Digital Microfluidic Delivery Device
PAOLINI, JR.; Richard J. ; et al.
U.S. patent application number 16/601641 was filed with the patent office on 2020-04-16 for digital microfluidic delivery device. The applicant listed for this patent is E INK CORPORATION. Invention is credited to Brian D. BEAN, Timothy J. O'MALLEY, Richard J. PAOLINI, JR., Stephen J. TELFER.
| Application Number | 20200114135 16/601641 |
| Document ID | / |
| Family ID | 70159689 |
| Filed Date | 2020-04-16 |




| United States Patent Application | 20200114135 |
| Kind Code | A1 |
| PAOLINI, JR.; Richard J. ; et al. | April 16, 2020 |
DIGITAL MICROFLUIDIC DELIVERY DEVICE
Abstract
An active molecule delivery system whereby active molecules can be released on demand and/or a variety of different active molecules can be delivered from the same system and/or different concentrations of active molecules can be delivered from the same system. The invention is well-suited for delivering pharmaceuticals to patients transdermally. In some embodiments, the system includes two separate reservoirs and a mixing area thereby allowing precursors to be mixed immediately before transdermal delivery.
| Inventors: | PAOLINI, JR.; Richard J.; (Framingham, MA) ; TELFER; Stephen J.; (Arlington, MA) ; O'MALLEY; Timothy J.; (Westford, MA) ; BEAN; Brian D.; (Newton, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 70159689 | ||||||||||
| Appl. No.: | 16/601641 | ||||||||||
| Filed: | October 15, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62745718 | Oct 15, 2018 | |||
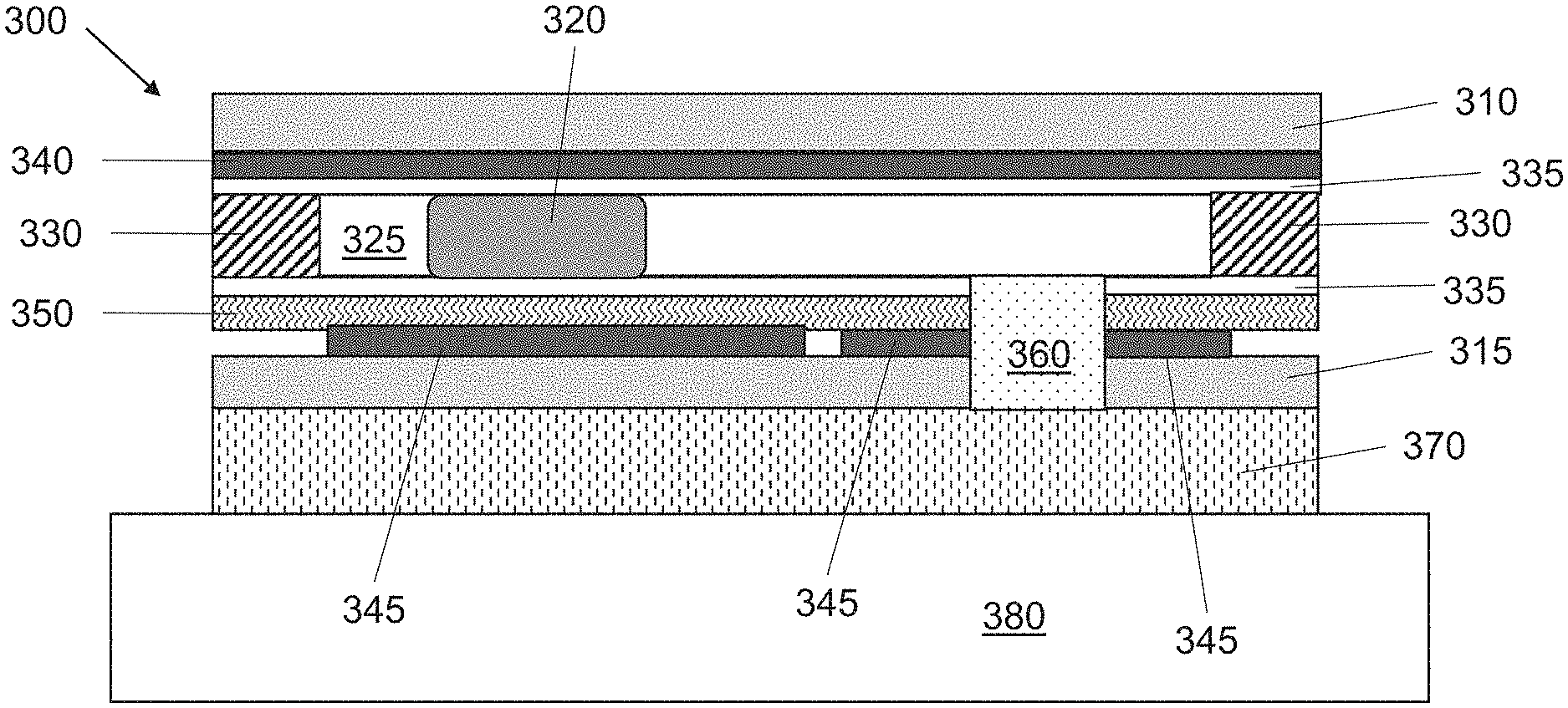
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61M 35/10 20190501; A61M 2205/0238 20130101; A61N 1/30 20130101; A61M 37/00 20130101; A61M 2205/82 20130101; A61M 2037/0007 20130101 |
| International Class: | A61M 35/00 20060101 A61M035/00 |
Claims
1. An active molecule delivery system comprising: a first substrate comprising: a plurality of driving electrodes, a dielectric layer covering the plurality of electrodes, and a first hydrophobic layer covering the dielectric layer; a second substrate comprising: a common electrode, a second hydrophobic layer covering the common electrode; a spacer separating the first and second substrates and creating a microfluidic region between the first and second substrates; a porous diffusion layer coupled to the first substrate on a side of the substrate opposed to the first hydrophobic layer, wherein the first substrate comprises a passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer; and a controller operatively coupled to the driving electrodes and configured to provide a voltage gradient between at least two driving electrodes.
