Method For Preparing Titanium Plating Solution And Method For Manufacturing Titanium Plated Product
NUMATA; Koma ; et al.
U.S. patent application number 16/462618 was filed with the patent office on 2020-02-27 for method for preparing titanium plating solution and method for manufacturing titanium plated product. This patent application is currently assigned to Sumitomo Electric Industries, Ltd.. The applicant listed for this patent is Kyoto University, Sumitomo Electric Industries, Ltd.. Invention is credited to Tomoyuki AWAZU, Masatoshi MAJIMA, Toshiyuki NOHIRA, Yutaro NORIKAWA, Koma NUMATA, Mitsuyasu OGAWA, Kouji YASUDA.
| Application Number | 20200063281 16/462618 |
| Document ID | / |
| Family ID | 62195781 |
| Filed Date | 2020-02-27 |

| United States Patent Application | 20200063281 |
| Kind Code | A1 |
| NUMATA; Koma ; et al. | February 27, 2020 |
METHOD FOR PREPARING TITANIUM PLATING SOLUTION AND METHOD FOR MANUFACTURING TITANIUM PLATED PRODUCT
Abstract
The titanium plating solution production method including measuring a titanium plating solution containing fluorine and titanium by cyclic voltammetry under the following conditions, and adding titanium to the titanium plating solution so that the potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is 0.75 V or more. Conditions: when the temperature of the titanium plating solution is 650.degree. C. to 850.degree. C. and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec to 500 mV/sec between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
| Inventors: | NUMATA; Koma; (Itami-shi, Hyogo, JP) ; MAJIMA; Masatoshi; (Itami-shi, Hyogo, JP) ; AWAZU; Tomoyuki; (Itami-shi, Hyogo, JP) ; OGAWA; Mitsuyasu; (Itami-shi, Hyogo, JP) ; NOHIRA; Toshiyuki; (Kyoto-shi, Kyoto, JP) ; YASUDA; Kouji; (Kyoto-shi, Kyoto, JP) ; NORIKAWA; Yutaro; (Kyoto-shi, Kyoto, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Sumitomo Electric Industries,
Ltd. Osaka-shi, Osaka JP Kyoto University Kyoto-shi, Kyoto JP |
||||||||||
| Family ID: | 62195781 | ||||||||||
| Appl. No.: | 16/462618 | ||||||||||
| Filed: | September 12, 2017 | ||||||||||
| PCT Filed: | September 12, 2017 | ||||||||||
| PCT NO: | PCT/JP2017/032796 | ||||||||||
| 371 Date: | May 21, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C25D 21/14 20130101; C25D 3/66 20130101; C25D 21/12 20130101 |
| International Class: | C25D 3/66 20060101 C25D003/66; C25D 21/12 20060101 C25D021/12 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 22, 2016 | JP | 2016-227050 |
Claims
1. A method for preparing a titanium plating solution comprising: measuring a titanium plating solution containing fluorine and titanium by cyclic voltammetry under the following conditions; and adding titanium to the titanium plating solution so that a potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is 0.75 V or more, conditions: when the temperature of the titanium plating solution is 650.degree. C. or more and 850.degree. C. or less and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec or more and 500 mV/sec or less between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
2. The method for preparing a titanium plating solution according to claim 1, wherein the titanium plating solution is obtained by dissolving titanium in a molten salt of potassium fluoride and potassium chloride.
3. The method for preparing a titanium plating solution according to claim 1, wherein the titanium plating solution is obtained by dissolving K.sub.2TiF.sub.6 in a molten salt of potassium fluoride and potassium chloride.
4. The method for preparing a titanium plating solution according to claim 3, wherein the content of K.sub.2TiF.sub.6 in the titanium plating solution is 0.1 mol % or more.
5. The method for preparing a titanium plating solution according to claim 2, wherein the molar mixing ratio between potassium fluoride and potassium chloride is 10:90 to 90:10.
6. The method for preparing a titanium plating solution according to claim 1, wherein the titanium added to the titanium plating solution is titanium sponge.
7. A method for manufacturing a titanium plated product which includes an electrolyzing step of carrying out a molten salt electrolysis by using a cathode and an anode provided in a titanium plating solution containing fluorine and titanium so as to electrodeposit titanium on the surface of the cathode, the titanium plating solution being prepared by the method for preparing a titanium plating solution according to claim 1.
8. The method for manufacturing a titanium plated product according to claim 7, wherein the titanium plating solution used in the electrolyzing step is measured by cyclic voltammetry under the following conditions, and a potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is controlled to be 0.75 V or more, conditions: when the temperature of the titanium plating solution is 650.degree. C. or more and 850.degree. C. or less and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec or more and 500 mV/sec or less between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
Description
TECHNICAL FIELD
[0001] The present disclosure relates to a method for preparing a titanium plating solution and a method for manufacturing a titanium plated product. The present application claims the benefit of priority to Japanese Patent Application No. 2016-227050 filed on Nov. 22, 2016, the entire contents of which are incorporated herein by reference.
BACKGROUND ART
[0002] Titanium is a metal that is excellent in corrosion resistance, heat resistance and specific strength. However, titanium is costly to manufacture and difficult to smelt and work, which hampers the wide use of titanium. Dry deposition, such as chemical vapor deposition (CVD) and physical vapor deposition (PVD), is now partially used in industry as one of the methods that take advantage of high corrosion resistance, high strength, and other properties of titanium and titanium compounds. However, the dry deposition cannot be applied to a complex-shaped substrate. As a method for depositing titanium that would solve this problem, electrodeposition of titanium in a molten salt may be adopted.
[0003] For example, Japanese Patent Laying-open No. 2015-193899 (PTL 1) describes that an alloy film of Fe and Ti is formed on a Fe wire surface by using a molten salt bath of KF--KCl to which K.sub.2TiF.sub.6 or TiO.sub.2 is added.
