Use of an Acyclic Picolinamide Compound as a Fungicide for Control of Phytopathogenic Fungi in Vegetables
Gallup; Courtney ; et al.
U.S. patent application number 16/610100 was filed with the patent office on 2020-03-19 for use of an acyclic picolinamide compound as a fungicide for control of phytopathogenic fungi in vegetables. This patent application is currently assigned to Dow AgroSciences LLC. The applicant listed for this patent is Dow AgroSciences LLC. Invention is credited to Valentino Bosco, Alejandro Calixto, Courtney Gallup, Marsha Martin, Chenglin Yao, Alisa Ye Yu.
| Application Number | 20200085047 16/610100 |
| Document ID | / |
| Family ID | 64016718 |
| Filed Date | 2020-03-19 |


| United States Patent Application | 20200085047 |
| Kind Code | A1 |
| Gallup; Courtney ; et al. | March 19, 2020 |
Use of an Acyclic Picolinamide Compound as a Fungicide for Control of Phytopathogenic Fungi in Vegetables
Abstract
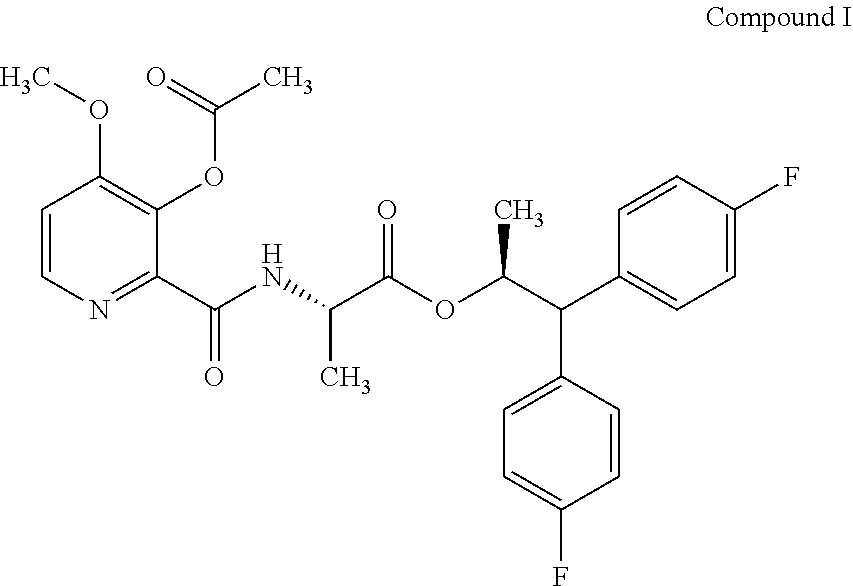
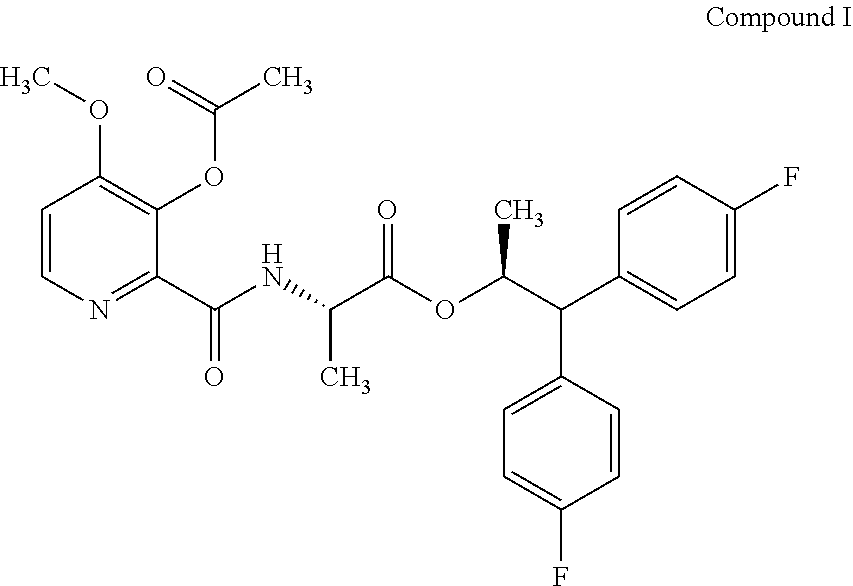
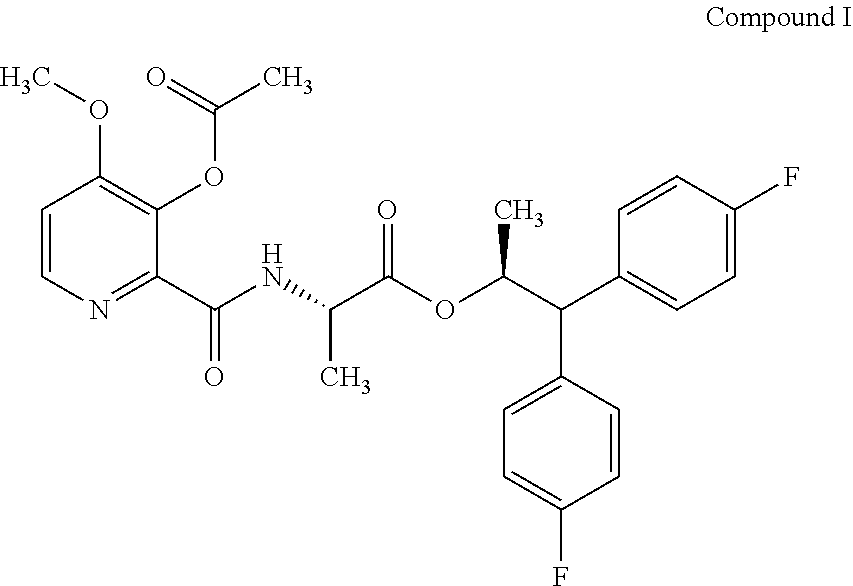
The present disclosure is related to the field of agrochemicals, including compound I and its use to control fungal diseases in agriculturally useful vegetable crops. ##STR00001##
| Inventors: | Gallup; Courtney; (Davenport, IA) ; Bosco; Valentino; (Lauzacco, IT) ; Yao; Chenglin; (Westfield, IN) ; Yu; Alisa Ye; (Shanghai, CN) ; Calixto; Alejandro; (Wesley Chapel, FL) ; Martin; Marsha; (Columbus, OH) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Dow AgroSciences LLC Indianapolis IN |
||||||||||
| Family ID: | 64016718 | ||||||||||
| Appl. No.: | 16/610100 | ||||||||||
| Filed: | May 2, 2018 | ||||||||||
| PCT Filed: | May 2, 2018 | ||||||||||
| PCT NO: | PCT/US2018/030554 | ||||||||||
| 371 Date: | November 1, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62500195 | May 2, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A01N 43/54 20130101; C07C 229/08 20130101; A01N 43/653 20130101; C07D 249/08 20130101; C07D 417/12 20130101; A01N 43/40 20130101; A01N 43/78 20130101; A01N 43/56 20130101; A61K 9/0053 20130101; A01N 43/40 20130101; A01N 25/04 20130101; A01N 25/30 20130101 |
| International Class: | A01N 43/40 20060101 A01N043/40; A01N 43/54 20060101 A01N043/54; C07C 229/08 20060101 C07C229/08; A01N 43/653 20060101 A01N043/653; A01N 43/56 20060101 A01N043/56 |
Claims
1. A method of controlling fungal diseases in vegetable crops that are at risk of being diseased comprising the steps of: contacting at least a portion of a plant and/or an area adjacent to a plant with a composition including compound I. ##STR00004## wherein said compound is effective against a plant pathogen.
2. The method of claim 1 wherein the composition is ##STR00005##
3. The method of claim 1, wherein the composition further includes at least one additional agriculturally active ingredient selected from the group consisting of: an insecticide, an herbicide, and a fungicide.
4. The method of claim 1, wherein the fungal pathogen is selected from the group consisting of the causal agents of: black leaf spot of cabbage (Alternaria brassicicola), Sclerotinia rot of lettuce (Sclerotinia sclerotiorum), anthracnose of hot pepper (Colletotrichum capsici), powdery mildew of cucumber (Erysiphe cichoracearum), early blight of tomato (Alternaria solani), leaf mold of tomato (Mycovellosiella fulva), gummy stem blight of watermelon (Stagonosporopsis cucurbitacearum), gray mold of broad bean (Botrytis cinerea), target spot of tomato (Corynespora cassiicola), and anthracnose of tomato (Colletotrichum coccodes).
5. The method of claim 2, wherein the composition further includes at least one additional agriculturally active ingredient selected from the group consisting of: an insecticide, an herbicide, and a fungicide.
6. The method of claim 2, wherein the fungal pathogen is selected from the group consisting of the causal agents of: black leaf spot of cabbage (Alternaria brassicicola), Sclerotinia rot of lettuce (Sclerotinia sclerotiorum), anthracnose of hot pepper (Colletotrichum capsici), powdery mildew of cucumber (Erysiphe cichoracearum), early blight of tomato (Alternaria solani), leaf mold of tomato (Mycovellosiella fulva), gummy stem blight of watermelon (Stagonosporopsis cucurbitacearum), gray mold of broad bean (Botrytis cinerea), target spot of tomato (Corynespora cassiicola), and anthracnose of tomato (Colletotrichum coccodes).
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This is a national phase entry under 35 U.S.C. .sctn. 371 of international patent application PCT/US18/030554, filed on May 2, 2018 and published in English as international patent publication WO2018204432 on Nov. 8, 2018, which claims the benefit of U.S. Provisional Patent Application Ser. No. 62/500,172 filed May 2, 2017, which is expressly incorporated by reference herein.
