Formulations Of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) Cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic Acid
Verwijs; Marinus Jacobus
U.S. patent application number 16/523493 was filed with the patent office on 2020-03-19 for formulations of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid. This patent application is currently assigned to Vertex Pharmaceuticals Incorporated. The applicant listed for this patent is Vertex Pharmaceuticals Incorporated. Invention is credited to Marinus Jacobus Verwijs.
| Application Number | 20200085750 16/523493 |
| Document ID | / |
| Family ID | 47679064 |
| Filed Date | 2020-03-19 |










View All Diagrams
| United States Patent Application | 20200085750 |
| Kind Code | A1 |
| Verwijs; Marinus Jacobus | March 19, 2020 |
FORMULATIONS OF 3-(6-(1-(2,2-DIFLUOROBENZO[D][1,3]DIOXOL-5-YL) CYCLOPROPANECARBOXAMIDO)-3-METHYLPYRIDIN-2-YL)BENZOIC ACID
Abstract
A pharmaceutical composition comprising Compound 1, (3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid), and at least one excipient selected from: a filler, a disintegrant, a surfactant, a binder, and a lubricant, the composition being suitable for oral administration to a patient in need thereof to treat a CFTR mediated disease such as Cystic Fibrosis. Processes of preparing pharmaceutical compositions comprising Compound 1 are also disclosed.
| Inventors: | Verwijs; Marinus Jacobus; (Framingham, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Vertex Pharmaceuticals
Incorporated Boston MA |
||||||||||
| Family ID: | 47679064 | ||||||||||
| Appl. No.: | 16/523493 | ||||||||||
| Filed: | July 26, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15655354 | Jul 20, 2017 | |||
| 16523493 | ||||
| 15093582 | Apr 7, 2016 | |||
| 15655354 | ||||
| 13750069 | Jan 25, 2013 | |||
| 15093582 | ||||
| 61708691 | Oct 2, 2012 | |||
| 61691898 | Aug 22, 2012 | |||
| 61651218 | May 24, 2012 | |||
| 61590479 | Jan 25, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 1/18 20180101; A61P 11/00 20180101; A61P 25/28 20180101; A61P 21/04 20180101; A61K 9/1623 20130101; A61P 25/00 20180101; A61K 9/2866 20130101; A61P 11/06 20180101; A61K 31/47 20130101; A61P 5/14 20180101; A61P 35/00 20180101; A61P 3/00 20180101; A61P 7/10 20180101; A61P 5/18 20180101; A61P 15/10 20180101; A61K 45/06 20130101; A61P 25/08 20180101; A61K 9/1652 20130101; A61P 1/10 20180101; A61P 25/14 20180101; A61P 21/02 20180101; A61K 9/2077 20130101; A61P 19/08 20180101; A61J 3/10 20130101; A61P 27/02 20180101; A61K 31/443 20130101; A61P 7/00 20180101; A61P 9/04 20180101; A61P 11/02 20180101; A61P 3/10 20180101; A61K 9/28 20130101; A61P 19/10 20180101; A61P 25/16 20180101; A61K 31/4709 20130101; A61P 1/16 20180101; A61P 3/06 20180101; A61P 43/00 20180101; A61K 9/2054 20130101; A61K 31/443 20130101; A61K 2300/00 20130101; A61K 31/4709 20130101; A61K 2300/00 20130101; A61K 31/47 20130101; A61K 2300/00 20130101 |
| International Class: | A61K 9/28 20060101 A61K009/28; A61K 31/443 20060101 A61K031/443; A61K 45/06 20060101 A61K045/06; A61J 3/10 20060101 A61J003/10; A61K 9/16 20060101 A61K009/16; A61K 9/20 20060101 A61K009/20; A61K 31/47 20060101 A61K031/47; A61K 31/4709 20060101 A61K031/4709 |
Claims
1-54. (canceled)
55. A continuous process for preparing a tablet comprising 3-(6-(1-(2,2-Difluorobenzo[d][1,3 ]dioxol-5-yl)cycl opropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (`Compound 1`) Form I, comprising the steps of: a) mixing Compound 1 Form I, a filler, and a disintegrant in a blender to form a blend; b) preparing a granulation solution with water, a binder, and a surfactant; c) feeding the blend from step a) into a continuous twin screw granulator while adding the granulation solution from step b) to produce granules; d) drying the granules from step c) and milling them; e) blending the milled granules from step d) with a filler, disintegrant, and lubricant to form a blend; f) compressing the blend from step e) into a tablet; and g) optionally coating the tablet from step f), wherein the particle size of Compound 1 Form I is between 0.1 and 10 microns.
56. The process of claim 55, wherein the tablet comprises at least 30 wt % by weight of Compound 1 Form I.
57. The process of claim 55, wherein the particle size of Compound 1 Form I is between 1 micron and 5 microns.
58. The process of claim 55, wherein Compound 1 Form I has a particle size D50 of 2.0 microns.
59. The process of claim 55, wherein the tablet has a target friability of less than 1.0% after 400 revolutions.
60. The process of claim 55, wherein the tablet has a hardness of at least 5 kP.
61. The process of claim 55, wherein Compound 1 Form I is characterized by one or more peaks within one or more 2.theta. ranges, selected from 15.2 to 15.6 degrees; 16.1 to 16.5 degrees; and 14.3 to 14.7 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
62. The process of claim 61, wherein Compound 1 Form I is characterized by a peak within the range of 16.1 to 16.5 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
63. The process of claim 62, wherein Compound 1 Form I is characterized by a peak having a 2.theta. value at 16.3 degrees in an X-ray powder diffraction.
64. The process of claim 61, wherein Compound 1 Form I is characterized by a peak within the range of 14.3 to 14.7 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
65. The process of claim 64, wherein Compound 1 Form I is characterized by a peak having a 2.theta. value at 14.5 degrees in an X-ray powder diffraction.
66. The process of claim 61, wherein Compound 1 Form I is characterized by a peak within the range of 15.2 to 15.6 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
67. The process of claim 66, wherein Compound 1 Form I is characterized by a peak having a 2.theta. value at 15.4 degrees in an X-ray powder diffraction.
68. The process of claim 61, wherein Compound 1 Form I is characterized by a peak within the range of 17.6 to 18.0 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
69. The process of claim 61, wherein Compound 1 Form I is further characterized by a peak within the range of 7.6 to 8.0 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation.
70. The process of claim 55, wherein Compound 1 Form I is characterized by one or more peaks having a 2.theta. value selected from 14.41 degrees, 14.64 degrees, 15.23 degrees, 16.11 degrees, 17.67 degrees, 19.32 degrees, 21.67 degrees, 23.40 degrees, 23.99 degrees, 26.10 degrees, and 28.54 degrees, all .+-.0.2 degrees, in an X-ray powder diffraction obtained using Cu K alpha radiation.
71. The process of claim 55, wherein Compound 1 Form I is characterized by one or more peaks having a 2.theta. value selected from 7.83 degrees; 14.51 degrees; 14.78 degrees; 15.39 degrees; 16.26 degrees; 16.62 degrees; 17.81 degrees;
21. 59 degrees; 23.32 degrees; 24.93 degrees; and 25.99 degrees, all .+-.0.2 degrees, in an X-ray powder diffraction obtained using Cu K alpha radiation.
72. The process of claim 55, wherein Compound 1 Form I is characterized by a diffraction pattern substantially similar to that of FIG. 1.
73. The process of claim 55, wherein Compound 1 Form I is characterized by a diffraction pattern substantially similar to that of FIG. 2.
74. The process of claim 55, wherein Compound 1 Form I is characterized as a monoclinic crystal system in P2.sub.1/n space group with the following unit cell dimensions: a=4.9626(7) .ANG., b=12.299(2) .ANG., c=33.075 (4) .ANG., .beta.=93.938(9).degree..
75. The process of claim 55, wherein the tablet further comprises N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide.
76. A tablet prepared by the process of claim 55.
77. A method of treating cystic fibrosis in a patient comprising administering a tablet prepared by the process of claim 55.
78. The method of claim 77, wherein the patient has a F508.DELTA. mutation.
79. The method of claim 78, wherein the patient is homozygous for F508.DELTA..
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application is a Continuation of U.S. patent application Ser. No. 15/655,354 filed Jul. 20, 2017, which is now abandoned, which is a Continuation of U.S. patent application Ser. No. 15/093,582 filed Apr. 7, 2016, which is now abandoned; which is a Continuation of U.S. patent application Ser. No. 13/750,069 filed Jan. 25, 2013, which is now abandoned; which claims priority to U.S. provisional patent application Ser. Nos. 61/590,479, filed Jan. 25, 2012; 61/651,218, filed May 24, 2012; 61/691,898, filed Aug. 22, 2012; and 61/708,691, filed Oct. 2, 2012, the entire contents of all applications are incorporated herein by reference in their entirety.
TECHNICAL FIELD OF INVENTION
[0002] The invention relates to pharmaceutical compositions comprising 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1), methods for manufacturing such compositions and methods for administering pharmaceutical compositions comprising same.
BACKGROUND
[0003] CFTR is a cAMP/ATP-mediated anion channel that is expressed in a variety of cells types, including absorptive and secretory epithelia cells, where it regulates anion flux across the membrane, as well as the activity of other ion channels and proteins. In epithelia cells, normal functioning of CFTR is critical for the maintenance of electrolyte transport throughout the body, including respiratory and digestive tissue. CFTR is composed of approximately 1480 amino acids that encode a protein made up of a tandem repeat of transmembrane domains, each containing six transmembrane helices and a nucleotide binding domain. The two transmembrane domains are linked by a large, polar, regulatory (R)-domain with multiple phosphorylation sites that regulate channel activity and cellular trafficking.
[0004] The gene encoding CFTR has been identified and sequenced (See Gregory, R. J. et al. (1990) Nature 347:382-386; Rich, D. P. et al. (1990) Nature 347:358-362), (Riordan, J. R. et al. (1989) Science 245:1066-1073). A defect in this gene causes mutations in CFTR resulting in cystic fibrosis ("CF"), the most common fatal genetic disease in humans. Cystic fibrosis affects approximately one in every 2,500 infants in the United States. Within the general United States population, up to 10 million people carry a single copy of the defective gene without apparent ill effects. In contrast, individuals with two copies of the CF associated gene suffer from the debilitating and fatal effects of CF, including chronic lung disease.
[0005] In patients with cystic fibrosis, mutations in CFTR endogenously expressed in respiratory epithelia lead to reduced apical anion secretion causing an imbalance in ion and fluid transport. The resulting decrease in anion transport contributes to enhance mucus accumulation in the lung and the accompanying microbial infections that ultimately cause death in CF patients. In addition to respiratory disease, CF patients typically suffer from gastrointestinal problems and pancreatic insufficiency that, if left untreated, results in death. In addition, the majority of males with cystic fibrosis are infertile and fertility is decreased among females with cystic fibrosis. In contrast to the severe effects of two copies of the CF associated gene, individuals with a single copy of the CF associated gene exhibit increased resistance to cholera and to dehydration resulting from diarrhea--perhaps explaining the relatively high frequency of the CF gene within the population.
[0006] Sequence analysis of the CFTR gene of CF chromosomes has revealed a variety of disease-causing mutations (Cutting, G. R. et al. (1990) Nature 346:366-369; Dean, M. et al. (1990) Cell 61:863:870; and Kerem, B-S. et al. (1989) Science 245:1073-1080; Kerem, B-S et al. (1990) Proc. Natl. Acad. Sci. USA 87:8447-8451). To date, greater than 1000 disease-causing mutations in the CF gene have been identified as reported by the scientific and medical literature. The most prevalent mutation is a deletion of phenylalanine at position 508 of the CFTR amino acid sequence, and is commonly referred to as F508del-CFTR. This mutation occurs in approximately 70 percent of the cases of cystic fibrosis and is associated with a severe disease. Other mutations include the R117H and G551D.
[0007] The deletion of residue 508 in F508del-CFTR prevents the nascent protein from folding correctly. This results in the inability of the mutant protein to exit the ER, and traffic to the plasma membrane. As a result, the number of channels present in the membrane is far less than observed in cells expressing wild-type CFTR. In addition to impaired trafficking, the mutation results in defective channel gating. Together, the reduced number of channels in the membrane and the defective gating lead to reduced anion transport across epithelia leading to defective ion and fluid transport. (Quinton, P. M. (1990), FASEB J. 4: 2709-2727). Studies have shown, however, that the reduced numbers of F508del-CFTR in the membrane are functional, albeit less than wild-type CFTR. (Dalemans et al. (1991), Nature Lond. 354: 526-528; Denning et al., supra; Pasyk and Foskett (1995), J. Cell. Biochem. 270: 12347-50). In addition to F508del-CFTR, other disease causing mutations in CFTR that result in defective trafficking, synthesis, and/or channel gating could be up- or down-regulated to alter anion secretion and modify disease progression and/or severity.
[0008] Although CFTR transports a variety of molecules in addition to anions, it is clear that this role (the transport of anions) represents one element in an important mechanism of transporting ions and water across the epithelium. The other elements include the epithelial Na.sup.+ channel, ENaC, Na.sup.+/2Cl.sup.-/K.sup.+ co-transporter, Na.sup.+--K.sup.+ ATPase pump and the basolateral membrane K.sup.+ channels, that are responsible for the uptake of chloride into the cell.
[0009] These elements work together to achieve directional transport across the epithelium via their selective expression and localization within the cell. Chloride absorption takes place by the coordinated activity of ENaC and CFTR present on the apical membrane and the Na.sup.+--K.sup.+-ATPase pump and Cl-- channels expressed on the basolateral surface of the cell. Secondary active transport of chloride from the luminal side leads to the accumulation of intracellular chloride, which can then passively leave the cell via Cl.sup.- channels, resulting in a vectorial transport. Arrangement of Na.sup.+/2Cl.sup.-/K.sup.+ co-transporter, Na.sup.+--K.sup.+-ATPase pump and the basolateral membrane K.sup.+ channels on the basolateral surface and CFTR on the luminal side coordinate the secretion of chloride via CFTR on the luminal side. Because water is probably never actively transported itself, its flow across epithelia depends on tiny transepithelial osmotic gradients generated by the bulk flow of sodium and chloride.
[0010] As discussed above, it is believed that the deletion of residue 508 in F508del-CFTR prevents the nascent protein from folding correctly, resulting in the inability of this mutant protein to exit the ER, and traffic to the plasma membrane. As a result, insufficient amounts of the mature protein are present at the plasma membrane and chloride transport within epithelial tissues is significantly reduced. In fact, this cellular phenomenon of defective endoplasmic reticulum (ER) processing of ATP-binding cassette (ABC) transporters by the ER machinery, has been shown to be the underlying basis not only for CF disease, but for a wide range of other isolated and inherited diseases. The two ways that the ER machinery can malfunction is either by loss of coupling to ER export of the proteins leading to degradation, or by the ER accumulation of these defective/misfolded proteins [Aridor M, et al., Nature Med., 5(7), pp 745-751 (1999); Shastry, B. S., et al., Neurochem. International, 43, pp 1-7 (2003); Rutishauser, J., et al., Swiss Med Wkly, 132, pp 211-222 (2002); Morello, J P et al., TIPS, 21, pp. 466-469 (2000); Bross P., et al., Human Mut., 14, pp. 186-198 (1999)].
[0011] Compound 1 in salt form is disclosed in International PCT Publication WO 2007056341 as a modulator of CFTR activity and thus as a useful treatment for CFTR-mediated diseases such as cystic fibrosis. Compound 1 Form I, which is substantially crystalline and salt-free, is disclosed in United States Published Patent Application US20090170905, filed Dec. 4, 2008. Compound 1 Form II and Compound 1 HCl salt Form A are disclosed in United States Published Patent Application US20110263654, filed Apr. 7, 2011. All applications are incorporated in their entirety by reference herein.
[0012] Compound 1, as part of a combination with ivacaftor (N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide), has been granted a Breakthrough Therapy Designation from the Food and Drug Administration (FDA) for the treatment of cystic fibrosis, one of only two such grants at the time of the filing of this application (the other being for ivacaftor). This demonstrates a significant unmet need for the effective treatment of the cause of cystic fibrosis over symptomatic treatments. Additionally, a common challenge for drugs approved by the FDA is the occasional lack of drug availability for patients in need thereof. Accordingly, a significant unmet need exists for the presently disclosed Compound 1 formulations and processes for preparing them in a continuous and controlled manner.
SUMMARY
[0013] The invention relates to pharmaceutical compositions, pharmaceutical preparations, and solid dosage forms comprising 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1) which has the structure below:
##STR00001##
[0014] In one aspect, the invention provides a pharmaceutical composition comprising:
[0015] a. Compound 1;
[0016] b. a filler;
[0017] c. a disintegrant;
[0018] d. a surfactant;
[0019] e. a lubricant; and
[0020] f. a glidant or a binder.
[0021] In other embodiments, Compound 1 is in substantially one of its crystalline solid forms. In one embodiment, Compound 1 is in substantially crystalline Form I (Compound 1 Form I). In one embodiment, Compound 1 is in substantially crystalline Form II (Compound 1 Form II). In one embodiment, Compound 1 is in substantially crystalline HCl salt form (Compound 1 HCl Salt Form A). It is understood that the term "Compound 1," as used throughout, includes, amongst other forms, including non-crystalline forms, the following solid state forms: Compound 1 Form I, Compound 1 Form II, and/or Compound 1 HCl Salt Form A.
[0022] In some embodiments, the pharmaceutical composition comprises 25 mg to 400 mg. In some embodiments, the pharmaceutical composition comprises 25 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 50 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 100 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 125 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 150 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 200 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 250 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 300 mg of Compound 1. In some embodiments, the pharmaceutical composition comprises 400 mg of Compound 1.
[0023] In one aspect, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00001 Roller Compaction Granule Blend (% w/w) Compound 1 20-40 Microcrystalline cellulose 30-50 Mannitol 10-30 Croscarmellose Sodium 1-5 Sodium Lauryl Sulfate 0.1-2 Colloidal Silica 0.1-1 Magnesium Stearate 1-3 Tablet Composition (% w/w) Roller Compaction Granule Blend 99-99.9 Magnesium Stearate 0.1-1
[0024] In one aspect, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00002 High Shear Granule Blend (% w/w) Compound 1 60-70 Microcrystalline cellulose 5-15 Croscarmellose Sodium 1-5 Sodium Lauryl Sulfate 0.1-2 Polyvinylpyrrolidone 1-5 Tablet Composition (% w/w) High Shear Granule Blend 75-89 Microcrystalline cellulose 10-15 Croscarmellose Sodium 1-5 Magnesium Stearate 0.1-5
[0025] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00003 High Shear Granule Blend (% w/w) Compound 1 Form I 60-70 Microcrystalline cellulose 5-15 Croscarmellose Sodium 1-5 Polyvinylpyrrolidone 1-5 Sodium Lauryl Sulfate 0.1-2 Tablet Composition (% w/w) High Shear Granule Blend 78-89 Microcrystalline cellulose 10-15 Croscarmellose Sodium 1-5 Magnesium Stearate 0.1-2 Film Coated Tablet (% w/w) Core Tablet Composition 95-99 Film Coat 1-5 Wax Trace
[0026] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00004 Roller Compaction Granule Blend (% w/w) Compound 1 Form I 30 Microcrystalline cellulose 42.3 Mannitol 21.2 Croscarmellose Sodium 3 Sodium Lauryl Sulfate 1 Colloidal Silica 0.5 Magnesium Stearate 2 Tablet Composition (% w/w) Roller Compaction Granule Blend 99.5 Magnesium Stearate 0.5
[0027] In another aspect, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00005 High Shear Granule Blend (% w/w) Compound 1 Form I 40-80 Microcrystalline cellulose 20-40 Mannitol 10-15 Croscarmellose Sodium 1-5 Polyvinylpyrrolidone 1-10 Sodium Lauryl Sulfate 0.1-2 Tablet Composition (% w/w) High Shear Granule Blend 95-99 Croscarmellose Sodium 1-4 Magnesium Stearate 0.1-1
[0028] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00006 High Shear Granule Blend (% w/w) Compound 1 Form I 50 Microcrystalline cellulose 30 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (% w/w) High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0029] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00007 High Shear Granule Blend (% w/w) Compound 1 Form I 60 Microcrystalline cellulose 20 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (% w/w) High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0030] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00008 High Shear Granule Blend (% w/w) Compound 1 Form I 60 Microcrystalline cellulose 20 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (% w/w) High Shear Granule Blend 83 Microcrystalline cellulose 14 Croscarmellose Sodium 2 Magnesium Stearate 1
[0031] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00009 Twin Screw Granule Blend (% w/w) Compound 1 Form I 60 Microcrystalline cellulose 20 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (% w/w) Twin Screw Granule Blend 83 Microcrystalline cellulose 14 Croscarmellose Sodium 2 Magnesium Stearate 1
[0032] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00010 Twin Screw Wet Granule Blend (% w/w) Compound 1 Form I 80.0 Microcrystalline cellulose 13.6 Croscarmellose Sodium 2.5 Polyvinylpyrrolidone 3.1 Sodium Lauryl Sulfate 0.7 Tablet Composition (% w/w) Twin Screw Granule Blend 83 Microcrystalline cellulose 12 Croscarmellose Sodium 4 Magnesium Stearate 1
[0033] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00011 Twin Screw Granule Blend (% w/w) Compound 1 Form I 80.0 Microcrystalline cellulose 13.6 Croscarmellose Sodium 2.5 Polyvinylpyrrolidone 3.1 Sodium Lauryl Sulfate 0.7 Tablet Composition (% w/w) Twin Screw Granule Blend 83 Microcrystalline cellulose 12 Croscarmellose Sodium 4 Magnesium Stearate 1 Film Coated Tablet (% w/w) Core Tablet Composition 97 Film Coat 3 Wax Trace
[0034] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00012 High Shear Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 66 Mannitol 43 Croscarmellose Sodium 7 Polyvinylpyrrolidone 13 Sodium Lauryl Sulfate 3 Core Tablet Composition (200 mg dose) mg High Shear Granule Blend 332 Microcrystalline cellulose 56 Croscarmellose Sodium 8 Magnesium Stearate 4 Film Coated Tablet (200 mg dose) mg Core Tablet Composition 400 Film Coat 12 Wax trace
[0035] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00013 Twin Screw Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 66 Mannitol 43 Croscarmellose Sodium 7 Polyvinylpyrrolidone 13 Sodium Lauryl Sulfate 3 Core Tablet Composition (200 mg dose) mg Twin Screw Granule Blend 332 Microcrystalline cellulose 56 Croscarmellose Sodium 8 Magnesium Stearate 4
[0036] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00014 High Shear Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 67 Mannitol 45 Croscarmellose Sodium 7 Polyvinylpyrrolidone 10.4 Sodium Lauryl Sulfate 2.6 Core Tablet Composition (200 mg dose) mg High Shear Granule Blend 332 Microcrystalline cellulose 56 Croscarmellose Sodium 8 Magnesium Stearate 4 Film Coated Tablet (200 mg dose) mg Core Tablet Composition 400 Film Coat 12 Wax trace
[0037] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00015 mg High Shear Granule Blend Compound 1 Form I 300 Microcrystalline cellulose 99 Mannitol 64.5 Croscarmellose Sodium 10.5 Polyvinylpyrrolidone 19.5 Sodium Lauryl Sulfate 4.5 Core Tablet Composition (300 mg dose) High Shear Granule Blend 498 Microcrystalline cellulose 84 Croscarmellose Sodium 12 Magnesium Stearate 6 Film Coated Tablet (300 mg dose) Core Tablet Composition 600 Film Coat 18 Wax trace
[0038] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00016 mg High Shear Granule Blend Compound 1 Form I 300 Microcrystalline cellulose 100.5 Mannitol 67.5 Croscarmellose Sodium 10.5 Polyvinylpyrrolidone 15.6 Sodium Lauryl Sulfate 3.9 Core Tablet Composition (300 mg dose) High Shear Granule Blend 498 Microcrystalline cellulose 84 Croscarmellose Sodium 12 Magnesium Stearate 6 Film Coated Tablet (300 mg dose) Core Tablet Composition 600 Film Coat 18 Wax trace
[0039] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00017 (% w/w) High Shear Granule Blend Compound 1 Form I 70 Microcrystalline cellulose 12 Mannitol 11 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0040] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00018 (% w/w) High Shear Granule Blend Compound 1 Form I or Form II 61 Microcrystalline cellulose 20.3 Mannitol 13.2 Croscarmellose Sodium 2 Polyvinylpyrrolidone 2.7 Sodium Lauryl Sulfate 0.7 Tablet Composition High Shear Granule Blend 83 Microcrystalline cellulose 14 Croscarmellose Sodium 2 Magnesium Stearate 1
[0041] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00019 mg High Shear Granule Blend Compound 1 Form I or Form II 100 Microcrystalline cellulose 33.3 Mannitol 21.7 Croscarmellose Sodium 3.3 Polyvinylpyrrolidone 4.4 Sodium Lauryl Sulfate 1.1 Core Tablet Composition (100 mg dose) High Shear Granule Blend 163.9 Microcrystalline cellulose 27.6 Croscarmellose Sodium 3.9 Magnesium Stearate 2.0
[0042] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00020 mg Twin Screw Granule Blend Compound 1 Form I 200 Microcrystalline cellulose 34.0 Croscarmellose Sodium 6.3 Polyvinylpyrrolidone 7.8 Sodium Lauryl Sulfate 1.8 Core Tablet Composition (200 mg dose) Twin Screw Granule Blend 249.9 Microcrystalline cellulose 36.1 Croscarmellose Sodium 12.0 Magnesium Stearate 3.0
[0043] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00021 mg Twin Screw Granule Blend Compound 1 Form I 400 Microcrystalline cellulose 68.0 Croscarmellose Sodium 12.6 Polyvinylpyrrolidone 15.6 Sodium Lauryl Sulfate 3.6 Core Tablet Composition (400 mg dose) Twin Screw Granule Blend 499.8 Microcrystalline cellulose 72.2 Croscarmellose Sodium 24.0 Magnesium Stearate 6.0
[0044] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00022 mg Twin Screw Granule Blend Compound 1 Form I 200 Microcrystalline cellulose 34.0 Croscarmellose Sodium 6.3 Polyvinylpyrrolidone 7.8 Sodium Lauryl Sulfate 1.8 Core Tablet Composition (200 mg dose) Twin Screw Granule Blend 249.9 Microcrystalline cellulose 36.1 Croscarmellose Sodium 12.0 Magnesium Stearate 3.0 Film Coated Tablet (200 mg dose, 310 mg total) Core Tablet Composition 301 Film Coat 9.0 Wax trace
[0045] In another embodiment, the invention provides a pharmaceutical composition comprising the following components:
TABLE-US-00023 mg Twin Screw Granule Blend Compound 1 Form I 400 Microcrystalline cellulose 68.0 Croscarmellose Sodium 12.6 Polyvinylpyrrolidone 15.6 Sodium Lauryl Sulfate 3.6 Core Tablet Composition (400 mg dose) Twin Screw Granule Blend 499.8 Microcrystalline cellulose 72.2 Croscarmellose Sodium 24.0 Magnesium Stearate 6.0 Film Coated Tablet (400 mg dose, 620 mg total) Core Tablet Composition 602 Film Coat 18.0 Wax trace
[0046] In another aspect, the invention provides a pharmaceutical composition in the form of a tablet that comprises Compound 1, and one or more pharmaceutically acceptable excipients, for example, a filler, a disintegrant, a surfactant, a diluent, a binder, a glidant, and a lubricant and any combination thereof, where the tablet has a dissolution of at least about 50% in about 30 minutes. In another embodiment, the dissolution rate is at least about 75% in about 30 minutes. In another embodiment, the dissolution rate is at least about 90% in about 30 minutes.
