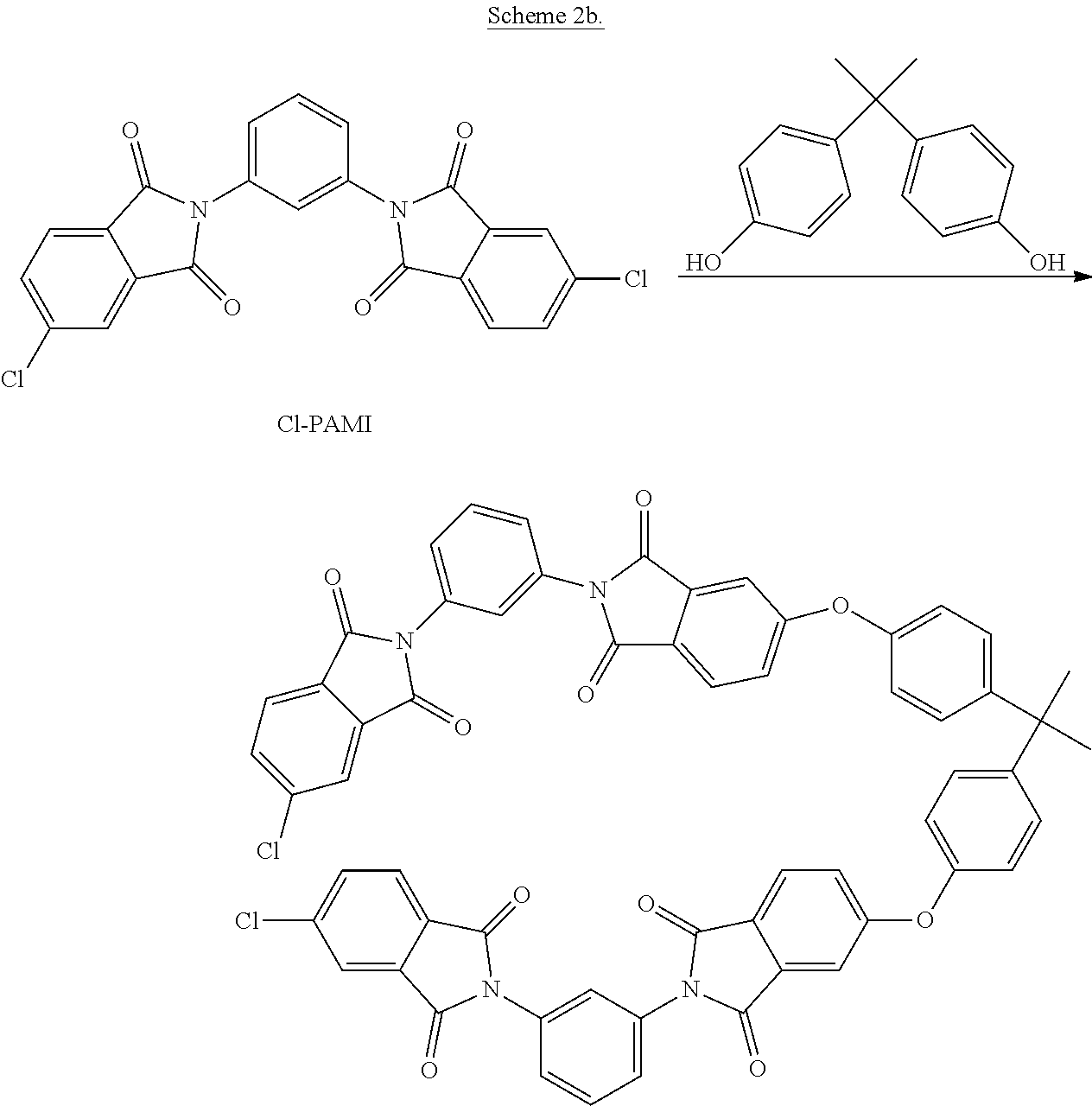
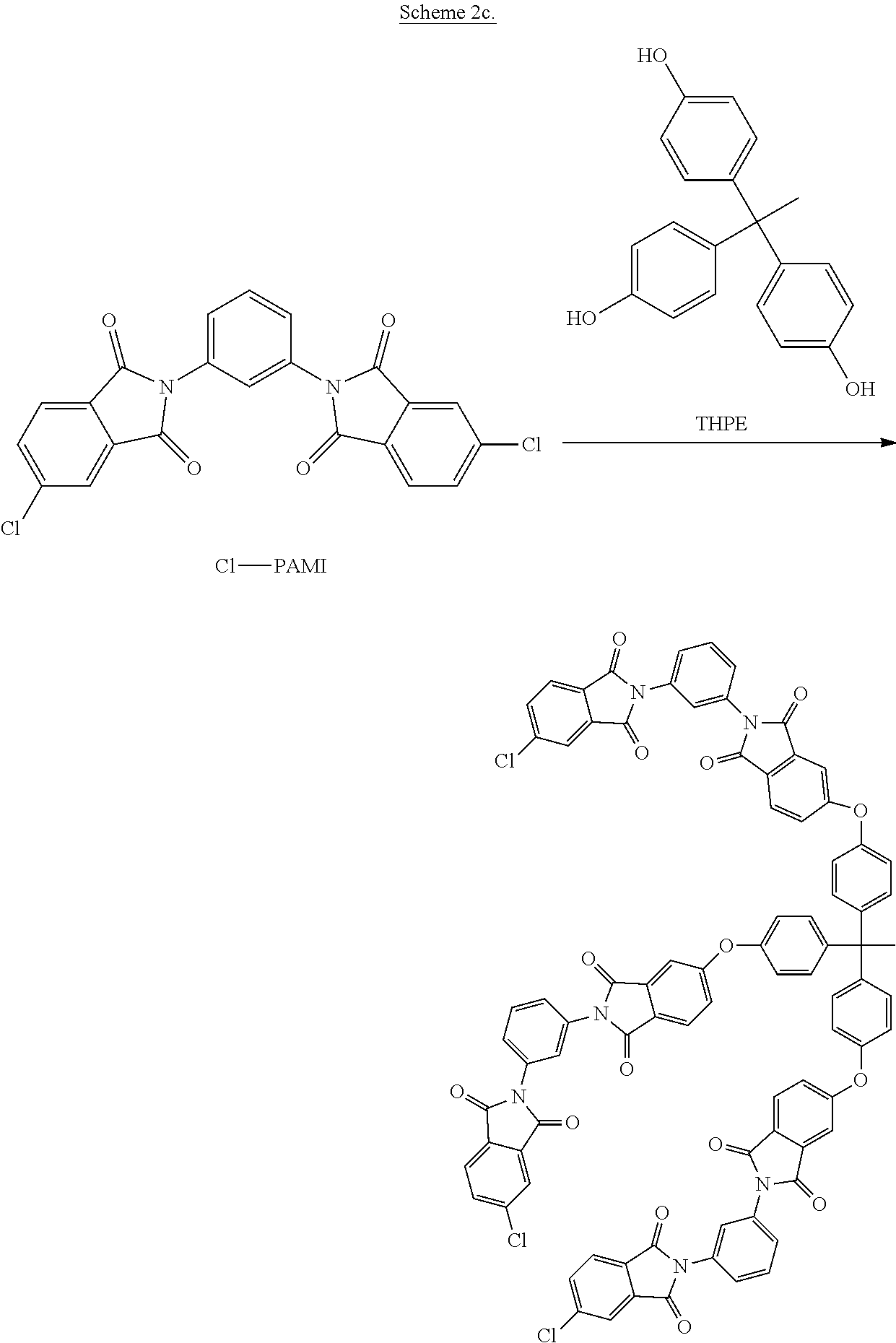
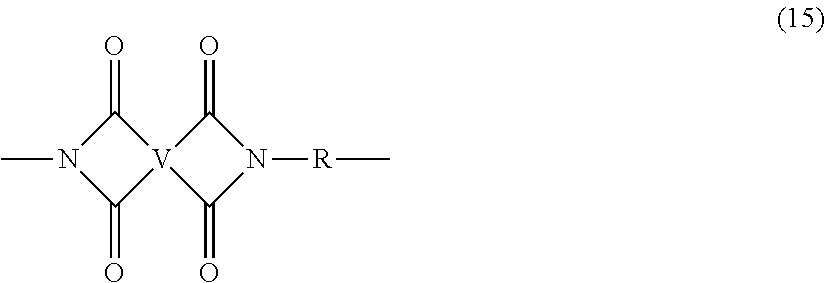
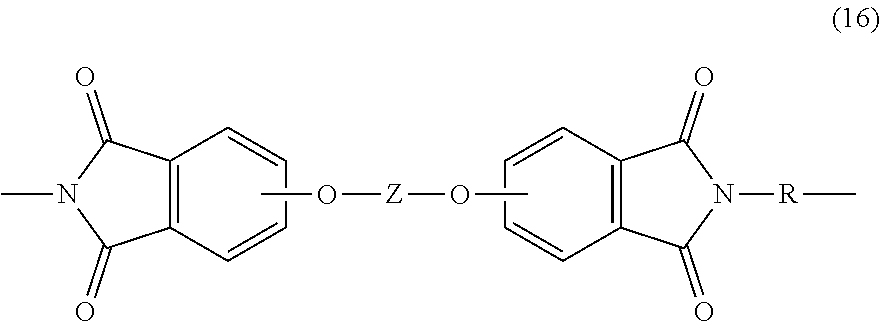