FIELD
[0002] This present disclosure is related to the field of the use of (S)-1,1-bis(4-fluorophenyl)propan-2-yl (3-acetoxy-4-methoxypicolinoyl)-L-alaninate to control fungal diseases in vegetables.
BACKGROUND AND SUMMARY
[0003] Fungicides are compounds, of natural or synthetic origin, which act to protect and cure plants against damage caused by agriculturally-relevant fungi. Generally, no single fungicide is useful in all situations. Consequently, research is ongoing to produce fungicides that may have better performance, are easier to use, and cost less.
[0004] The present disclosure relates to (S)-1,1-bis(4-fluorophenyl)propan-2-yl (3-acetoxy-4-methoxypicolinoyl)-L-alaninate (compound I) and its use as a fungicide. Compound I may offer protection against ascomycetes, basidiomycetes, and deuteromycetes.
[0005] One embodiment of the present disclosure includes a method of controlling a pathogen-induced disease in a plant that is at risk of being diseased from the pathogen comprising contacting the plant or an area adjacent to the plant with a composition including compound I.
[0006] Another embodiment of the present disclosure is a use of compound I for protection of a plant against attack by a phytopathogenic organism or the treatment of a plant infested by a phytopathogenic organism, comprising the application of compound I, or a composition including compound I to soil, a plant, a part of a plant, foliage, and/or seeds.
[0007] Additionally, another embodiment of the present disclosure is a composition useful for protecting a plant against attack by a phytopathogenic organism and/or treatment of a plant infested by a phytopathogenic organism comprising compound I and a phytologically acceptable carrier material.
DETAILED DESCRIPTION
[0008] One exemplary embodiment of the present disclosure includes mixtures for controlling the growth of fungi, the mixture including compound I:
##STR00002##
[0009] Compound I of the present disclosure may be applied by any of a variety of known techniques, either as compound I or as formulations comprising compound I. For example, compound I may be applied to the roots, stems, seeds, flowers, or foliage of plants for the control of various fungi, without damaging the commercial value of the plants. Compound I may also be applied as a foliar spray, soil spray, soil incorporation, chemigation, soil drench, soil injection, or seed treatment. The material may be applied in the form of any of the generally used formulation types, for example, as solutions, dusts, wettable powders, flowable concentrates, or emulsifiable concentrates.
[0010] Preferably, compound I of the present disclosure is applied in the form of a formulation, including compound I with a phytologically acceptable carrier. Concentrated formulations may be dispersed in water or other liquids for application, or formulations may be dust-like or granular, which may then be applied without further treatment. The formulations can be prepared according to procedures that are conventional in the agricultural chemical art.
[0011] The present disclosure contemplates all vehicles by which compound I may be formulated for delivery and use as a fungicide. Typically, formulations are applied as aqueous suspensions or emulsions. Such suspensions or emulsions may be produced from water-soluble, water-suspendible, or emulsifiable formulations which are solids, usually known as wettable powders; or liquids, usually known as emulsifiable concentrates, aqueous suspensions, or suspension concentrates. As will be readily appreciated, any material to which compound I may be added may be used, provided it yields the desired utility without significant interference with the activity of compound I as an antifungal agent.
[0012] Wettable powders, which may be compacted to form water-dispersible granules, comprise an intimate mixture including compound I, an inert carrier and surfactants. The concentration of compound I in the wettable powder may be from about 10 percent to about 90 percent by weight based on the total weight of the wettable powder, more preferably about 25 weight percent to about 75 weight percent. In the preparation of wettable powder formulations, compound I may be compounded with any finely divided solid, such as prophyllite, talc, chalk, gypsum, Fuller's earth, bentonite, attapulgite, starch, casein, gluten, montmorillonite clays, diatomaceous earths, purified silicates or the like. In such operations, the finely divided carrier and surfactants are typically blended with compound I and milled.
[0013] Emulsifiable concentrates of compound I may comprise a convenient concentration, such as from about 10 weight percent to about 50 weight percent of compound I, in a suitable liquid, based on the total weight of the concentrate. Compound I may be dissolved in an inert carrier, which is either a water-miscible solvent or a mixture of water-immiscible organic solvents, and emulsifiers. The concentrates may be diluted with water and oil to form spray mixtures in the form of oil-in-water emulsions. Useful organic solvents include aromatics, especially the high-boiling naphthalenic and olefinic portions of petroleum, such as heavy aromatic naphtha. Other organic solvents may also be used, for example, terpenic solvents, including rosin derivatives, aliphatic ketones, such as cyclohexanone, and complex alcohols, such as 2-ethoxyethanol.
[0014] Emulsifiers which may be advantageously employed herein may be readily determined by those skilled in the art and include various nonionic, anionic, cationic and amphoteric emulsifiers, or a blend of two or more emulsifiers. Examples of nonionic emulsifiers useful in preparing the emulsifiable concentrates include the polyalkylene glycol ethers and condensation products of alkyl and aryl phenols, aliphatic alcohols, aliphatic amines or fatty acids with ethylene oxide, propylene oxides such as the ethoxylated alkyl phenols and carboxylic esters solubilized with the polyol or polyoxyalkylene. Cationic emulsifiers include quaternary ammonium compounds and fatty amine salts. Anionic emulsifiers include the oil-soluble salts (e.g., calcium) of alkylaryl sulphonic acids, oil-soluble salts or sulfated polyglycol ethers and appropriate salts of phosphated polyglycol ether.
[0015] Representative organic liquids which may be employed in preparing the emulsifiable concentrates of compound I of the present invention are the aromatic liquids such as xylene, propyl benzene fractions; or mixed naphthalene fractions, mineral oils, substituted aromatic organic liquids such as dioctyl phthalate; kerosene; dialkyl amides of various fatty acids, particularly the dimethyl amides of fatty glycols and glycol derivatives such as the n-butyl ether, ethyl ether or methyl ether of diethylene glycol, and the methyl ether of triethylene glycol and the like. Mixtures of two or more organic liquids may also be employed in the preparation of the emulsifiable concentrate. Organic liquids include xylene, and propyl benzene fractions, with xylene being most preferred in some cases. Surface-active dispersing agents are typically employed in liquid formulations and in an amount of from 0.1 to 20 percent by weight based on the combined weight of the dispersing agent with compound I. The formulations can also contain other compatible additives, for example, plant growth regulators and other biologically active compounds used in agriculture.
[0016] Aqueous suspensions including compound I may be dispersed in an aqueous vehicle at a concentration in the range from about 5 to about 50 weight percent, based on the total weight of the aqueous suspension. Suspensions are prepared by finely grinding compound I, and vigorously mixing the ground material into a vehicle comprised of water and surfactants chosen from the same types discussed above. Other components, such as inorganic salts and synthetic or natural gums, may also be added to increase the density and viscosity of the aqueous vehicle.
[0017] Compound I may also be applied as a granular formulation, which is particularly useful for applications to the soil. Granular formulations generally contain from about 0.5 to about 10 weight percent, based on the total weight of the granular formulation of compound I, dispersed in an inert carrier which consists entirely or in large part of coarsely divided inert material such as attapulgite, bentonite, diatomite, clay or a similar inexpensive substance. Such formulations are usually prepared by dissolving compound I in a suitable solvent and applying it to a granular carrier which has been preformed to the appropriate particle size, in the range of from about 0.5 to about 3 mm. A suitable solvent is a solvent in which compound I is substantially or completely soluble. Such formulations may also be prepared by making a dough or paste of the carrier and compound I and solvent, and crushing and drying to obtain the desired granular particle.
[0018] Dusts containing compound I may be prepared by intimately mixing compound I in powdered form with a suitable dusty agricultural carrier, such as, for example, kaolin clay, ground volcanic rock, and the like. Dusts can suitably contain from about 1 to about 10 weight percent of compound I, based on the total weight of the dust.
[0019] The formulations may additionally contain adjuvant surfactants to enhance deposition, wetting and penetration of compound I onto the target crop and organism. These adjuvant surfactants may optionally be employed as a component of the formulation or as a tank mix. The amount of adjuvant surfactant will typically vary from 0.01 to 1.0 percent by volume, based on a spray-volume of water, preferably 0.05 to 0.5 volume percent. Suitable adjuvant surfactants include, but are not limited to ethoxylated nonyl phenols, ethoxylated synthetic or natural alcohols, salts of the esters or sulphosuccinic acids, ethoxylated organosilicones, ethoxylated fatty amines and blends of surfactants with mineral or vegetable oils. The formulations may also include oil-in-water emulsions such as those disclosed in U.S. patent application Ser. No. 11/495,228, the disclosure of which is expressly incorporated by reference herein.
[0020] In certain instances, it would be beneficial for formulations of compound I to be sprayed via an aerial application using aircraft or helicopters. The exact components of these aerial applications depends upon the crop being treated. Aerial applications for vegetables utilize spray volumes preferably from 15 to 50 liters per hectare (L/ha) with standard surfactant, wetting, sticking, spreading or penetrating type additives such as non-ionic surfactants, organosilicones, or crop oil concentrates, preferably from 0.05 to 15 percent, based on a spray volume of water.
[0021] The formulations may optionally include combinations that contain other pesticidal compounds. Such additional pesticidal compounds may be fungicides, insecticides, herbicides, nematocides, miticides, arthropodicides, bactericides, additives such as non-ionic surfactants, organosilicones, or crop oil concentrates, or combinations thereof that are compatible with the compounds of the present invention in the medium selected for application, and not antagonistic to the activity of the present compounds. Accordingly, in such embodiments, the other pesticidal compound is employed as a supplemental toxicant for the same or for a different pesticidal use. Compound I and the pesticidal compound in the combination can generally be present in a weight ratio of from 1:100 to100:1.
