Novel Glycan Conjugates And Methods Of Use Thereof
WONG; Chi-Huey ; et al.
U.S. patent application number 16/502844 was filed with the patent office on 2020-03-12 for novel glycan conjugates and methods of use thereof. The applicant listed for this patent is ACADEMIA SINICA. Invention is credited to Chi-Huey WONG, Chung-Yi WU.
| Application Number | 20200078452 16/502844 |
| Document ID | / |
| Family ID | 56417553 |
| Filed Date | 2020-03-12 |










View All Diagrams
| United States Patent Application | 20200078452 |
| Kind Code | A1 |
| WONG; Chi-Huey ; et al. | March 12, 2020 |
NOVEL GLYCAN CONJUGATES AND METHODS OF USE THEREOF
Abstract
The present disclosure is directed to vaccines, antibodies, and/or immunogenic conjugate compositions targeting the SSEA3/SSEA4/GloboH associated epitopes (natural and modified) which elicit antibodies and/or binding fragment production useful for modulating the globo-series glycosphingolipid synthesis. The present disclosure relates to methods and compositions which can modulate the globo-series glycosphingolipid synthesis. Particularly, the present disclosure is directed to glycoenzyme inhibitor compound and compositions and methods of use thereof that can modulate the synthesis of globo-series glycosphingolipid SSEA3/SSEA4/GloboH in the biosynthetic pathway; particularly, the glycoenzyme inhibitors target the alpha-4GalT; beta-4GalNAcT-I; or beta-3GalT-V enzymes in the globo-series synthetic pathway. Moreover, the present disclosure is also directed to the method of using the compositions described herein for the treatment or detection of hyperproliferative diseases and/or conditions.
| Inventors: | WONG; Chi-Huey; (La Jolla, CA) ; WU; Chung-Yi; (TAIPEI, TW) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 56417553 | ||||||||||
| Appl. No.: | 16/502844 | ||||||||||
| Filed: | July 3, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14832993 | Aug 21, 2015 | 10342858 | ||
| 16502844 | ||||
| 62107378 | Jan 24, 2015 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07H 15/04 20130101; A61K 31/715 20130101; A61K 2039/55511 20130101; C07H 15/26 20130101; C07H 5/06 20130101; A61K 39/0011 20130101; C07K 16/3076 20130101; A61K 2039/6037 20130101; C07H 5/02 20130101; A61K 39/001169 20180801; A61K 31/7028 20130101; C08B 37/006 20130101; C07K 16/44 20130101; C07H 5/04 20130101; A61K 2039/627 20130101; A61K 39/001173 20180801 |
| International Class: | A61K 39/00 20060101 A61K039/00; C07K 16/30 20060101 C07K016/30; C07H 15/04 20060101 C07H015/04; C08B 37/00 20060101 C08B037/00; C07H 5/02 20060101 C07H005/02; C07H 5/06 20060101 C07H005/06; A61K 31/715 20060101 A61K031/715; C07H 5/04 20060101 C07H005/04; A61K 31/7028 20060101 A61K031/7028; C07H 15/26 20060101 C07H015/26; C07K 16/44 20060101 C07K016/44 |
Claims
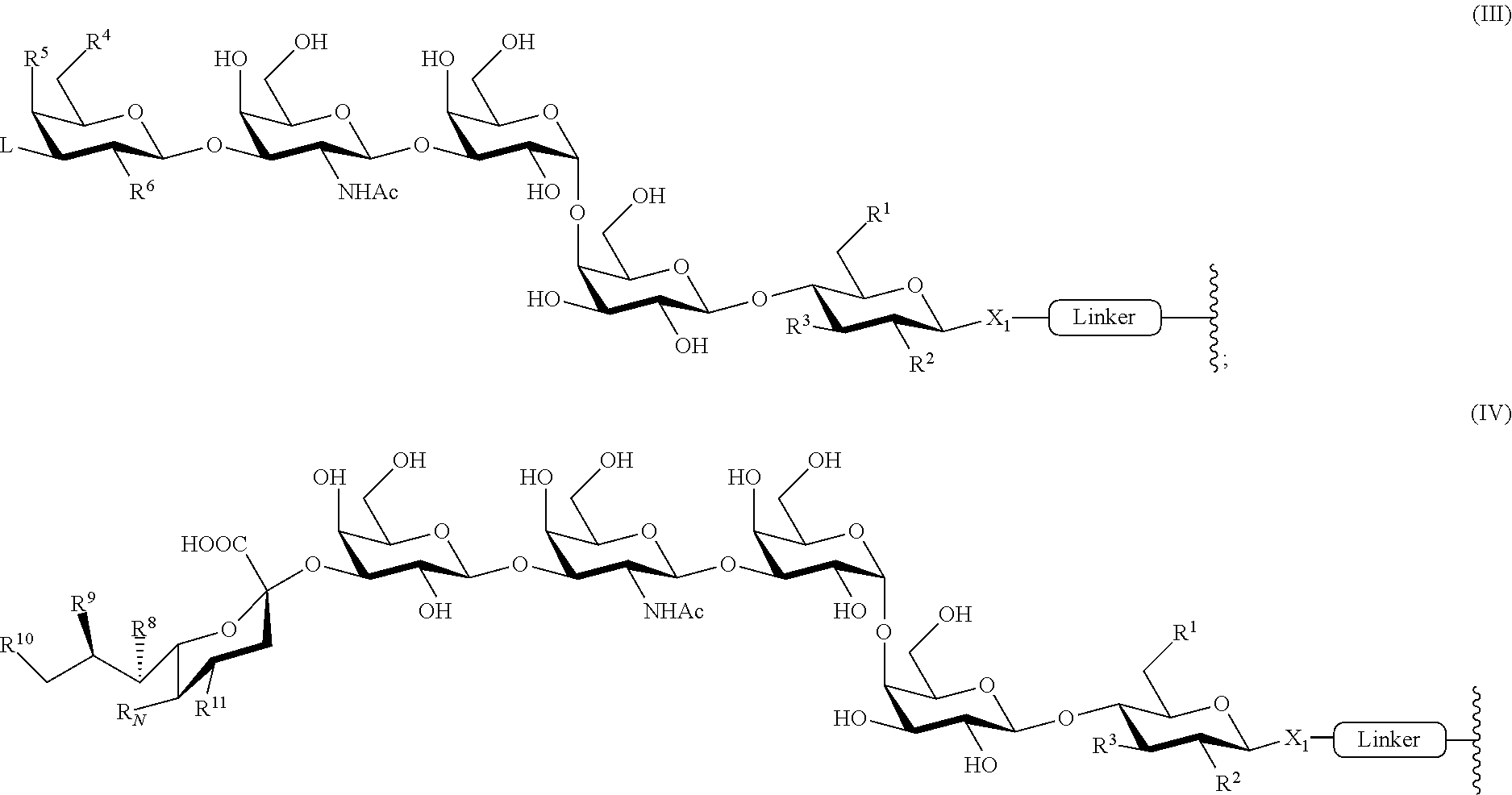
1. An immunogenic composition comprising: (a) a glycan conjugate including a carrier and one or more glycans, and optionally (b) an adjuvant; wherein each of the one or more glycans is conjugated with the carrier through a linker having the formula (III): ##STR00026## wherein: X.sub.1 is --OR or --SR, wherein R is an oxygen or sulfur protecting group, optionally substituted C.sub.1-10 alkyl, optionally substituted aryl, optionally substituted acyl, or optionally substituted imidoyl; each instance of R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6 and L is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --CR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; each instance of R.sup.A is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; each instance of R.sup.B is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; and provided the glycan conjugate is not of the formula (III-a) and (III-b): ##STR00027##
2. The immunogenic composition of claim 1, wherein L is --OH.
3. The immunogenic composition of claim 2, wherein at least one instance of R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5 and R.sup.6 is --N.sub.3.
4. The immunogenic composition of claim 2, wherein at least one instance of R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5 and R.sup.6 is --F.
5. The immunogenic composition of claim 1, wherein L is of the formula: ##STR00028## wherein: each instance of R.sup.8, R.sup.9, R.sup.10 and R.sup.11 is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --CR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; wherein R.sup.12 is H, OH, or halogen; R.sub.N is selected from --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --SR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; each instance of R.sup.A is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; each instance of R.sup.B is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl.
6. The immunogenic composition of claim 5, wherein the glycan conjugate is of formula (IV): ##STR00029##
7. The immunogenic composition of claim 6, wherein at least one instance of R.sup.1, R.sup.2, R.sup.3, R.sup.8, R.sup.9, R.sup.10, and R.sup.11 is --N.sub.3.
8. The immunogenic composition of claim 6, wherein at least one instance of R.sup.2, R.sup.3, R.sup.8, R.sup.9, R.sup.10, and R.sup.11 is --F.
9. The immunogenic composition of claim 1, wherein the carrier is a protein, a lipid, a lipolized protein, a virus, a peptide, or a dendrimer of glycopeptides.
10. The immunogenic composition of claim 9, wherein the carrier is a protein selected from the group consisting of tetanus toxoid (TT), diphtheria toxoid (DT), diphtheria toxin cross-reacting material .sub.197 (CRM.sub.197), fragment C of TT, Keyhole limpet hemocyanin (KLH), bovine serum albumin (BSA), protein D, outer-membrane protein (OMP) and pneumolysin.
11. The immunogenic composition of claim 10, wherein the carrier protein is selected from the group consisting of TT, DT and CRM.sub.197.
12. The immunogenic composition of claim 11, wherein the glycan conjugate is of the formula (IV-a) or (IV-b): ##STR00030## wherein m is an integer of 1 to 40, inclusive.
13. The immunogenic composition of claim 1, wherein the linker is a hetero- or homo-bifunctional linker.
14. The immunogenic composition of claim 1, wherein the adjuvant is a glycolipid capable of binding a CD1d molecule on a dendritic cell.
15. The immunogenic composition of claim 1, wherein the adjuvant is C34, 7DW8-5, C17, C23, Gluco-C34, Aluminum salt, Squalene, MF59, or QS-21.
16. The immunogenic composition of claim 1, wherein the immunogenic composition is capable of eliciting an immune response against a cancer cell.
17. The immunogenic composition of claim 16, wherein the cancer cell is selected from the group consisting of a brain cancer cell, a lung cancer cell, a breast cancer cell, an oral cancer cell, an esophagus cancer cell, a stomach cancer cell, a liver cancer cell, a bile duct cancer cell, a pancreatic cancer cell, a colon cancer cell, a kidney cancer cell, a bone cancer cell, a skin cancer cell, a cervical cancer cell, an ovarian cancer cell, and a prostate cancer cell.
18. The immunogenic composition of claim 16, wherein the immune response includes generation of antibodies that specifically bind to one or more of the antigens selected from the group consisting of SSEA3 and SSEA4.
19. The immunogenic composition of claim 18, wherein the antibodies neutralize one or more of SSEA3 and SSEA4 antigen expressed on the surface of cancer cells or cancer stem cells.
20. The immunogenic composition of claim 18, wherein the antibodies predominantly includes IgG antibodies.
21. A cancer vaccine, comprising a therapeutically effective amount of the immunogenic composition of claim 1 and a pharmaceutically acceptable excipient.
22. The cancer vaccine of claim 21, wherein the cancer vaccine is able to induce an anti-cancer immune response in a subject.
23. A method of treating cancer in a subject in need thereof wherein the method comprising administering a therapeutically effective amount of the immunogenic composition of claim 1.
24. The method of claim 23 wherein the vaccine is co-administered in combination with another therapeutic agent.
25. The method of claim 23, wherein the cancer is selected from the group consisting of brain cancer, lung cancer, breast cancer, oral cancer, esophageal cancer, stomach cancer, liver cancer, bile duct cancer, pancreatic cancer, colon cancer, kidney cancer, bone cancer, skin cancer, cervical cancer, ovarian cancer, and prostate cancer.
26. The method of claim 25, wherein the cancer cell expresses SSEA3 and/or SSEA4 antigen on the surface of the cell.
27. The method of claim 26, wherein the subject is a human.
28. The method of claim 23, wherein the immunogenic composition or the cancer vaccine is administered subcutaneously.
29. An isolated monoclonal antibody and/or a binding fragment thereof raised against the immunogenic composition of claim 1.
30. A composition comprising an effective amount of the antibody or antigen-binding fragment of claim 29 and a pharmaceutically acceptable carrier.
31. A method for making the immunogenic composition of claim 1.
32. The method of claim 31 wherein the method comprising: providing a carrier; conjugating one or more glycan to the carrier by conjugation reaction; wherein each of the one or more glycan is SSEA3 or SSEA4.
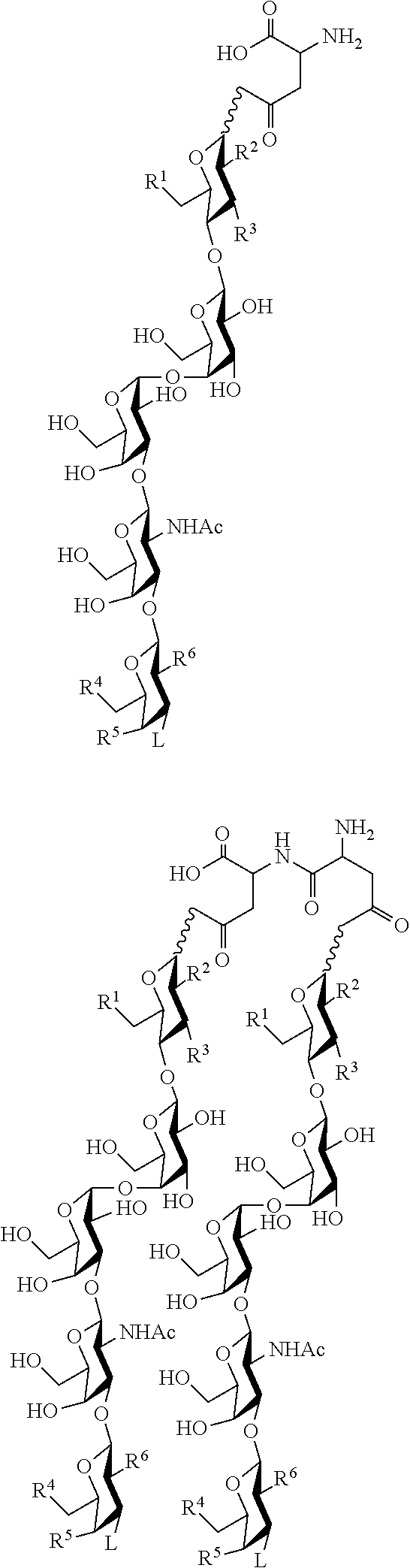
33. An immunogenic composition comprising multivalent construct targeting one or more of SSEA4 and SSEA3 and their analogs thereof wherein the glycans are linked to a template and a carrier, ##STR00031## wherein n can be an integer from 1 to 10; wherein glycan can be selected from the group consisting of Formulas I, II, III, and IV; wherein if n is 2 or more, each glycan can be the same as another glycan on the aspartyl peptide or a difference glycan on the aspartyl peptide.
34. The composition of claim 33 wherein the glycan is selected from the group consisting of SSEA3 and SSEA4.
35. The composition of claim 33 wherein the multivalent construct has the structure: ##STR00032## ##STR00033## wherein R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, and L on each glycan moiety can be the same or different.
36. A compound having the formula (I): ##STR00034## or a salt thereof, wherein: X.sub.1 is --OR or --SR, wherein R is hydrogen, a oxygen or sulfur protecting group, optionally substituted C.sub.1-10 alkyl, optionally substituted aryl, optionally substituted acyl, or optionally substituted imidoyl; each instance of R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6 and L is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --SR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; each instance of R.sup.A is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; each instance of R.sup.B is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; and provided the compound is not of the formula (I-a) or (I-b): ##STR00035##
37. The compound of claim 35, wherein L is --OH.
38. The compound of claim 35, wherein L is of the formula: ##STR00036## wherein: each instance of R.sup.8, R.sup.9, R.sup.10 and R.sup.11 is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --SR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; wherein R.sup.12 is H, OH, or halogen; R.sub.N is selected from --N.sub.3, --NO.sub.2, --N(R.sup.B).sub.2, --N(R.sup.A)C(O)R.sup.A, --OR.sup.A, --OC(O)R.sup.A, --SR.sup.A, --C(O)N(R.sup.B).sub.2, --CN, --C(O)R.sup.A, --C(O)OR.sup.A, --S(O)R.sup.A, --SO.sub.2R.sup.A, --SO.sub.2N(R.sup.B).sub.2, and --NHSO.sub.2R.sup.B; each instance of R.sup.A is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; and each instance of R.sup.B is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl.
39. The compound of claim 35, wherein the compound is of Formula (II): ##STR00037##
40. The compound of claim 39, wherein at least one instance of R.sup.1, R.sup.2, R.sup.3, R.sup.8, R.sup.9, R.sup.10 and R.sup.11 is --F.
41. The compound of claim 39, wherein at least one instance of R.sup.1, R.sup.2, R.sup.3, R.sup.8, R.sup.9, R.sup.10 and R.sup.11 is --N.sub.3.
42. The method of treating hyperproliferative disease or condition comprising the administering to a subject in need thereof a therapeutically effective amount of the compound of claim 36.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. application Ser. No. 14/832,993, filed Aug. 21, 2015, which claims the benefit under 35 U.S.C. 119(e) of U.S. Provisional Patent Application No. 62/107,378, filed on Jan. 24, 2015, entitled NOVEL GLYCAN CONJUGATES AND METHODS OF USE THEREOF, the contents of which is hereby incorporated by reference as if set forth in its entirety.
FIELD
[0002] The present disclosure relates to methods and compositions which can modulate the globo-series glycosphingolipid synthesis. Particularly, the present disclosure is directed to glycoenzyme inhibitor compound and compositions and methods of use thereof that can modulate the synthesis of globo-series glycosphingolipid SSEA3/SSEA4/GloboH in the biosynthetic pathway; particularly, the glycoenzyme inhibitors target the alpha-4GalT; beta-4GalNAcT-I; or beta-3GalT-V enzymes in the globo-series synthetic pathway. Additionally, the present disclosure is also directed to vaccines, antibodies, and/or immunogenic conjugate compositions targeting the SSEA3/SSEA4/GloboH associated epitopes (natural and modified) which can elicit antibodies and/or binding fragment production useful for modulating the globo-series glycosphingolipid synthesis. Moreover, the present disclosure is also directed to the method of using the compositions described herein for the treatment or detection of hyperproliferative diseases and/or conditions.
BACKGROUND OF THE INVENTION
[0003] The carbohydrate antigens GloboH, stage-specific embryonic antigen-3 (SSEA3), and stage-specific embryonic antigen-4 (SSEA4) are closely related to one another in either structure or in function. GloboH, SSEA3 and SSEA4 are globo-series glycosphingolipids, with SSEA3 being the non-fucosylated pentasaccharide precursor structure of GloboH, SSEA4 is sialylated SSEA3 with sialic acid a2-3 links to the non-reducing end of galactose of SSEA3.
[0004] Stage-specific embryonic antigen-3 (SSEA3) was first identified and defined by the reactivity of an IgM monoclonal antibody generated in a rat immunized with 4- to 8-cell stage mouse embryos. This monoclonal antibody reacted with all mouse preimplantation embryos from oocytes up to the early blastocyst stage where its expression became more restricted, in the primitive endoderm after implantation. The SSEA3 antigenic determinant was determined to be a carbohydrate present on glycolipids and glycoproteins; it was also found on human teratocarcinoma cells and human erythrocytes. In a panel of structures isolated from the 2102Ep human teratocarcinoma cell line, the SSEA3 antibody had the highest affinity for Gal.beta.(1-3)GalNAc .beta.(1-3)Gal.alpha.(1-4)Gal.beta.(1-4)Glc.beta.(1)Cer. This structure is also known as Gb5, galactosyl-globoside, or globopentaosylceramide.
[0005] Synthesis of SSEA3 occurs when 31,3-.sub.galactosyltransferase V (.beta.3GalT-V) transfers galactose to the GalNAc of globoside to form Gb5 or galactosyl-globoside. It was determined that SSEA3 was not expressed in hematopoietic or mesenchymal stem cells. Based on immortalized lymph node lymphocytes from primary lung cancer patients, generated hybridomas, and selected for antibody secreting clones; monoclonal antibodies were then generated from two of these clones--J309 and D579, which recognized the SSEA3 antigenic determinant. The antibodies recognized SSEA3 on several tumor cell lines including lung and breast cancer cell lines, and a teratocarcinoma cell line; in an immune adherence assay, rodent monoclonal SSEA3 antibody, also referred to as MC631, reacted against the same cell lines as the J309 and D579 antibodies. SSEA3 has also been found on testicular germ cell tumors, as well as in breast cancer and in BCSCs (breast cancer stem cells).
[0006] Chang et al. looked at SSEA3 expression on normal tissues using a tissue microarray because its location outside of cancer and development was largely unknown. The group found SSEA3 to be expressed on normal epithelium of colon, esophagus, small intestine, kidney, prostate, rectum, skin, testis, thymus, and uterine cervix. Expression was located only on the apical surfaces of epithelial cells or in the cytoplasm, which are considered immune system restricted or inaccessible sites. In an experiment using a KLH conjugated GloboH monovalent vaccine in mice, an antibody response was made to only the GloboH antigen. When .alpha.-GalCer was added as an adjuvant, the amount of overall antibody production increased and the mice made polyclonal antibodies to both the GloboH, the SSEA3 and the SSEA4 antigen structures, which vaccination was unable to generate in the absence of the adjuvant. This result showed that SSEA3, GloboH and SSEA4 could make promising targets for cancer vaccines and could be targeted simultaneously.
[0007] However, most tumor associated carbohydrate antigens have poor immunogenicity and many approaches have been developed to increase the immune response of carbohydrate-based vaccines, including conjugation with a carrier protein,administration with an immunologic adjuvant using unnatural glycosidic linkage, clustered antigens, unimolecular polyvalent vaccine or hetero-glycan multivalent vaccine. Using these strategies, a few carbohydrate-based vaccines that could elicit significant immune responses to target glycan structures were designed for cancer therapy and entered clinical trials. Among them, the clinical trials of Theratope and GMK with adjuvant QS-21 failed to produce statistically significant difference between time-to-disease and overall survival rate. Mot likely these two vaccines could not elicit robust T cell-dependent immune response in patients. Specifically, Theratope and GMK induced a higher level of IgM in patients but could not induce a strong immune IgG response, which is a major problem in carbohydrate-based vaccine development.
