Techniques For Neuromodulation Using Electromagnetic Energy
Tracey; Kevin J. ; et al.
U.S. patent application number 16/342554 was filed with the patent office on 2020-02-13 for techniques for neuromodulation using electromagnetic energy. This patent application is currently assigned to THE FEINSTEIN INSTITUTE FOR MEDICAL RESEARCH. The applicant listed for this patent is THE FEINSTEIN INSTITUTE FOR MEDICAL RESEARCH. Invention is credited to Chad E. Bouton, Sangeeta S. Chavan, Kevin J. Tracey.
| Application Number | 20200046992 16/342554 |
| Document ID | / |
| Family ID | 62025502 |
| Filed Date | 2020-02-13 |










View All Diagrams
| United States Patent Application | 20200046992 |
| Kind Code | A1 |
| Tracey; Kevin J. ; et al. | February 13, 2020 |
TECHNIQUES FOR NEUROMODULATION USING ELECTROMAGNETIC ENERGY
Abstract
The subject matter of the present disclosure generally relates to techniques for neuromodulation of tissue that include applying energy (e.g., electromagnetic energy) into the target tissue to cause altered activity of a neuron in the tissue. In certain embodiments, the altered activity causes a change in one or molecules in the tissue or blood.
| Inventors: | Tracey; Kevin J.; (Old Greenwich, CT) ; Chavan; Sangeeta S.; (Syosset, NY) ; Bouton; Chad E.; (Darien, CT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | THE FEINSTEIN INSTITUTE FOR MEDICAL
RESEARCH Manhasset NY |
||||||||||
| Family ID: | 62025502 | ||||||||||
| Appl. No.: | 16/342554 | ||||||||||
| Filed: | October 31, 2017 | ||||||||||
| PCT Filed: | October 31, 2017 | ||||||||||
| PCT NO: | PCT/US2017/059163 | ||||||||||
| 371 Date: | April 17, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62415212 | Oct 31, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61N 1/40 20130101; A61B 5/6825 20130101; A61B 5/418 20130101; A61N 1/36014 20130101; A61B 5/6823 20130101; A61B 5/40 20130101; A61B 5/04001 20130101; A61N 2/02 20130101; A61N 7/00 20130101; A61N 2/006 20130101; A61N 2007/0026 20130101; A61B 2090/378 20160201; A61B 5/407 20130101 |
| International Class: | A61N 1/40 20060101 A61N001/40 |
Goverment Interests
STATEMENT OF GOVERNMENT SUPPORT
[0002] This invention was made with government support under grant number W911NF-09-1-0125 awarded by the Defense Advanced Research Projects Agency (DARPA). The government has certain rights in the invention.
Claims
1. A method of neuromodulation, comprising: focusing an electromagnetic energy source on an internal tissue field in a patient in need of neuromodulation, the internal tissue field comprising one or more neurons, wherein the electromagnetic energy source is not in direct contact with the tissue field of focus; and applying one or more energy pulses to the patient's internal tissue in the field of focus via the electromagnetic energy source to cause a change in activity in the patient's internal tissue, wherein the change is relative to a baseline activity; wherein the electromagnetic energy source is extracorporeal and the one or more energy sources are applied transdermally.
2. The method of claim 1, wherein the patient's internal tissue is a peripheral tissue.
3. The method of claim 1, wherein the patient's internal tissue is a spleen.
4. The method of claim 1, wherein the patient's internal tissue is a lymph node.
5. The method of claim 1, wherein the baseline is indicative of the activity of the internal tissue before applying the one or more energy pulses.
6. The method of claim 5, wherein a time at which the one or more energy pulses are applied is designated as time zero, and wherein the baseline is indicative of the activity at a time immediately before the energy pulses are applied.
7-9. (canceled)
10. The method of claim 1, wherein the change in activity is a change in a level of cytokine release in the internal tissue.
11. The method of claim 1, wherein the change in activity is a change in a level of a cytokine in the patient's blood.
12. The method of claim 1, wherein the change in activity is a decrease in a level of a cytokine in the patient's blood.
13. The method of claim 10, wherein the cytokine is one or more of TNF-alpha, IL-1, IL-6 and HMGB1.
14. (canceled)
15. The method of claim 1, wherein the electromagnetic energy source generates an electromagnetic field.
16. The method of claim 1, wherein the patient has one or more of endotoxemia, sepsis, septicemia, septic shock, inflammation, and a pathogenic consequence of an inflammatory condition or an inflammatory cytokine cascade.
17. The method of claim 1 comprising: controlling an electromagnetic energy source to apply one or more energy pulses to a patient's internal immune tissue and according to one or more parameters of the energy pulses to cause a change in a level of cytokine release in the internal tissue in direct response to the one or more energy pulses; receiving information related to the release of the cytokine relative to a baseline before the one or more energy pulses are applied; and changing the one or more parameters based on the information; wherein the electromagnetic energy source is configured to be positioned on or above a patient's skin.
18. The method of claim 17, wherein the cytokine is one or more of TNF-alpha, IL-1, IL-6 and HMGB1.
19. (canceled)
20. The method of claim 17, wherein the patient's internal immune tissue is a spleen.
21. The method of claim 17, wherein the patient's internal immune tissue is a lymph node.
22. The method of claim 1, wherein the patient has one or more of endotoxemia, sepsis, septicemia, septic shock, inflammation, and a pathogenic consequence of an inflammatory condition or an inflammatory cytokine cascade.
23. The method of claim 1 comprising: positioning an electromagnetic field generator at a location at which the electromagnetic field generator is capable of stimulating one or more neurons in an internal lymphatic tissue field; and applying one or more energy pulses to the tissue field via the electromagnetic field generator to alter activity in the one or more neurons in response to the one or more energy pulses to modulate the lymphatic tissue in the field; wherein the energy pulses are applied non-invasively.
24. The method of claim 23, wherein the lymphatic tissue is a lymph node and/or spleen.
25. (canceled)
26. The method of claim 24, wherein the number of lymphocytes is increased in a tissue and/or serum levels of one or more proinflammatory cytokines is decreased in a subject, thereby treating an infection and/or an inflammation in the subject.
27-32. (canceled)
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Patent Application No. 62/415,212, filed on Oct. 31, 2016, the entirety of which is incorporated by reference herein for all purposes.
BACKGROUND
[0003] The subject matter disclosed herein relates to neuromodulation and more specifically, to techniques for modulating a physiological response using energy applied from an energy source, in particular an electromagnetic energy source.
[0004] Neuromodulation has been used to treat a variety of clinical conditions. For example, electrical stimulation at various locations along the spinal cord has been used to treat chronic back pain. Such treatment may be performed by an implantable device that periodically generates electrical energy that is applied to a tissue to activate certain nerve fibers, which in turn may result in a decreased sensation of pain. In the case of spinal cord stimulation, the stimulating electrodes are generally positioned in the epidural space, although the pulse generator may be positioned somewhat remotely from the electrodes, e.g., in the abdominal or gluteal region, but connected to the electrodes via conducting wires. In other implementations, deep brain stimulation may be used to stimulate particular areas of the brain to treat movement disorders, and the stimulation locations may be guided by neuroimaging.
