Plating Solution And Method For Producing Plated Product
NAWAFUNE; Hidemi ; et al.
U.S. patent application number 15/576892 was filed with the patent office on 2020-02-06 for plating solution and method for producing plated product. The applicant listed for this patent is FUSO COMPANY, LTD., TECHNO ROLL CO., LTD.. Invention is credited to Yoshiyuki KAMEGAWA, Toshikazu MURATA, Hidemi NAWAFUNE, Hiroshi NISHIWAKI, Hidehiro YOSHIOKA.
| Application Number | 20200040477 15/576892 |
| Document ID | / |
| Family ID | 63107327 |
| Filed Date | 2020-02-06 |








| United States Patent Application | 20200040477 |
| Kind Code | A1 |
| NAWAFUNE; Hidemi ; et al. | February 6, 2020 |
PLATING SOLUTION AND METHOD FOR PRODUCING PLATED PRODUCT
Abstract
The present invention provides a plating solution containing chromium sulfate and formic acid at a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less and a concentration of formic acid of 0.05 mol/L or more and 0.2 mol/L or less.
| Inventors: | NAWAFUNE; Hidemi; (Osaka, JP) ; NISHIWAKI; Hiroshi; (Osaka, JP) ; MURATA; Toshikazu; (Hyogo, JP) ; YOSHIOKA; Hidehiro; (Hyogo, JP) ; KAMEGAWA; Yoshiyuki; (Osaka, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 63107327 | ||||||||||
| Appl. No.: | 15/576892 | ||||||||||
| Filed: | August 25, 2017 | ||||||||||
| PCT Filed: | August 25, 2017 | ||||||||||
| PCT NO: | PCT/JP2017/030445 | ||||||||||
| 371 Date: | November 27, 2017 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C25D 3/10 20130101; C25D 3/06 20130101 |
| International Class: | C25D 3/06 20060101 C25D003/06; C25D 3/10 20060101 C25D003/10 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 8, 2017 | JP | 2017-021006 |
Claims
1. A plating solution used for trivalent chromium plating, the plating solution comprising chromium sulfate and formic acid at a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less and a concentration of formic acid of 0.05 mol/L or more and 0.2 mol/L or less.
2. The plating solution according to claim 1 used for trivalent chromium plating with a plating thickness of 5 .mu.m or more.
3. A method for producing a plated product, the method comprising a plating step of performing electric plating in a plating bath that contains a plating solution comprising chromium sulfate so as to produce the plated product to which trivalent chromium plating is applied by the plating step, wherein the plating solution used in the plating step has a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less, the plating bath has a temperature of 20.degree. C. or more and less than 40.degree. C., and the electric plating is performed at a current density of 2 A/dm.sup.2 or more and 20 A/dm.sup.2 or less.
Description
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application claims priority to Japanese Patent Application No. 2017-21006, the disclosure of which is incorporated herein by reference in its entirety.
FIELD
[0002] The present invention relates to a plating solution and a method for producing a plated product, more specifically, to a plating solution used for trivalent chromium plating and a method for producing a plated product by applying trivalent chromium plating.
BACKGROUND
[0003] Conventionally, plated products obtained by applying chromium plating to metal products or plastic products are widely used. In producing the plated products of this type, the cases of using plating solutions containing trivalent chromium, which is environmentally friendly, instead of plating solutions containing hexavalent chromium are increasing (see Patent Literature 1 below).
CITATION LIST
Patent Literature
[0004] Patent Literature 1: JP 2000-249340 A
SUMMARY
Technical Problem
[0005] There has been a strong demand for employing trivalent chromium plating instead of hexavalent chromium plating due to a recent increase in environmental awareness. However, the cases of employing trivalent chromium plating are limited in the current situation since the covering power on the product serving as an object to be plated is poorer than in the cases of employing hexavalent chromium plating. Further, it is difficult to obtain a linear relationship between the current density and the thickness of plating deposition and to exert throwing power in trivalent chromium plating. Therefore, trivalent chromium plating is rarely employed in the cases of producing plated products having complex shapes such as dies and thick plated products, though there are some cases of employing trivalent chromium plating in decorative plating or the like where the plating thickness is several .mu.m. It is therefore an object of the present invention to provide a plating solution for trivalent chromium plating with which plated products equivalent to those produced by hexavalent chromium plating can be produced, so as to enlarge the application range of environmentally friendly plated products.
Solution to Problem
[0006] As a result of diligent studies in order to achieve the aforementioned object, the inventors have found that a plating solution containing chromium sulfate in which the concentration of Cr.sup.3+ ions is adjusted to a specific range exhibits a covering power equivalent to that of a plating solution containing hexavalent chromium, so as to accomplish the present invention.
