Nucleophilic Catalysts For Oxime Linkage
Haider; Stefan ; et al.
U.S. patent application number 16/532212 was filed with the patent office on 2020-01-16 for nucleophilic catalysts for oxime linkage. The applicant listed for this patent is Baxalta GmbH, Baxalta Incorporated. Invention is credited to Stefan Haider, Andreas Ivens, Hanspeter Rottensteiner, Jurgen Siekmann, Peter Turecek, Oliver Zoechling.
| Application Number | 20200017543 16/532212 |
| Document ID | / |
| Family ID | 45556599 |
| Filed Date | 2020-01-16 |







View All Diagrams
| United States Patent Application | 20200017543 |
| Kind Code | A1 |
| Haider; Stefan ; et al. | January 16, 2020 |
NUCLEOPHILIC CATALYSTS FOR OXIME LINKAGE
Abstract
The invention relates to materials and methods of conjugating a water soluble polymer to an oxidized carbohydrate moiety of a therapeutic protein comprising contacting the oxidized carbohydrate moiety with an activated water soluble polymer under conditions that allow conjugation. More specifically, the present invention relates to the aforementioned materials and methods wherein the water soluble polymer contains an active aminooxy group and wherein an oxime or hydrazone linkage is formed between the oxidized carbohydrate moiety and the active aminooxy group on the water soluble polymer, and wherein the conjugation is carried out in the presence of a nucleophilic catalyst.
| Inventors: | Haider; Stefan; (Prinzersdorf, AT) ; Ivens; Andreas; (Zurich, CH) ; Rottensteiner; Hanspeter; (Vienna, AT) ; Siekmann; Jurgen; (Vienna, AT) ; Turecek; Peter; (Klosterneuburg, AT) ; Zoechling; Oliver; (Vienna, AT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 45556599 | ||||||||||
| Appl. No.: | 16/532212 | ||||||||||
| Filed: | August 5, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15281616 | Sep 30, 2016 | 10414793 | ||
| 16532212 | ||||
| 14136233 | Dec 20, 2013 | 9492555 | ||
| 15281616 | ||||
| 13194038 | Jul 29, 2011 | 8642737 | ||
| 14136233 | ||||
| 12843542 | Jul 26, 2010 | 8637640 | ||
| 13194038 | ||||
| 61369186 | Jul 30, 2010 | |||
| 61347136 | May 21, 2010 | |||
| 61228828 | Jul 27, 2009 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 47/60 20170801; C07K 1/1077 20130101; C07K 1/34 20130101; A61K 47/61 20170801; C07K 14/755 20130101; C07K 1/20 20130101 |
| International Class: | C07K 1/107 20060101 C07K001/107; C07K 1/20 20060101 C07K001/20; C07K 1/34 20060101 C07K001/34; C07K 14/755 20060101 C07K014/755; A61K 47/61 20060101 A61K047/61; A61K 47/60 20060101 A61K047/60 |
Claims
1.-72. (canceled)
73. A modified therapeutic protein comprising an activated water soluble polymer conjugated to an oxidized carbohydrate moiety on a therapeutic protein through forming a hydrazone linkage, wherein said activated water soluble polymer contains an active hydrazide group and is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulan, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, and poly(l-hydroxymethylethylene hydroxymethylformal) (PHF); wherein the modified therapeutic protein is prepared by a method comprising the steps of: a) oxidizing a carbohydrate moiety on a therapeutic protein by incubating said protein with an oxidizing agent selected from the group consisting of sodium periodate (NaIO.sub.4), lead tetraacetate (Pb(OAc).sub.4) and potassium perruthenate (KRuO.sub.4); and b) forming a hydrazone linkage between the oxidized carbohydrate moiety of the therapeutic protein and the activated water soluble polymer containing an active hydrazide group in the presence of a nucleophilic catalyst under conditions allowing formation of said hydrazone linkage; and wherein the nucleophilic catalyst is m-toluidine.