[0008] Previous studies showed that modification of carbohydrate antigen structures (MCAS) could effectively elicit a higher level of immune response. For example, in the modification study of the capsular polysaccharide of group B meningococci, the N-acetyl groups of .alpha.-(2,8)-linked polysialic acid (PSA) was replaced with the N-propinoyl group and such a modification elicited a high antibody response to recognize not only the N-propinoyl PSA, but also the nature N-acetyl PSA. Similar approaches were applied to STn and GM3 antigens to produce high antibody titers against modified and nature forms. The results indicated that N-phenylacetyl, N-fluoroacetyl or N-difluoroacetyl modifications on glycan antigens could improve the immunogenicity. Moreover, the Schultz group reported that incorporation of a p-nitrophenylalanine into the tumor necrosis factor-.alpha. (TNF-.alpha.) could break immune tolerance and induce more antibody response to TNF-.alpha.. Using glycans as antigens, although some progress has been achieved, most cases are the N-modification of disaccharide (STn), trisaccharide (GM3) and polysialic acid (PSA) and some are based on fluorinated MUC1 glycopeptide antigens.
SUMMARY OF THE INVENTION
[0009] The present disclosure is based on the discovery that the modification of the stage-specific embryonic antigens (SSEA3 and SSEA4) with certain groups disclosed herein elicited robust IgG antibody response to specifically recognize SSEA3 and SSEA4, respectively. The antibodies induced by an immunogenic composition comprising such unnatural glycan moiety are able to mediate the complement-dependent cell cytotoxicity against tumor cells.
[0010] Accordingly, the present invention features the design of antibodies against SSEA3 and/or SSEA4 for treating cancers. The present invention also features novel compounds consisting of the modified carbohydrate antigens (SSEA3 and SSEA4), glycan conjugates comprising such, and immunogenic compositions and vaccines thereof.
[0011] The present disclosure also provides methods of using synthetic glycan conjugates described herein to treat or reduce hyperproliferative disease such as cancer.
[0012] Additionally, the present disclosure is also directed to vaccines and/or immunogenic conjugate compositions targeting the SSEA3/SSEA4/GloboH associated epitopes (natural and modified) which can elicit antibodies and/or binding fragment production useful for modulating the globo-series glycosphingolipid synthesis. Moreover, the present disclosure is also directed to the method of using the compositions described herein for the treatment or detection of hyperproliferative diseases and/or conditions.
[0013] Accordingly, the present invention features the design of antibodies against SSEA3 for treating cancers. The present invention also features novel compounds consisting of the modified carbohydrate antigens (SSEA3, SSEA4), glycan conjugates comprising such, and immunogenic compositions and vaccines thereof
[0014] In one aspect, the present invention provides a compound of formula (I):
##STR00001##
or a salt thereof, wherein X.sub.1, R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6 and L are as described herein. In certain embodiments, a compound of Formula (I) is useful for making an immunogenic composition for treating cancers.
[0015] In another aspect, the present invention provides a compound of Formula (II):
##STR00002##
[0016] or a salt thereof, wherein X.sub.1, R.sup.1, R.sup.2, R.sup.3, R.sup.8, R.sup.9, R.sup.10, R.sup.11, and R.sub.N are as described herein. In certain embodiments, a compound of Formula (II) is useful for making an immunogenic composition for treating cancers.
[0017] In another aspect, the present invention provides an immunogenic composition, comprising (a) a glycan conjugate including a carrier and one or more glycans, and optionally (b) an adjuvant,
[0018] wherein: each of the one or more glycans is conjugated with the carrier through a linker, having the formula (III) or (IV):
##STR00003##
[0019] wherein X.sub.1, R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.8, R.sup.9, R.sup.10, R.sup.11, and R.sub.N are as described herein.
[0020] In certain aspects, it is contemplated that any construct of vaccine containing a combination of any one or more of the three glycans (SSEA3, SSEA4 and GloboH) and analogs thereof in any ratio can be linked to a carrier.
##STR00004##
[0021] wherein n can be an integer from 1 to 10;
[0022] wherein Glycan can be selected from the group consisting of Formulas I, II, III, and IV;
[0023] wherein if n is 2 or more, each Glycan can be the same as another Glycan on the aspartyl peptide or a difference Glycan on the aspartyl peptide.
[0024] In some embodiments, Glycan can be selected from the group consisting of SSEA3, SSEA4, and GloboH.
[0025] In some embodiments, the exemplary multivalent construct can be:
##STR00005## ##STR00006##
[0026] wherein R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, and L on each glycan moiety can be the same or different.
[0027] In certain embodiments, the immunogenic composition of the invention comprises an adjuvant. Exemplary adjuvants suitable for the invention are as described herein.
[0028] In certain embodiments, the immunogenic composition is capable of eliciting an immune response against a cancer cell in a subject. In certain embodiments, the cancer cell is selected from the group consisting of a brain cancer cell, a lung cancer cell, a breast cancer cell, an oral cancer cell, an esophageal cancer cell, a stomach cancer cell, a liver cancer cell, a bile duct cancer cell, a pancreatic cancer cell, a colon cancer cell, a kidney cancer cell, a bone cancer cell, a skin cancer cell, a cervical cancer cell, an ovarian cancer cell, and a prostate cancer cell.
[0029] In certain embodiments, the immune response includes generation of antibodies that specifically bind to one or more of the antigens selected from the group consisting of GloboH, SSEA3 and SSEA4. In certain embodiments, the antibodies are developed to target one or more of GloboH, SSEA3 and SSEA4 expressed on the surface of cancer cells or cancer stem cells , and trigger CDC and/or ADCC to kill these cells. In certain embodiments, the antibodies predominantly include IgG antibodies. In certain embodiments, the immunogenic compositions provided herein mainly induce IgG1, IgG2b, IgG2c and IgG3.
[0030] Further, the present disclosure features monoclonal antibodies and binding fragments raised against the immunogenic composition described herein.
[0031] In one embodiment, the antibody is a human antibody.
[0032] In one embodiment, the antibody is a humanized antibody.
[0033] In one embodiment, the antibody is specifically targeted against one or more of SSEA4, SSEA3, or GloboH.
[0034] In one embodiment, the antibody is specifically targeted against SSEA3.
[0035] In one embodiment, the antibody is specifically targeted against SSEA4.
[0036] In one embodiment, the antibody is a homogeneous antibody having the biantennary glycan terminated by two sialic acid in alpha-2,6-linkage.
[0037] In one aspect, the present disclosure provides a pharmaceutical composition comprising an effective amount of the antibody or antigen-binding fragment specifically targeted against one or more of SSEA4, SSEA3, or GloboH and a pharmaceutically acceptable carrier
[0038] In one embodiment, the pharmaceutical composition comprises a combination of antibodies and/or binding fragment thereof each independently targeting one or more of the SSEA4, SSEA3, and/or GloboH glycans.
[0039] In one embodiment, the pharmaceutical composition is useful for the treatment of cancer, infectious diseases, and/or anti-inflammatory diseases,
[0040] In one embodiment, the pharmaceutical composition comprises antibodies or binding fragments thereof having universal biantennary N-glycan terminated with sialic acid in alpha-2,6-linkage.
[0041] In another aspect, the present invention provides a cancer vaccine comprising an immunogenic composition described herein and a pharmaceutically acceptable excipient.
[0042] In another aspect, the present invention provides methods for treating and/or reducing the risk for cancer in a subject comprising administering to a subject in need thereof a therapeutically effective amount of an immunogenic composition or a cancer vaccine as described herein.
[0043] The treatment results in reduction of tumor size, elimination of malignant cells, prevention of metastasis, prevention of relapse, reduction or killing of disseminated cancer, prolongation of survival and/or prolongation of time to tumor cancer progression.
[0044] In some embodiments, the treatment further comprises administering an additional therapy to the subject prior to, during or subsequent to the administering of the immunogenic composition or the cancer vaccine described herein. In some embodiments, the additional therapy is treatment with a chemotherapeutic agent. In some embodiments, the additional therapy is radiation therapy.
[0045] Another aspect of the present disclosure features a method of vaccinating a mammal against cancers, comprising administering to the mammal a pharmacologically effective amount of an immunogenic composition or a cancer vaccine as described herein.
[0046] In some embodiments, the mammal is a human. In some embodiments, the immunogenic composition or the cancer vaccine described herein is administered subcutaneously.
[0047] Examples of the cancer include, but are not limited to, brain cancer, lung cancer, breast cancer, oral cancer, esophagus cancer, stomach cancer, liver cancer, bile duct cancer, pancreas cancer, colon cancer, kidney cancer, cervix cancer, ovary cancer and prostate cancer. In some embodiments, the cancer is brain cancer, lung cancer, breast cancer, ovarian cancer, prostate cancer, colon cancer, or pancreas cancer.
[0048] In another aspect, the present invention provides methods of synthesizing the compounds of the invention as described herein.
[0049] In yet another aspect, the present disclosure features the process for making an immunogenic composition or a cancer vaccine as described herein.
[0050] The details of certain embodiments of the invention are set forth herein. Other features, objects, and advantages of the invention will be apparent from the Detailed Description, the Figures, the Examples, and the Claims.
BRIEF DESCRIPTION OF THE DRAWINGS
[0051] FIG. 1: Biosynthetic Pathway of Globo Series of Glycosphingolipids.
[0052] FIG. 2: The induced GloboH-IgG collected from different epitope ratios of SSE4-CRM197 or SSEA4-Gc-CRM197 immunization.
[0053] FIG. 3A: Native SSEA4, as well as all eight SSEA4 analogs, could elicit IgG antibodies against SSEA4 when combining the use of Gal-C34.
[0054] FIG. 3B: Native SSEA4, as well as all eight SSEA4 analogs, could elicit IgM antibodies against SSEA4 when combining the use of Gal-C34.
[0055] FIG. 4A: Native SSEA4, as well as all eight SSEA4 analogs, could elicit IgG antibodies against SSEA4 when combining the use of Glc-C34.
[0056] FIG. 4B: Native SSEA4, as well as all eight SSEA4 analogs, could elicit IgM antibodies against SSEA4 when combining the use of Glc-C34.
[0057] FIG. 5: The glycan-protein conjugation method affects the immune response.
DETAILED DESCRIPTIONS
[0058] The present disclosure is based on the surprising discovery that the modification of the stage-specific embryonic antigens (SSEA3 and SSEA4) with certain groups elicited robust IgG antibody response to specifically recognize SSEA3 and SSEA4, respectively.
[0059] In some examples, the modification of SSEA3 comprises a fluoro, an azido or an O-phenyl group at the one or more positions of the glucose of SSEA3. In some examples, the modification of SSEA3 comprises a fluoro, an azido or an O-phenyl group at the one or more positions of the non-reducing end galactose. In some examples, the modification of SSEA4 comprises a fluoro, an azido or an O-phenyl group at one or more positions of the glucose of SSEA4. In some examples, the modification of SSEA4 comprises a fluoro, an azido or an O-phenyl group at one or more positions of the sialic acid residue.
[0060] Described herein are SSEA3 and SSEA4 analogs having the modification at the reducing and/or non-reducing end. Such SSEA3 and SSEA4 analogs can elicit a stronger immune response (e.g., induction of IgG antibodies against SSEA3 and/or SSEA4) as compared to the native SSEA3 and SSEA4. The antibodies induced by an immunogenic composition comprising such unnatural glycan moiety are able to mediate the complement-dependent cell cytotoxicity against tumor cells.
Chemical Definitions
[0061] Definitions of specific functional groups and chemical terms are described in more detail below. The chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 75th Ed., inside cover, and specific functional groups are generally defined as described therein. Additionally, general principles of organic chemistry, as well as specific functional moieties and reactivity, are described in Thomas Sorrell, Organic Chemistry, University Science Books, Sausalito, 1999; Smith and March, March's Advanced Organic Chemistry, 5th Edition, John Wiley & Sons, Inc., New York, 2001; Larock, Comprehensive Organic Transformations, VCH Publishers, Inc., New York, 1989; and Carruthers, Some Modern Methods of Organic Synthesis, 3rd Edition, Cambridge University Press, Cambridge, 1987. Moreover, exemplary glycan and antibody methodologies are described in Wong et al, US20100136042, US20090317837, and US20140051127, the disclosures of each of which are hereby incorporated by reference.
[0062] Compounds described herein can comprise one or more asymmetric centers, and thus can exist in various isomeric forms, e.g., enantiomers and/or diastereomers. For example, the compounds described herein can be in the form of an individual enantiomer, diastereomer or geometric isomer, or can be in the form of a mixture of stereoisomers, including racemic mixtures and mixtures enriched in one or more stereoisomer. Isomers can be isolated from mixtures by methods known to those skilled in the art, including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts; or preferred isomers can be prepared by asymmetric syntheses. See, for example, Jacques et al., Enantiomers, Racemates and Resolutions (Wiley Interscience, New York, 1981); Wilen et al., Tetrahedron 33:2725 (1977); Eliel, Stereochemistry of Carbon Compounds (McGraw-Hill, NY, 1962); and Wilen, Tables of Resolving Agents and Optical Resolutions p. 268 (E.L. Eliel, Ed., Univ. of Notre Dame Press, Notre Dame, Ind. 1972). The invention additionally encompasses compounds described herein as individual isomers substantially free of other isomers, and alternatively, as mixtures of various isomers.
[0063] When a range of values is listed, it is intended to encompass each value and sub-range within the range. For example "C1-6" is intended to encompass C1, C2, C3, C4, C5, C6, C1-6, C1-5, C1-4, C1-3, C1-2, C2-6, C2-5, C2-4, C2-3, C3-6, C3-5, C3-4, C4-6, C4-5, and C5-6.
[0064] "Alkyl" refers to a radical of a straight-chain or branched saturated hydrocarbon group having from 1 to 20 carbon atoms ("C1-20 alkyl"). In some embodiments, an alkyl group has 1 to 10 carbon atoms ("C1-10 alkyl"). In some embodiments, an alkyl group has 1 to 9 carbon atoms ("C1-9 alkyl"). In some embodiments, an alkyl group has 1 to 8 carbon atoms ("C1-8 alkyl"). In some embodiments, an alkyl group has 1 to 7 carbon atoms ("C1-7 alkyl"). In some embodiments, an alkyl group has 1 to 6 carbon atoms ("C1-6 alkyl"). In some embodiments, an alkyl group has 1 to 5 carbon atoms ("C1-5 alkyl"). In some embodiments, an alkyl group has 1 to 4 carbon atoms ("C1-4 alkyl"). In some embodiments, an alkyl group has 1 to 3 carbon atoms ("C1-3 alkyl"). In some embodiments, an alkyl group has 1 to 2 carbon atoms ("C1-2 alkyl"). In some embodiments, an alkyl group has 1 carbon atom ("C1 alkyl"). In some embodiments, an alkyl group has 2 to 6 carbon atoms ("C2-6 alkyl"). Examples of C1-6 alkyl groups include methyl (C1), ethyl (C2), n-propyl (C3), iso-propyl (C3), n-butyl (C4), tert-butyl (C4), sec-butyl (C4), iso-butyl (C4), n-pentyl (C5), 3-pentanyl (C5), amyl (C5), neopentyl (C5), 3-methyl-2-butanyl (C5), tertiary amyl (C5), and n-hexyl (C6). Additional examples of alkyl groups include n-heptyl (C7), n-octyl (C8) and the like.
[0065] Unless otherwise specified, each instance of an alkyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted alkyl") or substituted (a "substituted alkyl") with one or more substituents. In certain embodiments, the alkyl group is unsubstituted C1-10 alkyl (e.g., --CH3). In certain embodiments, the alkyl group is substituted C1-10 alkyl.
[0066] "Alkenyl" refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon double bonds, and no triple bonds ("C2-20 alkenyl"). In some embodiments, an alkenyl group has 2 to 10 carbon atoms ("C2-10 alkenyl"). In some embodiments, an alkenyl group has 2 to 9 carbon atoms ("C2-9 alkenyl"). In some embodiments, an alkenyl group has 2 to 8 carbon atoms ("C2-8 alkenyl"). In some embodiments, an alkenyl group has 2 to 7 carbon atoms ("C2-7 alkenyl"). In some embodiments, an alkenyl group has 2 to 6 carbon atoms ("C2-6 alkenyl"). In some embodiments, an alkenyl group has 2 to 5 carbon atoms ("C2-5 alkenyl"). In some embodiments, an alkenyl group has 2 to 4 carbon atoms ("C2-4 alkenyl"). In some embodiments, an alkenyl group has 2 to 3 carbon atoms ("C2-3 alkenyl"). In some embodiments, an alkenyl group has 2 carbon atoms ("C2 alkenyl"). The one or more carbon-carbon double bonds can be internal (such as in 2-butenyl) or terminal (such as in 1-butenyl). Examples of C2-4 alkenyl groups include ethenyl (C2), 1-propenyl (C3), 2-propenyl (C3), 1-butenyl (C4), 2-butenyl (C4), butadienyl (C4), and the like. Examples of C2-6 alkenyl groups include the aforementioned C2-4 alkenyl groups as well as pentenyl (C5), pentadienyl (C5), hexenyl (C6), and the like. Additional examples of alkenyl include heptenyl (C7), octenyl (C8), octatrienyl (C8), and the like. Unless otherwise specified, each instance of an alkenyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted alkenyl") or substituted (a "substituted alkenyl") with one or more substituents. In certain embodiments, the alkenyl group is unsubstituted C2-10 alkenyl. In certain embodiments, the alkenyl group is substituted C2-10 alkenyl.
[0067] "Alkynyl" refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon triple bonds, and optionally one or more double bonds ("C2-20 alkynyl"). In some embodiments, an alkynyl group has 2 to 10 carbon atoms ("C2-10 alkynyl"). In some embodiments, an alkynyl group has 2 to 9 carbon atoms ("C2-9 alkynyl"). In some embodiments, an alkynyl group has 2 to 8 carbon atoms ("C2-8 alkynyl"). In some embodiments, an alkynyl group has 2 to 7 carbon atoms ("C2-7 alkynyl"). In some embodiments, an alkynyl group has 2 to 6 carbon atoms ("C2-6 alkynyl"). In some embodiments, an alkynyl group has 2 to 5 carbon atoms ("C2-5 alkynyl"). In some embodiments, an alkynyl group has 2 to 4 carbon atoms ("C2-4 alkynyl"). In some embodiments, an alkynyl group has 2 to 3 carbon atoms ("C2-3 alkynyl"). In some embodiments, an alkynyl group has 2 carbon atoms ("C2 alkynyl"). The one or more carbon-carbon triple bonds can be internal (such as in 2-butynyl) or terminal (such as in 1-butynyl). Examples of C2-4 alkynyl groups include, without limitation, ethynyl (C2), 1-propynyl (C3), 2-propynyl (C3), 1-butynyl (C4), 2-butynyl (C4), and the like. Examples of C2-6 alkenyl groups include the aforementioned C2-4 alkynyl groups as well as pentynyl (C5), hexynyl (C6), and the like. Additional examples of alkynyl include heptynyl (C7), octynyl (C8), and the like. Unless otherwise specified, each instance of an alkynyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted alkynyl") or substituted (a "substituted alkynyl") with one or more substituents. In certain embodiments, the alkynyl group is unsubstituted C2-10 alkynyl. In certain embodiments, the alkynyl group is substituted C2-10 alkynyl.
[0068] "Heterocyclyl" or "heterocyclic" refers to a radical of a 3- to 10-membered non-aromatic ring system having ring carbon atoms and 1 to 4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon ("3-10 membered heterocyclyl"). In certain embodiments, the heteroatom is independently selected from nitrogen, sulfur, and oxygen. In heterocyclyl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. A heterocyclyl group can either be monocyclic ("monocyclic heterocyclyl") or a fused, bridged or spiro ring system such as a bicyclic system ("bicyclic heterocyclyl"), and can be saturated or partially unsaturated. Heterocyclyl bicyclic ring systems can include one or more heteroatoms in one or both rings. "Heterocyclyl" also includes ring systems wherein the heterocyclic ring, as defined above, is fused with one or more carbocyclyl groups wherein the point of attachment is either on the carbocyclyl or heterocyclic ring, or ring systems wherein the heterocyclic ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclic ring, and in such instances, the number of ring members continue to designate the number of ring members in the heterocyclic ring system. Unless otherwise specified, each instance of heterocyclyl is independently optionally substituted, i.e., unsubstituted (an "unsubstituted heterocyclyl") or substituted (a "substituted heterocyclyl") with one or more substituents. In certain embodiments, the heterocyclyl group is unsubstituted 3-10 membered heterocyclyl. In certain embodiments, the heterocyclyl group is substituted 3-10 membered heterocyclyl.
[0069] "Aryl" refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 .pi. electrons shared in a cyclic array) having 6-14 ring carbon atoms and zero heteroatoms in the aromatic ring system ("C6-14 aryl"). In some embodiments, an aryl group has six ring carbon atoms ("C6 aryl"; e.g., phenyl). In some embodiments, an aryl group has ten ring carbon atoms ("C10 aryl"; e.g., naphthyl such as 1-naphthyl and 2-naphthyl). In some embodiments, an aryl group has fourteen ring carbon atoms ("C14 aryl"; e.g., anthracyl). "Aryl" also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system. Unless otherwise specified, each instance of an aryl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted aryl") or substituted (a "substituted aryl") with one or more substituents. In certain embodiments, the aryl group is unsubstituted C6-14 aryl. In certain embodiments, the aryl group is substituted C6-14 aryl.
[0070] Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, as defined herein, which are divalent bridging groups are further referred to using the suffix -ene, e.g., alkylene, alkenylene, alkynylene, carbocyclylene, heterocyclylene, arylene, and heteroarylene.
[0071] The term "alkoxy" or "alkyloxy" refers to an --O-alkyl radical, wherein alkyl is optionally substituted alkyl as defined herein. Examples of alkoxy include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, sec-butoxy, and tert-butoxy.
[0072] The term "aryloxy" refers to an --O-aryl, wherein aryl is optionally substituted aryl as defined herein.
[0073] As used herein, the term "optionally substituted" refers to a substituted or unsubstituted moiety.
[0074] Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, as defined herein, are optionally substituted (e.g., "substituted" or "unsubstituted" alkyl, "substituted" or "unsubstituted" alkenyl, "substituted" or "unsubstituted" alkynyl, "substituted" or "unsubstituted" carbocyclyl, "substituted" or "unsubstituted" heterocyclyl, "substituted" or "unsubstituted" aryl or "substituted" or "unsubstituted" heteroaryl group). In general, the term "substituted", whether preceded by the term "optionally" or not, means that at least one hydrogen present on a group (e.g., a carbon or nitrogen atom) is replaced with a permissible substituent, e.g., a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction. Unless otherwise indicated, a "substituted" group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position. The term "substituted" is contemplated to include substitution with all permissible substituents of organic compounds, any of the substituents described herein that results in the formation of a stable compound. The present invention contemplates any and all such combinations in order to arrive at a stable compound. For purposes of this invention, heteroatoms such as nitrogen may have hydrogen substituents and/or any suitable substituent as described herein which satisfy the valencies of the heteroatoms and results in the formation of a stable moiety.
[0075] "Halo" or "halogen" refers to fluorine (fluoro, --F), chlorine (chloro, --Cl), bromine (bromo, --Br), or iodine (iodo, --I).
