Lipid Combinations
HODGSON; Charles ; et al.
U.S. patent application number 16/481444 was filed with the patent office on 2020-03-26 for lipid combinations. The applicant listed for this patent is PHARMALINK INTERNATIONAL LIMITED. Invention is credited to Charles HODGSON, Stephen MYERS, Christopher OLIVER.
| Application Number | 20200093867 16/481444 |
| Document ID | / |
| Family ID | 66993218 |
| Filed Date | 2020-03-26 |







View All Diagrams
| United States Patent Application | 20200093867 |
| Kind Code | A1 |
| HODGSON; Charles ; et al. | March 26, 2020 |
LIPID COMBINATIONS
Abstract
A combination or composition of mussel lipid and krill oil is disclosed, which is used to treat inflammation or pain. A process for preparing hill oil having a phospholipid content of about 50% or greater is also disclosed.
| Inventors: | HODGSON; Charles; (Nelson, NZ) ; MYERS; Stephen; (Evans Head, AU) ; OLIVER; Christopher; (Tregeagle, AU) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 66993218 | ||||||||||
| Appl. No.: | 16/481444 | ||||||||||
| Filed: | December 21, 2018 | ||||||||||
| PCT Filed: | December 21, 2018 | ||||||||||
| PCT NO: | PCT/IB2018/060482 | ||||||||||
| 371 Date: | July 26, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 35/612 20130101; A61P 29/00 20180101; C11B 1/10 20130101; A61K 35/618 20130101; A61K 9/48 20130101 |
| International Class: | A61K 35/618 20060101 A61K035/618; A61K 35/612 20060101 A61K035/612; A61K 9/48 20060101 A61K009/48; A61P 29/00 20060101 A61P029/00; C11B 1/10 20060101 C11B001/10 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 22, 2017 | AU | 2017905181 |
Claims
1. A combination comprising mussel lipid and krill oil, wherein the combination is adapted for separate or sequential administration.
2. The combination according to claim 1 in the form of a composition comprising mussel lipid and krill oil.
3. The combination according to claim 1 wherein the krill oil has a phospholipid content of at least about 40% w/w or at least about 60% w/ww.
4. (canceled)
5. The combination according to claim 1, wherein the krill oil has a water content of about 5% w/w or less.
6.-7. (canceled)
8. The combination according to claim 1, wherein the krill oil has an extraction solvent content of about 5% w/w or less, about 3% w/w or less, or about 1% w/w or less.
9.-10. (canceled)
11. The combination according to claim 1 wherein the mussel lipid is in the form of mussel powder.
12. The combination according to claim 1 wherein the mussel lipid is in the form of mussel lipid extract, optionally containing vitamin E.
13. The combination according to claim 1 wherein the weight ratio of mussel lipid to krill oil is in the range of 1:99 to 99:1.
14. The combination according to claim 13 wherein the weight ratio of mussel lipid to krill oil is about 5:95, or about 10:90, or about 15:85, or about 20:80, or about 25:75, or about 30:70, or about 35:65, or about 40:60, or about 45:55, or about 50:50, or about 55:45, or about 60:40, or about 65:35, or about 70:30, or about 75:25, or about 80:20, or about 85:15, or about 90:10, or about 95:5.
15. The combination according claim 1 in oral unit dosage form.
16. The combination according to claim 15 wherein the oral unit dosage form is a soft gel capsule.
17. The combination according to claim 15 wherein the oral unit dosage form comprises from about 10 mg to about 10 g, of mussel lipid.
18. The combination according to claim 17 wherein the oral unit dosage form comprises about 10 mg, 20 mg, 30mg, 40 mg 50 mg, 100 mg, 150 mg, 200mg, 250 mg, 300 mg, 350 mg, 400 mg, 450 mg, 500 mg, 550 mg, 600 mg, 650 mg, 700 mg, 750 mg, 800 mg, 850 mg, 900 mg, 950 mg 1 g, 1.1 g, 1.2 g, 1.3 g, 1.4, g, 1.5 g, 1.6 g, 1.7 g, 1.8 g, 1.9 g, 2.0 g, 2.1 g, 2.2 g, 2.3 g, 2.4 g, 2.5 g, 2.6 g, 2.7 g, 2.8 g, 2.9 g, 3.0 g, 3.2 g 3.5 g, 3.7 g, 4.0 g, 4.5 g, 5.0 g, 5.5 g, 6.0 g, 6.5 g, 7.0 g, 7.5 g, 8.0 g, 8.5 g, 9.0 g, or about 9.5 g of mussel lipid.
19. The combination according to claim 15 wherein the oral unit dosage form comprises from about 10 mg to about 10 g, of krill oil.
20. The combination according to claim 19 wherein the oral unit dosage form comprises about 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 100 mg, 150 mg, 200 mg, 250 mg, 300 mg, 350 mg, 400 mg, 450 mg, 500 mg, 550 mg, 600 mg, 650 mg, 700 mg, 750 mg, 800 mg, 850 mg, 900 mg, 950 mg 1 g, 1.1 g, 1.2 g, 1.3 g, 1.4, g, 1.5 g, 1.6 g, 1.7 g, 1.8 g, 1.9 g, 2.0 g, 2.1 g, 2.2 g, 2.3 g, 2.4 g, 2.5 g, 2.6 g, 2.7 g, 2.8 g, 2.9 g, 3.0 g, 3.2 g 3.5 g, 3.7 g, 4.0 g, 4.5 g, 5.0 g, 5.5 g, 6.0 g, 6.5 g, 7.0 g, 7.5 g, 8.0 g, 8.5 g, 9.0 g, or about 9.5 g of krill oil.
21. The combination according to claim 15 wherein the oral unit dosage form comprises about 10-500 mg of the combination.
22. The combination according to claim 21, wherein the oral unit dosage form comprises about 50-300 mg of the combination.
23. The combination according to claim 1, further comprising one or more pharmaceutically acceptable carriers and/or additives.
24. The combination according to claim 1 consisting or consisting essentially of mussel lipid and krill oil.
25. A composition comprising mussel lipid and krill oil.
26. The composition according to claim 25 wherein the krill oil has a phospholipid content of at least about 50% w/w.
27. The composition according to claim 25 wherein the mussel lipid is in the form of mussel lipid extract, optionally containing vitamin E.
28. The composition according to claim 25 wherein the weight ratio of mussel lipid to krill oil is about 5:95, or about 10:90, or about 15:85, or about 20:80, or about 25:75, or about 30:70, or about 35:65, or about 40:60, or about 45:55, or about 50:50, or about 55:45, or about 60:40, or about 65:35, or about 70:30, or about 75:25, or about 80:20, or about 85:15, or about 90:10, or about 95:5.
29. The composition according to claim 25, further comprising a carrier oil.
30. The composition according to claim 29 wherein the carrier oil comprises from about 10% w/w to about 90% w/w of the total composition.
31. The composition according to claim 29 wherein the weight ratio of carrier oil to combined amount of mussel lipid and krill oil is from about 3:1, to about 1:3.
32. The composition according to claim 25 in unit dosage form.
33. The composition according to claim 32 encapsulated in a soft gel capsule.
34. The composition according to claim 32 comprising about 10-500 mg of combined mussel lipid and krill oil.
35. The combination according to claim 34, comprising about 50-300 mg of combined mussel lipid and krill oil.
36. (canceled)
37. A method of treating inflammation in a subject in need thereof, comprising administering to said subject a combination according to claim 1 or a composition according to claim 25.
38.-43. (canceled)
44. A method of treating pain in a subject in need thereof, comprising administering to said subject a combination according to claim 1 or a composition according to claim 25.
45.-49. (canceled)
50. A process for preparing krill oil having a phospholipid content of about 50% or greater, comprising the steps of: (a) contacting a krill biomass feed material with a mixture of CO.sub.2 and ethanol, to extract a krill oil; and (b) contacting said krill oil with CO.sub.2 to extract at least a proportion of non-polar lipid components such that the oil has a phospholipid content of at least 50% w/w.
51. The process of claim 50, wherein the krill biomass feed material is contacted with a mixture of about 15% w/w to about 30% w/w ethanol in CO.sub.2.
52. The process of claim 50 wherein step (a) is performed at a temperature of about 60.degree. or less.
53. The process of claim 50, wherein step (a) is performed at a pressure at or greater than about 300 bar.
54. The process of claim 50 wherein step (b) is performed at a temperature of about 60.degree. or less.
55. The process of claim 50 wherein step (a) is performed at a pressure at or greater than about 300 bar.
56. The process of claim 50 wherein the oil obtained from step (b) has a phospholipid content in the range of about 60% w/w to about 90% w/w.
57. The process of claim 50 wherein ethanol is removed from the extracted oil obtained in step (a).
58. The process according to claim 57 wherein the ethanol is removed under vacuum at a temperature of about 60.degree. C. or less.
Description
FIELD
[0001] The present disclosure relates generally to combinations of marine lipids. In particular the disclosure relates to a combination of lipids obtained from Perna canaliculus and krill, compositions and preparations comprising said combinations and the use of said combinations and compositions in therapy. The disclosure further relates to processes for the manufacture of krill oils and their use in the combinations and compositions.
BACKGROUND
[0002] The reference in this specification to any prior publication (or information derived from it), or to any matter which is known, is not, and should not be taken as an acknowledgment or admission or any form of suggestion that that prior publication (or information derived from it) or known matter forms part of the common general knowledge in the field of endeavour to which this specification relates.
[0003] Inflammation is a necessary physiological adaptive response to injury and infection, without which humans and animals could not survive. Its function is to eliminate the initial cause of injury, remove offending factors and initiate repair of tissue structure and function. Its early acute phase is typically characterised by heat, pain, redness and swelling. The usual outcome of acute inflammation is restoration and repair of damage, however, disproportionate acute and longer-term chronic inflammatory responses in conditions such sepsis can be harmful.
[0004] Importantly, chronic inflammation has now been implicated in the pathology of a wide number of diseases affecting all tissues and organs including osteoarthritis, rheumatoid arthritis, cardiovascular disease, cerebrovascular disease, respiratory disease, autoimmune disease and sarcopenia; indeed chronic inflammation has been implicated in the process of ageing itself.
[0005] As a response to the inflammatory process, there has been the development of a range of drugs, the most effective being glucocorticoid steroid drugs which are capable of suppressing excessive inflammation. However, steroid drug therapy is limited in its widespread clinical use due to significant side-effects, and is generally restricted to short-term use only. A second class of inflammatory drugs was developed called the non-steroidal anti-inflammatory drugs (NSAIDS) such as ibuprofen etc see Table 1. It was recognised that aspirin had anti-inflammatory activity and that it was part of this second class of anti-inflammatory drugs. These drugs were safer than steroid drugs and could be used for more chronic inflammatory conditions, such as osteoarthritis.
TABLE-US-00001 TABLE 1 List of commonly used non-steroidal anti-inflammatory drugs (NSAIDS) Generic name Brand name(s) celecoxib Celebrex diclofenac Cambia, Cataflam, Dyloject, Flector, Pennsaid, Solaraze, Voltaren, Voltaren-XR, Zipsor, Zorvolex, Arthrotec (combination with misoprostol) diflunisal No brand name currently marketed etodolac No brand name currently marketed fenoprofen Nalfon flurbiprofen Ansaid ibuprofen Advil, Caldolor, Children's Advil, Children's Elixsure IB, Children's Motrin, Ibu-Tab, Ibuprohm, Motrin IB, Motrin Migraine Pain, Profen, Tab-Profen, Duexis (combination with famotidine), Reprexain (combination with hydrocodone), Vicoprofen (combination with hydrocodone) indomethacin Indocin, Tivorbex ketoprofen No brand name currently marketed ketorolac Sprix mefenamic acid Ponstel meloxicam Mobic nabumetone No brand name currently marketed naproxen Aleve, Anaprox, Anaprox DS, EC-Naprosyn, Naprelan, Naprosyn, Treximet (combination with sumatriptan), Vimovo (combination with esomeprazole) oxaprozin Daypro piroxicam Feldene sulindac Clinoril tolmetin No brand name currently marketed
[0006] Prostaglandins play a key role in the inflammatory response, with their presence significantly increased in inflamed tissue, contributing to pain and fever by raising temperature, and dilating blood vessels, which causes redness and swelling in the place they are released. NSAIDs are competitive site inhibitors of both cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2), and thereby reduce the synthesis of prostaglandins. By reducing production of prostaglandins NSAIDs help relieve the discomfort of fever and reduce inflammation and the associated pain. NSAIDS are usually used for the treatment of acute and chronic conditions characterised by pain and inflammation, such as osteoarthritis, rheumatoid arthritis, headache and migraine, and fever. However, NSAIDS also present considerable issues around safety, and can exhibit gastrointestinal, renal and cardiovascular toxicities. For example, aspirin can cause gastric bleeding within several days of use and in July 2015, the FDA repeated a previous warning about the heart hazards of common NSAID pain relievers, excluding aspirin. These include ibuprofen (Advil, Motrin) and naproxen (Aleve) and prescription-only NSAIDs.
[0007] Accordingly, there is a need for further alternative anti-inflammatory therapies.
SUMMARY
[0008] It has now been found that certain combinations of mussel lipid and krill oil may advantageously provide an additive or synergistic inhibitory effect against the production or release of one or more pro-inflammatory mediators implicated in the inflammatory process. In some embodiments, combinations of the present disclosure may therefore afford new therapeutic treatments for disorders characterised by inflammation, or having an inflammatory component. In some embodiments, combinations of the present disclosure may afford new therapeutic treatments for pain, such as pain associated with inflammation.
[0009] Thus, in one aspect, there is provided a combination comprising mussel lipid and krill oil. The mussel lipid and krill oil combination may be adapted for separate or simultaneous administration to a subject. In some embodiments, the combination is a composition comprising mussel lipid and a krill oil.
[0010] In another aspect, there is provided a combination consisting, or consisting essentially, of mussel lipid and krill oil. The mussel lipid and krill oil combination may be adapted for separate or simultaneous administration to a subject. In some embodiments, the combination is a composition consisting, or consisting essentially of mussel lipid and a krill oil.
[0011] In another aspect there is provided a combination comprising mussel lipid and krill oil for use in therapy. In some embodiments, there is provided a combination comprising mussel lipid and krill oil for use in treating inflammation in a subject. There is also provided a combination comprising mussel lipid and krill oil for use in treating pain in a subject. The mussel lipid and krill oil combination may be adapted for separate or simultaneous administration to a subject. In some embodiments, the combination is a composition comprising mussel lipid and a krill oil.
[0012] In another aspect, the disclosure provides a method of treating inflammation in a subject in need thereof, comprising administering to said subject a combination comprising mussel lipid and krill oil. The disclosure also provides a method of treating pain in a subject in need thereof, comprising administering to said subject a combination comprising mussel lipid and krill oil. The mussel lipid and krill oil combination may be adapted for separate or simultaneous administration to a subject. In some embodiments, the combination is a composition comprising mussel lipid and a krill oil.
[0013] In another aspect, the disclosure provides use of mussel lipid and krill oil in the manufacture of a combination medicament for treating inflammation. The disclosure also provides use of mussel lipid and krill oil in the manufacture of a combination medicament for treating pain. The medicament may be adapted for separate or simultaneous administration. In some embodiments, the combination is a composition comprising mussel lipid and a krill oil.
[0014] In another aspect, the disclosure provides a combination agent for treating inflammation, said combination comprising mussel lipid and krill oil. The disclosure also provides a combination agent for treating pain, said combination comprising mussel lipid and krill oil. The mussel lipid and krill oil may be adapted for separate or simultaneous administration to a subject. In some embodiments, the combination agent is a composition comprising mussel lipid and a krill oil.
[0015] In some embodiments, the mussel lipid is in the form of a dried mussel powder. In other embodiments, the mussel lipid is in the form of a lipid extract obtained from mussel ("mussel lipid extract"). In still further embodiments, the mussel lipid may be in the form of a combination or composition of dried mussel powder and mussel lipid extract.
[0016] In some embodiments, the krill oil has a phospholipid content in the range of about 40-99% w/w, and in further embodiments a phospholipid content in the range of about 50-99% w/w, such as about 60-80% w/w.
[0017] In some embodiments, the combinations of the disclosure may be useful in treating one or more disorders in a subject, wherein the disorder has an inflammatory component, and whereby the inhibition of one or more pro inflammatory molecules is therapeutically beneficial. In some embodiments, the combinations may be suitable for treating one or more chronic disorders.
[0018] In some embodiments, such as for use in treating chronic inflammation, the combinations may eliminate, avoid or otherwise mitigate the extent, severity or duration of one or more adverse effects associated with commonly available NSAIDS.
[0019] In another aspect, there is provided a process for preparing krill oil having a phospholipid content of about 50% or greater, such as about 60% or greater, comprising the steps of:
(a) contacting a krill biomass feed material with a mixture of CO.sub.2 and ethanol, to extract a krill oil; and (b) contacting said krill oil with CO.sub.2 to extract at least a proportion of non-polar lipid components such that the oil has a phospholipid content of at least 50% w/w.
[0020] In some embodiments, the krill oil obtained from this process has a phospholipid content of about 60% w/w or greater, such as at least about 65% w/w, or 70% w/w, or 75% w/w, or 80% w/w, or 85% w/w, or 90% w/w.
[0021] The disclosure also relates to processes for enriching the phospholipid content of a krill oil.