74. The modified therapeutic protein of claim 73, wherein the therapeutic protein is selected from the group consisting of Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor V (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF), ADAMTS 13 protease, IL-1 alpha, IL-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-11, human growth hormone (HGH), tumor necrosis factor-alpha (TNF-alpha), colony stimulating factor-1 (CSF-1), M-CSF, SCF, GM-CSF, granulocyte colony stimulating factor (G-CSF), EPO, interferon-alpha (IFN-alpha), consensus interferon, IFN-beta, IFN-gamma, IFN-omega, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-14, IL-15, IL-16, IL-17, IL-18, IL-19, IL-20, IL-21, IL-22, IL-23, IL-24, IL-31, IL-32 alpha, IL-33, thrombopoietin (TPO), Ang-1, Ang-2, Ang-4, Ang-Y, angiopoietin-like polypeptide 1 (ANGPTL1), angiopoietin-like polypeptide 2 (ANGPTL2), angiopoietin-like polypeptide 3 (ANGPTL3), angiopoietin-like polypeptide 4 (ANGPTL4), angiopoietin-like polypeptide 5 (ANGPTL5), angiopoietin-like polypeptide 6 (ANGPTL6), angiopoietin-like polypeptide 7 (ANGPTL7), vitronectin, vascular endothelial growth factor (VEGF), angiogenin, activin A, activin B, activin C, bone morphogenic protein-1, bone morphogenic protein-2, bone morphogenic protein-3, bone morphogenic protein-4, bone morphogenic protein-5, bone morphogenic protein-6, bone morphogenic protein-7, bone morphogenic protein-8, bone morphogenic protein-9, bone morphogenic protein-10, bone morphogenic protein-11, bone morphogenic protein-12, bone morphogenic protein-13, bone morphogenic protein-14, bone morphogenic protein-15, bone morphogenic protein receptor IA, bone morphogenic protein receptor IB, bone morphogenic protein receptor II, brain derived neurotrophic factor, cardiotrophin-1, ciliary neurotrophic factor, ciliary neurotrophic factor receptor, cripto, cryptic, cytokine-induced neutrophil chemotactic factor 1, cytokine-induced neutrophil chemotactic factor 2.alpha., cytokine-induced neutrophil chemotactic factor 2.beta., .beta. endothelial cell growth factor, endothelin 1, epidermal growth factor, epigen, epiregulin, epithelial-derived neutrophil attractant, fibroblast growth factor 4, fibroblast growth factor 5, fibroblast growth factor 6, fibroblast growth factor 7, fibroblast growth factor 8, fibroblast growth factor 8b, fibroblast growth factor 8c, fibroblast growth factor 9, fibroblast growth factor 10, fibroblast growth factor 11, fibroblast growth factor 12, fibroblast growth factor 13, fibroblast growth factor 16, fibroblast growth factor 17, fibroblast growth factor 19, fibroblast growth factor 20, fibroblast growth factor 21, fibroblast growth factor acidic, fibroblast growth factor basic, glial cell line-derived neurotrophic factor receptor .alpha.1, glial cell line-derived neurotrophic factor receptor .alpha.2, growth related protein, growth related protein .alpha., growth related protein .alpha., growth related protein .gamma., heparin binding epidermal growth factor, hepatocyte growth factor, hepatocyte growth factor receptor, hepatoma-derived growth factor, insulin-like growth factor I, insulin-like growth factor receptor, insulin-like growth factor II, insulin-like growth factor binding protein, keratinocyte growth factor, leukemia inhibitory factor, leukemia inhibitory factor receptor .alpha., nerve growth factor, nerve growth factor receptor, neuropoietin, neurotrophin-3, neurotrophin-4, oncostatin M (OSM), placenta growth factor, placenta growth factor 2, platelet-derived endothelial cell growth factor, platelet derived growth factor, platelet derived growth factor A chain, platelet derived growth factor AA, platelet derived growth factor AB, platelet derived growth factor B chain, platelet derived growth factor BB, platelet derived growth factor receptor .alpha., platelet derived growth factor receptor 13, pre-B cell growth stimulating