Anti-tnf Alpha Antibody Formulations
CALLAHAN; William J. ; et al.
U.S. patent application number 16/471492 was filed with the patent office on 2020-03-19 for anti-tnf alpha antibody formulations. This patent application is currently assigned to AMGEN INC.. The applicant listed for this patent is AMGEN INC.. Invention is credited to Joy BRENNAN, William J. CALLAHAN, Rahul Rajan KAUSHIK.
| Application Number | 20200087390 16/471492 |
| Document ID | / |
| Family ID | 61006357 |
| Filed Date | 2020-03-19 |












View All Diagrams
| United States Patent Application | 20200087390 |
| Kind Code | A1 |
| CALLAHAN; William J. ; et al. | March 19, 2020 |
ANTI-TNF ALPHA ANTIBODY FORMULATIONS
Abstract
Stable adalimumab formulations are disclosed.
| Inventors: | CALLAHAN; William J.; (Thousand Oaks, CA) ; KAUSHIK; Rahul Rajan; (Newbury Park, CA) ; BRENNAN; Joy; (Thousand Oaks, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | AMGEN INC. Thousand Oaks CA |
||||||||||
| Family ID: | 61006357 | ||||||||||
| Appl. No.: | 16/471492 | ||||||||||
| Filed: | December 20, 2017 | ||||||||||
| PCT Filed: | December 20, 2017 | ||||||||||
| PCT NO: | PCT/US2017/067723 | ||||||||||
| 371 Date: | June 19, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62437640 | Dec 21, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 9/08 20130101; A61K 47/10 20130101; A61K 47/183 20130101; A61K 39/39591 20130101; C07K 16/241 20130101; A61K 9/0019 20130101 |
| International Class: | C07K 16/24 20060101 C07K016/24; A61K 47/10 20060101 A61K047/10; A61K 47/18 20060101 A61K047/18 |
Claims
1. A stable aqueous formulation comprising about 180 mg/mL adalimumab, about 20 mM glutamate, and about 160 mM monoethanolamine (MEA), wherein the formulation has a pH of about 5.2, and demonstrates less than about a 2.1-fold increase in acidic species as measured by cation-exchange high-performance liquid chromatography (CEX-HPLC) after storage for 28 days at 40.degree. C.
Description
REFERENCE TO PRIOR APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application No. 62/437,640, filed Dec. 21, 2016, which is hereby incorporated by reference.
[0002] The present application is being filed along with a sequence listing in electronic format. The sequence listing is provided as a file entitled A-2102-WO-PCT_SeqListing_122017.txt, created Dec. 19, 2017, which is 8 kb in size. The information in the electronic format of the sequence listing is incorporated herein by reference in its entirety.
BACKGROUND
[0003] Elevated levels of tumor necrosis factor alpha (TNF.alpha.) have been associated with a number of human disorders such as arthritis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, axial spondyloarthritis, juvenile idiopathic arthritis, enthesitis related arthritis, osteoarthritis, peripheral spondyloarthritis, acute disc prolapse, inflammatory bowel disease, Crohn's disease, ulcerative colitis, intestinal Behcet's disease, chronic pouchitis, small bowel lesions, Hermansky-Pudlak syndrome, psoriasis, psoriasis vulgaris, psoriasis arthropica, plaque psoriasis, hidradenitis suppurativa, interstitial cystitis, sleep apnea, sarcoidosis, retinal vascular disorders, uveitis, choroidal neovascularization, Pyoderma Gangrenosum, giant cell arteritis, Netherton syndrome, anaplastic thyroid cancers, asthma, and refractory asthma. TNF.alpha. inhibitors are frequently used to treat these disorders. One such inhibitor is adalimumab, also known as D2E7, a recombinant human IgG1 monoclonal antibody specific for human TNF.alpha..
SUMMARY
[0004] The present disclosure is directed to stable aqueous adalimumab formulations, to methods of making stable aqueous adalimumab formulations, to use of a formulation as disclosed herein, and to methods of treating a disease comprising administering to a patient a formulation as disclosed herein.
[0005] In one aspect, the disclosure includes a stable aqueous formulation comprising about 180 mg/mL adalimumab, about 20 mM glutamate, and about 160 mM monoethanolamine (MEA), wherein the formulation has a pH of about 5.2, and demonstrates less than about a 2.1-fold increase in acidic species as measured by cation-exchange high-performance liquid chromatography (CEX-HPLC) after storage for 28 days at 40.degree. C.
[0006] In another aspect, the disclosure provides a stable adalimumab formulation as described in the Tables provided herein. In some aspects, the stable adalimumab formulation provided herein demonstrates one or more of the following parameters: (i) less than about a 2.1-fold increase in acidic species, as measured by cation-exchange high-performance liquid chromatography (CEX-HPLC) after storage for 28 days at 40.degree. C.; (ii) less than about a 5-fold increase in high molecular weight species (HMWS) species, as measured by size-exclusion chromatography (SE-HPLC) after storage for 28 days at 40.degree. C.; and/or (iii) less than about 500 nephelometric turbidity units (NTUs) after stirring at room temperature for 5 days.
BRIEF DESCRIPTION OF THE DRAWINGS
[0007] FIG. 1 is a bar graph of stability of adalimumab formulations as determined by cation-exchange high-performance liquid chromatography (CEX-HPLC).
[0008] FIG. 2 is a bar graph of stability of adalimumab formulations as determined by size-exclusion high-performance liquid chromatography (SE-HPLC).
[0009] FIG. 3 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0010] FIG. 4 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0011] FIG. 5 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0012] FIG. 6 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0013] FIG. 7 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0014] FIG. 8 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC. The symbol " . . . " indicates a value above the maximum shown on the y-axis.
[0015] FIG. 9 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0016] FIG. 10 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0017] FIG. 11 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0018] FIG. 12 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0019] FIG. 13 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0020] FIG. 14 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0021] FIG. 15 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0022] FIG. 16 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0023] FIG. 17 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0024] FIG. 18 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0025] FIG. 19 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0026] FIG. 20 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0027] FIG. 21 is a bar graph of stability of adalimumab formulations as determined by opalescence measurement.
[0028] FIG. 22 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0029] FIG. 23 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0030] FIG. 24 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0031] FIG. 25 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0032] FIG. 26 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0033] FIG. 27 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0034] FIG. 28 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0035] FIG. 29 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0036] FIG. 30 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0037] FIG. 31 is a bar graph of stability of adalimumab formulations as determined by micro-flow imaging (MFI).
[0038] FIG. 32 is a bar graph of stability of adalimumab formulations as determined by MFI. The symbol " . . . " indicates a value above the maximum shown on the y-axis.
[0039] FIG. 33 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0040] FIG. 34 is a bar graph of stability of adalimumab formulations as determined by MFI. The symbol " . . . " indicates a value above the maximum shown on the y-axis.
[0041] FIG. 35 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0042] FIG. 36 is a bar graph of stability of adalimumab formulations as determined by MFI. The symbol " . . . " indicates a value above the maximum shown on the y-axis.
[0043] FIG. 37 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0044] FIG. 38 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0045] FIG. 39 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0046] FIG. 40 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0047] FIG. 41 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0048] FIG. 42 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0049] FIG. 43 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0050] FIG. 44 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0051] FIG. 45 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0052] FIG. 46 is a bar graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0053] FIG. 47 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0054] FIG. 48 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0055] FIG. 49 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0056] FIG. 50 is a bar graph of stability of adalimumab formulations as determined by SE-HPLC.
[0057] FIG. 51 is a line graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0058] FIG. 52 is a line graph of stability of adalimumab formulations as determined by CEX-HPLC.
[0059] FIG. 53 is a line graph of stability of adalimumab formulations as determined by SE-HPLC.
[0060] FIG. 54 is a line graph of stability of adalimumab formulations as determined by SE-HPLC.
[0061] FIG. 55 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0062] FIG. 56 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0063] FIG. 57 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0064] FIG. 58 is a bar graph of stability of adalimumab formulations (non-transport at 4.degree. C.) as determined by CEX-HPLC.
[0065] FIG. 59 is a bar graph of stability of adalimumab formulations (transport at 4.degree. C.) as determined by CEX-HPLC.
[0066] FIG. 60 is a bar graph of stability of adalimumab formulations (non-transport at 25.degree. C.) as determined by CEX-HPLC.
[0067] FIG. 61 is a bar graph of stability of adalimumab formulations (transport at 25.degree. C.) as determined by CEX-HPLC.
[0068] FIG. 62 is a bar graph of stability of adalimumab formulations (non-transport at 40.degree. C.) as determined by CEX-HPLC.
[0069] FIG. 63 is a bar graph of stability of adalimumab formulations (transport at 40.degree. C.) as determined by CEX-HPLC.
[0070] FIG. 64 is a bar graph of stability of adalimumab formulations (non-transport at 4.degree. C.) as determined by SE-HPLC.
[0071] FIG. 65 is a bar graph of stability of adalimumab formulations (transport at 4.degree. C.) as determined by SE-HPLC.
[0072] FIG. 66 is a bar graph of stability of adalimumab formulations (non-transport at 25.degree. C.) as determined by SE-HPLC.
[0073] FIG. 67 is a bar graph of stability of adalimumab formulations (transport at 25.degree. C.) as determined by SE-HPLC.
[0074] FIG. 68 is a bar graph of stability of adalimumab formulations (non-transport at 40.degree. C.) as determined by SE-HPLC.
[0075] FIG. 69 is a bar graph of stability of adalimumab formulations (transport at 40.degree. C.) as determined by SE-HPLC.
[0076] FIGS. 70A-B are a bar graph of stability of adalimumab formulations as determined by MFI. FIG. 70B is the same data presented in FIG. 70A but graphed at a different scale.
[0077] FIGS. 71A-B are a bar graph of stability of adalimumab formulations as determined by MFI. FIG. 71B is the same data presented in FIG. 71A but graphed at a different scale.
[0078] FIGS. 72A-B are a bar graph of stability of adalimumab formulations as determined by determined by MFI. FIG. 72B is the same data presented in FIG. 72A but graphed at a different scale.
[0079] FIG. 73 is a bar graph of stability of adalimumab formulations (non-transport at 4.degree. C.) as determined by CEX-HPLC.
[0080] FIG. 74 is a bar graph of stability of adalimumab formulations (transport at 4.degree. C.) as determined by CEX-HPLC.
[0081] FIG. 75 is a bar graph of stability of adalimumab formulations (non-transport at 25.degree. C.) as determined by CEX-HPLC.
[0082] FIG. 76 is a bar graph of stability of adalimumab formulations (transport at 25.degree. C.) as determined by CEX-HPLC.
[0083] FIG. 77 is a bar graph of stability of adalimumab formulations as (non-transport at 40.degree. C.) determined by CEX-HPLC.
[0084] FIG. 78 is a bar graph of stability of adalimumab formulations (transport at 40.degree. C.) as determined by CEX-HPLC.
[0085] FIG. 79 is a bar graph of stability of adalimumab formulations (non-transport at 4.degree. C.) as determined by SE-HPLC.
[0086] FIG. 80 is a bar graph of stability of adalimumab formulations (transport at 4.degree. C.) as determined by SE-HPLC.
[0087] FIG. 81 is a bar graph of stability of adalimumab formulations (non-transport at 25.degree. C.) as determined by SE-HPLC.
[0088] FIG. 82 is a bar graph of stability of adalimumab formulations (transport at 25.degree. C.) as determined by SE-HPLC.
[0089] FIG. 83 is a bar graph of stability of adalimumab formulations (non-transport at 40.degree. C.) as determined by SE-HPLC.
[0090] FIG. 84 is a bar graph of stability of adalimumab formulations (transport at 40.degree. C.) as determined by SE-HPLC.
[0091] FIG. 85 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0092] FIG. 86 is a bar graph of stability of adalimumab formulations as determined by MFI.
[0093] FIG. 87 is a bar graph of stability of adalimumab formulations as determined by MFI.
DETAILED DESCRIPTION
[0094] The present disclosure provides stable aqueous adalimumab formulations and related methods of making stable aqueous adalimumab formulations. Also provided are lyophilized forms of the aqueous adalimumab formulations disclosed herein. The present disclosure also provides related uses of the formulations disclosed herein and related methods of administering these formulations to treat diseases such as arthritis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, axial spondyloarthritis, juvenile idiopathic arthritis, enthesitis related arthritis, osteoarthritis, peripheral spondyloarthritis, acute disc prolapse, inflammatory bowel disease, Crohn's disease, ulcerative colitis, intestinal Behcet's disease, chronic pouchitis, small bowel lesions, Hermansky-Pudlak syndrome, psoriasis, psoriasis vulgaris, psoriasis arthropica, plaque psoriasis, hidradenitis suppurativa, interstitial cystitis, sleep apnea, sarcoidosis, retinal vascular disorders, uveitis, choroidal neovascularization, Pyoderma Gangrenosum, giant cell arteritis, Netherton syndrome, anaplastic thyroid cancers, asthma, and refractory asthma.
[0095] The stable aqueous adalimumab formulations include adalimumab and one or more excipients selected such that the formulation demonstrates characteristics suitable for use as a pharmaceutical composition. A formulation suitable for use as a pharmaceutical composition generally exhibits a low amount of high molecular weight species (HMWS), such as aggregates and dimers, and/or also exhibits a low degree of oxidation over time. For example, a suitable adalimumab formulation may exhibit minimal oxidation of residues TRP 53, MET 34, MET 256, and MET 432. A suitable formulation may also exhibit a minimal amount of sub-visible particles (e.g., particles having a diameter of .gtoreq.10 .mu.m or .gtoreq.25 .mu.m) and/or non-spherical particles (e.g., particles having an aspect ratio of .gtoreq.5 .mu.m). High amounts of HMWS, oxidation, and/or particles may impact the shelf-life, safety and/or potency of a formulation. Stable aqueous adalimumab formulations are described in the embodiments set forth below.
[0096] In some cases, the stable aqueous adalimumab formulations include adalimumab, calcium chloride, and optionally one or more (typically one, two, or three) additional excipients as described herein.
[0097] In some cases, the stable aqueous adalimumab formulations include adalimumab, a buffer, and optionally one or more (typically one, two, or three) additional excipients as described herein. Suitable buffers include glutamate/glutamic acid buffers ("glutamate buffer"), adipate/adipic acid buffers ("adipate buffer"), glucuronate/glucuronic acid buffers ("glucuronate buffer"), acetate/acetic acid buffers ("acetate buffer"), benzoate/benzoic acid buffers ("benzoate buffer"), glycolate/glycolic acid buffers ("glycolate buffer"), lactate/lactic acid buffers ("lactate buffer"), and histidine buffers.
[0098] In some cases, the stable aqueous adalimumab formulations include adalimumab and do not include a buffer. Optionally, these adalimumab formulations additionally include one or more (typically one, two, or three) excipients as described herein.
[0099] In some cases, the stable aqueous adalimumab formulation includes adalimumab, a buffer (e.g., lactate buffer), calcium chloride, and optionally one or more (typically one, two, or three) additional excipients as described herein.
[0100] As used herein, a "stable" formulation demonstrates stability sufficient to permit administration to a patient. For example, a stable formulation may demonstrate long-term stability, such as stability upon storage for 6 months or 1 year. Stability of a formulation may, for example, be assessed by growth of acidic species over time, growth of high molecular weight species over time, or increase in opalescence over time. When stability is assessed by growth of acidic species over time, a stable formulation may demonstrate less than about a 4-fold increase (e.g., less than about a 3.5-fold increase, less than about a 3-fold increase, less than about a 2.5-fold increase, less than about a 2.4-fold increase, less than about a 2.3-fold increase, less than about a 2.25-fold increase, less than about a 2.2-fold increase, less than about a 2.15-fold increase, less than about a 2.1-fold increase, less than about a 2.05-fold increase, or less than about a 2-fold increase) in acidic species as measured by CEX-HPLC after storage for 28 days at 40.degree. C. When stability is assessed by growth of high molecular weight species over time, a stable formulation may demonstrate less than about a 5-fold increase (e.g., less than about a 4.5-fold increase, less than about a 4-fold increase, less than about a 3.9-fold increase, less than about a 3.8-fold increase, less than about a 3.7-fold increase, less than about a 3.6-fold increase, less than about a 3.5-fold increase, less than about a 3.4-fold increase, less than about a 3.3-fold increase, less than about a 3.2-fold increase, less than about a 3.15-fold increase, less than about a 3.1-fold increase, less than about a 3.05-fold increase, less than about a 3-fold increase, less than about a 2.95-fold increase, or less than about a 2.9-fold increase) in HMWS species as measured by SE-HPLC after storage for 28 days at 40.degree. C. When stability is assessed by increase in opalescence over time, a stable formulation may demonstrate less than about 500 nephelometric turbidity units (NTUs) (e.g., less than about 400 NTUs, less than about 350 NTUs, less than about 300 NTUs, less than about 250 NTUs, less than about 200 NTUs, less than about 150 NTUs, less than about 140 NTUs, less than about 130 NTUs, less than about 125 NTUs, less than about 120 NTUs, less than about 115 NTUs, less than about 110 NTUs, less than about 100 NTUs, less than about 90 NTUs, less than about 80 NTUs, or less than about 70 NTUs) after stirring at room temperature for 5 days.
[0101] As used herein, an "aqueous" formulation contains water. Aqueous formulations can be in a liquid state or a frozen state, and preferably are liquid formulations.
[0102] As used herein, an "excipient" is a component of a formulation other than water and the active agent (e.g., adalimumab or biosimilar thereof) added to the formulation. Examples of excipients include buffers; stabilizers such as amino acids and amino acid derivatives, polyethylene glycols and polyethylene glycol derivatives, polyols, acids, amines, polysaccharides or polysaccharide derivatives, salts, and surfactants; and pH-adjusting agents.
[0103] As used herein, a "biosimilar," particularly an adalimumab biosimilar, is a biological product that is highly similar to HUMIRA (also known as adalimumab or D2E7) notwithstanding minor differences in clinically inactive components; and there are no clinically meaningful differences between the biological product and HUMIRA in terms of safety, purity, and potency of the product.
[0104] As used herein, the term "about," when used to modify a particular value or range, generally means within 20 percent, e.g., within 10 percent, 5 percent, 4 percent, 3 percent, 2 percent, or 1 percent of the stated value or range.
[0105] Adalimumab is a fully human monoclonal antibody of the immunoglobulin G1 (IgG1) subclass expressed in the Chinese hamster ovary (CHO) cell line and consists of 2 heavy chains (HC), and 2 light chains (LC) of the kappa subclass. Adalimumab contains 32 total cysteine residues involved in both intrachain and interchain disulfide bonds. Each HC contains 451 amino acids with 4 intrachain disulfides. Each LC contains 214 amino acids with 2 intrachain disulfides. Each HC contains an N-linked glycan at the consensus glycosylation site on Asn301. The amino acid sequences of the adalimumab variable LC and variable HC are set out at SEQ ID NO: 1 and 2, respectively and the full length LC and HC are set out as SEQ ID NO: 3 and 4; respectively. In addition, the adalimumab LC CDRs are set out as SEQ ID NO: 5 (LC CDR1), SEQ ID NO: 6 (LC CDR2) and SEQ ID NO: 7 (LC CDR3). Adalimumab HC CDRs are set out as SEQ ID NO: 8 (HC CDR1), SEQ ID No: 9 (HC CDR2), and SEQ ID NO: 10 (HC CDR3). Adalimumab has been described and claimed in U.S. Pat. No. 6,090,382, the disclosure of which is hereby incorporated by reference in its entirety. As used herein, the term "adalimumab" includes biosimilars of adalimumab.
Formulations of Adalimumab with Calcium Chloride
[0106] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 80 to about 120 mg/mL, about 90 to about 110 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, calcium chloride at a concentration of about 1 to about 150 mM, such as about 5 to about 50 mM, about 5 to about 30 mM, about 10 to about 30 mM, about 12.5 to about 17.5 mM about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, about 10 mM, about 15 mM, about 20 mM, about 25 mM, or about 30 mM, and one or more excipients as discussed below. The presence of calcium chloride in the formulations advantageously and unexpectedly provides improved stability over time as compared to other salts, particularly with respect to levels of acidic species at 40.degree. C. as detected by cation-exchange high-performance liquid chromatography (CEX-HPLC). Further, the presence of calcium chloride at low concentrations (e.g., about 20 to about 25 mM, about 10 mM to about 20 mM, about 12.5 mM to about 17.5 mM, or about 15 mM) advantageously lowers the rate of growth of acidic species without significantly increasing the growth of high molecular weight species (HMWS) at 40.degree. C. as detected by size-exclusion high-performance liquid chromatography (SE-HPLC).
[0107] Increased levels of acidic species over time are generally due to protein deamidation. It is therefore beneficial for the stable aqueous adalimumab formulations to demonstrate minimal growth of acidic species over time. Similarly, it is beneficial for the stable aqueous adalimumab formulations to demonstrate minimal growth over time of HMWS because HMWS provide a measure of soluble aggregation.
[0108] Further still, the presence of calcium chloride at low concentrations (e.g., about 10 mM to about 20 mM, about 12.5 mM to about 17.5 mM or about 15 mM) advantageously minimizes formation of sub-visible and/or non-spherical particles in a formulation, as detected by micro-flow imaging ("MFI") even if the formulation has been subjected to transport conditions.
[0109] Suitable excipients for combination with the calcium chloride-containing adalimumab formulations include certain buffers, and certain stabilizers such as certain amino acids and amino acid derivatives, certain polyethylene glycols and polyethylene glycol derivatives, certain polyols, certain acids, certain amines, certain polysaccharides or polysaccharide derivatives, and certain surfactants. Examples of suitable buffers include glutamate (e.g., at a concentration of about 5 mM to about 50 mM, about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), adipate (e.g., at a concentration of about 5 mM to about 50 mM, about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), glucuronate (e.g., at a concentration of about 5 mM to about 50 mM, about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), acetic acid and/or acetate (e.g., at a concentration of about 0.1 mM to about 300 mM, about 2 mM to about 30 mM, about 5 mM to about 50 mM, about 5 mM to about 15 mM, about 10 mM to about 20 mM, about 10 mM to about 30 mM, about 15 mM to about 25 mM, about 30 mM to about 40 mM, about 35 mM to about 45 mM, about 40 mM to about 50 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), benzoate (e.g., at a concentration of about 5 mM to about 50 mM, about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), glycolate (e.g., at a concentration of about 5 mM to about 50 mM, about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), lactic acid and/or lactate (e.g., at a concentration of about 0.1 mM to about 300 mM, about 2 mM to about 30 mM, about 10 mM to about 30 mM, about 5 mM to about 15 mM, about 7 mM to about 12 mM, about 9 mM to about 11 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and histidine (e.g., at a concentration of about 5 mM to about 50 mM, about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM). Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 50 to about 320 mM, and/or about 50 to about 300 mM), N-acetyl arginine (e.g., at a concentration of about 0.1 to about 450 mM or about 90 to about 150 mM), citruline (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), sarcosine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), N-acetyl proline (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), N-acetyl ornithine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), ornithine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), beta-alanine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), alanine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), asparagine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), isoleucine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), serine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), aspartic acid (e.g., at a concentration of about 0.1 to about 450 mM or about 10 to about 30 mM), creatine (e.g., at a concentration of about 0.1 to about 450 mM or about 15 to about 35 mM), glutamine (e.g., at a concentration of about 0.1 to about 450 mM or about 40 to about 60 mM), phenylalanine (e.g., at a concentration of about 0.1 to about 450 mM or about 40 to about 60 mM), tryptophan (e.g., at a concentration of about 0.1 to about 450 mM or about 15 to about 35 mM), and arginine-HCl (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM). Examples of suitable polyethylene glycols and polyethylene glycol derivatives include PEG 15 hydroxystearate (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v) or about 3% (w/v) to about 6% (w/v)), PEG 3350 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v) or about 1% (w/v) to about 7% (w/v)), PEG 200 (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v) or about 0.6% (w/v) to about 4.8% (w/v)), PEG 600 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v) or about 1.2% (w/v) to about 14.5% (w/v)), and PEG 400 (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v) or about 0.3% (w/v) to about 1.5% (w/v)). Examples of suitable polyols include inositol (e.g., at a concentration of about 0.1 to about 450 mM or about 150 to about 210 mM), glycerol (also referred to as glycerin) (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.5% (w/v) to about 1% (w/v)), sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 6% (w/v) to about 8.5% (w/v), about 6.2% (w/v) to about 7.3% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 7.4% (w/v), or about 9% (w/v)), and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include glycolic acid (e.g., at a concentration of about 0.1 to about 300 mM or about 50 to about 70 mM), pyrollidone carboxylic acid (PCA) (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.05% (w/v) to about 2% (w/v)), medronic acid (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), benzene sulfonic acid (e.g., at a concentration of about 0.1 to about 300 mM or about 60 to about 90 mM), and methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, and/or about 10 to about 30 mM). Examples of suitable amines include monoethanolamine hydrochloride (MEA-HCl) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM), monoethanolamine (MEA) (e.g., at a concentration of about 0.1 to about 300 mM, about 0.1 to about 50 mM, and/or about 30 to about 160 mM), and triethanolamine (TEA) (e.g., at a concentration of about 0.1 to about 170 mM or about 30 to about 150 mM). Examples of suitable polysaccharides or polysaccharide derivatives include hyaluronic acid (e.g., at a concentration of about 0.05% (w/v) to about 2.5% (w/v) or about 0.1% (w/v) to about 0.05% (w/v)), sodium carboxymethylcellulose (NaCMC) (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.1% (w/v) to about 2% (w/v)), and dextran (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 8% (w/v) to about 12% (w/v)). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 0.2% (w/v), about 0.03% (w/v) to about 0.06% (w/v), about 0.01% (w/v), about 0.05% (w/v), about 0.06% (w/v), and/or about 0.1% (w/v)), Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), Docusate sodium (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), benzalkonium chloride (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.05% (w/v) to about 0.5% (w/v)), Span 40 (sorbitan monopalmitate) (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.05% (w/v) to about 0.5% (w/v)), and Triton X-100 (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)). Examples of other suitable excipients include imidazole (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.5% (w/v) to about 2% (w/v)), taurine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), betaine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), gelatin (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.5% (w/v) to about 2% (w/v)), niacinamide (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 120 mM), polyvinylpyrrolidone (PVP), for example, 10K PVP, (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v) or about 0.05% (w/v) to about 2% (w/v)), guanidine hydrochloride (GnHCl) (e.g., at a concentration of about 0.1 to about 150 mM or about 10 to about 30 mM), and ethanol (e.g., at a concentration of about 0.05% (w/v) to about 2.5% (w/v) or about 0.25% (w/v) to about 1% (w/v)). Optionally, the calcium chloride-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0110] Suitable excipients for combination with the calcium chloride-containing adalimumab formulations also include, but are not limited to, glutamate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), adipate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), glucuronate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), acetic acid and/or acetate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 5 mM to about 50 mM, about 5 mM to about 15 mM, about 10 mM to about 20 mM, about 10 mM to about 30 mM, about 15 mM to about 25 mM, about 30 mM to about 40 mM, about 35 mM to about 45 mM, about 40 mM to about 50 mM, about 10 mM, about 15 mM, about 20 mM, and/or 25 mM), benzoate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), glycolate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), lactic acid and/or lactate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 10 mM to about 30 mM, about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), histidine at a concentration of about 5 mM to about 50 mM (e.g., about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 320 mM and/or about 50 to about 300 mM), N-acetyl arginine at a concentration of about 0.1 to about 450 mM (e.g., about 90 to about 150 mM), citruline at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), sarcosine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), N-acetyl proline at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), N-acetyl ornithine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), ornithine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), beta-alanine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), alanine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), asparagine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), isoleucine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), serine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), aspartic acid at a concentration of about 0.1 to about 450 mM (e.g., about 10 to about 30 mM), creatine at a concentration of about 0.1 to about 450 mM (e.g., about 15 to about 35 mM), glutamine at a concentration of about 0.1 to about 450 mM (e.g., about 40 to about 60 mM), phenylalanine at a concentration of about 0.1 to about 450 mM (e.g., about 40 to about 60 mM), tryptophan at a concentration of about 0.1 to about 450 mM (e.g., about 15 to about 35 mM), arginine-HCl at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), PEG 15 hydroxystearate at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 3% (w/v) to about 6% (w/v)), PEG 3350 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 1% (w/v) to about 7% (w/v)), PEG 200 at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 0.6% (w/v) to about 4.8% (w/v)), PEG 600 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 1.2% (w/v) to about 14.5% (w/v)), PEG 400 at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 0.3% (w/v) to about 1.5% (w/v)), inositol at a concentration of about 0.1 to about 450 mM (e.g., about 150 to about 210 mM), glycerol (also referred to as glycerin) at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.5% (w/v) to about 1% (w/v)), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 6.2% (w/v) to about 7.3% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), glycolic acid at a concentration of about 0.1 to about 300 mM (e.g., about 50 to about 70 mM), PCA at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.05% (w/v) to about 2% (w/v)), medronic acid at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), benzene sulfonic acid at a concentration of about 0.1 to about 300 mM (e.g., about 60 to about 90 mM), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM and/or about 10 to about 30 mM), monoethanolamine hydrochloride (MEA-HCl) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), monoethanolamine (MEA) at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM and/or about 30 to about 160 mM), triethanolamine (TEA) at a concentration of about 0.1 to about 170 mM (e.g., about 30 to about 150 mM), hyaluronic acid at a concentration of about 0.05% (w/v) to about 2.5% (w/v) (e.g., about 0.1% (w/v) to about 0.05% (w/v)), sodium carboxymethylcellulose (NaCMC) at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.1% (w/v) to about 2% (w/v)), dextran at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 8% (w/v) to about 12% (w/v)), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), Docusate sodium at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), benzalkonium chloride at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.05% (w/v) to about 0.5% (w/v)), Span 40 (sorbitan monopalmitate) at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.05% (w/v) to about 0.5% (w/v)), Triton X-100 at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), imidazole at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), taurine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), betaine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), gelatin at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), niacinamide at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 120 mM), polyvinylpyrrolidone (PVP), for example, 10K PVP, at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.05% (w/v) to about 2% (w/v)), guanidine hydrochloride (GnHCl) at a concentration of about 0.1 to about 150 mM (e.g., about 10 to about 30 mM), and ethanol at a concentration of about 0.05% (w/v) to about 2.5% (w/v) (e.g., about 0.25% (w/v) to about 1% (w/v)). Optionally, the calcium chloride-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0111] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 5% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0112] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 140 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, dextran at a concentration of about 5% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0113] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.5% (w/v) to about 2% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0114] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0115] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0116] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0117] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), PEG 3350 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0118] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, TEA at a concentration of about 100 mM to about 200 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0119] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, TEA at a concentration of about 20 mM to about 40 mM, calcium chloride at a concentration of about 60 mM to about 90 mM, and has a pH of about 5.0 to about 5.5.