[0022] Thus, in a further aspect, there is provided a process for increasing the phospholipid content of a krill oil having a phospholipid content of less than 50% w/w to about 50% w/w or greater, comprising the step of contacting a krill oil having a phospholipid content of less than 50% w/w with CO.sub.2 to selectively remove non-polar lipid components.
[0023] In some embodiments, the preliminary krill oil has a phospholipid content of less than about 50%, w/w, such as less than about 40%, or less than about 30% w/w or 20% w/w. In some embodiments, the enriched oil so obtained has a phospholipid content of at least about 55% w/w, or at least about 60% w/w, or at least about 65% w/w or at least about 70% w/w or at least about 75% w/w or at least about 80% w/w or at least about 85% w/w or at least about 90% w/w.
[0024] A further embodiment provides krill oil having a phospholipid content of about 50% w/w or greater, (e.g. .gtoreq.60% w/w) obtained by a process of the disclosure.
[0025] Still further embodiments provide krill oil having a phospholipid content of about 50% or 60% w/w or greater for use in the combinations and compositions described herein.
FIGURES
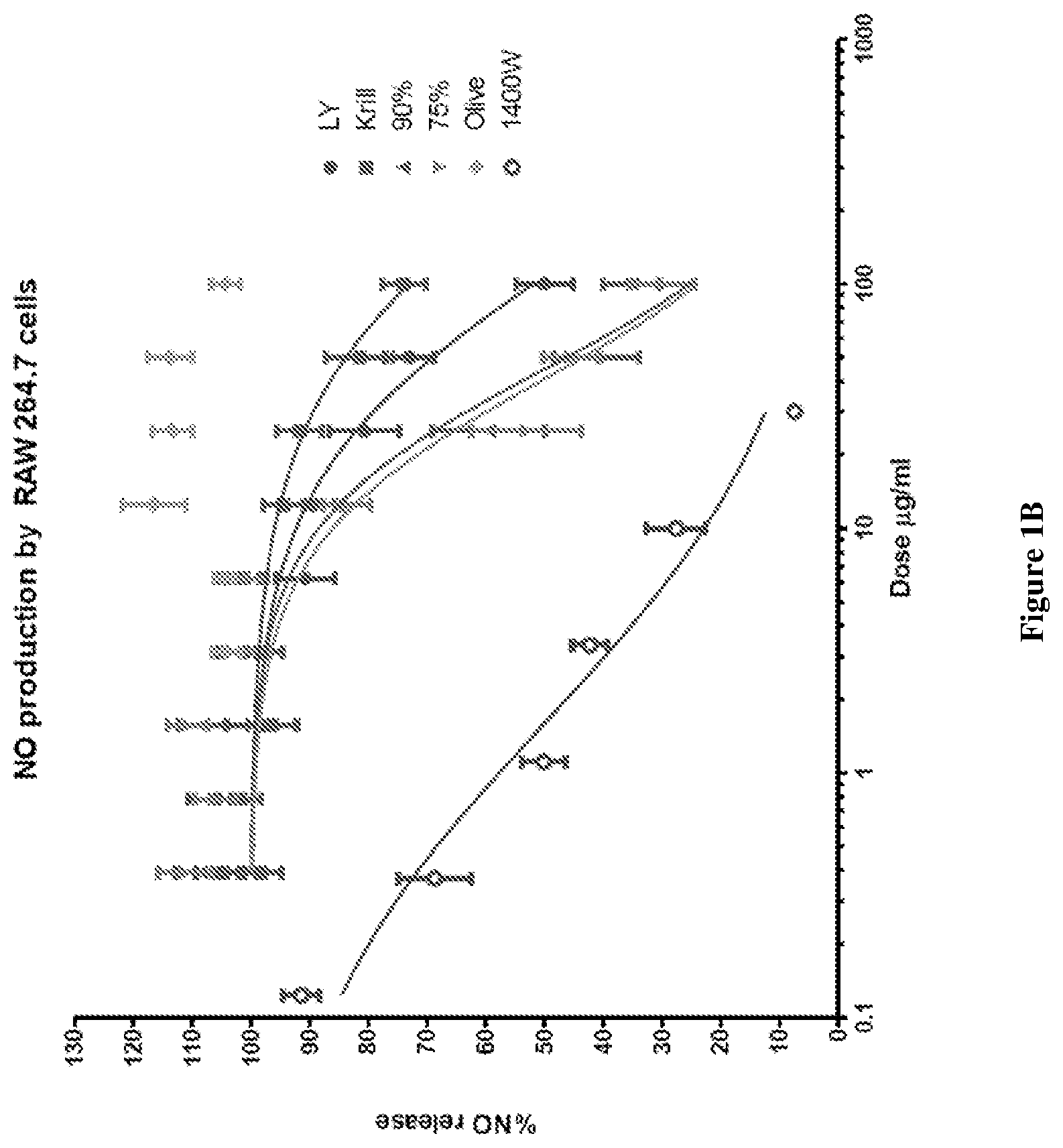
[0026] FIG. 1A graphically depicts the NO inhibition effect in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, olive oil and N-(3-(Aminomethyl)benzyl)acetamidine (1400W).
[0027] FIG. 1B graphically depicts NO release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, olive oil and N-(3 -(Aminomethyl)benzyl)acetamidine (1400W).
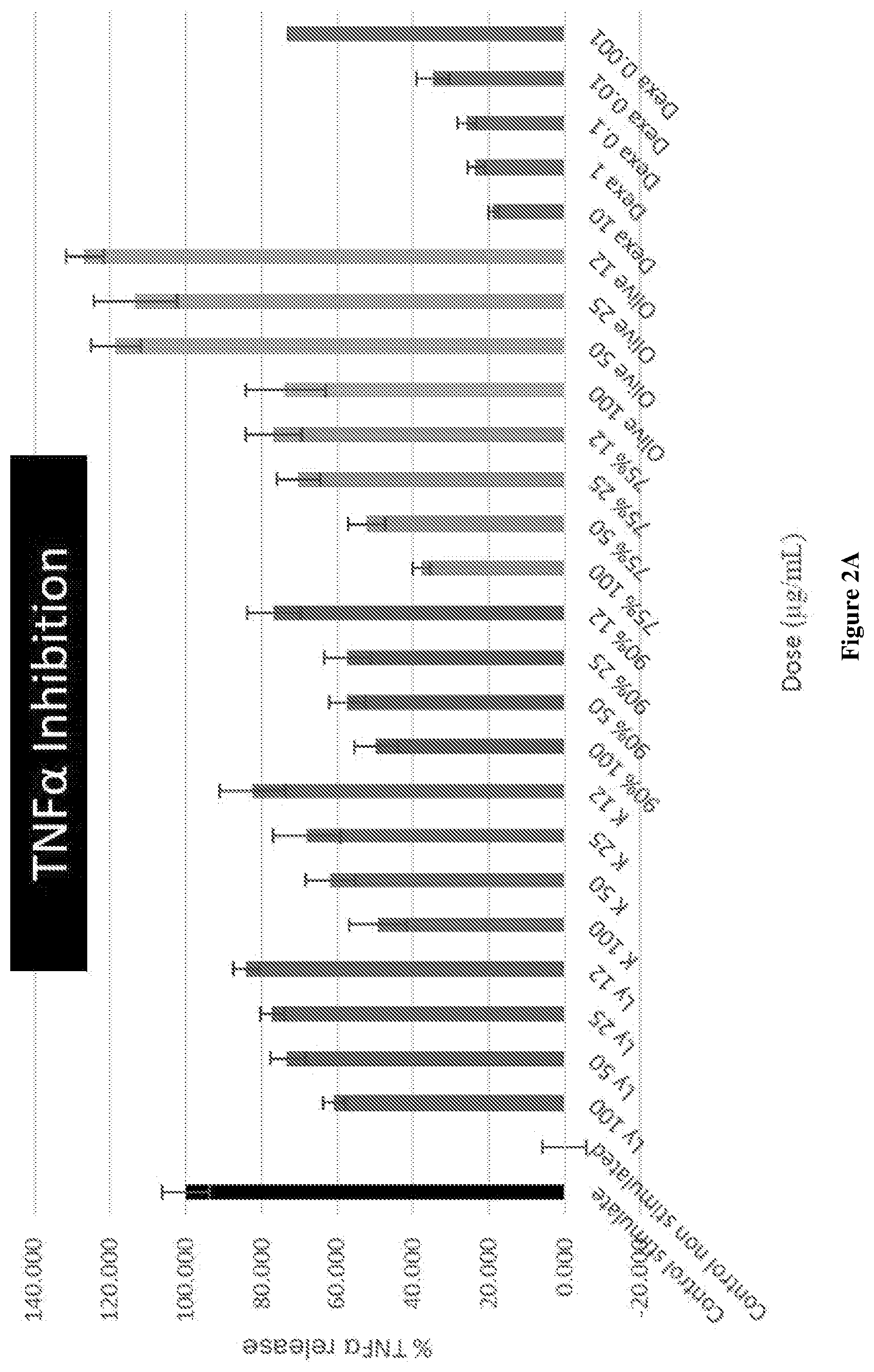
[0028] FIG. 2A graphically depicts the TNF.alpha. inhibition effect in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, olive oil and Dexamethasone.
[0029] FIG. 2B graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone and olive oil.
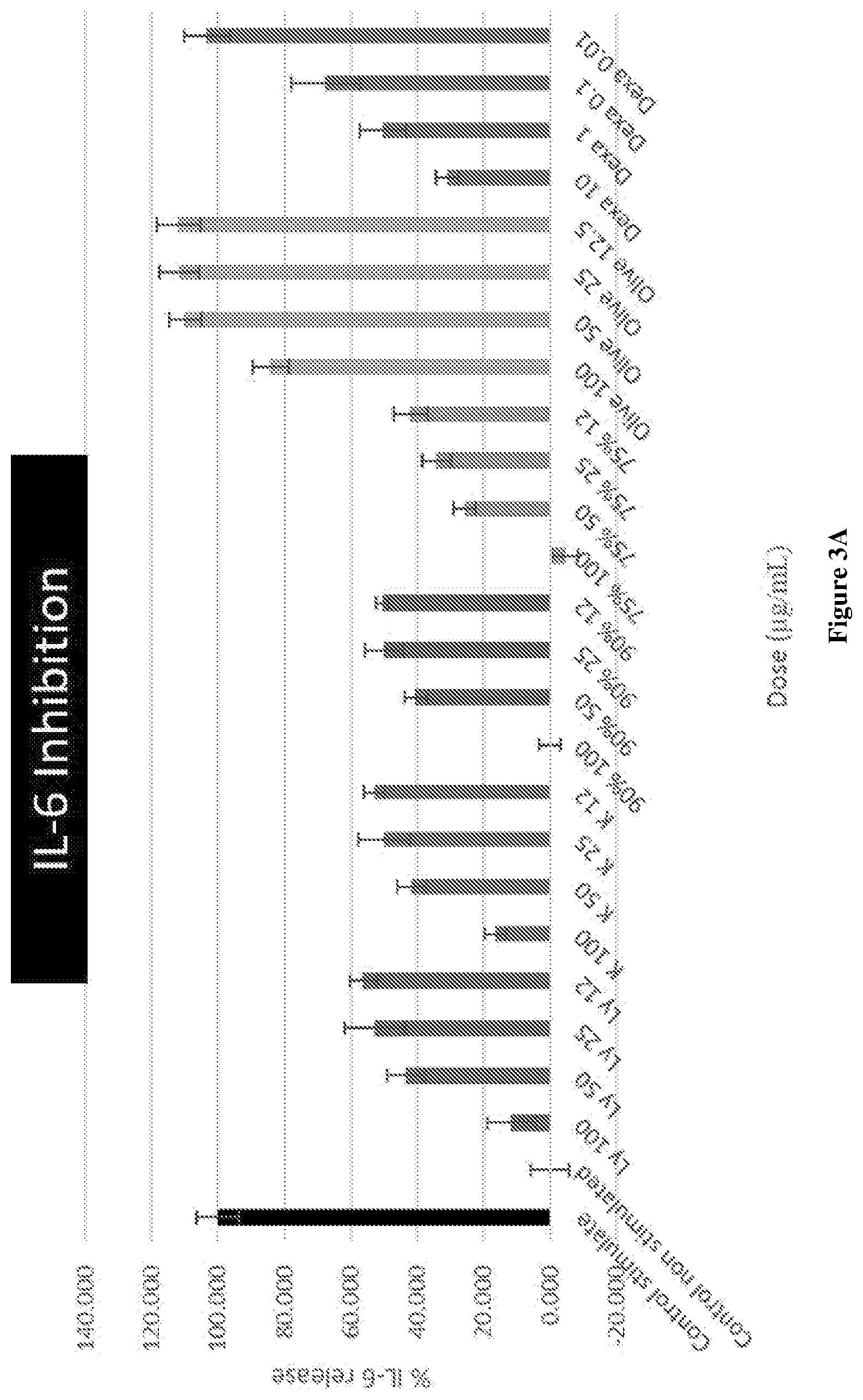
[0030] FIG. 3A graphically depicts the IL-6 inhibition effect in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, olive oil and Dexamethasone.
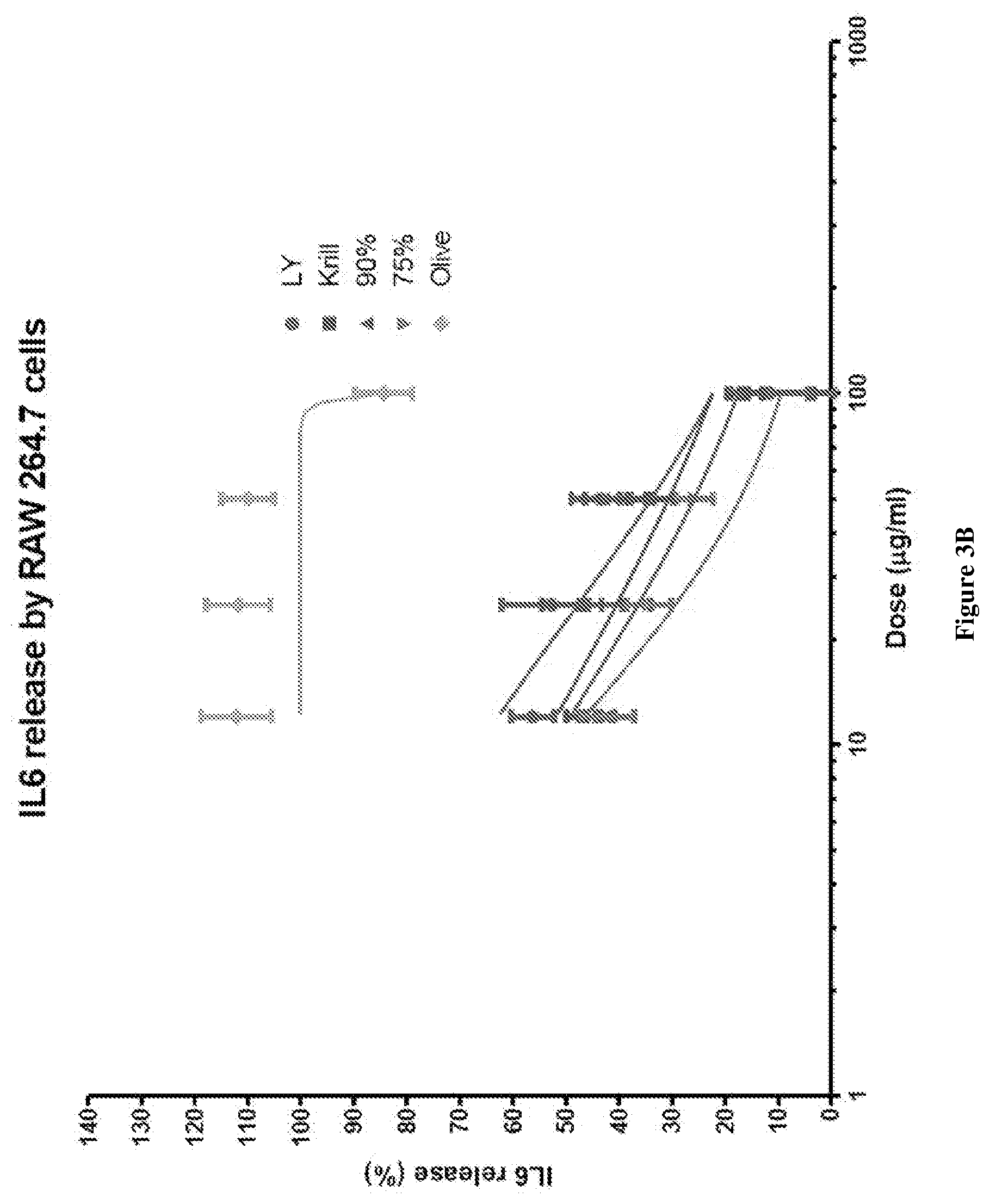
[0031] FIG. 3B graphically depicts the IL-6 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, and olive oil.
[0032] FIG. 4A graphically depicts the PGE2 inhibition effect in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, olive oil and diclofenac.
