Substituted Benzamides And Methods Of Use Thereof
ANDREZ; Jean-Christophe ; et al.
U.S. patent application number 16/390957 was filed with the patent office on 2020-04-09 for substituted benzamides and methods of use thereof. This patent application is currently assigned to GENENTECH, INC.. The applicant listed for this patent is GENENTECH, INC. XENON PHARMACEUTICALS INC.. Invention is credited to Jean-Christophe ANDREZ, Paul Robert BICHLER, Chien-An CHEN, Sultan CHOWDHURY, Shannon Marie DECKER, Christoph Martin DEHNHARDT, Thilo FOCKEN, Michael Edward GRIMWOOD, Ivan William HEMEON, Qi JIA, Jun LI, Zhiguo LIU, Daniel F. ORTWINE, Brian Salvatore SAFINA, Tao SHENG, Shaoyi SUN, Daniel SUTHERLIN, Andrew D. WHITE, Michael Scott WILSON, Alla Yurevna ZENOVA, Jiuxiang ZHU.
| Application Number | 20200108054 16/390957 |
| Document ID | / |
| Family ID | 53198372 |
| Filed Date | 2020-04-09 |









View All Diagrams
| United States Patent Application | 20200108054 |
| Kind Code | A1 |
| ANDREZ; Jean-Christophe ; et al. | April 9, 2020 |
SUBSTITUTED BENZAMIDES AND METHODS OF USE THEREOF
Abstract
The invention provides compounds having the general formula I: ##STR00001## and pharmaceutically acceptable salts thereof, wherein the variables R.sup.A, R.sup.AA, subscript n, ring A, X.sup.2, L, subscript m, X.sup.1, R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, and R.sup.N have the meaning as described herein, and compositions containing such compounds and methods for using such compounds and compositions.
| Inventors: | ANDREZ; Jean-Christophe; (Burnaby, CA) ; BICHLER; Paul Robert; (Burnaby, CA) ; CHEN; Chien-An; (Shanghai, CN) ; CHOWDHURY; Sultan; (Burnaby, CA) ; DECKER; Shannon Marie; (Burnaby, CA) ; DEHNHARDT; Christoph Martin; (Burnaby, CA) ; FOCKEN; Thilo; (Burnaby, CA) ; GRIMWOOD; Michael Edward; (Burnaby, CA) ; HEMEON; Ivan William; (Burnaby, CA) ; JIA; Qi; (Burnaby, CA) ; LI; Jun; (South San Francisco, CA) ; LIU; Zhiguo; (Shanghai, CN) ; ORTWINE; Daniel F.; (South San Francisco, CA) ; SAFINA; Brian Salvatore; (South San Francisco, CA) ; SUTHERLIN; Daniel; (South San Francisco, CA) ; SHENG; Tao; (Burnaby, CA) ; SUN; Shaoyi; (Burnaby, CA) ; WHITE; Andrew D.; (Shanghai, CN) ; WILSON; Michael Scott; (Burnaby, CA) ; ZENOVA; Alla Yurevna; (Burnaby, CA) ; ZHU; Jiuxiang; (Shanghai, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | GENENTECH, INC. South San Francisco CA XENON PHARMACEUTICALS INC. Burnaby |
||||||||||
| Family ID: | 53198372 | ||||||||||
| Appl. No.: | 16/390957 | ||||||||||
| Filed: | April 22, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16130703 | Sep 13, 2018 | |||
| 16390957 | ||||
| 15875992 | Jan 19, 2018 | |||
| 16130703 | ||||
| 15620597 | Jun 12, 2017 | |||
| 15875992 | ||||
| 15275131 | Sep 23, 2016 | 9694002 | ||
| 15620597 | ||||
| 14603273 | Jan 22, 2015 | 9546164 | ||
| 15275131 | ||||
| PCT/CN2014/092269 | Nov 26, 2014 | |||
| 14603273 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 19/02 20180101; C07D 211/32 20130101; C07D 417/06 20130101; C07F 7/0812 20130101; C07D 211/96 20130101; C07D 295/155 20130101; A61P 25/24 20180101; C07D 265/30 20130101; C07D 305/08 20130101; C07D 413/12 20130101; A61P 11/00 20180101; A61P 35/00 20180101; C07D 207/12 20130101; C07D 237/08 20130101; C07D 233/68 20130101; A61P 25/00 20180101; A61P 29/00 20180101; C07D 211/60 20130101; A61K 31/4412 20130101; A61P 9/10 20180101; A61P 25/18 20180101; A61P 25/22 20180101; A61K 31/397 20130101; C07D 205/12 20130101; A61P 25/06 20180101; A61K 31/4545 20130101; C07D 403/04 20130101; A61K 31/4015 20130101; C07D 205/04 20130101; C07D 413/06 20130101; C07D 451/02 20130101; A61P 17/02 20180101; C07D 405/12 20130101; C07D 487/08 20130101; A61K 31/337 20130101; C07D 451/06 20130101; C07D 211/42 20130101; C07D 211/54 20130101; C07D 295/26 20130101; A61P 9/00 20180101; A61P 13/10 20180101; A61P 31/18 20180101; C07D 453/02 20130101; C07D 241/04 20130101; C07D 401/04 20130101; C07D 401/12 20130101; A61K 31/415 20130101; A61K 31/445 20130101; A61K 31/495 20130101; A61P 1/02 20180101; A61P 1/04 20180101; A61P 25/04 20180101; C07D 211/22 20130101; C07D 401/06 20130101; C07D 403/12 20130101; A61P 17/00 20180101; C07D 405/04 20130101; A61K 31/4523 20130101; A61P 1/00 20180101; A61P 25/08 20180101 |
| International Class: | A61K 31/445 20060101 A61K031/445; C07D 205/12 20060101 C07D205/12; C07D 417/06 20060101 C07D417/06; C07D 405/12 20060101 C07D405/12; C07D 413/12 20060101 C07D413/12; C07D 413/06 20060101 C07D413/06; C07D 211/22 20060101 C07D211/22; C07D 295/155 20060101 C07D295/155; C07D 451/06 20060101 C07D451/06; C07D 401/04 20060101 C07D401/04; C07D 451/02 20060101 C07D451/02; C07D 405/04 20060101 C07D405/04; C07D 295/26 20060101 C07D295/26; C07F 7/08 20060101 C07F007/08; C07D 211/96 20060101 C07D211/96; A61K 31/4545 20060101 A61K031/4545; A61K 31/4523 20060101 A61K031/4523; C07D 211/60 20060101 C07D211/60; C07D 211/54 20060101 C07D211/54; C07D 211/32 20060101 C07D211/32; C07D 487/08 20060101 C07D487/08; C07D 403/04 20060101 C07D403/04; C07D 453/02 20060101 C07D453/02; C07D 403/12 20060101 C07D403/12; C07D 401/12 20060101 C07D401/12; C07D 401/06 20060101 C07D401/06; C07D 305/08 20060101 C07D305/08; C07D 207/12 20060101 C07D207/12; C07D 241/04 20060101 C07D241/04; C07D 237/08 20060101 C07D237/08; C07D 233/68 20060101 C07D233/68; A61K 31/495 20060101 A61K031/495; A61K 31/4412 20060101 A61K031/4412; A61K 31/415 20060101 A61K031/415; A61K 31/4015 20060101 A61K031/4015; A61K 31/397 20060101 A61K031/397; A61K 31/337 20060101 A61K031/337; C07D 265/30 20060101 C07D265/30; C07D 211/42 20060101 C07D211/42; C07D 205/04 20060101 C07D205/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 27, 2013 | CN | PCT/CN2013/001452 |
| Nov 28, 2013 | CN | PCT/CN2013/088062 |
| Nov 3, 2014 | CN | PCT/CN2014/090171 |
Claims
1. A compound of formula I: ##STR02185## or a pharmaceutically acceptable salt thereof, wherein: R.sup.1 is C.sub.1-8 alkyl, C.sub.2-8 alkenyl, C.sub.1-8 haloalkyl, C.sub.1-8 alkoxy, C.sub.3-8 carbocycle, C-linked C.sub.2-7 heterocycle, or --NR.sup.1AR.sup.1B, wherein R.sup.1A and R.sup.1B are each independently selected from the group consisting of hydrogen, C.sub.1-8 alkyl, C.sub.1-8 alkoxy, and wherein R.sup.1A and R.sup.1B are optionally combined to form a 3 to 8 membered heterocyclic ring optionally comprising 1 additional heteroatom selected from N, O and S; and wherein R.sup.1 is optionally substituted with from 1 to 5 substituents selected from the group consisting of C.sub.1-4 alkyl, C.sub.1-4 haloalkyl, F, Cl, Br, I, --OH, --CN, --NO.sub.2, --NR.sup.R1aR.sup.R1b, --OR.sup.R1a, --SR.sup.R1a, --Si(R.sup.R1a).sub.3 and C.sub.3-6 carbocycle; wherein R.sup.R1a and R.sup.R1b are independently selected from the group consisting of hydrogen, C.sub.1-8 alkyl, C.sub.1-8 haloalkyl; R.sup.N is hydrogen, C.sub.1-4alkyl or C.sub.1-4haloalkyl; R.sup.2 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8 alkyl, C.sub.1-8 haloalkyl and C.sub.1-8 alkoxy; R.sup.3 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8 alkyl, C.sub.1-8 haloalkyl and C.sub.1-8 alkoxy; R.sup.4 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8 alkyl, C .sub.1-8 haloalkyl and C.sub.1-8 alkoxy; R.sup.5 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8 alkyl, C.sub.1-8 haloalkyl, C.sub.1-8 alkoxy, C.sub.3-8 cycloalkyl and C.sub.2-7 heterocycle, wherein said C.sub.3-8 cycloalkyl and C.sub.2-7 heterocycle is optionally substituted with 1-3 substituents selected from F, Cl, Br and I; L is a linker selected from the group consisting of C.sub.1-4 alkylene, C.sub.2-4 alkenylene and C.sub.2-4 alkynylene, wherein L is optionally substituted with from 1 to 3 substituents selected from the group consisting of .dbd.O, C.sub.1-4 alkyl, halo, and C.sub.1-4 haloalkyl; the subscript m represents the integer 0 or 1; X.sup.1 and X.sup.2 are each independently selected from the group consisting of absent, --O--, --S(O)--, --S(O).sub.2-- and --N(R.sup.X)-- wherein R.sup.X is H, C.sub.1-8 alkyl, C.sub.1-8 alkanoyl, or --S(O).sub.2(C.sub.1-8 alkyl), and wherein if the subscript m is 0 then one of X.sup.1 or X.sup.2 is absent; the subscript in is an integer from 0 to 5; the ring A is a C.sub.2-11 heterocycle comprising a nitrogen atom and further optionally comprising 1-2 heteroatoms selected from N, O and S; each R.sup.AA is independently selected from the group consisting of C.sub.1-6 alkyl, C.sub.1- 6 haloalkyl, C.sub.1-6 heteroalkyl, CN, F, Cl, Br and I; and R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10 aryl-(X.sup.RA)--, C.sub.1-20 heteroaryl-(X.sup.RA)--, C.sub.3-12 carbocycle-(X.sup.RA)--, --R.sup.A2, --S(O).sub.2--R.sup.A2, and C.sub.2-11 heterocycle-(X.sup.RA)--, wherein said C.sub.6-10 aryl, C.sub.5-9 heteroaryl, C.sub.3-12 carbocycle and C.sub.2-11 heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4 alkyl, C.sub.1-4 haloalkyl, C.sub.1-4 alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4 alkylamino, C.sub.1-4 dialkylamino, C.sub.1-4 alkanoyl, C.sub.1-4 alkyl-OC(.dbd.O)--, C.sub.1-4 alkyl-S(O).sub.2--, C.sub.3-6carbocycle, and phenyl that is optionally substituted with one or more substituents selected from fluoro, chloro, and bromo; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8 alkyl, C.sub.2-8 alkenyl, C.sub.1-8 haloalkyl, C.sub.3-8 cycloalkyl, phenyl and benzyl; R.sup.A2 is selected from the group consisting of C.sub.1-8 alkyl that is optionally substituted with one or more substituents selected from oxo (.dbd.O), fluoro, amino, C.sub.1-4 alkylamino and C.sub.1-4 dialkylamino; X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4 alkyl)-, --S(O)--, --S(O).sub.2--, --C(.dbd.O)--, C.sub.1-4 alkylene, C.sub.1-4 heteroalkylene, C.sub.2-4 alkenylene and C.sub.2-4 alkynylene; X.sup.RB is selected from the group consisting of absent, C.sub.1-4 alkylene, C.sub.1-4 heteroalkylene, C.sub.2-4 alkenylene and C.sub.2-4 alkynylene; wherein any C.sub.1-4 alkylene, C.sub.1-4 heteroalkylene, C.sub.2-4 alkenylene and C.sub.2-4 alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4 alkyl, C.sub.1-4 haloalkyl, C.sub.1-4 heteroalkyl, oxo (.dbd.O), hydroxy, and phenyl that is optionally substituted with 1 to 5 substitutents selected from F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4 alkyl, C.sub.1-4 haloalkyl, C.sub.1-4 alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4 alkylamino and C.sub.1-4 dialkylamino; or wherein X.sup.RA or X.sup.RB is optionally substituted with 2 substituents that combine to form a 3 to 5 membered carbocycle or a 3-5 membered heterocycle; provided the compound of formula I is not: ##STR02186## ##STR02187## ##STR02188## ##STR02189##
2-64. (canceled)
Description
[0001] The present invention relates to organic compounds useful for therapy and/or prophylaxis in a mammal, and in particular to inhibitors of sodium channel (e.g., NAV1.7) that are useful for treating sodium channel-mediated diseases or conditions, such as pain, as well as other diseases and conditions associated with the mediation of sodium channels.
[0002] Voltage-gated sodium channels, transmembrane proteins that initiate action potentials in nerve, muscle and other electrically excitable cells, are a necessary component of normal sensation, emotions, thoughts and movements (Catterall, W. A., Nature (2001), Vol. 409, pp. 988-990). These channels consist of a highly processed alpha subunit that is associated with auxiliary beta subunits. The pore-forming alpha subunit is sufficient for channel function, but the kinetics and voltage dependence of channel gating are in part modified by the beta subunits (Goldin et al., Neuron (2000), Vol. 28, pp. 365-368).
Electrophysiological recording, biochemical purification, and molecular cloning have identified ten different sodium channel alpha subunits and four beta subunits (Yu, F. H., et al., Sci. STKE (2004), 253; and Yu, F. H., et al., Neurosci. (2003), 20:7577-85).
[0003] The hallmarks of sodium channels include rapid activation and inactivation when the voltage across the plasma membrane of an excitable cell is depolarized (voltage-dependent gating), and efficient and selective conduction of sodium ions through conducting pores intrinsic to the structure of the protein (Sato, C., et al., Nature (2001), 409:1047-1051). At negative or hyperpolarized membrane potentials, sodium channels are closed. Following membrane depolarization, sodium channels open rapidly and then inactivate. Channels only conduct currents in the open state and, once inactivated, have to return to the resting state, favoured by membrane hyperpolarization, before they can reopen. Different sodium channel subtypes vary in the voltage range over which they activate and inactivate as well as their activation and inactivation kinetics.
[0004] The sodium channel family of proteins has been extensively studied and shown to be involved in a number of vital body functions. Research in this area has identified variants of the alpha subunits that result in major changes in channel function and activities, which can ultimately lead to major pathophysiological conditions. The members of this family of proteins are denoted NaV1.x, where x=1 to 9. NaV1.1 and NaV1.2 are highly expressed in the brain (Raymond, C. K., et al., J. Biol. Chem. (2004), 279(44):46234-41) and are vital to normal brain function. Some loss of function mutations in NaV1.1 in humans result in epilepsy, apparently because many of these channels are expressed in inhibitory neurons (Yu, F. H., et al., Nat Neurosci (2006), 9 (9), 1142-9). Thus, block of NaV1.1 in the CNS may be counter-productive because it can produce hyperexcitability. However, NaV1.1 is also expressed in the peripheral nervous system and block may afford analgesic activity.
[0005] NaV1.3 is expressed primarily in the fetal central nervous system. It is expressed at very low levels or not at all in the peripheral nervous system, but expression is upregulated in the dorsal horn sensory neurons of rats after nervous system injury (Hains, B. D., et al., J. Neurosci. (2003), 23(26):8881-92). Thus, it is an inducible target for treatment of pain following nerve injury.
[0006] NaV1.4 is expressed primarily in skeletal muscle (Raymond, C. K., et al., op. cit.). Mutations in this gene have been shown to have profound effects on muscle function including paralysis, (Tamaoka A., Intern. Med. (2003), (9):769-70).
[0007] NaV1.5, is expressed mainly in cardiac myocytes (Raymond, C. K., et al., op. cit.), including atria, ventricles, the sino-atrial node, atrio-ventricular node and cardiac Purkinje fibers. The rapid upstroke of the cardiac action potential and the rapid impulse conduction through cardiac tissue is due to the opening of NaV1.5. Abnormalities in the function of NaV1.5 can result in the genesis of a variety of cardiac arrhythmias. Mutations in human NaV1.5 result in multiple arrhythmic syndromes, including, for example, long QT3 (LQT3), Brugada syndrome (BS), an inherited cardiac conduction defect, sudden unexpected nocturnal death syndrome (SUNDS) and sudden infant death syndrome (SIDS) (Liu, H., et al., Am. J. Pharmacogenomics (2003), 3(3):173-9). Sodium channel blocker therapy has been used extensively in treating cardiac arrhythmias.
[0008] NaV1.6 is a widely distributed voltage-gated sodium channel found throughout the central and peripheral nervous systems. It is expressed at high density in the nodes of Ranvier of myelinated neurons (Caldwell, J. H., et al., Proc. Natl. Acad. Sci. USA (2000), 97(10): 5616-20).
[0009] NaV1.7 is a tetrodotoxin-sensitive voltage-gated sodium channel encoded by the gene SCN9A. Human NaV1.7 was first cloned from neuroendocrine cells (Klugbauer, N., et al., 1995 EMBO J., 14 (6): 1084-90.) and rat NaV1.7 was cloned from a pheochromocytoma PC12 cell line (Toledo-Aral, J. J., et al., Proc. Natl. Acad. Sci. USA (1997), 94:1527-1532) and from rat dorsal root ganglia (Sangameswaran, L., et al., (1997), J. Biol. Chem., 272 (23): 14805-9). NaV1.7 is expressed primarily in the peripheral nervous system, especially nocieptors and olfactory neurons and sympathetic neurons. The inhibition, or blocking, of NaV1.7 has been shown to result in analgesic activity. Knockout of NaV1.7 expression in a subset of sensory neurons that are predominantly nociceptive results in resistance to inflammatory pain (Nassar, et al., op. cit.). Likewise, loss of function mutations in humans results in congenital indifference to pain (CIP), in which the individuals are resistant to both inflammatory and neuropathic pain (Cox, J. J., et al., Nature (2006); 444:894-898; Goldberg, Y. P., et al., Clin. Genet. (2007); 71:311-319). Conversely, gain of function mutations in NaV1.7 have been established in two human heritable pain conditions, primary erythromelalgia and familial rectal pain, (Yang, Y., et al., J. Med. Genet. (2004), 41 (3):171-4). In addition, a single nucleotide polymorphism (R1150W) that has very subtle effects on the time- and voltage-dependence of channel gating has large effects on pain perception (Estacion, M., et al., 2009. Ann Neurol 66: 862-6; Reimann, F., et al., Proc Natl Acad Sci USA (2010), 107: 5148-53). About 10% of the patients with a variety of pain conditions have the allele conferring greater sensitivity to pain and thus might be more likely to respond to block of NaV1.7. Because NaV1.7 is expressed in both sensory and sympathetic neurons, one might expect that enhanced pain perception would be accompanied by cardiovascular abnormalities such as hypertension, but no correlation has been reported. Thus, both the CIP mutations and SNP analysis suggest that human pain responses are more sensitive to changes in NaV1.7 currents than are perturbations of autonomic function.
[0010] NaV1.8 is expressed primarily in sensory ganglia of the peripheral nervous system, such as the dorsal root ganglia (Raymond, C. K., et al., op. cit.). There are no identified human mutations for NaV1.8 that produce altered pain responses. NaV1.8 differs from most neuronal NaV's in that it is insensitive to block by tetrodotoxin. Thus, one can isolate the current carried by this channel with tetrodotoxin. These studies have shown that a substantial portion of total sodium current is NaV1.8 in some dorsal root ganglion neurons (Blair, N. T., et al., J Neurosci (2002), 22: 10277-90). Knock-down of NaV1.8 in rats has been achieved by using antisense DNA or small interfering RNAs and virtually complete reversal of neuropathic pain was achieved in the spinal nerve ligation and chronic constriction injury models (Dong, X. W., et al., Neuroscience (2007), 146: 812-21; Lai J., et al. Pain (2002), 95: 143-52). Thus, NaV1.8 is considered a promising target for analgesic agents based upon the limited tissue distribution of this NaV isoform and the analgesic activity produced by knock-down of channel expression.
[0011] NaV1.9 is also a tetrodotoxin insensitive, sodium channel expressed primarily in dorsal root ganglia neurons (Dib-Hajj, S. D., et al. (see Dib-Hajj, S. D., et al., Proc. Natl. Acad. Sci. USA (1998), 95(15):8963-8). It is also expressed in enteric neurons, especially the myenteric plexus (Rugiero, F., et al., J Neurosci (2003), 23: 2715-25). The limited tissue distribution of this NaV isoform suggests that it may be a useful target for analgesic agents (Lai, J., et al., op. cit.; Wood, J. N., et al., op. cit.; Chung, J. M., et al., op. cit.). Knock-out of NaV1.9 results in resistance to some forms of inflammatory pain (Amaya, F., et al., J Neurosci (2006), 26: 12852-60; Priest, B. T., et al., Proc Natl Acad Sci USA (2005), 102: 9382-7).
[0012] This closely related family of proteins has long been recognized as targets for therapeutic intervention. Sodium channels are targeted by a diverse array of pharmacological agents. These include neurotoxins, antiarrhythmics, anticonvulsants and local anesthetics (England, S., et al., Future Med Chem (2010), 2: 775-90; Termin, A., et al., Annual Reports in Medicinal Chemistry (2008), 43: 43-60). All of the current pharmacological agents that act on sodium channels have receptor sites on the alpha subunits. At least six distinct receptor sites for neurotoxins and one receptor site for local anesthetics and related drugs have been identified (Cestele, S., et al., Biochimie (2000), Vol. 82, pp. 883-892).
[0013] The small molecule sodium channel blockers or the local anesthetics and related antiepileptic and antiarrhythmic drugs interact with overlapping receptor sites located in the inner cavity of the pore of the sodium channel (Catterall, W. A., Neuron (2000), 26:13-25). Amino acid residues in the S6 segments from at least three of the four domains contribute to this complex drug receptor site, with the IVS6 segment playing the dominant role. These regions are highly conserved and as such most sodium channel blockers known to date interact with similar potency with all channel subtypes. Nevertheless, it has been possible to produce sodium channel blockers with therapeutic selectivity and a sufficient therapeutic window for the treatment of epilepsy (e.g., lamotrignine, phenytoin and carbamazepine) and certain cardiac arrhythmias (e.g., lignocaine, tocainide and mexiletine). However, the potency and therapeutic index of these blockers is not optimal and have limited the usefulness of these compounds in a variety of therapeutic areas where a sodium channel blocker would be ideally suited.
[0014] Sodium channel blockers have been shown to be useful in the treatment of pain, including acute, chronic, inflammatory and/or neuropathic pain (see, e.g., Wood, J. N., et al., J. Neurobiol. (2004), 61(1), 55-71. Preclinical evidence demonstrates that sodium channel blockers can suppress neuronal firing in peripheral and central sensory neurons, and it is via this mechanism that they are considered to be useful for relieving pain. In some instances, abnormal or ectopic firing can original from injured or otherwise sensitized neurons. For example, it has been shown that sodium channels can accumulate in peripheral nerves at sites of axonal injury and may function as generators of ectopic firing (Devor et al., J. Neurosci. (1993), 132: 1976). Changes in sodium channel expression and excitability have also been shown in animal models of inflammatory pain where treatment with proinflammatory materials (CFA, Carrageenan) promoted pain-related behaviors and correlated with increased expression of sodium channel subunits (Gould et al., Brain Res., (1999), 824(2): 296-99; Black et al., Pain (2004), 108(3): 237-47). Alterations in either the level of expression or distribution of sodium channels, therefore, may have a major influence on neuronal excitability and pain-related behaviors.
[0015] Controlled infusions of lidocaine, a known sodium channel blocker, indicate that the drug is efficacious against neuropathic pain, but has a narrow therapeutic index. Likewise, the orally available local anesthetic, mexiletine, has dose-limiting side effects (Wallace, M. S., et al., Reg. Anesth. Pain Med. (2000), 25: 459-67). A major focus of drug discovery targeting voltage-gated sodium channels has been on strategies for improving the therapeutic index. One of the leading strategies is to identify selective sodium channel blockers designed to preferentially block NaV1.7, NaV1.8, NaV1.9 and/or NaV1.3. These are the sodium channel isoforms preferentially expressed in sensory neurons and unlikely to be involved in generating any dose-limiting side effects. For example, there is concern that blocking of NaV1.5 would be arrhythmogenic, so that selectivity of a sodium channel blocker against NaV1.5 is viewed as highly desirable. Furthermore, nearly 700 mutations of the SCN1A gene that codes for NaV1.1 have been identified in patients with Severe Myoclonic Epilepsy of Infancy (SMEI), making this the most commonly mutated gene in human epilepsy. Half of these mutations result in protein truncation (Meisler, M. H., et al., The Journal of Physiology (2010), 588: 1841-8). Thus, selectivity of a sodium channel blocker against NaV1.1 is also desirable.
[0016] In addition to the strategies of identifying selective sodium channel blockers, there is the continuing strategy of identifying therapeutic agents for the treatment of neuropathic pain. There has been some degree of success in treating neuropathic pain symptoms by using medications originally approved as anticonvulsants, such as gabapentin, and more recently pregabalin. However, pharmacotherapy for neuropathic pain has generally had limited success for a variety of reasons: sedation, especially by drugs first developed as anticonvulsants or anti-depressants, addiction or tachyphylaxis, especially by opiates, or lack of efficacy, especially by NSAIDs and anti-inflammatory agents. Consequently, there is still a considerable need to explore novel treatment modalities for neuropathic pain, which includes, but is not limited to, post-herpetic neuralgia, trigeminal neuralgia, diabetic neuropathy, chronic lower back pain, phantom limb pain, and pain resulting from cancer and chemotherapy, chronic pelvic pain, complex regional pain syndrome and related neuralgias.
[0017] There are a limited number of effective sodium channel blockers for the treatment of pain with a minimum of adverse side effects which are currently in the clinic. There is also an unmet medical need to treat neuropathic pain and other sodium channel associated pathological states effectively and without adverse side effects due to the blocking of sodium channels not involved in nociception. The present invention provides methods to meet these critical needs.
SUMMARY OF THE INVENTION
[0018] In one aspect the present invention provides for novel compounds. In a first embodiment of such compounds (Embodiment 1; abbreviated as "E1") the invention provides for a compound of formula I:
##STR00002##
[0019] or a pharmaceutically acceptable salt thereof, wherein:
[0020] R.sup.1 is C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8carbocycle, C-linked C.sub.2-7heterocycle, or --NR.sup.1AR.sup.1B, wherein R.sup.1A and R.sup.1B are each independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8alkoxy, and wherein R.sup.1A and R.sup.1B are optionally combined to form a 3 to 8 membered heterocyclic ring optionally comprising 1 additional heteroatom selected from N, O and S; and wherein R.sup.1 is optionally substituted with from 1 to 5 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, F, Cl, Br, I, --OH, --CN, --NO.sub.2, --NR.sup.R1aR.sup.R1b, --OR.sup.R1a, --SR.sup.R1a, --Si(R.sup.R1a).sub.3 and C.sub.3-6carbocycle; wherein R.sup.R1a and R.sup.R1b are independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8haloalkyl;
[0021] R.sup.N is hydrogen, C.sub.1-4alkyl or C.sub.1-4haloalkyl;
[0022] R.sup.2 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0023] R.sup.3 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0024] R.sup.4 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0025] R.sup.5 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8cycloalkyl and C.sub.2-7heterocycle, wherein said C.sub.3-8cycloalkyl and C.sub.2-7heterocycle is optionally substituted with 1-3 substituents selected from F, Cl, Br and I;
[0026] L is a linker selected from the group consisting of C.sub.1-4alkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene, wherein L is optionally substituted with from 1 to 3 substituents selected from the group consisting of .dbd.O, C.sub.1-4alkyl, halo, and C.sub.1-4haloalkyl;
[0027] the subscript m represents the integer 0 or 1;
[0028] X.sup.1 and X.sup.2 are each independently selected from the group consisting of absent, --O--, --S(O)--, --S(O).sub.2-- and --N(R.sup.X)-- wherein R.sup.X is H, C.sub.1-8alkyl, C.sub.1-8alkanoyl, or --S(O).sub.2(C.sub.1-8alkyl), and wherein if the subscript m is 0 then one of X.sup.1 or X.sup.2 is absent;
[0029] the subscript n is an integer from 0 to 5;
[0030] the ring A is a C.sub.2-11heterocycle comprising a nitrogen atom and further optionally comprising 1-2 heteroatoms selected from N, O and S;
[0031] each R.sup.AA is independently selected from the group consisting of C.sub.1-6alkyl, C.sub.1-6haloalkyl, C.sub.1-6heteroalkyl, CN, F, Cl, Br and I; and
[0032] R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10aryl-(X.sup.RA)--, C.sub.1-20heteroaryl-(X.sup.RA)--, C.sub.3-12carbocycle-(X.sup.RA)--, --R.sup.A2, --S(O).sub.2--R.sup.A2 and C.sub.2-11heterocycle-(X.sup.RA)--, wherein said C.sub.6-10aryl, C.sub.5-9heteroaryl, C.sub.3-12carbocycle and C.sub.2-11heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2--, C.sub.3-6carbocycle, and phenyl that is optionally substituted with one or more substituents selected from fluoro, chloro, and bromo; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.2-8haloalkyl, C.sub.3-8cycloalkyl, phenyl and benzyl; R.sup.A2 is selected from the group consisting of C.sub.1-8alkyl that is optionally substituted with one or more substituents selected from oxo (.dbd.O), fluoro, amino, C.sub.1-4alkylamino and C.sub.1-4dialkylamino; X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4alkyl)-, --S(O)--, --S(O).sub.2--, --C(.dbd.O)--, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; X.sup.RB is selected from the group consisting of absent, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), hydroxy, and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-3alkylamino and C.sub.1-4dialkylamino; or wherein X.sup.RA or X.sup.RB is optionally substituted with 2 substituents that combine to form a 3 to 5 membered carbocycle or a 3-5 membered heterocycle;
[0033] provided the compound of formula I is not:
##STR00003## ##STR00004## ##STR00005## ##STR00006##
E2 The compound or salt of E1 wherein:
[0034] R.sup.1 is C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8carbocycle, C-linked C.sub.2-7heterocycle, or --NR.sup.1AR.sup.1B, wherein R.sup.1A and R.sup.1B are each independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8alkoxy, and wherein R.sup.1A and R.sup.1B are optionally combined to form a 3 to 8 membered heterocyclic ring optionally comprising 1 additional heteroatom selected from N, O and S; and wherein R.sup.1 is optionally substituted with from 1 to 5 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, F, Cl, Br, I, --OH, --CN, --NO.sub.2, --NR.sup.R1aR.sup.R1b, --OR.sup.R1a, --SR.sup.R1a, --Si(R.sup.R1a).sub.3 and C.sub.3-6carbocycle; wherein R.sup.R1a and R.sup.R1b are independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8haloalkyl;
[0035] R.sup.N is hydrogen, C.sub.1-4alkyl or C.sub.1-4haloalkyl;
[0036] R.sup.2 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0037] R.sup.3 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0038] R.sup.4 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0039] R.sup.5 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8cycloalkyl and C.sub.2-7heterocycle, wherein said C.sub.3-8cycloalkyl and C.sub.2-7heterocycle is optionally substituted with 1-3 substituents selected from F, Cl, Br and I;
[0040] L is a linker selected from the group consisting of C.sub.1-4alkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene, wherein L is optionally substituted with from 1 to 3 substituents selected from the group consisting of .dbd.O, C.sub.1-4alkyl, halo, and C.sub.1-4haloalkyl;
[0041] the subscript m represents the integer 0 or 1;
[0042] X.sup.1 and X.sup.2 are each independently selected from the group consisting of absent, --O--, --S(O)--, --S(O).sub.213 and --N(R.sup.X)-- wherein R.sup.X is H, C.sub.1-8alkyl, C.sub.1-8alkanoyl, or --S(O).sub.2(C.sub.1-8alkyl), and wherein if the subscript m is 0 then one of X.sup.1 or X.sup.2 is absent:
[0043] the subscript n is an integer from 0 to 5;
[0044] the ring A is a C.sub.2-11heterocycle comprising a nitrogen atom and further optionally comprising 1-2 heteroatoms selected from N, O and S;
[0045] each R.sup.AA is independently selected from the group consisting of C.sub.1-6alkyl, C.sub.1-6haloalkyl, C.sub.1-6heteroalkyl, F, Cl, Br and I; and
[0046] R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10aryl-(X.sup.RA)--, C.sub.5-9heteroaryl-(X.sup.RA)--, C.sub.3-12carbocycle-(X.sup.RA)--, --R.sup.A2, --S(O).sub.2R.sup.A2, and C.sub.2-11heterocycle-(X.sup.RA)--, wherein said C.sub.6-10aryl, C.sub.5-9heteroaryl, C.sub.3-12carbocycle and C.sub.2-11heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2, C.sub.3-6carbocycle, and phenyl that is optionally substituted with one or more substituents selected from fluoro, chloro, and bromo; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.3-8cycloalkyl, phenyl and benzyl; R.sup.A2 is selected from the group consisting of C.sub.1-8alkyl that is optionally substituted with one or more substituents selected from oxo (.dbd.O), fluoro, amino, C.sub.1-4alkylamino and C.sub.1-4dialkylamino; X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(C.sub.1-8alkyl)-, --S(O)--, --S(O).sub.2--, --C(.dbd.O)--, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene: X.sup.RB is selected from the group consisting of absent, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.2-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino; or wherein X.sup.RA or X.sup.RB is optionally substituted with 2 substituents that combine to form a 3 to 5 membered carbocycle or a 3-5 membered heterocycle;
E3 The compound or salt of E1 or E2 wherein:
[0047] R.sup.1 is C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8carbocycle, C-linked C.sub.2-7heterocycle, or --NR.sup.1AR.sup.1B, wherein R.sup.1A and R.sup.1B are each independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8alkoxy, and wherein R.sup.1A and R.sup.1B are optionally combined to form a 3 to 8 membered heterocyclic ring optionally comprising 1 additional heteroatom selected from N, O and S; and wherein R.sup.1 is optionally substituted with from 1 to 5 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, F, Cl, Br, I, --OH, --CN, --NO.sub.2, --NR.sup.R1aR.sup.R1b, --OR.sup.R1a, --SR.sup.R1a, --Si(R.sup.R1a).sub.2 and C.sub.3-6carbocycle; wherein R.sup.R1a and R.sup.R1b are independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8haloalkyl;
[0048] R.sup.N is hydrogen, C.sub.1-4alkyl or C.sub.1-4haloalkyl;
[0049] R.sup.2 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0050] R.sup.3 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0051] R.sup.4 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl and C.sub.1-8alkoxy;
[0052] R.sup.5 is selected from the group consisting of H, F, Cl, Br, I, --CN, C.sub.1-8alkyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8cycloalkyl and C.sub.2-7heterocycle, wherein said C.sub.3-8cycloalkyl and C.sub.2-7heterocycle is optionally substituted with 1-3 substituents selected from F, Cl, Br and I;
[0053] L is a linker selected from the group consisting of C.sub.1-4alkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene, wherein L is optionally substituted with from 1 to 3 substituents selected from the group consisting of .dbd.O, C.sub.1-4alkyl, halo, and C.sub.1-4haloalkyl;
[0054] the subscript m represents the integer 0 or 1;
[0055] X.sup.1 and X.sup.2 are each independently selected from the group consisting of absent, --O--, --S(O)--, --S(O).sub.2-- and --N(R.sup.X)-- wherein R.sup.X is H, C.sub.1-8alkyl, C.sub.1-8alkanoyl, or --S(O).sub.2(C.sub.1-8alkyl), and wherein if the subscript m is 0 then one of X.sup.1 or X.sup.2 is absent;
[0056] the subscript n is an integer from 0 to 5;
[0057] the ring A is a C.sub.2-11heterocycle comprising a nitrogen atom and further optionally comprising 1-2 heteroatoms selected from N, O and S;
[0058] each R.sup.AA is independently selected from the group consisting of C.sub.1-6alkyl, C.sub.1-6haloalkyl, C.sub.1-6heteroalkyl, F, Cl, Br and I; and
[0059] R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10aryl-(X.sup.RA)--, C.sub.5-9heteroaryl-(X.sup.RA)--, C.sub.3-12carbocycle-(X.sup.RA)--, and C.sub.2-11heterocycle-(X.sup.RA)--, wherein said C.sub.6-10aryl, C.sub.5-9heteroaryl, C.sub.3-12carbocycle and C.sub.2-11heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2--, and C.sub.3-6carbocycle; R.sup.A1 is is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.3-8cycloalkyl, phenyl and benzyl; R.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4alkyl)-, --S(O).sub.2--, --C(.dbd.O)--, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; X.sup.RB is selected from the group consisting of absent, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino; or wherein X.sup.RA or X.sup.RB is optionally substituted with 2 substituents that combine to form a 3 to 5 membered carbocycle or a 3-5 membered heterocycle.
E4 The compound of E1, E2, or E3 wherein the compound has the formula Ia:
##STR00007##
E5 The compound of E1, E2, or E3, wherein the compound has the formula Ib:
##STR00008##
E6 The compound of E1, E2, or E3, wherein the compound has the formula Ic:
##STR00009##
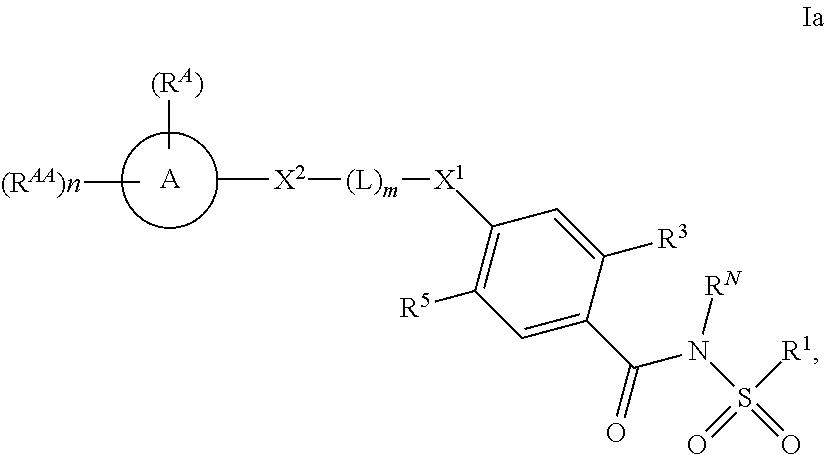
E7 The compound of E1, E2, E3, E4, E5, or E6 wherein R.sup.1 is selected from the group consisting of C.sub.1-8alkyl, C.sub.1-8haloalkyl, C.sub.3-8carbocycle, C.sub.2-7heterocycle, and --NR.sup.1AR.sup.1B, wherein R.sup.1A and R.sup.1B are each independently selected from the group consisting of C.sub.1-8alkyl and C.sub.1-8alkoxy, and wherein and R.sup.1A and R.sup.1B are optionally combined to form a 3 to 6 membered heterocyclic ring; and wherein R.sup.1 is optionally substituted with from 1 to 5 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, F, Cl, Br, I, --OH, --OR.sup.R1a, --SR.sup.R1a, --Si(R.sup.R1a).sub.3 and C.sub.3-8carbocycle; wherein R.sup.R1a and R.sup.R1b are independently selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.1-8haloalkyl E8 The compound of E1, E2, E3, E4, E5, or E6 wherein R.sup.1 is methyl, cyclopropyl, cyclopropylmethyl, 1-azetidinyl, 1-methylcycloprop-1-yl, difluoromethyl, N-methylamino, ethyl, 2-methoxyeth-1-yl, 2-trimethylsilyleth-1-yl, propyl, 1,1,1 -trifluoroprop-3-yl, butyl, morpholino, pyrrolidino, or 3-fluoroazetidin-1-yl. E9 The compound of E1, E2, E3, E4, E5, or E6 wherein R.sup.1 is methyl, cyclopropyl, 1-azetidinyl or 2-methoxyethyl. E10 The compound of E1, E2, E3, E7, E8, or E9 wherein R.sup.2 is H. E11 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, or E10 wherein R.sup.3 is F, Cl, or Br. E12 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, or E10 wherein R.sup.3 is F. E13 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, or E12 wherein R.sup.4 is H. E14 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, or E13 wherein R.sup.5 is C.sub.3-5cycloalkyl. E15 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, or E13 wherein R.sup.5 is cyclopropyl. E16 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein X.sup.1 is --O-- or --N(H)--; X.sup.2 is absent; the subscript m is 1; and --(L)-- is an optionally substituted group selected from the group consisting of C.sub.1-4alkylene, C.sub.2-4alkenylene or C.sub.2-4alkynylene. E17 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein X.sup.1 is --O-- or --N(H)--; X.sup.2 is absent; the subscript m is 1; and --(L)-- is selected from the group consisting of --CH.sub.2--, --C(.dbd.O)--, --C(H)(CH.sub.3)--, --CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--, --C(H)(CH.sub.3)--C(H.sub.2)--, --CH.sub.2CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--. E18 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein X.sup.1 is --O--; the subscript m is 1 and --(L)-- is --CH.sub.2-- or --CH.sub.2--CH.sub.2--. E19 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein X.sup.1 is absent; X.sup.2 is --O-- or --N(H)--; the subscript m is 1; and --(L)-- is selected from the group consisting of --C(H).sub.2--, --C(.dbd.O)--, --C(H)(CH.sub.3)--, --CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--, --C(H)(CH.sub.3)--C(H.sub.2)--, --CH.sub.2CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--. E20 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein X.sup.1 is X.sup.2 is absent; the subscript m is 1; and --(L)-- is selected from the group consisting of --C(H).sub.2--, --C(.dbd.O)--, --C(H)(CH.sub.3)--, --CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--, --C(H)(CH.sub.3)--C(H.sub.2)--, --CH.sub.2CH.sub.2CH.sub.2--, --CH.sub.2--C(H)(CH.sub.3)--CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--. E21 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, or E15 wherein m is 0; X.sup.1 is selected from --O--, and --N(H)--; and X.sup.2 is absent. E22 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, or E21 wherein A is optionally substituted and is selected from azetidine, pyrrolidine, piperidine, morpholine, homopiperazine, and piperazine. E23 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, or E21 wherein:
##STR00010##
is selected from the group consisting of:
##STR00011##
E24 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, or E21 wherein:
##STR00012##
is selected from the group consisting of:
##STR00013##
E25 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, or E24 wherein R.sup.AA is selected from the group consisting of methyl, trifluoromethyl, ethyl, CN, F, Cl, Br, and I. E26 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, or E24 wherein R.sup.AA is selected from the group consisting of methyl, trifluoromethyl, ethyl, F, Cl, Br, and I. E27 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of phenyl-(X.sup.RA)--, wherein said phenyl is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)-- and C.sub.3-6carbocycle; and wherein X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4alkyl)-, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; and wherein X.sup.RA is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino. E28 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is phenyl-(X.sup.RA)--, wherein said phenyl is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, C.sub.1-4alkyl, --CN, C.sub.3-6carbocycle and C.sub.1-4haloalkyl; wherein X.sup.RA is selected from the group consisting of absent and C.sub.1-4alkylene; and wherein X.sup.RA is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, C.sub.1-4alkyl, and C.sub.1-4haloalkyl. E29 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is --(X.sup.RB).sub.0-1OR.sup.A1; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.3-8cycloalkyl, phenyl and benzyl; and X.sup.RB is selected from the group consisting of absent and C.sub.1-4alkylene that is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino. E30 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of
##STR00014## ##STR00015##
E31 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of
##STR00016##
E32 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of phenyl, phenylmethyl, pyrazolyl, pyrazolylmethyl, cyclobutyl, cyclohexylmethyl, cyclopentyl, cyclopentylmethyl, cyclobutyl, cyclobutylmethyl, pyrimidinyl, pyrimidinylmethyl, pyrazinyl, pyrazinylmethyl, pyridazinyl, pyridazinylmethyl, indolinyl, indolinylmethyl, isoindolinyl, and isoindolinylmethyl, and wherein R.sup.A is optionally substituted with from 1 to 5 substitutents selected from F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4dialkylamino, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2--, C.sub.3-6carbocycle, and phenyl that is optionally substituted with one or more substituents selected from fluoro, chloro, and bromo. E33 The compound of claim E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10aryl-(X.sup.RA)--, C.sub.1-20heteroaryl-(X.sup.RA)--, C.sub.3-12carbocycle-(X.sup.RA)--, and C.sub.2-11heterocycle-(X.sup.RA)--, wherein said of R.sup.A C.sub.6-10aryl, C.sub.5-9heteroaryl, C.sub.3-12carbocycle and C.sub.2-11heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.3-6carbocycle; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.3-8cycloalkyl, phenyl, and benzyl; X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4alkyl)-, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; and X.sup.RB selected from the group consisting of absent, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, and C.sub.1-4heteroalkyl. E34 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of --(X.sup.RB).sub.0-1OR.sup.A1, C.sub.6-10aryl-(X.sup.RA)--, C.sub.5-9heteroaryl-(X.sup.RA)--, C.sub.3-12carbocycle-(X.sup.RA)--, and C.sub.2-11heterocycle-(X.sup.RA)--, wherein said C.sub.6-10aryl, C.sub.5-9heteroaryl, C.sub.3-12carbocycle and C.sub.2-11heterocycle of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)-- and C.sub.3-6carbocycle; R.sup.A1 is selected from the group consisting of hydrogen, C.sub.1-8alkyl, C.sub.2-8alkenyl, C.sub.1-8haloalkyl, C.sub.3-8cycloalkyl, phenyl, and benzyl; X.sup.RA is selected from the group consisting of absent, --O--, --S--, --N(H)--, --N(C.sub.1-4alkyl)-, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; and X.sup.RB selected from the group consisting of absent, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA or X.sup.RB is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, and C.sub.1-4heteroalkyl. E35 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2--, and C.sub.3-6carbocycle; and X.sup.RA is selected from the group consisting of --C(.dbd.O)--, C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene; wherein any C.sub.1-4alkylene, C.sub.1-4heteroalkylene, C.sub.2-4alkenylene and C.sub.2-4alkynylene of X.sup.RA is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino. E36 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26wherein R.sup.A is phenyl --(X.sup.RA)--, wherein said phenyl is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, --CN, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, and C.sub.1-4(halo)alkoxy; and X.sup.RA is C.sub.1-4alkylene that is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino. E37 The compound of E1, E2, E3, E7, E8, E9, E16, E17, E18, E19, E20, E20, E21, E22, E23, E24, E25, E26, E27, E28, E29, E30, E31, E32, E33, E34, E35, or E36 wherein the compound has the formula Id:
##STR00017##
E38 The compound of E37 wherein R.sup.1 is methyl, ethyl, cyclopropyl, or 1-azetidinyl. E39 The compound of E37 or E38 wherein --X.sup.2--(L).sub.m--X.sup.1-- is --O--, --CH.sub.2--, or --CH.sub.2CH.sub.2--O--. E40 The compound of E37, E38, or E39 wherein:
##STR00018##
E41 The compound of E37, E38, or E39 wherein:
##STR00019##
E42 The compound of E37, E38, or E39 wherein A is optionally substituted azetidine, pyrrolidine, piperidine, morpholine, homopiperazine, and piperazine. E43 The compound of E37, E38, or E39 wherein:
##STR00020##
is selected from the group consisting of:
##STR00021##
E44 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E37, E38, or E39 wherein:
##STR00022##
is selected from the group consisting of:
##STR00023##
E45 The compound of E1, E2, E3, E4, E7, E8, E9, E10, E11, E12, E13, E14, E15, E25, E26, E27, E28, E29, E30, E31, E32, E33, E34, E35, E36, E37, or E38 wherein: has the formula:
##STR00024##
E46 The compound of E41, wherein
##STR00025##
E47 The compound of claim E46, wherein R.sup.AA is selected from the group consisting of hydrogen, F, Cl and C.sub.1-4haloalkyl. E48 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, E26, E37, E38, E39, E40, E41, E42, E43, E44, E45, E46, or E47 wherein R.sup.A is
##STR00026## ##STR00027## ##STR00028## ##STR00029## ##STR00030## ##STR00031## ##STR00032## ##STR00033## ##STR00034## ##STR00035## ##STR00036## ##STR00037## ##STR00038## ##STR00039## ##STR00040## ##STR00041## ##STR00042## ##STR00043## ##STR00044##
E49 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, E26, E37, E38, E39, E40, E41, E42, E43, E44, E45, E46, or E47 wherein R.sup.A is
##STR00045## ##STR00046## ##STR00047## ##STR00048## ##STR00049## ##STR00050## ##STR00051## ##STR00052## ##STR00053## ##STR00054## ##STR00055## ##STR00056##
E50 The compound of E1 which is selected from:
##STR00057## ##STR00058## ##STR00059## ##STR00060## ##STR00061## ##STR00062## ##STR00063## ##STR00064## ##STR00065## ##STR00066## ##STR00067## ##STR00068## ##STR00069## ##STR00070## ##STR00071## ##STR00072## ##STR00073## ##STR00074## ##STR00075## ##STR00076## ##STR00077## ##STR00078## ##STR00079##
##STR00080## ##STR00081## ##STR00082## ##STR00083## ##STR00084## ##STR00085## ##STR00086## ##STR00087## ##STR00088## ##STR00089## ##STR00090## ##STR00091## ##STR00092## ##STR00093## ##STR00094## ##STR00095## ##STR00096## ##STR00097## ##STR00098## ##STR00099## ##STR00100## ##STR00101## ##STR00102##
##STR00103## ##STR00104## ##STR00105## ##STR00106## ##STR00107## ##STR00108## ##STR00109## ##STR00110## ##STR00111## ##STR00112## ##STR00113## ##STR00114## ##STR00115## ##STR00116## ##STR00117## ##STR00118## ##STR00119## ##STR00120## ##STR00121## ##STR00122## ##STR00123## ##STR00124## ##STR00125## ##STR00126## ##STR00127##
##STR00128## ##STR00129## ##STR00130## ##STR00131## ##STR00132## ##STR00133## ##STR00134## ##STR00135## ##STR00136## ##STR00137## ##STR00138## ##STR00139## ##STR00140## ##STR00141## ##STR00142## ##STR00143## ##STR00144## ##STR00145## ##STR00146## ##STR00147## ##STR00148##
##STR00149## ##STR00150## ##STR00151## ##STR00152## ##STR00153## ##STR00154##
and salts thereof. E51 The compound of E1, which is selected from:
##STR00155## ##STR00156## ##STR00157## ##STR00158## ##STR00159## ##STR00160## ##STR00161## ##STR00162## ##STR00163## ##STR00164## ##STR00165## ##STR00166## ##STR00167## ##STR00168## ##STR00169## ##STR00170## ##STR00171## ##STR00172## ##STR00173## ##STR00174## ##STR00175## ##STR00176## ##STR00177## ##STR00178## ##STR00179##
and salts thereof. E52 The compound of E1, which is selected from the compounds of Examples 162-593 and the free bases and salts thereof. E53 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of benzyl, 3,5-dichlorobenzyl, N-acetylpiperidin-3-yl, 2-chloro-4-fluorobenzyl, 2,4-difluorobenzyl, 2,6-dichlorobenzyl, N-(cyclohexylmethyl)piperidin-3-yl, 1-methyl-3-phenyl-1H-pyrazol-5-ylmethyl, pyridazin-4- ylmethyl, isoindolin-4-ylmethyl, alpha-phenylbenzyl, 3,4-dichlorobenzyl, 4-fluorobenzyl, 2-chlorobenzyl, 3-chlorobenzyl, 2,4-dichlorobenzyl, 4-methylbenzyl, 2-(trifluoromethyl)-4-fluorobenzyl, 4-fluorophenyl, phenyl, 3,5-dichlorophenyl, benzyl, alpha-methyl-3,5-dichlorobenzyl, 3,5-dichlorophenoxy, tert-butoxycarbonyl, 3-fluorobenzyl, 3-chloro-5-fluorobenzyl, and 4-(trifluoromethyl)-3-fluorobenzyl. E54 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is selected from the group consisting of:
##STR00180## ##STR00181## ##STR00182## ##STR00183## ##STR00184## ##STR00185##
E55 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is C.sub.6-10aryl-(X.sup.RA)--, wherein said C.sub.6-10aryl, of R.sup.A is optionally substituted with from 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino, C.sub.1-4dialkylamino, phenyl, C.sub.1-4alkanoyl, C.sub.1-4alkyl-OC(.dbd.O)--, C.sub.1-4alkyl-S(O).sub.2--, and C.sub.3-6carbocycle; and X.sup.RA is C.sub.1-4alkylene that is optionally substituted with 1 to 3 substituents selected from the group consisting of C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4heteroalkyl, oxo (.dbd.O), and phenyl that is optionally substituted with 1 to 5 substitutents selected from, F, Cl, Br, I, --NH.sub.2, --OH, --CN, --NO.sub.2, C.sub.1-4alkyl, C.sub.1-4haloalkyl, C.sub.1-4alkoxy, C.sub.1-4(halo)alkoxy, C.sub.1-4alkylamino and C.sub.1-4dialkylamino. E56 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is
##STR00186##
E57 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, or E26 wherein R.sup.A is
##STR00187##
E58 The compound of E1, E2, E3, E4, E5, E6, E7, E8, E9, E10, E11, E12, E13, E14, E15, E16, E17, E18, E19, E20, E21, E22, E23, E24, E25, E26, E37, E38, E39, E40, E41, E42, E43, E44, E45, E46, or E47 wherein R.sup.A is
##STR00188##
E59 The compound of E1, which is selected from:
##STR00189##
and salts thereof.
[0060] In another aspect the present invention provides for a pharmaceutical composition comprising a compound of formula I or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
[0061] In another aspect the present invention provides for a method a treating disease or condition in a mammal selected from the group consisting of pain, depression, cardiovascular diseases, respiratory diseases, and psychiatric diseases, and combinations thereof, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof. In another aspect of the present invention said disease or condition is selected from the group consisting neuropathic pain, inflammatory pain, visceral pain, cancer pain, chemotherapy pain, trauma pain, surgical pain, post-surgical pain, childbirth pain, labor pain, neurogenic bladder, ulcerative colitis, chronic pain, persistent pain, peripherally mediated pain, centrally mediated pain, chronic headache, migraine headache, sinus headache, tension headache, phantom limb pain, dental pain, peripheral nerve injury or a combination thereof. In another aspect of the present invention said disease or condition is selected from the group consisting of pain associated with HIV, HIV treatment induced neuropathy, trigeminal neuralgia, post-herpetic neuralgia, eudynia, heat sensitivity, tosarcoidosis, irritable bowel syndrome, Crohns disease, pain associated with multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), diabetic neuropathy, peripheral neuropathy, arthritis, rheumatoid arthritis, osteoarthritis, atherosclerosis, paroxysmal dystonia, myasthenia syndromes, myotonia, malignant hyperthermia, cystic fibrosis, pseudoaldosteronism, rhabdomyolysis, hypothyroidism, bipolar depression, anxiety, schizophrenia, sodium channel toxi related illnesses, familial erythromelalgia, primary erythromelalgia, familial rectal pain, cancer, epilepsy, partial and general tonic seizures, restless leg syndrome, arrhythmias, fibromyalgia, neuroprotection under ischaemic conditions cause by stroke or neural trauma, tach-arrhythmias, atria fibrillation and ventricular fibrillation.
[0062] In another aspect the present invention provides for a method of treating pain in a mammal by the inhibition of ion flux through a voltage-dependent sodium channel in the mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0063] In another aspect the present invention provides for a method of decreasing ion flux through a voltage-dependent sodium channel in a cell in a mammal, wherein the method comprises contacting the cell with a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0064] In another aspect the present invention provides for a method of treating pruritus in a mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0065] In another aspect the present invention provides for a method of treating cancer in mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0066] In another aspect the present invention provides for a method of treating, but not preventing, pain in a mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof. In another aspect of the present invention the pain as selected from the group consisting of neuropathic pain, inflammatory pain, visceral pain, cancer pain, chemotherapy pain, trauma pain, surgical pain, post-surgical pain, childbirth pain, labor pain, neurogenic bladder, ulcerative colitis, chronic pain, persistent pain, peripherally mediated pain, centrally mediated pain, chronic headache, migraine headache, sinus headache, tension headache, phantom limb pain, dental pain, peripheral nerve injury or a combination thereof. In another aspect the present invention the pain is associated with a disease or condition selected from the group consisting of HIV, HIV treatment induced neuropathy, trigeminal neuralgia, post-herpetic neuralgia, eudynia, heat sensitivity, tosarcoidosis, irritable bowel syndrome, Crohns disease, pain associated with multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), diabetic neuropathy, peripheral neuropathy, arthritis, rheumatoid arthritis, osteoarthritis, atherosclerosis, paroxysmal dystonia, myasthenia syndromes, myotonia, malignant hyperthermia, cystic fibrosis, pseudoaldosteronism, rhabdomyolysis, hypothyroidism, bipolar depression, anxiety, schizophrenia, sodium channel toxi related illnesses, familial erythromelalgia, primary erythromelalgia, familial rectal pain, cancer, epilepsy, partial and general tonic seizures, restless leg syndrome, arrhythmias, fibromyalgia, neuroprotection under ischaemic conditions cause by stroke or neural trauma, tach-arrhythmias, atrial fibrillation and ventricular fibrillation.
[0067] In another aspect the present invention provides for a method of treating, but not preventing, acute pain or chronic pain in a mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0068] In another aspect the present invention provides for a method of treating, but not preventing, neuropathic pain or inflammatory pain in a mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0069] In another aspect the present invention provides for a method for the treatment or prophylaxis of pain, depression, cardiovascular disease, respiratory disease, or psychiatric disease, or a combinations thereof, in an animal which method comprises administering an effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
[0070] In another aspect the present invention provides for a compound of formula I, or a pharmaceutically acceptable salt thereof for the use as a medicament for the treatment of diseases and disorders selected from the group consisting of pain, depression, cardiovascular diseases, respiratory diseases, and psychiatric diseases, or a combination thereof.
[0071] In another aspect the present invention provides for the use of a compound of formula I, or a pharmaceutically acceptable salt thereof for the manufacture of a medicament for the treatment of diseases and disorders selected from the group consisting of pain, depression, cardiovascular diseases, respiratory diseases, and psychiatric diseases, or a combination thereof.
[0072] In another aspect the present invention provides for the invention as described herein.
DETAILED DESCRIPTION OF THE INVENTION
Definitions
[0073] As used herein, the term "alkyl", by itself or as part of another substituent, means, unless otherwise stated, a straight or branched chain hydrocarbon radical, having the number of carbon atoms designated (i.e., C.sub.1-8 means one to eight carbons). Examples of alkyl groups include methyl, ethyl, n-propyl, iso-propyl, n-butyl, t-butyl, iso-butyl, sec-butyl, n-hexyl, n-heptyl, n-octyl, and the like. The term "alkenyl" refers to an unsaturated alkyl radical having one or more double bonds. Similarly, the term "alkynyl" refers to an unsaturated alkyl radical having one or more triple bonds. Examples of such unsaturated alkyl groups include vinyl, 2-propenyl, crotyl, 2-isopentenyl, 2-(butadienyl), 2,4-pentadienyl, 3-(1,4-pentadienyl), ethynyl, 1- and 3 -propynyl, 3-butynyl, and the higher homologs and isomers.
[0074] The term "heteroalkyl," by itself or in combination with another term, means, unless otherwise stated, a stable straight or branched chain hydrocarbon radical, consisting of the stated number of carbon atoms and from one to three heteroatoms selected from the group consisting of O, N, Si and S, and wherein the nitrogen and sulfur atoms can optionally be oxidized and the nitrogen heteroatom can optionally be quaternized. The heteroatom(s) O, N and S can be placed at any interior position of the heteroalkyl group. The heteroatom Si can be placed at any position of the heteroalkyl group, including the position at which the alkyl group is attached to the remainder of the molecule. A "heteroalkyl" can contain up to three units of unsaturation, and also include mono- and poly-halogenated variants, or combinations thereof. Examples include --CH.sub.2--CH.sub.2--O--CH.sub.3, --CH.sub.2--CH.sub.2--O--CF.sub.3, --CH.sub.2--CH.sub.2--NH--CH.sub.3, --CH.sub.2--CH.sub.2--N(CH.sub.3)--CH.sub.3, --CH.sub.2--S--CH.sub.2--C H.sub.3, --S(O)--CH.sub.3, --CH.sub.2--CH.sub.2--S(O).sub.2--CH.sub.3, --CH.dbd.CH--O--CH.sub.3, --Si(CH.sub.3).sub.3, --CH.sub.2--CH.dbd.N--OCH.sub.3, and --CH.dbd.CH.dbd.N(CH.sub.3)--CH.sub.3. Up to two heteroatoms can be consecutive, such as, for example, --CH.sub.2NH--OCH.sub.3 and --CH.sub.2--O--Si(CH.sub.3).sub.3.
[0075] The term "alkylene" by itself or as part of another substituent means a divalent radical derived from an alkane (including branched alkane), as exemplified by --CH.sub.2CH.sub.2CH.sub.2CH.sub.2-- and --CH(CH.sub.2)CH.sub.2CH.sub.2--. Typically, an alkyl (or alkylene) group will have from 1 to 24 carbon atoms, with those groups having 10 or fewer carbon atoms being preferred in the present invention. "Alkenylene" and "alkynylene" refer to the unsaturated forms of "alkylene" having double or triple bonds, respectively. "Alkylene", "alkenylene" and "alkynylene" are also meant to include mono and poly-halogenated variants.
[0076] The term "heteroalkylene" by itself or as part of another substituent means a divalent radical, saturated or unsaturated or polyunsaturated, derived from heteroalkyl, as exemplified by and --CH.sub.2--CH.sub.2--S--CH.sub.2--CH.sub.2-- and --CH.sub.2--S--CH.sub.2--NH--CH.sub.2--, --O--CH.sub.2--CH.dbd.CH--, --CH.sub.2--CH.dbd.CH(H)CH.sub.2--O--CH.sub.2-- and --S--CH.sub.2--C.dbd.C--. For heteroalkylene groups, heteroatoms can also occupy either or both of the chain termini (e.g., alkyleneoxy, alkylenedioxy, alkyleneamino, alkylenediamino, and the like). The term "heteroalkylene" is also meant to include mono and poly-halogenated variants.
[0077] The terms "alkoxy," "alkylamino" and "alkylthio", are used in their conventional sense, and refer to those alkyl groups attached to the remainder of the molecule via an oxygen atom ("oxy"), an amino group ("amino") or thio group, and further include mono- and poly-halogenated variants thereof. Additionally, for dialkylamino groups, the alkyl portions can be the same or different.
[0078] The terms "halo" or "halogen," by themselves or as part of another substituent, mean, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom. The term "(halo)alkyl" is meant to include both a "alkyl" and "haloalkyl" substituent. Additionally, the term "haloalkyl," is meant to include monohaloalkyl and polyhaloalkyl. For example, the term "C.sub.1-4haloalkyl" is mean to include trifluoromethyl, 2,2,2,-trifluoroethyl, 4-chlorobutyl, 3-bromopropyl, difluoromethyl, and the like.
[0079] The term "aryl" as used herein refers to a single all carbon aromatic ring or a multiple condensed all carbon ring system wherein at least one of the rings is aromatic. For example, in certain embodiments, an aryl group has 6 to 20 carbon atoms, 6 to 14 carbon atoms, or 6 to 12 carbon atoms. Aryl includes a phenyl radical. Aryl also includes multiple condensed ring systems (e.g., ring systems comprising 2, 3 or 4 rings) having about 9 to 20 carbon atoms in which at least one ring is aromatic and wherein the other rings may be aromatic or not aromatic (i.e., carbocycle). Such multiple condensed ring systems are optionally substituted with one or more (e.g., 1, 2 or 3) oxo groups on any carbocycle portion of the multiple condensed ring system. The rings of the multiple condensed ring system can be connected to each other via fused, spiro and bridged bonds when allowed by valency requirements. It is to be understood that the point of attachment of a multiple condensed ring system, as defined above, can be at any position of the ring system including an aromatic or a carbocycle portion of the ring. Non-limiting examples of aryl groups include, but are not limited to, phenyl, indenyl, naphthyl, 1, 2, 3, 4-tetrahydronaphthyl, anthracenyl, and the like.
[0080] The term "carbocycle" or "carbocyclyl" refers to a single saturated (i.e., cycloalkyl) or a single partially unsaturated (e.g., cycloalkenyl, cycloalkadienyl, etc.) all carbon ring having 3 to 7 carbon atoms (i.e., (C.sub.3-C.sub.7)carbocycle). The term "carbocycle" or "carbocyclyl" also includes multiple condensed, saturated and partially unsaturated all carbon ring systems (e.g., ring systems comprising 2, 3 or 4 carbocyclic rings). Accordingly, carbocycle includes multicyclic carbocyles such as a bicyclic carbocycles (e.g., bicyclic carbocycles having about 6 to 12 carbon atoms such as bicyclo[3.1.0]hexane and bicyclo[2.1.1]hexane), and polycyclic carbocycles (e.g tricyclic and tetracyclic carbocycles with up to about 20 carbon atoms). The rings of the multiple condensed ring system can be connected to each other via fused, spiro and bridged bonds when allowed by valency requirements. For example, multicyclic carbocyles can be connected to each other via a single carbon atom to form a spiro connection (e.g., spiropentane, spiro[4,5]decane, etc), via two adjacent carbon atoms to form a fused connection (e.g., carbocycles such as decahydronaphthalene, norsabinane, norcarane) or via two non-adjacent carbon atoms to form a bridged connection (e.g., norbornane, bicyclo[2.2.2]octane, etc). The "carbocycle" or "carbocyclyl" can also be optionally substituted with one or more (e.g., 1, 2 or 3) oxo groups. In one embodiment the term carbocycle includes a C.sub.3-12carbocycle. In one embodiment the term carbocycle includes a C.sub.3-8carbocycle. In one embodiment the term carbocycle includes a C.sub.3-6carbocycle. In one embodiment the term carbocycle includes a C.sub.3-5carbocycle. Non-limiting examples of carbocycles include cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopent-1-enyl, 1-cyclopent-2-enyl, 1-cyclopent-3-enyl, cyclohexyl, 1-cyclohex-1 -enyl, 1-cyclohex-2-enyl, bicyclo[2.2.1]heptane, pinane, adamantane, norborene, spirocyclic C.sub.3-12alkane, and 1-cyclohex-3-enyl.
[0081] The term "heteroaryl" as used herein refers to a single aromatic ring that has at least one atom other than carbon in the ring, wherein the atom is selected from the group consisting of oxygen, nitrogen and sulfur; "heteroaryl" also includes multiple condensed ring systems that have at least one such aromatic ring, which multiple condensed ring systems are further described below. Thus, "heteroaryl" includes single aromatic rings of from about 1 to 6 carbon atoms and about 1-4 heteroatoms selected from the group consisting of oxygen, nitrogen and sulfur. The sulfur and nitrogen atoms may also be present in an oxidized form provided the ring is aromatic. Exemplary heteroaryl ring systems include but are not limited to pyridyl, pyrimidinyl, oxazolyl or furyl. "heteroaryl" also includes multiple condensed ring systems (e.g., ring systems comprising 2, 3 or 4 rings) wherein a heteroaryl group, as defined above, is condensed with one or more rings selected from heteroaryls (to form for example a naphthyridinyl such as 1,8-naphthyridinyl), heterocycles, (to form for example a 1,2,3,4-tetrahydronaphthyridinyl such as 1,2,3,4-tetrahydro-1,8-naphthyridinyl), carbocycles (to form for example 5,6,7,8 -tetrahydroquinolyl) and aryls (to form for example indazolyl) to form the multiple condensed ring system. Thus, a heteroaryl (a single aromatic ring or multiple condensed ring system) has about 1-20 carbon atoms and about 1-6 heteroatoms within the heteroaryl ring. Such multiple condensed ring systems may be optionally substituted with one or more (e.g., 1, 2, 3 or 4) oxo groups on the carbocycle or heterocycle portions of the condensed ring. The rings of the multiple condensed ring system can be connected to each other via fused, spiro and bridged bonds when allowed by valency requirements. It is to be understood that the individual rings of the multiple condensed ring system may be connected in any order relative to one another. It is also to be understood that the point of attachment of a multiple condensed ring system (as defined above for a heteroaryl) can be at any position of the multiple condensed ring system including a heteroaryl, heterocycle, aryl or carbocycle portion of the multiple condensed ring system. It is also to be understood that the point of attachment for a heteroaryl or heteroaryl multiple condensed ring system can be at any suitable atom of the heteroaryl or heteroaryl multiple condensed ring system including a carbon atom and a heteroatom (e.g., a nitrogen). Exemplary heteroaryls include but are not limited to pyridyl, pyrrolyl, pyrazinyl, pyrimidinyl, pyridazinyl, pyrazolyl, thienyl, indolyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, furyl, oxadiazolyl, thiadiazolyl, quinolyl, isoquinolyl, benzothiazolyl, benzoxazolyl, indazolyl, quinoxalyl, quinazolyl, 5,6,7,8 tetrahydroisoquinolinyl benzofuranyl, benzimidazolyl, thianaphthenyl, pyrrolo[2,3-b]pyridinyl, quinazolinyl-4(3H)-one, triazolyl, 4,5,6,7-tetrahydro-1H-indazole and 3b,4,4a,5-tetrahydro-1H-cyclopropa[3,4]cyclo- penta[1,2-c]pyrazole.
[0082] The term "heterocyclyl" or "heterocycle" as used herein refers to a single saturated or partially unsaturated ring that has at least one atom other than carbon in the ring, wherein the atom is selected from the group consisting of oxygen, nitrogen and sulfur; the term also includes multiple condensed ring systems that have at least one such saturated or partially unsaturated ring, which multiple condensed ring systems are further described below. Thus, the term includes single saturated or partially unsaturated rings (e.g., 3, 4, 5, 6 or 7 -membered rings) from about 1 to 6 carbon atoms and from about 1 to 3 heteroatoms selected from the group consisting of oxygen, nitrogen and sulfur in the ring. The ring may be substituted with one or more (e.g., 1, 2 or 3) oxo groups and the sulfur and nitrogen atoms may also be present in their oxidized forms. Exemplary heterocycles include but are not limited to azetidinyl, tetrahydrofuranyl and piperidinyl. The term "heterocycle" also includes multiple condensed ring systems (e.g., ring systems comprising 2, 3 or 4 rings) wherein a single heterocycle ring (as defined above) can be condensed with one or more groups selected from heterocycles (to form for example a 1,8-decahydronapthyridinyl), carbocycles (to form for example a decahydroquinolyl) and aryls to form the multiple condensed ring system. Thus, a heterocycle (a single saturated or single partially unsaturated ring or multiple condensed ring system) has about 2-20 carbon atoms and 1-6 heteroatoms within the heterocycle ring. Such multiple condensed ring systems may be optionally substituted with one or more (e.g., 1, 2, 3 or 4) oxo groups on the carbocycle or heterocycle portions of the multiple condensed ring. The rings of the multiple condensed ring system can be connected to each other via fused, spiro and bridged bonds when allowed by valency requirements. It is to be understood that the individual rings of the multiple condensed ring system may be connected in any order relative to one another. It is also to be understood that the point of attachment of a multiple condensed ring system (as defined above for a heterocycle) can be at any position of the multiple condensed ring system including a heterocycle, aryl and carbocycle portion of the ring. It is also to be understood that the point of attachment for a heterocycle or heterocycle multiple condensed ring system can be at any suitable atom of the heterocycle or heterocycle multiple condensed ring system including a carbon atom and a heteroatom (e.g., a nitrogen). In one embodiment the term heterocycle includes a C.sub.2-20heterocycle. In one embodiment the term heterocycle includes a C.sub.2-7 heterocycle. In one embodiment the term heterocycle includes a C.sub.2-5heterocycle. In one embodiment the term heterocycle includes a C.sub.2-4heterocycle. Exemplary heterocycles include, but are not limited to aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, homopiperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, tetrahydrofuranyl, dihydrooxazolyl, tetrahydropyranyl, tetrahydrothiopyranyl, 1,2,3,4-tetrahydroquinolyl, benzoxazinyl, dihydrooxazolyl, chromanyl, 1,2-dihydropyridinyl, 2,3-dihydrobenzofuranyl, 1,3-benzodioxolyl, 1,4-benzodioxanyl, spiro[cyclopropane-1,1'-isoindolinyl]-3'-one, isoindolinyl-1-one, 2-oxa-6-azaspiro[3.3]heptanyl, imidazolidin-2-one N-methylpiperidine, imidazolidine, pyrazolidine, butyrolactam, valerolactam, imidazolidinone, hydantoin, dioxolane, phthalimide, 1,4-dioxane, thiomorpholine, thiomorpholine-S-oxide, thiomorpholine-S,S-oxide, pyran, 3-pyrroline, thiopyran, pyrone, tetrhydrothiophene, quinuclidine, tropane, 2-azaspiro[3.3]heptane, (1R,5S)-3-azabicyclo[3.2.1]octane, (1s,4s)-2-azabicyclo[2.2.2]octane, (1R,4R)-2-oxa-5-azabicyclo[2.2.2]octane and pyrrolidin-2-one.
[0083] the above terms (e.g., "alkyl," "aryl" and "heteroaryl"), in some embodiments, will include both substituted and unsubstituted forms of the indicated radical. Preferred substituents for each type of radical are provided below.
[0084] Substituents for the alkyl radicals (including those groups often referred to as alkylene, alkenyl, alkynyl, heteroalkyl, carbocycle, and heterocyclyl) can be a variety of groups including, but not limited to, -halogen, --OR', --NR'R'', --SR', --SiR'R''R''', --OC(O)R', --C(O)R', --CONR'R'', --OC(O)NR'R'', --NR''C( O)R', --NR'''C(O)NR'R'', --NR''C(O).sub.2R', --NHC(NH.sub.2).dbd.NH, --NRC(NH.sub.2).dbd.NH, --NHC(NH.sub.2).dbd.NR', --NR'''C(NR'R'').dbd.N--CN, --NR'''C(NR'R'').dbd.NOR', --NHC(NH.sub.2).dbd.NR',--S(O)R', --S(O).sub.2R', --S(O).sub.2NR'R'', --NR'S(O).sub.2R'', --NR'''S(O).sub.1NR'R'', --CN, --NO.sub.2, --(CH.sub.2).sub.1-4--OR', --(CH.sub.2).sub.1-4--SR', --(CH.sub.2).sub.1-4--SiR'R''R''', --(CH.sub.2).sub.1-4--OC( O)R', --(CH.sub.2).sub.1-4--C(O)R', --(CH.sub.2).sub.1-4--CO.sub.2R', --(CH.sub.2).sub.1-4CONR'R'', in a number ranging from zero to (2 m'+1), where m' is the total number of carbon atoms in such radical. R', R'' and R''' each independently refer groups including, for example, hydrogen, unsubstituted C.sub.1-6alkyl, unsubstituted heteroalkyl, unsubstituted aryl, aryl substituted with 1-3 halogens, unsubstituted C.sub.1-6alkyl, C.sub.1-6alkoxy or thioalkoxy groups, or unsubstituted aryl-C.sub.1-4alkyl groups, unsubstituted heteroaryl, substituted heteroaryl, among others. When R' and R'' are attached to the same nitrogen atom, they can be combined with the nitrogen atom to form a 3-, 4-, 5-, 6-, or 7-membered ring. For example, --NR'R'' is meant to include 1-pyrrolidinyl and 4-morpholinyl. Other substitutents for alkyl radicals, including heteroalkyl, alkylene, include for example, .dbd.O, .dbd.NR', .dbd.N--OR', .dbd.N--CN, .dbd.NH, wherein R' include substituents as described above.
[0085] Similarly, substituents for the aryl and heteroaryl groups are varied and are generally selected from the group including, but not limited to halogen, --OR', --OC(O)R', --NR'R'', --SR', --R',--CN, --NO.sub.2, --CONR'R'', --C(O)R', --OC(O)NR'R'', --NR''C(O)R', --NR''C(O).sub.2R', --NR'C(O)NR''R''', --NHC(NH.sub.2).dbd.NH, --NR'C(NH.sub.1).dbd.NH, --NHC(NH.sub.2).dbd.NR', --S(O)R', --S(O).sub.2R', --S(O).sub.2NR'R'', --NJ'S(O).sub.2R'', --N.sub.3, perfluoro-C.sub.1-4alkoxy, and perfluoro-C.sub.1-4alkyl, --(CH.sub.2).sub.1-4--OR', --(CH.sub.1).sub.1-4--NR'R'', --(CH.sub.2).sub.1-4--SR', --(CH.sub.2).sub.1-4SiR'R''R''', --C(CH.sub.2).sub.1-4-OC(O)R', --(CH.sub.2).sub.1-4--CO.sub.2R', --(CH.sub.2).sub.1-4CONR'R'', in a number ranging from zero to the total number of open valences on the aromatic ring system; and where and R', R'' and R''' are independently selected from hydrogen, C.sub.1-6alkyl, C.sub.3-6carbocycle, C.sub.2-6alkenyl, C.sub.2-6alkynyl, unsubstituted aryl and heteroaryl, (unsubstituted aryl)-C.sub.1-4alkyl, and unsubstituted aryloxy-C.sub.1-4alkyl. Other suitable substituents include each of the above aryl substituents attached to a ring atom by an alkylene tether of from 1-4 carbon atoms. When a substituent for the aryl heteroaryl group contains an alkylene linker (e.g., --(CH.sub.2).sub.1-4--NR'R''), the alkylene linker includes halo variants as well. For example, the linker "--(CH.sub.2).sub.1-4--" when used as part of a substituent is meant to include difluoromethylene, 1,2-difluoroethylene, etc.
[0086] As used herein, the term "heteroatom" is meant to include oxygen (O), nitrogen (N), sulfur (S) and silicon (Si).
[0087] As used herein, the term "chiral" refers to molecules which have the property of non-superimposability of the mirror image partner, while the term "achiral" refers to molecules which are superimposable on their mirror image partner.
[0088] As used herein, the term "stereoisomers" refers to compounds which have identical chemical constitution, but differ with regard to the arrangement of the atoms or groups in space.
[0089] As used herein a wavy line "" that intersects a bond in a chemical structure indicates the point of attachment of the bond that the wavy bond intersects in the chemical structure to the remainder of a molecule.
[0090] As used herein, the term "C-linked" means that the group that the term describes is attached the remainder of the molecule through a ring carbon atom.
[0091] As used herein, the term "N-linked" means that the group that the term describes is attached to the remainder of the molecule through a ring nitrogen atom.
[0092] "Diastereomer" refers to a stereoisomer with two or more centers of chirality and whose molecules are not mirror images of one another. Diastereomers have different physical properties, e.g. melting points, boiling points, spectral properties, and reactivities. Mixtures of diastereomers can separate under high resolution analytical procedures such as electrophoresis and chromatography.
[0093] "Enantiomers" refer to two stereoisomers of a compound which are non-superimposable mirror images of one another.
[0094] Stereochemical definitions and conventions used herein generally follow S. P. Parker, Ed., McGraw-Hill Dictionary of Chemical Terms (1984) McGraw-Hill Book Company, New York; and Eliel, E. and Wilen, S., "Stereochemistry of Organic Compounds", John Wiley & Sons, Inc., New York, 1994. The compounds of the invention can contain asymmetric or chiral centers, and therefore exist in different stereoisomeric forms. It is intended that all stereoisomeric forms of the compounds of the invention, including but not limited to, diastereomers, enantiomers and atropisomers, as well as mixtures thereof such as racemic mixtures, form part of the present invention. Many organic compounds exist in optically active forms, i.e., they have the ability to rotate the plane of plane-polarized light. In describing an optically active compound, the prefixes D and L, or R and S, are used to denote the absolute configuration of the molecule about its chiral center(s). The prefixes d and l or (+) and (-) are employed to designate the sign of rotation of plane-polarized light by the compound, with (-) or l meaning that the compound is levorotatory. A compound prefixed with (+) or d is dextrorotatory. For a given chemical structure, these stereoisomers are identical except that they are mirror images of one another. A specific stereoisomer can also be referred to as an enantiomer, and a mixture of such isomers is often called an enantiomeric mixture. A 50:50 mixture of enantiomers is referred to as a racemic mixture or a racemate, which can occur where there has been no stereoselection or stereospecificity in a chemical reaction or process. The terms "racemic mixture" and "racemate" refer to an equimolar mixture of two enantiomeric species, devoid of optical activity.
[0095] As used herein, the term "tautomer" or "tautomeric form" refers to structural isomers of different energies which are interconvertible via a low energy barrier. For example, proton tautomers (also known as prototropic tautomers) include interconversions via migration of a proton, such as keto-enol and imine-enamine isomerizations. Valence tautomers include interconversions by reorganization of some of the bonding electrons.
[0096] As used herein, the term "solvate" refers to an association or complex of one or more solvent molecules and a compound of the invention. Examples of solvents that form solvates include, but are not limited to, water, isopropanol, ethanol, methanol, DMSO, ethyl acetate, acetic acid, and ethanolamine. The term "hydrate" refers to the complex where the solvent molecule is water.
[0097] As used herein, the term "protecting group" refers to a substituent that is commonly employed to block or protect a particular functional group on a compound. For example, an "amino-protecting group" is a substituent attached to an amino group that blocks or protects the amino functionality in the compound. Suitable amino-protecting groups include acetyl, trifluoroacetyl, t-butoxycarbonyl (BOC), benzyloxycarbonyl (CBZ) and 9-fluorenylmethylenoxycarbonyl (Fmoc). Similarly, a "hydroxy-protecting group" refers to a substituent of a hydroxy group that blocks or protects the hydroxy functionality. Suitable protecting groups include acetyl and silyl. A "carboxy-protecting group" refers to a substituent of the carboxy group that blocks or protects the carboxy functionality. Common carboxy-protecting groups include phenylsulfonylethyl, cyanoethyl, 2-(trimethylsilyl)ethyl, 2-(trimethylsilyl)ethoxymethyl, 2-(p-toluenesulfonyl)ethyl, 2-(p-nitrophenylsulfenyl)ethyl, 2-(diphenylphosphino)-ethyl, nitroethyl and the like. For a general description of protecting groups and their use, see P. G. M. Wuts and T. W. Greene, Greene's Protective Groups in Organic Synthesis 4.sup.th edition, Wiley-Interscience, New York, 2006.
[0098] As used herein, the term "mammal" includes, but is not limited to, humans, mice, rats, guinea pigs, monkeys, dogs, cats, horses, cows, pigs, and sheep
[0099] As used herein, the term "pharmaceutically acceptable salts" is meant to include salts of the active compounds which are prepared with relatively nontoxic acids or bases, depending on the particular substituents found on the compounds described herein. When compounds of the present invention contain relatively acidic functionalities, base addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired base, either neat or in a suitable inert solvent. Examples of salts derived from pharmaceutically-acceptable inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic, manganous, potassium, sodium, zinc and the like. Salts derived from pharmaceutically-acceptable organic bases include salts of primary, secondary and tertiary amines, including substituted amines, cyclic amines, naturally-occurring amines and the like such as arginine, betaine, caffeine, choline, N,N'-dibenzylethylenediamine, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N-ethylmorpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine, tripropylamine, tromethamine and the like. When compounds of the present in invention contain relatively basic functionalities, acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent. Examples of pharmaceutically acceptable acid addition salts include those derived from inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phosphorous acids and the like, as well as the salts derived from relatively nontoxic organic acids like acetic, propionic, isobutyric, malonic, benzoic, succinic, suberic, fumaric, mandelic, phthalic, benzenesulfonic, p-tolylsulfonic, citric, tartaric, methanesulfonic, and the like. Also included are salts of amino acids such as arginate and the like, and salts of organic acids like glucuronic or galactunoric acids and the like (see, for example, Berge, S. M., et al., "Pharmaceutical Salts", Journal of Pharmaceutical Science, 1977, 66, 1-19). Certain specific compounds of the present invention contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts.
[0100] The neutral forms of the compounds can be generated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner. The parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present invention.
[0101] In addition to salt forms, the present invention provides compounds which are in a prodrug form. As used herein the term "prodrug" refers to those compounds that readily undergo chemical changes under physiological conditions to provide the compounds of the present invention. Additionally, prodrugs can be converted to the compounds of the present invention by chemical or biochemical methods in an ex vivo environment. For example, prodrugs can be slowly converted to the compounds of the present invention when placed in a transdermal patch reservoir with a suitable enzyme or chemical reagent.
[0102] Prodrugs of the invention include compounds wherein an amino acid residue, or a polypeptide chain of two or more (e.g., two, three or four) amino acid residues, is covalently joined through an amide or ester bond to a free amino, hydroxy or carboxylic acid group of a compound of the present invention. The amino acid residues include but are not limited to the 20 naturally occurring amino acids commonly designated by three letter symbols and also includes phosphoserine, phosphothreonine, phosphotyrosine, 4-hydroxyproline, hydroxylysine, demosine, isodemosine, gamma-carboxyglutamate, hippuric acid, octahydroindole-2-carboxylic acid, statine, 1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid, penicillamine, ornithine, 3-methylhistidine, norvaline, beta-alanine, gamma-aminobutyric acid, citrulline, homocysteine, homoserine, methyl-alanine, para-benzoylphenylalanine, phenylglycine, propargylglycine, sarcosine, methionine sulfone and tert-butylglycine.
[0103] Additional types of prodrugs are also encompassed. For instance, a free carboxyl group of a compound of the invention can be derivatized as an amide or alkyl ester. As another example, compounds of this invention comprising free hydroxy groups can be derivatized as prodrugs by converting the hydroxy group into a group such as, but not limited to, a phosphate ester, hemisuccinate, dimethylaminoacetate, or phosphoryloxymethyloxycarbonyl group, as outlined in Fleisher, D. et al., (1996) Improved oral drug delivery; solubility limitations overcome by the use of prodrugs Advanced Drug Delivery Reviews, 19:115. Carbamate prodrugs of hydroxy and amino groups are also included, as are carbonate prodrugs, sulfonate esters and sulfate esters of hydroxy groups. Derivatization of hydroxy groups as (acyloxy)methyl and (acyloxy)ethyl ethers, wherein the acyl group can be an alkyl ester optionally substituted with groups including, but not limited to, ether, amine and carboxylic acid functionalities, or where the acyl group is an amino acid ester as described above, are also encompassed. Prodrugs of this type are described in J. Med. Chem., (1996), 39:10. More specific examples include replacement of the hydrogen atom of the alcohol group with a group such as (C.sub.1-6)alkanoyloxymethyl, 1-((C.sub.1-6)alkanoyloxy)ethyl, 1-methyl-1-((C.sub.1-6)alkanoyloxy)ethyl, (C.sub.1-6)alkoxycarbonyloxymethyl, N--(C.sub.1-6)alkoxycarbonylaminomethyl, succinoyl, (C.sub.1-6)alkanoyl, alpha-amino(C.sub.1-4)alkanoyl, arylacyl and alpha-aminoacyl, or alpha-aminoacyl-alpha-aminoacyl, where each alpha-aminoacyl group is independently selected from the naturally occurring L-amino acids, P(O)(OH).sub.2, --P(O)(O(C.sub.1-6)alkyl).sub.2 or glycosyl (the radical resulting from the removal of a hydroxyl group of the hemiacetal form of a carbohydrate).
[0104] For additional examples of prodrug derivatives, see, for example, a) Design of Prodrugs, edited by H. Bundgaard, (Elsevier, 1985) and Methods in Enzymology, Vol. 42, p. 309-396, edited by K. Widder, et al. (Academic Press, 1985); b) A Textbook of Drug Design and Development, edited by Krogsgaard-Larsen and H. Bundgaard, Chapter 5 "Design and Application of Prodrugs," by H. Bundgaard p. 113-191 (1991); c) H. Bungaard, Advanced Drug Delivery Reviews, 8:1-38 (1992); d) H. Bundgaard, et al., Journal of Pharmaceutical Sciences, 77:285 (1988); and e) N. Kakeya, et al., Chem. Pharm. Bull., 32:692 (1984), each of which is specifically incorporated herein by reference.
[0105] Additionally, the present invention provides for metabolites of compounds of the invention. As used herein, a "metabolite" refers to a product produced through metabolism in the body of a specified compound or salt thereof. Such products can result for example from the oxidation, reduction, hydrolysis, amidation, deamidation, esterification, deesterification, enzymatic cleavage, and the like, of the administered compound.
[0106] Metabolite products typically are identified by preparing a radiolabelled (e.g., .sup.14C or .sup.3H) isotope of a compound of the invention, administering it parenterally in a detectable dose (e.g., greater than about 0.5 mg/kg) to an animal such as rat, mouse, guinea pig, monkey, or to man, allowing sufficient time for metabolism to occur (typically about 30 seconds to 30 hours) and isolating its conversion products from the urine, blood or other biological samples. These products are easily isolated since they are labeled (others are isolated by the use of antibodies capable of binding epitopes surviving in the metabolite). The metabolite structures are determined in conventional fashion, e.g., by MS, LC/MS or NMR analysis. In general, analysis of metabolites is done in the same way as conventional drug metabolism studies well known to those skilled in the art. The metabolite products, so long as they are not otherwise found in vivo, are useful in diagnostic assays for therapeutic dosing of the compounds of the invention.
[0107] Certain compounds of the present invention can exist in unsolvated forms as well as solvated forms, including hydrated forms. In general, the solvated forms are equivalent to unsolvated forms and are intended to be encompassed within the scope of the present invention. Certain compounds of the present invention can exist in multiple crystalline or amorphous forms. In general, all physical forms are equivalent for the uses contemplated by the present invention and are intended to be within the scope of the present invention.
[0108] Certain compounds of the present invention possess asymmetric carbon atoms (optical centers) or double bonds; the racemates, diastereomers, geometric isomers, regioisomers and individual isomers (e.g., separate enantiomers) are all intended to be encompassed within the scope of the present invention.
[0109] The compounds of the present invention can also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such compounds. For example, the present invention also embraces isotopically-labeled variants of the present invention which are identical to those recited herein, bur the for the fact that one or more atoms are replace by an atom haying the atomic mass or mass number different from the predominant atomic mass or mass number usually found in nature for the atom. All isotopes of any particular atom or element as specified are contemplated within the scope of the compounds of the invention, and their uses. Exemplary isotopes that can be incorporated in to compounds of the invention include istopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, fluorine, chlorine and iodine, such as .sup.2H ("D"), .sup.3H, .sup.11C, .sup.13C, .sup.14C, .sup.13N, .sup.15N, .sup.15O, .sup.17O, .sup.18O, .sup.32P, .sup.33P, .sup.35S, .sup.18F, .sup.36Cl, .sup.123I and .sup.125I. Certain isotopically labeled compounds of the present invention (e.g., those labeled with .sup.3H or .sup.14C) are useful in compound and/or substrate tissue distribution assays. Tritiated (.sup.3H) and carbon-14 (.sup.14C) isotopes are useful for their ease of preparation and detectability. Further substitution with heavier isotopes such as deuterium (i.e., .sup.2H) may afford certain therapeutic advantages resulting from greater metabolic stability (e.g., increased in vivo half-life or reduced dosage requirements) and hence may be preferred in some circumstances. Positron emitting isotopes such as .sup.15O, .sup.13N, .sup.11C, and .sup.18F are useful for positron emission tomography (PET) studies to examine substrate receptor occupancy. Isotopically labeled compounds of the present inventions can generally be prepared by following procedures analogous to those disclosed in the Schemes and/or in the Examples herein below, by substituting an isotopically labeled reagent for a non-isotopically labeled reagent.
[0110] The terms "treat" and "treatment" refer to both therapeutic treatment and/or prophylactic treatment or preventative measures, wherein the object is to prevent or slow down (lessen) an undesired physiological change or disorder, such as, for example, the development or spread of cancer. For purposes of this invention, beneficial or desired clinical results include, but are not limited to, alleviation of symptoms, diminishment of extent of disease or disorder, stabilized (i.e., not worsening) state of disease or disorder, delay or slowing of disease progression, amelioration or palliation of the disease state or disorder, and remission (whether partial or total), whether detectable or undetectable. "Treatment" can also mean prolonging survival as compared to expected survival if not receiving treatment. Those in need of treatment include those already with the disease or disorder as well as those prone to have the disease or disorder or those in which the disease or disorder is to be prevented.
[0111] The phrase "therapeutically effective amount" or "effective amount" of a compound of the present invention that (i) treats or prevents the particular disease, condition, or disorder, (ii) attenuates, ameliorates, or eliminates one or more symptoms of the particular disease, condition, or disorder, or (iii) prevents or delays the onset of one or more symptoms of the particular disease, condition, or disorder described herein. For cancer therapy, efficacy can, for example, be measured by assessing the time to disease progression (TTP) and/or determining the response rate (RR).
[0112] The term "bioavailability" refers to the systemic availability (i.e., blood/plasma levels) of a given amount of drug administered to a patient. Bioavailability is an absolute term that indicates measurement of both the time (rate) and total amount (extent) of drug that reaches the general circulation from an administered dosage form.
[0113] In another embodiment, the compound is selected from compounds of formula I as described in the Examples herein and salts thereof.
Synthesis of Compounds
[0114] Compounds of formula (I) may be prepared by the process illustrated in Schemes 1 and 2. Compounds of formula (I), wherein X.sup.1 is O, S, or NH, may be prepared by the processes illustrated in Scheme 1.
##STR00190##
[0115] Compounds of formula (I) can be made from compounds of formula (II) by displacement with formula (III) and a base (reaction step ii in Scheme 1). Suitable conditions include potassium tert-butoxide or cesium carbonate in DMSO, NaH in DMF, or K.sub.2CO.sub.3 in DMF. Formula (II) can be made according to step (i) by activation of the acid group of formula (IV) with reagents such as oxalyl chloride, carbonyl di-imidazole (CDI), propylphosphonic anhydride, a uronium based amide coupling agent or a carbodiimide reagent followed by displacement with a sulfonamide of formula (VII) in the presence of a nucleophilic base such as 4-dimethylaminopyridine. Illustrative conditions comprise N, N-dimethylaminopropyl-N'-ethylcarbodiimide and 4-dimethylamino- pyridine with N, N-diisopropylethylamine.
[0116] Alternatively, compounds of formula (I) can be made from compounds of formula (IV) by reversing steps (i) and (ii) as described in Scheme 1. Illustrative conditions for steps vi and vii are as previously described in steps (ii) and (i), respectively.
[0117] Compounds of formula (I) can also be made from compounds of formula (V) according to step (v) by displacement of the ester with compounds of formula (VII) and a suitable base such as potassium tert-butoxide, NaH or DBU. Compounds of formula (I) can also be made from compounds of formula (v) by a two steps sequence (see steps viii and vii in Scheme 1). Compounds of formula (V) can be made from compounds of formula (VIII) according to step (iv) via a nucleophilic substitution reaction using compounds of formula (III) and a base as described in step ii. Compounds of formula (VIII) can be made from compounds of formula (IV) according to step (iii) using protecting group methodology as described in references such as `Greene's Protective Groups in Organic Synthesis`. When Pg is tolyl, illustrative conditions comprise thionyl chloride or carbonyldiimidazole with para-cresol. When Pg is tert-butyl, illustrative conditions comprise di-tert-butyl dicarbonate and 4-dimethylaminopyridine in tert-butanol. Compounds of formula (I), wherein R.sup.5 is C.sub.1-8alkyl, C.sub.1-8haloalkyl, C.sub.1-8alkoxy, C.sub.3-8cycloalkyl or C.sub.2-7heterocycloalkyl can be prepared by the process illustrated in Scheme 2. In certain embodiment, W groups in compounds of formula (IX, X and XI) are an ester or cyano group.
##STR00191##
[0118] Compounds of formula (I) can be prepared from compounds of formula (XII) (--V.dbd.OH) according to reaction step (iv) by activation of the acid group with reagents such as oxalyl chloride, carbonyl di-imidazole (CDI), as uronium based amide coupling agent, propylphosphonic anhydride or a carbodiimide reagent followed by displacement with a suitable sulfonamide of formula (VII) in the presence of a nucleophilic base such as 4-dimethylaminopyridine.
[0119] Alternatively, compounds of formula (I) can be prepared from compounds of formula (XII) (--V.dbd.NH.sub.2) according to reaction step (v) by displacement of a sulfonyl chloride of formula (XIII) under basic reaction conditions.
[0120] Compounds of formula (XII) can be prepared by hydrolysis of the nitrile functional group in compounds of formula (XI, W.dbd.CN) or by hydrosis of the ester functional group in compounds of formula (XI, W.dbd.CO.sub.2Pg) by either acidic or basic methods according to step (iii) as required.
[0121] Compounds of formula (XI) can be prepared from compounds of formula (X) by palladium-catalyzed coupling of a compound of formula (R.sub.5M) according to step (ii). Conveniently the coupling is effective with a boronic acid or ester of formula (R.sub.5M). The coupling reaction can be carried out with a variety of palladium catalysis such as palladium acetate or tetrakistriphenylphosphine palladium (0) in various solvents and in the presence of bases such as sodium and potassium carbonate, cesium fluoride or potassium phosphate. Compounds of formula (X) can be prepared under similar conditions as described for the preparation of compounds of formula (V), (VI) and (I) in Scheme 1.
Pharmaceutical Composition and Administration
[0122] In addition to one or more of the compounds provided above (or stereoisomers, geometric isomers, tautomers, solvates, metabolites, isotopes, pharmaceutically acceptable salts, or prodrugs thereof), the invention also provides for compositions and medicaments comprising a compound of formula I or and embodiment thereof and at least one pharmaceutically acceptable carrier, diluent or excipient. The compositions of the invention can be used to selectively inhibit NaV1.7 in patients (e.g, humans).
[0123] The term "composition," as used herein, is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results, directly or indirectly, from combination of the specified ingredients in the specified amounts. By "pharmaceutically acceptable" it is meant the carrier, diluent or excipient must be compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
[0124] In one embodiment, the invention provides for pharmaceutical compositions (or medicaments) comprising a compound of formula I or an embodiment thereof, and its stereoisomers, geometric isomers, tautomers, solvates, metabolites, isotopes, pharmaceutically acceptable salts, or prodrugs thereof) and a pharmaceutically acceptable carrier, diluent or excipient. In another embodiment, the invention provides for preparing compositions (or medicaments) comprising compounds of the invention. In another embodiment, the invention provides for administering compounds of formula I or its embodiments and compositions comprising compounds of formula I or an embodiment thereof to a patient (e.g., a human patient) in need thereof.
[0125] Compositions are formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The effective amount of the compound to be administered will be governed by such considerations, and is the minimum amount necessary to inhibit NaV1.7 activity as required to prevent or treat the undesired disease or disorder, such as for example, pain. For example, such amount may be below the amount that is toxic to normal cells, or the mammal as a whole.
[0126] In one example, the therapeutically effective amount of the compound of the invention administered parenterally per dose will be in the range of about 0.01-100 mg/kg, alternatively about e.g., 0.1 to 20 mg/kg of patient body weight per day, with the typical initial range of compound used being 0.3 to 15 mg/kg/day. The daily does is, in certain embodiments, given as a single daily dose or in divided doses two to six times a day, or in sustained release form. In the case of a 70 kg adult human, the total daily dose will generally be from about 7 mg to about 1,400 mg. This dosage regimen may be adjusted to provide the optimal therapeutic response. The compounds may be administered on a regimen of 1 to 4 times per day, preferably once or twice per day.
[0127] The compounds of the present invention may be administered in any convenient administrative form, e.g., tablets, powders, capsules, solutions, dispersions, suspensions, syrups, sprays, suppositories, gels, emulsions, patches, etc. Such compositions may contain components conventional in pharmaceutical preparations, e.g., diluents, carriers, pH modifiers, sweeteners, bulking agents, and further active agents.
[0128] The compounds of the invention may be administered by any suitable means, including oral, topical (including buccal and sublingual), rectal, vaginal, transdermal, parenteral, subcutaneous, intraperitoneal, intrapulmonary, intradermal, intrathecal and epidural and intranasal, and, if desired for local treatment, intralesional administration. Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, intracerebral, intraocular, intralesional or subcutaneous administration.
[0129] The compositions comprising compounds of formula I or an embodiment thereof are normally formulated in accordance with standard pharmaceutical practice as a pharmaceutical composition. A typical formulation is prepared by mixing a compound of the present invention and a diluent, carrier or excipient. Suitable, diluents, carriers and excipients are well known to those skilled in the art and are described in detail in, e.g., Ansel, Howard C., et al., Ansel's Pharmaceutical Dosage Forms and Drug Delivery Systems. Philadelphia: Lippincott, Williams & Wilkins, 2004; Gennaro, Alfonso R., et al. Remington: The Science and Practice of Pharmacy. Philadelphia: Lippincott, Williams & Wilkins, 2000; and Rowe, Raymond C. Handbook of Pharmaceutical Excipients, Chicago, Pharmaceutical Press, 2005. The formulations may also include one or more buffers, stabilizing agents, surfactants, wetting agents, lubricating agents, emulsifiers, suspending agents, preservatives, antioxidants, opaquing agents, glidants, processing aids, colorants, sweeteners, perfuming agents, flavoring agents, diluents and other known additives to provide an elegant presentation of the drug (i.e., a compound of the present invention or pharmaceutical composition thereof) or aid in the manufacturing of the pharmaceutical product (i.e., medicament).
[0130] Suitable carriers, diluents and excipients are well known to those skilled in the art and include materials such as carbohydrates, waxes, water soluble and/or swellable polymers, hydrophilic or hydrophobic materials, gelatin, oils, solvents, water and the like. The particular carrier, diluent or excipient used will depend upon the means and purpose for which a compound of the present invention is being applied. Solvents are generally selected based on solvents recognized by persons skilled in the art as safe (GRAS) to be administered to a mammal. In general, safe solvents are non-toxic aqueous solvents such as water and other non-toxic solvents that are soluble or miscible in water. Suitable aqueous solvents include water, ethanol, propylene glycol, polyethylene glycols (e.g., PEG 400, PEG 300), etc. and mixtures thereof. The formulations can also include one or more buffers, stabilizing agents, surfactants, wetting agents, lubricating agents, emulsifiers, suspending agents, preservatives, antioxidants, opaquing agents, glidants, processing aids, colorants, sweeteners, perfuming agents, flavoring agents and other known additives to provide an elegant presentation of the drug (i.e., a compound of the present invention or pharmaceutical composition thereof) or aid in the manufacturing of the pharmaceutical product (i.e., medicament).
[0131] Acceptable diluents, carriers, excipients and stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone: amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g., Zn-protein complexes); and/or non-ionic surfactants such as TWEEN.TM., PLURONICS.TM. or polyethylene glycol (PEG). A active pharmaceutical ingredient of the invention (e.g., compound of formula I or an embodiment thereof) can also be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-microcapsules and poly-(methylmethacylate) microcapsules, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions. Such techniques are disclosed in Remington: The Science and Practice of Pharmacy: Remington the Science and Practice of Pharmacy (2005) 21.sup.st Edition, Lippincott Williams & Wilkins, Philadelphia, Pa.
[0132] Sustained-release preparations of a compound of the invention (e.g., compound of formula I or an embodiment thereof) can be prepared. Suitable examples of sustained-release preparations include semipermeable matrices of solid hydrophobic polymers containing a compound of formula I or an embodiment thereof, which matrices are in the form of shaped articles, e.g., films, or microcapsules. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2-hydroxyethyl-methacrylate), or poly(vinyl alcohol)), polylactides (U.S. Pat. No. 3,773,919), copolymers of L-glutamic acid and gamma-ethyl-L-glutamate (Sidman et al., Biopolymers 22:547, 1983), non-degradable ethylene-vinyl acetate (Langer et al., J. Biomed. Mater. Res. 15:167, 1981), degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOT.TM. (injectable microspheres composed of lactic acid glycolic acid copolymer and leuprolide acetate) and poly-D-(-)-3-hydroxybutyric acid (EP 133,988A). Sustained release compositions also include liposomally entrapped compounds, which can be prepared by methods known per se (Epstein et al., Proc. Natl. Acad. Sci. U.S.A. 82:3688, 1985; Hwang et al., Proc. Natl. Acad. Sci. U.S.A. 77:4030, 1980; U.S. Pat. Nos. 4,485,045 and 4,544,345; and EP 102,324A). Ordinarily, the liposomes are of the small (about 200-800 Angstroms) unilamelar type in which the lipid content is greater than about 30 mol % cholesterol, the selected proportion being adjusted for the optimal therapy.
[0133] The formulations include those suitable for the administration routes detailed herein. The formulations can conveniently be presented in unit dosage form and can be prepared by any of the methods well known in the art of pharmacy. Techniques and formulations generally are found in Remington: The Science and Practice of Pharmacy: Remington the Science and Practice of Pharmacy (2005) 21.sup.st Edition, Lippincott Williams & Wilkins, Philadelphia, Pa. Such methods include the step of bringing into association the active ingredient with the carrier which constitutes one or more accessory ingredients.
[0134] In general the formulations are prepared uniformly and intimately bringing into association the active ingredient with liquid carriers, diluents or excipients or finely divided solid carriers, diluents or excipients, or both, and then, if necessary, shaping the product. A typical formulation is prepared by a compound of the present invention and a carrier, diluent or excipient. The formulations can be prepared using conventional dissolution and mixing procedures. For example, the bulk drug substance (i.e., compound of the present invention or stabilized form of the compound (e.g., complex with a cyclodextrin derivative or other known complexation agent) is dissolved in a suitable solvent in the presence of one or more of the excipients described above. A compound of the present invention is typically formulated into pharmaceutical dosage forms to provide an easily controllable dosage of the drug and to enable patient compliance with the prescribed regimen.
[0135] In one example, compounds of formula I or an embodiment thereof may be formulated by mixing at ambient temperature at the appropriate pH, and at the desired degree of purity, with physiologically acceptable carriers, i.e., carriers that are non-toxic to recipients at the dosages and concentrations employed into a galenical administration form. The pH of the formulation depends mainly on the particular use and the concentration of compound, but preferably ranges anywhere from about 3 to about 8. In one example, a compound of formula I (or an embodiment thereof) is formulated in an acetate buffer, at pH 5. In another embodiment, the compounds of formula I or an embodiment thereof are sterile. The compound may be stored, for example, as a solid or amorphous composition, as a lyophilized formulation or as an aqueous solution.
[0136] Formulations of a compound of the invention (e.g., compound of formula I or an embodiment thereof) suitable for oral administration can be prepared as discrete units such as pills, capsules, cachets or tablets each containing a predetermined amount of a compound of the invention.
[0137] Compressed tablets can be prepared by compressing in a suitable machine the active ingredient in a free-flowing form such as a powder or granules, optionally mixed with a binder, lubricant, inert diluent, preservative, surface active or dispersing agent. Molded tablets can be made by molding in a suitable machine a mixture of the powdered active ingredient moistened with an inert liquid diluent. The tablets can optionally be coated or scored and optionally are formulated so as to provide slow or controlled release of the active ingredient therefrom.
[0138] Tablets, troches, lozenges, aqueous or oil suspensions, dispersible powders or granules, emulsions, hard or soft capsules, e.g., gelatin capsules, syrups or elixirs can be prepared for oral use. Formulations of a compound of the invention (e.g., compound of formula I or an embodiment thereof) intended for oral use can be prepared according to any method known to the art for the manufacture of pharmaceutical compositions and such compositions can contain one or more agents including sweetening agents, flavoring agents, coloring agents and preserving agents, in order to provide a palatable preparation. Tablets containing the active ingredient in admixture with non-toxic pharmaceutically acceptable excipient which are suitable for manufacture of tablets are acceptable. These excipients can be, for example, inert diluents, such as calcium or sodium carbonate, lactose, calcium or sodium phosphate; granulating and disintegrating agents, such as maize starch, or alginic acid; binding agents, such as starch, gelatin or acacia; and lubricating agents, such as magnesium stearate, stearic acid or talc. Tablets can be uncoated or can be coated by known techniques including microencapsulation to delay disintegration and adsorption in the gastrointestinal tract and thereby provide a sustained action over a longer period. For example, a time delay material such as glyceryl monostearate or glyceryl distearate alone or with a wax can be employed.
[0139] An example of a suitable oral administration form is a tablet containing about 1 mg, 5 mg, 10 mg, 25 mg, 30 mg, 50 mg, 80 mg, 100 mg, 150 mg, 250 mg, 300 mg and 500 mg of the compound of the invention compounded with about 90-30 mg anhydrous lactose, about 5-40 mg sodium croscarmellose, about 5-30 mg polyvinylpyrrolidone (PVP) K30, and about 1-10 mg magnesium stearate. The powdered ingredients are first mixed together and then mixed with a solution of the PVP. The resulting composition can be dried, granulated, mixed with the magnesium stearate and compressed to tablet form using conventional equipment. An example of an aerosol formulation can be prepared by dissolving the compound, for example 5-400 mg, of the invention in a suitable buffer solution, e.g. a phosphate buffer, adding a tonicifier, e.g. a salt such sodium chloride, if desired. The solution may be filtered, e.g., using a 0.2 micron filter, to remove impurities and contaminants.
[0140] For treatment of the eye or other external tissues, e.g., mouth and skin, the formulations are preferably applied as a topical ointment or cream containing the active ingredient(s) in an amount of, for example, 0.075 to 20% w/w. When formulated in an ointment, the active ingredient can be employed with either a paraffinic or a water-miscible ointment base. Alternatively, the active ingredients can be formulated in a cream with an oil-in-water cream base. If desired, the aqueous phase of the cream base can include a polyhydric alcohol, i.e., an alcohol having two or more hydroxyl groups such as propylene glycol, butane 1,3-diol, mannitol, sorbitol, glycerol and polyethylene glycol (including PEG 400) and mixtures thereof. The topical formulations can desirably include a compound which enhances absorption or penetration of the active ingredient through the skin or other affected areas. Examples of such dermal penetration enhancers include dimethyl sulfoxide and related analogs.
[0141] The oily phase of the emulsions invention can be constituted from known ingredients in a known manner. While the phase can comprise merely an emulsifier, it desirably comprises a mixture of at least one emulsifier with a fat or an oil or with both a fat and an oil. Preferably, a hydrophilic emulsifier is included together with a lipophilic emulsifier which acts as a stabilizer. It is also preferred to include both oil and a fat. Together, the emulsifier(s) with or without stabilizer(s) make up the so-called emulsifying wax, and the wax together with the oil and fat make up the so-called emulsifying ointment base which forms the dispersed phase of the cream formulations. Emulsifiers and emulsion stabilizers suitable for use in the formulation of the invention include Tween.RTM. 60, Span.RTM. 80, cetostearyl alcohol, benzyl alcohol, myristyl alcohol, glyceryl mono-stearate and sodium lauryl sulfate.
[0142] In one aspect of topical applications, it is desired to administer an effective amount of a pharmaceutical composition according to the invention to target area, e.g., skin surfaces, mucous membranes, and the like, which are adjacent to peripheral neurons which are to be treated. This amount will generally range from about 0.0001 mg to about 1 g of a compound of the invention per application, depending upon the area to be treated, whether the use is diagnostic, prophylactic or therapeutic, the severity of the symptoms, and the nature of the topical vehicle employed. A preferred topical preparation is an ointment, wherein about 0.001 to about 50 mg of active ingredient is used per cc of ointment base. The pharmaceutical composition can be formulated as transdermal compositions or transdermal delivery devices ("patches"). Such compositions include, for example, a backing, active compound reservoir, a control membrane, liner and contact adhesive. Such transdermal patches may be used to provide continuous pulsatile, or on demand delivery of the compounds of the present invention as desired.
[0143] Aqueous suspensions of a compound of the invention (e.g., compound of formula I or an embodiment thereof) contain the active materials in admixture with excipients suitable for the manufacture of aqueous suspensions. Such excipients include a suspending agent, such as sodium carboxymethylcellulose, croscarmellose, povidone, methylcellulose, hydroxypropyl methylcellulose, sodium alginate, polyvinylpyrrolidone, gum tragacanth and gum acacia, and dispersing or wetting agents such as a naturally occurring phosphatide (e.g., lecithin), a condensation product of an alkylene oxide with a fatty acid (e.g., polyoxyethylene stearate), a condensation product of ethylene oxide with a long chain aliphatic alcohol (e.g., heptadecaethyleneoxycetanol), a condensation product of ethylene oxide with a partial ester derived from a fatty acid and a hexitol anhydride (e.g., polyoxyethylene sorbitan monooleate). The aqueous suspension can also contain one or more preservatives such as ethyl or n-propyl p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents and one or more sweetening agents, such as sucrose or saccharin.
[0144] Formulations of a compound of the invention (e.g., compound of formula I or an embodiment thereof) can be in the form of a sterile injectable preparation, such as a sterile injectable aqueous or oleaginous suspension. This suspension can be formulated according to the known art using those suitable dispersing or wetting agents and suspending agents which have been mentioned above. The sterile injectable preparation can also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, such as a solution in 1,3-butanediol or prepared as a lyophilized powder. Among the acceptable vehicles and solvents that can be employed are water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile fixed oils can conventionally be employed as a solvent or suspending medium. For this purpose any bland fixed oil can be employed including synthetic mono- or diglycerides. In addition, fatty acids such as oleic acid can likewise be used in the preparation of injectables.
[0145] The amount of active ingredient that can be combined with the carrier material to produce a single dosage form will vary depending upon the host treated and the particular mode of administration. For example, a time-release formulation intended for oral administration to humans can contain approximately 1 to 1000 mg of active material compounded with an appropriate and convenient amount of carrier material which can vary from about 5 to about 95% of the total compositions (weight:weight). The pharmaceutical composition can be prepared to provide easily measurable amounts for administration. For example, an aqueous solution intended for intravenous infusion can contain from about 3 to 500 .mu.g of the active ingredient per milliliter of solution in order that infusion of a suitable volume at a rate of about 30 mL/hr can occur.
[0146] Formulations suitable for parenteral administration include aqueous and non-aqueous sterile injection solutions which can contain anti-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which can include suspending agents and thickening agents.
[0147] Formulations suitable for topical administration to the eye also include eye drops wherein the active ingredient is dissolved or suspended in a suitable carrier, especially an aqueous solvent for the active ingredient. The active ingredient is preferably present in such formulations in a concentration of about 0.5 to 20% w/w, for example about 0.5 to 10% w/w, for example about 1.5% w/w.
[0148] Formulations suitable for topical administration in the mouth include lozenges comprising the active ingredient in a flavored basis, usually sucrose and acacia or tragacanth; pastilles comprising the active ingredient in an inert basis such as gelatin and glycerin, or sucrose and acacia; and mouthwashes comprising the active ingredient in a suitable liquid carrier.
[0149] Formulations for rectal administration can be presented as a suppository with a suitable base comprising for example cocoa butter or a salicylate.
[0150] Formulations suitable for intrapulmonary or nasal administration have a particle size for example in the range of 0.1 to 500 microns (including particle sizes in a range between 0.1 and 500 microns in increments microns such as 0.5, 1, 30 microns, 35 microns, etc.), which is administered by rapid inhalation through the nasal passage or by inhalation through the mouth so as to reach the alveolar sacs. Suitable formulations include aqueous or oily solutions of the active ingredient. Formulations suitable for aerosol or dry powder administration can be prepared according to conventional methods and can be delivered with other therapeutic agents such as compounds heretofore used in the treatment of disorders as described below.
[0151] The formulations can be packaged in unit-dose or multi-dose containers, for example sealed ampoules and vials, and can be stored in a freeze-dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example water, for injection immediately prior to use. Extemporaneous injection solutions and suspensions are prepared from sterile powders, granules and tablets of the kind previously described. Preferred unit dosage formulations are those containing a daily dose or unit daily sub-dose, as herein above recited, or an appropriate fraction thereof, of the active ingredient.
[0152] When the binding target is located in the brain, certain embodiments of the invention provide for a compound of formula I (or an embodiment thereof) to traverse the blood-brain barrier. Certain neurodegenerative diseases are associated with an increase in permeability of the blood-brain barrier, such that a compound of formula I (or an embodiment thereof) can be readily introduced to the brain. When the blood-brain barrier remains intact, several art-known approaches exist for transporting molecules across it, including, but not limited to, physical methods, lipid-based methods, and receptor and channel-based methods.
[0153] Physical methods of transporting a compound of formula I (or an embodiment thereof) across the blood-brain barrier include, but are not limited to, circumventing the blood-brain barrier entirely, or by creating openings in the blood-brain barrier.
[0154] Circumvention methods include, but are not limited to, direct injection into the brain (see, e.g., Papanastassiou et al., Gene Therapy 9:308-406, 2000, interstitial infusion/convection-enhanced delivery (see, e.g., Bobo et al., Proc. Natl. Acad. Sci. U.S.A. 91 :2076-2080, 1994), and implanting a delivery device in the brain (see, e.g., Gill et al., Nature Med, 9:589-595, 2003; and Gliadel Wafers.TM., Guildford.
[0155] Pharmaceutical). Methods of creating openings in the barrier include, but are not limited to, ultrasound (see, e.g., U.S. Patent Publication No. 2002/0038086), osmotic pressure (e.g., by administration of hypertonic mannitol (Neuwelt, E. A., Implication of the Blood-Brain Barrier and its Manipulation, Volumes 1 and 2, Plenum Press, N.Y., 1989)), and permeabilization by, e.g., bradykinin or permeabilizer A-7 (see, e.g., U.S. Pat. Nos. 5,112,596, 5,268,164, 5,506,206, and 5,686,416).
[0156] Lipid-based methods of transporting a compound of formula I (or an embodiment thereof) across the blood-brain barrier include, but are not limited to, encapsulating the a compound of formula I (or an embodiment thereof) in liposomes that are coupled to antibody binding fragments that bind to receptors on the vascular endothelium of the blood-brain barrier (see, e.g., U.S. Patent Application Publication No. 2002/0025313), and coating a compound of formula I (or an embodiment thereof) in low-density lipoprotein particles (see, e.g., U.S. Patent Application Publication No. 2004/0204354) or apolipoprotein E (see, e.g., U.S. Patent Application Publication No. 2004/0131692).
[0157] Receptor and channel-based methods of transporting a compound of formula I (or an embodiment thereof) across the blood-brain barrier include, but are not limited to, using glucocorticoid blockers to increase permeability of the blood-brain barrier (see, e.g., U.S. Patent Application Publication Nos. 2002/0065259, 2003/0162695, and 2005/0124533); activating potassium channels (see, e.g., U.S. Patent Application Publication No. 2005/0089473), inhibiting ABC drug transporters (see, U.S. Patent Application Publication No. 2003/0073713); coating compound of formula I (or an embodiment thereof) with a transferrin and modulating activity of the one or more transferrin receptors (see, e.g., U.S. Patent Application Publication No. 2003/0129186), and cationizing the antibodies (see, e.g., U.S. Pat. No. 5,004,697).
[0158] For intracerebral use, in certain embodiments, the compounds can be administered continuously by infusion into the fluid reservoirs of the CNS, although bolus injection may be acceptable. The inhibitors can be administered into the ventricles of the brain or otherwise introduced into the CNS or spinal fluid. Administration can be performed by use of an indwelling catheter and a continuous administration means such as a pump, or it can be administered by implantation, e.g., intracerebral implantation of a sustained release vehicle. More specifically, the inhibitors can be injected through chronically implanted cannulas or chronically infused with the help of osmotic minipumps. Subcutaneous pumps are available that deliver proteins through a small tubing to the cerebral ventricles. Highly sophisticated pumps can be refilled through the skin and their delivery rate can be set without surgical intervention. Examples of suitable administration protocols and delivery systems involving a subcutaneous pump device or continuous intracerebroventricular infusion through a totally implanted drug delivery system are those used for the administration of dopamine, dopamine agonists, and cholinergic agonists to Alzheimer's disease patients and animal models for Parkinson's disease, as described by Harbaugh, J. Neural Transm. Suppl. 24:271, 1987; and DeYebenes et al., Mov. Disord. 2: 143, 1987.
[0159] A compound of formula I (or an embodiment thereof) used in the invention are formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. A compound of formula I (or an embodiment thereof) need not be, but is optionally formulated with one or more agent currently used to prevent or treat the disorder in question. The effective amount of such other agents depends on the amount of a compound of the invention present in the formulation, the type of disorder or treatment, and other factors discussed above.
[0160] These are generally used in the same dosages and with administration routes as described herein, or about from 1 to 99% of the dosages described herein, or in any dosage and by any route that is empirically/clinically determined to be appropriate.
[0161] For the prevention or treatment of disease, the appropriate dosage of a compound of formula I (or an embodiment thereof) (when used alone or in combination with other agents) will depend on the type of disease to be treated, the properties of the compound, the severity and course of the disease, whether the compound is administered for preventative or therapeutic purposes, previous therapy, the patient's clinical history and response to the compound, and the discretion of the attending physician. The compound is suitably administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 .mu.g/kg to 15 mg/kg (e.g., 0.1 mg/kg-10 mg/kg) of compound can be an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion. One typical daily dosage might range from about 1 .mu.g kg to 100 mg/kg or more, depending on the factors mentioned above. For repeated administrations over several days or longer, depending on the condition, the treatment would generally be sustained until a desired suppression of disease symptoms occurs. One exemplary dosage of a compound of formula I (or an embodiment thereof) would be in the range from about 0.05 mg/kg to about 10 mg/kg. Thus, one or more doses of about 0.5 mg/kg, 2.0 mg/kg, 4.0 mg/kg, or 10 mg/kg (or any combination thereof) may be administered to the patient. Such doses may be administered intermittently, e.g., every week or every three weeks (e.g., such that the patient receives from about two to about twenty, or, e.g., about six doses of the antibody). An initial higher loading dose, followed by one or more lower doses may be administered. An exemplary dosing regimen comprises administering an initial loading dose of about 4 mg/kg, followed by a weekly maintenance dose of about 2 mg kg of the compound. However, other dosage regimens ma be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
[0162] Other typical daily dosages might range from, for example, about 1 g/kg to up to 100 mg/kg or more (e.g., about 1 .mu.g kg to 1 mg/kg, about 1 .mu.g/kg to about 5 mg/kg, about 1 mg kg to 10 mg/kg, about 5 mg/kg to about 200 mg/kg, about 50 mg/kg to about 150 mg/mg, about 100 mg/kg to about 500 mg/kg, about 100 mg/kg to about 400 mg/kg, and about 200 mg/kg to about 400 mg/kg), depending on the factors mentioned above. Typically, the clinician will administer a compound until a dosage is reached that results in improvement in or, optimally, elimination of, one or more symptoms of the treated disease or condition. The progress of this therapy is easily monitored by conventional assays. One or more agent provided herein may be administered together or at different times (e.g., one agent is administered prior to the administration of a second agent). One or more agent may be administered to a subject using different techniques (e.g., one agent may be administered orally, while a second agent is administered via intramuscular injection or intranasally). One or more agent may be administered such that the one or more agent as a pharmacologic effect in a subject at the same time. Alternatively, one or more agent may be administered, such that the pharmacological activity of the first administered agent is expired prior the administration of one or more secondarily administered agents (e.g., 1, 2, 3, or 4 secondarily administered agents).
Indications and Methods of Treatment
[0163] The compounds of the invention modulate, preferably inhibit, ion flux through a voltage-dependent sodium channel in a mammal, (e.g. a human). Any such modulation, whether it be partial or complete inhibition or prevention of ion flux, is sometimes referred to herein as "blocking" and corresponding compounds as "blockers" or "inhibitors". In general, the compounds of the invention modulate the activity of a sodium channel downwards by inhibiting the voltage-dependent activity of the sodium channel, and/or reduce or prevent sodium ion flux across a cell membrane by preventing sodium channel activity such as ion flux.
[0164] The compounds of the invention inhibit the ion flux through a voltage-dependent sodium channel. In one aspect, the compounds are state or frequency dependent modifers of the sodium channels, having a low affinity for the rested/closed state and a high affinity for the inactivated state. Without being bound by any particular theory, it is thought that these compounds are likely to interact with overlapping sites located in the inner cavity of the sodium conducting pore of the channel similar to that described for other state-dependent sodium channel blockers (Cestele, S., et al., op. cit.). These compounds may also be likely to interact with sites outside of the inner cavity and have allosteric effects on sodium ion conduction through the channel pore.
[0165] Any of these consequences may ultimately be responsible for the overall therapeutic benefit provided by these compounds.
[0166] Accordingly, the compounds of the invention are sodium channel blockers and are therefore useful for treating diseases and conditions in mammals, for example humans, and other organisms, including all those diseases and conditions which are the result of aberrant voltage-dependent sodium channel biological activity or which may be ameliorated by modulation of voltage-dependent sodium channel biological activity. In particular, the compounds of the invention, i.e., the compounds of formula (I) and embodiments and (or stereoisomers, geometric isomers, tautomers, solvates, metabolites, isotopes, pharmaceutically acceptable salts, or prodrugs thereof), are useful for treating diseases and conditions in mammals, for example humans, which are the result of aberrant voltage-dependent NaV1.7 biological activity or which may be ameliorated by the modulation, preferably the inhibition, of NaV1.7 biological activity. In certain aspects, the compounds of the invention selectively NaV1.7 over NaV1.5.
[0167] As defined herein, a sodium channel-mediated disease or condition refers to a disease or condition in a mammal, preferably a human, which is ameliorated upon modulation of the sodium channel and includes, but is not limited to, pain, central nervous conditions such as epilepsy, anxiety, depression and bipolar disease; cardiovascular conditions such as arrhythmias, atrial fibrillation and ventricular fibrillation; neuromuscular conditions such as restless leg syndrome and muscle paralysis or tetanus; neuroprotection against stroke, neural trauma and multiple sclerosis; and channelopathies such as erythromyalgia and familial rectal pain syndrome.
[0168] In one aspect, the present invention relates to compounds, pharmaceutical compositions and methods of using the compounds and pharmaceutical compositions for the treatment of sodium channel-mediated diseases in mammals, preferably humans and preferably diseases and conditions related to pain, central nervous conditions such as epilepsy, anxiety, depression and bipolar disease; cardiovascular conditions such as arrhythmias, atrial fibrillation and ventricular fibrillation; neuromuscular conditions such as restless leg syndrome and muscle paralysis or tetanus; neuroprotection against stroke, neural trauma and multiple sclerosis; and channelopathies such as erythromyalgia and familial rectal pain syndrome, by administering to a mammal, for example a human, in need of such treatment an effective amount of a sodium channel blocker modulating, especially inhibiting, agent.
[0169] A sodium channel-mediated disease or condition also includes pain associated with HIV, HIV treatment induced neuropathy, trigeminal neuralgia, glossopharyngeal neuralgia, neuropathy secondary to metastatic infiltration, adiposis dolorosa, thalamic lesions, hypertension, antoimmune disease, asthma, drug addiction (e.g., opiate, benzodiazepine, amphetamine, cocaine, alcohol, butane inhalation), Alzheimer, dementia, age-related memory impairment, Korsakoff syndrome, restenosis, urinary dysfunction, incontinence, Parkinson's disease, cerebrovascular ischemia, neurosis, gastrointestinal disease, sickle cell anemia, transplant rejection, heart failure, myocardial infarction, reperfusion injury, intermittant claudication, angina, convulsion, respiratory disorders, cerebral or myocardial ischemias, long-QT syndrome, Catecholeminergic polymorphic ventricular tachycardia, ophthalmic diseases, spasticity, spastic paraplegia, myopathies, myasthenia gravis, paramyotonia congentia, hyperkalemic periodic paralysis, hypokalemic periodic paralysis, alopecia, anxiety disorders, psychotic disorders, mania, paranoia, seasonal affective disorder, panic disorder, obsessive compulsive disorder (OCD), phobias, autism, Aspergers Syndrome, Retts syndrome, disintegrative disorder, attention deficit disorder, aggressivity, impulse control disorders, thrombosis, pre clampsia, congestive cardiac failure, cardiac arrest, Freidrich's ataxia, Spinocerebellear ataxia, myelopathy, radiculopathy, systemic lupus erythamatosis, granulomatous disease, olivo-ponto-cerebellar atrophy, spinocerebellar ataxia, episodic ataxia, myokymia, progressive pallidal atrophy, progressive supranuclear palsy and spasticity, traumatic brain injury, cerebral oedema, hydrocephalus injury, spinal cord injury, anorexia nervosa, bulimia, Prader-Willi syndrome, obesity, optic neuritis, cataract, retinal haemorrhage, ischaemic retinopathy, retinitis pigmentosa, acute and chronic glaucoma, macular degeneration, retinal artery occlusion, Chorea, Huntington's chorea, cerebral edema, proctitis, post-herpetic neuralgia, eudynia, heat sensitivity, sarcoidosis irritable bowel syndrome, Tourette syndrome, Lesch-Nyhan Syndrome, Brugado syndrome, Liddle syndrome, Crohns disease, multiple sclerosis and the pain associated with multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), disseminated sclerosis, diabetic neuropathy, peripheral neuropathy, charcot marie tooth syndrome, arthritic, rheumatoid arthritis, osteoarthritis, chondrocalcinosis, atherosclerosis, paroxysmal dystonia, myasthenia syndromes, myotonia, myotonic dystrophy, muscular dystrophy, malignant hyperthermia, cystic fibrosis, pseudoaldosteronism, rhabdomyolysis, mental handicap, hypothyroidism, bipolar depression, anxiety, schizophrenia, sodium channel toxin related illnesses, familial erythromelalgia, primary erythromelalgia, rectal pain, cancer, epilepsy, partial and general tonic seizures, febrile seizures, absence seizures (petit mal), myoclonic seizures, atonic seizures, clonic seizures, Lennox Gastaut, West Syndome (infantile spasms), multiresistant seizures, seizure prophylaxis (anti-epileptogenic), familial Mediterranean fever syndrome, gout, restless leg syndrome, arrhythmias, fibromyalgia, neuroprotection under ischaemic conditions caused by stroke or neural trauma, tachy-arrhythmias, atrial fibrillation and ventricular fibrillation and as a general or local anaesthetic.
[0170] As used herein, the term "pain" refers to all categories of pain and is recognized to include, but is not limited to, neuropathic pain, inflammatory pain, nociceptive pain, idiopathic pain, neuralgic pain, orafacial pain, burn pain, burning mouth syndrome, somatic pain, visceral pain, myofacial pain, dental pain, cancer pain, chemotherapy pain, trauma pain, surgical pain, post-surgical pain, childbirth pain, labor pain, chronic regional pain syndrome (CRPS), reflex sympathetic dystrophy, brachial plexus avulsion, neurogenic bladder, acute pain (e.g., musculoskeletal and post-operative pain), chronic pain, persistent pain, peripherally mediated pain, centrally mediated pain, chronic headache, migraine headache, familial hemiplegic migraine, conditions associated with cephalic pain, sinus headache, tension headache, phantom limb pain, peripheral nerve injury, pain following stroke, thalamic lesions, radiculopathy, HIV pain, post-herpetic pain, non-cardiac chest pain, irritable bowel syndrome and pain associated with bowel disorders and dyspepsia, and combinations thereof.
[0171] Furthermore, sodium channel blockers have clinical uses in addition to pain. The present invention therefore also relates to compounds, pharmaceutical compositions and methods of using the compounds and pharmaceutical compositions for the treatment of diseases or conditions such as cancer and pruritus (itch).
[0172] Pruritus, commonly known as itch, is a common dermatological condition. While the exact causes of pruritus are complex and incompletely understood, there has long been evidence that itch involves sensory neurons, especially C fibers, similar to those that mediate pain (Schmelz, M., et al., Neurosci. (1997), 17: 8003-8). In particular, it is believed that sodium influx through voltage-gated sodium channels is essential for the propagation of itch sensation from the skin. Transmission of the itch impulses results in the unpleasant sensation that elicits the desire or reflex to scratch.
[0173] Multiple causes and electrical pathways for eliciting itch are known. In humans, pruritis can be elicited by histamine or PAR-2 agonists such as mucunain that activate distinct populations of C fibers (Namer, B., et al., J. Neurophysiol. (2008),100: 2062-9). A variety of neurotrophic peptides are known to mediate itch in animal models (Wang, H., and Yosipovitch, G., International Journal of Dermatology (2010), 49: 1-11). Itch can also be elicited by opioids, evidence of distinct pharmacology from that of pain responses.
[0174] There exists a complex interaction between itch and pain responses that arises in part from the overlapping sensory input from the skin (Ikoma, A., et al., Arch. Dermatol. (2003),139: 1475-8) and also from the diverse etiology of both pain and pruritis. Pain responses can exacerbate itching by enhancing central sensitization or lead to inhibition of painful scratching. Particularly severe forms of chronic itch occur when pain responses are absent, as in the case of post-herpetic itch (Oaklander, A. L., et al., Pain (2002), 96: 9-12).
[0175] The compounds of the invention can also be useful the treating pruritus. The rationale for treating itch with inhibitors of voltage-gated sodium channels, especially NaV1.7, is as follows:
[0176] The propagation of electrical activity in the C fibers that sense pruritinergic stimulants requires sodium entry through voltage-gated sodium channels.
[0177] NaV1.7 is expressed in the C fibers and kerotinocytes in human skin (Zhao, P., et al., Pain (2008), 139: 90-105).
[0178] A gain of function mutation of NaV1.7 (L858F) that causes erythromelalgia also causes chronic itch (Li, Y., et al., Clinical and Experimental Dermatology (2009), 34: e313-e4).
[0179] Chronic itch can be alleviated with treatment by sodium channel blockers, such as the local anesthetic lidocaine (Oaklander, A. L., et al., Pain (2002), 96: 9-12; Villamil, A. G., et al. The American Journal of Medicine (2005), 118: 1160-3). In these reports, lidocaine was effective when administered either intravenously or topically (a Lidoderm patch). Lidocaine can have multiple activities at the plasma concentrations achieved when administered systemically, but when administered topically, the plasma concentrations are only about 1 .mu.M (Center for Drug Evaluation and Research NDA 20-612). At these concentrations, lidocaine is selective for sodium channel block and inhibits spontaneous electrical activity in C fibers and pain responses in animal models (Xiao, W. H., and Bennett, G. J., Pain (2008), 137: 218-28). The types of itch or skin irritation, include, but are not limited to:
[0180] psoriatic pruritus, itch due to hemodyalisis, aguagenic pruritus, and itching caused by skin disorders (e.g., contact dermatitis), systemic disorders, neuropathy, psychogenic factors or a mixture thereof:
[0181] itch caused by allergic reactions, insect bites, hypersensitivity (e.g., dry skin, acne, eczema, psoriasis), inflammatory conditions or injury;
[0182] itch associated with vulvar vestibulitis; and
[0183] skin irritation or inflammatory effect from administration of another therapeutic such as, for example, antibiotics, antivirals and antihistamines.
[0184] The compounds of the invention are also useful in treating certain cancers, such as hormone sensitive cancers, such as prostate cancer (adenocarcinoma), breast cancer, ovarian cancer, testicular cancer and thyroid neoplasia, in a mammal, preferably a human. The voltage gated sodium channels have been demonstrated to be expressed in prostate and breast cancer cells. Up-regulation of neonatal NaV1.5 occurs as an integral part of the metastatic process in human breast cancer and could serve both as a novel marker of the metastatic phenotype and a therapeutic target (Clin. Cancer Res. (2005), August 1; 11(15): 5381-9). Functional expression of voltage-gated sodium channel alpha-subunits, specifically NaV1.7, is associated with strong metastatic potential in prostate cancer (CaP) in vitro. Voltage-gated sodium channel alpha-subunits immunostaining, using antibodies specific to the sodium channel alpha subunit was evident in prostatic tissues and markedly stronger in CaP vs non-CaP patients (Prostate Cancer Prostatic Dis., 2005; 8(3): 266-73). See also Diss, J. K. J., et al., Mol. Cell. Neurosci. (2008), 37:537-547 and Kis-Toth, K., et al., The Journal of Immunology (2011), 187:1273-1280.
[0185] In consideration of the above, in one embodiment, the present invention provides a method for treating a mammal for, or protecting a mammal from developing, a sodium channel-mediated disease, especially pain, comprising administering to the mammal, especially a human, in need thereof, a therapeutically effective amount of a compound of the invention or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention wherein the compound modulates the activity of one or more voltage-dependent sodium channels.
[0186] In another embodiment of the invention is a method of treating a disease or a condition in a mammal, preferably a human, wherein the disease or condition is selected from the group consisting of pain, depression, cardiovascular diseases, respiratory diseases, and psychiatric diseases, and combinations thereof, and wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of an embodiment of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, and a pharmaceutically acceptable excipient.
[0187] One embodiment of this embodiment is wherein the disease or condition is selected from the group consisting of neuropathic pain, inflammatory pain, visceral pain, cancer pain, chemotherapy pain, trauma pain, surgical pain, post surgical pain, childbirth pain, labor pain, neurogenic bladder, ulcerative colitis, chronic pain, persistent pan, peripherally mediated pain, centrally mediated pain, chronic headache, migraine headache, sinus headache, tension headache, phantom limb pain, peripheral nerve injury, and combinations thereof.
[0188] Another embodiment of this embodiment is wherein the disease or condition is selected from the group consisting of pain associated with HIV, HIV treatment induced neuropathy, trigeminal neuralgia, post herpetic neuralgia, eudynia, heat sensitivity, tosarcoidosis, irritable bowel syndrome, Crohns disease, pain associated with multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), diabetic neuropathy, peripheral neuropathy, arthritic, rheumatoid arthritis, osteoarthritis, atherosclerosis, paroxysmal dystonia, myasthenia syndromes, myotonia, malignant hyperthermia, cystic fibrosis, pseudoaldosteronism, rhabdomyolysis, hypothyroidism, bipolar depression, anxiety, schizophrenia, sodium channel toxin related illnesses, familial erythromelalgia, primary erythromelalgia, familial rectal pain, cancer, epilepsy, partial and general tonic seizures, restless leg syndrome, arrhythmias, fibromyalgia, neuroprotection under ischaemic conditions caused by stroke or neural trauma, tachy arrhythmias, atrial fibrillation and ventricular fibrillation.
[0189] Another embodiment of the invention is a method of treating, but not preventing, pain in a mammal, wherein the method comprises administering to the mammal in need thereof therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, and a pharmaceutically acceptable excipient.
[0190] One embodiment of this embodiment is a method wherein the pain is selected from the group consisting of neuropathic pain, inflammatory pain, visceral pain, cancer pain, chemotherapy pain, trauma pain, surgical pain, post surgical pain, childbirth pain, labor pain, dental pain, chronic pain, persistent pain, peripherally mediated pain, centrally mediated pain, chronic headache, migraine headache, sinus headache, tension headache, phantom limb pain, peripheral nerve injury, trigeminal neuralgia, post herpetic neuralgia, eudynia, familial erythromelalgia, primary erythromelalgia, familial rectal pain or fibromyalgia, and combinations thereof.
[0191] Another embodiment of this embodiment is a method wherein the pain is associated with a disease or condition selected from HIV, HIV treatment induced neuropathy, heat sensitivity, tosarcoidosis, irritable bowel syndrome, Crohns disease, multiple sclerosis, amyotrophic lateral sclerosis, diabetic neuropathy, peripheral neuropathy, rheumatoid arthritis, osteoarthritis, atherosclerosis, paroxysmal dystonia, myasthenia syndromes, myotonia, malignant hyperthermia, cystic fibrosis, pseudoaldosteronism, rhabdomyolysis, hypothyroidism, bipolar depression, anxiety, schizophrenia, sodium channel toxin related illnesses, neurogenic bladder, ulcerative colitis, cancer, epilepsy, partial and general tonic seizures, restless leg syndrome, arrhythmias, ischaemic conditions caused by stroke or neural trauma, tachy arrhythmias, atrial fibrillation and ventricular fibrillation.
[0192] Another embodiment of the invention is the method of treating pain in a mammal, preferably a human, by the inhibition of flux through a voltage dependent sodium channel in the mammal, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of an embodiment of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, and a pharmaceutically acceptable excipient.
[0193] Another embodiment of the invention is the method of treating pruritus in a mammal, preferably a human, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of an embodiment of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, and a pharmaceutically acceptable excipient.
[0194] Another embodiment of the invention is the method of treating cancer in a mammal, preferably a human, wherein the method comprises administering to the mammal in need thereof a therapeutically effective amount of an embodiment of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, or a pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof, and a pharmaceutically acceptable excipient.
[0195] Another embodiment of the invention is the method of decreasing ion flux through a voltage dependent sodium channel in a cell in a mammal, wherein the method comprises contacting the cell with an embodiment of a compound of the invention, as set forth above, as a stereoisomer, enantiomer or tautomer thereof or mixtures thereof, or a pharmaceutically acceptable salt, solvate or prodrug thereof.
[0196] Another embodiment of the invention is the method of selectively inhibiting a first voltage-gated sodium channel over a second voltage-gated sodium channel in a mammal, wherein the method comprises administering to the mammal an inhibitory amount of a compound of formula (I), or an embodiment of a compound of formula (I).
[0197] Another embodiment of the invention is the method of selectively inhibiting NaV1.7 in a mammal or a mammalian cell as compared to NaV1.5, wherein the method comprises administering to the mammal in need thereof an inhibitory amount of a compound of formula (I) or an embodiment of an embodiment thereof.
[0198] For each of the above embodiments described related to treating diseases and conditions in a mammal, the present invention also contemplates relatedly a compound of formula I or an embodiment thereof for the use as a medicament in the treatment of such diseases and conditions.
[0199] For each of the above embodiments described related to treating diseases and conditions in a mammal, the present invention also contemplates relatedly the use of a compound of formula I or an embodiment thereof for the manufacture of a medicament for the treatment of such diseases and conditions.
[0200] Another embodiment of the invention is a method of using the compounds of formula (I) as standards or controls in in vitro or in vivo assays in determining the efficacy of test compounds in modulating voltage-dependent sodium channels.
[0201] In another embodiment of the invention, the compounds of formula (I) are isotopically-labeled by having one or more atoms therein replaced by an atom having a different atomic mass or mass number. Such isotopically-labeled (i.e., radiolabelled) compounds of formula (I) are considered to be within the scope of this invention. Examples of isotopes that can be incorporated into the compounds of formula (I) include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, fluorine, chlorine, and iodine, such as, but not limited to, .sup.2H, .sup.3H, .sup.11C, .sup.13C, .sup.14C, .sup.13N, .sup.15N, .sup.15O, .sup.17O, .sup.18O, .sup.31P, .sup.32P, .sup.35S, .sup.18F, .sup.36Cl, .sup.123I, and .sup.125I, respectively. These isotopically-labeled compounds would be useful to help determine or measure the effectiveness of the compounds, by characterizing, for example, the site or mode of action on the sodium channels, or binding affinity to pharmacologically important site of action on the sodium channels, particularly NaV1.7. Certain isotopically-labeled compounds of formula (I), for example, those incorporating a radioactive isotope, are useful in drug and/or substrate tissue distribution studies. The radioactive isotopes tritium, i.e. .sup.3H, and carbon-14, i.e., .sup.14C, are particularly useful for this purpose in view of their ease of incorporation and ready means of detection.
[0202] Substitution with heavier isotopes such as deuterium, i.e. .sup.2H, may afford certain therapeutic advantages resulting from greater metabolic stability, for example, increased in vivo half-life or reduced dosage requirements, and hence may be preferred in some circumstances.
[0203] Substitution with positron emitting isotopes, such as .sup.11C, .sup.18F, .sup.15O and .sup.13N, can be useful in Positron Emission Topography (PET) studies for examining substrate receptor occupancy. Isotopically-labeled compounds of formula (I) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the Examples as set out below using an appropriate isotopically-labeled reagent in place of the non-labeled reagent previously employed.
Testing Compounds
[0204] The assessment of the compounds of the invention in mediating, especially inhibiting, the sodium channel ion flux can be determined using the assays described hereinbelow. Alternatively, the assessment of the compounds in treating conditions and diseases in humans may be established in industry standard animal models for demonstrating the efficacy of compounds in treating pain. Animal models of human neuropathic pain conditions have been developed that result in reproducible sensory deficits (allodynia, hyperalgesia, and spontaneous pain) over a sustained period of time that can be evaluated by sensory testing. By establishing the degree of mechanical, chemical, and temperature induced allodynia and hyperalgesia present, several physiopathological conditions observed in humans can be modeled allowing the evaluation of pharmacotherapies.
[0205] In rat models of peripheral nerve injury, ectopic activity in the injured nerve corresponds to the behavioural signs of pain. In these models, intravenous application of the sodium channel blocker and local anesthetic lidocaine can suppress the ectopic activity and reverse the tactile allodynia at concentrations that do not affect general behaviour and motor function (Mao, J. and Chen, L. L., Pain (2000), 87:7-17). Allometric scaling of the doses effective in these rat models, translates into doses similar to those shown to be efficacious in humans (Tanelian, D. L. and Brose, W. G., Anesthesiology (1991), 74(5):949-951). Furthermore, Lidoderm.RTM., lidocaine applied in the form of a dermal patch, is currently an FDA approved treatment for post-herpetic neuralgia (Devers, A. and Glaler, B. S., Clin. J. Pain (2000), 16(3):205-8).
[0206] The present invention readily affords many different means for identification of sodium channel modulating agents that are useful as therapeutic agents. Identification of modulators of sodium channel can be assessed using a variety of in vitro and in vivo assays, e.g., measuring current, measuring membrane potential, measuring ion flux, (e.g., sodium or guanidinium), measuring sodium concentration, measuring second messengers and transcription levels, and using e.g., voltage-sensitive dyes, radioactive tracers, and patch-clamp electrophysiology.
[0207] One such protocol involves the screening of chemical agents for ability to modulate the activity of a sodium channel thereby identifying it as a modulating agent.
[0208] A typical assay described in Bean et al., J. General Physiology (1983), 83:613-642, and Leuwer, M., et al., Br. J. Pharmacol (2004), 141(1):47-54, uses patch-clamp techniques to study the behaviour of channels. Such techniques are known to those skilled in the art, and may be developed, using current technologies, into low or medium throughput assays for evaluating compounds for their ability to modulate sodium channel behaviour.
[0209] Throughput of test compounds is an important consideration in the choice of screening assay to be used. In some strategies, where hundreds of thousands of compounds are to be tested, it is not desirable to use low throughput means. In other cases, however, low throughput is satisfactory to identify important differences between a limited number of compounds. Often it will be necessary to combine assay types to identify specific sodium channel modulating compounds.
[0210] Electrophysiological assays using patch clamp techniques is accepted as a gold standard for detailed characterization of sodium channel compound interactions, and as described in Bean et al., op. cit. and Leuwer, M., et al., op. cit. There is a manual low-throughput screening (LTS) method which can compare 2-10 compounds per day; a recently developed system for automated medium-throughput screening (MTS) at 20-50 patches (i.e. compounds) per day; and a technology from Molecular Devices Corporation (Sunnyvale, Calif.) which permits automated high-throughput screening (HTS) at 1000-3000 patches (i.e. compounds) per day.
[0211] One automated patch-clamp system utilizes planar electrode technology to accelerate the rate of drug discovery. Planar electrodes are capable of achieving high-resistance, cells-attached seals followed by stable, low-noise whole-cell recordings that are comparable to conventional recordings. A suitable instrument is the PatchXpress 7000A (Axon Instruments Inc, Union City, Calif.). A variety of cell lines and culture techniques, which include adherent cells as well as cells growing spontaneously in suspension are ranked for seal success rate and stability. Immortalized cells (e.g. HEK and CHO) stably expressing high levels of the relevant sodium ion channel can be adapted into high-density suspension cultures.
[0212] Other assays can be selected which allow the investigator to identify compounds which block specific states of the channel, such as the open state, closed state or the resting state, or which block transition from open to closed, closed to resting or resting to open. Those skilled in the art are generally familiar with such assays.
[0213] Binding assays are also available. Designs include traditional radioactive filter based binding assays or the confocal based fluorescent system available from Evotec OAI group of companies (Hamburg, Germany), both of which are HTS.
[0214] Radioactive flux assays can also be used. In this assay, channels are stimulated to open with veratridine or aconitine and held in a stabilized open state with a toxin, and channel blockers are identified by their ability to prevent ion influx. The assay can use radioactive 22[Na] and 14[C] guanidinium ions as tracers. FlashPlate & Cytostar-T plates in living cells avoids separation steps and are suitable for HTS. Scintillation plate technology has also advanced this method to HTS suitability. Because of the functional aspects of the assay, the information content is reasonably good.
[0215] Yet another format measures the redistribution of membrane potential using the FLIPR system membrane potential kit (HTS) available from Molecular Dynamics (a division of Amersham Biosciences, Piscataway, N.J.). This method is limited to slow membrane potential changes. Some problems may result from the fluorescent background of compounds. Test compounds may also directly influence the fluidity of the cell membrane and lead to an increase in intracellular dye concentrations. Still, because of the functional aspects of the assay, the information content is reasonably good.
[0216] Sodium dyes can be used to measure the rate or amount of sodium ion influx through a channel. This type of assay provides a very high information content regarding potential channel blockers. The assay is functional and would measure Na+ influx directly. CoroNa Red, SBFI and/or sodium green (Molecular Probes, Inc. Eugene Oreg.) can be used to measure Na influx; all are Na responsive dyes. They can be used in combination with the FLIPR instrument. The use of these dyes in a screen has not been previously described in the literature. Calcium dyes may also have potential in this format.
[0217] In a another assay, FRET based voltage sensors are used to measure the ability of a test compound to directly block Na influx. Commercially available HTS systems include the VIPR.TM. II FRET system (Life Technologies, or Aurora Biosciences Corporation, San Diego, Calif., a division of Vertex Pharmaceuticals, Inc.) which may be used in conjunction with FRET dyes, also available from Aurora Biosciences. This assay measures sub-second responses to voltage changes. There is no requirement for a modifier of channel function. The assay measures depolarization and hyperpolarizations, and provides ratiometric outputs for quantification. A somewhat less expensive MTS version of this assay employs the FLEXstation.TM. (Molecular Devices Corporation) in conjunction with FRET dyes from Aurora Biosciences. Other methods of testing the compounds disclosed herein are also readily known and available to those skilled in the art.
[0218] Modulating agents so identified are then tested in a variety of in vivo models so as to determine if they alleviate pain, especially chronic pain or other conditions such as cancer and pruritus (itch) with minimal adverse events. The assays described below in the Biological Assays Section are useful in assessing the biological activity of the instant compounds.
[0219] Typically, the efficacy of a compound of the invention is expressed by its IC50 value ("Inhibitory Concentration--50%"), which is the measure of the amount of compound required to achieve 50% inhibition of the activity of the target sodium channel over a specific time period. For example, representative compounds of the present invention have demonstrated IC50's ranging from less than 100 nanomolar to less than 10 micromolar in the patch voltage clamp NaV1.7 electrophysiology assay described herein.
[0220] In another aspect of the invention, the compounds of the invention can be used in in vitro or in vivo studies as exemplary agents for comparative purposes to find other compounds also useful in treatment of, or protection from, the various diseases disclosed herein.
[0221] Another aspect of the invention relates to inhibiting NaV1.1, NaV1.2, NaV1.3, NaV1.4, NaV1.5, NaV1.6, NaV1.7, NaV1.8, or NaV1.9 activity, preferably NaV1.7 activity, in a biological sample or a mammal, preferably a human, which method comprises administering to the mammal, preferably a human, or contacting said biological sample with a compound of formula (I) or a pharmaceutical composition comprising a compound of formula (I). The term "biological sample", as used herein, includes, without limitation, cell cultures or extracts thereof; biopsied material obtained from a mammal or extracts thereof; and blood, saliva, urine, feces, semen, tears, or other body fluids or extracts thereof.
[0222] Inhibition of NaV1.1, NaV1.2, NaV1.3, NaV1.4, NaV1.5, NaV1.6, NaV1.7, NaV1.8, or NaV1.9 activity in a biological sample is useful for a variety of purposes that are known to one of skill in the art. Examples of such purposes include, but are not limited to, the study of sodium ion channels in biological and pathological phenomena; and the comparative evaluation of new sodium ion channel inhibitors.
[0223] The compounds of the invention (or stereoisomers, geometric isomers, tautomers, solvates, metabolites, isotopes, pharmaceutically acceptable salts, or prodrugs thereof) and/or the pharmaceutical compositions described herein which comprise a pharmaceutically acceptable excipient and one or more compounds of the invention, can be, used in the preparation of a medicament for the treatment of sodium channel-mediated disease or condition in a mammal.
Combination Therapy
[0224] The compounds of the invention may be usefully combined with one or more other compounds of the invention or one or more other therapeutic agent or as any combination thereof, in the treatment of sodium channel-mediated diseases and conditions. For example, a compound of the invention may be administered simultaneously, sequentially or separately in combination with other therapeutic agents, including, but not limited to:
[0225] opiates analgesics, e.g., morphine, heroin, cocaine, oxymorphine, levorphanol, levallorphan, oxycodone, codeine, dihydrocodeine, propoxyphene, nalmefene, fentanyl, hydrocodone, hydromorphone, meripidine, methadone, nalorphine, naloxone, naltrexone, buprenorphine, butorphanol, nalbuphine and pentazocine;
[0226] non-opiate analgesics, e.g., acetomeniphen, salicylates (e.g., aspirin); nonsteroidal antiinflammatory drugs (NSAIDs), e.g., ibuprofen, naproxen, fenoprofen, ketoprofen, celecoxib, diclofenac, diflusinal, etodolac, fenbufen, fenoprofen, flufenisal flurbiprofen, ibuprofen, indomethacin, ketoprofen, ketorolac, meclofenamic acid, mefenamic acid, meloxicam, nabumetone, naproxen, nimesulide, nitroflurbiprofen, olsalazine, oxaprozin, phenylbutazone, piroxicam, sulfasalazine, sulindac, tolmetin and zomepirac;
[0227] anticonvulsants, e.g., carbamazepine, oxcarbazepine, lamotrigine, valproate, topiramate, gabapentin and pregabalin;
[0228] antidepressants such as tricyclic antidepressants, e.g., amitriptyline, clomipramine, despramine, imipramine and nortriptyline;
[0229] COX-2 selective inhibitors, e.g., celecoxib, rofecoxib, parecoxib, valdecoxib, deracoxib, etoricoxib, and lumiracoxib;
[0230] alpha-adrenergics, e.g., doxazosin, tamsulosin, clonidine, guanfacine, dexmetatomidine, modafinil, and 4-amino-6,7-dimethoxy-2-(5-methane sulfonamido-1,2,3,4-tetrahydroisoquinol-2-yl)-5-(2-pyridyl) quinazoline;
[0231] barbiturate sedatives, e.g., amobarbital, aprobarbital, butabarbital, butabital, mephobarbital, metharbital, methohexital, pentobarbital, phenobartital, secobarbital, talbutal, theamylal and thiopental;
[0232] tachykinin (NK) antagonist, particularly NK-3, NK-2 or NK-1 antagonist, e.g., (oR, 9R)-7-[3,5-bis(trifluoromethyl)benzyl)]-8,9,10,11-tetrahydro-9-methyl-5-(- 4-methylphenyl)-7H-[1,4]diazoc ino[2,1-g][1,7]-naphthyridine-6-13-dione (TAK-637), 5-[[2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethylphenyl]ethoxy-3-(4-fluorophe- nyl)-4-morpholinyl]-methyl]-1,2-dihydro-3H-1,2,4-triazol-3-one (MK-869), aprepitant, lanepitant, dapitant or 3-[[2-methoxy5-(trifluoromethoxy)phenyl]-methylamino]-2-phenylpiperidine (2S,3S);
[0233] coal-tar analgesics, in particular paracetamol;
[0234] serotonin reuptake inhibitors, e.g., paroxetine, sertraline, norfluoxetine (fluoxetine desmethyl metabolite), metabolite demethylsertraline, '3 fluvoxamine, paroxetine, citalopram, citalopram metabolite desmethylcitalopram, escitalopram, d,l-fenfluramine, femoxetine, ifoxetine, cyanodothiepin, litoxetine, dapoxetine, nefazodone, cericlamine, trazodone and fluoxetine;
[0235] noradrenaline (norepinephrine) reuptake inhibitors, e.g., maprotiline, lofepramine, mirtazepine, oxaprotiline, fezolamine, tomoxetine, mianserin, buproprion, buproprion metabolite hydroxybuproprion, nomifensine and viloxazine (Vivalan.RTM.)), especially a selective noradrenaline reuptake inhibitor such as reboxetine, in particular (S,S)-reboxetine, and venlafaxine duloxetine neuroleptics sedative/anxiolytics;
[0236] dual serotonin-noradrenaline reuptake inhibitors, such as venlafaxine, venlafaxine metabolite O-desmethylvenlafaxine, clomipramine, clomipramine metabolite desmethylclomipramine, duloxetine, milnacipran and imipramine;
[0237] acetylcholinesterase inhibitors such as donepezil;
[0238] 5-HT3 antagonists such as ondansetron;
[0239] metabotropic glutamate receptor (mGluR) antagonists;
[0240] local anaesthetic such as mexiletine and lidocaine;
[0241] corticosteroid such as dexamethasone;
[0242] antiarrhythimics, e.g., mexiletine and phenytoin;
[0243] muscarinic antagonists, e.g., tolterodine, propiverine, tropsium t chloride, darifenacin, solifenacin, temiverine and ipratropium;
[0244] cannabinoids;
[0245] vanilloid receptor agonists (e.g., resinferatoxin) or antagonists (e.g., capsazepine);
[0246] sedatives, e.g., glutethimide, meprobamate, methaqualone, and dichloralphenazone;
[0247] analytics such as benzodiazepines,
[0248] antidepressants such as mirtazapine,
[0249] topical agents (e.g., lidocaine, capsacin and resiniferotoxin);
[0250] muscle relaxants such as benzodiazepines, baclofen, carisoprodol, chlorzoxazone, cyclobenzaprine, methocarbamol and orphrenadine;
[0251] anti-histamines or H1 antagonists;
[0252] NMDA receptor antagonists;
[0253] 5-HT receptor agonists/antagonists;
[0254] PDEV inhibitors;
[0255] Tramadol.RTM.;
[0256] cholinergic (nicotine) analgesics;
[0257] alpha-2-delta ligands;
[0258] prostaglandin E2 subtype antagonists;
[0259] leukotriene B4 antagonists;
[0260] 5-lipoxygenase inhibitors; and
[0261] 5-HT3 antagonists.
[0262] Sodium channel-mediated diseases and conditions that may be treated and/or prevented using such combinations include but not limited to, pain, central and peripherally mediated, acute, chronic, neuropathic as well as other diseases with associated pain and other central nervous disorders such as epilepsy, anxiety, depression and bipolar disease; or cardiovascular disorders such as arrhythmias, atrial fibrillation and ventricular fibrillation; neuromuscular disorders such as restless leg syndrome and muscle paralysis or tetanus; neuroprotection against stroke, neural trauma and multiple sclerosis; and channelopathies such as erythromyalgia and familial rectal pain syndrome.
[0263] As used herein "combination" refers to any mixture or permutation of one or more compounds of the invention and one or more other compounds of the invention or one or more additional therapeutic agent. Unless the context makes clear otherwise, "combination" may include simultaneous or sequentially delivery of a compound of the invention with one or more therapeutic agents. Unless the context makes clear otherwise, "combination" may include dosage forms of a compound of the invention with another therapeutic agent. Unless the context makes clear otherwise, "combination" may include routes of administration of a compound of the invention with another therapeutic agent. Unless the context makes clear otherwise, "combination" may include formulations of a compound of the invention with another therapeutic agent. Dosage forms, routes of administration and pharmaceutical compositions include, but are not limited to, those described herein.
[0264] The invention will be more fully understood by reference the following examples. They should not, however, be construed limiting the scope of the invention.
EXAMPLES
[0265] These examples serve to provide guidance to a skilled artisan to prepare and use the compounds, compositions and methods of the invention. While particular embodiments of the present invention are described, the skilled artisan will appreciate that various changes and modifications can be made without departing from the spirit and scope of the inventions.
[0266] The chemical reactions in the examples described can be readily adapted to prepare a number of other compounds of the invention, and alternative methods for preparing the compounds of this invention are deemed to be within the scope of this invention. For example, the synthesis of non-examplified compounds according to the invention can be successfully performed by modifications apparent to those skilled in the art, for example, by appropriately protecting interferring group, by utilizing other suitable reagents known in the art, for example, by appropriately protecting interferring groups by utilizing other suitable reagents known in the art other than those described, and/or by making routine modifications of reaction conditions.
[0267] In the examples below, unless otherwise indicated all temperatures are set forth in degrees Celsius. Commercially aviable reagents were purchased from suppliers such as Aldrich Chemical Company, Lancaster, TCI or Maybridge and were used without further purification unless otherwise indicated. The reactions set forth below were done generally under a positive pressure of nitrogen or argon or with a drying tube (unless otherwise stated) in anhydrous solvents, and the reaction flasks were typically fitted with rubber septa for the introduction of substrates and reagents via syringe. Glassware was oven dried and/or heat dried. .sup.1H NMR spectra were obtained in deuterated CDCl.sub.3, d.sub.6-DMSO, CH.sub.3OD or d.sub.6-acetone solvent solutions (reported in ppm) using or trimethylsilane (TMS) or residual non-deuterated solvent peaks as the reference standard. When peak multiplicities are reported, the following abbreviates are used: s (singlet), d (doublet), t (triplet), q (quartet), m (multiplet, br (broadened), dd (doublet of doublets), dt (doublet of triplets). Coupling constants, when given, ar reported in Hz, (Hertz).
[0268] All abbreviations used to describe reagents, reaction conditions or equipment are intended to be consistent with the definitions set forth in the "List of standard abbreviates and acronyms". The chemical names of discrete compounds of the invention were obtained using the structure naming feature of ChemDraw naming program.
LCMS Analytical Methods
[0269] Final compounds were analyzed using three different LC/MS conditions, with UV detector monitoring at 214 nm and 254 nm, and mass spectrometry scanning 110-800 amu in ESI+ ionization mode.
[0270] LC/MS Method A (8.0 min LC-MS Run): XBridge C18 column (4.6.times.50 mm, 3.5 .mu.m, 40.degree. C.); mobile phase: A=10 mM ammonium hydrogen carbonate in water, B-=acetonitrile; gradient: 0.0-8.0 min, 5%-95% B; flow rate=1.2 mL/min.
[0271] LC/MS Method B (8.0 min LC-MS Run): XBridge, C18 column (4.6.times.50 mm, 3.5 .mu.m, 40.degree. C.); mobile phase: A=0.1% ammonia in water, B=acetonitrile; gradient: 0.0-8.0 min, 5%-95% B; flow rate=1.2 mL/min.
[0272] LC/MS Method C (8.0 min LC-MS Run): XBridge C18 column (4.6.times.50 mm, 3.5 .mu.m, 40.degree. C.); mobile phase: A=0.1% TFA in water, B=acetonitrile; gradient: 0.0-8.0 min, 5%-95% B; flow rate=1.2 mL/min.
[0273] LC/MS Method D: Agilent SB C18, 2.1.times.30 min, 1.8 .mu.m; mobile phase: A water (0.05TFA), B CH.sub.3CN (0.05% TFA); gradient: 3% B (0.3 min), followed by 3-95% B (6.5 min), 95% B (1.5 min); flow rate: 0.4 ml/min; oven temperature 25.degree. C.
[0274] LC/MS Method E: Acquity BEH C18, 2.1.times.50 mm, 1.8 .mu.m; mobile phase: A water (0.1% FA), B CH.sub.3CN (0.1% FA); gradient: 3% B (0.4 min), followed by 3-95% B (7.5 min), 95% B (0.5 min); flow rate: 0.5 mL/min; oven temperature 25.degree. C.
[0275] LC/MS Method F: Agilent SB C18, 2.1.times.30 mm, 1.8 .mu.m; mobile phase: A water (0.05% TFA), B CH.sub.3CN (05% TFA); gradient: 3% B (0.3 min), followed by 3-95% B (6.5 min), 95% B (1.5 min); flow rate: 0.4 mL/min; oven temperature 25.degree. C.
[0276] LC/MS Method G: Acquity BEH C18, 2.1.times.50 mm, 1.8 .mu.m; mobile phase: A water (0.1% FA), B CH.sub.3CN (0.1% FA); gradient: 3% B (0.4 min), followed by 3-95% B (7.5 min), 95% B (0.5 min); flow rate: 0.5 mL/min; oven temperature 25.degree. C.
Abbreviations
[0277] MeCN Acetonitrile [0278] EtOAc Ethyl acetate [0279] DCE Dichloroethane [0280] DCM Dichloromethane [0281] DIPEA Diisopropylethylamine [0282] DEA Diethylamine [0283] DMAP 4-dimethylaminopyridine [0284] DMF N,N-Dimethylformamide [0285] DMSO Dimethyl sulfoxide [0286] FA Formic acid [0287] IPA Isopropyl alcohol [0288] TFA Trifluoroacetic acid [0289] EDCl 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride [0290] HCl Hydrochloric acid [0291] HPLC High Pressure Liquid Chromatography [0292] LCMS Liquid Chromatography Mass Spectrometry [0293] MeOH Methanol [0294] NMP N-methyl-2-pyrrolidone [0295] RPHPLC Reverse phase high pressure chromatography [0296] RT Retention time [0297] THF Tetrahydrofuran
EXAMPLES
Example 1
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzylpiperidin-3-yl)oxy)- 5-cyclopropyl-2-fluorobenzamide
##STR00192##
[0298] Step 1. Preparation of (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-fluorobenzoate
##STR00193##
[0300] To a solution of (R)-1-benzylpiperidin-3-ol(0.38 g, 2.00 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (0.50 g, 2.00 mmol) in anhydrous dimethyl sulfoxide (6 mL) was added cesium carbonate (2.16 g, 4.00 mmol). The reaction mixture was stirred at 70.degree. C. for 2 hours under an atmosphere of nitrogen and then cooled to ambient temperature and quenched by addition of 10 mL of water. The mixture was extracted with ethyl acetate (3.times.15 mL); the organic layers were combined and washed with brine (15 mL), dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by column chromatography eluting with a gradient of ethyl acetate in hexanes to (0 to 25%) to give the title compound (0.66 g, 78%) as a white solid: .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.85 (d, J=7.74 Hz, 1H), 7.36-7.18 (m, 5H), 6.63 (d, J=12.2 Hz, 1H), 4.49-4.31 (m, 1H), 3.57 (s, 2H), 3.10-2.96 (m, 1H), 2.82-2.66 (m, 1H), 2.27 (m, 1H), 2.20-2.02 (m, 2H), 1.92-1.75 (m, 1H), 1.73-1.59 (m, 1H), 1.60-1.50 (m, 10H).
Step 2. Preparation of (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy-5-cyclopropyl- 2-fluorobenzoate
##STR00194##
[0302] To a solution of (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate (0.38 g, 0.90 mmol) and cyclopropylboronic acid (0.12 g, 1.35 mmol) in toluene (3 mL) and water (0.3 mL) was added potassium phosphate tribasic (0.64 g, 1.80 mmol), palladium (II) acetate (0.02 g, 0.09 mmol), and tricyclohexyl phosphonium tetrafluoroborate (0.07 g, 0.18 mmol) and the mixture was degassed thoroughly and the reaction vessel filled up with nitrogen before heating at 115.degree. C. for 40 min under microwave irradiation. The reaction mixture was then cooled to ambient temperature and quenched by addition of 10 mL of water. The mixture was then extracted with diethyl ether (2.times.15 mL). The organic layers were combined, concentrated and The residue was purified by column chromatography (10 to 30% gradient of ethyl acetate in hexanes) to give the title compound (0.37 g, 98%) as a colorless oil: .sup.1H NMR (300 MHz, CDCl.sub.3) d7.36 (d, J=8.4 Hz, 1H), 7.33-7.13 (m, 5H), 6.53 (d, J=12.8 Hz, 1H), 4.44-4.30 (m, 1H), 3.61-3.49 (m, 2H), 3.09-2.94 (m, 1H), 2.76-2.63 (m, 1H), 2.31-1.96 (m, 4H), 1.90-1.60 (m, 3H), 1.59-1.54 (m, 9H), 0.92-0.83 (m, 2H), 0.67-0.60 (m, 2H); MS(ES+) m/z 426.2 (M +1).
Step 3. Preparation of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzylpiperidin-3-yl)oxy)-5- cyclopropyl-2-fluorobenzamide
##STR00195##
[0304] T a solution of (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate (0.127 g, 0.30 mmol) in dichloromethane (1 mL) was added trifluoroacetic acid (1 mL). After stirring at ambient temperature for 1 hour, reaction mixture was concentrated, diluted with dichloromethane (10 mL) and washed with aqueous hydrochloric acid (1.0 N, 10 mL). The aqueous layers was extracted with dichloromethane (10 mL), the organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated to give the corresponding carboxylic acid which was used directly for the next step. To a solution of the carboxylic acid (0.11 g, 0.30 mmol) in dichloromethane (2 mL) was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.124 g, 0.48 mmol) and 4-dimethylaminopyridine (0.091 g, 0.75 mmol) and azetidine sulfonamide (0.052 g, 0.39 mmol). The reaction mixture was stirred at ambient temperature for 16 hours and then diluted with dichloromethane (10 mL) and washed with aqueous hydrochloric acid (1.0 N, 10 mL). The aqueous layer was extracted with dichloromethane (10 mL); the organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was first purified by column chromatography eluting with a gradient of methanol in dichlorometharte (0% to 15%) and further purified by preparative HPLC (gradient of acetonitrile in water) to give the title compound as a white solid (0.021 g, 14%): .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.54 (d, J=9.1 Hz, 1H), 7.33-7.22 (m, 5H), 6.56 (d, J=14.5 Hz, 1H), 4.48-4.34 (m, 1H), 4.22 (t, J=7.7 Hz, 4H), 3.57 (s, 2H), 3.07-2.93 (m, 1H), 2.78-2.67 (m, 1H), 2.35-2.13 (m, 4H), 2.13-1.99 (m, 2H), 1.91-1.78 (m, 1H), 1.76-1.46 (m, 2H), 0.95-0.85 (m, 2H), 0.69-0.61 (m, 2H). MS(ES+) m/z: 488.1 (M+1); MS(ES-) m/z 486.2 (M-1).
Example 2
Synthesis of (S)-N-(azetidin-1-sulfonyl)-4-((1-benzylpiperidin-3- yl)oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00196##
[0306] Following the procedure as described in Example 1 step 1 to step 3, and making variation as required to replace (R)-1-benzylpiperdin-3-ol with (S)-1-benzylpiperidin-3-ol, the title compound was obtained as a white solid (0.012 g, 45%): .sup.1H NMR (300 MHz, CDCl.sub.3)d 7.54 (d, J=9.15 Hz, 1H), 7.33-7,22 (m, 5H), 6.56 (d, J=14.5 Hz, 1H), 4.48-4.34 (m, 1H), 4.22 (t, J=7.7 Hz, 4H), 3.57 (s, 2H), 3.07-2.93 (m, 1H), 2.78-2.67 (m, 1H), 2.35-2.13 (m, 4H), 2.13-1.99 (m, 2H), 1.9-1.78 (m, 1H), 1.76-1.46 (m, 2H), 0.95-.085 (m, 2H), 0.69-0.61 (m, 2H); MS(E+) m/z 488.1 (M+1); MS(ES-) m/z 486.2 (M-1).
Example 3
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,5-dichlorobenzyl)piper- idin- 3-yl)oxy)-2-fluorobenzamide
##STR00197##
[0307] Step 1. Preparation of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-chloro-5- fluorophenoxy)piperidine-1-carboxylate
##STR00198##
[0309] To a solution of (R)-tert-butyl 3-hydroxypiperidine-1-carboxylate (10.05 g, 50.00 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (13.02 g, 52.50 mmol) in anhydrous DMSO (200 mL) was added cesium carbonate (40.62 g, 75.00 mmol). The reaction mixture was stirred at 70.degree. C. for 1 hour under an atmosphere of nitrogen and then cooled to ambient temperature and quenched by addition of 50 mL of water. The mixture was extracted with ethyl acetate (3.times.100 mL); the organic layers were combined and washed with brine (150 mL), dried over anhydrous magnesium sulfate, filtered and concentrated. The crude material (22.50 g, 99%) was used directly for the next step without further purification; MS(ES+) m/z 430.2, 431.2 (M+1).
Step 2. Preparation of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine-1-car- boxylate
##STR00199##
[0311] To a solution of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)piperidine- 1-carboxylate (22.50 g, 50.00 mmol) and cyclopropylboronic acid (7.22 g, 83.90 mmol) in toluene (150 mL) and water (15 mL) was added potassium phosphate tribasic (39.53 g, 111.90 mmol), palladium (II) acetate (1.25 g, 5.60 mmol), and tricyclohexyl phosphonium tetrafluoroborate (4.10 g, 11.20 mmol). The mixture was degassed thoroughly and the reaction vessel filled up with nitrogen before heating at 115.degree. C. for 16 hours. The reaction mixture was then cooled to ambient temperature and quenched by addition of 100 mL of water. The mixture was extracted with diethyl ether (2.times.100 mL). The organic layers were combined, concentrated. The residue was purified by column chromatography (10 to 30% gradient of ethyl acetate in hexanes) to give the title compound as an colorless oil (16.50 g, 75%). .sup.1H NMR (300 MHz, CDCl.sub.3)d 7.36 (d, J=8.4 Hz, 1H), 6.55 (d, J=12.6 Hz, 1H), 4.37-4.21 (m, 3.81-3.32 (m, 4H), 2.03-1.76 (m, 5H), 1.55 (s, 9H), 0.92-0.79 (m, 2H), 0.73-0.50 (m, 2H).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid, trifluoroacetic acid salt
##STR00200##
[0313] To a solution of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)- piperidine-1-carboxylate (9.5 g, 21.8 mmol) in dichloromethane (200 mL), was added trifluoroacetic acid (40 ml). The reaction mixture was stirred at ambient temperature for 3 hours and then concentrated in vacuo. The residue was purified by column chromatography (5% to 100% methanol in water on C18 column) afforded the title compound as colorless solid (5.3 g, 64%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 9.83 (brs, 2H), 7.27 (d, J=8.5 Hz, 1H), 7.02 (d, J=13.1 Hz, 1H), 4.76 (brs, 1H), 3.36-3.32 (m, 1H), 3.22-3.16 (m, 1H), 3.04 (brs, 2H), 2.27-2.18 (m, 1H), 1.96-1.66 (m, 4H), 0.92-0.87 (m, 2H), 0.66-0.52 (m, 2H); MS(ES+) m/z 280.3 (M+1); MS(ES-) m/z 278.4 (M-1).
Step 4. Preparation of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3- yl)oxy)-2-fluorobenzoic acid hydrochloride
##STR00201##
[0315] To a stirred solution of (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid trifluoroacetate (0.20 g, 0.53 mmol) in tetrahydrofuran (1 mL) under an atmosphere of nitrogen were introduced 3,5-dichlorobenzaldehyde (0.11 g, 0.64 mmol) and sodium triacetoxyborohydride (0.31 g, 0.96 mmol) and the mixture was stirred for 16 hours. Aqueous hydrochloric acid (1M, 5 mL) was added and the mixture was extracted with ethyl acetate (3.times.10 mL) and concentrated. The residue was purified by column chromatography eluting with 5% methanol in dichloromethane to give the title compound as an oil (0.16 g, 63%): MS(ES+) m/z 438.1, 440.1 (M+1); MS(ES-) m/z 436.1, 438.1 (M-1).
Step 5. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,5-dichlorobenzyl)piper- idin-3-yl)oxy)-2-fluorobenzamide
##STR00202##
[0317] To a solution of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid hydrochloride (0.07 g, 0.17 mmol) in dichloromethane (1 mL) was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.04 g, 0.25 mmol) and 4-dimethylaminopyridine (0.05 g, 0.42 mmol) and cyclopropylsulfonamide (0.02 g, 0.17 mmol). The reaction mixture was stirred at ambient temperature for 16 hours and then diluted with dichloromethane (10 mL) and washed with aqueous hydrochloric acid (1M, mL). The aqueous layer was extracted with dichloromethane (10 mL), the organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated to give an oil which was purified over silica gel chromatography (0 to 15% gradient of methanol containing 1% ammonia solution in dichloromethane) to give the title compound (0.02 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.41 (d, J=12.6 Hz, 1H), 7.23-7.18 (m, 3H), 6.54 (d, J=12.6 Hz, 1H), 4.49-4.33 (m, 1H), 3.56-3.39 (m, 2H), 2.94-2.81 (m, 1H), 2.68-2.53 (m, 1H), 2.46-2.31 (m, 1H), 2.31-2.16 (m, 1H), 2.11-1.97 (m, 2H), 1.92-1.78 (m, 1H), 1.73-1.52 (m, 3H), 0.98-0.79 (m, 6H), 0.72-0.57 (m, 2H); MS(ES+) m/z 541.1, 543.1 (M+1); MS(ES-) m/z 539.2, 541.2 (M-1).
Example 4
Synthesis of (R)-4-((1-acetylpiperidin-3-yl)oxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)- - 2-fluorobenzamide
##STR00203##
[0319] This compound was isolated as a side product during the synthesis of Example 3 in step 5 (0.02 g, 24%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 7.21-7.00 (m, 2H), 4.83-4.52 (m, 1H), 4.06-3.93 (m, 1H), 3.86-3.69 (m, 1H), 3.63-3.38 (m, 2H), 3.13-2.93 (m, 2H), 1.99-1.76 (m, 5H), 172-1.36 (m, 2H), 1.29-1.03 (m, 4H), 0.90-0.79 (m, 2H), 0.69-0.60 (m, 2H); MS(ES+) m/z 425.2 (M+1); MS(ES-) m/z 423.3 (M-1).
Example 5
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)pip- eridin- 3-yl)oxy)-2-fluorobenzamide
##STR00204##
[0321] Following the procedure as described in Example 3 step 5, and making variation as required to replace cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.02 g, 25%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.63-11.45 (m, 1H), 7.46-7.40 (m, 1H), 7.38-7.29 (m, 2H), 7.11 (d, J=8.36 Hz, 1H), 6.97 (d, J=13.0 Hz, 1H), 4.68-4.53 (m, 1H), 4.05-3.92 (m, 4H), 3.63-3.43 (m, 2H), 2.74-2.63 (m, 1H), 2.44-2.25 (m, 2H), 2.20-1.99 (m, 3H), 1.93-1.67 (m, 2H), 1.63-1.48 (m, 3H), 0.96-0.79 (m, 2H), 0.77-0.60 (m, 2H); MS(ES+) m/z 556.1, 558.1 (M+1); MS(ES-) m/z 554.2, 556.2 (M-1).
Example 6
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-- yl)oxy-5-cyclopropyl-2-fluorobenzamide
##STR00205##
[0323] Following the procedure as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with 2-chloro-4-fluorobenzaldehyde and cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.035 g, 50%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.62-11.47 (m, 1H), 7.45-7.40 (m, 1H), 7.38-7.28 (m, 2H), 7.11 (d, J=8.3 Hz, 1H), 4.68-4.53 (m, 1H), 4.05-3.92 (m, 4H), 3.62-3.42 (m, 2H), 2.75-2.63 (m, 1H), 2.44-2.26 (m, 2H), 2.20-2.00 (m, 3H), 1.90-1.67 (m, 2H), 1.63-1.48 (m, 3H), 0.94-0.81 (m, 2H), 0.76-0.62 (m, 2H); MS(ES+) m/z 540.1, 542.1 (M+1); MS(ES-) m/z 538.2, 540.2 (M-1).
Example 7
Synthesis of (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl- N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00206##
[0325] Following the procedures as described in Example 3 steps 4 and 5, and making variation as required to replace 3,5-dichlorobenzaldehyde with 2-chloro-4-fluorobenzaldehyde, the title compound was obtained (0.038 g, 37%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89-11.60 (m, 1H), 7.49 (dd, J=8.5, 6.5 Hz, 1H), 7.37 (dd, J=8.8, 2.6 Hz, 1H), 7.17-7.05 (m, 2H), 6.98 (d, J=13.2 Hz, 1H), 4.68-4.50 (m, 1H), 3.63-3.52 (m, 2H), 3.11-2.97 (m, 1H), 2.84-2.70 (m, 1H), 2.61-2.49 (m, 2H), 2.43-2.29 (m, 1H), 2.12-1.98 (m, 1H), 1.98-1.65 (m, 2H), 1.64-1.45 (m, 2H), 1.13-1.02 (m, 4H), 0.91-0.81 (m, 2H), 0.72-0.62 (m, 2H); MS(ES+) m/z 525.1, 527.1 (M+1); MS(ES-) m/z 523.2, 525.2 (M-1).
Example 8
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(2,4-difluorobenzyl)- piperidin-3-yl)oxy)-2-fluorobenzamide
##STR00207##
[0327] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with 2,4-difluorobenzaldehyde and cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.048 g, 57%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.95-11.21 (m, 1H), 7.44 (dd, J=15.4, 8.53 Hz, 1H), 7.26-6.81 (m, 4H), 4.67-4.52 (m, 1H), 4.08-3.97 (m, 4H), 3.62-3.54 (m, 2H), 2.90-2.77 (m, 1H), 2.64-2.53 (m, 1H), 2.46-2.22 (m, 2H), 2.22-2.05 (m, 3H), 2.01-1.87 (m, 1H), 1.85-1.69 (m, 1H), 1.68-1.39 (m, 2H), 0.95-0.82 (m, 2H), 0.76-0.64 (m, 2H); MS(ES+) m/z 524.1 (M+1); MS(ES-) m/z 522.2 (M-1).
Example 9
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(2,4-difluorobenzyl)- piperidin-3-yl)oxy)-2-fluorobenzamide
##STR00208##
[0329] Following the procedures as described in Example 3 steps 4 and 5, and making variation as required to replace 3,5-dichlorobenzaldehyde with 2,4-difluorobenzaldehyde, the title compound was obtained (0.035 g, 39%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87-11.57 (m, 1H), 7.51-7.38 (m, 1H), 7.26-6.95 (m, 4H), 4.67-4.51 (m, 1H), 3.62-3.55 (m, 2H), 3.13-3.00 (m, 1H), 2.90-2.77 (m, 1H), 2.65-2.52 (m, 1H), 2.45-2.16(m, 2H), 2.11-2.01 (m, 1H), 1.99-1.87 (m, 1H), 1.83-1.68 (m, 1H), 1.67-1.38 (m, 2H), 1.14-1.01 (m, 4H), 0.93-0.83 (m, 2H), 0.75-0.61 (m, 2H); MS(ES+) m/z 509.2 (M+1); MS(ES-) m/z 507.3 (m-1).
Example 10
Synthesis of (R)-5-cyclopropyl-4-((1-(2.6-dichlorobenzyl)piperidin-3-yl)oxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00209##
[0331] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with 2,6-dichlorobenzaldehyde and cyclopropylsulfonamide with methylsulfonamide, the title compound was obtained (0.018 g, 13%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 7.46-7.34 (m, 2H), 7.28 (dd, J=8.8, 7.18 Hz, 1H), 7.14 (d, J=8.8 Hz, 1H), 4.39-4.24 (m, 1H), 3.67 (m, 2H), 2.94-2.80 (m, 1H), 2.80-2.72 (m, 3H), 2.61-2.53 (m, 1H), 2.44-2.37 (m, 1H), 2.37-2.23 (m, 1H), 2.05-1.83 (m, 2H), 1.78-1.60 (m, 1H), 1.54-1.36 (m, 2H), 0.86-0.73 (m, 2H), 0.56-0.42 (m, 2H); MS(ES+) m/z 515.2, 517.2 (M+1); MS(ES-) m/z 513.3, 515.3 (M+1).
Example 11
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-(cyclohexylmethyl)piperidin-3- yl)oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00210##
[0333] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with cyclohexanecarbaldehyde and cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.041 g, 51%) as a colorless solid: .sup.1H NMR (300 MHz, acetonitrile-d.sub.3) .delta. 736-7.26 (m, 1H), 7.07-6.95 (m, 1H), 5.01-4.88 g (m, 1H), 4.18-4.08 (m, 4H), 2.98-2.83 (m, 2H), 2.31-2.15 (m, 7H), 1.84-1.57 (m, 9H), 1.36-1.11 (m, 4H), 1.08-0.87 (m, 4H), 0.70-0.62 (m, 2H); MS(ES+) m/z 494.3 (M+1); MS(ES-) m/z 492.4 (M-1).
Example 12
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(1-methyl-3-phenyl-1H-pyrazol- 5-yl)methyl)piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00211##
[0335] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with 1-methyl-3-phenyl-1H-pyrazole-5-carbaldehyde and cyclopropylsulfonamide with methylsulfonamide, the title compound was obtained (0.023 g, 39%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 7.75-7.61 (m, 2H), 7.33 (t, J=7.5 Hz, 2H), 7.25-7.13 (m, 2H), 6.81-6.67 (m, 1H), 6.52 (d, J.ltoreq.3.3 Hz, 1H), 4.53-4.41 (m, 1H), 3.79 (s, 3H), 3.61-3.50 (m, 2H), 2.88-2.74 (m, 4H), 2.31-2.19 (m, 1H), 2.07-1.83 (m, 3H), 1.79-1.68 (m, 1H), 1.57-1.38 (m, 3H), 0.86-0.78 (m, 2H), 0.58-0.48 (m, 2H); MS(ES+) m/z 527.3 (M+1); MS(ES-) m/z 525.3 (M-1).
Example 13
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-methylsulfonyl)-4-((1-(pyridazin-4- ylmethyl)piperidin-3-yl)oxy)benzamide
##STR00212##
[0337] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with 1-methyl-3-phenyl-1H- pyrazole-5-carbaldehyde and cyclopropylsulfonamide with methylsulfonamide, the title compound was obtained (0.035 g, 40%) as a colorless solid: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.87-8.82 (m, 1H), 8.75 (dd, J=5.2, 0.8 Hz, 1H), 7.23-7.18 (m, 1H), 7.08 (d, J=8.8 Hz, 1H), 6.36 (d, J=13.5 Hz, 1H), 4.28-4.16 (m, 1H), 3.35 (d, J=15.0 Hz, 1H), 3.26 (d, J=15.0 Hz, 1H), 2.88 (s, 3H), 2.59-2.48 (m, 1H), 2.38-2.02 (m, 4H), 1.85-1.76 (m, 2H), 1.46-1.29 (m, 3H), 0.68-0.59 (m, 2H), 0.46-0.34 (m, 2H); MS(ES+) m/z 449.1 (M+1).
Example 14
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(isoindolin-4-ylmethyl)piperidin- 3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00213##
[0338] Step 1. Preparation of (R)-tert-butyl 4-((3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)-phenoxy)piper- idin-1-yl)methyl)isoindoline-2-carboxylate
##STR00214##
[0340] Following the procedures as described in Example 3 steps 4 and 5, and making variations as required to replace 3,5-dichlorobenzaldehyde with tert-butyl 4-formylisoindoline-2-carboxylate and cyclopropylsulfonamide with methylsulfonamide, the title compound was obtained (0.030 g, 16%): MS(ES+) m/z 588.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(isoindolin-4-ylmethyl)- piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00215##
[0342] To a stirred solution of (R)-tert-butyl 4-((3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl)-carabamoyl)phenoxy)pipe- ridin-1-yl)methyl)isoindoline-2-carboxylate (0.030 g, 0.051 mmol) in dichloromethane (1 mL) was added trifluoroacetic acid (0.3 mL) and the mixture was stirred at ambient temperature for 1 hour and then concentrated. The residue was purified by silica gel chromatography (0 to 15% gradient of methanol plus 1% ammonia solution in dichloromethane) to give the title compound (0.01 g, 38%): .sup.1H NMR (300 MHz, MeOD-d.sub.4) d 7.41-7.18 (m, 4H), 6.69 (d, J=13.1 Hz, 1H), 4.77-4.54 (m, 2H), 4.53-4.43 (m, 1H), 3.61-3.55 (m, 2H), 3.54-3.40 (m, 1H), 3.37-3.03 (m, 4H), 2.88-2.79 (m, 1H), 2.61-2.52 (m, 1H), 2.51-2.41 (m, 1H), 2.38-2.26 (m, 1H), 2.10-1.99 (m, 2H), 1.94-1.82 (m, 1H), 1.78-1.70 (m, 1H), 1.69-1.56 (m, 1H), 0.94-0.85 (m, 2H), 0.70-0.57 (m, 2H); MS(ES+) m/z 488.3 (M+1); MS(ES-) m/z 486.4 (M-1).
Example 15
Synthesis of(R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzhydrylpiperidin-3-yl)o- xy)- 5-cyclopropyl-2-fluorobenzamide
##STR00216##
[0343] Step 1. Preparation of (R)-4-((1-benzhydrylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoic acid.
##STR00217##
[0345] To a stirred solution of (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid trifluoroacetate (0.20 g, 0.53 mmol) in acetonitrile (2 mL) under an atmosphere of nitrogen were added (bromomethylene)dibenzene (0.16 g, 0.64 mmol), potassium carbonate (0.17 g, 1.28 mmol) and sodium iodide (0.09 g, 0.64 mmol) and the mixture was stirred at reflux for 16 hours. After cooled to ambient temperature, 1M aqueous hydrochloric acid (5 mL) was added slowly and the mixture was extracted with ethyl acetate (3.times.10 mL) and concentrated. The residue was purified over silica gel chromatography eluting with 30% ethyl acetate (containing 1% Formic acid) in hexanes to give compound the title compound as an oil (0.16 g, 70%): MS(ES+) m/z 446.1 (M+1).
Step 2. Preparation of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzhydrylpiperidin-3-yl)oxy)-5-cyclo- propyl-2-fluorobenzamide
##STR00218##
[0347] Following the procedure as described in Example 3 step 5, and making variations as required to replace cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.035 g, 34%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.65-11.53 (m, 1H), 7.43-7.35 (m, 2H), 7.31-7.19 (m, 4H), 7.19-7.05 (m, 5H), 6.86 (d, J=13.0 Hz, 1H), 4.71-4.57 (m, 1H), 4.39-4.33 (m, 1H), 4.08-3.95 (m, 4H), 2.62-2.49 (m, 1H), 2.44-2.21 (m, 3H), 2.20-2.05 (m, 3H), 1.94-1.70 (m, 2H), 1.67-1.48 (m, 0,96-0.87 (m, 2H), 0.79-0.69 (m, 2H); MS(ES+) m/z 564.3 (M+1); MS(ES-) m/z 562.4 (M-1).
Example 16
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzhydrylpiperidin-3-yl)oxy)-5-cyclo- propyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00219##
[0349] Following the procedure as described in Example 15 step 2, and making variations as required to replace azetidine-1-sulfonamide with cyclopropylsulfonamide, the title compound was obtained (0.048 g, 52%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.90-11.73 (m, 1H), 7.43-7.34 (m, 2H), 7.31-7.20 (m, 4H), 7.19-7.04 (m, 5H), 6.86 (d, J=13.2 Hz, 1H), 4.73-4.55 (m, 1H), 4.40-4.32 (m, 1H), 3.12-2.99 (m, 1H), 2.62-2.49 (m, 1H), 2.44-2.21 (m, 3H), 2.20-2.05 (m, 1H), 1.94-1.71 (m, 2H), 1.68-1.46 (m, 2H), 1.15-1.02 (m, 4H), 0.96-0.87 (m, 2H), 0.79-0.68 (m, 2H); MS(ES+) m/z 549.3 (M+1); MS(ES-) m/z 547.4 (M-1).
Example 17
Synthesis of (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00220##
[0350] Step 1. Preparation of (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3- yl)oxy)-2-fluorobenzoic acid
##STR00221##
[0352] To a solution of (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid (0.40 g, 1.43 mmol) and 3,4-dichlorobenzaldehyde (0.3 g, 1.72 mmol) in tetrahydrofuran (2 mL) was added sodium triacetoxyborohydride (0.55 g, 2.58 mmol). The reaction mixture was stirred at ambient temperature for 2 hours, and concentrated in vacuo. The residue was diluted with ethyl acetate (50 mL), washed with aqueous ammonium chloride (25% solution, 2.times.25 mL); dried over anhydrous sodium sulfate and concentrated invacuo. The crude product was purified by column chromatography (5% to 100% methanol in water on C18 column) afforded the title compound as colorless solid (0.42 g, 56%): MS(ES+) m/z 438.2, 440.2 (M+1); MS(ES-) m/z 436.3, 438.3 (M-1).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00222##
[0354] To a mixture of (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid (0.10 g, 0.23 mmol), 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.10 g, 0.52 mmol) and 4-dimethylaminopyridine (0.06 g, 0.52 mmol) in anhydrous dichloromethane (2 mL) was added methanesulfonamide (0.03 g, 0.34 mmol) at ambient temperature. The resulting mixture was stirred at ambient temperature for 16 hours. The mixture was diluted with ethyl acetate (50 mL), washed with aqueous ammonium chloride (25% solution, 2.times.25 mL), dried over anhydrous sodium sulfate, and filtered.
[0355] The filtrate was concentrated in vacuo, the crude product was purified by silica gel column chromatography using 10-100% ethyl acetate in hexanes as an eluent to afford the title compound as colorless solid (0.07 g, 58%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.74 (brs, 1H), 7.57-7.53 (m, 2H), 7.29 (dd, J=1.8, 8.3 Hz, 1H), 7.14 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.2 Hz, 1H), 4.63-4.61 (m, 3.61 (d, J =14.0 Hz, 1H), 3.53 (d, j=14.0 Hz, 1H), 3.29 (s, 3H), 2.79-2.75 (m, 1H), 2.55-2.32 (m, 3H), 2.12-2.03 (m, 1H), 1.92-1.89 (m, 1H), 1.83-1.77 (m, 1H), 1.63-1.53 (m, 2H), 0.93-0.85 (m, 2H), 0.76-0.65 (m, 2H); MS(ES+) m/z 515.2, 517.2 (M+1); MS(ES-) m/z 513.1, 515.1 (M-1).
Example 18
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,4-dichlorobenzyl)- piperidin-3-yl)oxy-2-fluorobenzamide
##STR00223##
[0357] Following the procedure as described in Example 17 step 2, and making variations as required to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (06 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.72 (brs, 1H), 7.56-7.53 (m, 2H), 7.28 (dd, J=1.8, 8.3 Hz, 1H), 7.13 (d, J=8.4 Hz, 1H), 7.00 (d, J=13.2 Hz, 1H), 4.63-4.61 (m, 1H), 3.60 (d, J=14.0 Hz, 1H), 3.51 (d, J=14.0 Hz, 1H), 3.11-3.02 (m, 1H), 2.77-2.74 (m, 1H), 2.54-2.34 (m, 3H), 2.13-2.03 (m, 1H), 1.92-1.77 (m, 2H), 1.60-1.56 (m, 2H), 0.12-1.07 (m, 4H), 0.92-0.88 (m, 2H), 0.74-0.69 (m, 2H); MS(ES+) m/z 541.2, 543.2 (M+1); MS(ES-) m/z 539.1, 541.1 (M-1).
Example 19
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)- piperidin-3-yl)oxy-2-fluorobenzamide
##STR00224##
[0359] Following the procedure as described in Example 17 step 2, and making variations as required to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.01 g, 11%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.65 (brs, 1H), 7.58 (d, J=9.1 Hz, 1H), 7.44-7.35 (m, 2H), 7.16-7.14 (m, 1H), 6.58 (d, J=14.1 Hz, 1H), 4.44 (brs, 1H), 4.27-4.22 (m, 4H), 3.56-3.45 (m, 2H), 2.92-2.88 (m, 1H), 2.67-2.63 (m, 1H), 2.41-2.22 (m, 4H), 2.12-2.03 (m, 2H), 1.91-1.87 (m, 1H), 1.68-1.63 (m, 2H), 0.97-0.91 (m, 2), 0.71-0.67 (m, 2H); m/z 556.2, 558.2 (M+1); MS(ES-) m/z 554.2, 556.2 (M-1).
Example 20
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-3-yl)oxy-N-(me- thylsulfonyl)benzamide
##STR00225##
[0360] Step 1. Preparation of (5)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin- 3-yl)oxy)benzoic acid
##STR00226##
[0362] Following the procedure as described in Example 17 step 1, and making variations as required to replace 3,4-dichlorobenzaldehyde with 4-fluorobenzaldehyde, the title compound was obtained as is colorless solid (0.22 g, 41%): MS(ES+) m/z 388.2(M+1); MS(ES-) m/z 386.2 (M-1).
Step 2. Preparation of ((R )-5-cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-3- yl)oxy)-N-(methylsulfonyl)benzamide
##STR00227##
[0364] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5 -cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-3-yl)oxy)benzoic acid, the title compound was obtained as colorless solid (0.01 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.68 (brs, 1H), 7.36-7.31 (m, 2H), 7.15-7.09 (m, 3H), 6.93 (d, J=13.1 Hz, 1H), 4.59-4.55 (m, 1H), 3.62-3.50 (m, 2H), 3.21 (m, 3H), 2.84-2.81 (m, 1H), 2.60-2.56 (m, 1H), 2.44-2.26 (m, 2H), 2.10-2.01 (m, 1H), 1.98-1.92 (m, 1H), 1.82-1.76 (m, 1H), 1.64-1.46 (m, 2H), 0.90-0.87 (m, 2H), 0.69-0.65 (m, 2H); MS(ES+) m/z 465.3 (M+1); MS(ES-) m/z 463.2 (M-1).
Example 21
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(4- fluorobenzyl)piperidin-3-yl)oxy)benzamide
##STR00228##
[0366] Following the procedure as described in Example 17 step 2, and making variations required to replace (R)-5-cyclopropyl-4-((1 -(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorobenzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (0.05 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.62 (brs, 1H), 7.36-7.31 (m, 2H), 7.15-7.09 (m, 3H), 6.97 (d, J=13.2 Hz, 1H), 4.62-4.57 (m, 1H), 3.63-3.52 (m, 2H), 3.09-3.01 (m, 1H), 2.83-2.80 (m, 1H), 2.60-2.56 (m, 1H), 2.42-2.27 (m, 2H), 2.11-2.02 (m, 1H), 1.98-1.92 (m, 1H), 1.82-1.76 (m, 1H), 1.61-1.50 (m, 2H), 1.10-1.06 (m, 4H), 0.91-0.87 (m, 2H), 0.71-0.67 (m, 2H); MS(ES+) m/z 491.3 (M+1); MS(ES-) m/z 489.3 (M-1).
Example 22
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-(4- fluorobenzyl)piperidin-3-yl)oxy)benzamide
##STR00229##
[0368] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.04 g, 44%): .sup.1HNMR (300 MHz, DMSO-d.sub.6) .delta. 11.48 (brs, 1H), 7.35-7.31 (m, 2H), 7.14-7.09 (m, 3H), 6.98 (d, J=13.0 Hz, 1H), 4.60-4.58 (m, 1H), 4.01 (t, J=7.7 Hz, 4H), 3.62-3.50 (m, 2H), 2.82-2.79 (m, 1H), 2.60-2.56 (m, 1H), 2.41-2.26 (m, 2H), 2.20-2.04 (m, 3H), 1.98-1.92 (m, 1H), 1.82-1.76 (m, 1H), 1.64-1.46 (m, 2H), 0.91-0.85 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 506.3 (M+1); MS(ES-) m/z 504.3 (M-1).
Example 23
Synthesis of (R)-4-((1-(2-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-N- (cyclopropylsulfonyl)-2-fluorobenzamide
##STR00230##
[0369] Step 1. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(2-chlorobenzyl)piperidin-3- yl)oxy)benzoic acid
##STR00231##
[0371] Following the procedure as described in Example 17 step 1, and making variations as required to replace 3,4-dichlorobenzaldehyde with 2-chlorobenzaldehyde, the title compound was obtained as colorless solid (0.28 g, 41%): MS(ES+) m/z 404.2, 406.2 (M+1); MS(ES-) m/z 402.2, 404.2 (M-1).
Step 2. Preparation of (R)-4-((1-(2-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-N-(cyclopropy- lsulfonyl)-2-fluorobenzamide
##STR00232##
[0373] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2-chlorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (0.09 g, 98%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.71 (brs, 1H), 7.48-7.45 (m, 1H), 7.39-7.36 (m, 1H), 7.24-7.21 (m, 2H), 7.08 (d, J=8.4 Hz, 1H), 6.97 (d, J=13.2 Hz, 1H), 4.60-4.58 (m, 1H), 3.59 (s, 2H), 3.07-2.99 (m, 1H), 2.79-2.76 (m, 1H), 2.58-2.51 (m, 1H), 2.50-2.36 (m, 2H), 2.10-2.00 (m, 1H), 1.91-1.88 (m, 1H), 1.81-1.75 (m, 1H), 1.58-1.51 (m, 2H), 1.08-1.04 (m, 4H), 0.87-0.83 (m, 2H), 0.68-0.65 (m, 2H); MS(ES+) m/z 507.3, 509.3 (M+1); MS(ES-) m/z 505.3, 507.3 (M-1).
Example 24
Synthesis of (R)-4-((1-(2-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-2- fluoro-N-(methylsulfonyl)benzamide
##STR00233##
[0375] Following the procedure as described in Example 17 step 3, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2chlorobenzyl)piperidin-3-yl)oxy)benzoi- c acid the title compound was obtained as colorless solid (0.05 g, 58%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (brs, 1H), 7.52-7.49 (m, 1H), 7.43-739 (m, 1H), 7.28-7.24 (m, 2H), 7.13 (d, J=8.4 Hz, 1H), 7.00 (d, J=13.2 Hz, 1H), 4.64-4.561 (m, 1H), 3.64 (s, 2H), 3.31 (s, 3H), 2.83-2.80 (m, 1H), 2.63-2.57 (m, 1H), 2.53-2.38 (m, 2H), 2.33-2.04 (m, 1H), 1.95-1.92 (m, 1H), 1.84-1.77 (m, 1H), 1.62-1.57 (m, 2H), 0.92-0.85 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 481.2, 483.2 (M+1); MS(ES-) m/z 479.3, 481.3 (M-1).
Example 25
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-(2-chlorobenzyl)piperidin-3- yl)oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00234##
[0377] Following the procedure as described to Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2-chlorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.07 g, 74%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.56 (brs, 1H), 7.53-7.49 (m, 1H), 7.43-7.38 (m, 110, 7.29-7.23 (m, 2H), 7.13 (d, J=8.3 Hz, 1H), 7.02 (d, J=13.0 Hz, 1H), 4.64-4.62 (m, 1H), 4.03 (t J=7.7 Hz, 4H), 3.63 (s, 2H), 2.84-2.80 (m, 1H), 2.62-2.57 (m, 1H), 2.49-2.39 (m, 2H), 2.21-2.05 (m, 3H), 1.95-1.92 (m, 1H), 1.84-1.79 (m, 1H), 1.62-1.54 (m, 2H), 0.92-0.87 (m, 2H), 0.73-0.69 (m, 2H); MS(ES+) m/z 522.3, 524.2 (M+1); MS(ES-) m/z 520.3, 522.3 (M-1).
Example 26
Synthesis of (R)-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-2- fluoro-N-(methylsulfonyl)benzamide
##STR00235##
[0378] Step 1. Preparation of (R)-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)-5- cyclopropyl-2-fluorobenzoic acid
##STR00236##
[0380] Following the procedure as described to Example 17 step 1, and making variations as required to replace 3,4-dichlorobenzaldehyde with 3-chlorobenzaldehyde, the title compound was obtained as colorless solid (0.23 g, 41%): MS(ES+) m/z 404.2, 406.2 (M+1); MS(ES-) m/z 402.2, 404.2 (M-1).
Step 2. Preparation of (R)-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-N-(cyclopropy- lsulfonyl)-2-fluorobenzamide
##STR00237##
[0382] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)benzo- ic acid, the title compound was obtained as colorless solid (0.04 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.71 (brs, 1H), 7.39-7.25 (m, 4H), 7.13 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.1 Hz, 1H), 4.63-4.61 (m, 1H), 3.66-3.54 (m, 2H), 3.29 (s, 3H), 2.81-2.78 (m, 1H), 2.56-2.36 (m, 3H), 2.11-2.03 (m, 1H), 1.93-1.89 (m, 1H), 1.83-1.77 (m, 1H), 1.63-1.53 (m, 2H), 0.92-0.88 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 481.2, 483.2 (M+1); MS(ES-) m/z 479.3, 481.3 (M-1).
Example 27
Synthesis of (R)-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-N-(cyclopropy- lsulfonyl)-2-fluorobenzamide
##STR00238##
[0384] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy-2-fluorobe- nzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (0.06 g, 65%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.71 (brs, 1H), 7.38-7.24 (m, 4H), 7.12 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.2 Hz, 1H), 4.3-4.61 (m, 1H), 3.63-3.51 (m, 2H), 3.10-3.01 (m, 2H), 2.79-2.75 (m, 1H), 2.57-2.33 (m, 3H), 2.13-2.04 (m, 1H), 1.93-1.77 (m, 2H), 1.61-1.52 (m, 2H), 1.11-1.06 (m, 4H), 0.92-0.89 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 507.2, 509.2 (M+1); MS(ES-) m/z 505.3, 507.3 (M-1).
Example 28
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)- 5-cyclopropyl-2-fluorobenzamide
##STR00239##
[0386] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(3-chlorobenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.02 g, 22%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.55 (brs, 1H), 7.37-7.24 (m, 4H), 7.14 (d, J=8.4 Hz, 1H), 6.98 (d, J=13.0 Hz, 1H), 4.61-4.59 (m, 1H), 3.99 (t, J=7.6 Hz, 4H), 3.61-3.49 (m, 2H), 2.78-2.74 (m, 1H), 2.54-2.27 (m, 3H), 2.18-2.04 (m, 3H), 1.95-1.90 (m, 1H), 1.82-1.76 (m, 1), 1.60-1.52 (m, 2H), 0.93-0.85 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 522.2, 524.2 (M+1); MS(ES-) m/z 520.3, 522.3 (M-1).
Example 29
Synthesis of (R)-5-cyclopropyl-4-((1-(2,4-dichlorobenzyl)piperidin-3-yl)oxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00240##
[0387] Step 1. Preparation of (R)-5-cyclopropyl-4-((1-(2,4-dichlorobenzyl)piperidin-3- yl)oxy-2-fluorobenzoic acid
##STR00241##
[0389] Following the procedure as described in Example 17 step 1, and making variations as required to replace 3,4-dichlorobenzaldehyde with 2,4-dichlorobenzaldehyde, the title compound was obtained as colorless solid (0.35 g, 56%): MS(ES+) m/z 438.2, 440.2 (M+1); MS(ES-) m/z 436.2, 438.2 (M-1).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(2,4-dichlorobenzyl)piperidin-3-yl)oxy-2- fluoro-N-(methylsulfonyl)benzamide
##STR00242##
[0391] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2,4-dichlorobenzyl)piperidin-3-yl)oxy)b- enzoic acid, the title compound was obtained as colorless solid (0.02 g, 25%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.84 (brs, 1H), 7.57 (d, J=2.1 Hz, 1H), 7.50 (d, J=8.3 Hz, 1H), 7.33 (dd, J=2.1 Hz, 8.3 Hz, 1H), 7.13 (d, J=8.4 Hz, 1H), 6.99 (d, J =13.2 Hz, 1H), 4.63-4.61 (m, 1H), 3.59 (m, 2H), 3.30 (s, 3H), 2.81-2.76 (m, 1H), 2.58-2.39 (m, 3H), 2.12-2.04 (m, 1H), 1.94-1.77 (m, 2H), 1.61-1.53 (m, 2H), 0.91-0.88 (m, 2H), 0.72-0.69 (m, 2H); MS(ES+) m/z 515.1, 517.1 (M+1); MS(ES-) m/z 513.2, 515.2 (M-1).
Example 30
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(2,4-dichlorobenzyl)pip- eridin- 3-yl)oxy)-2fluorobenzamide
##STR00243##
[0393] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2,4-dichlorobenzyl)piperidin-3-yl)oxy)b- enzoic acid and methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.06 g, 64%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.58 (brs, 1H), 7.56 (d, J=2.1 Hz, 1H), 7.50 (d, J=8.4 Hz, 1H), 7.33 (dd, J=2.1 Hz, 8.3 Hz, 1H), 7.13 (d, J=8.4 Hz, 1H), 7.01 (d, J=13.1 Hz, 1H), 4.63-4.61 (m, 1H), 4.03 (t, J=7.7 Hz, 4H), 3.59 (s, 2H), 2.79-2.76 (m, 1H), 2.58-2.39 (m, 3H), 2.21-2.04 (m, 3H), 1.94-1.79 (m, 2H), 1.59-1.56 (m, 2H), 0.92-0.88 (m, 2H), 0.73-0.69 (m, 2H); MS(ES-) m/z 556.2, 558.2 (M+1); MS(ES-) m/z 554.3, 556.3 (M-1).
Example 31
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(2,4-dichlorobenzyl)piper- idin- 3-yl)oxy)-2-fluorobenzamide
##STR00244##
[0395] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(2,4-dichlorobenzyl)piperidin-3-yl)oxy)b- enzoic acid and methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (0.05 g, 48%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (brs, 1H), 7.57 (d, J=2.1 Hz, 1H), 7.50 (d, J=8.3 Hz, 1H), 7.33 (dd, J=2.1 Hz, 8.3 Hz, 1H), 7.12 (d, J=8.4 Hz, 1H), 7.01 (d, J=13.2 Hz, 1H), 4.63-4.61 (m, 1H), 3.59 (s, 2H), 3.11-3.03 (m, 1H), 2.80-2.76 (m, 1H), 2.58-2.39 (m, 3H), 2.12-2.04 (m, 1H), 1.94-1.77 (m, 2H), 1.60-1.55 (m, 2H), 1.12-1.07 (m, 4H), 0.91-0.88 (m, 2H), 0.72-0.69 (m, 2H); MS(ES+) m/z 541.2, 543.1 (M+1); MS(ES-) m/z 539.3, 542,2 (M-1).
Example 32
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(4- methylbenzyl)piperidin- 3-yl)oxy)benzamide
##STR00245##
[0396] Step 1. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-methylbenzyl)piperidin- 3-yl)oxy)benzoic acid
##STR00246##
[0398] Following the procedure as described in Example 17 step 1, and making variations as required to replace 3,4-dichlorobenzaldehyde with 4-methylbenzaldehyde, the title compound was obtained as colorless solid (0.24 g, 44%): MS(ES+) m/z 384.3 (M+1); MS(ES-) m/z 382.3 (M-1).
Step 2. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(4-methylbenzyl)- piperidin-3-yl)oxy)benzamide
##STR00247##
[0400] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid and methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as colorless solid (0.01 g, 18%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.46 (brs, 1H), 7.20-7.09 (m, 5H), 6.96 (d, J=13.1 Hz, 1H), 4.59-4.57 (m, 1H), 3.63-3.52 (m, 2H), 3.08-3.00 (m, 1H), 2.86-2.82 (m, 1H), 2.63-2.59 (m, 1H), 2.43-2.33 (m, 2H), 2.26 (s, 3H), 2.11-2.02 (m, 1H), 1.98-1.92 (m, 1H), 1.82-1.77 (m, 1H), 1.64-1.49 (m, 2H), 1.08-1.03 (m, 4H), 0.90-0.87 (m, 2H), 0.70-0.66 (m, 2H); MS(ES+) m/z 487.2 (M+1); MS(ES-) m/z 485.3 (M-1).
Example 33
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-(4-methylbenzy- l)piperidin- 3-yl)oxy)benzamide
##STR00248##
[0402] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(4-methylbenzyl)piperidin-3-yl)oxy)benzo- ic acid and methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as off-white solid (0.05 g, 54%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.28 (brs, 1H), 7.25-7.12 (m, 5H), 7.00 (d, J=13.0 Hz, 1H), 4.66-4.64 (m, 1H), 4.01 (t, J=7.7 Hz, 4H), 3.75-3.63 (m, 2H), 2.96-2.92 (m, 1H), 2.71-2.67 (m, 1H), 2.56-2.42 (m, 2H), 2.27 (s, 3H), 2.19-2.05 (m, 3H), 1.99-1.80 (m, 2H), 1.69-1.50 (m, 2H), 0.92-0.86 (m, 2H), 0.71-0.68 (m, 2H); MS(ES+) m/z 502.2 (M+1); MS(ES-) m/z 500.3 (M-1).
Example 34
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1 -(4-fluoro-2-(trifluoromethyl)benzyl)piperidin- 3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00249##
[0403] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate
##STR00250##
[0405] To a solution of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)piperidine-1-carboxylate (22.50 g 51.80 mmol) in anhydrous methanol (400 mL), was added sulfuric acid (10.0 mL). The reaction mixture was refluxed for 16 hours and then concentrated in vacuo. The pH of the residue was adjusted to 8.about.9 with 1M aqueous sodium hydroxide solution, and extracted with ethyl acetate (2.times.300 mL). Organic layers were combined, washed with saturated sodium bicarbonate (50 mL), brine solution (50 mL), dried over anhydrous sodium sulfate and concentrated invacuo. The crude product was purified by column chromatography (5% to 20% methanol in dichloromethane) afforded the title compound as an oil (10.00 g, 66%): MS(ES+) m/z 294.3 (M+1).
Step 2. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2-(trifluoromethyl)-benzyl)piperid- in-3-yl)oxy)benzoate
##STR00251##
[0407] To a solution of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (0.225 g, 0.768 mmol) in anhydrous dimethylformamide (10 mL) was added potassium carbonate (0.269 g, 1.95 mmol) and 1-(bromomethyl)-4-fluoro-2-(trifluoromethyl)benzene (0.13 mL, 0.84 mmol). The mixture was stirred at ambient temperature for 1 hour, then poured into water (50 mL) and extracted with ethyl acetate (3.times.30 mL). The combined organic layer was washed with water (2.times.30 mL), brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated invacuo. The residue was purified by column chromatography (0 to 30% ethyl acetate in hexanes) to give the title compound (0.286 g, 79%): MS(ES+) m/z 470.2 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2- (trifluoromethyl)benzyl)piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00252##
[0409] To a solution of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2-(trifluoromethyl)- benzyl)piperidin-3-yl)oxy)benzoate (0.525 g, 1.12 mmol) in water and tetrahydrofuran (1:1, 20 mL) was added lithium hydroxide (0.265 g, 11.10 mmol). The mixture was heated to reflux for 2 hours and then stirred for an additional 16 hours at ambient temperature before neutralized with a 1 M aqueous hydrochloric acid solution. The aqueous layer was then extracted with ethyl acetate (3.times.10 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The crude product was used directly for the next step without further purification. To a solution of crude (R)-5-cyclopropyl-2-fluoro-4- ((1 -(4-fluoro-2-(trifluoromethyl)benzyl)piperidin-3-yl)oxy)benzoic acid (0.161 g, 0.354 mmol) in anhydrous dichloromethane (5 mL) was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (0.212 g, 1.11 mmol), 4-dimethylaminopyridine (0.199 g, 1.63 mmol) and methanesulfonamide (0.105 g, mmol). The mixture was stirred at ambient temperature for 32 hours, then diluted with ethyl acetate (50 mL) and washed with a 5% aqueous hydrochloric acid solution (2.times.25 mL). The combined aqueous layers were extracted with ethyl acetate (3.times.50 mL). The combined organic layers were then washed with water (50 mL) and brine (50 mL); dried over anhydrous sodium sulfate; filtered and concentrated in vacuo. The residue was purified by column chromatography (0 to 100% ethyl acetate (containing 0.2% acetic acid) in hexanes) to afford the title compound (0.033 g, 17%). .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (brs, 1H), 7.84-7.79 (m, 1H), 7.58-7.54 (m, 1H), 7.46-7.40 (m, 1H), 7.13 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.2 Hz, 1H), 4.63 (brs, 1H), 3.63 (m, 2H), 3.32 (s, 3H), 2.74-2.70 (m, 1H), 2.44-2.32 (m, 2H), 2.14-2.05 (m, 1H), 1.98-1.74 (m, 3H), 1.66-1.53 (m, 2H), 0.97-0.87 (m, 2H), 0.76-0.67 (m, 3H); .sup.19F NMR (282 MHz, DMSO -d.sub.6) .delta.-114.1, -113.1, -58.4; MS(ES+) m/z 533.2 (M+H).
Example 35
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(4-fluoro-2-(tri- fluoromethyl)-benzyl)piperidin-3-yl)oxy)benzamide
##STR00253##
[0411] Following the procedure as described in example 34 step 3 and making variations as required to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained (0.04 g, 20%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.81 (brs, 1H), 7.83-7.79 (m, 1H), 7.58-7.54 (m, 1H), 7.46-7.40 (m, 1H), 7.12 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.2 Hz, 1H), 4.63 (brs, 1H), 3.62 (m, 2H), 3.11-3.03 (m, 1H), 2.73-2.69 (m, 1H), 2.44-2.32 (m, 1H), 2.16-2.03 (m, 1H), 1.99-1.73 (m, 2H), 1.67-1.53 (m, 2H), 1.23 (s, 2H), 1.13-1.08 (m, 4H), 0.93-0.86 (m, 2H), 0.77-0.65 (m, 2H); .sup.19F NMR (282 MHz, DMSO-d.sub.6) .delta. -114.1, -112.8, -58.4; MS(ES+) m/z 559.2 (M+H).
Example 36
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro- 4-((1-(4-fluoro-2-(trifluoromethyl)benzyl)piperidin-3-yl)oxy)benzamide
##STR00254##
[0413] Following the procedure as described in example 34 step 3 and making variations as required to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.053 g, 26%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.60 (br s, 1H), 7.83-7.78 (m, 1H), 7.58-7.54 (m, 1H), 7.46-7.40 (m, 1H), 7.14 (d, J=8.3 Hz, 1H), 6.99 (d, J=13.1 Hz, 1H), 4.63 (br s, 1H), 4.04 (t, J=7.7 Hz, 4H), 3.63 (m, 2H), 2.73-2.69 (m, 1H), 2.45-2.33 (m, 2H), 2.21-2.06 (m, 3H), 1.96-1.74 (m, 2H), 1.67-1.52 (m, 2H), 1.23 (s, 1H), 0.93-0.89 (m, 2H), 0.79-0.67 (m, 2H), .sup.1F NMR (282 MHz, DMSO-d.sub.6) .delta. -114.1, -113.1, -58.4; MS (ES+) m/z 574.2 (M+H).
Example 37
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro- 4-((1-(4-fluorophenyl)piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR00255##
[0414] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(4-fluorophenyl)piperidin- 3-yl)oxy)benzoate
##STR00256##
[0416] To a mixture (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yl)oxy)benzoate (0.587 g, 2.0 mmol), 4-fluorophenylboronic acid (0.56 g, 4.0 mmol), and copper(II) acetate (0.363 g, 2.0 mmol in anhydrous dichloromethane (8 mL) was added triethylamine (0.56 mL, 4.0 mmol) and the reaction mixture was stirred for 72 hours at ambient temperature under an atmosphere of dry air. The mixture was filtered through a plug of celite, the filter cake was washed with a mixture of dichloromethane and methanol (1:1, 20 mL), and the combined filtrate was concentrated in vacuo. Purification of the residue by column chromatography (0 to 50% ethyl acetate in hexanes) afforded the title compound as a light yellow oil (0.448 g 58%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.42 (d, J=8.4 Hz, 1H), 6.97-6.81 (m, 4H), 6.62 (d, J=12.8 Hz, 1H), 4.54-4.45 (m, 1H), 3.86 (s, 3H), 3.63-3.55 (m, 1H), 3.36-3.26 (m, 1H), 3.01 (dd, J=11.9, 8.0 Hz, 1H), 2.89 (ddd, J=12.0, 9.3, 2.9 Hz, 1H), 2.21-2.09 (m, 1H), 2.04-1.91 (m, 2H), 1.84-1.64 (m, 1H), 1.60-1.53 (m, 1H), 0.90-0.81 (m, 2H), 0.65-0.59 (m, 2H); MS(ES+) m/z 388.3 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorophenyl)piperidin- 3-yl)oxy)benzoic acid
##STR00257##
[0418] To a mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(4-fluorophenyl)piperidin-3- yl)oxy)benzoate (0.448 g, 1:16 mmol) in tetrahydrofuran (10 mL) was added a solution of lithium hydroxide (0.139 g, 5.8 mmol) in water (3 mL). The reaction mixture was stirred for 16 hours at ambient temperature and subsequently for 1 hour at 60.degree. C. After cooling to ambient temperature, the reaction mixture was adjusted to pH 1 with 1 N hydrochloric acid solution and extracted with dichloromethane (3.times.20 mL). The combined organic phase was washed with brine (5 mL), dried over anhydrous sodium sulfate, and filtered. Concentration of the filtrate in vacuo gave the title compound as a yellowish oil (0.43 g, 99%), which was used without further purification; MS(ES+) m/z 374.3 (M+1).
Step 3. Preparation of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-(4-fluoropheny- l)piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR00258##
[0420] To a mixture of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluorophenyl)piperidin-3-yl)oxy)benzo- ic acid (0.215 g, 0.58 mmol), N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (0.167 g, 0.87 mmol), and 4-(dimethylamino)pyridine (0.213 g, 1.74 mmol) in anhydrous dichloromethane was added azetidine-1-sulfonamide (0.119 g, 0.87 mmol). The reaction mixture was stirred for 48 hours at ambient temperature. The mixture was diluted with ethyl acetate (100 mL) and then quenched by addition of 1 N hydrochloric acid solution (10 mL). The organic phase was washed with 1 N hydrochloric acid solution (5 mL), water (5 mL) and brine (5 mL); dried over anhydrous sodium sulfate and filtered. Concentration of the filtrate gave a residue which was purified first by column chromatography (0 to 50% ethyl acetate in hexanes) and then by reverse-phase preparative HPLC to afford the title compound as an off-white solid (0.168 g, 48%): .sup.1H NMR (300 MHz:, DMSO-d.sub.6) .delta. 11.61 (s, 1H), 7.30 (brs, 1H), 7.18-6.93 ((m, 6H), 4.78-4.68 (m, 1H), 4.04 (t, J=7.7, 7.7 Hz, 4H), 3.52 (dd, J=12.2, 2.7 Hz, 1H), 3.31-3.18 (m, 2H), 3.13-3.03 (m, 1H), 2.23-2.10 (m, 2H), 2.10-1.99 (m, 1H), 1.99-1.84 (m, 2H), 1.76-1.61 (m, 2H), 0.86-0.77 (m, 2H), 0.70-0.62 (m, 2H); .sup.19F NMR (282 MHz, DMSO-d.sub.6) .delta. -75.0, -112.9, -125.2; MS(ES-) m/z 490.3 (M-1).
Example 38
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1- (4-fluorophenyl)piperidin-3-yl)oxy)benzamide
##STR00259##
[0422] Following the procedure as described in Example 37 Step 3 and making variations as required to replace azetidine-1-sulfonamide with cyclopropanesulfonamide and purification by column chromatography (0 to 50% ethyl acetate in hexanes), the title compound was obtained as a colorless solid (0.177 g, 64%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (s, 1H), 7.18-6.89 (m, 6H), 4.77-4.66 (m, 1H), 3.50 (dd, J=12.3, 2,9 Hz, 1H), 3.29-3.15 (m, 2H), 3.13-3.01 (m, 2H), 2.09-1.99 (m, 1H), 1.97-1.81 (m, 2H), 1.75-1.60 (m, 2H), 1.16-1.06 (m, 4H), 0.86-0.77 (m, 2H), 0.69-0.61 (m, 2H); .sup.19F NMR (282 MHz, DMSO-d.sub.6) .delta. -112.63, -125.87; MS(ES-) m/z 475.3 (M-1).
Example 39
Synthesis of (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3- yl)oxy)-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00260##
[0423] Step 1. Preparation of (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin- 3-yl)oxy)-2-fluorobenzoic acid
##STR00261##
[0425] To a mixture of (R)-methyl 5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)- 2-fluorobenzoate (0.15 g, 0.35 mmol) in tetrahydrofuran (20 mL) and water (20 mL) was added lithium hydroxide monohydrate (0.15 g, 3.5 mmol). The reaction mixture was heated to reflux for 4 hours. The reaction mixture was diluted with ethyl acetate (80 mL), washed with 1 M hydrochloric acid solution (50 mL) and brine (2.times.50 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to provide the title compound as a solid (0.15 g, quant.); MS(ES+) m/z 414.2, 416.2 (M+1).
Step 2. Preparation of (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-N-(cyclop- ropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00262##
[0427] A mixture of (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid (0.15 g, 0.35 mmol), cyclopropanesulfonamide (0.064 g, 0.53 mmol), N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (0.10 g, 0.53 mmol), and 4-dimethylaminopyridine (0.13 g, 1.05 mmol) in dichloromethane (20 mL) was stirred at ambient temperature for 18 hours. The reaction was concentrated in vacuo and the residue was first purified by flash chromatography (0 to 4% methanol in dichloromethane), then by reverse phase HPLC (acetonitrile in water+0.1% TFA) to provide the title compound (0.03 g, 17%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.03 (brs, 1H), 9.45 (brs 1H), 7.77 (d, J=7.6 Hz, 1H), 7.74-7.64 (m, 1H), 7.60-7.49 (m, 1H), 7.41-7.26 (m, 2H), 4.85-4.67 (m, 1H), 4.53-3.99 (m, 5H), 3.43-3.29 (m, 1H), 3.18-2.99 (m, 2H), 2.01-1.88 (m, 1H), 1.81-1.56 (m, 2H), 1.15-1.04 (m, 4H); MS (ES+) m/z 519.1, 521.1 (M+H).
Example 40
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-phenylpiperidin- 3-yl)oxy)benzamide
##STR00263##
[0428] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-3-yl)oxy)benzoate
##STR00264##
[0430] To a degassed mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (0.76 g, 2.68 mmol), iodobenzene (1.49 mL, 13.4 mmol), L-proline (0.62 g, 5.36 mmol), and potassium carbonate (1.88 g, 13.4 mmol) in anhydrous dimethylsulfoxide (30 mL) was added copper (1) iodide (0.51 g, 2.68 mmol). The resulting mixture was heated to 15.degree. C. under nitrogen for 2 hours. And then iodobenzene (1.0 mL, 9.0 mmol) was added to the reaction mixture stirring was continued at 75.degree. C. under nitrogen for 24 hours. The reaction mixture was diluted with ethyl acetate (100 mL), washed with water (50 mL), saturated ammonium chloride (3.times.50 mL) and brine (50 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (R.sub.f=0.2 in 9:1 hexanes:ethyl acetate) to provide the title compound as an oil (0.74 g, 75%): .sup.1H NMR (300 MHz, CDCl.sub.6) .delta. 7.46-7.39 (m, 1H), 7.31-7.17 (m, 3H), 6.95-6.88 (m, 1H), 6.86-6.79 (m, 1H), 6.63 (d, J=12.8 Hz, 1H), 4.55-4.44 (m, 1H), 3.87 (s, 3H), 3.76-3.68 (m, 1H), 3.48-3.39 (m, 1H), 3.10 (dd, J=13.1, 8.0 Hz, 1H), 3.03-2.93 (m, 1H), 2.21-2.12 (m, 1H), 2.03-1.91 (m, 2H), 1.56-1.66 (m, 2H), 0.89-0.82 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 370.2 (M+H).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-3-yl)oxy)benzoic acid
##STR00265##
[0432] Following the procedure as described in Example 39 step 1 and making variation as required to replace (R)-methyl 5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorobenzo- ate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-3-yl)oxy)benzoate, the title compound was obtained as a colorless solid (0.61 g, 86%): MS(ES+) m/z 356.2 (M+H).
Step 3. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-phenylpiperidin-- 3-yl)oxy)benzamide
##STR00266##
[0434] Following the procedure as described in Example 39 step 2 and making variation as required to replace (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-3-yl)oxy)benzoic acid and purification by flash chromatography [(R.sub.f=0.2 in 2.1 hexanes:ethyl acetate (containing 0.2% acetic acid)], the title compound was obtained as a colorless solid (0.075 g, 18%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.78 (brs, 1H), 7.17-7.02 (m, 4H), 6.90-6.84 (m, 2H), 6.72-6.65 (m, 1H), 4.72-4.64 (m, 1H), 3.56-3.48 (m, 1H), 3.29-3.20 (m, 2H), 3.15-3.07 (m, 1H), 3.07-2.98 (m, 1H), 2.05-1.96 (m, 1H), 1.91-1.79 (m, 2H), 1.73-1.58 (m, 2H), 1.11-1.03 (m, 4H), 0.79-0.72 (m, 2H), 0.63-0.56 (m, 2H); MS(ES+) m/z 459.2 (M+H).
Example 41
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-phenylpiperidi- n- 3-yl)oxy)benzamide
##STR00267##
[0436] Following the procedure as described in Example 39 step 2 and making variations as required to replace (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-3-yl)oxy)benzoic acid and cyclopropanesulfonamide with 1-azetidinesulfonamide and purification by flash chromatography [(R.sub.f=0.2 in 2:1 hexanes:ethyl acetate (containing 0.2% acetic acid)], the title compound was obtained as a colorless solid (0.08 g, 20%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.57 (brs, 1H), 7.18-7.03 (m, 4H), 6.91-6.49 (m, 2H), 6.72-6.65 (m, 1H), 4.72-4.63 (m, 1H), 4.00 (t, J=7.6 Hz, 4H), 3.56-3.48 (m, 1H), 3.28-3.19 (m, 2H), 3.15-3.05 (m, 1H), 2.12 (quintet, J=7.6 Hz, 2H), 2.05-1.95 (m, 1H), 1.91-1.78 (m, 2H), 1.73-1.58 (m, 2H), 0.80-0.71 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 474.25 (M+H).
Example 42
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,5-dichlorophenyl)piper- idin- 3-yl)oxy)-2-fluorobenzamide
##STR00268##
[0437] Step 1. Preparation of (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorobenzo- ate
##STR00269##
[0439] Following the procedure as described in Example 40 step 1 and making variation as required to replace iodobenzene with 1,3-dichloro-5-iodobenzene, the title compound was obtained as a colorless oil (1.08 g, 47%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.39 (d, J=8.3 Hz, 1H), 6.74-6.69 (m, 3H), 6.57 (d, J=12.7 Hz, 1H), 4.52-4.43 (m, 1H), 3.86 (s, 3H), 3.59 (dd, J=12.8, 3.07 Hz, 1H), 3.39-3.29 (m, 2H), 3.24-3.12 (m, 1H), 2.15-2.04 (m, 1H), 2.00-1.91 (m, 1H), 1.90-1.78 (m, 2H), 1.75-1.62 (m, 1H), 0.86-0.77 (m, 2H), 0.60-0.53 (m, 2H); MS(ES+) m/z 438.2, 440.2 (M+H).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3- yl)oxy)-2-fluorobenzoic acid
##STR00270##
[0441] Following the procedure as described in Example 39 step 1 and making variation as required to replace (R)-methyl 5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorobenzo- ate with (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorobenzo- ate, the title compound was obtained as a colorless solid (1.04 g, quant.); MS(ES+) m/z 424.2, 426.2 (M+H).
Step 3. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1- (3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorobenzamide
##STR00271##
[0443] Following the procedure as described in Example 39 step 2 and making variation as required to replace (R)-5-chloro-4-((1 -(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorobenzoic acid with (R)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid and purification by flash chromatography [R.sub.f=0.25 in 2:1 hexanes:ethyl acetate (containing 0.2% acetic acid)), the title compound was obtained as a colorless solid (0.175 g, 47%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.77 (brs, 1H), 7.06 (d, J=13.2 Hz, 1H), 7.01 (d, J=8.3 Hz, 1H), 6.85 (d, J=1.8 Hz, 2H), 6.69 (dd, J=1.7, 1.7 Hz, 1H), 4.73-4.65 (m, 1H), 3.73-3.63 (m, 1H), 3.57-3.49 (m, 1H), 3.48-3.39 (m, 2H), 3.27-3.21 (m, 1H), 3.08-2.97 (m, 1H), 2.00-1.88 (m, 1H), 1.84-1.71 (m, 1H), 1.67-1.59 (m, 1H), 1.57-1.46 (m, 1H), 1.13-1.03 (m, 4H), 0.70-0.60 (m, 2H), 0.57-0.48 (m, 2H); MS(ES+) m/z 527.1, 529.1 (M+H).
Example 43
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)pip- eridin-3-yl)oxy-2-fluorobenzamide
##STR00272##
[0445] Following the procedure as described in Example 39 step 2 and making variation as required to replace (R)-5-chloro-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid and cyclopropanesulfonamide with 1-azetidinesulfamide and purification by flash chromatography (R.sub.f=0.25 in 2:1 hexanes:ethyl acetate (+0.2% acetic acid)), the title compound was obtained as a colorless solid (0.16 g, 42%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.55 (brs, 1H), 7.09-7.00(m, 2H), 6.86 (d, J=1.7 Hz, 2H), 6.71-6.68 (m, 1H), 4.72-4.65 (m, 1H), 3.99 (t, J=7.7 Hz, 4H), 3.67 (dd, J=13.7, 5.5 Hz, 1H), 3.52 (dd, J=13.7, 2.3 Hz, 1H), 3.48-3.39 (m, 1H), 3.27-3.21 (m, 1H), 2.11 (p, J=7.7 Hz, 2H), 1.99-1.88 (m, 1H), 1.84-1.71 (m, 2H), 1.68-1.59 (m, 1H), 1.59-1.46 (m, 1H), 0.69-0.61 (m, 2H), 0.57-0.50 (m, 2H); MS (ES+) m/z 542.2, 544.1 (M+H).
Example 44
Synthesis of (R)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00273##
[0447] Following the procedure as described in Example 3 step 2 and making variations as required to replace (R)-5-chloro-4-((1 -(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-2-fluorobenzoic acid with (R)-5-cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid and cyclopropanesulfonamide with methanesulfonamide and purification by flash chromatography (R.sub.f=0.5 in 2:1 hexanes:ethyl acetate (+0.2% acetic acid)), the title compound was obtained as a colorless solid (0.15 g 42%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.83 (brs 1H), 7.08-6.99 (m, 2H), 6.85 (d, J=1.7 Hz, 2H), 6.70-6.68 (m, 1H), 4.72-4.65 (m, 1H), 3.68 (dd, J=13.7, 5.4 Hz, 1H), 3.53 (dd, J=13.7, 2.4 Hz, 1H), 3.48-3.38 (m, 1H), 3.30 (s, 3H), 3.26-3.21 (m, 1H), 1.99-1.88 (m, 1H), 1.83-1.70 (m, 2H), 1.69-1.59 (m, 1H), 1.57-1.45 (m, 1H), 0.69-0.61 (m, 2H), 0.57-0.49 (m, 2H); MS(ES+) m/z 501.1, 503.1 (M+H)
Example 45
Synthesis of (S)-N-(azetidin-1-ylsulfonyl)-4-((1-benzylpyrrolidin-3-yl)oxy)-5-cyclopro- pyl-2-fluorobenzamide
##STR00274##
[0449] Following the procedure as described in Example 3 steps 1 to 5, and making variations as required to replace (R)-tert-butyl 3-hydroxypiperidine-1-carboxylate with (S)-tert-butyl 3-hydroxypyrrolidine-1-carboxylate, 3,5-dichlorobenzaldehyde with benzaldehyde and cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.027 g, 5%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 7.40-7.17 (m, 5H), 7.13 (d, J=8.8 Hz, 1H), 6.69 (d, J=12.61 Hz, 1H), 5.00-4.87 (m, 1H), 3.86-3.71 (m, 4H), 3.65-3.59 (m, 2H), 2.98-2.84 (m, 1H), 2.78-2.56 (m, 3H), 2.37-1.77 (m, 5H), 0.92-0.78 (m, 2H), 0.62-0.52 (m, 2H); MS(ES+) m/z 474.2 (M+1); MS(ES-) m/z 472.3 (M-1).
Example 46
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-benzylpyrrolidin-3-yl)oxy)-5-cyclopro- pyl-2-fluorobenzamide
##STR00275##
[0451] Following the procedures as described in Example 3 steps 1 to 5, and making variations as required to replace (R)-tert-butyl 3-hydroxypiperidine-1-carboxylate with (R)-tert-butyl 3-hydroxypyrrolidine-1-carboxylate, 3,5-dichlorobenzaldehyde with benzaldehyde and cyclopropylsulfonamide with azetidine-1-sulfonamide, the title compound was obtained (0.095 g, 27% ): .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 7.45-7.18 (m, 5H), 7.10 (d, J=8.3 Hz, 1H), 6.82 (d, J=12.7 Hz, 1H), 5.07-4.94 (m, 1H), 3.98-3.87 (m, 4H), 3.76 (s, 2H), 3.06 (dd, J=11.0, 5.8 Hz, 1H), 2.91-2.74 (m, 2H), 2.72-2.57 (m, 1H), 2.34 (dt, J=13.9, 7.1 Hz, 1H), 2.17-1.94 (m, 3H), 1.93-1.79 (m, 1H), 0.90-0.77 (m, 2H), 0.67-0.56 (m, 2H); MS(ES+) m/z 474.2 (M+1); MS(ES-) m/z 472.3 (M-1)
Example 47
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((8-(2-chlorobenzyl)-8-azabicyclo[3.2.1]octan- -3-yl)methoxy)-5-cyclopropyl-2-fluorobenzamide
##STR00276##
[0452] Step 1. Preparation of benzyl 3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR00277##
[0454] To a solution of 8-Azabicyclo[3.2.1]octan-3-ylmethanol (1.00 g, 7.09 mmol) in dichloromethane (15 mL) and a saturated aqueous sodium bicarbonate solution (10 mL) at 0.degree. C. was added benzyl chloroformate (1.26 g, 7.45 mmol) and the reaction mixture was stirred at 0.degree. C. for 30 min. The reaction mixture was then extracted with dichloromethane (3.times.100 mL). The organic layers were combined and washed with brine (150 mL); dried over anhydrous sodium sulfate, filtered and concentrated to give the title compound, which was used directly for the next step. (1.50 g, 73%). .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.43-7.24 (m, 5H), 5.15-5.09 (m, 2H), 4.59-4.57 (m, 1H), 4.40-4.27 (m, 2H), 3.41 (m, 2H), 2.14-1.90 (m, 3H), 1.88-1.80 (m, 1H), 1.73-1.48 (m, 4H).
Step 2. Preparation of benzyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)- 8-azabicyclo[3.2.1]octane-8-carboxylate
##STR00278##
[0456] To a solution of benzyl 3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate (1.92 g, 7.00 mmol) in anhydrous dimethylsulfoxide (15 mL) was added cesium carbonate (5.69 g, 10.50 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (1.82 g, 7.35 mmol). The reaction mixture was stirred at 70.degree. C. for 16 hours; cooled to ambient temperature and acidified to pH=1 with 5% aqueous hydrochloric acid solution and extracted with ethyl acetate (2.times.15 mL), the combined organics were washed with brine (15 mL); dried over anhydrous sodium sulfate; filtered and concentrated in vacuo. Purification of the residue by column chromatography (0 to 10% gradient of ethyl acetate in hexanes) afforded the title compound (2.00 g, 54%): .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.85 (d, J=7.6 Hz, 1H), 7.39-7.26 (m, 5H), 6.56 (d, J=12.1 Hz, 1H), 5.13(s, 2H), 4.46-4.28 (m, 2H), 3.81-3.73 (m, 2H), 2.54-2.32 (m, 1H), 2.03-1.96 (m, 4H), 1.79-1.65 (m, 4H), 1.60-1.51 (m, 9H); MS(ES+) m/z 504.2, 506.2 (m+1).
Step 3. Preparation of benzyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)- 8-azabicyclo[3.2.1]octane-8-carboxylate.
##STR00279##
[0458] To a solution of benzyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)- methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate (1.50 g, 2.98 mmol), cyclopropylboronic acid (0.38 g, 4.46 mmol), potassium phosphate (2.10 g, 5.95 mmol) and tricyclohexylphosphine tetrafluoroborate (0.22 g, 0.60 mmol) in toluene (15 mL) and water (1.5 mL) under a nitrogen atmosphere was added palladium acetate (0.06 g, 0.31 mmol). The reaction mixture was heated at reflux for 16 hours, and then cooled to ambient temperature. Water (50 mL) was added and the mixture was extracted with diethyl ether (2.times.100 mL), the combined organics were washed with brine (30 mL); dried over anhydrous sodium sulfate and concentrated in vacuo. Purification of the residue by column chromatography (10 to 30% gradient of ethyl acetate in hexanes) afforded the title compound (1.05 g, 66%). .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=8.4 Hz, 1H), 6.58 (d, J=12.9 Hz, 1H), 3.87 (dd, J=6.9, 2.3 Hz, 2H), 2.17-1.91 (m, 3H), 1.76-1.31 (m, 8H), 0.98-0.84 (m, 5H), 0.67-0.58 (m, 2H).
Step 4. Preparation of benzyl 3-((4-((azetidin-1-ylsulfonyl)carbamoyl)-2-cyclopropyl- 5-fluorophenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate.
##STR00280##
[0460] To a stirred solution of benzyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate (0.56 g, 1.10 mmol) in dichloromethane (5 mL) at 0.degree. C. was added trifluoroacetic acid (1 mL) and the mixture was stirred for 1.5 hours at ambient temperature and then concentrated. The residue was further concentrated 2 times with toluene (5 mL) and then diluted with dichloromethane (5 mL). To this solution was added N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (0.426 g, 1.65 mmol) and 4-(dimethylamino)pyridine (0.335 g, 2.75 mmol) and azetidine-1-sulfonamide (0.165 g, 1.21 mmol). The reaction mixture was stirred at room temperature for 16 hours and then diluted with dichloromethane (10 mL) and washed with aqueous hydrochloric acid (1M, 10 mL). The aqueous layer was extracted with dichloromethane (10 mL); the organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by column chromatography (0 to 15% gradient of methanol, containing 1% NH.sub.3 in dichloromethane) to give the title compound (460 mg, 73%): MS(ES+) m/z 572.2 (M+1 ); MS(ES-) m/z 570.2 (M-1).
Step 5. Preparation of 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-N-(azetidin-1-ylsulfonyl)-5-cycl- opropyl-2-fluorobenzamideate.
##STR00281##
[0462] To a stirred solution of benzyl 3-((4-((azetidin-1-ylsulfonyl)carbamoyl)-2-cyclopropyl- 5-fluorophenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate (0.46 g, 0.81 mmol) in degassed ethyl acetate was added palladium 20% on carbon (0.050 g). The reaction mixture was stirred for 2 hours under an atmosphere of hydrogen. The reaction mixture was then filtered over a plug of silica gel and rinsed (2.times.15 mL) with a solution of 20% methanol and 2% acetic acid in dichloromethane. The filtrate was concentrated to give the title compound (0.2 g, 56%). .sup.1H NMR (300 MHz, MeO-d.sub.4) .delta. 7.37-7.13 (m, 1H), 6.87-6.61 (m, 1H), 4.33-3.73 (m, 1H), 3.38-3.23 (m, 1H), 2.68-1.64 (m, 2H), 1.03-0.75 (m, 2H), 0.69-0.51 (m, 2H).
Step 6. Preparation of N-(azetidin-1-ylsulfonyl)-4-((8-(2-chlorobenzyl)-8- azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzamide
##STR00282##
[0464] To a stirred solution of 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-N-(azetidin-1- ylsulfonyl)-5-cyclopropyl-2-fluorobenzamideate (50 mg, 0.11 mmol) in tetrahydrofuran (1 mL) under an atmosphere of nitrogen were introduced 2-chlorobenzaldehyde (19 mg, 0.14 mmol) and sodium triacetoxyborohydride (66 mg, 0.21 mmol) and the mixture was stirred for 16 hours. 1N aqueous hydrochloric acid (5 mL) was added and the mixture was extracted with ethyl acetate (3.times.10 mL) and concentrated. The residue was purified by chromatography eluting with 5% methanol in dichloromethane to give the title compound, which was lyophilized to give a white solid (0.035 g, 29%). .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 7.71 (d, J=6.5 Hz, 1H), 7.44 (d, J=7.3 Hz, 1H), 7.40-7.28 (m, 2H), 7.12 (d, J=8.3 Hz, 1H), 6.86 (d, J=12.7 Hz, 1H), 4.00-3.79 (m, 8H), 3.55-3.42 (m, 2H), 2.33-1.94 (m, 6H), 1.83-1.60 (m, 6H), 0.90-0.80 (m, 2H), 0.68-0.56 (m, 2H). MS(ES+) m/z 562.2, 564.2 (M+1); MS(ES-) m/z 560.3, 562.3 (M-1).
Example 48
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((8-benzyl-8-azabicyclo[3.2.1]octan-3-yl)meth- oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00283##
[0466] Following the procedure as described in Example 47 step 6, and making variation as required to replace 2-chlorobenzaldehyde with benzyaldehyde, the title compound was obtained as a white solid (0.025 g, 41%). .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 7.66-7.52 (m, 2H), 7.47-7.36 (m, 3H), 7.22-7.12 (m, 1H), 6.82-6.71 (m, 1H), 4.18-4.01 (m, 2H), 3.97-3.62 (m, 8H), 2.42-2.16 (m, 3H), 2.14-1.71 (m, 9H), 0.91 -0.76 (m, 2H), 0.63-0.52 (m, 2H); MS(ES+) m/z 528.2 (M+1).
Example 49
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((8-benzhydryl-8-azabicyclo[3.2.1]-octan-3-yl- )methoxy)-5-cyclopropyl-2-fluorobenzamide
##STR00284##
[0468] To a stirred solution of 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-N-(azetidin-1- ylsulfonyl)-5-cyclopropyl-2-fluorobenzamideate (0.05 g, 011 mmol) in acetonitrile (2 mL) under an atmosphere of nitrogen was added (bromomethylene)dibenzene (0.034 g, 0.14 mmol), potassium carbonate (0.038 g, 0.27 mmol) and sodium iodide (0.021 g, 0.14 mmol). The reaction mixture was stirred at reflux for 16 hours and then cooled to ambient temperature. 1N aqueous hydrochloric acid (5 mL) was added and the mixture was extracted with ethyl acetate (3.times.10 mL) and concentrated. The residue was first purified by column chromatography eluting with 30% ethyl acetate (containing 1% formic acid) in hexanes and then purified by reverse phase chromatography eluting with a gradient of acetonitrile in water (containing 0.1% trifluoroacetic acid) and finally crystallized in isopropyl alcohol to give the title compound (0.03 g, 43%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) d 12.18-11.67 (m, 1H), 8.75-8.53 (m, 1H), 7.98-7.76 (m, 4H), 7.61-7.49 (m, 1H), 7.47-7.30 (m, 6H), 6.59 (d, J=14.1 1H), 4.77-4.65 (m, 1H), 4.28-4.17 (m, 4H), 4.07-3.98 (m, 2H), 3.97-3.87 (m, 2H), 2.90-2.56 (m, 3H), 2.51-2.34 (m, 3H), 2.24 (m, 2H), 2.12-2.01 (m, 2H), 1.95-1.81 (m, 2H), 0.90-0.76 (m, 2H), 0.67-0.55 (m, 2H); MS(ES+) m/z 564.3 (M+1); MS(ES-) m/z 562.3.
Example 50
Synthesis of (5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin-3- yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00285##
[0469] Step 1. Preparation of methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)- piperidin-3-yl)oxy)-2-fluorobenzoate and methyl 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)o- xy)-2-fluorobenzoate
##STR00286##
[0471] To a mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (0.52 g, 1.77 mmol), potassium carbonate (0.73 g, 5.30 mmol) and sodium iodide (0.26 g, 1.77 mmol) in acetonitrile (50 mL) was added 1,3-dichloro-5-(1-chloroethyl)benzene (0.37 g, 1.77 mmol). The reaction mixture was heated at reflux for 16 hours, and concentrated in vacuo. To the he residue was added 25% aqueous ammonium chloride solution (40 mL) and extracted with ethyl acetate (2.times.70 mL). The combined organic layer was washed with brine (40 mL), dried over anhydrous sodium sulfate and concentrated in vacuo. The residue was purified by column chromatography (0-20% ethyl acetate in hexanes) afforded the title compound. The first eluting fraction was arbitrarily assigned as methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl)oxy)-2-fluorobenzoate (0.18 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.42 (d, J=8.4 Hz, 1H), 7.22-7.21 (m, 3H), 6.54 (d, J=12.9 Hz, 1H), 4.39-4.37 (m, 1H), 3.87 (s, 3H), 3.47 (q, J=1 6.6 Hz, 1H), 2.97-2.94 (m, 1H), 2.57-2.54 (m, 1H), 2.35-2.23 (m, 2H), 2.11-1.98 (m, 2H), 1.85-1.77 (m, 1H), 1.65-1.51 (m, 2H), 1.31 (d, J=6.7 Hz, 3H), 0.94-0.90 (m, 2H), 0.68-0.63 (m, 2H); MS(ES+)m/z 466.1, 468.1 (M+1). The second eluting fraction was arbitrarily assigned methyl 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)o- xy)-2-fluorobenzoate (0.18 g, 22%): .sup.1H NMR (300 Hz, CDCl.sub.3) .delta. 7.42 (d, J=8.4 Hz, 1H), 7.22-7.21 (m, 3H), 6.54 (d, J=12.9 Hz, 1H), 4.39-4.37 (m, 1H), 3.87 (s, 3H), 3.47 (q, J=1 6.6 Hz, 1H), 2.97-2.94 (m, 1H), 2.57-2.54 (m, 1H), 2.35-2.23 (m, 2H), 2.11-1.98 (m, 2H), 1.85-1.77 (m, 1H), 1.65-1.51 (m, 2H), 1.31 (d, J=6.7 Hz, 3H), 0.94-0.90 (m, 2H), 0.68-0.63 (m, 2H); MS(ES+) m/z 466.1, 468.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3-yl)- oxy)-2-fluorobenzoic acid
##STR00287##
[0473] To a mixture of 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3-yl)- oxy-2-fluorobenzoate (0.20 g, 0.43 mmol) in tetrahydrafurane (30 mL) and water (5 mL) was added lithium hydroxide (0.10 g, 4.3 mmol). The reaction mixture was heated to reflux for 16 hours and adjusted pH to 7 with 1N aqueous hydrochloric acid solution, extracted with ethyl acetate (2.times.50) mL), the combined organics were washed with 25% aqueous ammonium chloride solution (2.times.30 mL), dried over anhydrous sodium sulfate and concentrated in vacuo. Purification of the residue by column chromatography (20%-100% ethyl acetate in hexanes) afforded the title compound (0.15 g, 77%): MS(ES+) m/z 452.1, 454.1 (M +1); MS(ES-) m/z 450.2, 452.2(M-1).
Step 3. Preparation of (5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3-yl- )oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00288##
[0475] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)-ethyl)piperidin-3- yl)oxy)-2-fluorobenzoic acid, the title compound was obtained as colorless solid (0.05 g, 33%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (brs, 1H), 7.46-7.37 (m, 3H), 7.13 (d, J=8.4 Hz, 1H), 6.95 (d, J=13.2 Hz, 1H), 4.59-4.57 (m, 1H), 3.63-3.62 (m, 1H), 3.28 (s, 3H), 2.77-2.73 (m, 1H), 2.43-2.33 (m, 3H), 2.13-2.03 (m, 1H), 2.18-1.73 (m, 2H), 1.55-1.53 (m, 2H), 1.27 (d, J=6.7 Hz, 3H), 0.91-0.88 (m, 2H), 0.73-0.66 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1); MS(ES-) m/z 527.2, 529.2 (M-1).
Example 51
Synthesis of (5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)- oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00289##
[0476] Step 1. Preparation of 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5 -dichlorophenyl)ethyl)-piperidin-3-yl)oxy)-2-fluorobenzoic acid
##STR00290##
[0478] Following the procedure as described in Example 50 step 2 and making variations as required to replace 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl)oxy)-2-fluorobenzoate with 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)- piperidin-3-yl)oxy)-2-fluorobenzoate, the title compound was obtained as beige color solid (0.18 g, 2%): MS(ES+) m/z 452.2, 454.2 (M+1); MS(ES-) m/z 450.2, 452.2 (M-1).
Step 2. Preparation of (5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3-yl- )oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00291##
[0480] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3- yl)oxy)-2-fluorobenzoic acid, the title compound was obtained as colorless solid (0.06 g, 30%): .sup.1H NMR (300 MHz, DMSO.sub.6) .delta. 11.87 (brs, 1H), 7.47-7.40 (m, 3H), 7.12 (d, J=8.4 Hz, 1H), 6.96 (d, J=13.2 Hz, 1H), 4.58-4.56 (m, 1H), 3.68 (q, J=6.3 Hz, 1H), 3.29 (s, 3H), 2.80-2.77 (m, 1H), 2.60-2.56 (m, 1H), 2.37-2.20 (m, 2H), 2.08-2.03 (m, 1H), 1.91-1.74 (m, 2H), 1.57-1.46 (m, 2H), 1.27 (d, J=6.7 Hz, 3H), 0.90-0.87 (m, 2H), 0.75-0.63 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1); MS(ES-) m/z 527.2, 529.2 (M-1).
Example 52
Synthesis of 5-((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cycl- opropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00292##
[0481] Step 1. Preparation of (2R,5R)-benzyl 5-hydroxy-2-methylpiperidine-1-carboxylate
##STR00293##
[0483] To a cooled (0.degree. C.) solution of (3R,6R)-6-methylpiperidin-3-ol (1.06 g, 9.19 mmol) (Ian A. O'Neil et al., Synlett, 2000, 5, 695) and triethylamine (1.35 mL, 9.65 mmol) dichloromethane (60 mL) was added benzyl chloroformate (1.38 mL, 9.65 mmol) dropwise. The reaction mixture was stirred at ambient temperature for 16 h, diluted with aqueous saturated ammonium chloride solution (35 mL), and extracted with ethyl acetate (3.times.70 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous magnesium sulfate, filtered and concentrated in vacuo. The residue was purified by column chromatography eluting with a gradient of ethyl acetate in hexanes (0 to 60%) to give the title compound (1.30 g, 57%) as a colorless oil: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.40-7.29 (m, 5H), 5.14 (s, 2H), 4.57-4.45 (m, 1H), 4.13-4.02 (m, 1H), 3.94 (s, 1H), 3.18-3.03 (m, 1H), 2.19-2.03 (m, 1H), 1.88-1.62 (m, 3H), 1.39-1.26 (m, 1H), 1.19-1.13 (m, 3H); MS(ES+) m/z 250.2 (M+1).
Step 2. Preparation of (2R,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)- 2-methylpiperidine-1-carboxylate
##STR00294##
[0485] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (2R,5R)-benzyl-5-hydroxy-2-methylpiperidine-1-carboxylate, the title compound was obtained (1.80 g, 72%) as a colorless oil: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.84-7.77 (m, 1H), 7.32-7.08 (m, 5H), 6.64-6.53 (m, 1H), 5.09-4.92 (m, 2H), 4.69-4.56 (m, 1H), 4.46 (s, 1H), 4.39-4.29 (m, 1H), 3.19-3.07 (m, 1H), 2.32-2.16 (m, 1H), 2.01-1.89 (m, 2H), 1.56 (s, 9H), 1.41-1.31 (m, 1H), 1.24-1.18 (m, 3H); MS(ES+) m/z 478.2 (M+1).
Step 3. Preparation of (2R,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-methylpiperid- ine-1-carboxylate
##STR00295##
[0487] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (2R,5R)-benzyl 5(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)- 2-methylpiperidine-1-carboxylate, the title compound was obtained (1.62 g, 89%) as a colorless oil: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.39-7.32 (m, 1H), 7.30-7.03 (m, 5H), 6.52-6.41 (m, 1H), 5.06-4.92 (m, 2H), 4.68-4.55 (m, 1H), 4.46 (s, 1H), 4.42-4.29 (m, 1H), 3.20-3.07 (m, 1H), 2.27-2.08(m, 1H), 2.04-1.82 (m, 3H), 1.56 (s, 9H), 1.42-1.31 (m, 1H), 1.24-1.19 (m, 3H), 0.83-0.73 (m, 2H), 0.58-0.47 (m) 2H); MS(ES+) m/z 484.3 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6R)-6-methylpiperidin- 3-yl)oxy)benzoate
##STR00296##
[0489] To a solution of (2R,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-methylpiperid- ine-1-carboxylate (1.62 g, 3.35 mmol) in ethyl acetate (15 mL) and methanol (30 mL) was added 10% palladium on carbon (0.5 g). The reaction mixture was stirred at ambient temperature under hydrogen atmosphere using a balloon for 1 h and filtered through a pad of Celite. The filtrate was concentrated in vacuo to give the title compound (0.94 g, 80%) as colorless oil: MS(ES+) m/z 350.3 (M+1).
Step 5. Preparation of tert-butyl-4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)piperidin-3- yl)oxy)-5-cyclopropyl-2-fluorobenzoate
##STR00297##
[0491] Following the procedure as described in Example 34 step 2, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yl)oxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6R)-6-methylpiperidin-3-yl)oxy)benzoate, and to replace 1-(bromomethyl)-4-fluoro-2-(trifluoromethyl)benzene with 1-(bromomethyl)-2- chloro-4-fluorobenzene, the title compound was obtained (1.29 g, 98%) as a colorless oil: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.58-7.48 (m, 1H), 7.36-7.29 (m, 1H), 7.11-7.03 (m, 1H), 7.00-6.90 (m, 1H), 6.50-6.39 (m, 1H), 4.36-4.23 (m, 1H), 4.05-3.92 (m, 1H), 3.43-3.32 (m, 1H), 3.10-2.99 (m, 1H), 2.58-2.41 (m, 1H), 2.29-2.11 (m, 2H), 2.04-1.83 (m, 2H), 1.64-1.39 (m, 11H), 1.21-1.10 (m, 3H), 0.94-0.80 (m, 2H), 0.65-0.55 (m, 2H); MS(ES+) m/z 492.2, 494.4 (M+1).
Step 6. Preparation of 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)- 5-cyclopropyl-2-fluorobenzoic acid
##STR00298##
[0493] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with of tert-butyl 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3- yl)oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained (1.10 g, 97%) as a colorless solid; MS(ES+) m/z 436.2, 438.2 (M+1).
Step 7. Preparation of 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00299##
[0495] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl- 2-fluorobenzoic acid, the title compound was obtained (0.065 g, 22%) as a colorless solid: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.80-8.56 (m, 1H), 7.59-7.46 (m, 2H), 7.13-7.05 (m, 1H), 7.01-6.89 (m, 1H), 6.54-6.40 (.English Pound.(m, 1H), 4.38-4.26 (m, 1H), 4.06-3.95 (m, 1H), 3.46-3.33 (m, 4H), 3.09-3.00 (m, 1H), 2.59-2.45 (m, 1H), 2.32-2.11 (m, 2H), 2.09-1.86 (m, 2H), 1.70-1.41 (m, 2H), 1.22-1.16 (m, 3H), 0.96-0.85 (m, 2H), 0.67-0.59 (m, 2H); MS(ES+) m/z 513.1, 515.1 (M+1).
Example 53
Synthesis of (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((3-(3,5-dichlorophenoxy)-pip- eridin-1-yl)methyl)-2-fluorobenzamide.
##STR00300##
[0496] Step 1. Preparation of (S)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate
##STR00301##
[0498] A mixture of (S)-tert-butyl 3-hydroxypiperidine-1-carboxylate (1.00 g, 4.97 mmol), 3,5-dichloroiodobenzene (1.36 g, 4.97 mmol), copper(I) iodide (0.142 g, 0.75 mmol), 3,4,7,8-tetramethyl-1,10-phenanthroline (0.352 g, 1.49 mmol), molecular sieves 4 .ANG. (1.00 g), and cesium carbonate (4.86 g, 14.9 mmol) in toluene (6 mL) was degassed and then heated to 90.degree. C. for 16 hours. After cooling to ambient temperature, the mixture was filtered through a plug of celite. The filter cake was washed with dichloromethane (100 mL), and the combined filtrate concentrated in vacuo. Purification of the residue by column chromatography (0 to 30% ethyl acetate in hexanes) afforded the title compound as a colorless oil: (1.20 g, 70%): MS(ES+) m/z 346.1, 348.1 (M+1).
Step 2. Preparation of (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt
##STR00302##
[0500] A solution of (S)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate (1.17 g, 3.38 mmol) in dichloromethane (30 mL) was treated with trifluoroacetic acid (10 mL). The mixture was stirred at ambient temperature for 1 hour and then concentrated in vacuo to provide the title compound as an oil (1.22 g, quant.); MS(ES+) m/z 246.2, 248.1 (M+1)
Step 3. Preparation of (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1- yl)methyl)-2-fluorobenzoate
##STR00303##
[0502] A mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(((methylsulfonyl)oxy)methyl)benzoate (0.64 g, 1.86 mmol), (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt (0.74 g, 2.05 mmol), and potassium carbonate (0.64 g, 4.65 mmol) in anhydrous dimethylformamide (15 mL) was stirred at ambient temperature for 18 hours. The reaction mixture was diluted with ethyl acetate (100 mL), washed with water (80 mL), saturated ammonium chloride (2.times.80 mL), brine (80 mL), dried over anhydrous sodium sulfate, filtered the solid, and concentrated in vacuo. The residue was purified by flash chromatography (0 to 25% ethyl acetate in hexanes) to provide the title compound as an oil (0.70 g, 76%): .sup.1H NMR (300 MHz, CDCl.sub.3) d 7.48 (d, J=7.3 Hz, 1H), 7.15 (d, J=11.8 Hz, 1H), 6.90-6.87 (m, 1H), 6.76-6.73 (m, 2H), 4.35-4.23 (m, 1H), 3.72-3.58 (m, 2H), 2.97-2.88 (m, 1H), 2.71-2.61 (m, 1H), 2.31-2.12 (m, 2H), 2.06-1.88 (m, 2H), 1.85-1.74 (m, 1H), 1.70-1.38 (m, 11H), 0.94-0.82 (m, 2H), 0.64-0.54 (m, 2H); MS(ES+) m/z 494.3, 496.3 (M+1).