[0047] In another aspect, the invention provides a pharmaceutical composition consisting of a tablet that comprises a powder blend or granules comprising Compound 1; and, one or more pharmaceutically acceptable excipients, for example, a filler, a disintegrant, a surfactant, a diluent, a binder, a glidant, and a lubricant, wherein the tablet has a hardness of at least about 5 kP (kP=kilo Ponds; 1 kP=.about.9.8 N). In another embodiment, the tablet has a target friability of less than 1.0% after 400 revolutions. In another aspect, the invention provides a pharmaceutical composition consisting of a tablet that comprises a powder blend or granules comprising Compound 1 Form II, Compound 1; and, one or more pharmaceutically acceptable excipients, for example, a filler, a disintegrant, a surfactant, a diluent, a binder, a glidant, and a lubricant, wherein the tablet has a hardness of at least about 5 kP (kP=kilo Ponds; 1 kP=.about.9.8 N). In another embodiment, the tablet has a target friability of less than 1.0% after 400 revolutions.
[0048] In another aspect, the invention provides a pharmaceutical composition as described herein further comprising an additional therapeutic agent. In some embodiments, the additional therapeutic agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3 -carboxamide.
[0049] In another aspect, the invention provides a method of treating a CFTR mediated disease in a mammal comprising administering to the mammal an effective amount of a pharmaceutical composition as described herein. In some embodiments, the CFTR mediated disease is cystic fibrosis, emphysema, COPD, or osteoporosis. In other embodiments, the CFTR mediated disease is cystic fibrosis. This method may further comprise administering an additional therapeutic agent, wherein in some embodiments, the additional therapeutic agent is selected from a mucolytic agent, bronchodilator, an anti-biotic, an anti-infective agent, an anti-inflammatory agent, a CFTR potentiator, or a nutritional agent. In another embodiment, the additional therapeutic agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide. In another embodiment, the patient has a F508del-CFTR mutation. In another embodiment, the patient is homozygous for F508del. In another embodiment, the patient is heterozygous for F508del.
[0050] In another aspect, the invention features a kit comprising a tablet of the present invention, and a separate therapeutic agent or pharmaceutical composition thereof. In another embodiment, the Compound 1 in the tablet is in Form I. In another embodiment, the therapeutic agent is a cystic fibrosis corrector other than Compound 1. In another embodiment, the therapeutic agent is a cystic fibrosis potentiator. In another embodiment, the therapeutic agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide. In another embodiment, the tablet and the therapeutic agent are in separate containers. In another embodiment, the separate containers are bottles. In another embodiment, the separate containers are vials. In another embodiment, the separate containers are blister packs.
[0051] In another aspect, the invention provides a process for making the pharmaceutical compositions described herein by a roller compaction process comprising the steps of screening and weighing Compound 1 and excipients; blending Compound 1 and excipients for a suitable amount of time; roller compacting the blend into ribbons and milling the ribbons into granules; blending the granules with extra-granular excipients for a suitable amount of time; compressing the blend into tablets; coating the tablets; and, optionally, printing a monogram on one or both tablet faces.
[0052] In another aspect, the invention provides a process for making the pharmaceutical compositions described herein by a high shear granulation process comprising the steps of screening and weighing Compound 1 and excipients; mixing Compound 1 and excipients while adding a granulation fluid comprising surfactant and a binder at a suitable mixing speed for a suitable amount of time and chopping the mixture into granules; drying the granules; blending the granules with extra-granular excipients for a suitable amount of time; compressing the blend into tablets; coating the tablets; and, optionally, printing a monogram on one or both tablet faces.
[0053] In another aspect, the invention provides a continuous or semi-continuous process for making the pharmaceutical compositions described herein by a twin screw wet granulation process comprising the steps of screening and weighing Compound 1 and excipients; mixing Compound 1 and excipients in a blender and feeding the blend into a continuous granulator while adding a granulation fluid comprising surfactant and a binder at a suitable rate for a suitable amount of time and chopping the mixture into granules; drying the granules; blending the granules with extra-granular excipients for a suitable amount of time; compressing the blend into tablets; coating the tablets; and, optionally, printing a monogram on one or both tablet faces.
BRIEF DESCRIPTION OF DRAWINGS
[0054] FIG. 1 is an X-ray diffraction pattern calculated from a single crystal structure of Compound 1 Form I.
[0055] FIG. 2 is an actual X-ray powder diffraction pattern of Compound 1 Form I.
[0056] FIG. 3 is an X-ray powder diffraction pattern of Compound 1 Form II.
[0057] FIG. 4 provides X-ray diffraction patterns of Compound 1 Form II's selected from:
[0058] 1) Compound 1 Form II, Methanol Solvate;
[0059] 2) Compound 1 Form II, Ethanol Solvate;
[0060] 3) Compound 1 Form II, Acetone Solvate;
[0061] 4) Compound 1 Form II, 2-Propanol Solvate;
[0062] 5) Compound 1 Form II, Acetonitrile Solvate;
[0063] 6) Compound 1 Form II, Tetrahydrofuran Solvate;
[0064] 7) Compound 1 Form II, Methyl Acetate Solvate;
[0065] 8) Compound 1 Form II, 2-Butanone Solvate;
[0066] 9) Compound 1 Form II, Ethyl Formate Solvate; and
[0067] 10) Compound 1 Form II, 2-Methyltetrahydrofuran Solvate.
[0068] FIG. 5 provides an X-ray diffraction pattern of Compound 1 Form II, Methanol Solvate.
[0069] FIG. 6 provides an X-ray diffraction pattern of Compound 1 Form II, Ethanol Solvate.
[0070] FIG. 7 provides an X-ray diffraction pattern of Compound 1 Form II, Acetone Solvate.
[0071] FIG. 8 provides an X-ray diffraction pattern of Compound 1 Form II, 2-Propanol Solvate.
[0072] FIG. 9 provides an X-ray diffraction pattern of Compound 1 Form II, Acetonitrile Solvate.
[0073] FIG. 10 provides an X-ray diffraction pattern of Compound 1 Form II, Tetrahydrofuran Solvate.
[0074] FIG. 11 provides an X-ray diffraction pattern of Compound 1 Form II, Methyl Acetate Solvate.
[0075] FIG. 12 provides an X-ray diffraction pattern of Compound 1 Form II, 2-Butanone Solvate.
[0076] FIG. 13 provides an X-ray diffraction pattern of Compound 1 Form II, Ethyl Formate Solvate.
[0077] FIG. 14 provides an X-ray diffraction pattern of Compound 1 Form II, 2-Methyltetrahydrofuran Solvate.
[0078] FIG. 15 is a differential scanning calorimetry (DSC) trace of Compound 1 Form II, Acetone Solvate.
[0079] FIG. 16 is a Thermogravimetric analysis (TGA) plot of Compound 1 Form II, Acetone Solvate.
[0080] FIG. 17 is a conformational image of Compound 1 Form II, Acetone Solvate based on single crystal X-ray analysis.
[0081] FIG. 18 is a conformational image of the dimer of Compound 1 HCl Salt Form A.
[0082] FIG. 19 is an X-ray diffraction pattern of Compound 1 HCl Salt Form A calculated from the crystal structure.
[0083] FIG. 20 is an .sup.1H NMR spectrum of Compound 1.
[0084] FIG. 21 is an .sup.1H NMR spectrum of Compound 1 HCl salt.
[0085] FIG. 22 is a differential scanning calorimetry (DSC) trace of Compound 1 Form I.
[0086] FIG. 23 is a conformational picture of Compound 1 Form I based on single crystal X-ray analysis.
[0087] FIG. 24 is a conformational image of Compound 1 Form II, Acetone Solvate, based on single crystal X-ray analysis.
[0088] FIG. 25 is a solid state .sup.13C NMR spectrum (15.0 kHz spinning) of Compound 1 Form II, Acetone Solvate.
[0089] FIG. 26 is a solid state .sup.19F NMR spectrum (12.5 kHz spinning) of Compound 1 Form II, Acetone Solvate.
[0090] FIG. 27 is an X-ray diffraction pattern of Compound 1 HCl Salt Form A calculated from the crystal structure.
[0091] FIG. 28 is a graph depicting Compound 1 pH gradient dissolution profiles for a tablet made by a high shear granulation (HSG) process and a twin screw wet granulation (TSWG) process (LOD stands for loss on drying, a measure to define the amount of water in a powder/granule).
DETAILED DESCRIPTION
Definitions
[0092] As used herein, "CFTR" stands for cystic fibrosis transmembrane conductance regulator.
[0093] As used herein, a ".DELTA.F508" or "F508del" is a specific mutation within the CFTR protein. The mutation is a deletion of the three nucleotides that comprise the codon for amino acid phenylalanine at position 508, resulting in CFTR protein that lacks this particular phenylalanine.
[0094] As used herein, a patient who is "homozygous" for a particular mutation, e.g. F508del, has the same mutation on both alleles.
[0095] As used herein, a patient who is "heterozygous" for a particular mutation, e.g. F508del, has this mutation on one allele, and a different mutation on the other allele.
[0096] As used herein, the term "CFTR corrector" refers to a compound that augments or induces the amount of functional CFTR protein to the cell surface, resulting in increased functional activity.
[0097] As used herein, the term "CFTR potentiator" refers to a compound that augments or induces the channel activity of CFTR protein located at the cell surface, resulting in increased functional activity.
[0098] As used herein, the term "active pharmaceutical ingredient" or "API" refers to a biologically active compound. Exemplary APIs include 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1).
[0099] The terms "solid form", "solid forms" and related terms, when used herein to refer to 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl) benzoic acid (Compound 1), refer to a solid form e.g. crystals and the like, comprising Compound 1 which is not predominantly in a liquid or a gaseous state.
[0100] As used herein, the term "substantially amorphous" refers to a solid material having little or no long range order in the position of its molecules. For example, substantially amorphous materials have less than about 15% crystallinity (e.g., less than about 10% crystallinity or less than about 5% crystallinity). It is also noted that the term `substantially amorphous` includes the descriptor, `amorphous`, which refers to materials having no (0%) crystallinity.
[0101] As used herein, the term "substantially crystalline" (as in the phrase substantially crystalline Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A) refers to a solid material having predominantly long range order in the position of its molecules. For example, substantially crystalline materials have more than about 85% crystallinity (e.g., more than about 90% crystallinity or more than about 95% crystallinity). It is also noted that the term `substantially crystalline` includes the descriptor, `crystalline`, which refers to materials having 100% crystallinity.
[0102] The term "crystalline" and related terms used herein, when used to describe a substance, component, product, or form, means that the substance, component or product is substantially crystalline as determined by X-ray diffraction. (See, e.g., Remington: The Science and Practice of Pharmacy, 21st Ed., Lippincott Williams & Wilkins, Baltimore, Md. (2003); The United States Pharmacopeia, 23.sup.rd ed., 1843-1844 (1995)).
[0103] As used herein, the term "composition" generally refers to a composition of two or more components, usually one or more drugs (e.g., one drug (e.g., Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A)) and one or more pharmaceutical excipients.
[0104] As used herein, the term "solid dosage form" generally refers to a pharmaceutical composition, which when used in an oral mode of administration include capsules, tablets, pills, powders and granules. In such solid dosage forms, the active compound is mixed with at least one inert, pharmaceutically acceptable excipient or carrier.
[0105] As used herein, an "excipient" includes functional and non-functional ingredients in a pharmaceutical composition.
[0106] As used herein, a "disintegrant" is an excipient that hydrates a pharmaceutical composition and aids in tablet dispersion. As used herein, a "diluent" or "filler" is an excipient that adds bulkiness to a pharmaceutical composition.
[0107] As used herein, a "surfactant" is an excipient that imparts pharmaceutical compositions with enhanced solubility and/or wetability.
[0108] As used herein, a "binder" is an excipient that imparts a pharmaceutical composition with enhanced cohesion or tensile strength (e.g., hardness).
[0109] As used herein, a "glidant" is an excipient that imparts a pharmaceutical compositions with enhanced flow properties.
[0110] As used herein, a "colorant" is an excipient that imparts a pharmaceutical composition with a desired color. Examples of colorants include commercially available pigments such as FD&C Blue #1 Aluminum Lake, FD&C Blue #2, other FD&C Blue colors, titanium dioxide, iron oxide, and/or combinations thereof In one embodiment, the pharmaceutical composition provided by the invention is purple.
[0111] As used herein, a "lubricant" is an excipient that is added to pharmaceutical compositions that are pressed into tablets. The lubricant aids in compaction of granules into tablets and ejection of a tablet of a pharmaceutical composition from a die press.
[0112] As used herein, "cubic centimeter" and "cc" are used interchangeably to represent a unit of volume. Note that 1 cc=1 mL.
[0113] As used herein, "kiloPond" and "kP" are used interchangeably and refer to the measure of force where a kP=approximately 9.8 Newtons.
[0114] As used herein, "friability" refers to the property of a tablet to remain intact and withhold its form despite an external force of pressure. Friability can be quantified using the mathematical expression presented in equation 1:
% friabiliy = 100 .times. ( W 0 - W f ) W 0 ( 1 ) ##EQU00001##
wherein W.sub.0 is the original weight of the tablet and W.sub.f is the final weight of the tablet after it is put through the friabilator. Friability is measured using a standard USP testing apparatus that tumbles experimental tablets for 100 or 400 revolutions. Some tablets of the invention have a friability of less than 5.0%. In another embodiment, the friability is less than 2.0%. In another embodiment, the target friability is less than 1.0% after 400 revolutions.
[0115] As used herein, "mean particle diameter" is the average particle diameter as measured using techniques such as laser light scattering, image analysis, or sieve analysis. In one embodiment, the granules used to prepare the pharmaceutical compositions provided by the invention have a mean particle diameter of less than 1.0 mm.
[0116] As used herein, "bulk density" is the mass of particles of material divided by the total volume the particles occupy. The total volume includes particle volume, inter-particle void volume and internal pore volume. Bulk density is not an intrinsic property of a material; it can change depending on how the material is processed. In one embodiment, the granules used to prepare the pharmaceutical compositions provided by the invention have a bulk density of about 0.5-0.7 g/cc.
[0117] An effective amount or "therapeutically effective amount" of a drug compound of the invention may vary according to factors such as the disease state, age, and weight of the subject, and the ability of the compound of the invention to elicit a desired response in the subject. Dosage regimens may be adjusted to provide the optimum therapeutic response. An effective amount is also one in which any toxic or detrimental effects (e.g., side effects) of the compound of the invention are outweighed by the therapeutically beneficial effects.
[0118] As used herein, and unless otherwise specified, the terms "therapeutically effective amount" and "effective amount" of a compound mean an amount sufficient to provide a therapeutic benefit in the treatment or management of a disease or disorder, or to delay or minimize one or more symptoms associated with the disease or disorder. A "therapeutically effective amount" and "effective amount" of a compound mean an amount of therapeutic agent, alone or in combination with one or more other agent(s), which provides a therapeutic benefit in the treatment or management of the disease or disorder. The terms "therapeutically effective amount" and "effective amount" can encompass an amount that improves overall therapy, reduces or avoids symptoms or causes of disease or disorder, or enhances the therapeutic efficacy of another therapeutic agent.
[0119] "Substantially pure" as used in the phrase "substantially pure Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A," means greater than about 90% purity. In another embodiment, substantially pure refers to greater than about 95% purity. In another embodiment, substantially pure refers to greater than about 98% purity. In another embodiment, substantially pure refers to greater than about 99% purity.
[0120] With respect to Compound 1 (e.g., Compound 1 Form I, Compound 1 Form II, Compound 1 HCl Salt Form A), the terms "about" and "approximately", when used in connection with doses, amounts, or weight percent of ingredients of a composition or a dosage form, mean a dose, amount, or weight percent that is recognized by one of ordinary skill in the art to provide a pharmacological effect equivalent to that obtained from the specified dose, amount, or weight percent. Specifically the term "about" or "approximately" means an acceptable error for a particular value as determined by one of ordinary skill in the art, which depends in part on how the value is measured or determined. In certain embodiments, the term "about" or "approximately" means within 1, 2, 3, or 4 standard deviations. In certain embodiments, the term "about" or "approximately" means within 30%, 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.5%, 0.1%, or 0.05% of a given value or range.
[0121] Unless otherwise specified, the term "Compound 1" includes, but is not limited to, the solid forms of Compound 1 as described herein, e.g. Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A, as well as combinations thereof.
Pharmaceutical Compositions
[0122] The invention provides pharmaceutical compositions, pharmaceutical formulations and solid dosage forms comprising Compound 1 which may be in substantially crystalline form. In some embodiments, Compound 1 is in crystalline Form I (Compound 1 Form I). In some embodiments, Compound 1 is in crystalline Form II (Compound 1 Form II). In some embodiments, Compound 1 is in crystalline HCl salt form (Compound 1 HCl Salt Form A). In some embodiments of this aspect, the amount of Compound 1 that is present in the pharmaceutical composition is 25 mg, 50 mg, 75 mg, 100 mg, 125 mg, 150 mg, 200 mg, 250 mg, or 400 mg. In some embodiments of this aspect, weight/weight relative percent of Compound 1 that is present in the pharmaceutical composition is from 10 to 75 percent. In these and other embodiments, 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid is present as substantially pure Compound 1. "Substantially pure" means greater than ninety percent pure; preferably greater than 95 percent pure; more preferably greater than 99.5 percent pure (i.e., not mixed with other crystalline forms of Compound 1).
[0123] Thus in one aspect, the invention provides a pharmaceutical composition comprising:
[0124] a. Compound 1;
[0125] b. a filler;
[0126] c. a disintegrant;
[0127] d. a surfactant;
[0128] e. a diluent;
[0129] f. a lubricant; and
[0130] g. a glidant or a binder.
[0131] In one embodiment of this aspect, the pharmaceutical composition comprises 25 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 50 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 100 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 125 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 150 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 200 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 250 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 300 mg of Compound 1. In another embodiment of this aspect, the pharmaceutical composition comprises 400 mg of Compound 1.
[0132] In some embodiments, the pharmaceutical compositions comprises Compound 1, wherein Compound 1 is present in an amount of at least 15 wt % (e.g., at least 20 wt %, at least 30 wt %, at least 40 wt %, at least 50 wt %, at least 60 wt %, or at least 70 wt %) by weight of the composition.
[0133] In some embodiments, the pharmaceutical composition comprises Compound 1, a filler, a diluent, a disintegrant, a surfactant, a glidant, and a lubricant. In this embodiment, the composition comprises from about 20 wt % to about 50 wt % (e.g., about 25-35 wt %) of Compound 1 by weight of the composition, and more typically, from 25 wt % to about 45 wt % (e.g., about 28-32 wt %) of Compound 1 by weight of the composition.
[0134] In some embodiments, the pharmaceutical composition comprises Compound 1, a filler, a diluent, a disintegrant, a surfactant, a binder, and a lubricant. In this embodiment, the composition comprises from about 30 wt % to about 60 wt % (e.g., about 40-55 wt %) of Compound 1 by weight of the composition, and more typically from 35 wt % to about 70 wt % (e.g., about 45-55 wt %) of Compound 1 by weight of the composition.
[0135] The concentration of Compound 1 in the composition depends on several factors such as the amount of pharmaceutical composition needed to provide a desired amount of Compound 1 and the desired dissolution profile of the pharmaceutical composition.
[0136] In another embodiment, the pharmaceutical composition comprises Compound 1, in which Compound 1 in its solid form has a mean particle diameter, measured by light scattering (e.g., using a Malvern Mastersizer available from Malvern Instruments in England) of 0.1 microns to 10 microns. In another embodiment, the particle size of Compound 1 is 1 micron to 5 microns. In another embodiment, Compound 1 has a particle size D50 of 2.0 microns.
[0137] As indicated, in addition to Compound 1, in some embodiments of the invention, the pharmaceutical compositions which are oral formulations also comprise one or more excipients such as fillers, disintegrants, surfactants, diluents, binders, glidants, lubricants, colorants, or fragrances and any combination thereof.
[0138] Fillers suitable for the invention are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the hardness, the chemical stability, the physical stability, or the biological activity of the pharmaceutical composition. Exemplary fillers include: celluloses, modified celluloses, (e.g. sodium carboxymethyl cellulose, ethyl cellulose hydroxymethyl cellulose, hydroxypropylcellulose), cellulose acetate, microcrystalline cellulose, calcium phosphates, dibasic calcium phosphate, starches (e.g. corn starch, potato starch), sugars (e.g., sorbitol) lactose, sucrose, or the like), or any combination thereof.