CITATION LIST
Patent Literature
[0004] PTL 1: Japanese Patent Laying-open No. 2015-193899
SUMMARY OF INVENTION
[0005] A method for preparing a titanium plating solution according to an embodiment of the present disclosure includes measuring a titanium plating solution containing fluorine and titanium by cyclic voltammetry under the following conditions, and adding titanium to the titanium plating solution so that the potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is 0.75 V or more, conditions: when the temperature of the titanium plating solution is 650.degree. C. or more and 850.degree. C. or less and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec or more and 500 mV/sec or less between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
BRIEF DESCRIPTION OF DRAWINGS
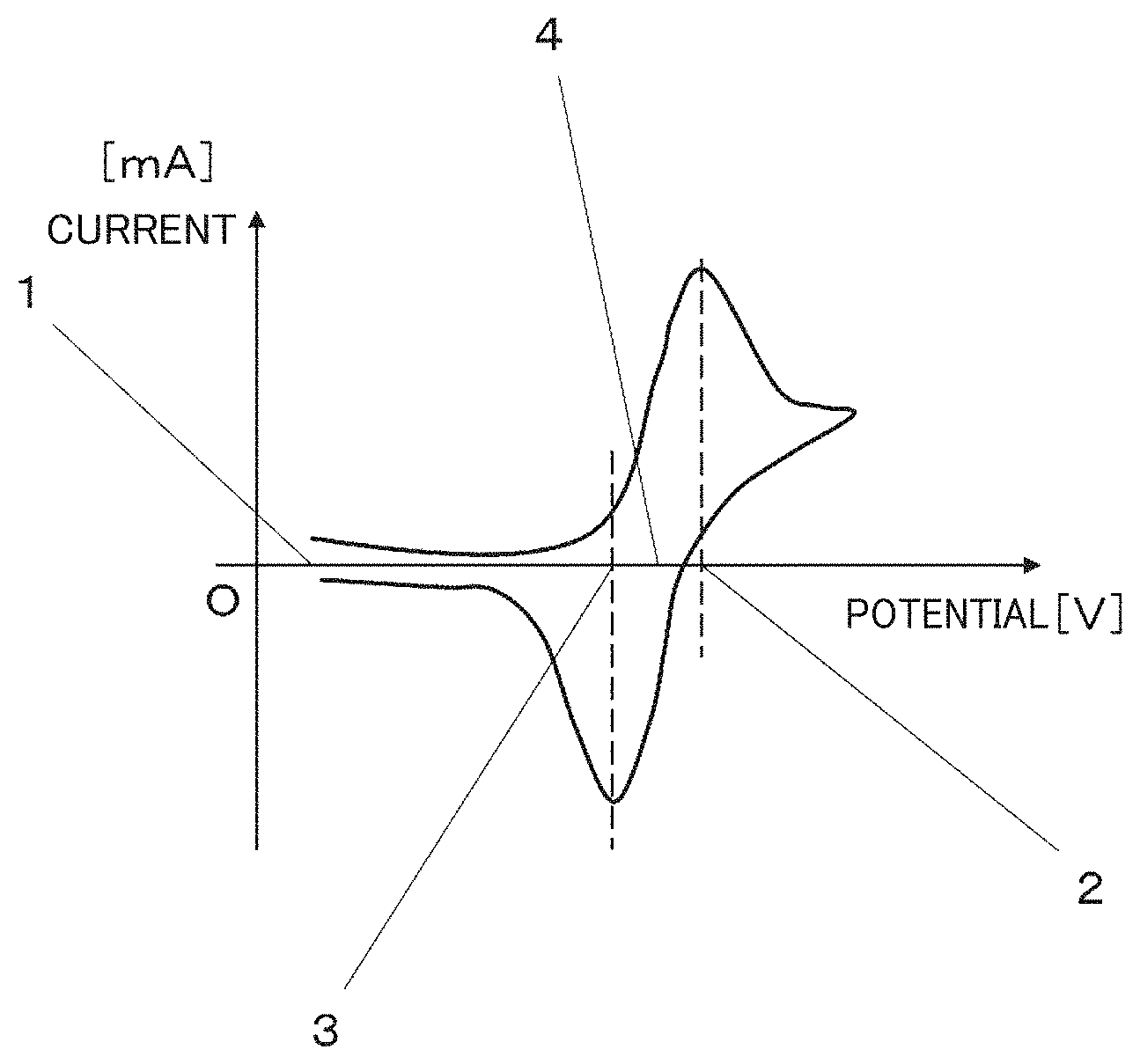
[0006] FIG. 1 is a graph schematically illustrating a measurement result of a titanium plating solution by cyclic voltammetry;