Step 4. Preparation of (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid
##STR00304##
[0504] A solution of (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin- 1-yl)methyl)-2-fluorobenzoate (1.04 g, 2.10 mmol) in dichloromethane (30 mL) was treated with trifluoroacetic acid (10 mL). The resulting solution was stirred at ambient temperature for 1 hour and then concentrated in vacuo to provide the title compound as a colorless solid (0.92 g, quant.); MS(ES+) m/z 438.1, 440.1 (M+1).
Step 5. Preparation of (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((3-(3,5-dichlorophenoxy)pipe- ridin-1-yl)methyl)-2-fluorobenzamide
##STR00305##
[0506] A mixture of (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid (0.30 g, 0.68 mmol), cyclopropanesulfonamide (0.12 g, 1.02 mmol), 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide (0.20 g, 1.02 mmol), and 4-dimethylaminopyridine (0.25 g, 2.04 mmol) in dichloromethane (12 mL) was stirred at ambient temperature for 24 hours. The reaction mixture was treated with acetic acid (1.0 mL) and purified by flash chromatography (0 to 30% ethyl acetate (containing 0.2% acetic acid) in hexanes) to provide the title compound as a colorless solid (0.175 g, 48%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.93 (br s, 1H), 7.24 (d, J=11.7 Hz, 1H), 7.18 (d, J=7.0 Hz, 1H), 7.09-7.07 (m, 1H), 7.01 (d, J=1.9 Hz, 2H), 4.61-4.51 (m, 1H), 3.69 (s, 2H), 3.08-2.99 (m, 1H), 2.85-2.77 (m, 1H), 2.63-2.52 (m, 1H), 2.41-2.21 (m, 2H), 2.05-1.97 (m, 1H), 1.96-1.84 (m, 1H), 1.78-1.67 (m, 1H), 1.63-1.51 (m, 1H), 1.49-1.34 (m, 1H), 1.11-1.03 (m, 4H), 0.91-0.79 (m, 2H), 0.68-0.56 (m, 2H); MS(ES+) m/z 541.2, 543.2 (M+1).
Example 54
Synthesis of (S)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)pi- peridin- 1-yl)methyl)-2-fluorobenzamide, acetic acid salt
##STR00306##
[0508] Following the procedure as described in Example 53, step 5, and making variation as required to replace cyclopropanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.16 g, 42%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.74 (br s, 1H), 7.72 (d, J=7.9 Hz, 1H), 7.35-7.26 (m, 1H), 6.91 (s, 1H), 6.77 (s, 2H), 4.39-4.28 (m, 1H), 4.24 (t, J=7.7 Hz, 4H), 3.37-3.65 (m, 2H), 2.96-2.86 (m, 1H), 2.74-2.62 (m, 1H), 2.39-2.19 (m, 4H), 2.08 (s, 3H), 1.98-1.79 (m, 3H), 1.72-1.44 (m, 3H), 1.00-0.88 (m, 2H), 0.71-0.59 (m, 2H); MS(ES+) m/z 556.2, 558.2 (M+1).
Example 55
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((3-(3,5-dichlorophenoxy)pipe- ridin- 1-yl)methyl)-2-fluorobenzamide
##STR00307##
[0509] Step 1. Preparation of (R)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate
##STR00308##
[0511] Following the procedure as described in Example 53 step 1 and making variation as required to replace (S)-tert-butyl 3-hydroxypiperidine-1-carboxylate with (R)-tert-butyl 3-hydroxypiperidine-1-carboxylate, the title compound was obtained as a colorless oil (0.461 g, 83%): .sup.1H NMR (300 MHz, CDCl.sub.3) d 6.91-6.87 (m, 1H), 6.76-6.74 (m, 2H), 4.23-4.12 (m, 1H), 3.92-3.03 (m, 4H), 2.01-1.88 (m, 1H), 1.85-1.63 (m, 2H), 1.55-1.27 (m, 10H).
Step 2. Preparation of (R)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt
##STR00309##
[0513] Following the procedure as described in Example 53 step 2, and making variation as required to replace (S)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate with (R)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate, the title compound was obtained as an oil (0.43 g, quant.); MS (ES+) m/z 246.2, 248.2 (M+1).
Step 3. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate
##STR00310##
[0515] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with (R)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt, the title compound was obtained as an oil (0.38 g, 72%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.3 Hz, 1H), 7.15 (d, J=11.7 Hz, 1H), 6.90-6.87 (m, 1H), 6.76-6.73 (m, 2H), 4.35-4.24 (m, 1H), 3.72-3.58 (m, 2H), 2.97-2.88 (m, 1H), 2.71-2.61 (m, 1H), 2.31-2.12 (m, 2H), 2.08-1.98 (m, 1H), 1.97-1.88 (m, 1H), 1.85-1.75 (m, 1H), 1.68-1.39 (m, 11H), 11), 0.93-0.84 (m, 2H), 0.64-0.56 (m, 2H); MS (ES+) m/z 494.3, 496.3 (M+1).
Step 4. Preparation of (R)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid
##STR00311##
[0517] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with (R)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate, the title compound was obtained as an colorless solid (0.34 g, quant.): MS(ES+) m/z 438.1, 440.1 (M+1).
Step 5. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((3-(3,5-dichlorophenoxy)- piperidin-1-yl)methyl)-1-fluorobenzamide
##STR00312##
[0519] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with (R)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid, the title compound was obtained as a colorless solid (0.12 g, 57%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.85-8.67 (m, 1H), 7.72 (d, J=7.7 Hz, 1H), 7.29 (d, J=14.0 Hz, 1H), 6.91 (s, 1H), 6.75 (s, 2H), 4.37-4.26 (m, 1H), 3.78-3.61 (m, 2H), 3.13-3.03 (m, 1H), 2.94-2.85 (m, 1H), 2.72-2.61 (m, 1H), 2.37-2.18 (m, 2H), 2.09-1.98 (m, 1H), 1.97-1.79 (m, 2H), 1.71-1.49 (m, 2H), 1.48-1.41 (m, 2H), 1.18-1.09 (m, 2H), 0.99-0.88 (m, 2H), 0.70-0.59 (m, 2H); MS(ES+) m/z 541.2, 543.2 (M+1).
Example 56
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)pi- peridin-1-yl)methyl)-2-fluorobenzamide
##STR00313##
[0521] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with (R)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid and cyclopropanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.30 g, 58%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.80-8.62 (m, 1H), 7.73 (d, J=7.3 Hz, 1H), 7.35-7.26 (m, 1H), 6.91 (s, 1H), 6.76 (s, 2H), 4.38-4.28 (m, 1 H), 4.23 (t, J=7.7 Hz, 4H), 3.77-3.62 (m, 2H), 2.95-2.86 (m, 1H), 2.72-2.62 (m, 1H), 2.37-2.18 (m, 3H), 2.06-1.99 (m, 1H), 1.97-1.88 (m, 1H), 1.86-1.79 (m, 1H), 1.72-1.45 (m, 3H), 1.01-0.89 (m, 2H), 0.71-0.59 (m, 2H); MS(ES+) m/z 556.2, 558.2 (M+1).
Example 57
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(3,5-dichlorophenoxy)piperidi- n-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid
##STR00314##
[0522] Step 1. Preparation of tert-butyl 4-(3,5-dichlorophenoxy)piperidine-1-carboxylate
##STR00315##
[0524] Following the procedure as described in Example 53 step 1, and making variations as required to replace (S)-tert-butyl 3-hydroxypiperidine-1-carboxylate with tert-butyl 4-hydroxypiperidine-1-carboxylate, the title compound was obtained as an oil (0.84 g, 41%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 6.92-6.89 (m, 1H), 6.77-6.74 (m, 2H), 4.44-4.34 (m, 1H), 3.69-3.58 (m, 2H), 3.37-3.25 (m, 2H), 1.93-1.80 (m, 2H), 1.75-1.63 (m, 2H), 1.43 (s, 9H).
Step 2. Preparation of 4-(3,5dichlorophenoxy)piperidine, trifluoroacetic acid salt
##STR00316##
[0526] Following the procedure as described in Example 53 step 2, and making variation as required to replace (S)-tert-butyl 3-(3,5-dichlorophenoxy)piperidine-1-carboxylate with tert-butyl 4-(3,5-dichlorophenoxy)piperidine-1-carboxylate, the title compound was obtained as an oil (0.88 g, quant.); MS (ES+) m/z 246.2, 248.2 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate
##STR00317##
[0528] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt, the title compound was obtained as an oil (0.74 g, 62%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.3 Hz, 1H), 7.17 (d, J=11.8 Hz, 1H), 6.92-6.88 (m, 1H), 6.79-6.74 (m, 2H), 4.35-4.25 (m, 1H), 3.65 (s, 2H), 2.75-164 (m, 2H), 2.42-2.30 (m, 2H), 1.77-1.90 (m, 3H), 1.86-1.73 (m, 2H), 1.55 (s, 9H), 0.96-0.87 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 494.3, 496.3 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid
##STR00318##
[0530] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin- 1-yl)methyl)-2-fluorobenzoate with tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)- piperidin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as an colorless solid (0.66 g, quant.); MS(ES+) m/z 438.2, 440.2 (M+1).
Step 5. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(3,5-dichlorophenoxy)- piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00319##
[0532] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid and purification by reverse phase HPLC, the title compound was obtained as a colorless solid (0.20 g, 49%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.25 (brs, 1H), 9.76 (brs, 1H), 7.52 (d, J=10.9 Hz, 1H), 7.24 (d, J=6.7 Hz, 1H), 7.20-7.06 (m, 3H), 4.90-4.77 (m, 1H), 4.56 (s, 2H), 3.55-3.41 (m, 1H), 3.34-3.18 (m, 3H), 3.11-3.01 (m, 1H), 2.30-1.92 (m, 4H), 1.86-1.72 (m, 1H), 1.15-1.06 (m, 4H), 1.03-0.95 (m, 2H), 0.80-0.72 (m, 2H); MS (ES+) m/z 541.2, 543.2 (M+1).
Example 58
Synthesis of N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)piperi- din-1-yl)- methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00320##
[0534] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid and cyclopropanesulfonamide with azetidine-1-sulfonamide and purification by reverse phase HPLC, the title compound was obtained as a colorless solid (0.19 g, 46%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.00 (brs, 1H), 9.69 (brs, 1H), 7.52 (d, J=10.8 Hz, 1H), 7.26 (d, J=6.5 Hz, 1H), 7.20-7.07 (m, 3H), 4.89-4.79 (m, 1H), 4.56 (s, 2H), 4.03 (t, J=7.6 Hz, 4H), 3.54-3.42 (m, 1H), 3.39-3.18 (m, 3H), 2.24-2.09 (m, 4H), 2.08-1.98 (m, 2H), 1.87-1.70 (m, 1H), 1.04-0.96 (m, 2H), 0.82-0.74 (m, 2H); MS(ES+) m/z 556.2, 558.2 (M+1).
Example 59
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-chloro-2-fluoro- N-(methylsulfonyl)benzamide
##STR00321##
[0536] To a solution of (1-benzhydrylazetidin-3-yl)methanol (40.4 mg, 0.159 mmol) and 5-chloro-2,4-difluoro-N-methylsulfonyl-benzamide (43.0 mg, 0.159 mmol) in DMSO (0.80 mL) at rt was added potassium tert-butoxide in 1:10 THF-DMSO (0.38 mL, 0.93 M). The mixture was stirred at rt for 1 hr. LCMS showed major product. Diluted with EtOAc, the contents were washed with 1:4 mixture of 1M HCl and 1M NaH.sub.2PO.sub.4 (2.times.) and brine (1.times.), dried (Na.sub.2SO.sub.4). After filtration and concentration, the crude was purified with HPLC (55.7 mg). LCMS (Method D): RT=5.37 min, m/z: 503.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.79 (d, J=7.5 Hz, 1H), 7.47-7.40 (m, 4H), 7.32 (t, J=7.5 Hz, 4H), 7.27-7.15 (m, 3H), 4.78 (s, 1H), 4.29 (d, J=6.1 Hz, 2H), 3.45 (s, 2H), 3.20 (m, 3H), 2.96 (s, 1H).
Example 60
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-((2-(trim- ethylsilyl)ethyl)sulfonyl)benzamide
##STR00322##
[0537] Step 1: Preparation of tert-butyl 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-fluoro-2-fluorobenzoate
[0538] To solution of (1-benzhydrylazetidin-3-yl)methanol (1.14 g) and tert-butyl 5-chloro-2,4-difluoro-benzoate (1.244 g, .about.90% pure) in DMSO (13.5 mL) at 14.degree. C. (bath) was added potassium tert-butoxide (0.606 g). The mixture was stirred at rt for 1 hr. Diluted with EtOAc, the contents were washed with dilute NaHCO.sub.3 (2.times.) and brine (1.times.), and dried (Na.sub.2SO.sub.4). After filtration and concentration, the crude was purified with silica gel flash chromatography (0-40% EtOAc/heptane) to give the product (1.359 g, 63%).
Step 2: Preparation of tert-butyl 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
[0539] A mixture of tert-butyl 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-chloro-2-fluorobenzoate (1.35 g), cyclopropylboronic acid (506 mg), potassium phosphate (1.52 g), and potassium fluoride (163 mg) in toluene (16.8 mL) and water (0.56 mL) was purge with nitrogen. Tricyclohexylphosphine tetrafluoroborate (213 mg) and palladium acetate (64 mg) were added. The mixture was heated at 90.degree. C. for 7 hours. The mixture was diluted with EtOAc and filtered. The filtrate was concentrated. The residue was purified with silica gel flash chromatography (0-20% EtOAc/heptane with 0.5% Et.sub.3N) to give the product (1.092 g, 80%.
Step 3: Preparation of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
[0540] A to of tert-butyl 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate (248 mg) and potassium hydroxide (228 mg) in DMSO (2.0 mL) was slimed at rt for 16 hr. The contents were acidified with 1M NaH.sub.2PO.sub.4. Solid was collected with filtration, washed with water, and dried under vacuum (171 rug, 78%).
Step 4: Preparation of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-((2-(trim- ethylsilyl)ethyl)sulfonyl)benzamide
[0541] A mixture of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid (26.2 mg), 2-(trimethylsilyl)ethanesulfonamide (44.0 mg), HBTU (30.9 mg), and DIPEA (0.053 mL) DCE (0.83 mL) was heated at 40.degree. C. for 16 hr. Acidified with 0.5 M NaH.sub.2PO.sub.4, the contents were extracted with DCM (2.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with HPLC (18.0 mg, 50%). LCMS (Method D): RT=5.47 min, m/z: 595.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.66 (s, 1H), 7.45-7.36 (m, 4H), 7.25 (t, J=7.5 Hz, 4H), 7.19-7.07 (m, 3H), 6.93 (d, J=12.7 Hz, 1H), 4.45 (s, 1H), 4.19 (d, J=6.2 Hz, 2H), 3.38-3.29 (m, 2H), 2.99 (s, 2H), 2.84 (s, 1H), 2.07-1.97 (m, 1H), 0.93-0.81 (m, 4H), 0.66-0.59 (m, 2H), -0.00 (s, 7H), -0.03 (s, 4H).
Example 61
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-((2-metho- xyethyl)sulfonyl)benzamide
##STR00323##
[0543] The compound was prepared in a similar manner to Example 60 from 4-((1-benzhydrylazetidin-3-yl)-methoxy)-5-cyclopropyl-2-fluorobenzoic acid and 2-methoxyethanesulfonamide. LCMS (Method D): RT=4.53 min, m/z: 553.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.46-7.37 (m, 4H), 7.33-7.23 (m, 4H), 7.21-7.12 (m, 3H), 6.84 (d, J=12.6 Hz, 1H), 4.48 (s, 1H), 4.18 (d, J=6.1 Hz, 2H), 3.64 (t, J=6.6 Hz, 3H), 3.51 (s, 2H), 3.26 (s, 2H), 3.21 (s, 3H), 3.14 (s, 1H), 3.02 (d, J=6.5 Hz, 2H), 2.92-2.79 (m, 1H), 2.10-1.98 (m, 1H), 1.25 (q, J=7.6, 6.4 Hz, 6H), 0.94-0.83 (m, 2H), 0.66-0.55 (m, 2H).
Example 62
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-N-((difluoromethyl)- sulfonyl)-2-fluorobenzamide
##STR00324##
[0545] The compound was prepared in a similar manner to Example 60 from 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and difluoromethanesulfonamide, LCMS (Method E): RT=3.95 min, m/z: 545.2 [M+H].sup.+.
Example 63
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N- ((3,3,3-trifluoropropyl)sulfonyl)benzamide
##STR00325##
[0547] The compound was prepared in a similar manner to Example 60 from 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and 3,3,3-trifluoropropane-1-sulfonamide. LCMS (Method E): RT=5.14 min, m/z: 591.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.44 (d, J=7.2 Hz, 4H), 7.40-7.30 (m, 4H), 7.26 (s, 2H), 7.21 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.7 Hz, 1H), 4.22 (d, J=6.2 Hz, 2H), 3.53 (s, 3H), 2.76-2.60 (m, 2H), 2.09-1.98 (m, 1H), 0.93-0.82 (m, 2H), 0.66-0.57 (m, 2H).
Example 64
Synthesis of 4-((1 -benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-N-((cyclopropylmethyl)- sulfonyl)-2-fluorobenzamide
##STR00326##
[0549] The compound was prepared in a similar manner to Example 60 from 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and cyclopropylmethanesulfonamide. LCMS (Method E): RT=4.89 min, m/z: 549.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.84 (s, 1H), 7.46-7.37 (m, 4H), 7.28 (t, J=7.6 Hz, 4H), 7.23-7.10 (m, 3H), 6.95 (d, J=12.8 Hz, 1H), 4.49 (s, 1H), 4.22 (d, J=6.2 Hz, 2H), 3.37 (d, J=7.1 Hz, 2H), 3.02 (s, 2H), 2.94-2.80 (m, 1H), 2.10-1.99 (m, 1H), 1.11-0.97 (m, 1H), 0.94-0.84 (m, 2H), 0.71-0.63 (m, 2H), 0.60-0.52 (m, 2H), 0.37 0.28 (m, 2H), 0.37-0.28 (m, 2).2H hidden under water
Example 65
Synthesis of 4-((1-benzhydrylazetidin-3-yl)methoxy)-N-(butylsulfonyl)-5-cyclopropyl- 2-fluorobenzamide
##STR00327##
[0551] The compound was prepared in a similar manner to Example 60 from 4-((1-benzhydrylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and butane-1-sulfonamide. LCMS (Method E): RT=5.08 min, m/z: 551.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.75 (s, 1H), 7.42 (dd, J=8.2, 1.4 Hz, 4H), 7.27 (dd, J=8.3, 6.9, Hz, 4H), 7.21-7.11 (m, 3H), 6.92 (d, J=12.7 Hz, 1H), 4.47 (s, 1H), 4.20 (d, J=6.2 Hz, 2H), 3.37 (t, J=7.9 Hz, 2H), 3.01 (t, J=6.6 Hz, 2H), 2.93-2.81(m, 1H), 2.09-1.99 (m, 1H), 1.71-1.59 (m, 2H), 1.45-1.34 (m, 2H), 0.94-0.83 (m, 5H), 0.69-0.59 (m, 2H). 2H hidden under water
Example 66
Synthesis of 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(met- hylsulfonyl)benzamide
##STR00328##
[0552] Step 1: Preparation of 4-((1-(tert-butoxycarbonyl)-3-fluoroazetidin-3-yl)methoxy)-5- chloro-2-fluorobenzoic acid
[0553] To a solution of tert-butyl 3-fluoro-3-(hydroxymethyl)azetidine-1-carboxylate (0.16 g, 0.784 mmol) and 5-chloro-2,4-difluoro-benzoic acid (151 mg, 0.784 mmol) in dimethyl sulfoxide (4.00 mL/mmol, 44.1 mmol, 99.8 mass %) at 14.degree. C. (bath) was added potassium tert-butoxide (194 mg, 1.73 mmol). The mixture was stirred at that temp for 5 min then at rt for 30 min. Diluted with EtOAc, the contents were washed with 1:4 mixture of 0.3M HCl and 0.3M NaH.sub.2PO.sub.4 (3.times.) and brine, and dried (Na.sub.2SO.sub.4). After filtration and concentration, the white solid crude (327 mg) was used as-is.
Step 2: Preparation of tert-butyl 3-((2-chloro-4-(ethoxycarbonyl)-5-fluorophenoxy)methyl)-3-fluoroazetidine- -1-carboxylate
[0554] To a suspension of product from step 1 (278 mg) and potassium carbonate (185 mg, 1,32 mmol) in N,N-dimethylformamide (2.65 mL) was added iodoethane (156 mg, 0.99 mmol). The mixture was heated at 50.degree. C. for 2 hr. LCMS showed completion. Diluted with EtOAc (50 mL), the contents were washed with diluted NaHCO.sub.3 (2.times.) and brine, and dried (Na.sub.2SO.sub.4). After filtration and concentration, the residue was purified with silica gel flash chromatography (0-40% EtOAc/heptane) to give the product (141 mg).
Step 3: Preparation of ethyl 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate
[0555] A mixture of product from step 2 (57 mg, 0.14 mmol) and trifluoroacetic acid (0.28 mL, 3.6 mmol) in dichloromethane (0.83 mL) was stirred at 0.degree. C. for 30 min then at rt for 2 h. The contents were concentrated and used as-is.
Step 4: Preparation of ethyl 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate
[0556] To the residue from step 3 was added dichloromethane (0.83 mL), followed by N,N-diisopropylethylamine (0.19 mL, 1.1 mmol) and benzyl bromide (26 mg, 0.15 mmol). The mixture The mixture was stirred at rt for 16 hr. Dilute aq Na.sub.2CO.sub.3 was added. The contents were extracted with DCM (2.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with silica gel flash chromatography (0-25% EtOAc/heptane) to give the product as viscous oil (43.8 mg).
Step 5: Preparation of 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
[0557] A mixture of product from previous step (41.7 mg, 0.104 mmol) and potassium hydroxide (11.7 mg, 0.208 mmol) in methanol (0.62 mL) and water (0.16 mL) was stirred at rt for 3 hr then heated at 50.degree. C. for 1.5 hr. Diluted with water and acidified with 1M NaH.sub.2PO.sub.4, the contents were extracted with DCM (3x). The combined extracts were dried (Na.sub.2SO.sub.4) and concentrated. The crude (37.6 mg) was used as-is.
Step 6: Preparation of 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro- N-(methylsulfonyl)benzamide
[0558] A Mixture of product from previous step (37.6 mg, 0.101 mmol), methanesulfonamide (28.7 mg, 0.302 mmol), N,N-diisopropylethylamine (0.089 mL, 0.503 mmol), and HBTU ((51.2 mg, 0.131 mmol) in 1,2-dichloroethane (1 mL) was stirred at 50.degree. C. for 2 hr then at 65.degree. C. for 1 hr. To the mixture was added 1M NaH.sub.2PO.sub.4. The contents were extracted with DCM (2.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with HPLC (23.0 mg, 50.7%). LCMS (Method D): RT=4.24 min, m/z: 451.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.39-7.22 (m, 5H), 7.17 (d, J=8.3 J 8.3 Hz, 1H), 7.01 (d, J=12.7 Hz, 1H), 4.43 (d, J=23.8 Hz, 2H), 3.74 (s, 2H), 3.59 (dd, J=13.3, 9.1 Hz, 2H), 3.25 (s, 3H), 2.06-1.95 (m, 1H), 0.92-0.82 (m, 2H), 0.71-0.62 (m, 2H).
Example 67
Synthesis of 4-((1-benzyl-3-fluoroazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro- N-(methylsulfonyl)benzamide
##STR00329##
[0560] The compound was prepared in a similar manner to Example 66 while in step 3 benzyl bromide being replaced by bromodiphenylmethane and DCM being replaced by DMF. LCMS (Method D): RT=4.67 min, m/z: 427.2 [M+H].sup.+. .sup.1H NMR (400 DMSO-d6) .delta. 11.90 (s, 1H), 7.49-7.40 (m, 4H), 7.29 (dd, J=8.4, 6.8 Hz, 4H), 7.23-7.13 (m, 3H), 7.02 (d, J=12.7 Hz, 1H), 4.60 (s, 1H), 4.48 (d, J=23.1 Hz, 2H), 3.49-3.38 (m, 2H), 3.27-3.15 (m, 5H), 2.06-1.91 (m, 1H), 0.90-0.79 (m, 2H), 0.70-0.60 (m, 2H).
Example 68
Synthesis of 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2- fluoro-N-(methylsulfonyl)benzamide
##STR00330##
[0561] Step 1: Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)- 3-methylazetidine-1-carboxylate
[0562] The compound was prepared from to tert-butyl 3-(hydroxymethyl)-3-methylazetidine-1-carboxylate and tert-butyl 5-chloro-2,4-difluorobenzoate in a similar manner to step 1 of Example 60.
Step 2: Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl- 5-fluorophenoxy)methyl)- 3-methylazetidine-1-carboxylate
[0563] The compound was prepared from tert-butyl 3-((4-(tert-butoxycarbonyl)-2 -chloro-5 -fluorophenoxy)methyl)-3-methylazetidine-1-carboxylate in a similar manner to step 2 of Example 60.
Step 3: Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3- yl)methoxy)benzoate
[0564] To a solution of tert-butyl 3-[(4-tert-butoxycarbonyl-2-cyclopropyl-5-fluorophenoxy)methyl]- 3-methyl-azetidine-1-carboxylate (73.3 mg, 0.168 mmol) in acetonitrile (1.35 mL) at 7.degree. C. (bath) was added p-toluenesulfonic acid hydrate (38.4 mg, 0.202 mmol). After 10 min, the mixture was stirred at rt for 20 hr. Tert-butyl acetate (0.23 mL) and p-toluenesulfonic acid hydrate (16.0 mg, 0.842 mmol) were added. After 1 hr, acetonitrile (3 mL) and K.sub.2CO.sub.3 was added (500 mg). After well mixing, the contents were diluted with EtOAc and filtered. The filtrated was concentrated. The residue was used as-is.
Step 4: Preparation of tert-butyl 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5- cyclopropyl-2-fluorobenzoate
[0565] A mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-[(3-methylazetidin-3-yl)methoxy]benzoate (56.3 mg, 0.168 mmol), bromodiphenylmethane (62.3 mg, 0.252 mmol), and cesium carbonate (164 mg, 0.504 mmol) in acetonitrile (1.68 mL) was heated at 50.degree. C. for 16 hr. The contents were concentrated. The residue was suspended in water and extracted with DCM (2.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with silica gel flash chromatography (0-30% EtOAc/heptane) to give the product (96.9 mg).
Step 5: Preparation of 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoic acid
[0566] A mixture of product from previous step (96.9 mg) and potassium hydroxide (86.7 mg, 1.55 mmol) in DMSO (1.93 mL) was stirred at rt for 40 hr. Acidified with 0.5M NaH.sub.2PO.sub.4, the contents were extracted with DCM (3.times.). The combined extracts were dried (Na.sub.2SO.sub.4) and concentrated. The crude was used as-is (103 mg).
Step 6: Preparation of 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoic acid
[0567] The compound was prepared in a similar manner to step 4 of Example 60 from 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-f- luorobenzoic acid and methanesulfonamide. LCMS (Method D): RT=4.49 min, m/z: 523.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.83 (s, 1H), 7.48-7.38 (m, 4H), 7.28 (t, J=7.6 Hz, 4H), 7.23-7.13 (m, 3H), 6.98 (d, J=12.8 Hz, 1H), 4.53 (s, 1H), 4.11 (s, 2H), 3.17 (s, 2H), 2.88 (d, J=11.8 Hz, 2H), 2.12-2.00 (m, 1H), 1.35 (s, 3H), 0.94-0.85 (m, 2H), 0.72-0.62 (m, 2H).
Example 69
Synthesis of 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-N- (ethylsulfonyl)-2-fluorobenzamide
##STR00331##
[0569] The compound was prepared in a similar manner to step 4 of Example 60 from 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-f- luorobenzoic acid and ethanesulfonamide. LCMS (Method D): RT=4.63 min, m/z: 537.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.75 (s, 1H), 7.47-7.37 (m, 4H), 7.28 (t, J=7.5 Hz, 4H), 7.18 (t, J=7.3 Hz, 3H), 6.99 (d, J=12.8 Hz, 1H), 4.50 (s, 1H), 4.10 (s, 2H), 3.50-3.38 (m, 2H), 3.15 (s, 2H), 2.95-2.81 (m, 2H), 2.12-1.99 (m, 1H), 1.35 (s, 3H), 1.24 (t, J =7.4 Hz, 3H), 0.94-0.83 (m, 2H), 0.71-0.60 (m, 2H).
Example 70
Synthesis of 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-N- (cyclopropylsulfonyl)-2-fluorobenzamide
##STR00332##
[0571] The compound was prepared in a similar manner to step 4 of Example 60 from 4-((1 -benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and cyclopropanesulfonamide. LCMS (Method E): RT=4.69 min, m/z: 549.2 [M+H].sup.+. .sup.1H NMR (400 MHZ, DMSO-d6) .delta. 11.79 (s, 1H), 7.46-7.38 (m, 4H), 7.32-7.23 (m, 4H), 7.22-7.13 (m, 3H), 6.94 (d, J=12.8 Hz, 1H), 4.50 (s, 1H), 4.09 (s, 2H), 3.14 (d, J=7.1 Hz, 2H), 3.07-2.96 (m, 1H), 2.92-2.80 (m, 2H), 2.13-2.00 (m, 1H), 1.30-1.20 (m, 3H), 1.03 (d, J=17.8 Hz, 4H), 0.94-0.83 (m, 2H), 0.69-0.60 (m, 2H).
Example 71
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)- - 5-cyclopropyl-2-fluorobenzamide
##STR00333##
[0573] The compound was prepared is a similar manner to step 4 of Example 60 from 4-((1-benzhydryl-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-f- luorobenzoic acid and azetidine-1-sulfonamide. LCMS (Method D): RT=4.74 min, m/z: 564.2 [M+H].sup.+.
Example 72
Synthesis of tert-butyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)-methyl)a- zetidine-1-carboxylate
##STR00334##
[0574] Steps 1-2: Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)-azetidi- ne-1-carboxylate
[0575] The compound was prepared from tert-butyl 3-(hydroxymethyl)azetidine-1-carboxylate and tert-butyl 5-chloro-2,4-difluorobenzoate in a similar manner to steps 1-2 of Example 60.
Step 3: Preparation of 4-((1-(tert-butoxycarbonyl)azetidin-3-yl)methoxy)-5- cyclopropyl-2-fluorobenzoic acid
[0576] To a mixture of tert-butyl 3-[(4-tert-butoxycarbonyl-2-cyclopropyl-5-fluorophenoxy)- methyl]azetidine-1-carboxylate (0.711 g, 1.69 mmol) and potassium hydroxide (1.14 g, 20.2 mmol) in methanol (8.43 mL) was slowly added water (0.84 mL). The resulting mixture was heated at 60.degree. C. for 16 hr. LCMS showed completion. The contents were diluted with water and acidified with 1:4 mixture of 1M HCl and 1M NaH.sub.2PO.sub.4, and extracted with DCM (2.times.). The combined DCM solutions were dried (Na.sub.2SO.sub.4). After filtration, and concentration, the crude was used as-is.
Step 4: Preparation of tert-butyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)-phenoxy)methyl)a- zetidine-1-carboxylate
[0577] The compound was prepared at a similar manner to step 4 of Example 60 from 4-((1-(tert-butoxycarbonyl)azetidin-3-yl)methoxy)-5-cyclopropyl-2- -fluorobenzoic acid and methanesulfonamide. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.88 (s, 1H), 7.17 (d, J=8.3 Hz, 1H), 6.97 (d, J=12.8 Hz, 1H), 4.29-4.15 (m, 2H), 3.96 (s, 2H), 3.85-3.71 (m, 2H), 3.31 (s, 3H), 3.06-2.93 (m, 1H), 2.04-1.89 (m, 1H), 1.36 (s, 9H), 0.93-0.80 (m, 2H), 0.74-0.59 (m, 2H).
Example 73
Synthesis of 5-cyclopropyl-2-fluoro-4-((1-(3-fluorobenzyl)azetidin-3-yl)methoxy)- N-(methylsulfonyl)benzamide
##STR00335##
[0579] A mixture of tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]-methyl]aze- tidine-1-carboxylate (39.2 mg, 0.0753 mmol, Example 77) and trifluoroacetic acid (0.15 mL, 1.9 mmol) in dichloromethane (0.45 mL) was stirred at 0.degree. C. for 10 min then at rt for 1 h. The contents were concentrated. To the residue was added 1, 2-dichloroethane (1.5 mL). The mixture was cooled at 0.degree. C. N, N-diisopropylethylamine (0.158 mL, 0.904 mmol) was added, followed by 3-fluorobenzaldehyde (28.0 mg, 0.226 mmol) and sodium triacetoxyborohydride (63.8 mg, 0.301 mmol). The mixture was stirred at rt for 20 hr. Diluted with 0.5M NaH.sub.2PO.sub.4, the contents were extracted with 1:5 mixture of IPA-DCM (3.times.x). The combined org solutions were dried (Na.sub.2SO.sub.4). The crude purified with HPLC (10.9 mg). LCMS (Method E): RT=3.29 min, m/z: 451.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.43 (q, J=7.4 Hz, 1H), 7.27-7.12 (m, 4H), 6.85 (d, J=12.7 Hz, 1H), 4.19 (d, J=6.1 Hz, 2H), 3.98 (s, 2H), 3.74 (s, 2H), 3.56 (s, 2H), 3.06 (s, 4H), 2.11-1.97 (m, 1H), 0.94-0.82 (m, 2H), 0.66-0.57 (m, 2H).
Example 74
Synthesis of -((1-benzylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N- (methylsulfonyl)benzamide
##STR00336##
[0581] The compound was prepared in a similar manner to Example 73 from tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]methyl]azet- idine-1-carboxylate and benzaldehyde. LCMS (Method D): RT=3.88 min, m/z: 433.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.49-7.31 (m, 5H), 7.23 (d, J=8.5 Hz, 1H), 6.80 (d, J=12.7 Hz, 1H), 4.18 (d, J=6.1 Hz, 2H), 4.08 (s, 2H), 3.92-3.78 (m, 2H), 3.68 (d, J=8.1 Hz, 2H), 3.18-3.05 (m, 1H), 2.99 (s, 3H), 2.11-1.94 (m, 1H), 0.94-0.81 (m, 2H), 0.65-0.53 (m, 2H).
Example 75
Synthesis of 4-((1-(3-chloro-5-fluorobenzyl)azetidin-3-yl)methoxy)-5-cyclopropyl-2-flu- oro- N-(methylsulfonyl)benzamide
##STR00337##
[0583] The compound was prepared in a similar manner to Example 73 from tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]methyl]azet- idine-1-carboxylate and 3-chloro-5-fluorobenzaldehyde. LCMS (Method D): RT=4115 min, m/z: 485.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.41-7.34 (m, 1H), 7.29 (s, 1H), 7.24-7.15 (m, 2H), 6.90 (d, J=12.8 Hz, 1H), 4.20 (d, J=6.2 Hz, 2H), 3.88 (s, 2H), 3.65 (t, J=8.1 Hz, 2H), 3.44 (t, J=7.4 Hz, 2H), 3.17 (s, 3H), 3.08-2.95 (m, 1H), 2.04 (tt, J=8.6, 5.3 Hz, 1H), 0.94-0.83 (m, 2H), 0.69-0.60 (m, 2H).
Example 76
Synthesis of 5-cyclopropyl-2 -fluoro-4-((1-(3-fluoro-4-(trifluoromethyl)piperidin)azetidin- 3-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR00338##
[0585] The compound was prepared in a similar manner to Example 73 from tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]methyl]azet- idine-1-carboxylate and 3-fluoro-4-(trifluoromethyl)benzaldehyde. LCMS (Method D): RT=4.33 min, m/z: 519.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.76 (t, J=7.8 Hz, 1H), 7.45 (d, J=11.9 Hz, 1H), 7.37 (d, J=8.1 Hz, 1H), 7.19 (d, J =8.4 Hz, 1H), 6.91 (d, J=12.8 Hz, 1H), 4.21 (d, J=6.2 Hz, 2H), 3.87 (s, 2H), 3.58 (t, J=7.9 Hz, 2H), 3.18 (s, 3H), 3.05-2.93 (m, 1H), 2.06-1.98 (m, 1H), 0.94-0.85 (m, 2H), 0.68-0.61 (m, 2H).
Example 77
Synthesis of 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-3-methylazetidin-3-yl)methoxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00339##
[0586] Steps 1-4: Preparation of tert-butyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)-phenoxy)methyl)-- 3-methylazetidine-1-carboxylate
[0587] The compound was prepared in a similar manner to Example 72 while in step 1 tert-butyl 3-(hydroxymethyl)azetidine-1-carboxylate was replaced by tert-butyl 3-(hydroxymethyl)-3-methylazetidine-1-carboxylate.
Step 5: Preparation of 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-3-methylazetidin-3-yl)methoxy)-2- -fluoro-N-(methylsulfonyl)benzamide
[0588] The compound was prepared in a similar manner to Example 73 from tert-butyl 3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)-3-methyla- zetidine-1-carboxylate and 3,5-dichlorobenzaldehyde. LCMS (Method D): RT=4.42 min, m/z: 515.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.54 (t, J=2.0 Hz, 1H), 7.39 (d, J=1.9 Hz, 2H), 7.22 (d, J=8.4 Hz, 1H), 6.91 (d, J=12.8 Hz, 1H), 4.08 (s, 2H), 3.84 (s, 2H), 3.49 (s, 2H), 3.17 (s, 3H), 2.09-1.98 (m, 1H), 1.37 (s, 3H), 0.94-0.83 (m, 2H), 0.70-0.59 (m, 2H).
Example 78
Synthesis of tert-butyl 4-((2-chloro-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)- methyl)piperidine-1-carboxylate
##STR00340##
[0590] The compound was prepared in a similar manner to Example 59 from tert-butyl 4-(hydroxymethyl)-piperidine-1-carboxylate and 5-difluoro-N- methylsulfonylbenzamide. LCMS (Method D): RT=6.77 min, m/z: 409.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.77 (d, J=7.7 Hz, 1H), 7.13 (d, J=12.3 Hz, 1h), 4.08-3.88 (m, 4H), 3.13 (s, 3H), 2.74 (d, J=9.9 Hz, 2H), 1.97 (q, J=7.2, 5.8 Hz, 1H), 1.81-1.68 (m, 2H), 1.40 (s, 9H), 1.19 (qd, J=12.4, 4.1 Hz, 2H).
Example 79
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-phenylpiperidin- 4-yl)methoxy)benzamide
##STR00341##
[0592] The compound was prepared in a similar manner to Example 60 from (1-phenylpiperidin-4-yl)methanol and 5-chloro-2,4-difluoro-N-methylsulfonyl-benzamide.
[0593] LCMS (Method E): RT=4.68 min, m/z: 447.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.82 (s, 1H), 7.26-7.11 (m, 3H), 7.00-6.89 (m, 3H), 6.74 (t, J=7.2 Hz, 1H), 4.00 (d, J=6.2 Hz, 3H), 3.79-3.67 (m, 2H), 3.29 (s, 3H), 2.78-2.56 (m, 2H), 2.07-1.92 (m, 2H), 1.87 (d, J=11.9 Hz, 2H), 1.55-1.41 (m, 2H), 0.94-0.83 (m, 2H), 0.72-0.61 (m, 2H).
Example 80
##STR00342##
[0594] 5-Cyclopropyl-4-((1-(3,4-dichlorophenyl)(phenyl)methyl)azetidin-3-y- l)methoxy-2-fluoro-N-(methylsulfonyl)benzamide
##STR00343##
[0595] Step 1
##STR00344##
[0596] (3,4-Dichlorophenyl)(phenyl)methanol
[0597] A mixture of (3,4-dichlorophenyl)(phenyl)methanone (2.0 g, 8.0 mmol) and sodium borohydride (456 mg, 12 mmol), in EtOH (10 mL) was stirred at 25.degree. C. for 2 h. After removal of the solvent, the residue was diluted with water (20 mL) and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the desired product (2.0 g, 100%) as yellow oil. LCMS(ESI) m/z: 251.1 [M-H].sup.-.
Step 2
##STR00345##
[0598] 1,2-Dichloro-4-(chloro(phenyl)methyl)benezene
[0599] A solution of (3,4-dichlorophenyl)(phenyl)methanol (2.0 g, 7.9 mmol) in thionyl chloride (10 mL) was stirred at 60.degree. C. for 3 h. After cooling to room temperature, the reaction mixture was concentrated and purified by silica gel column (eluting with petroleum ether/ethyl acetate=100/1) to give the desired product (1.6 g, 76%) as yellow oil.
Step 3
##STR00346##
[0600] tert-Butyl 5-cyclopropyl-4-((1-((3,4-dichlorophenyl)(phenyl)methyl)-azetidin-3-yl)me- thoxy)-2-fluorobenzoate
[0601] A mixture of methyl tert-butyl 4-(azetidin-3-ylmethoxy)-5-cyclopropyl-2- fluorobenzoate (100 mg, 0.31 mmol), 1,2-dichloro-4-(chloro(phenyl)methyl)benzene (126 mg, 0.47 mmo), sodium iodide (93 mg, 0.62 mmol) and potassium carbonate (128 mg, 0.93 mmol) in MeCN (10 mL) was stirred at 80.degree. C. for 16 h. After cooling to room temperature, the reaction mixture was diluted with EtOAc (100 mL) and brine (50 mL). The organic layer was separated and washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=4/1) to give the target compound (100 mg, 58%) as a pale yellow oil. LCMS(ESI) m/z: 556.0 [M+H].sup.+.
Step 4
##STR00347##
[0602] 5-Cyclopropyl-4-((1-(3,4-dichlorophenyl)(phenyl)methyl)azetidin-3-y- l)methoxy)-2-fluorobenzoic acid
[0603] Trifluoroacetic acid (1 mL) was added to a solution of tert-butyl 5-cyclopropyl-4-((1- ((3,4-dichlorophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-2-fluorobenzoa- te (100 mg, 0.18 mmol) in DCM (2 mL) and the reaction stirred at room temperature for 1 h. The mixture was then concentrated to give the desired product (80 mg, crude) as a pale yellow solid. LCMS(ESI): 500.0 [M-H].sup.-.
Step 5
##STR00348##
[0604] 5-Cyclopropyl-4-((1-((3,4-dichlorophenyl)(phenyl)methyl)azetidin-3-- yl)methoxy-2- fluoro-N-(methylsulfonyl)benzamide
[0605] 1. A mixture of 5-cyclopropyl-4-((1-((3,4-dichlorophenyl)(phenyl)methyl)- azetidin-3-yl)methoxy)-2-fluorobenzoic acid (80 mg, 0.16 mmol), methanesulfonamide (23 mg, 0.24 mmol), EDCI (61 mg, 0.32 mmol) and DMAP (39 mg, 0.32 mmol) in DCM (4 mL) was stirred at 25.degree. C. for 16 h. The reaction mixture was diluted with EtOAc (100 mL), washed with HCl (2.0 M, 20 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase combiflash (30-40% MeCN in 0.1% NH.sub.4HCO.sub.3) to give the target product (35 mg, 38%) as an off-white solid. LCMS(ESI) Method A: RT=6.24 min, m/z: 577.2. [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta.7.60 (d, J=2.0 Hz, 1H), 7.48-7.24 (m, 8H), 6.85 (d, J=13.0 Hz, 1H), 4.58 (s, 1H), 4.23 (d, J=6.0 Hz, 2H), 3.48-3.42 (m, 2H), 3.32 (s, 3H), 3.27-3.21 (m, 2H), 3.06-3.00 (m, 1H), 2.13-2.09 (m, 1H), 0.97-0.94 (m, 2H), 0.70-0.67 (m, 2H).
##STR00349##
Example 81
(R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin-3-yl)m- ethyl)azetidin-3-yl)methoxy)benzamide
##STR00350##
[0606] Step 1
##STR00351##
[0607] Phenyl(pyridin-3-yl)methanol
[0608] A mixture of phenyl(pyridin-3-yl)methanone (2.0 g, 11 mmol) and sodium borohydride (623 mg, 16 mmol) in EtOH (30 mL) was stirred at 25.degree. C. for 2 h. The mixture was then concentrated, diluted with water (20 mL) and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the desired product (2.0 g, 100%) as yellow oil. LCMS(ESI): m/z 184.3 [M+H].sup.+.
Step 2
##STR00352##
[0609] 3-(Chloro(phenyl)methyl)pyridine
[0610] A solution of phenyl(pyridin-3-yl)methanol (1.5 g, 8.1 mmol) in thionyl chloride (10 mL) was stirred at 80.degree. C. for 16 h. The mixture was then cooled to room temperature, concentrated and purified silica gel chromatography (eluting with DCM/MeOH from 100/1 to 20/1) to give the desired product (1.5 g, 94%) as a brown solid. LCMS(ESI): m/z 204.3 [M+H].sup.+.
Step 3
##STR00353##
[0611] tert-butyl-5-Cyclopropyl-2-fluoro-4-((1-(phenyl(pyridin-3-yl)methyl- )azetidin- 3-yl)methoxy)benzoate
[0612] The compound was synthesized as described in step 3, Example 80. LCMS(ESI) m/z: 489.1 [M+H].sup.+.
Step 4
##STR00354##
[0613] 5-Cyclopropyl-2-fluoro-4-((1-(phenyl(pyridin-3-yl)methyl)azetidin-3- -yl)methoxy)benzoic acid
[0614] The compound was synthesized as described in step 4, Example 80. LCMS(ESI) m/z: 433.1 [M+H].sup.+.
Step 5
##STR00355##
[0615] (R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin- -3-yl)methyl)azetidin-3-yl)methoxy)benzamide
[0616] The compound was synthesized as described in step 5, Example 80. The enantionmer was separated by chiral SFC from the racemate. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=4.83 min). LCMS(ESI) Method B: RT=4.56 min, m/z: 510.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.90 (s, 1H), 8.66 (s, 1H), 8.43 (s, 1H), 7.80 (d, J=8.0 1H), 7.45-7.44 (m, 2H), 7.32-7.29 (m, 3H), 7.22-7.16 (m, 2H), 6.92 (d, J=13.0 Hz, 1H), 4.55 (s, 1H), 4.21 (d, J=6.5 Hz, 2H), 3.29-3.24 (m, 2H), 3.22 (s, 3H), 3.04-3.00 (m, 2H), 2.90-2.84 (m, 1H), 2.08-2.02 (m, 1H), 0.91-0.87 (m, 2H), 0.66-0.63 (m, 2H).
##STR00356##
Example 82
(S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin-3-yl)m- ethyl)azetidin-3-yl)methoxy)benzamide
[0617] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=4.08 mm). LCMS(ESI) Method B: RT=4.56 min, m/z: 510.2 [M+H].sup.+.sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.90 (s, 1H), 8.64 (s, 1H), 8.41 (s, 1H), 7.79 (d, J=8.0 Hz, 1H), 7.45-7.44 (m, 2H), 7.32-7.29 (m, 3H), 7.22-7.16 (m, 2H), 6.92 (d, J=13.0 Hz, 1H), 4.55 (s, 1H), 4.21 (d, J=6.5 Hz, 2H), 3.29-3.24 (m, 2H), 3.20 (s, 3H), 3.04-3.00 (m, 2H), 2.90-2.84 (m, 1H), 2.07-2.02 (m, 1H), 0.91-0.87 (m, 2H), 0.66-0.63 (m, 2H).
##STR00357##
Example 83
4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)azetidin-3-yl)methoxy)-5-- cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00358##
[0618] Step 1
##STR00359##
[0619] 5-Bromo-3-chloro-2-isopropoxypyridine
[0620] A mixture of potassium tert-butoxide (10 g, 88.8 mmol) in isopropanol (15 mL) was stirred at 95.degree. C. for 3 h, 5-bromo-2,3-dichloropyridine (5 g, 22.2 mmol) was then added. The reaction mixture was refluxed overnight then partitioned between ethyl acetate and water. The organic layer was washed with water, brine, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica column chromatography (eluting with petroleum ether/ethyl acetate=100/1) to give 5-bromo-3-chloro-2-isopropoxypyridine (3.2 g, 58% yield) as colorless oil. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 8.25-8.20 (m, 2H), 5.27-5.23 (m, 1H), 1.32 (d, J=5.5 Hz, 6H).
Step 2
##STR00360##
[0621] 5-Chloro-6-isopropoxynicotinaldehyde
[0622] n-BuLi (2.5 M, 9.6 mL, 24 mmol) was added dropwise to a solution of 5-bromo-2,3-dichloropyridine (3.0 g, 12 mmol) in anhydrous THF (20 mL) at -78.degree. C. The resulting mixture was stirred at this temperature for 10 min then DMF (2.6 g, 36 mmol) was added at -50.degree. C. The mixture was warmed to room temperature and partitioned with EtOAc (100 mL) and 1N HCl (10 mL). The organic layer was washed with saturated brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=50/1) to give 5-chloro-6-isopropoxynicotinaldehyde (700 mg, 29% yield) as colorless oil. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 9.94 (s, 1H), 8.70 (d, J=1.5 Hz, 1H), 8.24 (d, J=2.0 Hz, 1H), 5.40-5.31 (m, 1H), 1.37 (d, J=6.0 Hz, 6H).
Step 3
##STR00361##
[0623] (5-Chloro-6-isopropoxypyridin-3-yl)methanol
[0624] The compound was synthesized as described in step 1, Example 80. LCMS(ESI) m/z: 200.1 [M-H].sup.-.
Step 4
##STR00362##
[0625] 3-Chloro-5-(chloromethyl)-2-isopropoxypyridine
[0626] The compound was synthesized as described in step 2, Example 80.
Step 5
##STR00363##
[0627] 2tert-butyl 4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)azetidin-3-yl)methoxy)-5- -cyclopropyl-2-fluorobenzoate
[0628] The compound was synthesized as described in step 3, Example 80. LCMS(ESI) m/z: 505.1 [M+H].sup.+.
Step 6
##STR00364##
[0629] 4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)azetidin-3-yl)meth- oxy)-5-cyclopropyl-2-fluorobenzoic acid
[0630] The compound was synthesized as described in step 4 Example 80. LCMS(ESI) m/z: 449.1 [M+H].sup.+.
##STR00365##
Step 7
4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)azetidin-3-yl)methoxy-5-c- yclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0631] The compound was synthesized as described in step 5, Example 80. LCMS(ESI) Method A: RT=5.06 min, m/z: 526.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.99 (d, J=2.0 Hz, 1H), 7.55 (d, J=2.0 Hz, 1H), 7.23 (d, J=9.0 Hz, 1H), 6.71 (d, J=13.0 Hz, 1H), 5.28-5.26 (m, 1H), 4.11 (d, J=6.0 Hz, 2H), 3.55 (s, 2H), 3.39-3.33 (m, 2H), 3.13-3.11 (m, 2H), 2.86-2.83 (m, 4H), 2.00 (m, 1H), 1.31 (d, J=6.0 Hz, 6H), 0.88-0.85 (m, 2H), 0.56-0.53 (m, 2H).
##STR00366##
Example 84
(S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-phenylethyl)azetidi- n-3 -yl)methoxy)benzamide
##STR00367##
[0632] Step 1
##STR00368##
[0633] tert-Butyl 5-chloro-4-((2,2-dimethyl-1,3-dioxan-5-yl)methoxy)-2-fluorobenzoate
[0634] Potassium tert-butoxide (7.8 g, 70 mmol) was added to a solution of (2,2-dimethyl-1,3-dioxan-5-yl)methanol (9.3 g, 63.7 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (16.6 g, 66.9 mmol) in DMSO (200 mL) at 14.degree. C. After stirring at room temperature for 1 h, the reaction mixture was diluted with water (500 mL) and extracted with EtOAc (200 mL.times.3). The combined organics was washed with brine, dried over anhydrous sodium sulfate and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethylacetate, 5/1) to afford the target compound (16.4 g, yield: 69%) as a white solid.
Step 2
##STR00369##
[0635] tert-Butyl 5-cyclopropyl-4-(2,2-dimethyl-1,3-dioxan-5-yl)methoxy)-2-fluorobenzoate
[0636] Palladium acetate (23 mg 0.1 mmol) was added to a mixture of tert-butyl 5-chloro-4-((2,2-dimethyl-1,3-dioxan-5-yl)methoxy)-2-fluorobenzoate (375 mg, 1 mmol), cyclopropylboronic acid (176 mg, 2 mmol), potassium phosphate (1.06 g, 5 mmol) and tricyclohexylphosphine tetrafluoroborate (74 mg, 0.2 mmol) in toluene (5 mL) and water (0.25 mL) under a nitrogen atmosphere. The reaction mixture was heated at 100.degree. C. for 16 hours then cooled to room temperature. The mixture was then diluted with water (200 mL) and extracted with ethyl acetate (100 mL.times.3). The combined organics were washed with brine, dried over anhydrous sodium sulfate and concentrated in vacuo. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=5/1) to afford tert-butyl 5-cyclopropyl-4-((2,2-dimethyl-1,3-dioxan-5-yl)methoxy)-2-fluorobenzoate (350 mg, yield: 92%) as a white solid.
Step 3
##STR00370##
[0637] tert-Butyl 5-cyclopropyl-2-fluoro-4-(3-hydroxy-2-(hydroxymethyl)-propoxy)benzoate
[0638] A solution of tert-butyl 5-cyclopropyl-4-((2,2-dimethyl-1,3-dioxan-5-yl)methoxy)- 2-fluorobenzoate (350 mg, 0.92 mmol) in a mixture of THF (10 mL) and HCl (1 M, 10 mL) was stirred at room temperature for 2 h. The reaction mixture was diluted with DCM (20 mL.times.2) and washed with saturated aqueous NaHCO.sub.3 (10 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (eluting with hexanes/ethyl acetate=2/1) to give tert-butyl 5-cyclopropyl-2-fluoro-4-(3-hydroxy-2-(hydroxymethyl)propoxy)benzoate (300 mg, yield: 96%) as a yellow solid. LCMS(ESI) m/z: 339.1 [M-H].sup.-.
Step 4
##STR00371##
[0639] (S)-tert-Butyl 5-cyclopropyl-2-fluoro-4-((1-(1-phenylethyl)azetidin-3- yl)methoxy)benzoate
[0640] Trifluoromethanesulfonic anhydride (200 mg, 0.71 mmol) was added dropwise to a 0.degree. C. mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(3-hydroxy-2-(hydroxymethyl)- propoxy)benzoate (60 mg, 0.18 mmol) and N,N-diisopropylethylamine (91 mg, 0.71 mmol) in acetonitrile (5 mL). The mixture was stirred at 0.degree. C. for 1 h, then (S)-1-phenylethanamine (21 mg, 0.18 mmol) added, and the solution stirred for a further 1 h at room temperature. The reaction was quenched with water (10 mL) and extracted with ethyl acetate (10 mL.times.3). The combined organic layers were washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The crude product was purified by silica column chromatography (eluting with petroleum ether/ethyl acetate=10/1) to give (S)-tert-butyl 5-cyclopropyl-2- fluoro-4-((1-(1-phenylethyl)azetidin-3-yl)methoxy)benzoate (32 mg, 43%) as an oil. LCMS(ESI): m/z: 426.8 [M+H].sup.+.
Step 5
##STR00372##
[0641] (S)-5-Cyclopropyl-2-fluoro-4-((1-(1-phenylethyl)azetidin-3-yl)metho- xy)benzoic acid
[0642] The compound was synthesized as described in step 4, Example 80. LCMS(ESI) m/f: 370.1 [M+H].sup.+.
Step 6
##STR00373##
[0643] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-phenylethyl)- azetidin-3-yl)methoxy)benzamide
[0644] The compound was synthesized as described in step 5, Example 80. LCMS(ESI) Method A: RT=4.31 min, m/z: 447.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.37-7.32 (m, 5H), 7.20 (d, J=8.5 Hz, 1H), 6.83 (d, J=12.5 Hz, 1H), 4.17 (d, J=6.5 Hz, 2H), 3.75-3.46 (m, 5H), 3.04 (s, 3H), 3.00-2.98 (m, 1H), 2.03-2.00 (m, 1H), 1.29 (d, J=5.0 Hz, 3H), 0.88-0.87 (m, 2H), 0.62-0.59 (m, 2H).
Example 85
##STR00374##
[0645] (R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-phenylethyl)- azetidin- 3 -yl)methoxy)benzamide
[0646] The compound was synthesized as described in Example 5. LCMS(ESI) Method A: RT=4.32 min, m/z: 447.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.39-7.33 (m, 5H), 7.20 (d, J=8.5 Hz, 1H), 6.82 (d, J=12.5 Hz, 1H), 4.17 (d, J=5.5 Hz, 2H), 3.93-3.49 (m, 5H), 3.04 (s, 3H), 3.02-2.98 (m, 1H), 2.04-2.00 (m, 1H), 1.29 (d, J=6.0 Hz, 3H), 0.88-0.85 (m, 2H), 0.62-0.59(m, 2H).
Example 86
##STR00375##
[0647] 5-Cyclopropyl-4-((1-(1-(3,5-difluorophenyl)ethyl)azetidin-3-yl)meth- oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00376##
[0649] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=4.79 min, m/z: 483.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.18-7.07 (m, 4H), 6.91 (d, J=13.0 Hz, 1H), 4.19 (d, J=6.0 Hz, 2H), 3.64-3.63 (m, 1H), 3.52-3.49 (m, 1H), 3.40-3.33 (m, 1H), 3.22-3.17 (m, 5H), 2.90-2.88 (m, 1H), 2.04-2.01 (m, 1H), 1.18 (d, J=6.0 Hz, 3H), 0.89-0.86 (m, 2H), 0.66-0.64 (m, 2H).
Example 87
##STR00377##
[0650] 4-((1-Benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methy- lsulfonyl)benzamide
##STR00378##
[0651] Step 1
##STR00379##
[0652] tert-Butyl 4-((1-benzylpiperidin-4-yl)methoxy)-5-chloro-2-fluorobenzoate
[0653] Potassium tert-butoxide (135 mg, 1.12 mmol) was added to a mixture of tert-butyl 5-chloro-2,4-difluorobenzoate (300 mg, 0.93 mmol) and (1-benzylpiperidin-4- yl)methanol (230 g, 1.12 mmol) in DMSO (5 mL) at 15.degree. C. After stirring at room temperature for 1 h, the mixture was diluted with EtOAc, washed with brine, dried over Na.sub.2SO.sub.4, filtered and concentrated. The resulting residue was purified by silica gel column chromatography (eluting with petroleum ether/ethyl acetate=10/1) to give the desired product (105 mg, 26% yield) as an oil. LCMS(ESI): m/z 434.0 [M+H].sup.+.
Step 2.
##STR00380##
[0654] tert-Butyl 4-((1-benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
[0655] Palladium acetate (8 mg, 0.037 mmol) was added to a solution of tert-butyl 4-((1-benzylpiperidin-4-yl)methoxy)-5-chloro-2-fluorobenzoate (160 mg, 0.37 mmol), cyclopropylboronic acid (47 mg, 0.55 mmol), potassium phosphate (157 mg, 0.74 mmol) and tricyclohexylphosphine tetrafluoroborate (27 mg, 0.074 mmol) in toluene (2mL) and water (0.1 mL) under a nitrogen atmosphere. The reaction mixture was heated at 100.degree. C. for 18 hours then cooled to room temperature. The mixture was diluted with water (10 mL) and extracted with ethyl acetate (10 mL.times.3). The combined organic layers were washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=10/1) to give the desired product (110 mg, 68% yield) as an oil. LCMS(ESI): m/z 440.0 [M+H].sup.+.
Step 3
##STR00381##
[0656] 4-((1-Benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
[0657] The synthetic procedure was same as the step 4 of Example 80. LCMS(ESI) m/z: 384.0 [M+H].sup.+.
Step 4
##STR00382##
[0658] 4-((1-benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N- (methylsulfonyl)benzamide
[0659] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=4.76 min, m/z: 461.1 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.41-7.35 (m, 5H), 7.19 (d, J=8.5 Hz, 1H), 6.78 (d, J=12.5 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.89 (s, 2H), 3.13-3.11 (m, 2H), 3.02 (s, 3H), 2.52-2.50 (m, 2H), 2.02-1.97 (m, 1H), 1.93-1.86 (m, 3H), 1.51-1.45 (m, 2H), 0.89-0.85 (m, 2H), 0.60-0.57 (m, 2H).
Example 88
##STR00383##
[0660] 4-((1-(3-Fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- o-N-(methylsulfonyl)benzamide
##STR00384##
[0661] Step 1
##STR00385##
[0662] tert-butyl 4-((4-(tert-Butoxycarbonyl)-2-chloro-5-fluorophenoxy)- methyl)piperidine-1-carboxylate
[0663] Potassium tert-butoxide (6.2 g, 55.6 mmol) was added to a solution of tert-butyl 4-(hydroxymethyl)piperidine-1-carboxylate (10.0 g, 6.3 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (12.6 g, 50.9 mmol) in DMSO (200 mL). After stirring at room temperature for 1 h, the reaction mixture was diluted with water (500 mL) and extracted with EtOAc (200 mL.times.3). The combined organic layers were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (eluting with petroleum ether/ethyl acetate, from 20/1 to 5/1) to afford the target compound (12.3 g, yield: 60%) as a pale yellow liquid. LCMS(ESI) m/z: 331.9. [M-111].sup.+.
##STR00386##
Step 2
tert-Butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)- methyl)piperidine-1-carboxylate
[0664] Palladium acetate (672 mg, 3 mmol) was added to a solution of tert-butyl 4-((4-(tert-butoxycarbonyl)cyclohexyl)methoxy)-5-chloro-2-fluorobenzoate (13.3 g, 30 mmol), cyclopropylboronic acid (5.16 g, 60 mmol), potassium phosphate (25.5 g, 120 mmol) and tricyclohexylphosphine tetrafluoroborate (2.2 g, 6 mmol) in toluene (200 mL) and water (10 mL) under a nitrogen atmosphere. The reaction mixture was heated at 100.degree. C. for 16 hours then cooled to room temperature. The mixture was diluted with water (200 mL) was and extracted with ethyl acetate (100 mL.times.3). The combined organic layers were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (eluting with petroleum ether/ethyl acetate, from 10/1 to 2/) to afford the target compound (10.8 g, yield 80%) as a pale yellow liquid. LCMS(ESI) m/z: 338.0 [M-111].sup.+.
Step 3
##STR00387##
[0665] 5-Cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoic acid
[0666] A solution of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)methyl)piperidine-1-carboxylate (11.0 g, 24.5 mmol) in DCM (20 mL) and TFA (20 mL) was stirred at room temperature for 1 h. The reaction was quenched with saturated aqueous sodium bicarbonate and extracted with DCM (50 mL.times.3). The combined organic layers were dried over anhydrous sodium sulfate, filtered and concentrated to give target compound (6.5 g, yield: 90%) as a white solid which was used in the next step without further purification. LCMS(ESI) m/z: 294.1 [M+H].sup.+.
Step 4
##STR00388##
[0667] Methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate
[0668] Thionyl chloride (8 mL) was added dropwise to a solution of 5-cyclopropyl-2-fluoro-4- (piperidin-4-ylmethoxy)benzoic acid (5.0 g, 17 mmol) in MeOH (80 ml). After stirring at 70.degree. C. for 1 h, the solution was concentrated to give a brown solid, which was recrystallized (petroleum ether/ethyl acetate=5/1) to give the target compound as a gray solid (yield: 80%). LCMS(ESI) m/z: 308.1 [M+H].sup.+.
Step 5
##STR00389##
[0669] Methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5- cyclopropyl-2-fluorobenzoate
[0670] A mixture of methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (100 mg, 0.33 mmol), 1-(chloromethyl)-3-fluorobenzene (48 mg, 0.33 mmo), sodium iodide (149 mg, 0.99 mmol) and potassium carbonate (137 mg, 0.99 mol) in MeCN (10 mL) was stirred at 80.degree. C. for 1 h. The reaction mixture was diluted with EtOAc (100 mL) and brine (50 mL). The organic layer was separated, washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/EtOAc=5/1) to give the target compound (110 mg, 81%) as a pale yellow oil. LCMS(ESI) m/z: 416.0 [M+H].sup.+.
Step 6
##STR00390##
[0671] 4-((1-(3-Fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- obenzoic acid
[0672] A mixture of methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate (110 mg, 0.27 mmol) and lithium hydroxide (64 mg, 2.7 mmol) in THF (5 mL) and water 15 mL) was stirred at 50.degree. C. for 3 h. After cooling to room temperature, the mixture was adjusted to a pH of 2-3 with HCl (2M) then extracted with EtOAc (10.times.2 mL). The combined organic layers were washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the product (68 mg, 64%) as a pale yellow solid. LCMS(ESI) m/z: 402.1 [M+H].sup.+.
Step 7
##STR00391##
[0673] 4-((1-(3-Fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- o-N- (methylsulfonyl)benzamide
[0674] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=5.02 min, m/z: 479.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.43-7.42 (m, 1H), 7.41-7.13 (m, 4H), 6.81 (d, J=12.5 Hz, 1H), 3.92 (d, J=6.0 Hz, 2H), 3.74 (s, 2H), 3.18 (s, 3H), 3.07-2.99 (m, 2H), 2.30-2.28 (m, 2H), 2.04-1.98 (m, 1H), 1.87-1.82 (m, 3H), 1.47-1.24 (m, 2H), 0.89-0.85 (m, 2H), 0.62-0.59 (m, 2H).
Example 89
##STR00392##
[0675] 5-Cyclopropyl-4-((1-(1-(3,5-difluorophenyl)ethyl)piperidin-4-yl)met- hoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0676] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.39 min, m/z: 511.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.17-7.10 (m, 4H), 6.85 (d, J=13.0 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H). 3.81-3.74 (m, 1H), 3.16 (s, 3H), 3.11-3.08(m, 1H), 2.92-2.90 (m, 1H), 2.20-2.10 (m, 2H), 2.03-1.97 (m, 1H), 1.85-1.78 (m, 3H), 1.45-1.36 (m, 5H), 0.89-0.85 (m, 2H), 0.64-0.61 (m, 2H).
Example 90
##STR00393##
[0677] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidi- n-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00394##
[0678] Step 1
##STR00395##
[0679] 2-(3,5-Dichlorophenyl)oxirane
[0680] Sodium hydride (280 mg, 6.9 mmol) was added to an ice-cooled solution of trimethylsulfonium iodide (1.4 g, 6.9 mmol) in DMSO (40 mL). After stirring at room temperature for 30 min, 3,5-dichlorobenzaldehyde (1 g, 5.7 mmol) was added and mixture stirred further at room temperature for 1 h. The mixture was then quenched with water (40 mL) and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (40 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The crude product was used in the next step without further purification.
Step 2
##STR00396##
[0681] 1-(3,5-Dichlorophenyl)-2-methoxyethanol
[0682] 2-(3,5-Dichlorophenyl)oxirane (1 g, crude) was added to a solution of sodium (1.2 g, 53 mmol) in methanol (50 mL) and the mixture heated at 60.degree. C. for 1 h. After cooling to room temperature, the mixture was diluted with water (50 mL) and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (40 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/EtOAc=50/1) to give the target compound (160 mg, 14%) as a pale yellow oil. LCMS(ESI) m/z: 219.0 [M+H].sup.+.
Step 3
##STR00397##
[0683] 1,3-dichloro-5-(1-chloro-2-methoxyethyl)benzene
[0684] The compound was synthesized as described in step 2 of Example 80.
Step 4
##STR00398##
[0685] Methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin- 4-yl)methoxy)-2-fluorobenzoate
[0686] The compound was synthesized as described in step 5 of Example 88. LCMS(ESI) m/z: 510.1 [M+H].sup.+.
Step 5
##STR00399##
[0687] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidi- n-4-yl)methoxy)-2-fluorobenzoic acid
[0688] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 496.1 [M+H].sup.+.
Step 6
##STR00400##
[0689] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidi- n-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0690] The compound synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=5.78 min, m/z: 572.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.73 (brs, 1H), 7.50-7.49 (m, 1H), 7.39-7.38 (m, 2H), 7.16 (d, J=8.0 Hz, 1H), 6.82 (d, J=13.0 Hz, 1H), 3.88 (d, J=5.5 Hz, 2H), 3.73-3.65 (m, 3H), 3.22 (s, 3H), 3.14 (s, 3H), 2.99-2.97 (m, 1H), 2.81-2.79 (m, 1H), 2.11-2.07 (m, 1H), 2.02-1.95 (m, 2H), 1.79-1.71 (m, 3H), 1.38-1.28 (m, 2H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 91
##STR00401##
[0691] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)azetidin- -3-yl)methoxy)- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00402##
[0692] Step 1
##STR00403##
[0693] tert-Butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-3- azetidin-3-yl)methoxy)-2-fluorobenzoate
[0694] The compound was synthesized as described in step 3 of Example 80. LCMS(ESI) m/z: 524.0 [M+H].sup.+.
Step 2
##STR00404##
[0695] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)azetidin- -3-yl)methoxy)- 2-fluorobenzoic acid
[0696] The compound was synthesized as described in step 4 of Example 80. LCMS(ESI) m/z: 467.9 [M+H].sup.+.
Step 3
##STR00405##
[0697] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)azetidin- -3- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0698] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=5.26 min, m/z: 544.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.50 (m, 1H), 7.35 (m, 2H), 7.17 (d, J=8.0 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 4.16 (d, J=6.0 Hz, 2H), 3.60 (m, 1H), 3.48-3.45 (m, 1H), 3.42-3.32 (m, 3H), 3.20 (s, 3H), 3.18 (s, 3H), 3.15-3.12 (m, 1H), 3.09-3.07 (m, 1H), 2.87-2.85 (m, 1H), 2.04-2.00 (m, 1H), 0.89-0.87 (m, 2H), 0.64-0.63 (m, 2H).
Example 92
##STR00406##
[0699] 4-((1-(3-Chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- o- N-(methylsulfonyl)benzamide
[0700] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.35 min, m/z 494.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.42-7.31 (m, 4H), 7.19-7.17 (d, J=8.5 Hz, 1H), 6.81-6.79 (d, J=13.0 Hz, 1H), 3.92-3.91 (m, 2H), 3.68 (br s, 2H), 3.04-2.96 (m, 5H), 2.24-2.22 (m, 2H), 2.02-1.99 (m, 1H), 1.84-1.81 (d, J=12.5 Hz, 3H), 1.43-1.40 (m, 2H), 0.89-0.86 (m, 2H), 0.61-0.58 (m, 2H).
Example 93
##STR00407##
[0701] 5-Cyclopropyl-2-fluoro-4-((1-(4-methylbenzyl)piperidin-4-yl)methoxy- )-N- (methylsulfonyl)benzamide
[0702] The compound was synthesized as described n Example 88. LCMS(ESI) Method A: RT=4.88 min, m/z 475.0 [M+H].sup.+. .sup.1H NMR, (500 MHz, DMSO-.sub.6): .delta. 7.31-7.29 (d, J=7.5 Hz, 2H), 7.23-7.18 (m, 3H), 6.79-6.77 (d, J=12.5 Hz, 1H), 3.92 (m, 4H), 3.16-3.14 (m, 2H), 3.00 (s, 3H), 2.50 (s, 2H), 2.31 (s, 3H), 2.01-1.87 (m, 4H), 1.50-1.48 (m, 2H), 0.88-0.86 (m, 2H), 0.59-0.58 (m, 2H).
Example 94
##STR00408##
[0703] 5-Cyclopropyl-2-fluoro-4-((1-(3-fluoro-4-methoxybenzyl)piperidin-4- yl)methoxy)-N-(methylsulfonyl)benzamide
[0704] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.70 min, m/z 509.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.29-7.27 (d, J=12.5 Hz, 1H), 7.19-7.18 (d, J=6.0 Hz, 3H), 6.81-6.78 (d, J=13.0 Hz, 1H), 3.92 (m, 2H), 3.88 (m, 5H), 3.14-3.12 (m, 2H), 3.04 (s, 3H), 2.50 (s, 2H), 2.00-1.86 (m, 4H), 1.50-1.48 (m, 2H), 0.88-0.86 (m, 2H), 0.60-0.59 (m, 2H).
Example 95
##STR00409##
[0705] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(methylsulf- onyl)-phenyl)(phenyl)methyl)azetidin-3-yl)methoxy)benzamide
[0706] The compound was synthesized as described in Example 81. LCMS(ESI) Method A: RT=4.99 min, m/z 587.3 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-.sub.4): .delta. 7.90 (d, J=8.0 Hz, 2H), 7.73 (d, J=8.5 Hz, 2H), 7.45 (d, J =7.0 Hz, 2H), 7.37-7.25 (m, 3H), 7.23 (d, J=7.0 Hz, 1H), 6.76 (d, J=12.5 Hz, 1H), 4.66 (s, 1H), 4.20 (d, J=6.0 Hz, 2H), 3.47-3.33 (m, 2H), 3.24-3.16 (m, 5H), 3.09 (s, 3H), 3.03-3.00 (m, 1H), 2.10 (t, J=10.5 Hz, 1H), 0.94-0.90 (m, 2H), 0.70-0.67 (m, 2H).
Example 96
##STR00410##
[0707] (R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(methylsulf- onyl)-phenyl)(phenyl)methyl)azetidin-3-yl)methoxy)benzamide
[0708] The compound was synthesized as described in Example 81. LCMS(ESI) Method A: RT=4.99 min, m/z 587.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.91 (d, J=8.5 Hz, 2H), 7.74 (d, J=8.0 Hz, 2H), 7.45 (d, J =7.0 Hz, 2H), 7.34-7.31 (m, 3H), 7.26 (d, J=7.0 Hz, 1H), 6.85 (d, J=13.0 Hz, 1H), 4.71 (s, 1H), 4.25 (d, J=6.0 Hz, 2H), 3.50-3.43 (m, 2H), 3.34-3.21 (m, 5H), 3.09 (s, 3H), 3.06-3.04 (m, 1H), 2.10 (t, J=10.5 Hz, 1H), 0.97-0.93 (m, 2H), 0.70-0.67 (m, 2H).
Example 97
##STR00411##
[0709] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl)met- hoxy)- 2-fluoro-N-(methylsulfonyl)benzamide
[0710] The compound was synthesized as described in Example 88. LCMS(ESI): Method A: RT=6.17 min, m/z 543.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.51 (s, 3H), 7.35 (d, J=8.5 Hz, 1H), 6.63 (d, J=12.5 Hz, 1H), 4.14 (s, 1H), 3.82 (s, 2H), 3.54 (d, J=10.0 Hz, 1H), 3.21 (d, J=12.5 Hz, 4H), 2.66-2.62 (m, 2H), 2.09-2.00 (m, 4H), 1.69-1.59 (m, 5H), 0.88 (m, 2H), 0.63 (m, 2H).
Example 98
##STR00412##
[0711] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3-yl)meth- oxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0712] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=5.51 min, m/z 515.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.43 (s, 1H), 7.38-7.35 (m, 3H), 6.77 (d, J=13.0 Hz, 1H), 4.20-4.17 (m, 2H), 3.79 (s, 2H), 3.58 (d, J=7.0 Hz, 1H), 3.49 (d, J=4.0 Hz, 2H), 3.25 (s, 3H), 3.11 (t, J=13.0 Hz, 1H), 2.09-2.06 (m, 1H), 1.36 (d, J=6.5 Hz, 3H), 0.94 (m, 2H), 0.68 (m, 2H).
Example 99
##STR00413##
[0713] (R)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin)-4-y- l)methoxy)-2-fluoro-N-(methylsulfonyl)-benzamide
[0714] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate. Chiral HPLC (column: AD-3, 4.6.times.150 mm, 3 .mu.m; mobile Phase: n-hexane (0.1% DEA)/ EtOH 0.1% DEA)=90/10; flow: 1 mL/min; column temperature: 40.degree. C.; RT=6.59 min). LCMS(ESI) Method A: RT=6.13 min. m/z 543.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.51 (s, 1H), 7.40 (d, J=1.5 Hz, 2H), 7.17 (d, J=3 8.5 Hz, 1H), 6.82 (d, J=12.5 Hz, 1H), 3.91 (d, J=5.5 Hz, 2H), 3.70 (t, J=4.0 Hz, 1H), 3.32 (s, 3H), 3.03 (d, J=8.5 Hz, 1H), 2.86 (t, J=12.0 Hz, 1H), 2.05-1.99 (m, 3H), 1.83-1.76 (m, 3H), 1.40-1.33 (m, 5H), 0.89-0.86 (m, 2H), 0.63-0.59 (m, 2).
Example 100
##STR00414##
[0715] (S)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl- )methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0716] The compound was synthesized as described in Example 99. Chiral HPLC (column: AD-3, 4.6.times.150 mm, 3 .mu.m; mobile Phase: n -hexane (0.1% DEA)/EtOH 0.1% DEA)=90/10; flow: 1 mL/min; column temperature: 40.degree. C.; RT=9.38 min). LCMS(ESI) Method A: RT=6.06 min, m/z 542.9 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.50 (s, 1H), 7.39 (s, 2H), 7.19 (d, J=8.5 Hz, 1H), 6.78 (d, J=12.5 Hz, 1H), 3.89 (d, J=5.5 Hz, 2H), 3.65 (s, 1H), 3.03-2.99 (m, 4H), 2.84 (d, J=11.0 Hz, 1H), 2.04-1.98 (m, 3H), 1.82-1.75 (m, 3H), 1.38-1.31 (m, 5H), 0.88-0.85 (m, 2H), 0.61-0.57 (m, 2H).
Example 101
##STR00415##
[0717] 5-Cycloproyl-4-((1-(1-(3 ,5-dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluor- obenzamide
[0718] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.22 min, m/z 557.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.52 (s, 1H), 7.42 (s, 2H), 7.14 (d, J=9.0 Hz, 1H), 6.89 (d, J=12.5 Hz, 1H), 3.92 (d, J=6.0 Hz, 2H), 3.75 (s, 1H), 3.38 (d, J=7.0 Hz, 2H), 3.06 (d, J=10.0 Hz, 1H), 2.89 (d, J=9.5 Hz, 1H), 2.12-1.99 (m, 3H), 1.84-1.77 (m, 3H), 1.40-1.35 (m, 5H), 1.21 (t, J=14.5 Hz, 3H), 0.89-0.85 (m, 2H), 0.65-0.62 (m, 2H).
Example 102
##STR00416##
[0719] 5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)- -ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0720] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.26 min, m/z 569.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.52 (s, 1H), 7.41 (s, 2H), 7.14 (d, J=8.0 Hz, 1H), 6.90 (d, J=13.0 Hz, 1H), 3.93 (d, J=5.5 Hz, 2H), 3.73 (s, 1H), 3.06-3.02 (m, 2H), 2.89 (d, J=8.5 Hz, 1H), 2.11-1.99 (m, 3H), 1.84-1.77 (m, 3H), 1.41-1.34 (m, 5H), 1.06-1.01 (m, 4H), 0.90-0.86 (m, 2H), 0.65-0.62 (m, 2H).
Example 103
##STR00417##
[0721] N-(Azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichloropheny- l)-ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0722] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.34 min, m/z 583.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.48 (s, 1H), 7.38 (d, J=1.5 Hz, 2H), 7.19 (d, J=7.5 Hz, 1H), 6.81 (d, J=12.5 Hz, 1H), 3.88 (d, J=22.5 Hz, 6H), 3.61 (d, J=6.5 Hz, 1H), 2.97 (d, J=10.5 Hz, 1H), 2.81 (d, J=10.0 Hz, 1H), 2.08-1.96 (m, 5H), 1.82-1.74 (m, 3H), 1.37-1.30 (m, 5H), 0.88 (m, 2H), 0.61 (m, 2H).
Example 104
##STR00418##
[0723] 4-((1-(4-Chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- o- N-(methylsulfonyl)benzamide
[0724] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.29 min, m/z 494.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.41 (d, J=8.0 Hz, 2H), 7.36 (d, J=8.5 Hz, 2H), 7.20 (t, J =8.5 Hz, 1H), 6.74 (d, J=13.0 Hz, 1H), 3.90 (d, J=5.5 Hz, 2H), 3.61 (s, 2H), 2.93 (d, J=9.0 Hz, 5H), 2.16 (s, 2H), 1.99 (d, J=5.5 Hz, 1H), 1.80 (d, J=11.0 Hz, 3H), 1.40 (d, J=11.0 Hz, 2H), 0.87-0.86 (m, 2H), 0.56 (m, 2H).
Example 105
##STR00419##
[0725] 4-((1-(2-Chloro-4-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropy- l-2-fluoro-N-(methylsulfonyl)benzamide
[0726] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.69 min, m/z 513.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.57 (t, J=15.0 Hz, 1H), 7.45 (t, J=9.0 Hz, 1H), 7.27-7.24 (m, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.89 (d, J=12.5 Hz, 1H), 3.95 (d, J=5.5 Hz, 2H), 3.70 (s, 2H), 3.21 (s, 3H), 2.97 (d, J=10.5 Hz, 2H), 2.28 (t, J=22 Hz, 2H), 2.02 (t, J=10.0 Hz, 1H), 1.87-1.81 (m, 3H), 1.41 (d, J =11.5 Hz, 2H), 0.89 (m, 2H), 0.64 (m, 2H).
Example 106
##STR00420##
[0727] 5-Cyclopropyl-4-((1-(2,4-dichlorobenzyl)piperidin-4-yl)methoxy)-2- fluoro-N-(methylsulfonyl)benzamide
[0728] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.03 min, m/z 528.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.61 (s, 1H), 7.54 (d, J=8.0 Hz, 1H), 7.45 (t, J=9.0 Hz, 1H), 7.15 (d, J=8.5 Hz, 1H), 6.90 (d, J=13.0 Hz, 1H), 3.95 0), (d, J=6.0 Hz, 2H), 3.67 (s, 2H), 3.23 (s, 3H), 2.94 (d, J=10.5 Hz, 2H), 2.24 (t, J=22 Hz, 2H), 2.03-1.99 (m, 1H), 1.86-1.80 (m, 3H), 1.13-1.37 (m, 2H), 0.89 (m, 2H), 0.65 (m, 2H).
Example 107
##STR00421##
[0729] 4-((1-(2,5-bis(trifluoromethyl)piperidin)-4-methylpiperidin-4-yl)me- thoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0730] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 2,5-bis(trifluoromethyl)benzaldehyde. LCMS (Method F): RT=4.76 min, m/z: 577.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.76 (s, 1H), 8.15 (d, J=1.7 Hz, 1H), 7.96 (d, J=8.2 Hz, 1H), 7.85 (dd, J=8.3, 1.5 1H), 7.18 (d, J=8.3 Hz, 1H), 6.96 (d, J=12.9 Hz, 1H), 3.86 (s, 2H), 3.77 (s, 2H), 2.64-2.53 (m, 2H), 2.47-2.36 (m, 2H), 2.08-1.98 (m, 1H), 1.81-1.68 (m, 2H), 1.52-1.39 (m, 2), 1.09 (s, 3H), 0.94-0.83 (m, 2H), 0.71-0.62 (m, 2H).
Example 108
##STR00422##
[0731] (S)-4-((1-((3-Cyanophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-c- yclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0732] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75.25; flow: 2.25 mL/min; column temperature: 36.degree. C.; RT=6.85 min). LCMS(ESI) Method A: RT=5.66, m/z: 534.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.82 (s, 1H), 7.77 (d, J=8.0 Hz, 1H), 7.60 (d, J=7.5 Hz, 1H), 7.52-7.49 (m, 1H), 7.45-7.43 (m, 2H), 7.35-7.32 (m, 3H), 7.27-7.24 (m, 1H), 6.85 (d, J=13.0 Hz, 1H), 4.63 (s, 1H), 4.24 (d, J=6.5 Hz, 2H), 3.47-3.43 (m, 2H), 3.33 (s, 3H), 3.23-3.20 (m, 2H), 3.04-3.02 (m, 1H), 2.13-2.10 (m, 1H), 0.97-0.94 (m, 2H), 0.70-0.67 (m, 2H).
Example 109
##STR00423##
[0733] (R)-4-((1-((3-Cyanophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-c- yclopropyl-2-fluoro-N-(methylsulfonyl)benazmide
[0734] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ_H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 2.25 mL/min; column temperature: 36.degree. C.; RT=8.24 min). LCMS(ESI) Method A: RT=5.34 min, m/z: 534.3 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.82 (s, 1H), 7.77 (d, J=8.5 Hz, 1H), 7.60 (d, J=7.5 Hz, 1H), 7.52-7.49 (m, 1H), 7.45-7.43 (m, 2H), 7.35-7.32 (m, 3H), 7.27-7.24 (m, 1H), 6.85 (d, J=12.5 Hz, 1H), 4.63 (s, 1H), 4.24 (d, J=6.0 Hz, 2H), 3.47-3.41 (m, 2H), 3.33 (s, 3H), 3.23-3.20 (m, 2H), 3.04-3.02 (m, 1H), 2.12-2.09 (m, 1H), 0.96-0.94 (m, 2H), 0.69-0.68 (m, 2H).
##STR00424##
Example 110
(S)-4-((1-((4-Cyanophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-cyclopro- pyl-2-fluoro-N-(methylsulfonyl)benazmide
[0735] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=4.13 min). LCMS(ESI) Method A: RT=5.64 min, m/z: 534.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.69 -7.64 (m, 4H), 7.43 (d, J=7.0 Hz, 2H), 7.34-7.31 (m, 3H), 7.26-7.23 (m, 1H), 6.84 (d, J=13.0 Hz, 1H), 4.65 (s, 1H), 4.23 (d, J=6.0 Hz, 2H), 3.48-3.40 (m, 2H), 3.31 (s, 3H), 3.25-3.18 (m, 2H), 3.05-3.01 (m, 1H), 2.12-2.08 (m, 1H), 0.97-0.93 (m, 2H), 0.70-0.67 (m, 2H).
Example 111
##STR00425##
[0736] (R)-4-((1-((4-Cyanophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-c- yclopropyl-2-fluoro-N-(methylsulfonyl)benazmide
[0737] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=6.36 min). LCMS(ESI) Method A: RT=5.64 min, m/z: 534.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.69-7.64 (m, 4H), 7.43 (d, J=7.5 Hz, 2H), 7.34-7.30 (m, 3H), 7.26-7.23 (m, 1H), 6.82 (d, J=13.0 Hz, 1H), 4.63 (s, 1H), 4.22 (d, J=5.5 Hz, 2H), 3.47-3.39 (m, 2H), 3.29 (s, 3H), 3.24-3.16 (m, 2H), 3.05-3.00 (m, 1H), 2.13-2.07 (m, 1H), 0.96-0.92 (m, 2H), 0.70-0.66 (m, 2H).
Example 112
##STR00426##
[0738] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(4-(trif- luoromethoxy)phenyl)-methyl)azetidin-3-yl)methoxy)benazmide
[0739] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.degree. C.; RT=3.62 min). LCMS(ESI) Method A: RT=6.36 min, m/z: 593.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.55-7.53 (m, 2H), 7.44-7.42 (m, 2H), 7.34-7.31 (m, 3H), 7.26-7.22 (m, 3H), 6.80 (d, J=12.0 Hz, 1H), 4.63 (s, 1H), 4.21 (d, J=6.0 Hz, 2H), 3.47-3.45 (m, 2H), 3.33-3.31 (m, 5H), 3.10-3.00 (m, 1H), 2.10-2.09 (m, 1H), 0.95-0.92 (m, 2H), 0.68-0.67 (m, 2H).
Example 113
##STR00427##
[0740] (R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(4-(trif- luoromethoxy)phenyl)methyl)-azetidin-3-yl)methoxy)benzamide
[0741] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 4.degree. C.; RT=4.11 min). LCMS(ESI) Method B: RT=6.38 min, m/z: 593.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 7.55-7.53 (m, 2H), 7.44-7.42 (m, 2H), 7.36-7.32 (m, 3H), 7.27-7.23 (m, 3H), 6.84 (d, J=12.5 Hz, 1H), 4.71 (s, 1H), 4.23 (d, J=6.0 Hz, 2H), 3.53-3.51 (m, 2H), 3.40-3.20 (m, 5H), 3.08-3.07 (m, 1H), 2.12-2.09 (m, 1H), 0.97-0.93 (m, 2H), 0.70-0.67 (m, 2H).
Example 114
##STR00428##
[0742] (S)-4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)(phenyl)methyl)azetid- in- 3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0743] The compound was synthesized as described in Example 81. Chiral HPLC (column: OH-J, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=4.49 min). LCMS(ESI) Method A: RT=5.88 min, m/z: 602.1 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 8.04 (d, J=5.0 Hz, 1H), 7.46 (m, 2H), 7.36-7.25 (m, 5H), 6.84 (d, J=13.0 Hz, 1H), 5.34-5.29 (m, 1H), 5.01 (s, 1H), 4.19 (d, J=5.5 Hz, 2H), 3.53-3.50 (m, 1H), 3.30-3.28 (m, 5H), 3.15-3.12 (m, 1H), 3.04-3.01 (m, 1H), 2.19-2.15 (m, 1H), 1.36 (d, J=6.5 Hz, 3H), 1.32 (d, J=6.5 Hz, 3H), 1.01-0.97 (m, 2H), 0.71-0.70 (m, 2H).
Example 115
##STR00429##
[0744] (R)-4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)(phenyl)methyl)azetid- in-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0745] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=4.49 min). LCMS(ESI) Method A: RT=5.56 min, m/z: 602.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d4): .delta. 8.04 (d, J=5.0 Hz, 1H), 7.46 (m, 2H), 7.36-7.25 (m, 5H), 6.85 (d, J=13.0 Hz, 1H), 5.34-5.30 (m, 1H), 5.01 (s, 1H), 4.19 (d, J=5.0 Hz, 2H), 3.53-3.50 (m, 1H), 3.32-3.30 (m, 5H), 3.15-3.12 (m, 1H), 3.04-3.01 (m, 1H), 2.19-2.15 (m, 1H), 1.36 (d, J=6.5 Hz, 3H), 1.32 (d, J=6.5 Hz, 3H), 1.01-0.97 (m, 2H), 0.71-0.70 (m, 2H).
Example 116
##STR00430##
[0746] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(2,2,2-trifluoroeth- yl)-piperidin-4-yl)methoxy)-benzamide
##STR00431##
[0747] Step 1
##STR00432##
[0748] Methyl 5-cyclopropyl-2-fluoro-4-((1-(2,2,2-trifluoroethyl)piperidin- 4-yl)methoxy)benzoate
[0749] A Mixture of methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (180 mg, 0.59 mmol), 2,2,2-trifluoroethyl trifluoromethanesulfonate (151 mg, 0.65 mmol) and DIPEA (152 mg, 1.18 mmol) in THF (15 mL) was stirred at 60.degree. C. for 2 h. The reaction mixture was diluted with ethyl acetate (30 mL), washed with brine (50.times.2 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=5/1) to give the target compound (160 mg, 69%) as a yellow solid. LCMS(ESI) m/z: 390.2 [M+H].sup.+.
Step 2
##STR00433##
[0750] 5-Cyclopropyl-2-fluoro-4-((1-(2,2,2-trifluoroethyl)piperidin-4- yl)methoxy)benzoic acid
[0751] A mixture of methyl 5-cyclopropyl-2-fluoro-4-((1-(2,2,2-triflouroethyl)piperidin- 4-yl)methoxy)benzoate (160 mg, 0.4 mmol) and lithium hydroxide (250 mg, 10.0 mmol) in THF (10 mL) and H.sub.2O (10 mL) was stirred at room temperature for 2 h. The mixture was diluted with EtOAc (50 mL), washed with HCl (2.0 M, 10 mL), brine (50.times.2 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the target compound (120 mg) as yellow solid which was used in the next step without further purification. LCMS(ESI) m/z: 376.0 [M+H].sup.+.
Step 3
##STR00434##
[0752] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(2,2,2-trifluoroeth- yl)-piperidin-4-yl)methoxy)-benzamide
[0753] A mixture of 5-cyclopropyl-2-fluoro-4-((1-(2,2,2-trifluoroethyl)piperidin- 4-yl)methoxy)benzoic acid (120 mg, 0.32 mmol), methanesulfonamide (45.6 mg, 0.48 mmol), EDCI (92 mg, 0.48 mmol) and DMAP (59 mg, 0.48 mmol) in DCM (20 mL) was stirred at 25.degree. C. for 1 h. The reaction mixture was diluted with EtOAc (100 mL), washed with HCl (2.0 M, 20 mL), brine (50.times.2 mL), dried over anhydrous sodium sulfide, filtered and concentrated. The residue was purified by reverse phase combiflash (25%-30% MeCN in 0.5% NH.sub.4HCO.sub.3) to give the target product (65.0 mg, 45%) as a white solid. LCMS(ESI) Method A: RT=5.97 min, m/z: 453.1 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOH-d.sub.4): .delta. 7.36 (d, J=8.5 Hz, 1H), 6.65 (d, J =12.5 Hz, 1H), 3.90 (d, J=5.5 Hz, 2H), 3.11-3.05 (m, 7H), 2.46-2.42 (m, 2H), 2.08-2.06 (m, 1H), 1.89-1.86 (m, 3H), 1.55-1.52 (m, 2H), 0.91-0.87 (m, 2H), 0.67-0.05 (m, 2H).
Example 117
##STR00435##
[0754] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(3,3,3-trifluoropro- pyl) piperidin-4-yl)methoxy)-benzamide
[0755] The compound was synthesized as described in Example 37. LCMS(ESI) Method A: RT=4.66 min, m/z: 467.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.24 (d, J=8.5 Hz, 1H), 6.56 (d, J=12.5 Hz, 1H), 3.81 (d, J=5.5 Hz, 2H), 3.01 (s, 3H), 2.96-2.94 (m, 2H), 2.60 (m, 2H), 2.39-2.33 (m, 2H), 2.11-2.07 (m, 2H), 1.97-1.93 (m, 1H), 1.84-1.81 (m, 2H), 1.47-1.42 (m, 2H), 1.22-1.20 (m, 1H), 0.79-0.76 (m, 2H), 0.54-0.53 (m, 2H).
Example 118
##STR00436##
[0756] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin- -2-yl)methyl)azetidin-3-yl)methoxy)benzamide
[0757] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=4.55 min). LCMS(ESI) Method A: RT=4.86 min, m/z 510.3 [M+H].sup.+. .sup.1H NMR (500 MHz, CDCl.sub.3): .delta. 8.51 (d, J=3.0 Hz, 1H), 7.62-7.57 (m, 2H), 7.50-7.47 (m, 3H), 7.29-7.22 (m, 4H), 7.11 (s, 1H), 6.61 (d, J=14.5 Hz, 1H), 4.61 (s, 1H), 4.20 (d, J=6.0 Hz, 2H), 3.41 (d, J=5H), 3.14 (s, 1H), 3.01 (s, 1H), 2.04 (m, 1H), 0.94 (m, 2H), 0.65 (m, 2H).
Example 119
##STR00437##
[0758] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin- -2-yl)methyl)azetidin-3-yl)methoxy)benzamide
[0759] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=5.70 min). LCMS(ESI) Method A: RT=4.86 min m/z: 510.3 [M+H].sup.+. .sup.1H NMR (500 MHz, CDCl.sub.3): 8.50 (d, J=4.5 Hz, 1H), 7.64-7.57 (m, 2H), 7.50-7.47 (m, 3H), 7.31-7.21 (m, 4H), 7.12-7.10 (m, 1H), 6.60 (d, J=14.5 Hz, 1H), 4.60 (s, 1H), 4.20 (d, J=6.0 Hz, 2H), 3.41 (s, 5H), 3.15 (s, 1H), 3.01 (s, 1H), 2.06-2.01 (m, 1H), 0.93 (m, 2H), 0.65 (m, 2H).
Example 120
##STR00438##
[0760] (S)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00439##
[0761] Step 1
##STR00440##
[0762] (S,E)-N-(1-(3,5-Dichlorophenyl)ethylidene)-2-methylpropane-2-sulfin- amide
[0763] Tetraisopropoxytitanium (6.14 g, 21.2 mmol) was added to a solution of 3,5-dichlorophenyl ethanone (1.0 g, 5.3 mmol) and (S)-2-methylpropane-2- sulfinamide (128 g, 10.6 mmol) in dry THF (20 mL). After stirring at 80.degree. C. for 16 h, ethyl acetate (15 mL) and brine was added to quench the reaction. The white precipitate was filtered and the filtrate was concentrated and purified by silica gel chromatography (eluting with ethyl acetate/petroleum ether=1/10) to give the target compound as a yellow solid (1.24 g, 80%). LCMS(ESI): m/z 292.0 [M+H].sup.+.
Step 2
##STR00441##
[0764] (S)-N-(S)-1-(3,5-Dichlorophenyl)ethyl-2-methylpropane-2-sulfinamide
[0765] Sodium borohydride (0.49 g, 12.9 mmol) was added to a solution (S, E)-N-(1-(3,5-dichlorophenyl)ethylidene)-2-methylpropane-2-sulfinamide (1.24 g, 4.3 mmol) in THF (20 mL, with 2% H.sub.2O) at -60.degree. C. After stirring at room temperature for 3 h, the solvent was removed under reduced pressure and the crude product was purified by silica gel chromatography (eluting with ethyl acetate/petroleum ether=1/12) to afford target compound (1.07 g, 85%) as white solid. LCMS(ESI): m/z 294.0 [M+H].sup.+. .sup.1H NMR (CDCl.sub.3, 500 MHz): .delta. 7.28 (t, J=2.0 Hz, 1H), 7.23 (d, J=2.0 Hz, 2H), 4.48 (m, 1 H), 3.41 (d, J=2.5 Hz, 1H), 1.49 (d, J=6.5 Hz, 3H), 1.24 (s, 9H).
Step 3
##STR00442##
[0766] (S)-1-(3,5-Dichlorophenyl)ethanamine hydrochloride
[0767] HCl (1M in MeOH, 10 mL) was added to a solution of (S)-N-(S)-1-(3,5-dichlorophenyl)ethyl)-2-methylpropane-2-sulfinamide (1.07 g, 3.66 mmol) in MeOH (5 ml). After stirring at room temperature for 2 h, the mixture was diluted with ethyl acetate (30 mL). The resultant white precipitate was filtered and washed with ethyl acetate (10 mL) to provide the target compound (820 mg, 99%) as a white solid. LCMS (ESI): m/z 190.0 [M+H].sup.+.
Step 4
##STR00443##
[0768] (S)-tert-Butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3- yl)methoxy)-2-fluorobenzoate
[0769] The compound was synthesized as described in step 4 of Example 5 LCMS(ESI): m/z 494.1 [M+H].sup.+.
Step 5
##STR00444##
[0770] (S)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3-yl)- methoxy)- 2-fluorobenzoic acid
[0771] The compound was synthesized as described in step 5 of Example 5. LCMS(ESI): m/z 438.1 [M+H].sup.+.
Step 6
##STR00445##
[0772] (S)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3-yl)- methoxy)- 2-fluoro-N-(methylsulfonyl)benzamide
[0773] The compound was synthesized as described in step 6 of Example 5. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=7.28 min). LCMS(ESI) Method A: RT=5.55 min m/z 514.9 [M+H].sup.+. .sup.1H-NMR (CDCl.sub.3, 500 MHz: .delta. 7.58 (d, J=9.0 Hz, 1H), 7.23 (m, 1H), 7.19 (d, J=2.0 Hz, 2H), 6.59 (d, J=14.5 Hz, 1H), 4.17 (m, 2H), 3.38 (m, 4H), 3.30 (m, 2H). 3.07 (m, 2H), 2.92 (m, 1H), 2.02 (m, 1H), 1.18 (d, 3H), 0.94 (m, 2H), 0.66 (m, 2H).
Example 121
##STR00446##
[0774] (R)-5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)azetidin-3-yl)- methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0775] The compound was synthesized as described in Example 41. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C,; RT=5.37 min). LCMS(ESI) Method A: RT=5.41 min m/z 514.8 [M+H].sup.+. .sup.1H-NMR CDCl.sub.3, 500 MHz): .delta. 7.58 (d, J=9.0 Hz, 1H), 7.23 (m, 1H), 7.19 (d, J=2.0 Hz, 2H), 6.59 (d, J=14.5 Hz, 1H), 4.17 (m, 2H), 3.38 (m, 4H), 3.30 (m, 2H), 3.07 (m, 2H), 2.92 (m, 1H), 2.02 (m, 1H), 1.18 (d, 3H), 0.94 (m, 2H), 0.66 (m, 2H).
Example 122
##STR00447##
[0776] 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-methylpiperidin-4-y- l)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0777] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 5-chloro-2-(trifluoromethyl)benzaldehyde. LCMS (Method F): RT=4.76 min, m/z: 577.2 [M+H].sup.+.
Example 123
##STR00448##
[0778] 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-4-methylpiperidin-4-yl)met- hoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0779] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 3,5-dichlorobenzaldehyde. LCMS (Method F): RT=4.64 min, m/z: 543.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.54 (t, J=1.9 Hz, 1H), 7.41 (d, J=1 -2.0 Hz, 2H), 7.20 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.9 Hz, 1H), 3.84 (s, 2H), 3.72 (s, 2H), 3.19 (s, 3H), 2.76-2.61 (m, 2H), 2.06-1.96 (m, 1H), 1.76-1.64 (m, 2H), 1.50 (d, J=14.0 Hz, 2H), 1.09 (s, 3H), 0.94-0.83 (m, 2H), 0.69-0.59 (m, 2H).
Example 124
##STR00449##
[0780] (S)-5-Cyclopropyl-2-fluoro-4-((1-((2-fluorophenyl)(phenyl)methyl)az- etidin-3-yl)methoxy)-N-(methylsulfonyl)benzamide
[0781] The compound was synthesized as described in Example 81. Chiral HPLC (column: OH-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=85:15; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=14.2 min). LCMS(ESI) Method A: RT=5.48 min, m/z: 527.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6,): .delta.7.64-7.62 (m, 1H), 7.43-7.03 (m, 9H), 6.81 (d, J=12.5 Hz, 1H), 5.00 (s, 1H), 4.18 (d, J=5.0 Hz, 2H), 3.55-3.46 (m, 3H), 3.28 (m, 4H), 3.07-3.05 (m, 1H), 2.16-2.13 (m, 1H), 0.98-0.96 (m, 2H), 0.69-0.68 (m, 2H).
Example 125
##STR00450##
[0782] (R)-5-Cyclopropyl-2-fluoro-4-((1-((2-fluorophenyl)(phenyl)methyl)az- etidin-3-yl)methoxy)-N-(methylsulfonyl)benzamide
[0783] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=85:15; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=16.3 min). LCMS(ESI) Method A: RT=5.69 min, m/z: 527.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): (m, 1H), 7.43-7.03 (m, 9H), 6.83 (d, J=13.0 Hz, 1H), 5.05 (s, 1H), 4.19 (d, J=5.5 Hz, 2H), 3.59-3.40 (m, 4H), 3.28 (s, 3H), 3.10-3.08 (m, 1H), 2.17-2.13 (m, 1H), 0.98-0.96 (m, 2H), 0.71-0.68 (m, 2H).
Example 126
##STR00451##
[0784] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(4-(trifluoromethyl- )piperidin)piperidin-4-yl)methoxy)benzamide
[0785] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.54 min, m/z: 528.9 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6,): .delta.7.74-7.58 (m, 4H), 7.16 (d, J=8.5 Hz, 1H), 6.86 (d, J =12.5 Hz, 1H), 3.94 (d, J=6.0 Hz, 2H), 3.79 (s, 2H), 3.16 (s, 3H), 2.99-2.98 (m, 2H), 2.29-2.26 (m, 2H), 2.02-1.99 (m, 1H), 1.88-1.82 (m, 3H), 1.44-1.42 (m, 2H), 0.89-0.86 (m, 2H), 0.64-0.61 (m, 2H).
Example 127
##STR00452##
[0786] 5-Cyclopropyl-2-fluoro-4-((1-(2-fluorobenzyl)piperidin-4-yl)methoxy- )- N-(methylsulfonyl)benzamide
[0787] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.89 min, m/z: 479.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, MeOH-d.sub.4,): .delta.7.46-7.36 (m, 2H), 7.23-7.16 (m, 3H), 6.83 (d, J=12.5 Hz, 1H), 3.92-3.91 (m, 2H), 3.76 (s, 2H), 3.11 (s, 3H), 3.03-3.01 (m, 2H), 2.33-2.30 (m, 2H), 2.02-1.82 (m, 4H), 1.46-1.42 (m, 2H), 0.89-0.86 (m, 2H), 0.62-0.60 (m, 2H).
Example 128
##STR00453##
[0788] 4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)piperidin-4-yl)met- hoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0789] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.86 min, m/z: 553.8 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6,): .delta.8.06 (d, J=5.0 Hz, 1H), 7.16-6.88 (m, 3H), 5.30-5.28 (m, 1H), 3.94 (d, J=3 6.0 Hz, 2H), 3.60 (s, 2H), 3.21 (s, 3H), 2.89-2.87 (m, 2H), 2.16-2.12 (m, 2H), 2.03-2.01 (m, 1H), 1.80-1.78 (m, 3H), 1.41-1.39 (m, 2H), 1.31 (d, J=6.5 Hz, 6H), 0.90-0.87 (m, 2H), 0.66-0.63 (m, 2H).
Example 129
##STR00454##
[0790] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenylsulfonyl)- azetidin-3-yl)methoxy)benzamide
##STR00455##
[0791] Step 1
##STR00456##
[0792] tert-Butyl 5-cyclopropyl-2-fluoro-4-((1-(phenylsulfonyl)azetidin-3- yl)methoxy)benzoate
[0793] Benzenesulfonyl chloride (54 mg, 0.31 mol) was added to a mixture of tert-butyl 4-(azetidin-3-ylmethoxy)-5-cyclopropyl-2-fluorobenzoate (100 mg, 0.31 mmol) and triethylamine (94 mg, 0.93 mmol) in DCM (10 ml). After stirring at room temperature for 2 h, the mixture was quenched with water (10 ml), extracted with DCM (10 ml.times.3), dried over sodium sulfate, filtered and concentrated to give target compound as a yellow solid. (112 mg, 78%). LCMS(ESI) m/z: 462.1 [M+H].sup.+.
Step 2
##STR00457##
[0794] 5-Cyclopropyl-2-fluoro-4-((1-(phenylsulfonyl)azetidin-3-yl)methoxy)- benzoic acid
[0795] The compound was synthetized as described in step 3 of Example 88.
Step 3
##STR00458##
[0796] 5-Cyclopropyl-2-fluoro-4-((1-(phenylsulfonyl)azetidin-3-yl)methoxy)- benzamide
[0797] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=4.66 min, m/z: 483.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6,): .delta.7.89-7.66 (m, 5H), 7.26 (d, J=8.0 Hz, 1H), 6.76 (d, J=12.5 Hz, 1H), 4.02 (d, J=5.5 Hz, 2H), 3.97-3.94 (m, 2H), 3.83-3.81 (m, 2H), 3.36 (s, 3H), 2.99-2.96 (m, 1H), 1.91-1.88 (m, 1H), 0.91-0.88 (m, 2H), 0.63-0.60 (m, 2H).
Example 130
##STR00459##
[0798] 5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2-(trifluoromethyl)piperidin- )-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0799] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidin-1-carboxylate (Example 73 step 1-2) and 4-fluoro-2-trifluoromethylbenzaldehyde. LCMS (Method F): RT=4.62 min, m/z: 561.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.68 (s, 1H), 7.87-7.78 (m, 1H), 7.62-7.49 (m, 2H), 7.18 (d, J=8.3 Hz, 1H), 6.95 (d, J=13.0 Hz, 1H), 3.85 (s, 2H), 3.66 (s, 2H), 3.28 (s, 3H), 2.61-2.52 (m, 2H), 2.46-2.36 (m, 2H), 2.06-1.95 (m, 1H), 1.74-1.61 (m, 2H), 1.47 (d, J=13.3 Hz, 2H), 1.09 (s, 3H), 0.93-0.84 (m, 2H), 0.70-0.61 (m, 2H).
Example 131
##STR00460##
[0800] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(4-(trifluor- omethyl)phenyl)methyl)- azetidin-3-yl)methoxy)benzamide
[0801] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=6.00 min, m/z: 577.3 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.65-7.60 (m, 4H), 7.44-7.43 (m, 2H), 7.34-7.31 (m, 3H), 7.25-7.23 (m, 1H), 6.81-6.78 (m, 1H), 4.65 (s, 1H), 4.21 (d, J=6.0 Hz, 2H), 3.48-3.41 (m, 2H), 3.29-3.19 (m, 5H), 3.04-3.01 (m, 1H), 2.12-2.08 (m, 1H), 0.96-0.68 (m, 2H), 0.67 (s, 2H).
Example 132
##STR00461##
[0802] 5-Cyclopropyl-4-((1-(3,5-dichlorobenzyl)azetidin-3-yl)methoxy)-2-fl- uoro-N-(methylsulfonyl)benzamide
[0803] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=5.26 min, m/z: 501.1 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.56 (s, 1H), 7.41-7.41 (m, 2H), 7.20-7.18 (m, 1H), 6.90-6.88 (m, 1H), 4.20-4.18(d, J=6.5 Hz, 2H), 3.85 (s, 2H), 3.63 (s, 2H), 3.42 (s, 2H), 3.14 (s, 3H), 3.02-3.00 (m, 1H), 2.06-2.02 (m, 1H), 0.91-0.87 (m, 2H), 0.65-0.62 (m, 2H).
Example 133
##STR00462##
[0804] 4-((1-(4-Chloro-3-fluorobenzyl)azetidin-3-yl)methoxy)-5cyclopropyl-- 2-fluoro-N-(methylsulfonyl)benzamide
[0805] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=4.81 min, m/z: 485.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.58 (t, J=8.0 Hz, 1H), 7.38 (d, J=9.5, 1H), 7.22-7.19 (m, 2H), 6.86 (d, J=12.5 Hz, 1H), 4.19 (d, J=6.0 Hz, 2H), 3.87 (s, 2H), 3.63 (s, 2H), 3.49 (s, 2H), 3.10 (s, 3H), 3.02-3.00 (m, 1H), 2.04-2.00 (m, 1H), 0.90-0.86 (m, 2H), 0.64-0.61 (m, 2H).
Example 134
##STR00463##
[0806] 5-Cyclopropyl-4-((1-(3,5-dichlorobenzoyl)azetidin-3-yl)methoxy)-2- fluoro-N-(methylsulfonyl)benzamide
##STR00464##
[0807] Step 1
##STR00465##
[0808] tert-Butyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzoyl)azetidin-3-yl)methoxy)-2- fluorobenzoate
[0809] A mixture of 3,5-dichlorobenzoic acid (100 mg 0.52 mmol), tert-butyl 4-(azetidin-3-ylmethoxy)- 5-cyclopropyl-2-fluorobenzoate (140 mg, 0.44 mmol), EDCI (140 mg, 0.72 mmol) and DMAP (27 mg, 0.22 mmol) in DCM (5 mL) was stirred at room temperature for 18 h. The mixture was diluted with DCM (10 mL) and washed with HCl (2 N, 15 mL.times.2). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/EtOAc=4/1) to give the target compound (200 mg, 92%) as a white solid. LCMS(ESI) m/z: 437.9 [M+H].sup.+. .sup.1H NMR
Step 2
##STR00466##
[0810] 5-Cyclopropyl-4-((1-(3,5-dichlorobenzoyl)azetidin-3-yl)methoxy)-2-f- luorobenzoic acid
[0811] The compound was synthesized as described in step 3 of Example 88. LCMS(ESI) m/z: 438.0 [M+H].sup.+. .sup.1H NMR
Step 3
##STR00467##
[0812] 5-Cyclopropyl-4-((1-(3,5-dichlorobenzoyl)azetidin-3-yl)methoxy)-2-f- luoro- N-(methylsulfonyl)benzamide
[0813] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=4.65 min, m/z: 515.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.90 (s, 1H), 7.80 (t, J=1.5 Hz, 1H), 7.8 (d, J=2.0 Hz, 2H), 7.15 (d, J=8.0 Hz, 1H), 6.96 (d, J=12.5 Hz, 1H), 4.52 (t, J=8.5 Hz, 1H), 4.27-4.16 (m, 1H), 3.99-3.97 (m, 1H), 3.28 (s, 3H), 3.12-3.09 (m, 1.95-1.89 (m, 1H), 0.89-0.84 (m, 1H), 0.76-0.60 (m, 3H).
Example 135
##STR00468##
[0814] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(4-(trifluoromethox- y) benzyl)piperidin-4-yl)methoxy)benzamide
[0815] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.55 min, m/z: 544.8 [M+H].sup.+. .sup.1H NMR R (500 MHz, DMSO-d.sub.6): .delta. 7.51 (d, J=8.5 Hz, 2H), 7.38 (d, J=8.0 Hz, 2H), 7.17 (d, J=8.5 Hz, 1H), 6.83 (d, J=12.5 Hz, 1 H), 3.92 (d, J=6.5 Hz, 2H), 3.827 (s, 2H), 3.11 (s, 3H), 3.05 (d, J=11.0 Hz, 2H), 2.41-2.37 (m, 2H), 2.03-1.98 (m, 1H), 1.90-1.85 (m, 3H), 1.48-1.42 (m, 2H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 136
##STR00469##
[0816] 4-((1-(3-Chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl)methoxy)-- 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0817] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.15 min, m/z: 578.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.48 (s, 2H), 7.35 (s, 1H), 7.15 (d, J=8.0 Hz, 1 H), 6.87 (d, J=13.5 Hz, 1H), 3.93 (d, J=5.5 Hz, 2H), 3.67 (s, 2H), 3.18 (s, 3H), 2.90 (d, J=10 Hz, 2H), 2.20-2.15 (m, 2H), 2.07-1.98 (m, 1H), 1.88-1.80 (m, 3H), 1.44-1.36 (m, 2H), 0.89-0.86 (m, 2H), 0.65-0.62 (m, 2H).
Example 137
##STR00470##
[0818] 4-((1-(3-Chloro-5-(trifluoromethyl)piperidin)piperidin-4-yl)methoxy- )-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0819] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.06 min, m/z: 562.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.76 (s, 2H), 7.73 (s, 1H), 7.67 (s, 1H), 7.18 (d, J=8.5 Hz, 1 H), 6.82 (d, J=11.5 1 H), 3.93 (d, J=5.5 2H), 3.67 (s, 2H), 3.10 (s, 3H), 2.89 (d, J=10.5 Hz,
Example 138
##STR00471##
[0820] 4-((1-(4-Chloro-3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropy- l-2-fluoro-N-(methylsulfonyl)benzamide
[0821] The compound synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.72 min, m/z: 513.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.56-7.23 (m, 1H), 7.60-7.58 (m, 1H), 7.38 (d, J=8.0 Hz, 1H), 7.13 (d, J=8.0 Hz, 1 H), 6.97 (d, J=12.5 Hz, 1 H), 4.32 (s, 2H), 3.98 (s, 2H), 3.42-3.34 (m, 2H), 3.33 (s, 3H), 2.97 (s, 2H), 2.06-1.97 (m, 4), 1.58-1.56 (m, 2H), 0.90-0.86 (m, 2H), 0.70-0.67(m, 2H).
Example 139
##STR00472##
[0822] 5-Cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-4-yl)methoxy)- 2-fluoro-N-(methylsulfonyl)benzamide
[0823] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.75 min, m/z: 528.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.53 (s, 1H), 7.40 (s, 2H), 7.16 (d, J=8.5 Hz, 1H), 6.84 (d, J=12.0 Hz, 1H), 3.93 (d, J=5.5 Hz, 2H), 3.64-3.61 (m, 2H), 3.14 (s, 3H), 2.93-2.91 (m, 2H), 2.17-2.16 (m, 2H), 2.03-2.00 (m, 1H), 1.82-1.80 (m, 3H), 1.41-1.39 (m, 2H), 0.90-0.86 (m, 2H), 0.63-0.62 (d, 2H).
Example 140
##STR00473##
[0824] 4-((1-(3-chloro-5-(trifluoromethyl)piperidin)-4-methylpiperidin-4-y- l)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0825] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 3-chloro-5-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=5.81 min, m/z: 577.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.81-7.65 (m, 3H), 7.20 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.9 Hz, 1H), 3.84 (s, 2H), 3.76 (s, 2H), 3.20 (s, 3H), 2.72-2.58 (m, 2H), 2.07-1.96 (m, 1H), 1.78-1.65 (m, 2H), 1.56-1.43 (m, 2H), 1.09 (s, 3H), 0.92-0.83 (m, 2H), 0.68-0.59 (m, 2H).
Example 141
##STR00474##
[0826] 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-methylpipe- ridin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0827] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 3-chloro-2-fluoro-5-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=6.67 min, m/z: 595.14 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.58 (s, 1H), 8.02 (dd, J=6.4, 2.3 Hz, 1H), 7.80 (dd, J=5.9, 2.3 Hz, 1H), 7.19 (d, J=8.5 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 3.84 (s, 2H), 3.75 (s, 2H), 3.24 (s, 3H), 2.72-2.57 (m, 2H), 2.08-1.96 (m, 1H), 1.77-1.62 (m, 2H), 1.53-1.40 (m, 2H), 1.07 (s, 3H), 0.93-0.82 (m, 2H), 0.69-0.59 (m, 2H).
Example 142
##STR00475##
[0828] (S)-5-Cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)azetidi- n-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0829] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=65:35; flow: 195 mL/min; column temperature: 40.degree. C.; RT=4.15 min). LCMS(ESI) Method A: RT=6.20 mm, m/z: 577.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, MeOD-d.sub.4): .delta.7.30-7.28 (m, 4H), 7.23-7.20 (m, 3H), 7.17-7.12 (m, 2H), 6.70 (d, J=12.5 Hz, 1H), 4.42 (s, 1H), 4.10 (d, J=7.0 Hz, 2H), 3.33-3.26 (m, 2H), 3.16 (s, 3H), 3.10-3.05 (m, 2H), 2.90-2.87 (m, 1H), 2.00-1.96 (m, 1H), 0.86-0.80 (m, 2H), 0.56-0.54 (m, 2H).
Example 143
##STR00476##
[0830] (R)-5-Cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)azetidi- n-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0831] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=5.47 min). LCMS(ESI) Method A: RT=6.19 min, m/z: 577.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, MeOD-d.sub.4): .delta.7.30-7.28 (m, 4H), 7.23-7.20 (m, 3H), 7.17-7.12 (m, 2H), 6.70 (d, J=12.5 Hz, 1H), 4.42 (s, 1H), 4.10 (d, J=7.0 Hz, 2H), 3.33-3.26 (m, 2H), 3.16 (s, 3H), 3.10-3.05 (m, 2H), 2.90-2.87 (m, 1H), 2.00-1.96 (m, 1H), 0.86-0.80 (m, 2H), 0.56-0.54 (m, 2H).
Example 144
##STR00477##
[0832] (S)-5-Cyclopropyl-2-fluoro-4-((1-((4-fluorophenyl)(phenyl)methyl)az- etidin-3-yl)methoxy)-N-(methylsulfonyl)benzamide
[0833] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=5.77 min). LCMS(ESI) Method A: RT=4.83 min, m/z: 527.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, MeOH-d.sub.4): .delta. 7.35-7.29 (m, 4H), 7.24-7.20 (m, 3H), 7.16-7.12 (m, 1H), 6.97-6.93 (m, 2H), 6.67 (d, J=13.0 Hz, 1H), 4.63 (s, 1H), 4.08 (d, J=6.0 Hz, 2H), 3.45-3.43 (m, 2H), 3.25-3.22 (m, 2H), 3.15 (s, 3H), 2.98-2.95 (m, 1H), 1.99-1.95 (m, 1H), 0.84-0.79 (m, 2H), 0.56-0.54 (m, 2H).
Example 145
##STR00478##
[0834] (R)-5-Cyclopropyl-2-fluoro-4-((1-((4-fluorophenyl)(phenyl)methyl)-a- zetidin-3-yl)methoxy)-N-(methylsulfonyl)benzamide
[0835] The compound was synthesized as described in Example 88. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B:75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=6.50 min). LCMS(ESI) Method A: RT=4.79 min, m/z: 527.2 [M+H].sup.+. .sup.1H-NMR (500 MHz, MeOH-d.sub.4): .delta. 7.35-7.29 (m, 3H), 7.24-7.20 (m, 3H), 7.16-7.12 (m, 1H), 6.97-6.93 (m, 2H), 6.67 (d, J=13.0 Hz, 1H), 4.63 (s, 1H), 4.08 (d, J=6.0 Hz, 2H), 3.45-3.43 (m, 2H), 3.25-3.22 (m, 2H), 3.15 (s, 3H), 2.98-2.95 (m, 1H), 1.99-1.95 (m, 1H), 0.84-0.79 (m, 2H), 0.56-0.54 (m, 2H).
Example 146
##STR00479##
[0836] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-phenylethyl)- piperidin-4-yl)methoxy)benzamide
[0837] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.70 min, m/z: 476.2 [M+H].sup.+. 1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.42-7.36 (m, 5H), 7.18 (d, J=8.0 Hz, 1H), 6.78 (d, J=13.0 Hz, 1H), 4.04 (brs, 1H), 3.90 (d, J=5.0 Hz, 2H), 3.32 (s, 3H), 3.02 (m, 4H), 2.02-1.97 (m, 1H), 1.90-1.84 (m, 3H), 1.50-1.49 (m, 5), 0.88-0.83 (m, 2H), 0.60-0.57 (m, 2H).
Example 147
##STR00480##
[0838] 5-Cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-4-yl)methoxy)-2-f- luoro-N-(methylsulfonyl)benzamide
[0839] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.75 min, m/z: 528.9 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.64-7.63 (m, 2H), 7.37-7.36 (m, 1H), 7.17-7.16 (m, 1H), 6.85 (d, J=13.0 Hz, 1H), 3.94 (s, 2H), 3.72 (s, 2H), 3.15 (s, 3H), 3.00-298 (m, 2H), 2.29 (s, 2H), 2.02-2.01 (m, 1H), 1.85-1.82 (m, 3H), 1.44-1.42 (m, 2H), 0.88-0.87 (m, 2H), 0.63 (s, 2H).
Example 148
##STR00481##
[0840] 4-((1-(1-(3-Chlorophenyl)ethyl)piperidin-4-yl)methoxy)-5-cyclopropy- l-2-fluoro-N-(methylsulfonyl)benzamide
[0841] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.52 min, m/z: 508.9 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.40-7.31 (m, 4H), 7.17 (d, J=8.5 Hz, 1H), 6.83 (d, J =12.5 Hz, 1H), 3.92-3.91 (m, 3H), 3.31-3.27 (m, 4H), 3.17-3.16 (m, 1H), 2.98-2.96 (m, 1H), 2.27-2.26 (m, 1H), 2.02-1.98 (m, 1H), 1.88-1.80 (m, 3H), 1.46-1.41 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 149
##STR00482##
[0842] 4-((1-(2-Cyanobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro- - N-(methylsulfonyl)benzamide
[0843] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.86 min, m/z: 486.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.83-7.81 (m, 1H), 7.71-7.67 (m, 1H), 7.60-7.59 (m, 1H), 7.50-7.47 (m, 1H), 7.15 (d, J=8.0 Hz, 1H), 6.91 (d, J=12.5 Hz, 1H), 3.94 (d, J=6.0 Hz, 2H), 3.70 (s, 2H), 3.25 (s, 3H), 2.90-2.88 (m, 2H), 2.19-2.15 (m, 2H), 2.03-2.00 (m, 1H), 1.84-1.78 (m, 3H), 1.42-1.34 (m, 2H), 0.90-0.86 (m, 2H), 0.66-0.63 (m, 2H).
Example 150
##STR00483##
[0844] (S)-4-((1-((2-Chlorophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-- cyclopropyl-2-fluoro- N-(methylsulfonyl)benzamide
[0845] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=4.38 min). LCMS(ESI) Method A: RT=6.04 min, m/z: 543.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.89 (s, 1H), 7.81 (d, J=6.6 Hz, 1H), 7.39 (dd, J=19.1, 7.6 Hz, 4H), 7.29 (t, J=7.6 Hz, 2H), 7.23 (dd, J=13.4, 7.6 Hz, 2H), 7.16 (d, J=8.3 Hz, 1H), 6.95 (d, J=12.6 Hz, 1H), 4.88 (s, 1H), 4.18 (d, J=5.7 Hz, 2H), 3.30-3.29 (m, 1H), 3.29-3.23 (m, 3H), 3.15 (s, 2H), 2.93 (s, 2H), 2.10 (s, 1H), 0.91 (t, J=8.9 Hz, 2H), 0.69 (s, 2H).
Example 151
##STR00484##
[0846] (R)-4-((1 -((2-Chlorophenyl)(phenyl)methyl)azetidin-3-yl)methoxy)-5-cyclopropyl-2-f- luoro-N-(methylsulfonyl)benzamide
[0847] The compound was synthesized as described in Example 81. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=5.23 min). LCMS(ESI) Method A: RT=6.11 min, m/z: 543.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.89 (s, 1H), 7.81 (d, J=6.6 Hz, 1H), 7.39 (dd, J=19.1, 7.6 Hz, 4H), 7.29 (t, J=7.6 Hz, 2H), 7.23 (dd, J=13.4, 7.6 Hz, 2H), 7.16 (d, J=8.3 Hz, 1H), 6.95 (d, J=12.6 Hz, 1H), 4.88 (s, 1H), 4.18 (d, J=5.7 Hz, 2H), 3.30-3.29 (m, 1H), 3.29-3.23 (m, 3H), 3.15 (s, 2H), 2.93 (s, 2H), 2.10 (s, 1H), 0.91 (t, J=8.9 Hz, 2H), 0.69 (s, 2H).
Example 152
##STR00485##
[0848] (S)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin- -4-yl)methyl)azetidin-3-yl)methoxy)benzamide
[0849] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=4.20 min). LCMS(ESI) Method A: RT=4.72 min, m/z: 510.3 [M+H].sup.+. (500 MHz, MeOD-d.sub.4) .delta. 8.46 (d, J=5.1 Hz, 2H), 7.54 (d, J=5.9 Hz, 2H), 7.44 (d, 7.3 Hz, 2H), 7.33 (t, J=7.5 Hz, 3H), 7.26 (t, J=7.3 Hz, 1H), 6.83 (d, J=12.8 Hz, 1H), 4.59 (s, 1H), 4.23 (d, J=1H), 4.23 (d, J=6.0 Hz, 2H), 3.49 (t, J=7.7 Hz, 1H), 3.39 (t, J=7.6 Hz, 1H), 3.30 (s, 3H), 3.26-3.22 (m, 1H), 3.17 (t, J=6.9 Hz, 1H), 3.06-3.00 (m, 1H), 2.13-2.05 (m, 1H), 0.97-0.92 (m, 2H), 0.68 (d, J=4.3 Hz, 2H).
Example 153
##STR00486##
[0850] (R)-5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(phenyl(pyridin- -4-yl)methyl)azetidin-3-yl)methoxy)benzamide
[0851] The compound was synthesized as described in Example 81. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=7.60 min). LCMS(ESI) Method B: RT=4.70 min, m/z: 510.3 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 8.34 (t, J=4.6 Hz, 2H), 7.41 (d, J=5.8 Hz, 2H), 7.32 (d, J=7.3 Hz, 2H), 7.21 (t, J=7.5 Hz, 3H), 7.13 (t, J=7.3 Hz, 1H), 6.70 (d, J=12.8 Hz, 1H), 4.47 (s, 1H), 4.11 (d, J=6.1 Hz, 2H), 3.37 (t, J=7.7 Hz, 1H), 3.27 (t, J=7.7 Hz, 1H), 3.17 (s, 3H), 3.14-3.10 (m, 2H), 3.07-3.03 (m, 1H), 2.02-1.92 (m, 1H), 0.86-0.78 (m, 2H), 0.55 (d, J=4.3 Hz, 2H).
Example 154
##STR00487##
[0852] 5-Cyclopropyl-2-fluoro-4-((1-((3-fluorophenyl)(phenyl)methyl)azetid- in-3-yl)methoxy)-N-(methylsulfonyl)benzamide
[0853] The compound was synthesized as described in Example 80. LCMS(ESI) Method B: RT=b 5.84 min, m/z: 527.2 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.43 (d, J=7.3 Hz, 2H), 7.34 (dd, J=7.9, 5.0 Hz, 4H), 7.26 (dd, J=7.4, 4.4 Hz, 2H), 7.19 (d, J=9.9 Hz, 1H), 6.98 (t, J=8.5 Hz, 1H), 6.83 (d, J=12.9 Hz, 1H), 4.66 (s, 1H), 4.22 (d, J=5.9 Hz, 2H), 3.56-3.45 (m, 2H), 3.32-3.32 (m, 2H), 3.30 (s, 3H), 3.06 (s, 1H), 2.11 (t, J=Hz, 1H), 0.98-0.91 (m, 2H), 0.69 (q, J=5.9 Hz, 2H).
Example 155
##STR00488##
[0854] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)propyl)piperidin-4-yl)me- thoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0855] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.40 min, m/z: 556.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.52 (s, 1H), 7.34 (s, 2H), 7.16 (d, J=8.4 Hz, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.87 (d, J=5.9 Hz, 2H), 3.51 (s, 1H), 3.10 (s, 3H), 3.03 (s, 1H), 2.91 (s, 1H), 1.99 (s, 2H), 1.88 (s, 2H), 1.76 (d, J=14.0 Hz, 4H), 1.35 (m, 2H), 0.89-0.81 (m, 2H), 0.72 (t, J=7.2 Hz, 3H), 0.60 (d, J=4.1 Hz, 2H).
##STR00489##
Example 156
5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-(trifluoromethoxy)-phen- yl)ethyl)piperidin-4-yl)methoxy)benzamide
[0856] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.68 min, m/z: 558.9 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.63 (s, 2H), 7.37 (m, 3H), 6.58 (s, 1H), 4.33 (s, 1H), 3.79 (m, 3H), 3.16 (s, 3H), 2.76 (s, 2H), 2.00 (m, 4H), 1.85-1.47 (m, 6H), 0.86 (s, 2H), 0.63 (s, 2H).
##STR00490##
Example 157
5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(1-(4-(trifluoromethoxy)-p- henyl)ethyl)azetidin-3-yl)methoxy)benzamide
[0857] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=5.24 min, m/z: 516.9 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.50 (d, J=8.6 Hz, 2H), 7.28 (m, 3H), 6.49 (d, J=12.3 Hz, 1H), 4.92 (m, 2H), 4.05 (m, 1H), 3.77 (m, 2H), 3.44-3.35 (m, 2H), 3.26 (m, 1H), 3.22 (s, 3H), 2.12 (m, 1H), 1.35 (d, J=6.5 Hz, 3H), 0.98-0.88 (m, 2H), 0.68 (m, 2H).
Example 158
##STR00491##
[0858] 5-Cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)propyl)azetidin-3-yl)met- hoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0859] The compound was synthesized as described in Example 80. LCMS(ESI) Method A: RT=5.74 min, m/z: 529.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.50-7.41 (m, 1H), 7.40-7.30 (m, 3H), 6.83-6.69 (m, 1H), 4.18 (s, 2H), 3.76 (m, 1H), 3.47 (m, 4H), 3.24 (s, 3H), 3.09 (m, 1H), 2.08 (m, 1H), 1.88 (m, 1H), 1.56 (m, 1H), 0.93 (m, 2H), 0.76 (m, 3H), 0.68 (s, 2H).
##STR00492##
Example 159
5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(2-(trifluoromethyl)-benzy- l)piperidin-4-yl)methoxy)benzamide
[0860] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.94 min, m/z: 529.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.88 (d, J=7.8 Hz, 1H), 7.73 (d, J=7.9 Hz, 1H), 7.66 (t, J =7.6 Hz, 1H), 7.50 (t, J=7.6 Hz, 1H), 7.35 (s, 1H), 6.72 (d, J=12.9 Hz, 1H), 3.93 (m, 4H), 3.22 (m, 3H), 3.12 (m, 2H), 2.43 (m, 2H), 2.07 (m, 1H), 2.01 (m, 1H), 1.95 (m, 2H), 1.68-1.55 (m, 2H), 0.91 (m, 2H), 0.65 (m, 2H).
Example 160
##STR00493##
[0861] 5-Cyclopropyl-4-((1-(3,4-difluorobenzyl)piperidin-4-yl)methoxy)-2- fluoro-N-(methylsulfonyl)benzamide
[0862] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.46 min, m/z: 497.2 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.43 (m, 2H), 7.25-7.13 (m, 2H), 6.83 (d, J=12.8 Hz, 1H), 3.93 (d, J=5.9 Hz, 2H), 3.70 (s, 2H), 3.11 (s, 3H), 2.99 (m, 2H), 2.28 (m, 2H), 2.05-1.90 (m, 1H), 1.83 (m, 3H), 1.42 (m, 2H), 0.87 (m, 2H), 0.61 (m, 2H).
##STR00494##
Example 161
5-Cyclopropyl-2-fluoro-4-((1-(4-fluorobenzyl)piperidin-4-yl)methoxy)- N-(methylsulfonyl)benzamide
[0863] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=4.90 min, m/z: 479.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.46-7.39 (m, 2H), 7.20 (m, 3H), 6.80 (d, J=12.8 Hz, 1H), 3.92 (d, J=6.0 Hz, 2H), 3.80 (m, 2H), 3.04 (m, 5H), 2.40 (m, 2H), 2.00 (m, 1H), 1.86 (m, 3H), 1.45 (m, 2H), 0.87 (m, 2H), 0.59 (m, 2H).
##STR00495##
Example 162
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00496##
[0864] Step 1
##STR00497##
[0865] 2-(3,5-Dichlorophenyl)oxirane
[0866] The compound was synthesized as described in step 1 of Example 90.
Step 2
##STR00498##
[0867] 1-(3,5-Dichlorophenyl)-2-methoxyethanol
[0868] The compound was synthesized as described in step 2 of Example 90.
Step 3
##STR00499##
[0869] 1,3-Dichloro-5-(1-chloro-2-methoxyethyl)benzene
[0870] The compound was synthesized as described in step 2 of Example 80.
Step 4
##STR00500##
[0871] (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-4-yl- )methoxy)-2-fluorobenzoate
[0872] The compound was synthesized as described in step 5 of Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-4-yl- )methoxy)-2-fluorobenzoate. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: EtOH, A:B=85:15; flow: mL/min; column temperature: 40.degree. C.; RT=3.89 min). LCMS(ESI) m/z: 510.1 [M+H].sup.+.
##STR00501##
Step 5
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-4- -yl)methoxy)-2-fluorobenzoic acid
[0873] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 496.1 [M+H].sup.+.
##STR00502##
Step 6
(R)-5-cyclopropyl-4-((1-((1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-- 4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0874] The compound was synthesized as described in step 5 of Example 80. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 2.55 mL/min; column temperature: 39.8.degree. C.; RT=6.09 min). LCMS(ESI) Method A: RT=5.79 min, m/z: 572.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.65 (brs, 1H), 7.51-7.50 (m, 1H), 7.41-7.40 (m, 2H), 7.14 (d, J=9.0 Hz, 1H), 6.88 (d, J=13.0 Hz, 1H), 3.91 (d, J=5.5 Hz, 2H), 3.78-3.67 (m, 3H), 3.25 (s, 3H), 3.23 (s, 3H), 3.03-3.01 (m, 1H), 2.84-2.82(m, 1H), 2.16-2.12 (m, 1H), 2.06-1.97 (m, 2H), 1.80-1.73 (m, 3H), 1.41-1.31 (m, 2H), 0.89-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00503##
Example 163
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0875] The compound was synthesized as described in Example 162. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-- 4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 2.55 mL/min; column temperature: 40.6.degree. C.; RT=6.48 min). LCMS(ESI) Method A: RT=5.79 min, m/z: 572.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.63 (brs, 1H), 7.52-7.51 (m, 1H), 7.41-7.40 (m, 2H), 7.14 (d, J=8.5 Hz, 1H), 6.89 (d, J=12.5 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.79-3.68 (m, 3H), 3.25 (s, 3H), 3.23 (s, 3H), 3.03-3.02 (m, 1H), 2.85-2.82 (m, 1H), 2.17-2.13 (m, 1H), 2.05-1.97 (m, 2H), 1.80-1.73 (m, 3H), 1.41-1.31 (m, 2H), 0.89-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00504##
Example 164
(R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)-2-- methoxyethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0876] The compound was synthesized as described in Example 162. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)-2- -methoxyethyl) piperidin-4-yl)methoxy)-2-fluorobenzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=80:20; flow: 2.4 mL/min; column temperature: 38.0.degree. C.; RT=13.16 min). LCMS(ESI) Method A: RT=6.27 min, m/z: 598.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta.11.67 (brs, 1H), 7.50 (s, 1H), 7.39 (s, 2H), 7.13 (d, J=8.0 Hz, 1H), 6.89 (d, J=12.5 Hz, 1H), 3.91 (d, J=5.5 Hz, 2H), 3.74-3.67 (m, 3H), 3.23 (s, 3H), 3.06-2.98 (m, 2.82-2.80 (m, 1H), 2.12-2.08 (m, 1H), 2.03-1.97 (m, 2H), 1.79-1.72 (m, 3H), 1.39-1.23 (m, 2H), 1.08-1.03 (m, 4H), 0.89-0.86 (m, 2H), 0.65-0.64 (m, 2H).
##STR00505##
Example 165
(S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)-2-- methoxyethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0877] The compound was synthesized as described in Example 163. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)-2- -methoxyethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide, Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=80:20; flow: 2.4 mL/min; column temperature: 39.7.degree. C.; RT=11.96 min). LCMS(ESI) Method A: RT=6.19 min, m/z: 598.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.69 (brs, 1H), 7.50 (s, 1H), 7.39 (s, 2H), 7.13 (d, J=8.0 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 3.90 (d, J=6.0 Hz, 2H), 3.73-3.67 (m, 3H), 3.22 (s, 3H), 3.06-2.98 (m, 2H), 2.82-2.80 (m, 1H), 2.11-2.07 (m, 1H), 2.03-1.96 (m, 2H), 1.79-1.72 (m, 3H), 1.37-1.23 (m, 2H), 1.07-1.02 (m, 4H), 0.89-0.85 (m, 2H), 0.65-0.62 (m, 2H).
Example 166
##STR00506##
[0878] (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorop- henyl)-2-methoxyethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0879] The compound was synthesized as described in Example 162. The enantiomer was arbitrarily assigned as (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)- -2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluorobenzamide. Chiral HPLC (column: (s,s-whelk-ol, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: n-hexane, B: MeOH, B:=80:20, flow: 1 mL/min; column temperature: 40.degree. C.; RT=11.97 min). LCMS(ESI) Method A: RT=6.34 min, m/z: 614.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.52 (brs, 1H), 7.50-7.49 (m, 1H), 7.39-7.38 (m, 2H), 7.15 (d, J=8.5 Hz, 1H), 6.87 (d, J=12.5 Hz, 1H), 3.97-3.94 (m, 4H), 3.90 (d, J=6.5 Hz, 2H), 3.74-3.66 (m, 3H), 3.22 (s, 3H), 2.99-2.97 (m, 1H), 2.81-2.79 (m, 1H), 2.15-2.06 (m, 3H), 2.02-1.94 (m, 2H), 1.79-1.69 (m, 3H), 1.37-1.28 (m, 2H), 0.89-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00507##
Example 167
(S)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-- 2-methoxyethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0880] The compound was synthesized as described in Example 163. The enantiomer was arbitrarily assigned as (S)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)- -2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluorobenzamide. Chiral HPLC (column: (s,s-whelk-ol, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: n-hexane, B: MeOH, B:=80:20, flow: 1 mL/min; column temperature: 40.degree. C.; RT=12.14 min). LCMS(ESI) Method A: RT=6.36 min, m/z: 614.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.49-7.48 (m, 1H), 7.38-7.37 (m, 2H), 7.18 (d, J=9.0 Hz, 1H), 6.87 (d, J=12.5 Hz, 1H), 3.88-3.85 (m, 6H), 3.72-3.65 (m, 3H), 3.21 (s, 3H), 2.98-2.96 (m, 1H), 2.80-2.78 (m, 1H), 2.09-1.92 (m, 5H), 1.79-1.71 (m, 3H), 1.37-1.28 (m, 2H), 0.89-0.86 (m, 2H), 0.62-0.59 (m, 2H).
Example 168
##STR00508##
[0881] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[0882] The compound was synthesized as described in Example 162. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-- 4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide LCMS(ESI) Method A: RT=5.99 :min, m/z: 587.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.49-7.48 (m, 1H), 7.38-7.37 (m, 2H), 7.16 (d, J=8.8 Hz, 1H), 6.77 (d, J=12.8 Hz, 1H), 3.87 (d, J=6.0 Hz, 2H), 3.73-3.65 (m, 3H), 3.21-3.19 (m, 5H), 2.98-2.95 (m, 1H), 2.80-2.77 (m, 1H), 2.06-1.94 (m, 3H), 1.79-1.70 (m, 3H), 1.36-1.26 (m, 2H), 1.15 (t, J=7.4 Hz, 3H), 0.89-0.84 (m, 2H), 0.61-0.57 (m, 2H).
Example 169
##STR00509##
[0883] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[0884] The compound was synthesized as described in Example 163. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)piperidin-- 4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. LCMS(ESI) Method A: RT=5.99 min, m/z: 587.2 [M+H].sup.+. .sup.1H NMR (400 DMSO-d.sub.6): .delta. 7.49-7.48 (m, 1H), 7.38-7.37 (m, 2H), 7.16 (d, J=8.4 Hz, 1H), 6.77 (d, J=12.8 Hz, 1H), 3.87 (d, J=6.0 Hz, 2H), 3.73-3.65 (m, 3H), 3.24-3.19 (m, 5H), 2.98-2.95 (m, 1H), 2.80-2.77 (m, 1H), 2.09-1.91 (m, 3H), 1.79-1.67 (m, 3H), 1.36-1.26 (m, 2H), 1.16 (t, J=7.4 Hz, 3H), 0.89-0.84 (m, 2H), 0.61-0.57 (m, 2H).
##STR00510##
Example 170
(R)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluoro-5-(trifluoromethyl)phenyl)et- hyl)-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0885] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluoro-5-(trifluoromethyl)phenyl)e- thyl)-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: IC-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=4.46 min). LCMS(ESI) Method A: RT=6.13 min, m/z: 575.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.45 (brs, 1H), 7.58-7.54 (m, 3H), 7.20 (d, J=8.4 Hz, 1H), 6.87 (d, J=13.2 Hz, 1H), 3.80 (m, 3H), 3.18 (s, 3H), 2.68-2.66 (m, 1H), 2.56-2.54 (m, 2H), 2.39-2.33 (m, 1H), 2.04-1.97 (m, 1H), 1.71-1.66 (m, 2H), 1.50-1.44 (m, 2H), 1.37 (d, J=6.8 Hz, 3H), 1.04 (s, 3H), 0.89-0.85 (m, 2H), 0.65-0.61 (m, 2H).
Example 171
##STR00511##
[0886] (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluoro-5-(trifluoromethyl)ph- enyl)ethyl)-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0887] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluoro-5-(trifluoromethyl)phenyl)e- thyl)-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: IC-H, 4.6.times.250 mm, mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 2.25 mL/min; column temperature: 39.9.degree. C.; RT=4.96 min). LCMS(ESI) Method A: RT=6.11 min, m/z: 575.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.58-7.54 (m, 3H), 7.19 (d, J=8.4 Hz, 1H), 6.87 (d, J=12.8 Hz, 1H), 3.80 (m, 3H), 3.17 (s, 3H), 2.71-2.67 (m, 1H), 2.56-2.54 (m, 2H), 2.38-2.32 (m, 1H), 2.04-1.97 (m, 1H), 1.71-1.66 (m, 2H), 1.49-1.44 (m, 2H), 1.37 (d, J=6.8 Hz, 3H), 1.04 (s, 3H), 0.89-0.85 (m, 2H), 0.64-0.61 (m, 2H).
##STR00512##
Example 172
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-4-methylpi- peridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00513##
[0888] Step 1
##STR00514##
[0889] 2-(4-((2-Cyclopropyl-5-fluoro-4-(methoxycarbonyl)phenxoy)methyl)-4-- methylpiperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid
[0890] A mixture of methyl 5-cyclopropyl-2-fluoro-4-((4-methylpiperidin-4-yl)methoxy)benzoate (1.8 g, 5.6 mmol), 3,5-dichlorophenylboronic acid (1.6 g, 8.4 mmol), 2-oxoacetic acid (638 mg, 8.4 mmol) and 1.8 g of 4 A molecular sieve in toluene (30 mL) was stirred at 100.degree. C. for 2 h. The mixture was then filtered, washed with DCM (20 mL) and concentrated. The residue was purified by silica gel chromatography (eluting with DCM/MeOH from 100/1 to 10/1) to afford the target compound (2.8 g, 96%) as a white solid. LCMS(ESI) m/z: 524.0 [M+H].sup.+.
##STR00515##
Step 2
(R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)-4-m- ethylpiperidin-4-yl)methoxy)-2-fluorobenzoate
[0891] A mixture of 2-(4-((2-cyclopropyl-5-fluoro-4-(methoxycarbonyl)phenoxy)methyl)-4-methyl- piperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid (2.0 g, 8 mmol) in borane-THF (20 mL) was stirred at room temperature for 2 h, quenched with MeOH (20 mL) and concentrated. The residue was purified by silica gel chromatography (eluting with DCM/MeOH from 300/1 to 100/1) to afford the racemate (2.8 g, 96%) as an oil. The enantiomer was separated by chiral SFC from the racemate. The enantiomer was arbitrarily assigned as (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)-4-methylpiper- idin-4-yl)methoxy)-2-fluorobenzoate. Chiral HPLC (column: OZ-H 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: n-Hexane, B: EtOH, A: B=85:15; flow: 1 mL/min: column temperature: 40.degree. C.; RT=6.83 min). LCMS(ESI) m/z: 510.1 [M+H].sup.+.
Step 3
##STR00516##
[0892] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-4-m- ethylpiperidin-4-yl)methoxy)-2-fluorobenzoic acid
[0893] To a solution of (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)-4-methylpiper- idin-4-yl)methoxy)-2-fluorobenzoate (100 mg, 0.2 mmol) in THF (5 mL) was added sodium hydride (39 mg, 0.98 mmol) at 0.degree. C. The mixture was stirred at room temperature for 30 min, then methyl iodide (56 mg, 0.39 mmol) was added. The mixture was stirred for another 16 h, quenched with water (15 mL), acidified with HCl (1 M) to pH 2-3, and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (20 mL) and dried over anhydrous sodium sulfate. The solvent was distilled off under reduce pressure to afford a yellow solid. LCMS(ESI) m/z: 510.1 [M+H].sup.+.
##STR00517##
Step 4
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-4-methylpi- peridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0894] The compound was synthesized as described in step 5 of Example 80. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=5.13 min). LCMS(ESI) Method A: RT=6.28 min, m/z: 587.2 [M+H].sup.+. .sup.1H NMR (400 Hz, DMSO-d.sub.6): .delta. 11.69 (brs, 1H), 7.50 (s, 1H), 7.41 (s, 2H), 7.18 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.8 Hz, 1H), 3.79-3.69 (m, 5H), 3.23 (s, 3H), 3.22 (s, 3H), 2.67-2.66 (m, 1H), 2.58-2.50 (m, 1H), 2.50-2.35 (m, 2H), 2.03-1.97 (m, 1H), 1.68-1.62 (m, 2H), 1.49-1.37 (m, 2H), 1.01 (s, 3H), 0.90-0.86 (m, 2H), 0.65-0.62 (m, 2H).
Example 173
##STR00518##
[0895] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-4-m- ethylpiperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0896] The compound was synthesized as described in Example 172. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxyethyl)-4-methylp- iperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=65:35; flow: 1.95 mL/min; column temperature: 40.1.degree. C.; RT=4.52 min). LCMS(ESI) Method A: RT=6.28 min, m/z: 587.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.64 (brs, 1H), 7.50 (s, 1H), 7.41 (s, 2H), 7.18 (d, J=8.4 Hz, 1H), 6.89 (d, J=12.8 Hz, 1H), 3.78-3.66 (m, 5H), 3.22 (s, 6H), 2.67-2.66 (m, 1H), 2.58-2.50 (m, 1H), 2.45-2.33 (m, 2H), 2.03-1.97 (m, 1H), 1.68-1.62 (m, 2H), 1.49-1.37(m, 2H), 1.01 (s, 3H), 0.90-0.86 (m, 2H), 0.65-0.62 (m, 2H).
##STR00519##
Example 174
5-Cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)piperidin-4-yl)meth- oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00520##
[0897] Step 1
##STR00521##
[0898] 3-(3,5-Dichlorophenyl)oxetan-3-ol
[0899] To a solution of 1-bromo-3,5-dichlorobenzene (1.0 g, 4.5 mmol) in dry THF (20 mL) was added n-BuLi (2.2 mL, 5.4 mmol) dropwise and the mixture stirred 30 min at -78.degree. C. Oxetan-3-one (386 mg, 5.4 mmol) was then added and the mixture was left to warm to room temperature. The mixture was then quenched with aqueous ammonium chloride (20 mL) and extracted with EtOAc (20 mL.times.3). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate and concentrated. The residue was purified by silica gel chromatography (eluting with ethyl acetate/petroleum ether=1/10) to afford the target compound (0.7 g, 71%) as an oil. LCMS(ESI) m/z: 217.1 [M-H].sup.-.
##STR00522##
Step 2
Methyl 5-cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)piperidin-4-- yl)methoxy)-2fluorobenzoate
[0900] To solution of 3-(3,5-dichlorophenyl)oxetan-3-ol (400 mg, 1.8 mmol) and DIPEA (1.4 g, mmol) in DCM (10 mL), trifluoromethanesulfonic anhydride (0.9 mL, 5.5 mmol) was added dropwise at -20.degree. C. The mixture was then stirred at room temperature for 3 h, and methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (280 mg, 0.92 mmol) in acetonitrile (10 mL) was added. Then the reaction mixture was stirred at room temperature for another 16 h. The mixture was then concentrated in vacuo and the residue purified by silica gel chromatography (eluting with ethyl acetate/petroleum ether=1/3) to afford the target compound (320 mg, 69%) as an oil. LCMS(ESI) m/z: 508.1 [M+H].sup.+.
##STR00523##
Step 3
5-Cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)piperidin-4-yl)meth- oxy)-2-fluorobenzoic acid
[0901] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 494.1 [M+H].sup.+.
Step 4
##STR00524##
[0902] 5-Cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)piperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0903] The compound was synthesized as described step 5 of Example 80. LCMS(ESI) Method A: RT=5.76 min, m/z: 571.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.82 (brs, 1H), 7.47-7.44 (m, 2H), 7.39-7.37 (m, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.88 (d, J=13.2 Hz, 1H), 3.90 (d, J=6.0 Hz, 2H), 3.24 (m, 1H), 3.23 (s, 3H), 3.02-2.94 (m, 2H), 2.87-2.79 (m, 3H), 2.15-2.08 (m, 2H), 2.02-1.98 (m, 1H), 1.75-1.69 (m, 3H), 1.33-1.30 (m, 2H), 0.90-0.85 (m, 2H), 0.66-0.62 (m, 2H).
##STR00525##
Example 175
5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3-(3,5-dichlorophenyl)oxetan-- 3-yl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0904] The compound was synthesized as described in Example 174. LCMS(ESI) Method A: RT=5.91 min, m/z: 597.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.79 (brs, 1H), 7.47-7.44 (m, 2H), 7.39-7.35 (m, 1H), 7.15 (d, J=8.4 1Hz, 1H), 6.85 (d, J=12.8 Hz, 1H), 3.89 (d, J=5.6 Hz, 2H), 3.23-3.22 (m, 3H), 3.01-2.93 (m, 3H), 2.87-2.79 (m, 3H), 2.12-2.07 (m, 2H), 2.01-1.97 (m, 1H), 1.72-1.69 (m, 3H), 1.33-1.30 (m, 2H), 1.03-0.97 (m, 4H), 0.90-0.85 (m, 2H), 0.65-0.61 (m, 2H).
Example 176
##STR00526##
[0905] N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(3-(3,5-dichloropheny- l)oxetan-3-yl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[0906] The compound was synthesized as described in Example 174. LCMS(ESI) Method A: RT=6.06 min, m/z: 612.5 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-d.sub.4): .delta.7.41-7.38 (m, 2H), 7.34-7.30 (m, 2H), 6.75 (d, J=13.2 Hz, 1H), 4.12-3.91 (m, 4H), 3.91 (d, J=6.0 Hz, 2H), 3.31 (m, 1H), 3.16-3.07 (m, 2H), 2.93-2.91 (m, 3H), 2.28-2.20 (m, 4H), 2.09-2.05 (m, 1H), 1.86-1.80 (m, 3H), 1.52-1.49 (m, 2H), 0.95-0.90 (m, 2H), 0.68-0.64 (m, 2H).
Example 177
##STR00527##
[0907] 5-Cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)piperidin-4-- yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[0908] The compound was synthesized as described in Example 174. LCMS(ESI) Method A: RT=6.86 min, m/z: 585.2 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-d.sub.4): .delta.7.42-7.39 (m, 2H), 7.34-7.28 (m, 2H), 6.87 (d, J=13.2 Hz, 1H), 3.92 (d, J=6.0 Hz, 2H), 3.51-3.45 (m, 2H), 3.35 (s, 1H), 3.21-3.13 (m, 2H), 2.98-2.92 (m, 3H), 2.34-2.67 (m, 2H), 2.09-2.05 (m, 1H), 1.90-1.82 (m, 3H), 1.57-1.51 (m, 2H), 1.37 (d, J=7.4 Hz, 3H), 0.95-0.90 (m, 2H), 0.68-0.64 (m, 2H).
##STR00528##
Example 178
5-Cyclopropyl-4-((1-(3-(3,5-dichlorophenyl)oxetan-3-yl)-4-methylpiperidin-- 4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0909] The compound was synthesized in a similar manner to Example 174 from methyl 5-cyclopropyl-2-fluoro-4-((4-methylpiperidin-4-yl)methoxy)benzoate and 3-(3,5-Dichlorophenyl)oxetan-3-ol. LCMS(ESI) Method A: RT=5.91 min, m/z: 585.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.47-7.44 (m, 2H), 7.39-7.35 (m, 1H), 7.23-7.19 (m, 1H), 6.84 (d, J=10.8 Hz, 1H), 3.76 (s, 2H), 3.23-3.22 (m, 1H), 3.12 (s, 3H), 2.87-2.76 (m, 3H), 2.69-2.61 (m, 2H), 2.45-2.39 (m, 2H), 2.03-1.95 (m, 1H), 1.66-1.61 (m, 2H), 1.38-1.33 (m, 2H), 1.03 (s, 3H), 0.88-0.85 (m, 2H), 0.61-0.60 (m, 2H).
##STR00529##
Example 179
(R)-5-cyclopropyl-4-(1-(2-(diisopropylamino)-2-oxoethyl)piperidin-3-yloxy)- -2-fluoro-N-(methylsulfonyl)benzamide
##STR00530##
[0910] Step 1
##STR00531##
[0911] (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate
[0912] To a solution of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine-1-car- boxylate (1.0 g, 2.3 mmol) in tert-butylacetate (4 mL) was added sulfuric acid (0.6 mL, 11.5 mmol) at room temperature. The reaction mixture was stirred for 2 h, quenched with aqueous ammonium bicarbonate (10 mL) and extracted with ethyl acetate (20 mL.times.3). The combined organic layers were distilled off under reduced pressure to afford the target compound (500 mg, 65%) as a pale yellow oil. LCMS(ESI) m/z: 336.0 [M+H].sup.+.
Step 2
##STR00532##
[0913] 2-Bromo-N,N-diisopropylacetamide
[0914] Diisopropylamine (1.0 g, 5.0 mmol) was added to a solution of 2-bromoacetyl bromide (1.0 g, 10.0 mmol) in DCM (30 mL) at 0.degree. C. The reaction mixture was left to warm to room temperature and stirred for 1 h, then quenched with saturated ammonium chloride, extracted with DCM (2.0 mL.times.3), washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=10/1) to afford the target compound (810 mg, 81%) as a pale yellow oil. LCMS(ESI) m/z: 222.0 [M+H].sup.+. .sup.1H NMR (500 MHz, CDCl.sub.3-d) .delta.3.98-3.95 (m, 1H), 3.81 (s, 2H), 3.45-3.42 (m, 1H), 1.40-1.88 (m, 6H), 1.28-1.25 (m, 6H).
Step 3
##STR00533##
[0915] (R)-tert-butyl 5-cyclopropyl-4-(1-(2-(diisopropylamino)--oxoethyl)piperidin-3-yloxy)-2-f- luorobenzoate
[0916] A mixture of (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (56 mg, 0.17 mmol), 2-bromo-N,N-diisopropylacetamide (44 mg, 0.20 mmol) and potassium carbonate (69 mg, 0.50 mmol) in acetonitrile (2 mL) was heated at 80.degree. C. for 16 h. The reaction mixture was filtered, washed with ethyl acetate (10 mL) and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=5/1) to afford the target compound (61 mg, 75%) as a pale yellow oil. LCMS(ESI) m/z: 477.0 [M+H].sup.+.
Step 4
##STR00534##
[0917] (R)-5-cyclopropyl-4-(1-(2-(diiospropylamino)-2-oxoethyl)piperidin-3- -yloxy)-2-fluorobenzoic acid
[0918] The compound was synthesized as described in step 4, Example 80. LCMS(ESI) m/z: 421.1 [M+H].sup.+.
Step 5
##STR00535##
[0919] (R)-5-cyclopropyl-4-(1-(2-(diisopropylamino)-2-oxoethyl)piperidin-3- -yloxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0920] The compound was synthesized as described in step 5, Example 80. LCMS(ESI) Method A: RT=4.61 min, m/z: 498.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.14 (d, J=8.5 Hz, 1H), 6.92 (d, J=13.0 Hz, 1H), 4.58 (m, 1H), 4.09 (m, 1H), 3.41-3.38 (m, 3H), 3.15 (s, 3H), 2.97 (m, 1H), 2.70-2.63 (m, 2H), 2.49-2.46 (m, 1H), 2.07 (m, 1H), 1.95 (m, 1H), 1.83 (m, 1H), 1.64-1.57 (m, 2H), 1.28-1.24 (m, 6H), 1.14-1.10 (m, 6H), 0.89-0.85 (m, 2H), 0.68-0.61 (m, 2H).
##STR00536##
Example 180
5-Cyclopropyl-4-((1-(2-(diisopropylamino)-2-oxoethyl)piperidin-4-yl)methox- y)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00537##
[0921] Step 1
##STR00538##
[0922] Methyl 5-cyclopropyl-4-((1-(2-(diisopropylamino)-2-oxoethyl)piperidin-4-yl)metho- xy)-2-fluorobenzoate
[0923] The compound was synthesized as described in step 3 of Example 179. LCMS(ESI) m/z: 449.1 [M+H].sup.+.
Step 2
##STR00539##
[0924] 5-Cyclopropyl-4-((1-(2-(diisopropylamino)-2-oxoethyl)piperidin-4-yl- )methoxy)-2-fluorobenzoic acid
[0925] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 435.1 [M+H].sup.+.
Step 3
##STR00540##
[0926] 5-Cyclopropyl-4-((1-(2-(diiospropylamino)-2-oxoethyl)piperidin-4-yl- )methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0927] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=4.52 min, m/z: 512.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.21 (d, J=8.5 Hz, 1H), 6.77 (d, J=12.0 Hz, 1H), 3.93-3.77 (m, 5H), 3.51-3.49 (m, 1H), 3.23 (m, 2H), 2.96 (s, 3H), 2.75-2.57 (m, 2H), 2.04-1.88 (m, 4H), 1.69-1.56 1 (m, 2H), 1.33-1.32 (m, 6H), 1.16-1.15 (m, 6H), 0.89-0.85 (m, 2H), 0.60-0.57 (m, 2H).
##STR00541##
Example 181
(R)-4-((1-(1-(3-chloro-4-fluorophenyl)-2-methoxyethyl)piperidin-4-yl)metho- xy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0928] The compound was synthesized as described in Example 172. The enantiomer was arbitrarily assigned as (R)-4-((1-(1-(3-chloro-4-fluorophenyl)-2-methoxyethyl)piperidin-4-yl)meth- oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 2.25 mL/min; column temperature: 41.7.degree. C.; RT=2.97 min). LCMS(ESI) Method A: RT=5.51 mm, m/z: 557.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.58 (brs, 1H), 7.55 (d, J=6.0 Hz, 1H), 7.41-7.36 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.85 (d, 12.8 Hz, 1H), 3.89 (d, J=6.0 Hz, 2H), 3.78-3.70 (m, 3H), 3.23 (s, 3H), 3.17 (s, 3H), 3.05-3.03 (m, 1H), 2.85-2.82 1H), 2.15-2.12 (m, 1H), 2.03-1.97 (m, 2H), 1.80-1.72 (m, 3H), 1.41-1.31 (m, 2H), 0.89-0.85 (m, 2H), 0.64-0.60 (m, 2H).
Example 182
##STR00542##
[0929] (S)-4-((1-(1-(3-chloro-4-fluorophenyl)-2-methoxyethyl)piperidin-4-y- l)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0930] The compound was synthesized as described in Example 172. The enantiomer was arbitrarily assigned as (S)-4-((1-(1-(3-chloro-4-fluorophenyl)-2-methoxyethyl)piperidin-4-yl)meth- oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 2.25 mL/min; column temperature: 39.5.degree. C.; RT=3.58 min). LCMS(ESI) Method A: RT=5.50 min, m/z: 557.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 11.60 (brs, 1H), 7.55 (d, J=6.8 Hz, 1H), 7.41-7.36 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.85 (d, J=12.8 Hz, 1H), 3.89 (d, J=5.6 Hz, 2H), 3.78-3.70 (m, 3H), 3.23 (s, 3H), 3.17 (s, 3H), 3.05-3.03 (m, 1H), 2.85-2.82. (m, 1H), 2.20-2.12 (m, 1H), 2.03-1.97 (m, 2H), 1.80-1.72 (m, 3H), 1.41-1.31 (m, 2H), 0.89-0.85 (m, 2H), 0.64-0.60 (m, 2H).
Example 183
##STR00543##
[0931] 4-((1-(3-Chloro-4-methoxybenzyl)piperidin-4-yl)methoxy)-5-cycloprop- yl-2-fluoro-N-(methylsulfonyl)benzamide
[0932] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.08 min, m/z: 525.2 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-d.sub.4) .delta.10.51 (brs, 1H), 7.48 (s, 1H), 7.35-7.33 (m, 1H), 7.20-7.16 (m, 2H), 6.89 (d, J=12.8 Hz, 1H), 3.92 (d, J=5.6 Hz, 2H), 3.86 (s, 3H), 3.82 (s, 2H), 3.11-3.08 (m, 2H), 3.01 (s, 3H), 2.50-2.39 (m, 2H), 2.03-1.97 (m, 1H), 1.91-1.85 (m, 3H), 1.48-1.45 (m, 2H), 0.89-0.85 (m, 2H), 0.61-0.57 (m, 2H).
##STR00544##
Example 184
5-Cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-4-yl)methoxy)-N-(ethylsu- lfonyl)-2-fluorobenzamide
[0933] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.34 min, m/z: 543.2 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-d.sub.4) .delta.11.47 (brs, 1H), 7.54-7.52 (m, 1H), 7.40-7.39 (m, 2H), 7.15-7.13(m, 1H), 6.90-6.87 (m, 1H), 3.93 (d, J=6.0 Hz, 2H), 3.62 (m, 2H), 3.35-3.32 (m, 2H), 2.93-2.90 (m, 2H), 2.19-1.99 (m, 3H), 1.82-1.79 (m, 3H), 1.44-1.38 (m, 2H), 1.22-1.18 (m, 3H), 0.90-0.85 (m, 2H), 0.61-0.57 (m, 2H).
Example 185
##STR00545##
[0934] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazeti- din-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0935] The compound was synthesized as described in Example 81. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazetidin-3-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.5% DEA), A:B=70:30; flow: 3 mL/min; column temperature: 38.5.degree. C.; RT=5.7 min). LCMS(ESI) Method C: RT =5.48 min, m/z: 528.8 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-.sub.4) .delta.7.44 (s, 1H), 7.39-7.35 (m, 3H), 6.81 (d, J =12.4 Hz, 1H), 4.10-4.04 (m, 2), 3.83-3.81 (m, 1H), 3.65-3.59 (m, 2H), 3.40-3.38 (m, 1H), 3.27-3.25 (m, 4H), 2.10-2.07 (m, 1H), 1.48 (s, 3H), 1.37-1.35 (m, 3H), 0.95-0.93 (m, 2H), 0.69-0.68 (m, 2H).
Example 186
##STR00546##
[0936] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazeti- din-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[0937] The compound was synthesized as described in Example 81. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazetidin-3-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.5% DEA), A:B=70:30; flow: 3 mL/min; column temperature: 38.5.degree. C.; RT=8.13 min). LCMS(ESI) Method C: RT =5.42 min, m/z: 528.7 [M+H].sup.+. .sup.1H NMR (400 MHz, MeOD-d.sub.4).delta. 7.42 (s, 1H), 7.38-7.36 (m, 3H), 6.81 (d, J =12.8 Hz, 1H), 4.10-4.03 (m, 2H), 3.79-3.77 (m, 1H), 3.60-3.50 (m, 2H), 3.34-3.32 (m, 1H), 3.25-3.23 (m, 4H), 2.09-2.01(m, 1H), 1.47 (s, 3H), 1.35-1.30 (m, 3H), 0.97-0.91 (m, 2H), 0.68-0.66 (m, 2H).
Example 187
##STR00547##
[0938] (R)-5-cyclopropyl-2-fluoro-4-((1-(1-(4-fluoro-3-(trifluoromethyl)ph- enyl)-2-methoxyethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0939] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-2-fluoro-4-((1-(1-(4-fluoro-3-(trifluoromethyl)phenyl)-- 2-methoxyethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 3 mL/min; column temperature: 39.5.degree. C.; RT =3.55 min). LCMS(ESI) Method C: RT=4.82 min, m/z: 591.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.72-7.70) (m, 2H), 7.51-7.46 (m, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.87 (d, J=12.8 Hz, 1H), 3.91-3.84 (m, 5H), 3.28-3.27 (m, 1H), 3.22-3.21 (m, 5H), 3.06-3.03 (m, 1H), 2.83-2.81 (m, 1H), 2.16-1.96 (m, 3H), 1.77-1.72 (m, 3H), 1.40-1.30 (m, 2H), 0.89-0.84 (m, 2H), 0.65-0.61 (m, 2H).
Example 188
##STR00548##
[0940] (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(4-fluoro-3-(trifluoromethyl)ph- enyl)-2-methoxyethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0941] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(4-fluoro-3-(trifluoromethyl)phenyl)-- 2-methoxyethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 min, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 3 mL/min; column temperature: 39.5.degree. C.; RT =6.51 min). LCMS(ESI) Method C: RT=4.83 min, m/z 591.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.72-7.70 (m, 2H), 7.51-7.46 (m, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.87 (d, J=13.2 Hz, 1H), 3.91-3.72 (m, 4H), 3.29-3.28 (m, 1H), 3.23-3.21 (m, 5H), 3.05-3.02 (m, 1H), 2.83-2.81 (m, 2H), 2.02-1.96 (m, 3H), 1.83-1.72 (m, 3H), 1.40-1.30 (m, 2H), 0.89-0.84 (m, 2H), 0.66-0.62 (m, 2H).
##STR00549##
Example 189
5-Cyclopropyl-2-fluoro-4-((1-(4-fluoro-3-(trifluoromethyl)piperidin)piperi- din-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0942] The compound was synthesized as described in Example 88. LCMS(ESI) Method C: RT=3.95 min, m/z: 547.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.33-7.72 (m, 2H), 7.52-7.47 (m, 1H), 7.18 (d, J=8.8 Hz, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.92 (d, J=5.6 Hz, 2H), 3.68 (s, 2H), 3.04 (s, 3H), 2.94-2.91 (m, 2H), 2.03-1.99 (m, 3H), 1.83-1.80 (m, 3H), 1.44-1.38 (m, 2H), 0.89-0.86 (m, 2H), 0.60-0.59 (m, 2H).
##STR00550##
Example 190
4-((1-((5-Chloro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)methyl)piperidin-4-- yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00551##
##STR00552##
[0943] Step 1
5-Chloro-6-(2,2,2-trifluoroethoxy)nicotinic acid
[0944] A mixture of 2,2,2-trifluoroethanol (1.0 g, 5.2 mmol) and potassium hydroxide (874 mg, 15.6 mmol) in DMSO (15 mL) was stirred at 120.degree. C. for 24 h. The reaction mixture was acidified with HCl (1M), extracted with DCM (30 mL.times.3), dried over anhydrous sodium sulfate and concentrated. The crude compound (960 mg) was used in next step without further purification. LCMS(ESI) m/z: 255.8 [M+H].sup.+.
Step 2
##STR00553##
[0945] (5-Chloro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)methanol
[0946] The compound was synthesized as described to step 2 of Example 172. LCMS(ESI) m/z: 241.9 [M+H].sup.+.
##STR00554##
Step 3
3-Chloro-5-(chloromethyl)-2-(2,2,2-trifluoroethoxy)pyridine
[0947] The compound was synthesized as described in step 2 of Example 80.
Step 4
##STR00555##
[0948] Methyl 4-((1-((5-chloro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)methyl)piperidin-4- -yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
[0949] The compound was synthesized as described in step 3 of Example 80. LCMS(ESI) m/z: 531.0 [M+H].sup.+.
##STR00556##
Step 5
4-((1-((5-Chloro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)methyl)piperidin-4-- yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
[0950] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 517.0 [M+H].sup.+.
##STR00557##
Step 6
4-((1-((5-Chloro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)methyl)piperidin-4-- yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[0951] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI) Method A: RT=5.70 min, m/z 594.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 8.14-8.11 (m, 1H), 7.97-7.94 (m, 1H), 7.35-7.34 (m, 1H), 6.70-6.66 (m, 1H), 5.0-4.94 (m, 2H), 3.90-3.71 (m, 4H), 3.30-3.15 (m, 5H), 2.50-2.49 (m, 2H), 2.07-1.93 (m, 4H), 1.61-1.55 (m, 2H), 0.90-0.89 (m, 2H), 0.65-0.63 (m, 2H).
Example 191
##STR00558##
[0952] 4-((1-(2-Chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluor- o-N-(methylsulfonyl)benzamide
[0953] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.67 min, m/z: 495.0 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4): .delta. 7.58-7.57 (m, 1H), 7.47-7.46 (m, 1H), 7.36-7.33 (m, 3H), 6.68 (d, J=12.0 Hz, 1H), 3.94-3.90 (m, 4H), 3.27-3.12 (m, 5H), 2.54-2.47 (m, 1H), 2.08-1.95 (m, 4H), 1.63-1.60 (m, 2H), 1.28-1.25 (m, 1H), 0.91-0.87 (m, 2H), 0.67-0.64 (m, 2H).
Example 192
##STR00559##
[0954] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(2,2,3,3-tetrafluor- opropyl)piperidin-4-yl)methoxy)benzamide
[0955] The compound was synthesized as described in Example 116. LCMS(ESI) Method A: RT=5.34 min, m/z: 485.1 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 11.87 (s, 1H), 7.15 (d, J=9.0 Hz, 1H), 6.90 (d, J=13.0 Hz, 1H), 6.58-6.37 (m, 1H), 3.92 (d, J=6.0 Hz, 2H), 3.23 (s, 3H), 3.0-2.89 (m, 4H), 2.32-2.27 (m, 2H), 2.03-2.0 (m, 1H), 1.78-1.74 (m, 3H), 1.40-1.32 (m, 2H), 0.91-0.87 (m, 2H), 0.66-0.63 (m, 2H).
Example 193
##STR00560##
[0956] 5-Cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(3,3,3-trifluo- ropropyl)piperidin-4-yl)methoxy)benzamide
[0957] The compound was synthesized as described in Example 116. LCMS(ESI) Method A: RT=4.98 min, m/z: 493.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta.7.15 (d, J=8.0 Hz, 1H), 6.88 (d, J=12.5 Hz, 1H), 3.94 (d, J=5.5 Hz, 2H), 3.04-3.00 (m, 3H), 2.65-2.63 (m, 2H), 2.56-2.50 (m, 2H), 2.17-2.12 (m, 2H), 2.04-1.99 (m, 1H), 1.82-1.79 (m, 3H), 1.42-1.35 (m, 2H), 1.06-0.97 (m, 4H), 0.90-0.87 (m, 2H), 0.65-0.62 m, 2H).
Example 194
##STR00561##
[0958] N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-(3,3,3-trifl- uoropropyl)piperidin-4-yl)methoxy)benzamide
[0959] The compound was synthesized as described in Example 116. LCMS(ESI) Method A: RT=5.13 min, m/z: 508.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.17 (d, J=8.5 Hz, 1H), 6.87 (d, J=13.0 Hz, 1H), 3.94-3.93 (m, 6H), 2.98-2.96 (m, 2H), 2.61-2.58 (m, 2-H), 2.52-2.50 (m, 1H), 2.49-2.48 (m, 1H), 2.12-2.01 (m, 5H), 1.38-1.35 (m, 3H), 0.90-0.87 (m, 2H), 0.90-0.87 (m, 2H), 0.65-0.62 (m, 2H).
Example 195
##STR00562##
[0960] 4-((1-(3-Chlorobenzoyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluo- ro-N-(methylsulfonyl)benzamide
[0961] The compound was synthesized as described in step Example 134. LCMS(ESI) Method A: RT 4.61 min, m/z: 509.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.87 (s, 1H), 7.53-7.43 (m, 3H), 7.34 (d, J=8.0 Hz, 1H), 7.15 (d, J=8.5 Hz, 1H), 6.96 (d, J=10.4 Hz, 1H), 4.51-4.49 (m, 1H), 3.99 (d, J=4.4 Hz, 2H), 3.57-3.51 (m, 1H), 3.28 (s, 3H), 3.12-3.04 (m, 1H), 2.89-2.81(m, 1H), 2.13-1.72 (m, 4H), 1.32-1.23 (m, 2H), 0.91-0.84 (m, 2H), 0.68-0.64 (m, 2H).
Example 196
##STR00563##
[0962] 4-((1-(3-Chlorobenzoyl)piperidin-4-yl)methoxy)-5-cyclopropyl-N-(cyc- lopropylsulfonyl)-2-fluorobenzamide
[0963] The compound was synthesized as described in step Example 134. LCMS(ESI) Method A: RT=4.74 min, m/z: 535.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.87 (s, 1H), 7.53-7.43 (m, 3H), 7.34 (d, J=8.0 Hz, 1H), 7.15 (d, J=8.5 Hz, 1H), 6.96 (d, J=12.5 Hz, 1H), 4.52-4.50 (m, 1H), 4.00 (d, J=5.0 Hz, 2H), 3.58-3.32 (m, 1H), 3.13-3.00 (m, 2H), 2.85-2.83 (m, 1H), 2.14-1.90 (m, 4H), 1.34-1.33 (m, 2H), 1.24-1.22 (m, 4H), 0.91-0.87 (m, 2H), 0.69-0.66 (m, 2H).
##STR00564##
Example 197
4-((1-Benzoylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfo- nyl)benzamide
[0964] The compound was synthesized as described in step Example 134. LCMS(ESI) Method A: RT=4.17 min, m/z: 475.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta.11.87 (s, 1H), 7.45-7.44 (m, 3H), 7.38-7.36 (m, 2H), 7.15 (d, J=8.5 Hz, 1H), 6.96 (d, J=13.0 Hz, 1H), 4.0-3.99 (m, 1H), 3.99 (d, J=4.4 Hz, 2H), 3.62-3.60 (m, 1H), 3.32 (s, 3H), 3.17-3.09 (m, 1H), 3.02-3.01 (m, 1H), 2.13-1.76 (m, 4H), 1.32-1.24 (m, 2H), 0.91-0.84 (m, 2H), 0.69-0.63 (m, 2H).
Example 198
##STR00565##
[0965] 4-((1-Benzoylpiperidin-4-yl)methoxy)-5-cyclopropyl-N-(cyclopropylsu- lfonyl)-2-fluorobenzamide
[0966] The compound was synthesized as described in step Example 134. LCMS(ESI) Method A: RT=4.38 min, m/z: 501.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta.11.81 (s, 1H), 7.45.sub.7-7.44 (m, 3H), 7.38-7.36 (m, 2H), 7.15 (d, J=8.0 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 4.52-4.51 (m, 1H), 3.98 (d, J=4.8 Hz, 2H), 3.62-3.60 (m, 1H), 3.09-3.01 (m, 2H), 2.83-2.81 (m, 1H), 2.14-1.71 (m, 4H), 1.32-1.31 (m, 2H), 1.07-1.03 (m, 4H), 0.90-0.84 (m, 2H), 0.66-0.61 (m, 2H).
Example 199
##STR00566##
[0967] 5-Cyclopropyl-2-fluoro-4-((1-((1R,2S)-2-hydroxy-2,3dihydro-1H-inden- -1-yl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR00567## ##STR00568##
[0968] Step 1
##STR00569##
[0969] tert-Butyl 5-chloro-4-(cyclopent-3-en-1-ylmethoxy)-2-fluorobenzoate
[0970] A mixture of cyclopent-3-enylmethanol (0.9 g, 9.2 mmol), tert-butyl 5-chloro-2,4-difluorobenzoate (2.3 g, 9.3 mmol) and cesium carbonate. (6.0 g, 18.4 mmol) in DMSO (20 mL) was stirred at 80.degree. C. for 10 h. The reaction mixture was diluted with EtOAc (50 mL) and brine (100 mL). The organic layer was washed by brine (30 mL.times.3), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by flash column (petroleum ether) to afford the title compound as a white solid (2.1 g, 70%).
Step 2
##STR00570##
[0971] tert-Butyl 5-chloro-2-fluoro-4-(4-hydroxy-2-(2-hydroxyethyl)butoxy)benzoate
[0972] Ozone was bubbled into a solution of tert-butyl 5-chloro-4-(cyclopent-3-enylmethoxy)-2-fluorobenzoate (1.63 g, 5 mmol) in dry DCM (50 mL) at -78.degree. C. until the solution turned blue. Then the reaction was purged with nitrogen gas until the reaction mixture turned colorless. Sodium borohydride (0.76 g, 20 mmol) and methanol (50 mL) was then added and the resultant mixture allowed to warm to room temperature and stirred for 5 h. The mixture was then concentrated in vacuo and the residue purified by silica gel chromatography (eluting with 50% ethyl acetate in petroleum ether) to afford the title compound as a colorless oil (1.63 g, 90%).
##STR00571##
Step 3
tert-Butyl 5-cyclopropyl-2-fluoro-4-(4-hydroxy-2-(2-hydroxyethyl)butoxy)be- nzoate
[0973] A mixture a tert-butyl 5-chloro-2-fluoro-4-(4-hydroxy-2-(2-hydroxyethyl)butoxy)benzoate (1.09 g, 3 mmol), cyclopropylboronic acid (0.515 g, 6 mmol), diacetoxypalladium (0.067 g, 0.3 mmol) and potassium phosphate (1.3 g, 6 mmol) in toluene (30 mL) and H.sub.2O (1.5 mL) was stirred at 90.degree. C. for 16 h. The reaction mixture was filtered, concentrated in vacuo and the residue purified by silica gel chromatography (eluting with 40% ethyl acetate in petroleum ether) to afford the target compound as a colorless oil (0.77 g, 70%).
##STR00572##
Step 4
tert-Butyl 4-(4-bromo-2-(2-bromoethyl)butoxy)-5-cyclopropyl-2-fluorobenzoa- te
[0974] A mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(4-hydroxy-2-(2-hydroxyethyl)butoxy)benzoate (0.73 g, 2 mmol), triphenylphosphine (2.1 g, 8 mmol), carbon tetrabromide (2.65 g, 8 mmol) in DCM (30 mL) was stirred at room temperature for 20 h. The mixture was concentrated in vacuo and the residue purified by silica gel chromatography (eluting with 10% ethyl acetate in petroleum ether) to afford the target compound as a pale yellow oil (0.84 g, 85%).
Step 5
##STR00573##
[0975] tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(1R,2S)-2-hydroxy-2,3-dihydro-1H-inden-1-yl)- piperidin-4-yl)methoxy)benzoate
[0976] A mixture of tert-butyl 4-(4-bromo-2-(2-bromoethyl)butoxy)-5-cyclopropyl-2-fluorobenzoate (0.84 g, 1.7 mmol), (1R,2S)-1-amino-2,3-dihydro-1H-inden-2-ol (0.253 g, 1.7 mmol), potassium carbonate (0.47 g, 3.4 mmol) and sodium iodide (0.020 g, 0.17 mmol) in acetonitrile (10 mL) was stirred at 80.degree. C. for 8 h. The reaction mixture was quenched with brine (40 mL), extracted with ethyl acetate (10 mL.times.3), dried over anhydrous sodium sulfate, and concentrated. The residue was purified by silica gel chromatography (eluting with 15% ethyl acetate in petroleum ether) to afford the target compound as a white solid (0.712 g, 87%). MS(ESI): m/z: 482.3 [M+1].sup.+.
Step 6
##STR00574##
[0977] 5-Cyclopropyl-2-fluoro-4-((1-((1R,2S)-2-hydroxy-2,3-dihydro-1H-inde- n-1-yl)piperidin-4-yl)methoxy)benzoic acid
[0978] The compound was synthesized as described in step 3 of Example 88. LCMS(ESI) m/z: 426.2 [M+1].sup.+.
Step 7
##STR00575##
[0979] 5-Cyclopropyl-2-fluoro-4-((1-((1R,2S)-2-hydroxy-2,3-dihydro-1H-inde- n-1-yl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0980] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI): Method A: RT=4.71 min, m/z: 503.2 [M+1].sup.+. .sup.1HNMR (500 MHz, DMSO-d.sub.6) .delta. 7.43 (d, J=7.6 Hz, 1H), 7.33-7.21 (m, 3H), 7.21 (d, J=8.4 Hz, 1H), 6.72 (d, J=2.8 Hz, 1H), 4.65-4.60 (m, 1H), 4.32 (s, 1H), 3.88 (d, J=5.6 Hz, 2H), 3.27-3.25 (m, 2H), 3.13-3.04 (m, 3H), 2.91-2.83 (m, 4H), 2.03-1.96 (m, 1H), 1.88-1.80 (m, 3H), 1.57-1.48 (m, 2H), 0.88-0.83 (m, 2H), 0.58-0.54 (m, 2H).
##STR00576##
Example 200
5-Cyclopropyl-2-fluoro-4-((1-((1R,2R)-2-hydroxy-2,3-dihydro-1H-inden-1-yl)- piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[0981] The compound was synthesized as described in Example 199. LCMS(ESI) Method A: RT=4.30 min, m/z: 503.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta.7.35-7.33 (m, 1H), 7.25-7.24 (m, 3H), 7.17 (d, J=8.4 Hz, 1H), 6.85 (d, J=12.8 Hz, 1H), 5.18-5.16 (m, 1H), 4.60-4.58 (m, 1H), 4.19-4.17 (m, 1H), 3.93 (d, J =6.0 Hz, 1H), 3.31-3.29 (m, 1H), 3.26-3.12 (m, 5H), 2.77-2.66 (m, 3H), 2.05-2.00 (m, 1H), 1.89-1.79 (m, 3H), 1.48-1.36 (m, 2H), 0.90-0.82 (m, 2H), 0.64-0.60 (m, 2H).
##STR00577##
Example 201
(R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piper- idin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00578## ##STR00579##
[0982] Step 1
##STR00580##
[0983] Methyl 2-bromo-2-(3,5-dichlorophenyl)acetate
[0984] To a solution of methyl 2-(3,5-dichlorophenyl)acetate (0.43 g, 90 mmol) and N-bromo- succinimide (0.72 g, 4 mmol) in carbon tetrachloride (10 mL) was added benzoyl peroxide (0.048 g, 0.2 mmol). The mixture was stirred at 80.degree. C. for 8 h, cooled to ambient temperature, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 15% ethyl acetate in petroleum ether) to give the title compound as a yellow oil (0.46 g, 80%). MS(EI) m/z: 295.9 [M].sup.+.
Step 2
##STR00581##
[0985] Methyl 2-(3,5-dichlorophenyl)-2-(4-(hydroxymethyl)piperidin-1-yl)acetate
[0986] To a solution of methyl 2-bromo-2-(3,5-dichlorophenyl)acetate (0.46 g, 1.57 mmol) acetonitrile (10 mL) was added potassium carbonate (0.65 g, 4.7 mmol) and piperidin-4-yl-methanol (0.37 g, 3.2 mmol). The reaction mixture was stirred at 80.degree. C. for 4 h, diluted with dichloromethane (30 mL) and washed with brine (20 mL.times.3). The combined organic layers were dried over anhydrous sodium sulfate, filtered and concentrated to give the crude product as colorless oil (0.5 g) which was used directly without further purification. LCMS(ESI) m/z: 332.1 [M+1].sup.+.
Step 3
##STR00582##
[0987] Methyl 2-(4-(((tert-butyldimethylsilyl)oxy)methyl)piperidin-1-yl)-2-(3,5-dichlor- ophenyl)acetate
[0988] To a solution of methyl 2-(3,5-dichlorophenyl)-2-(4-(hydroxymethyl)piperidin-1-yl)acetate (0.51 g, 1.54 mmol, crude) dichloromethane (20 mL) was added imidazole (0.35 g, 5.14 mmol) and tert-butylchlorodimethylsilane (0.47 g, 3.1 mmol). The mixture was stirred at room temperature for 2 h, then filtered, concentrated and purified by silica gel chromatography (eluting with 5% ethyl acetate in petroleum ether) to give the title compound (0.6 g, 86%) as a colorless oil. LCMS(ESI) m/z: 446.1 [M+1].sup.+.
Step 4
##STR00583##
[0989] Methyl 2-(4-(((tert-butyldimethylsilyl)oxy)methyl)piperidin-1-yl)-2-(3,5-dichlor- ophenyl)propanoate
[0990] To a solution of methyl 2-(4-(((tert-butyldimethylsilyl)oxy)methyl)piperidin-1-yl)-2-(3,5-dichlor- ophenyl)acetate (0.58 g, 1.3 mmol) in dry THF (10 mL) at -78.degree. C., was added sodium bis(trimethylsilyl) amide (1 M in THF, 2.6 mL). The resultant mixture was maintained at -78.degree. C. and stirred for 1 hour. Methyl iodide (0.55 g, 3.9 mmol) was then added and the mixture allowed to warm to room temperature overnight. The mixture was diluted with EtOAc (15 mL) and brine (30 mL), extracted with EtOAc (10 mL.times.3), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 5% EtOAc in petroleum ether) to afford the title compound as a colorless oil (0.49 g, 82%). LCMS(ESI) m/z: 460.1 [M+1].sup.+.
Step 5
##STR00584##
[0991] 2-(4-(((tert-Butyldimethylsilyl)oxy)methyl)piperidin-1-yl)-2-(3,5-d- ichlorophenyl)propan-1-ol
[0992] To a solution of methyl 2-(4-((tert-butyldimethylsilyloxy)methyl)piperidin-1-yl)-2-(3,5-dichlorop- henyl)propanoate (0.49 g , 1.06 mmol) in THF (20 mL) at -78.degree. C., diisobutyl aluminium hydride (1M in THF, 4.3 mL) was added dropwise. The resultant mixture was allowed to warm to room temperature and stirred for 16 h. The mixture was quenched with brine and the white precipitate filtered and washed with EtOAc (30 mL). The combined organic layers were dried over anhydrous sodium sulfate and concentrated to give the title compound which was used in next step without further purification. LCMS(ESI) m/z: 432.1 [M+1].sup.+.
Step 6
##STR00585##
[0993] 4-(((tert-Butyldimethylsilyl)oxy)methyl)-1-(2-(3,5-dichlorophenyl)-- 1-methoxypropan-2-yl)piperidine
[0994] To a solution of 2-(4-((tert-butyldimethylsilyloxy)methyl)piperidin-1-yl)-2-(3,5-dichlorop- henyl)propan-1-ol (0.46 g, 106 mmol, crude) in dry THF (20 mL) was added sodium hydride (60% in mineral oil, 0.4 g, 10 mmol) under N.sub.2 atmosphere at room temperature. Methyl iodide (1.41 g, 10 mmol) was added after 2 h and the resultant mixture was stirred overnight. The mixture was quenched with water (20 mL), extracted with EtOAc (10 mL.times.3), dried over sodium sulfate and concentrated. The residue was purified by silica gel chromatography (eluting vial 8% EtOAc in petroleum ether) to afford the title compound as a viscous oil (0.4 g, 85%). LCMS(ESI) m/z: 446.1 [M+1].sup.+.
Step 7
##STR00586##
[0995] (1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piperidin-4-yl)meth- anol
[0996] To a solution of 4-((tert-butyldimethylsilyloxy)methyl)-1-(2-(3,5-dichlorophenyl)-1-methox- ypropan-2-yl)piperidine (0.4 g, 0.9 mmol) in DCM (20 mL), HCl in methanol (4M, 2mL) was added dropwise. The mixture was stirred at room temperature for 2 h and concentrated in vauo to give the title compound as a pale yellow oil. LCMS(ESI) m/z: 332.1 [M+1].sup.+.
Step 8
##STR00587##
[0997] (R)-methyl 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piperidi- n-4-yl)methoxy)-2-fluorobenzoate
[0998] A mixture of (1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piperidin-4-yl)methanol (0.298 g, 0.9 mmol), methyl 5-cyclopropyl-2-fluoro-4-hydroxybenzoate (0.19 g, 0.9 mmol), triphenylphosphine (0.472 g, 1.8 mmol) and diisopropyl azodiformate (0.365 g, 1.8 mmol) in dry THF (10 mL) was stirred under an N.sub.2 atmosphere for 24 h. The reaction mixture was filtered, concentrated in vacuo, and the residue was purified by silica gel chromatography (eluting with 10% EtOAc in petroleum ether) to give the racemate (0.33 g, 70%). The enantiomer was separated by chiral SFC from the racemate. The enantiomer was arbitrarily assigned as (R)methyl 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piperidi- n-4-yl)methoxy)-2-fluorobenzoate. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, MeOH, A:B=90:10; flow: 3 mL/min; column temperature: 38.9.degree. C., RT=4.36 min). LCMS(ESI) m/z: 524.1 [M+1].sup.+.
Step 9
##STR00588##
[0999] (R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-y- l)piperidin-4-yl)methoxy)-2-fluorobenzoic acid
[1000] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 510.1 [M+1].sup.+.
Step 10
##STR00589##
[1001] (R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-y- l)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1002] The compound was synthesized as described in step 5 of Example 80. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. LCMS(ESI): Method A: RT=6.39 min, m/z: 587.31 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.49 (d, J=1.6 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.4 Hz, 1H), 3.91 (d, J=5.6 Hz, 2H), 3.53-3.40 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 2.88 (d, J=9.6 Hz, 1H), 2.57 (m, 1H), 2.28-2.13 (m, 2H), 2.01-2.02 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
##STR00590##
Example 202
(S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piper- idin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1003] The compound was synthesized as described n Example 201. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. LCMS(ESI): Method A: RT=6.52 min, m/z: 587.2 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.49 (d, J=2.0 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.8 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.53-3.40 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 2.88 (d, J=9.6 Hz, 1H), 2.57 (m, 1H), 2.28-2.13 (m, 2H), 2.02-2.00 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 203
##STR00591##
[1004] (S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-y- l)piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1005] The compound was synthesized as described in Example 201. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. LCMS(ESI): Method A: RT=6.19 min, m/z: 601.31 [M+1].sup.+. .sup.1HNMR (400 MHz, , DMSO-d.sub.6) .delta. 7.49 (d, J=2.0 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.8 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.53-3.40 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 3.08-3.06 (m, 2H), 2.88 (d, J=9.6 Hz, 1H), 2.58-2.56 (m, 1H), 2.28-2.13 (m, 2H), 2.02-2.00 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
##STR00592##
Example 204
(S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)piper- idin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1006] The compound was synthesized as described in Example 201. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)-1-methoxypropan-2-yl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. LCMS(ESI): Method A: RT=6.21 min, m/z: 601.2 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.49 (d, J=2.0 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.8 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.53-3.40 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 3.08-3.06 (m, 2H), 2.88 (d, J=9.6 Hz, 1H), 2.58-2.56 (m, 1H), 2.28-2.13 (m, 2H), 2.02-2.00 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 205
##STR00593##
[1007] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylp- ropyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00594##
[1008] Step 1
##STR00595##
[1009] 1-(4-(((tert-Butyldimethylsilyl)oxy)methyl)piperidin-1-yl)-1-(3,5-d- ichlorophenyl)-2-methylpropan-2-ol
[1010] To a solution of methyl 2-(4-((tert-butyldimethylsilyloxy)methyl)piperidin-1-yl)-2-(3,5-dichlorop- henyl)acetate (0.58 g, 1.3 mmol) in dry THF (10 mL) at -78.degree. C. was added methylmagnesium bromide (3M in THF, 2.6 mL). The resultant mixture was allowed to warm to room temperature and stirred overnight, quenched with water (20 mL), extracted with EtOAc (10 mL.times.3), dried over sodium sulfate, filtered and concentrated in vacuo. The residue was purified by silica gel chromatography (eluting with 5% EtOAc in petroleum ether) to afford title compound as a colorless oil (0.47 g, 81%). LCMS(ESI) m/z: 445.1 [M+1].sup.+.
Step 2
##STR00596##
[1011] 4-(((tert-Butyldimethylsilyl)oxy)methyl)-1-(1-(3,5-dichlorophenyl)-- 2-methoxy-2-methylpropyl)piperidine
[1012] The compound was synthesized as described in step 6 of Example 201. LCMS(ESI) m/z: 460.2 [M+1].sup.+.
Step 3
##STR00597##
[1013] (1-(1-(3,5-Dichlorophenyl)-2-methoxy-2-methylpropyl)piperidin-4-yl)- methanol
[1014] The compound was synthesized as described in step 7 of Example 201. LCMS(ESI) m/z: 346.1 [M+1].sup.+.
Step 4
##STR00598##
[1015] (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)pipe- ridin-4-yl)methoxy)-2-fluorobenzoate
[1016] The compound was synthesized as described in step 8 of Example 201. The enantiomer was separated by chiral SFC from the racemate, the enantiomer was arbitrarily assigned as (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)pipe- ridin-4-yl)methoxy)-2-fluorobenzoate. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 3 mL/min; column temperature: 40.4.degree. C., RT=2.85 min). LCMS(ESI) m/z: 538.1 [M+1].sup.+.
Step 5
##STR00599##
[1017] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl-2-methoxy-2-methylpr- opyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid
[1018] The compound was synthesized as described in step 9 of Example 201. LCMS(ESI) m/z: 524.1 [M+1].sup.+.
Step 6
##STR00600##
[1019] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylp- ropyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1020] The compound synthesized as described in step 9 of Example 201. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)- piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. LCMS(ESI): Method A: RT=6.95 min, m/z: 601.31 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 11.83 (brs, 1H), 7.50-7.44 (m, 3H), 7.15-7.13 (m, 1H), 6.86 (d, J=12.8 Hz, 1H), 3.86-3.85 (m, 2H), 3.46-3.44 (m, 2H), 3.29 (s, 1H), 3.24 (s, 3H), 3.16 (s, 3H), 2.82 (d, J=10.8 Hz, 1H), 2.00-1.95 (m, 2H), 1.72-1.71 (m, 3H), 1.55-1.54 (m, 1H), 1.40-1.23 (m, 4H), 0.93 (s, 3H), 0.88-0.83 (m, 2H), 0.65-0.61 (m, 2H).
##STR00601##
Example 206
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)p- iperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1021] The compound was synthesized as described in Example 205. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)- piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. LCMS(ESI): Method A: RT=6.95 min, m/z: 601.3 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 11.83 (brs, 1H), 7.50-7.44 (m, 3H), 7.15-7.13 (m, 1H), 6.86 (d, J=12.8 Hz, 1H), 3.86-3.85 (m, 2H), 3.46-3.44 (m, 2H), 3.29 (s, 1H), 3.24 (s, 3H), 3.16 (s, 3H), 2.82 (d, J=10.8 Hz, 1H), 2.00-1.95 (m, 2H), 1.72-1.71 (m, 3H), 1.55-1.54 (m, 1H), 1.40-1.23 (m, 4H), 0.93 (s, 3H), 0.88-0.83 (m, 2H), 0.65-0.61 (m, 2H).
##STR00602##
Example 207
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)p- iperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1022] The compound was synthesized as described in Example 205. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)- piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. LCMS(ESI): Method A: RT=7.04 min, m/z: 615.3 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.49 (d, J=2.0 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.8 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.53-3.40 (m, 2H), 3.42-3.37 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 3.07 (m, 2H), 2.88 (d, J=9.6 Hz, 1H), 2.57-2.56 (m, 1H), 2.28-2.13 (m, 2H), 2.01-2.00 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 208
##STR00603##
[1023] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-methoxy-2-methylp- ropyl)piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1024] The compound was synthesized as described in Example 205. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-4-((1 -(1-(3,5-dichlorophenyl)-2-methoxy-2-methylpropyl)piperidin-4-yl)methoxy)- -N-(ethylsulfonyl)-2-fluorobenzamide. LCMS(ESI): Method A: RT=7.12 min, m/z: 615.2 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.49 (d, J=2.0 Hz, 2H), 7.44 (m, 1H), 7.19 (d, J=8.8 Hz, 1H), 6.82 (d, J=12.8 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.53-3.40 (m, 2H), 3.42-3.37 (m, 2H), 3.18 (s, 3H), 3.10 (s, 3H), 3.07-3.06 (m, 2H), 2.88 (d, J=9.6 Hz, 1H), 2.58-2.56 (m, 1H), 2.28-2.13 (m, 2H), 2.01-2.00 (m, 1H), 1.80-1.70 (m, 3H), 1.37-1.24 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.60 (m, 2H).
##STR00604##
Example 209
5-Cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl)piperidin-4-yl)meth- oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00605##
[1025] Step 1
##STR00606##
[1026] Methyl 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl)piperidin-4-yl)met- hoxy)-2-fluorobenzoate
[1027] To a solution of methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzoyl)piperidin-4-yl)methoxy)-2-fluoro- benzoate (0.81 g, 1.7 mmol) and 2,6-di-tert-butyl-4-methylpyridine (1.4 g, 6.8 mmol) in dry THF (15 mL) under N.sub.2 atmosphere, was added trifluoromethanesulfonic anhydride dropwise at -78.degree. C. The mixture was stirred at -40.degree. C. for 3 h followed by the addition of MeLi (3M in THF, 2.3 mL). The reaction mixture was allowed to warm to room temperature and stirred overnight, quenched with water (10 mL), extracted with EtOAc (10 mL.times.3), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 1.5% ethyl acetate in petroleum ether) to afford the target compound as a white solid (0.17 g, 20%). LCMS(ESI): m/z: 494.2 [M+1].sup.+.
Step 2
##STR00607##
[1028] 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl)piperidin-4-- yl)methoxy)-2-fluorobenzoic acid
[1029] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 480.2 [M+1].sup.+.
Step 3
##STR00608##
[1030] 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl)piperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1031] The compound was synthesized as described in step 5 of Example 80. LCMS(ESI): Method A: RT=7.01 min, m/z: 557.0 [M+1].sup.+. .sup.1HNMR (500 MHZ, DMSO-d.sub.6) .delta. 7.51 (s, 2H), 7.44 (s, 1H), 7.21 (d, J=8.5 Hz, 1H), 6.76 (d, J=13.0 Hz, 1H), 3.90 (d, J=5.5 Hz, 2H), 3.00 (s, 3H), 2.75-2.73 (m, 2H), 2.12-2.10 (m, 2H), 2.01-1.98 (m, 1H), 1.78-1.76 (m, 3H), 1.35-1.23 (m, 8H), 0.89-0.85 (m, 2H), 0.60-0.57 (m, 2H).
Example 210
##STR00609##
[1032] 5-Cyclopropyl-4-((1-(2-((3,5-dichlorophenyl)propan-2-yl)piperidin-4- -yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1033] The compound was synthesized as described in Example 209. LCMS(ESI): Method A: RT=6.81 min, m/z: 571.2 [M+1].sup.+. .sup.1HNMR (400 MHz, DMSO-d.sub.6) .delta. 7.51 (s, 2H), 7.44 (s, 1H), 7.17 (d, J=8.8 Hz, 1H), 6.81 (d, J=12.8 Hz, 1H), 3.92 (d, J=5.2 Hz, 2H), 3.29-3.24 (m, 2H), 2.76-2.67 (m, 2H), 2.14-2.09 (m, 2H), 2.03-1.96 (m, 1H), 1.77-1.76 (m, 3H), 1.37-1.24 (m, 8H), 1.17 (t, J=7.6 Hz, 3H), 0.89-0.85 (m, 2H), 0.63-0.59 (m, 2H).
##STR00610##
Example 211
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-methylpiperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1034] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-methylpiperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: IC 4.6.times.150 mm 5.mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=70:30; flow: 3.0 mL/min; column temperature: 39.9.degree. C.; RT=6.78min). LCMS(ESI) Method A: RT=5.50 min, m/z: 556.7 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.53-7.43 (m, 3H), 7.20 (d, J=, 8.4 Hz, 1H), 6.89 (d, J=12.4 Hz, 1H), 3.80 (s, 3H), 3.14 (s, 3H), 3.21-3.16 (m, 2H), 2.42-2.40 (m, 1H), 2.01-1.99 (m, 1H), 1.69-1.68 (m, 2H), 1.47-1.35 (m, 3H), 0.89-0.86 (m, 2H), 0.62-0.61 (m, 2H).
##STR00611##
Example 212
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-methylpiperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1035] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-methylpiperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: IC 4.6.times.150 mm 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: Method A:B=70:30; flow: 3.0 as mL/min; column temperature: 39.9.degree. C.; RT=5.75 min). LCMS(ESI) Method A: RT=5.46 min, mm/z: 556.7 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.53-7.43 (m, 3H), 7.20 (d, J=8.4 Hz, 1H), 6.89 (d, J=12.4 Hz, 1H), 3.80 (s, 3H), 3.14 (s, 3H), 3.21-3.16 (m, 2H), 2.44-2.40 (m, 1H), 2.01-1.99 (m, 1H), 1.69-1.68 (m, 2H), 1.47-1.35 (m, 6H), 1.04 (s, 3H), 0.90-0.85 (m, 2H), 0.64-0.60 (m, 2H).
##STR00612##
Example 213
5-Cyclopropyl-4-((1-(2,4-dichlorobenzyl)piperidin-4-yl)methoxy)-N-(ethylsu- lfonyl)-2-fluorobenzamide
[1036] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.25 min, m/z: 542.8 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.60-7.51 (m, 3H), 7.13 (d, J=8.4 Hz, 1H), 6.91 (d, J=12.8 Hz, 1H), 3.94 (d, J=6.0 Hz, 2H), 3.62 (s, 2H), 3.40-3.37 (m, 2H), 2.92-2.90 (m, 2H), 2.20-1.79 (m, 6H), 1.42-1.37 (m, 2H), 1.23-1.19 (m, J=7.4 Hz, 3H), 0.89-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00613##
Example 214
(R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)piperidin-4-yl)methox- y)-2-fluoro-N-(methylsulfonyl)benzamide
[1037] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)piperidin-4-yl)metho- xy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m: mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=70:30; flow: 3.0 mL/min; column temperature: 37.7.degree. C.; RT=6.44 min). LCMS(ESI) Method A: RT=6.11 min, m/z: 542.9 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.63-7.62 (m, 2H), 7.39-7.38 (m, 1H), 7.17 (d, J=6.4 Hz, 1H), 6.80 (d, J=10.4 Hz, 1H), 3.90 (d, J=4.4 Hz, 3H), 3.08 (s, 3H), 2.86-2.84 (m, 2H), 2.01-1.98 (m, 3H), 1.85-1.78 (m, 3H), 1.39-1.35 (m, 5H), 0.88-0.86 (m, 2H), 0.62-0.59 (m, 2H).
##STR00614##
Example 215
(S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)piperidin-4-yl)methox- y)-2-fluoro-N-(methylsulfonyl)benzamide
[1038] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)piperidin-4-yl)metho- xy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=70:30; flow: 3.0 mL/min; column temperature: 37.7.degree. C.; RT=10.00 min). LCMS(ESI) Method A: RT=6.11 min, m/z: 542.9 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.64-7.62 (m, 2H), 7.38-7.36 (m, 1H), 7.17 (d, J=6.8 Hz, 1H), 6.80(d, J=10.4 Hz, 1H), 3.91 (d, J=4.4 Hz, 3H), 3.10 (s, 3H), 2.92-2.91 (m, 2H), 2.19-1.17 (m, 2H), 2.01-1.78 (m, 4H), 1.42-1.35 (m, 5)H, 0.88-0.86) (m, 2H), 0.64-0.61 (m, 2H).
Example 216
##STR00615##
[1039] 4-((1-(3-Chloro-5-(trifluoromethyl)piperidin)piperidin-4-yl)methoxy- )-5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
[1040] The compound was synthesized as described as Example 88. LCMS(ESI) Method A: RT=6.31 min, m/z: 576.8 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.77-7.67 (m, 3H), 7.14 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.8 Hz, 1H), 3.94 (d, J=5.6 Hz, 2H), 3.68 (s, 2H), 3.35-3.33 (m, 2H), 2.91-2.88 (m, 2H), 2.14-1.79 (m, 6H), 1.40-1.38 (m, 2H), 1.20 (t, J=7.4 Hz, 3H), 0.89-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00616##
Example 217
4-((1-((5-Chloro-6-isopropoxypyridin-3-yl)methyl)piperidin-4-yl)methoxy)-5- -cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1041] The compound was synthesized as described in Example 190. LCMS(ESI) Method A: RT=5.88 min, m/z: 553.8 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.8.06 (d, J=4.0 Hz, 1H), 7.15 (d, J=9.6 Hz, 1H), 7.10 (d, J=4.4 Hz, 1H), 6.89 (d, J=10.4 Hz, 1H), 5.30-5.28 (m, 1H), 3.95-3.94 (m, 2H), 3.60 (s, 2H), 3.21 (s, 3H), 2.89-2.87 (m, 2H), 2.16-2.11 (m, 2H), 2.03-2.01 (m, 1H), 1.80-1.78 (m, 3H), 1.41-1.39 (m, 2H), 1.31 (d, J=5.2 Hz, 6H), 0.91-0.87 (m, 2H), 0.66-0.63 (m, 2H).
##STR00617##
Example 218
(R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)-4-methylpiperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1042] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)-4-methylpiperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H (4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A: B=75:25; flow: 3.0 mL/min; column temperature: 40.0.degree. C.; RT=4.18 min). LCMS(ESI) Method A: RT=6.25 min, m/z: 556.8 [M+1].sup.+. .sup.1H NMR (300 MHz, DMSO-d.sub.6): .delta.7.65-7.63 (m, 2H), 7.40-7.38 (m, 1H), 7.20(d, J=8.1 Hz, 1H), 6.84 (d, J=12.9 Hz, 1H), 3.79-3.78 (m, 2H), 3.11 (s, 3H), 2.57-2.56 (m, 1H), 2.45-2.42 (m, 3H), 2.01-1.99 (m, 1H), 1.69-1.37 (m, 8H), 1.04 (s, 3H), 0.89-0.85 (m, 2H), 0.62-0.60 (m, 2H).
Example 219
##STR00618##
[1043] (S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)-4-methylpiper- idin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1044] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)ethyl)-4-methylpiperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H (4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A: B=75:25; flow: 3.0 mL/min; column temperature: 40.0.degree. C.; RT=6.43 min). LCMS(ESI) Method A: RT=6.26 min, m/z: 556.8 [M+1].sup.+. .sup.1H NMR (300 MHz, DMSO-d.sub.6): .delta.7.65-7.63 (m, 2H), 7.40-7.38 (m, 1H), 7.20 (d, J=8.1 Hz, 1H), 6.83 (d, J=12.9 Hz, 1H), 3.79-3.78 (m, 2H), 3.11 (s, 3H), 2.57-2.56 (m, 1H), 2.45-2.42 (m, 3H), 2.01-1.99 (m, 1H), 1.69-1.37 (m, 8H), 1.04 (s, 3H), 0.88-0.86 (m, 2H), 0.62-0.60 (m, 2H).
##STR00619##
Example 220
4-(((1-(3-Chloro-4-(trifluoromethyl)piperidin)piperidin-4-yl)methoxy)-5-cy- clopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1045] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.25 min, m/z: 562.8 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.86-7.84 (m, 1H), 7.69-7.52 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.8 Hz, 1H), 3.94 (d, J=6.0 Hz, 2H), 3.72 (s, 2H), 3.20 (s, 3H), 2.95-2.92 (m, 2H), 2.20-1.80 (m, 6H), 1.43-1.40 (m, 2H), 0.90-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00620##
Example 221
(R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzoyl)piperidin-3-yl)oxy)-2-fluoro-- N-(methylsulfonyl)benzamide
[1046] The compound was synthesized as described in step Example 134. LCMS(ESI) Method A: RT=4.69 min, m/z: 528.8 [M+1].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.74-7.28 (m, 3H), 7.17 (d, J=8.0 Hz, 1H), 6.78 (d, J=12.5 Hz, 1H), 4.68-4.67 (m, 1H), 4.19-4.16 (m, 1H), 3.68-3.50 (m, 2H), 3.28-3.14 (m, 4H), 2.02-1.60 (m, 5H), 0.93-0.92 (m, 2H), 0.71-0.68 (m, 2H).
Example 222
##STR00621##
[1047] (R)-5-cyclopropyl-2-fluoro-4-((1-(2-methoxy-1-phenylethyl)piperidin- -4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1048] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-2-fluoro-4-((1-(2-methoxy-1-phenylethyl)piperidin-4-yl)- methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250mm, 5 .mu.m; mobile Phase: A :supercritical CO.sub.2, B: MeOH, A:B=75:25; flow: 3.0 mL/min; column temperature: 40.2.degree. C.; RT=3.18 min). LCMS(ESI) Method A: RT=4.83 min, m/z: 505.0 [M+1].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta.7.36-7.32 (m, 5H), 7.16 (d, J=8.0 Hz, 1H), 6.82 (d, J=13.5 Hz, 1H), 3.90-3.71 (m, 5H), 3.29-3.25 (m, 4H), 3.11-2.90 (m, 4H), 2.35-1.75 (m, 6H), 1.49-1.38 (m, 2H), 0.88-0.85 (m, 2H), 0.61-0.60 (m, 2H).
##STR00622##
Example 223
(S)-5-cyclopropyl-2-fluoro-4-((1-(2-methoxy-1-phenylethyl)piperidin-4-yl)m- ethoxy)-N-(methylsulfonyl)benzamide
[1049] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-2-fluoro-4-((1-(2-methoxy-1-phenylethyl)piperidin-4-yl)- methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, MeOH, A:B=75:25; flow: 3.0 mL/min; column temperature: 40.2.degree. C.; RT=5.20 min). LCMS(ESI) Method A: RT=4.83 min, m/z: 504.9 [M+1].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta.7.36-7.32 (m, 5H), 7.16 (d, J=9.0 Hz, 1H), 6.82 (d, J=13.0 Hz, 1H), 3.90-3.69 (m, 5H), 3.31 (s, 3H), 3.11-2.90 (m, 5H), 2.01-1.75 (m, 6H), 1.49-1.38 (m, 2H), 0.88-0.85 (m, 2H), 0.61-0.59 (m, 2H).
##STR00623##
Example 224
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperidi- n-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00624##
[1050] Step 1
##STR00625##
[1051] 1-(3,5 -Dichlorophenyl)-2,2-difluoroethanol
[1052] A mixture of 3,5-dichlorobenzaldehyde (800 mg, 4.6 mmol), (difluoromethyl)trimethylsilane (850 mg, 6.8 mmol) and CsF (441 mg, 2.3 mmol) in DMF (20 mL) was stirred for 36 h at room temperature. The reaction mixture was diluted with ethyl acetate (50 mL), washed with brine (30 mL.times.3), dried over anhydrous sodium sulfate and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=10/1) to afford the title compound as a pale yellow oil. (500 mg, 48%). LCMS(ESI) m/z: 225.0 [M-1].sup.-.
Step 2
##STR00626##
[1053] Methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperidin-4- -yl)methoxy)-2-fluorobenzoate
[1054] To a mixture of 1-(3,5-dichlorophenyl)-2,2-difluoroethanol (200 mg, 0.9 mmol) and DIPEA (270 mg, 2.7 mmol) in DCM (20 mL) was added trifluoromethanesulfonic anhydride (507 mg, 1.8 mmol) at 0.degree. C., and the mixtures stirred for 2 h. Methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (277 mg, 0.9 mmol) was then added at 0.degree. C. and the mixture left to warm to room temperature and stirred for 16 h. The reaction was quenched with ethyl acetate (40 mL) and water (40 mL), extracted with ethyl acetate (20 mL.times.3), dried over anhydrous sodium sulfate and concentrated. The residue was purified by silica gel chromatography (eluting with petroleum ether/ethyl acetate=5/1) to afford the title compound as a pale yellow oil. (230 mg, 20%).
Step 3
##STR00627##
[1055] 5-Cyclopropyl-4-((1-(1-(3,5dichlorophenyl)-2,2-difluoroethyl)piperi- din-4-yl)methoxy)-2-fluorobenzoic acid
[1056] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 502.1 [M+1].sup.+.
Step 4
##STR00628##
[1057] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)p- iperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1058] The compound was synthesized as described in step 5 of Example 80. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperid- in-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 3.0 mL/min; column temperature: 40.1.degree. C.; RT=15.85 min). LCMS(ESI) Method A: RT=6.16 min, m/z: 579.0 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.62-7.47 (m, 3H), 7.14 (d, J=8.4 Hz, 1H), 6.89-6.65 (m, 2H), 4.04-3.89 (m, 3H), 3.29-3.24 (m, 3H), 2.98-2.92 (m, 2H), 2.17-2.15 (m, 1H), 2.00-1.73 (m, 5H), 1.35-1.32 (m, 2H), 0.89-0.84 (m, 2H), 0.66-0.62 (m, 2H).
Example 225
##STR00629##
[1059] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)p- iperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1060] The compound was synthesized as described in Example 224. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperid- in-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 3.0 mL/min; column temperature: 40.1.degree. C.; RT=13.82 min). LCMS(ESI) Method A: RT=6.16 min, m/z: 579.0 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.62-7.42 (m, 3H), 7.14 (d, J=8.4 Hz, 1H), 6.77-6.65 (m, 2H), 4.04-3.85 (m, 3H), 3.29-3.27 (m, 1H), 3.02-2.92 (m, 4H), 2.16-2.14 (m, 1H), 2.00-1.68 (m, 5H), 1.39-1.29 (m, 2H), 0.88-0.83 (m, 2H), 0.60-0.58 (m, 2H).
Example 226
##STR00630##
[1061] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)p- iperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1062] The compound was synthesized as described in Example 224. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperid- in-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5.mu.m: mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 3.0 mL/min; column temperature: 42.0.degree. C.; RT=13.5 min). LCMS(ESI) Method A: RT=6.15 min, m/z: 593.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.62-7.61 (m, 1H), 7.47-7.46 (m, 2H), 7.12 (d, J=8.4 Hz, 1H), 6.90-6.58 (m, 2H), 4.08-3.89 (m, 3H), 3.39-3.37 (m, 2H), 3.01-2.92 (m, 2H), 2.15-2.13 (m, 1H), 1.98-1.72 (m, 4H), 1.35-1.19 (m, 6H), 0.87-0.84 (m, 2H), 0.62-0.60 (m, 2H).
##STR00631##
Example 227
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperidi- n-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1063] The compound was synthesized as described in Example 224. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2-difluoroethyl)piperid- in-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH, A:B=85:15; flow: 3.0 mL/min; column temperature: 42.0.degree. C.; RT=12.33 min). LCMS(ESI) Method A: RT=6.15 min, m/z: 593.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta.7.62-7.61 (m, 1H), 7.47-7.46 (m, 2H), 7.12 (d, J=8.4 Hz, 1H), 6.90-6.58 (m, 2H), 4.08-3.89 (m, 3H), 3.39-3.37 (m, 2H), 3.01-2.94 (m, 2H), 2.15-1.91 (m, 3H), 1.76-1.68 (m, 3H), 1.35-1.32 (m, 2H), 1.21 (t, J=7.4 Hz, 3H), 0.89-0.84 (m, 2H), 0.66-0.63 (m, 2H).
Example 228
##STR00632##
[1064] 5-Cyclopropyl-2-fluoro-4-((1-(3-fluoro-5-(trifluoromethyl)piperidin- )piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1065] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.71 min, m/z 546.9 [M+1].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.34 (s, 1H), 7.62-7.53 (m, 3H), 7.16 (d, J=9.0 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 3.95 (d, J=6.0 Hz, 2H), 3.77 (s, 2H), 3.21 (s, 3H), 2.97-2.95 (m, 2H), 2.27-2.23 (m, 2H), 2.04-2.00 (m, 1H), 1.87-1.82 (m, 3H), 1.46-1.40 (m, 2H), 0.90-0.86 (m, 2H), 0.66-0.63 (m, 2H).
##STR00633##
Example 229
4-((1-(4-Chloro-3-(trifluoromethyl)piperidin)piperidin-4-yl)methoxy)-5-cyc- lopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1066] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.88 min, m/z 563.0 [M+1].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.24 (brs, 1H), 7.83 (s, 1H), 7.75 (d, J=8.0 Hz, 1H), 7.67 (d, J=8.0 Hz, 1H), 7.17 (d, J=8.5 Hz, 1H), 6.85 (d, J=12.5 Hz, 1H), 3.93 (d, J=6.0 Hz, 2H), 3.74 (s, 2H), 3.14 (s, 3H), 2.97-2.95 (m, 2H), 2.24 (s, 2H), 2.04-1.98 (m, 1H), 1.86-1.77 (m, 3H), 1.45-1.38 (m, 2H), 0.89-0.86 (m, 2H), 0.64-0.61 (m, 2H).
Example 230
##STR00634##
[1067] (R)-4-((1-(1-(3-chloro-5-fluorophenyl)ethyl)piperidin-4-yl)methoxy)- -5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1068] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(3-chloro-5-fluorophenyl)ethyl)piperidin-4-yl)methoxy)-5-cyc- lopropyl-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=70:30; flow: 2.1 mL/min; column temperature: 40.degree. C.; RT=4.01 min). LCMS(ESI) Method A: RT =5.99 min, m/z 527.2 [M+1].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.7.35 (d, J=8.8 Hz, 1H), 7.30 (s, 1H), 7.21 (d, J=9.2 Hz, 1H), 7.17 (d, J=8.4 Hz, 1H), 6.83 (d, J=12.4 Hz, 1H), 3.91 (d, J=5.6Hz, 2H), 3.74 (brs, 1H), 3.11 (s, 3H), 3.07-3.03 (m, 1H), 2.91-2.87 (m, 1H), 2.12-2.09 (m, 2H), 2.03-1.98 (m, 1H), 1.84-1.77 (m, 3H), 1.41-1.34 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.59 (m, 2H).
##STR00635##
Example 231
(S)-4-((1-(1-(3-chloro-5-fluorophenyl)ethyl)piperidin-4-yl)methoxy)-5-cycl- opropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1069] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(3-chloro-5-fluorophenyl)ethyl)piperidin-4-yl)methoxy)-5-cyc- lopropyl-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=70:30; flow: 2.1 mL/min; column temperature: 40.degree. C.; RT=4.83 min). LCMS(ESI) Method A: RT =5.99 min, m/z 527.2 [M+1].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.7.35 (d, J=8.8 Hz, 1H), 7.30 (s, 1H), 7.21 (d, J=9.2 Hz, 1H), 7.17 (d, J=8.4 Hz, 1H), 6.83 (d, J=12.4 Hz, 1H), 3.91 (d, J=5.6 Hz, 2H), 3.74 (brs, 1H), 3.11 (s, 3H), 3.07-3.03 (m, 1H), 2.91-2.87 (m, 1H), 2.12-2.09 (m, 2H), 2.03-1.98 (m, 1H), 1.84-1.77 (m, 3H), 1.41-1.34 (m, 5H), 0.89-0.85 (m, 2H), 0.63-0.59 (m, 2H).
##STR00636##
Example 232
5-Cyclopropyl-4-((1-(3,4-dichlorobenzoyl)piperidin-4-yl)methoxy)-2-fluoro-- N-(methylsulfonyl)benzamide
[1070] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=5.03 min, m/z 542.8 [M+1].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.89 (s, 1H), 7.72 (d, J=8.0 Hz, 1H), 7.66 (d, J=2.0 Hz, 1H), 7.39-7.37 (m, 1H), 7.15 (d, J=9.0 Hz, 1H), 6.95 (d, J=12.5 Hz, 1H), 4.48 (brs, 1H), 3.98 (d, J=6.0 Hz, 2H), 3.56 (brs, 1H), 3.29 (s, 3H), 3.18-3.12 (m, 1H), 2.84-2.76 (m, 1H), 2.13-2.10 (m, 1H), 2.05-1.99 (m, 1H), 1.89-1.76(m, 2H), 1.33 (brs, 2H), 0.91-0.87 (m, 2H), 0.68-0.65 (m, 2H).
Example 233
##STR00637##
[1071] 5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,4-dichlorobenzoyl)pi- peridin-4-yl)methoxy)-2-fluorobenzamide
[1072] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=5.18 min, m/z 568.8 [M+1].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.81 (s, 1H), 7.72 (d, J=8.5 Hz, 1H), 7.66 (d, J=2.0 Hz, 1H), 7.39-7.37 (m, 1H), 7.14 (d, J=8.0 Hz, 1H), 6.97 (d, J=12.3 Hz, 1H), 4.48 (brs, 1H), 3.99 (d, J=5.0 Hz, 2H), 3.56 (brs, 1H), 3.12-3.04 (m, 2H), 2.84 (s, 1H), 2.13-2.12 (m, 1H), 2.04-1.99 (m, 1H), 1.89 (s, 1H), 1.77 (s, 1H), 1.33 (s, 2H), 1.12-1.07 (m, 4H), 0.91-0.87 (m, 2H), 0.68-0.65 (m, 2H).
##STR00638##
Example 234
(R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl)methoxy)-- 2-fluoro-N-(methylsulfonyl)benzamide
##STR00639##
[1073] Step 1
##STR00640##
[1074] 1-(3,5-Dichlorobenzyl)-2-oxopiperidine-4-carboxylic acid
[1075] A mixture of methyl 2-oxopiperidine-4-carboxylate (1.00 g, 6.36 mmol), 1,3-dichloro-5-(chloromethyl)benzene (1.49 g, 7.63 mmol) and potassium hydroxide (1.79 g, 31.8 mmol) in DMSO (30 was stirred at room temperature for 4 h. The reaction was quenched with HCl (3.0 M, 50 mL) and ethyl acetate (150 mL). The organic layer was separated and washed with brine (50 mL.times.3), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 2% ethyl acetate in petroleum ether) to afford target compound as white solid (1.65 g, 85%). LCMS(ESI) m/z: 302.0 [M+1].sup.+.
Step 2
##STR00641##
[1076] Methyl 1-(3,5-dichlorobenzyl)-2-oxopiperidine-4-carboxylate
[1077] A solution of 1-(3,5-dichlorobenzyl)-2-oxopiperidine-4-carboxylic acid (1.65 g, 5.46 mmol) in thionyl chloride (30 mL) was refluxed for 2 h. Then the solution was added dropwise to methanol (100 mL) at 0.degree. C. and the mixture stirred at room temperature For 2 h. The mixture was concentrated in vacuo to afford the target compound as brown oil which was used without further purification. LCMS(ESI) m/z: 316.0 [M+1].sup.+.
Step 3
##STR00642##
[1078] 1-(3,5-Dichlorobenzyl)-4-(hydroxymethyl)piperidin-2-one
[1079] A mixture of methyl 1-(3,5-dichlorobenzyl)-2-oxopiperidine-4-carboxylate (1.80 g, 5.69 mmol) and sodium borohydride (1.08 g, 28.5 mmol) in THF (20 mL) and MeOH (10 mL) was stirred at room temperature for 16 h. The solvent was removed and the residue was purified by reverse phase chromatography (eluting with 25-30% CH.sub.3CN in 0.5% NH.sub.4HCO.sub.3) to give target compound as brown oil (630 mg, 38%). LCMS(ESI) m/z: 288.0 [M+1].sup.+.
Step 4
##STR00643##
[1080] (1-(3,5-Dichlorobenzyl)-2-oxopiperidin-4-yl)methyl 4-methylbenzenesulfonate
[1081] A mixture of 1-(3,5-dichlorobenzyl)-4-(hydroxymethyl)piperidin-2-one (630 mg, 2.19 mmol) and sodium hydride (437 mg, 10.9 mmol) in THF (30 mL) was stirred at room temperature for 30 min. Tosyl chloride (500 mg, 2.62 mmol) in THF (10 mL) was then added and the resulting mixture was stirred at room temperature for 16 h. The react was quenched with ice water, extracted with ethyl acetate (100 mL), washed with brine (50 mL.times.2), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase chromatography (eluting with 50-60% CH.sub.3CN in 0.5% NH.sub.4HCO.sub.3) to afford the target compound as brown oil (380 mg, 39%). LCMS(ESI) m/z: 442.0 [M+1].sup.+.
Step 5
##STR00644##
[1082] (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl)me- thoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1083] A mixture of (1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl)methyl 4-methylbenzenesulfonate (380 mg, 0.859 mmol), potassium carbonate (1.19 g, 8.61 mmol) and 5-cyclopropyl-2-fluoro-4-hydroxy-N-(methylsulfonyl)benzamide (352 mg, 1.29 mmol), in DMF (20 mL) was stirred at 85.degree. C. for 16 h. The reaction was quenched with ethyl acetate (100 mL) and HCl (2.0 M, 40 mL), washed with brine (50 mL.times.2), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase chromatography (eluting with 25-40% CH.sub.3CN in 0.5% NH.sub.4HCO.sub.3) to afford the racemate as a white solid. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.degree. C.; RT=5.59 min). LCMS(ESI) Method A: RT=5.14 min, m/z: 542.80 [M+1].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.11.92 (s, 1H), 7.53-7.52 (m, 1H), 7.31-7.29 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.95 (d, J=12.8 Hz, 1H), 4.56-4.47 (m, 2H), 4.00 (d, J=6.4 Hz, 2H), 3.32-3.30 (m, 2H), 3.28 (s, 3H), 2.57-2.51 (m, 1H), 2.44-2.39 (m, 1H), 2.30-2.24 (m, 1H), 2.07-1.98 (m, 2H), 1.69-1.63 (m, 1H), 0.92-0.87 (m, 2H), 0.69-0.65 (m, 2H).
##STR00645##
Example 235
(S)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl)methoxy)-- 2-fluoro-N-(methylsulfonyl)benzamide
[1084] The compound was synthesized as described in Example 234. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)-2-oxopiperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, (0.1% DEA), A: B=75:25; flow: 2.25 mL/min: column temperature: 40.degree. C.; RT=7.28 min). LCMS(ESI) Method A: RT=5.12 min, m/z: 542.80 [M+1].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.11.92 (s, 1H), 7.53-7.52 (m, 1H), 7.31-7.29 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.95 (d, J=12.8 Hz, 1H), 4.56-4.47 (m, 2H), 4.00 (d, J=6.4 Hz, 2H), 3.32-3.30 (m, 2H), 3.28 (s, 3H), 2.57-2.51 (m, 1H), 2.44-2.39 (m, 1H), 2.30-2.24 (m, 1H), 2.07-1.98 (m, 2H), 1.69-1.63 (m, 1H), 0.92-0.87 (m, 2H), 0.69-0.65 (m, 2H).
##STR00646##
Example 236
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichloro-2-fluorophenyl)ethyl)piperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1085] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichloro-2-fluorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1 DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.8.degree. C.; RT=3.74 min). LCMS(ESI) Method A: RT=6.22 min, m/z: 561.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) 7.69 (dd, J=6.1, 2.6 Hz, 1H), 7.47 (dd, J=5.3, 2.6 Hz, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.83 (d, J=12.8 Hz, 1H), 3.90-3.89 (m, 3H), 3.14 (s, 3H), 3.03-3.02 (m, 1H), 2.87-2.85 (m, 1H), 2.01-2.00 (m, 2H), 1.94-1.68 (m, 4H), 1.42-1.26 (m, 5H), 0.91-0.82 (m, 2H), 0.61 (d, J=5.1 Hz, 2H).
##STR00647##
Example 237
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichloro-2-fluorophenyl)ethyl)piperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1086] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichloro-2-fluorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1 DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.8.degree. C.; RT=3.11 min). LCMS(ESI) Method A: RT=5.83 min, m/z: 561.0 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.69 (dd, J=1.2.6 Hz, 1H), 7.47 (dd, J=5.3, 2.5 Hz, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.82 (d, J=12.7 Hz, 1H), 3.88-3.91 (m, 3H), 3.12 (s, 3H), 3.03 (d, J=9.7 Hz, 1H), 2.86-2.85 (m, 1H), 1.99-1.97 (m, 2H), 1.93-1.69 (m, 4H), 1.40-1.25 (m, 5H), 0.86-0.84 (m, 2H), 0.61-0.60 (m, 2H).
##STR00648##
Example 238
(S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)piperidin-4-yl)methox- y)-2-fluoro-N-(methylsulfonyl)benzamide
[1087] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1 DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 38.1.degree. C.; RT=4.08 min). LCMS(ESI) Method A: RT=6.43 min, m/z: 542.9 [M+1].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.56 (d, J=8.5 Hz, 1H), 7.44 (d, J=2.1 Hz, 1H), 7.33 (dd, J=8.4, 2.1 Hz, 1H), 7.22 (d, J=8.3 Hz, 1H), 6.52 (d, J=12.8 Hz, 1H), 4.34 (brs, 1H), 3.74 (s, 2H), 3.45 (s, 1H), 3.06-3.04 (m, 3H), 2.97-2.96 (m, 1H), 2.52-2.29 (m, 2H), 1.91-1.90 (m, 3H), 1.80-1.79 (m, 1H), 1.56-1.55 (m, 1H), 1.42-1.40 (m, 4H), 0.80-0.73 (m, 2H), 0.51-0.50 (m, 2H).
##STR00649##
Example 239
(R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)piperidin-4-yl)methox- y)-2-fluoro-N-(methylsulfonyl)benzamide
[1088] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6=250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A: B=75:25; flow: 2.25 mL/min; column temperature: 41.5.degree. C.; RT=2.87 min). LCMS(ESI) Method A: RT =6.48 min, m/z: 542.9 [M+1].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.56 (d, J=8.5 Hz, 1H), 7.44 (d, J=2.1 Hz, 1H), 7.33 (dd, J=8.4, 2.1 Hz, 1H), 7.22 (d, J=8.3 Hz, 1H), 6.52 (d, J=12.8 Hz, 1H), 4.34-4.33 (m, 1H), 3.74 (brs, 2H), 3.43-3.44 (m, 1H), 3.06 (s, 3H), 2.97-2.96 (m, 1H), 2.52-2.29 (m, 2H), 1.91-1.90 (m, 3H), 1.80 (m, 1H), 1.56 (m, 1H), 1.42 (m, 4H), 0.75 (m, 2H), 0.51 (m, 2H).
Example 240
##STR00650##
[1089] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)propyl)piperidin-4-y- l)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1090] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) propyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.5% DEA), A: B=70:30; flow: 2.1 mL/min; column temperature: 41.0.degree. C.; RT=10.67 min). LCMS(ESI) Method A: RT =6.42 min, m/z: 557.3 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.52 (m, 1H), 7.34 (d, J=1.8 Hz, 2H), 7.16 (d, 8.5 Hz, 1H), 6.81 (d, J=12.9 Hz, 1H), 3.88 (d, J=6.0 Hz, 2H), 3.53 (m, 1H), 3.13 (s, 3H), 3.05 (m, 1H), 2.93 (m, 1H), 2.01 (m, 4H), 1.76 (m, 4H), 1.34 (m, 2H), 0.86 (m, 2H), 0.72 (m, 3H), 0.60 (m, 2H).
##STR00651##
Example 241
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)propyl)piperidin-4-yl)metho- xy)-2-fluoro-N-(methylsulfonyl)benzamide
[1091] The compound was synthesized as described in Example 88. The enantiomer was separated by Chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl propyl)piperidin-4-yl)methoxy)-2-fluoro-N- (methylsulfonyl)benzamide. Chiral HPLC (column: OZ-H, 4.6.times.250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.5% DEA), A:B=70:30; flow: 2.1 mL/min; column temperature: 39.3.degree. C.; RT=9.34 min). LCMS(ESI) Method A: RT=6.35 min, m/z: 557.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.52 (m, 1H), 7.34 (d, J=1.8 Hz, 2H), 7.16 (d, J=8.5 Hz, 1H), 6.81 (d, J=12.9 Hz, 1H), 3.88 (d, J=6.0 Hz, 2H), 3.53 (m, 1H), 3.13 (s, 3H), 3.05 (m, 1H), 2.93 (m, 1H), 2.01 (m, 4H), 1.76 (m, 4H), 1.34 (m, 2H), 0.85 (m, 2H), 0.72 (m, 3H), 0.61 (m, 2H).
##STR00652##
Example 242
5-Cyclopropyl-4-((1-(3,5-dichloro-2-fluorobenzyl)piperidin-4-yl)methoxy)-2- -fluoro-N-(methylsulfonyl)benzamide
[1092] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.98 min, m/z: 547.0 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.72 (dd, J=6.1, 2.6 Hz, 1H), 7.47 (d, J=5.5, 2.6 Hz, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.85 (d, J=12.9 Hz, 1H), 3.92 (d, J=5.8 Hz, 2H), 3.63 (s, 2H), 3.16 (s, 3H), 2.89 (m, 2H), 2.12 (m, 2H), 2.01 (m, 1H), 1.79 (m, 3H), 1.38 (m, 2H), 0.85 (m, 2H), 0.63 (m, 2H).
Example 243
##STR00653##
[1093] (S)-4-((1-(1-(2-chloro-4-fluorophenyl)ethyl)piperidin-4-yl)methoxy)- -5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1094] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second fraction was arbitrarily assigned as (S)-4-((1-(1-(2-chloro-4-fluorophenyl) ethyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)be- nzamide. Chiral HPLC (column: AD-H, 4.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=70:30; flow: 2.1 mL/min; column temperature: 41.6.degree. C.; RT=5.88 min). LCMS(ESI) Method A: RT=5.88 min, m/z: 526.9 [M+1].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.61 (dd, J=8.6, 6.6 Hz, 1H), 7.41 (dd, J=8.8, 2.5 Hz, 1H), 7.24 (m, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.84 (d, J=12.8 Hz, 1H), 3.91 (m, 3H), 3.15 (m, 4H), 2.75 (m, 1H), 2.12 (m, 1H), 2.01 (m, 2H), 1.75(m, 3H), 1.39 (m, 1H), 1.28 (m, 4H), 0.87 (m, 2H), 0.62 (m, 2H).
Example 244
##STR00654##
[1095] (R)-4-((1-(1-(2-chloro-4-fluorophenyl)ethyl)piperidin-4-yl)methoxy)- -5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1096] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(2-chloro-4-fluorophenyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamid- e. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=70:30; flow: 2.1 mL/min; column temperature: 38.1.degree. C.; RT=3.41 min). LCMS(ESI) Method A: RT=5.88 min, m/z: 526.9 [M+1].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.65 (dd, J=8.6, 6.6 Hz, 1H), 7.43 (dd, J=8.8, 2.5 Hz, 1H), 7.27 (m, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 4.03 (m, 3H), 3.21 (m, 4H), 2.81 (m, 1H), 2.17 (m, 2H), 2.01 (m, 1H), 1.81 (m, 1H), 1.44 (m, 1H), 1.33. (m, 4H), 0.88 (m, 2H), 0.64 (m, 2H).
Example 245
##STR00655##
[1097] (S)-4-((1-(1-(2-chloro-4-fluorophenyl)ethyl)piperidin-4-yl)methoxy)- -5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
[1098] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-4-((1-(1-(2-chloro-4-fluorophenyl) ethyl)piperidin-4-yl)methoxy)-5-cyclopropyl-N-(ethylsulfonyl)-2-fluoroben- zamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 39.6.degree. C.; RT=3.31 min). LCMS(ESI) Method A: RT=5.85 min, m/z: 541.3 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 11.46 (s, 1H), 7.61 (dd, J=8.7, 6.5 Hz, 1H), 7.41 (dd, J=8.9, 2.6 Hz, 1H), 7.25 (m, 1H), 7.14 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.9 Hz, 1H), 3.92 (d, J=6.1 Hz, 3H), 3.40 (m, 2H), 3.18 (m, 1H), 2.77 (m, 1H), 2.15 (s, 1H), 2.01 (m, 2H), 1.84 (m, 2H), 1.72 (m, 1H), 1.40 (m, 1H), 1.29 (m, 4H), 1.21 (t, J=7.4 Hz, 3H), 0.85 (m, 2H), 0.64 (m, 2H).
Example 246
##STR00656##
[1099] (R)-4-((1-(1-(2-chloro-4-fluorophenyl)ethyl)piperidin-4-yl)methoxy)- -5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
[1100] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(2-chloro-4-fluorophenyl) ethyl)piperidin-4-yl)methoxy)-5-cyclopropyl-N-(ethylsulfonyl)-2-fluoroben- zamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical MeOH (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 40.degree. C.; RT=2.62. min). LCMS(ESI) Method A: RT=5.86 min, m/z: 541.3 [M+1].sup.+. .sup.1H NMR (400 DMSO-d.sub.6) .delta. 11.46 (s, 1H), 7.61 (dd, J=8.7, 6.5 Hz, 1H), 7.41 (dd, J=8.9, 2.6 Hz, 1H), 7.25 (m, 1H), 7.14 (d, J=8.4 Hz, 1H), 6.90 (d, J=2.9 Hz, 1H), 3.92 (d, J=6.1 Hz, 3H), 3.40 (m, 2H), 3.18 (m, 1H), 2.77 (m, 1H), 2.15 (m, 1H), 2.01 (m, 2H), 1.84 (m, 2H), 1.72 (m, 1H), 1.40 (m, 1H), 1.29 (m, 4H), 1.21 (t, J=7.4 Hz, 3H), 0.85 (m, 2H), 0.64 (m, 2H).
Example 247
##STR00657##
[1101] 5-cyclopropyl-2-fluoro-4-((1-(3-fluoro-4-(trifluoromethyl)piperidin- )piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1102] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.77 min, m/z: 547.2 [M+1].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.78 (m, 1H), 7.45 (m, 2H), 7.17 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 3.94 (d, J=5.9 Hz, 2H), 3.74 (s, 2H), 3.19 (s, 3H), 2.95 (m, 2H), 2.23 (m, 2H), 2.02 (m, 1H), 1.84 (m, 3H), 1.43 (m, 2H), 0.88 (m, 2H), 0.65 (m, 2H).
Example 248
##STR00658##
[1103] (S)-5-cyclopropyl-2-fluoro-4-((4-1-(1-(4-(trifluoromethoxy)phenyl)e- thyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1104] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-2-fluoro-4-((4-methyl-1-(1-(4-(trifluoromethoxy)phenyl)- ethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.1.degree. C.; RT=4.93 min). LCMS(ESI) Method A: RT=5.98 min, m/z: 573.3 [M+1].sup.+. .sup.1H NMR (b 400 MHz, DMSO-d.sub.6) .delta. 7.52 (d, J=8.1 Hz, 2H), 7.37 (d, J=8.1 Hz, 2H), 7.21 (d, J=5 Hz, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.79 (brs, 2H), 3.30 (m, 1H), 3.04 (s, 3H), 2.61 (m, 1H), 2.49 (S, 3H), 1.99 (m, 1H), 1.70 (m, 2H), 1.46 (m, 5H), 1.04 (s, 3H), 0.87 (m, 2H), 0.59 (m, 2H).
Example 249
##STR00659##
[1105] (R)-5-cyclopropyl-2-fluoro-4-((4-methyl-1-(1-(4-(trifluoromethoxy)p- henyl)ethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1106] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-2-fluoro-4-((4-methyl-1-(1-(4-(trifluoromethoxy) phenyl)ethyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature; 39.9.degree. C.; RT=2.52 min). LCMS(ESI) Method A: RT=5.95 min, m/z: 573.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.52 (m, 2H), 7.37 (m, 2H), 7.21 (d, J=8.5 Hz, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.79 (brs, 2H), 3.31 (m, 1H), 3.04 (s, 3H), 2.61 (m, 1H), 2.49 (s, 3H), 1.99 (m, 1H), 1.70 (m, 2H), 1.46 (m, 5H), 1.04 (s, 3H), 0.87 (m, 2H), 0.59 (m, 2H).
##STR00660##
Example 250
(R)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluorophenyl)ethyl-4-methylpiperidi- n-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1107] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluorophenyl) ethyl-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 min, 5 .mu.m; Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 38.8.degree. C.; RT=4.89 min). LCMS(ESI) Method A: RT =5.42 min, m/z: 507.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.43 (m, 1H), 7.29 (m, 2H), 7.23 (d, J =8.6 Hz, 1H), 7.16 (m, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.95 (m, 1H), 3.79 (s, 2H), 3.03 (s, 3H), 2.91 (m, 1H), 2.70 (m, 2H), 2.59 (m, 1H), 2.03 (m, 1H), 1.76 (m, 2H), 1.50 (m, 5H), 1.05 (s, 3H), 0.88 (m, 2H), 0.60 (m, 2H).
Example 251
##STR00661##
[1108] (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluorophenyl)ethyl-4-methylp- iperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
[1109] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-2-fluoro-4-((1-(1-(3-fluorophenyl) ethyl-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.6.degree. C.; RT=6.92 min). LCMS(ESI) Method A: RT =5.42 min, m/z: 507.3 [M+H].sup.+. .sup.1H NMR (400 DMSO-d.sub.6), .delta. 7.43 (m, 1H), 7.29 (m, 2H), 7.23 (d, J =8.6 Hz, 1H), 7.16 (m, 1H), 6.80 (d, J=12.8 Hz, 1H), 3.95 (m, 1H), 3.79 (s, 2H), 3.03 (s, 3H), 2.91 (m, 1H), 2.70 (m, 2H), 2.59 (m, 1H), 2.03 (m, 1H), 1.76 (m, 2H), 1.50 (m, 5H), 1.05 (s, 3H), 0.85 (m, 2H), 0.60 (m, 2H).
##STR00662##
Example 252
5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl)cyclo- propyl)methyl)piperidin-4-yl)methoxy)benzamide
[1110] The compound was synthesized as described in Example 88. LCMS(ESI) Method B: RT=5.42 min, m/z: 492.9 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6), .delta. 7.16 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.9 Hz, 1H), 3.94 (d, J=5.8 Hz, 2H), 3.24 (s, 3H), 3.01 (m, 2H), 2.59 (s, 2H), 2.03 (m, 3H), 1.79 (m, 3H), 1.37 (m, 2H), 0.99 (m, 2H), 0.88 (m, 2H), 0.76 (m, 2H), 0.66 (m, 2H).
Example 253
##STR00663##
[1111] 5-Cyclopropyl-4-((1-((3,5-dichlorobenzoyl)piperidin-4-yl)methoxy)-2- -fluoro-N-(methylsulfonyl)benzamide
[1112] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=4.92 min, m/z 542.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.71 (s, 1H), 7.45 (s, 2H), 7.15 (d, J=8.5 Hz, 1H), 6.92 (d, J=13.0 Hz, 1H), 4.49-4.47 (m, 1H), 3.97 (d, J=4.5 Hz, 2H), 3.53-3.50 (m, 1H), 3.26 (s, 3H), 3.15-3.12 (m, 1H), 2.85-2.84 (m, 1H), 2.14-2.10 (m, 1H), 2.05-1.99 (m, 1H), 1.89-1.88 (m, 1H), 1.78-1.75 (m, 1H), 1.35-1.32 (m, 2H), 0.89-0.88 (m, 2H), 0.66-0.65 (m, 2H).
##STR00664##
Example 254
5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,5-dichlorobenzoyl)piperidin- -4-yl)methoxy)-2-fluorobenzamide
[1113] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=5.28 min, m/z: 569.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 11.80 (s, 1H), 7.71 (s, 1H), 7.40 (m, 2H), 7.16 (d, J =8.0 Hz, 1H), 6.92 (d, J=12.1 Hz, 1H), 4.47 (m, 1H), 3.97 (s, 2H), 3.51 (m, 1H), 3.08 (m, 2H), 2.84 (m, 1H), 2.07 (m, 2H), 1.82 (m, 2H), 1.34 (m, 2H), 1.01 (m, 4H), 0.89 (m, 2H), 0.62 (m, 2H).
Example 255
##STR00665##
[1114] 5-Cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(2-(trifluoromethyl- )benzoyl)piperidin-4-yl)methoxy)benzamide
[1115] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=4.54 min, m/z: 542.9 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.82 (d, J=7.9 Hz, 1H), 7.75 (m, 1H), 7.67 (d, J=7.4 Hz, 1H), 7.46 (m, 1H), 7.31 (m, 1H), 6.82 (m, 1H), 4.77 1(m, 1H), 4.01 (d, J=5.2 Hz, 2H), 3.43 (m, 1H), 3.33 (s, 3H), 3.17 (m, 1H), 2.96 (m, 1H), 2.20 (m, 1H), 2.07 (m, 2H), 1.81 (m, 1H), 1.42 (m, 2H), 0.94 (m, 2H), 0.66 (m, 2H).
Example 256
##STR00666##
[1116] 5-Cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(2-(trifluorom- ethyl)benzoyl)piperidin-4-yl)methoxy)benzamide
[1117] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=4.69 min, m/z: 568.9 [M+H].sup.+. .sup.1H NMR (500 MHz, MeOD-d.sub.4) .delta. 7.82 (m, 1H), 7.76 (m, 1H), 7.67 (m, 1H), 7.46 (m, 1H), 7.30 (m, 1H), 6.82 (dd, J=12.8, 4.8 Hz, 1H), 4.77 (m, 1H), 4.01 (m, 2H), 3.43 (m, 1H), 3.17 (m, 2H), 2.96 (m, 1H), 2.20 (m, 1H), 2.02 (m, 2H), 1.81 (m, 1H), 1.47 (m, 2H), 1.29 (m, 2H), 1.13 (m, 2H), 0.92 (m, 2H), 0.66 (m, 2H).
Example 257
##STR00667##
[1118] 5-Cyclopropyl-4-((1-(2,4-dichlorobenzoyl)piperidin-4-yl)methoxy)-2-- fluoro-N-(methylsulfonyl)benzamide
[1119] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=4.82 min, m/z 542.8 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.73-7.72 (m, 1H), 7.53-7.49 (m, 1H), 7.43-7.37 (m, 1H), 7.15 (d, J=8.5 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 4.57-4.52 (m, 1H), 3.99-3.95 (m, 2H), 3.30-3.28 (m, 1H), 2.25 (s, 3H), 3.13-3.03 (m, 1H), 2.85-2.82 (m, 1H), 2.13-1.97 (m, 2H), 1.92-1.89 (m, 1H), 1.75-1.72 (m, 1H), 1.43-1.25 (m, 2H), 0.89-0.85 (m, 2H), 0.67-0.63 (m, 2H).
Example 258
##STR00668##
[1120] 5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(2,4-dichlorobenzoyl)pi- peridin-4-yl)methoxy)-2-fluorobenzamide
[1121] The compound was synthesized as described in Example 134. LCMS(ESI) Method A: RT=5.00 min, m/z 568.1 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.73 (s, 1H), 7.53-7.49 (m, 1H), 7.43-7.37 (m, 1H), 7.14 (d, J=8.5 Hz, 1H), 6.94 (d, J=13.0 Hz, 1H), 4.57-4.52 (m, 1H), 3.99-3.96 (m, 2H), 3.13-3.04 (m, 2H), 2.87-2.83 (m, 1H), 2.12-1.89 (m, 3H), 1.75-1.72 (m, 1H), 1.42-1.23 (m, 3H), 1.10-1.06 (m, 4H), 0.89-0.86 (m, 2H), 0.69-0.63 (m, 2H).
##STR00669##
Example 259
5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(3,5-dichlorobenzoyl)piperidin- -4-yl)methoxy)-2-fluorobenzamide
[1122] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.21 min, m/z 555.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.52 (s, 1H), 7.39 (s, 2H), 7.14 (d, J=8.0 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 3.94 (d, J=5.5 Hz, 2H), 3.62 (s, 2H), 3.05-2.90 (m, 1H), 2.91 (d, J=11.51 Hz, 2H), 2.16 (t, J=11.3 Hz, 2H), 2.03-1.99 (m, 1H), 1.82-1.79 (m, 3H), 1.43-1.37(m, 2H), 1.05-0.99 (m, 4H), 0.90-0.86 (m, 2H), 0.65-0.52 (m, 2H).
##STR00670##
Example 260
N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidi- n-4-yl)methoxy)-2-fluorobenzamide
[1123] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.47 min, m/z 570.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. 7.52 (s, 1H), 7.39 (s, 2H), 7.15 (d, J=8.0 Hz, 1H), 6.90 (d, J =12.5 Hz, 1H), 3.98-3.94 (m, 6H), 3.60 (s, 2H), 2.90 (d, J=11.0 Hz, 2H), 2.15-2.11 (m, 4H), 2.03-2.01 (m, 1H), 1.82-1.79 (m, 3H), 1.43-1.37 (m, 2H), 0.90-0.87 (m, 2H), 0.66-0.64 (m, 2H).
Example 261
##STR00671##
[1124] 4-((1-(3-Chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl)methoxy)-- 5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
[1125] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=6.34 min, m/z 593.3 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.48 (s, 2H), 7.35 (s, 1H), 7.14 (d, J=8.4 Hz, 1H), 6.90 (d, J=12.8 Hz, 1H), 3.95 (d, J=6.0 Hz, 2H), 3.67 (s, 2H), 3.40-3.32 (m, 1H), 2.92 (d, J=10.4 Hz, 2H), 2.20-2.15 (m, 2H), 2.03-1.98 (m, 1H), 1.82-1.79 (m, 3H), 1.45-1.37 (m, 2H), 1.21 (t, J=74 Hz, 3H), 0.90-0.86 (m, 2H), 0.65-0.62 (m, 2H).
Example 262
##STR00672##
[1126] (R)-4-((1-(1-(3-chlorophenyl)-2-methoxyethyl)piperidin-4-yl)methoxy- )-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1127] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(3-chlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamid- e. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75: 25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=3.05 min). LCMS(ESI) Method A: RT=5.47 min, m/z 538.9 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.41-7.30 (m, 4H), 7.15 (d, J=8.4 Hz, 1H), 6.86 (d, J=12.8 Hz, 1H), 3.89 (d, J=5.6 Hz, 2H), 3.81-3.78 (m, 1H), 3.73-3.71 (m, 2H), 3.23 (s, 3H), 3.18 (s, 3H), 3.09-3.06 (m, 1H), 2.87-2.84 (m, 1H), 2.22-2.16 (m, 1H), 2.10-2.06 (m, 1H), 2.03-2.96 (m, 1H), 1.81-1.73 (m, 3H), 1.45-1.26 (m, 2), 0.89-0.84 (m, 2H), 0.65-0.61 (m, 2H).
Example 263
##STR00673##
[1128] (S)-4-((1-(1-(3-chlorophenyl)-2-methoxyethyl)piperidin-4-yl)methoxy- )-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1129] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-4-((1-(1-(3-chlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamid- e. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=75: 25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=4.35 min). LCMS(ESI) Method A: RT=5.46 min, m/z 539.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.41-7.30 (m, 4H), 7.15 (d, J=8.4 Hz, 1H), 6.86 (d, J =12.8 Hz, 1H), 3.89 (d, J=5.6 Hz, 2H), 3.81-3.78 (m, 1H), 3.73-3.71 (m, 2H), 3.23 (s, 3H), 3.18 (s, 3H), 3.09-3.06 1(m, 1H), 2.87-2.84 (m, 1H), 2.22-2.16 (m, 1H), 2.10-2.06 (m, 1H), 2.03-2.96 (m, 1H), 1.81-1.73 (m, 3H), 1.45-1.26 (m, 2H), 0.89-0.84 (m, 2H), 0.65-0.61 (m, 2
Example 264
##STR00674##
[1130] (R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1131] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A: B=75: 25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=3.71 min). LCMS(ESI) Method A: RT=5.74 min, m/z 573.0 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sub.6): .delta. 7.60-7.58 (m, 2H), 7.32 (d, J=8.5 Hz, 1H), 7.15 (d, J=8.0 Hz, 1H), 6.83 (d, J=13.0 Hz, 1H), 3.89 (d, J=6 Hz, 2H), 3.74-3.68 (m, 3H), 3.22 (s, 3H), 3.15 (s, 3H), 3.01-3.00 (m, 1H), 2.82-2.78 (m, 1H), 2.14-2.09 (m, 1H), 2.02-1.96 (m, 2H), 1.79-1.71 (m, 3H), 1.38-1.28 (m, 1H), 0.88-0.85 (m, 2H), 0.63-0.60 (m, 2H).
##STR00675##
Example 265
(S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)-2-methoxyethyl)piperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1132] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A: B=75: 25; flow: 3 mL/min; column temperature: 40.degree. C.; RT=5.12 min). LCMS(ESI) Method A: RT=5.71 min, m/z 573.0 [M+H].sup.+. .sup.1H NMR (500 DMSO-d.sub.6): .delta. 7.60-7.58 (m, 2H), 7.32 (d, J=8.5 Hz, 1H), 7.15 (d, J=8.0 Hz, 1H), 6.83 (d, J=13.0 Hz, 4H), 3.89 (d, J=6 Hz, 2H), 3.74-3.68 (m, 3H), 3.22 (s, 3H), 3.15 (s, 3H), 3.01-3.00 (m, 1H), 2.82-2.78 (m, 1H), 2.14-2.09 (m, 1H), 2.02-1.96 (m, 2H), 1.79-1.71 (m, 3H), 1.38-1.28 (m, 2H), 0.88-0.85 (m, 2H), 0.63-0.60 (m, 2H).
Example 266
##STR00676##
[1133] ((R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)pip- eridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00677##
##STR00678##
[1134] Step 1
Methyl 2-bromo-2-(3,5-dichlorophenyl)acetate
[1135] A mixture of methyl 2-(3,5-dichlorophenyl)acetate (2.0 g, 9.11 mol), N-bromosuccinimide (6.5 g, 36.51 mmol) and 2,2'-azobis(2-methylpropionitrile) (600 mg, 3.64 mmol) in carbon tetrachloride (50 mL) was stirred at 80.degree. C. for 16 h. The reaction mixture was diluted with dichloromethane (200 mL) and brine (50 mL). The organic layer was separated, washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 10% ethylacetate in petroleum ether) to afford the target compound (2.5 g, 92%) as a pale yellow oil.
##STR00679##
Step 2
Methyl 2-(4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)- methyl)piperidin-1-yl)-2-(3,5-dichlorophenyl)acetate
[1136] The compound was synthesized as described in step 5 of Example 88. LCMS(ESI) m/z: 587.2 [M+H].sup.+.
Step 3
##STR00680##
[1137] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1138] A mixture of methyl 2-(4-((2-cyclopropyl-5-fluoro-4-(methysulfonylcarbamoyl)phenoxy)methyl)pi- peridin-1-yl)-2-(3,5-dichlorophenyl) acetate (200 mg, 0.37 mmol) and sodium borohydride (142 mg, 3.7 mmol) in MeOH (5 mL) was stirred at 25.degree. C. for 2 h. The mixture was then concentrated, diluted with water (20 mL) and extracted with DCM (20 mL.times.3). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase chromatography (eluting with 30-40% MeCN in 0.1% NH.sub.4HCO.sub.3) to afford the racemate (100 mg, 48.3%) as an white solid. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)piperidin-- 4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OJ-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80: 20; flow: 3 mL/min; column temperature: 40.degree. C.; RT=5.25 min). LCMS(ESI) Method A: RT=5.28 min, m/z 559.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.52 (s, 1H), 7.42 (s, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.85 (d, J=12.8 Hz, 1H), 4.77 (brs, 1H), 3.90 (d, J=5.6 Hz, 2H), 3.78 (s, 2H), 3.65-3.61 (m, 1H), 3.17-3.11 (m, 4H), 2.86-2.85 (m, 1H), 2.20-1.97 (m, 3H), 1.82-1.74 (m, 3H), 1.44-1.16 (m, 2H), 0.89-85 (m, 2H), 0.65-0.61 (m, 2H).
Example 267
##STR00681##
[1139] (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1140] The compound was synthesized as described in Example 266. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: OH-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A: B=80:20; flow: 3 mL/min; column temperature: 40.degree. C.; RT=6.45 min). LCMS(ESI) Method A: RT=5.35 min, m/z 559.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.52 (s, 1H), 7.42 (s, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.85 (d, J=12.8 Hz, 1H), 4.77 (br, 1H), 3.90 (d, J=5.6 Hz, 2H), 3.78 (s, 2H), 3.65-3.61 (m, 1H), 3.17-3.11 (m, 4H), 2.86-2.85 (m, 1H), 2.20-1.97 (m, 3H), 1.82-1.74 (m, 3H), 1.44-1.16 (m, 2H), 0.89-85 (m, 2H), 0.65-0.61 (m, 2H).
Example 268
##STR00682##
[1141] 4-((1-(3-Chloro-5-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropy- l-2-fluoro-N-(methylsulfonyl)benzamide
[1142] The compound was synthesized as described in Example 88. LCMS(ESI) Method A: RT=5.75 min, m/z: 513.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6): .delta. 7.35-7.34 (m, 1H), 7.28-7.27 (m, 1H), 7.20-7.16 (m, 2H), 6.86-6.83 (m, 1H), 3.94-3.92 (d, J=5.0 Hz, 2H), 3.63 (s, 2H), 3.13 (s, 3H), 2.94-2.91 (m, 2H), 2.19-2.15 (m, 2H), 2.01 (m, 1H), 1.82-1.80 (m, 3H), 1.42-1.40 (m, 2H), 0.89-0.86(m, 2H), 0.64-0.62 (m, 2H).
##STR00683##
Example 269
(R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)eth- yl)piperidin-4-yl)methoxy)-2-fluorobenzamide
##STR00684##
[1143] Step 1
##STR00685##
[1144] (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-- 2-fluorobenzoate
[1145] The compound was synthesized as described in step 5 of Example 88. The enantiomer was separated by chiral SFC from the racemate, the enantiomer was arbitrarily assigned as (R)-methyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzoate. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOH (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 39.9.degree. C.; RT=4.49 min). LCMS(ESI) m/z: 480.1 [M+H].sup.+.
Step 2
##STR00686##
[1146] (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl- )methoxy)-2-fluorobenzoic acid
[1147] The compound was synthesized as described in step 6 of Example 88. LCMS(ESI) m/z: 460.0 [M+H].sup.+.
##STR00687##
Step 3
(R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)eth- yl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[1148] The compound was synthesized as described in step 5 of Example 80. The enantiomer was arbitrarily assigned as (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide. LCMS(ESI) Method A: RT=6.52 min, m/z: 569.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.52 (m, 1H), 7.41 (m, 2H), 7.15-7.13 (d, J=5.0 Hz, 1H), 6.91-6.88 (d, J=13.0 Hz, 1H), 3.93-3.73 (m, 2H), 3.72-3.70 (m, 1H), 3.05-3.03 (m, 2H), 2.89-2.87 (m, 1H), 2.36-2.00 (m, 3H), 1.84-1.77 (m, 3H), 1.42-1.30 (m, 5H), 1.06-1.02 (m, 4H), 0.91-0.83 (m, 2H), 0.71-0.62 (m, 2H).
##STR00688##
Example 270
(S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)eth- yl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[1149] The compound was synthesized as described in Example 269. The enantiomer was arbitrarily assigned as (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide. LCMS(ESI) Method A: RT=6.51 min, m/z: 569.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.52 (m, 1H), 7.41 (m, 2H), 7.15-7.13 (d, J=5.0 Hz, 1H), 6.90-6.88 (d, J=13.0 Hz, 1H), 3.93-3.73 (m, 2H), 3.72-371 (m, 1H), 3.05-3.03 (m, 2H), 2.89-2.87 (m, 1H), 2.21-1.98 (m, 3H), 1.84-1.77 (m, 3H), 1.42-1.30 (m, 5H), 1.29-0.98 (m, 4H), 0.91-0.83 (m, 2H), 0.71-0.62 (m, 2H).
Example 271
##STR00689##
[1150] (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorop- henyl)ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[1151] The compound was synthesized as described in Example 269. The enantiomer was arbitrarily assigned as (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide. LCMS(ESI) Method A: RT=6.66 min, m/z: 584.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.49 (s, 1H), 7.39-7.38 (d, J=1.0 Hz, 2H), 7.19-7.17 (d, J=7.5 Hz, 1H), 6.86-6.83 (d, J=13.0 Hz, 1H), 3.91-3.90 (m, 6H), 3.64-3.63 (m, 1H), 2.98 (m, 1H), 2.84-2.82 (m, 1H), 2.11-1.98 (m, 5H), 1.82-1.75 (m, 3H), 1.36-1.31 (m, 5H), 0.88-0.86 (m, 2H), 0.63-0.62 (m, 2H).
##STR00690##
Example 272
(S)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide
[1152] The compound was synthesized as described in Example 269. The enantiomer was arbitrarily assigned as (S)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzamide. LCMS(ESI) Method A: RT=6.67 min, m/z: 584.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta. 7.49 (s, 1H), 7.39-7.38 (d, J=1.0 Hz, 2H), 7.19-7.17 (d, J=7.5 Hz, 1H), 6.83-6.80 (d, J=13.0 Hz, 1H), 3.91-3.86 (m, 6H), 3.64-3.63 (m, 1H), 2.98 (m, 1H), 2.82-2.80 (m, 1H), 2.11-1.98 (m, 5H), 1.82-1.75 (m, 3H), 1.36-1.31 (m, 5H), 0.88-0.86 (m, 2H), 0.63-0.62 (m, 2H).
Example 273
##STR00691##
[1153] 5-Cyclopropyl-4-((1-((3,5-dichlorophenyl)sulfonyl)piperidin-4-yl)me- thoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1154] The compound was synthesized as described in Example 129. LCMS(ESI) Method A: RT=5.54 min, m/z: 579.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.87 (brs, 1H), 8.13 (s, 1H), 8.04 (d, J=5.0 Hz, 2H), 7.14 (d, J=8.5 Hz, 1H), 6.90 (d, J=10.0 Hz, 1H), 3.94 (d, J=5 Hz, 2H), 3.75-3.74 (m, 2H), 3.26-3.22 (m, 3H), 3.46-3.36 (m, 2H), 1.99-1.85 (m, 4H), 1.42-1.35 (m, 2H), 0.89-0.83 (m, 2H), 0.67-0.62 (m, 2H).
##STR00692##
Example 274
5-Cyclopropyl-4-((1-((3,5-dichlorophenyl)sulfonyl)azetidin-3-yl)methoxy)-2- -fluoro-N-(methylsulfonyl)benzamide
[1155] The compound was synthesized as described in Example 129. LCMS(ESI) Method A: RT=5.01 min, m/z: 551.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.11.90 (brs, 1H), 8.09 (s, 1H), 7.84-7.83 (d, J=1.5 Hz, 2H), 7.15-7.13 (d, J=8.5 Hz, 1H), 6.89-6.87 (d, J=13.0 Hz, 1H), 4.08-4.06 (m, 2H), 3.96-3.92 (m, 2H), 3.78-3.75 (m, 2H), 3.24 (s, 3H), 3.02-2.96 (m, 1H), 1.81-1.76 (m, 1H), 0.83-0.79 (m, 2H), 0.63-0.61 (m, 2H).
##STR00693##
Example 275
5-Cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-((3,5-dichlorophenyl)sulfonyl)- piperidin-4-yl)methoxy)-2-fluorobenzamide
[1156] The compound was synthesized as described in Example 129. LCMS(ESI) Method A: RT=5.68 min, m/z: 604.8 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.8.05 (s, 1H), 7.76 (d, J=2.5 Hz, 2H), 7.14-7.12 (d, J=8.5 Hz, 1H), 6.91-6.88 (d, J=13.0 Hz, 1H), 3.94-3.93 (d, J=5.0 Hz, 2H), 3.75-3.73 (m, 2H), 3.05-3.02 (m, 1H), 2.50-2.42 (m, 2H), 1.98-1.85 (m, 4H), 1.42-1.37 (m, 2H), 0.87-0.85 (m, 4H), 0.66-0.64 (m, 2H), 0.62-0.56 (m, 2H).
Example 276
##STR00694##
[1157] (R)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl)etnyl)piperidin-4-- yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1158] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-4-((1-(1-(3-chloro-5-(trifluoromethyl) phenyl)ethyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulf- onyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.1.degree. C.; RT=5.80 min). LCMS(ESI) Method A: RT=6.43 min, m/z: 577.0 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.80 (s, 1H), 7.73 (s, 2H), 7.34-7.32 (m, 1H), 6.67-6.64 (d, J=12.8 Hz, 1H), 4.05-4.03 (m, 1H), 3.86-3.85 (m, 2H), 3.44-3.42 (m, 1H), 3.21 (s, 3H), 3.11-3.08 (m, 1H), 2.49-2.44 (m, 2H), 2.05-1.93 (m, 4H), 1.66-1.54 (m, 5H), 0.91-0.87 (m, 2H), 0.83-0.80 (m, 2H).
##STR00695##
Example 277
(S)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl)ethyl)piperidin-4-yl)meth- oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1159] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-4-((1-(1-(3-chloro-5-(trifluoromethyl) phenyl)ethyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulf- onyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.1.degree. C.; RT=7.02 min). LCMS(ESI) Method A: RT=6.42 min, m/z: 577.0 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.79 (s, 1H), 7.74 (s, 2H), 7.34-7.32 (m, 1H), 6.71-6.65 (m, 1H), 4.11-4.05 (m, 1H), 3.88-3.83 (m, 2H), 3.46-3.45 (m, 1H), 3.23 (s, 3H), 3.15-3.11 (m, 1H), 2.51-2.48 (m, 2H), 2.04-1.93 (m, 4H), 1.69-1.51 (m, 5H), 0.91-0.86 (m, 2H), 0.73-0.68 (m, 2H).
##STR00696##
Example 278
(R)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl)ethyl)-4-methylpiperidin-- 4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1160] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl) ethyl)-4-methylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsu- lfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical MeOD (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.0.degree. C.; RT=5.35 min). LCMS(ESI) Method A: RT=6.42 min, m/z: 591.2 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.77-7.72 (m, 3H), 7.36-7.34 (d, J=8.8 Hz, 1H), 6.76-6.68 (m, 1H), 4.00-3.95 (m, 1H), 3.81-3.78 (s, 2H), 3.23-3.22 (s, 3H), 3.10-3.02 (m, 1H), 2.78-2.67 (m, 3H), 2.08-2.02 (m, 1H), 1.94-1.84 (m, 2H), 1.68-1.55 (m, 5H), 1.16 (s, 3H), 0.92-0.88 (m, 2H), 0.67-0.63 (m, 2H).
##STR00697##
Example 279
(S)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl)ethyl)-4-methylpiperidin-- 4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[1161] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-4-((1-(1-(3-chloro-5-(trifluoromethyl)phenyl)ethyl)-4-methylpiperidin- -4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AD-H, 4.6.times.250 min, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=80:20; flow: 2.4 mL/min; column temperature: 40.0.degree. C.; RT=4.69 min). LCMS(ESI) Method A: RT=6.42 min, m/z: 591.3 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.79-7.72 (m, 3H), 7.36-7.34 (d, J=8.8 Hz, 1H), 6.76-6.68 (m, 1H), 4.00-3.95 (m, 1H), 3.81-3.78 (s, 2H), 3.23-3.22 (s, 3H), 3.07-3.05 (m, 1H), 2.78-2.67 (m, 3H), 2.08-2.02 (m, 1H), 1.94-1.84 (m, 2H), 1.69-1.53 (m, 5H), 1.16 (s, 3H), 0.92-0.88 (m, 2H), 0.67-0.63 (m, 2H)
Example 280
##STR00698##
[1162] (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)-4-methylpiper- idin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1163] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl) ethyl)-4-methylpiperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzami- de. Chiral HPLC (column: AD-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=70:30; flow: 2.4 mL/min; column temperature: 39.9.degree. C.; RT=3.89 min). LCMS(ESI) Method A: RT=6.46 min, m/z: 557.2 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.68-7.66 (d, J=11.5 Hz, 1H), 7.57-7.56 (d, J=2.5 Hz, 1H), 7.46-7.42 (m, 1H), 7.38-7.36 (d, J=8.5 Hz, 1H), 6.75-6.72 (d, H=13.0 Hz, 1H), 4.47-4.46 (m, 1H), 3.82 (s, 2H), 3.20-3.17 (m, 4H), 2.90-2.79 (m, 3H), 2.06-2.04 (m, 1H), 1.94-1.88 (m, 2H), 1.66 (s, 2H), 1.54-1.53 (d, J=7.0 Hz, 3H), 1.18 (s, 3H), 0.93-0.89 (m, 2H), 0.67-0.63 (m, 2H).
##STR00699##
Example 281
(R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)-4-methylpiperidin-4-- yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1164] The compound was synthesized as described in Example 170. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl) ethyl)-4-methylpiperidin-4-yl)methoxy)-2-fluoro-N-(methyl)sulfonyl)benzam- ide. Chiral HPLC (column: AD-H, 4.6.times.250 min, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=70:30; flow: 2.4 mL/min; column temperature: 39.9.degree. C.; RT=5.67 min). LCMS(ESI) Method A: RT=6.40 min, m/z: 557.2 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta. 7.70-7.68 (d, J=11.5 Hz, 1H), 7.57-7.56 (d, J=2.5 Hz, 1H), 7.47-7.45 (m, 1H), 7.36-7.34 (d, J=8.5 Hz, 1H), 6.72-6.69 (d, J=13.0 Hz, 1H), 4.47-4.46 (m, 1H), 3.82 (s, 2H), 3.20-3.17 (m, 4H), 2.88-2.82 (m, 3H), 2.06-2.04 (m, 1H), 1.96-1.85 (m, 2H), 1.66 (s, 2H), 1.54-1.53 (d, J=7.0 Hz, 3H), 1.18 (s, 3H), 0.93-0.89 (m, 2H), 0.67-0.63 (m, 2H).
Example 282
##STR00700##
[1165] (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1166] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.0.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.3.degree. C.; RT=2.95 min). LCMS(ESI) Method A: RT=6.00 min, m/z: 573.1 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.7.69-7.66 (m, 1H), 7.55-7.54 (d, J=2.0 Hz, 1H), 7.41-7.39 (m, 1H), 7.33-7.31 (d, J=8.4 Hz, 1H), 6.75-6.71 (m, 1H), 4.41 (m, 1H), 3.94-3.92 (m, 2H), 3.77 (s, 2H), 3.43-3.32 (m, 4H), 3.24 (s, 3H), 2.98 (m, 1H), 2.55-2.41 (m, 2H), 2.08-1.80 (m, 4H), 1.67-1.51 (m, 2H), 0.93-0.89 (m, 2H), 0.66-0.64 (d, J=4.8 Hz, 2H).
##STR00701##
Example 283
(R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl)piperidin-4- -yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
[1167] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.3.degree. C.; RT=3.85 min). LCMS(ESI) Method A: RT=6.01 min, m/z: 573.1 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.7.69-7.66 (m, 1H), 7.55-7.54 (d, J=2.0 Hz, 1H), 7.41-7.39 (m, 1H), 7.33-7.31 (d, J=8.4 Hz, 1H), 6.75-6.71 (m, 1H), 4.21 (m, 1H), 3.81-3.78(m, 2H), 3.77 (s, 2H), 3.43-3.32 (m, 4H), (s, 3H), 2.98 (m, 1H), 2.55-2.41 (m, 2H), 2.08-1.86 (m, 4H), 1.67-1.51 (m, 2H), 0.93-0.89 (m, 2H), 0.66-0.64 (m, 2H).
Example 284
##STR00702##
[1168] (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)piperidin-4-yl- )methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1169] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m, mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 37.7.degree. C.; RT=4.46 min). LCMS(ESI) Method A: RT=6.28 min, m/z: 557.1 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.7.69-7.64 (m, 1H), 7.55-7.54 (m, 1H), 7.44-7.42 (d, J=8.4 Hz, 1H), 7.31-7.29 (d, J=8.4 Hz, 1H), 6.73-6.70 (d, J=12.8 Hz, 1H), 4.36-4.35 (m, 1H), 3.92-3.86 (m, 2H), 3.53-3.50 (m, 1H), 3.34-3.32 (m, 2H), 3.06-3.03 (m, 1H), 2.49-2.41 (m, 2H), 2.08-1.89 (m, 4H), 1.68-1.65 (m, 1H), 1.54-1.49 (m, 4H), 1.38-1.33 (s, 3H), 0.93-0.82 (m, 2H), 0.61-0.59 (m, 2H).
##STR00703##
Example 285
(S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)ethyl)piperidin-4-yl)methox- y)-N-(ethylsulfonyl)-2-fluorobenzamide
[1170] The compound was synthesized as described in Example 88. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 37.7.degree. C.; RT=2.99 min). LCMS(ESI) Method A: RT=6.29 min, m/z: 557.2 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.7.71-7.65 (m, 1H), 7.56-7.55 (m, 1H), 7.46-7.42 (d, J=8.4 Hz, 1H), 7.31-7.30 (d, J=8.4 Hz, 1H), 6.73-6.70 (d, J=12.8 Hz, 1H), 4.49 (m, 1H), 3.89-3.86 (m, 2H), 3.58-3.55 (m, 1H), 3.34-3.32 (m, 2H), 3.06-3.03 (m, 1H), 2.49-2.41 (m, 2H), 2.08-1.89 (m, 4H), 1.68-165 (m, 1H), 1.54-1.49 (m, 4H), 1.38-1.33 (s, 3H), 0.93-0.82 (m, 2H), 0.61-0.59 (m, 2H).
Example 286
##STR00704##
[1171] (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1172] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40..degree. C.; RT=2.87 min). LCMS(ESI) Method A: RT=6.10 min, m/z: 587.0 [M+H].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.7.58-7.54 (m, 2H), 7.43-7.40 (m, 1H), 7.16-7.13 (d, J=10.5 Hz, 1H), 6.86-6.83 (d, J=12.8 Hz, 1H), 4.05-4.02 (m, 1H), 3.90-3.88 (m, 2H), 3.68-3.58 (m, 2H), 3.29-3.28 (m, 2H), 3.19 (s, 3H), 3.10-3.08 (m, 1H), 2.73-2.69 (m, 1H), 2.19-2.13 (m, 1H), 2.03-1.97 (m, 2H), 1.81-1.67 (m, 3H), 1.35-1.18 (m, 5H), 0.95-0.82 (m, 2H), 0.72-0.61 (m, 2H).
Example 287
##STR00705##
[1173] (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl)pipe- ridin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1174] The compound was synthesized as described in Example 172. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(2,4-dichlorophenyl)-2-methoxyethyl) piperidin-4-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide. Chiral HPLC (column: AS-H, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=75:25; flow: 2.25 mL/min; column temperature: 40.5.degree. C.; RT=3.66 min). LCMS(ESI) Method A: RT=6.10 min, m/z: 587.0 [M+H].sup.+. .sup.1H-NMR (400 MHz, DMSO-d.sub.6): .delta.7.58-7.54 (m, 2H), 7.43-7.40 (m, 1H), 7.16-7.14(d, J=10.5 Hz, 1H), 6.86-6.82 (d, J=12.8 Hz, 1H), 4.05-4.02 (m, 1H), 3.90-3.88 (m, 2H), 3.67-3.58 (m, 2H), 3.30-3.28 (m, 2H), 3.19 (s, 3H), 3.10-3.08 (m, 1H), 2.71-2.69 (m, 1H), 2.18-2.13 (m, 1H), 2.03-1.97 (m, 2H), 1.81-1.67 (m, 3H), 1.35-1.18 (m, 5H), 0.89-0.81 (m, 2H), 0.61-0.59 (m, 2H).
##STR00706##
Example 288
(R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazetidin-3-y- l)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1175] The compound was synthesized as described in Example 68. The enantiomer was separated by chiral SFC from the racemate, the first eluting fraction was arbitrarily assigned as (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-3-methylazetidin-3-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide- . Chiral HPLC (column: IC, 4.6.times.150 mm, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 39.9.degree. C.; RT=2.73 min). LCMS(ESI) Method A: RT=6.04 min, m/z: 543.1 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.7.42-7.33 (m, 4H), 6.83-6.76 (m, 1H), 4.10-4.04 (m, 2H), 3.81 (m, 1H), 3.64-3.45 (m, 2H), 3.43-3.36 (m, 2H), 3.36-3.35 (m, 1H), 3.12-3.22 (m, 1H), 2.09-2.06 (m, 1H), 1.47 (s, 3H), 1.39-1.30 (m, 6H), 0.94-0.92 (m, 2H), 0.70-0.65 (m, 2H).
##STR00707##
Example 289
(S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-3-methylazetidin-3-y- l)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
[1176] The compound was synthesized as described in Example 68. The enantiomer was separated by chiral SFC from the racemate, the second eluting fraction was arbitrarily assigned as (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-3-methylazetidin-3-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide- . Chiral HPLC (column: IC, 4.6.times.150 mm, 4.6.times.250 mm, 5 .mu.m; mobile Phase: A: supercritical CO.sub.2, B: MeOD (0.1% DEA), A:B=65:35; flow: 1.95 mL/min; column temperature: 39.9.degree. C.; 3.22 min). LCMS(ESI) Method A: RT=6.04 min, m/z: 543.1 [M+H].sup.+. .sup.1H-NMR (400 MHz, MeOD-d.sub.4): .delta.57.40-7.33 (m, 4H), 6.84-6.76 (m, 1H), 4.10-4.04 (m, 2H), 3.72 (m, 1H), 3.55-3.43 (m, 2H), 3.45-3.33 (m, 2H), 3.36-3.35 (m, 1H), 3.12-3.22 (m, 1H), 2.09-2.06 (m, 1H), 1.47 (s, 3H), 1.39-1.30 (m, 6H), 0.94-0.92 (m, 2H), 0.71-0.65 (m, 2H).
##STR00708##
Example 290
5-Cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-phenylpiperidin-4-yl)- methoxy)benzamide
[1177] The compound was synthesized as described in Example 79. LCMS(ESI) Method A: RT=5.51 min, m/z: 473.1 [M+H].sup.+. .sup.1H-NMR (500 DMSO-d.sub.6): .delta.7.27-7.23 (m, 1H), 7.25-7.23 (d, J=7 Hz, 2H), 7.04-7.03 (d, J=8.0 Hz, 2H), 6.87-6.84 (m, 1H), 6.78-6.75 (d, J=12.5 Hz, 1H), 4.00-3.99 (d, J=6.0 Hz, 2H), 3.77-3.75 (m, 2H), 3.26 (s, 1H), 2.82-1.72 (m, 2H), 2.09-1.99 (m, 4H), 1.71-1.65 (m, 2H), 1.06-1.05 (m, 2H), 0.93-0.66 (m, 2H), 0.65 (m, 1H).
##STR00709##
Example 291
N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-phenylpiperidin-4-y- l)methoxy)benzamide
[1178] The compound was synthesized as described in Example 79. LCMS(ESI) Method A: RT=5.40 min, m/z: 488.0 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.7.23-7.18 (m, 3H), 6.95-6.94 (m, 2H), 6.75-6.69 (m, 2H), 3.93-3.92 (d, J=6.0 Hz, 2H), 3.75-3.73 (m, 2H), 3.68-3.65 (m, 2.72-2.68 (m, 2H), 2.01-1.89 (m, 6H), 1.49-1.46 (m, 2H), 0.87-0.84 (m, 2H), 0.54-0.52 (m, 2H).
##STR00710##
Example 292
5-Cyclopropyl-4-((1-(3,5-dichlorophenyl)piperidin-4-yl)methoxy)-2-fluoro-N- -(methylsulfonyl)benzamide
[1179] The compound was synthesized as described in Example 79. LCMS(ESI) Method A: RT=6.07 min, m/z: 514.8 [M+H].sup.+. .sup.1H-NMR (500 MHz, CDCl.sub.3-d.sub.4): .delta.7.20-7.19 (d, J=8.5 Hz, , 1H), 6.93 (d, J=1.5 2H), 6.82-6.79 (m, 2H), 3.95-3.94 (d, J=6.5 Hz, 2H), 3.86-3.83 (m, 2H), 3.04 (s, 3H), 2.82 (m, 2H), 1.98 (m, 2H), 1.84-1.82 (m, 2H), 1.43 (m, 2H), 0.86-0.84 (m, 2H), 0.60-0.59 (m, 2H).
##STR00711##
Example 293
4-((1-(2-Chlorophenyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(me- thylsulfonyl)benzamide
[1180] The compound was synthesized as described in Example 79. LCMS(ESI) Method A: RT=5.53 min, m/z: 480.9 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.7.41-7.39 (m, 1H), 7.39-7.27 (m, 1H), 7.21-7.16 (m, 2H), 7.04-7.01 (m, 1H), 6.85-6.82 (m, 1H), 4.00-3.99 (d, J=5.5 Hz, 2H), 3.32 (m, 2H), 3.05-3.01 (m, 3H), 2.71-2.54 (m, 2H), 2.05-1.90 (m, 4H), 1.58-1.50 (m, 2H), 0.91-0.89 (m, 2H), 0.62-0.61 (m, 2H).
##STR00712##
Example 294
4-((1-(3-Chlorophenyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(me- thylsulfonyl)benzamide
[1181] The compound was synthesized as described in Example 79. LCMS(ESI) Method A: RT=5.55 min, m/z: 480.9 [M+H].sup.+. .sup.1H-NMR (500 MHz, DMSO-d.sub.6): .delta.7.20-7.17 (m, 2H), 6.94-6.90 (m, 3H), 6,74-6.72 (d, J=7.5 Hz 1H), 3.97-3.95 (d, J=6.5 Hz, 2H), 3.80-3.78 (m, 2H), 3.12-3.11 (s, 3H), 2.78-2.74 (m, 2H), 2.00-1.98 (m, 2H), 1.87-1.84 (m, 2H), 1.45-1.43 (m, 2H), 0.87-0.85 (m, 2H), 0.62-0.61 (m, 2H).
Example 295
Synthesis of 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00713##
[1182] Step 1. Preparation of methyl 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)ethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate and methyl 4-(((R)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy-5-cyclopropyl- -2-fluorobenzoate
##STR00714##
[1184] Following the procedure as described in Example 50 step 1, and making variations as required to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 2-chloro-1-(1-chloroethyl)-4-fluorobenzene. The first eluting fraction was arbitrarily assigned as methyl 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)ethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate was obtained as a colorless solid (0.17 g, 15%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.50-7.42 (m, 2H), 7.06 (dd, J=2.6 Hz, 8.6 Hz, 1H), 6.87 (dt, J=1.5 Hz, 8.4 Hz, 1H), 6.52 (d, J=12.9 Hz, 1H), 4.38-4.36 (m, 1H), 3.92 (q, J=6.6 Hz, 1H), 3.88 (s, 3H), 3.07-3.04 (m, 1H), 2.55-2.51 (m, 1H), 2.39-2.28 (m, 2H), 2.13-2.01 (m, 2H), 1.84-1.77 (m, 1H), 1.67-1.56 (m, 3H), 1.27 (d, J=6.5 Hz, 3H), 0.96-0.89 (m, 2H), 0.69-0.65 (m, 2H); MS(ES+) m/z 450, 452 (M+1).
[1185] The second eluting fraction was arbitrarily assigned as methyl 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl) ethyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate as a colorless solid (0.26 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.56-7.51 (m, 1H), 7.38 (d, J=8.4 Hz, 1H), 7.06-6.95 (m, 2H), 6.46(d, J=13.0 Hz, 1H), 4.32-4.26 (m, 1H), 3.94 (q, J=6.6 Hz, 1H), 3.86 (s, 3H), 2.96-2.89 (m, 2H), 2.39-1.83 (m, 6H), 1.68-1.45 (m, 2H), 1.27 (d, J=6.6 Hz, 3H), 0.90-0.84 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 450.2, 452.2 (M+1).
Step 2. Preparation of 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoic acid
##STR00715##
[1187] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)- piperidin-3-yl)oxy)-2-fluorobenzoate with methyl 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.04 g, 24%): MS(ES+) m/z 436.2, 438.2 (M+1); MS(ES-) m/z 434.3, 436.3 (M-1).
Step 3. Preparation of 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl) ethyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzam- ide
##STR00716##
[1189] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.01. g, 32%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (br s, 1H), 7.51 (dd, J=6.6 Hz, 8.6 Hz, 1H), 7.37 (dd, J=2.6 Hz, 8.9 Hz, 1H), 7.14 (d, J=8.4 Hz, 1H), 7.05 (dt, J=2.6 Hz, 8.6 Hz, 1H), 6.92 (d, J=13.1 Hz, 1H), 4.56-4.55 (m, 1H), 3.91-3.89 (m, 1H), 3.32 (s, 3H), 2.78-2.74 (m, 2H), 2.47-2.42 (m, 2H), 2.15-2.06 (m, 1H), 1.87-1.77 (m, 2H), 1.59-1.46 (m, 2H), 1.23 (d, J=6.4 Hz, 3H), 0.93-0.90 (m, 2H), 0.70-0.67 (m, 2H); MS(ES+) m/z: 513.2, 515.1 (M+1); MS(ES-) m/z 511.2, 513.2 (M-1).
Example 296
Synthesis of 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00717##
[1190] Step 1. Preparation of 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)ethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00718##
[1192] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl)oxy)-2-fluorobenzoate with methyl 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a beige color solid (0.23 g, 99%): MS(ES+) m/z 436.2, 438.1 (M+1); MS(ES-) m/z 434.2, 436.2 (M-1).
Step 2. Preparation of 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00719##
[1194] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((R)-1-(R)-1-(2-chloro-4-fluorophenyl)ethyl)piperidin-3-yl)oxy)-5-cycl- opropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.17 g, 48%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 7.61 (dd, J=6.5 Hz, 8.7 Hz, 1H), 7.38 (dd, J=2.4 Hz, 8.8 Hz, 1H), 7.22 (dt, J=2.6 Hz, 8.5 Hz, 1H), 7.09 (d, J=8.4 Hz, 1H), 6.91 (d, J=13.1 Hz, 1H), 4.55-4.50 (m, 1H), 3.92 (q, J=6.3 Hz, 1H), 3.32 (s, 3H), 2.77-2.74 (m, 2H), 2.36-2.19 (m, 2H), 2.08-1.94 (m, 2H), 1.81-1.75 (m, 1H), 1.59-1.46 (m, 2H), 1.23 (d, J=6.6 Hz, 3H), 0.89-0.85 (m, 2H), 0.70-0.65 (m, 2H); MS(ES+) m/z 513.2, 515.1 (M+1); MS(ES-) m/z 511.2, 513.2 (M-1).
Example 297 and Example 298
Synthesis of 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin- -3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00720##
[1195] And 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl- )piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00721##
[1196] Step 1. Preparation of 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate
##STR00722##
[1198] To a solution of 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethanol (1.14 g, 5.00 mmol) and 2,6-dimethylpyridine (0.86 g, 8.00 mmol) in cyclohexane (10 mL) was added dropwise trifluoromethanesulfonic anhydride (2.12 g, 7.50 mmol) at 10.degree. C. The reaction mixture was stirred at ambient temperature for 5 hours, and diluted with hexanes (200 mL). The organic layer was washed with 1N aqueous hydrochloric acid solution (30 mL), 25% aqueous ammonium chloride solution (2.times.40 mL); dried over anhydrous sodium sulfate and concentrated in vacuo to afford the title compound as a colorless oil (1.30 g, 72%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.66-7.61 (m, 1H), 7.26-7.22 (m, 1H), 7.19-7.12 (m, 1H), 6.46 (q, J=5.6 Hz, 1H).
Step 2. Preparation of methyl 4-(((3R)-1-(1-(2 -chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate
##STR00723##
[1200] To a mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (1.07 g, 3.66 mmol) and potassium carbonate (0.69 g, 4.99 mmol) in cyclohexane (10 mL) was added 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate (1.20 g, 3.33 mmol). The reaction mixture was heated a 70.degree. C. for 72 hours, cooled to ambient temperature and diluted with ethyl acetate (250 mL). The organic layer was washed with 1N aqueous hydrochloric acid solution (30 mL) and 25% aqueous ammonium chloride solution (2.times.40 mL); dried over anhydrous sodium sulfate and concentrated in vacuo. The residue was purified by column chromatography (0-25% ethyl acetate in hexanes) afforded the title compound as a colorless oil (1.20 g, 71%): MS(ES+) m/z 504.1, 506.1 (M+1);
Step 3. Preparation of 4-(((3R)-1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00724##
[1202] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl- piperidin-3-yl) oxy)-2-fluorobenzoate and methyl with methyl 4-(((3R)-1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless oil (0.98 g, 92%), .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.53-7.43 (m, 2H), 7.25-7.21 (m, 0.5H), 7.17-7.13 (m, 0.5H), 7.03-6.95 (m, 1H), 6.47 (d, J=12.9 Hz, 0.5H) 6.37 (d, J=12.9 Hz, 0.5H), 4.76-4.66 (m, 1H), 4.38-4.31 (m, 0.5H), 4.27-4.20 (m, 0.5H), 3.14-3.10 (m, 1H), 2.94-2.84 (m, 1H), 2.75-2.60 (m, 2H), 2.05-1.96 (m, 2H), 1.91-1.80 (m, 1H), 1.70-1.52 (m, 2H), 0.91-0.85 (m, 2H), 0.70-0.57 (m, 2H); MS(ES+) m/z 490.2, 492.2 (M+1); MS(ES-) m/z 488.2, 490.2 (M-1).
Step 4. Preparation of 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00725##
[1203] And 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl- ) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00726##
[1205] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((3R)-1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-3-- yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid. The residue was purified by preparative-HPLC, the first eluting fraction was arbitrarily assigned as 4-(((R)-1-((R)-1-(2-chloro-4-fluorophenyl)-2,2,2trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide as a colorless solid (0.02 g, 4%): .sup.1H NMR (300 MHz, CDC3) .delta.8.69 (d, J=16.0 Hz, 1H), 7.52-7.46 (m, 2H), 7.12 (dd, J=2.6 Hz, 8.4 Hz, 1H), 7.02-6.95 (m, 1H), 6.44 (d, J=14.6 Hz, 1H), 4.70 (q, J=8.8 Hz, 1H), 4.37-4.32 (m, 1H), 3.42 (s, 3H), 3.13-3.08 (m, 1H), 2.93-2.89 (m, 1H), 2.75-2.69 (m, 2H), 2.10-1.85 (m, 3H), 1.72-1.53 (m, 2H), 0.95-0.87 (m, 2H), 0.72-0.66 (m, 1H), 0.62-0.56 (m, 1H); MS(ES+) m/z 567.1,569.1 (M+1); MS(ES-) m/z 565.1, 567.1 (M-1).
[1206] The second eluting fraction was arbitrarily assigned as 4-(((R)-1-((S)-1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide as a colorless solid (0.01 g, 2%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.68 (d, J=16.0 Hz, 1H), 7.57-7.51 (m, 2H), 7.26-7.22 (m, 1H), 7.04-6.97 (m, 1H), 6.38 (d, J=14.6 Hz, 1H), 4.73 (q, J=8.8 Hz, 1H), 4.31-4.23 (m, 1H), 3.41 (s, 3H), 3.16-3.12 (m, 1H), 2.91-2.85 (m, 1H), 2.78-2.60 (m, 2H), 2.09-1.99 (m, 2H), 1.70-1.54 (m, 3H), 0.94-0.91 (m, 2H), 0.67-0.63 (m, 2H); MS(ES+) m/z 567.1,569.1 (M+1); MS(ES-) m/z 565.1, 567.1 (M-1)
Example 299
Synthesis of 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00727##
[1207] Step 1. Preparation of 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate
##STR00728##
[1209] Following the procedure as described in Example 297 step 1, and making variation as required to replace 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethanol with 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethanol, the title compound was obtained as a colorless oil (1.30 g, 72%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.66-7.61 (m, 1H), 7.19-7.12 (m, 2H), 6.46 (q, J=5.6 Hz, 1H).
Step 2. Preparation of tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR00729##
[1211] Following the procedure as described in Example 297 step 2, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate and to replace 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate with 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate, the title compound was obtained as a colorless oil (1.20 g, 90%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.37-7.29 (m, 4H), 6.47 (d, J=12.6 Hz, 1H), 4.49-4.01 (m, 1H), 3.80 (d, J=5.9 Hz, 2H), 2.99-2.93 (m, 2H), 2.47-2.29 (m, 2H), 2.01-1.92 (m, 1H), 1.84-1.81 (m. 3H), 1.55 (s, 9H), 1.50-1.33 (m, 2H), 0.89-0.83 (m, 2H), 0.63-0.57 (m, 2H).
Step 3. Preparation of 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-2-fluorobenzoic acid
##STR00730##
[1213] To a solution of tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate (1.10 g, 1.91 mmol) in dichloromethane (30 mL) was added trifluoroacetic acid (10 mL). The resulting solution was stirred at ambient temperature for 1 hour and then concentrated in vacuo to provide the title compound as a gummy solid (1.0 g, 99%): MS(ES+) m/z 520.1, 522.1 (M+1); MS(ES-) m/z 518.1, 520.1 (M-1).
Step 4. Preparation of 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00731##
[1215] Following the procedure as described in Example 3 step 5, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl)piperidi- n-4-yl) methoxy)-2-fluorobenzoic acid, and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.35 g, 50%), .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.88 (br s, 1H), 7.72 (s, 1H), 7.45-7.44 (m, 2H), 7.12 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 4.77 (q, J=9.3 Hz, 1H), 3.91 (d, J=5.7 Hz, 2H), 3.33 (s, 3H), 3.00-2.99 (m, 2H), 2.33-2.25 (m, 1H), 2.04-1.95 (m, 2H), 1.79-1.70 (m, 3H), 1.41-1.30 (m, 2H), 0.90-0.84 (m, 2H), 0.69-0.64 (m, 2H); MS(ES+) m/z 597.1, 599.0 (M+1).
Example 300
Synthesis of 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00732##
[1216] Step 1. Preparation of methyl 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-fluorobenzoate
##STR00733##
[1218] Following the procedure as described in Example 297 step 2, and making variation as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate, the title compound was obtained as a colorless solid (0.80 g, 47%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.59-7.54 (m, 1H), 7.43 (d, J=8.3 Hz, 1H), 7.20 (dd, 2.6 Hz, 8.4 Hz, 1H), 7.03 (d, J=2.6 Hz, 8.3 Hz, 1H), 6.51 (d, J=12.7 Hz, 1H), 4.69 (q, J=8.7 Hz, 1H), 3.87 (s, 3H), 3.81 (d, J=6.0 Hz, 2H), 3.13-3.09 (m, 1H), 3.00-2.96 (m, 1H), 2.52 (t, J=11.2 Hz, 1H), 2.30 (t, J=11.1 Hz, 1H), 2.03-1.94 (m, 1H), 1.83-1.77 (m, 3H), 1.48-1.31 (m, 2H), 0.90-1.85 (m, 2H), 0.65-0.59 (m, 2H); MS(ES+) m/z 518.1, 520.1 (M+1).
Step 2. Preparation of 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy-5-cyclopropyl-2-fluorobenzoic acid
##STR00734##
[1220] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)o- xy)-2-fluorobenzoate with methyl 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.75 g, 96%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.12.82 (s, 1H), 7.65-7.59 (m, 2H), 7.36-7.28 (m, 2H), 6.87 (d, J=13.1 Hz, 1H), 4.84 (q, J=9.1 Hz, 1H), 3.90 (d, J=5.7 Hz, 2H), 3.07-2.95 (m, 2H), 2.49-2.40 (m, 1H), 2.20-2.13 (m, 1H), 2.02-1.93 (m, 1H), 1.77-1.74 (m, 3H), 1.33-1.27 (m, 2H), 0.93-0.83 (m, 2H), 0.59-0.54 (m, 2H); MS(ES+) m/z 504.1, 506.1 (M+1);
Step 3. Preparation of 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00735##
[1222] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-((1-(1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.32 g, 56%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.88 (br s, 1H), 7.65-7.59 (m, 2H), 7.33 (dt, J=2.7 Hz, 8.5 Hz, 1H), 7.12 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 4.84 (q, J=9.0 Hz, 1H), 3.91 (d, J=5.6 Hz, 2H), 3.33 (s, 3H), 3.01 (dd, J=11.1 Hz, 27.2 Hz, 2H), 2.49-2.40 (m, 1H), 2.16 (t, J=10.9 Hz, 1H), 2.03-1.94 (m, 1H), 1.77-1.74 (m, 3H), 1.38-1.27 (m, 2H), 0.90-0.83 (m, 2H), 0.68-0.63 (m, 2H); MS(ES+) m/z 581.2, 583.2 (M+1); MS(ES-) m/z 579.2, 581.2 (M-1).
Example 301
Synthesis of 4-((1-(1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00736##
[1223] Step 1. Preparation of 1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate
##STR00737##
[1225] Following the procedure as described in Example 297 step 1, and making variation as required to replace 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethanol with 1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethanol, the title compound was obtained as a brown oil (4.60 g, 76%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.58-7.55 (m, 1H), 7.42-7.37 (m, 1H), 7.29-7.24 (m, 1H), 5.78 (q, J=5.7 Hz, 1H).
Step 2. Preparation of tert-butyl 4-((1-(1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00738##
[1227] Following the procedure as described in Example 297 step 2, and making variation as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate and to replace 1-(2-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate with 1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl trifluoromethanesulfonate, the title compound was obtained as a pale yellow oil (0.70 g, 54%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.47-7.44 (m, 1H), 7.36 (d, J=8.4 Hz, 1H), 7.29-7.24 (m, 1H), 7.13 (t, J=8.6 Hz, 1H), 6.46 (d, J=12.6 Hz, 1H), 4.05 (q, J=8.7 Hz, 1H), 3.79 (d, J=5.9 Hz, 2H), 2.98-2.95 (m, 2H), 2.41 (t, J=11.2 Hz, 1H), 2.27 (t, J=11.4 Hz, 1H), 2.01-1.91 (m, 1H), 1.83-1.79 (m, 3H), 1.54 (m, 9H), 1.48-1.33 (m, 2H), 0.88-0.82 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 506.0, 508.0 (M+1).
Step 3. Preparation of 4-((1-(1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00739##
[1229] Following the procedure as described in Example 299 step 3, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl)piperidi- n-4-yl) methoxy)-2-fluorobenzoate with tert-butyl 4-((1-(1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.55 g, 87%): MS(ES+) m/z 504.1, 506.0 (M+1); MS(ES-) m/z 502.1, 504.1 (M-1).
Step 4. Preparation of 4-((1-(1-(3,5-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00740##
[1231] Following the procedure as described in Example 3 step 5, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-((1-(1-(3-chloro-4-fluorophenyl)-2,2,2-trifluoroethyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.23 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.88 (br s, 1H), 7.63-7.60 (m, 1H), 7.53-7.41 (m, 2H), 7.12 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 4.72 (q, J=9.4 Hz, 1H), 3.91 (d, J=5.9 Hz, 2H), 3.33 (s, 3H), 3.01-2.98 (m, 2H), 2.31-2.24 (m, 1H), 2.05-1.94 (m, 2H), 1.78-1.66 (m, 3H), 1.42-1.29 (m, 2H), 0.90-0.84 (m, 2H), 0.69-0.63 (m, 2H); MS(ES+) m/z 581.1, 583.1 (M+1); MS(ES-) m/z 579.1, 581.0 (M-1).
Example 302
Synthesis of 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl) ethoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00741##
[1232] Step 1. Preparation of tert-butyl 4-(2-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) ethyl)piperidine-1-carboxylate
##STR00742##
[1234] Following the procedure as described in Example 3 step 1, and making variation as required to replace (R)-tert-butyl-3-hydroxypiperidine-1-carboxylate with tert-butyl 4-(2-hydroxyethyl)piperidine-1-carboxylate, the title compound was obtained as a colorless oil. (3.80 g, 95%): MS(ES+) m/z 458.2, 460.2 (M+1).
Step 2. Preparation of tert-butyl 4-(2-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenyl)ethyl)piperidi- ne-1-carboxylate
##STR00743##
[1236] Following the procedure as described in Example 3 step 2, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 4-(2-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)ethyl)piperidine-1- -carboxylate, the title compound was obtained as a light yellow oil (1.90 g, 95%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.34 (d, J=8.4 Hz, 1H), 6.48 (d, J=12.7 Hz, 1H), 4.10-3.99 (m, 4H), 2.67 (d, J=12.4 Hz, 2H), 2.01-1.92 (m, 1H), 1.80-1.63 (m, 5H), 1.53 (s, 9H), 1.42 (s, 9H), 1.22-1.11 (m, 2H), 0.89-0.82 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 464.3 (M=1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-4-(2-(piperidin-4-yl)ethoxy)benzoic acid
##STR00744##
[1238] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)- piperidine-1-carboxylate with tert-butyl 4-(2-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)ethyl)piperid- ine-1-carboxylate, the title compound was obtained as trifluoroacetic acid salt (1.72 g, 99%): MS(ES+) m/z 308.1 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl) ethoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR00745##
[1240] Following the procedure as described in Example 3 step 4, and making variation as required to replace (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid with 5-cyclopropyl-2-fluoro-4-(2-(piperidin-4-yl) ethoxy)benzoic acid, the title compound was obtained (1.37 g, 56%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.87 (brs, 1H), 9.62 (br, 1H), 7.76 (s, 1H), 7.61-7.60 (m, 2H), 7.31 (d, J=8.5 Hz, 1H), 6.92 (d, J=13.1 Hz, 1H), 4.29-4.12 (m, 4H), 3.40-3.36 (m, 2H), 2.95-2.88 (m, 2H), 2.02-1.74 (m, 6H), 1.48-1.37 (m, 2H), 0.92-0.86 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 466.1, 468.0 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl) ethoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00746##
[1242] Following the procedure as described in 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl)ethoxy)-2-fluoro- benzoic acid and cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.02 g, 10%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (br s, 1H), 9.55 (br s, 1H), 7.74-7.72 (m, 1H), 7.58-7.57 (m, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.95 (d, J=13.0 Hz, 1H), 4.26 (s, 2H), 4.10-4.08 (m, 2H), 3.37-3.33 (m, 2H), 3.30 (s, 3H), 2.94-2.86 (m, 2H), 2.01-1.89 (m, 3H), 1.72-1.68 (m, 3H), 1.45-1.37 (m, 2H), 0.88-0.82 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 543.0, 545.0 (M+1).
Example 303
Synthesis of 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl) ethoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00747##
[1244] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl)ethoxy)-2-fluoro- benzoic acid and to replace cyclopropylsulfonamide with ethanesulfonamide, the title compound was obtained as a colorless solid (0.06 g, 34%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.78 (br s, 1H), 9.73 (br s, 1H), 7.73-7.72 (m, 1H), 7.58-7.57 (m, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.94 (d, J=13.0 Hz, 1H), 4.26 (s, 2H), 4.10-4.08 (m, 2H), 3.44 (q, J=7.3 Hz, 2H), 3.37-3.33 (m, 2H), 2.94-2.86 (m, 2H), 2.01-1.88 (m, 3H), 1.72-1.68 (m, 3H), 1.45-1.37 (m, 2H), 1.21 (t, J=7.3 Hz, 3H), 0.88-0.82 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 557.0, 559.0 (M+1).
Example 304
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(2-(1-(3,5-dichlorobenzyl)piperid- in-4-yl) ethoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00748##
[1246] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-(2-(1-(3,5-dichlorobenzyl)piperidin-4-yl)ethoxy)-2-fluoro- benzoic acid, the title compound was obtained as a colorless solid (0.10 g, 56%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.81 (br s, 1H), 9.75 (br s, 1H), 7.73-7.72 (m, 1H), 7.58-7.57 (m, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.95 (d, J=3.0 Hz, 1H), 4.26 (s, 2H), 4.10-4.08 (m, 2H), 3.37-3.33 (m, 2H), 3.08-3.00 (m, 1H) (m, 2H), 2.01-1.89(m, 3H), 1.72-1.68 (m, 3H), 1.45-1.37 (m, 2H), 1.10-1.05 (m, 4H), 0.88-0.82 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 568.9, 570.9 (M+1).
Example 305
Synthesis of (R)-5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl) methyl)piperidin-3-yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00749##
[1248] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy)benzam- ide and to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 2-(chloromethyl)-6-cyclopropyl-4-(trifluoromethyl)pyridine, the title compound was obtained as a colorless solid (0.17 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.71 (br s, 1H), 7.54 (s, 1H), 7.47 (s, 1H), 7.13 (d, J=8.4 Hz, 1H), 6.99 (d, J=13.2 Hz, 1H), 4.66 (br s, 1H), 3.79-3.67 (m, 2H), 3.29 (s, 3H), 2.87-2.84 (m, 1H), 2.59-2.55 (m, 2H), 2.46-2.43 (m, 1H), 2.28-2.19 (m, 1H), 2.12-2.03 (m, 1H), 1.91-1.80 (m, 2H), 1.63-1.58 (m, 2H), 1.03-0.82 (m, 1H), 0.74-0.65 (m, 2H); MS(ES+) m/z 556.1 (M+1); MS(ES-) m/z 554.1 (M-1).
Example 306
Synthesis of 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00750##
[1249] Step 1. Preparation of tert-butyl 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl) methyl)piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR00751##
[1251] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate and to replace 1,3-dichloro-5-(1-chloroethyl) benzene with 2-(chloromethyl)-6-cyclopropyl-4-(trifluoromethyl)pyridine, the title compound was obtained as a colorless solid (1.00 g, 64%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.40-7.35 (m, 2H), 7.19 (s, 1H), 6.49 (d, J=12.7 Hz, 1H), 3.85 (d, J=5.9 Hz, 2H), 3.76 (s, 2H), 3.08-3.04 (m, 2H), 2.29-2.21 (m, 2H), 2.13-1.97 (m, 1H), 1.88-1.84 (m, 3H), 1.63-1.50 (m, 11H), 1.05-1.02 (m, 4H), 0.91-0.85 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 549.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl) methyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR00752##
[1253] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)- piperidine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l) piperidin-4-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless oil (1.31 g, 99%): MS(ES+) m/z 493.0 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl) pyridin-2-yl)methyl)piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)be- nzamide
##STR00753##
[1255] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l)piperidin-4-yl) methoxy)-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.10 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.00 (br s, 1H), 7.60 (s, 1H), 7.48 (s, 1H), 7.13 (d, J=8.4 Hz, 1H), 6.83 (d, J=12.9 Hz, 1H), 3.92-3.87 (m, 4H), 3.11 (s, 3H), 3.05-3.01 (m, 2H), 2.45-2.37 (m, 2H), 2.28-2.23 (m, 1H), 2.01-1.96 (m, 1H), 1.83-1.80 (m, 3H), 1.51-1.39 (m, 2H), 1.02-0.96 (m, 4H), 0.88-0.81 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 570.0 (M+1); MS(ES-) m/z 568.0 (M-1).
Example 307
Synthesis of 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l)piperidin-4-yl) methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
##STR00754##
[1257] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l)piperidin-4-yl) methoxy)-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with ethanesulfonamide, the title compound was obtained as a colorless solid (0.14 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.15 (br s, 1H), 7.62 (s, 1H), 7.50 (s, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 3.96-3.94 (m, 2H), 3.85 (s, 2H), 3.37-3.30 (m, 2H), 3.05-3.01 (m, 2H), 2.41-2.24 (m, 3H), 2.06-1.97 (m, 1H), 1.85-1.82 (m, 3H), 1.52-1.41 (m, 2H), 1.20 (t, J=7.3 Hz, 3H), 1.04-0.99 (m, 4H), 0.91-0.85 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 584.0 (M+1); MS(ES-) m/z 582.1 (M-1).
Example 308
Synthesis of 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l)piperidin-4-yl) methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide
##STR00755##
[1259] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-4-((1-((6-cyclopropyl-4-(trifluoromethyl)pyridin-2-yl)methy- l)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.13 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.22 (br s, 1H), 7.61 (s, 1H), 7.49 (s, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 3.95 (d, J=5.8 Hz, 2H), 3.83 (s, 2H), 3.04-2.99 (m, 3H), 2.38-2.24 (m, 3H), 2.07-1.97 (m, 1H), 1.85-1.81 (m, 3H), 1.52-1.41 (m, 2H), 1.04-0.99 (m, 8H), 0.91-0.85 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 596.1 (M+1); MS(ES-) m/z 594.2 (M-1).
Example 309
Synthesis of 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide (arbitrarily assigned)
##STR00756##
[1260] Step 1. Preparation of methyl 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluorobenzoate and methyl 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl) ethyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate
##STR00757##
[1262] Following the procedure as described in Example 50 step 1 and making variations as required to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 3-chloro-6-(1-chloroethyl)-2-cyclopropylpyridine. The residue was purified by preparative-HPLC, the first eluting fraction was arbitrarily assigned as methyl 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- )oxy)-5-cyclopropyl-2-fluorobenzoate (0.36 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.44 (d, J=8.2 Hz, 1H), 7.38 (d, J=8.4 Hz, 1H), 7.01 (d, J=8.2 Hz, 1H), 6.52 (d, J=12.9 Hz, 1H), 4.36-4.28 (m, 1H), 3.84 (s, 3H), 3.63 (q, J=6.8 Hz, 1H), 2.99-2.95 (m, 1H), 2.66-2.62 (m, 1H), 2.45-2.40 (m, 1H), 2.29-2.15 (m, 2H), 2.05-1.96 (m, 2H), 1.80-1.74 (m, 1H), 1.60-1.42 (m, 2H), 1.29 (d, J=6.8 Hz, 3H), 1.07-0.84 (m, 6H), 0.63-0.58 (m, 2H); MS(ES+) m/z 473.1, 475.1 (M+1).
[1263] The second eluting fraction was arbitrarily assigned as methyl 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate (0.36 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.45 (d, J=8.2 Hz, 1H), 7.41 (d, J=8.4 Hz, 1H), 7.04 (d, J=7.5 Hz, 1H), 6.53 (d, J=12.8 Hz, 1H), 4.35-4.29 (m, 1H), 3.85 (s, 3H), 3.67 (q, J=6.8 Hz, 1H), 3.039-3.00 (m, 1H), 2.72-2.68 (m, 1H), 2.49-2.41 (m, 1H), 2.28-2.13 (m, 2H), 2.05-1.96 (m, 2H), 1.82-1.77 (m, 1H), 1.64-1.38 (m, 2H), 1.31 (d, J=6.8 Hz, 3H), 1.06-0.86 (m, 6H), 0.65-0.60 (m, 2H); MS(ES+) m/z 473.1, 475.1 (M+1).
Step 2. Preparation of 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00758##
[1265] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl) oxy)-2-fluorobenzoate with methyl 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl) piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a pale yellow oil (0.06 g, 17%): MS(ES+) m/z 459.1, 461.1 (M+1); MS(ES-) m/z 457.1, 459.1 (M-1).
Step 3. Preparation of 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00759##
[1267] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((R)-1-((R)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- )oxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.01 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.77 (br s, 1H), 7.67 (d, J=7.9 Hz, 1H), 7.53 (d, J=8.8 Hz, 1H), 7.10 (d, J=7.8 Hz, 1H), 6.87 (d, J=13.5 Hz, 1H), 4.86-4.82 (m, 1H), 4.66-4.64 (m, 1H), 3.95-3.92 (m, 1H), 3.68-3.64 (m, 1H), 3.38 (s, 3H), 2.95-2.86 (m, 1H), 2.60-2.55 (m, 2H), 2.36-2.31 (m, 1H), 2.13-2.07 (m, 2H), 1.92-1.87 (m, 1H), 1.69 (d, J=6.4 Hz, 3H), 1.62-161 (m, 1H), 1.10-0.86 (m, 6H), 0.62-0.60 (m, 2H); MS(ES+) m/z 536.1, 538.1 (M+1).
Example 310
Synthesis of 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00760##
[1268] Step 1. Preparation of 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00761##
