Process And Apparatus For Treating Mercaptans In A Naphtha Boiling Range Feed
HE; Zunqing Alice ; et al.
U.S. patent application number 16/545001 was filed with the patent office on 2020-04-09 for process and apparatus for treating mercaptans in a naphtha boiling range feed. The applicant listed for this patent is CHEVRON U.S.A. INC.. Invention is credited to Zunqing Alice HE, Stephen Edward LEICHTY, Cameron Adams McCORD.
| Application Number | 20200109337 16/545001 |
| Document ID | / |
| Family ID | 70051987 |
| Filed Date | 2020-04-09 |
| United States Patent Application | 20200109337 |
| Kind Code | A1 |
| HE; Zunqing Alice ; et al. | April 9, 2020 |
PROCESS AND APPARATUS FOR TREATING MERCAPTANS IN A NAPHTHA BOILING RANGE FEED
Abstract
Processes and apparatuses are disclosed for treating a naphtha boiling range stream containing mercaptan compounds. The process includes oxidizing mercaptan compounds in the naphtha boiling range stream to provide a mercaptan-depleted naphtha stream rich in disulfide compounds; passing the mercaptan-depleted naphtha stream rich in disulfide compounds to a naphtha splitter column; and fractionating at least a portion of the mercaptan-depleted naphtha stream rich in disulfide compounds into at least two streams, a light naphtha stream lean in disulfide compounds and a heavy naphtha stream rich in disulfide compounds. The heavy naphtha stream rich in disulfide compounds may be passed to a hydroprocessing unit to convert organic disulfides in the stream to hydrocarbons and hydrogen sulfide. A heavy naphtha stream lean in disulfide compounds can be recovered and routed as desired by the refiner.
| Inventors: | HE; Zunqing Alice; (San Rafael, CA) ; LEICHTY; Stephen Edward; (Boulder, CO) ; McCORD; Cameron Adams; (Martinez, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 70051987 | ||||||||||
| Appl. No.: | 16/545001 | ||||||||||
| Filed: | August 20, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62742603 | Oct 8, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 7/00 20130101; C10G 2300/1025 20130101; C10G 2300/202 20130101; C10G 2300/1044 20130101; C10G 27/04 20130101; C10G 53/14 20130101; C10G 2300/4018 20130101; C10G 2300/104 20130101 |
| International Class: | C10G 53/14 20060101 C10G053/14; C10G 27/04 20060101 C10G027/04; C10G 7/00 20060101 C10G007/00 |
Claims
1. A process for treating a naphtha boiling range stream containing mercaptan compounds, the process comprising: (a) oxidizing mercaptan compounds in the naphtha boiling range stream to provide a mercaptan-depleted naphtha stream rich in disulfide compounds; (b) passing the mercaptan-depleted naphtha stream rich in disulfide compounds to a naphtha splitter column; and (c) fractionating at least a portion of the mercaptan-depleted naphtha stream rich in disulfide compounds into at least two streams, a light naphtha stream lean in disulfide compounds and a heavy naphtha stream rich in disulfide compounds.
2. The process of claim 1, wherein the naphtha boiling range stream comprises straight-run naphtha.
3. The process of claim 1, wherein oxidizing mercaptan compounds comprises contacting the mercaptan compounds in the naphtha boiling range stream with an oxidizing catalyst and oxygen.
4. The process of claim 3, wherein the oxidizing catalyst comprises a metal component retained on an inorganic oxide support, wherein the metal component is selected from one or more of vanadium, chromium, manganese, cobalt, nickel, copper, and zinc.
5. The process of claim 4, wherein the metal component comprises copper.
6. The process of claim 5, wherein the copper is present as copper metal, copper oxide, copper sulfide, or a combination thereof.
7. The process of claim 4, wherein the metal component is present in an amount of from 10 to 40 wt. %, based on a total weight of the catalyst.
8. The process of claim 4, wherein the inorganic oxide support is selected from alumina, silica, zirconia, titania, thoria, boria, magnesia, chromia, stannic oxide, and combinations thereof.
9. The process claim 1, wherein conditions for oxidizing mercaptan compounds include a pressure of from 101 to 2068 kPa; a temperature of from 35.degree. C. to 200.degree. C.; and a liquid hourly space velocity of from 1 to 10 h.sup.-1 (e.g., 1 to 5 h.sup.-1).
10. The process of claim 1, wherein mercaptan compounds in the naphtha boiling range stream are depleted in the substantial absence of any caustic solvent.