[0022] Compound I of the present disclosure may also be combined with other fungicides to form fungicidal mixtures and synergistic mixtures thereof. Compound I of the present disclosure is often applied in conjunction with one or more other fungicides to control a wider variety of undesirable diseases. When used in conjunction with other fungicide(s), the presently claimed compound I may be formulated with the other fungicide(s), tank-mixed with the other fungicide(s) or applied sequentially with the other fungicide(s). Such other fungicides may include 2-(thiocyanatomethylthio)-benzothiazole, 2-phenylphenol, 8-hydroxyquinoline sulfate, ametoctradin, amisulbrom, antimycin, Ampelomyces quisqualis, azaconazole, azoxystrobin, Bacillus subtilis, Bacillus subtilis strain QST713, benalaxyl, benomyl, benthiavalicarb-isopropyl, benzylaminobenzene-sulfonate (BABS) salt, bicarbonates, biphenyl, bismerthiazol, bitertanol, bixafen, blasticidin-S, borax, Bordeaux mixture, boscalid, bromuconazole, bupirimate, calcium polysulfide, captafol, captan, carbendazim, carboxin, carpropamid, carvone, chlazafenone, chloroneb, chlorothalonil, chlozolinate, Coniothyrium minitans, copper hydroxide, copper octanoate, copper oxychloride, copper sulfate, copper sulfate (tribasic), cuprous oxide, cyazofamid, cyflufenamid, cymoxanil, cyproconazole, cyprodinil, dazomet, debacarb, diammonium ethylenebis-(dithiocarbamate), dichlofluanid, dichlorophen, diclocymet, diclomezine, dichloran, diethofencarb, difenoconazole, difenzoquat ion, diflumetorim, dimethomorph, dimoxystrobin, diniconazole, diniconazole-M, dinobuton, dinocap, diphenylamine, dithianon, dodemorph, dodemorph acetate, dodine, dodine free base, edifenphos, enestrobin, enestroburin, epoxiconazole, ethaboxam, ethoxyquin, etridiazole, famoxadone, fenamidone, fenarimol, fenbuconazole, fenfuram, fenhexamid, fenoxanil, fenpiclonil, fenpropidin, fenpropimorph, fenpyrazamine, fentin, fentin acetate, fentin hydroxide, ferbam, ferimzone, fluazinam, fludioxonil, flumorph, fluopicolide, fluopyram, fluoroimide, fluoxastrobin, fluquinconazole, flusilazole, flusulfamide, flutianil, flutolanil, flutriafol, fluxapyroxad, folpet, formaldehyde, fosetyl, fosetyl-aluminium, fuberidazole, furalaxyl, furametpyr, guazatine, guazatine acetates, GY-81, hexachlorobenzene, hexaconazole, hymexazol, imazalil, imazalil sulfate, imibenconazole, iminoctadine, iminoctadine triacetate, iminoctadine tris(albesilate), iodocarb, ipconazole, ipfenpyrazolone, iprobenfos, iprodione, iprovalicarb, isoprothiolane, isopyrazam, isotianil, kasugamycin, kasugamycin hydrochloride hydrate, kresoxim-methyl, laminarin, mancopper, mancozeb, mandipropamid, maneb, mefenoxam, mepanipyrim, mepronil, meptyl-dinocap, mercuric chloride, mercuric oxide, mercurous chloride, metalaxyl, metalaxyl-M, metam, metam-ammonium, metam-potassium, metam-sodium, metconazole, methasulfocarb, methyl iodide, methyl isothiocyanate, metiram, metominostrobin, metrafenone, mildiomycin, myclobutanil, nabam, nitrothal-isopropyl, nuarimol, octhilinone, ofurace, oleic acid (fatty acids), orysastrobin, oxadixyl, oxine-copper, oxpoconazole fumarate, oxycarboxin, pefurazoate, penconazole, pencycuron, penflufen, pentachlorophenol, pentachlorophenyllaurate, penthiopyrad, phenylmercury acetate, phosphonic acid, phthalide, picoxystrobin, polyoxin B, polyoxins, polyoxorim, potassium bicarbonate, potassium hydroxyquinoline sulfate, probenazole, prochloraz, procymidone, propamocarb, propamocarb hydrochloride, propiconazole, propineb, proquinazid, prothioconazole, pyraclostrobin, pyrametostrobin, pyraoxystrobin, pyrazophos, pyribencarb, pyributicarb, pyrifenox, pyrimethanil, pyriofenone, pyroquilon, quinoclamine, quinoxyfen, quintozene, Reynoutria sachalinensis extract, sedaxane, silthiofam, simeconazole, sodium 2-phenylphenoxide, sodium bicarbonate, sodium pentachlorophenoxide, spiroxamine, sulfur, SYP-Z048, tar oils, tebuconazole, tebufloquin, tecnazene, tetraconazole, thiabendazole, thifluzamide, thiophanate-methyl, thiram, tiadinil, tolclofos-methyl, tolylfluanid, triadimefon, triadimenol, triazoxide, tricyclazole, tridemorph, trifloxystrobin, triflumizole, triforine, triphenyltin hydroxide, triticonazole, validamycin, valifenalate, valiphenal, vinclozolin, zineb, ziram, zoxamide, Candida oleophila, Fusarium oxysporum, Gliocladium spp., Phlebiopsis gigantea, Streptomyces griseoviridis, Trichoderma spp., (RS)--N-(3,5-dichlorophenyl)-2-(methoxymethyl)-succinimide, 1,2-dichloropropane, 1,3-dichloro-1,1,3,3-tetrafluoroacetone hydrate, 1-chloro-2,4-dinitronaphthalene, 1-chloro-2-nitropropane, 2-(2-heptadecyl-2-imidazolin-1-yl)ethanol, 2,3-dihydro-5-phenyl-1,4-dithi-ine 1,1,4,4-tetraoxide, 2-methoxyethylmercury acetate, 2-methoxyethylmercury chloride, 2-methoxyethylmercury silicate, 3-(4-chlorophenyl)-5-methylrhodanine, 4-(2-nitroprop-1-enyl)phenyl thiocyanateme, aminopyrifen, ampropylfos, anilazine, azithiram, barium polysulfide, Bayer 32394, benodanil, benquinox, bentaluron, benzamacril; benzamacril-isobutyl, benzamorf, benzovindiflupyr, binapacryl, bis(methylmercury) sulfate, bis(tributyltin) oxide, buthiobate, cadmium calcium copper zinc chromate sulfate, carbamorph, CECA, chlobenthiazone, chloraniformethan, chlorfenazole, chlorquinox, climbazole, copper bis(3-phenylsalicylate), copper zinc chromate, coumoxystrobin, cufraneb, cupric hydrazinium sulfate, cuprobam, cyclafuramid, cypendazole, cyprofuram, decafentin, dichlobentiazox, dichlone, dichlozoline, diclobutrazol, dimethirimol, dinocton, dinosulfon, dinoterbon, dipymetitrone, dipyrithione, ditalimfos, dodicin, drazoxolon, EBP, enoxastrobin, ESBP, etaconazole, etem, ethirim, fenaminosulf, fenaminstrobin, fenapanil, fenitropan, fenpicoxamid, fluindapyr, fluopimomide, fluotrimazole, flufenoxystrobin, furcarbanil, furconazole, furconazole-cis, furmecyclox, furophanate, glyodine, griseofulvin, halacrinate, Hercules 3944, hexylthiofos, ICIA0858, inpyrfluxam, ipfentrifluconazole, ipflufenoquin, isofetamid, isoflucypram, isopamphos, isovaledione, mandestrobin, mebenil, mecarbinzid, mefentrifluconazole, metazoxolon, methfuroxam, methylmercury dicyandiamide, metsulfovax, metyltetraprole, milneb, mucochloric anhydride, myclozolin, N-3,5-dichlorophenyl-succinimide, N-3-nitrophenylitaconimide, natamycin, N-ethylmercurio-4-toluenesulfonanilide, nickel bis(dimethyldithiocarbamate), OCH, oxathiapiprolin, phenylmercury dimethyldithiocarbamate, phenylmercury nitrate, phosdiphen, picarbutrazox, prothiocarb; prothiocarb hydrochloride, pydiflumetofen, pyracarbolid, pyrapropoyne, pyraziflumid, pyridachlometyl, pyridinitril, pyrisoxazole, pyroxychlor, pyroxyfur, quinacetol, quinacetol sulfate, quinazamid, quinconazole, quinofumelin, rabenzazole, salicylanilide, SSF-109, sultropen, tecoram, thiadifluor, thicyofen, thiochlorfenphim, thiophanate, thioquinox, tioxymid, triamiphos, triarimol, triazbutil, trichlamide, triclopyricarb, triflumezopyrim, urbacid, zarilamid, and any combinations thereof.
