Azaindoline Compounds As Granzyme B Inhibitors
Cameron; Dale R.
U.S. patent application number 16/372255 was filed with the patent office on 2020-02-27 for azaindoline compounds as granzyme b inhibitors. This patent application is currently assigned to viDA Therapeutics Inc.. The applicant listed for this patent is viDA Therapeutics Inc.. Invention is credited to Dale R. Cameron.
| Application Number | 20200062803 16/372255 |
| Document ID | / |
| Family ID | 55216535 |
| Filed Date | 2020-02-27 |











View All Diagrams
| United States Patent Application | 20200062803 |
| Kind Code | A1 |
| Cameron; Dale R. | February 27, 2020 |
AZAINDOLINE COMPOUNDS AS GRANZYME B INHIBITORS
Abstract
Azaindoline compounds as granzyme B inhibitors, compositions that include the compounds, and methods for using the compounds. Methods for treating cutaneous scleroderma, epidermolysis bullosa, radiation dermatitis, alopecia areata, and discoid lupus erythematosus are provided.
| Inventors: | Cameron; Dale R.; (Richmond, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | viDA Therapeutics Inc. North Vancouver CA |
||||||||||
| Family ID: | 55216535 | ||||||||||
| Appl. No.: | 16/372255 | ||||||||||
| Filed: | April 1, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15500781 | Jan 31, 2017 | 10246487 | ||
| PCT/CA2015/050724 | Jul 31, 2015 | |||
| 16372255 | ||||
| 62032471 | Aug 1, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/06 20130101; A61Q 7/00 20130101; C07K 5/0812 20130101; A61K 8/64 20130101; A61K 38/00 20130101; C07K 5/101 20130101; C07K 5/0202 20130101; C07K 5/06104 20130101; C07K 5/0808 20130101; A61P 17/00 20180101; A61K 38/05 20130101 |
| International Class: | C07K 5/103 20060101 C07K005/103; A61K 38/06 20060101 A61K038/06; A61K 38/05 20060101 A61K038/05; C07K 5/02 20060101 C07K005/02; C07K 5/087 20060101 C07K005/087; C07K 5/083 20060101 C07K005/083; C07K 5/072 20060101 C07K005/072; A61K 8/64 20060101 A61K008/64; A61Q 7/00 20060101 A61Q007/00 |
Claims
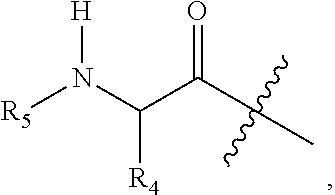
1. A compound having Formula (I): ##STR00087## its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is a heteroaryl group selected from (a) 1,2,3-triazolyl, and (b) 1,2,3,4-tetrazolyl; n is 1 or 2; R.sub.2a and R.sub.2b are independently selected from hydrogen and C1-C6 alkyl; R.sub.2c at each occurrence is independently selected from (a) hydrogen, (b) halogen, (c) C.sub.1-C.sub.6 alkyl, (d) --XR.sub.11, wherein X is selected from O, C(.dbd.O), S, S.dbd.O, or S(.dbd.O).sub.2, (e) --C(.dbd.O)N(R.sub.12)(R.sub.13), (f) --N(R.sub.11)(R.sub.12)(R.sub.13), (g) --N--C(.dbd.O)--R.sub.11, and (h) --N--C(.dbd.O)O--R.sub.11, wherein R.sub.11, R.sub.12, and R.sub.13 are independently selected from the group consisting of hydrogen, C.sub.1-C.sub.6 alkyl, C.sub.1-C.sub.6 heteroalkyl, C.sub.2-C.sub.6 alkenyl, C.sub.6-C.sub.10 aryl, aralkyl, and C.sub.3-C.sub.10 heteroaryl; m is 1, 2, or 3; R.sub.3 is selected from (a) hydrogen, (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2.sup.-, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; Z is an acyl group selected from the group (a) ##STR00088## and (b) ##STR00089## wherein Y is hydrogen, heterocycle, --NH.sub.2, or C.sub.1-C.sub.4 alkyl; R.sub.4 is selected from (i) C.sub.1-C.sub.12 alkyl, (ii) C.sub.1-C.sub.6 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, (iii) C.sub.3-C.sub.6 cycloalkyl, (iv) C.sub.6-C.sub.10 aryl, (v) heterocyclyl, (vi) C.sub.3-C.sub.10 heteroaryl, (vii) aralkyl, and (viii) heteroalkylaryl; R.sub.5 is heteroaryl or --C(.dbd.O)--R.sub.10, wherein R.sub.10 is selected from (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid, (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid, (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (v) heterocyclyl, (vi) C.sub.3-C.sub.10 heteroaryl, (vii) aralkyl, and (viii) heteroalkylaryl.
2. The compound of claim 1, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is a heteroaryl group selected from (a) 1,2,3-triazolyl, and (b) 1,2,3,4-tetrazolyl; n is 1; R.sub.2a, R.sub.2b, and R.sub.2c are hydrogen; R.sub.3 is selected from (a) hydrogen, (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2.sup.-, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; Z is an acyl group selected from the group (a) ##STR00090## and (b) ##STR00091##
3. The compound of claim 1, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; n is 1; R.sub.3 is hydrogen, C.sub.1-C.sub.4 alkyl substituted with a carboxylic acid or carboxylate group, C.sub.1-C.sub.4 alkyl substituted with an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; and Z is ##STR00092##
4. The compound of claim 1, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; n is 1; R.sub.3 is hydrogen, or C.sub.1-C.sub.4 alkyl substituted with a carboxylic acid or carboxylate group, an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; and Z is ##STR00093## wherein R.sub.4 is selected from (i) C.sub.1-C.sub.12 alkyl, (ii) C.sub.3-C.sub.6 cycloalkyl, (iii) C.sub.6-C.sub.10 aryl, and (iv) C.sub.3-C.sub.10 heteroaryl; R.sub.5 is --C(.dbd.O)--R.sub.10, wherein R.sub.10 is selected from (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid, (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid, (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (v) C.sub.3-C.sub.10 heteroaryl; and Y is hydrogen, C.sub.1-C.sub.4 alkyl, or --NH.sub.2.
5. A compound having Formula (II): ##STR00094## its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is a heteroaryl group selected from (a) 1,2,3-triazolyl, and (b) 1,2,3,4-tetrazolyl; R.sub.3 is selected from (a) hydrogen, (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2--, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; R.sub.4 is selected from (i) C.sub.1-C.sub.12 alkyl, (ii) C.sub.1-C.sub.6 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, (iii) C.sub.3-C.sub.6 cycloalkyl, (iv) C.sub.6-C.sub.10 aryl, (v) heterocyclyl, (vi) C.sub.3-C.sub.10 heteroaryl, (vii) aralkyl, and (viii) heteroalkylaryl; and R.sub.10 is selected from (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid, (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid, (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (v) heterocyclyl, (vi) C.sub.3-C.sub.10 heteroaryl, (vii) aralkyl, and (viii) heteroalkylaryl.
6. The compound of claim 5, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.10, when defined as C.sub.1-C.sub.12 alkyl substituted with a carboxylic acid or carboxylate group, is: --(CH.sub.2).sub.n--CO.sub.2H, where n is 2, 3, 4, 5, or 6; optionally wherein one or more single methylene carbons are substituted with a fluoro, hydroxy, amino, C.sub.1-C.sub.3 alkyl, or C.sub.6-C.sub.10 aryl group; optionally wherein one or more single methylene carbons are substituted with two fluoro or C.sub.1-C.sub.3 alkyl groups; optionally wherein one or more single methylene carbons are substituted with two alkyl groups that taken together with the carbon to which they are attached form a 3, 4, 5, or 6-membered carbocyclic ring; and optionally wherein adjacent carbon atoms from an unsaturated carbon-carbon bond or taken form a benzene ring; or
7. The compound of claim 5, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: wherein R.sub.10, when defined as C.sub.3-C.sub.6 cycloalkyl substituted with a carboxylic acid or carboxylate group, is: ##STR00095## wherein n is 1, 2, 3, or 4; and optionally, for n=3 or 4, wherein adjacent carbon atoms from an unsaturated carbon-carbon bond.
8. The compound of claim 5, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; R.sub.3 is hydrogen; C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; or C.sub.1-C.sub.4 alkyl optionally substituted with an amide, which may be optionally substituted with an alkylheteroaryl group; R.sub.4 is C.sub.1-C.sub.12 alkyl, C.sub.3-C.sub.6 cycloalkyl, C.sub.6-C.sub.10 aryl, C.sub.3-C.sub.10 heteroaryl, or heterocyclyl; and R.sub.10 is C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid.
9. The compound of claim 5, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; R.sub.3 is C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; R.sub.4 is C.sub.1-C.sub.8 alkyl or C.sub.3-C.sub.6 cycloalkyl; and R.sub.10 is selected from: (a) C.sub.1-C.sub.3 alkyl substituted with C.sub.6-C.sub.10 aryl (e.g., phenyl) or C.sub.1-C.sub.10 heteroaryl (e.g., triazolyl or tetrazolyl); (b) --(CH.sub.2).sub.n--CO.sub.2H, where n is 2, 3, 4, 5, or 6; (c) ##STR00096## wherein n is 1, 2, 3, or 4.
10. A compound having Formula (III): ##STR00097## its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein R.sub.1 is a heteroaryl group selected from (a) 1,2,3-triazolyl, and (b) 1,2,3,4-tetrazolyl; R.sub.3 is selected from (a) hydrogen, (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2--, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; Y is hydrogen, heterocycle, --NH.sub.2, or C.sub.1-C.sub.4 alkyl; and R.sub.4 is selected from (i) C.sub.1-C.sub.12 alkyl, (ii) C.sub.1-C.sub.6 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, (iii) C.sub.3-C.sub.6 cycloalkyl, (iv) C.sub.6-C.sub.10 aryl, (v) heterocyclyl, (vi) C.sub.3-C.sub.10 heteroaryl, (vii) aralkyl, and (viii) heteroalkylaryl.
11. The compound of claim 10, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; R.sub.3 is hydrogen; C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; or C.sub.1-C.sub.4 alkyl optionally substituted with an amide, which may be optionally substituted with an alkylheteroaryl group; R.sub.4 is C.sub.1-C.sub.12 alkyl, C.sub.3-C.sub.6 cycloalkyl, C.sub.6-C.sub.10 aryl, C.sub.3-C.sub.10 heteroaryl, or heterocyclyl; and Y is hydrogen, C.sub.1-C.sub.4 alkyl, or --NH.sub.2.
12. The compound of claim 10, its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein: R.sub.1 is tetrazole or triazole; R.sub.3 is C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; R.sub.4 is selected from (i) C.sub.1-C.sub.8 alkyl, (ii) C.sub.3-C.sub.6 cycloalkyl, (iii) C.sub.6-C.sub.10 aryl, (iv) C.sub.3-C.sub.10 heteroaryl, and (v) heterocyclyl; and Y is hydrogen.
13. A compound as shown in Table 1.
14. A pharmaceutical composition, comprising a compound of any one of claims 1-13 and a pharmaceutically acceptable carrier.
15. A method for inhibiting Granzyme B in a subject, comprising administering an effective amount of a compound of any one of claims 1-13 or a pharmaceutical composition of claim 14 to a subject in need thereof.
16. A method for treating a disease, disorder, or condition treatable by inhibiting Granzyme B, comprising administering a therapeutically effective amount of a compound of any one of claims 1-13 or a pharmaceutical composition of claim 14 to a subject in need thereof.
17. The method of claim 16, wherein the disease, disorder, or condition treatable by inhibiting Granzyme B is selected from treating dissection, aneurysm, and atherosclerosis.
18. The method of claim 16, wherein the condition treatable by inhibiting Granzyme B is a wound and administering the compound or composition promotes wound healing.
19. The method of any one of claims 15-18, wherein administering the compound or composition comprises topical administration, oral administration, and administration by injection.
20. A method for treating cutaneous scleroderma, epidermolysis bullosa, radiation dermatitis, alopecia areata, or discoid lupus erythematosus, comprising administering a therapeutically effective amount of a compound of any one of claims 1-13 or a pharmaceutical composition of claim 14 to a subject in need thereof.
21. The method of claim 20, wherein administering the compound or compositin comprises topical administration.
Description
CROSS-REFERENCES TO RELATED APPLICATIONS
[0001] This application in a continunation of U.S. application Ser. No. 15/500,781, filed Jan. 31, 2017, which is a National Stage of International Application No. PCT/CA2015/050724, filed Jul. 31, 2015, which claims the benefit of U.S. Provisional Application No. 62/032,471, filed Aug. 1, 2014, the disclosures of which are expressly incorporated herein by reference in their entirety.
FIELD OF THE INVENTION
[0002] The present invention disclosure relates generally to agents for treating diseases, disorders, and conditions treatable by inhibiting Granzyme B, and more specifically to azaindoline compounds that are inhibitors of Granzyme B.
BACKGROUND OF THE INVENTION
[0003] Granzyme B is a pro-apoptotic serine protease found in the granules of cytotoxic lymphocytes (CTL) and natural killer (NK) cells. Granzyme B is released towards target cells, along with the pore-forming protein, perforin, resulting in its perforin-dependent internalization into the cytoplasm and subsequent induction of apoptosis (see, for e.g., Medema et al., Eur. J. Immunol. 27:3492-3498, 1997). However, during aging, inflammation and chronic disease, Granzyme B can also be expressed and secreted by other types of immune (e.g., mast cell, macrophage, neutrophil, and dendritic cells) or non-immune (keratinocyte, chondrocyte) cells and has been shown to possess extracellular matrix remodeling activity (Choy et al., Arterioscler. Thromb. Vasc. Biol. 24(12):2245-2250, 2004 and Buzza et al., J. Biol. Chem. 280:23549-23558, 2005).
[0004] Inhibitors of Granzyme B in humans have been limited to (a) relatively weak, nonspecific inhibitors such as isocoumarins (Odake et al., (1991), Biochemistry, 30(8), 2217-2227); (b) biological inhibitors such as serpinB9 (Sun et al., (1996), J. Biol. Chem., 271(44), 27802-27809); (c) covalently coupled inhibitors such as aldehydes (Willoughby et al., (2002), Bioorg. Med. Chem. Lett., 12(16), 2197), halomethyl ketones (Kam et al., (2000), Biochim. Biophy. Acta, 1477(1-2), 307-323), and phosphonates (Mahrus and Craik, (2005), Chem. & Biol., 12, 567-77 and Kam et al., (2000)); and (d) tricyclic inhibitors (Willoughby et al., (2002)).
[0005] Nonspecific inhibitors (such as isocoumarins) are not sufficiently potent or specific to be effective treatments for Granzyme-B-related diseases, disorders, and conditions. Likewise, the use of biological inhibitors such as serpins is limited by the ability to deliver the inhibitor to the target mammal, the cost of manufacturing the biological agents, and other, off-target activities, such as inhibition of other serine proteases such as human neutrophil elastase (Dahlen et al., (1999), Biochim. Biophys. Acta, 1451(2-3), 233-41), Caspase-1 (Annaud et al., (1999), Biochem. J., September 15; 342 Pt3, 655-65; Krieg et al., (2001), Mol. Endocrinol., 15(11), 1971-82; and Young et al., (2000), J. Exp. Med., 191(9), 1535-1544); Caspase-4 and Caspase-8 (Annaud et al., (1999)).
[0006] The tricyclic inhibitors (Willoughby et al. (2001)) also suffer from synthetic complexity/high manufacturing cost due to the complex core and accompanying low water solubility.
[0007] Despite the advances in development of Granzyme B inhibitors, there exists a need for compounds that inhibit Granzyme B with selectivity, that are relatively simple to manufacture at low cost, and that do not present drug delivery challenges. The present invention seeks to fulfill this need and provides further related advantages.
SUMMARY OF THE INVENTION
[0008] The present invention provides Granzyme B inhibitor compounds, compositions that include the compounds, and methods for using the compounds.
[0009] In one aspect of the invention, the invention provides Granzyme B inhibitor compounds.
[0010] In one embodiment, the invention provides the compounds having Formula (I):
##STR00001##
[0011] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0012] R.sub.1 is a heteroaryl group selected from
[0013] (a) 1,2,3-triazolyl, and
[0014] (b) 1,2,3,4-tetrazolyl;
[0015] n is 1 or 2;
[0016] R.sub.2a and R.sub.2b are independently selected from hydrogen and C1-C6 alkyl;
[0017] R.sub.2c at each occurrence is independently selected from
[0018] (a) hydrogen,
[0019] (b) halogen,
[0020] (c) C.sub.1-C.sub.6 alkyl,
[0021] (d) --XR.sub.11, wherein X is selected from O, C(.dbd.O), S, S.dbd.O, or S(.dbd.O).sub.2,
[0022] (e) --C(.dbd.O)N(R.sub.12)(R.sub.13),
[0023] (f) --N(R.sub.11)(R.sub.12)(R.sub.13),
[0024] (g) --N--C(.dbd.O)--R.sub.1, and
[0025] (h) --N--C(.dbd.O)O--R.sub.11,
[0026] wherein R.sub.11, R.sub.12, and R.sub.13 are independently selected from the group consisting of hydrogen, C.sub.1-C.sub.6 alkyl, C.sub.1-C.sub.6 heteroalkyl, C.sub.2-C.sub.6 alkenyl, C.sub.6-C.sub.10 aryl, aralkyl, and C.sub.3-C.sub.10 heteroaryl;
[0027] m is 1, 2, or 3;
[0028] R.sub.3 is selected from
[0029] (a) hydrogen,
[0030] (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2--, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group;
[0031] Z is an acyl group selected from the group
[0032] (a)
##STR00002##
and
[0033] (b)
##STR00003##
[0034] wherein
[0035] Y is hydrogen, heterocycle, --NH.sub.2, or C.sub.1-C.sub.4 alkyl;
[0036] R.sub.4 is selected from
[0037] (i) C.sub.1-C.sub.12 alkyl,
[0038] (ii) C.sub.1-C.sub.6 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl,
[0039] (iii) C.sub.3-C.sub.6 cycloalkyl,
[0040] (iv) C.sub.6-C.sub.10 aryl,
[0041] (v) heterocyclyl,
[0042] (vi) C.sub.3-C.sub.10 heteroaryl,
[0043] (vii) aralkyl, and
[0044] (viii) heteroalkylaryl;
[0045] R.sub.5 is heteroaryl or --C(.dbd.O)--R.sub.10,
[0046] wherein R.sub.10 is selected from
[0047] (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0048] (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid,
[0049] (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0050] (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0051] (v) heterocyclyl,
[0052] (vi) C.sub.3-C.sub.10 heteroaryl,
[0053] (vii) aralkyl, and
[0054] (viii) heteroalkylaryl.
[0055] In another embodiment, the invention provides compounds having Formula (II):
##STR00004##
[0056] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0057] R.sub.1, R.sub.3, R.sub.4, and R.sub.10 are as above for Formula (I).
[0058] In a further embodiment, the invention provides compounds having Formula (III):
##STR00005##
[0059] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein R.sub.1, R.sub.3, R.sub.4, and Y are as defined above for Formula (I).
[0060] In another aspect, the invention provides pharmaceutical compositions comprising a Granzyme B inhibitor compound of the invention and a pharmaceutically acceptable carrier.
[0061] In a further aspect of the invention, a method for inhibiting Granzyme B is provided. In one embodiment, the method comprises administering an effective amount of a Granzyme B inhibitor compound of the invention or a pharmaceutical composition of the invention to a subject in need thereof.
[0062] In a further aspect of the invention, methods for treating a disease, disorder, or condition treatable by inhibiting Granzyme B is provided. In one embodiment, the method comprises administering a therapeutically effective amount of a Granzyme B inhibitor compound of the invention or a pharmaceutical composition of the invention to a subject in need thereof. Representative routes of administration include topical administration, oral administration, and administration by injection.
[0063] In one embodiment, the invention provides a method for treating discoid lupus erythematosus (DLE) comprising administering a therapeutically effective amount of a Granzyme B inhibitor compound of the invention or a pharmaceutical composition of the invention to a subject in need thereof. In certain embodiments, the Granzyme B inhibitor compound of the invention or pharmaceutical composition is administered topically.
[0064] Cosmetic compositions comprising a Granzyme B inhibitor compound of the invention and a cosmetically acceptable carrier are also provided, as are methods for using the compositions to treat, reduce, and/or inhibit the appearance of ageing in the skin.
BRIEF DESCRIPTION OF THE DRAWINGS
[0065] FIG. 1 is a schematic illustration of a representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from P1.
[0066] FIG. 2 is a schematic illustration of another representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from P5.) FIG. 3 is a schematic illustration of a further representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from a component other than P1 or P5.
DETAILED DESCRIPTION OF THE INVENTION
[0067] The present invention provides Granzyme B inhibitor compounds, compositions that include the compounds, and methods for using the compounds. The compounds of the invention effectively inhibit Granzyme B.
[0068] In one aspect of the invention, the invention provides Granzyme B inhibitor compounds.
[0069] In one embodiment, the invention provides the compounds having Formula (I):
##STR00006##
[0070] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0071] R.sub.1 is a heteroaryl group selected from
[0072] (a) 1,2,3-triazolyl, and
[0073] (b) 1,2,3,4-tetrazolyl;
[0074] n is 1 or 2;
[0075] R.sub.2a and R.sub.2b are independently selected from hydrogen and C1-C6 alkyl;
[0076] R.sub.2c at each occurrence is independently selected from
[0077] (a) hydrogen,
[0078] (b) halogen,
[0079] (c) C.sub.1-C.sub.6 alkyl,
[0080] (d) --XR.sub.11, wherein X is selected from O, C(.dbd.O), S, S.dbd.O, or S(.dbd.O).sub.2,
[0081] (e) --C(.dbd.O)N(R.sub.12)(R.sub.13),
[0082] (f) --N(R.sub.11)(R.sub.12)(R.sub.13),
[0083] (g) --N--C(.dbd.O)--R.sub.1, and
[0084] (h) --N--C(.dbd.O)O--R.sub.11,
[0085] wherein R.sub.11, R.sub.12, and R.sub.13 are independently selected from the group consisting of hydrogen, C.sub.1-C.sub.6 alkyl, C.sub.1-C.sub.6 heteroalkyl, C.sub.2-C.sub.6 alkenyl, C.sub.6-C.sub.10 aryl, aralkyl, and C.sub.3-C.sub.10 heteroaryl;
[0086] m is 1, 2, or 3;
[0087] R.sub.3 is selected from
[0088] (a) hydrogen,
[0089] (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2.sup.-, --C(.dbd.O)OC.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group;
[0090] Z is an acyl group selected from the group
[0091] (a)
##STR00007##
and
[0092] (b)
##STR00008##
[0093] wherein
[0094] Y is hydrogen, heterocycle, --NH.sub.2, or C.sub.1-C.sub.4 alkyl;
[0095] R.sub.4 is selected from
[0096] (i) C.sub.1-C.sub.12 alkyl,
[0097] (ii) C.sub.1-C.sub.6 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl,
[0098] (iii) C.sub.3-C.sub.6 cycloalkyl,
[0099] (iv) C.sub.6-C.sub.10 aryl,
[0100] (v) heterocyclyl,
[0101] (vi) C.sub.3-C.sub.10 heteroaryl,
[0102] (vii) aralkyl, and
[0103] (viii) heteroalkylaryl;
[0104] R.sub.5 is heteroaryl or --C(.dbd.O)--R.sub.10,
[0105] wherein R.sub.10 is selected from
[0106] (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0107] (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid,
[0108] (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0109] (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid, (v) heterocyclyl,
[0110] (vi) C.sub.3-C.sub.10 heteroaryl,
[0111] (vii) aralkyl, and
[0112] (viii) heteroalkylaryl.
[0113] In another embodiment, the invention provides compounds having Formula (I), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0114] R.sub.1 is a heteroaryl group selected from
[0115] (a) 1,2,3-triazolyl, and
[0116] (b) 1,2,3,4-tetrazolyl;
[0117] n is 1;
[0118] R.sub.2a, R.sub.2b, and R.sub.2c are hydrogen;
[0119] R.sub.3 is selected from
[0120] (a) hydrogen,
[0121] (b) C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or carboxylate C.sub.1-C.sub.8 ester group (--CO.sub.2H, --CO.sub.2.sup.-, --CO.sub.2C.sub.1-C.sub.8), an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group;
[0122] Z is an acyl group selected from the group
[0123] (a)
##STR00009##
and
[0124] (b)
##STR00010##
[0125] wherein R.sub.4, R.sub.5, and Y are as described above.
[0126] In further embodiments, the invention provides compounds having Formula (I), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0127] R.sub.1 is tetrazole or triazole; n is 1; R.sub.3 is hydrogen, C.sub.1-C.sub.4 alkyl substituted with a carboxylic acid or carboxylate group, C.sub.1-C.sub.4 alkyl substituted with an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; and Z is
##STR00011##
and
[0128] R.sub.1 is tetrazole or triazole; n is 1; R.sub.3 is hydrogen, or C.sub.1-C.sub.4 alkyl substituted with a carboxylic acid or carboxylate group, an amide optionally substituted with an alkylheteroaryl group, or a heteroaryl group; and Z is
##STR00012##
[0129] wherein
[0130] R.sub.4 is selected from
[0131] (i) C.sub.1-C.sub.12 alkyl,
[0132] (ii) C.sub.3-C.sub.6 cycloalkyl,
[0133] (iii) C.sub.6-C.sub.10 aryl, and
[0134] (iv) C.sub.3-C.sub.10 heteroaryl;
[0135] R.sub.5 is --C(.dbd.O)--R.sub.10, wherein R.sub.10 is selected from
[0136] (i) C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0137] (ii) C.sub.1-C.sub.10 heteroalkyl optionally substituted with C.sub.1-C.sub.6 alkyl or carboxylic acid,
[0138] (iii) C.sub.3-C.sub.6 cycloalkyl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0139] (iv) C.sub.6-C.sub.10 aryl optionally substituted with C.sub.1-C.sub.6 alkyl, optionally substituted C.sub.6-C.sub.10 aryl, optionally substituted C.sub.3-C.sub.10 heteroaryl, amino, or carboxylic acid,
[0140] (v) C.sub.3-C.sub.10 heteroaryl; and
[0141] Y is hydrogen, C.sub.1-C.sub.4 alkyl, or --NH.sub.2.
[0142] In another embodiment, the invention provides compounds having Formula (II):
##STR00013##
[0143] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0144] R.sub.1, R.sub.3, R.sub.4, and R.sub.10 are as above for Formula (I).
[0145] In certain embodiments, R.sub.10, when defined as C.sub.1-C.sub.12 alkyl substituted with a carboxylic acid or carboxylate group, is:
[0146] --(CH.sub.2).sub.n--CO.sub.2H, where n is 2, 3, 4, 5, or 6;
[0147] optionally wherein one or more single methylene carbons are substituted with a fluoro, hydroxy, amino, C.sub.1-C.sub.3 alkyl (e.g., methyl), or C.sub.6-C.sub.10 aryl group;
[0148] optionally wherein one or more single methylene carbons are substituted with two fluoro (e.g., difluoro, perfluoro) or C.sub.1-C.sub.3 alkyl (e.g., gem-dimethyl) groups;
[0149] optionally wherein one or more single methylene carbons are substituted with two alkyl groups that taken together with the carbon to which they are attached form a 3, 4, 5, or 6-membered carbocyclic ring (e.g., spiro groups such as cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl); and
[0150] optionally wherein adjacent carbon atoms from an unsaturated carbon-carbon bond (e.g., alkenyl such as --CH.dbd.CH--) or taken form a benzene ring (e.g., 1,2-, 1,3-, and 1,4-phenylene); or
[0151] wherein R.sub.10, when defined as C.sub.3-C.sub.6 cycloalkyl substituted with a carboxylic acid or carboxylate group, is:
##STR00014##
wherein n is 1, 2, 3, or 4; and optionally, for n=3 or 4, wherein adjacent carbon atoms from an unsaturated carbon-carbon bond (e.g., cyclopentenyl or cyclohexenyl).
[0152] In certain embodiments, the invention provides compounds having Formula (II), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0153] R.sub.1 is tetrazole or triazole;
[0154] R.sub.3 is hydrogen; C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; or C.sub.1-C.sub.4 alkyl optionally substituted with an amide, which may be optionally substituted with an alkylheteroaryl group;
[0155] R.sub.4 is C.sub.1-C.sub.12 alkyl, C.sub.3-C.sub.6 cycloalkyl, C.sub.6-C.sub.10 aryl, C.sub.3-C.sub.10 heteroaryl, or heterocyclyl; and
[0156] R.sub.10 is C.sub.1-C.sub.12 alkyl optionally substituted with C.sub.6-C.sub.10 aryl, C.sub.1-C.sub.10 heteroaryl, amino, or carboxylic acid.
[0157] In further embodiments, the invention provides compounds having Formula (II), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0158] R.sub.1 is tetrazole or triazole;
[0159] R.sub.3 is C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group;
[0160] R.sub.4 is C.sub.1-C.sub.8 alkyl or C.sub.3-C.sub.6 cycloalkyl; and
[0161] R.sub.10 is selected from:
[0162] (a) C.sub.1-C.sub.3 alkyl substituted with C.sub.6-C.sub.10 aryl (e.g., phenyl) or C.sub.1-C.sub.10 heteroaryl (e.g., triazolyl or tetrazolyl);
[0163] (b) --(CH.sub.2).sub.n--CO.sub.2H, where n is 2, 3, 4, 5, or 6;
[0164] (c)
##STR00015##
wherein n is 1, 2, 3, or 4.
[0165] Representative compounds of Formula (II) include A1, C1-C24, C26, C28-C40.
[0166] In a further embodiment, the invention provides compounds having Formula (III):
##STR00016##
[0167] its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein R.sub.1, R.sub.3, R.sub.4, and Y are as defined above for Formula (I).
[0168] In certain embodiments, the invention provides compounds having Formula (III), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0169] R.sub.1 is tetrazole or triazole;
[0170] R.sub.3 is hydrogen; C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group; or C.sub.1-C.sub.4 alkyl optionally substituted with an amide, which may be optionally substituted with an alkylheteroaryl group;
[0171] R.sub.4 is C.sub.1-C.sub.12 alkyl, C.sub.3-C.sub.6 cycloalkyl, C.sub.6-C.sub.10 aryl, C.sub.3-C.sub.10 heteroaryl, or heterocyclyl; and
[0172] Y is hydrogen, C.sub.1-C.sub.4 alkyl, or --NH.sub.2.
[0173] In further embodiments, the invention provides compounds having Formula (III), its stereoisomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
[0174] R.sub.1 is tetrazole or triazole;
[0175] R.sub.3 is C.sub.1-C.sub.4 alkyl optionally substituted with a carboxylic acid, carboxylate, or a carboxylate ester group;
[0176] R.sub.4 is selected from
[0177] (i) C.sub.1-C.sub.8 alkyl (e.g., methyl, ethyl, n-propyl, i-propyl),
[0178] (ii) C.sub.3-C.sub.6 cycloalkyl (i.e., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl),
[0179] (iii) C.sub.6-C.sub.10 aryl (e.g., phenyl),
[0180] (iv) C.sub.3-C.sub.10 heteroaryl (e.g., thiophenyl), and
[0181] (v) heterocyclyl (e.g., morpholinyl); and
[0182] Y is hydrogen.
[0183] Representative compounds of Formula (III) include C20, C25, C27, and C31-C40.