[0005] Such nervous system stimulation is generally targeted to the local nerve or brain cell function and is mediated by electrodes that deliver electrical pulses and that are positioned at or near the target nervous tissue. However, positioning electrodes at or near the target is challenging. For example, such techniques may involve surgical placement of the electrodes that deliver the energy. In addition, specific tissue targeting via neuromodulation is challenging. For example, electrodes that are positioned at or near certain target nerves mediate neuromodulation by triggering action potentials in nearby nerve fibers, which in turn results in the propagation of the action potentials through the nerves and neurotransmitter release at nerve synapses. This may result in a relatively larger or more diffuse physiological effect than desired. Because neural pathways are complex and interconnected, a more targeted modulated effect may be more clinically predictable and useful.
BRIEF DESCRIPTION OF THE DRAWINGS
[0006] These and other features, aspects, and advantages of the present invention will become better understood when the following detailed description is read with reference to the accompanying drawings in which like characters represent like parts throughout the drawings.
[0007] FIG. 1 is a schematic representation of a neuromodulation system using a pulse generator according to embodiments of the disclosure.
[0008] FIG. 2 is a block diagram of a neuromodulation system according to embodiments of the disclosure.
[0009] FIG. 3 is a schematic representation of an ultrasound energy application device in operation.
[0010] FIG. 4 is a flow diagram of a neuromodulation technique according to embodiments of the disclosure.
[0011] FIG. 5 shows the results of a standard electrode stimulation on direct and contralateral popliteal lymph nodes as well as distal axillary lymph nodes.
[0012] FIG. 6 shows the results of a comparison of lymphocyte counts in directly stimulated popliteal vs. non-stimulated popliteal and non-stimulated popliteal lymph nodes after ultrasound stimulation.
[0013] FIG. 7 shows the concentration of norepinephrine, epinephrine, and dopamine in lymph node dissociated tissue after ultrasound stimulation.
[0014] FIG. 8 shows the concentration of norepinephrine, epinephrine, acetylcholine, and dopamine in the spleen after ultrasound stimulation.
[0015] FIG. 9A shows the concentration of acetylcholine in the spleen after ultrasound stimulation for a group antigen-naive animals relative to a control group of unstimulated antigen-naive animals.
[0016] FIG. 9B shows the concentration of acetylcholine in the spleen after ultrasound stimulation for another group of antigen-naive animals relative to a control group of unstimulated antigen-naive animals.
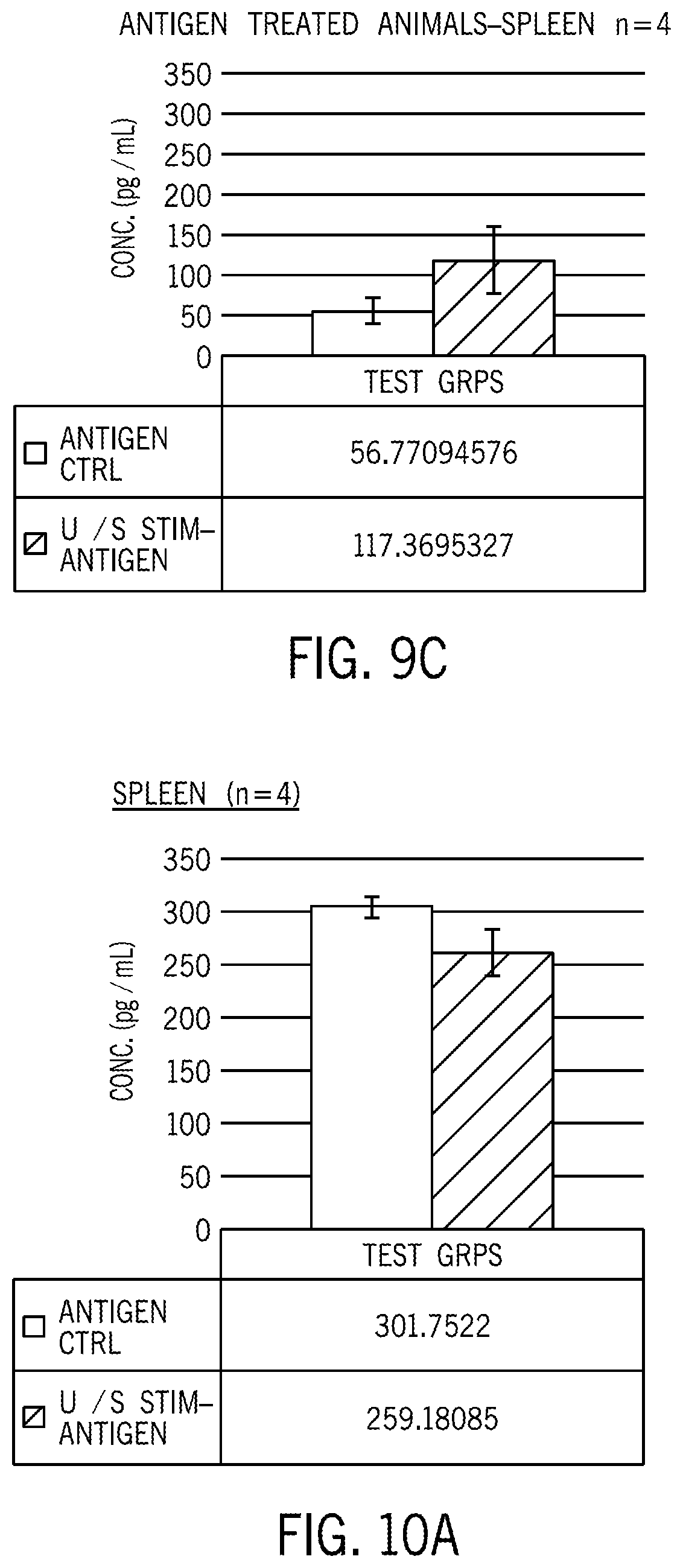
[0017] FIG. 9C shows the concentration of acetylcholine in the spleen after ultrasound stimulation for a group of antigen-treated animals relative to a control group of unstimulated but antigen-treated animals.