11. The process of claim 1, wherein the straight-run naphtha is derived from crude oil or natural gas condensate resources.
12. An apparatus for treating a naphtha boiling range stream containing mercaptan compounds, the apparatus comprising: (a) an oxidation unit to provide a mercaptan-depleted naphtha stream rich in disulfide compounds; and (b) a naphtha splitter column in downstream communication with the oxidation unit to provide a light naphtha stream lean in disulfide compounds in a naphtha splitter overhead line and a heavy naphtha stream rich in disulfide compounds in a naphtha splitter bottoms line.
13. The apparatus of claim 12, further comprising a hydroprocessing unit in downstream communication with the naphtha splitter bottoms line to provide a heavy naphtha stream lean in disulfide compounds.
14. The apparatus of claim 12, further comprising a distillation unit in upstream communication with the oxidation unit to provide a naphtha boiling range stream.
15. The apparatus of claim 14, wherein the distillation unit is a crude oil atmospheric distillation unit or a natural gas condensate splitter column.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and the benefit of U.S. Provisional Application Ser. No. 62/742,603, filed Oct. 8, 2018.
FIELD
[0002] The present disclosure generally relates to processes and apparatuses for treating petroleum fractions. More particularly, the field relates to an improved process and apparatus for removing mercaptan compounds from petroleum fractions such as naphtha.
BACKGROUND
[0003] Most distillate hydrocarbon streams produced from crude oil contain some amount of sulfur in one form or another unless these streams have been subjected to extensive sulfur removal procedures such as hydrotreating. Often a major amount of this sulfur is present in the form of mercaptans. Mercaptan sulfur content must be reduced in the hydrocarbon distillate stream in order to meet certain product specifications such as a limitation on the total sulfur content of a product. It may also be desirable to remove mercaptan compounds from a hydrocarbon stream for the purpose of eliminating the rather malodorous mercaptan compounds and thereby improve or reduce the odor associated with the hydrocarbon stream. Another reason for removing mercaptan compounds from a hydrocarbon stream would be to eliminate the passage of sulfur-containing compounds into a catalyst bed which is sensitive to the presence of sulfur. It may therefore be necessary to remove mercaptans from a hydrocarbon distillate stream for the purpose of preserving the activity of a catalyst employed in a downstream conversion unit.
[0004] In many regions, naphtha is useful for motor fuel and petrochemical feedstock and its further recovery is desirable. Due to environmental concerns and newly enacted rules and regulations, saleable fuels must meet lower and lower limits on contaminates, such as sulfur and nitrogen. For example, the U.S. Environmental Protection Agency (EPA) is implementing its Tier 3 Gasoline Sulfur Program, a goal of which is to decrease the average gasoline sulfur content from 30 parts per million (ppm) to 10 ppm across each refining company's total annual output.
[0005] Hydroprocessing, such as hydrotreating, can be effective for reducing the sulfur content of a naphtha boiling range fraction to a desired sulfur level. In many cases, however, the existing hydrotreating assets do not have sufficient available hydraulic capacity to process the petrochemical naphtha stream and building a grass roots hydrotreater for this purpose is a very expensive option.
[0006] Sweetening of petroleum fractions, such as naphtha boiling range hydrocarbons or other liquid hydrocarbons, that contain mercaptans (or sour petroleum fractions) are well-developed commercial processes commonly used in many petroleum refineries. In the sweetening process, mercaptans contained in the feed hydrocarbon stream (e.g., sour hydrocarbon stream) are converted to disulfide compounds that remain in the hydrocarbon stream (e.g., sweetened hydrocarbon stream). Sweetening processes, therefore, do not remove sulfur from the hydrocarbon stream but rather convert the sulfur to an acceptable form. The sweetening process involves an admixture of an oxygen-containing stream to the sour hydrocarbon stream to supply the required oxygen. Typically, the admixture of hydrocarbons and oxygen contacts an oxidation catalyst in an aqueous alkaline environment to oxidize the mercaptans. Typically, a caustic (e.g., an aqueous caustic solution) is combined with the sour hydrocarbon stream to create the aqueous alkaline environment. After contacting the oxidation catalysts, at least a portion of the caustic is carried with the sweetened hydrocarbon stream and can be problematic for further downstream processing. Current approaches for removing caustic from sweetened hydrocarbon streams often require additional downstream equipment and can be costly and/or are relatively inefficient.