[0184] For the compounds of Formulae (I), (II), or (III), representative substituents R.sub.3 include the following:
##STR00017##
[0185] For the compounds of Formulae (I), (II), or (III), representative substituents R.sub.4 include the following:
##STR00018##
[0186] For the compounds of Formulae (I), (II), or (III), representative substituents R.sub.5 include the following:
##STR00019## ##STR00020## ##STR00021## ##STR00022##
[0187] Each of the inhibitor compounds of the invention contain asymmetric carbon centers and give rise to stereoisomers (i.e., optical isomers such as diastereomers and enantiomers). It will be appreciated that the present invention includes such diastereomers as well as their racemic and resolved enantiomerically pure forms. It will also be appreciated that in certain configurations, the relative stereochemistry of certain groups may be depicted as "cis" or "trans" when absolute stereochemistry is not shown.
[0188] Some of the compounds described herein contain olefinic double bonds, and unless specified otherwise, are meant to include both E and Z geometric isomers.
[0189] Certain of the compounds of the invention may exist in one or more tautomeric forms (e.g., acid or basic forms depending on pH environment). It will be appreciated that the compounds of the invention include their tautomeric forms (i.e., tautomers).
[0190] When the compounds of the present invention are basic, salts may be prepared from pharmaceutically acceptable non-toxic acids, including inorganic and organic acids. Examples of such acids include acetic, benzenesulfonic, benzoic, camphorsulfonic, citric, ethanesulfonic, fumaric, gluconic, glutamic, hydrobromic, hydrochloric, isethionic, lactic, maleic, malic, mandelic, methanesulfonic, mucic, nitric, pamoic, pantothenic, phosphoric, succinic, sulfuric, tartaric, and p-toluenesulfonic acids.
[0191] The invention is described using the following definitions unless otherwise indicated.
[0192] As used herein, the term "alkyl" refers to a saturated or unsaturated, branched, straight-chain or cyclic monovalent hydrocarbon group derived by the removal of one hydrogen atom from a single carbon atom of a parent alkane, alkene, or alkyne. Representative alkyl groups include methyl; ethyls such as ethanyl, ethenyl, ethynyl; propyls such as propan-1-yl, propan-2-yl, cyclopropan-1-yl, prop-1-en-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), cycloprop-1-en-1-yl; cycloprop-2-en-1-yl, prop-1-yn-1-yl, and prop-2-yn-1-yl; butyls such as butan-1-yl, butan-2-yl, 2-methyl-propan-1-yl, 2-methyl-propan-2-yl, cyclobutan-1-yl, but-1-en-1-yl, but-1-en-2-yl, 2-methyl-prop-1-en-1-yl, but-2-en-1-yl, but-2-en-2-yl, buta-1,3-dien-1-yl, buta-1,3-dien-2-yl, cyclobut-1-en-1-yl, cyclobut-1-en-3-yl, cyclobuta-1,3-dien-1-yl, but-1-yn-1-yl, but-1-yn-3-yl, and but-3-yn-1-yl; and the like. Where a specific level of saturation is intended, the expressions "alkanyl," "alkenyl," and "alkynyl" are used. Alkyl groups include cycloalkyl groups. The term "cycloalkyl" refers to mono-, bi-, and tricyclic alkyl groups having the indicated number of carbon atoms. Representative cycloalkyl groups include cyclopropyl, cyclopentyl, cycloheptyl, adamantyl, cyclododecylmethyl, and 2-ethyl-1-bicyclo[4.4.0]decyl groups. The alkyl group may be unsubstituted or substituted as described below.
[0193] "Alkanyl" refers to a saturated branched, straight-chain, or cyclic alkyl group. Representative alkanyl groups include methanyl; ethanyl; propanyls such as propan-1-yl, propan-2-yl(isopropyl), and cyclopropan-1-yl; butanyls such as butan-1-yl, butan-2-yl (sec-butyl), 2-methyl-propan-1-yl(isobutyl), 2-methyl-propan-2-yl(t-butyl), and cyclobutan-1-yl; and the like. The alkanyl group may be substituted or unsubstituted. Representative alkanyl group substituents include
[0194] --R.sub.14, --OR.sub.14, --SR.sub.14, --NR.sub.14(R.sub.15),
[0195] --X, --CX.sub.3, --CN, --NO.sub.2,
[0196] --C(.dbd.O)R.sub.14, --C(.dbd.O)OR.sub.14, --C(.dbd.O)NR.sub.14(R.sub.15), --C(.dbd.O)SR.sub.14,
[0197] --C(.dbd.NR.sub.14)R.sub.14, --C(.dbd.NR.sub.14)OR.sub.14, --C(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --C(.dbd.NR.sub.14)SR.sub.14,
[0198] --C(.dbd.S)R.sub.14, --C(.dbd.S)OR.sub.14, --C(.dbd.S)NR.sub.14(R.sub.15), --C(.dbd.S)SR.sub.14,
[0199] --NR.sub.14C(.dbd.O)NR.sub.14(R.sub.15), --NR.sub.14(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --NR.sub.14C(.dbd.S)NR.sub.14(R.sub.15),
[0200] --S(.dbd.O).sub.2R.sub.14, --S(.dbd.O).sub.2OR.sub.14, --S(.dbd.O).sub.2NR.sub.14(R.sub.15),
[0201] --OC(.dbd.O)R.sub.14, --OC(.dbd.O)OR.sub.14, --OC(.dbd.O)NR.sub.14(R.sub.15), --OC(.dbd.O)SR.sub.14,
[0202] --OS(.dbd.O).sub.2OR.sub.14, --OS(.dbd.O).sub.2NR.sub.14(R.sub.15), and
[0203] --OP(.dbd.O).sub.2(OR.sub.14),
[0204] wherein each X is independently a halogen; and R.sub.14 and R.sub.15 are independently hydrogen, C1-C6 alkyl, C6-C14 aryl, arylalkyl, C3-C10 heteroaryl, and heteroarylalkyl, as defined herein.
[0205] In certain embodiments, two hydrogen atoms on a single carbon atom can be replaced with .dbd.O, .dbd.NR.sub.12, or .dbd.S.
[0206] "Alkenyl" refers to an unsaturated branched, straight-chain, cyclic alkyl group, or combinations thereof having at least one carbon-carbon double bond derived by the removal of one hydrogen atom from a single carbon atom of a parent alkene. The group may be in either the cis or trans conformation about the double bond(s). Representative alkenyl groups include ethenyl; propenyls such as prop-1-en-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), prop-2-en-2-yl, and cycloprop-1-en-1-yl; cycloprop-2-en-1-yl; butenyls such as but-1-en-1-yl, but-1-en-2-yl, 2-methyl-prop-1-en-1-yl, but-2-en-1-yl, but-2-en-1-yl, but-2-en-2-yl, buta-1,3-dien-1-yl, buta-1,3-dien-2-yl, cyclobut-1-en-1-yl, cyclobut-1-en-3-yl, and cyclobuta-1,3-dien-1-yl; and the like. The alkenyl group may be substituted or unsubstituted. Representative alkenyl group substituents include
[0207] --R.sub.14,
[0208] --X, --CX.sub.3, --CN,
[0209] --C(.dbd.O)R.sub.14, --C(.dbd.O)OR.sub.14, --C(.dbd.O)NR.sub.14(R.sub.15), --C(.dbd.O)SR.sub.14,
[0210] --C(.dbd.NR.sub.14)R.sub.14, --C(.dbd.NR.sub.14)OR.sub.14, --C(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --C(.dbd.NR.sub.14)SR.sub.14,
[0211] --C(.dbd.S)R.sub.14, --C(.dbd.S)OR.sub.14, --C(.dbd.S)NR.sub.14(R.sub.15), --C(.dbd.S)SR.sub.14,
[0212] wherein each X is independently a halogen; and R.sub.14 and R.sub.15 are independently hydrogen, C1-C6 alkyl, C6-C14 aryl, arylalkyl, C3-C10 heteroaryl, and heteroarylalkyl, as defined herein.
[0213] "Alkynyl" refers to an unsaturated branched, straight-chain, or cyclic alkyl group having at least one carbon-carbon triple bond derived by the removal of one hydrogen atom from a single carbon atom of a parent alkyne. Representative alkynyl groups include ethynyl; propynyls such as prop-1-yn-1-yl and prop-2-yn-1-yl; butynyls such as but-1-yn-1-yl, but-1-yn-3-yl, and but-3-yn-1-yl; and the like. The alkynyl group may be substituted or unsubstituted. Representative alkynyl group substituents include those as described above for alkenyl groups.
[0214] The term "haloalkyl" refers to an alkyl group as defined above having the one or more hydrogen atoms replaced by a halogen atom. Representative haloalkyl groups include halomethyl groups such as chloromethyl, fluoromethyl, and trifluoromethyl groups; and haloethyl groups such as chloroethyl, fluoroethyl, and perfluoroethyl groups.
[0215] The term "heteroalkyl" refers to an alkyl group having the indicated number of carbon atoms and where one or more of the carbon atoms is replaced with a heteroatom selected from O, N, or S. Where a specific level of saturation is intended, the expressions "heteroalkanyl," "heteroalkenyl," and "heteroalkynyl" are used. Representative heteroalkyl groups include ether, amine, and thioether groups. Heteroalkyl groups include heterocyclyl groups. The term "heterocyclyl" refers to a 5- to 10-membered non-aromatic mono- or bicyclic ring containing 1-4 heteroatoms selected from O, S, and N. Representative heterocyclyl groups include pyrrolidinyl, piperidinyl, piperazinyl, tetrahydrofuranyl, tetrahydropuranyl, and morpholinyl groups. The heteroalkyl group may be substituted or unsubstituted. Representative heteroalkyl substituents include
[0216] --R.sub.14, --OR.sub.14, --SR.sub.14, --NR.sub.14(R.sub.15),
[0217] --X, --CX.sub.3, --CN, --NO.sub.2,
[0218] --C(.dbd.O)R.sub.14, --C(.dbd.O)OR.sub.14, --C(.dbd.O)NR.sub.14(R.sub.15), --C(.dbd.O)SR.sub.14,
[0219] --C(.dbd.NR.sub.14)R.sub.14, --C(.dbd.NR.sub.14)OR.sub.14, --C(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --C(.dbd.NR.sub.14)SR.sub.14,
[0220] --C(.dbd.S)R.sub.14, --C(.dbd.S)OR.sub.14, --C(.dbd.S)NR.sub.14(R.sub.15), --C(.dbd.S)SR.sub.14,
[0221] --NR.sub.14C(.dbd.O)NR.sub.14(R.sub.15), --NR.sub.14(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --NR.sub.14C(.dbd.S)NR.sub.14(R.sub.15),
[0222] --S(.dbd.O).sub.2R.sub.14, --S(.dbd.O).sub.2OR.sub.14, --S(.dbd.O).sub.2NR.sub.14(R.sub.15),
[0223] --OC(.dbd.O)R.sub.14, --OC(.dbd.O)OR.sub.14, --OC(.dbd.O)NR.sub.14(R.sub.15), --OC(.dbd.O)SR.sub.14,
[0224] --OS(.dbd.O).sub.2OR.sub.14, --OS(.dbd.O).sub.2NR.sub.14(R.sub.15), and
[0225] --OP(.dbd.O).sub.2(OR.sub.14),
[0226] wherein each X is independently a halogen; and R.sub.14 and R.sub.15 are independently hydrogen, C1-C6 alkyl, C6-C14 aryl, arylalkyl, C3-C10 heteroaryl, and heteroarylalkyl, as defined herein.
[0227] In certain embodiments, two hydrogen atoms on a single carbon atom can be replaced with .dbd.O, .dbd.NR.sub.12, or .dbd.S.
[0228] The term "alkoxy" refers to an alkyl group as described herein bonded to an oxygen atom. Representative C1-C3 alkoxy groups include methoxy, ethoxy, propoxy, and isopropoxy groups.
[0229] The term "alkylamino" refers an alkyl group as described herein bonded to a nitrogen atom. The term "alkylamino" includes monoalkyl- and dialkylaminos groups. Representative C1-C6 alkylamino groups include methylamino, dimethylamino, ethylamino, methylethylamino, diethylamino, propylamino, and isopropylamino groups.
[0230] The term "alkylthio" refers an alkyl group as described herein bonded to a sulfur atom. Representative C1-C6 alkylthio groups include methylthio, propylthio, and isopropylthio groups.
[0231] The term "aryl" refers to a monovalent aromatic hydrocarbon group derived by the removal of one hydrogen atom from a single carbon atom of a parent aromatic ring system. Suitable aryl groups include groups derived from aceanthrylene, acenaphthylene, acephenanthrylene, anthracene, azulene, benzene, chrysene, coronene, fluoranthene, fluorene, hexacene, hexaphene, hexalene, as-indacene, s-indacene, indane, indene, naphthalene, octacene, octaphene, octalene, ovalene, penta-2,4-diene, pentacene, pentalene, pentaphene, perylene, phenalene, phenanthrene, picene, pleiadene, pyrene, pyranthrene, rubicene, triphenylene, trinaphthalene, and the like. In certain embodiments, the aryl group is a C5-C14 aryl group. In other embodiments, the aryl group is a C5-C10 aryl group. The number of carbon atoms specified refers to the number of carbon atoms in the aromatic ring system. Representative aryl groups are phenyl, naphthyl, and cyclopentadienyl. The aryl group may be substituted or unsubstituted. Representative aryl group substituents include
[0232] --R.sub.14, --OR.sub.14, --SR.sub.14, --NR.sub.14(R.sub.15),
[0233] --X, --CX.sub.3, --CN, --NO.sub.2,
[0234] --C(.dbd.O)R.sub.14, --C(.dbd.O)OR.sub.14, --C(.dbd.O)NR.sub.14(R.sub.15), --C(.dbd.O)SR.sub.14,
[0235] --C(.dbd.NR.sub.14)R.sub.14, --C(.dbd.NR.sub.14)OR.sub.14, --C(.dbd.NR.sub.14)NR.sub.14(R.sub.15), --C(.dbd.NR.sub.14)SR.sub.14,
[0236] --C(.dbd.S)R.sub.14, --C(.dbd.S)OR.sub.14, --C(.dbd.S)NR.sub.14(R.sub.15), --C(.dbd.S)SR.sub.14,
[0237] --NR.sub.14C(.dbd.O)NR.sub.14(R.sub.15), --NR.sub.14(.dbd.NR.sub.15)NR.sub.14(R.sub.15), --NR.sub.14C(.dbd.S)NR.sub.14(R.sub.15),
[0238] --S(.dbd.O).sub.2R.sub.14, --S(.dbd.O).sub.2OR.sub.14, --S(.dbd.O).sub.2NR.sub.14(R.sub.15),
[0239] --OC(.dbd.O)R.sub.14, --OC(.dbd.O)OR.sub.14, --OC(.dbd.O)NR.sub.14(R.sub.15), --OC(.dbd.O)SR.sub.14,
[0240] --OS(.dbd.O).sub.2OR.sub.14, --OS(.dbd.O).sub.2NR.sub.14(R.sub.15), and
[0241] --OP(.dbd.O).sub.2(OR.sub.14),
[0242] wherein each X is independently a halogen; and R.sub.14 and R.sub.15 are independently hydrogen, C1-C6 alkyl, C6-C14 aryl, arylalkyl, C3-C10 heteroaryl, and heteroarylalkyl, as defined herein.
[0243] The term "aralkyl" refers to an alkyl group as defined herein with an aryl group, optionally substituted, as defined herein substituted for one of the alkyl group hydrogen atoms. Suitable aralkyl groups include benzyl, 2-phenylethan-1-yl, 2-phenylethen-1-yl, naphthylmethyl, 2-naphthylethan-1-yl, 2-naphthylethen-1-yl, naphthobenzyl, 2-naphthophenylethan-1-yl, and the like. Where specific alkyl moieties are intended, the terms aralkanyl, aralkenyl, and aralkynyl are used. In certain embodiments, the aralkyl group is a C6-C20 aralkyl group, (e.g., the alkanyl, alkenyl, or alkynyl moiety of the aralkyl group is a C1-C6 group and the aryl moiety is a C5-C14 group). In other embodiments, the aralkyl group is a C6-C13 aralkyl group (e.g., the alkanyl, alkenyl, or alkynyl moiety of the aralkyl group is a C1-C3 group and the aryl moiety is a C5-C10 aryl group. In certain embodiments, the aralkyl group is a benzyl group.
[0244] The term "heteroaryl" refers to a monovalent heteroaromatic group derived by the removal of one hydrogen atom from a single atom of a parent heteroaromatic ring system, which may be monocyclic or fused ring (i.e., rings that share an adjacent pair of atoms). A "heteroaromatic" group is a 5- to 14-membered aromatic mono- or bicyclic ring containing 1-4 heteroatoms selected from O, S, and N. Representative 5- or 6-membered aromatic monocyclic ring groups include pyridine, pyrimidine, pyridazine, furan, thiophene, thiazole, oxazole, and isooxazole. Representative 9- or 10-membered aromatic bicyclic ring groups include benzofuran, benzothiophene, indole, pyranopyrrole, benzopyran, quionoline, benzocyclohexyl, and naphthyridine. Suitable heteroaryl groups include groups derived from acridine, arsindole, carbazole, .beta.-carboline, chromane, chromene, cinnoline, furan, imidazole, indazole, indole, indoline, indolizine, isobenzofuran, isochromene, isoindole, isoindoline, isoquinoline, isothiazole, isoxazole, naphthyridine, oxadiazole, oxazole, perimidine, phenanthridine, phenanthroline, phenazine, phthalazine, pteridine, purine, pyran, pyrazine, pyrazole, pyridazine, pyridine, pyrimidine, pyrrole, pyrrolizine, quinazoline, quinoline, quinolizine, quinoxaline, tetrazole, thiadiazole, thiazole, thiophene, triazole, xanthene, and the like. In certain embodiments, the heteroaryl group is a 5-14 membered heteroaryl group. In other embodiments, the heteroaryl group is a 5-10 membered heteroaryl group. Preferred heteroaryl groups are those derived from thiophene, pyrrole, benzothiophene, benzofuran, indole, pyridine, quinoline, imidazole, oxazole, and pyrazine. The heteroaryl group may be substituted or unsubstituted. Representative heteroaryl group substituents include those described above for aryl groups.
[0245] The term "heteroarylalkyl" refers to an alkyl group as defined herein with a heteroaryl group, optionally substituted, as defined herein substituted for one of the alkyl group hydrogen atoms. Where specific alkyl moieties are intended, the terms heteroarylalkanyl, heteroarylalkenyl, or heteroarylalkynyl are used. In certain embodiments, the heteroarylalkyl group is a 6-20 membered heteroarylalkyl (e.g., the alkanyl, alkenyl or alkynyl moiety of the heteroarylalkyl is a C1-C6 group and the heteroaryl moiety is a 5-14-membered heteroaryl group. In other embodiments, the heteroarylalkyl group is a 6-13 membered heteroarylalkyl (e.g., the alkanyl, alkenyl or alkynyl moiety is C1-C3 group and the heteroaryl moiety is a 5-10-membered heteroaryl group).
[0246] The term "acyl" group refers to the --C(.dbd.O)--R' group, where R' is selected from optionally substituted alkyl, optionally substituted aryl, and optionally substituted heteroaryl, as defined herein.
[0247] The term "halogen" or "halo" refers to fluoro, chloro, bromo, and iodo groups.
[0248] The term "substituted" refers to a group in which one or more hydrogen atoms are each independently replaced with the same or different substituent(s).
[0249] Representative compounds of the invention and related intermediates were prepared from commercially available starting materials or starting materials prepared by conventional synthetic methodologies. Representative compounds of the invention were prepared according to Methods A to J as described below and illustrated in FIGS. 1-3. The preparations of certain intermediates (I-1 to I-12) useful in the preparation of compounds of the invention are described in the Synthetic Intermediate section below.
[0250] FIGS. 1-3 present schematic illustrations of representative synthetic pathways for the preparation of representative compounds of the invention P5-P4-P3-P2-P1. As used herein, "P5-P4-P3-P2-P1" refers to compounds of the invention prepared from five (5) components: P1, P2, P3, P4, and P5. Protected version of the components useful in the preparation of the compounds of the invention are designated as, for example, "PG-P2," "PG-P2-P1," "PG-P3," and "PG-P3-P2-P1," where "PG" is refers to a protecting group that allows for the coupling of, for example, P1 to P2 or P3 to P1-P2, and that is ultimately removed to provide, for example, P1-P2 or P1-P2-P3.
[0251] FIG. 1 is a schematic illustration of another representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from P5. In this pathway, compound P5-P4-P3-P2-P1 is prepared in a stepwise manner starting with P5 by sequential coupling steps, separated as appropriate by deprotection steps and other chemical modifications. As shown in FIG. 1, P5 is coupled with PG-P4 to provide P5-P4-PG, which is then deprotected to provide P5-P4 and ready for coupling with the next component, P3-PG. The process is continued with subsequent couplings PG-P2 with P5-P4-P3 and PG-P1 with P5-P4-P3-P2 to ultimately provide P5-P4-P3-P2-P1. Example A1 was prepared by this method.
[0252] FIG. 2 is a schematic illustration of a representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from P1. In this pathway, compound P5-P4-P3-P2-P1 is prepared in a stepwise manner starting with P1 by sequential coupling steps, separated as appropriate by deprotection steps and other chemical modifications. As shown in FIG. 2, P1 is coupled with PG-P2 to provide PG-P2-P1, which is then deprotected to provide P2-P1 and ready for coupling with the next component, PG-P3. The process is continued with subsequent couplings PG-P4 with P3-P2-P1 and PG-P5 with P4-P3-P2-P1 to ultimately provide P5-P4-P3-P2-P1.
[0253] FIG. 3 is a schematic illustration of a further representative synthetic pathway for the preparation of representative compounds of the invention P5-P4-P3-P2-P1 starting from a component other than P1 or P5. In this pathway, compound P5-P4-P3-P2-P1 is prepared in a stepwise manner starting with P2 by sequential coupling steps, separated as appropriate by deprotection steps and other chemical modifications. As shown in FIG. 3, there are multiple pathways to P5-P4-P3-P2-P1. Examples C1-C41 were prepared by this method.
[0254] The preparation of representative compounds and their characterization are described in Examples A1 and C1-C41. The structures of representative compounds are set forth in Table 1
TABLE-US-00001 TABLE 1 Representative Compounds. Cmpd # Structure A1 ##STR00023## C1 ##STR00024## C2 ##STR00025## C3 ##STR00026## C4 ##STR00027## C5 ##STR00028## C6 ##STR00029## C7 ##STR00030## C8 ##STR00031## C9 ##STR00032## C10 ##STR00033## C11 ##STR00034## C12 ##STR00035## C13 ##STR00036## C14 ##STR00037## C15 ##STR00038## C16 ##STR00039## C17 ##STR00040## C18 ##STR00041## C19 ##STR00042## C20 ##STR00043## C21 ##STR00044## C22 ##STR00045## C23 ##STR00046## C24 ##STR00047## C25 ##STR00048## C26 ##STR00049## C27 ##STR00050## C28 ##STR00051## C29 ##STR00052## C30 ##STR00053## C31 ##STR00054## C32 ##STR00055## C33 ##STR00056## C34 ##STR00057## C35 ##STR00058## C36 ##STR00059## C37 ##STR00060## C38 ##STR00061## C39 ##STR00062## C40 ##STR00063## C41 ##STR00064##
[0255] A general kinetic enzyme assay useful for determining the inhibitory activity of the compounds of the invention is described in Examples D1 and D4.
[0256] A Granzyme B enzymatic inhibition assay is described in Example D2 and Example D5. The compounds of the invention identified in Table 1 exhibited Granzyme B inhibitory activity. In certain embodiments, select compounds exhibited IC.sub.50<50,000 nM. In other embodiments, select compounds exhibited IC.sub.50<10,000 nM. In further embodiments, select compounds exhibited IC.sub.50<1,000 nM. In still further embodiments, select compounds exhibited IC.sub.50<100 nM. In certain embodiments, select compounds exhibited IC.sub.50 from 10 nM to 100 nM, preferably from 1 nM to 10 nM, more preferably from 0.1 nM to 1 nM, and even more preferably from 0.01 nM to 0.1 nM.
[0257] A caspase enzymatic inhibition assay is described in Example D3 and Example D6. None of the compounds of the invention tested demonstrated an ability to significantly inhibit any of the caspases evaluated at a concentration of 50 .mu.M. In certain embodiments, the compounds exhibited less than 50% inhibition at 50 .mu.M. In other embodiments, the compounds exhibited greater than 50% inhibition at 50 .mu.M, but less than 10% inhibition at 25 .mu.M. The results demonstrate that select compounds of the invention selectively inhibit Granzyme B without significantly inhibiting caspases.
[0258] A cell detachment assay is described in Example D7.
[0259] A fibronectin cleavage assay is described in Example D8.
[0260] A cell adhesion based on fibronectin cleavage assay is described in Example D9.
[0261] Pharmaceutical Compositions
[0262] The pharmaceutical compositions of the present invention include an inhibitor compound of the invention (e.g., a compound of Formulae (I), (II), or (III)) as an active ingredient or a pharmaceutically acceptable salt thereof in combination with a pharmaceutically acceptable carrier, and optionally other therapeutic ingredients.
[0263] The term "pharmaceutically acceptable salts" refers to salts prepared from pharmaceutically acceptable bases including inorganic bases and organic bases. Representative salts derived from inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic, manganous, ammonium, potassium, sodium, and zinc salts. Representative salts derived from pharmaceutically acceptable organic bases include salts of primary, secondary and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, and basic ion exchange resins, such as arginine, betaine, caffeine, choline, N,N'-dibenzylethylenediamine, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N-ethyl-morpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine, tripropylamine, and trimethamine.
[0264] Compositions can include one or more carriers acceptable for the mode of administration of the preparation, be it by topical administration, lavage, epidermal administration, sub-epidermal administration, dermal administration, subdermal administration, transdermal administration, subcutaneous administration, systemic administration, injection, inhalation, oral, or any other mode suitable for the selected treatment. Topical administration includes administration to external body surfaces (e.g., skin) as well as to internal body surfaces (e.g., mucus membranes for vaginal or rectal applications by, for example, suppositories). Suitable carriers are those known in the art for use in such modes of administration.
[0265] Suitable compositions can be formulated by means known in the art and their mode of administration and dose determined by a person of skill in the art. For parenteral administration, the compound can be dissolved in sterile water or saline or a pharmaceutically acceptable vehicle used for administration of non-water soluble compounds. For enteral administration, the compound can be administered in a tablet, capsule, or dissolved or suspended in liquid form. The tablet or capsule can be enteric coated, or in a formulation for sustained release. Many suitable formulations are known including, polymeric or protein microparticles encapsulating a compound to be released, ointments, pastes, gels, hydrogels, foams, creams, powders, lotions, oils, semi-solids, soaps, medicated soaps, shampoos, medicated shampoos, sprays, films, or solutions which can be used topically or locally to administer a compound. A sustained release patch or implant may be employed to provide release over a prolonged period of time. Many techniques known to one of skill in the art are described in Remington: the Science & Practice of Pharmacy by Alfonso Gennaro, 20th ed., Williams & Wilkins, (2000). Formulations can contain excipients, polyalkylene glycols such as polyethylene glycol, oils of vegetable origin, or hydrogenated naphthalenes. Biocompatible, biodegradable lactide polymer, lactide/glycolide copolymer, or polyoxyethylene-polyoxypropylene copolymers can be used to control the release of a compound. Other potentially useful delivery systems for a modulatory compound include ethylene-vinyl acetate copolymer particles, osmotic pumps, implantable infusion systems, and liposomes. Formulations can contain an excipient, for example, lactose, or may be aqueous solutions containing, for example, polyoxyethylene-9-lauryl ether, glycocholate, and deoxycholate, or can be an oily solution for administration in the form of drops, as a gel, or for other semi-solid formulation.
[0266] Compounds or pharmaceutical compositions in accordance with this invention or for use in the methods disclosed herein can be administered in combination with one or more other therapeutic agents as appropriate. Compounds or pharmaceutical compositions in accordance with this invention or for use in the methods disclosed herein can be administered by means of a medical device or appliance such as an implant, graft, prosthesis, stents, and wound dressings. Also, implants can be devised that are intended to contain and release such compounds or compositions. An example would be an implant made of a polymeric material adapted to release the compound over a period of time.
[0267] One skilled in the art will appreciate that suitable methods of administering a Granzyme B inhibitor directly to the eye are available (i.e., invasive and noninvasive methods). Although more than one route can be used to administer the Granzyme B inhibitor, a particular route can provide a more immediate and more effective reaction than another route. The present use is not dependent on the mode of administering the agent to an animal, preferably a human, to achieve the desired effect, and the described routes of administration are exemplary. As such, any route of administration is appropriate so long as the agent contacts an ocular cell. Thus, the Granzyme B inhibitor can be appropriately formulated and administered in the form of an injection, eye lotion, ointment, and implant.
[0268] The Granzyme B inhibitor can be applied, for example, systemically, topically, intracamerally, subconjunctivally, intraocularly, retrobulbarly, periocularly (e.g., subtenon delivery), subretinally, or suprachoroidally. In certain cases, it can be appropriate to administer multiple applications and employ multiple routes to ensure sufficient exposure of ocular cells to the Granzyme B inhibitor (e.g., subretinal and intravitreous). Multiple applications of the Granzyme B inhibitor can also be required to achieve the desired effect.
[0269] Depending on the particular case, it may be desirable to non-invasively administer the Granzyme B inhibitor to a patient. For instance, if multiple surgeries have been performed, the patient displays low tolerance to anesthetic, or if other ocular-related disorders exist, topical administration of the Granzyme B inhibitor may be most appropriate. Topical formulations are well known to those of skill in the art. Such formulations are suitable in the context of the use described herein for application to the skin or to the surface of the eye. The use of patches, corneal shields (see, U.S. Pat. No. 5,185,152), and ophthalmic solutions (see, e.g., U.S. Pat. No. 5,710,182) and ointments is within the skill in the art.
[0270] The Granzyme B inhibitor also can be present in or on a device that allows controlled or sustained release, such as an ocular sponge, meshwork, mechanical reservoir, or mechanical implant. Implants (see U.S. Pat. Nos. 5,443,505, 4,853,224 and 4,997,652), devices (see U.S. Pat. Nos. 5,554,187, 4,863,457, 5,098,443 and 5,725,493), such as an implantable device (e.g., a mechanical reservoir, an intraocular device or an extraocular device with an intraocular conduit, or an implant or a device comprised of a polymeric composition are particularly useful for ocular administration of the expression vector). The Granzyme B inhibitor also can be administered in the form of sustained-release formulations (see U.S. Pat. No. 5,378,475) comprising, for example, gelatin, chondroitin sulfate, a polyphosphoester, such as bis-2-hydroxyethyl-terephthalate, or a polylactic-glycolic acid.
[0271] When used for treating an ocular disease the Granzyme B inhibitor is administered via an ophthalmologic instrument for delivery to a specific region of an eye. Use of a specialized ophthalmologic instrument ensures precise administration while minimizing damage to adjacent ocular tissue. Delivery of the Granzyme B inhibitor to a specific region of the eye also limits exposure of unaffected cells to the Granzyme B inhibitor. A preferred ophthalmologic instrument is a combination of forceps and subretinal needle or sharp bent cannula.
[0272] Alternatively, the Granzyme B inhibitor can be administered using invasive procedures, such as, for instance, intravitreal injection or subretinal injection, optionally preceded by a vitrectomy, or periocular (e.g., subtenon) delivery. The pharmaceutical composition of the invention can be injected into different compartments of the eye (e.g., the vitreal cavity or anterior chamber).