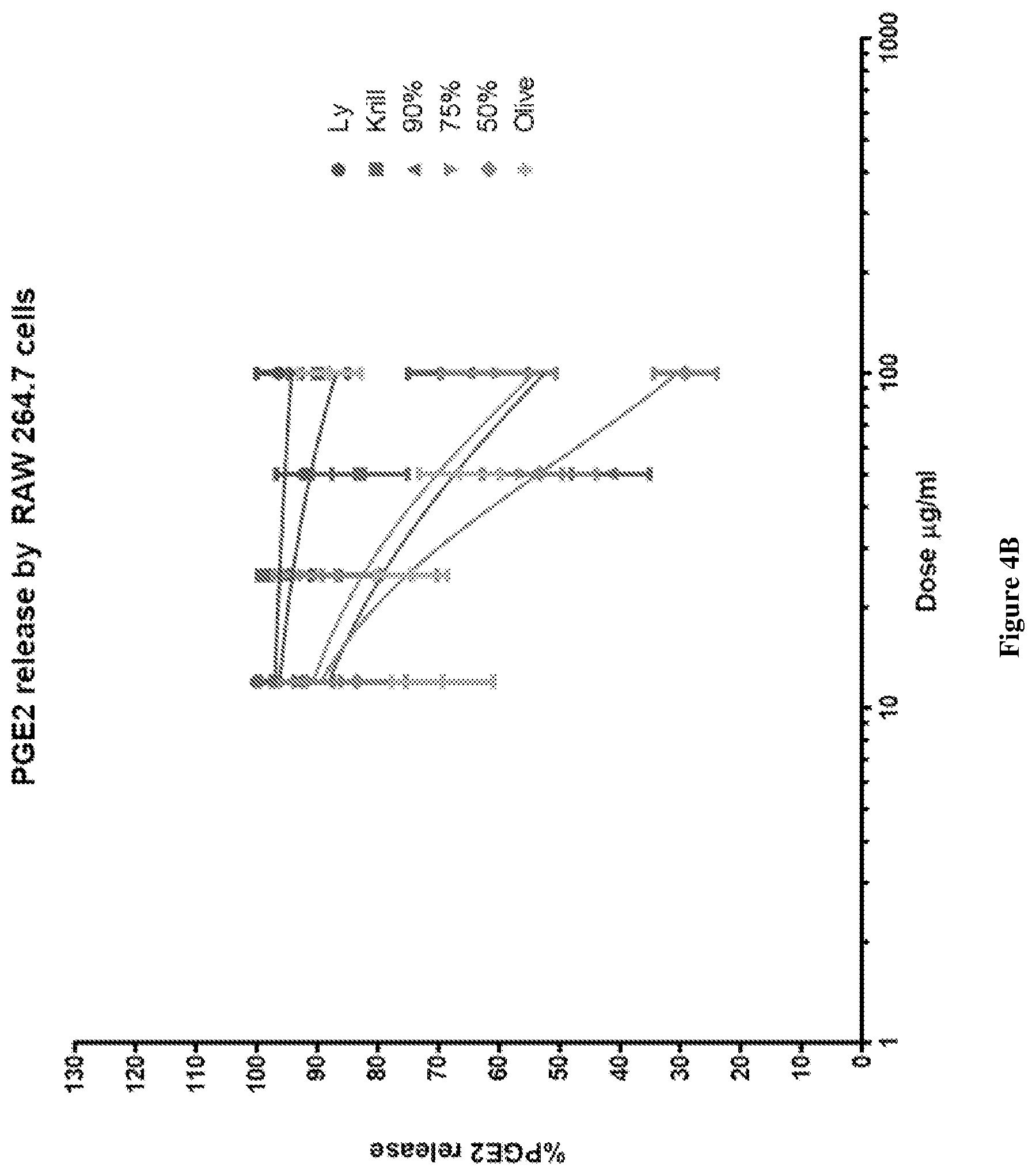
[0033] FIG. 4B graphically depicts the PGE2 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil, each of mussel lipid extract and krill oil alone, and olive oil.
[0034] FIG. 5A graphically depicts NO release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY50).
[0035] FIG. 5B graphically depicts NO release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY50-LY10).
[0036] FIG. 6A graphically depicts NO release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY75-LY60).
[0037] FIG. 6B graphically depicts NO release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY60-LY45).
[0038] FIG. 7 graphically depicts the isobologram for synergistic NO inhibition by various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY10).
[0039] FIG. 8A graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY60).
[0040] FIG. 8B graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY60-LY30).
[0041] FIG. 8C graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY40-LY10).
[0042] FIG. 9A graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY70-LY55).
[0043] FIG. 9B graphically depicts TNF.alpha. release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY50-LY35).
[0044] FIG. 10 graphically depicts the isobologram for synergistic TNF.alpha. inhibition by various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY10).
[0045] FIG. 11A graphically depicts the IL-6 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY50).
[0046] FIG. 11B graphically depicts the IL-6 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY50-LY10).
[0047] FIG. 12A graphically depicts the IL-6 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY70-LY50).
[0048] FIG. 12B graphically depicts the IL-6 release (%) in lipopolysaccharide (LPS) and interferon .gamma. (IF.gamma.) stimulated RAW264.7 cells for various concentrations of combinations of mussel lipid extract and krill oil (LY50-LY30).
[0049] FIG. 13 graphically depicts the isobologram for synergistic IL-6 inhibition by various concentrations of combinations of mussel lipid extract and krill oil (LY90-LY10).
DESCRIPTION
[0050] Throughout this specification and the claims which follow, unless the context requires otherwise, the word "comprise" and variations such as "comprises" and "comprising" will be understood to imply the inclusion of a stated integer or step or group of integers but not the exclusion of any other integer or step or group of integers or steps.
[0051] Throughout this specification and the claims which follow, unless the context requires otherwise the phrase "consisting essentially of" and variations such as "consists essentially of" will be understood to indicate that the recited element(s) is/are essential i.e. necessary elements of the invention. The phrase allows for the presence of other non-recited elements which do not materially affect the characteristics of the invention but excludes additional unspecified elements which would affect the basic and novel characteristics of the invention defined.
[0052] The singular forms "a", "an" and "the" include plural aspects unless the context clearly dictates otherwise.
[0053] The term "invention" includes all disclosures, aspects, embodiments and examples as described herein.
[0054] As used herein, "about" refers to a quantity, value or parameter that may vary by as much as 10%, 5%, or 2-1% of the stated quantity, value or parameter, and includes at least tolerances accepted within the art. When used in reference to a stated whole number value, "about" may include variation of one whole number either side of the stated value, for example "50%", may include 49% and 51%. When prefacing a recited range of values, it is intended to apply to both upper and lower limits of the range.
[0055] Unless the context indicates otherwise, features described below may apply independently to any aspect or embodiment.
[0056] As used herein, "mussel lipid" refers to a lipid component extracted or obtained from the New Zealand green-lipped (NZGL) (or greenshell) mussel (Perna canaliculus). The mussel lipid may comprise one or more of polyunsaturated long chain fatty acids (PUFAs), such as ALA, ETAs, EPA and DHA, sterols, sterol esters, triglycerides, non-polar lipds carotenoids and other components of (NZGL) mussel meat. The mussel lipid may be in the form of dried mussel powder, or a lipid fraction extracted from the mussel meat ("mussel lipid extract"). It is also envisaged that "mussel lipid" encompasses a mixture of a mussel powder and mussel lipid extract, for example a mussel lipid may be supplemented by the addition of mussel powder or vice versa. In some embodiments, the mussel lipid is an isolated lipid fraction.
[0057] The mussel lipid powder may be prepared from fresh (raw), frozen or heat treated NZGL mussel meat by any suitable drying means (e.g. freeze drying, flash drying or vacuum drying) and pulverizing means. In addition to fatty acids (including ALA, ETAs, EPA and DHA) the mussel powder obtained by drying mussel meat will also contain other potentially beneficial components, including minerals, amino acids, peptides, proteins, and glycosaminoglycans (for example chondroitin-4-sulfate and chondroitin-6-sulfate). Processes for preparing mussel powder are known in the art.
[0058] The mussel lipid extract may be obtained from fresh (raw), frozen, heat treated or dried (e.g. freeze-, flash- or vacuum drum-dried) NZGL mussel meat (e.g. powdered, spray dried or pulverized form) by any suitable method, such as. solvent extraction (e.g. acetone or ethanol see for example WO2005073354 A1, the contents of which are incorporated by reference), enzyme treatment (see for example WO2006128244, the contents of which are incorporated by reference) or supercritical fluid extraction. In some embodiments, the mussel lipid extract is advantageously obtained by extraction with supercritical CO.sub.2 from dried (e.g. freeze-dried) mussel meat (optionally stabilized to prevent oxidation). An exemplary method for obtaining mussel lipid extract is described in WO 97/09992 A1, the contents of which are incorporated by reference. Other methods will be known in the art.
[0059] In preferred embodiments processes are performed under conditions such that the beneficial components, such as fatty acids, not substantially destroyed and are significantly retained, for example cold processing.
[0060] One exemplary mussel lipid extract obtained in accordance with the process described in WO 97/09992 A1, is also known as PCSO-524.RTM. (Pharmalink International Limited, Hong Kong). PCSO-524.RTM. contains added vitamin E (0.15% w/w, added as an anti-oxidant preservative) and comprises a combination of free fatty acids, triglycerides, sterol esters, non-polar lipids, and carotenoids (Sinclair, A. J. et al, 2000), and is a source of the long chain omega-3 polyunsaturated fatty acids, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), as well as other long chain fatty acids, such as 5,9,12,15-octadecatetraenoic acid, 5,9,12,16-nonadecatetraenoic acid, 7,11,14,17-eicotetraenoic acid, and 5,9,12,15,18-heneicosapentaenoic acid. PCSO-254.RTM. (formulated together with olive oil in encapsulated oral dosage form), is marketed under the banner Lyprinol.RTM. and Omega XL.RTM., (for human consumption), and Antinol.RTM. (for dogs and cats).
[0061] In some embodiments, the mussel lipid extract used in the combinations of the disclosure is formulated with vitamin E, (added, for example in an amount of about 0.2% w/w, or 0.15% w/w, or 0.1% w/w, or 0.05% w/w or about 0.03% w/w or about 0.01% w/w). In some embodiments, mussel lipid is used in the form of PCSO-254 , i.e mussel lipid extract containing 0.15% w/w vitamin E. In some embodiments the mussel lipid, optionally containing vitamin E, is further formulated with a carrier oil, such as olive oil. While in some embodiments the mussel lipid is a mussel lipid extract and contains added vitamin E, the addition of vitamin E is optional, and thus, in some embodiments, the mussel lipid extract is used neat, i.e., does not contain any other additional ingredients, such as vitamin E.
[0062] Mussel lipid, in various forms, may also be purchased from commercial suppliers.
[0063] Krill oil may be prepared from any suitable krill species, including Euphausia superba (Antarctic krill), Euphausia pacifica (Pacific krill), Maganycitiphanes norvegica (Northern krill), Euphausia crystallorophias (ice krill), Euphausia frigida, Euphausia longirostris, Euphausia triacantha and Euphausia vallentini. In some preferred embodiments, the krill oil is obtained from Euphausia superba.
[0064] Marine lipids contain fatty acids, particularly omega-3 fatty acids such as EPA and DHA, in free and triglyceride form. Similarly, krill oil is also rich in omega-3 fatty acids, however, krill oil contains significant amounts of phospholipids, where the fatty acids are attached to a phosphate head group via a glycerol moiety. It is this phospholipid bound form of the fatty acids that is more efficiently taken up into cellular membranes than the triglyceride form, and thereby more readily bioavailable. Typical phospholipids found in krill oil may include: phosphatidylcholine, alkyl acyl phosphatidylcholine, phosphatidylinositol, phosphatidylserine, lysophosphatidylcholine, lyso alkyl acyl phosphatidylcholine, phosphatidylethanolamine, alkyl acyl phosphatidylethanolamine, cardiolipin+N-acyl phosphatidylethanolamine, lysophosphatidylethanolamine, and lyso alkyl acyl phosphatidylethanolamine. Krill oil also contains appreciable amounts of astaxanthin, an antioxidant, which is also responsible for its red colour.
[0065] In some embodiments, the krill oil contains at least about 1% w/w, 5% w/w, 10% w/w, or at least about 20% w/w phospholipids. In further embodiments, the oil contains at least about 25% w/w or at least about 30% w/w, or at least about 35% w/w, or at least about 40% w/w, or at least about 45% w/w, or at least about 50% w/w, or at least about 55% w/w, or at least about 60% w/w, or at least about 65% w/w or at least about 70% w/w, or at least about 75% w/w, or at least about 80% w/w or at least about 85% w/w phospholipids, or at least about 90% w/w phospholipids, or at least about 95% w/w phospholipids, or at least about 97% w/w phospholipids, or at least about 98% w/w phospholipids, or at least about 99%, w/w phospholipids. In some embodiments, the krill oil has a phospholipid content in the range of about 40-99% w/w. In some further embodiments, the krill oil has a phospholipid content in the range of about 60-99% w/w, for example in the range of about 65-90% w/w. As referred to herein, "enriched" krill oil refers to krill oil having a phospholipid content of least about 60% w/w. Phospholipid content may be determined by any suitable means in the art, for example .sup.31P NMR analysis.
[0066] Methods for preparing krill oil, including krill oil enriched in phospholipids are known in the art. Typically, fresh, frozen and/or heat treated krill (e.g. Euphasia superba or Euphasia pacifica) biomass may be extracted using solvents (e.g, alcohols, such as ethanol; ketones, such as acetone; or dimethoxyethane) and/or supercritical fluid (e.g. CO.sub.2). Some non-limiting exemplary processes for preparing krill oil are described in U.S. Pat. Nos. 9,028,877, 9,375,453, 6,800,299, 8,828,447, 9,150,815, 8,383,845, WO2007/123424, WO2011/050474, WO2015/104401 and WO2015/121378, the contents of which are incorporated herein by reference. Further methods are also described herein. Krill oil may also be purchased from commercial suppliers.
[0067] In some advantageous embodiments, the krill oil has a water content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. In some embodiments, the krill oil has a residual extraction solvent content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. In further embodiments, the krill oil has a water content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less, and a residual extraction solvent content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. Water and solvent may be removed by any suitable means, such as very short duration or gentle heating (e.g. 30 min, or 1 hour, or 2 hours or 3 hours, at a temperature of about or less than about 60.degree. C., or 50.degree. C., or 40.degree. C., and preferably such that the integrity of the constituents is not substantially compromised), nitrogen stream or lyophilisation (freeze-drying).
[0068] The inhibitory activity of the combinations against one or more inflammatory mediators such as nitrous oxide (NO), cytokines, such as interleukins, (e.g. IL-6), prostaglandins (e.g. PG-E.sub.2), and TNF.alpha., may be useful in treating one or more disorders or symptoms in a subject whereby the inhibition of one or more of such molecules is therapeutically beneficial. In particular, the combinations of the disclosure may be useful in treating excessive acute, or chronic inflammation, and/or one or more of the symptoms associated therewith, such as pain, fever, redness and swelling. In some embodiments the combinations may be useful in treating inflammation in disorders where the pathology includes an inflammatory component, and/or pain associated with such disorders. Some non-limiting examples of disorders that include an inflammatory aspect include atherosclerosis, allergy, asthma, autoimmune disease (e.g. coeliac disease, psoriasis, rheumatoid arthritis, psoriatic arthritis), fibromyalgia, gout, migraine, osteoarthritis, ulcerative colitis, cancer, impaired cognition, including Alzheimer's disease, type 2 diabetes, delayed onset muscle soreness (DOMS), Crohn's disease and ankylosing spondylitis. In some embodiments, the combinations of the disclosure may be useful in treating joint pain or improving joint mobility associated with osteoarthritis or rheumatoid arthritis. In some embodiments, the combinations of the disclosure may be useful in treating disorders in which inhibition of PGE.sub.2 may be beneficial, such as rheumatoid arthritis, migraine and pain (which may be nociceptive (somatic or visceral) pain, and/or neuropathic pain).
[0069] It will be appreciated that the combinations described herein may be adapted for separate or simultaneous administration. Where adapted for simultaneous administration, the combination may be provided and/or administered as an intimate composition or a mixture which comprises both the mussel lipid extract and krill oil, or as discrete dosage forms of each combination component. Where the mussel lipid extract and krill oil are each provided and/or administered separately, they may be administered simultaneously, one after the other, or each at a different time.
[0070] In further embodiments, the mussel lipid and krill oil may optionally be formulated, either together or individually, in combination with one or more pharmaceutically acceptable carriers and/or additives. Some examples of suitable carriers are edible oils, such as olive oil, castor oil, flaxseed oil, grapeseed oil, fish oil (e.g. tuna oil), canola oil, vegetable oil, sunflower oil, chia oil, soybean oil, sesame oil, algal oil, and mixtures thereof. One or more optional additives, such as anti-oxidants, vitamins (such as fat soluble vitamins (A, D, E and K), or water soluble vitamins (B1, B2, B3, B5, B6, B7, B9, B12, C), dietary minerals, amino acids, odour and taste masking agents, emulsifiers, pharmaceutically acceptable alcohols, (e.g. ethanol, glycerol, propylene glycol, and polyethylene glycol) or other viscosity modifiers, surfactants (e.g. polysorbates), suspension agents, lactose, dextrose, sucrose, mannitol, sorbitol, glucose, lubricants, binders, starches, absorption enhancers and preservatives etc. may also be included. A carrier or additive may perform one or more functions. The mussel lipid and/or krill oil may optionally be further supplemented or combined with one or more additional purified or partially purified components of mussel and krill oil, such as, astaxanthin and esters thereof, fatty acids (e.g. EPA, DHA), either in free acid, acid ester, triglyceride or phospholipid form, sterols, sterol esters, amino acids, peptides and proteins, and glycosaminoglycans (e.g. chondroitin sulfates). Other anti-inflammatory foods, such as whole ground form, or extracts thereof, e.g. turmeric (curcumin), ginger, garlic, cloves etc., may also be optionally incorporated.