[0007] Accordingly, it is desirable to provide improved processes and apparatuses for treating naphtha streams containing mercaptan compounds. Additionally, it is desirable to provide such processes and apparatuses that can sweeten naphtha streams without the need for caustic and separate washing apparatus/procedures.
SUMMARY
[0008] In one aspect, there is provided a process for treating a naphtha boiling range stream containing mercaptan compounds, the process comprising: (a) oxidizing mercaptan compounds in the naphtha boiling range stream to provide a mercaptan-depleted naphtha stream rich in disulfide compounds; (b) passing the mercaptan-depleted naphtha stream rich in disulfide compounds to a naphtha splitter column; and (c) fractionating at least a portion of the mercaptan-depleted naphtha stream rich in disulfide compounds into at least two streams, a light naphtha stream lean in disulfide compounds and a heavy naphtha stream rich in disulfide compounds.
[0009] In another aspect, there is provided an apparatus for treating a naphtha boiling range stream containing mercaptan compounds, wherein the apparatus comprises: (a) an oxidation unit to provide a mercaptan-depleted naphtha stream rich in disulfide compounds; and (b) a naphtha splitter column in downstream communication with the oxidation unit to provide a light naphtha stream lean in disulfide compounds in a naphtha splitter overhead line and a heavy naphtha stream rich in disulfide compounds in a naphtha splitter bottoms line.
BRIEF DESCRIPTION OF THE DRAWINGS
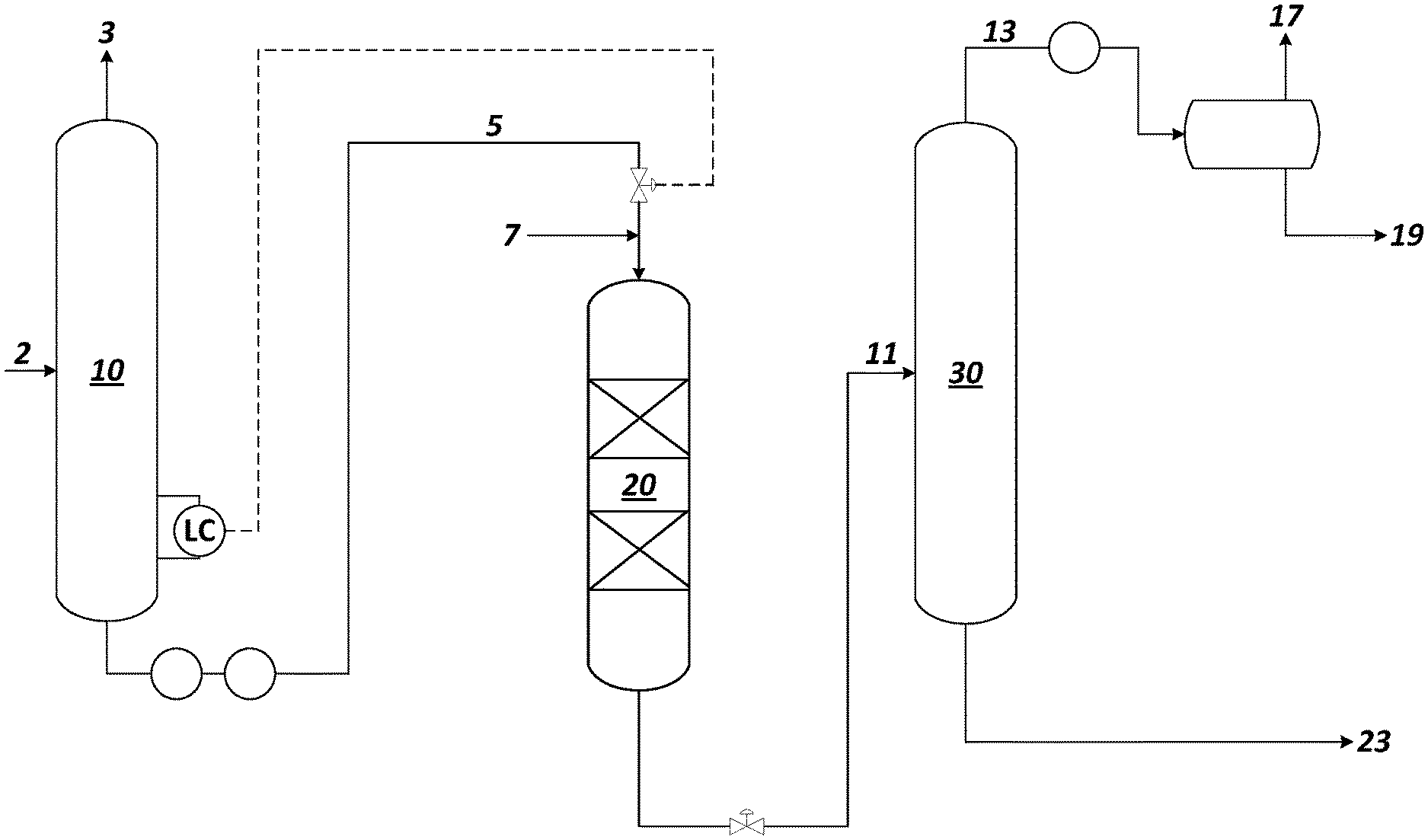
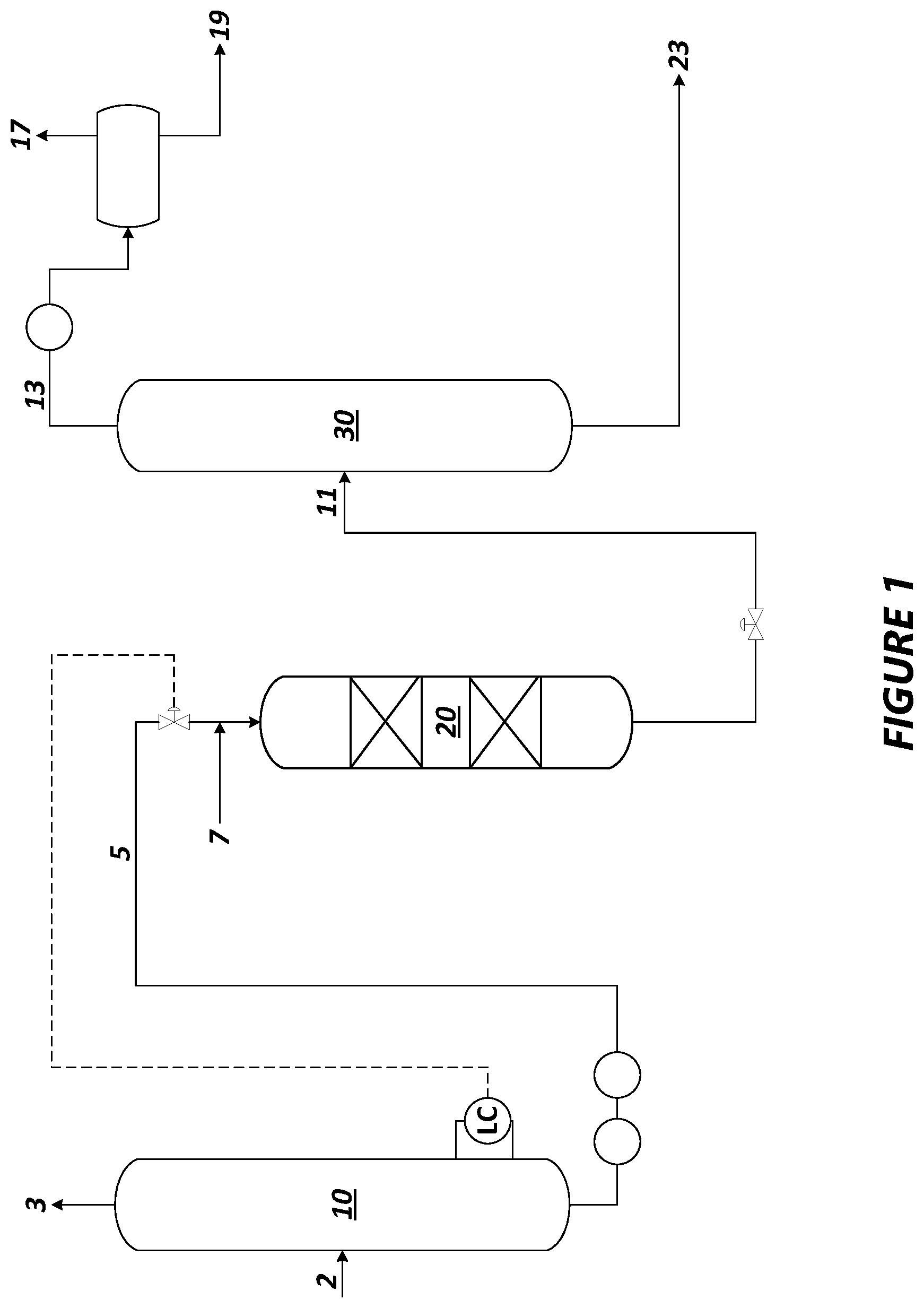
[0010] The FIG. 1s a process flow diagram illustrating an exemplary process and apparatus of the present disclosure.