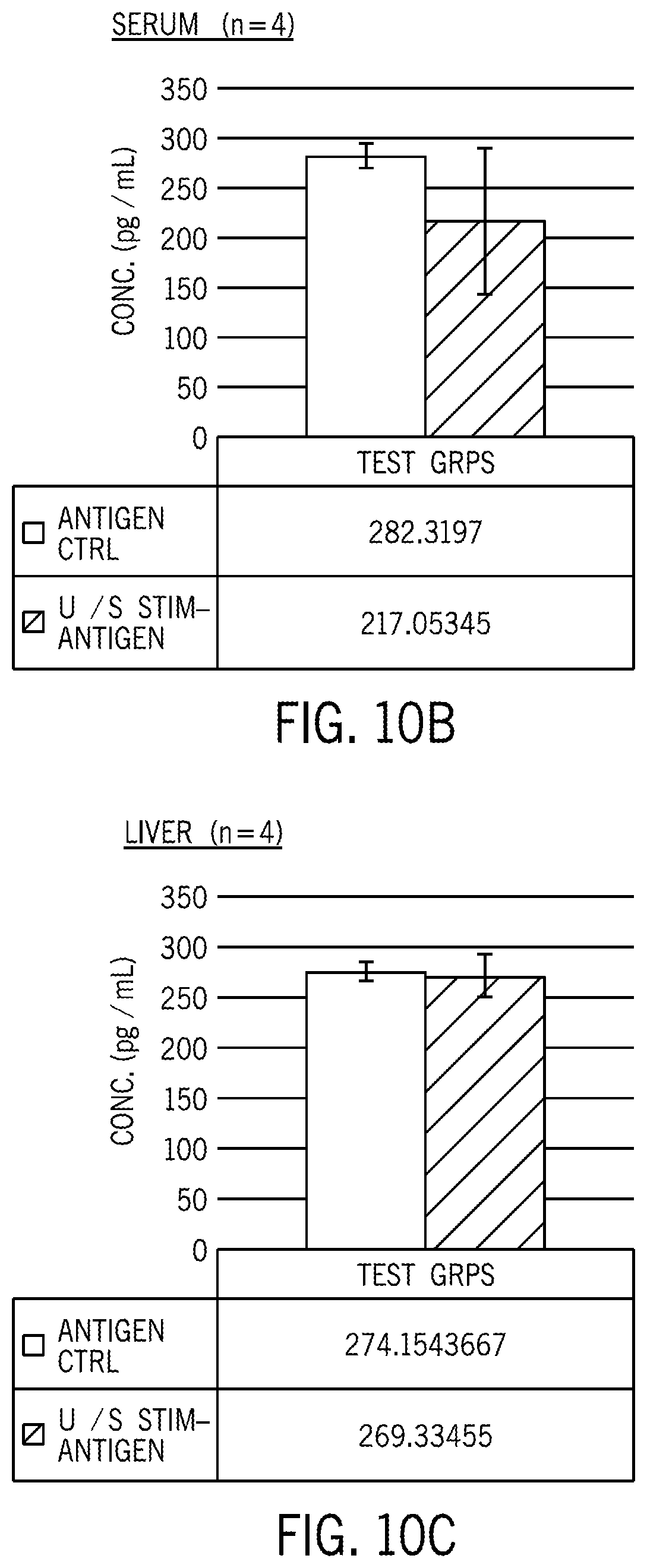
[0018] FIG. 10A shows the concentration of TNF-alpha in the spleen for a group of antigen-treated animals relative to a control group of unstimulated but antigen-treated animals.
[0019] FIG. 10B shows the concentration of TNF-alpha in the serum for a group of antigen-treated animals relative to a control group of unstimulated but antigen-treated animals.
[0020] FIG. 10C shows the concentration of TNF-alpha in the liver for a group of antigen-treated animals relative to a control group of unstimulated but antigen-treated animals.
[0021] FIG. 11A-11B are examples of a minimally-invasive ultrasound stimulator (A) and of a minimally-invasive electromagnetic stimulator (B) that can be inserted through a small incision or through the vasculature of a subject to be adjacent to a desired target tissue for localized stimulation of the tissue.
[0022] FIG. 12 shows the serum concentration of TNF after electromagnetic local tissue stimulation.
[0023] FIG. 13 shows a dose-dependent relationship of the serum concentration of TNF after electromagnetic local tissue stimulation.
[0024] FIG. 14 shows a dose-dependent relationship of the serum concentration of TNF after electromagnetic local tissue stimulation.
[0025] FIG. 15 shows the serum concentration of TNF after electromagnetic stimulation in mice after undergoing a vagotomy.
[0026] FIG. 16 shows the serum concentration of TNF after electromagnetic local tissue stimulation.
[0027] FIG. 17 shows the spleen concentration of TNF after electromagnetic local tissue stimulation.
DETAILED DESCRIPTION
[0028] One or more specific embodiments will be described below. In an effort to provide a concise description of these embodiments, not all features of an actual implementation are described in the specification. It should be appreciated that in the development of any such actual implementation, as in any engineering or design project, numerous implementation-specific decisions must be made to achieve the developers' specific goals, such as compliance with system-related and business-related constraints, which may vary from one implementation to another. Moreover, it should be appreciated that such a development effort might be complex and time consuming, but would nevertheless be a routine undertaking of design, fabrication, and manufacture for those of ordinary skill having the benefit of this disclosure.
[0029] Any examples or illustrations given herein are not to be regarded in any way as restrictions on, limits to, or express definitions of, any term or terms with which they are utilized. Instead, these examples or illustrations are to be regarded as being described with respect to various particular embodiments and as illustrative only. Those of ordinary skill in the art will appreciate that any term or terms with which these examples or illustrations are utilized will encompass other embodiments that may or may not be given therewith or elsewhere in the specification and all such embodiments are intended to be included within the scope of that term or terms. Language designating such non-limiting examples and illustrations includes, but is not limited to: "for example", "for instance", "such as", "e.g.", "including" and "in one (an) embodiment."
[0030] The present techniques relate to local modulation of axoextracellular or axo somatic or other synapses at axon terminals in the tissue via the application of energy by an energy source. Synapses between neurons and nonneuronal cells may be modulated to alter the activity in the synapse, e.g., the release of neurotransmitters from the presynaptic neuron. In turn, the altered activity may lead to local effects. The present techniques permit energy to be focused in a targeted manner on a volume of tissue that includes the axon terminals to achieve desired outcomes. In one example, the axoextracellular synapse is between a neuron and an immune cell. Accordingly, the application of energy leads to modulation of immune function in the targeted tissue.
[0031] The human nervous system is a complex network of nerve cells, or neurons, found centrally in the brain and spinal cord and peripherally in the various nerves of the body. Neurons have a cell body, dendrites and an axon. A nerve is a group of neurons that serve a particular part of the body. Nerves may contain several hundred neurons to several hundred thousand neurons. Nerves often contain both afferent and efferent neurons. Afferent neurons carry signals to the central nervous system and efferent neurons carry signals to the periphery. A group of neuronal cell bodies in one location is known as a ganglion. Electrical signals generated in the nerves (e.g., via upstream stimulation, which may be intrinsic or externally applied) are conducted via neurons and nerves. Neurons release neurotransmitters at synapses (connections) adjacent to a receiving cell to allow continuation and modulation of the electrical signal. In the periphery, synaptic transmission often occurs at ganglia.