[0007] That is, the present invention provides a plating solution used for trivalent chromium plating, the plating solution containing chromium sulfate and formic acid at a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less and a concentration of the formic acid of 0.05 mol/L or more and 0.2 mol/L or less.
[0008] Further, the present invention provides a method for producing a plated product, the method including a plating step of performing electric plating in a plating bath that contains a plating solution containing chromium sulfate, so as to produce the plated product to which trivalent chromium plating is applied by the plating step, wherein the plating solution used in the plating step has a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less, the plating bath has a temperature of 20.degree. C. or more and less than 40.degree. C., and the electric plating is performed at a current density of 2 A/dm.sup.2 or more and 20 A/dm.sup.2 or less.
BRIEF DESCRIPTION OF DRAWINGS
[0009] FIG. 1 is a relationship graph between current density and plating thickness in hexavalent chromium plating and trivalent chromium plating disclosed in general literatures or the like.
[0010] FIG. 2 is a graph showing the relationship between current density and plating thickness in trivalent chromium plating using the plating solution of the present invention.
[0011] FIG. 3 is a graph showing the results of a Hull cell test using a plating solution of an embodiment.
[0012] FIG. 4 is a graph showing the measurement results of plating thickness using a fluorescent x-ray film thickness meter.
[0013] FIG. 5 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
[0014] FIG. 6 is a graph showing the measurement results of plating thickness using a fluorescent x-ray film thickness meter.
[0015] FIG. 7 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
[0016] FIG. 8 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
[0017] FIG. 9 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
[0018] FIG. 10 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
[0019] FIG. 11 is a graph showing the results of a Hull cell test using a plating solution of another embodiment.
DESCRIPTION OF EMBODIMENTS
[0020] Hereinafter, a preferable embodiment of the present invention will be described. First, a method for producing a plated product will be described. In the method for producing a plated product of this embodiment, a pretreatment step of adjusting the surface properties of a product that serves as an object to be plated (which will be hereinafter referred to also as "original product") and a plating step of applying trivalent chromium plating to the pre-treated original product (which will be hereinafter referred to also as "pre-treated product") are performed. In the method for producing a plated product of this embodiment, a base plating step of applying base plating to the pre-treated product or an intermediate plating step of further applying intermediate plating to the pre-treated product to which base plating has been applied (which will be hereinafter referred to also as "base plated product") may be performed between the pretreatment step and the plating step, as needed. In such a case, trivalent chromium plating is performed as finish plating to the product to which the intermediate plating has been applied (which will be hereinafter referred to also as "intermediate plated product").
[0021] In the method for producing a plated product of this embodiment, chemical surface treatment, heat treatment, or the like may be further applied to the product to which trivalent chromium plating has been applied in the plating step. Further, coating such as clear coating may be applied to the plated product, as needed.
[0022] Examples of the original product to which the pretreatment step is applied include resin products, ceramic products, metal products, composite products combining resin parts and metal parts, and composite products obtained by coating metal parts with ceramics. Examples of the resins forming the original product include common thermoplastic resins and common thermosetting resins. The resins may be fiber reinforced plastic (FRP). Examples of the ceramics forming the original product include common ceramics mainly containing silicon oxide, aluminum oxide, or the like. Examples of the ceramics also include glassy materials such as enamels. Examples of the metals forming the original product include common metals such as iron and copper. The metals may be alloys. Examples of the pretreatment applied to the original product include polishing such as mechanical polishing, honing, and blasting, and degreasing such as alkali degreasing. In the base plating step and the intermediate plating step, various types of plating such as nickel plating, copper plating, and iron plating with a thickness of several .mu.m can be applied to the pre-treated product or the base plated product for purposes such as improving the aesthetic appearance and the corrosion resistance of the finish plated product.
[0023] In the plating step of applying trivalent chromium plating, electric plating is performed using the pre-treated product, the base plated product, the intermediate plated product, or the like as a workpiece. In the electric plating, trivalent chromium plating is applied to the workpiece using a plating bath that contains a plating solution containing chromium sulfate. Hereinafter, the plating solution used in the plating step will be described in detail.
[0024] For plating the workpiece with good covering power, it is important to use a plating solution having a concentration of Cr.sup.3+ ions of 0.1 mol/L or more and 1 mol/L or less as the plating solution in the plating step. The plating solution can contain a complexing agent, a pH buffer, a conductive agent, a surfactant, or the like, in addition to chromium sulfate as the main component. Examples of water that serves as the solvent of the plating solution include industrial water, tap water, deionized water, distilled water, and pure water.