DETAILED DESCRIPTION
[0011] In this specification, the following words and expressions, if and when used, have the meanings ascribed below.
[0012] The term "naphtha" or "naphtha boiling range" refers to hydrocarbons boiling in a range of about 25.degree. C. to 190.degree. C. atmospheric equivalent boiling point (AEBP), as determined by any standard gas chromatographic simulated distillation method such as ASTM D2887, and can include one or more C.sub.5-C.sub.10 hydrocarbons.
[0013] The term "light naphtha" refers to hydrocarbons boiling in a range of about 25.degree. C. to 85.degree. C., and can include one or more C.sub.5-C.sub.6 hydrocarbons.
[0014] The term "heavy naphtha" refers to hydrocarbons boiling in the range of about 85.degree. C. to 190.degree. C. (e.g., 110.degree. C. to 170.degree. C.), and can include one or more C.sub.6-C.sub.10 hydrocarbons.
[0015] The modifying term "straight-run" is used herein having its well-known meaning, that is, describing fractions derived directly from the atmospheric distillation unit, optionally subjected to steam stripping, without other refinery treatment such as hydroprocessing, fluid catalytic cracking or steam cracking. An example of this is "straight-run naphtha" and its acronym "SRN" which accordingly refers to "naphtha" defined above that is derived directly from the atmospheric distillation unit, optionally subjected to steam stripping, as is well known. Straight-run naphtha may also be derived from natural gas condensates.
[0016] The term "rich" refers to a stream exiting a vessel that has a concentration of one or more compounds exceeding a stream entering the vessel.
[0017] The term "lean" refers to a stream exiting a vessel that has a concentration of one or more compounds less than a stream entering the vessel.
[0018] The term "depleted" is synonymous with reduced from originally present. For example, removing a substantial portion of a material from a stream would produce a material-depleted stream that is substantially depleted of that material.
[0019] The term "mercaptan" means thiol compounds of the formula R--SH where R is a hydrocarbon group, such as an alkyl or aryl group, that is saturated or unsaturated and optionally substituted,
[0020] The term "disulfide" means compounds having the molecular formula R--S--S--R' where R and R' are each, independently, a hydrocarbon group, such as an alkyl or aryl group, that is saturated or unsaturated and optionally substituted. Generally, the term "disulfide" as used herein excludes carbon disulfide (CS.sub.2).
[0021] The notation "C.sub.x" means hydrocarbon molecules that have "x" number of carbon atoms, "C.sub.x" means hydrocarbon molecules that have "x" and/or more than "x" number of carbon atoms, and "C.sub.x-" means hydrocarbon molecules that have "x" and/or less than "x" number of carbon atoms.
[0022] The term "communication" means that material flow is operatively permitted between enumerated components.
[0023] The term "downstream communication" means that at least a portion of material flowing to the subject in downstream communication may operatively flow from the object with which it communicates.
[0024] The term "upstream communication" means that at least a portion of the material flowing from the subject in upstream communication may operatively flow to the object with which it communicates.
[0025] Numbers expressing quantities, percentages or proportions, and other numerical values used in the specification and claims are to be understood as being modified in all instances by the term "about." The term "about" can be understood as within 10%, 5%, 2.5%, 1%, 0.5%, 0.1%, 0.05%, or 0.01% of the reported numerical value.
[0026] As depicted, process flow lines in the FIGURE can be referred to interchangeably as, for example, lines, pipes, feeds, gases, products, discharges, parts, portions, conduits or streams.
[0027] The following description is merely exemplary in nature and is not intended to limit the various embodiments or the application and uses thereof. The FIGURE has been simplified by the deletion of a large number of apparatuses customarily employed in a process of this nature, such as vessel internals, temperature and pressure control systems, flow control valves, recycle pumps, etcetera which are not specifically required to illustrate the performance of the invention.
[0028] With reference to the FIGURE, a feed 2 that is a crude oil or a natural gas condensate stream containing a range of hydrocarbons is passed to a distillation unit 10 where the feed is separated into a light stream 3, a naphtha boiling range stream 5, and/or one or more heavier or bottoms fractions. In embodiments where the feed 2 is a crude oil, the distillation unit 10 may be an atmospheric distillation unit. In embodiments where the feed 2 is a natural gas condensate stream, the distillation unit 10 may be a natural gas condensate splitter.