2. The active molecule delivery system of claim 1, wherein each driving electrode is coupled to a thin-film-transistor.
3. The active molecule delivery system of claim 1, further comprising a plurality of passages providing fluidic communication between the hydrophobic layer and the porous diffusion layer.
4. The active molecule delivery system of claim 1, wherein the driving electrodes are flexible.
5. The active molecule delivery system of claim 1, wherein the passage includes capillary tubes or wicking fibers.
6. The active molecule delivery system of claim 5, wherein the capillary tubes or wicking fibers are coated with a hydrophobic coating.
7. The active molecule delivery system of claim 1, further comprising a reservoir in fluidic communication with the plurality of driving electrodes.
8. The active molecule delivery system of claim 7, further comprising a plurality of reservoirs in fluidic communication with the plurality of driving electrodes, and a plurality of passages providing fluidic communication between the hydrophobic layer and the porous diffusion layer, wherein each reservoir is in fluidic communication with only one passage.
9. The active molecule delivery system of claim 7, further comprising a plurality of reservoirs and a mixing region in fluidic communication with each of the plurality of reservoirs and the passages providing fluidic communication between the hydrophobic layer and the porous diffusion layer.
10. The active molecule delivery system of claim 1, wherein the porous diffusion layer comprises acrylate, methacrylate, polycarbonate, polyvinyl alcohol, cellulose, poly(N-isopropylacrylamide) (PNIPAAm), polylactic-co-glycolic acid) (PLGA), polyvinylidene chloride, acrylonitrile, amorphous nylon, oriented polyester, terephthalate, polyvinyl chloride, polyethylene, polybutylene, polypropylene, polyisobutylene, or polystyrene.
11. The active molecule delivery system of claim 1, further comprising a biocompatible adhesive in contact with the porous diffusion layer.
12. A method for delivering an active molecule to the skin of a subject, comprising: providing an active molecule delivery system including: a first substrate comprising a plurality of driving electrodes, a dielectric layer covering the plurality of electrodes, and a first hydrophobic layer covering the dielectric layer, a second substrate comprising a common electrode and a second hydrophobic layer covering the common electrode, a spacer separating the first and second substrates and creating a microfluidic region between the first and second substrates, a porous diffusion layer coupled to the first substrate on a side of the substrate opposed to the first hydrophobic layer, wherein the first substrate comprises a first passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer, and a controller operatively coupled to the driving electrodes and configured to provide a voltage gradient between at least two driving electrodes; coupling the porous diffusion layer to skin of a subject; moving a solution comprising an active molecule from a driving electrode to the first passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer; and allowing the active molecule to pass from the porous diffusion layer to the skin of the subject.
13. The method of claim 12, wherein the active molecule delivery system further comprises a first reservoir in fluidic communication with the plurality of driving electrodes, and wherein the solution comprising an active molecule is held in the reservoir until the solution comprising an active molecule is required for delivery.
14. The method of claim 13, wherein the active molecule delivery system further comprises a second reservoir and a second passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer, wherein the first reservoir is only in fluidic communication with the first passage and the second reservoir is only in fluidic communication with the second passage.
15. The method of claim 14, wherein the first reservoir contains a first solution comprising the active molecule at a first concentration and the second reservoir contains a second solution comprising the active molecule at a second concentration.
16. The method of claim 13, wherein the active molecule delivery system further comprises a second reservoir and a mixing area in fluidic communication with both the first reservoir and the second reservoir.
17. The method of claim 16, wherein the first reservoir contains a first solution comprising a first precursor molecule and the second reservoir contains a second solution comprising a second precursor molecule, and wherein delivering an active molecule to the skin of a subject further comprises: mixing the first precursor molecule with the second precursor molecule to create a mixture; and moving the mixture to the first passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer.
18. The method of claim 17, wherein the first precursor molecule is an antibody.
19. The method of claim 17, wherein the first precursor is an oligonucleotide.
20. The method of claim 17, wherein the mixture comprises an opioid.
Description
RELATED APPLICATIONS
[0001] This application claims priority to co-pending U.S. Provisional Patent Application No. 62/745,718, filed Oct. 15, 2018. All patents, publications, and pending applications disclosed herein are incorporated by reference in their entireties.
BACKGROUND
[0002] Digital microfluidic devices use independent electrodes to propel, split, and join droplets in a confined environment, thereby providing a "lab-on-a-chip." Digital microfluidic devices are alternatively referred to as electrowetting on dielectric, or "EWoD," to further differentiate the method from competing microfluidic systems that rely on electrophoretic flow and/or micropumps. A 2012 review of the electrowetting technology was provided by Wheeler in "Digital Microfluidics," Annu. Rev. Anal. Chem. 2012, 5:413-40, which is incorporated herein by reference in its entirety. The technique allows sample preparation, assays, and synthetic chemistry to be performed with tiny quantities of both samples and reagents. In recent years, controlled droplet manipulation in microfluidic cells using electrowetting has become commercially viable; and there are now products available from large life science companies, such as Oxford Nanopore.
[0003] Most of the literature reports on EWoD involve so-called "passive matrix" devices (a.k.a. "segmented" devices), whereby ten to twenty electrodes are directly driven with a controller. While segmented devices are easy to fabricate, the number of electrodes is limited by space and driving constraints. Accordingly, it is not possible to perform massive parallel assays, reactions, etc. in passive matrix devices. In comparison, "active matrix" devices (a.k.a. active matrix EWoD, a.k.a. AM-EWoD) devices can have many thousands, hundreds of thousands or even millions of addressable electrodes. The electrodes are typically switched by thin-film transistors (TFTs) and droplet motion is programmable so that AM-EWoD arrays can be used as general purpose devices that allow great freedom for controlling multiple droplets and executing simultaneous analytical processes.
[0004] Because of the restrictive requirements on the electric field leakage, most advanced AM-EWoD devices are constructed from polycrystalline silicon (a.k.a. polysilicon., a.k.a. poly-Si). However, polysilicon fabrication is substantially more expensive than amorphous silicon fabrication, i.e., the type used in mass-produced active matrix TFTs for the LCD display industry. Polysilicon fabrication processes are more expensive because there are unique handling and fabrication steps for working with polysilicon. There are also fewer facilities worldwide that are configured to fabricate devices from polysilicon. However, because of the improved functionality of polysilicon, Sharp Corporation has been able to achieve AM-EWoD devices that include propulsion, sensing, and heating capabilities on a single active matrix. See, e.g., U.S. Pat. Nos. 8,419,273, 8,547,111, 8,654,571, 8,828,336, 9,458,543, all of which are incorporated herein by reference in their entireties. An example of a complex poly-Si AM-EWoD is shown in FIG. 1.