[0025] As described above, the chromium sulfate is contained in the plating solution so that the concentration of Cr.sup.3+ ions in the plating bath is 0.1 mol/L or more and 1 mol/L or less. The chromium sulfate is preferably contained in the plating solution so that the concentration of Cr.sup.3+ ions in the plating bath is 0.1 mol/L or more and 0.3 mol/L or less. Use of such a preferable plating solution can give better covering power in trivalent chromium plating on the workpiece in the plating step.
[0026] Although it is also possible to partially replace the chromium sulfate contained in the plating solution of this embodiment as a supply source of the Cr.sup.3+ ions with one or more selected from the group consisting of chromium chloride, basic chromium sulfate, chrome alum, and chromium nitrate, there is a possibility of generating chlorine gas at the anode during plating when chromium chloride is contained in a large amount, and there is a possibility of reducing the current density during plating when chromium chloride is contained in a large amount. Accordingly, the ratio of chromium sulfate in the supply source of the Cr.sup.3+ ions in the plating solution is preferably 90 mol % or more. The ratio of chromium sulfate is more preferably 95 mol % or more, further preferably 99 mol % or more. It is particularly preferable that the supply source of the Cr.sup.3+ ions in the plating solution be substantially only chromium sulfate.
[0027] As the complexing agent contained in the plating solution of this embodiment, organic acids and salts thereof can be used. Examples of the organic acids include oxalic acid, citric acid, formic acid, maleic acid, malonic acid, tartaric acid, malic acid, acetic acid, phthalic acid, propionic acid, and ethylenediamine tetraacetic acid. Examples of the salts thereof include alkali metal salts such as lithium salts, potassium salts, and sodium salts, and alkaline earth metal salts such as magnesium salts and calcium salts. Among these, the formic acid is a particularly effective component as the complexing agent, and it is important that the formic acid be contained in the plating solution at a concentration of 0.05 mol/L or more and 0.2 mol/L or less. The concentration of the formic acid in the plating solution is more preferably 0.08 mol/L or more and 0.12 mol/L or less.
[0028] The aforementioned organic acids and the salts thereof exert at least a certain effect also as the pH buffer. Further, as the complexing agent, amino carbonyl compounds such as urea and carbamic acid also may be employed. In particular, urea functions not only as the pH buffer but also as a supply source of nitrogen to the plating film, which is effective in hardening the film. Further, urea can be expected to have an effect of suppressing generation of a precipitate such as chromium hydroxide in the plating solution. In view of such a point, the urea is preferably contained in the plating solution at a concentration of 0.1 mol/L or more and 1 mol/L or less. The concentration of the urea in the plating solution is more preferably 0.2 mol/L or more and 0.8 mol/L or less, particularly preferably 0.4 mol/L or more and 0.6 mol/L or less.
[0029] Other than the above, examples that are usable as the pH buffer include boric acid and borate. In the case where the boric acid is contained in the plating solution, the boric acid is generally contained in the plating solution at a concentration of 0.5 mol/L or more and 1 mol/L or less, though it also depends on the amount of the organic acids or the urea contained as the pH buffer. The pH of the plating solution is preferably adjusted to 1 or more and 2 or less, more preferably 1.3 or more and 1.7 or less, using the pH buffer or the like.
[0030] Examples of the conductive agent include ammonium chloride, sodium chloride, potassium chloride, ammonium sulfate, sodium sulfate, potassium sulfate, ammonium nitrate, sodium nitrate, and potassium nitrate. Further, examples of the surfactant include sodium lauryl sulfate, sodium dodecyl sulfate, polyethylene glycol, diisohexyl sulfosuccinate, 2-ethylhexyl sulfate, diisobutyl sulfosuccinate, diisoamyl sulfosuccinate, and isodecyl sulfosuccinate.
[0031] In the plating solution, various additives including a film forming agent such as polyethylene glycol, polyvinyl alcohol, and gelatin, and a defoamer can be contained as additional additives.
[0032] In the plating step using the plating solution containing such components, it is important to set the temperature of the plating bath to 20.degree. C. or more and less than 40.degree. C. The lower the temperature of the plating bath in the plating step, the better the covering power in trivalent chromium plating on the workpiece can be obtained. Meanwhile, when the temperature of the plating bath is a certain temperature or higher, the deposition of the components contained in the plating solution can be suppressed, and the occurrence of roughness on the plated surface can be reduced. In view of such a point, the temperature of the plating bath in the plating step is more preferably 23.degree. C. or more and 29.degree. C. or less, particularly preferably 24.degree. C. or more and 28.degree. C. or less. A plating film with uniform thickness and excellent surface brightness can be provided on the finish plated product by performing the plating step under such preferable temperature conditions.