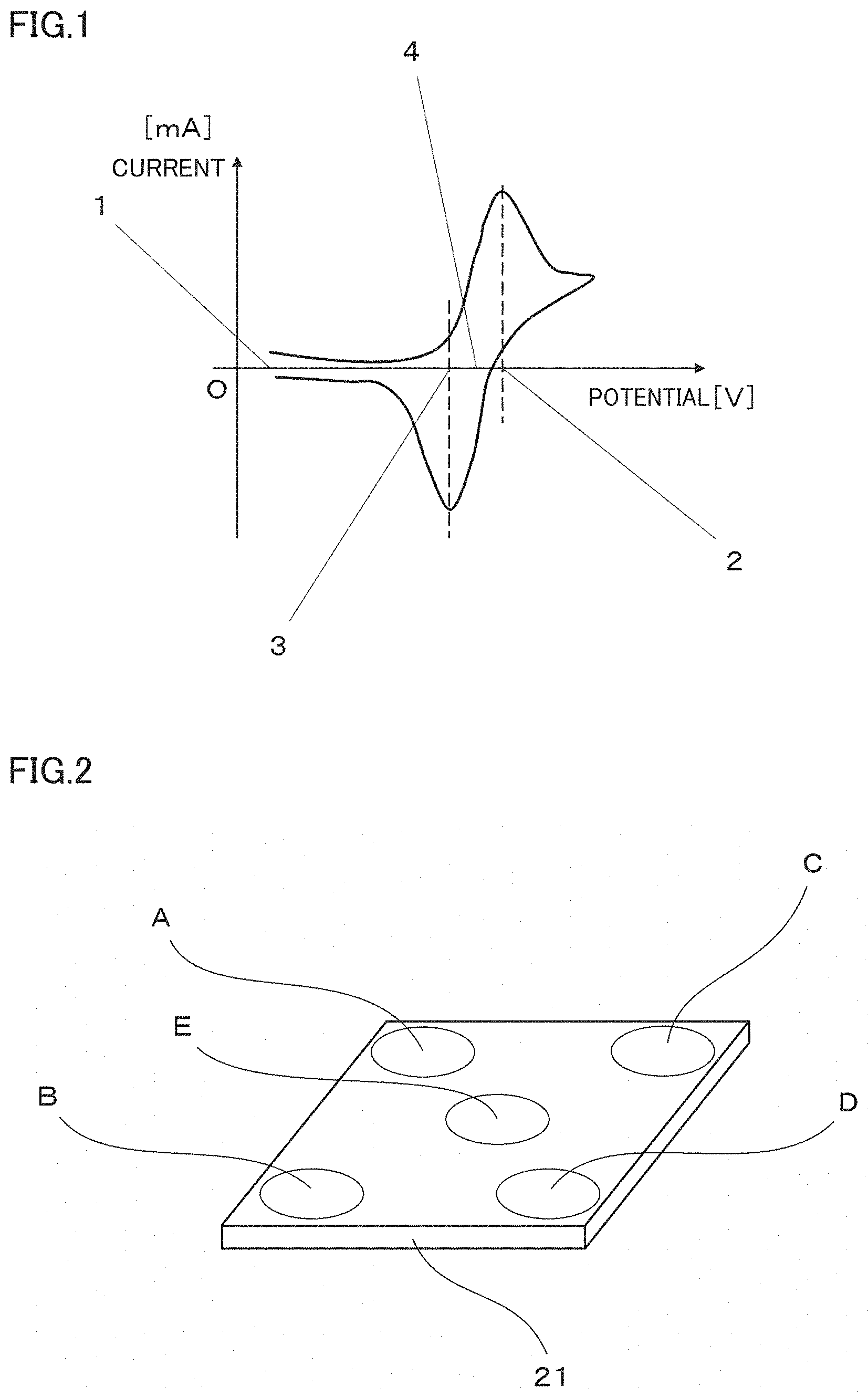
[0007] FIG. 2 is a schematic view illustrating an example of defining areas A to E on a titanium plated product in a method of measuring an average film thickness of a titanium plating film on the titanium plated product;
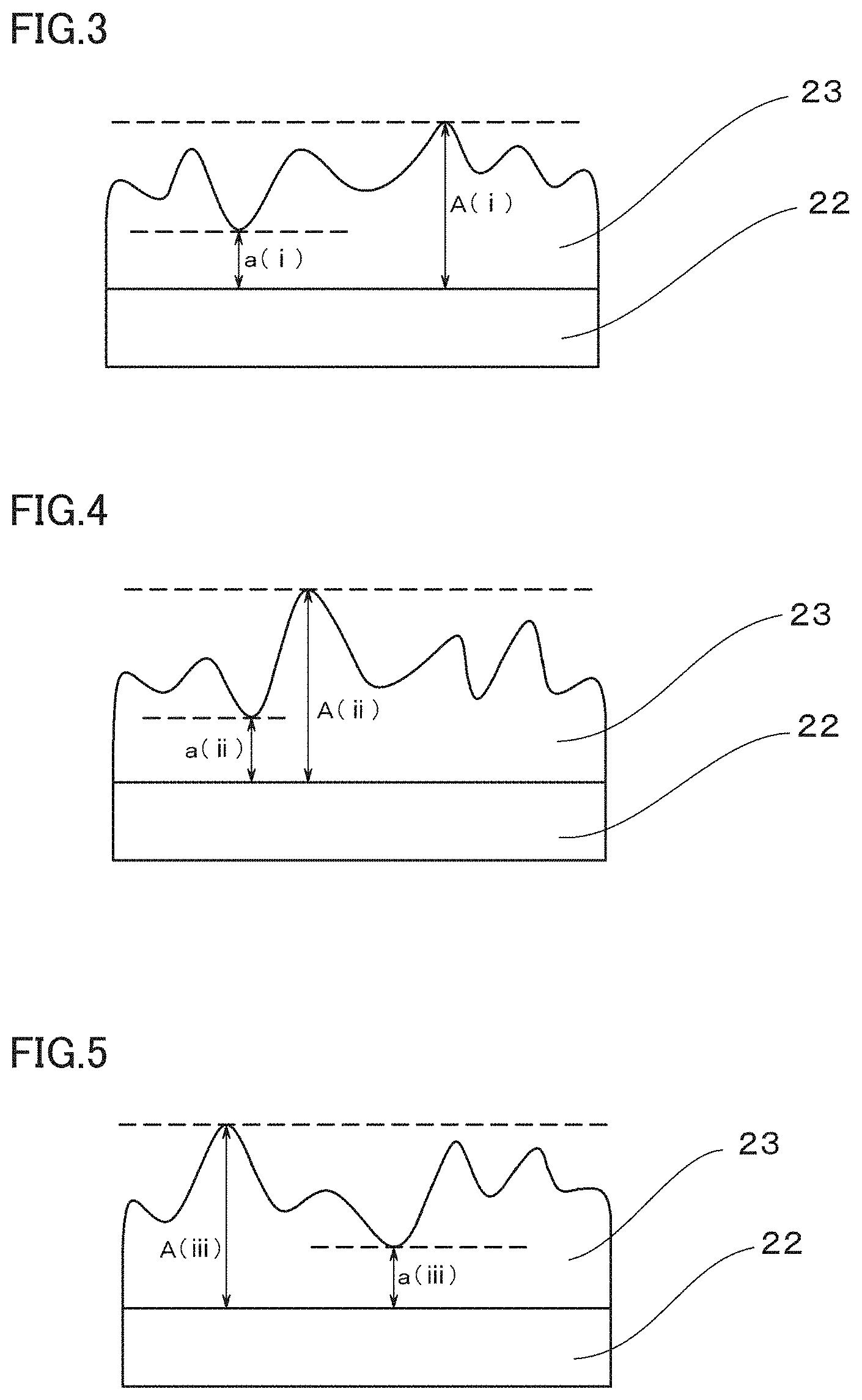
[0008] FIG. 3 is a conceptual diagram illustrating an example of a field of view (i) when the area A of the titanium plated product illustrated in FIG. 2 is observed with a scanning electron microscope.
[0009] FIG. 4 is a conceptual diagram illustrating an example of a field of view (ii) when the area A of the titanium plated product illustrated in FIG. 2 is observed with a scanning electron microscope;
[0010] FIG. 5 is a conceptual diagram illustrating an example of a field of view (iii) when the area A of the titanium plated product illustrated in FIG. 2 is observed with a scanning electron microscope.