[0071] The formulated combinations may be prepared according to methods known in the art. Such methods include the step of intimately bringing the mussel lipid extract and/or krill oil into association with the carrier, optionally together with one or more additive ingredients. It will be understood that any carrier or additive will be pharmaceutically acceptable.
[0072] Thus, in some embodiments, the mussel lipid and krill oil are formulated, either separately or together, with a carrier oil, such as olive oil. In some embodiments, the carrier oil comprises from about 10% w/w to about 90% w/w, such as about 20% w/w to about 80% w/w of the combination or composition. In further embodiments, the carrier oil comprises about 25% w/w, or about 30% w/w, or about 35% w/w, or about 40% w/w, or about 45% w/w, or about 50% w/w, or about 55% w/w, or about 60% w/w, or about 65% w/w, or about 70% w/w, or about 75% w/w of the combination or composition. In some embodiments, the weight ratio of carrier oil to combined amount of mussel lipid and krill oil is about 3:1, or about 2.5:1, or about 2:1, or about 1.5:1, or about 1:1, or about 1:1.5, or about 1:2, or about 1:2.5 or about 1:3.
[0073] While any form of administration is contemplated herein, such as oral, parenteral, topical, transdermal or subdermal, advantageously, in some embodiments the combinations of the disclosure may be provided and/or administered in an oral dosage form. In some embodiments the combinations can be presented in a bulk form, for example as liquids, syrups, pastes, semi-solid waxes, dispersions, suspensions, emulsions (e.g. water-in-oil or oil-in-water), pulverised powders or microencapsulated powders, from which individual dosages can be measured. Measurement and/or administration may be by any means, such as spoon or scoop, syringe, dropper, or measuring cup. Measured dosages may be administered to the subject directly or mixed through, poured on, or sprinkled on food or beverages.
[0074] In other embodiments, the combinations are advantageously presented in unit oral dosage forms, i.e. a fixed dosage form. Some examples of suitable unit oral dosages include individually packaged ampoules, tubes, filled syringes, sachets, chews and capsules (including hard and soft gel capsules).
[0075] One example, of a suitable unit oral dosage form is a capsule, in the form of a hard or soft shell. The shell may comprise one or more of gelatin, pullulan, hypromellose, PVA copolymer, carrageenan or other saccharide component such as starch or cellulose, or mixtures thereof, and may further include colouring agents, opacifying agents, plasticizers (e.g. sorbitol, xylose, maltitol and glycerine) etc. Methods for encapsulating marine oils and lipids, such as mussel lipid and krill oil, are known in the art. See for example WO2015/121378, the contents of which are incorporated herein by reference. In some embodiments, where the krill oil is encapsulated separately, the krill oil may be encapsulated in the absence of optional additional agents, such as viscosity modifying agents, that is to say, the capsule fill consists essentially of krill oil.
[0076] In some advantageous embodiments, the mussel lipid and krill oil combinations of the disclosure are presented in soft gel capsule form, for example a soft gel capsule comprising both the mussel lipid and krill oil, or individual soft gel capsules, where the mussel lipid and krill oil are separately encapsulated, optionally together with suitable carriers and/or additives. Suitable soft gel capsules may be prepared from gelatin (or alternatively, saccharide sources such as pullulan and hypromellose), optionally with one or more plasticizers such as sorbitol and glycerine (glycerol), and additives such as colouring and opacifying agents. In one example, a soft gel capsule shell may comprise gelatin, and one or both of sorbitol and glycerine.
[0077] Microencapsulation is a method by where tiny droplets or particles are surrounded by a coating wall or are embedded in a matrix to form a powder, with the coating or matrix forming a functional barrier that avoids or reduces the propensity for chemical reactions, such as oxidation. In addition, it may provide a potential taste or odour masking role. Thus, in some embodiments, the mussel lipid and/or krill oil may be microencapsulated, either separately or together, to form a powder. Commonly used microencapsulation methods include emulsification, spray-drying, freeze-drying, co-axial electrospray, extrusion, coacervation, supercritical fluid technology, and in situ polymerization. Coating materials include natural and synthetic polymers, carbohydrates (e.g. starches, glucose), proteins (e.g. casein, gelatin) and mixtures thereof (See, for example, Bakry, A. M., et al, Comprehensive Reviews in Food Science and Food Safety, 15, 143, 2016, and the references cited therein, WO2014/170464 and WO2014/169315, the contents of which are included herein by reference). The mussel lipid and/or krill oil may be microencapsulated with one or more carriers or additives as described above. The microencapsulated mussel lipid and/or krill oil powder may be further encapsulated, for example in a hard shell capsule unit dosage form. The powdered microencapsulated lipid or oil may be optionally combined with one or more carriers or additives.
[0078] In some embodiments the combinations of the disclosure may be taken with food or beverages, for example by sprinkling, stirring, mixing or other means of applying or incorporating the mussel lipid extract and krill oil combination into or onto a food or beverage. Thus, the combinations may be provided in a format for incorporation into or onto a beverage or foodstuff. In some embodiments the combination, may also be formulated in the preparation of foodstuffs and beverages to provide functional foods.
[0079] In some embodiments, the mussel lipid and krill oil are formulated, either separately or together, with a carrier oil, such as olive oil, optionally with an antioxidant (e.g. vitamin E).
[0080] Subjects to be treated by the combinations of the disclosure include mammalian subjects, such as humans, primates, felines, canines, bovines, equines, porcines, leporines, ovines, and caprines, and include livestock animals (e.g. cows, horses, sheep, pigs and goats), companion animals (e.g. dogs, cats, rabbits, guinea pigs), and captive wild animals. Laboratory animals such as rabbits, mice, rats, guinea pigs and hamsters are also contemplated as they may provide a convenient test system.
[0081] Any of the dosage forms described above may be applicable for human or veterinary use as appropriate.
[0082] A treatment effective amount is intended to include an amount of the combination which, when administered according to the desired dosing regimen, is jointly effective to at least partially attain the desired therapeutic effect, including one or more of: alleviating, eliminating or reducing the duration, severity and/or frequency of inflammation and/or one or more symptoms of inflammation (e.g. heat, pain, swelling, redness), preventing or delaying the onset of, inhibiting the progression of, or halting or reversing (partially or altogether) the onset or progression of the particular disorder or condition being treated.
[0083] Suitable dosage amounts and dosing regimens can be determined by the attending physician or veterinarian, and may depend on the particular condition/symptoms being treated, the severity of the condition as well as the general age, health and weight of the subject. Suitable daily dosage amounts of the mussel lipid and/or krill oil may independently lie in the range of from about 10 mg to about 10 g, for example about 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 100 mg, 150 mg, 200 mg, 250 mg, 300 mg, 350 mg, 400 mg, 450 mg, 500 mg, 550 mg, 600 mg, 650 mg, 700 mg, 750 mg, 800 mg, 850 mg, 900 mg, 950 mg 1 g, 1.1 g, 1.2g, 1.3 g, 1.4,g, 1.5 g, 1.6 g, 1.7 g, 1.8 g, 1.9 g, 2.0 g, 2.1 g, 2.2 g, 2.3 g, 2.4 g, 2.5 g, 2.6 g, 2.7 g, 2.8 g, 2.9 g, 3.0 g, 3.2 g 3.5 g, 3.7 g, 4.0 g, 4.5 g, 5.0 g, 5.5 g, 6.0 g, 6.5 g, 7.0 g, 7.5 g, 8.0 g, 8.5 g, 9.0 g, or about 9.5 g. In some further embodiments, daily dosages of the combination may lie in the range of from about 20 mg to about 15 g, for example, about 20 mg, 30mg, 40 mg, 50 mg, 100 mg, 150 mg, 200 mg, 250 mg, 300 mg, 350 mg, 400 mg, 450 mg, 500 mg, 550 mg, 600 mg, 650 mg, 700 mg, 750 mg, 800 mg, 850 mg, 900 mg, 950 mg 1 g, 1.1 g, 1.2 g, 1.3 g, 1.4, g, 1.5 g, 1.6 g, 1.7 g, 1.8g, 1.9g, 2.0 g, 2.1 g, 2.2 g, 2.3 g, 2.4 g, 2.5 g, 2.6 g, 2.7 g, 2.8 g, 2.9 g, 3.0 g, 3.2 g 3.5 g, 3.7 g, 4.0 g, 4.5 g, 5.0 g, 5.5 g, 6.0 g, 6.5 g, 7.0 g, 7.5 g, 8.0 g, 8.5 g, 9.0g, 9.5 g, 10.0 g, 10.5 g, 11.0 g, 11.5 g, 12.0 g, 12.5 g, 13.0 g, 13.5 g, 14.0 g, or 14.5 g.
[0084] In some embodiments, individual unit dosages (e.g. soft gel capsule) may contain about 10 mg, 20 mg, 25 mg 30 mg, 40 mg, 50 mg, 60 mg, 70 mg, 75 mg 80 mg, 90 mg, 100 mg, 110 mg, 120 mg, 125 mg, 130 mg, 140 mg, 150 mg, 160 mg, 165 mg, 170 mg, 175 mg, 180 mg, 190 mg, 200 mg, 210 mg, 220 mg, 225 mg, 230 mg, 240 mg, 250 mg, 260 mg, 265 mg, 270 mg, 275 mg, 280 mg, 290 mg, 300, mg 310 mg, 320 mg, 325 mg, 330 mg, 340 mg, 350 mg, 360 mg, 365 mg, 370 mg, 375 mg, 380 mg, 390 mg, 400, mg, 410 mg, 420 mg, 425 mg, 430 mg, 440 mg, 450 mg, 460 mg, 465 mg, 470 mg, 475 mg, 480 mg, 490 mg, or about 500 mg of the combination, optionally formulated together with a carrier oil (e.g olive oil). In some further embodiments thereof, the combination further comprises vitamin E. In some further embodiments, thereof, the combinations comprise mussel lipid (e.g. as PCSO-524) in an amount in the range of about 10% to about 90% w/w of the total amount of total mussel lipid and krill oil, and comprise krill oil in an amount in the range of about 90% to about 10% w/w of total mussel lipid and krill oil, that is, a weight ratio of mussel lipid to krill oil of from about 10:90 to 90:10, for example, mussel lipid to krill oil in a weight ratio of about 15:85, or about 20:80, or about 25:75, or about 30:70, or about 35:65, or about 40:60, or about 45:55, or about 50:50, or about 55:45, or about 60:40, or about 65:35, or about 70:30, or about 75:25, or about 80:20, or about 85:15.
[0085] Dosages may be conveniently administered once daily, or daily dosages may be divided and administered multiple times (e.g. two, three or four times) daily. In some embodiments, the combinations of the disclosure may be administered one, two, three or more times weekly, for example on alternate days. In some embodiments, treatment may be continuous or long term, for example, over a period of at least 6-12 months or at least 2-3 years, or ongoing.
[0086] Combinations of the disclosure, for example in any one of the daily dosage amounts, may comprise mussel lipid in an amount in the range of about 1% to about 99% w/w of the total amount of total mussel lipid and krill oil, and comprise krill oil in an amount in the range of about 99% to about 1% of total mussel lipid and krill oil, that is, a weight ratio of mussel lipid to krill oil of from about 1:99 to 99:1. In some embodiments the combinations comprise mussel lipid in an amount in the range of about 5% to about 95% w/w of the total amount of total mussel lipid and krill oil, and comprise krill oil in an amount in the range of about 95% to about 5% of total mussel lipid and krill oil, that is, a weight ratio of mussel lipid to krill oil of from about 5:95 to 95:5. In some embodiments the combinations comprise mussel lipid in an amount in the range of about 10% to about 90% w/w of the total amount of total mussel lipid and krill oil, and comprise krill oil in an amount in the range of about 90% to about 10% w/w of total mussel lipid and krill oil, that is, a weight ratio of mussel lipid to krill oil of from about 10:90 to 90:10. In still further embodiments, the combinations comprise mussel lipid to krill oil in a weight ratio of about 15:85, or about 20:80, or about 25:75, or about 30:70, or about 35:65, or about 40:60, or about 45:55, or about 50:50, or about 55:45, or about 60:40, or about 65:35, or about 70:30, or about 75:25, or about 80:20, or about 85:15.
[0087] While the combinations of the disclosure may be administered as the sole anti-inflammatory therapy for any one or more disorders, they may also be administered in conjunction with an administration regimen of one or more NSAIDS, such as celecoxib, diclofenac, diflunisal, etodolac, fenoprofen, flurbiprofen, ibuprofen, indomethacin, ketoprofen, telorolac, mefenamic, meloxicam, nabumetone, naproxen, oxaprozin, piroxicam, sulindac, and tolmetin. In some embodiments, combinations of the disclosure may eliminate or reduce the potential adverse effects associated with NSAIDS, for example, by eliminating or substantially eliminating the need for additional NSAIDS therapy, or by reducing the dosage amounts and/or dosing frequency of NSAIDS required to achieve a beneficial therapeutic effect
[0088] As discussed above, in one or more embodiments, the krill oil used in the combinations disclosed herein may advantageously have a phospholipid content of at least about 50% w/w, or higher, preferably at least about 60% w/w, or higher. Many prior art processes for extracting a high phospholipid (e.g. greater than about 50% or 60% w/w) krill oil from krill meal use a combination of CO.sub.2 and CO.sub.2/ethanol. However, it is well recognised that the higher the phospholipid content of a krill oil, the more viscous the krill oil is, with krill oils having a content of about 60% or greater typically presenting as a viscous paste at ambient temperatures. This presents manufacturing challenges, particularly on an industrial or commercial scale of oil production, as higher temperatures are required to evaporate the solvents used in the extraction process from the viscous material, with heat related damage to the oil more likely to occur. Additionally, increased pressures are then required to transfer the oil from the extraction tanks to packaging tanks.
[0089] One exemplary prior art process is described in WO2007123424. This document describes a twostep process, whereby a feed material is first extracted with pure CO.sub.2 to extract out only neutral lipids (i.e. non-polar triglycerides), leaving behind a material that is, by virtue of the removal of the non-polar triglycerides, phospholipid-enriched. This phospholipid enriched material is then extracted with, CO.sub.2+.gtoreq.10% ethanol co-solvent to extract the polar phospholipids and remaining non-polar triglycerides together from the krill feed biomass. This method inefficiently uses plant capacity at commercial scale, as during both stages of the process much of the volume in the high pressure extractors is filled with non-extractable protein, carbohydrates and ash components in the feed biomass. It is also fundamentally a less efficient batch operation (compared to more efficient continuous operation), which may adversely impact process costs. Effectively, enrichment to deliver a final polar phospholipid content in the finished oil is achieved in the first step, directly on a bulk non-homogenous solid raw material at industrial scale. Precise prior knowledge of the polar and non-polar lipid contents of the solid feed, and how well each will subsequently extract, is required for accurate final oil enrichment. In practice, uncertainty at commercial scale may translate to costly over enrichment, which then requires final blending back to specification as required. Again this adversely impacts the economics of the process.
[0090] U.S. Pat. Nos. 9,735,453, 9,028,877, 9,320,765, and 9,072,752 describe extraction of krill with CO.sub.2 or CO.sub.2 plus approximately 5% ethanol to extract neutral (non-polar triglyceride) lipids, followed by CO.sub.2/.about.20% ethanol to extract from the bulk non-homogenous solid material a krill oil with high amounts of phospholipids, astaxanthin esters and/or omega-3 fatty acids. These processes share the disadvantages described above.
[0091] The present disclosure now describes a process that, in some embodiments, may reduce, minimise or eliminate one or more of the disadvantages discussed above when preparing high, or enriched phospholipid-containing krill oil according to prior art methods, particularly on an industrial or commercial scale (e.g. when manufacturing batches of oil in the order of at least about 50, 100, 200, 300 or 500 kg and greater). Thus, the present disclosure also provides a process for preparing krill oil with a phospholipid content of at least about 50% w/w, or higher, preferably to a level of at least about 60% w/w or higher, and a process for enriching a lower phospholipid content (less than about 50% w/w) oil to a level of at least about 50% w/w or higher, preferably to a level of at least about 60% w/w or higher.
[0092] In some embodiments, the disclosure provides a 2-step process for preparing a krill oil having a phospholipid content of at least about 50% w/w wherein a first step involves extracting a first krill oil, having a phospholipid content of less than about 50% w/w from a krill biomass and then removing at least a proportion of non-polar lipid components (e.g. triglycerides) from the first krill oil to obtain a second krill oil which is enriched in phospholipids (i.e. has a higher phospholipid content) compared to the first krill oil.
[0093] In contrast to the prior art processes discussed above, some embodiments of the process commence with a non-selective extraction of oil from the krill biomass feed material. Extraction of both polar (e.g. phospholipid) and non-polar (e.g. triglyceride) lipids from solid feed meal may be achieved using a mix of CO.sub.2 and ethanol (e.g. azeotropic ethanol--a water-ethanol mixture comprising about 95% ethanol).