[0033] In the electric plating in the plating step, it is important to set the current density to 2 A/dm.sup.2 or more and 20 A/dm.sup.2 or less. The current density is more preferably 2 A/dm.sup.2 or more and 15 A/dm.sup.2 or less, particularly preferably 2 A/dm.sup.2 or more and 13 A/dm.sup.2 or less. The covering power in trivalent chromium plating can be further enhanced by plating the workpiece at such a preferable current density.
[0034] In the plating step, air bubbles are generated in the plating solution due to the hydrogen gas. Therefore, in order to prevent adhesion of the air bubbles to the workpiece, the workpiece may be vibrated during the plating or may be subjected to bubbling to generate air bubbles by an inert gas or the like from below the workpiece.
[0035] Although the aforementioned various conditions in the plating step such as the concentration of the components of the plating solution, the bath temperature of the plating solution, and the current density applied to the workpiece are not always necessarily maintained within the aforementioned ranges during the period from the very start of the plating step to the completion of the plating step in order to exert the effects thereof, it is preferable that almost the same conditions as those at the start of the plating be maintained over the entire period.
[0036] In the plating step of this embodiment, chromium plating with the same thickness as in flat portions is applied also to portions that conventionally tend to have different plating thickness from the flat portions, such as corner portions and fine uneven portions of the workpiece, and the same covering power and throwing power as in hexavalent chromium plating are exerted even in trivalent chromium plating. The plating thickness of the plated product produced in this embodiment can be appropriately set corresponding to the application or the like of the plated product, but portions with trivalent chromium plating alone, excluding base plating, preferably have a plating thickness of 5 .mu.m or more and 600 .mu.m or less, in order to exert the effects of the present invention more significantly. The plating thickness is more preferably 50 .mu.m or more, particularly preferably 100 .mu.m or more. The plating thickness may be measured, for example, using a fluorescent x-ray film thickness meter or the like, as needed. However, the measurement using the fluorescent x-ray film thickness meter is difficult when the plating thickness is 50 .mu.m or more, and therefore the cross sections of the plated product may be observed using a scanning electron microscope (SEM) in such a case. Then, the plating thickness can be determined by measuring the plating thickness of the plated product at several sites selected at random and arithmetically averaging the measurement results excluding abnormal values.
[0037] Detailed descriptions are not further repeated herein, but a conventionally known technical matter that is not specifically shown in the aforementioned examples can be appropriately employed for the method for producing a plated product and the plating solution used for the plating in this embodiment, without significantly impairing the effects of the present invention. That is, the present invention is not limited to those in the ranges of the aforementioned examples.
EXAMPLES
[0038] Next, the present invention will be described further in detail by way of examples. However, the present invention is not limited to these examples.
(Summary of the Present Invention: Difference from Conventional Trivalent Chromium Plating)
[0039] FIG. 1 shows the relationship between current density and plating thickness in hexavalent chromium plating and trivalent chromium plating disclosed in general literatures or the like. It is understood from this graph that it is difficult to exert throwing power in conventional trivalent chromium plating since the slope of the straight line that shows the relationship between current density and plating thickness rapidly increases at a current density of about 5 A/dm.sup.2. Further, it is understood from this graph that it is difficult to perform plating at a current density of 5 A/dm.sup.2 or less and to perform plating with excellent covering power in conventional trivalent chromium plating.
[0040] In contrast, in the case of using the plating solution of the present invention, the relationship between current density and plating thickness is as shown in FIG. 2, where trivalent chromium plating at a low current density of 5 A/dm.sup.2 or less is made possible as in hexavalent chromium plating. Further, in the case of using the plating solution of the present invention, the relationship between current density and plating thickness is made linear from the region at a low current density of 5 A/dm.sup.2 or less to the region at a high current density of about 30 A/dm.sup.2, and thus plating can be performed with excellent throwing power. Accordingly, it can be seen also from this graph that the present invention provides a plating solution for trivalent chromium plating that is excellent in covering power and throwing power. The results of the study on this point will be described in detail below.
Evaluation 1: Bath Temperature 1
[0041] A plating solution was prepared so as to contain the ingredients shown in Table 1. That is, a plating solution containing chromium sulfate and having a concentration of Cr.sup.3 ions of 1 mol/L was prepared. Formic acid and urea were added to the plating solution so that each has a concentration of 0.5 mol/L. Further, the plating solution was adjusted to have a pH of 1.5.