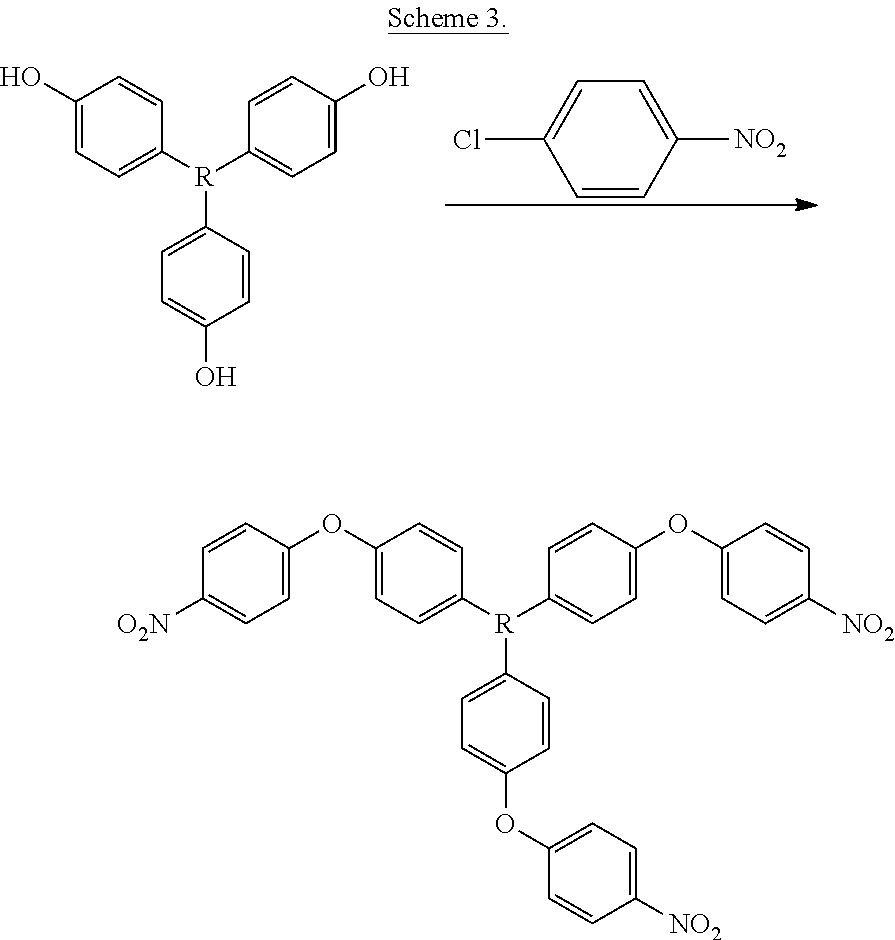
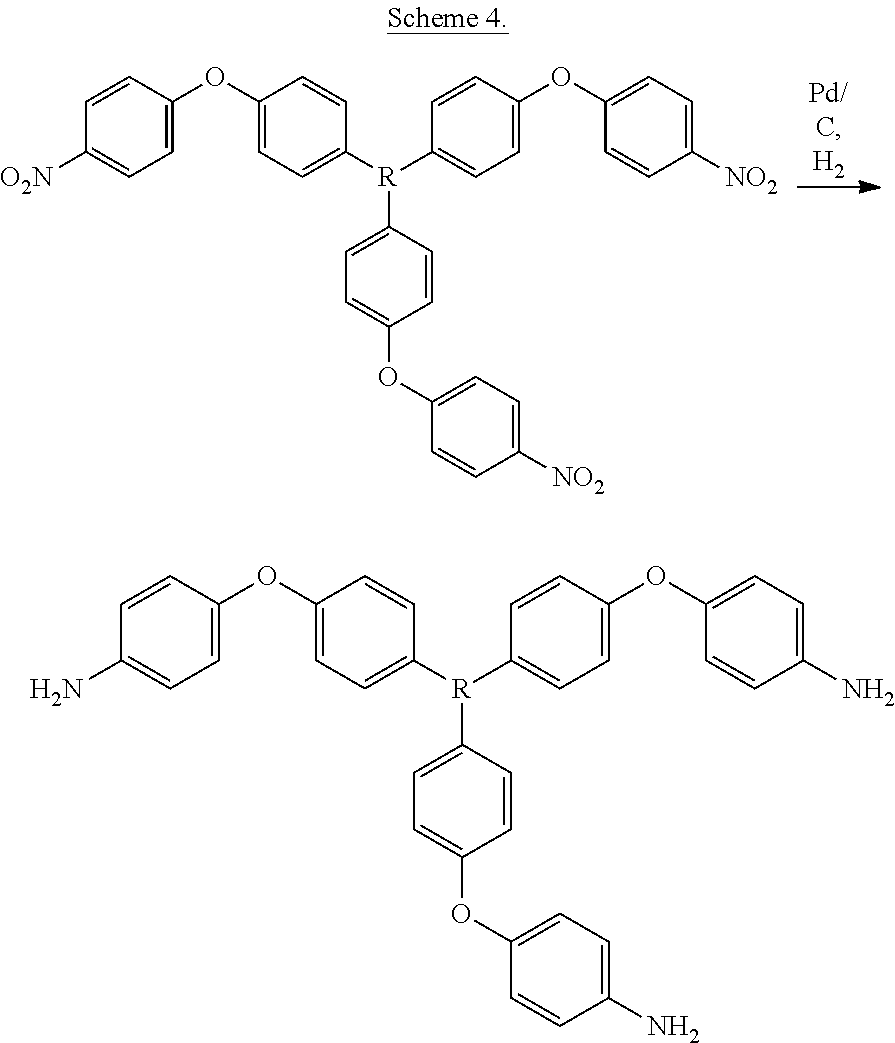
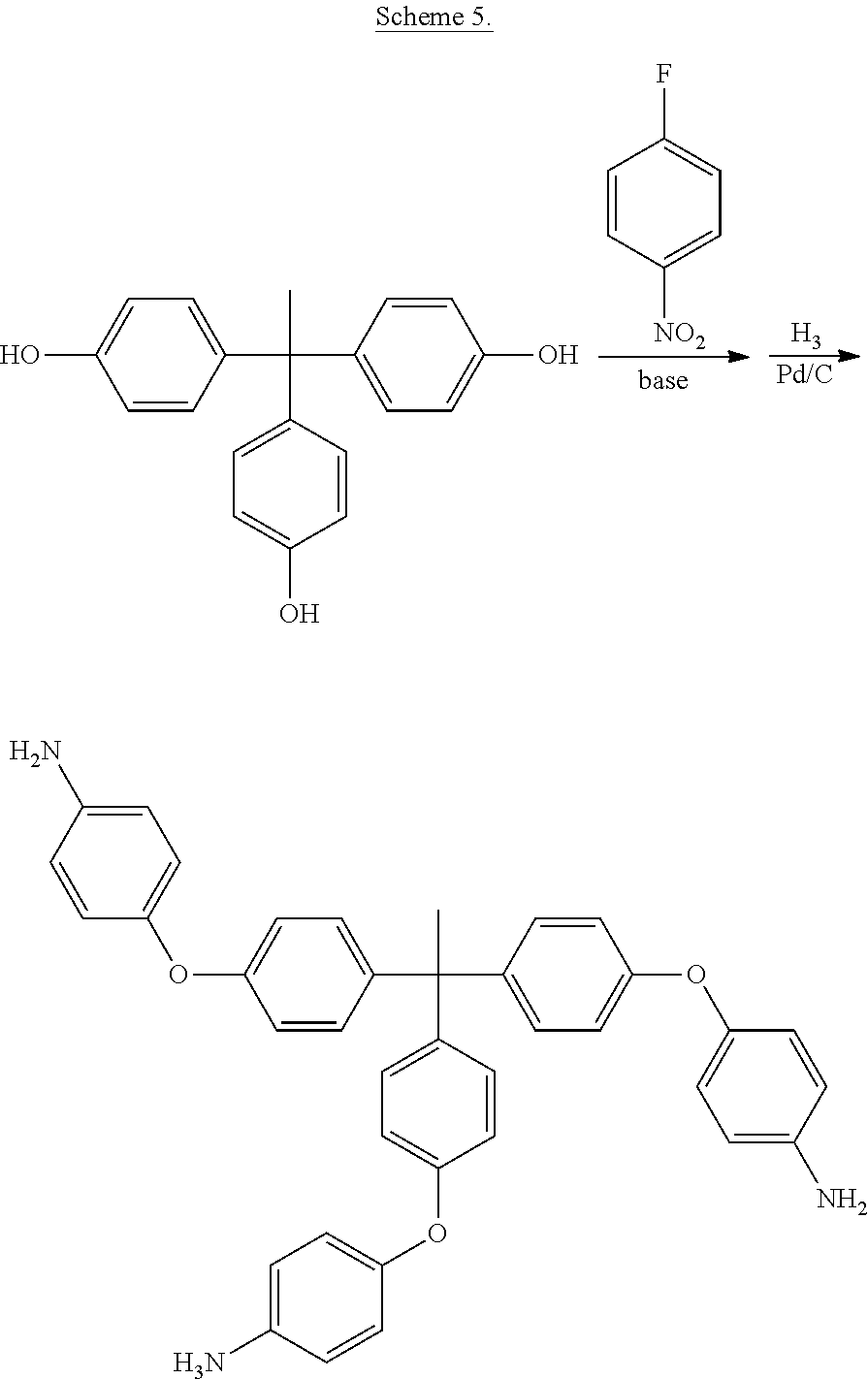
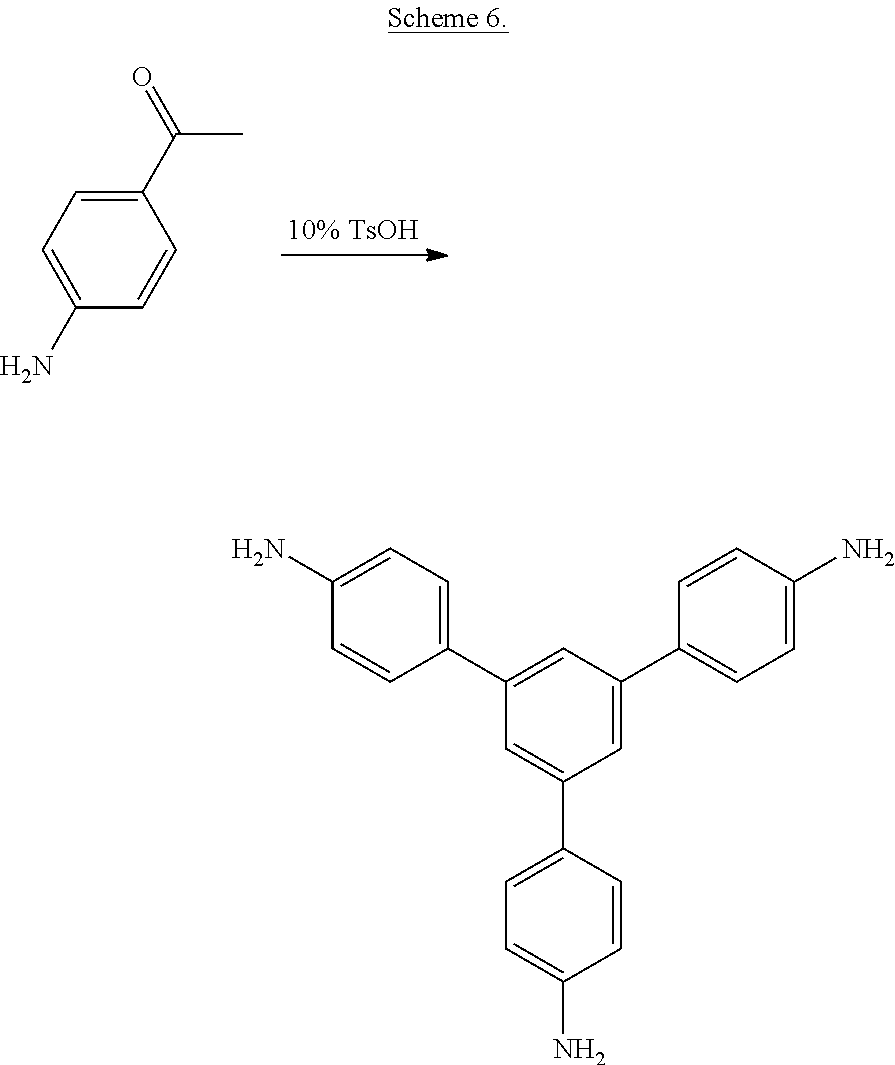