[0032] The electrical signal of a neuron is known as an action potential. Action potentials are initiated when a voltage potential across the cell membrane exceeds a certain threshold. This action potential is then propagated down the length of the neuron. The compound action potential of a nerve is complex and represents the sum of action potentials of the individual neurons in it. The junction between the axon terminals of a neuron and the receiving cell is called a synapse. Action potentials travel down the axon of the neurons to its axon terminal, the distal termination of the branches of an axon nerve that forms a presynaptic ending or a synaptic knob of the nerve fiber. The electrical impulse of the action potential triggers migration of vesicles containing neurotransmitters to a presynaptic membrane of the presynaptic ending and ultimately the release of the neurotransmitters into a synaptic cleft. A synapse that reaches a synaptic knob to convert the electrical signal of the action potential to a chemical signal of neurotransmitter release is a chemical synapse. Chemical synapses may be contrasted with electrical synapses in which the ionic currents flowing into a presynaptic cell can cross the barrier of the two cell membranes and enter a postsynaptic cell.
[0033] The physiological mechanism of the action potential is mediated by ion movement across the cell membrane. Neurons actively maintain a resting membrane potential via ion pumps that facilitate movement of ions such as Na.sup.+, K.sup.+, and Cl.sup.-through the neuronal membrane. Different types of neurons may maintain different resting potentials, e.g., -75 mV to -55 mV. An action potential is generated by an influx of ions, i.e., a movement of charge to generate a large deviation in the membrane potential that is associated with a temporary rise in voltage across the membrane, e.g., a rise to a membrane potential in a range of 30-60 mV. The action potential in an individual neuron may be initiated in response to the neurotransmitter release from a presynaptic (e.g., upstream) neuron, which in turn results in receptor binding at the postsynaptic neuron and a cascade of events which leads to the influx of ions and membrane depolarization that results in an action potential that is propagated through the nerve.
[0034] Synapses may be located at the junction between two neurons, which permits an action potential to be propagated from one nerve cell to another. However, axon terminals may also form synapses at the junctions between neurons and non-neuronal cells, such as with immune cells and a neuroimmune junction, muscle cells at a neuromuscular junction or gland cells. Release of neurotransmitters into the synaptic cleft and binding to receptors in the postsynaptic membrane of a postsynaptic cell results in downstream effects that are dependent on the nature of the presynaptic neuron and the specific neurotransmitters released as well as the nature of the postsynaptic cell, e.g., the type of available receptors. In addition, the post-synaptic effect of an action potential may be excitatory or inhibitory. An excitatory postsynaptic potential is a postsynaptic potential that makes the postsynaptic neuron more likely to fire an action potential while an inhibitory postsynaptic potential is a postsynaptic potential that makes the postsynaptic neuron less likely to fire an action potential. Further, several neurons may work together to release neurotransmitters in concert that trigger downstream action potentials or inhibit the triggering of downstream action potentials. Neuromodulation is a technique in which energy from an external energy source is applied to certain areas of the nervous system to activate or increase the nerve or nerve function and/or block or decrease the nerve or nerve function. In certain neuromodulation techniques, one or more electrodes are applied at or near target nerves, and the application of energy is carried through the nerve (e.g., as an action potential) to cause a physiological response in areas of the body downstream of the energy application site. However, because the nervous system is complex, it is difficult to predict the scope and eventual endpoint of the physiological response for a given energy application site.
[0035] While strategies for ultrasound modulation of the central nervous system (i.e. brain tissue) have demonstrated successful modulation of neural activity, attempts to modulate peripheral nerves have lagged. For example, ultrasound modulation of the central nervous system (CNS) involves stimulation of cortical regions of the brain, which are rich in synaptic structures while attempts at ultrasound stimulation of peripheral nerves have targeted nerve trunks that are less rich in/devoid of synaptic structures. In the present technique, ultrasound stimulation of peripheral nerves involves targeting one or more axon terminals. In addition, instead of targeting traditional neuron-neuron synapses in peripheral tissue, where stimulation results in action potential propagation, in present techniques, one or more energy pulses are applied to the patient's internal tissue comprising axon terminals that include axoextracellular synapses and/or neuronal junctions with other cell types, e.g., at neuroimmune synapses, where stimulation of axonal end terminals releases neurotransmitter/neuropeptide or induces altered neurotransmitter release in the vicinity of neighboring non-neuronal cells such as immune or other cells and modulates cell activity.
[0036] Benefits of the present techniques include local modulation at the target area of the tissue. This local modulation was demonstrated relative to other modulation techniques, such as electrical stimulation via electrodes. For example, the present techniques were examined relative to electrical stimulation, and changes in contralateral tissue were not observed for the focused and local energy application.
[0037] Provided herein are techniques for neuromodulation in which energy from an energy source (e.g., an external or extracorporeal energy source) is applied to axon terminals in a manner such that neurotransmitter release at the site of focus of the energy application, e.g., the axon terminals, is triggered in response to the energy application and not in response to an action potential. That is, the application of energy directly to the axon terminals acts in lieu of an action potential to facilitate neurotransmitter release into a neuroimmune junction or other neuronal junction with a non-neuronal cell. The application of energy directly to the axon terminals further induces an altered neurotransmitter release from the axon terminal within, e.g., an axoextracellular synapse into the vicinity of neighboring non-neuronal cells. In one embodiment, the energy source is an extracorporeal energy source, such as an ultrasound or electromagnetic energy source. In this manner, non-invasive and targeted neuromodulation may be achieved directly at the site of energy focus rather than via modulation at an upstream site that in turn triggers an action potential to activate downstream targets. However, it should be understood that modulation is complex and energy application to organ tissue that includes target neuronal junctions may also trigger downstream effects.
[0038] The present techniques may be used in conjunction with lymph node neuromodulation or modulation of the lymphocyte retention neuro-immune reflex in addition to modulation of any neuroimmune interfaces (e.g., the junction or synapse between an axon terminal and an immune cell). In addition, direct modulation of immune cells themselves is also contemplated, e.g., immune cells that are not part of a junction with a neuron. While certain embodiments of the disclosure are presented in the context of neuroimmune modulation, it should be understood that the disclosed techniques may be used in conjunction with other target tissues and with other types of non-neuronal cells. As provided herein, non-neuronal cells may include immune cells, muscular cells, secretory cells, etc.
[0039] In certain embodiments, the target tissues are internal tissues or organs that are difficult to access using electrical stimulation techniques. Other contemplated tissue targets include gastrointestinal tissue (stomach, intestines), muscle tissue, epithelial tissue, connective tissue, cardiac tissue, secretory tissue, etc. In one example, focused application of energy at a neuromuscular junction facilitates neurotransmitter release at the neuromuscular junction without an upstream action potential. Contemplated modulation targets may include portions of the spleen responsible for controlling TNF-alpha release from macrophages, a site in an adrenal gland for controlling dopamine release, or local sites within a mesenteric plexus controlling inflammation and macrophage function in gut. In addition, neuroimmune interfaces that may control antibody production or the functional state of lymphocytes may be modulated via ultrasound or magnetic energy.