[0273] While intraocular injection is preferred, injectable compositions can also be administered intramuscularly, intravenously, intraarterially, and intraperitoneally. Pharmaceutically acceptable carriers for injectable compositions are well-known to those of ordinary skill in the art (see Pharmaceutics and Pharmacy Practice, J. B. Lippincott Co., Philadelphia, Pa., Banker and Chalmers, eds., pages 238-250 (1982), and ASHP Handbook on Injectable Drugs, Toissel, 4th ed., pages 622-630 (1986)).
[0274] An "effective amount" of a Granzyme B inhibitor or a pharmaceutical composition of the invention as described herein includes a therapeutically effective amount or a prophylactically effective amount. A "therapeutically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic result, such as reduced levels of Granzyme B activity. A therapeutically effective amount of a compound may vary according to factors such as the disease state, age, sex, and weight of the subject, and the ability of the compound to elicit a desired response in the subject. Dosage regimens can be adjusted to provide the optimum therapeutic response. A therapeutically effective amount is also one in which any toxic or detrimental effects of the compound are outweighed by the therapeutically beneficial effects. A "prophylactically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired prophylactic result, such as Granzyme B activity. Typically, a prophylactic dose is used in subjects prior to or at an earlier stage of disease, so that a prophylactically effective amount may be less than a therapeutically effective amount.
[0275] It is to be noted that dosage values can vary with the severity of the condition to be alleviated. For any particular subject, specific dosage regimens can be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the compositions. Dosage ranges set forth herein are exemplary only and do not limit the dosage ranges that can be selected by a medical practitioner. The amount of active compound(s) in the composition can vary according to factors such as the disease state, age, sex, and weight of the subject. Dosage regimens can be adjusted to provide the optimum therapeutic response. For example, a single bolus can be administered, several divided doses can be administered over time or the dose can be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. It may be advantageous to formulate parenteral compositions in dosage unit form for ease of administration and uniformity of dosage.
[0276] In general, compounds of the invention should be used without causing substantial toxicity. Toxicity of the compounds of the invention can be determined using standard techniques, for example, by testing in cell cultures or experimental animals and determining the therapeutic index (i.e., the ratio between the LD.sub.50, the dose lethal to 50% of the population, and the LD.sub.100, the dose lethal to 100% of the population). In some circumstances however, such as in severe disease conditions, it may be necessary to administer substantial excesses of the composition.
[0277] Methods of Use
[0278] In a further aspect, the invention provides methods of using the compounds of the invention as Granzyme B inhibitors.
[0279] In one embodiment, the invention provides a method for inhibiting Granzyme B in a subject. In the method, an effective amount of a compound of the invention (e.g., a compound of Formulae (I), (II), or (III) is administered to a subject in need thereof.
[0280] In another embodiment, the invention provides a method for treating a disease, disorder, or condition treatable by inhibiting Granzyme B. In the method, a therapeutically effective amount of a compound of the invention (e.g., a compound of Formulae (I), (II), or (III)) is administered to a subject in need thereof.
[0281] As used herein, the term "disease, disorder, or condition treatable by inhibiting Granzyme B" refers to a disease, disorder, or condition in which Granzyme B is involved in the pathway related to for the disease, disorder, or condition, and that inhibiting Granzyme B results in the treatment or prevention of the disease, disorder, or condition.
[0282] Representative methods of treatment using the compounds of the invention include those described for Granzyme B inhibitors in WO 2007/101354 (Methods of Treating, Reducing, and Inhibiting the Appearance of Ageing in the Skin), WO 2009/043170 (Treatment of Dissection, Aneurysm, and Atherosclerosis Using Granzyme B Inhibitors), WO 2012/076985 (Granzyme B Inhibitor Compositions, Methods and Uses for Promoting Wound Healing), each expressly incorporated herein by reference in its entirety. The compounds of the invention are useful for treating, reducing, and inhibiting the appearance of aging of the skin; treating dissection, aneurysm, and atherosclerosis; and promoting wound healing.
[0283] Other disease and disorders described as treatable using the Granzyme B inhibitors are disclosed in WO 2003/065987 (Granzyme B Inhibitors), expressly incorporated herein by reference in its entirety. Disease and disorders described as treatable by Granzyme B inhibitors in this reference include autoimmune or chronic inflammatory diseases, such as systemic lupus erythematosis, chronic rheumatoid arthritis, type I diabetes mellitus, inflammatory bowel disease, biliary cirrhosis, uveitis, multiple sclerosis, Crohn's disease, ulcerative colitis, bullous pemphigoid, sarcoidosis, psoriasis, autoimmune myositis, Wegener's granulomatosis, ichthyosis, Graves ophthalmopathy, asthma, schleroderma and Sjogren's syndrome. The Granzyme B inhibitors described in the reference are noted as more particularly useful to treat or prevent diseases or disorders including diseases or disorders resulting from transplantation of organs or tissue, graft-versus-host diseases brought about by transplantation, autoimmune syndromes including rheumatoid arthritis, systemic lupus erythematosus, Hashimoto's thyroiditis, multiple sclerosis, myasthenia gravis, type I diabetes, uveitis, posterior uveitis, allergic encephalomyelitis, glomerulonephritis, post-infectious autoimmune diseases including rheumatic fever and post-infectious glomerulonephritis, inflammatory and hyperproliferative skin diseases, psoriasis, atopic dermatitis, contact dermatitis, eczematous dermatitis, seborrhoeic dermatitis, lichen planus, pemphigus, bullous pemphigoid, epidermolysis bullosa, urticaria, angioedemas, vasculitis, erythema, cutaneous eosinophilia, lupus erythematosus, acne, alopecia areata, keratoconjunctivitis, vernal conjunctivitis, uveitis associated with Behcet's disease, keratitis, herpetic keratitis, conical cornea, dystrophia epithelialis corneae, corneal leukoma, ocular pemphigus, Mooren's ulcer, scleritis, Graves' opthalmopathy, Vogt-Koyanagi-Harada syndrome, sarcoidosis, pollen allergies, reversible obstructive airway disease, bronchial asthma, allergic asthma, intrinsic asthma, extrinsic asthma, dust asthma, chronic or inveterate asthma, late asthma and airway hyper-responsiveness, bronchitis, gastric ulcers, vascular damage caused by ischemic diseases and thrombosis, ischemic bowel diseases, inflammatory bowel diseases, necrotizing enterocolitis, intestinal lesions associated with thermal burns, coeliac diseases, proctitis, eosinophilic gastroenteritis, mastocytosis, Crohn's disease, ulcerative colitis, migraine, rhinitis, eczema, interstitial nephritis, Goodpasture's syndrome, hemolytic-uremic syndrome, diabetic nephropathy, multiple myositis, Guillain-Barre syndrome, Meniere's disease, polyneuritis, multiple neuritis, mononeuritis, radiculopathy, hyperthyroidism, Basedow's disease, pure red cell aplasia, aplastic anemia, hypoplastic anemia, idiopathic thrombocytopenic purpura, autoimmune hemolytic anemia, agranulocytosis, pernicious anemia, megaloblastic anemia, anerythroplasia, osteoporosis, sarcoidosis, fibroid lung, idiopathic interstitial pneumonia, dermatomyositis, leukoderma vulgaris, ichthyosis vulgaris, photoallergic sensitivity, cutaneous T cell lymphoma, arteriosclerosis, atherosclerosis, aortitis syndrome, polyarteritis nodosa, myocardosis, scleroderma, Wegener's granuloma, Sjogren's syndrome, adiposis, eosinophilic fascitis, lesions of gingiva, periodontium, alveolar bone, substantia ossea dentis, glomerulonephritis, male pattern alopecia or alopecia senilis by preventing epilation or providing hair germination and/or promoting hair generation and hair growth, muscular dystrophy, pyoderma and Sezary's syndrome, Addison's disease, ischemia-reperfusion injury of organs which occurs upon preservation, transplantation or ischemic disease, endotoxin-shock, pseudomembranous colitis, colitis caused by drug or radiation, ischemic acute renal insufficiency, chronic renal insufficiency, toxinosis caused by lung-oxygen or drugs, lung cancer, pulmonary emphysema, cataracta, siderosis, retinitis pigmentosa, senile macular degeneration, vitreal scarring, corneal alkali burn, dermatitis erythema multiforme, linear IgA ballous dermatitis and cement dermatitis, gingivitis, periodontitis, sepsis, pancreatitis, diseases caused by environmental pollution, aging, carcinogenesis, metastasis of carcinoma and hypobaropathy, disease caused by histamine or leukotriene-C4 release, Behcet's disease, autoimmune hepatitis, primary biliary cirrhosis, sclerosing cholangitis, partial liver resection, acute liver necrosis, necrosis caused by toxin, viral hepatitis, shock, or anoxia, B-virus hepatitis, non-A/non-B hepatitis, cirrhosis, alcoholic cirrhosis, hepatic failure, fulminant hepatic failure, late-onset hepatic failure, "acute-on-chronic" liver failure, augmentation of chemotherapeutic effect, cytomegalovirus infection, HCMV infection, AIDS, cancer, senile dementia, trauma, and chronic bacterial infection. To the extent that the diseases and disorders noted in the reference are treatable by the Granzyme B inhibitors described in the reference, the Granzyme B inhibitors of the present invention are also useful in treating and/or ameliorating a symptom associated with these diseases and conditions.
[0284] Elevated Granzyme B levels have been identified in cells and tissues from subjects suffering from a variety of diseases and conditions including Rasmussen encephalitis, amyotrophic lateral sclerosis (ALS), chronic inflammation, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), Kawasaki disease, idiopathic pulmonary fibrosis, chronic obstructive pulmonary disease (COPD), coronary artery disease (CAD), transplant vascular disease (TVD), restenosis, acute respiratory distress syndrome (ARDS), chronic obstructive sialadentis (associated with sialolithiasis), vitiligo, allergic contact dermatitis (ACD), atopic dermatitis (AD), pityriasis rosea (PR), rheumatoid arthritis (RA), osteoarthritis (OA), vasculitic neuropathy, sensory perineuritis, ischemic stroke, spinal cord injury, myasthenia gravis (MG), lymphocytic gastritis, autoimmune cholangitis (AIC), nodular regenerative hyperplasia (NRH) of the liver, achalasia, esophagitis, eosinophilic fasciitis, cryptorchidism, necrotizing lymphadenitis, Duchenne muscular dystrophy, facioscapulo humeral muscular dystrophy, and Higashi syndrome. Other diseases and conditions in which elevated Granzyme B levels have been identified include those described in WO 2009/043167 (Granzyme A and Granzyme B Diagnostics), expressly incorporated herein by reference in its entirety. The Granzyme B inhibitors of the invention may be useful for treating, alleviating or ameliorating a symptom of, diminishing the extent of, stabilizing, or ameliorating or palliating the diseases and conditions noted above in which elevated Granzyme B levels have been identified. A description of intracellular versus extracellular Granzyme B in immunity and disease is provided in Granville et al., Laboratory Investigation, 2009, 1-26, expressly incorporated herein by reference in its entirety. The reference provides a listing of conditions in which the pathogenic role of Granzyme B has been identified.
[0285] The compounds of the invention are useful in treating cutaneous scleroderma, epidermolysis bullosa, radiation dermatitis, alopecia areata, and discoid lupus erythematosus.
[0286] Cutaneous Scleroderma.
[0287] Scleroderma refers to a heterogeneous group of autoimmune fibrosing disorders. Limited cutaneous systemic sclerosis (CREST syndrome or LcSSc) develop sclerosis of the skin distal to their elbows and knees and have facial involvement. Patients with diffuse cutaneous systemic sclerosis (DcSSc) develop proximal, in addition to distal, skin sclerosis. Both groups of patients are also at high risk of developing internal organ involvement. Patients with LcSSc and DcSSc suffer from Raynaud's phenomenon (excessively reduced blood flow in response to cold or emotional stress, causing discoloration of the fingers, toes, and occasionally other areas believed to be the result of vasospasms that decrease blood supply to the respective regions) with high frequencies. Management of progressive skin involvement is dependent on additional comorbidities. In patients with skin involvement only, mycophenolate mofetil (Cellsept, immunomodulator) or methotrexate (T cell modulator) have been recommended.
[0288] Epidermolysis Bullosa.
[0289] Epidermolysis bullosa acquisita (EBA) is a chronic mucocutaneous autoimmune skin blistering disease. EBA patients can be classified into two major clinical subtypes: noninflammatory (classical or mechanobullous) and inflammatory EBA, which is characterized by cutaneous inflammation. In patients with inflammatory EBA, widespread vesiculobullous eruptions are observed, typically involving the trunk, central body, extremities, and skin folds. Usually the patients suffer from pruritus (rashes). Autoantibodies targeting type VII collagen (COL7) has been implicated in the pathogenesis. Therefore, EBA is a prototypical autoimmune disease with a well-characterized pathogenic relevance of autoantibody binding to the target antigen. EBA is a rare disease with an incidence of 0.2-0.5 new cases per million and per year. The current treatment of EBA relies on general immunosuppressive therapy, which does not lead to remission in all cases.
[0290] Radiation Dermatitis.
[0291] Radiation Dermatitis (acute skin reaction) ranges from a mild rash to severe ulceration. Approximately 85-90% of patients treated with radiation therapy will experience a moderate-to-severe skin reaction. Acute radiation-induced skin reactions often lead to itching and pain, delays in treatment, and diminished aesthetic appearance--and subsequently to a decrease in quality of life. Skin reactions related to radiation therapy usually manifest within 1-4 weeks of radiation start, persist for the duration of radiation therapy, and may require 2-4 weeks to heal after completion of therapy. The severity of the skin reaction ranges from mild erythema (red rash) and dry desquamation (itchy, peeling skin) to more severe moist desquamation (open wound) and ulceration. Treatments that have been assessed for the management of radiation-induced skin reactions include topical steroid creams, nonsteroidal creams, dressings, and herbal remedies. Only three trials have showed a significant difference: one in favor of a corticosteroid cream, one favoring a nonsteroidal cream, and one for a dressing. However, all three of these trials were small and had limitations, thus there is still an unmet medical need.
[0292] Late effects of radiation therapy, typically months to years post exposure, occur at doses greater than a single dose of 20-25 Gy or fractionated doses of 70 Gy or higher. The major underlying histopathological findings at the chronic stage include telangiectasia, dense dermal fibrosis (round fibrosis), sebaceous and sweat gland atrophy, loss of hair follicles, and with higher doses, increased melanin deposition or depigmentation and skin ulcers.
[0293] Ramipril was very effective in reducing the late effects of skin injury, whereas its mitigating effects on the acute and sub-acute injury were modest. However, the dose required to mitigate these late effects may be pharmacologically too high to be clinically relevant. More recently, it has been shown that significant mitigation of acute skin injury using an adeno-associated virus encoding the manganese SOD gene, when injected subcutaneously shortly after irradiation. However, difficulties in delivery, application and cost limit the utility of this treatment strategy.
[0294] Alopecia Aerata.
[0295] Alopecia areata (AA) is a CD8+ T-cell dependent autoimmune disease of the hair follicle (HF) in which the collapse of HF immune privilege (IP) plays a key role. Mast cells (MCs) are crucial immunomodulatory cells implicated in the regulation of T cell-dependent immunity, IP, and hair growth. Many of these infiltrating immune cells express GzmB, suggesting it may be a key mediator in immune cell-mediated follicular attack. The peptide substance P was shown to increase the CD8+ cells expressing GzmB in the intrafollicular dermis, co-relating to a regression of follicles into the catagen stage of follicle growth cessation (Siebenhaar et al., J Invest Dermatol, 2007, 127: 1489-1497).
[0296] In mice fed a diet with excess vitamin A, AA was accelerated and GzmB expressing cells were found in excess surrounding hair follicles, including in the isthmus (the region of the follicle containing stem cells) (Duncan et al., J Invest Dermatol 2013, 133: 334-343). As GzmB is expressed in the immune cell infiltrate within and surrounding growing follicles, it may be a key protease involved in hair loss through autoimmunity, apoptosis and ECM degradation.
[0297] No drug is currently approved by the US FDA for the treatment of alopecia areata. A number of treatments have been found to be effective using the American College of Physician's criteria, for example, topical and oral corticosteroids and the sensitizing agents diphenylcyclopropenone and dinitrochlorobenzene. However, there is no cure for alopecia areata, nor is there any universally proven therapy that induces and sustains remission.
[0298] Discoid Lupus Erythematosus.
[0299] Granzyme B is a serine protease found in cytoplasmic granules of cytotoxic lymphocytes and natural killer cells that plays an important role in inducing apoptotic changes in target cells during granule exocytosis-induced cytotoxicity. When Granzyme B is secreted into the cytoplasm of a target cell through the pore formed by perforin, it triggers cytotoxic-induced cell death (Shah et al., Cell Immunology 2011, 269:16-21).
[0300] Lupus erythematosus (LE) is a chronic, autoimmune, multisystem disease that displays many diverse symptoms in which localized cutaneous LE (CLE) is on one end of the spectrum and severe systemic LE (SLE) on the other end. CLE is a disfiguring, chronic skin disease, with a significant impact on the patients' everyday life. CLE are further divided into four main subsets: Acute CLE (ACLE), subacute CLE (SCLE) and chronic CLE (CCLE), where classic discoid LE (DLE) is the most common form. There is also a drug-induced form of the disease. The disease often has a chronic and relapsing course that can be induced or aggravated by UV light. CLE patients display well-defined skin lesions, often in sun-exposed areas. Discoid LE is the most common subtype of CLE, 60-80% is localized above the neck and 20-40% is generalized (lesions both above and below the neck). 70-90% of the DLE patients suffer from photosensitivity and sun exposed areas such as the scalp, ears and cheeks, which are most commonly involved areas. The lesions start as erythematosus maculae or papules with a scaly surface and then grow peripherally into larger discoid plaques that heal with atrophic scar and pigmentary changes. DLE often results in scarring and alopecia. Mutilation with tissue loss can be seen when the lesions affect the ears and tip of the nose. CLE can be managed but so far, not cured. Avoidance of trigger factors is of utmost importance, such as, cessation of smoking and avoidance of sun exposure. The treatment is about the same for the different CLE subsets where first-line of treatment is sun-protection and local therapy with corticosteroids or calcineurin inhibitors. Antimalarial are the first choice of systemic treatment.
[0301] Strong co-expression of Granzyme B and the skin-homing molecule, cutaneous lymphocyte antigen (CLA) was found in lesional lymphocytes of patients with scarring localized chronic DLE and disseminated chronic DLE, which was enhanced compared with nonscarring subacute CLE and healthy controls (Wenzel et al., British Journal of Dermatology 2005, 153: 1011-1015). Wenzel et al. conclude that skin-homing cytotoxic Granzyme B-positive lymphocytes play an important role in the pathophysiology of scarring chronic DLE and that the potentially autoreactive cytotoxic lymphocytes targeting adnexal structures may lead to scarring lesions in chronic DLE.
[0302] Correlation between Granzyme B-positive lymphocytes and the presence of CLE was shown by Grassi (Grassi et al., Clinical and Experimental Dermatology 2009, 34:910-914). Granzyme B is an apoptosis immunological mediator that, once synthesized and free from activated cytotoxic lymphocytes, enters the target cell and starts apoptotic mechanisms involved at different levels in all apoptotic pathways. In CLE, apoptosis is characterized by the presence of colloid or Civatte bodies, which are evident in the epidermis and papillary dermis of CLE lesions, and since Granzyme B is mainly expressed in CLE lesions, Grassi et al. conclude that Granzyme B could play a role in the induction of apoptotic mechanisms in CLE.
[0303] The expression of Granzyme B and perforin was correlated with clinicopathological features in patients with DLE, where both Granzyme B and perforin were expressed in DLE, with absent expression in normal skin (Abdou et al., Ultrastructural Pathology 2013, Early Online 1-9). Abdou et al. concluded that cytotoxicity in dermal lymphocytic inflammation was due to expression of both Granzyme B and perforin.
[0304] Extracellular Granzymes B is also reported to play a role in DLE by Grassi et al. Further, UV light increases Granzyme B expression in keratinocytes as well as mast cells (Hernandez-Pigeon, J. Biol. Chem., 2007, 282:8157-8164). As Granzymes B is in abundance at the dermal-epidermal junction (DEJ), where many key extracellular matrix substrates are present (for example, laminin, fibronectin, decorin), it follows that Granzymes B may also be damaging the DEJ, as is observed in DLE. Given its expression in adnexal structures, Granzyme B may also be contributing to alopecia, as reduced Granzymes B is associated with reduced hair loss in a murine model of skin aging. Similarly, reduced extracellular Granzyme B activity is associated with improved collagen organization and reduced scarring in the skin and aorta.
[0305] In view of the established connection between Granzyme B and DLE, by virtue of their ability to inhibit Granzyme B, the compounds of the invention are useful in methods for treating lupus erythematosus (LE) including severe systemic LE (SLE) and localized cutaneous LE (CLE) (e.g., acute CLE (ACLE), subacute CLE (SCLE), chronic CLE (CCLE) and the most common form classic discoid LE (DLE)). In one embodiment, the invention provides a method for treating DLE comprising administering a therapeutically effective amount of a compound of the invention to a subject suffering from DLE.
[0306] Administration.
[0307] In the above methods, the administration of the Granzyme B inhibitor can be a systemic administration, a local administration (e.g., administration to the site, an inflamed microenvironment, an inflamed joint, an area of skin, a site of a myocardial infarct, an eye, a neovascularized tumor), or a topical administration to a site (e.g., a site of inflammation or a wound).
[0308] The term "subject" or "patient" is intended to include mammalian organisms. Examples of subjects or patients include humans and non-human mammals, e.g., nonhuman primates, dogs, cows, horses, pigs, sheep, goats, cats, mice, rabbits, rats, and transgenic non-human animals. In specific embodiments of the invention, the subject is a human.
[0309] The term "administering" includes any method of delivery of a Granzyme B inhibitor or a pharmaceutical composition comprising a Granzyme B inhibitor into a subject's system or to a particular region in or on a subject. In certain embodiments, a moiety is administered topically, intravenously, intramuscularly, subcutaneously, intradermally, intranasally, orally, transcutaneously, intrathecal, intravitreally, intracerebral, or mucosally.
[0310] As used herein, the term "applying" refers to administration of a Granzyme B inhibitor that includes spreading, covering (at least in part), or laying on of the inhibitor. For example, a Granzyme B inhibitor may be applied to an area of inflammation on a subject or applied to, for example the eye or an area of inflammation by spreading or covering the surface of the eye with an inhibitor, by injection, oral or nasal administration.
[0311] As used herein, the term "contacting" includes contacting a cell or a subject with a Granzyme B inhibitor. Contacting also includes incubating the Granzyme B inhibitor and the cell together in vitro (e.g., adding the inhibitor to cells in culture) as well as administering the inhibitor to a subject such that the inhibitor and cells or tissues of the subject are contacted in vivo.
[0312] As used herein, the terms "treating" or "treatment" refer to a beneficial or desired result including, but not limited to, alleviation or amelioration of one or more symptoms, diminishing the extent of a disorder, stabilized (i.e., not worsening) state of a disorder, amelioration or palliation of the disorder, whether detectable or undetectable. "Treatment" can also mean prolonging survival as compared to expected survival in the absence of treatment.
[0313] Cosmetic Compositions and Related Methods
[0314] In further aspects, the invention provides cosmetic compositions that include one or more granzyme B inhibitors of the invention and methods for using the compositions to treat, reduce, and/or inhibit the appearance of ageing of the skin.
[0315] This aspect of the invention is based, in part, on the observation that granzyme B expression is induced in keratinocytes and immune cells, such as mast cells in the skin during aging. When released by these cells, granzyme B cleaves extracellular matrix proteins such as decorin which can result in collagen disorganization. This invention is also based in part on the observation that granzyme B cleaves decorin, in addition to other extracellular matrix proteins, in the interstitial space surrounding cells.
[0316] Skin is comprised of three main layers: the epidermis, the dermis and subcutaneous layers. Each of these three layers has individual compositions. The functions and structures of these layers are known to a person of skill in the art. The epidermis is the outermost layer of skin and includes both living and dead cell layers. The dermis is the middle layer of skin and is comprised of arrangements of collagen fibers, which surround many specialized cells and structures. Hair follicles are found within the dermis, and produce the hair shaft which grows out through layers of the dermis and epidermis to become visible as hair. The lowermost layer of the skin is the subcutaneous layer, often called the sub-dermis. The subcutaneous layer is comprised largely of fat and connective tissue and houses larger blood vessels and nerves. Collagen may be found in all layers of the skin, but is most prominently in the dermis layer.
[0317] A youthful appearance is achieved by not having at least one of the characteristic signs of age. This is often achieved by being young. Nevertheless, there are circumstances in which being young does not confer a youthful appearance as a disease or disorder or other non-time related event has conferred the characteristics associated with age. A youthful appearance is often characterized by the condition of the skin and the following skin qualities are typically associated with, but not limited to, a youthful appearance: small pore size, healthy skin tone, radiance, clarity, tautness, firmness, plumpness, suppleness, elasticity, softness, healthy skin texture, healthy skin contours, such as few or no wrinkles, shallow wrinkle depth, few or no fine lines, healthy skin luster and brightness, moisturized skin, healthy skin thickness and resilient skin. If a skin of a subject comprises any one or more of these characteristics then a youthful appearance is achieved.
[0318] The appearance of ageing can occur for a variety of reasons, but typically happens at a normal rate associated with the passage of time. A rate of appearance of ageing will be different for different subjects, depending on a variety of factors including age, gender, diet and lifestyle. An appearance of ageing is often characterized by the condition of the skin. Characteristics associated with an appearance of ageing in the skin include, but are not limited to, skin fragility, skin atrophy, skin wrinkles, fine lines, skin discoloration, skin sagging, skin fatigue, skin stress, skin inelasticity, skin fragility, skin softening, skin flakiness, skin dryness, enlarged pore size, skin thinning, reduced rate of skin cell turnover, deep and deepening of skin wrinkles. The rate of appearance of ageing can be measured by measuring the rate at which any one or more of the above characteristics appear. An appearance of ageing may be inhibited, reduced, or treated by reducing or maintaining a state of any one or more of these skin characteristics.
[0319] In many circumstances a reduction in the appearance of ageing of skin occurs when the rate of collagen cleavage exceeds the rate of collagen formation. In many other circumstances, a youthful appearance of skin is maintained when the rate of collagen formation is equal to the rate of collagen cleavage. In many other circumstances, a reduction in a rate of appearance of ageing of skin is achieved when the rate of decorin cleavage and collagen disorganization and cleavage is slowed such that the rate of collagen fibrillogenesis exceeds the rate of collagen cleavage and the ratio of the rate of collagen fibrillogenesis to the rate of collagen cleavage is greater after application of granzyme B inhibitor compound compared to the ratio before application of the compound. In many other circumstances, an extracellular protein, other than decorin, is also cleaved by granzyme B, and the beneficial effects of inhibiting granzyme B can be enhanced beyond what is realized by inhibiting decorin cleavage alone.
[0320] In one aspect, the invention provides a cosmetic composition. The composition comprises a cosmetically acceptable carrier and one or more compounds of the invention (e.g., a compound of Formulae (I), (II), or (III), or stereoisomers, tautomers, and cosmetically acceptable salts thereof, as described herein).
[0321] As used herein, the term "cosmetically acceptable salt" refers to a salt prepared from a cosmetically acceptable base, such as an inorganic base and an organic base, or a salt prepared from a cosmetically acceptable acid. Representative salts derived from inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic, manganous, ammonium, potassium, sodium, and zinc salts. Representative salts derived from cosmetically acceptable organic bases include salts of primary, secondary and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, and basic ion exchange resins, such as arginine, betaine, caffeine, choline, N,N'-dibenzylethylenediamine, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N-ethyl-morpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine, tripropylamine, and trimethamine.
[0322] The cosmetic compositions can be formulated by means known in the art and their mode of administration and the amount of granzyme B inhibitor compound as described herein can be determined by a person of skill in the art. Compositions for use in the methods described herein can comprise one of more of a granzyme B inhibitor compound or a cosmetically acceptable salt thereof as an active ingredient, in combination with a cosmetically acceptable carrier.
[0323] The cosmetic compositions can include diluents, excipients, solubilizing agents, emulsifying agents, and salts known to be useful for cosmetic compositions. Examples of suitable agents include thickeners, buffers, preservatives, surface active agents, neutral or cationic lipids, lipid complexes, liposomes, and penetration enhancers. In certain embodiments, the cosmetic compositions further include other cosmetic ingredients known in the art.
[0324] In certain embodiments, the cosmetic composition can include one or more penetration enhancers. Numerous types of penetration enhancers are known, such as fatty acids, bile salts, chelating agents, surfactants and non-surfactants (Lee et al., Critical Reviews in Therapeutic Drug Carrier Systems 8:91-192, 1991; Muranishi, Critical Reviews in Therapeutic Drug Carrier Systems 7:1-33, 1990). Fatty acids and their derivatives which act as penetration enhancers include, for example, cabrylic acid, oleic acid, lauric acid, capric acid, caprylic acid, hexanoic acid, myristic acid, palmitic acid, valeric acid, stearic acid, linoleic acid, linolenic acid, arachidonic acid, oleic acid, elaidic acid, erucic acid, nervonic acid, dicaprate, tricaprate, recinleate, monoolein (also known as 1-monooleoyl-rac-glycerol), dilaurin, arachidonic acid, glyceryll-monocaprate, 1-dodecylazacycloheptan-2-one, acylcarnitines, acylcholines, mono- and di-glycerides and physiologically acceptable salts thereof (e.g., oleate, laurate, caprate, myristate, palmitate, stearate, linoleate) (Lee et al., Critical Reviews in Therapeutic Drug Carrier Systems page 92, 1991; Muranishi, Critical Reviews in Therapeutic Drug Carrier Systems 7:1, 1990; El-Hariri et al., J. Pharm. Pharmacol. 44:651-654, 1992).
[0325] In certain embodiments, the cosmetic composition further includes other cosmetic ingredients known in the art to be useful for cosmetic, skincare, and/or dermatological applications (e.g., anti-wrinkle active ingredients including flavone glycosides such as alpha-glycosylrutin; coenzyme Q10; vitamin E and derivatives; as well as sunblock ingredients, moisturizers, and perfumes).
[0326] The cosmetic compositions of the invention can be administered for "cosmetic" or "skincare" (e.g., dermatologic) applications, either alone or as an "additive" in combination with other suitable agents or ingredients. As used herein, "cosmetic" and "skincare" applications includes, for example, preventive and/or restorative applications in connection with dermatological changes in the skin, such as, for example, during pre-mature skin aging; dryness; roughness; formation of dryness wrinkles; itching; reduced re-fatting (e.g., after washing); visible vascular dilations (e.g., telangiectases, cuperosis); flaccidity; formation of wrinkles and lines; local hyperpigmentation; hypopigmentation; incorrect pigmentation (e.g., age spots); increased susceptibility to mechanical stress (e.g., cracking); skin-sagging (e.g., lack of firmness) and the appearance of dry or rough skin surface features.
[0327] The cosmetic compositions of the invention can be formulated for topical administration. Such compositions can be administered topically in any of a variety of forms. Such compositions are suitable in the context of the use described herein for application to the skin or to the surface of the eye. The use of patches, corneal shields (see, U.S. Pat. No. 5,185,152), and ophthalmic solutions (see, for example, U.S. Pat. No. 5,710,182) and ointments is within the skill in the art.