[0094] In some preferred embodiments, a mass ratio of at least about 15% w/w or about 20% w/w ethanol in CO.sub.2 may be used, for example in the range of about 17-22% w/w. In further embodiments a mass ratio of at least about 25% w/w ethanol in CO.sub.2 may be used, such as least about 26% w/w ethanol, or least about 27% w/w ethanol, or least about 28% w/w ethanol, or least about 29% w/w ethanol, or least about 30% w/w ethanol in CO.sub.2 is used.
[0095] In some embodiments, including any one of the other embodiments discussed in this paragraph, extraction temperatures are at or below about 60.degree. C., such as at or below about 55.degree. C., or at or below about 50.degree. C., or at or below about 45.degree. C., or at or below 40 .degree. C., or at or below 35.degree. C., or at or below 30.degree. C. to advantageously reduce or minimise risk of product degradation. The extraction pressure may be set to ensure super-, sub- and/or near critical conditions for the selected temperature and ratio of CO.sub.2 to ethanol. In some embodiments the pressure is in the range of about 200-350 bar, such as about 250-300 bar, although higher pressures e.g. 400 bar and greater, are technically effective and may also be used. In some embodiments, the pressure value or range produces adequate solvent density to ensure extraction of the non-polar lipids does not become rate limiting for the process. In some embodiments, extraction pressure conditions may be adjusted throughout the extraction process to move between super-, sub- or near critical conditions. In some embodiments, subcritical, and/or near critical conditions are used, the conditions for which will vary depending on the CO.sub.2 to ethanol binary mix ratio (or ternary mix ratio in the case of azeotropic ethanol, which also contains water). Extraction times can be determined by those skilled in the art and may depend, inter alia, on the extraction conditions and the desired economic optimisation. In some embodiments, extraction times are typically in the range of about 1-15 hours, such as about 2-10 hours, such as about 2-5 hours or about 3-6 hours, or about 4-5 hours. This in turn can depend on, inter alia, the quantity and particle size of the biomass feed material. Larger particles will permit higher solvent flow rates to be used while still retaining a static biomass and uniform solvent contact. However, a larger particle, also presents an increased diffusion requirement for the solvent to reach the centre of the particles, with more solvent being required. Thus, in some embodiments, the particle size of the feed material is, about 1-5 mm, such as about 2-3mm.
[0096] In some further embodiments, the extraction pressure is about 300 bar and the extraction temperature is about 60.degree. C.
[0097] Separation of the oil can be conducted at lower temperatures (e.g. about 25-35.degree. C.) and pressures (e.g. about 25-60 bar)
[0098] The resulting extracted oil contains both polar (e.g. phospholipid) and non-polar (e.g. triglyceride) lipids, and may have a phospholipid content of less than about 50%, w/w, or less than about 45% w/w, or less than about 40% w/w, or less than about 35% w/w, or less than about 30% w/w, or less than about 25% w/w, or less than about 20% w/w, or less than about 10% w/w.
[0099] In some advantageous embodiments, water and ethanol present in the oil can then be removed using any suitable method, such as evaporation under vacuum (optionally with gentle heating, e.g., about 65.degree. C. or less), nitrogen stream or lyophilisation. In some embodiments, the oil is subjected to evaporation under vacuum, optionally with gentle heating. This may further be followed by short residence time (e.g. 1-3 seconds) at a higher temperature, (e.g. about 70.degree. C., about 75.degree. C. or about 80.degree. C.) to remove water and ethanol co-solvent from the low viscosity polar and non-polar lipid mix, the high proportion of non-polar lipids affording the low viscosity. In some preferred embodiments, temperatures advantageously do not exceed about 60.degree. C. throughout the gentle heating under vacuum, in order to avoid or minimize degradation of the components of the extracted oil. In further embodiments, temperatures throughout the gentle heating under vacuum advantageously do not exceed about 55.degree. C., or about 50.degree. C. or about 45.degree. C., or about 40.degree. C., or about 35.degree. C., or about 30.degree. C., or about 25.degree. C.
[0100] In some embodiments, the residual volatile content (water and ethanol) after evaporation is about or less than about 3% w/w, as this may minimize the possibility of residual ethanol and water adversely impacting separation of the lipids in the later enrichment step. In further embodiments, the residual volatile content is about or less than about 2.5% w/w, or about or less than about 2.0% w/w, or about or less than about 1.5% w/w, or about or less than about 1.0% w/w, or less than about 0.5% w/w, or less than about 0.3% w/w, or less than about 0.1% w/w.
[0101] At this stage, after evaporation, due to the presence of the non-polar lipid components, the oil is still fluid and can be easily analysed, e.g. for phospholipid and/or omega-3 fatty acid content. This is important as an accurate analysis is required in order to calculate desired degree of enrichment, and hence phospholipid content of the final oil achieved in the following step. In particular, where a final high phospholipid content is desired, over-enriching (i.e. further removal of non-polar lipids), even by small margins can lead to processing problems due to excessive viscosity. The evaporated oil can be thoroughly mixed to ensure homogeneity, optionally after transferral to intermediate product tanks. Optionally, the oil may be gently heated (e.g. at a temperature of less than or about 60.degree. C. 55.degree. C., or about 50.degree. C. or about 45.degree. C., or about 40.degree. C., or about 35.degree. C., or about 30.degree. C., or about 25.degree. C.) to assist in maintaining a fluid and homogeneous material for analysis. The less viscous nature of unenriched oil obtained by the first step CO.sub.2/EtOH extraction compared to the first step bulk solid biomass used in the prior art processes discussed above, may, in some embodiments, advantageously allow for more accurate compositional analysis as bulk homogeneity can be more readily achieved. In some embodiments this may advantageously avoid, minimize or otherwise reduce over-enrichment of phospholipids in the subsequent selective extraction of non-polar lipids, which could otherwise result in an undesirably viscous or immovable solid product.
[0102] The second step of the process involves preferred or selective extraction of the non-polar (triglyceride) lipids from the oil obtained in the first step. If the unenriched oil obtained by the first extraction step has been transferred to intermediate tanks, it is returned to the extraction facility. The oil is further subjected to CO.sub.2 extraction, in some preferred embodiments at supercritical conditions, e.g. at or greater than about 300 bar and at about 60.degree. C., in order to selectively extract the non-polar (triglyceride) lipids. In some embodiments, the extraction can commence with a lower pressure and then be increased incrementally to the desired level (e.g. about 300 bar). The non-polar (triglyceride) lipids can be progressively extracted, thereby enriching the remaining raffinate, until the required compositional target is achieved, for example a phospholipid content of at least about 50% w/w, or at least about 55% w/w phospholipids, or at least about 60% w/w, phospholipids or at least about 65% w/w phospholipids. or at least about 70% w/w phospholipids, or at least about 75% w/w phospholipids, or at least about 80% w/w phospholipids, or at least about 85% w/w phospholipids, or at least about 90% w/w phospholipids, or at least about 95% w/w phospholipids, or at least about 97% w/w phospholipids, or at least about 98% w/w phospholipids, or at least about 99%, w/w phospholipids.
[0103] In some embodiments, once the required quantity of non-polar lipid has been extracted to achieve the desired level of phospholipid enrichment, partially depressurising the extraction vessel allows the remaining pressure to assist in discharge of the now enriched (and more viscous) raffinate as required. Draining of enriched high viscosity oil can be tolerated to a greater degree by draining from an extractor still partially pressurised. In this way, extremely viscous materials can be transferred for onwards blending and formulation.
[0104] In one or more embodiments, the process may allow for semi continuous processing, with individual extraction vessels being changed out on a continuous rotation but one at a time while other extraction vessels continue to operate. In this way, stopping to change multiple extractor batches may be avoided.
[0105] In other embodiments, the second step described herein can be used to enrich the phospholipid content of any krill oil having a phospholipid content of less than about 50%, w/w, in order to obtain a krill oil having a phospholipid content of .at least about 50% w/w.
[0106] In some embodiments, the starting krill oil has a phospholipid content of about 45% w/w or less, or about 40% w/w or less, or about 35% w/w or less, or about 30% w/w or less, or about 25% w/w or less, or about 20% w/w or less, or about 10% w/w, or less. In some embodiments, the final enriched oil may have a phospholipid content of at least about 55% w/w phospholipids, or at least about 60% w/w phospholipids or at least about 65% w/w phospholipids. or at least about 70% w/w phospholipids, or at least about 75% w/w phospholipids, or at least about 80% w/w phospholipids, or at least about 85% w/w phospholipids, or at least about 90% w/w phospholipids, or at least about 95% w/w phospholipids, or at least about 97% w/w phospholipids, or at least about 98% w/w phospholipids, or at least about 99%, w/w phospholipids.
[0107] In some embodiments, the enriched krill oil has final a water content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. In some embodiments, the krill oil has a residual extraction solvent content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. In further embodiments, the krill oil has a water content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less, and a residual extraction solvent content of about 5% w/w or less, or about 4, or 3, or 2 or 1, or 0.5% w/w, or less. In still further embodiments, the enriched krill has a residual solvent (water and ethanol) content of 5% w/w or less, or about 4, or 3, or 2 or 1.5, or 1, or 0.5, or 0.3, or 0.1% w/w, or less.
[0108] In still other embodiments, the final enriched krill oil has a phospholipid content of at least about 60% w/w phospholipids, and a residual solvent content of about 3% w/w or less.
[0109] Other embodiments relating to krill oil as described herein, may also, as appropriate, apply to krill oil produced by the process of the disclosure.
[0110] The following examples are provided for the purpose of illustrating some embodiments of the disclosure and are not intended to limit the generality hereinbefore described.
EXAMPLES
Example 1--Preparation of 62% w/w Phospholipid Krill Oil
1. Extraction of Krill Oil From Krill Meal
[0111] Krill meal was extracted with CO.sub.2/ethanol (feed ratio of ethanol to krill meal about 3.0-3.5:1 w/w), with an ethanol mass fraction in the range of about 17-22% w/w, at a temperature of 60.degree. C. and a pressure of 300 bar. The ethanol/CO.sub.2 stage extraction was between 10 and 15 hours in duration. The extracted oil/CO.sub.2/EtOH mixture was separated at a pressure of 45 bar and 25.degree. C.
[0112] Several batches of oil obtained by this method were blended to afford a krill oil containing both polar and non-polar components and having a phospholipid content of about 42% (see Table 1-1 below).
TABLE-US-00002 TABLE 1-1 .sup.31P NMR Analysis of PL in Krill oil sample wt % of total g/100 g Phospholipid (PL) PL sample Phosphatidylcholine PC 76.0 32.3 Alkyl acyl phosphatidylcholine AAPC 9.6 4.1 Phosphatidylinositol PI 0.6 0.3 Phosphatidylserine PS 0.3 0.1 Lysophosphatidylcholine LPC 7.4 3.1 Lyso alkyl acyl phosphatidylcholine LAAPC 0.5 0.2 Phosphatidylethanolamine PE 1.3 0.5 Alkyl acyl phosphatidylethanolamine AAPE 0.9 0.4 Cardiolipin + N-acyl CL/NAPE 2.6 1.1 phosphatidylethanolamine Lysophosphatidylethanolamine LPE 0.9 0.4 Lyso alkyl acyl LAAPE <0.1 <0.1 phosphatidylethanolamine Total PL content 42.5
2. Selective Extraction of Triglycerides From the Krill Oil Obtained From Step 1
[0113] 5.9 kg of feed krill oil (having the composition as set out in Table 1-1 above) was loaded directly into a single 10.7 L extraction vessel (diameter of 155 mm). The feed material was extracted with CO.sub.2 at a temperature of 60.degree. C. and a pressure of 300 bar.
[0114] Extracts, containing largely triglycerides, were recovered and were significantly less viscous than the feed krill oil that was loaded into the extraction vessel.
[0115] The depressurization process of the extraction vessel was commenced once the total mass of extracted material reached 97% of the theoretical amount of material that could be extracted. The facility was depressurized at a constant ramp rate over a period of 15 minutes from 300 to 100 bar with CO.sub.2 circulation continuing, although at 50% of the extraction flowrate. During this time the measured temperature at the exit to the extractor reduced from the 60.degree. C. operating temperature to 50.degree. C. The extractor was then further depressurized from 100 to 75 bar over a period of another 15 minutes without the pump running. Following that, the separation vessel was emptied of its contents.
[0116] While the extraction vessel was still at the processing temperature and 75 bar pressure, the vessel was emptied of its enriched krill oil contents from the base of the extractor vessel well below the raffinate surface level, thereby avoiding loss of high pressure CO2 with the raffinate oil being drained. The recovery of enriched oil took about an hour and in that time the pressure of the vessel decreased from 75 to 54 bar as the remaining CO2 in the extraction vessel expanded to occupy space previously taken up by the raffinate oil.
[0117] Once the discharge of enriched oil had ended, it was observed that some enriched krill oil remained in the extraction vessel on the distributor and bottom surface of the vessel, which was estimated to be less than 2% of the total mass of enriched oil. At commercial scale, any such remaining oil is recovered into the following krill oil batch for that extractor.
[0118] Prior to astaxanthin and phospholipid analysis, the enriched oil was heated in an oven for 55.degree. C. for 1 hour. This allowed the sample to be sufficiently fluid for stirring to achieve a homogenous sample for analysis.
[0119] Table 1-2 summarises the mass, and phospholipid (PL) and astaxanthin (Asta) contents for the feed, extracted and enriched oils. Very little phospholipid (<1 g/100 g extract) and astaxanthin (<2 mg/100 g extract) was co-extracted. Overall, the mass of extract obtained from the enrichment process was 98% of the theoretical extract required for enrichment to 62% phospholipid.
TABLE-US-00003 TABLE 1-2 Summary of Phospholipid (PL) and Astaxanthin (Asta) contents PL Asta Mass Mass % PL mass PL Asta mass Asta (g) (% of feed) (g/100 g) (g) (% of feed) (mg/100 g) (mg) (% of feed) Feed 5870.0 100 42.5 2494.8 100 21 1232.7 100 Extract 1939.0 33 1.0 19.4 0.8 1.7 32.3 2.6 Enriched 3835.6 65.3 62.0 2378.1 95.3 24 902.5 74.7 Total 5774.6 98.4 2397.5 96.1 952.9 77.3 out
Tables 1-3 and 1-4 summarize the compositional content of the enriched oil
TABLE-US-00004 TABLE 1-3 .sup.31P NMR Analysis of PL in Krill oil sample wt % of total g/100 g Phospholipid (PL) PL sample Phosphatidylcholine PC 72.9 45.2 Alkyl acyl phosphatidylcholine AAPC 10.0 6.2 Phosphatidylinositol PI 1.3 0.8 Phosphatidylserine PS 0.7 0.4 Lysophosphatidylcholine LPC 7.6 4.7 Lyso alkyl acyl phosphatidylcholine LAAPC 0.6 0.4 Phosphatidylethanolamine PE 1.1 0.7 Alkyl acyl phosphatidylethanolamine AAPE 1.3 0.8 Cardiolipin + N-acyl CL/NAPE 3.8 2.4 phosphatidylethanolamine Lysophosphatidylethanolamine LPE 0.5 0.3 Lyso alkyl acyl LAAPE 0.2 0.1 phosphatidylethanolamine Total PL content 62.0
TABLE-US-00005 TABLE 1-4 GC Analysis of Fatty acids g/100 g sample Fatty Acid (expressed as FFA) 14:0 4.3 15:0 0.3 16:0 14.9 16:1 n-9 0.3 16:1 n-7 2.4 16:1 n-5 0.4 i17:0 0.2 16:2 0.7 17:1 0.2 i18:0 0.3 16:4 n-1 0.3 18:0 0.8 18:1 n-9 6.2 18:1 n-7 4.4 18:1 n-5 0.2 18:2 n-6 1.3 18:3 n-3 0.9 18:4 n-3 2.2 20:1 n-9 0.5 20:1 n-7 0.2 20:4 n-6 0.3 20:4 n-3 0.3 20:5 n-3 13.6 (EPA) 22:1 n-11 0.6 21:5 n-3 0.4 22:5 n-3 0.4 22:6 n-3 8.2 DHA Others 1.1 Total n-3 26.0 Total fatty acids 66.5
Example 2
[0120] Mussel lipid extract was prepared according to WO97/09992 and used in the form of PCSO-524.RTM. (Pharmalink International Limited, Hong Kong). Krill oil was prepared by the process of Example 1, having the compositional content as set out in Tables 1-3 and 1-4 above.
Sample Preparation
[0121] Fresh samples were prepared daily. The samples were mixed by inversion before sampling. The samples were weighed in 1.5 mL centrifuge tubes and made to 100 mg/mL with ethanol to prepare the stock. The stock mixtures were prepared by weighing out the oils in the correct ratio and then making up to concentration with ethanol. Serial dilutions of the stock solution were made in ethanol. The serial dilutions were then diluted (1 in 100) in cell culture medium before being added to the cells (in triplicate) with a final 1 in 10 dilutions. This resulted in an ethanoic concentration of 0.1% for all doses and controls. Krill oil contained approximately 62% w/w phospholipids. Mussel lipid extract was used in the form of PCSO-524.RTM..