[0005] Transdermal delivery of bioactive materials is well-established technology. In the most straightforward embodiments the biologically-active component (typically a molecule having molecular weight below about 1000) is incorporated into a polymeric matrix or gel that is placed into contact with a patient's skin. Penetration of the molecule occurs by passive diffusion, often over a period of several hours. The initiation of drug delivery is by application of the patch to the skin. In the current state of the art it is difficult to modulate the rate of delivery of the active ingredient, however, from one particular patch.
SUMMARY
[0006] The invention addresses this need by providing a low power transdermal delivery system whereby the active molecules can be loaded with a digital microfluidic platform and released on demand. Additionally as described below, the invention provides a system for delivering varying concentrations of active molecules from the same delivery system at different times, and for delivering multiple drugs at the same, or different, times from the same patch.
[0007] The invention works by holding an active (i.e. a drug) in a reservoir until it is needed and then it is moved to a porous diffusion layer (e.g., a drug delivery gel) that is in contact with the skin. In one aspect, the invention is an active molecule delivery system comprising a first substrate, a second substrate, a spacer, a porous diffusion layer and a controller. The first substrate includes a plurality of driving electrodes, a dielectric layer covering the plurality of electrodes, and a first hydrophobic layer covering the dielectric layer. The second substrate includes a common electrode, and a second hydrophobic layer covering the common electrode. The spacer separates the first and second substrates and creates a microfluidic region between the first and second substrates. The porous diffusion layer is coupled to the first substrate on a side of the substrate opposed to the first hydrophobic layer, and the first substrate comprises a passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer. The controller is operatively coupled to the driving electrodes and configured to provide a voltage gradient between at least two driving electrodes. The porous diffusion layer may be constructed from a variety of materials, such acrylate, methacrylate, polycarbonate, polyvinyl alcohol, cellulose, poly(N-isopropylacrylamide) (PNIPAAm), polylactic-co-glycolic acid) (PLGA), polyvinylidene chloride, acrylonitrile, amorphous nylon, oriented polyester, terephthalate, polyvinyl chloride, polyethylene, polybutylene, polypropylene, polyisobutylene, or polystyrene. Typically, the reservoir has a volume greater than 100 nL, and the porous diffusion layer has an average pore size of between 1 nm and 100 nm. In some embodiments, a device includes a plurality of passages. In some embodiments, the passages include wicking materials such as capillary tubes or fibers. In some embodiments, the porous diffusion layer is coupled to a subject with a biocompatible adhesive.
[0008] Typically, the active molecule is a pharmaceutical compound; however, systems of the invention can be used to deliver hormones, nutraceuticals, proteins, nucleic acids, antibodies, or vaccines. The invention may include a plurality of reservoirs and the device may be configured to mix components prior to administering the components. For example, it is possible to have different reservoirs within the same device containing different mixtures or similar mixtures having different concentrations. For example, a system may include a first reservoir containing a mixture of first active molecules and a second reservoir containing a mixture of second active molecules, or a system may include a first reservoir containing active molecules at a first concentration and a second reservoir containing the same active molecules at a second concentration. In other embodiments, the system may include a first reservoir containing a mixture of active molecules and a second reservoir containing an adjuvant and/or a skin penetrant. Other combinations of active molecules, agents, and concentrations will be evident to one of skill in the art.
[0009] The invention additionally includes a controller for controlling an active molecule delivery system. This controller includes an active molecule delivery system as described above, that is, including a mixture of an active molecule dispersed in a first charged phase and a second phase that is oppositely charged or uncharged and immiscible with the first phase, or a mixture of an active molecule and charged particles. The controller may also include a switch configured to interrupt flow from the voltage source to the active molecule delivery system. The switch may be a mechanical switch or a digital switch, and the controller may include a processor for controlling the switch. In some embodiments, the controller will include a wireless receiver and a wireless transmitter, thereby allowing the controller to be interfaced with a device, such as a smart phone, docking station, smart watch, fitness tracker, etc.
[0010] Devices of the invention can be used to deliver active molecules to the skin of a subject. For example, using a device of the invention a the porous diffusion layer can be coupled to the skin of a subject, a solution comprising an active molecule can be moved from a driving electrode to the first passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer, and an active molecule allowed to pass from the porous diffusion layer to the skin of the subject. In some embodiments, the active is held in a first reservoir in fluidic communication with the plurality of driving electrodes, and the solution comprising an active molecule is held in the reservoir until the solution comprising an active molecule is required for delivery. In some embodiments, a device has two separate reservoirs and a mixing area and an active molecule is delivered to a patient. The first reservoir includes a first precursor solution, the second reservoir includes a second precursor solution, and the delivery function includes mixing the first precursor molecule with the second precursor molecule to create a mixture and moving the mixture to a passage providing fluidic communication between the hydrophobic layer and the porous diffusion layer.
BRIEF DESCRIPTION OF DRAWINGS
[0011] FIG. 1 shows a prior art EWoD device including both propulsion and sensing on the same active matrix.
[0012] FIG. 2 depicts the movement of an aqueous-phase droplet between adjacent electrodes by providing differing charge states on adjacent electrodes.
[0013] FIG. 3 depicts a cross-section of the invention when the device is in an "off state" that is, the active has not been loaded.
[0014] FIG. 4 depicts a cross-section of the invention when the device is in an "on state" that is, the active has been loaded.
[0015] FIG. 5 is a plan view of a device of the invention including a reservoir of active material, driving electrodes, passages and a porous diffusion layer. FIG. 5 shows step-by-step (bottom to top) dispensing a solution including an active molecule, moving the droplets toward passages, movement to the passages, and dispensing the droplets into the porous diffusion layer.