[0328] Compositions for topical administration include dermal patches, ointments, lotions, serums, creams, gels, hydrogels, pastes, foams, oils, semi-solids, shampoos, soaps, drops, sprays, films, liquids, and powders. Examples of such compositions include those in which a cosmetically effective amount of a compound of the invention is encapsulated in a vehicle selected from macro-capsules, micro-capsules, nano-capsules, liposomes, chylomicrons and microsponges. Another example of such a composition includes absorption of a compound of the invention on or to a material selected from powdered organic polymers, talcs, bentonites, and other mineral supports. A third example of such a composition or formulation includes a mixture of a cosmetically effective amount of a compound of the invention with other ingredients selected from extracted lipids, vegetable extracts, liposoluble active principles, hydrosoluble active principles, anhydrous gels, emulsifying polymers, tensioactive polymers, synthetic lipids, gelifying polymers, tissue extracts, marine extracts, vitamin A, vitamin C, vitamin D, vitamin E, solar filter compositions, and antioxidants. Other examples of suitable composition ingredients can be found in US2005/0249720.
[0329] In the cosmetic compositions, the compounds of the invention can be incorporated into any gelanic form, such as oil/water emulsions and water/oil emulsions, milks, lotions, gelifying and thickening tensioactive and emulsifying polymers, pomades, lotions, capillaries, shampoos, soaps, powders, sticks and pencils, sprays, and body oils.
[0330] Regardless of the compound or formulation described herein, application/administration to a subject as a colloidal dispersion system can be used as a delivery vehicle to enhance the in vivo stability of the compound and/or to target the granzyme B inhibitor compound to a particular skin layer, tissue or cell type. Colloidal dispersion systems include, but are not limited to, macromolecule complexes, nanocapsules, microspheres, beads and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, liposomes and lipid:inhibitor complexes of uncharacterized structure. An example of a colloidal dispersion system is a plurality of liposomes. Liposomes are microscopic spheres having an aqueous core surrounded by one or more outer layers made up of lipids arranged in a bilayer configuration (see, generally, Chonn et al., Current Op. Biotech. 6:698-708, 1995). Sustained-release dosage forms of the compounds described herein can also be used.
[0331] The amount of the granzyme B inhibitor compound administered or applied to a subject is not critical, except that it should be an amount sufficient to effect improvement of the condition for which the composition is administered/applied. Application can be dependent on a number of factors, including severity and responsiveness of the condition to be treated, and with the course of treatment lasting from several days to several months, or until improvement of a condition is effected or a diminution of a symptom is achieved.
[0332] A "cosmetically effective amount" of a granzyme B inhibitor compound includes a cosmetically effective amount or a prophylactically effective amount. A "cosmetically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired cosmetic result, such as improved skin elasticity, skin durability, skin firming, skin texture, decrease the appearance or decrease rate of appearance of aging, and the like. A cosmetically effective amount of a compound may vary according to factors such as the skin state, age, sex, and weight of the subject, and the ability of the compound to elicit a desired response in the subject. Dosage regimens can be adjusted to provide the optimum cosmetic response. A cosmetically effective amount is also one in which any toxic or detrimental effects of the compound are outweighed by the cosmetically beneficial effects. A "prophylactically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired prophylactic result, such as improved skin elasticity, skin durability, skin firming, skin texture, a decrease appearance or a decrease in the rate of appearance of aging, and the like. Typically, a prophylactic dose is used in subjects prior to or at an earlier stage of skin deterioration, so that a prophylactically effective amount may be less than a cosmetically effective amount.
[0333] The amount of granzyme B inhibitor administered/applied may vary with the severity of the appearance, or rate of appearance, of age of the skin. For any particular subject, specific dosage regimens may be adjusted over time according to the individual need and the judgment of the person applying or supervising the applying of the compositions. Dosage ranges set forth herein are exemplary only and do not limit the dosage ranges that may be selected. The amount of granzyme B inhibitor compound(s) in the composition or formulation can vary according to factors such as the skin state, age, sex, and weight of the subject. Dosage regimens can be adjusted to provide the optimum response. For example, a single application can be administered/applied, several divided doses can be administered/applied over time or the amount of the composition administered/applied can be proportionally reduced or increased as indicated by the exigencies of the situation. It can be advantageous to formulate the granzyme B inhibitor compounds in a composition into a dosage unit form for ease of administration and uniformity of application.
[0334] By way of example, a granzyme B inhibitor compound of the cosmetic composition can be administered/applied to achieve from about 0.01 micrograms per milliliter (.mu.g/mL) to about 10 milligrams per milliliter, from about 0.1 .mu.g/mL to about 500 .mu.g/mL, from about 0.1 .mu.g/mL to about 1500 .mu.g/mL, from about 1 .mu.g/mL to about 2000 .mu.g/mL, and from about 0.1 .mu.g/mL to about 5000 .mu.g/mL, including any range within these ranges, final concentrations at a target site.
[0335] Appropriate dosage values can depend on the characteristics of the site to which the composition is to be applied/administered and on the form of the granzyme B inhibitor compound used. Guidance as to particular dosages and methods of delivery is provided in the literature and generally available to practitioners in the art. Those skilled in the art will employ different formulations for different uses and the granzyme B inhibitor compound used. Persons of ordinary skill in the art can easily estimate repetition rates for dosing based on measured residence times and concentrations of the granzyme B inhibitor compound in, for example, a bodily fluid or a tissue. Following successful treatment, it can be desirable to have the subject undergo maintenance therapy to prevent the recurrence of the condition, wherein a selected compound is administered/applied in maintenance doses applied, for example, once or more daily, to once every few days. In certain embodiments, granzyme B inhibitor compounds are administered/applied in an amount to achieve ex vivo concentrations from about 1 micromolar to about 10 millimolar, from about 10 micromolar to about 5000 micromolar, or from about 30 micromolar to about 3000 micromolar, and from about 25 micromolar to about 3000 micromolar final concentration over a site of interest, and including, about 25 micromolar, or about 1600 micromolar, or about 3000 micromolar final concentration over the site, and still more typically between about 1 micromolar to about 1000 micromolar.
[0336] Compounds or compositions of granzyme B inhibitors can be administered/applied by means of a device or appliance such as an implant, graft, prosthesis, garment of clothing, stent, and the like. Also, implants can be devised which are intended to contain and release such compounds or compositions. An example would be an implant made of a polymeric material adapted to release the compound over a period of time. Such implants can be placed into a garment to be worn by a subject, for example a glove, shirt, mask or hat.
[0337] The cosmetic compositions of the invention can be used to inhibit or reduce the appearance of ageing. Ageing is a natural phenomenon that cannot be reversed per se, but the appearance of ageing, such as skin deterioration including, but not limited to, skin inelasticity, skin fragility, skin softening, skin flakiness, skin dryness, enlarged pore size, skin thinning, reduced rate of skin cell turnover, skin wrinkling, deepening of skin wrinkles, skin sagging, fine lines, and skin discoloration may be inhibited or reduced.
[0338] The cosmetic compositions can be used to increase or decrease a rate of increasing or a rate of decreasing occurrences of a particular skin characteristic. In other words, the composition, when applied to the skin or a portion of the skin of a subject delays the onset of an appearance of aging. For example, in a population of subjects where half of the population applies a granzyme B inhibitor to their skin and another half of the population does not apply a granzyme B inhibitor to their skin, the half which applied a granzyme B inhibitor would not appear as aged as the half which did not apply the granzyme B inhibitor after a period of time had elapsed. The half of the population which applied a granzyme B inhibitor to the skin would also have maintained a youthful appearance.
[0339] The rate at which a particular subject experiences a change in the rate of appearance of a particular skin characteristic, i.e., an increasing or decreasing rate of the appearance of a particular skin characteristic, will depend on a variety of factors, including, but not limited to age, weight, sex and lifestyle of the subject. As such, rates are not necessarily constant, but a normal rate of increase or of decrease of an appearance of a characteristic, defined as being the new occurrence of a particular characteristic over a predetermined period of time under a set of conditions that do not include the presence of a granzyme B inhibitor applied by a method or use of this invention, is increased or decreased by applying a granzyme B inhibitor in accordance with a method or use of this invention. Methods of measuring skin characteristics, rates of increasing appearance of skin characteristics and rates of decreasing appearance of skin characteristics are known to a person of skill in the art, see for example, Measuring the Skin by Agache et al., Springer (2004).
[0340] Surprisingly, granzyme B inhibitors can also be used to increase the density of hair follicles of a skin of a subject and may be used to reduce the occurrences of cutaneous xanthomas of a skin of a subject. Actively growing hair follicles contain melanocytes that transfer pigment to matrix keratinocytes, imparting color to hair. Additionally, sebum, produced in sebaceous glands, is often secreted via hair follicles. Increased density of hair follicles results in increased pigment production and increased sebum secretion resulting in improved hair appearance (e.g., hair that is less grey in color or not grey at all) as well as healthier hair and skin. Granzyme B inhibitors also cause hair follicles to appear deeper in the skin which provide stronger hair that is less susceptible to mechanical damage. Additionally, a characteristic sign of ageing is the reduction in hair follicle density. It is known in the art that age and follicular miniaturization are weak predictors of total hair count (see Chapman et al., Brit. J. Dermatol. 152:646-649, 2005). Consequently, the characteristic sign of age associated with hair follicle density is not predictive of hair density.
[0341] The cosmetic composition may be applied to a portion of the skin of a subject or to the whole of the skin of the subject. For example, the composition may be applied to the skin, only on the face, only on the scalp, on the whole head or to each part of the body.
INCORPORATION BY REFERENCE
[0342] Each reference cited is incorporated herein by reference in its entirety.
Abbreviations
[0343] As used herein, the following abbreviations have the indicated meanings.
[0344] .sup.1H NMR: proton nuclear magnetic resonance
[0345] .sup.19F NMR: fluorine-19 nuclear magnetic resonance
[0346] % Inh: Percent inhibition
[0347] Ac-IEPD-AMC: acetyl-isoleucyl-glutamyl-prolyl-aspartyl-(7-amino-4-methylcoumarin) substrate
[0348] ACN: acetonitrile
[0349] BHET: bis-2-hydroxyethyl-terephthalate
[0350] Boc: tert-butoxycarbonyl
[0351] BSA: Bovine serum albumin
[0352] CHAPS: 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate
[0353] DAPI: 4',6-diamidino-2-phenylindole
[0354] DCM: dichloromethane
[0355] DIPEA: diisopropylethylamine
[0356] DMAP: 4-dimethylaminopyridine
[0357] DMF: dimethylformamide
[0358] DMSO: dimethylsulfoxide
[0359] DMSO-d6: dimethylsulfoxide-d6
[0360] DTT: dithiothreitol
[0361] EDC: 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
[0362] EDTA: 2-({2-[Bis(carboxymethyl)amino]ethyl}(carboxymethyl)amino)acetic acid
[0363] ESI: Electrospray ionization
[0364] EtOAc: ethyl acetate
[0365] eq.: equivalent(s)
[0366] GzmB: Granzyme B
[0367] HATU: 2-(7-aza-1H-benzotriazole-1-yl)-1,1,1,1-tetramethyluronium hexafluorophosphate
[0368] HCl: hydrochloric acid
[0369] HEPES: 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid
[0370] hGzmB: human Granzyme B
[0371] HPLC: high performance liquid chromatography
[0372] HOBt: 1-hydroxy-benzotriazol
[0373] IC.sub.50: inhibitory concentration that provides 50% inhibition
[0374] LC/MS: liquid chromatography/mass spectrometry
[0375] MeOH: methanol
[0376] mGzmB: murine Granzyme B
[0377] MS: mass spectrometry
[0378] m/z: mass to charge ratio.
[0379] Oxyma: ethyl 2-cyano-2-(hydroxyimino)acetate
[0380] PBS: phosphate buffered saline (pH 7.4)
[0381] RPM: revolution per minute
[0382] RT: room temperature
[0383] tert-BuOH: tert-butyl alcohol
[0384] THF: tetrahydrofuran
[0385] TFA: trifluoroacetic acid
[0386] wt %: weight percent
General Methods A-J
[0387] Representative compounds of the invention were prepared according to Methods A to J as described below and illustrated in FIGS. 1-3.
[0388] It will be appreciated that in the following general methods and preparation of synthetic intermediates, reagent levels and relative amounts or reagents/intermediates can be changed to suit particular compounds to be synthesized, up or down by up to 50% without significant change in expected results.
[0389] Method A: General Method for Deprotection Followed by Coupling Reaction Using EDC/HOBt/DIPEA.
##STR00065##
[0390] HCl Solution in dioxane (4M, 5 ml) was added to respective carbamate compound (0.125 mmol) and stirred for 2 hrs at RT. The reaction mixture was concentrated to dryness under vacuum and swapped with MeOH (5 ml) three times. The resulting residue was dried well under vacuum and subjected to next reaction as it was. The residue obtained above, respective acid moiety (0.125 mmol), EDC (0.19 mmol), HOBt (0.16 mmol) and DIPEA (0.5 mmol) were stirred in anhydrous DCM (5 ml) for 16 hrs. The reaction mixture was concentrated under vacuum to give the crude product which was purified on a C18 column using 10-50% MeOH in water to yield product as an off-white solid (35-55%).
[0391] Method B: General Method for Deprotection Followed by Reaction with Anhydride.
##STR00066##
[0392] HCl Solution in dioxane (4M, 5 ml) was added to a representative Boc-protected compound (0.125 mmol) and stirred for 2 hrs at RT. The reaction mixture was concentrated to dryness under vacuum and washed with MeOH (5 ml) three times. The resulting residue was dried well under vacuum and subjected to next reaction as it was. The residue obtained above, the respective anhydride moiety (0.125 mmol), and triethylamine (0.5 mmol) were added to anhydrous DCM (5 mL) and stirred for 16 hrs. The mixture was concentrated under vacuum to give the crude product which was purified on a C18 column using 10-50% MeOH in water to yield product as an off-white solid (40-60%).
[0393] Method C: General Method of Coupling Reaction Using HATU/DIPEA.
##STR00067##
[0394] The respective acid moiety (0.125 mmol), HATU (0.17 mmol), DIPEA (0.5 mmol) and respective amine moiety (0.125 mmol) were stirred in anhydrous DCM (5 ml) for 16 hrs. The reaction mixture was concentrated under vacuum to give the crude product which was purified on a C18 column using 10-50% MeOH in water (or similar ratio as needed) to yield product as an off-white solid (35-55%).
[0395] Method D: General Method of Hydrolysis Using LiOH.
##STR00068##
[0396] To the stirring solution of the ester compound (0.08 mmol) in ethanol (1 ml) was added solution of lithium hydroxide monohydrate (0.4 mmol) in water (0.5 ml). After stirring the reaction mixture for 5 hrs at RT, the mixture was acidified using citric acid (saturated solution) and concentrated under vacuum to give the crude product which was purified on a C18 column using 10-40% MeOH in water to yield product as an off-white solid (50-65%).
[0397] Method E: General Method for Boc Deprotection.
##STR00069##
[0398] HCl Solution in dioxane (4M, 0.5 ml) was added to the respective carbamate compound (0.06 mmol) and stirred for 3 hrs at RT. The reaction mixture was concentrated under vacuum to give the crude product which was purified on a C18 column using 10-40% MeOH in water to yield product as an off-white solid (50-60%).
[0399] Method F: General Method for Deprotection Followed by Reaction with Anhydride).
##STR00070##
[0400] This method is an improved procedure for the method B. HCl Solution in dioxane (4M, 5 ml) was added to a representative Boc-protected compound (0.125 mmol) and stirred for 2 hrs at RT. The reaction mixture was concentrated to dryness under vacuum and swapped with MeOH (5 ml) three times. The resulting residue was dried well under vacuum and subjected to next reaction as it was. The residue obtained above, the respective anhydride moiety (0.19 mmol, 1.5 eq.), and triethylamine (0.5 mmol, 4 eq.) were added to anhydrous DCM (5 mL) and stirred for 16 hrs. The mixture was acidified with formic acid and then concentrated under vacuum to give the crude product which was purified on a C18 column using 25-65% MeOH in water to yield product as an off-white solid (30-80%).
[0401] Method G: General Method for Boc Protection.
##STR00071##
[0402] To respective amine compound (6.1 mmol) in dioxane (6 ml) and NaOH solution (0.5M, 12 ml) was added slowly solution of di-tert-butyl dicarbonate (6.7 mmol) in dioxane (6 ml) at 0.degree. C. The reaction mixture was warmed to RT and stirred overnight. The reaction mixture was then washed with hexanes (10 ml). The separated water layer was acidified using saturated solution of citric acid and extracted with ethyl acetate (3.times.15 ml). The organic layer was washed with brine, separated, dried over sodium sulfate and concentrated to give Boc protected amine compound as off-white solid (65-90%).
[0403] Method H: General Method for EDC/HOBt/DIPEA Coupling of an Intermediate Existing as an HCl Salt and a Free Carboxylic Acid.
##STR00072##
[0404] To an intermediate collected as an HCl salt (0.125 mmol) were added the carboxylic acid (0.125 mmol), EDC (0.19 mmol), HOBt (0.16 mmol), and anhydrous DCM (5 ml). The flask was purged with N.sub.2, sonicated for 20 s and DIPEA (0.5 mmol) was added. The reaction was stirred at room temperature for 6 hrs then concentrated under reduced pressure. The residue was purified on a C18 column using 10-80% MeOH in water to yield the product as an off-white solid (40-90%).
[0405] Method I: General Method for Coupling (2H-Tetrazol-5-Yl)Methylamine and a Free Carboxylic Acid.
##STR00073##
[0406] To the carboxylic acid (0.18 mmol), were added the (2H-tetrazol-5-yl)methylamine (0.22 mmol), EDC (0.275 mmol), HOBt (0.22 mmol), and anhydrous DMF (15 ml). The flask was purged with N.sub.2, sonicated for 20 s and DIPEA (0.73 mmol) was added. The reaction was stirred at room temperature for 16 hrs. Analysis of the reaction by LC/MS showed approximately 75% conversion of the acid. An additional one half of the portion of the amine, EDC, HOBt, and DIPEA were added and the reaction was heated at 45.degree. C. for another 6 hrs then concentrated under reduced pressure. The residue was purified on a C18 column using 10-70% MeOH in water to yield the product as an off-white solid (40-95%).
[0407] Method J: General Method for Hydrogenate Deprotection of Benzyl Esters or Benzyl Carbamates.
##STR00074##
[0408] To a flask containing the respective benzylated compound (1.0 eq.) under a nitrogen atmosphere was added palladium on carbon (10 wt %, wetted, 0.2 eq.) then MeOH (0.05 M). The atmosphere was changed to hydrogen (vacuum+H.sub.2 backfill.times.3) and the suspension of black solids was stirred for 3 hrs, then filtered over a pad of CELITE.TM. and washed with excess MeOH. The reaction mixture was concentrated under vacuum to give the crude product which was purified on a C18 column using 10-50% MeOH in water to yield the product (50-95%).
[0409] The following examples are provided for the purpose of illustrating, not limiting, the invention.
EXAMPLES
Synthetic Intermediates
[0410] The following is a description of synthetic intermediates (I-1 to I-12) useful for making representative compounds of the invention.
[0411] Intermediate I-1
##STR00075##
2-Bromo-3-(bromomethyl)pyridine (I-1)
[0412] This intermediate was generated by a modified procedure based on that disclosed in Rebek, J., et al., J. Am. Chem. Soc., 107, 7487 (1985)). A three-neck round bottom flask with a stir bar was flame dried, cooled under vacuum and purged with N.sub.2. To the flask were added 2-bromo-3-methylpyridine (5.2 mL, 29.1 mmol), N-bromosuccinimide (5.5 g, 32.0 mmol), and degassed benzene (126 mL). The flask was fitted with a condenser, heated to 40.degree. C. and AIBN (0.24 g, 1.5 mmol) was added in several portions. The reaction was irradiated using a sun lamp as it was stirred at 40.degree. C. The reaction was monitored using TLC and HPLC and was stopped after 80% conversion of the pyridine reagent (approximately 8 hrs). The reaction was concentrated under reduced pressure, then redissolved in 4:1 DCM/EtOAc (120 mL) and extracted once with 50 mL of a saturated solution of NaHCO.sub.3(aq), water and a saturated solution of NaCl.sub.(aq). The organic phase was dried over anhydrous sodium sulphate, filtered and concentrated. Upon standing the residue could not be fully redissolved in DCM and the resultant suspension was filtered to remove the insoluble solid. The filtrate was concentrated to near dryness and the residue was purified by normal phase flash chromatography (EtOAc/Hexanes) to give the title compound I-1 (3.0 g, 11.9 mmol, 41%) as a yellow solid. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.33 (1H, dd, J=5.2 Hz), 7.78 (1H, dd, J=7.2 Hz), 7.28 (1H, dd, J=5.4 Hz), 4.57 (2H, s), MS (LC/MS) m/z observed 249.97, expected 249.89 [M+H]
[0413] Intermediate I-2
##STR00076##
tert-butyl 2-((diphenylmethylene)amino)acetate (I-2)
[0414] This intermediate was generated by a generic procedure based on that disclosed in US2010/0189644 and O'Donnell, Acc. Chem. Res., 37, 506 (2004). A round bottom flask was charged with a stir-bar, diphenylmethanimine (8.6 g, 47.5 mmol), tert-butyl 2-bromoacetate (9.3 g, 47.5 mmol), and acetonitrile (40 mL). The reaction was heated to 70.degree. C. and DIPEA (8.3 mL, 47.5 mmol) was added slowly. The flask was fitted with a reflux condenser and heated at 70.degree. C. for 16 hrs. Analysis of the reaction by HPLC and TLC showed complete conversion of the reactants and the reaction was cooled to room temperature. A solution of 5:3 water/formic acid (1 mL) was added the reaction was concentrated under reduced pressure. The resultant solid was filtered and washed 2.times.60 mL of a cold solution of 3:1 water/ethanol and once with 30 mL of a cold solution of 1:1 water/ethanol. The solid was dried under high vacuum to give tert-butyl 2-((diphenylmethylene)amino)acetate (I-2) as a white solid (14.9 g, 47.0 mmol, 99%). .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 7.66 (2H, m), 7.47 (3H, m), 7.41 (1H, t, J=8 Hz), 7.34 (1H, t, J=8 Hz), 7.20 (2H, m), 4.13 (2H, s), 1.48 (9H, s), MS (LC/MS) m/z observed 295.93, expected 296.16 [M+H].
[0415] Intermediate I-3
##STR00077##
(1S,2S,4S,5R)-1-(anthracen-9-ylmethyl)-2-(hydroxy(quinolin-4-yl)methyl)-5- -vinylquinuclidin-1-ium Chloride (I-3)
[0416] This intermediate was generated by a procedure based on that disclosed in Corey, E. J., et al., J. Am. Chem. Soc., 119, 12414 (1997). A round bottom flask was charged with a stir-bar, quinolin-4-yl((1S,2S,4S,5R)-5-vinylquinuclidin-2-yl)methanol (1.5 g, 5.10 mmol) also known as cinchonine, 9-(chloromethyl)anthracene (1.21 g, 5.35 mmol) and toluene (15 mL). The flask was fitted with a condenser and heated for 2 hrs at 110.degree. C. Conversion of the amine was confirmed by LCMS and the reaction was cooled to room temperature and poured into 100 mL of diethyl ether. The formed yellow precipitate was filtered and washed with 2.times.10 mL of cold DCM. The solid was set aside and the filtrate was concentrated and suspended overnight in 10% Et.sub.2O/DCM at 0.degree. C. The cold suspension was filtered. The solids were pooled together and dried on high vacuum to give the title compound (1S,2S,4S,5R)-1-(anthracen-9-ylmethyl)-2-(hydroxy(quinolin-4-yl)methyl)-5- -vinylquinuclidin-1-ium chloride (I-3) as a bright yellow solid (2.6 g, 5.0 mmol, 98%). .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 9.06 (1H, d, J=8 Hz), 8.84 (2H, d, J=4 Hz), 8.73 (1H, d, J=8 Hz), 8.20 (1H, d, J=4 Hz), 8.03 (1H, d, J=4 Hz), 7.99 (1H, s), 7.70-7.55 (3H, m), 7.40 (1H, d, J=8 Hz), 7.30-7.15 (6H, m), 7.15-7.05 (2H, m), 6.83 (1H, t, J=14 Hz), 6.68 (1H, t, J=14 Hz), 5.44 (1H, m), 4.91 (1H, dd, J=10.4 Hz), 4.74 (2H, m), 6.83 (1H, d, J=14 Hz), 6.68 (1H, d, J=14 Hz), 5.44 (1H, m), 5.27 (1H, d, J=16 Hz), 6.68 (1H, dd, J=8.3 Hz), 4.74 (2H, m), 2.59 (1H, dd, J=14.12 Hz), 2.42 (1H, m), 2.36 (2H, s), 2.13 (1H, m), 1.90-1.75 (3H, m), 1.70 (1H, m), 2.42 (1H, m), 1.12 (1H, m), 1.01 (1H, m), MS (LC/MS) m/z observed 485.08, expected 485.26 [M-C1].
[0417] Intermediate I-4
##STR00078##
(1S,2S,4S,5R)-2-((allyloxy)(quinolin-4-yl)methyl)-1-(anthracen-9-ylmethyl- )-5-vinylquinuclidin-1-ium bromide (I-4)
[0418] This catalyst was generated by a procedure based on that disclosed in Corey, E. J., et al., J. Am. Chem. Soc., 119, 12414 (1997). A round bottom flask with a stir bar was flame dried, cooled under vacuum and purged with N.sub.2. To the flask were added I-3 (1.0 g, 1.92 mmol), allyl bromide (0.5 mL 5.76 mmol) and DCM (8 mL). To the yellow foamy mixture was added a solution of 50% w/w KOH (2 mL, 9.60 mmol) at RT. A slight exotherm was observed. Analysis of the reaction by HPLC after 4 hrs showed complete conversion of I-3 and the reaction was diluted with 30 mL of DCM and water and transferred to a separatory funnel. The organic phase was collected, then extracted 2.times.20 mL of water and washed with a saturated solution of NaCl.sub.(aq). The organic phase was dried over anhydrous sodium sulphate, filtered and concentrated. To the residue was added 8 mL of methanol, which produced a clear red solution with a small amount of precipitate. Diethyl ether was slowly added to the solution at 0.degree. C. and the solution became cloudy. After the addition of 50 mL of ether the precipitate was filtered, washed once with cold ether (10 mL) and dried under high vacuum to give the title compound (1S,2S,4S,5R)-2-((allyloxy)(quinolin-4-yl)methyl)-1-(anthracen-9-ylmethyl- )-5-vinylquinuclidin-1-ium bromide (I-4) (0.63 g, 1.03 mmol, 54%). MS (LC/MS) m/z observed 525.08, expected 525.29 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0419] Intermediate I-5
##STR00079##
(S)-tert-butyl 3-(2-bromopyridin-3-yl)-2-((diphenylmethylene)amino) propanoate (I-5)
[0420] This intermediate was generated by a modified procedure based on that disclosed in Viswanathan, R., et al., J. Am. Chem. Soc., 125, 163 (2003) and Synthesis 2, 330 (2005). A three neck round bottom flask with a stir bar was charged with I-2 (40.1 g, 135.7 mmol), I-4 (8.2 g, 13.6 mmol), powdered KOH (69.1 g, 1221.4 mmol), and DCM (600 mL). The opaque yellow suspension was cooled to -78.degree. C. and the flask fitted with a dropping funnel. A suspension of I-1 (152.0 g, 610.7 mmol) in 400 mL DCM was transferred to the dropping funnel and added to the reaction at -78.degree. C. over about 1 hr. The suspension in the dropping funnel would occasionally settle and the solid would be resuspended. After the end of the addition the funnel was rinsed with an additional 200 mL of DCM and the rinse was added to the reaction. After 10 hrs at -78.degree. C. the reaction was allowed to stir overnight as it warmed to room temperature. Analysis of the reaction by HPLC and TLC showed complete conversion of I-2. The reaction was diluted with 3 L of DCM, transferred to a 15 L reactor and extracted 2.times.1 L of water. During the separation the organic phase appeared cloudy due to a solid formed from I-1. The organic phase was collected, then washed with a saturated aqueous solution of NaCl, dried over anhydrous sodium sulphate, filtered and concentrated to near dryness and purified by normal phase flash chromatography. A three solvent mobile phase was used for the separation; initially DCM/hexanes to elute the excess I-1, followed by EtOAc/Hexanes to elute the title compound (S)-tert-butyl 3-(2-bromopyridin-3-yl)-2-((diphenylmethylene)amino) propanoate (I-5) obtained as a yellow solid (23.1 g, 226.0 mmol, 37%). .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.20 (1H, dd, J=4.2 Hz), 7.60 (2H, d, J=8 Hz), 7.56 (1H, dd, J=4.2 Hz), 7.45-7.25 (6H, m), 7.12 (1H, dd, J=8.4 Hz), 6.67 (1H, d, J=d Hz), 4.39 (1H, dd, J=8.4 Hz), 3.39 (1H, dd, J=12.4 Hz), 3.21 (1H, dd, J=12.4 Hz), 1.46 (9H, s), MS (LC/MS) m/z observed 464.87, expected 465.12 [M+H].
[0421] Intermediate I-6
##STR00080##
(S)-tert-butyl 1-benzhydryl-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate (I-6)
[0422] This intermediate was generated by a modified procedure based on that disclosed in Viswanathan, R., et al., J. Am. Chem. Soc., 125, 163 (2003) and Synthesis 2, 330 (2005). A three-neck round bottom flask with a stir bar was flame dried, cooled under vacuum and purged with N.sub.2. To the flask were added I-5 (3.0 g, 6.46 mmol), tri-n-butyltin hydride (3.8 mL, 14.2 mmol), and degassed toluene (646 mL). The flask was fitted with a condenser, and a dropping funnel and heated to 85.degree. C. A solution of AIBN (1.27 g, 7.8 mmol) in 40 mL toluene was prepared in the dropping funnel and added to the reaction over the course of 1 hr. After 2 hrs the reaction was monitored by LCMS and approximately 50% conversion. Another portion of tri-n-butyltin hydride was added and the reaction was heated at 85.degree. C. for another 4 hrs. Analysis of the reaction by TLC, HPLC and LCMS showed complete conversion of I-5. The reaction was concentrated to near dryness and to the residue was added 250 mL of diethyl ether and 100 mL of a saturated solution of KF.sub.(aq). The biphasic mixture was stirred vigorously at room temperature for 3 hrs during with time a white solid formed at the interface and on the flask wall. The mixture was filtered through CELITE.TM. and the cake washed with 200 mL of diethyl ether. The filtrate was transferred to a separatory funnel, the organic phase was collected, dried over anhydrous sodium sulphate, filtered and concentrated to near dryness. The residue was purified by normal phase flash chromatography (EtOAc/Hexanes) to give the title compound I-6 as an off-white solid (1.37 g, 3.5 mmol, 55%). .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 7.87 (1H, d, J=4 Hz), 7.41 (2H, d, J=8 Hz), 7.35-7.15 (8H, m), 6.55-6.45 (2H, m), 4.21 (1H, dd, J=10.6 Hz), 3.43 (1H, dd, J=18.10 Hz), 3.21 (1H, dd, J=18.6 Hz), 1.73 (9H, s), MS (LC/MS) m/z observed 487.04, expected 487.21 [M+H].