TABLE-US-00001 TABLE 1 Reference Complexing agent concentration 1-1 1-2 1-3 Chromium sulfate 1 mol/L 1 1 1 Formic acid 0.5 mol/L 0.5 0.5 0.5 Urea 0.5 mol/L 0.5 0.5 0.5 Boric acid 0.5 mol/L 0.5 0.5 0.5 Aluminum sulfate 0.15 mol/L 0.15 0.15 0.15 Sodium sulfate 0.3 mol/L 0.3 0.3 0.3 Sodium dodecyl 50 mg/L 50 50 50 sulfate Bath 30 35 40 temperature (.degree. C.) Plating condition at 1 A/dm.sup.2 x x x each current density 2 A/dm.sup.2 .smallcircle. x x 5 A/dm.sup.2 .smallcircle. .smallcircle. x 10 A/dm.sup.2 .smallcircle. .smallcircle. x 15 A/dm.sup.2 .smallcircle. .smallcircle. x 20 A/dm.sup.2 .smallcircle. .smallcircle. x 25 A/dm.sup.2 .smallcircle. .smallcircle. x Conditions: pH 1.5, Hull cell total current amount of 5 A, Plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating x: No plating
[0042] Using the aforementioned plating solution, a Hull cell test was conducted with a current value of 5 A. The test was performed on three patterns at a bath temperature of 30.degree. C., 35.degree. C., and 40.degree. C., and the test was conducted for 10 minutes. The results are shown in FIG. 3 and Table 1 together.
[0043] Further, the plating thickness of the sample after the test was measured using a fluorescent x-ray film thickness meter. The results are shown in FIG. 4. It was seen from the results shown in Table 1 and FIG. 4 that at least a certain plating thickness could be ensured when the current density was low due to the concentration of Cr.sup.3+ ions being 1 mol/L or less. Further, it was proved from the results shown in Table 1 and FIG. 4 that the bath temperature of less than 40.degree. C. was advantageous in order to perform trivalent chromium plating with excellent covering power.
Evaluation 2: Bath Temperature 2
[0044] Based on the results of "Evaluation 1", the Hull cell test was performed on five patterns at a bath temperature of 24.degree. C., 26.degree. C., 28.degree. C., 30.degree. C., and 32.degree. C. by further subdividing the bath temperature, and the test results were evaluated in the same manner as in "Evaluation 1". The results are shown in Table 2 and FIGS. 5 and 6.
TABLE-US-00002 TABLE 2 Reference Complexing agent concentration 2-1 2-2 2-3 2-4 2-5 Chromium sulfate 1 mol/L 1 1 1 1 1 Formic acid 0.5 mol/L 0.5 0.5 0.5 0.5 0.5 Urea 0.5 mol/L 0.5 0.5 0.5 0.5 0.5 Boric acid 0.5 mol/L 0.5 0.5 0.5 0.5 0.5 Aluminum sulfate 0.15 mol/L 0.15 0.15 0.15 0.15 0.15 Sodium sulfate 0.3 mol/L 0.3 0.3 0.3 0.3 0.3 Sodium dodecyl 50 mg/L 50 50 50 50 50 sulfate Bath 24 26 28 30 32 temperature (.degree. C.) Plating condition at 1 A/dm.sup.2 x x x x x each current density 2 A/dm.sup.2 .smallcircle. .smallcircle. x x x 5 A/dm.sup.2 .smallcircle. .smallcircle. x x x 8 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. x x 10 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. 15 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. 20 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. x 25 A/dm.sup.2 x x x x x Conditions: pH 1.5, Hull cell total current amount of 5 A, Plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating x: No plating or abnormal deposition
[0045] It is seen also from these table and graphs that deposition of the plating film at each current density can be performed at a substantially constant speed when the bath temperature falls within the range from 24.degree. C. to 28.degree. C. Further, it turned out that the covering power was most improved at a bath temperature of 24.degree. C., and preferable results could be obtained by performing the plating at a lower bath temperature. Then, it was also considered to further decrease the bath temperature, but when the bath temperature was excessively decreased, the bath composition partially crystallized to make the plating surface rough, or the bath viscosity increased to make it difficult to remove hydrogen at the cathode. Therefore, it was determined that the bath temperature was optimally within the range of 24.degree. C. to 28.degree. C. It was confirmed also in a bent cathode test, which was separately performed, that good plating tends to be made possible, even at a low current density, by employing a bath temperature of 24.degree. C. to 28.degree. C.