[0076] "Acyl" as used herein refers to a moiety selected from the group consisting of --C(.dbd.O)Raa,--CHO, --CO2Raa, --C(.dbd.O)N(Rbb)2, --C(.dbd.NRbb)Raa, --C(.dbd.NRbb)ORaa, --C(.dbd.NRbb)N(Rbb)2, --C(.dbd.O)NRbbSO2Raa, --C(.dbd.S)N(Rbb)2, --C(.dbd.O)SRaa, and --C(.dbd.S)SRaa, wherein Raa and Rbb are as defined herein.
[0077] Nitrogen atoms can be substituted or unsubstituted as valency permits, and include primary, secondary, tertiary, and quarternary nitrogen atoms. Exemplary nitrogen atom substituents include, but are not limited to, hydrogen, --OH, --ORaa, --N(Rcc)2, --CN, --C(.dbd.O)Raa, --C(.dbd.O)N(Rcc)2, --CO2Raa, --SO2Raa, --C(.dbd.NRbb)Raa, --C(.dbd.NRcc)ORaa, --C(.dbd.NRcc)N(Rcc)2, --SO2N(Rcc)2, --SO2Rcc, --SO2ORcc, --SORaa, --C(.dbd.S)N(Rcc)2, --C(.dbd.O)SRcc, --C(.dbd.S)SRcc, --P(.dbd.O)2Raa, --P(.dbd.O)(Raa)2, --P(.dbd.O)2N(Rcc)2, --P(.dbd.O)(NRcc)2, C1-10 alkyl, C1-10 perhaloalkyl, C2-10 alkenyl, C2-10 alkynyl, C3-10 carbocyclyl, 3-14 membered heterocyclyl, C6-14 aryl, and 5-14 membered heteroaryl, or two Rcc groups attached to a nitrogen atom are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups, and wherein Raa, Rbb, Rcc, and Rdd are as defined above.
[0078] In certain embodiments, the substituent present on an oxygen atom is an oxygen protecting group (also referred to as a hydroxyl protecting group). Oxygen protecting groups include, but are not limited to, --Raa, --N(Rbb)2, --C(.dbd.O)SRaa, --C(.dbd.O)Raa, --CO2Raa, --C(.dbd.O)N(Rbb)2, --C(.dbd.NRbb)Raa, --C(.dbd.NRbb)ORaa, --C(.dbd.NRbb)N(Rbb)2, --S(.dbd.O)Raa, --SO2Raa, --Si(Raa)3, --P(Rcc)2, --P(Rcc)3, --P(.dbd.O)2Raa, --P(.dbd.O)(Raa)2, --P(.dbd.O)(ORcc)2, --P(.dbd.O)2N(Rbb)2, and --P(.dbd.0)(NRbb)2, wherein Raa, Rbb, and Rcc are as defined herein. Oxygen protecting groups are well known in the art and include those described in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference.
[0079] Exemplary oxygen protecting groups include, but are not limited to, methyl, methoxylmethyl (MOM), methylthiomethyl (MTM), t-butylthiomethyl, (phenyldimethylsilyl)methoxymethyl (SMOM), benzyloxymethyl (BOM), p-methoxybenzyloxymethyl (PMBM), (4-methoxyphenoxy)methyl (p-AOM), guaiacolmethyl (GUM), t-butoxymethyl, 4-pentenyloxymethyl (POM), siloxymethyl, 2-methoxyethoxymethyl (MEM), 2,2,2-trichloroethoxymethyl, bis(2-chloroethoxy)methyl, 2-(trimethylsilyl)ethoxymethyl (SEMOR), tetrahydropyranyl (THP), 3-bromotetrahydropyranyl, tetrahydrothiopyranyl, 1-methoxycyclohexyl, 4-methoxytetrahydropyranyl (MTHP), 4-methoxytetrahydrothiopyranyl, 4-methoxytetrahydrothiopyranyl S,S-dioxide, 1-[(2-chloro-4-methyl)phenyl]-4-methoxypiperidin-4-yl(CTMP), 1,4-dioxan-2-yl, tetrahydrofuranyl, tetrahydrothiofuranyl, 2,3,3a,4,5,6,7,7a-octahydro-7,8,8-trimethyl-4,7-methanobenzofuran-2-yl, 1-ethoxyethyl, 1-(2-chloroethoxy)ethyl, 1-methyl-1-methoxyethyl, 1-methyl-1-benzyloxyethyl, 1-methyl-1-benzyloxy-2-fluoroethyl, 2,2,2-trichloroethyl, 2-trimethylsilylethyl, 2-(phenylselenyl)ethyl, t-butyl, allyl, p-chlorophenyl, p-methoxyphenyl, 2,4-dinitrophenyl, benzyl (Bn), p-methoxybenzyl, 3,4-dimethoxybenzyl, o-nitrobenzyl, p-nitrobenzyl, p-halobenzyl, 2,6-dichlorobenzyl, p-cyanobenzyl, p-phenylbenzyl, 2-picolyl, 4-picolyl, 3-methyl-2-picolyl N-oxido, diphenylmethyl, p,p'-dinitrobenzhydryl, 5-dibenzosuberyl, triphenylmethyl, .alpha.-naphthyldiphenylmethyl, p-methoxyphenyldiphenylmethyl, di(p-methoxyphenyl)phenylmethyl, tri(p-methoxyphenyl)methyl, 4-(4'-bromophenacyloxyphenyl)diphenylmethyl, 4,4',4''-tris(4,5-dichlorophthalimidophenyl)methyl, 4,4',4''-tris(levulinoyloxyphenyl)methyl, 4,4',4''-tris(benzoyloxyphenyl)methyl, 3-(imidazol-1-yl)bis(4',4''-dimethoxyphenyl)methyl, 1,1-bis(4-methoxyphenyl)-1'-pyrenylmethyl, 9-anthryl, 9-(9-phenyl)xanthenyl, 9- (9-phenyl-10-oxo)anthryl, 1,3-benzodithiolan-2-yl, benzisothiazolyl S,S-dioxido, trimethylsilyl (TMS), triethylsilyl (TES), triisopropylsilyl (TIPS), dimethylisopropylsilyl (IPDMS), diethylisopropylsilyl (DEIPS), dimethylthexylsilyl, t-butyldimethylsilyl (TBDMS), t-butyldiphenylsilyl (TBDPS), tribenzylsilyl, tri-p-xylylsilyl, triphenylsilyl, diphenylmethylsilyl (DPMS), t-butylmethoxyphenylsilyl (TBMPS), formate, benzoylformate, acetate, chloroacetate, dichloroacetate, trichloroacetate, trifluoroacetate, methoxyacetate, triphenylmethoxyacetate, phenoxyacetate, p-chlorophenoxyacetate, 3-phenylpropionate, 4-oxopentanoate (levulinate), 4,4-(ethylenedithio)pentanoate (levulinoyldithioacetal), pivaloate, adamantoate, crotonate, 4-methoxycrotonate, benzoate, p-phenylbenzoate, 2,4,6-trimethylbenzoate (mesitoate), methyl carbonate, 9-fluorenylmethyl carbonate (Fmoc), ethyl carbonate, 2,2,2-trichloroethyl carbonate (Troc), 2-(trimethylsilyl)ethyl carbonate (TMSEC), 2-(phenylsulfonyl) ethyl carbonate (Psec), 2-(triphenylphosphonio) ethyl carbonate (Peoc), isobutyl carbonate, vinyl carbonate, allyl carbonate, t-butyl carbonate (BOC), p-nitrophenyl carbonate, benzyl carbonate, p-methoxybenzyl carbonate, 3,4-dimethoxybenzyl carbonate, o-nitrobenzyl carbonate, p-nitrobenzyl carbonate, S-benzyl thiocarbonate, 4-ethoxy-1-napththyl carbonate, methyl dithiocarbonate, 2-iodobenzoate, 4-azidobutyrate, 4-nitro-4-methylpentanoate, o-(dibromomethyl)benzoate, 2-formylbenzenesulfonate, 2-(methylthiomethoxy)ethyl, 4-(methylthiomethoxy)butyrate, 2-(methylthiomethoxymethyl)benzoate, 2,6-dichloro-4-methylphenoxyacetate, 2,6-dichloro-4-(1,1,3,3-tetramethylbutyl)phenoxyacetate, 2,4-bis(1,1-dimethylpropyl)phenoxyacetate, chlorodiphenylacetate, isobutyrate, monosuccinoate, (E)-2-methyl-2-butenoate, o-(methoxyacyl)benzoate, .alpha.-naphthoate, nitrate, alkyl N,N,N',N'-tetramethylphosphorodiamidate, alkyl N-phenylcarbamate, borate, dimethylphosphinothioyl, alkyl 2,4-dinitrophenylsulfenate, sulfate, methanesulfonate (mesylate), benzylsulfonate, and tosylate (Ts).
[0080] It must be noted that as used herein and in the appended claims, the singular forms "a", "an", and "the" include plural reference unless the context clearly dictates otherwise. As well, the terms "a" (or "an"), "one or more" and "at least one" can be used interchangeably herein. It is also to be noted that the terms "comprising", "including", and "having" can be used interchangeably.
[0081] The practice of the present invention will employ, unless otherwise indicated, conventional techniques of molecular biology, microbiology, recombinant DNA, and immunology, which are within the skill of the art. Such techniques are explained fully in the literature. See, for example, Molecular Cloning A Laboratory Manual, 2nd Ed., ed. by Sambrook, Fritsch and Maniatis (Cold Spring Harbor Laboratory Press, 1989); DNA Cloning, Volumes I and II (D. N. Glover ed., 1985); Culture Of Animal Cells (R. I. Freshney, Alan R. Liss, Inc., 1987); Immobilized Cells And Enzymes (IRL Press, 1986); B. Perbal, A Practical Guide To Molecular Cloning (1984); the treatise, Methods In Enzymology (Academic Press, Inc., N.Y.); Gene Transfer Vectors For Mammalian Cells (J. H. Miller and M. P. Calos eds., 1987, Cold Spring Harbor Laboratory); Methods In Enzymology, Vols. 154 and 155 (Wu et al. eds.), Immunochemical Methods In Cell And Molecular Biology (Mayer and Walker, eds., Academic Press, London, 1987); Antibodies: A Laboratory Manual, by Harlow and Lane s (Cold Spring Harbor Laboratory Press, 1988); and Handbook Of Experimental Immunology, Volumes I-IV (D. M. Weir and C. C. Blackwell, eds., 1986).
[0082] As used herein, the term "glycan" refers to a polysaccharide, or oligosaccharide. Glycan is also used herein to refer to the carbohydrate portion of a glycoconjugate, such as a glycoprotein, glycolipid, glycopeptide, glycoproteome, peptidoglycan, lipopolysaccharide or a proteoglycan. Glycans usually consist solely of O-glycosidic linkages between monosaccharides. For example, cellulose is a glycan (or more specifically a glucan) composed of .beta.-1,4-linked D-glucose, and chitin is a glycan composed of .beta.-1,4-linked N-acetyl-D-glucosamine. Glycans can be homo or heteropolymers of monosaccharide residues, and can be linear or branched. Glycans can be found attached to proteins as in glycoproteins and proteoglycans. They are generally found on the exterior surface of cells. O- and N-linked glycans are very common in eukaryotes but may also be found, although less commonly, in prokaryotes. N-Linked glycans are found attached to the R-group nitrogen (N) of asparagine in the sequon. The sequon is a Asn-X-Ser or Asn-X-Thr sequence, where X is any amino acid except praline.
[0083] As used herein, the term "antigen" is defined as any substance capable of eliciting an immune response.
[0084] As used herein, the term "immunogenicity" refers to the ability of an immunogen, antigen, or vaccine to stimulate an immune response.
[0085] As used herein, the term "CD1d" refers to a member of the CD1 (cluster of differentiation 1) family of glycoproteins expressed on the surface of various human antigen-presenting cells. CD1d presented lipid antigens activate natural killer T cells. CD1d has a deep antigen-binding groove into which glycolipid antigens bind. CD1d molecules expressed on dendritic cells can bind and present glycolipids, including alpha-GalCer analogs such as C34.
[0086] As used herein, the term "epitope" is defined as the parts of an antigen molecule which contact the antigen binding site of an antibody or a T cell receptor.
[0087] As used herein, the term "vaccine" refers to a preparation that contains an antigen, consisting of whole disease-causing organisms (killed or weakened) or components of such organisms, such as proteins, peptides, or polysaccharides, that is used to confer immunity against the disease that the organisms cause. Vaccine preparations can be natural, synthetic or derived by recombinant DNA technology.
[0088] As used herein, the term "antigen specific" refers to a property of a cell population such that supply of a particular antigen, or a fragment of the antigen, results in specific cell proliferation.
[0089] As used herein, the term "specifically binding," refers to the interaction between binding pairs (e.g., an antibody and an antigen). In various instances, specifically binding can be embodied by an affinity constant of about 10-6 moles/liter, about 10-7 moles/liter, or about 10-8 moles/liter, or less.
[0090] As used herein, the terms glycoenzymes refers to at least in part the enzymes in the globo-series biosynthetic pathway; exemplary glycoenzymes include alpha-4GalT; beta-4GalNAcT-I; or beta-3GalT-V enzymes.
[0091] As used herein, the term "globo-series pathway" includes to a biosynthetic and enzymatic pathways described in FIG. 1.
[0092] An "isolated" antibody is one which has been identified and separated and/or recovered from a component of its natural environment. Contaminant components of its natural environment are materials which would interfere with research, diagnostic or therapeutic uses for the antibody, and may include enzymes, hormones, and other proteinaceous or nonproteinaceous solutes. In one embodiment, the antibody will be purified (1) to greater than 95% by weight of antibody as determined by, for example, the Lowry method, and in some embodiments more than 99% by weight, (2) to a degree sufficient to obtain at least 15 residues of N-terminal or internal amino acid sequence by use of, for example, a spinning cup sequenator, or (3) to homogeneity by SDS-PAGE under reducing or nonreducing conditions using, for example, Coomassie blue or silver stain. Isolated antibody includes the antibody in situ within recombinant cells since at least one component of the antibody's natural environment will not be present. Ordinarily, however, isolated antibody will be prepared by at least one purification step.
[0093] "Binding affinity" generally refers to the strength of the sum total of noncovalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, "binding affinity" refers to intrinsic binding affinity which reflects a 1:1 interaction between members of a binding pair (e.g., antibody and antigen). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (Kd). Affinity can be measured by common methods known in the art, including those described herein. Low-affinity antibodies generally bind antigen slowly and tend to dissociate readily, whereas high-affinity antibodies generally bind antigen faster and tend to remain bound longer. A variety of methods of measuring binding affinity are known in the art, any of which can be used for purposes of the present invention. Specific illustrative embodiments are described in the following.
[0094] "Antibody fragments" comprise only a portion of an intact antibody, wherein the portion retains at least one, and as many as most or all, of the functions normally associated with that portion when present in an intact antibody. In one embodiment, an antibody fragment comprises an antigen binding site of the intact antibody and thus retains the ability to bind antigen. In another embodiment, an antibody fragment, for example one that comprises the Fc region, retains at least one of the biological functions normally associated with the Fc region when present in an intact antibody, such as FcRn binding, antibody half life modulation, ADCC function and complement binding. In one embodiment, an antibody fragment is a monovalent antibody that has an in vivo half life substantially similar to an intact antibody. For example, such an antibody fragment may comprise an antigen binding arm linked to an Fc sequence capable of conferring in vivo stability to the fragment.
[0095] The monoclonal antibodies herein specifically include "chimeric" antibodies in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (U.S. Pat. No. 4,816,567; and Morrison et al., Proc. Natl. Acad. Sci. USA 81:6851-6855 (1984)).
[0096] Humanized" forms of non-human (e.g., murine) antibodies are chimeric antibodies that contain minimal sequence derived from non-human immunoglobulin. In one embodiment, a humanized antibody is a human immunoglobulin (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from a hypervariable region of a non-human species (donor antibody) such as mouse, rat, rabbit or nonhuman primate having the desired specificity, affinity, and/or capacity. In some instances, framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, humanized antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable loops correspond to those of a non-human immunoglobulin and all or substantially all of the FRs are those of a human immunoglobulin sequence. The humanized antibody optionally will also comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin. For further details, see Jones et al., Nature 321:522-525 (1986); Riechmann et al., Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol. 2:593-596 (1992). See also the following review articles and references cited therein: Vaswani and Hamilton, Ann. Allergy, Asthma & Immunol. 1:105-115 (1998); Harris, Biochem. Soc. Transactions 23:1035-1038 (1995); Hurle and Gross, Curr. Op. Biotech. 5:428-433 (1994).
[0097] A "blocking" antibody or an "antagonist" antibody is one which inhibits or reduces biological activity of the antigen it binds. Certain blocking antibodies or antagonist antibodies substantially or completely inhibit the biological activity of the antigen.
[0098] An "agonist antibody", as used herein, is an antibody which mimics at least one of the functional activities of a polypeptide of interest.
[0099] A "disorder" is any condition that would benefit from treatment with an antibody of the invention. This includes chronic and acute disorders or diseases including those pathological conditions which predispose the mammal to the disorder in question. Non-limiting examples of disorders to be treated herein include cancer.
[0100] The terms "cell proliferative disorder" and "proliferative disorder" refer to disorders that are associated with some degree of abnormal cell proliferation. In one embodiment, the cell proliferative disorder is cancer.
[0101] "Tumor," as used herein, refers to all neoplastic cell growth and proliferation, whether malignant or benign, and all pre-cancerous and cancerous cells and tissues. The terms "cancer," "cancerous," "cell proliferative disorder," "proliferative disorder" and "tumor" are not mutually exclusive as referred to herein.
[0102] The terms "cancer" and "cancerous" refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth/proliferation. Examples of cancer include, but are not limited to, carcinoma, lymphoma (e.g., Hodgkin's and non-Hodgkin's lymphoma), blastoma, sarcoma, and leukemia. More particular examples of such cancers include squamous cell cancer, small-cell lung cancer, non-small cell lung cancer, adenocarcinoma of the lung, squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastrointestinal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, breast cancer, colon cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney cancer, liver cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma, leukemia and other lymphoproliferative disorders, and various types of head and neck cancer.
[0103] The term "globo-series -related disorder" refers to or describes a disorder that is typically characterized by or contributed to by aberrant functioning or presentation of the pathway. Examples of such disorders include, but are not limited to, hyperproliferative diseases, including cancer.
[0104] Examples of immunologic deficiency syndromes include, but are not limited to, ataxia telangiectasia, leukocyte-adhesion deficiency syndrome, lymphopenia, dysgammaglobulinemia, HIV or deltaretrovirus infections, common variable immunodeficiency, severe combined immunodeficiency, phagocyte bactericidal dysfunction, agammaglobulinemia, DiGeorge syndrome, and Wiskott-Aldrich syndrome. Examples of hypersensitivity include, but are not limited to, allergies, asthma, dermatitis, hives, anaphylaxis, Wissler's syndrome, and thrombocytopenic purpura.
[0105] As used herein, "treatment" refers to clinical intervention in an attempt to alter the natural course of the individual or cell being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing or decreasing inflammation and/or tissue/organ damage, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, antibodies of the invention are used to delay development of a disease or disorder.
[0106] An "individual" or a "subject" is a vertebrate. In certain embodiments, the vertebrate is a mammal. Mammals include, but are not limited to, farm animals (such as cows), sport animals, pets (such as cats, dogs, and horses), primates, mice and rats. In certain embodiments, the vertebrate is a human.
[0107] "Mammal" for purposes of treatment refers to any animal classified as a mammal, including humans, domestic and farm animals, and zoo, sports, or pet animals, such as dogs, horses, cats, cows, etc. In certain embodiments, the mammal is human.
[0108] An "effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
[0109] A "therapeutically effective amount" of a substance/molecule of the invention may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the substance/molecule, to elicit a desired response in the individual. A therapeutically effective amount is also one in which any toxic or detrimental effects of the substance/molecule are outweighed by the therapeutically beneficial effects. A "prophylactically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired prophylactic result. Typically but not necessarily, since a prophylactic dose is used in subjects prior to or at an earlier stage of disease, the prophylactically effective amount would be less than the therapeutically effective amount.
[0110] The term "cytotoxic agent" as used herein refers to a substance that inhibits or prevents the function of cells and/or causes destruction of cells. The term is intended to include radioactive isotopes (e.g., At211, 1131, 1125, Y90, Rel86, Rel88, Sm153, Bi212, P32, Pb212 and radioactive isotopes of Lu), chemotherapeutic agents (e.g., methotrexate, adriamicin, vinca alkaloids (vincristine, vinblastine, etoposide), doxorubicin, melphalan, mitomycin C, chlorambucil, daunorubicin or other intercalating agents, enzymes and fragments thereof such as nucleolyticenzymes, antibiotics, and toxins such as small molecule toxins or enzymatically active toxins of bacterial, fungal, plant or animal origin, including fragments and/or variants thereof, and the various antitumor or anticancer agents disclosed below. Other cytotoxic agents are described below. A tumoricidal agent causes destruction of tumor cells.
[0111] A "chemotherapeutic agent" is a chemical compound useful in the treatment of cancer. Examples of chemotherapeutic agents include alkylating agents such as thiotepa and CYTOXAN.RTM. cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, trietylenephosphoramide, triethiylenethiophosphoramide and trimethylolomelamine; acetogenins (especially bullatacin and bullatacinone); delta-9-tetrahydrocannabinol (dronabinol, MARINOL.RTM.); beta-lapachone; lapachol; colchicines; betulinic acid; a camptothecin (including the synthetic analogue topotecan (HYCAMTIN.RTM.), CPT-11 (irinotecan, CAMPTOSAR.RTM.), acetylcamptothecin, scopolectin, and 9-aminocamptothecin); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogues); podophyllotoxin; podophyllinic acid; teniposide; cryptophycins (particularly cryptophycin 1 and cryptophycin 8); dolastatin; duocarmycin (including the synthetic analogues, KW-2189 and CB1-TM1); eleutherobin; pancratistatin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlomaphazine, cholophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, and ranimnustine; antibiotics such as the enediyne antibiotics (e.g., calicheamicin, especially calicheamicin gammall and calicheamicin omegaIl (see, e.g., Agnew, Chem. Intl. Ed. Engl., 33: 183-186 (1994)); dynemicin, including dynemicin A; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antiobiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, caminomycin, carzinophilin, chromomycinis, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN.RTM. doxorubicin (including morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins such as mitomycin C, mycophenolic acid, nogalamycin, olivomycins, peplomycin, potfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites such as methotrexate and 5-fluorouracil (5-FU); folic acid analogues such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6-mercaptopurine, thiamiprine, thioguanine; pyrimidine analogs such as ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replenisher such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; eniluracil; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elformithine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidainine; maytansinoids such as maytansine and ansamitocins; mitoguazone; mitoxantrone; mopidanmol; nitraerine; pentostatin; phenamet; pirarubicin; losoxantrone; 2-ethylhydrazide; procarbazine; PSK.RTM. polysaccharide complex (JHS Natural Products, Eugene, Oreg.); razoxane; rhizoxin; sizofuran; spirogermanium; tenuazonic acid; triaziquone; 2,2',2''-trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine (ELDISINE.RTM., FILDESIN.RTM.); dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside ("Ara-C"); thiotepa; taxoids, e.g., TAXOL.RTM. paclitaxel (Bristol-Myers Squibb Oncology, Princeton, N.J.), ABRAXANE.TM. Cremophor-free, albumin-engineered nanoparticle formulation of paclitaxel (American Pharmaceutical Partners, Schaumberg, Ill.), and TAXOTERE.RTM. doxetaxel (Rhone-Poulenc Rorer, Antony, France); chloranbucil; gemcitabine (GEMZAR.RTM.); 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine (VELBAN.RTM.); platinum; etoposide (VP-16); ifosfamide; mitoxantrone; vincristine (ONCOVIN.RTM.); oxaliplatin; leucovovin; vinorelbine (NAVELBINE.RTM.); novantrone; edatrexate; daunomycin; aminopterin; ibandronate; topoisomerase inhibitor RFS 2000; difluoromethylornithine (DMFO); retinoids such as retinoic acid; capecitabine (XELODA.RTM.); pharmaceutically acceptable salts, acids or derivatives of any of the above; as well as combinations of two or more of the above such as CHOP, an abbreviation for a combined therapy of cyclophosphamide, doxorubicin, vincristine, and prednisolone, and FOLFOX, an abbreviation for a treatment regimen with oxaliplatin (ELOXATIN.TM.) combined with 5-FU and leucovovin.