[1270] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)o- xy)-2-fluorobenzoate with methyl 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.17 g, 49%): MS(ES+) m/z 459.1, 461.0 (M+1); MS(ES-) m/z 457.1, 459.1 (M-1).
Step 2. Preparation of 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- ) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00762##
[1272] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((R)-1-((S)-1-(5-chloro-6-cyclopropylpyridin-2-yl)ethyl)piperidin-3-yl- )oxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.05 g, 44%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.77 (br s, 1H), 7.67 (d, J=7.7 Hz, 1H), 7.57 (d, J=8.0 Hz, 1H), 7.08 (d, J=7.4 Hz, 1H), 6.89 (d, J=13.6 Hz, 1H), 4.95-4.89 (m, 1H), 4.67-4.66 (m, 1H), 3.92-3.89 (m, 1H), 3.62-3.59 (m, 1H), 3.39 (s, 3H), 3.05-2.97 (m, 1H), 2.62-2.50 (m, 2H), 2.38-2.34 (m, 1H), 2.05-1.93 (m, 3H), 1.67 (d, J=4.4 Hz, 3H), 1.51-1.45 (m, 1H), 1.10-0.90 (m, 6H), 0.65-0.63 (m, 2H); MS(ES+) m/z 536.1, 538.1 (M+1).
Example 311
Synthesis of (R)-4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00763##
[1274] Following the procedure as described in Example 3 step 4, and making variation as required to replace (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid trifluoroacetate with (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy)benzam- ide and to replace 3,5-dichlorobenzaldehyde with 3-chloro-2-fluoro-5-(trifluoromethyl)benzaldehyde, the title compound was obtained as a colorless solid (0.18 g, 35%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 7.98 (dd, J=1.9 Hz, 6.3 Hz, 1H), 7.79 (dd, J=1.9 Hz, 5.6 Hz, 1H), 7.12 (d, J=8.4 Hz, 1H), 7.02 (d, J=13.2 Hz, 1H), 4.65-4.63 (m, 1H), 3.71 (d, J=2.0 Hz, 2H), 3.32 (s, 3H), 2.83-2.79 (m, 1H), 2.56-2.54 (m, 2H), 2.41-2.36 (m, 1H), 2.10-2.00 (m, 1H), 1.90-1.77 (m, 2H), 1.60-1.55 (m, 2H), 0.89-0.84 (m, 2H), 0.73-0.67 (m, 2H); MS(ES+) m/z 567.2, 569.0 (M+1); MS(ES-) m/z 565.1, 567.1 (M-1).
Example 312
Synthesis of (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fl- uoro-N-(methylsulfonyl)benzamide
##STR00764##
[1276] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy-5-cyclopropyl-2-flu- orobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.05 g, 60%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (br s, 1H), 7.55-7.50 (m, 1H), 7.42-7.38 (m, 1H), 7.17-7.11 (m, 2H), 7.00 (d, J=13.2 Hz, 1H), 4.62-4.60 (m, 1H), 3.60 (s, 2H), 3.31 (s, 3H), 2.82-2.79 (m, 1H), 2.60-2.55 (m, 1H), 2.49-2.39 (m, 2H), 2.12-2.03 (m, 1H), 1.94-1.91 (m, 1H), 1.83-1.79 (m, 1H), 1.61-1.57 (m, 2H), 0.91-0.85 (m, 2H), 0.72-0.68 (m, 2H); MS(ES+) m/z 499.2, 501.2 (M+1); MS(ES-) m/z 497.3, 499.3 (m-1).
Example 313
Synthesis of (R)-5-cyclopropyl)-N-(cyclpropylsulfonyl)-2-fluoro-4-((1-(3-fluoro-4-((1-- (3-fluoro-4-methoxybenzyl) piperidin-3-yl)oxy)benzamide
##STR00765##
[1278] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(3-fluoro-4-methoxybenzyl)piperidin-3-yl- )oxy)benzoic acid, the title compound was obtained as a colorless solid (0.03 g, 23%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.55 (d, J=9.1 Hz, 1H), 7.11-6.86 (m, 3H), 6.60 (d, J=18.1 Hz, 1H), 4.50 (s, 1H), 3.88 (s, 3H), 3.53 (s, 2H), 3.12-3.05 (m, 1H), 3.02-2.91 (m, 1H), 2.83-2.66 (m, 1H), 2.44-2.20 (m, 2H), 2.15-1.98 (m, 2H), 1.95-1.82 (m, 2H), 1.66-1.53 (m, 2H), 1.48-1.42 (m, 2H), 1.17-1.13 (m, 2H), 0.96-0.88 (m, 2H), 0.69-0.64 (m, 2H); MS(ES+) m/z 521.2 (M+1), MS(ES-+) m/z 519.3 (M-1).
Example 314
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(methylsulfonyl)piper- idin-3-yl)oxy)benzamide
##STR00766##
[1280] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl)piperidin-3-yl)oxy)benzo- ic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.07 g, 40%) .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.72 (d, J=15.9 Hz, 1H), 7.59 (d, J=9.0 Hz, 1H), 6.65 (d, J=14.2 Hz, 1H), 4.49 (brs, 1H), 3.74 (d, J=14.2 Hz, 1H), 3.51-3.45 (m, 1H), 3.41 (s, 3H), 3.25-3.12 (m, 2H), 2.83 (s, 3H), 2.11-2.01 (m, 3H), 1.85-1.79 (m, 2H), 0.94 (d, J=8.4 Hz, 2H), 0.66 (d, J=5.2 Hz, 2H); MS(ES+) m/z 435.1 (M+1); MS(ES-) m/z 433.2 (M-1).
Example 315
Synthesis of 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)-N-(methylsulfonyl)benzamide
##STR00767##
[1281] Step 1. Preparation of methyl 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)benzoate and methyl 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)benzoate
##STR00768##
[1283] Following the procedure as described in Example 50 step 1, and making variations as required to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1-(1-chloroethyl)-4-fluorobenzene, the first eluting fraction was arbitrarily assigned as methyl 5-cyclopropyl -2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl) ethyl)piperidin-3-yl)oxy)benzoate (0.50 g, 38%): .sup.1H NMR (300 MHz, CDCl3) .delta.7.41 (d, J=8.4 Hz, 1H), 7.27-7.22 (m, 2H), 6.95 (t, J=8.7 Hz, 2H), 6.53 (d, J=13 Hz, 1H), 4.39-4.32 (m, 1H), 3.86 (s, 3H), 3.54-3.46 (m, 1H), 3.02-3.00 (m, 1H), 2.62-2.59 (m, 1H), 2.25-1.96 (m, 4H), 1.83-1.76 (m, 1H), 1.63-1.43 (m, 2H), 1.32 (d, J=6.8 Hz, 3H), 0.91-0.88 (m, 2H), 0.66-0.62 (m, 2H); MS(ES+) m/z 416.2 (M+1).
[1284] The second eluting fraction was arbitrarily assigned as methyl 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl) ethyl)piperidin-3-yl)oxy)benzoate (0.5 g, 38%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.36 (d, J=8.4 Hz, 1H), 7.23-7.21 (m, 2H), 6.94 (t, J=8.9 Hz, 2H), 6.46 (d, J=12.9 Hz, 1H), 4.32-4.24 (m, 1), 3.82 (s, 3H), 3.50 (q, J=6.7 Hz, 1H), 2.94-2.91 (m, 1H), 2.77-2.73 (m, 1H), 2.15-1.93 (m, 4H), 1.88-1.76 (m, 1H), 1.65-1.37 (m, 2H), 1.30 (d, J=6.8 Hz, 3H), 0.85-0.81 (m, 2H), 0.61-0.57 (m, 2H); MS(ES+) m/z 416.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl) piperidin-3-yl)oxy)benzoic acid
##STR00769##
[1286] Following the procedure as described 1, ample 50 step 2, and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-piperidin-3-yl)- oxy)-2-fluorobenzoate with methyl 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl) oxy)benzoate, the title compound was obtained as a colorless solid (0.16 g, 33%): MS(ES+) m/z 402.2 (M+1); MS(ES-) m/z 400.2 (M-1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl) piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00770##
[1288] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-2-fluoro-4-(((R)-1-((S)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.12 g, 62%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.99-8.66 (m, 1H), 7.56 (d, J=8.0 Hz, 1H), 7.45-7.42 (m, 2H), 7.19 (t, J=8.0 Hz, 2H), 6.96 (d, J=12.9 Hz, 1H), 5.01-4.86 (m, 1H), 4.61-4.47 (m, 1H), 4.06-3.92 (m, 1H), 3.59-3.46 (m, 1H), 3.41 (s, 3H), 2.59-2.42 (m, 2H), 2.39-2.28 (m, 1H), 2.23-2.00 (m, 2H), 1.97-1.87 (m, 1H), 1.81 (d, J=5.3 Hz, 3H), 1.64-1.46 (m, 1H), 0.93-0.88 (m, 2H), 0.64-0.60 (m, 2H); MS(ES+) m/z 479.1 (M+1); MS(ES-) m/z 477.2 (M-1).
Example 316
Synthesis of 5-cyclopropyl-2-fluoro-4-(((R)-1-(4-fluorophenyl)ethyl)piperidin-3-yl)oxy- )-N-(methylsulfonyl)benzamide
##STR00771##
[1289] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl)ethyl) piperidin-3-yl)oxy benzoic acid
##STR00772##
[1291] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl)oxy)-2-fluorobenzoate with methyl 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy) benzoate, the title compound was obtained as a beige color solid (0.23 g, 99%): MS(ES-) m/z 400.2 (M-1); MS(ES+) m/z 402.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-(((R)-((R)-1-(4-fluorophenyl) ethyl)piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00773##
[1293] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.04 g, 15%) .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.9.07-8.61 (m, 1H), 7.54 (d, J=8.7 Hz, 1H), 7.45-7.41 (m, 2H), 7.18 (t, J=8.26 Hz, 2H), 7.02-6.89 (m, 1H), 5.10-4.91 (m, 1H), 4.63-4.46 (m, 1H), 3.82-3.56 (m, 2H), 3.40 (s, 3H), 2.63-2.41 (m, 2H), 2.40-2.28 (m, 1H), 2.25-1.87 (m, 3H), 1.80 (d, J=6.3 Hz, 3H), 1.63-1.43 (m, 1H), 0.95-0.87 (m, 2H), 0.67-0.59 (m, 2H); MS(ES+) m/z 479.1 (M+1); MS(ES-) m/z 477.2 (M-1).
Example 317
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-(((R)-1-((R)-1-(4-fluoro- phenyl) ethyl)piperidin-3-yl)oxy)benzamide
##STR00774##
[1295] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 5-cyclopropyl-2-fluoro-4-(((R)-1-((R)-1-(4-fluorophenyl)ethyl)piperidin-3- -yl)oxy)benzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%) .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.91-8.63 (m, 1H), 7.61-7.51 (m, 1H), 7.49-7.38 (m, 2H), 7.19-7.12 (m, 2H), 7.04-6.89 (m, 1H), 5.12-4.88 (m, 1H), 4.63-4.46 (m, 1H), 3.86-3.52 (m, 2H), 3.17-3.02 (m, 1H), 2.69-2.28 (m, 3H), 2.23-1.89 (m, 3H), 1.89-1.85 (m, 3H), 1.50-1.41 (m, 3H), 1.21-1.10 (m, 2H), 0.96-0.85 (m, 2H), 0.69-0.58 (m, 2H); MS(ES+) m/z 505.2 (M+1); MS(ES-) m/z 503.2 (M-1).
Example 318
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl)benzyl) piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00775##
[1296] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl) benzyl)piperidin-3-yl)oxy)benzoate
##STR00776##
[1298] Following the procedure as described in Example 50 step 1, and making variations as required to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 2-(bromomethyl)-4-fluoro-1-(trifluoromethyl) benzene, the title compound was obtained as a colorless oil (0.20 g, 99%): MS(ES+) m/z 470.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl)benzyl) piperidin-3-yl)oxy)benzoic acid
##STR00777##
[1300] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-(1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl) oxy)-2-fluorobenzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl) benzyl)piperidin-3-yl)oxy)benzoate, the title compound was obtained as a colorless oil (0.12 g, 59%): MS(ES+) m/z 456.1 (M+1); MS(ES-) m/z 454.2 (M-1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl)benzyl) piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide
##STR00778##
[1302] Following the procedure is described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(trifluoromethyl)piperidin)p- iperidin-3-yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.03 g, 18%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.99-8.55 (m, 1H), 7.78-7.74 (m, 2H), 7.56-7.53 (d, J=8.6 Hz, 1H), 7.27-7.22 (m, 1H), 4.97-4.83 (m, 1H), 4.61-4.56 (m, 1H), 4.37-4.33 (m, 1H), 3.81-3.57 (m, 1H), 3.38 (s, 3H), 3.35-3.25 (m, 1H), 3.03-2.78 (m, 2H), 2.37-2.22 (m, 1H), 2.17-1.98 (m, 3H), 1.87-1.69 (m, 1H), 0.92 (d, J=8.1 Hz, 2H), 0.68-0.56 (m, 2H); MS(ES+) m/z 533.2 (M+1); MS(ES-) m/z 531.2 (M-1).
Example 319
Synthesis of ((R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(3-(trifluoromethyl)- benzyl) piperidin-3-yl)oxy)benzamide
##STR00779##
[1303] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(3-(trifluoromethyl)benzyl) piperidin-3-yl)oxy)benzoate
##STR00780##
[1305] Following the procedure as described in Example 50 step 1, and making variations as required to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1-(chloromethyl)-3-(trifluoromethyl)benzene, the title compound was obtained as a colorless oil (0.24 g, 28%): MS(ES+) m/z 452.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(3-(trifluoromethyl)benzyl) piperidin-3-yl)oxy)benzoic acid
##STR00781##
[1307] Following the procedure as described in Example 50 step 2 and making variations as required to replace methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)piperidin- 3-yl)oxy)-2-fluorobenzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(3-(trifluoromethyl)piperidin)piperidin-3-yl- )oxy) benzoate, the title compound was obtained as colorless oil (0.25 g, 99%): MS(ES+) m/z 438.2 (M+1); MS(ES-) m/z 436.2 (M-1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(3-(trifluoromethyl) benzyl)piperidin-3-yl)oxy)benzamide
##STR00782##
[1309] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-(3-(trifluoromethyl)piperidin)piperidin-- 3-yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.04 g, 16%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.9.03-8.68 (m, 1H), 7.76-7.67 (m, 4H), 7.56-7.54 (m, 1H), 6.95-6.71 (m, 1H), 4.96-4.78 (m,1H), 4.47-4.23 (m, 2H), 4.02-3.72 (m, 1H), 3.40 (s, 3H), 2.96-2.58 (m, 2H), 2.43-2.26 (m, 1H), 2.21-1.91 (m, 3H), 1.83-1.56 (m, 2H), 0.98-0.80 (m, 2H), 0.68-0.51 (m, 2H); MS(ES+) m/z 515.1 (M+1).
Example 320
Synthesis of 4-((2-(2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00783##
[1310] Step 1. Preparation of tert-butyl 6-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)-2-azaspiro[3.3]hepta- ne-2-carboxylate
##STR00784##
[1312] Following the procedure as described in Example 3 step 1, and making variation as required to replace (R)-tert-butyl 3-hydroxypiperidine-1-carboxylate with tert-butyl 6-hydroxy-2-azaspiro[3.3]heptane-2-carboxylate, the title compound was obtained as a colorless solid (2.36 g, 53%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.85 (d, J=7.6 Hz, 1H), 6.42 (d, J=11.9 Hz, 1H), 4.64-4.55 (m, 1H), 3.95 (d, J=11.2 Hz, 4H), 2.78-2.71 (m, 2H), 2.44-2.37 (m, 2H), 1.55 (s, 9H), 1.42 (s, 9H); MS(ES+) m/z 442.1, 444.0 (M +1).
Step 2. Preparation of tert-butyl 6-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-azaspiro[3.3]- heptane-2-carboxylate
##STR00785##
[1314] Following the procedure as described in Example 3 step 2, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 6-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)-2-azaspiro[3.3]hepta- ne-2-carboxylate, the title compound was obtained as a colorless oil (0.96 g, 95%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.3.1 (d, J=8.3 Hz, 1H), 6.28 (d, J=12.4 Hz, 1H), 4.61-4.52 (m, 1H), 3.94 (d, J=12.2 Hz, 4H), 2.75-2.68 (m, 2H), 2.37-2.30 (m, 2H), 2.01-1.93 (m, 1H), 1.54 (s, 9H), 1.41 (s, 9H), 0.90-0.82 (m, 2H), 0.63-0.57 (m, 2H); MS(ES+) m/z 448.2 (M+1).
Step 3. Preparation of 4-(2-azaspiro[3.3]heptan-6-yloxy)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR00786##
[1316] Following the procedure as described in Example 3step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)- piperidine-1-carboxylate with tert-butyl 6-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-azaspiro[3.3]- heptane-2-carboxylate, the title compound was obtained (0.86 g, 99%): MS(ES+) m/z 292.2 (M+1); MS(ES-) m/z 290.3 (M-1)
Step 4. Preparation of 4-((2-(2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR00787##
[1318] Following the procedure as described in Example 3 step 4, and making variation as required to replace (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoic acid with 4-(2-azaspiro[3.3]heptan-6-yloxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace 3,5-dichlorobenzaldehyde with 2-chloro-4-fluorobenzaldehyde, the title compound was obtained as colorless solid (0.30 g, 37%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.87 (br s, 1H), 10.35 (br s, 1H), 7.70-7.62 (m, 2H), 7.42-7.36 (m, 1H), 7.27 (d, J=8.4 Hz, 1H), 6.72 (d, J=12.9 Hz, 1H), 4.79-4.71(m, 1H), 4.51-4.49 (m, 2H), 4.32-4.10 (m, 4H), 2.92-2.80 (m, 2H), 2.39-2.30 (m, 2H), 2.04-1.95 (m, 1H), 0.93-0.86 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 434.1, 436.1 (M+1); MS(ES-) m/z 432.2, 434.1 (M-1).
Step 5. Preparation of 4-((2-(2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl) oxy-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00788##
[1320] Following the procedure as described in Example 3 step 5, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy-2-fluorobe- nzoic acid with 4-((2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl)oxy)-5-cyclopropy- l-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as colorless solid (0.04 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.73 (br s, 1H), 7.59-7.52 (m, 2H), 7.25-7.21 (m, 1H), 7.14-7.08 (m, 1H), 6.31 (d, J=13.8 Hz, 1H), 4.66-4.62 (m, 1H), 4.52-4.40 (m, 4H), 3.95-3.93 (m, 2H), 3.40 (s, 3H), 3.06-3.00 (m, 1H), 2.86-2.80 (m, 1H), 2.60-2.45 (m, 2H), 2.02-1.93 (m, 1H), 0.96-0.90 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 511.8, 513.1 (M+1); MS(ES-) m/z 509.2, 511.2 (M-1).
Example 321
Synthesis of 4-((2-(2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl) oxy-5-cyclopropyl-2-fluoro-N-((3-fluoroazetidin-1-yl)sulfonyl)benzamide
##STR00789##
[1322] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-((2-(2-chloro-4-fluorobenzyl)-2-azaspiro[3.3]heptan-6-yl)oxy)-5-cyclopr- opyl-2-fluorobenzoic acid and to replace cyclopropylsulfonamide with 3-fluoroazetidine-1-sulfonamide, the title compound was obtained as colorless solid (0.08 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.48 (br s, 1H), 7.69-7.62 (m, 2H), 7.39 (dt, J=2.6 Hz, 8.4 Hz, 1H), 7.11 (d, J=8.2 Hz, 1H), 6.79 (d, J=12.7 Hz, 1H), 5.48-5.42 (m, 0.5H), 5.29-5.23 (m, 0.5H), 4.80-4.71 (m, 1H), 4.50 (s, 2H), 4.43-4.12 (m, 8H), 2.86 (br s, 2H), 2.37-2.30 (m, 2H), 2.06-1.97 (m, 1H), 0.93-0.86 (m, 2H), 0.71-0.66 (m, 1H); MS(ES+) m/z 570.1, 572.1 (M+1); MS(ES-) m/z 568.1, 570.1 (M-1).
Example 322
Synthesis of N-(azetidin-1-ylsulfonyl)-4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methy- lpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00790##
[1324] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl- 2-fluorobenzoic acid, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.07 g, 23%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.70-8.55 (m, 1H), 7.59-7.48 (m, 2H), 7.13-7.04 (m, 1H), 6.99-6.89 (m, 1H), 6.52-6.43 (m, 1 H), 4.40-4.14 (m, 5H), 4.06-3.94 (m, 1H), 3.45-3.32 (m, 1H), 3.09-3.00 (m, 1H), 2.60-2.46 (m, 1H), 2.33-2.13 (m, 4H), 2.09-1.87 (m, (m, 2H), 1.69-1.42 (m, 2H), 1.22-1.14 (m, 3H), 0.97-0.86 (m, 2H), 0.69-0.60 (m, 2H); MS(ES+) m/z 554.2, 556.2 (M+1).
Example 323
Synthesis of 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00791##
[1326] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)oxy)-2-fluorob- enzoic acid with 4-(((3R,6R)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl- 2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.14 g, 44%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.75-8.57 (m, 1H), 7.58-7.47 (m, 2H), 7.13-7.03 (m, 1H), 7.00-6.88 (m, 1H), 6.53-6.41 (m, 1H), 4.39-4.25 (m, 1H), 4.07-3.93 (m, 1H), 3.44-3.33 (m, 1H), 3.14-2.99 (m, 2H), 2.57-2.46 (m, 1H), 2.31-2.11 (m, 2H), 2.08-1.85 (m, 2H), 1.69-1.37 (m, 5H), 1.22-1.08 (m, 4H), 0.96-0.84 (m, 2H), 0.69-0.56 (m, 2H); MS(ES+) m/z 539.2, 541.2, (M+1).
Example 324
Synthesis of 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00792##
[1327] Step 1. Preparation of (2S,5R)-benzyl 5-hydroxy-2-methylpiperidine-1-carboxylate
##STR00793##
[1329] Following the procedure as described in Example 52 step 1, and making variations as required to replace (3R,6R)-6-methylpiperidin-3-ol with (3R,6S)-6-methylpiperidin-3-ol (Ian A. O'Neil et al., Synlett, 2000, 5, 695), the title compound was obtained (1.45 g, 27%) as a colorless oil: MS(ES+) m/z 250.2 (M+1).
Step 2. Preparation of (2S,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)-2-methylpiperidine-1- -carboxylate
##STR00794##
[1331] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (2S,5R)-benzyl 5-hydroxy-2-methylpiperidine-1-carboxylate, the title compound was obtained as a colorless oil (1.63 g, 59%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.91-7.84 (m, 1H), 7.43-7.28 (m, 5H), 6.78-6.61 (m, 1H), 5.23-5.06 (m, 2H), 4.61-4.09 (m, 3H), 3.07-2.88 (m, 1H), 2.17-1.99 (m, 1H), 1.96-1.50 (m, 12H), 1.31-1.17 (m, 3H); MS(ES+) m/z 478.2, 480.2 (M+1).
Step 3. Preparation of (2S,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-methylpiperid- ine-1-carboxylate
##STR00795##
[1333] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (2S,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)-2-methylpiperidine-1- -carboxylate, the title compound was obtained as a colorless oil (1.35 g, 82%): .sup.1H NMR (300 MHz, CDCl.sub.3) 67 7.43-7.28 (m, 6H), 6.64-6.53 (m, 1H), 5.23-5.07 (m, 2H), 4.63-4.10 (m, 3H), 3.01-2.82 (m, 1H), 2.14-1.94 (m, 2H), 1.89-1.75 (m, 2H), 1.73-1.62 (m, 1H), 1.57 (s, 9H), 1.28-1.19 (m, 3H), 0.94-0.84 (m, 2H), 0.67-0.58 (m, 2H); MS(ES+) m/z 484.3 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6S)-6-methylpiperidin-3-yl)oxy)benzoate
##STR00796##
[1335] Following the procedure as described in Example 52 step 4, and making variation as required to replace (2R,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-methylpiperid- ine-1-carboxylate with (2S,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)-2-methylpiperid- ine-1-carboxylate, the title compound was obtained as a colorless oil (0.97 g, 99%): MS(ES+) m/z 350.3 (M+1).
Step 5. Preparation of tert-butyl 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate
##STR00797##
[1337] Following the procedure as described in Example 34 step 2, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yl)oxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6S)-6-methylpiperidin-3-yl)oxy)benzoate, and to replace 1-(bromomethyl)-4-fluoro-2-(trifluoromethyl)benzene with 1-(bromomethyl)-2-chloro-4-fluorobenzene, the title compound was obtained as a colorless oil (1.10 g, 82%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.53-7.43 (m, 1H), 7.41-7.34 (m, 1H), 7.08-6.99 (m, 1H), 6.86-6.73 (m, 1H), 6.48-6.36 (m, 1H), 4.48-4.34 (m, 1H), 3.93-3.76 (m, 1H), 3.50-3.36 (m, 1H), 2.97-2.81 (m, 1H), 2.79-2.65 (m, 1H), 2.58-2.42 (m, 1H), 2.22-2.04 (m, 1H), 2.01-1.66 (m, 4H), 1.57 (s, 9H), 1.19-1.09 (m, 3H), 0.97-0.87 (m, 2H), 0.75-0.62 (m, 2H); MS(ES+) m/z 492.3, 494.3 (M+1).
Step 6. Preparation of 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00798##
[1339] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoate the title compound was obtained as a colorless solid (0.94 g, 96%): MS(ES+) m/z 436.2, 438.2 (M+1).
Step 7. Preparation of 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00799##
[1341] Following the procedure as described in Example 17 step 2, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.03 g, 9%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.77-8.58 (m, 1H), 7.62-7.53 (m, 1H), 7.48-7.40 (m, 1H), 7.08-6.99 (m, 1H), 6.82-6.72 (m, 1H), 6.49-6.38 (m, 1H), 4.47-4.37 (m, 1H), 3.92-3.79 (m, 1H), 3.49-3.35 (m, 4H), 2.96-2.81 (m, 1H), 2.80-2.67 (m, 1H), 2.56-2.43 (m, 1H), 2.20-2.05 (m, 1H), 2.03-1.66 (m, 4H), 1.19-1.07 (m, 3H), 1.02-0.87 (m, 2H), 0.78-0.60 (m, 2H); MS(ES+) m/z 513.2, 515.2 (M+1).
Example 325
Synthesis of N-(azetidin-1-ylsulfonyl)-4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methy- lpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzamide
##STR00800##
[1343] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-2-flu- orobenzoic acid, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.08 g, 27%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.69-8.55 (m, 1H), 7.63-7.57 (m, 1H), 7.50-7.41 (m, 1), 7.08-6.99 (m, 1H), 6.83-6.73 (m, 1H), 6.50-6.39 (m, 1H), 4.48-4.38 (m, 1H), 4.34-4.15 (m, 4H), 3.92-3.81 (m, 1H), 3.48-3.36 (m, 1H), 2.94-2.83 (m, 1H), 2.78-2.67 (m, 1H), 2.54-2.45 (m, 1H), 2.34-2.20 (m, 2H), 2.19-2.08 (m, 1H), 2.04-1.67 (m, 4H), 1.20-1.10 (m, 3H), 1.01-0.89 (m, 2H), 0.80-0.64 (m, 2H); MS(ES+) m/z 554.0, 556.0 (M+1).
Example 326
Synthesis of 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl) oxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00801##
[1345] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 4-(((3R,6S)-1-(2-chloro-4-fluorobenzyl)-6-methylpiperidin-3-yl)oxy)-5-cyc- lopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.11 g, 35%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.72-8.60 (m, 1H), 7.62-7.54 (m, 1H), 7.50-7.41 (m, 1H), 7.07-6.99 (m, 1H), 6.84-6.72 (m, 1H), 6.50-6.38 (m, 1H), 4.49-4.37 (m, 1H), 3.93-3.80 (m, 1H), 3.49-3.36 (m, 1H), 3.17-3.03 (m, 1H), 2.95-2.84 (m, 1H), 2.79-2.67 (m, 1H), 2.55-2.45 (m, 1H), 2.21-2.06 (m, 1H), 2.02-1.67 (m, 4H), 1.49-1.39 (m, 2H), 1.19-1.11 (m, 5H), 0.99-0.92 (m, 2H), 0.77-0.64 (m, 2H); MS(ES+) m/z 539.1, 541.1 (M+1).
Example 327
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-3-yl)oxy)benzamide
##STR00802##
[1346] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-3-yl)oxy)benzoate
##STR00803##
[1348] To a stirred solution of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (1.84 g, 6.26 mmol) in acetone (31 mL) was added triethylamine (4.36 mL, 31.30 mmol), followed by addition of (1-(trifluoromethyl)cyclopentyl)methyl trifluoromethanesulfonate (1.84 g, 6.26 mmol) (A. Wolniewicz et al., Journal of Fluorine Chemistry, 2001, 109, 95-102). The reaction mixture was stirred at reflux for 48 hours, cooled to ambient temperature, and concentrated in i vacuo. The residue was diluted with ethyl acetate (70 mL), and washed with saturated aqueous sodium bicarbonate solution (30 mL). The layers were separated and the aqueous layer was extracted with ethyl acetate (2.times.70 mL). The combined organic layers were washed with brine (80 mL), dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by column chromatography eluting with a gradient of ethyl acetate in hexanes to (0 to 25%) to give the title compound as a colorless oil (0.84 g, 30%): MS(ES+) m/z 444.4 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(1-(trifluoromethyl) cyclopentyl)methyl)piperidin-3-yl)oxy)benzoic acid
##STR00804##
[1350] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl)cyclopentyl)methyl)pipe- ridin-3-yl) oxy)benzoate, the title compound was obtained as a colorless solid (0.78 g, 90%): MS(ES+) m/z 430.2 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-3-yl)oxy)benzamide
##STR00805##
[1352] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (R)-5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl)cyclopentyl)methyl)- piperidin-3-yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.11 g, 17%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.71 (s, 1H), 7.60-7.52 (m, 1H), 6.66-6.54 (m, 1H), 4.45-4.29 (m, 1H), 3.41 (s, 3H), 3.08-2.98 (m, 1H), 2.76-2.63 (m, 1H), 2.60-2.38 (m, 4H), 2.12-1.94 (m, 2H), 1.91-1.55 (m, 11H), 0.98-0.86 (m, 2H), 0.72-0.60 (m, 2H); MS(ES+) m/z 507.2 (M+1).
Example 328
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclobutyl)methyl)piperidin-3-yl)oxy)benzamide
##STR00806##
[1353] Step 1. Preparation of (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclobutyl)methyl)piperidin-3-yl)oxy)benzoate
##STR00807##
[1355] To a mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (1.51 g, 5.15 mmol), (1-(trifluoromethyl)cyclobutyl)methyl trifluoromethanesulfonate (1.34 g, 4.68 mmol) (A. Wolniewicz, et al., Journal of Fluorine Chemistry, 2001, 109, 95-102), and potassium carbonate (0.97 g, 7.02 mmol) in cyclohexane (35 mL) was stirred as reflux for 96 hours. The mixture was cooled to ambient temperature, diluted with hexanes (100 mL), washed with saturated aqueous sodium bicarbonate solution (30 mL), brine (40 mL), dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by column chromatography eluting with a gradient of ethyl acetate in hexanes (0 to 25%) to give the title compound as a colorless oil (0.90 g, 39%): MS(ES+) m/z 430.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclobutyl)methyl)piperidin-3-yl)oxy)benzoic acid
##STR00808##
[1357] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl)cyclobutyl)methyl)piper- idin-3-yl) oxy)benzoate, the title compound was obtained as a colorless solid (0.58 g, 67%): MS(ES+) m/z 416.2 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclobutyl)methyl)piperidin-3-yl)oxy)benzamide
##STR00809##
[1359] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid With (R)-5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl)cyclobutyl)methyl)p- iperidin-3-yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.24 g, 35%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.70 (s, 1H), 7.61-7.50 (m, 1H), 6.67-6.52 (m, 1H), 4.49-4.31 (m, 1H), 3.41 (s, 3H), 3.05-2.96 (m, 1H), 2.75-2.64 (m, 1 H), 2.59 (s, 2H), 2.53-2.32 (m, 2H), 2.29-2.15 (m, 2H), 2.11-1.93 (m, 5H), 1.92-1.80 (m, 2H), 1.73-1.58 (m, 2H), 0.99-0.85 (m, 2H), 0.71-0.60 (m, 2H); MS(ES+) m/z 493.2 (M+1).
Example 329
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-4-yl)methoxy)benzamide trifluoroacetic acid salt
##STR00810##
[1360] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-4-yl)methoxy)benzoate
##STR00811##
[1362] Following the procedure as described in Example 328 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzonate, and to replace (1-(trifluoromethyl)cyclobutyl)methyl trifluoromethanesulfonate with (1-(trifluoromethyl)cyclopentyl)methyl trifluoromethanesulfonate, the title compound was obtained as a colorless oil (0.42 g, 54%): MS(ES+) m/z 500.3 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-4-yl)methoxy)benzoic acid
##STR00812##
[1364] Following the procedure as described in Example 3 step 3, and making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine-1-car- boxylate tert-butyl 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-4-yl)methoxy)benzoate, the title compound was obtained as a colorless solid (0.36 g, 97%): MS(ES+) m/z 444.3 (M+1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((1-(trifluoromethyl) cyclopentyl)methyl)piperidin-4-yl)methoxy)benzamide trifluoroacetic acid salt
##STR00813##
[1366] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((1-(trifluoromethyl)cyclopentyl)methyl)pipe- ridin-4-yl) methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.04 g, 17%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.82-8.64 (m, 1H), 7.64-7.53 (m, 1H), 6.63-6.49 (m, 1H), 4.00-3.68 (m, 4H), 3.42 (s, 3H), 3.28 (s, 2H), 3.06-2.70 (m, 2H), 2.28-1.69 (m, 14H), 1.01-0.88 (m, 2H), 0.71-0.59 (m, 2H); MS(ES+) m/z 521.2 (M+1).
Example 330
Synthesis of 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00814##
[1367] Step 1. Preparation of tert-butyl 4-(((2S,3S)-1-benzyl-2-methylpiperidin-3-yl) oxy)-5-chloro-2-fluorobenzoate
##STR00815##
[1369] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (2S,3S)-1-benzyl-2-methylpiperidin-3-ol (Peter H. Huy et al., Org. Lett., 2013, 15, 5178), the title compound was obtained as a pale yellow oil (0.15 g, 11%): MS(ES+) m/z 434.2, 436.2 (M+1).
Step 2. tert-butyl 4-(((2S,3S)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobe- nzoate
##STR00816##
[1371] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with tert-butyl 4-(((2S,3S)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoat- e, the title compound was obtained (0.343 g, 96%) as a colorless oil:.sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.39-7.18 (m, 6H), 6.57-6.48 (m, 1H), 4.51-4.38 (m, 1H), 3.74-3.52 (m, 2H), 3.32-3.18 (m, 1H), 2.59-2.32 (m, 2H), 2.04-1.93 (m, 1H), 1.91-1.58 (m, 4H), 1.54 (s, 9H), 1.07 (d, J=6.6 Hz, 3H), 0.91-0.79 (m, 2H), 0.67-0.52 (m, 2H).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((2S,3S)-2-methylpiperidin-3-yl)oxy)benzoate
##STR00817##
[1373] To a mixture of tert-butyl 4-((2S,3S)-1benzyl-2-methylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenz- oate (0.34 g, 0.78 mmol) and ammonium formate (0.49 g, 7.74 mmol) in methanol (16 mL) was added 10% palladium on carbon (0.04 g). The reaction mixture was stirred at reflux for 1 hour, cooled to ambient temperature, and filtered through a pad of diatomaceous earth. The filtrate was concentrated. The residue was diluted with ethyl acetate (100 mL), washed with aqueous saturated sodium bicarbonate solution (25 mL), water (25 mL), and brine (40 mL); dried over anhydrous magnesium sulfate; filtered and concentrated in vacuo to give the title compound as a colorless solid (0.22 g, 81%): MS(ES+) m/z 349.9 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl)o- xy)-2-fluorobenzoate
##STR00818##
[1375] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 4-(((2S,3S)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobe- nzoate, and to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained as a colorless oil (0.27 g, 84%): MS(ES+) m/z 508.1, 510.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluorobenzoic acid
##STR00819##
[1377] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.24 g, 99%): MS(ES+) m/z 452.1, 454.0 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide trifluoroacetic acid salt
##STR00820##
[1379] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((2S,3S)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl)o- xy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.93-8.61 (m, 1H), 7.70-7.53 (m, 1H), 7.50-7.35 (m, 3H), 6.89-6.67 (m, 1H), 5.02-4.82 (m, 1H), 4.46-4.12 (m, 2H), 4.09-3.85 (m, 1H), 3.47-3.33 (m, 3H), 3.31-2.87 (m, 2H), 2.18-1.86 (m, 5H), 1.62-1.39 (m, 3H), 1.03-0.83 (m, 2H), 0.73-0.54 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1).
Example 331
Synthesis of 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00821##
[1380] Step 1. Preparation of tert-butyl 4-(((2S,3R)-1-benzyl-2-methylpiperidin-3-yl) oxy)-5-chloro-2-fluorobenzoate
##STR00822##
[1382] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (2S,3R)-1-benzyl-2-methylpiperidin-3-ol (Peter H. Huy et al., Org. Lett., 2013, 15, 5178), the title compound was obtained as a colorless oil (0.26 g, 37%): MS(ES+) m/z 434.1, 436.1 (M+1).
Step 2. Preparation of tert-butyl 4-(((2S,3R)-1-benzyl-2-methylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate
##STR00823##
[1384] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with tert-butyl 4-(((2S,3R)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoat- e, the title compound was obtained as a pale yellow oil (0.21 g, 80%): MS(ES+) m/z 440.4 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((2S,3R)-2-methylpiperidin-3-yl)oxy)benzoate
##STR00824##
[1386] Following the procedure as described in Example 330 step 3, and making variations as required to replace tert-butyl 4-(((2S,3R)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobe- nzoate with tert-butyl 4-(((2S,3R)-1-benzyl-2-methylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobe- nzoate, the title compound was obtained as a colorless oil (0.16 g, 95%): MS(ES+) m/z 350.3 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl)o- xy)-2-fluorobenzoate
##STR00825##
[1388] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((2S,3R)-2-methylpiperidin-3-yl)oxy)benzoate, and to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained as a colorless oil (0.15 g, 68%): MS(ES+) m/z 508.1, 510.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluorobenzoic acid
##STR00826##
[1390] Following the procedure as described in Example 3 step 3, and making variation as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.08 g, 64%): MS(ES+) m/z 452.1, 454.0 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00827##
[1392] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((2S,3R)-1-(3,5-dichlorobenzyl)-2-methylpiperidin-3-yl)o- xy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.03 g, 27%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.88-8.66 (m, 1H), 7.67-7.58 (m, 1H), 7.52-7.36 (m, 3H), 6.79-6.64 (m, 1H), 4.89-4.58 (m, 2H), 4.12-3.90 (m, 1H), 3.54-3.21 (m, 5H), 3.00-2.75 (m, 1H), 2.44-1.93 (m, 5H), 1.84-1.64 (m, 3H), 1.06-0.90 (m, 2H), 0.80-0.54 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1).
Example 332
Synthesis of (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00828##
[1393] Step 1. Preparation of (R)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)piperidine-1-carboxylate
##STR00829##
[1395] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (R)-tert-butyl 3-(hydroxymethyl)piperidine-1-carboxylate, the title compound was obtained (13.11 g, 79%) as a colorless oil: MS(ES+) m/z 444.2, 446.2 (M+1).
Step 2. Preparation of (R)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)piperidine-1-carboxylate
##STR00830##
[1397] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (R)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)piperidine-1-- carboxylate, the title compound was obtained as a colorless solid (8.61 g, 65%): MS(ES+) m/z 450.3 (M+1).
Step 3. Preparation (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-ylmethoxy)benzoate
##STR00831##
[1399] Following the procedure as described in Example 34 step 1, and making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)piperidine-1-carboxylate with (R)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)piperidine-1-carboxylate, the title compound was obtained as a colorless oil (5.89 g, 99%): MS(ES+) m/z 308.2 (M+1).
Step 4. Preparation of (R)-methyl 4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00832##
[1401] Following the procedure as described in Example 34 step 2, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-ylmethoxy)benzoate, and to replace 1-(bromomethyl)-4-fluoro-2-(trifluoromethyl)benzene with 1-(bromomethyl)-2-chloro-4-fluorobenzene, the title compound was obtained as a colorless oil (4.06 g, 94%): MS(ES+) m/z 450.1, 452.1 (M+1).
Step 5. Preparation of (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00833##
[1403] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (R)-methyl 4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-2-fl- uorobenzoate, the title compound was obtained as a colorless solid (3.80 g, 97%): MS(ES+) m/z 436.1, 438.1 (M+1).
Step 6. Preparation of (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00834##
[1405] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-- 2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.25 g, 35%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.11.53 (br s, 1H), 7.59-7.48 (m, 1H), 7.47-7.39 (m, 1H), 7.25-7.09 (m, 2H), 6.96-6.85 (m, 1H), 4.09-3.85 (m, 2H), 3.78-3.54 (m, 2H), 3.26 (s, 3H), 3.01-2.78 (m, 2H), 2.36-2.02 (m, 3H), 1.91-1.47 (m, 4H), 1.29-1.12 (m, 1H), 0.86-0.68 (m, 2H), 0.64-0.52 (m, 2H); MS(ES+) m/z 513.2, 515.1 (M+1).
Example 333
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-- yl) methoxy)-5-cyclopropyl-2-fluorobenzamide
##STR00835##
[1407] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (R)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-- 2-fluorobenzoic acid, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.14 g, 19%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.65 (s, 1H), 7.62-7.55 (m, 1H), 7.47-7.38 (m, 1H), 7.12-7.04 (m, 1H), 6.96-6.86 (m, 1H), 6.61-6.51 (m, 1H), 4.33-4.14 (m, 4H), 4.01-3.82 (m, 2H), 3.64-3.49 (m, 2H), 3.00-2.86 (m, 1H), 2.83-2.69 (m, 1H), 2.37-2.03 (m, 5H), 1.99-1.61 (m, 4H), 1.37-1.17 (m, 1H), 0.97-0.75 (m, 2H), 0.71-0.53 (m, 2H); MS(ES+) m/z 554.2, 556.2 (M+1).
Example 334
Synthesis of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-flu- oro-N-(methylsulfonyl)benzamide
##STR00836##
[1408] Step 1. Preparation of (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-fluorob- enzoate
##STR00837##
[1410] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-ylmethoxy)benzoate, and to replace 1,3-dichloro-5-(1-chloroethyl) benzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained as a colorless oil (0.68 g, 89%): MS(ES+) m/z 466.2, 468.1 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluorobenzoic acid
##STR00838##
[1412] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-fluorob- enzoate, the title compound was obtained as a colorless solid (0.60 g, 91%): MS(ES+) m/z 452.1, 454.1 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00839##
[1414] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-flu- orobenzoic acid, the title compound was obtained as a colorless solid (0.14 g, 39%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.68 (s, 1H), 7.59-7.53 (m, 3H), 7.25-7.18 (m, 3H), 6.59-6.52 (m, 1H), 3.96-3.82 (m, 2H), 3.53-3.35 (m, 5H), 2.96-2.84 (m, 1H), 2.81-2.67 (m, 1H), 2.33-2.06 (m, 2H), 2.05-1.63 (m, 5H), 1.36-1.16 (m, 1H), 0.95-0.75 (m, 2H), 0.69-0.51 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1).
Example 335
Synthesis of (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00840##
[1415] Step 1. Preparation of (S)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)piperidine-1-carboxylate
##STR00841##
[1417] Following the procedure as described in Example 1 step 1, and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (S)-tert-butyl 3-(hydroxymethyl)piperidine-1-carboxylate, the title compound was obtained as a colorless oil (12.94 g, 78%): MS(ES+) m/z 444.2, 446.2 (M+1).
Step 2. Preparation of (S)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)piperidine-1-carboxylate
##STR00842##
[1419] Following the procedure as described in Example 1 step 2, and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (S)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)piperidine-1-- carboxylate, the title compound was obtained as a colorless solid (10.22 g, 78%): MS(ES+) m/z 450.3 (M+1).
Step 3. Preparation of (S)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3ylmethoxy)benzoate
##STR00843##
[1421] Following the procedure as described in Example 34 step 1, making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)piperidine-1-carboxylate with (R)-tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)piperidine-1-carboxylate, the title compound was obtained as a pale yellow oil (6.99 g, 99%): MS(ES+) m/z 308.2 (M+1).
Step 4. Preparation of (S)-methyl 4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00844##
[1423] Following the procedure as described in Example 34 step 2, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (S)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yl)methoxy)benzoate, and to replace 1-(bromomethyl)-4-fluoro-2-(trifluoromethyl) benzene with 1-(bromomethyl)-2- chloro-4-fluorobenzene, the title compound was obtained as a colorless oil (1.85 g, 42%): MS(ES+) m/z 450.2, 452.2 (M+1).
Step 5. Preparation of (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00845##
[1425] Following the procedure as described in Example 50 step 2, an making variations as required to replace 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (S)-methyl 4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-2-fl- uorobenzoate, the title compound was obtained as a colorless solid (1.78 g, 99%): MS(ES+) m/z 436.2, 438.1 (M+1).
Step 6. Preparation of (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00846##
[1427] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-- 2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.19 g, 37%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.53 (br s, 1H), 7.58-7.48 (m, 1H), 7.47-7.38 (m, 1H), 7.24-7.09 (m, 2H), 6.96-6.86 (m, 1H), 4.08-3.85 (m, 2H), 3.79-3.56 (m, 2H), 3.26 (s, 3H), 3.01-2.78 (m, 2H), 2.34-2.21 (m, 1H), 2.20-2.02 (m, 2H), 1.92-1.50 (m, 4H), 1.31-1.12 (m, 1H), 0.85-0.69 (m, 2H), 0.65-0.53 (m, 2H); MS(ES+) m/z 513.2, 515.2 (M+1).
Example 336
Synthesis of (S)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00847##
[1428] Step 1. Preparation of (S)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluorobenzoate
##STR00848##
[1430] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (S)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yl)methoxy)benzoate, and to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained as a colorless oil (0.65 g, 86%): MS(ES+) m/z 466.2, 468.2 (M+1).
Step 2. Preparation of (S)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluorobenzoic acid
##STR00849##
[1432] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with (S)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-fluorob- enzoate, the title compound was obtained as a colorless solid (0.52 g, 83%): MS(ES+) m/z 452.1, 454.2 (M+1).
Step 3. Preparation of (S)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR00850##
[1434] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (S)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)methoxy)-2-flu- orobenzoic acid, the title compound was obtained as a colorless solid (0.07 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.78 (s, 1H), 7.60-7.50 (m, 1H), 7.24-7.20 (m, 3H), 6.61-6.50 (m, 1H), 3.99-3.82 (m, 2H), 3.55-3.32 (m, 5H), 2.97-2.83 (m, 1H), 2.82-2.67 (m, 1H), 2.32-2.05 (m, 2H), 2.05-1.60 (m, 5H), 1.33-1.15 (m, 1H), 0.95-0.75 (m, 2H), 0.68-0.48 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1).
Example 337 and Example 338
Synthesis of 5-cyclopropyl-4-(((S)-1-((R)-1-(3,5-dichlorophenyl)ethyl) piperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00851##
[1435] And 5-cyclopropyl-4-(((S)-1-((S)-1-(3,5-dichlorophenyl)ethyl) piperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00852##
[1436] Step 1. Preparation of methyl 5-cyclopropyl-4-(((3S)-1-(1-(3,5-dichlorophenyl)ethyl) piperidin-3-yl)methoxy)-2-fluorobenzoate
##STR00853##
[1438] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with (S)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-ylmethoxy)benzoate, the title compound was obtained as a pale yellow oil (1.92 g, 75%): MS(ES+) m/z 480.2, 482.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((3S)-1-(1-(3,5-dichlorophenyl)ethyl) piperidin-3-yl)methoxy)-2-fluorobenzoic acid
##STR00854##
[1440] Following the procedure as described in Example 50 step 2, and making variations as required to replace methyl 4-((1-(3-fluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- 2-fluorobenzoate with methyl 5-cyclopropyl-4-(((3S)-1-(1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)meth- oxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.72 g, 92%): MS(ES+) m/z 452.1, 454.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-(((S)-1-((R)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00855##
[1441] And 5-cyclopropyl-4-(((S)-1-((S)-1-(3,5 -dichlorophenyl)ethyl)piperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00856##
[1443] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((3S)-1-(3,5-dichlorophenyl)ethyl)piperidin-3-yl)methoxy- )-2-fluorobenzoic acid, a mixture of diastereomers was obtained. The mixture of diastereomers was then purified by preparative HPLC. The first eluting fraction was arbitrarily assigned as 5-cyclopropyl-4-(((S)-1-((R)-1-(3,5-dichlorophenyl) ethyl)piperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt (0.03 g, 4%): .sup.1H NMR (300 MHz, DMSO-d.sub.6, 1 drop of D.sub.2O) .delta.7.80-7.75 (m, 1H), 7.68-7.63 (m, 2H), 7.19-7.12 (m, 1H), 6.98-6.90 (m, 1H), 4.62-4.51 (m, 1H), 4.11-4.04 (m, 1H), 3.94-3.84 (m, 1H), 3.79-3.67 (m, 1H), 3.36-3.28 (m, 4H), 2.85-2.63 (m, 2H), 2.36-2.20 (m, 1H), 2.04-1.91 (m, 1H), 1.88-1.73 (m, 2H), 1.73-1.58 (m, 4H), 1.39-1.26 (m, 1H), 0.80-0.51 (m, 4H); MS(ES+) m/z 543.2, 545.1 (M+1).
[1444] The second eluting fraction was arbitrarily assigned as 5-cyclopropyl-4-(((S)-1-((S)-1-(3,5-dichlorophenyl) ethyl)piperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt (0.03 g, 4%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.13.00 (br s, 1H), 8.81-8.64 (m, 1H), 7.66-7.57 (m, 1H), 7.50-7.43 (m, 1H), 7.37-7.30 (m, 2H), 6.58-6.49 (m, 1H), 4.51-4.36 (m, 1H), 4.07-3.77 (m, 3H), 3.64-3.52 (m, 1H), 3.41 (s, 3H), 2.83-2.65 (m, 1H), 2.58-2.39 (m, 2H), 2.26-2.09 (m, 1H), 2.08-1.91 (m, 2H), 1.91-1.71 (m, 4H), 1.50-1.32 (m, 1H), 0.92-0.76 (m, 2H), 0.71-0.50 (m, 2H); MS(ES+) m/z 543.2, 545.1 (M+1).
Example 339
Synthesis of (S)-N-(azetidin-1-ylsulfonyl)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-- yl) methoxy)-5-cyclopropyl-2-fluorobenzamide, trifluoroacetic acid salt
##STR00857##
[1446] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-- 2-fluorobenzoic acid, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.05 g, 7%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.12.89 (br s, 1H), 8.79-8.49 (m, 1H), 7.79-7.69 (m, 1H), 7.67-7.58 (m, 1H), 7.24-7.17 (m, 1H), 7.17-7.06 (m, 1H), 6.60-6.45 (m, 1H), 4.43 (s, 2H), 4.32-4.16 (m, 4H), 4.08-3.84 (m, 2H), 3.81-3.57 (m, 2H), 2.83-2.63 (m, 2H), 2.34-1.82 (m, 7H), 1.66-1.39 (m, 1H), 0.98-0.81 (m, 2H), 0.73-0.55 (m, 2H); MS(ES+) m/z 554.1, 556.1 (M+1).
Example 340
Synthesis of (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00858##
[1448] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)methoxy)-5-cyclopropyl-- 2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.17 g, 33%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.13.17 (br s, 1H), 8.78-8.58 (m, 1H), 7.80-7.70 (m, 1H), 7.64-7.56 (m, 1H), 7.22-7.16 (m, 1H), 7.14-7.05 (m, 1H), 6.58-6.44 (m, 1H), 4.41 (s, 2H), 4.05-3.81 (m, 2H), 3.77-3.55 (m, 2H), 3.14-3.01 (m, 1H), 2.82-2.60 (m, 3H), 2.22-1.79 (m, 4H), 1.58-1.36 (m, 3H), 1.20-1.05 (m, 2H), 0.96-0.79 (m, 2H), 0.72-0.50 (m, 2H); MS(ES+) m/z 539.2, 541.2 (M+1).
Example 341
Synthesis 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperid- in-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00859##
[1449] Step 1. Preparation of (2R,5R)-benzyl 5-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-2-methylpiperidin-1-carboxylate
##STR00860##
[1451] Following the procedure as described in Example 1 step 1, and making variations as required to replace (R)-1-benzylpiperidin-3-ol with (2R,5R)-benzyl 5-(hydroxymethyl)-2-methylpiperidine-1-carboxylate (WO 2010/048010 A1), the title compound was obtained as a colorless oil (1.61 g, 66%): MS(ES+) m/z 492.2, 494.1 (M+1).
Step 2. Preparation of (2R,5R)-benzyl 5-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-2-methylpiperidine-1-carboxylate
##STR00861##
[1453] Following the procedure as described in Example 1 step 2, and making variations as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (2R,5R)-benzyl 5-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)-2-methylpipe- ridine-1-carboxylate, the title compound was obtained as a colorless solid (1.46 g, 90%): MS(ES+) m/z 498.3 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6R)-6-methylpiperidin-3-yl)methoxy)benzoat- e
##STR00862##
[1455] Following the procedure as described in Example 52 step 4, and making variation as required to replace (2R,5R)-benzyl 5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5- fluorophenoxy)-2-methylpiperidine-1-carboxylate with (2R,5R)-benzyl 5-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-2-methylpiperidine-1-carboxylate, the title compound was obtained as a colorless oil (1.00 g, 94%): MS(ES+) m/z 364.3 (M+1).
Step 4. Preparation of tert-butyl 5cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperidin-3-yl)me- thoxy)-2-fluorobenzoate
##STR00863##
[1457] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6R)-6-methylpiperidin-3-yl)methoxy)benzoat- e, and to replace 1,3-dichloro-5-(1-chloroethyl)benzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained a colorless oil (0.71 g, 98%): MS(ES+) m/z 522.2, 524.2 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperidin-3-yl) methoxy)-2-fluorobenzoic acid
##STR00864##
[1459] Following the procedure as described in Example 3 step 3, and making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperidin-3-yl) methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.56 g, 88%): MS(ES+) m/z 466.1, 468.1 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperidin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00865##
[1461] Following the procedure as described in Example 17 step 2, and making variations required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorobenzyl)-6-methylpiperidin-3-yl)m- ethoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.09 g, 40%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.8.82-8.62 (m, 1H), 7.67-7.52 (m, 1H), 7.51-7.42 (m, 1H), 7.42-7.34 (m, 2H), 6.57-6.42 (m, 1H), 4.76-4.57 (m, 1H), 4.03-3.76 (m, 3H), 3.53-3.44 (m, 1H), 3.41 (s, 3H), 3.04-2.71 (m, 2H), 2.55-2.37 (m, 1H), 2.28-2.10 (m, 1H), 2.09-1.95 (m, 2H), 1.80-1.61 (m, 4H), 1.56-1.42 (m, 1H), 0.87-0.75 (m, 2H), 0.66-0.49 (m, 2H) MS(ES+) m/z 543.2, 545.2 (M+1).
Example 342
Synthesis of 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide trifluoroacetic acid salt (arbitrarily assigned)
##STR00866##
[1462] Step 1. Preparation of tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl) ethyl-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoate
##STR00867##
[1463] And tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-(3,5-dichlorophenyl)ethyl)-6-methylpiperidin-- 3-yl) methoxy)-2-fluorobenzoate
##STR00868##
[1465] Following the procedure as described in Example 50 step 1, and making variations as required to replace (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((3R,6R)-6-methylpiperidin-3-yl)methoxy)benzoat- e. The mixture of diastereomers was separated by column chromatography eluting with a gradient of methanol in dichloromethane (0 to 5%). The first eluting fraction was arbitrarily assigned as tert-butyl 5-cyclopropyl-4-(((3 R,6R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-6-methylpiperidin-3-yl) methoxy)-2-fluorobenzoate (0.23 g, 32%) as a pale yellow oil: MS(ES+) m/z 536.0, 538.2 (M+1).
[1466] The second eluting fraction was arbitrarily assigned as tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoate (0.23 g, 32%) as a pale yellow oil: MS(ES+) m/z 536.1, 538.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoic acid
##STR00869##
[1468] Following the procedure as described in Example 3 step 3, and making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.20 g, 99%): MS(ES+) m/z 480.1, 482.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzami- de, trifluoroacetic acid salt
##STR00870##
[1470] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.05 g, 38%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.12.12 (br s, 1H), 8.91-8.53 (m, 1H), 7.65-7.57 (m, 1H), 7.53-7.48 (m, 2H), 7.45-7.39 (m, 1H), 6.56-6.44 (m, 1H), 5.05-4.92 (m, 1H), 4.02-3.76 (m, 2H), 3.41 (s, 3H), 3.27-3.08 (m, 2H), 2.88-2.71 (m, 1H), 2.72-2.56 (m, 2H), 2.36-2.20 (m, 1H), 2.09-1.92 (m, 2H), 1.83-1.49 (m, 7H), 0.94-0.75 (m, 2H), 0.71-0.50 (m, 2H); MS(ES+) m/z 557.0, 559.0 (M+1).
Example 343
Synthesis of 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzami- de, trifluoroacetic acid salt
##STR00871##
[1471] Step 1. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoic acid
##STR00872##
[1473] Following the procedure as described in Example 3 step 3, and making variations as required to replace (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine-1-car- boxylate with tert-butyl 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.21 g, 99%): MS(ES+) m/z 480.1, 482.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzami- de, trifluoroacetic acid salt
##STR00873##
[1475] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoroben- zoic acid with 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.05 g, 28%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.13.00 (br s, 1H), 8.91-8.59 (m, 1H), 7.72-7.59 (m, 1H), 7.54-7.45 (m, 1H), 7.31-7.27 (m, 2H), 6.61-6.51 (m, 1H), 5.04-4.91 (m, 1H), 4.14-3.90 (m, 2H), 3.85-3.73 (m, 1H), 3.42 (s, 3H), 2.96-2.64 (m, 2H), 2.60-2.42 (m, 1H), 2.26-2.06 (m, 1H), 2.04-1.65 (m, 9H), 1.56-1.36 (m, 1H), 0.87-0.73 (m, 2H), 0.67-0.54 (m, 2H); MS(ES+) m/z 557.0, 559.0 (M+1).
Example 344
Synthesis of 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-N-(ethylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00874##
[1477] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((3R,6R)-1-((S)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-2-fluorobenzoic acid, and to replace methanesulfonamide with ethanesulfonamide, the title compound was obtained as a colorless solid (0.05 g, 31%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.80 (br s, 1H), 9.19-9.02 (m, 1H), 7.84-7.64 (m, 3H), 7.21-7.10 (m, 1H), 6.98-6.84 (m, 1H), 5.16-4.95 (m, 1H), 4.16-3.95 (m, 1H), 3.87-3.68 (m, 1H), 3.50-3.37 (m, 2H), 3.11-2.85 (m, 2H), 2.30-1.89 (m, 3H), 1.83-1.29 (m, 10H), 1.25-1.14 (m, 3H), 0.79-0.35 (m, 4H); MS(ES+) m/z 571.1, 573.1 (M+1).
Example 345
Synthesis of 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl) ethyl)-6-methylpiperidin-3-yl)methoxy)-N-(ethylsulfonyl)-2-fluorobenzamid- e, trifluoroacetic acid salt
##STR00875##
[1479] Following the procedure as described in Example 17 step 2, and making variations as required to replace (R)-5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)piperidin-3-yl)-oxy)-2-fluoro- benzoic acid with 5-cyclopropyl-4-(((3R,6R)-1-((R)-1-(3,5-dichlorophenyl)ethyl)-6-methylpip- eridin-3-yl) methoxy)-2-fluorobenzoic acid, and to replace to methanesulfonamide with ethanesulfonamide, the title compound was obtained as a colorless solid (0.02 g, 17%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.84 (br s, 1H), 9.71-9.51 (m, 1H), 7.86-7.60 (m, 3H), 7.24-7.14 (m, 1H), 7.03-6.90 (m, 1H), 5.21-4.98 (m, 1H), 4.15-3.99 (m, 1H), 3.94-3.81 (m, 1H), 3.52-3.39 (m, 2H), 2.97-2.80 (m, 1H), 2.47-2.22 (m, 2H), 2.04-1.35 (m, 11H), 1.29-1.18 (m, 3H), 0.82-0.55 (m, 4H); MS(ES+) m/z 571.1, 573.1 (M+1).
Example 346
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)benzyl)-4-fluoropiperidin-4-y- l) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide trifluoroacetic acid salt
##STR00876##
[1480] Step 1. Preparation of tert-Butyl 4-fluoro-4-(hydroxymethyl)piperidine-1-carboxylate
##STR00877##
[1482] To a solution of 1-(tert-butoxycarbonyl)-4-fluoropiperidine-4-carboxylic acid (5.00 g, 20.20 mmol) in tetrahydrofuran (100 mL) was added a solution of borane tetrahydrofuran complex (30.3 mL, 30.30 mmol, 1.0 M solution in tetrahydrofuran). The reaction mixture was refluxed for 16 hours, and then another 24 mL of borane tetrahydrofuran complex was added and continued to reflux for another 16 hours. After cooling to ambient temperature the reaction mixture poured onto ice-cold water (50 mL) and saturated ammonium chloride (100 mL), and extracted with ethyl acetate (3.times.100 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated to afford the title compound (4.60 g, 98%). Which was used in the next step without further purification; MS(ES+) m/z 234.1 (M +1).
Step 2. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate
##STR00878##
[1484] To a mixture of tert-butyl 4-fluoro-4-(hydroxymethyl)piperidine-1-carboxylate (4.60 g, 19.70 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (4.89 g, 19.70 mmol) in dimethyl sulfoxide (50 mL) was added cesium carbonate (9.65 g, 29.60 mmol) and the reaction mixture was heated at 80.degree. C. for 4 hours. After cooling to an ambient temperature, water (50 mL) was added, and then extracted with ethyl acetate (3.times.100 mL). The organic layer was washed with brine (2.times.50 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo and the residue was purified by silica gel column chromatography eluting with gradient of 0-30% ethyl acetate in hexanes to afford the title compound as a colorless gum (6.60 g, 73%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.82 (d, J=7.6 Hz, 1H), 6.58 (d, J=11.8 Hz, 1H), 3.90-4.10 (m, 3H), 3.07 (t, J=12.4 Hz, 2H), 1.98-1.89 (m, 3H), 1.84-1.56 (m, 2H), 1.51 (s, (H), 1.40 (s, 9H); MS(ES+) m/z 464.1, 462.1 (M+1).
Step 3. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate
##STR00879##
[1486] To a mixture of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)-4-fluoropipe- ridin-1-carboxylate (6.60 g, 14.30 mmol), cyclopropylboronic acid (3.07 g, 35.80 mmol), potassium phosphate (12.2 g, 57.3 mmol) and tricyclohexylphosphine tetrafluoroborate (0.79 g, 2.15 mmol) in toluene (120 mL) and water (12 mL) under a nitrogen atmosphere was added palladium acetate (0.32 g, 1.43 mmol). The reaction mixture was heated at 100.degree. C. for 16 hours and cooled to ambient temperature. To the reaction mixture was added water (20 mL) and extracted with ethyl acetate (3.times.100 mL). The combined organic extracts was washed with brine, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo and the residue was purified by column chromatography eluting with 5% ethyl acetate in hexanes to afford the title compound as a colorless gum (4.00 g, 60%): MS(ES+) m/z 468.6 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl) methoxy)benzoate hydrochloride
##STR00880##
[1488] To a solution of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate (4.00 g, 8.50 mmol) in dioxane (12 mL) was added a solution of hydrogen chloride in dioxane (4M, 4.0 mL, 16.00 mmol). The reaction solution was stirred at an ambient temperature for 3 hours. The title compound precipitated out from solution was collected by filtration. The filtrate was concentrated to afford another portion of product (1.90 g in total, 61%) as a pale yellow solid: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.97 (br s, 1H), 7.26 (d, J=8.4 Hz, 1H), 6.93 (d, J =12.9 Hz, 1H), 4.23 (d, J=19.9 Hz, 2H), 3.31-3.20 (m, 2H), 3.11-2.92 (m, 2H), 2.15-2.06 (m, 2H), 2.04-1.92 (m, 2H), 1.47 (s, 9H), 0.91-0.85 (m, 2H), 0.60-0.54 (m, 2H); MS(ES+) m/z 368.2 (M+1).
Step 5. Preparation of tert-butyl 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl) benzyl)-4-fluoropiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00881##
[1490] A mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate hydrochloride (0.29 g, 0.70 mmol), 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene (0.18 g, 0.70 mmol) and potassium carbonate (0.24 g, 2.10 mmol) in N,N-dimethylformamide (2 mL) was heated at 90.degree. C. for 16 hours. The solid was filtered off and the filtrate was subjected to column chromatography eluting with 40% ethyl acetate in hexanes to afford the title compound as a gum (0.29 g, 71%): MS(ES+) m/z 578.2, 580.2 (M+1).
Step 6. Preparation of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl) benzyl)-4-fluoropiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00882##
[1492] To a solution of tert-butyl 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (0.29 g, 0.50 mmol) in dichloromethane (5 mL) was added trifluoroacetic acid (2 mL). The reaction mixture was stirred for 3 hours and concentrated invacuo to afford the title compound as a colorless solid (0.20 g, 76%): MS(ES-) m/z 520.2, 518.2 (M-1).
Step 7. Preparation of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00883##
[1494] To a mixture of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid (0.10 g, 0.19 mmol), 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.068 g, 0.44 mmol), 4-dimethylaminopyridine (0.054 g, 0.44 mmol) was added methanesulfonamide (0.027 g, 0.29 mmol) in dichloromethane (3 mL). After stirring at ambient temperature for 16 hours, the reaction mixture was diluted with ethyl acetate (5 mL), washed with brine (10 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo and the residue was purified by preparative-HPLC to afford the title compound as a colorless solid (0.05 g, 44%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.91 (br s, 1H), 10.24 (br s, 1H), 8.22 (d, J=3 Hz, 1H), 8.00 (d, J=3 Hz, 1H), 7.13 (d, J=9 Hz, 1H), 6.98 (d, J=12 Hz, 1H), 4.65-4.10 (m, 5H), 3.53-3.01 (m, 6H), 2.32-1.81 (m, 5H), 0.94-0.78 (m, 2H), 0.72-0.56 (m, 2H); MS (ES+) m/z 599.1, 601.3 (M+1).
Example 347
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00884##
[1496] Following the procedure as described in Example 346 step 7 and making non-critical variation as required to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.04 g, 35%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89 (br s, 1H), 10.10 (br s, 1H), 8.28 (d, J=6 Hz, 1H), 8.05 (d, J=6 Hz, 1H), 7.15 (d, J=9 Hz, 1H), 7.02 (d, J=15 Hz, 1H), 4.60-4.20 (m, 5H), 3.59-3.16 (m, 4H), 3.14-2.99 (m, 1H), 2.37-1.88 (m, 5H), 1.18-1.04 (m, 3H), 0.96-0.83 (m, 2H), 0.74-0.64 (m, 2H); MS(ES+) m/z 625.1, 627.3 (M+1).
Example 348
Synthesis of 4-((1-(3-chloro-4-(trifluoromethoxy)benzyl)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00885##
[1497] Step 1. Preparation of tert-butyl 4-((1-(3-chloro-4-(trifluoromethoxy)benzyl)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00886##
[1499] To a solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate (0.250 g, 0.62 mmol) and 3-chloro-4-(trifluoromethoxy)benzaldehyde (0.17 g, 0.74 mmol) in tetrahydrofuran (5 mL) was added sodium triacetoxyborohydride (0.30 g, 1.40 mmol). The reaction mixture was stirred at ambient temperature for 16 hours and concentrated invacuo. The residue was subjected to column chromatography to afford the title compound as a colorless gum (0.10 g, 23%): MS(ES+) m/z 576.2, 578.2 (M+1).
Step 2. Preparation of 4-((1-(3-chloro-4-(trifluoromethoxy)benzyl)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00887##
[1501] Following the procedure as described in Example 346 step 6, and making non-critical variation as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)-4-fluor- opiperidine-1-carboxylate with tert-butyl 4-((1-(3-chloro-4-(trifluoromethoxy)benzyl)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.12 g, quant. yield): MS(ES+) m/z 522.2, 520.2 (M+1)
Step 3. Preparation of 4-((1-(3-chloro-4-(trifluoromethoxy)benzyl-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide trifluoroacetic acid salt
##STR00888##
[1503] Following the procedure as described in Example 346 step 7 and making non-critical variation as respired to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.02 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta..alpha.11.89 (br s, 1H), 10.00 (br s, 1H), 7.90 (s, 1H), 7.75-7.56 (m, 2H), 7.15 (d, J=9 Hz, 1H), 7.03 (d, J=12 Hz, 1H), 4.54-4.2 (m, 4H), 3.25-2.97 (m, 2H), 2.34-1.88 (m, 3H), 2.32-1.85 (m, 5H), 1.21-1.03 (m, 4H), 0.95-0.83 (m, 2H), 0.75-0.61 (m, 2H); MS(ES+) m/z 625.0, 623.1 (M+1).
Example 348
Synthesis of 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00889##
[1505] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin-4-fluoropiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(5-chloro-2-(trifluoromethyl)piperidin-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.02 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.95 (br, s, 1H), 8.13 (br, s, 1H), 7.96-7.79 (m, 2H), 7.16 (d, J=6 Hz, 1H), 7.02 (d, J=15 Hz, 1H), 4.68-4.41 (m, 2H), 4.39-4.18 (m, 3H), 3.46-3.23 (m, 2H), 3.34 (s, 3H), 2.33-1.93 (m, 6H), 0.95-0.85 (m, 2H), 0.74-0.65 (m, 2H); MS(ES+) m/z 583.1, 581.2 (M+1).
Example 349
Synthesis of (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00890##
[1506] Step 1. Preparation of (S)-tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoate
##STR00891##
[1508] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate, the title compound was obtained as a colorless solid (0.40 g, 60%): MS(ES+) m/z 542.2, 540.2 (M+1).
Step 2. Preparation of (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy)-2-fluorobenzoic acid
##STR00892##
[1510] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)-4-fluor- opiperidine-1-carboxylate with (R)-tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4-yl) methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.20 g, 41%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 12.9 (br s, 1H), 7.73 (s, 1H), 7.61 (s, 2H), 7.30 (d, J=8.4 Hz, 1H), 6.93 (d, J=12.9 Hz, 1H), 4.73-4.53 (m, 1H), 4.23 (d, J=20.3 Hz, 2H), 3.81-3.3.63 (m, 1H), 3.26-3.14 (m, 1H), 3.09-2.90 (m, 2H), 2.15-2.06 (m, 2H), 2.04-1.92 (m, 2H), 1.64 (brs, 3H), 0.89-0.83 (m, 2H), 0.60-0.55 (m, 2H); MS(ES+) m/z 486.3, 484.1 (M+1).
Step 3. Preparation of (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00893##
[1512] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin-4-fluoropiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (S)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoic acid, the title compound (0.05 g, 42% yield) was obtained as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.95 (br s, 1H), 7.87-7.54 (m, 3H), 7.16 (d, J=9 Hz, 1H), 7.02 (d, J=12 Hz, 1H), 4.57 (br, s, 1H), 4.39-4.18 (m, 2H), 3.52 (br, s, 6H), 2.36-1.85 (m, 8H), 1.33-1.14 (m, 1H), 0.89-0.80 (m, 2H), 0.74-0.61 (m, 2H); MS (ES+) m/z 563.3, 561.1 (M+1).
Example 350
Synthesis of (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzamide
##STR00894##
[1514] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.07 g, 57%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (br s, 1H), 10.01 (br s, 1H), 7.73 (s, 1H), 7.615 (d, J=3.0 Hz, 2H), 7.12 (d, J=6.0 Hz, 1H), 6.99 (d, J=15.0 Hz, 1H), 4.6 (brs, 1H), 4.24 (d, J=18.0 Hz, 2H), 3.81-3.62 (m, 1H), 3.27-3.11 (m, 1H), 3.09-2.89 (m, 2H), 2.36-1.83 (m, 5H), 1.73-1.51 (m, 3H), 1.16-1.01 (m, 3H), 0.91-0.78 (m, 2H), 0.71-0.60 (m, 2H); MS(ES+) m/z 589.3, 587.1 (M+1).
Example 351
Synthesis of (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR00895##
[1515] Step 1. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoate
##STR00896##
[1517] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with (S)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate, the title compound was obtained (0.04 g, 60%): MS(ES+) m/z 542.2, 540.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy)-2-fluorobenzoic acid
##STR00897##
[1519] Following the procedure as described in Example 346 step 6 and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with (S)-tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.12 g, quant. yield): MS(ES+) m/z 484.1, 486.3 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)ethyl)-4-fluoropiperidin-4- -yl) methoxy-2-fluoro-N-(methylsulfonyl)benzamide
##STR00898##
[1521] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (R)-5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl) ethyl)-4-fluoropiperidin-4-yl)methoxy)-2-fluorobenzoic acid, the compound was obtained as a colorless solid (0.01 g, 7%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.94 (br s, 1H), 7.87-7.54 (m, 3H), 7.16 (d, J=9.0 Hz, 1H), 7.02 (d, J=12.0 Hz, 1H), 4.57 (brs, 1H), 4.39-4.18 (m, 2H), 3.52 (brs, 6H), 2.36-1.85 (m, 8H), 1.33-1.14 (m, 1H), 0.89-0.80 (m, 2H), 0.74-0.61 (m, 2H); MS(ES+) m/z 563.3, 561.1 (M+1).
Example 352
Synthesis of 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00899##
[1522] Step 1. Preparation of tert-butyl 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00900##
[1524] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate and 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 3,3'(bromomethylene)bis(chlorobenzene), the title compound was obtained as a colorless solid (0.58 g, 35%): MS(ES+) m/z 586.1, 584.1 (M+1).
Step 2. Preparation of 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00901##
[1526] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with tert-butyl 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-- fluorobenzoate, the title compound was obtained as a colorless solid (0.31 g, 54%): MS(ES+) m/z 530.1, 528.1 (M+1).
Step 3. Preparation of 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00902##
[1528] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.01 g, 38%): .sup.1H NMR (300 MHz, DMSO-d.sub.61% D.sub.2O) .delta.7.72 (s, 2H), 7.63-7.61 (m, 2H), 7.54-7.46 (m, 4H), 7.10 (d, J=8.3 Hz, 1H), 6.93 (d, J=12.9 Hz, 1H), 5.67 (s, 1H), 3.98 (d, J=4.6 Hz, 2H), 3.30 (s, 3H), 3.25-3.21 (m, 2H), 3.09-2.98 (m, 2H), 2.21-2.08 (m, 1H), 2.04-1.97 (m, 3H), 1.72-1.64 (m, 2H), 0.89-0.83 (m, 2H), 0.65-0.60 (m, 2H); MS(ES+) m/z 605.1, 607.1 (M+1).
Example 353
Synthesis of 5-cyclopropyl-4-(((1R,3S,5S)-8-((R)-1-(3,5-dichlorophenyl)ethyl)-8-azabic- yclo[3.2.1]octan-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00903##
[1530] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl) benzene with (S)-1,3-dichloro-5-(1-chloroethyl)benzene, the title compound was obtained as a colorless solid (0.03 g, 54%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.89 (br s, 11H), 9.58 (br s, 1H), 7.84-7.74 (m, 2H), 7.73-7.68 (m, 1H), 7.09 (d, J=8.2 Hz, 1H), 6.98 (d, J=12.9 Hz, 1H), 4.54-4.36 (m, 1H), 4.31-4.17 (m, 1H), 4.16-4.06 (m, 1H), 3.96-3.80 (m, 2H), 3.73-3.42 (m, 3H), 3.43-3.34 (m, 1H), 2.39-2.22 (m, 1H), 2.17-1.73 (m, 7H), 1.72-1.52 (m, 4H), 0.91-0.79 (m, 2H), 0.70-0.59 (m, 2H); MS(ES+) m/z 570.1, 571.1 (M+1).
Example 354
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((2-methylthiazol-4-- yl) methyl)piperidin-4-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR00904##
[1531] Step 1. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl) methyl)piperidin-4-yl)methoxy)benzoate
##STR00905##
[1533] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 4-(chloromethyl)-2-methylthiazole, the title compound was obtained as a colorless solid (0.20 g, 66%): MS(ES+) m/z 419.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl) methyl)piperidin-4-yl)methoxy)benzoic acid
##STR00906##
[1535] To a solution of methyl 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl)methyl) piperidin-4-yl)methoxy)benzoate (0.20 g, 0.48 mmol) in tetrahydrofuran (10 mL) was a added a solution of lithium hydroxide (0.11 g, 4.80 mmol) in water (5 mL). The reaction mixture was refluxed for 5 hours, cooled to ambient temperature, and acidified with 1N hydrochloric acid solution. The reaction mixture was extracted with ethyl acetate (3.times.10 mL), dried over anhydrous sodium sulfate, filtered.
[1536] The filtrate was concentrated invacuo to afford the title compound as a colorless gum (0.14 g, 72% yield): MS(ES+) m/z 405.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((2-methylthiazol-4-- yl) methyl)piperidin-4-yl)methoxy)benzamide
##STR00907##
[1538] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl) methyl)piperidin-4-yl)methoxy)benzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.03 g, 20%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 9.85 (br s, 1H), 7.72 (s, 1H), 7.12 (d, J=9.0 Hz, 1H), 6.98 (d, J=12.8 Hz, 1H), 4.36 (s, 2H), 4.01-3.91 (m, 2H), 3.56-3.39 (m, 2H), 3.13-2.92 (m, 3H), 2.70 (s, 3H), 2.16-1.82 (m, 4H), 1.77-1.52 (m, 2H), 1.18-1.03 (m, 4H), 0.95-0.82 (m, 2H), 0.74-0.60 (m, 2H); MS(ES+) m/z 508.2 (M+1).
Example 355
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((2-isopropylthiazol- -4-yl) methyl)piperidin-4-yl)methoxy)benzamide trifluoroacetic acid salt
##STR00908##
[1539] Step 1. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((1-((2-isopropylthiazol-4-yl)methyl) piperidin-4-yl)methoxy)benzoate
##STR00909##
[1541] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 4-(chloromethyl)-2-isopropylthiazole, the title compound was obtained as a colorless gum (0.23 g, 70%): MS(ES+) m/z 447.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-fluoro-4-((1-((2-isopropylthiazol-4-yl)piperidin-4-yl) methoxy)benzoic acid
##STR00910##
[1543] Following the procedure as described in Example 354 step 2 and making non-critical variations as required to replace methyl 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl)methyl)piperidin-4-yl- ) methoxy)benzoate with methyl 5-cyclopropyl-2-fluoro-4-((1-((2-isopropylthiazol-4-yl) methyl)piperidin-4-yl)methoxy)benzoate, the title compound was obtained as a colorless solid (0.15 g, 77%): MS(ES+) m/z 432.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((2-isopropylthiazol- -4-yl) methyl)piperidin-4-yl)methoxy)benzamide 2,2,2-trifluoroacetate
##STR00911##
[1545] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace a 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((2-isopropylthiazol-4-yl) methyl)piperidin-4-yl)methoxy)benzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.08 g, 47%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 9.81 (s, 1H), 7.76 (s, 1H), 7.12 (d, J=8.2 Hz, 1H), 6.93 (d, J=12.9 Hz, 1H), 4.38 (s, 2H), 4.07-3.86 (m, 2H), 3.41-3.19 (m, 3H), 3.16-2.90 (m, 3H), 2.16-1.84 (m, 4H), 1.75-1.47 (m, 2H), 1.42-1.26 (m, 6H), 1.19-1.00 (m, 4H), 0.95-0.79 (m, 2H), 0.73-0.59 (m, 2H); MS(ES+) m/z 536.2 (M+1).
Example 356
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(5-fluoro-2-(isoprop- ylamino) benzyl)piperidin-4-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR00912##
[1546] Step 1. Preparation of methyl 4-((1-(2-amino-5-fluorobenzoyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fl- uorobenzoate
##STR00913##
[1548] To a mixture of 2-amino-5-fluorobenzoic acid (0.16 g, 1.00 mmol), benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate (PyBOP) (0.70 g, 1.50 mmol) and N,N-diisopropylethylamine (0.71 g, 4.00 mmol) in dichloromethane (2 mL) was added methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (0.52 g, 1.50 mmol). After stirring at an ambient temperature for 16 hours, the reaction mixture was diluted with dichloromethane (10 mL), washed with saturated solution of ammonium chloride (3.times.5 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated and the residue was purified by column chromatography eluting with ethyl acetate in hexane to afford the title compound as a colorless solid (0.05 g, 11%): MS(ES+) m/z 445.2 (M+1).
Step 2. Preparation of methyl 4-((1-(2-amino-5-fluorobenzoyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00914##
[1550] To a solution of methyl 4-((1-(2-amino-5-fluorobenzoyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fl- uorobenzoate (0.18 g, 0.40 mmol) in anhydrous tetrahydrofuran (4 ml), was added borane dimethyl sulfide complex (1.5 mL, 150 mmol). After stirring at ambient temperature for 1 hour, the reaction mixture was added slowly to methanol (50 mL), followed by addition of hydrogen chloride (4.0M in dioxane solution, 5 mL), and then concentrated invacuo to afford the title compound as a colorless solid (0.10 g, 58%): MS(ES+) m/z 431.2 (M+1).
Step 3. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(isopropylamino) benzyl)piperidin-4-yl)methoxy)benzoate
##STR00915##
[1552] To 20 mL microwave vial was added methyl 4-((1-(2-amino-5-fluorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (0.20 g, 0.50 mmol), methanol (5 mL), sodium cyanoborohydride (0.20 g, 3.00 mmol), acetone (1 mL) and acetic acid (1 mL). The reaction mixture was heated at 130.degree. C. for 15 minutes. After cooling to ambient temperature, the reaction mixture was concentrated and basified with sodium bicarbonate solution, extracted with ethyl acetate (2.times.20 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo to afford the title compound as a gum (0.12 g, 51%). Which was used in next step without further purification: MS(ES+) m/z 473.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(isopropylamino) benzyl)piperidin-4-yl)methoxy)benzoic acid
##STR00916##
[1554] Following the procedure as described in Example 354 step 2 and making non-critical variations as required to replace methyl 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl)methyl)piperidin-4-yl- ) methoxy)benzoate with methyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(isopropylamino) benzyl)piperidin-4-yl)methoxy)benzoate, the title compound was obtained as a gum (0.04 g, 34%). Which was used in next step without further purification; MS(ES+) m/z 459.6 (M+1).
Step 4. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(5-fluoro-2-(isoprop- ylamino) benzyl)piperidin-4-yl)methoxy)benzamide
##STR00917##
[1556] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(5-fluoro-2-(isopropylamino) benzyl)piperidin-4-yl)methoxy)benzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 7.33 (s, 1H), 7.23-7.06 (m, 3H), 7.04-6.94 (m, 1H), 6.76-6.68 (m, 1H), 4.30 (s, 2H), 3.98 (s, 2H), 3.70-3.54 (m, 1H), 3.50-3.34 (m, 2H), 3.19-2.97 (m, 3H), 2.54 (s, 4H), 2.18-1.92 (m, 3H), 1.75-1.57 (m, 2H), 1.22-1.07 (m, 8H), 0.05-0.85 (m, 2H), 0.73-0.63 (m, 2H); MS(ES+) m/z 562.2 (M+1).
Example 357
Synthesis of 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)methyl)pipe- ridin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00918##
[1557] Step 1. Preparation of methyl 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)methyl)pipe- ridin-4-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00919##
[1559] To a mixture or methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate (0.12 g, 0.40 mmol), 5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazole-4-carbaldehyde (0.08 g, 0.40 mmol) in dichloroethane (2 mL) was added sodium triacetoxyborohydride (0.25 g, 1.20 mmol) and acetic acid (0.07 g, 1.20 mmol). After stirring at ambient temperature for 16 hours, the reaction mixture was quenched by addition aqueous ammonium hydroxide solution (28%, 3 mL) and extracted with dichloromethane, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo to afford the title compound as a colorless gum (0.01 g, 50%). Which was used in next step without further purification; MS(ES+) m/z 506.2, 504.2 (M+1).
Step 2. Preparation of 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00920##
[1561] Following the procedure as described in Example 354 step 2 and making non-critical variations as required to replace methyl 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl)methyl)piperidin-4-yl- ) methoxy)benzoate with methyl 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.10 g, quant. yield): MS(ES+) m/z 492.3, 490.1 (M+1).
Step 3. Preparation of 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fl- uorobenazmide 2,2,2,-trifluoroacetate
##STR00921##
[1563] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((5-chloro-1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 10%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (br s, 1H), 7.3 (s, 1H), 7.12 (s, 1H), 6.98 (s, 1H), 4.21-4.03 (m, 1H), 3.95 (s, 3H), 3.80-3.20 (m, 6H), 3.1-2.8 (m, 2H), 2.1-1.8 (m, 4H), 1.61-1.42 (m, 2H), 1.21-1.01 (m, 4H), 0.95-0.85 (m, 2H), 0.72-0.55 (m, 2H); MS(ES+) m/z 595.1, 593.2 (M+1).
Example 358
Synthesis of 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-fluoroethyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00922##
[1564] Step 1. Preparation of tert-butyl, 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR00923##
[1566] To a mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate(1.31 g, 3.75 mmol), glyoxylic acid monohydrate (0.55 g, 5.99 mmol) and 4 .ANG. molecular sieves (1.0 g) in anhydrous toluene (15 mL) was added 3,5-dichlorophenyl)boronic acid (0.79 g, 4.12 mmol). The reaction mixture was refluxed for 16 hours, cooled to ambient temperature and filtered off the solid. The filtrated was concentrated and the residue was redissolved in anhydrous tetrahydrofuran (10 mL). A boron tetrahydrofuran complex (1.0 M tetrahydrofuran, 25 mL, 25.00 mmol) was added to the solution and stirred at ambient temperature for 16 hours. The reaction mixture was quenched with 1N hydrochloric acid solution (15 mL) and extracted with ethyl acetate (3.times.25 mL). The combined organic layer was dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated and the residue was purified by column chromatography eluting with ethyl acetate in hexanes to afford the title compound as a gum (0.95 g, 48%). Which was used in the next step without further purification: MS(ES+) m/z 540.2, 538.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-fluoroethyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00924##
[1568] To a solution of tert-butyl 5-cyclopropyl-4-((1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)piperidin-4-yl- ) methoxy)-2-fluorobenzoate (0.95 g, 1.77 mmol) in dichloromethane (15 mL) was added methoxyethyl)aminosulfur trifluoride (BAST) (0.57 g, 3.54 mmol). The reaction mixture was stirred at ambient temperature for 16 hours and washed with aqueous sodium bicarbonate solution (3.times.5 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated invacuo and the residue was redissolved in dichloromethane (15 mL). To this solution was added trifluoroacetic acid (5 mL), after stirring at ambient temperature for 3 hours, the reaction mixture was concentrated invacuo. The residue was redissolved in anhydrous tetrahydrofuran (15 mL). To this solution was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.43 g, 3.54 mmol), 4-dimethylaminopyridine (1.08 g, 8.85 mmol) and methanesulfonamide (0.34 g, 3.54 mmol). After stirring at a ambient temperature for 16 hours. The reaction mixture was diluted with ethyl acetate (15 mL), washed with brine (10 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated in vacuo and the residue was purified by preparative HPLC afford the title compound as a colorless solid (0.06 g, 6% in 3 steps): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 11.84 (br s, 1H), 10.06 (br s, 1H), 7.72 (dd, J=1.79, 1.79 Hz, 1H), 7.54 (s, 2H), 7.11 (d, J=8.31 Hz, 1H), 6.96 (d, J=12.93 Hz, 1H), 6.33-6.03 (m, 1H), 4.11-3.37 (m, 8H), 3.22-2.95 (m, 2H), 2.50 (s, 3H), 2.17-1.87 (m, 2H), 1.83-1.58 (m, 2H), 0.94-0.78 (m, 2H), 0.71-0.61 (m, 2H); MS(ES+) m/z (M+1) 563.1
Example 359
Synthesis of (S)-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-- 4-yl) methoxy)-2-fluoro-5-methylbenzamide, trifluoroacetic acid salt
##STR00925##
[1569] Step 1. Preparation of tert-butyl 4-((1-benzylpiperidin-4-yl)methoxy)-2-fluoro-5-methylbenzoate
##STR00926##
[1571] Following the procedure as described in Example 346 step 2 and making non-critical variations as required to replace tert-butyl 4-fluoro-4-(hydroxymethyl)piperidine-carboxylate with (1-benzylpiperidin-4-yl)methanol and to replace tert-butyl 5-chloro-2,4-difluorobenzoate with tert-butyl 2,4-difluoro-5-methylbenzoate, the title compound was obtained as a colorless foam (0.85 g, 43%): MS(ES+) m/z 416.1, 414.2 (M+1).
Step 2. Preparation of tert-butyl 2-fluoro-5-methyl-4-(piperidin-4-yl)methoxy)benzoate
##STR00927##
[1573] To a mixture of tert-butyl 4-((1-benzylpiperidin-4-yl)methoxy)-2-fluoro-5-methylbenzoate (0.63 g, 1.52 mmol), ammonium formate (0.12 g, 1.82 mmol) was added 10% Pd/C (0.21 g) in methanol (15 mL). The reaction mixture was refluxed for 1 hour cooled to ambient temperature, filtered through a pad of diatomaceous earth, and washed with methanol. The filtrate was concentrated in vacuo and the residue was directly used in the next step; MS(ES+) m/z 324.2 (M+1).
Step 3. Preparation of (S)-tert-butyl 4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-5-methylbenzoate
##STR00928##
[1575] Following the procedure as described as Example 345 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate hydrochloride with tert-butyl 2-fluoro-5-methyl-4-(piperidin-4-ylmethoxy)benzoate, and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate, the title compound was obtained as a colorless foam (0.30 g, 82%): MS (ES+) m/z 498.1, 496.2 (M+1).
Step 4. Preparation of (S)-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-5-methylbenzoic acid
##STR00929##
[1577] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with (S)-tert-butyl 4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluoro-5-methylbenzoate, the title compound was obtained as a gum (0.25 g, 95%). Which was used in the next step without further purification; MS(ES-) m/z 438.1, 437.0 (M-1).
Step 5. Preparation of (S)-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluoro-5-methylbenzamide trifluoroacetic acid salt
##STR00930##
[1579] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (S)-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluoro-5-methylbenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.83 (br s, 1H), 9.49 (br s, 1H), 7.76 (brs, 1H), 7.65 (br s, 2H), 7.49 (d, J=9.0 Hz, 1H), 6.97 (d, J=12.0 Hz, 1H), 4.55 (br s, 1H), 4.00-3.19 (m, 2H), 3.17-3.01 (m, 2H), 2.94-2.75 (m, 2H), 2.31-2.24 (m, 1H), 2.13 (s, 3H), 2.08-1.89 (m, 3H), 1.72-1.48 (m, 5H), 1.19-1.05 (m, 4H); MS(ES+) m/z 545.1, 543.1 (M+1).
Example 360
Synthesis of (S)-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-5-methyl-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR00931##
[1581] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (S)-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy-2-fluoro-5-methylbenzoic acid, the title compound was obtained as a colorless solid (0.04 g, 63%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89 (br s, 1H), 9.60 (br s, 1H), 7.76 (s, 1H), 7.65 (d, J=3.0 Hz, 2H), 7.50 (d, J=9.0 Hz, 1H), 6.97 (d, J=12.0 Hz, 1H), 4.62-4.49 (m, 1H), 4.01-3.89 (m, 2H), 3.73-3.63 (m, 1H), 3.41-3.33 (m, 1H), 3.34 (s, 3H), 2.92-2.75 (m, 2H), 2.13 (s, 3H), 2.08-1.89 (m, 3H), 1.73-1.53 (m, 5H); MS(ES+) m/z 519.1, 517.1 (M+1).
Example 361
Synthesis of 4-((1-(bis(2-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR00932##
[1583] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(2-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.07 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D2O) .delta. 7.76-7.73 (m, 2H), 7.57-7.40 (m, 6H), 7.09 (d, J=1 8.3 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 6.05 (s, 1H), 3.99-3.92 (m, 2H), 3.29 (s, 3H), 3.27-3.18 (m, 2H), 3.17-3.08 (m, 2H), 2.22-2.09 (m, 1H), 2.05-1.94 (m, 3H), 1.69-1.54 (m, 2H), 0.90-0.84 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 605.2, 607.1 (M+1).
Example 362
Synthesis of 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR00933##
[1584] Step 1. Preparation of tert-butyl 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00934##
[1586] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-yl)methoxy)benzoate, and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 4,4'-(bromomethylene)bis(chlorobenzene), the title compound was obtained as a colorless foam (0.59 g, 95%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.37-7.21 (m, 9H), 6.48 (d, J=12.7 Hz, 1H), 4.23(s, 1H), 3.8 1(d, J=6.3 Hz, 2H), 2.87 (d, J=12.7 Hz, 2H), 1.99-1.74 (m, 6H), 1.54 (s, 9H), 1.54-1.38 (m, 2H), 0.88-0.81 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 554.2, 552.2 (M+1).
Step 2. Preparation of 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00935##
[1588] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with tert-butyl 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a gum (0.25 g, 95%). Which was used directly in the next step without further purification; MS(ES-) m/z 438.1, 437.0 (M-1).
Step 3. Preparation of 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00936##
[1590] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D.sub.2O) .delta. 7.64-7.61 (m, 4H), 7.55-7.52 (m, 4H), 7.09 (d, J=8.3 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 5.53 (s, 1H), 3.97-3.96 (m, 2H), 3.30 (s, 3H), 3.25-3.18 (m, 2H), 3.08-2.96 (m, 2H), 2.18-2.06 (m, 1H), 2.04-1.90 (m, 3H), 1.71-1.55 (m, 2H), 0.89-0.83 (m, 2H), 0.64-0.59 (m, 2H) (acidic protons were not observed): MS(ES+) m/z 604.9, 606.9 (M+1).
Example 363
Synthesis of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00937##
[1591] Step 1. Preparation of tert-butyl 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR00938##
[1593] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate, and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 4,4'-(bromomethylene)bis(fluorobenzene), the title compound was obtained as a colorless foam (0.59 g, 95%): MS(ES+) m/z 554.2, 552.2 (M+1).
Step 2. Preparation of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00939##
[1595] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with tert-butyl 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a gum (0.57 g, quant. yield). Which was used directly in the next step without further purification; MS(ES+) m/z 493.1, 492.1 (M-1).
Step 3. Preparation of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR00940##
[1597] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.02 g, 5%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 9.99 (br s, 1H), 7.76-7.62 (m, 2H), 7.38-7.27 (m, 2H), 7.12-6.94 (m, 4H), 6.84-6.72 (m, 2H), 4.02-3.99 (m, 4H), 3.30-3.18 (m, 2H), 3.08-3.00 (m, 1H), 2.07-1.93 (m, 3H), 1.87-1.78 (m, 2H), 1.17-1.04 (m, 4H), 0.87-0.77 (m, 2H), 0.06-0.61 (m, 2H); MS(ES+) m/z 600.1, 599.2 (M+1).
Example 364
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((3-(trifluoromethyl- )pyridin-2-yl) methyl)piperidin-4-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR00941##
[1598] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl)pyridin-2-yl) methyl)piperidin-4-yl)methoxy)benzoate
##STR00942##
[1600] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate, and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 2-(chloromethyl)-3-(trifluoromethyl)pyridine, the title compound was obtained as a colorless foam (0.55 g, 95%): MS(ES+) m/z 510.1, 509.0 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl)pyridin-2-yl) methyl)piperidin-4-yl)methoxy)benzoic acid
##STR00943##
[1602] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with tert-butyl 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl) pyridin-2-yl)methyl)piperidin-4-yl)methoxy)benzoate, the title compound was obtained as a gum (0.49 g, quant. yield). Which was used directly in the next step without further purification; MS(ES-) m/z 493.1, 492.1 (M-1).
Step 3. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((3-(trifluoromethyl- ) pyridin-2-yl)methyl)piperidin-4-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR00944##
[1604] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl) pyridin-2-yl)methyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.07 g, 24%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.83 (br s, 1H), 9.49 (br s, 1H), 8.95 (d, J=4.7 Hz, 1H), 8.32 (d, J=8.2 Hz, 1H), 7.71 (d, J=7.9, 4.9 Hz, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.97 (d, J=12.9 Hz, 1H), 4.69 (s, 2H), 3.97 (d, J=5.2 Hz, 2H) 1H), 3.64-3.39 (m, 2H), 3.30-3.18 (m, 2H), 3.08-3.00 (m, 1H), 2.07-1.93 (m, 3H), 1.87-1.78 (m, 2H), 1.19-1.05 (m, 4H), 0.92-0.82 (m, 2H), 0.68-0.63 (m, 2H); MS(ES+) m/z 557.1, 556.1 (M+1).
Example 365
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((3-(trifluoromethyl)pyri- din-2-yl) methyl)piperidin-4-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR00945##
[1606] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl) pyridin-2-yl)methyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.06 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89 (br s, 1H), 9.98 (br s, 1H), 8.95 (d, J=4.8 Hz, 1H), 8.32 (d, J=7.7 Hz, 1H), 7.71 (dd, J=8.0, 4.9 Hz, 1H), 7.11 (d, J=8.3 Hz, 1H), 6.97 (d, J=12.9 Hz, 1H), 4.69 (s, 2H), 3.97 (d, J=5.1 Hz, 2H) 1H), 3.60-3.48 (m, 2H), 3.30-3.18 (m, 2H), 3.08-3.00 (m, 1H), 2.07-1.93 (m, 3H), 1.87-1.78 (m, 2H), 1.19-1.05 (m, 4H), 0.92-0.82 (m, 2H), 0.68-0.63 (2H); MS(ES+) m/z 531.0, 530.0 (M+1).
Example 366
Synthesis of 4-((1-((4-bromo-5-chloro-2-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00946##
[1607] Step 1. Preparation of 4-((1-((4-bromo-5-chloro-2-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00947##
[1609] To a mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-yl)methoxy)benzoate (0.50 g, 1.43 mmol) and triethylamine (0.4 mL) in anhydrous tetrahydrofuran (5 mL) was added 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride (0.66 g, 2.15 mmol). After stirring at ambient temperature for 16 hours, the reaction mixture was diluted with ethyl acetate (5 mL), washed with 1M aqueous hydrochloric acid solution (3.times.2 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated invacuo and the residue was redissolved in dichloromethane (5 mL) and trifluoroacetic acid (2 mL). The reaction mixture was stirred at ambient temperature for 3 hours and concentrated invacuo. The residue was triturated with ether to afford the title compound as a colorless solid (0.30 g, 37%). Which was used directly in the next step without any further purification: MS(ES+) m/z 567.1, 565.1 (M+1).
Step 2. Preparation of 4-((1-((4-bromo-5-chloro-2-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)
##STR00948##
[1611] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin-4-fluoropiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-5-chloro-2-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.70 (d, J=15.0 Hz, 1H), 7.93 (d, J=6.0 Hz, 1H), 7.62-7.49 (m, 2H), 6.56 (d, J=15.0 Hz, 1H), 4.03-3.93 (m, 2H), 3.93-3.86 (m, 2H), 3.41 (s, 3H), 2.73-2.60 (m, 2H), 2.03-1.79 (m, 4H), 1.48-1.27 (m, 2H), 0.95-0.81 (m, 2H), 0.71-0.59 (m, 2H); MS(ES+) m/z 645.0, 643.0 (M+1).
Example 367
Synthesis of 4-((1-((4-bromo-5-chloro-2-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00949##
[1613] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-5-chloro-2-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 8.16 (d, J=12.0 Hz, 1H), 7.91 (d, J=12.0 Hz, 1H), 7.12 (d, J=6.0 Hz, 1H), 6.94 (d, J=6.0 Hz, 1H), 4.02-3.91 (m, 2H), 3.80-3.68 (m, 2H), 3.13-2.99 (m, 1H), 2.75-2.60 (m, 2H), 2.05-1.79 (m, 2H), 1.44-1.18 (m, 4H), 1.17-1.03 (m, 4H), 0.93-0.79 (m, 2H), 0.71-0.6 (m, 2H); MS(ES+) m/z 671.0, 669.0 (M+1).
Example 368
Synthesis of 4-((1-((4-bromo-3-fluorophenyl)sulfonyl)piperidin-4-yl)methoxy)-5-cyclopr- opyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00950##
[1614] Step 1. Preparation of 4-((1-((4-bromo-3-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00951##
[1616] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-bromo-3-fluorobenzene-1-sulfonyl chloride, the title compound was obtained as a colorless solid (0.30 g, 75%): MS(ES-) m/z 528.1, 526.1 (M-1).
Step 2. Preparation of 4-((1-((4-bromo-3-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00952##
[1618] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-3-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.05 g, 30%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (s, 1H), 7.98 (dd, J=8.3, 6.8 Hz, 1H), 7.72 (dd, J=8.2, 1.9 Hz, 1H), 7.49 (d, J=8.3, 1.8 Hz, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 3.93 (d, J=5.9 Hz, 2H), 3.74 (d, J=12.2 Hz, 2H), 3.29 (s, 3H), 2.40-2.32 (m, 2H), 1.99-1.89 (m, 2H), 1.89-1.76 (m, 2H), 1.42-1.30 (m, 2H), 0.89-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 609.1, 607.0 (M+1).
Example 369
Synthesis of 4-((1-((4-bromo-3-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00953##
[1620] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-3-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.08 g, 46%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (br s, 1H), 7.98 (dd, J=8.3, 6.4 Hz, 1H), 7.72 (dd, J=8.2, 1.9 Hz, 1H), 7.49 (d, J=8.3, 1.8 Hz, 1H), 7.08 (d, J=8.3 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 3.90 (d, J=5.4 Hz, 2H), 3.70 (d, J=11.4 Hz, 2H), 3.07-2.98 (m, 1H), 2.36 (t, J=10.9, 10.9, 2H), 1.98-1.87 (m, 1H), 1.43-1.27 (m, 2H), 1.11-1.04 (m, 3H), 0.88-0.78 (m, 4H), 0.64-0.59 (m, 2H); MS(ES+) m/z 635.1, 633.1 (M+1).
Example 370
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((2-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00954##
[1621] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((2-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid
##STR00955##
[1623] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 2-(trifluoromethyl)benzene-1-sulfonyl chloride, the title compound (0.24 g, 59% yield) was obtained as a colorless solid; MS(ES-) m/z 500.1 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((2-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)
##STR00956##
[1625] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((2-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.88 (br s, 1H), 8.05-7.98 (m, 2H), 7.98-7.84 (m, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 3.93 (d, J=5.9 Hz, 2H), 3.74 (d, J=12.2 Hz, 2H), 3.28 (s, 3H), 2.81-2.69 (m, 2H), 1.98-1.89 (m, 2H), 1.85-1.76 (m, 2H), 1.42-1.27 (m, 2H), 0.87-0.80 (m, 2H), 0.65-0.60 (m, 2H); MS(ES+) m/z 579.1 (M+1).
Example 371
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((2-(trifluoromethyl- ) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00957##
[1627] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((2-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.10 g, 65%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 8.05-7.98 (m, 2H), 7.90-7.87 (m, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 6.75 (brs, 1H), 3.94 (d, J=5.9 Hz, 2H), 3.70 (d, J=11.1 Hz, 2H), 3.74 (d, J=12.4 Hz, 2H), 3.07-2.91 (m, 1H), 2.82-2.71 (m, 2H), 2.35-1.89 (m, 2H), 1.89-1.77 (m, 2H), 1.42-1.26 (m, 2H), 1.11-1.02 (m, 3H), 0.89-0.78 (m, 4H), 0.86-0.58 (m, 2H); MS(ES+) m/z 605.2, 607.1 (M+1).
Example 372
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00958##
[1628] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid
##STR00959##
[1630] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-(trifluoromethyl)benzene-1-sulfonyl chloride, the title compound was obtained as a colorless solid (0.27 g, 66%): MS(ES+) m/z 502.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00960##
[1632] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.05 g, 29%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (br s, 1H), 8.01 (d, J=8.5 Hz, 2H), 7.93 (d, J=8.4 Hz, 2H), 7.08 (d, J=8.3 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 3.89 (d, J=5.4 Hz, 2H), 3.69 (d, J=11.4 Hz, 2H), 3.29 (s, 3H), 2.38-2.27 (m, 2H), 1.98-1.88 (m, 1H), 1.87-1.74 (m, 3H), 1.44-1.26 (m, 2H), 0.86-0.78 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 579.1 (M+1).
Example 373
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((4-(trifluoromethyl- ) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide (
##STR00961##
[1634] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.09 g, 58%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.78 (br s, 1H), 8.00 (d, J=8.6 Hz, 2H), 7.94 (d, J=8.4 Hz, 2H), 7.08 (d, J=8.3 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 6.75 (s, 1H), 3.89 (d, J=5.4 Hz, 2H), 3.70 (d, J=11.1 Hz, 2H), 3.07-2.98 (m, 1H), 2.42-2.26 (m, 2H), 1.98-1.89 (m, 1H), 1.88-1.72 (m, 2H), 1.44-1.28 (m, 2H), 1.11-1.02 (m, 3H), 0.89-0.78 (m, 4H), 0.67-0.58 (m, 2H); MS(ES+) m/z 605.2, 607.1 (M+1).
Example 374
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((3-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00962##
[1635] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid
##STR00963##
[1637] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 3-(trifluoromethyl)benzene-1-sulfonyl chloride, the title compound was obtained as a colorless solid (0.21 g, 52%): MS(ES-) m/z 500.1 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((3-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00964##
[1639] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-((3-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.08 g, 50%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89 (br s, 1H), 8.18-8.06 (m, 2H), 8.00-7.88 (m, 2H), 7.11 (d, J=9.0 Hz, 1H), 6.92 (d, J=15.0 Hz, 1H), 3.97-3.88 (m, 2H), 3.82-3.70 (m, 2H), 3.33 (s, 3H), 2.41-2.28 (m, 2H), 2.02-1.91 (m, 1H), 1.88-1.75 (m, 3H), 1.49-1.30 (m, 2H), 0.9-0.8 (m, 2H), 0.7-0.6 (m, 2H); MS(ES-) m/z 577.2 (M-1).
Example 375
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((3-(trifluoromethyl- ) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00965##
[1641] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin-4-fluoropiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((3-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.04 g, 70%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 8.18-8.04 (m, 3H), 8.00-7.88 (m, 2H), 7.10 (d, J=9.0 Hz, 1H), 6.91 (d, J=15.0 Hz, 1H), 3.98-3.87 (m, 2H), 3.82-3.70 (m, 2H), 3.12-3.00 (m, 1H), 2.42-2.29 (m, 2H), 2.01-1.91 (m, 1H), 1.91-1.76 (m, 2H), 1.48-1.30 (m, 2H), 1.28-1.04 (m, 4H), 0.93-0.81 (m, 2H), 0.70-0.60 (m, 2H); MS(ES+) m/z 605.2 (M+1).
Example 376
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((tetrahydro-2H-pyran-4-y- l) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00966##
[1642] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((tetrahydro-2H-pyran-4-yl)sulfonyl)piperidi- n-4-yl) methoxy)benzoic acid
##STR00967##
[1644] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with tetrahydro-2H-pyran-4-yl) sulfonyl chloride, the title compound (0.10 g, 26% yield) was obtained as a colorless gum; MS(ES-) m/z 400.2 (M+1).
Step 2. Preparation of 5 -cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((tetrahydro-2H-pyran-4-yl- ) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00968##
[1646] Following the procedure as described as Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((tetrahydro-2H-pyran-4-yl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.05 g, 76%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (br s, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 3.96 (d, J=6.1 Hz, 2H ), 3.90-3.85 (m, 2H), 3.67-3.63 (m, 2H), 3.43-3.25 (m, 6H), 2.97-2.89 (m, 2H), 2.02-1.93 (m, 2H), 1.83-1.79 (m, 4H), 1.57 (ddd, J=12.4, 12.4, 4.7 Hz, 2H), 1.37-1.23 (m, 2H), 0.89-0.82 (m, 2H), 0.67-0.61 (m, 2H); MS(ES+) m/z 519.1 (M+1).
Example 377
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((tetrahydro-2H-pyra- n-4-yl) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00969##
[1648] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((tetrahydro-2H-pyran-4-yl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.04 g, 68%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 3.94 (d, J=6.1 Hz, 2H), 3.90-3.85 (m, 2H), 3.67-3.63 (m, 2H), 3.43-3.25 (m, 3H), 3.08-2.89 (m, 3H), 2.02-1.93 (m, 2H), 1.83-1.79 (m, 4H), 1.57 (ddd, J=4.6, 12.4, 12.4 Hz, 2H), 1.37-1.23 (m, 2H), 1.09-1.05 (m, 4H), 0.89-0.82 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 545.1 (M+1).
Example 378
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(trifluoromethoxy) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00970##
[1649] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((4-(trifluoromethoxy) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid
##STR00971##
[1651] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-(trifluoromethoxy)benzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.25 g, 60%): MS(ES-) m/z 516.2 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((4-(trifluoromethoxy) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00972##
[1653] Following the procedure described in Example 346 step 7 and making non-critical variations as required to replace -4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-((1-((4-(triflu- oromethoxy) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (br s, 1H), 7.86 (d, J=8.5 Hz, 2H), 7.60 (d, J=8.5 Hz, 2H), 7.09 (d, J=8.3 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 3.89 (d, J=5.1 Hz, 2H), 3.70-3.66 (m, 2H), 3.27 (m, 3H), 2.34-2.26 (m, 2H), 1.98-1.89 (m, 1H), 1.84-1.80 (m, 3H), 1.41-1.29 (m, 2H), 0.86-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 595.0 (M+1).
Example 379
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((4-(trifluoromethox- y) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00973##
[1655] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((4-(trifluoromethoxy) phenyl)sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.79 (br s, 1H), 7.88-7.84 (m, 2H), 7.62-7.59 (m, 2H), 7.08 (d, J=8.3 Hz, 1H), 6.89 (d, J=13.0 Hz, 1H), 3.90 (d, J=5.4 Hz, 2H), 3.70-3.66 (m, 2H), 3.07-2.98 (m, 1H), 2.33-2.26 (m, 2H), 1.98-1.89 (m, 1H), 1.84-1.80 (m, 3H), 1.41-1.28 (m, 2H), 1.08-1.04 (m, 4H), 0.86-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 621.0 (M+1).
Example 380
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((3,4,5-trifluorophenyl) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00974##
[1656] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((3,4,5-trifluorophenyl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid
##STR00975##
[1658] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 3,4,5-trifluorobenzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.23 g, 59%): MS(ES+) m/z 488.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-((3,4,5-trifluorophenyl) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00976##
[1660] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((3,4,5-trifluorophenyl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.08 g, 49%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.89 (br s, 1H), 7.86-7.71 (m, 2H), 7.13 (d, J=9.0 Hz, 1H), (d, J=12.0 Hz, 1H), 4.00-3.89 (m, 2H), 3.77-3.64 (m, 2H), 2.47-2.36 (m, 2H), 2.03-1.91 (m, 2H), 1.90-1.74 (m, 3H), 1.48-1.20 (m, 4H), 0.92-0.81 (m, 2H), 0.70-0.62 (m, 2H).
Example 381
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((3,4,5-trifluorophe- nyl) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00977##
[1662] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(3,4,5-trifluorophenyl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.05 g, 31%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 7.88-7.75 (m, 2H), 7.11 (d, J=9.0 Hz, 1H), 6.94 (d, J=15.0 Hz, 1H), 3.98-3.89 (m, 2H), 3.77-3.66 (m, 2H), 3.11-3.00 (m, 1H), 2.47-2.37 (m, 3H), 2.04-1.92 (m, 1H), 1.90-1.76 (m, 3H), 1.47-1.31 (m, 2H), 1.16-1.04 (m, 3H), 0.90-0.80 (m, 2H), 0.71-0.61 (m, 2H); MS(ES+) m/z 591.2 (M+1).
Example 382
Synthesis of 4-((1-((3-chloro-4-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00978##
[1663] Step 1. Preparation of 4-((1-((3-chloro-4-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00979##
[1665] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 3-chloro-4-fluorobenzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.23 g, 59%): MS(ES-) m/z 488.1, 486.1 (M-1).
Step 2. Preparation of 4-((1-((3-chloro-4-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00980##
[1667] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((3-chloro-4-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.03 g, 17%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.88 (br s, 1H), 7.93 (dd, J=2.0, 6.8 Hz, 1H), 7.78-7.64 (m, 2H), 7.09 (d, J=8.3 1H), 6.87 (d, J=12.9 Hz, 1H), 3.90 (d, J=5.1 Hz, 2H), 3.70-3.66 (m, 2H), 3.25 (s, 3H), 2.38-2.30 (m, 2H), 1.96-1.80 (m, 4H), 1.41-1.27 (m, 2H), 0.86-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES-) m/z 561.1 (M-1).
Example 383
Synthesis of 4-((1-((3-chloro-4-fluorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00981##
[1669] Following the procedure as described as Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((3-chloro-4-fluorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.04 g, 25%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.78 (br s, 1H), 7.93 (dd, J=2.1, 6.8 Hz, 1H), 7.78-7.64 (m, 2H), 7.08 (d, J=8.3 Hz, 1H), 6.90 (d, J=13.0 Hz, 1H), 3.90 (d, J=5.3 Hz, 2H), 3.70-3.66 (m, 2H), 3.07-2.99 (m, 1H), 2.37-2.30 (m, 2H), 1.98-1.80 (m, 4H), 1.41-1.29 (m, 2H), 1.09-1.05 (m, 4H), 0.86-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 589.0. 591.0 (M+1).
Example 384
Synthesis of 4-((1-((2-chloro-4-(trifluoromethyl)phenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00982##
[1670] Step 1. Preparation of 4-((1-((2-chloro-4-(trifluoromethyl)phenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00983##
[1672] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 2-chloro-4-(trifluoromethyl)benzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum: (0.26 g, 61%) MS(ES-) m/z 534.0, 532.1 (M-1).
Step 2. Preparation of 4-((1-((2-chloro-4-(trifluoromethyl)phenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00984##
[1674] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-((3-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((2-chloro-4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.08 g, 49%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 8.16-8.15 (m, 1H), 8.07-8.04 (m, 1H), 7.94 (d, J=8.3 Hz, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 3.93 (d, J=5.8 Hz, 2H), 3.80-3.76 (m, 2H), 3.30 (s, 3H), 2.86-2.79 (m, 2H), 1.98-1.80 (m, 4H), 1.39-1.26 (m, 2H), 0.86-0.80 (m, 2H), 0.65-0.60 (m, 2H); MS(ES+) m/z 613.0, 615.0 (M+1).
Example 385
Synthesis of 4-((1-((2-chloro-4-(trifluoromethyl)phenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00985##
[1676] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((2-chloro-4-(trifluoromethyl) phenyl)sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.03 g, 14%): .sup.1H NMR (300 MHZ, DMSO-d.sub.6) .delta. 11.79 (br s, 1H), 8.16-8.15 (m, 1H), 8.07-8.04 (m, 1H), 7.94 (d, J=8.3 Hz, 1H), 7.08 (d, J=8.3 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 3.93 (d, J=5.8 Hz, 2H), 3.80-3.76 (m, 2H), 3.07-2.99 (m, 1H), 2.86-2.79 (m, 2H), 1.98-1.80 (m, 4H). 1.39-1.26 (m, 2H), 1.09-1.056 (m, 4H), 0.86-0.80 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 639.0, 641.0 (M+1).
Example 386
Synthesis of 5-cyclopropyl-2-fluoro-4-((1-((4-fluoro-2-methylphenyl)sulfonyl) piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR00986##
[1677] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((4-fluoro-2-methylphenyl)sulfonyl) piperidin-4-yl)methoxy)benzoic acid
##STR00987##
[1679] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-fluoro-2-methylbenzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.28 g, 75%): MS(ES-) m/z 464.1 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((1-((4-fluoro-2-methylphenyl)sulfonyl) piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR00988##
[1681] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((4-fluoro-2-methylphenyl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 7.84 (dd, J=8.8, 5.9 Hz, 1H), 7.33 (dd, J=10.0, 2.6 Hz, 1H), 7.23 (ddd, J=8.6, 2.8. Hz, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.91 (d, J=13.0 Hz, 1H), 3.92 (d, J=5.8, 2H), 3.62 (d, J=12.0, 2H), 3.30 (s, 3H), 2.68-1.54 (m, 2H), 2.53 (s, 3H), 1.99-1.75 (m, 4H), 1.40-1.26 (m, 2H), 0.87-0.81 (m, 2H), 0.65-0.60 (m, 2H); MS(ES-) m/z 542.0, 541.0 (M-1).
Example 387
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-((4-fluoro-2-methylp- henyl) sulfonyl)piperidin-4-yl)methoxy)benzamide
##STR00989##
[1683] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-((4-fluoro-2-methylphenyl) sulfonyl)piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%), .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (br s, 1H), 7.84 (dd, J=8.8, 5.9 Hz, 1H), 7.33 (dd, J=10.0, 2.6 Hz, 1H), 7.23 (ddd, J=8.5, 8.5, 2.7 Hz, 1H), 7.08 (d, J=8.3 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 3.92 (d, J =5.8, 2H), 3.63 (d, J=12.1, 2H), 3.30 (s, 3H), 3.08-2.99 (m, 1H), 2.68-1.54 (m, 2H), 2.53 (s, 3H), 1.99-1.75 (m, 4H), 1.41-1.22 (m, 2H), 1.11-1.01 (m, 4H), 0.87-0.81 (m, 2H), 0.65-0.60 (m, 2H); MS(ES+) m/z 570.0, 569.0 (M+1).
Example 388
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(naphthalen-2-ylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR00990##
[1684] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-((1-(naphthalen-2-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid
##STR00991##
[1686] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-fluoro-2-methylbenzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.28 g, 72%): MS(ES-) m/z 482.1 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(naphthalen-2-ylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR00992##
[1688] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(naphthalen-2-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.84 (br s, 1H), 8.41 (s, 1H), 8.19-8.12 (m, 2H), 8.06-8.04 (m, 1H), 7.75-7.63 (m, 3H), 7.07 (d, J=8.3 Hz, 1H), 6.87 (d, J=13.0 Hz, 1H), 3.87 (d, J=5.6 Hz, 2H), 3.77-3.73 (m, 2H), 3.28 (s, 3H), 2.34-2.27 (m, 2H), 1.93-1.78 (m, 4H), 1.44-1.30 (m, 2H), 0.83-0.76 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 561.1 (M+1).
Example 389
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(naphthalen-2-ylsulf- onyl) piperidin-4-yl)methoxy)benzamide
##STR00993##
[1690] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(naphthalen-2-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.77 (br s, 1H), 8.41 (s, 1H), 8.19-8.12 (m, 2H), 8.06-8.03 (m, 1H), 7.75-7.63 (m, 3H), 7.06 (d, J=8.3 Hz, 1H), 6.87 (d, J=13.0 Hz, 1H), 3.87 (d, J=5.5 Hz, 2H), 3.77-3.74 (m, 2H), 3.07-2.98 (m, 1H), 2.34-2.27 (m, 2H), 1.93-1.73 (m, 4H), 1.43-1.32 (m, 2H), 1.09-1.05 (m, 4H), 0.82-0.76 (m, 2H), 0.62-0.56 (m, 2H); MS(ES+) m/z 585.1 (M+1).
Example 390
Synthesis of 4-((1-((4-bromo-3-chlorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00994##
[1691] Step 1. Preparation of 4-((1-((4-bromo-3-chlorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR00995##
[1693] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with 4-bromo-3-chlorobenzene-1-sulfonyl chloride, the title compound was obtained as a colorless gum (0.34 g, 78%): MS(ES-) m/z 544.0, 542.1 (M-1).
Step 2. Preparation of 4-((1-((4-bromo-3-chlorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR00996##
[1695] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-3-chlorophenyl)sulfonyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, d6-DMSO) .delta. 11.89 (br s, 1H), 8.07 (d, J=9.0 Hz, 1H), 7.95-7.89 (d, J=3.0 Hz, 1H), 7.62 (dd, J=12.0, 3.0 Hz, 1H), 7.13 (d, J=9.0, 1H), 6.93 (d, J=12.0 Hz, 1H), 3.98-3.87 (m, 2H), 3.77-3.64 (m, 2H), 3.31 (s, 3H), 2.46-2.32 (m, 2H), 2.03-1.92 (m, 1H), 1.91-1.76 (m, 3H), 1.49-1.30 (m, 2H), 0.94-0.81 (m, 2H), 0.71-0.61 (m, 2H); MS(ES+) m/z 625.0, 627.0 (M+1)
Example 391
Synthesis of 4-((1-((4-bromo-3-chlorophenyl)sulfonyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR00997##
[1697] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-((4-bromo-3-chlorophenyl) sulfonyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.01 g, 6%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 8.06 (d, J=9.0 Hz, 1H), 7.92 (d, J=3 Hz, 1H), 7.62 (dd, J=12 Hz, 3 Hz, 1H), 7.11 (d, J=9 Hz, 1H), 6.93 (d, J=12 Hz, 1H), 3.99-3.89 (m, 2H), 3.76-3.63 (m, 2H), 3.12-2.99 (m, 1H), 2.43-2.32 (m, 3H), 2.01-1.91 (m, 1H), 1.91-1.74 (m, 4H), 1.48-1.29 (m, 2H), 1.16-1.05 (m, 2H), 0.93-0.82 (m, 2H), 0.70-0.61 (m, 2H); MS(ES+) m/z 653.0, 651.0 (M+1).
Example 392
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(oxetan-3-ylsulfonyl) piperidin-4-yl)methoxy)benzamide (
##STR00998##
[1698] Step 1. Preparation of 5-cyclopropyl-2-fluoro-4-(( 1-(oxetan-3-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid
##STR00999##
[1700] Following the procedure as described in Example 366 step 1 and making non-critical variations as required to replace 4-bromo-5-chloro-2-fluorobenzene-1-sulfonyl chloride with oxetane-3-sulfonyl chloride, the title compound was obtained as a colorless gum (0.14 g, 39%): MS(ES-) m/z 512.1 (M-1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(oxetan-3-ylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR01000##
[1702] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(oxetan-3-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.03 g, 35%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.92 (d, J=13.0 Hz, 1H), 4.80-4.66 (m, 5H), 3.94 (d, J=6.0 Hz, 2H), 3.62-3.58 (m, 2H), 3.29 (s, 3H), 2.80-2.72 (m, 2H), 2.02-1.80 (m, 4H), 1.34-1.20 (m, 2H), 0.89-0.82 (m, 2H), 0.66-0.61 (m, 2H); MS(ES-) m/z 489.1 (M-1).
Example 393
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(oxetan-3-ylsulfonyl- ) piperidin-4-yl)methoxy)benzamide
##STR01001##
[1704] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(oxetan-3-ylsulfonyl) piperidin-4-yl)methoxy)benzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.07 g, 76%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.79 (br s, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.93 (d, J=13.0 Hz, 1H), 4.80-4.66 (m, 5H), 3.95 (d, J=6.0 Hz, 2H), 3.62-3.58 (m, 2H), 3.08-3.00 (m, 1H), 2.80-2.73 (m, 2H), 2.02-1.80 (m, 4H), 1.35-1.20 (m, 2H), 1.10-1.06 (m, 4H), 0.89-0.83 (m, 2H), 0.66-0.61 (m, 2H); MS(ES-) m/z 515.2 (M-1).
Example 394
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR01002##
[1705] Step 1. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzoate
##STR01003##
[1707] To a cooled (0.degree. C.) solution of 4-bromo-2-chlorobenzyl alcohol (0.75 g, 3.40 mmol) in anhydrous tetrahydrofuran (20 mL) was added methanesulfonyl chloride (0.66 mL, 8.50 mmol) and N,N-diisopropylethylamine (1.5 mL, 8.50 mmol). After stirring at 0.degree. C. under a nitrogen atmosphere for 40 minutes, the reaction mixture was diluted with ethyl acetate (100 mL), washed with 1 M hydrochloric acid solution (100 mL) and brine (100 mL), dried over anhydrous magnesium sulfate, filtered and concentrated invacuo. The residue was dissolved in anhydrous N,N-dimethylformamide (15 mL), and methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride (0.98 g, 3.2 mmol) and potassium carbonate (1.52 g, 11.0 mmol) were added to this solution. The reaction mixture was heated at 90.degree. C. under a nitrogen atmosphere for 17 hours, cooled to ambient temperature, diluted with brine (200 mL) and extracted with ethyl acetate (2.times.150 mL). The combined organic layers were dried over anhydrous magnesium sulfate, filtered, and concentrated to in vacuo. The residue was purified by column chromatography, eluting with a 0-50% gradient of ethyl acetate with 10% isopropanol, 10% triethylamine in hexanes to afford the title compound as a colorless solid (0.30 g, 25%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.43 (d, J=8.2 Hz, 1H), 6.52 (d, J=12.6 Hz, 1H), 3.89-3.83 (m, 7H), 2.78 (s, 3H), 2.75-2.67 (m, 2H), 2.02-1.94 (m, 4H), 1.63-1.50 (m, 2H), 0.92-0.87 (m, 2H), 0.65-0.60 (m, 2H); MS(ES+) m/z 386.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl)piperidin-4-yl) methoxy)benzoic acid
##STR01004##
[1709] To a solution of methyl 5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl)piperidin-4-yl) methoxy)benzoate (0.30 g, 0.77 mmol) in tetrahydrofuran (10 mL), water (5 mL) was added lithium hydroxide (0.40 g, 17.00 mmol). The mixture was refluxed for 4.5 hours, cooled to ambient temperature. The reaction mixture was acidified with 1 M hydrochloric acid solution (100 mL), extracted with ethyl acetate (100 mL) and dichloromethane (2.times.100 mL). The combined organic layers were dried over anhydrous magnesium sulfate, filtered and concentrated in vacuo to afford the title compound as a colorless solid (0.16 g, 57%). The aqueous layer contained solid that was filtered, washed with water (50 mL) and diethyl ether (30 mL) to afford additional amount of the title compound as a colorless solid (0.05 g, 18%): MS(ES-) m/z 370.1 (M-1).
Step 3. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR01005##
[1711] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.06 g, 45%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.94 (d, J=13.0 Hz, 1H), 3.97 (d, J=5.8 Hz, 2H), 3.58-3.54 (m, 2H), 3.08-3.00 (m, 1H), 2.82 (s, 3H), 2.75-2.67 (m, 2H), 2.02-1.84 (m, 4H), 1.43-1.30 (m, 2H), 1.09-1.05 (m, 4H), 0.90-0.83 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 475.1 (M+1).
Example 395
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzamide
##STR01006##
[1713] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((1-(methylsulfonyl) piperidin-4-yl)methoxy)benzoic acid, the title compound was obtained as a colorless solid (0.03 g, 25%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (br s, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.94 (d, J=13.0 Hz, 1H), 3.96 (d, J=5.8 Hz, 2H), 3.58-3.54 (m, 2H), 3.30 (s, 3H), 2.82 (s, 3H), 2.75-2.67 (m, 2H), 2.04-1.84 (m, 4H), 1.42-1.30 (m, 2H), 0.90-0.83 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 449.0 (M+1).
Example 396
Synthesis of 4-((1R,3S,5S)-8-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-8-azabic- yclo[3.2.1]octan-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01007##
[1714] Step 1. Preparation of (1R, 3S, 5S)-benzyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01008##
[1716] Following the procedure as described in Example 346 step 2 and making non-critical variations as required to replace of tert-butyl-4-fluoro-4-(hydroxymethyl)piperidine-1-carboxylate (4.60 g, 19.70 mmol) with (1R, 3S, 5S)-benzyl 3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as a colorless gum (7.10 g, 98%): MS(ES+) m/z 506.2, 504.2 (M+1).
Step 2. Preparation of (1R,3S,5S)-benzyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01009##
[1718] Following the procedure as described in Example 346 step 3 and making non-critical variations as required to replace of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with (1R,3S,5S)-benzyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as a colorless solid (5.7 g, 80%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 7.34-7.27 (m, 6H), 6.44 (d, J=12.6 Hz, 1H), 5.12 (s, 2H), 4.05 (br s, 2H), 3.75 (d, J=4.8 Hz, 2H), 2.49-2.34 (m, 1H), 2.03-1.88 (m, 3H), 1.76-1.64 (m, 5H), 1.55 (s, 9H), 0.91-0.80 (m, 2H), 0.66-0.61 (m, 2H).
Step 3. Preparation of 4-(((1R,3S,5S)-8-((benzyloxy)carbonyl)-8-azabicyclo[3.2.1]octan-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01010##
[1720] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with (1R,3S,5S)-benzyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as a colorless solid (1.30 g, 27%): MS(ES+) m/z 455.1, 454.1 (M+1).
Step 4. Preparation of (1R,3S,5S)-benzyl 3-((2-cyclopropyl-4-((cyclopropylsulfonyl)carbamoyl)-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]-octane-8-carboxylate
##STR01011##
[1722] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-(((1R,3S,5S)-8-((benzyloxy) carbonyl)-8-azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-2-fluorobe- nzoic acid, the title compound was obtained as a colorless solid (1.19 g, quant. yield): MS(ES+) m/z 556.2, 555.2 (M+1).
Step 5. Preparation of 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-N-(cycl- opropylsulfonyl)-2-fluorobenzamide
##STR01012##
[1724] A mixture of (1R,3S,5S)-benzyl 3-((2-cyclopropyl-4-((cyclopropylsulfonyl)carbamoyl)-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate (1.70 g, 3.01 mmol), Pd/C (0.080 g) and acetic acid (0.10 g) in ethanol (60 mL) was stirred at ambient temperature under hydrogen balloon for 2 hours. The solid was filtered and the solvent was concentrated invacuo to afford the title compound as a beige solid (1.19 g, 94%): MS(ES+) m/z 423.1 (M+1).
Step 6. Preparation of 4-(((1R,3S,5S)-8-(3-chloro-2-fluoro-5-(trifluoromethyl) benzyl)-8-azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-N-(cycloprop- ylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01013##
[1726] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-N-(cycl- opropylsulfonyl)-2-fluorobenzamide, the title compound was combined as a colorless solid (0.09 g, 5%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.81 (br s, 1H), 9.88 (br s, 1H), 8.25 (d, J=4.9 Hz, 1H), 8.06 (d, J=5.5 Hz, 1H), 7.07 (d, J=8.3 Hz, 1H), 6.98 (d, J=12.9 Hz, 1H), 4.33 (s, 2H), 4.05 (brs, 2H), 3.86 (d, J=6.2 Hz, 2H), 3.08-2.99 (m, 2H), 2.41-2.27 (m, 2H), 2.06-1.96 (m, 3H), 1.83-1.67 (m, 2H), 1.11-1.03 (m, 4H), 0.88-0.80 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 634.0, 633.0 (M+1).
Example 397
Synthesis of 4-(((1R,3S,5S)-8-(2-fluoro-5-(trifluoromethyl)piperidin)-8-azabicyclo[3.2- .1]octan-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01014##
[1728] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with 4-((1R,3s,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-N-(cycl- opropylsulfonyl)-2-fluorobenzamide, and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 1-chloro-2-(chloromethyl)-4-(trifluoromethyl)benzene, the title compound was obtained as a colorless solid (0.01 g, 6%) .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 9.36 (br s, 1H), 8.19 (s, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 6.05 (s, 1H), 3.99-3.92 (m, 2H), 3.29 (s, 3H), 3.27-3.18 (m, 2H), 3.17-3.08 (m, 2H), 2.22-2.09 (m, 1H), 2.05-1.94 (m, 3H), 1.69-1.54 (m, 2H), 0.90-0.84 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 617.1, 615.1 (M+1).
Example 398
Synthesis of 4-(((1R,3S,5S)-8-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-8-azabi- cyclo[3.2.1]octan-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01015##
[1729] Step 1. Preparation of (1R,3S,5S)-benzyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01016##
[1731] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-(((1R,3S,5S)-8-((benzyloxy) carbonyl)-8-azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-2-fluorobe- nzoic acid and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.45 g, 14%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 11.86 (br s, 1H), 7.39-7.25 (m, 5H), 7.10 (d, J=8.3 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 5.03 (s, 2H), 4.22-4.14 (m, 2H), 3.85 (d, J=5.1 Hz, 2H), 3.29 (s, 3H), 2.41-2.31 (m, 1H), 1.94-1.84 (m 3H), 1.72-1.44 (m, 6H), 0.85-0.76 (m, 2H), 0.65-0.59 (m, 2H); MS(ES+) m/z 532.1, 531.1 (M+1).
Step 2. Preparation of 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-2-fluor- o-N-(methylsulfonyl)benzamide
##STR01017##
[1733] Following the procedure as described in Example 396 step 5 and making non-critical variations as required to replace (1R,3S,5S)-benzyl 3-((2-cyclopropyl-4-((cyclopropylsulfonyl)carbamoyl)-5-fluorophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate with (1R,3S,5S)-benzyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-8- -azabicyclo[3.2.1]octane-8-carboxylate, title compound was obtained as a beige solid (1.10 g, 88%): MS(ES+) m/z 398.1, 397.1 (M+1).
Step 3. Preparation of 4-(((1R,3S,5S)-8-(3-chloro-2-fluoro-5-(trifluoromethyl) benzyl)-8-azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(- methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01018##
[1735] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-2fluoro- -N-(methylsulfonyl) benzamide, the title compound was obtained as a colorless solid (0.02 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D.sub.2) .delta. 8.21-8.18 (m, 1), 8.03-8.01 (m, 1H), 7.08 (d, J=8.2 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 4.31 (br s, 2H), 4.04-3.96 (m, 2H), 3.91-3.82 (m, 2H), 3.29 (s, 3H), 2.42-2.27 (m, 3H), 2.04-1.84 (m, 3H), 1.95-1.88 (m, 2H), 1.78-1.67 (m, 2H), 0.90-0.84 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 609.0, 607.0 (M+1).
Example 399
Synthesis of 4-(((1R,3S,5S)-8-(2-chloro-5-(trifluoromethyl) benzyl)-8-azabicyclo[3.2.1]octan-3-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(- methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01019##
[1737] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoropiperidin-4-yl)methoxy)benzoate with 4-((1R,3S,5S)-8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-2-fluor- o-N-(methylsulfonyl) benzamide and to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 1-chloro-2-(chloromethyl)-4-(trifluoromethyl)benzene, the title compound was obtained as a colorless solid (0.04 g, 29%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D.sub.2O) .delta. 8.10 (s, 1H), 7.97-7.76 (m, 2H), 7.04 (d, J=8.1 Hz, 1H), 6.87 (d, J=12.9 Hz, 1H), 4.31 (s, 2H), 4.04-3.94 (m, 2H), 3.92-3.82 (m, 2H), 3.25 (s, 3H), 2.46-2.29 (m, 3H), 2.07-1.95 (m, 3H), 1.94-1.84 (m, 2H), 1.78-1.64 (m, 2H), 0.87-0.81 (m, 2H), 0.59-53 (m, 2H); MS(ES+) m/z 591.0, 589.1 (M +1).
Example 400
Synthesis of 4-((1-(bis(4-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01020##
[1739] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)benzyl)-4-fluoropiperidin-4-y- l) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(4-chlorophenyl)methyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.07 g, 55%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D.sub.2O) .delta.7.63-7.60 (m, 4H), 7.54-7.51 (m, 4H), 7.07 (d, J=8.3 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 5.51 (s, 1H), 4.02-3.96 (m, 2H), 3.27-3.19 (m, 2H), 3.06-2.98 (m, 3H), 2.18-2.06 (m, 1H), 2.03-1.94 (m, 3H), 1.71-1.56 (m, 2H), 1.11-1.08 (m, 4H), 0.89-0.83 (m, 2H), 0.62-1.57 (m, 2H); MS(ES+) m/z 631.1, 633.1 (M+1).
Example 401
Synthesis of 4-((1-(bis(3-chlorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01021##
[1741] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)benzyl)-4-fluoropiperidin-4-y- l) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(bis(4-chlorophenyl)methyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropylsulfonamide, the title compound was obtained as a colorless solid (0.06 g, 34%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+1% D.sub.2O) .delta.7.70 (br s, 2H), 7.61-7.59 (m, 2H), 7.54-7.45 (m, 4H), 7.08 (d, J=8.3 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 5.51 (s, 1H), 3.98-3.96 (m, 2H), 3.24-3.21 (m, 2H), 3.07-2.98 (m, 3H), 2.19-2.07 (m, 1H), 2.04-1.95 (m, 3H), 1.73-1.57 (m, 2H), 1.11-1.06 (m, 4H), 0.90-0.83 (m, 2H), 0.63-0.58 (m, 2H); MS(ES+) m/z 631.0, 633.0 (M+1).
Example 402
Synthesis of (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01022##
[1742] Step 1. Preparation of (S)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR01023##
[1744] To a solution of tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride (0.13 g, 0.33 mmol) in anhydrous N,N-dimethylformamide (5 mL) was added (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate (0.14 g, 0.39 mmol) and potassium carbonate (0.16 g, 1.1 mmol). The reaction mixture was heated to 90.degree. C. under a nitrogen atmosphere for 15 hours, then cooled and diluted with water (100 mL). The mixture was extracted with ethyl acetate (2.times.100 mL) and the combined organic layers were dried over anhydrous magnesium sulfate, filtered, and concentrated in vacuo. The residue was purified by column chromatography eluting with a gradient of 0-50% ethyl acetate containing 10% isopropanol and 10% triethylamine in hexanes to afford the title compound as a colorless syrup (0.13 g, 73%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.84 (d, J=7.7 Hz, 1H), 7.20-7.19 (m, 3H), 6.59 (d, J=12.2 Hz, 1H), 4.84 (q, J=6.5 Hz, 1H), 3.83 (d, J=6.1 Hz, 2H), 3.40-3.33 (m, 1H), 3.03-3.00 (m, 1H), 2.81-2.77 (m, 1H), 2.04-1.76 (m, 6H), 1.55 (s, 9H), 1.31 (d, J=6.5 Hz, 3H); MS(ES+) m/z 516.0, 518.0, 520.0 (M+1).
Step 2. Preparation of (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01024##
[1746] To a solution of (S)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoate (0.13 g, 0.24 mmol) in dichloromethane (6 mL) was added trifluoroacetic acid (3 mL). The reaction mixture was stirred at ambient temperature for 40 minutes and concentrated in vacuo to afford the title compound as a colorless syrup that was used directly in the next step; MS(ES+) m/z 460.0, 462.0 (M+1).
Step 3. Preparation of (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide trifluoroacetic acid salt
##STR01025##
[1748] To a solution of (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt in anhydrous dichloromethane (10 mL) was added N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (0.09 g, 0.48 mmol), 4-(dimethylamino)pyridine (0.07 g, 0.54 mmol), and methanesulfonamide (0.04 g, 0.41 mmol). The reaction mixture was stirred at ambient temperature for 16 hours, and then diluted with ethyl acetate (100 mL). The mixture was washed with saturated aqueous ammonium chloride (100 mL), brine (2.times.100 mL), dried over anhydrous magnesium sulfate, filtered and concentrated invacuo. The residue was purified by reverse-phase HPLC, eluting with a gradient of 20-80% acetonitrile in water with 0.1% trifluoroacetic acid to afford the title compound as a colorless solid (0.01 g, 15% in 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.11 (br s, 1H), 9.42 (br s, 1H), 7.76-7.72 (m, 2H), 7.60 (s, 2H), 7.22 (d, J=12.4 Hz, 1H), 4.50 (br s, 1H), 4.02-4.00 (m, 2H), 3.66-3.59 (m, 1H), 3.34 (brs, 1H), 3.29 (s, 3H), 2.86-2.69 (m, 2H), 2.05-1.89 (m, 3H), 1.61-1.47 (m, 5H); MS(ES+) m/z 537.0, 539.0, 541.0 (M+1).
Example 403
Synthesis of (S)-5-chloro-N-(cyclopropylsulfonyl)-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01026##
[1750] Following the procedure as described in Example 402 Step 3 and making non-critical variations as required to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.01 g, 13% in 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.03 (br s, 1H), 9.42 (br s, 1H), 7.75-7.72 (m, 2H), 7.60 (s, 2H), 7.23 (d, J=12.4 Hz, 1H), 4.51 (brs, 1H), 4.02-4.00 (m, 2H), 3.67-3.60 (m, 1H), 3.45 (br s, 1H), 3.08-2.99 (m, 1H), 2.86-2.76 (m, 2H), 2.05-1.89 (m, 3H), 1.62-1.47 (m, 5H), 1.10-1.07 (m, 4H); MS(ES+) m/z 563.1, 565.1, 567.0 (M+1).
Example 404
Synthesis of (R)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR01027##
[1751] Step 1. Preparation of (R)-tert-butyl 5-chloro-4-((1-(1-(3,5dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR01028##
[1753] Following the procedure as described in Example 402 step 1 and making non-critical variations to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with (S)-1-(3,5-dichlorophenyl) ethyl 4-methylbenzenesulfonate, the title compound was obtained as a colorless syrup (0.10 g, 62%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.84 (d, J=7.6 Hz, 1H), 7.20-7.19 (m, 3H), 6.59 (d, J=12.1 Hz, 1H), 4.83 (q, J=6.5, 6.5, 6.4 Hz, 1H), 3.83 (d, J=5.9 Hz, 2H), 3.40-3.34 (m, 1H), 3.03-3.00 (m, 1H), 2.81-2.77 (m, 1H), 2.04-1.76 (m, 6H), 1.55 (s, 9H), 1.31 (d, J=6.2 Hz, 3H); MS(ES+) m/z 516.0, 518.0, 520.0 (M+1).
Step 2. Preparation of (R)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01029##
[1755] Following the procedure as described in Example 402 step 2 and making non-critical variations as required to replace (S)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate with (R)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a colorless syrup (0.11 g, quant. yield) that was used directly in the next step; MS(ES+) m/z 460.0, 462.0 (M+1).
Step 3. Preparation of (R)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01030##
[1757] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-2- -fluorobenzoic acid, trifluoroacetic acid salt with (R)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt, the title compound was obtained as a colorless solid (0.01 g, 20% in 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.08 (br s, 1H), 9.46 (br s, 1H), 7.76-7.72 (m, 2H), 7.60 (s, 2H), 7.21 (d, J=12.4 Hz, 1H), 4.48 (br s, 1H), 4.02-4.00 (m, 2H), 3.64-3.57 (m, 1H), 3.32 (br s, 1H), 3.27 (s, 3H), 2.83-2.73 (m, 2H), 2.04-1.88 (m, 3H), 1.60-1.46 (m, 5H); MS(ES+) m/z 537.0, 539.0, 541.0 (M+1).
Example 405
Synthesis of (R)-5-fluoro-N-(cyclopropylsulfonyl)-4-((1-((1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01031##
[1759] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with (R)-5-chloro-4-((1-(1-(3,5-dichlorophenyl) ethyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.01 g, 13% over 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.01 (br s, 1H), 9.48 (br s, 1H), 7.75-7.70 (m, 2H), 7.58 (s, 2H), 7.21 (d, J=12.4 Hz, 1H), 4.44 (br s, 1H), 4.02-4.00 (m, 2H), 3.62-3.53 (m, 1H), 3.44 (br s, 1H), 3.06-2.97 (m, 1H), 2.77-2.69 (m, 2H), 2.01-1.87 (m, 3H), 1.58-1.45 (m, 5H), 1.08-1.04 (m, 4H); MS(ES+) m/z 563.2, 565.3 (M+1).
Example 406
Synthesis of 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01032##
[1760] Step 1. Preparation of 3-chloro-5-(trifluoromethoxy)benzyl 4-methylbenzenesulfonate
##STR01033##
[1762] To a cooled (0.degree. C.) solution of 3-chloro-5-(trifluoromethoxy)benzaldehyde (0.45 g, 2.00 mmol) in anhydrous methanol (10 mL) was added sodium borohydride (0.15 g, 4.00 mmol). After stirring at 0.degree. C. under a nitrogen atmosphere for 1 hour, the reaction mixture was quenched with saturated aqueous ammonium chloride (3 mL), diluted with ethyl acetate (100 mL), washed with 1 M hydrochloric acid solution (2.times.75 mL), dried over anhydrous magnesium sulfate, filtered, and concentrated in vacuo. The residue was dissolved in anhydrous dichloromethane (20 mL) and to this solution was added 4-methylbenzene-1-sulfonyl chloride (0.47 g, 2.5 mmol), triethylamine (1.4 mL, 9.9 mmol), and 4-dimethylaminopyridine (0.031 g, 0.25 mmol). The reaction mixture was stirred at ambient temperature for 2.5 hours, then diluted with dichloromethane (100 mL), washed with water (100 mL) and brine (100 mL), dried over anhydrous magnesium sulfate, filtered, and concentrated in vacuo. The residue was purified by column chromatography, eluting with a gradient of 0-10% ethyl acetate in hexanes to afford the title compound as a colorless syrup (0.25 g, 33%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.75 (d, J=8.1 Hz, 2H), 7.31 (d, J=8.1 Hz, 2H), 7.14 (br s, 2H), 6.96 (br s, 1H), 5.01 (s, 2H), 2.42 (s, 3H).
Step 2. Preparation of tert-butyl 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR01034##
[1764] Following the procedure as described in Example 402 step 1 and making non-critical variations to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with 3-chloro-5-(trifluoromethoxy) benzyl 4-methylbenzenesulfonate, the title compound was obtained as a colorless syrup (0.27 g, 83%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.58 (d, J=7.7 Hz, 1H), 7.26 (br s, 1H), 7.10 (brs, 2H), 6.60 (d, J=12.1 Hz, 1H), 3.85 (d, J=6.3 Hz, 2H), 3.47 (s, 2H), 2.89-2.85 (m, 2H), 2.07-2.00 (m, 2H), 1.90-1.82 (m, 3H), 1.56 (s, 9H), 1.48-1.39 (m, 2H); MS(ES+) m/z 552.0, 554.0 (M+1).
Step 3. Preparation of 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy)benzyl) piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01035##
[1766] Following the procedure as described in Example 402 Step 2 and making non-critical variations as required to replace (S)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl) piperidin-4-yl)methoxy)-2-fluorobenzoate with tert-butyl 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzoate, the title compound was obtained as a light yellow syrup (0.29 g, 98%) that was used directly in the next reaction; MS(ES+) m/z 498.0, 496.0 (M+1).
Step 4. Preparation of 5-chloro-4-((1-(3-Chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01036##
[1768] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt, the title compound (0.05. g, 41% in 2 steps) as obtained as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.90 (br s, 1H), 9.81) (br s, 1H), 7.75 (d, J=7.5 Hz, 1H), 7.67-7.64 (m, 2H), 7.52 (s, 1H), 7.21 (d, J=12.4 Hz, 1H), 4.27 (brs, 2H), 4.04-4.02 (m, 2H), 3.32 (brs, 2H), 3.27 (s, 3H), 2.97-2.85 (m, 2H), 2.06-1.90 (m, 3H), 1.59-1.46 (m, 2H); MS(ES+) m/z 573.0, 575.0 (M+1).
Example 407
Synthesis of 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01037##
[1770] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.05 g, 46% over 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.04 (br s, 1H), 9.75 (br s, 1H), 7.74 (d, J=7.5 Hz, 1H), 7.69-7.66 (m, 2H), 7.54 (s, 1H), 7.24 (d, J=12.4 Hz, 1H), 4.33 (brs, 2H), 4.04-4.02 (m, 2H), 3.43-3.40 (m, 2H), 3.08-2.93 (m, 3H), 2.07-1.92 (m, 3H), 1.60-1.47 (m, 2H), 1.11-1.08 (m, 4H); MS(ES+) m/z 599.3, 601.0 (M+1).
Example 408
Synthesis of N-(azetidin-1-ylsulfonyl)-5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01038##
[1772] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-chloro-4-((1-(3-chloro-5-(trifluoromethoxy) benzyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.06 g, 53% in 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.82 (br s, 1H), 9.57 (br s, 1H), 7.77 (d, J=7.4 Hz, 1H), 7.70-7.66 (m, 2H), 7.54 (s, 1H), 7.24 (d, J=12.2 Hz, 1H), 4.32 (br s, 2H), 4.04-3.99 (m, 6H), 3.43-3.39 (m, 2H), 3.01-2.92 (m, 2H), 2.19-1.93 (m, 5H), 1.58-1.45 (m, 2H); MS(ES+) m/z 613.9, 615.9 (M+1).
Example 409
Synthesis of 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-5-cyano-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01039##
[1773] Step 1. Preparation of tert-butyl 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-5-cyano-2-fluorobenzoate
##STR01040##
[1775] Following the procedure as described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-yl)methoxy)benzoate hydrochloride with tert-butyl 5-cyano-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate, trifluoroacetic acid salt and to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with 3-chloro-5-(trifluoromethoxy) benzyl 4-methylbenzenesulfonate, the title compound was obtained as a colorless syrup (0.03 g, 7%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.12 (d, J=8.0 Hz, 1H), 7.26 (br s, 1H), 7.09 (br s, 2H), 6.65 (d, J=12.1 Hz, 1H), 3.90 (d, J=6.6 Hz, 2H), 3.47 (s, 2H), 2.89-2.85 (m, 2H), 2.07-1.84 (m, 5H), 1.56 (s, 9H), 1.45-1.32 (m, 2H); MS(ES+) m/z 543.3, 545.1 (M+1).
Step 2. Preparation of 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-5-cyano-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01041##
[1777] Following the procedure as described in Example 402 step 2 and making non-critical variations as required to replace (S)-tert-butyl 5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoate with tert-butyl 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-5-cyano-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.03 g, quant. yield) that was used directly in the next step; MS(ES+) m/z 487.0 (M+1).
Step 3. Preparation of 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-4-yl) methoxy)-5-cyano-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01042##
[1779] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(3-chloro-5-(trifluoromethoxy)benzyl) piperidin-4-yl)methoxy)-5-cyano-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound (0.02 g, 43% in 2 steps) was obtained as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 8.12 (d, J=7.8 Hz, 1H), 7.67-7.64 (m, 2H), 7.52 (s, 1H), 7.31 (d, J=12.6 Hz, 1H), 4.31 (s, 2H), 4.09-4.07 (m, 2H), 3.43-3.39 (m,2H), 3.06-2.93 (m, 3H), 2.11-1.03 (m, 1H), 1.96-1.92 (m, 2H), 1.57-1.44 (m, 2H), 1.10-1.07 (m, 4H); MS(ES+) m/z 592.0, 590.0 (M+1).
Example 410
Synthesis of 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01043##
[1780] Step 1. Preparation of methyl 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01044##
[1782] Following the procedure as described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace (R)-(1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with 5-(chloromethyl)-3-(2-chlorophenyl)-1,2,4-oxadiazole and adding tetra-n-butylammonium iodide to the reaction mixture, the title compound was obtained as a colorless syrup (1.13 g, 85%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.92 (dd, J =2.0, 7.4 Hz, 1H), 7.52 (dd, J=1.3, 7.8 Hz, 1H), 7.46-7.34 (m, 3H), 6.52 (d, J=12.7 Hz, 1H), 3.97 (s, 2H), 3.86-3.83 (m, 5H), 3.09-3.05 (m, 2H), 2.37-2.29 (m, 2H), 2.05-1.96 (m, 1H), 1.89-1.86 (m, 3H), 1.61-1.47 (m, 2H), 0.92-0.85 (m, 2H), 0.64-0.59 (m, 2H); MS(ES+) m/z 500.0, 502.0 (M+1).
Step 2. Preparation of 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01045##
[1784] Following the procedure as described in Example 354 step 2 and making non-critical variations as required to replace 5-cyclopropyl-2-fluoro-4-((1-((2-methylthiazol-4-yl)methyl)piperidin-4-yl- ) methoxy)benzoate with methyl 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl)methyl) piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained following trituration in diethyl ether (10 mL) as a colorless solid (0.43 g, 39%): MS(ES+) m/z 488.0, 486.0 (M+1).
Step 3. Preparation of 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01046##
[1786] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5 -dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-((3-(2-chlorophenyl)-1,2,4-oxadiazol-5-yl) methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.08 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.76 (br s, 2H), 7.93 (dd, J=1.8, 7.6 Hz, 1H), 7.70-7.52 (m, 3H), 7.10 (d, J=8.3 Hz, 1H), 6.94 (d, J=13.0 Hz, 1H), 4.68 (br s, 2H), 3.97 (d, J=5.1 Hz, 2H), 3.53-3.48 (m, 2H), 3.30 (s, 3H), 3.03-2.92 (m, 2H), 2.05-1.92 (m, 4H), 1.66-1.54 (m, 2H), 0.89-0.83 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 562.9, 564.9 (M+1).
Example 411
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-((3,5-dichlorophenyl) (phenyl)methyl)piperidin-4-yl)methoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01047##
[1787] Step 1. Preparation of methyl 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)piperidin-4-yl) methoxy)-2-fluorobenzoate
##STR01048##
[1789] Following the procedure as described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzensulfonate with 1-(bromo(phenyl)methyl)-3,5-dichlorobenzene and adding tetra-n-butylammonium iodide to the reaction mixture, the title compound was obtained as a colorless syrup (0.66 g, 70%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.42 (d, J =8.3 Hz, 1H), 7.33-7.21 (m, 7H), 7.16-7.15 (m, 1H), 6.52(d, J=12.8 Hz, 1H), 4.21 (s, 1H), 3.86-3.82 (m, 5H), 2.92-2.84 (m, 2H), 2.02-1.76 (m, 6H), 1.54-1.37 (m, 2H), 0.90-0.84 (m, 2H), 0.64-0.58 (m, 2H); MS(ES+) m/z 544.0, 542.0 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl) piperidin-4-yl)methoxy)-2-fluorobenzoic acid
##STR01049##
[1791] To a solution of methyl 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)piperidin-4-yl) methoxy)-2-fluorobenzoate (0.66 g, 1.20 mmol) in tetrahydrofuran (20 mL) and water (5 mL) was added lithium hydroxide (0.30 g, 12.00 mmol). The reaction mixture was refluxed for 16 hours, then cooled to ambient temperature, diluted with 1 M hydrochloric acid solution (100 mL) and extracted with dichloromethane (100 mL). The solid present in the organic layer was filtered and washed with diethyl ether (20 mL) to afford the title compound as a colorless solid (0.25 g, 39%). The aqueous layer was further extracted with dichloromethane (2.times.75 mL). The organic layers were combined with the filtrate from the previous filtration, dried over anhydrous magnesium sulfate, filtered, and concentrated in vacuo to afford additional amount of the title compound as a colorless solid (0.32 g, 50%): MS(ES+) m/z 528.1, 530.0 (M+1).
Step 3. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-((3,5-dichlorophenyl) (phenyl)methyl)piperidin-4-yl)methoxy)-2-fluorobenzamide trifluoroacetic acid salt
##STR01050##
[1793] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-((1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-cyclopropyl-4-((1-(3,5-dichlorophenyl) (phenyl)methyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.09 g, 30%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.69 (s, 2H), 7.61-7.56 (m, 3H), 7.47-7.35 (m, 3H), 7.06 (d, J=8.3 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 5.44 (br s, 1H), 3.96-3.94 (m, 2H), 3.29-3.11 (m, 2H), 3.05-2.86 (m, 3H), 2.10-1.92 (m, 4H), 1.66-1.54 (m, 2H), 1.08-1.02 (m, 4H), 0.86-0.80 (m, 2H), 0.61-0.56 (m, 2H); MS(ES+) m/z 631.1, 633.0 (M+1).
Example 412
Synthesis of 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01051##
[1795] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-cyclopropyl-4-((1-((3,5-dichlorophenyl) (phenyl)methyl)piperidin-4-yl)methoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.03 g, 11%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.69 (s, 2H), 7.62-7.57 (m, 3H), 7.48-7.35 (m, 3H), 7.07 (d, J=8.3 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 5.44 (brs, 1H), 3.96-3.95 (m, 2H), 3.28 (s, 3H), 3.19-2.28 (m, 4H), 2.07-1.92 (m, 4H), 1.65-1.55 (m, 2H), 0.86-0.80 (m, 2H), 0.63-0.58 (m, 2H); MS(ES+) m/z 605.1, 607.1 (M+1).
Example 413
Synthesis of 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-flu- oro-N-(methylsulfonyl) benzamide, trifluoroacetic acid salt
##STR01052##
[1796] Step 1. Preparation of methyl 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01053##
[1798] Following the procedure described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with 4-bromo-2-chloro-1-(chloromethyl)benzene and adding tetra-n-butylammonium iodide to the reaction mixture, the title compound was obtained as a light yellow solid (1.55 g, 86%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.49 (br s, 1H), 7.44-7.35 (m, 3H), 6.52 (dd, J=2.5, 12.8 Hz, 1H), 3.87-3.82 (m, 5H), 3.55 (d, J=2.5 Hz, 2H), 2.93-2.89 (m, 2H), 2.16-1.81 (m, 6H), 1.50-1.38 (m, 2H), 0.92-0.85 (m, 2H), 0.65-0.61 (m, 2H); MS(ES+) m/z 514.0, 512.0, 510,0 (M+1).
Step 2. Preparation of 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-flu- orobenzoic acid
##STR01054##
[1800] Following the procedure as described in Example 411 step 2 and making non-critical variations as required to replace methyl 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-- fluorobenzoate with methyl 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained was obtained following trituration in diethyl ether (1mL) as a light pink solid (0.10 g, 22%): MS(ES+) m/z 500.0, 498.2, 496.0 (M+1).
Step 3. Preparation of 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01055##
[1802] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-2- -fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.02 g, 21%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 9.48 (br s, 1H), 7.89 (d, J=1.7 Hz, 1H), 7.70 (dd, J=1.7, 8.3 Hz, 1H), 7.60 (d, J=8.3 Hz, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.94 (d, J=12.9 Hz, 1H), 4.35 (s, 2H), 3.94 (brs, 2H), 3.40 (br s, 2H), 3.30 (s, 3H), 3.14-3.04 (m, 2H), 2.08-1.92 (m, 4H), 1.62-1.47 (m, 2H), 0.88-0.82 (m, 2H), 0.68-0.62 (m, 2H); MS(ES+) m/z 577.0, 575.0, 573.0 (M+1).
Example 414
Synthesis of 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01056##
[1804] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.02 g, 26%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.80 (br s, 1H), 9.46 (br s, 1H), 7.89 (d, J=1.6 Hz, 1H), 7.70 (dd, J=1.6, 8.3 Hz, 1H), 7.60 (d, J=8.3 Hz, 1H), 7.09 (d, J=8.3 Hz, 1H), 6.95 (d, J=12.9 Hz, 1H), 4.35 (s, 2H), 3.95-3.94 (m, 2H), 3.39 (br s, 2H), 3.13-2.99 (m, 3H), 2.04-1.93 (m, 4H), 1.61-1.46 (m, 2H), 1.09-1.06 (m, 4H), 0.89-0.82 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 603.0, 601.0, 599.0 (M=1).
Example 415
Synthesis of 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01057##
[1805] Step 1. Preparation of methyl 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01058##
[1807] To a solution of methyl 4-((1-(4-bromo-2-chlorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-flu- orobenzoate (1.03 g, 2.02 mmol) in anhydrous 1,4-dioxane (15 mL) was added cyclopropylboronic acid (1.06 g, 12.30 mmol), tribasic potassium phosphate (1.74 g, 8.22 mmol), and tetrakis(triphenylphosphine)palladium(0) (0.48 g, 0.42 mmol). The suspension was degassed used with argon and heated at 100.degree. C. for 30 minutes in a microwave reactor. After cooling to ambient temperature, the reaction mixture was diluted with ethyl acetate (100 mL), washed with saturated aqueous ammonium chloride (2.times.100 mL), dried over anhydrous magnesium sulfate, filtered and concentrated in vacuo. The residue was purified by column chromatography eluting with a 0-20% gradient of ethyl acetate with 10% isopropanol, 10% triethylamine in hexanes to afford the title compound as a yellow syrup (0.94 g, 99%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.42 (d, J=8.3 Hz, 1H), 7.31 (d, J=7.9 Hz, 1H), 7.02 (d, J=1.6 Hz, 1H), 6.92 (dd, J=1.6, 7.9 Hz, 1H), 6.52 (d, J=12.8 Hz, 1H), 3.86-3.81 (m, 5H), 3.57 (s, 2H), 2.96-2.92 (m, 2H), 2.13-1.96 (m, 3), 1.87-1.80 (m, 4H), 1.49-1.37 (m, 2H), 0.98-0.85 (m, 4H), 0.55-0.50 (m, 4H); MS(ES+) m/z 474.1, 472.1 (M=1).
Step 2. Preparation of 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01059##
[1809] To a solution of methyl 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl- -2-fluorobenzoate (0.93 g, 2.00 mmol) in tetrahydrofuran (25 mL) and water (7 mL) was added lithium hydroxide (0.48 g, 20.00 mmol). The mixture was refluxed for 15 hours and cooled to ambient temperature. The reaction mixture was acidified with 1 M aqueous hydrochloric acid solution (10 mL), diluted with saturated aqueous ammonium chloride (150 mL) and extracted with ethyl acetate (2.times.150 mL). The combined organic layers were dried over anhydrous magnesium sulfate, filtered and concentrated in vacuo. The residue was triturated in diethyl ether (15 mL) to afford the title compound as a colorless solid (0.16 g, 18%). The aqueous layer contained white solid that was filtered, washed with water (100 mL) and diethyl ether (40 mL) to afford additional amount of the title compound as a colorless solid (0.24 g, 27%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 10.25 (br s, 1H), 7.70 (d, J=7.9 Hz, 1H), 7.27-7.25 (m, 2H), 7.11 (d, J=7.9 Hz, 1H), 6.88 (d, J=13.0 Hz, 1H), 4.31 (d, J=3.7 Hz, 2H), 3.92 (d, J=5.4 Hz, 2H), 3.40-3.31 (m, 5H), 3.11-3.00 (m, 2H), 2.06-1.90 (m, 5H), 1.76-1.68 (m, 2H), 1.01-0.95 (m, 2H), 0.89-0.83 (m, 2H), 0.75-0.70 (m, 2H), 0.57-0.52 (m, 2H); MS(ES+) m/z 460.0, 558.2. (M=1).
Step 3. Preparation of 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01060##
[1811] Following the procedure as described in Example 402 Step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.06 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.84 (br s, 1H), 9.57 (s, 1H), 7.51 (d, J=8.0 Hz, 1H), 7.28-7.27 (m, 1H), 7.14-7.08 (m, 2H), 6.94 (d, J=12.9 Hz, 1H), 4.33 (s, 2H), 3.94 (s, 2H), 3.39 (br s, 2H), 3.30 (s, 3H), 3.12-3.04 (m, 2H), 2.10-1.92 (m, 5H), 1.64-1.55 (m, 2H), 1.02-0.96 (m, 2H), 0.88-0.82 (m, 2H), 0.75-0.70 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 537.1, 535.1 (M=1).
Example 416
Synthesis of 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01061##
[1813] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl)methoxy)-2- -fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.05 g, 26%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.81 (br s, 1H), 9.47 (br s, 1H), 7.51 (d, J=8.0 Hz, 1H), 7.29-7.28 (m, 1H), 7.14-7.07 (m, 2H), 6.95 (d, J=13.0 Hz, 1H), 4.33 (s, 2H), 3.94-3.93 (m, 2H), 3.43-3.40 (m, 2H), 3.13-2.99 (m, 3H), 2.07-1.92 (m, 5H), 1.62-1.50 (m, 2H), 1.09-1.06 (m, 4H), 1.02-0.96 (m, 2H), 0.88-0.82 (m, 2H), 0.75-0.70 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 563.1, 561.2 (M=1).
Example 417
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzamide, trifluoroacetic acid salt
##STR01062##
[1815] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(2-chloro-4-cyclopropylbenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.06 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.59 (br s, 1H), 9.38 (br s, 1H), 7.51 (d, J=8.0 Hz, 1H), 7.29-7.28 (m, 1H), 7.15-7.07 (m, 2H), 6.95 (d, J=12.8 Hz, 1H), 4.33 (s, 2H), 4.01 (t, J=7.7 Hz, 4H), 3.94-3.93 (m, 2H), 3.44-3.40 (m, 2H), 3.12-3.04 (m, 2H), 2.18-1.91 (m, 7H), 1.62-1.50 (m, 2H), 1.02-0.96 (m, 2H), 0.89-0.82 (m, 2H), 0.75-0.70 (m, 2H), 0.68-0.63 (m, 2H); MS(ES+) m.z 578.0, 576.0 (M=1).
Example 418
Synthesis of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01063##
[1816] Step 1. Preparation of methyl 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01064##
[1818] Following the procedure as described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonamide with 4,4'-(bromomethylene)bis(fluorobenzene) and adding tetra-N-butylammonium iodide to the reaction mixture, the title compound was obtained as a light yellow syrup (0.19 g, 18%): .sup.1H NMR (300 MHz, CDCl.sub.3), .delta. 7.42 (d, J=8.3 Hz, 1H), 7.36-7.29 (m, 4H), 6.98-6.92 (m, 4H), 6.52 (d, J=12.8 Hz, 1H), 4.26 (s, 1H), 3.86 (s, 3H), 3.83 (d, J=6.1 Hz, 2H), 2.90-2.86 (m, 2H), 2.01-1.96 (m, 1H), 1.91-1.77 (m, 5H), 1.49-1.38 (m, 2H), 0.90-0.83 (m, 2H), 0.64-0.58 (m, 2H); MS(ES+) m/z 510.2 (M=1).
Step 2. Preparation of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01065##
[1820] To a solution of methyl 4-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (0.19 g, 0.38 mmol) in tetrahydrofuran (10 mL) and water (3 mL) was added lithium hydroxide (0.10 g, 4.10 mmol). The mixture was refluxed for 6 hours, and then cooled to ambient temperature. The mixture was diluted with 1 M hydrochloric acid solution (100 mL) and extracted with ethyl acetate (2.times.100 mL). The combined organic layers were dried over anhydrous magnesium sulfate, filtered, and concentrated in vacuo to afford the title compound as a colorless syrup (0.19 g, quant. yield) that was used directly in the next reaction: MS(ES+) m/z 496.0 (M=1).
Step 3. Preparation of 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01066##
[1822] Following the procedure as described in Example 402 Step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.04 g, 27% in 2 steps): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.0 11.88 (br s, 1H), 9.98 (br s, 1H), 7.67 (br s, 4H), 7.31 (brs, 4H), 7.09 (d, J=8.2 Hz, 1H), 6.95 (d, J=13.0 Hz, 1H), 5.62-5.59 (m, 1H), 3.95 (d, J =5.0 Hz, 2H), 3.30 (s, 3H), 3.26-3.22 (m, 2H), 3.04-2.95 (m, 2H), 2.05-1.96 (m, 3H), 1.84-1.81 (m, 1H), 1.60-1.55 (m, 2H), 0.87-0.82 (m, 2H), 0.66-0.61 (m, 2H); MS(ES+) m/z 573.1 (M=1).
Example 419
Synthesis of N-azetidin-1-ylsulfonyl)-4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoorobenzamide, trifluoroacetic acid salt
##STR01067##
[1824] Following the procedure as described in Example 402 Step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.04 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.59 (br s, 1H), 10.24 (br s, 1H), 7.72 (br s, 4H), 7.31 (br s, 4H), 7.09 (d, J=8.2 Hz, 1H), 6.96 (d, J=12.5 Hz, 1H), 4.01 (t, J=7.7, 7.7 Hz, 4H), 3.95 (d, J=4.4 Hz, 2H), 3.26-3.22 (m, 2H), 2.15-1.97 (m, 4H), 1.85-1.65 (m, 2H), 1.14 (t, J=7.3, 7.3 Hz, 2H), 0.87-0.82 (m, 2), 0.66-0.61 (m, 2H); MS(ES+) m/z 614.1 (M=1).
Example 420
Synthesis of 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl)methyl)-3-methylazetidin- -3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01068##
[1825] Step 1. Preparation of methyl 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl) methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzoate
##STR01069##
[1827] Following the procedure as described in Example 402 step 1 and making non-critical variations as required to replace tert-butyl 5-chloro-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride, and to replace (R)-1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate with 1-(bromo(phenyl)methyl)-3,5-dichlorobenzene and adding tetra-n-butylammonium iodide to the reaction mixture, the title compound was obtained as a light yellow syrup (0.56 g, 62%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=8.3 Hz, 1H), 7.37-7.20 (m, 2H), 7.16-7.15 (m, 1H), 6.61 (d, J=12.6 Hz, 1H), 4.32 (s, 1H), 4.02 (s, 2H), 3.88 (s, 3H), 3.21 (dd, J=7.4, 12.0 Hz, 2H), 2.89 (dd, J=7.4, 9.8 Hz, 2H), 2.06-1.97 (m, 1H), 1.38 (s, 3H), 0.92-0.86 (m, 2H), 0.67-0.62 (m, 2H); MS(ES+) m/z 530.0, 528.0 (M=1).
Step 2. Preparation of 5-cyclopropyl-4-((1-(3,5-dichlorophenyl)(phenyl) methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzoic acid
##STR01070##
[1829] Following the procedure as described in Example 402 step 2 and making non-critical variations as required to replace methyl 4-((1-(bis(4-fluorophenyl)methyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2-- fluorobenzoate with methyl 5-cyclopropyl-4-((1-(3,5-dichlorophenyl)(phenyl) methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzoate, the title compound was obtained following trituration in diethyl ether (15 mL) (0.44 g, 80%) as a colorless solid: MS(ES+) m/z 516.1, 514.1 (M=1).
Step 3. Preparation of 5-cyclopropyl-4-((1-((3,5-dichlorophenyl)(phenyl) methyl)-3-methylazetidin-3-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzami- de, trifluoroacetic acid salt
##STR01071##
[1831] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzinc acid, trifluoroacetic acid salt with 5-cyclopropyl-4-((1-((3,5-dichlorophenyl) (phenyl)methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.11 g, 37%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.60 (s, 1H), 7.52-7.51 (m, 2H), 7.45-7.35 (m, 5H), 7.13 (d, J=8.3 Hz, 1H), 6.91 (d, J=12.8 Hz, 1H), 5.50 (br s, 1H), 4.13 (br s, 2H), 3.89-3.57 (m, 4H), 3.29 (s, 3H), 2.04-1.95 (m, 1H), 1.40 (s, 3H), 0.85-0.78 (m, 2H), 0.65-0.56 (m, 2H); MS(ES+) m/z 592.9, 590.9 (M+1).
Example 421
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-((3,5-dichlorophenyl)(phenyl) methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01072##
[1833] Following the procedure as described in Example 402 step 3 and making non-critical variations as required to replace (S)-5-chloro-4-((1-(1-(3,5-dichlorophenyl)ethyl)piperidin-4-yl) methoxy)-2-fluorobenzoic acid, trifluoroacetic acid salt with 5-cyclopropyl-4-((1-((3,5-dichlorophenyl) (phenyl)methyl)-3-methylazetidin-3-yl)methoxy)-2-fluorobenzoic acid and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.14 g, 47%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.61 (s, 1H), 7.54-7.53 (m, 2H), 7.47-7.35 (m, 5H), 7.12 (d, J=8.3 Hz, 1H), 6.91 (d, J=12.8 Hz, 1H), 5.60 (br s, 1H), 4.14 (br s, 2H), 3.98-3.91 (m, 2H), 3.82-3.76 (m, 2H), 3.06-2.98 (m, 1H), 2.04-1.94 (m, 1H), 1.42 (s, 3H), 1.09-1.06 (m, 4H), 0.84-0.78 (m, 2H), 0.62-0.57 (m, 2H); MS(ES+) m/z 616.9, 618.9 (M+1).
Example 422
Synthesis of (R)-4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluuoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01073##
[1834] Step 1. Preparation of (R)-4-((1-(tert-butoxycarbonyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01074##
[1836] To a solution of (R)-tert-butyl 3-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)piperidine-1-car- boxylate (3.50 g, 8.05 mmol) in dichloromethane (20 mL) trifluoroacetic acid (5 mL) was added. The mixture was stirred for 1.5 hours and then a saturated aqueous solution of sodium bicarbonate (25 mL) was added followed by di-tert-butyl dicarbonate (1.93 g, 8.85 mmol). The reaction mixture was stirred for 1 hour, acidified with 1M aqueous hydrochloric acid solution to pH=1 and then extracted with dichloromethane (2.times.20 mL). The organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated to give the title compound as a colorless solid (3.05 g, quant. yield): MS(ES+) m/z 380.2 (M+1).
Step 2. Preparation of (R)-tert-butyl 3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)piperidine-1-carboxylate
##STR01075##
[1838] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (R)-4-((1-(tert-butoxycarbonyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid and the residue was purified by column chromatography eluting with gradient of 0% to 10% methanol containing 1% ammonia in dichloromethane, the title compound was obtained as a colorless solid (2.76 g, 75%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.89-8.45 (brs, 1H), 7.56 (d, J=9.0 Hz, 1H), 6.61 (d, J=14.5 Hz, 1H), 4.41-4.25 (m, 1H), 3.81-3.25 (m, 7H), 2.10-1.75 (m, 4H), 1.66-1.17 (m, 10H), 0.96-0.81 (m, 2H), 0.75-0.52 (m, 2H).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy)benzam- ide, trifluoroacetic acid salt
##STR01076##
[1840] Following the procedure as described in Example 346 step6, and making non-critical variations as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-fluoropiperidine-1-carboxylate with (R)-tert-butyl 3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (2.75 g, quant. yield): MS(ES+) m/z 357.1 (M+1).
Step 4. (R)-4-((1-(3-chloro-5-(trifluoromethoxy)benzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01077##
[1842] To a stirred solution of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy) benzamide trifluoroacetic (0.05 g, 0.10 mmol) and 3-chloro-5-(trifluoromethoxy)benzaldehyde (0.033 g, 0.15 mmol) in tetrahydrofuran (1 mL) was added sodium triacetoxyborohydride (0.073 g, 0.23 mmol). After stirring at ambient temperature for 16 hours, the reaction was quenched by adding 1 n aqueous hydrochloric acid solution (0.4 mL) and the mixture was purified by column chromatography eluting with 5% methanol in dichloromethane to give an oil, which was further purified by preparative HPLC to afford the title compound as a colorless solid (0.03 g, 45%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.19-11.69 (m, 1H), 10.04-9.66 (m, 1H), 7.76-7.62 (m, 2H), 7.61-7.49 (m, 1H), 7.14-7.01 (m, 2H), 5.01-4.58 (m, 1H), 4.57-4.18 (m, 2H), 3.67-2.77 (m, 7H), 2.42-1.49 (m, 5H), 0.96-0.76 (m, 2H), 0.73-0.60 (m, 2H); MS(ES+) m/z 567.0, 565.0 (M+1).
Example 423
Synthesis of (R)-5-cyclopropyl-4-((1-(2,4-dimethylbenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR01078##
[1844] Following the procedure as described in Example 422 step 4, and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2,4-dimethylbenzaldehyde, the title compound was obtained as a colorless solid (0.02 g, 50%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. b 12.21-11.69 (m, 1H), 9.97-8.99 (m, 1H), 7.45-7.28 (m, 1H), 7.20-6.98 (m, 4H), 4.98-4.62 (m, 1H), 4.45-4.22 (m, 2H), 3.74-2.86 (m, 7H), 2.42-2.28 (m, 3H), 2.25 (s, 3H), 2.21-1.42 (m, 5H), 0.94-0.79 (m, 2H), 0.75-0.59 (m, 2H); MS(ES+) m/z 475.2 (M+1).
Example 424
Synthesis of (R)-4-((1-(2-chloro-4-methylbenzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01079##
[1846] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2-choro-4-methylbenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 61%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.24-11.69 (m, 1H), 9.78-9.46 (m, 1H), 7.57 (d, J=7.41 Hz, 1H), 7.41 (s, 1H), 7.25 (d, J=8.00 Hz, 1H), 7.18-7.01 (m, 2H), 5.03-4.65 (m, 1H), 4.54-4.37 (m, 2H), 3.75-3.35 (m, 2H), 3.31 (s, 3H), 3.28-2.89 (m, 2H), 2.30 (s, 3H), 2.25-1.51 (m, 5H), 0.94-0.79 (m, 2H), 0.74-0.60 (m, 2H); MS(ES+) m/z 497.0, 495.0 (M+1).
Example 425
Synthesis of (R)-4-((1-(4-(tert-butyl)piperidin)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01080##
[1848] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 4-(tert-butyl)benzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 61%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.14-11.80 (m, 1H), 9.96-9.16 (m, 1H), 7.52-7.33 (m, 4H), 7.17-6.98 (m, 2H), 4.99-4.58 (m, 1H), 4.46-4.17 (m, 2H), 3.72-3.37 (m, 2H), 3.36-2.78 (m, 5H), 2.42-1.49 (m, 5H), 1.25 (s, 9H), 0.94-0.80 (m, 2H), 0.74-0.59 (m, 2H); MS(ES+) m/z 503.2 (M+1).
Example 426
Synthesis of (R)-4-((1-([1,1'-biphenyl]-4-ylmethyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01081##
[1850] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with [1,1'-biphenyl]-4-carbaldehyde, the title compound was obtained as a colorless solid (0.02 g, 36%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.14-11.77 (m, 1H), 10.15-9.40 (m, 1H), 7.76 (d, J=8.1 Hz, 2H), 7.67 (d, J=7.3 Hz, 2H), 7.64-7.54 (m, 2H), 7.46 (t, J=7.5 Hz, 2H), 7.41-7.33 (m, 1H), 7.19-7.01 (m, 2H), 5.02-4.61 (m, 1H), 4.55-4.29 (m, 2H), 3.76-3.44 (m, 2H), 3.40-2.86 (m, 5H), 2.42-1.53 (m, 5H), 0.95-0.79 (m, 2H), 0.72-0.61 (m, 2H); MS(ES+) m/z 523.2 (M+1).
Example 427
Synthesis of (R)-4-((1-(5-chloro-2-hydroxybenzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-4-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01082##
[1852] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 5-chloro-2-hydroxybenzaldehyde, the title compound was obtained as a colorless solid (0.04 g, 59%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.10-11.77 (m, 1H), 10.72-10.50 (m, 1H), 10.16-9.18 (m, 1H), 7.58-7.47 (m, 1H), 7.30 (dd, J=8.6, 2.1 Hz, 1H), 7.17-7.03 (m, 2H), 6.93 (d, J=8.4 Hz, 1H), 5.01-4.68 (m, 1H), 4.35-4.21 (m, 2H), 3.70-2.85 (m, (m, 7H), 2.41-1.56 (m, 5H), 0.95-0.79 (m, 2H), 0.72-0.61 (m, 2H); MS(ES+) m/z 499.1, 497.1 (M+1).
Example 428
Synthesis of (R)-5-cyclopropyl-4-((1-(2,4-diethoxybenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01083##
[1854] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2,4-diethoxybenzaldehyde, the title compound was obtained as a colorless solid (0.04 g, 77%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta.12.13-11.74 (m, 1H), 9.87-9.15 (m, 1H), 7.34 (dd, J=8.7, 8.7 Hz, 1H), 7.18-7.02 (m, 2H), 6.62-6.52 (m, 2H), 5.02-4.65 (m, 1H), 4.38-4.16 (m, 2H), 4.11-3.96 (m, 4H), 3.69-2.82 (m, 7H), 2.43-2.09 (m, 1H), 2.08-1.56 (m, 4H), 1.38-1.22 (m, 6H), 0.93-0.78 (m, 2H), 0.76-0.57 (m, 2H); MS(ES+) m/z 535.1. (M+1).
Example 429
Synthesis of (R)-5-cyclopropyl-4-((1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)methyl)piper- idin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01084##
[1856] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2,3-dihydrobenzo[b][1,4]dioxine-6-carbaldehyde, the title compound was obtained as a colorless solid (0.03 g, 64%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.09-11.82 (m, 1H), 9.90-9.22 (m, 1H), 7.18-6.99 (m, 3H), 6.98-6.87 (m, 2H), 4.98-4.60 (m, 1H), 4.39-4.09 (m, 6H), 3.69-2.75 (m, 7H), 2.38-1.49 (m, 5H), 0.95-0.79 (m, 2H), 0.75-0.60 (m, 2H); MS(ES+) m/z 505.1 (M+1).
Example 430
Synthesis of (R)-5-cyclopropyl-4-((1-(3,4-dimethoxybenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01085##
[1858] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluorormethoxy)benzaldehyde with 3,4-dimethoxybenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 71%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.09-11.81 (m, 1H), 9.95-9.19 (m, 1H), 7.18-6.94 (m, 5H), 4.97-4.62 (m, 1H), 4.43-4.13 (m, 2H), 3.80-3.68 (m, 6H), 3.67-3.36 (m, 2H), 3.31 (s, 3H), 3.25-2.82 (m, 2H), 2.39-2.10 (m, 1H), 2.09-1.52 (m, 4H), 0.97-0.76 (m, 2H), 0.74-0.61 (m, 2H); MS(ES+) m/z 507.1 (M+1).
Example 431
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2-methylbenzyl)piperidin-3-yl) oxy)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01086##
[1860] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 4-fluoro-2-methylbenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 82%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.13-11.78 (m, 1H), 9.82-9.03 (m, 1H), 7.63-7.43 (m, 1H), 7.23-7.00 (m, 4H), 4.96-4.62 (m, 1H), 4.44-4.27 (m, 2H), 3.71-2.91 (m, 7H), 2.43-2.13 (m, 4H), 2.13-1.47 (m, 4H), 0.94-0.79 (m, 2H), 0.75-0.60 (m, 2H); MS(ES+) m/z 479.0 (M+1).
Example 432
Synthesis of (R)-4-((1-(2-chloro-4-methoxybenzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01087##
[1862] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2-chloro-4-methoxybenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 67%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.17-11.73 (m, 1H), 9.99-9.27 (m, 1H), 7.67-7.52 (m, 1H), 7.20-6.97 (m, 4H), 5.03-4.60 (m, 1H), 4.48-4.34 (m, 2H), 3.78 (s, 3H), 3.67-2.88 (m, 7H), 2.42-1.51 (m, 5H), 0.95-0.78 (m, 2H), 0.76-0.61 (m, 2H); MS(ES+) m/z 513.1, 511.1 (M+1).
Example 433
Synthesis of (R)-4-((1-(3-chloro-4-(trifluoromethyl)piperidin)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01088##
[1864] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 3-chloro-4-(trifluoromethyl)benzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 69%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.19-11.63 (m, 1H), 9.79-9.50 (m, 1H), 8.12-7.95 (m, 1H), 7.91-7.69 (m, 2H), 7.16-6.98 (m, 2H), 5.00-4.58 (m, 1H), 4.56-4.27 (m, 2H), 3.70-2.77 (m, 7H), 2.42-1.48 (m, 5H), 0.97-0.76 (m, 2H), 0.75-0.59 (m, 2H); MS(ES+) m/z 551.0, 549.0 (M+1).
Example 434
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(4-methoxy-2-methylbenzyl)piperidin-3-yl- ) oxy)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01089##
[1866] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 4-methoxy-2-methylbenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 71%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.09-11.80 (m, 1H), 9.72-8.85 (m, 1H), 7.41 (dd, J=16.1, 8.6 Hz, 1H), 7.17-7.00 (m, 2H), 6.89-6.78 (m, 2H), 4.96-4.63 (m, 1H), 4.39-4.20 (m, 2H), 3.72 (s, 3H), 3.68-3.36 (m, 2H), 3.31 (s, 3H), 3.24-2.91 (m, 2H), 2.34 (s, 3H), 2.27-1.51 (m, 5H), 0.94-0.79 (m, 2H), 0.74-0.58 (m, 2H); MS(ES+) m/z 491.2 (M+1).
Example 435
Synthesis of (R)-4-((1-(3-chloro-4-methylbenzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01090##
[1868] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzylaldehyde with 3-chloro-4-methylbenzaldehyde, the title compound was obtained as a colorless solid (0.04 g, 91%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.16-11.76 (m, 1H), 9.74-9.43 (m, 1H), 7.58-7.43 (m, 2H), 7.41-7.28 (m, 1H), 7.19-7.01 (m, 2H), 4.97-4.64 (m, 1H), 4.44-4.20 (m, 2H), 3.70-3.39 (m, 2H), 3.31 (s, 3H), 3.26-2.85 (m, 2H), 2.31 (s, 3H), 2.26-1.46 (m, 5H), 0.95-0.77 (m, 2H), 0.73-0.61 (m, 2H); MS(ES+) m/z 497.1, 495.1 (M+1).
Example 436
Synthesis of (R)-5-cyclopropyl-4-((1-(3,5-dimethylbenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01091##
[1870] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 3,5-dimethylbenzaldehyde, the title compound was obtained as a colorless solid (0.02 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.21-11.75 (m, 1H), 10.27-9.47 (m, 1H), 7.20-6.96 (m, 5H), 5.00-4.62 (m, 1H), 4.43-4.12 (m, 2H), 3.70-3.37 (m, 2H), 3.31 (s, 3H), 3.27-2.85 (m, 2H), 2.42-2.11 (m, 7H), 2.07-1.54 (m, 4H), 0.94-0.77 (m, 2H), 0.75-0.57 (m, 2H); MS(ES+) m/z 475.2 (M+1).
Example 437
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(3-(trifluoromethoxy) benzyl)piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01092##
[1872] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 3-(trifluoromethoxy)benzaldehyde, the title compound was obtained as a colorless solid (0.02 g, 33%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.23-11.62 (m, 1H), 9.77-9.45 (m, 1H), 7.68-7.39 (m, 3H), 7.20-6.98 (m, 2H), 5.03-4.59 (m, 1H), 4.59-4.26 (m, 2H), 3.67-2.77 (m, 7H), 2.42-1.48 (m, 5H), 1.20-0.57 (m, 4H); MS(ES+) m/z 531.1 (M+1).
Example 438
Synthesis of (R)-5-cyclopropyl-2-fluoro-4-((1-(2-methoxy-4-(trifluoromethoxy) benzyl)piperidin-3-yl)oxy)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01093##
[1874] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 2-methoxy-4-(trifluoromethoxy)benzaldehyde, the title compound was obtained as a colorless solid (0.04 g, 60%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.18-11.73 (m, 1H), 9.25-9.01 (m, 1H), 7.63-7.49 (m, 1H), 7.19-6.98 (m, 4H), 5.00-4.61 (m, 1H), 4.41-4.24 (m, 2H), 3.90-3.77 (m, 3H), 3.74-2.83 (m, 7H), 2.42-1.53 (m, 5H), 0.95-0.79 (m, 2H), 0.74-0.61 (m, 2H); MS(ES+) m/z 561.1 (M+1).
Example 439
Synthesis of (R)-5-cyclopropyl-4-((1-(3,4-dimethylbenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01094##
[1876] Following the procedure as described in Example 426 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 3,4-dimethylbenzaldehyde, the title compound was obtained as a colorless solid (0.03 g, 60%): .sup.1H NMR (300 MHz, DMSO-.sub.6).delta. 12.09-11.80 (m, 1H), 10.35-9.35 (m, 1H), 7.34-7.16 (m, 3H), 7.16-6.98 (m, 3H), 4.98-4.67 (m, 1H), 4.42-4.12 (m, 2H), 3.69-3.36 (m, 2H), 3.31 (s, 3H), 3.27-2.79 (m, 2H), 2.42-2.12 (m, 7H), 2.06-1.51 (m, 4H), 0.95-0.78 (m, 2H), 0.73-0.59 (m, 2H); MS(ES+) m/z 475.2 (M+1).
Example 440
Synthesis of (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01095##
[1878] Following the procedure as described in Example 422 step 4 and making variation as required to replace 3-chloro-5-(trifluoromethoxy)benzaldehyde with 3,5-dichlorobenzaldehyde and to purify the residue using column chromatography eluting with 5% methanol containing 0.5% ammonia in dichloromethane, the title compound was obtained as a colorless solid (0.12 g, 72%): .sup.1H NMR (300 MHz, CDCl.sub.3).delta. 7.53 (d, J=9.1 Hz, 1H), 7.27-7.16 (m, 3H), 6.55 (d, J=14.5 Hz, 1H), 4.50-4.36 (m, 1H), 3.56-3.41 (m, 2H), 3.39 (s, 3H), 2.86 (d, J=10.4 Hz, 1H), 2.67-2.56 (m, 1H), 2.45-2.33 (m, 1H), 2.32-2.21 (m, 1H), 2.13-1.97 (m, 2H), 1.94-1.78 (m, 1H), 1.74-1.54 (m, 2H), 1.00-0.87 (m, 2H), 0.73-0.59 (m, 2H); MS(ES+) m/z: 515.0, 517.1 (M+1).
Example 441
Synthesis of (R)-4-((1-(2-chloro-4-(methylsulfonyl)piperidin)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01096##
[1880] To a stirred solution of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy) benzamide trifluoroacetate (0.05 g, 0.14 mmol) and 1-(bromomethyl)-2-chloro-4-(methylsulfonyl)benzene (0.06 g, 0.21 mmol) in acetonitrile (2 mL) was added potassium carbonate (0.05 g, 0.34 mmol) and potassium iodide (0.03 g, 0.17 mmol). The reaction mixture was stirred at reflux for 16 hours, cooled to ambient temperature, and then 1N aqueous hydrochloric acid solution (5 mL) was added; extracted with ethyl acetate (3.times.10 mL) and concentrated invacuo. The residue was purified by column chromatography eluting with gradient of 0% to 30% ethyl acetate containing 1% formic acid in hexanes to obtained an oil, which was further purified by preparative HPLC (gradient of acetonitrile in water) to afford the title compound (0.04 g, 44%) as a colorless solid: .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 7.90 (s, 1H), 7.78-7.71 (m, 2H), 7.16 (d, J=8.70 Hz, 1H), 6.85-6.67 (m, 1H), 4.57-4.45 (m, 1H), 3.66 (m, 2H), 3.22 (s, 3H), 2.85-2.72 (m, 2H), 2.62-2.50 (m, 1H), 2.40-2.29 (m, 1H), 2.10-1.97 (m, 1), 1.97-1.85 (m, 1H), 1.84-1.72 (m, 1H), 1.63-1.44 (m, 2H), 0.90-0.79 (m, 2H), 0.63-0.51 (m, 2H); MS(ES+) m/z: 559.0, 561.0 (M+1).
Example 442
Synthesis of (R)-4-((1-([1,1'-biphenyl]-2-yl)methyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01097##
[1882] Following the procedure as described in Example 441 and making variation as required to replace 1-(bromomethyl)-2-chloro-4-(methylsulfonyl)benzene with 2-(bromomethyl)-1,1'-biphenyl, the title compound was obtained as a colorless solid (0.02 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.22-11.69 (m, 1H), 9.66-8.78 (m, 1H), 7.80-7.62 (m, 1H), 7.57-7.21 (m, 8H), 7.17-7.04 (m, 1H), 6.96 (d, J =12.9 Hz, 1H), 4.89-4.11 (m, 5H), 3.41-2.87 (m, 5H), 2.42-1.38 (m, 5H), 0.96-0.77 (m, 2H), 0.76-0.59 (m, 2H); MS(ES+) m/z 523.2 (M+1).
Example 443
Synthesis of (R)-4-((1-([1,1'-biphenyl]-3-ylmethyl)piperidin-3-yl) oxy-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01098##
[1884] Following the procedure as described in Example 441, and making variation as required to replace 1-(bromomethyl)-2-chloro-4-(methylsulfonyl)benzene with 3-(bromomethyl)-1,1'-biphenyl, the title compound was obtained as a colorless solid (0.01 g, 8%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.06-11.82 (m, 1H), 10.20-9.29 (m, 1H), 7.92-7.60 (m, 2H), 7.60-7.42 (m, 6H), 7.42-7.34 (m, 1H), 7.17-6.98 (m, 2H), 4.99-4.62 (m, 1H), 4.60-4.27 (m, 2H), 3.75-3.43 (m, 2H), 3.39-2.89 (m, 5H), 2.41-1.50 (m, 5H), 0.93-0.54 (m, 4H); MS(ES+) m/z 523.2 (M+1).
Example 444
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(spiro[3.5]nonan-7-yl- methyl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01099##
[1885] Step 1. Preparation of spiro[3.5]nonan-7-ylmethyl methanesulfonate
##STR01100##
[1887] To a solution of spiro[3.5]nonan-7-ylmethanol (0.77 g, 5.00 mmol) and triethylamine (1.05 mL, 7.50 mmol) in dichloromethane (20 mL) was added dropwise methanesulfonyl chloride (0.42 mL, 5.5 mmol) at 0.degree. C. The mixture was stirred for 1.5 hours, and then saturated aqueous solution of ammonium chloride (10 mL) was added and the mixture was extracted with dichloromethane (2.times.20 mL). The combined organic layers were washed with brine (15 mL), dried over anhydrous sodium sulfate, filtered and concentrated invacuo to afford the title compound (1.16 g, quant. yield), which was used next step without further purification.
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(spiro[3.5]nonan-7-yl- methyl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01101##
[1889] Following the procedure as described in Example 441, and making variation as required to replace 1-(bromomethyl)-2-chloro-4-(methylsulfonyl)benzene with spiro[3.5]nonan-7-ylmethyl methanesulfonate, the title compound was obtained as a colorless solid (0.02 g, 31%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.09-11.80 (m, 1H), 9.28-8.58 (m, 1H), 7.18-7.01 (m, 2H), 5.03-4.62 (m, 1H), 3.74-3.59 (m, 2H), 3.31 (s, 3H), 3.14-2.81 (m, 4H), 2.43-2.11 (m, 2H), 2.06-1.85 (m, 2H), 1.85-1.44 (m, 12H), 1.30-1.12 (m, 2H), 1.07-0.79 (m, 4H), 0.78-0.56 (m, 2H); MS(ES+) m/z 493.2 (M+1).
Example 445
Synthesis of (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide (
##STR01102##
[1890] Step 1. Preparation of (R)-3-((tert-butyldimethylsilyl)oxy)-1-(4-methoxy)piperidine-2,6-dione
##STR01103##
[1892] To a solution of (R)-3-hydroxy-1-(4-methoxybenzyl)piperidine-2,6-dione (prepared accordingly to Yuan-Ping et al., Chirality, 2005, 17, 595-599) (3.50 g, 14.00 mmol) in dichloromethane (20 mL) was added tert-butylchlorodimethylsilane (2.30 g, 15.50 mmol) and imidazole (1.30 g, 19.70 mmol). After stirring at ambient temperature for 1 hour, the reaction mixture was quenched with saturated aqueous ammonium chloride solution (25 mL and extracted with dichloromethane (2.times.40 mL), washed with brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated invacuo. The residue was purified by column chromatography (0% to 15% ethyl acetate in hexanes) to give the title compound (2.80 g, 55% 0); MS(ES+) m/z 364.1 (M+1).
Step 2. Preparation of (5R)-5-((tert-butyldimethylsilyl) oxy)-6-hydroxy-1-(4-methoxybenzyl)piperidin-2-one
##STR01104##
[1894] To a solution of (R)-3-((tert-butyldimethylsilyl)oxy)-1-(4-methoxybenzyl)piperidine-2,6-di- one (2.80 g. 7.70 mmol) in methanol (10 mL) and dichloromethane (10 mL) under nitrogen atmosphere at -30.degree. C. was added sodium borohydride (1.40 g, 38.50 mmol). After stirring for 30 minutes at -30.degree. C., the reaction mixture was warmed to 0.degree. C. over 30 minutes and quenched with saturated aqueous ammonium chloride solution (20 mL) and extracted with dichloromethane (2.times.15 mL). The combined extracts were dried over anhydrous sodium sulfate, filtered and concentrated invacuo to give an oil (2.70 g, 76%) which was used in the next step without further purification: MS(ES+) m/z 366.2 (M+1).
Step 3. Preparation of (R)-5-((tert-butyldimethylsilyl)oxy)-1-(4-methoxybenzyl)piperidin-2-one
##STR01105##
[1896] To a solution of (5R)-5-((tert-butyldimethylsilyl)oxy) 6-hydroxy-1-(4-methoxybenzyl)piperidin-2-one (2.70 g, 7.60 mmol) and triethylsilane (11.50 g, 77.10 mmol) in dry dichloromethane (20 mL) was added boron trifluoride diethyl ether complex (2.9 mL, 23.00 mmol) at -78.degree. C. under nitrogen atmosphere. After stirring for 30 minutes at -78.degree. C., the reaction mixture was warmed up slowly to 0.degree. C. and stirred at this temperature for 2 hours before it was quenched with saturated aqueous solution of sodium bicarbonate solation (5 mL) and extracted with dichloromethane (2.times.10 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered, an concentrated invacuo to afford the title compound as an oil (1.26 g, 47%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.16 (d, J=8.5 Hz, 2H), 6.82 (d, J=8.6 Hz, 2H), 4.71 (d, J=14.6 Hz, 1H), 4.27 (d, J=14.6 Hz, 1H), 4.08-3.98 (m, 1H), 3.77 (s, 3H), 3.24 (dd, J=12.4, 3.7 Hz, 1H), 3.05 (dd, J=12.3, 4.5 Hz, 1H), 2.68 (m, 1H), 2.39 (td, J=17.6, 5.7 Hz, 1H), 1.91-1.78 (m, 2H), 0.81 (s, 9H), 0.00 (s, 3H), -0.06 (s, 3H).
Step 4. Preparation of (R)-5-hydroxy-1-(4-methoxybenzyl)piperidin-2-one
##STR01106##
[1898] To a solution of (R)-5-((tert-butyldimethylsilyl)oxy)-1-(4-methoxybenzyl)piperidin-2-one (1.25 g, 3.58 mmol) in tetrahydrofuran (10 mL) was added tetra-butylammonium fluoride (1.0M in THF, 5.3 mL, 5.3 mmol) under nitrogen atmosphere. After stirring a ambient temperature for 30minutes, the reaction mixture was quenched with saturated aqueous ammonium chloride solution (2.5 mL) and extracted with dichloromethane (2.times.10 mL). The combined extracts were concentrated invacuo and filtered over a plug of silica gel eluting with 10% hexanes in ethyl acetate followed by ethyl acetate to afford the title compound as oil (0.84 g, quant. yield): MS(ES+) m/z 236.2 (M+1).
Step 5. Preparation of (R)-tert-butyl 5-chloro-2-fluoro-4-((1-(4-methoxybenzyl)-6-oxopiperidin-3-yl)oxy)benzoat- e
##STR01107##
[1900] To a solution of (R)-5-hydroxy-1-(4-methoxybenzyl)piperidin-2-one (0.84 g, 3.58 mmol) and tert-butyl 5-chloro-2,4-difluorobenzoate (0.98 g, 3.95 mmol) in dimethylsulfoxide (10 mL) was added cesium carbonate (1.52 g, 10.7 mmol). After stirring for 48 hours at 80.degree. C., the reaction mixture was quenched with saturated aqueous solution of ammonium chloride solution (20 mL) and extracted with dichloromethane (2.times.10 mL). The organic layers were concentrated under reduced pressure and the residue was purified by chromatography eluting with a gradient of ethyl acetate in hexanes (50% to 100%) to afford the title compound as a gum (1.38 g, 83%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.80 (d, J =7.7 Hz, 1H), 7.08 (d, J=8.6 Hz, 2H), 6.71 (d, J=8.6 Hz, 2H), 6.46 (d, J=11.9 Hz, 1H), 4.79 (d, J =14.6 Hz, 1H), 4.71-4.62 (m, 1H), 4.17 (d, J=14.6 Hz, 1H), 3.69 (s, 3H), 3.43-3.37 (m, 2H), 2.71 (ddd, J=17.6, 11.1, 6.6 Hz, 1H), 2.46 (ddd, J=17.7, 6.2, 3.7 Hz, 1H), 2.26-2.13 (m, 1H), 2.12-2.00 (m, 1H), 1.52 (s, 9H); MS(ES+) m/z 466.1, 464.1 (M+1).
Step 6. Preparation of (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(4-methoxybenzyl)-6-oxopiperidin-3-yl)oxy)be- nzoate
##STR01108##
[1902] Following the procedure as described in Example 346 step 3 and making variation as required to replace tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)-4-fluoropipe- ridine-1-carboxylate with (R)-tert-butyl 5-chloro-2-fluoro-4-((1-(4-methoxybenzyl)-6-oxopiperidin-3-yl) oxy)benzoate and the reaction mixture was heated at 150.degree. C. for 1 hour under microwave irradiation, the title compound was obtained as a colorless oil (0.90 g, 64%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.32 (d, J=8.4 Hz, 1H), 7.15-7.09 (m, 2H), 6.77-6.70 (m, 2H), 6.39 (d, J=12.4 Hz, 1H), 4.73-4.64 (m, 2H), 4.35-4.28 (m, 1H), 3.72 (s, 3H), 3.47-3.42 (m, 2H), 2.75-2.59 (m, 1H), 2.55-2.42 (m, 1H), 2.30-2.17 (m, 1H), 2.12-2.01 (m, 1H), 1.89-1.76 (m, 1H), 1.53 (s, 9H), 0.89-0.78 (m, 2H), 0.60-0.49 (m, 2H).
Step 7. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((5,5-difluoro-1-(4-methoxybenzyl)-6-oxopiperidin-3-yl) oxy)-2-fluorobenzoate
##STR01109##
[1904] To a solution of diisopropyl amine (0.10 mL, 0.75 mmol) in anhydrous tetrahydrofuran (1 mL) under nitrogen was added butyl lithium solution (1.5M solution in hexanes, 0.46 mL, 0.69 mmol) at -78.degree. C. After stirring at -78.degree. C. for 1 hour, a solution of (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(4-methoxybenzyl)-6-oxopiperidin-3-yl) oxy)benzoate (0.25 g, 0.53 mmol) in tetrahydrofuran (1.5 mL) was added, stirring was continued at -78.degree. C. for another 1 hour. To this reaction mixture, was added a solution of N-fluorobenzenesulfonimide (0.39 g, 0.75 mmol) in tetrahydrofuran (2 mL) slowly over 10 minutes. The reaction mixture was stirred for 30 minutes at -78.degree. C. and then slowly warmed up to -30.degree. C. over 1 hour. The reaction mixture was cooled to -78.degree. C. again and lithium bis(trimethylsilyl)amide (1.0 M solution in toluene, 0.8 mL, 0.8 mmol) was added. The reaction mixture was stirred for 30 min at -78.degree. C., a solution of N-fluorobenzenesulfonimide (0.39 g, 0.75 mmol) in tetrahydrofuran (2 mL) was added and the reaction mixture was slowly warmed up to -30.degree. C. over 1 hour and then quenched with a saturated aqueous solution of ammonium chloride solution (10 mL), extracted with ethyl acetate (2.times.15 mL) and concentrated. The residue was purified by column chromatography eluting with a gradient of 30% to 80% ethyl acetate in hexanes to afford the title compound (0.05 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.92 (d, J=7.0 Hz, 1H), 7.62-7.45 (m, 2H), 7.22-7.12 (m, 2H), 6.77 (d, J=8.6 Hz, 2H), 6.40-6.16 (m, 1H), 4.94-4.82 (m, 1H), 4.76-4.64 (m, 1H), 4.47-4.28 (m, 1H), 3.75 (s, 3H), 3.70-3.54 (m, 2H), 2.72 (dt, J=14.6, 4.8 Hz, 1H), 1.88-1.59 (m, 2H), 1.54 (s, 9H), 0.90-0.72 (m, 2H), 0.61-0.47 (m, 2H); MS(ES+) m/z 450.1 (M-t-Bu+1).
Step 8. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((5,5-difluoro-1-(4-methoxybenzyl)piperidin-3-yl) oxy)-2-fluorobenzoate
##STR01110##
[1906] To a solution of (R)-tert-butyl 5-cyclopropyl-4-((5,5-difluoro-1-(4-methoxybenzyl) -6-oxopiperidin-3-yl)oxy)-2-fluorobenzoate (0.05 g, 0.10 mmol) in tetrahydrofuran (1 mL), borane (1.0M in tetrahydrofuran, 0.3 mL, 0.3 mmol,) was added. The reaction mixture was stirred for 2 hours at ambient temperature and then quenched with methanol (1 mL). The reaction mixture was concentrated and the residue was purified by column chromatography eluting with a gradient of 0% to 20% ethyl acetate in hexanes to afford the title compound as an oil (0.03 g, 61%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.35 (d, J=8.3 Hz, 1H), 7.21 (d, J=8.5 Hz, 2H), 6.85 (d, J=8.5 Hz, 2H), 6.52 (d, J=12.4 Hz, 1H), 4.64-4.49 (m, 1H), 3.78 (s, 3H), 3.68 (d, J=13.1 Hz, 1H), 3.61 (d, J=13.1 Hz, 1H), 3.20-3.09 (m, 1H), 3.08-2.92 (m, 1H), 2.68-2.22 (m, 3H), 2.01-1.89 (m, 2H), 1.55 (s, 9H), 0.85-0.78 (m, 2H), 0.65-0.55 (m, 2H); MS(ES+) m/z 492.1 (M+1).
Step 9. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((5,5-difluoropiperidin-3-yl) oxy)-2-fluorobenzoate trifluoroacetate
##STR01111##
[1908] To a stirred solution of 4(R)-tert-butyl 5-cyclopropyl-4-((5,5-difluoro-1-(4-methoxybenzyl)piperidin-3-yl) oxy)-2-fluorobenzoate (0.23 g, 0.47 mmol)) in degassed ethyl acetate (5 mL), and trifluoroacetic acid (0.2 mL) was added 10% palladium on carbon (20 mg) and the flask was put under 1 atm of hydrogen gas. The reaction mixture was stirred for 4 hours at ambient tendperature, and then filtered over diatomaceous earth and rinsed with ethyl acetate (2.times.20 mL). The filtrate was concentrated to afford the title compound (0.23 g, quant. yield): MS(ES+) m/z 372.2 (M+1).
Step 10. Preparation of (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid hydrochioric acid
##STR01112##
[1910] To a stirred solution of (R)-tert-butyl 5-cyclopropyl-4-(5,5-difluoropiperidin-3-yl) oxy)-2-fluorobenzoate trifluoroacetate (0.23 g, 0.47 mmol) in dichloromethane (1 mL) was added trifluoroacetic acid (1 mL). After stirring at ambient temperature for 2 hours, the reaction mixture was concentrated in vacuo. The residue was dissolved in tetrahydrofuran (1 mL), and to the solution was added 2-chloro-4-fluorobenzaldehyde (0.07 g, 0.50 mL) and sodium triacetoxyborohydride (0.24 g, 0.74 mmol). The reaction mixture was stirred for 16 hours at ambient temperature, quenched with 1N aqueous hydrochloric acid solution (5 mL) and extracted with ethyl acetate (3.times.10 mL), and concentrated in vacuo. The residue was purified by column chromatography eluting with 40% ethyl acetate (plus 0.5% trifluoroacetic acid) in hexanes to afford the title compound as a colorless solid (0.16 g, 81%): .sup.1H NMR (300 MHz, CDDl.sub.3) .delta. 7.55-7.41 (m, 2H), 7.10 (dd, J=8.4, 2.6 Hz, 1H), 6.96 (dt, J=8.4, 2.6 Hz, 1H), 6.57 (d, J=12.5 Hz, 1H), 6.47-6.12 (brs, 2H), 4.63 (m, 1H), 3.77 (s, 2H), 3.13 (dd, J=11.4, 3.1 Hz, 1H), 3.02 (dd, J=20.3, 9.1 Hz, 1H), 2.72-2.48 (m, 3H), 2.15-2.05 (m, 1H), 2.01-1.96 (m, 1H), 0.88-0.79 (m, 2H), 0.66-0.58 (m, 2H).
Step 11. Preparation of (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01113##
[1912] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy-5-cyclopropyl-2-fluorobenzoic acid hydrochloric acid, the title compound was obtained as a colorless solid (0.02 g, 41%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 8.68 (d, J=15.8 Hz, 1H), 7.56 (d, J=9.0 Hz, 1H), 7.46 (dd, J=8.5, 6.2 Hz, 1H), 7.11 (dd, J=8.4, 2.5 Hz, 1H), 6.96 (dt, J=8.3, 2.5 Hz, 1H), 6.57 (d, J=14.1 Hz, 1H), 4.71-4.57 (m, 1H), 3.77 (s, 2H), 3.39 (s, 3H), 3.18-2.94 (m, 2H), 2.75-2.49 (m, 3H), 2.19-1.98 (m, 2H), 0.98-0.83 (m, 2H), 0.68-0.59 (m, 2H); MS(ES+) m/z 537.2, 535.2 (M+1).
Example 446
Synthesis of (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01114##
[1913] Step 1. Preparation of N-(2-chloro-4-fluorobenzyl)-5-oxotetrahydrofuran-2-carboxamide
##STR01115##
[1915] A mixture of (R)-5-oxotetrahydrofuran-2-carboxylic acid (10.0 g, 76.9 mmol) and thionyl chloride (20.0 g, 307.0 mmol) was refluxed for 10 hours, and then the excess thionyl chloride was removed under reduced pressure. The residue was redissolved in anhydrous dichloromethiane (150 mL) and cooled to 0.degree. C. To this solution was added triethylamine (10.1 g, 100.0 mmol) and (2-chloro-4-fluorophenyl)methanamine (12.2 g, 76.9 mmol) successively. After stirring at 0.degree. C. for 3 hours, the reaction mixture was directly poured on a silica column and eluted with 20% to 70% ethyl acetate in hexanes to afford the title compound as a colorless solid (20.80 g, quant. yield): .sup.1H NMR (300MHz, CDCl.sub.3) .delta. 7.34 (dd, J=8.5, 6.0 Hz, 1H), 7.13 (dd, J=8.4, 2.6 Hz, 1H), 6.95 (dt, J=8.3, 2.6 Hz, 1H), 6.82-6.68 (m, 1H), 4.86 (t, J=7.4 Hz, 1H), 4.51 (d, J=6.0 Hz, 2H), 2.72-2.50 (m, 3H), 2.41-2.28 (m, 1H).
Step 2. Preparation of (R)-3-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)piperidin- e-2,6-dione
##STR01116##
[1917] To a Solution of N-(2-chloro-4-fluorobenzyl)-5-oxotetrahydrofuran-2-carboxamide (20.8 g, 77.0 mmol) in anhydrous tetrahydrofuran (200 mL) was added a cooled 1.0 M solution of potassium tert-butoxide in anhydrous tetrahydrofuran (46.0 mL, 46.0 mmol) at -78.degree. C. under nitrogen atmosphere. After 10 minutes of stirring at -78.degree. C., the temperature was allowed to arise to -60.degree. C. over 10 min, and then the reaction mixture was stirred at -60.degree. C. for 1.5 hours. Another portion of potassium tert-butoxide (46.0 mL, 46.0 mmol) was added and the reaction mixture was stirred for another 1.5 hours at -45.degree. C. and then a solution of tert-butyldimethylsilyl chloride (12.80 g, 85.70 mmol) in tetrahydrofuran (20 mL) was added dropwise and stirring was continued for 30 minutes at -45.degree. C. The reaction mixture was quenched with saturated aqueous ammonium chloride solution (25 mL) at -45.degree. C., and extracted with ethyl acetate (2.times.100 mL). The combined extracts were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of 0 to 15% ethyl acetate in hexanes to give the title compound (28.1 g, 93%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.03 (dd, J=8.4, 2.4 Hz, 1H), 6.89 (d, J=8.5, 6.1 Hz, 1H), 6.79 (dt, J=8.5, 2.4 Hz, 1H), 4.94 (d, J=15.7 Hz, 1H), 4.88 (d, J=15.7 Hz, 1H), 4.35 (dd, J=6.3, 3.7 Hz, 1H), 2.94 (ddd, J=17.8, 8.9, 5.7 Hz, 1H), 2.62 (td, J =17.8, 5.7 Hz, 1H), 2.13-1.97 (m, 2H), 0.85 (s, 9H), 0.08 (s, 3H), 0.05 (s, 3H).
Step 3. Preparation of (5R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)-6-hydro- xypiperidin-2-one
##STR01117##
[1919] Following the procedure as described in Example 445 step 2 and step 3, and making variation as required to replace (R)-3-((tert-butyldimethylsilyl)oxy)-1-(4-methoxybenzyl)piperidine-2,6-di- one with (R)-3-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)p- iperidine-2,6-dione, the title compound was obtained (0.68 g, 79% in 2 steps) and used without further purification: MS(ES+) m/z 390.1, 388.1 (M+1).
Step 4. Preparation of (R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)piperidin- -2-one
##STR01118##
[1921] To a cooled solution (-78.degree. C.) of (5R)-5-((tert-butyldimethylsilyl)oxy)-6-hydroxy-1-(4-methoxybenzyl)piperi- din-2-one (2.70 g, 7.60 mmol) and triethylsilane (11.50 g, 77.10 mmol) in anhydrous dichloromethane (20 mL) under nitrogen atmosphere was added boron trifluoride diethyl ether complex (2.9 mL, 23 mmol). The reaction mixture was stirred for 30 minutes at -78.degree. C., and then warmed up slowly to 0.degree. C. and stirred at this temperature for 2 hours. The reaction mixture was quenched with a saturated aqueous solution of sodium bicarbonate (5 mL) and extracted with dichloromethane (2.times.10 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to give the title compound as an oil (1.26 g, 47%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.16 (d, J=8.5 Hz, 2H), 6.82 (d, J=8.6 Hz, 2H), 4.71 (d, J=14.6 Hz, 1H), 4.27 (d, J=14.6 Hz, 1H), 4.08-3.98 (m, 1H), 3.77 (s, 3H), 3.24 (dd, J=12.4, 3.7 Hz, 1H), 3.05 (dd, J=12.3, 4.5 Hz, 1H), 2.68 (m, 1H), 2.39 (td, J=17.6, 5.7 Hz, 1H), 1.91-1.78 (m, 2H), 0.81 (s, 9H), 0.00 (s, 3H), -0.06 (s, 3H).
Step 4. Preparation of (5R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)-3-fluor- opiperidin-2-one
##STR01119##
[1923] To a stirred solution of diisopropylamine (0.068 g, 0.67 mmol) in tetrahydrofuran (2 mL) at -78.degree. C. was added n-butyllithium (1.5 M solution in hexanes, 0.41 mL, 0.62 mmol). After stirring for 45 minutes at -78.degree. C., a solution of (R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)piperidin- -2-one (0.18 g, 0.48 mmol) tetrahydrofuran (2 mL) was added dropwise and the reaction mixture was stirred for 40 minutes at -78.degree. C. A solution of N-fluorobenzenesulfonimide (0.21 g, 0.67 mmol) in tetrahydrofuran (1.5 mL) was added via syringe pump over a 10 minutes period and the reaction mixture was stirred at -78.degree. C. for 5 hours and then slowly warmed up to -40.degree. C., The reaction mixture was quenched by addition of saturated aqueous solution of ammonium chloride (10 mL), and extracted with ethyl acetate (2.times.10 mL). The combined organic phases were washed with water (10 mL) and brine (10 mL), dried over sodium sulfate, filtered and concentrated invacuo. To the residue was added dichloromethane and methyl tert-butylether (10 mL, 1/1, v/v) the resulting precipitate was removed by filtration. The filtrate was concentrated to give a light yellow residue which was purified by silica gel chromatography using a gradient (0 to 30%) of ethyl acetate in hexanes to give the title compound as a gum (0.08 g, 39%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.29 (dd, J =8.6, 6.1 Hz, 1H), 7.10 (dd, J=8.4, 2.6 Hz, 1H), 6.92 (dt, J=8.4, 2.6 Hz, 1H), 5.18 (ddd, J=47.6, 10.1, 6.1 Hz, 1H), 4.99 (d, J=15.5 Hz, 1H), 4.37 (d, J=15.5 Hz, 1H), 4.29-4.20 (m, 1H), 3.44 (dd, J =12.6, 3.3 Hz, 1H), 3.18-3.09 (m, 1H), 2.51-2.35 (m, 1H), 2.23-2.07 (m, 1H), 0.83 (s, 9H), 0.06 (s, 3H), -0.03 (s, 3H).
Step 5. Preparation of (R)-5-((tert-butyldimethylsilyl) oxy)-1-(2-chloro-4-fluorobenzyl)-3,3-difluoropiperidin-2-one
##STR01120##
[1925] Following the procedure as described in Example 445 step 4, and making variation as required to replace (R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)piperidin- -2-one with (5R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)-3-fluor- opiperidin-2-one, the title compound was obtained as a colorless solid (0.075 g, 99%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.35 (dd, J =8.6, 6.1 Hz, 1H), 7.12 (dd, J=8.4, 2.6 Hz, 1H), 6.96 (dt, J=8.4, 8.3, 2.6 Hz, 1H), 4.83 (d, J=15.3 Hz, 1H), 4.56 (d, J=15.2 Hz, 1H), 4.23-4.13 (m, 1H), 3.44 (dd, J=12.4, 4.0 Hz, 1H), 3.21 (dd, J=12.5, 6.3 Hz, 1H), 2.62-2.27 (m, 2H), 0.82 (s, 9H), 0.05 (s, 3H), -0.01 (s, 3H); MS(ES+) m/z 410.2, 408.2 (M+1).
Step 6. Preparation of (R)-1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-ol
##STR01121##
[1927] To a solution of (R)-5-((tert-butyldimethylsilyl)oxy)-1-(2-chloro-4-fluorobenzyl)-3,3-difl- uorpiperidin-2-one (0.015 g, 0.04 mmol) in tetrahydrofuran (1 mL) was added borane (1.0 M solution in tetrahydrofuran 0.9 mL, 0.9 mmol) under nitrogen atmosphere. After 30 minutes stirring, the reaction mixture was quenched by addition of 6 N hydrochloric acid solution (2.5 mL) and stirring was continued for 2.5 hours. The solution was neutralized by addition of a saturated aqueous solution of sodium bicarbonate and extracted with dichloromethane (2.times.10 mL). The combined organic extracts were concentrated under reduced pressure and filtered over a plug of silica gel eluting with 10% ethyl acetate in hexanes followed by 100% ethyl acetate to give the title compound as a colorless oil (0.01 g, 85%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.38 (dd, J=8.5, 6.2 Hz, 1H), 7.11 (dd, J=8.5, 2.6 Hz, 1H), 6.97 (dt, J=8.3, 2.6 Hz, 1H), 4.05-3.96 (m, 1H), 3.72-3.67 (m, 2H), 2.98-2.83 (m, 1H), 2.82-2.71 (m, 1H), 2.60-2.45 (m, 2H), 2.24-1.90 (m, 2H); MS(ES+) m/z 282.1, 280.1 (M+1).
Step 7. Preparation of (R)-tert-butyl 5-chloro-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl)oxy)-- 2-fluorobenzoate
##STR01122##
[1929] Following the procedure as described in Example 445 step 4 and making variation as required to replace (R)-5-hydrozyl-1-(4-methoxybenzyl)piperidin-2-one with (R)-1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-ol, the title compound was obtained as a colorless solid (0.01 g, 67%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.87 (d, J=7.7 Hz, 1H), 7.47 (dd, J=8.6, 6.3 Hz, 1), 7.10 (dd, J=8.5, 2.6 Hz, 1H), 6.97 (dt, J=8.3, 8.3, 2.6 Hz, 1H), 6.64 (d, J=11.8 Hz, 1H), 4.65-4.51 (m, 1H), 3.75 (s, 2H), 3.19-2.96 (m, 2H), 2.69-2.42 (m, 3H), 2.12-2.03 (m, 1H), 1.56 (s, 9H); MS(ES+) m/z 510.0, 508.0 (M +1).
Step 8. Preparation of (R)-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01123##
[1931] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with with (R)-tert-butyl 5-chloro-4-((1-(2-chloro-4-fluorobenzyl)-5,5-difluoropiperidin-3-yl) oxy)-2-fluorobenzoate, the title compound was obtained as a solid (0.004 g, 32%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.77 (d, J=7.4 Hz, 1H), 7.63 (dd, J=8.6, 6.3 Hz, 1H), 7.19 (dd, J=8.7, 2.6 Hz, 1H), 7.09-6.97 (m, 2H), 4.84-4.75 (m, 1H), 3.86-3.79 (m, 2H), 3.33 (s, 3H), 3.09-2.79 (m, 3H), 2.77-2.64 (m, 1H), 2.59-2.41 (m, 1H), 2.33-2.14 (m, 1H); MS(ES+) m/z 531.1, 529.1 (M+1).
Example 447
Synthesis of 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-methoxyethyl)piperi- din-3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01124##
[1932] Step 1. Preparation of 2-((R)-3-(2-cyclopropyl-5-fluoro-4-(methoxycarbonyl) phenoxy)piperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid
##STR01125##
[1934] To a stirred solution of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (0.59 g, 2.00 mmol) in toluene (8 mL) under an atmosphere of nitrogen were added 50% glyoxylic acid in water (0.24 g, 3.20 mmol), molecular sieve 4 .ANG. (0.5 g) and (3,5-dichlorophenyl)boronic acid (0.420 g, 2.20 mmol). The reaction mixture was stirred at 100.degree. C. for 2 hours, cooled to ambient temperature and filtered to remove the molecular sieve. The filtrate was concentrated. The residue (0.15 g, 15%) was used directly in the next step without further purification.
Step 2. Preparation of methyl 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-3-yl)(oxy)-2-fluorobenzoate
##STR01126##
[1936] To a stirred solution of 2-((R)-3-(2-cyclopropyl-5-fluoro-4-(methoxycarbonyl) phenoxy)piperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid (0.15 g, 0.30 mmol) in tetrahydrofuran (3 mL) under an atmosphere of nitrogen was added borate in tetrahydrofuran (1.0 M solution in tetrahydrofuran, 1 mL, 1.00 mmol). After stirring at ambient temperature for 16 hours, the reaction mixture was quenched by addition of methanol (2 mL), and concentrated. The residue was purified by chromatography eluting with a gradient (0 to 30%) of ethyl acetate in hexanes to give the title compound (0.09 g, 55%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.43 (d, J=8.4 Hz, 1H), 7.29 (t, J=1.8 Hz, 1H), 7.06 (d, J=1.9 Hz, 2H), 6.54 (d, J=12.8 Hz, 1H), 4.50-4.39 (m, 1H), 3.93-3.82 (m, 4H), 3.69-3.58 (m, 2H), 2.72-2.57 (m, 3H), 2.40-2.27 (m, 1H), 2.01-1.49 (m, 5H), 0.87-0.78 (m, 2H), 0.68-0.57 (m, 2H). The other diastereoisomer methyl 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoate was also isolated (0.07 g, 45%). .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.42 (d, J=8.4 Hz, 1H), 7.33-7.29 (m, 1H), 7.10-7.07 (m, 2H), 6.52 (d, J=12.7 Hz, 1H), 4.48-4.35 (m, 1H), 3.96-3.82 (m, 4H), 3.73-3.58 (m, 2H), 2.97-2.88 (m, 1H), 2.61-2.39 (m, 3H), 1.94-1.53 (m, 5H), 0.85-0.79 (m, 2H), 0.67-0.56 (m, 2H).
Step 3. Preparation of methyl 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoate
##STR01127##
[1938] To a stirred solution of methyl 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoate (0.08 g, 0.14 mmol) in dimethoxyethane (1 mL) under an atmosphere of nitrogen was added sodium hydride (60% in mineral oil, 0.03 g, 0.69 mmol) and the reaction mixture was stirred for 30 minutes. Methyl iodide (0.030 g, 0.21 mmol) was added to the reaction mixture. After stirring for 16 hours, water (10 mL) was added and the reaction mixture was extracted with dichloromethane (2.times.10 mL), the combined organic layers were concentrated. The residue was purified by chromatography eluting with a gradient of methanol in dichloromethane (0 to 5%) to give the title compound (0.04g, 58%): MS(ES+) m/z 498.1, 496.1 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoic acid hydrochloride
##STR01128##
[1940] To a stirred solution of methyl 5-cyclopropyl-4-(((3R)-1-(1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoate (0.075 g, 0.15 mmol) in DMSO (1 mL) under an atmosphere of nitrogen were added lithium hydroxide (0.036 g, 1.52 mmol) and the mixture was stirred for 1 hour at 70.degree. C. and then quenched with 1N aqueous hydrochloric acid solution (5 mL) and extracted with dichloromethane (3.times.10 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the title compound (0.06 g, 76%) as a colorless solid: MS(ES+) m/z 484.2, 482.2 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(((R)-1-((R)-1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01129##
[1942] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-fluoropiperidin-- 4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 5-cyclopropyl-4-(((3R)-1-(1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluorobenzoic acid hydrochloride (0.06 g, 0.12 mmol), the title compound was obtained as a colorless solid (0.006 g, 9%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.86-8.62 (m, 1H), 7.56 (d, J=8.9 Hz, 1H), 7.52-7.42 (m, 2H), 6.94-6.83 (m, 1H), 5.06-4.92 (m, 1H), 4.48-4.36 (m, 1H), 4.48-4.36 (m, 1H), 4.20-4.08 (m, 1H), 4.06-3.34 (m, 1H), 2.94-2.73 (m, 1H), 2.33-1.94 (m, 3H), 1.77-1.58 (m, 1H), 0.96-0.86 (m, 2H), 0.69-0.59 (m, 2H); MS(ES+) m/z 561.1, 559.1 (M+1).
Example 448
Synthesis of 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01130##
[1943] Step 1. Preparation of 2-((R)-3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)piperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid
##STR01131##
[1945] To a stirred solution of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-3-yloxy)benzam- ide (1.50 g, 3.31 mmol) in toluene (10 mL) under an atmosphene of nitrogen was introduced 50% glyoxylic acid in water (0.39 g, 5.30 mmol), molecular sieve 4 .ANG. (0.5 g) and (3,5-dichlorophenyl)boronic acid (0.69 g, 3.64 mmol). The reaction mixture was stirred at 100.degree. C. for 2 hours, cooled to ambient temperature and filtered to remove the molecular sieve. The filtrate was concentrated to give the title compound as a colorless solid (2.16 g, 99%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.90-11.82 (m, 1H), 7.70-7.58 (m, 1H), 7.52-7.42 (m, 2H), 7.10 (t, J=7.9 Hz, 1H), 6.98 (dd, J=13.0, 6.0 Hz, 1H), 4.78-4.56 (m, 2H), 3.30 (s, 3H), 3.04-2.92 (m, 1H), 2.90-2.77 (m, 1H), 2.76-2.63 (m, 1H), 2.62-2.51 (m, 1H), 2.19-1.97 (m, 1H), 1.98-1.77 (m, 2H), 1.74-1.47 (m, 2H), 0.95-0.78 (m, 2H), 0.76-0.58 (m, 2H); MS(ES+) m/z 561.0, 559.0 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((3R)-1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl) piperidin-3-yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01132##
[1947] To a stirred solution of 2-((R)-3-(2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl) phenoxy)piperidin-1-yl)-2-(3,5-dichlorophenyl)acetic acid (0.80 g, 1.43 mmol) in tetrahydrofuran (8 mL) was added carbonyl diimidazole (0.26 g, 1.57 mmol) under nitrogen. The reaction mixture was reflux for 1 hour and cooled to ambient temperature. To the reaction mixture was added sodium borohydride (0.064 g, 2.0 mmol). After stirring for 2 hours, the reaction mixture was quenched by addition of 1N aqueous hydrochloric acid solution (10 mL) and extracted with dichloromethane (3.times.20 mL). The combined organics was concentrated, the residue was purified by chromatography eluting with a gradient of 5 to 10% dichloromethane in methanol containing 2% ammonia to afford the title compound as a colorless solid (0.13 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 7.47-7.40 (m, 1H), 7.38-7.83 (m, 2H), 7.18-7.09 (m, 1H), 6.86-6.67 (m, 1H), 4.64-4.53 (m, 1H), 4.51-4.37 (m, 1H), 3.80-3.59 (m, 2H), 3.58-3.48 (m, 1H), 3.06-2.57 (m, 5H), 2.38-2.11 (m, 2H), 2.07-1.93 (m, 1H), 1.93-1.77 (m, 1H), 1.76-1.61 (m, 1H), 1.59-1.29 (m, 2H), 0.90-0.76 (m, 2H), 0.65-0.48 (m, 2H); MS(ES+) m/z 547.1, 545.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-(((R)-1-((S)-1-(3,5-dichlorophenyl)-2-methoxyethyl) piperidin-3-yl)oxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01133##
[1949] To a solution of 5-cyclopropyl-4-(((3R)-1-(1-(3,5-dichlorophenyl)-2-hydroxyethyl)piperidin- -3-yl) oxy)-2-fluoro-N-(methylsulfonyl)benzamide (0.08 g, 0.12 mmol) in dimethoxyethane (1 mL) was added sodium hydride (60% in mineral oil, 0.025 g, 0.62 mmol). The reaction mixture was stirred for 30 minutes and methyl iodide (0.026 g, 0.19 mmol) was added. After stirring for 16 hours, the reaction mixture was quenched by addition of water (10 mL) and extracted with dichloromethane (2.times.10 mL). The combined organic layers was concentrated and the residue purified by preparative HPLC eluting with a gradient of acetonitrile in water containing 0.1% trifluoroacetic acid to afford the title compound as a colorless solid (0.011 g, 14%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.82-8.64 (m, 1H), 7.55 (d, J=8.8 Hz, 1H), 7.47-7.38 (m, 3H), 6.88 (d, J=14.4 Hz, 1H), 4.92-4.81 (m, 1H), 4.26-4.19 (m, 1H), 4.12-4.02 (m, 1H), 3.92-3.79 (m, 2H), 3.39 (s, 3H), 3.36 (s, 3H), 3.35-3.28 (m, 1H), 2.79-2.65 (m, 2H), 2.28-2.18 (m, 1), 2.09-1.96 (m, 2H), 1.71-1.56 (m, 2H), 0.96-0.87 (m, 2H), 0.67-0.59 (m, 2H); MS(ES+) m/z 561.1, 559.1 (M+1).
Example 449
Synthesis of 5-cyclopropyl-4-(((1R,3R,5S)-8-(3,5-dichlorobenzyl)-8-azabicyclo[3.2.1]oc- tan-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01134##
[1950] Step 1. Preparation of benzyl 3-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01135##
[1952] Following the procedure as described in Example 346 Step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)benzyl)-4-fluoropiperidin-4-y- l) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((8-((benzyloxy)carbonyl)-8-azabicyclo[3.2.1]octan-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.75 g, 72%): MS(ES+) m/z 531.1 (M+1).
Step 2. Preparation of 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-2-fluoro-N-(methyl- sulfonyl)benzamide
##STR01136##
[1954] Following the procedure as described in Example 396 step 5 and making non-critical variations as required to replace (1R, 3S,5S)-benzyl-3-((2-cyclopropyl-4-((cyclopropylsulfonyl)carbamoyl)-5-fluo- rophenoxy) methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate with (1 R,3,5S)-benzyl 3-((2-cyclopropyl-5-fluoro-4-(methylsulfonyl) carbamoyl)phenoxy)methyl)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained (0.55 g, 98%): MS(ES+) m/z 367.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-(((1B,3r,5S)-8-(3,5-dichlorobenzyl)-8-azabicyclo[3.2.1]oc- tan-3-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01137##
[1956] Following the procedure as described in Example 441, and making variation as required to replace 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-N-(azetidin-1- ylsulfonyl)-5-cyclopropyl-2-fluorobenzamideate with 4-(8-azabicyclo[3.2.1]octan-3-ylmethoxy)-5-cyclopropyl-2-fluoro-N-(methyl- sulfonyl)benzamide and to replace (bromomethylene)dibenzene with 1,3-dichloro-5-(chloromethyl)benzene, the title compound was obtained (0.04 g, 41%). .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 7.67-7.44 (m, 3H), 7.14 (d, J=8.4 Hz, 1H), 6.79 (d, J=12.9 Hz, 1H), 3.92-3.73 (m, 4H) 3.54-3.36 (m, 2H), 3.05 (s, 3H), 2.33-1.55 (m, 10H), 0.91-0.77 (m, 2H), 0.63-0.53 (m, 2H); MS(ES+) m/z 557.1, 555.1 (M+1).
Example 450
Synthesis of 4-(((1R,4S,6R)-2-((S)-1-(2-chloro-4-fluorophenly)ethyl)-2-azabicyclo[2.2.- 1]heptan-6-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01138##
[1957] Step 1. Preparation of tert-butyl 5-chloro-2-fluoro-4-(((1R,4S,6R)-2-((S)-1-phenlyethyl)-2-azabicyclo[2.2.1- ]heptan-6-yl)oxy)benzoate
##STR01139##
[1959] Following the procedure as described in Example 445 step 4 and making variation as required to replace (R)-5-hydroxy-1-(4-methoxybenzyl)piperidin-2-one with (1R,4S,6R)-2-((S)-1-phenylethyl)-2-azabicyclo[2.2.1]heptan-6-ol, and purifying the compound by column chromatognaphy eluting with a gradient of ethyl acetate in hexanes (50% to 100%), the title compound was obtained (2.76 g, 49%). MS(ES+) m/z: 448.1, 446.1 (M+1).
Step 2. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,4S,6R)-2-((S)-1-phenylethyl)-2-azabicyclo[- 2.2.1]heptan-6-yl)oxy)benzoate
##STR01140##
[1961] Following the procedure as described in Example 445 step 5, and making variation as required to replace (R)-tert-butyl 5-chloro-2-fluoro-4-((1-(4-methoxybenzyl)-6-oxopiperidin-3-yl)oxy)benzoat- e with tert-butyl 5-chloro-2-fluoro-4-(((1R,4S,6R)-2-((S)-1-phenylethyl)-2-azabicyclo[2.2.1- ]heptan-6-yl) oxy)benzoate, the title compound was obtained as a colorless gum (1.82 g, 66%): MS(ES+) m/z 452.2 (M+1).
Step 3. Preparation of tert-butyl 4-((1R,4S,6R)-2-azabicyclo[2.2.1]heptan-6-yloxy)-5-cyclopropyl-2-fluorobe- nzoate
##STR01141##
[1963] To a stirred solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((1R,4S,6R)-2-((S)-1-phenylethyl)-2-azabicyclo[2- .2.1]heptan-6-yl) oxy)benzoate (1.75 g, 3.88 mmol) and ammonium formate (7.3 g, 116 mmol) in methanol (50 mL) and water (5 mL) was added 10% palladium on carbon (0.05 g). After stirring for 2 hours at reflux, the reaction mixture was filtered over diatomaceous earth and rinsed with methanol (2.times.30 mL). The filtrate was concentrated, basified with saturated sodium bicarbonate solution and extracted with dichloromethane (2.times.40 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered and concentrated to afford the title compound (1.22 g, 80%): MS(ES+) m/z 348.2 (M+1).
Step 4. Preparation of tert-butyl 4-(((1R,4S,6R)-2-((S)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluorobenzoate
##STR01142##
[1964] And tert-butyl 4-(((1R,4S,6R)-2-((R)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluorobenzoate
##STR01143##
[1966] To a stirred solution of tert-butyl 4-((1R,4S,6R)-2-azabicyclo[2.2.1]heptan-6-yloxy)-5-cyclopropyl-2-fluorobe- nzoate (1.22 g, 3.51 mmol) in acetonitrile (15 mL) under a nitrogen atmosphere was added 2-chloro-1-(1-chloroethyl)-4-fluorobenzene (0.87 g, 4.55 mmol), potassium carbonate (1.38 g, 10 mmol) and potassium iodide (0.165 g, 1 mmol). After stirring at reflux for 16 hours, the reaction mixture was was cooled to ambient temperature, quenched with water (15 mL) and extracted with ethyl acetate (3.times.20 mL). The combined organic layers was concentrated and the residue was purified by column chromatography (0% to 30% ethyl acetate in hexanes) to give the title compound (0.28 g, 16%). MS(ES+) m/z 504.1 (M+1); its diastereoisomer tert-butyl 4-(((1R,4S,6R)-4-fluorophenyl)ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy)-- 5-cyclopropyl-2-fluorobenzoate was also isolated (0.25 g, 14%): MS(ES+) m/z 504.1 (M+1).
Step 5. Preparation of 4-(((1R,4S,6R)-2-((S)-1-(2-Chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluoro-N-(methy- lsulfonyl)benzamide, trifluoroacetic acid salt
##STR01144##
[1968] To a stirred solution of tert-butyl 4-(((1R,4S,6R)-2-((S)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy)-5-cyclopropyl-2-fluorobenzoate (0.13 g, 0.26 mmol) in dichloromethane (2 ml) was added trifluoroacetic acid (1 mL). After stirring at ambient temperature for 30 minutes, the reaction mixture was concentrated and co-concentrated with toluene (2.times.10 mL). The residue was dissolved in dichloromethane (0.5 mL), and to this solution was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (0.11 g, 0.44 mmol) 4-dimethylaminopyridine (0.09 g, 0.78 mmol) and methanesulfonamide (0.03 g, 0.34 mmol). After stirring at at ambient temperature for 16 hours, the reaction mixture was quenched with aqueous hydrochloric acid solution (6N, 0.2 mL), diluted with acetonitrile (0.5 mL) and purified by preparative HPLC (gradient of acetonitrile in water +0.1% trifluoroacetic acid) to afford the title compound as a colorless solid (0.06 g, 71%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.30-11.64 (br s, 1H), 11.36-10.08 (br s, 1H), 7.99-7.84 (m, 1H), 7.67-7.51 (m, 1H), 7.49-7.38 (m, 1H), 7.23-6.98 (m, 1H), 6.91-6.00 (m, 1H), 5.25-4.47 (m, 2H), 3.86-2.51 (m, 7H), 2.29-2.06 (m, 1H), 2.06-1.79 (m, 3H), 1.78-1.41 (m, 4H), 0.95-0.48 (m, 4H); MS(ES+) m/z 527.1, 525.1 (M+1).
Example 451
Synthesis of 4-(((1R,4S,6R)-2-((R)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluoro-N-(methy- lsulfonyl)benzamide, trifluoroacetic acid salt
##STR01145##
[1970] Following the procedure as described in Example 450 step 5 and making variation as required to replace tert-butyl 4-(((1R,4S,6R)-2-((S)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluorobenzoate with its diastereoisomer tert-butyl 4-(((1R,4S,6R)-2-((R)-1-(2-chloro-4-fluorophenyl) ethyl)-2-azabicyclo[2.2.1]heptan-6-yl)oxy-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.04 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.12.14-11.78 (br s, 1H), 10.49-10.27 (br s, 1H), 8.15-8.00 (m, 1H), 7.64-7.53 (m, 1H), 7.52-7.39 (m, 1H), 7.11-7.03 (m, 1H), 6.36-6.22 (m, 1H), 5.01-4.80 (m, 2H), 3.64-2.64 (m, 7H), 2.30-1.99 (m, 2H), 1.96-1.78 (m, 2H), 1.75-1.46 (m, 4H), 0.87-0.72 (m, 2H), 0.65-0.53 (m, 2H); MS(ES+) m/z 527.1, 525.1. (M+1).
Example 452
Synthesis of 4-(((R)-1-(R)-1-(2-chloro-4-fluorophenyl)ethyl)-5,5-dimethylpiperidin-3-y- l) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01146##
[1971] Step 1. Preparation of (R)-tert-butyl 4-((1-benzyl-5,5-dimethylpiperidin-3-yl) oxy)-5-chloro-2-fluorobenzoate
##STR01147##
[1973] Following the procedure as described in Example 422 step 1 and making variation as required to replace (R)-1-benzylpiperidin-3-ol with (R)-1-benzyl)-5,5-dimethylpiperidin-3-ol (prepared accordingly to Ma Y., Lahue B. R. et al.; U.S. patent 2008/0004287 A1), the title compound waa obtained as a colorless solid (0.73 g, 68%): MS(ES+) m/z 450.2, 448.2 (M+1).
Step 2. Preparation of (R)-tert-butyl 4-((1-benzyl-5,5dimethylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoat- e
##STR01148##
[1975] Following the procedure as described in Example 422 step 2 and making variation as required to replace (R)-tert-butyl 4-((1-benzylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate with (R)-tert-butyl 4-((1-benzyl-5,5-dimethylpiperidin-3-yl)oxy)-5-chloro-2-fluorobenzoate, the title compound was obtained as oil (0.47 g, 63%): MS(ES+) m/z 454.1 (M+1).
Step 3. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((5,5-dimethylpiperidin-3-yl)oxy)-2-fluorobenzoate
##STR01149##
[1977] Following the procedure as described in Example 450 .sub.step 3, and making variation as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,4S,6R)-2-((S)-1-phenylethyl)-2-azabicyclo[- 2.2.1]heptan-6-yl) oxy)benzoate with (R)-tert-butyl 4-((1-benzyl-5,5-dimethylpiperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as an oil (0.38 g, 96%): MS(ES+) m/z 364.2 (M+1).
Step 4. Preparation of tert-butyl 4-(((3R)-1-(1-(2-fluorophenyl)ethyl)-5,5-dimethylpiperidin-3-yl) oxy-5-cyclopropyl-2-fluorobenzoate
##STR01150##
[1979] Following the procedUre as described in Example 450 step 4, and making variation as required to replace tert-butyl 4-((1R,4S,6R)-2-azabicyclo[2.2.1]heptan-6-yloxy)-5-cyclopropyl-2-fluorobe- nzoate with tert-butyl 5-cyclopropyl-4-((5,5-dimethylpiperidin-3-yl)oxy)-2-fluorobenzoate, the title compound was obtained as an oil (0.22 g, 43%): MS(ES+) m/z 522.1, 520.1 (M+1).
Step 5. Preparation of 4-(((R)-1-((R)-1-(2-Chloro-4-fluorophenyl)ethyl)-5,5-dimethylpiperidin-3-- yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01151##
[1981] Following the procedure as described in Example 450 step 5, and making variation as required to replace tert-butyl 4-(((1R,4S,6R)-2-((S)-1-(2-chloro-4-fluorophenyl)ethyl)-2-azabicyclo[2.2.- 1]heptan-6-yl) oxy)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 4-(((3R)-1-(1-(2-chloro-4-fluorophenyl) ethyl)-5,5-dimethylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.02 g, 15%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.7.61 (dd, J=8.7, 6.5 Hz, 1H), 7.35 (dd, J=8.9, 2.6 Hz, 1H), 7.25-7.02 (m, 3H), 6.65-6.50 (m, 2H), 4.56-4.39 (m, 1H), 3.87 (q, J=6.3 Hz, 1H), 3.06-2.74 (m, 4H), 2.39 (d, J=10.7 Hz, 1H), 2.19-2.01 (m, 1H), 2.00-1.85 (m, 2H), 1.80-1.67 (m, 1H), 1.32-1.17 (m, 4H), 0.97 (s, 3), 0.93 (s, 3H), 0.86-0.73 (m, 2H), 0.60-0.44 (m, 2H); MS(ES+) m/z 543.1, 541.1 (M+1).
Example 453
Synthesis of 5-cyclopropyl-4-((1-(2,4-dichloro-5-fluorobenzyl)piperidin-4-yl)methoxy)-- 2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01152##
[1982] Step 1. Preparation of 4-((1-benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfo- nyl)benzamide
##STR01153##
[1984] To a solution of tert-butyl 4-((1-benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate (1.55 g, 3.54 mmol) in dichlormethane (10 mL) was added trifluoroacetic acid (5 mL). The reaction mixture was stirred for 1.5 h and the solvent was concentration invacuo. The residue was redissolved in anhydrous dichloromethane (10 mL). To this solution, was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (1.37 g, 5.30 mmol), 4-dimethylaminopyridine (1.08 g, 8.84 mmol) and methanesulfonamide (0.37 g, 3.89 mmol). The reaction mixture was stirred at ambient tenperature for 16 hours, and then diluted with dichloromethane (20 mL), washed with 1N aqueous hydrochloric acid solution (10 mL), the aqueous layer was extracted with dichloromethane (20 mL). The combined organic layer dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by column chromatography (0% to 10% methanol (1% ammonia) in dichloromethane) to afford the title compound as a gum (1.32 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.55 (d, J=9.04 Hz, 1H), 7.44-7.36 (m, 5H), 6.52 (d, J=14.25 Hz, 1H), 4.16-4.02 (m, 2H), 3.94-3.86 (m, 2H), 3.64-3.43 (m, 2H), 3.39 (s, 3H), 2.68-2.43 (m, 2H), 2.09-1.87 (m, 6H), 0.97-0.85 (m, 2H), 0.65-0.55 (m, 2H).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-4-ylmethoxy) benzamide hydrochloride
##STR01154##
[1986] To a stirred solution of 4-((1-benzylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfo- nyl)benzamide (1.25 g, 2.72 mmol)) in degassed ethyl acetate (5mL) was added 10% palladium on carbon (0.02 g) and the flask was put under 1 atm of hydrogen. The reaction mixture was stirred for 24 hours at ambient temperature, then filtered over diatomaceous earth and rinsed with a 1:1 mixture of methanol and 1 M aqueous hydrochloric acid solution (2.times.20 mL). The filtrate was concentrated to give the title compound (0.55 g, 55%). MS(ES+) m/z 371.1 (M-1).
Step 3. Preparation of 5-cyclopropyl-4-((1-(2,4-dihloro-5-fluorobenzyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetate
##STR01155##
[1988] To a stirred solution of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-(piperidin-4-ylmethoxy)benzam- ide hydrochloride (0.05 g, 0.12. mmol) in acetonitrile (2 mL) under a nitrogen atmosphere was added 1,5-dichloro-2-(chloromethyl)-4-fluorobenzene (0.04 g, 0.18 mmol), potassium carbonate (0.04 g, 0.30 mmol) and potassium iodide (0.024 g, 0.15 mmol). The reaction mixture was stirred at reflux for 16 hours, cooled to ambient temperature, 1N aqueous hydrochloric acid solution (5 mL) was added and the mixture was extracted with ethyl acetate (3.times.10 mL), concentrated and purified by column chromatography (0% to 30% ethyl acetate (+1% formic acid) in hexanes) to give an oil which was further purified by preparative HPLC (gradient of acetonitrile in water +0.1% trifluoroacetic acid) to give the title compound as a colorless solid (0.04, 43%). .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.15-11.63 (m, 1H), 9.91-9.31 (m, 1H), 7.99 (d, J=6.88 Hz, 1H), 7.77 (d, J=9.55 Hz, 1H), 7.10 (d, J=8.24 Hz, 1H), 6.95 (d, J=13.00 Hz, 1H), 4.44-4.31 (m, 2H), 4.01-3.90 (m, 2H), 3.53-3.40 (m, 2H), 3.30 (s, 3H), 3.21-3.00 (m, 2H), 2.17-1.88 (m, 4H), 1.68-1.46 (m, 2H), 0.93-0.79 (m, 2H), 0.72-0.60 (m, 2H); MS(ES+) m/z: 547.1, 549.1 (M+1).
Example 454
Synthesis of 5-cyclopropyl-4-((3,5-dichloro-4-fluorobenzyl)piperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01156##
[1990] Following the procedure as described in Example 453 step 3 and making variation as required to replace 1,5-dichloro-2-(chloromethyl)-4-fluorobenzene with 1,3-dichloro-5-(chloromethyl)-2-fluorobenzene, the title compound was obtained as a colorless solid (0.03 g, 33%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.12-11.60 (m, 1H), 9.87-9.63 (m, 1H), 7.83-7.62 (m, 2H), 7.14-7.06 (m, 1H), 6.98-6.90 (m, 1H), 4.35-4.21 (m, 2H), 4.00-3.91 (m, 2H), 3.52-3.36 (m, 2H), 3.30 (s, 3H), 3.04-2.85 (m, 2H), 2.15-1.80 (m, 4H), 1.67-1.45 (m, 2H), 0.92-0.79 (m, 2H), 0.71-0.60 (m, 2H); MS(ES+) m/z 547.0, 549.0 (M+1).
Example 455
Synthesis of 4-((1-(5-chloro-2,4-difluorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01157##
[1992] Following the procedure as described in Example 453 step 3, and making variation as require to replace 1,5-dichloro-2-(chloromethyl)-4-fluorobenzene with 1-chloro-5-(chloromethyl)-2,4-difluorobenzene, the title compound was obtained as a colorless solid (0.02 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 12.06-11.74 (m, 1H), 9.65-9.36 (m, 1H), 7.87 (dd, J=7.9, 7.9 Hz, 1H), 7.71 (dd, J =9.6, 9.6 Hz, 1H), 7.10 (d, J=8.2 Hz, 1H), 6.94 (d, J=12.9 Hz, 1H), 4.35-4.22 (m, 2H), 3.99-3.91 (m, 2H), 3.50-3.42 (m, 2H), 3.30 (s, 3H), 3.11-2.93 (m, 2H), 2.13-1.88 (m, 4H), 1.62-1.43 (m, 2H), 0.92-0.80 (m, 2H), 0.70-0.61 (m, 2H); MS(ES+) m/z 533.1, 531.1 (M+1).
Example 456
Synthesis of 4-((1-(3-chloro-4,5-difluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-- 2-fluoro-N-(methysulfonnyl)benzamide, trifluoroacetic acid salt
##STR01158##
[1994] Following the procedure as described in Example 453 step 3, and making variation as required to replace 1,5-dichloro-2-(chloromethyl)-4-fluorobenzene with 1-chloro-5-(chloromethyl)-2,3-difluorobenzene, the title compound was obtained as a colorless solid (0.03 g, 31%): .sup.1H NMR (300 MHz, DMSO-d.sub.6).delta. 11.40-11.06 (m, 1H), 7.50-7.35 (m, 2H), 7.12 (d, J=8.4 Hz, 1H), 6.85 (d, J=12.9 Hz, 1H), 3.91 (d, J=6.0 Hz, 2H), 3.67-3.59 (m, 2H), 3.16 (s, 3H), 2.99-2.82 (m, 2H), 2.23-2.06 (m, 2H), 2.04-1.91 (m, 1H), 1.86-1.72 (m, 3H), 1.46-1.27 (m, 2H), 0.90-0.80 (m, 2H), 0.66-0.55 (m, 2H); MS(ES+) m/z 533.2, 531.2 (M+1).
Example 457
Synthesis of 4-((1-(5-chloro-2-trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01159##
[1995] Step 1. Preparation of tert-butyl 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01160##
[1997] Following the procedure as described in Example 346 step 5 and making non-critical variations as required to replace 1-chloro-3-(chloromethyl)-2-fluoro-5-(trifluoromethyl)benzene with 5-chloro-2-(trifluoromethyl)piperidin methanesulfonate, the title compound was obtained as a colorless gum (0.45 g, 69%): MS(ES+) m/z 562.1, 560.1 (M+1).
Step 2. Preparation of 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01161##
[1999] Following the procedure as described in Example 346 step 6, and making non-critical variations as required to replace terr-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)-4-fluor- opiperidine-1-carboxylate with tert-butyl 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate to afford the title compound as a colorless solid (0.36 g, 88%): MS(ES+) m/z 506.2, 504.0 (M+1).
Step 3. Preparation of 4-((1-(5-chloro-2-(trifluoromethyl)piperidin)-4-fluoropiperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01162##
[2001] Following the procedure as described in Example 346 step 7 and making non-critical variations as required to replace 4-((1-(3-chloro-2-fluoro-5-(4trifluoromethyl)piperidin)-4-fluoropiperidin- -4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid with 4-((1-(5-chloro-2-(trifluoromethyl) benzyl)-4-fluoropiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid and to replace methanesulfonamide with cyclopropanesulfonamide the title compound was obtained as a colorless solid (0.03 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.87 (br s, 1H), 9.99 (br s, 1H), 8.13 (br s, 1H), 7.61 (br s, 2H), 7.15 (d, J=8.3 Hz, 1H), 7.03 (d, J=12.8 Hz, 1H), 4.80-4.42 (m, 2H), 4.28 (d, J =20.6 Hz, 2H), 3.60-3.17 (m, 4H), 3.12-3.03 (m, 1H), 2.31-2.17 (m, 2H), 2.08-1.99 (m, 3H), 1.14-1.09 (m, 4H), 0.94-0.87 (m, 2H), 0.73-0.67 (m, 2H); MS(ES+) m/z 506.2, 504.0 (M+1).
Example 458
Synthesis of (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluoro-N- -(methylsulfonyl)benzamide
##STR01163##
[2002] Step 1. Preparation of (R)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol
##STR01164##
[2004] A 50 mL flask charged magnesium turnings (0.32 g, 13.33 mmol) was heated via heat gun under hi-vac. The flask was cooled to ambient temperature and flushed with argon before freshly distilled diethyl ether (1 mL) and 1,2-dibromoethane (2 drops) were added. The flask was equipped with a condenser before a solution of 4-bromo-1,2-dichlorobenzene (1.68 mL, 13.00 mmol) in diethyl ether (14 mL) was added dropwise so as to maintain a gentle reflux. The cloudy solution was stirred for 1 hour at ambient temperature. After cooling to 0.degree. C., copper iodide (0.21 g, 1.08 mmol) was added. After 10 minutes stiring, a solution of (R)-epichlorohydrin (0.85 mL, 10.80 mmol) in diethyl ether (14 mL) was added dropwise. The reaction mixture was slowly warmed to ambient temperature and stirred overnight, quenched with saturated aqueous ammonium chloride solution (10 mL) at 0.degree. C., and then poured into water (40 mL). The biphasic mixture was stirred until all solids dissolved. The blue aqueous layer was isolated and extracted with ethyl acetate (3.times.30 mL). The combined organic Layers were washed with brine (150 mL); dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified using flash chromatography [0% to 5% to 10% ethyl acetate in hexanes] to yield the title compound as a colourless oil (2.50 g, 80%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.38 (d, J=8.2 Hz, 1H), 7.35 (d, J=2.0 Hz, 1H), 7.09 (dd, J=2.0, 8.2 Hz, 1H), 4.06-3.99 (m, 1H), 3.62 (dd, J=3.9, 11.2 Hz, 1H), 3.49 (dd, J=6.3, 11.2 Hz, 1H), 2.84 (d, J=5.8 Hz, 1H), 2.83 (d, J=7.1 Hz, 1H), 2.22 (br s, 1H).
Step 2. Preparation of (R)-2-(3,4-dichlorobenzyl)morpholine
##STR01165##
[2006] To a solution of sodium hydroxide (2.50 g, 62.40 mmol) in water (5 mL) was added a solution of (R)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol (2.50 g, 10.40 mmol) in methanol (11 mL). After 5 minutes, 2-aminoethyl hydrogen sulfate (5.86 g, 41.60 mmol) was added in portions (4.times.1.47 g). The resulting suspension was heated at 40.degree. C. for 5 hours. Toluene (25 mL) and powdered sodium hydroxide (2.50 g, 62.40 mmol) were added. The reaction mixture was stirred overnight at 6.degree. C. After cooling to ambient temperature, the reaction mixture was quenched with water (300 mL). The aqueous layer was isolated and extracted with toluene (2.times.150 mL). The combined organics were washed with water (50 mL) and brine (50 mL), and concentrated. The residue was purified using flash chromatography [0% to 100% (80:10:10 ethyl acetate/isopropanol/triethylamine) in hexanes] to yield the title compound as a colourless oil (0.83 g, 32%): MS(ES+) m/z 246.1, 248.1 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(iodomethyl)benzoate
##STR01166##
[2008] To a suspension of polymer bound triphenylphosphine (2.60 g, 7.80 mmol) in dichloromehane (60 mL) was added imidazole (0.53 g, 7.80 mmol) and iodine (2.00 g, 7.80 mmol). After 5 minutes, tert-butyl 5-cyclopropyl-2-fluoro-4-(hydroxymethyl)benzoate (1.60 g, 6.0 mmol) was added and the reaction mixture was stirred at ambient temperature overnight. The reaction mixture was filtered and the filtrate was washed with saturated aqueous sodium bisulfate solution (50 mL) and water (50 mL). The organic layer was dried over sodium sulfate, decanted and concentrated. The residue was dissolved in ethyl acetate (100 mL) and washed with saturated aqueous sodium bisulfate solution (20 mL), 1N hydrochloric acid solution (20 mL) and brine (20 mL). The organic layer was dried over sodium sulfate, decanted and concentrated to yield the title compound as a yellow solid (2.14 g, 94%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.50 (d, J=7.3 Hz, 1H), 7.05 (d, J=10.9 Hz, 1H), 4.58 (m, 2H), 1.97-1.84 (m, 1H), 1.58 (s, 9H), 1.08-0.99 (m, 2H), 0.78-0.70 (m, 2H).
Step 4. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) metoxy)-2-fluorobenzoate
##STR01167##
[2010] A suspension of tert-butyl 5-cyclopropyl -2-fluoro-4-(iodomethyl)benzoate (0.46 g, 1.22 mmol), (R)-2-(3,4-dichlorobenzyl)morpholine (0.60 g, 2.44 mmol), potassium phosphate (0.52 g, 2.44 mmol) in N,N-dimethyl formamide (22 mL) was heated as 80.degree. C. for 2 hours. The reaction mixture was cooled to ambient temperature, and diluted with water (400 mL) and ethyl acetate (200 mL). The aqueous layer was isolated and extracted with ethyl acetate (3.times.100 mL). The combined organics were concentrated and the residue was purified by flash chromatography (0% to 10% ethyl acetate in hexanes) to yield the title compound (0.59 g, 97%): .sup.1NMR (300 MHz, CDCl.sub.3) .delta.7.49 (d, J=7.2 Hz, 1H), 7.33 (d, J=8.2 Hz, 1H), 7.32 (d, J=2.0 Hz, 1H), 7.15 (d, J=11.7 Hz, 1H), 7.05 (dd, J=2.0, 8.2 Hz, 1H), 3.84 (d, J=10.6 Hz, 1H), 3.77-3.67 (m, 1H), 3.67-3.54 (m, 3H), 2.80-2.58 (m, 4H), 2.30-2.16 (m, 1H), 2.07-1.90 (m, 2H), 1.58 (s, 9H), 1.00-0.87 (m, 2H), 0.70-0.58 (m, 2H); MS(ES+) m/z 494.2, 496.1 (M+1).
Step 5. Preparation of (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoic acid hydrochloride
##STR01168##
[2012] A solution of (R)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobenzoa- te (0.17 g, 0.34 mmol) and hydrogenchloride (4.0 N solution in 1,4-dioxane, 1.72 mL, 6.88 mmol) in 1,4-dioxane (5 mL) was stirred overnight at ambient temperature, and then heated at 60.degree. C. for 2 hours. The reaction mixture was cooled to ambient temperature and diluted with toluene (10 mL) and concentrated to yield the title compound (0.17 g, quant. yield), which was used without purification: MS(ES+) m/z 438.1, 440.0 (M+1); MS(ES-) m/z 436.1, 438.1 (M-1).
Step 6. Preparation of (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluoro-N- -(methylsulfonyl)benzamide
##STR01169##
[2014] To a solution of (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride (0.17 g, 0.36 mmol) in dichloromethane (10 mL) was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (0.10 g, 0.54 mmol), and 4-dimethylaminopyridine (0.10 g, 0.82 mmol). The reaction was stirred at ambient temperature for 10 minutes, methanesulfonamide (0.06 g, 0.58 mmol) was added and stirring was continued at ambient temperature overnight. The reaction mixture was concentrated, diluted with ethyl acetate (20 mL) and washed with 5% aqueous hydrochloric acid solution (10 mL). The organic layer was separated, washed with water and brine, dried over anhydrous sodium sulfate and concentrated. The residue was purified by flash chromatography (0% to 50% ethyl acetate in hexanes) to yield the title compound as a colorless solid (0.09 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.94-8.69 (m, 1H), 7.73 (d, J=7.8 Hz, 1H), 7.36-7.31 (m, 3H), 7.05 (d, J=8.1 Hz, 1H), 3.87 (d, J=11.2 Hz, 1H), 3.81-3.57 (m, 4H), 3.42 (s, 3H), 2.83-2.58 (m, 4H), 2.34-2.19 (m, 1H), 2.12-1.99 (m, 1H), 1.99-1.88 (m, 1H), 1.02-0.92 (m, 2H), 0.70-0.59 (m, 2H); MS(ES+) m/z 515.1, 517.1 (M+1), MS(ES-) m/z 513.2, 515.1 (M-1).
Example 459
Preparation of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl) morpholino)methyl)-2-fluorobenzamide
##STR01170##
[2016] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.08 g, 41): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.73 (d, J=15.7 Hz, 1H), 7.74 (d, J=7.8 Hz, 1H), 7.36-7.31 (m, 3H), 7.06 (dd, J=2.0, 8.2 Hz, 1H), 4.26 (t, J=7.7 Hz, 4H), 3.89-3.85 (m, 1H), 3.79-3.58 (m, 4H), 2.81-2.58 (m, 4H), 2.33-2.20 (m, 3H), 2.11-1.91 (m, 2H), 1.00-0.92 (m, 2H), 0.71-0.63 (m, 2H); MS(ES+) m/z 556.1, 558.1 (M+1), MS(ES-) m/z 554.2, 556.1 (M -1).
Example 460
Preparation or (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((2-(3,4-dichlorobenzyl) morpholino)methyl)-2-fluorobenzamide
##STR01171##
[2018] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.07g, 35%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.78 (d, J=15.0 Hz, 1H), 7.74 (d, J=7.8 Hz, 1H), 7.36-7.31 (m, 3H), 7.05 (dd, J=1.9, 8.2 Hz, 1H), 3.91-3.85 (m, 1H), 3.81-3.57 (m, 4H), 3.16-3.02 (m, 1H), 2.83-2.52 (m, 4H), 2.32-2.19 (m, 1H), 2.08-1.85 (m, 2H), 1.51-1.38 (m, 2H), 1.20-1.06 (m, 2H), 1.00-0.87 (m, 2H), 0.70-0.56 (m, 2H); MS(ES+) m/z 541.1, 543.1 (M+1), MS(ES-) m/z 539.1, 541.1 (M-1).
Example 461
Synthesis of (S)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluoro-N- -(methylsulfonyl)benzamide
##STR01172##
[2019] Step 1. Preparation of (S)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol
##STR01173##
[2021] Following the procedure as described in Example 458 step 1 and making no variations as required to replace (R)-epichlorohydrin with (S)-epichlorohydrin, the title compound was obtained as a colorless oil (2.70 g, 87%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.39 (d, J=8.3 Hz, 1H), 7.35 (d, J=2.0 Hz, 1H), 7.09 (dd, J=2.0, 8.2 Hz, 1H), 4.10-3.98 (m, 1H), 3.62 (dd, J=3.9, 11.2 Hz, 1H), 3.49 (dd, J=6.3 Hz, 1H), 2.85-2.82 (m, 2H), 2.20 (br s, 1H).
Step 2. Preparation of (S)-2-(3,4-dichlorobenzyl)morpholine
##STR01174##
[2023] Following the procedure as described in Example 458 step 2 and making non-critical variations as required to replace (R)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol with (S)-1-chloro-3-(3,4-dichlorophenyl) propan-2-ol, the title compound was obtained as a colorless oil (1.04 g, 37%): MS(ES+) m/z 246.1, 248.1 (M+1).
Step 3. Preparation of (S)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate
##STR01175##
[2025] Following the procedure as described in Example 458 step 4 and making non-critical variations as required to replace (R)-2-(3,4-dichlorobenzyl)morpholine with (S)-2-(3,4-dichlorobenzyl)morpholine, the title compound was obtained (0.46 g, 98%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.50 (d, J=7.3 Hz, 1H), 7.34 (d, J=8.2 Hz, 1H), 7.32 (d, J=2.0 Hz, 1H), 7.14 (d, J=11.7 Hz, 1H), 7.05 (dd, J=2.0, 8.2 Hz, 1H), 3.85 (d, J=10.6 Hz, 1H), 3.78-3.66 (m, 1H), 3.66-3.55 (m, 3H), 2.80-2.57 (m, 4H), 2.22 (dt, J=3.0,11.2 Hz, 1H), 2.05-1.92 (m, 2H), 1.58 (s, 9H), 0.97-0.89 (m, 2H), 0.67-0.59 (m, 2H); MS(ES+) m/z 494.0, 496.0 (M+1).
Step 4. Preparation of (S)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride
##STR01176##
[2027] Following the procedure as described in Example 458 step 5 and making non-critical variations as required to replace (R)-tert-butyl 5-cyclopropyl -4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate with (S)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate, the title compound was obtained as a cotoriess solid (0.43 g, 98%): MS(ES+) m/z 438.1, 440.0 (M+1); MS(ES-) 436.1, 438.1 (M-1).
Step 5. Preparation of (S)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluoro-N- -(methylsulfonyl)benzamide
##STR01177##
[2029] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (S)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride, the title compound was obtained as a colorless solid (0.04 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.94-8.69 (m, 1H), 7.73 (d, J=7.8 Hz, 1H), 7.36-7.31 (m, 3H), 7.05 (d, J=8.1 Hz, 1H), 3.87 (d, J=11.2 Hz, 1H), 3.81-3.57 (m, 4H), 3.42 (s, 3H), 2.83-2.58 (m, 4H), 2.34-2.19 (m, 1H), 2.12-1.99 (m, 1H), 1.99-1.88 (m, 1H), 1.02-0.92 (m, 2H), 0.70-0.59 (m, 2H); MS(ES+) m/z 515.1, 517.1 (M+1), MS(ES-) m/z 513.2, 515.2 (M-1).
Example 462
Preparation of (S)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((2-(3,4-dichlorobenzyl) morpholino)methyl)-2-fluorobenzamide
##STR01178##
[2031] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (S)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoic acid hydrochloride, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.02 g, 11%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.78 (d, J=13.5 Hz, 1H), 7.74 (d, J=7.8 Hz, 1H), 7.38-7.23 (m, 3H), 7.07-7.04 (m, 1H), 3.94-3.82 (m, 1H), 3.82-3.57 (m, 4H), 3.16-3.04 (m, 1H), 2.83-2.55 (m, 4H), 237-2.21 (m, 1H), 2.12-1.88 (m, 2H), 1.53-1.42 (m, 2H), 1.22-1.10 (m, 2H), 1.03-0.92 (m, 2H), 0.72-0.61 (m, 2H); MS(ES+) m/z 541.1, 543.1 (M+1), MS(ES-) m/z 539.2, 541.2 (M-1).
Example 463
Synthesis of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01179##
[2032] Step 1. Preparation of (R)-1-chloro-3-(3,5-dichlorophenyl)propan-2-ol
##STR01180##
[2034] Following the procedure as described in Example 458 step 1 and making non-critical variations as required to replace 4-bromo-1,2-dichlorobenzene with 1-bromo-3,5-dichlorobenzene, the title compound was obtained as a colorless oil (12.93 g, 83%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.27-7.24 (m, 1H), 7.18-7.12 (m, 2H), 4.09-3.98 (m, 1H), 3.62 (dd, J=3.9, 11.2 Hz, 1H), 3.49 (dd, J=6.3, 11.2 Hz, 1H), 2.87-2.79 (m, 2H), 2.22 (br s, 1H).
Step 2. Preparation of (R)-2-(3,5-dichlorobenzyl)morpholine
##STR01181##
[2036] Following the procedure as described in Example 458 step 2 and making non-critical variations as required to replace (R)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol with (R)-1-chloro-3-(3,5-dichlorophenyl)propan-2-ol, the title compound was obtaind as a colorless oil (6.98 g, 53%): MS(ES+) m/z 246.1, 248.1 (M+1).
Step 3. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate
##STR01182##
[2038] To a microwave vial was charged with tert-butyl 5-cyclopropyl-2-fluoro-4-(iodomethyl)benzoate (0.14 g, 0.37 mmol), (R)-2-(3,5-dichlorobenzyl)morpholine (0.18 g, 0.75 mmol), potassium phosphate (0.16 mg, 0.75 mmol) and N,N-dimethyl formamide (8 mL). The suspension was heated in the microwave reactor at 80.degree. C. for 2 hours. The reaction mixture was cooled to ambient temperature and diluted with water (200 mL) and ethyl acetate (100 mL). The aqueous layer was isolated and extracted with ethyl acetate (3.times.50 mL). The combined organics were concentrated and the residue was purified by flash chromatography (0% to 10% ethyl acetate in hexanes) to yield the title compound (0.18 g, 98%): MS(ES+) m/z 494.2, 496.2 (M+1).
Step 4. Preparation of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoic acid hydrochloride
##STR01183##
[2040] Following the procedure as described in Example 458 step 5 and making non-critical variations as required to replace (R)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate with (R)-tert-butyl 5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.08 g, 49%): MS(ES+) m/z 437.9, 439.9 (M+1); MS(ES-) m/z 436.0, 438.0 (M-1).
Step 5. Preparation of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01184##
[2042] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride, the title compound was obtained as a colorless solid (0.05 g, 24%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.95-8.69 (m, 1H), 7.73 (d, J=7.8 Hz, 1H), 7.34-7.20 (m, 2H), 7.14-7.11 (m, 2H), 3.92-3.83 (m, 1H), 3.81-3.31 (m, 2H), 3.67 (s, 2H), 3.42 (s, 3H), 2.82-2.59 (m, 4H), 2.27 (dt, J=3.1, 11.2 Hz, 1H), 2.11-1.89 (m, 2H), 1.02-0.92 (m, 2H), 0.70-0.59 (m, 2H); MS(ES+) m/z 515.1, 517.1 (M+1), MS(ES-) m/z 513.1, 515.1 (M+1).
Example 464
Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((2-(3,5-dichlorobenzyl) morpholino)methyl)-2-fluorobenzamide
##STR01185##
[2044] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoic acid hydrochloride, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.03 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.78 (d, J=15.7 Hz, 1H), 7.74 (d, J=7.8 Hz, 1H), 7.33-7.19 (m, 2H), 7.11-7.07 (m, 2H), 3.92-3.85 (m, 1H), 3.81-3.71 (m, 1H), 3.67 (s, 2H), 3.65-3.60 (m, 1H), 3.16-3.04 (m, 1H), 2.82-2.62 (m, 4H), 2.27 (dr, J=3.1, 11.2 Hz, 1H), 2.05 (t, J=10.4 Hz, 2H), 2.01-1.90 (m, 1H), 1.51-1.44 (m, 2H), 1.21-1.11 2H), 1.01-0.94 (m, 2H), 0.71-0.64 (m, 2H); MS(ES+) m/z 541.1, 543.0 (M+1), MS(ES-) m/z 539.1, 541.1 (M-1).
Example 465
Preparation of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)mor- pholino) methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01186##
[2046] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained by reverse-phase HPLC purification as a colorless solid (0.04 g, 37%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.77 (d, J=7.4 Hz, 1H), 7.43 (d, J=11.5 Hz, 1H), 7.27-7.26 (m, 1H), 7.07 (s, 2H), 4.49 (s, 2H), 4.25 (t, J=7.7 Hz, 4H), 4.15-3.88 (m, 3H), 3.55 d, J=11.1 Hz, 2H), 2.97-2.77 (m, 2H), 2.75-2.64 (m, 1H), 2.63-2.49 (m, 1H), 2.36-2.22 (m, 2H), 1.51-1.44 (m, 2H), 1.93-1.79 (m, 1H), 1.17-1.03 (m, 2H), 0.84-0.70 (m, 2H); MS(ES+) m/z 556.1, 558.1 (M+1), MS(ES-) m/z 554.2, 556.2 (M-1).
Example 466
Synthesis of (R)-4-((2-(2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide trifluoroacetic acid salt
##STR01187##
[2047] Step 1. Preparation of (R)-1-chloro-3-(2-chloro-4-fluorophenyl)propan-2-ol
##STR01188##
[2049] Following the procedure as described in Example 458 step 1 and making non-critical variations as required to replace 4-bromo-1,2-dichlorobenzene with 1-bromo-2-chloro-4-fluorobenzene, the title compound was obtained as a colorless oil (3.22 g, 55%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.28 (dd, J=6.2 Hz, 1H), 7.12 (dd, J=2.6, 8.5 Hz, 2H), 6.95 (dt, J=2.6, 8.3 Hz, 1H), 4.19-4.09 (m, 1H), 3.72-3.66 (m, 1H), 3.59-3.50 (m, 1H), 3.06-2.90 (m, 2H), 2.17 (br s, 1H).
Step 2. Preparation of (R)-2-(2-chloro-4-fluorobenzyl)morpholine
##STR01189##
[2051] Following the procedure as described in Example 458 step 2 and making non-critical variations as required to replace (R)-1-chloro-3-(3,4-dichlorophenyl)propan-2-ol with (R)-1-chloro-3-(2-chloro-4-fluorophenyl) propan-2-ol, the title compound was obtained as a colorless oil (0.66 g, 20%): MS(ES+) m/z 230.2, 232.2 (M+1).
Step 3. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((-2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-2-fluorobenzoate
##STR01190##
[2053] Following the procedure as described in Example 455 step 4 and making non-critical variations as required to replace (R)-2-(3,4-dichlorobenzyl)morpholine with (R)-2-(2-chloro-4-fluorobenzyl)morpholine, the title compound was obtained (0.47 g, quant. yield): MS(ES+) m/z 478.2, 480.2 (M+1).
Step 4. Preparation of (R)-4-((2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01191##
[2055] Following the procedure as described in Example 458 step 5 and making non-critical variations as required to replace (R)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate with (R)-tert-butyl 5-cyclopropyl-4-((2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-2-fluorobenzoate, the title compound was obtainned as a colorless solid (0.45 g, 97%): MS(ES+) m/z 422.0, 424.0(M+1); MS(ES-) m/z 420.1, 422.1 (M-1).
Step 5. Preparation of (R)-4-((-2-(2-chloro-4-fluorobenzyl)morphlino) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01192##
[2057] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholine)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-4-((2-(2-chloro-4-fluorobenzyl)morpholino)methyl)-5-cyclopropyl-2-flu- orobenzoic acid hydrochloride, the title compound was obtained as a colorless solid (0.07 g, 38%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.71 (d, J=7.7 Hz, 1H), 7.38 (d, J=11.5 Hz, 1H), 7.27-7.16 (m, 1H), 7.14-7.07 (m, 1H), 6.98-6.90 (m, 1H), 4.51 (s, 2H), 4.19-4.09 (m, 1H), 4.09-3.88 (m, 2H), 3.57 (dd, J =11.5, 26.0 Hz, 2H), 3.39 (s, 3H), 3.02-2.88 (m, 3H), 2.68 (t, J=11.0 Hz, 1H), 1.94-1.83 (m, 1H), 1.16-1.06 (m, 2H), 0.82-0.75 (m, 2H); MS(ES+) m/z 499.1, 501.1 (M+1); MS(ES-) m/z 497.1, 499.1 (M-1).
Example 467
Preparation of (R)-4-((2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01193##
[2059] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-4-((2-(2-chloro-4-fluorobenzyl)morpholino)methyl)-5-cyclopropyl-2-flu- orobenzoic acid hydrochloride, and to replace methanesulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.06 g, 32%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.73 (d, J=7.3 Hz, 1H), 7.37 (d, J=11.5 Hz, 1H), 7.20 (dd, J=6.2, 8.4 Hz, 1H), 7.11 (dd, J=2.3, 8.4 Hz, 1H), 6.94 (dt, J=2.4, 8.2 Hz, 1H), 4.52 (s, 2H), 4.21-3.81 (m, 3H), 3.59 (dd, J=11.2, 26.5 Hz, 2H), 3.14-2.95 (m, 1H), 3.01-2.93 (m, 3H), 2.70 (t, J=10.8 Hz, 1H), 1.95-1.80 (m, 1H), 1.49-1.37 (m, 2H), 1.23-1.37 (m, 4H), 0.80-0.68 (m, 2H); MS(ES+) m/z 525.1, 527.1 (M+1); MS(ES-) m/z 523.1, 525.1 (M-1).
Example 468
Preparation of (R)-N-(azetidin-1-ylsulfonyl)-4-((2-(2-chloro-4-fluorobenzyl)morpholino) methyl)-5-cyclopropyl-2-fluorobenzamide, trifluoroacetic acid salt
##STR01194##
[2061] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-4-((2-(2-chloro-4-fluorobenzyl)morpholino)methyl)-5-cyclopropyl-2-flu- orobenzoic acid hydrochloride, and to replace methanesulfonamide with azetidine-1-sulfonamide, the title cumpouad was obtanied as a colorless solid (0.06 g, 37%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.77 (d, J=7.2 Hz, 1H), 7.42 (d, J=11.6 Hz, 1H), 7.25-7.20 (m, 1H), 7.14-7.10 (m, 1H), 6.98-6.91 (m, 1H), 4.49 (s, 2H), 4.25 (t, J=7.7 Hz, 4H), 4.20-4.15 (m, 1H), 4.05-3.92 (m, 2H), 3.55 (dd, J=11.2, 26.5 Hz, 2H), 3.02-2.82 (m, 3H), 2.65 (t, J=10.6 Hz, 1H), 2.37-2.22 (m, 2H), 1.96-1.82 (m, 1H), 1.16-1.03 (m, 2H), 0.82-0.69 (m, 2H); MS(ES+) m/z 540.1, 542.1 (M+1); MS(ES-) m/z 538.1, 540.1 (M-1).
Example 469
Synthesis of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5-dimethylmorpholino) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01195##
[2062] Step 1. Preparation of (R)-1-amino-3-(3,5-dichlorophenyl)propan-2-ol
##STR01196##
[2064] To a solution of (R)-1-chloro-3-(3,5-dichlorophenyl)propan-2-ol (1.44 g, 6.01 mmol) at N,N-dimethyl formamide (45 mL) was added sodium azide (1.95 g, 30.05 mmol) and sodium iodide (0.54 g, 3.60 mmol). The resulting suspension was heated at 75.degree. C. overnight, cooled to ambient temperature, diluted with ethyl acetate (150 mL) and washed with water (50 mL), 5% aqueous lithium chloride solution (50 mL) and brine (50 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated. The residue was dissolved in tetrahydrofuran (55 mL) and water (18 mL), and to this solution was added triphenylphosphine (1.85 g, 7.07 mmol). The resulting reaction mixture was heated at 50.degree. C. for 24 hours. Upon cooling to ambient temperature, the reaction mixture was diluted with dichloromethane (200 mL) and 10% aqueous sodium bicarbonate solution (100 mL). The aqueous layer was isolated and extracted with dichloromethane (2.times.100 mL). The combined organics were washed with brine (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by flash chromatography [0% to 100% (85:14:1 dichloromethane/ethanol/ammonium hydroxide) in dichloromethane] to yield the title compound (0.93 g, 70% over 2 steps): MS(ES+) m/z 220.1, 222.1 (M+1).
Step 2. Preparation of (R)-2-chloro-N-(3-(3,5-dichlorophenyl)-2-hydroxypropyl)acetamide
##STR01197##
[2066] To a cooled (0.degree. C.) solution of (R)-1-amino-3-(3,5-dichlorophenyl)propan-2-ol (0.86 g, 3.91 mmol) and triethylamine (0.718 mL, 5.08 mmol) in 9:1 dichloromethane/acetonitrile (60 mL) was added 2-chloroacetyl chloride (340 .mu.L, 4.29 mmol) dropwise. The resulting solution was stirred at 0.degree. C. for 1.5 hours, and then warmed to ambient temperature and stirred for 30 minutes. The reaction mixture was re-cooled (0.degree. C.) and quenched with 5% aqueous hydrochloric acid solution (15 mL). The aqueous layer was isolated and extracted with dichloromethane (2.times.30 mL). The combined organics were washed with brine (20 mL), dried over sodium sulfate, filtered and concentrated. The residue was triturated with diethyl ether to yield the title compound as a colorless solid (0.87 g, 75%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.27-7.26 (m, 2H), 7.13-7.11 (m, 1H), 6.98 (br s, 1H), 4.09 (s, 2H), 4.05-3.95 (m, 1H), 3.64-3.53 (m, 1H), 3.33-3.21 (m, 1H), 2.84-2.64 (m, 2H), 1.58 (br s, 1H); MS(ES+) m/z 296.0, 298.0 (M+1).
Step 3. Prepartion of (R)-6-(3,5-dichlorobenzyl)morpholin-3-one
##STR01198##
[2068] To a cooled (0.degree. C.) solution of (R)-2-chloro-N-(3-(3,5-dichlorophenyl)-2-hydroxypropyl)acetamide (0.31 g, 1.03 mmol) in isopropanol (2 mL) and toluene (1 mL) was added a solution of potassium tert-butoxide (0.32 g, 2.88 mmol) in isopropanol (3 mL). The reaction was stirred at 0.degree. C. for 1 hour, and then slowly warmed to ambient temperature and stirred overnight. The reaction mixture was neutralized to pH=6 with 5% aqueous hydrochloric acid solution, and then concentrated. The aqueous residue was diluted with toluene (75 mL) and saturated aqueous sodium bicarbonate solution (25 mL). The toluene layer was isolated, washed with brine (20 mL) and then concentrated. The combined aqueous layers were extracted with ethyl acetate (2.times.50 mL). The combined organics were dried over sodium sulfate, filtered and concentrated. The residue was purified using flash chromatography [0% to 5% methanol in dichloromethane] to yield the title compound as a colorless solid (0.23 g, 87%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.32-7.12 (m, 2H), 7.11 (s, 1H), 6.32 (br s, 1H), 4.29 (d, J=16.9 Hz, 1H), 4.13 (d, J=16.9 Hz, 1H), 3.96-3.82 (m, 1H), 3.41-3.22 (m, 2H), 2.88 (dd, J =7.4, 14.2 Hz, 1H), 2.74 (dd, J=5.1, 14.2 Hz, 1H), 1.87 (br s, 1H).
Step 4. Preparation of (R)-6-(3,5-dichlorobenzyl)-4-(4-methoxybenzyl)morpholin-3-one
##STR01199##
[2070] To a cooled (0.degree. C.) solution of (R)-6-(3,5-dichlorobenzyl)morpholin-3-one (0.23 g, 0.89 mmol) in N,N-dimethyl formamide (3 mL) was added sodium hydride (60% dispersion, 0.04 g, 1.08 mmol). The resulting suspension was warmed to ambient temperature and stirred for 15 minutes. The pink slurry was re-cooled (0.degree. C.) before 4-methoxybenzylchloride (0.15 mL, 1.08 mmol) was added dropwisely. At this point the slurry was too thick to stir and N,N-dimethyl formamide (2 mL) was added before the reaction mixture was slowly warmed to ambient temperature and stirred overnight. The reaction was cooled (0.degree. C.), quenched with saturated aqueous ammonium chloride solution (5 mL) and diluted with ethyl acetate (50 mL). The aqueous layer was isolated and extracted with ethyl acetate (2.times.20 mL). The combined organics were washed with brine (20 mL), dried over sodium sulfate, flitered and concentrated. The residue was purified using flash chromatography [0% to 20% to 40% ethyl acetate in hexanes] to yield the title compound as a colourless oil (0.27 g, 79): .sup.1H NMR (300 CDCl.sub.3) .delta.7.28-7.16 (m, 3H), 7.08-7.03 (m, 2H), 6.91-6.84 (m, 2H), 4.62 (d, J=14.5 Hz, 1H), 4.43 (d, J=14.5 Hz, 1H), 4.19-4.07 (m, 1H), 3.94-3.81 (m, 1H), 3.80 (s, 3H), 3.24-3.02 (m, 2H), 2.96 (s, 1H), 2.88 (s, 1H), 2.77 (dd, J=7.8, 14.3 Hz, 1H), 2.67 (dd, J=4.6, 14.3 Hz, 1H).
Step 5. Preparation of (R)-2-(3,5-dichlorobenzyl)-4-(4-methoxybenzyl)-5,5-dimethylmorpholine
##STR01200##
[2072] To a cooled (-10.degree. C.) solution of (R)-6-(3,5-dichlorobenzyl)-4-(4-methoxybenzyl)morpholin-3-one (0.27 g, 0.71 mmol) tetrahydrofuran (1.5 mL) was added anhydrous zirconium(IV) chloride (0.17 g, 0.72 mmol). After 30 minutes, a solution of methylmagnesium bromide (3.0M in diethyl ether, 1.4 4.26 mmol) was added dropwisely. After 1 hour at -10.degree. C., the slurry was too thick to stir, so tetrahydrofuran (2 mL) was added. The resulting suspension was stirred -10.degree. C. for an additional 1 hour, and then slowly warmed to ambient temperature and stirred overnight. The reaction was cooled (0.degree. C.), diluted with diethyl ether (10 mL) and saturated aqueous sodium/potassium tartarate solution (5 mL). The thick slurry was stirred for 15 minutes, resulting in a cloudy yellow biphasic solution. The solid was removed by filtration and rinsed with ethyl acetate and water. The organic layer was isolated and the aqueous layer was extracted with ethyl acetate (2.times.50 mL). The combined organics were washed with water (20 mL) then brine (20 mL), dried over sodium sulfate, and concentrated. The residue was purified using flash chromatography [0% to 15% to 30% ethyl acetate in hexanes] to yield the title compound as a colourless oil (0.13 g, 46%): MS(ES+) m/z 394.0, 396.0 (M+1).
Step 6. Preparation of (R)-2-(3,5-dichlorobenzyl)-5,5-dimethylmorpholine
##STR01201##