[0040] To that end, the disclosed neuromodulation techniques may be used in conjunction with a neuromodulation system. FIG. 1 is a schematic representation of a system 10 for neuromodulation to achieve neurotransmitter release into a synapse in response to an application of energy. The depicted system includes a pulse generator 14 coupled to an energy application device 12 (e.g., an ultrasound transducer, or a magnetic coil). The energy application device 12 is configured to receive energy pulses, e.g., via leads, that in use are directed to a target tissue of the patient, which in turn results in a clinical effect at the site of energy focus. In certain embodiments, the pulse generator 14 and/or the energy application device 12 may be implanted at a biocompatible site (e.g., the abdomen), and the lead or leads couple the energy application device 12 and the pulse generator 14 internally. For example, the energy application device 12 may be a MEMS transducer, such as a capacitive micromachined ultrasound transducer.
[0041] In certain embodiments, the energy application device 12 and/or the pulse generator 14 may communicate wirelessly, for example with a controller 16 that may in turn provide instructions to the pulse generator 14. In other embodiments, the pulse generator 14 may be an extracorporeal device, e.g., may operate to apply energy transdermally or in a noninvasive manner from a position outside of a patient's body, and may, in certain embodiments, be integrated within the controller 16. In embodiments in which the pulse generator 14 is extracorporeal, the energy application device 12 may be operated by a caregiver and positioned at a spot on or above a patient's skin such that the energy pulses are delivered transdermally to a desired internal tissue. Once positioned to apply energy pulses to the desired site, the system 10 may initiate neuromodulation to achieve desired clinical effects.
[0042] In certain embodiments, the system 10 may include an assessment device 20 that is coupled to the controller 16 and that assesses proxy characteristics that are indicative of whether the modulation goals have been achieved. In one embodiment, the modulation goal may be local. For example, the modulation may result in local tissue or function changes, such as tissue structure changes, increased drainage, etc. In embodiments in which the energy is applied to a neuroimmune junction, the modulation may also result in immune function changes, such as a change in a population of immune cells or a change in a presence or concentration of chemical compounds by the lymphatic tissue. Based on the assessment, the modulation parameters of the controller 16 may be altered. For example, if a successful modulation is associated with a decrease in norepinephrine or tumor necrosis factor concentration within a defined time window relative to the start of the procedure (e.g., 5 minutes, 30 minutes), a change may be desired in the frequency or voltage or other parameters, which in turn may be provided by an operator to the controller 16 for defining the energy pulses of the pulse generator 14.
[0043] The system 10 as provided herein may provide energy pulses according to various modulation parameters. For example, the modulation parameters may include various stimulation time patterns, ranging from continuous to intermittent. With intermittent stimulation, energy or energy pulses delivered for a period of time at a certain frequency during the signal-on time. The signal-on time is followed by a period of time with no energy delivery, referred to as signal-off time. The modulation parameters may also include stimulation application frequency and duration. The application frequency may be continuous or delivered at various time periods within the day or week. The treatment duration may last for as little as a few minutes to as long as several hours. Treatment duration with a specified stimulation pattern may last for one hour, repeated at, e.g., 12 hour intervals. Alternatively, treatment may be delivered at a higher frequency, say every three hours, for shorter durations, for example, 30 minutes. The treatment duration and frequency can be tailored to achieve the desired result.
[0044] FIG. 2 is a block diagram of certain components of the system 10. As provided herein, the system 10 for neuromodulation may include a pulse generator 14 that is adapted to generate a plurality of energy pulses for application to a tissue of a patient. The pulse generator 14 may be separate or may be integrated into an external device, such as a controller 16. The controller 16 includes a processor 30 for controlling the device. Software code or instructions are stored in memory 32 of the controller 16 for execution by the processor 30 to control the various components of the device. The controller 16 and/or the pulse generator 14 may be connected to the energy application device 12 via one or more leads 33.
[0045] The controller 16 also includes a user interface with input/output circuitry 34 and a display 36 that are adapted to allow a clinician to provide selection inputs or stimulation parameters to stimulation programs. Each stimulation program may include one or more sets of modulation parameters including pulse amplitude, pulse width, pulse frequency, etc. The pulse generator 14 modifies its internal parameters in response to the control signals from controller device 16 to vary the stimulation characteristics of energy pulses transmitted through lead 33 to the patient. Any suitable type of pulse generating circuitry may be employed including constant current, constant voltage, multiple-independent current or voltage sources, etc. The energy applied is a function of the current amplitude and pulse width duration.
[0046] In one embodiment, the memory 32 stores different operating modes that are selectable by the operator. For example, the stored operating modes may include instructions for executing a set of modulation parameters associated with a particular treatment site. Different sites may have different associated modulation parameters. Rather than having the operator manually input the modes, the controller 16 may be configured to execute the appropriate instruction based on the selection. In another embodiment, the memory 32 stores operating modes for different types of treatment. For example, activation may be associated with a different stimulating voltage or frequency range relative to those associated with depressing or blocking tissue function. In a specific example, the frequencies are in the range of between 10 mW/cm.sup.2-3 W/cm.sup.2 and Positive Pressure Peak range between 0.5 MPa to 7 MPa. The selected frequencies may depend on the mode of energy application, e.g., ultrasound or magnetic energy.
[0047] In another embodiment, the memory 32 stores a calibration or setting mode that permits adjustment or modification of the modulation parameters to achieve a desired result. In one example, the stimulation starts at a lower energy parameter and increases incrementally, either automatically or upon receipt of an operator input. In this manner, the operator may observe the effects as the modulation parameters are being changed.
[0048] The controller 16 may also be configured to receive inputs related to immune and lymphatic function as an input to the selection of the modulation parameters. For example, when an imaging modality is used to assess a tissue characteristic, the controller 16 may be configured to receive a calculated index or parameter of the characteristic. Based on whether the index or parameter is above or below a threshold, the modulation parameters may be modified.
[0049] In another implementation, a successful modulation parameter set may also be stored by the controller 16. In this manner, patient-specific parameters may be determined. Further, the effectiveness of such parameters may be assessed over time. If a particular set of parameters is less effective over time, the patient may be developing insensitivity to activated pathways. If the system 10 includes an assessment device 20, the assessment device may provide feedback to the controller 16. Based on the feedback, the processor 16 may automatically alter the modulation parameters (e.g., step up the frequency or pulse width).
[0050] The system may also include an imaging device 38 that facilitates focusing the energy application device 12. In one embodiment, the imaging device 38 may be integrated with the energy application device 12 such that different ultrasound energies are applied for targeting and subsequently neuromodulation.
[0051] FIG. 3 is a specific example in which the energy application device 12 includes an ultrasound transducer 42 that is capable of applying energy, shown by way of example at a spleen. The energy application device 12 may include control circuitry for controlling the ultrasound transducer 42. The ultrasound transducer 42 may also be configured to acquire image data to assist with focusing the applied energy on a desired target location.
[0052] The desired target may be an internal tissue or organ that includes axon end terminals that can be stimulated by direct application of energy to the axon terminals within a field of focus of the energy application device 12 to release neurotransmitters. The energy may be focused on only part of the internal tissue, e.g., less than 50% of the total volume of the tissue, such that only axon terminals in the portions of the tissue would directly receive the energy and release neurotransmitters while the unstimulated axon terminals outside of the focus area do not. In some embodiments, axon terminals in the portions of the tissue directly receiving the energy would induce altered neurotransmitters release. In this manner, tissue compartments may be targeted for neuromodulation in a granular manner. In certain embodiments, the energy may be focused to a volume of less than 25 mm.sup.3. The focal volume and focal depth may be influenced by the size/configuration of the energy application device 12. The focal volume of the energy application may be defined by the field of focus of the energy application device 12.