[0120] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, alanine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0121] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, asparagine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0122] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, isoleucine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0123] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, serine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0124] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, aspartic acid at a concentration of about 10 mM to about 30 mM, proline at a concentration of about 60 mM to about 100 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0125] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, creatine at a concentration of about 15 mM to about 35 mM, proline at a concentration of about 60 mM to about 90 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0126] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, glutamine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0127] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, leucine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0128] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, phenylalanine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0129] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, tryptophan at a concentration of about 10 mM to about 40 mM, proline at a concentration of about 60 mM to about 90 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0130] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0131] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, proline at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0132] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 3% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0133] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 3% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, proline at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0134] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, imidazole at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0135] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0136] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, inositol at a concentration of about 200 to about 300 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0137] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0138] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, taurine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0139] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, citruline at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0140] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, betaine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0141] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sarcosine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0142] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, glycolic acid at a concentration of about 40 to about 80 mM, calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0143] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PCA at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0144] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, gelatin at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0145] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, hyaluronic acid at a concentration of about 0.1% (w/v) to about 0.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0146] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl proline at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0147] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl ornithine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0148] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ornithine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0149] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, beta-alanine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0150] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0151] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, medronic acid at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0152] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0153] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), and calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0154] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 80 to about 120 mM, calcium chloride at a concentration of about 60 to about 100 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0155] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzene sulfonic acid at a concentration of about 60 to about 90 mM, calcium chloride at a concentration of about 25 to about 75 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0156] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.25% (w/v) to about 0.75% (w/v), calcium chloride at a concentration of about 40 to about 80 mM, glycerin at a concentration of about 0.5% (w/v) to about 1% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0157] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 50 mM, and MSA at a concentration of about 10 to about 30 mM, and has a pH of about 5.0 to about 5.5.
[0158] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 60 to about 90 mM, PEG 400 at a concentration of about 0.1% (w/v) to about 0.5% (w/v), and 10K PVP at a concentration of about 0.5% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0159] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0160] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 60 to about 90 mM, and has a pH of about 5.0 to about 5.5.
[0161] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), PEG 400 at a concentration of about 1% (w/v) to about 3% (w/v), and calcium chloride at a concentration of about 10 to about 30 mM, and has a pH of about 5.0 to about 5.5.
[0162] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0163] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0164] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0165] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0166] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Docusate sodium at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0167] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and benzalkonium chloride at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0168] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Span 40 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0169] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0170] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, and arginine-HCl at a concentration of about 80 to about 120 mM, and has a pH of about 5.1 to 5.3.
[0171] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0172] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0173] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0174] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0175] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0176] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0177] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0178] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0179] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0180] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0181] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0182] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.2% (w/v) to about 0.6% (w/v), and has a pH of about 5.1 to 5.3.
[0183] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.1% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0184] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.0% (w/v) to about 6.5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0185] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.7% (w/v) to about 7.3% (w/v), calcium chloride at a concentration of about 5 to about 15 mM, guanidine hydrochloride (GnHCl) at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0186] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.7% (w/v) to about 7.3% (w/v), calcium chloride at a concentration of about 5 to about 15 mM, NaCMC at a concentration of about 0.2% (w/v) to about 1% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0187] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0188] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0189] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0190] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.6% (w/v) to about 7% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0191] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0192] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0193] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0194] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 240 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0195] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 200 to about 250 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0196] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0197] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0198] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0199] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 3% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.2% (w/v) to about 2% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0200] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 200 at a concentration of about 3.5% (w/v) to about 4.2% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0201] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 5% (w/v) to about 5.7% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 90 to about 130 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0202] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0203] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, and/or Ca(OH).sub.2.
[0204] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, histidine at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 270 to about 370 mM, and calcium chloride at a concentration of about 10 to about 30 mM, and has a pH of about 6.7 to 6.9. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0205] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 8% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0206] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 6.5% (w/v) to about 7.3% (w/v), PEG 200 at a concentration of about 0.3% (w/v) to about 1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0207] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 4% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 2.1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0208] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 0.9% (w/v) to about 1.5% (w/v), PEG 200 at a concentration of about 2.5% (w/v) to about 3.5% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0209] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 200 at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0210] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 6.9% (w/v) to about 7.7% (w/v), proline at a concentration of about 40 to about 80 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0211] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 5% (w/v) to about 6% (w/v), proline at a concentration of about 90 to about 150 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0212] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 2% (w/v) to about 3% (w/v), proline at a concentration of about 150 to about 210 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0213] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, proline at a concentration of about 200 to about 300 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0214] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl and/or NaOH.
[0215] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 5% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0216] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, proline at a concentration of about 200 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0217] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0218] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, sucrose at a concentration of about 5% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0219] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0220] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 200 at a concentration of about 4.5% (w/v) to about 5.1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0221] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl.
[0222] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 13% (w/v) to about 16% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0223] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 3% (w/v) to about 3.6% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 150 to about 210 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0224] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), and calcium chloride at a concentration of about 15 to about 35 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0225] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 6% (w/v) to about 7% (w/v), and calcium chloride at a concentration of about 40 to about 60 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0226] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 3.4% (w/v) to about 4% (w/v), and calcium chloride at a concentration of about 65 to about 85 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0227] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL and calcium chloride at a concentration of about 80 to about 120 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0228] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 200 to about 250 mM, and calcium chloride at a concentration of about 15 to about 35 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0229] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 120 to about 180 mM, and calcium chloride at a concentration of about 40 to about 60 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0230] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 60 to about 90 mM, and calcium chloride at a concentration of about 65 to about 85 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0231] In an embodiment, a lyophilized form of any one of the foregoing calcium chloride-containing adalimumab formulations is provided.
[0232] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 mg/ml to about 200 mg/ml, or about 160 mg/ml to about 190 mg/mL, or about 80 mg/ml to about 120 mg/ml, or about 90 mg/ml to about 110 mg/ml, or about 95 mg/ml to about 105 mg/ml, or about 40 mg/ml, or about 45 mg/ml, or about 50 mg/ml, or about 55 mg/ml, or about 60 mg/ml, or about 65 mg/ml, or about 70 mg/ml, or about 75 mg/ml, or about 80 mg/ml, or about 85 mg/ml, or about 90 mg/ml, or about 95 mg/ml, or about 100 mg/ml, or about 105 mg/ml, or about 110 mg/ml, or about 115 mg/ml, or about 120 mg/ml, or about 125 mg/ml, or about 130 mg/ml, or about 135 mg/ml, or about 140 mg/ml, or about 145 mg/ml, or about 150 mg/ml, or about 155 mg/ml, or about 160 mg/ml, or about 165 mg/ml, or about 170 mg/ml, or about 175 mg/ml, or about 180 mg/ml, or about 185 mg/ml, or about 190 mg/mL, with the following excipients:
(a) lactate buffer at a concentration of about 5 mM to about 15 mM, or about 7 mM to about 12 mM, or about 9 mM to about 11 mM, or about 5 mM, or about 6 mM, or about 7 mM, or about 8 mM, or about 8 mM, or about 9 mM, or about 10 mM, or about 11 mM, or about 12 mM, or about 13 mM, or about 14 mM, or about 15 mM; (b) calcium chloride at a concentration of about 5 to about 30 mM, about 10 to about 20 mM, or about 12.5 mM to about 17.5 mM, or about 14 mM to about 16 mM, or about 10 mM, or about 10.5 mM, or about 11 mM, or about 11.5 mM, or about 12 mM, or about 12.5 mM, or about 13 mM, or about 13.5 mM, or about 14 mM, or about 14.5 mM, or about 15 mM, or about 15.5 mM, or about 16 mM, or about 16.5 mM, or about 17 mM, or about 17.5 mM, or about 18 mM, or about 18.5 mM, or about 19 mM, or about 19.5 mM, or about 20 mM; (c) sucrose at a concentration of about 4% (w/v) to about 10% (w/v), or about 6% (w/v) to about 8.5% (w/v), or about 4% (w/v), or about 4.5% (w/v), or about 5% (w/v), or about 5.5% (w/v), or about 6% (w/v), or about 6.5% (w/v), or about 7% (w/v), or about 7.1% (w/v), or about 7.2% (w/v), or about 7.3% (w/v), or about 7.4% (w/v), or about 7.5% (w/v), or about 7.6% (w/v), or about 7.7% (w/v), or about 7.8% (w/v), or about 8% (w/v), or about 8.5% (w/v); (d) Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.09% (w/v), or about 0.03% (w/v) to about 0.06% (w/v), about 0.01% (w/v) to about 0.2% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), or about 0.05% (w/v) to about 0.07% (w/v), or about 0.03% (w/v), or about 0.04% (w/v), or about 0.05% (w/v), or about 0.06% (w/v), or about 0.07% (w/v), or about 0.08% (w/v), about 0.09% (w/v), or about 0.1% (w/v); and (e) has a pH of about 3.5 to about 8, or about 4 to about 7, or about 4.5 to about 6, or about 5 to about 5.5, or about 3.5, or about 4, or about 4.5, or about 4.6, or about 4.7, or about 4.8, or about 4.9, or about 5.0, or about 5.1, or about 5.2, or about 5.3, or about 5.4, or about 5.5, or about 5.6, or about 5.7, or about 5.8, or about 5.9, or about 6.0, or about 6.5, or about 7.0, or about 7.5, or about 8.0.
[0233] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration or about 40 mg/ml to 200 mg/ml, lactate buffer at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 5 to about 30 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 3.5 to 8.
[0234] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration or about 40 mg/ml to 200 mg/ml, lactate buffer at a concentration of about 1 mM to about 15 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 10 to about 20 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 3.5 to 8.
[0235] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 mg/ml to about 120 mg/mL, lactate buffer at a concentration of about 7 mM to about 12 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 12.5 to about 17.5 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.07% (w/v), and a pH of about 4 to about 7.
[0236] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 mg/ml to about 110 mg/mL, lactate buffer at a concentration of about 9 mM to about 11 mM, sucrose at a concentration of about 6% (w/v) to about 8.5% (w/v), calcium chloride at a concentration of about 14 to about 16 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.07% (w/v), and a pH of about 5.0 to about 5.5.
[0237] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 100 mg/mL, lactate buffer at a concentration of about 10 mM, sucrose at a concentration of about 7.4% (w/v), calcium chloride at a concentration of about 15 mM, and Pluronic F68 at a concentration of about 0.06% (w/v), and a pH of about 5.2.
[0238] In an embodiment, the stable aqueous adalimumab formulation is one of the lactate buffer formulations described in Tables G, H, I, 1, 11, 12, 13, 15, 16 or 17 provided herein.
[0239] In an embodiment, the stable aqueous adalimumab formulations described above, which include lactate buffer, calcium chloride, and Pluronic F68, are in lyophilized form. In an embodiment, the stable aqueous adalimumab formulations described above, which include lactate, calcium chloride, and Pluronic F68, are not in lyophilized form (e.g., are hydrated).
[0240] In an embodiment, the pH of the stable aqueous adalimumab formulations described above, which include lactate buffer, calcium chloride, and Pluronic F68, can be adjusted using HCl/Ca(OH).sub.2. In an embodiment, the pH of the stable aqueous adalimumab formulations described above, which include lactate buffer, calcium chloride, and Pluronic F68, is not adjusted using a pH adjusting agent.
[0241] The stable aqueous adalimumab formulations described above, which include lactate buffer, calcium chloride, and Pluronic F68, exhibit a conductivity of less than about 4 mS/cm, or less than about 3.5 mS/cm, or less than about 3 mS/cm, or less than about 2.5 mS/cm, or less than about 2 mS/cm, or less than about 1.5 mS/cm, or less than about 1 mS/cm, or less than about 0.5 mS/cm, or about 0.5 mS/cm to about 3.5 mS/cm at ambient room temperature.
[0242] The stable aqueous adalimumab formulations described above, which include lactate buffer, calcium chloride, and Pluronic F68, exhibit an osmolality of 270-330 mOsM, or about 300 mOsM.
[0243] Aqueous adalimumab formulations having the specific combination of about 5 to about 30 mM (e.g., 12.5 mM to about 17.5 mM, or about 15 mM) calcium chloride, about 5 mM to about 15 mM (e.g., about 7 mM to about 12 mM, or about 9 mM to about 11 mM, or about 10 mM) lactate buffer, and Pluronic F68 surfactant (e.g., about 0.03% (w/v) to about 0.1% (w/v); about 0.03% (w/v) or about 0.06% (w/v)), advantageously exhibit a lowered growth rate of acidic species, as detected by CEX-HPLC), a lowered growth rate of HMWS, as detected by SE-HPLC, and a minimal amount of sub-visible and/or non-spherical particles, as detected by MFI. See, e.g., Examples 15-17 below.
Formulations of Adalimumab with Glutamate Buffer
[0244] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glutamate at a concentration of about 5 mM to about 50 mM (e.g., about 5 mM to about 30 mM, about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyethylene glycols and polyethylene glycol derivatives, certain polyols, certain acids, certain amines, certain polysaccharides or polysaccharide derivatives, certain salts, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM or about 50 to about 300 mM), arginine (e.g., at a concentration of about 0.1 to about 450 mM or about 60 to about 90 mM), N-acetyl arginine (e.g., at a concentration of about 0.1 to about 450 mM or about 90 to about 150 mM), citruline (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), sarcosine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), N-acetyl proline (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), N-acetyl ornithine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), ornithine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), beta-alanine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), alanine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), asparagine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), isoleucine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), serine (e.g., at a concentration of about 0.1 to about 450 mM or about 80 to about 120 mM), aspartic acid (e.g., at a concentration of about 0.1 to about 450 mM or about 10 to about 30 mM), creatine (e.g., at a concentration of about 0.1 to about 450 mM or about 15 to about 35 mM), glutamine (e.g., at a concentration of about 0.1 to about 450 mM or about 40 to about 60 mM), leucine (e.g., at a concentration of about 0.1 to about 450 mM or about 40 to about 60 mM), phenylalanine (e.g., at a concentration of about 0.1 to about 450 mM or about 40 to about 60 mM), and tryptophan (e.g., at a concentration of about 0.1 to about 450 mM or about 15 to about 35 mM). Examples of suitable polyethylene glycols and polyethylene glycol derivatives include PEG 15 hydroxystearate (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v) or about 2.5% (w/v) to about 5% (w/v)), PEG 3350 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v), about 0.5% (w/v) to about 2% (w/v), about 1% (w/v) to about 7% (w/v), about 5% (w/v) to about 10% (w/v), and/or about 6% (w/v) to about 8% (w/v)), PEG 600 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v), about 1% (w/v) to about 4% (w/v), and/or about 1% (w/v) to about 2% (w/v)), PEG 400 (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v), about 0.5% (w/v) to about 10% (w/v), about 0.2% (w/v) to about 2% (w/v), and/or about 6% (w/v) to about 12% (w/v)), and PEG 200 (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 1% (w/v) to about 4% (w/v), and/or about 0.05% (w/v) to about 5% (w/v)). Examples of suitable polyols include glycerol (also referred to as glycerin) (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 0.7% (w/v) to about 2.5% (w/v), and/or about 0.5% (w/v) to about 4% (w/v)), inositol (e.g., at a concentration of about 0.1 to about 450 mM or about 180 to about 250 mM), sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include glycolic acid (e.g., at a concentration of about 0.1 to about 300 mM or about 50 to about 70 mM), PCA (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.05% (w/v) to about 2% (w/v)), medronic acid (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, and/or about 20 to about 125 mM), benzene sulfonic acid (e.g., at a concentration of about 0.1 to about 300 mM or about 75 to about 150 mM), and adipic acid (e.g., at a concentration of about 0.1 to about 300 mM or about 120 to about 180 mM). Examples of suitable amines include triethanolamine (TEA) (e.g., at a concentration of about 0.1 to about 170 mM or about 30 to about 150 mM), monoethanolamine hydrochloride (MEA-HCl) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM), monoethanolamide (MEA) (e.g., at a concentration of about 0.1 to about 300 mM, about 0.1 to about 50 mM, about 0.1 to about 170 mM, and/or about 30 to about 160 mM). Examples of suitable polysaccharides or polysaccharide derivatives include dextran (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 2% (w/v) to about 10% (w/v)) and hyaluronic acid (e.g., at a concentration of about 0.05% (w/v) to about 2.5% (w/v) or about 0.1% (w/v) to about 0.05% (w/v)). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sodium borate (e.g., at a concentration of about 0.1 to about 150 mM or about 60 to about 90 mM), sodium bicarbonate (e.g., at a concentration of about 0.1 to about 150 mM or about 60 to about 90 mM), sodium sulfate (e.g., at a concentration of about 0.1 to about 150 mM or about 60 to about 90 mM), calcium sulfate (e.g., at a concentration of about 0.1 to about 150 mM or about 10 to about 30 mM), ammonium sulfate (e.g., at a concentration of about 0.1 to about 150 mM or about 60 to about 90 mM), sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), and magnesium chloride (e.g., at a concentration of about 0.1 to about 150 mM or about 60 to about 90 mM). Examples of suitable surfactants include benzalkonium chloride (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), guanidine HCl (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), lecithin (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), oleic acid (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)), polyvinyl alcohol, for example, 31K or 205 K polyvinyl alcohol, (e.g., at a concentration of about 0.01% (w/v) to about 10% (w/v) or about 0.05% (w/v) to about 0.5% (w/v)), polyvinylpyrrolidone (PVP), for example, 10K PVP, (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.01% (w/v) to about 1% (w/v), and/or about 0.005% (w/v) to about 2% (w/v)), and protamine sulfate (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)). Examples of other suitable excipients include imidazole (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 1% (w/v) to about 2% (w/v)), taurine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), betaine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM), gelatin (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 0.5% (w/v) to about 2% (w/v)), niacinamide (e.g., at a concentration of about 0.1 to about 450 mM, about 100 to about 270 mM, and/or about 100 to about 150 mM), and ethanol (e.g., at a concentration of about 0.05% (w/v) to about 2.5% (w/v) or about 0.25% (w/v) to about 1.4% (w/v)). Optionally, the glutamate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0245] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glutamate at a concentration of about 5 mM to about 50 mM (e.g., about 5 mM to about 30 mM, about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of PEG 3350 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v), about 1% (w/v) to about 7% (w/v), about 5% (w/v) to about 10% (w/v), and/or about 6% (w/v) to about 8% (w/v)), PEG 600 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 1% (w/v) to about 4% (w/v) and/or about 1% (w/v) to about 2% (w/v)), PEG 400 at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 0.5% (w/v) to about 10% (w/v), about 0.2% (w/v) to about 2% (w/v) and/or about 6% (w/v) to about 12% (w/v)), PEG 200 at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 1% (w/v) to about 4% (w/v) and/or about 0.05% (w/v) to about 5% (w/v)), glycerol (also referred to as glycerin) at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.7% (w/v) to about 2.5% (w/v) and/or about 0.5% (w/v) to about 4% (w/v)), polyvinylpyrrolidone (PVP), for example, 10K PVP, at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.01% (w/v) to about 1% (w/v) and/or about 0.005% (w/v) to about 2% (w/v)), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM), PEG 15 hydroxystearate at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 2.5% (w/v) to about 5% (w/v)), arginine at a concentration of about 0.1 to about 450 mM (e.g., about 60 to about 90 mM), dextran at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 2% (w/v) to about 10% (w/v)), imidazole at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 1% (w/v) to about 2% (w/v)), N-acetyl arginine at a concentration of about 0.1 to about 450 mM (e.g., about 90 to about 150 mM), inositol at a concentration of about 0.1 to about 450 mM (e.g., about 180 to about 250 mM), taurine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), citruline at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), betaine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), sarcosine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), glycolic acid at a concentration of about 0.1 to about 300 mM (e.g., about 50 to about 70 mM), PCA at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.05% (w/v) to about 2% (w/v)), gelatin at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), hyaluronic acid at a concentration of about 0.05% (w/v) to about 2.5% (w/v) (e.g., about 0.1% (w/v) to about 0.05% (w/v)), N-acetyl proline at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), N-acetyl ornithine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), ornithine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), beta-alanine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), niacinamide at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 270 mM and/or about 100 to about 150 mM), medronic acid at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM and/or about 20 to about 125 mM), triethanolamine (TEA) at a concentration of about 0.1 to about 170 mM (e.g., about 30 to about 150 mM), monoethanolamide hydrochloride (MEA-HCl) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), monoethanolamide (MEA) at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM, about 0.1 to about 170 mM, and/or about 30 to about 160 mM), ethanol at a concentration of about 0.05% (w/v) to about 2.5% (w/v) (e.g., about 0.25% (w/v) to about 1.4% (w/v)), benzene sulfonic acid at a concentration of about 0.1 to about 300 mM (e.g., about 75 to about 150 mM), adipic acid at a concentration of about 0.1 to about 300 mM (e.g., about 120 to about 180 mM), sodium borate at a concentration of about 0.1 to about 150 mM (e.g., about 60 to about 90 mM), sodium bicarbonate at a concentration of about 0.1 to about 150 mM (e.g., about 60 to about 90 mM), sodium sulfate at a concentration of about 0.1 to about 150 mM (e.g., about 60 to about 90 mM), calcium sulfate at a concentration of about 0.1 to about 150 mM (e.g., about 10 to about 30 mM), ammonium sulfate at a concentration of about 0.1 to about 150 mM (e.g., about 60 to about 90 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), magnesium chloride at a concentration of about 0.1 to about 150 mM (e.g., about 60 to about 90 mM), alanine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), asparagine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), isoleucine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), serine at a concentration of about 0.1 to about 450 mM (e.g., about 80 to about 120 mM), aspartic acid at a concentration of about 0.1 to about 450 mM (e.g., about 10 to about 30 mM), creatine at a concentration of about 0.1 to about 450 mM (e.g., about 15 to about 35 mM), glutamine at a concentration of about 0.1 to about 450 mM (e.g., about 40 to about 60 mM), leucine at a concentration of about 0.1 to about 450 mM (e.g., about 40 to about 60 mM), phenylalanine at a concentration of about 0.1 to about 450 mM (e.g., about 40 to about 60 mM), tryptophan at a concentration of about 0.1 to about 450 mM (e.g., about 15 to about 35 mM), benzalkonium chloride at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), guanidine HCl at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), lecithin at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), oleic acid at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)), polyvinyl alcohol, for example, 31K or 205 K polyvinyl alcohol, at a concentration of about 0.01% (w/v) to about 10% (w/v) (e.g., about 0.05% (w/v) to about 0.5% (w/v)), and protamine sulfate at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)). Optionally, the glutamate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0246] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted within this range with MEA or sodium hydroxide.