[2074] To a solution of (R)-2-(3,5-dichlorobenzyl)-4-(4-methoxybenzyl)-5,5-dimethylmorpholine (0.13 g, 0.32 mmol) in water/acetonitrile (1:1, v/v, 7 mL) was added a solution of ceric ammonium nitrate (535 mg, 0.975 mmol) in methanol (56 mL). The resulting orange solution was stirred at ambient temperature overnight, quenched with 5% aqueous hydrochloric acid solution (20 mL), and then extracted with diethyl ether (3.times.20 mL). The combined aqueous layer was basified with solid sodium bicarbonate to pH=9 and extracted with ethyl acetate (3.times.75 mL). The combined ethyl acetate extracts were washed with brine (20 mL), dried ever sodium sulfate, filtered and concentrated to give the title compound as a colorless solid (0.08 g, 98%): MS(ES+) m/z 274.1, 276.1 (M+1).
Step 7. Preparation of (R)-tert-butyl 5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5-dimethyl morpholino)methyl)-2-fluorobenzoate
##STR01202##
[2076] To a suspension of (R)-2-(3,5-dichlorobenzyl)-5,5-dimethylmorpholine (0.08 g, 0.30 mmol) and potassium carbonate (0.05 g, 0.34 mmol) in N,N-dimethyl formamide (2 mL) was added tert-butyl 5-cyclopropyl-2-fluoro-4-(((methylsulfonyl)oxy)methyl) benzoate (0.12 g, 0.34 mmol). The suspension was stirred at ambient temperature overhight, diluted with water (30 mL ) and ethyl acetate (75 mL). The aqueous layer was isolated and extracted with ethyl acetate (3.times.50 mL). The combined organics were washed with brine (50 mL), and concentrated. The residue was purified by flash chromatogpaphy [0% to 10% ethyl acetate in hexanes] to yield the title compound (0.08 g, 54%): MS(ES+) m/z 522.1, 524.1 (M+1).
Step 8. Preparation of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5dimethylmorpholino) methyl)-2-fluorobenzoic acid hydrochloride
##STR01203##
[2078] Following the procedure as described in Example 458 step 5 and making non-critical variations as required to replace (R)-tert-butyl 5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino) methyl)-2-fluorobenzoate with (R)-tert-butyl 5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5-dimethyl morpholino)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.08 g, quant. yield): MS(ES+) m/z 466.0, 468.0 (M+1); MS(ES-) m/z 464.1, 466.1 (M-1).
Step 9. Preparation of (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5-dimethylmorpholino) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR01204##
[2080] Following the procedure as described in Example 458 step 6 and making non-critical variations as required to replace (R)-5-cyclopropyl-4-((2-(3,4-dichlorobenzyl)morpholino)methyl)-2-fluorobe- nzoic acid hydrochloride with (R)-5-cyclopropyl-4-((2-(3,5-dichlorobenzyl)-5,5-dimethylmorpholino) methyl)-2-fluorobenzoic acid hydrochloride, the title compound was obtained by reverse-phase HPLC purification as a colorless solid (0.06 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46 (d, J=7.0 Hz, 1H), 7.31-7.19 (m, 2H), 7.09-7.03 (m, 2H), 4.58 (d, J=13.2 Hz, 1H), 4.06-3.96 (m, 1H), 3.91 (d, J=13.2 Hz, 1H), 3.84 (d, J=12.7 Hz, 1H), 3.58 (d, J=12.6 Hz, 1H), 3.36 (s, 3H), 3.20 (d, J=11.9 Hz, 1H), 2.78 (dd, J=14.5 Hz, 1H), 1.90-1.77 (m, 1H), 1.50 (s, 3H), 1.42 (s, 3H), 1.10-0.94 (m, 2H), 0.88-0.79 (m, 2H), 0.61-0.51 (m, 2H); MS(ES+) m/z 543.0, 545.0 (M+1); MS(ES-) m/z 541.0, 543.0 (M-1).
Example 470
Synthesis of 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-N-(cycloprop- ylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01205##
[2081] Step 1. Preparation of (1R,3s,5S)-tert-butyl 3-((methylsulfonyl)oxy)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01206##
[2083] To a cold (0.degree. C.) mixture of (1R,3s,5S)-tert-butyl 3-hydroxy-8-azabicyclo[3.2.1]octane-8-carboxylate (7.68 g, 33.80 mmol) and triethylamine (7.1 mL, 50.70 mmol) in anhydrous dichloromethane (100 mL) was added methanesulfonyl chloride (3.1 mL, 40.60 mmol) and the reaction mixture was stirred for 1 hour at 0.degree. C. The organic phase was washed with hydrochloric acid solution (1 N, 10 mL), water (20 mL), and brine (20 mL); dried over anhydrous sodium sulfate. Filtration and concentration of filtrate in vacuo provided the title compound as a yellowish solid (10.40 Hz, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 5.09-4.96 (m, 1H), 4.29-4.17 (m, 2H), 2.97 (s, 3H), 2.10-1.93 (m, 4H), 1.90-1.75 (m, 2H), 1.68-1.59 (m, 2H) 1.44 (s, 9H).
Step 2. Preparation of (1R,3s,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01207##
[2085] To a mixture or (1R,3s,5S)-tert-butyl 3-hydroxy-8-azabicyclo[3.2.1]octane-8-8-carboxylate (1.44 g, 4.70 mmol) in anhydrous dimethylformamide (10 mL) was added cesium carbonate (3.06 g, 9.40 mmol) and 3-chloro-2-fluoro-5-(trifluoromethyl)phenol (1.10 g, 4.70 mmol) and the reaction mixture was heated at 80.degree. C. for 16 hours. After cooling to ambient temperature, the reaction mixture was diluted with ethyl acetate (150 mL) and water (20 mL). The organic phase was washed with water (2.times.15 mL), brine (15 mL), and dried over anhydrous sodium sulfate. After filtration and concentration of the filtrate in vacuo, the residue was purified by flash chromatography (0 to 30% ethyl acetate in hexanes) to afford the title compound as a colorless oil (2.00 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.19 (d, J=6.5 Hz, 1H), 6.98-6.93 (m, 1H), 4.71-4.65 (m, 1H), 4.25-4.19 (m, 2H), 2.24-2.09 (m, 4H), 2.06-1.90 (m, 4H), 1.46 (s, 9H); MS(ES+) m/z 368.0, 370.0 (M-55).
Step 3. Preparation of (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane
##STR01208##
[2087] To a mixture of (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate (2.00 g, 4.70 mmol) in dichloromethane (10 mL) was added trifluoroacetic acid (2 mL) and the reaction mixture was stirred at ambient temperature for 16 hours. After evaporation of all volatiles in vacuo, the residue was dissolved in dichloromethane (100 mL). The organic phase was washed with sodium hydroxide solution (1 N, 10 mL), water (10 mL), and brine (10 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate in vacuo yielded the title compound as a colorless oil (1.50 g, quant. yield): MS(ES+) m/z 324.1, 326.1 (M+1).
Step 4. Preparation of tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octane-8-yl)methyl)-5- cyclopropyl-2-fluorobenzoate
##STR01209##
[2089] To a mixture of (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane (1.50 g, 4.70 mmol) in anhydrous dimethylformamide (10 mL) was added potassium carbonate (1.30 g, 9.40 mmol) and tert-butyl 5-cyclopropyl-2-fluoro-4-(((methylsulfonyl) oxy)methyl)benzoate (1.62 g, 4.70 mmol) and the reaction mixture was stirred at ambient temperature for 16 hours. After dilution with ethyl acetate (150 mL) and addition of water (20 mL), the organic phase was washed with water (2.times.15 mL), brine (15 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate in vacuo gave a residue which was purified by flash chromatography (0 to 20% ethyl acetate in hexanes) to afford the title compound as a colorless oil (1.30 g, 48%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.2 Hz, 1H), 7.35-7.28 (m, 1H), 7.24-7.20 (m, 1H), 4.68-4.60 (m, 1H), 6.98-6.94 (m, 1H), 3.73-3.61 (m, 2H), 3.25-3.08 (m, 2H), 2.27-1.85 (m, 9H), 1.57 (s, 9H), 0.95-0.85 (m, 2H), 0.64-0.58 0 (m,2H); MS(ES+) m/z 572.1, 574.1 (M +1).
Step 5. 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenz- oic acid hydrochloride
##STR01210##
[2091] A mixture of tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzozte (1.30 g, 2.27 mmol) and concentrated hydrochloric acid (3 mL) in dioxane (20 mL) was stirred at ambient temperature for 16 hours. Concentration of the reaction mixture in vacuo followed by co-evaporation with toluene (2.times.10 mL) provided the title compound as an off-white solid containing traced of toluene (1.30 g, quant. yield): MS(ES+) m/z 514.1, 516.1 (M+1).
Step 6. Preparation of 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-N-(cycloprop- ylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01211##
[2093] A mixture of 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenz- oic acid hydrochloride (0.63 g, 1.10 mmol), cyclopropanesulfonamide (0.34 g, 3.60 mmol), N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (0.69 g, 3.60 mmol), and 4-dimethylaminopyridine (0.88 g, 7.20 mmol) in anhydrous dichloromethane (10 mL) was stirred at ambient temperature for 16 hours. The reaction mixture was diluted with ethyl acetate (150 mL) and washed with aqueous hydrochloric acid solution (1 M, 10 mL), water (10 mL) and brine (10 mL). The organic phase was dried over anhydrous sodium sulfate, filtered, and the filtrate concentrated in vacuo. The residue was purified reverse phase HPLC (acetonitrile in water with 0.1% trifluoroacetic acid) to give the title compound as a colorless solid (0.38 g, 47%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 9.58 (br s, 1H), 7.67-7.53 (m, 3H), 7.27 (d, J=7.1 Hz, 1H), 5.00-4.92 (m, 1H), 4.42 (brs, 2H), 4.01 (br s, 2H), 3.12-2.99 (m, 1H), 2.46-2.23 (m, 6H), 2.22-2.05 (m, 3H), 1.15-1.05 (m, 4H), 1.04-0.94 (m, 2H), 0.82-0.73 (m, 2H); MS(ES-) m/z 617.1, 619.1 (M-1).
Example 471
Synthesis of 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(- methylsulfonyl)benzamide 2,2,2-trifluoroacetate
##STR01212##
[2095] Following the procedure as described in Example 470 step 6, and making variation as required to replace cyclopropanesulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.35 g, 45%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.32 (br s, 1H), 9.46 (br s, 1H), 7.67-7.51 (m, 3H), 7.29 (d, J=7.2 Hz, 1H), 4.97 (s, 1H), 4.42 (s, 2H), 4.01 (s, 2H), 3.34 (s, 3H), 2.44-2.26 (m, 6H), 2.22-2.06 (m, 3H), 1.03-0.96 (m, 2H), 0.81-0.74 (m, 2H); MS(ES-) m/z 591.1, 593.1 (M-1).
Example 472
Synthesis of 4-((4-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-4-methylpiperidin-1-- yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01213##
[2096] Step 1. Preparation of tert-butyl 4-(bromomethyl)-5-cyclopropyl-2-fluorobenzoate
##STR01214##
[2098] To a cold (0.degree. C.) mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-(hydroxymethyl)benzoate (1.0 g, 3.80 mmol) and triphenylphosphine (1.48 g, 5.60 mmol) in anhydrous tetrahydrofuran (20 mL) was added tetrabromomethane (1.87 g, 5.60 mmol) and the reaction mixture was stirred for 3 hours at 0.degree. C. After concentration in vacuo, the residue was purified by flash chromatography (0 to 20% ethyl acetate in hexanes) to afford the title compound as an off-white solid (1.24 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) 7.52 (d, J=7.2 Hz, 1H), 7.06 (d, J=10.8 Hz, 1H), 4.62. (s, 2H), 2.04-1.92 (m, 1H), 1.56 (s, 9H), 1.04-0.90 (m, 2H), 0.74-0.67. (m, 2H).
Step 2. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxy-4-methylpiperidin-1-yl)methyl)benzoa- te
##STR01215##
[2100] Following the procedure as described in Example 470 step 4, and making variations as required to replace (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with 4-hydroxy-methylpiperidine, and to replace tert-butyl 5-cyclopropyl-2-fluoro-4-(((methylsulfonyl) oxy)methyl)benzoate with tert-butyl 4-(bromomethyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as an orange oil (1.10 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.2 Hz, 1H), 7.17 (d, J=11.9 Hz, 1H), 3.64 (s, 2H), 2.59-2.49 (m, 2H), 2.47-2.36 (m, 2H), 1.99-1.88 (m, 1H), 1.73-1.51 (m, 14H), 1.24 (s, 3H), 0.93-0.85 (m, 2H), 0.63-0.57 (m, 2H).
Step 3. Preparation of tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-4-methylpiperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01216##
[2102] To a mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxy-4-methylpiperidin-1-yl) methyl)benzoate (1.08 g, 2.96 mmol) and 2,5-dichloro-4-(trifluoromethyl)pyridine (0.96 g, 4.40 mmol) in anhydrous tetrahydrofuran (5 mL) was added lithium bis(trimethylsilyl)amide (1.0 M in tetrahydrofuran, 4.4 mL, 4.4 mmol) and the reaction mixture was heated at 80-100.degree. C. for 48 hours. After cooling to ambient temperature, the reaction mixture was diluted with ethyl acetate (100 mL). The organic phase was washed with 1 M aqueous hydrochloric acid solution (10 ml), water (2.times.10 mL), brine (10 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate provided a residue which was purified by flash chromatography (0 to 20% ethyl acetate in hexanes) to afford the title compound as a colorless oil (0.19 g, 12%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.16 (s, 1H), 7.47 (d, J=7.3 Hz, 1H), 7.17 (d, J=11.8 Hz, 1H), 6.98 (s, 1H), 3.62 (s, 2H), 2.62-2.53 (m, 2H), 2.43-2.30 (m, 4H), 1.99-1.88 (m, 1H), 1.83-1.71 (m, 2H), 1.60 (s, 3H), 1.58 (d, J=13.7 Hz, 9H), 0.93-0.85 (m, 2H), 0.63-0.57 (m, 2H); MS(ES+) m/z 543.2, 545.2 (M+1).
Step 4. Preparation of 4-((4-(-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-4-methylpiperidin-1-yl)methyl)-5cyclopropyl-2-fluorobenzoic acid
##STR01217##
[2104] Following the procedure as described in Example 470 step 5, and making variation as required to replace tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl) pyridin-2-yl)oxy)-4-methylpiperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobe- nzoate, the title compound was obtained as a yellowish solid (0.17 g, quant. yield): MS(ES+) m/z 485.2, 487.1 (M+1).
Step 5. Preparation of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-4-methylpiperidin-1- -yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide 2,2,2,-trifluoroacetate
##STR01218##
[2106] Following the procedure as described in Example 470 Step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((4-((5-chloro-4-(trifluoromethyl) pyridin-2-yl)oxy)-4-methylpiperidin-1-yl)methyl)-5-cyclopropyly2-fluorobe- nzoic acid, the title compound was obtained as a colorless solid (0.07 mg, 31%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.47 (br s, 1H), 9.24 (br s, 1H), 8.22 (s, 1H), 7.74 (d, J=7.4 Hz, 1H), 7.49 (d, J=11.8 Hz, 1H), 7.03 (s, 1H), 4.46 (s, 2H), 3.58-3.44 (m, 2H), 3.12-2.97 (m, 3H), 2.78-2.67 (m, 2H), 2.28-2.11 (m, 2H), 1.91-1.80 (m, 1H), 1.59 (s, 3H), 1.48-1.40 (m, 2H), 1.19-1.11 (m, 2H), 1.10-1.02 (m, 2H), 0.76-0.60 (m, 2H); MS(ES-) m/z 588.1, 590.0 (M-1).
Example 473
Synthesis of 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoracetic acid saLt
##STR01219##
[2107] Step 1. Preptiration of tert-butyl 4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidine-1-carboxylate
##STR01220##
[2109] To a mixture of sodium hydride (60% dispersion in mineral oil, 0.44 g, 11.00 mmol) in anhydrous dimethylformamide (20 mL) was added 1-Boc-4-hydroxypiperidine (2.00 g, 10.00 mmol). The reaction mixture was stirred for 1 hour at ambient temperature, after which 5-bromo-2,3-dichloropyridine (2.27 g, 10.00 mmol) was added. The reaction mixture was heated to 70.degree. C. for 16 hours. After cooling to ambient temperature, the reaction mixture was partitioned between water (50 mL) and ethyl acetate (200 mL). The organic phase was washed with brine (20 mL), dried over anhydrous sodium sulfate, and filtered. Concentration of the filtrate in vacuo provided the title compound as yellowish oil (3.90 g, quant. yield), which was used without further purification: MS(ES+) m/z 335.0, 337.0 (M-55).
Step 2. Preparation of 5-bromo-3-chloro-2-(piperidin-4-yloxy)pyridine
##STR01221##
[2111] Following the procedure as described in Example 470 step 3, and making variation as required to replace (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate with tert-butyl 4-((5-bromo-3-chloropyridin-2-yl) oxy)piperidine-1-carboxylate, the title compound was isolated as a yellowish oil (2.90 g, quant. yield): MS(ES+) m/z 291.0, 293.0, 295.0 (M+1).
Step 3. Preparation of tert-butyl 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01222##
[2113] Following the procedure as described in Example 470 Step 4, and making variation as required to replace (1R,3r5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[3- .2.1]-octane with 5-bromo-3-chloro-2-(piperidin-4-yloxy)pyridine, the title compound was obtined as a yellowish solid (2.70 g, quant. yield): MS(ES+) m/z 539.2, 541.2 (M+1).
Step 4: Preparation of 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01223##
[2115] Following the procedure as described in Example 470 step 5, and making variation as required to replace tert-butyl 4-(((1R,3r5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabicy- clo[3.2.1]-octan-80-yl) methyl)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 4-((4-((5-bromo-3-chloropyridin-2-yl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was isolated as an off-white solid (1.09 g, quant. yield): MS(ES+) m/z 483.0, 485.00 (M+1).
Step 5: Preparation of 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01224##
[2117] To a mixture of 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl)methyl)-5-cyclopr- opyl-2-fluorobenzoic acid hydrochloride (1.09 g, 2.10 mmol) in anhydrous tetrahydrofuran (10 mL) was added 1,1'-carbonyldiimidazole (0.68 g, 4.20 mmol) and 1,8-diazabicyclo[5.4.0]undec-7-ene (0.63 mL, 4.20 mmol) and the reaction mixture was heated at 70.degree. C. for 30 minutes. After cooling to ambient temperature, cyclopropanesulfonamide (0.51 g, 4.20 mmol) and 1,8-diazabicylo[5.4.0]undec-7-ene (0.63 mL, 4.20 mmol) was added and the reaction mixture was heated at 70.degree. C. for 4 hours. After cooling to ambient temperature, the reaction mixture was diluted with ethyl acetate (200 mL). The organic phase washed with hydrochloric acid (1 N, mL), water (10 mL), brine (10 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate in vacuo provided the crude product (0.78 g). Half of the crude material (0.39 g) was purified by reverse phase preparative HPLC to give the title compound as a colorless solid (0.20 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.38 (br s, 1H), 9.38 (br s, 1H), 8.07 (s, 1H), 7.79 (d, J=2.1 Hz, 1H), 7.74 (d, J =7.3 Hz, 1H), 7.47 (d, J=11.8 Hz, 1H), 5.45 (s, 1H), 4.52 (s, 2H), 3.61-3.45 (m, 2H), 3.27-3.11 (m, 2H), 3.11-3.00 (m, 1H), 2.43-2.27 (m, 2H), 2.27-2.14 (m, 2H), 1.95-1.83 (m, 1H), 1.48-1.38 (m, 2H), 1.18-1.05 (m, 4H), 0.79-0.72 (m, 2H); MS(ES-) m/z 584.0, 586.0, 588.0 (M-1).
Example 474
Synthesis of 4-((4-((3-chloro-5-cyclopropylpyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluurobenzamide hydrochloride
##STR01225##
[2119] To a mixture of 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide (0.39 g, 0.67 mmol), cyclopropylboronic acid (0.17 g, 2.00 mmol), potassium phosphate tribasic (0.57 g, 2.7 mmol) in dioxane (10 mL) was added tetrakis(triphenylphosphine)palladium(0) (0.08 g, 0.07 mmol). The reaction mixture was degassed by passing a stream of argon through it and then heated at 110.degree. C. in a sealed vial for 16 hours. After cooling to ambient temperature, the reaction mixture was filtered over diatomaceous earth. The filter cake was washed with dichloromethane (50 mL), and the combined filtrate was concentrated in vacuo. The residue was purified by reverse phase HPLC (acetonitrile in water with 0.1% ammonium hydroxide). The combined fractions were adjusted to pH 1-2 with 1 N hydrochloric acid, and extracted with dichloromethane (3.times.30 mL). The orgrmic phase was dried over anhydrous sodium sulfate, filtered, and the filtrate concentrated in vacuo to give the title compound as an off-white solid (0.11 g, 29%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.66 (br s, 1H), 9.22 (br s, 1H), 8.15 (d, J=11.6 Hz, 1H), 7.83 (d, J=2.0 Hz, 1H), 7.78 (d, J=7.5 Hz, 1H), 7.29 (d, J=2.1 Hz, 1H), 5.40 (s, 1H), 4.39 (s, 2H), 3.37-3.00 (m, 5H), 2.70-2.50 (m, 2H), 2.24-2.07 (m, 2H), 2.01-1.88 (m, 1H), 1.80-1.75 (m, 1H), 1.50-1.40 (m, 2H), 1.19-1.03 (m, 4H), 1.00-0.92 (m, 2H), 0.77-0.70 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 548.2, 550.2 (M+1).
Example 475
Synthesis of 5-cyclopropyL-N-(cyclopropylsulfonyl)-4-(((1R,3r,5S)-3-(3,5-dichloropheno- xy)-8-azabicyclo[3.2.1]octan-8-yl) methyl)-2-fluorobenzamide hydrochloride
##STR01226##
[2120] Step 1. Preparation of (1R,3r,5S)-tert-butyl (3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]octan-8-carboxylate
##STR01227##
[2122] To a mixture of (1R,3r,5S)-tert-butyl 3-hydroxy-8-azabicyclo[3.2.1]octan-8-carboxylate (5.00 g, 22.00 mmol) in toluene (50 mL) was added 3,5-dichloroiodobenzene (6.00 g, 22.00 mmol), cesium carbonate (21.50 g, 66.00 mmol), copper(I) iodide (0.63 g, 3.30 mmol), 3,4,7,8-tetramethyl-1,10-phenantroline (1.60 g, 6.60 mmol) and molecular sieves (4 A, 5.00 g). The reaction mixture was degassed by passing a stream of argon through it and then heated at 80.degree. C. in a sealed vial for 120 hours. After cooling to ambient temperature, the reaction mixture was diluted with ethyl acetate (150 mL) and filtered over diatomaceous earth. Concentration of the filtrate in vacuo gave a residue which was purified by flash chromatography (0 to 20% ethyl acetate in hexanes) to afford the title compound as a yellowish oil (3.80 g, 46%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.6.93-6.90 (m, 1H), 6.71-6.69 (m, 2H), 4.59-4.53 (m, 1H), 4.28-4.09 (m, 2H), 2.24-1.85 (m, 8H), 1.45 (s, 9H); MS(ES+) m/z 316.1, 318.1 (M -55).
Step 2. Preparation of (1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]octane
##STR01228##
[2124] Following the procedure as described in Example 470 Step 3, and making variation as required to replace (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-aziabicyclo[3.2.1]octa- ne-8-carboxylate with (1R,3r,5S)-tert-butyl 3-(3,5-dichlorophenoxy)-8-aziabicyclo[3.2.1]octane-8-carboxylate, the title compound was isolated as an off-white solid (2.80 g, quant. yield): MS(ES+) m/z 272.1, 274.1 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-(((1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoate
##STR01229##
[2126] Following the procedure as described in Example 470 step 4, and making variation as required to replace (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with (1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]octane, the title compound was obtained as an off-white foam (4.50 g, 86%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.47 (d, J=7.3 Hz, 1H), 7.32 (d, J=12.1 Hz, 1H), 6.92-6.89 (m, 1H), 6.72-6.69 (m, 2H), 4.54-4.46 (m, 1H), 3.65 (s, 2H), 3.14 (s, 2H), 2.19-2.09 (m, 2H), 2.07-1.99 (m, 4H), 1.95-1.85 (m, 3H), 1.57 (s, 9H), 0.93-0.85 (m, 2H), 0.64-0.58 (m, 2H); MS(ES+) m/z 520.2, 522.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-(((1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]-- octan-8-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01230##
[2128] Following the procedure as described in Example 470 Step 5, and making variation as required to replace tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 5-cyclopropyl-4-(((1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid after trituration with diethyl ether (4.40 g, quant. yield): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.13.43 (br s, 1H), 10.56 (br s, 1H), 7.92 (d, J=11.6 Hz, 1H), 7.49 (d, J=7.3 Hz, 1H), 7.17-7.09 (m, 3H), 4.84-4.78 (m, 1H), 4.48-4.38 (m, 2H), 3.98 (s, 2H), 2.77-2.65 (m, 2H), 2.40-2.02 (m, 7H), 1.07-0.97 (m, 2H), 0.76-0.66 (m, 2H); MS(ES+) m/z 464.1, 466.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1R,3r,5S)-3-(3,5-dichloropheno- xy)-8-azabicyclo[3.2.1]octan-8-yl) methyl)-2-fluorobenzamide hydrochloride
##STR01231##
[2130] A mixture of 5-cyclopropyl-4-(((1R,3r,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoic acid hydrochloride (4.00 g, 8.00 mmol), cyclopropanesulfonamide (1.16 g, 9.60 mmol), N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (1.84 g, 9.60 mmol), and 4-dimethylaminopyridine (3.52 g, 28.80 mmol) in anhydrous dichloromethane (50 mL) and anhydrous tetrahydrofuran (50 mL) was stirred at ambient temperature for 16 hours. Additional cyclopropanesulfonamide (1.16 g, 9.60 mmol), N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (1.84 g, 9.60 mmol), and 4-dimethylaminopyridine (3.52 g, 28.80 mmol) was added and the reaction mixture was stirred for another 16 hours at ambient temperature. The reaction mixture was partitioned between ethyl acetate (300 mL) and 1M aqueous hydrochloric acid solution (20 mL). The organic phase was washed with hydrochloric acid (1 N, 20 mL), water (20 mL) and brine (2.times.10 mL), dried over anhydrous sodium sulfate and filtered. After concentration of the filtrate in vacuo, the obtained solid residue was triturated with acetonitrile (100 mL) and filtered off. The solid was re-dissolved in dichloromethane (50 mL), filtered and concentrated in vacuo. The residue was dryed in vacuo at 40-50.degree. C. provided the title compound as a colorless solid (2.45 g, 51%): .sup.1H NMR (300 MHz, CDCl.sub.3/CDCl.sub.3OD) .delta. 8.42 (d, J=12.3 Hz, 1H), 7.73 (d, J=7.4 Hz, 1H), 6.97 (t, J=1.6, 1H), 6.71 (d, J=1.7 Hz, 2H), 4.73-4.67 (m, 1H), 4.45 (s, 2H), 3.84 (br s, 2H), 3.33-3.21 (m, 2H), 3.12-3.02 (m, 1H), 2.52-2.42 (m, 2H), 2.33-2.21 (m, 2H), 2.18-2.09 (m, 2H), 1.93-1.82 (m, 1H), 1.50-1.42 (m, 2H), 1.19-1.10 (m, 2H), 1.10-1.01 (m, 2H), 0.78-0.70 (m, 2H); MS(ES-) 567.1, 559.0 (M +1).
Example 476
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01232##
[2131] Step 1. Preparation of tert-butyl 4-((5-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate
##STR01233##
[2133] Following the procedure as described in Example 470 step 1, and making variation as required to replace 5-bromo-2,3-dichloropyridine with 2-chloro-5-trifluoromethylpyridine, the title compound was isolated as a colorless solid (3.65 g, quant. yield): MS(ES+) m/z 291.1 (M-55).
Step 2. Preparation of 2-(piperidin-4-yloxy)-5-(trifluoromethyl)pyridine
##STR01234##
[2135] Following the procedure as described in Example 470 step 3, and making variation as required to replace (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate with tert-butyl 4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidine-1-carboxylate, the title compound was obtained as an off-white solid (2.40 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.38 (d, J=0.7 Hz, 1H), 7.75 (dd, J=8.7, 2.4 Hz, 1H), 6.78 (d, J=8.7 Hz, 1H), 6.48 (s, 1H), 5.36-5.22 (m, 1H), 3.30-3.16 (m, 2H), 3.06-2.93 (m, 2H), 2.22-2.10 (m, 2H), 1.97-1.83 (m, 2H); MS(ES+) m/z 247.2 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4 4-((4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzoate
##STR01235##
[2137] Following the procedure as described in Example 470 step 4, and making variation as required to replace (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with 2-(piperidin-4-yloxy)-5-(trifluoromethyl)pyridine, the title compound was obtained as light yellowish oil (2.50 g, quant. yield): MS(ES+) m/z 495.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-2-fluoro-4-((4-((5-(trifluoromethyl)pyridin-2-yl) oxy)piperidin-1-yl)methyl)benzoic acid hydrochloride
##STR01236##
[2139] Following the procedure as described in Example 470 step 5, and making variation as required to replace tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzoate, the title compound was obtained au a colorless solid (1.10 g, 95%): MS(ES+) m/z 439.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01237##
[2141] Following the procedure as described in Example 470 Step 6, and making variations as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 5-cyclopropyl-2-fluoro-4-((4-((5-(trifluoromethyl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzoic acid hydrochloride and cyclopropanesulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.24 g, 33%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.44 (br s, 1H), 9.94 (br s, 1H), 8.40 (s, 1H), 7.80 (dd, J=8.7, 1.9 Hz, 1H), 7.68 (d, J=7.3 Hz, 1H), 7.48 (d, J=11.7 Hz, 1H), 6.83 (d, J=8.7 Hz, 1H), 5.46 (s, 1H), 4.52 (s, 2H), 3.81-3.42 (m, 2H), 3.37 (s, 3H), 3.31-2.88 (m, 2H), 2.44-2.29 (m, 2H), 2.29-2.14 (m, 2H), 1.96-1.84 (m, 1H), 1.16-1.02 (m, 2H), 0.80-0.71 (m, 2H); MS(ES-) m/z 514.2 (M-1).
Example 477
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-((5-(trifluoromethyl- ) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01238##
[2143] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 5-cyclopropyl-2-fluoro-4-((4-((5-(trifluoromethl) pyridin-2-yl)oxy)piperidin-1-yl)methyl)benzoic acid hydrochloride, the title compound was obtained as a colorless solid (0.31 g, 40%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.12.88 (br s, 1H), 9.49 (br s, 1H), 8.40 (s, 1H), 7.80 (dd, J=8.7, 2.1 Hz, 1H), 7.72 (d, J=7.4 Hz, 1H), 7.49 (d, J =11.9 Hz, 1H), 6.83 (d, J=8.7 Hz, 1H), 5.45 (s, 1H) 4.50 (s, 2H), 3.75-3.31 (m, 2H), 3.28-2.83 (m, 3H), 2.46-2.29 (m, 2H), 2.28-2.14 (m, 2H), 1.98-1.83 (m, 1H), 1.47-1.37 (m, 2H), 1.19-1.04 (m, 4H), 0.79-0.71 (m, 2H); MS(ES-) m/z 540.2 (M+1).
Example 478
Synthesis of 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01239##
[2144] Step 1. Preparation of tert-butyl 4-((2-chloro-4-fluorobenzyl)oxy)piperidine-1-carboxylate
##STR01240##
[2146] To a mixture of 1-Boc-4-hydroxypiperidine (2.00 g, 10.00 mmol) in anhydrous dimethylformamide (20 mL) was added sodium hydride (60% dispersion in mineral oil, 0.40 g, 10.10 mmol) at 0.degree. C. The reaction mixture was allowed to warm to ambient temperature and stirred for 1 hour. To this reaction mixture was added 2-chloro-4-fluorobenzylbromide (2.45 g, 11.00 mmol) and tetrabutylammonium iodide (0.37 g, 1.00 mmol) and the reaction mixture was stirred for 48 hours at ambient temperature. After addition of water (20 mL) and dilution with ethyl acetate (200 mL), the organic phase was washed with water (3.times.20 mL), brine (20 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate in vacuo provided the title compound as yellowish oil (3.50 g, quant. yield), which was used without further putificaton: MS(ES+) m/z 288.1, 290.1 (M-55).
Step 2. Preparation of 4-((2-chloro-4-fluorobenzyl)oxy)piperidine
##STR01241##
[2148] Following the procedure as described in Example 470 step 3, and making variation as required to replace (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxyy)-8-azabicyclo[3.2.1]octa- ne-8-carboxylate with tert-butyl 4-((2-chloro-4-fluorobenzyl)oxy)piperidine-1-carboxylate, the title compound was obtained as a yellowish oil (1.70 g, 71%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46 (dd, J=8.4, 6.4 Hz, 1H), 7.08 (dd, J=8.5, 2.6 Hz, 1H), 6.96 (dt, J=8.4, 8.3, 2.6 Hz, 1H), 4.56 (s, 2H), 3.56-3.43 (m, 1H), 3.14-3.04 (m, 2H), 2.68-2.54 (m, 2H), 2.02-1.91 (m, 2H), 1.88-1.79 (m, 1H), 1.58-1.43 (m, 2H).
Step 3. Preparation of tert-butyl 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl) methyl-5-cyclopropyl-2-fluorobenzoate
##STR01242##
[2150] Following the procedure as described in Example 470 step 4, and making variation as required to replace (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with 4-((2-chloro-4-fluorobenzyl)oxy)piperidine, the title compound was obtained as a yellowish oil (3.40 g, 97%): MS(ES+) m/z 492.2, 494.2 (M+1).
Step 4. Preparation of 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01243##
[2152] To a mixture of tert-butyl 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl)methyl)-5-cyclopropyl-- 2-fluorobenzoate (3.41 g, 6.90 mmol) na dichloromethane (40 mL) was added trifluoroacetic acid (5 mL) and the reaction mixture was stirred at ambient temperature for 16 hours. Concentration of the reaction mixture in vacuo provided the title compound as an off-white solid (3.20 g, 84%): MS(ES+) m/z 436.1, 438.1 (M+1).
Step 5. Preparation of 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01244##
[2154] Following the procedure as described in Example 470 step 6, and making variations as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((4-((2-chloro-4-fluorobenzyl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace cyclopropanesulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.52 g, 53%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.46 (br s, 1H), 9.97 (br s, 1H), 7.68 (d, J=7.4 Hz, 1H), 7.43 (d, J=11.9 Hz, 1H), 7.37 (dd, J=8.4, 6.3 Hz, 1H), 7.13 (dd, J=8.4, 2.3 Hz, 1H), 6.99 (dt, J=8.3, 8.3, 2.4 Hz, 1H), 4.52 (s, 2H), 4.46 (s, 2H), 3.85 (br s, 1H), 3.50-3.40 (m, 2H) 3.37 (s, 3H), 3.25-3.09 (m, 2H), 2.24-2.02 (m, 4H), 1.91-1.80 (m, 1H), 1.12-1.03 (m, 2H), 0.77-0.69 (m, 2H); MS(ES+) m/z 513.2, 515.2 (M+1).
Example 479
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1- -yl) methyl)-5-cyclopropyl-2-fluorobenzamide, triflouroacetic acid salt
##STR01245##
[2156] Following the procedure as described in Example 470 step 6, and making variations as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-6-(trifluoromethyl) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((4-((2-chloro-4-fluorobenzyl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt and to replace cyclopropanesulfonamide with azetidine-1-sulfonamide, the title compound was obtained as a colorless solid (0.18 g, 17%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.13.04 (br s, 1H), 9.04 (br s, 1H), 7.75 (d, J=7.5 Hz, 1H), 7.52 (d, J=12.1 Hz, 1H), 7.37 (dd, J=8.4, 6.2 Hz, 1H), 7.13 (dd, J=8.4, 2.5 Hz, 1H), 6.99 (dt, J=8.3, 8.3, 2.6 Hz, 1H), 4.53 (s, 2H), 4.43 (s, 2H), 4.23 (t, J=7.7, 7.7 Hz, 4H), 3.84 (br s, 1H), 3.48-3.33 (m, 2H), 3.22-3.05 (m, 2H), 2.34-2.14 (m, 4H), 2.13-2.01 (m, 3H), 1.13-1.04 (m, 2H), 0.78-0.71 (m, 2H); MS(ES+) m/z 554.2, 556.2 (M+1).
Example 480
Synthesis of 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01246##
[2158] Following the procedure described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((4-((2-chloro-4-fluorobenzyl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid 2,2,2-trifluoroacetate, the title compound was obtained as a colorless solid (0.62 g, 60%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.12.13 (br s, 1H), 9.34 (br s, 1H), 7.72 (d, J=7.4 Hz, 1H), 7.46 (d, J=11.9 Hz, 1H), 7.37 (dd, J=8.5, 6.1 Hz, 1H), 7.13 (dd, J=8.4, 2.4 Hz, 1H), 6.99 (dt, J=8.3, 8.3, 2.6 Hz, 1H), 4.52 (s, 2H), 4.45 (s, 2H), 3.85 (s, 1H), 3.50-3.37 (m, 2H), 3.22-2.99 (m, 3H), 2.28-2.01 (m, 4H), 1.92-1.80 (m, 1H), 1.48-1.39 (m, 2H), 1.19-1.03 (m, 4H), 0.77-0.70 (m, 2H); MS(ES+) m/z 539.2, 541.1 (M+1).
Example 481
Synthesis of 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01247##
[2159] Step 1. Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)-3-methylazet- idine-1-carboxylate
##STR01248##
[2161] Following the procedure as described in Example 3 step 1, and making as required to (R)-tert-butyl 3-hydroxypiperidine-1-carboxlate with 1-Boc-3-(hydroxymethyl)-3-methylazetidine and purification by flash chromatography (0 to 40% ethyl acetate in hexanes), the title compound was obtained as a yellowish oil (12.40 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.86 (d, J=7.6 Hz, 1H), 6.62 (d, J=11.9 Hz, 1H), 3.95 (s, 2H), 3.90 (d, J=8.5 Hz, 2H), 3.66 (d, J=8.5 Hz, 2H), 1.56 (s, 9H), 1.42 (s, 9H), 1.41 (s, 3H); MS(ES+) m/z 430.1, 432.1 (M+1).
Step 2. Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-3-methylazetidine-1-carboxylate
##STR01249##
[2163] Following the procedure as described in Example 3 step 2, and making variation as required to (R)-tert-butyl 3-(4-tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)piperidine- 1-carboxylate with tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)-3-methylazet- idine-1-carboxylate, the title compound was obtained as a brownish gum (10.80 g, 86%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.40 (d, J=8.3 Hz, 1H), 6.50 (d, J=12.4 Hz, 1H), 3.95 (d, J=8.4 Hz, 2H), 3.90 (s, 2H), 3.64 (d, J =8.4 Hz, 2H), 1.98-1.87 (m, 1H), 1.55 (s, 9H), 1.41 (s, 9H), 1.40 (s, 3H), 0.91-0.82 (m, 2H), 0.62-0.56 (m, 2H).
Step 3. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride
##STR01250##
[2165] Thionyl chloride (9.0 mL) was added slowly methanol (200 mL) at 0.degree. C. and the mixture was stirred for 1 hour at 0.degree. C. A solution of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluoromethoxy) methyl)-3-methylazetidine-1-carboxylate (10.80 g, 24.80 mmol) in methanol (20 mL) was then added and the reaction mixture was heated under reflux for 6 hours. After cooling to ambient temperature, the reaction mixture was stirred for 16 hours and then concentrated in vacuo. The residue was co-evaporated with toluene (3.times.10 mL) and triturated in hexanes (20 mL) to provide the title compound as a brownish gum (8.20 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.9.98 (br s, 1H), 9.79 (br s, 1H), 7.47 (d, J=8.2 Hz, 1H), 6.57 (d, J=12.2 Hz, 1H), 4.26-4.15 (m, 2H), 4.01 (s, 2H), 3.86 (s, 3H), 3.86-3.79 (m, 2H), 2.11-2.01 (m, 1H), 1.55 (s, 3H), 1.01-0.93 (m, 2H), 0.62-0.56 (m, 2H); MS(ES+) m/z 294.2 (M+1).
Step 4. Preparation of methyl 4-((1-(3-chloro-5-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoa- te
##STR01251##
[2167] To a mixture of methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride (0.60 g, 1.82 mmol) in toluene (10 mL) was added 2-bromo-3-chloro-5-(trifluoromethyl) pyridine (0.71 g, 2.73 mmol), cesium carbonate (1.78 g, 5.46 mmol), bis(dibenzylideneacetone)palladium(0) (0.21 g, 0.30 mmol) and 2,2'-bis(diphenyl-phosphino)-1,1'-binaphthalene (0.22 g, 0.36 mmol). The reaction mixture was degassed by passing a stream of argon through it and then heated at 110.degree. C. in a sealed vial for 16 hours. After cooling to ambient temperature, the reaction mixture was filtered over diatomaceous earth. The filter cake was washed with ethyl acetate (50 mL) and the combined filtrate was concentrated in vacuo. Purification of the residue by purification by flash chromatography (0 to 25% ethyl acetate in hexanes) provided the title compound as an orange gum (0.60 g, 70%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.24-8.22 (m, 1H), 7.56 (d, J=2.0 Hz, 1H), 7.45 (d, J=8.3 Hz, 1H), 6.56 (d, J=12.5 Hz, 1H), 4.39 (d, J=9.0 Hz, 2H), 4.08 (d, J=9.0 Hz, 2H), 4.00 (s, 2H), 3.86 (s, 3H), 1.88-1.74 (m, 1H), 1.48 (s, 3H), 0.75-0.68 (m, 2H), 0.59-0.53 (m, 2H).
Step 5. Preparation of 4-((1-(3-chloro-5-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoi- c acid
##STR01252##
[2169] To a mixture of methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (0.60 g, 1.27 mmol) in tetrahydrofuran (5 mL) was added a solution of lithium hydroxide monohydrate (0.29 g, 6.95 mmol) in water (5 mL) and the reaction mixture was heated at 80.degree. C. for 3 hours. After cooling to ambient temperature, the reaction mixture was partitioned between dichloromethane (50 mL) and 1 M aqueous hydrochloric acid solution (10 mL). The organic phase was dried over anhydrous sodium sulfate, filtered, and the filtrate concentrated in vacuo to provide the title compound as an off-white solid (0.58 g, 99%): MS(ES+) m/z 459.0, 461.0 (M+1).
Step 6. Preparation of 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01253##
[2171] Following the procedure as described in Example 470 Step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((1-(3-chloro-5-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoi- c acid and purification by flash chromatography (0 to 50% ethyl acetate containing 0.1% trifluoroacetic acid in hexanes), the title compound was obtained as an off-white solid (0.19 g, 54%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.85 (br s, 1H), 8.40-8.38 (m, 1H), 7.99 (d, J=1.9 Hz, 1H), 7.12 (d, J=8.3 Hz, 1H), 7.01 (d, J=12.9 Hz, 1H), 4.35 (d, J=9.0 Hz, 2H), 4.12 (s, 2H), 4.06 (d, J=9.0 Hz, 2H), 3.12-3.02 (m, 1H), 1.80-1.69 (m, 1H), 1.43 (s, 3H), 1.15-1.07 (m, 4H), 0.64-0.58 (m, 4H); MS(ES-) m/z 560.1, 562.1 (M-1).
Example 482
Synthesis of 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
##STR01254##
[2173] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((1-(3-chloro-5-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoi- c acid and cyclopropanesulfonamide with ethanesulfonamide and purification by flash chromatography (0 to 50% ethyl acetate containing 0.1% trifluoracetic acid in hexanes), the title compound was obtained as an off-white solid (0.19 g, 56%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.83 (br s, 1H), 8.40-8.38 (m, 1H), 7.99 (d, J=2.1 Hz, 1H), 7.12 (d, J=8.3 Hz, 1H), 7.00 (d, J=12.8 Hz, 1H), 4.35 (d, J=9.0 Hz, 2H), 4.12 (s, 2H), 4.06 (d, J=9.0 Hz, 2H), 3.46 (d, J=7.3 Hz, 2H), 1.81-1.68 (m, 1H), 1.43 (s, 3H), 1.24 (t, J=7.3 Hz, 3H), 0.64-0.58 (m, 4H); MS(ES-) m/z 548.1, 550.1 (M-1).
Example 483
Synthesis of 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01255##
[2174] Step 1. Preparation of methyl 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01256##
[2176] Following the procedure as described in Example 481 step 4, and making variation as required to replace 2-bromo-3-chloro-5-(trifluoromethyl)pyridine with 2-bromo-5-chloro-3-fluoropyridine, the title compound was obtained as an orange gum (0.43 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.87-7.85 (m, 1H), 7.44 (d, J=8.3 Hz, 1H), 7.16 (d, J=11.2 Hz, 1H), 6.56 (d, J=12.5 Hz, 1H), 4.18 (d, J =8.3 Hz, 2H), 4.01 (s, 2H), 3.89 (d, J=7.9 Hz, 2H), 3.86 (s, 3H), 1.87-1.75 (m, 1H), 1.48 (s, 1H), 0.77-0.68 (m, 2H), 0.60-0.53 (m, 2H).
Step 2. Preparation of 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01257##
[2178] Following the procedure as described in Example Example 481 step 5, and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as an off-white solid (0.41 g, 98%): MS(ES-) m/z 409.0, 411.0 (M-1).
Step 3. Preparation of 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01258##
[2180] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and purification by flash chromatography (0 to 50% ethyl acetate containing 0.1% trifluoroacetic acid in hexanes), the title compound was obtained as an off-white solid (0.13 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.85 (s, 1H), 7.97 (dd, J=2.0, 0.6 Hz, 1H), 7.74 (dd, J=11.8, 2.1 Hz, 1H), 7.12 (d, J=8.3 Hz, 1H), 7.00 (d, J=12.9 Hz, 1H), 4.16-4.10 (m, 4H), 3.85 (dd, J=8.3, 1.2 Hz, 2H), 3.12-3.02 (m, 1H), 1.80-1.69 (m, 1H), 1.43 (s, 3H), 1.16-1.07 (m, 4H), 0.68-0.58 (m, 4H); MS(ES-) m/z 510.1, 512.1 (M-1).
Example 484
Synthesis of 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(ethylsulfonyl)-2-fluorobenzamide
##STR01259##
[2182] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with 4-((1-(5-chloro-3-fluoropyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and cyclopropanesulfonamide with ethanesulfonamide, the title compound was obtained as an off-white solid (0.08 g, 30%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.83 (br s, 1H), 7.97 (d, J=1.9 Hz, 1H), 7.74 (dd, J=11.8, 2.0 Hz, 1H), 7.12 (d, J=8.3 Hz, 1H), 7.00 (d, J=12.9 Hz, 1H), 4.17-4.09 (m, 4H), 3.85 (dd, J=8.3, 1.2 Hz, 2H), 3.46 (q, J=7.4, 7.3, 7.3 Hz, 2H), 1.81-1.70 (m, 1H), 1.43 (s, 3H), 1.24 (t, J=7.3 Hz, 3H), 0.68-0.58 (m, 4H); MS(ES-) m/z 498.1, 500.1 (M-1).
Example 485
Synthesis of 4-((1-(5-chloro-6-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01260##
[2183] Step 1. Preparation of methyl 4-((1-(5-chloro-6-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01261##
[2185] Following the procedure as described in Example 481 step 4, and making variation as required to replace 2-bromo-3-chloro-5-(trifluoromethyl)pyridine with 3,6-dichloro-2-(trifluoromethyl)pyridine, the title compound was obtained as an orange gum (0.66 g, 51%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.48 (d, J=8.8 Hz, 1H), 7.43 (d, J=8.3 Hz, 1H), 6.55 (d, J=12.4 Hz, 1H), 6.36 (d, J=8.8 Hz, 1H), 4.10 (d, J=8.2 Hz, 2H), 3.99 (s, 2H), 3.86 (s, 3H), 3.81 (d, J=8.2 Hz, 2H), 1.82-1.70 (m, 1H), 1.50 (s, 3H), 0.74-0.65 (m, 2H), 0.58-0.52 (m, 2H).
Step 2. Preparation of 4-((1-(5-chloro-6-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01262##
[2187] Following the procedure as described in Example 481 Step 5, and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 4-((1-(5-chloro-6-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as an off-white solid (0.63 g, 99%): MS(ES+) m/z 459.0, 461.0 (M+1).
Step 3. Preparation of 4-((1-(5-chloro-6-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01263##
[2189] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with -((1-(5-chloro-6-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoi- c acid and purification by flash chromatography (0 to 50% ethyl acetate containing 0.1% trifluoroacetic acid in hexanes), the title compound was obtained as a colorless solid (0.11 g, 28%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.11.85 (br s, 1H), 7.81 (d, J=8.9 Hz, 1H), 7.10 (d, J=8.3 Hz, 1H), 6.99 (d, J=12.9 Hz, 1H), 6.78-6.68 (m, 1H), 4.10 (s, 2H), 4.05 (d, J=8.5 Hz, 2H), 3.81 (d, J=8.5 Hz, 2H), 3.13-3.00 (m, 1H), 1.72-1.61 (m, 1H), 1.44 (s, 3H), 1.15-1.06 (m, 4H), 0.62-0.55 (m, 4H); MS(ES-) m/z 560.1, 562.1 (M-1).
Example 486 and Example 487
Synthesis of 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-3-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01264##
[2190] And 4-((2-(2-chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetid- in-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01265##
[2191] Step 1. Prepapation of methyl 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate and methyl 4-((1-(2-chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01266##
[2193] Following the procedure as described in Example 481 Step 4, and making variation as required to replace 2-bromo-3-chloro-5-(trifluoromethyl)pyridine with 2,4-dichloro-5-(trifluoromethyl)pyridine, a 2.5:1 mixture of the title compounds was obtained as an orange gum (0.40 g, 31%). Major isomer: .sup.19F NMR (282 MHz, CDCl.sub.3) .delta. -55.5 (s, 3F), -108.8 (s, 1F). Minor isomer: .sup.19F NMR (2.82 MHz, CDCl.sub.3) .delta. -60.5 (s, 3F), -108.9 (s, 1F).
Step 2. Prepartion of 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid and 4-((1-(2-chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01267##
[2195] Following the procedure as described in Example 481 Step 5, and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethlyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with a mixture of methyl 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate and methyl 4-((1-(2-chloro-5-trifluoromethyl) pyridin-4-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoa- te, a mixture of the title compounds was obtained as a yellowish solid (0.33 g, 76%): MS(ES+) m/z 459.1, 461.1 (M+1).
Step 3. Preparation of 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01268##
[2196] And 4-((1-(2-chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetid- in-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01269##
[2198] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with a mixture of 4-((1-(4-chloro-5-(trifluoromethyl) pyridin-2-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoi- c acid and 4-((1-(2-chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetid- in-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the compounds were obtained after reverse phase HPLC purification: the first fraction is 4-((1-(2-Chloro-5-(trifluoromethyl)pyridin-4-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt (0.07 g, 18%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.86 (br s, 1H), 8.27 (s, 1H), 7.14 (d, J=8.3 Hz, 1H), 7.02 (d, J=12.9 Hz, 1H), 6.58 (s, 1H), 4.19 (d, J=8.9 Hz, 2H), 4.14 (s, 2H), 3.96 (d, J=8.9 Hz, 2H), 3.13-3.02 (m, 1H), 1.88-1.76 (m, 1H), 1.42 (s, 3H), 1.16-1.07 (m, 4H), 0.66-0.58 (m, 4H); MS(ES+) m/z 562.1, 564.1 (M+1). The second fraction is 4-((1-(4-Chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt (0.19 g, 46%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.85 (s, 1H), 8.39 (s, 1H), 7.11 (d, J=8.3 Hz, 1H), 7.01 (d, J=12.9 Hz, 1H), 6.75 (s, 1H), 4.14-4.07 (m, 4H), 3.86 (d, J=8.9 Hz, 2H), 3.12-3.02 (m, 1H), 1.76-1.64 (m, 1H), 1.44 (s, 3H), 1.14-1.07 (m, 4H), 0.64-0.58 (m, 4H); MS(ES+) m/z 562.1, 564.1 (M+1).
Example 488
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((3-methyl-1-(5-(trifluo- romethyl)pyridin-2-yl) azetidin-3-yl)methoxy)benzamide, trifluoroacetic acid salt
##STR01270##
[2200] To a mixture of 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluorcacetic acid salt (0.10 g, 0.18 mmol) in ethyl acetate (5 mL) and acetic acid (1mL) was added palladium on carbon (10 wt %, wet, 50 mg) and the reaction mixture was stirred under an atmosphere of hydrogen the 16 hours. The reaction mixture was filtered over diatomaceous earth, the filter cake washed with ethyl acetate (20 mL), and the combined filtrate concentrated in vacuo. Purification of the residue by reverse phase HPLC provided the title compound as a colorless solid (0.02 g, 16%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 8.67 (d, J=16.3 Hz, 1H), 8.43(s, 1H), 7.80 (d, J=8.2 Hz, 1H), 7.61 (d, J=9.1 Hz, 1H), 6.61 (d, J=13.3 Hz, 1H), 6.51 (d, J=8.8 Hz, 1H), 4.41 (d, J=8.7 Hz, 2H), 4.12 (d, J=8.8 Hz, 2H), 4.07 (s, 2H), 3.14-3.02 (m, 1H), 1.86-1.73 (m, 1H), 1.57 (s, 3H), 1.48-1.40 (m, 2H), 1.19-1.09 (m, 2H), 0.78-0.70 (m, 2H), 0.63-0.55 (m, 2H); MS(ES+) m/z 528.0 (M+1).
Example 489
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((3-methyl-1-(3-(trifluo- romethyl) pyridin-4-yl)azetidin-3-yl)methoxy)benzamide, trifluoroacetic acid salt (
##STR01271##
[2202] Following the procedure as described in Example 488, and making variation as required to replace 4-((1-(4-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt with 4-((1-(2-chloro-5-trifluoromethyl) pyridin-4-yl)-3-methylazetidin-3-yl)methoxy)-5-cyclopropyl-N-(cyclopropyl- sulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt, the title compound was obtained as a colorless solid (0.01 g, 33%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 8.79-8.55 (m, 2H), 8.36 (br s, 1H), 7.63 (d, J=8.9 Hz, 1H), 6.62. (d, J=13.7 Hz, 1H), 6.53 (br s, 1H), 4.53 (br s, 2H), 4.19 (br s, 2H), 4.07 (s, 2H), 3.13-3.00 (m, 1H), 1.84-1.72 (m, 1H), 1.59 (s, 3H), 1.48-1.38 (m, 2H), 1.17-1.08 (m, 2H), 0.77-0.68 (m, 2H), 0.62-0.54 (m, 2H); MS(ES+) m/z 528.1 (M+1).
Example 490
Synthesis of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(5-(trifluoromethyl)p- yridin-2-yl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01272##
[2203] Step 1. Preparation of (R) methyl 5-cyclopropyl-2-fluoro-4-((1-(5-(trifluoromethyl) pyridin-2-yl)piperidin-3-yl)oxy)benzoate
##STR01273##
[2205] Following the procedure as described in Example 481 Step 4 and making variation as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate and 2-bromo-3-chloro-5-(trifluoromethyl)pyridine with 2-chloro-5-(trifluoromethyl)pyridine, the title compound was obtained as a yellowish oil (0.58 g, 55%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.35-8.33 (m, 1H), 7.56 (dd, J=9.1, 2.5 Hz, 1H), 7.36 (d, J=8.3 Hz, 1H), 6.74 (d, J=12.9 Hz, 1H), 6.61 (d, J=9.0 Hz, 1H), 4.47-4.38 (m, 1H), 4.17 (dd, J=13.5, 2.7 Hz, 1H), 3.86 (s, 3H), 3.80-3.64 (m, 3H), 2.18-2.05 (m, 1H), 2.04-1.87 (m, 2H), 1.78-1.57 (m, 2H), 0.74-0.67 (m, 2H), 0.55-0.46 (m, 2H); MS(ES+) m/z 439.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(5-(trifluoromethl) pyridin-2-yl)piperidin-3-yl)oxy)benzoic acid
##STR01274##
[2207] Following the procedure as described in Example 481 Step 5 and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with (R)-methyl 5-cyclopropyl-2-fluoro-4-((1-(5-(trifluoromethyl) pyridin-2-yl)piperidin-3-yl)oxy)benzoate, the title compound was obtained as an orange gum (0.55 g, quant. yield): MS(ES+) m/z 425.1 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(5-(trifluoromethyl) pyridin-2-yl)piperidin-3-yl)oxy)benzanmde, trifluoroacetic acid salt
##STR01275##
[2209] Following the procedure as described in Example 470 Step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-2-fluoro-4-((1-(5-(trifluoromethyl) pyridin-2-yl)piperidin-3-yl)oxy)benzoic acid and cyclopropanesulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.16 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.10.59 (br s, 1H), 8.86-8.71 (m, 1H), 8.37-8.34 (m, 1H), 7.73 (dd, J=9.3, 2.3 Hz, 1H), 7.47 (d, J=9.0 Hz, 1H), 6.83 (d, J=9.3 Hz, 1H), 6.76-6.69 (m, 1H), 4.65-4.56 (m, 1H), 4.10 (dd, J=14.0, 5.3 Hz, 1H), 4.02 (dd, J=14.0, 2.7 Hz, 1H), 3.90 (ddd, J=13.5, 5.9, 3.2 Hz, 1H), 3.63 (ddd, J=13.7, 8.6, 3.4 Hz, 1H), 3.39 (s, 3H), 2.15-1.94 (m, 3H), 1.81-1.60 (m, 2H), 0.78-0.61 (m, 2H), 0.58-0.41 (m, 2H); MS(ES-) m/z 500.2 (M-1).
Example 491
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(5-(trifluoromet- hyl)pyridin-2-yl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01276##
[2211] Following the proocedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-2-fluoro-4-((1-(5-(trifluoromethyl) pyridin-2-yl)piperidin-3-yl)oxy)benzoic acid, the title compound was obtained as a colorless solid (0.16 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta.9.89 (br s, 1H), 8.75 (d, J=15.1 Hz, 1H), 7.72 (dd, J=9.3, 2.3 Hz, 1H), 7.48 (d, J=9.0 Hz, 1H), 6.82 (d, J=9.3 Hz, 1H), 6.73 (d, J=14.2 Hz, 1H), 4.64-4.53 (m, 1H), 4.12-3.99 (m, 2H), 3.89 (ddd, J=13.4, 6.3, 3.3 Hz, 1H), 3.63 (ddd, J=13.7, 8.2, 3.3 Hz, 1H), 3.07 (tt, J=8.1, 8.1, 4.8, 4.8 Hz, 1H), 2.15-1.92 (m, 3H), 1.80-1.60 (m, 3H), 1.47-1.38 (m, 2H), 1.18-1.08 (m, 2H), 0.79-0.61 (m, 2H), 0.58-0.43 (m, 2H); MS(ES-) m/z 526.2 (M-1).
Example 492
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(pyridin-2-yl)pi- peridin-3-yl)oxy)benzamide
##STR01277##
[2212] Step 1. Preparation of (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl)piperidin-3-yl)oxy)benzoate
##STR01278##
[2214] Following the procedure as described in Example 481 Step 4 and making variations as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate, and to replace 2-bromo-3-chloro-5-(trifluoromethyl)pyridine with 2-bromopyridine, and to replace cesium carbonate with potassium tert-butoxide, the title compound was obtained as a yellowish oil (0.27 g, 32%): MS(ES+) m/z 413.3 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl)piperidin-3-yl)oxy)benzoic acid
##STR01279##
[2216] Following the procedure as described in Example 478 step 4, and making variation as required to replace tert-butyl 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl)methyl-5-cyclopropyl-2- -fluorobenzoate with (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl)piperidin-3-yl)oxy)benzoate, the title compound was obtained as a yellowish oil (0.23 g, quant. yield): MS(ES+) m/z 357.2 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(pyridin-2-yl) piperidin-3-yl)oxy)benzamide
##STR01280##
[2218] Following the procedure as described in Example 470 Step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl) piperidin-3-yl)oxy)benzoic acid and purification by flash chromatography (0-100% ethyl acetate in hexanes), the title compound was obtained as a colorless solid (0.03 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.71 (s, 1H), 8.15 (ddd, J=5.0, 1.9, 0.6 Hz, 1H), 7.52 (d, J=9.1 Hz, 1H), 7.44 (ddd, J=8.9, 7.1 Hz, 1H), 6.95 (d, J=15.0 Hz, 1H), 6.65 (d, J=8.6 Hz, 1H), 6.59 (dd, J=7.1, 5.0 Hz, 1H), 4.46-4.32 (m, 2H), 3.81-3.71 (m, 1H), 3.45-3.32 (m, 2H), 3.13-3.01 (m, 1H), 2.23-2.11 (m, 1H), 1.97-1.81 (m, 3H), 1.70-1.60 (m, 1H), 1.47-1.39 (m, 2H), 1.16-1.08 (m, 2H), 0.85-0.76 (m, 2H), 0.60-0.53 (m, 2H); MS(ES-) m/z 458.3 (M-1).
Example 493
Synthesis of (R)-N-(azetidin-1-ylsulfonyl)-5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl)- piperidin-3-yl)oxy)benzamide
##STR01281##
[2220] Following the procedore as described in Example 470 Step 6, and making variations as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-2-fluoro-4-((1-(pyridin-2-yl) piperidin-3-yl)oxy)benzoic acid, and to replace cyclopropanesulfonamide with azetidine-1-sulfonamide and purification by flash chromatography (0-100% ethyl acetate in hexanes), the title compound was obtained as colorless solid (0.04 g, 28%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.8.67 (d, J=14.1 Hz, 1H), 8.15 (dd, J=4.9, 1.8 Hz, 1H), 7.53 (d, J=9.1 Hz, 1H), 7.44 (ddd, J=8.8, 7.2, 1.9 Hz, 1H), 6.96 (d, J=14.7 Hz, 1H), 6.65 (d, J=8.6 Hz, 1H), 6.59 (dd, J=7.1, 5.0 Hz, 1H), 4.46-4.33 (m, 2H), 4.23 (t, J=8.1, 8.1 Hz, 4H), 3.81-3.71 (m, 1H), 3.45-3.33 (m, 2H), 2.31-2.12 (m, 3H), 1.97-1.82 (m, 3H), 1.72-1.60 (m, 1H), 0.85-0.77 (m, 2H), 0.62-0.55 (m, 2H); MS(ES-) m/z 473.3 (M-1).
Example 494
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((1-(5-fluoropyridin- -2-yl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01282##
[2221] Step 1. Preparation of (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoropyridin-2-yl) piperidin-3-yl)oxy)benzoate
##STR01283##
[2223] Following the procedure as described in Example 481 step 4 and making variation as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate, 2-bromo-3-chloro-5-(trifluoromethyl) pyridine with 2-chloro-5-fluoropyridine, and cesium carbonate with potassium tert-butoxide, the title compound was obtained as a yellowish oil (0.22 g, 26%): MS(ES+) m/z 431.1 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoropyridin-2-yl)piperidin-3-yl)oxy- )benzoic acid
##STR01284##
[2225] Following the procedure as described in Example 478 step 4, and making variation as required to replace tert-butyl 4-((4-((2-chloro-4-fluorobenzyl)oxy)piperidin-1-yl)methyl)-5-cyclopropyl-- 2-fluorobenzoate with (R)-tert-butyl 5-cyclopropyl-2-fluoro-4-((1-(5-fluoropyridin-2-yl)piperidin-3-yl) oxy)benzoate, the title compound was obtained as an orange oil (0.04 g, quant. yield): MS(ES+) m/z 374.9 (M+1).
Step 3. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-((1-(5-fluoropyridin-2- -yl) piperidin-3-yl)oxy)benzamide, trifluoroacetic acid salt
##STR01285##
[2227] Following the procedure as described in Example 470 step 6, and making variation as required to replace 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-2-fluoro-4-((1-(5-fluoropyridin-2-yl) ppieridin-3-yl)oxy)benzoic acid and purification by flash chromatoaraphy (50% ethyl acetate in hexanes, then 0-10% methanol in dichloromethane) and treatment with trifluoroacetic acid, the title compound was obtained as a colorless solid (0.12 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 8.72 (d, J=15.5 Hz, 1H), 8.01 (d, J=2.8 Hz, 1H), 7.51 (d, J=9.1 Hz, 1H), 7.32-7.25 (m, 1H), 6.86 (d, J=14.5 Hz, 1H), 6.66 (dd, J=9.3, 3.2 Hz, 1H), 5.06 (br s, 1H), 4.51-4.42 (m, 1H), 4.19 (dd, J=13.2, 2.0 Hz, 1H), 3.72-3.61 (m, 1H), 3.54-3.39 (m, 2H), 3.15-3.02 (m, 1H), 2.21-2.08 (m, 1H), 2.00-1.78 (m, 3H), 1.75-1.60 (m, 1H), 1.47-1.37 (m, 2H), 1.20-1.08 (m, 2H), 0.84-0.76 (m, 2H), 0.61-0.52 (m, 2H); MS(ES+) m/z 478.2 (M+1).
Example 495
Synthesis of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(2-(3,5-dichlorophenyl)pr- opan-2-yl) piperidin-3-yl)oxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01286##
[2228] Step 1. Preparation of (R)-methyl 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl) piperidin-3-yl)oxy)-2-fluorobenzoate
##STR01287##
[2230] To a mixture of (R)-methyl 5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (1.00 g, 3.41 mmol) in anhydrous tetrahydrofuran (20 mL) was added 4-dimethylaminopyridine (0.08 g, 0.68 mmol), triethylamine (0.95 mL, 6.82 mmol) and 3,5-dichlorobenzoyl chloride (0.86 g, 4.11 mmol). After stirring at ambient temperature for 16 hours, the reaction mixture was diluted with ethyl acetate (200 mL), washed with aqueous hydrochloric acid solution (1 N, 10 mL), water (10 mL), brine (10 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of filtrate in vacuo gave a residue which was purified by flash chromatography (0-100% ethyl acetate in hexanes) to provide (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzoyl)piperidin-3-yl)oxy)-2-fluorobenz- oate as yellowish oil (1.31 g, 82%).
[2231] To a mixture of (R)-methyl 5-cyclopropyl-4-((1-(3,5-dichlorobenzoyl)piperidin-3-yl)oxy)-2-fluorobenz- oate (1.31 g, 2.81 mmol) and 2,6-di-tert-butyl-4-methylpyridine (0.69 g, 3.37 mmol) in anhydrous dichloromethane (40 mL) was added trifluoromethanesulfonic anhydride (0.57 ml, 3.37 mmol) at -78.degree. C. The reaction mixture was stirred for 2 hours at -78.degree. C. after which methyl lithium (1.6 M solution in diethyl ether, 8.8 mL 14.10 mmol) was added to it. After stirring for 1 hour at -78.degree. C., the reaction mixture was warmed to -60.degree. C., and stirred for additional 2 hours. After addition of saturated ammonium chloride solution (20 mL) and dichloromethane (200 mL), the mixture was allowed to warm to ambient temperature. The organic phase was washed with aqueous hydrochloric acid solution (1 N, 10 mL), water (10 mL), brine (10 mL), and dried over anhydrous sodium sulfate. Filtration and concentration of the filtrate in vacuo gave a residue which was purified by flash chromatography (0-30% ethyl acetate in hexanes) to provide the title compound as a colorless oil (0.23 g, 17%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.42-7.37 (m, 3H), 7.19 (t, J=1.9, 1.9 Hz, 1H), 6.42 (d, J=12.9 Hz, 1H), 4.37-4.28 (m, 1H), 3.85 (s, 3H), 2.93-2.85 (m, 1H), 2.67-2.58 (m, 1H), 2.39-2.21 (m, 2H), 2.09-1.97 (m, 2H), 1.86-1.74 (m, 1H), 1.64-1.48 (m, 2H), 1.29 (s, 3H), 1.27 (s, 3H), 0.94-0.82 (m, 2H), 0.66-0.59 (m, 2H); MS(ES+) m/z 480.2, 482.2 (M+1).
Step 2. Preparation of (R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl) piperidin-3-yl)oxy)-2-fluorobenzoic acid
##STR01288##
[2233] Following the procedure as described in Example 481 Step 5 and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with (R)-methyl 5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl) piperidin-3-yl)oxy)-2-fluorobenzoate, the title compound was obtained as a yellowish oil (0.23 g, quant. yield): MS(ES-) m/z 466.2, 468.2 (M-1).
Step 3. Preparation of (R)-5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((1-(2-(3,5-dichlorophenyl) propan-2-yl)piperidin-3-yl)oxy)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01289##
[2235] Following the procedure as described in Example 473 Step 5, and making variation as required to replace 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl)methyl)-5-cyclopr- opyl-2-fluorobenzoic acid hydrochloride with (R)-5-cyclopropyl-4-((1-(2-(3,5-dichlorophenyl)propan-2-yl) piperidin-3-yl)oxy)-2-fluorobenzoic acid, the title compound was as a colorless solid (0.19 g, 67%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.74 (d, J=13.3 Hz, 1H), 7.55 (d, J=1.7 Hz, 2H), 7.53 (d, J=9.0 Hz, 1H), 7.45 (t, J=1.7 Hz, 1H), 6.92 (d, J=14.1 Hz, 1H), 5.15-5.04 (m, 1H), 3.75-3.65 (m, 1H), 3.43-3.35 (m, 1H), 3.11-3.01 (m, 1H), 2.67-2.50 (m, 2H), 2.39-1.89 (m, 4H), 1.89 (s, 3H), 1.86 (s, 3H), 1.60-1.48 (m, 1H), 1.47-1.38 (m, 2H), 1.16-1.08 (m, 2H), 0.93-0.84 (m, 2H), 0.65-0.58 (m, 2H); MS(ES-) m/z 567.2, 569.2 (M-1)
Example 496
Synthesis of 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.- 1]octan-8-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01290##
[2236] Step 1. Preparation of (1R,3r,5S)-tert-butyl 3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.1]octane-8-carb- oxylate
##STR01291##
[2238] Following the procedure as described in Example 470 step 2, and making variation as required to replace 3-chloro-2-fluoro-5-(trifluoromethyl)phenol with 3-chloro-5-(trifluoromethoxy)-phenol, the title compound was obtained as a colorless oil (2.0 t, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.6.82-6.79 (m, 1H), 6.75-6.72 (m, 1H), 6.57-6.53 (m, 1H), 4.60-4.54 (m, 1H), 4.23-4.17 (m, 2H), 2.19-1.87 (m, 8H), 1.46 (s, 9H); MS(ES+) m/z 366.0, 368.0 (M-55).
Step 2. Preparation of (1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.1]oc- tane
##STR01292##
[2240] Following the procedure as described in Example 470 step 3, and making variation as required to replace (1R,3r,5S)-tert-butyl 3-(3-chloro-2-fluoro-5-(trifluoromethoxy) phenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate with (1R,3r,5S)-tert-butyl 3-(3-chloro-5-(trifluoromethoxy-) phhenoxy)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was isolaled as a colorless oil (1.50 g, quant. yield): MS(ES+) m/z 322.1, 324.1 (M+1).
Step 3. Preparation of tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy) phhenoxy)-8-azabicyclo[3.2.1]octane-8-yl)methyl)-5-cyclopropyl-2-fluorobe- nzoate
##STR01293##
[2242] Following the procedure as described in Example 470 Step 4, and making variation as required to replace (1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with (1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.1]-o- ctane, the title compound was isolated as a colorless oil (1.75 g, 65%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.1 Hz, 1H), 7.36-7.28 (m, 1H), 6.81-6.77 (m, 1H), 6.74 (t, J=2.0 Hz, 1H), 6.58-6.53 (m, 1H), 4.58-4.46 (m, 1H), 3.74-3.58 (m, 2H), 3.23-3.05 (m, 2H), 2.23-1.83 (m, 9H), 1.57 (s, 9H), 0.95-0.83 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 570.1, 572.1 (M+1).
Step 4. Preparation of 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenz- oic acid hydrochloride
##STR01294##
[2244] Following the procedure as described in Example 470 Step 5, and making variation as required to replace tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)-phenoxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoate with tert-butyl 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.- 1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was isolated as a colorless solid (1.64 g, 96%): MS(ES+) m/z 516.1, 518.1 (M+1).
Step 5. Preparation of 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy) phenoxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-N-(cycloprop- ylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01295##
[2246] Following the procedure as described in Example 473 step 5, and making variation as required to replace 4-((4-((5-bromo-3-chloropyridin-2-yl)oxy)piperidin-1-yl)methyl)-5-cyclopr- opyl-2-fluorobenzoic acid hydrochloride with (of 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.- 1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, the title compound was as a colorless solid (0.45 g, 62%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.41 (d, J=11.2 Hz, 1H), 6.79 (br s, 1H), 6.74-6.71 (m, 1H), 6.53 (br s, 1H), 4.62-4.55 (m, 1H), 4.37 (s, 2H), 3.88 (br s, 2H), 3.04-2.93 (m, 1H), 2.75-2.62 (m, 2H), 2.49-2.38 (m, 2H), 2.33-2.22 (m, 2H), 2.12 (d, J=15.6 Hz, 2H), 1.85-1.74 (m, 1H), 1.37-1.28 (m, 2H), 1.11-0.94 (m, 4H), 0.71-0.62 (m, 2H); MS(ES+) m/z 617.1, 619.1 (M+1).
Example 497
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(2-(trifluoromethyl)pheno- xy)piperidin-1-yl)methyl)benzamide
##STR01296##
[2247] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(2-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoate
##STR01297##
[2249] Following the procedure as described to Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(2-(trifluoromethyl)phenoxy)piperidine, hydrochloride salt, the title compound was obtained as an oil (1.38 g, 66%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.55 (d, J=8.3 Hz, 1H), 7.50-7.39 (m, 2H), 7.20 (d, J =12.1 Hz, 1H), 6.99-6.90 (m, 2H), 4.58-4.47 (m, 1H), 3.64 (s, 2H), 2.74-2.62 (m, 2H), 2.50-2.35 (m, 2H), 2.03-1.83 (m, 5H), 1.56 (s, 9H), 0.96-0.86 (m, 2H), 0.64-0.56 (m, 2H); MS(ES+) m/z 494.3 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(2-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)benzoic acid, trifluoroacetic acid salt
##STR01298##
[2251] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluorobenzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(2-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzoate, tne title compound was obtained as an colorless solid (1.54 g, quant. yield): MS(ES+) m/z 438.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(2-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide
##STR01299##
[2253] Following the procedure as described in Example 53 step 5, and making variation as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(2-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzoic acid, trifluoroacetic acid salt, and purification by flash chromatography (0-75% methanol+0.4% ammonium hydroxide in dichloromethane), the title compound was obtained as a colorless solid (0.20 g, 53%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.69 (d, J=7.6 Hz, 1H), 7.57 (d, J=7.7 Hz, 1H), 7.51-7.37 (m, 2H), 7.04-6.93 (m, 2H), 6.50 (br s, 1H), 4.77-4.68 (m, 1H), 4.09 (s, 2H), 3.29 (s, 3H), 3.04-2.89 (m, 4H), 2.36-2.22 (m, 2H), 2.12-2.00 (m, 2H), 1.94-1.82 (m, 1H), 1.05-0.97 (m, 2H), 0.70-0.63 (m, 2H); MS(ES+) m/z 515.2 (M+1H).
Example 498
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-(2-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide
##STR01300##
[2255] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(2-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoic acid, trifluoroacetic acid salt the title compound was obtained as a colorless solid (0.22 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.70 (d, J=7.7 Hz, 1H), 7.56 (d, J=7.7 Hz, 1H), 7.50-7.37 (m, 2H), 7.02-6.92 (m, 2H), 6.57 (br s, 1H), 4.73-4.62 (m, 1H), 3.99 (s, 2H), 3.08-2.97 (m, 1H), 2.95-2.76 (m, 4H), 2.27-2.12 (m, 2H), 2.08-1.96 (m, 2H), 1.95-1.84 (m, 1H), 1.45-1.36 (m, 2H), 1.16-1.05 (m, 2H), 1.04-0.94 (m, 2H), 0.70-0.61 (m, 2H); MS(ES+) m/z 541.2 (M+1).
Example 499
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(4-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide
##STR01301##
[2256] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzoate
##STR01302##
[2258] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(4-(trifluoromethyl)phenoxy)piperidine, hydrochloride salt, the title compound was obtained as an oil (1.24 g, 60%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.54-7.45 (m, 3H), 7.18 (d, J=11.8 Hz, 1H), 6.94 (d, J=9.1 Hz, 2H), 4.46-4.34 (m, 1H), 3.65 (s, 2H), 2.79-2.67 (m, 2H), 2.44-2.27 (m, 2H), 2.07-1.90 (m, 3H), 1.89-1.75 (m, 2H), 1.56 (s, 9H), 0.95-0.87 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 494.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(4-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)benzoic acid, 2trifluoroacetic acid
##STR01303##
[2260] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoate, the title compound was obtained a an colorless solid (1.38 g, quant. yield): MS(ES+) m/z 438.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(4-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide
##STR01304##
[2262] Following the procedure as described in Example 53 step 5, and making variation as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(4-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoic acid, trifluoroacetic acid salt, the title compound was obtained as a colorless solid (0.18 g, 48%): .sup.1H NMR (300 mHz, CDCl.sub.3) .delta. 7.69 (d, J=7.6 Hz, 1H), 7.52 (d, J=8.6 Hz, 2H), 7.39 (d, J=13.1 Hz, 1H), 6.94 (d, J=8.6 Hz, 2H), 6.22 (br s, 1H), 4.56-4.43 (m, 1H), 3.88 (s, 2H), 3.37 (s, 3H), 2.90-2.79 (m, 2H), 2.72-2.59 (m, 2H), 2.21-2.08 (m, 2H), 1.99-1.86 (m, 3H), 1.03-0.94 (m, 2H), 0.70-0.62 (m, 2H); MS(ES+) m/z 515.2 (M+1).
Example 500
Synthesis of 5-cyclopropyl-N-cyclopropylsulfonyl)-2-fluoro-4-((4-(4-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide
##STR01305##
[2264] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(4-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoic acid, trifluoroacetic acid salt, the title compound was obtained as a colorless solid (0.20 g, 51%): 7.69 (d, J =7.6 Hz, 1H), 7.53 (d, J=8.6 Hz, 2H), 7.46 (d, J=12.5 Hz, 1H), 6.95 (d, J=8.6 Hz, 2H), 4.67-4.55 (m, 1H), 4.17 (s, 2H), 3.16-2.91 (m, 5H), 2.37-2.18 (m, 2H), 2.11-1.97 (m, 2H), 1.96-1.85 (m, 1H), 1.46-1.37 (m, 2H), 1.18-0.99 (m, 4H), 0.74-0.65 (m, 2H); MS(ES+) m/z 541.2 (M+1).
Example 501
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(3-(trifluoromethyl)pheno- xy) piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01306##
[2265] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(3-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzoate
##STR01307##
[2267] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3-(trifluoromethyl) phenoxy)piperidine, the title compound was obtained as an oil (0.69 g, 79%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.3 Hz, 1H), 7.35 (dd, J=8.0, 8.0 Hz, 1H), 7.22-7.09 (m, 3H), 7.07-7.02 (m, 1H), 4.43-4.34 (m, 1H), 3.67 (s, 2H), 2.79-2.68 (m, 2H), 2.43-2.32 (m, 2H), 2.05-1.92 (m, 3H), 1.89-1.76 (m, 2H), 1.56 (s, 9H), 0.95-0.88 (m, 2H), 0.65-0.58 (m, 2H); MS(ES+) m/z 494.3 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(3-(trifluoromethyl)phenoxy) piperidin-1-yl)methyl)benzoic acid, trifluoroacetic acid salt
##STR01308##
[2269] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(3-(trifluoromethyl)phenoxy) piperidin-1-yl)methyl)benzoate, the title compound was obtained as an colorless solid (0.77 g, quant. yield): MS(ES+) m/z 438.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((4-(3-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01309##
[2271] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(3-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzoic acid trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.06 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 9.49 (br s, 1H), 7.65-7.45 (m, 2H), 7.34-7.22 (m, 4H), 4.92-4.74 (m, 1H), 4.52 (br s, 2H), 3.78-3.49 (m, 2H), 3.33 (s, 3H), 3.27-3.15 (m, 2H), 2.25-1.95 (m, 5H), 1.03-0.95 (m, 2H), 0.81-0.73 (m, 2H); MS(ES+) m/z 515.0 (M+1).
Example 502
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-(3-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01310##
[2273] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(3-(trifluoromethyl)phenoxy)piperidin-1-yl)m- ethyl)benzoic acid trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtabled as a colorless solid (0.10 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 12.78 (br s, 1H), 9.24 (br s, 1H), 7.76 (d, J=7.6 Hz, 1H), 7.63 (d, J=11.9 Hz, 1H), 7.46-7.38 (m, 1H), 7.28-7.21 (m, 1H), 7.14-7.10 0 (m, 1H), 7.07-7.02 (m, 1H), 4.78-4.69 (m, 1H), 4.49 (s, 2H), 3.55-3.42 (m, 2H), 3.25-3.11 (m, 2H), 3.11-3.00 (m, 1H), 2.56-2.38 (m, 2H), 2.23-2.10 (m, 2H), 1.97-1.86 (m, 1H), 1.48-1.40 (m, 2H), 1.19-1.07 (m, 4H), 0.80-0.72 (m, 2H); MS(ES+) m/z 541.0 (M+1).
Example 503
Synthesis of 4-((4-(4-chlorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluoro-N-(m- ethylsulfonyl) benzmide, trifluoroacetic acid salt
##STR01311##
[2274] Step 1. Preparation of tert-butyl 4-((4-(4-chlorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzo- ate
##STR01312##
[2276] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(4-chlorophenoxy)piperidine, the title compound was obtained as an oil (0.85 g, 93%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.4 Hz, 1H), 7.23-7.14 (m, 3H), 6.84-6.78 (m, 2H), 4.33-4.23 (m, 1H), 3.65 (s, 2H), 2.77-2.66 (m, 2H), 2.40-2.28 (m, 2H), 2.03-1.90 (m, 3H), 1.86-1.72 (m, 2H), 1.56 (s, 9H), 0.94-0.87 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 460.3, 462.2 (M+1).
Step 2. Preparation of 4-((4-(4-chlorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzo- ic acid, trifluoroacetic acid salt
##STR01313##
[2278] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 4-((4-(4-chlroophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzo- ate, the title compound was obtained as an colorless solid (0.96 g, quant. yield): MS(ES+) m/z 404.2, 406.2 (M+1).
Step 3. Preparation of 4-((4-(4-chlorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluoro-N-(m- ethylsulfonyl) benzamide, trifluoroacetic acid salt
##STR01314##
[2280] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 4-((4-(4-chlorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.06 g, 11%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.35-7.28 (m, 2), 7.14-7.07 (m, 2H), 6.76-6.69 (m, 2H), 4.51-4.45 (m, 1H), 4.36 (s, 2H), 3.23 (s, 3H), 3.21-3.09 (m, 4H), 2.19-2.07 (m, 2H), 2.05-1.94 (m, 2H), 1.88-1.77 (m, 1H), 1.00-0.92 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 481.1, 483.2 (M+1).
Example 504
Synthesis of 4-((4-(4-chlorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01315##
[2282] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(4-chlorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzo- ic acid, trifluoroacetic acid salt, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.07 g, 12%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.19 (br s, 1H), 9.49 (br s, 1H), 7.49 (d, J=11.1 Hz, 1H), 7.32 (d, J=8.6 Hz, 2H), 7.23 (d, J=7.0 Hz, 1H), 7.01 (d, J=8.5 Hz, 2H), 4.77-4.35 (m, 3H), 3.71-3.16 (m, 4H), 3.09-2.99 (m, 1H), 2.28-1.93 (m, 4H), 1.86-1.67 (m, 1H), 1.15-1.06 (m, 4H), 1.02-0.94 (m, 2H), 0.79-0.72 (m, 2H); MS(ES+) m/z 507.2, 509.1 (M+1).
Example 505
Synthesis of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01316##
[2283] Step 1. Preparation of tert-butyl 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01317##
[2285] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(2-chloro-4-fluorophenoxy)piperidine, the title compound was obtained as an oil (1.42 g, 68%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.3 Hz, 1H), 7.22-7.15 (m, 1H), 6.98-6.80 (m, 3H), 4.33-4.18 (m, 1H), 3.65 (s, 2H), 2.80-2.68 (m, 2H), 2.41-2.26 (m, 2H), 2.02-1.72 (m, 5H), 1.56 (s, 9H), 0.95-0.87 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 478.3, 480.2 (M+1).
Step 2. Preparation of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01318##
[2287] Following the procedure as described in Example 53 step 4, and making variation as required tb replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoate, the title compound was obtained as an colorless solid (1.59 g, quant. yield): MS(ES+) m/z 4.22, 424.1 (M+1).
Step 3. Preparation of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyrl)benzamide, trifluoroacetic acid salt
##STR01319##
[2289] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with -((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid, trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.10 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.19 (br s, 1H), 9.81 (br s, 1H), 7.53-7.41 (m, 2H), 7.31-7.23 (m, 2H), 7.21-7.13 (m, 1H), 4.74-4.38 (m, 3H), 3.77-3.44 (m, 2H), 3.32 (s, 3H), 3.29-3.17 (m, 2H), 2.18-2.03 (m, 3H), 2.00-1.80 (m, 2H), 1.02-0.94 (m, 2H), 0.80-0.72 (m, 2H); MS(ES+) m/z 499.2, 501.1 (M+1).
Example 506
Synthesis of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01320##
[2291] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoic acid, trifluoroacetic acid salt, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.13 g, 29%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.28 (br s, 1H), 9.92 (br s, 1H), 7.56-7.49 (m, 1H), 7.44 (dd, J=8.4, 3.1 Hz, 1H), 7.31-7.22 (m, 2H), 7.21-7.13 (m, 1H), 4.76-4.68 (m, 1H), 4.56 (s, 2H), 3.53-3.33 (m, 2H), 3.31-3.15 (m, 2H), 3.11-3.00 (m, 1H), 2.31-1.97 (m, 4H), 1.91-1.77 (m, 1H), 1.15-1.06 (m, 4H), 1.03-0.94 (m, 2H), 0.81-0.73 (m, 2H); MS(ES+) m/z 525.2, 527.2 (M+1).
Example 507
Synthesis of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl)-5-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01321##
[2292] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenoxy) piperidin-1-yl)methyl)benzoate
##STR01322##
[2294] Following the prccedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(4-fluorophenoxy)piperidine, the title compound was obtained as an oil (1.92 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.5 Hz, 1H), 7.17 (d, J=11.8 Hz, 1H), 6.97-6.88 (m, 2H), 6.86-6.79 (m, 2H), 4.27-4.17 (m, 1H), 3.63 (s, 2H), 2.77-2.67 (m, 2H), 2.36-2.25 (m, 2H), 2.00-1.89 (m, 3H), 1.1.84-1.71 (m2H), 1.55 (s, 9H), 0.94-0.86 (m, 2H), 0.63-0.57 (m, 2H); MS(ES+) m/z 444.2 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenoxy)piperidin-1-yl) methyl)benzoic acid, trifluoroacetic acid salt
##STR01323##
[2296] Following the procedure as described in Example 53 step 4, and making variatin as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenoxy)piperidin-1-yl) methyl)benzoate, the title compound was obtained as an colorless solid (2.43 g, quant. yield): MS(ES+) m/z 388.2 (M+1).
Step 3. Preparation of 4-((4-(2-chloro-4-fluorophenoxy)piperidin-1-yl) methyl-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01324##
[2298] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenoxy) piperidin-1-yl)methyl)benzoic acid, trifluoroacetic acid salt, and purification by preparative HPLC the title compound was obtained as a colorless solid (0.05 g, 12%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.28 (br s, 1H), 9.68 (br s, 1H), 7.51 (d, J=11.2 Hz, 1H), 7.25 (d, J=6.9 Hz, 1H), 7.16-7.06 (m, 2H), 7.05-6.94 (m, 2H), 4.71-4.41 (m, 3H), 4.21-3.72 (m, 4H), 3.33 (s, 3H), 2.26-2.09 (m, 2H), 2.07-1.93 (m, 2H), 1.86-1.70 (m, 1H), 1.04-0.95 (m, 2H), 0.81-0.73 (m, 2H); MS(ES+) m/z 465.2 (M+1).
Example 508
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-(4-fluorophenoxy)pip- eridin-1-yl) methyl)benzamide, trifluoroacetic acid salt
##STR01325##
[2300] Following the procedure as described in Example 53 step 5, and making variations as required replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenoxy)piperidin-1-yl)methyl)benzo- ic acid, trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.06 g, 14%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.26 (br s, 1H), 9.86 (br s, 1H), 7.52 (d, J=11.1 Hz, 1H), 7.23 (d, J=7.2 Hz, 1H), 7.16-7.06 (m, 2H), 7.05-6.95 (m, 2H), 4.73-4.70 (m, 3H), 4.13-3.70 (m, 2H), 3.50-3.31 (m, 2H), 3.11-3.00 (m, 1H), 2.27-1.93 (m, 4H), 1.88-1.70 (m, 1H), 1.15-1.06 (m, 4H), 1.03-0.94 (m, 2H), 0.80-0.72 (m, 2H); MS(ES+) m/z 491.2 (M+1).
Example 509
Synthesis of 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01326##
[2301] Step 1. Preparation of tert-butyl 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoate
##STR01327##
[2303] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3,4-dichlorophenoxy)piperidine, the title compound was obtained as an oil (1.75 g, quant.): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.2 Hz, 1H), 7.26 (d, J=8.9 Hz, 1H), 7.15 (d, J=11.8 Hz, 1 H), 6.97 (d, J=2.7 Hz, 1H), 6.72 (dd, J=8.9, 2.8 Hz, 1H), 4.32-4.21 (m, 1H), 3.63 (s, 2H), 2.74-2.64 (m, 2H), 2.38-2.27 (m, 2H), 2.00-1.88 (m, 3H), 1.84-1.71 (m, 2H), 1.55 (s, 9H), 0.94-0.85 (m, 2H), 0.64-0.56 (m, 2H); MS(ES+) m/z 494.1, 406.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy)piperidin-1-yl) methyl-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01328##
[2305] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate and purification by trituration with diethyl ether, the title compound was obtained as an colorless solid (1.54 g, 69%): MS(ES+) m/z 438.1, 440.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01329##
[2307] Following the procedure as described in Example 53, step 5, and making variation as required to replace cyclopropanesulfonamide with methylsulfonamide and (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid, difluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.40 g, 69%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.28 (br s, 1H), 10.00 (br s, 1H), 7.55-7.47 (m, 2H), 7.32 (d, J=2.4 Hz, 1H), 7.25 (d, J=7.1 Hz, 1H), 7.07-6.98 (m, 1H), 4.82-4.65 (m, 1H), 4.56 (s, 2H), 4.38-3.94 (m, 2H), 3.34 (s, 3H), 3.30-3.18 (m, 2H), 2.24-2.09 (m, 3H), 2.01-1.76 (m, 2H), 1.05-0.94 (m, 2H), 0.80-0.72 (m, 2H); MS(ES+) m/z 515.7, 517.1 (M+1).
Example 510
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(3,4-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01330##
[2309] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,4-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid, trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.45 g, 69%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.28 (br s, 1H), 9.98 (br s, 1H), 7.57-7.47 (m, 2H), 7.36-7.28 (m, 1H), 7.23 (d, J=7.0 Hz, 1H), 7.08-6.94 (m, 1H), 4.85-4.69 (m, 1H), 4.57 (s, 2H), 3.59-3.16 (m, 4H), 3.10-2.98 (m, 1H), 2.26-2.09 (m, 2H), 2.09-1.94 (m, 2H), 1.89-1.71 (m, 1H), 1.15-1.06 (m, 4H), 1.03-0.94 (m, 2H), 0.81-0.72 (m, 2H); MS(ES+) m/z 541.1, 543.1 (M+1).
Example 511
Synthesis of 5-cyclopropyl-4-((4-((3,5-dichlorobenzyl)oxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01331##
[2310] Step 1. Preparation of tert-butyl 4-((3,5 dichlorobenzyl)oxy)piperidin-1-yl)carboxylate
##STR01332##
[2312] Following the procedure as described in Example 478 step 1, and making variation as required to replace 2-chloro-4-fluorobenzylbromide with 3,5-dichlorobenzyl chloride, the title compound was isolated as a yellowish oil (1.29 g, 77%).
Step 2. Preparation of 4-((3,5-dichlorobenzyl)oxy)piperidine, trifluoroacetic acid salt
##STR01333##
[2314] A solution of tert-butyl 4-((3,5-dichlorobenzyl)oxy)piperidine-1-carboxylate (1.98 g, 5.50 mmol) in dichloromethane (50 mL) was treated with trifluoroacetic acid (15 mL). The resulting solution was stirred at ambient temperature for 1 hour and then concentrated to vacuo. The residue was used in the next step without further purification.
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorobenzyl)oxy) piperidin-1-yl)methyl)-4-fluorobenzoate
##STR01334##
[2316] Following the procedure as described in Example 53 step 3, and-making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-((3,5-dichlorobenzyl) oxy)piperidine, trifluoroacetic acid salt, the title compound was obtained as an oil (1.44 g, 51% in 2 steps): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.25-7.19 (m, 3H), 7.16 (d, J=11.8 Hz, 1H), 4.46 (s, 2H), 3.61 (s, 2H), 3.48-3.37 (m, 1H), 2.78-2.68 (m, 2H), 2.27-2.14 (m, 2H), 1.99-1.84 (m, 3H), 1.74-1.60 (m, 2H), 1.56 (s, 9H), 0.94-0.85 (m, 2H), 0.63-0.56 (m, 2H); MS(ES+) m/z 508.2, 510.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorobenzyl)oxy)piperidin-1-yl) methyl)-2-fluorobenzoic acid, trifluoroacetic acid salt
##STR01335##
[2318] Following the procedure as described in Example 53 step 4, and making variation as required to replace (S)-tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoate with tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorobenzyl)oxy)piperidin-1-yl)methyl)-2-flu- orobenzoate, the title compound was obtained as an colorless solid (1.29 g, 81%): MS(ES+) m/z 452.1, 454.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)oxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01336##
[2320] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonamide with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorobenzyl)oxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid, trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.25 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.26 (br s, 1H), 9.63 (br s, 1H), 7.54-7.45 (m, 2H), 7.42-7.32 (m, 2H), 7.24 (d, J=7.3 Hz, 3H), 4.51 (br s, 4H), 3.81-3.66 (m, 1H), 3.34 (s, 3H), 3.30-3.20 (m, 2H), 3.18-3.07 (m, 1H), 2.24-1.97 (m, 3H), 1.93-1.80 (m, 2H), 1.73-1.54 (m, 1H), 1.02-0.93 (m, 2H), 0.80-0.71 (m, 2H); MS(ES+) m/z 529.1, 531.1 (M+1).
Example 512
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5-dichlorobenzyl)oxy) piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01337##
[2322] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)oxy)piperidin-1-yl)methyl)-2-fluo- robenzoic acid, trifluoroacetic acid salt and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.31 g, 61%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 9.53 (br s, 1H), 7.53-7.45 (m, 2H), 7.42-7.33 (m, 2H), 7.23 (d, J=7.3 Hz, 1H), 4.51 (br s, 3H), 3.79-3.66 (m, 1H), 3.64-3.49 (m, 1H), 3.42-3.12 (m, 3H), 3.11-3.00 (m, 1H), 2.21-1.98 (m, 3H), 1.93-1.79 (m, 1H), 1.53 (m, 1H), 1.16-1.06 (m, 4H), 1.01-0.93 (m, 2H), 0.79-0.71 (m, 2H); MS(ES+) m/z 555.2, 557.2 (M+1).
Example 513
Synthesis of 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl)piperidin-1- -yl) methyl)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01338##
[2323] Step 1. Preparation of (4-(4-fluorophenyl)piperidin-4-yl)methanol, trifluoroacetic acid salt
##STR01339##
[2325] To a solution of tert-butyl 4-(4-fluorophenyl)-4-(hydroxymethyl)piperidine-1-carboxylate (1.51 g, 4.88 mmol) in dichloromethane (50 mL) was added trifluoracetic acid (15 mL). The resulting solution was stirred at ambient temperature for 1.5 hours and then concentrated in vacuo to provided the title compound (1.58 g, quant. yield): MS(ES+) m/z 210.2 (M+1).
Step 2. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(hydroxymethyl) piperidin-1-yl)methyl)benzoate
##STR01340##
[2327] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with (4-(4-fluorophenyl) piperidin-4-yl)methanol, trifluoroacetic acid salt, the title compound was obtained as an oil (0.78 g, 35%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.45 (d, J=7.3 Hz, 1H), 7.33-7.26 (m, 2H), 7.17 (d, J=11.9 Hz, 1H), 7.12-7.00 (m, 2H), 4.19-4.11 (m, 1H), 3.60-3.50 (m, 4H), 3.32-3.21 (m, 1H), 3.14-3.03 (m, 1H), 2.68-2.57 (m, 2H), 2.33-2.21 (m, 1H), 2.20-2.10 (m, 1H), 1.98-1.79 (m, 3H), 1.55 (s, 9H), 0.90-0.82 (m, 2H), 0.61-0.54 (m, 2H); MS(ES+) m/z 458.3 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-fluoromethyl)-4-(4-fluorophenyl) piperidin-1-yl)methyl)benzoate
##STR01341##
[2329] To a solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(hydroxymethyl) piperidin-1-yl)methyl)benzoate (0.78 g, 1.70 mmol) in dichloromethane (40 mL) under nitrogen, was added with diethylaminosulfur trifluoride (0.45 mL, 3.4 mmol) at 0.degree. C. The resulting solution was stirred at 0.degree. C. for 2.5 hours and then quenched with saturated sodium bicarbonate (30 mL). The mixture was extracted with ethyl acetate (80 mL) and the combied organic extracts were washed with saturated sodium bicarbonate (3.times.30 mL), saturated ammonium chloride (40 mL), brine (40 mL), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo. The residue was purified by flash chromatography (0-25% ethyl acetate in hexanes) to provide the title compound as a colorless oil (0.39 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.2 Hz, 1H), 7.21-7.09 (m, 3H), 7.01-6.91 (m, 2H), 3.64 (s, 2H), 2.85 (d, J=22.9 Hz, 2H), 2.71-2.59 (m, 2H), 2.41-2.28 (m, 2H), 1.97-1.85 (m, 1H), 1.79-1.61 (m, 4H), 1.55 (s, 9H), 0.93-0.85 (m, 2H), 0.63-0.55 (m, 2H); MS(ES+) m/z 460.3 (M+1).
Step 4. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl) piperidin-1-yl)methyl)benzoic acid
##STR01342##
[2331] To a solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl) piperidin-1-yl)methyl)benzoate (0.39 g, 0.85 mmol) in 1,4-dioxane (10 mL) was added concentrated hydrochloric acid (5 mL). The resulting solution was stirred at ambient temperature for 4 hours. The reaction mixture was diluted with ethyl acetate (60 mL), washed with water (60 mL), saturated ammonium chloride (50 mL), brine (2.times.40 mL), dried over anhydrous sodium sulfate, filtered the solid, and concentrated in vacuo to provide the title compound (0.24 g, 71%): MS(ES+) m/z 404.2 (M+1).
Step 5. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl) piperidin-1-yl)methyl)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01343##
[2333] Following the procedure as described in Example 53 step 5, and making variations as required to replace cyclopropanesulfonaimde with methylsulfonamide and to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl) piperidin-1-yl)methyl)benzoic acid and purification by preparative HPLC, the title compoind was obtained as a colorless solid (0.045 g, 26%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.22 (br s, 1H), 9.49 (br s, 1H), 7.45 (d, J=10.8 Hz, 1H), 7.28-7.19 (m, 3H), 7.17-7.08 (m, 2H), 4.50 (s, 2H), 4.14-3.60 (m, 2H), 3.33 (s, 3H), 3.30-3.10 (m, 2H), 2.96 (d, J=20.6 Hz, 2H), 2.17-2.05 (m, .sub.1H), 2.02-1.75 (m, 4H), 1.03-0.89 (m, 2H), 0.78-0.69 (m, 2H); MS(ES+) m/z 481.2 (M +1).
Example 514
Synthesis of 5-Cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-(fluoromethyl)-4-(4-- flurorophenyl) piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01344##
[2335] Following the procedure as described in Example 53 step 5, and making variations as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl)piperidin-1- -yl)methyl)benzoic acid, and purification by preparative HPLC s), the title compound was obtained as a colorless solid (0.07 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 9.53 (br s, 1H), 7.46 (d, J=11.1 Hz, 1H), 7.27-7.18 (m, 3H), 7.17-7.08 (m, 2H), 4.52 (s, 2H), 4.09-3.64 (m, 2), 3.37-3.13 (m, 2H), 3.09-3.02 (m, 1H), 2.96 (d, J=19.7 Hz, 2H), 2.17-2.05 (m, 1H), 2.02-1.73 (m, 4H), 1.16-1.04 (m, 4H), 1.01-0.91 (m, 2H), 0.79-0.68 (m, 2H); MS(ES+) m/z 507.2 (M+1).
Example 515
Synthesis of 5-cyclopropyl-2-fluoro-4-((4-(4-fluorephenyl)-4-(methoxymethyl) piperidin-1-yl)methyl)-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01345##
[2336] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl) piperidin-1-yl)methyl)benzoate
##STR01346##
[2338] To a solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(hydroxymethyl) piperidin-1-yl)methyl)benzoate (1.39 g, 3.04 mmol) and methyliodide (0.53 mL, 8.5 mmol) in anhydrous tetrahydrofuran (40 mL) at -78.degree. C. was treated with 1.0 M lithium bis(trimethylsilyl)amide in tetrahydrofuran (8.5 mL, 8.5 mmol) under nitrogen. The reulting solution was stirred -78.degree. C. for 30 minutes and then warmed to ambient temperature and stirred for 18 hours. The reaction mixture was diluted with ethyl acetate (100 mL), washed with saturated ammonium chloride (2.times.50 mL), brine (2.times.40 mL), dried over anhydrous sodium sulfate, filtered the solid, and concentrated iu vacuo. The residue was purified by flash chromatography (0-25% ethyl acetate in hexanes) to provide the title compound as a colorless oil (0.48 g, 34%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.45 (d, J=7.3 Hz, 1H), 7.33-7.26 (m, 2H), 7.17 (d, J=11.8 Hz, 1H), 7.05-6.96 (m, 2H), 3.54 (s, 2H), 3.31 (s, 2H), 3.19 (s, 3H), 2.66-2.54 (m, 2H), 2.33-2.21 (m, 2H), 2.17-2.06 (m, 2H), 2.02-1.84 (m, 3H), 1.56 (s, 9H), 0.90-0.81 (m, 2H), 0.61-0.53 (m, 2H); MS(ES+) m/z 472.3 (M+1).
Step 2. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl) piperidin-1-yl)methyl)benzoic acid
##STR01347##
[2340] Following the procedure as described in Example 513 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(fluoromethyl)-4-(4-fluorophenyl)piperidin-1- -yl) methyl)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl) piperidin-1-yl)methyl)benzoate, the title compound was obtained a colorless solid (0.26 g, 62%): MS(ES+) m/z 461.2 (M+1).
Step 3. Preparation of 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl) piperidin-1-yl)methyl)-N-methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01348##
[2342] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with with 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl)piperidin-- 1-yl) methyl)benzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.04 g, 21%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.31 (br s, 1H), 9.54 (br s, 1H), 7.54-7.30 (m, 3H), 7.25-7.02 (m, 3H), 5.74-4.82 (m, 1H), 4.63-4.34 (m, 2H), 3.73-3.55 (m, 1H), 3.38-3.27 (m, 5H), 3.18-3.08 (m, 4H), 2.92-2.74 (m, 1H), 2.43-2.37 (m1H), 2.28-2.02 (m, 3H), 1.99-1.84 (m, 1H), 1.04-0.85 (m, 2H), 0.81-0.66 (m, 2H), MS(ES+) m/z 493.2 (M+1).
Example 516
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluoro-4-((4-(4-fluorophenyl)-4-(- methoxymethyl) piperidin-1-yl)methyl)benzamide, trifluoroacetic acid salt
##STR01349##
[2344] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-2-fluoro-4-((4-(4-fluorophenyl)-4-(methoxymethyl)piperidin-- 1-yl) methyl)benzoic acid, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.045 g, 23%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 9.51 (br s, 1H), 7.54-7.31 (m, 3H), 7.26-7.07 (m, 3H), 4.64-4.53 (m, 2H), 3.72-3.57 (m, 1H), 3.42-3.26 (m, 2H), 3.19-3.09 (m, 4H), 3.08-3.00 (m, 1H), 2.92-2.75 (m, 1H), 2.44-2.35 (m, 1H), 2.29-2.01 (m, 3H), 2.00-1.85 (m, 1H), 1.17-1.06 (m, 4H), 1.04-0.84 (m, 2H), 0.82-0.65 (m, 2H); MS(ES+) m/z 519.2 (M+1).
Example 517
Synthesis of 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01350##
[2345] Step 1. Preparation of tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate
##STR01351##
[2347] To a mixture of tert-butyl piperazine-1-carboxylate (1.00 g, 5.37 mmol) and potassium carbonate (1.34 g, 9.76 mmol) in anhydrous dimethylformamide (50 mL) was added 1,3-dichloro-5-(chloromethyl)benzene (0.95 g, 4.88 mmol) under nitrogen. The resulting mixture was stirred at ambient temperature for 2 days. The reaction mixture was diluted with ethyl acetate (200 mL), washed with water (100 mL), saturated ammonium chloride (2.times.50 mL), brine (2.times.40 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue purified by flash chromatography (3:1 hexanes:ethyl acetate) to provide the title compound as a colorless oil (1.22 g, 73%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.25-7.17 (m, 3H), 3.45-3.35 (m, 6H), 2.39-2.30 (m, 4H), 1.42 (s, 9H); MS(ES+) m/z 345.1, 347.1 (M+1).
Step 2. Preparation of 1-(3,5-dichlorobenzyl)piperazine dihydrochloride
##STR01352##
[2349] To a mixture of tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate (1.22 g, 3.53 mmol) in 1,4-dioxane (20 mL) was added concentrated hydrochloric acid (5 mL). The mixture was stirred for 2 hours and then concentrated in vacuo to provide the title compound as a colorless solid (1.12 quant. yield): MS(ES+) m/z 245.1, 247.1 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl) methyl)-2-fluorobenzoate
##STR01353##
[2351] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 1-(3,5-dichlorobenzyl)piperazine dihydrochloride, the title compound was obtained as an oil (1.74 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46 (d, J=7.2 Hz, 1H), 7.25-7.10 (m, 4H), 3.63 (s, 2H), 3.43 2H), 2.59-2.35 (m, 8H), 1.98-1.87 (m, 1H), 1.55 (s, 9H), 0.94-0.86 (m, 2H), 0.67-0.55 (m, 2H); MS(ES+) m/z 508.2, 510.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl) methyl)-2-fluorobenzoic acid dihydrochloride
##STR01354##
[2353] To a solution tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate, (1.81 g, 3.67 mmol) in 1,4-dioxane (35 mL) was added concentrated hydrochloric acid (15 mL). The resulting mixture was stirred for 2 hours and then concentrated in vacuo. The residue was triturated with water to provide the title compound as a colorless solid (1.5 g, 94%:) MS(ES+) m/z 437.0, 439.0 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01355##
[2355] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoic acid dihydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as colorless solid (0.08 g, 21%): .sup.1NMR (300 MHz, DMSO-d.sub.6) .delta. 12.19 (br s, 1H), 9.74 (br s, 1H), 7.66 (s, 1H), 7.51 (s, 2H), 7.32 (d, J=11.6 Hz, 1H), 7.22 (d, J=7.1 Hz, 1H), 5.76 (br s, 1H), 4.11 (br s, 2H), 3.97 (br s, 2H), 3.33 (s, 3H), 3.27-2.65 (m, 8H), 2.11-1.98 (m, 1H), 1.01-0.86 (m, 2H), 0.77-0.59 (m, 2H); MS(ES+) m/z 514.1, 516.1 (M+1).
Example 518
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(3,5-dichlorobenzyl) piperazin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01356##
[2357] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piparazin-1-yl)methyl)-2-fluorobe- nzoic acid dihydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.20 g, 34%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.13 (br s, 1H), 7.65 (s, 1H), 7.54-7.49 (m, 2H), 7.33 (d, J=11.3 Hz, 1H), 7.20 (d, J=7.6 Hz, 1H), 4.10 (br s, 2H), 3.97 (br s, 2H), 3.33-2.59 (m, 9H), 2.10-1.99 (m, 1H), 1.15-1.03 (m, 4H), 0.96-0.87 (m, 2H), 0.72-0.63 (m, 2H); MS(ES+) m/z 540.2, 542.2 (M+1).
Example 519
Synthesis of 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01357##
[2358] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxypiperidin-1-yl)methyl)benzoate
##STR01358##
[2360] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with piperidin-4-ol, the title compound was obtained as an oil (0.98 g, 94%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.45 (d, J=7.3 Hz, 1H), 7.16 (d, J=11.8 Hz, 1H), 3.57-3.64 (m, 1H), 3.61 (s, 2H), 2.77-2.68 (m, 2H), 2.25-2.13 (m, 2H), 1.97-1.76 (m, 4H), 1.64-1.57 (m, 1H), 1.54 (s, 9H), 0.92-0.84 (m, 2H), 0.62-0.55 (m, 2H); MS(ES+) m/z 350.3 (M+1).
Step 2. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((methylsulfonyl)oxy)piperidin-1-yl)methyl)b- enzoate
##STR01359##
[2362] To a solution of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxypiperidin-1-yl)methyl)benzoate (0.98 g, 2.80 mmol) and triethylamine (0.59 mL, 4.2 mmol) in dichloromethane (40 mL) at 0.degree. C. was added methanesulfonyl chloride (0.26 mL, 3.36 mmol) under nitrogen. The resulting mixture was stirred at 0.degree. C. for 1 hour. The mixture was washed with saturated ammonium chloride (40 mL), brine (40 mL), dried over anhydrous sodium sulfate, filtered the solid, and concentrated in vacuo to dryness to provide the title compound as a colorless oil (1.20 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.14 (d, J=11.8 Hz, 1H), 7.81-4.71 (m, 1H), 3.64 (s, 2H), 3.00 (s, 3H), 2.76-2.65 (m, 2H), 2.43-2.28 (m, 2H), 2.09-1.83 (m, 5H), 1.55 (s, 9H), 0.94-0.86 (m, 2H), 0.63-0.56 (m, 2H); MS(ES+) m/z 428.2 (M+1).
Step 3. Preparation of tert-butyl 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01360##
[2364] To a mixture of tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((methylsulfonyl)oxy)piperidin-1-yl) methyl)benzoate (1.20 g, 2.80 mmol) and 4-chloro-3-(trifluoromethyl)phenol (0.61 g, 3.08 mmol) in anhydrous dimethylformamide (50 mL) was added potassium carbonate (0.43 g, 3.08 mmol). The reaction mixture was heated at 90.degree. C. under nitrogen for 7 hours, cooled to ambient temperature and diluted with ethyl acetate (150 mL), washed with water (100 mL), saturated ammonium chloride (100 mL), brine (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-25% ethyl acetate in hexanes) to provide the title compound as a colorless oil (0.71 g, 48%): MS(ES+) m/z 528.1, 530.1 (M+1).
Step 4. Preparation of 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01361##
[2366] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl)methyl)-5-cyclo- propyl-2-fluorobenzoate and purification by trituration with diethyl ether, the title compound was obtained as a colorless solid (0.32 g, 47%): MS(ES+) m/z 472.0, 474.0 (M+1).
Step 5. Preparation of 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01362##
[2368] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl)methyl)-5-cyclo- propyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.05 g, 24%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.30 (br s, 1H), 9.67 (br s, 1H), 7.63 (d, J=8.9 Hz, 1H), 7.51 (d, J=11.1 Hz, 1H), 7.44-7.30 (m, 2H), 7.25 (d, J=7.0 Hz, 1H), 4.95-4.76 (m, 1H), 4.54 (s, 2H), 4.26-3.74 (m, 2H), 3.47-3.12 (m, 6H), 2.28-1.74 (m, 4H), 1.06-0.92 (m, 2H), 0.82-0.71 (m, 2H); MS(ES+) m/z 549.1, 551.1 (M+1).
Example 520
Synthesis of 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01363##
[2370] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(4-chloro-3-(trifluoromethyl)phenoxy)piperidin-1-yl)methyl)-5-cyclo- propyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless (0.07 g, 32%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.22 (br s, 1H), 9.69 (br s, 1H), 7.63 (d, J=8.7 Hz, 1H), 7.51 (d, J=11.1 Hz, 1H), 7.44-7.30 (m, 2H), 7.23 (d, J=7.2 Hz, 1H), 7.92-4.79 (m, 1H), 4.54 (s, 2H), 4.23-3.76 (m, 2H), 3.50-3.31 (m, 2H), 3.10-3.00 (m, 1H), 2.28-1.70 (m, 5H), 1.15-1.06 (m, 4H), 1.03-0.95 (m, 2H), 0.81-0.72 (m, 2H); MS(ES+) m/z 575.2, 577.1 (M+1).
Example 521
Synthesis of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01364##
[2371] Step 1. Preparation of tert-butyl 4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate
##STR01365##
[2373] To a solution of tert-butyl 4-hydroxypiperidine-1-carboxylate (1.03 g, 5.09 mmol) in anhydrous 1,2-dimethoxyethane (35 mL) was added (60% sodium hydride in mineral oil, 0.20 g, 5.09 mmol) under nitrogen. The resulting mixture was stirred for 1 hour at ambient temperature and then added 2,5-dichloro-4-(trifluoromethyl)pyridine (1.00 g, 4.63 mmol). The reaction mixture was refluxed for 2 hours and cooled to ambient temperature. The reaction atixture was diluted with ethyl acetate (100 mL), washed with water (50 mL), saturated ammonium chloride (2.times.50 mL), brine (50 mL), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo to provide the title compound as an oil (1.76 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.18 (s, 1H), 6.99 (s, 1H), 5.21-5.11 (m, 1H), 3.80-3.66 (m, 2H), 3.30-3.19 (m, 2H), 1.99-1.88 (m, 2H), 1.75-1.61 (m, 2H), 1.43 (s, 9H); MS(ES+) m/z 325.0, 326.9 (M-55).
Step 2. Preparation of tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01366##
[2375] To a mixture of tert-utyl 4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate (1.76 g, 4.63 mmol) in 1,4-dioxane (30 mL) was added concentrated hydrochloric acid (10 mL). The resulting mixture was stirred for 5 hours and then concentrated in vacuo. The residue was dissolved in anhydrous dimethylformamide (70 mL), and to this solution was added potassium carbonate (2.21 g, 16.00 mmol) and tert-butyl 5-cyclopropyl-2-fluoro-4-(((methylsulfonyl)oxy)methyl)benzoate (1.38 g, 4.00 mmol). The resulting mixture was stirred under nitrogen at ambient temperature for 18 hours diluted with ethyl acetate (150 mL), washed with water (100 mL), saturated ammonium chloride (2.times.70 mL), brine (70 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-80% ethyl acetate in hexanes) to provide the title compound as a colorless oil (1.92 g, 91%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.19 (s. 1H), 7.48 (d, J=7.1 Hz, 1H), 7.18 (d, J=11.8 Hz, 1H), 7.00 (s, 1H), 5.12-5.02 (m, 1H), 3.64 (s, 2H), 2.78-2.67 (m, 2H), 2.41-2.31 (m, 2H), 2.05-1.90 (m, 3H), 1.86-1.73 (m, 2H), 1.56 (s, 9H), 0.94-0.86 (m, 2H), 0.65-0.58 (m, 2H); MS(ES+) m/z 529.2, 531.2 (M+1H).
Step 3. Preparation of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01367##
[2377] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.85 g, quant. yield): MS(ES+) m/z 472.9, 475.1 (M+1H).
Step 4. Preparation of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01368##
[2379] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.45 g, 67%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.28 (br s, 1H), 9.92 (br s, 1H), 8.49 (s, 1H), 7.51 (d, J=10.9 Hz, 1H), 7.30 (s, 1H), 7.25 (d, J=7.1 Hz, 1H), 5.33-5.15 (m, 1H), 4.55 (s, 2H), 3.51-3.24 (m, 7H), 2.34-2.00 (m, 4H), 1.93-1.75 (m, 1H), 1.04-0.91 (m, 2H), 0.81-0.70 (m, 2H); MS(ES+) m/z 550.1, 552.1 (M+1).
Example 522
Synthesis of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01369##
[2381] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.35 g, 51%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.24 (br s, 1H), 10.16 (br s, 1H), 8.48 (s, 1H), 7.53 (d, J=10.9 Hz, 1H), 7.30 (s, 1H), 7.23 (d, J=7.1 Hz, 1H), 5.32-5.14 (m, 1H), 4.56 (s, 2H), 3.52-3.24 (m, 4H), 3.11-3.00 (m, 1H), 2.33-1.98 (m, 4H), 1.96-1.82 (m, 1H), 1.17-1.06 (m, 4H), 1.03-0.94 (m, 2H), 0.81-0.71 (m, 2H); MS(ES+) m/z 576.1, 578.1 (M+1).
Example 523
Synthesis of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01370##
[2382] Step 1. Preparation of tert-butyl 4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate
##STR01371##
[2384] Following the procedure as described in Example 521 step 1, and making variation as required to replace 2,5-dichloro-4-(trifluoromethyl)pyridine with 3-chloro-2-fluoro-5-(trifluoromethyl)pyridine, the title compound was obtained as a colorless solid (1.76 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.25 (s, 1H), 7.80 (s, 1H), 5.38-5.28 (m, 1H), 3.87-3.72 (m, 4H), 3.04-2.90 (m, 2H), 1.86-1.72 (m, 2H), 1.40 (s, 9H); MS(ES+) m/z 324.9, 326.9 (M-55).
Step 2. Preparation of tert-butyl 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01372##
[2386] Following the procedure as described in Example 521 step 2, and making vatiation as required to replace tert-butyl 4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate with tert-butyl 4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidine-1-carboxylate- , the title compound was obtained as a colorless solid (0.52 g, 25%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.29-8.26 (m, 1H), 7.81 (d, J=2.1 Hz, 1H), 7.48 (d, J=7.1 Hz, 1H), 7.19 (d, J=11.7 Hz, 1H), 5.28-5.19 (m, 1H), 3.66 (s, 2H), 2.79-2.68 (m, 2H), 2.46-2.35 (m, 2H), 2.09-1.82 (m, 5H), 1.56 (s, 9H), 0.96-0.87 (m, 2H), 0.65-0.58 (m, 2H); MS(ES+) m/z 529.3, 531.2 (M+1).
Step 3. Preparation of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01373##
[2388] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.50 g, quant. yield): MS(ES+) m/z 472.9, 475.1 (M+1).
Step 4. Preparation of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01374##
[2390] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl)methy- l)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.11 g, 39%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.31 (br s, 1H), 9.87 (br s, 1H), 8.55-8.51 (m1H), 8.43-8.39 (m, 1H), 7.52 (d, J=11.1 Hz, 1H), 7.26 (d, J=7.1 Hz, 1H), 5.49-5.24 (m, 1H), 4.56 (s, 2H), 3.55-3.14 (m, 7H), 2.38-2.06 (m, 4H), 2.03-1.82 (m, 1H), 1.05-0.93 (m, 2H), 0.83-0.70 (m, 2H); MS(ES+) m/z 550.1, 552.1 (M+1).
Example 524
Synthesis of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01375##
[2392] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl)methy- l)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.11 g, 38%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.22 (br s, 1H), 9.96 (br s, 1H), 8.55-8.51 (m, 1H), 8.43-8.39 (m, 1H), 7.55 (d, J=10.9 Hz, 1H), 7.24 (d, J=7.1 Hz, 1H), 5.48-5.25 (m, 1H), 4.56 (s, 2H), 3.56-3.16 (m, 4H), 3.11-3.00 (m, 1H), 2.38-2.07 (m, 4H), 2.01-1.82 (m, 1H), 1.15-1.06 (m, 4H), 1.04-0.94 (m, 2H), 0.80-0.72 (m, 2H); MS(ES+) m/z 576.1, 578.1 (M+1).
Example 525
Synthesis of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01376##
[2393] Step 1. Preparation of methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01377##
[2395] To a degassed mixture of methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride (0.15 g, 0.46 mmol), 2,5-dichloro-4-(trifluoromethyl)pyridine (0.15 g, 0.68 mmol) and cesium carbonate (0.58 g, 1.77 mmol) in anhydrous toluene (5.0 mL) was added rac-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl (0.06 g, 0.091 mmol) and bis(dibenzylideneacetone)palladium(0) (0.03 g, 0.046 mmol). The resulting mixture was heated at 100.degree. C. in a sealed tube for 30 hours. The reaction mixture was diluted with ethyl acetate (30 mL), washed with water (30 mL), saturated ammonium chloride (30 mL), brine (30 mL), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo. The residue was purified by HPLC to provide the title compound as a colorless solid (0.10 g, 46%): MS(ES+) m/z 473.1, 475.1 (M+1).
Step 2. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01378##
[2397] To a mixture of methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (0.10 g, 0.21 mmol) and lithium hydroxide monohydrate (0.09 g, 2.10 mmol) in tetrahydrofuran (20 mL) and water (10 mL) was refluxed for 7 hours. The mixture was diluted with ethyl acetate (50 mL), washed with 1.0 N hydrochloride acid solution (30 mL), brine (3.times.30 mL), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo to provide the title compound as a colorless solid (0.07 g, 67%): MS(ES+) m/z 458.9, 460.9 (M+1).
Step 3. Preparation of 4-((4-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01379##
[2399] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and purification by preparative HPLC, the title compound was obtained as a colorless solid, (0.03 g, 38%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.81 (s, 1H), 8.27 (s, 1H), 7.06 (d, J=8.3 Hz, 1H), 6.96 (d, J=13.0 Hz, 1H), 6.75 (s, 1H), 4.07 (s, 2H), 4.03 (d, J=8.6 Hz, 2H), 3.79 (d, J=8.6 Hz, 2H), 3.08-2.98 (m, 1H), 1.71-1.59 (m, 1H), 1.40 (s, 3H), 1.12-1.03 (m, 4H), 0.59-0.53 (m, 4H); MS(ES+) m/z 562.1, 564.1 (M+1).
Example 526
Synthesis of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperazin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01380##
[2400] Step 1. Preparation of tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperazine-1-carboxylate
##STR01381##
[2402] To a solution of perfluorophenyl 3,5-dichlorobenzenesulfonate (2.30 g, 5.85 mmol) and triethylamine (1.63 mL, 11.7 mmol) in dichlocomethane (50 mL) was added with tert-butyl piperazine-1-carboxylate (1.31 g, 7.02 mmol) at ambient temperature. The resulting mixture was stirred at ambient temperature for 18 hours under nitrogen, diluted with ethyl acetate (100 mL), washed with saturated ammonium chloride (2.times.50 mL), saturated sodium bicarbonate (50 mL), brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-20% ethyl acetate in hexanes) to provide the compound as a colorless solid (1.87 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.60-7.54 (m, 3H), 3.50 (t, J=4.8 Hz, 4H), 2.99 (t, J=5.1 Hz, 4H), 1.39 (s, 9H); MS(ES+) m/z 294.9, 296.9 (M-Boc+2).
Step 2. Preparation of 1-((3,5-dichlorophenyl)sulfonyl)piperazine hydrochloride
##STR01382##
[2404] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperazine-1-carboxylate, the title compound was obtained as a colorless solid (1.57 g, quant. yield): MS(ES+) m/z 295.0, 297.0 (M+1).
Step 2. Preparation of tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl) piperazin-1-yl)methyl)-2-fluorobenzoate
##STR01383##
[2406] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 1-(3,5-dichlorophenyl)sulfonyl)piperazine hydrochloride and purification by flash chromatography (0-20% ethyl acetate in hexanes), the title compound was obtained as an foam (1.81 g, 70%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.61-7.55 (m, 3H), 7.45 (d, J=7.5 Hz, 1H), 7.04 (d, J=11.7 Hz, 1H), 3.64 (s, 2H), 3.11-3.00 (m, 4H), 2.61-2.51 (m, 4H), 1.92-1.79 (m, 1H), 1.54 (s, 9H), 0.91-0.83 (m, 2H), 0.62-0.54 (m, 2H); MS(ES+) m/z 543.0, 545.0 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperazin-1-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01384##
[2408] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperazin-1-yl) methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.74 g, quant. yield): MS(ES+) m/z 478.0, 489.0 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperazin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01385##
[2410] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperazin-1-yl)methyl)-- 2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.18 g, 43%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 11.97 (br s, 1H), 8.07-8.04 (m, 1H), 7.74 (d, J=1.7 Hz, 2H), 7.32 (d, J=11.3 Hz, 1H), 7.21 (d, J=7.2 Hz, 1H), 5.68 (br s, 1H), 4.20 (s, 2H), 3.32 (s, 3H), 3.29-2.90 (m, 8H), 2.08-1.97 (m, 1H), 0.97-0.87 (m, 2H), 0.72-0.64 (m, 2H); MS(ES+) m/z 564.0, 566.0 (M+1).
Example 527
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5-dichlorophenyl)sulfonyl- ) piperazin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01386##
[2412] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenoxy)sulfonyl)piperazin-1-yl)methyl)- -2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.19 g, 43%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.01 (br s, 1H), 8.04 (d, J=1.7 Hz, 1H), 7.74 (d, J=1.7 Hz, 2H), 7.31 (d, J=11.4 Hz, 1H), 7.19 (d, J=7.1 Hz, 1H), 5.05 (br s, 1H), 4.13 (s, 2H), 3.29-2.76 (m, 9H), 2.07-1.95 (m, 1H), 1.14-1.05 (m, 4H), 0.96-0.87 (m, 2H), 0.71-0.63 (m, 2H); MS(ES+) m/z 590.0, 592.0 (M+1).
Example 528
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1R,3s,5S)-3-(3,5-dichloropheno- xy)-8-azabicyclo[3.2.1]octan-8-yl) methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01387##
[2413] Step 1. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,3s,5S)-3-hydroxy-8-azabicyclo[3.2.1]octan-- 8-yl) methyl)benzoate
##STR01388##
[2415] Following the procedure as described in Example 470 Step 4, and making variation as required to replace (1R,3s,5S)-3-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)-8-azabicyclo[- 3.2.1]-octane with nortropine, the title compound was obtained as a yellowish solid (2.16 g, 99%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.46 (d, J=7.3 Hz, 1H), 7.31 (d, J=12.2 Hz, 1H), 4.09-4.03 (m, 1H), 3.63 (s, 2H), 3.13-3.07 (m, 2H), 2.16-1.94 (m, 6H), 1.92-1.81 (m, 1H), 1.70 (br s, 1H), 1.65 (br s, 1H), 1.57-1.57 (m, 1H), 1.56 (s, 9H), 0.91-0.83 (m, 2H), 0.62-0.56 (m, 2H).
Step 2. Preparation of tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,3r,5S)-3-((methylsulfonyl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)benzoate
##STR01389##
[2417] Following the procedure as described in Example 519 step 2, and making variation as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxypiperidin-1-yl)methyl)benzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,3r,5S)-3-hydroxy-8-azabicyclo[3.2.1]octan-- 8-yl)methyl)benzoate, the title compound was obtained as an foam (1.21 g, quant. yield): MS(ES+) m/z 454.2 (M+1).
Step 5. Preparation of tert-butyl 5-cyclopropyl-4-(((1R,3s,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoate
##STR01390##
[2419] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 3,5-dichlorophenol and to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(methylsulfonyl)oxy)piperidin-1-yl)methyl)be- nzoate with tert-butyl 5-cyclopropyl-2-fluoro-4-(((1R,3r,5S)-3-((methylsulfonyl)oxy)-8-azabicycl- o[3.2.1]octan-8-yl) methyl)benzoate, the title compound was obtained as an colorless oil (0.70 g, 36%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.30 (d, J=12.1 Hz, 1H), 6.91-6.87 (m, 1H), 6.76-6.69 (m, 2H), 4.53-4.40 (m, 1H), 3.75 (s, 2H), 3.32-3.24 (m, 2H), 2.15-1.76 (m, 7H), 1.72-1.64 (m, 2H), 1.57 (s, 9H), 0.94-0.85 (m, 2H), 0.64-0.56 (m, 2H); MS(ES+) m/z 520.1, 522.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-(((1R,3s,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01391##
[2421] Following the procedure as described in Example 517 step 4, and making variation as required to replace tert-butyl 5-cyclopropyl-4-((4-(3,5-dichlorobenzyl)piperazin-1-yl)methyl)-2-fluorobe- nzoate with tert-butyl 5-cyclopropyl-4-(((1R,3s,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.67 g, quant. yield): MS(ES+) m/z 464.1, 466.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1R,3s,5S)-3-(3,5-dichloropheno- xy)-8-azabicyclo[3.2.1]octan-8-yl) methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01392##
[2423] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((1R,3s,5S)-3-(3,5-dichlorophenoxy)-8-azabicyclo[3.2.1]o- ctan-8-yl) methyl)-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtaned as a colorless solid (0.04 g, 4%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.25 (br s, 1H), 9.63 (br s, 1H), 7.57 (d, J=11.4 Hz, 1H), 7.26 (d, J=7.1 Hz, 1H), 7.16-7.09 (m, 3H), 4.97-4.83 (m, 1H), 4.38 (s, 2H), 4.03 (s, 2H), 3.10-2.98 (m, 1H), 2.42-2.17 (m, 6H), 2.15-1.94 (m, 3H), 1.17-1.06 (m, 4H), 1.03-0.93 (m, 2H), 0.82-0.72 (m, 2H); MS(ES+) m/z 567.1, 569.1 (M+1).
Example 529
Synthesis of (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01393##
[2424] Step 1. Preparation of methyl (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate
##STR01394##
[2426] Following the procedure as described in Example 495 step 1 and making variation as required to replace 3,5-dichlorobenzoyl chloride with 3-chlorobenzoyl chloride, the title compound was obtained as a yellowish oil (0.72 g, 24% in 2 steps): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.53-7.48 (m, 1H), 7.42-7.35 (m, 2H), 7.21-7.16 (m, 2H), 6.39 (d, J=13.0 Hz, 1H), 4.35-4.24 (m, 1H), 3.85 (s, 3H), 2.96-2.87 (m, 1H), 2.74-2.64 (m, 1H), 2.34-2.20 (m, 2H), 2.09-1.97 (m, 2H), 1.86-1.77 (m, 1H), 1.63-1.50 (m, 2H), 1.30 (s, 3H), 1.30 (s, 3H), 0.92-0.84 (m, 2H), 0.65-0.59 (m, 2H); MS(ES+) m/z 446.2, 448.2 (M+1).
Step 2. Preparation of (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01395##
[2428] Following the procedure as described in Example 481 step 5 and making variation as required to replace methyl 4-((1-(3-chloro-5-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with (R)-methyl 4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as an orange amorphous solid (1.47 g, 68%): MS(ES+) m/z 432.2, 434.2 (M+1).
Step 3. Preparation of (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01396##
[2430] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl)oxy-5-cyclopropyl- -2-fluorobenzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.08 g, 24%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 12.12 (br s, 1H), 8.85-8.65 (m, 1H), 7.67-7.58 (m, 1H), 7.57-7.49 (m, 2H), 7.46-7.40 (m, 2H), 6.96 (d, J=14.0 Hz, 1H), 5.18-5.05 (m, 1H), 3.78-3.66 (m, 1H), 3.47-3.32 (m, 4H), 2.60-2.43 (m, 2H), 2.38-2.08. (m, 2H), 2.03-1.93 (m, 2H), 1.90 (s, 3H), 1.88 (s, 3H), 1.57-1.41 (m, 1H), 0.93-0.82 (m, 2H), 0.65-0.56 (m, 2H); MS(ES+) m/z 509.1, 511.1 (M+1).
Example 530
Synthesis of (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl) oxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01397##
[2432] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with (R)-4-((1-(2-(3-chlorophenyl)propan-2-yl)piperidin-3-yl)oxy)-5-cyclopropy- l-2-fluorobenzoic acid, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.08 g, 23%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 11.92 (br s, 1H), 8.72 (d, J=15.5 Hz, 1H), 7.67-7.58 (m, 1H), 7.57-7.49 (m, 2H), 7.47-7.40 (m, 2H), 6.96 (d, J=14.0 Hz, 1H), 5.18-5.04 (m, 1H), 3.78-3.65 (m, 1H), 3.46-3.34 (m, 1H), 3.11-3.01 (m, 1H), 2.61-2.43 (m, 2H), 2.37-2.10 (m, 2H), 2.04-1.93 (m, 2H), 1.91 (s, 3H), 1.87 (s, 3H), 1.57-1.39 (m, 3H), 1.16-1.07 (m, 2H), 0.92-0.84 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 535.2, 537.1 (M+1).
Example 531
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5 dichlorophenyl)thio)piperidin-1-yl) methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01398##
[2433] Step 1. Preparation of tert-butyl 4-((methylsulfonyl)oxy)piperidine-1-carboxylate
##STR01399##
[2435] Following the procedure as described in Example 519 step 2, and making variation as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxypiperidin-1-yl)methyl)benzoate with tert-buzyl 4-hydroxypiperidine-1-carboxylate, the title compound was obtained as a colorless solid (7.32 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 4.88-4.78 (m, 1H), 3.71-3.60 (m, 2H), 3.30-3.19 (m, 2H), 2.99 (s, 3H), 1.97-1.86 (m, 2H), 1.82-1.70 (m, 2H) 1.41 (s, 9H); MS(ES+) m/z 302.1 (M+23).
Step 2. Preparation of tert-butyl 4-((3,5-dichlorophenyl)thio)piperidine-1-carboxylate
##STR01400##
[2437] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 3,5-dichiorobenzenethiol and tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(methylsulfonyl)oxy)piperidin-1-yl)methyl)be- nzoate with tert-butyl 4-((methylsulfonyl)oxy)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (3.66 g, 64%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.22-7.16 (m, 3H), 3.99-3.81 (m, 2H), 3.29-3.17 (m, 1H), 2.98-2.85 (m, 2H), 1.94-1.83 (m, 2H), 1.58-1.45 (m, 2H), 1.41 (s, 9H); MS(ES+) m/z 306.0, 308.0 (M-Boc+2H).
Step 3. Preparation of 4-((3,5-dichlorophenyl)thio)piperidine hydrochloride
##STR01401##
[2439] Following the procedare as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-(3,5-dichlorophenyl) thio)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (0.19 g, quant. yield): MS(ES+) m/z: 262.1, 264.1 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl) thio)piperidin-1-yl)methyl)-2-fluorobenzoate
##STR01402##
[2441] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3,5-dichlorophenyl) thio)piperidine hydrochloride, the title compound was obtained as an oil (0.29 g, quant. yield): MS(ES+) m/z 510.0, 512.0 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)thio) piperidin-1-yl)methyl)-2-fluorobenzoic acid hydrochloride
##STR01403##
[2443] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl) thio)piperidin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.28 g, quant. yield): MS(ES+) m/z 454.0, 456.1 (M+1).
Step 6. Preparation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5-dichlorophenyl) thio)piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01404##
[2445] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)thio)piperidin-1-yl)methyl)-2-fl- uorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.09 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 9.43 (br s, 2H), 7.71 (d, J=7.2 Hz, 1H), 7.43 (d, J=12.1 Hz, 1H), 7.27-7.20 (m, 3H), 4.43 (s, 2H), 3.61-3.15 (m, 5H), 3.09-2.99 (m, 1H), 2.50-2.20 (m, 2H), 2.07-1.91 (m, 2H), 1.91-1.81 (m, 1H), 1.46-1.37 (m, 2H), 1.19-1.04 (m, 4H), 0.79-0.69 (m, 2H); MS(ES+) m/z 557.1, 559.1 (M+1).
Example 532
Synthesis of 5-cyclopropyl-4-((4-(3,5-dichlorophenyl)sulfonyl)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01405##
[2446] Step 1. Preparation of tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate and tert-butyl 4-((3,5-dichlorophenyl)sulfinyl)piperidine-1-carboxylate
##STR01406##
[2448] To a solution of tert-butyl 4-((3,5-dichlorophenyl)thio)piperidine-1-carboxylate (3.66 g, 10.10 mmol) in dichloromethane (100 mL) at 0.degree. C. was added m-chloroperoxybenzoic acid (77%, 3.40 g, 15.15 mmol). The resulting mixture was stirred at 0.degree. C. for 3 hours and then concentrated in vacuo to about 40 mL volume. The mixture was diluted with ethyl acetate (150 mL), washed with saturated sodium thiosulfate (100 mL), 1.0 M aqueous sodium hydroxide solution (3.times.50 mL), brine (50 mL), dried over anhydrous sodium sulfate, filtered the solid, and concentrated in vacuo. The residue was purified by flash chromatography (0-50% ethyl acetate in hexanes) to provide tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate (2.12 g, 53%) and tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate (1.35 g, 34%) as a colorless solids. Analytical data for tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.73-7.68 (m, 2H), 7.64-7.60 (m, 1H), 4.31-4.15 (m, 2H), 3.10-2.97 (m, 1H), 2.72-2.56 (m, 2H), 1.99-1.89 (m, 2H), 1.67-1.51 (m, 2H), 1.41 (s, 9H); MS(ES+) m/z 294.0, 296.0 (M-Boc+H). Analytical data for tert-butyl 4-((3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46-7.44 (m, 1H), 7.43-7.41 (m, 2H), 4.27-4.12 (m, 2H), 2.76-2.59 (m, 3H), 1.86-1.76 (m, 1H), 1.68-1.54 (m, 3H), 1.40 (s, 9H); MS(ES+) m/z 378.0, 380.0 (M+1).
Step 2. Preparation of 4-((3,5-dichlorophenyl)sulfonyl)piperidine hydrochloride
##STR01407##
[2450] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-(3,5-dichlorophenyl)sulfonyl)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (1.78 g, quant. yield): MS(ES+) m/z 294.1, 296.1 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl) piperidin-1-yl)methyl)-2-fluorobenzoate
##STR01408##
[2452] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-((3,5-dichlorophenyl) sulfonyl)piperidine hydrochloride, the title compound was obtained as a colorless solid (2.63 quant. yield): MS(ES+) m/z 542.0, 544.0 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperidin-1-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01409##
[2454] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl) sulfonyl)piperidin-1-yl)methyl)-2-fluorobenzoate, the title compoond was obtained as a colorless solid (2.59 g, 90%): MS(ES+) m/z 486.0, 488.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl) piperidin-1-yl)methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01410##
[2456] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperidin-1-yl)methyl)-- 2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.11 g, 20%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.17 (br s, 1H), 9.85 (br s, 1H), 8.12 (dd, J=1.7, 1.7 Hz, 1H), 7.84 (d, J=1.7 Hz, 2H), 7.39 (d, J=11.1 Hz, 1H), 7.23 (d, J=7.1 Hz, 1H), 4.32 (s, 2H), 3.75-3.59 (m, 3H), 3.32 (s, 3H), 2.98-2.75 (m, 2H), 2.10-1.96 (m, 3H), 1.88-1.68 (m, 2H), 0.98-0.88 (m, 2H), 0.76-0.66 (m, 2H); MS(ES+) m/z 563.0, 565.0 (M+1).
Example 533
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5-dichlorophenyl) sulfonyl)piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01411##
[2458] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopopyl-4-((4-((3,5-dichlorophenyl)sulfonyl)piperidin-1-yl)methyl)-2- -fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the the compound was obtained as a colorless solid (0.04 g, 7%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.65-7.61 (m, 2H), 7.60-7.57 (m, 1H), 7.38 (d, J=7.1 Hz, 1H), 7.16 (d, J=11.4 Hz, 1H), 4.02 (s, 2H), 3.28-3.20 (m, 2H), 3.17-3.06 (m, 1H), 3.02-2.91 (m, 1H), 2.66-2.51 (m, 2H), 2.11-2.00 (m, 2H), 1.97-1.74 (m, 3H), 1.34-1.25 (m, 2H), 1.08-0.98 (m, 2H), 0.95-0.86 (m, 2H), 0.62-0.54 (m, 2H); MS(ES+) m/z 589.1, 591.1 (M+1).
Example 534
Synthesis of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl)pipperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01412##
[2459] Step 1. Preparation of 4-((3,5-dichlorophenyl)sulfinyl)pipperidine hydrochloride
##STR01413##
[2461] Following the procedure described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-((3,5-dichlorophenyl) sulfinyl)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (1.12 g, quant. yield): MS(ES+) m/z 278.1, 280.1 (M+1).
Step 2. Preparation of tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl) piperidin-1-yl)methyl)-2-fluorobenzoate
##STR01414##
[2463] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-((3,5-dichlorophenyl) sulfinyl)piperidine, hydrochloride, the title compound was obtained as a colorless solid (1.40 g, 80%): MS(ES+) m/z 526.1, 528.1 (M+1).
Step 3. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl) piperidin-1-yl)methyl)-2-fluorobenzoic acid hydrochloride
##STR01415##
[2465] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((4-((3,5-dichlorophenyl) sulfinyl)piperidin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.35 g, quant. yield): MS(ES+) m/z 470.0, 472.1 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01416##
[2467] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl)piperidin-1-yl)methyl)-- 2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.05 g, 9%): .sup.1H NMR (300 MHz, CDCl.sub.310% CD.sub.3OD) .delta. 7.46-7.37 (m, 3H), 7.26-7.20 (m, 2H), 4.19 (s, 2H), 3.42-3.33 (m, 2H), 3.31-3.24 (m, 4H), 2.88-2.70 (m, 2H), 2.08-1.87 (m, 4H), 1.86-1.76 (m, 1H), 1.00-0.91 (m, 2H), 0.66-0.58 (m, 2H) (Note: Note: exchangeable protons not observed.); MS(ES+) m/z 547.0, 549.0 (M+1).
Example 535
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-((3,5-dichlorophenyl)sulfinyl- ) piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01417##
[2469] Following the prooedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-((3,5-dichlorophenyl)sulfinyl)piperidin-1-yl)methyl)-- 2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.05 g, 8%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.47-7.42 (m, 2H), 7.42-7.39 (m, 2H), 7.29-7.23 (m, 1H), 4.26 (s, 2H), 3.33-3.27 (m, 2H), 3.05-2.95 (m, 1H), 2.94-2.84 (m, 2H), 2.82-2.72 (m, 1H), 2.13-2.03 (m, 2H), 2.02-1.93 (m, 2H), 1.88-1.77 (m, 1H), 1.39-1.30 (m, 2H), 1.12-1.03 (m, 2H), 1.02-0.95 (m, 2H), 0.68-0.60 (m, 2H); MS(ES+) m/z 573.1, 575.1. (M+1).
Example 536
Synthesis of 4-((4-(3-Chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifuoroacetic acid salt
##STR01418##
[2470] Step 1. Preparation of tert-butyl 4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidine-1-carboxylate
##STR01419##
[2472] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 3-chloro-2-fluoro-5-(trifluoromethyl)phenol and tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((methylsulfonyl)oxy)piperidin-1-yl)methyl)b- enzoate with tert-butyl 4-((methylsulfonyl)oxy)piperidine-1-carboxylate, the title compound was obtained as an colorless oil (2.07 g, 69%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.29-7.25 (m, 1H), 7.11-7.06 (m, 1H), 4.55-4.46 (m, 1H), 3.74-3.61 (m, 2H), 3.41-3.29 (m, 2H), 1.97-1.85 (m, 2H), 1.83-1.71 (m, 2H), 1.44 (s, 9H); MS(ES+) m/z 342.0, 344.0 (M-55 ).
Step 2. Preparation of 4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidine hydrochloride
##STR01420##
[2474] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (2.24 g, quant. yield): MS(ES+) m/z 298.1, 300.1 (M+1).
Step 3. Preparation of tert-butyl 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01421##
[2476] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)piperidine hydrochloride, the title compound was obtained as a colorless solid (2.00, 66%): MS(ES+) m/z: 546.2, 548.2 (M+1).
Step 4. Preparation of 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy) piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01422##
[2478] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl) phenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.93 g, quant. yield): MS(ES+) m/z 490.1, 492.1 (M+1).
Step 5. Preparation of -((4-(3-Chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01423##
[2480] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidin-1-yl)methyl- )-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.22 g, 38%): .sup.1H NMR (300 MHz, DMSO-d.sub.6) .delta. 12.19 (br s, 1H), 10.27 (br s, 1H), 7.68-7.61 (m, 2H), 7.52 (d, J=11.1 Hz, 1H), 7.25 (d, J=7.1 Hz, 1H), 4.95-4.77 (m, 1H), 4.57 (s, 2H), 3.45-3.36 (m, 2H), 3.34 (s, 3H), 3.31-3.23 (m, 2H), 2.24-2.10 (m, 3H), 2.06-1.98 (m, 2H), 1.03-0.95 (m, 2H), 0.81-0.73 (m, 2H); MS(ES+) m/z 567.1, 569.1 (M+1).
Example 537
Synthesis of 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01424##
[2482] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(3-chloro-2-fluoro-5-(trifluoromethyl)phenoxy)piperidin-1-yl)methyl- )-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.25 g, 43%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.40 (d, J =7.1 Hz, 1H), 7.34-7.25 (m, 2H), 7.13-7.08 (m, 1H), 4.67-4.59 (m, 1H), 4.41 (s, 2H), 3.42-3.30 (m, 2H), 3.29-3.15 (m, 2H), 3.02-2.91 (m, 1H), 2.32-2.17 (m, 2H), 2.13-2.01 (m, 2H), 1.90-1.79 (m, 1H), 1.34-1.26 (m, 2H), 1.08-0.96 (m, 4H), 0.69-0.61 (m, 2H); MS(ES+) m/z 593.1, 595.1 (M+1).
Example 538
Synthesis of (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-N-(methylsulfon- yl)benzamide
##STR01425##
[2483] Step 1. Preparation of methyl (R)-4-((1-acetylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate
##STR01426##
[2485] To a solution of methyl (R)-5-cyclopropyl-2-fluoro-4-(piperidin-3-yloxy)benzoate (2.00 g, 6.82 mmol), triethylamine (1.90 mL, 13.64 mmol), and 4-dimethylaminopyridine (0.20 g, 1.64 mmol) in dichloromethane (60 mL) was added acetic anhydride (1.29 m.L, 13.64 mmol) under nitrogen. The resulting solution was stirred at ambient temperature for 18 hours. The reaction mixture was concentrated in vacuo, diluted with ethyl acetate (100 mL), washed with aqueous saturated ammonium chloride solution (2.times.50 mL), aqueous saturated sodium bicarbonate solution (2.times.50 mL), and brine (30 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (50-100% ethyl acetate hexanes) to provide the title compound as a colorless solid (1.28 g, 56%): MS(ES+) m/z 336.1 (M+1).
Step 2. Preparation of methyl (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate
##STR01427##
[2487] To a solution of methyl (R)-4-((1-acetylpiperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate (1.28 g, 3.82 mmol) and 2,6-di-tert-butyl-4-methylpyridine (0.94 g, 4.58 mmol) in anhydrous dichloromethane (70 mL) at -78.degree. C. under nitrogen was added trifluoromethanesulfonic anhydride (0.77 mL, 4.58 mmol) dropwise. The resulting mixture was stirred at -78.degree. C. for 2 hours and then added methyl lithium (1.6 M in diethyl ether, 11.9 mL, 19.04 mmol) dropwise. The resulting mixture was stirred at -78.degree. C. for 1 hour and then quenched cold with aqueous saturated ammonium chloride solution (50 mL). The mixture was warmed to ambient temperature, the organic layer was washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by preparative HPLC to provide the title compound as a colorless solid (0.40 g, 30%): .sup.1H NMR (300 MHz, CDC.sub.3) .delta. 7.39 (d, J=8.4 Hz, 1H), 6.46 (d, J=12.8 Hz, 1H), 4.67-4.39 (m, 1H), 3.84 (s, 3H), 3.46-3.31 (m, 1H), 3.16-2.98 (m, 1H), 2.32-2.11 (m, 3H), 2.05-1.93 (m, 1H), 1.91-1.69 (m, 2H), 1.50-1.34 (m, 1H), 1.15 (s, 9H), 0.90-0.81 (m, 2H), 0.66-0.55 (m, 2H); MS(ES+) m/z 350.3 (M+1).
Step 3. Preparation of (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluoro-N-(methy- lsulfonyl)benzamide
##STR01428##
[2489] To a mixture of (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate (0.14 g, 0.40 mmol) and sodium hydroxide (0.12 g, 2.85 mmol) in tetrahydrofuran (10 mL) and water (2.5 mL) was refluxed for 6 hours. The reaction mixture was cooled to ambient temperature and added 1.0 N aqueous hydrochloride acid solution (5 mL) and concentrated in vacuo. The residue was dissolved in anhydrous dimethylformamide (5 mL), and dichloromethane (5 mL). To this solution was added methanesulfonamide (0.06 g, 0.60 mmol), N-(3-dimethylaminopropyl)-N-ethylcarbodiimide hydrochloride (0.12 g, 0.60 mmol) and 4-dimethylaminopyridine (0.20 g, 1.60 mmol). The resulting mixture was stirred for 20 hours at ambient temperature, and diluted with 1.0 N aqueous hydrochloric acid solution (15 mL) and brine (15 mL); extracted with dichloromethane:methanol (20:1 v/v, 2.times.50 mL); dried dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by preparative HPLC to provide the title compound as a colorless solid (0.03 g, 18%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.29 (d, J=8.9 Hz, 1H), 6.54 (d, J=13.1 Hz, 1H), 4.51-4.38 (m, 1H), 3.47-3.37 (m, 1H), 3.29-3.18 (m, 1H), 3.09 (s, 3H), 2.37-2.23 (m, 2H), 2.19-2.07 (m, 1H), 1.94-1.73 (m, 3H), 1.44-1.29 (m, 1H), 1.19 (s, 9H), 0.81-0.73 (m, 2H), 0.58-0.49 (m, 2H); MS(ES+) m/z 413.2 (M+1).
Example 539
Synthesis of (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-N-(cyclopropysulf- onyl)-2-fluorobenzamide
##STR01429##
[2491] Following the procedure as described in Example 538 step 3, and making variation as required to replace methylsulfonamide with cyclopropanesulfonamide, the title compound was obtained as a colorless solid (0.04 g, 23%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.33 (d, J=8.7 Hz, 1H), 6.58 (d, J=13.4 Hz, 1H), 4.44-4.31 (m, 1H), 3.36-3.24 (m, 1H), 3.11-2.98 (m, 1H), 2.95-2.84 (m, 1H), 2.23-2.05 (m, 3H), 1.98-1.87 (m, 1H), 1.85-1.57 (m, 2H), 1.43-1.29 (m, 1H), 1.27-1.20 (m, 2H), 1.09 (s, 9H), 1.01-0.92 (m, 2H), 0.84-0.74 (m, 2H), 0.58-0.50 (m, 2H); MS(ES+) m/z 439.2 (M+1).
Example 540
Synthesis of 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl)ethyl)piperazin-1-yl)methyl)-2- -fluoro-N-(methylsulfonyl)benzamide
##STR01430##
[2492] Step 1. Preparation of tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate
##STR01431##
[2494] To a solution of tert-butyl piperazine-1-carboxylate (3.13 g, 16.38 mmol) and 1-(3,5-dichlorophenyl)ethan-1-one (3.50 g, 18.51 mmol) in anhydrous tetrahydrofuran (80 mL) was added titanitium isopropoxide (IV) (10.2 mL, 33.06 mmol) under nitrogen. The resulting mixture was reflux for 18 hours, cooled to -42 .degree. C. and added anhydrous methanol (3.0 mL) and sodium triacetoxyborohydride (7.13 g, 33.66 mmol). After stirring for 5 hours at -42.degree. C., the reaction mixture was added acetic acid (3.85 mL, 67.32 mmol) and allowed to warm to ambient temperature. The reaction mixture was stirred for 18 hours and then refluxed for 16 hours, cooled to ambient temperature and diluted with ethyl acetate (200 mL) and 1.0 M aqueous sodium hydroxide solution (100 mL). The mixture was filtered and the layers were separated. The organic layer was washed with 1.0 M aqueous sodium hydroxide solution (3.times.50 mL) and brine (3.times.50 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-50% ethyl acetate in hexanes) to provide the title compound as an oil (2.70 g, 45%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.21-7.15 (m, 3H), 3.43-3.33 (m, 4H), 3.29 (q, J=6.7 Hz, 1H), 2.45-2.21 (m, 4H), 1.41 (s, 9H), 1.27 (d, J=6.6 Hz, 3H); MS(ES+) m/z 359.1, 361.1 (M+1).
Step 2. Preparation of 1-(1-(3,5-dichlorophenyl)ethyl)piperazine
##STR01432##
[2496] To a solution of tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate (2.70 g, 7.51 mmol) in dichloromethane (100 mL) was added trifluoroacetic acid (25 mL). The resulting solution was stirred at ambient temperature for 1 hour and then concentrated in vacuo. To the residue was added 1.0 M aqueous sodium hydroxide solution (50 mL) and extracted with dichloromethane (2.times.80 mL). The combined organic extracts were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to provide the title compound as a colorless oil (1.80 g, 92%): MS(ES+) m/z 259.1, 261.2 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-(1-(3,5-diclorophenyl) ethyl)-piperazin-1-yl)methyl)-2-fluorobenzoate
##STR01433##
[2498] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 1-(1-(3,5-dichlorophenyl) ethyl)piperazine, the title compound was obtained as a colorless oil (3.53 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.51-7.43 (m, 1H), 7.27-7.09 (m, 2H), 3.61 (s, 2H), 3.34-3.22 (m, 1H), 2.58-2.28 (m, 8H), 1.98-1.85 (m, 1H), 1.55 (s, 9H), 1.33-1.23 (m, 3H), 0.94-0.83 (m, 2H), 0.66-0.53 (m, 2H); MS(ES+) m/z 507.2, 509.2 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl)ethyl) piperazin-1-yl)methyl)-2-fluorobenzoic acid dihydrochloride
##STR01434##
[2500] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl) ethyl)piperazin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.82 g, quant. yield): MS(ES+) m/z 451.0, 453.0 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl)ethyl)piperazin-1-yl) methyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01435##
[2502] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl)ethyl)piperazin-1-yl)methyl)-2- -fluorobenzoic acid dihydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.01, g, 1%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CO.sub.3OD) .delta. 7.50-7.43 (m, 1H), 7.18-7.09 (m, 4H), 3.59 (s, 2H), 3.31-3.19 (m, 4H), 2.56-2.25 (m, 8H), 1.18-1.79 (m, 1H), 1.29-1.20 (m, 3H), 0.90-0.81 (m, 2H), 0.59-0.50 (m, 2H); MS(ES+) m/z 528.1, 530.1 (M+1).
Example 541
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(1-(3,5-dichlorophenyl) ethyl)piperazin-1-yl)methyl)-2-fluorobenzamide
##STR01436##
[2504] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(1-(3,5-dichlorophenyl)ethyl)piperazin-1-yl)methyl)-2- -fluorobenzoic acid dihydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.08 g, 8%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CO.sub.3OD) .delta. 7.47 (d, J=7.9 Hz, 1H), 7.17-7.09 (m, 4H) 3.28-3.18 (m, 3H), 3.01-2.91 (m, 1H), 2.55-2.39 (m, 8H), 1.89-1.78 (m, 1H), 1.35-1.26 (m, 2H), 1.24 (d, J=6.8 Hz, 3H), 1.07-0.98 (m, 2H), 0.89-0.80 (m, 2H), 0.57-0.49 (m, 2H); MS(ES+) m/z 554.1, 556.1 (M+1).
Example 542
Synthesis of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl)methoxy)-5-- cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01437##
[2505] Step 1. Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)azetidine-1-c- arboxylate
##STR01438##
[2507] To a mixture of tert-butyl 3-(hydroxymethyl)azetidine-1-carboxylate (8.96 g, 47.86 mmol), tert-butyl 5-chloro-2,4-difluorobenzoate (14.28 g, 57.43 mmol) anhydrous dimethylsulfoxide (250 mL) was added cesium carbonate (28.10 g, 86.15 mmol). The reaction mixture was heated at 85.degree.0 C. under nitrogen for 6 hours, cooled to ambient temperature and diluted with ethyl acetate (500 mL), washed with water (250 mL), aqueous saturated ammonium chloride solution (2.times.200 mL) and brine (2.times.100 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-25% ethyl acetate in hexanes) to provide the title compound as a colorless solid (19.30 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.83 (d, J=7.7 Hz, 1H), 6.61 (d, J=11.8 Hz, 1H), 4.13-.4.07 (m, 2H), 4.4 (d, J=8.4 Hz, 2H), 3.80 (dd, J=8.6, 5.2 Hz, 2H), 3.07-2.91 (m, 1H), 1.53 (s, 9H), 1.40 (s, 9H); MS(ES+) m/z 416.2, 418.2 (M+1).
Step 2. Preparation of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)azetidin- e-1-carboxylate
##STR01439##
[2509] To a degassed mixture of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)azetidine-1-carboxylate (8.45 g, 20.32 mmol), cyclopropylboronic acid (43.49 g, 40.64 mmol), and potassium phosphate (tribasic, 17.30 g, 81.28 mmol) in toluene (200 mL) and water (20 mL) was added tricyclohexylphosphine tetrafluoroborate (1.50 g, 4.06 mmol) and palladium (II) acetate (0.46 g, 2.03 mmol). The resulting mixture was refluxed under nitrogen for 18 hours, cooled to ambient temperature and filtered. The filtrate was diluted with ethyl acetate (200 mL), washed with water (100 mL), aqueous saturated ammonium chloride solution (100 mL) and brine (100 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-18% ethyl acetate in hexanes) to provide the title compound as a colorless solid (8.35 g, 93%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.37 (d, J=8.3 Hz, 1H), 6.49 (d, J=12.4 Hz, 1H), 4.10-4.01 (m, 4H), 3.86 (dd, J=8.6, 5.5 Hz, 2H), 3.07-2.91 (m, 1H), 1.99-1.87 (m, 1H), 1.54 (s, 9H), 1.40 (s, 9H), 0.90-0.30 (m, 2H), 0.63-0.53 (m, 2H); MS(ES+) m/z 422.2 (M+1).
Step 3. Preparation of methyl 4-(azetidin-3-ylmethoxy)-5-cyclopropyl-2-fluorobenzoate hydrochloride
##STR01440##
[2511] To anhydrous methanol (80 mL) under nitrogen was added thionyi chloride (5.00 mL, 68.97 mmol) dropwise at 0.degree. C. The resulting mixture was stirred at 0.degree. C. for 1 hour and then added a solution of tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)azetidin- e-1-carboxylate (8.35 g, 19.81 mmol) in anhydrous methanol (10 mL). The resulting mixture was stirred at 0.degree. C. for 2 hours, at ambient temperature for 18 hours and refluxed for 4 hours, and then concentrated in vacuo. The residue was dissolved in anhydrous toluene (50 mL) and concentrated in vacuo to provide the title compound as a colorless solid (6.26 g, quant. yield): MS(ES+) m/z 280.2 (M+1).
Step 4. Preparation of methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01441##
[2513] Following the procedure as described in Example 525 step 1, and making variation as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with methyl 4-(azetidin-(3-ylmethoxy)-5-cyclopropyl-2-fluorobenzoate hydrochloride and purification by flash chromatography (2:1 of hexanes:ethyl acetate), the title compound was obtained as a colorless oil (0.51 g, 53%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.18 (s, 1H), 7.42 (d, J=8.4 Hz, 1H), 6.56 (d, J=12.4 Hz, 1H), 6.48 (s, 1H), 4.28-4.20 (m, 2H), 4.18 (d, J=5.7 Hz, 2H), 4.02 (d, J=8.2, 5.3 Hz, 2H), 3.86 (s, 3H), 3.34-3.20 (m, 1H), 1.86-1.75 (m, 1H), 0.77-0.69 (m, 2H), 0.60-0.53 (m, 2H); MS(ES+) m/z 459.1, 461.0 (M+1).
Step 5. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl) azetidin-3-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01442##
[2515] Following the procedure as described in Example 525 step 2, and making variation as required to replace methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.49 g, quant. yield): MS(ES+) m/z 445.1, 447.0 (M+1).
Step 6. Preparation of -((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01443##
[2517] Following the procedure asd described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl)methoxy)-5-- cyclopropyl-2-fluorobenzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, the title compound was obtained as a colorless solid (0.12 g, 42%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.70 (d, J=14.0 Hz, 1H), 8.17 (s, 1H), 7.54 (d, J=9.1 Hz, 1H), 6.59 (d, J=14.2 Hz, 1H), 6.48 (s, 1H), 4.28-4.19 (m, 4H), 4.06-3.98 (m, 2H), 3.38 (s, 3H), 3.33-3.23 (m, 1H), 1.87-1.76 (m, 1H), 0.80-0.72 (m, 2H), 0.61-0.54 (m, 2H); MS(ES+) m/z 522.0, 524.0 (M+1).
Example 543
Synthesis of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01444##
[2519] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenxy)piperidin-1-yl)methyl)-2-fluo- robenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)azetidin-3-yl)methoxy)-5-- cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.07 g, 23%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.66 (d, J=15.9 Hz, 1H), 8.18 (s, 1H), 7.56 (d, J=8.9 Hz, 1H), 6.59 (d, J=14.1 Hz, 1H), 6.49 (s, 1H), 4.30-4.19 (m, 4H), 4.06-3.98 (m, 2H), 3.35-3.22 (m, 1H), 3.12-3.02 (m, 1H), 1.89-1.77 (m, 1H), 1.47-1.39 (m, 2H), 1.17-1.08 (m, 2H), 0.81-0.72 (m, 2H), 0.64-0.54 (m, 2H); MS(ES+) m/z 548.0, 550.0 (M+1).
Example 544
Synthesis of 5-cyclopropyl-4-(3,5-dichlorophenoxy)azetidin-1-) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01445##
[2520] Step 1. Preparation of tert-butyl 3-((methylsulfonyl)oxy)azetidin-1-carboxylate
##STR01446##
[2522] Following the procedure as described in Example 519 step 2, and making variation as required to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-hydroxypiperidin-1-yl)methyl)benzoate with tert-butyl 3-hydroxyazetidine-1-carboxylate, the title compound was obtained as a colorless oil (16.76 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 5.18-5.08 (m, 1H), 4.21 (dd, J=11.1, 6.7 Hz, 2H), 4.02 (dd, J =4.1, 11.0 Hz, 2H), 3.00 (s, 3H), 1.37 (s, 9H).
Step 2. Preparation of tert-butyl 3-(3,5-dichlorphenoxy)azetidin-1-carboxylate
##STR01447##
[2524] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 3,5-dichlorophenol, and to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((methylsulfonyl)oxy)piperidin-1-yl)methyl)b- enzoate with tert-butyl 3-((methylsulfonyl)oxy)azetidin-1-carboxylate, the title compound was obtained as an colorless solid (7.06 g, 97%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 6.98-6.93 (m, 1H), 6.62-6.57 (m, 2H), 4.84-4.75 (m, 1H), 4.26 (dd, J=9.8, 6.4 Hz, 2H), 3.94 (dd, J=9.9, 4.1 Hz, 2H), 1.41 (s, 9H); MS(ES+) m/z 262.1, 264.1 (M-55).
Step 3. Preparation of 3-(3,5-dichlorophenoxy)azetidine
##STR01448##
[2526] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate with tert-butyl 3-(3,5-dichlorophenoxy)azetidine-1-carboxylate, the title compound was obtained as an colorless oil (4.84 g, quant. yield): MS(ES+) m/z 218.1, 220.1 (M+1).
Step 4. Preparation of tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)azetidin-1-yl)methyl)-2-fluorobe- nzoate
##STR01449##
[2528] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 3-(3,5-dichlorophenoxy)azetidine, the title compound was obtained as a colorless oil (1.81 g, 65%): .sup.1H NMR (300 MHz, CDC.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.09 (d, J=11.7 Hz, 1H), 6.95-6.92 (m, 1H), 6.67-6.62 (m, 2H), 4.81-4.71 (m, 1H), 3.91-3.80 (m, 4H), 3.22-3.11 (m, 2H), 1.90-1.76 (m, 1H), 1.55 (s, 9H), 0.96-0.85 (m, 2H), 0.66-0.55 (m, 2H); MS(ES+) m/z 466.1, 468.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)azetidin-1-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01450##
[2530] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy) azetidin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.73 g, quant. yield): MS(ES+) m/z 410.0, 412.0 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)azetidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01451##
[2532] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)azetidin-1-yl)methyl)-2-fluorobe- nzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.60 g, 99%): .sup.1H NMR (300 MHz, CDCl.sub.3+50% CD.sub.3OD) .delta. 8.00-7.93 (m, 1H), 7.78-7.68 (m, 1H), 7.55-7.49 (m, 1H), 7.26-7.18 (m, 2H), 5.66-5.55 (m, 1H), 5.24-5.08 (m, 4H), 4.60-4.49 (m, 2H), 3.84 (s, 3H), 2.46-2.35 (m, 1H), 1.62-1.49 (m, 2H), 1.26-1.16 (m, 2H); MS(ES+) m/z 487.0, 489.0 (M+1).
Example 545
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-((3-(3,5-dichlorophenoxy)azetidin- -1-yl) methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01452##
[2534] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)azetidin-1-yl)methyl)-2-fluorobe- nzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.40 g, 64%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.48 (d, J=7.1 Hz, 1H), 7.17 (d, J =10.8 Hz, 1H), 7.00-6.95 (m, 1H), 6.67-6.62 (m, 2H), 5.10-5.00 (m, 1H), 4.67-4.66 (m, 2H), 4.59 (s, 2H), 3.99-3.90 (m, 2H), 3.05-2.94 (m, 1H), 1.88-1.76 (m, 1H), 1.38-1.30 (m, 2H), 1.12-0.96 (m, 4H), 0.69-0.62 (m, 2H) (Note: exchangeable protons not observed.); MS(ES+) m/z 513.0, 515.0 (M+1).
Example 546
Synthesis of 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoracetic acid salt
##STR01453##
[2535] Step 1. Preparation of tert-butyl 4-(2,4-dichlorophenoxy)piperidine-1-carboxylate
##STR01454##
[2537] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 2,4-dichlorophenol, and to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-((methylsulfonyl)oxy)piperidin-1-yl)methyl)b- enozoate with tert-butyl 4-((methylsulfonyl)oxy)piperidine-1-carboxylate, the title compound was obtained as an colorless solid (3.16 g, 81%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.33 (d, J=2.4 Hz, 1H), 7.12 (dd, J=8.7, 2.6 Hz, 1H), 6.84 (d, J=8.7 Hz, 1H), 4.50-4.41 (m, 1H), 3.60 (ddd, J=1.13, 8.0, 3.9 Hz, 2H), 3.48-3.35 (m, 2H), 1.91-1.71 (m, 4H), 1.42 (s, 9H); MS(ES+) m/z 290.1, 292.1 (M-t-Bu+2H).
Step 2. Preparation of 4-(2,4-dichlorophenoxy)piperidine
##STR01455##
[2539] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate with tert-butyl 4-(2,4-dichlorophenoxy)piperidine-1-carboxylate, the title compound was obtained as an colorless oil (1.96 g, 87%): MS(ES+) m/z 246.1, 248.1 (M+1).
Step 3. Preparation of tert-butyl 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluorobenzoate
##STR01456##
[2541] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(2,4-dichlorophenoxy)piperidine, the title compound was obtained as a colorless oil(2.76 g, 93%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.34 (d, J=2.3 Hz, 1H), 7.21-7.10 (m, 2H), 6.85 (d, J=8.9 Hz, 1H), 4.4-4.30 (m, 1H), 3.64 (s, 2H), 2.78-2.67 (m, 2H), 2.42-2.30 (m, 2H), 2.01-1.80 (m, 5H), 1.56 (s, 9H), 0.94-0.86 (m, 2H), 0.65-0.56 (m, 2H); MS(ES+) m/z 494.1, 496.1 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01457##
[2543] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy) piperidin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (2.65 g, quant. yield): MS(ES+) m/z 438.1, 440.1 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(2,4-dichlorophenoxy)piperidin-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01458##
[2545] Following the procedure as described in Example 53 step 5 and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.25 g, 40%): .sup.1H NMR (300 MHz, CD.sub.3+10% CD.sub.3OD) .delta. 7.42 (d, J=7.1 Hz, 1H), 7.36-7.28 (m, 2H), 7.16-7.09 (m, 1H), 6.86-6.80 (m, 1H), 4.69-4.59 (m, 2H), 3.40-3.17 (m, 7H), 2.27-2.00 (m, 4H), 1.89-1.77 (m, 1H), 1.06-0.95 (m, 2H), 0.70-0.61 (m, 2H); MS(ES+) m/z 515.0, 517.0 (M+1).
Example 547
Synthesis of 5-cyclopropyl-N-(cyclopropyl-N-(cyclopropylsulfonyl)-4-((4-(2,4-dichlorop- henoxy) piperidin-1-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01459##
[2547] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-((4-(2,4-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluorob- enzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.33 g, 50%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.49-7.43 (m, 1H), 7.37-7.28 (m, 2H), 7.16-7.10 (m, 1 H), 6.87-6.80 (m, 1H), 4.69-4.61 (m, 1H), 4.43 (s, 2H), 3.48-3.15 (m, 4H), 3.04-2.92 (m, 1H), 2.30-2.14 (m, 2H), 2.13-2.01 (m, 2H), 1.89-1.77 (m, 1H), 1.39-1.28 (m, 2H), 1.13-0.94 (m, 4H), 0.72-0.63 (m, 2H); MS(ES+) m/z 541.1, 543.1 (M+1).
Example 548
Synthesis of 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01460##
[2548] Step 1. Preparation of tert-butyl 4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidine-1-carboxylate
##STR01461##
[2550] Following the procedure as described in Example 519 step 3, and making variation as required to replace 4-chloro-3-(trifluoromethyl)phenol with 3-chloro-5-(trifluoromethoxy)phenol, and to replace tert-butyl 5-cyclopropyl-2-fluoro-4-((4-(methylsulfonyl)oxy)piperidin-1-yl) methyl)benzoate with tert-butyl 4-(methylsulfonyl)oxy)piperidine-1-carboxylate, the title compound was obtaitied as an colorless solid (2.41 g, 65%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 6.82-6.78 (m, 2H), 6.64-6.61 (m, 1H), 4.46-4.37 (m, 1H), 3.64 (ddd, J=13.4, 7.6, 3.9 Hz, 2H), 3.33 (ddd, J=13.5, 7.6, 3.9 Hz, 2H), 1.95-1.82 (m, 2H), 1.77-1.64 (m, 2H), 1.44 (s, 9H); MS(ES+) m/z 340.1, 342.1 (M-t-Bu+2H).
Step 2. Preparation of 4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidine
##STR01462##
[2552] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate with tert-butyl 4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidine-1-carboxylate, the title compound was obtained as an colorless oil (1.55 g, 86%): MS(ES+) m/z 296.1, 298.1 (M+1).
Step 3. Preparation of tert-butyl 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01463##
[2554] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 4-(3-chloro-5-(trifluoromethoxy) phenoxy)piperidine, the title compound was obtained as a colorless oil (2.15 g, 76%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.48 (d, J=7.1 Hz, 1H), 7.17 (d, J=11.6 Hz, 1H), 6.84-6.76 (m, 2H), 6.67-6.61 (m, 1H), 4.35-4.26 (m, 1H), 3.64 (s, 2H), 2.76-2.64 (m, 2H), 2.41-2.29 (m, 2H), 2.03-1.89 (m, 3H), 1.87-1.74 (m, 2H), 1.56 (s, 9H), 0.96-0.87 (m, 2H), 0.65-0.57 (m,2H); MS(ES+) m/z 544.2, 546.2 (M+1).
Step 4. Preparation of 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01464##
[2556] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate tert-butyl 4-((4-(3-chloro-5-(trifluoromethoxy) phenoxy)piperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (2.07 g, quant. yield): MS(ES+) m/z 488.1, 490.1 (M+1).
Step 5. Preparation of 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01465##
[2558] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlororphenoxy)piperidin-1-yl)methyl)-2-fl- uorobenzoic acid with 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl)methyl)-5-cycl- opropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.15 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.42 (d, J=7.3 Hz, 1H), 7.33 (d, J=11.1 Hz, 1H), 6.84-6.78 (m, 2H), 6.65-6.60 (m, 1H), 4.64-4.55 (m, 1H), 4.41 (s, 2H), 3.45-3.33 (m, 2H), 3.30 (s, 3H), 3.24-3.11 (m, 2H), 2.33-2.19 (m, 2H), 2.15-2.03 (m, 2H), 1.91-1.80 (m, 1H), 1.08-0.99 (m, 2H), 0.71-0.64 (m, 2H); MS(ES+) m/z 565.2, 567.2 (M+1).
Example 549
Synthesis of 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01466##
[2560] Following the procedure as described in Example 53 step 5, and making variation as required tO replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-(3-chloro-5-(trifluoromethoxy)phenoxy)piperidin-1-yl)methyl)-5-cycl- opropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtaned as a colorless solid (0.15 g, 21%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.43 (d, J=7.0 Hz, 1H), 7.32 (d, 10.9 Hz, 1H), 6.82-6.77 (m, 2H), 6.64-6.59 (m, 1H), 4.62-4.57 (m, 1H), 4.41 (s, 2H), 3.35-3.25 (m, 2H), 3.23-3.09 (m, 2H), 3.04-2.93 (m, 1H), 2.32-2.17 (m, 2H), 2.13-2.01 (m, 2H), 1.91-1.79 (m, 1H), 1.37-1.28 (m, 2H), 1.11-0.97 (m, 4H), 0.71-0.62 (m, 2H); MS(ES+) m/z 591.1, 593.1 (M+1).
Example 550
Synthesis of 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(meth- ylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01467##
[2561] Step 1. Preparation of tert-butyl (1R,3r,5S)-3-hydroxy-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01468##
[2563] To a mixture of (1R,3r,5S)-8-azabicyclo[3.2.1]octan-3-ol (4.20 g, 33.02 mmol) and 1.0 M aqueous sodium hydroxide solution (50 mL, 50.0 mmol) in tetrahydrofuran (100 mL) was added di-tert-butyl dicarbonate (8.65 g, 39.62 mmol). The resulting mixture was stirred at ambient temperature for 18 hours, and diluted with diethyl ether (200 mL); washed with brine (4.times.70 mL); dried over anhydrous sodium sulfate, filtered concentrated in vacuo. The residue was triturated with hexanes to provide the title compound as a colorless solid (6.87 g, 91%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 4.24-4.02 (m, 2.19-1.95 (m, 4H), 1.95-1.83 (m, 2H), 1.76-1.59 (m, 3H), 1.41 (s, 9H).
Step 2. Preparation tert-butyl (1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01469##
[2565] Following the procedure as described in Example 521 step 1, and making variation as required to replace tert-butyl 4-hydroxypiperidine-1-carboxylate with tert-butyl (1R,3r,5S)-3-hydroxy-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as a colorless solid (1.57 g, 84%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.20 (s, 1H), 6.95 (s, 1H), 5.37-5.29 (m, 1H), 4.29-4.08 (m, 2H), 2.26-1.82 (m, 8H), 1.45 (s, 9H); MS(ES+) m/z 351.1, 353.1 (M-55).
Step 3. Preparation of (1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octane
##STR01470##
[2567] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-(3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate with tert-butyl (1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-8-arahicyclo- [3.2.1]octane-8-carboxylate, the title compound was obtained as an colorless oil (1.18 g, quant. yield): MS(ES+) m/z 307.2, 309.2 (M+1).
Step 4. Preparation of tert-butyl 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01471##
[2569] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidin, trifluoroacetic acid salt with (1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl) pyridin-2-yl)oxy)-8-azabicyclo[3.2.1]octane, the title compound was obtained as a colorless oil (1.62 g, 76%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.20 (s, 1H), 7.47 (d, J=7.3 Hz, 1H), 7.32 (d, J=12.1 Hz, 1H), 6.95 (s, 1H), 5.30-5.24 (m, 1H), 3.67 (s, 2H), 3.19-3.12 (m, 2H), 2.23-2.13 (m, 2H), 2.10-2.00 (m, 4H), 1.95-1.82. (m, 3H), 1.56 (s, 9H), 0.93-0.84 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 555.2, 557.2 (M+1).
Step 5. Preparation of 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01472##
[2571] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl) pyridin-2-yl)oxy)-8-aziabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-- fluorobenzoate, the title compound was obtained as a colorless solid (1.56 g, quant. yield): MS(ES+) m/z: 499.0, 501.0 (M+1).
Step 6. Preparation of 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyrdin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(meth- ylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01473##
[2573] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-8-azabic- yclo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.14 g, 20%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 8.18 (s, 1H), 7.50-7.37 (m, 2H), 6.94 (s, 1H), 5.37-5.27 (m, 1H), 4.40 (d, 2H), 3.98-3.83 (m, 2H), 3.29 (s, 3H), 2.80-2.63 (m, 2H), 2.52-2.41 (m, 2H), 2.38-2.25 (m, 2H), 2.19-2.08 (m, 2H), 1.86-1.75 (m, 1H), 1.05-0.95 (m, 2H), 0.71-0.62 (m, 2H); MS(ES+) m/z 576.1, 578.1 (M+1).
Example 551
Synthesis of 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-N-(cyclopropylsu- lfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01474##
[2575] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-(((1R,3r,5S)-3-((5-chloro-4-(trifluoromethyl)phyridin-2-yl) oxy-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.16 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 8.18 (s, 1H), 7.51-7.40 (m, 2H), 6.94 (s, 1H), 5.36-5.28 (m, 1H), 4.38 (s, 2H), 3.95-3.85 (m, 2H), 3.04-2.94 (m, 1H), 2.76-2.63 (m, 2H), 2.52-2.41 (m, 2H), 2.37-2.23 (m, 2H), 2.20-2.07 (m, 2H), 1.87-1.74 (m, 1H), 1.39-1.29 (m, 2H), 1.11-0.93 (m, 4H), 0.72-0.63 (m, 2H); MS(ES+) m/z 602.1, 604.1 (M+1).
Example 552
Synthesis of 4-(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(meth- ylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01475##
[2576] Step 1. Preparation of tert-butyl (1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octane-8-carboxylate
##STR01476##
[2578] Following the procedure as described in Example 521 step 1, and making variation as required to replace 2,5-dichloro-4-(trifluoromethyl)pryidine with 3-chloro-2-fluoro-5-(trifluoromethyl)pyridine and tert-butyl 4-hydroxypiperidine-1-carboxylate with tert-butyl (1R,3r,5S)-3-hydroxy-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as a colorless solid (1.22 g, 34%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.77 (s, 1H), 7.71 (s, 1H), 5.53-5.46 (m, 1H), 4.31-4.12 (m, 2H), 2.30-2.08 (m, 4H), 2.05-1.81 (m, 4H), 1.45 (s, 9H); MS(ES+) m/z 351.1, 353.1 (M-55).
Step 2. Preparation of (1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octane
##STR01477##
[2580] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-(3,5-dichlorophenyl)ethly)piperazine-1-carboxylate with tert-butyl (1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin -2-yl)oxy)-8-azabicyclo[3.2.1]octane-8-carboxylate, the title compound was obtained as an colorless oil (0.39 g, 42%): MS(ES+) m/z 307.0, 309.0 (M+1).
Step 3. Preparation of tert-butyl 4-(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01478##
[2582] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with (1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl) pyridin-2-yl)oxy)-8-azabicyclo[3.2.1]octane, the title compound was obtained as a colorless oil (0.68 g, 97%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.30-8.26 (m, 1H), 7.82 (d, J=2.2 Hz, 1H), 7.48 (d, J=7.3 Hz, 1H), 7.34 (d, J=12.1 Hz, 1H), 5.44 (t, J=5.2 Hz, 1H), 3.68 (s, 2H), 3.21-3.13 (m, 2H), 2.27-2.16 (m, 4H), 2.10-1.99 (m, 2H), 1.96-1.84 (m, 3H), 1.56 (s, 9H), 0.93-0.85 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 555.2, 557.2 (M+1).
Step 4. Preparation of 4-(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01479##
[2584] Following the procedure as described in Example 517 step 2, and making variation as required to replace rept-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with in tert-butyl 4-(((1R,3r,5S)-3-((3-chloro-5-trifluoromethyl) pyridin-2-yl)oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-f- luorobenzoate, the title compound was obtained as a colorless solid (0.66 quant. yield): MS(ES+) m/z 499.1, 501.1 (M+1).
Step 5. Preparation of 4-(((1R3r,5S)-3-((3-chloro-5-trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(meth- ylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01480##
[2586] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with -(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.12 g, 29%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 8.24 (s,1H), 7.83 (s, 1H), 7.50-7.41 (m, 2H), 5.53-5.46 (m, 1H), 4.42 (s, 2H), 3.97-3.87 (m, 2H), 3.30 (s, 3H), 2.80-2.68 (m, 2H), 2.66-2.54 (m, 2H), 2.38-2.25 (m, 2H), 2.20-2.09 (m, 2H), 1.87-1.75 (m, 1H), 1.05-0.95 (m, 2H), 0.72-0.63 (m, 2H); MS(ES+) m/z 576.0, 578.0 (M+1).
Example 553
Synthesis of 4-(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl) oxy)-8-azabicyclo[3.2.1]octan-8-yl)methyl)-5-cyclopropyl-2-fluoro-N-(meth- ylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01481##
[2588] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with -(((1R,3r,5S)-3-((3-chloro-5-(trifluoromethyl)pyridin-2-yl)oxy)-8-azabicy- clo[3.2.1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.12 g, 27%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 8.24 (s, 1H), 7.83 (s,1H), 7.52-7.41 (m, 2H), 5.53-5.46 (m, 1H), 4.42 (s, 2H), 3.97-3.87 (m, 2H), 3.05-2.95 (m, 1H), 2.80-2.68 (m, 2H), 2.66-2.54 (m, 2H), 2.38-2.25 (m, 2H), 2.20-2.09 (m, 2H), 1.87-1.75 (m, 1H), 1.38-1.29 (m, 2H), 1.13-0.94 (m, 1H), 0.73-0.62 (m, 2H); MS(ES+) m/z 602.1, 604.0 (M+1).
Example 554
Synthesis of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01482##
[2589] Step 1. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)piperidin-1-carboxylate
##STR01483##
[2591] Following the procedure as described in Example 542 step 1, and making variation as required to replace tert-butyl 3-(hydroxymethyl)azetidine-1-carboxylate with tert-butyl 4-(hydroxymethyl)piperidine-1-carboxylate, the title compound was obtained as a colorless solid (7.39 g, 69%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.85 (d, J=7.6 Hz, 1H), 6.59 (d, J=11.9 Hz, 1H), 4.23-4.05 (m, 2H), 3.84 (d, J=6.2 Hz, 2H), 2.81-2.64 (m, 2H), 2.10-1.94 (m, 1H), 1.87-1.77 (m, 2H), 1.55 (s, 9H), 1.44 (s, 9H), 1.36-1.20 (m, 2H); MS(ES+) m/z 444.1, 446.2 (M+1).
Step 2. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)piperidine-1-carboxylate
##STR01484##
[2593] Following the procedure as described in Example 542 step 2, and making variation as required to replace tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)azetidine-1-c- arboxylate with tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)piperidine-1-- carboxylate, the title compound was obtained as a colorless solid (8.12 g, 9.7%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.35 (d, J=8.3 Hz, 1H), 6.47 (d, J=12.4 Hz, 1H), 4.23-4.04 (m, 2H), 3.81 (d, J=6.2 Hz, 2H), 2.81-2.04 (m, 2H), 2.05-1.91 (m, 2H), 1.85-1.75 (m, 2H), 1.53 (s, 9H), 1.43 (s, 9H), 1.38-1.26 (m, 2H), 0.89-0.81 (m, 2H), 0.63-0.56 (m, 2H); MS(ES+) m/z: 450.2 (M+1).
Step 3. Preparation of methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride
##STR01485##
[2595] Following the procedure as described in Example 542 step 3, and making variation as required to tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)azetidin- e-1-carboxylate with tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)piperidi- ne-1-carboxylate, the title compound was obtained as a colorless solid (6.21 g, quant. yield): MS(ES+) m/z 308.2 (M+1).
Step 4. Preparation of methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01486##
[2597] Following the procedure as described in Example 525 step 1, and making variation as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride and purification by flash chromatography, the title compound was obtained as a colorless oil (0.71 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.19 (s, 1H), 7.42 (d, J=8.4 Hz, 1H), 6.86 (s, 1H), 6.52 (d, J=12.5 Hz, 1H), 4.41-4.31 (m, 2H), 3.90-3.83 (m, H), 2.94 (d, J=13.0, 2.6 Hz, 2H), 2.23-2.08 (m, 1H), 2.01-1.90 (m, 3H), 1.44 (dq, J=12.4, 4.0 Hz, 2H), 0.94-0.82 (m, 2H), 0.67-0.57 (m, 2H); MS(ES+) m/z 487.1, 489.1 (M+1).
Step 5. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl)methoxy)-5- -cyclopropyl-2-fluorobenzoic acid
##STR01487##
[2599] Following the procedure as described in Example 525 step 2, and making variation as required, to replace methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl)me- thoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.49 g, 71%): MS(ES+) m/z 473.1, 475.1 (M+1).
Step 6. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01488##
[2601] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl)methoxy)-5- -cyclopropyl-2-fluorobenzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, the title compound was obtained as a colorless solid (0.15 g, 55%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.68 (br s, 1H), 8.20 (s, 1H), 7.56 (d, J=8.7 Hz, 1H), 6.86 (s, 1H), 6.55 (d, J=14.1 Hz, 1H), 4.41-4.32 (m, 2H), 3.89 (d, J=6.28 Hz, 1H), 3.39 (s, 3H), 2.95 (dt, J=13.0, 2.6 Hz, 2H), 2.25-2.10 (m, 1H), 2.05-1.91 (m, 3H), 1.45 (dq, J=12.5, 3.6 Hz, 2H), 0.95-0.86 (m, 2H), 0.68-0.58 (m, 2H); MS(ES+) m/z 550.0, 552.0 (M+1).
Example 555
Synthesis of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl) methoxy)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01489##
[2603] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)piperidin-4-yl)methoxy)-5- -cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.14 g, 49%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.67 (br s, 1H), 8.20 (s, 1H), 7.57 (d, J=9.1 Hz, 1H), 6.86 (s, 1H), 6.55(d, J=14.1 Hz, 1H), 4.41-4.31 (m, 2H), 3.89 (d, J=6.1 Hz, 2H), 3.12-3.03 (m, 1H), 3.00-2.89 (m, 2H), 2.25-2.09 (m, 1H), 2.05-1.90 (m, 3H), 1.53-1.37 (m, 4H), 1.17-1.07 (m, 2H), 0.94-0.85 (m, 2H), 0.67-0.59 (m, 2H); MS(ES+) m/z 576.0, 578.0 (M+1).
Example 556
Synthesis of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01490##
[2604] Step 1. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-4-methylpiperidine-1-carboxylate
##STR01491##
[2606] Following the procedure as described in Example 542 step 1, and making variation as required to replace tert-butyl 3-(hydroxymethyl)azetidine-1-carboxylate with tert-butyl 4-(hydroxymethyl)-4-methylpiperidine-1-carboxylate, the title compound was obtained as a colorless solid (15.19 g, 70%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 77.83 (d, J=7.6 Hz, 1H), 6.58 (d, J=11.9 Hz, 1H), 3.76-3.62 (m, 4H), 3.22-3.10 (m, 2H), 1.65-1.56 (m, 2H), 1.54 (s, 9H), 1.48-1.39 (m, 11H), 1.13 (s, 3H); MS(ES+) m/z 458.1, 460.0 (M+1).
Step 2. Preparation of tert-butyl 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy) methyl)-4-methylpiperidine-1-carboxylate
##STR01492##
[2608] Following the procedure as described in Example 542 step 2, and making variation as required to replace tert-butyl 3-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy)methyl)azetidine-1-c- arboxylate with tert-butyl 4-((4-(tert-butoxycarbonyl)-2-chloro-5-fluorophenoxy) methyl)-4-methylpiperidine-1-carboxylate, the title empoond was obtained as a colorless solid (9.91 g, 69%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.38 (d, J=8.4 Hz, 1H), 6.47 (d, J=12.5 Hz, 1H), 3.77-3.61 (m, 4H), 3.23-3.09 (m, 2H), 2.00-1.88 (m, 1H), 1.69-1.57 (m, 2H), 1.54 (s, 9H), 1.48-1.38 (m, 11H), 1.13 (s, 3H), 0.89-0.82 (m, 2H), 0.62-0.55 (m, 2H); MS(ES+) m/z: 464.2 (M+1).
Step 3. Preparation of methyl 5-cyclopropyl-2-fluoro-4-((4-methylpiperidin-4-yl) methoxy)benzoate hydrochloride
##STR01493##
[2610] Following the procedure as described in Example 542 step, 3, and making variation as required to tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)azetidin- e-1-carboxylate with tert-butt 4-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)-4-methy- lpiperidine-1-carboxylate, the title compound was obtained as a colorless solid (7.65 g, quant. yield): MS(ES+) m/z 322.2 (M+1).
Step 4. Preparation of methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01494##
[2612] Following the procedure as described in Example 525 step 1, and making variation as required to replace methyl 5-cyclopropyl-2-fluoro-4-((3-methylazetidin-3-yl)methoxy)benzoate hydrochloride with methyl 5-cyclopropyl-2-fluoro-4-((4-methylpiperidin-4-yl)methoxy)benzoate hydrochloride, the title compound was obtained as a colorless oil (0.87 g, 67%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.19 (s, 1H), 7.45 (d, J=8.4 Hz, 1H), 6.84 (s, 1H), 6.52 (d, J=12.5 Hz, 1H), 3.94-3.83 (m, 5H), 3.73 (s, 2H), 3.43-3.31 (m, 2H), 1.99-1.86 (m, 1H), 1.84-1.72 (m, 2H), 1.63-1.52 (m, 2H), 1.20 (s, 3H), 0.88-0.79 (m, 2H), 0.64-0.55 (m, 2H); MS(ES+) m/z 501.1, 503.1 (M+1).
Step 5. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01495##
[2614] Following the procedure as described in Example 525 step 2, and making variation as required to replace methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl)me- thoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl)m- ethoxy)-5-cyclopropyl-2-fluorobenzoate, the title emnponad was obtained as a colorless solid (0.85 g, quant. yield): MS(ES+) m/z 487.1, 489.1 (M+1).
Step 6. Preparation of 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01496##
[2616] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, the title compound was obtained as a colorless solid (0.40 g, 82%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.71 (br s, 1H), 8.18 (s, 1H), 7.65 (d, J=9.1 Hz, 1H), 8.84 (s, 1H), 6.56 (d, J=14.1 Hz, 1H), 3.95-3.84 (m, 2H), 3.76 (s, 2H), 3.42-3.30 (m, 5H), 1.98-1.87 (m, 1H), 1.84-1.71 (m, 2H), 1.63-1.53 (m, 2H), 1.21 (s, 3H), 0.91-0.82 (m, 2H), 0.64-0.56 (m, 2H); MS(ES+) m/z 564.0, 566.0 (M+1).
Example 557
Synthesis of 4-((1-(5chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methyoxy)5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01497##
[2618] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, the title compound was obtained as a colorless solid (0.35 g, 68%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.67 (d, J=13.6 Hz, 1H), 8.19 (s, 1H), 7.58 (d, J=9.1 Hz, 1H), 6.84 (s, 1H), 6.55 (d, J=14.1 Hz, 1H), 3.97-3.84 (m, 2H), 3.76 (s, 2H), 3.43-3.29 (m, 2H), 3.13-3.01 (m, 1H), 1.99-1.87 (m, 1H), 1.85-1.72 (m, 2H), 1.64-1.51 (m, 2H), 1.46-1.38 (m, 2H), 1.21 (s, 3H), 1.16-1.07 (m, 2H), 0.91-0.81 (m, 2H), 0.65-0.56 (m, 2H); MS(ES+) m/z 590.0, 592.0 (M+1).
Example 558
Synthesis of 5-cyclopropyl-4-(((1S,4S)-5-((3,5-dichlorophenyl)sulfonyl)-2,5-diazabicyc- lo[2.2.1]heptan-2-yl)methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01498##
[2619] Step 1. Preparation of tert-butyl (1S,4S)-5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2,5-diaz- abicyclo[2.2.]heptane-2-carboxylate
##STR01499##
[2621] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoromacetic acid salt with tert-butyl (1S,4S)-2,5-diazabicyclo[2.2.1]heptane-2-carboxylate, the title compound was obtained as a colorless solid (2.61 g, 67%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46 (d, J=7.1 Hz, 1H), 7.18 (d, J=12.1 Hz, 1H), 4.41-4.22 (m, 1H), 3.94-3.77 (m, 2H), 3.65-3.39 (m, 2H), 3.23-3.13 (m, 1H), 2.98-2.82 (m, 1H), 2.75-2.43 (m, 1H), 1.93-1.80 (m, 2H), 1.77-1.60 (m, 1H), 1.55 (s, 9H), 1.44 (s, 9H), 0.94-0.86 (m, 2H), 0.64-0.55 (m, 2H); MS(ES+) m/z 447.2 (M+1).
Step 2. Preparation of methyl 4-((((1S,4S)-2,5-diazabicyclo[2.2.1]heptan-2-yl) methyl)-5-cyclopropyl-2-fluorobenzoate hydrochloride
##STR01500##
[2623] Following the procedure as described in Example 542 step 3, and making variation as required to tert-butyl 3-((4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorophenoxy)methyl)azetidin- e-1-carboxylate with tert-butyl (1S,4S)-5-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2,5-diaz- abicyclo[2.2.1]heptane-2-carboxylate, the title compound was obtained as a colorless solid (2.12 g, 96%): MS(ES+) m/z 305.2 (M+1).
Step 3. Preparation of methyl 5-cyclopropyl-4-(((1S,4S)-5-((3,5-dichlorophenyl) sulfonyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzoate
##STR01501##
[2625] To a mixture of of methyl 4-(((1S,4S)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoate hydrochloride (0.38 g, 1.00 mmol) and 4-dimethyhaminopyridine (0.61 g, 5.00 mmol) in dichloromethane (10 mL) was added 3,5-dichlorobenzenesulfonyl chloride (0.37 g, 1.50 mmol). The reaction mixture was stirred at ambient temperature in a sealed tube for 5 hours, was diluted with ethyl acetate (50 mL); washed with water (40 mL), aqueous saturated sodium bicarbonate solution (50 mL) and brine (50 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-40% ethyl acetate in hexanes) to provide the title compound as a colorless solid (0.51 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.73-7.68 (m, 2H), 7.58-7.50 (m, 2H), 7.11 (d, J=11.8 Hz, 1H), 4.34 (s, 1H), 3.93-3.81 (m, 5H), 3.64 (d, J=9.5 Hz, 1H), 3.49 (s, 1H), 3.10 (d, J=9.5, 2.1 Hz, 1H), 2.89 (dd, J=9.7, 2.1 Hz, 1H), 2.68 (d, J=9.7 Hz, 1H), 1.88-1.76 (m, 2H), 1.30 (d, J=9.7 Hz, 1H), 0.94-0.86 (m, 2H), 0.63-0.56 (m, 2H); MS(ES+) m/z 513.0, 515.0 (M+1).
Step 4. Preparation of 5-cyclopropyl-4-(((1S,4S)-5-((3,5-dichlorophenyl) sulfonyl)-5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzoic acid
##STR01502##
[2627] Following the procedure as described in Example 525 step 2, and making variation required to replace methyl 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-3-methylazetidin-3-yl) methoxy)-5-cyclopropyl-2-fluorobenzoate with methyl 5-cyclopropyl-4-(((1S,4S)-5-((3,5-dichlorophenyl) sulfonyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (0.51 g, quant. yield): MS(ES+) m/z 499.0, 501.0 (M+1).
Step 5. Preparation of 5-cyclopropyl-4-(((1S,4S)-5-((3,5-dichlorophenyl)sulfonyl)-2.5-diazabicyc- lo[2.2.1]heptan-2-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid
##STR01503##
[2629] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.15 g, 43%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.67-7.62 (m, 2H), 7.57-7.53 (m, 1H), 7.48-7.41 (m, 1H), 7.30-7.22 (m, 1H), 4.55-4.33 (m, 3H), 4.24 (s, 1H), 3.84 (d, J=11.1 Hz, 1H), 3.39-3.20 (m, 6H), 2.14 (d, J=11.1 Hz, 1H), 1.86-1.74 (m, 1H), 1.63 (d, J=11.6 Hz, 1H), 1.05-0.95 (m, 2H), 0.69-0.59 (m, 2H); MS(ES+) m/z 575.9, 577.9 (M+1).
Example 559
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1S,4S)-5-((3,5-dichlorophenyl) sulfonyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01504##
[2631] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((1-(5-chloro-4-(trifluoromethyl)pyridin-2-yl)-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzoic acid, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.20 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.64 (d, J=1.7 Hz, 2H), 7.57-7.54 (m, 1H), 7.47 (d, J=7.3 Hz, 1H), 7.258 (d, J=11.4 Hz, 1H), 4.49-4.28 (m, 3H), 4.17 (s, 1H), 3.82 (d, J=11.4 Hz, 1H), 3.35-3.20 (m, 3H), 3.05-2.94 (m, 1H), 2.15-2.05 (m, 1H), 1.86-1.74 (m, 1H), 1.60 (d, J=11.3 Hz, 1H), 1.37-1.29 (m, 2H), 1.22-0.94 (m, 4H), 0.67-0.59 (m, 2H); MS(ES+) m/z 602.3, 603.9 (M+1).
Example 560
Synthesis of 4-(((1R,3r,5s)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.- 1]octan-8-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01505##
[2633] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorphenoxy)piperidin-1-yl)methyl)-2-fluo- robenzoic acid with 4-(((1R,3r,5S)-3-(3-chloro-5-(trifluoromethoxy)phenoxy)-8-azabicyclo[3.2.- 1]octan-8-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.36 g, 51%): .sup.1H NMR (300 MHz, CDCl.sub.3+10% CD.sub.3OD) .delta. 7.47-7.39 (m, 2H), 6.81-6.78 (m, 1H), 6.75-6.72 (m, 1H), 6.56-6.52 (m, 1H), 4.62-4.56 (m, 1H), 4.38 (s, 2H), 3.90 (br s, 2H), 3.29 (s, 3H), 2.75-2.64 (m, 2H), 2.50-2.40 (m, 2H), 2.16-2.24 (m, 2H), 2.13 (d, J=16.4 Hz, 2H), 1.85-1.74 (m, 1H), 1.03-0.95 (m, 2H), 0.70-0.63 (m, 2H); MS(ES+) m/z 591.1, 593.1 (M+1).
Example 561
Synthesis of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethylpiperid- in-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01506##
[2634] Step 1. Preparation of tert-butyl 3,3-dimethyl-4-oxopiperidine-1-carboxylate
##STR01507##
[2636] To a solution of tert-butyl 4-oxopiperidine-1-carboxylate (15.0 g, 75.30 mmol) in anhydrous tetrahydrofuran (400 mL) at 0.degree. C. was added sodium hydride (60% in mineral oil, 6.04 g, 151.00 mmol) under nitrogen. After stirring for 10 minutes, methyl iodide (11.7 mL, 188.00 mmol) was added; the reaction mixture was stirred at 0.degree. C. for 1 hour and stirred at ambient temperature for 3 hours. The reaction mixture was quenched by slowly addition of water (10 mL) and concentrated in vacuo to remove about 300 mL tetrahydrofuran. The residue was diluted with ethyl acetate (300 mL), washed with aqueous saturated ammonium chloride solution (2.times.250 mL) and brine (100 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was triturated with hexanes to provide the title compound as a colorless solid (6.68 g, 39%): .sup.1H NMR (300 MHz, CDC.sub.3) .delta. 3.68 (t, J=6.2 Hz, 2H), 3.39 (s, 2H), 2.45 (t, J=6.2 Hz, 2H), 1.45 (s, 9H), 1.07 (s, 6H).
Step 2. Preparaton of tert-butyl 4-hydroxy-3,5-dimethylpiperidine-1-carboxylate
##STR01508##
[2638] To a solution of tert-butyl 3,3-dimethyl-4-oxopiperidine-1-carboxylate (4.88 g, 21.47 mmol) in anhydrous methanol (50 mL) at 0.degree. C. was added sodium borohydride (0.87 g, 21.97 mmol) portionwise under nitrogen. The resulting mixture was stirred at 0.degree. C. for 1 hour and then stirred at ambient temperature for 2.hours. The reaction mixture was concentrated in vacuo and the residue was diluted with ethyl acetate (200 mL), washed with 1.0 M aqueous hydrochloric acid solution (100 mL) and brine (2.times.70 mL); dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to provide the title compound as a colorless solid (4.92 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 3.92-3.68 (m, 1H), 3.58-3.41 (m, 1H), 3.37 (dd, J=9.2, 4.0 Hz, 1H), 3.07-2.93 (m, 1H), 2.69 (d, J=13.3 Hz, 1H), 1.77-1.65 (m, 1H), 1.60-1.46 (m, 1H), 1.42 (s, 9H), 0.92 (s, 3H), 0.85 (s, 3H); MS(ES+) m/z 230.2 (M+1).
Step 3. Preparation of tert-butyl 4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-3,3-dimethylpiperidine-1-carboxylate
##STR01509##
[2640] To a mixture of tert-butyl 4-hydroxy-3,3-dimethylpiperidine-1-carboxylate (4.58 g, 19.97 mmol) and 2,5-dichloro-4-(trifluoromethyl)pyridine (5.18 g, 23.96 mmol) in anhydrous dimethylsulfoxide (150 mL) was added cesium carbonate (19.52 g, 59.91 mmol). The reaction mixture was heated at 90.degree. C. under nitrogen for 18 hours, cooled to ambient temperature, diluted with ethyl acetate (300 mL), washed with water (150 mL) and brine (3.times.100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by flash chromatography (0-12% ethyl acetate in hexanes) to provide the title compound as a colorless oil (4.55 g, 56%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.18 (s, 1H), 7.01 (s, 1H), 4.91 (dd, J=8.3, 3.8 Hz, 1H), 3.80-3.60 (m, 1H), 3.55-3.43 (m, 1H), 3.25 (ddd, J=13.5, 8.7, 3.9 Hz, 1H), 2.97 (d, J=13.7 Hz, 1H), 1.98-1.83 (m, 1H), 1.73-1.58 (m, 1H), 1.44 (s, 9H), 0.97 (s, 3H), 0.95 (s, 3H); MS(ES+) m/z 353.0, 355.0 (M-55).
Step 4. Preparation of 5-chloro-2-((3,3-dimethylpiperidin-4-yl)oxy)-4-(trifluoromethyl)pyridine
##STR01510##
[2642] Following the procedure as described in Example 540 step 2, and making variation as required to replace tert-butyl 4-(1-((3,5-dichlorophenyl)ethyl)piperazine-1-carboxylate with tert-butyl 4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethylpiperidine-- 1-carboxylate, the title compound was obtained as an colorless oil (3.77 g, quant. yield): MS(ES+) m/z 309.1, 311.1 (M+1).
Step 5. Preparation of tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl) oxy)-3,3-dimethylpiperidin-1-yl)methyl)-5-cyclopropyl-2-fluorobenzoate
##STR01511##
[2644] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with 5-chloro-2-((3,3-dimethylpiperidin-4-yl) oxy)-4-(trifluoromethyl)pyridine, the title compound was obtained as a colorless solid (5.46 g, 79%): .sup.1H NMR (300 MHz, CDC.sub.3) .delta. 8.18 (s, 1H), 7.47 (d, J=7.3 Hz, 1H), 7.17 (d, J=11.9 Hz, 1H), 7.01 (s, 1H), 4.83 (dd, J=8.6, 3.9 Hz, 1H), 3.59 (s, 2H), 2.74-2.62 (m, 1H), 2.43 (d, J=10.9 Hz, 1H), 2.37-2.26 (m, 1H), 2.08-1.91 (m, 3H), 1.83-1.68 (m, 1H), 1.56 (s, 9H), 1.07 (s, 3H), 0.98-0.86 (m, 5H), 0.65-0.58 (m, 2H); MS(ES+) m/z 515.0, 516.9 (M+1).
Step 6. Preparation of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride
##STR01512##
[2646] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 4-((4-((5-chloro-4-(trifluoromethyl) pyridin-2-yl)oxy)-3,3-dimethylpiperidin-1-yl)methyl)-5-cyclopropyl-2-fluo- robenzoate, the title compound was obtained as a colorless solid (1.59 g, quant. yield): MS(ES+) m/z 501.1, 503.1 (M+1).
Step 7. Preparation of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethylpiperid- in-1-yl) methyl)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide, trifluoroacetic acid salt
##STR01513##
[2648] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethylpiperid- in-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.35 g, 34%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 8.45 (s, 1H), 7.49 (d, J=11.2 Hz, 1H) 7.32 (s, 1H), 7.25 (d, J=7.1 Hz, 1H), 5.06-4.91 (m, 1H), 4.43 (br s, 2H), 3.33 (s, 3H), 3.29-2.91 (m, 4H), 2.20-2.04 (m, 2H), 2.02-1.80 (m, 1H), 1.21-0.86 (m, 8H), 0.81-0.68 (m, 2H); MS(ES+) m/z 578.1, 580.1 (M+1).
Example 562
Synthesis of 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethyipiperid- in-1-yl) methyl)-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide, trifluoroacetic acid salt
##STR01514##
[2650] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 4-((4-((5-chloro-4-(trifluoromethyl)pyridin-2-yl)oxy)-3,3-dimethylpiperid- in-1-yl) methyl)-5-cyclopropyl-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.50 g, 47%): .sup.1H NMR (300 MHz, CDC.sub.3+10% CD.sub.3OD) .delta. 8.14 (s, 1H), 7.46 (d, J=7.3 Hz, 1H), 7.39 (d, J=11.2 Hz, 1H), 7.00 (s,1H), 4.99-4.92 (m, 1H), 4.48-4.32 (m, 2H), 3.56-3.32 (m, 1H), 3.21-3.08 (m, 1H), 3.05-2.97 (m, 1H), 2.96-2.88 (m, 1H), 2.87-2.74 (m, 1H), 2.35-2.19 (m, 1H), 2.08-1.95 (m, 1H), 1.92-1.81 (m, 1H), 1.38-1.29 (m, 2H), 1.20-0.89 (m, 10H), 0.70-0.63 (m, 2H); MS(ES+) m/z 604.2, 606.2 (M+1).
Example 563
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1S,4S)-5-((R)-1-(3,5-dichlorop- henyl) ethyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzamide- , trifluoroacetic acid salt
##STR01515##
[2651] Step 1. Preparation of methyl 5-cyclopropyl-4-(((1S,4S)-5-((R)-1-(3,5-dichlorophenyl)ethyl)-2,5-diazabi- cyclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate and methyl 5-cyclopropyl-4-((1S,4S)-5-((S)-1-(3,5-dichlorophenyl) ethyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzoate
##STR01516##
[2653] To a solution of methyl 4-(((1S,4S)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoate dihydrochloride (0.38 g, 1.00 mmol) and 1-(3,5-dichlorophenyl)ethyl 4-methylbenzenesulfonate (0.41 g, 1.20 mmol) in anhydrous dimethylformamide (10 mL) was added potassium carbonate (0.55 g, 4.00 mmol). The reaction mixture was heated at 80.degree. C. in a sealed tube for 6 hours, cooled to ambient temperature, diluted with ethyl acetate (80 mL), washed with water (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by preparative HPLC. The first eluting fraction was arbitrarily assigned as methyl 5-cyclopropyl-4-(((1S,4S)-5-((R)-1-(3,5-dichlorophenyl)ethyl)-2,5-diazabi- cyclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate as a colorless oil (0.05 g, 10%)) and the second eluting fraction was arbitrarily assigned as methyl 5-cyclopropyl-4-(((1S,4S)-5-((S)-(3,5-dichlorophenyl)ethyl)-2,5-di- azabicyclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate as a colorless oil (0.05 g, 10%). Analytical data for methyl 5-cyclopropyl-4-(((1S,4S)-5-((R)-1-(3,5-dichlorophenyl)ethyl)-2,5-diazabi- cyclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.54 (d, J=7.3 Hz, 1H), 7.31-7.17 (m, 4H), 3.97-3.85 (m, 4H), 3.75 (d, J=15.6Hz, 1H), 3.65-3.54 (m, 1H), 3.25 (d, J=11.4 Hz, 2H), 2.94-2.85 (s, 2H), 2.70 (d, J=9.7 Hz, 1H), 2.48 (dd, J=10.2, 2.4 Hz, 1H), 1.93-1.81 (m, 1H), 1.77-1.67 (m, 2H), 1.31 (d, J=6.4 Hz, 3H), 0.96-0.88 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 477.1, 479.1 (M+1). Analytical data for methyl 5-cyclopropyl-4-((1S,4S)-5-((S)-1-(3,5-dichlorophenyl) ethyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzoate: .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.54 (d, J=7.3 Hz, 1H), 7.30-7.17 (m, 4H), 3.93 (d, J=15.7 Hz, 1H), 3.88 (s, 3H), 3.79 (d, J=15.7 Hz, 1H), 3.54 (q, J=5.9 Hz, 1H), 3.48 (s, 1H), 3.25 (s, 1H), 2.88 (d, J=9.9 Hz, 1H), 2.72-2.60 (m, 2H), 2.54 (d, J=9.6 Hz, 1H), 1.93-1.82 (m, 1H), 1.81-1.69 (m, 2H), 1.22 (d, J=5.9 Hz, 3H), 0.96-0.88 (m, 2H), 0.68-0.56 (m, 2H); MS(ES+) m/z 477.1, 479.1 (M+1).
Step 2. Prepatation of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1S,4S)-5-((R)-1-(3,5-dichlorop- henyl) ethyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzamide- , trifluoroacetic acid salt
##STR01517##
[2655] Following the procedure as described in Example 538 step 3, and making variation as required to replace (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate with methyl 5-cyclopropyl-4-(((1S,4S)-5-((R)-1-(3,5-dichlorophenyl)ethyl)-2,5-diazabi- cyclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate, and to replace methylsulfonamide with cyclopropanesulfonamide, and purification by HPLC, the title compound was obtained as a colorless solid (0.07 g, 26%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.64-7.57 (m, 3H), 7.37 (d, J=11.5 Hz, 1H), 7.19 (d, J=7.1 Hz, 1H), 4.37-3.98 (m, 4H), 3.79 (br s, 1H), 3.38-3.23 (m, 2H), 3.10-2.86 (m, 3H), 2.20-2.03 (m, 2H), 2.02-1.92 (m, 1H), 1.52 (d, J=6.2 Hz, 3H), 1.13-1.04 (m, 4H), 0.95-0.87 (m, 2H), 0.70-0.60 (m, 2H); MS(ES+) m/z 566.1, 568.1 (M+1).
Example 564
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((1S,4S)-5-((S)-1-(3,5-dichlorop- henyl) ethyl)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-2-fluorobenzamide- , trifluoroacetic acid salt
##STR01518##
[2657] Following the procedure as described in Example 538 step 3, and making variation as required to replace (R)-4-((1-(tert-butyl)piperidin-3-yl)oxy)-5-cyclopropyl-2-fluorobenzoate with methyl 5-cyclopropyl-4-((1S,4S)-5-((S)-1-(3,5-dichlorophenyl)ethyl)-2,5-diazabic- yclo[2.2.1]heptan-2-yl) methyl)-2-fluorobenzoate, and to replace methylsulfonamide with cyclopropanesulfonamide, and purification by HPLC, the title compound was obtained as a colorless solid (0.07 g, 17%): .sup.1H NMR (300 MHz, DMSO-d.sub.6+5% D.sub.2O) .delta. 7.62 (s, 1H), 7.55 (s, 2H), 7.39 (d, J=11.7 Hz, 1H), 7.21 (d, J=7.1 Hz, 1H), 4.39-4.08 (m, 3H), 3.90 (br s, 2H), 3.39-3.25 (m, 2H), 3.22-3.10 (m, 1H), 3.08-2.99 (m, 1H), 2.98-2.89 (m, 1H), 2.26-2.08 (m, 2H), 2.04-1.93 (m, 1H), 1.41 (d, J=6.5 Hz, 3H), 1.16-1.03 (m, 4H), 0.97-0.87 (m, 2H), 0.73-0.61 (m, 2H) (Note: Exchangeable protons not observed); MS(ES+) m/z 566.2, 568.2 (M+1).
Example 565
Synthesis of 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01519##
[2658] Step 1. Preparation of 1-benzyl-4-(tert-butyl) (S)-2-methylpiperazine-1,4-dicarboxylate
##STR01520##
[2660] To a solution of tert-butyl (S)-3-methylpiperazine-1-carboxylate (6.35 g, 31.71 mmol), triethylamine (4.98 mL, 34.88 mmol) and 4-dimethylaminopyridine (0.10 g, 0.82 mmol) in dichloromethane (80 mL) under nitrogen at 0.degree. C. was added dropwise benzyl chloroformate (8.84 mL, 63.42 mmol). The resulting mixture was stirred at 0.degree. C. for 2 hours and then at ambient temperature for 18 hours, quenched with water (10 mL) and stirred for 1 hour. The mixture was diluted with diluted with ethyl acetate (150 mL), washed with aqueous saturated ammonium chloride solution (2.times.80 mL) and brine (80 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to provide the title compound as an oil (10.45 g, 99%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.39-7.25 (m, 5H), 5.11 (s, 2H), 4.37-4.23 (m, 1H), 4.10-3.71 (m, 3H), 3.15-2.91 (m, 2H), 2.86-2.69 (m, 1H), 1.43 (s, 9H), 1.14 (d, J=6.8 Hz, 3H); MS(ES+) m/z 235.2 (M-99).
Step 2. Preparation of benzyl (S)-2-methylpiperazine-1-carboxylate
##STR01521##
[2662] To a solution of 1-benzyl 4-(tert-butyl) (S)-2-methylpiperazine-1,4-dicarboxylate (10.45 g, 31.25 mmol) in dichloromethane (70 mL) was added trifluoroacetic acid (35 mL). After stirring at ambient temperature for 5 hours, the reaction mixture was concentrated in vacuo and the residue was dissolved in diethyl ether (80 mL) and extracted with 1.0 M aqueous hydrochloric acid solution (2.times.25 mL).
[2663] The combined aqueous layers were basified with 2.0 M aqueous sodium hydroxide solution to pH=9, and extracted with diethyl ether (2.times.100 mL), the combined organic layers were washed with brine (2.times.50 mL), dried over anhydrous sodium sulfate; filtered and concentrated in vacuo to provide the title compound as an oil (6.28 g, 86%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.36-7.23 (m, 5H), 5.13 (d, J=12.7 Hz, 1H), 5.08 (d, J=12.7 Hz, 1H), 4.27-4.16 (m, 1H), 3.88-3.79 (m, 1H), 3.02 (td, J=12.7, 3.3 Hz, 1H), 2.95-2.83 (m, 2H), 2.76-2.70 (m, 1H), 2.64 (td, J=12.7, 3.4 Hz, 1H), 1.21 (d, J=7.0 Hz, 3H); MS(ES+) m/z 235.1 (M+1).
Step 3. Preparation of benzyl (S)-4-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2-methylpipe- razine-1-carboxylate
##STR01522##
[2665] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with benzyl (S)-2-methylpiperazine-1-carboxylate, the title compound was obtained as a colorless solid (6.58 g, 84%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.3 Hz, 1H), 7.38-7.25 ) (m, 5H), 7.14 (d, J=11.7 Hz, 1H), 5.17-5.07 (m, 2H), 4.34-4.23 (m, 1H), 3.95-3.85 (m, 1H), 3.58 (s, 2H), 3.17 (td, J=12.4-3.0 Hz, 1H), 2.74 (d, J=11.1 Hz, 1H), 2.59 (d, J=11.1 Hz, 1H), 2.22 (dd, J=11.1, 3.8 Hz, 1H), 2.08 (td, J=11.8, 3.3 Hz, 1H), 2.01-1.90 (m, 1H), 1.56 (s, 9H), 1.26 (d, J=6.7 Hz, 3H), 0.94-0.86 (m, 2H), 0.64-0.57 (m, 2H); MS(ES+) m/z 483.1 (M+1).
Step 4. Preparation of tert-butyl (S)-5-cyclopropyl-2-fluoro-4-((3-methylpiperazin-1-yl)methyl)benzoate
##STR01523##
[2667] To a degassed mixture of 10% palladium on carbon (50% wetted powder, 2.0 g) in methanol (50 mL) was added a solution of benzyl (S)-4-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2-methylpipe- razine-1-carboxylate (5.51 g, 11.42 mmol) in methanol (50 mL). The resulting mixture was bubbled with hydrogen gas for 2 minutes and then held under 1 atmosphere of hydrogen for 2 hours. The reaction mixture was bubbled with nitrogen, filtered through diatomaceous earth, and concentrated in vacuo to provide the title compound as a colorless oil (3.98 g, quant. yield): MS(ES+) m/z: 349.3 (M+1).
Step 5. Preparation of tert-butyl 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoate and tert-butyl 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate
##STR01524##
[2669] Following the procedure as described in Example 563 step 1, and making variation as required to replace methyl 4-(((1S,4S)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoate dihydrochloride with tert-butyl (S)-5-cyclopropyl-2-fluoro-4-((3-methylpiperazin-1-yl) methyl)benzoate and purification by flash chromatography (0-15% ethyl acetate (+10% isopropanol, 10% triethylamine) in hexanes. The first eluting fraction was arbitrarily assigned as tert-butyl 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoate (1.44 g, 29%): MS(ES+) m/z: 521.3, 523.2 (M+1). The second eluting fraction was arbitrarily assigned as tert-butyl 5-cycloproppyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate (3.27 g, 66%) MS(ES+) m/z: 521.2, 523.2 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride
##STR01525##
[2671] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.49 g, quant. yield): MS(ES+) m/z 465.0, 467.0 (M+1).
Step 7. Preparation of 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01526##
[2673] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.07 g, 9%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.69 (d, J=7.7 Hz, 1H), 7.32-7.25 (m, 3H), 7.20-7.18 (m, 1H), 3.98-3.85 (m, 1H), 3.62 (s, 2H), 3.39 (s, 3H), 3.04-2.92 (m, 1H), 2.65-2.57 (m, 1H), 2.50-2.41 (m, 1H), 2.36-2.19 (m, 4H), 1.98-1.87 (m, 1H), 1.24 (d, J=6.7 Hz, 3H), 1.10 (d, J=6.3 Hz, 3H), 0.98-0.91 (m, 2H), 0.66-0.59 (m, 2H); MS(ES+) m/z 542.1, 544.1 (M+1).
Example 566
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((S)-4-((R)-1-(3,5-dichloropheny- l)ethyl)-3-methylpiperazin-1-yl) methyl)-2-fluorobenzamide
##STR01527##
[2675] Following the ptocedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((S)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.11 g, 14%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.74 (br s, 1H), 7.70 (d, J=7.8 Hz, 1H), 7.34-7.25 (m, 3H), 7.21-7.17 (m, 1H), 3.97-3.84 (m, 1H), 3.61 (m, 2H), 3.13-3.03 (m, 1H), 3.02-2.91 (m, 1H), 2.65-2.57 (m, 1H), 2.50-2.40 (m, 1H), 2.35-2.17 (m, 4H), 1.98-1.87 (m, 1H), 1.48-1.40 (m, 2H), 1.24 (d, J=6.5 Hz, 3H), 1.17-1.06 (m, 5H), 0.98-0.90 (m, 2H), 0.66-0.59 (m, 2H); MS(ES+) m/z 568.2, 570.2 (M+1).
Example 567
Synthesis of 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluoro-N-(methytsulfonyl)benzamide
##STR01528##
[2676] Step 1. Preparation of 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid dihydrochloride
##STR01529##
[2678] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-(((S)-4-((S)-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (3.38 g, quant.): MS(ES+) m/z 465.0, 467.0 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-yl) methyl)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01530##
[2680] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzeic acid with 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.003 g, 0.2%): MS(ES+) m/z 542.2, 544.2 (M+1).
Example 568
Synthesis of 5-cyclopropyl-N-cyclopropylsulfonyl)-4-(((S)-4-((S)-1-(3,5-dichlorophenyl- )ethyl)-3-)methylpiperazin-1-yl) methyl)-2-fluorobenzamide
##STR01531##
[2682] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((S)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.004 g, 0.2%): MS(ES+) m/z 568.2, 570.2 (M+1).
Example 569
Synthesis of 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl)ethyl-3-methylpiperazi- n-1-yl) methyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01532##
[2683] Step 1. Preparation of 1-benzyl 4-(tert-butyl) (R)-2-methylpiperazine-1,4-dicarboxylate
##STR01533##
[2685] Following the procedure as described in Example 565 step 1, and making variation as required to replace tert-butyl (S)-3-methylpiperazine-1-carboxylate with tert-butyl (R)-3-methylpiperazine-1-carboxylate, the title compound was obtained as a colorless oil (10.60 g, quant. yield): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.39-7.26 (m, 5H) 5.16-5.06 (m, 2H), 4.37-4.23 (m, 1), 4.12-3.70 (m, 3H), 3.16-2.91 (m, 2H), 2.88-2.68 (m, 1H), 1.43 (s, 9H), 1.14 (d, J=6.7 Hz, 3H); MS(ES+) m/z 235.2 (M-Boc+H).
Step 2. Preparation of benzyl (R)-2-methylpiperazine-1-carboxylate
##STR01534##
[2687] Following the procedure as described in Example 565 step 2, and making variation as required to replace 1-benzyl 4-(tert-butyl) (S)-2-methylpiperazine-1,4-dicarboxylate with 1-benzyl 4-(tert-butyl) (R)-2-methylpiperazine-1,4-dicarboxylate, the title compound was obtained as a colorless oil (6.80 g, 82%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.36-7.24 (m, 5H), 5.16-5.05 (m, 2H), 4.27-4.17 (m, 1H), 3.88-3.79 (m, 1H), 3.02. (td, J=12.5, 3.3 Hz, 1H), 2.95-2.82 (m, 2H), 2.77-2.70 (m, 1H), 2.64 (td, J=12.3, 3.5 Hz, 1H), 1.49 (br s, 1H), 1.21 (d, J=6.5 Hz, 3H); MS(ES+) m/z: 235.1 (M+1).
Step 3. Preparation of benzyl (R)-4-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2-methylpipe- razine-1-carboxylate
##STR01535##
[2689] Following the procedure as described in Example 53 step 3, and making variation as required to replace (S)-3-(3,5-dichlorophenoxy)piperidine, trifluoroacetic acid salt with benzyl (R)-2-methylpiperazine-1-carboxylate, the title compound was obtained as a colorless solid (7.04 g, 90%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.47 (d, J=7.4 Hz, 1H), 7.36-7.25 (m, 5H), 7.14 (d, J=11.8 Hz, 1H), 5.17-5.07 (m, 2H), 4.34-4.23 (m, 1H), 3.95-3.86 (m, 1H), 3.58 (s, 2H), 3.17 (td, J=12.7, 3.2 Hz, 1H), 2.74 (d, J=11.0 Hz, 1H), 2.59 (d, J=11.1 Hz, 1H), 2.22 (dd, J=11.0, 3.5 Hz, 1H), 2.08 (td, J=11.6, 3.3 Hz, 1H), 2.01-1.90 (m, 1H), 1.56 (s, 9H), 1.26 (d, J=6.8 Hz, 3H), 0.94-0.86 (m, 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 483.1 (M+1).
Step 4. Preparation of tert-butyl (R)-5-cyclopropyl-2-fluoro-4-((3-methylpiperazin-1-yl)methyl)benzoate
##STR01536##
[2691] Following the procedure as described in Example 565 step 4, and making variation as required to replace benzyl (S)-4-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2-methylpipe- razine-1-carboxylate with benzyl (R)-4-(4-(tert-butoxycarbonyl)-2-cyclopropyl-5-fluorobenzyl)-2-methylpipe- razine-1-carboxylate, the title compound was obtained as a colorless oil (4.19 g, 98%): MS(ES+) m/z 349.1 (M+1).
Step 5. Preparation of tert-butyl 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate and tert-butyl 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl-2-fluorobenzoate
##STR01537##
[2693] Following the procedure as described in Example 563 step 1, and making variation as required to replace methyl 4-(((1S,4S)-2,5-diazabicyclo[2.2.1]heptan-2-yl)methyl)-5-cyclopropyl-2-fl- uorobenzoate dihydrochloride with tert-butyl (R)-5-cyclopropyl-2-fluoro-4-((3-methylpiperazin-1-yl) methyl)benzoate and purification by flash chromatography (0-15% ethyl acetate (+10% isopropanol, +10% triethylamine) in hexanes. The first eluting fraction was arbitrarily assigned as tert-butyl 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-piperazin-1-y- l) methyl)-2-fluorobenzoate (1.12 g, 24%)): MS(ES+) m/z: 521.2, 523.2 (M+1). The second eluting fraction was arbitrarily assigned as tert-butyl 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorephenyl) ethyl)-3-methylpiperazin-1-yl)methyl-2-fluorobenzoate (2.07 g, 44%): MS(ES+) m/z 521.2, 523.2 (M+1).
Step 6. Preparation of 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoic acid dihydrochloride
##STR01538##
[2695] Following the procedure as described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (1.16 g, quant. yield): MS(ES+) m/z 465.1, 467.1 (M+1).
Step 7. Preparation of 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluoro-N-(methylsulfonyl)benzamid- e
##STR01539##
[2697] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S )-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-fluor- obenzoic acid with 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl)methyl)-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.13 g, 22%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.44 (d, J=7.7 Hz, 1H), 7.27 (d, J=1.6 Hz, 2H), 7.17 (dd, J=1.7 Hz, 1H), 7.02 (d, J=12.2 Hz, 1H), 6.78 (br s, 1H), 3.93-3.81 (m, 1H), 3.52 (s, 2H), 3.02 (s, 3H), 2.96-2.84 (m, 1H), 2.61-2.51 (m, 1H), 2.45-2.35 (m, 1H), 2.31-2.11 (m, 3H), 1.99-1.96 (m, 1H), 1.93-1.84 (m, 1H), 1.20 (d, J=1.05, 3H), (d, J=6.2Hz, 3H), 0.87-0.80 (m, 2H), 0.60-0.52 (m, 2H); MS(ES+) m/z 542.1, 544.1 (M+1).
Example 570
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((R)-4-((R)-1-(3,5-dichloropheny- l) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenamide
##STR01540##
[2699] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl-2-fluo- robenzoic acid with 5-cyclopropyl-4-(((R)-4-((R)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiparaz- in-1-yl) methyl)-2-fluorobenzoic acid hydrochloride, and purificafion by preparative HPLC, the title compound was obtained as a colorless solid (0.18 g, 30%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.69 (d, J=7.8 Hz, 1H), 7.32-7.23 (m, 3H), 7.21-7.17 (m, 1H), 3.98-3.83 (m, 1H), 3.61 (s, 2H), 3.12-3.02 (m, 1H), 3.02-2.92 (m, 1H), 2.65-2.57 (m, 1H), 2.51-2.40 (m, 1H), 2.35-2.19 (m, 4H), 2.00-1.87 (m, 1H), 1.47-1.39 (m, 2H), 1.24 (d, J=6.5 Hz, 3H), 1.17-1.06 (m, 5H), 0.98-0.90 (m, 2H) 0.66-0.59 (m, 2H); MS(ES+) m/z 568.2, 570.2 (M+1).
Example 571
Synthesis of 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluoro-N-(methylsulfonyl)benzamid- e
##STR01541##
[2700] Step 1. Preparation of 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoic acid hydrochloride
##STR01542##
[2702] Following the procedure described in Example 517 step 2, and making variation as required to replace tert-butyl 4-(3,5-dichlorobenzyl)piperazine-1-carboxylate with tert-butyl 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzoate, the title compound was obtained as a colorless solid (2.14 g, quant.): MS(ES+) m/z 465.1, 467.1 (M+1).
Step 2. Preparation of 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluoro-N-(methylsulfonyl)benzamid- e
##STR01543##
[2704] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl)ethyl)-3-methylpiperaz- in-1-yl)methyl)-2-fluorobenzoic acid hydrochloride, and to replace cyclopropanesulfonamide with methylsulfonamide, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.005 g, 0.5%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 7.46 (d, J=7.5 Hz, 1H), 7.22-7.18 (m, 1H), 7.17-7.13 (m, 2H), 6.99 (d, J=12.2 Hz, 1H), 3.91-3.78 (m, 1H), 3.51 (s, 2H), 3.02 (s, 3H), 2.79-2.67 (m, 1H), 2.54-2.35 (m, 3H), 2.21-2.11 (m, 2H), 1.94-1.82 (m, 1H), 1.31 (d, J=6.8 Hz, 3H), 1.25-1.20 (m, 2H), 1.00 (d, J =6.1 Hz, 3H), 0.88-0.78 (m, 2H), 0.59-0.52 (m, 2H); MS(ES-) m/z 540.2, 542.2 (M-1).
Example 572
Synthesis of 5-cyclopropyl-N-(cyclopropylsulfonyl)-4-(((R)-4-((S)-1-(3,5-dichloropheny- l) ethyl)-3-methylpiperazin-1-yl)methyl)-2-fluorobenzamide
##STR01544##
[2706] Following the procedure as described in Example 53 step 5, and making variation as required to replace (S)-5-cyclopropyl-4-((3-(3,5-dichlorophenoxy)piperidin-1-yl)methyl)-2-flu- orobenzoic acid with 5-cyclopropyl-4-(((R)-4-((S)-1-(3,5-dichlorophenyl)ethyl-3-methylpiperazi- n-1-yl)methyl)-2-fluorobenzoic acid hydrochloride, and purification by preparative HPLC, the title compound was obtained as a colorless solid (0.01 g, 1%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta. 8.74 (br s, 1H), 7.69 (d, J=7.8 Hz, 1H), 7.28-7.20 (m, 2H), 7.19-7.13 (m, 2H), 3.94-3.81 (m, 1H), 3.59 (s, 2H), 3.12-3.02 (m, 1H), 2.93-2.72 (m, 1H), 2.58-2.42 (m, 3H), 2.25-2.16 (m, 1H), 1.97-1.86 (m, 1H), 1.66-1.49 (m, 2H), 1.47-1.40 (m, 2H), 1.33 (d, J=6.5 Hz, 3H), 1.17-1.08 (m, 2H), 1.05 (d, J=6.3 Hz, 3H), 0.97-0.89 2H), 0.65-0.57 (m, 2H); MS(ES+) m/z 568.2, 570.2 (M+1).
Example 573
Synthesis of (S)-4-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl) oxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)
##STR01545##
[2708] Following the procedure as described in Example 3 step 5, and making variation as required to replace (R)-5-cyclopropyl-4-((1-(3,5-dichlorobenzyl)piperidin-3-yl)-2-fluorobenzo- ic acid with (S)-1-((1-(2-chloro-4-fluorobenzyl)piperidin-3-yl)oxy-5-cyclopropyl-2-flu- orobenzoic acid, and to replace cyclopropylsulfonamide with methanesulfonamide, the title compound was obtained as a colorless solid (0.09 g, 55%): .sup.1H NMR (300 MHz, CDCl.sub.3) .delta.7.55 (d, J=9.2 Hz, 1H), 7.46-7.41 (m, 1H), 7.11-7.08 (m, 1H), 6.96-6.89 (m, 1H), 6.58 (d, J=14.6 Hz, 1H), 4.46-4.41 (m, 1H), 3.63 (s, 2H), 3.41 (s, 3H), 2.99-2.96 (m, 1H), 2.73-2.70 (m, 1H), 2.48-2.42 (m, 1H), 2.36-2.29 (m, 1H), 2.10-2.02 (m, 2H), 1.92-1.86 (m, 1H), 1.72-1.56 (m, 2H), 0.96-0.90 (m, 2H), 0.69-0.64 (m, 2H); MS(ES+) m/z 498.9, 500.9 (M+1); MS(ES-) m/z 497.2, 499.2 (M-1).
Example 574
Synthesis of 4-((1-benzhydryl-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01546##
[2709] Steps 1-2: Preparation of tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)-phenoxy)methyl)-4-methylpiperidine-1-carboxylate
[2710] The compound was prepared in a similar manner to Example 598 starting from 5-chloro-2,4-difluoro-N-methylsulfonyl-benzamide and tert-butyl 3-(hydroxymethyl)-3-methylazetidine-1-carboxylate.
Step 3: Preparation of 4-((1-benzhydryl-4-methylpiperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[2711] A mixture of 4-((1-benzhydryl-4-methylpiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluoro-N- -(methylsulfonyl)benzamide (55 mg), trifluoroacetic acid (0.23 mL) in dichloromethane (0.68 mL) was stirred at 0.degree. C. for 10 min then at rt for 1 h. The contents were concentrated under vacuum. To the residue were added acetonitrile (2.3 mL), benzhydryl bromide (29 mg), and cesium carbonate (266 mg). The mixture was heated at 50.degree. C. for 4 hr. Extra benzhydryl bromide (6 mg) was added. The mixture was heated at 50.degree. C. for 16 hr. Acidified with 0.5M NaH.sub.2PO.sub.4, the contents were extracted with DCM (2.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with HPLC (19.8 mg). LCMS (Method F): RT=4.71 min, m/z: 551.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.84 (s, 1H), 7.49-7.36 (m, 4H), 7.36-7.24 (m, 4H), 7.24-7.11 (m, 3H), 6.93 (d, J=12.9 Hz, 1H), 4.35 (s, 1H), 3.84 (s, 2H), 2.31-2.15 (m, 2H), 2.04-1.94 (m, 1H), 1.76-1.60 (m, 2H), 1.55-1.35 (m, 1H), 1.06 (s, 3H), 0.92-0.80 (m, 2H), 0.68-0.58 (m, 2H).
Example 575
Synthesis of 5-cyclopropyl-4-((1-(3,4-dichlorobenzyl)-4-methylpiperidin-4-yl)methoxy)-- 2-fluoro-N-(methylsulfonyl)benzamide
##STR01547##
[2713] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluporo-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-- 4-methylpiperidin-1-carboxylate (Example 73 step 1-2) and 3,4-dichlorobenzaldehyde. LCMS (Method F): RT=4.73 min, m/z: 543.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.67-7.58 (m, 2H), 7.35 (dd, J=8.3, 2.0 Hz, 1H), 7.20 (d, J=8.4 Hz, 1H), 6.87 (d, J=12.8 Hz, 1H), 3.83 (s, 2H), 3.72 (s, 2H), 3.15 (s, 3H), 2.78-2.62 (m, 2H), 2.07-1.95 (m, 1H), 1.76-1.64 (m, 2H), 1.57-1.43 (m, 2H), 1.09 (s, 3H), 0.94-0.83 (m, 2H), 0.68-0.55 (m, 2H).
Example 576
Synthesis of 5-cyclopropyl-2-fluoro-4-((1-((6-methoxypyridin-2-yl) methyl)-4-methylpiperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR01548##
[2715] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl)carbamoyl)phenoxy)methyl)-4- -methylpiperidine-1-carboxylate (Example 73 step 1-2) and 6-methoxypicolinaldehyde.
[2716] LCMS (Method F): RT=4.25 min, m/z: 506.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.77-7.69 (m, 1H), 7.22 (d, J=8.5 Hz, 1H), 7.06 (d, J=7.3 Hz, 1H), 6.80 (dd, J=26.4, 10.5 Hz, 2H), 4.02-3.89 (m, 2H), 3.86 (s, 3H), 3.85 (d, J=1.2 Hz, 2H), 3.05 (s, 3H), 3.01-2.75 (m, 4H), 2.06-1.97 (m, 1H), 1.87-1.74 (m, 2H), 1.58 (s, 2H), 1.12 (s, 3H), 0.91-0.83 (m, 2H), 0.63-0.56 (m, 2H).
Example 577
Synthesis of 5-cyclopropyl-4-((1-(4,5-dichloro-2-fluorobenzoyl)-4-methylpiperidin-4-yl- ) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01549##
[2718] To a solution of tert-butyl 4-((2-cyclopropyl-5-fluoro-4-(methylsulfonyl)carbamoyl)-phenoxy) methyl)-4-methylpiperidine-1-carboxylate (Example 73 steps 1-2, 56 mg) in dichloromethane (0.7 mL) at 0.degree. C. was added trifluoroacetic acid (0.17 mL). The mixture was stirred at 0.degree. C. for 10 min then at rt for 1 h. The contents were concentrated under vacuum. To the residue was added dichloromethane (2.3 mL), 4,5-dichloro-2-fluorobenzoic acid (28 mg), DIPEA (0.09 mL, cooled with ice-bath), and HBTU (30 mg). The mixture was stirred at rt for 1 hr. Acidified with 1:4 mixture of 0.5 M HCl and 0.5 M NaH.sub.2PO.sub.4, the contents were extracted with DCM (2.times.). The combined DCM solutions were dried (Na.sub.2SO.sub.4). After filtration and concentration. The crude was purified with HPLC (41 mg). LCMS (Method. F): RT=6.50 min, m/z: 575.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.85 (s, 1H), 7.83 (d, J=8.9 Hz, 1H), 7.73 (d, J=6.4 Hz, 1H), 7.18 (d, J=8.3 Hz, 1H), 7.00-6.91 (m, 1H), 4.01-3.85 (m, 3H), 3.49-3.38 (m, 1H), 2.05-1.95 (m, 1H), 1.74-1.55 (m, 2H), 1.56-1.45 (m, 1H), 1.45-1.35 (m, 1H), 1.15 (s, 3H), 0.93-0.84 (m, 2H), 0.70-0.61 (m, 2H).
Example 578
Synthesis of 5-cyclopropyl-4-((1-(2,5-dichlorobenzoyl)-4-methylpiperidin-4-yl) methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01550##
[2720] The compound was prepared in a similar manner to Example 589 from tert-butyl 4-((2-cyclopropyl-5-fluoro-4-((methylsulfonyl) carbamoyl)phenoxy)methyl)-4-methylpiperidine-1-carboxylate and 2,5-dichlorobenzoic acid. LCMS (Method F): RT=6.256 min, m/z: 557.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.85 (s, 1H), 7.62-7.43 (m, 3H), 7.19 (t, J=8.0 Hz, 1H), 6.93 (dd, J=12.9, 6.2 Hz, 1H), 3.99 (s, 1H), 3.89 (d, J=4.9 Hz, 1H), 3.41 (d, J=13.9 Hz, 1H), 3.26-3.17 (m, 4H), 2.01 (d, J=4.9 Hz, 1H), 1.78-1.46 (m, 4H), 1.40 (d, J=13.6 Hz, 1H), 1.14 (d, J=9.5 Hz, 3H), 0.93-0.82 (m, 2H), 0.70-0.58 (m, 2H).
Example 579
Synthesis of 4-((1-(4-chloro-2-(trifluoromethyl)piperidin)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01551##
[2722] To a solution of 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy)benzamide hydrochloride (60 mg) in 1,2-dichloroethane (2.2 mL) at 0.degree. C. was added DIPEA (0.075 mL), followed by 4-fluoro-2-(trifluoromethyl)benzaldehyde (39 mg) and sodium triacetoxyborohydride (91 mg). The mixture was stirred at rt for 20 hr. Diluted with 0.5 M NaH.sub.2PO.sub.4, the contents were extracted with DCM (3.times.). The combined org solutions were dried (Na.sub.2SO.sub.4). After filtration and concentration, the residue was purified with HPLC (55 mg). LCMS (Method G): RT=4.36 min, m/x: 563.14 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.74 (s, 1H), 7.81 (d, J=8.5 Hz, 1H), 7.78-7.72 (m, 2H), 7.15 (d, J =8.4 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 3.95 (d, J=6.0 Hz, 2H), 3.62 (s, 2H), 3.27 (s, 3H), 2.83 (d, J =11.1 Hz, 2H), 2.16-1.97 (m, 3H), 1.90-1.74 (m, 3H), 1.46-1.31 (m, 2H), 0.92-0.85 (m, 2H), 0.69-0.62 (m, 2H).
Example 580
Synthesis of 4-((1-(2-chloro-5-(trifluoromethyl)piperidin)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01552##
[2724] The compound was prepared in a similar manner to Example 591 from 5-cyclopropyl-2-fluoro-N-methylsuifonyl-4-(4-piperidylmethoxy)benzamide hydrochloride and 2-chloro-5-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=4.25 min, m/z: 563.14 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.61 (s, 1H), 7.86 (d, J=2.0 Hz, 1H), 7.73-7.64 (m, 2H), 7.16 (d, J=8.4 Hz, 1H), 6.91 (d, J=12.9 Hz, 1H), 3.97 (d, J=6.0 Hz, 2H), 3.72 (s, 2H), 3.26 (s, 3H), 2.92 (d, J=11.2 Hz, 2H), 2.24 (t, J=11.6 Hz, 2H), 2.08-1.96 (m, 1H), 1.92-1.75 (m, 3H), 1.51-1.35 (m, 2H), 0.92-0.83 (2H), 0.69-0.61 (m, 2H).
Example 581
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01553##
[2725] Step 1: Preparation of methyl 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoate
[2726] To a solution of methyl 5-cyclopropyl-2-fluoro-4-(4-piperidylmethoxy)benzoate hydrochloride (A, 0.362 g, 1.00 mmol, .about.95% pure) in 1,2-dichloroethane (6.0 mL) at 0.degree. C. was added DIPEA (2.0 equiv., 2.00 mmol, 100 mass %), followed by 3-chloro-2-fluoro-5-(trifluoromethyl)benzaldehyde (350 mg) and sodium triacetoxyborohydride (636 mg). The mixture was stirred at rt for 20 hr. Diluted with aqueous sodium bicarbonate solution, the contents were extracted with DCM (3.times.). The combined org solutions were dried (Na.sub.2SO.sub.4). After filtration and concentration, the crude was purified with flash chromatography (0-40% EtOAc/heptane) to afford the product (498 mg).
Step 2: Preparation of 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoic acid
[2727] To a mixture of methyl 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoate (A, 0.480 g, 0.927 mmol) and KOH (57 mg) in methanol (4.6 mL) was slowly added water (0.46 mL). The resulting mixture was stirred at 40.degree. C. for 16 hr. Extra 0.1 eq of KOH was added. The mixture was heated at 60.degree. C. for 20 hr. LCMS showed completion. The contents were concentrated under vacuum. Used as-is.
Step 3: Preparation of 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-N-methylsulfon- yl-benzamide
[2728] A mixture of crude 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoic acid potassium salt (60.0 mg) from the previous step, methanesulfonamide (41 mg), HBTU (62 mg) and DIPEA (0.037 mL) in 1,2-dichloroethane (1.6 mL) was stirred at 4.degree. C. for 16 hr. LCMS showed completion. Acidified with 0.5M NaH.sub.2PO.sub.4, the contents were extracted with DCM (3.times.). The combined extracts were dried (Na.sub.2SO.sub.4). The crude was purified with HPLC (16.2 mg). LCMS (Method G): RT=5.71 min, m/z: 581.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.59 (s, 1H), 8.05-7.98 (m, 1H), 7.83-7.75 (m, 1H), 7.15 (d, J=8.3 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 3.94 (d, J=5.9 Hz, 2H), 3.73 (s, 2H), 3.24 (s, 3H), 2.91 (d, J=11.2 Hz, 2H), 2.25-2.10 (m, 2H), 2.05-1.96 (m, 1H), 1.80 (d, J=11.9 Hz, 3H), 1.39 (d, J=11.9 Hz, 2H), 0.93-0.83 (m, 2H), 0.69-0.61 (m, 2H).
Example 582
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)piperidin-4-yl) methoxy-5-cyclopropyl-N-(cyclopropylsulfonyl)-2-fluorobenzamide
##STR01554##
[2730] The compound was prepared in a similar manner to Example 587 from 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoic acid and cyclopropanesulfonamide. LCMS (Method F): RT=4.98 min, m/z: 607.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.69 (s, 1H), 8.03-7.97 (m, 1H), 7.80-7.74 (m, 1H), 7.16 (d, J=8.5 Hz, 1H), 6.83 (d, J=12.9 Hz, 1H), 3.92 (d, J=5.8 Hz, 2H), 3.70-3.63 (m, 2H), 3.04-2.93 (m, 1H), 2.86 (d, J=11.1 Hz, 2H), 2.14-2.05 (m, 3H), 2.04-1.96 (m, 1H), 1.78 (d, J=10.9 Hz, 3H), 1.44-1.31 (m, 2H), 1.04-0.91 (m, 3H), 0.90-0.83 (m, 2H), 0.65-0.58 (m, 2H).
Example 583
Synthesis of N-(azetidin-1-ylsulfonyl)-4-((1-(3-chloro-2-fluoro)-5-(trifluoromethyl)pi- peridin)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluorobenzamide
##STR01555##
[2732] The compound was prepared in a similar manner to Example 587 from 4-[[1-[[3-chloro-2-fluoro-5-(trifluoromethyl) phenyl]methyl]-4-piperidyl]methoxy]-5-cyclopropyl-2-fluoro-benzoic acid and azetidine-1-sulfonamide. LCMS (Method F): RT=5.04 min, m/z: 622.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSP-d6) .delta. 11.41 (s, 1H), 8.03-7.97 (m, 1H), 7.81-7.74 (m, 1H), 7.15 (d, J=8.3 Hz, 1H), 6.90 (d, J=12.8 Hz, 1H), 4.06-3.90 (m, 6H), 3.68 (s, 2H), 2.88 (d, J=11.1 Hz, 2H), 2.19-2.06 (m, 4H), 2.06-1.97 (m, 1H), 1.79 (d, J=11.4 Hz, 3H), 1.46-1.31 (m, 2H), 0.93-0.84 (m, 2H), 0.69-0.61 (m, 2H).
Example 584
Synthesis of 4-((1-(4-chloro-2-(trifluoromethyl)piperidin)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01556##
[2734] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy)benzamide hydrochloride and 4-chloro-2-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=4.36 min, m/z: 563.14 [M+H].sup.+.
[2735] .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.74 (s, 1H), 7.81 (d, J=8.5 Hz, 1H), 7.78-7.72 (m, 2H), 7.15 (d, J=8.4 Hz, 1H), 6.92 (d, J=12.9 Hz, 1H), 3.95 (d, J=6.0 Hz, 2H), 3.62 (s, 2H), 3.27 (s, 3H), 2.83 (d, J=11.1 Hz, 2H), 2.16-1.97 (m, 3H), 1.90-1.74 (m, 3H), 1.46-1.31 (m, 2H), 0.92-0.85 (m, 2H), 0.69-0.62 (m, 2).
Example 585
Synthesis of 4-((1-(4-bromo-2-(trifluoromethyl)piperidin)piperadin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01557##
[2737] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluror-N-methylsulfonyl-4-(4-piperidylmethoxy)benzamide hydrochloride and 4-bromo-2-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=4.44 min, m/z: 609.09 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.74 (s, 1H), 7.93-7.82 (m, 2H), 7.74 (d, J=8.4 Hz, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.92 (d, J=13.0 Hz, 1H), 3.96 (d, J=6.1 Hz, 2H), 3.61 (s, 2H), 3.28 (s, 3H), 2.89-2.77 (m, 2H), 2.17-1.96 (m, 3H), 1.90-1.72 (m, 3H), 1.46-1.31 (m, 2H), 0.94-0.82 (m, 2H), 0.71-0.61 (m, 2H).
Example 586
Synthesis of 5-cyclopropyl-2-fluoro-4-((1-(4-fluoro-2-(trifluoromethyl) benzyl)piperidin-4-yl)methoxy)-N-(methylsulfonyl)benzamide
##STR01558##
[2739] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethyoxy)benzamide hydrochloride and 4-fluoro-2-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=4.18 min, m/z: 547.17 [M+H].sup.+.
[2740] .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.71 (s, 1H), 7.82 (dd, J=8.6, 5.8 Hz, 1H), 7.61-7.49 (m, 2H), 7.15 (d, J=8.3 Hz, 1H), 6.92 (d, J=13.0 Hz, 1H), 3.95 (d, J=6.2 Hz, 2H), 3.62 (s, 2H), 2.84 (d, J=11.1 Hz, 2H), 2.17-1.96 (m, 3H), 1.90-1.73 (m, 3H), 1.46-1.31 (m, 2H), 0.93-0.84 (m, 2H), 0.69-0.62 (m, 2H).
Example 587
Synthesis of 4-((1-(4-bromo-3-chlorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01559##
[2742] The compound was prepared in a similar manner of Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy) benzamide hydrochloride and 4-bromo-2-chlorobenzaldehyde. LCMS (Method G): RT=4.13 min, m/z: 575.06 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.76 (d, J =8.2 Hz, 1H), 7.61 (d, J=2.0 Hz, 1H), 7.27 (dd, J=8.2, 2.0 Hz, 1H), 7.17 (d, J=8.4 Hz, 1H), 6.86 (d, J=12.9 Hz, 1H), 3.94 (d, J=5.9 Hz, 2H), 3.67 (s, 2H), 3.16 (s, 3H), 2.97 (d, J=11.2 Hz, 2H), 2.25 (s, 2H), 2.06-1.96 (m, 1H), 1.83 (d, J=12.8 Hz, 3H), 1.42 (d, J=12.2 Hz, 2H), 0.92-0.83 (m, 2H), 0.67-0.58 (m, 2H).
Example 588
Synthesis of 4-((1-(4-bromo-2,5-difluorobenzyl)piperidin-4-yl)methoxy)-5-cyclopropyl-2- -fluoro-N-(methylsulfonyl)benzamide
##STR01560##
[2744] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy)benzamide hydrochloride and 4-bromo2,5-difluorobenzaldehyde. LCMS (Method G): RT=4.19 min, m/z: 577.08 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.46 (s, 1H), 7.71 (dd, J=8.9, 5.7 Hz, 1H), 7.44 (dd, J=9.1, 6.2 Hz, 1H), 7.15 (d, J=8.3 Hz, 1H), 6.89 (d, J=12.9 Hz, 1H), 3.94 (d, J=5.9 Hz, 2H), 3.62 (s, 2H), 3.23 (s, 3H), 2.93 (d, J=11.2 Hz, 2H), 2.19 (t, J=11.5 Hz, 2H), 2.05-1.96 (m, 1H), 1.80 (d, J=12.4 Hz, 3H), 1.48-1.32 (m, 2H), 0.92-0.83 (m, 2H), 0.68-0.60 (m, 2H).
Example 589
Synthesis of 4-((1-(4-bromo-2-fluorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benazamide
##STR01561##
[2746] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy)benzamide hydrochloride and 4-bromo-2,5-difluorobenzaldehyde. LCMS (Method G): RT=4.18 min, m/z: 559.09 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.54 (dd, J=9.7, 1.8 Hz, 1H), 7.47-7.37 (m, 2H), 7.16 (d, J=8.5 Hz, 1H), 6.87 (d, J=12.9 Hz, 1H), 3.93 (d, J=5.97 Hz, 2H), 3.67 (s, 2H), 3.19 (s, 3H), 2.96 (d, J=11.2 Hz, 2H), 2.24 (s, 2H), 2.05-1.96 (m, 1H), 1.81 (d, J=11.8 Hz, 3H), 1.47-1.32 (m, 2H), 0.92-0.83 (m, 2H), 0.67-0.59 (m, 2H).
Example 590
Synthesis of 4-((1-(3-chloro-5-cyanobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01562##
[2748] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy) benzamide hydrochloride and 3-chloro-5-formylbenzonitrile. LCMS (Method G): RT=3.96 min, m/z: 520.15 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.42 (s, 1H), 7.96 (t, J=1.8 Hz, 1H), 7.81-7.74 (m, 2H), 7.16 (d, J=8.4 Hz, 1H), 6.89 (d, J=11.6 Hz, 2H), 3.95 (d, J=5.9 Hz, 2H), 3.68 (s, 2H), 3.22 (s, 3H), 2.92 (d, J=11.2 Hz, 2H), 2.19 (t, J=11.6 Hz, 2H), 2.06-1.95 (m, 1H), 1.91-1.74 (m, 3H), 1.50-1.32 (m, 2H), 0.94-0.82 (m, 2H), 0.69-0.60 (m, 2H).
Example 591
Synthesis of 4-((1-(3-cyano-4-fluorobenzyl)piperidin-4-yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01563##
[2750] The compound was prepared in a similar manner to Example 585 from 5-cyclopropyl-2-fluoro-N-methylsulfonyl-4-(4-piperidylmethoxy) benzamide hydrochloride and 2-fluoro-5-formylbenzonitrile.
[2751] LCMS (Method G): RT=3.85 min, m/z: 504.17 [M+H].sup.+.
[2752] .sup.1H NMR (400 MHz, DMSO-d6) .delta. 7.87 (dd, J=6.3, 2.2 Hz, 1H), 7.80-7.72 (m, 1H), 7.57-7.48 (m, 1H), 7.16 (d, J=8.4 Hz, 1H), 6.88 (d, J=12.9 Hz, 1H), 3.94 (d, J=6.0 Hz, 2H), 3.70 (s, 2H), 3.19 (s, 3H), 2.96 (d, J=11.3 Hz, 2H), 2.24 (t, J=11.6 Hz, 2H), 2.05-1.96 (m, 1H), 1.92-1.76 (m, 3H), 1.49-1.33 (m, 2H), 0.92-0.83 (m, 2H), 0.68-0.60 (m, 2H).
Example 592
Synthesis of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(6-(trifluorormethyl) pyridin-2-yl)piperidin-4-yl)methoxy)benzamide
##STR01564##
[2753] Step 1: Preparation of 5-chloro-2-fluoro-N-methylsulfonyl-4-[[1-[6-(trifluoromethyl)-2-pyridyl]-- 4-piperidyl]methoxy]benzamide
[2754] To solution of (1-[6-(trifluoromethyl)pyridin-2-yl]piperidin-4-yl)methanol (169 mg) and 5-chloro-2,4-difluoro-N-methylsulfonyl-benzamide (175 mg) in DMSO (2.5 mL) at 14.degree. C. (bath) was added potassium tert-butoxide (175 mg). The mixture was stirred at rt for 1 hr. Diluted with EtOAc, the contents were washed with 1/4 mixture of 0.5M HCl and 0.5M NaH.sub.2PO.sub.4 (2.times.) and brine (1.times.), dried (Na.sub.2PO.sub.4). After filtration and concentration, the crude was purified with flash chromatography (0-2% MeOH/DCM with 0.5% HCO.sub.2H) to give the product (205 mg).
Step 2: Preparation of 5-cyclopropyl-2-fluoro-N-(methylsulfonyl)-4-((1-(6-(trifluoromethyl) pyridin-2-yl)piperidin-4-yl)methoxy)benzamide
[2755] A mixture of 5-chloro-2-fluoro-N-methylsulfonyl-4-[[1-[6-(trifluoromethyl)-2-pyridyl]-- 4-piperidly]methoxy]benzamide (205 mg), cyclopropylboronic acid (109 mg), and potassium phosphate (523 mg) in water (0.4 mL) and toluene (8.0 mL) was purged with nitrogen for 10 min. Tricyclohexylphosphoniumtetrafluoroborate (46 mg) and palladium acetate (14 mg) were added. The resulting mixture was stirred under nitrogen at 95.degree. C. for 40 hr. Acidified with 1/4 mixture of 0.5 M HCl and 0.5 M NaH.sub.2PO.sub.4, the contents were extracted with DCM (2.times.). The combined DCM solutions were dried (Na.sub.2SO.sub.4). Afier filtration and concentration, the crude was purified with HPLC (37.5 mg). LCMS (Method G): RT=7.99 min, m/z: 516.2 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.84 (s, 1H), 7.76-7.67 (m, 1H), 7.15 (dd, J=9.8, 8.6 Hz, 2H), 6.98 (d, J=7.2 Hz, 1H), 6.90 (d, J=12.9 Hz, 1H), 4.38 (d, J=13.2 Hz, 2H), 3.98 (d, J=6.2 Hz, 2H), 3.23 (s, 3H), 2.94 (td, J=12.7, 2.6 Hz, 2H), 2.12 (s, 1H), 2.04-1.94 (m, 1H), 1.92-1.81 (m, 2H), 1.43-1.28 (m, 2H), 0.88-0.80 (m, 2H), 0.68-0.59 (m, 2H).
Example 593
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-3-methylazetidin-3- -yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01565##
[2756] Steps 1-2: Preparation of tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]methyl]-3-m- ethyl-azetidine-1-carboxylate
[2757] The compound was prepared in a similar manner to Example 598 starting 5-chloro-2,4-difluoro-N-methylsulfonyl-benzamide and tert-butyl 3-(hydroxymethyl)-3-methylazetidine-1-carboxylate
Step 3: Preparation of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-3-methylazetidin-3- -yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
[2758] The compound was prepared in a similar manner to Example 73 starting from tert-butyl 3-[[2-cyclopropyl-5-fluoro-4-(methylsulfonylcarbamoyl)phenoxy]methyl]-3-m- ethyl-azetidine-1-carboxylate and 3-chloro-2-fluoro-5-(trifluoromethyl)benzaldehyde. LCMS (Method G): RT=5.66 min, m/z: 567.1 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 11.68 (s, 1H), 8.01 (dd, J=6.5, 2.2 Hz, 1H), 7.74 (dd, J=5.9, 2.3 Hz, 1H), 7.20 (d, J=8.5 Hz, 1H), 6.94 (d, J=12.8 Hz, 1H), 4.06 (s, 2H), 3.84 (s, 2H), 3.39 (d, J=7.3 Hz, 2H), 3.23 (s, 3H), 3.14 (d, J=7.3 Hz, 2H), 2.09-1.98 (m, 1H), 1.36 (s, 3H), 0.92-0.82 (m, 2H), 0.70-0.61 (m, 2H).
Example 594
Synthesis of 5-Cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl)piperidin-4-yl) methoxy)-2-fluoro-N-(mehylsulfonyl)benzamide
##STR01566##
[2759] Step 1: Preparation of 1-(bromomethyl)-3,5-dichloro-2-iodobenzene
##STR01567##
[2761] 2,2'-azobis(2-methylpropionitrile) (149 mg, 0.91 mmol) was added to a solution of 1,5-dichloro-2-iodo-3-methylbenzene (2.6 g, 9.1 mmol) and N-bromosuccinimide (1.8 g, 10.0 mmol) in acetonitrile (70 mL), the reaction mixture was stirred at 80.degree. C. for 16 h, diluted with water (50 mL) and extracted with ethyl acetate (100 mL.times.3), the combined organic layers were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (eluting with petroleum ether) to afford the target compound (1.26 g, yield: 38%) as a white solid. .sup.1H NMR (500 MHz, CDCl.sub.3): .delta. 7.44-7.43 (m, 1H), 7.41-7.40 (m, 1H), 4.61 (s, 2H).
Step 2: Preparation of methyl 5-cyclopropyl-4-((1-(3,5-dichloro-2-iodobenzyl) piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR01568##
[2763] A mixture of 1-(bromomethyl)-3,5-dichloro-2-iodobenzene (200 mg, 0.55 mmol), methyl 5-cyclopropyl-2-fluoro-4-(piperidin-4-ylmethoxy)benzoate hydrochloride (188.7 mg, 0.55 mmo), sodium iodide (245.9 mg, 1.65 mmol) and potassium carbonate (227.7 mg, 1.65 mmol) in acetonitrile (40 mL) was stirred at 80.degree. C. for 16 h. The reaction mixture was diluted with ethyl acetate (100 mL) and brine (50 mL), and the organic layer was separated, washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 20% ethyl acetate in petroleum ether), to give the target compound (240 mg, 74%) as a pale yellow oil. LCMS (ESI) m/z: 592.0 [M+H].sup.+.
Step 3: Preparation of methyl 5-cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl) piperidin-4-yl)methoxy)-2-fluorobenzoate
##STR01569##
[2765] A mixture of methyl 5-cyclopropyl-4-((1-(3,5-dichloro-2-iodobenzyl)piperidin-4-yl) methoxy)-2-fluorobenzoate (150 mg, 0.25 mmol) and copper cyanide (45.0 mg, 0.50 mmo) in N-methylpyrolidone (5.0 mL) was stirred under microwave at 150.degree. C. for 1.0 h. The reaction mixture was diluted with ethyl acetate (300 mL), filtered, the filtrate was washed with water (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 20% ethyl acetate in petroleum ether to give the target compound (35.0 mg, 14%) as a pale yellow oil. LCMS(ESI) m/z: 491.1 [M+H].sup.+.
Step 4: Preparation of 5-cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl) piperidin-4-yl)methoxy)-2-fluorobenzoic acid
##STR01570##
[2767] A mixture of methyl 5-cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl)piperidin-4-yl)methoxy)-2- -fluorobenzoate (35 mg, 0.07 mmol) and lithium hydroxide (42 mg, 1.75 mmol) in THF (5 mL) and water (5 mL) was stirred at room temperature for 16 h. The mixture was adjusted to pH 2-3 with HCl (2M), extracted with ethyl acetate (10.times.2 mL), the combined organic layers were washed with brine (10 mL), dried over anhydtous sodium sulfate, filtered and concentrated to give the crude product as a pale yellow solid. The solid was used in next step without further purification. LCMS(ESI) m/z: 477.1 [M+H].sup.+.
Step 5: Preparation of 5-Cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl) piperidin-4-yl)methoxy)-2-fluoro-N-(methylsulfonyl)benzamide
##STR01571##
[2769] A mixture of 5-cyclopropyl-4-((1-(3,5-dichloro-2-cyanobenzyl)piperidin-4-yl)methoxy)-2- -fluorobentzoic acid (30 mg, 0.07 mmol), methanesulfonamide (11 mg, 0.11 mmol), 1-ethyl-(3-dimethylaminopropyl)carbodiimide hydrochloride (27 mg, 0.14 mmol) and N,N-dimethyl-4-aminopyridine (17 mg, 0.14 mmol) in DCM (4 mL) was stirred at room temperature for 16 h. The reaction mixture was diluted with DCM (30 mL), washed with HCl (2.0 M, 5 mL), water (5 mL) and brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase Combiflash (30-40% MeCN in 0.1% NH.sub.4HCO.sub.3) to give the target product (23 mg, 38%) as a white solid. LCMS(ESI) Method A. RT=5.68 min, m/z: 554.1 [M+H].sup.+..sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 7.91 (d, J=4.0 Hz, 1H), 7.65 (d, J=4.0 Hz, 1H), 7.15 (d, J=8.4 Hz, 1H), 6.90 (d, J=16.0 Hz, 1H), 3.95 (d, J=4.0 Hz, 2H), 3.67 (s, 2H), 3.23 (s, 3H), 2.87-2.84 (m, 2H), 2.18-2.12 (m, 2H), 1.99-2.02 (m, 1H), 1.80-1.77 (m, 3H), 1.39-1.36 (m, 2H), 0.90-0.87 (m, 2H), 0.67-0.64 (m, 2H).
Example 595
Synthesis of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-cyanopiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01572## ##STR01573##
[2770] Step 1: Preparation of 1-(tert-butyl) 4-ethyl 4-cyanopiperidine-1,4-dicarboxylate
##STR01574##
[2772] Lithium hexamethyldisilazide (1 M, 20 mL, 20 mmol) was added dropwise to a solution of tert-butyl 4-cyanopiperidine-1-carboxylate (2.1 g, 10 mmol) in anhydrous THF (30 mL) at -78.degree. C. The resulting mixture was stirred at this temperature for 1 h, then ethyl carbonochloridate (2.2 g, 20 mmol) was added at -78.degree. C. and stirred at this temperature for 1 h. The reaction was quenched with sodium bicarbonate aqueous solution (1 M, 30 mL) and extracted with ethyl acetate (100 mL.times.3), the combined organic layers were washed with brine (20 mL). dried over anhydrous sodium sulfate, filtered and concentrated. The crode product was purified by silica column chromatography (eluting with 10-25% ethyl acetate of petroleum ether) to give product as colorless tui (2.8 g, 99%). LCMS (ESI) m/z: 1.83 [M-99].sup.+.
Step 2: Preparation of tert-butyl 4-cyano-4-(hydroxymethyl)piperidine-1-carboxylate
##STR01575##
[2774] Sodium borohydride (1.5 g, 40 mmol) was added to a solution of 1-tert-butyl 4-ethyl 4-cyanopiperidine-1,4-dicarboxylate (2.8 g, 10 mmol) in MeOH (30 mL) at 0.degree. C., the reaction mixture was stirred at room temperature for 1 h. The solvent was removed under reduced pressure and the crude was diluted with water (30 ml), extracted with ethyl acetate (50 mL.times.3), washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated to get crude product (2.4 g, 99%). The crude was used directly in the next step without further purification.
Step 3: Preparation of tert-butyl 4-cyano-4-(tosyloxymethyl)piperidine-1-carboxylate
##STR01576##
[2776] Sodium hydride (2.0 g, 50 mmol) was added to an ice-cooled solution of tert-butyl 4-cyano-4-(hydroxymethyl) piperidine-1-carboxylate (2.4 g, 10 mmol) in anhydrous THF (20 mL) and stirred for 1 h, then a solution of tosyl chloride (3.8 g, 20 mmol) in THF (10 mL) was added and the mixture was stirred at room temperature for 16 h. The mixture was quenched with water (40 mL), extracted with ethyl acetate (50 mL.times.3), washed with brine (40 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The crude product was purified by silica column chromatography (eluting with 25% ethyl acetate in petroleum ether) to give product as white solid (2.0 g, 50%). LCMS(ESI) m/z: 295.1 [M-99].sup.+.
Step 4: Preparation of tert-butyl 4-cyano-4-((2-cyclopropyl-5-fluoro-4-(methoxycarbonyl)-phenoxy) methyl) piperidine-1-carboxylate
##STR01577##
[2778] A mixture of tert-butyl 4-cyano-4-(tosyloxymethyl)piperidine-1-carboxylate (1.0 g, 2.5 mmol), methyl 5-cyclopropyl-2-fluoro-4-hydroxybenzoate (525 mg, 2.5 mmol) and potassium carbonate (1.1 g, 7.5 mmol) in DMF (10 mL) in a sealed tube was stirred at 120.degree. C. for 4 h. The reaction mixture was diluted with water (40 mL), extracted with ethyl acetate (50 mL.times.3), washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated to get crude product (1.0 g, 93%). The crude was used directly in the next step without further purification. LCMS(ESI) m/z: 433.1 [M+H].sup.+.
Step 5: Preparation of methyl 4-((4-cyanopiperidin-4-yl)methyoxy)-5-cyclopropyl-2-fluorobenzoate hydrochloride
##STR01578##
[2780] Hydrochloride in 1,4-dioxane (4 M, 20 mL) was added to a solution of tert-butyl 4-cyano-4-((2-cyclopropyl-5-fluoro-4-(methoxycarbonyl)phenoxy)methyl)pipe- ridine-1-carboxylate (1.0 g, 2.3 mmol) in 1,4-dioxane, the reaction mixture was stirred at room temperature for 1 h. The solution was concentrated to give a brown solid, which was recrystallized in ethyl acetate (4 mL) to give the target compound as a gray solid (0.6 g, 74%). LCMS(ESI) m/z: 333.1 [M-HCl+1].sup.+.
Step 6: Preparation of (3-chloro-2-fluoro-5-(trifluoromethyl)phenyl)methanol
##STR01579##
[2782] Borane-tehrahydrofuran complex (1M, 20 mL, 20 mmol) was mixed with 3-chloro-2-fluoro-5-(trifluoromethyl)benzoic acid (480 mg, 2 mmol) at 0.degree. C. The reaction mixture was stirred at room temperature the 1 h, quenched with MeOH (20 mL). The solvents were removed under reduced pressure to give crude product (400 mg, 87%) which was used directly in the next step without further purification. LCMS(ESI) m/z: 227.1 [M-1].sup.+.
Step 7: Preparation of 3-chloro-2-fluoro-5-(trifluoromethyl)piperidin methanesulfonate
##STR01580##
[2784] Methanesulfonyl chloride (60 mg, 0.52 mmol) was added dropwise to a mixture of (3-chloro-2-fluoro-5-(trifluoromethyl) phenyl)methanol (100 mg, 0.44 mmol) and triethylamine (90 mg, 0.88 mmol) in DCM (10 mL) at 0.degree. C. The reaction mixture was stirred at room temperature for 16 h, diluted with DCM (20 mL), washed with water (10 mL.times.2), dried over anhydrous sodium sulfate, filtered and concentrated to get crude product (130 mg, 96%). The crude was used directly in the next step without further purification. LCMS(ESI) m/z: 307.1 [M+1].sup.+.
Step 8: Preparation of methyl 4-((1-(3chloro-2-fluoro-5-(trifluoromethyl) benzyl)-4-cyanopiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate
##STR01581##
[2786] A mixture of 3-chloro-2-fluoro-5-(trifluoromethyl)piperidin methanesulfonate (100 mg, 0.33 mmol), methyl 4-((4-cyanopiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoate hydrochloride (122 mg, 0.33 mmol) and potassium carbonate (137 mg, 0.99 mmol) in acetonitrile (10 mL) was stirred at 80.degree. C. for 16 h. The reaction mixture was diluted with ethyl acetate (100 mL) and brine (50 mL), washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (eluting with 20% ethyl acetate in petroleum ether) to give the target compound (80 mg, 45%) as a pale yellow oil. LCMS(ESI) m/z: 543.0 [M+H].sup.+.
Step 9: Preparation of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl) benzyl)-4-cyanopiperidin-4-yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid
##STR01582##
[2788] A mixture of methyl 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-cyanopiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluorobenzoate (80 mg, 0.15 mmol) and lithium hydroxide (36 mg, 1.5 mmol) in THF (3 mL) and water (3 mL) was stirred at 50 .degree. C. for 3 h. The reaction mixture was adjusted pH 2-3 with HCl (2M), extracted with ethyl acetate (10.times.2 mL), washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated to give the product (77 mg, 99%) as a pale yellow solid. LCMS(ESI) m/z: 529.1 [M+H].sup.30 .
Step 10: Preparation of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-cyanopiperidin-4- -yl) methoxy)-5-cyclopropyl-2-fluoro-N-(methylsulfonyl)benzamide
##STR01583##
[2790] A mixture of 4-((1-(3-chloro-2-fluoro-5-(trifluoromethyl)piperidin)-4-cyanopiperidin-4- -yl)methoxy)-5-cyclopropyl-2-fluorobenzoic acid (77 mg, 0.16 mmol), methanesulfonamide (23 mg, 0.24 mmol), 1-ethyl-(3-dimethylaminopropyl)carbodiimide hydrochloride (61 mg, 0.32 mmol) and N,N-dimethyl-4-aminopyridine (39 mg, 0.32 mmol) in DCM (4 mL) was stirred at room temperature for 16 h. The reaction mixture was diluted with DCM (100 mL), washed with HCl (2.0 M, 20 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by reverse phase combiflash (40-50% MeCN in 0.1% NH.sub.4HCO.sub.3) to give the target product (40 mg, 41%) as white solid. LCMS(ESI) Method A: RT=5.91 min, m/z: 606.0 [M+H].sup.+. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 8.03 (d, J=5.2 Hz, 1H), 7.8 (d, J=3.2 Hz, 1H), 7.21 (d, J=7.6 Hz, 1H), 6.92-6.87 (m, 1H), 4.21 (s, 2H), 3.72 (s, 2H), 3.14 (s, 3H), 2.91-2.88 (m, 2H), 2.33-2.76 (m, 2H), 2.06-2.03 (m, 3H), 1.77-1.71 (m, 2H), 0.90-0.87 (m, 2H), 0.65-0.62 (m, 2H).
Examples 596-598
[2791] Using procedures similar to those described herein the following compounds of fornula (I) were also prepared.
##STR01584##
[2792] Example 596: M+H 588.23; Example 597: M+H 568.22; and Example 598: M+H 568.22
Example 599
Electrophysiological Assay (EP) (In Vitro Assay)
[2793] Patch voltage clamp electrophysiology allows for the direct measurement and quantification of block of voltage-gated sodium channels (NaV's), and allows the determination of the time- and voltage-dependence of block which has been interpreted as differential binding to the resting, open, and inactivated states of the sodium channel (Hille B., Journal of General Physiology (1977), 69: 497-515).
[2794] The following patch voltage clamp electrophysiology studies were performed on representative compounds of the invention using human embryonic kidney cells (HEK), permanently transfected with an expression vector containing the full-length cDNA coding for the desired human sodium channel .alpha.-subunit, grown in culture media containing 10% FBS, 1% PSG, and 0.5 mg/mL G418 at 37.degree. C. with 5% CO2. HEK cells used for the electrophysiology (EP) recordings had a passage number of less than 40 for all studies and were used within three days from the time of plating. NaV1.7 and NaV1.5 cDNAs (NM_002977 and AC137587; SCN5A, respectively) were stably expressed in HEK-293 cells. The .beta.1 subunit was coexpressed in both the NaV1.7 and NaV1.5 cell lines.
[2795] Sodium currents were measured using the patch clamp technique in the whole-cell configuration using either a PatchXpress automated voltage clamp or manually using an Axopatch 200B (Axon Instruments) or Model 2400 (A-M systems) amplifier. The manual voltage clamp protocol was as follows: Borosilicate glass micropipettes were fire-polished to a tip diameter yielding a resistance of 2-4 Mohms in the working solutions. The pipette was filled with a solution comprised of: 5 mMNaCl, 10 mMCsCl, 120 mMCsF, 0.1 mM CaCl2, 2 mM MgCl2, 10 mM HEPES, 10 mM EGTA; and adjusted to pH 7.2 with CsOH. The external solution had the following composition: 140 mMNaCl, 5 mMKCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM HEPES; and adjusted to pH 7.4 with NaOH. In some studies, the external sodium was reduced by equimolar replacement with choline. Osmolarity in the CsF internal and NaCl external solutions was adjusted to 300 mOsm/kg and 310 mOsm/kg with glucose, respectively. All recordings were performed at ambient temperature in a bath chamber with a volume of 150 .mu.L. Control sodium currents were measured in 0.5% DMSO. Controls and representative compounds of the invention were applied to the recording chamber through a 4-pinch or 8-pinch valve bath perfusion system manufactured by ALA Scientific Instruments.
[2796] Currents were recorded at 40 kHz sampling frequency, filtered at 5 Hz, and stored using a Digidata-1322A analogue/digital interface with the pClamp software (Axon Instruments). Series resistance compensation was applied (60-80%). Cells were rejected if currents showed inadequate voltage control (as judged by the IV relationship during stepwise activation). All statistics in this study are given as mean .+-.SD.
[2797] The membrane potential was maintained at a voltage where inactivation of the channel is complete (which was -60 mV for both NaV1.7 and NaV1.5). The voltage is then stepped back to a very negative (Vhold=150 mV) voltage for 20 ms and then a test pulse is applied to quantify the compound block. The 20 ms brief repolarization was long enough for compound-free channels to completely recover from fast inactivation, but the compound-bound channels recovered more slowly such that negligible recovery could occur during this interval. The percent decrease in sodium current following wash-on of compound was taken as the percent block of sodium channels. Data for representative compounds of fornula (I) is provided in Table 1.
Example 600
Tritiated Sulfonamide Binding to Membranes Isolated from Cells that Heterologously Express hNav1.7 and the .beta.1 Subunit
[2798] Preparation of membranes containing recombinantly expressed sodium channels: Frozen recombinant cell pellets were thawed on ice and diluted to 4 times the cell pellet weight with ice cold 50 mMTrisHCl, pH 7.4 buffer. The cell suspensions were homogenized on ice using a motorized glass douncehomogeniser. Homogenates were further diluted 8.4 times with ice cold 50 mMTrisHCl, pH 7.4 buffer and then centrifuged at 200.times.g, at 4.degree. C. for 15 min. The supetnatants were collected and centrifuged at 10000.times.g, at 4.degree. C. for 50 min. The pellets were then re-suspended in 100 mMNaCl, 20 mMTrisHCl, pH 7.4 buffer containing 1% v/v protease inhibitor (Calbiochem) and re-homogenized on ice. The homogenized membranes were then processed through a syringe equipped with a 26 gauge needle. Protein concentrations were determined by Bradford Assay and the membranes were stored at -80.degree. C.
[2799] Radioligand Binding Studies: Saturation experiments. A representative compound of formula (I) having a methyl group was tritiated. Three tritiums were incorporated in place of methyl hydrogens to generate [.sup.3H] compound. Binding of this radioligand was preformed in 5 mL borosilicate glass test tubes at room temperature. Binding was initiated by adding membranes to increasing concentrations of [.sup.3H] compound in 100 mMNaCl, 20 mMTrisHCl, pH 7.4 buffer containing 0.01% w/v bovine serum albumin (BSA) for 18 h. Non-specific binding was determined in the presence of 1 .mu.M unlabelled compound. After 18 h, the reactants were filtered through GF/C glass fiber filters presoaked in 0.5% w/v polyethylene imine. Filters were washed with 15 mL ice cold 100 mMNaCl, 20 mMTrisHCl, pH 7.4 buffer containing 0.25% BSA to separate bound from free ligand. [.sup.3H] compound bound to filters was quantified by liquid scintillation counting.
[2800] Competitive binding experiments: Binding reactions were preformed in 96-well polypropylene plates at room temperature for 18 h. In 360 .mu.L, membranes were incubated with 100 pM [.sup.3H] compound and increasing concentrations of Test Compound. Non-specific binding was defined in the presence of 1 .mu.M unlabelled compound. Reactions were transferred and filtered through 96-well glass fiber/C filter plates presoaked with 0.5% polyethylene imine. The filtered reactions were washed 5 times with 200 .mu.L ice cold buffer containing 0.25% BSA. Bound radioactivity was determined by liquid scintillation counting.
[2801] Data Analysis: For saturation experiments, non-specific binding was subtracted from total binding to provide specific binding and these values were recalculated in terms of pmol ligand bound per mg protein. Saturation curves were constructed and dissociation constants were calculated using the single site ligand binding model: Beq=(Bmax*X)/(X+Kd), where Beq is the amount of ligand bound at equilibrium, Bmax is the maximum receptor density, Kd is the dissociation constant for the ligand, and X is the free ligand concentration. For competition studies percent inhibition was determined and IC.sub.50 values were calculated using a 4 parameter logistic model (% inhibition=(A+((B-A) /(l+((x/C){circumflex over ( )}D)))) using XLfit, where A and B are the maximal and minimum inhibitin respectively, C is the IC.sub.50 concentration and D is the (Hill) slope.
[2802] Representative compounds, when tested in this model, demonstrated affinities as set forth in Table 1.
TABLE-US-00001 TABLE 1 Ligand Binding NaV1.7 NaV1.5 Assay EP EP Example Structure (.mu.M) (.mu.M) (.mu.M) 1 ##STR01585## 0.0356 0.0226 2.7559 2 ##STR01586## 4.1167 1.0769 3 ##STR01587## 0.0026 4 ##STR01588## 6.5995 5 ##STR01589## 0.0071 6 ##STR01590## 0.0030 0.0009 0.0331 7 ##STR01591## 0.0035 8 ##STR01592## 0.0081 9 ##STR01593## 0.0035 10 ##STR01594## 0.0041 11 ##STR01595## 0.0474 12 ##STR01596## 0.0113 13 ##STR01597## 1.4894 14 ##STR01598## 0.3181 15 ##STR01599## 0.0048 16 ##STR01600## 0.0034 17 ##STR01601## 0.0048 18 ##STR01602## 0.0026 19 ##STR01603## 0.0049 20 ##STR01604## 0.0309 21 ##STR01605## 0.0112 22 ##STR01606## 0.0182 23 ##STR01607## 0.0017 24 ##STR01608## 0.0060 25 ##STR01609## 0.0032 26 ##STR01610## 0.0061 27 ##STR01611## 0.0036 0.1689 28 ##STR01612## 0.0074 29 ##STR01613## 0.0017 30 ##STR01614## 0.0027 31 ##STR01615## 0.0026 32 ##STR01616## 0.0054 33 ##STR01617## 0.0132 34 ##STR01618## 0.0019 35 ##STR01619## 0.0024 36 ##STR01620## 0.0036 37 ##STR01621## 0.0151 38 ##STR01622## 0.0039 39 ##STR01623## 0.0068 40 ##STR01624## 0.0084 41 ##STR01625## 0.0134 42 ##STR01626## 0.0021 43 ##STR01627## 0.0049 44 ##STR01628## 0.0036 45 ##STR01629## 4.5841 46 ##STR01630## 0.9205 47 ##STR01631## 0.0251 48 ##STR01632## 2.8179 49 ##STR01633## 0.0043 0.0054 0.199 50 ##STR01634## 0.0019 51 ##STR01635## 0.0021 52 ##STR01636## <0.0016 53 ##STR01637## 0.0080 54 ##STR01638## 0.0220 55 ##STR01639## 0.0233 56 ##STR01640## 0.0635 57 ##STR01641## 0.0038 ##STR01642## 58 ##STR01643## ##STR01644## 59 ##STR01645## 0.076 60 ##STR01646## 1.6 61 ##STR01647## 0.057 62 ##STR01648## 0.047 63 ##STR01649## 0.046 64 ##STR01650## 0.0047 65 ##STR01651## 0.034 66 ##STR01652## 0.091 67 ##STR01653## 0.0079 68 ##STR01654## 0.0064 69 ##STR01655## 0.0061 70 ##STR01656## 0.006 71 ##STR01657## 0.0031 72 ##STR01658## 0.75 73 ##STR01659## 1.3 74 ##STR01660## 3.6 75 ##STR01661## 0.074 76 ##STR01662## 0.042 77 ##STR01663## 0.0064 78 ##STR01664## 0.49 79 ##STR01665## 0.0099 80 ##STR01666## 0.00405 81 ##STR01667## 0.63 82 ##STR01668## 0.408 83 ##STR01669## 0.059 84 ##STR01670## 1.24 85 ##STR01671## 1.86 86 ##STR01672## 0.0495 87 ##STR01673## 0.278 88 ##STR01674## 0.0928 89 ##STR01675## 0.0223 90 ##STR01676## 0.00287 91 ##STR01677## 0.0147 92 ##STR01678## 0.0305 93 ##STR01679## 0.173 94 ##STR01680## 0.15 95 ##STR01681## 1.06 96 ##STR01682## 0.429 97 ##STR01683## 0.00269 98 ##STR01684## 0.00751 99 ##STR01685## 0.00389 100 ##STR01686## 0.00419 0.0058 0.552 101 ##STR01687## 0.00376 102 ##STR01688## 0.00254 103 ##STR01689## 0.00524 104 ##STR01690## 0.0451 105 ##STR01691## 0.0163 106 ##STR01692## 0.0048 107 ##STR01693## 0.0021 108 ##STR01694## 0.094 109 ##STR01695## 0.0536 110 ##STR01696## 0.0411 111 ##STR01697## 0.0204 112 ##STR01698## 0.00335 113 ##STR01699## 0.0111 114 ##STR01700## 0.00727 115 ##STR01701## 0.0042 116 ##STR01702## 0.0775 117 ##STR01703## 0.411 118 ##STR01704## 0.417 119 ##STR01705## 0.306
120 ##STR01706## 0.0206 121 ##STR01707## 0.0301 122 ##STR01708## 0.0024 123 ##STR01709## 0.0029 124 ##STR01710## 0.0713 125 ##STR01711## 0.025 126 ##STR01712## 0.282 127 ##STR01713## 0.578 128 ##STR01714## 0.0056 129 ##STR01715## 1.37 130 ##STR01716## 0.0035 131 ##STR01717## 0.00299 132 ##STR01718## 0.014 133 ##STR01719## 0.118 134 ##STR01720## 0.346 135 ##STR01721## 0.0155 136 ##STR01722## 0.00188 137 ##STR01723## 0.00205 138 ##STR01724## 0.0112 139 ##STR01725## 0.0049 140 ##STR01726## 0.0035 141 ##STR01727## 0.0039 142 ##STR01728## 0.00657 143 ##STR01729## 0.0026 144 ##STR01730## 0.00915 145 ##STR01731## 0.00622 146 ##STR01732## 0.276 147 ##STR01733## 0.00603 148 ##STR01734## 0.015 149 ##STR01735## 0.0737 150 ##STR01736## 0.100 151 ##STR01737## 0.00994 152 ##STR01738## 0.443 153 ##STR01739## 0.439 154 ##STR01740## 0.00912 155 ##STR01741## 0.00202 156 ##STR01742## 0.021 157 ##STR01743## 0.0351 158 ##STR01744## 0.0123 159 ##STR01745## 0.00542 160 ##STR01746## 0.0628 161 ##STR01747## 0.284 162 ##STR01748## 0.0039 0.0028 163 ##STR01749## 0.0036 164 ##STR01750## 0.0043 0.0033 165 ##STR01751## 0.0023 0.0024 0.049 166 ##STR01752## 0.0039 167 ##STR01753## 0.0049 0.0042 168 ##STR01754## 0.0062 0.003 0.078 169 ##STR01755## 0.0063 0.003 0.28 170 ##STR01756## 0.005 171 ##STR01757## 0.005 172 ##STR01758## 0.0039 173 ##STR01759## 0.0039 174 ##STR01760## 0.012 175 ##STR01761## 0.0052 176 ##STR01762## 0.0091 177 ##STR01763## 0.0083 178 ##STR01764## 0.0076 179 ##STR01765## 1.8 180 ##STR01766## 9.9 181 ##STR01767## 0.011 182 ##STR01768## 0.014 183 ##STR01769## 0.047 184 ##STR01770## 0.0051 185 ##STR01771## 0.007 186 ##STR01772## 0.004 187 ##STR01773## 0.005 188 ##STR01774## 0.0052 189 ##STR01775## 0.016 190 ##STR01776## 0.056 191 ##STR01777## 0.032 192 ##STR01778## 0.034 193 ##STR01779## 0.052 194 ##STR01780## 0.18 195 ##STR01781## 0.35 196 ##STR01782## 0.043 197 ##STR01783## 3.2 198 ##STR01784## 0.55 199 ##STR01785## 1.4 200 ##STR01786## 9.4 201 ##STR01787## 0.005 202 ##STR01788## 0.0016 203 ##STR01789## 0.0036 204 ##STR01790## 0.0027 205 ##STR01791## 0.0025 206 ##STR01792## 0.0018 207 ##STR01793## 0.0017 208 ##STR01794## 0.0016 0.003 0.013 209 ##STR01795## 0.0017 0.0158 210 ##STR01796## 0.0019 211 ##STR01797## 0.0017 212 ##STR01798## 0.0027 213 ##STR01799## 0.0025 214 ##STR01800## 0.0031 215 ##STR01801## 0.0048 216 ##STR01802## 0.005 217 ##STR01803## 0.0056 0.0025 0.19 218 ##STR01804## 0.0078 219 ##STR01805## 0.0083 220 ##STR01806## 0.017 221 ##STR01807## 1.8 222 ##STR01808## 0.16 223 ##STR01809## 0.25 224 ##STR01810## 0.0045 225 ##STR01811## 0.017 226 ##STR01812## 0.012 227 ##STR01813## 0.0081 228 ##STR01814## 0.0051 0.031 4.1 229 ##STR01815## 0.0073 230 ##STR01816## 0.0044 231 ##STR01817## 0.0059 232 ##STR01818## 0.064 233 ##STR01819## 0.031 234 ##STR01820## 0.17 235 ##STR01821## 0.21 236 ##STR01822## 0.0016 237 ##STR01823## 0.0029 238 ##STR01824## 0.0022 239 ##STR01825## 0.0034 240 ##STR01826## 0.0024 241 ##STR01827## 0.0027 242 ##STR01828## 0.003 243 ##STR01829## 0.011 244 ##STR01830## 0.012 245 ##STR01831## 0.0079
246 ##STR01832## 0.0077 247 ##STR01833## 0.0089 248 ##STR01834## 0.01 249 ##STR01835## 0.039 250 ##STR01836## 0.043 251 ##STR01837## 0.049 252 ##STR01838## 0.05 253 ##STR01839## 0.02 254 ##STR01840## 0.026 255 ##STR01841## 1.1 256 ##STR01842## 0.29 257 ##STR01843## 0.27 258 ##STR01844## 0.065 259 ##STR01845## 0.0022 0.023 0.11 260 ##STR01846## 0.0048 261 ##STR01847## 0.0048 262 ##STR01848## 0.018 263 ##STR01849## 0.02 264 ##STR01850## 0.0092 265 ##STR01851## 0.10 266 ##STR01852## 0.091 267 ##STR01853## 0.047 268 ##STR01854## 0.006 0.0028 269 ##STR01855## 0.0034 270 ##STR01856## 0.005 0.0058 271 ##STR01857## 0.0037 272 ##STR01858## 0.0042 273 ##STR01859## 0.0085 274 ##STR01860## 0.068 275 ##STR01861## 0.0069 276 ##STR01862## 0.0057 277 ##STR01863## 0.004 278 ##STR01864## 0.0033 279 ##STR01865## 0.004 280 ##STR01866## 0.0081 281 ##STR01867## 0.0047 282 ##STR01868## 0.0044 283 ##STR01869## 0.004 284 ##STR01870## 0.0036 285 ##STR01871## 0.0033 286 ##STR01872## 0.0054 287 ##STR01873## 0.0035 288 ##STR01874## 0.0035 289 ##STR01875## 0.0035 290 ##STR01876## 0.0048 291 ##STR01877## 0.011 292 ##STR01878## 0.0029 293 ##STR01879## 0.0052 294 ##STR01880## 0.0038 295 ##STR01881## 0.0023 296 ##STR01882## 0.0081 297 ##STR01883## 0.0108 298 ##STR01884## 0.0046 299 ##STR01885## 0.0019 300 ##STR01886## 0.0016 301 ##STR01887## 0.003 302 ##STR01888## 0.0057 303 ##STR01889## 0.0065 304 ##STR01890## 0.0037 305 ##STR01891## 0.0039 306 ##STR01892## 0.0060 307 ##STR01893## 0.0067 308 ##STR01894## 0.0060 309 ##STR01895## 0.0027 0.003 0.38 310 ##STR01896## 0.0023 311 ##STR01897## 0.0029 312 ##STR01898## 0.0022 <0.001 0.31 313 ##STR01899## 0.0091 314 ##STR01900## >10 315 ##STR01901## 0.0191 316 ##STR01902## 0.0372 317 ##STR01903## 0.0060 318 ##STR01904## 0.0026 319 ##STR01905## 0.0054 320 ##STR01906## 0.2133 321 ##STR01907## 0.1907 322 ##STR01908## 0.0019 323 ##STR01909## 0.0019 324 ##STR01910## 0.0027 325 ##STR01911## 0.0024 326 ##STR01912## 0.0029 327 ##STR01913## 0.0027 328 ##STR01914## 0.0064 329 ##STR01915## 0.0033 330 ##STR01916## 0.0319 331 ##STR01917## 0.0322 332 ##STR01918## 0.0352 333 ##STR01919## 0.0207 334 ##STR01920## 0.0071 335 ##STR01921## 0.0143 336 ##STR01922## 0.0049 337 ##STR01923## 0.01108 338 ##STR01924## 0.0074 339 ##STR01925## 0.0165 340 ##STR01926## 0.0047 341 ##STR01927## 0.0044 342 ##STR01928## 0.0057 343 ##STR01929## 0.0084 344 ##STR01930## 0.0069 345 ##STR01931## 0.0078 346 ##STR01932## 0.0044 0.0022 682 347 ##STR01933## 0.0063 348 ##STR01934## 0.0045 349 ##STR01935## 0.0040 350 ##STR01936## 0.0045 351 ##STR01937## 0.0053 352 ##STR01938## 0.0020 353 ##STR01939## 0.011 354 ##STR01940## 2.6695 355 ##STR01941## 0.11876 356 ##STR01942## 0.0058 357 ##STR01943## 0.0130 358 ##STR01944## 0.0140 359 ##STR01945## 0.0114 360 ##STR01946## 0.0128 361 ##STR01947## 0.0020 362 ##STR01948## 0.0029 363 ##STR01949## 0.0058 364 ##STR01950## 0.0806 365 ##STR01951## 0.3272 366 ##STR01952## 0.0091 367 ##STR01953## 0.0083 368 ##STR01954## 0.0251 369 ##STR01955## 0.0145 370 ##STR01956## 0.0308 371 ##STR01957## 0.0203
372 ##STR01958## 0.04923 373 ##STR01959## 0.0197 374 ##STR01960## 0.0231 375 ##STR01961## 0.0146 376 ##STR01962## 2.4491 377 ##STR01963## 1.2589 378 ##STR01964## 0.0459 379 ##STR01965## 0.0172 380 ##STR01966## 0.0409 381 ##STR01967## 0.0148 382 ##STR01968## 0.0407 383 ##STR01969## 0.0148 384 ##STR01970## 0.0163 385 ##STR01971## 0.0100 386 ##STR01972## 0.0521 387 ##STR01973## 0.0133 388 ##STR01974## 0.0163 389 ##STR01975## 0.0088 390 ##STR01976## 0.0185 391 ##STR01977## 0.0101 392 ##STR01978## 2.0625 393 ##STR01979## 1.3750 394 ##STR01980## 6.0057 395 ##STR01981## >10 396 ##STR01982## 0.0148 397 ##STR01983## 0.0057 398 ##STR01984## 0.0051 399 ##STR01985## 0.0046 400 ##STR01986## 0.0021 401 ##STR01987## 0.0031 402 ##STR01988## 0.0224 403 ##STR01989## 0.0170 404 ##STR01990## 0.0201 405 ##STR01991## 0.0150 406 ##STR01992## 0.0176 407 ##STR01993## 0.0067 408 ##STR01994## 0.0227 409 ##STR01995## 0.0540 410 ##STR01996## 0.0109 411 ##STR01997## 0.0008 412 ##STR01998## 0.0015 413 ##STR01999## 0.0060 414 ##STR02000## 0.0038 415 ##STR02001## 0.0061 416 ##STR02002## 0.0053 417 ##STR02003## 0.0057 418 ##STR02004## 0.0032 419 ##STR02005## 0.0052 420 ##STR02006## 0.0024 421 ##STR02007## 0.0025 422 ##STR02008## 0.0013 423 ##STR02009## 0.0021 424 ##STR02010## 0.0025 425 ##STR02011## 0.0029 426 ##STR02012## 0.0031 427 ##STR02013## 0.0190 428 ##STR02014## 0.0234 429 ##STR02015## 0.0519 430 ##STR02016## 0.5327 431 ##STR02017## 0.0032 432 ##STR02018## 0.0033 433 ##STR02019## 0.0062 434 ##STR02020## 0.0050 0.0128 435 ##STR02021## 0.0054 436 ##STR02022## 0.0054 437 ##STR02023## 0.0073 438 ##STR02024## 0.0095 439 ##STR02025## 0.0110 440 ##STR02026## 0.0003 441 ##STR02027## 0.1378
