[0122] The abbreviations used in presenting the results are set out in Table 2-1 below:
TABLE-US-00006 TABLE 2-1 Abbreviations Abbreviation Description LY PCSO-524 .RTM. Krill Enriched Krill oil (62% PL) 90% A mixture containing 90% w/w LY and 10% w/w Krill. 75% A mixture containing 75% LY w/w and 25% w/w Krill 50% A mixture containing 50% LY w/w and 50% w/w Krill Olive Food grade olive oil 1400W CAS No 214358-33-5 Dexa Dexamethasone Diclo Diclofenac
Assays
[0123] The anti-inflammatory activity was determined in lipopolysaccharide (LPS) and interferon .gamma. (IFN.gamma.)-stimulated murine macrophages, RAW264.7 cells cultured in standard cell culture media and incubated with LPS and IFN.gamma. in the presence or absence of different test compounds/extracts and positive controls. The production of inflammatory mediators, including NO, PGE.sub.2 and LTB.sub.4, cytokines TNF.alpha. and IL-6, were measured by established methods using commercial kits. Each sample was tested with at least 3 concentrations (using 3 replicates, maximum concentration was 100 .mu.g/ml) (n=9), with relevant internal controls (Table 2-2). The cytotoxicity of each sample was also determined by MTT assay. No cytotoxicity was detected for any concentration tested.
[0124] The assay parameters for each assay are summarised in Table 2-2. Briefly, to perform the anti-inflammatory assay, the cultured RAW264.7 cells were counted and plated (0.8.times.105 cells/well) in 96 well plates and incubated for the indicated plating time. The medium was then aspired and replaced with fresh medium followed by the addition of the test compounds. The compounds were incubated for 1 h prior to the addition of the stimulant. The plates were then incubated for between 4-18 h and the supernatant analysed for the mediator of interest, the remaining cell viability was determined by MTT.
[0125] The positive controls were selected based on their widespread use in similar assays, including N-(3-(Aminomethyl)benzyl)acetamidine (1400W), a slow, tight binding inhibitor of inducible nitric-oxide synthase (iNOS) (Garvey, E. P., et al, J Biol Chem, 1997, 21:272(8):4959-63, and dexamethasone, a commonly used cytokine inhibitor. Diclofenac is a common non-steroidal anti-inflammatory agent and known inhibitor of cyclooxygenase (COX) which produces PGE2.
TABLE-US-00007 TABLE 2-2 Assay Parameters Plat- Incu- ing bation Kit Assay time Stimulation time Supplier Control used NO 48 h LPS (50 ng/ml) 18 h Griess 1400W IF.gamma. (50 units/ml) Reagent (Cayman) TNF.alpha. 48 h LPS (50 ng/ml) 18 h Peprotech Dexamethasone IF.gamma. (50 units/nil) (USA) (Sigma) IL-6 48 h LPS (50 ng/ml) 18 h Peprotech Dexamethasone IF.gamma. (50 units/nil) (USA) (Sigma) PGE.sub.2 24 h LPS (50 ng/ml) 18 h Cayman Diclofenac IF.gamma. (50 units/nil) Chemical (Sigma) (USA)
Results
1. Nitric Oxide Assay
[0126] NO is a radical metabolite, which has been shown to have numerous physiological functions both as a signalling molecule and as a toxic agent in inflammation (Coleman, 2001). NO is derived from the oxidation of L-arginine by three types of nitric oxide synthases (NOS); the constitutive forms, neuronal NOS and endothelial NOS, and the inducible form, iNOS, originally described in murine macrophages (Nathan & Xie, 1994; Stuehr & Marletta, 1985). The inducible form is continually activated once expressed, and is therefore regulated at the transcription level by NF-.kappa.B, stimulated by inflammatory molecules like LPS and IFN-.gamma.. The production of NO by iNOS experiences hours of lag time before NO is produced in much higher (nM) sustained levels (Nathan & Xie, 1994). The inducible form of NOS is most likely implicated in inflammation and due to the higher levels of NO produced it is more easily assessed in-vitro.
[0127] NO is an unusual signalling molecule. As there is no specific cell surface receptor for NO it enters cells indiscriminately, where the effect is dependent on cell type and NO concentration, thus producing a wide range of physiological responses. NO causes increased vascular permeability, vasodilatation and generation of radicals which causes tissue damage and eliminates pathogens (Guzik, Korbut, & Adamek-Guzik, 2003). These physiological changes are associated with inflammation with the increased blood flow, allowing more immune cells to enter affected tissue thereby destroying the pathogen.
[0128] The results for the NO inhibition assay are depicted in FIGS. 1A and 1B and Table 2-3
TABLE-US-00008 TABLE 2-3 IC.sub.50 for NO Inhibition LY Krill 90% LY 75% LY Olive Oil 1400 W IC.sub.50 ~107 ~286 44.8 40.9 Not Active 1.6
2. Tumour Necrosis Factor-Alpha
[0129] TNF.alpha. is a cell signalling protein (cytokine) involved mainly in the acute phase inflammatory response. Macrophages are the major source of TNF.alpha., although it can be released by many other cell types such as CD4+ lymphocytes, natural killer (NK) cells, neutrophils, mast cells, eosinophils, and neurons. TNF.alpha. is produced by activation of MAPK and NF-.kappa.B. It acts to increase its own production and that of other inflammatory cytokines such as interleukin-1 beta (IL-1.beta.). TNF.alpha. induces fever, apoptotic cell death, cachexia, inflammation and inhibits tumorigenesis and viral replication. TNF.alpha. is implicated in many disease states, including, sepsis, traumatic injury, ischemia, asthma, burns, irritable bowel syndrome, Alzheimer's disease, cancer, major depression, arthritis and multiple sclerosis (Cairns, Panacek, Harken, & Banerjee, 2000; Dowlati et al., 2010; Swardfager et al., 2010).
[0130] The results for the TNF.alpha. inhibition assay are depicted in FIGS. 2A and 2B and Table 2-4.
TABLE-US-00009 TABLE 2-4 IC.sub.50 for TNF.alpha. Inhibition LY Krill 90% LY 75% LY Olive Oil Dexa IC.sub.50 ~238.7 ~103.3 89 57 DNF* 3.328e-005 *Did not fit mathematical model of a dose response curve
3. Interleukin-6
[0131] Like TNF.alpha., IL-6 is considered a pro-inflammatory cytokine. IL-6 is secreted by T cells and macrophages which stimulates an immune response. IL-6 is responsible for increased production of neutrophils in bone marrow. It supports the growth of B cells and is antagonistic to differentiation of T cells into regulatory T cells. It is capable of crossing the blood-brain barrier and initiating synthesis of PGE.sub.2 in the hypothalamus, thereby changing the body's temperature set point (Banks, Kastin, & Gutierrez, 1994).
[0132] The results for IL-6 inhibition assay are depicted in FIGS. 3A and 3B and Table 2-5
TABLE-US-00010 TABLE 2-5 IC.sub.50 for IL-6 Inhibition LY Krill 90% LY 75% LY Olive Oil Dexa IC.sub.50 22.4 13.4 11.5 10.4 ~106.8 1.19
4. Prostaglandin E.sub.2
[0133] Prostaglandin E2 (PGE) is one of the lipid mediators produced from arachidonic acid (AA) by the action of the enzyme cyclooxygenase (COX) and is involved in inducing pyrexia, pain sensation, and inflammation. Aspirin and non-steroidal anti-inflammatory drugs (NSAIDS) inhibit the biosynthesis of prostanoids (including PGE.sub.2), resulting in antipyretic, analgesic, and anti-inflammatory effects (Kawahara, K., et al, 2015, and Kawabata, A., 2011).
[0134] The results are depicted in FIGS. 4A and 4B and Table 2-6.
TABLE-US-00011 TABLE 2-6 IC.sub.50 for PG-E.sub.2 Inhibition LY Krill 90% LY 75% Ly 50% LY Diclofenac Olive Oil Not Not ~112 ~118 55.3 0.12 DNF* Active Active *Did not fit mathematical model of a dose response curve
Summary of Results
[0135] Mussel lipid extract and krill oil were demonstrated in this assay system at the concentrations tested to individually inhibit NO, TNF.alpha. and IL-6, but not PGE.sub.2.
[0136] The combination of mussel lipid extract and krill oil was more effective than either mussel lipid extract or krill oil alone in inhibiting NO, TNF.alpha. and IL-6. In the PGE.sub.2, assay, neither mussel lipid extract or krill oil alone demonstrated inhibitory activity, but in combination, demonstrated inhibition.
Example 3--Synergy
[0137] Mussel lipid extract was prepared according to WO97/09992 and used in the form of PCSO-524.RTM.. Krill oil was prepared by the process of Example 1, having the compositional content as set out in Tables 1-3 and 1-4 above.
Samples, Preparation and Combination
[0138] Stock samples of PCSO-524.RTM. and high-phospholipid krill oil were mixed by inversion before experimental sampling. The samples were weighed in 15 mL centrifuge tubes and made to 100 mg/mL with ethanol to prepare the stock. The mixtures were prepared by mixing the diluted oils in the correct ratio. Serial dilutions were made in ethanol. The serial dilutions were then diluted (1 in 100) in cell culture medium before being added to the cells (in triplicate) with a final 1 in 10 dilutions. This resulted in an ethanoic concentration of 0.1% for all doses and controls. These doses were prepared fresh daily. Table 3-1 shows the abbreviations used for each sample.
TABLE-US-00012 TABLE 3-1 Sample name abbreviations Abbreviation Description LY PCSO-524 .RTM. Krill Enriched Krill oil (62% PL) LY90 A mixture containing 90% w/w LY and 10% w/w Krill. LY80 A mixture containing 80% LY w/w and 20% w/w Krill LY70 A mixture containing 70% LY w/w and 30% w/w Krill LY65 A mixture containing 65% LY w/w and 35% w/w Krill LY60 A mixture containing 60% LY w/w and 40% w/w Krill LY55 A mixture containing 55% LY w/w and 45% w/w Krill LY50 A mixture containing 50% LY w/w and 50% w/w Krill LY45 A mixture containing 45% LY w/w and 55% w/w Krill LY40 A mixture containing 40% LY w/w and 60% w/w Krill LY35 A mixture containing 35% LY w/w and 65% w/w Krill LY30 A mixture containing 30% LY w/w and 70% w/w Krill LY20 A mixture containing 20% LY w/w and 80% w/w Krill LY10 A mixture containing 10% LY w/w and 90% w/w Krill Olive Food grade olive oil 1400W CAS No 214358-33-5 Dexa Dexamethasone CI Confidence Interval
Assays
[0139] The anti-inflammatory activity was determined in lipopolysaccharide (LPS) and interferon .gamma. (IFN.gamma.)-stimulated murine macrophages, RAW264.7 cells cultured in standard cell culture media (DMEM, Foetal bovine serum 5%) and incubated in the presence or absence of different test compounds/extracts and controls. The production of inflammatory mediators, including NO, cytokines TNF.alpha. and IL-6, were measured by established methods using commercial ELISA kits (suppliers listed in
[0140] Table 3-2). Each sample was tested with at least 6 concentrations (using 3 replicates, maximum concentration was 100 .mu.g/mL) (n=9), with relevant internal controls (shown in
[0141] Table 3-2). The cytotoxicity of each sample tested was determined by MTT assay. There was no cytotoxicity detected for any concentration tested.
[0142] The assay parameters for each assay are summarised in
[0143] Table 3-2. Briefly, for the NO, TNF.alpha. and IL-6 assays the cultured RAW264.7 cells were counted and plated (0.8.times.10.sup.5 cells/well) in 96 well plates and incubated for 48 h. The medium was then aspired and replaced with fresh medium followed by the addition of the test compounds. The compounds were incubated for 1 h prior to the addition of the stimulant. The plates were then incubated for between 18 h and the supernatant analysed for the mediator of interest, the remaining cell viability was determined by MTT.
[0144] The positive controls were selected based on their widespread use in similar assays, including N-(3-(Aminomethyl)benzyl)acetamidine (1400W), a slow, tight binding inhibitor of inducible nitric-oxide synthase (iNOS) (Garvey et al., 1997), and dexamethasone, a commonly used cytokine inhibitor.
TABLE-US-00013 TABLE 3-2 Anti-inflammatory assays and positive controls Plating Incubation Kit Positive Assay time Stimulation time supplier Control NO 48 h LPS 18 h Griess 1400W (50 ng/ml), Reagent (Cayman) IFN.gamma. (50 units/ml) TNF.alpha. 48 h LPS 18 h Peprotech Dexamethasone (50 ng/ml), (USA) (Sigma) IFN.gamma. (50 units/ml) IL-6 48 h LPS 18 h Biogems Dexamethasone (50 ng/ml), (USA) (sigma) IFN.gamma. (50 units/ml)
Synergy Calculations
[0145] Synergism is expressed as a Combination Index. The synergism combination index (CI) and isobologram IC50 weightings were calculated using Compsyn software. The dose response curve generated in Graphpad Prism was transformed into 10 points representing the curve. These 10 points were then entered into the Compsyn program that generated a curve to fit the data points. This method was preferable to closely replicate the complete dose response curve in the synergy program. The Compsyn fit curve was then used for synergy calculations. The pattern in the synergy over the range tested can be observed with an isobologram, which plots the relative contribution of each component to the activity at the IC50. There is a straight line drawn between the two drugs being blended (Biavatti, 2009). Values below the line indicate synergy, on the line is considered additive and above the line is antagonistic.
Results
1. Nitric Oxide Assay
[0146] The combinations were assayed to determine if they demonstrated synergistic inhibition on the inflammatory signalling molecule NO. The combinations were first tested in 10% increments. The most active combination observed was LY60. Further 5% increments were then tested around LY60.
[0147] The dose response curves of the tested combinations LY90-LY10 are depicted in FIGS. 5A, 5B, 6A and 6B. IC50 values for NO inhibition and Combination Index are presented in Tables 3-3 and 3-4. A combination index less than 1 indicates synergy. The isobologram for the 10% increments is depicted in FIG. 7.
TABLE-US-00014 TABLE 3-3 IC.sub.50 values for NO inhibition of combinations LY90-LY10 LY K LY90 LY80 LY70 LY60 LY50 LY40 LY30 LY20 LY10 1400 W IC.sub.50 204 265 105 109 74 67 83 85 111 125 134 25 (.mu.g/mL) 95% CI 136 to 144 to 87 to 88 to 63 to 59 to 64 to 68 to 81 to 98 to 98 to 21 to (.mu.g/mL) 392 1108 136 144 90 77 117 115 183 194 248 32
TABLE-US-00015 TABLE 3-4 IC50s (CI) and Combination Index for NO inhibition of combinations LY75 to LY45 LY75 LY70 LY65 LY60 LY55 LY50 LY45 IC.sub.50 123 68 84 65 86 79 84 (.mu.g/mL) 95% CI 97 to 65 to 69 to 60 to 70 to 69 to 73 to for IC.sub.50 160 85 96 73 90 89 100 (.mu.g/mL) Combi- 0.80 0.50 0.55 0.48 0.54 0.77 0.61 nation Index
2. TNF.alpha. Assay
[0148] The combinations were assayed to determine if they demonstrated synergistic inhibition on the inflammatory cytokine TNF.alpha.. The combinations were first tested in 10% increments. The most active combination observed was LY50. Further 5% increments were then tested around LY50.
[0149] The dose response curves of the tested combinations LY90-LY10 are depicted in FIGS. 8A, 8B, 8C, 9A and 9B. IC50 values for TNF.alpha. inhibition and combination index are presented in Tables 3-5 and 3-6. A combination index less than 1 indicates synergy. The isobologram for the 10% increments is depicted in FIG. 10.
TABLE-US-00016 TABLE 3-5 IC50 values for TNF inhibition LY K LY90 LY80 LY70 LY60 LY50 LY40 LY30 LY20 LY10 IC50 ~606 ~966 -- 56 64 53 43 48 64 61 77 95% CI -- -- -- 49 to -- to 75 44 to 35 to 37 to -- to 71 39 to 56 to 67 64 50 58 90 134 *-- no estimate possible
TABLE-US-00017 TABLE 3-6 IC50's and CI for LY70-LY35 (n = 9) LY70 LY65 LY60 LY55 LY50 LY45 LY40 LY35 IC50 91 80 78 68 69 90 70 73 95% CI 64 to 189 60 to 137 64 to 98 54 to 87 60 to 80 68 to 128 57 to 89 59 to 92 Combination 0.14 0.12 0.114 0.097 0.096 0.123 0.094 0.095 Index
3. IL-6 Assay
[0150] The combinations were assayed to determine if they demonstrated synergistic inhibition on the inflammatory cytokine IL-6. The combinations were first tested in 10% increments. The most active combination observed was LY60. Further 5% increments were then tested around LY60.
[0151] The dose response curves of the tested combinations LY90-LY10 are depicted in FIGS. 11A, 11B, 12A and 12B. IC50 values for IL-6 inhibition and Combination Index are presented in Tables 3-7 and 3-8. A combination index less than 1 indicates synergy. The isobologram for the 10% increments is depicted in FIG. 13.
TABLE-US-00018 TABLE 3-7 IC50s for IL-6 inhibition (n = 3) LY K LY90 LY80 LY70 LY60 LY50 LY40 LY30 LY20 LY10 Dex IC50 173 114 ~100 53 36 34 37 52 41 60 ~60 0.010 (.mu.g/mL) 95% CI 104 to 92 to 41 to 29 to 27 to 30 to 44 to 32 to 50 to 0.004 to (.mu.g/mL) 1311 191 61 45 42 44 60 51 72 0.03
TABLE-US-00019 TABLE 3-8 IC50s and Combination Index for IL-6 inhibition LY70 LY65 LY60 LY55 LY50 LY45 LY40 LY35 LY30 IC50 36 39 34 43 37 49 52 41 41 95% 29 to 33 to 27 to 37 to 30 to 40 to 44 to 30 to 32 to CI 45 45 42 -- 44 58 60 54 51 Combi- 0.27 0.23 0.31 0.27 0.37 0.4 0.32 0.32 nation Index
Summary of Results
[0152] Mussel lipid extract and krill oil, used in combination, were demonstrated in this assay system to meet the mathematical criteria for synergy in inhibiting NO, TNF.alpha. and IL-6.