[0112] Unless defined otherwise, all technical and scientific terms used herein have the same meanings as commonly understood by one of ordinary skill in the art to which this invention belongs. Although any methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present invention, the preferred methods and materials are now described. All publications and patents specifically mentioned herein are incorporated by reference for all purposes including describing and disclosing the chemicals, cell lines, vectors, animals, instruments, statistical analysis and methodologies which are reported in the publications which might be used in connection with the invention. All references cited in this specification are to be taken as indicative of the level of skill in the art. Nothing herein is to be construed as an admission that the invention is not entitled to antedate such disclosure by virtue of prior invention.
[0113] In one aspect, the present disclosure is based on the surprising discovery that the modification of the stage-specific embryonic antigens (SSEA3 and SSEA4) with certain groups elicited robust IgG antibody response to specifically recognize SSEA3 and SSEA4, respectively.
[0114] In some examples, the modification of SSEA3 comprises a fluoro, an azido or an O-phenyl group at the one or more positions of the glucose of SSEA3. In some examples, the modification of SSEA3 comprises a fluoro, an azido or an O-phenyl group at the one or more positions of the non-reducing end galactose. In some examples, the modification of SSEA4 comprises a fluoro, an azido or an O-phenyl group at one or more positions of the glucose of SSEA4. In some examples, the modification of SSEA4 comprises a fluoro, an azido or an O-phenyl group at one or more positions of the sialic acid residue.
[0115] In certain aspects, the present disclosure provides SSEA3 and SSEA4 analogs having the modification at the reducing and/or non-reducing end. Such SSEA3 and SSEA4 analogs can elicit a stronger immune response (e.g., induction of IgG antibodies against SSEA3 and/or SSEA4) as compared to the native SSEA3 and SSEA4. The antibodies induced by an immunogenic composition comprising such unnatural glycan moiety are able to mediate the complement-dependent cell cytotoxicity against tumor cells.
[0116] Compounds
[0117] Accordingly, the present invention also features novel compounds consisting of the modified carbohydrate antigens (SSEA3 and SSEA4), glycan conjugates comprising such, and immunogenic compositions and vaccines thereof
[0118] In one aspect, the present invention provides a compound of formula (I):
##STR00007##
[0119] or a salt thereof,
[0120] wherein: [0121] X1 is --OR or --SR, wherein R is hydrogen, a oxygen or sulfur protecting group, optionally substituted C1-10 alkyl, optionally substituted aryl, optionally substituted acyl, or optionally substituted imidoyl; [0122] each instance of R1, R2, R3, R4, R5, R6 and L is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N3, --NO2, --N(RB)2, --N(RA)C(O)RA, --ORA, --OC(O)RA, --SRA, --C(O)N(RB)2, --CN, --C(O)RA, --C(O)ORA, --S(O)RA, --SO2RA, --SO2N(RB)2, and --NHSO2RB; [0123] each instance of RA is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; [0124] each instance of RB is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; and [0125] provided the compound is not of the formula:
##STR00008##
[0126] In certain embodiments, X1 is in the alpha configuration. In certain embodiments, X1 is in the beta configuration.
[0127] In some embodiments, X1 is --ORA. In some embodiments, X1 is --OH. In some embodiments, X1 is --O(protecting group). In some embodiments, X1 is --ORA, wherein RA is unsubstituted C1-10 alkyl. In some embodiments, X1 is --ORA, wherein RA is substituted C1-10 alkyl. In some embodiments, X1 is --ORA, wherein RA is unsubstituted aryl. In some embodiments, X1 is --ORA, wherein RA is substituted aryl. In some embodiments, X1 is --ORA, wherein RA is unsubstituted acyl. In some embodiments, X1 is --ORA, wherein RA is substituted acyl. In some embodiments, X1 is --ORA, wherein RA is unsubstituted imidoyl. In some embodiments, X1 is --ORA, wherein RA is substituted imidoyl.
[0128] In some embodiments, X1 is --SRA. In some embodiments, X1 is --SH. In some embodiments, X1 is --S(protecting group). In some embodiments, X1 is --SRA, wherein RA is unsubstituted C1-10 alkyl. In some embodiments, X1 is --SRA, wherein RA is substituted C1-10 alkyl. In certain embodiments, X1 is --SCH3. In some embodiments, X1 is --SRA, wherein RA is unsubstituted aryl. In some embodiments, X1 is --SRA, wherein RA is substituted aryl. In some embodiments, X1 is --SRA, wherein RA is unsubstituted acyl. In some embodiments, X1 is --SRA, wherein RA is substituted acyl. In some embodiments, X1 is --SRA, wherein RA is unsubstituted imidoyl. In some embodiments, X1 is --SRA, wherein RA is substituted imidoyl.
[0129] In some embodiments, X1 is C1-10 alkoxy. In some embodiments, X1 is C1-3 alkoxy.
[0130] In some embodiments, X1 is selected from the group consisting of alpha-thiomethyl, beta-thiomethyl, alpha-thiocresyl, beta-thiocresyl, alpha-t-butyldiphenylsilyloxy, beta-t-butyldiphenylsilyloxy, and alpha-methoxy.
[0131] In some embodiments, R1 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R1 is --N3. In certain embodiments, R1 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R1 is --NH2. In certain embodiments, R1 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R1 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R1 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2.
[0132] In certain embodiments, R1 is --NH(Cbz). In certain embodiments, R1 is --NH(Fmoc). In certain embodiments, R1 is --NHC(O)CCl3. In certain embodiments, R1 is --NHC(O)CH3. In certain embodiments, R1 is --N(C(O)CH3)2.
[0133] In some embodiments, R2 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R2 is --N3. In certain embodiments, R2 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R2 is --NH2. In certain embodiments, R2 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R2 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R2 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R2 is --NH(Cbz). In certain embodiments, R2 is --NH(Fmoc). In certain embodiments, R2 is --NHC(O)CCl3. In certain embodiments, R2 is --NHC(O)CH3. In certain embodiments, R2 is --N(C(O)CH3)2.
[0134] In some embodiments, R3 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R3 is --N3. In certain embodiments, R3 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R3 is --NH2. In certain embodiments, R3 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R3 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R3 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R3 is --NH(Cbz). In certain embodiments, R3 is --NH(Fmoc). In certain embodiments, R3 is --NHC(O)CCl3. In certain embodiments, R3 is --NHC(O)CH3. In certain embodiments, R3 is --N(C(O)CH3)2.
[0135] In some embodiments, R4 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R4 is --N3. In certain embodiments, R4 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R4 is --NH2. In certain embodiments, R4 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R4 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R4 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2.
[0136] In certain embodiments, R4 is --NH(Cbz). In certain embodiments, R4 is --NH(Fmoc). In certain embodiments, R4 is --NHC(O)CCl3. In certain embodiments, R4 is --NHC(O)CH3. In certain embodiments, R4 is --N(C(O)CH3)2.
[0137] In some embodiments, R5 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R5 is --N3. In certain embodiments, R5 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R5 is --NH2. In certain embodiments, R5 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R5 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R5 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R5 is --NH(Cbz). In certain embodiments, R5 is --NH(Fmoc). In certain embodiments, R5 is --NHC(O)CCl3. In certain embodiments, R5 is --NHC(O)CH3. In certain embodiments, R5 is --N(C(O)CH3)2.
[0138] In some embodiments, R6 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R6 is --N3. In certain embodiments, R6 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R6 is --NH2. In certain embodiments, R6 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R6 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R6 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R6 is --NH(Cbz). In certain embodiments, R6 is --NH(Fmoc). In certain embodiments, R6 is --NHC(O)CCl3. In certain embodiments, R6 is --NHC(O)CH3. In certain embodiments, R6 is --N(C(O)CH3)2.
[0139] In some embodiments, R1, R2 and R3 are the same. In some embodiments, R1, R2 and R3 are --OH. In some embodiments, R4, R5 and R6 are the same. In some embodiments, R4, R5 and R6 are --OH.
[0140] In certain embodiments, L is --OH.
[0141] In certain embodiments, L is --OH and R1 is --N3. In certain embodiments, L is --OH, R1 is --N3, and each instance of R2, R3, R4, R5 and R6 is --OH.
[0142] In certain embodiments, L is --OH and R2 is --N3. In certain embodiments, L is --OH, R2 is --N3, and each instance of R1, R3, R4, R5 and R6 is --OH.
[0143] In certain embodiments, L is --OH and R3 is --N3. In certain embodiments, L is --OH, R3 is --N3, and each instance of R1, R2, R4, R5 and R6 is --OH.
[0144] In certain embodiments, L is --OH and R4 is --N3. In certain embodiments, L is --OH, R4 is --N3, and each instance of R1, R2, R3, R5 and R6 is --OH.
[0145] In certain embodiments, L is --OH and R5 is --N3. In certain embodiments, L is --OH, R5 is --N3, and each instance of R1, R2, R3, R4 and R6 is --OH.
[0146] In certain embodiments, L is --OH and R6 is --N3. In certain embodiments, L is --OH, R6 is --N3, and each instance of R1, R2, R3, R4 and R5 is --OH.
[0147] In certain embodiments, each instance of R1, R2, R3, R4, R5, R6 and L is --F. In certain embodiments, R1 is --F. In certain embodiments, R2 is --F. In certain embodiments, R3 is --F. In certain embodiments, R4 is --F. In certain embodiments, R5 is --F. In certain embodiments, R6 is --F. In certain embodiments, L is --F.
[0148] In certain embodiments, L is of the following structure:
##STR00009##
[0149] wherein: [0150] each instance of R8, R9, R10 and R11 is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, optionally substituted aryl, --N3, --NO2, --N(RB)2, --N(RA)C(O)RA, --ORA, --OC(O)RA, --SRA, -- C(O)N(RB)2, --CN, --C(O)RA, --C(O)ORA, --S(O)RA, --SO2RA, --SO2N(RB)2, and --NHSO2RB; [0151] R.sub.N is selected from --N3, --NO2, --N(RB)2, --N(RA)C(O)RA, --ORA, --OC(O)RA, --SRA, --C(O)N(RB)2, --CN, --C(O)RA, --C(O)ORA, --S(O)RA, --SO2RA, --SO2N(RB)2, and --NHSO2RB; [0152] each instance of RA is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl; and [0153] each instance of RB is independently selected from hydrogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted heterocyclyl, and optionally substituted aryl.
[0154] In some embodiments, the compound is of Formula (II)
##STR00010##
[0155] wherein: R1, R2, R3, R8, R9, R10, R11 and R.sub.N and X1 are as described herein, and
[0156] provided the compound is not of the formula:
##STR00011##
[0157] In some embodiments, R8 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R8 is --N3. In certain embodiments, R8 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R8 is --NH2. In certain embodiments, R8 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R8 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R8 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R8 is --NH(Cbz). In certain embodiments, R8 is --NH(Fmoc). In certain embodiments, R8 is --NHC(O)CCl3. In certain embodiments, R8 is --NHC(O)CH3. In certain embodiments, R8 is --N(C(O)CH3)2.
[0158] In some embodiments, R9 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R9 is --N3. In certain embodiments, R9 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R9 is --NH2. In certain embodiments, R9 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R9 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R9 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R9 is --NH(Cbz). In certain embodiments, R9 is --NH(Fmoc). In certain embodiments, R9 is --NHC(O)CCl3. In certain embodiments, R9 is --NHC(O)CH3. In certain embodiments, R9 is --N(C(O)CH3)2.
[0159] In some embodiments, R10 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R10 is --N3. In certain embodiments, R10 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R10 is --NH2. In certain embodiments, R10 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R10 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R10 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R10 is --NH(Cbz). In certain embodiments, R10 is --NH(Fmoc). In certain embodiments, R10 is --NHC(O)CCl3. In certain embodiments, R10 is --NHC(O)CH3. In certain embodiments, R10 is --N(C(O)CH3)2.
[0160] In some embodiments, R11 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R11 is --N3. In certain embodiments, R11 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R11 is --NH2. In certain embodiments, R11 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R11 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R11 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R11 is --NH(Cbz). In certain embodiments, R11 is --NH(Fmoc). In certain embodiments, R11 is --NHC(O)CCl3. In certain embodiments, R11 is --NHC(O)CH3. In certain embodiments, R11 is --N(C(O)CH3)2.
[0161] In some embodiments, R12 is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R12 is --N3. In certain embodiments, R12 is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R12 is --NH2. In certain embodiments, R12 is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R12 is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R12 is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R12 is --NH(Cbz). In certain embodiments, R12 is --NH(Fmoc). In certain embodiments, R12 is --NHC(O)CCl3. In certain embodiments, R12 is --NHC(O)CH3. In certain embodiments, R12 is --N(C(O)CH3)2.
[0162] In some embodiments, R.sub.N is --N3 or --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, RN is --N3. In certain embodiments, R.sub.N is --N(RW)2, wherein each RW is independently hydrogen or a nitrogen protecting group. In certain embodiments, R.sub.N is --NH2. In certain embodiments, R.sub.N is --NHRW, wherein RW is a nitrogen protecting group. In certain embodiments, R.sub.N is --N(RW)2, wherein each RW is a nitrogen protecting group. In certain embodiments, R.sub.N is selected from the group consisting of --N3, --NH(Cbz), --NH(Boc), --NH(Fmoc), --NHC(O)CCl3, --NHC(O)CH3, and --N(C(O)CH3)2. In certain embodiments, R.sub.N is --NH(Cbz). In certain embodiments, R.sub.N is --NH(Fmoc). In certain embodiments, R.sub.N is --NHC(O)CCl3. In certain embodiments, R.sub.N is --NHC(O)CH3. In certain embodiments, R.sub.N is --N(C(O)CH3)2.
[0163] Immunogenic Compositions
[0164] In another aspect, the present invention provides an immunogenic composition, comprising (a) a glycan conjugate including a carrier and one or more glycans, and optionally (b) an adjuvant,
[0165] wherein: each of the one or more glycans is conjugated with the carrier through a linker, having the formula (III) or (IV):
##STR00012##
[0166] wherein X1, R1, R2, R3, R4, R5, R6, R8, R9, R10, R11, L and R.sub.N are as described herein.
[0167] In certain embodiments, the linker is a hetero- or homo-bifunctional linker.
[0168] In certain embodiments, the linker is a homo-bifunctional p-nitrophenyl linker.
[0169] In certain embodiments, the linker includes at least one sulfur atom, carboxylate group, amide group, carbamate group, carbonate group, thiocarbamate group, thiocarbonate group, thioether group, succinamide group, n-hydroxy succinamide group, or any combination thereof.
[0170] In certain embodiments, the linker is wherein --L.sup.1--L.sup.2-- is a bond, O, S , NRL1a--, --C(.dbd.O)--, --NRL1 aC(.dbd.O)--, --NRL1 aC(.dbd.O)O--, --C(.dbd.O)NRL1a--, --OC(.dbd.O)NRL1a--, --SC(.dbd.O)--, --C(.dbd.O)S--, --OC(.dbd.O)--, --C(.dbd.O)O--, --NRL1aC(.dbd.S)--, --C(.dbd.S)NRL1a--, trans-CRL1b=CRL1b--, cis-CRL1b=CRL1b , C.ident.C , OC(RL1b)2-, --C(RL1b)2O--, --NRL1aC(RL1b)2--, --C(RL1b)2NRL 1 a--, --SC(RL1b)2-, --C(RL1b)2S--, --S(.dbd.O)2O--, --OS(.dbd.O)2--, --S(.dbd.O)2NRL1a--, --NRL1aS(.dbd.O)2-, or an optionally substituted C1-20 hydrocarbon chain, optionally wherein one or more carbon units of the hydrocarbon chain is replaced with --O--, --S--, --NRL1a--, --C(.dbd.O)--, NRL1 aC(.dbd.O)--, --NRL1 aC(.dbd.O)O--, --C(.dbd.O)NRL 1 a--, --OC(.dbd.O)NRL 1 a--, --S C(.dbd.O)--, --C(.dbd.O)S--, --OC(.dbd.O)--, --C(.dbd.O)O--, --NRL1aC(.dbd.S)--, --C(.dbd.S)NRL1a--, trans-CRL1b=CRL1b--, cis-CRL1b=CRL1b , C.ident.C , S(.dbd.O)20-, --OS(.dbd.O)2--, --S(.dbd.O)2NRL1a--, or --NRL1aS(.dbd.O)2--, wherein RL1a is hydrogen, optionally substituted C1-6 alkyl, or a nitrogen protecting group, or RL1a is joined with the adjacent carbon atom to form an optionally substituted heterocyclic ring, and wherein each occurrence of RL1b is independently selected from the group consisting of hydrogen, halogen, optionally substituted C1-10 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted carbocyclyl, optionally substituted heterocyclyl, optionally substituted aryl, and optionally substituted heteroaryl, or RL1b is joined with the adjacent carbon or nitrogen or oxygen atom to form an optionally substituted carbocyclic or heterocyclic ring, or two RL1b groups are joined to form an optionally substituted carbocyclic or optionally substituted heterocyclic ring; and L2 is a moiety derived from a crosslinking reagent capable of crosslinking the carrier and L1.
[0171] The carrier can be a protein, a lipid, a lipolized protein, a virus, a peptide, or a dendrimer of glycopeptides. In certain embodiments, the carrier is a peptide comprising a T cell epitope.
[0172] Examples of carrier proteins which may be used in the present invention are tetanus toxoid (TT), diphtheria toxoid (DT), diphtheria toxin cross-reacting material 197 (CRM197), fragment C of TT, Keyhole limpet hemocyanin (KLH), bovine serum albumin (BSA), protein D, outer-membrane protein (OMP) and pneumolysin, diphtheria toxin cross-reacting material 197 (CRM197) or other DT point mutants, such as CRM176, CRM228, CRM 45 (Uchida et al J. Biol. Chem. 218; 3838-3844, 1973); CRM 9, CRM 45, CRM102, CRM 103 and CRM107 and other mutations described in the art.
[0173] In certain embodiments, the glycan conjugate is of the formula (IV-a) or (IV-b):
##STR00013##
[0174] wherein m is an integer of 1 to 40, inclusive.
[0175] In certain embodiments, m is an integer of 1 to 30, inclusive. As generally defined herein, m is an integer of 1 to 20 inclusive. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 4. In certain embodiments, m is 6. In certain embodiments, m is 8. In certain embodiments, m is 10. In certain embodiments, m is 15. In certain embodiments, m is 20. In certain embodiments, m is 30. In certain embodiments, m is 40.
[0176] In another aspect, the present invention provides a glycan conjugate mixture comprising at least two of the glycan conjugates as described herein. In certain embodiments, the average value of w in the glycan mixture is from about 1.0 to about 40.0. In certain embodiments, the average value of w in the glycan mixture is from about 1.0 to 10.0. In certain embodiments, the average value of w in the glycan mixture is about 5.7, 4.9, 2.9, 2.8, or 3.1. In certain embodiments, the average value of w in the glycan mixture is about 4.9, 2.9, 2.8, or 3.1.
[0177] In certain embodiments, the immunogenic compositions described herein include an immunogenically effective amount of a glycan conjugate of the invention. In certain embodiments, the immunogenic composition includes a pharmaceutically effective amount of the inventive glycan conjugate.
[0178] The compounds of the invention can be synthesized using procedures described herein and also see US20140051127.
[0179] The immunogenic conjugate of the invention may include one or more molecules (e.g., 1-40, 1-20, 1-25, 1-30,) of the same or different SSEA3 and/or SSEA4 analogs and/or related derivatives. Additional descriptions and related procedures for generating glycan conjugates are described below. Also see U.S. Pat. No. 8,268,969. The contents of which is hereby incorporated by reference.
[0180] In certain embodiments, the immunogenic composition of the invention may include one or more adjuvants. Suitable adjuvants can include, for example, C34, 7DW8-5, C17, C23, C-30, alpha-galactoceramide, Gluco-C34, Aluminum salt, Squalene, MF59, and QS-21).
[0181] As used herein, the term "alum adjuvant" refers to an aluminum salt with immune adjuvant activity. This agent adsorbs and precipitates protein antigens in solution; the resulting precipitate improves vaccine immunogenicity by facilitating the slow release of antigen from the vaccine depot formed at the site of inoculation.
[0182] As used herein, the term "immunologic adjuvant" refers to a substance used in conjunction with an immunogen which enhances or modifies the immune response to the immunogen. The .alpha.-GalCer analogs of the present disclosure are used as immunologic adjuvants to modify or augment the effects of a vaccine by stimulating the immune system of a patient who is administered the vaccine to respond to the vaccine more vigorously. In an exemplary implementation, the analog C34 is used as an adjuvant. The structures of C34 and other alpha-galactosyl ceramide analogs and their use as adjuvants are disclosed in detail in U.S. Pat. No. 7,928,077.
[0183] As used herein, the term "glycolipid" refers to a carbohydrate-attached lipid that serves as a marker for cellular recognition.
[0184] The glycolipids C34, Gluco-C34, C23 and 7DW8-5 have the following structures:
##STR00014##
[0185] The immunogenic composition can further include a pharmaceutically acceptable excipient. In certain embodiments, the immunogenic compositions described herein include a pharmaceutically effective amount of a glycan conjugate of the invention.
[0186] In another aspect, the present invention provides a cancer vaccine comprising an immunogenic composition described herein and a pharmaceutically acceptable excipient.