Evaluation 3: Bath Concentration
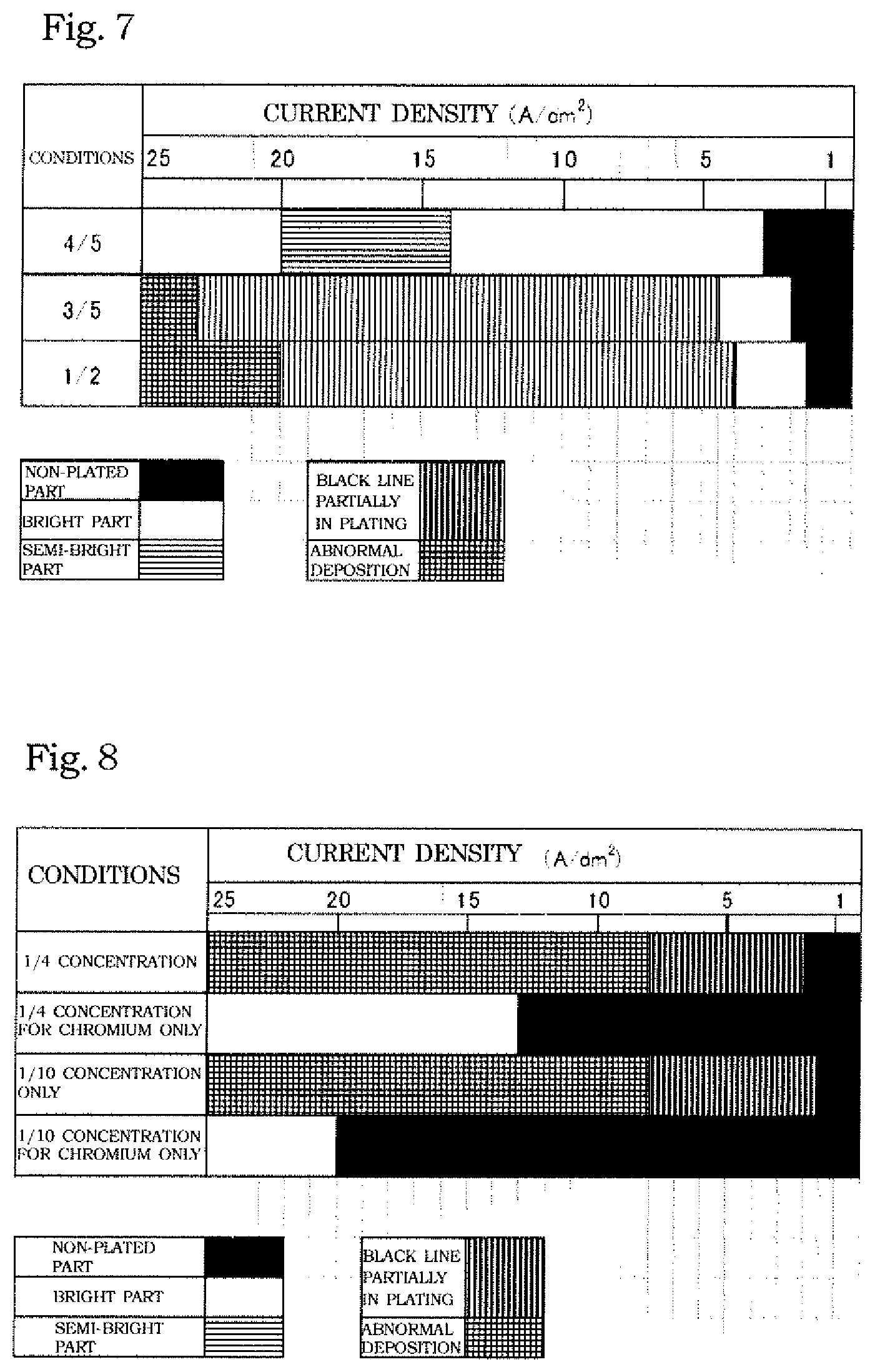
[0046] A Hull cell test was conducted with a reduced concentration of the bath composition itself, in order to improve the covering power by suppressing decrease in the bath viscosity and crystallization of the bath liquid. Specifically, the Hull cell test was conducted at a bath temperature of 25.degree. C. with the bath composition reduced to 4/5 (at a concentration of Cr.sup.3+ ions of 0.8 mol/L and a concentration of each of formic acid and urea of 0.4 mol/L), 3/5 (at a concentration of Cr.sup.3+ ions of 0.6 mol/L and a concentration of each of formic acid and urea of 0.3 mol/L), and 1/2 (at a concentration of Cr.sup.3+ ions of 0.5 mol/L and a concentration of each of formic acid and urea of 0.25 mol/L). The results are shown in Table 3 and FIG. 7. As is obvious also from Table 3 and FIG. 7, it turned out in this "Evaluation 3" that a better plating film was obtained at a lower bath concentration. It was confirmed also in a bent cathode test, which was separately performed, that the best results were obtained by using a plating solution containing 0.5-fold components.