[0023] Additionally, compound I of the present invention may be combined with other pesticides, including insecticides, nematicides, miticides, arthropodicides, bactericides or combinations thereof that are compatible with compound I of the present invention in the medium selected for application, and not antagonistic to the activity of compound I, to form pesticidal mixtures and synergistic mixtures thereof. Compound I of the present disclosure may be applied in conjunction with one or more other pesticides to control a wider variety of undesirable pests. When used in conjunction with other pesticides, the presently claimed compound I may be formulated with the other pesticide(s), tank mixed with the other pesticide(s) or applied sequentially with the other pesticide(s). Typical insecticides include, but are not limited to: antibiotic insecticides such as allosamidin and thuringiensin; macrocyclic lactone insecticides such as spinosad and spinetoram; avermectin insecticides such as abamectin, doramectin, emamectin, eprinomectin, ivermectin and selamectin; milbemycin insecticides such as lepimectin, milbemectin, milbemycin oxime and moxidectin; carbamate insecticides such as bendiocarb and carbaryl; benzofuranyl methylcarbamate insecticides such as benfuracarb, carbofuran, carbosulfan, decarbofuran and furathiocarb; dimethylcarbamate insecticides dimitan, dimetilan, hyquincarb and pirimicarb; oxime carbamate insecticides such as alanycarb, aldicarb, aldoxycarb, butocarboxim, butoxycarboxim, methomyl, nitrilacarb, oxamyl, tazimcarb, thiocarboxime, thiodicarb and thiofanox; phenyl methylcarbamate insecticides such as allyxycarb, aminocarb, bufencarb, butacarb, carbanolate, cloethocarb, dicresyl, dioxacarb, EMPC, ethiofencarb, fenethacarb, fenobucarb, isoprocarb, methiocarb, metolcarb, mexacarbate, promacyl, promecarb, propoxur, trimethacarb, XMC and xylylcarb; dessicant insecticides such as boric acid, diatomaceous earth and silica gel; diamide insecticides such as broflanilide, chlorantraniliprole, cyantraniliprole, cyclaniliprole, cyhalodiamide, flubendiamide, tetrachlorantraniliprole, and tetraniliprole; diarylisoxazoline insecticides such as fluxametamide; dinitrophenol insecticides such as dinex, dinoprop, dinosam and DNOC; fluorine insecticides such as barium hexafluorosilicate, cryolite, sodium fluoride, sodium hexafluorosilicate and sulfluramid; formamidine insecticides such as amitraz, chlordimeform, formetanate and formparanate; fumigant insecticides such as acrylonitrile, carbon disulfide, carbon tetrachloride, chloroform, chloropicrin, para-dichlorobenzene, 1,2-dichloropropane, ethyl formate, ethylene dibromide, ethylene dichloride, ethylene oxide, hydrogen cyanide, iodomethane, methyl bromide, methylchloroform, methylene chloride, naphthalene, phosphine, sulfuryl fluoride and tetrachloroethane; inorganic insecticides such as borax, calcium polysulfide, copper oleate, mercurous chloride, potassium thiocyanate and sodium thiocyanate; chitin synthesis inhibitors such as bistrifluron, buprofezin, chlorfluazuron, cyromazine, diflubenzuron, flucycloxuron, flufenoxuron, hexaflumuron, lufenuron, novaluron, noviflumuron, penfluron, teflubenzuron and triflumuron; juvenile hormone mimics such as epofenonane, fenoxycarb, hydroprene, kinoprene, methoprene, pyriproxyfen and triprene; juvenile hormones such as juvenile hormone I, juvenile hormone II and juvenile hormone III; mesoionic insecticides such as dicloromezotiaz and triflumezopyrim; moulting hormone agonists such as chromafenozide, halofenozide, methoxyfenozide and tebufenozide; moulting hormones such as .alpha.-ecdysone and ecdysterone; moulting inhibitors such as diofenolan; precocenes such as precocene I, precocene II and precocene III; unclassified insect growth regulators such as dicyclanil; nereistoxin analogue insecticides such as bensultap, cartap, thiocyclam and thiosultap; pyridylpyrazole insecticides such as tyclopyrazoflor; nicotinoid insecticides such as flonicamid; nitroguanidine insecticides such as clothianidin, dinotefuran, imidacloprid and thiamethoxam; nitromethylene insecticides such as nitenpyram and nithiazine; pyridylmethyl-amine insecticides such as acetamiprid, cycloxaprid, imidacloprid, nitenpyram, and thiacloprid; organochlorine insecticides such as bromo-DDT, camphechlor, DDT, pp'-DDT, ethyl-DDD, HCH, gamma-HCH, lindane, methoxychlor, pentachlorophenol and TDE; cyclodiene insecticides such as aldrin, bromocyclen, chlorbicyclen, chlordane, chlordecone, dieldrin, dilor, endosulfan, alpha-endosulfan, endrin, HEOD, heptachlor, HHDN, isobenzan, isodrin, kelevan and mirex; organophosphate insecticides such as bromfenvinfos, chlorfenvinphos, crotoxyphos, dichlorvos, dicrotophos, dimethylvinphos, fospirate, heptenophos, methocrotophos, mevinphos, monocrotophos, naled, naftalofos, phosphamidon, propaphos, TEPP and tetrachlorvinphos; organothiophosphate insecticides such as dioxabenzofos, fosmethilan and phenthoate; aliphatic organothiophosphate insecticides such as acethion, amiton, cadusafos, chlorethoxyfos, chlormephos, demephion, demephion-O, demephion-S, demeton, demeton-O, demeton-S, demeton-methyl, demeton-O-methyl, demeton-S-methyl, demeton-S-methylsulphon, disulfoton, ethion, ethoprophos, IPSP, isothioate, malathion, methacrifos, oxydemeton-methyl, oxydeprofos, oxydisulfoton, phorate, sulfotep, terbufos and thiometon; aliphatic amide organothiophosphate insecticides such as amidithion, cyanthoate, dimethoate, ethoate-methyl, formothion, mecarbam, omethoate, prothoate, sophamide and vamidothion; oxime organothiophosphate insecticides such as chlorphoxim, phoxim and phoxim-methyl; heterocyclic organothiophosphate insecticides such as azamethiphos, coumaphos, coumithoate, dioxathion, endothion, menazon, morphothion, phosalone, pyraclofos, pyridaphenthion and quinothion; benzothiopyran organothiophosphate insecticides such as dithicrofos and thicrofos; benzotriazine organothiophosphate insecticides such as azinphos-ethyl and azinphos-methyl; isoindole organothiophosphate insecticides such as dialifos and phosmet; isoxazole organothiophosphate insecticides such as isoxathion and zolaprofos; pyrazolopyrimidine organothiophosphate insecticides such as chlorprazophos and pyrazophos; pyridine organothiophosphate insecticides such as chlorpyrifos and chlorpyrifos-methyl; pyrimidine organothiophosphate insecticides such as butathiofos, diazinon, etrimfos, lirimfos, pirimiphos-ethyl, pirimiphos-methyl, primidophos, pyrimitate and tebupirimfos; quinoxaline organothiophosphate insecticides such as quinalphos and quinalphos-methyl; thiadiazole organothiophosphate insecticides such as athidathion, lythidathion, methidathion and prothidathion; triazole organothiophosphate insecticides such as isazofos and triazophos; phenyl organothiophosphate insecticides such as azothoate, bromophos, bromophos-ethyl, carbophenothion, chlorthiophos, cyanophos, cythioate, dicapthon, dichlofenthion, etaphos, famphur, fenchlorphos, fenitrothion fensulfothion, fenthion, fenthion-ethyl, heterophos, j odfenphos, mesulfenfos, parathion, parathion-methyl, phenkapton, phosnichlor, profenofos, prothiofos, sulprofos, temephos, trichlormetaphos-3 and trifenofos; phosphonate insecticides such as butonate and trichlorfon; phosphonothioate insecticides such as mecarphon; phenyl ethylphosphonothioate insecticides such as fonofos and trichloronat; phenyl phenylphosphonothioate insecticides such as cyanofenphos, EPN and leptophos; phosphoramidate insecticides such as crufomate, fenamiphos, fosthietan, mephosfolan, phosfolan and pirimetaphos; phosphoramidothioate insecticides such as acephate, isocarbophos, isofenphos, isofenphos-methyl, methamidophos and propetamphos; phosphorodiamide insecticides such as dimefox, mazidox, mipafox and schradan; oxadiazine insecticides such as indoxacarb; oxadiazoline insecticides such as metoxadiazone; phthalimide insecticides such as dialifos, phosmet and tetramethrin; pyrazole insecticides such as tebufenpyrad, tolefenpyrad; phenylpyrazole insecticides such as acetoprole, ethiprole, fipronil, pyrafluprole, pyriprole and vaniliprole; pyrethroid ester insecticides such as acrinathrin, allethrin, bioallethrin, barthrin, bifenthrin, kappa-bifenthrin, bioethanomethrin, chloroprallethrin, cyclethrin, cycloprothrin, cyfluthrin, beta-cyfluthrin, cyhalothrin, gamma-cyhalothrin, 1ambda-cyhalothrin, cypermethrin, alpha-cypermethrin, beta-cypermethrin, theta-cypermethrin, zeta-cypermethrin, cyphenothrin, deltamethrin, dimefluthrin, dimethrin, empenthrin, fenfluthrin, fenpirithrin, fenpropathrin, fenvalerate, esfenvalerate, flucythrinate, fluvalinate, tau-fluvalinate, furethrin, heptafluthrin, imiprothrin, meperfluthrin, metofluthrin, epsilon-metofluthrin, momfluorothrin, epsilon-momfluorothrin, permethrin, biopermethrin, transpermethrin, phenothrin, prallethrin, profluthrin, pyresmethrin, resmethrin, bioresmethrin, cismethrin, tefluthrin, kappa-tefluthrin, terallethrin, tetramethrin, tetramethylfluthrin, tralomethrin and transfluthrin; pyrethroid ether insecticides such as etofenprox, flufenprox, halfenprox, protrifenbute and silafluofen; pyrimidinamine insecticides such as flufenerim and pyrimidifen; pyrrole insecticides such as chlorfenapyr; tetramic acid insecticides such as spiropidion and spirotetramat; tetronic acid insecticides such as spiromesifen; thiourea insecticides such as diafenthiuron; urea insecticides such as flucofuron and sulcofuron; unclassified nematicides such as fluazaindolizine and tioxazafen; and unclassified insecticides such as benzpyrimoxan, closantel, copper naphthenate, crotamiton, EXD, fenazaflor, fenoxacrim, fluhexafon, flupyrimin, hydramethylnon, isoprothiolane, malonoben, metaflumizone, nifluridide, oxazolsulfyl, plifenate, pyridaben, pyridalyl, pyrifluquinazon, rafoxanide, sulfoxaflor, triarathene and triazamate, and any combinations thereof.