[0187] The cancer vaccines of the invention may include a single dose or multiple doses of the inventive glycan conjugates, a glycan conjugate mixture thereof, or immunogenic compositions thereof. The provided cancer vaccines may be useful for treating or reducing the risk of cancers. The cancer vaccines may also include packaging information describing the use or prescribing information for the subject or a health care professional. Such information may be required by a regulatory agency such as the U.S. Food and Drug Administration (FDA). The cancer vaccine may also optionally include a device for administration of the compound or composition, for example, a syringe for parenteral administration.
[0188] Pharmaceutical Formulations
[0189] The immune composition is administered in a manner compatible with the dosage formulation, and in an amount that is therapeutically effective, protective and immunogenic. The quantity to be administered depends on the subject to be treated, including, for example, the capacity of the individual's immune system to synthesize antibodies, and if needed, to produce a cell-mediated immune response. Precise amounts of active ingredient required to be administered depend on the judgment of the practitioner. However, suitable dosage ranges are readily determinable by one skilled in the art. Suitable regimes for initial administration and booster doses are also variable, but may include an initial administration followed by subsequent administrations. The dosage of the vaccine may also depend on the route of administration and varies according to the size of the host.
[0190] The immune composition of this invention can also be used to generate antibodies in animals for production of antibodies, which can be used in both cancer treatment and diagnosis. Methods of making monoclonal and polyclonal antibodies and fragments thereof in animals (e.g., mouse, rabbit, goat, sheep, or horse) are well known in the art. See, for example, Harlow and Lane, (1988) Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, New York. The term "antibody" includes intact immunoglobulin molecules as well as fragments thereof, such as Fab, F(ab')2, Fv, scFv (single chain antibody), and dAb (domain antibody; Ward, et. al. (1989) Nature, 341, 544).
[0191] The compositions disclosed herein can be included in a pharmaceutical composition together with additional active agents, carriers, vehicles, excipients, or auxiliary agents identifiable by a person skilled in the art upon reading of the present disclosure.
[0192] The pharmaceutical compositions preferably comprise at least one pharmaceutically acceptable carrier. In such pharmaceutical compositions, the compositions disclosed herein form the "active compound," also referred to as the "active agent." As used herein the language "pharmaceutically acceptable carrier" includes solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like, compatible with pharmaceutical administration. Supplementary active compounds can also be incorporated into the compositions. A pharmaceutical composition is formulated to be compatible with its intended route of administration. Examples of routes of administration include parenteral, e.g., intravenous, intradermal, subcutaneous, oral (e.g., inhalation), transdermal (topical), transmucosal, and rectal administration. Solutions or suspensions used for parenteral, intradermal, or subcutaneous application can include the following components: a sterile diluent such as water for injection, saline solution, fixed oils, polyethylene glycols, glycerine, propylene glycol, or other synthetic solvents; antibacterial agents such as benzyl alcohol or methyl parabens; antioxidants such as ascorbic acid or sodium bisulfite; chelating agents such as ethylenediaminetetraacetic acid; buffers such as acetates, citrates, or phosphates and agents for the adjustment of tonicity such as sodium chloride or dextrose. pH can be adjusted with acids or bases, such as hydrochloric acid or sodium hydroxide. The parenteral preparation can be enclosed in ampoules, disposable syringes, or multiple dose vials made of glass or plastic.
[0193] Clinical Applications
[0194] The present invention provides glycan conjugates, immunogenic compositions or vaccines useful for the treatment of a proliferative disease such as cancer (e.g. lung cancer, large bowel cancer, pancreas cancer, biliary tract cancer, or endometrial cancer), benign neoplasm, or angiogenesis in a subject.
[0195] The immunogenic compositions or vaccines described herein can also be used to generate antibodies in human or animals for production of antibodies, which can be used in both cancer treatment and diagnosis. In some embodiments, the immunogenic compositions or vaccines described herein can also be used to generate the production of GloboH, SSEA3 and/or SSEA4 antibodies. Methods of making monoclonal and polyclonal antibodies and fragments thereof in human and/or animals (e.g., mouse, rabbit, goat, sheep, or horse) are well known in the art. See, for example, Harlow and Lane, (1988) Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, New York. The term "antibody" includes intact immunoglobulin molecules as well as fragments thereof, such as Fab, F(ab').sub.2, Fv, scFv (single chain antibody), and dAb (domain antibody; Ward, et. al. (1989) Nature, 341, 544).
[0196] Compositions comprising at least one anti-SSEA3/SSEA4/GloboH antibody or at least one polynucleotide comprising sequences encoding an anti-SSEA3/SSEA4/GloboH antibody are provided. In certain embodiments, a composition may be a pharmaceutical composition. As used herein, compositions comprise one or more antibodies that bind to one or more SSEA3/SSEA4/GloboH and/or one or more polynucleotides comprising sequences encoding one or more antibodies that bind to one or more SSEA3/SSEA4/GloboH. These compositions may further comprise suitable carriers, such as pharmaceutically acceptable excipients including buffers, which are well known in the art.
[0197] Isolated antibodies and polynucleotides are also provided. In certain embodiments, the isolated antibodies and polynucleotides are substantially pure.
[0198] In one embodiment, anti-SSEA3/SSEA4/GloboH antibodies are monoclonal. In another embodiment, fragments of the anti-SSEA3/SSEA4/GloboH antibodies (e.g., Fab, Fab'-SH and F(ab!)2 fragments) are provided. These antibody fragments can be created by traditional means, such as enzymatic digestion, or may be generated by recombinant techniques. Such antibody fragments may be chimeric, humanized, or human. These fragments are useful for the diagnostic and therapeutic purposes set forth below.
[0199] Pharmaceutical Formulations
[0200] Therapeutic formulations comprising an pharmaceutical agents of the invention are prepared for storage by mixing the antibody having the desired degree of purity with optional physiologically acceptable carriers, excipients or stabilizers (Remington's Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980)), in the form of aqueous solutions, lyophilized or other dried formulations. Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate, histidine and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g., Zn-protein complexes); and/or non-ionic surfactants such as TWEEN.TM., PLURONICS.TM. or polyethylene glycol (PEG).
[0201] The formulation herein may also contain more than one active compound as necessary for the particular indication being treated, including, but not limited to those with complementary activities that do not adversely affect each other. Such molecules are suitably present in combination in amounts that are effective for the purpose intended.
[0202] The active ingredients may also be entrapped in microcapsule prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-microcapsule and poly-(methylmethacylate) microcapsule, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions. Such techniques are disclosed in Remington's Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980).
[0203] The formulations to be used for in vivo administration must be sterile. This is readily accomplished by filtration through sterile filtration membranes.
[0204] Sustained-release preparations may be prepared. Suitable examples of sustained-release preparations include semipermeable matrices of solid hydrophobic polymers containing the immunoglobulin of the invention, which matrices are in the form of shaped articles, e.g., films, or microcapsule. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2-hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides (U.S. Pat. No. 3,773,919), copolymers of L-glutamic acid and .gamma. ethyl-L-glutamate, non-degradable ethylene-vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOT.TM. (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid. While polymers such as ethylene-vinyl acetate and lactic acid-glycolic acid enable release of molecules for over 100 days, certain hydrogels release proteins for shorter time periods. When encapsulated immunoglobulins remain in the body for a long time, they may denature or aggregate as a result of exposure to moisture at 37.degree. C., resulting in a loss of biological activity and possible changes in immunogenicity. Rational strategies can be devised for stabilization depending on the mechanism involved. For example, if the aggregation mechanism is discovered to be intermolecular S--S bond formation through thiol-disulfide interchange, stabilization may be achieved by modifying sulfhydryl residues, lyophilizing from acidic solutions, controlling moisture content, using appropriate additives, and developing specific polymer matrix compositions.
[0205] Pharmaceutical compositions of the invention can be used to treat, inhibit, delay progression of, prevent/delay recurrence of, ameliorate, or prevent diseases, disorders or conditions associated with abnormal expression and/or activity of SSEA3/SSEA4/GloboHs and SSEA3/SSEA4/GloboH related proteins, including but not limited to cancer, muscular disorders, ubiquitin-pathway-related genetic disorders, immune/inflammatory disorders, neurological disorders, and other ubiquitin pathway-related disorders.
[0206] In one aspect, a blocking antibody of the invention is specific for a SSEA3/SSEA4/GloboH.
[0207] Pharmaceutical compositions of the invention can be used either alone or in combination with other compositions in a therapy. For instance, an antibody of the invention may be co-administered with another antibody, and/or adjuvant/therapeutic agents (e.g., steroids). For instance, an antibody of the invention may be combined with an anti-inflammatory and/or antiseptic in a treatment scheme, e.g. in treating any of the diseases described herein, including cancer, muscular disorders, ubiquitin-pathway-related genetic disorders, immune/inflammatory disorders, neurological disorders, and other ubiquitin pathway-related disorders. Such combined therapies noted above include combined administration (where the two or more agents are included in the same or separate formulations), and separate administration, in which case, administration of the antibody of the invention can occur prior to, and/or following, administration of the adjunct therapy or therapies.
[0208] Pharmaceutical compositions of the invention (and adjunct therapeutic agent) can be administered by any suitable means, including parenteral, subcutaneous, intraperitoneal, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration. Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, or subcutaneous administration. In addition, the Pharmaceutical composition can be suitably administered by pulse infusion, particularly with declining doses of the antibody. Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic.
[0209] The location of the binding target of an antibody of the invention may be taken into consideration in preparation and administration of the antibody. When the binding target is an intracellular molecule, certain embodiments of the invention provide for the antibody or antigen-binding fragment thereof to be introduced into the cell where the binding target is located. In one embodiment, an antibody of the invention can be expressed intracellularly as an intrabody. The term "intrabody," as used herein, refers to an antibody or antigen-binding portion thereof that is expressed intracellularly and that is capable of selectively binding to a target molecule, as described in Marasco, Gene Therapy 4: 11-15 (1997); Kontermann, Methods 34: 163-170 (2004); U.S. Pat. Nos. 6,004,940 and 6,329,173; U.S. Patent Application Publication No. 2003/0104402, and PCT Publication No. WO2003/077945. Intracellular expression of an intrabody is effected by introducing a nucleic acid encoding the desired antibody or antigen-binding portion thereof (lacking the wild-type leader sequence and secretory signals normally associated with the gene encoding that antibody or antigen-binding fragment) into a target cell. Any standard method of introducing nucleic acids into a cell may be used, including, but not limited to, microinjection, ballistic injection, electroporation, calcium phosphate precipitation, liposomes, and transfection with retroviral, adenoviral, adeno-associated viral and vaccinia vectors carrying the nucleic acid of interest.
[0210] Pharmaceutical compositions of the invention would be formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The antibody need not be, but is optionally formulated with one or more agents currently used to prevent or treat the disorder in question. The effective amount of such other agents depends on the amount of antibodies of the invention present in the formulation, the type of disorder or treatment, and other factors discussed above. These are generally used in the same dosages and with administration routes as described herein, or about from 1 to 99% of the dosages described herein, or in any dosage and by any route that is empirically/clinically determined to be appropriate.
[0211] For the prevention or treatment of disease, the appropriate dosage of an Pharmaceutical compositions of the invention (when used alone or in combination with other agents such as chemotherapeutic agents) will depend on the type of disease to be treated, the type of antibody, the severity and course of the disease, whether the antibody is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the antibody, and the discretion of the attending physician. The antibody is suitably administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 .mu.g/kg to 15 mg/kg (e.g. 0.1 mg/kg-10 mg/kg) of antibody can be an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion. One typical daily dosage might range from about 1 .mu.g/kg to 100 mg/kg or more, depending on the factors mentioned above. For repeated administrations over several days or longer, depending on the condition, the treatment would generally be sustained until a desired suppression of disease symptoms occurs. One exemplary dosage of the antibody would be in the range from about 0.05 mg/kg to about 10 mg/kg. Thus, one or more doses of about 0.5 mg/kg, 2.0 mg/kg, 4.0 mg/kg or 10 mg/kg (or any combination thereof) may be administered to the patient. Such doses may be administered intermittently, e.g. every week or every three weeks (e.g. such that the patient receives from about two to about twenty, or e.g. about six doses of the antibody). An initial higher loading dose, followed by one or more lower doses may be administered. An exemplary dosing regimen comprises administering an initial loading dose of about 4 mg/kg, followed by a weekly maintenance dose of about 2 mg/kg of the antibody. However, other dosage regimens may be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
[0212] Articles of Manufacture
[0213] In another aspect of the invention, an article of manufacture containing materials useful for the treatment, prevention and/or diagnosis of the disorders described above is provided. The article of manufacture comprises a container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, etc. The containers may be formed from a variety of materials such as glass or plastic. The container holds a composition which is by itself or when combined with another composition effective for treating, preventing and/or diagnosing the condition and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). At least one active agent in the composition is an antibody of the invention. The label or package insert indicates that the composition is used for treating the condition of choice. Moreover, the article of manufacture may comprise (a) a first container with a composition contained therein, wherein the composition comprises an antibody of the invention; and (b) a second container with a composition contained therein, wherein the composition comprises a further cytotoxic or otherwise therapeutic agent. The article of manufacture in this embodiment of the invention may further comprise a package insert indicating that the compositions can be used to treat a particular condition. Alternatively, or additionally, the article of manufacture may further comprise a second (or third) container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
[0214] The following are examples of the methods and compositions of the invention. It is understood that various other embodiments may be practiced, given the general description provided above.
[0215] In some embodiments, the provided glycan conjugates, immunogenic compositions or vaccines are useful in treating, or diagnosing a cancer, including, but are not limited to, acoustic neuroma, adenocarcinoma, adrenal gland cancer, anal cancer, angiosarcoma (e.g., lymphangiosarcoma, lymphangioendotheliosarcoma, hemangiosarcoma), appendix cancer, benign monoclonal gammopathy, biliary cancer (e.g., cholangiocarcinoma), bladder cancer, breast cancer (e.g., adenocarcinoma of the breast, papillary carcinoma of the breast, mammary cancer, medullary carcinoma of the breast), brain cancer (e.g., meningioma; glioma, e.g., astrocytoma, oligodendroglioma; medulloblastoma), bronchus cancer, carcinoid tumor, cervical cancer (e.g., cervical adenocarcinoma), choriocarcinoma, chordoma, craniopharyngioma, colorectal cancer (e.g., colon cancer, rectal cancer, colorectal adenocarcinoma), epithelial carcinoma, ependymoma, endotheliosarcoma (e.g., Kaposi's sarcoma, multiple idiopathic hemorrhagic sarcoma), endometrial cancer (e.g., uterine cancer, uterine sarcoma), esophageal cancer (e.g., adenocarcinoma of the esophagus, Barrett's adenocarinoma), Ewing sarcoma, eye cancer (e.g., intraocular melanoma, retinoblastoma), familiar hypereosinophilia, gall bladder cancer, gastric cancer (e.g., stomach adenocarcinoma), gastrointestinal stromal tumor (GIST), head and neck cancer (e.g., head and neck squamous cell carcinoma, oral cancer (e.g., oral squamous cell carcinoma (OSCC), throat cancer (e.g., laryngeal cancer, pharyngeal cancer, nasopharyngeal cancer, oropharyngeal cancer)), hematopoietic cancers (e.g., leukemia such as acute lymphocytic leukemia (ALL) (e.g., B-cell ALL, T-cell ALL), acute myelocytic leukemia (AML) (e.g., B-cell AML, T-cell AML), chronic myelocytic leukemia (CML) (e.g., B-cell CML, T-cell CML), and chronic lymphocytic leukemia (CLL) (e.g., B-cell CLL, T-cell CLL); lymphoma such as Hodgkin lymphoma (HL) (e.g., B-cell HL, T-cell HL) and non-Hodgkin lymphoma (NHL) (e.g., B-cell NHL such as diffuse large cell lymphoma (DLCL) (e.g., diffuse large B-cell lymphoma (DLBCL)), follicular lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), mantle cell lymphoma (MCL), marginal zone B-cell lymphomas (e.g., mucosa-associated lymphoid tissue (MALT) lymphomas, nodal marginal zone B-cell lymphoma, splenic marginal zone B-cell lymphoma), primary mediastinal B-cell lymphoma, Burkitt lymphoma, lymphoplasmacytic lymphoma (i.e., "Waldenstrom's macroglobulinemia"), hairy cell leukemia (HCL), immunoblastic large cell lymphoma, precursor B-lymphoblastic lymphoma and primary central nervous system (CNS) lymphoma; and T-cell NHL such as precursor T-lymphoblastic lymphoma/leukemia, peripheral T-cell lymphoma (PTCL) (e.g., cutaneous T-cell lymphoma (CTCL) (e.g., mycosis fungiodes, Sezary syndrome), angioimmunoblastic T-cell lymphoma, extranodal natural killer T-cell lymphoma, enteropathy type T-cell lymphoma, subcutaneous panniculitis-like T-cell lymphoma, anaplastic large cell lymphoma); a mixture of one or more leukemia/lymphoma as described above; and multiple myeloma (MM)), heavy chain disease (e.g., alpha chain disease, gamma chain disease, mu chain disease), hemangioblastoma, inflammatory myofibroblastic tumors, immunocytic amyloidosis, kidney cancer (e.g., nephroblastoma a.k.a. Wilms' tumor, renal cell carcinoma), liver cancer (e.g., hepatocellular cancer (HCC), malignant hepatoma), lung cancer (e.g., bronchogenic carcinoma, small cell lung cancer (SCLC), non-small cell lung cancer (NSCLC), adenocarcinoma of the lung), leiomyosarcoma (LMS), mastocytosis (e.g., systemic mastocytosis), myelodysplastic syndrome (MDS), mesothelioma, myeloproliferative disorder (MPD) (e.g., polycythemia Vera (PV), essential thrombocytosis (ET), agnogenic myeloid metaplasia (AMM), a.k.a. myelofibrosis (MF), chronic idiopathic myelofibrosis, chronic myelocytic leukemia (CML), chronic neutrophilic leukemia (CNL), hypereosinophilic syndrome (HES)), neuroblastoma, neurofibroma (e.g., neurofibromatosis (NF) type 1 or type 2, schwannomatosis), neuroendocrine cancer (e.g., gastroenteropancreatic neuroendoctrine tumor (GEP-NET), carcinoid tumor), osteosarcoma, ovarian cancer (e.g., cystadenocarcinoma, ovarian embryonal carcinoma, ovarian adenocarcinoma), papillary adenocarcinoma, pancreatic cancer (e.g., pancreatic andenocarcinoma, intraductal papillary mucinous neoplasm (IPMN), islet cell tumors), penile cancer (e.g., Paget's disease of the penis and scrotum), pinealoma, primitive neuroectodermal tumor (PNT), prostate cancer (e.g., prostate adenocarcinoma), rectal cancer, rhabdomyosarcoma, salivary gland cancer, skin cancer (e.g., squamous cell carcinoma (SCC), keratoacanthoma (KA), melanoma, basal cell carcinoma (BCC)), small bowel cancer (e.g., appendix cancer), soft tissue sarcoma (e.g., malignant fibrous histiocytoma (MFH), liposarcoma, malignant peripheral nerve sheath tumor (MPNST), chondrosarcoma, fibrosarcoma, myxosarcoma), sebaceous gland carcinoma, sweat gland carcinoma, synovioma, testicular cancer (e.g., seminoma, testicular embryonal carcinoma), thyroid cancer (e.g., papillary carcinoma of the thyroid, papillary thyroid carcinoma (PTC), medullary thyroid cancer), urethral cancer, vaginal cancer and vulvar cancer (e.g., Paget's disease of the vulva). In certain embodiments, the provided glycan conjugates, immunogenic compositions or vaccines are useful for treating brain cancer, lung cancer, breast cancer, oral cancer, esophagus cancer, stomach cancer, liver cancer, bile duct cancer, pancreas cancer, colon cancer, kidney cancer, bone cancer, skin cancer, cervix cancer, ovary cancer, and prostate cancer.
[0216] To perform the treatment methods described herein, an effective amount of any of the glycan conjugates or immunogenic compositions or vaccines described herein may be administered to a subject in need of the treatment via a suitable route, as described above. The subject, such as a human subject, can be a patient having cancer, suspected of having cancer, or susceptible to cancer. The amount of the glycan conjugate or immunogenic composition administered to the subject may be effective in eliciting immune responses specific to the glycan moiety in the conjugate or composition. In some embodiments, the amount of the glycan conjugate or immunogenic composition is sufficient to elicit immune responses leading to the inhibition of cancer growth and/or reduction of tumor mass. In other embodiments, the amount of the glycan conjugate or immunogenic composition may be effective in delaying the onset of the target cancer or reducing the risk for developing the cancer. The exact amount of the provided glycan conjugates, immunogenic compositions or vaccines required to achieve an effective amount will vary from subject to subject, depending, for example, on species, age, and general condition of a subject, severity of the side effects or disorder, identity of the particular compound(s), mode of administration, and the like. The desired dosage can be delivered three times a day, two times a day, once a day, every other day, every third day, every week, every two weeks, every three weeks, or every four weeks. In certain embodiments, the desired dosage can be delivered using multiple administrations (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, or more administrations).
[0217] In certain embodiments, an effective amount of the provided glycan conjugates, immunogenic compositions or vaccines for administration one or more times a day to a 70 kg adult human may comprise about 0.0001 mg to about 3000 mg, about 0.0001 mg to about 2000 mg, about 0.0001 mg to about 1000 mg, about 0.001 mg to about 1000 mg, about 0.01 mg to about 1000 mg, about 0.1 mg to about 1000 mg, about 1 mg to about 1000 mg, about 1 mg to about 100 mg, about 10 mg to about 1000 mg, or about 100 mg to about 1000 mg, of a compound per unit dosage form.
[0218] In certain embodiments, the provided glycan conjugates, immunogenic compositions or vaccines may be administered orally or parenterally at dosage levels sufficient to deliver from about 0.001 mg/kg to about 100 mg/kg, from about 0.01 mg/kg to about 50 mg/kg, preferably from about 0.1 mg/kg to about 40 mg/kg, preferably from about 0.5 mg/kg to about 30 mg/kg, from about 0.01 mg/kg to about 10 mg/kg, from about 0.1 mg/kg to about 10 mg/kg, and more preferably from about 1 mg/kg to about 25 mg/kg, of subject body weight per day, one or more times a day, to obtain the desired therapeutic effect.
[0219] It will be appreciated that dose ranges as described herein provide guidance for the administration of the provided glycan conjugates, immunogenic compositions or vaccines to an adult. The amount to be administered to, for example, a child or an adolescent can be determined by a medical practitioner or person skilled in the art and can be lower or the same as that administered to an adult.
[0220] It will be also appreciated that the provided glycan conjugates, immunogenic compositions or vaccines can be administered in combination with one or more additional therapeutically active agents. The provided glycan conjugates, immunogenic compositions or vaccines can be administered in combination with additional therapeutically active agents that improve their bioavailability, reduce and/or modify their metabolism, inhibit their excretion, and/or modify their distribution within the body. It will also be appreciated that the therapy employed may achieve a desired effect for the same disorder, and/or it may achieve different effects.