[0011] FIG. 6 is a graph schematically illustrating a measurement result by cyclic voltammetry of a titanium plating solution No. 4 prepared in Example 4; and
[0012] FIG. 7 is a graph schematically illustrating a measurement result by cyclic voltammetry of a titanium plating solution No. A prepared in Comparative Example 4.
DETAILED DESCRIPTION
Problem to be Solved by the Present Disclosure
[0013] According to the studies conducted by the inventors of the present disclosure, although an alloy film of Fe and Ti can be electrodeposited on the surface of a cathode used in the molten salt electrolysis by the method described in PTL 1, a metal Ti film cannot be electrodeposited by the method. Specifically, the alloy film of Fe and Ti is stable in the molten salt bath, whereas the metal Ti dissolves in the molten salt bath due to a comproportionation reaction.
[0014] After further studies, the inventors of the present disclosure have found that it is effective to carry out the molten salt electrolysis by adding titanium to a titanium plating solution which is a molten salt containing KF, KCl and K.sub.2TiF.sub.6 in at least a minimum amount necessary for converting Ti.sup.4+ to Ti.sup.3+ according to the comproportionation reaction represented by the following formula (A)
3Ti.sup.4++metal Ti.fwdarw.4Ti.sup.3+ Formula (A):
[0015] According to the above method, it is possible to form a smooth titanium plating film on the surface of the cathode used in the molten salt electrolysis.
[0016] However, since it is impossible to confirm whether or not the comproportionation reaction has sufficiently progressed in the above method, it is necessary to wait longer than the time required for the molten salt electrolysis to finish after titanium is added to the titanium plating solution. In addition, if oxygen is mixed into the titanium plating solution from the external environment for some reasons, the titanium ions may be oxidized from Ti.sup.3+ to Ti.sup.4+, and thereby it is impossible to know whether or not Ti.sup.3+ is sufficiently present in the titanium plating solution.
[0017] In view of the above problems, it is an object of the present disclosure to provide a method for preparing a titanium plating solution in which the concentration ratio between Ti.sup.3+ and Ti.sup.4+ in the titanium plating solution is monitored so that the concentration of Ti.sup.3+ is maintained sufficiently high.
Advantageous Effect of the Present Disclosure
[0018] According to the present disclosure, it is possible to provide a method for preparing a titanium plating solution in which the concentration ratio between Ti.sup.3+ and Ti.sup.4+ in the titanium plating solution is monitored so that the concentration of Ti.sup.3+ is maintained sufficiently high.
Description of Embodiments
[0019] First, embodiments of the present disclosure are enumerated hereinafter.
[0020] (1) A method for preparing a titanium plating solution according to one embodiment of the present disclosure includes: measuring a titanium plating solution containing fluorine and titanium by cyclic voltammetry under the following conditions; and adding titanium to the titanium plating solution so that the potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is 0.75 V or more, conditions: when the temperature of the titanium plating solution is 650.degree. C. or more and 850.degree. C. or less and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec or more and 500 mV/sec or less between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
[0021] According to the embodiment described in the above (1), it is possible to provide a method for preparing a titanium plating solution in which the concentration ratio between Ti.sup.3+ and Ti.sup.4+ in the titanium plating solution is monitored so that the concentration of Ti.sup.3+ is maintained sufficiently high.
[0022] (2) In the method for preparing a titanium plating solution described in the above (1), it is preferable that the titanium plating solution is obtained by dissolving titanium in a molten salt of potassium fluoride and potassium chloride.
[0023] (3) In the method for preparing a titanium plating solution described in the above (1) or (2), it is preferable that the titanium plating solution is obtained by dissolving K.sub.2TiF.sub.6 in a molten salt of potassium fluoride and potassium chloride.
[0024] According to the embodiment described in the above (2) or (3), it is possible to provide a titanium plating solution that can be maintained in liquid state at a temperature lower than a titanium plating solution which is obtained by dissolving titanium in potassium fluoride.
[0025] (4) In the method for preparing a titanium plating solution described in the above (3), it is preferable that the content of the K.sub.2TiF.sub.6 in the titanium plating solution is 0.1 mol % or more.
[0026] According to the embodiment described in the above (4), it is possible to provide a titanium plating solution enabling the titanium plating to be carried out stably.
[0027] (5) In the method for preparing a titanium plating solution described in any one of the above (2) to (4), it is preferable that the molar mixing ratio between potassium fluoride and potassium chloride is 10:90 to 90:10.
[0028] According to the embodiment described in the above (5), it is possible to provide a titanium plating solution enabling the formation of a smooth titanium plating film.
[0029] (6) In the method for preparing a titanium plating solution described in any one of the above (1) to (5), it is preferable that the titanium added to the titanium plating solution is titanium sponge.
[0030] According to the embodiment described in the above (6), it is possible to facilitate the progress of the comproportionation reaction of titanium in the titanium plating solution.
[0031] Note that the titanium sponge refers to a porous metal titanium having a porosity of 1% or more. The porosity of the titanium sponge is calculated by the following formula:
100-(the volume calculated from the mass)/(the apparent volume).times.100.
[0032] (7) A method for manufacturing a titanium plated product according to one embodiment of the present disclosure includes an electrolyzing step of carrying out a molten salt electrolysis by using a cathode and an anode provided in a titanium plating solution containing fluorine and titanium so as to electrodeposit titanium on the surface of the cathode, and the titanium plating solution is prepared by the method for preparing a titanium plating solution according to any one of the above (1) to (6).
[0033] According to the embodiment described in the above (7), the method for manufacturing a titanium plated product may be used to produce a titanium plated product with a smooth titanium plating film formed on its surface.
[0034] (8) In the method for manufacturing a titanium plated product described in the above (7), the titanium plating solution used in the electrolyzing step is measured by cyclic voltammetry under the following conditions, and the potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is controlled to be 0.75 V or more,
[0035] conditions: when the temperature of the titanium plating solution is 650.degree. C. or more and 850.degree. C. or less and when glassy carbon is used as a working electrode, platinum is used as a pseudo-reference electrode and titanium is used as a counter electrode, the potential scanning is repeatedly performed on the working electrode for at least five times at a scanning speed of 1 mV/sec or more and 500 mV/sec or less between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
[0036] According to the embodiment described in the above (8), the method for manufacturing a titanium plated product may be used to continuously and stably produce a titanium plated product with a smooth titanium plating film formed on its surface.