[0016] FIG. 6 illustrates a device of the invention wherein each passage is coupled to only a single reservoir. In some embodiments, each reservoir contains a different concentration of active molecules, thereby allowing the dosage to be dynamically controlled.
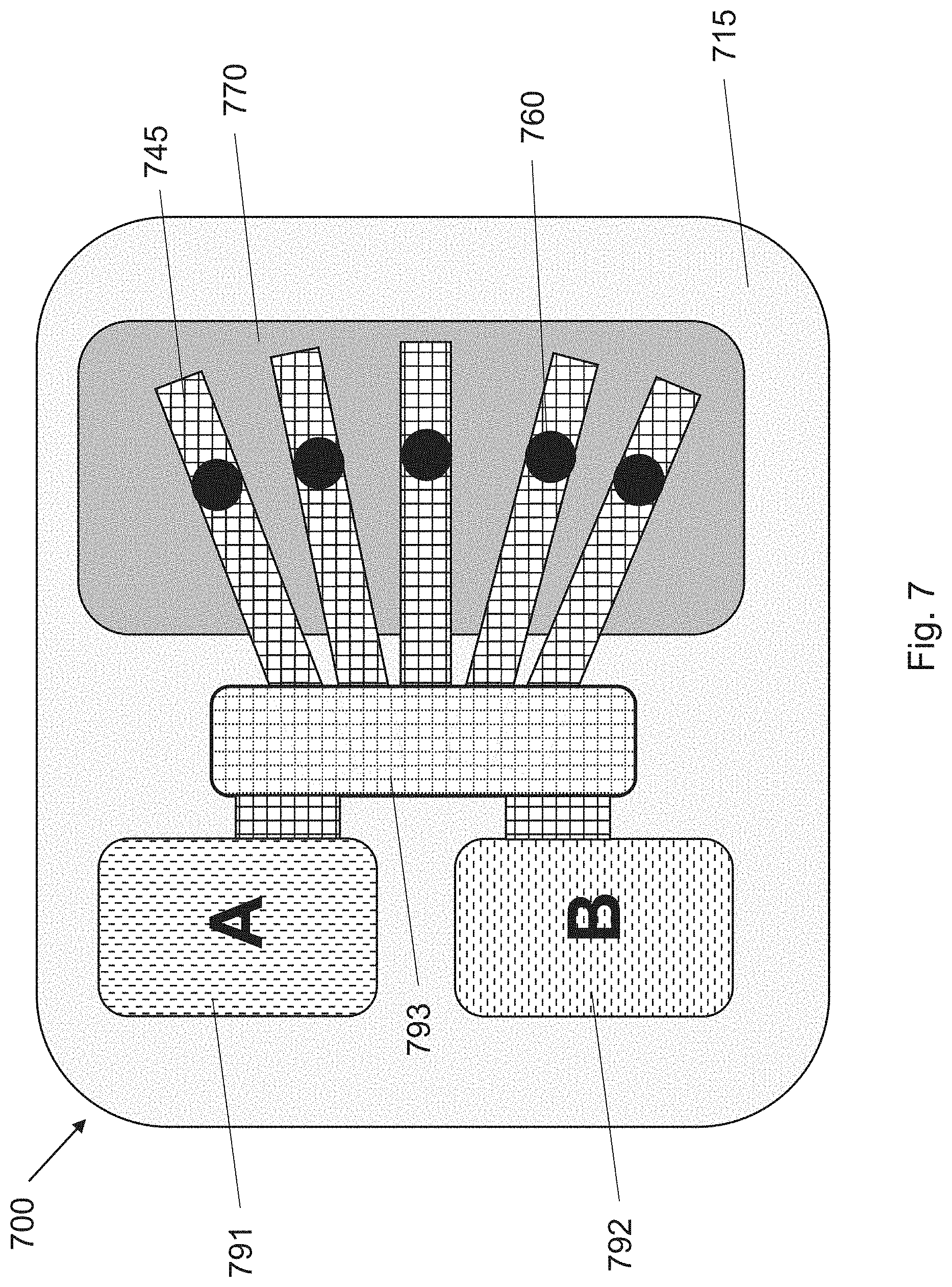
[0017] FIG. 7 illustrates a device of the invention whereby two different reservoirs are coupled to a mixing area whereby two components "A" and "B" can be mixed prior to delivery to the porous diffusion layer.
[0018] FIG. 8 illustrates a delivery device of the invention wherein the driving electrodes are flexible, thereby allowing the delivery device to be wrapped around a limb, e.g., an arm or a leg.
DETAILED DESCRIPTION
[0019] The invention provides an active molecule delivery system whereby active molecules can be released on demand and/or a variety of different active molecules can be delivered from the same system and/or different concentrations of active molecules can be delivered from the same system. The invention is well-suited for delivering pharmaceuticals to patients transdermally, however the invention may be used to deliver active ingredients to animals, generally. The active delivery system includes a plurality of reservoir(s), wherein the reservoir(s) are filled with a medium for delivering the active molecules. In some embodiments the medium includes active molecules dispersed in a first charged phase as well as a second phase that is oppositely charged or uncharged and immiscible with the first phase.
[0020] One molecule of interest for transdermal delivery is naloxone, a competitive opioid receptor antagonist that is used to prevent or reduce the effects of overdose of opioid narcotics. Naloxone is poorly absorbed when taken by mouth and is typically administered by injection or by means of nasal spray. Unfortunately, the effects of the drug diminish after less than about an hour, requiring the administration of several doses to maintain therapeutic levels over long periods. Prior work has suggested, however, that use of a transdermal patch of about 40 cm.sup.2 in area may be able maintain useful plasma concentrations of naloxone over a period of about 4-48 hours. See, e.g., Panchagula, R., Bokalial, R.., Sharma, P. and Khandavilli, S International Journal of Pharmaceutics, 293 (2005), 213-223. Thus, a combination of an initial injected dose with a longer-acting transdermal infusion might provide a practical means to maintain a therapeutic concentration of naloxone while avoiding subjecting a patient to multiple injections.