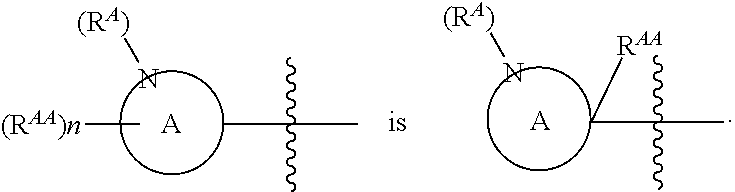
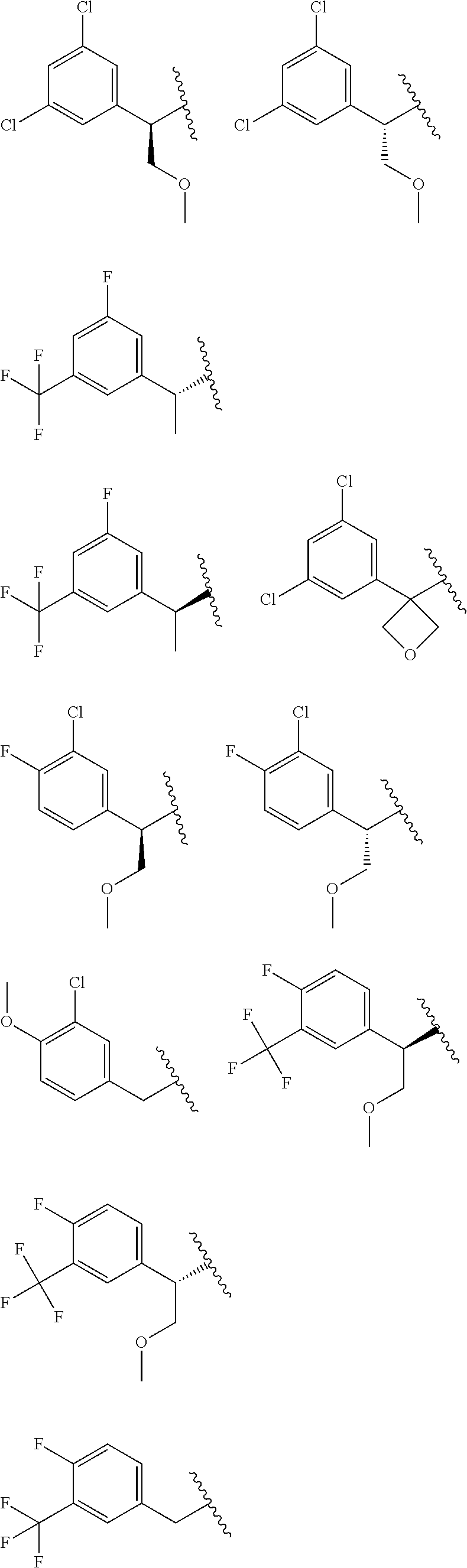