Details of Embodiment of the Present Disclosure
[0037] Specific examples of the method for preparing a titanium plating solution and the method for manufacturing a titanium plated product according to an embodiment of the present disclosure will be described hereinafter in more detail. It should be noted that the present invention is not limited to the specific examples but defined by the scope of the claims, and it is intended that the present invention encompasses all modifications equivalent in meaning and scope to the claims.
Method for Preparing Titanium Plating Solution
[0038] In the method for preparing a titanium plating solution according to an embodiment of the present disclosure, first, a titanium plating solution containing fluorine and titanium is prepared. Then, the titanium plating solution is measured by cyclic voltammetry (hereinafter, abbreviated to "CV" where necessary), and titanium is added to the titanium plating solution so that the potential difference between the spontaneous potential and the Ti3.sup.+/Ti.sup.4+ redox potential is 0.75 V or more.
[0039] The CV measurement may be carried out in a non-oxidizing atmosphere which does not react with titanium to form a compound. For example, the CV measurement may be carried out in an inert gas atmosphere such as argon gas. The CV measurement may be carried out under such a condition that the temperature is set to 650.degree. C. or more and 850.degree. C. or less so as to maintain the titanium plating solution in liquid state and the scanning speed of the potential scanning is set at 1 mV/sec or more and 500 mV/sec or less. From the viewpoint of preventing the conductivity of the titanium plating solution from decreasing, the temperature of the titanium plating solution is more preferably 650.degree. C. or more and 850.degree. C. or less, and further preferably 650.degree. C. or more and 750.degree. C. or less. From the viewpoint of shortening the measurement time or increasing the measurement accuracy, the scanning speed of the potential scanning is more preferably 50 mV/sec or more and 300 mV/sec or less, and further preferably 100 mV/sec or more and 200 mV/sec or less.
[0040] The working electrode may be, for example, graphite, glassy carbon or the like.
[0041] The reference electrode may be, for example, Pt, Ni or the like.
[0042] The counter electrode may be, for example, titanium, glassy carbon, graphite or the like.
[0043] In the CV measurement, the potential scanning is repeatedly performed on the working electrode for at least five times between a lower potential limit which is the immersion potential of the working electrode and an upper potential limit which is a potential that is 2 V to 4 V higher than the lower potential limit.
[0044] FIG. 1 illustrates the CV measurement result of a titanium plating solution. In FIG. 1, the vertical axis represents the current (mA) and the horizontal axis represents the potential (V) of the reference electrode.
[0045] The spontaneous potential 1 refers to a potential difference between the working electrode and the reference electrode when no current is flowing therethrough.
[0046] The Ti.sup.3+/Ti.sup.4+ redox potential 4 refers to a midpoint potential between the peak potential 2 resulting from the oxidation of Ti.sup.3+ to Ti.sup.4+ and the peak potential 3 resulting from the reduction of Ti.sup.4+ to Ti.sup.3+. The peak potential 2 resulting from the oxidation of Ti.sup.3+ to Ti.sup.4+ is an average value of the potentials obtained by repeating the potential scanning on the working electrode for at least five times. Likewise, the peak potential 3 resulting from the reduction of Ti.sup.4+ to Ti.sup.3+ is an average value of the potentials obtained by repeating the potential scanning on the working electrode for at least five times.
[0047] When a titanium plating solution has a potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 of 0.75 V or more, the concentration of Ti.sup.3+ in the titanium plating solution is much greater than the concentration of Ti.sup.4+. Therefore, if the molten salt electrolysis is carried out by using a titanium plating solution having a potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 of 0.75 V or more, a titanium plating film which is silvery white and highly smooth can be formed on the surface of the cathode. On the other hand, when the molten salt electrolysis is carried out by using a titanium plating solution having a potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 of less than 0.75 V, a titanium plating film cannot be formed on the surface of the cathode. From the viewpoint of forming a smooth titanium plating film, the potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 is more preferably 1.0 V or more, and further preferably 1.1 V or more.
[0048] If the potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 is known, the ratio between the concentration of Ti.sup.3+ and the concentration of Ti.sup.4+ in the titanium plating solution may be calculated by using the Nernst equation represented by the following formula (B):
E=E.sub.0-(RT/zF)ln(a.sub.Ti3+/a.sub.Ti4+) Equation (B):
wherein E: electrode potential; E.sub.0: standard electrode potential; R: gas constant; T: absolute temperature; Z: number of mobile electrons; F: Faraday constant; and a: activity.
[0049] When using the Nernst equation to calculate the ratio between the concentration of Ti.sup.3+ and the concentration of Ti.sup.4+, it is assumed that the electrode potential E in the formula (B) is dominantly affected by a reaction of oxidizing Ti.sup.3+ to Ti.sup.4+ and a reaction of reducing Ti.sup.4+ to Ti.sup.3+, and it is also assumed that the ratio between the activity of Ti.sup.3+ and the activity of Ti.sup.4+ (Ti.sup.3+ activity/Ti.sup.4+ activity) is the same as the ratio between the concentration of Ti.sup.3+ and the concentration of Ti.sup.4+ (Ti.sup.3+ concentration/Ti.sup.4+ concentration).
[0050] The titanium plating solution before the CV measurement may be a molten salt containing fluorine and titanium. For example, the titanium plating solution may be a molten salt obtained by dissolving K.sub.2TiF.sub.6 in KF--KCl, a molten salt obtained by dissolving K.sub.2TiF.sub.6 in LiF--KCl, or a molten salt obtained by dissolving K.sub.2TiF.sub.6 in NaF--KCl. The titanium compound to be dissolved in the molten salt is not limited to K.sub.2TiF.sub.6, it may be TiCl.sub.4 or the like. Among the molten salts mentioned above, the molten salt obtained by dissolving K.sub.2TiF.sub.6 in KF--KCl is preferable. The molten salt obtained by dissolving K.sub.2TiF.sub.6 in KF--KCl is a titanium plating solution that may be used to form a smooth titanium plating film.