factor, stem cell factor (SCF), stem cell factor receptor, TNF, TNF0, TNF1, TNF2, transforming growth factor .alpha., transforming growth factor .beta., transforming growth factor .beta.1, transforming growth factor .beta.1.2, transforming growth factor .beta.2, transforming growth factor .beta.3, transforming growth factor .beta.5, latent transforming growth factor .beta.1, transforming growth factor .beta. binding protein I, transforming growth factor .beta. binding protein II, transforming growth factor .beta. binding protein III, thymic stromal lymphopoietin (TSLP), tumor necrosis factor receptor type I, tumor necrosis factor receptor type II, urokinase-type plasminogen activator receptor, phospholipase-activating protein (PUP), insulin, lectin, ricin, prolactin, chorionic gonadotropin, follicle-stimulating hormone, thyroid-stimulating hormone, tissue plasminogen activator, IgG, IgE, IgM, IgA, and IgD, .alpha.-galactosidase, .beta.-galactosidase, DNAse, fetuin, luteinizing hormone, estrogen, albumin, lipoproteins, fetoprotein, transferrin, thrombopoietin, urokinase, integrin, thrombin, leptin, Humira (adalimumab), Prolia (denosumab), Enbrel (etanercept), a protein in Table 1, or a biologically active fragment, derivative or variant thereof.
75. The modified therapeutic protein of claim 74, wherein the therapeutic protein has biological activity of a blood coagulation protein.
76. The modified therapeutic protein of claim 75, wherein the blood cogulation protein is selected from the group consisting of FVIIa, FVIII and FIX.
77. The modified therapeutic protein of claim 73, wherein the water soluble polymer is PEG or PSA.
78. The modified therapeutic protein of claim 73, wherein the oxidizing agent is sodium periodate (NaIO.sub.4) and is added in an amount to result in a final concentration between about 50 .mu.M and about 1000 .mu.M under conditions comprising a time period between about 0.1 minutes and 120 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring.
79. The modified therapeutic protein of claim 73, wherein the m-toluidine is provided at a concentration between 1 mM and about 50 mM.
80. The modified therapeutic protein of claim 74, wherein the therapeutic protein has biological activity of FVIIa, FVIII or FIX; wherein the water soluble polymer is PEG or PSA; wherein the oxidizing agent is sodium periodate (NaIO4) and is added in an amount to result in a final concentration between about 50 .mu.M and about 1000 .mu.M; and wherein the m-toluidine is provided at a concentration between 1 mM and about 50 mM.
81. The modified therapeutic protein of claim 80, wherein the oxidizing agent is sodium periodate (NaIO.sub.4) and is added in an amount to result in a final concentration of 400 .mu.M; and wherein the m-toluidine is provided at a concentration of 10 mM.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation application of U.S. patent application Ser. No. 15/281,616, filed Sep. 30, 2016, which is a continuation application of U.S. patent application Ser. No. 14/136,233, filed Dec. 20, 2013, now U.S. Pat. No. 9,492,555, which is a continuation application of U.S. patent application Ser. No. 13/194,038, filed Jul. 29, 2011, now U.S. Pat. No. 8,642,737, which claims benefit to U.S. Provisional No. 61/369,186, filed Jul. 30, 2010, and is a Continuation-In-Part of U.S. patent application Ser. No. 12/843,542, filed Jul. 26, 2010, now U.S. Pat. No. 8,637,640, which claims benefit of to U.S. Provisional No. 61/347,136, filed May 21, 2010 and U.S. Provisional No. 61/228,828, filed Jul. 27, 2009, all of which are incorporated herein by reference in its entirety.
REFERENCE TO A SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, is named "SEQUENCE LISTING" and is 4,096 kilobytes in size.
FIELD OF THE INVENTION
[0003] The present invention relates to materials and methods for conjugating a water soluble polymer to a protein.