[0021] Military and civilian first responders, including law-enforcement personnel, face the possibility of exposure to high concentrations of opioid narcotics in dangerous situations in which access to conventional medical care may be impossible. Ideally, they would be able to self-administer an initial bolus of naloxone (or a similar drug) and at the same time be able to trigger longer-term release of a maintenance dose. It might in some situations even be necessary that release of the drug be triggered remotely, especially when the affected individual's ability to function independently has been compromised. In these situations it would be preferred to use a pre-applied transdermal patch in. a state in which the drug to be delivered is in some way unavailable. A triggering event would release the drug, allowing it to begin to diffuse through the skin. Of course, delivery device are not limited to these examples and can be used to deliver, e.g., hormones, nutraceuticals, proteins, nucleic acids, antibodies, or vaccines.
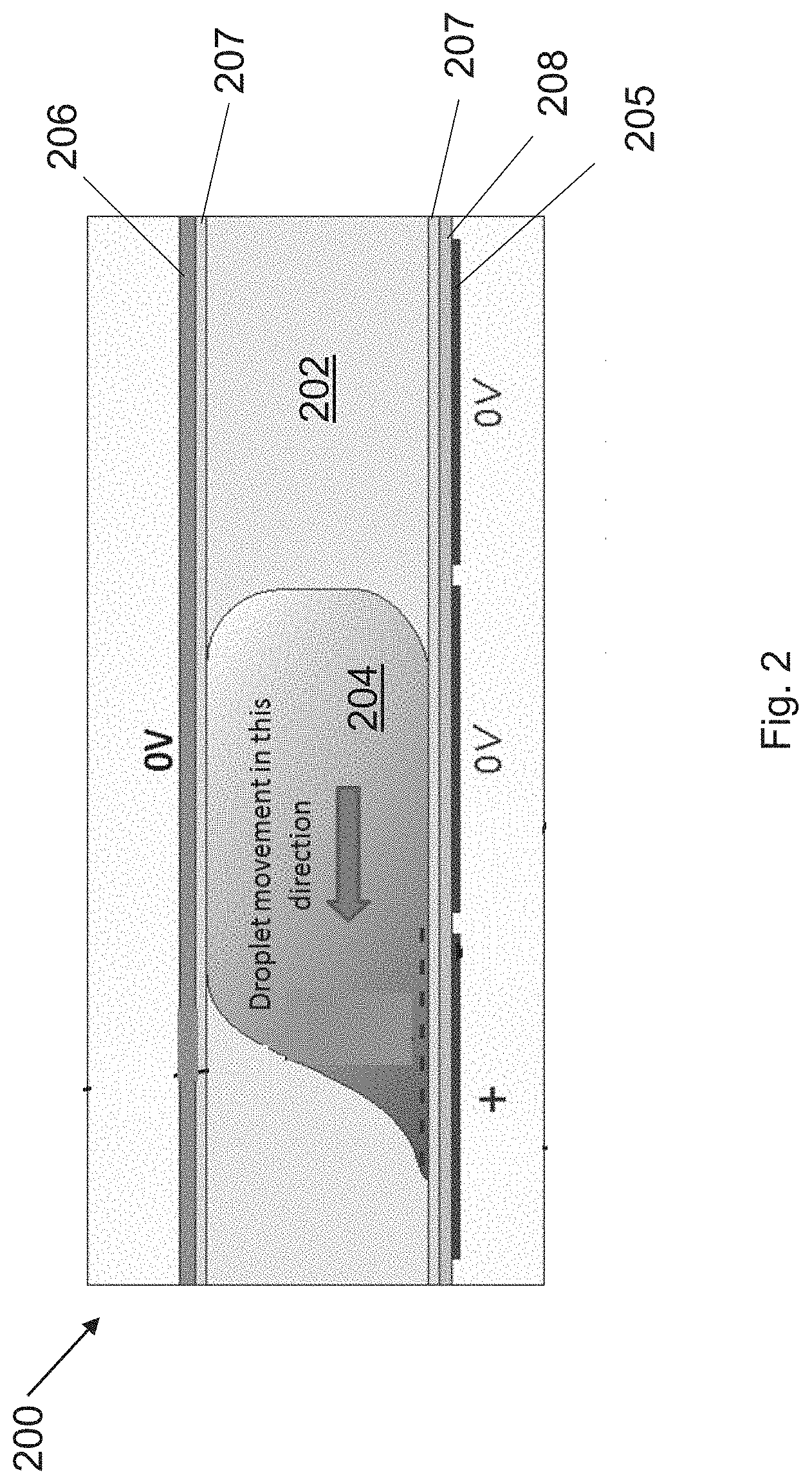
[0022] Devices of the invention function by moving aqueous droplets of actives using electrowetting on dielectric (EWoD). The fundamental operation of an EWoD device is illustrated in the sectional image of FIG. 2. The EWoD 200 includes a cell filled with an oil 202 and at least one aqueous droplet 204. The cell gap is typically in the range 50 to 200 .mu.m, but the gap can be larger. In a basic configuration, as shown in FIG. 2, a plurality of driving electrodes 205 are disposed on one substrate and a singular top electrode 206 is disposed on the opposing surface. The cell additionally includes hydrophobic coatings 207 on the surfaces contacting the oil layer, as well as a dielectric layer 208 between the driving electrodes 205 and the hydrophobic coating 207. (The upper substrate may also include a dielectric layer, but it is not shown in FIG. 2). The hydrophobic layer prevents the droplet from wetting the surface. When no voltage differential is applied between adjacent electrodes, the droplet will maintain a spheroidal shape to minimize contact with the hydrophobic surfaces (oil and hydrophobic layer). Because the droplets do not wet the surface, they are less likely to contaminate the surface or interact with other droplets except when that behavior is desired.
[0023] While it is possible to have a single layer for both the dielectric and hydrophobic functions, such layers typically require thick inorganic layers (to prevent pinholes) with resulting low dielectric constants, thereby requiring more than 100V for droplet movement. To achieve low voltage actuation, it is better to have a thin inorganic layer for high capacitance and to be pinhole free, topped by a thin organic hydrophobic layer. With this combination, it is possible to have electrowetting operation with voltages in the range +1-10 to +/-50V, which is in the range that can be supplied by conventional TFT controllers.