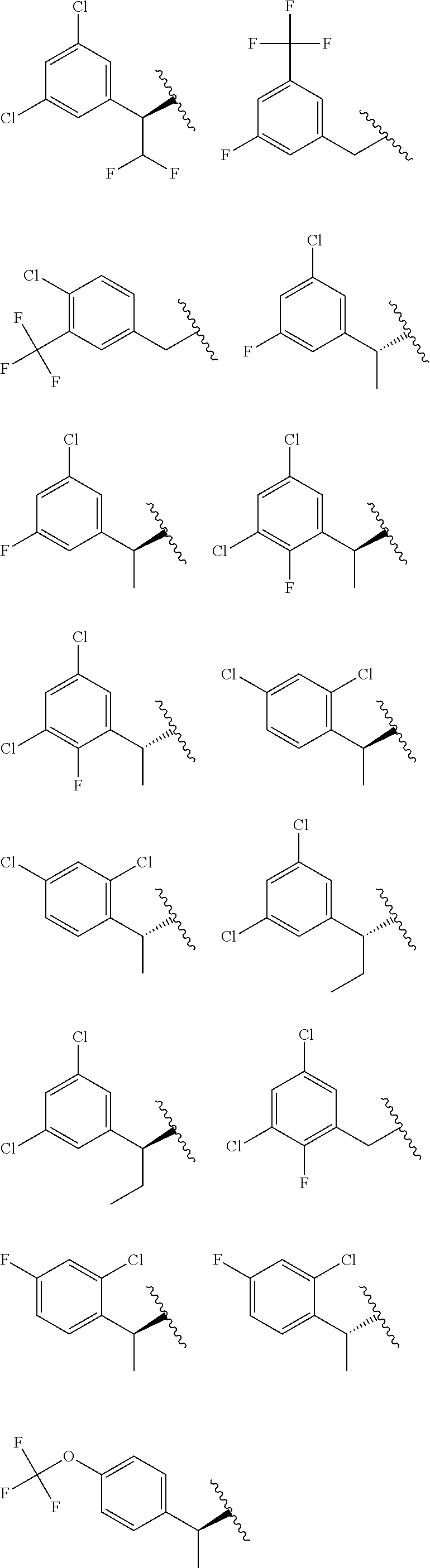










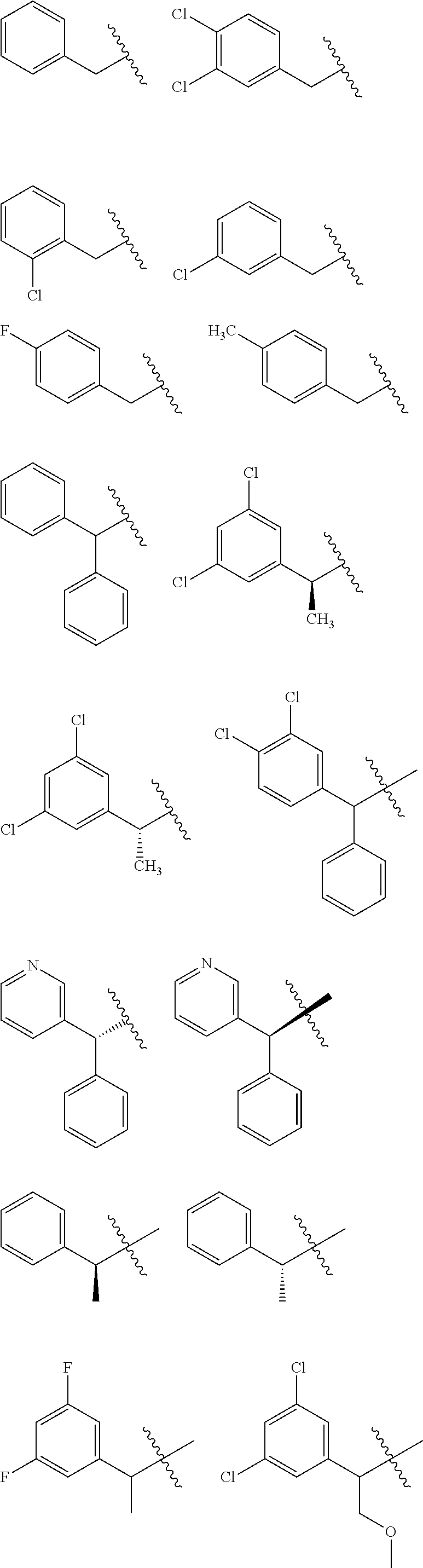




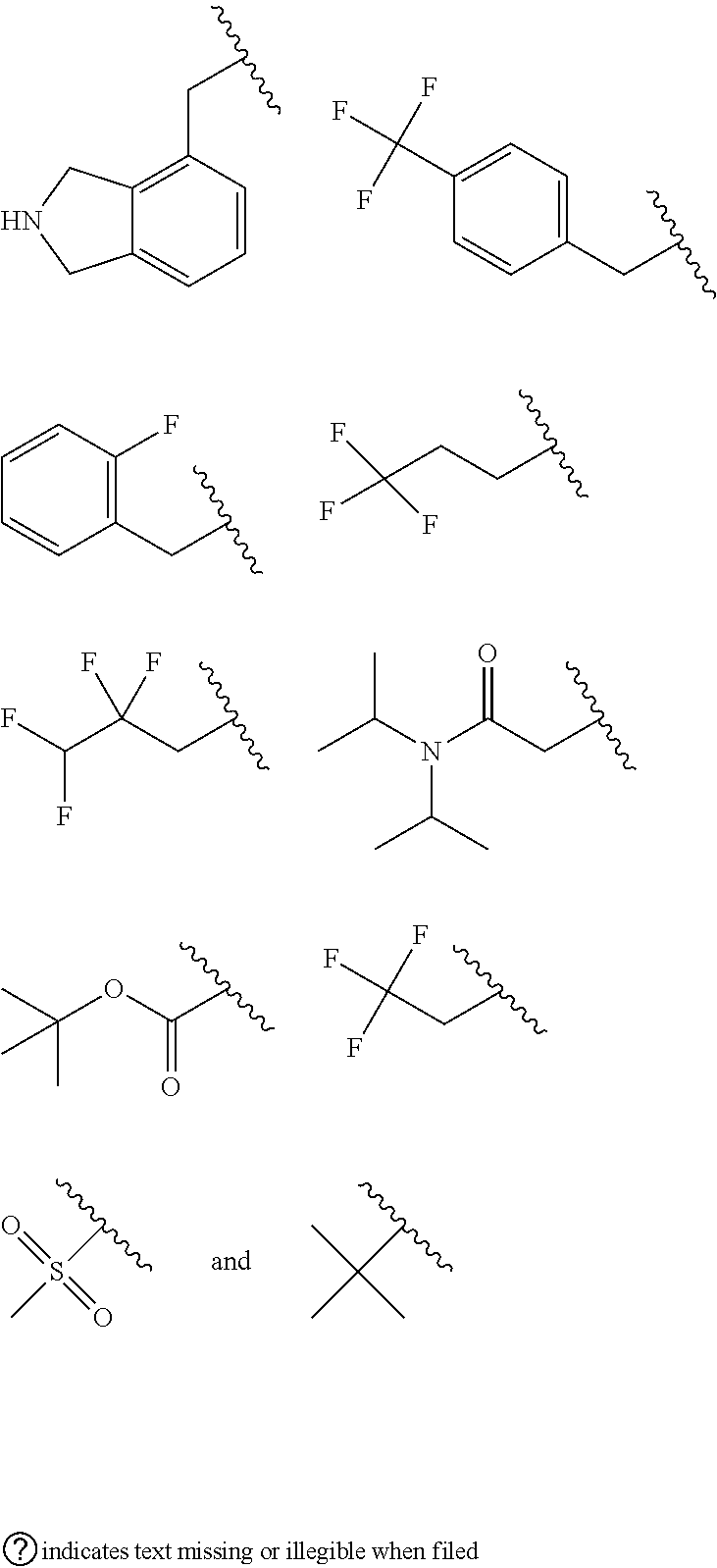


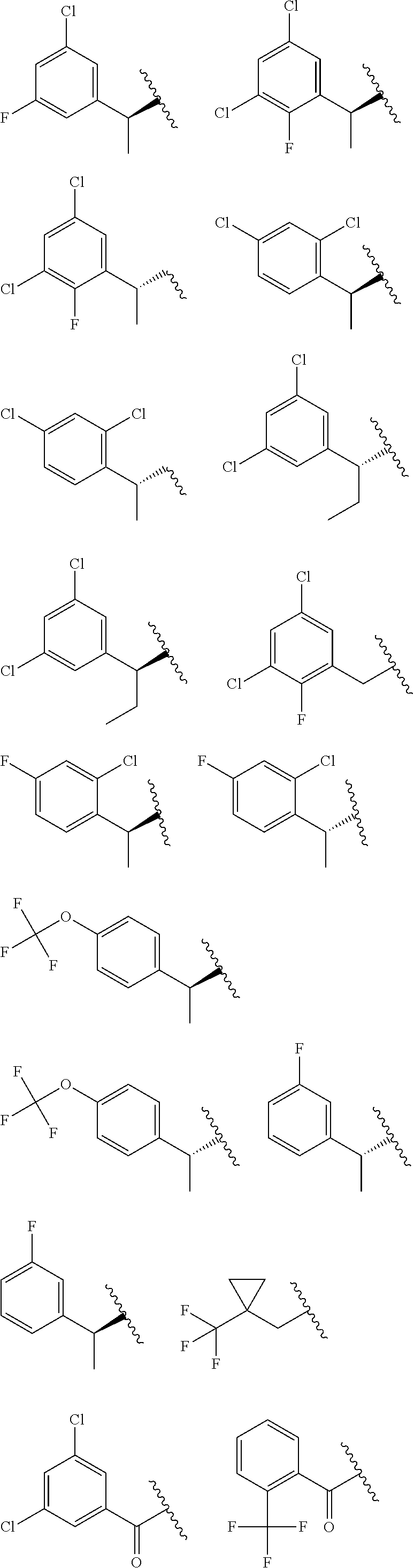




















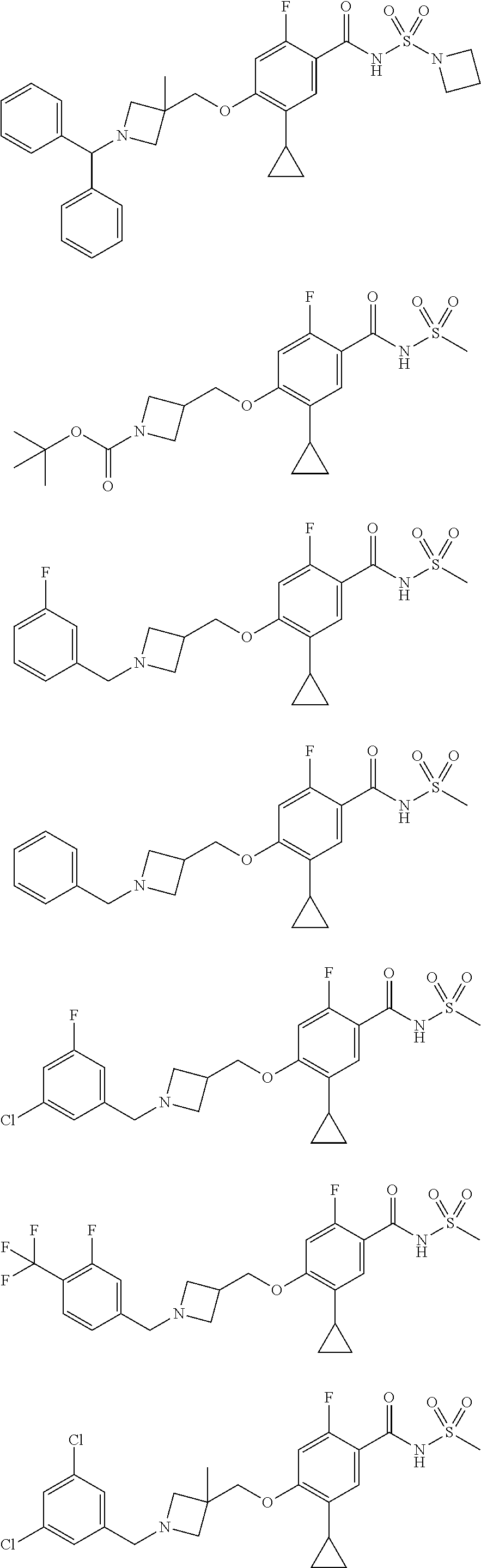







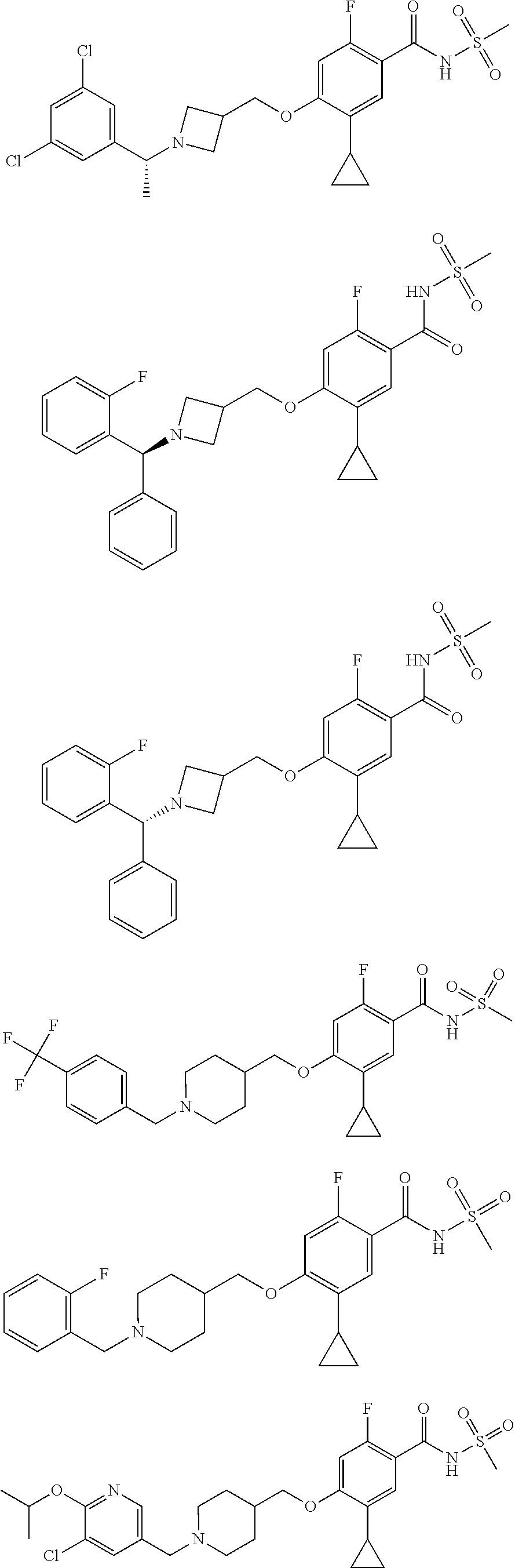





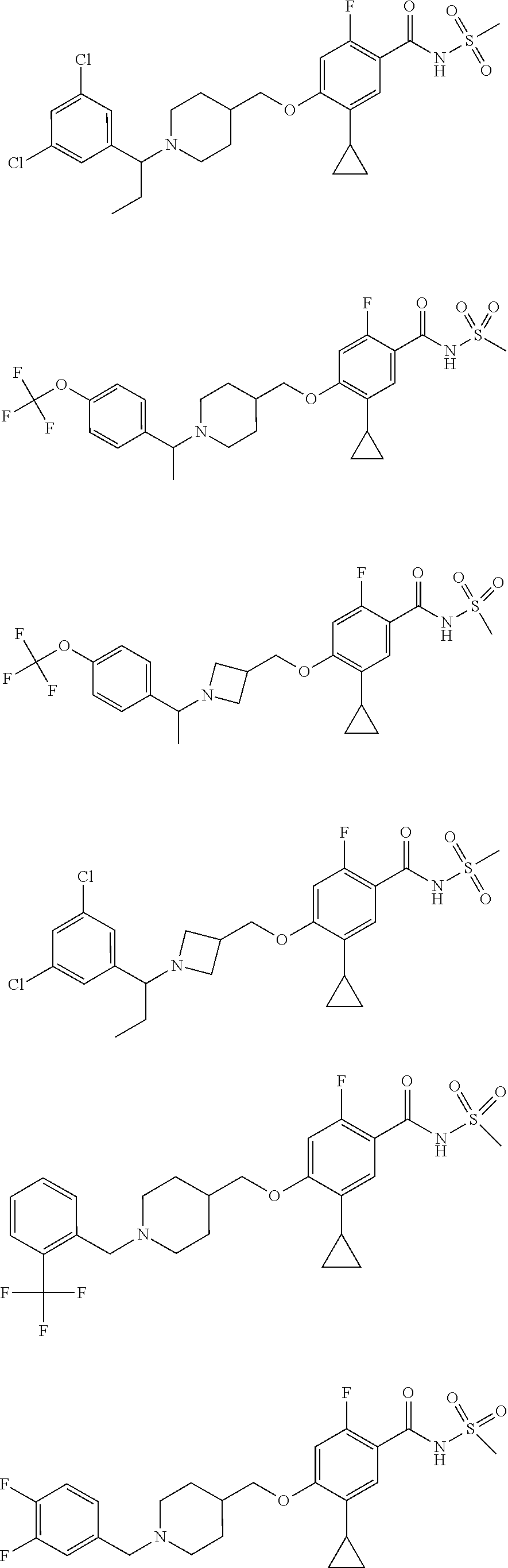


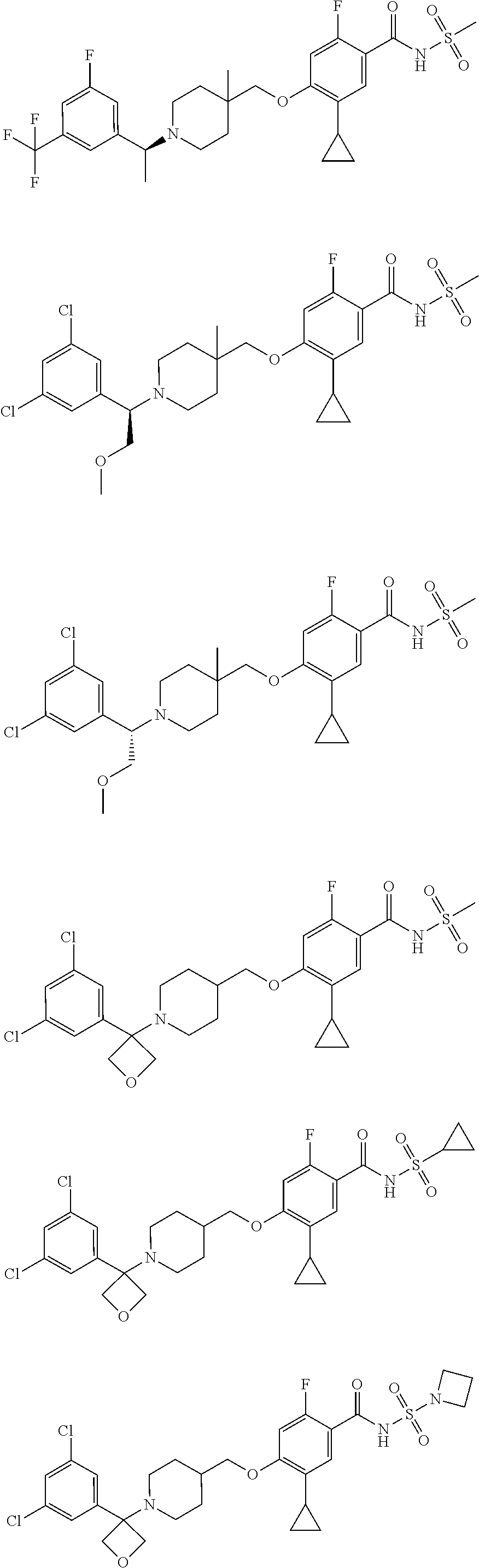
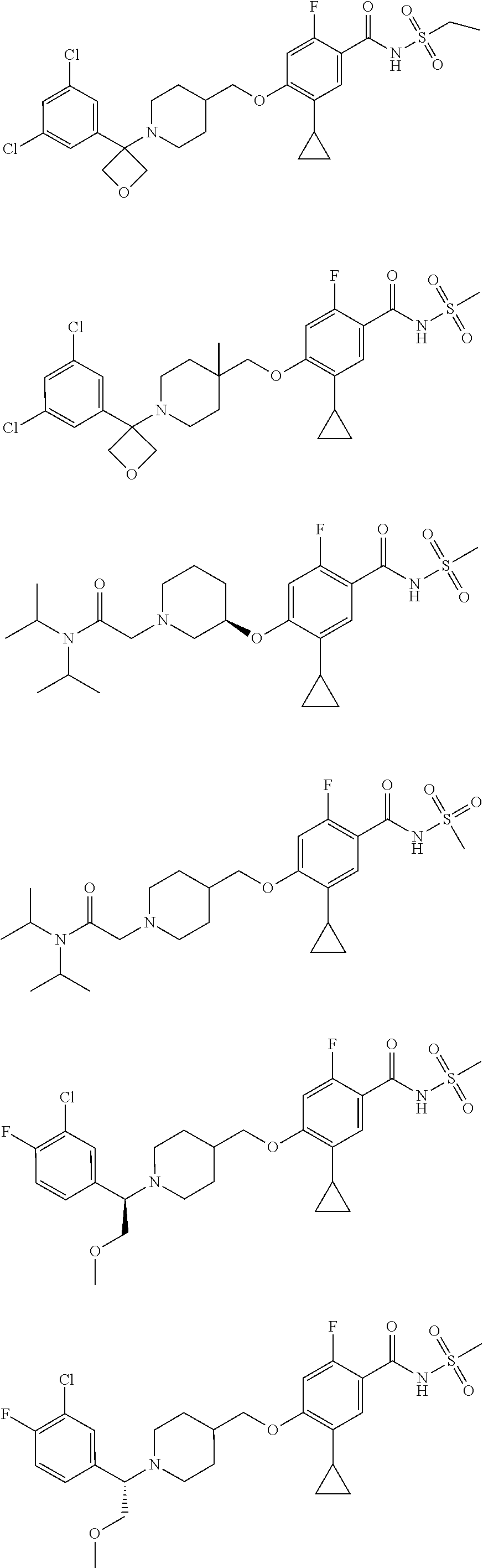




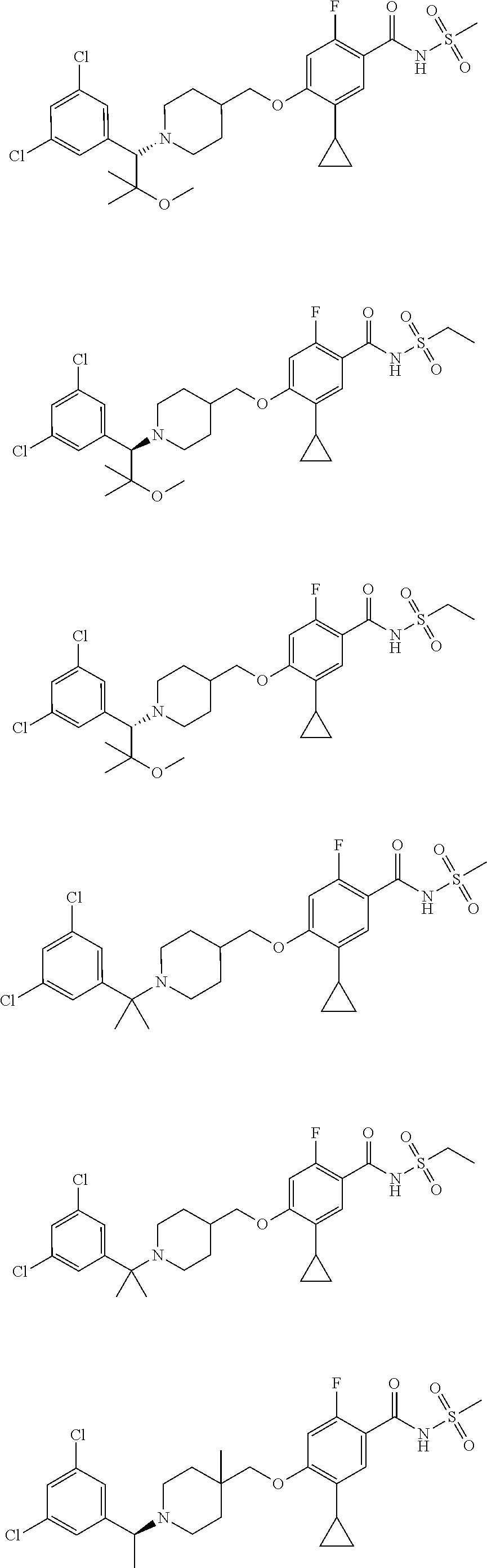






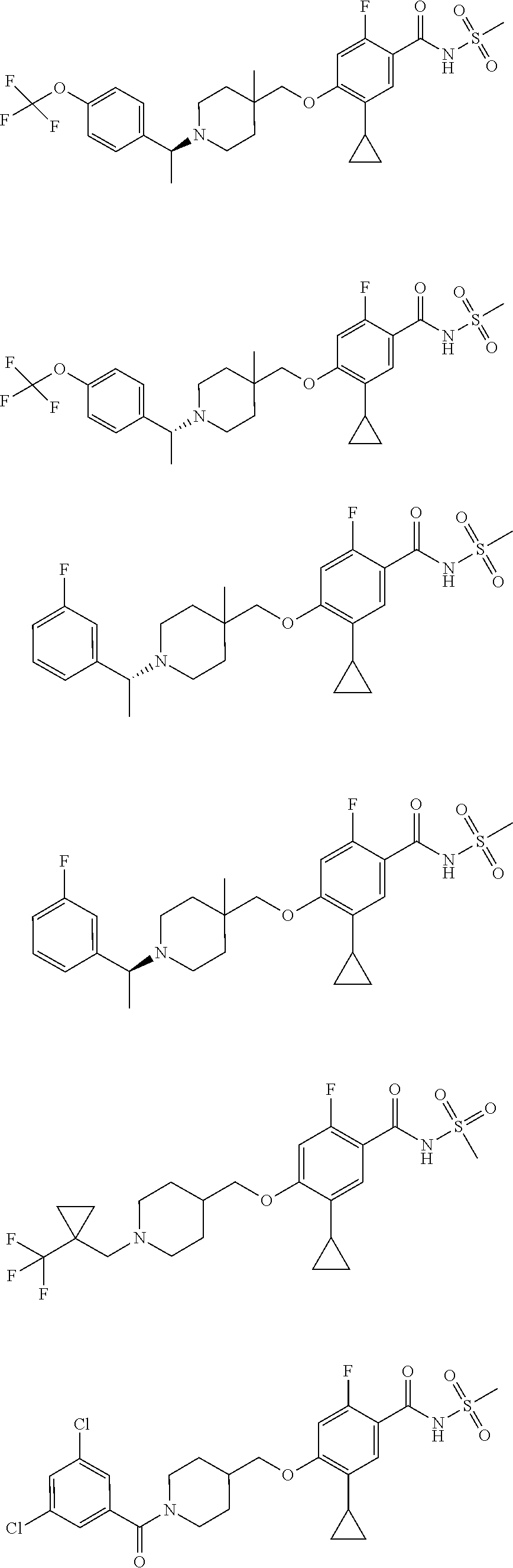



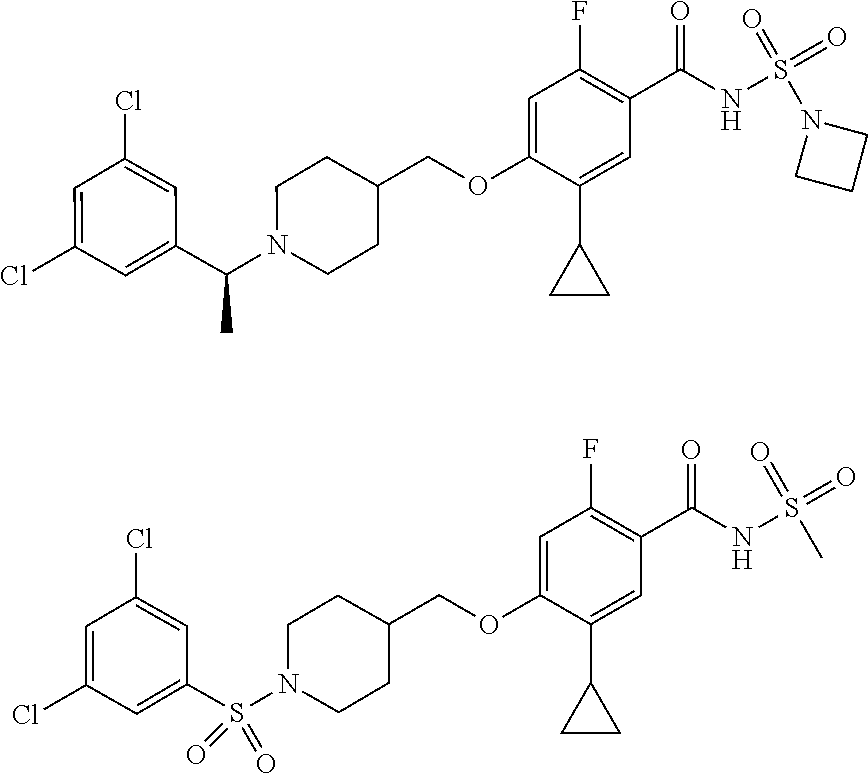

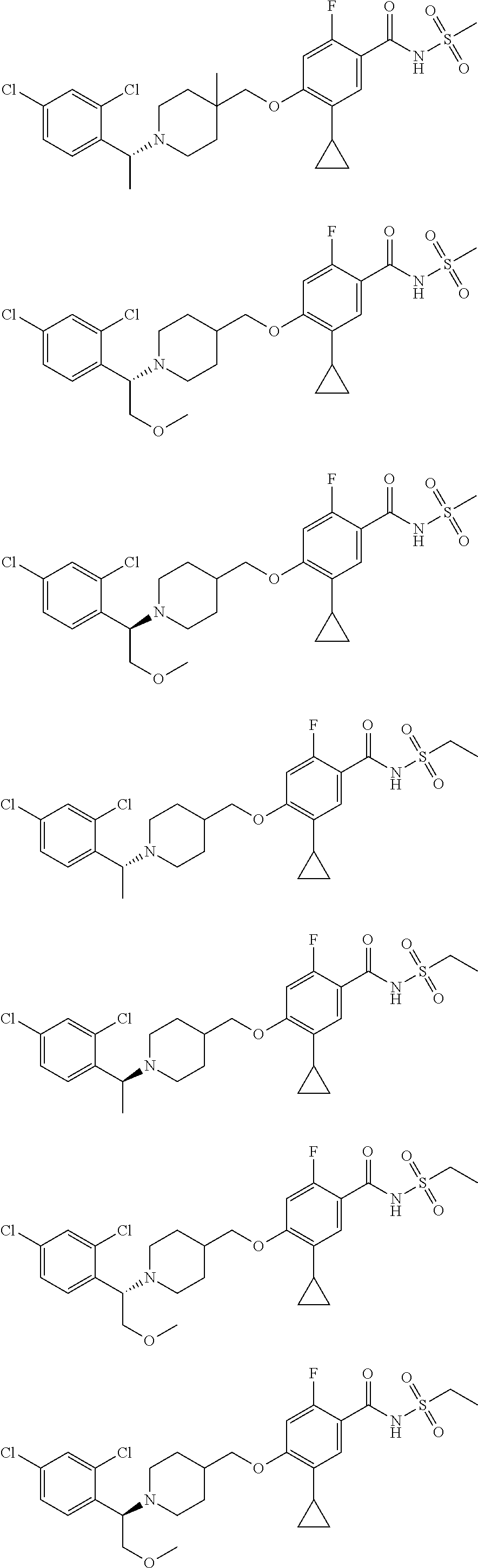

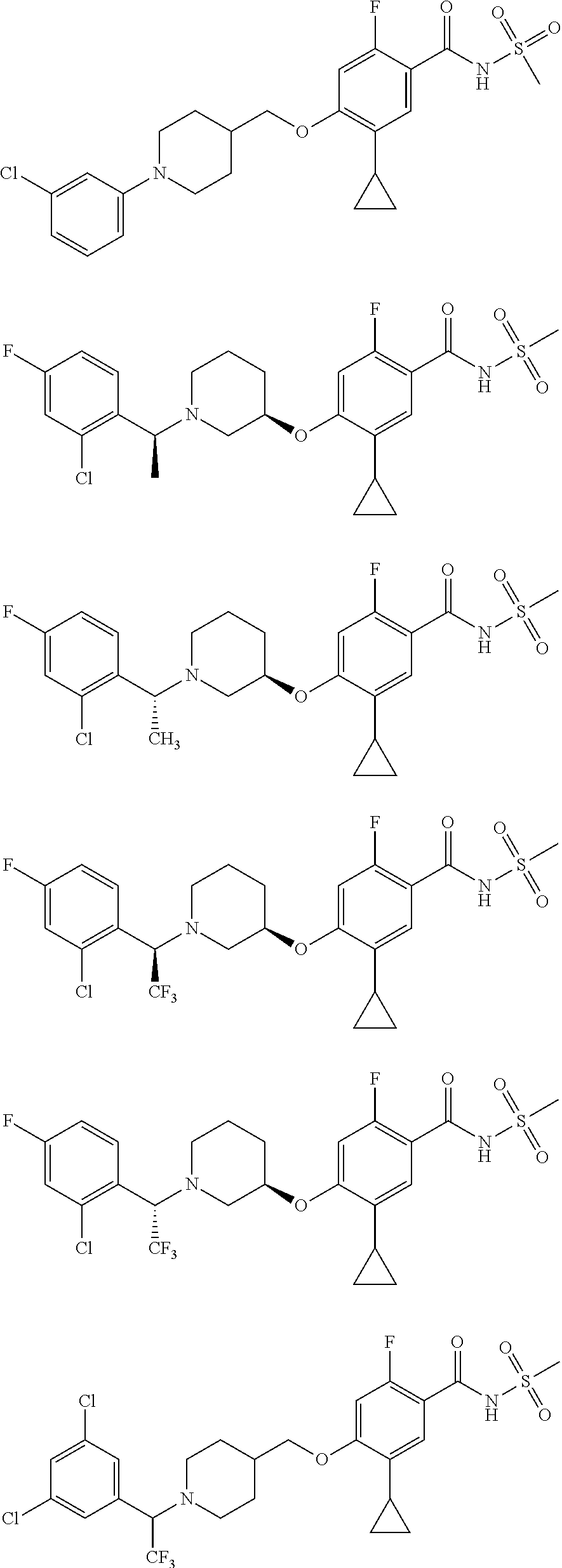



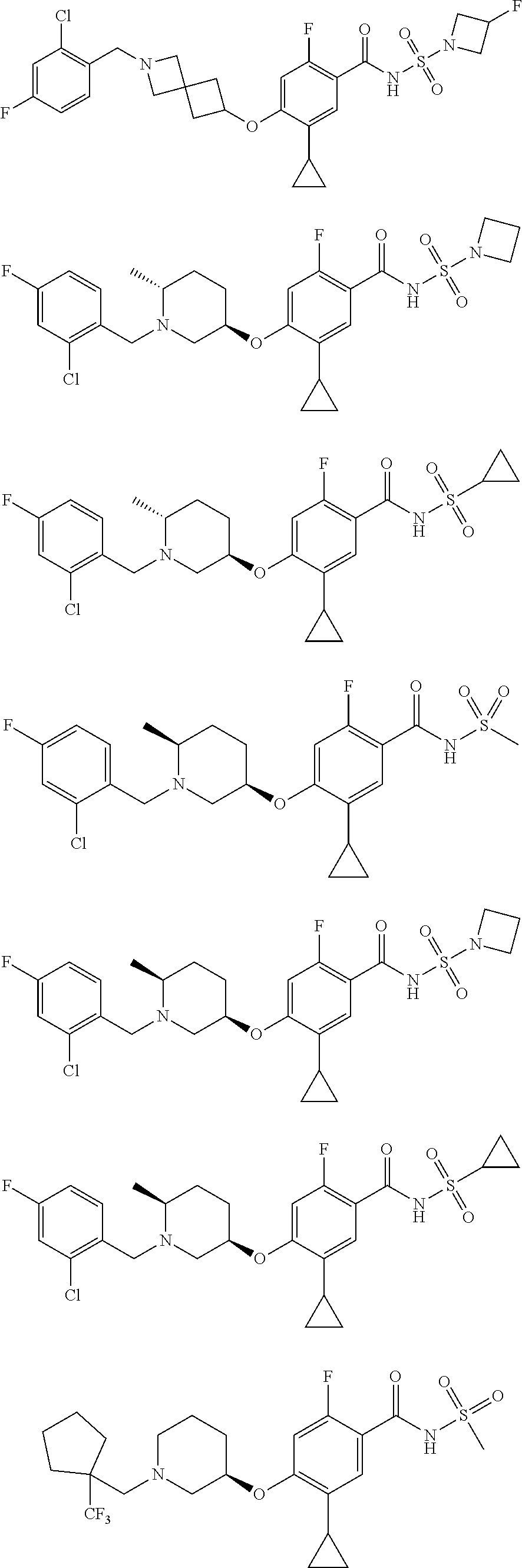




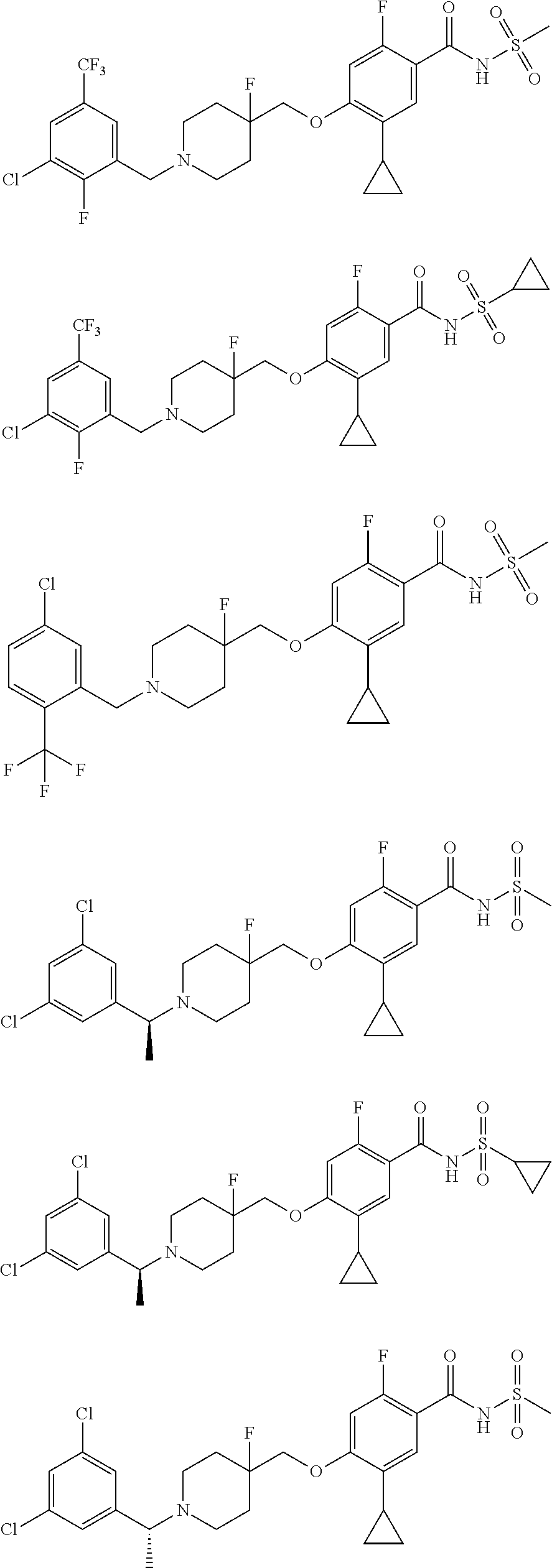





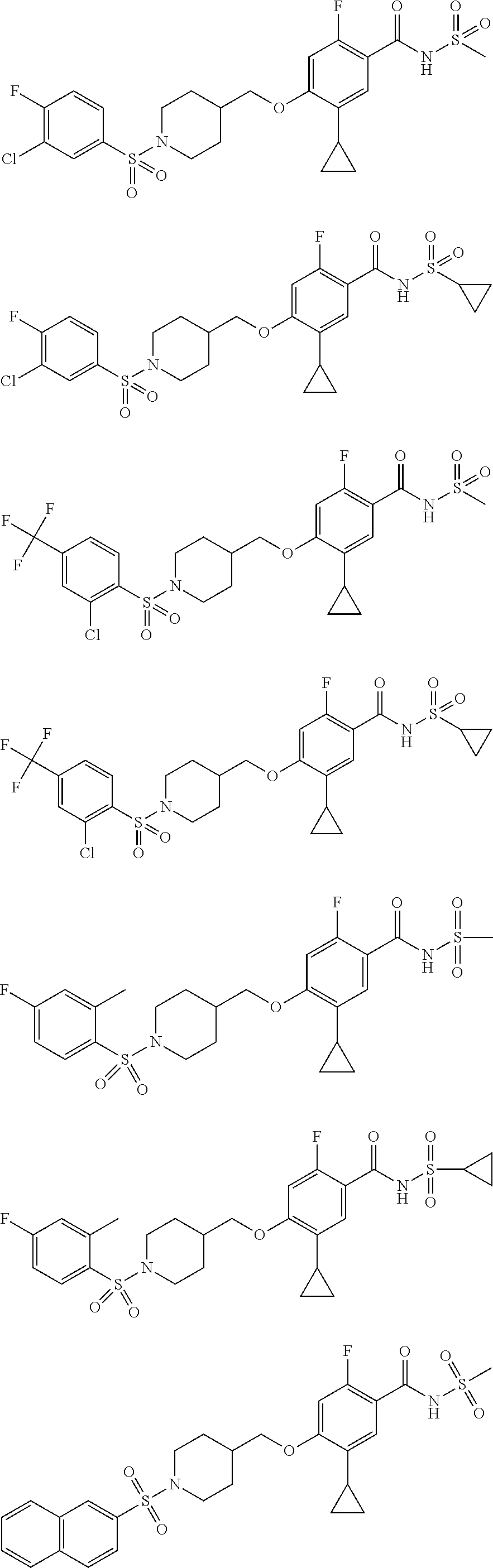

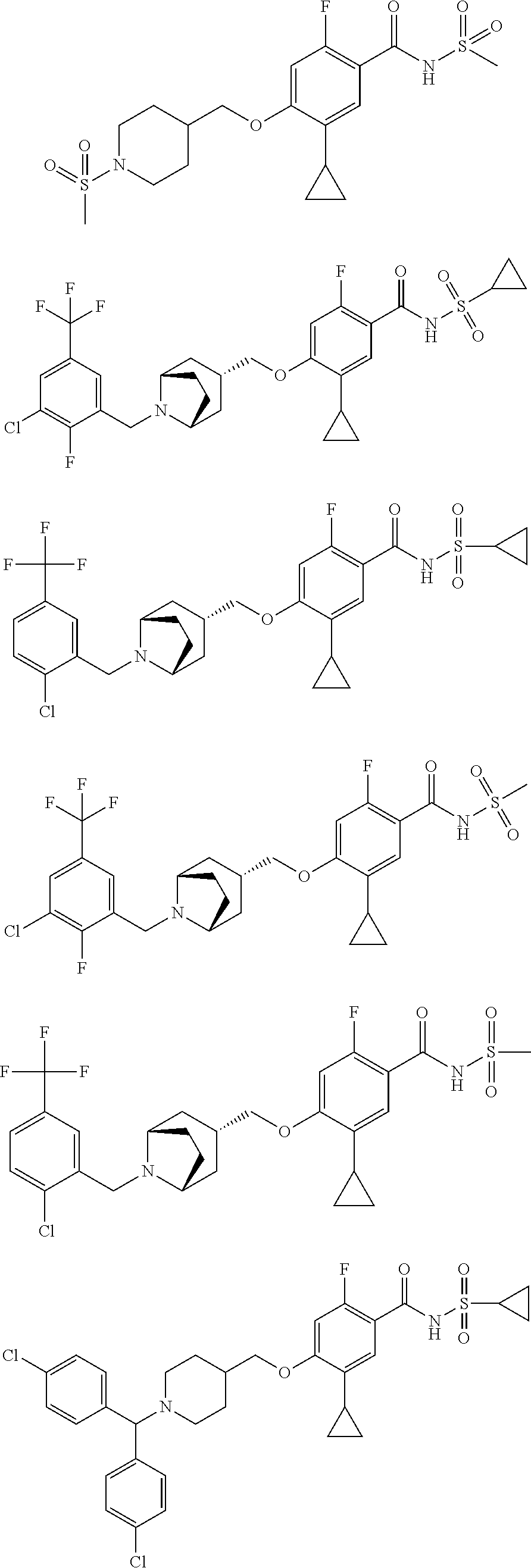


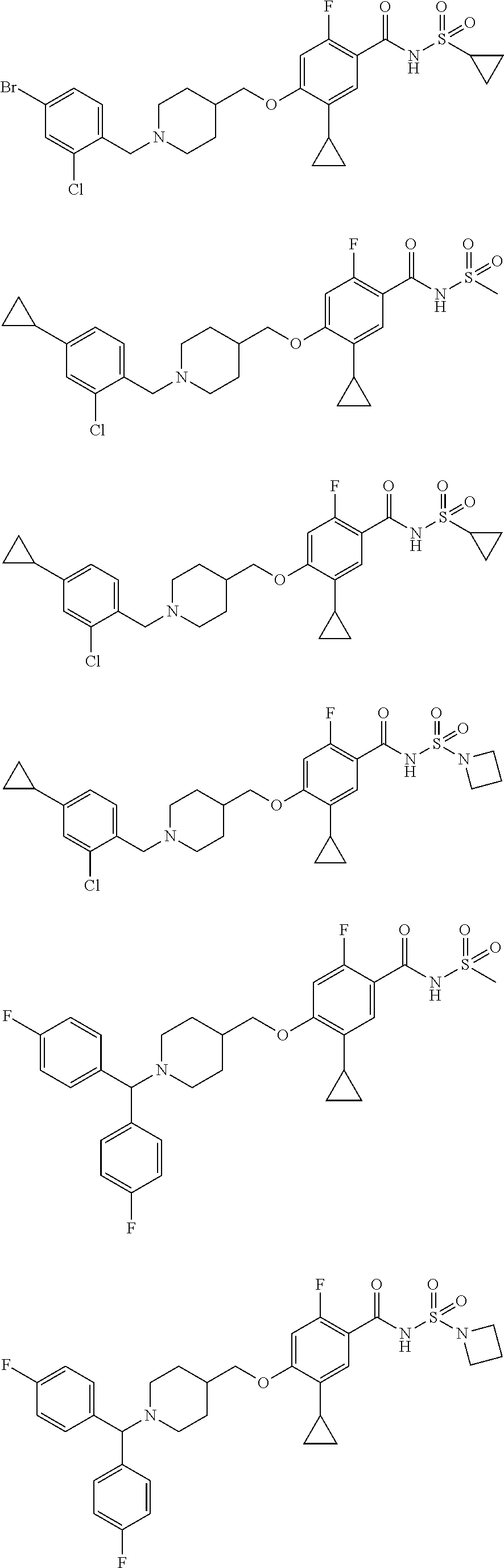











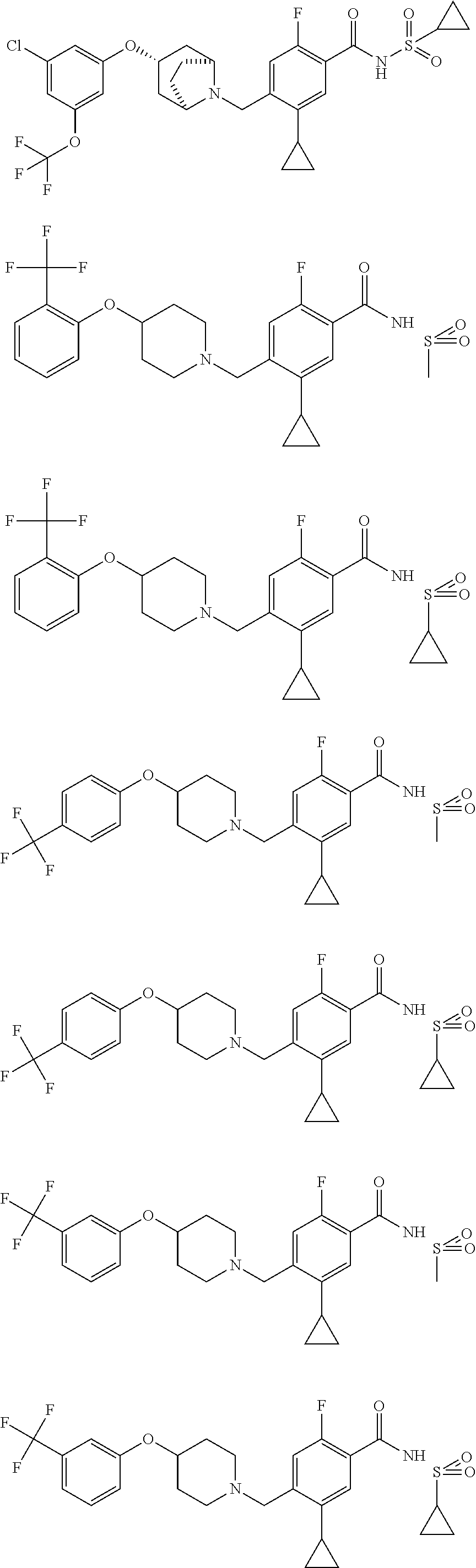
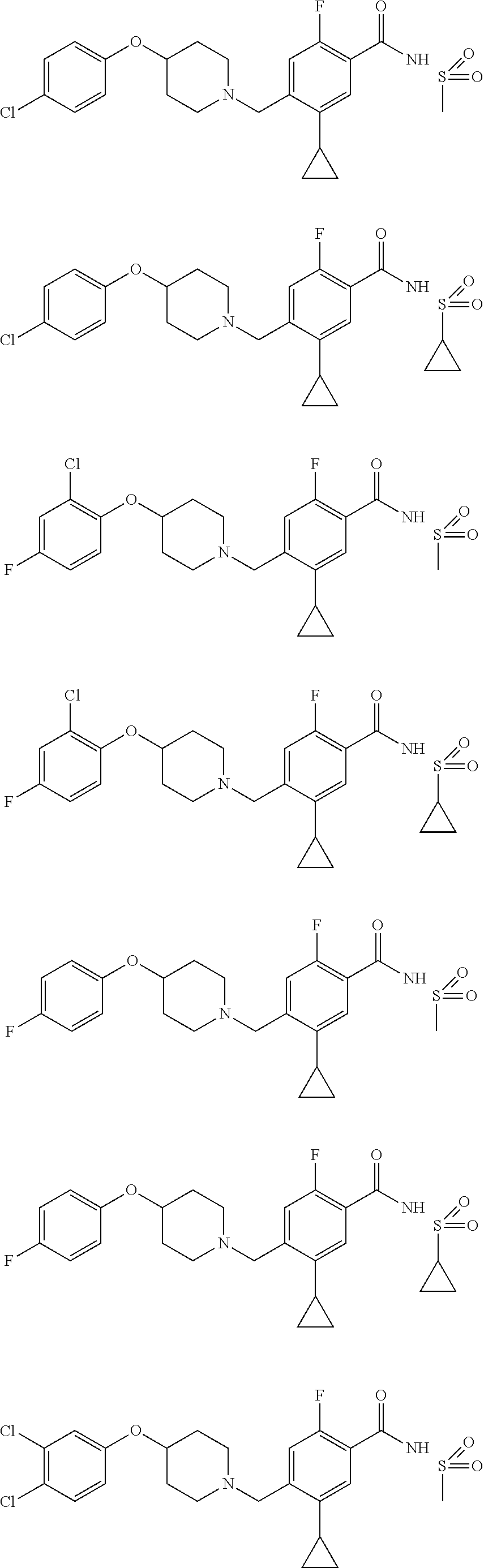