[0051] When KF--KCl is used in the molten salt, the molar mixing ratio between KF and KCl is preferably 10:90 to 90:10. If the content ratio of KF in KF--KCl is 10 mol % or more, a smooth titanium plating film may be electrodeposited on the surface of the cathode. If the content ratio of KF in KF--KCl is 90 mol % or less, the melting point may be made lower than that of the molten salt of KF alone. From these viewpoints, the molar mixing ratio between KF and KCl is more preferably 20:80 to 80:20, and further preferably 40:60 to 60:40.
[0052] When carrying out the CV measurement on the titanium plating solution made of the molten salt, if the potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 is less than 0.75 V, titanium should be added to the titanium plating solution so that the potential difference between the spontaneous potential 1 and the Ti.sup.3+/Ti.sup.4+ redox potential 4 is 0.75 V or more.
[0053] Although the form of Ti to be added to the titanium plating solution is not particularly limited, it is preferable to use titanium sponge, titanium powder which is processed as fine as possible or the like. Since titanium sponge has a higher porosity, the specific surface area is larger, which makes it easier to be dissolved in the molten salt bath. Thus, the porosity of titanium sponge to be used is more preferably 20% or more, and further preferably 40% or more. Using titanium sponge which has a higher porosity may facilitate the progress of the comproportionation reaction in the titanium plating solution.
[0054] If the titanium plating solution obtained by the method for preparing a titanium plating solution according to an embodiment of the present disclosure is used to carry out the molten salt electrolysis, it is possible to manufacture a titanium plated product with a smooth titanium plating film having a small thickness distribution on its surface.
Method for Manufacturing Titanium Plated Product
[0055] A method for manufacturing a titanium plated product according to an embodiment of the present disclosure includes an electrolyzing step of carrying out a molten salt electrolysis by using a cathode and an anode provided in the titanium plating solution which is obtained according to the method for preparing a titanium plating solution described in the above embodiment so as to electrodeposit titanium on the surface of the cathode.
Cathode
[0056] In the electrolyzing step, a titanium plating film will be formed on the surface of the cathode. Thus, a material suitable for forming the titanium plating film on the surface may be used as the cathode. As an example, a metal, a conductive sintered body or the like may be given. Specifically, nickel, iron, SUS304, molybdenum, tungsten, copper, carbon or the like may be preferably used.
[0057] The base material used as the cathode is simply required to be conductive at least at its surface. If the base material is made of a material to be alloyed with titanium, a titanium alloy layer may be formed on the cathode side of the titanium plating film. On the other hand, if a high-purity titanium plating film is to be formed without a titanium alloy layer, a material that cannot be alloyed with Ti in the titanium plating solution may be used as the cathode.
Anode
[0058] The anode is not particularly limited, and it may be made of any conductive material such as glassy carbon or titanium, for example. From the viewpoint of stably and continuously producing the titanium plating film, the anode is preferably made of Ti.
Other Conditions
[0059] The atmosphere in which the molten salt electrolysis is carried out may be vacuum or a non-oxidative atmosphere that does not form a compound with titanium. For example, the molten salt electrolysis may be carried out in a glove box filled or circulated with an inert gas such as argon gas.
[0060] The current density for carrying out the molten salt electrolysis is not particularly limited, and it may be, for example, 10 mA/cm.sup.2 or more and 500 mA/cm.sup.2 or less. By setting the current density to 10 mA/cm.sup.2 or more, it is possible to stably form a titanium plating film on the surface of the cathode. By setting the current density to 500 mA/cm.sup.2 or less, the diffusion of titanium ions in the titanium plating solution is not a rate-limiting factor, and thus the resulting titanium plating film can be prevented from becoming black. From these viewpoints, the current density is more preferably 50 mA/cm.sup.2 or more and 250 mA/cm.sup.2 or less, and further preferably 100 mA/cm.sup.2 or more and 200 mA/cm.sup.2 or less.
[0061] In the electrolyzing step, the temperature of the titanium plating solution is preferably 650.degree. C. or more and 850.degree. C. or less. If the temperature of the titanium plating solution is set to 650.degree. C. or more, it is possible to maintain the titanium plating solution in liquid state so as to carry out the molten salt electrolysis stably. If the temperature of the titanium plating solution is set to 850.degree. C. or less, it is possible to prevent the titanium plating solution from becoming unstable due to the evaporation of the components in the titanium plating solution. From these viewpoints, the temperature of the titanium plating solution is more preferably 650.degree. C. or more and 750.degree. C. or less, and further preferably 650.degree. C. or more and 700.degree. C. or less.
[0062] The time for carrying out the molten salt electrolysis is not particularly limited, and it may be carried out for a period of time in which the target titanium plating film is sufficiently formed on the surface of the cathode.
CV Measurement in Electrolyzing Step
[0063] In the electrolyzing step, it is preferable that the titanium plating solution is measured regularly or irregularly by cyclic voltammetry and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential is controlled to be 0.75 V or more.
[0064] If oxygen or water is mixed into the titanium plating solution from the external environment for some reasons during the electrolyzing step, the titanium ions may be oxidized from Ti.sup.3+ to Ti.sup.4+, and thereby a smooth titanium plating film may not be formed. Further, when an electrode other than the titanium electrode is used as the anode, the concentration of the titanium ions in the titanium plating solution may change at anytime.
[0065] Even under these circumstances, the titanium plating solution is subjected to CV measurement regularly or irregularly and controlled so that the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential is 0.75 V or more, which makes it possible to stably and continuously form a smooth titanium plating film on the surface of the cathode.
[0066] The conditions for the CV measurement are the same as those for the CV measurement in the method for preparing a titanium plating solution according to the embodiment of the present disclosure described above. When the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential of the titanium plating solution determined by the CV measurement becomes less than 0.75 V, for example, titanium sponge or the like may be added and dissolved in the titanium plating solution.
[0067] According to the method for manufacturing a titanium plated product according to the embodiment of the present disclosure, it is possible to manufacture a titanium plated product with a smooth titanium plating film having a small thickness distribution on its surface. A smooth titanium plating film having a small film thickness distribution refers to such a film that either the maximum thickness or the minimum thickness of the titanium plating film measured at any of arbitrary five spots is preferably within .+-.50% of the average film thickness.