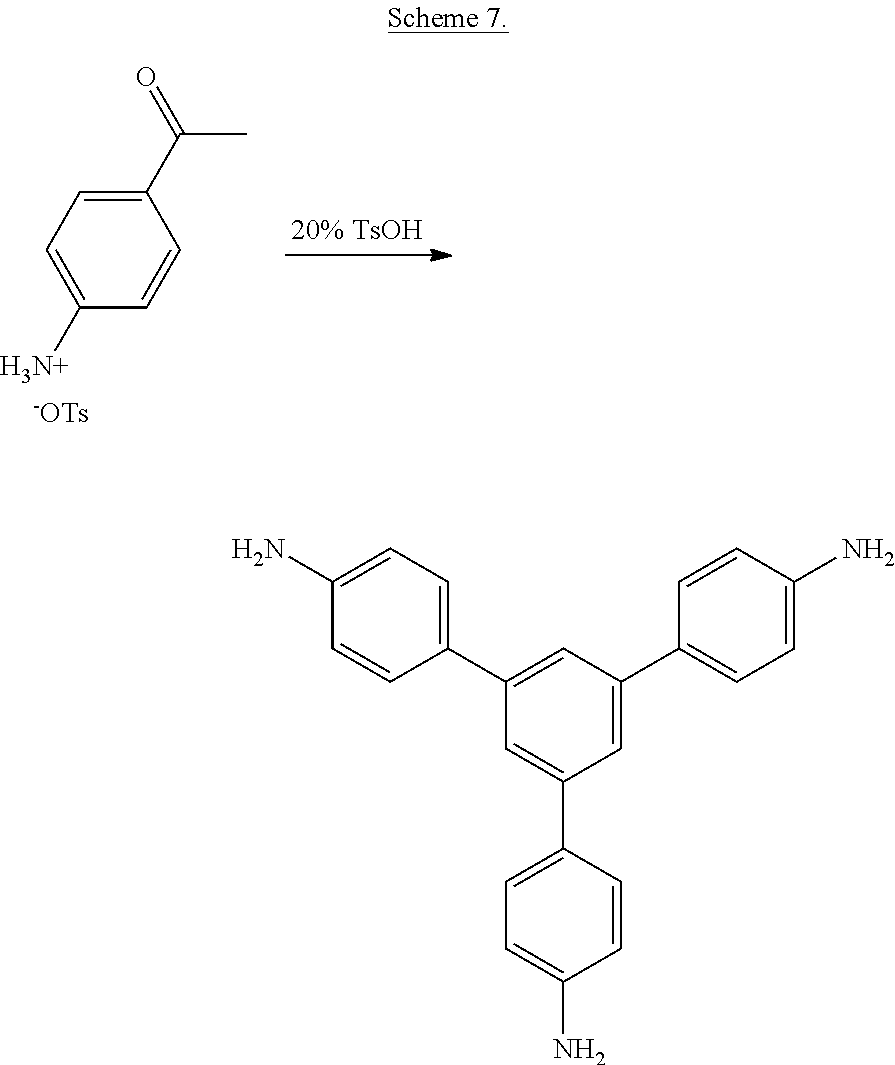

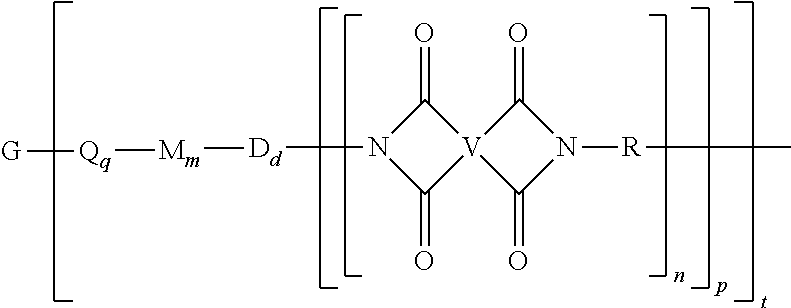
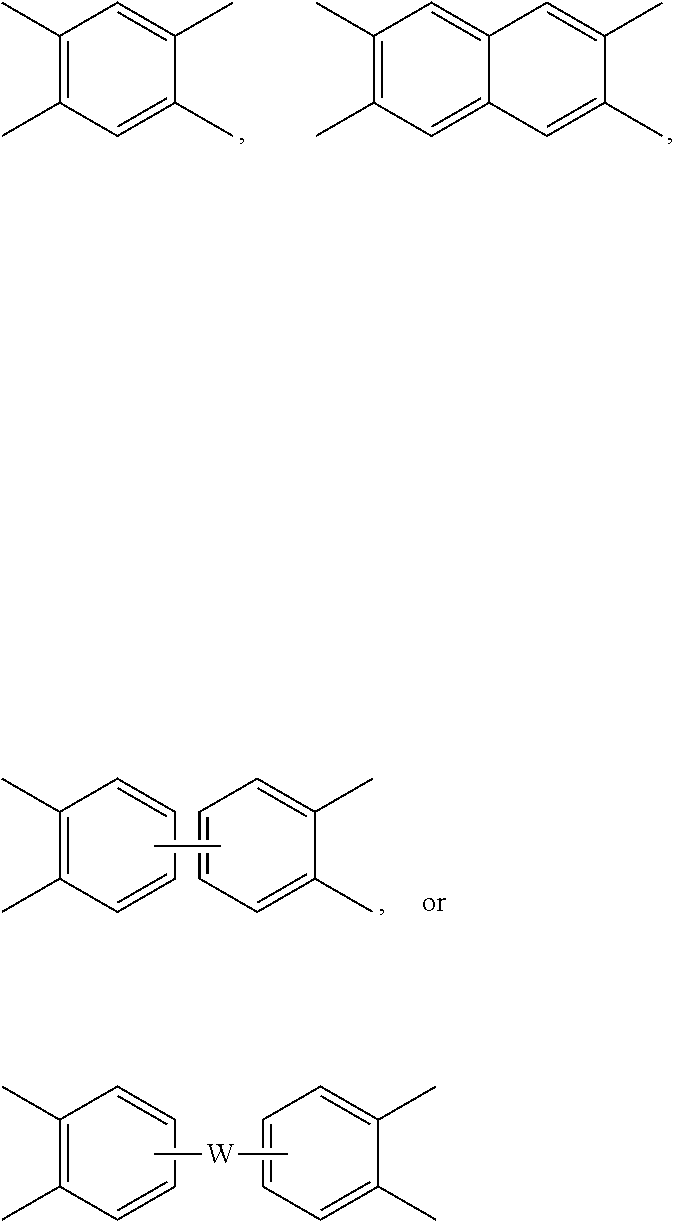
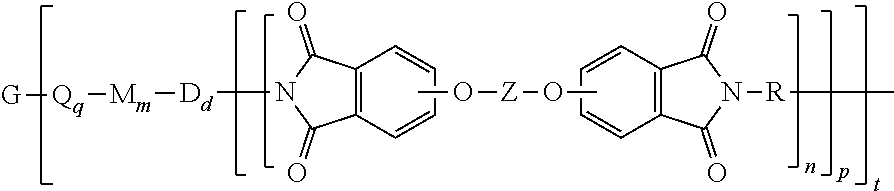