[0247] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 5% (w/v) to about 10% (w/v), and has a pH of about 5.0 to about 5.5.
[0248] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 5% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0249] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 140 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, dextran at a concentration of about 5% (w/v) to about 15% (w/v), and has a pH of about 5.0 to about 5.5.
[0250] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 140 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, dextran at a concentration of about 5% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0251] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.5% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0252] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.5% (w/v) to about 2% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0253] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), and has a pH of about 5.0 to about 5.5.
[0254] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0255] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 140 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, proline at a concentration of about 200 to about 300 mM, and has a pH of about 5.0 to about 5.5.
[0256] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0257] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MSA at a concentration of about 50 mM to about 150 mM, TEA at a concentration of about 25 mM to about 75 mM, and has a pH of about 5.0 to about 5.5.
[0258] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sodium borate at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0259] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sodium bicarbonate at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0260] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sodium sulfate at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0261] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium sulfate at a concentration of about 10 mM to about 30 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0262] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ammonium sulfate at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0263] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0264] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sodium chloride at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0265] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 200 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, magnesium chloride at a concentration of about 50 mM to about 100 mM, proline at a concentration of about 50 mM to about 150 mM, and has a pH of about 5.0 to about 5.5.
[0266] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MEA at a concentration of about 20 mM to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0267] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MEA at a concentration of about 70 mM to about 90 mM, and has a pH of about 5.0 to about 5.5.
[0268] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MEA at a concentration of about 100 mM to about 130 mM, and has a pH of about 5.0 to about 5.5.
[0269] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 1% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0270] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0271] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), PEG 3350 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0272] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, TEA at a concentration of about 100 mM to about 200 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0273] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, TEA at a concentration of about 20 mM to about 40 mM, calcium chloride at a concentration of about 60 mM to about 90 mM, and has a pH of about 5.0 to about 5.5.
[0274] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), and has a pH of about 5.0 to about 5.5.
[0275] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 4% (w/v), and has a pH of about 5.0 to about 5.5.
[0276] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.1% (w/v) to about 0.4% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 4% (w/v), and has a pH of about 5.0 to about 5.5.
[0277] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), MSA at a concentration of about 80 mM to about 120 mM, and has a pH of about 5.0 to about 5.5.
[0278] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), 10K PVP at a concentration of about 0.3% (w/v) to about 0.8% (w/v), MSA at a concentration of about 80 mM to about 120 mM, and has a pH of about 5.0 to about 5.5.
[0279] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.3% (w/v) to about 0.8% (w/v), MEA at a concentration of about 80 mM to about 120 mM, and has a pH of about 5.0 to about 5.5.
[0280] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, alanine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0281] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, asparagine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0282] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, isoleucine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0283] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, serine at a concentration of about 80 mM to about 120 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0284] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, aspartic acid at a concentration of about 10 mM to about 30 mM, proline at a concentration of about 60 mM to about 100 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0285] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, creatine at a concentration of about 15 mM to about 35 mM, proline at a concentration of about 60 mM to about 90 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0286] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, glutamine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0287] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, leucine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0288] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, phenylalanine at a concentration of about 40 mM to about 60 mM, proline at a concentration of about 40 mM to about 60 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0289] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, tryptophan at a concentration of about 10 mM to about 40 mM, proline at a concentration of about 60 mM to about 90 mM, calcium chloride at a concentration of about 30 mM to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0290] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzalkonium chloride at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0291] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, guanidine HCl at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0292] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, lecithin at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0293] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, oleic acid at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0294] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0295] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, polyvinyl alcohol 205K at a concentration of about 0.05% (w/v) to about 0.5% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0296] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, polyvinyl alcohol 31K at a concentration of about 0.05% (w/v) to about 0.5% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0297] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PVP at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0298] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, protamine sulfate at a concentration of about 0.005% (w/v) to about 0.05% (w/v), proline at a concentration of about 250 mM to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0299] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 5% (w/v) to about 12% (w/v), glycerol at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0300] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 2% (w/v) to about 6% (w/v), glycerol at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0301] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), glycerol at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0302] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 8% (w/v) to about 12% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0303] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PVP at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0304] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0305] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, proline at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0306] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0307] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 1.5% (w/v) to about 4% (w/v), arginine at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0308] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 3% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0309] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 15 hydroxystearate at a concentration of about 3% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, proline at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0310] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, dextran at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0311] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, imidazole at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0312] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, imidazole at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0313] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0314] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0315] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, inositol at a concentration of about 200 to about 300 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0316] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, inositol at a concentration of about 200 to about 300 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0317] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0318] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, taurine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0319] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, citruline at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0320] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, betaine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0321] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, sarcosine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0322] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 1% (w/v) to about 1.5% (w/v), glycolic acid at a concentration of about 40 to about 80 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0323] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, glycolic acid at a concentration of about 40 to about 80 mM, calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0324] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PCA at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0325] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PCA at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0326] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, gelatin at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0327] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, hyaluronic acid at a concentration of about 0.1% (w/v) to about 0.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0328] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, hyaluronic acid at a concentration of about 0.1% (w/v) to about 0.5% (w/v), calcium chloride at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0329] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl proline at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0330] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, N-acetyl ornithine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0331] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ornithine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0332] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, beta-alanine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0333] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0334] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, medronic acid at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0335] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0336] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 200 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), calcium chloride at a concentration of about 30 to about 60 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0337] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0338] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 600 at a concentration of about 1.5% (w/v) to about 4% (w/v), and calcium chloride at a concentration of about 30 to about 60 mM, and has a pH of about 5.0 to about 5.5.
[0339] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MSA at a concentration of about 80 to about 120 mM, TEA at a concentration of about 30 to about 70 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0340] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, MSA at a concentration of about 110 to about 140 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0341] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 250 to about 300 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0342] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0343] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 80 to about 120 mM, calcium chloride at a concentration of about 60 to about 100 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0344] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 80 to about 120 mM, MSA at a concentration of about 60 to about 100 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0345] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, niacinamide at a concentration of about 80 to about 120 mM, PEG 200 at a concentration of about 0.7% (w/v) to about 1.5% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0346] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.25% (w/v) to about 0.75% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0347] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.25% (w/v) to about 0.75% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0348] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.1% (w/v) to about 0.4% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 4% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0349] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.1% (w/v) to about 0.4% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 4% (w/v), MSA at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0350] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.25% (w/v) to about 0.75% (w/v), MSA at a concentration of about 80 to about 120 mM, 10K PVP at a concentration of about 0.25% (w/v) to about 0.75% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0351] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, ethanol at a concentration of about 0.25% (w/v) to about 0.75% (w/v), MEA at a concentration of about 80 to about 120 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0352] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzene sulfonic acid at a concentration of about 100 to about 200 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5. Optionally, the pH of the formulation is adjusted within this range with sodium hydroxide or calcium hydroxide.
[0353] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzene sulfonic acid at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0354] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzene sulfonic acid at a concentration of about 60 to about 90 mM, calcium chloride at a concentration of about 25 to about 75 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0355] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, benzene sulfonic acid at a concentration of about 60 to about 90 mM, MEA at a concentration of about 60 to about 90 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0356] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, adipic acid at a concentration of about 100 to about 200 mM, and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0357] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, glutamate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.25% (w/v) to about 0.75% (w/v), calcium chloride at a concentration of about 40 to about 80 mM, glycerin at a concentration of about 0.5% (w/v) to about 1% (w/v), and optionally Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.4% (w/v) or Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.1% (w/v), and has a pH of about 5.0 to about 5.5.
[0358] Exemplary aqueous adalimumab formulations are provided in Table A. Each formulation in Table A may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table A has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table A is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00001 TABLE A Protein conc. Buffer Excipient(s).sup.1 100 mg/mL 15 mM glutamate 8% PEG 400 1% glycerol 100 mg/mL 15 mM glutamate 4% PEG 200 1% glycerol 100 mg/mL 15 mM glutamate 1% PEG 400 2.5% glycerol 100 mg/mL 15 mM glutamate 10% PEG 400 100 mg/mL 15 mM glutamate 1% PVP 110 mg/mL 20 mM glutamate 45 mM CaCl.sub.2 110 mg/mL 20 mM glutamate 45 mM CaCl.sub.2 100 mM proline 110 mg/mL 20 mM glutamate 2.5% PEG 15 hydroxystearate 110 mg/mL 20 mM glutamate 2.5% PEG 15 hydroxystearate 75 mM arginine 110 mg/mL 20 mM glutamate 5% PEG 15 hydroxystearate 45 mM CaCl.sub.2 110 mg/mL 20 mM glutamate 5% PEG 15 hydroxystearate 45 mM CaCl.sub.2 100 mM proline 110 mg/mL 20 mM glutamate 2% dextran 110 mg/mL 20 mM glutamate 2% imidazole 110 mg/mL 20 mM glutamate 1% imidazole 45 mM CaCl.sub.2 110 mg/mL 20 mM glutamate 120 mM N-acetyl arginine 110 mg/mL 20 mM glutamate 120 mM N-acetyl arginine 45 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 250 mM inositol 105 mg/mL 20 mM glutamic acid 180 mM inositol 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 1% PEG 3350 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 120 mM taurine 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 120 mM citruline 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 120 mM betaine 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 120 mM sarcosine 40 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 1.2% PEG 400 60 mM glycolic acid 105 mg/mL 20 mM glutamic acid 60 mM glycolic acid 75 mM CaCl.sub.2 105 mg/mL 20 mM glutamic acid 1% PCA 105 mg/mL 20 mM glutamic acid 1% PCA 75 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 1% Type B gelatin 75 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 0.25% hyaluronic acid 110 mg/mL 20 mM glutamic acid 0.25% hyaluronic acid 75 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM N-acetyl proline 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM N-acetyl ornithine 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM ornithine 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM beta-alanine 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM niacinamide 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 120 mM medronic acid 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 1% PEG 200 110 mg/mL 20 mM glutamic acid 1% PEG 200 40 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 2% PEG 600 110 mg/mL 20 mM glutamic acid 2% PEG 600 40 mM CaCl2 110 mg/mL 20 mM glutamic acid 100 mM MSA 50 mM TEA 110 mg/mL 20 mM glutamic acid 125 mM MSA 110 mg/mL 20 mM glutamic acid 270 mM niacinamide 110 mg/mL 20 mM glutamic acid 100 mM niacinamide 110 mg/mL 20 mM glutamic acid 100 mM niacinamide 80 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 100 mM niacinamide 80 mM MSA 110 mg/mL 20 mM glutamic acid 100 mM niacinamide 1% PEG 200 110 mg/mL 20 mM glutamic acid 0.5% ethanol 110 mg/mL 20 mM glutamic acid 0.5% ethanol 2% PEG 200 110 mg/mL 20 mM glutamic acid 0.25% ethanol 2% PEG 200 110 mg/mL 20 mM glutamic acid 0.25% ethanol 2% PEG 200 100 mM MSA 110 mg/mL 20 mM glutamic acid 0.5% ethanol 100 mM MSA 0.5% 10K PVP 110 mg/mL 20 mM glutamic acid 0.5% ethanol 100 mM MEA 110 mg/mL 20 mM glutamic acid 150 mM benzene sulfonic acid (adjust pH with NaOH) 110 mg/mL 20 mM glutamic acid 150 mM benzene sulfonic acid (adjust pH with Ca(OH).sub.2) 110 mg/mL 20 mM glutamic acid 75 mM benzene sulfonic acid 110 mg/mL 20 mM glutamic acid 75 mM benzene sulfonic acid 50 mM CaCl.sub.2 110 mg/mL 20 mM glutamic acid 75 mM benzene sulfonic acid 75 mM MEA 110 mg/mL 20 mM glutamic acid 150 mM adipic acid 180 mg/mL 20 mM glutamic acid 0.5% PEG 400 60 mM CaCl.sub.2 0.7% glycerin .sup.1All percentages are (w/v).
[0359] In an embodiment, a lyophilized form of any one of the foregoing glutamate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Adipate Buffer
[0360] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 170 to about 190 mg/mL, about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, adipate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyethylene glycols and polyethylene glycol derivatives, certain polyols, certain acids, certain amines, certain salts, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM or about 50 to about 300 mM) and N-acetyl arginine (e.g., at a concentration of about 0.1 to about 450 mM or about 100 to about 150 mM). Examples of suitable polyethylene glycols and polyethylene glycol derivatives include PEG 400 (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v) or about 0.3% (w/v) to about 1.5% (w/v)) and PEG 3350 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v) or about 0.5% (w/v) to about 2% (w/v)). Examples of suitable polyols include glycerol (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 1% (w/v) to about 2% (w/v)), sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include MSA (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, and/or about 10 to about 40 mM). Examples of suitable amines include monoethanolamine hydrochloride (MEA-HCl) (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 40 mM, or about 50 to about 100 mM) and methanolamine (MEA) (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, or about 50 to about 100 mM). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 20 to about 75 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM) and sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM). Examples of suitable surfactants Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Examples of other suitable excipients include imidazole (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) or about 1% (w/v) to about 1.5% (w/v)) and PVP, for example, 10K PVP, (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v) or about 0.5% (w/v) to about 2% (w/v)). Optionally, the adipate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0361] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 170 to about 190 mg/mL, about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, adipate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 25 mM, about 15 mM to about 20 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of N-acetyl arginine at a concentration of about 0.1 to about 450 mM (e.g., about 100 to about 150 mM), PEG 400 at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 0.3% (w/v) to about 1.5% (w/v)), MSA at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM and/or about 10 to about 40 mM), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 20 to about 75 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), monoethanolamide hydrochloride (MEA-HCl) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM or about 50 to about 100 mM), monoethanolamide (MEA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM or about 50 to about 100 mM), glycerol at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 1% (w/v) to about 2% (w/v)), imidazole at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 1% (w/v) to about 1.5% (w/v)), PVP, for example, 10K PVP, at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), PEG 3350 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the adipate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0362] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, PEG 400 at a concentration of about 0.5% (w/v) to about 1% (w/v), and MSA at a concentration of about 10 to about 30 mM, and has a pH of about 5.0 to about 5.5.
[0363] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, calcium chloride at a concentration of about 30 to about 50 mM, and MSA at a concentration of about 10 to about 30 mM, and has a pH of about 5.0 to about 5.5.
[0364] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, N-acetyl arginine at a concentration of about 100 to about 150 mM, MEA-HCl at a concentration of about 30 to about 70 mM, and MSA at a concentration of about 5 to about 15 mM, and has a pH of about 5.0 to about 5.5.
[0365] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, glycerol at a concentration of about 1% (w/v) to about 2% (w/v), and MSA at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0366] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), and MSA at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0367] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, and imidazole at a concentration of about 1% (w/v) to about 1.5% (w/v), and has a pH of about 5.0 to about 5.5.
[0368] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, calcium chloride at a concentration of about 60 to about 90 mM, PEG 400 at a concentration of about 0.1% (w/v) to about 0.5% (w/v), and 10K PVP at a concentration of about 0.5% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0369] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, MEA-HCl at a concentration of about 80 to about 120 mM, PEG 400 at a concentration of about 0.1% (w/v) to about 0.5% (w/v), and 10K PVP at a concentration of about 0.5% (w/v) to about 2% (w/v), and has a pH of about 5.0 to about 5.5.
[0370] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0371] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 60 to about 90 mM, and has a pH of about 5.0 to about 5.5.
[0372] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), PEG 400 at a concentration of about 1% (w/v) to about 3% (w/v), and calcium chloride at a concentration of about 10 to about 30 mM, and has a pH of about 5.0 to about 5.5.
[0373] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 170 to about 190 mg/mL, adipate at a concentration of about 10 mM to about 25 mM, PEG 3350 at a concentration of about 0.5% (w/v) to about 2% (w/v), PEG 400 at a concentration of about 0.5% (w/v) to about 2% (w/v), and calcium chloride at a concentration of about 30 to about 50 mM, and has a pH of about 5.0 to about 5.5.
[0374] Exemplary aqueous adalimumab formulations are provided in Table B. Each formulation in Table B may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table B has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table B is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00002 TABLE B Protein conc. Buffer Excipient(s) 180 mg/mL 15 mM adipate 120 mM N-acetyl arginine 0.7% PEG 400 20 mM MSA 180 mg/mL 15 mM adipate 120 mM N-acetyl arginine 40 mM CaCl.sub.2 20 mM MSA 180 mg/mL 15 mM adipate 120 mM N-acetyl arginine 50 mM MEA-HCl 10 mM MSA 180 mg/mL 15 mM adipate 1.4% glycerol 40 mM MSA 180 mg/mL 15 mM adipate 1% PEG 400 40 mM MSA 180 mg/mL 15 mM adipate 1.2% imidazole 180 mg/mL 20 mM adipate 75 mM CaCl.sub.2 0.3% PEG 400 1% PVP 10K 180 mg/mL 20 mM adipate 100 mM MEA-HCl 0.3% PEG 400 1% PVP 10K 180 mg/mL 20 mM adipate 1% PEG 400 40 mM CaCl.sub.2 180 mg/mL 20 mM adipate 1% PEG 3350 75 mM CaCl.sub.2 180 mg/mL 20 mM adipate 1% PEG 3350 1.5% PEG 400 20 mM CaCl2 180 mg/mL 20 mM adipate 1% PEG 3350 1% PEG 400 40 mM CaCl.sub.2
[0375] In an embodiment, a lyophilized form of any one of the foregoing adipate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Glucuronate Buffer
[0376] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 170 to about 190 mg/mL, about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glucuronate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyols, certain acids, certain amines, certain salts, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 250 to about 350 mM, or about 50 to about 300 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 50 mM). Examples of suitable amines include monoethanolamide hydrochloride (MEA-HCl) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM) and monoethanolamide (MEA) (e.g., at a concentration of about 0.1 to about 300 mM or about 0.1 to about 50 mM). Examples of suitable salts include sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM) and calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the glucuronate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0377] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 170 to about 190 mg/mL, about 90 to about 120 mg/mL, about 160 to about 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glucuronate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 250 to about 350 mM or about 50 to about 300 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), or about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM), monoethanolamide hydrochloride (MEA-HCl) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), monoethanolamide (MEA) at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the glucuronate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0378] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glucuronate at a concentration of about 10 mM to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.0 to about 5.5.
[0379] Exemplary aqueous adalimumab formulations are provided in Table C. Each formulation in Table C may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table C has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table C is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00003 TABLE C Protein conc. Buffer Excipient(s) 105 mg/mL 20 mM MEA glucuronate 300 mM proline 105 mg/mL 20 mM Na glucuronate 300 mM proline
[0380] In an embodiment, a lyophilized form of any one of the foregoing glucuronate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Acetate Buffer