[0024] Additionally, compound I of the present invention may be combined with herbicides that are compatible with compound I of the present invention in the medium selected for application, and not antagonistic to the activity of compound I to form pesticidal mixtures and synergistic mixtures thereof. The fungicidal compound I of the present disclosure may be applied in conjunction with one or more herbicides to control a wide variety of undesirable plants. When used in conjunction with herbicides, the presently claimed compound I may be formulated with the herbicide(s), tank mixed with the herbicide(s) or applied sequentially with the herbicide(s). Typical herbicides include, but are not limited to: amide herbicides such as allidochlor, beflubutamid, benzadox, benzipram, bromobutide, cafenstrole, CDEA, cyprazole, dimethenamid, dimethenamid-P, diphenamid, epronaz, etnipromid, fentrazamide, flupoxam, fomesafen, halosafen, isocarbamid, isoxaben, napropamide, naptalam, pethoxamid, propyzamide, quinonamid, tebutam and tiafenacil; anilide herbicides such as chloranocryl, cisanilide, clomeprop, cypromid, diflufenican, etobenzanid, fenasulam, flufenacet, flufenican, mefenacet, mefluidide, metamifop, monalide, naproanilide, pentanochlor, picolinafen and propanil; arylalanine herbicides such as benzoylprop, flamprop and flamprop-M; chloroacetanilide herbicides such as acetochlor, alachlor, butachlor, butenachlor, delachlor, diethatyl, dimethachlor, metazachlor, metolachlor, S-metolachlor, pretilachlor, propachlor, propisochlor, prynachlor, terbuchlor, thenylchlor and xylachlor; sulfonanilide herbicides such as benzofluor, perfluidone, pyrimisulfan and profluazol; sulfonamide herbicides such as asulam, carbasulam, fenasulam and oryzalin; thioamide herbicides such as chlorthiamid; antibiotic herbicides such as bilanafos; benzoic acid herbicides such as chloramben, dicamba, 2,3,6-TBA and tricamba; pyrimidinyloxybenzoic acid herbicides such as bispyribac and pyriminobac; pyrimidinylthiobenzoic acid herbicides such as pyrithiobac; phthalic acid herbicides such as chlorthal; picolinic acid herbicides such as aminopyralid, clopyralid, florpyrauxifen, halauxifen, and picloram; quinolinecarboxylic acid herbicides such as quinclorac and quinmerac; arsenical herbicides such as cacodylic acid, CMA, DSMA, hexaflurate, MAA, MAMA, MSMA, potassium arsenite and sodium arsenite; benzoylcyclohexanedione herbicides such as fenquinotrione, lancotrione, mesotrione, sulcotrione, tefuryltrione and tembotrione; benzofuranyl alkylsulfonate herbicides such as benfuresate and ethofumesate; benzothiazole herbicides such as benzazolin; carbamate herbicides such as asulam, carboxazole chlorprocarb, dichlormate, fenasulam, karbutilate and terbucarb; carbanilate herbicides such as barban, BCPC, carbasulam, carbetamide, CEPC, chlorbufam, chlorpropham, CPPC, desmedipham, phenisopham, phenmedipham, phenmedipham-ethyl, propham and swep; cyclohexene oxime herbicides such as alloxydim, butroxydim, clethodim, cloproxydim, cycloxydim, profoxydim, sethoxydim, tepraloxydim and tralkoxydim; cyclopropylisoxazole herbicides such as isoxachlortole and isoxaflutole; dicarboximide herbicides such as cinidon-ethyl, flumezin, flumiclorac, flumioxazin and flumipropyn; dinitroaniline herbicides such as benfluralin, butralin, dinitramine, ethalfluralin, fluchloralin, isopropalin, methalpropalin, nitralin, oryzalin, pendimethalin, prodiamine, profluralin and trifluralin; dinitrophenol herbicides such as dinofenate, dinoprop, dinosam, dinoseb, dinoterb, DNOC, etinofen and medinoterb; diphenyl ether herbicides such as ethoxyfen; nitrophenyl ether herbicides such as acifluorfen, aclonifen, bifenox, chlomethoxyfen, chlornitrofen, etnipromid, fluorodifen, fluoroglycofen, fluoronitrofen, fomesafen, furyloxyfen, halosafen, lactofen, nitrofen, nitrofluorfen and oxyfluorfen; dithiocarbamate herbicides such as dazomet and metam; halogenated aliphatic herbicides such as alorac, chloropon, dalapon, flupropanate, hexachloroacetone, iodomethane, methyl bromide, monochloroacetic acid, SMA and TCA; imidazolinone herbicides such as imazamethabenz, imazamox, imazapic, imazapyr, imazaquin and imazethapyr; inorganic herbicides such as ammonium sulfamate, borax, calcium chlorate, copper sulfate, ferrous sulfate, potassium azide, potassium cyanate, sodium azide, sodium chlorate and sulfuric acid; nitrite herbicides such as bromobonil, bromoxynil, chloroxynil, cyclopyranil, dichlobenil, iodobonil, ioxynil and pyraclonil; organophosphorus herbicides such as amiprofos-methyl, anilofos, bensulide, bilanafos, butamifos, 2,4-DEP, DMPA, EBEP, fosamine, glufosinate, glufosinate-P, glyphosate and piperophos; phenoxy herbicides such as bromofenoxim, clomeprop, 2,4-DEB, 2,4-DEP, difenopenten, disul, erbon, etnipromid, fenteracol and trifopsime; oxadiazoline herbicides such as methazole, oxadiargyl, oxadiazon; oxazole herbicides such as fenoxasulfone; phenoxyacetic herbicides such as 4-CPA, 2,4-D, 3,4-DA, MCPA, MCPA-thioethyl and 2,4,5-T; phenoxybutyric herbicides such as 4-CPB, 2,4-DB, 3,4-DB, MCPB and 2,4,5-TB; phenoxypropionic herbicides such as cloprop, 4-CPP, dichlorprop, dichlorprop-P, 3,4-DP, fenoprop, mecoprop and mecoprop-P; aryloxyphenoxypropionic herbicides such as chlorazifop, clodinafop, clofop, cyhalofop, diclofop, fenoxaprop, fenoxaprop-P, fenthiaprop, fluazifop, fluazifop-P, haloxyfop, haloxyfop-P, isoxapyrifop, metamifop, propaquizafop, quizalofop, quizalofop-P and trifop; phenylenediamine herbicides such as dinitramine and prodiamine; pyrazole herbicides such as pyroxasulfone; benzoylpyrazole herbicides such as benzofenap, pyrasulfotole, pyrazolynate, pyrazoxyfen, tolpyralate, and topramezone; phenylpyrazole herbicides such as fluazolate, nipyraclofen, pioxaden and pyraflufen; pyridazine herbicides such as credazine, cyclopyrimorate, pyridafol and pyridate; pyridazinone herbicides such as brompyrazon, chloridazon, dimidazon, flufenpyr, metflurazon, norflurazon, oxapyrazon and pydanon; pyridine herbicides such as aminopyralid, cliodinate, clopyralid, dithiopyr, florpyrauxifen, fluroxypyr, halauxifen, haloxydine, picloram, picolinafen, pyriclor, thiazopyr and triclopyr; pyrimidinediamine herbicides such as iprymidam and tioclorim; quaternary ammonium herbicides such as cyperquat, diethamquat, difenzoquat, diquat, morfamquat and paraquat; thiocarbamate herbicides such as butylate, cycloate, di-allate, EPTC, esprocarb, ethiolate, isopolinate, methiobencarb, molinate, orbencarb, pebulate, prosulfocarb, pyributicarb, sulfallate, thiobencarb, tiocarbazil, tri-allate and vernolate; thiocarbonate herbicides such as dimexano, EXD and proxan; thiourea herbicides such as methiuron; triazine herbicides such as dipropetryn, indaziflam, triaziflam and trihydroxytriazine; chlorotriazine herbicides such as atrazine, chlorazine, cyanazine, cyprazine, eglinazine, ipazine, mesoprazine, procyazine, proglinazine, propazine, sebuthylazine, simazine, terbuthylazine and trietazine; methoxytriazine herbicides such as atraton, methometon, prometon, secbumeton, simeton and terbumeton; methylthiotriazine herbicides such as ametryn, aziprotryne, cyanatryn, desmetryn, dimethametryn, methoprotryne, prometryn, simetryn and terbutryn; triazinone herbicides such as ametridione, amibuzin, hexazinone, isomethiozin, metamitron, metribuzin, and trifludimoxazin; triazole herbicides such as amitrole, cafenstrole, epronaz and flupoxam; triazolone herbicides such as amicarbazone, bencarbazone, carfentrazone, flucarbazone, ipfencarbazone, propoxycarbazone, sulfentrazone and thiencarbazone-methyl; triazolopyrimidine herbicides such as cloransulam, diclosulam, florasulam, flumetsulam, metosulam, penoxsulam and pyroxsulam; uracil herbicides such as benzfendizone, bromacil, butafenacil, flupropacil, isocil, lenacil, saflufenacil and terbacil; urea herbicides such as benzthiazuron, cumyluron, cycluron, dichloralurea, diflufenzopyr, isonoruron, isouron, methabenzthiazuron, monisouron and noruron; phenylurea herbicides such as anisuron, buturon, chlorbromuron, chloreturon, chlorotoluron, chloroxuron, daimuron, difenoxuron, dimefuron, diuron, fenuron, fluometuron, fluothiuron, isoproturon, linuron, methiuron, methyldymron, metobenzuron, metobromuron, metoxuron, monolinuron, monuron, neburon, parafluron, phenobenzuron, siduron, tetrafluron and thidiazuron; pyrimidinylsulfonylurea herbicides such as amidosulfuron, azimsulfuron, bensulfuron, chlorimuron, cyclosulfamuron, ethoxysulfuron, flazasulfuron, flucetosulfuron, flupyrsulfuron, foramsulfuron, halosulfuron, imazosulfuron, mesosulfuron, metazosulfuron, nicosulfuron, orthosulfamuron, oxasulfuron, primisulfuron, propyrisulfuron, pyrazosulfuron, rimsulfuron, sulfometuron, sulfosulfuron and trifloxysulfuron; triazinylsulfonylurea herbicides such as chlorsulfuron, cinosulfuron, ethametsulfuron, iodosulfuron, iofensulfuron, metsulfuron, prosulfuron, thifensulfuron, triasulfuron, tribenuron, triflusulfuron and tritosulfuron; thiadiazolylurea herbicides such as buthiuron, ethidimuron, tebuthiuron, thiazafluron and thidiazuron; and unclassified herbicides such as acrolein, allyl alcohol, aminocyclopyrachlor, azafenidin, bentazone, benzobicyclon, bicyclopyrone, buthidazole, calcium cyanamide, cambendichlor, chlorfenac, chlorfenprop, chlorflurazole, chlorflurenol, cinmethylin, clomazone, CPMF, cresol, cyanamide, cyclopyrimorate, ortho-dichlorobenzene, dimepiperate, endothal, fluoromidine, fluridone, flurochloridone, flurtamone, fluthiacet, indanofan, methyl isothiocyanate, OCH, oxaziclomefone, pentachlorophenol, pentoxazone, phenylmercury acetate, prosulfalin, pyribenzoxim, pyriftalid, quinoclamine, rhodethanil, sulglycapin, thidiazimin, tridiphane, trimeturon, tripropindan and tritac.