[0139] Thus, in one embodiment, the pharmaceutical composition comprises at least one filler in an amount of at least 5 wt % (e.g., at least about 20 wt %, at least about 30 wt %, or at least about 40 wt %) by weight of the composition. For example, the pharmaceutical composition comprises from about 10 wt % to about 60 wt % (e.g., from about 20 wt % to about 55 wt %, from about 25 wt % to about 50 wt %, or from about 27 wt % to about 45 wt %) of filler, by weight of the composition. In another example, the pharmaceutical composition comprises at least about 20 wt % (e.g., at least 30 wt % or at least 40 wt %) of microcrystalline cellulose, for example MCC Avicel PH102, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 10 wt % to about 60 wt % (e.g., from about 20 wt % to about 55 wt % or from about 25 wt % to about 45 wt %) of microcellulose, by weight of the composition.
[0140] Disintegrants suitable for the invention enhance the dispersal of the pharmaceutical composition and are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the chemical stability, the physical stability, the hardness, or the biological activity of the pharmaceutical composition. Exemplary disintegrants include croscarmellose sodium, sodium starch glycolate, or a combination thereof
[0141] Thus, in one embodiment, the pharmaceutical composition comprises disintegrant in an amount of about 10 wt % or less (e.g., about 7 wt % or less, about 6 wt % or less, or about 5 wt % or less) by weight of the composition. For example, the pharmaceutical composition comprises from about 1 wt % to about 10 wt % (e.g., from about 1.5 wt % to about 7.5 wt % or from about 2.5 wt % to about 6 wt %) of disintegrant, by weight of the composition. In another example, the pharmaceutical composition comprises about 10 wt % or less (e.g., 7 wt % or less, 6 wt % or less, or 5 wt % or less) of croscarmellose sodium, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 1 wt % to about 10 wt % (e.g., from about 1.5 wt % to about 7.5 wt % or from about 2.5 wt % to about 6 wt %) of croscarmellose sodium, by weight of the composition. In some examples, the pharmaceutical composition comprises from about 0.1% to about 10 wt % (e.g., from about 0.5 wt % to about 7.5 wt % or from about 1.5 wt % to about 6 wt %) of disintegrant, by weight of the composition. In still other examples, the pharmaceutical composition comprises from about 0.5% to about 10 wt % (e.g., from about 1.5 wt % to about 7.5 wt % or from about 2.5 wt % to about 6 wt %) of disintegrant, by weight of the composition.
[0142] Surfactants suitable for the invention enhance the wettability of the pharmaceutical composition and are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the chemical stability, the physical stability, the hardness, or the biological activity of the pharmaceutical composition. Exemplary surfactants include sodium lauryl sulfate (SLS), sodium stearyl fumarate (SSF), polyoxyethylene 20 sorbitan mono-oleate (e.g., Tween.TM.), any combination thereof, or the like.
[0143] Thus, in one embodiment, the pharmaceutical composition comprises a surfactant in an amount of about 10 wt % or less (e.g., about 5 wt % or less, about 2 wt % or less, about 1 wt % or less, about 0.8 wt % or less, or about 0.6 wt % or less) by weight of the composition. For example, the pharmaceutical composition includes from about 10 wt % to about 0.1 wt % (e.g., from about 5 wt % to about 0.2 wt % or from about 2 wt % to about 0.3 wt %) of surfactant, by weight of the composition. In another example, the pharmaceutical composition comprises 10 wt % or less (e.g., about 5 wt % or less, about 2 wt % or less, about 1 wt % or less, about 0.8 wt % or less, or about 0.6 wt % or less) of sodium lauryl sulfate, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 10 wt % to about 0.1 wt % (e.g., from about 5 wt % to about 0.2 wt % or from about 2 wt % to about 0.3 wt %) of sodium lauryl sulfate, by weight of the composition.
[0144] Binders suitable for the invention enhance the tablet strength of the pharmaceutical composition and are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the chemical stability, the physical stability, or the biological activity of the pharmaceutical composition. Exemplary binders include polyvinylpyrrolidone, dibasic calcium phosphate, sucrose, corn (maize) starch, modified cellulose (e.g., hydroxymethyl cellulose), or any combination thereof.
[0145] Thus, in one embodiment, the pharmaceutical composition comprises a binder in an amount of at least about 0.1 wt % (e.g., at least about 1 wt %, at least about 3 wt %, at least about 4 wt %, or at least about 5 wt %) by weight of the composition. For example, the pharmaceutical composition comprises from about 0.1 wt % to about 10 wt % (e.g., from about 1 wt % to about 10 wt % or from about 2 wt % to about 7 wt %) of binder, by weight of the composition. In another example, the pharmaceutical composition comprises at least about 0.1 wt % (e.g., at least about 1 wt %, at least about 2 wt %, at least about 3 wt %, or at least about 4 wt %) of polyvinylpyrrolidone, by weight of the composition. In yet another example, the pharmaceutical composition comprises a glidant in an amount ranging from about 0.1 wt % to about 10 wt % (e.g., from about 1 wt % to about 8 wt % or from about 2 wt % to about 5 wt %) of polyvinylpyrrolidone, by weight of the composition.
[0146] Diluents suitable for the invention may add necessary bulk to a formulation to prepare tablets of the desired size and are generally compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the hardness, the chemical stability, the physical stability, or the biological activity of the pharmaceutical composition. Exemplary diluents include: sugars, for example, confectioner's sugar, compressible sugar, dextrates, dextrin, dextrose, lactose, mannitol, sorbitol, cellulose, and modified celluloses, for example, powdered cellulose, talc, calcium phosphate, starch, or any combination thereof.
[0147] Thus, in one embodiment, the pharmaceutical composition comprises a diluent in an amount of 40 wt % or less (e.g., 35 wt % or less, 30 wt % or less, or 25 wt % or less, or 20 wt % or less, or 15 wt % or less, or 10 wt % or less) by weight of the composition. For example, the pharmaceutical composition comprises from about 40 wt % to about 1 wt % (e.g., from about 35 wt % to about 5 wt % or from about 30 wt % to about 7 wt %, from about 25 wt % to about 10 wt %, from about 20 wt % to about 15 wt %) of diluent, by weight of the composition. In another example, the pharmaceutical composition comprises 40 wt % or less (e.g., 35 wt % or less, 25 wt % or less, or 15 wt % or less) of mannitol, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 35 wt % to about 1 wt % (e.g., from about 30 wt % to about 5 wt % or from about 25 wt % to about 10 wt %) of mannitol, by weight of the composition.
[0148] Glidants suitable for the invention enhance the flow properties of the pharmaceutical composition and are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the hardness, the chemical stability, the physical stability, or the biological activity of the pharmaceutical composition. Exemplary glidants include colloidal silicon dioxide, talc, or a combination thereof.
[0149] Thus, in one embodiment, the pharmaceutical composition comprises a glidant in an amount of 2 wt % or less (e.g., 1.75 wt %, 1.25 wt % or less, or 1.00 wt % or less) by weight of the composition. For example, the pharmaceutical composition comprises from about 2 wt % to about 0.05 wt % (e.g., from about 1.5 wt % to about 0.07 wt % or from about 1.0 wt % to about 0.09 wt %) of glidant, by weight of the composition. In another example, the pharmaceutical composition comprises 2 wt % or less (e.g., 1.75 wt %, 1.25 wt % or less, or 1.00 wt % or less) of colloidal silicon dioxide, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 2 wt % to about 0.05 wt % (e.g., from about 1.5 wt % to about 0.07 wt % or from about 1.0 wt % to about 0.09 wt %) of colloidal silicon dioxide, by weight of the composition.
[0150] In some embodiments, the pharmaceutical composition can include an oral solid pharmaceutical dosage form which can comprise a lubricant that can prevent adhesion of a granulate-bead admixture to a surface (e.g., a surface of a mixing bowl, a compression die and/or punch). A lubricant can also reduce interparticle friction within the granulate and improve the compression and ejection of compressed pharmaceutical compositions from a die press. The lubricant is also compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the hardness, or the biological activity of the pharmaceutical composition. Exemplary lubricants include magnesium stearate, calcium stearate, zinc stearate, sodium stearate, stearic acid, aluminum stearate, leucine, glyceryl behenate, hydrogenated vegetable oil or any combination thereof. In one embodiment, the pharmaceutical composition comprises a lubricant in an amount of 5 wt % or less (e.g., 4.75 wt %, 4.0 wt % or less, or 3.00 wt % or less, or 2.0 wt % or less) by weight of the composition. For example, the pharmaceutical composition comprises from about 5 wt % to about 0.10 wt % (e.g., from about 4.5 wt % to about 0.5 wt % or from about 3 wt % to about 1 wt %) of lubricant, by weight of the composition. In another example, the pharmaceutical composition comprises 5 wt % or less (e.g., 4.0 wt % or less, 3.0 wt % or less, or 2.0 wt % or less, or 1.0 wt % or less) of magnesium stearate, by weight of the composition. In yet another example, the pharmaceutical composition comprises from about 5 wt % to about 0.10 wt % (e.g., from about 4.5 wt % to about 0.15 wt % or from about 3.0 wt % to about 0.50 wt %) of magnesium stearate, by weight of the composition.
[0151] Pharmaceutical compositions of the invention can optionally comprise one or more colorants, flavors, and/or fragrances to enhance the visual appeal, taste, and/or scent of the composition. Suitable colorants, flavors, or fragrances are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the chemical stability, the physical stability, the hardness, or the biological activity of the pharmaceutical composition. In one embodiment, the pharmaceutical composition comprises a colorant, a flavor, and/or a fragrance. In one embodiment, the pharmaceutical compositions provided by the invention are purple.
[0152] In some embodiments, the pharmaceutical composition includes or can be made into tablets and the tablets can be coated with a colorant and optionally labeled with a logo, other image and/or text using a suitable ink. In still other embodiments, the pharmaceutical composition includes or can be made into tablets and the tablets can be coated with a colorant, waxed, and optionally labeled with a logo, other image and/or text using a suitable ink. Suitable colorants and inks are compatible with the ingredients of the pharmaceutical composition, i.e., they do not substantially reduce the solubility, the chemical stability, the physical stability, the hardness, or the biological activity of the pharmaceutical composition. The suitable colorants and inks can be any color and are water based or solvent based. In one embodiment, tablets made from the pharmaceutical composition are coated with a colorant and then labeled with a logo, other image, and/or text using a suitable ink. For example, tablets comprising pharmaceutical composition as described herein can be coated with about 3 wt % (e.g., less than about 6 wt % or less than about 4 wt %) of film coating comprising a colorant. The colored tablets can be labeled with a logo and text indicating the strength of the active ingredient in the tablet using a suitable ink. In another example, tablets comprising pharmaceutical composition as described herein can be coated with about 3 wt % (e.g., less than about 6 wt % or less than about 4 wt %) of a film coating comprising a colorant.
[0153] In another embodiment, tablets made from the pharmaceutical composition are coated with a colorant, waxed, and then labeled with a logo, other image, and/or text using a suitable ink. For example, tablets comprising pharmaceutical composition as described herein can be coated with about 3 wt % (e.g., less than about 6 wt % or less than about 4 wt %) of film coating comprising a colorant. The colored tablets can be waxed with Carnauba wax powder weighed out in the amount of about 0.01% w/w of the starting tablet core weight. The waxed tablets can be labeled with a logo and text indicating the strength of the active ingredient in the tablet using a suitable ink. In another example, tablets comprising pharmaceutical composition as described herein can be coated with about 3 wt % (e.g., less than about 6 wt % or less than about 4 wt %) of a film coating comprising a colorant The colored tablets can be waxed with Carnauba wax powder weighed out in the amount of about 0.01% w/w of the starting tablet core weight. The waxed tablets can be labeled with a logo and text indicating the strength of the active ingredient in the tablet using a pharmaceutical grade ink such as a black ink (e.g., Opacode.RTM. S-1-17823, a solvent based ink, commercially available from Colorcon, Inc. of West Point, Pa.).
[0154] One exemplary pharmaceutical composition comprises from about 15 wt % to about 70 wt % (e.g., from about 15 wt % to about 60 wt %, from about 15 wt % to about 50 wt %, or from about 15 wt % to about 40 wt %, or from about 20 wt % to about 70 wt %, or from about 30 wt % to about 70 wt %, or from about 40 wt % to about 70 wt %, or from about 50 wt % to about 70 wt %) of Compound 1, by weight of the composition. The aforementioned compositions can also include one or more pharmaceutically acceptable excipients, for example, from about 20 wt % to about 50 wt % of a filler; from about 1 wt % to about 5 wt % of a disintegrant; from about 2 wt % to about 0.3 wt % of a surfactant; from about 0.1 wt % to about 5 wt % of a binder; from about 1 wt % to about 30 wt % of a diluent; from about 2 wt % to about 0.05 wt % of a glidant; and from about 5 wt % to about 0.1 wt % of a lubricant. Or, the pharmaceutical composition comprises a composition containing from about 15 wt % to about 70 wt % (e.g., from about 20 wt % to about 40 wt %, from about 25 wt % to about 60 wt %, or from about 30 wt % to about 55 wt %) of Compound 1, by weight of the composition; and one or more excipients, for example, from about 20 wt % to about 50 wt % of a filler; from about 1 wt % to about 5 wt % of a disintegrant; from about 2 wt % to about 0.3 wt % of a surfactant; from about 0.1 wt % to about 5 wt % of a binder; from about 1 wt % to about 30 wt % of a diluent; from about 2 wt % to about 0.05 wt % of a glidant; and from about 5 wt % to about 0.1 wt % of a lubricant.
[0155] Another exemplary pharmaceutical composition comprises from about 15 wt % to about 70 wt % (e.g., from about 15 wt % to about 60 wt %, from about 15 wt % to about 50 wt %, or from about 15 wt % to about 40 wt % or from about 20 wt % to about 70 wt %, or from about 30 wt % to about 70 wt %, or from about 40 wt % to about 70 wt %, or from about 50 wt % to about 70 wt %) of Compound 1 by weight of the composition, and one or more excipients, for example, from about 20 wt % to about 50 wt % of a filler; from about 1 wt % to about 5 wt % of a disintegrant; from about 2 wt % to about 0.3 wt % of a surfactant; from about 0.1 wt % to about 5 wt % of a binder; from about 1 wt % to about 30 wt % of a diluent; from about 2 wt % to about 0.05 wt % of a glidant; and from about 2 wt % to about 0.1 wt % of a lubricant.
[0156] Another exemplary pharmaceutical composition comprises from about 15 wt % to about 70 wt % (e.g., from about 15 wt % to about 60 wt %, from about 15 wt % to about 50 wt %, or from about 15 wt % to about 40 wt % or from about 20 wt % to about 70 wt %, or from about 30 wt % to about 70 wt %, or from about 40 wt % to about 70 wt %, or from about 50 wt % to about 70 wt %) of Compound 1 by weight of the composition, and one or more excipients, for example, from about 20 wt % to about 50 wt % of a filler; from about 1 wt % to about 5 wt % of a disintegrant; from about 2 wt % to about 0.3 wt % of a surfactant; from about 0.1 wt % to about 5 wt % of a binder; from about 1 wt % to about 30 wt % of a diluent; from about 2 wt % to about 0.05 wt % of a glidant; and from about 2 wt % to about 0.1 wt % of a lubricant.
[0157] Another exemplary pharmaceutical composition comprises from about 15 wt % to about 70 wt % (e.g., from about 15 wt % to about 60 wt %, from about 15 wt % to about 50 wt %, or from about 15 wt % to about 40 wt % or from about 20 wt % to about 70 wt %, or from about 30 wt % to about 70 wt %, or from about 40 wt % to about 70 wt %, or from about 50 wt % to about 70 wt %) of Compound 1 and one or more excipients, for example, from about 20 wt % to about 50 wt % of a filler; from about 1 wt % to about 5 wt % of a disintegrant; from about 2 wt % to about 0.3 wt % of a surfactant; from about 0.1 wt % to about 5 wt % of a binder; from about 1 wt % to about 30 wt % of a diluent; from about 2 wt % to about 0.05 wt % of a glidant; and from about 2 wt % to about 0.1 wt % of a lubricant.
[0158] In one embodiment, the invention is a granular pharmaceutical composition comprising:
[0159] a. about 30 wt % of Compound 1 by weight of the composition;
[0160] b. about 42 wt % of microcrystalline cellulose by weight of the composition;
[0161] c. about 21 wt % of mannitol by weight of the composition;
[0162] d. about 3 wt % of sodium croscarmellose sodium by weight of the composition;
[0163] e. about 1 wt % of sodium lauryl sulfate by weight of the composition;
[0164] f. about 2 wt % of magnesium stearate by weight of the composition; and
[0165] g. about 0.5 wt % of colloidal silica by weight of the composition.
[0166] Another granular composition formulated into an oral formulation of the invention comprises:
[0167] a. about 50 wt % of Compound 1;
[0168] b. about 30 wt % of microcrystalline cellulose by weight of the composition;
[0169] c. about 13 wt % of mannitol by weight of the composition;
[0170] d. about 2 wt % of sodium croscarmellose sodium by weight of the composition;
[0171] e. about 4 wt % of polyvinylpyrrolidone by weight of the composition; and
[0172] f. about 1 wt % of sodium lauryl sulfate by weight of the composition.
[0173] In one embodiment, a pharmaceutical oral formulation of the invention comprises:
[0174] a. about 30 wt % of a Compound 1 by weight of the composition;
[0175] b. about 42 wt % of microcrystalline cellulose by weight of the composition;
[0176] c. about 21 wt % of mannitol by weight of the composition;
[0177] d. about 3 wt % of sodium croscarmellose sodium by weight of the composition;
[0178] e. about 1 wt % of sodium lauryl sulfate by weight of the composition;
[0179] f. about 2.5 wt % of magnesium stearate by weight of the composition; and
[0180] g. about 0.5 wt % of colloidal silica by weight of the composition.
[0181] Another pharmaceutical oral formulation of the invention comprises:
[0182] a. about 50 wt % of a Compound 1 by weight of the composition;
[0183] b. about 30 wt % of microcrystalline cellulose by weight of the composition;
[0184] c. about 13 wt % of mannitol by weight of the composition;
[0185] d. about 4 wt % of sodium croscarmellose sodium by weight of the composition;
[0186] e. about 4 wt % of polyvinylpyrrolidone by weight of the composition
[0187] f. about 1 wt % of sodium lauryl sulfate by weight of the composition; and
[0188] g. about 0.5 wt % of magnesium stearate by weight of the composition.
[0189] Another pharmaceutical oral formulation of the invention comprises:
[0190] a. about 60 wt % of a Compound 1 by weight of the composition;
[0191] b. about 20 wt % of microcrystalline cellulose by weight of the composition;
[0192] c. about 13 wt % of mannitol by weight of the composition;
[0193] d. about 4 wt % of sodium croscarmellose sodium by weight of the composition;
[0194] e. about 4 wt % of polyvinylpyrrolidone by weight of the composition
[0195] f. about 1 wt % of sodium lauryl sulfate by weight of the composition; and
[0196] g. about 0.5 wt % of magnesium stearate by weight of the composition.
[0197] Another pharmaceutical oral formulation of the invention comprises:
[0198] a. about 150 to 250 mg of Compound 1;
[0199] b. about 40 to 50 mg of mannitol;
[0200] c. about 120 to 130 mg of microcrystalline cellulose;
[0201] d. about 10 to 20 mg of croscarmellose sodium;
[0202] e. about 10 to 20 mg of polyvinylpyrrolidone;
[0203] f. about 1 to 5 mg of sodium lauryl sulfate; and
[0204] g. about 1 to 5 mg of magnesium stearate.
[0205] Another pharmaceutical oral formulation of the invention comprises:
[0206] a. about 200 mg of Compound 1;
[0207] b. about 43 mg of mannitol;
[0208] c. about 123 mg of microcrystalline cellulose;
[0209] d. about 15 mg of croscarmellose sodium;
[0210] e. about 13 mg of polyvinylpyrrolidone;
[0211] f. about 3 mg of sodium lauryl sulfate; and
[0212] g. about 4 mg of magnesium stearate.
[0213] Another pharmaceutical oral formulation of the invention comprises:
[0214] a. about 200 mg of Compound 1;
[0215] b. about 45 mg of mannitol;
[0216] c. about 123 mg of microcrystalline cellulose;
[0217] d. about 15 mg of croscarmellose sodium;
[0218] e. about 10.4 mg of polyvinylpyrrolidone;
[0219] f. about 2.6 mg of sodium lauryl sulfate; and
[0220] g. about 4 mg of magnesium stearate.
[0221] Another pharmaceutical oral formulation of the invention comprises:
[0222] a. about 70 wt % of a Compound 1 by weight of the composition;
[0223] b. about 12 wt % of microcrystalline cellulose by weight of the composition;
[0224] c. about 11 wt % of mannitol by weight of the composition;
[0225] d. about 4 wt % of sodium croscarmellose sodium by weight of the composition;
[0226] e. about 4 wt % of polyvinylpyrrolidone by weight of the composition
[0227] f. about 1 wt % of sodium lauryl sulfate by weight of the composition; and
[0228] g. about 0.5 wt % of magnesium stearate by weight of the composition.
[0229] The pharmaceutical compositions of the invention can be processed into a tablet form, capsule form, pouch form, lozenge form, or other solid form that is suited for oral administration. Thus in some embodiments, the pharmaceutical compositions are in tablet form.
[0230] In still another pharmaceutical oral formulation of the invention, a shaped pharmaceutical tablet composition having an initial hardness of 5-21 kP.+-.20 percent comprises: about 30 wt % of Compound 1; about 42 wt % of microcrystalline cellulose by weight of the composition; about 21 wt % of mannitol by weight of the composition; about 3 wt % of sodium croscarmellose sodium by weight of the composition; about 1 wt % of sodium lauryl sulfate by weight of the composition; about 2.5 wt % of magnesium stearate by weight of the composition; and about 0.5 wt % of colloidal silica by weight of the composition. Wherein the amount of Compound 1 in the shaped pharmaceutical tablet ranges from about 25 mg to about 250 mg, for example, 50 mg, or 75 mg, or 100 mg, or 150 mg, 200 mg, or 250 mg Compound 1 per tablet.
[0231] In still another pharmaceutical oral formulation of the invention, a shaped pharmaceutical tablet composition having an initial hardness of 5-21 kP.+-.20 percent comprises: about 49 wt % of a Compound 1; about 29 wt % of microcrystalline cellulose by weight of the composition; about 12.6 wt % of mannitol by weight of the composition; about 4 wt % of sodium croscarmellose sodium by weight of the composition; about 4 wt % of polyvinylpyrrolidone by weight of the composition; about 1 wt % of sodium lauryl sulfate by weight of the composition; and about 0.5 wt % of magnesium stearate by weight of the composition. The amount of Compound 1 in the shaped pharmaceutical tablet ranges from about 25 mg to about 250 mg, for example, 50 mg, or 75 mg, or 100 mg, or 150 mg, 200 mg, or 250 mg Compound 1 per tablet.
[0232] In certain embodiments, the shaped pharmaceutical tablet contains about 100 mg of Compound 1. In certain embodiments, the shaped pharmaceutical tablet contains about 200 mg of Compound 1.