[0029] The feed in line 2 may be dried and/or pre-treated to reduce and/or remove one or more of undesired components such as carbon dioxide, mercury, and water prior to fractionation.
[0030] Naphtha boiling range streams typically contain one or more mercaptan compounds. The mercaptans occurring in naphtha boiling range streams are generally C.sub.1-C.sub.10 mercaptans (e.g., C.sub.1-C.sub.6 mercaptans). The mercaptans are generally concentrated in the light fractions of the naphtha and more precisely in the fraction with a boiling point of less than 120.degree. C. The mercaptan sulfur may be present in the naphtha boiling range stream in an amount ranging from about 2 ppm to 300 wppm or more, depending on the particular stream to be treated.
[0031] Upon exiting the distillation unit 10, the mercaptan-containing naphtha boiling range stream 5 is passed to an oxidation unit 20 where mercaptan compounds in the naphtha stream are converted to disulfide compounds. The flow direction in oxidation unit 20 can be down flow or up flow. The naphtha stream 5 is mixed with an air or other oxygen-containing gas stream 7 supplied at a rate that supplies at least the stoichiometric amount of oxygen necessary to oxidize the mercaptan compounds in the naphtha boiling range stream 5 to disulfide compounds. The mole ratio of oxygen to mercaptan sulfur may range from about 1:4 to 10:1 (e.g., about 1:1 to 10:1, or about 1:1 to 3:1). The oxidation of the mercaptan compounds is promoted through the presence of a catalytically effective amount of an oxidation catalyst capable of functioning at the conditions found in the oxidation unit 20. The oxidation unit 20 may be configured in a packed bed configuration to ensure adequate mixing between the naphtha boiling range feed, the catalyst, and the oxygen. The oxidation unit 20 may comprise a cylindrical fixed bed of catalyst through which the reactants move in a vertical direction.
[0032] The oxidation conditions utilized in oxidation unit 20 may include a pressure of from about 101 to 2068 kPa (gauge) (15 to 300 psig), such as from 172 to 689 kPa (gauge) (25 to 100 psig); a temperature of from 35.degree. C. to 200.degree. C. (e.g. 50.degree. C. to 150.degree. C.); and a liquid hourly space velocity of from 1 to 10 h.sup.-1 (e.g., 1 to 5 h.sup.-1).
[0033] In an aspect, mercaptan compounds are depleted in the naphtha boiling range stream in the substantial absence of any caustic solvent, such as an aqueous solution of alkali metal hydroxide (e.g., sodium hydroxide, potassium hydroxide).
[0034] Suitable oxidation catalysts are any known conventional catalysts for oxidizing mercaptans to disulfides and can include those which are comprised of a Group 5-12 metal component (e.g., one or more of vanadium, chromium, manganese, cobalt, nickel, copper, zinc) retained on a refractory inorganic oxide support.
[0035] The inorganic oxide binder of the catalyst may comprise materials such as alumina, silica, zirconia, titania, thoria, boria, magnesia, chromia, stannic oxide, and the like as well as combinations and composites thereof, for example silica-alumina, alumina-zirconia, alumina-titania, aluminum phosphate, and the like. Alumina is a preferred refractory inorganic oxide binder. As is well known in the art, a precursor of the desired refractory inorganic oxide may be used to form, bind, and/or otherwise prepare the catalyst. Such binder precursors or sources may be converted into a refractory inorganic oxide binder, for example, by calcination. The alumina may be any of the various aluminum oxides, hydroxides, and gels, including boehmite, pseudo-boehmite, gibbsite, bayerite, and the like, especially transition and gamma aluminas. Suitable aluminas are commercially available, for example, under the trade names CATAPAL.COPYRGT. B and VERSAL.TM. 250.
[0036] The metal component of the catalyst may comprise a metal selected from the group consisting of vanadium, chromium, manganese, cobalt, nickel, copper, zinc, and combinations thereof. In one embodiment, the metal component comprises copper. The metal content of the catalyst can range from 10 to 40 wt. % (e.g., 15 to 30 wt. %) as the metal based upon the total weight of the catalyst.
[0037] The metal component may be incorporated into the catalyst in any suitable manner such as co-mulling, co-precipitation or co-gellation with the carrier material, ion exchange, or impregnation. The metal component may exist within the final catalyst as a compound such as an oxide, sulfide, halide, or oxyhalide, in chemical combination with one or more of the other ingredients of the composite, or as an elemental metal. In an embodiment where the metal component is copper, the metal component may be present as copper metal, copper, oxide, copper sulfide, or a combination thereof.