[0025] Compound I of the present invention can also comprise or may be applied together and/or sequentially with further active compounds. These further compounds can be plant health stimulants, such as organic compounds, inorganic fertilizers, or micronutrient donors or other preparations that influence plant growth, such as inoculants.
[0026] In another embodiment, Compound I can also comprise or may be applied together and/or sequentially with other biological organisms, such as, but not limited to the group consisting of Bacillus strains, for example Bacillus subtilis var. amyloliquefaciens FZB24 (TAEGRP.RTM.) and Bacillus amyloliquefaciens FZB42 (RHIZOVITAL.RTM.), VotiVo.TM. Bacillus firmus, Clariva.TM. (Pasteuria nishizawae), Bacillus thuringiensis, Trichoderma spp., and/or mutants and metabolites of the respective strains that exhibit activity against insects, mites, nematodaes, and/or phytopathogens.
[0027] One embodiment of the present disclosure is a method for the control or prevention of fungal attack. This method comprises applying to the soil, plant, roots, foliage, seed or locus of the fungus, or to a locus in which the infestation is to be prevented (for example applying to cereal or grape plants), a fungicidal effective amount of compound I. Compound I is suitable for treatment of various plants at fungicidal levels, while exhibiting low phytotoxicity. Compound I may be useful both in a protectant and/or an eradicant fashion.
[0028] The compound of Formula I has been found to have significant fungicidal effects particularly for agricultural use. The compound of Formula I is particularly effective for use with agricultural crops and horticultural plants. Additional benefits may include, but are not limited to, improving the health of a plant; improving the yield of a plant (e.g. increased biomass and/or increased content of valuable ingredients); improving the vigor of a plant (e.g. improved plant growth and/or greener leaves); improving the quality of a plant (e.g. improved content or composition of certain ingredients); and improving the tolerance to abiotic and/or biotic stress of the plant.
[0029] In particular, the composition is effective in controlling a variety of undesirable fungi that infect useful vegetable crops. The composition maybe used against a variety of Ascomycete and Basidiomycete fungi, including, for example, the following representative fungi species:
[0030] On cucurbits: powdery mildew (Erysiphe cichoracearum, Sphaerotheca fuliginea), ring spot (Mycosphaerella brassicicola), blackleg (Plenodomus lingam), anthracnose (Colletotrichum higginsianum, Colletotrichum obiculare, Colletotrichum spp., Glomerella lagenarium), gummy stem blight (Mycosphaerella melonis, Didymella bryoniae, Stagonosporopsis cucurbitacearum); Alternaria leaf spot and blight (Alternaria cucumerina, Alternaria alternata f sp. cucurbitae, Alternaria spp.), Cercospora leaf spot (Cercospora citrullina, Cercospora spp.), Fusarium wilt (Fusarium oxysporum), belly rot (Rhizoctonia solani), Corynespora blight/target spot (Corynespora cassiicola), scab (Cladosporium cucumerinum), Verticillium wilt (Verticillium dahliae, Verticillium albo-atrum), black root rot (Thielaviopsis basicola), blue mold rot (Penicillium spp.), charcoal rot (Macrophomina phaseolina), and crater rot (fruit) (Myrothecium roridum);
[0031] On tomato: powdery mildew (Leveillula taurica), Septoria leaf spot (Septoria lycopersici), white mold (Sclerotinia sclerotiorum, Sclerotinia minor), target spot (Corynespora cassiicola), southern blight (Sclerotium rolfsii), gray mold (Botrytis cinerea), gray leaf spot (Stemphylium spp.), Fusarium wilt, Fusarium crown and root rot (Fusarium oxysporum), anthracnose (Colletotrichum coccodes, Colletotrichum dematium, Colletotrichum gloeosporioides, Glomerella cingulate), Alternaria stem canker and black mold (Alternaria alternata), black root rot (Chalara elegans), Cercospora leaf mold (Cercospora fuligena), charcoal rot (Macrophomina phaseolina), corky root rot (Pyrenochaeta lycopersici), Didymella stem rot (Didymella lycopersici), early blight (Alternaria solani), leaf mold (Passalora fulva, Mycovellosiella fulva), phoma rot (Phoma destructiva), and sour rot (Geotrichum candidum);
[0032] On leafy vegetables: Cercospora leaf spot (Cercospora longissimi, Cercospora spp.), gray mold/Botrytis rot (Botrytis cinerea), Alternaria leaf spot and blight (Alternaria sonchi, Alternaria spp.), powdery mildew (Erysiphe cichoracearum, Golovinomyces cichoracearum, Uncinula spp.), rust (Puccinia dioicae), Septoria leaf spot (Septoria lactucae, Septoria spp.), southern blight (Sclerotium rolfsii), basal rot (Phoma exigua), Rhizoctonia bottom rot (Rhizoctonia solani), Sclerotinia drop (Sclerotinia minor, Sclerotinia sclerotiorum), and Stemphylium leaf spot (Stemphylium botryosum);
[0033] On potato: anthracnose or black dot (Colletotrichum coccodes), brown spot and black pit (Alternaria alternata), Cercospora leaf blotch (Cercospora concors), charcoal rot (Macrophomina phaseolina), common rust (Puccinia pittieriana), deforming rust (Aecidium cantensis), early blight (Alternaria solani), Fusarium dry rot and Fusarium wilt (Fusarium spp.), gangrene (Phoma exigua f. sp. foveata), gray mold (Botrytis cinerea), black blight or Phoma leaf spot (Stagonosporopsis andigena, Phoma andigena var. andinai), powdery mildew (Erysiphe cichoracearum), Rhizoctonia canker and black scurf (Rhizoctonia solani), Rosellinia black rot (Rosellinia spp.), Septoria leaf spot (Septoria lycopersici var. malagutil), silver scurf (Helminthosporium solani), skin spot (Polyscytalum pustulans), stem rot (Sclerotium rolfsii), Thecaphora smut (Thecaphora solani), Ulocladium blight (Ulocladium atrum), Verticillium wilt (Verticillium albo-atrum), and white mold (Sclerotinia sclerotiorum);
[0034] On peppers: southern blight (Sclerotium rolfsii), powdery mildew (Leveillula taurica), anthracnose (Colletotrichum capsici, Colletorichum acutatum, Colletorichum spp.), Cercospora (frogeye) leaf spot (Cercospora capsici, Cercospora spp.), charcoal rot (Macrophomina phaseolina), damping-off and root rot (Rhizoctonia solani), Fusarium stem rot (Fusarium solani), Fusarium wilt (Fusarium oxysporum f. sp. capsici), gray leaf spot (Stemphylium spp.), gray mold (Botrytis cinerea), Verticillium wilt (Verticillium albo-atrum), and white mold (Sclerotinia sclerotiorum);
[0035] On cole crops: Alternaria leaf spot and blight (Alternaria brassicicola, Alternaria spp.), Anthracnose (Colletotrichum spp.), black leg (Leptosphaeria maculans, Phoma lingam), Cercospora leaf spot (Cercopsora spp.), Fusarium yellows and other diseases (Fusarium spp.), gray mold/Botrytis blight (Botrytis cinerea), powdery mildew (Erysiphe polygoni), Rhizoctonia rot and blight (Rhizoctonia solani), ring spot (Mycosphaerella brassicicola), Sclerotinia rot (Sclerotinia spp.), white leaf spot (Pseudocercosporella capsellae).