[0024] When a voltage differential is applied between adjacent electrodes, the voltage on one electrode attracts opposite charges in the droplet at the dielectric-to-droplet interface, and the droplet moves toward this electrode, as illustrated in FIG. 2. The voltages needed for acceptable droplet propulsion depend on the properties of the dielectric and hydrophobic layers. AC driving is used to reduce degradation of the droplets, dielectrics, and electrodes by various electrochemistries. Operational frequencies for. EWoD can be in the range 100 Hz to 1 MHz, but lower frequencies of 1 kHz or lower are preferred for use with TFTs that have limited speed of operation.
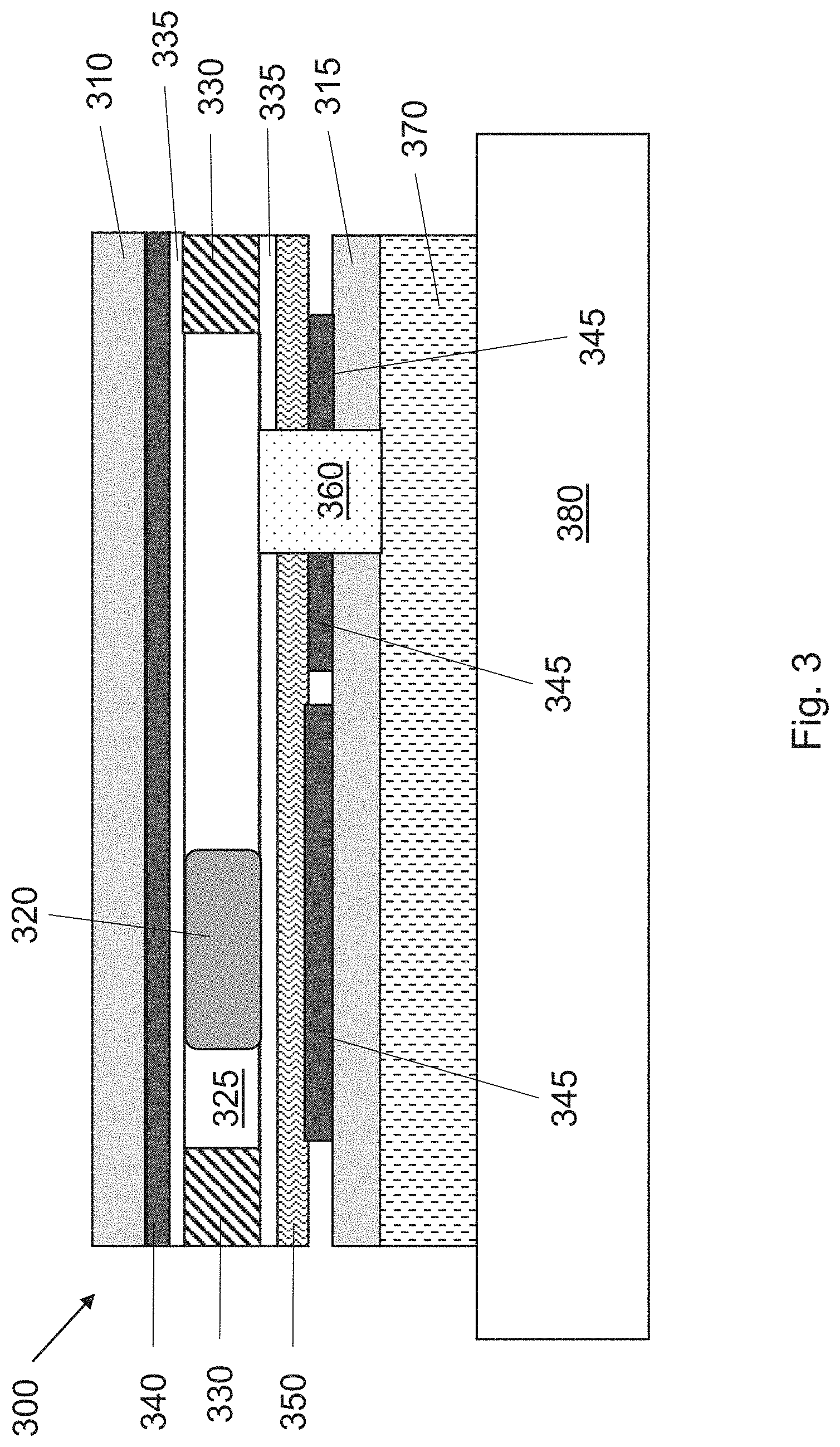
[0025] FIG. 3 shows the principle of operation of an active molecule delivery device 300 of the invention in cross-sectional form (not to scale) when disposed upon skin 380. The device 300 is constructed on one or more substrates 310/315, which may he a flexible substrate. A common electrode 340 is spaced from a driving electrode 345 by a spacer 330. The active molecule (drug) to be delivered is dissolved in a water drop 320 suspended in an incompatible solvent 325 (for example, a hydrocarbon, silicone, or fluorinated organic oil). As discussed above, application of appropriate voltages to the common electrode 340 and the driving electrodes 345 can be used to move the water drop 320 laterally toward the passage 360 which is coupled to the porous diffusion layer 370 (from left to right). To facilitate movement of the water drop 320, a hydrophobic layer 335 is provided below the common electrode 340 and above the driving electrodes 345. A dielectric layer 350 is intermediate between the hydrophobic layer 335 and the driving electrodes 345.