[0221] The provided glycan conjugates, immunogenic compositions or vaccines can be administered concurrently with, prior to, or subsequent to, one or more additional therapeutically active agents. In general, each agent will be administered at a dose and/or on a time schedule determined for that agent. In will further be appreciated that the additional therapeutically active agent utilized in this combination can be administered together in a single composition or administered separately in different compositions. The particular combination to employ in a regimen will take into account compatibility of the inventive compound with the additional therapeutically active agent and/or the desired therapeutic effect to be achieved. In general, it is expected that additional therapeutically active agents utilized in combination be utilized at levels that do not exceed the levels at which they are utilized individually. In some embodiments, the levels utilized in combination will be lower than those utilized individually.
[0222] In certain embodiments, the provided glycan conjugate, immunogenic composition or vaccine is administered in combination with one or more additional pharmaceutical agents described herein. In certain embodiments, the additional pharmaceutical agent is an anti-cancer agent. Anti-cancer agents encompass biotherapeutic anti-cancer agents as well as chemotherapeutic agents.
[0223] Exemplary biotherapeutic anti-cancer agents include, but are not limited to, interferons, cytokines (e.g., tumor necrosis factor, interferon a, interferon y), vaccines, hematopoietic growth factors, monoclonal serotherapy, immunostimulants and/or immunodulatory agents (e.g., IL-1, 2, 4, 6, or 12), immune cell growth factors (e.g., GM-CSF) and antibodies (e.g. Herceptin (trastuzumab), T-DM1, AVASTIN (bevacizumab), ERBITUX (cetuximab), Vectibix (panitumumab), Rittman (rituximab), Bexxar (tositumomab)).
[0224] Exemplary chemotherapeutic agents include, but are not limited to, anti-estrogens (e.g. tamoxifen, raloxifene, and megestrol), LHRH agonists (e.g. goscrclin and leuprolide), anti-androgens (e.g. flutamide and bicalutamide), photodynamic therapies (e.g. vertoporfin (BPD-MA), phthalocyanine, photosensitizer Pc4, and demethoxy-hypocrellin A (2BA-2-DMHA)), nitrogen mustards (e.g. cyclophosphamide, ifosfamide, trofosfamide, chlorambucil, estramustine, and melphalan), nitrosoureas (e.g. carmustine (BCNU) and lomustine (CCNU)), alkylsulphonates (e.g. busulfan and treosulfan), triazenes (e.g. dacarbazine, temozolomide), platinum containing compounds (e.g. cisplatin, carboplatin, oxaliplatin), vinca alkaloids (e.g. vincristine, vinblastine, vindesine, and vinorelbine), taxoids (e.g. paclitaxel or a paclitaxel equivalent such as nanoparticle albumin-bound paclitaxel (Abraxane), docosahexaenoic acid bound-paclitaxel (DHA-paclitaxel, Taxoprexin), polyglutamate bound-paclitaxel (PG-paclitaxel, paclitaxel poliglumex, CT-2103, XYOTAX), the tumor-activated prodrug (TAP) ANG1005 (Angiopep-2 bound to three molecules of paclitaxel), paclitaxel-EC-1 (paclitaxel bound to the erbB2-recognizing peptide EC-1), and glucose-conjugated paclitaxel, e.g., 2'-paclitaxel methyl 2-glucopyranosyl succinate; docetaxel, taxol), epipodophyllins (e.g. etoposide, etoposide phosphate, teniposide, topotecan, 9-aminocamptothecin, camptoirinotecan, irinotecan, crisnatol, mytomycin C), anti-metabolites, DHFR inhibitors (e.g. methotrexate, dichloromethotrexate, trimetrexate, edatrexate), IMP dehydrogenase inhibitors (e.g. mycophenolic acid, tiazofurin, ribavirin, and EICAR), ribonuclotide reductase inhibitors (e.g. hydroxyurea and deferoxamine), uracil analogs (e.g. 5-fluorouracil (5-FU), floxuridine, doxifluridine, ratitrexed, tegafur-uracil, capecitabine), cytosine analogs (e.g. cytarabine (ara C), cytosine arabinoside, and fludarabine), purine analogs (e.g. mercaptopurine and Thioguanine), Vitamin D3 analogs (e.g. EB 1089, CB 1093, and KH 1060), isoprenylation inhibitors (e.g. lovastatin), dopaminergic neurotoxins (e.g. 1-methyl-4-phenylpyridinium ion), cell cycle inhibitors (e.g. staurosporine), actinomycin (e.g. actinomycin D, dactinomycin), bleomycin (e.g. bleomycin A2, bleomycin B2, peplomycin), anthracycline (e.g. daunorubicin, doxorubicin, pegylated liposomal doxorubicin, idarubicin, epirubicin, pirarubicin, zorubicin, mitoxantrone), MDR inhibitors (e.g. verapamil), Ca2+ ATPase inhibitors (e.g. thapsigargin), imatinib, thalidomide, lenalidomide, tyrosine kinase inhibitors (e.g., axitinib (AG013736), bosutinib (SKI-606), cediranib (RECENTIN.TM., AZD2171), dasatinib (SPRYCEL.RTM., BMS-354825), erlotinib (TARCEVA.RTM.), gefitinib (IRESSA.RTM.), imatinib (Gleevec.RTM., CGP57148B, STI-571), lapatinib (TYKERB.RTM., TYVERB.RTM.), lestaurtinib (CEP-701), neratinib (HKI-272), nilotinib (TASIGNA.RTM.), semaxanib (semaxinib, SU5416), sunitinib (SUTENT.RTM., SU11248), toceranib (PALLADIA.RTM.), vandetanib (ZACTIMA.RTM., ZD6474), vatalanib (PTK787, PTK/ZK), trastuzumab (HERCEPTIN.RTM.), bevacizumab (AVASTIN.RTM.), rituximab (RITUXAN.RTM.), cetuximab (ERBITUX.RTM.), panitumumab (VECTIBIX.RTM.), ranibizumab (Lucentis.RTM.), nilotinib (TASIGNA.RTM.), sorafenib (NEXAVAR.RTM.), everolimus (AFINITOR.RTM.), alemtuzumab (CAMPATH.RTM.), gemtuzumab ozogamicin (MYLOTARG.RTM.), temsirolimus (TORISEL.RTM.), ENMD-2076, PCI-32765, AC220, dovitinib lactate (TKI258, CHIR-258), BIBW 2992 (TOVOKTM), SGX523, PF-04217903, PF-02341066, PF-299804, BMS-777607, ABT-869, MP470, BIBF 1120 (VARGATEF.RTM.), AP24534, JNJ-26483327, MGCD265, DCC-2036, BMS-690154, CEP-11981, tivozanib (AV-951), OSI-930, MM-121, XL-184, XL-647, and/or XL228), proteasome inhibitors (e.g., bortezomib (Velcade)), mTOR inhibitors (e.g., rapamycin, temsirolimus (CCI-779), everolimus (RAD-001), ridaforolimus, AP23573 (Ariad), AZD8055 (AstraZeneca), BEZ235 (Novartis), BGT226 (Norvartis), XL765 (Sanofi Aventis), PF-4691502 (Pfizer), GDC0980 (Genetech), SF1126 (Semafoe) and OSI-027 (OSI)), oblimersen, gemcitabine, carminomycin, leucovorin, pemetrexed, cyclophosphamide, dacarbazine, procarbizine, prednisolone, dexamethasone, campathecin, plicamycin, asparaginase, aminopterin, methopterin, porfiromycin, melphalan, leurosidine, leurosine, chlorambucil, trabectedin, procarbazine, discodermolide, carminomycin, aminopterin, and hexamethyl melamine.
[0225] In certain embodiments, the subject being treated is a mammal. In certain embodiments, the subject is a human. In certain embodiments, the subject is a domesticated animal, such as a dog, cat, cow, pig, horse, sheep, or goat. In certain embodiments, the subject is a companion animal such as a dog or cat. In certain embodiments, the subject is a livestock animal such as a cow, pig, horse, sheep, or goat. In certain embodiments, the subject is a zoo animal. In another embodiment, the subject is a research animal such as a rodent, dog, or non-human primate. In certain embodiments, the subject is a non-human transgenic animal such as a transgenic mouse or transgenic pig.
EXAMPLES
[0226] The following examples are included to demonstrate preferred embodiments of the invention. It should be appreciated by those of skill in the art that the techniques disclosed in the examples which follow represent techniques discovered by the inventor to function well in the practice of the invention, and thus can be considered to constitute preferred modes for its practice. However, those of skill in the art should, in light of the present disclosure, appreciate that many changes can be made in the specific embodiments which are disclosed and still obtain a like or similar result without departing from the spirit and scope of the invention.
Example 1
Exemplary Syntheses of SSEA3 Analogues
[0227] A: Chemo-enzymatic synthesis of SSEA3 analog-NH2
##STR00015##
[0228] The combined compounds Gb4 analog, ATP, UTP, galactose analog, phosphoenolpyruvate, MgCl.sub.2 with enzymes galactokinase (GalK), UDP-sugar pyrophosphorylase (AtUSP), beta-1,3-galactosyltransferase (LgtD,), pyruvate kinase (PK), and inorganic pyrophosphatase (PPA) in the solution, and the reaction was initiated at room temperature with the pH controlled at 7.0, and the reaction was monitored by TLC until no more product could be observed. After completion of the reaction, the proteins in the reaction mixture were removed by heating for 30 min followed by centrifugation and filtration with 0.22 .quadrature.M filter. The filtrate was then purified by C-18 gel chromatography. Fractions were collected and monitored by TLC.
Example 2
Exemplary Syntheses of SSEA4 Analogues
[0229] A : Chemical Synthesis of SSEA4-Gc-NH2
##STR00016## ##STR00017##
[0230] Powdered molecular sieves (4 A, 0.5 g) was added to a solution of acceptor 3 (93 mg, 0.045 mmol) and imidates 6 (76 mg, 0.068 mmol) in 6 mL of dichloromethane (CH2C12). The mixture was stirred at room temperature for 2 hrs. After cooled to -10.degree. C., TMSOTf (5 .mu.L, 0.03 mmol) was added, and the mixture was stirred at 5.degree. C. (cold room) overnight. The reaction mixture was quenched by the addition of triethylamine (0.5 mL), diluted with CH.sub.2Cl.sub.2 and filtered through a pad of celite. The filtrate was washed with saturated sodium bicarbonate (NaHCO.sub.3) aqueous solution, dried over sodium sulfate (Na.sub.2SO.sub.4), filtered, and concentrated. The residue was purified by flash silica gelchromatography (50-100% EtOAc in Hexane) to afford hexasaccharide 7 contaminated with impurities from disaccharide imidates 6. The yield was determined by NMR (90 mg, 68%).
[0231] Zinc dust (1g) was added to a solution of hexasaccharide 7 (90 mg, 0.03 mmol) in glacial acetic acid (5.0 mL) and the mixture was stirred for 1-2 hrs, until compound 7 was consumed by TLC analysis. The reaction mixture was diluted with CH2C12, filtered through a pad of celite, and concentrated under reduced pressure. The residue was dissolved in pyridine/Ac.sub.2O (1:1, 2.0 mL), stirred for 1 h, and concentrated. The residue was purified by flash silica get chromatography. The acylated material was dissolved in anhydrous CH.sub.2Cl.sub.2 and MeOH (2:8, 10 mL) and treated with NaOMe (45 mg). After stirring at room temperature for 4 hrs, water (0.2 mL) was added, and the resulting mixture was stirred for 16 hrs. The reaction mixture was neutralized with amberlyst IR-120, filtered, and concentrated. The residue was purified by reverse phase chromatography (RP-18).
[0232] Palladium hydroxide (20% in Charcoal, 50 mg) was added to the adduct in a mixture of methanol/water/ Acetic acid (10:10:0.5, 6 mL) and the reaction mixture was stirred at room temperature under a positive pressure of hydrogen for 16 hrs. The reaction mixture was filtered through a pad of celite and concentrated. The residue was purified by reverse phase chromatography to afford 8 (17 mg, 43%).
[0233] B: Chemoenzymatic Synthesis of SSEA4 analog-NH2
##STR00018##
[0234] SSEA4 analogs-NH2 were synthesized via enzymatic regeneration strategy as described in Scheme 3. In this system, ManNAc derivatives were reacted with pyruvate and transformed into Neu5Ac analogs by aldolase catalysis, followed by incorporation with Gb5-NH2 in the regeneration system (J. Am. Chem. Soc. 2013, 135, 14831-14839) to obtain the exemplary SSEA4 analogs-NH2.
[0235] Detail of the reaction condition is described as follows: Gb5-NH2 (18 .mu.mol), CTP (5 .mu.mol), ManNAc derivative (27 .mu.mol), sodium pyruvate (81 .mu.mol), PEP (55 .mu.mol), and ATP (5 .mu.mol), were dissolved in 50 mM Tris-HCl buffer (pH 8.0). Enzymes alpha-(2,3)-sialyltransferase (20 units), sialic acid aldolase(20 units) CMK (10 units), Pykf (10 units), PPA (10 units), and Pmcss (10 units) were added to the solution, and the reaction was incubated at 37.degree. C. for 8 hours and monitored by TLC plate. At the end of reaction, enzyme was denatured by heating at 100.degree. C. for 5 minutes. The desired SSEA4 analog-NH2 was purified by G25, DEAE, and SP column (80%).
[0236] .sup.1H NMR of SSEA4analogs-NH2
[0237] B-1. SSEA4-pentylamine (RN=NHAc, R10=OH)
[0238] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.94 (d, J=3.8 Hz, 1H), 4.72 (d, J=8.5 Hz, 1H), 4.54-4.50 (m, 3H), 4.40 (t, J=6.4 Hz, 1H), 4.27 (d, J=2.0 Hz, 1H), 4.20 (d, J=2.8 Hz, 1H), 4.10-3.54 (m, 37 H), 3.34-3.31 (m, 1H), 3.02 (t, J=7.6 Hz, 2H), 2.78 (dd, J=12.4, 4.6 Hz, 1H), 2.05 (m, 6H), 1.80 (t, 12.2 Hz, 1H), 1.74-1.67 (m, 4H), 1.51-1.45 (m, 2H)
[0239] B-2. Neu5Gc_SSEA4-pentylamine (RN=NHGc, R10=OH)
[0240] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.89 (d, J=3.6 Hz, 1H), 4.66 (d, J=8.2 Hz, 1H), 4.52-4.45 (m, 3H), 4.37 (t, J=6.8 Hz, 1H), 4.23 (d, J=3.2 Hz, 1H), 4.15 (d, J=2.8 Hz, 1H), 4.10-3.48 (m, 35 H), 3.27 (m, 1H), 2.98 (t, J=7.6 Hz, 2H), 2.73 (dd, J=4.8, 12.4 Hz, 1H), 2.00 (s, 3H), 1.77 (t, J=12.0 Hz, 1H), 1.72-1.61 (m, 4H), 1.48-1.39 (m, 2 H).
[0241] B-3. Ac-Alkynyl_SSEA4-pentylamine (RN=NHCOC.sub.2H.sub.4C.ident.CH, R10=OH)
[0242] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.89 (d, J=4.0 Hz, 1H), 4.67 (d, J=8.4 Hz, 1H), 4.52-4.45 (m, 3H), 4.37 (t, J=6.4 Hz, 1H), 4.23 (d, J=2.4 Hz, 1H), 4.08-3.54 (m, 38 H), 3.28 (m, 1H), 2.99 (t, J=7.6 Hz, 2H), 2.53-2.4 (m, 4H), 2.37 (s, 1H), 2.01 (s, 3H), 1.77 (t, J=12.0 Hz, 1H), 1.72-1.62 (m, 4H), 1.49-1.41 (m, 2 H).
[0243] B-4. Ac-Fluoride_SSEA4-pentylamine (RN=NHCOCH.sub.2F, R10=OH)
[0244] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.90 (d, J=46.4 Hz, 2H), 4.90 (d, J=4.0 Hz, 1 H), 4.67 (d, J=8.8 Hz, 1H), 4.53-4.46 (m, 3H), 4.37 (t, J=6.8 Hz, 1 H), 4.24 (d, J=2.8 Hz, 2H), 4.16 (d, J=3.2 Hz, 1 H), 4.09-3.51 (m, 34H), 3.28 (m, 1H), 2.99 (t, J=7.2 Hz, 1H), 2.75 (dd, J=4.8, 12.4 Hz, 1H), 2.01 (s, 3H), 1.79 (t, J=12.0 Hz, 1H), 1.72-1.62 (m, 4H), 1.48-1.40 (m, 2 H).
[0245] B-5. Ac-Phenyl _SSEA4-pentylamine (RN=NHCOCH.sub.2Ph, R10=OH)
[0246] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.39-7.30 (m, 5H), 4.90 (d, J=4.0 Hz, 1H), 4.66 (d, J=8.4 Hz, 1H), 4.52-4.46 (m, 3H), 4.37 (t, J=6.8 Hz, 1H), 4.23 (d, J=2.8 Hz, 1H), 4.15 (d, J=3.2 Hz, 1H), 4.08-3.47 (m, 38H), 3.36(dd, J=1.6, 9.2 Hz, 1H), 3.28 (m, 1H), 2.99 (t, J=7.6 Hz, 2H), 2.73 (dd, J=4.8, 12.4 Hz, 1H), 2.00 (s, 3H), 1.76 (t, J=12.0 Hz, 1H), 1.72-1.61 (m, 4H), 1.51-1.40 (m, 2 H).
[0247] B-6. Ac-Azido_SSEA4-pentylamine (RN=NHCOCH.sub.2N.sub.3, R10=OH)
[0248] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.88 (d, J=3.6 Hz, 1H), 4.66 (d, J=8.4 Hz, 1H), 4.52-4.44 (m, 3H), 4.36 (t, J=6.4 Hz, 1H), 4.23 (d, J=2.4 Hz, 1H), 4.08-3.54 (m, 35 H), 3.27 (m, 1H), 2.98 (t, J=7.2 Hz, 2H), 2.73 (dd, J=4.8, 12.4 Hz, 1H), 2.00 (s, 3H), 1.77 (t, J=12.4 Hz, 1H), 1.72-1.60 (m, 4H), 1.48-1.39 (m, 2 H).
[0249] B-7. 5'-Azido_SSEA4-pentylamine (RN=N.sub.3, R10=OH)
[0250] .sup.1H NMR (400 MHz, D.sub.2O): .delta. 4.90 (d, J=3.6 Hz, 1H), 4.67 (d, J=8.4 Hz, 1H), 4.51-4.47 (m, 3H), 4.37 (t, J=6.4 Hz, 1H), 4.23 (d, J=2.8 Hz, 1H), 4.15 (d, J=3.2 Hz, 1H), 4.08-3.44 (m, 35H), 3.31-3.27 (m, 1H), 2.99 (t, J=7.2 Hz, 1H), 2.73 (dd, J=4.8, 12.4 Hz, 1H), 2.01 (s, 3H), 1.76 (t, J=12.0 Hz, 1H), 1.72-1.63 (3, 4H), 1.48-1.41 (m, 2H); HRMS (ESI-TOF, M-H-) C.sub.46H.sub.78N.sub.5O.sub.33- calcd for 1228.4579, found 1228.4621.
[0251] B-8. 9'-Azido_SSEA4-pentylamine (RN=NHAc, R10=N.sub.3)
[0252] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.85 (d, J=3.8 Hz, 1H), 4.67 (d, J=8.4 Hz, 1H), 4.51-4.44 (m, 3H), 4.37 (t, J=6.4 Hz, 1H), 4.23 (d, J=2.8 Hz, 1H), 4.10-3.40 (m, 33 H), 3.27 (m, 1H), 2.98 (t, J=7.6 Hz, 2H), 2.72 (dd, J=4.8, 12.8 Hz, 1H), 2.00 (s, 3H), 2.00 (s, 3H), 1.75 (t, J=12.4 Hz, 1H), 1.72-1.60 (m, 4H), 1.58-1.38 (m, 2 H).
[0253] B-9. NHBz_SSEA4-pentylamine (RN=NHBz, R10=OH)
[0254] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.80-7.73 (m, 2H), 7.63 (m, 1H), 7.56-7.51 (m, 2H), 4.92 (d, J=4.0 Hz, 1H), 4.70 (d, J=8.4 Hz, 1H), 4.58-4.47 (m, 3H), 4.40 (t, J=6.4 Hz, 1H), 4.26 (d, J=2.8 Hz, 1H), 4.19 (d, J=3.2 Hz, 1H), 4.15-3.53 (m, 36H), 3.31 (m, 1H), 3.01 (t, J=7.6 Hz, 2H), 2.82 (dd, J=4.4, 12.4 Hz, 1H), 2.00 (s, 3H), 1.87 (t, J=12.0 Hz, 1H), 1.72-1.60 (m, 4H), 1.48-1.39 (m, 2 H).
[0255] C: Cross-Linking Reaction for SSEA4 Analog-SH
##STR00019##
[0256] In certain embodiments, DTSSP (2.0 eq) and SSEA4 analog-NH2 (1.0 eq) was mixed in 0.1 M phosphate buffer, pH 7.4 (.about.3 mg/ml). The solution was stirred at room temperature for overnight. Then the reaction mixture was warmed to 40.degree. C. and added with DTT (9.0 eq). After stirring for 1.5 hrs at 40.degree. C., the reaction mixture was concentrated in vacuo, and the residue was purified by LH-20 column to afford a white solid SSEA4 analogs-SH. (Scheme 4)
[0257] .sup.1H NMR of SSEA4 analogs-SH
[0258] C-1: SSEA4-SH (RN=NHAc, R10=OH)
[0259] .sup.1H NMR (400 Hz, D.sub.2O) .delta. 4.88 (d, J=4.0 Hz, 1H), 4.65 (d, J=8.5 Hz, 1H), 4.50-4.44 (m, 3H), 4.36 (t, J=6.5 Hz, 1H), 4.22 (d, J=2.9 Hz, 1H), 4.14 (d, J=3.1 Hz, 1H), 4.04-3.55 (m, 35H), 3.26 (t, J=8.5 Hz, 1H), 3.18 (t, J=6.8 Hz, 2H), 2.74-2.70 (m, 3H), 2.49 (t, J=6.8 Hz, 2H), 1.994 (s, 3H), 1.992 (s, 3H), 1.75 (t, J=12.2 Hz, 1H), 1.61 (tt, J=6.7, 6.7 HZ, 2H), 1.52 (tt, J=7.1, 7.1 Hz, 2H), 1.40-1.36 (m, 2H);
[0260] C-2: Neu5Gc_SSEA4-SH (RN=NHGc, R10=OH)
[0261] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.89 (d, J=3.9 Hz, 1H), 4.66 (d, J=8.5 Hz, 1H), 4.53- 4.43 (m, 3H), 4.36 (t, J=6.5 Hz, 1H), 4.22 (d, J=3.0 Hz, 1H), 4.15 (d, J=3.1 Hz, 1H), 4.11-3.48 (m, 38H), 3.27 (t, J=8.4 Hz, 1H), 3.19 (t, J=6.7 Hz, 2H), 2.78-2.71 (m, 3H), 2.51 (t, J=6.7 Hz, 2H), 2.00 (s, 3H), 1.78 (t, J=12.1 Hz, 1H), 1.61 (q, J=7.1 Hz, 2H), 1.52 (q, J=7.1 Hz, 2H), 1.39 (q, J=8.0 Hz, 2H).