[0068] The average film thickness of the titanium plating film is measured in the following manner. FIG. 2 is a conceptual diagram for illustrating a method for measuring the average film thickness.
[0069] First, the titanium plated product with a titanium plating film formed on its surface is arbitrarily and equally divided into areas, and five spots (area A to area E) are selected as measurement spots. Then, the cross section of the titanium plating film in each area is observed with a scanning electron microscope (SEM). The magnifying power of the SEM is set in such a manner that the entire titanium plating film can be observed in the thickness direction and is enlarged in the thickness direction as much as possible in one field of view. The maximum thickness and the minimum thickness of the titanium plating film in each area are measured at three points by changing the field of view, and the average value is defined as the average film thickness of the titanium plating film.
[0070] As an example, FIG. 2 illustrates a schematic view of a titanium plated product 21 with a titanium plating film formed on the surface of a substantially square-shaped base material, in which four corners are defined as areas A to D, and a central portion is defined as an area E. FIG. 3 illustrates a conceptual view of a field of view (i) when the area A of the titanium plated product 21 illustrated in FIG. 2 is observed by SEM. Similarly, FIG. 4 illustrates a conceptual view of a field of view (ii) of the area A, and FIG. 5 illustrates a conceptual view of a field of view (iii) of the area A.
[0071] In each field of view (i) to (iii) for the area A of the titanium plated product 21 observed by SEM, the maximum thickness of the titanium plating film 23 (the maximum thickness A(i), the maximum thickness A(ii), and the maximum thickness A(iii)) and the minimum thickness of the titanium plating film 23 (the minimum thickness a(i), the minimum thickness a(ii), and the minimum thickness a(iii)) are measured. The thickness of the titanium plating film 23 is defined as the length of the titanium plating film 23 extending in the vertical direction from the base material 22. In the case where a titanium alloy layer made of titanium and a base metal is formed between the titanium plating film 23 and the base material 22, the thickness of the titanium plating film 23 is defined as the total length of the titanium alloy layer and the titanium plating film extending in the vertical direction from the base material 22. Thus, in the area A, the maximum thickness A(i) to A(iii) and the minimum thickness a(i) to a(iii) are determined in three fields of view. Regarding the areas B, C, D and E, the maximum thickness and the minimum thickness of the titanium plating film are measured in three fields of view in the same manner as the area A.
[0072] Thus, the average of the maximum thickness A(i) to A(iii), B(i) to B(iii), C(i) to C(iii), D(i) to D(iii) and E(i) to E(iii) and the minimum thickness a(i) to a(iii), b(i) to b(iii), c(i) to c(iii), d(i) to d(iii) and e(i) to e(iii) of the titanium plating film measured as described above are averaged, and the average value is defined as the average film thickness of the titanium plating film.
EXAMPLES
[0073] Hereinafter, the present disclosure will be described in more detail by examples. It should be noted that the examples are illustrative, and the method for preparing a titanium plating solution and the method for manufacturing a titanium plated product of the present invention are not limited to the examples. The scope of the present invention is defined by the scope of the claims, and encompasses all modifications equivalent in meaning and scope to the claims.
Example 1
Preparation of Titanium Plating Solution
[0074] KCl, KF and K.sub.2TiF.sub.6 were mixed so that the molar mixing ratio between KCl and KF was 55:45 and the concentration of K.sub.2TiF.sub.6 was 0.1 mol %, and were heated to 650.degree. C. to prepare a titanium plating solution.
[0075] While the obtained titanium plating solution was maintained at 650.degree. C., the CV measurement was carried out at a potential scanning speed of 200 mV/sec under an atmosphere in which argon gas was circulated. A graphite rod with a diameter of 3 mm was used as a working electrode, a Pt wire with a diameter of 1 mm was used as a reference electrode, and a titanium rod with a diameter of 3 mm was used as a counter electrode. The potential scanning was repeatedly performed on the working electrode for five times.
[0076] According to the CV measurement, the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential of the titanium plating solution was 0.65 V.
[0077] Therefore, 0.3 mg of titanium sponge per 1 g of the titanium plating solution was added to the titanium plating solution, and sufficiently dissolved therein. The used titanium sponge has a porosity of 50%.
[0078] Again, the titanium plating solution was subjected to the CV measurement under the same conditions, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 0.75 V. This titanium plating solution was used as the titanium plating solution No. 1.
Manufacture of Titanium Plated Product
[0079] A cathode and an anode were provided in the titanium plating solution No. 1, and the molten salt electrolysis was carried out for 40 minutes.
[0080] The molten salt electrolysis was carried out in a glove box under an argon flow atmosphere. A Ni plate of 0.5 cm.times.2.5 cm.times.0.1 mm was used as the cathode, and a Ti rod was used as the anode. A Pt wire was used as a pseudo-reference electrode. The current density was set to 25 mA/cm.sup.2. The potential of the pseudo-reference electrode was calibrated with the potential (K.sup.+/K potential) of metallic potassium electrochemically deposited on the Pt wire.
[0081] As a result, titanium was electrodeposited on the surface of the Ni plate serving as the cathode, and a titanium plated product with a titanium plating film formed on the surface was obtained.
[0082] After the molten salt electrolyzing step, the titanium plated product was washed with water. The salt that adhered to the surface of the titanium plated product was highly soluble in water and was easily removed. Through the above-described operation, the titanium plated product No. 1 with a titanium plating film formed on its surface was obtained.
Example 2
Preparation of Titanium Plating Solution
[0083] Titanium plating solution No. 2 was prepared in the same manner as Example 1 except that the amount of titanium sponge added to the titanium plating solution after the CV measurement in Example 1 was modified to 0.5 mg of titanium sponge per 1 g of the titanium plating solution. The titanium plating solution No. 2, to which titanium sponge was added, was subjected to the CV measurement under the same conditions as Example 1, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 0.85 V.
Manufacture of Titanium Plated Product
[0084] Titanium plated product No. 2 was manufactured in the same manner as Example 1 except that the titanium plating solution No. 2 was used in place of the titanium plating solution No. 1 of Example 1.
Example 3
Preparation of Titanium Plating Solution
[0085] Titanium plating solution No. 3 was prepared in the same manner as Example 1 except that the amount of titanium sponge added to the titanium plating solution after the CV measurement in Example 1 was modified to 1 mg of titanium sponge per 1 g of the titanium plating solution. The titanium plating solution No. 3, to which titanium sponge was added, was subjected to the CV measurement under the same conditions as Example 1, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 1.00 V.