[0381] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 90 to about 110 mg/mL, about 100 to about 110 mg/mL, about 120 to about 160 mg/mL, about 130 to about 150 mg/mL, about 160 to about 190 mg/mL, about 160 to about 180 mg/mL, about 170 to about 180 mg/mL, about 40 to about 60 mg/mL, about 40 to about 50 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, acetic acid and/or acetate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 5 mM to about 50 mM, about 5 mM to about 15 mM, about 10 mM to about 20 mM, about 10 mM to about 30 mM, about 15 mM to about 25 mM, about 30 mM to about 40 mM, about 35 mM to about 45 mM, about 40 mM to about 50 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyols, certain acids, certain amines, certain salts, certain polysaccharides or polysaccharide derivatives, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include arginine-HCl (e.g., at a concentration of about 0.1 to about 450 mM, about 20 to about 200 mM, about 50 to about 150 mM, about 80 to about 120 mM, and/or about 100 mM) and proline (e.g., at a concentration of about 0.1 to about 450 mM, about 20 to about 400 mM, about 50 to about 350 mM, about 50 to about 300 mM, about 80 to about 300 mM, about 100 to about 250 mM, about 150 to about 230 mM, about 100 to about 140 mM, about 130 to about 170 mM, about 160 to about 200 mM, about 190 to about 230 mM, about 220 to about 260 mM, about 250 to about 290 mM and/or about 220 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v) about 4% (w/v) to about 12% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 5% (w/v) to about 10% (w/v), about 6% (w/v) to about 8% (w/v), about 8% (w/v) to about 10% (w/v), about 8.5% (w/v) to about 9.5% (w/v), about 9% (w/v), about 5.8% (w/v) to about 6.6% (w/v), about 6% (w/v) to about 6.4% (w/v), about 6.2% (w/v), about 6.1% (w/v) to about 6.9% (w/v), about 6.3% (w/v) to about 6.7% (w/v), about 6.5% (w/v), about 6.4% (w/v) to about 7.2% (w/v), about 6.6% (w/v) to about 7% (w/v), about 6.8% (w/v), about 6.5% (w/v) to about 7.3% (w/v), about 6.7% (w/v) to about 7.1% (w/v), about 6.9% (w/v), about 6.6% (w/v) to about 7.4% (w/v), about 6.8% (w/v) to about 7.2% (w/v), about 7% (w/v), about 6.9% (w/v) to about 7.7% (w/v), about 7.1% (w/v) to about 7.5% (w/v), and/or about 7.3% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include MSA (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM). Examples of suitable amines include MEA-HCl (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 40 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM) and MEA (e.g., at a concentration of about 0.1 to about 300 mM, about 0.1 to about 50 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 5 to about 15 mM, about 10 to about 40 mM, about 10 to about 30 mM, about 15 to about 35 mM, about 20 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 50 mM, about 40 to about 60 mM, about 40 to about 80 mM, about 50 to about 100 mM, about 10 mM, about 25 mM, and/or about 45 mM) and sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM). Examples of suitable polysaccharides or polysaccharide derivatives include sodium carboxymethylcellulose (NaCMC) (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 0.1% (w/v) to about 5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.1% (w/v) to about 1% (w/v), and/or about 0.1% (w/v) to about 0.5% (w/v)). Examples of suitable surfactants include guanidine hydrochloride (GnHCl) (e.g., at a concentration of about 0.1 to about 150 mM, 5 to about 50 mM, about 10 to about 40 mM, about 15 to about 30 mM, and/or 20 mM), Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.2% (w/v) to about 1% (w/v), about 0.01% (w/v), about 0.05% (w/v), about 0.1% (w/v), and/or about 0.4% (w/v)), Docusate sodium (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.005% (w/v) to about 0.05% (w/v)), benzalkonium chloride (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.05% (w/v) to about 0.5% (w/v)), Span 40 (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v) or about 0.05% (w/v) to about 0.5% (w/v)), Triton X-100 (e.g., at a concentration of about 0.001% (w/v) to about 1% (w/v), about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.2% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the acetate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0382] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 90 to about 110 mg/mL, about 100 to about 110 mg/mL, about 120 to about 160 mg/mL, about 130 to about 150 mg/mL, about 160 to about 190 mg/mL, about 160 to about 180 mg/mL, about 170 to about 180 mg/mL, about 40 to about 60 mg/mL, about 40 to about 50 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, acetic acid and/or acetate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 5 mM to about 50 mM, about 5 mM to about 15 mM, about 10 mM to about 20 mM, about 10 mM to about 30 mM, about 15 mM to about 25 mM, about 30 mM to about 40 mM, about 35 mM to about 45 mM, about 40 mM to about 50 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 12% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 5% (w/v) to about 10% (w/v), about 6% (w/v) to about 8% (w/v), about 8% (w/v) to about 10% (w/v), about 8.5% (w/v) to about 9.5% (w/v), about 9% (w/v), about 5.8% (w/v) to about 6.6% (w/v), about 6% (w/v) to about 6.4% (w/v), about 6.2% (w/v), about 6.1% (w/v) to about 6.9% (w/v), about 6.3% (w/v) to about 6.7% (w/v), about 6.5% (w/v), about 6.4% (w/v) to about 7.2% (w/v), about 6.6% (w/v) to about 7% (w/v), about 6.8% (w/v), about 6.5% (w/v) to about 7.3% (w/v), about 6.7% (w/v) to about 7.1% (w/v), about 6.9% (w/v), about 6.6% (w/v) to about 7.4% (w/v), about 6.8% (w/v) to about 7.2% (w/v), about 7% (w/v), about 6.9% (w/v) to about 7.7% (w/v), about 7.1% (w/v) to about 7.5% (w/v), and/or about 7.3% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 5 to about 15 mM, about 10 to about 40 mM, about 10 to about 30 mM, about 15 to about 35 mM, about 20 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 50 mM, about 40 to about 60 mM, about 40 to about 80 mM, about 50 to about 100 mM, about 10 mM, about 25 mM, and/or about 45 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), MSA at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM), MEA-HCl at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM), MEA at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM, about 10 to about 50 mM, about 20 to about 40 mM, about 30 mM, about 30 to about 50 mM, about 40 to about 60 mM, about 50 to about 70 mM, about 60 to about 80 mM, about 70 to about 90 mM, and/or about 80 to about 100 mM), guanidine hydrochloride (GnHCl) at a concentration of about 0.1 to about 150 mM (e.g., 5 to about 50 mM, about 10 to about 40 mM, about 15 to about 30 mM, and/or 20 mM), sodium carboxymethylcellulose (NaCMC) at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 0.1% (w/v) to about 5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.1% (w/v) to about 1% (w/v), and/or about 0.1% (w/v) to about 0.5% (w/v)), arginine-HCl at a concentration of about 0.1 to about 450 mM (e.g., about 20 to about 200 mM, about 50 to about 150 mM, about 80 to about 120 mM, and/or about 100 mM), proline at a concentration of about 0.1 to about 450 mM (e.g., about 20 to about 400 mM, about 50 to about 350 mM, about 50 to about 300 mM, about 80 to about 300 mM, about 100 to about 250 mM, about 150 to about 230 mM, about 100 to about 140 mM, about 130 to about 170 mM, about 160 to about 200 mM, about 190 to about 230 mM, about 220 to about 260 mM, about 250 to about 290 mM and/or about 220 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.2% (w/v) to about 1% (w/v), about 0.01% (w/v), about 0.05% (w/v), about 0.1% (w/v), and/or about 0.4% (w/v)), Docusate sodium at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.005% (w/v) to about 0.05% (w/v)), benzalkonium chloride at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.05% (w/v) to about 0.5% (w/v)), Span 40 at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.05% (w/v) to about 0.5% (w/v)), Triton X-100 at a concentration of about 0.001% (w/v) to about 1% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.2% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the acetate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0383] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0384] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0385] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0386] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Docusate sodium at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0387] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and benzalkonium chloride at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0388] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Span 40 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0389] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0390] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, and arginine-HCl at a concentration of about 80 to about 120 mM, and has a pH of about 5.1 to 5.3.
[0391] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0392] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0393] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 80 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0394] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0395] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0396] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Polysorbate 20 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0397] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.0025% (w/v) to about 0.025% (w/v), and has a pH of about 5.1 to 5.3.
[0398] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3.
[0399] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Triton X-100 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0400] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.025% (w/v) to about 0.25% (w/v), and has a pH of about 5.1 to 5.3.
[0401] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0402] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 120 to about 160 mg/mL, acetate at a concentration of about 15 mM to about 25 mM, calcium chloride at a concentration of about 30 to about 60 mM, arginine-HCl at a concentration of about 80 to about 120 mM, and Pluronic F68 at a concentration of about 0.2% (w/v) to about 0.6% (w/v), and has a pH of about 5.1 to 5.3.
[0403] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 150 to about 180 mg/mL, acetate at a concentration of about 5 mM to about 10 mM, sucrose at a concentration of about 7% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0404] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.1% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0405] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.0% (w/v) to about 6.5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0406] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.6% (w/v) to about 7.2% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0407] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.7% (w/v) to about 7.3% (w/v), calcium chloride at a concentration of about 5 to about 15 mM, guanidine hydrochloride (GnHCl) at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0408] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 50 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.7% (w/v) to about 7.3% (w/v), calcium chloride at a concentration of about 5 to about 15 mM, NaCMC at a concentration of about 0.2% (w/v) to about 1% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0409] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, and sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl and/or NaOH.
[0410] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0411] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8.5% (w/v) to about 9.5% (w/v), and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0412] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0413] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8.5% (w/v) to about 9.5% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0414] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0415] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0416] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0417] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0418] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0419] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0420] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8.5% (w/v) to about 9.5% (w/v), and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0421] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0422] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0423] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0424] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0425] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0426] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0427] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0428] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0429] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0430] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0431] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0432] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0433] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8.5% (w/v) to about 9.5% (w/v), and Polysorbate 80 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0434] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0435] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.6% (w/v) to about 7% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Polysorbate 80 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0436] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0437] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0438] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0439] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0440] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.7% (w/v) to about 7.1% (w/v), sodium chloride at a concentration of about 25 to about 40 mM, and Polysorbate 80 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0441] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0442] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0443] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0444] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0445] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0446] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0447] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0448] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.4% (w/v) to about 7.2% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0449] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0450] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), MEA-HCl at a concentration of about 20 to about 40 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0451] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), MSA at a concentration of about 20 to about 40 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH, Ca(OH).sub.2, or MEA.
[0452] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, acetate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 6.5% (w/v) to about 7.3% (w/v), sodium chloride at a concentration of about 20 to about 45 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0453] Exemplary aqueous adalimumab formulations are provided in Table D. Each formulation in Table D may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.2% (w/v) to about 1% (w/v), about 0.01% (w/v), about 0.05% (w/v), about 0.1% (w/v), and/or about 0.4% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.2% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table D has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table D is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00004 TABLE D Protein conc. Buffer Excipient(s) 50 mg/mL 10 mM sodium acetate 6.8% (w/v) sucrose 25 mM CaCl.sub.2 50 mg/mL 10 mM calcium acetate 6.2% (w/v) sucrose 25 mM CaCl.sub.2 50 mg/mL 10 mM sodium acetate 6.9% (w/v) sucrose 30 mM MEA-HCl 50 mg/mL 10 mM sodium acetate 7% (w/v) sucrose 10 mM CaCl.sub.2 20 mM GnHCl 50 mg/mL 10 mM sodium acetate 7% (w/v) sucrose 10 mM CaCl.sub.2 0.5% NaCMC 50 mg/mL 10 mM acetate 9% (w/v) sucrose 100 mg/mL 10 mM acetate 9% (w/v) sucrose 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 25 mM CaCl.sub.2 100 mg/mL 10 mM acetate 220 mM proline 25 mM CaCl.sub.2 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 30 mM MEA-HCl 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 30 mM MSA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 35 mM NaCl
[0454] Exemplary aqueous adalimumab formulations are provided in Table E. Each formulation in Table E has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table E is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00005 TABLE E Protein conc. Buffer Excipient(s) Surfactant 50 mg/mL 10 mM sodium 6.8% (w/v) sucrose 0.1% (w/v) acetate 25 mM CaCl.sub.2 Pluronic F68 50 mg/mL 10 mM calcium 6.2% (w/v) sucrose 0.1% (w/v) acetate 25 mM CaCl.sub.2 Pluronic F68 50 mg/mL 10 mM sodium 6.9% (w/v) sucrose 0.1% (w/v) acetate 30 mM MEA-HCl Pluronic F68 50 mg/mL 10 mM sodium 7% (w/v) sucrose 0.1% (w/v) acetate 10 mM CaCl.sub.2 Pluronic F68 20 mM GnHCl 50 mg/mL 10 mM sodium 7% (w/v) sucrose 0.1% (w/v) acetate 10 mM CaCl.sub.2 Pluronic F68 0.5% NaCMC 50 mg/mL 10 mM acetate 9% (w/v) sucrose 0.1% (w/v) Polysorbate 80 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.1% (w/v) Pluronic F68 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.1% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM acetate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 30 mM MEA-HCl Pluronic F68 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.1% (w/v) 30 mM MSA Pluronic F68 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 35 mM NaCl Pluronic F68 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.05% (w/v) Pluronic F68 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.05% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM acetate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 30 mM MEA-HCl Pluronic F68 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.05% (w/v) 30 mM MSA Pluronic F68 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 35 mM NaCl Pluronic F68 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.1% (w/v) Polysorbate 80 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM acetate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 30 mM MEA-HCl Polysorbate 80 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.1% (w/v) 30 mM MSA Polysorbate 80 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 35 mM NaCl Polysorbate 80 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.05% (w/v) Polysorbate 80 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM acetate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 30 mM MEA-HCl Polysorbate 80 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.05% (w/v) 30 mM MSA Polysorbate 80 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 35 mM NaCl Polysorbate 80 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.1% (w/v) Polysorbate 20 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM acetate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 30 mM MEA-HCl Polysorbate 20 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.1% (w/v) 30 mM MSA Polysorbate 20 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.1% (w/v) 35 mM NaCl Polysorbate 20 100 mg/mL 10 mM acetate 9% (w/v) sucrose 0.05% (w/v) Polysorbate 20 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM acetate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 30 mM MEA-HCl Polysorbate 20 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose 0.05% (w/v) 30 mM MSA Polysorbate 20 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose 0.05% (w/v) 35 mM NaCl Polysorbate 20
[0455] Exemplary aqueous adalimumab formulations are provided in Table F.
TABLE-US-00006 TABLE F Protein pH Adjusting conc. Buffer Excipient(s) Agent Surfactant pH 50 mg/mL 10 mM sodium 6.8% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 acetate 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 50 mg/mL 10 mM calcium 6.2% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 acetate 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 50 mg/mL 10 mM sodium 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 acetate 30 mM MEA-HCl HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 50 mg/mL 10 mM sodium 7% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 acetate 10 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 20 mM GnHCl HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 50 mg/mL 10 mM sodium 7% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 acetate 10 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 0.5% NaCMC HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 50 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.1% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Pluronic F68 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.05% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Pluronic F68 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.1% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 80 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.05% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 80 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.04% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.04% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.04% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.04% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.04% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 80 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.04% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.03% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.03% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.03% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.03% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.03% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 80 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.03% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.1% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 20 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.8% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 30 mM MEA-HCl HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or
MSA/MEA 100 mg/mL 10 mM acetate 7.3% (w/v) sucrose NaOH, 0.05% (w/v) 5.2 30 mM MSA Ca(OH).sub.2, or Polysorbate 20 MEA 100 mg/mL 10 mM acetate 6.9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 35 mM NaCl HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA
[0456] Exemplary aqueous adalimumab formulations are also provided in Tables 8, 9, 12, 14 and 15. In an embodiment, the stable aqueous adalimumab formulation is one of the acetate formulations described in Tables D, E, F, 8, 9, 12, 14 or 15 provided herein.
[0457] In an embodiment, a lyophilized form of any one of the foregoing acetate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Benzoate Buffer
[0458] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 170 to about 190, about 160 to about 190 mg/mL, about 60 to about 90 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, benzoate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyols, certain acids, certain amines, certain salts, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 50 to about 300 mM, or about 250 mM to about 350 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 50 mM). Examples of suitable amines include MEA-HCl (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM) and MEA (e.g., at a concentration of about 0.1 to about 300 mM or about 0.1 to about 50 mM). Examples of suitable salts include sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM) and calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the benzoate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0459] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 170 to about 190, about 160 to about 190 mg/mL, about 60 to about 90 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, benzoate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM or about 250 mM to about 350 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM), MEA-HCl at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), MEA at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.01% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the benzoate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0460] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 60 to about 90 mg/mL, benzoate at a concentration of about 10 mM to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA.
[0461] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, benzoate at a concentration of about 10 mM to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with NaOH.
[0462] In an embodiment, a lyophilized form of any one of the foregoing benzoate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Glycolate Buffer
[0463] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 170 to about 190, about 160 to about 190 mg/mL, about 60 to about 90 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glycolate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyols, certain acids, certain amines, certain salts, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 250 mM to about 350 mM, or about 50 to about 300 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 50 mM). Examples of suitable amines include MEA-HCl (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM) and MEA (e.g., at a concentration of about 0.1 to about 300 mM or about 0.1 to about 50 mM). Examples of suitable salts include sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM) and calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the glycolate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0464] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 170 to about 190, about 160 to about 190 mg/mL, about 60 to about 90 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, glycolate at a concentration of about 5 mM to about 50 mM (e.g., about 10 mM to about 30 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 250 mM to about 350 mM or about 50 to about 300 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM), MEA-HCl at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), MEA at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the glycolate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0465] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, glycolate at a concentration of about 10 mM to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA or NaOH.
[0466] In an embodiment, a lyophilized form of any one of the foregoing glycolate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Lactate Buffer
[0467] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, lactic acid and/or lactate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 10 mM to about 30 mM, about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyethylene glycols and polyethylene glycol derivatives, certain salts, certain polyols, certain acids, certain amines, and certain surfactants. Examples of suitable amino acids include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 50 to about 300 mM, about 110 to about 300 mM, about 250 to about 350 mM, about 190 to about 250 mM, and/or about 220 mM). Examples of suitable polyethylene glycols include PEG 600 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v) or about 4% (w/v) to about 13% (w/v)), PEG 400 (e.g., at a concentration of about 0.1% (w/v) to about 20% (w/v) or about 0.5% (w/v) to about 2% (w/v)), and PEG 200 (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v) or about 1.6% (w/v) to about 3.8% (w/v)). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM) and sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, or about 35 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 50 mM). Examples of suitable amines include monoethanolamide hydrochloride (MEA-HCl) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM) and monoethanolamide (MEA) (e.g., at a concentration of about 0.1 to about 300 mM or about 0.1 to about 50 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the lactate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0468] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, or about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, lactic acid and/or lactate at a concentration of about 0.1 mM to about 300 mM (e.g., about 2 mM to about 30 mM, about 10 mM to about 30 mM, about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM, about 110 to about 300 mM, about 250 to about 350 mM, about 190 to about 250 mM, and/or about 220 mM), PEG 600 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 4% (w/v) to about 13% (w/v)), PEG 400 at a concentration of about 0.1% (w/v) to about 20% (w/v) (e.g., about 0.5% (w/v) to about 2% (w/v)), PEG 200 at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 1.6% (w/v) to about 3.8% (w/v)), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM), monoethanolamide hydrochloride (MEA-HCl) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), monoethanolamide (MEA) at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the lactate-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0469] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 10 mM to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA or NaOH.
[0470] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0471] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 3% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.2% (w/v) to about 2% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0472] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0473] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 3% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.2% (w/v) to about 2% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0474] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 200 at a concentration of about 3.5% (w/v) to about 4.2% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0475] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3.
[0476] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 190 to about 240 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0477] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 200 to about 250 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0478] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0479] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0480] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0481] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 3% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.2% (w/v) to about 2% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0482] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 200 at a concentration of about 3.5% (w/v) to about 4.2% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0483] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, PEG 600 at a concentration of about 5% (w/v) to about 5.7% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 90 to about 130 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0484] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MSA and/or MEA.
[0485] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0486] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0487] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0488] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0489] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8.5% (w/v) to about 9.5% (w/v), and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0490] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 15 mM to about 35 mM, and Pluronic F68 at a concentration of about 0.005% (w/v) to about 0.05% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0491] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 15 mM to about 35 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0492] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0493] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0494] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0495] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0496] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0497] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, calcium chloride at a concentration of about 15 to about 35 mM, proline at a concentration of about 190 to about 250 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0498] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0499] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, lactate at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 8% (w/v) to about 10% (w/v), and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, Ca(OH).sub.2, MEA, and/or MSA.
[0500] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 mg/ml to about 200 mg/ml, or about 160 mg/ml to about 190 mg/mL, or about 80 mg/ml to about 120 mg/ml, or about 90 mg/ml to about 110 mg/ml, or about 95 mg/ml to about 105 mg/ml, or about 40 mg/ml, or about 45 mg/ml, or about 50 mg/ml, or about 55 mg/ml, or about 60 mg/ml, or about 65 mg/ml, or about 70 mg/ml, or about 75 mg/ml, or about 80 mg/ml, or about 85 mg/ml, or about 90 mg/ml, or about 95 mg/ml, or about 100 mg/ml, or about 105 mg/ml, or about 110 mg/ml, or about 115 mg/ml, or about 120 mg/ml, or about 125 mg/ml, or about 130 mg/ml, or about 135 mg/ml, or about 140 mg/ml, or about 145 mg/ml, or about 150 mg/ml, or about 155 mg/ml, or about 160 mg/ml, or about 165 mg/ml, or about 170 mg/ml, or about 175 mg/ml, or about 180 mg/ml, or about 185 mg/ml, or about 190 mg/mL, with the following excipients:
(a) lactate buffer at a concentration of about 5 mM to about 15 mM, or about 7 mM to about 12 mM, or about 9 mM to about 11 mM, or about 5 mM, or about 6 mM, or about 7 mM, or about 8 mM, or about 8 mM, or about 9 mM, or about 10 mM, or about 11 mM, or about 12 mM, or about 13 mM, or about 14 mM, or about 15 mM; (b) calcium chloride at a concentration of about 5 to about 30 mM, about 10 to about 20 mM, or about 12.5 mM to about 17.5 mM, or about 14 mM to about 16 mM, or about 10 mM, or about 10.5 mM, or about 11 mM, or about 11.5 mM, or about 12 mM, or about 12.5 mM, or about 13 mM, or about 13.5 mM, or about 14 mM, or about 14.5 mM, or about 15 mM, or about 15.5 mM, or about 16 mM, or about 16.5 mM, or about 17 mM, or about 17.5 mM, or about 18 mM, or about 18.5 mM, or about 19 mM, or about 19.5 mM, or about 20 mM; (c) sucrose at a concentration of about 4% (w/v) to about 10% (w/v), or about 6% (w/v) to about 8.5% (w/v), or about 4% (w/v), or about 4.5% (w/v), or about 5% (w/v), or about 5.5% (w/v), or about 6% (w/v), or about 6.5% (w/v), or about 7% (w/v), or about 7.1% (w/v), or about 7.2% (w/v), or about 7.3% (w/v), or about 7.4% (w/v), or about 7.5% (w/v), or about 7.6% (w/v), or about 7.7% (w/v), or about 7.8% (w/v), or about 8% (w/v), or about 8.5% (w/v); (d) Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.09% (w/v), or about 0.03% (w/v) to about 0.06% (w/v), about 0.01% (w/v) to about 0.2% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), or about 0.05% (w/v) to about 0.07% (w/v), or about 0.03% (w/v), or about 0.04% (w/v), or about 0.05% (w/v), or about 0.06% (w/v), or about 0.07% (w/v), or about 0.08% (w/v), about 0.09% (w/v), or about 0.1% (w/v); and (e) has a pH of about 3.5 to about 8, or about 4 to about 7, or about 4.5 to about 6, or about 5 to about 5.5, or about 3.5, or about 4, or about 4.5, or about 4.6, or about 4.7, or about 4.8, or about 4.9, or about 5.0, or about 5.1, or about 5.2, or about 5.3, or about 5.4, or about 5.5, or about 5.6, or about 5.7, or about 5.8, or about 5.9, or about 6.0, or about 6.5, or about 7.0, or about 7.5, or about 8.0.
[0501] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration or about 40 mg/ml to 200 mg/ml, lactate buffer at a concentration of about 5 mM to about 15 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 5 to about 30 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 3.5 to 8.
[0502] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration or about 40 mg/ml to 200 mg/ml, lactate buffer at a concentration of about 1 mM to about 15 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 10 to about 20 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 3.5 to 8.
[0503] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 80 mg/ml to about 120 mg/mL, lactate buffer at a concentration of about 7 mM to about 12 mM, sucrose at a concentration of about 4% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 12.5 to about 17.5 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.07% (w/v), and a pH of about 4 to about 7.