[0233] Another aspect of the invention provides a pharmaceutical formulation consisting of a tablet or capsule that includes a Compound 1 and other excipients (e.g., a filler, a disintegrant, a surfactant, a binder, a glidant, a colorant, a lubricant, or any combination thereof), each of which is described above and in the Examples below, wherein the tablet has a dissolution of at least about 50% (e.g., at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 99%) in about 30 minutes. In one example, the pharmaceutical composition consists of a tablet that includes Compound 1 in an amount ranging from 25 mg to 250 mg, for example, 25 mg, or 50 mg, or 75 mg, or 100 mg, or 150 mg, 200 mg, or 250 mg and one or more excipients (e.g., a filler, a disintegrant, a surfactant, a binder, a glidant, a colorant, a lubricant, or any combination thereof), each of which is described above and in the Examples below, wherein the tablet has a dissolution of from about 50% to about 100% (e.g., from about 55% to about 95% or from about 60% to about 90%) in about 30 minutes. In another example, the pharmaceutical composition consists of a tablet that comprises a composition comprising Compound 1; and one or more excipients from: a filler, a diluent, a disintegrant, a surfactant, a binder, a glidant, and a lubricant, wherein the tablet has a dissolution of at least about 50% (e.g., at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 99%) in about 30 minutes.
[0234] In one embodiment, the tablet comprises a composition comprising at least about 25 mg (e.g., at least about 30 mg, at least about 40 mg, or at least about 50 mg) of Compound 1; and one or more excipients from: a filler, a diluent, a disintegrant, a surfactant, a binder, a glidant, and a lubricant. In another embodiment, the tablet comprises a composition comprising at least about 25 mg (e.g., at least about 30 mg, at least about 40 mg, at least about 50 mg, at least about 100 mg, or at least 150 mg) of Compound 1 and one or more excipients from: a filler, a diluent, a disintegrant, a surfactant, a binder, a glidant, and a lubricant.
[0235] Dissolution can be measured with a standard USP Type II apparatus that employs a dissolution media of 0.1% CTAB dissolved in 900 mL of DI water, buffered at pH 6.8 with 50 mM potassium phosphate monoasic, stirring at about 50-75 rpm at a temperature of about 37.degree. C. A single experimental tablet is tested in each test vessel of the apparatus. Dissolution can also be measured with a standard USP Type II apparatus that employs a dissolution media of 0.7% sodium lauryl sulfate dissolved in 900 mL of 50 mM sodium phosphate buffer (pH 6.8), stirring at about 65 rpm at a temperature of about 37.degree. C. A single experimental tablet is tested in each test vessel of the apparatus. Dissolution can also be measured with a standard USP Type II apparatus that employs a dissolution media of 0.5% sodium lauryl sulfate dissolved in 900 mL of 50 mM sodium phosphate buffer (pH 6.8), stirring at about 65 rpm at a temperature of about 37.degree. C. A single experimental tablet is tested in each test vessel of the apparatus.
Methods for Making Compound 1, Compound 1 Form I, Compound 1 Form II, Compound 1 HCl Salt Form A
Compound 1
[0236] Compound 1 is used as the starting point for the other solid state forms and can be prepared by coupling an acid chloride moiety with an amine moiety according to Schemes 1-4.
##STR00002##
[0237] Scheme 1 depicts the preparation of 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarbonyl chloride, which is used in Scheme 3 to make the amide linkage of Compound 1.
[0238] The starting material, 2,2-difluorobenzo[d][1,3]dioxole-5-carboxylic acid, is commercially available from Saltigo (an affiliate of the Lanxess Corporation). Reduction of the carboxylc acid moiety in 2,2-difluorobenzo[d][1,3]dioxole-5-carboxylic acid to the primary alcohol, followed by conversion to the corresponding chloride using thionyl chloride (SOCl.sub.2), provides 5-(chloromethyl)-2,2-difluorobenzo[d][1,3]dioxole, which is subsequently converted to 2-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)acetonitrile using sodium cyanide. Treatment of 2-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)acetonitrile with base and 1-bromo-2-chloroethane provides 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarbonitrile. The nitrile moiety in 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarbonitrile is converted to a carboxylic acid using base to give 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarboxylic acid, which is converted to the desired acid chloride using thionyl chloride.
##STR00003##
[0239] Scheme 2 depicts an alternative synthesis of the requisite acid chloride. 5-bromomethyl-2,2-difluoro-1,3-benzodioxole is coupled with ethyl cyanoacetate in the presence of a palladium catalyst to form the corresponding alpha cyano ethyl ester. Saponification of the ester moiety to the carboxylic acid gives the cyanoethyl compound. Alkylation of the cyanoethyl compound with 1-bromo-2-chloro ethane in the presence of base gives the cyanocyclopropyl compound. Treatment of the cyanocyclopropyl compound with base gives the carboxylate salt, which is converted to the carboxylic acid by treatment with acid. Conversion of the carboxylic acid to the acid chloride is then accomplished using a chlorinating agent such as thionyl chloride or the like.
##STR00004##
[0240] Scheme 3 depicts the preparation of the requisite tert-butyl 3-(6-amino-3-methylpyridin-2-yl)benzoate, which is coupled with 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarbonyl chloride in Scheme 3 to give Compound 1. Palladium-catalyzed coupling of 2-bromo-3-methylpyridine with 3-(tert-butoxycarbonyl)phenylboronic acid gives tert-butyl 3-(3-methylpyridin-2-yl)benzoate, which is subsequently converted to the desired compound.
##STR00005##
[0241] Scheme 4 depicts the coupling of 1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarbonyl chloride with tert-butyl 3-(6-amino-3-methylpyridin-2-yl)benzoate using triethyl amine and 4-dimethylaminopyridine to initially provide the tert-butyl ester of Compound 1.
Compound 1 Form I
[0242] Compound 1 Form I is prepared by dispersing or dissolving a salt form, such as the HCl salt, of Compound 1 in an appropriate solvent for an effective amount of time. Treatment of the tert-butyl ester with an acid such as HCl, gives the HCl salt of Compound 1, which is typically a crystalline solid. Compound 1 Form I may also be prepared directly from the t-butyl ester precursor by treatment with an appropriate acid, such as formic acid.
[0243] The HCl salt of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid can be used to make Form I by dispersing or dissolving the HCl salt of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid in an appropriate solvent for an effective amount of time. Other salts of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid may be used, such as, for example, salts derived from other mineral or organic acids. The other salts result from acid-mediated hydrolysis of the t-butyl ester moiety. Salts derived from other acids may include, for example, nitric, sulfuric, phosphoric, boric, acetic, benzoic and malonic. These salt forms of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2yl)benzoic acid may or may not be soluble, depending upon the solvent used, but lack of solubility does not hinder formation of Form I. For example, in one embodiment, the appropriate solvent may be water or an alcohol/water mixture such as 50% methanol/water mixture, even though the HCl salt form of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid is only sparingly soluble in water. In one embodiment, the appropriate solvent is water.
[0244] The effective amount of time for formation of Form I from the salt of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid can be any time between 2 to 24 hours or greater. It is recognized that the amount of time needed is inversely proportional to the temperature. That is, the higher the temperature the less time needed to affect dissociation of acid to form Form I. When the solvent is water, stirring the dispersion for approximately 24 hours at room temperature provides Form I in an approximately 98% yield. If a solution of the salt of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid is desired for process purposes, an elevated temperature may be used. After stirring the solution for an effective amount of time at the elevated temperature, recrystallization upon cooling provides substantially pure Form 1. In one embodiment, substantially pure refers to greater than about 90% purity. In another embodiment, substantially pure refers to greater than about 95% purity. In another embodiment, substantially pure refers to greater than about 98% purity. In another embodiment, substantially pure refers to greater than about 99% purity. The temperature selected depends in part on the solvent used and is well within the determination capabilities of one of ordinary skill in the art. In one embodiment, the temperature is between room temperature and about 80.degree. C. In another embodiment, the temperature is between room temperature and about 40.degree. C. In another embodiment, the temperature is between about 40.degree. C. and about 60.degree. C. In another embodiment, the temperature is between about 60.degree. C. and about 80.degree. C.
[0245] Compound 1 Form I may also be formed directly from 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (cf. Scheme 3), which is a precursor to the salt of Compound 1. Thus, 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate is allowed to undergo reaction with an appropriate acid, such as, for example, formic acid under appropriate reaction conditions to give Compound 1 Form I.
[0246] Compound 1 Form I may be further purified by recrystallization from an organic solvent. Examples of organic solvents include, but are not limited to, toluene, cumene, anisol, 1-butanol, isopropyl acetate, butyl acetate, isobutyl acetate, methyl t-butyl ether, methyl isobutyl ketone and 1-propanol-water mixtures. The temperature may be as described above. For example, Form I is dissolved in 1-butanol at 75.degree. C. until it is completely dissolved. Cooling down the solution to 10.degree. C. at a rate of 0.2.degree. C./min yields crystals of Form I which may be isolated by filtration.
[0247] In one embodiment, Compound 1 Form I is characterized by one or more peaks at 15.2 to 15.6 degrees, 16.1 to 16.5 degrees, and 14.3 to 14.7 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation. In another embodiment, Compound 1 Form I is characterized by one or more peaks at 15.4, 16.3, and 14.5 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 14.6 to 15.0 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 14.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 17.6 to 18.0 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 17.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 16.4 to 16.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 16.4 to 16.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 16.6 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 7.6 to 8.0 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 7.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 25.8 to 26.2 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 26.0 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 21.4 to 21.8 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 21.6 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 23.1 to 23.5 degrees. In another embodiment, Compound 1 Form I is further characterized by a peak at 23.3 degrees. In some embodiments, Compound 1 Form I is characterized by a diffraction pattern substantially similar to that of FIG. 1. In some embodiments, Compound 1 Form I is characterized by a diffraction pattern substantially similar to that of FIG. 2.
[0248] In some embodiments, the particle size distribution of D90 is about 82 .mu.m or less for Compound 1 Form I. In some embodiments, the particle size distribution of D50 is about 30 or less for Compound 1 Form I.
Compound 1 Form II
[0249] Compound 1 Form II is prepared by slurrying Compound 1 Form I in an appropriate solvent at a sufficient concentration for a sufficient time. The slurry is then filtered centrifugally or under vacuum and dried at ambient conditions for sufficient time to yield Compound 1 Form II.
[0250] In some embodiments, about 20 to 40 mg of Compound 1 Form I is slurried in about 400 to 600 .mu.L of an appropriate solvent. In another embodiment, about 25 to 35 mg of Compound 1 Form I is slurried in about 450 to 550 .mu.L of an appropriate solvent. In another embodiment, about 30 mg of Compound 1 Form I is slurried in about 500 .mu.L of an appropriate solvent.
[0251] In some embodiments, the time that Compound 1 Form I is allowed to slurry with the solvent is froml hour to four days. More particularly, the time that Compound 1 Form I is allowed to slurry with the solvent is from l to 3 days. More particularly, the time is 2 days.
[0252] In some embodiments, the appropriate solvent is selected from an organic solvent of sufficient size to fit the voids in the crystalline lattice of Compound 1 Form II. In other embodiments, the solvate is of sufficient size to fit in voids measuring about 100 .ANG..sup.3.
[0253] In other embodiments, the solvent is selected from the group consisting of methanol, ethanol, acetone, 2-propanol, acetonitrile, tetrahydrofuran, methyl acetate, 2-butanone, ethyl formate, and 2-methyl tetrahydrofuran.
[0254] In other embodiments, a mixture of two or more of these solvents may be used to obtain Compound 1 Form II. Alternatively, Compound 1 Form II may be obtained from a mixture comprising one or more of these solvents and water.
[0255] In some embodiments, the effective amount of time for drying Compound 1 Form II is 1 to 24 hours. More particularly, the time is 6 to 18 hours. More particularly, the time is about 12 hours.
[0256] In another embodiment, Compound 1 Form II is prepared by dispersing or dissolving a salt form of Compound 1, such as an HCl salt of Compound 1 in an appropriate solvent for an effective amount of time.
[0257] Compound 1 Form II as disclosed herein comprises a crystalline lattice of Compound 1 in which voids in the crystalline lattice are empty, or occupied, or partially occupied by one or more molecules of a suitable solvent. Suitable solvents include, but are not limited to, methanol, ethanol, acetone, 2-propanol, acetonitrile, tetrahydrofuran, methyl acetate, 2-butanone, ethyl formate, and 2-methyl tetrahydrofuran. Certain physical characterisics of Compound 1 isostructural solvate forms, such as X-ray powder diffraction, melting point and DSC, are not substantially affected by the particular solvent molecule in question.
[0258] In one embodiment, Compound 1 Form II is characterized by one or more peaks at 21.50 to 21.90 degrees, 8.80 to 9.20 degrees, and 10.80 to 11.20 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation. In another embodiment, Compound 1 Form II is characterized by one or more peaks at 21.50 to 21.90 degrees, 8.80 to 9.20 degrees, 10.80 to 11.20 degrees, 18.00 to 18.40 degrees, and 22.90 to 23.30 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation. In another embodiment, Compound 1 Form II is characterized by one or more peaks at 21.70, 8.98, and 11.04 degrees. In another embodiment, Compound 1 Form II is characterized by one or more peaks at 21.70, 8.98, 11.04, 18.16, and 23.06 degrees. In another embodiment, Compound 1 Form II is characterized by a peak at 21.50 to 21.90 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 21.70 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 8.80 to 9.20 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 8.98 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 10.80 to 11.20 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 11.04. In another embodiment, Compound 1 Form II is further characterized by a peak at 18.00 to 18.40 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 18.16 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 22.90 to 23.30 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 23.06 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 20.40 to 20.80 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 20.63 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 22.00 to 22.40 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 22.22 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 18.40 to 18.80 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 18.57 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 16.50 to 16.90 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 16.66 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 19.70 to 20.10 degrees. In another embodiment, Compound 1 Form II is further characterized by a peak at 19.86 degrees.
[0259] In some embodiments, Compound 1 Form II is characterized by a diffraction pattern substantially similar to that of FIG. 3. In some embodiments, Compound 1 Form II is characterized by diffraction patterns substantially similar to those provided in FIG. 4.
[0260] In another embodiment, the solvate that forms Compound 1 Form II is selected from the group consisting of methanol, ethanol, acetone, 2-propanol, acetonitrile, tetrahydrofuran, methyl acetate, 2-butanone, ethyl formate, and 2-methyl tetrahydrofuran. Diffraction patterns are provided for the following Compound 1 Form II: methanol (FIG. 5), ethanol (FIG. 6), acetone (FIG. 7), 2-propanol (FIG. 8), acetonitrile (FIG. 9), tetrahydrofuran (FIG. 10), methyl acetate (FIG. 11), 2-butanone (FIG. 12), ethyl formate (FIGS. 13), and 2-methytetrahydrofuran (FIG. 14).
[0261] In another embodiment, the invention provides Compound 1 Form II which exhibits two or more phase transitions as determined by DSC or a similar analytic method known to the skilled artisan. In some embodiments, the DSC of Compound 1 Form II is substantially similar to the DSC trace depicted in FIG. 15. In another embodiment of this aspect, the DSC gives two phase transitions. In another embodiment, the DSC gives three phase transitions. In another embodiment, one of the phase transitions occurs between 200 and 207.degree. C. In another embodiment, one of the phase transitions occurs between 204 and 206.degree. C. In another embodiment, one of the phase transitions occurs between 183 and 190.degree. C. In another embodiment, one of the phase transitions occurs between 185 and 187.degree. C. In another embodiment, the melting point of Compound 1, Solvate Form A is between 183.degree. C. to 190.degree. C. In another embodiment, the melting point of Compound 1, Solvate Form A is between 185.degree. C. to 187.degree. C.
[0262] In another embodiment, Compound 1 Form II comprises 1 to 10 weight percent (wt. %) solvate as determined by TGA. In some embodiments, the TGA of Compound 1 Form II is substantially similar to the TGA trace depicted in FIG. 16. In another embodiment, Compound 1 Form II comprises 2 to 5 wt. % solvate as determined by TGA or a similar analytic method known to the skilled artisan.
[0263] In another embodiment, the conformation of Compound 1 Form II acetone solvate is substantially similar to that depicted in FIG. 17, which is based on single X-ray analysis.
[0264] In another embodiment, Compound 1 Form II acetone solvate has a P2.sub.1/n space group, and the following unit cell dimensions:
a=16.5235 (10) .ANG. .alpha.=90.degree.
b=12.7425 (8) .ANG. .beta.=103.736 (4).degree.
c=20.5512 (13) .ANG. .gamma.=90.degree..
Compound 1 HCl Salt Form A
[0265] Compound 1 HCl Salt Form A can be prepared from the HCl salt of Compound 1, by dissolving the HCl salt of Compound 1 in a minimum of solvent and removing the solvent by slow evaporation. In another embodiment, the solvent is an alcohol. In another embodiment, the solvent is ethanol. Slow evaporation is generally carried out by impeding the evaporation of the solvent. For example, in one embodiment, slow evaporation involves dissolving the HCl salt of Compound 1 in a vial and covering the vial with parafilm that contains a hole poked in it.
[0266] In one embodiment, Compound 1 HCl Salt Form A is characterized by one or more peaks at 8.80 to 9.20 degrees, 17.30 to 17.70 degrees, and 18.20 to 18.60 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation. In another embodiment, Compound 1 HCl Salt Form A is characterized by one or more peaks at 8.80 to 9.20 degrees, 17.30 to 17.70 degrees, 18.20 to 18.60 degrees, 10.10 to 10.50, and 15.80 to 16.20 degrees in an X-ray powder diffraction obtained using Cu K alpha radiation. In another embodiment, Compound 1 HCl Salt Form A is characterized by one or more peaks at 8.96, 17.51, and 18.45 degrees. In another embodiment, Compound 1 HCl Salt Form A is characterized by one or more peaks at 8.96, 17.51, 18.45. 10.33, and 16.01 degrees. In another embodiment, Compound 1 HCl Salt Form A is characterized by a peak at 8.80 to 9.20 degrees. In another embodiment, Compound 1 HCl Salt Form A is characterized by a peak at 8.96 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 17.30 to 17.70 degrees. In another embodiment, Compound 1 HCl Salt Form A is characterized by a peak at 17.51 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 18.20 to 18.60 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 18.45degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 10.10 to 10.50 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 10.33 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 15.80 to 16.20 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 16.01 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 11.70 to 12.10 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 11.94 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 7.90 to 8.30 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 8.14 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 9.90 to 10.30 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 10.10 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 16.40 to 16.80 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 16.55 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 9.30 to 9.70 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 9.54 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 16.40 to 16.80 degrees. In another embodiment, Compound 1 HCl Salt Form A is further characterized by a peak at 16.55 degrees. In some embodiments, Compound 1 HCl Salt Form A is characterized as a dimer as depicted in FIG. 18.
[0267] In some embodiments, Compound 1 HCl Salt Form A is characterized by a diffraction pattern substantially similar to that of FIG. 19.
[0268] In another embodiment, the invention features crystalline Compound 1 HCl Salt Form A having a P.sup.-1 space group, and the following unit cell dimensions:
a=10.2702 (2) .ANG. .alpha.=67.0270 (10).degree.
b=10.8782 (2) .ANG. .beta.=66.1810 (10).degree.
c=12.4821 (3) .ANG. .gamma.=72.4760 (10).degree..
Methods For Making the Pharmaceutical Compositions
[0269] The dosage unit forms of the invention can be produced by compacting or compressing an admixture or composition, for example, a powder or granules, under pressure to form a stable three-dimensional shape (e.g., a tablet). As used herein, "tablet" includes compressed pharmaceutical dosage unit forms of all shapes and sizes, whether coated or uncoated.
[0270] The expression "dosage unit form" as used herein refers to a physically discrete unit of agent appropriate for the patient to be treated. In general, a compacted mixture has a density greater than that of the mixture prior to compaction. A dosage unit form of the invention can have almost any shape including concave and/or convex faces, rounded or angled corners, and a rounded to rectilinear shape. In some embodiments, the compressed dosage forms of the invention comprise a rounded tablet having flat faces. The solid pharmaceutical dosage forms of the invention can be prepared by any compaction and compression method known by persons of ordinary skill in the art of forming compressed solid pharmaceutical dosage forms. In particular embodiments, the formulations provided herein may be prepared using conventional methods known to those skilled in the field of pharmaceutical formulation, as described, e.g., in pertinent textbooks. See, e.g., Remington: The Science and Practice of Pharmacy, 21st Ed., Lippincott Williams & Wilkins, Baltimore, Md. (2003); Ansel et al., Pharmaceutical Dosage Forms And Drug Delivery Systems, 7th Edition, Lippincott Williams & Wilkins, (1999); The Handbook of Pharmaceutical Excipients, 4.sup.th edition, Rowe et al., Eds., American Pharmaceuticals Association (2003); Gibson, Pharmaceutical Preformulation And Formulation, CRC Press (2001), these references hereby incorporated herein by reference in their entirety.
Granulation and Compression
[0271] In some embodiments, solid forms, including powders comprising the active agent Compound 1 and the included pharmaceutically acceptable excipients (e.g. filler, diluent, disintegrant, surfactant, glidant, binder, lubricant, or any combination thereof) can be subjected to a dry granulation process. The dry granulation process causes the powder to agglomerate into larger particles having a size suitable for further processing. Dry granulation can improve the flowability of a mixture in order to be able to produce tablets that comply with the demand of mass variation or content uniformity.
[0272] Formulations as described herein may be produced using one or more mixing and dry granulations steps. The order and the number of the mixing and granulation steps do not seem to be critical. However, at least one of the excipients and Compound 1 can be been subject to dry granulation or wet high shear granulation before compression into tablets. Dry granulation of Compound 1 and the excipients made together prior to tablet compression seem, surprisingly, to be a simple, inexpensive and efficient way of providing close physical contact between the ingredients of the present compositions and formulations and thus results in a tablet formulation with good stability properties. Dry granulation can be carried out by a mechanical process, which transfers energy to the mixture without any use of any liquid substances (neither in the form of aqueous solutions, solutions based on organic solutes, or mixtures thereof) in contrast to wet granulation processes, also contemplated herein. Generally, the mechanical process requires compaction such as the one provided by roller compaction. An example of an alternative method for dry granulation is slugging.
[0273] In some embodiments, roller compaction is a granulation process comprising highly intensive mechanical compacting of one or more substances. In some embodiments, a pharmaceutical composition comprising an admixture of powders is pressed, that is roller compacted, between 2 counter rotating rollers to make a solid sheet which is subsequently crushed in a sieve to form a particulate matter. In this particulate matter, a close mechanical contact between the ingredients can be obtained. An example of roller compaction equipment is Minipactor.RTM. a Gerteis 3W-Polygran from Gerteis Maschinen+Processengineering A G.
[0274] In some embodiments, tablet compression according to the invention can occur without any use of any liquid substances (neither in the form of aqueous solutions, solutions based on organic solutes, or mixtures thereof), i.e. a dry granulation process. In a typical embodiment the resulting core or tablet has a compressive strength in the range of 1 to 15 kP; such as 1.5 to 12.5 kP, preferably in the range of 2 to 10 kP.
Brief Manufacturing Procedure
[0275] In some embodiments, the ingredients are weighed according to the formula set herein. Next, all of the intragranular ingredients are sifted and mixed well. The ingredients can be lubricated with a suitable lubricant, for example, magnesium stearate. The next step can comprise compaction/slugging of the powder admixture and sized ingredients. Next, the compacted or slugged blends are milled into granules and sifted to obtain the desired size. Next, the granules can be further lubricated with, for example, magnesium stearate. Next the granular composition of the invention can be compressed on suitable punches into various pharmaceutical formulations in accordance with the invention. Optionally the tablets can be coated with a film, colorant or other coating.
[0276] Another aspect of the invention provides a method for producing a pharmaceutical composition comprising providing an admixture of a composition comprising Compound 1 and one or more excipients selected from: a filler, a diluent, a binder, a glidant, a surfactant, a lubricant, a disintegrant, and compressing the composition into a tablet having a dissolution of at least about 50% in about 30 minutes.