[0053] The disclosed techniques may be used in assessment of neuromodulation effects, which in turn may be used as an input to selecting or modifying neuromodulation parameters. The disclosed techniques may use direct assessments of tissue condition or function. The assessment may occur before (i.e., baseline assessment), during, and/or after the neuromodulation. For example, for a patient in need of increase lymphatic drainage, such drainage may be monitored before, during and/or after neuromodulation to determine if the selected parameters have achieved a sufficient increase in the drainage. Accordingly, lymphatic drainage may be assessed by one or more in vivo techniques that determine lymphatic drainage. In one embodiment, an exogenous contrast agent is administered either directly into the lymphatic tissue or indirectly via intradermal injection. For example, a gadolinium-based contrast media may be used. Either local or systemic flow may be addressed, depending on the desired clinical outcome. For example, MR lymphangiography may be used to assess lymphatic drainage in the limbs. In another embodiment, fluorescence microlymphangiography (FML) may also be used to assess lymphatic drainage. FML employs the intradermal administration of a fluorescent dye, FITC conjugated to dextran (FITC-dextran), and video fluorescence microscopy techniques to acquire high-resolution images. In another embodiment, quantum-dot optical lymphatic imaging may be used for in vivo lymphatic imaging and lymphatic flow assessment. In yet another embodiment, imaging may include dyes or indicators that target lymph-specific markers, such as LYVE-1, Prox-1, podoplanin, and VEGFR3.
[0054] The images from the assessment techniques may be received by the system for automatic or manual assessment. Based on the image data, the modulation parameters may also be modified. If the lymphatic drainage has increased in the presence of stable vital signs and other health indicators, the stimulation frequency or voltage may be stepped back to the lowest energy that maintains the desired elevated lymphatic drainage. In other embodiments, the change in lymphatic drainage is utilized as a marker of local neurotransmitter concentration, and used as a surrogate marker for exposure of local immune (immune interacting) cells to phenotype modulating neurotransmitters, and effectively a marker of the predicted effect on immune function. The local concentration may refer to concentration within a field of focus of the energy application.
[0055] Additionally or alternatively, the system may assess the presence or concentration of neurotransmitters or cells in the lymph tissue or lymphatic fluid. Lymphatic fluid or tissue may be acquired by a fine needle aspirate, and the assessment of the presence or levels of neurotransmitters (e.g., peptide transmitters, catecholamines) may be performed by any suitable technique.
[0056] In another embodiment, a change in the types and/or numbers of cells in the lymph node or lymphatic tissue may be an indication of lymphatic tissue function. The cell population may be assessed by ex vivo techniques, such as flow cytometry. In another example, the lymphatic cell population may be assessed by laser-scanning in vivo confocal microscopy (IVCM) using endogenous contrast.
[0057] FIG. 4 is a flow diagram of a method 50 for stimulating tissue. In the method, the energy application device is positioned such that the energy pulses are focused at the desired internal tissue location at step 52, and the pulse generator applies a plurality of energy pulses to the internal tissue to stimulate the target axon terminals to release neurotransmitters and/or induce altered neurotransmitter release at step 54. Then, the effect of the stimulation is assessed at step 56. For example, one or more direct or indirect assessments of a state of tissue function or condition may be used. Based on the tissue function as assessed, the modulation parameters of the one or more energy pulses may be modified at step 58 to achieve the desired clinical result.
[0058] In one embodiment, assessments may be performed before and after stimulation to assess a change in lymphatic function as a result of the stimulation. If a desired change in the state of the assessed characteristic of lymphatic function is above or below a threshold, appropriate modification in the modulation parameters may be made. For example, if the change in the characteristic relative to the threshold is associated with successful activation of the lymph tissue, the energy applied during neuromodulation may be stepped back to the minimum level that supports the desired outcome. If the change in the characteristic relative to the threshold is associated with insufficient activation of the lymph tissue, certain modulation parameters, such as the modulation voltage or frequency, the pulse shape, the stimulation pattern, and/or the stimulation location may be changed. It should also be understood that certain desired clinical outcomes may be instead associated with blocking activation. In such embodiments, an assessment of decreased neural and/or lymphatic function is associated with maintaining the modulation parameters, and the modulation parameters may be modified if an undesired level of lymphatic activity persists.
[0059] Further, the assessed characteristic or condition may be a value or index (e.g., a flow rate, a concentration, a cell population), which in turn may be analyzed by any suitable technique. For example, a relative change exceeding a threshold may be used to determine if the modulation parameters are modified. The successful modulation may be assessed via a measured clinical outcome, such as a presence or absence of an increase in tissue structure size (e.g., lymph node size) or a change in concentration of released molecules e.g., relative to the baseline concentration before the neuromodulation). In one embodiment, a successful modulation may involve an increase in concentration above a threshold, e.g., above a 50%, 100%, 200%, 400%, 1000% increase in concentration relative to baseline. For blocking treatments, the assessment may involve tracking a decrease in concentration of a molecule over time, e.g., at least a 10%, 20%, 30%, 50%, or 75% decrease in the molecule of interest. Further, for certain subjects, the successful blocking treatment may involve keeping a relatively steady concentration of a particular molecule in the context of other clinical events that may tend to increase the concentration of the molecule. That is, successful blocking may block a potential increase. The increase or decrease or other observable effect may be measured within a certain time window from the start of treatment, e.g., within 5 minutes, within 30 minutes. In certain embodiments, if the neuromodulation is determined to be successful, the change in the neuromodulation is an instruction to stop applying energy pulses. In another embodiment, one parameter of the neuromodulation is changed if the neuromodulation is not successful. For example, the change in modulation parameters may be an increase in modulation frequency, such as a stepwise increase in frequency of 10-100 Hz and assessment of the desired characteristic until successful neuromodulation is achieved. In another implementation, the pulse width may be changed. In other embodiments, two or more of the parameters may be changed together. If the neuromodulation is not successful after multiple parameter changes, the focus of energy application may be changed.
[0060] Provided herein are techniques for neuromodulation based on direct and focused stimulation of lymphatic tissue, e.g., lymph node tissue, spleen tissue, liver tissue, etc. Neuromodulation of lymph tissue may alter the drainage rate and/or the population of cells in the drained fluid. Due to co-localization of nerves innervating the immune cell and lymphatic vessel compartments of the lymph node, neurotransmitter release may have a simultaneous effect on both lymphatic and immune function; therefore, the observable changes in lymphatic function (i.e. easily observable size and/or flow change in lymphatic tissue using non-invasive imaging technology) may be used as a surrogate measure of immune cell neuromodulation (i.e. the simultaneous changes in immune cell phenotypes due to release of the local neurotransmitters).