BACKGROUND OF THE INVENTION
[0004] The preparation of conjugates by forming a covalent linkage between the water soluble polymer and the therapeutic protein can be carried out by a variety of chemical methods. PEGylation of polypeptide drugs protects them in circulation and improves their pharmacodynamic and pharmacokinetic profiles (Harris and Chess, Nat Rev Drug Discov. 2003; 2:214-21). The PEGylation process attaches repeating units of ethylene glycol (polyethylene glycol (PEG)) to a polypeptide drug. PEG molecules have a large hydrodynamic volume (5-10 times the size of globular proteins), are highly water soluble and hydrated, non-toxic, non-immunogenic and rapidly cleared from the body. PEGylation of molecules can lead to increased resistance of drugs to enzymatic degradation, increased half-life in vivo, reduced dosing frequency, decreased immunogenicity, increased physical and thermal stability, increased solubility, increased liquid stability, and reduced aggregation. The first PEGylated drugs were approved by the FDA in the early 1990s. Since then, the FDA has approved several PEGylated drugs for oral, injectable, and topical administration.
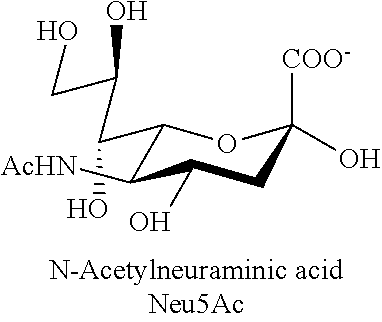
[0005] Polysialic acid (PSA), also referred to as colominic acid (CA), is a naturally occurring polysaccharide. It is a homopolymer of N-acetylneuraminic acid with .alpha.(2.fwdarw.8) ketosidic linkage and contains vicinal diol groups at its non-reducing end. It is negatively charged and a natural constituent of the human body. It can easily be produced from bacteria in large quantities and with pre-determined physical characteristics (U.S. Pat. No. 5,846,951). Because the bacterially-produced PSA is chemically and immunologically identical to PSA produced in the human body, bacterial PSA is non-immunogenic, even when coupled to proteins. Unlike some polymers, PSA acid is biodegradable. Covalent coupling of colominic acid to catalase and asparaginase has been shown to increase enzyme stability in the presence of proteolytic enzymes or blood plasma. Comparative studies in vivo with polysialylated and unmodified asparaginase revealed that polysialylation increased the half-life of the enzyme (Fernandes and Gregoriadis, Int J Pharm. 2001; 217:215-24).
[0006] Coupling of PEG-derivatives to peptides or proteins is reviewed by Roberts et al. (Adv Drug Deliv Rev 2002; 54:459-76). One approach for coupling water soluble polymers to therapeutic proteins is the conjugation of the polymers via the carbohydrate moieties of the protein. Vicinal hydroxyl (OH) groups of carbohydrates in proteins can be easily oxidized with sodium periodate (NaIO4) to form active aldehyde groups (Rothfus et Smith, J Biol Chem 1963; 238:1402-10; van Lenten et Ashwell, J Biol Chem 1971; 246:1889-94). Subsequently the polymer can be coupled to the aldehyde groups of the carbohydrate by use of reagents containing, for example, an active hydrazide group (Wilchek M and Bayer E A, Methods Enzymol 1987; 138:429-42). A more recent technology is the use of reagents containing aminooxy groups which react with aldehydes to form oxime linkages (WO 96/40662, WO2008/025856).
[0007] Additional examples describing conjugation of a water soluble polymer to a therapeutic protein are described in WO 06/071801 which teaches the oxidation of carbohydrate moieties in Von Willebrand factor and subsequent coupling to PEG using hydrazide chemistry; US Publication No. 2009/0076237 which teaches the oxidation of rFVIII and subsequent coupling to PEG and other water soluble polymers (e.g. PSA, HES, dextran) using hydrazide chemistry; WO 2008/025856 which teaches oxidation of different coagulation factors, e.g. rFIX, FVIII and FVIIa and subsequent coupling to e.g., PEG, using aminooxy chemistry by forming an oxime linkage; and U.S. Pat. No. 5,621,039 which teaches the oxidation of FIX and subsequent coupling to PEG using hydrazide chemistry.