[0277] In another embodiment, a wet granulation process is performed to yield the pharmaceutical formulation of the invention from an admixture of powdered and liquid ingredients. For example, a pharmaceutical composition comprising an admixture of a composition comprising Compound 1 and one or more excipients selected from: a filler, a diluent, a binder, a glidant, a surfactant, a lubricant, a disintegrant, are weighed as per the formula set herein. Next, all of the intragranular ingredients are sifted and mixed in a high shear or low shear granulator or a twin screw granulator using water or water with a surfactant or water with a binder or water with a surfactant and a binder to granulate the powder blend. A fluid other than water can also be used with or without surfactant and/or binder to granulate the powder blend. Next, the wet granules can optionally be milled using a suitable mill. Next, water may optionally be removed from the admixture by drying the ingredients in any suitable manner. Next, the dried granules can optionally be milled to the required size. Next, extra granular excipients can be added by blending (for example a filler, a diluent, and a disintegrant). Next, the sized granules can be further lubricated with magnesium stearate and a disintegrant, for example, croscarmellose sodium. Next the granular composition of the invention can be compressed on suitable punches into various pharmaceutical formulations in accordance with the invention. Optionally, the tablets can be coated with a film, colorant or other coating.
[0278] In a particularly favored embodiment, the pharmaceutical compositions of the present invention are prepared by a continuous twin screw wet granulation (TSWG) process. Continuous manufacturing delivers high quality and highly consistent product with on-line monitoring and control. Continuous manufacturing also facilitates quality by design development with a "data rich" design space and an easier to understand impact of upstream variables on the downstream process and final product quality. In addition, the pharmaceutical compositions of the present invention can be finalized early on commercial scale equipment which avoids scale-up risks and formulation changes late in development. Finally, continuous manufacturing has commercial manufacturing advantages such as improved process control, reduced product handling, and real time release efficiencies. The overall result is a more robust, controllable, and scalable process that has fewer process checks resulting in increased product quality and therefore greater patient safety.
[0279] For example, high shear granulation (HSG), a common granulation technique is well known for the risk of over-granulation and poor process control. Scale-up of this process is very challenging and involves significant risk. Changing from a HSG process to a continuous TSWG process, allows scale-up using the same equipment to produce different batch sizes, by running for a longer time. This eliminates the scale-up risk commonly encountered with wet granulation processes. Additionally, it was found that the TSWG process is more robust, being less sensitive to over-granulation. As can be seen in FIG. 28 for a Compound 1 tablet, the HSG process showed significant dissolution slow-down with increasing water content, while the TSWG process did not show a change for a similar range of water addition. Surprisingly, no performance changes were found with the tablet formulations comprising Compound 1 between 45-55 percent by weight and the tablet formulations comprising Compound 1 between 60-70 percent by weight using the twin screw wet granulation process. This was not the case with the HSG process. Additionally, this continuous and increased product quality process addresses a common complaint by the FDA regarding the lack of drug availability for patients in need thereof.
[0280] In one embodiment the continuous process starts with feeding individual excipients and Compound 1 into a continuous in-line blender through loss-in-weight feeding. From this blender, the material is continuously conveyed and processed through twin screw wet granulation, drying, milling, extra-granular excipient addition, blending, compression and film coating.
[0281] For example, in one embodiment, a tablet comprising Compound 1 may be prepared continuously according to the below flow chart.
[0282] Each of the ingredients of this exemplary admixture is described above and in the Examples below. Furthermore, the admixture can comprise optional additives, such as, one or more colorants, one or more flavors, and/or one or more fragrances as described above and in the Examples below. In some embodiments, the relative concentrations (e.g., wt %) of each of these ingredients (and any optional additives) in the admixture are also presented above and in the Examples below. The ingredients constituting the admixture can be provided sequentially or in any combination of additions; and, the ingredients or combination of ingredients can be provided in any order. In one embodiment, the lubricant is the last component added to the admixture.
[0283] In another embodiment, the admixture comprises a composition of Compound 1, and any one or more of the excipients; a binder, a glidant, a surfactant, a diluent, a lubricant, a disintegrant, and a filler, wherein each of these ingredients is provided in a powder form (e.g., provided as particles having a mean or average diameter, measured by light scattering, of 250 .mu.m or less (e.g., 150 .mu.m or less, 100 .mu.m or less, 50 .mu.m or less, 45 .mu.m or less, 40 .mu.m or less, or 35 .mu.m or less)). For instance, the admixture comprises a composition of Compound 1, a diluent, a glidant, a surfactant, a lubricant, a disintegrant, and a filler, wherein each of these ingredients is provided in a powder form (e.g., provided as particles having a mean diameter, measured by light scattering, of 250 .mu.m or less (e.g., 150 .mu.m or less, 100 .mu.m or less, 50 .mu.m or less, 45 .mu.m or less, 40 .mu.m or less, or 35 .mu.m or less)). In another example, the admixture comprises a composition of Compound 1, a diluent, a binder, a surfactant, a lubricant, a disintegrant, and a filler, wherein each of these ingredients is provided in a powder form (e.g., provided as particles having a mean diameter, measured by light scattering, of 250 .mu.m or less (e.g., 150 .mu.m or less, 100 .mu.m or less, 50 .mu.m or less, 45 .mu.m or less, 40 .mu.m or less, or 35 .mu.m or less))
[0284] In another embodiment, the admixture comprises a composition of Compound 1, and any combination of: a binder, a glidant, a diluent, a surfactant, a lubricant, a disintegrant, and a filler, wherein each of these ingredients is substantially free of water. Each of the ingredients comprises less than 5 wt % (e.g., less than 2 wt %, less than 1 wt %, less than 0.75 wt %, less than 0.5 wt %, or less than 0.25 wt %) of water by weight of the ingredient. For instance, the admixture comprises a composition of Compound 1, a diluent, a glidant, a surfactant, a lubricant, a disintegrant, and a filler, wherein each of these ingredients is substantially free of water. In some embodiments, each of the ingredients comprises less than 5 wt % (e.g., less than 2 wt %, less than 1 wt %, less than 0.75 wt %, less than 0.5 wt %, or less than 0.25 wt %) of water by weight of the ingredient.
[0285] In another embodiment, compressing the admixture into a tablet is accomplished by filling a form (e.g., a mold) with the admixture and applying pressure to admixture. This can be accomplished using a die press or other similar apparatus. In some embodiments, the admixture of Compound 1 and excipients can be first processed into granular form. The granules can then be sized and compressed into tablets or formulated for encapsulation according to known methods in the pharmaceutical art. It is also noted that the application of pressure to the admixture in the form can be repeated using the same pressure during each compression or using different pressures during the compressions. In another example, the admixture of powdered ingredients or granules can be compressed using a die press that applies sufficient pressure to form a tablet having a dissolution of about 50% or more at about 30 minutes (e.g., about 55% or more at about 30 minutes or about 60% or more at about 30 minutes). For instance, the admixture is compressed using a die press to produce a tablet hardness of at least about 5 kP (at least about 5.5 kP, at least about 6 kP, at least about 7 kP, at least about 10 kP, or at least 15 kP). In some instances, the admixture is compressed to produce a tablet hardness of between about 5 and 20 kP.
[0286] In some embodiments, tablets comprising a pharmaceutical composition as described herein can be coated with about 3.0 wt % of a film coating comprising a colorant by weight of the tablet. In certain instances, the colorant suspension or solution used to coat the tablets comprises about 20% w/w of solids by weight of the colorant suspension or solution. In still further instances, the coated tablets can be labeled with a logo, other image or text.
[0287] In another embodiment, the method for producing a pharmaceutical composition comprises providing an admixture of a solid forms, e.g. an admixture of powdered and/or liquid ingredients, the admixture comprising Compound 1 and one or more excipients selected from: a binder, a glidant, a diluent, a surfactant, a lubricant, a disintegrant, and a filler; mixing the admixture until the admixture is substantially homogenous, and compressing or compacting the admixture into a granular form. Then the granular composition comprising Compound 1 can be compressed into tablets or formulated into capsules as described above or in the Examples below. Alternatively, methods for producing a pharmaceutical composition comprises providing an admixture of Compound 1, and one or more excipients, e.g. a binder, a glidant, a diluent, a surfactant, a lubricant, a disintegrant, and a filler; mixing the admixture until the admixture is substantially homogenous, and compressing/compacting the admixture into a granular form using a roller compactor using a dry granulation composition as set forth in the Examples below or alternatively, compressed/compacted into granules using a high shear wet granule compaction process as set forth in the Examples below. Pharmaceutical formulations, for example a tablet as described herein, can be made using the granules prepared incorporating Compound 1 in addition to the selected excipients described herein.
[0288] In some embodiments, the admixture is mixed by stirring, blending, shaking, or the like using hand mixing, a mixer, a blender, any combination thereof, or the like. When ingredients or combinations of ingredients are added sequentially, mixing can occur between successive additions, continuously throughout the ingredient addition, after the addition of all of the ingredients or combinations of ingredients, or any combination thereof. The admixture is mixed until it has a substantially homogenous composition.
[0289] In another embodiment, the present invention comprises jet milling Compound 1, Compound 1 Form I, Compound 1 Form II, Compound 1 HCl Salt Form A in a suitable, conventional milling apparatus using air pressure suitable to produce particles having a significant particle size fraction between 0.1 microns and 50 microns. In another embodiment, the particle size is between 0.1 microns and 20 microns. In another embodiment, the particles size is between 0.1 microns and 10 microns. In another embodiment, the particle size is between 1.0 microns and 5 microns. In still another embodiment, Compound 1, Compound 1 Form I, Compound 1 Form II, Compound 1 HCl Salt Form A has a particle size D50 of 2.0 microns.
[0290] In various embodiments, a second therapeutic agent can be formulated together with Compound 1 to form a unitary or single dose form, for example, a tablet or capsule.
[0291] Dosage forms prepared as above can be subjected to in vitro dissolution evaluations according to Test 711 "Dissolution" in United States Pharmacopoeia 29, United States Pharmacopeial Convention, Inc., Rockville, Md., 2005 ("USP"), to determine the rate at which the active substance is released from the dosage forms. The content of active substance and the impurity levels are conveniently measured by techniques such as high performance liquid chromatography (HPLC).
[0292] In some embodiments, the invention includes use of packaging materials such as containers and closures of high-density polyethylene (HDPE), low-density polyethylene (LDPE) and or polypropylene and/or glass, glassine foil, aluminum pouches, and blisters or strips composed of aluminum or high-density polyvinyl chloride (PVC), optionally including a desiccant, polyethylene (PE), polyvinylidene dichloride (PVDC), PVC/PE/PVDC, and the like. These package materials can be used to store the various pharmaceutical compositions and formulations in a sterile fashion after appropriate sterilization of the package and its contents using chemical or physical sterilization techniques commonly employed in the pharmaceutical arts.
Methods For Administering the Pharmaceutical Compositions
[0293] In one aspect, the pharmaceutical compositions of the invention can be administered to a patient once daily or about every twenty four hours. Alternatively, the pharmaceutical compositions of the invention can be administered to a patient twice daily or about every twelve hours. These pharmaceutical compositions are administered as oral formulations containing about 25 mg, 50 mg, 100 mg, 125 mg, 150 mg, 200 mg, 250 mg, or 400 mg of Compound 1. In this aspect, in addition to Compound 1, the pharmaceutical compositions comprise a filler; a diluent; a disintegrant; a surfactant; at least one of a binder and a glidant; and a lubricant. For instance, a dose of 400 mg of Compound 1, may comprise two tablets of the invention each containing 200 mg of Compound 1, or four tablets of the invention each containing 100 mg of Compound 1.
[0294] It will also be appreciated that the compound and pharmaceutically acceptable compositions and formulations of the invention can be employed in combination therapies; that is, Compound 1 and pharmaceutically acceptable compositions thereof can be administered concurrently with, prior to, or subsequent to, one or more other desired therapeutics or medical procedures. The particular combination of therapies (therapeutics or procedures) to employ in a combination regimen will take into account compatibility of the desired therapeutics and/or procedures and the desired therapeutic effect to be achieved. It will also be appreciated that the therapies employed may achieve a desired effect for the same disorder (for example, an inventive compound may be administered concurrently with another agent used to treat the same disorder), or they may achieve different effects (e.g., control of any adverse effects). As used herein, additional therapeutic agents that are normally administered to treat or prevent a particular disease, for example, a CFTR mediated disease, or condition, are known as "appropriate for the disease or condition being treated."
[0295] In one embodiment, the additional therapeutic agent is selected from a mucolytic agent, bronchodialator, an antibiotic, an anti-infective agent, an anti-inflammatory agent, a CFTR modulator other than Compound 1 of the invention, or a nutritional agent.
[0296] In one embodiment, the additional agent is (R)-1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)-N-(1-(2,3-dihydroxypropyl)-6- -fluoro-2-(1-hydroxy-2-methylpropan-2-yl)-1H-indol-5-yl)cyclopropanecarbox- amide. In another embodiment, the additional agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide. In another embodiment, the additional agent is selected from Table 1:
TABLE-US-00024 TABLE 1 ##STR00006## 1 ##STR00007## 2 ##STR00008## 3 ##STR00009## 4 ##STR00010## 5 ##STR00011## 6 ##STR00012## 7 ##STR00013## 8 ##STR00014## 9 ##STR00015## 10 ##STR00016## 11 ##STR00017## 12 ##STR00018## 13 ##STR00019## 14
[0297] In another embodiment, the additional agent is any combination of the above agents. For example, the composition may comprise Compound 1, (R)-1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)-N-(1-(2,3-dihydroxypropyl)-6- -fluoro-2-(1-hydroxy-2-methylpropan-2-yl)-1H-indol-5-yl)cyclopropanecarbox- amide, and N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carb- oxamide. In another example, the composition may comprise Compound 1, N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide, and any one of the compounds from Table 1, i.e. compounds 1 through 14 of Table 1, or any combination thereof.
[0298] In one embodiment, the additional therapeutic agent is an antibiotic. Exemplary antibiotics useful herein include tobramycin, including tobramycin inhaled powder (TIP), azithromycin, aztreonam, including the aerosolized form of aztreonam, amikacin, including liposomal formulations thereof, ciprofloxacin, including formulations thereof suitable for administration by inhalation, levoflaxacin, including aerosolized formulations thereof, and combinations of two antibiotics, e.g., fosfomycin and tobramycin.
[0299] In another embodiment, the additional agent is a mucolyte. Exemplary mucolytes useful herein includes Pulmozyme.RTM..
[0300] In another embodiment, the additional agent is a bronchodialator. Exemplary bronchodialtors include albuterol, metaprotenerol sulfate, pirbuterol acetate, salmeterol, or tetrabuline sulfate.
[0301] In another embodiment, the additional agent is effective in restoring lung airway surface liquid. Such agents improve the movement of salt in and out of cells, allowing mucus in the lung airway to be more hydrated and, therefore, cleared more easily. Exemplary such agents include hypertonic saline, denufosol tetrasodium ([[(3S,5R)-5-(4-amino-2-oxopyrimidin-1-yl)-3-hydroxyoxolan-2-yl]methoxy-h- ydroxyphosphoryl] [[[(2R,3 S,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-3, 4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-hydroxyphosphoryl]hy- drogen phosphate), or bronchitol (inhaled formulation of mannitol).
[0302] In another embodiment, the additional agent is an anti-inflammatory agent, i.e., an agent that can reduce the inflammation in the lungs. Exemplary such agents useful herein include ibuprofen, docosahexanoic acid (DHA), sildenafil, inhaled glutathione, pioglitazone, hydroxychloroquine, or simavastatin.
[0303] In another embodiment, the additional agent is a CFTR modulator other than Compound 1, i.e., an agent that has the effect of modulating CFTR activity. Exemplary such agents include ataluren ("PTC124.RTM."; 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]benzoic acid), sinapultide, lancovutide, depelestat (a human recombinant neutrophil elastase inhibitor), and cobiprostone (7-{(2R, 4aR, 5R, 7aR)-2-[(3S)-1,1-difluoro-3-methylpentyl]-2-hydroxy-6-oxooctahydrocyclope- nta[b]pyran-5-yl}heptanoic acid).
[0304] In another embodiment, the additional agent is a nutritional agent. Exemplary nutritional agents include pancrelipase (pancreating enzyme replacement), including Pancrease.RTM., Pancreacarb.RTM., Ultrase.RTM., or Creon.RTM., Liprotomase.RTM. (formerly Trizytek.RTM.), Aquadeks.RTM., or glutathione inhalation. In one embodiment, the additional nutritional agent is pancrelipase.
[0305] In another embodiment, the additional agent is a compound selected from gentamicin, curcumin, cyclophosphamide, 4-phenylbutyrate, miglustat, felodipine, nimodipine, Philoxin B, geniestein, Apigenin, cAMP/cGMP modulators such as rolipram, sildenafil, milrinone, tadalafil, amrinone, isoproterenol, albuterol, and almeterol, deoxyspergualin, HSP 90 inhibitors, HSP 70 inhibitors, proteosome inhibitors such as epoxomicin, lactacystin, etc.
[0306] In another embodiment, the additional agent is a compound selected from 3-amino-6-(4-fluoro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid (3,3,3-trifluoro-2-hydroxy -2-methyl-propyl)-amide; 5-amino-6'-methyl-3-trifluoromethyl-[2,3]bipyridinyl-6-carboxylic acid (3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-cyclopropyl-N-(3,3,3-trifluoro-2-hydroxy-2-methylpropyl)-5-(tri- fluoromethyl)picolinamide; 3-amino-6-methoxy-N-(3,3,3-trifluoro-2-hydroxy-2-(trifluoromethyl)propyl)- -5-(trifluoro methyl)picolinamide; 3-amino-6-(4-fluoro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid ((S)-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-methoxy-5-trifluoromethyl-pyridine-2-carboxylic acid ((S-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-methoxy-5-trifluoromethyl-pyridine-2-carboxylic acid ((R)-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-(2,4-dichloro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid ((S)-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-(2,4-dichloro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid ((R)-3,3,3-trifluoro -2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-(4-fluoro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid (2-hydroxy-2-methyl-propyl)-amide; 3-amino-5,6-bis-trifluoromethyl-pyridine-2-carboxylic acid ((S)-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-5,6-bis-trifluoromethyl-pyridine-2-carboxylic acid ((R)-3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; (S)-3-amino-6-ethoxy-N-(3,3,3-trifluoro-2-hydroxy-2-methylpropyl)-5-(trif- luoro methyl)picolinamide; 3-amino-6-methoxy-5-trifluoromethyl-pyridine-2-carboxylic acid ((S)-3,3,3-trifluoro -2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-methoxy-5-trifluoromethyl-pyridine-2-carboxylic acid ((R)-3,3,3-trifluoro -2-hydroxy-2-methyl-propyl)-amide; 3-amino-6-(4-fluoro-phenyl)-5-trifluoromethyl-pyridine-2-carboxylic acid (3,3,3-trifluoro-2-hydroxy-2-methyl-propyl)-amide; 3-amino-5,6-bis-trifluoromethyl-pyridine-2-carboxylic acid ((S)-3,3,3-trifluoro -2-hydroxy-2-methyl-propyl)-amide; 3-amino-5,6-bis-trifluoromethyl-pyridine-2-carboxylic acid ((R)-3,3,3-trifluoro -2-hydroxy-2-methyl-propyl)-amide, or pharmaceutically acceptable salts thereof. In another embodiment, the additional agent is a compound disclosed in U.S. Pat. No. 8,247,436 and International PCT Publication WO 2011113894, each incorporated herein in their entirety by reference.
[0307] In one embodiment, the additional agent is trimethylangelicin. In another embodiment, the additional agent is a compound disclosed in WO 2012171954, incorporated herein in its entirety by reference.
[0308] In other embodiments, the additional agent is a compound disclosed in WO 2004028480, WO 2004110352, WO 2005094374, WO 2005120497, or WO 2006101740. In another embodiment, the additional agent is a benzo[c]quinolizinium derivative that exhibits CFTR modulation activity or a benzopyran derivative that exhibits CFTR modulation activity. In another embodiment, the additional agent is a compound disclosed in U.S. Pat. No. 7,202,262, U.S. Pat. No. 6,992,096, US20060148864, US20060148863, US20060035943, US20050164973, WO2006110483, WO2006044456, WO2006044682, WO2006044505, WO2006044503, WO2006044502, or WO2004091502. In another embodiment, the additional agent is a compound disclosed in WO2004080972, WO2004111014, WO2005035514, WO2005049018, WO2006099256, WO2006127588, or WO2007044560. In another embodiment, the additional agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carbo- xamide.
[0309] In one embodiment, 600 mg of Compound 1 may be administered to a subject in need thereof followed by co-administration of 250 mg of N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide (Compound 2). In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 600 mg of Compound 1 may be achieved by administering three tablets each containing 200 mg of Compound 1, four tablets each containing 150 mg of Compound 1, or one table of 400 mg Compound 1 and one tablet of 200 mg Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 600 mg of Compound 1 for 2 weeks followed by co-administration of 250 mg of Compound 2 for 1 additional week. In another embodiment, 600 mg of Compound 1 may be administered bid (twice daily) for 28 days followed by 250 mg of Compound 2 administered bid (twice daily) for 28 days. In another embodiment, 600 mg of Compound 1 may be administered qd (once a day) for 28 days followed by 250 mg of Compound 2 administered qd (once a day) for 28 days. In another embodiment, 600 mg of Compound 1 may be administered qd (once a day) for 28 days followed by co-administration of 600 mg of Compound 1 qd (once a day) and 250 mg of Compound 2 ql2h (once every 12 hours) for 28 days. In another embodiment, 600 mg of Compound 1 may be administered qd (once a day) and 250 mg of Compound 2 administered qd (once a day).
[0310] In one embodiment, 600 mg of Compound 1 may be administered to a subject in need thereof followed by co-administration of 450 mg of N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide (Compound 2). In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 600 mg of Compound 1 may be achieved by administering three tablets each containing 200 mg of Compound 1, or four tablets each containing 150 mg of Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 600 mg of Compound 1 for 2 weeks followed by co-administration of 450 mg of Compound 2 for 1 additional week. In another embodiment, 600 mg of Compound 1 may be administered bid (twice daily) for 28 days followed by 450 mg of Compound 2 administered bid (twice daily) for 28 days.
[0311] In one embodiment, 400 mg of Compound 1 may be administered to a subject in need thereof followed by co-administration of 350 mg of N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide (Compound 2). In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 400 mg of Compound 1 may be achieved by administering two tablets each containing 200 mg of Compound 1, or four tablets each containing 100 mg of Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 400 mg of Compound 1 for 2 weeks followed by co-administration of 350 mg of Compound 2 for 1 additional week. In another embodiment, 400 mg of Compound 1 may be administered q8h (every 8 hours) for 28 days followed by 350 mg of Compound 2 administered q8h (every 8 hours) for 28 days.
[0312] In one embodiment, 400 mg of Compound 1 may be administered to a subject in need thereof followed by co-administration of 250 mg of N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide (Compound 2). In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 400 mg of Compound 1 may be achieved by administering two tablets each containing 200 mg of Compound 1, or four tablets each containing 100 mg of Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 400 mg of Compound 1 for 2 weeks followed by co-administration of 150 mg or 250 mg of Compound 2 for 1 additional week. In another embodiment, 400 mg of Compound 1 may be administered bid (twice daily) for 28 days followed by 250 mg of Compound 2 administered bid (twice daily) for 28 days. In another embodiment, 400 mg of Compound 1 may be administered bid (twice daily) for 28 days followed by 250 mg of Compound 2 administered qd (once daily) for 28 days. In another embodiment, 400 mg of Compound 1 may be administered qd (once a day) for 28 days followed by co-administration of 400 mg of Compound 1 qd (once a day) and 250 mg of Compound 2 ql2h (once every 12 hours) for 28 days. In another embodiment, 400 mg of Compound 1 may be administered bid (twice daily) and 250 mg of Compound 2 administered qd (once daily).
[0313] In one embodiment, 400 mg of Compound 1 may be administered once a day to a subject in need thereof followed by co-administration of 150 mg of Compound 2 once a day. In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 400 mg of Compound 1 may be achieved by administering two tablets each containing 200 mg of Compound 1, or four tablets each containing 100 mg of Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 400 mg of Compound 1 for 2 weeks followed by co-administration of 150 mg or 250 mg of Compound 2 for 1 additional week.