[0038] For practical applications, the catalyst is most preferably used in particulate form, for example as pellets, extrudate, spheres or granules, although other solid forms also are suitable. Particle size of the catalyst is selected such that a bed of catalyst particles is easily maintained in a suitable reactor for the oxidation process but permits flow of the naphtha boiling range through the bed without undesirable pressure drop. Preferred average particle sizes are such that catalyst particles pass through a 2-mesh screen but are retained on a 24-mesh screen (U.S. Sieve Series) and more preferably pass through a 4-mesh screen but are retained on a 12-mesh screen.
[0039] The oxidation catalyst may have a BET surface area in a range of 30 to 300 m.sup.2/g (e.g., 50 to 200 m.sup.2/g).
[0040] In one embodiment, the oxidation catalyst may be a fresh or spent sulfur sorbent having application in eliminating residual sulfur from conventionally desulfurized reformer or isomerization feed streams, such as described in U.S. Pat. No. 4,259,213.
[0041] The effluent from oxidation unit 20 comprises a mercaptan-depleted naphtha stream rich in disulfide compounds. Organic disulfides have higher boiling points than those of their mercaptan precursors. Thus, mercaptans that typically boil in a light naphtha fraction are converted in the oxidation unit 20 into disulfides that typically boil in a heavy naphtha fraction.
[0042] The mercaptan-depleted naphtha stream rich in disulfide compounds in line 11 is passed to a naphtha splitter column 30 in which it is fractionated to provide a light naphtha stream lean in disulfide compounds in a naphtha splitter overhead line 13 and a heavy naphtha stream rich in disulfide compounds in a naphtha splitter bottoms line 23. The light naphtha stream, typically a C.sub.5-C.sub.6 or a C.sub.5-C.sub.7 stream, with reduced sulfur content may be condensed and separated in a receiver with a portion of the condensed liquid being sent in line 19 for blending to the gasoline pool. Any light ends may be vented to an appropriate combustion destination such as a furnace burner of a flare equipped for streams with high levels of oxygen via line 17. The heavy naphtha stream, typically comprising C.sub.7+ naphtha, is rich in disulfide compounds, and may be taken from a bottoms outlet in the naphtha splitter bottoms line 23 for further processing.
[0043] The naphtha splitter column 30 may be operated with a top pressure of 69 to 448 kPa (gauge) (10 to 65 psig) and a bottom temperature of 121.degree. C. to 232.degree. C. (250.degree. F. to 450.degree. F.). Alternatively, the naphtha splitter column 30 may be operated at a vacuum. The naphtha splitter column 30 may include a reboiler at a bottom of the column to vaporize and send a portion of the heavy naphtha stream back to the bottom of the column.
[0044] The heavy naphtha stream rich in disulfide compounds in line 23 may be passed to a hydroprocessing unit to convert organic disulfides in the stream to hydrocarbons and hydrogen sulfide. A heavy naphtha stream lean in disulfide compounds can be recovered and routed as desired by the refiner.
EXAMPLES
[0045] The following illustrative examples are intended to be non-limiting.
Example 1
[0046] A mercaptan-containing whole straight-run naphtha (boiling range of 30.degree. F.-330.degree. F.; API gravity of 62.5) mixed with a selected air rate was flowed continuously through a 10 mL reactor bed loaded with a spent copper-containing oxidation catalyst that was previously used as an adsorbent in a sulfur guard bed for a refinery process. The catalyst contained about 30 wt. % copper and was prepared by co-mulling and impregnating an alumina support with copper, as described in U.S. Pat. No. 4,259,213. The unit was operated at various temperatures and liquid hourly space velocities (LHSV) at a pressure of 65 psig. The results are summarized in Table 1.
TABLE-US-00001 TABLE 1 SRN Treated Naphtha Feed Run 1 Run 2 Run 3 Run 4 Run 5 Sulfur as RSH, 20.66 Not De- 0.07 0.58 0.68 0.25 wppm (ASTM tectable D3227) Process Conditions T, .degree. F. 210 210 230 230 230 LHSV, h.sup.-1 2.0 1.2 2.0 2.0 3.0 O.sub.2/RSH mole 1.5 1.5 1.5 0.75 0.75 ratio
[0047] As shown in Table 1, the treated naphtha products in Runs 1-5 contained less than 1 ppm mercaptans.