[0262] C-3: Ac-Alkynyl_SSEA4-SH (RN=NHCOC.sub.2H.sub.4CCH, R10=OH)
[0263] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.94 (d, J=3.9 Hz, 1H), 4.72 (d, J=8.4 Hz, 1H), 4.58- 4.48 (m, 3H), 4.41 (t, J=6.5 Hz, 1H), 4.30-4.26 (m, 1H), 4.21 (d, J=3.1 Hz, 1H), 4.14 -3.54 (m, 37H), 3.32 (t, J=8.6 Hz, 1H), 3.24 (t, J=6.8 Hz, 2H), 2.83-2.74 (m, 3H), 2.59-2.49 (m, 5H), 2.43 (s, 1H), 2.06 (s, 3H), 1.82 (t, J=12.1 Hz, 1H), 1.67 (p, J=6.9 Hz, 2H), 1.58 (p, J=6.9 Hz, 2H), 1.48-1.38 (m, 2H).
[0264] C-4: Ac-Fluoride_SSEA4-SH (RN=NHCOCH.sub.2F, R10=OH)
[0265] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.90 (d, J=46.4 Hz, 2H), 4.95 (d, J=4.0 Hz, 1H), 4.72 (d, J=8.5 Hz, 1H), 4.59-4.48 (m, 3H), 4.41 (t, J=6.6 Hz, 1H), 4.31- 4.26 (m, 1H), 4.23 -4.18 (m, 1H), 4.14-3.54 (m, 36H), 3.36-3.29 (m, 1H), 3.25 (t, J=6.8 Hz, 2H), 2.80 (m, 3H), 2.57 (t, J=6.7 Hz, 2H), 2.06 (s, 3H), 1.84 (t, J=12.2 Hz, 1H), 1.67 (p, J=6.9 Hz, 2H), 1.58 (p, J=7.0 Hz, 2H), 1.43 (q, J=8.3 Hz, 2H).
[0266] C-5: Ac-Phenyl_SSEA4-SH (RN=NHCOCH.sub.2Ph, R10=OH)
[0267] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.48-7.32 (m, 5H), 4.94 (d, J=3.6 Hz, 1H), 4.73-4.68 (d, J=8.4 Hz, 1H), 4.52 (m, 3H), 4.41 (t, J=6.4 Hz, 1H), 4.29-4.26 (m, 1H), 4.20 (d, J=3.0 Hz, 1H), 4.13-3.51 (m, 37H), 3.39 (dd, J=9.0, 1.8 Hz, 1H), 3.32 (t, J=8.6 Hz, 1H), 3.25 (t, J=6.7 Hz, 2H), 2.83-2.74 (m, 3H), 2.56 (t, J=6.7 Hz, 2H), 2.04 (s, 3H), 1.80 (t, J=12.1 Hz, 1H), 1.67 (q, J=7.2 Hz, 2H), 1.57 (q, J=7.1 Hz, 2H), 1.48-1.38 (m, 2H).
[0268] C-6: Ac-Azido_SSEA4-SH (RN=NHCOCH.sub.2N.sub.3, R10=OH)
[0269] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.88 (d, J=3.9 Hz, 1H), 4.66 (d, J=8.5 Hz, 1H), 4.52- 4.43 (m, 3H), 4.36 (t, J=6.5 Hz, 1H), 4.22 (d, J=3.1 Hz, 1H), 4.14 (d, J=3.1 Hz, 1H), 4.08-3.47 (m, 38H), 3.26 (t, J=8.4 Hz, 1H), 3.19 (t, J=6.8 Hz, 2H), 2.74 (m, 3H), 2.51 (t, J=6.7 Hz, 2H), 2.00 (s, 3H), 1.76 (t, J=12.1 Hz, 1H), 1.61 (q, J=7.1 Hz, 2H), 1.53 (p, J=7.0 Hz, 2H), 1.38 (q, J=8.3 Hz, 2H).
[0270] C-7: 5'-Azido_SSEA4-SH (RN=N.sub.3, R10=OH)
[0271] .sup.1H NMR (400 Hz, D.sub.2O) .delta. 4.90 (d, J=4.0 Hz, 1H), 4.67 (d, J=8.4 Hz, 1H), 4.51-4.46 (m, 3H), 4.37 (t, J=6.4 Hz, 1H), 4.24 (d, J=2.8 Hz, 1H), 4.15 (d, J=2.8 Hz, 1H), 4.01-3.44 (m, 35H), 3.28 (t, J=8.4 Hz, 1H), 3.21 (t, J=6.8 Hz, 2H), 2.78-2.72 (m, 3H), 2.52 (t, J=7.2 Hz, 2H), 2.02 (s, 3H), 1.77 (t, J=12.0 Hz, 1H), 1.67-1.60 (m, 2H), 1.58-1.50 (m, 2H), 1.43-1.37 (m, 2H)
[0272] C-8: 9'-Azido_SSEA4-SH (RN=NHAc, R10=N3)
[0273] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 4.90 (d, J=3.9 Hz, 1H), 4.68 (d, J=8.5 Hz, 1H), 4.48 (dd, J=13.2, 7.9 Hz, 3H), 4.37 (t, J=6.5 Hz, 1H), 4.26-4.22 (m, 1H), 4.16 (d, J=3.3 Hz, 1H), 4.09-3.44 (m, 36H), 3.31-3.24 (m, 1H), 3.20 (t, J=6.8 Hz, 2H), 2.79-2.70 (m, 3H), 2.52 (t, J=6.7 Hz, 2H), 2.02 (d, J=2.0 Hz, 6H), 1.76 (t, J=12.1 Hz, 1H), 1.63 (p, J=6.9 Hz, 2H), 1.54 (p, J=6.9 Hz, 2H), 1.39 (q, J=8.3 Hz, 2H).
[0274] C-9: NHBz_SSEA4-SH (RN=NHBz, R10=OH)
[0275] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.80-7.73 (m, 2H), 7.66-7.58 (m, 1H), 7.52 (dd, J=8.4, 7.0 Hz, 2H), 4.91 (d, J=3.9 Hz, 1H), 4.69 (d, J=8.5 Hz, 1H), 4.57-4.44 (m, 3H), 4.38 (t, J=6.5 Hz, 1H), 4.27-4.22 (m, 1H), 4.18 (d, J=3.1 Hz, 1H), 4.16-3.52 (m, 36H), 3.29 (t, J=8.5 Hz, 1H), 3.20 (t, J=6.8 Hz, 2H), 2.84-2.72 (m, 3H), 2.52 (t, J=6.7 Hz, 2H), 2.03 (s, 3H), 1.89 (t, J=12.2 Hz, 1H), 1.63 (p, J=6.8 Hz, 2H), 1.53 (q, J=7.1 Hz, 2H), 1.40 (q, J=8.2 Hz, 2H).
[0276] D: Chemoenzymatic Synthesis of SSEA4 analog-allyl
##STR00020##
[0277] SSEA4 analogs-allyl were synthesized via enzymatic regeneration strategy as described in Scheme5. In this system, ManNAc derivatives were reacted with pyruvate and transformed into NeuSAc analogs by aldolase catalysis, followed by incorporation with Gb5-ally in the regeneration system (J. Am. Chem. Soc. 2013, 135, 14831-14839) to obtain the exemplary SSEA4 analogs-allyl. (Scheme 5)
[0278] Detail of the reaction condition is described as follows: Gb5-allyl (18 .mu.mol), CTP (5 .mu.mop, ManNAc derivative (27 .mu.mol), sodium pyruvate (81 .mu.mol), PEP (55 .mu.mol), and ATP (5 .mu.mol), were dissolved in 50 mM Tris-HCl buffer (pH 8.0). Enzymesalpha-(2,3)-sialyltransferase (20 units), sialic acid aldolase(20 units) CMK (10 units), Pykf (10 units), PPA (10 units), and Pmcss (10 units) were added to the solution, and the reaction was incubated at 37.degree. C. for 8 hours and monitored by TLC plate. At the end of reaction, enzyme was denatured by heating at 100.degree. C. for 5 minutes. The desired SSEA4-analog-allyl was purified by G25, DEAE, and SP column (80%).
[0279] .sup.1H NMR of SSEA4 analogs-allyl
[0280] D-1. SSEA4-allyl (R1=OH, RN=NHAc, R10=OH)
[0281] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 6.00 (m, 1H), 5.40-5.37 (d, J=17.3 Hz, 1H), 5.30-5.28 (d, J=10.4 Hz, 1H), 4.92 (d, J=3.9 Hz, 1H), 4.70 (d, J=8.5 Hz, 1H), 4.54-4.51 (m, 3H), 4.40-4.38 (m, 2H), 4.25-4.18 (m, 3H), 4.10-3.52 (m, 34 H), 3.35-3.32 (t, J=8.6 Hz, 1H), 2.77 (dd, J=12.5, 4.6 Hz, 1H), 2.03 (s, 6H), 1.80 (t, J=12.1 Hz, 1H)
[0282] D-2. Neu5Gc_SSEA4-allyl (R1=OH, RN=NHGc, R10=OH)
[0283] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.99 (m, 1H), 5.38 (dd, J=1.2, 17.2 Hz, 1H), 5.29 (dd, J=1.2, 10.0 Hz, 1H), 4.93 (d, J=4.0 Hz, 1H), 4.69 (d, J=8.4 Hz, 1H), 4.58-4.51 (m, 3H), 4.43-4.37 (m, 2H), 4.28-4.17 (m, 3H), 4.14-3.52 (m, 34 H), 3.33 (t, J=8.8 Hz, 1H), 2.77 (dd, J=4.8, 12.4 Hz, 1H), 2.03 (s, 3H), 1.81 (t, J=12.0 Hz, 1H).
[0284] D-3. Ac-Fluoride_SSEA4-allyl (R1=OH, RN=NHCOCH.sub.2F, R10=OH)
[0285] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.96 (m, 1H), 5.36 (dd, J=1.6, 17.2 Hz, 1H), 5.25 (dd, J=1.6, 10.4 Hz, 1H), 4.89 (d, J=46.4 Hz, 2H), 4.88 (d, J=3.6 Hz, 1 H), 4.65 (d, J=8.4 Hz, 1H), 4.53-4.45 (m, 3H), 4.39-4.32 (m, 2H), 4.22-3.51 (m, 37H), 3.30 (t, J=8.4 Hz, 1H), 2.73 (dd, J=4.4, 12.4 Hz, 1H), 2.00 (s, 3H), 1.85 (t, J=12.4 Hz, 1H).
[0286] D-4. Ac-Phenyl_SSEA4-allyl (R1=OH, RN=NHCOCH.sub.2Ph, R10=OH)
[0287] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.45-7.34 (m, 5H), 6.02 (m, 1H), 5.42 (dd, J=1.2, 17.2 Hz, 1H), 5.32 (dd, J=1.2, 10.4 Hz, 1H), 4.94 (d, J=4.0 Hz, 1H), 4.72 (d, J=8.4 Hz, 1H), 4.59-4.52 (m, 3H), 4.46-4.38 (m, 2H), 4.30-3.50 (m, 38 H), 3.42-3.32 (m, 4H), 2.77 (dd, J=4.4, 12.8 Hz, 1H), 2.05 (s, 3H), 1.90 (t, J=12.0 Hz, 1H).
[0288] D-5. Ac-Azido_SSEA4-allyl (R1=OH, RN=NHCOCH.sub.2N.sub.3, R10=OH)
[0289] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.95 (m, 1H), 5.35 (dd, J=1.6, 17.2 Hz, 1H), 5.25 (dd, J=1.2, 10.4 Hz, 1H), 4.88 (d, J=3.6 Hz, 1H), 4.65 (d, J=8.4 Hz, 1H), 4.52-4.46 (m, 3H), 4.40-4.32 (m, 2H), 4.23-4.18 (m, 3H), 4.12-3.50 (m, 36 H), 3.30 (t, J=5.6 Hz, 1H), 2.72 (dd, J=4.8, 12.8 Hz, 1H), 2.00 (s, 3H), 1.84 (t, J=12.4 Hz, 1H).
[0290] D-6. 5'-Azido_SSEA4-allyl (R1=OH, RN=N.sub.3, R10=OH)
[0291] .sup.1H NMR (400 MHz, D.sub.2O): 6 5.99 (m, 1H), 4.40 (dd, J=1.6, 17.2 Hz, 1H), 5.29 (d, J=10.4 Hz, 1H), 4.92 (d, J=3.6 Hz, 1H), 4.70 (d, J=8.4 Hz, 1H), 4.56-4.51 (m, 3H), 4.43-4.38 (m, 2H), 4.26 (d, J=3.6 Hz, 2H), 4.22 (d, J=6.4 Hz, 1H), 4.10-3.46 (m, 35H), 3.36-3.32 (m, 1H), 2.74 (dd, J=4.8, 12.4 Hz, 1H), 2.04 (s, 3H), 1.79 (t, J=12.4 Hz); HRMS (ESI-TOF, M-H-) C.sub.44H.sub.71N.sub.4O.sub.33 calcd for 1183.4001, found 1183.4056.
[0292] D-7. 9'-Azido_SSEA4-allyl (R1=OH, RN=NHAc, R10=N.sub.3)
[0293] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.96 (m, 1H), 5.36 (dd, J=1.6, 17.3 Hz, 1H), 5.26 (dd, J=1.6, 10.4 Hz, 1H), 4.90 (d, J=3.6 Hz, 1H), 4.68 (d, J=8.4 Hz, 1H), 4.55-4.47 (m, 3H), 4.41-4.35 (m, 2H), 4.25-4.14 (m, 3H), 4.10-3.41 (m, 34 H), 3.31 (t, J=6.8 Hz, 1H), 2.72 (dd, J=4.8, 12.8 Hz, 1H), 2.02 (s, 3H), 1.79 (t, J=12.0 Hz, 1H).
[0294] D-8. NHBz_SSEA4-allyl (R1=OH, RN=NHBz, R10=OH)
[0295] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.76-7.73 (m, 2H), 7.59 (m, 1H), 7.51-7.46 (m, 2H), 5.90 (m, 1H), 5.30 (dd, J=1.6, 17.2 Hz, 1H), 5.25 (dd, J=1.6, 10.8 Hz, 1H), 4.89 (d, J=3.6 Hz, 1H), 4.67 (d, J=8.8 Hz, 1H), 4.55-4.45 (m, 3H), 4.39-4.38 (m, 2H), 4.24-3.50 (m, 34H), 3.30 (t, J=8.0 Hz, 1H), 2.77 (dd, J=4.4, 12.4 Hz, 1H), 2.01 (s, 3H), 1.90 (t, J=12.4 Hz, 1H).
[0296] E: Oxidation reaction for SSEA4 analog-aldehyde
##STR00021##
[0297] In certain exemplary embodiments, a stirred solution of the SSEA4 analogs-allyl in methanol and H.sub.2O was ozonolysis for 15 minutes under O.sub.3 gas atmosphere at -70.degree. C. The reaction mixture was quenched by dimethyl sulfide (Me.sub.2S) and then the solution was evaporated in vacuo. The desired SSEA4 analogs-aldehyde was then purified by G15. (Scheme 6)
[0298] .sup.1HNMR of SSEA4 analogs-aldehyde
[0299] E-1: SSEA4-aldehyde (RN=NHAc, R10=OH)
[0300] 1H NMR (400 MHz, D.sub.2O) .delta. 5.19 (t, J=4.9 Hz, 1H), 4.89 (d, J=3.9 Hz, 1H), 4.66 (d, J=8.4 Hz, 1H), 4.54-4.45 (m, 3H), 4.36 (t, J=6.5 Hz, 1H), 4.25-4.20 (m, 1H), 4.15 (d, J=3.1 Hz, 1H), 4.08-3.47 (m, 32H), 3.37-3.30 (m, 1H), 2.73 (dd, J=12.4, 4.6 Hz, 1H), 2.00 (d, J=0.9 Hz, 6H), 1.76 (t, J=12.1 Hz, 1H).
[0301] E-2: Neu5Gc_SSEA4-aldehyde (RN=NHGc, R10=OH)
[0302] .sup.1HNMR (400 MHz, D.sub.2O) .delta. 5.20 (t, J=4.9 Hz, 1H), 4.91 (d, J=3.9 Hz, 1H), 4.68 (d, J=8.5 Hz, 1H), 4.52 (dt, J=8.5, 4.5 Hz, 3H), 4.38 (t, J=6.5 Hz, 1H), 4.27-4.22 (m, 1H), 4.17 (d, J=3.1 Hz, 1H), 4.13-3.51 (m, 34H), 3.38-3.32 (m, 1H), 2.76 (dd, J=12.4, 4.6 Hz, 1H), 2.02 (s, 3H), 1.80 (t, J=12.1 Hz, 1H).
[0303] E-3: Ac-Fluoride_SSEA4-aldehyde (RN=NHCOCH.sub.2F, R10=OH)
[0304] 1H NMR (400 MHz, D.sub.2O) .delta. 5.21 (t, J=4.9 Hz, 1H), 4.90 (d, J=46.4 Hz, 2H), 4.69 (d, J=8.5 Hz, 1H), 4.52 (t, J=8.0 Hz, 3H), 4.38 (t, J=6.4 Hz, 1H), 4.24 (d, J=3.1 Hz, 1H), 4.17 (d, J=3.2 Hz, 1H), 4.10-3.45 (m, 33H), 3.40-3.32 (m, 1H), 2.78 (dd, J=12.4, 4.6 Hz, 1H), 2.03 (s, 3H), 1.81 (t, J=12.2 Hz, 1H).
[0305] E-4: Ac-Phenyl_SSEA4-aldehyde (RN=NHCOCH.sub.2Ph, R10=OH)
[0306] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.48-7.27 (m, 5H), 5.22 (t, J=4.9 Hz, 1H), 4.92 (d, J=4.0 Hz, 1H), 4.69 (d, J=8.4 Hz, 1H), 4.56-4.49 (m, 3H), 4.39 (t, J=6.5 Hz, 1H), 4.26 (m, 1H), 4.18 (m, 1H), 4.10-3.45 (m, 34H), 3.43-3.34 (m, 1H), 2.76 (dd, J=12.4, 4.6 Hz, 1H), 2.03 (s, 3H), 1.78 (t, J=12.3 Hz, 1H).
[0307] E-5: Ac-Azido_SSEA4-aldehyde (RN=NHCOCH.sub.2N.sub.3, R10=OH)
[0308] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.20 (t, J=4.9 Hz, 1H), 4.90 (d, J=3.9 Hz, 1H), 4.68 (d, J=8.5 Hz, 1H), 4.54-4.48 (m, 3H), 4.38 (t, J=6.4 Hz, 1H), 4.24 (d, J=3.1 Hz, 1H), 4.17 (d, J=3.1 Hz, 1H), 4.13-3.51 (m, 34H), 3.39-3.32 (m, 1H), 2.75 (dd, J=12.4, 4.6 Hz, 1H), 2.02 (s, 3H), 1.79 (t, J=12.2 Hz, 1H).
[0309] E-6: 9'-Azido_SSEA4-aldehyde (RN=NHAc, R10=N.sub.3)
[0310] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 5.20 (t, J=4.9 Hz, 1H), 4.91 (d, J=3.9 Hz, 1H), 4.69 (d, J=8.5 Hz, 1H), 4.52 (t, J=8.0 Hz, 3H), 4.38 (t, J=6.4 Hz, 1H), 4.24 (d, J=3.1 Hz, 1H), 4.17 (d, J=3.2 Hz, 1H), 4.10-3.45 (m, 32H), 3.39-3.32 (m, 1H), 2.74 (dd, J=12.5, 4.6 Hz, 1H), 2.03 (d, J=2.1 Hz, 6H), 1.77 (t, J=12.1 Hz, 1H).
[0311] E-7: NHBz_SSEA4-aldehyde (RN=NHBz, R10=OH)
[0312] .sup.1H NMR (400 MHz, D.sub.2O) .delta. 7.83-7.76 (m, 2H), 7.63 (t, J=7.3 Hz, 1H), 7.53 (t, J=7.7 Hz, 2H), 5.21 (t, J=4.9 Hz, 1H), 4.92 (d, J=3.8 Hz, 1H), 4.70 (d, J=8.5 Hz, 1H), 4.57-4.49 (m, 3H), 4.39 (t, J=6.5 Hz, 1H), 4.26 (d, J=3.1 Hz, 1H), 4.19 (d, J=3.3 Hz, 1H), 4.16-3.52 (m, 32H), 3.40-3.34 (m, 1H), 2.82 (dd, J=12.4, 4.6 Hz, 1H), 2.04 (d, J=4.7 Hz, 3H), 1.87 (t, J=12.1 Hz, 1H).
Example 3
Synthesis of SSEA3/SSEA4 Analog CRM197-Conjugates Via Sulfo-EMCS Crosslink
##STR00022##
[0314] General Methods:
[0315] Step A. To modify SSEA3 analog-NH2 or SSEA4 analog-NH2 into SSEA3 analog- SH or SSEA4-analog-SH
[0316] To synthesize SSEA3/4 analog CRM197-conjugates, the amine-terminated SSEA3/4 analogs were reacted with the DTSSP linker in PBS buffer (pH 7.4) at room temperature. To monitor the pH value of solution by pH paper, and add some NaOH solution to the solution when the solution became neuter or acid. After the reaction was stirred at room temperature for 12 hours, DTT was added to the solution at room temperature. The solution was kept stirring at 40.degree. C., and then the solvent was removed under reduced pressure. The residue was purified by LH-20 column chromatography to give SSEA3/4 analog-SH.
[0317] Step B: To modify CRM197 into CRM197-maleimide.
[0318] After the salt of commercial CRM197 (1.0 mg) was removed via alternate dissolving in water and dialyzing (Amicon Ultra-0.5, 10 kDa,), the residue was dissolved in PBS buffer (pH 6.5, 1.0 mL) and transferred into a sample vial. Sulfo-EMCS (1.0 mg, 8.22.times.10.sup.-6 mol) was added to the solution, and then the reaction was kept stirring at room temperature for 2 hours. The mixture was purified by Amicon Ultra-0.5 (10 kDa). After using MALDI-TOF to check the molecular weight and BCA assay to calculate the amount of protein, the CRM197-maleimid was stored in PBS buffer (pH 7.2, 1.0 mg/mL) for next step. According to the data of MALDI-TOF, the amount of maleimid function groups could be calculated. For example, when the molecular weight of CRM197-maleimid was 61841, the numbers of maleimide function groups on CRM197-maleimid were (61841-58326)/193=18.2.