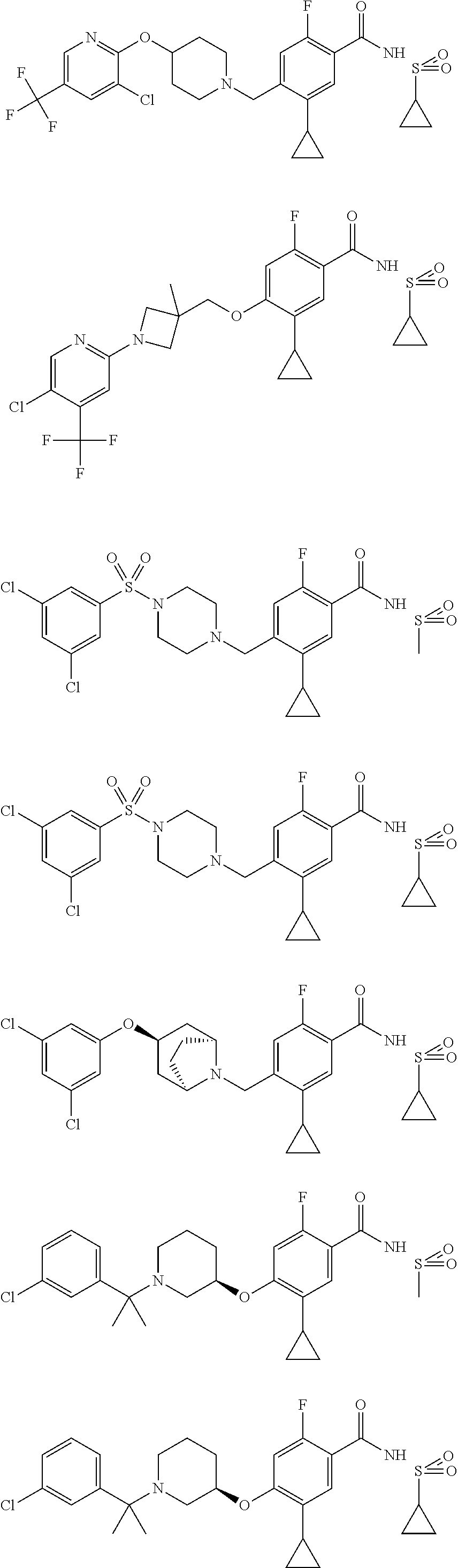
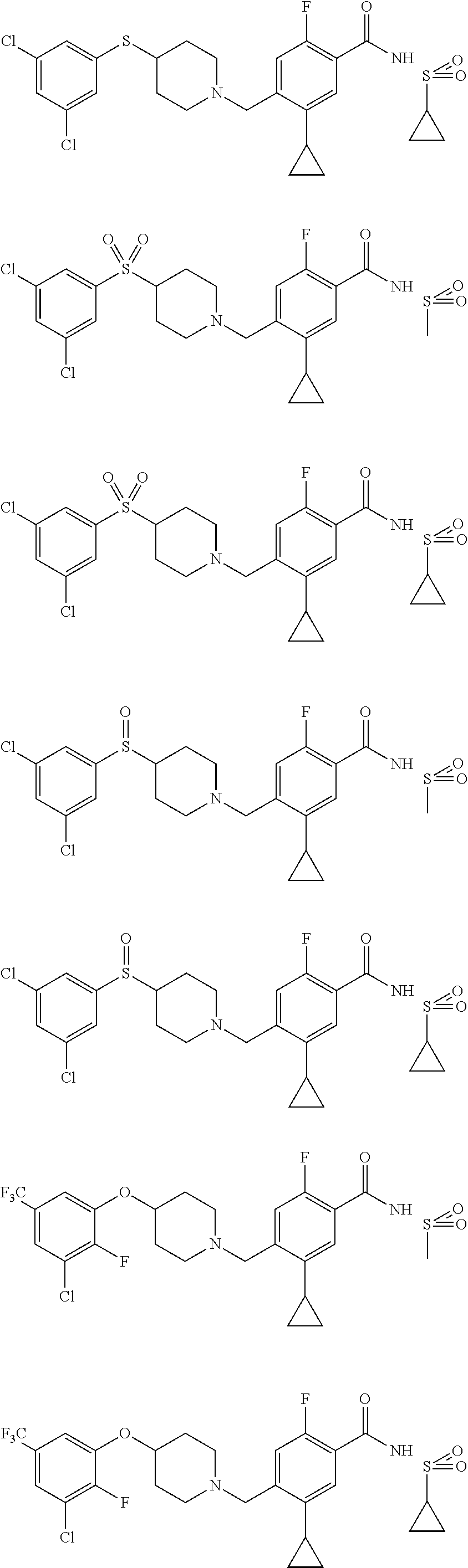


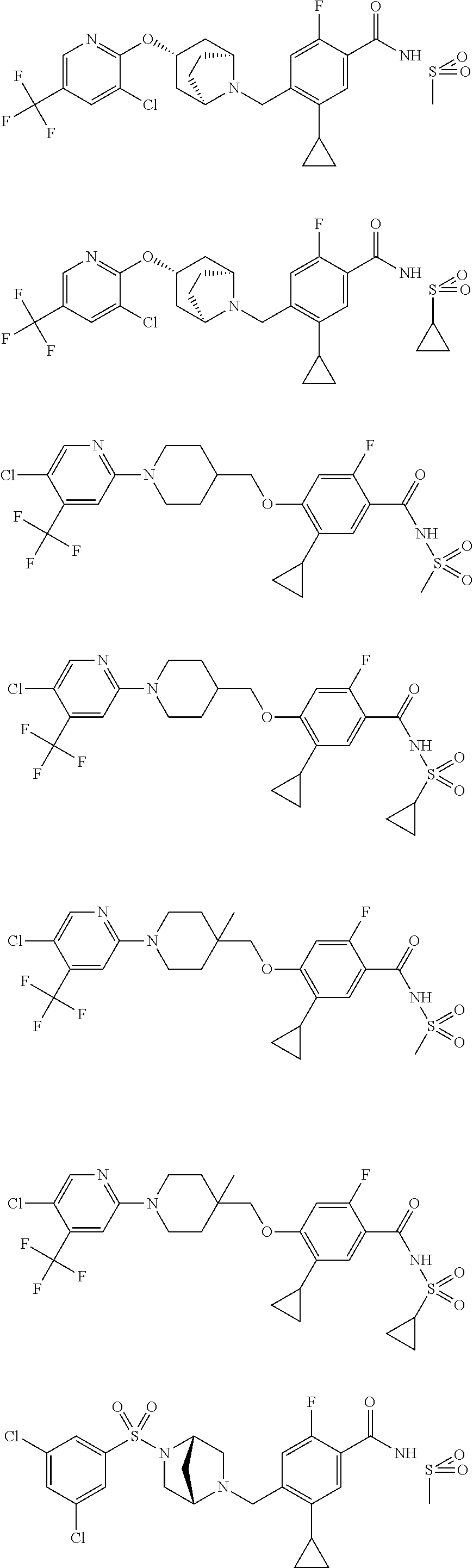
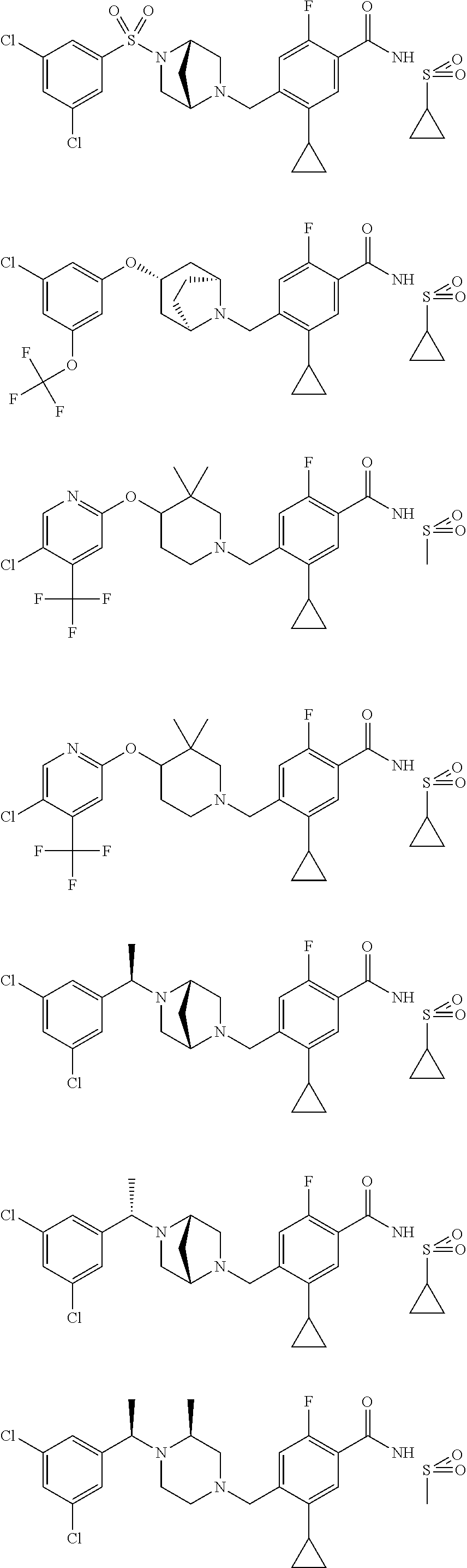







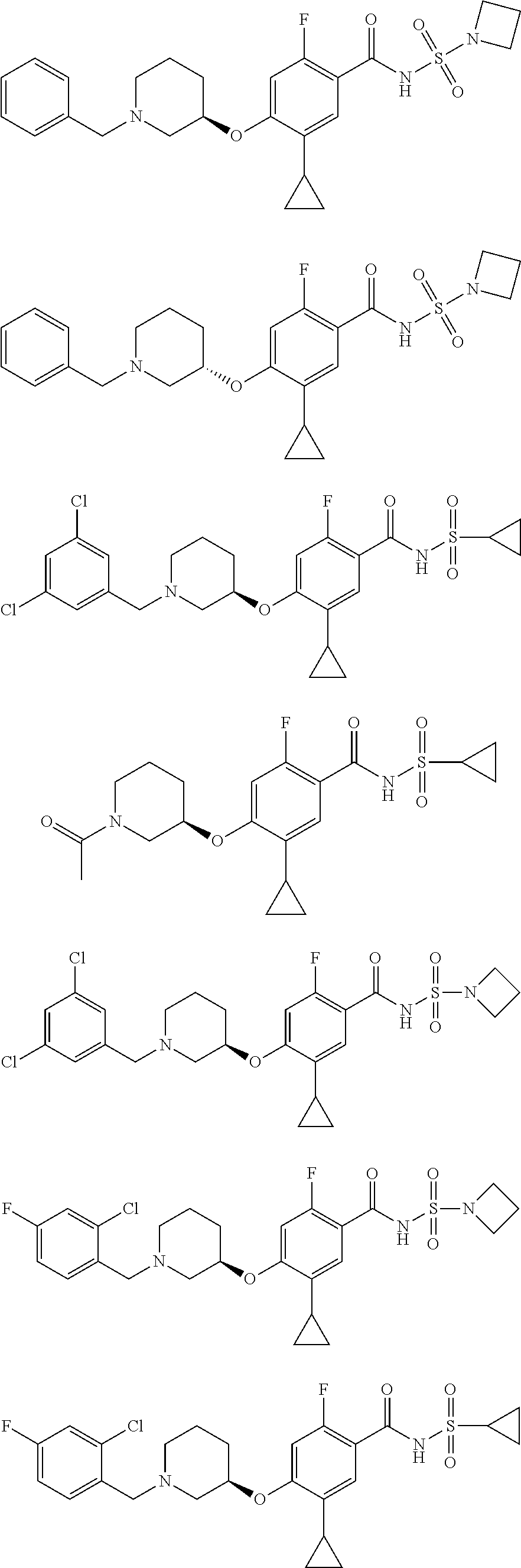
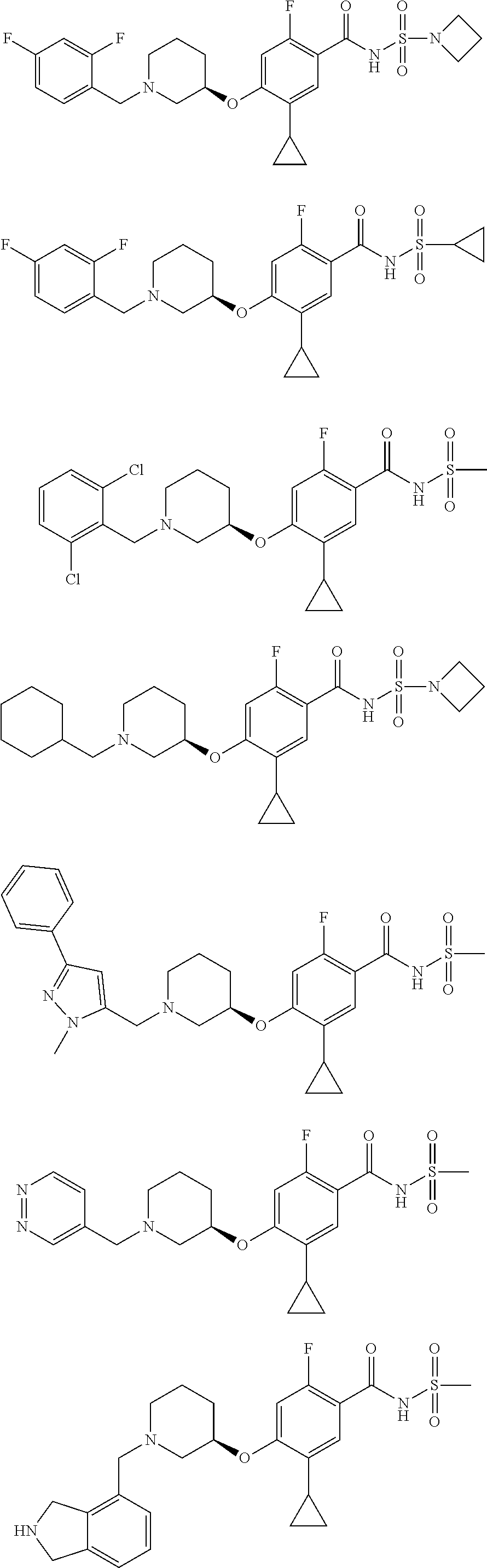



















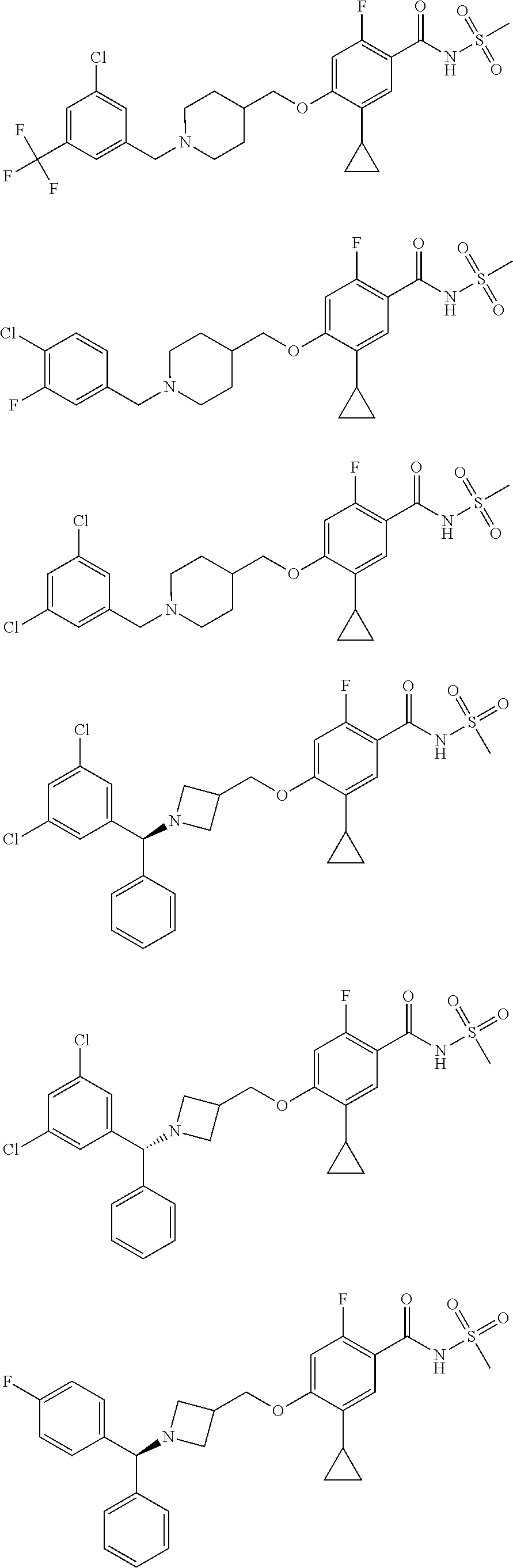









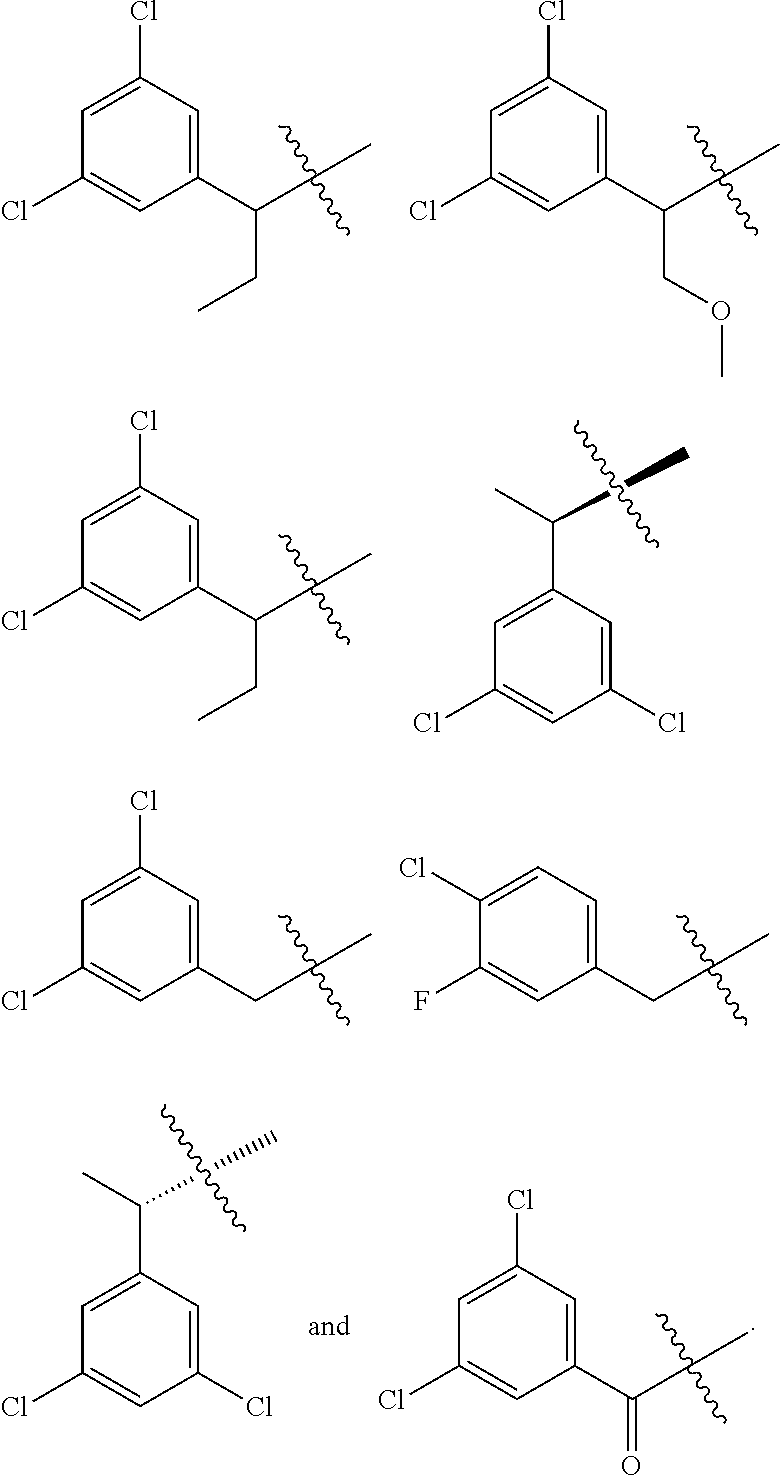
































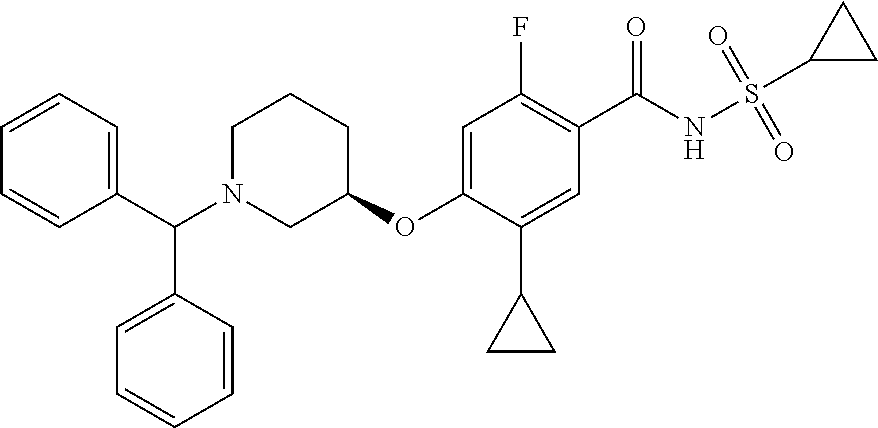
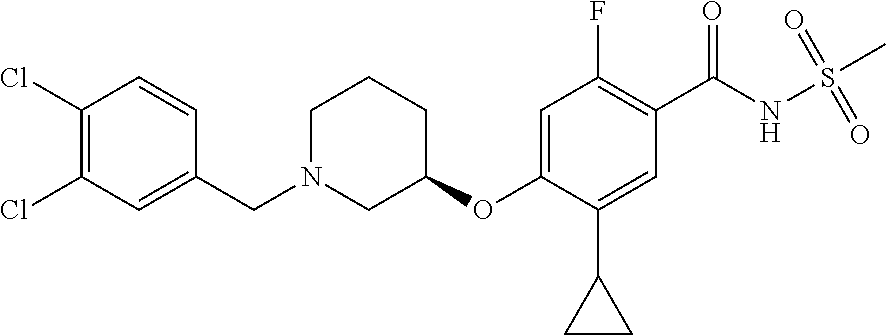



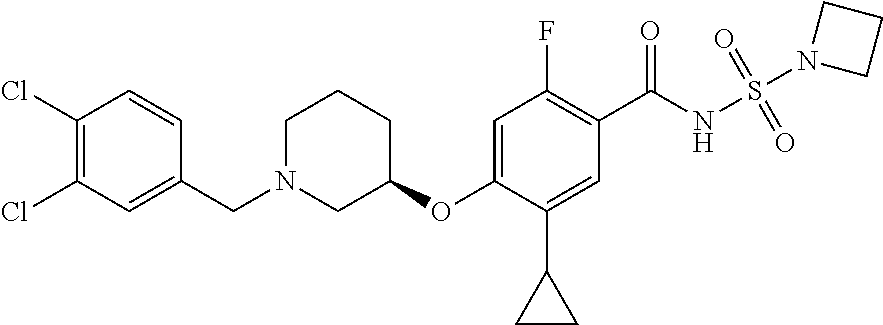














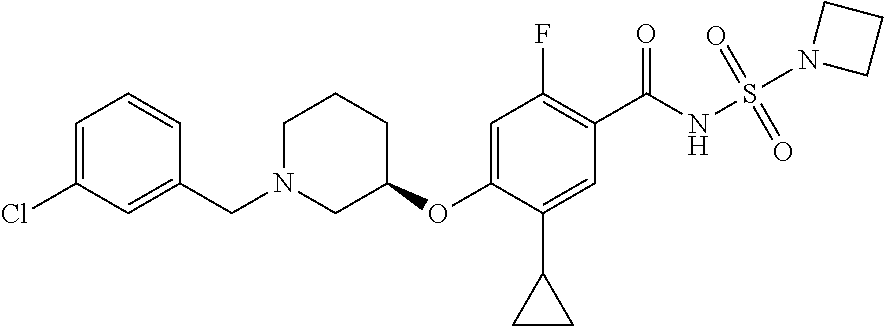
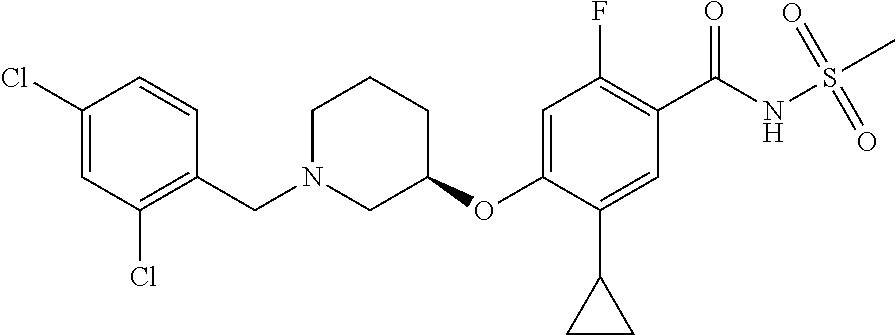
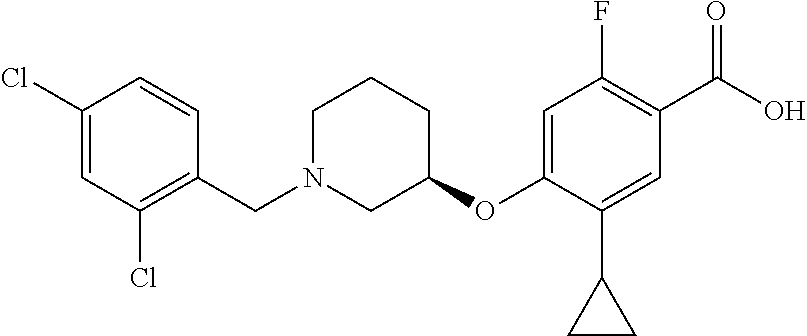







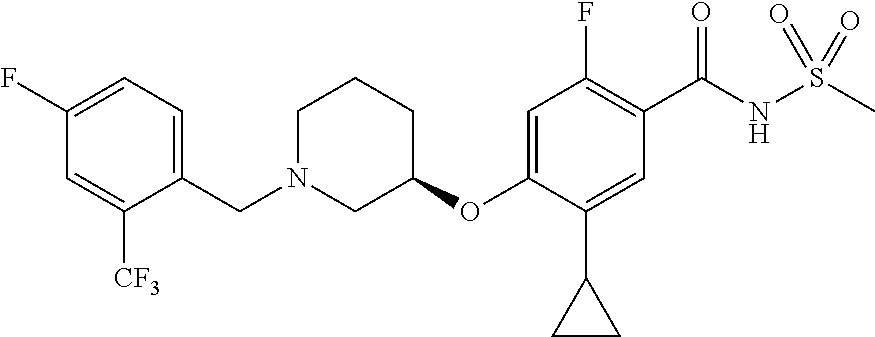

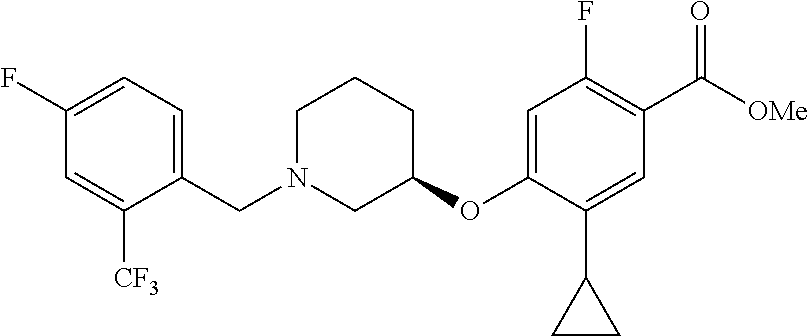















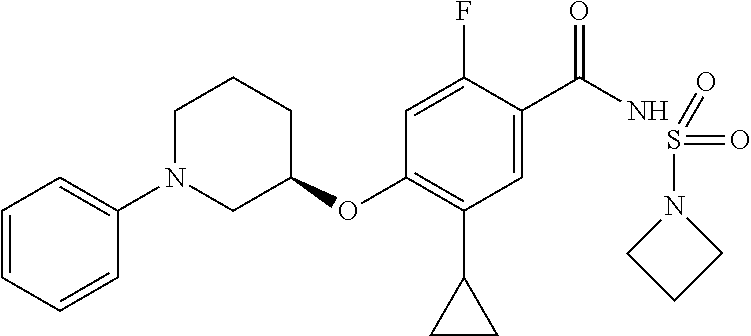
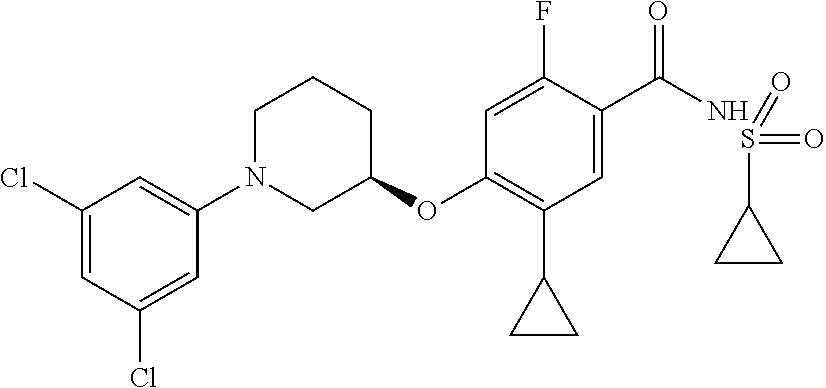








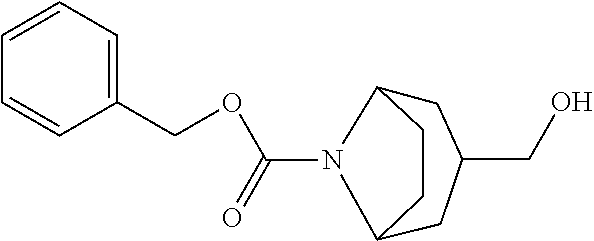


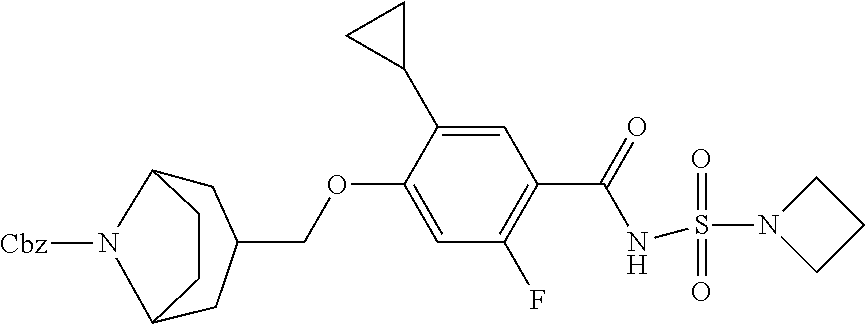

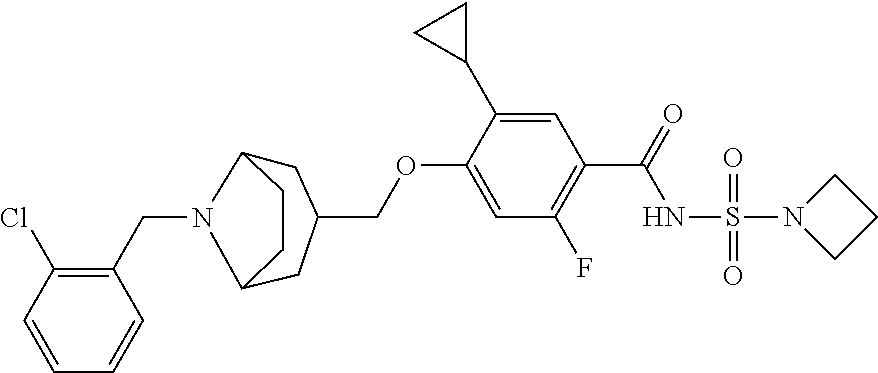



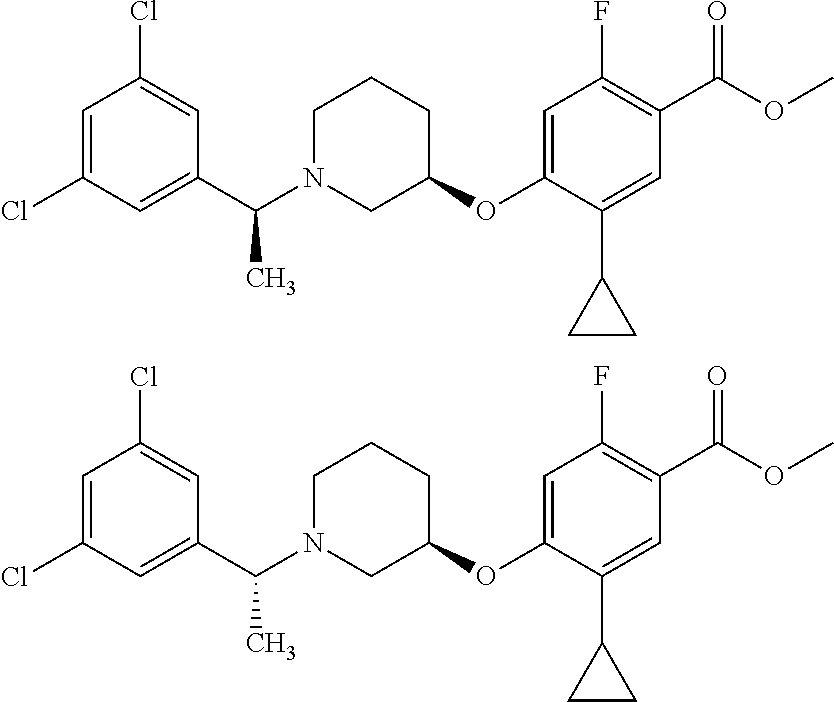



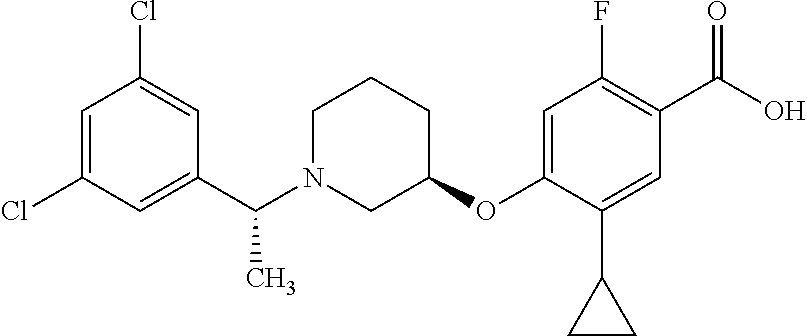





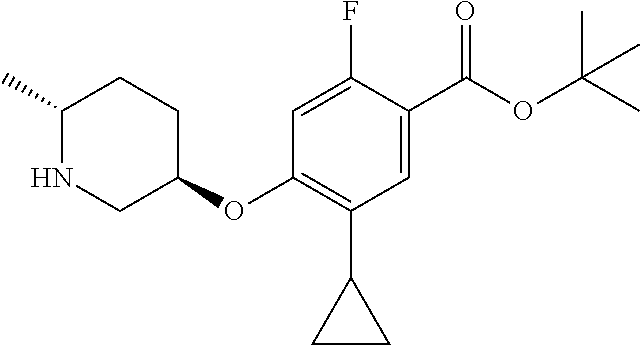
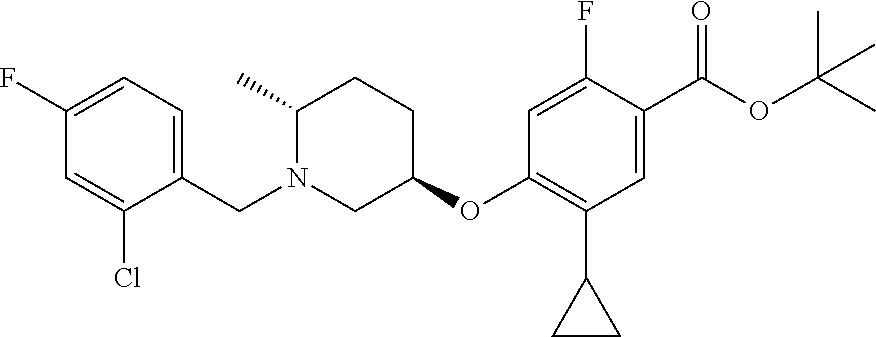


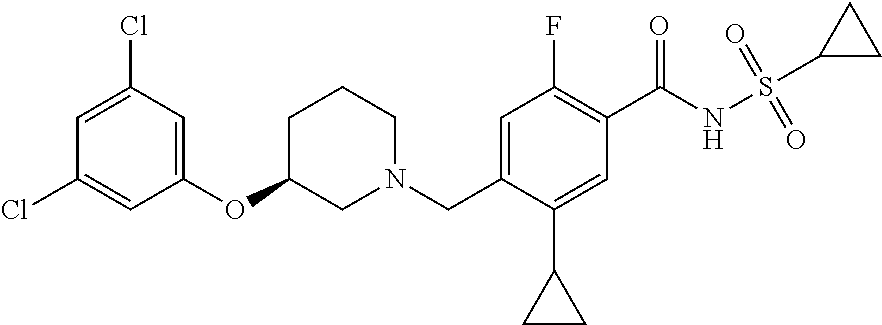
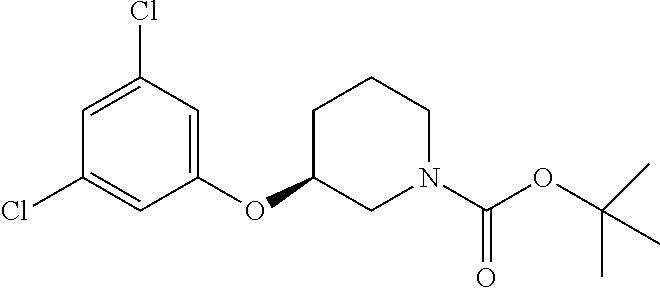
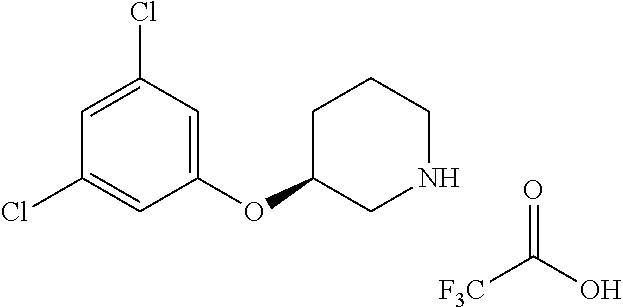
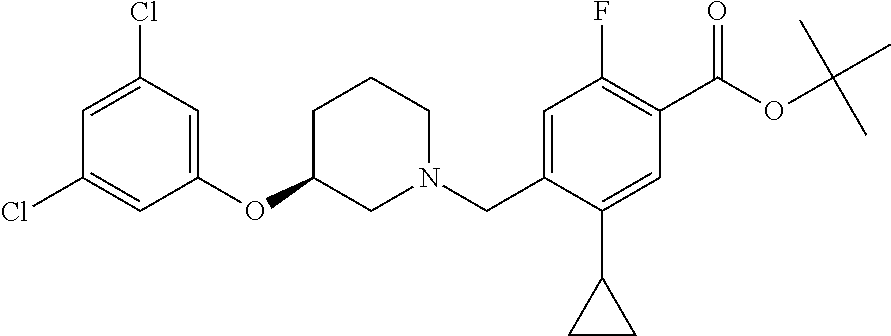
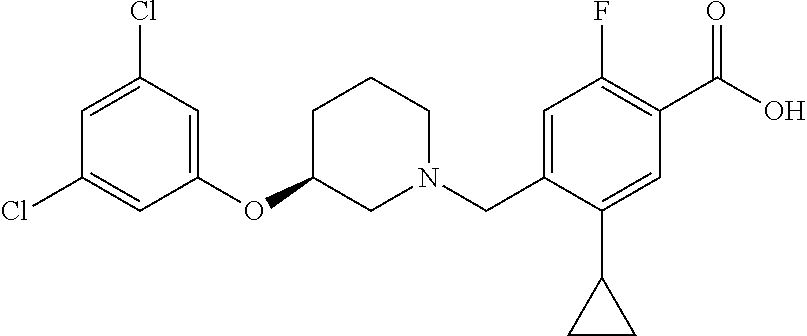





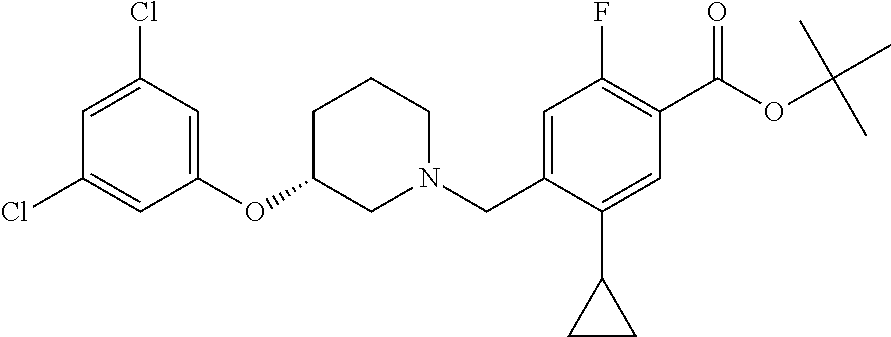



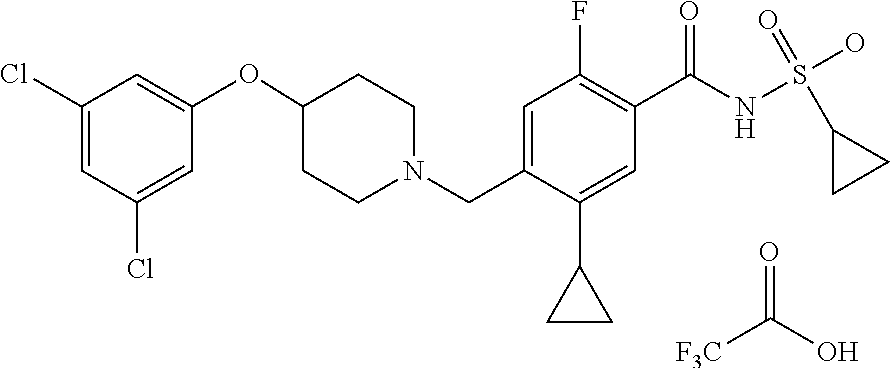

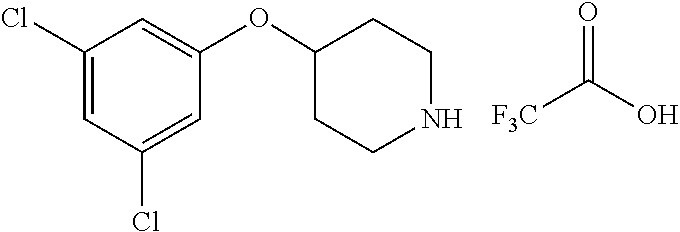

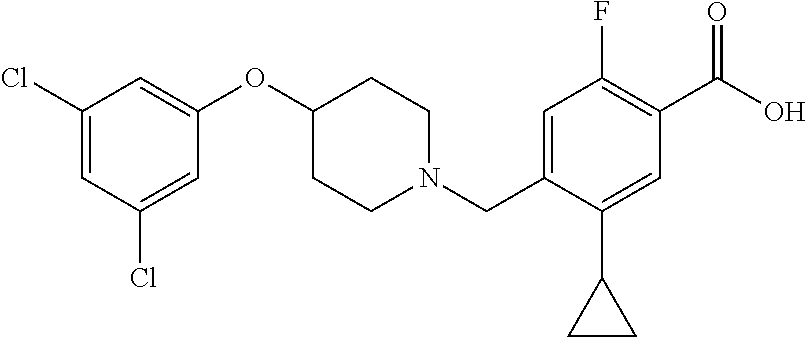

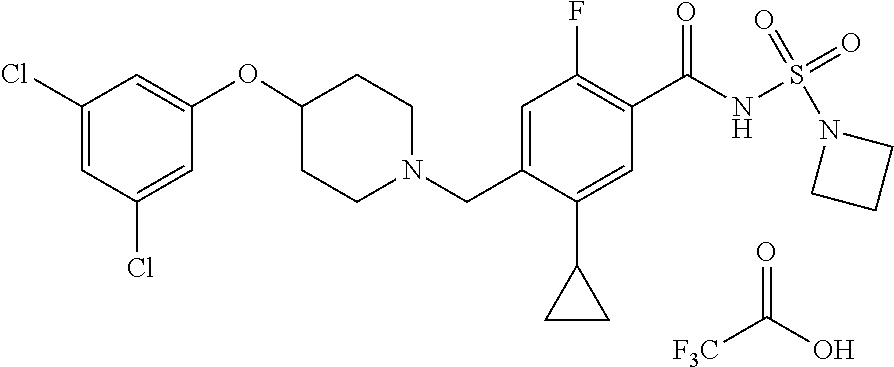
















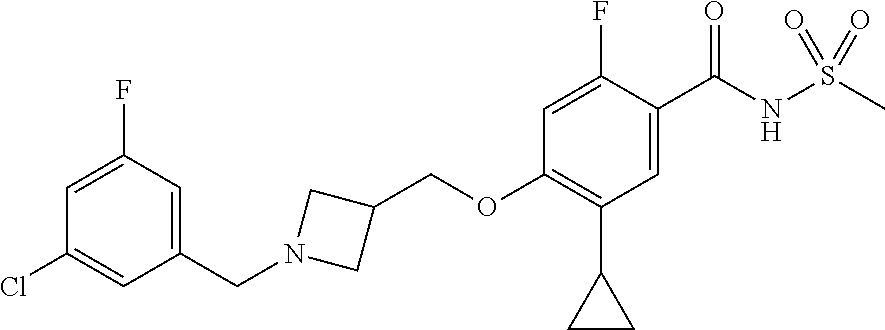





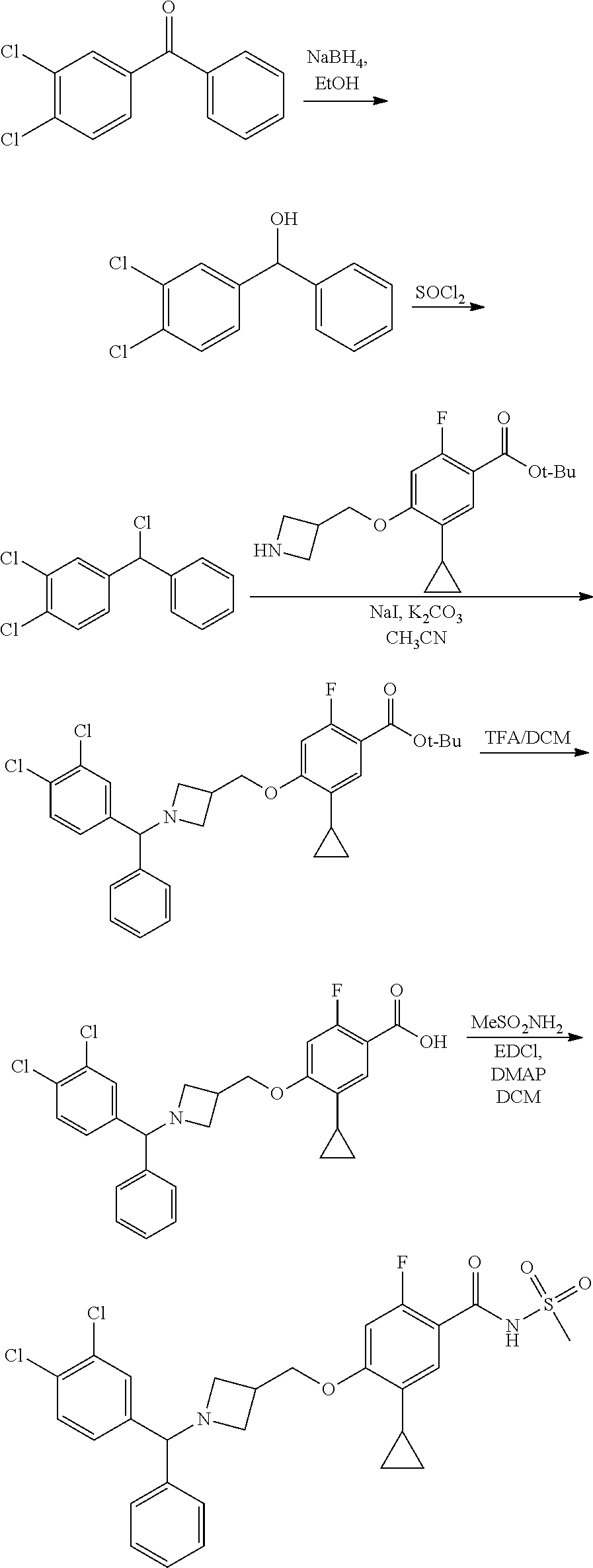



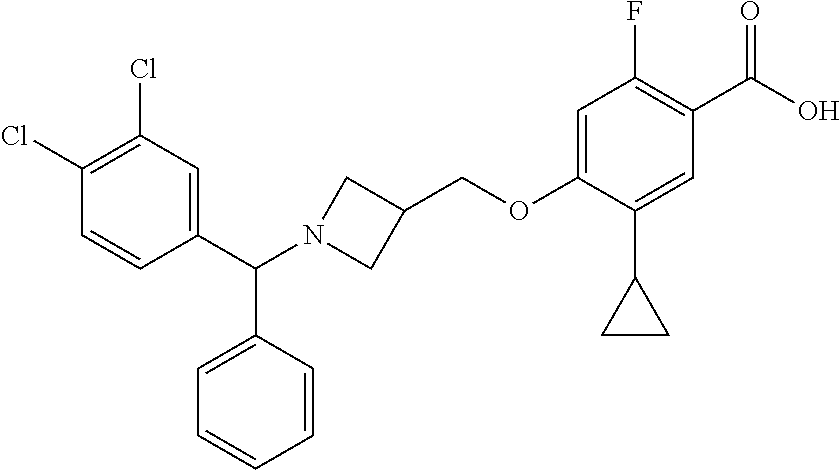
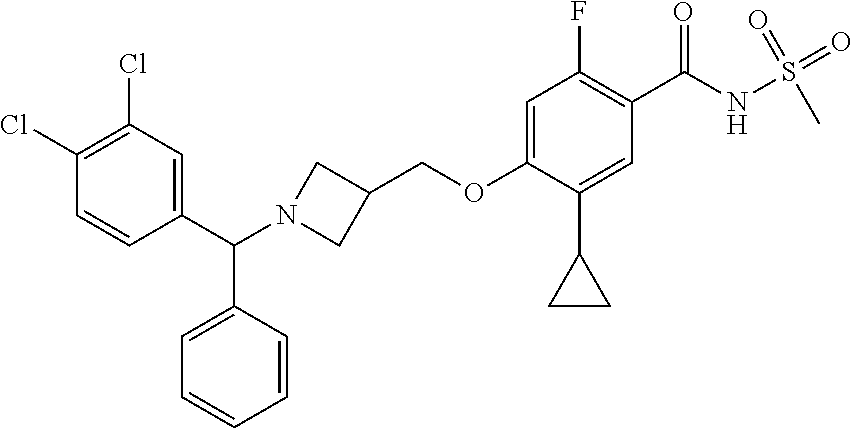
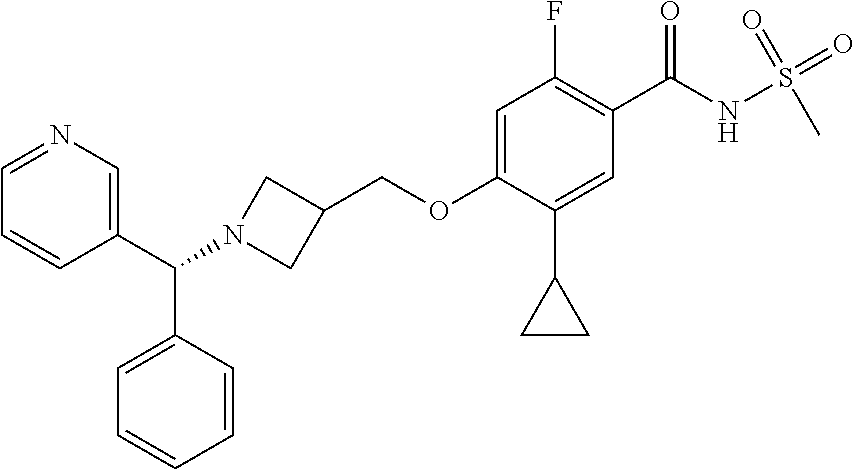








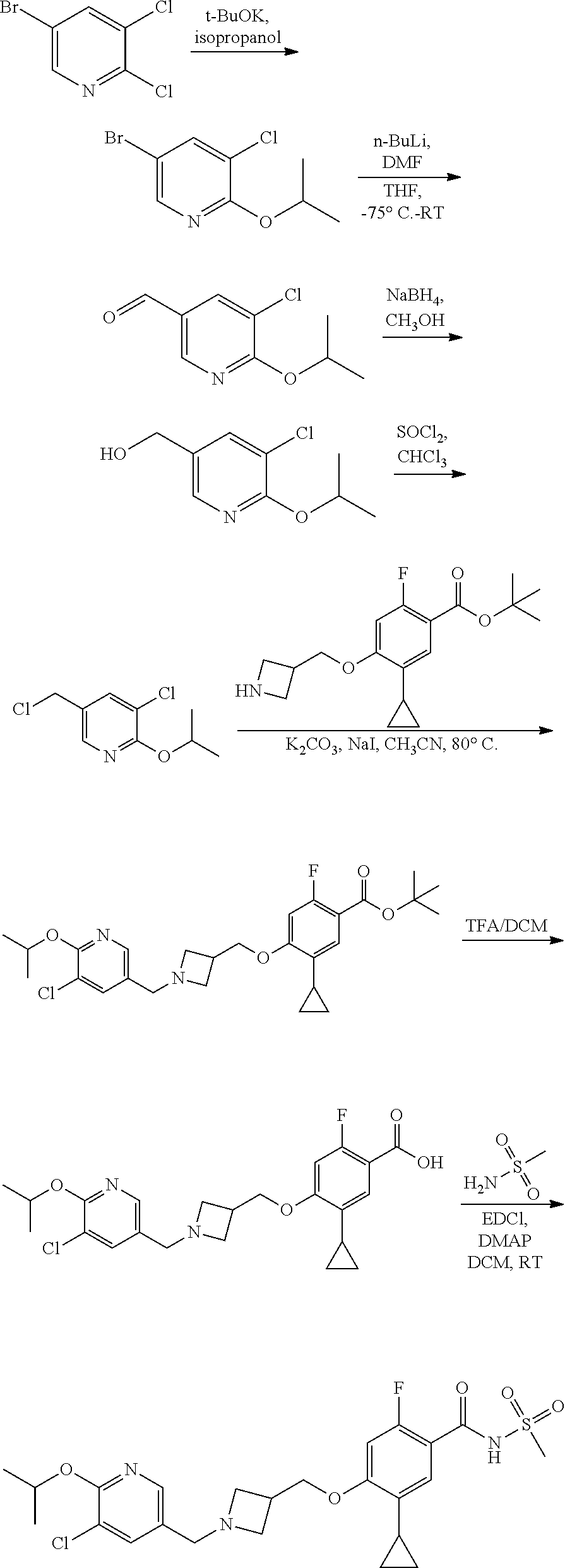


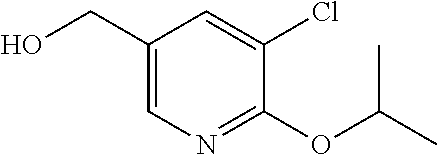




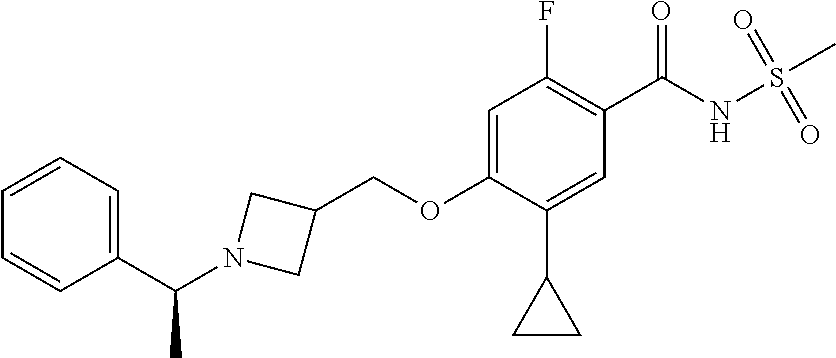

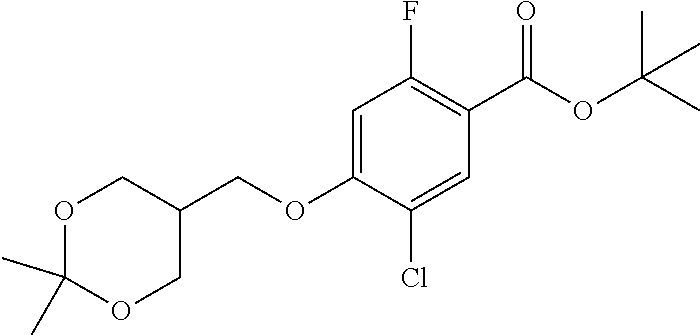






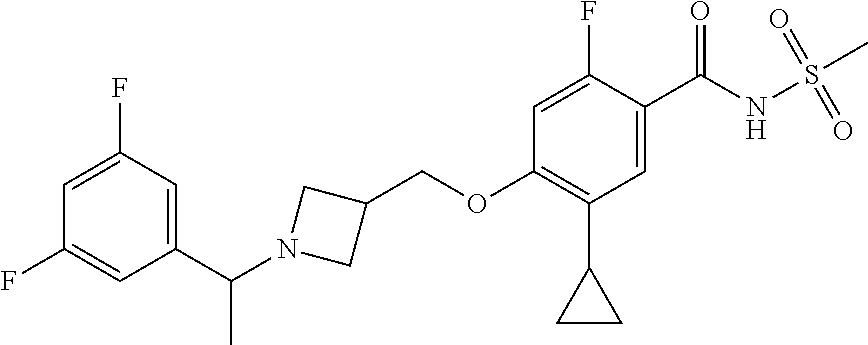





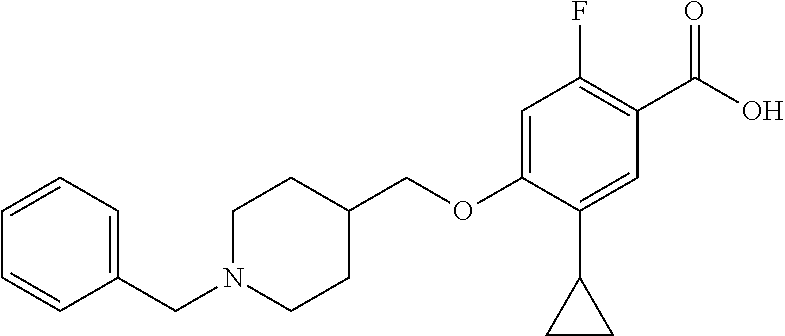






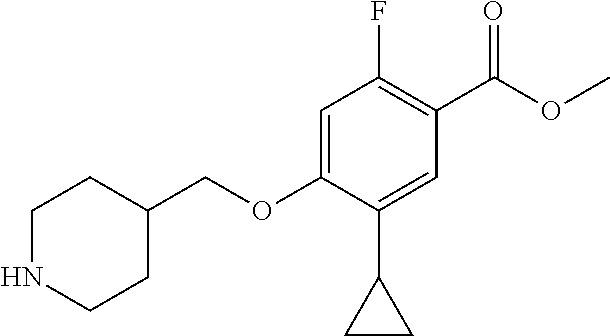









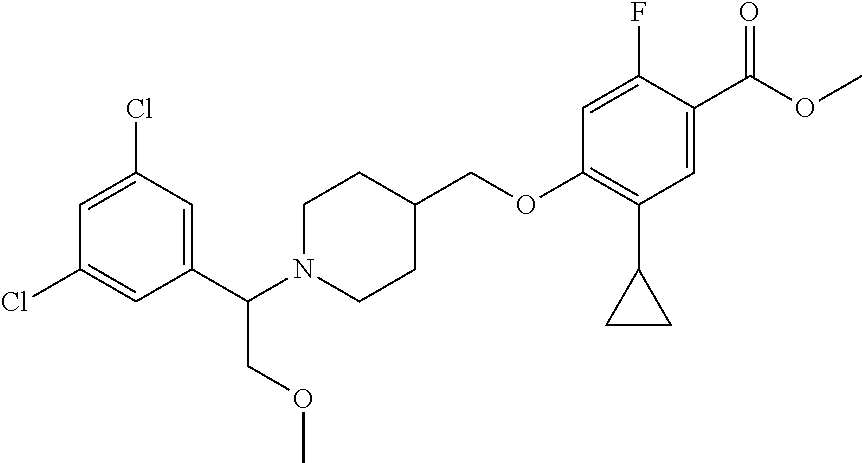


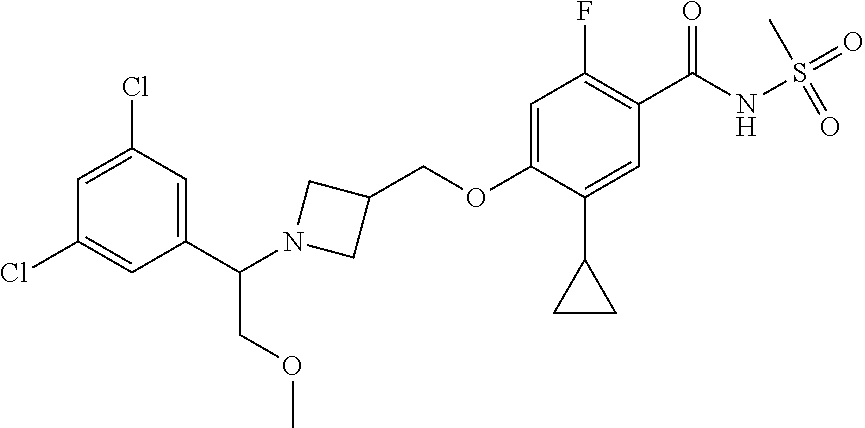







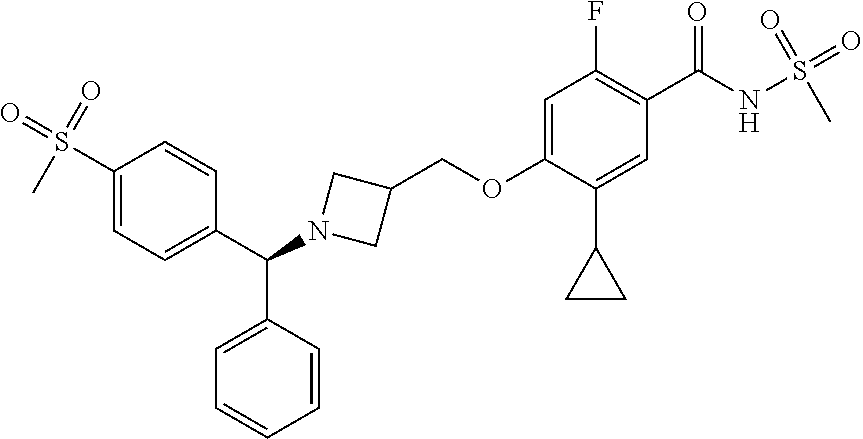










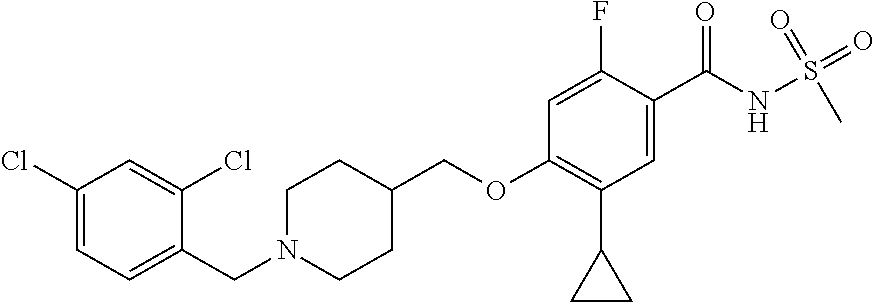

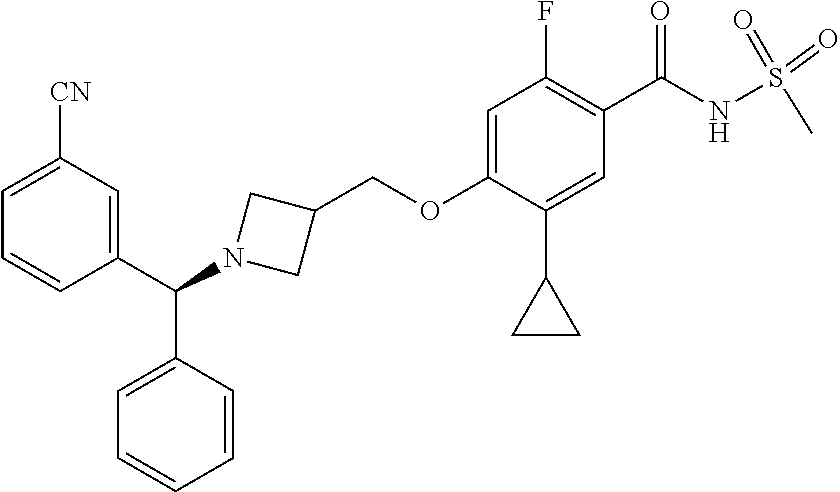

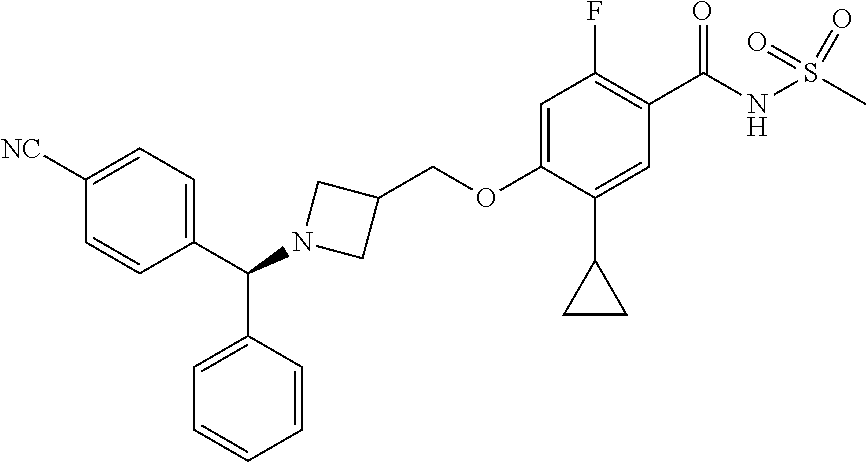







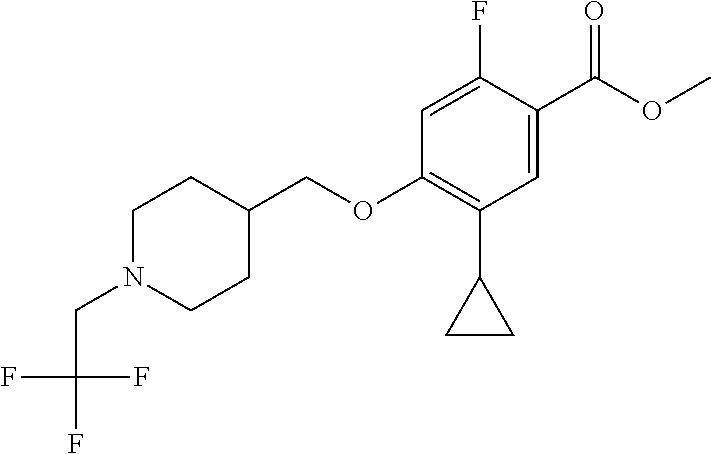







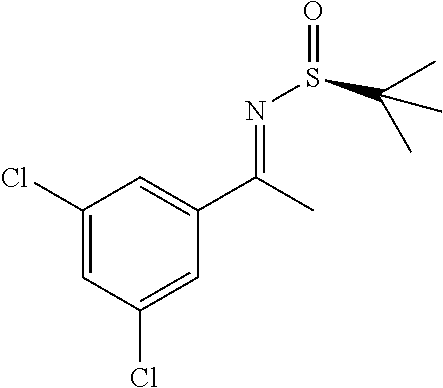















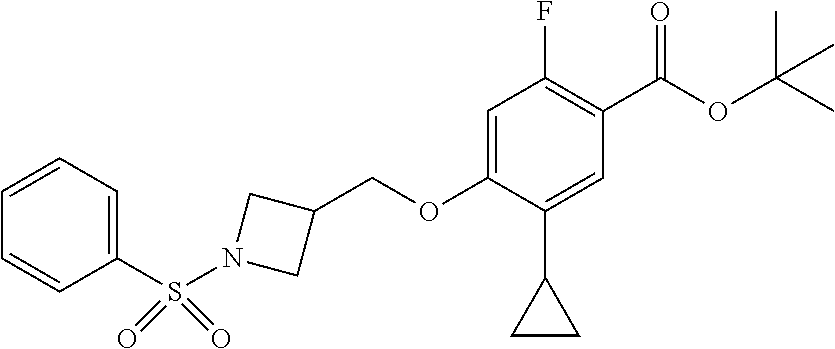





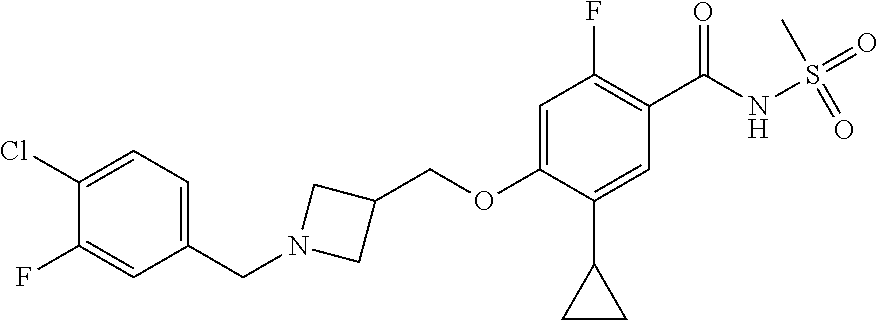
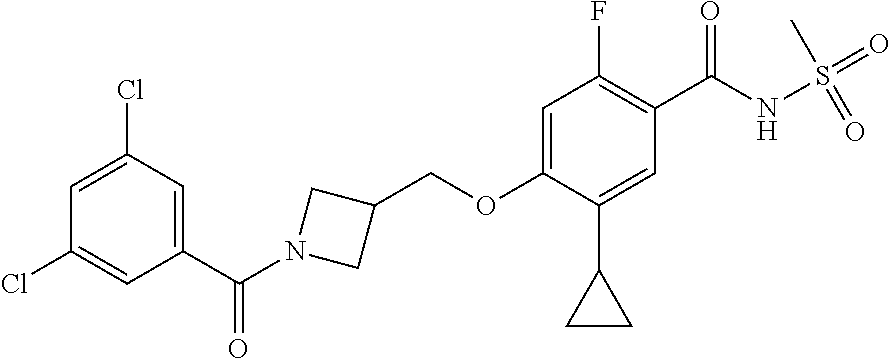







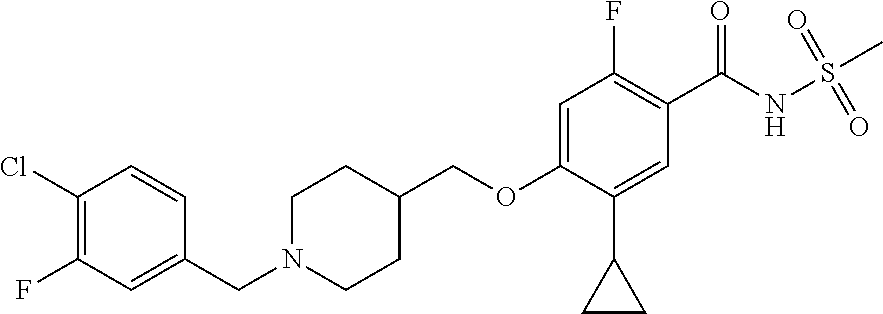




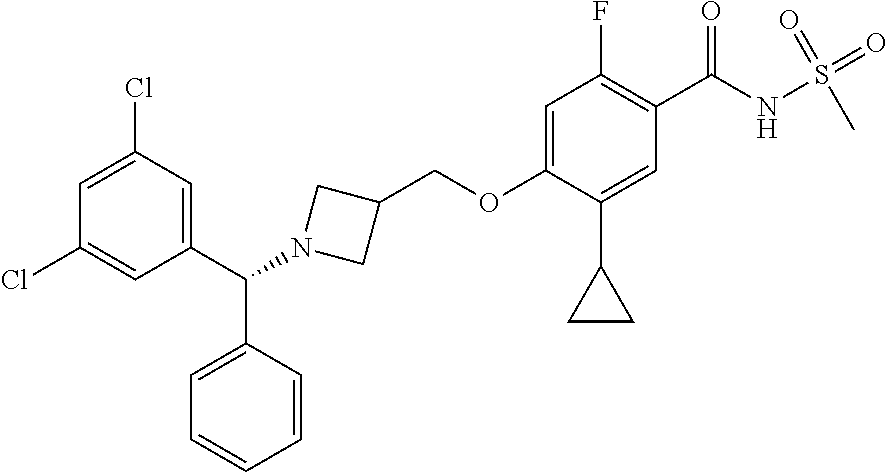






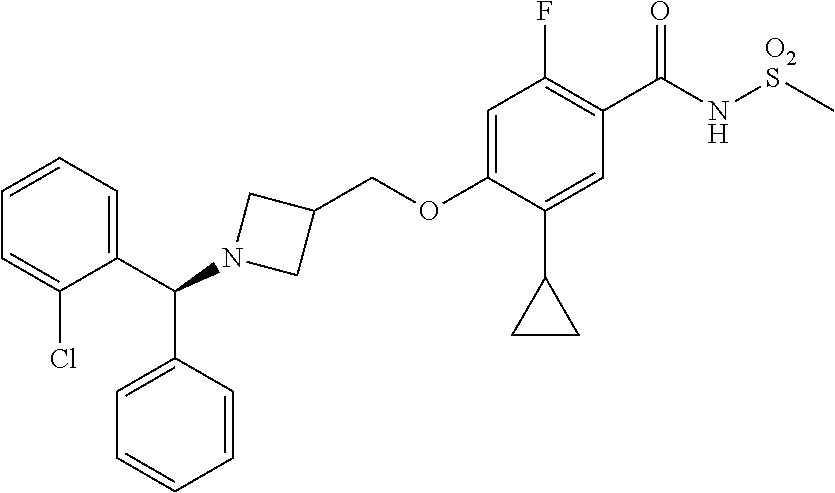
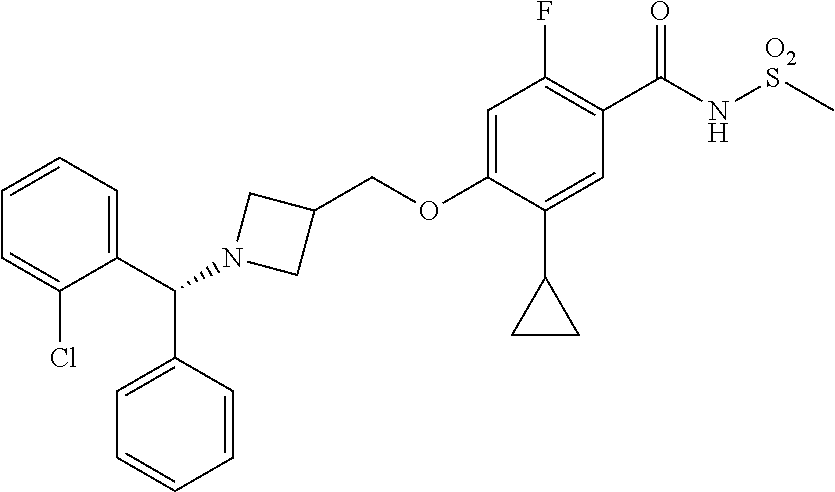











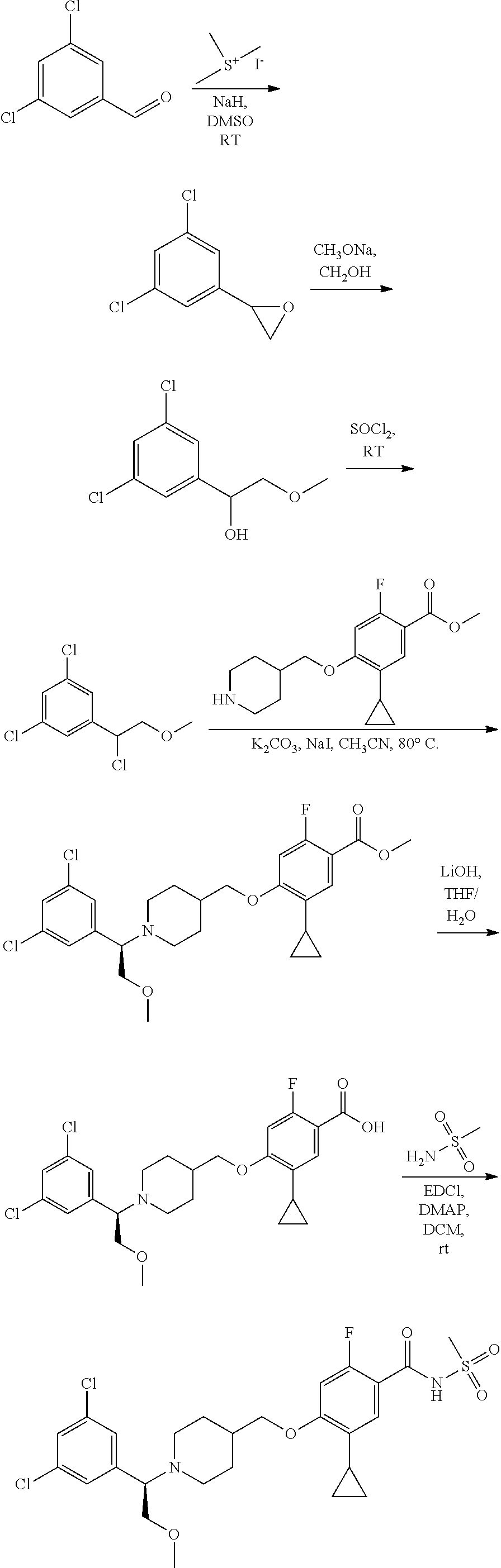







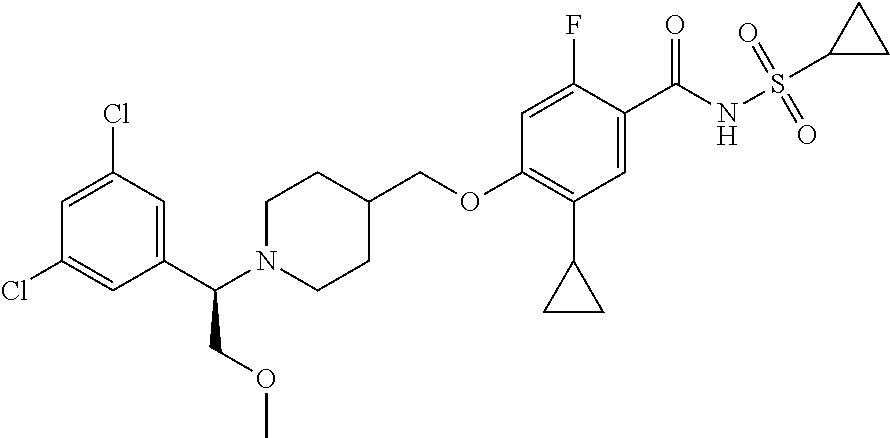





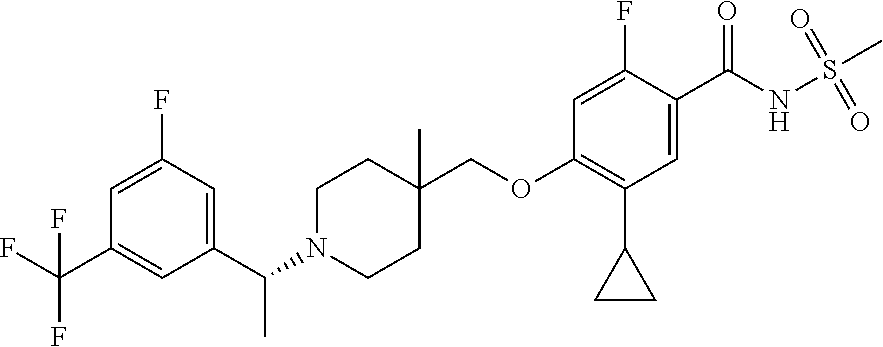






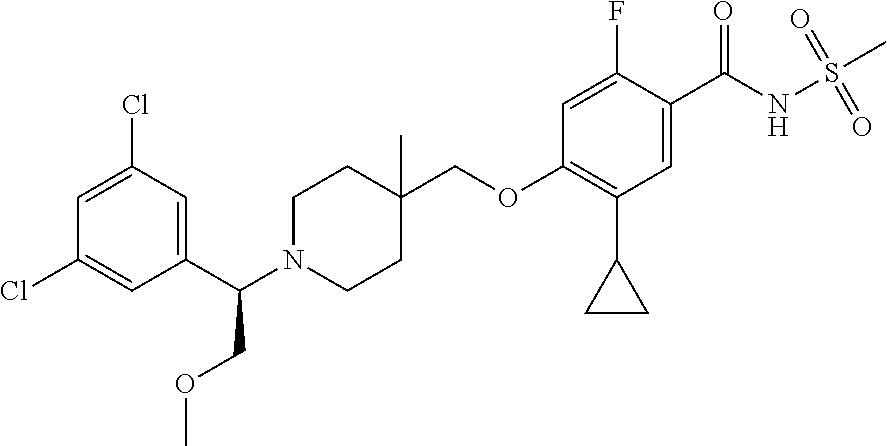
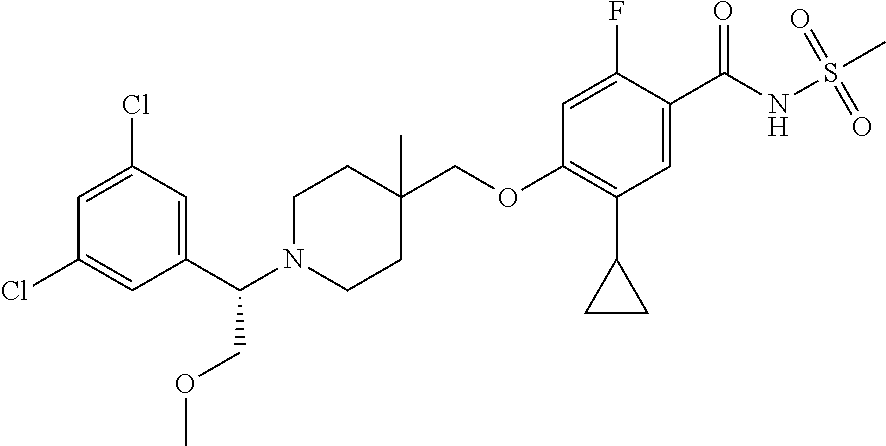





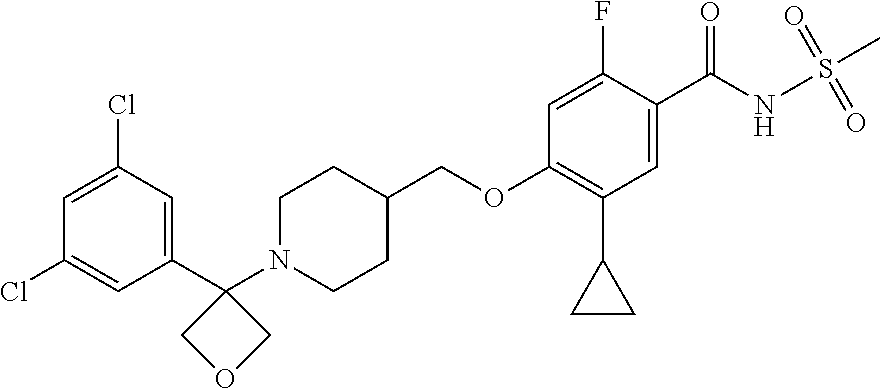







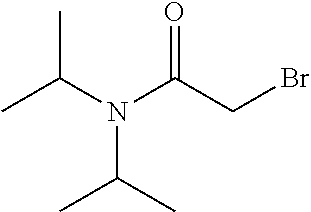



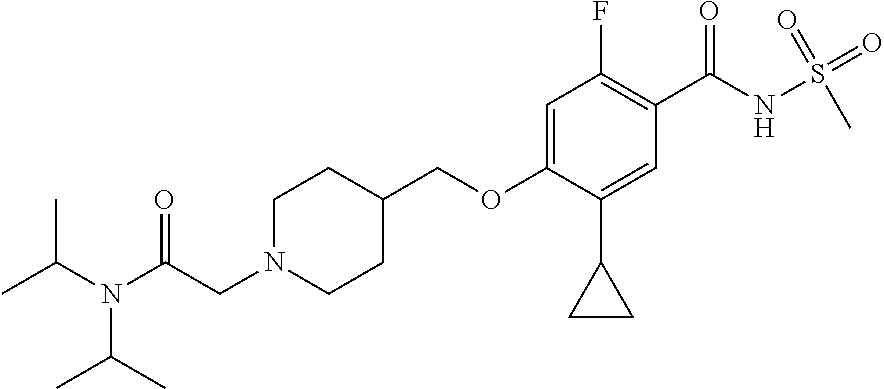










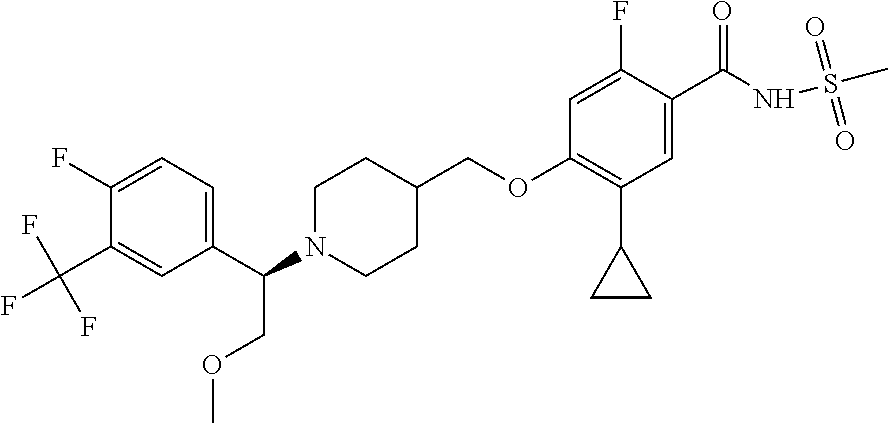





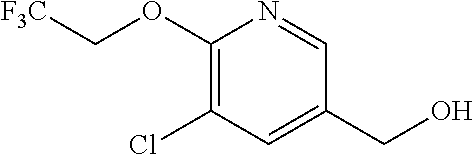


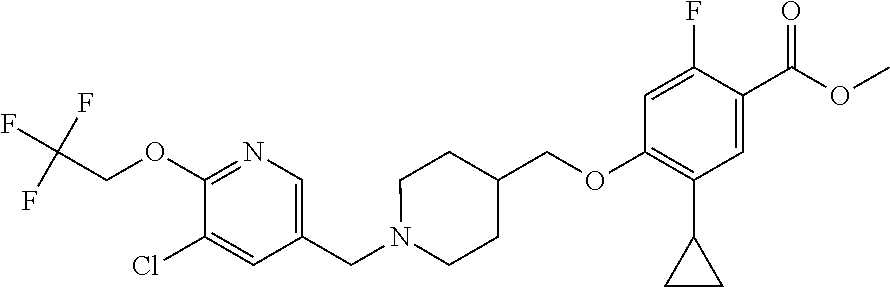





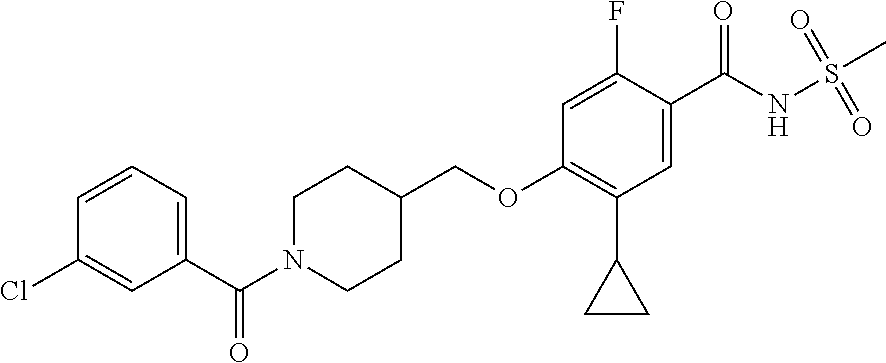





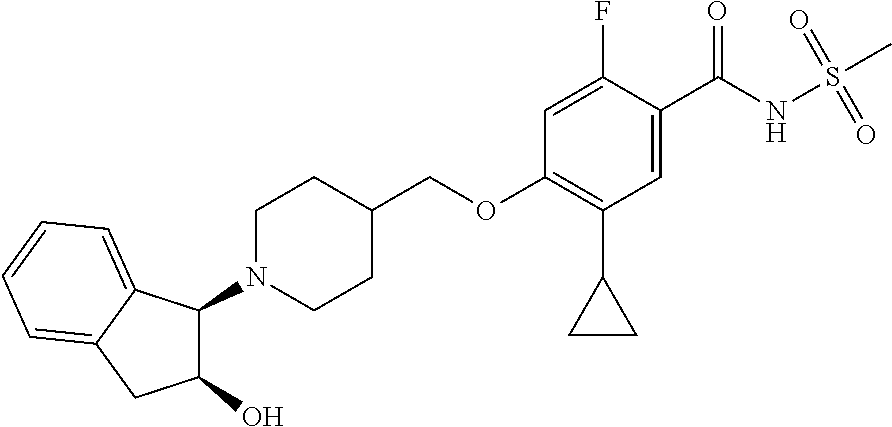
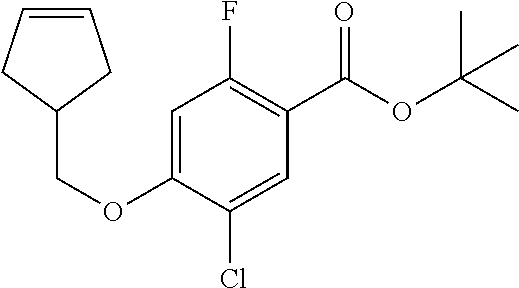


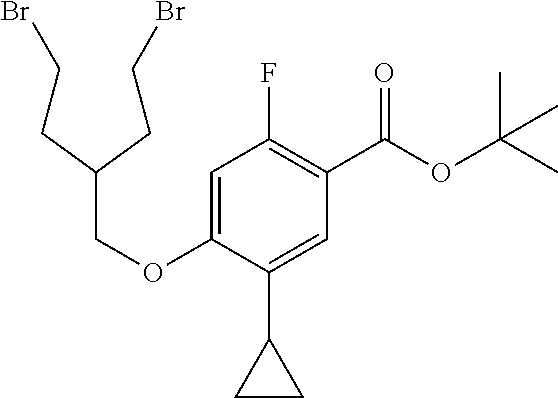

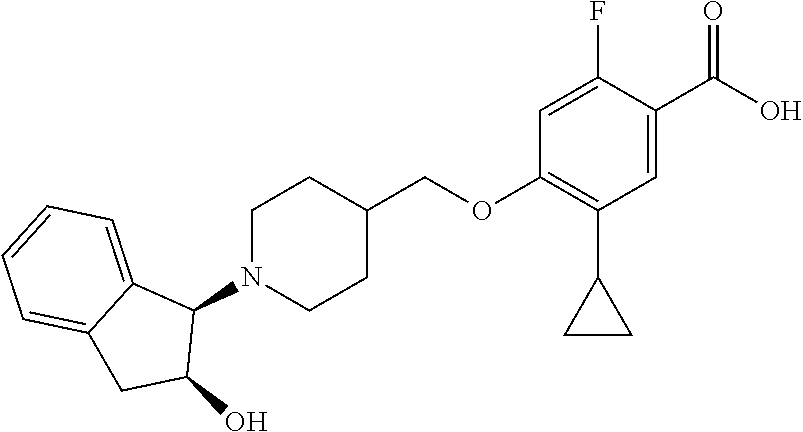
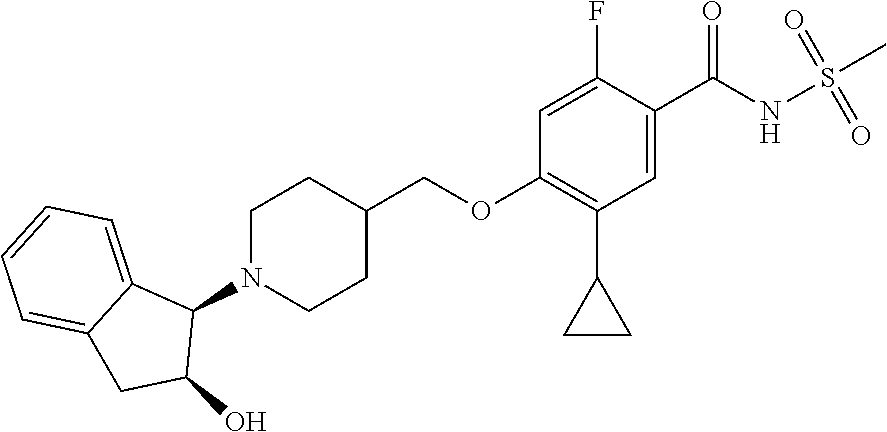


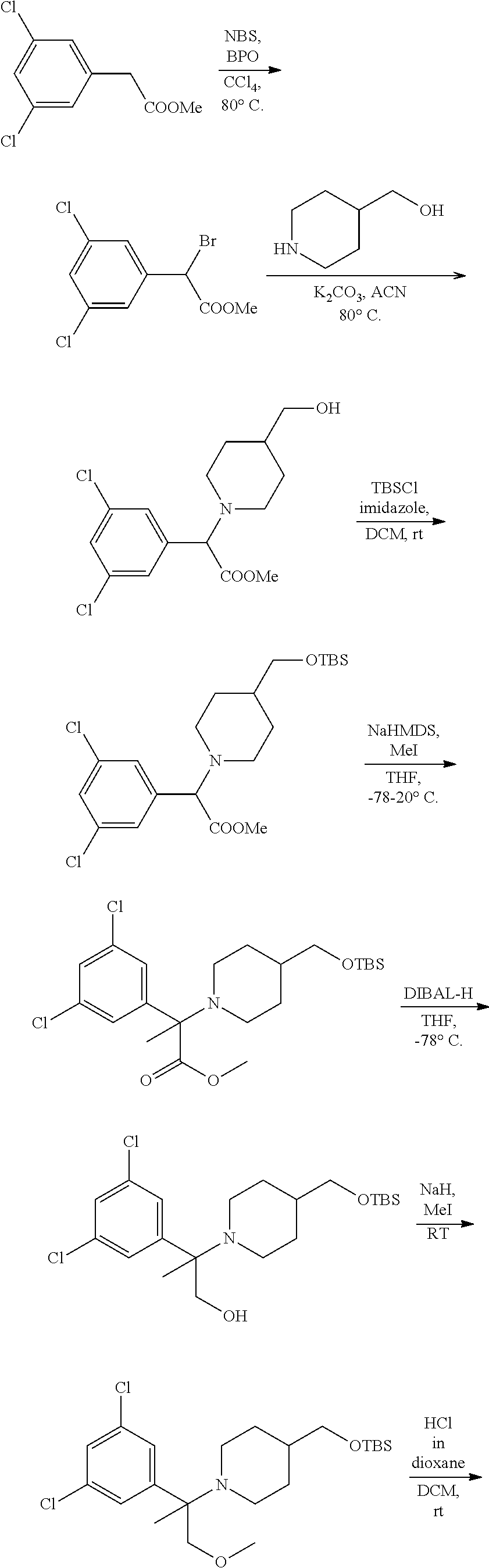
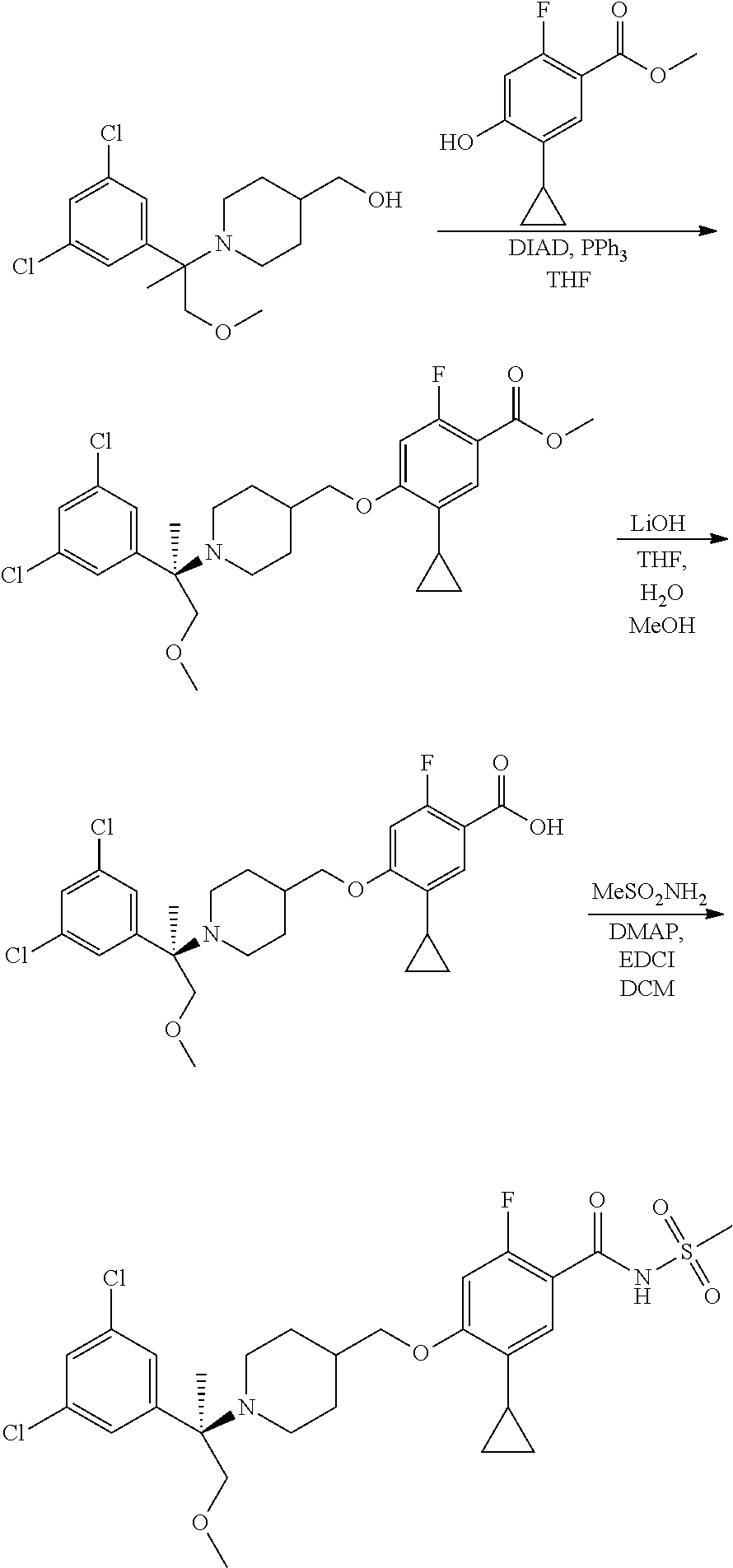
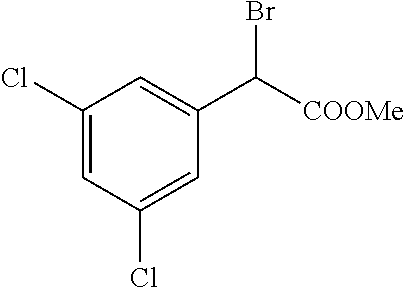


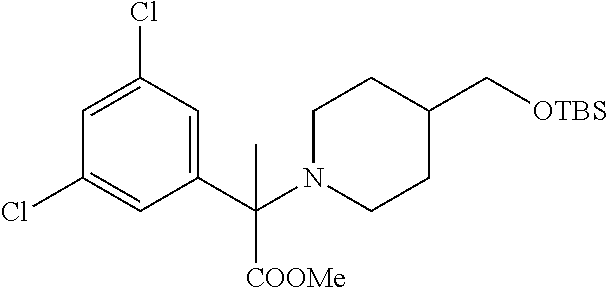
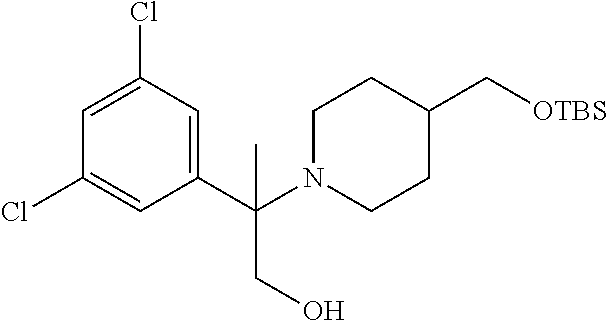



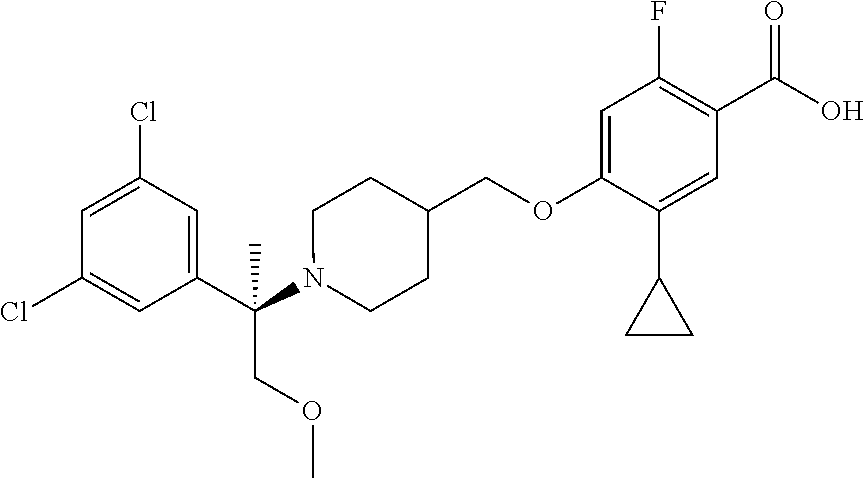

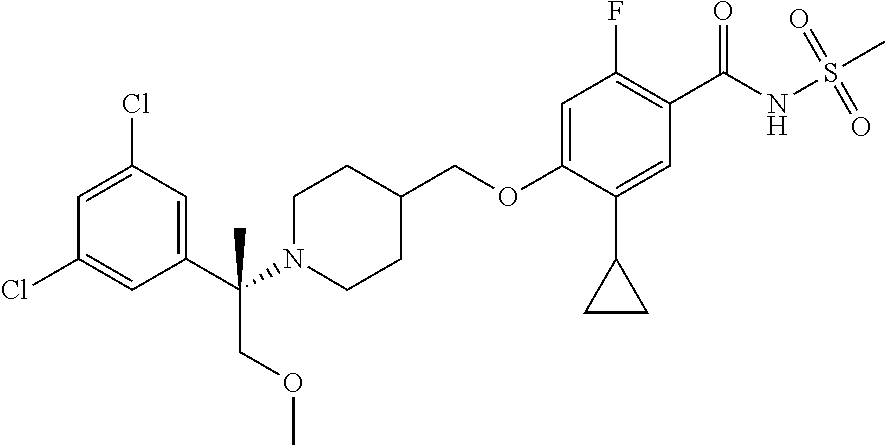








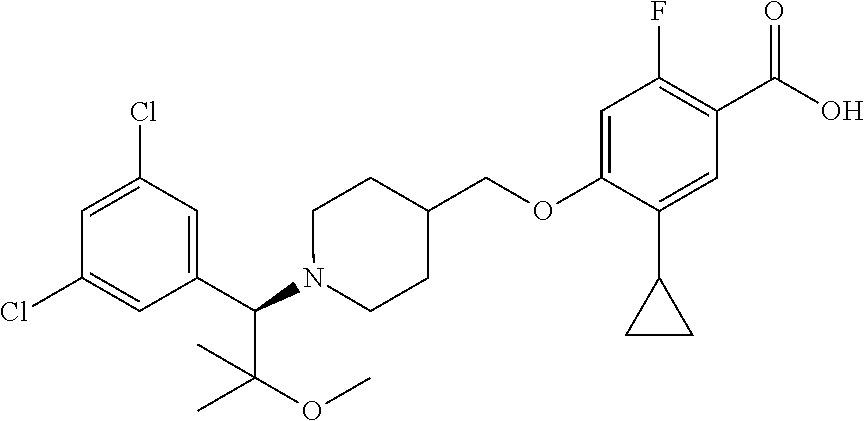
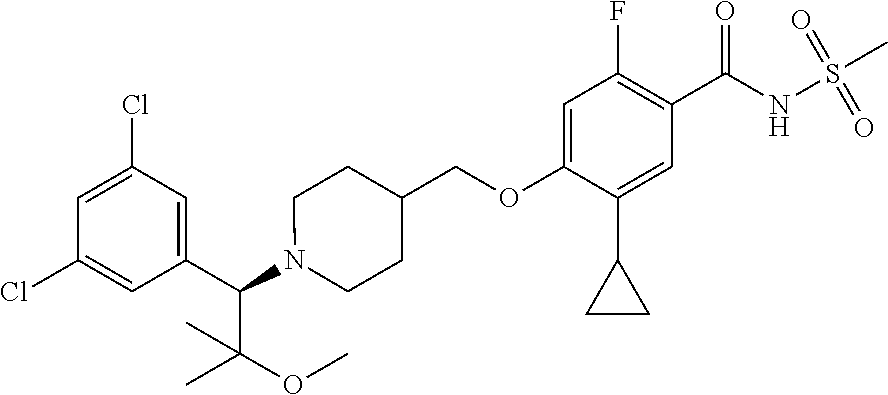








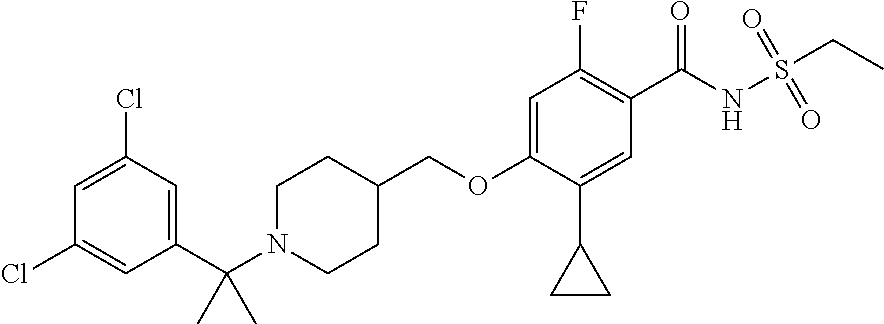


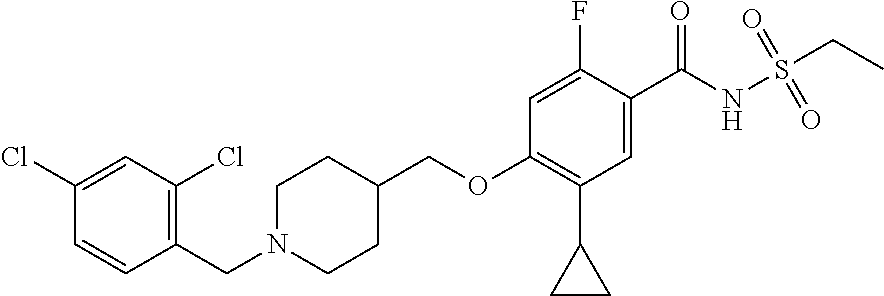








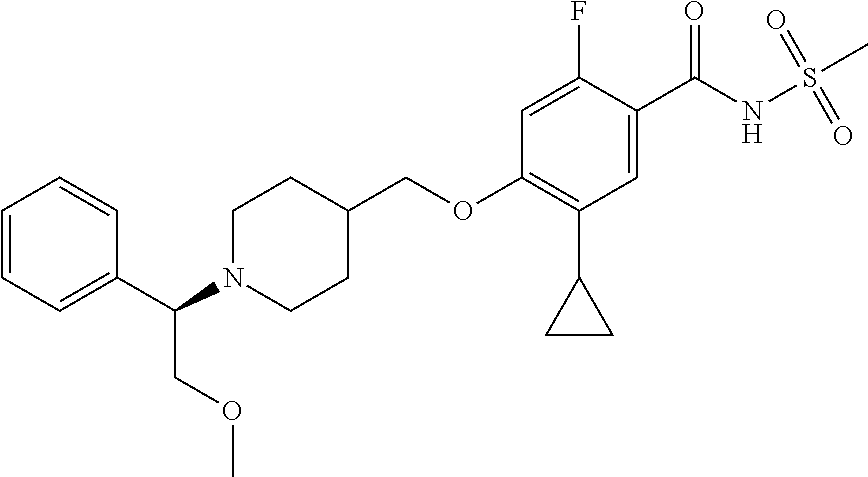
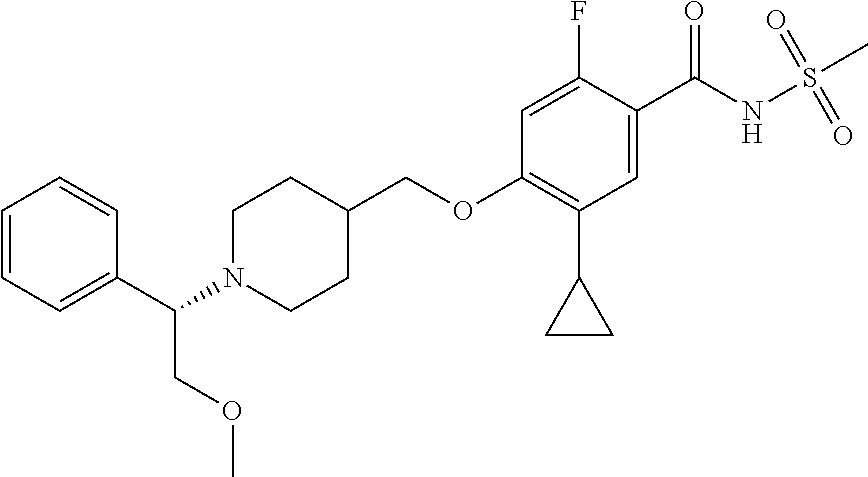

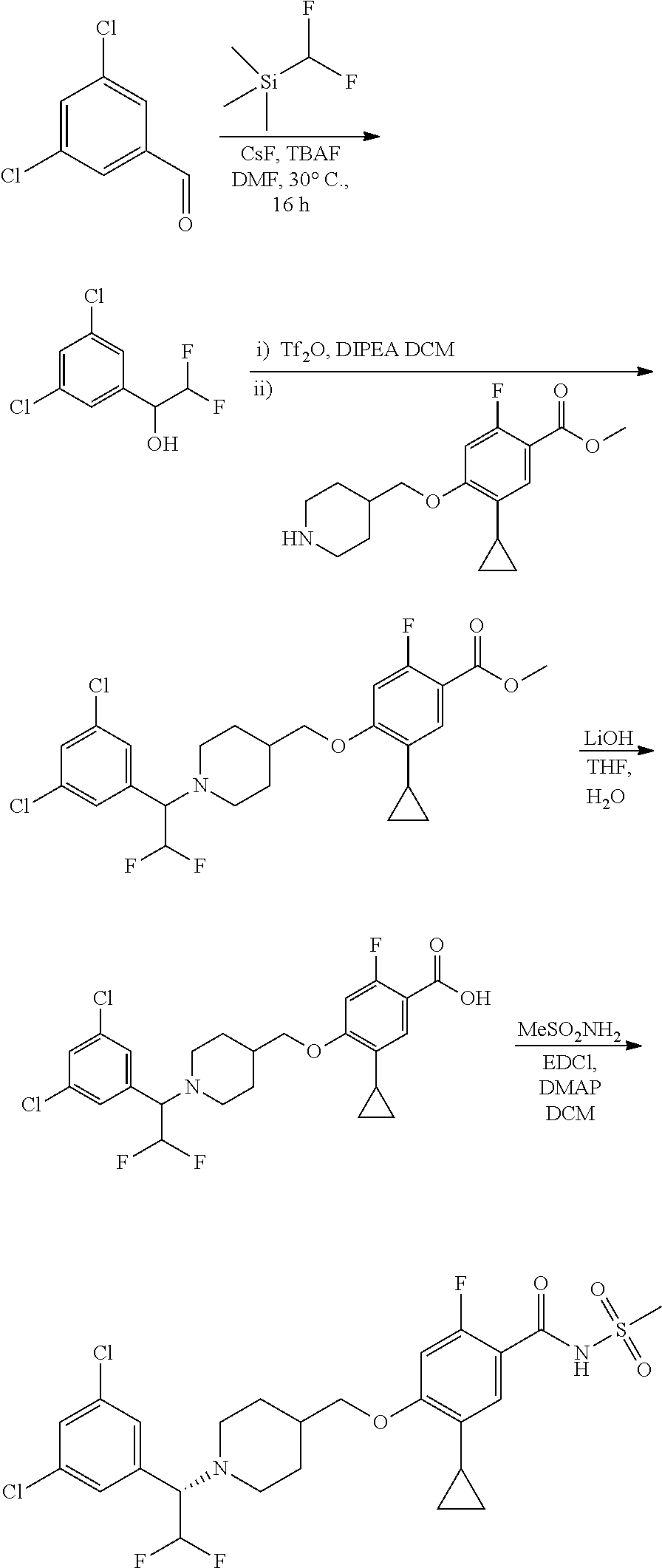
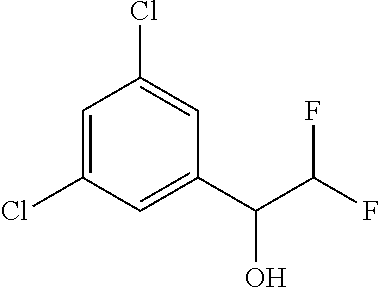







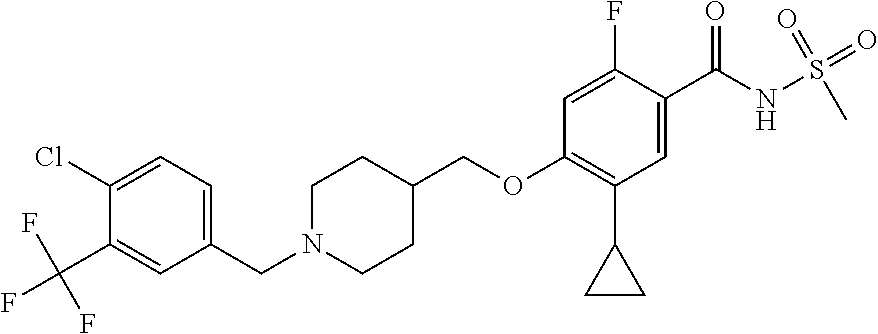


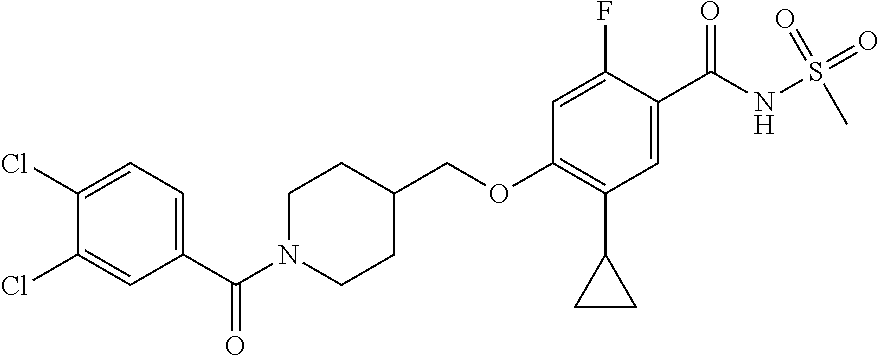
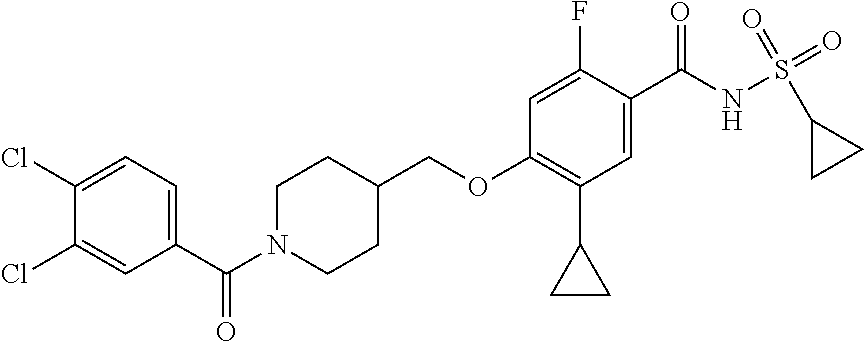


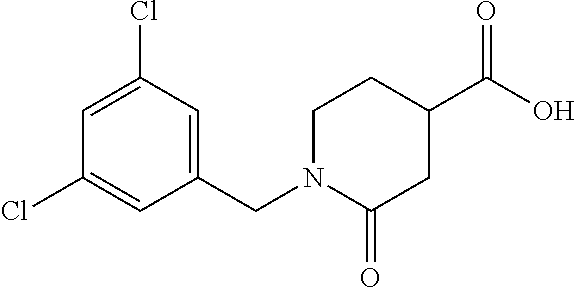
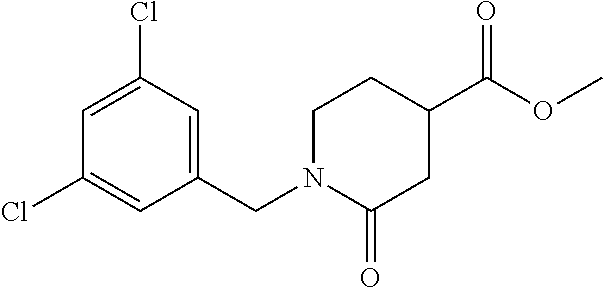










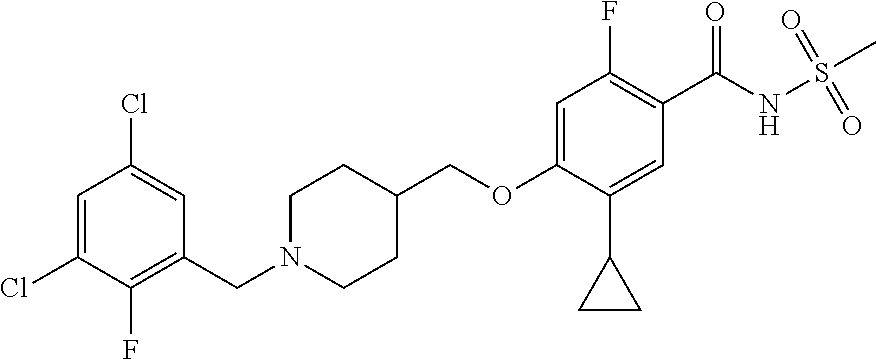









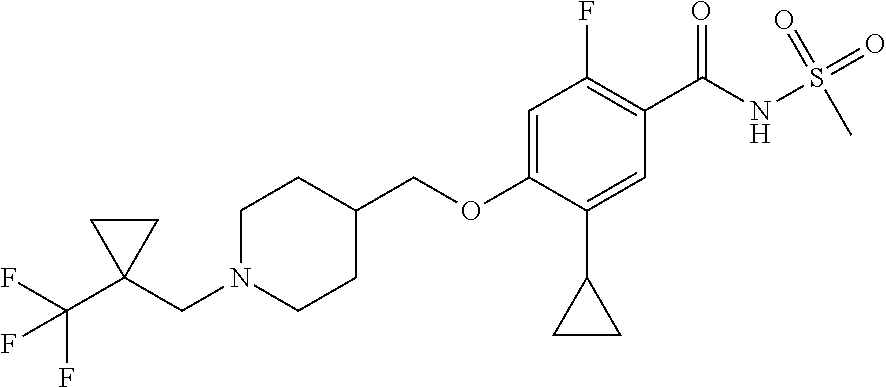

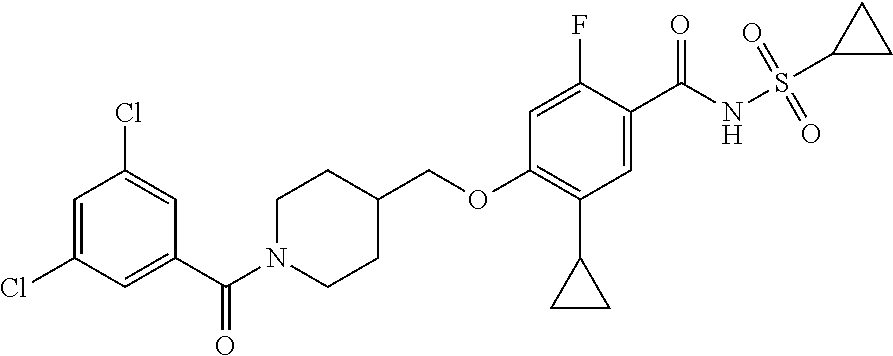




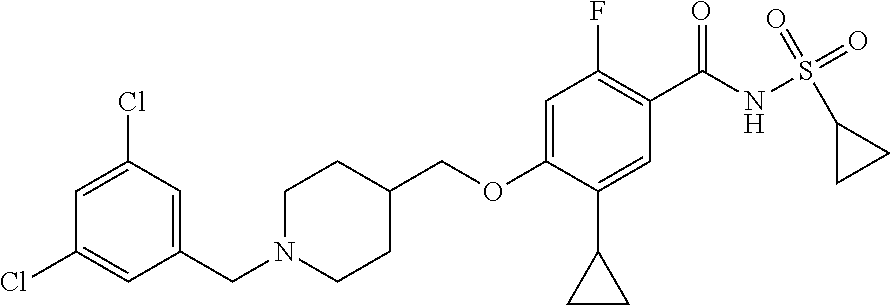




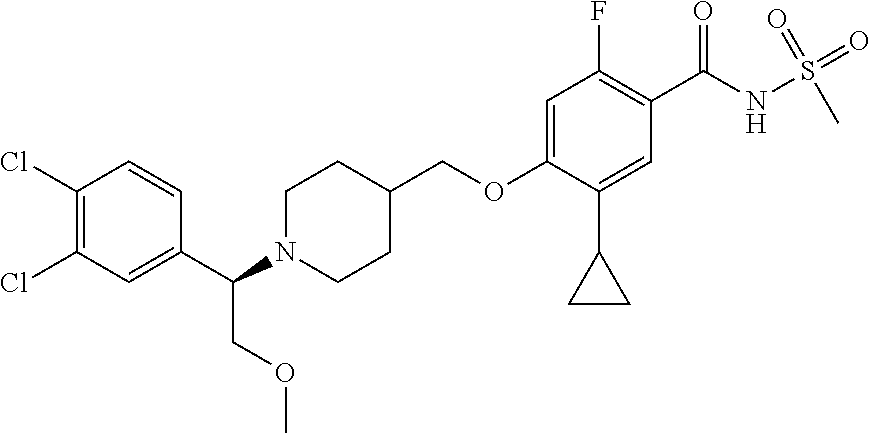






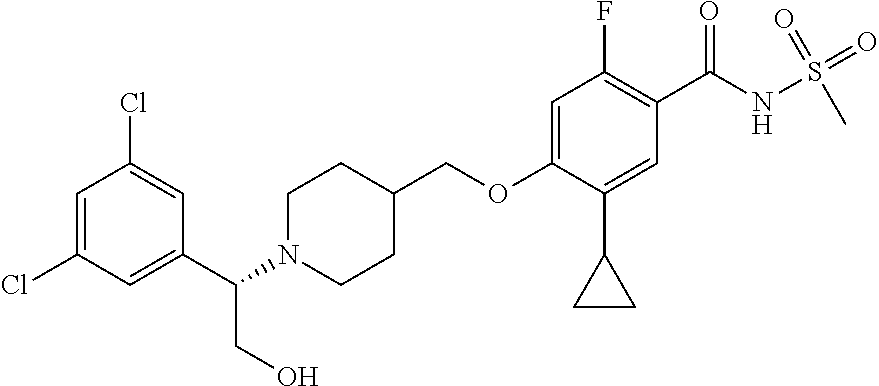

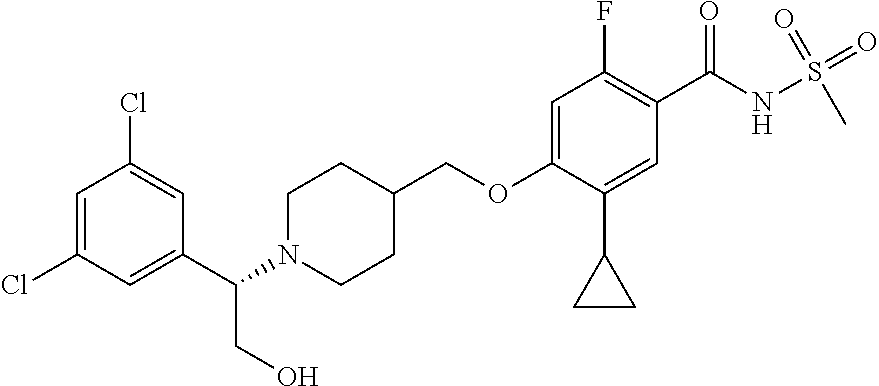
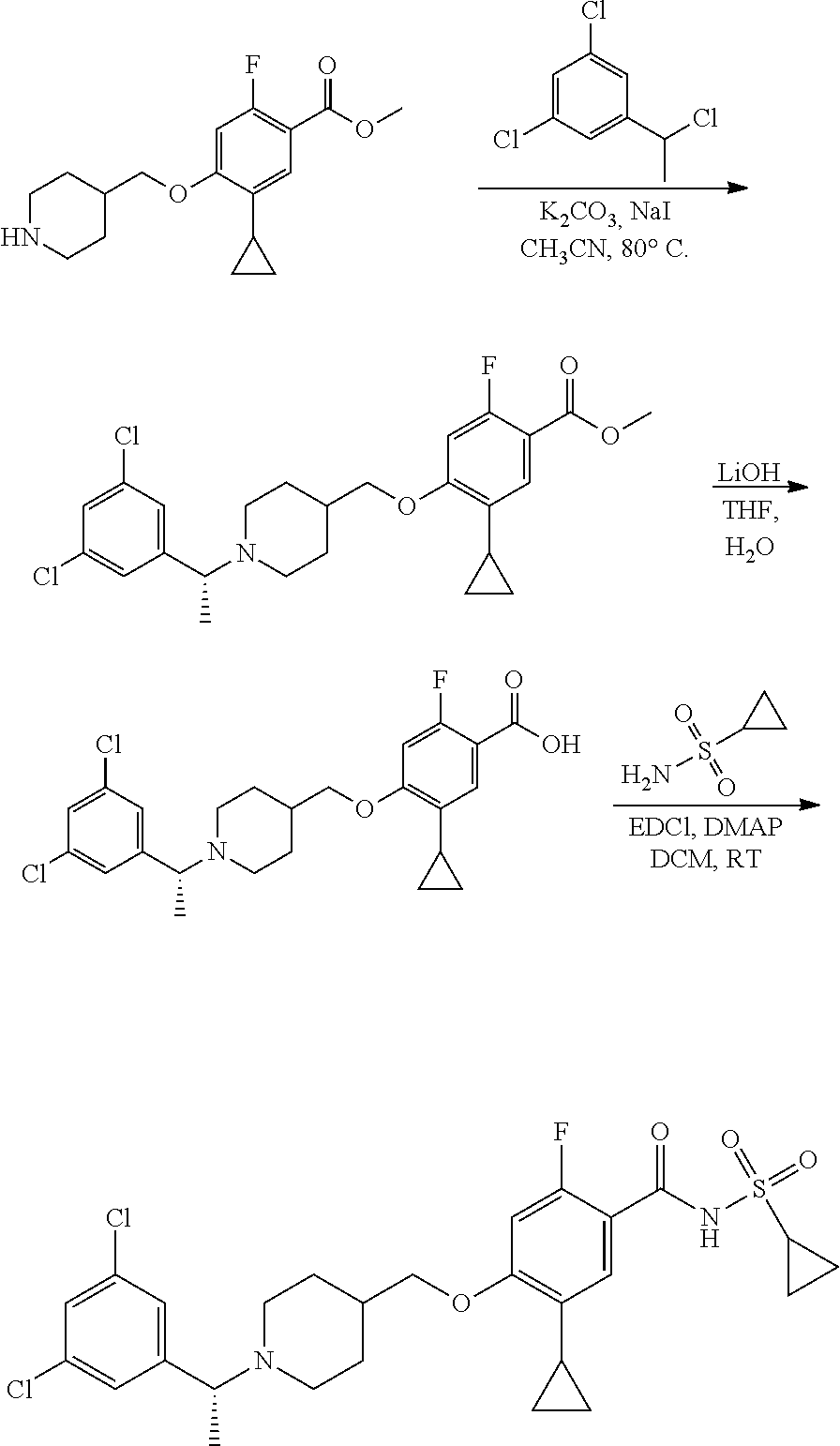


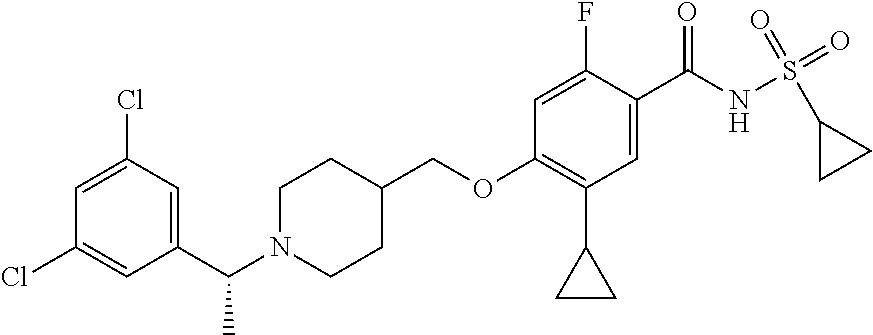





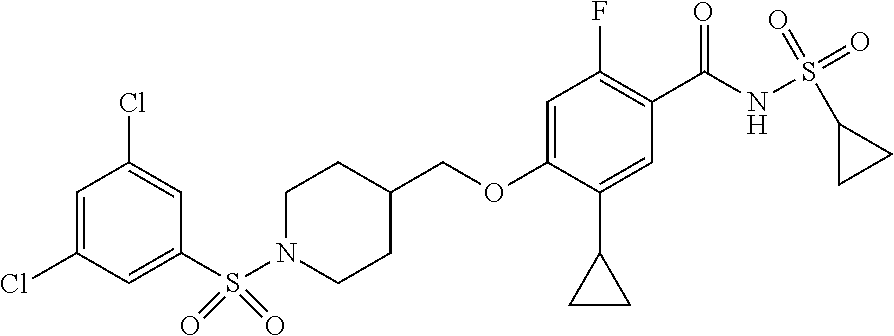








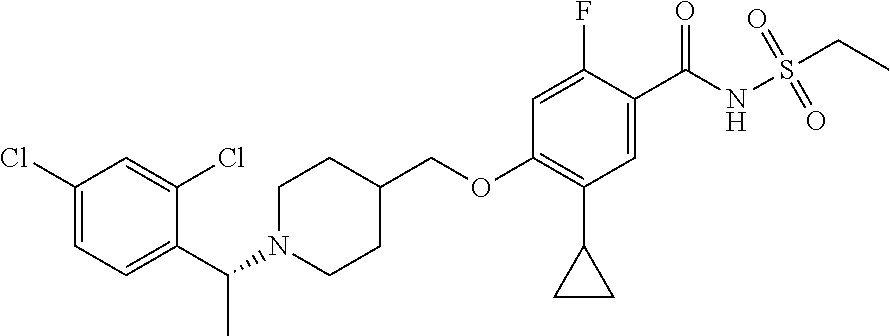







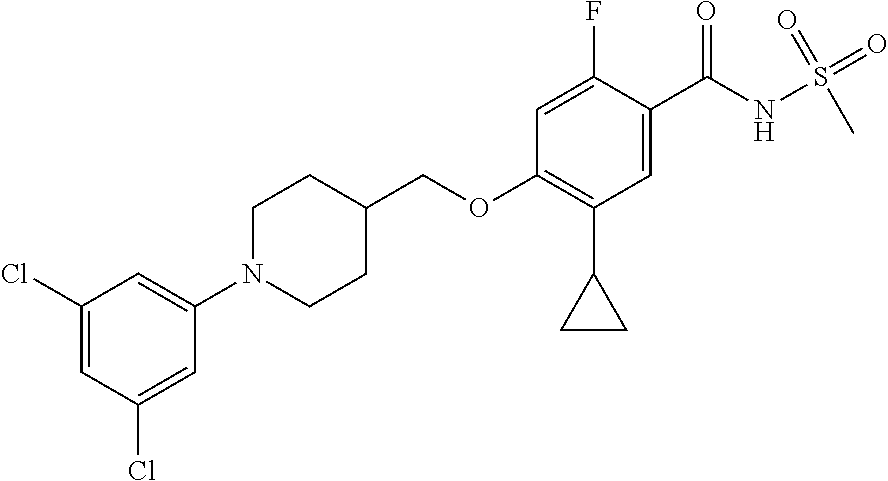






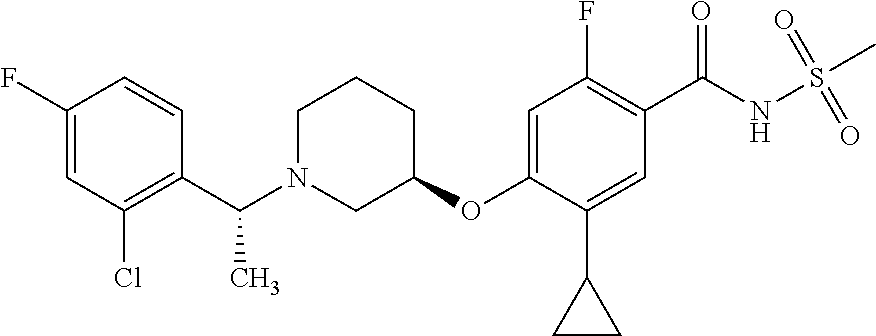




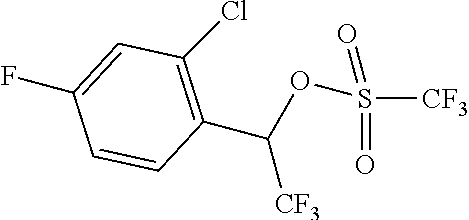







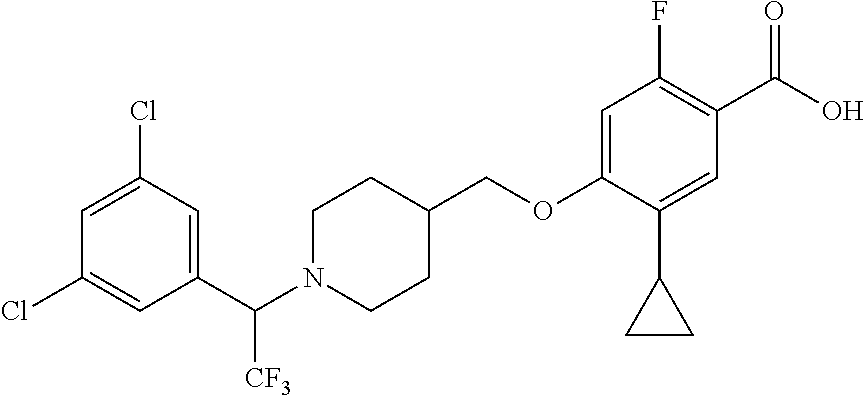





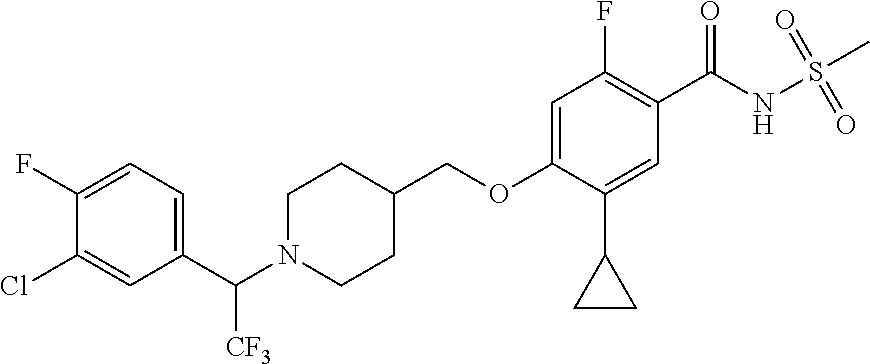
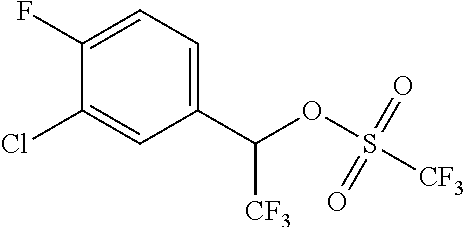


















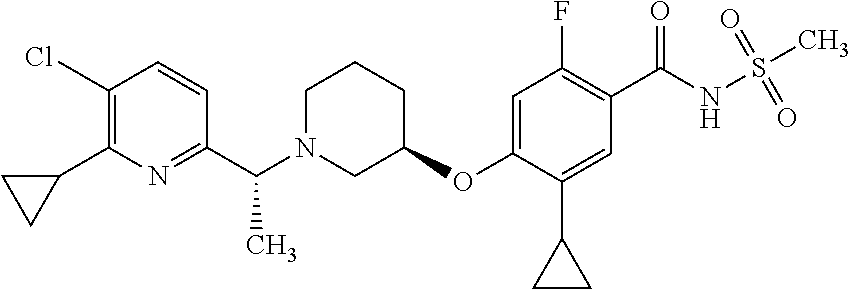





















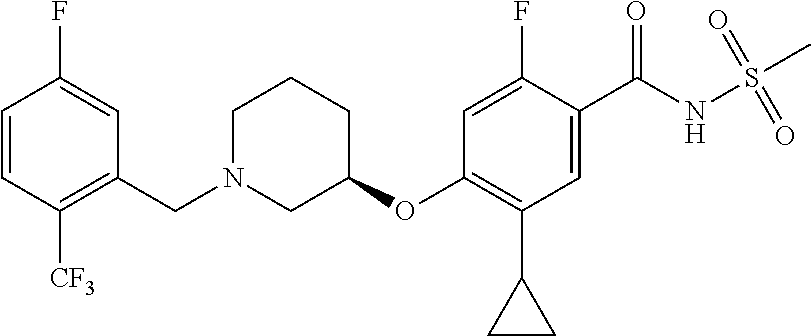




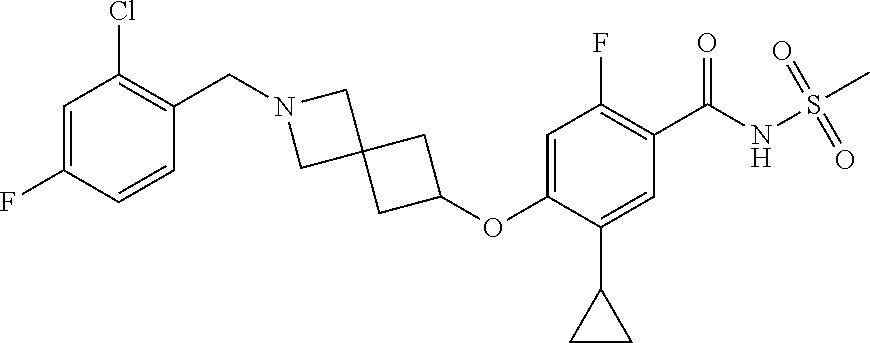


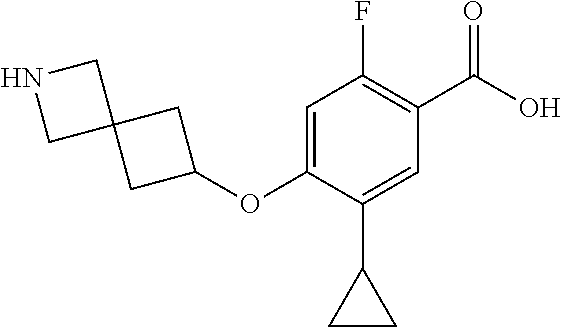
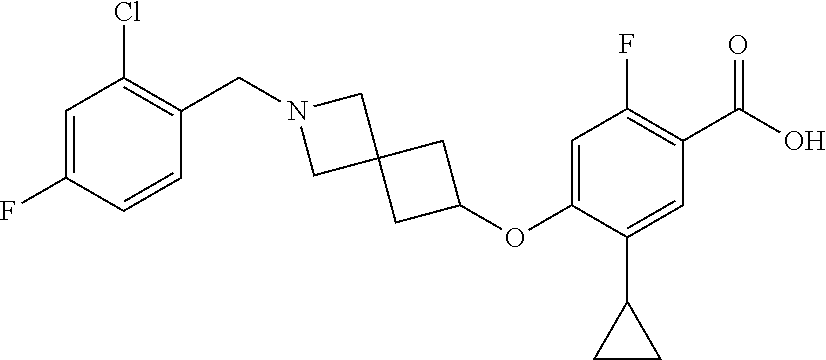



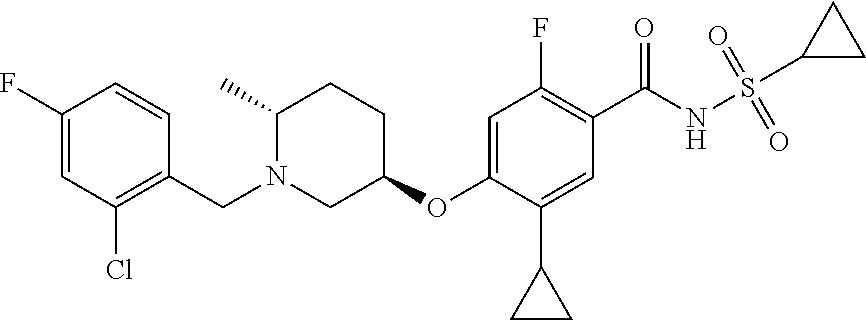





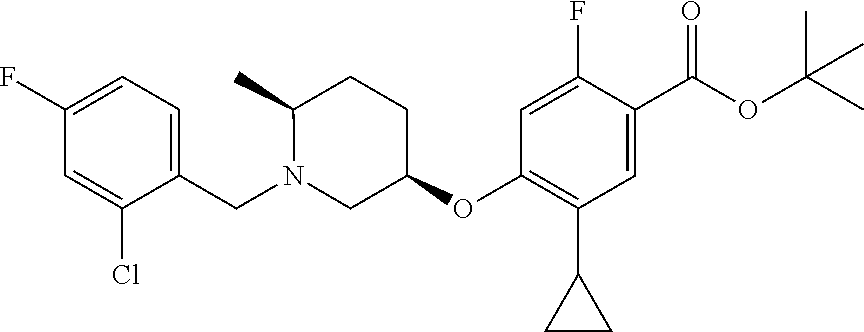




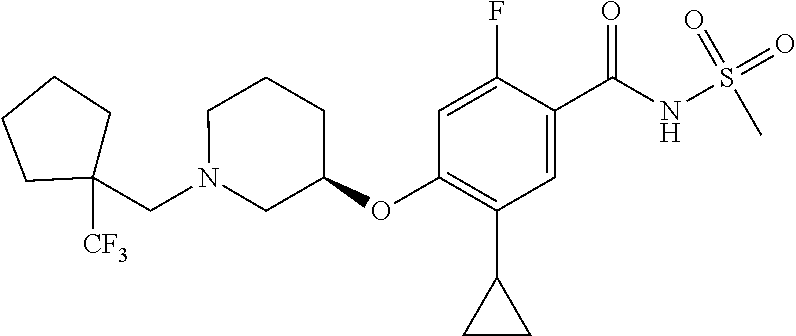














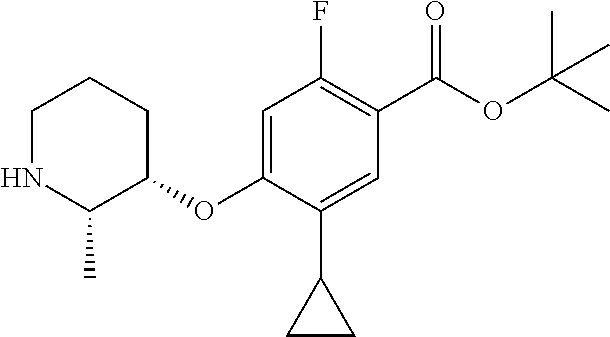














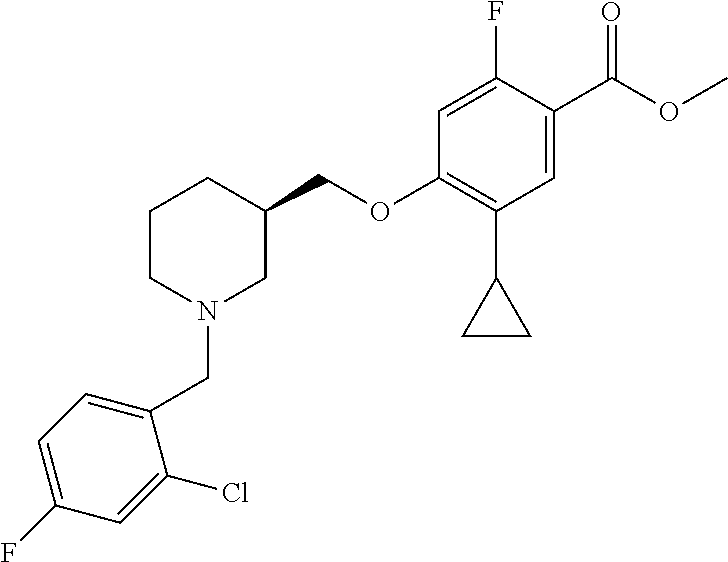



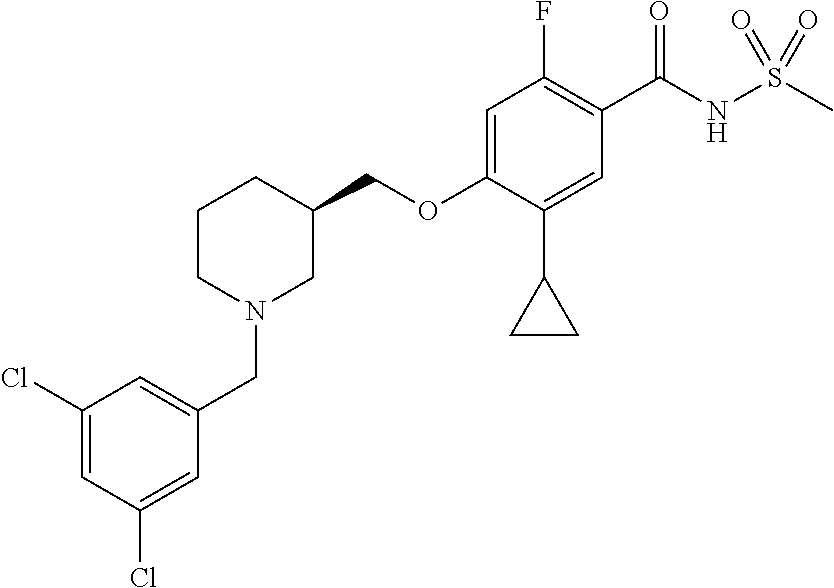








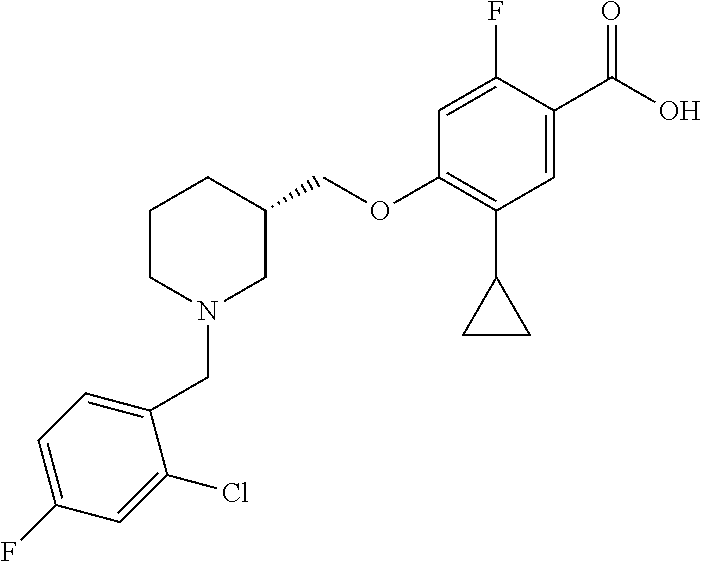






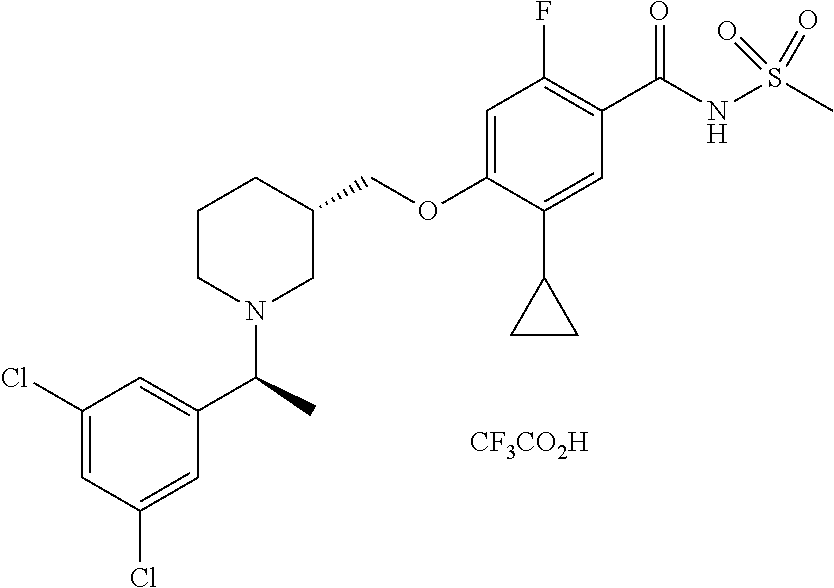




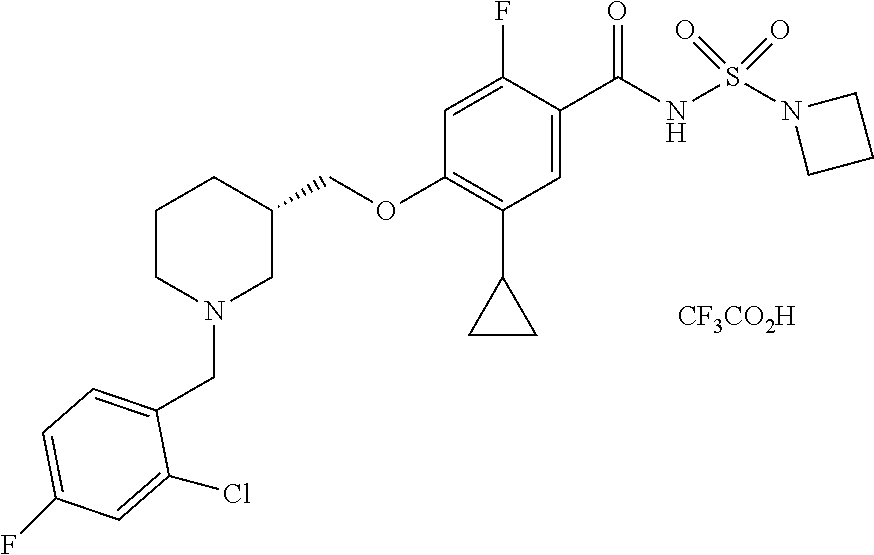
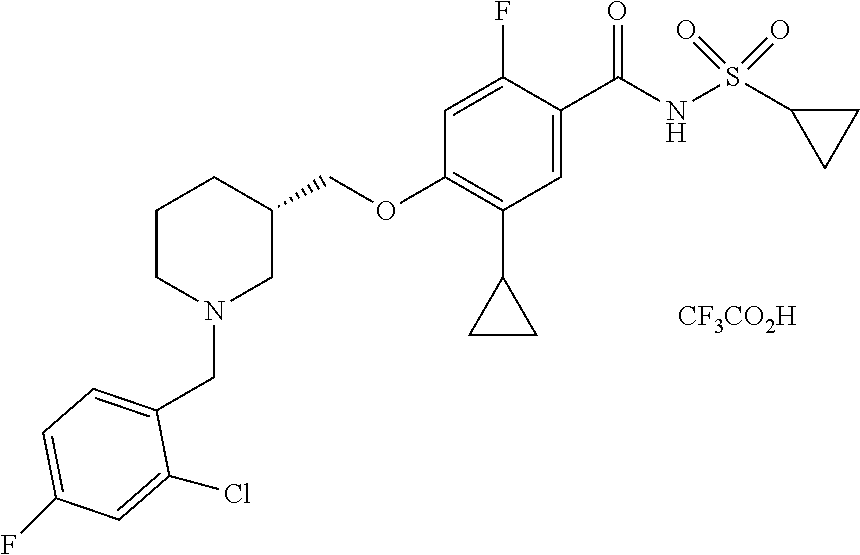



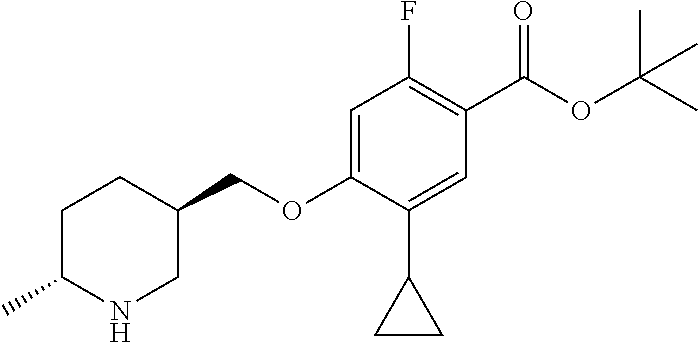












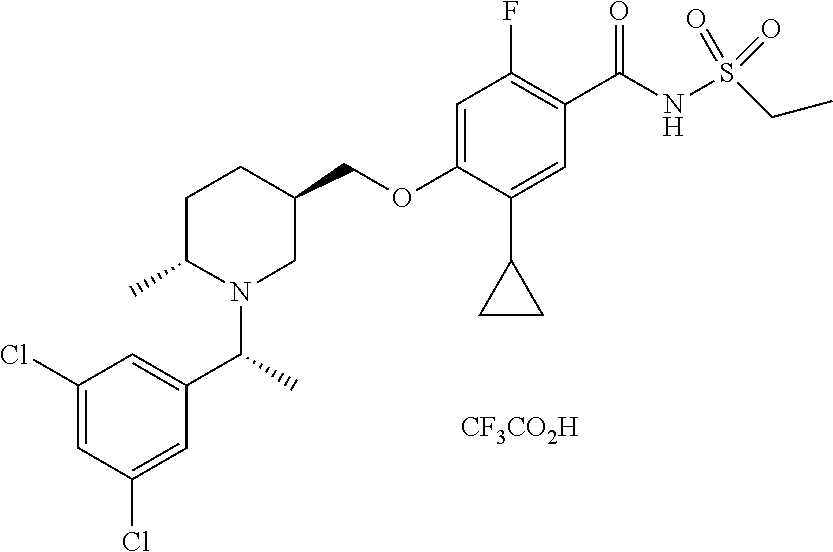






















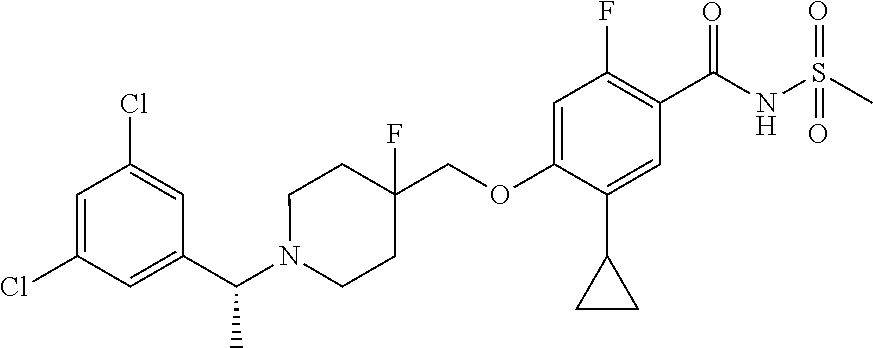









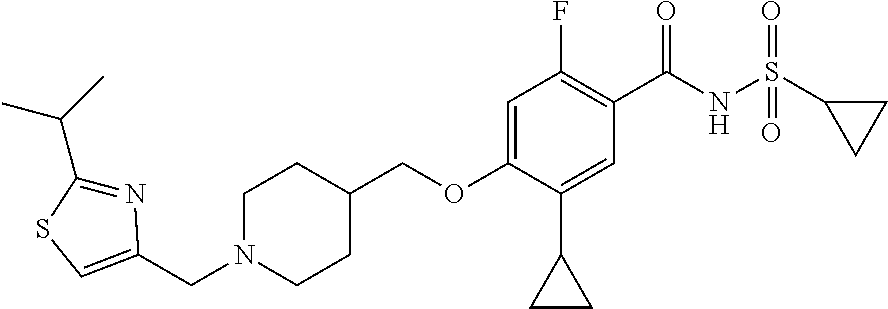
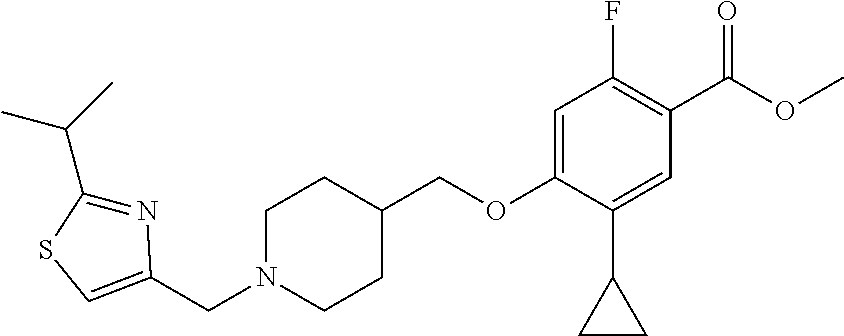




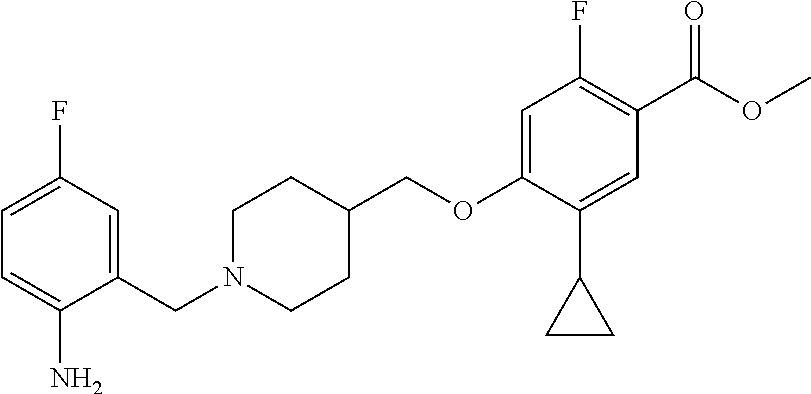
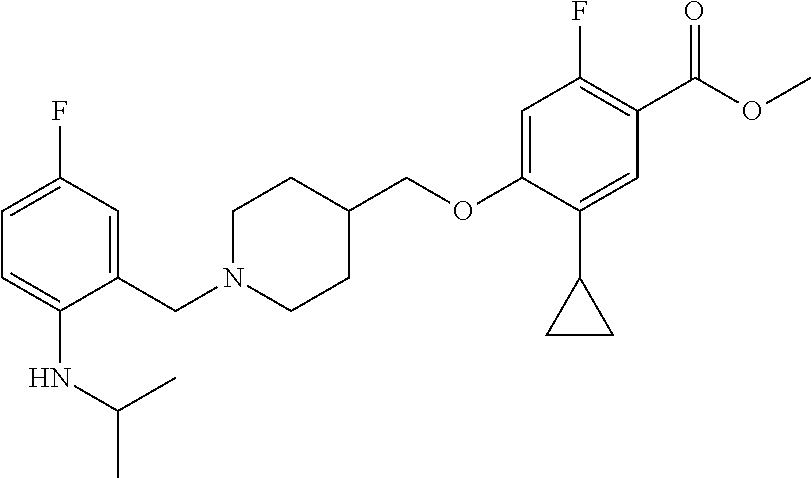









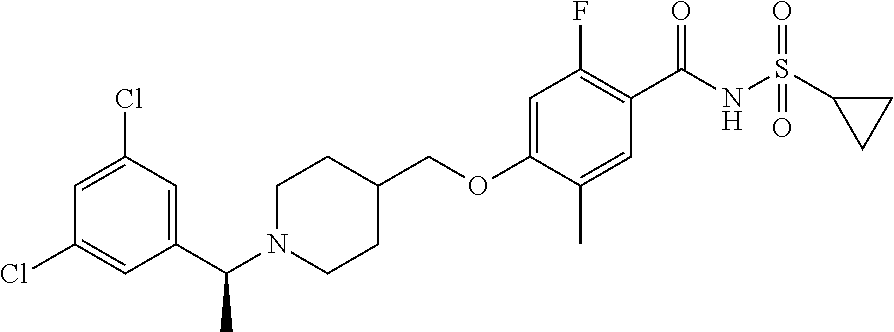












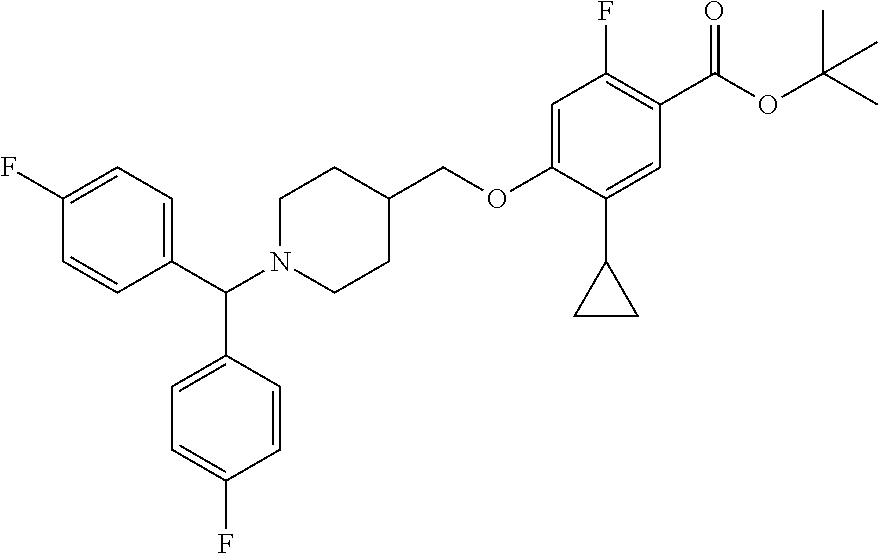


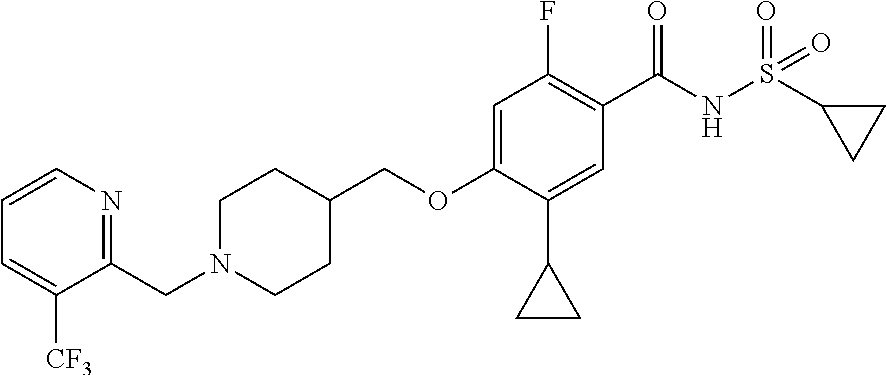

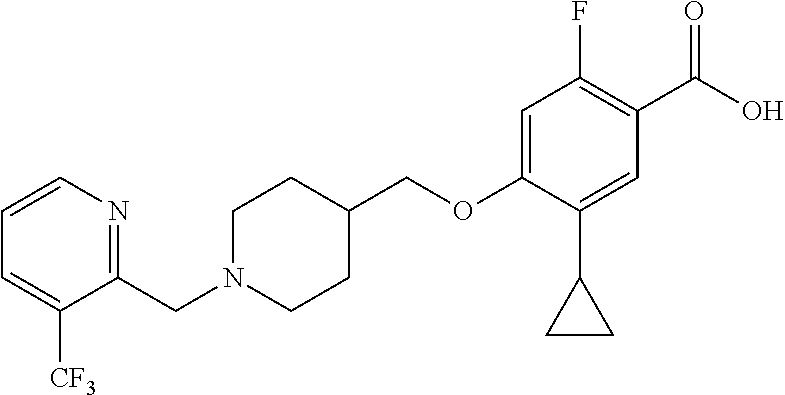


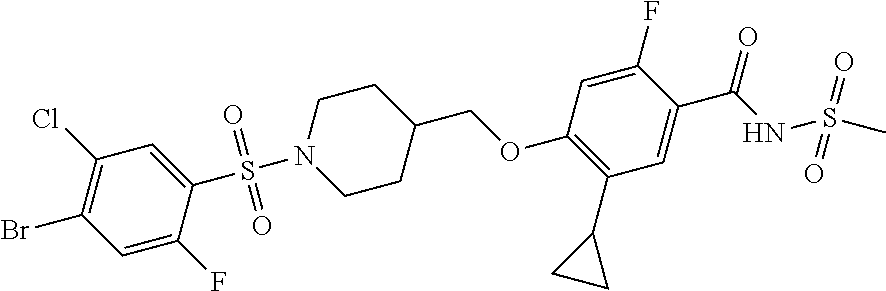




















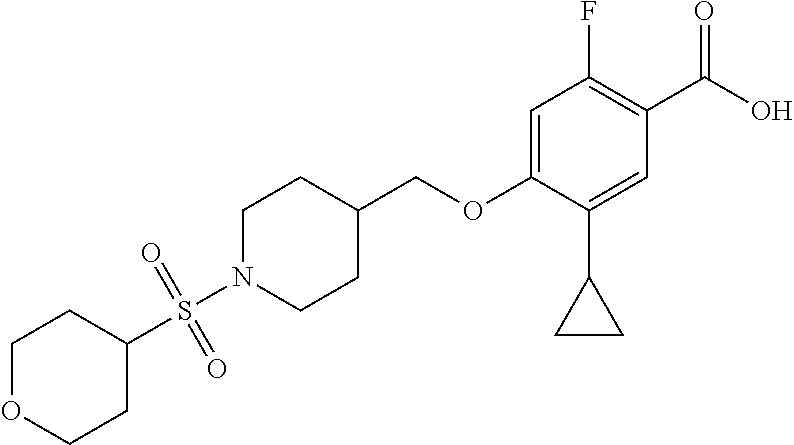
















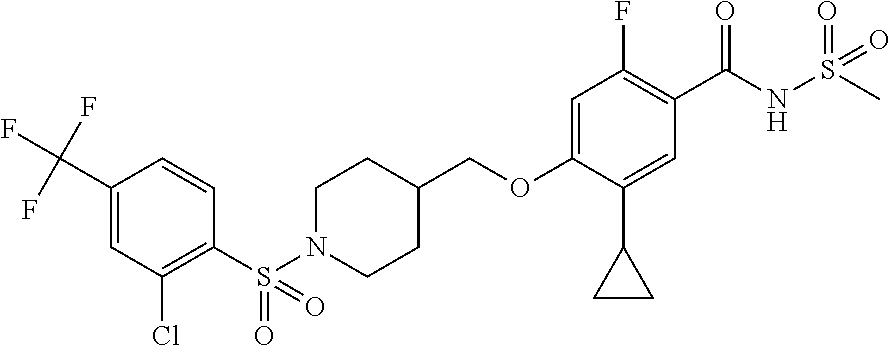

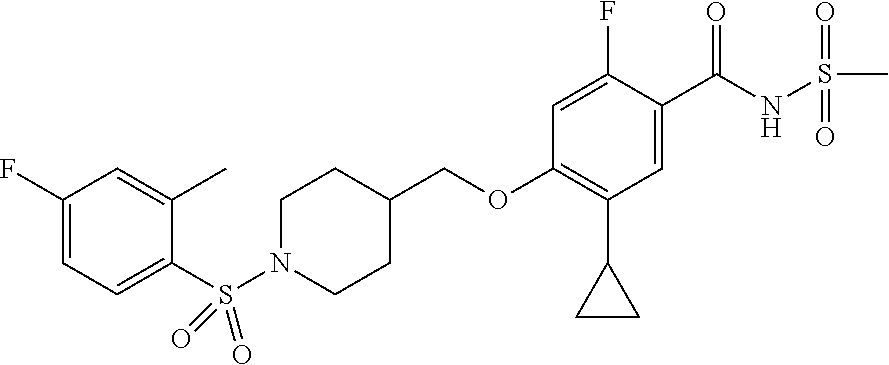







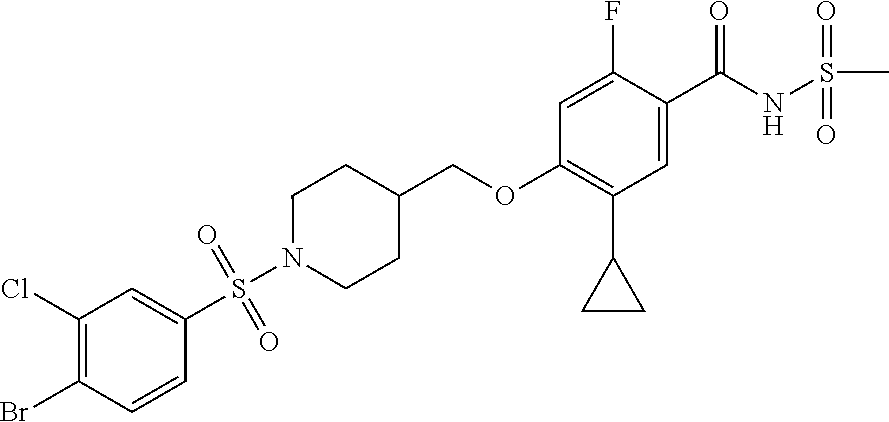
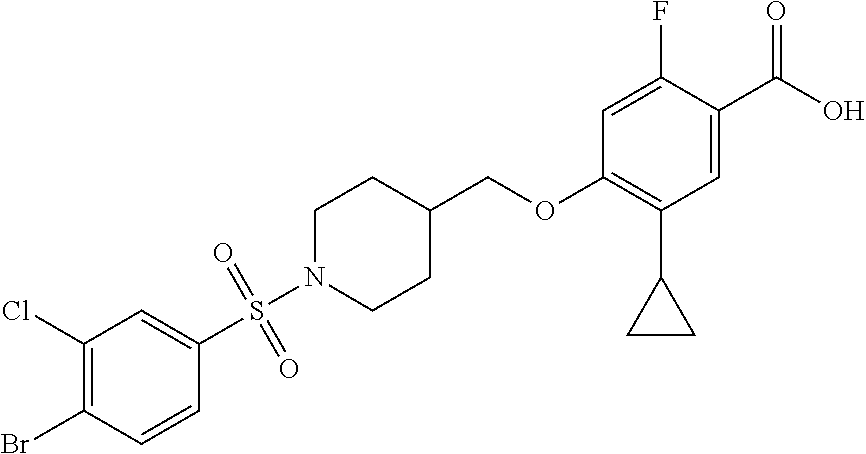

















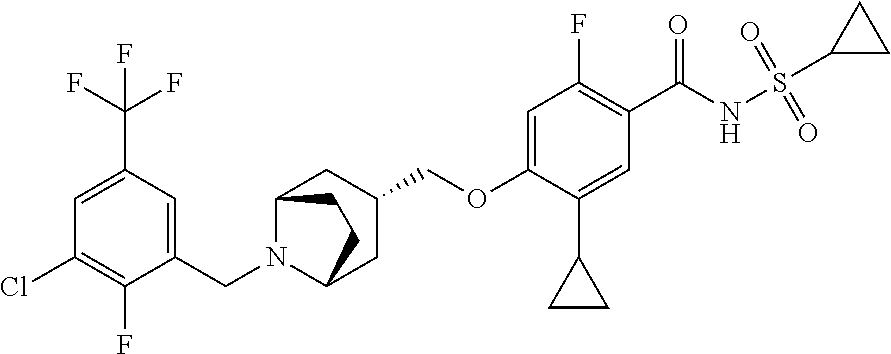

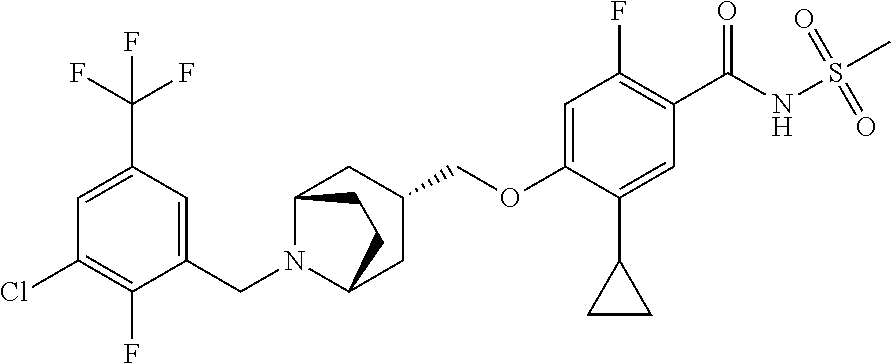
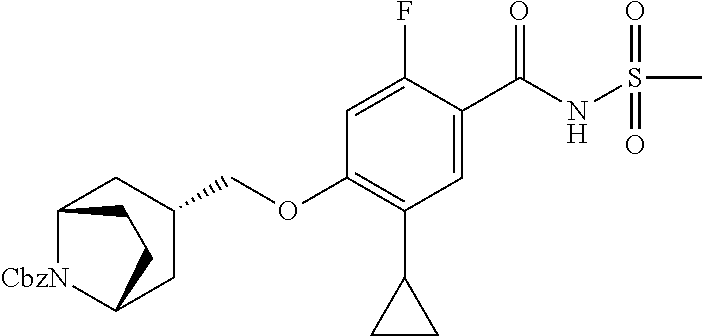