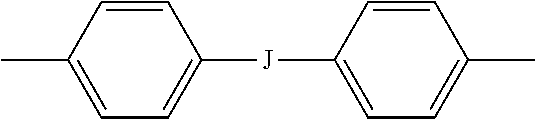
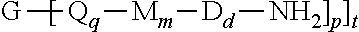
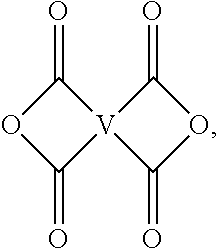
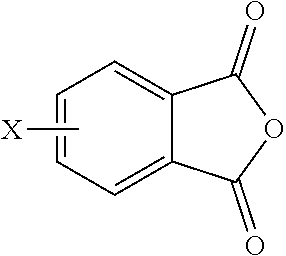
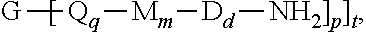

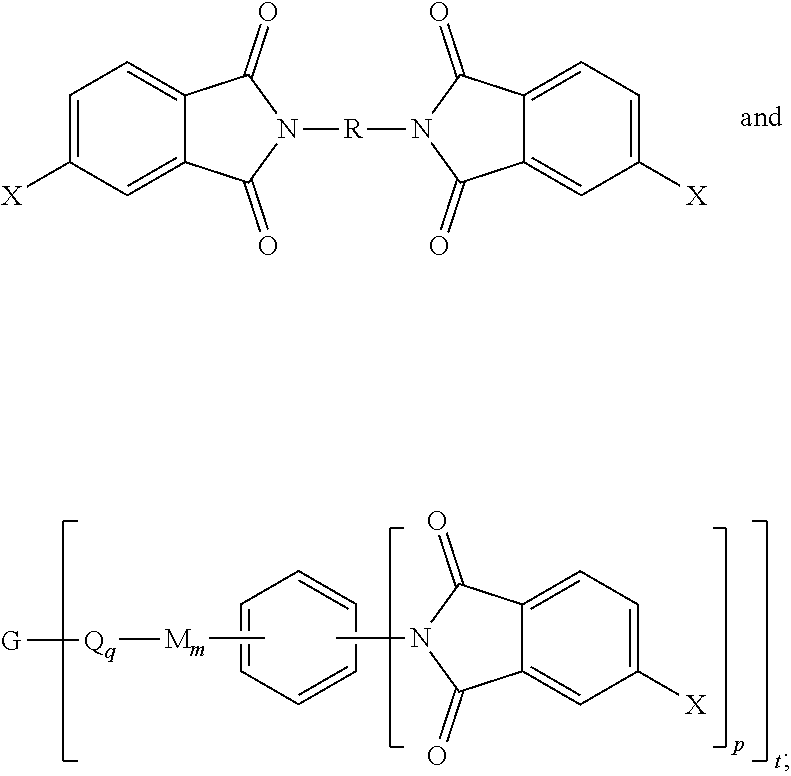
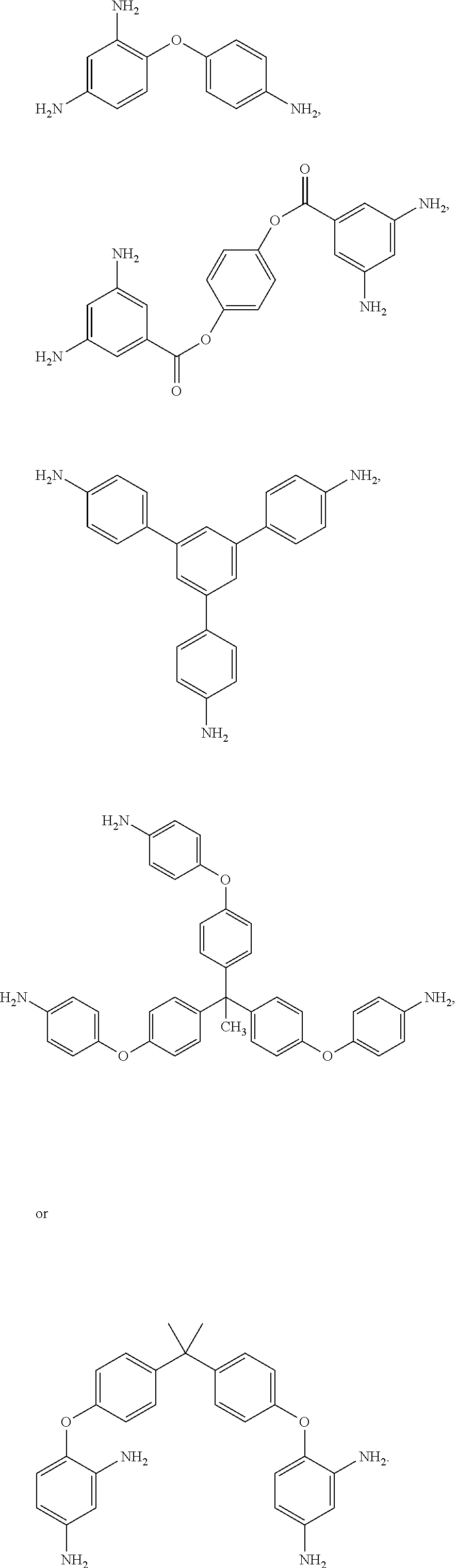
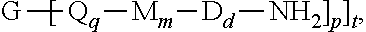