[0504] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 mg/ml to about 110 mg/mL, lactate buffer at a concentration of about 9 mM to about 11 mM, sucrose at a concentration of about 6% (w/v) to about 8.5% (w/v), calcium chloride at a concentration of about 14 to about 16 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.07% (w/v), and a pH of about 5.0 to about 5.5.
[0505] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 100 mg/mL, lactate buffer at a concentration of about 10 mM, sucrose at a concentration of about 7.4% (w/v), calcium chloride at a concentration of about 15 mM, and Pluronic F68 at a concentration of about 0.06% (w/v), and a pH of about 5.2.
[0506] Exemplary aqueous adalimumab formulations are provided in Table G. Each formulation in Table G may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.01% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.2% (w/v) to about 1% (w/v), about 0.05% (w/v), about 0.1% (w/v), and/or about 0.4% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.2% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table G has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table G is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00007 TABLE G Protein conc. Buffer Excipient(s) 100 mg/mL 10 mM lactate 220 mM proline 25 mM CaCl.sub.2 100 mg/mL 10 mM lactate 9% (w/v) sucrose 100 mg/mL 10 mM lactate 9% (w/v) sucrose 25 mM CaCl.sub.2
[0507] Exemplary aqueous adalimumab formulations are provided in Table H. Each formulation in Table H has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table H is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00008 TABLE H Protein conc. Buffer Excipient(s) Surfactant 100 mg/mL 10 mM lactate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.1% (w/v) Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.1% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.05% (w/v) Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.05% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 220 mM proline 0.01% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.01% (w/v) Pluronic F68 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.01% (w/v) 25 mM CaCl.sub.2 Pluronic F68 100 mg/mL 10 mM lactate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.1% (w/v) Polysorbate 80 100 mg/mL 10 mM lactate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 80 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.05% (w/v) Polysorbate 80 100 mg/mL 10 mM lactate 220 mM proline 0.1% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.1% (w/v) Polysorbate 20 100 mg/mL 10 mM lactate 220 mM proline 0.05% (w/v) 25 mM CaCl.sub.2 Polysorbate 20 100 mg/mL 10 mM lactate 9% (w/v) sucrose 0.05% (w/v) Polysorbate 20
[0508] Exemplary aqueous adalimumab formulations are provided in Table I.
TABLE-US-00009 TABLE I Protein pH Adjusting conc. Buffer Excipient(s) Agent Surfactant pH 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.01% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.01% (w/v) 5.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.01% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Pluronic F68 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 80 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.1% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.1% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 220 mM proline HCl/NaOH, 0.05% (w/v) 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 10 mM lactate 9% (w/v) sucrose HCl/NaOH, 0.05% (w/v) 5.2 HCl/Ca(OH).sub.2, Polysorbate 20 HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA
[0509] Exemplary aqueous adalimumab lactate formulations are also provided in Tables 1, 11, 12, 13, 15, 16 and 17. In an embodiment, the stable aqueous adalimumab formulation is one of the lactate buffer formulations described in Tables G, H, I, 1, 11, 12, 13, 15, 16 or 17 provided herein.
[0510] In an embodiment, a lyophilized form of any one of the foregoing lactate-containing adalimumab formulations is provided.
Formulations of Adalimumab with Histidine Buffer
[0511] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, histidine at a concentration of about 5 mM to about 50 mM (e.g., about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain salts, certain polyols, certain acids, certain amines, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 50 to about 300 mM, or about 270 to about 370 mM). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM) and sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM). Examples of suitable polyols include sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)) and sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)). Examples of suitable acids include methane sulfonic acid (MSA) (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 50 mM). Examples of suitable amines include MEA-HCl (e.g., at a concentration of about 0.1 to about 150 mM or about 0.1 to about 40 mM) and MEA (e.g., at a concentration of about 0.1 to about 300 mM or about 0.1 to about 50 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the histidine-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, about 6.4 to about 7.2, about 6.5 to about 7.1, about 6.6 to about 7.0, about 6.7 to about 6.9, about 6.7 to about 6.8, about 6.8 to about 6.9, about 5.2, and/or about 6.8. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0512] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, histidine at a concentration of about 5 mM to about 50 mM (e.g., about 5 mM to about 15 mM, about 10 mM, about 15 mM, about 20 mM, and/or about 25 mM), and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM or about 270 to about 370 mM), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 50 to about 100 mM, or about 25 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v) and/or about 4% (w/v)), methane sulfonic acid (MSA) at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM), MEA-HCl at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM), MEA at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), about 0.04% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the histidine-containing stable aqueous adalimumab formulation has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, about 6.4 to about 7.2, about 6.5 to about 7.1, about 6.6 to about 7.0, about 6.7 to about 6.9, about 6.7 to about 6.8, about 6.8 to about 6.9, about 5.2, and/or about 6.8. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0513] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, histidine at a concentration of about 5 mM to about 15 mM, proline at a concentration of about 270 to about 370 mM, and calcium chloride at a concentration of about 10 to about 30 mM, and has a pH of about 6.7 to 6.9. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0514] Exemplary aqueous adalimumab formulations are provided in Table J.
TABLE-US-00010 TABLE J Protein pH Adjusting conc. Buffer Excipient(s) Agent pH 100 mg/mL 10 mM histidine 320 mM proline HCl/NaOH, 5.2 20 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA
[0515] In an embodiment, a lyophilized form of any one of the foregoing histidine-containing adalimumab formulations is provided.
Formulations of Adalimumab without Buffer
[0516] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, and one or more excipients. Suitable excipients include certain stabilizers such as certain amino acids and amino acid derivatives, certain polyethylene glycols and polyethylene glycol derivatives, certain salts, certain polyols, certain acids, certain amines, and certain surfactants. Examples of suitable amino acids and amino acid derivatives include proline (e.g., at a concentration of about 0.1 to about 450 mM, about 50 to about 300 mM, or about 60 to about 300 mM). Examples of suitable polyethylene glycols and polyethylene glycol derivatives include PEG 600 (e.g., at a concentration of about 0.1% (w/v) to about 30% (w/v) or about 1.2% (w/v) to about 14.5% (w/v)) and PEG 200 (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v) or about 0.6% (w/v) to about 4.8% (w/v)). Examples of suitable salts include calcium chloride (e.g., at a concentration of about 1 to about 150 mM, about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 20 to about 100 mM, about 50 to about 100 mM, or about 25 mM) and sodium chloride (e.g., at a concentration of about 10 to about 100 mM, about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM). Examples of suitable polyols include sorbitol (e.g., at a concentration of about 0.1% (w/v) to about 10% (w/v), about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)) and sucrose (e.g., at a concentration of about 0.1% (w/v) to about 15% (w/v), about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v) to about 7.3% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)). Examples of suitable acids include MSA (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 50 mM, or about 20 to about 90 mM). Examples of suitable amines include MEA-HCl (e.g., at a concentration of about 0.1 to about 150 mM, about 0.1 to about 40 mM, or about 60 to about 90 mM) and MEA (e.g., at a concentration of about 0.1 to about 300 mM, about 0.1 to about 50 mM, or about 60 to about 90 mM). Examples of suitable surfactants include Pluronic F68 (e.g., at a concentration of about 0.001% (w/v) to about 10% (w/v), about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v) or about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 (e.g., at a concentration of about 0.001% (w/v) to about 2% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the stable aqueous adalimumab formulation without buffer has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0517] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 200 mg/mL, such as about 90 to about 120 mg/mL, about 160 to 190 mg/mL, about 40 to about 60 mg/mL, about 50 mg/mL, about 100 mg/mL, or about 170 mg/mL, and one or more excipients selected from the group consisting of proline at a concentration of about 0.1 to about 450 mM (e.g., about 50 to about 300 mM or about 60 to about 300 mM), PEG 600 at a concentration of about 0.1% (w/v) to about 30% (w/v) (e.g., about 1.2% (w/v) to about 14.5% (w/v)), PEG 200 at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 0.6% (w/v) to about 4.8% (w/v)), calcium chloride at a concentration of about 1 to about 150 mM (e.g., about 5 to about 50 mM, about 10 to about 30 mM, about 20 to about 40 mM, about 30 to about 50 mM, about 40 to about 80 mM, about 20 to about 100 mM, about 50 to about 100 mM, or about 25 mM), sodium chloride at a concentration of about 10 to about 100 mM (e.g., about 60 to about 90 mM, about 15 to about 50 mM, and/or about 35 mM), MSA at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 50 mM or about 20 to about 90 mM), MEA-HCl at a concentration of about 0.1 to about 150 mM (e.g., about 0.1 to about 40 mM or about 60 to about 90 mM), MEA at a concentration of about 0.1 to about 300 mM (e.g., about 0.1 to about 50 mM or about 60 to about 90 mM), sorbitol at a concentration of about 0.1% (w/v) to about 10% (w/v) (e.g., about 3% (w/v) to about 5% (w/v), and/or about 4% (w/v)), sucrose at a concentration of about 0.1% (w/v) to about 15% (w/v) (e.g., about 4% (w/v) to about 10% (w/v), about 4% (w/v) to about 9% (w/v), about 6.5% (w/v) to about 7.3% (w/v), about 6.5% (w/v), about 6.8% (w/v), about 6.9% (w/v), about 9% (w/v)), Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.01% (w/v), about 0.05% (w/v), and/or about 0.1% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v)), and Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Optionally, the stable aqueous adalimumab formulation without buffer has a pH of about 4.8 to about 5.7, for example, about 4.9 to about 5.6, about 5.0 to about 5.5, about 5.1 to about 5.4, about 5.1 to about 5.3, about 5.1 to about 5.2, about 5.2 to about 5.3, and/or about 5.2. Optionally, the pH of the stable aqueous adalimumab formulation is adjusted using a strong acid and/or a strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
[0518] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, MSA at a concentration of about 10 to about 30 mM, and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA or NaOH.
[0519] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 8% (w/v) to about 10% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0520] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 6.5% (w/v) to about 7.3% (w/v), PEG 200 at a concentration of about 0.3% (w/v) to about 1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0521] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 4% (w/v) to about 5% (w/v), PEG 200 at a concentration of about 1.5% (w/v) to about 2.1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0522] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 0.9% (w/v) to about 1.5% (w/v), PEG 200 at a concentration of about 2.5% (w/v) to about 3.5% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0523] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 200 at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0524] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 6.9% (w/v) to about 7.7% (w/v), proline at a concentration of about 40 to about 80 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0525] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 5% (w/v) to about 6% (w/v), proline at a concentration of about 90 to about 150 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0526] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 2% (w/v) to about 3% (w/v), proline at a concentration of about 150 to about 210 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0527] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, proline at a concentration of about 200 to about 300 mM, calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0528] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0529] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, sorbitol at a concentration of about 3.5% (w/v) to about 4.5% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0530] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0531] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, sorbitol at a concentration of about 3.5% (w/v) to about 4.5% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0532] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0533] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0534] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0535] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0536] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 5% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0537] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0538] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, sucrose at a concentration of about 6.3% (w/v) to about 6.7% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.2% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0539] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0540] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 110 mg/mL, sucrose at a concentration of about 6.3% (w/v) to about 6.7% (w/v), calcium chloride at a concentration of about 20 to about 30 mM, and Pluronic F68 at a concentration of about 0.03% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0541] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0542] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 80 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0543] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0544] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.1% (w/v) to about 6.9% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Polysorbate 20 at a concentration of about 0.01% (w/v) to about 0.1% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl, NaOH, CaCl.sub.2), MEA, and/or MSA.
[0545] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, proline at a concentration of about 200 to about 250 mM, calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0546] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 11% (w/v) to about 15% (w/v), calcium chloride at a concentration of about 20 to about 40 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0547] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 160 to about 190 mg/mL, sucrose at a concentration of about 5% (w/v) to about 8% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0548] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sucrose at a concentration of about 6.9% (w/v) to about 7.7% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0549] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 200 at a concentration of about 4.5% (w/v) to about 5.1% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0550] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, sorbitol at a concentration of about 3% (w/v) to about 5% (w/v), calcium chloride at a concentration of about 15 to about 35 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with HCl.
[0551] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 13% (w/v) to about 16% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0552] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 90 to about 120 mg/mL, PEG 600 at a concentration of about 3% (w/v) to about 3.6% (w/v), calcium chloride at a concentration of about 10 to about 30 mM, proline at a concentration of about 150 to about 210 mM, and Pluronic F68 at a concentration of about 0.05% (w/v) to about 0.5% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0553] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL and PEG 600 at a concentration of about 10% (w/v) to about 11% (w/v), and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0554] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 8% (w/v) to about 9% (w/v), and calcium chloride at a concentration of about 15 to about 35 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0555] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 6% (w/v) to about 7% (w/v), and calcium chloride at a concentration of about 40 to about 60 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0556] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, PEG 600 at a concentration of about 3.4% (w/v) to about 4% (w/v), and calcium chloride at a concentration of about 65 to about 85 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0557] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL and calcium chloride at a concentration of about 80 to about 120 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0558] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL and proline at a concentration of about 250 to about 350 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0559] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 200 to about 250 mM, and calcium chloride at a concentration of about 15 to about 35 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0560] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 120 to about 180 mM, and calcium chloride at a concentration of about 40 to about 60 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0561] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 60 to about 90 mM, and calcium chloride at a concentration of about 65 to about 85 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0562] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 200 to about 300 mM, MEA at a concentration of about 40 to about 80 mM, and MSA at a concentration of about 40 to about 80 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0563] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 150 to about 210 mM, MEA at a concentration of about 40 to about 80 mM, and MSA at a concentration of about 40 to about 80 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0564] In an embodiment, the stable aqueous adalimumab formulation includes adalimumab at a concentration of about 40 to about 60 mg/mL, proline at a concentration of about 90 to about 150 mM, MEA at a concentration of about 70 to about 110 mM, and MSA at a concentration of about 70 to about 110 mM, and has a pH of about 5.1 to 5.3. Optionally, the pH of the formulation is adjusted within this range with MEA and/or MSA.
[0565] Exemplary aqueous adalimumab formulations are provided in Table J. Each formulation in Table J may optionally include Pluronic F68 at a concentration of about 0.001% (w/v) to about 10% (w/v) (e.g., about 0.005% (w/v) to about 1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 1% (w/v), about 0.05% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.4% (w/v), about 0.01% (w/v), about 0.05% (w/v) to about 0.5% (w/v), about 0.1% (w/v) to about 2% (w/v), about 0.2% (w/v) to about 1% (w/v), about 0.05% (w/v), about 0.1% (w/v), and/or about 0.4% (w/v)), Polysorbate 20 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.02% (w/v) to about 0.1% (w/v), and/or about 0.05% (w/v) to about 0.2% (w/v)), or Polysorbate 80 at a concentration of about 0.001% (w/v) to about 2% (w/v) (e.g., about 0.002% (w/v) to about 0.01% (w/v), about 0.005% (w/v) to about 0.1% (w/v), about 0.005% (w/v) to about 0.05% (w/v), about 0.02% (w/v) to about 0.1% (w/v), about 0.03% (w/v) to about 0.1% (w/v), 0.04% (w/v) to about 0.1% (w/v), about 0.05% (w/v) to about 0.2% (w/v), about 0.03% (w/v), about 0.04% (w/v), and/or about 0.1% (w/v)). Each formulation in Table J has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table J is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00011 TABLE J Protein conc. Excipient(s) 100 mg/mL 4% (w/v) sorbitol 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 25 mM CaCl.sub.2
[0566] Exemplary aqueous adalimumab formulations are provided in Table K. Each formulation in Table K has a pH of about 5.0 to about 5.5, e.g., about 5.1 to about 5.3 and/or about 5.2. Optionally, the pH of each formulation in Table K is adjusted using a strong acid and/or strong base including, but not limited to, hydrochloric acid, sodium hydroxide, calcium hydroxide, MSA, and/or MEA.
TABLE-US-00012 TABLE K Protein conc. Excipient(s) Surfactant 100 mg/mL 4% (w/v) sorbitol 0.1% (w/v) Pluronic F68 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.1% (w/v) Pluronic F68 25 mM CaCl.sub.2 100 mg/mL 4% (w/v) sorbitol 0.05% (w/v) Pluronic F68 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.05% (w/v) Pluronic F68 25 mM CaCl.sub.2 100 mg/mL 4% (w/v) sorbitol 0.1% (w/v) Polysorbate 80 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.1% (w/v) Polysorbate 80 25 mM CaCl.sub.2 100 mg/mL 4% (w/v) sorbitol 0.05% (w/v) Polysorbate 80 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.05% (w/v) Polysorbate 80 25 mM CaCl.sub.2 100 mg/mL 4% (w/v) sorbitol 0.1% (w/v) Polysorbate 20 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.1% (w/v) Polysorbate 20 25 mM CaCl.sub.2 100 mg/mL 4% (w/v) sorbitol 0.05% (w/v) Polysorbate 20 25 mM CaCl.sub.2 100 mg/mL 6.5% (w/v) sucrose 0.05% (w/v) Polysorbate 20 25 mM CaCl.sub.2
[0567] Exemplary aqueous adalimumab formulations are provided in Table L.
TABLE-US-00013 TABLE L Protein pH Adjusting conc. Excipient(s) Agent Surfactant pH 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.1% (w/v) Pluronic F68 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.1% (w/v) Pluronic F68 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.05% (w/v) Pluronic F68 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.05% (w/v) Pluronic F68 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.1% (w/v) Polysorbate 80 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.1% (w/v) Polysorbate 80 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.05% (w/v) Polysorbate 80 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.05% (w/v) Polysorbate 80 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.1% (w/v) Polysorbate 20 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.1% (w/v) Polysorbate 20 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 4% (w/v) sorbitol HCl/NaOH, 0.05% (w/v) Polysorbate 20 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA 100 mg/mL 6.5% (w/v) sucrose HCl/NaOH, 0.05% (w/v) Polysorbate 20 5.2 25 mM CaCl.sub.2 HCl/Ca(OH).sub.2, HCl/MEA, MSA/Ca(OH).sub.2, or MSA/MEA
[0568] In an embodiment, a lyophilized form of any one of the foregoing adalimumab formulations without buffer is provided.
Methods of Treatment
[0569] The invention provides for methods of treating a patient suffering from a TNF-.alpha. associated disease or disorder comprising administering to the patient any of the stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection). The TNF-.alpha. diseases and disorders include, but are not limited to, inflammatory diseases and disorders, intestinal diseases and disorders, autoimmune diseases and disorders, eye diseases and disorders, pulmonary diseases and disorders, and infectious diseases and disorders. The term "patient" includes humans and non-human animal subjects.
[0570] The invention also provides for compositions comprising any of the stable aqueous or lyophilized adalimumab formulations of the invention for the treatment of a TNF-.alpha. associated disease or disorder, such as inflammatory diseases and disorders, intestinal diseases and disorders, autoimmune diseases and disorders, eye diseases and disorders, pulmonary diseases and disorders, and infectious diseases and disorders.
[0571] The invention further provides for use of the stable aqueous or lyophilized adalimumab formulations of the invention for the preparation of a medicament for the treatment of TNF-.alpha. associated diseases or disorders, such as inflammatory diseases and disorders, intestinal diseases and disorders, autoimmune diseases and disorders, eye diseases and disorders, pulmonary diseases and disorders, and infectious diseases and disorders.
[0572] Inflammatory diseases and disorders include, but are not limited to, arthritis, adult and juvenile rheumatoid arthritis, juvenile idiopathic arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, osteoarthritis including erosive osteoarthritis and hund osteoarthritis, plaque psoriasis, general pustular psoriasis, nail and scalp psoriasis, hidradenitis suppurativa, ankylosing spondylitis, interstitial cystitis, spondyloarthritis including peripheral spondyloarthritis and axial spondyloarthritis, spondylarthropathy, pulmonary inflammation disorder, allergy, uveitis, chronic pulmonary inflammation disease, vascular inflammation, enthesitis related arthritis, enthesopathy, coronary atherosclerosis, inflammatory bone disorders, bone resorption disease, hepatitis including alcoholic hepatitis, chronic pouchitis, inflammatory bowel disease, adult and pediatric Crohn's disease, ulcerative colitis, schleritis, sarcoidosis, cutaneous sarcoidosis, Netherton syndrome, and giant cell arteritis.
[0573] Intestinal diseases and disorders include, but are not limited to, chronic pouchitis, inflammatory bowel disease, adult and pediatric Crohn's disease, ulcerative colitis, small bowel lesions, anal squamous intraepithelial lesions, anal fissures, and intestinal Behcet's disease.
[0574] Autoimmune diseases and disorders include, but are not limited to, adult and juvenile rheumatoid arthritis, juvenile idiopathic arthritis, psoriasis, plaque psoriasis, general pustular psoriasis, nail and scalp psoriasis, psoriasis vulgaris, psoriasis arthropica, psoriatic arthritis, pyoderma gangrenosum, gouty arthritis, allergy, multiple sclerosis, autoimmune diabetes, autoimmune uveitis, nephrotic syndrome, diabetic ulcers, and graft vs. host disease
[0575] Eye diseases and disorders include, but are not limited to, uveitis, anterior uveitis, intermediate uveitis and posterior uveitis, refractory diabetic retinopathy, choroid diseases, choroidal neovascularization, macular degeneration including age-related macular degeneration, albinism, Behcet's syndrome, Hermanski-Pudluk syndrome, panuveitis, pars planitis, retinal degeneration, uveal diseases, retinal vascular disorders, and schleritis.
[0576] Infectious diseases and disorders include, but are not limited to, malaria, acquired immune deficiency (AIDS), cytomegalovirus infection and influenza.
[0577] Pulmonary disease and disorders include, but are not limited to, adult respiratory distress syndrome, asthma, refractory asthma, pulmonary inflammation disorder, shock lung, chronic pulmonary inflammatory disease, pulmonary sarcoidosis, pulmonary fibrosis and silicosis.
[0578] Other TNF-.alpha. associated diseases and disorders include, but are not limited to, mucopolysaccharidosis including mucopolysaccharidosis type I, mucopolysaccharidosis type II, mucopolysaccharidosis type IV, cancers, cachexia, ischemia of the heart, coagulation disturbances, acute disc prolapse, sleep apnea, anaplastic thyroid cancer and focal segmental glomeruloschelorisis.
[0579] Stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) may be administered subcutaneously, intravenously, parenterally, intradermally, intramuscularly, and/or intraperitoneally using standard techniques. For example, the stable aqueous adalimumab formulations of the invention may be prepared to be subcutaneously administered using a pre-filled syringe. Specifically, any of the formulations of the invention may be administered once every week or 6 to 8 days or 7 to 10 days, or every other week or every two weeks or 12 to 16 days or 7 to 14 days or 13 to 15 days, or every three weeks or 19 to 23 days, or every month or 26 to 30 days or 29 to 31 days, or every five weeks or 33 to 34 days, or every six weeks or 40 to 44 days, or every seven weeks or 47 to 51 days, or every two months or 54 to 58 days subcutaneously, intravenously, parenterally, intradermally, intramuscularly, and/or intraperitoneally at a therapeutically effective dosage and in the formulations described herein for an indefinite period of time for the treatment of the diseases and conditions described above.
[0580] Administration and dosage regimens of stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) can be adjusted to provide an effective amount for an optimum therapeutic response. For example, a single bolus can be administered, two or more divided doses can be administered over time or the dose can be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. For example, a unit dose can be administered over two consecutive days every two weeks. Unit dosing refers to a physically discrete amount of adalimumab or a biosimilar thereof suited as unitary dosages for the patients to be treated; each unit contains a predetermined quantity of active biopharmaceutical calculated to produce a desired therapeutic effect.
[0581] The dosing regimen of stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) may comprise administering a dose given on Day one, followed by the administration of the same dose every other week. For example, a dose of 40 mg adalimumab or biosimilar thereof is administered every other week in patients suffering from rheumatoid arthritis, psoriatic arthritis or ankylosing spondylitis. For patients suffering from juvenile idiopathic arthritis that are 10 kg (22 lbs) to less than 15 kg (33 lbs), a dose of 10 mg adalimumab or biosimilar thereof is administered every other week, for example. For patients suffering from juvenile idiopathic arthritis that are 15 kg (33 lbs) to less than 30 kg (66 lbs), a dose of 20 mg adalimumab or biosimilar thereof is administered every other week, for example. For patients suffering from juvenile idiopathic arthritis that are greater or equal to 30 kg (66 lbs), a dose of 40 mg adalimumab or biosimilar thereof is administered every other week, for example. This dosing regimen may also include administering methotrexate (MTX), other non-biologic DMARDS, glucocorticoid, nonsteroidal anti-inflammatory drugs (NSAIDS) and/or analgesics throughout the administration or for a portion of the time of administration of any of the stable aqueous adalimumab formulations of the invention.
[0582] The dosing regimen of stable aqueous adalimumab formulations of the invention may (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) comprise administering an initial dose given on day one or split over two consecutive days, followed by the administration of the same or a reduced dose two weeks later (Day 15), e.g. the initial dose reduced by half. The dosing regimen may further comprise administration of the same or further reduced dose two weeks later (Day 29); e.g. a dose that is a fourth of the initial dose which will be continued as a maintenance dose every two weeks. For example, for patients suffering from adult Crohn's disease or ulcerative colitis, an initial dose of 160 mg adalimumab or biosimilar thereof is administered on Day 1, a second dose of 80 mg adalimumab or biosimilar thereof is administered two weeks later (Day 15), followed by a maintenance dose of 40 mg adalimumab or biosimilar thereof administered two weeks later (Day 29) that is continued every other week. For patients suffering from pediatric Crohn's disease that are 17 kb (37 lbs) to less than 40 kg (88 lbs), an initial dose of 80 mg adalimumab or biosimilar thereof is administered on Day 1, a second dose of 40 mg adalimumab or biosimilar thereof is administered two weeks later (Day 15), followed by a maintenance dose of 20 mg adalimumab or biosimilar thereof administered two weeks later (Day 29) that is continued every other week, for example. For patients suffering from pediatric Crohn's disease that are greater than 40 kg (88 lbs), an initial dose of 160 mg adalimumab or biosimilar thereof is administered on Day 1, a second dose of 80 mg adalimumab or biosimilar thereof is administered two weeks later (Day 15), followed by a maintenance dose of 40 mg adalimumab or biosimilar thereof administered two weeks later (Day 29) that is continued every other week, for example. This dosing regimen may also include administering aminosalicylates and/or corticosteroids, azathioprine, 6-mercaptopurine (6-MP) or MTX throughout the administration or for a portion of the time of administration of any of the stable aqueous adalimumab formulations of the invention.
[0583] The dosing regimen of stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) may comprise administering an initial dose given on Day one or split over two consecutive days, followed by the administration of the same or a reduced dose two weeks later (Day 15), e.g. an initial dose reduced by half. The dosing regimen may further comprise administration of the same or further reduced dose two weeks later (Day 29), e.g. a dose that is a fourth of the initial dose. For example, an initial dose of 160 mg adalimumab or biosimilar thereof is administered on Day 1, a second dose of 80 mg adalimumab or biosimilar thereof is administered two weeks later (Day 15), and a third dose of 40 mg adalimumab or biosimilar thereof is administered on Day 29 followed by administration of 40 mg adalimumab or biosimilar thereof every week. This dosing regimen may be administered to patients suffering from hidradenitis suppurativa.