TABLE-US-00003 TABLE 3 Reference Complexing agent concentration 3-1 3-2 3-3 Chromium sulfate 1 mol/L 0.8 0.6 0.5 Formic acid 0.5 mol/L 0.4 0.3 0.25 Urea 0.5 mol/L 0.4 0.3 0.25 Boric acid 0.5 mol/L 0.4 0.3 0.25 Aluminum sulfate 0.15 mol/L 0.12 0.09 0.075 Sodium sulfate 0.3 mol/L 0.24 0.18 0.15 Sodium dodecyl 50 mg/L 40 30 25 sulfate Plating condition at 1 A/dm.sup.2 x x x each current density 2 A/dm.sup.2 x .smallcircle. .smallcircle. 5 A/dm.sup.2 .smallcircle. .DELTA. .DELTA. 10 A/dm.sup.2 .smallcircle. .DELTA. .DELTA. 15 A/dm.sup.2 .DELTA. .DELTA. .DELTA. 20 A/dm.sup.2 .DELTA. .DELTA. .DELTA. 25 A/dm.sup.2 .DELTA. x x Conditions: pH 1.5, Bath temperature of 25.degree. C., Hull cell total current amount of 5 A, Plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating .DELTA.: Partly faulty x: No plating or abnormal deposition
Evaluation 4: Added Amount 1 of Formic Acid and Urea
[0047] Based on the results of "Evaluation 3", attempts to further reduce the bath concentration were made. Specifically, a Hull cell test was conducted with the bath composition reduced to 1/4 and 1/10. As a result, the covering power was further improved, but abnormal plating deposition was found. Therefore, while the chromium ion concentration was set to 1/4 (0.25 mol/L) and 1/10 (0.1 mol/L), the concentration of formic acid and urea was reset to the initial value (0.5 mol/L). As a result, the abnormal deposition was eliminated as shown in Table 4 and FIG. 8, but the covering power decreased.
TABLE-US-00004 TABLE 4 Reference Complexing agent concentration 4-1 4-2 4-3 4-4 Chromium sulfate 1 mol/L 0.25 0.25 0.1 0.1 Formic acid 0.5 mol/L 0.125 0.5 0.05 0.5 Urea 0.5 mol/L 0.125 0.5 0.05 0.5 Boric acid 0.5 mol/L 0.125 0.5 0.05 0.5 Aluminum sulfate 0.15 mol/L 0.0375 0.15 0.015 0.15 Sodium sulfate 0.3 mol/L 0.075 0.3 0.03 0.3 Sodium dodecyl 50 mg/L 12.5 50 5 5 sulfate Plating condition at 1 A/dm.sup.2 x x x x each current density 2 A/dm.sup.2 x x x x 5 A/dm.sup.2 x .smallcircle. x .DELTA. 10 A/dm.sup.2 x .smallcircle. x .DELTA. 15 A/dm.sup.2 x .smallcircle. x .DELTA. 20 A/dm.sup.2 .smallcircle. .smallcircle. x x 25 A/dm.sup.2 .smallcircle. x .smallcircle. x Conditions: pH 1.5, Bath temperature of 25.degree. C., Hull cell total current amount of 5 A, Plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating .DELTA.: Partly faulty x: No plating or abnormal deposition
Evaluation 5: Added Amount 2 of Formic Acid and Urea
[0048] Based on the results of "Evaluation 4", attempts to reduce the abnormal deposition by varying the amounts of formic acid and urea were made. As a result of the Hull cell test (see Table 5 and FIG. 9), it turned out that the abnormal deposition was suppressed and the covering power was good by setting the concentration of formic acid to a low value of 0.1 mol/L and the concentration of urea to the initial value (0.5 mol/L).
TABLE-US-00005 TABLE 5 Reference Complexing agent concentration 5-1 5-2 5-3 5-4 Chromium sulfate 1 mol/L 0.1 0.1 0.25 0.25 Formic acid 0.5 mol/L 0.5 0.1 0.5 0.1 Urea 0.5 mol/L 0.1 0.5 0.1 0.5 Boric acid 0.5 mol/L 0.5 0.5 0.5 0.5 Aluminum sulfate 0.15 mol/L 0.15 0.15 0.15 0.15 Sodium sulfate 0.3 mol/L 0.3 0.3 0.3 0.3 Sodium dodecyl 50 mg/L 50 50 50 50 sulfate Plating condition at 1 A/dm.sup.2 x x x x each current density 2 A/dm.sup.2 x x x x 5 A/dm.sup.2 x x x .smallcircle. 10 A/dm.sup.2 x x x .smallcircle. 15 A/dm.sup.2 x x x .smallcircle. 20 A/dm.sup.2 x .smallcircle. .smallcircle. .smallcircle. 25 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. x Conditions: pH 1.5, Bath temperature of 25.degree. C., Hull cell total current amount of 5 A, Plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating .DELTA.: Partly faulty x: No plating or abnormal deposition
[0049] Subsequently, whether the covering power was improved was checked, with the concentration of urea fixed to 0.5 mol/L and the concentration of formic acid varied from 0.1 mol/L to 0.22 mol/L. The results are shown in Table 6 and FIG. 10.