[0314] In one embodiment, 400 mg of Compound 1 may be administered once a day to a subject in need thereof followed by co-administration of 150 mg of Compound 2 every 12 hours. In another embodiment, 400 mg of Compound 1 may be administered once a day to a subject in need thereof followed by co-administration of 250 mg of Compound 2 every 12 hours. In these embodiments, the dosage amounts may be achieved by administration of one or more tablets of the invention. For example, administration of 400 mg of Compound 1 may be achieved by administering two tablets each containing 200 mg of Compound 1, or four tablets each containing 100 mg of Compound 1. Compound 2 may be administered as a pharmaceutical composition comprising Compound 2 and a pharmaceutically acceptable carrier. The duration of administration may continue until amelioration of the disease is achieved or until a subject's physician advises, e.g. duration of administration may be less than a week, 1 week, 2 weeks, 3 weeks, or a month or longer. The co-administration period may be preceded by an administration period of just Compound 1 alone. For example, there could be administration of 400 mg of Compound 1 for 2 weeks followed by co-administration of 150 mg or 250 mg of Compound 2 for 1 additional week.
[0315] In another embodiment, 200 mg of Compound 1 may be administered qd (once a day) for 28 days followed by co-administration of 200 mg of Compound 1 qd (once a day) and 250 mg of Compound 2 q12h (once every 12 hours) for 28 days.
[0316] In one embodiment, the 100 mg, 200 mg, and 300 mg of Compound 1 tablets may be combined to form a number of different dosage amounts. For example, dosage amounts of 100 mg, 200 mg, 300 mg, 400 mg, 500 mg, 600 mg, 700 mg, 800 mg, 900 mg, 1000 mg, 1100 mg, or 1200 mg of Compound 1 may be administered by using the 100 mg, 200 mg, and 300 mg tablet formulations and multiples thereof. For example, a dosage amount of 900 mg of Compound 1 may be administered using 3 300 mg tablets of Compound 1. A dosage amount of 600 mg of Compound 1 may be administered using 3 200 mg tablets of Compound 1 or 2 300 mg tablets of Compound 1. Any of the preceding dosage amounts of this paragraph my be administered with the amounts of Compound 2 and/or dosage schedules of the preceding 3 paragraphs.
[0317] These combinations are useful for treating the diseases described herein including cystic fibrosis. These combinations are also useful in the kits described herein.
[0318] The amount of additional therapeutic agent present in the compositions of this invention will be no more than the amount that would normally be administered in a composition comprising that therapeutic agent as the only active agent. Preferably the amount of additional therapeutic agent in the presently disclosed compositions will range from about 50% to 100% of the amount normally present in a composition comprising that agent as the only therapeutically active agent.
[0319] In another aspect, the invention features a kit comprising a tablet of the present invention, and a separate therapeutic agent or pharmaceutical composition thereof. In another embodiment, the Compound 1 in the tablet is in Form I. In another embodiment, the therapeutic agent is a cystic fibrosis corrector other than Compound 1. In another embodiment, the therapeutic agent is a cystic fibrosis potentiator. In another embodiment, the therapeutic agent is N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide. In another embodiment, the tablet and the therapeutic agent are in separate containers. In another embodiment, the separate containers are bottles. In another embodiment, the separate containers are vials. In another embodiment, the separate containers are blister packs.
Therapeutic Uses of the Composition
[0320] In one aspect, the invention also provides a method of treating, lessening the severity of, or symptomatically treating a disease in a patient, the method comprising administering an effective amount of the pharmaceutical composition of the invention to the patient, wherein the disease is selected from cystic fibrosis, asthma, smoke induced COPD, chronic bronchitis, rhinosinusitis, constipation, pancreatitis, pancreatic insufficiency, male infertility caused by congenital bilateral absence of the vas deferens (CBAVD), mild pulmonary disease, idiopathic pancreatitis, allergic bronchopulmonary aspergillosis (ABPA), liver disease, hereditary emphysema, hereditary hemochromatosis, coagulation-fibrinolysis deficiencies, such as protein C deficiency, Type 1 hereditary angioedema, lipid processing deficiencies, such as familial hypercholesterolemia, Type 1 chylomicronemia, abetalipoproteinemia, lysosomal storage diseases, such as I-cell disease/pseudo-Hurler, mucopolysaccharidoses, Sandhof/Tay-Sachs, Crigler-Najjar type II, polyendocrinopathy/hyperinsulemia, Diabetes mellitus, Laron dwarfism, myleoperoxidase deficiency, primary hypoparathyroidism, melanoma, glycanosis CDG type 1, congenital hyperthyroidism, osteogenesis imperfecta, hereditary hypofibrinogenemia, ACT deficiency, Diabetes insipidus (DI), neurophyseal DI, neprogenic DI, Charcot-Marie Tooth syndrome, Perlizaeus-Merzbacher disease, neurodegenerative diseases such as Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, progressive supranuclear plasy, Pick's disease, several polyglutamine neurological disorders such as Huntington's, spinocerebullar ataxia type I, spinal and bulbar muscular atrophy, dentatorubal pallidoluysian, and myotonic dystrophy, as well as spongiform encephalopathies, such as hereditary Creutzfeldt-Jakob disease (due to prion protein processing defect), Fabry disease, Straussler-Scheinker syndrome, COPD, dry-eye disease, or Sjogren's disease, osteoporosis, osteopenia, bone healing and bone growth (including bone repair, bone regeneration, reducing bone resorption and increasing bone deposition), Gorham's Syndrome, chloride channelopathies such as myotonia congenita (Thomson and Becker forms), Bartter's syndrome type III, Dent's disease, hyperekplexia, epilepsy, lysosomal storage disease, Angelman syndrome, and Primary Ciliary Dyskinesia (PCD), a term for inherited disorders of the structure and/or function of cilia, including PCD with situs inversus (also known as Kartagener syndrome), PCD without situs inversus and ciliary aplasia.
[0321] Compound 1, as part of a combination with ivacaftor (N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1H-quinoline-3-carboxamide), has been granted a Breakthrough Therapy Designation from the Food and Drug Administration (FDA) for the treatment of cystic fibrosis, one of only two such grants at the time of the filing of this application (the other being for ivacaftor). This demonstrates a significant unmet need for the effective treatment of the cause of cystic fibrosis over symptomatic treatments. Additionally, a common challenge for drugs approved by the FDA is the occasional lack of drug availability for patients in need thereof. Accordingly, a significant unmet need exists for the presently disclosed Compound 1 formulations and processes for preparing them in a continuous and controlled manner.
[0322] In one aspect, the invention also provides a method of treating, lessening the severity of, or symptomatically treating a disease in a patient comprising administering an effective amount of the pharmaceutical composition of the invention to the patient, wherein the disease is selected from generalized epilepsy with ferbrile seizures plus (GEFS+), general epilepsy with ferbile and aferbrile seizures, myotonia, paramyotonia congenital, potassium-aggravated myotonia, hyperkalemic periodic paralysis, LQTS, LQTS/Brugada syndrome, autosomal-dominant LQTS with deafness, autosomal-recessive LQTS, LQTS with dysmorphic features, congenital and acquired LQTS, Timothy syndrome, persistent hyperinsulinemic hypolglycemia of infancy, dilated cardiomyopathy, autosomal-dominant LQTS, Dent disease, Osteopetrosis, Bartter syndrome type III, central core disease, malignant hyperthermia, and catecholaminergic polymorphic tachycardia.
[0323] In one aspect, the present invention is directed to a method of treating, lessening the severity of, or symptomatically treating cystic fibrosis in a patient comprising administering an effective amount of the pharmaceutical composition of the invention to the patient, wherein the patient possesses the CFTR genetic mutation N1303K, .DELTA.I507, or R560T.
[0324] In one aspect, the present invention is directed to a method of treating, lessening the severity of, or symptomatically treating cystic fibrosis in a patient comprising administering an effective amount of the pharmaceutical composition of the invention to the patient, wherein the patient possesses the CFTR genetic mutation G551D. In another embodiment, the patient is homozygous for G551D. In another embodiment, the patient is heterozygous for G551D wherein the other CFTR genetic mutation is any one of F508del, G542X, N1303K, W1282X, R117H, R553X, 1717-1G->A, 621+1G->T, 2789+5G->A, 3849+10kbC->T, R1162X, G85E, 3120+1G->A, .DELTA.I507, 1898+1G->A, 3659delC, R347P, R560T, R334W, A455E, 2184delA, or 711+1G->T.
[0325] In one aspect, the present invention is directed to a method of treating, lessening the severity of, or symptomatically treating cystic fibrosis in a patient comprising administering an effective amount of the pharmaceutical composition of the invention to the patient, wherein the patient possesses the CFTR genetic mutation F508del. In another embodiment, the patient is homozygous for F508del. In another embodiment, the patient is heterozygous for F508del wherein the other CFTR genetic mutation is any one of G551D, G542X, N1303K, W1282X, R117H, R553X, 1717-1G->A, 621+1G->T, 2789+5G->A, 3849+10kbC->T, R1162X, G85E, 3120+1G->A, .DELTA.I507, 1898+1G->A, 3659delC, R347P, R560T, R334W, A455E, 2184delA, or 711+1G->T.
[0326] In certain embodiments, the pharmaceutically acceptable compositions of the present invention comprising Compound 1 are useful for treating, lessening the severity of, or symptomatically treating cystic fibrosis in patients who exhibit residual CFTR activity in the apical membrane of respiratory and non-respiratory epithelia. The presence of residual CFTR activity at the epithelial surface can be readily detected using methods known in the art, e.g., standard electrophysiological, biochemical, or histochemical techniques. Such methods identify CFTR activity using in vivo or ex vivo electrophysiological techniques, measurement of sweat or salivary Cl.sup.- concentrations, or ex vivo biochemical or histochemical techniques to monitor cell surface density. Using such methods, residual CFTR activity can be readily detected in patients heterozygous or homozygous for a variety of different mutations, including patients homozygous or heterozygous for the most common mutation, F508del, as well as other mutations such as the G551D mutation, or the R117H mutation. In certain embodiments, the pharmaceutical compositions comprising Compound 1 are useful for treating, lessening the severity of, or symptomatically treating cystic fibrosis in patients who exhibit little to no residual CFTR activity. In certain embodiments, the pharmaceutical compositions comprising Compound 1 are useful for treating, lessening the severity of, or symptomatically treating cystic fibrosis in patients who exhibit little to no residual CFTR activity in the apical membrane of respiratory epithelia.
[0327] In another embodiment, the compounds and compositions of the present invention are useful for treating or lessening the severity of cystic fibrosis in patients who have residual CFTR activity induced or augmented. Such a residual CFTR inducer or augmenter can be done using pharmacological methods. In another embodiment, the compounds and compositions of the present invention are useful for treating or lessening the severity of cystic fibrosis in patients who have residual CFTR activity induced or augmented using or gene therapy. Such methods increase the amount of CFTR present at the cell surface, thereby inducing a hitherto absent CFTR activity in a patient or augmenting the existing level of residual CFTR activity in a patient.
[0328] In one embodiment, pharmaceutical compositions of the present invention comprising Compound 1, as described herein, are useful for treating or lessening the severity of cystic fibrosis in patients within certain genotypes exhibiting residual CFTR activity, e.g., Class I mutations (not synthesized), class II mutation (misfolding), class III mutations (impaired regulation or gating), class IV mutations (altered conductance), or class V mutations (reduced synthesis).
[0329] In one embodiment, pharmaceutical compositions of the present invention comprising Compound 1, as described herein, are useful for treating, lessening the severity of, or symptomatically treating cystic fibrosis in patients within certain clinical phenotypes, e.g., a moderate to mild clinical phenotype that typically correlates with the amount of residual CFTR activity in the apical membrane of epithelia. Such phenotypes include patients exhibiting pancreatic sufficiency.
[0330] In one embodiment, pharmaceutical compositions of the present invention comprising Compound 1, as described herein, are useful for treating, lessening the severity of, or symptomatically treating patients diagnosed with pancreatic sufficiency, idiopathic pancreatitis and congenital bilateral absence of the vas deferens, or mild lung disease wherein the patient exhibits residual CFTR activity.
[0331] In one embodiment, pharmaceutical compositions of the present invention comprising Compound 1, as described herein, are useful for treating, lessening the severity of, or symptomatically treating patients diagnosed with pancreatic sufficiency, idiopathic pancreatitis and congenital bilateral absence of the vas deferens, or mild lung disease wherein the patient has wild type CFTR.
[0332] In addition to cystic fibrosis, modulation of CFTR activity may be beneficial for other diseases not directly caused by mutations in CFTR, such as secretory diseases and other protein folding diseases mediated by CFTR. These include, but are not limited to, chronic obstructive pulmonary disease (COPD), dry eye disease, and Sjogren's Syndrome. COPD is characterized by airflow limitation that is progressive and not fully reversible. The airflow limitation is due to mucus hypersecretion, emphysema, and bronchiolitis. Activators of mutant or wild-type CFTR offer a potential treatment of mucus hypersecretion and impaired mucociliary clearance that is common in COPD. Specifically, increasing anion secretion across CFTR may facilitate fluid transport into the airway surface liquid to hydrate the mucus and optimized periciliary fluid viscosity. This would lead to enhanced mucociliary clearance and a reduction in the symptoms associated with COPD. Dry eye disease is characterized by a decrease in tear aqueous production and abnormal tear film lipid, protein and mucin profiles. There are many causes of dry eye, some of which include age, Lasik eye surgery, arthritis, medications, chemical/thermal burns, allergies, and diseases, such as cystic fibrosis and Sjogrens's syndrome. Increasing anion secretion via CFTR would enhance fluid transport from the corneal endothelial cells and secretory glands surrounding the eye to increase corneal hydration. This would help to alleviate the symptoms associated with dry eye disease. Sjogrens's syndrome is an autoimmune disease in which the immune system attacks moisture-producing glands throughout the body, including the eye, mouth, skin, respiratory tissue, liver, vagina, and gut. Symptoms, include, dry eye, mouth, and vagina, as well as lung disease. The disease is also associated with rheumatoid arthritis, systemic lupus, systemic sclerosis, and polymypositis/dermatomyositis. Defective protein trafficking is believed to cause the disease, for which treatment options are limited. Augmenters or inducers of CFTR activity may hydrate the various organs afflicted by the disease and help to elevate the associated symptoms.
[0333] In one embodiment, the invention relates to a method of augmenting or inducing anion channel activity in vitro or in vivo, comprising contacting the channel with a pharmaceutical composition of the present invention. In another embodiment, the anion channel is a chloride channel or a bicarbonate channel. In another embodiment, the anion channel is a chloride channel.
[0334] The exact amount required will vary from subject to subject, depending on the species, age, and general condition of the subject, the severity of the infection, the particular agent, its mode of administration, and the like. The compounds of the invention are preferably formulated in dosage unit form for ease of administration and uniformity of dosage. The expression "dosage unit form" as used herein refers to a physically discrete unit of agent appropriate for the patient to be treated. It will be understood, however, that the total daily usage of the compounds and compositions of the invention will be decided by the attending physician within the scope of sound medical judgment. The specific effective dose level for any particular patient or organism will depend upon a variety of factors including the disorder being treated and the severity of the disorder; the activity of the specific compound employed; the specific composition employed; the age, body weight, general health, sex and diet of the patient; the time of administration, route of administration, and rate of excretion of the specific compound employed; the duration of the treatment; drugs used in combination or coincidental with the specific compound employed, and like factors well known in the medical arts. The term "patient", as used herein, means an animal, preferably a mammal, and most preferably a human.
[0335] Anywhere in the present application where a name of a compound may not correctly describe the structure of the compound, the structure supersedes the name and governs.
EXAMPLES
[0336] XRPD (X-ray Powder Diffraction)
[0337] The X-Ray diffraction (XRD) data of Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A were collected on a Bruker D8 DISCOVER powder diffractometer with HI-STAR 2-dimensional detector and a flat graphite monochromator. Cu sealed tube with K.alpha. radiation was used at 40 kV, 35mA. The samples were placed on zero-background silicon wafers at 25.degree. C. For each sample, two data frames were collected at 120 seconds each at 2 different .theta..sub.2 angles: 8.degree. and 26.degree. . The data were integrated with GADDS software and merged with DIFFRACT.sup.plusEVA software. Uncertainties for the reported peak positions are .+-.0.2 degrees.
[0338] Jet Milling Description
[0339] Unmicronized Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A is sieved to de-lump it prior to placing it into the jet mill hopper. All sieves are disposable and received a wipe prior to use. Unmicronized Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A is added to the jet mill hopper at a controlled feeding rate using compressed nitrogen gas. The gas pressure range is 40-45/45-70 (Venturi/Mill) PSI and the feeding rate range is 0.5-1.6 Kg/Hour. The Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A is micronized in the mill through particle-particle and particle-wall collisions and the processed Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A is emptied into the micronized product containers. It is believed that one of ordinary skill in the art may also achieve Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A with a favorable particle size through pin milling based in part on the conditions described above.
[0340] Differential Scanning Calorimetry (DSC)
[0341] The Differential scanning calorimetry (DSC) data of Compound 1, Compound 1 Form I, Compound 1 Form II, or Compound 1 HCl Salt Form A were collected using a DSC Q100 V9.6 Build 290 (TA Instruments, New Castle, Del.). Temperature was calibrated with indium and heat capacity was calibrated with sapphire. Samples of 3-6 mg were weighed into aluminum pans that were crimped using lids with 1 pin hole. The samples were scanned from 25.degree. C. to 350.degree. C. at a heating rate of 1.0.degree. C./min and with a nitrogen gas purge of 50 ml/min. Data were collected by Thermal Advantage Q Series.TM. version 2.2.0.248 software and analyzed by Universal Analysis software version 4.1D (TA Instruments, New Castle, Del.). The reported numbers represent single analyses.
[0342] Compound 1 Form I, Compound 1 Form II, and Compound 1 HCl Salt Form A Single Crystal Structure Determination
[0343] Diffraction data were acquired on Bruker Apex II diffractometer equipped with sealed tube Cu K-alpha source and an Apex II CCD detector. The structure was solved and refined using SHELX program (Sheldrick, G. M., Acta Cryst., (2008) A64, 112-122). Based on systematic absences and intensities statistics the structure was solved and refined in P2.sub.1/n space group.
[0344] Vitride.RTM. (sodium bis(2-methoxyethoxy)aluminum hydride [or NaAlH.sub.2(OCH.sub.2CH.sub.2OCH.sub.3).sub.2], 65 wgt % solution in toluene) was purchased from Aldrich Chemicals.
[0345] 2,2-Difluoro-1,3-benzodioxole-5-carboxylic acid was purchased from Saltigo (an affiliate of the Lanxess Corporation).
Preparation of (2,2-difluoro-1,3-benzodioxol-5-yl)-methanol
##STR00020##
[0347] Commercially available 2,2-difluoro-1,3-benzodioxole-5-carboxylic acid (1.0 eq) was slurried in toluene (10 vol). Vitride.RTM. (2 eq) was added via addition funnel at a rate to maintain the temperature at 15-25.degree. C. At the end of the addition, the temperature was increased to 40.degree. C. for 2 hours (h), then 10% (w/w) aqueous (aq) NaOH (4.0 eq) was carefully added via addition funnel, maintaining the temperature at 40-50.degree. C. After stirring for an additional 30 minutes (min), the layers were allowed to separate at 40.degree. C. The organic phase was cooled to 20.degree. C., then washed with water (2.times.1.5 vol), dried (Na2SO4), filtered, and concentrated to afford crude (2,2-difluoro-1,3-benzodioxol-5-yl)-methanol that was used directly in the next step.
Preparation of 5-chloromethyl-2,2-difluoro-1,3-benzodioxole
##STR00021##
[0349] (2,2-difluoro-1,3-benzodioxol-5-yl)-methanol (1.0 eq) was dissolved in MTBE (5 vol). A catalytic amount of 4-(N,N-dimethyl)aminopyridine (DMAP) (1 mol %) was added and SOCl.sub.2 (1.2 eq) was added via addition funnel. The SOCl.sub.2 was added at a rate to maintain the temperature in the reactor at 15-25.degree. C. The temperature was increased to 30.degree. C. for 1 h, and then was cooled to 20.degree. C. Water (4 vol) was added via addition funnel while maintaining the temperature at less than 30.degree. C. After stirring for an additional 30 min, the layers were allowed to separate. The organic layer was stirred and 10% (w/v) aq NaOH (4.4 vol) was added. After stirring for 15 to 20 min, the layers were allowed to separate. The organic phase was then dried (Na.sub.2SO.sub.4), filtered, and concentrated to afford crude 5-chloromethyl-2,2-difluoro-1,3-benzodioxole that was used directly in the next step.
Preparation of (2,2-difluoro-1,3-benzodioxol-5-yl)-acetonitrile
##STR00022##
[0351] A solution of 5-chloromethyl-2,2-difluoro-1,3-benzodioxole (1 eq) in DMSO (1.25 vol) was added to a slurry of NaCN (1.4 eq) in DMSO (3 vol), while maintaining the temperature between 30-40.degree. C. The mixture was stirred for 1 h, and then water (6 vol) was added, followed by methyl tent-butyl ether (MTBE) (4 vol). After stirring for 30 min, the layers were separated. The aqueous layer was extracted with MTBE (1.8 vol). The combined organic layers were washed with water (1.8 vol), dried (Na.sub.2SO.sub.4), filtered, and concentrated to afford crude (2,2-difluoro-1,3-benzodioxol-5-yl)-acetonitrile (95%) that was used directly in the next step.
Synthesis of (2,2-difluoro-1,3-benzodioxol-5-yl)-1-ethylacetate-acetonitrile
##STR00023##
[0353] A reactor was purged with nitrogen and charged with 900 mL of toluene. The solvent was degassed via nitrogen sparge for no less than 16 h. To the reactor was then charged Na.sub.3PO.sub.4 (155.7 g, 949.5 mmol), followed by bis(dibenzylideneacetone) palladium(0) (7.28 g, 12.66 mmol). A 10% w/w solution of tert-butylphosphine in hexanes (51.23 g, 25.32 mmol) was charged over 10 min at 23.degree. C. from a nitrogen purged addition funnel. The mixture was allowed to stir for 50 min, at which time 5-bromo-2,2-difluoro-1,3-benzodioxole (75 g, 316.5 mmol) was added over 1 min. After stirring for an additional 50 min, the mixture was charged with ethyl cyanoacetate (71.6 g, 633.0 mmol) over 5 min followed by water (4.5 mL) in one portion. The mixture was heated to 70.degree. C. over 40 min and analyzed by HPLC every 1-2 h for the percent conversion of the reactant to the product. After complete conversion was observed (typically 100% conversion after 5-8 h), the mixture was cooled to 20-25.degree. C. and filtered through a celite pad. The celite pad was rinsed with toluene (2.times.450 mL) and the combined organics were concentrated to 300 mL under vacuum at 60-65.degree. C. The concentrate was charged with 225 mL DMSO and concentrated under vacuum at 70-80.degree. C. until active distillation of the solvent ceased. The solution was cooled to 20-25.degree. C. and diluted to 900 mL with DMSO in preparation for Step 2. .sup.1H NMR (500 MHz, CDCl.sub.3) .delta. 7.16-7.10 (m, 2H), 7.03 (d, J=8.2 Hz, 1H), 4.63 (s, 1H), 4.19 (m, 2H), 1.23 (t, J=7.1 Hz, 3H).