[0584] The dosing regimen of stable aqueous adalimumab formulations of the invention (or lyophilized formulations thereof once reconstituted, e.g., with sterile water for injection) may comprise administering an initial dose given on Day one or split over two consecutive days, followed by the administration of the same or a reduced dose one week after the initial dose e.g. an initial dose reduced by half and continued administration every other week. For example, an initial dose of 80 mg adalimumab or biosimilar thereof, followed by administration of 40 mg adalimumab or biosimilar thereof every other week starting one week after the initial dose. This dosing regimen may be administered to patients suffering from plaque psoriasis or uveitis.
[0585] The invention provides for a method of preparing a stable aqueous adalimumab formulations of the invention, comprising combining an aqueous solution comprising one or more excipients and an therapeutically effective amount of adalimumab using techniques standard in the art. The invention further provides for a method of preparing the stable lyophilized adalimumab formulations of the invention, comprising lyophilizing an aqueous adalimumab formulation comprising one or more excipients and a therapeutically effective amount of adalimumab using techniques standard in the art.
[0586] The foregoing detailed description is not intended to define every aspect of the invention, and other features and advantages of the present disclosure will be apparent to those skilled in the art. The present disclosure is intended to be related as a unified document, and it should be understood that all combinations of features described herein are contemplated, even if the combination of features are not found together in the same sentence, paragraph, or section of this disclosure. In addition, the disclosure includes, as an additional aspect, all embodiments of the invention narrower in scope in any way than the variations specifically mentioned above. With respect to aspects of the disclosure described or claimed with "a" or "an," it should be understood that these terms mean "one or more" unless context unambiguously requires a more restricted meaning. With respect to elements described as one or more within a set, it should be understood that all combinations within the set are contemplated. If aspects of the disclosure are described as "comprising" a feature, embodiments also are contemplated "consisting of" or "consisting essentially of" the feature. Additional features and variations of the disclosure will be apparent to those skilled in the art from the entirety of this application, and all such features are intended as aspects of the disclosure.
[0587] The present disclosure will be more readily understood by reference to the following examples, which are provided by way of illustration and are not intended to be limiting.
EXAMPLES
[0588] The following Examples describe formulations of the present disclosure.
[0589] General Materials: In the following examples, an adalimumab biosimilar as described in Velayudhan et al., BioDrugs 30:339-351 (2016) (i.e., ABP 501) was used.
[0590] General Analytical Methods: Cation-exchange high-performance liquid chromatography (CEX-HPLC) and size-exclusion high-performance liquid chromatography (SE-HPLC) were used to assess stability. CEX-HPLC examines changes in charge, mainly due to deamidation, which is measured as pre-peak or acidic growth, and SE-HPLC is used to resolve and measure soluble aggregation, also known as high molecular weight species (HMWS), which is determined as a growth in pre-peak area. To be considered significant, changes in degradation should be greater than the intermediate precision of the assays: the standard deviation is +/-0.16 for the CEX-HPLC method and +/-0.032 for the SE-HPLC method. CEX-HPLC was performed using, a Pro Pac WCX-10 analytical column, 4.0 mm.times.250 mm (Dionex, 054993) for the charge separation of protein in a gradient mobile phase. Mobile Phase A was 20 mM sodium phosphate, pH 6.8 and Mobile Phase B was 20 mM sodium phosphate, 0.5 M NaCl, pH 6.8. Samples were injected onto the column at a 50 .mu.g mass load and detected at a wavelength of 230 nm. SE-HPLC was performed using an Agilent 1200 system. A TSK-GEL G3000SWXL column, 5 .mu.M particle size, 7.8.times.300 nm (Tosoh Bioscience, 08541) to separate protein by size. A UV detector with a wavelength of 220 nm was used to detect samples injected at a mass load of 35 .mu.g. The mobile phase which allows for separation on the column was 100 mM sodium phosphate, 250 mM sodium chloride, pH 6.8.
[0591] Opalescence measurement was also used to assess stability. Opalescence examines physical instability of a formulation due to the presence of aggregates. Opalescence was measured in a 2100 AN Turbidimeter, using 13 cm.times.100 mm glass tubes and a 13 mm sample tube adapter. Samples of at least 2.5 mL were used for each measurement. A standard curve was generated using Stablcal.RTM. turbidity standard (Hach Company) prepared to expected turbidimetry readings of 3, 6, 18 and 30 nephelometric turbidity units (NTUs) by diluting a stock standard in water.
[0592] Transport stress studies were also used to assess stability. Frozen formulations were thawed statically at room temperature protected from light using a TempTale.RTM. temperature monitor to monitor temperature conditions in the cold room during the thaw. After fully thawing, the cap on the bottle was tightened to avoid leakage. The bottle was placed on its side and rolled gently for 4-6 minutes to ensure thorough mixing. The bottle was then placed at -30.degree. C. for 8 hours to re-freeze. Freezing was confirmed visually. The freeze-thaw process was repeated for a total of 3 cycles. Next, the formulation was mixed with an overhead mixer for 15 minutes to simulate shear from a mixing operation, and then filtered at 2-8.degree. C. under pressure using a 0.2 .mu.m PVDF filter. The formulation was sterile-filtered a second time prior to filling into syringes followed by stoppering. The formulations were pressure-filtered through a 0.2 .mu.m PVDF filter, then hand-filled into syringes or vials. The hand-filled syringes were stoppered using an ASPU (autoclavable stopper placement unit) system. The filled and stoppered syringes were placed in a laminar air flow hood at room temperature for 3 days to mimic room temperature and light exposure stresses expected during manufacturing. Temperature was recorded using a TempTale.RTM. temperature monitor, and UV and visible light levels were recorded using a photometer. A portion of the filled syringes were subjected to simulated air and ground transportation stresses. Air and ground simulated transport stress studies were carried out. for a total duration of 91.5 hours, with air transportation vibration of 48 hours and truck transportation vibration for 43.5 hours. Samples were also subjected to the International Safe Transit Association (ISTA 3A) drop test sequence six times which included drops 1 thru 9 prior to the 91.5 hour air and ground vibrational simulation and drops 10 thru 17 after the air and ground vibrational transport simulation. The syringes were stored for 2 weeks at 40.degree. C. Stability was assessed by SE-HPLC, CEX-HPLC, and micro-flow imaging (MFI). MFI measures the presence of sub-visible particles by passing a sample through a visual flow cell, counting particles as they pass through the cell, and categorizing into different bins based on size. An aspect ratio is applied to resolve silicone oil, if present, from proteinaceous particles. MFI (Micro-Flow Imaging) was performed on MFI 5200 systems. The size range of particles measured was from 1 to 70 .mu.m. The sample volume measured was 1 mL, with pre filled syringes pooled into a clean glass vial to allow for adequate volume of at least 1 mL for measurement. Between each measurement, the system was flushed and a baseline established before proceeding. For each sample measurement, a digital camera is used to magnify, record the size, shape and morphology of visible particles.
[0593] Conductivity is the ability of an aqueous solution to conduct an electric current between two electrodes. Because a current flows via ion transport, the more ions in a particular solution, the higher the conductivity. Conductivity of the formulations described herein was assessed on a Model CDM83, Thermo Orion 4 or Model 5230 Seven Compact, Mettler Toledo instrument, using a conductivity cell. At minimum of at least 20 mL sample (Thermo Orion) or 3 mL sample (Mettler Toledo) was used for each measurement. The cell was rinsed with water and dried between sample measurements. Conductivity measurements were performed at ambient room temperature, and is reported using the standard SI unit of siemens per meter (S/m).
[0594] Osmolality is the concentration of a solution in terms of amount of solute quantity of solvent. For example, serum has an osmolality ranging from about 270-300 mOsM. Osmolality of the formulations described herein can be determined using, for example, Freezing Point Depression Osmometry.
Example 1
Effect of Buffer Composition on Formulation Stability
[0595] Adalimumab biosimilar was prepared in a target formulation buffer by centrifuge concentration using a 30 kD MW cutoff filter tube. 2 mL of adalimumab biosimilar was diluted with the target formulation buffer to a volume of 15 mL, followed by a centrifuge concentration step to a final volume of around 2 mL before repeating the dilution and centrifuge concentration step three times. Around 3 mL were collected after the last centrifuge concentration step, which was diluted with the desired formulation buffer to 100 mg/mL, sterile filtered and aliquoted into 5 cc glass vials. Following the filling step, samples were placed at 40.degree. C. and examined for stability at approximately 1 week, 2 weeks and 4 weeks.
[0596] The formulation buffers used for the centrifuge concentration step contained various buffers, with the pH of the buffer adjusted to 5.2.+-.0.1 using either NaOH or monoethanolamine (MEA). Each buffer contained isotonic proline as an additional excipient. Formulations without buffer were prepared by adjusting the pH of methane sulfonic acid (MSA) to pH 5.2.+-.0.1 with MEA or NaOH. The composition of the formulation buffer, and the pH and antibody (Ab) concentration of the formulations are provided in Table 1.
TABLE-US-00014 TABLE 1 pH adjusting Ab conc. Ref. Buffer Excipient agent pH (mg/mL) 1A 20 mM benzoate 300 mM proline MEA 5.2 73 1B 20 mM benzoate 300 mM proline NaOH 5.2 109 1C 20 mM glutamate 300 mM proline MEA 5.2 105 1D 20 mM glutamate 300 mM proline NaOH 5.1 111 1E 20 mM glycolate 300 mM proline MEA 5.2 110 1F 20 mM glycolate 300 mM proline NaOH 5.2 108 1G 20 mM lactate 300 mM proline MEA 5.1 109 1H 20 mM lactate 300 mM proline NaOH 5.1 104 1I -- 20 mM MSA MEA 5.3 100 300 mM proline 1J -- 20 mM MSA NaOH 5.3 107 300 mM proline
[0597] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 6, 13, and 28 days at 40.degree. C. As shown in FIG. 1, the formulations containing lactate or glycolate buffers demonstrated less acidic peak growth over time compared to the formulations containing benzoate or glutamate buffers. The formulations without buffer, but including MSA, demonstrated stability comparable to the buffered formulations and improved compared to the benzoate and glutamate-buffered formulations at the 28 day time point.
[0598] Stability also was assessed by measuring high molecular weight species (HMWS) by SE-HPLC after 0, 6, 13, and 28 days at 40.degree. C. As shown in FIG. 2, the formulations containing lactate demonstrated significantly less HMWS growth over time compared to the other formulations. Compared to glycolic acid, lactic acid has a structure that differs only by including an additional methyl group. As shown in FIG. 2, HMWS were surprisingly lower for the formulations containing lactate compared to the formulations containing glycolate. The formulations without buffer, but including MSA, demonstrated stability comparable to the buffered formulations.
Example 2
Effect of PEG, Dextran, and Calcium Chloride on Formulation Stability
[0599] Adalimumab biosimilar solution at a concentration of 220 mg/mL ("UF DF stock") was prepared by ultrafiltration/diafiltration (UF DF) into a buffer of 20 mM glutamate, pH 5.1 using a Cogent .mu.Scale tangential flow filtration (TFF) system with a delta pressure set to about 23 psi. A Millipore Pellicon 3 Ultracell 30 kD 0.11 m.sup.2 cassette was used as the exchange filter. The resulting material was then further concentrated to 220 mg/mL to obtain the UF DF stock.
[0600] Stock 2.times. excipient solutions were prepared, and were then diluted into UF DF stock adalimumab biosimilar solution. Upon final dilution and mixing in of the 2.times. excipients, the adalimumab biosimilar concentration was adjusted to about 100 mg/mL. If needed, the pH was adjusted to 5.2.+-.0.2 using NaOH or HCl. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 2. Following sterile filtration, aliquots were filled into 5 cc glass vials and stored at -30.degree. C., 4.degree. C., and 40.degree. C.
TABLE-US-00015 TABLE 2 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 2A 20 mM glutamate 7% PEG 3350 5.4 114 2B 20 mM glutamate 7% PEG 3350 5.3 114 45 mM calcium chloride 2C 20 mM glutamate 10% dextran 5.1 128 2D 20 mM glutamate 10% dextran 5.2 119 45 mM calcium chloride 2E 20 mM glutamate 1% PEG 200 5.1 110 2F 20 mM glutamate 1% PEG 200 5.1 115 40 mM calcium chloride 2G 20 mM glutamate 2% PEG 600 5.1 110 2H 20 mM glutamate 2% PEG 600 5.1 109 40 mM calcium chloride
[0601] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 3, the formulations containing calcium chloride and one of PEG 200, PEG 600, PEG 3350, or dextran demonstrated less acidic peak growth over time compared to otherwise similar formulations without calcium chloride. In addition, the formulations containing PEG 200 or PEG 600 demonstrated less acidic peak growth over time compared to the formulations containing PEG 3350 or dextran.
[0602] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 4, the formulations containing PEG 200, PEG 600, or PEG 3350 (alone or in combination with calcium chloride) demonstrated less HMWS growth over time compared to the formulations containing dextran.
Example 3
Effect of Proline, PEG, MSA, TEA, and Calcium Chloride on Frozen Stability
[0603] Stock 2.times. excipient solutions in 20 mM glutamate buffer, pH 5.2 were added to the UF DF stock adalimumab biosimilar solution described in Example 2 to a final protein concentration of 100 mg/mL. If needed, the pH was adjusted to 5.2.+-.0.2 using NaOH or HCl. The final formulations were sterile filtered and placed into 5 cc glass vials for subsequent tests.
[0604] Three free thaw (F/T) cycles were executed, with room temperature thaws of samples stored at -30.degree. C. at each cycle. After the freeze thaw cycles, each formulation was then analyzed for stability and the remainder of the material stored at -30.degree. C. for long term storage stability. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 3.
TABLE-US-00016 TABLE 3 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 3A 20 mM glutamate 250 mM proline 5.1 119 3B 20 mM glutamate 1% PEG 3350 5.4 114 2A 20 mM glutamate 7% PEG 3350 5.4 114 2B 20 mM glutamate 7% PEG 3350 5.3 114 45 mM calcium chloride 2E 20 mM glutamate 1% PEG 200 5.1 110 2F 20 mM glutamate 1% PEG 200 5.1 115 40 mM calcium chloride 2G 20 mM glutamate 2% PEG 600 5.1 109 2H 20 mM glutamate 2% PEG 600 5.1 109 40 mM calcium chloride 3C 20 mM glutamate 100 mM MSA 5.2 109 50 mM TEA
[0605] Stability was assessed by measuring HMWS by SE-HPLC after 0 days, after 3 F/T cycles, and after 83 days at -30.degree. C. As shown in FIG. 5, the formulations containing proline demonstrated almost no HMWS growth over time.
[0606] Stability was also assessed by measuring HMWS by SE-HPLC after 0 days, after 3 F/T cycles, after 35 days at -30.degree. C., and after 56 days at -30.degree. C. As shown in FIG. 6, the formulations containing PEG 600 and a formulation with MSA and triethanolamine (TEA) demonstrated a small decrease in HMWS growth upon F/T stress and storage at -30.degree. C. compared to the formulations containing PEG 200.
Example 4
Effect of Salts on Formulation Stability
[0607] The OF DF stock adalimumab biosimilar solution described in Example 2 was diluted to 170 mg/mL with 20 mM glutamate, pH 5.2 buffer and various salts were added directly by weight until dissolved at a final concentration of 75 mM. A stock concentrated solution of proline in the 20 mM glutamate, pH 5.2 buffer was also added to achieve a final concentration of 100 mM. After preparing the protein solutions, the pH was adjusted if needed with either HCl or NaOH to 5.2.+-.0.4. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 4. Following sterile filtration, aliquots were filled into 5 cc glass vials and stored at -30.degree. C., 4.degree. C., and 40.degree. C.
TABLE-US-00017 TABLE 4 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 4A 20 mM glutamate 75 mM sodium borate 5.6 175 100 mM proline 4B 20 mM glutamate 75 mM sodium bicarbonate 5.2 180 100 mM proline 4C 20 mM glutamate 75 mM sodium sulfate 5.2 183 100 mM proline 4D 20 mM glutamate 15 mM calcium sulfate 5.1 178 100 mM proline 4E 20 mM glutamate 75 mM ammonium sulfate 5.2 175 100 mM proline 4F 20 mM glutamate 75 mM calcium chloride 5.1 172 100 mM proline 4G 20 mM glutamate 75 mM sodium chloride 5.2 167 100 mM proline 4H 20 mM glutamate 75 mM magnesium chloride 5.1 193 100 mM proline
[0608] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 7, the formulations containing calcium chloride or magnesium chloride demonstrated less acidic peak growth over time compared to the formulations containing sodium borate, sodium bicarbonate, sodium sulfate, calcium sulfate, ammonium sulfate, or sodium chloride.
[0609] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 8, the formulations containing calcium sulfate, ammonium sulfate, calcium chloride, sodium chloride, or magnesium chloride demonstrated less HMWS growth over time compared to the formulations containing sodium borate, sodium bicarbonate, or sodium sulfate.
Example 5
Effect of MEA on Formulation Stability
[0610] The OF DF stock adalimumab biosimilar solution described in Example 2 was diluted with 20 mM glutamate buffer, pH 5.0 and 2M MEA in volumes needed to generate the final MEA concentrations shown in Table 5. After preparing the protein solutions, the pH was adjusted if needed with either HCl or NaOH to 5.2. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 5. Following sterile filtration, aliquots were filled into 5 cc glass vials and stored at 4.degree. C. or 40.degree. C.
TABLE-US-00018 TABLE 5 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 5A 20 mM glutamate 30 mM MEA 5.2 182 5B 20 mM glutamate 80 mM MEA 5.2 182 5C 20 mM glutamate 115 mM MEA 5.2 185 5D 20 mM glutamate 160 mM MEA 5.2 177
[0611] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 9, the formulations containing higher concentrations of MEA (e.g., 160 mM MEA) demonstrated less acidic peak growth over time compared to the formulations containing lower concentrations of MEA (e.g., 30 mM MEA). The ratio of % acidic peak after 28 days to the % acidic peak at 0 days was 2.23 for formulation 5A, 2.17 for formulation 5B, 2.11 for formulation 5C, and 2.09 for formulation 5D.
[0612] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 10, the formulations containing higher concentrations of MEA (e.g., 160 mM MEA) demonstrated slightly less HMWS growth over time compared to the formulations containing lower concentrations of MEA (e.g., 30 mM MEA).
Example 6
Effect of Excipients on Formulation Stability
[0613] The OF DF stock adalimumab biosimilar solution described in Example 2 was diluted to around 105 mg/mL once excipients were added. In each formulation, 2.times. stock excipient solutions in 20 mM glutamate buffer, pH 5.2 were added to a final protein concentration of around 105 mg/mL. The pH was adjusted to 5.2.+-.0.1 with HCl or NaOH if needed. The final formulations were sterile filtered and placed into 5 cc glass vials for subsequent tests. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 6.
TABLE-US-00019 TABLE 6 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 6A 20 mM glutamate 1.4% ethanol 5.1 102 6B 20 mM glutamate 0.5% ethanol 5.2 106 40 mM calcium chloride 6C 20 mM glutamate 0.5% ethanol 5.2 108 1% PEG 3350 40 mM calcium chloride 6D 20 mM glutamate 150 mM TEA 5.2 107 40 mM calcium chloride 6E 20 mM glutamate 30 mM TEA 5.2 105 75 mM calcium chloride 6F 20 mM glutamate 0.5% ethanol 5.2 106 6G 20 mM glutamate 0.5% ethanol 5.2 106 2% PEG 200 6H 20 mM glutamate 0.25% ethanol 5.2 102 2% PEG 200 6I 20 mM glutamate 0.5% ethanol 5.1 105 100 mM MSA 6J 20 mM glutamate 0.5% ethanol 5.2 105 0.5% poly(vinylpyrrolidone) (PVP) 10K 100 mM MSA 6K 20 mM glutamate 0.5% ethanol 5.2 101 100 mM MEA
[0614] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 11, the formulations containing ethanol with calcium chloride, ethanol with calcium chloride and PEG 3350, TEA with calcium chloride, and ethanol with MEA demonstrated less than 30% of acidic peak after 4 weeks at 40.degree. C. The formulations containing 1.4% ethanol with no additional excipients demonstrated stability comparable to the formulations containing 0.5% ethanol with no additional excipients at the 4 week time point.
[0615] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 7, 14, and 28 days at 40.degree. C. As shown in FIG. 12, the formulation containing 1.4% ethanol with no additional excipients demonstrated less than 1% of HMWS after 4 weeks at 40.degree. C. The formulations containing 0.5% ethanol, ethanol in combination with calcium chloride, ethanol in combination with calcium chloride and PEG 3350, ethanol in combination with PEG 200, and ethanol in combination with MEA demonstrated less than 1.5% of HMWS at the 4 week time point.
Example 7
Effect of Amino Acids on Formulation Stability
[0616] Adalimumab biosimilar was prepared in a buffer containing 15 mM glutamate, pH 5.2 using a Cogent .mu.Scale TFF system with a 30 kD Millipore cassette and a pressure difference of about 23 psi. The protein was concentrated to 114 mg/mL, and the resulting material in 15 mM glutamic acid, pH 5.2 buffer was then concentrated by centrifugation concentration to 186 mg/mL. In each formulation, 2.times. stock excipient solutions in 15 mM glutamate buffer, pH 5.2 were added to the starting material, resulting in a final protein concentration of around 90-100 mg/mL. The pH was adjusted to 5.2 with HCl or NaOH if needed. The final formulations were sterile filtered and placed into 5 cc glass vials for subsequent tests. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 7.
TABLE-US-00020 TABLE 7 Ab conc. Ref. Buffer Excipient(s) pH (mg/mL) 7A 15 mM glutamate 100 mM alanine 5.2 92 45 mM calcium chloride 7B 15 mM glutamate 100 mM asparagine 5.2 95 45 mM calcium chloride 7C 15 mM glutamate 100 mM isoleucine 5.2 96 45 mM calcium chloride 7D 15 mM glutamate 100 mM serine 5.2 98 45 mM calcium chloride 7E 15 mM glutamate 20 mM aspartic acid 5.2 96 80 mM proline 45 mM calcium chloride 7F 15 mM glutamate 25 mM creatine 5.2 96 75 mM proline 45 mM calcium chloride 7G 15 mM glutamate 50 mM glutamine 5.2 97 50 mM proline 45 mM calcium chloride 7H 15 mM glutamate 50 mM leucine 5.2 97 50 mM proline 45 mM calcium chloride 7I 15 mM glutamate 50 mM phenylalanine 5.2 97 50 mM proline 45 mM calcium chloride 7J 15 mM glutamate 25 mM tryptophan 5.2 90 75 mM proline 45 mM calcium chloride
[0617] To assess stability, the % acidic peak was measured by CEX-HPLC after 0 and 7 days at 40.degree. C. As shown in FIG. 13, the formulations containing alanine, serine, proline in combination with glutamine, or proline in combination with creatine, for example, demonstrated 18-19% of acidic peak after 7 days incubation at 40.degree. C., in comparison to the formulation containing proline in combination with phenylalanine, which demonstrated almost 20% of acidic peak after 7 days.
[0618] Stability also was assessed by measuring HMWS by SE-HPLC after 0 and 7 days at 40.degree. C. As shown in FIG. 14, the formulations containing alanine, asparagine, isoleucine, serine, proline in combination with aspartic acid, proline in combination with creatine, proline in combination with glutamine, and proline in combination with tryptophan demonstrated about 0.5% or less of HMWS after 7 days at 40.degree. C.
Example 8
Effect of Nonionic, Anionic, or Cationic Surfactants on Formulation Stability
[0619] Adalimumab biosimilar was prepared using a Cogent .mu.Scale TFF system with a 30 kD Millipore cassette and a pressure difference of about 23 psi in a buffer containing 20 mM acetate, 45 mM calcium chloride, and 100 mM arginine, with a final pH of 5.2. The resulting protein was concentrated to greater than 143 mg/mL. The material was then used for the addition of stock surfactant solutions to the final concentrations of surfactant and protein listed in Table 8. Final pH adjustment to 5.2 was accomplished with NaOH or HCl. For accelerated stability tests, aliquots were filled into 5 cc glass vials and examined for stability at 40.degree. C. For shaking stress studies, 36 mL of each formulation in 50 cc containers was subjected to continued shaking stress at room temperature. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 8.
TABLE-US-00021 TABLE 8 Ab conc. Ref. Buffer Surfactant Excipient(s) pH (mg/mL) 8A 20 mM acetate 0.1% Polysorbate 20 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl 8B 20 mM acetate 0.1% Polysorbate 80 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl 8C 20 mM acetate 0.1% Pluronic F68 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl 8D 20 mM acetate 0.01% Docusate 45 mM calcium 5.2 140 sodium chloride 100 mM arginine-HCl 8E 20 mM acetate 0.1% benzalkonium 45 mM calcium 5.2 140 chloride chloride 100 mM arginine-HCl 8F 20 mM acetate 0.1% Span 40 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl 8G 20 mM acetate 0.1% Triton X-100 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl 8H 20 mM acetate -- 45 mM calcium 5.2 140 chloride 100 mM arginine-HCl
[0620] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 3, and 7 days at 40.degree. C. As shown in FIG. 15, the formulations containing nonionic, anionic, or cationic surfactants demonstrated similar amounts of acidic peak after 7 days at 40.degree. C., in comparison to a formulation without surfactant.
[0621] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 3, 7, and 14 days at 40.degree. C. As shown in FIG. 16, the formulations containing nonionic, anionic, or cationic surfactants demonstrated similar amounts of HMWS after 14 days at 40.degree. C., in comparison to a formulation without surfactant.
[0622] Stability was further assessed after subjecting the formulations to shaking stress. The % acidic peak was measured by CEX-HPLC after continuous shaking at room temperature for 0 and 158 hours. As shown in FIG. 17, the formulations containing surfactants demonstrated around 0.3% or less of acidic peak after continuous shaking at room temperature for 158 hours, in comparison to a formulation without surfactant. HMWS were measured by SE-HPLC after continuous shaking at room temperature for 0 and 158 hours. As shown in FIG. 18, the formulation containing benzalkonium chloride demonstrated about twice the percentage of HMWS, in comparison to the formulations containing other surfactants and a formulation without surfactant.
Example 9
Effect of Nonionic, Anionic, or Cationic Surfactants on Formulation Stability
[0623] Adalimumab biosimilar in a buffer consisting of 20 mM acetate, with 45 mM calcium chloride and 100 mM arginine, with a final pH set at 5.2 was used to assess stability upon stirring stress. The final formulation pH was adjusted to 5.2 using NaOH or HCl. Surfactants were chosen and selected at low, medium and high levels to assess stirring stability with adalimumab biosimilar at a final concentration of approximately 140 mg/mL. Stock surfactant solutions were made and diluted with the starting material to achieve the final surfactant concentrations as shown in Table 9. For each formulation condition, 30 mL were prepared, transferred to 50 cc containers and stirred continuously for 5 days at room temperature.
TABLE-US-00022 TABLE 9 Ab conc. Ref. Buffer Surfactant Excipient(s) pH (mg/mL) 9A 20 mM acetate 0.005% Polysorbate 80 45 mM calcium 5.2 137 chloride 100 mM arginine-HCl 9B 20 mM acetate 0.01% Polysorbate 80 45 mM calcium 5.2 135 chloride 100 mM arginine-HCl 9C 20 mM acetate 0.05% Polysorbate 80 45 mM calcium 5.2 138 chloride 100 mM arginine-HCl 9D 20 mM acetate 0.005% Polysorbate 20 45 mM calcium 5.2 138 chloride 100 mM arginine-HCl 9E 20 mM acetate 0.01% Polysorbate 20 45 mM calcium 5.2 136 chloride 100 mM arginine-HCl 9F 20 mM acetate 0.05% Polysorbate 20 45 mM calcium 5.2 136 chloride 100 mM arginine-HCl 9G 20 mM acetate 0.005% Triton X-100 45 mM calcium 5.2 134 chloride 100 mM arginine-HCl 9H 20 mM acetate 0.01% Triton X-100 45 mM calcium 5.2 139 chloride 100 mM arginine-HCl 9I 20 mM acetate 0.05% Triton X-100 45 mM calcium 5.2 138 chloride 100 mM arginine-HCl 9J 20 mM acetate 0.05% Pluronic F68 45 mM calcium 5.2 139 chloride 100 mM arginine-HCl 9K 20 mM acetate 0.1% Pluronic F68 45 mM calcium 5.2 139 chloride 100 mM arginine-HCl 9L 20 mM acetate 0.4% Pluronic F68 45 mM calcium 5.2 134 chloride 100 mM arginine-HCl
[0624] To assess stability, the % acidic peak was measured by CEX-HPLC after stirring at room temperature for 0, 1, 2, and 5 days. The results are shown in FIG. 19. With the exception of the 0.005% Polysorbate 80 formulation (9A), which had a slightly higher level of % acidic peak, there was no meaningful difference due to the level of surfactant in the range tested on stability as measured by CEX-HPLC.
[0625] Stability also was assessed by measuring HMWS by SE-HPLC after stirring at room temperature for 0, 1, 2, and 5 days. The results are shown in FIG. 20. No meaningful differences in the amount of HMWS were observed in the concentration range of surfactants examined.
[0626] Stability additionally was assessed by measuring opalescence after stirring at room temperature for 0, 1, 2, and 5 days. As shown in FIG. 21, the formulations containing low levels of Polysorbate 20 or Polysorbate 80 demonstrated the highest opalescence of the formulations tested after stirring at room temperature for 5 days. In comparison, the formulations containing Pluronic F68 demonstrated the lowest opalescence of the formulations tested after stirring at room temperature for 5 days.
Example 10
Effect of Nontraditional Surfactants on Formulation Stability
[0627] Adalimumab biosimilar was prepared in a buffer containing 15 mM glutamate, 300 mM proline, pH 5.2 using the Cogent .mu.Scale TFF system described in Example 2. Surfactant stock solutions were then added to obtain a final protein concentration close to 110 mg/mL. The pH was adjusted to 5.2 using NaOH or HCl if needed. Aliquots were then filled into 5 cc glass vials for evaluation of stability at accelerated temperature. The composition of the formulation buffer, and the pH and antibody concentration of the formulations are provided in Table 10.
TABLE-US-00023 TABLE 10 Ab conc. Ref. Buffer Surfactant Excipient(s) pH (mg/mL) 10A 15 mM glutamate 0.01% benzalkonium 300 mM proline 5.2 109 chloride 10B 15 mM glutamate 0.01% guanidine HCl 300 mM proline 5.2 109 10C 15 mM glutamate 0.01% lecithin 300 mM proline 5.2 109 10D 15 mM glutamate 0.01% oleic acid 300 mM proline 5.2 109 10E 15 mM glutamate 0.01% Polysorbate 80 300 mM proline 5.2 109 10F 15 mM glutamate 0.1% polyvinyl alcohol 300 mM proline 5.2 100 205K 10G 15 mM glutamate 0.1% polyvinyl alcohol 300 mM proline 5.2 100 31K 10H 15 mMglutamate 0.01% PVP 300 mM proline 5.2 109 10I 15 mM glutamate 0.01% protamine 300 mM proline 5.2 109 sulfate
[0628] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 7, 14, and 28 days at 40.degree. C. The results are shown in FIG. 22. The amount of acidic peak formed after storage at 40.degree. C. for 28 days was found to be similar. Meaningful differences were not observed.
[0629] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 7, 14, and 28 days at 40.degree. C. The results are shown in FIG. 23. The formulations containing benzalkonium chloride and protamine sulfate (10A and 10I) appeared to have the lowest amount of HMWS after 28 days at 40.degree. C.
Example 11
Effect of PEG, Proline, and Calcium Chloride on Formulation Stability
[0630] Adalimumab biosimilar frozen in a buffer with 20 mM glutamic acid, pH 5.2 was thawed and subjected to dialysis using dialysis tubing into one of the following buffers: 20 mM calcium chloride; 10 mM lactate; 4.2% mannitol; and 14.4 mM sodium phosphate with 7.7 mM citrate, 105 mM sodium chloride and 1.2% mannitol. Stock excipient solutions were then added to achieve the final concentrations as shown in Table 11. If needed, the pH was adjusted to 5.2.+-.0.1 with MEA or MSA. Comparative formulations were also prepared and adjusted to pH 5.2.+-.0.1 with NaOH or HCl as needed. The final formulation compositions, pH, and antibody concentration are listed in Table 11.
TABLE-US-00024 TABLE 11 Buffer/pH Adjusting Ab conc. Osmolality Ref. Agent Excipient(s) Surfactant pH (mg/mL) (mOsm/kg) Comp. ---/HCl, NaOH 4.2% mannitol 0.1% 5.2 102 271 1A polysorbate 80 Comp. 7.7 mM citrate, 105 mM NaCl 0.1% 5.1 110 309 2A 14.1 mM 1.2% mannitol polysorbate 80 sodium phosphate 11A ---/MEA, 9% PEG 600 0.1% Pluronic 5.1 105 383 MSA 20 mM calcium F68 chloride 11B ---/MEA, 6.9% PEG 600 0.1% Pluronic 5.1 97 367 MSA 0.6% PEG 200 F68 20 mM calcium chloride 11C ---/MEA, 4.5% PEG 600 0.1% Pluronic 5.1 104 321 MSA 1.8% PEG 200 F68 20 mM calcium chloride 11D ---/MEA, 1.2% PEG 600 0.1% Pluronic 5.1 112 349 MSA 3% PEG 200 F68 20 mM calcium chloride 11E ---/MEA, 4% PEG 200 0.1% Pluronic 5.1 99 321 MSA 20 mM calcium F68 chloride 11F ---/MEA, 7.3% PEG 600 0.1% Pluronic 5.2 114 439 MSA 60 mM proline F68 20 mM calcium chloride 11G ---/MEA, 5.5% PEG 600 0.1% Pluronic 5.1 107 404 MSA 120 mM F68 proline 20 mM calcium chloride 11H ---/MEA, 2.5% PEG 600 0.1% Pluronic 5.2 105 410 MSA 180 mM F68 proline 20 mM calcium chloride 11I ---/MEA, 240 mM 0.1% Pluronic 5.1 109 367 MSA proline F68 20 mM calcium chloride 11J 10 mM lactate 8.4% PEG 600 0.1% Pluronic 5.1 107 379 F68 11K 10 mM lactate 4% PEG 600 0.1% Pluronic 5.1 102 307 1.6% PEG 200 F68 Comp. ---/HCl, NaOH 4.2% mannitol 0.1% 5.2 173 301 1B polysorbate 80 Comp. 7.7 mM citrate 105 mM NaCl 0.1% 5.1 174 317 2B 14.1 mM 1.2% mannitol polysorbate 80 sodium phosphate 11L 10 mM lactate 8.4% PEG 600 0.1% Pluronic 5.2 177 488 F68 11M 10 mM lactate 4% PEG 600 0.1% Pluronic 5.2 178 413 1.6% PEG 200 F68 11N 10 mM lactate 3.8% PEG 200 0.1% Pluronic 5.2 182 310 F68 11O 10 mM lactate 220 mM 0.1% Pluronic 5.2 170 389 proline F68