Example 4--Patient Study
[0153] Patients suffering from a variety of pain/inflammatory conditions were administered a combination of mussel lipid extract (in the form of PCSO-542) and krill oil (61% PL) in a ratio of PCSO-542 to krill oil of 75:25, in capsule form. The composition of said capsules in presented in Table 4-1.
TABLE-US-00020 TABLE 4-1 Composition of 150 mg oil blend capsules Ingredient Weight % Fill Component Olive oil 66.67% PCSO-542* Mussel 25% (37.5 mg) oil (Perna canaliculus) Krill oil* 8.33% (12.5 mg) (Euphausia superba) Shell Component (before drying) Gelatin (bovine) 46.5 Glycerine 12.6 Sorbtiol 6.00 water 34.9 *contains 0.15% w/w vitamin E (i.e. approx. 0.056 mg/capsule)
[0154] Dosage typically ranged from 2-8 capsules per day, over one, two or three dosages. Prior to commencing treatment with the combination, patients had typically been taking one or more NSAIDS, including paracetamol or ibuprofen, to manage pain. The results are depicted in Table 4-2.
TABLE-US-00021 TABLE 4-2 Summary of patient results Pain level Pain level prior to after Patient treatment treatment (gender/age) Condition (1-10) (1-10) Other comments F, 80-84 Severe pain in knee 8 4 Conditions present and shoulders for 3-12 years. Improvement after 2-3 weeks of treatment (2 caps per day). M, 60-64 Lumbar spine stenosis 6-8 0 Condition present for 2-3 years. Improvement after 4-5 days of treatment (6-8 caps/day). M, 40-44 Lumbar pain 7 4 Condition present for 15 years. Improvement after 7 days of treatment (3 caps/day). F, 45-49 Chronic back pain 8 4-5 Condition present for 2 years. Improvement after 2-3 weeks of treatment (3 caps/day). M, 50-54 Joint pain in hands- 6 2 Condition present nerve damage/swollen for 2 years. knuckles Improvement after 1 week of treatment (3 caps/day). M, 50-54 Chronic back pain - 2-8, 2-3 Condition present herniated disc depending for 25 years. on activity Improvement after 3 days of treatment (4 caps/day). F, 65-69 Chronic knee and 10 5 Condition present shoulder pain for 5 years. Improvement after 2 weeks of treatment (4 caps/day). M, 20-24 Severe allergy - 8 5 Condition present coughing, inflamed for 4 years. nasal passage and Improvement after 3 nasal drip weeks of treatment (4 caps/day). M, 40-44 Back pain - herniated 9 5 Condition present disc for 8 years. Improvement after 3 weeks of treatment F, 50-54 Back pain 7 01-1 Condition present for 1 month. Improvement after 1 week of treatment F, 55-59 Osteoporosis - pain 7-8 1-3 Condition present varies with activity for 15 years. Improvement after 11/2 weeks of treatment (4 caps/day). M, 65-69 Knee pain 8-10 2-3 Condition present for 14 years. Improvement after 11/2 weeks of treatment (4 caps/day). M, 50-54 Neck pain, stiff knees 5 3 Condition present for several years. Improvement after 3 weeks of treatment (3 caps/day). M, 50-54 Body aches and pain 7-8 2-3 Improvement after a in knees and elbow few days of post-surgery treatment F, 65-69 Arthritis/tendonitis in 6 2 Condition present finger joints for several months. Improvement after few days of treatment (6 caps/day). F Rheumatoid arthritis 7 Joint ache Condition present and pain for 10+ years. reduced Improvement after tremendously few days of treatment (4 caps/day) F, 50-54 Lupus/fibromyalgia 8 4 Condition present for 10+ years. Improvement after 7-8 days of treatment M, 30-34 Shoulder pain, 7 4 Improvement after 2 increasing after weeks of treatment exercise (4 caps/day) F Pre-existing knee and 5 3 Improvement after ankle injury 2-3 weeks of treatment (4 caps/day) M, 45-49 Herniated discs, 10 General Condition present shoulder and hand improvement for