[0423] Intermediate I-7
##STR00081##
(2S)-2-Carboxy-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-ium (I-7)
[0424] This intermediate was generated by a modified procedure based on that disclosed in Viswanathan, R., et al., J Am. Chem. Soc., 125, 163 (2003) and Synthesis 2, 330 (2005). A round bottom flask was charged with I-6 (670 mg, 1.7 mmol), DCM (5 mL) and triethylsilane (1 mL, 8.65 mmol). To the clear yellow solution was added TFA (3.3 mL) at room temperature and the yellow/orange reaction was stirred at room temperature for 16 hrs. Analysis of the reaction by HPLC showed complete conversion of I-6 and the reaction was concentrated to approximately one quarter of the volume. Diethyl ether (60 mL) was added slowly to the residue, which resulted in the precipitation of a fine white solid. The mixture was cooled to 0.degree. C. for 10 min then sonicated and filtered. The white solid was washed with 10 mL of cold diethyl ether to give (2S)-2-carboxy-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-ium trifluoroacetate (I-7) (263 mg, 0.93 mmol, 54%). .sup.1H NMR (400 MHz, DMSO) .delta. 8.52 (1H, bs), 7.69 (1H, d, J=4 Hz), 7.55 (1H, d, J=4 Hz), 6.68 (2H, dd, J=8.4 Hz), 4.59 (1H, dd, J=12.4 Hz), 3.45 (1H, dd, J=16.12 Hz), 3.15 (1H, dd, J=20.4 Hz), MS (LC/MS) m/z observed 165.02, expected 165.07 [M-C1].
[0425] Intermediate I-8
##STR00082##
2-Phenylacetic acid (I-8)
[0426] A solution of methyl 2-phenylacetate (10 g, 64 mmol) in methanol (60 ml) was treated with solution of sodium hydroxide (5.1 g, 127 mmol) in water (40 ml) at 70.degree. C. for 3 hrs. The resulting mixture was concentrated under vacuum to remove the methanol. The residue was diluted with water (40 ml) and washed with diethyl ether (40 ml). The separated water layer was acidified to pH 2 using a mixture of water and HCl (1:1) and extracted with DCM (3.times.80 ml). Combined organic extracts were washed with brine, 80 ml, separated, dried over sodium sulfate and concentrated to give 2-phenylacetic acid (I-8) as a white solid (9 g, 96%) used without further characterization. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 3.64 (2H, s), 7.27-7.35 (5H, m), 11.5 (1H, bs).
[0427] Intermediate I-9
##STR00083##
(2S,3S)-3-Methyl-2-(2-phenylacetamido)pentanoic acid (I-9)
[0428] I-8 (2.0 g, 14.7 mmol) and thionyl chloride (6.6 ml, 90.3 mmol) were stirred together for 1 hr at RT. Thionyl chloride was removed by distillation under vacuum. The acid chloride was added to the stirring solution of L-isoleucine (1.75 g, 13.4 mmol) in NaOH (2N, 17 ml) at 0.degree. C. The resulting mixture was warmed to RT and stirred overnight. The mixture was washed with diethyl ether (20 ml) and acidified to pH 4-5 by adding citric acid (aqueous, saturated solution). The precipitated solid was filtered, washed with diethyl ether and dried to yield (2S,3S)-3-methyl-2-(2-phenylacetamido)pentanoic acid (I-9) as a white solid (1.6 g, 44%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.78-0.82 (6H, t, J=8 Hz), 1.12-1.18 (1H, m), 1.34-1.40 (1H, m), 1.72-1.78 (1H, m), 3.41-3.53 (2H, q, J=16 Hz), 4.13-4.16 (1H, dd, J=4.12 Hz), 7.15-7.19 (1H, m), 7.22-7.28 (1H, m), 8.19-8.21 (1H, d, J=8 Hz), 12.54 (1H, s), MS (LC/MS) m/z observed 250.02, expected 250.14 [M+H].
[0429] Intermediate I-10
##STR00084##
(S)-3-methyl-2-(2-phenylacetamido)butanoic (I-10)
[0430] I-8 (0.5 g, 3.67 mmol) and thionyl chloride (1.6 ml, 22 mmol) were stirred together for 1 hr at room temperature. Thionyl chloride was removed by distillation under vacuum. The acid chloride was added to the stirring solution of L-valine (0.39 g, 3.31 mmol) in NaOH (2N, 4.2 ml) at 0.degree. C. The resulting reaction mixture was warmed to RT and stirred overnight. The mixture was washed with diethyl ether (5 ml) and acidified to pH 4-5 by adding citric acid (aqueous, saturated solution). The precipitated solid was filtered, washed with diethyl ether and dried to yield (S)-3-methyl-2-(2-phenylacetamido)butanoic acid (I-10) as a white solid (0.64 g, 74%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82-0.84 (3H, d, J=8 Hz), 0.85-0.87 (3H, d, J=8 Hz), 1.99-2.06 (1H, m), 3.44-3.55 (2H, q, J=12 Hz), 4.10-4.14 (1H, dd, J=8.12 Hz), 7.16-7.21 (1H, m), 7.24-7.29 (4H, m), 7.19-7.21 (1H, d, J=8 Hz), 12.55 (1H, s), MS (LC/MS) m/z observed 236.04, expected 236.13 [M+H].
[0431] Intermediate I-11
##STR00085##
Ethyl 2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)aceta- te (I-11)
[0432] Boc-L-Isoleucine and glycine ethyl ester hydrochloride were combined using method A except the purification was performed on normal phase using 0% to 30% ethyl acetate in hexanes as the eluent to yield ethyl 2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)aceta- te (I-11). .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 0.91 (3H, t, J=7 Hz), 0.96 (3H, d, J=7 Hz), 1.14 (1H, m), 1.28 (3H, t, J=7 Hz), 1.45 (9H, s), 1.51 (1H, m), 1.92 (1H, m), 3.95-4.12 (3H, m), 4.22 (2H, q, J=7 Hz), 5.55 (1H, d, J=9 Hz), 6.52 (1H, bs), MS (LC/MS) m/z observed 317.42, expected 317.21 [M+H].
[0433] Intermediate I-12
##STR00086##
Ethyl 2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)aceta- te (I-12)
[0434] Intermediate I-12 was prepared from I-11 using method D with 2 eq. of LiOH.H.sub.2O. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.74-0.85 (6H, m), 1.08 (1H, m), 1.31-1.41 (10H, m), 1.71 (1H, m), 3.38-4.50 (2H, m), 3.80 (1H, t, J=8 Hz), 6.85 (1H, d, J=9 Hz), 7.50 (1H, bs), MS (LC/MS) m/z observed 288.88, expected 289.18 [M+H].
Representative Granzyme B Inhibitor Compounds
[0435] The following is a description of the preparation of representative Granzyme B inhibitor compounds of the invention.
[0436] Example A1 was prepared by the representative synthetic pathway illustrated schematically in FIG. 1.
Example A1
(S)-1-(2-((2S,3S)-2-(2-(2H-TETRAZOL-5-YL)ACETAMIDO)-3-METHYLPENTANAMIDO)AC- ETYL)-N-((2H-TETRAZOL-5-YL)METHYL)-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRIDINE-2- -CARBOXAMIDE
[0437] Ethyl 2-((2S,3S)-2-(2-(1H-tetrazol-5-yl)acetamido)-3-methylpentanamido) acetate (620 mg, 1.64 mmol, 57%) was collected as an off-white solid from I-12 (0.91 g, 2.88 mmol) and 2-(2H-tetrazol-5-yl)acetic (307 mg, 2.4 mmol) using method A in DMF. MS (LC/MS) m/z observed 326.86, expected 327.18 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0438] A round bottom flask was charged with a stir bar, ethyl 2-((2S,3S)-2-(2-(1H-tetrazol-5-yl)acetamido)-3-methylpentanamido) acetate (290 mg, 0.89 mmol), LiOH (94 mg, 2.23 mmol), tert-BuOH (6.6 ml), and water (3.3 mL). The reaction was stirred at room temperature for 2 hrs. Analysis of the reaction by LC/MS showed complete conversion and concentrated HCl (aqueous) was added to reach pH 2. The reaction was concentrated under reduced pressure and reconcentrated from tert-BuOH. The remaining off white solid contained 2-((2S,3S)-2-(2-(1H-tetrazol-5-yl)acetamido)-3-methylpentanamido)acetic acid. (MS (LC/MS) m/z observed 298.89, expected 299.15 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0439] (S)-1-(2-((2S,3S)-2-(2-(1H-Tetrazol-5-yl)acetamido)-3-methylpentana- mido)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and 2-((2S,3S)-2-(2-(1H-tetrazol-5-yl)acetamido)-3-methylpentanamido)acetic acid (3 eq.) using method C in DMF. (LC/MS) m/z observed 445.04, expected 445.19 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0440] (S)-1-(2-((2S,3S)-2-(2-(1H-Tetrazol-5-yl)acetamido)-3-methylpentana- mido)acetyl)-N-((2H-tetrazol-5-yl)methyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyr- idine-2-carboxamide (A1) was prepared from (S)-1-(2-((2S,3S)-2-(2-(1H-tetrazol-5-yl)acetamido)-3-methylpentanamido)a- cetyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82 (3H, t, J=7 Hz), 0.90 (3H, d, J=7 Hz), 1.12 (1H, m), 1.48 (1H, m), 1.75 (1H, m), 2.99 (1H, dd, J=4, 17 Hz), 3.46 (1H, dd, J=11, 18 Hz), 3.93-4.04 (2H, m), 4.31 (1H, t, J=8 Hz), 4.45-4.53 (2H, m), 4.62 (1H, dd, J=6, 16 Hz), 4.71 (1H, dd, J=4, 18 Hz), 4.96 (1H, dd, J=4, 11 Hz), 7.03 (1H, dd, J=5, 7 Hz), 7.65 (1H, d, J=7 Hz), 8.15 (1H, d, J=5 Hz), 8.35 (1H, t, J=6 Hz), 8.47 (1H, d, J=9 Hz), 8.95 (1H, t, J=5 Hz), (MS (LC/MS) m/z observed 526.06, expected 526.24 [M+H].
[0441] Examples C1-C41 were prepared by the representative synthetic pathway illustrated schematically in FIG. 3.
Example C1
3-{[(1S,2S)-2-METHYL-1-({2-OXO-2-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)- CARBAMOYL]-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRIDIN-1-YL]ETHYL}CARBAMOYL)BUTYL- ]CARBAMOYL}PROPANOIC ACID
[0442] Intermediate I-7 (600 mg, 2.160 mmol) was suspended in EtOH (40 mL) at 0.degree. C. and thionyl chloride (0.313 mL, 4.320 mmol, 2 eq.) was added dropwise. The resulting clear mixture was allowed to come to RT and stirred for 16 hours. The reaction mixture was then concentrated to dryness and swapped with EtOH (2.times.25 mL). The solid obtained was dried well under reduced pressure to give (S)-ethyl 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate as a white solid (494 mg, quantitative). MS (LC/MS) m/z observed 193.52, expected 193.10 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0443] (S)-Ethyl 1-(2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)acetyl)-- 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate was prepared from (S)-ethyl 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate and I-12 using method C. (LC/MS) m/z observed 463.05, expected 463.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0444] (S)-1-(2-((2S,3S)-2-((tert-Butoxycarbonyl)amino)-3-methylpentanamid- o)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from (S)-ethyl 1-(2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)acetyl)-- 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate using method D with 2 eq of LiOH.H.sub.2O. MS (LC/MS) m/z observed 435.05, expected 435.22 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0445] tert-Butyl ((2S,3S)-1-((2-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1- H-pyrrolo[2,3-b]pyridin-1-yl)-2-oxoethyl)amino)-3-methyl-1-oxopentan-2-yl)- carbamate was prepared from (S)-1-(2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)acet- yl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 516.04, expected 516.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0446] 3-{[(1S,2S)-2-Methyl-1-({2-oxo-2-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-yl- methyl)carbamoyl]-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl]ethyl}carbamoy- l)butyl]carbamoyl}propanoic acid (C1) was prepared from tert-butyl ((2S,3S)-1-((2-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1- H-pyrrolo[2,3-b]pyridin-1-yl)-2-oxoethyl)amino)-3-methyl-1-oxopentan-2-yl)- carbamate and succinic anhydride using method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.81 (3H, t, J=7 Hz), 0.87 (3H, d, J=7 Hz), 1.10 (1H, m), 1.46 (1H, m), 1.72 (1H, m), 2.34-2.46 (4H, m), 3.00 (1H, dd, J=4, 17 Hz), 3.42 (1H, dd, J=11, 18 Hz), 4.24 (1H, t, J=8 Hz), 4.43-4.52 (2H, m), 4.58-4.74 (2H, m), 4.96 (1H, dd, J=4, 11 Hz), 7.00 (1H, dd, J=5, 7 Hz), 7.64 (1H, d, J=7 Hz), 7.91 (1H, d, J=9 Hz), 8.12-8.18 (2H, m), 8.92 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 516.11, expected 516.23 [M+H].
Example C2
(S)-N-((1H-1,2,3-TRIAZOL-4-YL)METHYL)-1-(2-((2S,3S)-3-METHYL-2-(2-PHENYLAC- ETAMIDO)PENTANAMIDO)ACETYL)-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRIDINE-2-CARBOX- AMIDE
[0447] (S)-1-(2-((tert-Butoxycarbonyl)amino)acetyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Boc-glycine (3 eq.) using method C in DMF. (LC/MS) m/z observed 322.63, expected 322.14 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0448] (S)-tert-Butyl (2-(2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2- ,3-b]pyridin-1-yl)-2-oxoethyl)carbamate was prepared from (S)-1-(2-((tert-butoxycarbonyl)amino)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b- ]pyridine-2-carboxylic acid and (2H-1,2,3-triazol-4-yl)methyl-amine using method A but without HCl treatment. MS (LC/MS) m/z observed 402.55, expected 402.19 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0449] (S)-N-((1H-1,2,3-Triazol-4-yl)methyl)-1-(2-((2S,3S)-3-methyl-2-(2-p- henylacetamido)pentanamido)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2- -carboxamide (C2) was prepared from (S)-tert-butyl (2-(2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2- ,3-b]pyridin-1-yl)-2-oxoethyl)carbamate and I-9 using method A. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76-0.87 (6H, m), 1.10 (1H, m), 1.44 (1H, m), 1.74 (1H, m), 2.93 (1H, dd, J=4, 17 Hz), 3.40-3.62 (3H, m), 4.27 (1H, m), 4.32-4.37 (2H, m), 4.47 (1H, dd, J=5, 18 Hz), 4.72 (1H, dd, J=6, 18 Hz), 4.94 (1H, dd, J=4, 11 Hz), 7.02 (1H, dd, J=5, 7 Hz), 7.20 (1H, m), 7.24-7.30 (4H, m), 7.60-7.70 (2H, m), 8.10-8.18 (2H, m), 8.25 (1H, t, J=6 Hz), 8.72 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 533.11, expected 533.26 [M+H].
Example C3
(S)-N-((2H-TETRAZOL-5-YL)METHYL)-1-(2-((2S,3S)-3-METHYL-2-(2-PHENYLACETAMI- DO)PENTANAMIDO)ACETYL)-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRIDINE-2-CARBOXAMIDE
[0450] (S)-tert-Butyl (2-(2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]- pyridin-1-yl)-2-oxoethyl)carbamate was prepared from (S)-1-(2-((tert-butoxycarbonyl)amino)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b- ]pyridine-2-carboxylic acid (from Example C2) and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 403.35, expected 403.18 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0451] (S)-N-((2H-Tetrazol-5-yl)methyl)-1-(2-((2S,3S)-3-methyl-2-(2-phenyl- acetamido)pentanamido)acetyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carb- oxamide (C3) was prepared from (S)-tert-butyl (2-(2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]- pyridin-1-yl)-2-oxoethyl)carbamate and I-9 using method A. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76-0.87 (6H, m), 1.08 (1H, m), 1.35 (1H, m), 1.78 (1H, m), 2.98 (1H, dd, J=4, 17 Hz), 3.40-3.62 (3H, m), 4.42 (1H, m), 4.48-4.54 (2H, m), 4.58-4.75 (2H, m), 4.97 (1H, m), 7.02 (1H, dd, J=5, 7 Hz), 7.20 (1H, m), 7.24-7.30 (4H, m), 7.64 (1H, d, J=7 Hz), 7.99 (1H, d, J=9 Hz), 8.10-8.18 (2H, m), 8.95 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 556.15, expected 556.24 [M+Na].
Example C4
(S)-5-((S)-2(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[2- ,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTANAMIDO- )-5-OXOPENTANOIC ACID
[0452] To a suspension of Boc-L-glutamic acid y-benzyl ester (2.54 g, 7.2 mmol, 4.0 eq.) in a mixture of DCM:DMF (29 ml, 5:1 (v/v)) was added HATU (1.0 g, 2.7 mmol, 1.5 eq.), then DIPEA (1.6 ml, 9.0 mmol, 5.0 eq.) in that order. The reaction mixture was stirred for 20 minutes whereupon the reaction mixture became a yellow solution. Intermediate I-7 (0.5 g, 1.8 mmol, 1.0 eq.) was added and the reaction mixture was stirred and additional 30 minutes. The reaction mixture was concentrated under vacuum and was purified on a C18 column using 10-65% MeOH in water to yield the (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid as an off-orange solid. MS (LC/MS) m/z observed 484, expected 484.20 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0453] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate was prepared from (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid via method O but without the second amine addition. MS (LC/MS) m/z observed 565, expected 565.24 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0454] (S)-Benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate and Boc-L-isoleucine via method A but without swapping with MeOH; and DMF as the solvent of the coupling step. MS (LC/MS) m/z observed 678; expected 678.33 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0455] To a solution of (S)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpent- anamido)-5-oxopentanoate benzyl ester (327 mg, 0.48 mmol, 1.0 eq.) in dioxane (3 ml) was added hydrochloric acid (2 ml, 4.0 M solution in dioxane, 8.0 mmol, 16 eq.), then stirred at RT for 2 h whereupon a white paste forms upon the walls of the reaction vessel. The reaction mixture was concentrated under vacuum to dryness, then methanol (6 ml), DIPEA (0.67 ml, 3.8 mmol, 8.0 eq.), and succinic anhydride (236.9 mg, 2.4 mmol, 5.0 eq.) were added in that order. The reaction mixture was stirred at RT for 1 hr, then acidified to pH<4 with formic acid, then concentrated under vacuum to dryness. The reaction vessel was purged with nitrogen, then palladium on carbon (98 mg, 10 wt %, wetted) and MeOH (10 ml) were added in that order. The atmosphere changed to hydrogen (vacuum+H.sub.2 backfill.times.3) and the suspension of black solids was stirred for 3 hr, then filtered over a pad of CELITE.TM. and washed with excess MeOH. The reaction mixture was concentrated under vacuum and purified on a C18 column using 10-60% MeOH in water to yield the title compound (S)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpentanamid- o)-5-oxopentanoic acid (C4) as a white solid (166 mg, 59% over 3 steps). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.72-0.91 (m, 6H), 1.08 (ddd, J=14.4, 10.7, 4.7 Hz, 1H), 1.35-1.47 (m, 1H), 1.59-1.73 (m, 1H), 1.77-1.92 (m, 1H), 1.94-2.07 (m, 1H), 2.25-2.47 (m, 6H), 2.89-3.01 (m, 1H), 3.36-3.53 (m, 1H), 4.20 (t, J=8.2 Hz, 1H), 4.47 (dd, J=15.8, 5.0 Hz, 1H), 4.69 (dd, J=15.9, 6.0 Hz, 1H), 4.99 (dd, J=11.1, 3.9 Hz, 1H), 5.84 (s, 1H), 7.02 (dd, J=7.3, 5.0 Hz, 1H), 7.65 (d, J=7.4 Hz, 1H), 7.83 (d, J=8.8 Hz, 1H), 8.10-8.22 (m, 2H), 8.97 (t, J=5.5 Hz, 1H). MS (LC/MS) m/z observed 588, expected 588.25 [M+H].sup.+.
Example C5
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-ACETAMIDO-3-METHYLPENTANAMIDO)-5-OXOPENTA- NOIC ACID
[0456] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-2-acetamido-3-methylpentanamido)-5-oxopentanoa- te was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate from Example C4 and acetic acid via method A but with DMF as the solvent. MS (LC/MS) m/z observed 620, expected 620.29 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0457] (S)-5-((S)-2-(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-p- yrrolo[2,3-b]pyridin-1-yl)-4-((2S,3S)-2-acetamido-3-methylpentanamido)-5-o- xopentanoic acid (C5) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-2-acetamido-3-methylpentanamido)-5-oxopentanoa- te via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.72-0.87 (m, 6H), 0.99-1.13 (m, 1H), 1.33-1.46 (m, 1H), 1.62 (q, J=8.4, 7.9 Hz, 1H), 1.75-1.88 (m, 4H), 1.91-2.04 (m, 1H), 2.37 (td, J=11.5, 5.1 Hz, 2H), 2.93 (dd, J=17.6, 3.9 Hz, 1H), 3.42 (dd, J=17.4, 11.3 Hz, 1H), 4.15 (t, J=8.3 Hz, 1H), 4.44 (dd, J=15.8, 5.0 Hz, 1H), 4.65 (dd, J=15.9, 6.0 Hz, 1H), 4.96 (dd, J=11.3, 3.9 Hz, 1H), 5.81 (ddd, J=10.9, 7.2, 3.7 Hz, 1H), 7.00 (dd, J=7.4, 5.1 Hz, 1H), 7.63 (d, J=7.4 Hz, 1H), 7.82 (d, J=8.7 Hz, 1H), 8.07-8.22 (m, 2H), 8.92 (t, J=5.5 Hz, 1H). MS (LC/MS) m/z observed 503, expected 530.24 [M+H].sup.+.
Example C6
3-{[(1S,2S)-2-METHYL-1-{[(2S)-1-OXO-1-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLME- THYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PROPAN-2-YL]CARBAMOYL}- BUTYL]CARBAMOYL}PROPANOIC ACID
[0458] (S)-1-((S)-2-((tert-Butoxycarbonyl)amino)propanoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Boc-L-alanine (3 eq.) using method C in DMF. (LC/MS) m/z observed 335.85, expected 336.16 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0459] tert-Butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)carbamate was prepared from (S)-1-((S)-2-((tert-butoxycarbonyl)amino)propanoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 416.85, expected 417.20 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0460] tert-Butyl ((2S,3S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihyd- ro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)amino)-3-methyl-1-oxope- ntan-2-yl)carbamate was prepared from tert-butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)carbamate and Boc-L-Isoleucine using method A. MS (LC/MS) m/z observed 529.91, expected 530.28 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0461] Title compound 3-{[(1S,2S)-2-methyl-1-{[(2S)-1-oxo-1-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylm- ethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]propan-2-yl]carbamoyl- }butyl]carbamoyl}propanoic acid (C6) was prepared from tert-butyl ((2S,3S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihyd- ro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)amino)-3-methyl-1-oxope- ntan-2-yl)carbamate and succinic anhydride using method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76 (3H, t, J=7 Hz), 0.80 (3H, d, J=7 Hz), 1.05 (1H, m), 1.28 (3H, d, J=7 Hz), 1.40 (1H, m), 1.63 (1H, m), 2.29-2.42 (4H, m), 2.93 (1H, dd, J=4, 17 Hz), 3.42 (1H, dd, J=11, 18 Hz), 4.16 (1H, t, J=8 Hz), 4.44 (1H, dd, J=5, 16 Hz), 4.64 (1H, dd, J=6, 16 Hz), 4.96 (1H, dd, J=4, 11 Hz), 5.82 (1H, m), 6.99 (1H, dd, J=5, 7 Hz), 7.63 (1H, d, J=7 Hz), 7.79 (1H, d, J=9 Hz), 8.12 (1H, d, J=6 Hz), 8.18 (1H, d, J=7 Hz), 8.90 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 529.97, expected 530.25 [M+H].
Example C7
(3S)-3-[(2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTANAMIDO]-4-OXO-4-[(2S- )-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYR- IDIN-1-YL]BUTANOIC ACID
[0462] (S)-1-((S)-4-(tert-Butoxy)-2-((tert-butoxycarbonyl)amino)-4-oxobuta- noyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-2-carboxylic acid was prepared in the same manner as (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid in Example C4, starting from Boc-L-aspartic acid .beta.-tert-butyl ester. MS (LC/MS) m/z observed 436; expected 436.20 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0463] (S)-tert-Butyl 4-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-3-((tert-butoxycarbonyl)amino)-4-oxobutanoate was prepared from (S)-1-((S)-4-(tert-butoxy)-2-((tert-butoxycarbonyl)amino)-4-oxobutan- oyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-2-carboxylic acid via method O but without the second amine addition. MS (LC/MS) m/z observed 517; expected 517.24 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0464] (S)-4-((S)-2(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-py- rrolo[2,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-meth- ylpentanamido)-4-oxobutanoic acid was prepared from (S)-tert-butyl 4-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-3-((tert-butoxycarbonyl)amino)-4-oxobutanoate and Boc-L-isoleucine (4 eq.) via method A but without swapping with MeOH; and DMF as the solvent of the coupling step. MS (LC/MS) m/z observed 574; expected 574.27 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0465] Title compound (3S)-3-[(2S,3S)-2-(3-carboxypropanamido)-3-methylpentanamido]-4-oxo-4-[(2- S)-2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]py- ridin-1-yl]butanoic acid (C7) was prepared from (S)-4-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-H-pyrrolo[2- ,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpenta- namido)-4-oxobutanoic acid via method I but with MeOH as the solvent for anhydride opening. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.72-0.89 (m, 6H), 1.10-1.24 (m, 1H), 1.32-1.48 (m, 1H), 1.67-1.82 (m, 1H), 2.26-2.47 (m, 6H), 2.71-2.81 (m, 1H), 2.97 (d, J=17.3 Hz, 1H), 3.45 (dd, J=17.4, 11.2 Hz, 1H), 4.12-4.26 (m, 3H), 4.46 (dd, J=15.9, 5.2 Hz, 1H), 4.62 (td, J=14.5, 13.2, 6.2 Hz, 1H), 4.96 (dd, J=11.1, 3.5 Hz, 1H), 5.97-6.08 (m, 1H), 7.00 (dd, J=7.4, 5.2 Hz, 1H), 7.64 (d, J=7.4 Hz, 1H), 7.75 (d, J=9.3 Hz, 1H), 7.95-8.07 (m, 2H), 8.25 (d, J=6.7 Hz, 1H), 8.86 (t, J=5.8 Hz, 1H). MS (LC/MS) m/z observed 574, expected 574.23 [M+H].sup.+.
Example C8
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((S)-2-(3-CARBOXYPROPANAMIDO)-2-CYCLOPENTYLACETAMIDO- )-5-OXOPENTANOIC ACID
[0466] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-2-cyclopentylacetam- ido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) and boc-L-cyclopentylglycine dicyclohexylammonium salt via method A but with DMF as the solvent. MS (LC/MS) m/z observed 690, expected 690.33 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0467] 4-(((S)-2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)- amino)-1-cyclopentyl-2-oxoethyl)amino)-4-oxobutanoic acid was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-2-cyclopentylacetam- ido)-5-oxopentanoate via method I. MS (LC/MS) m/z observed 690, expected 690.29 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0468] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((S)-2-(3-carboxypropanamido)-2-cyclopentylacetamid- o)-5-oxopentanoic acid (C8) was prepared from 4-(((S)-2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydr- o-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)- -1-cyclopentyl-2-oxoethyl)amino)-4-oxobutanoic acid via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.15-1.63 (m, 8H), 1.92-2.01 (m, 1H), 2.23-2.44 (m, 6H), 2.91 (d, J=17.6 Hz, 1H), 4.13-4.26 (m, 2H), 4.35-4.51 (m, 1H), 4.92-5.04 (m, 1H), 5.75-5.89 (m, 1H), 6.73-6.85 (m, 1H), 6.95-7.02 (m, 1H), 7.62 (d, J=7.3 Hz, 1H), 7.88 (d, J=8.6 Hz, 1H), 8.07-8.17 (m, 2H), 8.31-8.41 (m, 1H). MS (LC/MS) m/z observed 600, expected 600.25 [M+H].sup.+.
Example C9
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-3-METHYL-2-(2-PHENYLACETAMIDO)PENTANAMIDO)-- 5-OXOPENTANOIC ACID
[0469] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(2-phenylacetamido)pentanamido)-5-o- xopentanoate was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) and intermediate I-9 via method A but without swapping with MeOH; and DMF as the solvent of the coupling step. MS (LC/MS) m/z observed 696, expected 696.32 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0470] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(2-phenylacetamido)pentanamido)- -5-oxopentanoic acid (C9) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(2-phenylacetamido)pentanamido)-5-o- xopentanoate via Method Q. C9 exists as a mixture of rotamers in a 1.5:1 ratio; only the major signals are reported. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.71-0.88 (m, 7H), 0.98-1.16 (m, 1H), 1.33-1.47 (m, 1H), 1.74-1.92 (m, 1H), 1.95-2.07 (m, 1H), 2.20-2.45 (m, 2H), 2.96 (d, J=17.1 Hz, 1H), 3.39-3.67 (m, 4H), 4.20 (t, J=8.3 Hz, 1H), 4.48 (dt, J=15.8, 4.8 Hz, 1H), 4.67 (dt, J=14.7, 7.0 Hz, 1H), 4.99 (dd, J=11.2, 3.8 Hz, 1H), 5.85 (tt, J=7.5, 3.6 Hz, 1H), 7.03 (dd, J=7.3, 5.2 Hz, 1H), 7.17-7.23 (m, 1H), 7.23-7.31 (m, 5H), 7.66 (d, J=7.3 Hz, 1H), 8.02 (d, J=8.9 Hz, 1H), 8.14-8.18 (m, 1H), 8.21 (dd, J=7.5, 3.2 Hz, 1H), 8.96 (q, J=5.2 Hz, 1H). MS (LC/MS) m/z observed 606, expected 606.27 [M+H].sup.+.
Example C10
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((S)-3-METHYL-2-(2-PHENYLACETAMIDO)BUTANAMIDO)-5-OXO- PENTANOIC ACID
[0471] To a solution of (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) (85 mg, 0.15 mmol, 1.0 eq.) in dioxane (1 ml) was added hydrochloric acid (1.5 ml, 4.0 M solution in dioxane, 6.0 mmol, 40 eq.), then stirred at RT for 2 h whereupon a white paste forms upon the walls of the reaction vessel. The reaction mixture was concentrated under vacuum to dryness and coupled to intermediate I-10 via method C but with DMF as the solvent to obtain the (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-3-methyl-2-(2-phenylacetamido)butanamido)-5-oxopen- tanoate. MS (LC/MS) m/z observed 682, expected 682.30 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0472] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((S)-3-methyl-2-(2-phenylacetamido)butanamido)-5-ox- opentanoic acid (C10) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-3-methyl-2-(2-phenylacetamido)butanamido)-5-oxopen- tanoate via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82 (dd, J=11.7, 6.7 Hz, 6H), 1.74-2.05 (m, 3H), 2.37 (q, J=13.9, 11.9 Hz, 1H), 2.96 (d, J=17.7 Hz, 1H), 3.21-3.62 (m, 4H), 4.20 (t, J=7.9 Hz, 1H), 4.46 (dd, J=15.8, 5.0 Hz, 1H), 4.67 (dd, J=15.9, 6.0 Hz, 1H), 4.93-5.04 (m, 1H), 5.84 (s, 1H), 7.03 (dd, J=7.3, 5.1 Hz, 1H), 7.21 (d, J=6.7 Hz, 1H), 7.27 (d, J=6.4 Hz, 4H), 7.66 (d, J=7.3 Hz, 1H), 7.98 (d, J=8.8 Hz, 1H), 8.16 (d, J=5.2 Hz, 1H), 8.21 (d, J=7.4 Hz, 1H), 8.94 (t, J=5.4 Hz, 1H). MS (LC/MS) m/z observed 592, expected 592.26 [M+H].sup.+.