[0631] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 2, and 4 weeks at 40.degree. C. and at 25.degree. C. The results are shown in FIG. 24 and FIG. 25. At 25.degree. C., meaningful differences were not observed. After storage at 40.degree. C., the formulations labeled 2A and 2B appeared to have higher growth of the % acidic peak, however the other formulations tested did not show apparent differences that could be considered meaningful.
[0632] Stability also was assessed by measuring HMWS by SE-HPLC after 0, 2, and 4 weeks at 40.degree. C. and at 25.degree. C. The results are shown in FIG. 26 and FIG. 27. In formulations at a protein concentration of around 170 mg/mL, the formulation buffered with lactate and containing proline had superior stability, both at 25.degree. C. and at 40.degree. C. In formulations containing PEG 200 or PEG 600, stability was worse than the other formulations at 170 mg/mL. Formulations at approximately 100 mg/mL also showed a preference for proline in that the amount of HMWS was reduced as the proline concentration increased, a trend which was most apparent at 40.degree. C. and to a lesser extent at 25.degree. C.
[0633] Stability was assessed after freeze/thaw cycling as described in Example 3. The percentage of HMWS was measured by SE-HPLC after 0 days and after 3 F/T cycles. The results are shown in FIG. 28. In general, at around 100 mg/mL, the comp 1A and comp 2A formulations appeared to have the largest increase in HMWS compared to the other formulations examined. At 170 mg/mL, the formulation with PEG 600 and PEG 200 (11M) appeared to have the largest increase in HMWS.
Example 12
Effect of Sorbitol, Sucrose, Proline, PEG, and Calcium Chloride on Formulation Stability
[0634] Adalimumab biosimilar starting material was prepared using a Cogent .mu.Scale TFF with a 30 kD Millipore cassette and a pressure difference of about 23 psi in the following buffers: 4.2% mannitol; 14.4 mM sodium phosphate with 7.7 mM citrate, 105 mM sodium chloride and 1.2% mannitol; 7.3% sucrose with 20 mM calcium chloride; 20 mM calcium chloride; 4% sorbitol with 25 mM calcium chloride; 320 mM proline with 20 mM calcium chloride; 10 mM lactate with 225 mM proline and 20 mM calcium chloride; 10 mM lactate with 20 mM calcium chloride; 10 mM acetate with 9% sucrose. The pH was adjusted as shown in Table 12. The resulting protein was concentrated to achieve the concentration provided in Table 12, with the exception of Comp 3A which was diluted to 100 mg/mL with buffer from a bulk preparation at 170 mg/mL. The formulations are provided in Table 12. The formulations were stored at -30.degree. C.
TABLE-US-00025 TABLE 12 Buffer/pH Adjusting Ab conc. Ref. Agent Excipient(s) Surfactant pH (mg/mL) Comp. 3A -- 4.2% mannitol 0.1% 5.2 100 Polysorbate 80 Comp. 3B -- 4.2% mannitol 0.1% 5.2 170 Polysorbate 80 Comp. 4 14.1 mM sodium 105 mM NaCl 0.1% 5.2 50 phosphate 1.2% mannitol Polysorbate 80 7.7 mM citrate 12A --/MSA, MEA 7.3% sucrose 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 12B --/MSA, MEA 4.8% PEG 200 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 12C --/HCl 4% sorbitol, 0.1% Pluronic 5.2 100 25 mM calcium F68 chloride 12D --/MSA, MEA 14.5% PEG 600 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 12E 10 mM histidine/ 320 mM proline -- 6.8 100 MSA, MEA 20 mM calcium chloride 12F 10 mM lactate/ 225 mM proline 0.1% Pluronic 5.2 170 MSA, MEA 20 mM calcium F68 chloride 12G 10 mM lactate/ 13% PEG 600 0.1% Pluronic 5.2 170 MSA, MEA 20 mM calcium F68 chloride 12H 10 mM acetate/ 9% sucrose 0.1% 5.2 170 MSA, MEA Polysorbate 80
[0635] To assess stability, the % acidic peak was measured by CEX-HPLC after 0 days, after transport, and after storing the transported sample at 40.degree. C. for 2 weeks. The results are shown in FIG. 29. After 2 weeks at 40.degree. C., the lowest amount of % acidic peak was found in the formulation buffered with lactate and containing calcium chloride (12F). Otherwise, formulations buffered with histidine, containing PEG 600 at a high concentration or the formulation buffered with sodium phosphate were less stable (12E, 12D, Comp 4). The antibody concentration of each formulation, i.e. whether at 50, 100 or 170 mg/mL did not appear to influence the amount of % acidic peak.
[0636] Stability also was assessed by measuring HMWS by SE-HPLC after 0 days, after transport, and after storing the transported sample at 40.degree. C. for 2 weeks. The results are shown in FIG. 30. The lower the concentration, the lower the amount of HMWS. This is shown in comparing Comp 3B, Comp 3A and Comp 4 in which as the concentration changes from 170 mg/mL to 100 mg/mL to 50 mg/mL the HMWS is correspondingly reduced. For the other formulations tested, at 170 mg/mL, there are differences in the amount of HMWS after two weeks at 40.degree. C. The lowest amount of HMWS was measured for the acetate formulation with sucrose (12H), followed by the lactate buffered formulation with proline (12F) and finally the formulation with the high amount of PEG 600 (12G). In formulations at 100 mg/mL, the lowest HMWS was found in the self-buffering formulation with sorbitol and calcium chloride (12C). The addition of PEG 200 resulted in more HMWS at 100 mg/mL (12B).
[0637] Stability was further assessed by MFI. The results are shown in FIGS. 31-36. High particle counts at 5 .mu.M were measured in the formulation containing proline and calcium chloride after transport stress (12E). In general, particle counts were higher at 100 mg/mL than at 170 mg/mL at the 5 .mu.M particle size. At 100 mg/mL, formulation Comp 3A and the self-buffered formulation with sorbitol had lower particle counts at 5 .mu.M post transport than the other formulations tested at this concentration (compare 12C and Comp 3A to 12A, 12B, 12D and 12E). The particle count trends observed at 5 .mu.M were also observed at the 10 .mu.M particle size. The lowest particle counts at 10 .mu.M were measured at 170 mg/mL. At 100 mg/mL, a low particle count increase post transport was also observed in the self-buffered formulation with sorbitol (12C) and in Comp 3A. Finally, the particle count trends reported for the 5 .mu.M and 10 particle sizes were also observed at 25 .mu.M. The lowest particle counts were again at the 25 size. At 100 mg/mL, the lowest particle counts post transport were measured in formulations Comp 3A, 12A and 12C.
Example 13
Effect of PEG, Proline, and Calcium Chloride on Formulation Stability
[0638] Adalimumab biosimilar starting material at 200 mg/mL was diluted to 180 mg/mL and then subjected to dialysis using 3 kD cutoff dialysis tubing in the following buffers: 20 mM calcium chloride, pH adjusted with MSA or MEA; 14.4 mM sodium phosphate with 7.7 mM citrate, 105 mM sodium chloride and 1.2% mannitol, pH adjusted with HCl or NaOH to a final pH of 4.8; 10 mM lactate with 20 mM calcium chloride, pH adjusted with MSA or MEA; and 4.2% mannitol, pH adjusted with MSA or MEA. Stock excipient solutions were then added to achieve the final concentrations as shown in Table 13 and the pH was adjusted to 5.2 if needed. The formulations are provided in Table 13.
TABLE-US-00026 TABLE 13 Buffer/pH Adjusting Ab conc. Ref. Agent Excipient(s) Surfactant pH (mg/mL) Comp. 5A --/HCl, NaOH 4.2% mannitol 0.1% 5.2 100 Polysorbate 80 Comp. 6A 14.1 mM sodium 105 mM NaCl 0.1% 5.2 100 phosphate 1.2% mannitol Polysorbate 80 7.7 mM citrate/ HCl, NaOH 13A --/MSA, MEA 9% PEG600 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 13B --/MSA, MEA 6.9% PEG600 0.1% Pluronic 5.2 100 0.6% PEG200 F68 20 mM calcium chloride 13C --/MSA, MEA 4.5% PEG600 0.1% Pluronic 5.2 100 1.8% PEG200 F68 20 mM calcium chloride 13D --/MSA, MEA 1.2% PEG600 0.1% Pluronic 5.2 100 3% PEG200 F68 20 mM calcium chloride 13E --/MSA, MEA 4% PEG200 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 13F --/MSA, MEA 7.3% PEG600 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 60 mM proline 13G --/MSA, MEA 5.5% PEG600 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 120 mM proline 13H --/MSA, MEA 2.5% PEG600 0.1% Pluronic 5.2 100 20 mM calcium F68 chloride 180 mM proline 131 --/MSA, MEA 20 mM calcium 0.1% Pluronic 5.2 100 chloride F68 240 mM proline 13J 10 mM lactate/ 20 mM calcium 0.1% Pluronic 5.2 100 MSA, MEA chloride F68 8.4% PEG 600 13K 10 mM lactate/ 20 mM calcium 0.1% Pluronic 5.2 100 MSA, MEA chloride F68 4% PEG 600, 1.6% PEG 200 Comp. 5B --/HCl, NaOH 4.2% mannitol 0.1% 5.2 170 Polysorbate 80 Comp. 6B 14.1 mM sodium 105 mM NaCl 0.1% 5.2 170 phosphate 1.2% mannitol Polysorbate 80 7.7 mM citrate/ HCl, NaOH 13L 10 mM lactate/ 8.4% PEG600 0.1% Pluronic 5.2 170 MSA, MEA 20 mM calcium F68 chloride 13M 10 mM lactate/ 4% PEG600 0.1% Pluronic 5.2 170 MSA, MEA 1.6% PEG200 F68 20 mM calcium chloride 13N 10 mM lactate/ 3.8% PEG200 0.1% Pluronic 5.2 170 MSA, MEA 20 mM calcium F68 chloride 13O 10 mM lactate/ 20 mM calcium 0.1% Pluronic 5.2 170 MSA, MEA chloride F68 220 mM proline
[0639] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 2, and 4 weeks at 40.degree. C. and at 25.degree. C. The results are shown in FIG. 37 and FIG. 38. At 25.degree. C., after 4 weeks, the lowest % acidic peak was found in the formulation containing proline and calcium chloride (13I) and the formulation with PEG 200 and PEG 600 and calcium chloride (13D). Lower growth was also observed in formulations with a mixture of PEG 600, proline and calcium chloride (13H) and in the formulation buffered with sodium phosphate (Comp 6A). In formulations at a concentration of 170 mg/mL, the lowest % acidic peak after storage for 4 weeks at 25.degree. C. was found in the lactate buffer formulation with proline (13O) and in the sodium phosphate buffered formulation (Comp 6B). After 4 weeks at 40.degree. C., meaningful differences between formulations were not as apparent. The highest amount of % acidic peak found after 4 weeks at 40.degree. C. were in the formulations buffered with sodium phosphate at both 100 and 170 mg/mL (Comp 6A, Comp 6B respectively.)
[0640] Stability was assessed by measuring HMWS by SE-HPLC after 0, 2, and 4 weeks at 40.degree. C. and at 25.degree. C. The results are shown in FIG. 39 and FIG. 40. At 25.degree. C., after 4 weeks, the formulation with proline and calcium chloride had the lowest HMWS (13I). Otherwise, increasing concentrations of PEG 600 and lower proline levels in formulations resulted in higher amounts of HMWS (compare 13I, 13H, 13G and 13F). In formulations at 170 mg/mL stored for 4 weeks at 25.degree. C., the proline formulation with calcium chloride was again superior in having the lowest HMWS (13O). At 40.degree. C., similar trends to those observed at 25.degree. C. were noted. The proline formulations had the lowest amount of HMWS at both 100 mg/mL and 170 mg/mL (13I, 13O). At 4.degree. C., stability was assessed by measuring HMWS by SE-HPLC after 0, 4, and 8 weeks. The results are shown in FIG. 41. Minimal growth of HMWS is observed at 4.degree. C. in most formulations. At time zero, the lowest HMWS in the 100 mg/mL formulation is found in the proline formulation with calcium chloride (13I). Likewise, at 170 mg/mL, HMWS is minimized in the lactate buffer formulation with proline at time zero (13O). Finally, stability was assessed by measuring HMWS by SE-HPLC after 0 weeks, after 3 F/T cycles, and after 8 weeks at -30.degree. C. The results are shown in FIG. 42. In agreement with the results obtained at 4.degree. C., the proline formulation at 100 and 170 mg/mL (13I, 13O) had the lowest amount of HMWS and no meaningful change in HMWS after the freeze thaw cycles. Most formulations did not show meaningful change upon repeated freezing and thawing at -30.degree. C. The lone exception appears to be the Comp 5A formulation, in which HMWS increased upon repeated freezing and thawing and after 8 weeks at -30.degree. C.
Example 14
Effect of PEG, Proline, MEA, MSA and Calcium Chloride on Formulation Stability
[0641] Adalimumab biosimilar starting material at 50 mg/mL was prepared using 3.5 kD cutoff dialysis tubing in the following buffers: 5 mM MEA with 5 mM MSA, pH adjusted using MSA or MEA to a final pH of 4.8; 14.4 mM sodium phosphate with 7.7 mM citrate, 105 mM sodium chloride and 1.2% mannitol, pH adjusted with HCl or NaOH to a final pH of 4.8; 4.2% mannitol, pH adjusted with HCl or NaOH to a final pH of 4.8; 10 mM acetate with 9% sucrose, pH adjusted with HCl or NaOH to a final pH of 4.8. Stock excipient solutions were then added to achieve the final concentrations as shown in Table 14 and the pH was adjusted to 5.2 if needed. The formulations are provided in Table 14.
TABLE-US-00027 TABLE 14 pH Adjusting Ab conc. Ref. Buffer Excipient(s) Agent pH (mg/mL) Comp. 7 14.1 mM sodium 105 mM NaCl HCl, NaOH 5.2 50 phosphate 1.2% mannitol 7.7 mM citrate Comp. 8 -- 4.2% mannitol HCl, NaOH 5.2 50 14A 10 mM acetate 9% sucrose HCl, NaOH 5.2 50 14B -- 10.4% PEG600 MSA, MEA 5.2 50 14C -- 8.5% PEG600 MSA, MEA 5.2 50 25 mM calcium chloride 14D -- 6.4% PEG600 MSA, MEA 5.2 50 50 mM calcium chloride 14E -- 3.7% PEG600 MSA, MEA 5.2 50 75 mM calcium chloride 14F -- 100 mM calcium MSA, MEA 5.2 50 chloride 14G -- 300 mM proline MSA, MEA 5.2 50 14H -- 225 mM proline MSA, MEA 5.2 50 25 mM calcium chloride 14I -- 150 mM proline MSA, MEA 5.2 50 50 mM calcium chloride 14J -- 75 mM proline MSA, MEA 5.2 50 75 mM calcium chloride 14K -- 240 mM proline MSA, MEA 5.2 50 60 mM MEA 60 mM MSA 14L -- 180 mM proline MSA, MEA 5.2 50 60 mM MEA 60 mM MSA 14M -- 120 mM proline MSA, MEA 5.2 50 90 mM MEA 90 mM MSA
[0642] To assess stability, the % acidic peak was measured by CEX-HPLC after 0, 2, and 4 weeks at 40.degree. C. and after 0, 2, 4, and 8 weeks at 25.degree. C. The results are shown in FIG. 43 and FIG. 44. At 25.degree. C., after 8 weeks, lower amounts of HMWS were measured in the formulation with proline and high MEA and MSA (14M) and in the formulations with the higher concentrations of calcium chloride (14E, 14F and 14J). As is shown by comparing formulations 14B, 14C, 14D, 14E and 14F, the level of HMWS increased as the concentration of PEG 600 increased after 8 weeks at 25.degree. C. Similar trends were observed at 40.degree. C. that had been apparent at 25.degree. C. Increasing levels of calcium chloride were beneficial in minimizing the formation of HMWS. PEG 600 was not preferred for stability at 40.degree. C. Low HMWS was observed in the formulation containing proline and high MEA and MSA after 4 weeks at 40.degree. C. Stability was assessed by measuring acidic peak by CEX-HPLC after 0, 4 and 8 weeks at 4.degree. C. The results are shown in FIG. 45. Minor differences were measured in the % acidic peak at time zero and after storage for 8 weeks at 4.degree. C. These differences were not considered meaningful. Stability was assessed by measuring acidic peak by CEX-HPLC after 0 days, after 3 F/T cycles, and after 4 weeks at -30.degree. C. The results are shown in FIG. 46. Consistent with the results obtained at 4.degree. C., differences between formulations were minor and apparent growth of the % acidic peak was not observed after multiple freeze thaws and 8 weeks storage at -30.degree. C.
[0643] Stability also was assessed by measuring HMWS by SE-HPLC after 0 days and after 3 F/T cycles. The results are shown in FIG. 47. For most formulations, multiple freezing and thawing did not result in growth of HMWS, with the exception of formulation Comp 8. Several formulations, although not showing an increase in HMWS after multiple freeze thaw steps, did grow in HMWS after storage at -30.degree. C. following the freeze thaw steps (formulations Comp. 8, self-buffered, and 14M, containing high amounts of MEA and MSA). Stability was assessed by measuring HMWS by SE-HPLC after 0 and 2 weeks at 40.degree. C. and at 25.degree. C. The results are shown in FIG. 48 and FIG. 49. After storage at 40.degree. C. for 4 weeks, HMWS growth appeared to be higher in formulations containing PEG 600 or higher amounts of calcium chloride and in the formulation buffered with sodium phosphate (Comp 7, 14B, 14C, 14D, 14E, 14F). Overall, the highest amount of HMWS at >3% was measured in the formulation with higher MEA and MSA (14M). Low amounts of HMWS were observed in the formulation buffered with acetate and the proline formulation (14A, 14G). Similar trends were observed at 25.degree. C. as were found at 40.degree. C. Low HMWS were apparent in formulations 14A and 14G, with PEG 600 not preferred for stability, along with high levels of MEA and MSA. Finally, stability was also assessed at 4.degree. C. by SE-HPLC after 0, 4 and 8 weeks. The results are shown in FIG. 50. Meaningful growth of HMWS at 4.degree. C. was not observed in most formulations, with the exception of the formulations containing higher amounts of PEG 600 (14B, 14C). As discussed previously, at time zero the highest HMWS was observed in the formulation containing high levels of MEA and MSA.
Example 15
Stability of Self-Buffered, Lactate Buffered, and Acetate Buffered Formulations
[0644] Three adalimumab formulations were prepared, as shown in Table 15.
TABLE-US-00028 TABLE 15 Buffer/pH Adjusting Ab conc. Ref. Agent Excipients Surfactant pH (mg/mL) Conductivity 15A --/HCl, 4% 0.05% Pluronic 5.2 100 4.7 mS/cm Ca(OH).sub.2 sorbitol, 30 mM F68 calcium chloride 15B 10 mM 6% 0.006% 5.1 100 4.78 mS/cm Lactate/HCl, sucrose, Pluronic F68 Ca(OH).sub.2 30 mM calcium chloride 15C 10 mM 6% 0.1% 5.2 100 4.05 mS/cm Acetate/HCl, sucrose, 45 mM Polysorbate 80 NaOH NaCl
[0645] To assess stability, the % acidic peak was measured by CEX-HPLC at 0 days, 1 week, 2 weeks, 1 month, and 2 months, at 2-8.degree. C., and at 0 days, 1 week, 2 weeks, 1 month, and 2 months at 25.degree. C. The results are shown in FIG. 51 (2-8.degree. C.) and FIG. 52 (25.degree. C.). All three formulation (formulations 15A-C) exhibited similar stability with respect to acidic species at 2-8.degree. C. At 25.degree. C., formulation 15C (acetate buffer with sodium chloride) had the highest amount of % acidic peak.
[0646] Stability also was assessed by measuring HMWS by SE-HPLC after 0 days, 1 week, 2 weeks, 1 month, and 2 months at 2-8.degree. C., and after 0 days, 1 week, 2 weeks, 1 month, and 2 months at 25.degree. C. The results are shown in FIGS. 53 (2-8.degree. C.) and 54 (25.degree. C.). At time zero, the highest HMWS was measured in the self-buffered formulation, followed by the acetate buffer formulation with sodium chloride and the lactate buffer formulation with calcium chloride, respectively. This trend was maintained at 4.degree. C. over time, with the lactate buffer formulation having the least amount of HMWS. Likewise, at 25.degree. C., the lactate buffer formulation exhibited the least growth of HMWS. At the 1 and 2 month time point, the rate of degradation also slowed down compared to the earlier time points at 25.degree. C. This trend was observed for all formulations at 25.degree. C. At 4.degree. C., the rate of growth of HMWS were similar for each formulation, with a minor increase observed in all formulations after time zero and up to two weeks, followed by a leveling off of the rate after the two week time point. The lactate buffer formulation also had the lowest amount of HMWS at time zero.
[0647] Stability also was assessed by measuring the count of 5 .mu.M, 10 .mu.M, and 25 .mu.M sub-visible particles by MFI in non-transported and transported samples at 2-8.degree. C. for 1, 2, and 4 weeks. The particles exhibited an equivalent circular diameter of at least 5.000 and an aspect ratio of less than 0.700. The results are shown in FIGS. 55-57. All formulations at .ltoreq.25 .mu.M had low particle counts. For the 10 .mu.M sub-visible counts, the acetate formulation (formulation 15B) showed the lowest amount of sub-visible particles. For the non-spherical .ltoreq.5 .mu.M particle counts, the self-buffered and lactate buffer formulations (formulations 15A and 15C) showed higher particle counts at t=0 (pre-transport) and at later time points compared to the acetate formulation with sodium chloride. In general, for .ltoreq.5 .mu.M particle counts, each formulation showed an increase in particles post-transport, followed by a trend of lower particle counts at the 1 week, 2 week and 4 week time points.
Example 16
Effect of Surfactant and Salt on Formulation Stability
[0648] Several adalimumab formulations (formulations 16A-16L) were prepared, as shown in Table 16.
TABLE-US-00029 TABLE 16 Buffer/pH Conductivity Adjusting CaCl.sub.2 Ab conc. Osmolality Ref. Agent (mM) Excipient Surfactant pH (mg/mL) Viscosity 16A --/-- 0 4% 0.09% 5.21 97.89 0.76 mS/cm sorbitol Polysorbate 20 241 mOsm 2.54 mPa s 16B --/60 .mu.L 15 4% 0.09% 5.09 99.37 3.01 mS/cm Ca(OH).sub.2 sorbitol Polysorbate 20 283 mOsm 2.59 mPa s 16C --/40 .mu.L 30 4% 0.03% 5.06 103.47 4.98 mS/cm Ca(OH).sub.2 sorbitol Polysorbate 20 317 mOsm 2.62 mPa s 16D --/40 .mu.L 15 4% 0.03% 5.1 99.99 3.07 mS/cm Ca(OH) sorbitol Polysorbate 20 283 mOsm 2.46 mPa s 16E --/60 .mu.L 30 4% 0.09% 5.03 103.77 4.97 mS/cm Ca(OH).sub.2 sorbitol Polysorbate 20 316 mOsm 2.54 mPa s 16F 10 mM 0 6% sucrose 0.10% Pluronic 5.25 97.68 0.744 mS/cm Lactate/-- F68 219 mOsm 2.82 mPa s 16G 10 mM 15 6% sucrose -- 5.09 111.79 2.99 mS/cm Lactate/-- 268 mOsm 3.03 mPa s 16H 10 mM 15 6% sucrose 0.08% Pluronic 5.09 106.72 2.97 mS/cm Lactate/-- F68 266 mOsm 3.00 mPa s 16I 10 mM 15 6% sucrose 0.02% Pluronic 5.06 109.35 2.99 mS/cm Lactate/-- F68 268 mOsm 2.93 mPa s 16J 10 mM 30 6% sucrose 0.2% Pluronic 5.04 101.79 4.99 mS/cm Lactate/-- F68 305 mOsm 2.75 mPa s 16K 10 mM 30 6% sucrose 0.08% Pluronic 5.05 102.27 5.03 mS/cm Lactate/-- F68 309 mOsm 2.70 mPa s 16L 10 mM 30 6% sucrose 0.03% Pluronic 5.04 104.89 5.06 mS/cm Lactate/-- F68 308 mOsm 2.68 mPa s
[0649] To assess stability, the % acidic peak was measured by CEX-HPLC in non-transported samples and transported samples at 0, 1, and 2 weeks at 4.degree. C., 25.degree. C., and 40.degree. C. The results are shown in FIGS. 58-63. No meaningful differences in the % acidic peak were exhibited at time 0 for all twelve formulations. The lack of meaningful differences between formulations was also apparent after 1 month at 4.degree. C. for both transport stressed and non-transport stressed conditions. At 25.degree. C., and 40.degree. C., as a general trend, formulations without calcium chloride (16A and 16F) had the highest amount of % acidic peak after 1 month. In addition, differences between transported stressed and non-transported stressed formulations were not apparent at 25.degree. C. At 40.degree. C., the non-transported formulations that were self-buffered and containing calcium chloride (16B-16E) had lower rates of growth of % acidic peak compared to the identical formulations that were transport stressed. Otherwise, the presence of calcium chloride appears to reduce the formation of the acidic peak at both 25.degree. C. and at 40.degree. C.
[0650] Stability also was assessed by measuring HMWS by SE-HPLC after 0 days, after transport, and after storing the non-transported and transported samples at 4.degree. C., 25.degree. C., or 40.degree. C. for 1, 2, and 4 weeks. The results are shown in FIGS. 64-69. The formulations having the combination of lactate buffer and calcium chloride (e.g., formulations 16G-16L), versus self-buffered and formulations with sorbitol (formulations 16A-E), exhibited the lowest amount of HMWS at time 0. Formulations having lactate buffer exhibited better stability than formulations without lactate buffer at 25.degree. C. Formulations having 15 mM of calcium chloride (formulations 16G-16I) exhibited better stability than formulations having 0 mM or 30 mM of calcium chloride. This trend was more apparent in the self-buffered formulations (16A-16E) in which 15 mM calcium chloride was superior for stability at 25.degree. C. compared to self-buffered formulations with 30 mM calcium chloride. Overall, formulations having the combination of lactate buffer and calcium chloride exhibited surprisingly superior stability at 25.degree. C. After storage for 1 month at 40.degree. C., the growth of HMWS appeared to increase faster in the lactate buffer formulations compared to the self-buffered formulations, however the amount of HMWS formed did not exceed that of the HMWS measured in the self-buffered formulations at this same temperature and time point. In general, formulations having the combination of lactate buffer and 15 mM calcium chloride exhibited the lowest amount of HMWS, in part, because they initially had the lowest amount of HMWS.
[0651] Stability also was assessed by measuring the count of 5 .mu.M, 10 .mu.M, and 25 .mu.M sub-visible particles by MFI in non-transported and transported samples at 4.degree. C., 25.degree. C., or 40.degree. C. for 1, 2, and 4 weeks. The particles exhibited an equivalent circular diameter of at least 5.000 and an aspect ratio of less than 0.700. The results are shown in FIGS. 70-72. For the 5 .mu.M size particles, the lactate buffered formulation with 0.25% Pluronic F68 had the highest number of particles following transport and at later time points. In the self-buffered formulations, the absence of calcium chloride resulted likewise in a high 5 .mu.M particle count initially and over time. These results were not expected, especially with the higher level of Pluronic F68 in the lactate buffer formulation associated with high particle counts. In the lactate buffer formulations in general at 5 .mu.M, 0.13% Pluronic F68 appeared best at minimizing particles in formulations with either 15 mM or 30 mM calcium chloride. FIGS. 70A-B. At the 10 .mu.M particle level, a similar trend to that observed for the 5 .mu.M results was observed. The lactate buffer formulation with 0.13% Pluronic F68 was effective in reducing the number of particles compared to higher Pluronic F68 at 0.25%. FIGS. 71A-B. Finally, at 25 Pluronic F68 at 0.13% minimized particle growth in lactate buffer formulations. Calcium chloride at concentrations of either 15 or 30 mM minimized particle growth in self-buffered formulations in the presence of PS 20. FIGS. 72A-B.
Example 17
Effect of Calcium Chloride Concentration on the Stability of Lactate Buffer Formulations
[0652] Several adalimumab formulations (formulations 17A-17E) were prepared, as shown in Table 17.
TABLE-US-00030 TABLE 17 Buffer/pH Adjusting CaCl.sub.2 Ab conc. Conductivity Ref. Agent (mM) Excipient Surfactant pH (mg/mL) Osmolality 17A 10 mM 5 8.3% 0.03% Pluronic 5.11 100 1.339 mS/cm Lactate/HCl, sucrose F68 292 mOsm Ca(OH).sub.2 17B 10 mM 10 7.8% 0.03% Pluronic 5.12 100 2.125 mS/cm Lactate/HCl, sucrose F68 304 mOsm Ca(OH).sub.2 17C 10 mM 15 7.4% 0.03% Pluronic 5.10 100 2.792 mS/cm Lactate/HCl, sucrose F68 304 mOsM Ca(OH).sub.2 17D 10 mM 20 6.9% 0.03% Pluronic 5.13 100 3.527 mS/cm Lactate/HCl, sucrose F68 302 mOsM Ca(OH).sub.2 17E 10 mM -- mM 8.8% 0.03% Pluronic 5.23 100 0.534 mS/cm Lactate/HCl, sucrose F68 314 mOsM Ca(OH).sub.2
[0653] To assess stability, the % acidic peak was measured by CEX-HPLC after 0 days, after transport, and after 1 week, 2 weeks and 4 weeks at 4.degree. C., 25.degree. C. and 40.degree. C. The results are shown in FIGS. 73-78. At time zero, the % acidic peak was similar either with or without transport stress, with only minor variability observed. After 1 month at 25.degree. C., the rate of growth was similar in all formulations, with the level of calcium chloride in the range evaluated in this study having minor to no impact, tested in both transport stressed and non-transport stressed samples. In formulations stored at 40.degree. C., a smaller amount of % acidic peak was observed as the level of calcium chloride increased after 1 month's storage. This trend was observed for both the transport and non-transport stressed formulations. Otherwise, transport stress did not appear to result in higher rates of degradation.
[0654] Stability also was assessed by measuring HMWS by SE-HPLC after 0 days, after transport, and after storing the non-transported and transported samples at 4.degree. C., 25.degree. C., or 40.degree. C. for 1, 2, and 4 weeks. The results are shown in FIGS. 79-84. At time zero, a very minor increase in HMWS was observed as the calcium chloride level increased. This is most likely not meaningful, considering assay variability. Formulations that were transport stressed did not appear to have higher HMWS than non-transport stressed formulations. After one month at 25.degree. C. and at 40.degree. C., it is difficult to detect a correlation in formulations with a higher amount of HMWS and increasing amounts of calcium chloride. These results suggest that levels of calcium chloride in the range tested do not adversely affect stability with respect to the formation of HMWS.
[0655] Stability also was assessed by measuring the count of 5 .mu.M, 10 .mu.M, and 25 .mu.M sub-visible particles by MFI in non-transported and transported samples. The particles exhibited an equivalent circular diameter of at least 5.000 and an aspect ratio of less than 0.700. The results are shown in FIGS. 85-87. In formulations tested for particle counts at .ltoreq.5 .mu.m, counts appeared to spike after transport stress, however the particle counts dropped at all time points and temperature conditions thereafter. High temperature exposure (25.degree. C., 40.degree. C.) resulted in only a modest increase in particles. There was a less apparent spike in particles .ltoreq.10 .mu.m in most formulations that had been transport stressed, but the counts were also reduced at the 1 month time point at all temperatures tested. A modest particle count increase was also observed in samples stored after 1 month at 25.degree. C. and 40.degree. C. MFI results measured for particles in the range .ltoreq.25 .mu.m did not show an apparent trend, as the number of particles was low or not detected.
[0656] All publications, patents and patent applications cited in this specification are herein incorporated by reference as if each individual publication or patent application were specifically and individually indicated to be incorporated by reference. Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, it will be readily apparent to those of ordinary skill in the art in light of the teachings of this disclosure that certain changes and modifications may be made thereto without departing from the spirit or scope of the disclosed embodiments.
Sequence CWU 1
1
101107PRTArtificial SequenceLight chain variable region 1Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Gly Ile Arg Asn Tyr 20 25
30Leu Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45Tyr Ala Ala Ser Thr Leu Gln Ser Gly Val Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Val Ala Thr Tyr Tyr Cys Gln Arg Tyr Asn
Arg Ala Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 1052121PRTArtificial SequenceHeavy chain variable region 2Glu
Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Arg1 5 10
15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Asp Asp Tyr
20 25 30Ala Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp
Val 35 40 45Ser Ala Ile Thr Trp Asn Ser Gly His Ile Asp Tyr Ala Asp
Ser Val 50 55 60Glu Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn
Ser Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Lys Val Ser Tyr Leu Ser Thr Ala Ser
Ser Leu Asp Tyr Trp Gly 100 105 110Gln Gly Thr Leu Val Thr Val Ser
Ser 115 1203214PRTArtificial SequenceFull length light chain 3Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Gly Ile Arg Asn Tyr
20 25 30Leu Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu
Ile 35 40 45Tyr Ala Ala Ser Thr Leu Gln Ser Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Val Ala Thr Tyr Tyr Cys Gln Arg Tyr
Asn Arg Ala Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro
Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys
Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp
Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu
Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170
175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr
180 185 190Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr
Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys 2104224PRTArtificial
SequenceFull length heavy chain 4Glu Val Gln Leu Val Glu Ser Gly
Gly Gly Leu Val Gln Pro Gly Arg1 5 10 15Ser Leu Arg Leu Ser Cys Ala
Ala Ser Gly Phe Thr Phe Asp Asp Tyr 20 25 30Ala Met His Trp Val Arg
Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Ser Ala Ile Thr Trp
Asn Ser Gly His Ile Asp Tyr Ala Asp Ser Val 50 55 60Glu Gly Arg Phe
Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu Tyr65 70 75 80Leu Gln
Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala
Lys Val Ser Tyr Leu Ser Thr Ala Ser Ser Leu Asp Tyr Trp Gly 100 105
110Gln Gly Thr Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser
115 120 125Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
Thr Ala 130 135 140Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu
Pro Val Thr Val145 150 155 160Ser Trp Asn Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe Pro Ala 165 170 175Val Leu Gln Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val Thr Val 180 185 190Pro Ser Ser Ser Leu
Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His 195 200 205Lys Pro Ser
Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys Ser Cys 210 215
220511PRTArtificial SequenceL-CDR1 5Arg Ala Ser Gln Gly Ile Arg Asn
Tyr Leu Ala1 5 1067PRTArtificial SequenceL-CDR2 6Ala Ala Ser Thr
Leu Gln Ser1 579PRTArtificial SequenceL-CDR3MISC_FEATURE(9)..(9)Xaa
is Thr or Ala 7Gln Arg Tyr Asn Arg Ala Pro Tyr Xaa1
585PRTArtificial SequenceH-CDR1 8Asp Tyr Ala Met His1
5917PRTArtificial SequenceH-CDR2 9Ala Ile Thr Trp Asn Ser Gly His
Ile Asp Tyr Ala Asp Ser Val Glu1 5 10 15Gly1012PRTArtificial
SequenceH-CDR3MISC_FEATURE(12)..(12)Xaa is Tyr or Asn 10Val Ser Tyr
Leu Ser Thr Ala Ser Ser Leu Asp Xaa1 5 10
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