Manufacture of Titanium Plated Product
[0086] Titanium plated product No. 3 was manufactured in the same manner as Example 1 except that the titanium plating solution No. 3 was used in place of the titanium plating solution No. 1 of Example 1.
Example 4
Preparation of Titanium Plating Solution
[0087] Titanium plating solution No. 4 was prepared in the same manner as Example 1 except that the amount of titanium sponge added to the titanium plating solution after the CV measurement in Example 1 was modified to 1.2 mg of titanium sponge per 1 g of the titanium plating solution. The titanium plating solution No. 4, to which titanium sponge was added, was subjected to the CV measurement under the same conditions as Example 1, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 1.10 V.
[0088] FIG. 6 illustrates the result of the CV measurement (the result of the fifth potential scanning) for the titanium plating solution No. 4. In FIG. 6, the vertical axis represents the current (mA), and the horizontal axis represents the potential (V) of the reference electrode.
Manufacture of Titanium Plated Product
[0089] Titanium plated product No. 4 was manufactured in the same manner as Example 1 except that the titanium plating solution No. 4 was used in place of the titanium plating solution No. 1 of Example 1.
Comparative Example 1
Preparation of Titanium Plating Solution
[0090] Titanium plating solution No. A was prepared in the same manner as Example 1 except that titanium sponge was not added to the titanium plating solution after the CV measurement in Example 1. The titanium plating solution No. A was subjected to the CV measurement under the same conditions as Example 1, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 0.67 V. FIG. 7 illustrates the result of the CV measurement (the result of the fifth potential scanning) for the titanium plating solution No. A. In FIG. 7, the vertical axis represents the current (mA), and the horizontal axis represents the potential (V) of the reference electrode.
Manufacture of Titanium Plated Product
[0091] Titanium plated product No. A was manufactured in the same manner as Example 1 except that the titanium plating solution No. A was used in place of the titanium plating solution No. 1 of Example 1.
Comparative Example 2
Preparation of Titanium Plating Solution
[0092] Titanium plating solution No. B was prepared in the same manner as Example 1 except that the amount of titanium sponge added to the titanium plating solution after the CV measurement in Example 1 was modified to 0.2 mg of titanium sponge per 1 g of the titanium plating solution. The titanium plating solution No. B, to which titanium sponge was added, was subjected to the CV measurement under the same conditions as Example 1, and the potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential was 0.70 V.
Manufacture of Titanium Plated Product
[0093] Titanium plated product No. B was manufactured in the same manner as Example 1 except that the titanium plating solution No. B was used in place of the titanium plating solution No. 1 of Example 1.
Evaluation
[0094] The ratio between the concentration of Ti.sup.3+ and the concentration of Ti.sup.4+ (Ti.sup.3+ concentration/Ti.sup.4+ concentration) in each of the titanium plating solution No. 1 obtained in Example 1, the titanium plating solution No. 2 obtained in Example 2, the titanium plating solution No. 3 obtained in Example 3, the titanium plating solution No. 4 obtained in Example 4, the titanium plating solution No. A obtained in Comparative Example 1, and the titanium plating solution No. B obtained in Comparative Example 2 was calculated by using the Nernst equation. The results are listed in Table 1.
[0095] In addition, the surface condition of each of the titanium plated product No. 1 obtained in Example 1, the titanium plated product No. 2 obtained in Example 2, the titanium plated product No. 3 obtained in Example 3, the titanium plated product No. 4 obtained in Example 4, the titanium plated product No. A obtained in Comparative Example 1, and the titanium plated product No. B obtained in Comparative Example 2 was visually observed. The results are listed in Table 1.
[0096] The current efficiency of the cathode in the electrolyzing step of each of Examples 1 to 4 and Comparative Examples 1 and 2 was calculated by using the following formula (C). The results are listed in Table 1.
Current efficiency (%)=(actual plating amount)/(theoretical plating amount).times.100 Formula (C):
TABLE-US-00001 TABLE 1 Titanium plating solution Difference (V) between Titanium plated product spontaneous Ti.sup.3+/Ti.sup.4+ Surface Current potential and Concen- condition efficiency Ti.sup.3+/Ti.sup.4- tration of titanium of cathode No. redox potential ratio No. plating film (%) 1 0.75 10000 1 silvery white 20 2 0.85 40000 2 silvery white 30 3 1.00 290000 3 silvery white 90 4 1.10 1000000 4 silvery white 90 A 0.67 3500 A black plating impossible B 0.70 7000 B black plating impossible
[0097] As listed in Table 1, when the molten salt electrolysis was carried out by using the titanium plating solutions No. 1 to No. 4 having a potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential of 0.75 V or more, it is possible to obtain the titanium plated products No. 1 to No. 4 each with a silvery white and smooth titanium plating film formed on its surface. In particular, when the titanium plating solution No. 3 or No. 4 having a potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential of 1.00 V or more was used, the current efficiency of the cathode could be improved to 90% or more.
[0098] On the other hand, when the molten salt electrolysis was carried out by using the titanium plating solution No. A or No. B having a potential difference between the spontaneous potential and the Ti.sup.3+/Ti.sup.4+ redox potential of less than 0.75 V, a titanium plating film could not be successfully formed on the cathode surface, and the electrodeposited film was black.
REFERENCE SIGNS LIST
[0099] 1: spontaneous potential; 2: peak potential resulting from the oxidation of Ti.sup.3+ to Ti.sup.4+; 3: peak potential resulting from the reduction of Ti.sup.4+ to Ti.sup.3+; 4: Ti.sup.3+/Ti.sup.4+ redox potential; A: arbitrary area on titanium plated product; B: arbitrary area on titanium plated product; C: arbitrary area on titanium plated product; D: arbitrary area on titanium plated product; E: arbitrary area on titanium plated product; 21: titanium plated product; 22: base material; 23: titanium plating film; 61: spontaneous potential; 64: Ti.sup.3+/Ti.sup.4+ redox potential; 71: spontaneous potential; 74: Ti.sup.3+/Ti.sup.4+ redox potential
* * * * *
D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.