[0036] Compound I has been found to have significant fungicidal effects on phytopathogenic fungi of agriculturally useful vegetable crops. These diseases include Alternaria brassicicola, which causes black leaf spot of cabbage; Alternaria solani, which causes tomato early blight; Sclerotinia sclerotiorum, which causes Sclerotinia rot of lettuce; Colletotrichum capsici, which causes anthracnose of hot pepper; Erysiphe cichoracearum, which causes powdery mildew of cucumber; Mycovellosiella fulva, which causes leaf mold of tomato; Stagonosporopsis cucurbitacearum, which causes gummy stem blight of watermelon; and Botrytis cinerea, which causes broad bean grey mold, particularly for agricultural use. Compound I is particularly effective for use with agricultural crops and horticultural plants.
[0037] Compound I has a broad range of efficacy as a fungicide. The exact amount of the active material to be applied is dependent not only on the specific active material being applied, but also on the particular action desired, the fungal species to be controlled, and the stage of growth thereof, as well as the part of the plant or other product to be contacted with the compound. Thus, compound I, and formulations containing the same, may not be equally effective at similar concentrations or against the same fungal species.
[0038] Compound I is effective in use with plants in a disease-inhibiting and phytologically acceptable amount. The term "disease-inhibiting and phytologically acceptable amount" refers to an amount of a compound that kills or inhibits the plant disease for which control is desired, but is not significantly toxic to the plant. This amount will generally be from about 0.1 to about 1000 ppm (parts per million), with 1 to 500 ppm being preferred. The exact concentration of compound required varies with the fungal disease to be controlled, the type of formulation employed, the method of application, the particular plant species, climate conditions, and the like. A suitable application rate is typically in the range from about 0.10 to about 4 pounds/acre (about 0.01 to 0.45 grams per square meter, g/m.sup.2).
[0039] Any range or desired value given herein may be extended or altered without losing the effects sought, as is apparent to the skilled person for an understanding of the teachings herein.
Examples
##STR00003##
[0040] Field assessment of Alternaria brassicicola (ALTEBI) on cabbage:
[0041] A fungicidal treatment containing Compound I, applied in a 5% EC formulation and tank mixed with an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed three times on head cabbage plants (BRSOL) with the first application at 10-12 LF stage of cabbage. The following applications were done at 7 day intervals will all applications being sprayed at rates of 50, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with black leaf spot pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with compound I being applied at water volume of 800 L/ha.
[0042] Disease severity was assessed as the percent diseased area of cabbage bottom foliage (6 plants per plot randomly). Visual infection was assessed three times during the trial at 7 days after each application. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
Field Assessment of Sclerotinia sclerotiorum (SCLESC) on Lettuce:
[0043] A fungicidal treatment containing a 5% EC formulation of compound I plus an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed on head lettuce plants (LACSC) twice, the first application at the heading stage 36 days after planting, and the second application 7 days later. Formulations of compound I were applied at rates of 50, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with sclerotinia rot pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with formulations of compound I being applied at water volume of 800 L/ha.
[0044] Disease infection percentage was calculated by the equation (stem lesion lengths)/(total stem lengths).times.100% per plant (6 plants per plot randomly). Sclerotinia rot infection was assessed four times, 7 days after application A (DAAA) plus 7, 14 and 21 DAAB. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded visual infection data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
Field Assessment of Colletotrichum capsici (COLLCA) on Hot Peppers:
[0045] Assessment of compound I of COLLCA on hot peppers, in both protectant and curative fashion, was performed in two separate field trials. For the 2 day protectant test, a fungicidal treatment containing a 5% EC formulation of compound I plus an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed on hot pepper plants (CPSAN) three times, the first application at the flowering and fruiting stage 43 days after planting, with the following applications in 7 day intervals. Formulations of compound I were applied at rates of 50, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the anthracnose pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with formulations of compound I being applied at water volume of 1000 L/ha.
[0046] Disease severity was assessed as the percent fruit area diseased per plant (6 plants per plot randomly). Anthracnose infection was assessed four times, 7 days after application A (DAAA), 7 DAAB, plus 7 and 14 DAAC. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded visual infection data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
[0047] For the 4 day curative trial, a fungicidal treatment containing a 5% EC formulation of compound I plus an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed on hot pepper plants (CPSAN) twice, the first application at the flowering and fruiting stage 59 days after planting, with the second application after 7 days. Formulations of compound I were applied at rates of 50, 100, 150 and 200 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the anthracnose pathogen 4 days before the first application (curative). The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with formulations of compound I being applied at water volume of 1200 L/ha.
[0048] Disease severity was assessed as the percent fruit area diseased per plant (6 plants per plot randomly). Anthracnose infection was assessed four times, 7 days after application A (DAAA), plus 7, 14 and 21 DAAB. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded visual infection data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
Field Assessment of Erysiphe Cichoracearum (ERYSCI) on Cucumber:
[0049] Assessment of compound I of ERYSCI on cucumbers, in both protectant and curative fashion, was performed in two separate field trials. For the two day protectant test (2DP), a fungicidal treatment containing a 5% EC formulation of compound I plus an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed on cucumber plants (CUMSA) twice, the first application at the fruiting stage 41 days after planting, with the following application after 7 days. Formulations of compound I were applied at rates of 50, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the powdery mildew pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with formulations of compound I being applied at water volume of 1200 L/ha based on seedling sizes.
[0050] Disease severity was assessed as percent diseased area of cucumber foliage per plant (6 plants per plot randomly). Cucumber powdery mildew infection was assessed three times, 7 days after application A (DAAA), and 7 and 14 DAAB. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded visual infection data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
[0051] In the curative trial, a fungicidal treatment containing a 5% EC formulation of compound I plus an adjuvant (Trycol, 50% w/w at 0.2% v/v), was sprayed on cucumber plants (CUMSA) twice, the first application at the fruiting stage 29 days after planting, with the second application after 7 days. Formulations of compound I were applied at rates of 50, 100 and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the powdery mildew pathogen 2 days before the first application (curative). The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.1 m, with formulations of compound I being applied at water volume of 1200 L/ha based on seedling sizes.
[0052] Disease severity was assessed as percent diseased area of cucumber foliage per plant (6 plants per plot randomly). Powdery mildew disease severity was assessed four times, 7 days after application A (DAAA), plus 7, 14 and 21 DAAB. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded visual infection data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 1.
Field Assessment of Alternaria solani (ALTESO) on Tomato:
[0053] Assessment of compound I of ALTESO on tomato was performed in two separate field trials. In the first trial, a fungicidal treatment containing Compound I, applied in both a 5% EC and 10% SC formulation and tank mixed with an adjuvant (Agnique BP420, 50% w/w at 0.3% v/v), was sprayed on tomato plants (LYPES) at approximately 60-70 cm in height at rates of 100 and 200 grams of active ingredient per hectare (g ai/ha). The trial was based on five foliar applications at 7 day intervals with inoculation with the early blight pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 1.5.times.4 m. Compound I was applied at water volume of 1000 L/ha with an AZO backpack sprayer using compressed air.
[0054] Disease severity was recorded as percent diseased area on a random selection of 20 leaves and 20 fruits per plot. Percent control was assessed with 6 evaluations made between 0-35 days after the first application, DAA1. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 2.
[0055] In a replicated trial, a fungicidal treatment containing Compound I, applied in both a 5% EC and 10% SC formulation and tank mixed with an adjuvant (Agnique BP420, 50% w/w at 0.3% v/v), was sprayed on tomato plants (LYPES, Taylor variety) at approximately 21 days after planting. Formulations of compound I was applied at rates of 100 and 200 grams of active ingredient per hectare (g ai/ha). The trial was based on six foliar applications at 10 day intervals under natural disease pressure of tomato early blight. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.times.4 m. Formulations of compound I were applied at water volume of 800 L/ha, using a backpack plot sprayer (BKPCKENG, Solo 443; HCSOLID--Albutz ATR80 Orange Nozzle) and pressurized at 300 kPa. Disease severity (percent control) was recorded as percent visual leaf infection per plot and was assessed at 10 days after the last application. Results are given in Table 2.
Field assessment of Mycovellosiella fulva (FULVFU) on tomato:
[0056] A 10% SC formulation of compound I was tanked mixed with four different adjuvants: Agnique BP420 (50% w/w at 0.3% v/v); Trycol (50% w/w at 0.1% v/v); Ethomeen T18H (50% w/w at 0.2% v/v); and Phase II (50% w/w at 0.2% v/v). A fungicidal treatment containing formulations of compound I, either alone or with adjuvants, was sprayed on tomato plants four times, the first application approximately 3 months after planting, with the following three applications in 7-10 day intervals. Formulations of compound I were applied at rates of 50, 100, 150 and 200 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the leaf mold pathogen 7 days before the first application (curative). The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.5.times.1.4 m. Formulations of compound I were applied at water volume of 675 L/ha.
[0057] Disease severity was recorded as a percentage of visual diseased foliage (6 random plants per plot). Tomato leaf mold infection was assessed five times at 7 days after the first application (7 DAAA), 7 DAAB, 5 DAAC, followed by 7 and 12 DAAD. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 3.