[0008] Recently, an improved method was described comprising mild periodate oxidation of sialic acids to generate aldehydes followed by reaction with an aminooxy group containing reagent in the presence of catalytic amounts of aniline (Dirksen A., and Dawson P E, Bioconjugate Chem. 2008; 19, 2543-8; and Zeng Y et al., Nature Methods 2009; 6:207-9). The aniline catalysis dramatically accelerates the oxime ligation, allowing the use of very low concentrations of the reagent. The use of nucelophilic catalysts are also described in Dirksen, A., et al., J Am Chem Soc., 128:15602-3 (2006); Dirksen, A., et al., Angew chem. Int Ed., 45:7581-4 (2006); Kohler, J. J., ChemBioChem., 10:2147-50 (2009); Giuseppone, N., et al., J Am Chem Soc., 127:5528-39 (2005); and Thygesen, M. B., et al., J Org Chem., 75:1752-5 (2010).
[0009] Although aniline catalysis can accelerate the oxime ligation allowing short reaction times and the use of low concentrations of the aminooxy reagent, aniline has toxic properties that must be considered when, for example, the conjugated therapeutic protein to form the basis of a pharmaceutical. For example, aniline has been shown to induce methemoglobinemia (Harrison, J. H., and Jollow, D. J., Molecular Pharmacology, 32(3) 423-431, 1987). Long-term dietary treatment of rats has been shown to induce tumors in the spleen (Goodman, D G., et al., J Natl Cancer Inst., 73(1):265-73, 1984). In vitro studies have also shown that aniline has the potential to induce chromosome mutations and has the potentially genotoxic activity (Bombhard E. M. et Herbold B, Critical Reviews in Toxicology 35, 783-835, 2005).
[0010] Considering the potentially dangerous properties of aniline and notwithstanding the methods available of conjugating water soluble polymers to therapeutic proteins, there remains a need to develop materials and methods for conjugating water soluble polymers to proteins that improves the protein's pharmacodynamic and/or pharmacokinetic properties while minimizing the costs associated with the various reagents and minimizing the health risks to the patient recipient.
SUMMARY OF THE INVENTION
[0011] The present invention provides materials and methods for conjugating polymers to proteins that improves the protein's pharmacodynamic and/or pharmacokinetic properties while minimizing the costs associated with the various reagents and the health risks to the patient recipients when the conjugation reaction is catalyzed by a nucleophilic catalyst. In various embodiments of the invention, alternative catalysts to substitute for aniline are provided.
[0012] In one embodiment, a method of conjugating a water soluble polymer to an oxidized carbohydrate moiety of a therapeutic protein is provided comprising contacting the oxidized carbohydrate moiety with an activated water soluble polymer under conditions that allow conjugation; said water soluble polymer containing an active aminooxy group and is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); and said carbohydrate moiety oxidized by incubation with a buffer comprising an oxidizing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); wherein an oxime linkage is formed between the oxidized carbohydrate moiety and the active aminooxy group on the water soluble polymer; and wherein said oxime linkage formation is catalyzed by a nucleophilic catalyst selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0013] In another embodiment, a method of conjugating a water soluble polymer to an oxidized carbohydrate moiety of a therapeutic protein is provided comprising contacting the oxidized carbohydrate moiety with an activated water soluble polymer under conditions that allow conjugation; said therapeutic protein selected from the group consisting of Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF), ADAMTS 13 protease, IL-1 alpha, IL-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-11, colony stimulating factor-1 (CSF-1), M-CSF, SCF, GM-CSF, granulocyte colony stimulating factor (G-CSF), EPO, interferon-alpha (IFN-alpha), consensus interferon, IFN-beta, IFN-gamma, IFN-omega, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-14, IL-15, IL-16, IL-17, IL-18, IL-19, IL-20, IL-21, IL-22, IL-23, IL-24, IL-31, IL-32 