[0319] Step C: The Synthesis of SSEA3/4 analog-CRM197 Conjugates
[0320] The CRM197-maleimids were dissolved in PBS buffer (pH 7.2, the concentration was 1.0 mg/mL) and then different amount of SSEA3/4 analog-SH (5.0 mg/mL in PBS buffer, pH 7.2) were added into the solution. The mixtures were stirred at room temperature for 2 hours. The SSEA3/4 analog-CRM197 conjugates were purified by using Amicon Ultra-0.5 (10 kDa) to remove the nonreactive SSEA3/4 analog-SH and sodium phosphate salt via dialysis. The obtained SSEA3/4 analog-CRM197 conjugates could be characterized by MALDI-TOF analysis to determine the carbohydrate incorporation rate. The nonreactive SSEA3/4 analog-SH could be recovered after reacting with DTT and purifying by LH-20 column chromatography.
TABLE-US-00001 TABLE 1 Carbohydrate incorporation rate of SSEA4 analog with CRM-197 via Sulfo-EMCS Molecular weight (y) average Code Sugar after glycosylation incorporation rate M1 SSEA4 75465 8.84 M2 Neu5Gc_SSEA4 70750 5.83 M3 Ac-Alkynyl_SSEA4 68965 5.94 M4 Ac-Fluoride_SSEA4 69190 4.59 M5 Ac-Phenyl_SSEA4 75454 8.10 M6 Ac-Azido_SSEA4 70274 5.30 M7 9'-Azido_SSEA4 76596 9.87 M8 Glc-azido_SSEA4 73047 8.00
Example 4
Syntheses of SSEA4-Gc-CRM197 Conjugates Via Sulfo-EMCS Crosslink
##STR00023##
[0322] Step A: To modify SSEA4-Gc-NH2 into SSEA4-Gc-SH
[0323] DTSSP (5.0 mg, 8.22.times.10.sup.-6 mol) was added to a flask of SSEA4-Gc-NH2 (5.0 mg, 4.01.times.10.sup.-6 mol) in PBS buffer (pH 7.4, 1.0 mL) at room temperature. To monitor the pH value of solution by pH paper, NaOH (1 M/water) was added to the solution when the solution became neuter or acid. After the reaction was stirred at room temperature for 12 hours, DTT (5.0 mg, 32.41.times.10.sup.-6 mol) was added to the solution at room temperature. The solution was kept stirring at 40.degree. C. for 1 hour, and then the solvent was removed under reduced pressure. The residue was purified by LH-20 column chromatography to give SSEA4-Gc-SH (5.0 mg, 93%)
[0324] Step B: To modify CRM197 into CRM197-maleimide.
[0325] After the salt of commercial CRM197 (1.0 mg) was removed via alternate dissolving in water and dialyzing (Amicon Ultra-0.5, 10 kDa,), the residue was dissolved in PBS buffer (pH 6.5, 1.0 mL) and transferred into a sample vial. Sulfo-EMCS (1.0 mg, 8.22.times.10.sup.-6 mol) was added to the solution, and then the reaction was kept stirring at room temperature for 2 hours. The mixture was purified by Amicon Ultra-0.5 (10 kDa). After using MALDI-TOF to check the molecular weight and BCA assay to calculate the amount of protein, the CRM197-maleimid was stored in PBS buffer (pH 7.2, 1.0 mg/mL) for next step. According to the data of MALDI-TOF, the amount of maleimid function groups could be calculated. For example, when the molecular weight of CRM197-maleimid was 61841, the numbers of maleimide function groups on CRM197-maleimid were (61841-58326)/193=18.2.
[0326] The CRM197-maleimids were dissolved in PBS buffer (pH 7.2, the concentration was 1.0 mg/mL) and then different amount of SSE4Gc-SH (5.0 mg/mL in PBS buffer, pH 7.2) were added into the solution. The mixtures were stirred at room temperature for 2 hours. The SSEA4-Gc-CRM197 conjugates were purified by using Amicon Ultra-0.5 (10 kDa) to remove the nonreactive SSEA4-Gc-SH and sodium phosphate salt via dialysis. The obtained SSEA4-Gc-CRM197 conjugates could be characterized by MALDI-TOF analysis to determine the carbohydrate incorporation rate as showing in Table 2. The nonreactive SSEA4-Gc-SH could be recovered after reacting with DTT and purifying by LH-20 column chromatography.
[0327] Step C: To trap the nonreactive maleimides of CRM197-maleimide
[0328] The SSEA4-Gc-CRM197 conjugates were dissolved in PBS buffer (pH 7.2, the concentration was 1.0 mg/mL) and 10.0 equivalent of 2-mercaptoethanol (5 mg/mL, PBS buffer, pH 7.2) were added to the solution. The mixtures were stirred at room temperature for 2 hours. The SSEA4-Gc-CRM197 conjugates were purified by using Amicon Ultra-0.5 (10 kDa) to remove the nonreactive 2-mercaptoethanol and sodium phosphate salt via dialysis and then lyphophilized to a white powder.
TABLE-US-00002 TABLE 2 Conjugation of CRM197 with SSEA4-Gc Number Amount of Number HSC.sub.2H.sub.4OH CRM197 of Linkers PBS Buffer SSEA4-Gc Reaction of (5 mg/mL) CRM197 (.mu.g) Linkers (mol) (pH 7.4, .mu.L) (5 mg/mL) Time Sugars 10.0 eq. (.mu.g) 1 1388 21.9 5.21 .times. 10.sup.-7 1000 28.1 .mu.L 2 hr 2.0 81.4 .mu.L 1550.4 (0.2 eq.) 2 694 21.9 2.61 .times. 10.sup.-7 500 28.2 .mu.L 2 hr 4.2 40.8 .mu.L 657.4 (0.4 eq.) 3 694 21.9 2.61 .times. 10.sup.-7 500 56.4 .mu.L 2 hr 6.5 40.8 .mu.L 665.0 (0.8 eq.) 4 694 21.9 2.61 .times. 10.sup.-7 500 84.5 .mu.L 2 hr 6.9 40.8 .mu.L 627.0 (1.2 eq.) 5 694 21.9 2.61 .times. 10.sup.-7 500 140.9 .mu.L 2 hr 7.1 40.8 .mu.L 615.6 (2.0 eq.) 6 694 21.9 2.61 .times. 10.sup.-7 500 281.8 .mu.L 2 hr 7.0 40.8 .mu.L 665.0 (4.0 eq.) 7 694 21.9 2.61 .times. 10.sup.-7 500 704.4 .mu.L 2 hr 6.8 40.8 .mu.L 695.4 (10.0 eq.) a) M.W. of CRM197 = 58326 .fwdarw. 1000 .mu.g = 0.1715 .times. 10.sup.-7 mol b) M.W. of SSEA4-Gc-SH = 1349.479 .fwdarw. 5 mg/mL = 37.051 .times. 10.sup.-7 mol/mL c) M.W. of 2-Mercaptoethanol = 78.13 .fwdarw. 5 mg/mL = 639.91 .times. 10.sup.-7 mol/mL
Example 5
SSEA4 Analog-CRM197 Conjugate Via SBAP Crosslink
##STR00024##
[0330] CRM-197 was dissolved in 0.1 M phosphate buffer pH 7.4 (.about.1 mg/ml), and SBAP (1.0 mg) was added to the solution. The solution was stirred gently for 2 hrs at room temperature. The mixture was then diluted with PBS buffer and centrifuge against 5 changes of 0.1 M phosphate buffer pH 7.4 by Amicon Ultra-0.5 (10 kDa, 2.times.). The obtained modified CRM-197 can be characterized by MALDI-TOF (positive mode, matrix was sinapinic acid, H.sub.2O) analysis to determine the SBAP incorporation rate.
[0331] Modified CRM-197 was dissolved in 0.1 M phosphate buffer pH 8.0 (.about.1 mg/ml), and SSEA4-SH analog was added to the solution. The mixture was stirred for 1 day at room temperature. The mixture was then diluted with PBS buffer and centrifuge against 5 changes of 0.1 M PBS buffer pH 7.4 by Amicon Ultra-0.5 (10 kDa, 2.times.). The obtained sugar-protein conjugate could be characterized by MALDI-TOF (positive mode, matrix was sinapinic acid, H.sub.2O) analysis to determine the carbohydrate incorporation rate. (Scheme 9)
TABLE-US-00003 TABLE 3 Carbohydrate incorporation rate of SSEA4 analog with CRM-197 via SBAP Molecular weight (x) average Code Sugar after glycosylation incorporation rate S1 SSEA4 68212 4.79 S2 Neu5Gc_SSEA4 67651 4.84 S3 Ac-Alkynyl_SSEA4 70308 5.70 S4 Ac-Fluoride_SSEA4 69309 5.01 S5 Ac-Phenyl_SSEA4 68891 5.05 S6 Ac-Azido_SSEA4 68359 4.50 S7 5'-Azido_SSEA4 71638 7.06 S8 9'-Azido_SSEA4 72545 7.90 S9 Glc-azido_SSEA4 67131 3.9 S10 NHBz_SSEA4 69636 5.50
Example 6
SSEA4 Analog-CRM197 Conjugate Via Reductive Amination Crosslink
##STR00025##
[0333] In certain embodiments, CRM197 was dissolved in 0.1 M phosphate buffer (pH 6-9) (.about.1 mg/ml), and enough quantity SSEA4-aldehyde analogs and NaCNBH3 were added to the solution. The solution was stirred gently for 3 days at room temperature. The mixture was then diluted with deionized water and centrifuge against 5 changes of 0.1 M phosphate buffer pH 7.4 by Amicon Ultra-0.5 (10 kDa, 2.times.). The obtained sugar-protein conjugate was characterized by MALDI-TOF (positive mode, matrix was sinapinic acid, H.sub.2O) analysis to determine the carbohydrate incorporation rate. (Scheme 10)
TABLE-US-00004 TABLE 4 Carbohydrate incorporation rate of SSEA4 analog with CRM-197 via reductive amination Molecular weight (m) average Code Sugar after glycosylation incorporation rate R1 SSEA4 69025 8.89 R2 Neu5Gc_SSEA4 65154 5.6 R3 Ac-Fluoride_SSEA4 69315 9 R4 Ac-Phenyl_SSEA4 71329 10.1 R5 Ac-Azido_SSEA4 67765 7.6 R6 9'-Azido_SSEA4 67635 7.58 R7 NHBz_SSEA4 67124 6.95
Example 7
Immunization Determination of the SSEA4 Analog-CRM197 Conjugates
[0334] Exemplary Method
[0335] To demonstrate the efficacy/immunogenicity of the SSEA4 analog CRM197 conjugates (S1.about.S10), female C57BL/6 mice (n=5 for each group) were vaccinated intramuscularly with 0.5 .mu.g of SSEA4 analog CRM197-conjugates combining the use of 2.0 .mu.g of glycolipid adjuvant. Control mice were given only phosphate buffer saline with 2.0 .mu.g of glycolipid adjuvant. The vaccination was conducted at biweekly intervals for 2 months, and the antisera from the immunized mice were collected one week after each vaccination. The antibody titers against SSEA4 were examined by ELISA using SSEA4 immobilized 96-well titer plates. ELISA was conducted using SSEA4 immobilized 96-well titer plate. Briefly, the diluted antisera were incubated with the immobilized SSEA4 at room temperature for 2 hr. After the washing cycle, the captured anti-SSEA4 antibodies were then detected using HPR-conjugated anti-IgG or IgM specific antibody.
[0336] To determine if the glycan-protein conjugation method would interfere the immune response, native SSEA4 was conjugated with CRM197 through EMCS linker (M1), SBAP linker (S1) or reductive amination (R1) and used for immunogenicity study as described above.
[0337] Representative Result
[0338] After four times of immunization, native SSEA4, as well as all eight SSEA4 analogs, could positively elicit both IgG (FIG. 3A) and IgM (FIG. 3B) antibodies against SSEA4 when combining the use of Gal-C34 adjuvant. There is no significant difference in the titers of anti-SSEA4 IgG and IgM antibodies among different analog groups. In addition, Glc-C34 can also be used as vaccine adjuvant for inducing both IgG (FIG. 4A) and IgM (FIG. 4B) antibodies against SSEA4 when co-administering with native SSEA4 and the other analogs.
[0339] Furthermore, the results shown in FIG. 5 indicated that the glycan-protein conjugation method can affect the immune response. Combining the use of Gal-C34, the SSEA4-EMCS-CRM197 (M1) elicited a higher anti-SSEA4 IgG antibody titer when comparing to SSEA4-SBAP-CRM197 (S1) and SSEA4-CRM197 (conjugated through reductive amination, R1).
Example 8
Immunogenicity Study of the SSEA4 Analogs CRM197-Conjugates
[0340] To demonstrate the immunogenicity of the SSEA4 analog CRM197-conjugates, five female BALB/c mice were immunized intramuscularly with 2 .mu.g of SSEA4 analog CRM197-conjugates and 2 .mu.g of the glycolipid adjuvant C34 three times at biweekly intervals. In the previous study, the anti-GH antibodies titer was low with SSEA4 analog-protein conjugates alone without any adjuvants. The antisera from each immunogen were obtained ten days after the third immunization and were tested on the glycan microarray containing 94 chemically synthesized glycans, including globo series glycans and other tumor-associated carbohydrate antigens. Because some chemical modifications were carried out on the glycan, some functional linkers were also included in the glycan array to check the cross reactivity.
[0341] Antibodies induced by the SSEA4-Gc CRM197-conjugates were specifically recognized by SSEA4-Gc, native SSEA4 or SSEA4 tetrasaccharide fragments but not by other TACAs and functional linkers. The sera obtained from the glycoconjugates induced high IgG antibody titers, indicating a T-cell-dependent immune response. Interestingly, no significant IgM production was observed for SSEA4-Gc or native SSEA4. Regarding the IgG level against GloboH, the titers of antibodies induced by SSEA4-Gc CRM197 was much higher than the nature form native SSEA4-CRM197 conjugate. Among them the 6.9 molecule of SSEA4-Gc conjugated with one molecule of CRM197 can induce the highest antibody titers.
[0342] Mice Dosage and Immunization Schedule
[0343] For comparing the immunogenicity of SSEA4 analog CRM197, ten groups of five mice (8-week-old female Balb/c mice, BioLASCO, Taiwan) were immunized intramuscularly with glycolipid C34. Three immunizations were given at 2-week intervals. Each vaccination contained 2 .mu.g SSEA4 analog and 2 .mu.g C34. Control mice were injected with phosphate buffer saline (PBS). Mice were bled before the first immunization (preimmune) and 10 days after the third immunization. All of the sera were obtained by centrifugation at 4,000.times.g for 10 min. The serologic responses were analyzed by glycan microarray.
[0344] Serologic Assay with Glycan Array
[0345] Mouse sera were diluted with 1% BSA/PBST buffer (PBST buffer: PBS and 0.05% Tween-20, pH 7.4). The glycan microarray was blocked with Superblock blocking buffer (Pierce) for 1 h at 4.degree. C. and washed three times with PBST buffer before use. The serum dilutions were then introduced to the glycan microarray and incubated at 4.degree. C. for 1 h. Excess serum antibodies were washed out and the microarrays were incubated individually with Alexa Fluor 647-conjugated goat anti-mouse IgG antibody or DyLight 649-conjugated goat anti-mouse IgM antibody as the 2nd antibody at 4.degree. C. in dark for 1 h. The slides were then washed three times with PBST and scanned at 635 nm wavelength with a microarray fluorescence chip reader (GenePix 4300A; Molecular Devices Corporation) and scanned images were analyzed with GenePix Pro-6.0 analysis software (Axon Instruments, Union City, Calif., USA).
OTHER EMBODIMENTS
[0346] All of the features disclosed in this specification may be combined in any combination. Each feature disclosed in this specification may be replaced by an alternative feature serving the same, equivalent, or similar purpose. Thus, unless expressly stated otherwise, each feature disclosed is only an example of a generic series of equivalent or similar features. From the above description, one skilled in the art can easily ascertain the essential characteristics of the described embodiments, and without departing from the spirit and scope thereof, can make various changes and modifications of the embodiments to adapt it to various usages and conditions. Thus, other embodiments are also within the claims.
* * * * *




























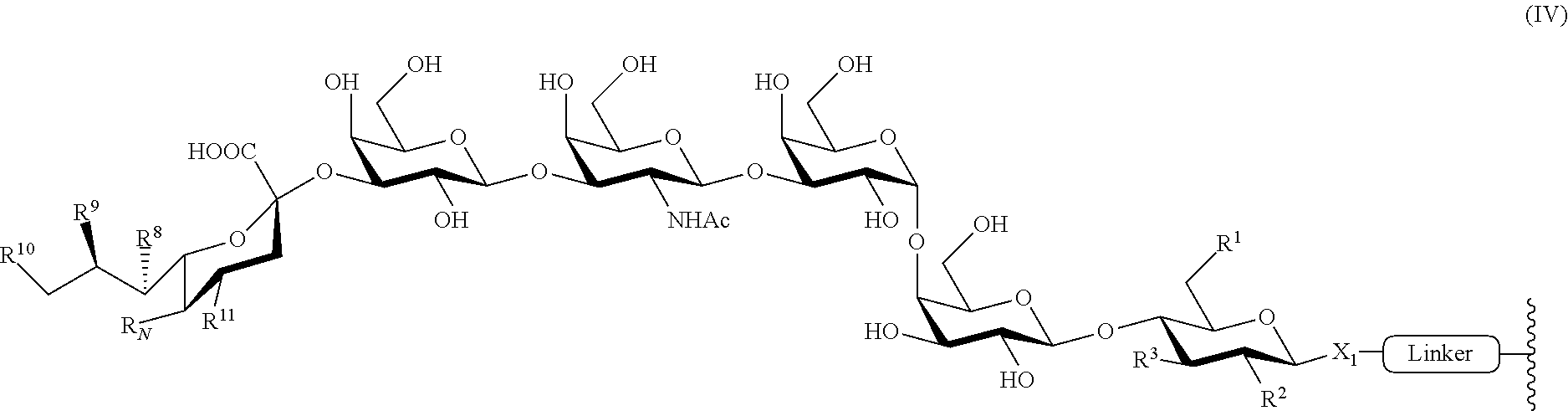


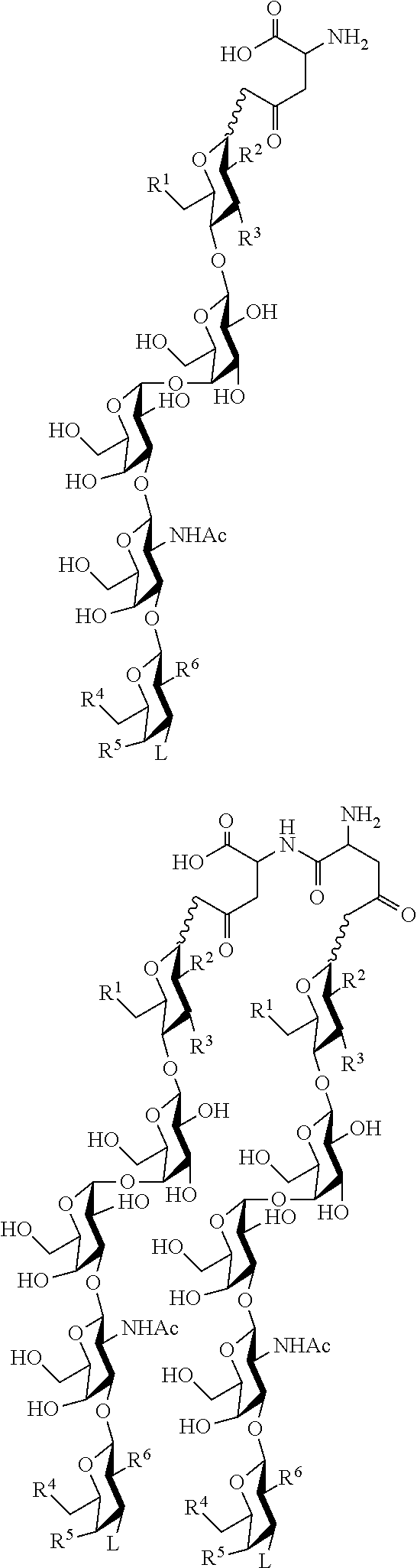

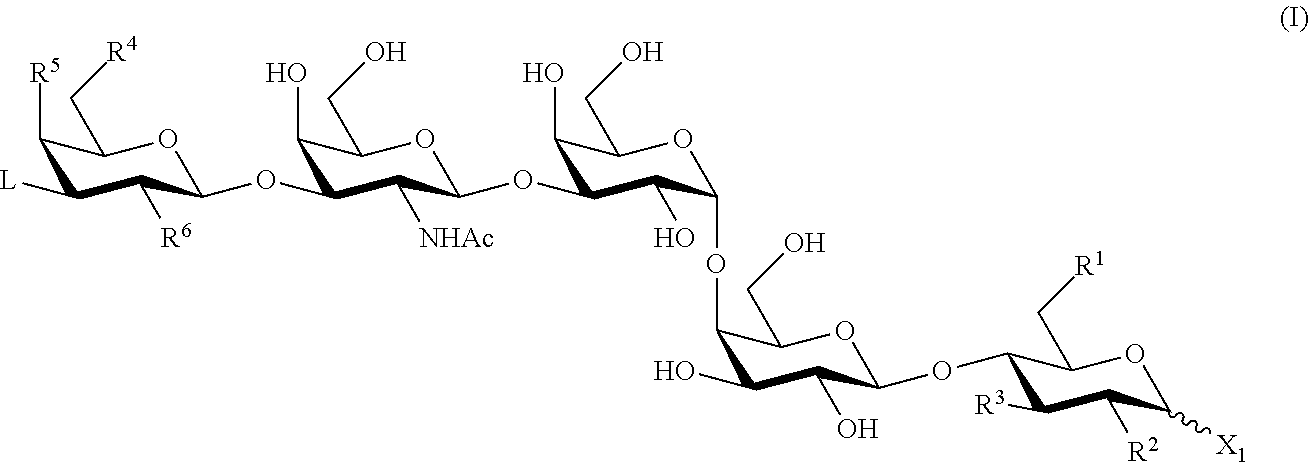

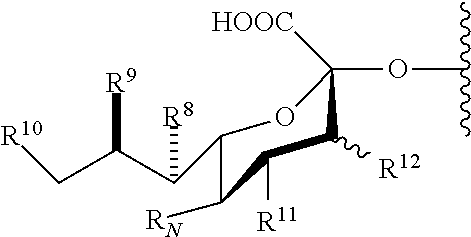

D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.