[0061] In one embodiment, the invention provides a method of neuromodulation, comprising:
[0062] focusing an electromagnetic energy source on an internal tissue field in a patient in need of neuromodulation, the internal tissue field comprising one or more neurons, wherein the electromagnetic energy source is not in direct contact with the tissue field of focus; and
[0063] applying one or more energy pulses to the patient's internal tissue in the field of focus via the electromagnetic energy source to cause a change in activity in the patient's internal tissue, wherein the change is relative to a baseline activity.
[0064] The patient's internal tissue can be, for example, a peripheral tissue, the spleen, or a lymph node.
[0065] In one embodiment, the baseline is indicative of the activity of the internal tissue before applying the one or more energy pulses. In one embodiment, the time at which the one or more energy pulses are applied is designated as time zero, and the baseline is indicative of the activity at a time immediately before the energy pulses are applied.
[0066] The change in activity can comprise, for example, an alteration in neurotransmitter release in the internal tissue, or a decrease in a concentration or amount of neurotransmitters released in the internal tissue, or an increase in a concentration or amount of neurotransmitters released in the internal tissue.
[0067] The change in activity can comprise, for example, a change in a level of cytokine release in the internal tissue, a change in a level of a cytokine in the patient's blood, and/or a decrease or increase in a level of a cytokine in the patient's blood. The cytokine can be, for example, one or more of tumor necrosis factor alpha (TNF-alpha), interleukin-1 (IL-1), IL-6 and high mobility group box 1 protein (HMGB1).
[0068] The electromagnetic energy source can be extracorporeal and the one or more energy sources applied transdermally. In a preferred embodiment, the electromagnetic energy source generates an electromagnetic field.
[0069] The patient can have, for example, one or more of endotoxemia, sepsis, septicemia, septic shock, inflammation, and a pathogenic consequence of an inflammatory condition or an inflammatory cytokine cascade.
[0070] The invention also provides a method of immunomodulation, comprising:
[0071] controlling an electromagnetic energy source to apply one or more energy pulses to a patient's internal immune tissue and according to one or more parameters of the energy pulses to cause a change in a level of cytokine release in the internal tissue in direct response to the one or more energy pulses;
[0072] receiving information related to the release of the cytokine relative to a baseline before the one or more energy pulses are applied; and
[0073] changing the one or more parameters based on the information.
[0074] The cytokine can be, for example, one or more of TNF-alpha, IL-1, IL-6 and HMGB1.
[0075] The electromagnetic energy source can be configured to be positioned on or above a patient's skin.
[0076] The patient's internal immune tissue can be, for example the spleen or a lymph node.
[0077] The patient can have, for example, one or more of endotoxemia, sepsis, septicemia, septic shock, inflammation, and a pathogenic consequence of an inflammatory condition or an inflammatory cytokine cascade.
[0078] The invention further provides a method of neuromodulation, comprising: positioning an electromagnetic field generator at a location at which the electromagnetic field generator is capable of stimulating at least one neuron in a lymphatic tissue; and
[0079] applying one or more energy pulses to the lymphatic tissue via the electromagnetic field generator to alter activity in the at least one neuron in response to the one or more energy pulses to modulate the lymphatic tissue.
[0080] Preferably, the lymphatic tissue is the spleen or a lymph node.
[0081] Preferably, the energy pulses are applied non-invasively.
[0082] The invention further provides a method of treating an infection and/or an inflammation in a subject comprising applying ultrasound and/or electromagnetic stimulation to one or more of lymph nodes and/or spleen of the subject in an amount effective to increase the number of lymphocytes in a tissue and/or decrease serum levels of one or more proinflammatory cytokines in a subject, thereby treating an infection and/or an inflammation in the subject, wherein the ultrasound and/or electromagnetic stimulation is applied by inserting an ultrasound transducer and/or an electromagnetic coil through vasculature of the subject or through a small incision so that the transducer and/or electromagnetic coil is adjacent to the spleen or lymph nodes.
[0083] The proinflammatory cytokines can comprise, for example, one or more of tumor necrosis factor alpha and interleukin-1.
[0084] Preferably, the ultrasound and/or electromagnetic stimulation is effective to modulate neurotransmitter release at the level of the spleen and/or lymph nodes.
[0085] In regard to electromagnetic stimulation, electric current flowing through a magnetic coil results in the generation of a magnetic field. A time varying magnetic field can be used to induce an electric current in the tissue. By focusing the coil, the magnetic field can be targeted at a selective site, leading to stimulation of the neural circuits in the specific target area. Using magnetic stimulation, it is therefore possible to activate the neural circuits in vivo in a noninvasive manner. Advantages of this technique include that it is noninvasive, painless and independent of clothing and bone/tissue structure. MagPro R30 is an example of a stimulation and Coil MC-B35 is an example of a coil that can be used.
EXAMPLES
[0086] Provided herein are techniques that may be applied to "neuroimmune synapses" or "neuroimmune interfaces." The depicted examples are directed to application of energy to lymph nodes that are proximate to the exit lymphatic vessels on lymph nodes. These nerves may alter the transit of lymphocytes through the lymph node and when stimulated result in increased lymphocyte retention (i.e. more lymphocytes in the lymph node). One molecular mechanism for decreasing lymphocyte egress from the exit lymphatic vessels is modulation of the ccr7 chemoreceptor activity through coupling and signaling through a beta-adrenergic receptor.
[0087] Nerves from the sciatic nerve entering the popliteal lymph node were stimulated in a rat. A bipolar electrode was utilized to stimulate for 5 minutes at a range of applied current using a 20 Hz stimulation frequency with 200 microsecond (balanced, biphasic pulses) After stimulation, the lymph node was excised, dissociated into a single cells suspension, and then analyzed using a Hemavet cell counter. The number of lymphocyte cells per cubic mL of dissociated tissue was plotted for the directly stimulated popliteal lymph node, the contralateral popliteal lymph node, and the axillary lymph node. Lymphocyte numbers were statistically increased only in the stimulated lymph node. Results are shown in FIG. 5
[0088] In contrast to the electrical stimulation, cells and nerve terminals within the popliteal lymph node were stimulated by exposing the lymph node to an ultrasound transducer. Ultrasound parameters used were as follows: [0089] Frequency: 1.1 MHz [0090] Burst Cycles: 150 [0091] Burst Period: 500 .mu.s (duty cycle is close to 30%) [0092] Time of Burst on: 1 min [0093] Estimated Positive Pressure: 5 MPa [0094] Estimated Negative Pressure: -2 MPa [0095] Transducer Geometry: Spherically focused with an opening in the middle [0096] O.D. --70 mm [0097] Hole diameter .about.20 mm [0098] Focus distance .about.54.8 mm (F number .about.=1) [0099] Focus diameter 1.8 mm [0100] Focus length 11.7 mm.
[0101] After stimulation the stimulated (and other) lymph nodes were excised and examined using a cell counter. Only the stimulated lymph node showed a lymphocyte increase as shown in FIG. 6. Neurotransmitter levels in the ultrasound stimulated lymph node differed relative to the electrically stimulated lymph node. However, the cell count data for lymphocytes was similar in both the ultrasound and electrically stimulated lymph nodes.