Example C11
(S)-5-((S)-2(((1H-1,2,3-TRIAZOL-4-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRR- OLO[2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTAN- AMIDO)-5-OXOPENTANOIC ACID
[0473] (S)-Benzyl 5-((S)-2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate was prepared from (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid (from Example C4) via method O but with 1H-1,2,3-triazole methylamine hydrochloride in place of (2H-tetrazol-5-yl)methylamine and without the second amine addition. MS (LC/MS) m/z observed 564, expected 564.25 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0474] (S)-Benzyl 5-((S)-2(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpen- tanamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate and Boc-L-isoleucine via method A but without swapping with MeOH; and DMF as the solvent for the coupling step described therein. MS (LC/MS) m/z observed 677; expected 677.33 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0475] Title compound (S)-5-((S)-2(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyr- rolo[2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpenta- namido)-5-oxopentanoic acid (C11) was prepared in the same manner as (S)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpentanamid- o)-5-oxopentanoic acid (from Example C4) starting from (S)-benzyl 5-((S)-2(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpen- tanamido)-5-oxopentanoate. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.73-0.88 (m, 6H), 1.01-1.15 (m, 1H), 1.33-1.46 (m, 1H), 1.59-1.74 (m, 1H), 1.78-1.95 (m, 1H), 1.95-2.08 (m, OH), 2.27-2.45 (m, 7H), 2.88 (d, J=17.2 Hz, 1H), 3.40-3.49 (m, 2H), 4.21 (t, J=8.1 Hz, 1H), 4.30 (dd, J=15.3, 5.4 Hz, 1H), 4.39 (dd, J=15.4, 5.7 Hz, 1H), 4.95 (dd, J=11.3, 4.1 Hz, 1H), 5.79-5.90 (m, 1H), 6.99-7.06 (m, 1H), 7.63 (d, J=7.5 Hz, 1H), 7.67 (s, 1H), 7.84 (d, J=8.8 Hz, 1H), 8.09-8.21 (m, 2H), 8.64-8.74 (m, 1H). MS (LC/MS) m/z observed 587, expected 587.25 [M+H].sup.+.
Example C12
(4S)-4-[(2S,3S)-3-METHYL-2-[2-(2H-1,2,3,4-TETRAZOL-5-YL)ACETAMIDO]PENTANAM- IDO]-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-- PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0476] (4S)-4-[(2S,3S)-3-Methyl-2-[2-(2H-1,2,3,4-tetrazol-5-yl)acetamido]p- entanamido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H- ,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid benzyl ester was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate from Example C4 and 2H-tetrazole-5-acetic acid via method A. MS (LC/MS) m/z observed 688, expected 688.30 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0477] Title compound (4S)-4-[(2S,3S)-3-methyl-2-[2-(2H-1,2,3,4-tetrazol-5-yl)acetamido]pentana- mido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H- -pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C12) was prepared from (S)-benzyl 4-((2S,3S)-2-(2-(1H-tetrazol-5-yl)methyl)acetamido)-3-methylpentamamido)-- 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-b- ]pyridin-1-yl)-5-oxopentanoate via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.81 (t, J=7.4 Hz, 3H), 0.86 (d, J=6.8 Hz, 3H), 0.99-1.23 (m, 1H), 1.44 (s, 1H), 1.69 (s, 1H), 1.85 (d, J=10.9 Hz, 1H), 1.94-2.06 (m, 1H), 2.27-2.46 (m, 2H), 2.96 (d, J=17.4 Hz, 1H), 3.40-3.52 (m, 1H), 3.88-4.03 (m, 2H), 4.25 (t, J=8.1 Hz, 1H), 4.48 (dd, J=15.9, 5.1 Hz, 1H), 4.69 (dd, J=16.0, 6.0 Hz, 1H), 4.94-5.05 (m, 1H), 5.79-5.91 (m, 1H), 6.98-7.06 (m, 1H), 7.66 (d, J=7.4 Hz, 1H), 8.15 (d, J=5.1 Hz, 1H), 8.29-8.39 (m, 2H), 8.97 (bs, 1H). MS (LC/MS) m/z observed 598, expected 598.25 [M+H].sup.+.
Example C13
(S)-BENZYL 5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-P- YRROLO[2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-ACETAMIDO-3-METHYLPENTANAMIDO)-5-O- XOPENTANOATE
[0478] To a solution of (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) (85 mg, 0.15 mmol, 1.0 eq.) in dioxane (1 ml) was added hydrochloric acid (1.5 ml, 4.0 M solution in dioxane, 6.0 mmol, 40 eq.), then stirred at RT for 2 h whereupon a white paste forms upon the walls of the reaction vessel. The reaction mixture was concentrated under vacuum to dryness and coupled to intermediate N-acetyl-L-valine via method C but with DMF as the solvent to obtain the title compound (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-2-acetamido-3-methylpentanamido)-5-oxopentanoa- te (C13). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.79-0.82 (d, J=6.7 Hz, 3H), 0.85 (d, J=6.7 Hz, 3H), 1.84 (s, 3H), 1.86-1.96 (m, 1H), 2.96 (d, J=17.2 Hz, 1H), 3.17 (s, 1H), 3.38-3.50 (m, 2H), 4.15 (t, J=8.0 Hz, 1H), 4.44 (d, J=16.9 Hz, 1H), 4.65 (dd, J=15.8, 6.1 Hz, 1H), 4.99 (d, J=10.7 Hz, 1H), 5.08 (s, 2H), 5.79-5.92 (m, 1H), 7.01 (t, J=6.3 Hz, 1H), 7.25-7.42 (m, 5H), 7.65 (d, J=7.5 Hz, 1H), 7.81 (d, J=8.7 Hz, 1H), 8.14 (d, J=5.1 Hz, 1H), 8.18 (d, J=7.4 Hz, 1H), 8.87 (s, 1H). MS (LC/MS) m/z observed 606, expected 606.27 [M+H].sup.+.
Example C14
(S)-N-((2H-TETRAZOL-5-YL)METHYL)-1-((S)-2-((2S,3S)-3-METHYL-2-(2-PHENYLACE- TAMIDO)PENTANAMIDO)PROPANOYL)-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRIDINE-2-CARB- OXAMIDE
[0479] Title compound (S)-N-((2H-tetrazol-5-yl)methyl)-1-((S)-2-((2S,3S)-3-methyl-2-(2-phenylac- etamido)pentanamido)propanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-car- boxamide (C14) was prepared tert-butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)carbamate from Example C6 and intermediate I-9 using method A. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.71-0.87 (6H, m), 1.05 (1H, m), 1.28 (3H, d, J=7 Hz), 1.40 (1H, m), 1.63 (1H, m), 2.95 (1H, dd, J=4, 17 Hz), 3.38-3.54 (3H, m), 4.17 (1H, t, J=8 Hz), 4.45 (1H, dd, J=5, 16 Hz), 4.65 (1H, dd, J=6, 16 Hz), 4.98 (1H, dd, J=4, 11 Hz), 5.84 (1H, m), 7.01 (1H, dd, J=5, 7 Hz), 7.16-7.30 (5H, m), 7.64 (1H, d, J=7 Hz), 7.99 (1H, d, J=9 Hz), 8.13 (1H, d, J=6 Hz), 8.24 (1H, d, J=7 Hz), 8.88 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 547.96, expected 548.27 [M+H].
Example C15
(2S)-1-[(2R)-2-[(2S,3S)-3-METHYL-2-(3-CARBOXYPROPANAMIDO)PENTANAMIDO]PROPA- NOYL]-N-(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)-2,3-DIHYDRO-1H-PYRROLO[2,3-B]PYRI- DINE-2-CARBOXAMIDE
[0480] (S)-1-((R)-2-((tert-Butoxycarbonyl)amino)propanoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Boc-D-alanine (3 eq.) using method C in DMF. (LC/MS) m/z observed 335.97, expected 336.16 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0481] tert-Butyl ((R)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)carbamate was prepared from (S)-1-((R)-2-((tert-butoxycarbonyl)amino)propanoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 416.86, expected 417.20 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0482] tert-Butyl ((3S)-1-(((R)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-- 1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)amino)-3-methyl-1-oxopenta- n-2-yl)carbamate was prepared from tert-butyl ((R)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)carbamate and Boc-L-Isoleucine using method A. MS (LC/MS) m/z observed 530.96, expected 530.28 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0483] Title compound (2S)-1-[(2R)-2-[(2S,3 S)-3-methyl-2-(3-carboxypropanamido)pentanamido]propanoyl]-n-(2H-1,2,3,4-- tetrazol-5-ylmethyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxamide (C15) was prepared from tert-butyl ((3S)-1-(((R)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-- 1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxopropan-2-yl)amino)-3-methyl-1-oxopenta- n-2-yl)carbamate and succinic anhydride using method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76-0.85 (6H, m), 1.07 (1H, m), 1.19 (3H, d, J=7 Hz), 1.41 (1H, m), 1.70 (1H, m), 2.30-2.43 (4H, m), 2.96 (1H, dd, J=4, 17 Hz), 3.42 (1H, dd, J=11, 18 Hz), 4.31 (1H, t, J=8 Hz), 4.46 (1H, dd, J=5, 16 Hz), 4.59 (1H, dd, J=6, 16 Hz), 4.93 (1H, dd, J=4, 11 Hz), 6.11 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.63 (1H, d, J=7 Hz), 7.91 (1H, d, J=9 Hz), 8.12-8.19 (2H, m), 8.75 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 530.00, expected 530.25 [M+H].
Example C16
(S)-6-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-5-((2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTANAMID- O)-6-OXOHEXANOIC ACID
[0484] (S)-1-((S)-6-(tert-butoxy)-2-((tert-butoxycarbonyl)amino)-6-oxohexa- noyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared in the same manner as (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid in Example C4, starting from boc-L-.alpha.-aminoadipic acid .delta.-tert-butyl ester (prepared from L-a-aminoadipic acid .delta.-tert-butyl ester hydrochloride via method K). MS (LC/MS) m/z observed 464; expected 464.23 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0485] (S)-tert-Butyl 6-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-((tert-butoxycarbonyl)amino)-6-oxohexanoate was prepared from (S)-1-((S)-6-(tert-butoxy)-2-((tert-butoxycarbonyl)amino)-6-oxohexanoyl)-- 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid via method O but without the second amine addition. MS (LC/MS) m/z observed 545; expected 545.28 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0486] (S)-6-((S)-2-(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-p- yrrolo[2,3-b]pyridin-1-yl)-5-((2 S,3 S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)-6-oxohexanoic acid was prepared from (S)-tert-butyl 6-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-((tert-butoxycarbonyl)amino)-6-oxohexanoate and boc-L-isoleucine via method A. MS (LC/MS) m/z observed 602; expected 602.30 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0487] Title compound (S)-6-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-5-((2S,3 S)-2-(3-carboxypropanamido)-3-methylpentanamido)-6-oxohexanoic acid (C16) was prepared from (S)-6-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-5-((2S,3 S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanamido)-6-oxohexanoic acid via method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.72-0.90 (m, 6H), 1.03-1.15 (m, 1H), 1.33-1.47 (m, 1H), 1.47-1.59 (m, 1H), 1.59-1.74 (m, 3H), 1.77-1.89 (m, 1H), 2.21-2.46 (m, 6H), 2.88-2.99 (m, 1H), 3.43 (dd, J=17.7, 11.2 Hz, 1H), 4.21 (t, J=8.2 Hz, 1H), 4.43 (dd, J=15.8, 5.0 Hz, 1H), 4.64 (dd, J=15.8, 6.0 Hz, 1H), 4.98 (dd, J=11.1, 3.9 Hz, 1H), 5.86 (t, J=7.8 Hz, 1H), 7.01 (dd, J=7.4, 5.1 Hz, 1H), 7.64 (d, J=7.4 Hz, 1H), 7.81 (d, J=8.9 Hz, 1H), 8.03 (d, J=7.7 Hz, 1H), 8.13 (d, J=5.0 Hz, 1H), 8.85 (t, J=5.4 Hz, 1H). MS (LC/MS) m/z observed 602, expected 602.26 [M+H].sup.+.
Example C17
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLBUTANAMIDO)-5-- OXOPENTANOIC ACID
[0488] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-3-methylbutanamido)- -5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) and boc-L-valine via method A but with DMF as the solvent. MS (LC/MS) m/z observed 664, expected 663.31 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0489] 4-(((S)-1-(((S)-1-((S)-2-(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)- amino)-3-methyl-1-oxobutan-2-yl)amino)-4-oxobutanoic acid was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-3-methylbutanamido)- -5-oxopentanoate via method I. MS (LC/MS) m/z observed 664, expected 663.28 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0490] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((S)-2-(3-carboxypropanamido)-3-methylbutanamido)-5- -oxopentanoic acid (C17) was prepared from 4-(((S)-1-(((S)-1-((S)-2-(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydr- o-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)- -3-methyl-1-oxobutan-2-yl)amino)-4-oxobutanoic acid via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82 (d, J=6.8 Hz, 3H), 0.86 (d, J=6.8 Hz, 3H), 1.77-2.07 (m, 3H), 2.27-2.48 (m, 6H), 2.95 (dd, J=17.5, 3.9 Hz, 1H), 3.45 (dd, J=17.4, 11.3 Hz, 1H), 4.19 (dd, J=8.8, 6.9 Hz, 1H), 4.46 (dd, J=15.9, 5.1 Hz, 1H), 4.68 (dd, J=15.9, 6.0 Hz, 1H), 4.98 (dd, J=11.2, 3.9 Hz, 1H), 5.78-5.90 (m, 1H), 7.02 (dd, J=7.3, 5.1 Hz, 1H), 7.65 (d, J=7.4 Hz, 1H), 7.79 (d, J=8.8 Hz, 1H), 8.14 (dd, J=7.6, 3.3 Hz, 2H), 8.94 (t, J=5.6 Hz, 1H). MS (LC/MS) m/z observed 574, expected 573.23 [M+H].sup.+.
Example C18
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((S)-2-CYCLOPENTYL-2-(2-PHENYLACETAMIDO)ACETAMIDO)-5- -OXOPENTANOIC ACID
[0491] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-cyclopentyl-2-(2-phenylacetamido)acetamido)-5-ox- opentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-2-cyclopentylacetam- ido)-5-oxopentanoate (from Example C8) and phenylacetic acid via method A. MS (LC/MS) m/z observed 708, expected 708.32 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0492] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((S)-2-cyclopentyl-2-(2-phenylacetamido)acetamido)-- 5-oxopentanoic acid (C18) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-cyclopentyl-2-(2-phenylacetamido)acetamido)-5-ox- opentanoate via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.10-1.65 (m, 9H), 1.77-1.90 (m, 1H), 1.94-2.18 (m, 2H), 2.29-2.47 (m, 2H), 2.96 (dd, J=17.6, 3.8 Hz, 1H), 3.39-3.56 (m, 3H), 4.21 (t, J=8.6 Hz, 1H), 4.48 (dd, J=15.9, 5.1 Hz, 1H), 4.69 (dd, J=15.9, 6.0 Hz, 1H), 5.00 (dd, J=11.3, 3.9 Hz, 1H), 5.84 (td, J=8.2, 3.7 Hz, 1H), 7.03 (dd, J=7.4, 5.1 Hz, 1H), 7.15-7.35 (m, 5H), 7.66 (dd, J=7.4, 1.7 Hz, 1H), 8.10 (d, J=8.5 Hz, 1H), 8.16 (dd, J=5.2, 1.6 Hz, 1H), 8.22 (d, J=7.5 Hz, 1H), 8.97 (t, J=5.6 Hz, 1H). MS (LC/MS) m/z observed 618, expected 618.27 [M+H].sup.+.
Example C19
(4S)-4-[(2S)-2-ACETAMIDO-3-METHYLBUTANAMIDO]-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-- TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANO- IC ACID
[0493] Title compound (4S)-4-[(2S)-2-acetamido-3-methylbutanamido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4- -tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentan- oic acid (C19) was prepared from C13 via Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82 (d, J=6.8 Hz, 3H), 0.86 (d, J=6.8 Hz, 3H), 1.85 (s, 3H), 1.88-2.07 (m, 1H), 2.39 (td, J=10.9, 5.2 Hz, 1H), 2.90-3.01 (m, 1H), 3.39-3.49 (m, 2H), 4.16 (t, J=7.9 Hz, 1H), 4.43 (dd, J=15.6, 4.9 Hz, 1H), 4.65 (dd, J=15.8, 5.9 Hz, 1H), 4.98 (dd, J=11.3, 3.9 Hz, 1H), 5.82 (d, J=9.6 Hz, 1H), 7.02 (dd, J=7.4, 5.1 Hz, 1H), 7.65 (d, J=7.3 Hz, 1H), 7.81 (d, J=8.8 Hz, 1H), 8.11-8.19 (m, 2H), 8.87 (t, J=5.6 Hz, 1H). MS (LC/MS) m/z observed 516, expected 516.22 [M+H].sup.+.
Example C20
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((S)-2-AMINO-3-METHYLBUTANAMIDO)-5-OXOPENTANOIC ACID HYDROCHLORIDE
[0494] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((S)-2-amino-3-methylbutanamido)-5-oxopentanoic acid hydrochloride (C20) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-3-methylbutanamido)- -5-oxopentanoate (from Example C17) via Method Q immediately followed by method E. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.92 (t, J=5.9 Hz, 6H), 1.80-2.15 (m, 3H), 2.44 (t, J=8.3 Hz, 2H), 2.94 (dd, J=17.5, 3.9 Hz, 1H), 3.37 (dd, J=17.3, 11.1 Hz, 1H), 3.56 (d, J=5.8 Hz, 1H), 4.27 (dd, J=14.9, 4.5 Hz, 1H), 4.45 (dd, J=14.9, 5.7 Hz, 1H), 5.00 (dd, J=11.2, 4.0 Hz, 1H), 5.87-6.00 (m, 1H), 7.03 (dd, J=7.4, 5.1 Hz, 1H), 7.63 (d, J=7.3 Hz, 1H), 8.16 (d, J=5.0 Hz, 1H), 8.46 (t, J=5.3 Hz, 1H), 8.64 (d, J=7.6 Hz, 1H). MS (LC/MS) m/z observed 510, expected 509.19 [M+H].sup.+.
Example C21
(R)-5-((S)-2(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[2- ,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTANAMIDO- )-5-OXOPENTANOIC ACID
[0495] (S)-1-((R)-5-(Benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentan- oyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared in the same manner as (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid in Example C4, starting from Boc-D-glutamic acid y-benzyl ester (365 mg). MS (LC/MS) m/z observed 484, expected 484.20 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0496] (R)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate was prepared from (S)-1-((R)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid via method O but without the second amine addition. MS (LC/MS) m/z observed 565, expected 565.24 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0497] (R)-Benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate was prepared from (R)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate via method A but without swapping with MeOH; and DMF as the solvent for the coupling step described therein. MS (LC/MS) m/z observed 678; expected 678.33 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0498] Title compound (R)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpentanamid- o)-5-oxopentanoic acid (C21) was prepared in the same manner as (S)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpentanamid- o)-5-oxopentanoic acid (from Example C4) starting from (R)-5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[- 2,3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpent- anamido)-5-oxopentanoate benzyl ester. C21 exists as a mixture of rotamers in a 4:1 ratio; only the major signals are reported. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.73-0.88 (m, 6H), 1.00-1.15 (m, 1H), 1.42 (d, J=13.7 Hz, 1H), 1.63-1.93 (m, 2H), 2.13-2.47 (m, 6H), 2.95 (dd, J=17.6, 3.4 Hz, 1H), 3.21-3.51 (m, 7H), 4.32-4.40 (m, 1H), 4.45 (dd, J=15.5, 5.3 Hz, 1H), 4.59 (dd, J=15.6, 5.8 Hz, 1H), 4.91 (d, J=10.5 Hz, 1H), 6.16 (s, 1H), 6.97-7.10 (m, 1H), 7.66 (d, J=7.3 Hz, 1H), 7.90 (d, J=9.1 Hz, 1H), 8.19 (dd, J=10.4, 6.5 Hz, 2H), 8.77 (t, J=5.6 Hz, 1H). MS (LC/MS) m/z observed 588, expected 588.25 [M+H].sup.+.
Example C22
4-(((S)-1-(((S)-1-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO- -1H-PYRROLO[2,3-B]PYRIDIN-1-YL)-5-AMINO-1,5-DIOXOPENTAN-2-YL)AMINO)-3-METH- YL-1-OXOBUTAN-2-YL)AMINO)-4-OXOBUTANOIC ACID
[0499] (S)-1-((S)-5-Amino-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared in the same manner as (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid in Example C4, starting from boc-L-glutamine. MS (LC/MS) m/z observed 393; expected 393.17 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0500] tert-Butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-5-amino-1,5-dioxopentan-2-yl)carbamate was prepared from (S)-1-((S)-5-amino-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2,- 3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid via method O but without the second amine addition. MS (LC/MS) m/z observed 474; expected 474.21 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0501] tert-Butyl ((S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1- H-pyrrolo[2,3-b]pyridin-1-yl)-5-amino-1,5-dioxopentan-2-yl)amino)-3-methyl- -1-oxobutan-2-yl)carbamate was prepared from tert-butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-5-amino-1,5-dioxopentan-2-yl)carbamate and boc-L-valine via method A. MS (LC/MS) m/z observed 573; expected 573.28 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0502] Title compound 4-(((S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydr- o-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-amino-1,5-dioxopentan-2-yl)amino)-3-met- hyl-1-oxobutan-2-yl)amino)-4-oxobutanoic acid (C22) was prepared from tert-butyl ((S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1- H-pyrrolo[2,3-b]pyridin-1-yl)-5-amino-1,5-dioxopentan-2-yl)amino)-3-methyl- -1-oxobutan-2-yl)carbamate via method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.82 (d, J=6.9 Hz, 3H), 0.85 (d, J=6.8 Hz, 3H), 1.71-1.86 (m, 1H), 1.87-2.05 (m, 2H), 2.13-2.44 (m, 6H), 2.94 (dd, J=17.7, 3.9 Hz, 1H), 3.38-3.51 (m, 1H), 3.60-3.85 (m, 4H), 4.02-4.33 (m, 3H), 4.47 (dd, J=15.8, 5.2 Hz, 1H), 4.67 (dd, J=15.9, 5.9 Hz, 1H), 4.98 (dd, J=11.3, 3.9 Hz, 1H), 5.74-5.87 (m, 1H), 6.75 (s, 1H), 7.02 (dd, J=7.3, 5.1 Hz, 1H), 7.24 (s, 1H), 7.65 (d, J=7.3 Hz, 1H), 7.78 (d, J=8.9 Hz, 1H), 8.10-8.23 (m, 2H), 8.93 (t, J=5.8 Hz, 1H). MS (LC/MS) m/z observed 573, expected 573.25 [M+H].sup.+.
Example C23
(S)-METHYL 5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-P- YRROLO[2,3-B]PYRIDIN-1-YL)-4-((S)-3-METHYL-2-(2-PHENYLACETAMIDO)BUTANAMIDO- )-5-OXOPENTANOATE
[0503] (S)-Methyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-amino-3-methylbutanamido)-5-oxopentanoate hydrochloride was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-((tert-butoxycarbonyl)amino)-3-methylbutanamido)- -5-oxopentanoate (from Example C17) via method E but with MeOH as a co-solvent in a 1:1 (v/v) ratio. MS (LC/MS) m/z observed 488, expected 487.23 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0504] Title compound (S)-methyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-3-methyl-2-(2-phenylacetamido)butanamido)-5-oxopen- tanoate (C23) was prepared from (S)-Methyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-amino-3-methylbutanamido)-5-oxopentanoate hydrochloride and phenylacetic acid via method M but with DMF as the solvent. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.80 (d, J=6.9 Hz, 3H), 0.83 (d, J=6.6 Hz, 3H), 1.56-1.66 (m, 1H), 1.83-2.07 (m, 4H), 2.86-3.08 (m, 3H), 3.36-3.45 (m, 2H), 3.57 (s, 3H), 4.19 (dd, J=8.9, 6.9 Hz, 1H), 4.29 (dd, J=15.2, 4.3 Hz, 1H), 4.51 (dd, J=15.0, 5.9 Hz, 1H), 5.01 (dd, J=11.2, 3.9 Hz, 1H), 5.81-5.89 (m, 1H), 7.01 (dd, J=7.4, 5.0 Hz, 1H), 7.16-7.23 (m, 1H), 7.23-7.33 (m, 4H), 7.65 (d, J=7.3 Hz, 1H), 7.99 (d, J=8.9 Hz, 1H), 8.14 (d, J=5.1 Hz, 1H), 8.23 (d, J=7.6 Hz, 1H), 8.54 (t, J=5.4 Hz, 1H). MS (LC/MS) m/z observed 606, expected 606.27 [M+H].sup.+.
Example C24
METHYL (4S)-4-[(2S)-2-ACETAMIDO-3-METHYLBUTANAMIDO]-5-OXO-5-[(2S)-2-[(2H-1- ,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]- PENTANOATE
[0505] Title compound methyl (4S)-4-[(2S)-2-acetamido-3-methylbutanamido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4- -tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentan- oate (C24) was prepared from (S)-methyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((S)-2-amino-3-methylbutanamido)-5-oxopentanoate hydrochloride (from Example C23) and acetic acid via method M but with DMF as a solvent. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.81 (d, J=6.8 Hz, 3H), 0.84 (d, J=6.8 Hz, 3H), 1.83 (s, 3H), 1.89 (dd, J=13.5, 7.4 Hz, 2H), 1.96-2.06 (m, 1H), 2.93 (d, J=16.4 Hz, 1H), 3.37-3.46 (m, 1H), 3.56 (s, 3H), 4.13 (t, J=7.9 Hz, 1H), 4.40 (dd, J=15.5, 4.9 Hz, 1H), 4.61 (dd, J=15.6, 6.0 Hz, 1H), 4.97 (dd, J=11.2, 4.0 Hz, 1H), 5.79-5.88 (m, 1H), 7.00 (dd, J=7.3, 5.1 Hz, 1H), 7.64 (d, J=7.3 Hz, 1H), 7.79 (d, J=8.7 Hz, 1H), 8.13 (d, J=5.5 Hz, 1H), 8.16 (d, J=7.5 Hz, 1H), 8.81 (s, 1H). MS (LC/MS) m/z observed 530, expected 530.24 [M+H]+.
Example C25
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-(3-METHYLBUTANAMIDO)-5-OXOPENTANOIC ACID
[0506] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(3-methylbutanamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) and and isovaleric acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 549.98, expected 549.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0507] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-(3-methylbutanamido)-5-oxopentanoic acid (C25) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(3-methylbutanamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.83-0.89 (6H, d, J=7 Hz), 1.77 (1H, m), 1.88-2.05 (4H, m), 2.30-2.45 (2H, m), 2.95 (1H, d, J=16 Hz), 3.45 (1H, dd, J=11, 17 Hz), 4.45 (1H, dd, J=5, 16 Hz), 4.65 (1H, dd, J=6, 16 Hz), 4.98 (1H, dd, J=4, 11 Hz), 5.83 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.66 (1H, d, J=7 Hz), 8.05 (1H, d, J=8 Hz), 8.17 (1H, d, J=4 Hz), 8.93 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 458.83, expected 459.21 [M+H].
Example C26
4-(((S)-1-(((S)-1-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO- -1H-PYRROLO[2,3-B]PYRIDIN-1-YL)-4-AMINO-1,4-DIOXOBUTAN-2-YL)AMINO)-3-METHY- L-1-OXOBUTAN-2-YL)AMINO)-4-OXOBUTANOIC ACID
[0508] (S)-1-((S)-2-((tert-Butoxycarbonyl)amino)-4-oxo-4-(tritylamino)buta- noyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Na-Boc-N.sup..gamma.-trityl-L-asparagine (3 eq.) using method C in DMF. MS (LC/MS) m/z observed 620.77, expected 621.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0509] tert-Butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1,4-dioxo-4-(tritylamino)butan-2-yl)carbamate was prepared from (S)-1-((S)-2-((tert-butoxycarbonyl)amino)-4-oxo-4-(tritylamino)butanoyl)-- 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 701.77, expected 702.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0510] tert-Butyl ((S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1- H-pyrrolo[2,3-b]pyridin-1-yl)-1,4-dioxo-4-(tritylamino)butan-2-yl)amino)-3- -methyl-1-oxobutan-2-yl)carbamate was prepared from tert-butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1,4-dioxo-4-(tritylamino)butan-2-yl)carbamate and Boc-L-valine using method A but without swapping with MeOH. DMF was used as the solvent of the coupling step. MS (LC/MS) m/z observed 800.79, expected 801.38 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0511] tert-Butyl ((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-1,4-dioxo-4-(tritylamino)butan-2-yl)carbamate (217 mg, 0.271 mmol, 1 eq.) was treated with HCl in dioxane (20 mL) for 1 h at rt. The solvent was then concentrated to dryness. The residue and succinic anhydride (41 mg, 0.406 mmol, 1.5 eq.) were suspended in dry DCM (10 mL) under N.sub.2 and DIPEA (0.151 mL, 1.08 mmol, 4 eq.) was added to the mixture. The reaction mixture was left at rt for 2 hrs and trifluoroacetic acid (30 mL) was added. The reaction was left at rt for 1 h until full deprotection of the trityl group. The solvent were then evaporated and the product was purified by preparative HPLC using a gradient from 20% to 32% of methanol in water in 10 minutes to give the title compound 4-(((S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydr- o-1H-pyrrolo[2,3-b]pyridin-1-yl)-4-amino-1,4-dioxobutan-2-yl)amino)-3-meth- yl-1-oxobutan-2-yl)amino)-4-oxobutanoic acid (C26) as a white solid (56 mg, 37%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.81 (3H, t, J=7 Hz), 0.84 (3H, d, J=7 Hz), 1.92 (1H, m), 2.32-2.50 (5H, m), 2.66 (1H, dd, J=5, 15 Hz), 3.01 (1H, dd, J=4, 17 Hz), 3.48 (1H, dd, J=11, 18 Hz), 4.22 (1H, dd, J=7, 9 Hz), 4.51 (1H, dd, J=5, 16 Hz), 4.62 (1H, dd, J=6, 16 Hz), 4.97 (1H, dd, J=4, 11 Hz), 6.10 (1H, m), 6.97 (1H, s), 7.03 (1H, dd, J=5, 7 Hz), 7.31 (1H, s), 7.66 (1H, d, J=7 Hz), 7.77 (1H, d, J=9 Hz), 8.12 (1H, d, 5 Hz), 8.25 (1H, d, J=7 Hz), 8.97 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 558.98, expected 559.24 [M+H].
Example C27
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-(2-CYCLOPENTYLACETAMIDO)-5-OXOPENTANOIC ACID
[0512] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclopentylacetamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-- b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentanam- ido)-5-oxopentanoate (from Example C4) and cyclopentylacetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 574.92, expected 575.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0513] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-(2-cyclopentylacetamido)-5-oxopentanoic acid (C27) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclopentylacetamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.06-1.18 (2H, m), 1.41-1.50 (2H, m), 1.52-1.58 (22H, m), 1.62-1.70 (2H, m), 1.80 (1H, m), 1.96-2.14 (4H, m), 2.30-2.47 (2H, m), 2.95 (1H, dd, J=4, 17 Hz), 3.45 (1H, dd, J=l 1, 17 Hz), 4.47 (1H, dd, J=5, 16 Hz), 4.67 (1H, dd, J=6, 16 Hz), 4.98 (1H, dd, J=4, 11 Hz), 5.83 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.66 (1H, d, J=7 Hz), 8.03 (1H, d, J=8 Hz), 8.17 (1H, d, J=4 Hz), 8.95 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 484.89, expected 485.23 [M+H].