Synthesis of (2,2-difluoro-1,3-benzodioxol-5-yl)-acetonitrile
##STR00024##
[0355] The DMSO solution of (2,2-difluoro-1,3-benzodioxol-5-yl)-1-ethylacetate-acetonitrile from above was charged with 3 N HCl (617.3 mL, 1.85 mol) over 20 min while maintaining an internal temperature <40.degree. C. The mixture was then heated to 75.degree. C. over 1 h and analyzed by HPLC every 1-2 h for % conversion. When a conversion of >99% was observed (typically after 5-6 h), the reaction was cooled to 20-25.degree. C. and extracted with MTBE (2.times.525 mL), with sufficient time to allow for complete phase separation during the extractions. The combined organic extracts were washed with 5% NaCl (2.times.375 mL). The solution was then transferred to equipment appropriate for a 1.5-2.5 Torr vacuum distillation that was equipped with a cooled receiver flask. The solution was concentrated under vacuum at <60.degree. C. to remove the solvents. (2,2-Difluoro-1,3-benzodioxol-5-yl)-acetonitrile was then distilled from the resulting oil at 125-130.degree. C. (oven temperature) and 1.5-2.0 Torr. (2,2-Difluoro-1,3-benzodioxol-5-yl)-acetonitrile was isolated as a clear oil in 66% yield from 5-bromo-2,2-difluoro-1,3-benzodioxole (2 steps) and with an HPLC purity of 91.5% AUC (corresponds to a w/w assay of 95%). .sup.1H NMR (500 MHz, DMSO) .delta. 7.44 (br s, 1H), 7.43 (d, J=8.4 Hz, 1H), 7.22 (dd, J=8.2, 1.8 Hz, 1H), 4.07 (s, 2H).
Preparation of (2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarbonitrile
##STR00025##
[0357] A mixture of (2,2-difluoro-1,3-benzodioxol-5-yl)-acetonitrile (1.0 eq), 50 wt % aqueous KOH (5.0 eq) 1-bromo-2-chloroethane (1.5 eq), and Oct4NBr (0.02 eq) was heated at 70.degree. C. for 1 h. The reaction mixture was cooled, then worked up with MTBE and water. The organic phase was washed with water and brine. The solvent was removed to afford (2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarbonitrile.
Preparation of 1-(2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarboxylic acid
##STR00026##
[0359] (2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarbonitrile was hydrolyzed using 6 M NaOH (8 equiv) in ethanol (5 vol) at 80.degree. C. overnight. The mixture was cooled to room temperature and the ethanol was evaporated under vacuum. The residue was taken up in water and MTBE, 1 M HCl was added, and the layers were separated. The MTBE layer was then treated with dicyclohexylamine (DCHA) (0.97 equiv). The slurry was cooled to 0.degree. C., filtered and washed with heptane to give the corresponding DCHA salt. The salt was taken into MTBE and 10% citric acid and stirred until all the solids had dissolved. The layers were separated and the MTBE layer was washed with water and brine. A solvent swap to heptane followed by filtration gave 1-(2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarboxylic acid after drying in a vacuum oven at 50.degree. C. overnight.
Preparation of 1-(2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarbonyl chloride
##STR00027##
[0361] 1-(2,2-difluoro-1,3-benzodioxol-5-yl)-cyclopropanecarboxylic acid (1.2 eq) is slurried in toluene (2.5 vol) and the mixture was heated to 60.degree. C. SOCl.sub.2 (1.4 eq) was added via addition funnel. The toluene and SOCl.sub.2 were distilled from the reaction mixture after 30 minutes. Additional toluene (2.5 vol) was added and the resulting mixture was distilled again, leaving the product acid chloride as an oil, which was used without further purification.
Preparation of tert-butyl-3-(3-methylpyridin-2-yl)benzoate
##STR00028##
[0363] 2-Bromo-3-methylpyridine (1.0 eq) was dissolved in toluene (12 vol). K.sub.2CO.sub.3 (4.8 eq) was added, followed by water (3.5 vol). The resulting mixture was heated to 65.degree. C. under a stream of N.sub.2 for 1 hour. 3-(t-Butoxycarbonyl)phenylboronic acid (1.05 eq) and Pd(dppf)Cl.sub.2.CH.sub.2Cl.sub.2 (0.015 eq) were then added and the mixture was heated to 80.degree. C. After 2 hours, the heat was turned off, water was added (3.5 vol), and the layers were allowed to separate. The organic phase was then washed with water (3.5 vol) and extracted with 10% aqueous methanesulfonic acid (2 eq MsOH, 7.7 vol). The aqueous phase was made basic with 50% aqueous NaOH (2 eq) and extracted with EtOAc (8 vol). The organic layer was concentrated to afford crude tent-butyl-3-(3-methylpyridin-2-yl)benzoate (82%) that was used directly in the next step.
Preparation of 2-(3-(tert-butoxycarbonyl)phenyl)-3-methylpyridine-1-oxide
##STR00029##
[0365] tent-Butyl-3-(3-methylpyridin-2-yl)benzoate (1.0 eq) was dissolved in EtOAc (6 vol). Water (0. 3 vol) was added, followed by urea-hydrogen peroxide (3 eq). Phthalic anhydride (3 eq) was then added portionwise to the mixture as a solid at a rate to maintain the temperature in the reactor below 45.degree. C. After completion of the phthalic anhydride addition, the mixture was heated to 45.degree. C. After stirring for an additional 4 hours, the heat was turned off 10% w/w aqueous Na.sub.2SO.sub.3 (1.5 eq) was added via addition funnel. After completion of Na.sub.2SO.sub.3 addition, the mixture was stirred for an additional 30 min and the layers separated. The organic layer was stirred and 10% wt/wt aqueous. Na.sub.2CO.sub.3 (2 eq) was added. After stirring for 30 minutes, the layers were allowed to separate. The organic phase was washed 13% w/v aq NaCl. The organic phase was then filtered and concentrated to afford crude 2-(3-(tert-butoxycarbonyl)phenyl)-3-methylpyridine-1-oxide (95%) that was used directly in the next step.
Preparation of tert-butyl-3-(6-amino-3-methylpyridin-2-yl)benzoate
##STR00030##
[0367] A solution of 2-(3-(tent-butoxycarbonyl)phenyl)-3-methylpyridine-1-oxide (1 eq) and pyridine (4 eq) in acetonitrile (8 vol) was heated to 70.degree. C. A solution of methanesulfonic anhydride (1.5 eq) in MeCN (2 vol) was added over 50 min via addition funnel while maintaining the temperature at less than 75.degree. C. The mixture was stirred for an additional 0.5 hours after complete addition. The mixture was then allowed to cool to ambient. Ethanolamine (10 eq) was added via addition funnel. After stirring for 2 hours, water (6 vol) was added and the mixture was cooled to 10.degree. C. After stirring for 3 hours, the solid was collected by filtration and washed with water (3 vol), 2:1 acetonitrile/water (3 vol), and acetonitrile (2.times.1.5 vol). The solid was dried to constant weight (<1% difference) in a vacuum oven at 50.degree. C. with a slight N.sub.2 bleed to afford tent-butyl-3-(6-amino-3-methylpyridin-2-yl)benzoate as a red-yellow solid (53% yield).
Preparation of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)-cyclopropanecarboxamido)-3- -methylpyridin-2-yl)-t-butylbenzoate
##STR00031##
[0369] The crude acid chloride described above was dissolved in toluene (2.5 vol based on acid chloride) and added via addition funnel to a mixture of tert-butyl-3-(6-amino-3-methylpyridin-2-yl)benzoate (1 eq), DMAP, (0.02 eq), and triethylamine (3.0 eq) in toluene (4 vol based on tent-butyl-3-(6-amino-3-methylpyridin-2-yl)benzoate). After 2 hours, water (4 vol based on tent-butyl-3-(6-amino-3-methylpyridin-2-yl)benzoate) was added to the reaction mixture. After stirring for 30 minutes, the layers were separated. The organic phase was then filtered and concentrated to afford a thick oil of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (quantitative crude yield). Acetonitrile (3 vol based on crude product) was added and distilled until crystallization occurs. Water (2 vol based on crude product) was added and the mixture stirred for 2 h. The solid was collected by filtration, washed with 1:1 (by volume) acetonitrile/water (2.times.1 volumes based on crude product), and partially dried on the filter under vacuum. The solid was dried to a constant weight (<1% difference) in a vacuum oven at 60.degree. C. with a slight N.sub.2 bleed to afford 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate as a brown solid.
Preparation of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid.HCL Salt
##STR00032##
[0371] To a slurry of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in MeCN (3.0 vol) was added water (0.83 vol) followed by concentrated aqueous HCl (0.83 vol). The mixture was heated to 45.+-.5.degree. C. After stirring for 24 to 48 h, the reaction was complete, and the mixture was allowed to cool to ambient. Water (1.33 vol) was added and the mixture stirred. The solid was collected by filtration, washed with water (2.times.0.3 vol), and partially dried on the filter under vacuum. The solid was dried to a constant weight (<1% difference) in a vacuum oven at 60.degree. C. with a slight N.sub.2 bleed to afford 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid.HCl as an off-white solid.
[0372] An .sup.1HNMR spectrum of Compound 1 is shown in FIG. 20 and FIG. 21 depicts an .sup.1HNMR spectrum of Compound 1 as an HCl salt.
[0373] Table 2 below recites the .sup.1HNMR data for Compound I.
TABLE-US-00025 TABLE 2 Compound LC/MS LC/RT No M + 1 minutes NMR 1 453.3 1.93 .sup.1HNMR (400 MHz, DMSO-d6) 9.14 (s, 1H), 7.99-7.93 (m, 3H), 7.80-7.78 (m,1H), 7.74-7.72 (m,1H), 7.60-7.55 (m,2H), 7.41-7.33 (m,2H), 2.24 (s, 3H), 1.53- 1.51 (m, 2H), 1.19-1.17 (m, 2H).
Preparation of Compound 1 Form I, Method A
##STR00033##
[0375] A slurry of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid.HCl (1 eq) in water (10 vol) was stirred at ambient temperature. A sample was taken after stirring for 24 h. The sample was filtered and the solid was washed with water (2 times). The solid sample was submitted for DSC analysis. When DSC analysis indicated complete conversion to Form I, the solid was collected by filtration, washed with water (2.times.1.0 vol), and partially dried on a filter under vacuum. The solid was then dried to a constant weight (<1% difference) in a vacuum oven at 60.degree. C. with a slight N.sub.2 bleed to afford Compound 1 Form I as an off-white solid (98% yield). .sup.1H NMR (400 MHz, DMSO-d6) 9.14 (s, 1H), 7.99-7.93 (m, 3H), 7.80-7.78 (m, 1H), 7.74-7.72 (m, 1H), 7.60-7.55 (m, 2H), 7.41-7.33 (m, 2H), 2.24 (s, 3H), 1.53-1.51 (m, 2H), 1.19-1.17 (m, 2H).
Preparation of Compound 1 Form I, Method B
##STR00034##
[0377] A solution of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in formic acid (3.0 vol) was heated with stirring to 70.+-.10.degree. C. , for 8 h. The reaction was deemed complete when no more than 1.0% AUC by chromatographic methods of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate) remained. The mixture was allowed to cool to ambient. The solution was added to water (6 vol), heated at 50.degree. C., and the mixture was stirred. The mixture was then heated to 70.+-.10.degree. C. until the level of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate was no more than 0.8% (AUC). The solid was collected by filtration, washed with water (2.times.3 vol), and partially dried on the filter under vacuum. The solid was dried to a constant weight (<1% difference) in a vacuum oven at 60.degree. C. with a slight N.sub.2 bleed to afford Compound 1 Form I as an off-white solid.
[0378] The DSC trace of Compound 1 Form I is shown in FIG. 22. Melting for Compound 1 Form I occurs at about 204.degree. C.
[0379] An X-ray diffraction pattern was calculated from a single crystal structure of Compound 1 Form I and is shown in FIG. 1. Table 3 lists the calculated peaks for FIG. 1.
TABLE-US-00026 TABLE 3 2.theta. Angle Relative Peak Rank [degrees] Intensity [%] 11 14.41 48.2 8 14.64 58.8 1 15.23 100.0 2 16.11 94.7 3 17.67 81.9 7 19.32 61.3 4 21.67 76.5 5 23.40 68.7 9 23.99 50.8 6 26.10 67.4 10 28.54 50.1
[0380] An actual X-ray powder diffraction pattern of Compound 1 Form I is shown in FIG. 2. Table 4 lists the actual peaks for FIG. 2.
TABLE-US-00027 TABLE 4 2.theta. Angle Relative Peak Rank [degrees] Intensity [%] 7 7.83 37.7 3 14.51 74.9 4 14.78 73.5 1 15.39 100.0 2 16.26 75.6 6 16.62 42.6 5 17.81 70.9 9 21.59 36.6 10 23.32 34.8 11 24.93 26.4 8 25.99 36.9
[0381] Colorless crystals of Compound 1 Form I were obtained by cooling a concentrated 1-butanol solution from 75.degree. C. to 10.degree. C. at a rate of 0.2.degree. C./min. A crystal with dimensions of 0.50.times.0.08.times.0.03 mm was selected, cleaned with mineral oil, mounted on a MicroMount and centered on a Bruker APEXII system. Three batches of 40 frames separated in reciprocal space were obtained to provide an orientation matrix and initial cell parameters. Final cell parameters were obtained and refined based on the full data set.
[0382] A diffraction data set of reciprocal space was obtained to a resolution of 0.82 .ANG. using 0.5.degree. steps using 30 s exposure for each frame. Data were collected at 100 (2) K. Integration of intensities and refinement of cell parameters were accomplished using APEXII software. Observation of the crystal after data collection showed no signs of decomposition.
[0383] A conformational picture of Compound 1 Form I based on single crystal X-ray analysis is shown in FIG. 23. Compound 1 Form I is monoclinic, P.sub.21/n, with the following unit cell dimensions: a=4.9626(7) .ANG., b=12.299(2) .ANG., c=33.075 (4) .ANG., .beta.3=93.938(9).degree., V=2014.0 .ANG..sup.3, Z=4. Density of Compound 1 Form I calculated from structural data is 1.492 g/cm.sup.3 at 100 K.
[0384] Preparation of Compound 1 Form II from Compound 1 Form I.
[0385] Compound 1 Form I (approximately 30 mg) was slurried in 500 .mu.L of an appropriate solvent (for example, methanol, ethanol, acetone, 2-propanol, acetonitrile, tetrahydrofuran, methyl acetate, 2-butanone, ethyl formate, and -methyl tetrahydrofuran for two days. The slurry was then filitered centrifugally or under vacuum and was left to dry at ambient temperature overnight to yield Compound 1 Form II.
[0386] The DSC trace of Compound 1 Form II Acetone Solvate is shown in FIG. 15, showing two phase transitions. The melting point for Compound 1 Form II Acetone Solvate occurs at about 188.degree. C. and 205.degree. C.
[0387] An actual X-ray powder diffraction pattern of Compound 1 Form II is shown in FIG. 3. Table 5 lists the actual peaks for FIG. 3 in descending order of relative intensity.
TABLE-US-00028 TABLE 5 2.theta. Angle Relative Intensity [degrees] [%] 21.70 100.0 8.98 65.5 11.04 57.4 18.16 55.9 23.06 55.4 20.63 53.1 22.22 50.2 18.57 49.1 16.66 47.2 19.86 35.0
[0388] Conformational depictions of Compound 1 Form II Acetone Solvate based on single crystal X-ray analysis are shown in FIG. 24. The stoichiometry between Compound 1 Form II and acetone is approximately 4.4:1 (4.48:1 calculated from .sup.1H NMR; 4.38:1 from X-ray). The crystal structure reveals a packing of the molecules where there are two voids or pockets per unit cell, or 1 void per host molecule. In the acetone solvate, approximately 92 percent of voids are occupied by acetone molecules. Compound 1 Form II is a monoclinic P2.sub.1/n space group with the following unit cell dimensions: a=16.5235(10) .ANG., b=12.7425(8) .ANG., c=20.5512 (13) .ANG., .alpha.=90.degree., .beta.=103.736(4).degree., .gamma.=90.degree., V=4203.3(5) .ANG..sup.3, =4. The density of Compound 1 in Compound 1 Form II calculated from structural data is 1.430/cm.sup.3 at 100 K.
[0389] A solid state .sup.13C NMR spectrum of Compound 1 Form II Acetone Solvate is shown in FIG. 25. Table 6 provides chemical shifts of the relevant peaks.
TABLE-US-00029 TABLE 6 Compound 1 Form II, Acetone Solvate Peak .sup.13C Chem. Shifts # F1 [ppm] Intensity 1 202.8 6.05 2 173.3 62.66 3 171.9 20.53 4 153.5 28.41 5 150.9 21.68 6 150.1 19.49 7 143.2 45.74 8 142.3 42.68 9 140.1 37.16 10 136.6 26.82 11 135.9 30.1 12 134.6 39.39 13 133.2 23.18 14 131.0 60.92 15 128.5 84.58 16 116.0 34.64 17 114.2 23.85 18 112.4 25.3 19 110.9 24.12 20 107.8 18.21 21 32.0 54.41 22 22.2 20.78 23 18.8 100
[0390] A solid state .sup.19F NMR spectrum of Compound 1 Form II Acetone Solvate is shown in FIG. 26. Peaks with an asterisk denote spinning side bands. Table 7 provides chemical shifts of the relevant peaks.
TABLE-US-00030 TABLE 7 Compound 1 Form II, Acetone Solvate Peak .sup.19F Chem. Shifts # F1 [ppm] Intensity 1 -41.6 12.5 2 -46.4 6.77 3 -51.4 9.05
[0391] Preparation of Compound 1 HCl Salt Form A.
[0392] Colorless crystals of Compound 1 HCl Salt Form A were obtained by slow evaporation from a concentrated solution of the HCl salt of Compound 1 in ethanol. A crystal with dimensions of 0.30.times.1/5.times.0.15 mm was selected, cleaned using mineral oil, mounted on a MicroMount and centered on a Bruker APEXII diffractometer. Three batches of 40 frames separated in reciprocal space were obtained to provide an orientation matrix and initial cell parameters. Final cell parameters were obtained and refined based on the full data set.
[0393] FIG. 18 provides a conformational image of Compound 1 HCl Salt Form A as a dimer, based on single crystal analysis. An X-ray diffraction pattern of Compound 1 HCl Salt Form A calculated from the crystal structure is shown in FIG. 27. Table 8 contains the calculated peaks for FIG. 27 in descending order of relative intensity.
TABLE-US-00031 TABLE 8 2.theta. Relative Intensity [degrees] [%] 8.96 100.00 17.51 48.20 18.45 34.60 10.33 32.10 16.01 18.90 11.94 18.40 8.14 16.20 10.10 13.90 16.55 13.30 9.54 10.10 16.55 13.30
[0394] Exemplary Oral Pharmaceutical Formulations Comprising Compound 1
[0395] A tablet was prepared with the components and amounts listed in Table 9 for Exemplary Tablet 1A comprising 100 mg of API, i.e. Compound 1 Form I. Exemplary Tablet 1A (formulated to have 100 mg of Compound 1) is prepared using a dry roller compaction device formulation process. In Table 9, grades/brands were microcrystalline cellulose: Avicel PH102; mannitol: Pearlitol SD 100; croscarmellose sodium: Acdisol; and colloidal silica: Cabosil.
TABLE-US-00032 TABLE 9 Roller Compaction Granule Blend (% w/w) Compound 1 Form I 30 Microcrystalline cellulose 42.3 Mannitol 21.2 Croscarmellose Sodium 3 Sodium Lauryl Sulfate 1 Colloidal Silica 0.5 Magnesium Stearate 2 Tablet Composition (100 mg dose, 335 mg image) (% w/w) Roller Compaction Granule Blend 99.5 Magnesium Stearate 0.5
[0396] A tablet was prepared with the components and amounts listed in Table 10 for Exemplary Tablet 1B comprising 100mg of API, i.e. Compound 1 Form I. Exemplary Tablet 1B (formulated to have 100 mg of Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 10, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Tablet Composition--croscarmellose sodium: Acdisol.
TABLE-US-00033 TABLE 10 High Shear Granule Blend (% w/w) Compound 1 Form I 50 Microcrystalline cellulose 30 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (100 mg dose, 205 mg image) (% w/w) High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0397] A tablet was prepared with the components and amounts listed in Table 11 for Exemplary Tablet 1C comprising 100 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1C (formulated to have 100 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 11, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Tablet Composition--croscarmellose sodium: Acdisol.
TABLE-US-00034 TABLE 11 High Shear Granule Blend (% w/w) Compound 1 Form I 60 Microcrystalline cellulose 20 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (100 mg dose, 171 mg image) (% w/w) High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0398] A tablet was prepared with the components and amounts listed in Table 12 for Exemplary Tablet 1D comprising 200mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1D (formulated to have 200 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 12, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712.
TABLE-US-00035 TABLE 12 High Shear Granule Blend (% w/w) Compound 1 Form I 60 Microcrystalline cellulose 20 Mannitol 13 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (200 mg dose, 402 mg image) (% w/w) High Shear Granule Blend 83 Microcrystalline cellulose 14 Croscarmellose Sodium 2 Magnesium Stearate 1
[0399] A tablet was prepared with the components and amounts listed in Table 13 for Exemplary Tablet 1E comprising 200 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1E (formulated to have 200 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 13, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00036 TABLE 13 High Shear Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 66 Mannitol 43 Croscarmellose Sodium 7 Polyvinylpyrrolidone 13 Sodium Lauryl Sulfate 3 Core Tablet Composition (200 mg dose, 400 mg image) mg High Shear Granule Blend 332 Microcrystalline cellulose 56 Croscarmellose Sodium 8 Magnesium Stearate 4 Film Coated Tablet (200 mg dose, 412 mg image) mg Core Tablet Composition 400 Film Coat 12 Wax trace
[0400] A tablet was prepared with the components and amounts listed in Table 14 for Exemplary Tablet 1F comprising 200 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1F (formulated to have 200 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 14, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00037 TABLE 14 High Shear Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 67 Mannitol 45 Croscarmellose Sodium 7 Polyvinylpyrrolidone 10.4 Sodium Lauryl Sulfate 2.6 Core Tablet Composition (200 mg dose, 400 mg image) mg High Shear Granule Blend 332 Microcrystalline cellulose 56 Croscarmellose Sodium 8 Magnesium Stearate 4 Film Coated Tablet (200 mg dose, 412 mg image) mg Core Tablet Composition 400 Film Coat 12 Wax 0.04
[0401] A tablet was prepared with the components and amounts listed in Table 15 for Exemplary Tablet 1G comprising 100 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1G (formulated to have 100 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 15, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Tablet Composition--croscarmellose sodium: Acdisol.
TABLE-US-00038 TABLE 15 High Shear Granule Blend (% w/w) Compound 1 Form I 70 Microcrystalline cellulose 12 Mannitol 11 Croscarmellose Sodium 2 Polyvinylpyrrolidone 4 Sodium Lauryl Sulfate 1 Tablet Composition (100 mg dose, 147 mg image) (% w/w) High Shear Granule Blend 97.5 Croscarmellose Sodium 2.0 Magnesium Stearate 0.5
[0402] A tablet was prepared with the components and amounts listed in Table 16 for Exemplary Tablet 1H comprising 100 mg of API, i.e. crystalline Compound 1 Form I or Form II. Exemplary Tablet 1H (formulated to have 100 mg of crystalline Compound 1 Form I or Form II) is prepared using a wet high shear granule formulation process. In Table 16, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712.
TABLE-US-00039 TABLE 16 High Shear Granule Blend (% w/w) Compound 1 Form I or Form II 61 Microcrystalline cellulose 20.3 Mannitol 13.2 Croscarmellose Sodium 2 Polyvinylpyrrolidone 2.7 Sodium Lauryl Sulfate 0.7 Tablet Composition (100 mg dose, 197 mg image) (% w/w) High Shear Granule Blend 83 Microcrystalline cellulose 14 Croscarmellose Sodium 2 Magnesium Stearate 1
[0403] A tablet was prepared with the components and amounts listed in Table 17 for Exemplary Tablet 1I comprising 100 mg of API, i.e. crystalline Compound 1 Form I or Form II. Exemplary Tablet 1I (formulated to have 100 mg of crystalline Compound 1 Form I or Form II) is prepared using a wet high shear granule formulation process. In Table 17, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712.