D00026

D00027

D00028

D00029

D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037

D00038

D00039

D00040

D00041

D00042

D00043

D00044
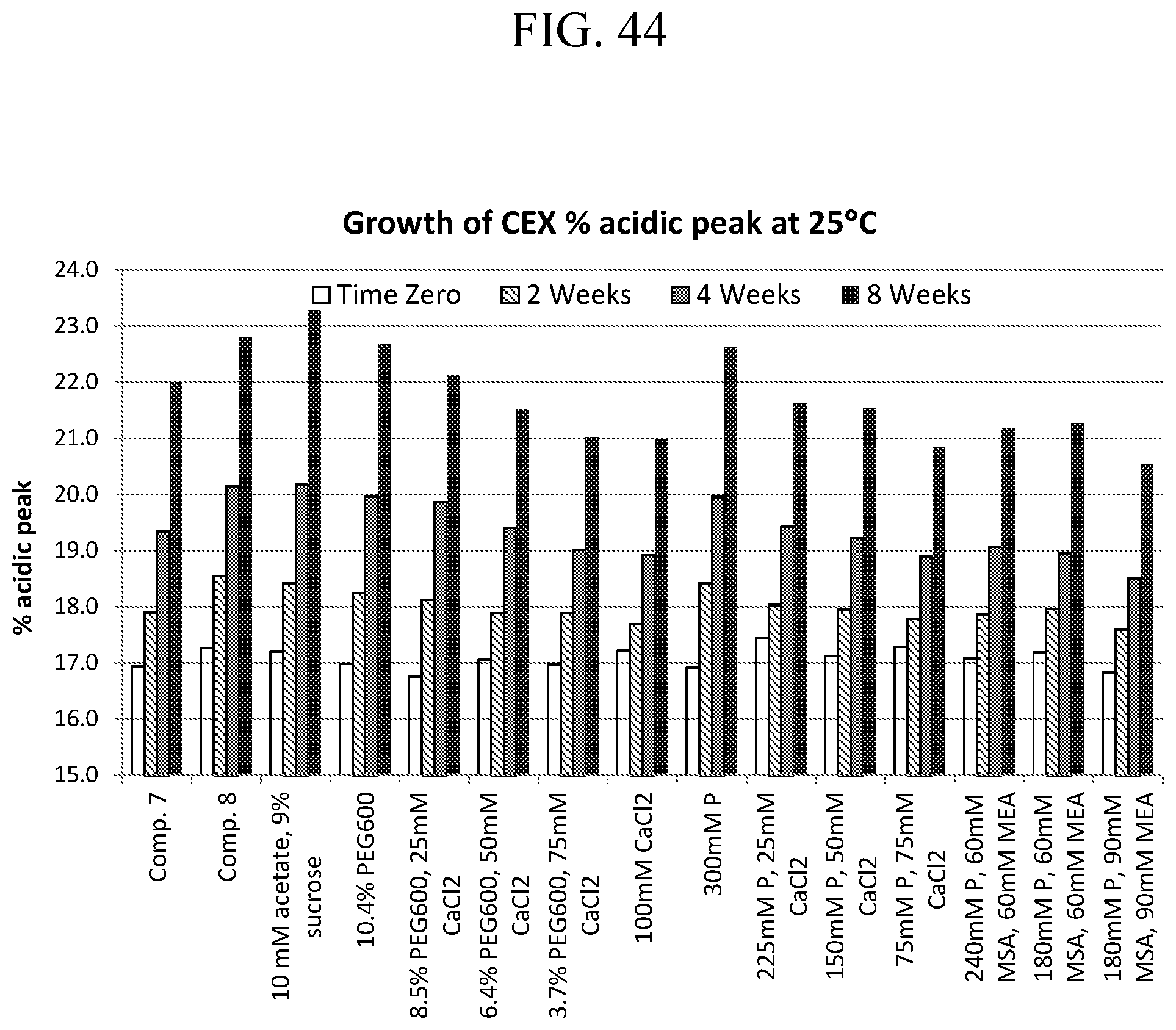
D00045

D00046

D00047

D00048

D00049

D00050

D00051

D00052

D00053

D00054

D00055

D00056

D00057

D00058

D00059

D00060

D00061

D00062

D00063

D00064

D00065

D00066

D00067

D00068

D00069

D00070

D00071

D00072

D00073

D00074

D00075

D00076

D00077

D00078

D00079

D00080

D00081

D00082

D00083

D00084

D00085

D00086

D00087

D00088

D00089

D00090

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.