Example C28
(S)-5-((S)-2-((2-(2H-TETRAZOL-5-YL)ETHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO- [2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-2-(3-CARBOXYPROPANAMIDO)-3-METHYLPENTANAMI- DO)-5-OXOPENTANOIC ACID
[0514] (S)-Benzyl 5-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,- 3-b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate was prepared from (S)-1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2- ,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid (from Example C4) and (2H-tetrazol-5-yl)ethyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 578.91, expected 579.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0515] (S)-Benzyl 5-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,- 3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentan- amido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,- 3-b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate and Boc-L-Isoleucine using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 691.95, expected 692.35 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0516] 4-(((2S,3S)-1-(((S)-1-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)- -2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-- 2-yl)amino)-3-methyl-1-oxopentan-2-yl)amino)-4-oxobutanoic acid was prepared from (S)-benzyl 5-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,- 3-b]pyridin-1-yl)-4-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentan- amido)-5-oxopentanoate and succinic anhydride using method I. MS (LC/MS) m/z observed 691.97, expected 692.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0517] Title compound (S)-5-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-((2S,3S)-2-(3-carboxypropanamido)-3-methylpentanam- ido)-5-oxopentanoic acid (C28) was prepared from 4-(((2S,3S)-1-(((S)-1-((S)-2-((2-(2H-tetrazol-5-yl)ethyl)carbamoyl)-2,3-d- ihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)a- mino)-3-methyl-1-oxopentan-2-yl)amino)-4-oxobutanoic acid using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76-0.85 (6H, m), 1.08 (1H, m), 1.40 (1H, m), 1.65 (1H, m), 1.85 (1H, m), 2.00 (1H, m), 2.25-2.48 (6H, m), 2.77 (1H, dd, J=4, 17 Hz), 2.95-3.10 (2H, m), 3.28-3.43 (2H, m), 3.56 (1H, m), 4.20 (1H, m), 4.87 (1H, dd, J=4, 11 Hz), 5.85 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.63 (1H, d, J=7 Hz), 7.82 (1H, d, J=9 Hz), 8.12-8.18 (2H, m), 8.36 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 601.96, expected 602.27 [M+H].
Example C29
3-{[(1S,2S)-2-METHYL-1-{[(2S)-1-OXO-3-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CAR- BAMOYL]-1-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYR- ROLO[2,3-B]PYRIDIN-1-YL]PROPAN-2-YL]CARBAMOYL}BUTYL]CARBAMOYL}PROPANOIC ACID
[0518] (S)-1-((S)-4-(Benzyloxy)-2-((tert-butoxycarbonyl)amino)-4-oxobutano- yl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Boc-L-aspartic acid .beta.-benzyl ester (3 eq.) using method C in DMF. MS (LC/MS) m/z observed 469.90, expected 470.19 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0519] (S)-Benzyl 4-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-3-((tert-butoxycarbonyl)amino)-4-oxobutanoate was prepared from (S)-1-((S)-4-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-4-oxobutanoyl)-2,- 3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 550.87, expected 551.24 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0520] (S)-Benzyl 4-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-3-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentana- mido)-4-oxobutanoate was prepared from (S)-benzyl 4-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-3-((tert-butoxycarbonyl)amino)-4-oxobutanoate and Boc-L-Isoleucine using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 663.87, expected 664.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0521] (S)-4-((S)-2-(((2H-Tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-p- yrrolo[2,3-b]pyridin-1-yl)-3-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-met- hylpentanamido)-4-oxobutanoic acid was prepared from (S)-benzyl 4-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-3-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpentana- mido)-4-oxobutanoate using Method Q. MS (LC/MS) m/z observed 573.89, expected 574.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0522] tert-Butyl ((2S,3S)-1-(((S)-4-(((2H-tetrazol-5-yl)methyl)amino)-1-((S)-2-(((2H-tetra- zol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1,4-- dioxobutan-2-yl)amino)-3-methyl-1-oxopentan-2-yl)carbamate was prepared from (S)-4-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-py- rrolo[2,3-b]pyridin-1-yl)-3-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-meth- ylpentanamido)-4-oxobutanoic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 654.83, expected 655.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0523] Title compound 3-{[(1S,2S)-2-methyl-1-{[(2S)-1-oxo-3-[(2H-1,2,3,4-tetrazol-5-ylmethyl)ca- rbamoyl]-1-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H-py- rrolo[2,3-b]pyridin-1-yl]propan-2-yl]carbamoyl}butyl]carbamoyl}propanoic acid (C29) was prepared from tert-butyl ((2S,3S)-1-(((S)-4-(((2H-tetrazol-5-yl)methyl)amino)-1-((S)-2-(((2H-tetra- zol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1,4-- dioxobutan-2-yl)amino)-3-methyl-1-oxopentan-2-yl)carbamate and succinic anhydride using method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.70-0.84 (6H, m), 1.04 (1H, m), 1.36 (1H, m), 1.62 (1H, m), 2.26-2.47 (4H, m), 2.58 (1H, dd, J=9, 15 Hz), 2.77 (1H, dd, J=5, 16 Hz), 3.00 (1H, dd, J=4, 17 Hz), 3.45 (1H, dd, J=11, 17 Hz), 4.16 (1H, t, J=8 Hz), 4.45-4.65 (4H, m), 4.96 (1H, dd, J=4, 11 Hz), 6.11 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.63 (1H, d, J=7 Hz), 7.82 (1H, d, J=9 Hz), 8.00 (1H, d, J=4 Hz), 8.22 (1H, d, J=7 Hz), 8.58 (1H, t, J=6 Hz), 8.93 (1H, t, J=6 Hz), MS (LC/MS) m/z observed 654.95, expected 655.28 [M+H].
Example C30
(S)-5-((S)-2-(((2H-TETRAZOL-5-YL)METHYL)CARBAMOYL)-2,3-DIHYDRO-1H-PYRROLO[- 2,3-B]PYRIDIN-1-YL)-4-((2S,3S)-3-METHYL-2-(PYRIMIDIN-2-YLAMINO)PENTANAMIDO- )-5-OXOPENTANOIC ACID
[0524] I-7 (200 mg, 0.719 mmol) was dissolved in a mixture of allyl alcohol and HCl in dioaxane (4M) (20 mL, 1:1 (v/v)) and the reaction mixture was stirred at rt for 3 hours. The reaction mixture was then concentrated to dryness and swapped with allyl alcohol (2.times.25 mL). The solid obtained was dried well under reduced pressure to give (S)-allyl 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate hydrochloride as a white solid (173 mg, quantitative). .sup.1H NMR (400 MHz, DMSO-d6) 3.23 (1H, dd, J=5, 18 Hz), 3.55 (1H, dd, J=11, 18 Hz), 4.65 (2H, d, J=5 Hz), 4.86 (1H, dd, J=5, 11 Hz), 5.25 (1H, d, J=10 Hz), 5.36 (1H, d, J=17 Hz), 5.94 (1H, m), 6.80 (1H, t, J=7 Hz), 7.68-7.74 (2H, m), 9.29 (1H, bs), MS (LC/MS) m/z observed 204.98, expected 205.10 [M+H].
[0525] (S)-Allyl 1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2,3-d- ihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate was prepared from (S)-allyl 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate hydrochloride and Boc-L-glutamic acid y-benzyl ester (1.2 eq.) using method C in DMF. MS (LC/MS) m/z observed 523.95, expected 524.24 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0526] (S)-Allyl 1-((S)-5-(benzyloxy)-2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpe- ntanamido)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxy- late was prepared from (S)-allyl 1-((S)-5-(benzyloxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)-2,3-d- ihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate and Boc-L-Isoleucine using method A but without swapping with MeOH. MS (LC/MS) m/z observed 636.97, expected 637.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0527] (S)-Allyl 1-((S)-5-(benzyloxy)-2-((2S,3S)-2-((tert-butoxycarbonyl)amino)-3-methylpe- ntanamido)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxy- late (218 mg, 0.342 mmol, 1 eq.) and 2-bromopyrimidine (136 mg, 0.855 mmol, 2.5 eq.) were added in a microwave vial. DMF (8 mL) and DIPEA (0.174 mL, 1.710 mmol, 5 eq.) were then added. The reaction mixture was irradiated (microwave) at 145.degree. C. for 4 hours. The solvent was then evaporated and the product was purified by column chromatography using 15% to 80% ethyl acetate in hexanes as the eluent to give (S)-allyl 1-((S)-5-(benzyloxy)-2-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanami- do)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate as an orange glass (40 mg, 19%). MS (LC/MS) m/z observed 614.98, expected 615.29 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0528] (S)-Allyl 1-((S)-5-(benzyloxy)-2-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanami- do)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate (40 mg, 0.0651 mmol, 1 equiv) and Pd(PPh.sub.3).sub.4(15 mg, 0.0130, 0.2 equiv) were dissolved in CH.sub.2Cl.sub.2 (10 mL) under N.sub.2. Morpholine (0.017 mL, 0.195 mmol, 3 equiv) was then added and the reaction was left at rt for 1 h. The solvent was then evaporated and the product was purified by column chromatography reverse phase using 10% to 50% methanol in water as the eluent to give (S)-1-((S)-5-(benzyloxy)-2-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)penta- namido)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid as a colorless glass 37 mg, quantitative). MS (LC/MS) m/z observed 574.94, expected 575.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0529] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanamido)-5- -oxopentanoate was prepared from (S)-allyl 1-((S)-5-(benzyloxy)-2-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanami- do)-5-oxopentanoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 655.98, expected 656.31 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0530] Title compound (S)-5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo- [2,3-b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanamid- o)-5-oxopentanoic acid (C30) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((2S,3S)-3-methyl-2-(pyrimidin-2-ylamino)pentanamido)-5- -oxopentanoate using Method Q. MS (LC/MS) m/z observed 565.95, expected 566.26 [M+H].
Example C31
(4S)-4-(2-CYCLOHEXYLACETAMIDO)-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLM- ETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0531] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclohexylacetamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and 2-cyclohexylacetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 588.97, expected 589.29 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0532] Title compound (4S)-4-(2-cyclohexylacetamido)-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-yl- methyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C31) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclohexylacetamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.78-0.92 (2H, m), 1.02-1.22 (3H, m), 1.52-1.65 (6H, m), 1.75 (1H, m), 1.93-2.02 (3H, m), 2.25-2.45 (2H, m), 2.93 (1H, d, J=18 Hz), 3.43 (1H, m), 4.47 (1H, d, J=16 Hz), 4.67 (1H, d, J=16 Hz), 4.95 (1H, d, J=11 Hz), 5.81 (1H, s), 7.00 (1H, s), 7.62 (1H, d, J=7 Hz), 8.03 (1H, d, J=8 Hz), 8.15 (1H, s), 8.92 (1H, s), MS (LC/MS) m/z observed 498.96, expected 498.23 [M+H].
Example C32
(4S)-5-OXO-4-(2-PHENYLACETAMIDO)-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHY- L)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0533] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-(2-phenylacetamido)pentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and 2-phenylacetic acid acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 582.92, expected 583.24 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0534] Title compound (4S)-5-oxo-4-(2-phenylacetamido)-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylmeth- yl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C32) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-(2-phenylacetamido)pentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.82 (1H, m), 2.05 (1H, m), 2.25-2.45 (2H, m), 2.95 (1H, d, J=17 Hz), 3.25-3.50 (3H, m), 4.47 (1H, d, J=16 Hz), 4.67 (1H, d, J=16 Hz), 4.98 (1H, d, J=11 Hz), 5.87 (1H, m), 7.03 (1H, m), 7.17-7.35 (5H, m), 7.66 (1H, d, J=7 Hz), 8.15 (1H, s), 8.40 (1H, d, J=8 Hz), 8.96 (1H, s), MS (LC/MS) m/z observed 492.92, expected 493.19 [M+H].
Example C33
(4S)-5-OXO-4-[(2R)-2-PHENYLPROPANAMIDO]-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-- YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0535] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-((R)-2-phenylpropanamido)pentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and (R)-2-phenylpropanoic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 596.93, expected 597.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0536] Title compound (4S)-5-oxo-4-[(2R)-2-phenylpropanamido]-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5- -ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-((R)-2-phenylpropanamido)pentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.28 (3H, d, J=8 Hz), 1.78 (1H, m), 1.99 (1H, m), 2.18 (1H, m), 2.33 (1H, m), 2.95 (1H, d, J=17 Hz), 3.45 (1H, m), 3.73 (1H, m), 4.47 (1H, d, J=16 Hz), 4.67 (1H, dd, J=8, 16 Hz), 5.00 (1H, d, J=11 Hz), 5.77 (1H, m), 7.03 (1H, m), 7.20 (1H, m), 7.25-7.36 (4H, m), 7.66 (1H, d, J=7 Hz), 8.12 (1H, s), 8.28 (1H, d, J=8 Hz), 8.96 (1H, s), MS (LC/MS) m/z observed 506.94, expected 507.21 [M+H].
Example C34
(4S)-5-OXO-4-[(2S)-2-PHENYLPROPANAMIDO]-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-- YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0537] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-((S)-2-phenylpropanamido)pentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and (S)-2-phenylpropanoic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 596.94, expected 597.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0538] Title compound (4S)-5-oxo-4-[(2S)-2-phenylpropanamido]-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5- -ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C34) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-5-oxo-4-((S)-2-phenylpropanamido)pentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.32 (3H, d, J=8 Hz), 1.80 (1H, m), 2.01 (1H, m), 2.30-2.48 (2H, m), 2.95 (1H, d, J=17 Hz), 3.43 (1H, m), 3.75 (1H, m), 4.47 (1H, d, J=16 Hz), 4.67 (1H, m), 4.95 (1H, dd, J=11.18 Hz), 5.90 (1H, m), 7.03 (1H, m), 7.18 (1H, m), 7.25-7.35 (4H, m), 7.66 (1H, d, J=7 Hz), 8.12 (1H, m), 8.28 (1H, d, J=8 Hz), 8.96 (1H, s), MS (LC/MS) m/z observed 506.94, expected 507.21 [M+H].
Example C35
(4S)-4-(2-CYCLOBUTYLACETAMIDO)-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLM- ETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0539] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclobutylacetamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and cyclobutylacetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 561.00, expected 561.26 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0540] Title compound (4S)-4-(2-cyclobutylacetamido)-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-yl- methyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C35) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclobutylacetamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.60-1.71 (2H, m), 1.72-1.82 (3H, m), 1.93-2.04 (3H, m), 2.17-2.25 (2H, m), 2.31-2.43 (2H, m), 2.53 (1H, m), 2.95 (1H, d, J=18 Hz), 3.45 (1H, dd, J=11, 18 Hz), 4.46 (1H, dd, J=5, 16 Hz), 4.66 (1H, dd, J=6, 16 Hz), 4.98 (1H, dd, J=4, 11 Hz), 5.83 (1H, m), 7.02 (1H, dd, J=5, 7 Hz), 7.65 (1H, d, J=7 Hz), 8.01 (1H, d, J=8 Hz), 8.17 (1H, d, J=5 Hz), 8.92 (1H, m), MS (LC/MS) m/z observed 470.98, expected 471.21 [M+H].
Example C36
(4S)-4-(2-CYCLOPROPYLACETAMIDO)-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YL- METHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0541] (S)-Benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclopropylacetamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and 2-cyclopropylacetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 546.98, expected 547.24 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0542] Title compound (4S)-4-(2-cyclopropylacetamido)-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-y- lmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C36) was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-(2-cyclopropylacetamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.08-0.15 (2H, m), 0.37-0.44 (2H, d, J=9 Hz), 0.95 (1H, m), 1.80 (1H, m), 1.96-2.06 (3H, m), 2.30-2.45 (2H, m), 2.96 (1H, d, J=18 Hz), 3.43 (1H, dd, J=11, 18 Hz), 4.47 (1H, dd, J=5, 16 Hz), 4.67 (1H, dd, J=6, 16 Hz), 5.00 (1H, dd, J=4, 11 Hz), 5.88 (1H, m), 7.02 (1H, dd, J=5, 7 Hz), 7.65 (1H, d, J=7 Hz), 7.99 (1H, d, J=8 Hz), 8.17 (1H, d, J=5 Hz), 8.92 (1H, m), MS (LC/MS) m/z observed 456.92, expected 457.19 [M+H].
Example C37
(4S)-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-- PYRROLO[2,3-B]PYRIDIN-1-YL]-4-[2-(THIOPHEN-3-YL)ACETAMIDO]PENTANOIC ACID
[0543] (S)-1-((S)-5-(tert-Butoxy)-2-((tert-butoxycarbonyl)amino)-5-oxopent- anoyl)-2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid was prepared from I-7 and Boc-L-glutamic acid tert-butyl ester (3 eq.) using method C in DMF. MS (LC/MS) m/z observed 449.98, expected 450.22 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0544] (S)-tert-Butyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate was prepared from (S)-1-((S)-5-(tert-butoxy)-2-((tert-butoxycarbonyl)amino)-5-oxopentanoyl)- -2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 530.96, expected 531.27 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0545] (S)-tert-Butyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (49.7 mg, 0.0880 mmol) was dissolved in 4M HCl in dioxane (10 mL) and the reaction was heated to 50.degree. C. for 3 hours. Both tert-butyl ester and Boc group were removed. The solvent was evaporated to give a residue as a colorless glass. In a separated flask were dissolved 2-(thiophen-3-yl)acetic acid (125.1 mg, 0.880 mmol, 10 eq.), HOBt (16.1 mg, 0.106 mmol, 1.2 eq.) and EDC (30.2 mg, 0.106 mmol, 1.2 eq.) in DMF (5 mL). DIPEA was then added (0.230 mL, 1.320 mmol, 15 eq.) and the mixture was stirred at RT for 10 minutes. A solution in DMF (2 mL) of the residue obtained previously was slowly added to the mixture containing the 2-(thiophen-3-yl)acetic acid and the reaction was left at RT for 10 minutes. The solvent was then evaporated and the product was purified by first a preparative reverse phase HPLC purification using a 10 minutes gradient from 40% to 52% methanol in water. The excess 2-(thiophen-3-yl)acetic acid and the desired product co-eluted on this column. The product was then repurified by normal phase chromatography using 5% methanol in DCM as the eluent to remove the excess acid and then 10% methanol, 1% HCOOH and 89% DCM to elute desired the title compound (4S)-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H- -pyrrolo[2,3-b]pyridin-1-yl]-4-[2-(thiophen-3-yl)acetamido]pentanoic acid (C37) that was obtained as a white solid (12.5 mg, 27%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.82 (1H, m), 2.03 (1H, m), 2.30-2.45 (2H, m), 2.48-2.53 (2H, m), 2.96 (1H, d, J=18 Hz), 3.43 (1H, dd, J=11, 18 Hz), 4.47 (1H, dd, J=5, 16 Hz), 4.67 (1H, dd, J=6, 16 Hz), 5.00 (1H, dd, J=4, 11 Hz), 5.88 (1H, m), 6.98-7.04 (2H, m), 7.23 (1H, s), 7.43 (1H, m), 7.65 (1H, d, J=7 Hz), 8.14 (1H, d, J=5 Hz), 8.35 (1H, d, J=8 Hz), 8.92 (1H, m), MS (LC/MS) m/z observed 498.96, expected 499.15 [M+H].
Example C38
(4S)-4-[2-(MORPHOLIN-2-YL)ACETAMIDO]-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL- -5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0546] (9H-Fluoren-9-yl)methyl 2-(2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-2-ox- oethyl)morpholine-4-carboxylate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and 2-(4-(((9H-fluoren-9-yl)methoxy)carbonyl)morpholin-2-yl)acetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 813.99, expected 814.33 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0547] (9H-Fluoren-9-yl)methyl 2-(2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-2-ox- oethyl)morpholine-4-carboxylate (41.3 mg, 0.0516 mmol) was dissolved in a 1:1 mixture methanol/DCM (20 mL) and palladium on charcoal 10% by wt (10 mg) was added to the solution under N.sub.2. The flask was then flushed with H.sub.2 and H.sub.2 was bubbled into the reaction mixture for 4 hrs. The flask was flushed with N.sub.2 and the reaction mixture was filtered over CELITE.TM.. The solids were washed with methanol (3.times.10 mL) and CH.sub.2Cl.sub.2 (3.times.10 mL) and the filtrate and washings were then concentrated to give a light brown solid that was dissolved in DMF (5 mL) and morpholine (5 mL). The reaction was left at RT for 1 h and the solvent was evaporated. The product was then purified by reverse phase preparative HPLC using a 10 minutes gradient from 0% to 15% methanol in water (containing 0.1% HCOOH) to give title compound (4S)-4-[2-(morpholin-2-yl)acetamido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazo- l-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid as a light brown solid (15.5 mg, 56%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.80 (1H, m), 2.02 (1H, m), 2.27-2.45 (4H, m), 2.76 (1H, m), 2.87-3.02 (2H, m), 3.06-3.25 (2H, m), 3.45 (1H, dd, J=11, 18 Hz), 3.66 (1H, m), 3.90 (1H, m), 4.02 (1H, m), 4.47 (1H, dd, J=5, 16 Hz), 4.67 (1H, dd, J=6, 16 Hz), 5.00 (1H, m), 5.88 (1H, m), 7.04 (1H, m), 7.65 (1H, d, J=7 Hz), 8.14 (1H, d, J=5 Hz), 8.35 (1H, m), 9.01 (1H, m), 9.23-9.37 (2H, m), MS (LC/MS) m/z observed 502.01, expected 502.22 [M+H].
Example C39
(4S)-4-[2-(MORPHOLIN-3-YL)ACETAMIDO]-5-OXO-5-[(2S)-2-[(2H-1,2,3,4-TETRAZOL- -5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0548] tert-Butyl 3-(2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-2-ox- oethyl)morpholine-4-carboxylate was prepared from (S)-benzyl 5-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2,3- -b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C4) and 2-(4-(tert-butoxycarbonyl)morpholin-3-yl)acetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 691.89, expected 692.32 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0549] tert-Butyl 3-(2-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-- pyrrolo[2,3-b]pyridin-1-yl)-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-2-ox- oethyl)morpholine-4-carboxylate (41.3 mg, 0.0597 mmol) was dissolved in methanol (10 mL) and palladium on charcoal 10% by wt (10 mg) was added to the solution under N.sub.2. The flask was then flushed with H.sub.2 and H.sub.2 was bubbled into the reaction mixture for 4 hrs. The flask was flushed with N.sub.2 and the reaction mixture was filtered over CELITE.TM.. The solids were washed with methanol (3.times.10 mL) and the filtrate and washings were then concentrated to give a light brown solid that was dissolved 4M HCl in dioxane (5 mL). The reaction was left at RT for 1 h and the solvent was evaporated. The product was then purified by reverse phase preparative HPLC using a 10 minutes gradient from 0% to 15% methanol in water (containing 0.1% HCOOH) to give title compound (4S)-4-[2-(morpholin-3-yl)acetamido]-5-oxo-5-[(2S)-2-[(2H-1,2,3,4-tetrazo- l-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C39) as white solid (12.1 mg, 29%). .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.90 (1H, m), 2.02 (1H, m), 2.30-2.45 (4H, m), 2.91-3.06 (3H, m), 3.25-3.32 (2H, m), 3.35-3.55 (2H, m), 2.75-3.85 (2H, m), 4.33 (1H, d, J=16 Hz), 4.50 (1H, d, J=16 Hz), 5.00 (1H, d, J=11 Hz), 5.92 (1H, s), 7.02 (1H, m), 7.65 (1H, d, J=7 Hz), 8.14 (1H, s), 8.50 (1H, m), 8.60 (1H, m), MS (LC/MS) m/z observed 502.02, expected 502.22 [M+H].
Example C40
(4S)-4-(2-CYCLOPENTYLACETAMIDO)-5-OXO-5-[(2S)-2-[(1H-1,2,3-TRIAZOL-4-YLMET- HYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN-1-YL]PENTANOIC ACID
[0550] (S)-Benzyl 5-((S)-2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-(2-cyclopentylacetamido)-5-oxopentanoate was prepared from (S)-benzyl 5-((S)-2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-((tert-butoxycarbonyl)amino)-5-oxopentanoate (from Example C11) and cyclopentylacetic acid using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 574.05, expected 574.28 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0551] Title compound (4S)-4-(2-cyclopentylacetamido)-5-oxo-5-[(2S)-2-[(1H-1,2,3-triazol-4-ylme- thyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyridin-1-yl]pentanoic acid (C40) was prepared from (S)-benzyl 5-((S)-2-(((1H-1,2,3-triazol-4-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrol- o[2,3-b]pyridin-1-yl)-4-(2-cyclopentylacetamido)-5-oxopentanoate using Method Q. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 1.06-1.18 (2H, m), 1.41-1.50 (2H, m), 1.52-1.58 (2H, m), 1.62-1.70 (2H, m), 1.80 (1H, m), 1.96-2.14 (4H, m), 2.30-2.47 (2H, m), 2.95 (1H, dd, J=4, 17 Hz), 3.45 (1H, dd, J=11, 17 Hz), 4.28-4.41 (2H, m), 4.96 (1H, dd, J=4, 11 Hz), 5.83 (1H, m), 7.03 (1H, dd, J=5, 7 Hz), 7.60-7.70 (2H, m), 8.05 (1H, d, J=8 Hz), 8.17 (1H, d, J=4 Hz), 8.70 (1H, m), MS (LC/MS) m/z observed 483.93, expected 484.23 [M+H].
Example C41
3-{[(1S,2S)-2-METHYL-1-{[(2S)-1-OXO-3-(2H-1,2,3,4-TETRAZOL-5-YL)-1-(2S)-2-- [(2H-1,2,3,4-TETRAZOL-5-YLMETHYL)CARBAMOYL]-1H,2H,3H-PYRROLO[2,3-B]PYRIDIN- -1-YL]PROPAN-2-YL]CARBAMOYL}BUTYL]CARBAMOYL}PROPANOIC ACID
[0552] (S)-2-(Fmoc-amino)-3-(2H-tetrazol-5-yl) propanoic acid (400 mg, 1.054 mmol, 1 eq.) was dissolved in DMF (5 mL). Morpholine (5 mL) was then added and the reaction was left at RT for 10 minutes, where it went to completion. The solvents were concentrated and the residue was dissolved in dioxane (10 mL) and Boc.sub.2O (276.1 mg, 1.265 mmol, 1.2 eq.) was added, followed by triethylamine (0.365 mL, 2.635 mmol, 2.5 eq.). The reaction was left at RT for 2 hrs and was then acidified to pH 4 with a saturated solution of citric acid. The solvent was evaporated and the product was purified by reverse phase C18 column chromatography using 10% methanol in water as the eluent. (S)-2-((tert-Butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoic acid was obtained as a colorless glass (202 mg, 75%). MS (LC/MS) m/z observed 257.87, expected 258.12 [M+H].sup.+. Compound was confirmed using LC/MS and moved to next step as it was.
[0553] Allyl 1-((S)-2-((tert-butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate was prepared from (S)-2-((tert-butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoic acid and (S)-allyl 2,3-dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate hydrochloride (from Example C30) using method C in DMF. MS (LC/MS) m/z observed 443.82, expected 444.20 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0554] Allyl 1-((S)-2-((tert-butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylate (90 mg, 0.203 mmol, 1 equiv) and Pd(PPh.sub.3).sub.4(46.9 mg, 0.0406, 0.2 equiv) were dissolved in CH.sub.2Cl.sub.2 (15 mL) under N.sub.2. Morpholine (0.053 mL, 0.609 mmol, 3 equiv) was then added and the reaction was left at RT for 1 h. The solvent was then evaporated and the product was purified by column chromatography reverse phase using 10% to 50% methanol in water as the eluent to give 1-((S)-2-((tert-butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid as a colorless glass 72 mg, 88%). MS (LC/MS) m/z observed 403.83, expected 404.17 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0555] tert-Butyl ((2S)-1-(2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2- ,3-b]pyridin-1-yl)-1-oxo-3-(2H-tetrazol-5-yl)propan-2-yl)carbamate was prepared from 1-((S)-2-((tert-butoxycarbonyl)amino)-3-(2H-tetrazol-5-yl)propanoyl)-2,3-- dihydro-1H-pyrrolo[2,3-b]pyridine-2-carboxylic acid and (2H-tetrazol-5-yl)methyl-amine using method A in DMF but without HCl treatment. MS (LC/MS) m/z observed 484.83, expected 485.21 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0556] tert-Butyl ((2S,3S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihyd- ro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxo-3-(2H-tetrazol-5-yl)propan-2-yl)am- ino)-3-methyl-1-oxopentan-2-yl)carbamate was prepared from tert-butyl ((2S)-1-(2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihydro-1H-pyrrolo[2- ,3-b]pyridin-1-yl)-1-oxo-3-(2H-tetrazol-5-yl)propan-2-yl)carbamate and Boc-L-Isoleucine using method A but the solvent was DMF for the coupling reaction. MS (LC/MS) m/z observed 597.88, expected 598.30 [M+H]. Compound was confirmed using LC/MS and moved to next step as it was.
[0557] Title compound 3-{[(1S,2S)-2-methyl-1-{[(2S)-1-oxo-3-(2H-1,2,3,4-tetrazol-5-yl)-1-[(2S)-- 2-[(2H-1,2,3,4-tetrazol-5-ylmethyl)carbamoyl]-1H,2H,3H-pyrrolo[2,3-b]pyrid- in-1-yl]propan-2-yl]carbamoyl}butyl]carbamoyl}propanoic acid (C41) was prepared from tert-butyl ((2S,3S)-1-(((S)-1-((S)-2-(((2H-tetrazol-5-yl)methyl)carbamoyl)-2,3-dihyd- ro-1H-pyrrolo[2,3-b]pyridin-1-yl)-1-oxo-3-(2H-tetrazol-5-yl)propan-2-yl)am- ino)-3-methyl-1-oxopentan-2-yl)carbamate and succinic anhydride using method I. .sup.1H NMR (400 MHz, DMSO-d6) .delta. 0.76-0.85 (6H, m), 1.05 (1H, m), 1.39 (1H, m), 1.65 (1H, m), 2.35-2.45 (4H, m), 2.60-2.70 (2H, m), 2.75 (1H, m), 3.25 (1H, m), 4.15 (1H, m), 4.51-4.61 (2H, m), 5.02 (1H, m), 6.25 (1H, s), 7.03 (1H, m), 7.63 (1H, d, J=7 Hz), 7.90 (1H, d, J=9 Hz), 8.08 (1H, m), 8.36 (1H, m), 9.25 (1H, s), MS (LC/MS) m/z observed 597.91, expected 598.26 [M+H].
Example D1
General Kinetic Enzyme Assay Protocol
[0558] A specific 2.times. assay buffer was prepared for the enzyme to be tested (see Table 2 for final 1.times. assay buffer compositions). If the assay buffer included DTT, it was added immediately prior to running the assay. A 2.times. enzyme mix was prepared (see Table 3 for enzyme assay conditions) at 80 uL per well. Compounds were screened at one or two appropriate concentrations (to determine the percent inhibition at those concentrations) and/or a full dose response curve (typically 8 points, to identify the IC.sub.50) in duplicate, triplicate, or higher replicates as needed. An appropriate control was also assessed in full dose response, in duplicate for each assay/plate. Background control wells consisted of 1.times. assay buffer, DMSO (5% v/v) and substrate. Positive control wells consisted of enzyme, DMSO (5% v/v) and substrate. Test compounds and control compounds were diluted in DMSO to 40.times. the final desired concentration. For example, a test compound may be tested in dose response, in serial, tripling dilution condition starting at 20 uM and ending at 9.1 nM (or any appropriate concentration range and dilution scheme). Control compounds were prepared similarly. Diluted compounds were prepared in a dilution plate and transferred to the reaction plate (96-well medium binding plate (Greiner Bio-One FLUOTRAC.TM.)) to allow for the desired final concentrations when added to the enzyme with AB. After mixing, the reaction plate was placed on a shaker (at 300 RPM) for 5 min, followed by incubation (covered) on the bench, for 20 min. Plates were warmed to reaction temperature (see Table 3) for a total incubation time of 30 min. Plates so prepared were ready for addition of substrate and the subsequent reaction.