TABLE-US-00040 TABLE 17 High Shear Granule Blend mg Compound 1 Form I or Form II 100 Microcrystalline cellulose 33.3 Mannitol 21.7 Croscarmellose Sodium 3.3 Polyvinylpyrrolidone 4.4 Sodium Lauryl Sulfate 1.1 Core Tablet Composition (100 mg dose, 197 mg image) mg High Shear Granule Blend 163.9 Microcrystalline cellulose 27.6 Croscarmellose Sodium 3.9 Magnesium Stearate 2.0
[0404] A tablet was prepared with the components and amounts listed in Table 18 for Exemplary Tablet 1J comprising 300 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1J (formulated to have 300 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 18, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00041 TABLE 18 High Shear Granule Blend mg Compound 1 Form I 300 Microcrystalline cellulose 99 Mannitol 64.5 Croscarmellose Sodium 10.5 Polyvinylpyrrolidone 19.5 Sodium Lauryl Sulfate 4.5 Core Tablet Composition (300 mg dose, 600 mg image) mg High Shear Granule Blend 498 Microcrystalline cellulose 84 Croscarmellose Sodium 12 Magnesium Stearate 6 Film Coated Tablet (300 mg dose, 618 mg image) mg Core Tablet Composition 600 Film Coat 18 Wax 0.06
[0405] A tablet was prepared with the components and amounts listed in Table 19 for Exemplary Tablet 1K comprising 300 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1K (formulated to have 300 mg of crystalline Compound 1 Form I) is prepared using a wet high shear granule formulation process. In Table 19, grades/brands were as follows. High Shear Granule Blend--microcrystalline cellulose: Avicel PH101; mannitol: Pearlitol C50; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00042 TABLE 19 High Shear Granule Blend mg Compound 1 Form I 300 Microcrystalline cellulose 100.5 Mannitol 67.5 Croscarmellose Sodium 10.5 Polyvinylpyrrolidone 15.6 Sodium Lauryl Sulfate 3.9 Core Tablet Composition (300 mg dose, 600 mg image) mg High Shear Granule Blend 498 Microcrystalline cellulose 84 Croscarmellose Sodium 12 Magnesium Stearate 6 Film Coated Tablet (300 mg dose, 618 mg image) mg Core Tablet Composition 600 Film Coat 18 Wax 0.06
[0406] A tablet was prepared with the components and amounts listed in Table 20 for Exemplary Tablet 1L comprising 200 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1L (formulated to have 200 mg of crystalline Compound 1 Form I) is prepared using a twin screw wet granulation formulation process. In Table 20, grades/brands were as follows. Twin Screw Granule Blend--microcrystalline cellulose: Avicel PH101; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00043 TABLE 20 Twin Screw Granule Blend mg Compound 1 Form I 200 Microcrystalline cellulose 34.0 Croscarmellose Sodium 6.3 Polyvinylpyrrolidone 7.8 Sodium Lauryl Sulfate 1.8 Core Tablet Composition (200 mg dose) mg Twin Screw Granule Blend 249.9 Microcrystalline cellulose 36.1 Croscarmellose Sodium 12.0 Magnesium Stearate 3.0 Film Coated Tablet (200 mg dose, 310 mg total) mg Core Tablet Composition 301 Film Coat 9.0 Wax trace
[0407] A tablet was prepared with the components and amounts listed in Table 21 for Exemplary Tablet 1M comprising 400 mg of API, i.e. crystalline Compound 1 Form I. Exemplary Tablet 1M (formulated to have 400 mg of crystalline Compound 1 Form I) is prepared using a twin screw wet granule formulation process. In Table 21, grades/brands were as follows. Twin Screw Granule Blend--microcrystalline cellulose: Avicel PH101; croscarmellose sodium: Acdisol; polyvinylpyrrolidone: Kollidon PVP K30; and in the Core Tablet Composition--microcrystalline cellulose: Avicel PH200; croscarmellose sodium: Acdisol; and magnesium stearate: 5712; and in the film coat--film coat: Opadry II; wax: Carnauba.
TABLE-US-00044 TABLE 21 Twin Screw Granule Blend mg Compound 1 Form I 400 Microcrystalline cellulose 68.0 Croscarmellose Sodium 12.6 Polyvinylpyrrolidone 15.6 Sodium Lauryl Sulfate 3.6 Core Tablet Composition (400 mg dose) mg Twin Screw Granule Blend 499.8 Microcrystalline cellulose 72.2 Croscarmellose Sodium 24.0 Magnesium Stearate 6.0 Film Coated Tablet (400 mg dose, 620 mg total) mg Core Tablet Composition 602 Film Coat 18.0 Wax trace
[0408] Tablet Formation from Roller Compaction Granule Composition
[0409] Equipment/Process
[0410] Equipment
[0411] Roller Compactors: Alexanderwerk WP 120, Vector TF-Mini, or Vector TF-Labo.
[0412] Screening/Weighing
[0413] Compound 1 and excipients may be screened prior to or after weigh-out. Appropriate screen sizes are mesh 20, mesh 40, or mesh 60. Compound 1 may be pre-blended with one or more of the excipients to simplify screening.
[0414] Blending
[0415] Compound 1 and excipients may be added to the blender in different order. The blending may be performed in a Turbula blender or a v-shell blender. The components may be blended for 10 minutes without lubricant followed by additional blending with lubricant for 3 minutes.
[0416] Roller Compaction
[0417] The blend may be roller compacted in ribbons and milled into granules using an Alexanderwerk WP 120. The rolls used may be the 25 mm rolls using a compaction pressure of 18 to 50 bar, a roller speed of 3 to 12 RPM, and a screw feeder speed of 20 to 80 RPM. The screen sizes of the integrated mill may be 2 mm for the top screen and 0.8 mm for the bottom screen.
[0418] Blending
[0419] The roller compacted granules may be blended with extra-granular excipients such as fillers and lubricant using a V-shell blender. The blending time may be 5, 3 or 1 minute(s).
[0420] Compression
[0421] The compression blend has been compressed into tablets using a single station Riva MiniPress with 10 mm tooling. The weight of the tablets for a 100 mg dose may be about 200, 250, or 300 mg.
[0422] Film Coating
[0423] Tablets may be film coated using a pan coater, such as, for example an O'Hara Labcoat.
[0424] Printing
[0425] Film coated tablets may be printed with a monogram on one or both tablet faces with, for example, a Hartnett Delta printer.
[0426] Tablet Formation From High Shear Granule Composition
[0427] Equipment/Process
[0428] Equipment
[0429] Granulator: Procept MiPro with a 250 ml or 1 L granulation bowl.
[0430] Screening/Weighing
[0431] Compound 1 and excipients may be screened prior to or after weigh-out. Possible screen sizes are mesh 20, mesh 40, or mesh 60. Compound 1 may be pre-blended with one or more of the excipients to simplify screening.
[0432] Granulation Operation
[0433] Granulation Fluid--SLS and binder are added to purified water and mixed until dissolved. A suitable ratio is 2.5% w/w SLS and 10.0% w/w PVP K30 in water.
[0434] Granulation--The excipients and compound 1 are added to the granulation bowl. The order of addition may be Compound 1, disintegrant, diluent, and filler. The components may be mixed in the 250 ml bowl for 1 minute at impeller speed 1000 RPM and chopper speed 1000 RPM. Granulation may be performed at an impeller speed of 2000 RPM with a chopper speed of 4000 RPM while adding the granulation fluid with a syringe pump at 1.5 to 4.5 g/min. The fluid addition time may be 4 to 12 minutes. After the required binder fluid is added, the granules may be wet-massed for about 10 seconds to about 1 minute. One notable advantage of the present high shear granulation process is using a granulation fluid that comprises both a surfactant and the binder for better granulation through increased wettability. In one embodiment, the surfactant is SLS.
[0435] Milling
[0436] The granules may be reduced in size using a screen mill or a cone mill.
[0437] Drying
[0438] The granules may be dried using a vacuum oven, tray dryer, bi-conical dryer, or fluid bed drier. The granules have been dried using a vacuum oven with a nitrogen purge.
[0439] Blending
[0440] The granules may be blended with extra-granular excipients. The granules have been blended with extra-granular disintegrant, diluent, filler, and lubricant. The granules have been blended using the Turbula blender for 3 minutes pre-lubricant and 1 minute with lubricant. A larger scale blender such as a 4-quart V-shell blender may be used.
[0441] Compression
[0442] The compression blend has been compressed into tablets using a single station Riva MiniPress with 8 mm, or 10 mm tooling. The weight of the tablets for a 100 mg dose may be about 160, 200, or 250 mg.
[0443] Film Coating
[0444] Tablets may be film coated using a pan coater, such as, for example an O'Hara Labcoat.
[0445] Printing
[0446] Film coated tablets may be printed with a monogram on one or both tablet faces with, for example, a Hartnett Delta printer.
[0447] Tablet Formation From Continuous Twin Screw Wet Granulation Process
[0448] Equipment/Process
[0449] Equipment
[0450] Granulator: ConsiGma or Leistritz or Thermo Fisher twin screw granulator.
[0451] Screening/Weighing
[0452] Compound 1 and excipients may be screened prior to or after weigh-out. Possible screen sizes are mesh 20, mesh 40, or mesh 60. Compound 1 may be pre-blended with one or more of the excipients to simplify screening.
[0453] Blending
[0454] Compound 1 and excipients may be added to the blender in different order. The blending may be performed in a Turbula blender, a v-shell blender, a bin blender, or a continuous blender. The components may be blended for 10 minutes for batch blenders or continuously for a continuous blender.
[0455] Granulation Operation
[0456] Granulation Fluid--SLS and binder are added to purified water and mixed until dissolved. A suitable ratio is 2.5% w/w SLS and 10.0% w/w PVP K30 in water.
[0457] Granulation--The blend containing Compound 1 and excipients may be dosed into the twin screw granulator using a Loss in Weight feeder at a rate of 10 kg/hr. The granulation fluid may be added using a peristaltic pump at a rate of 3.5 kg/hr. The granulator may be run at a speed of 400 RPM. A notable advantage of the present twin screw wet granulation process is using a granulation fluid that comprises both a surfactant and the binder for better granulation through increased wettability. In one embodiment, the surfactant is SLS. Another notable advantage is that because the process is continuous and at any moment in time only a limited amount of material is processed, the process can be well controlled and results in a high quality product.
[0458] Milling
[0459] The granules may be reduced in size using a screen mill or a cone mill
[0460] Drying
[0461] The granules may be dried using a vacuum oven, tray dryer, bi-conical dryer, or fluid bed drier.
[0462] Blending
[0463] The granules may be blended with extra-granular excipients. The granules have been blended using a 300 liter bin blender for 60 revolutions.
[0464] Compression
[0465] The compression blend has been compressed into tablets using a Courtoy Modul P rotary press
[0466] Film Coating
[0467] Tablets may be film coated using a pan coater, such as, for example an O'Hara Labcoat.
[0468] Printing
[0469] Film coated tablets may be printed with a monogram on one or both tablet faces with, for example, a Hartnett Delta printer.
[0470] Dosing Administration Schedule
[0471] In another aspect, the invention relates to a method of treating a CFTR mediated disease in a subject comprising administering to a subject in need thereof an effective amount of the pharmaceutical composition provided by the invention. In another embodiment, the pharmaceutical composition is administered to the subject once every two weeks. In another embodiment, the pharmaceutical composition is administered to the subject once a week. In another embodiment, the pharmaceutical composition is administered to the subject once every three days. In another embodiment, the pharmaceutical composition is administered to the subject once a day. In one embodiment, when the pharmaceutical composition is a tablet according to Table 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 dosing is once a day.
Assays
[0472] Assays for Detecting and Measuring F508del-CFTR Correction Properties of Compounds
[0473] Membrane potential optical methods for assaying F508del-CFTR modulation properties of compounds.
[0474] The optical membrane potential assay utilized voltage-sensitive FRET sensors described by Gonzalez and Tsien (See Gonzalez, J. E. and R. Y. Tsien (1995) "Voltage sensing by fluorescence resonance energy transfer in single cells" Biophys J 69(4): 1272-80, and Gonzalez, J. E. and R. Y. Tsien (1997) "Improved indicators of cell membrane potential that use fluorescence resonance energy transfer" Chem Biol 4(4): 269-77) in combination with instrumentation for measuring fluorescence changes such as the Voltage/Ion Probe Reader (VIPR) (See Gonzalez, J. E., K. Oades, et al. (1999) "Cell-based assays and instrumentation for screening ion-channel targets" Drug Discov Today 4(9): 431-439).
[0475] These voltage sensitive assays are based on the change in fluorescence resonant energy transfer (FRET) between the membrane-soluble, voltage-sensitive dye, DiSBAC.sub.2(3), and a fluorescent phospholipid, CC2-DMPE, which is attached to the outer leaflet of the plasma membrane and acts as a FRET donor. Changes in membrane potential (V.sub.m) cause the negatively charged DiSBAC.sub.2(3) to redistribute across the plasma membrane and the amount of energy transfer from CC2-DMPE changes accordingly. The changes in fluorescence emission were monitored using VIPR.TM. II, which is an integrated liquid handler and fluorescent detector designed to conduct cell-based screens in 96- or 384-well microtiter plates.
1. Identification of Correction Compounds
[0476] To identify small molecules that correct the trafficking defect associated with F508del-CFTR; a single-addition HTS assay format was developed. The cells were incubated in serum-free medium for 16 hrs at 37.degree. C. in the presence or absence (negative control) of test compound. As a positive control, cells plated in 384-well plates were incubated for 16 hrs at 27.degree. C. to "temperature-correct" F508del-CFTR. The cells were subsequently rinsed 3.times. with Krebs Ringers solution and loaded with the voltage-sensitive dyes. To activate F508del-CFTR, 10 .mu.M forskolin and the CFTR potentiator, genistein (20 .mu.M), were added along with Cl.sup.--free medium to each well. The addition of Cl.sup.--free medium promoted Cl.sup.- efflux in response to F508del-CFTR activation and the resulting membrane depolarization was optically monitored using the FRET-based voltage-sensor dyes.
2. Identification of Potentiator Compounds
[0477] To identify potentiators of F508del-CFTR, a double-addition HTS assay format was developed. During the first addition, a Cl.sup.--free medium with or without test compound was added to each well. After 22 sec, a second addition of Cl.sup.--free medium containing 2-10 .mu.M forskolin was added to activate F508del-CFTR. The extracellular Cl.sup.- concentration following both additions was 28 mM, which promoted Cl.sup.- efflux in response to F508del-CFTR activation and the resulting membrane depolarization was optically monitored using the FRET-based voltage-sensor dyes.
3. Solutions
[0478] Bath Solution #1: (in mM) NaCl 160, KCl 4.5, CaCl.sub.2 2, MgCl.sub.2 1, HEPES 10, pH 7.4 with NaOH.
[0479] Chloride-free bath solution: Chloride salts in Bath Solution #1 are substituted with gluconate salts.
[0480] CC2-DMPE: Prepared as a 10 mM stock solution in DMSO and stored at -20.degree. C. DiSBAC.sub.2(3): Prepared as a 10 mM stock in DMSO and stored at -20.degree. C.
4. Cell Culture
[0481] NIH3T3 mouse fibroblasts stably expressing F508del-CFTR are used for optical measurements of membrane potential. The cells are maintained at 37.degree. C. in 5% CO.sub.2 and 90% humidity in Dulbecco's modified Eagle's medium supplemented with 2 mM glutamine, 10% fetal bovine serum, 1.times. NEAA, .beta.-ME, 1.times. pen/strep, and 25 mM HEPES in 175 cm.sup.2 culture flasks. For all optical assays, the cells were seeded at 30,000/well in 384-well matrigel-coated plates and cultured for 2 hrs at 37.degree. C. before culturing at 27.degree. C. for 24 hrs for the potentiator assay. For the correction assays, the cells are cultured at 27.degree. C. or 37.degree. C. with and without compounds for 16-24 hours.
[0482] Electrophysiological Assays for Assaying F508del-CFTR Modulation Properties of Compounds
1. Ussing Chamber Assay
[0483] Using chamber experiments were performed on polarized epithelial cells expressing F508del-CFTR to further characterize the F508del-CFTR modulators identified in the optical assays. FRT.sup..DELTA.F508-CFTR epithelial cells grown on Costar Snapwell cell culture inserts were mounted in an Ussing chamber (Physiologic Instruments, Inc., San Diego, Calif.), and the monolayers were continuously short-circuited using a Voltage-clamp System (Department of Bioengineering, University of Iowa, Iowa, and, Physiologic Instruments, Inc., San Diego, Calif.). Transepithelial resistance was measured by applying a 2-mV pulse. Under these conditions, the FRT epithelia demonstrated resistances of 4 K.OMEGA./cm.sup.2 or more. The solutions were maintained at 27.degree. C. and bubbled with air. The electrode offset potential and fluid resistance were corrected using a cell-free insert. Under these conditions, the current reflects the flow of Cl.sup.- through F508del-CFTR expressed in the apical membrane. The I.sub.SC was digitally acquired using an MP100A-CE interface and AcqKnowledge software (v3.2.6; BIOPAC Systems, Santa Barbara, Calif.).
2. Identification of Correction Compounds
[0484] Typical protocol utilized a basolateral to apical membrane Cl.sup.- concentration gradient. To set up this gradient, normal ringer was used on the basolateral membrane, whereas apical NaCl was replaced by equimolar sodium gluconate (titrated to pH 7.4 with NaOH) to give a large Cl.sup.- concentration gradient across the epithelium. All experiments were performed with intact monolayers. To fully activate F508del-CFTR, forskolin (10 .mu.M) and the PDE inhibitor, IBMX (100 .mu.M), were applied followed by the addition of the CFTR potentiator, genistein (50 .mu.M).
[0485] As observed in other cell types, incubation at low temperatures of FRT cells stably expressing F508del-CFTR increases the functional density of CFTR in the plasma membrane. To determine the activity of correction compounds, the cells were incubated with 10 .mu.M of the test compound for 24 hours at 37.degree. C. and were subsequently washed 3.times. prior to recording. The cAMP- and genistein-mediated I.sub.SC in compound-treated cells was normalized to the 27.degree. C. and 37.degree. C. controls and expressed as percentage activity. Preincubation of the cells with the correction compound significantly increased the cAMP- and genistein-mediated I.sub.SC compared to the 37.degree. C. controls.
3. Identification of Potentiator Compounds
[0486] Typical protocol utilized a basolateral to apical membrane Cl.sup.- concentration gradient. To set up this gradient, normal ringers was used on the basolateral membran























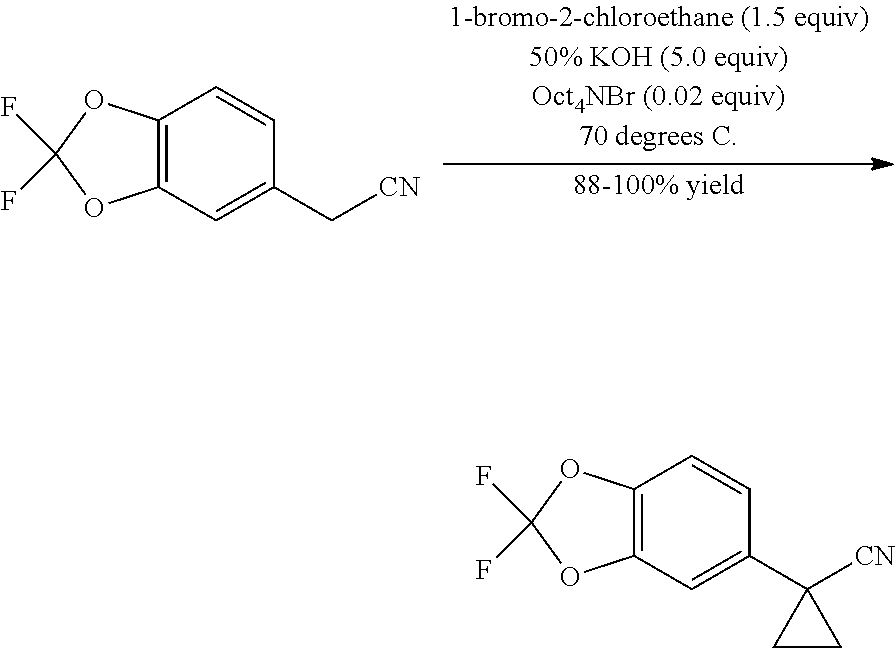









D00001

D00002

D00003

D00004

D00005

D00006

D00007
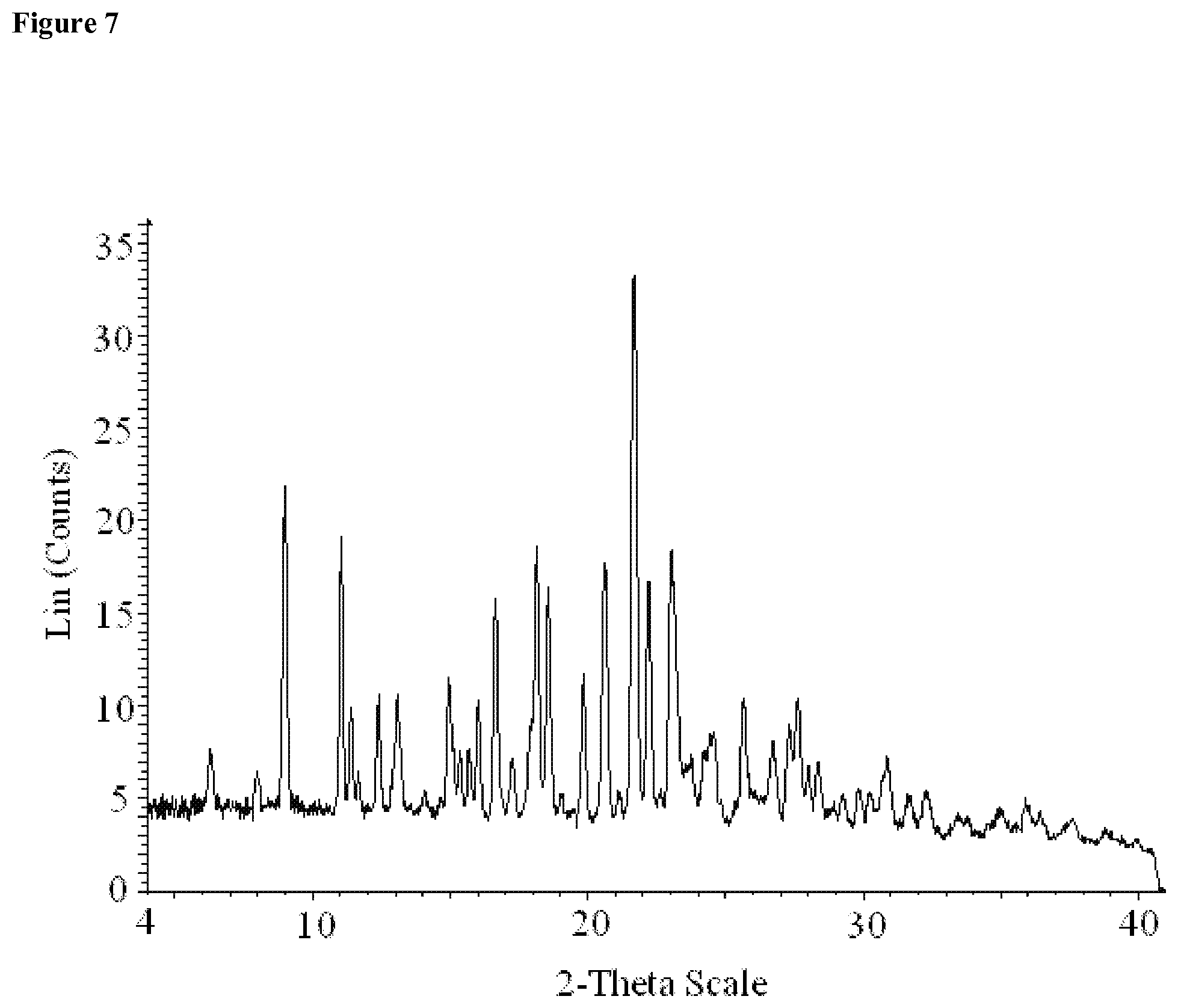
D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

D00026

D00027

D00028


P00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.