several months. injuries Improvement after 1 week of treatment (2 caps/day). F, 40-44 Knee discomfort 4 0 Condition present (crunching sensation) for several years. Improvement after 2 weeks of treatment (2 caps/day) F Joint stiffness and Varies 5 Condition present back pain daily Improved for several years. hand Improvement after 1 movement weeks of treatment (2 caps/day) F, 55-59 Back pain 6 4 Condition present for a few years. Improvement after 2 weeks of treatment (3 caps/day) M (elderly) Joint pain 6 2 Condition present for several years. Improvement after 4 weeks of treatment (2 caps/day) M Shoulder and knee 8 4 Condition present pain for several years. Improvement after 1 week of treatment (2 caps/day) M Knee pain 8 0 Improvement after 1 week of treatment (4 caps/day). F Arthritis 8 5 Improvement after treatment (4 caps/day) M Arthritis 8 5 Condition present for several years. Improvement after treatment (patient noted increased improvement compared to use of Omega XL) (4 caps/day) F Arthritis 6 3 Improvement after treatment (patient noted increased improvement compared to use of Omega XL) (4 caps/day)
REFERENCES
[0155] Biavatti, M. W. (2009). Synergy: an old wisdom, a new paradigm for pharmacotherapy. Brazilian Journal of Pharmaceutical Sciences, 45(3), 371-378. Cairns, C. B., Panacek, E. A., Harken, A. H., & Banerjee, A. (2000). Bench to bedside: Tumor necrosis factor-alpha: From inflammation to resuscitation. Academic Emergency Medicine, 7(8), 930-941. Coleman, J. W. (2001). Nitric oxide in immunity and inflammation. Int Immunopharmacol, 1(8), 1397-1406. Dowlati, Y., Herrmann, N., Swardfager, W., Liu, H., Sham, L., Reim, E. K., & Lanctot, K. L. (2010). A Meta-Analysis of Cytokines in Major Depression. Biological Psychiatry, 67(5), 446-457. doi:10.1016/j.biopsych.2009.09.033 Garvey, E. P., Oplinger, J. A., Furfine, E. S., Kiff, R. J., Laszlo, F., Whittle, B. J. R., & Knowles, R. G. (1997). 1400W is a slow, tight binding, and highly selective inhibitor of inducible nitric-oxide synthase in vitro and in vivo. Journal of Biological Chemistry, 272(8), 4959-4963. Guzik, T. J., Korbut, R., & Adamek-Guzik, T. (2003). Nitric oxide and superoxide in inflammation and immune regulation. J Physiol Pharmacol, 54(4), 469-487. Kawabata, A., (2011). Prostaglandin E.sub.2 and Pain--An Update. Biol Pharm Bull, 34(8), 1170-1173 Kawahara, K. (2015). Prostaglandin E.sub.2-induced inflammation: Relevance of prostaglandin E receptors. Biochim Biophys Acta, 1851 (4), 414-421 Nathan, C., & Xie, Q. W. (1994). Nitric oxide synthases: roles, tolls, and controls. Cell, 78(6), 915-918. Sinclair, A. J., Murphy, K. J. and Li, D. (2000) Marine lipids overview "news insights and lipid composition of Lyprinol". Allerg Immunol (Paris) 32(7), 261-271 Stuehr, D. J., & Marletta, M. A. (1985). Mammalian nitrate biosynthesis: mouse macrophages produce nitrite and nitrate in response to Escherichia coli lipopolysaccharide. Proc Natl Acad Sci USA, 82(22), 7738-7742. Swardfager, W., Lanctot, K., Rothenburg, L., Wong, A., Cappell, J., & Herrmann, N. (2010). A Meta-Analysis of Cytokines in Alzheimer's Disease. Biological Psychiatry, 68(10), 930-941.
* * * * *
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015
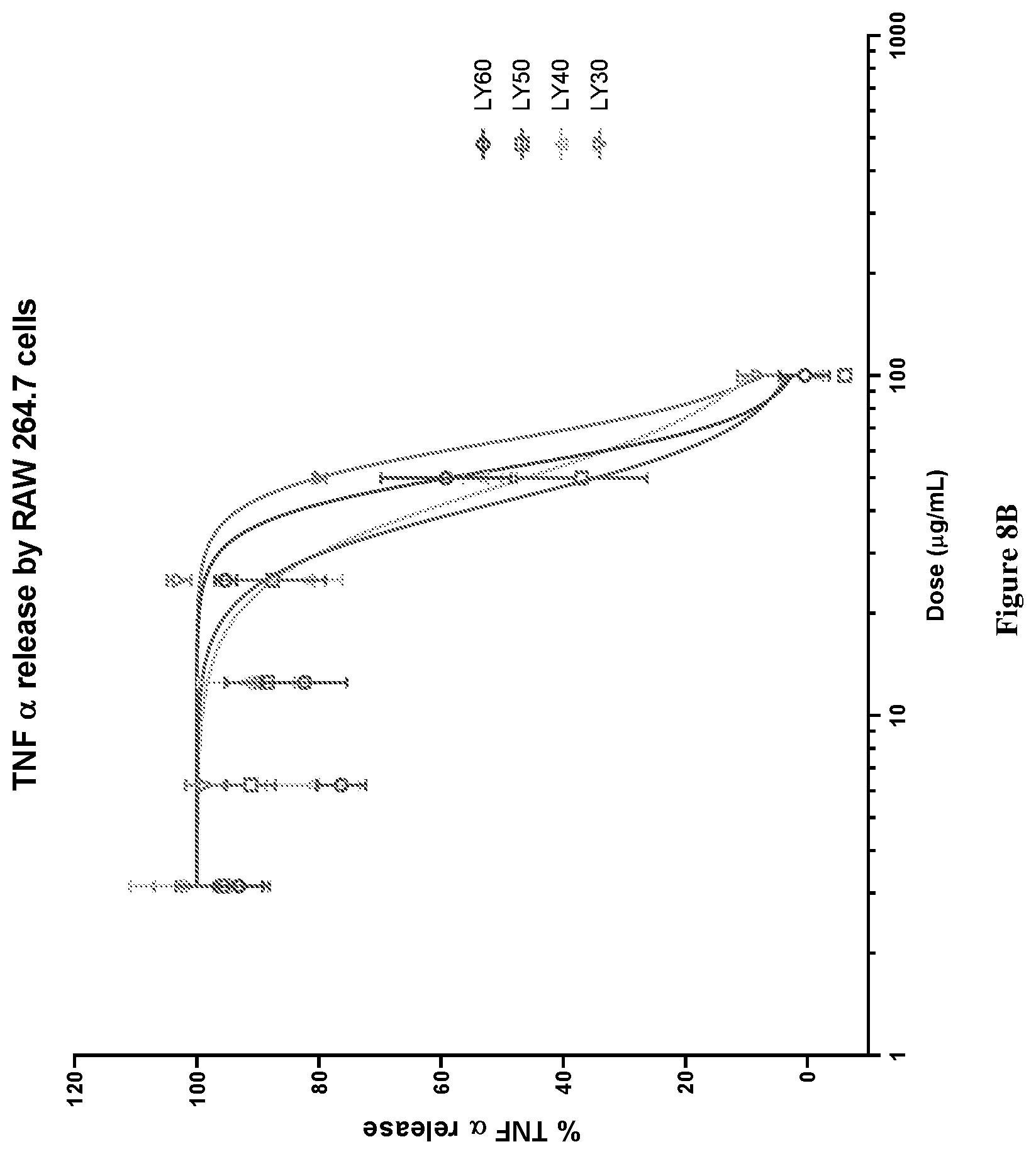
D00016
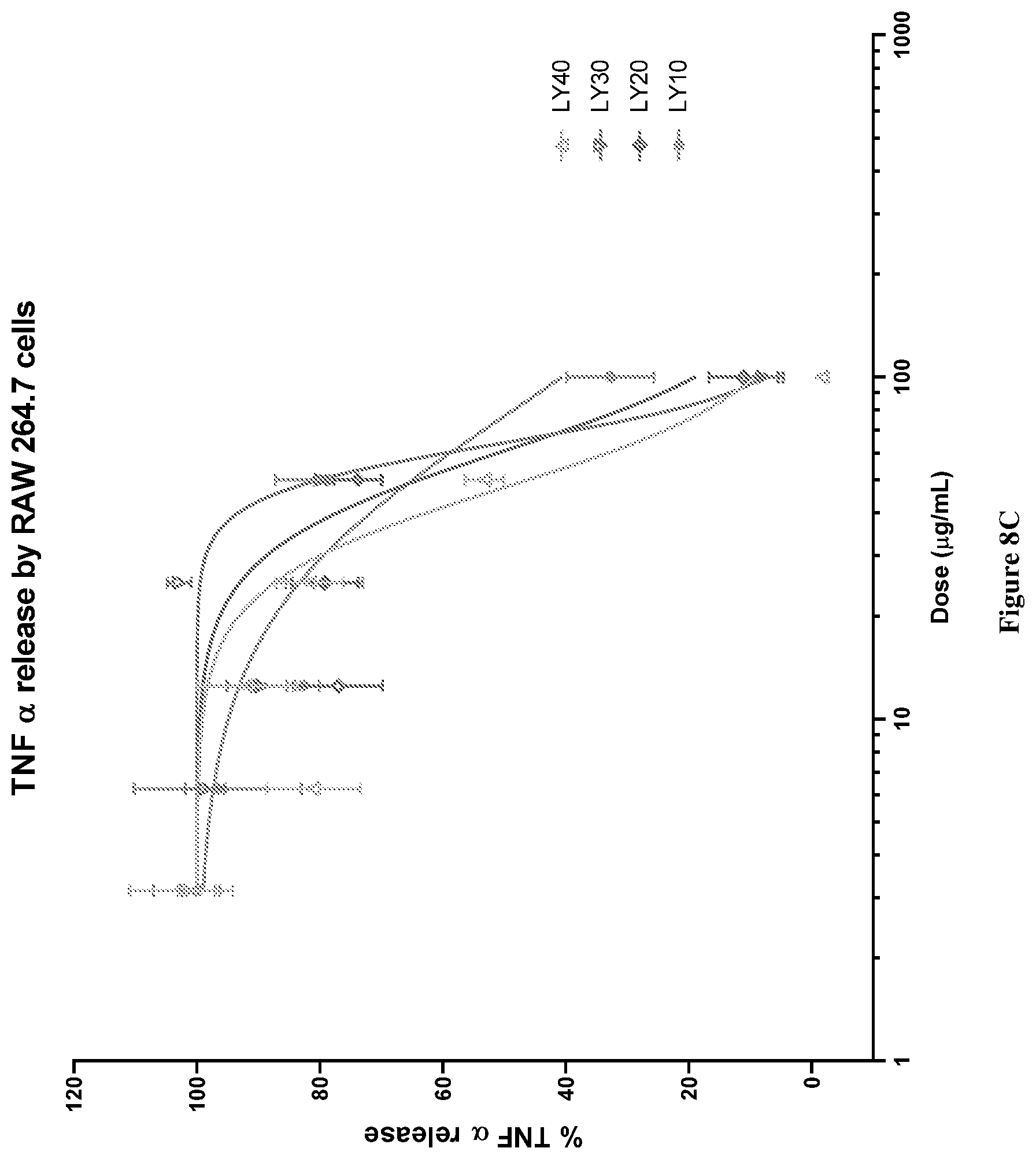
D00017
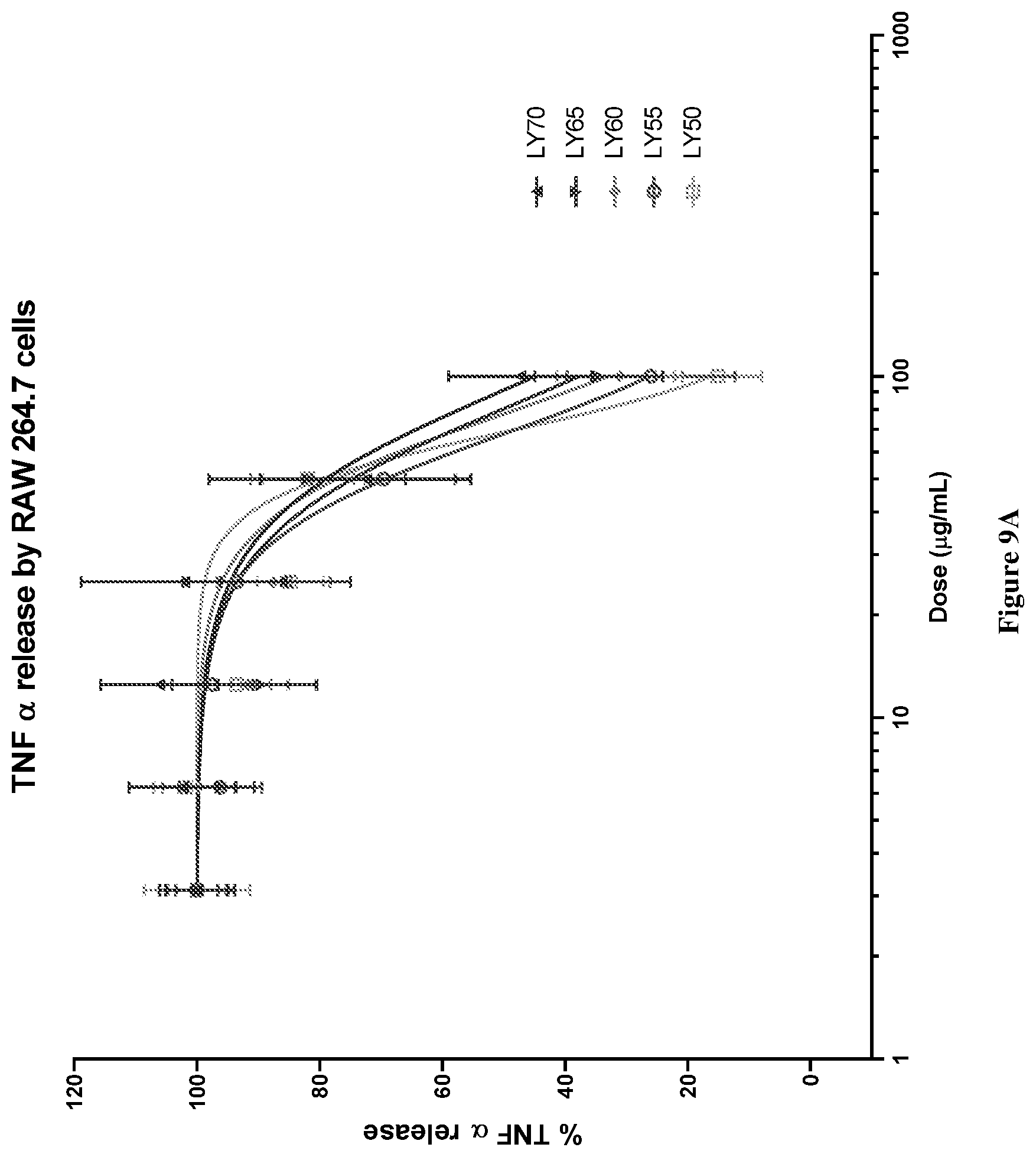
D00018

D00019
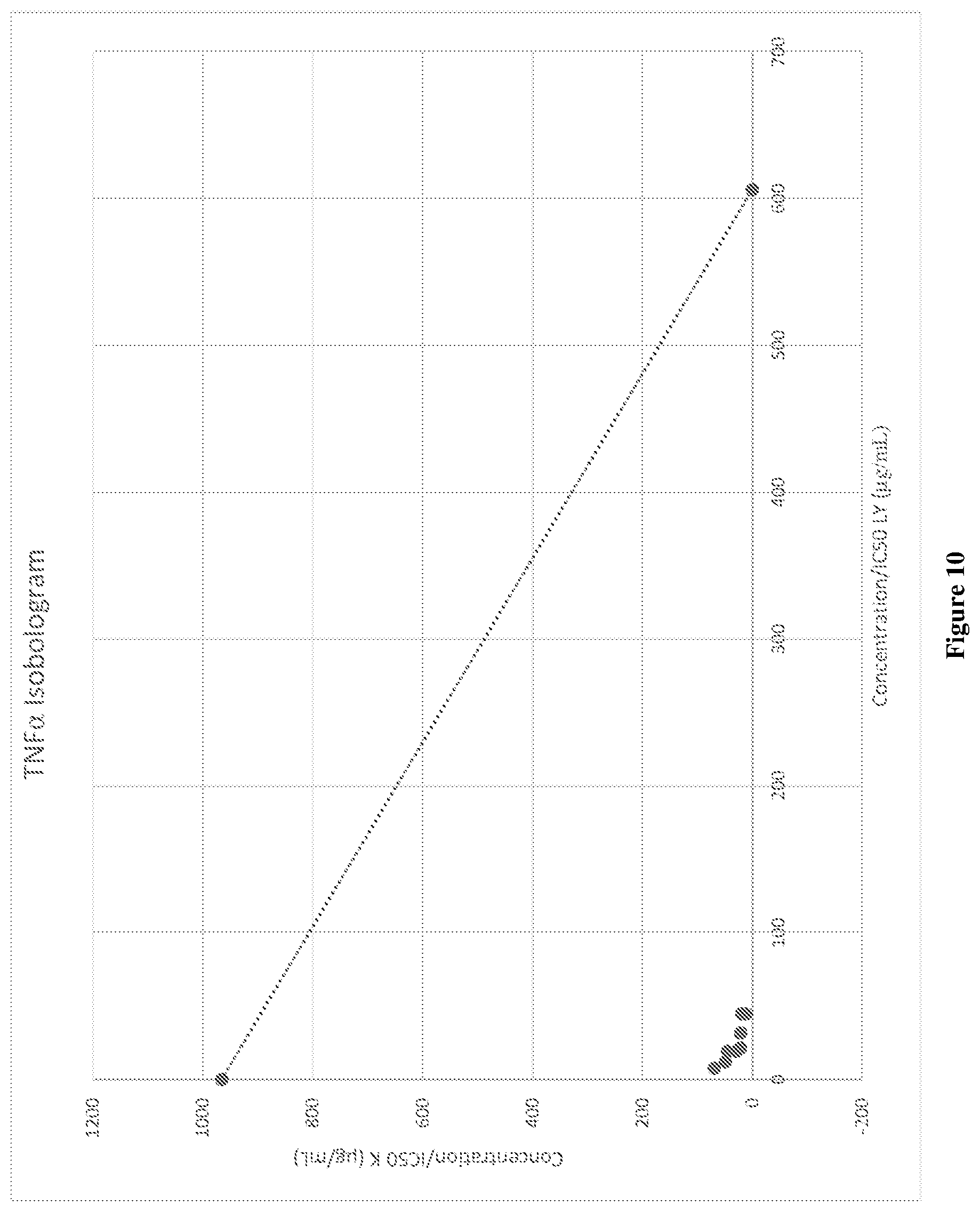
D00020

D00021
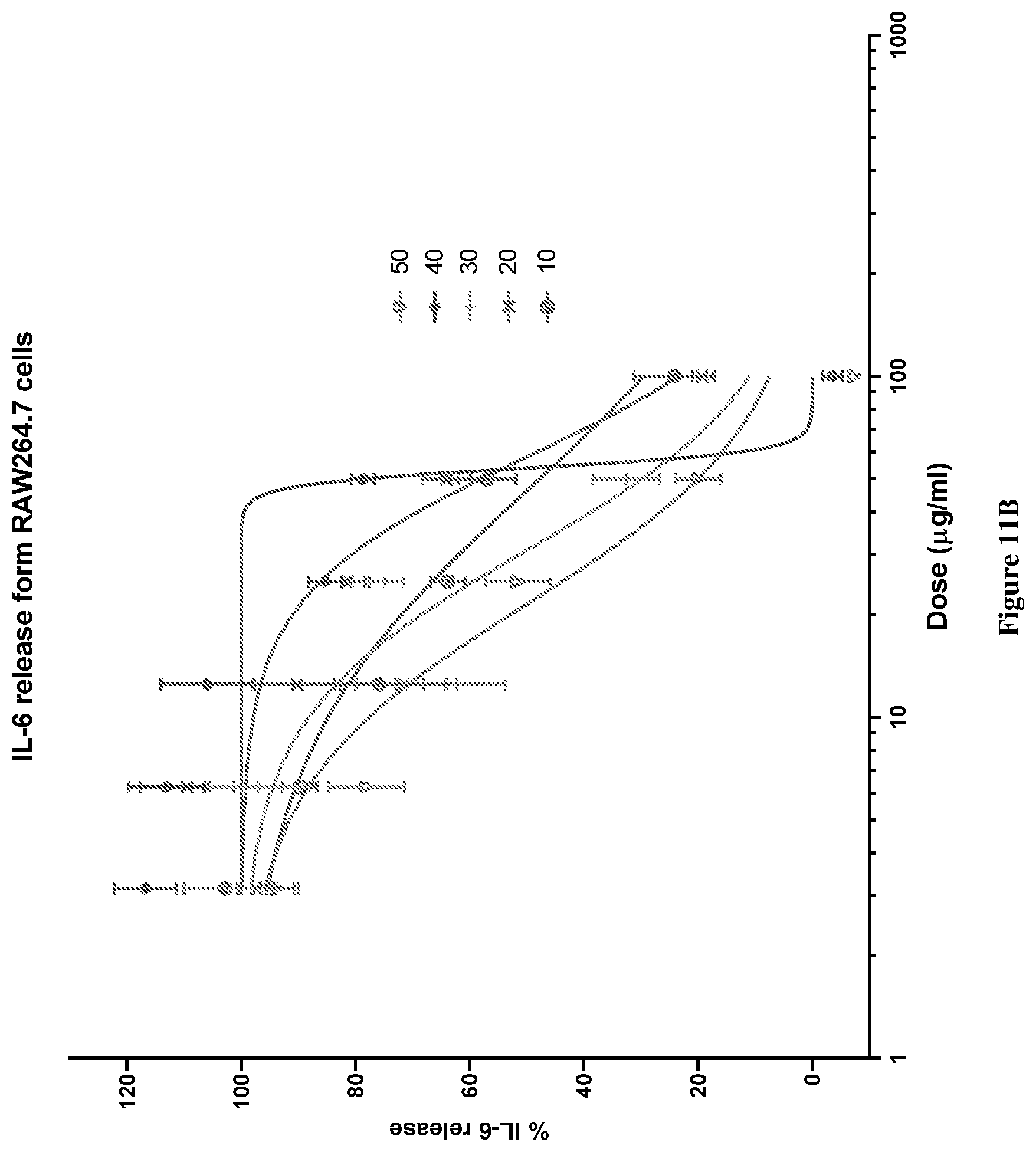
D00022
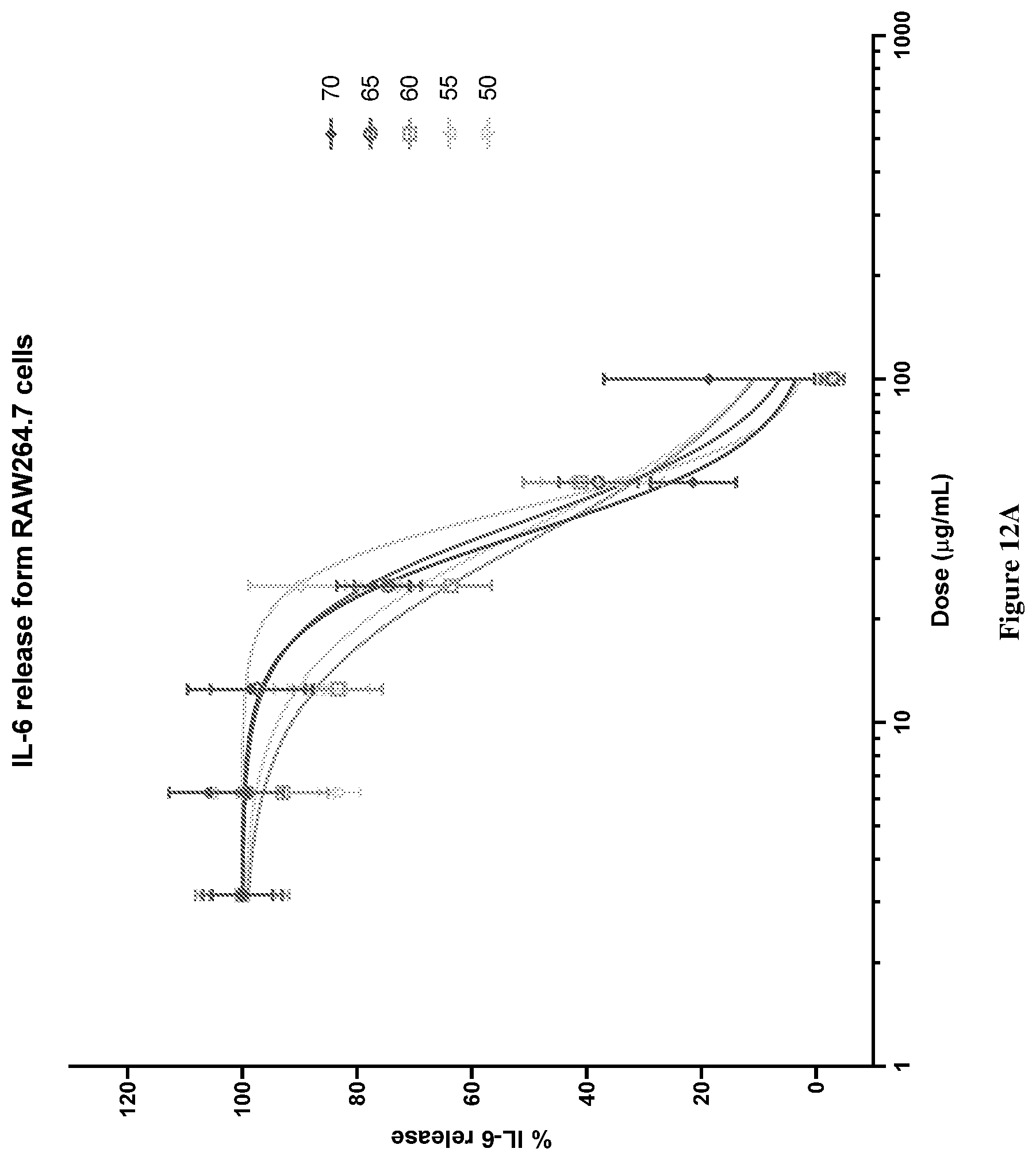
D00023

D00024
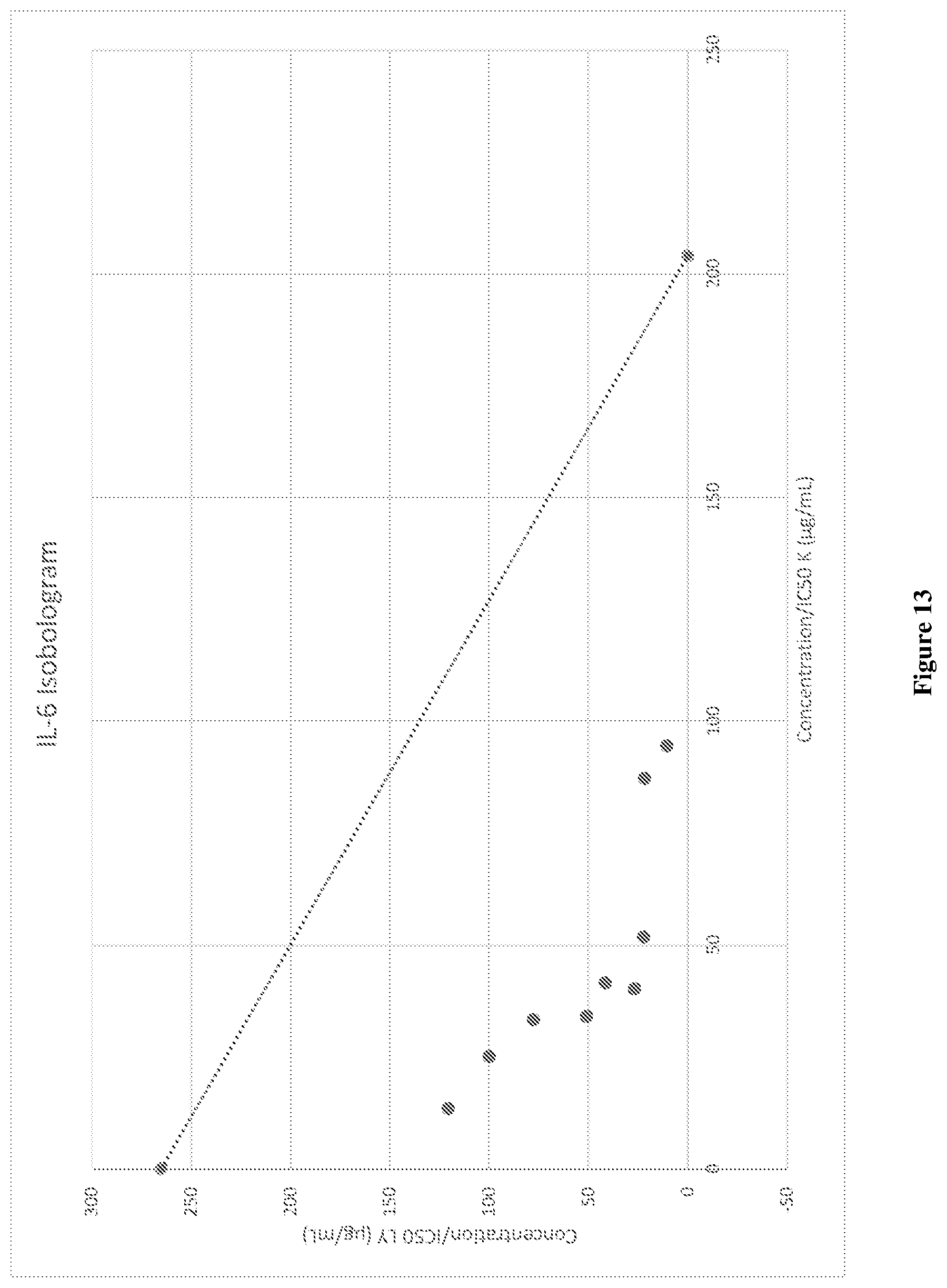
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.