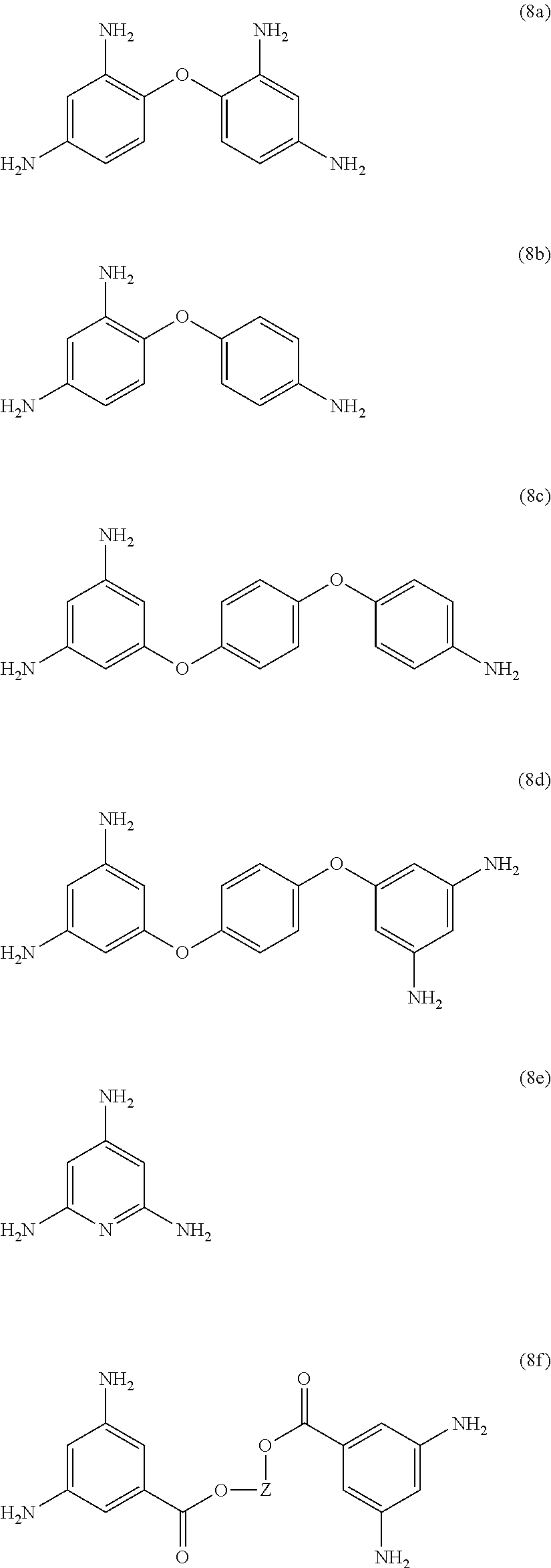
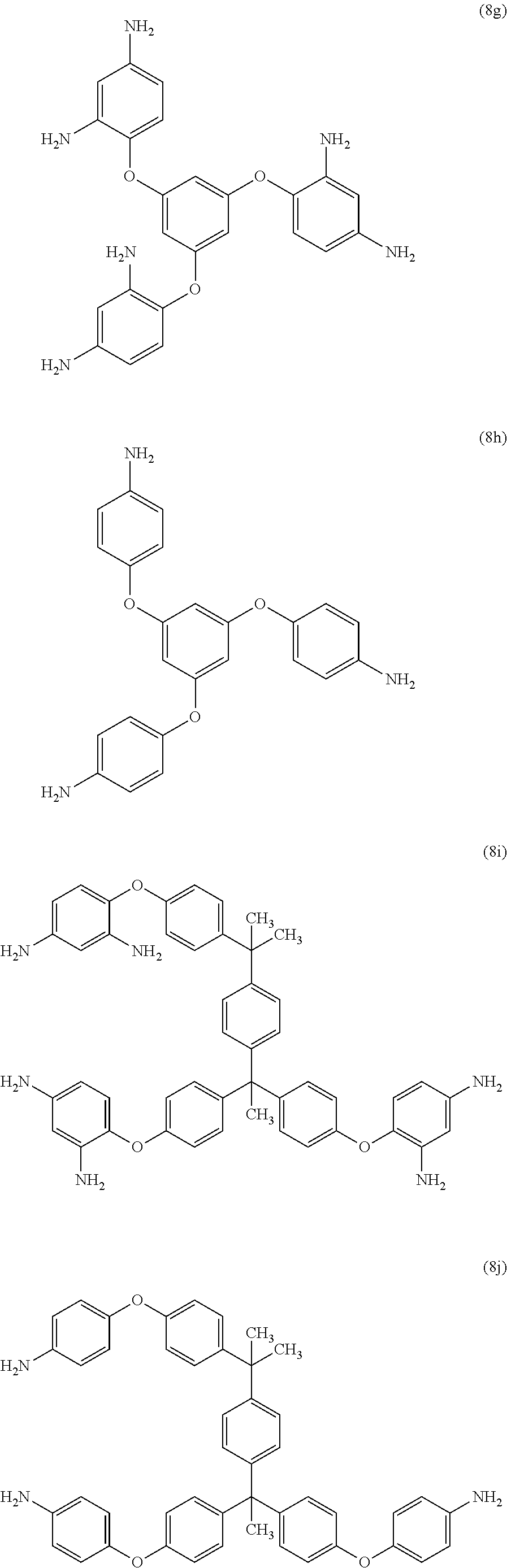
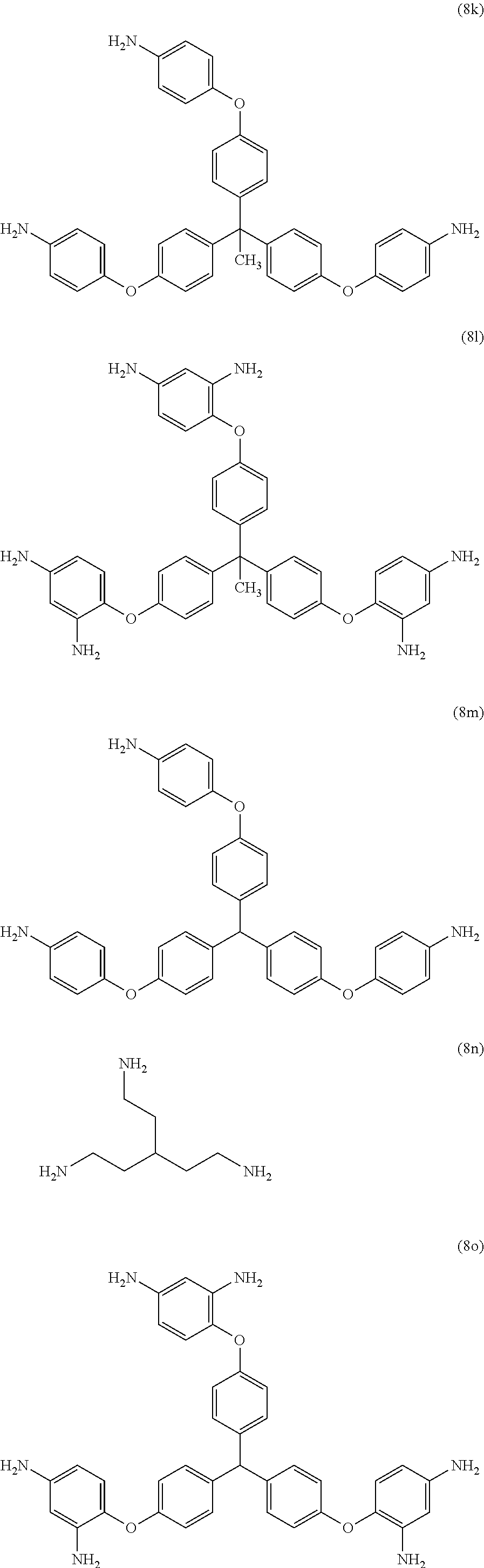
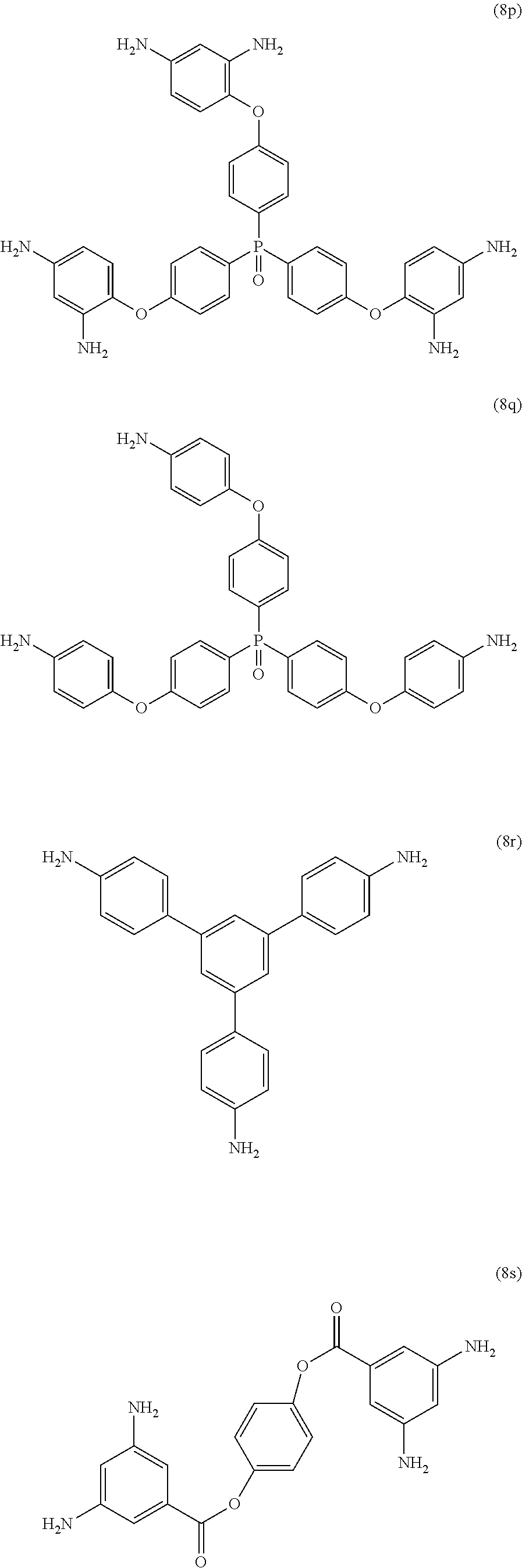