[0102] In addition to local modulation (through efferent nerve fibers) of lymphocyte egress, electrical stimulation of the nerves entering the lymph node was found to have distal effects on immune cell concentrations in the liver and spleen.
[0103] FIG. 7 shows results for neurotransmitter concentration in ultrasound stimulated lymph nodes. Epinephrine and dopamine went up in ultrasound stimulated lymph nodes while norepinephrine went down after ultrasound stimulation. FIG. 8 shows results from ultrasound stimulation of the spleen. Results were collected 15 minutes after a 1 minute ultrasound stimulation.
[0104] FIG. 8 shows results for norepinephrine, epinephrine, dopamine and acetylcholine concentrations in ultrasound stimulated spleens. FIGS. 9A and 9B show results of acetylcholine concentration in the spleen for ultrasound-stimulated animals relative to a control group of unstimulated animals. FIG. 9C shows acetylcholine concentration in the spleen for a control group of antigen-treated animals vs. ultrasound-stimulated and antigen-treated animals. FIGS. 10A-C show TNF-alpha concentrations for various experimental and control groups. FIG. 10A shows results from ultrasound stimulation in the spleen, FIG. 10B shows results from the serum, and FIG. 10C shows results from the liver for a control group of antigen-treated animals vs. ultrasound-stimulated and antigen-treated animals. FIGS. 11A-11B show schematic images of example stimulators.
[0105] In addition to ultrasound energy, the effect of noninvasive electromagnetic stimulation on local tissue was also examined. In the technique, a magnetic field was generated via an electric current applied through a magnetic coil. The magnetic energy technique may be associated with certain advantages relative to electrode stimulation. For example, the technique is noninvasive, painless, and independent of clothing and bone/tissue structure.
[0106] Mice were anesthetized (ketamine [100 mg/kg, i.m.] and xylazine [10 mg/kg, i.m.]) and placed in supine position. Mice were then subjected to transcutaneous electromagnetic stimulation (EMS) at the cervical region at increasing amplitude (5, 50, 100%) at 2 Hz frequency (total 120 cycles). Change in amplitude is proportional to the amount of current delivered. One group of mice did not receive stimulation (0% amplitude) and served as the sham control. Two hours after stimulation, animals were challenged with endotoxin to induce endotoxemia (where endotoxemia is a model of biological threat attack or of hemorrhagic shock or of a condition is which the inflammatory cytokine cascade is active). Serum samples were collected after 90 min and levels of TNF were assessed by ELISA.
[0107] FIG. 12 shows the results of electromagnetic stimulation on serum TNF levels. The results show that transcutaneous electromagnetic stimulation attenuates serum TNF levels in endotoxemia-challenged mice. Mice were subjected to electromagnetic stimulation at the cervical region for 1 min (50% amplitude, 2 Hz frequency, total 120 cycles). The control group did not receive any stimulation. After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and blood was collected after 90 min. Serum TNF levels were determined by ELISA.
[0108] FIG. 13 shows the results of electromagnetic stimulation on serum TNF levels in response to endotoxin challenge. The results show that transcutaneous electromagnetic stimulation attenuates serum TNF levels in a dose-dependent manner in endotoxemic mice. Mice were subjected to electromagnetic stimulation at the cervical region (1 min--0, 5, 50% amplitude and 6 sec--100% amplitude, 2 Hz frequency, total 120 cycles). After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and blood was collected after 90 min. Serum TNF levels were determined by ELISA.
[0109] FIG. 14 shows the results of electromagnetic stimulation on serum TNF levels. The results show that transcutaneous electromagnetic stimulation attenuates serum TNF levels in dose dependent manner in endotoxemic mice. Mice were subjected to electromagnetic stimulation at the cervical region (1 min--0, 12, 25, 50% amplitude, 2 Hz frequency, total 120 cycles). After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and blood was collected after 90 min. Serum TNF levels were determined by ELISA.
[0110] Another set of experiments examined the effect of noninvasive electromagnetic energy on mice that had previously been subjected to left vagotomy or sham surgery. After 7 days of recovery, mice were subjected to transcutaneous electromagnetic stimulation or sham stimulation at the cervical region (50% amplitude, 2 Hz frequency, total 120 cycles). Two hours after stimulation, animals were challenged with endotoxin to induce endotoxemia. Serum samples were collected after 90 min and levels of TNF were assessed by ELISA.
[0111] FIG. 15 shows the results of electromagnetic stimulation on serum TNF levels. The results show that vagotomy attenuates the effect of subsequent transcutaneous electromagnetic stimulation. Mice were subjected to left cervical vagotomy or sham vagotomy. After 7 days, EMS was carried out at the cervical region (1 min--0, 50% amplitude, 2 Hz frequency, total 120 cycles). After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and blood was collected after 90 min. Serum TNF levels were determined by ELISA.
[0112] In an additional study to analyze whether localized transcutaneous electromagnetic stimulation attenuates systemic and/or local immune responses, mice were anesthetized (ketamine [100 mg/kg, i.m.] and xylazine [10 mg/kg, i.m.]). Anesthetized mice were placed in supine position. Mice were subjected to transcutaneous electromagnetic stimulation (EMS) targeted at the spleen tissue field at 50% amplitude at 2 Hz frequency (total 120 cycles). One group of mice did not receive stimulation (0% amplitude) and served as the sham control. Two hours after stimulation, animals were challenged with endotoxin to induce endotoxemia (where endotoxemia is a model of biological threat attack and/or hemorrhagic shock and/or a condition in which the inflammatory cytokine cascade is active). Serum and spleen samples were collected after 90 min and levels of TNF were assessed by ELISA.
[0113] FIG. 16 shows the results of electromagnetic stimulation on serum TNF levels. The results show that local transcutaneous electromagnetic stimulation attenuates serum TNF levels in endotoxemic mice. Mice were subjected to electromagnetic stimulation at the spleen region (1 min--0, 50% amplitude, 2 Hz frequency, total 120 cycles). After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and blood was collected after 90 min. Serum TNF levels were determined by ELISA.
[0114] FIG. 17 shows the results of electromagnetic stimulation on serum TNF levels. The results show that local transcutaneous electromagnetic stimulation attenuates local TNF levels in endotoxemic mice. Mice were subjected to electromagnetic stimulation at the spleen region (1 min--0, 50% amplitude, 2 Hz frequency, total 120 cycles). After 2 hrs, mice were subjected to endotoxemia (10 mg/kg endotoxin, i.p.), and spleen was collected after 90 min. Serum TNF levels were determined by ELISA.
[0115] This written description uses examples to disclose the invention and also to enable any person skilled in the art to practice the invention, including making and using any devices or systems and performing any incorporated methods. The patentable scope of the invention is defined by the claims, and may include other examples that occur to those skilled in the art. Such other examples are intended to be within the scope of the claims if they have structural elements that do not differ from the literal language of the claims, or if they include equivalent structural elements with insubstantial differences from the literal languages of the claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.