Example 2
[0048] A treated naphtha product prepared as described in Example 1 was then distilled into light (200.degree. F.-) and heavy (200.degree. F.+) naphtha cuts. The physical properties and composition of the light and heavy naphtha cuts are summarized in Tables 2.
TABLE-US-00002 TABLE 2 Light Naphtha Heavy Naphtha Fraction Fraction Cu Strip Corrosion.sup.(a) 1a 1a (ASTM D130) Ag Strip Corrosion.sup.(b) 1 1 (ASTM D7667) Unwashed Gum, mg/100 mL 3.4 (ASTM D381) Washed Gum.sup.(c), mg/100 mL 0 (ASTM D381) Hydrocarbon Class Distibution.sup.(d), vol % (Modified ASTM D6729) Aromatics 2.72 13.03 i-Paraffins 37.48 28.43 n-Paraffins 37.10 22.75 Naphthenes 22.56 32.08 Olefins 0.07 0.02 Oxygen 0.00 0.00 Unclassified 0.07 3.69 .sup.(a)A copper strip rating of 1a indicates slight tarnish, almost the same as a freshly polished strip. ASTM D4814 requires that fuels for automotive spark engines have a copper strip corrosion maximum of 1. .sup.(b)A silver strip rating of 1 indicates slight tarnish. ASTM D4814 requires that fuels for automotive spark engines have a silver strip corrosion maximum of 1. .sup.(c)ASTM D4814 requires that fuels for automotive spark engines have a maximum solvent-washed gum content of 5 mg/100 mL. .sup.(d)Hydrocarbon class distribution was analyzed via detailed hydrocarbon analysis using gas chromatography (GC-DHA) by a method derived from the ASTM D6729 method. To improve identifications of GC species, 60 meter high resolution dual columns (one polar column and one non-polar column) were used. Those peaks that were not identified were grouped in the "Unclassified" category.
[0049] The GC-DHA analysis showed that very small amounts of olefins were found in either the light or heavy treated naphtha. This indicates the mercaptan oxidation process did not dehydrogenate hydrocarbons to any substantial extent. Furthermore, the heavy naphtha has a higher relative concentration of aromatics as compared to the light naphtha. Most aromatics are higher boiling than paraffins with the same carbon number. These results indicate that the mercaptan oxidation process does not reveal any product quality concerns nor require a need for additives in order meet US EPA Tier 3 requirements for gasoline.
[0050] The thermal stability of disulfides in the light and heavy naphtha cuts was determined at the naphtha splitter bottom temperature. Disulfides are products from mercaptan oxidation and can convert reversibly back to mercaptans at higher temperature. Each cut was heated to 350.degree. F. in a Parr bomb type reactor for one and four hours. Sulfur speciation studies using gas chromatography sulfur chemiluminescence detection (GC-SCD) indicated no detectable reappearance of any mercaptans in the light and heavy naphtha cuts.
Example 3
[0051] Example 1 was repeated except that the whole straight-run naphtha feed was a 300.degree. F.- cut from a blend of Canadian condensates CFT and CRW. The feed contained 90.4 wppm of C.sub.1-C.sub.9 mercaptans including cyclic and aromatic mercaptans. The mercaptan conversion results for this "tough feed" are summarized in Table 3.
TABLE-US-00003 TABLE 3 Treated Naphtha SRN Feed Run 6 Run 7 Run 8 Sulfur as RSH, wppm 90.4 Not Not Not (ASTM D3227) Detectable Detectable Detectable Process Conditions T, .degree. F. 210 210 210 LHSV, h.sup.-1 1.2 2.0 3.0 O.sub.2/RSH mole ratio 0.75 0.75 0.75
[0052] The results show complete mercaptan conversion of the high mercaptan content feed.
[0053] A treated naphtha product was then distilled into light (200.degree. F.-) and heavy (200.degree. F.+) naphtha cuts. Both cuts were tested for their thermal stability at 302.degree. F. GC-SCD indicated no detectable reappearance of any mercaptans in the light and heavy naphtha cuts.
Example 4
[0054] Example 1 was repeated except that a fresh copper-containing oxidation catalyst was used. The test was conducted on a conventional whole straight-run naphtha under the following conditions:
[0055] Pressure=70 psig
[0056] Temperature=210.degree. F.
[0057] LHSV=1.2 h-1
[0058] O.sub.2/RSH mole ratio=3
[0059] Time=2 months
Complete mercaptan conversion was achieved during the run.
* * * * *
D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.