TABLE-US-00006 TABLE 6 Reference Complexing agent concentration 5-5 5-6 5-7 5-8 5-9 5-10 5-11 Chromium sulfate 1 mol/L 0.25 0.25 0.25 0.25 0.25 0.25 0.25 Formic acid 0.5 mol/L 0.1 0.12 0.14 0.16 0.18 0.2 0.22 Urea 0.5 mol/L 0.5 0.5 0.5 0.5 0.5 0.5 0.5 Boric acid 0.5 mol/L 0.5 0.5 0.5 0.5 0.5 0.5 0.5 Aluminum sulfate 0.15 mol/L 0.15 0.15 0.15 0.15 0.15 0.15 0.15 Sodium sulfate 0.3 mol/L 0.3 0.3 0.3 0.3 0.3 0.3 0.3 Sodium dodecyl 50 mg/L 50 50 50 50 50 50 50 sulfate Plating condition at 1 A/dm.sup.2 x x x x x x x each current density 2 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. x x x x 5 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. x x 10 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. 15 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. 20 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. .smallcircle. 25 A/dm.sup.2 x x x x x x x Conditions: pH 1.5, Bath temperature of 25.degree. C., Hull cell total current amount of 5 A, plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating .DELTA.: Partly faulty x: No plating or abnormal deposition
[0050] It turned out from these table and graph that good results were obtained when the concentration of formic acid was 0.2 mol/L or less.
[0051] Further, the covering power was evaluated by further reducing the concentration of formic acid. The results are shown in Table 7 and FIG. 11.
TABLE-US-00007 TABLE 7 Reference Complexing agent concentration 5-12 5-13 5-14 5-15 Chromium sulfate 1 mol/L 0.25 0.25 0.25 0.25 Formic acid 0.5 mol/L 0 0.04 0.06 0.08 Urea 0.5 mol/L 0.5 0.5 0.5 0.5 Boric acid 0.5 mol/L 0.5 0.5 0.5 0.5 Aluminum sulfate 0.15 mol/L 0.15 0.15 0.15 0.15 Sodium sulfate 0.3 mol/L 0.3 0.3 0.3 0.3 Sodium dodecyl 50 mg/L 50 50 50 50 sulfate Plating condition at 1 A/dm.sup.2 x x x x each current density 2 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. 5 A/dm.sup.2 .smallcircle. .smallcircle. .smallcircle. .smallcircle. 10 A/dm.sup.2 x .smallcircle. .smallcircle. .smallcircle. 15 A/dm.sup.2 x x .smallcircle. .smallcircle. 20 A/dm.sup.2 x x .smallcircle. .smallcircle. 25 A/dm.sup.2 x x x .smallcircle. Conditions: pH 1.5, Bath temperature of 25.degree. C., Hull cell total current amount of 5 A, plating time of 10 mins Evaluation Symbol: .smallcircle.: Normal plating .DELTA.: Partly faulty x: No plating or abnormal deposition
[0052] It turned out from these table and graph that good results were obtained when the concentration of formic acid was 0.05 mol/L or more. As a result of a bent cathode test conducted under the same conditions as shown in Tables 6 and 7, using a cathode with a recessed portion of a "C-shape" in side view obtained by folding a strip-shaped metal plate, it was confirmed that plating was applied also to a range of 60% or more of the recessed portion when the concentration of formic acid falls within the range of 0.05 mol/L or more and 0.1 mol/L or less.
[0053] As described above, it was able to be confirmed that good covering power was exhibited in the plating solution for trivalent chromium plating containing chromium sulfate when the concentration of Cr.sup.3+ ions was set to the range of 0.1 mol/L or more and 1 mol/L or less, and the concentration of formic acid was set to the range of 0.05 mol/L or more and 0.2 mol/L or less. Further, it was able to be confirmed that particularly good covering power was exhibited in the plating solution when the concentration of Cr.sup.3+ ions was set to the range of 0.1 mol/L to 0.3 mol/L. Further, it was able to be confirmed from the aforementioned evaluation that it was effective to set the temperature of the plating bath to 20.degree. C. or more and less than 40.degree. C. Also from these facts, it is understood that the present invention can provide a plating solution for trivalent chromium plating with excellent covering power so as to enlarge the application range of environmentally friendly plated products.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.