alpha, IL-33, thrombopoietin (TPO), Ang-1, Ang-2, Ang-4, Ang-Y, angiopoietin-like polypeptide 1 (ANGPTL1), angiopoietin-like polypeptide 2 (ANGPTL2), angiopoietin-like polypeptide 3 (ANGPTL3), angiopoietin-like polypeptide 4 (ANGPTL4), angiopoietin-like polypeptide 5 (ANGPTL5), angiopoietin-like polypeptide 6 (ANGPTL6), angiopoietin-like polypeptide 7 (ANGPTL7), vitronectin, vascular endothelial growth factor (VEGF), angiogenin, activin A, activin B, activin C, bone morphogenic protein-1, bone morphogenic protein-2, bone morphogenic protein-3, bone morphogenic protein-4, bone morphogenic protein-5, bone morphogenic protein-6, bone morphogenic protein-7, bone morphogenic protein-8, bone morphogenic protein-9, bone morphogenic protein-10, bone morphogenic protein-11, bone morphogenic protein-12, bone morphogenic protein-13, bone morphogenic protein-14, bone morphogenic protein-15, bone morphogenic protein receptor IA, bone morphogenic protein receptor IB, bone morphogenic protein receptor II, brain derived neurotrophic factor, cardiotrophin-1, ciliary neutrophic factor, ciliary neutrophic factor receptor, cripto, cryptic, cytokine-induced neutrophil chemotactic factor 1, cytokine-induced neutrophil, chemotactic factor 2.alpha., cytokine-induced neutrophil chemotactic factor 2.beta.,.beta. endothelial cell growth factor, endothelin 1, epidermal growth factor, epigen, epiregulin, epithelial-derived neutrophil attractant, fibroblast growth factor 4, fibroblast growth factor 5, fibroblast growth factor 6, fibroblast growth factor 7, fibroblast growth factor 8, fibroblast growth factor 8b, fibroblast growth factor 8c, fibroblast growth factor 9, fibroblast growth factor 10, fibroblast growth factor 11, fibroblast growth factor 12, fibroblast growth factor 13, fibroblast growth factor 16, fibroblast growth factor 17, fibroblast growth factor 19, fibroblast growth factor 20, fibroblast growth factor 21, fibroblast growth factor acidic, fibroblast growth factor basic, glial cell line-derived neutrophic factor receptor .alpha.1, glial cell line-derived neutrophic factor receptor .alpha.2, growth related protein, growth related protein .alpha., growth related protein .beta., growth related protein .gamma., heparin binding epidermal growth factor, hepatocyte growth factor, hepatocyte growth factor receptor, hepatoma-derived growth factor, insulin-like growth factor I, insulin-like growth factor receptor, insulin-like growth factor II, insulin-like growth factor binding protein, keratinocyte growth factor, leukemia inhibitory factor, leukemia inhibitory factor receptor .alpha., nerve growth factor nerve growth factor receptor, neuropoietin, neurotrophin-3, neurotrophin-4, oncostatin M (OSM), placenta growth factor, placenta growth factor 2, platelet-derived endothelial cell growth factor, platelet derived growth factor, platelet derived growth factor A chain, platelet derived growth factor AA, platelet derived growth factor AB, platelet derived growth factor B chain, platelet derived growth factor BB, platelet derived growth factor receptor .alpha., platelet derived growth factor receptor .beta., pre-B cell growth stimulating factor, stem cell factor (SCF), stem cell factor receptor, TNF, TNF0, TNF1, TNF2, transforming growth factor .alpha., transforming growth factor (3, transforming growth factor .beta.1, transforming growth factor .beta.1.2, transforming growth factor .beta.2, transforming growth factor .beta.3, transforming growth factor .beta.5, latent transforming growth factor .beta.1, transforming growth factor .beta. binding protein I, transforming growth factor .beta. binding protein II, transforming growth factor .beta. binding protein III, thymic stromal lymphopoietin (TSLP), tumor necrosis factor receptor type I, tumor necrosis factor receptor type II, urokinase-type plasminogen activator receptor, phospholipase-activating protein (PUP), insulin, lectin ricin, prolactin, chorionic gonadotropin, follicle-stimulating hormone, thyroid-stimulating hormone, tissue plasminogen activator, IgG, IgE, IgM, IgA, and IgD, .alpha.-galactosidase, .beta.