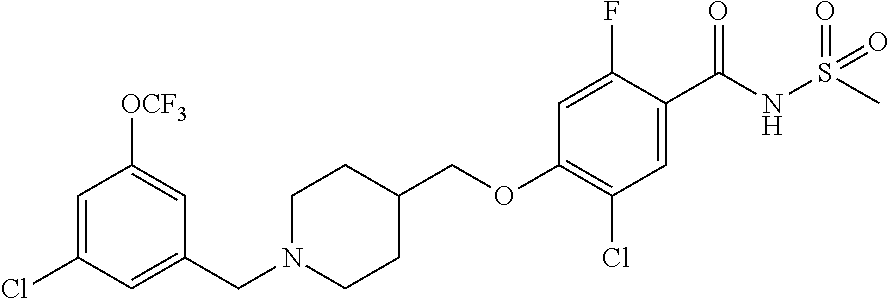










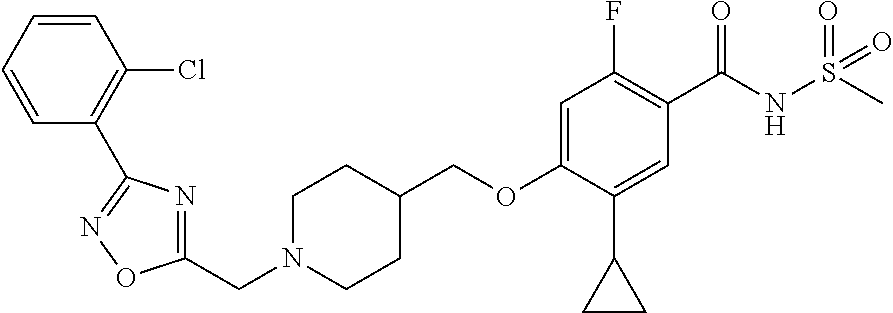



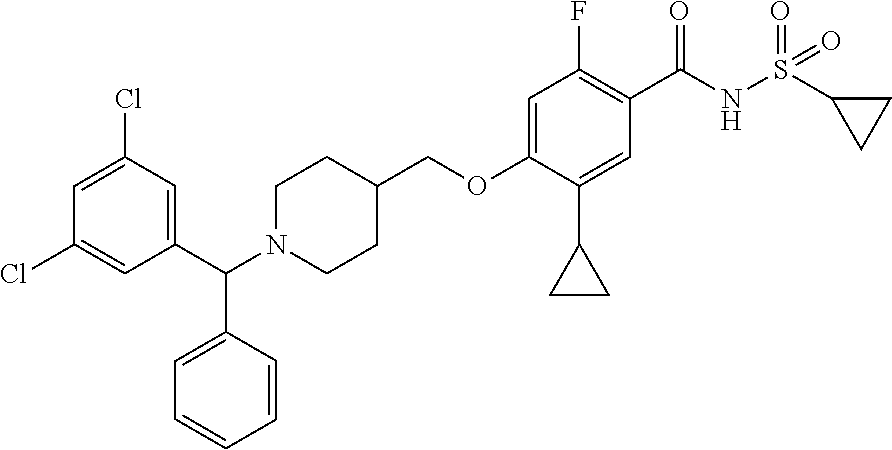

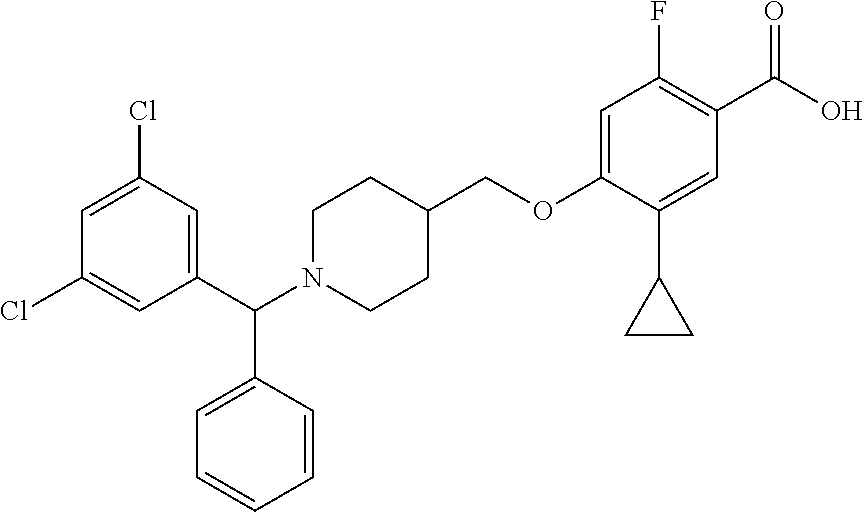

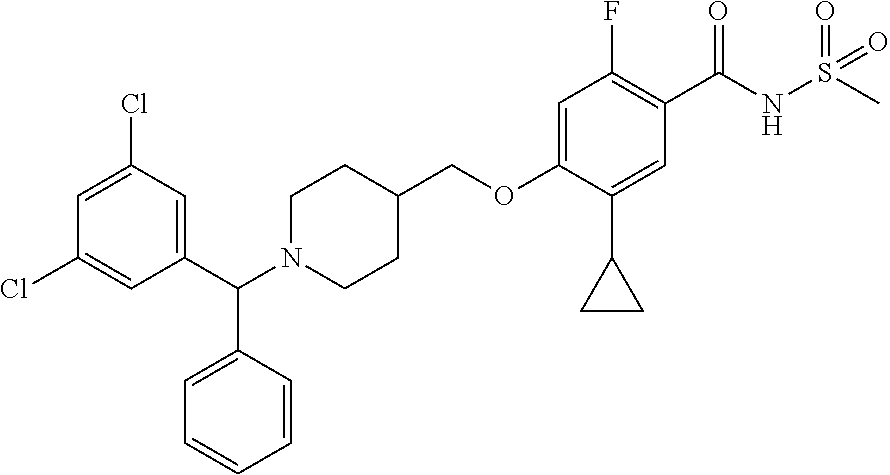




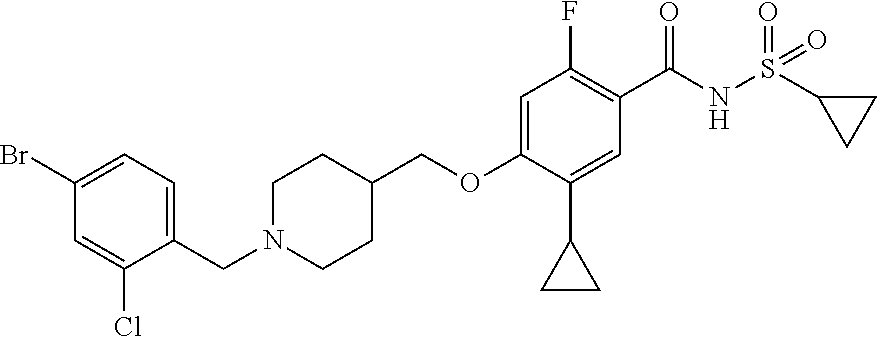








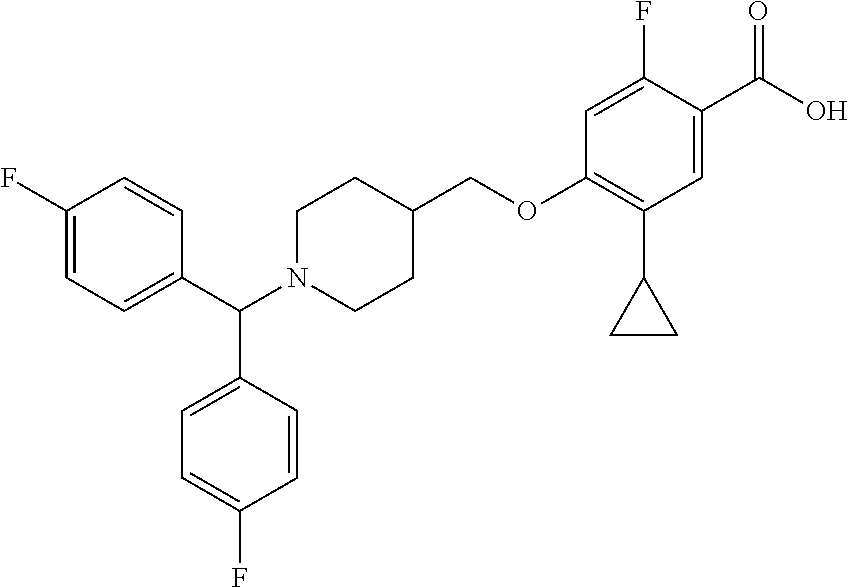

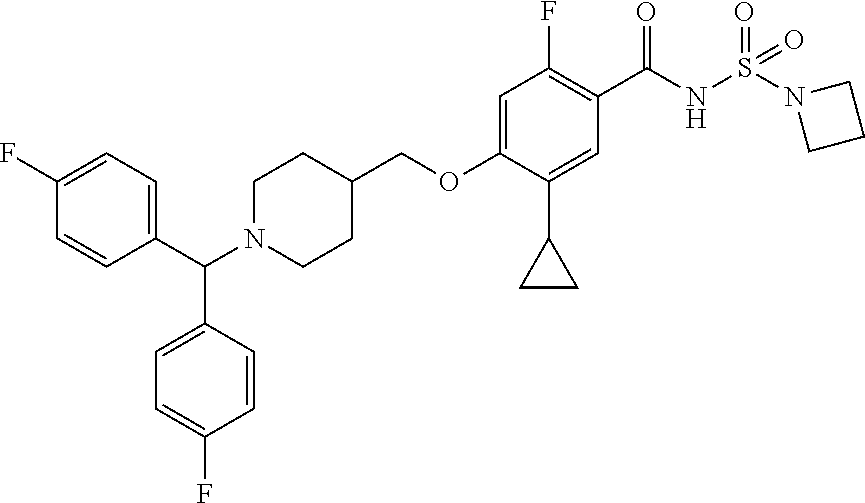







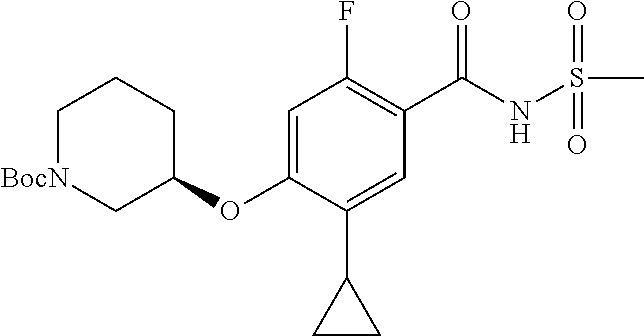








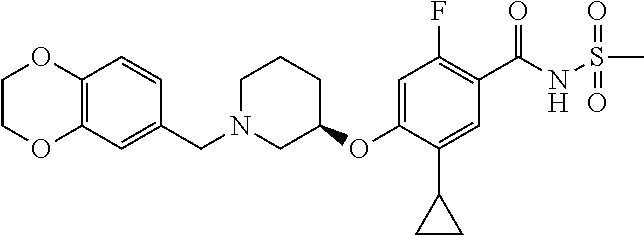




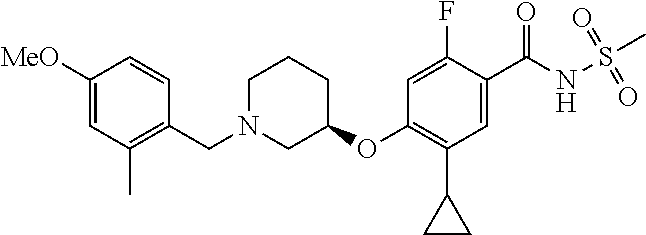


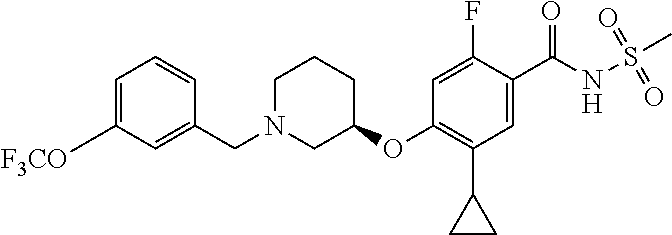

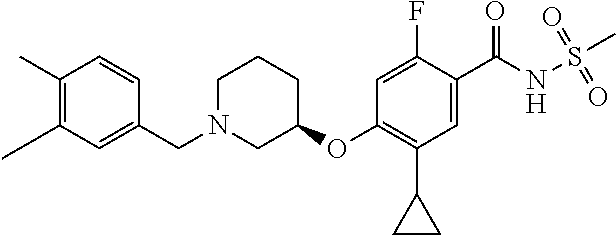


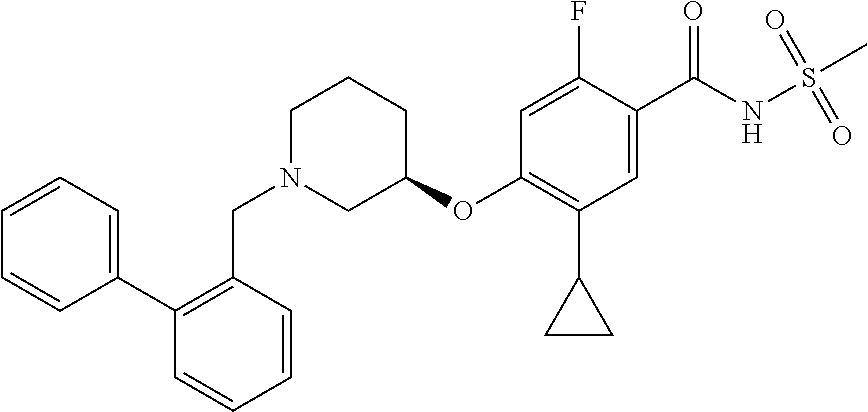

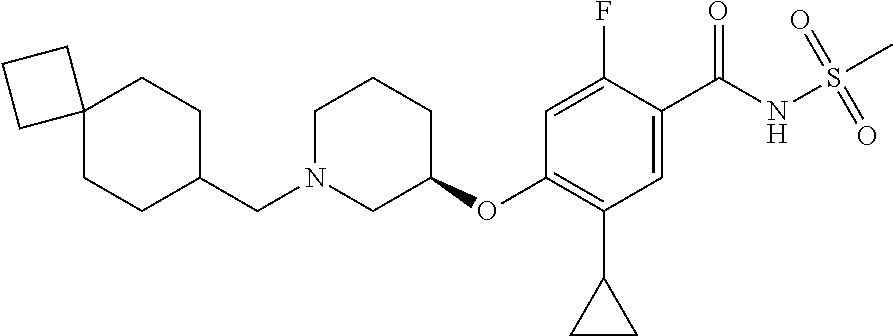









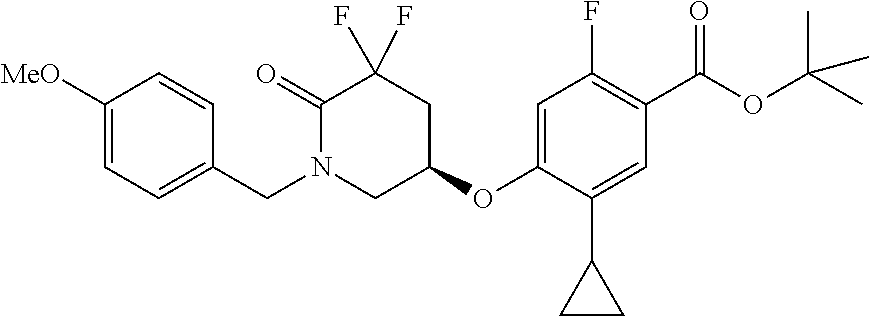





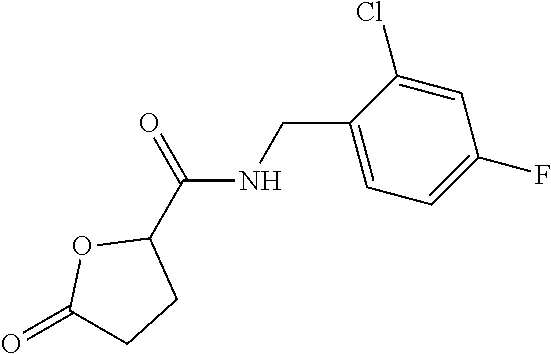













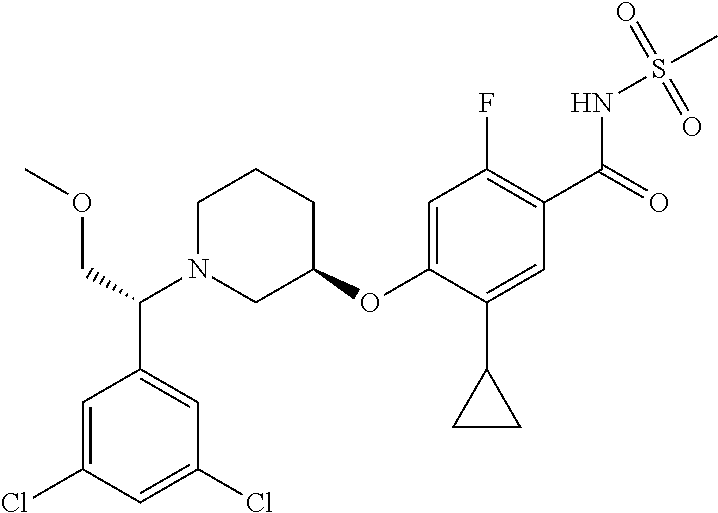

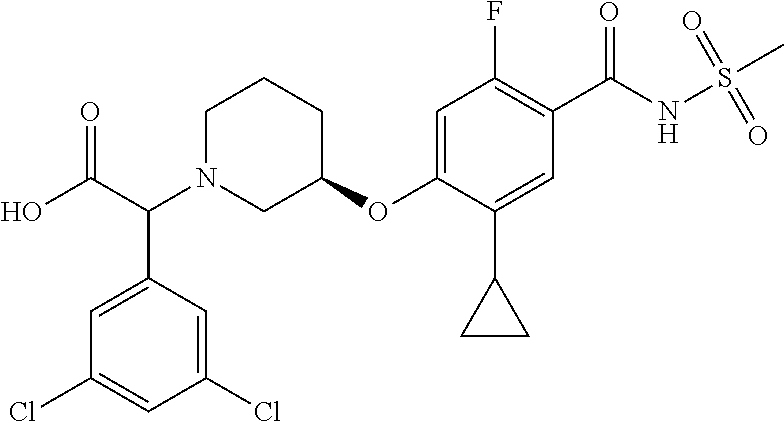







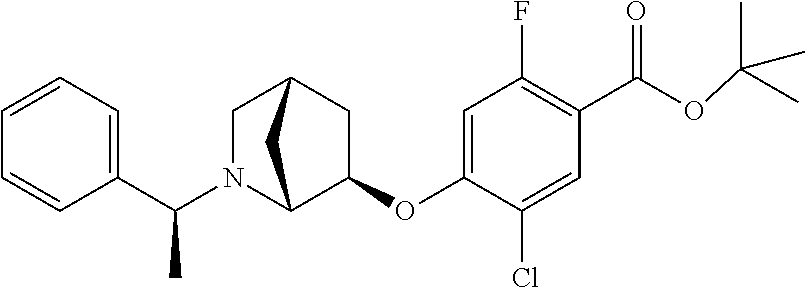


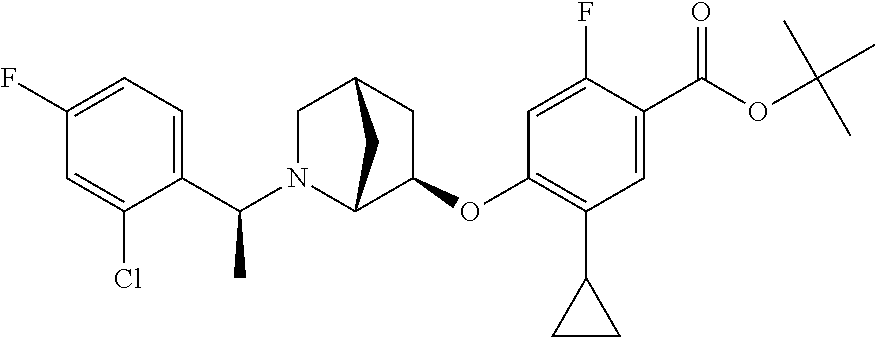









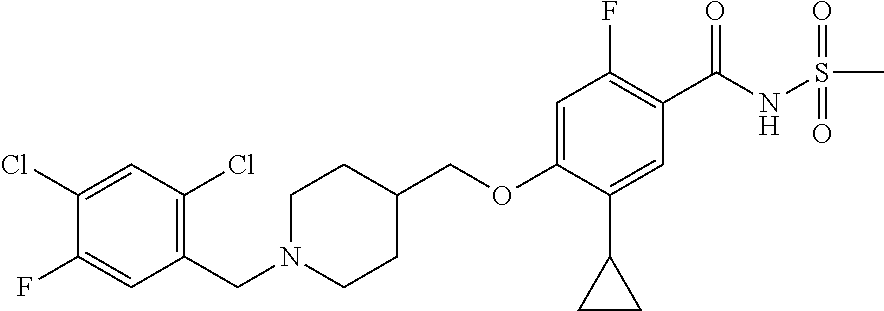










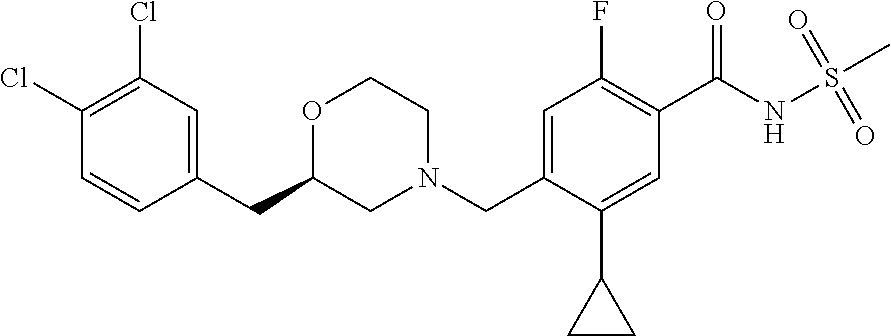






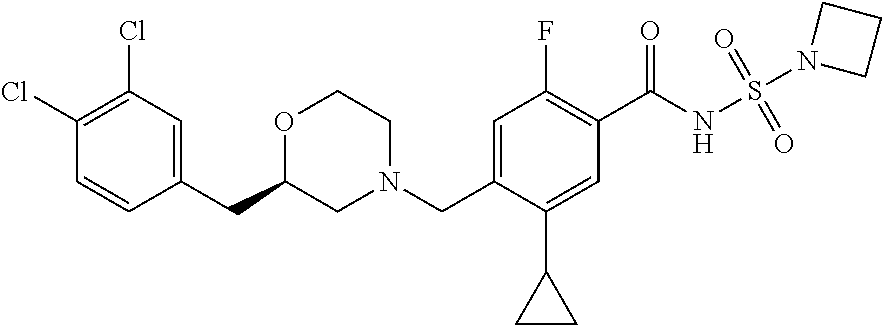


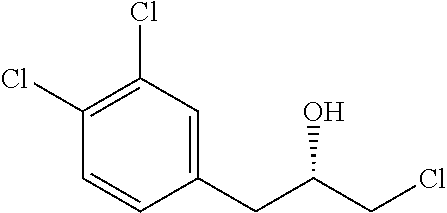

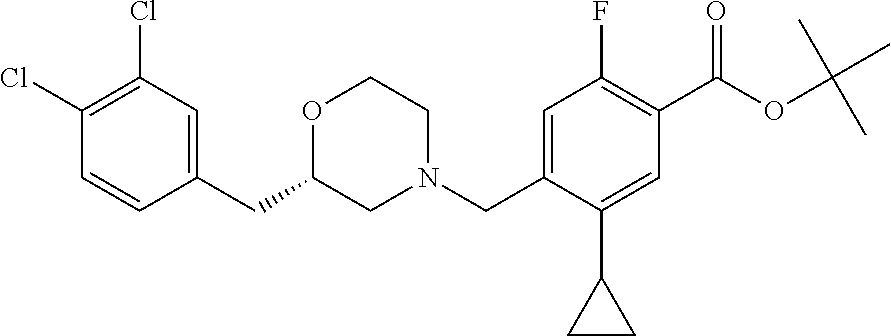










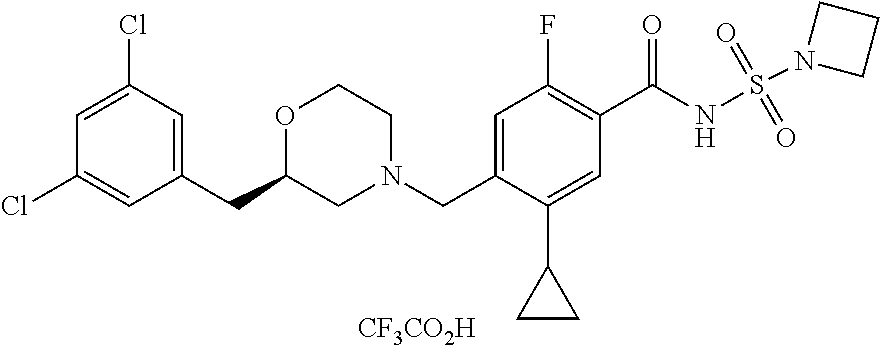
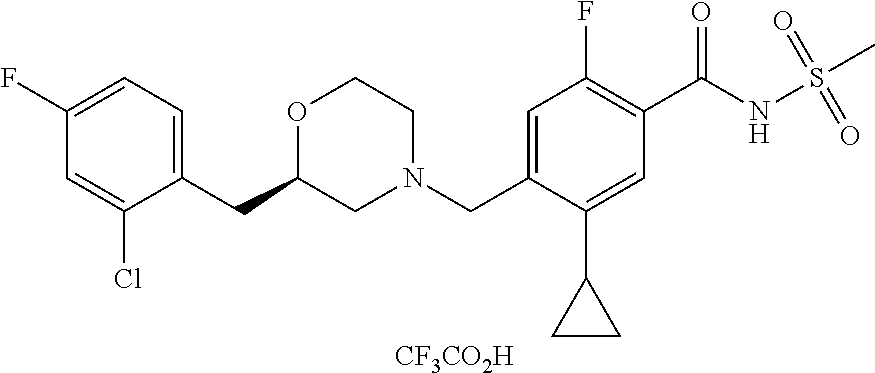





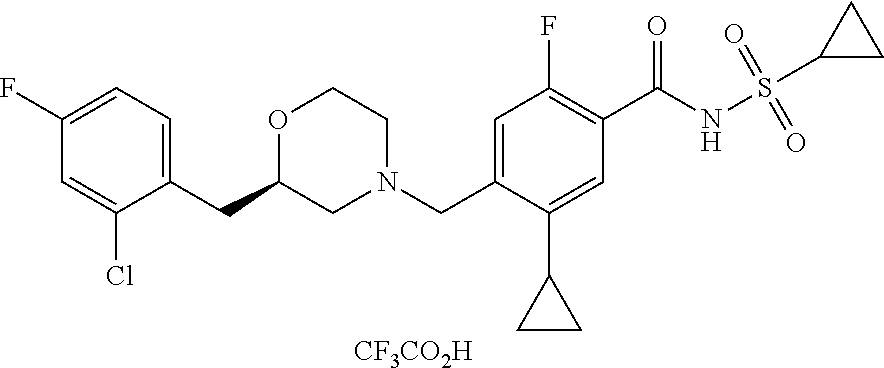

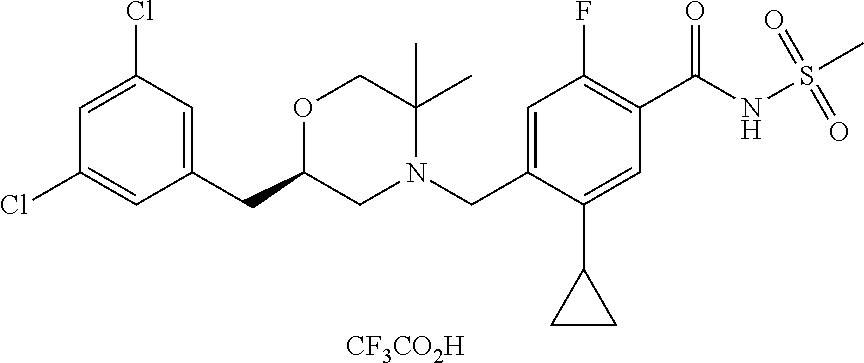

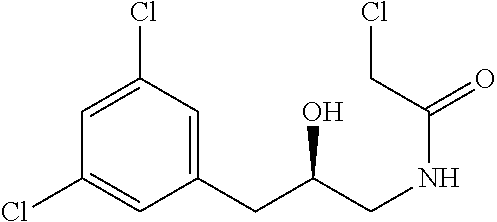























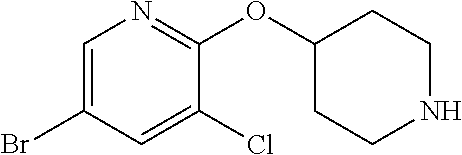



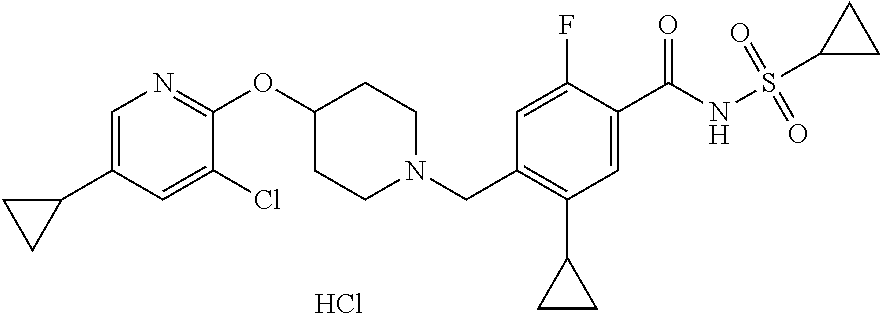
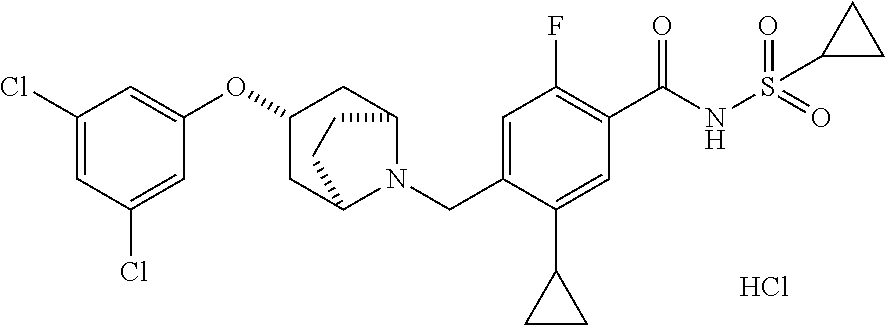


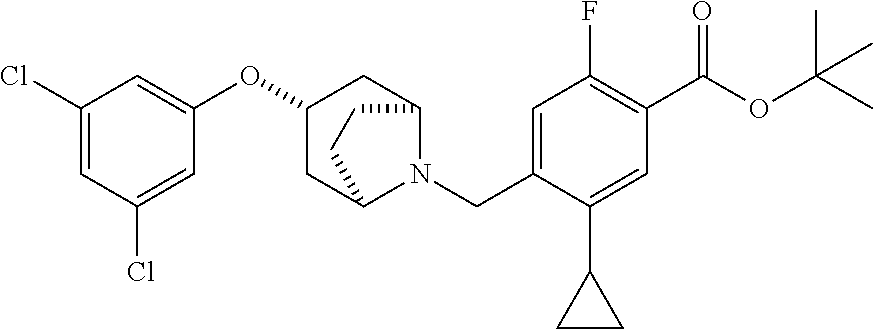





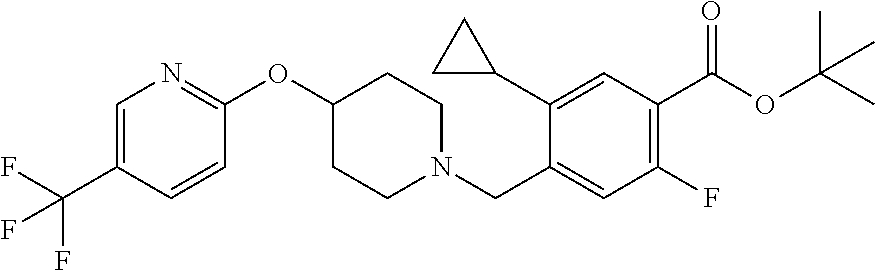



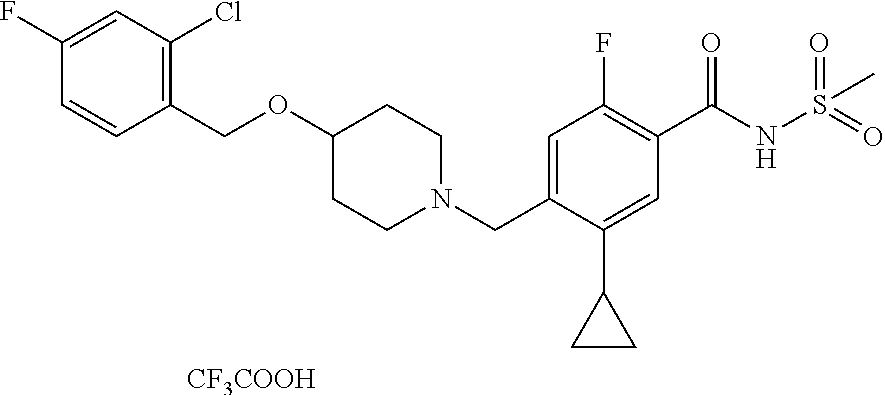















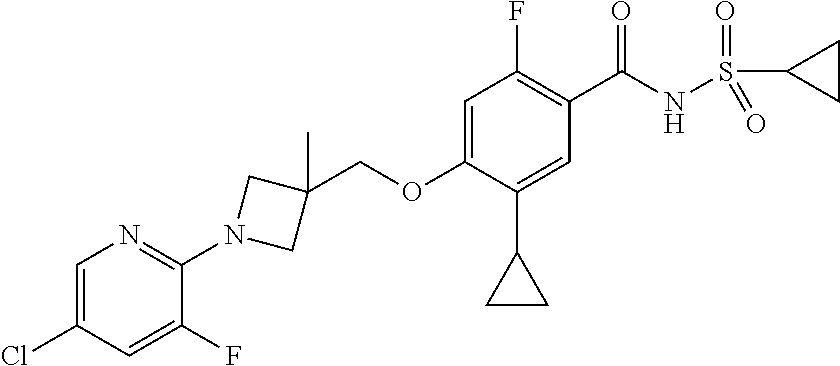
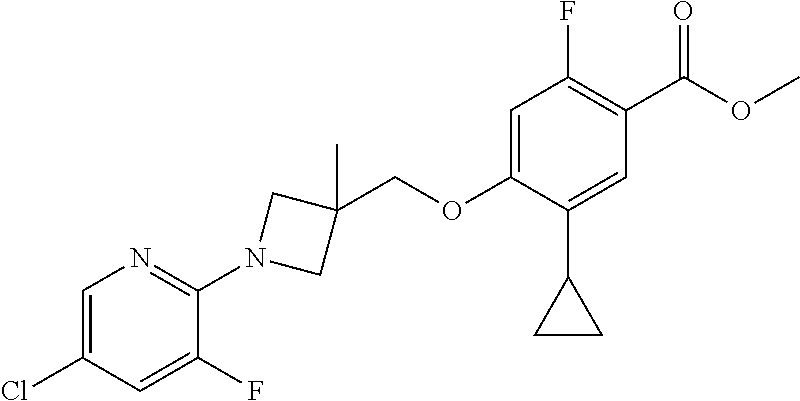









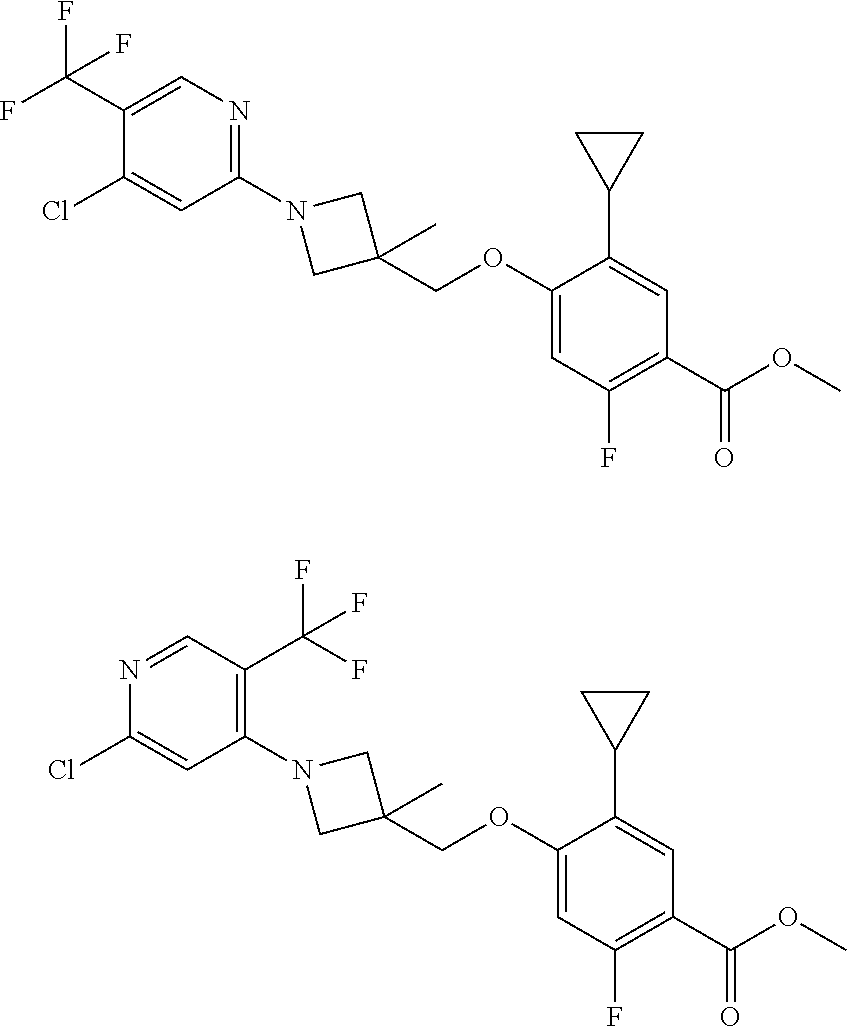
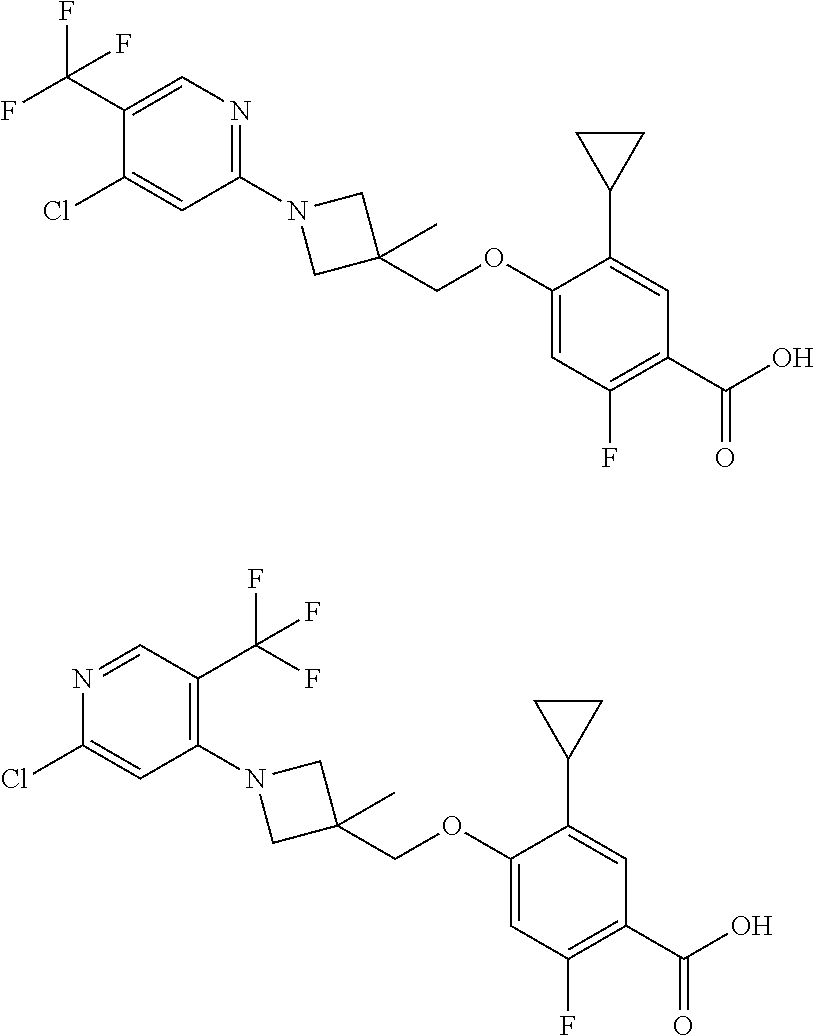
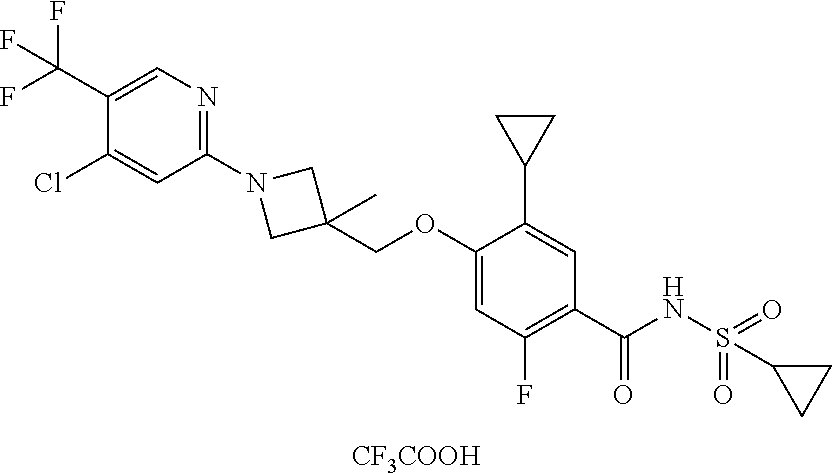

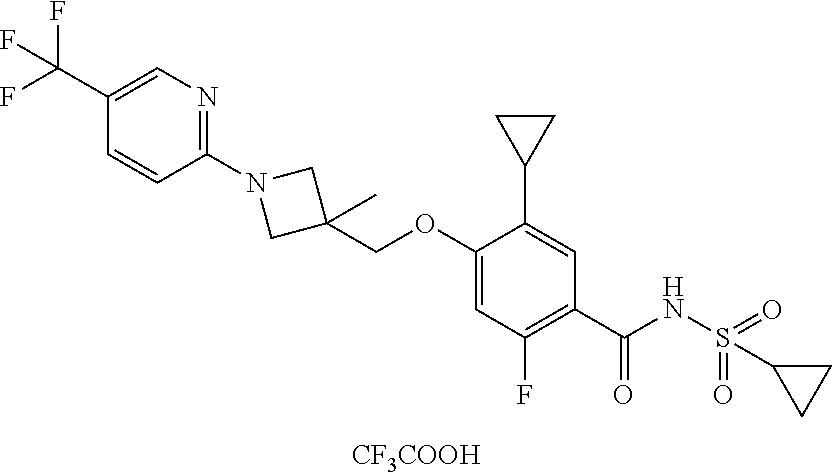
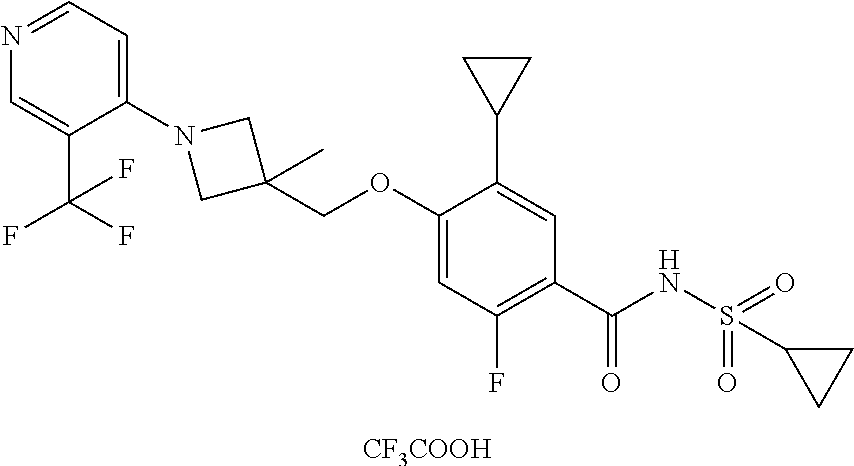


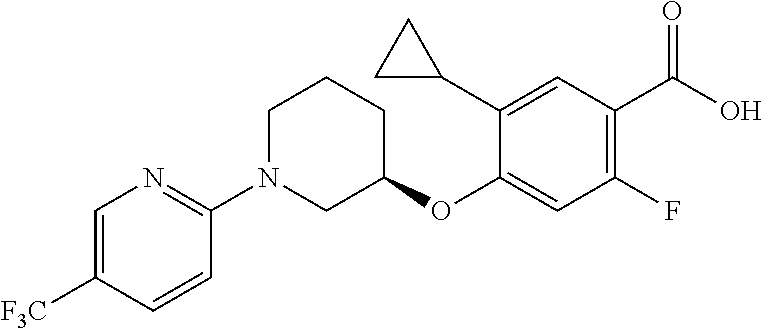








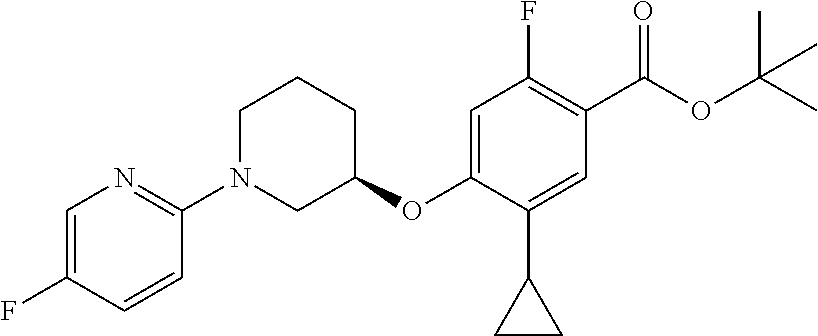



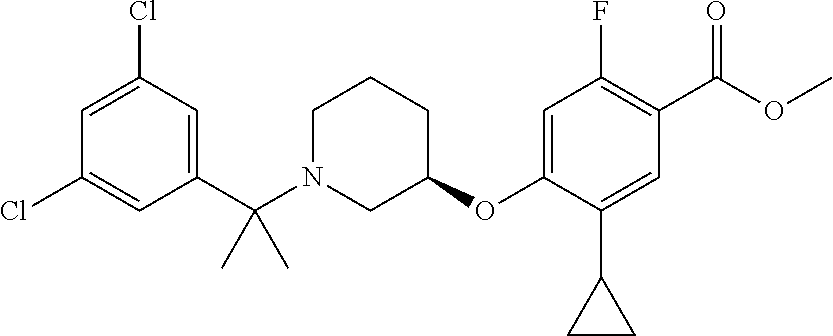
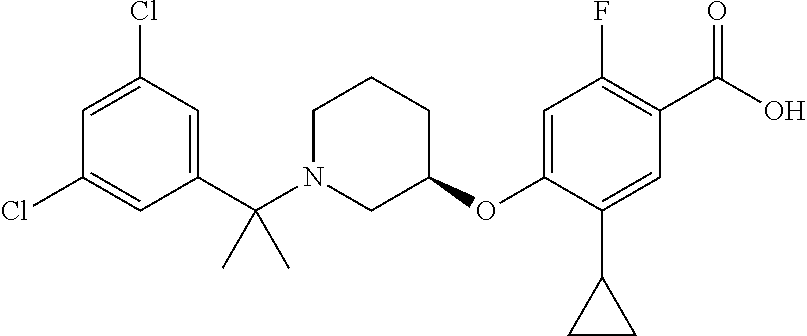




























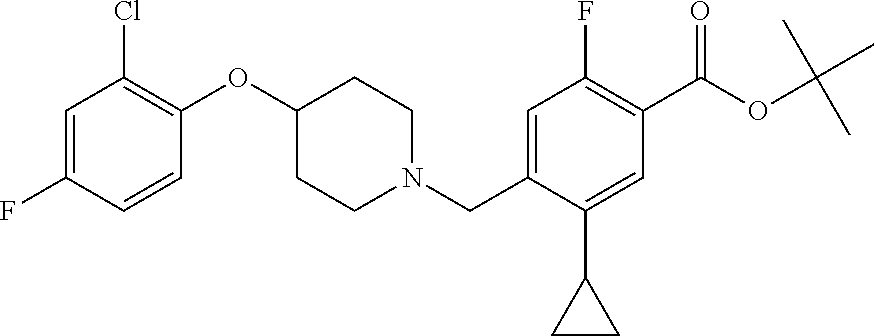



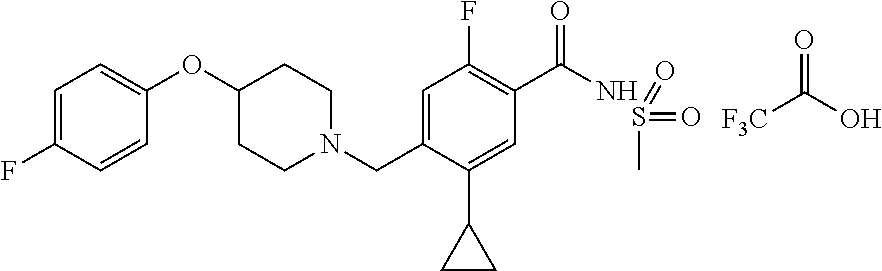

























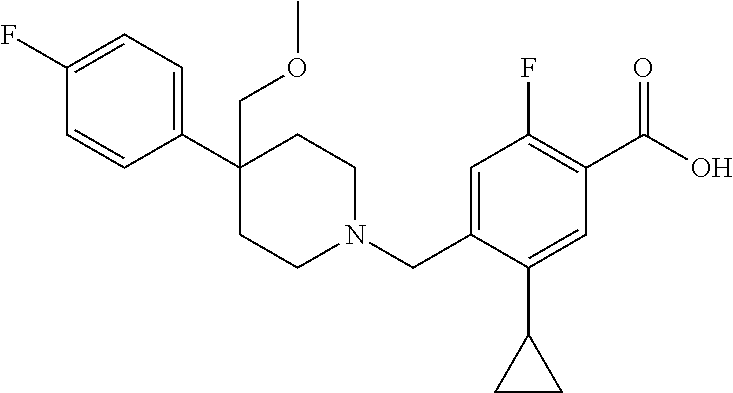

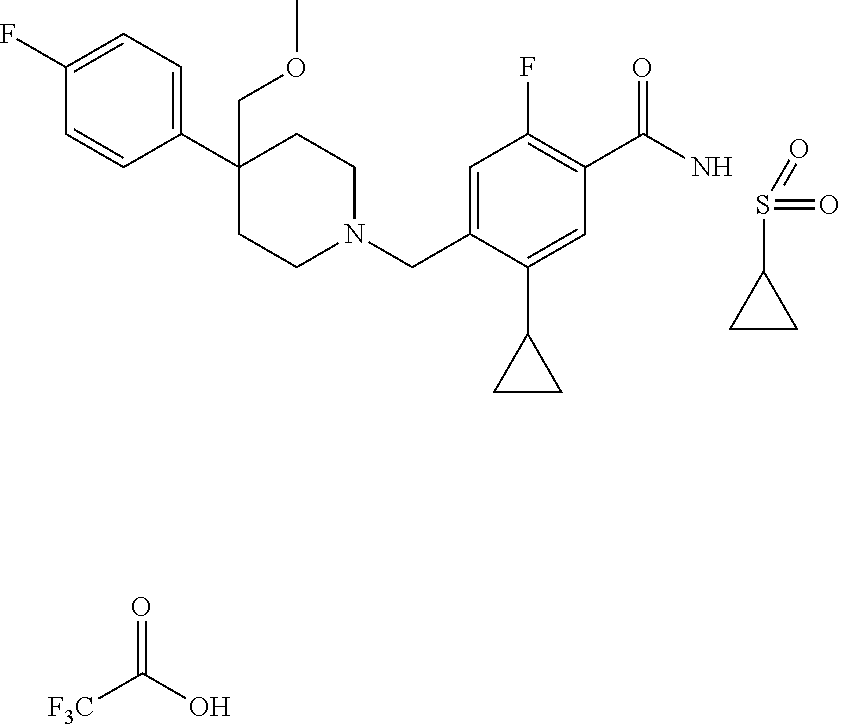





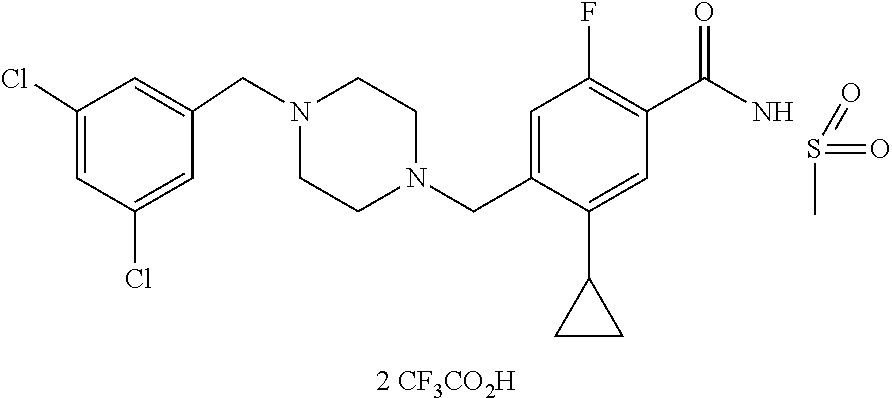
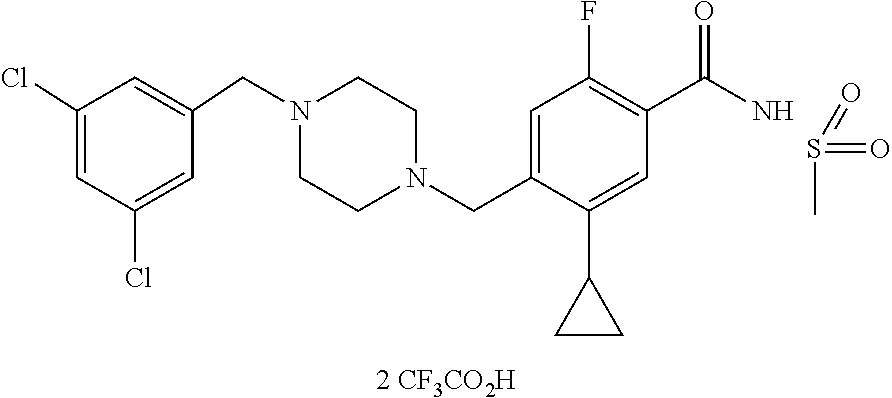

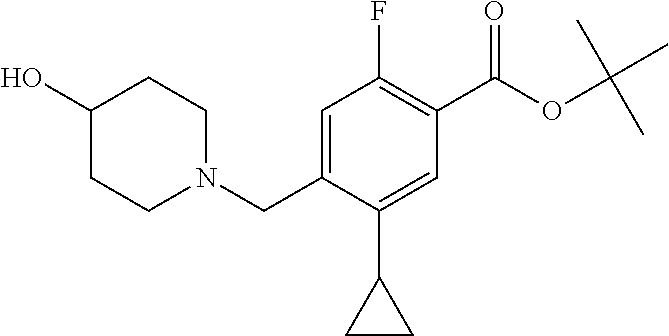








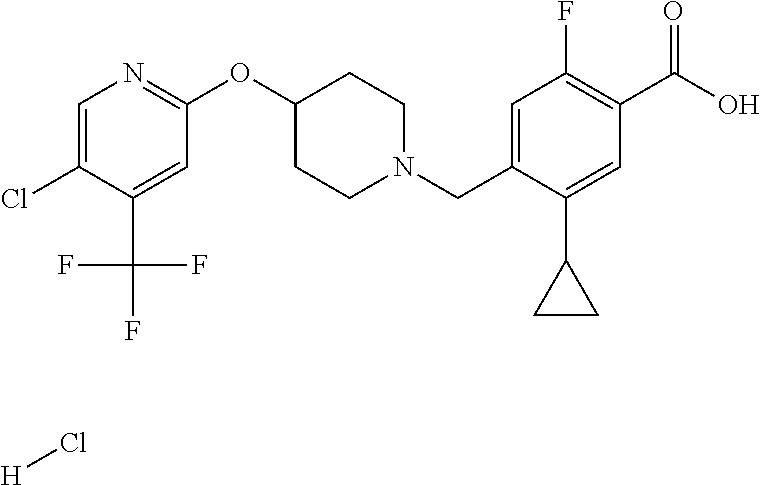

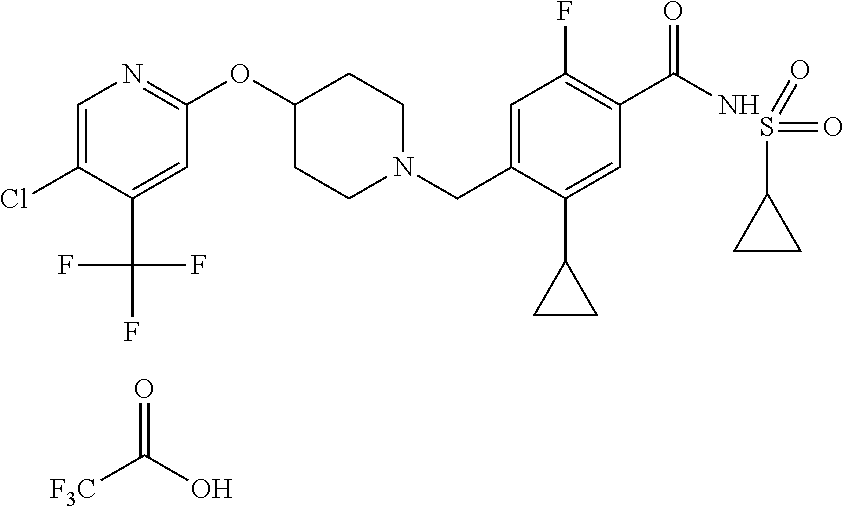











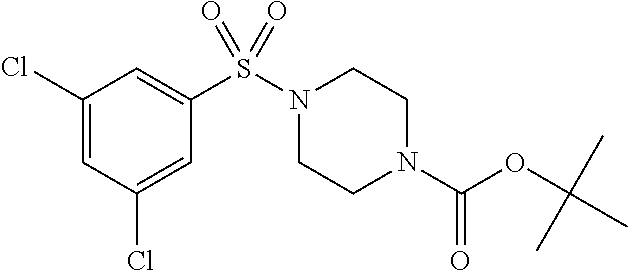

































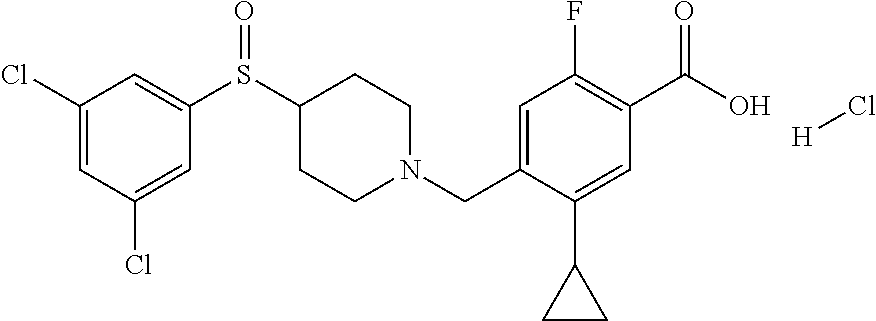


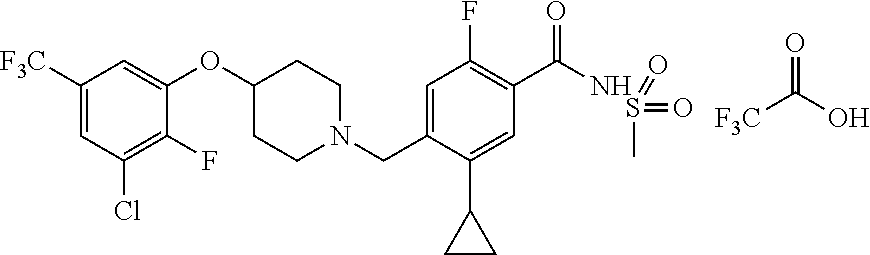








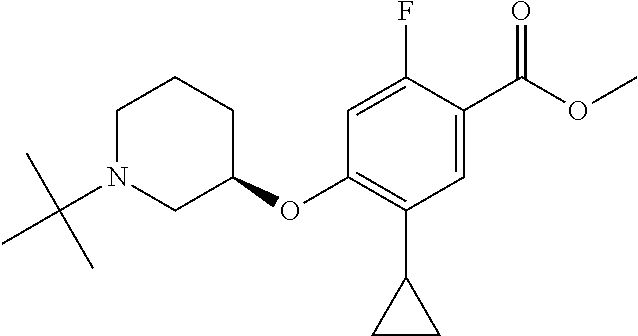

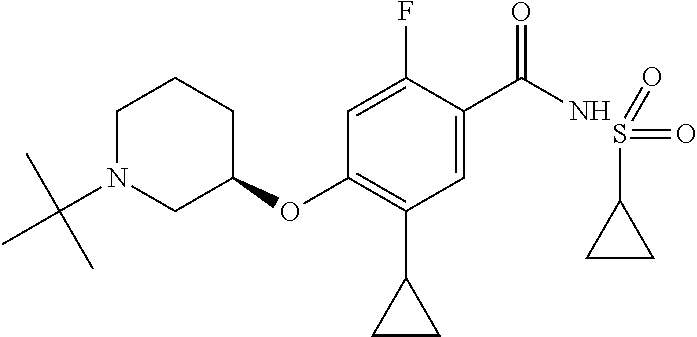

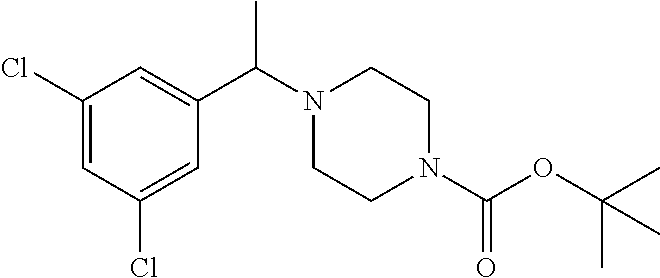

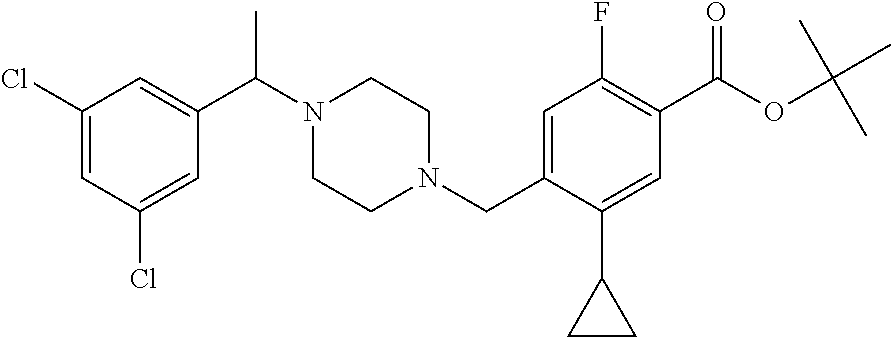





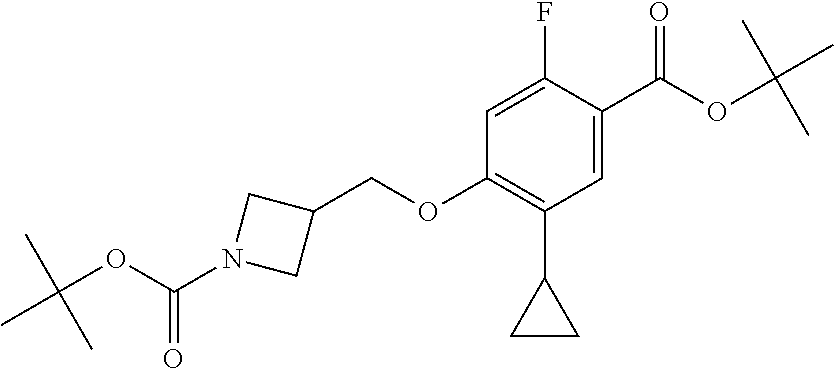




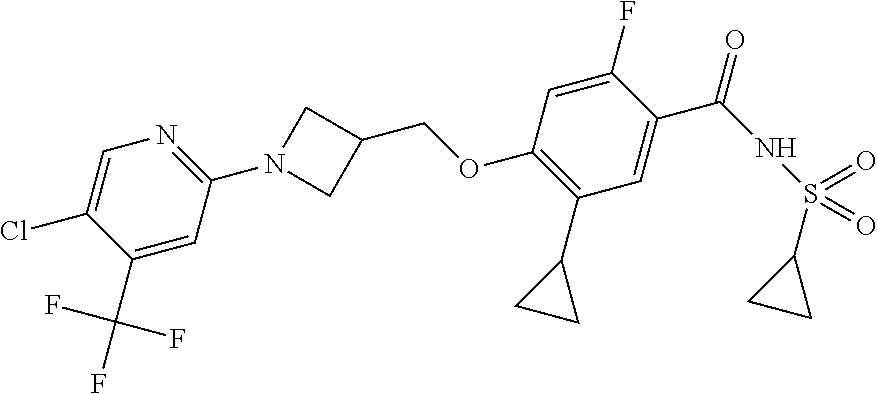



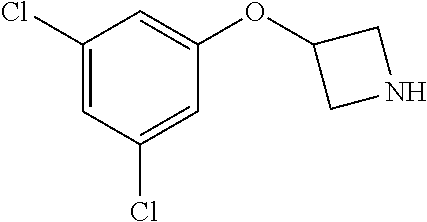





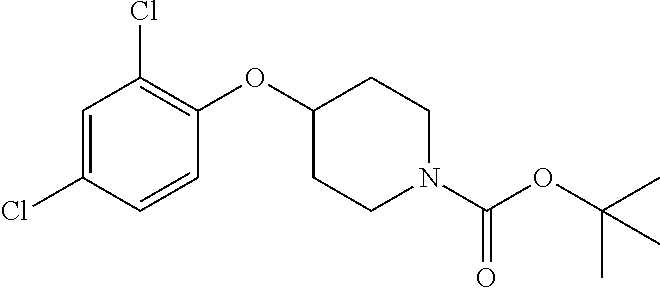




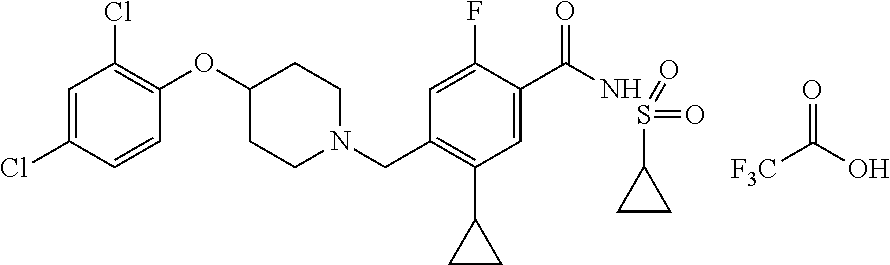




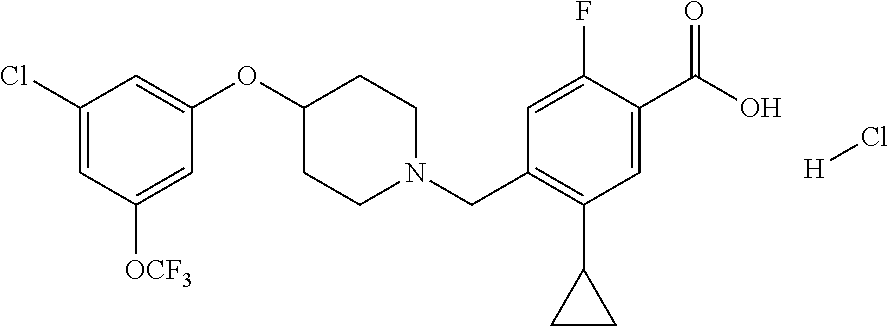




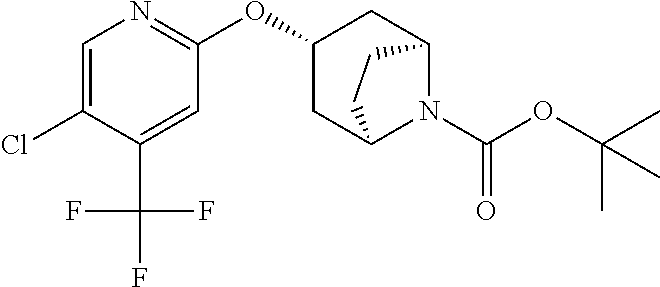

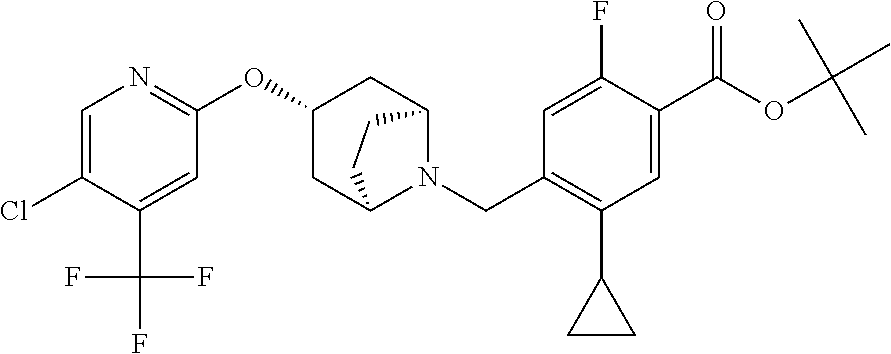


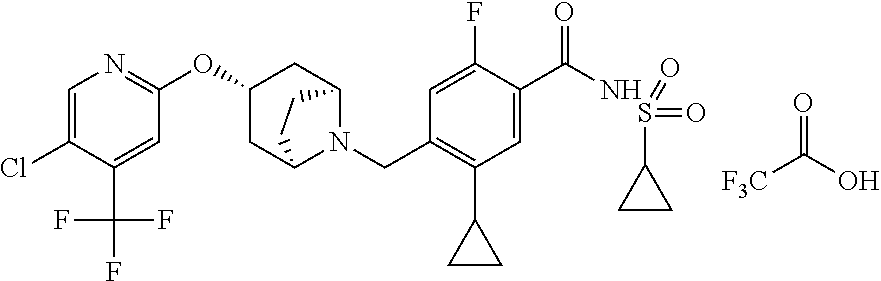









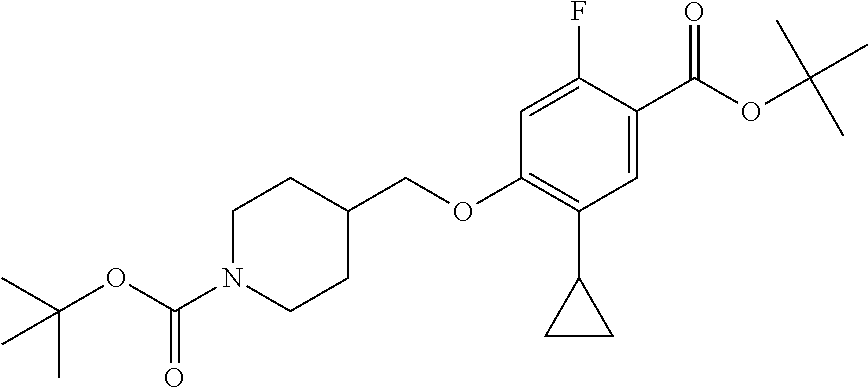









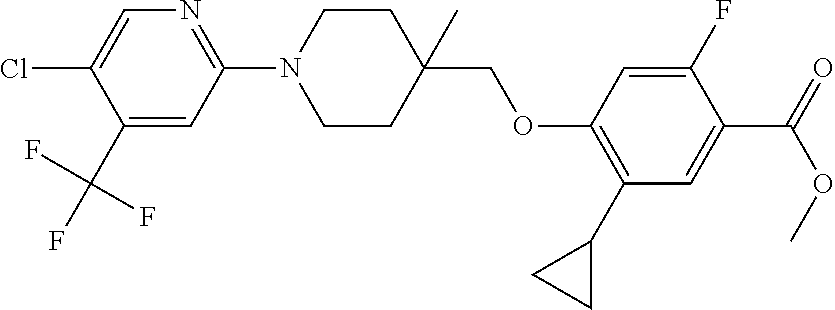







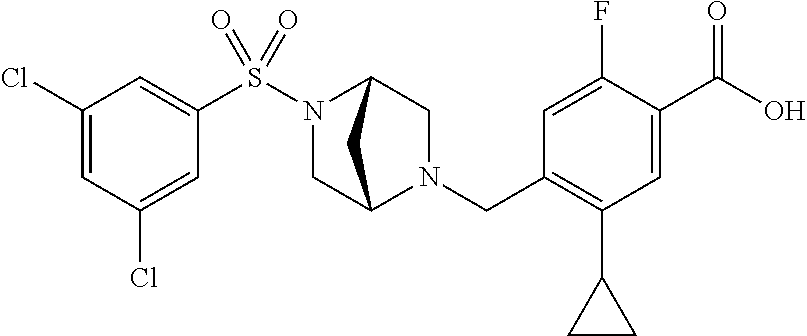
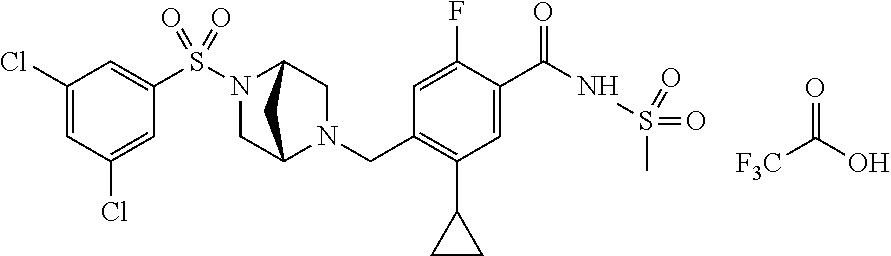


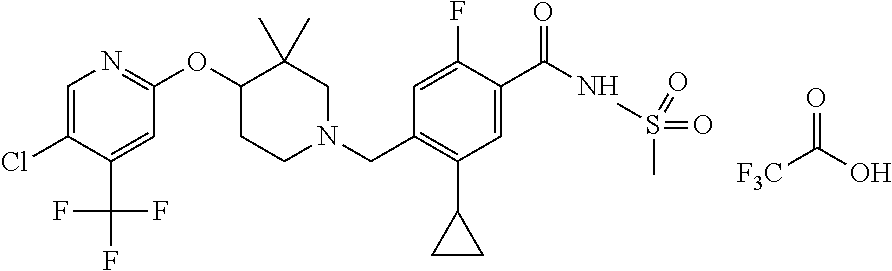



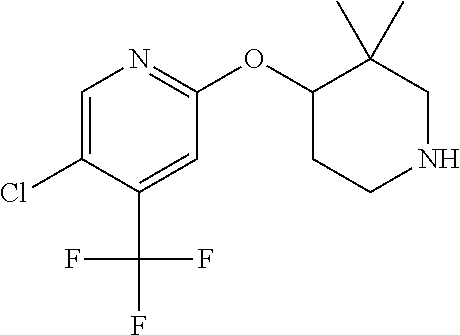



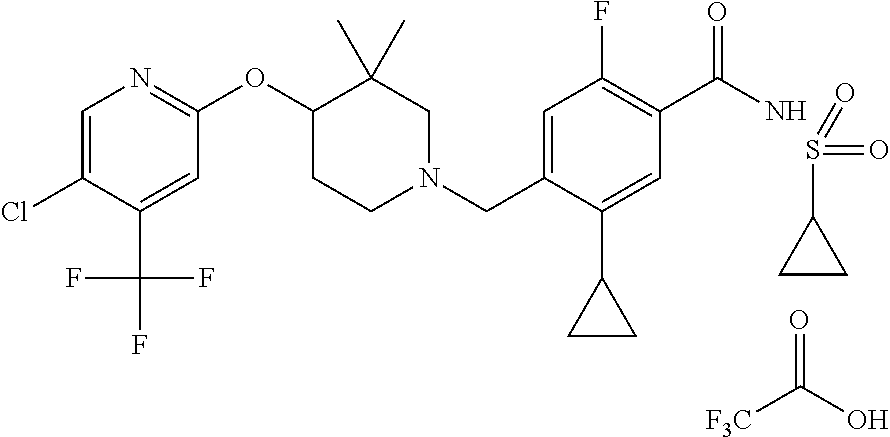
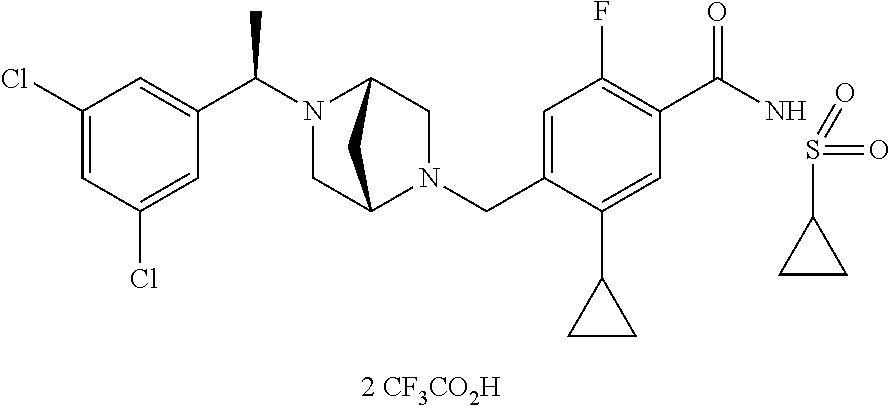

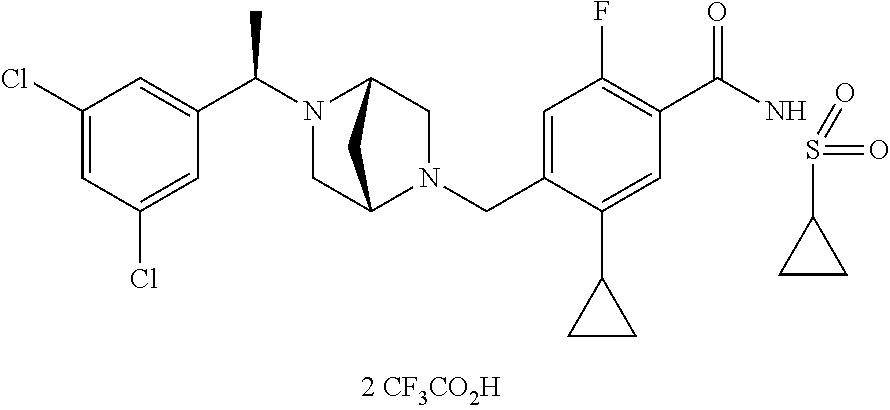
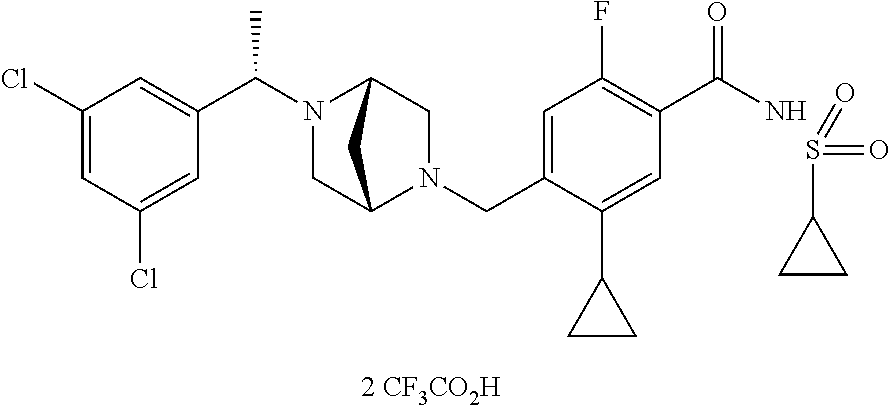





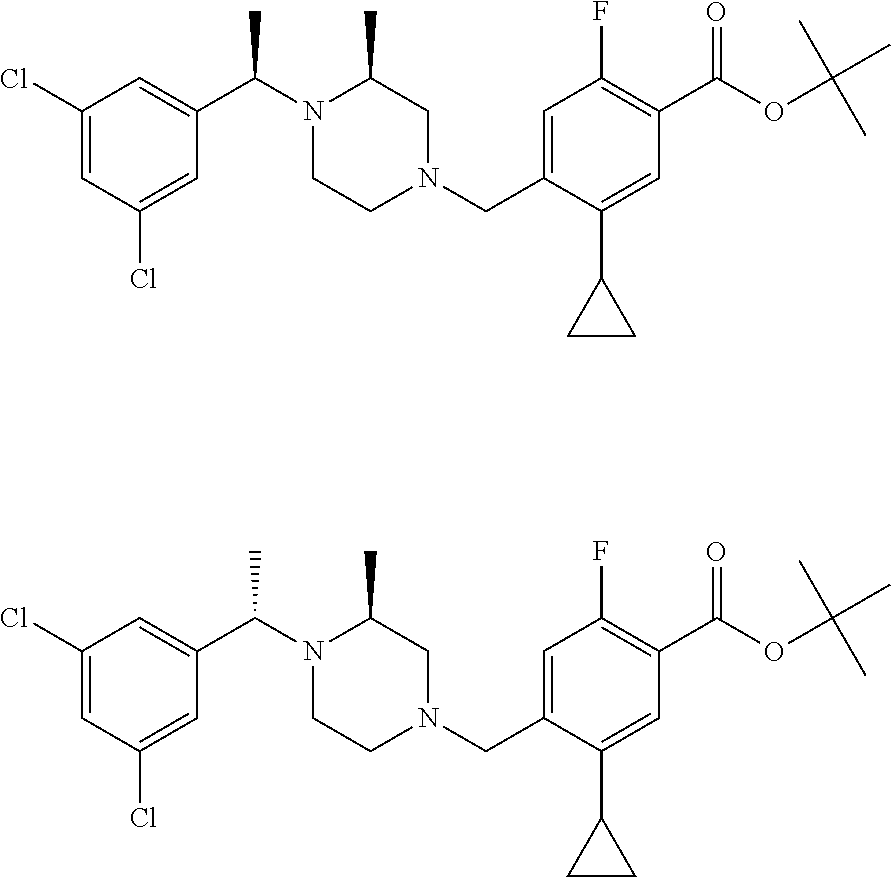


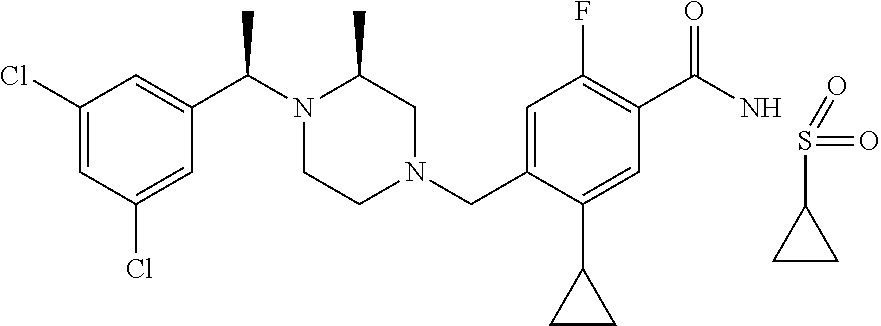
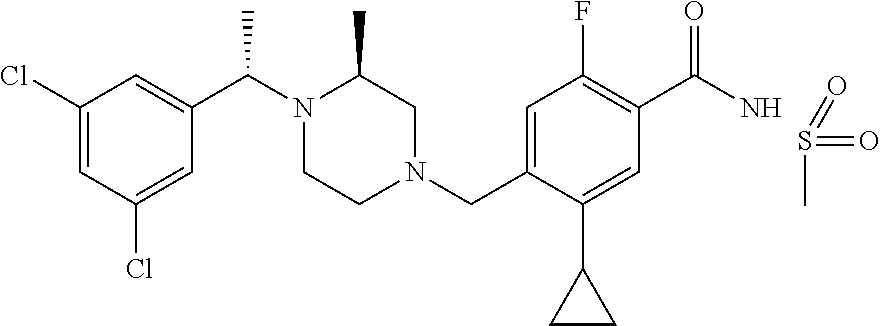


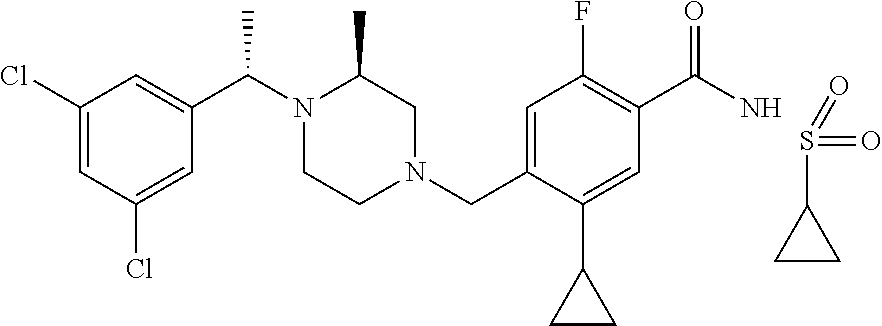


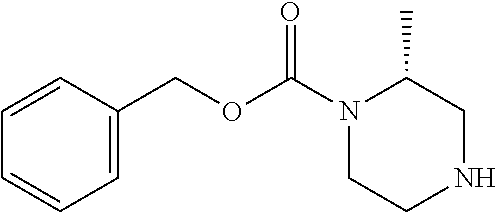



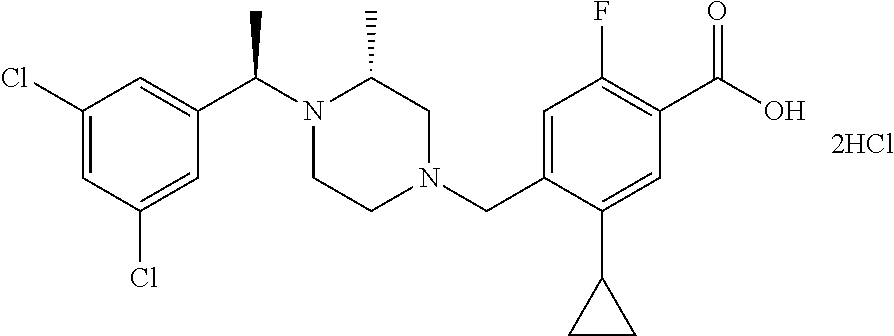




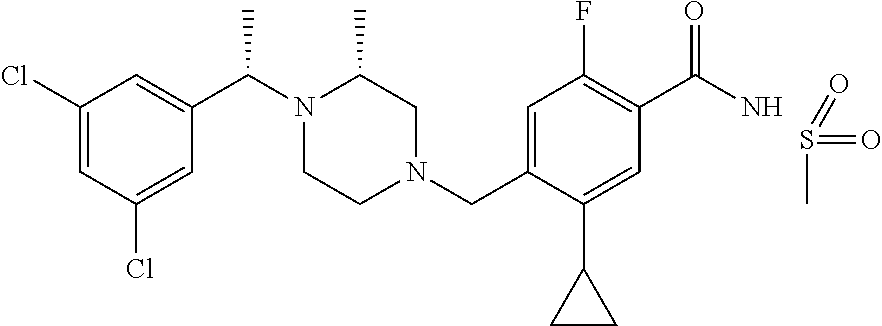
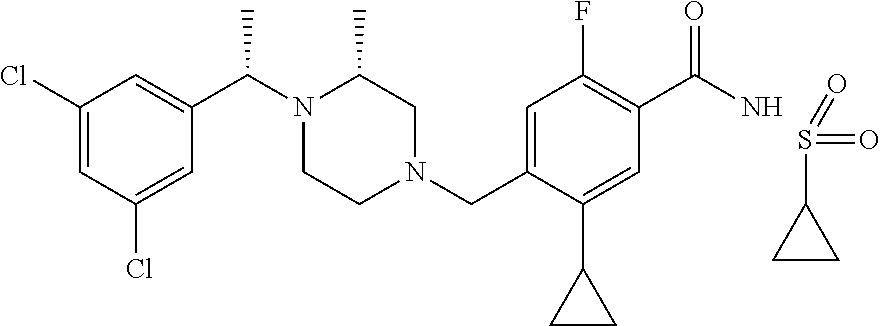










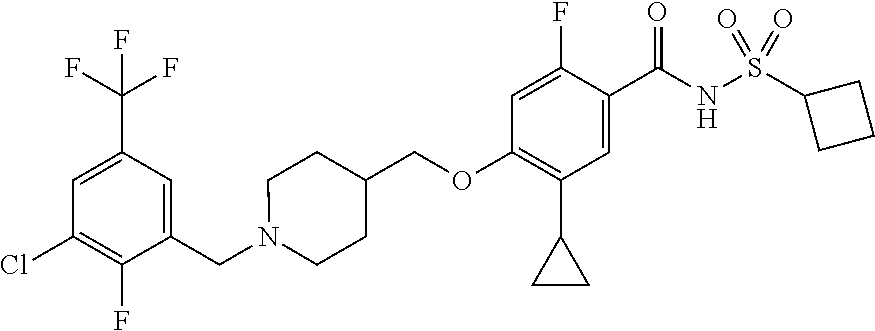
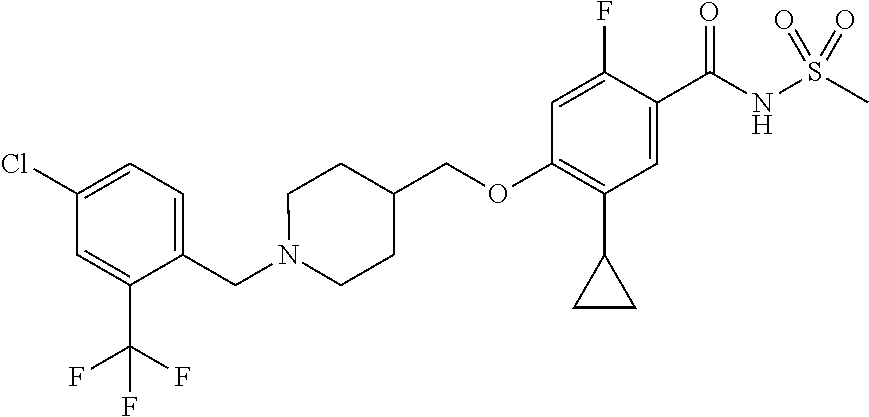







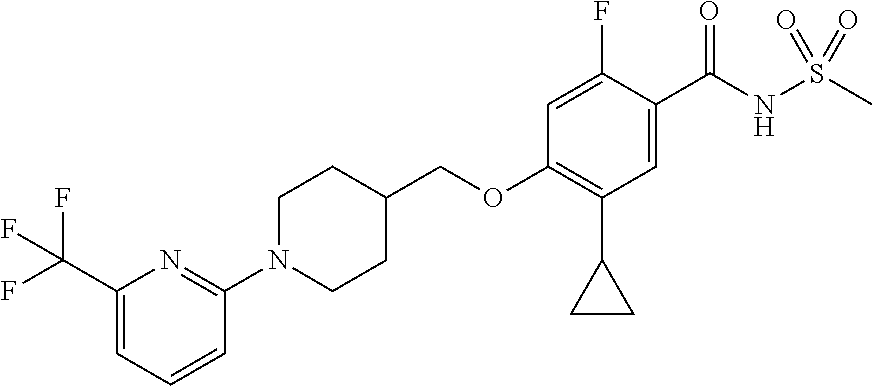
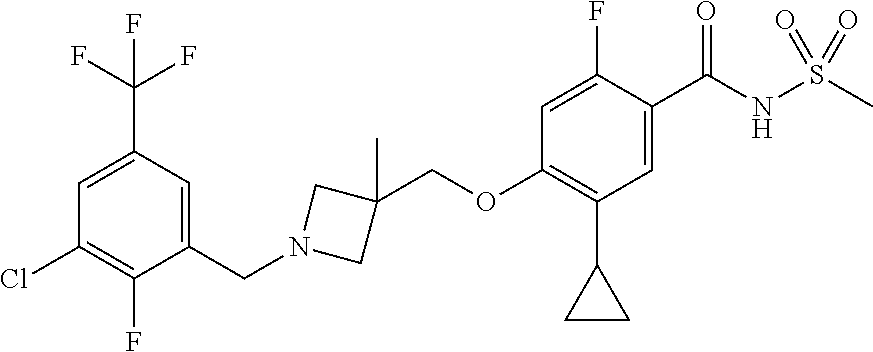


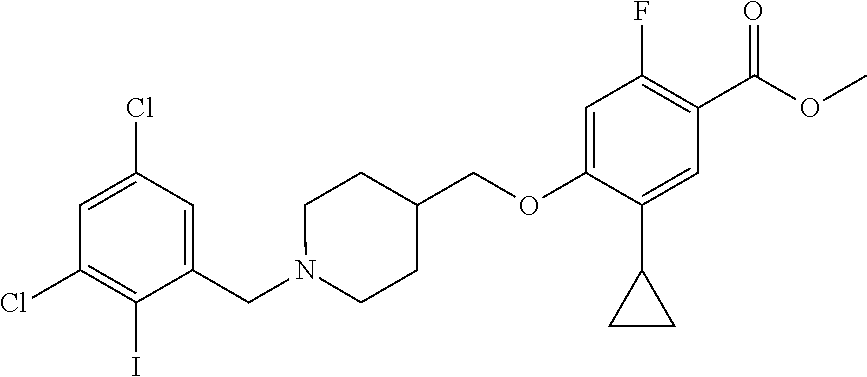


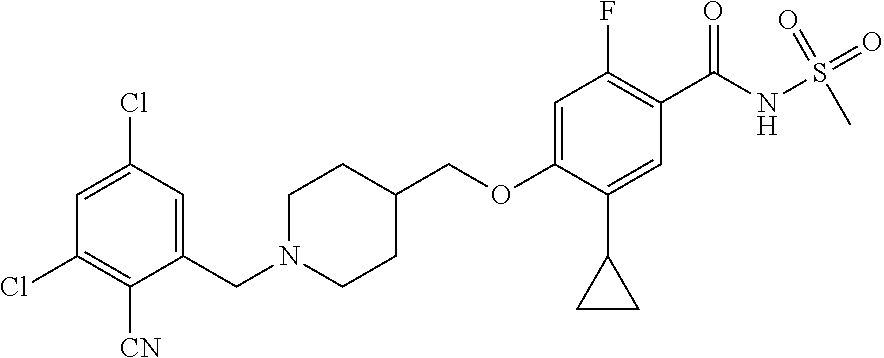











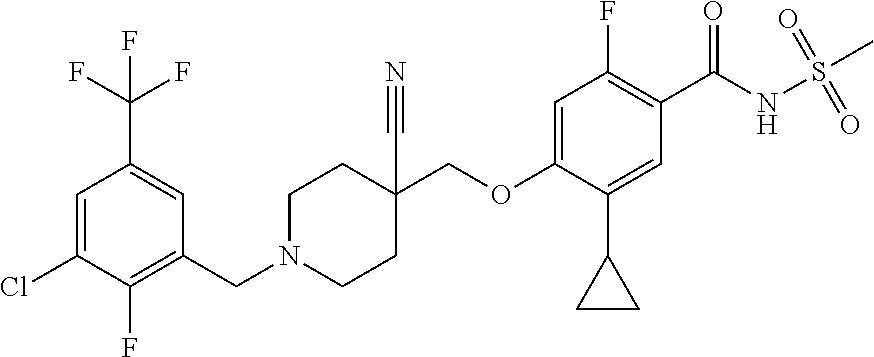

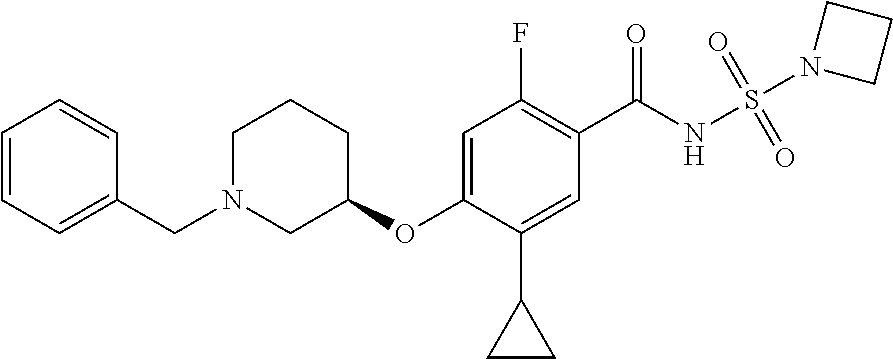
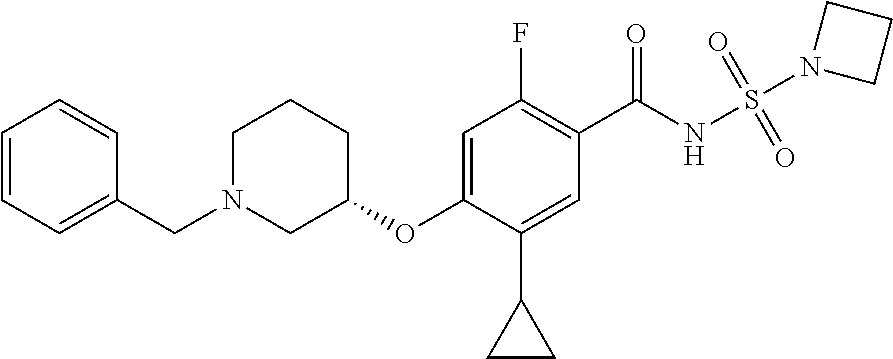



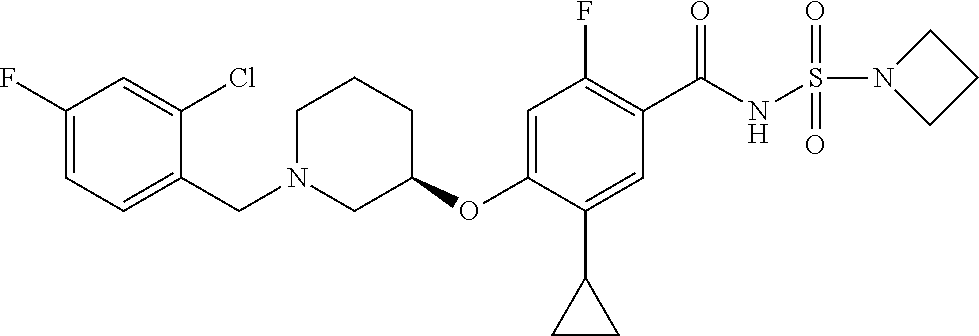












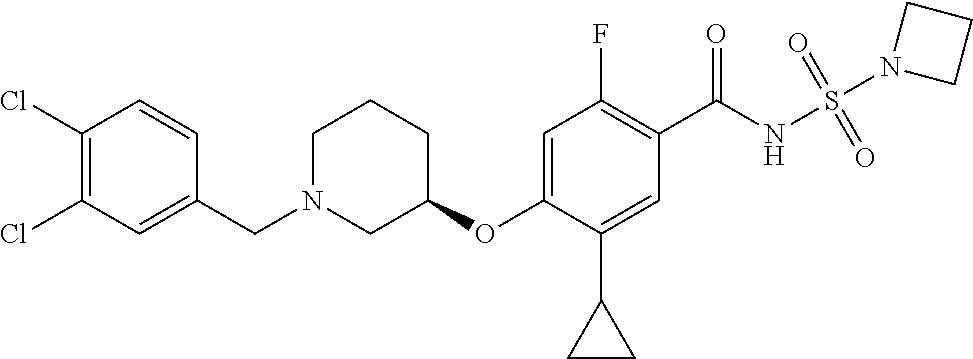




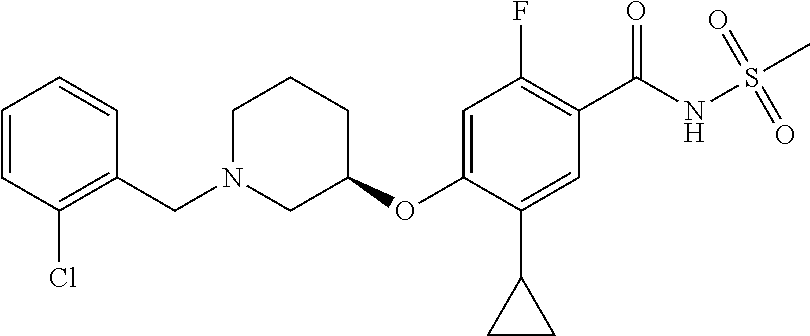
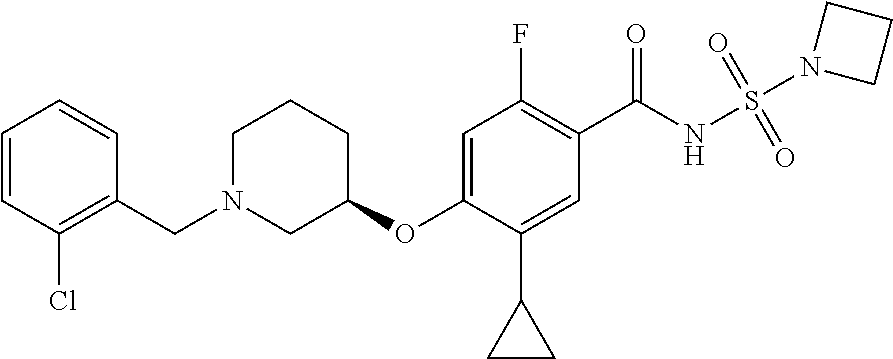







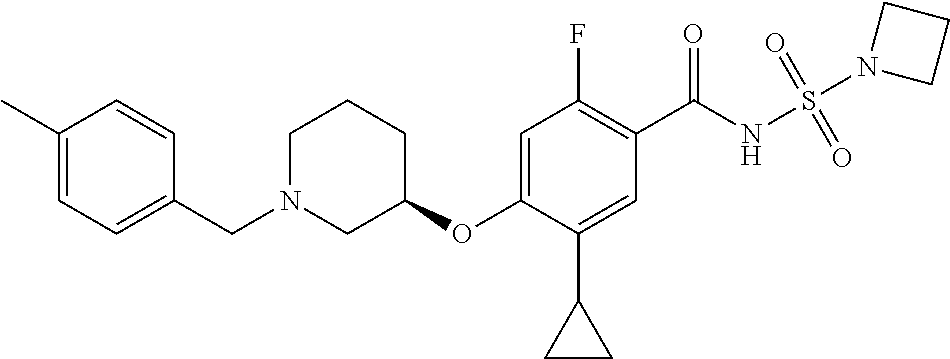



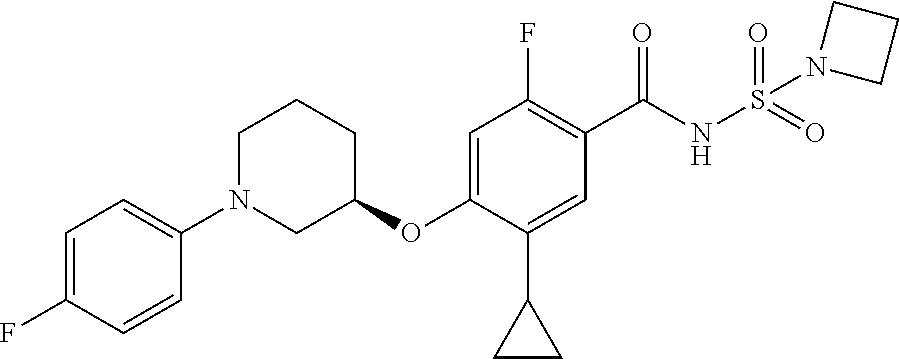

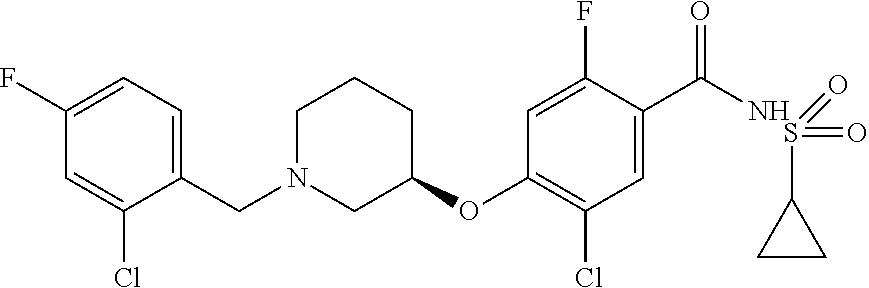





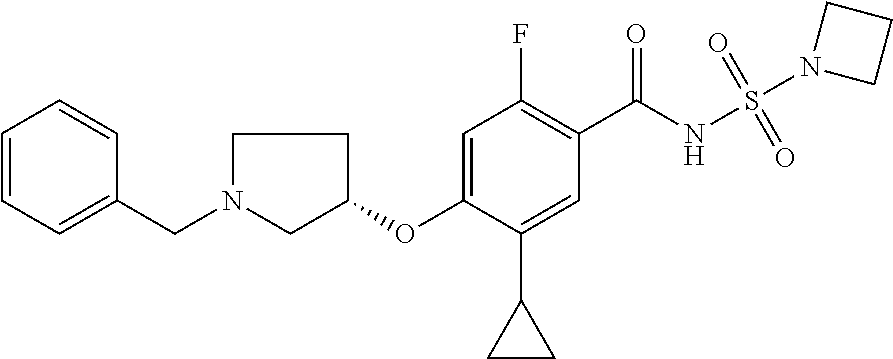
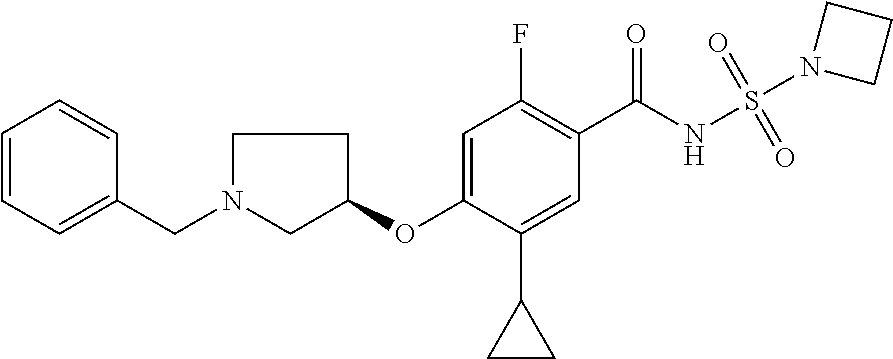









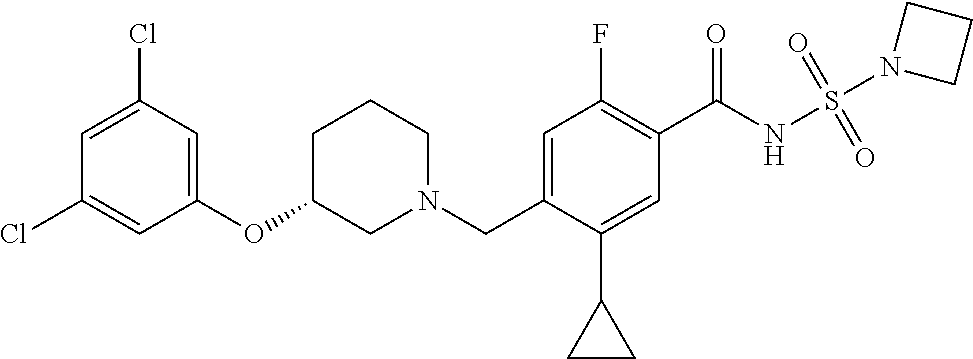











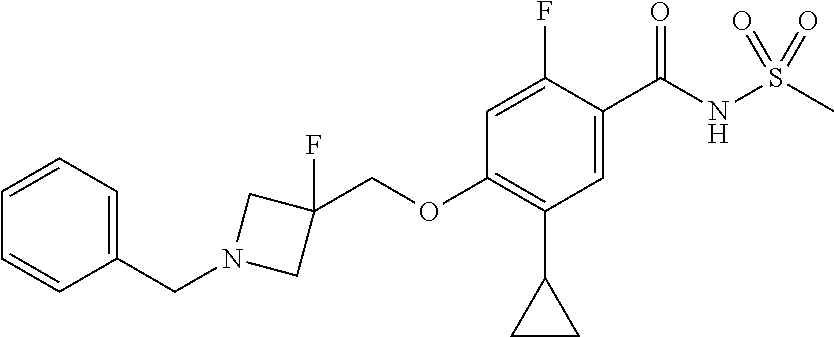











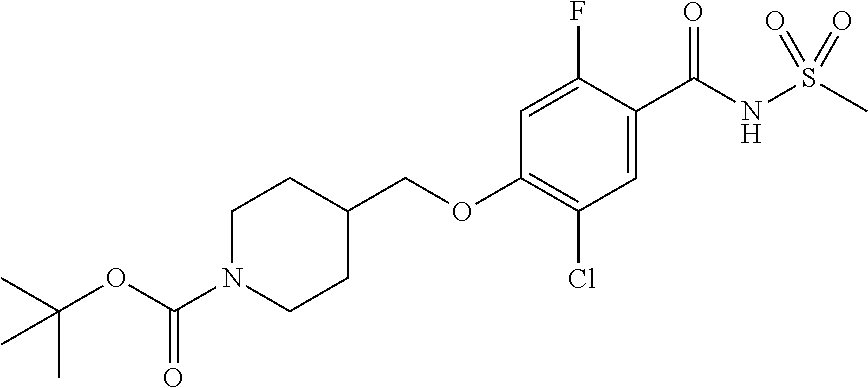




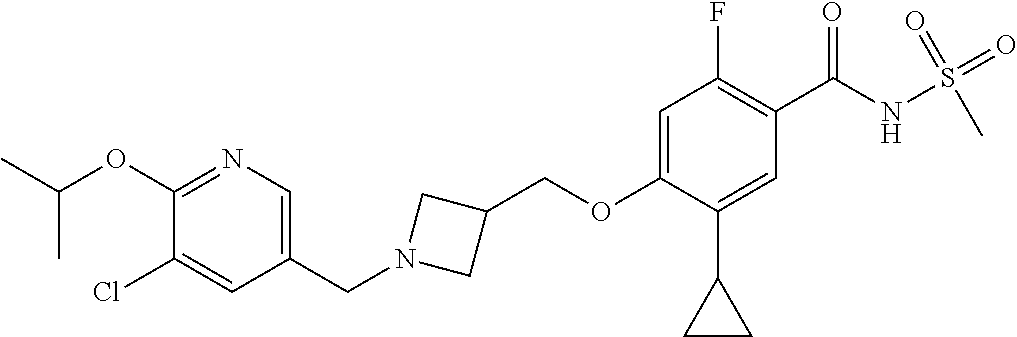




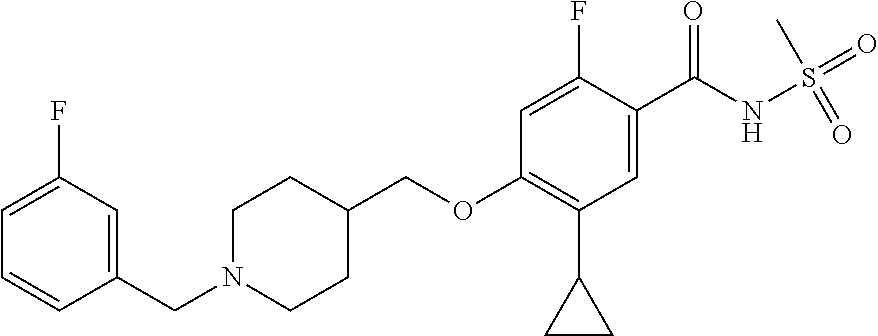
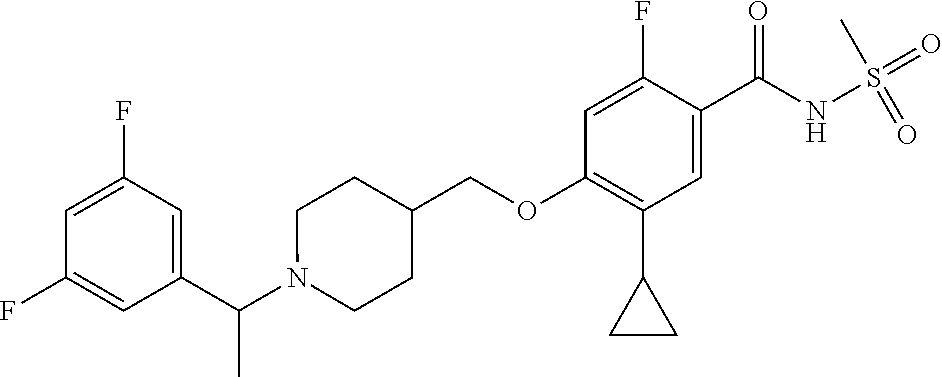




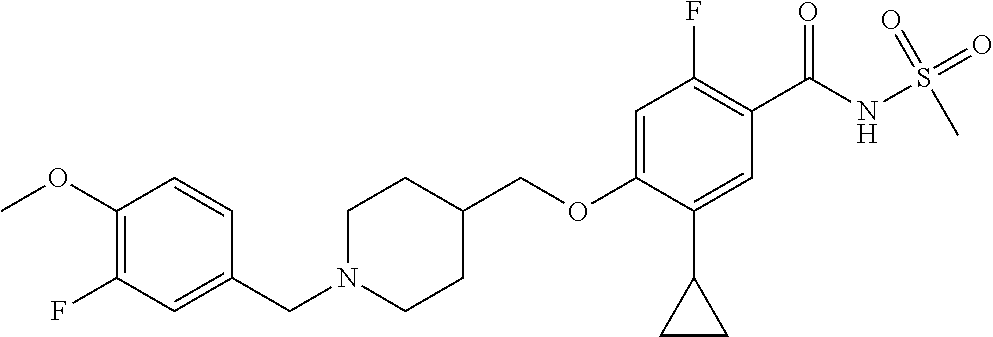
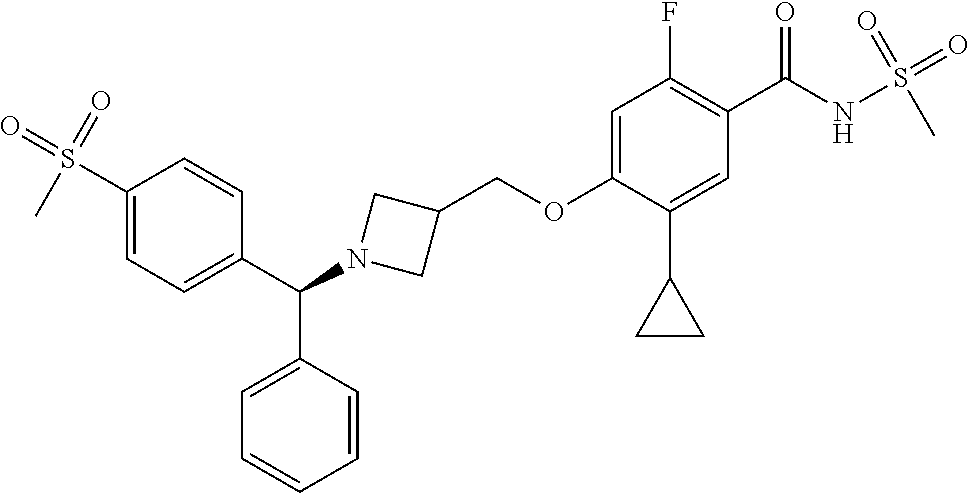









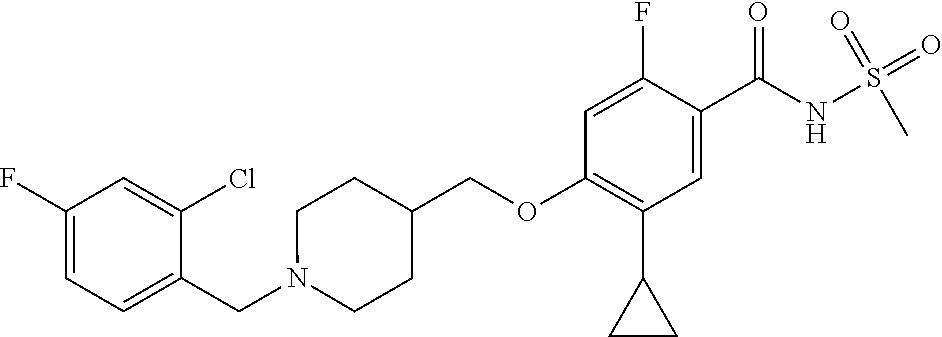
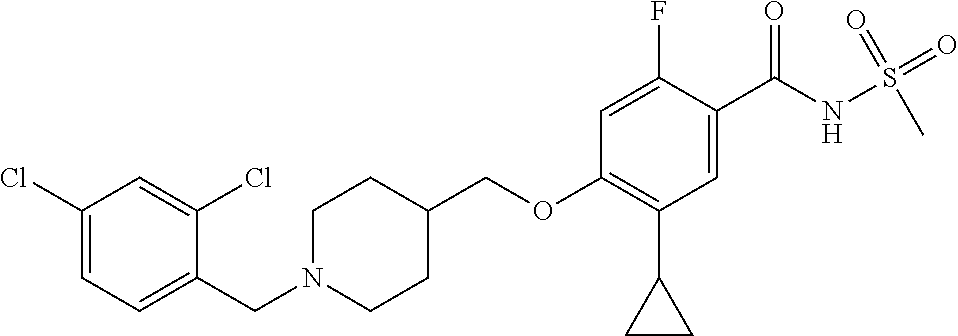


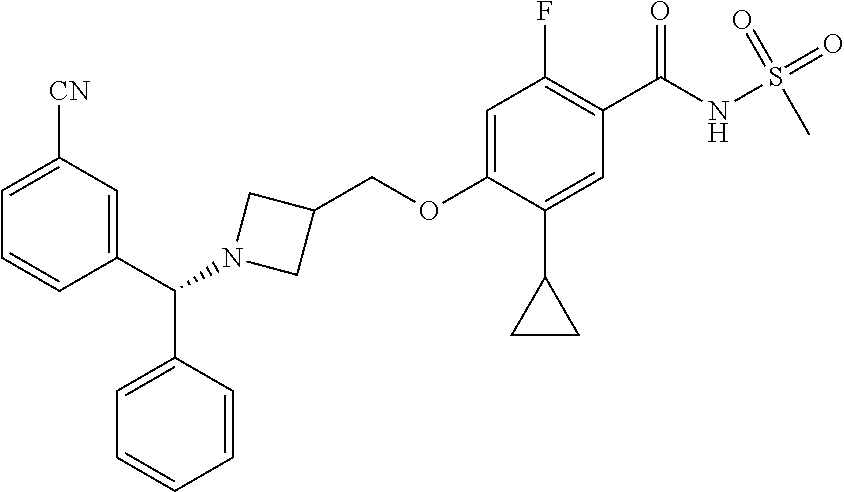

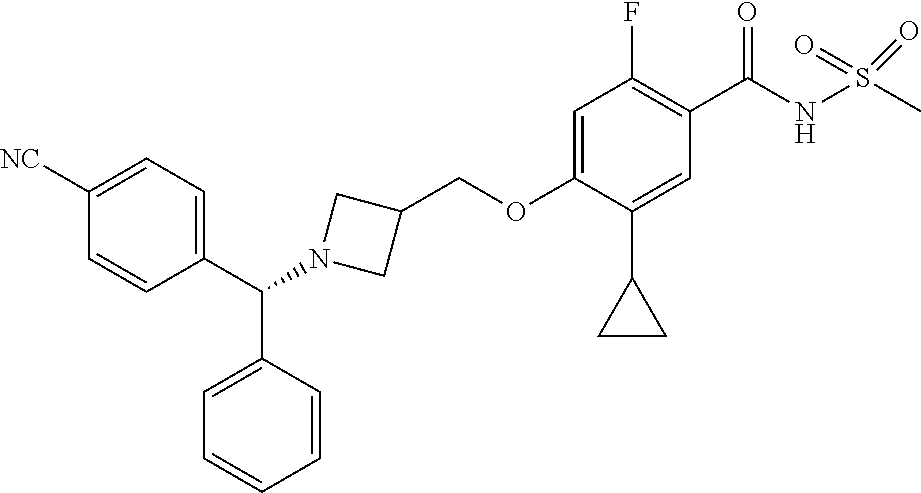












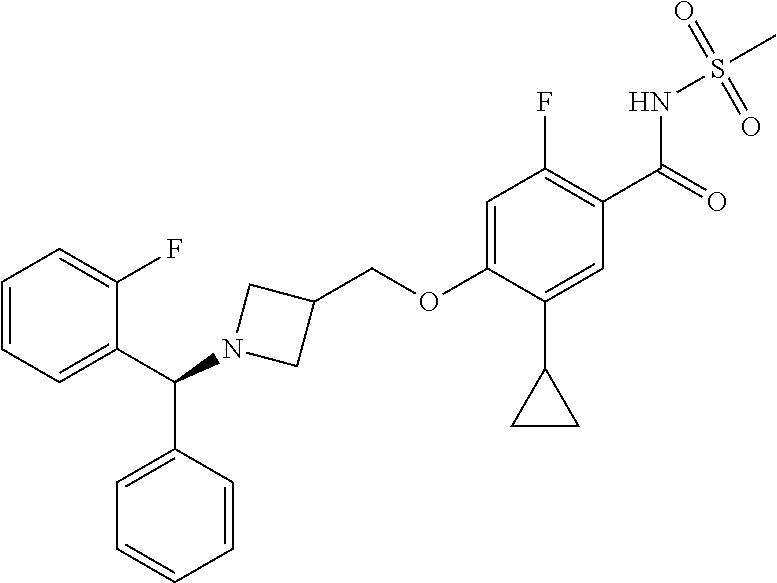


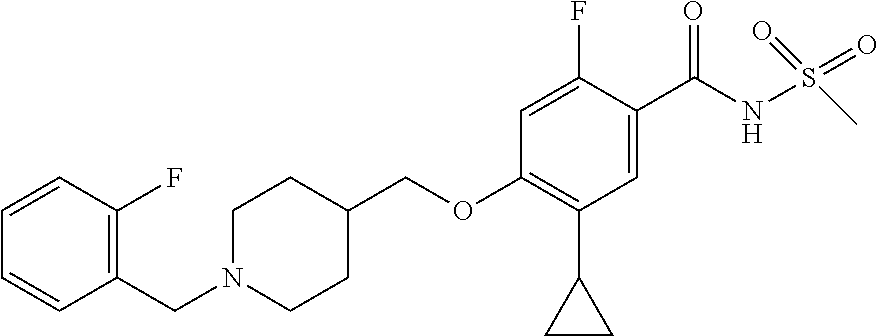



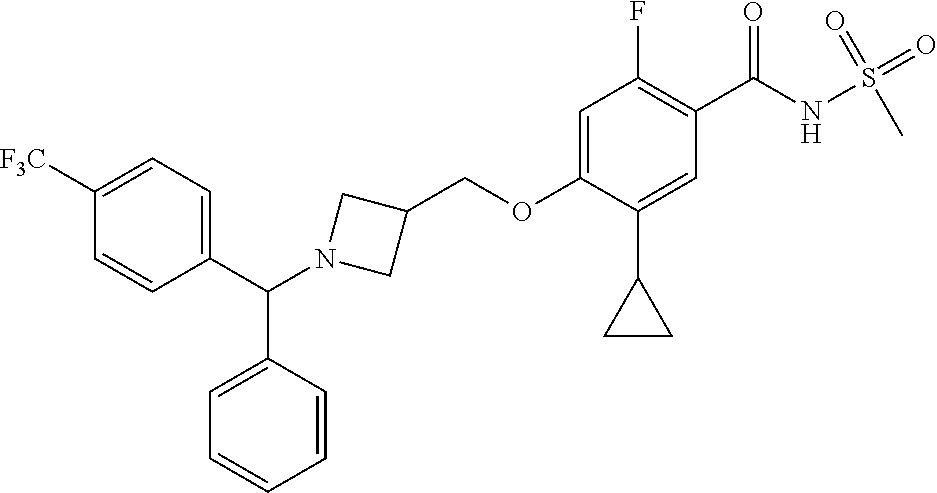





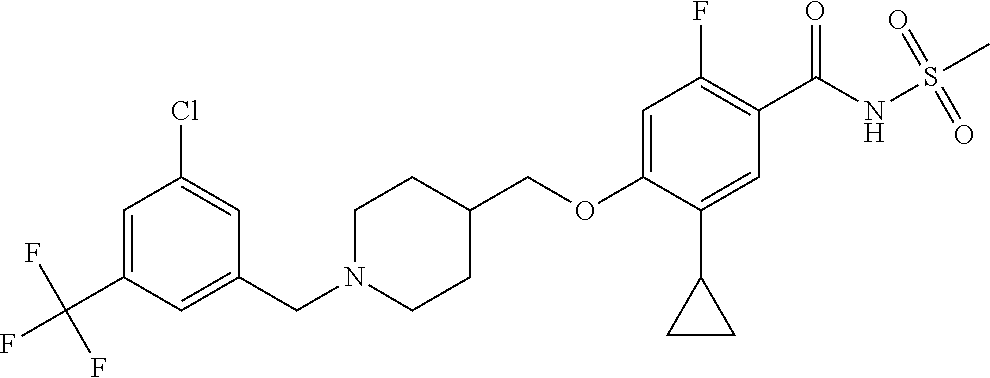

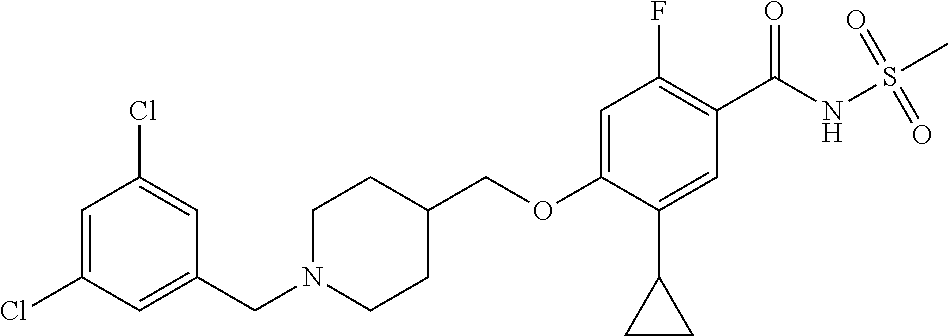


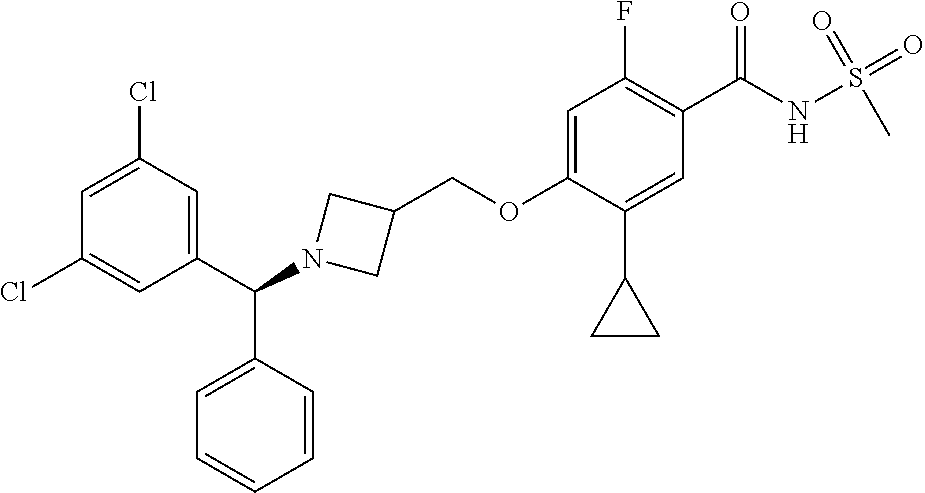











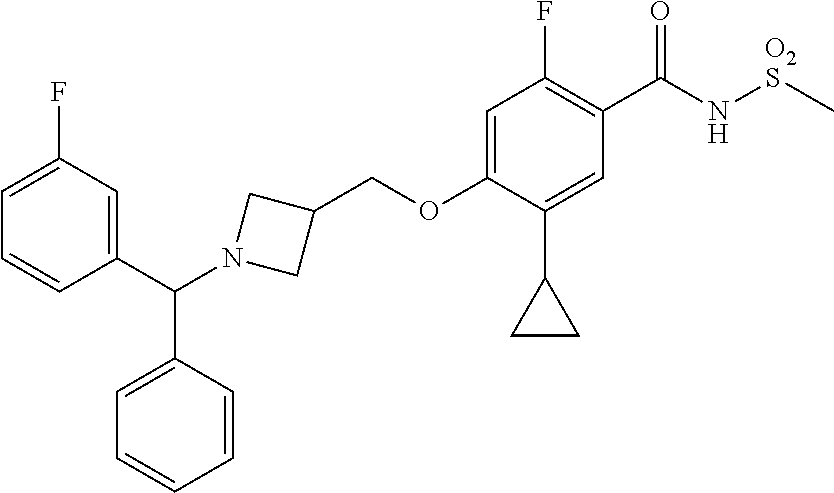
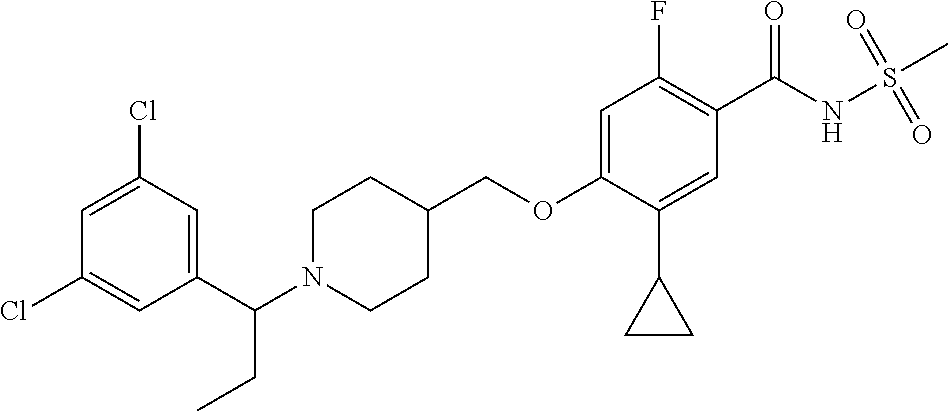

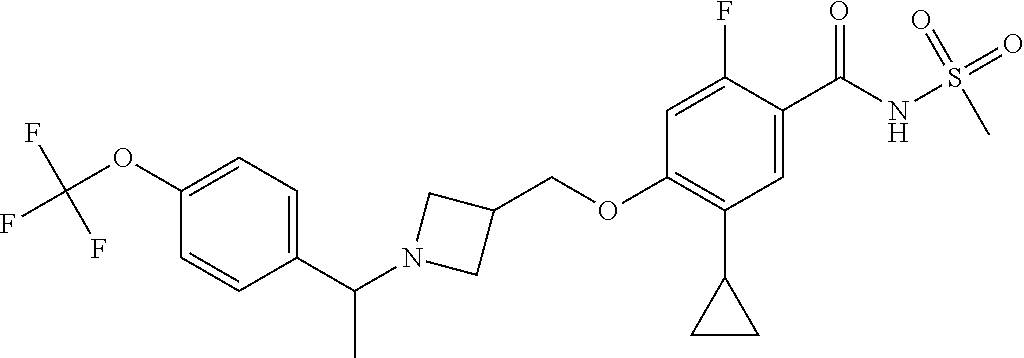
















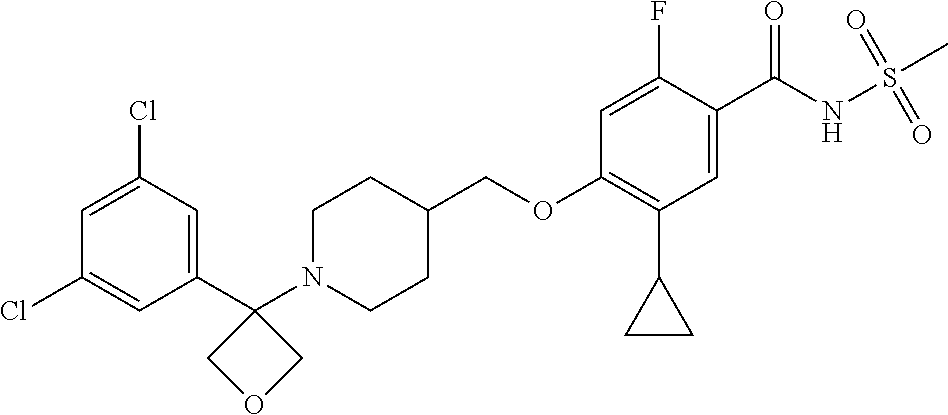







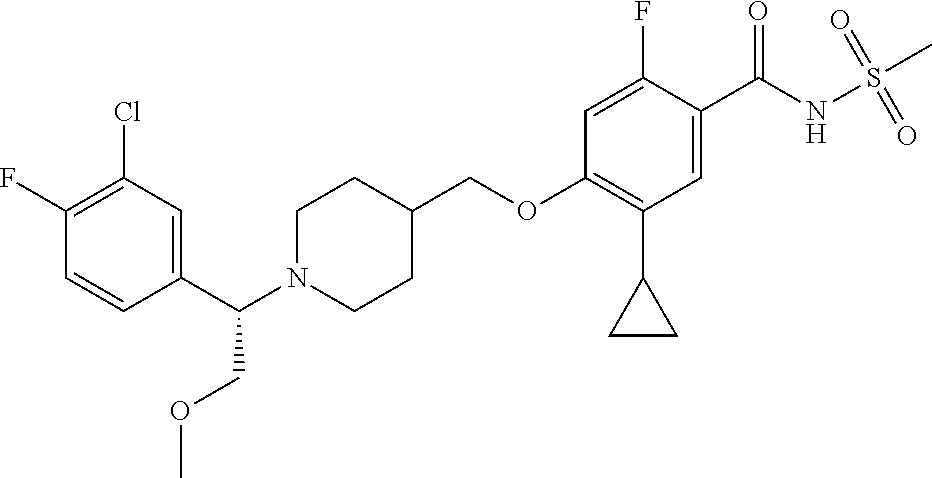







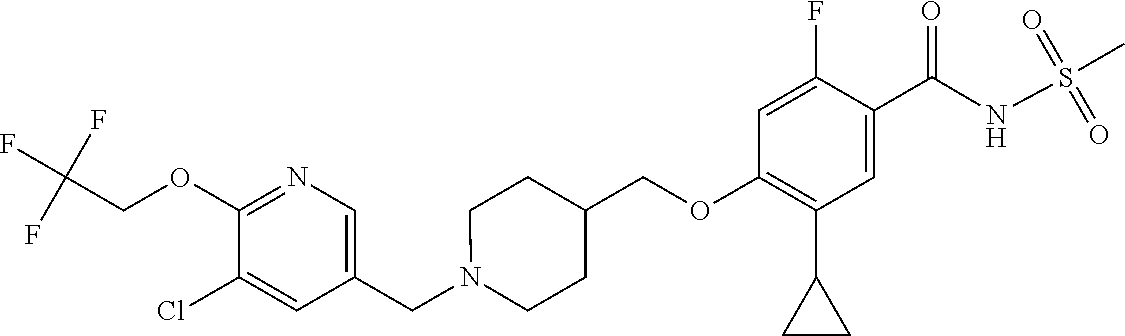


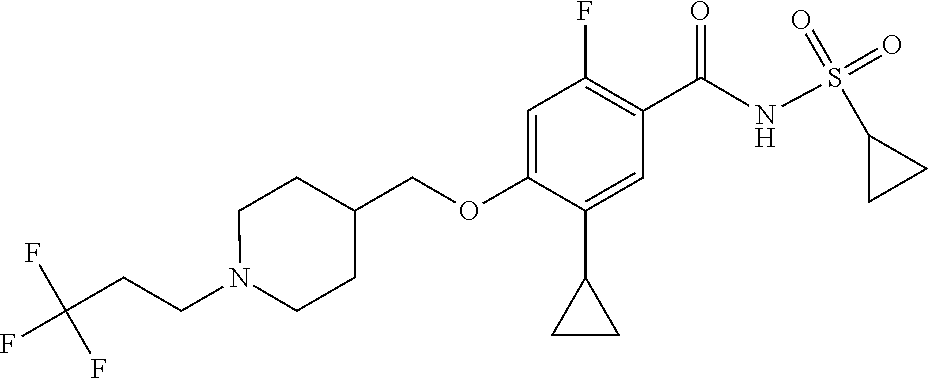


























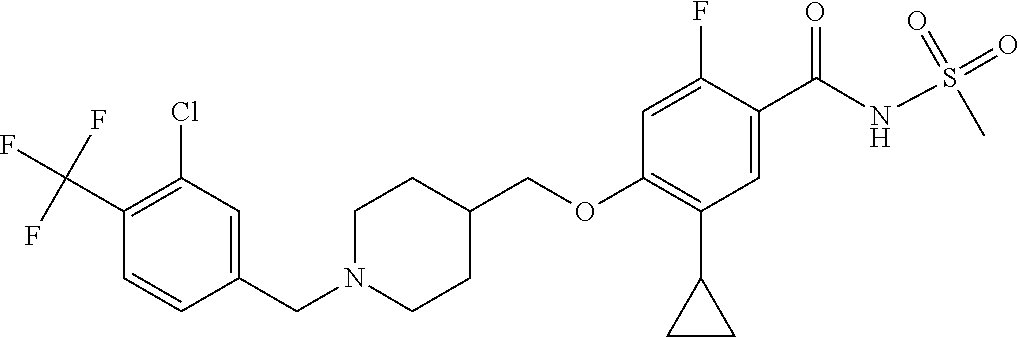
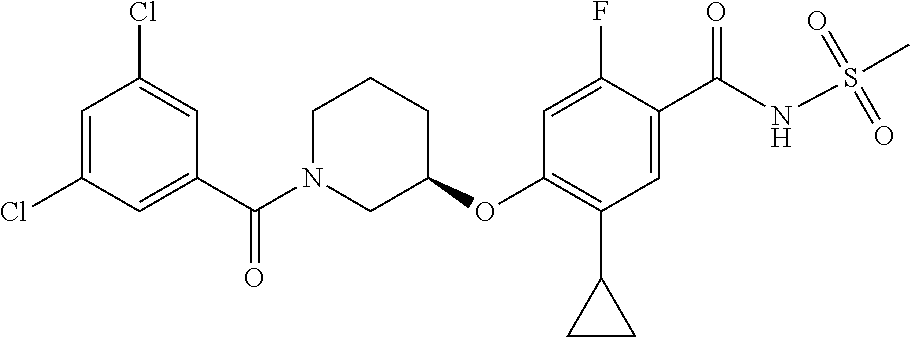



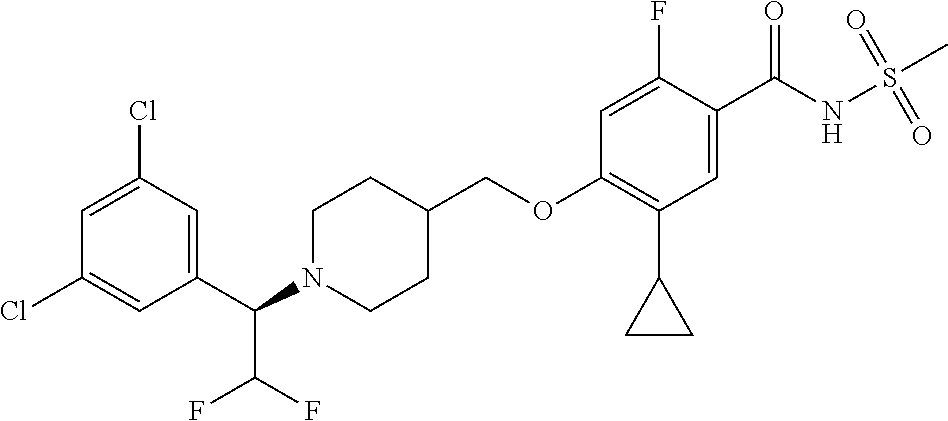














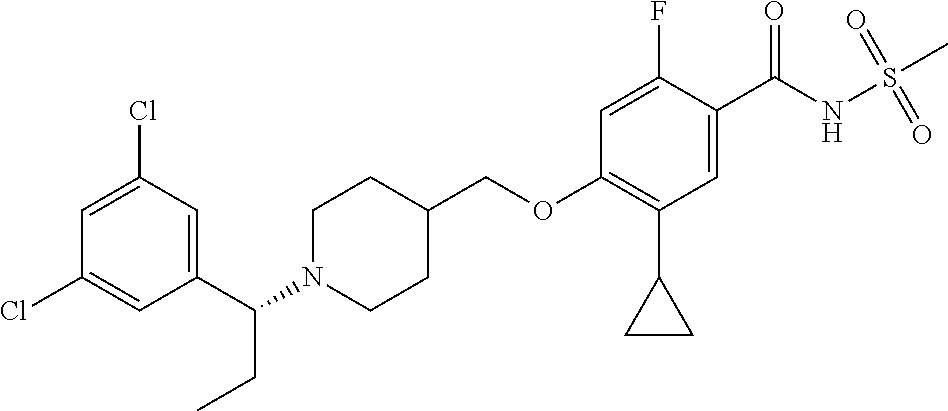



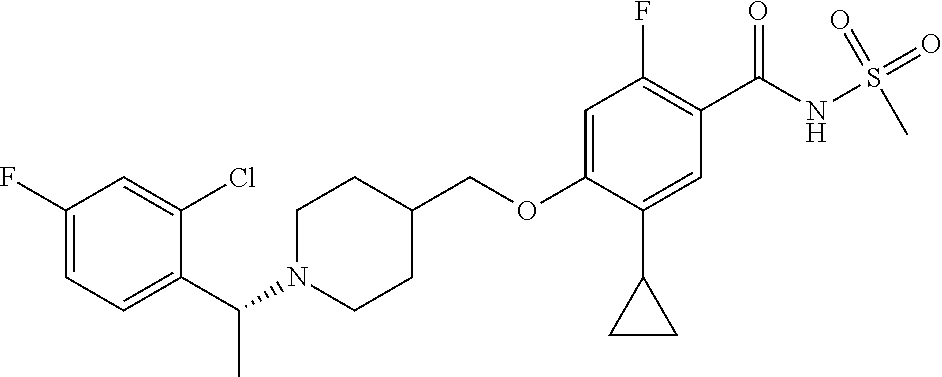







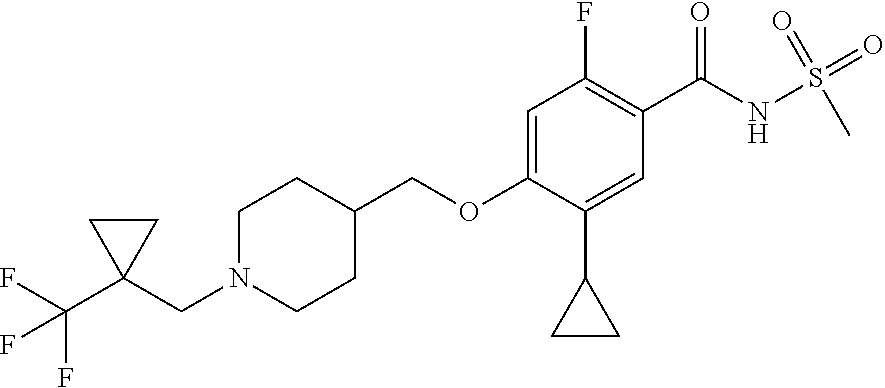




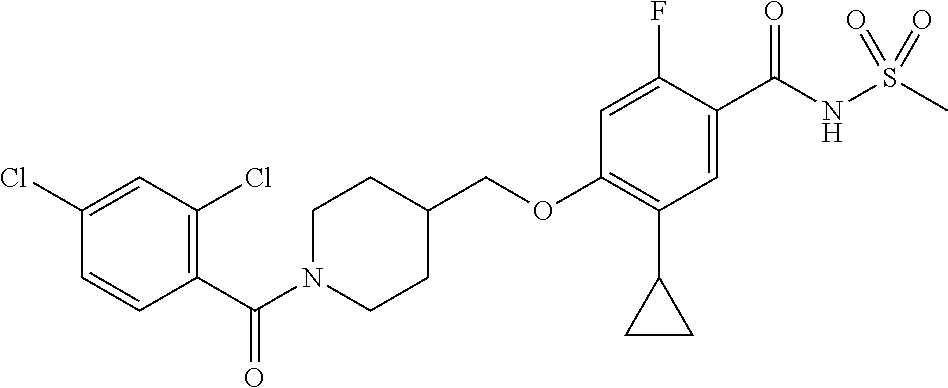
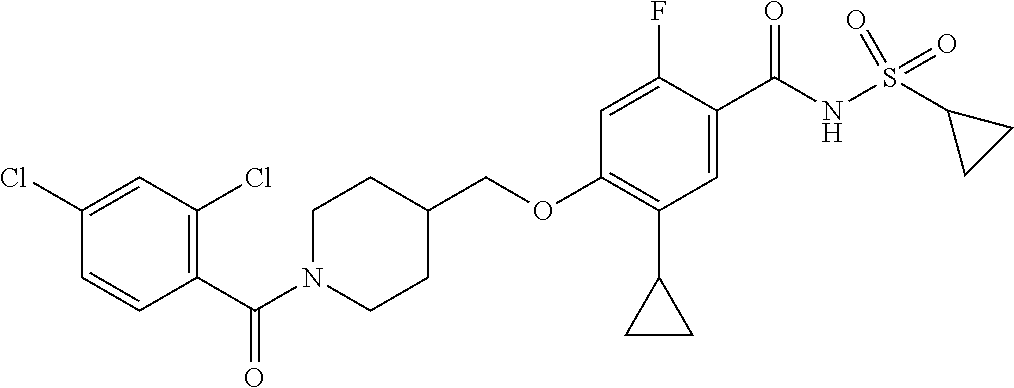


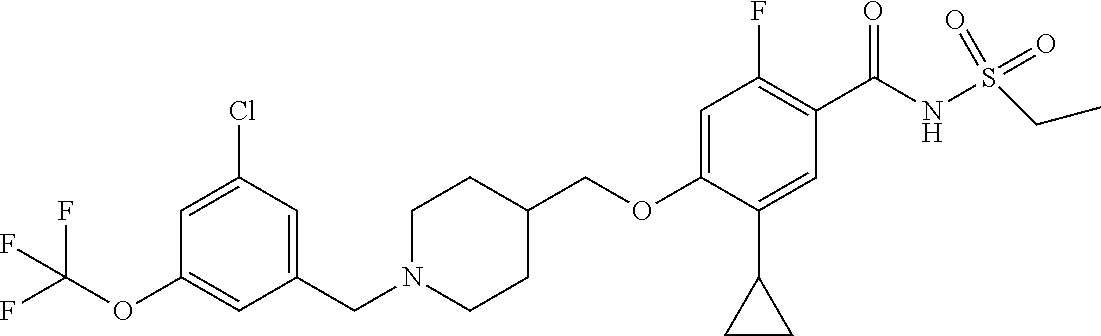


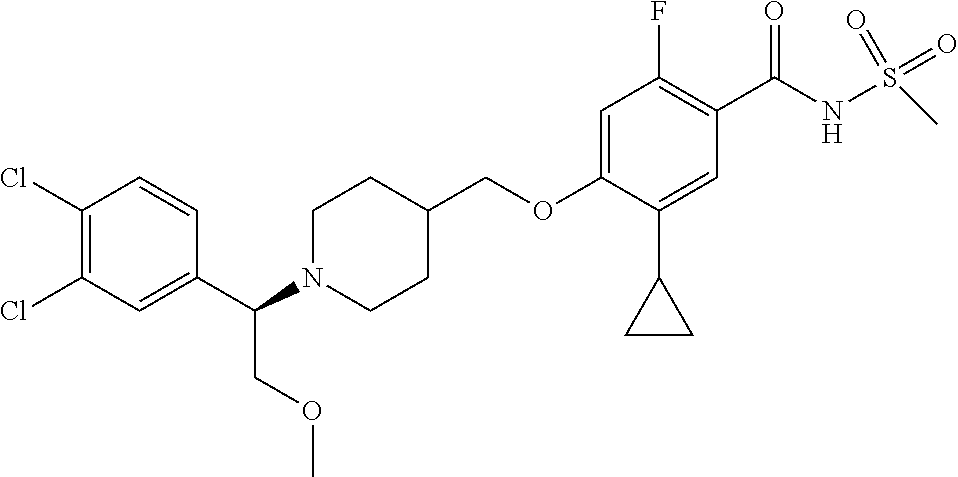


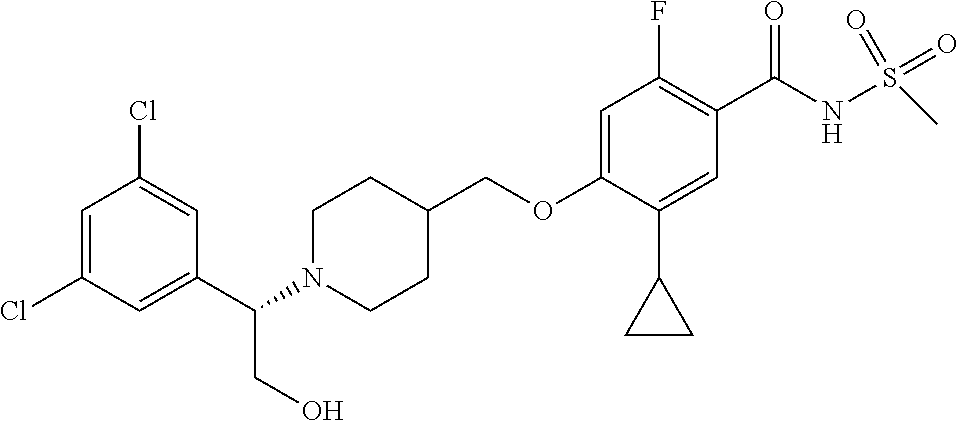





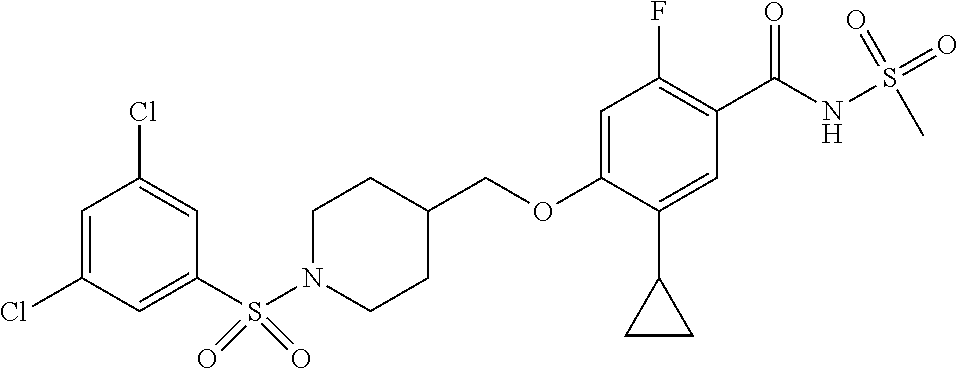
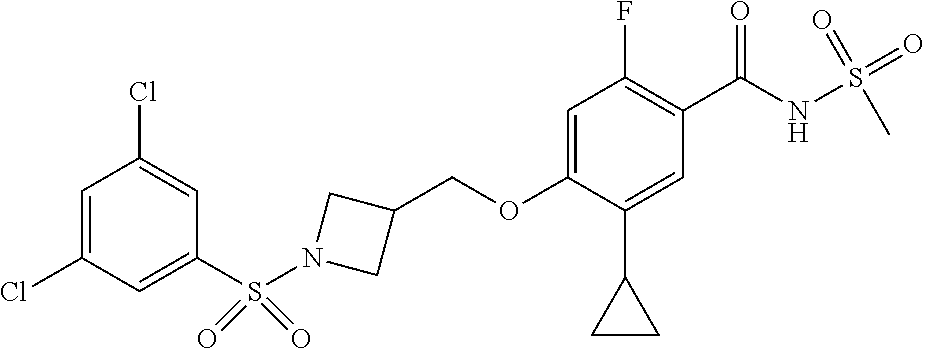




























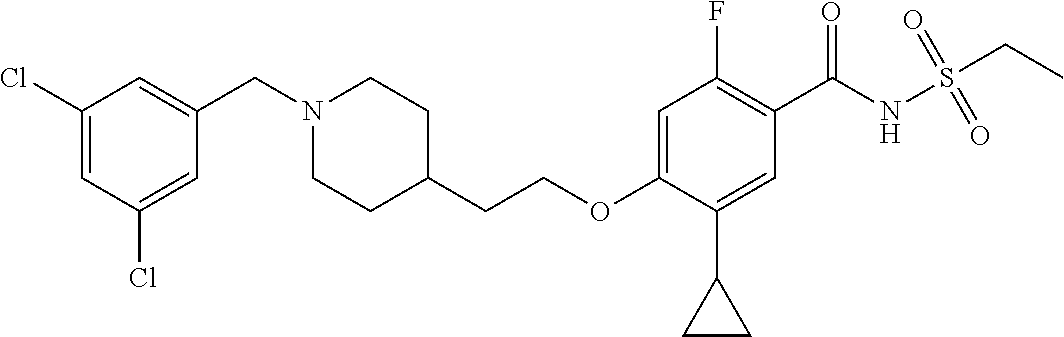
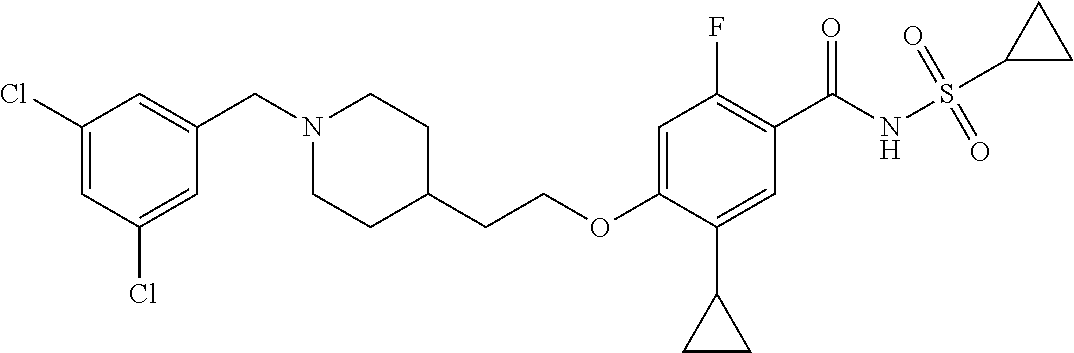





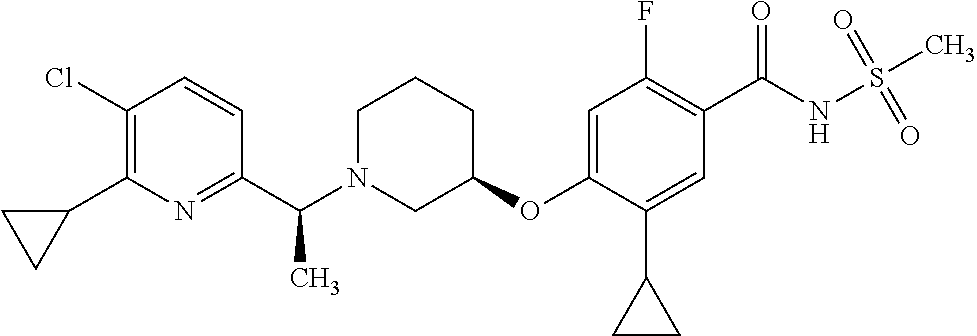
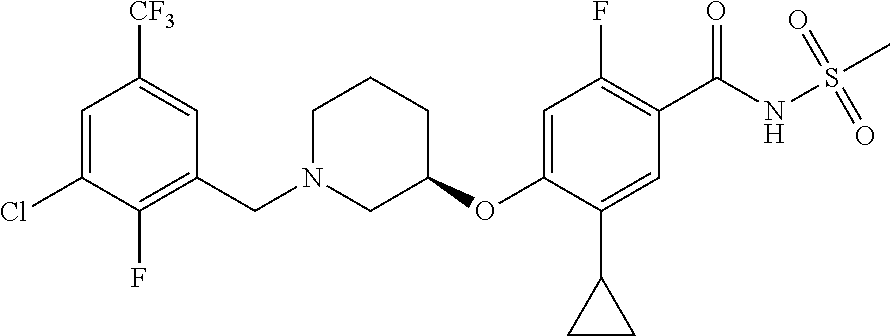






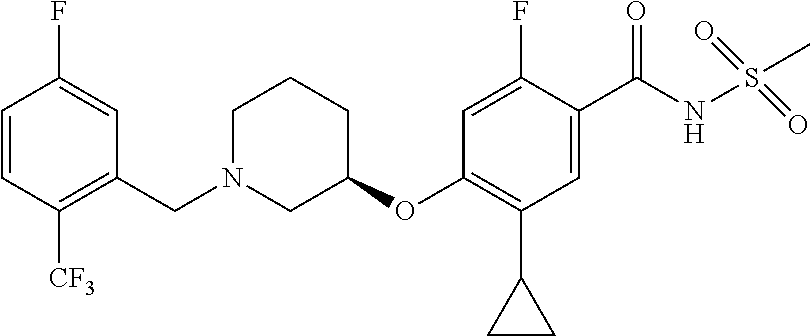




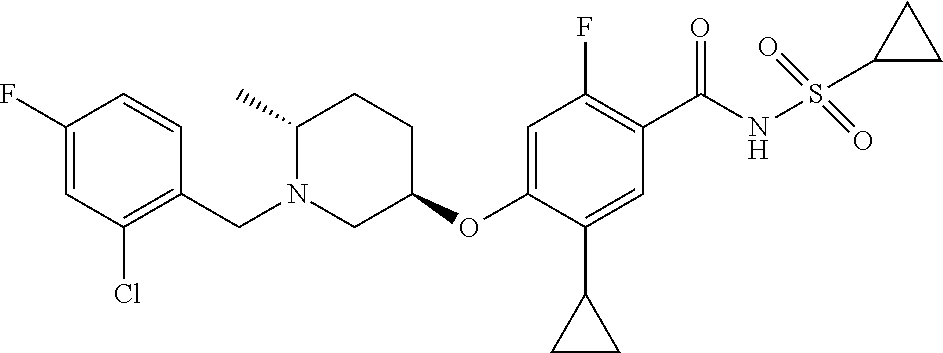


















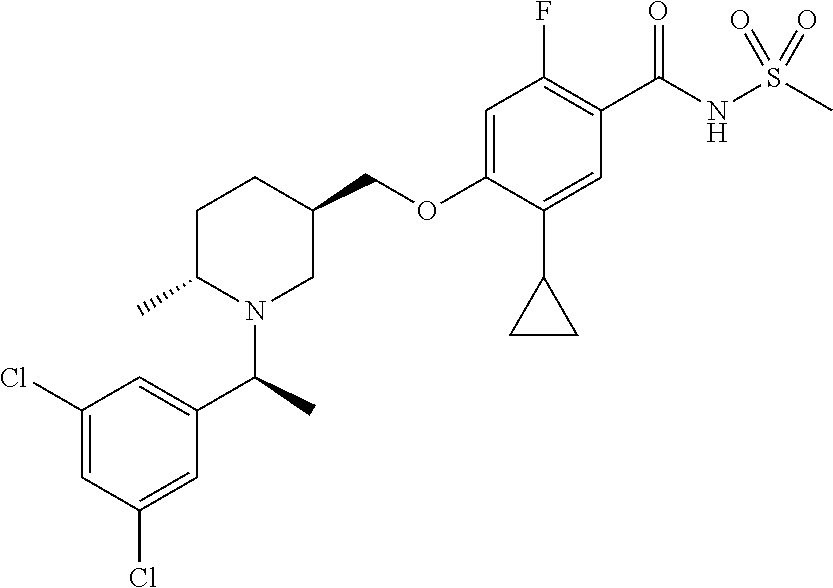
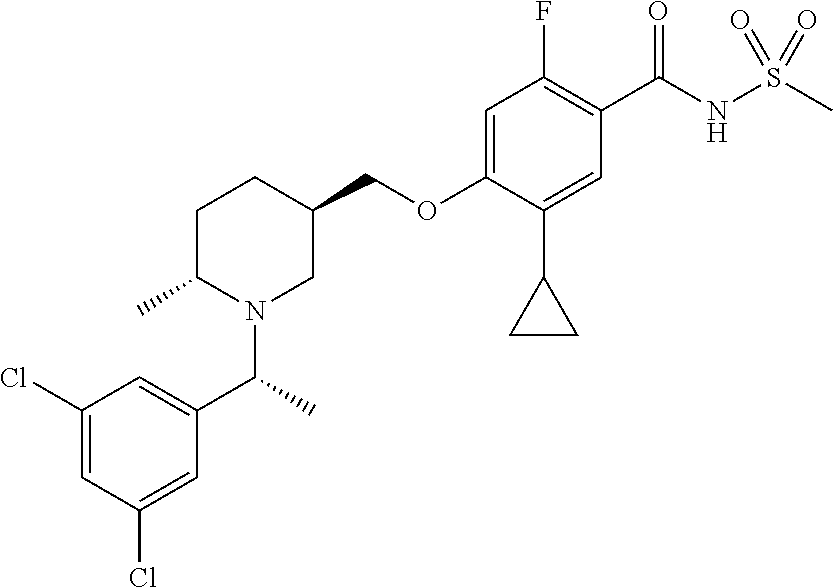








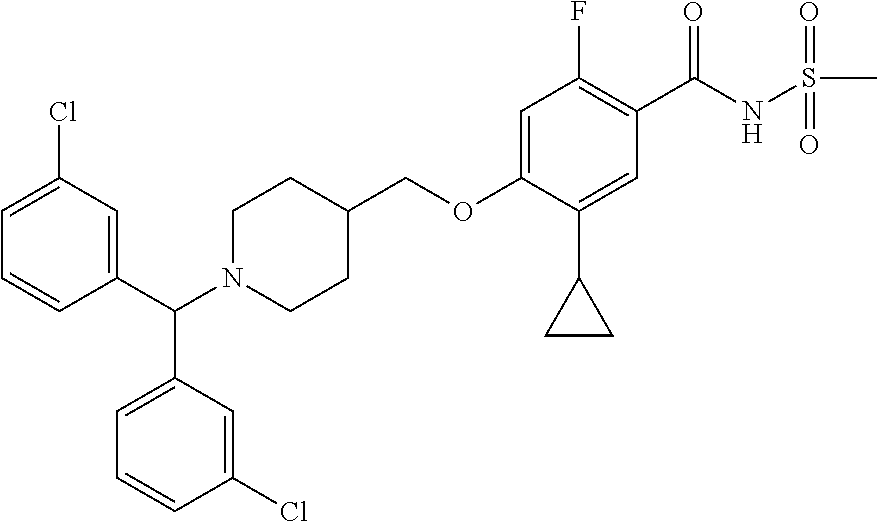






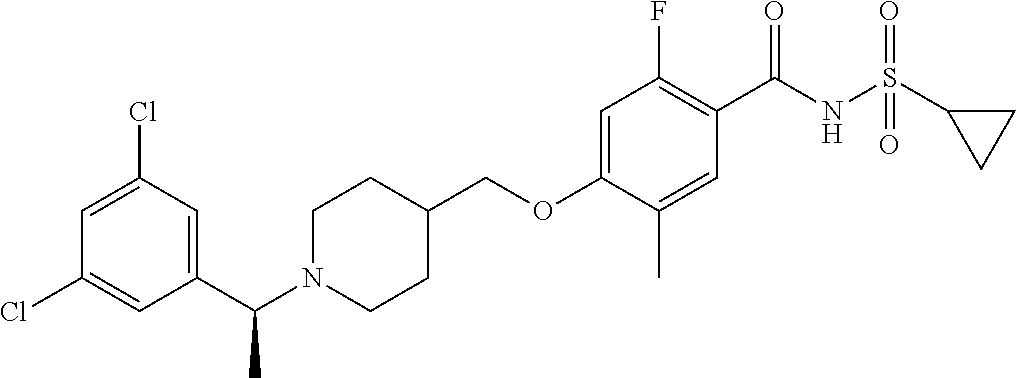




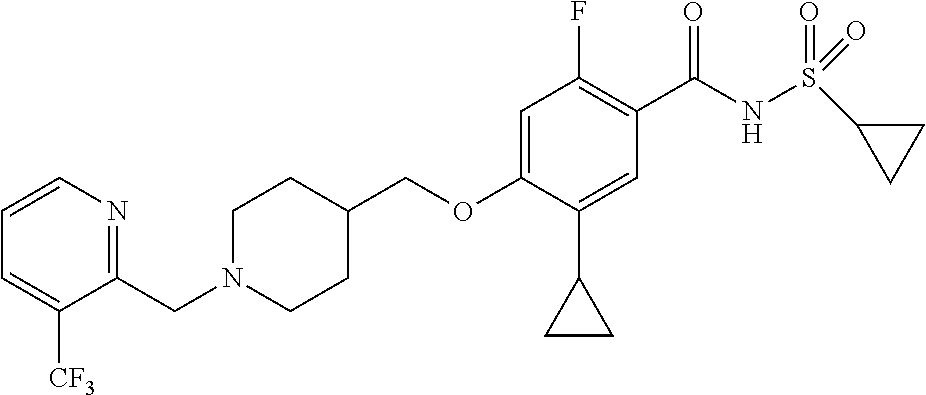






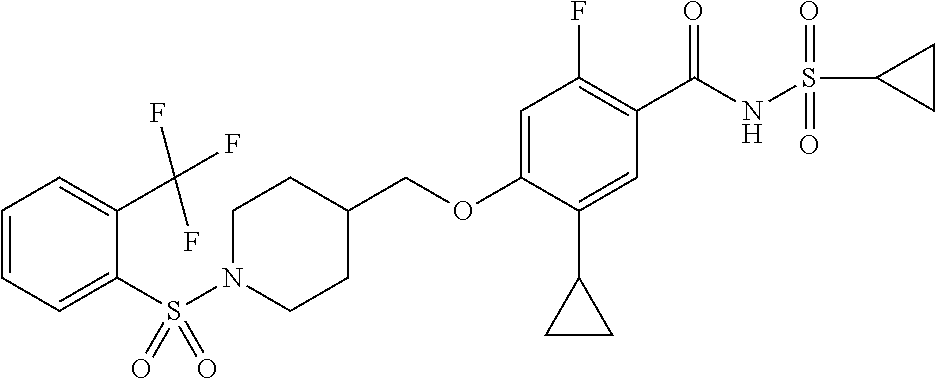





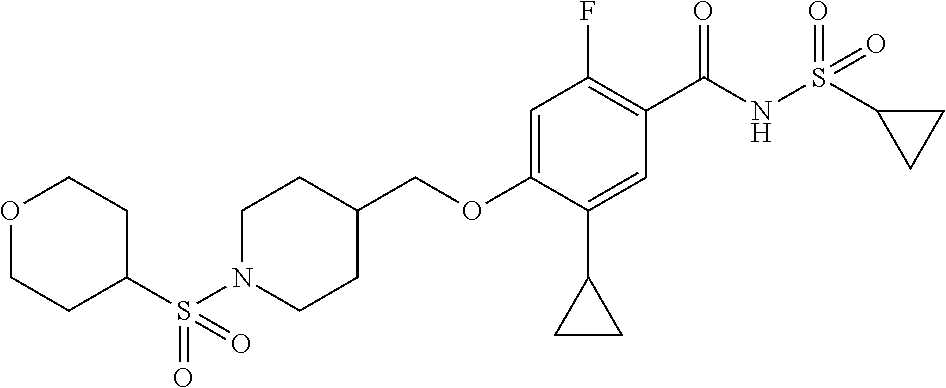






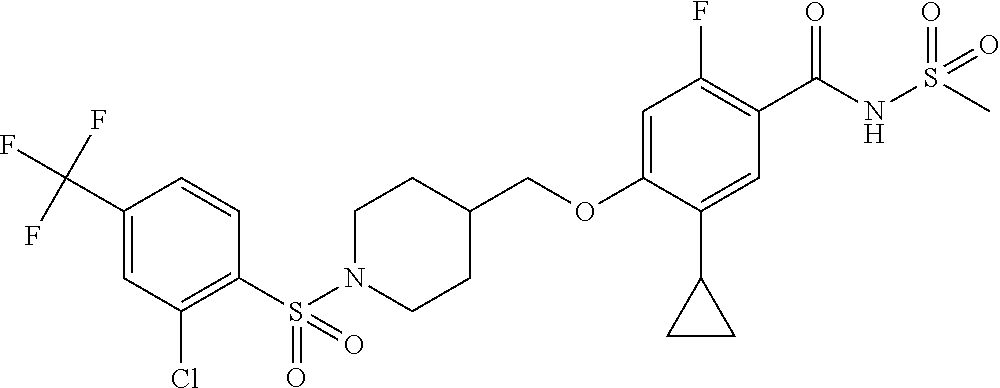



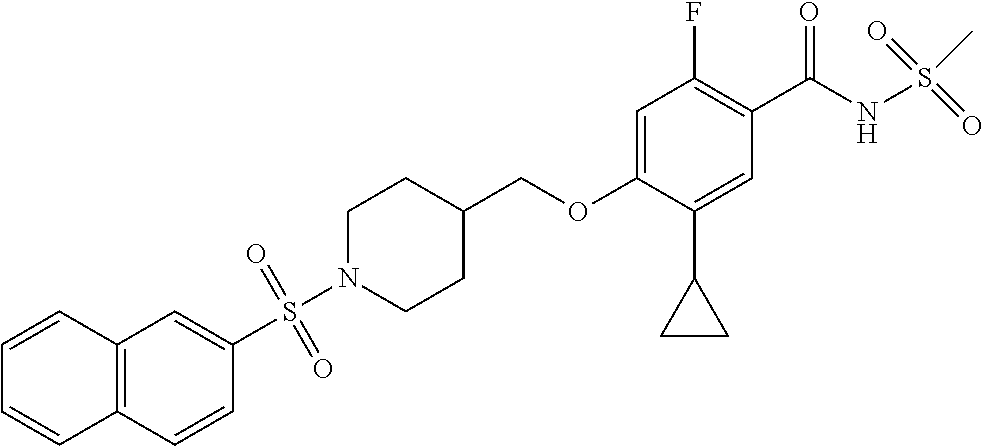

























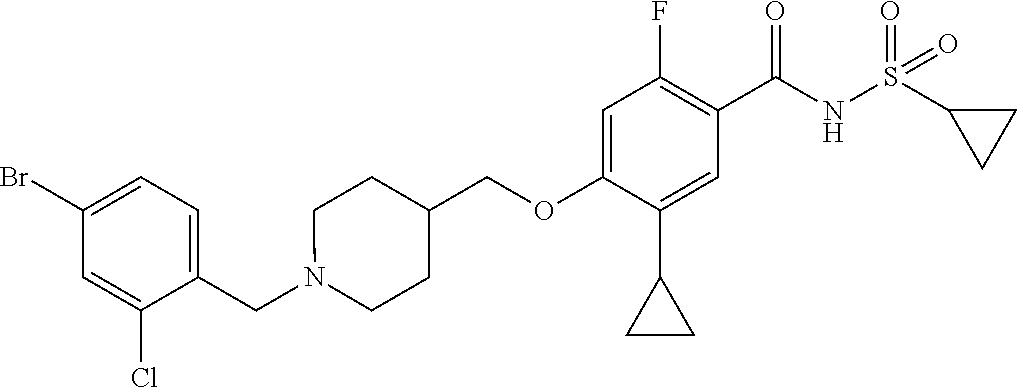


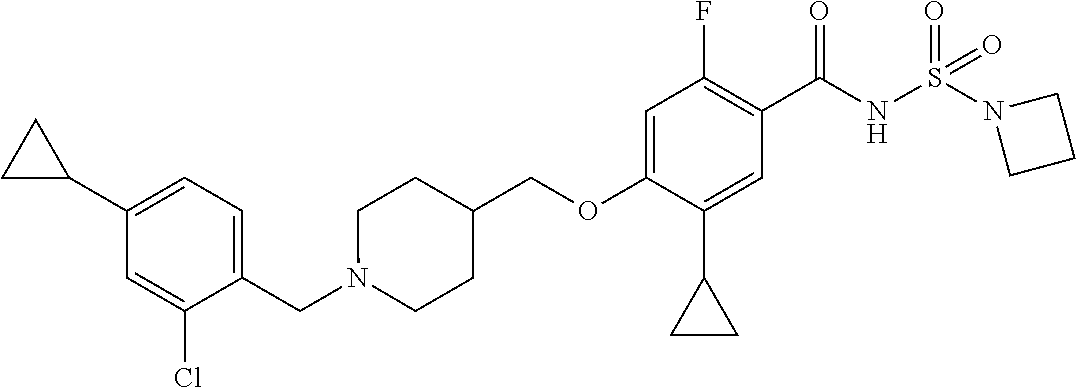









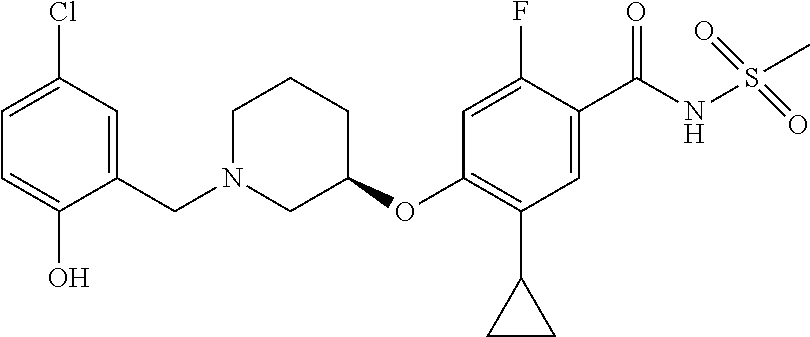

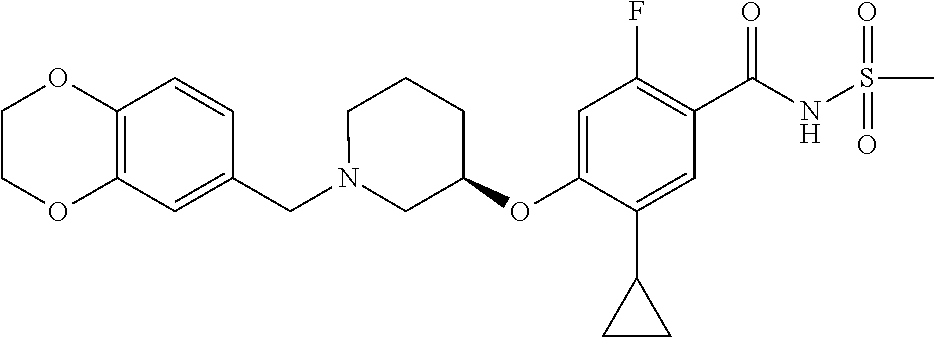





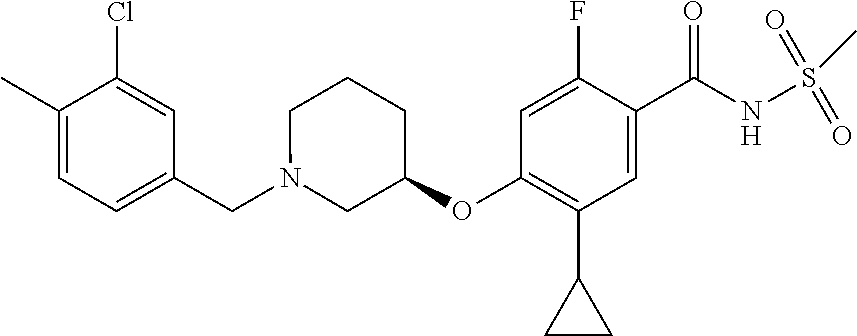








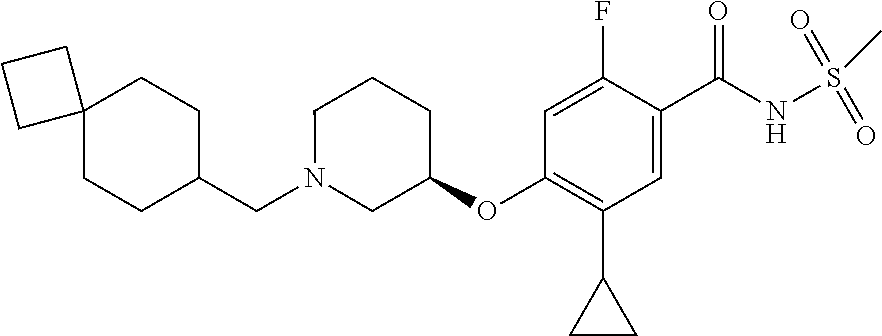











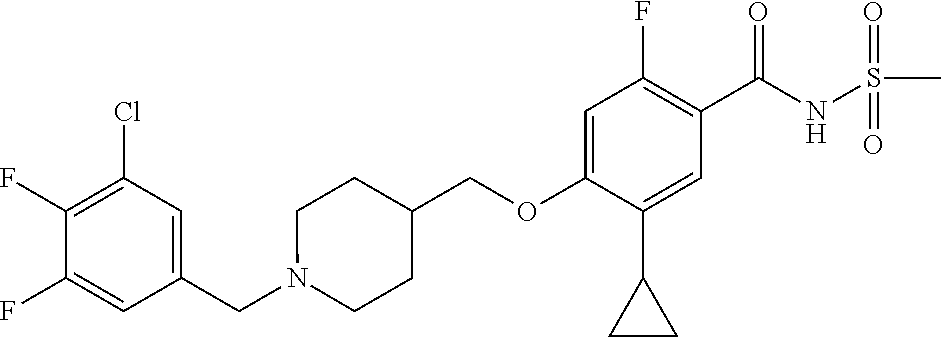




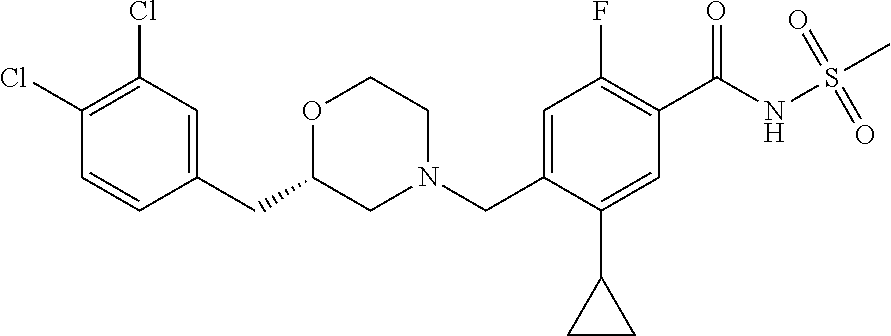

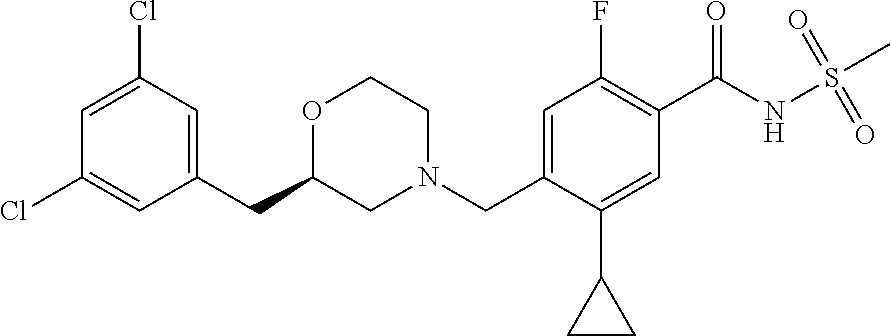


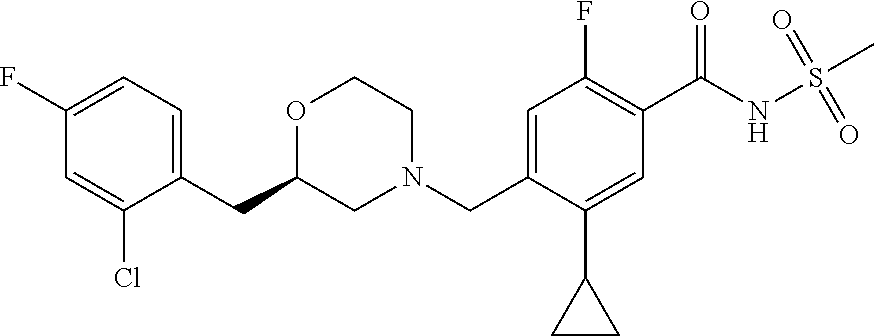
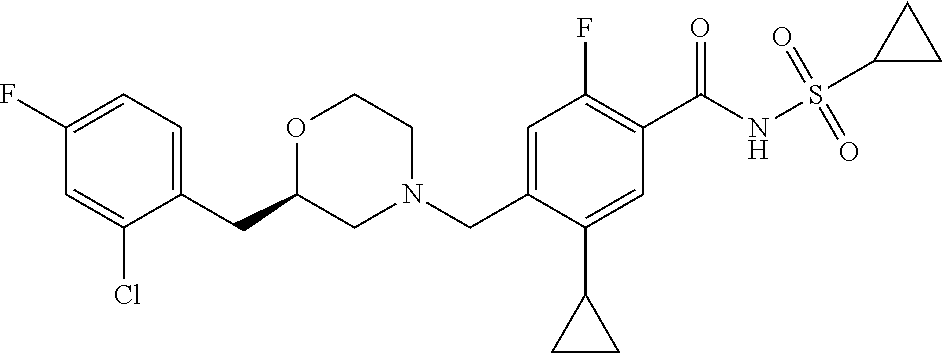


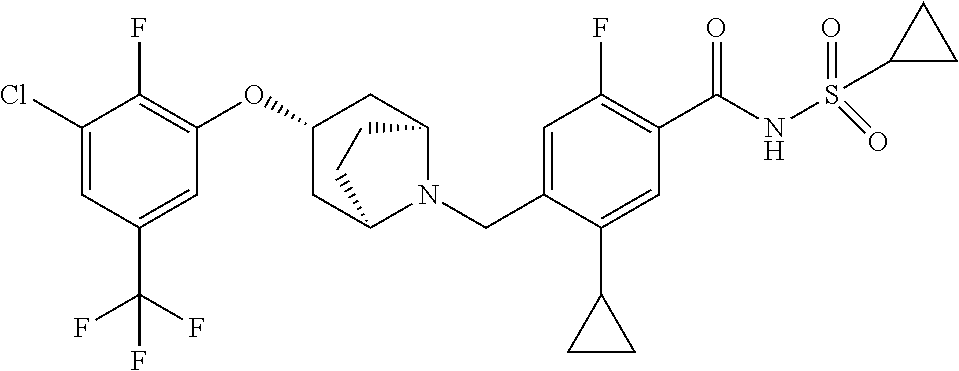




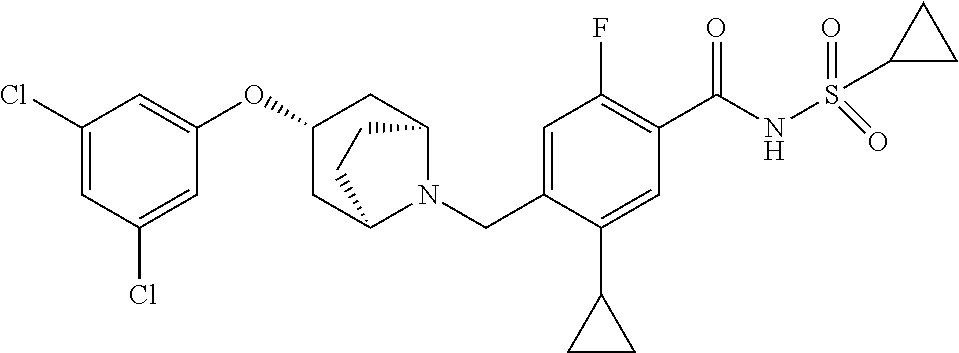








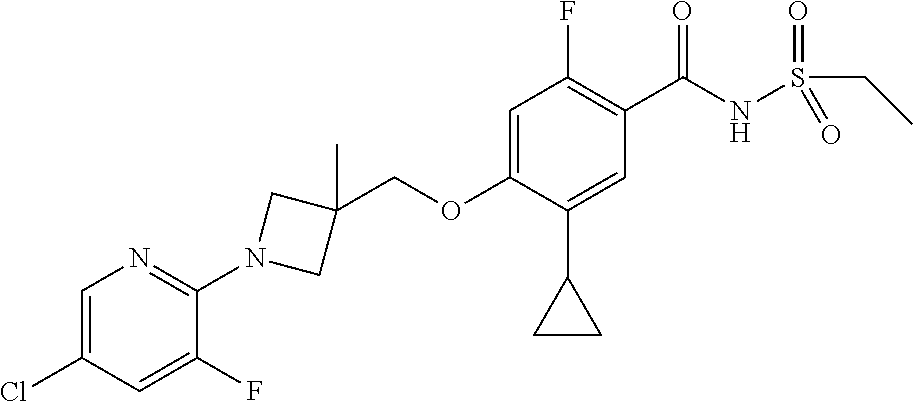






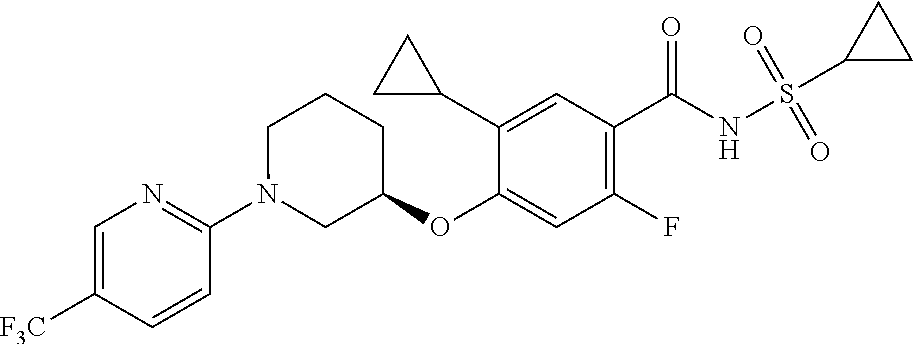



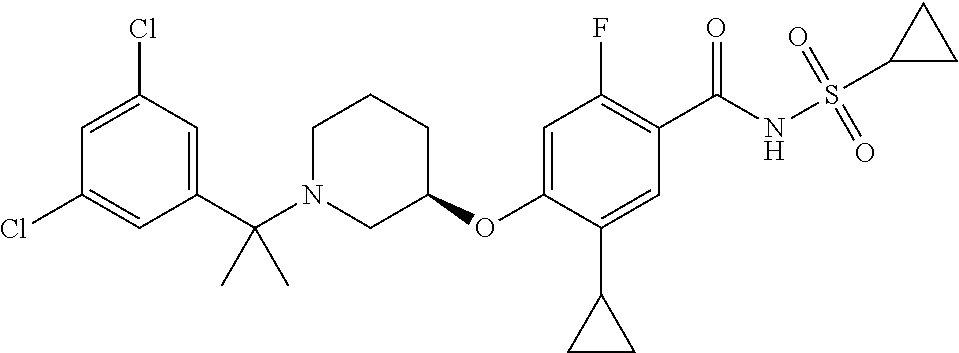
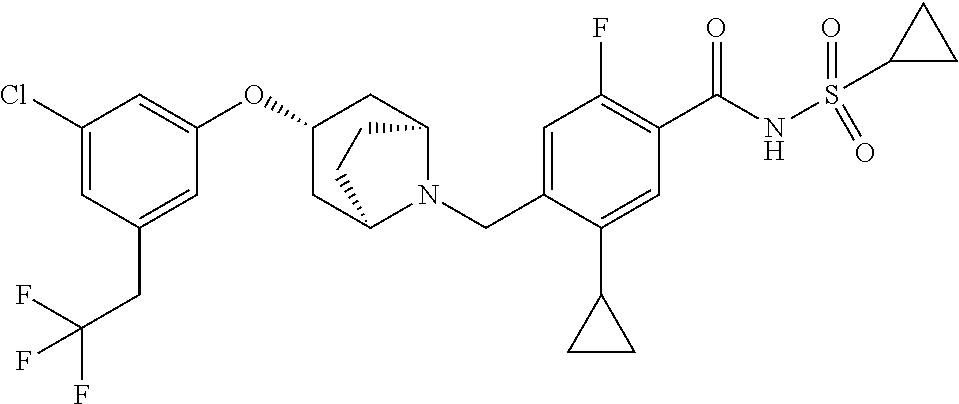







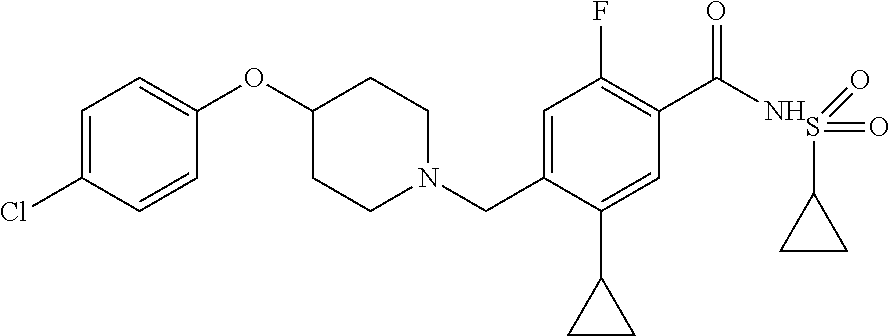
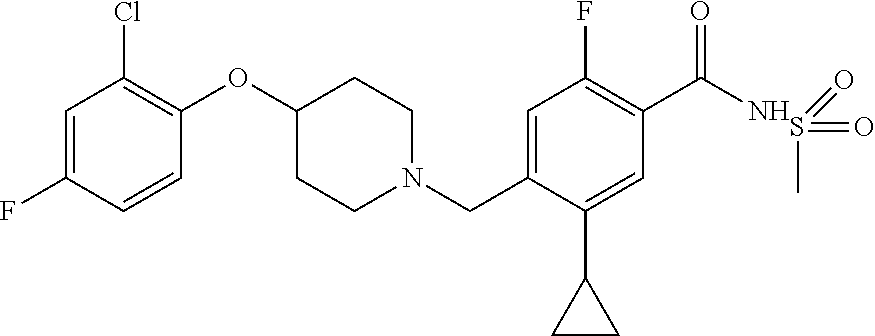








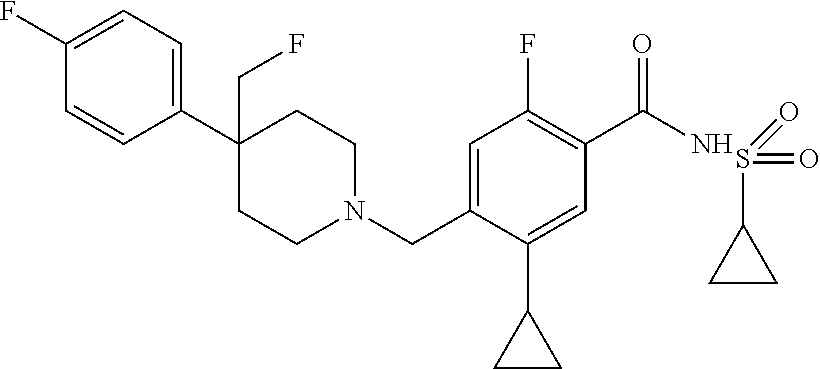




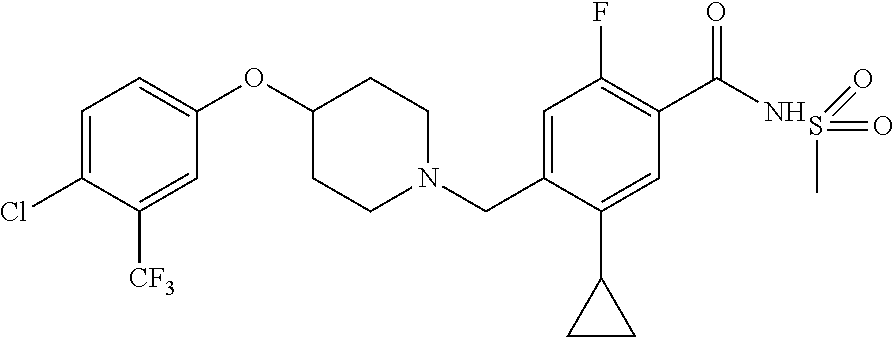



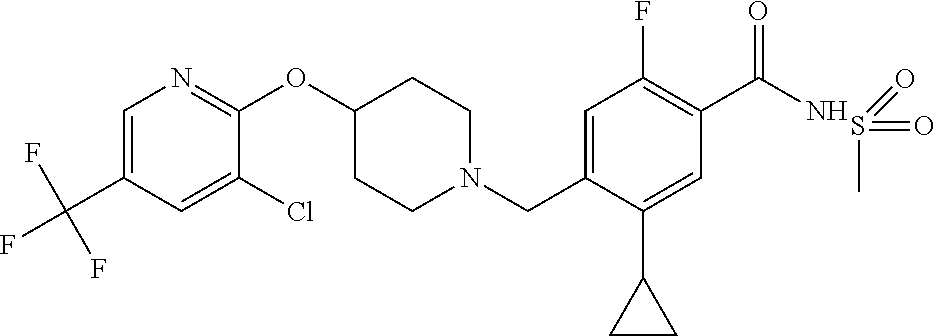

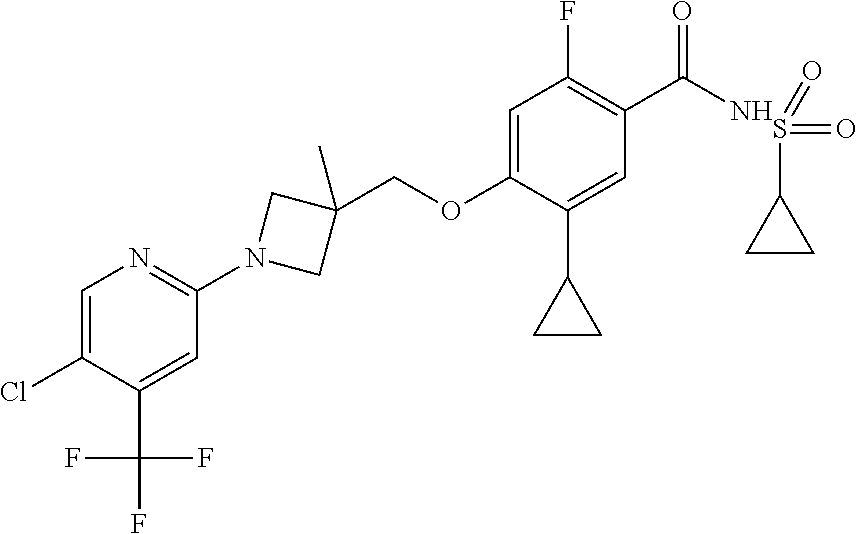






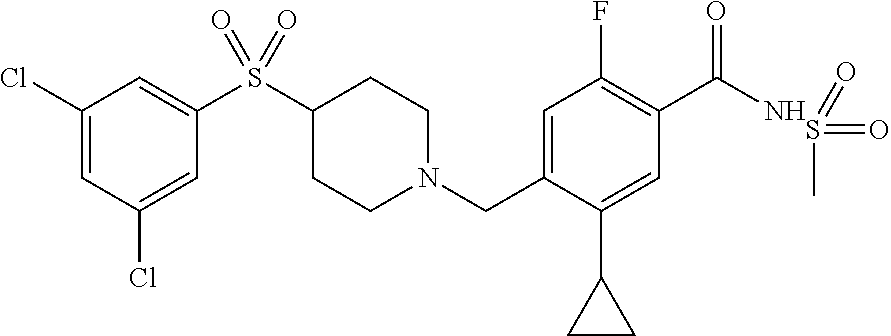






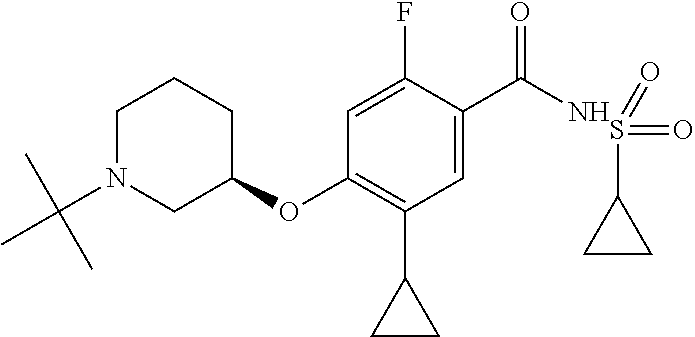








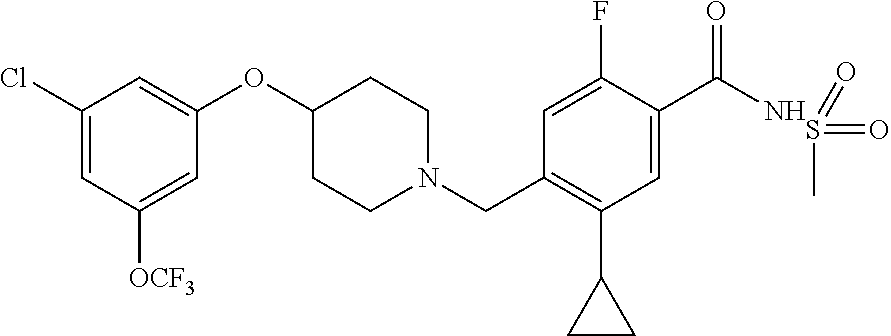
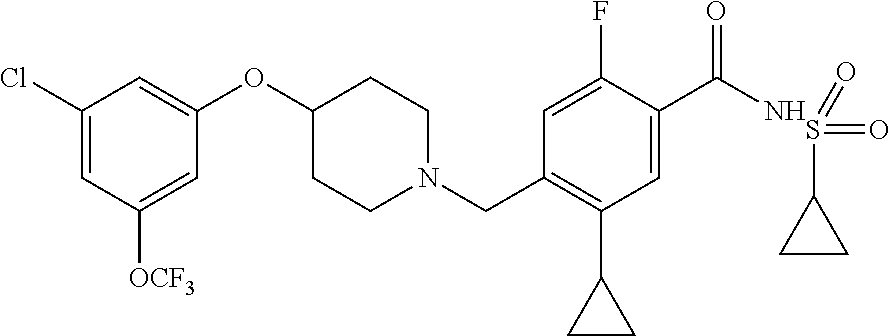


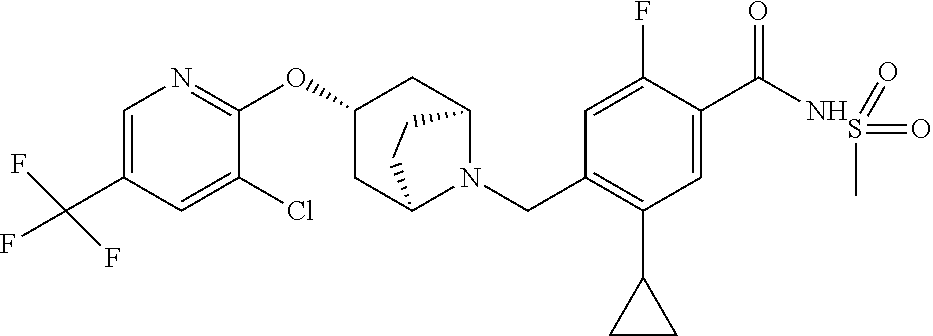

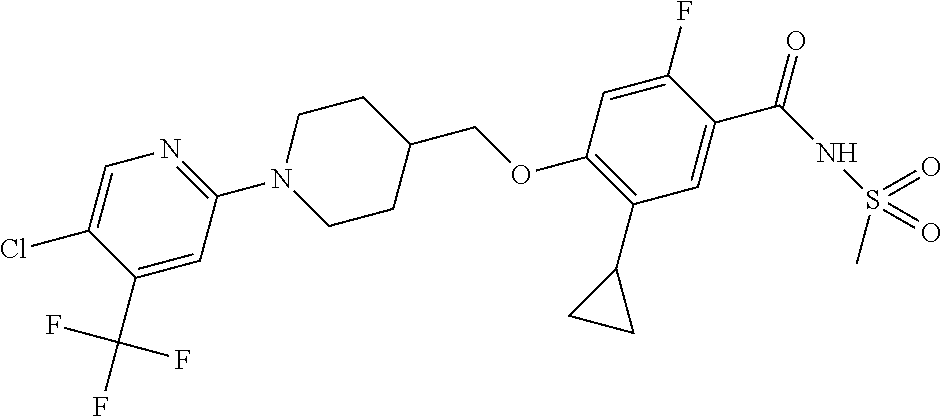











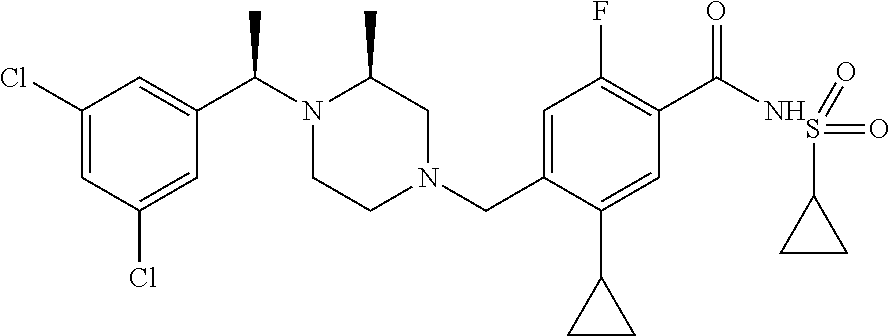




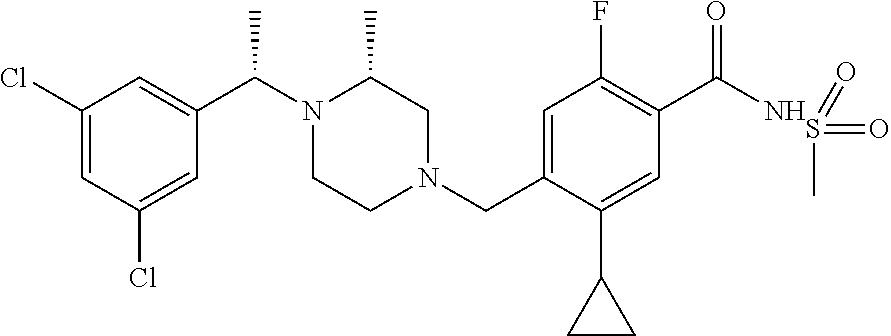











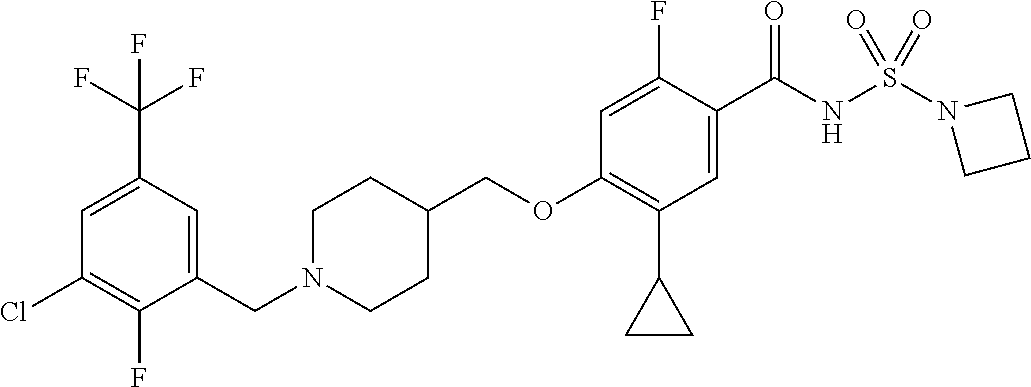














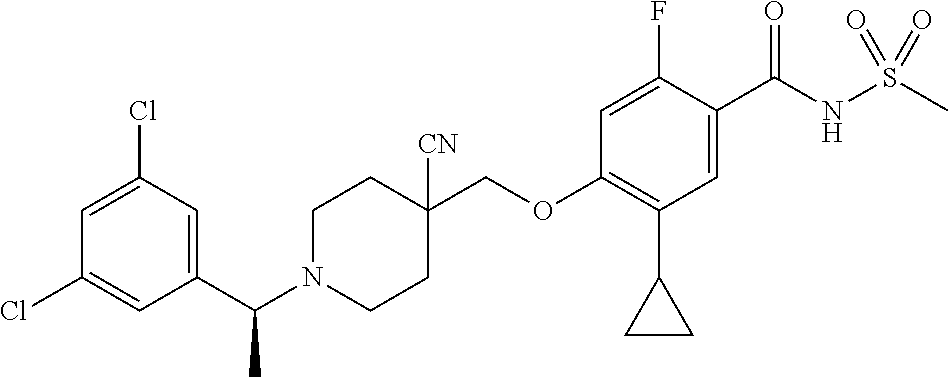
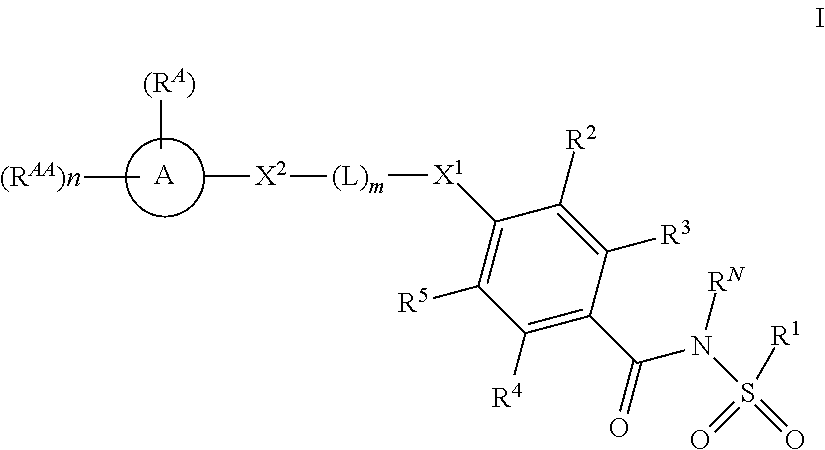
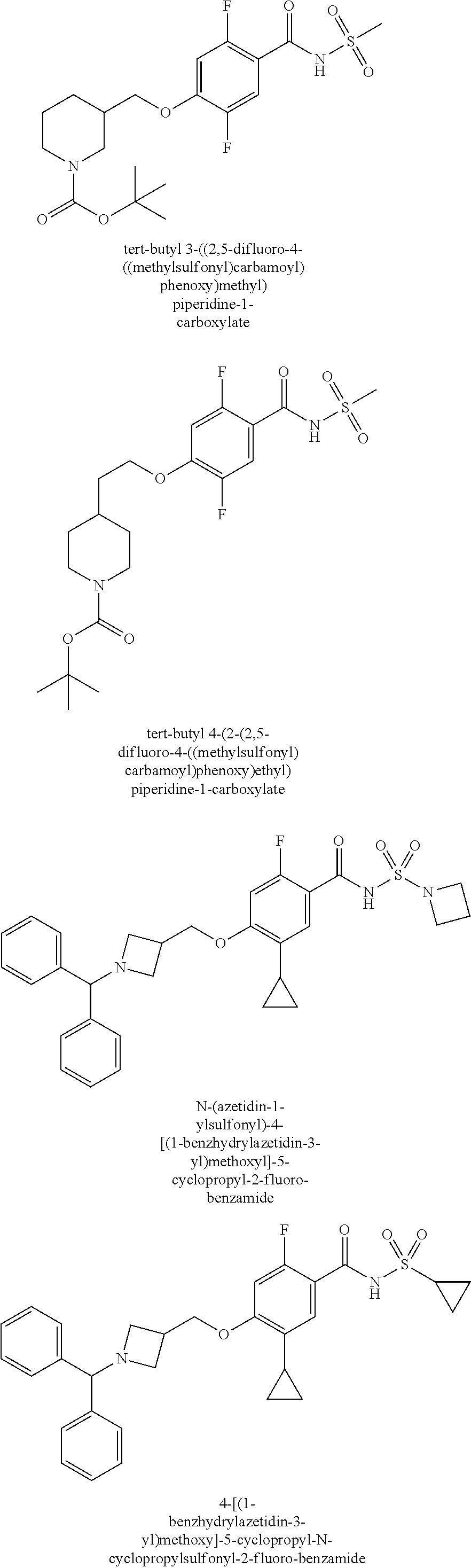
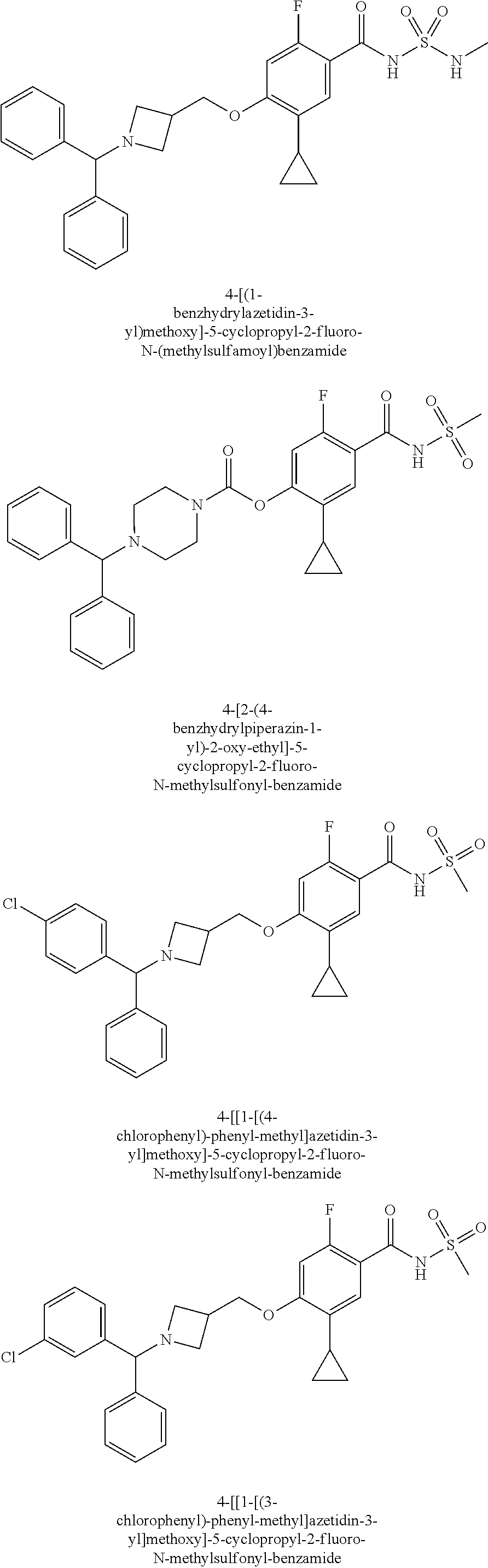



P00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.