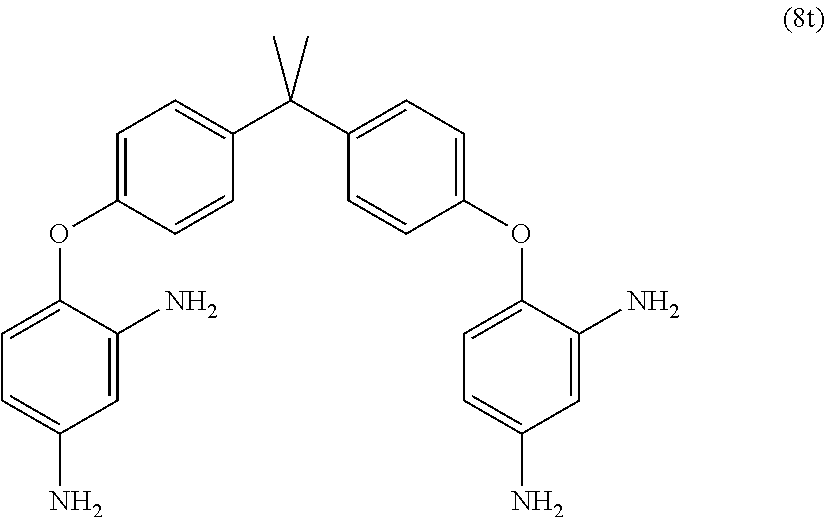
D00000
D00001
D00002
D00003
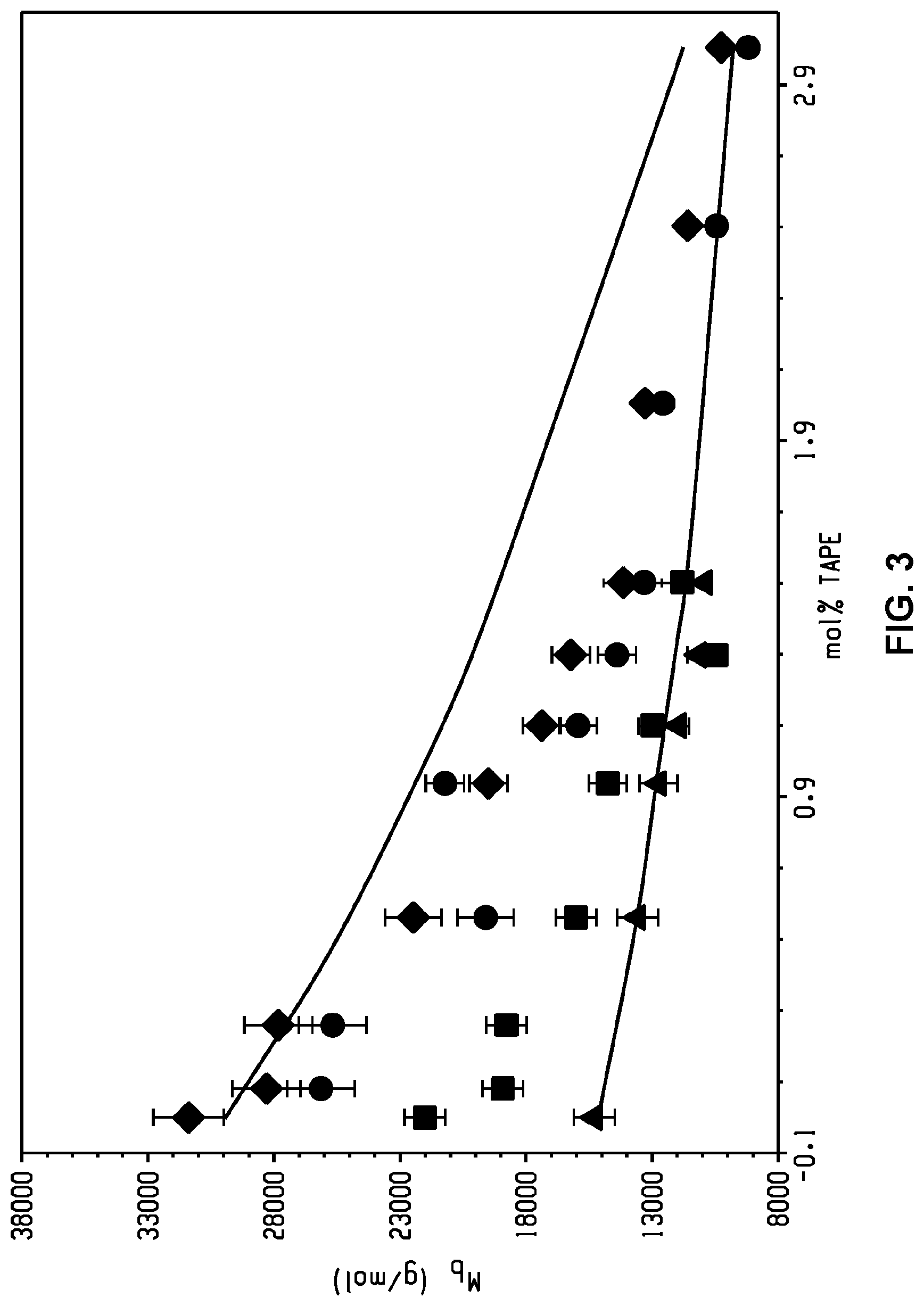
D00004
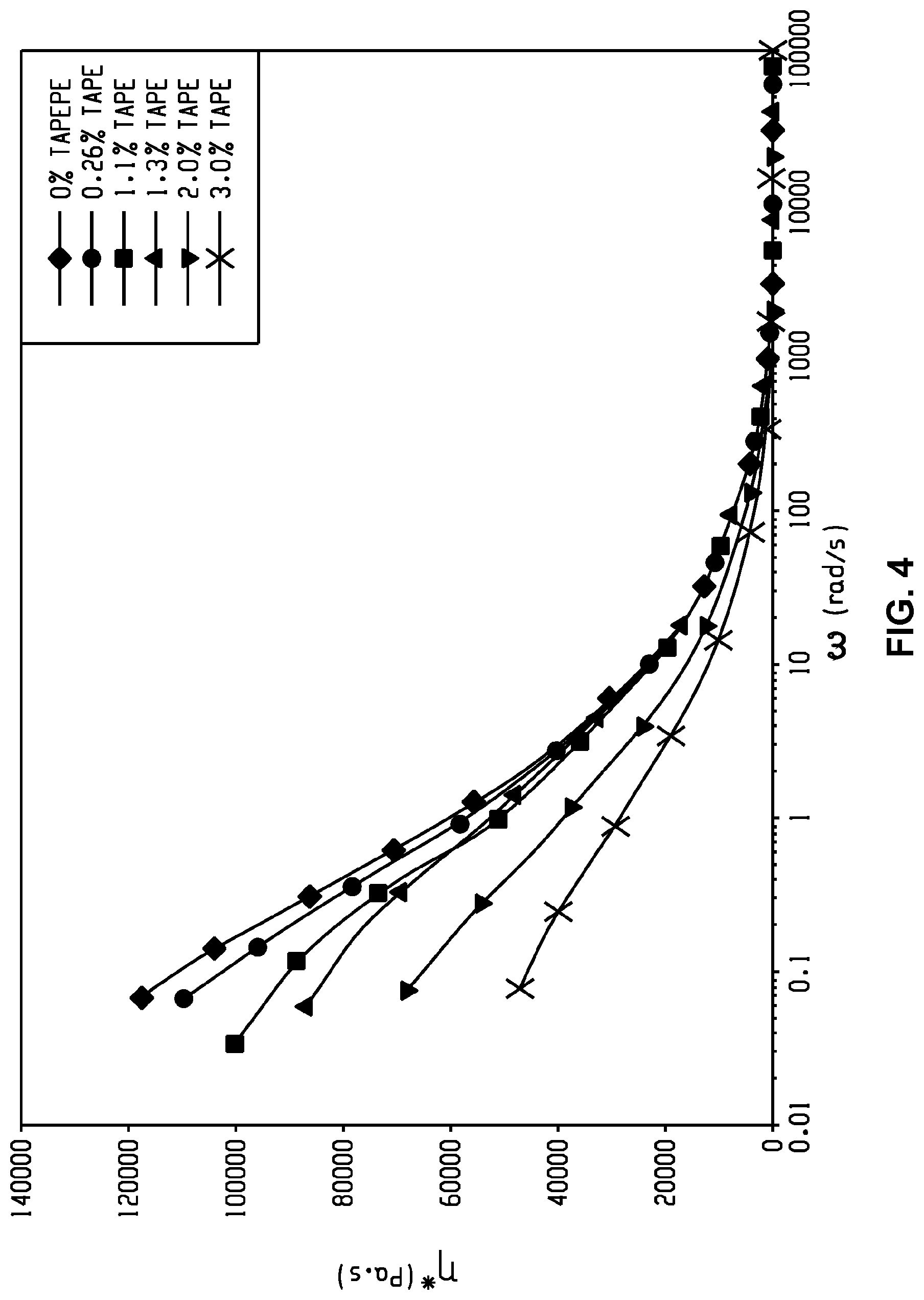