[0559] An appropriate substrate for each assay was prepared in advance at 2.times. the final desired concentration (see Table 2) in DMSO. The appropriate substrate mix was added to each appropriate well on the reaction plate, and the plate was read immediately in the TECAN plate reader (TECAN INFINITE.RTM. M1000 Pro), set to the correct wavelength as needed for each assay (see Table 3) using 25 cycles, kinetic interval of 1 min, number of reads per well of 20 with shaking set to is, double orbital, 2 mm amplitude. For fluorescent assays the gain was set to optimal (50%).
TABLE-US-00002 TABLE 2 Assay Buffer Composition. Enzyme Assay Buffer Composition Caspase 1, 3, 4, 5, 7, 8*, 9 & 10/a 50 mM HEPES pH 7.2 (General caspase assay buffer) 50 mM NaCl 0.1% (w/v) CHAPS 10 mM EDTA 5% (v/v) Glycerol 10 mM DTT GzmB & Caspase 8 50 mM HEPES pH 7.5 10% (w/v) sucrose 0.2% (w/v) CHAPS 5 mM DTT *Can also use GzmB assay buffer for the Caspase-8 assay; Assay buffer components were sourced as follows: HEPES, DTT, Glycerol and sucrose: Sigma-Aldrich, St. Louis, MO, USA, NaCl and EDTA: Fisher Scientific, Pittsburgh, PA, USA, CHAPS: Calbiochem, Billerica, MA, USA.
TABLE-US-00003 TABLE 3 Enzyme assay conditions. Substrate Assay Enzyme Conc. Ex/Em .lamda.* Temp Control Name Conc. Name (.mu.M) (nm) (.degree. C.) Inhibitor hGzmB 10 nM Ac-IEPD-AMC 150 380/460 30 Ac-IEPD-CHO Caspase-1 6.25 mU/.mu.l YVAD-AFC 25 400/505 37 Z-VAD-FMK Caspase-3 and 6.25 mU/.mu.l Ac-DEVD-AMC 20 380/460 37 Z-VAD-FMK Caspase 7 Caspase-4 and 3.125 mU/ul Ac-WEHD-AFC 100 400/505 37 Z-WEHD-FMK Caspase-5 Caspase-8 3.125 mU/ul Ac-IEPD-AMC 75 380/460 30 Ac-IEPD-CHO Caspase-9 3.125 mU/ul LEHD-AFC 50 400/505 37 Q-LEHD-Oph Caspase-10/a 6.25 mU/.mu.l Ac-IETD-AMC 100 400/505 30 Ac-AEVD-CHO *Ex/Em .lamda. is the excitation and emission wavelengths at which to measure fluorescence. Enzyme and substrate concentrations are the final concentrations in the well. Note that most protocols require preparing 2X enzyme and substrate mixes, as they are diluted 2-fold in the well.
[0560] Enzymes were sourced as follows: hGzmB, Froelich Lab, Northshore University Health Systems Research Institute, Evanston, Ill., USA; Caspases, Biovision Inc., Milpitas, Calif., USA. Substrates were sourced as follows: Ac-IEPD-AMC, California Peptide Research Inc., Napa, Calif., USA; YVAD-AFC, Biovision Inc., Milpitas, Calif., USA; Ac-DEVD-AMC, LEHD-AFC, AC-WEHD-AFC and Ac-IETD-AMC, Enzo Life Sciences Inc, Farmingdale, N.Y., USA. Control inhibitors were sourced as follows: Ac-IEPD-CHO, Ac--WEHD-FMK and Q-LEHD-Oph, Biovision Inc., Milpitas, Calif., USA; Z-VAD-FMK, R&D Systems, Minneapolis, Minn., USA; and Ac-AEVD-CHO, Enzo Life Sciences Inc, Farmingdale, N.Y., USA.
Example D2
Human Granzyme B Enzymatic Inhibition Assay
[0561] An in vitro fluorogenic detection assay for assessing the IC.sub.50 and/or percent inhibition at a given concentration of inhibitors against human Granzyme B (hGzmB) enzyme was performed as described in Example D1. When appropriate, percent inhibition data was collected and fitted to generate IC.sub.50 data using GraphPad Prism 5 (GraphPad Software, La Jolla Calif. USA, www.graphpad.com) and its non-linear regression analysis tools or other equivalent tools.
[0562] Select compounds of Examples A1 and C1-C41 exhibited inhibitory activity against hGzmB. Each of the compounds of the invention identified in Table 1 exhibited Granzyme B inhibitory activity.
[0563] In certain embodiments, select compounds exhibited IC.sub.50<50,000 nM. In other embodiments, select compounds exhibited IC.sub.50<10,000 nM. In further embodiments, select compounds exhibited IC.sub.50<1,000 nM. In still further embodiments, select compounds exhibited IC.sub.50<100 nM. In certain embodiments, select compounds exhibited IC.sub.50 from 10 nM to 100 nM, preferably from 1 nM to 10 nM, more preferably from 0.1 nM to 1 nM, and even more preferably from 0.01 nM to 0.1 nM.
Example D3
Human Caspase Enzymatic Inhibition Assay
[0564] In vitro fluorogenic detection assays for assessing the IC.sub.50 and/or percent inhibition at a given concentration of inhibitors, against a set of human Caspase enzymes, was performed as described in Example D1. Representative compounds of the invention do not significantly inhibit any caspase enzyme tested at a concentration of 50 .mu.M.
[0565] In certain embodiments, the compounds exhibited less than 50% inhibition at 50 .mu.M. In other embodiments, the compounds exhibited greater than 50% inhibition at 50 .mu.M, but less than 10% inhibition at 25 .mu.M.
Example D4
General Kinetic Enzyme Assay Protocol (384 Well)
[0566] A specific 2.times. assay buffer was prepared for the enzyme to be tested (see Table 4 for final 1.times. assay buffer compositions). If the assay buffer included DTT, it was added immediately prior to running the assay. A 2.times. enzyme mix was prepared (see Table 3 for enzyme assay conditions) at 26 uL per well. Compounds were screened at one or two appropriate concentrations (to determine the percent inhibition at those concentrations) and/or a full dose response curve (typically 12 points, to identify the IC.sub.50) in duplicate, triplicate, or higher replicates as needed. An appropriate control was also assessed in full dose response, in duplicate for each assay/plate. Background control wells consisted of 1.times. assay buffer and substrate. Positive control wells consisted of enzyme (no DMSO) and substrate. Test compounds and control compounds were diluted in 1.times. Assay Buffer to 15.times. the final desired concentration. For example, a test compound may be tested in dose response, in serial, tripling dilution condition starting at 20 uM and ending at 0.1 nM (or any appropriate concentration range and dilution scheme). Control compounds were prepared similarly. Diluted compounds were prepared in a dilution plate and transferred to the reaction plate (384-well medium binding plate (Greiner Bio-One FLUOTRAC.TM.)) to allow for the desired final concentrations when added to the enzyme with AB. After mixing, the reaction plate was placed on a shaker (at 300 RPM) for 5 min, followed by incubation (covered) on the bench, for 20 min. Plates were warmed to reaction temperature (see Table 5) for 5 mins for a total incubation time of 30 min. Plates so prepared were ready for addition of substrate and the subsequent reaction.
[0567] An appropriate substrate for each assay was prepared in advance at 2.times. the final desired concentration (see Table 4) in assay buffer. 30 uL of the appropriate substrate mix was added to each appropriate well on the reaction plate, and the plate was read immediately in the TECAN plate reader (TECAN INFINITE.RTM. M1000 Pro), set to the correct wavelength as needed for each assay (see Table 5) using 15 cycles, kinetic interval of 1 min, number of reads per well of 20 with shaking set to is, double orbital, 2 mm amplitude. For fluorescent assays the gain was set to optimal (100% with gain regulation) for all assays except human GzmB which was set to 85 (with the z set at 23000 um).
TABLE-US-00004 TABLE 4 Assay Buffer Composition. Enzyme Assay Buffer Composition Caspase 1, 3, 4, 5, 7, 8*, 9 & 10/a 50 mM HEPES pH 7.2 (General caspase assay buffer) 50 mM NaCl 0.1% (w/v) CHAPS 10 mM EDTA 5% (v/v) Glycerol 10 mM DTT GzmB & Caspase 8 50 mM HEPES pH 7.5 0.2% (w/v) CHAPS 5 mM DTT Cathepsin G 320 mM Tris-HCL pH 7.4 3.2M NaCl *Can also use GzmB assay buffer for the Caspase-8 assay; Assay buffer components were sourced as follows: HEPES, DTT, Glycerol and sucrose: Sigma-Aldrich, St. Louis, MO, USA, NaCl and EDTA: Fisher Scientific, Pittsburgh, PA, USA, CHAPS: Calbiochem, Billerica, MA, USA.
TABLE-US-00005 TABLE 5 Enzyme assay conditions. Substrate Enzyme Conc. Ex/Em .lamda.* Assay Control Name Conc. Name (.mu.M) (nm) Temp (.degree. C.) Inhibitor hGzmB 10 nM Ac-IEPD- 50 380/460 30 V2248 AMC Caspase-1 12.5 mU/.mu.L YVAD- 5 400/505 37 Z-VAD- AFC FMK Caspase-3 and 0.8 mU/.mu.L Ac-DEVD- 40 380/460 37 Z-VAD- Caspase 7 &1.5 mU/.mu.L AMC &5 FMK Caspase-4 and 3.125mU/uL & Ac-WEHD- 40 400/505 37 Z-WEHD- Caspase-5 1.5mU/uL AFC & 100 FMK Caspase-8 4 mU/uL Ac-IEPD- 80 380/460 37 Ac-IEPD- AMC CHO Caspase-9 2mU/uL LEHD-AFC 50 400/505 37 Q-LEHD- Oph Caspase-10/a 3 mU/.mu.L Ac-IETD- 10 400/505 37 Ac-AEVD- AMC CHO Cathepsin G 200 nM Suc-AAPF- 200 uM 410 25 Cat G pNA absorbance inhibitor Human 0.125 ug/mL MeOSuc- 50 384/500 37 Sivelestat Neutrophil AAPF-AFC Elastase *Ex/Em .lamda. is the excitation and emission wavelengths at which to measure fluorescence. Enzyme and substrate concentrations are the final concentrations in the well. Note that most protocols require preparing 2X enzyme and substrate mixes, as they are diluted 2-fold in the well.
[0568] Enzymes were sourced as follows: hGzmB, Froelich Lab, Northshore University Health Systems Research Institute, Evanston, Ill., USA; Caspases and Elastase, Biovision Inc., Milpitas, Calif., USA; Cathepsin G, Athens Research and Technologies, Athens, Ga., USA. Substrates were sourced as follows: Ac-IEPD-AMC, California Peptide Research Inc., Napa, Calif., USA; YVAD-AFC and MeOSuc-AAPF-AFC Biovision Inc., Milpitas, Calif., USA; LEHD-AFC and Suc-AAPF-pNA Millipore, Billerica Mass., USA. Ac-DEVD-AMC, AC-WEHD-AFC and Ac-IETD-AMC, Enzo Life Sciences Inc, Farmingdale, N.Y., USA. Control inhibitors were sourced as follows: Ac-IEPD-CHO, Ac--WEHD-FMK, Q-LEHD-Oph and CatG inhibito,r Biovision Inc., Milpitas, Calif., USA; Z-VAD-FMK, R&D Systems, Minneapolis, Minn., USA; and Ac-AEVD-CHO, Enzo Life Sciences Inc, Farmingdale, N.Y., USA. Sivelestat, Tocris Bioscience, Bristol, UK.
Example D5
Human Granzyme B Enzymatic Inhibition Assay
[0569] An in vitro fluorogenic detection assay for assessing the IC.sub.50 and/or percent inhibition at a given concentration of inhibitors against human Granzyme B (hGzmB) enzyme was performed as described in Example D4. When appropriate, percent inhibition data was collected and fitted to generate IC.sub.50 data using GraphPad Prism 5 (GraphPad Software, La Jolla Calif. USA, www.graphpad.com) and its non-linear regression analysis tools or other equivalent tools.
[0570] Select compounds of Examples A1 and C1 to C41 exhibited inhibitory activity against hGzmB. Each of the compounds of the invention identified in Table 1 exhibited Granzyme B inhibitory activity.
[0571] In certain embodiments, select compounds exhibited IC.sub.50<50,000 nM. In other embodiments, select compounds exhibited IC.sub.50<10,000 nM. In further embodiments, select compounds exhibited IC.sub.50<1,000 nM. In still further embodiments, select compounds exhibited IC.sub.50<100 nM. In certain embodiments, select compounds exhibited IC.sub.50 from 10 nM to 100 nM, preferably from 1 nM to 10 nM, more preferably from 0.1 nM to 1 nM, and even more preferably from 0.01 nM to 0.1 nM.
Example D6
Human Caspase Enzymatic Inhibition Assay
[0572] In vitro fluorogenic detection assays for assessing the IC.sub.50 and/or percent inhibition at a given concentration of inhibitors, against a set of human Caspase enzymes, was performed as described in Example D4. Representative compounds of the invention do not significantly inhibit any caspase enzyme tested at a concentration of 50 .mu.M.
[0573] In certain embodiments, the compounds exhibited less than 50% inhibition at 50 .mu.M. In other embodiments, the compounds exhibited greater than 50% inhibition at 50 .mu.M, but less than 10% inhibition at 25 .mu.M.
Example D7
Inhibition of Cell Detachment by GzmB Assay
[0574] HDFa primary human fibroblasts were plated at 10 k/well in 200 ul, approximately hrs before treatment. The next day, controls and 100 nM GzmB (recombinant, human) plus or minus inhibitor treatments were prepared in serum-free media. GzmB and inhibitor were incubated for 20 minutes at RT before adding to cells. Before addition, media and serum was removed from the cells and the cells were washed with PBS (1.times.), using pipettes to prevent disturbing the cells. Treatment preparations (100 ul) were added to the wells and incubated for 7 hours in a tissue culture incubator. After 7 hours, media and treatments were removed and the cells were washed with PBS (1.times.) to removed detached cells, using pipettes only. Phase pictures were taken then the PBS was removed and replaced with 100 uL of serum-free media and 20 uL of MTS and the cells were allowed to incubate for 3 hours in a cell culture incubator. After 3 hours the absorbance was read at 490 nm and a percent inhibition value for the treatments with inhibitors was determined from the control wells. The resulting date is shown in Table 6.
TABLE-US-00006 TABLE 6 Inhibition of Cell Detachment by GzmB Results. Compound Percent Inhibition of cell detachment at 50 uM C4 100%
Example D8
Inhibition of Fibronectin Cleavage by GzmB
[0575] Black, 96 well high-binding assay plates (Griener Bio-one) were treated overnight at 4.degree. C. with 40 uL of 8 ug/mL Hilyte Fluor 488 labeled Fibronectin (Cytoskeleton, Inc). After fibronectin coating, plates were washed 3 times in buffer (20 mM Tris-HCl, pH 7.4, 20 mM NaCl) then once with granzyme B assay buffer (50 mM HEPES, pH 7.5, 0.1% CHAPS). After washing, 50 uL of granzyme B assay buffer was added to each fibronectin-coated well. In a separate non-binding 96 well assay plate 5 uL of 20.times. inhibitor serial dilution stocks were added to 45 uL of 2.22.times.GzmB mix to establish inhibition (enzyme/inhibitor mixes were all prepared in granzyme B assay buffer and were incubated first at room temperature for 20 minutes, then at 30.degree. C. for another 10 minutes). After incubation, 50 uL of this 2.times. enzyme/inhibitor mix was added to the corresponding coated well to initiate fibronectin cleavage (20 nM final granzyme B concentration, 8-point inhibitor dilution series starting at 50 uM). The assay was conducted at 30.degree. C. in the TECAN plate reader (TECAN INFINITE.RTM. M1000 Pro), which was programmed to monitor the kinetic fluorescence polarization signal (filter set Ex/Em 470 nm/527 nm) with readings taken every minute, for 1 hour. Proteolytic activity was evaluated as the rate of fluorescence enhancement in the parallel emission over the linear range of the reaction. % Inhibition values were calculated from assay controls and the resulting date is shown in Table 7.
TABLE-US-00007 TABLE 7 Inhibition of Fibronectin Cleavage by GzmB Results. Percent Inhibition at Inhibitor Concentration Compound 50 uM 5.56 uM 0.62 uM A1 88% 82% 66 C4 98% 79% 45% C9 91% 81% 58% C17 94% 77% 56%
Example D9
Inhibition of Cell Adhesion by GzmB Cleavage of Fibronectin
[0576] Black, 96 well high-binding clear-bottom assay plates (Griener Bio-one) were treated overnight at 4.degree. C. with 40 uL of 5 ug/mL Fibronectin (Sigma-Aldrich). After fibronectin coating, wells were washed 3 times in Tris wash buffer (20 mM Tris-HCl, pH 7.4, 20 mM NaCl) then once with granzyme B assay buffer (HEPES, (50 mM, pH 7.5), CHAPS (0.1%)). After washing, 50 uL of granzyme B assay buffer was added to each fibronectin-coated well. In a separate non-binding 96 well assay plate 5 uL of relevant 20.times. inhibitor dilution stocks were added to 45 uL of 2.22.times.GzmB mix to establish inhibition (enzyme/inhibitor mixes were all prepared in granzyme B assay buffer and were incubated first at room temperature for 20 minutes, then at 30.degree. C. for another 10 minutes). After incubation, 50 uL of this 2.times. enzyme/inhibitor mix was added to the corresponding coated well to initiate fibronectin cleavage (20 nM final granzyme B concentration, 3 final inhibitor concentrations--0.1 uM, 10 uM and 100 uM). The assay was conducted at 30.degree. C. in a plate warmer for 2 hours. After incubation, wells were washed 3 times with PBS, and then blocked with 2% BSA in PBS for 1 hour at room temperature. After sufficient blocking, wells were washed an additional 3 times with PBS to remove residual BSA. 3T3 fibroblasts, harvested from sub-confluent conditions, were prepared in serum free DMEM and introduced into the treated wells at 10,000 cells/well. Cells were allowed to adhere for 90 minutes or until appropriate attached phenotype was detected. After attachment, wells were agitated with gentle repeat pipetting 3 times, gently aspirated manually and washed once with PBS. Wells were then fixed with 4% paraformaldehyde in PBS for 1 hr, washed twice with PBS and stained with the nuclear dye DAPI. Microscopic detection and counting of stained nuclei was performed using IMAGE-PRO.RTM. Plus software. Cell count was normalized to % Cell Adhesion. The results are shown in Table 8.
TABLE-US-00008 TABLE 8 Inhibition of Cell Adhesion by GzmB Results. Percent Cell Adhesion at Inhibitor Concentration Compound 100 uM 10 uM 0.1 uM C4 146% 88% 13%
* * * * *
References

















































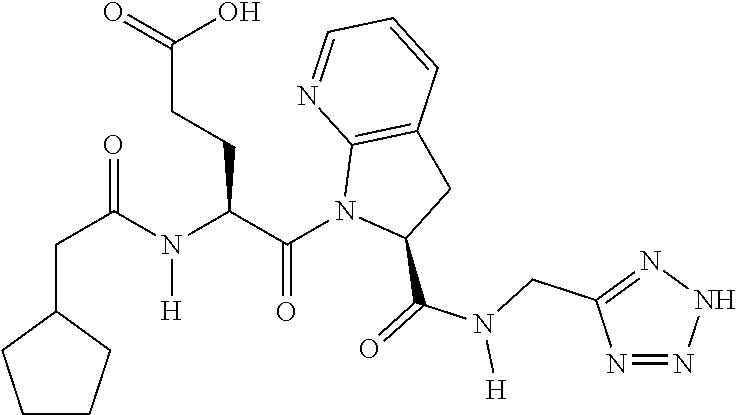
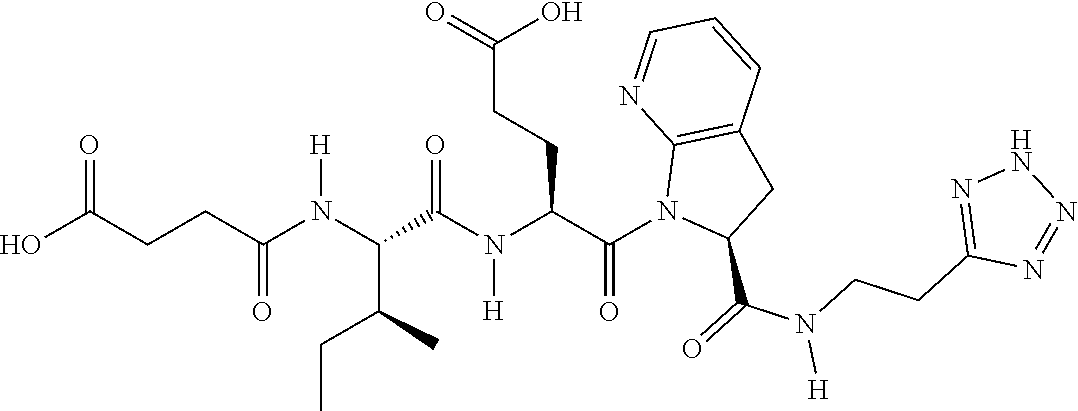




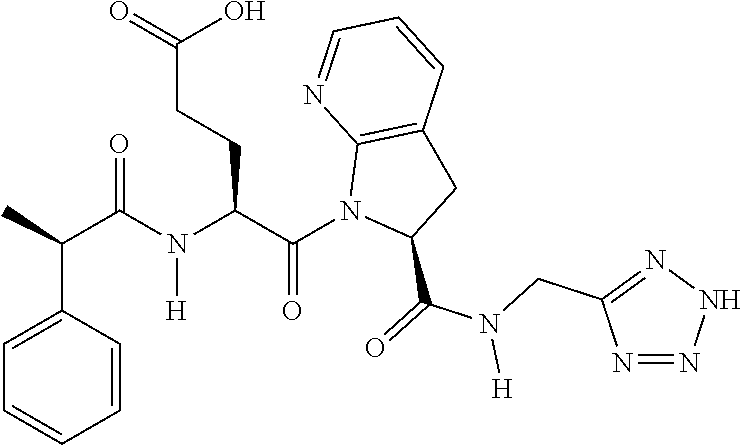









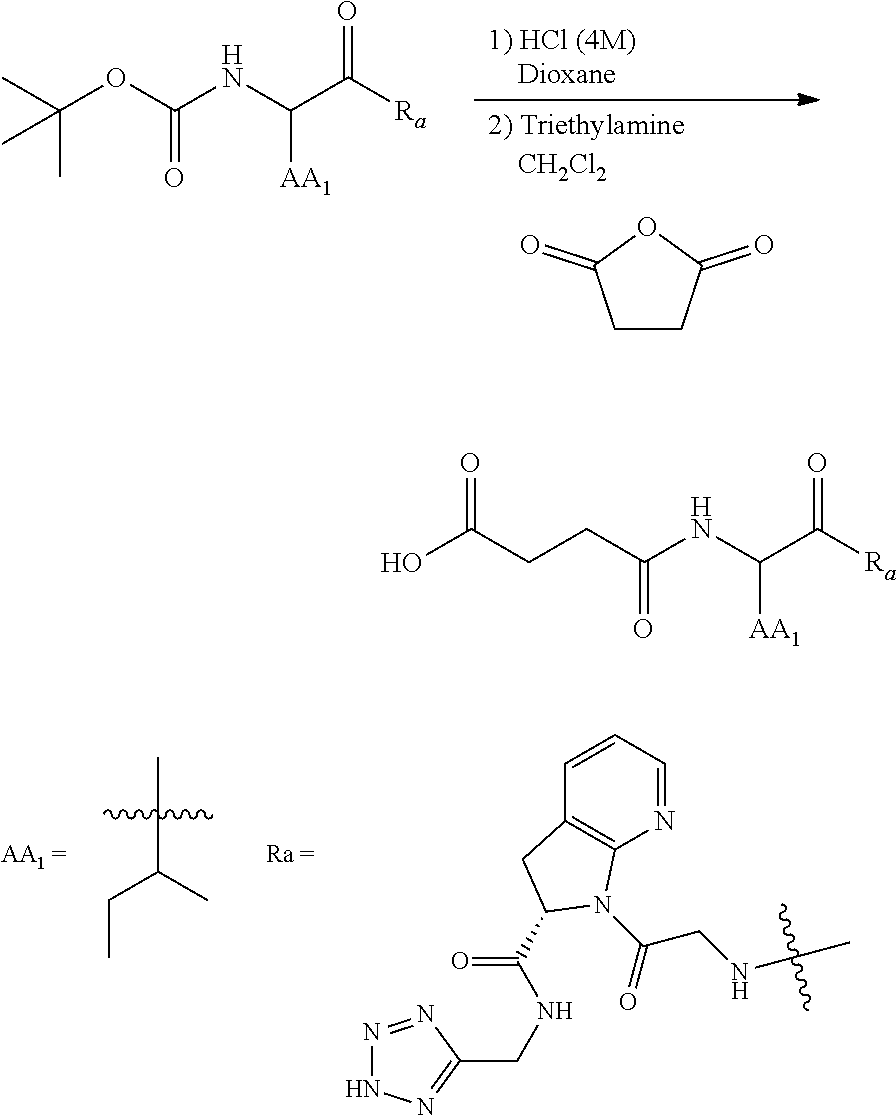









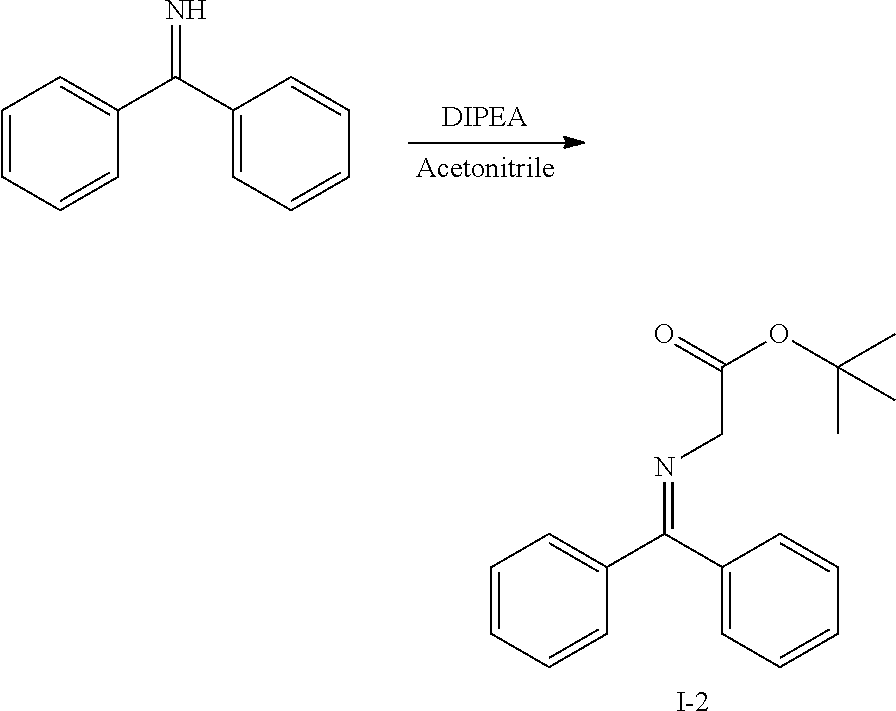

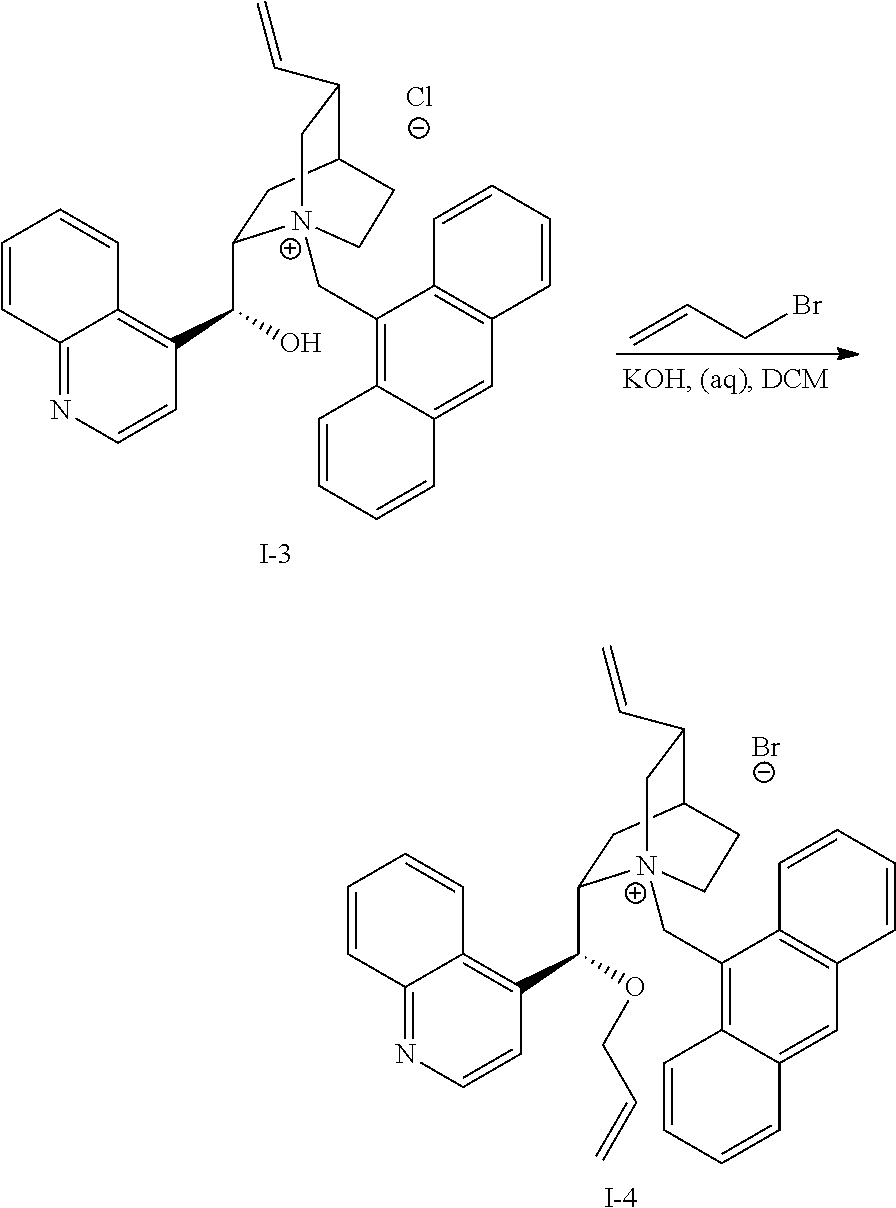









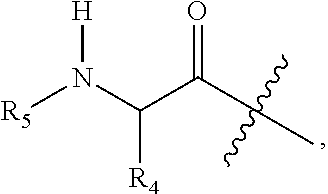









D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.