Field Assessment of Stagonosporopsis Cucurbitacearum (DIDYBR) on Watermelon:
[0058] A 10% SC formulation of compound I was tanked mixed with four different adjuvants: Agnique BP420 (50% w/w at 0.3% v/v); Trycol (50% w/w at 0.1% v/v); Ethomeen T18H (50% w/w at 0.2% v/v); and Phase II (50% w/w at 0.2% v/v). A fungicidal treatment containing formulations of compound I, either alone or with adjuvants, was sprayed on watermelon plants four times, the first application approximately 2 months after planting, with the following three applications in 7-10 day intervals. Formulations of compound I were applied at rates of 50, 100, 150 and 200 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the gummy stem blight pathogen 2 days after the first application. The treatment was part of an experimental trial designed as a randomized complete block with four replications and a plot of approximately 2.5.times.1.4 m. Formulations of compound I were applied at water volume of 675 L/ha.
[0059] Disease severity was recorded as the length of stem lesion and as percent area diseased foliage (3 random plants per plot). Watermelon gummy stem blight infection was assessed twice, the first at 6 days after the first application (6 DAAA), followed by 19 DAAD. Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the nontreated control. Results are given in Table 4.
Greenhouse Assessment of Botrytis cinerea (BOTRCI) on Broad Bean:
[0060] Technical grades of material were dissolved in acetone, which were then mixed with nine volumes of water containing 100 ppm of Triton X-100. The fungicide formulations were applied onto seedling plants using an automated booth sprayer to run off. All sprayed plants were allowed to dry prior to further handling. Test plants were inoculated with Botrytis cinerea 1-day post application. When disease symptoms were fully expressed on the untreated plants, percent diseased area of the plant was assessed on a scale of 0 to 100 percent disease severity. Percent disease control was calculated using the ratio of disease severity on treated plants relative to untreated plants. Results are given in Table 5.
[0061] In each case of Table 1-5 the rating scale of percent control based on AUDPC is as follows:
TABLE-US-00001 % Control Rating 76-100 A 51-75 B 26-50 C 1-25 D Not tested E
TABLE-US-00002 TABLE 1 Percent Control by Compound I.sup.a on Fungal Diseases of Vegetables Based on Area Under Disease Progression Curve (AUDPC) in Protectant and Curative Tests. Rate ALTEBI.sup.b SCLESC.sup.c COLLCA.sup.d ERYSCI.sup.e (g ai/ha).sup.f 2 DP.sup.g 2 DP 2 DP 4 DC.sup.h 2 DP 2 DC 50 C B A C A A 100 B A A B A B 150 B A A B A C 200 E E E B E E .sup.aCompound I applied as a 5% EC formulation with Trycol adjuvant (50% w/w at 0.2% v/v) .sup.bBlack leaf spot of cabbage-Alternaria brassicicola .sup.cLettuce Sclerotinia rot-Sclerotinia sclerotiorum .sup.dHot pepper anthracnose-Colletotrichum capsici .sup.eCucumber powdery mildew-Erysiphe cichoracearum .sup.fGrams of active ingredient per hectare .sup.g2 Day protectant .sup.h4 Day curative
TABLE-US-00003 TABLE 2 Efficacy of Compound I.sup.a against Early Blight (ALTESO, Alternariasolani) on Leaves and Fruits of Tomato in EC and SC Formulations. Leaves.sup.b Fruits.sup.c Leaves.sup.b Rate Trial 1 Trial 1 Trial 2 (g ai/ha).sup.d 5% EC 10% SC 5% EC 10% SC 5% EC 10% SC 100 A A A A A A 200 A A A A A A .sup.aCompound I applied with Agnique BP420 (50% w/w at 0.3% v/v) as an adjuvant .sup.bPercent disease control on tomato leaves based on Area Under Disease Progression Curve (AUDPC) .sup.cPercent disease control on tomato fruits based on Area Under Disease Progression Curve (AUDPC) .sup.dGrams of active ingredient per hectare
TABLE-US-00004 TABLE 3 Efficacy.sup.a of Compound I against Tomato Leaf Mold (FULVFU, Mycovellosiella fulva) in a 10% SC Formulation with or without Adjuvants. Rate Adjuvant (g ai/ha).sup.b I.sup.c II.sup.d III.sup.e IV.sup.f V.sup.g 50 C A NT NT NT 100 C A A B A 150 NT A NT NT NT 200 NT A NT NT NT .sup.aPercent control based on Area Under Disease Progression Curve (AUDPC) .sup.bGrams of active ingredient per hectare .sup.cI-No adjuvant .sup.dII-Agnique BP420 (50% w/w at 0.3% v/v) .sup.eIII-Trycol (50% w/w at 0.1% v/v) .sup.fIV-Ethomeen T18H (50% w/w at 0.2% v/v) .sup.gV-Phase II (50% w/w at 0.2% v/v)
TABLE-US-00005 TABLE 4 Efficacy.sup.a of Compound I against Watermelon Gummy Stem Blight (DIDYBR, Stagonosporopsis cucurbitacearum) in a 10% SC Formulation with or without Adjuvants. Rate Adjuvant (g ai/ha).sup.b I.sup.c II.sup.d III.sup.e IV.sup.f V.sup.g 50 NT A NT NT NT 100 B A B B B 150 NT A NT NT NT 200 NT A NT NT NT .sup.aPercent control based on Area Under Disease Progression Curve (AUDPC) .sup.bGrams of active ingredient per hectare .sup.cI--No adjuvant .sup.dII--Agnique BP420 (50% w/w at 0.3% v/v) .sup.eIII--Trycol (50% w/w at 0.1% v/v) .sup.fIV--Ethomeen T18H (50% w/w at 0.2% v/v) .sup.gV--Phase II (50% w/w at 0.2% v/v)
TABLE-US-00006 TABLE 5 Efficacy of Compound I against Broad Bean Gray Mold (BOTRCI, Botrytis cinerea). Rate (ppm).sup.a % Control.sup.b 400 A 100 A 25 A 6.25 B .sup.aParts per million .sup.bPercent control calculated using the ratio of disease severity on treated plants relative to untreated plants
Field Assessment of Corynespora cassiicola (CORYCA) in Tomato:
[0062] A fungicidal treatment containing Compound I, applied in an SC formulation (MSO built-in) and tank mixed with an adjuvant (Agnique BP-420, 50% w/w at 0.2% v/v or Adsee C80W 80%), was sprayed on tomato plants (LYPES, Charger variety) at growth stage BBCH64 (4.sup.th flower open) at rates of 50, 75, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were inoculated with the target spot pathogen one day after initial application. The treatment was part of an experimental trial designed as a randomized complete block (RCB) with four replications and a plot of approximately 6.times.25 feet (ft). Compound I was applied at water volume of 100 gallons per acre (gal/acre), using a tractor sprayer (SCDISC D1 Nozzles) and pressurized at 200 psi.
[0063] Disease severity (percentage of visual diseased foliage (leaf) on whole plot) was assessed four times during the trial (1-20 days after application, DAA). Area under the disease progress curve (AUDPC) was calculated for each plot using the sets of recorded severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the untreated control. Results are given in Table 6.
Field Assessment of Colletotrichum Coccodes (COLLCA) in Tomato:
[0064] A fungicidal treatment containing Compound I, applied in an SC formulation (MSO built-in) and tank mixed with an adjuvant (Agnique BP-420, 50% w/w at 0.2% v/v or Adsee C80W 80%), was sprayed on tomato plants (LYPES, Roma VS Saladette type variety) at growth stage 12 in vegetative growth at rates of 50, 75, 100, and 150 grams of active ingredient per hectare (g ai/ha). The experimental plots were run with natural infestation of the anthracnose pathogen and supplemented with additional inoculum about one day after the 1.sup.st application. The treatment was part of an experimental trial designed as a randomized complete block (RCB) with four replications and a plot of approximately 6.67.times.30 ft. Compound I was applied at water volume of 40 gal/acre, using a backpack sprayer (carbon dioxide (CO.sub.2), 8004VS FF nozzles) and pressurized at 38 psi.
[0065] Percent leaf severity was assessed three times during the trial (0-23 days after application 1). Area under the disease progress curve (AUDPC) was calculated for each plot using the recorded leaf severity data. Relative AUDPC (% control based on AUDPC) was calculated as percent of the untreated control. Results are given in Table 7.
TABLE-US-00007 TABLE 6 Formulation Comparison of Compound I with MSO or Tank-Mixed with Agnique BP-420 of Calculated Percent Control of Target Spot of Tomato (CORYCA, Corynespora cassiicola) Percent Control Compound of CORYCA I.sup.a Adjuvant.sup.a or b (AUDPC) 50 MSO, 100.sup.a 55.2 75 MSO, 150.sup.a 56.1 100 MSO, 200.sup.a 71.6 150 MSO, 300.sup.a 84.0 50 Agnique BP420, 200.sup.b 53.1 75 Agnique BP420, 300.sup.b 56.9 100 Agnique BP420, 400.sup.b 74.1 150 Agnique BP420, 600.sup.b 83.9 100 Adsee C80W 80%, 250.sup.a 61.4 .sup.aRate in g/ha .sup.bRate in mL/ha
TABLE-US-00008 TABLE 7 Formulation Comparison of Compound I with MSO or Tank- Mixed with Agnique BP-420 of Calculated Percent Control of Anthracnose of Tomato (COLLCA, Colletotrichum coccodes) Percent Control Compound of COLLCA I.sup.a Adjuvant.sup.a or b (AUDPC) 50 MSO, 100.sup.a 46.0 75 MSO, 150.sup.a 75.5 100 MSO, 200.sup.a 92.6 150 MSO, 300.sup.a 95.3 50 Agnique BP420, 200.sup.b 67 75 Agnique BP420, 2300.sup.b 91.1 100 Agnique BP420, 400.sup.b 91.0 150 Agnique BP420, 600.sup.b 95.7 100 Adsee C80W 80%, 250.sup.a 57.3 .sup.aRate in g/ha .sup.bRate in mL/ha
* * * * *





XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.