D00005
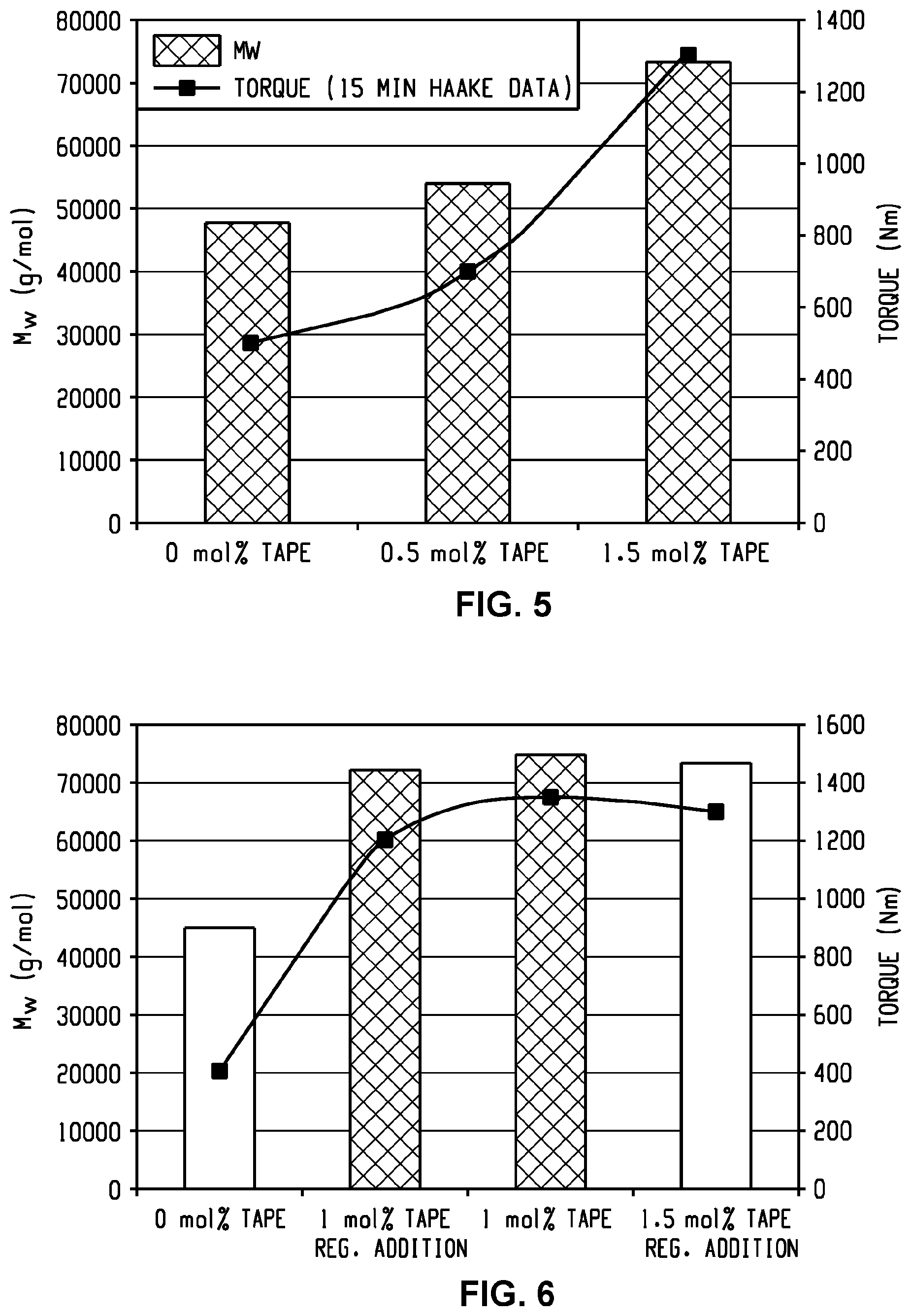
D00006
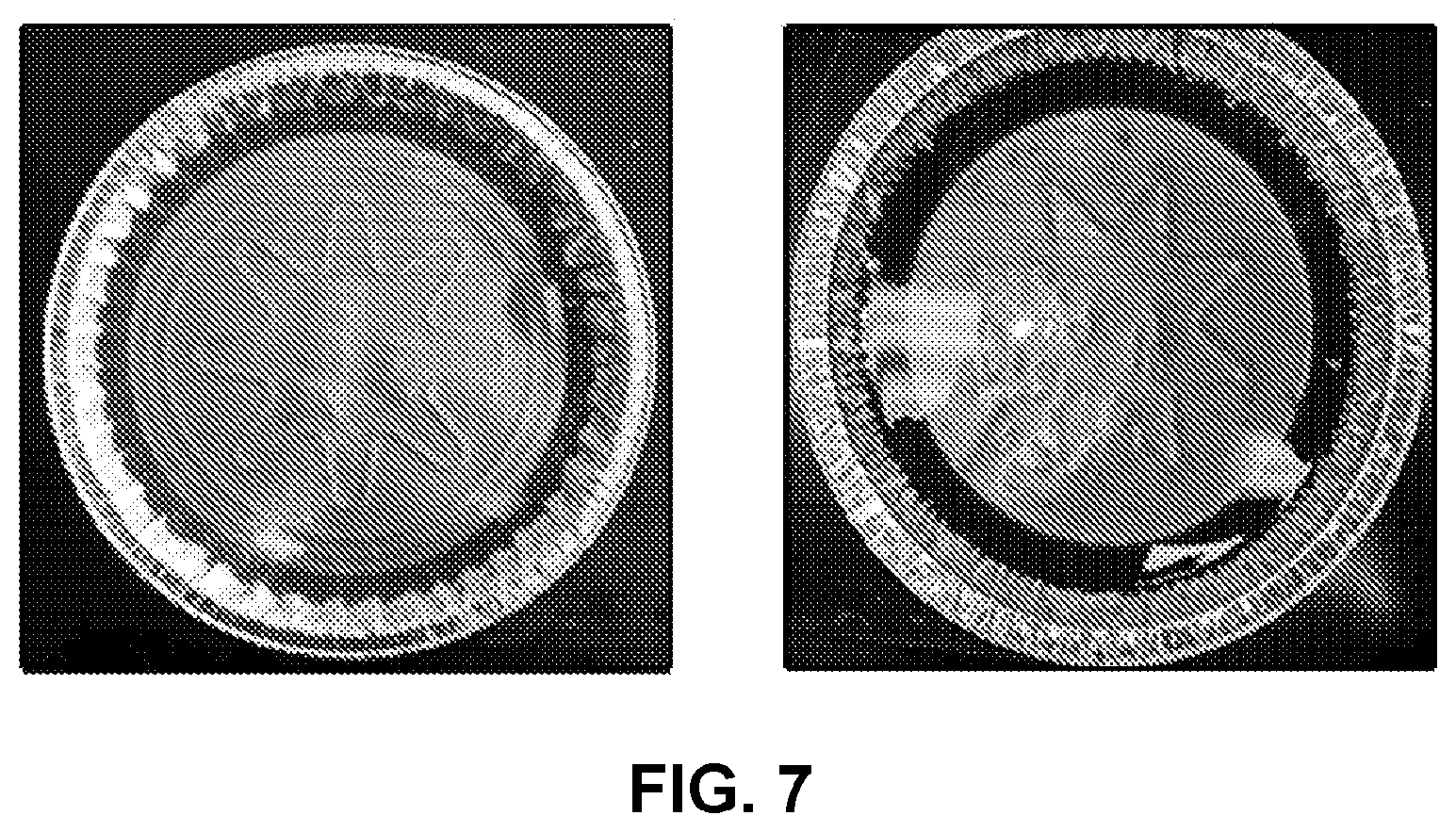
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.