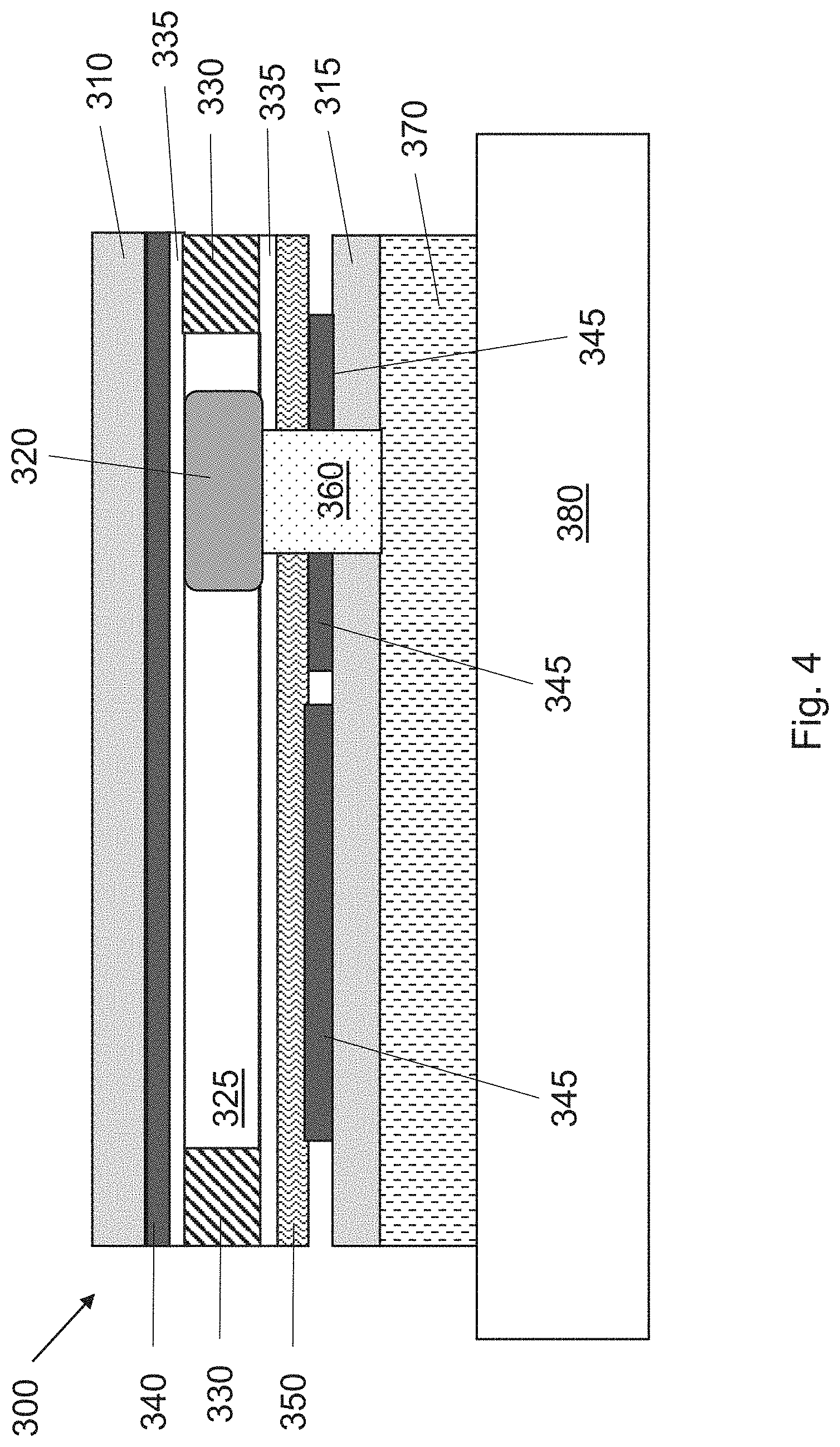
[0026] A device of the invention includes one or more passages 360 (i.e., a channel in the z-direction), either through or adjacent to a driving electrode 345. When a water drop 320 is positioned over such a passage 360, as shown in FIG. 4, the active molecules can move through the passage 360 to the porous diffusion layer 370 that is in contact with the delivery surface, e.g., a patient's skin 380. At this location, diffusional contact is established between the water droplets 320 and the porous diffusion layer 370 that is in contact with the skin 380. Typically, the passage 360 will include structures to facilitate movement of the solvent and active mixture from the hydrophobic surface 335 to the porous diffusion layer 370. For example, the passage may include materials that move hydrophilic materials through capillary action, such as wicking fibers, cellulose, or cotton. Such materials may be coated with an additional hydrophobic coating to facilitate movement of the aqueous solution from the electrowetting surface to the porous diffusion layer 370. In some embodiments, a biocompatible adhesive (not shown) may laminated to the porous diffusion layer. The biocompatible adhesive will allow active molecules to pass through while keeping the device immobile on a user. Suitable biocompatible adhesives are available from 3M (Minneapolis, Minn.).
[0027] FIG. 5 shows a top view of an embodiment of an active molecule delivery device 500 of the invention as if the top electrode and the top substrate have been removed. The device 500 includes a substrate 515, driving electrodes 545, passages 560, and a porous diffusion layer 570. The device additionally includes a controller 543 that is coupled to the driving electrodes 545 with traces 547. As shown in FIG. 5 a hydrophilic liquid containing a target molecule (drug) to be delivered is located in a reservoir 590, i.e., a region of the device remote from the porous diffusion layer that contacts the skin.
[0028] The sequence of delivery of a solution including an active molecule is illustrated in FIG. 5 running from the bottom driving electrodes to the top driving electrodes. Initially, droplets 520 are snapped off from the reservoir 590 thereby determining the dosage that will be delivered (concentration.times.volume.times.number of droplets). The droplets 520 are advanced until they are adjacent one or more passages 560, whereupon an auxiliary driving electrode 548 that surrounds the passage 560 is used to move the droplets 520 above the passages 560 using orthogonal electrowetting. Due to the wicking action of the passage 560, the droplets 520 will move into and through the passages 560 whereupon they are delivered to the porous diffusion layer 570 below. Accordingly, the active ingredients in the reservoir(s) 590 can be moved into the porous diffusion layer.
[0029] It is possible to imagine numerous different arrangements of driving electrodes 545 with respect to the passages 560. In a second embodiment, illustrated in FIG. 6, electrowetting forces are used to withdraw fluid 620 from the reservoir 690 and transfer it directly to a passage 660. The device 600 includes a substrate 615, driving electrodes 645, passages 660, and a porous diffusion layer 670. The device additionally includes a controller 643 that is coupled to the driving electrodes 645 with traces 647. No orthogonal motion is needed in FIG. 6, however. Capillary forces, provided by suitable materials between the driving electrodes 645 and the porous diffusion layer 670, withdraw fluid 620 from the reservoir 690. While the fluid 620 is shown to be continuous, it is to be understood that the fluid can be delivered in droplets as in FIG. 5. As shown in FIG. 6, each passage 660 is coupled to a unique reservoir 690. This allows each reservoir to act as a single dose, thereby reducing the complexity of the system, e.g., whereby a specific number of droplets of a specific volume of a specific dosage must be snapped off and delivered. For example, a device 600 may include seven identical reservoirs and the controller 643 configured to administer the contents of one reservoir each morning for seven consecutive days. Alternatively, different reservoirs 690 may each contain different concentrations of the same active so that, for example, a subject can receive a first stronger dose of the active and then receive one or more lower concentration maintenance doses during the day. Such a device may be particularly well suited for delivering hormones.
[0030] Another embodiment of the invention is shown in FIG. 7, where the device 700 includes a substrate 715, driving electrodes 745, passages 760, and a porous diffusion layer 770. While a controller is not shown, it is understood that a controller is needed to coordinate functions of the driving electrodes 745. FIG. 7 illustrates that it is possible to do reactions "on chip" before delivering an active molecule. As shown in FIG. 7, a first reservoir "A" 791 and a second reservoir "B" 792 are both in fluidic communication with a mixing area 793. A first precursor molecule can be contained in a first solution in the first reservoir 791 while a second precursor molecule can be contained in a second solution in the second reservoir 792. Prior to administering an active, the first solution and the second solution are brought to the mixing area 793 where they are allowed to mix to create the target active, which is then delivered to the porous diffusion layer 770 in a method described above.
[0031] There are many advantages to a device 700 with the ability to mix precursors prior to delivering a target active to the porous diffusion layer 770. For example, the first precursor may be a sensitive biologic, such as an antibody or an oligonucleotide that must be stabilized for storage in a solution that is not suitable for delivery through the porous diffusion layer. Accordingly, when it is appropriate to deliver the biologic, an amount is transferred from the first reservoir 791 to the mixing area 793 where the biologic can be activated, cleaned, or targeted for delivery via conjugation with promotors, markers, or other target specific molecules). Such configurations may greatly increase the shelf life of the biologics and they may allow a patient to avoid having to go to a clinic to have the biologic delivered via intravenous injection. In other alternatives, the first and second precursors may be prodrugs that combine to create an opioid. Using a device of the invention, it may he possible to prevent illegal opioid administration because only users having the device and appropriate security authorization could combine the precursors to create the opioid.
[0032] The system of FIG. 7 may also be suitable for delivering so-called "drug cocktails" that include active molecules that deactivate each other over time and typically must be administered in a clinic, e.g., a chemotherapy clinic. The system of FIG. 7 may also be used to deliver, e.g,, a patient's own cells, antibodies, etc. In such embodiments, the patient's own biological materials may be held in a first reservoir and when it is appropriate to deliver the therapeutic, the patient's own biological materials are moved to the mixing area where they are combined with another active ingredient prior to being delivered to the porous diffusion layer.
[0033] The controller 543, 643, 743, may comprise a battery and electronics required to initiate the electrowetting motion, and means to communicate with the outside world such as appropriate electronics/antenna, etc. In preferred arrangements, it will not be possible to transfer drops of water containing the drug to the passages without applying electrical signals. This will ensure that the unactivated patch may be subjected to a variety of stresses (mechanical, thermal, etc.) without releasing the active ingredient.
[0034] In one embodiment, a device of the invention can be used to deliver naloxone (NARCAN.TM.). The device would deliver about 20-100 mg of the drug from the reservoir to the porous diffusion area. Assuming a near-saturated concentration of active (e.g., naloxone) in water in the reservoir of 50 mg/mL, the volume of drops required to be delivered would be about 400-2000 .mu.L, which is within the capability of the devices. In other embodiments, devices of the invention can be used to deliver opioids, e.g., hydromorphone, hydrocodone, fentanyl, methadone, or oxycodone. Devices of the invention may be used to deliver stimulants such as nicotine, steroids (e.g., prednisone), and hormones (e.g., epinephrine).
[0035] In some embodiments, a device of the invention can be made to be flexible so that the device can deployed on a curved surface 880 and/or integrated into a flexible package to improve user comfort and compliance. An embodiment of such a device 800 is shown in FIG. 8, whereby flexible drive electrodes 845 are coupled to a porous diffusion layer 870 and passages 860 provide fluidic communication between the drive electrodes 845 and the porous diffusion layer 870. As shown in FIG. 8, the controller 843 and the reservoir 890 may be combined into the same housing. In an embodiment, the device 800 may take the shape of a wristband, whereby the device 800 may be additionally augmented with decorative designs to hide that the device 800 is, in fact, for delivering drugs transdermally.
[0036] Advanced embodiments of an active molecule delivery system will include circuitry to allow the active molecule delivery system to be controlled wirelessly with a secondary device, such as a smart phone or smart watch. With such improvements, a user can control, for example, the type of active molecule that is delivered and the amount that is delivered. Using an application on, e.g., a smart phone or watch, it may be possible to program the device to modify the amount of active molecule delivered based upon the time of day. In other embodiments, the device may be operatively coupled with biometric sensors, e.g., a fitness tracker or heart rate monitor, whereby the application causes the dosage to be turned off if, e.g., the pulse rate of the user exceeds a preset threshold. Other embodiments may couple, e.g., a readout from a glucose monitor to the device to allow for automatic delivery of insulin when a patient is outside of their desired blood glucose level.
[0037] When desired, devices of the invention can be activated and/or controlled remotely. For example, NFC, Bluetooth, WIFE, or other wireless communication function may be used to activate a device and cause the agent to be administered. Furthermore, the same wireless communication may be used to monitor the performance of the device, e.g., the percentage and area for all of the reservoir(s) at different driving status is known, which means all of the usage data will be available to a provider or therapist, including when the patch is activated and what amount of active is administered. For the "programmable" feature, because each reservoir can be turned independently, the overall release profile of the device can be programmed by driving differing concentrations of actives or different actives from different reservoirs at different times. Additionally, patient compliance is also good because the smart device that is used to activate the patch can also communicate with doctors remotely for data sharing.
[0038] It will be apparent to those skilled in the art that numerous changes and modifications can be made in the specific embodiments of the invention described above without departing from the scope of the invention. Accordingly, the whole of the foregoing description is to be interpreted in an illustrative and not in a limitative sense.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.