-galactosidase, DNAse, fetuin, leutinizing hormone, estrogen, insulin, albumin, lipoproteins, fetoprotein, transferrin, thrombopoietin, urokinase, integrin, thrombin, leptin, Humira (adalimumab), Prolia (denosumab), Enbrel (etanercept), a protein in Table 1, or a biologically active fragment, derivative or variant thereof; said water soluble polymer containing an active aminooxy group and is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); and said carbohydrate moiety oxidized by incubation with a buffer comprising an oxidizing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); wherein an oxime linkage is formed between the oxidized carbohydrate moiety and the active aminooxy group on the water soluble polymer; and wherein in said oxime linkage formation is catalyzed by a nucleophilic catalyst selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0014] In still another embodiment, an aforementioned method is provided wherein a solution comprising an initial concentration of the therapeutic protein between about 0.3 mg/ml and about 3.0 mg/ml is adjusted to a pH value between about 5.0 and about 8.0 prior to contacting with the activated water soluble polymer.
[0015] As used herein, the term "about" means a value above or below a stated value. In various embodiments, the term "about" includes the stated value plus or minus 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10% of the stated value.
[0016] In yet another embodiment, an aforementioned method is provided wherein the initial concentration of the therapeutic protein is about 1.0 mg/ml and the pH is about 6.0. In a related embodiment, the initial concentration of the therapeutic protein is about 0.75 mg/ml and the pH is about 6.0. In still another related embodiment, the initial concentration of the therapeutic protein is about 1.25 mg/ml and the pH is about 6.0.
[0017] In another embodiment, an aforementioned method is provided wherein the therapeutic protein is contacted by a desired excess concentration of activated water soluble polymer, wherein the excess concentration is between about 1-molar and about 300-molar excess. In another embodiment, the excess concentration is about 50-fold molar excess.
[0018] In still another embodiment, an aforementioned method is provided wherein the therapeutic protein is incubated with the activated water soluble polymer under conditions comprising a time period between about 0.5 hours and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring. In another embodiment, the conditions comprise a time period of about 120 minutes, a temperature of about 22.degree. C., the absence of light; and with stirring. As used herein, the term "stirring" is meant to include stirring at various speeds and intensities (e.g., gentle stirring) by commonly used laboratory or manufacturing equipment and products.
[0019] In another embodiment, an aforementioned method is provided wherein the nucleophilic catalyst is added in an amount to result in a final concentration between about 1.0 mM and about 50 mM nucleophilic catalyst, under conditions comprising a time period between about 0.1 minutes and about 30 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring. In another embodiment, the final concentration of the nucleophilic catalyst is about 10 mM, and the conditions comprise a time period of up to about 15 minutes, a temperature of about 22.degree. C., the absence of light; and with stirring.
[0020] In still another embodiment, an aforementioned method is provided wherein the oxidizing agent is added in an amount to result in a final concentration between about 50 .mu.M and about 1000 .mu.M oxidizing agent, under conditions comprising a time period between about 0.1 minutes and 120 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring. In another embodiment, the final concentration of oxidizing agent is about 400 .mu.M, and the conditions comprise a time period of about 10 minutes, a temperature of about 22.degree. C., the absence of light and with stirring.
[0021] In yet another embodiment, an aforementioned method is provided wherein the conjugating the water soluble polymer to the oxidized carbohydrate moiety of the therapeutic protein is stopped by the addition of a quenching agent selected from the group consisting of L-cysteine, methionine, glutathione, glycerol, sodium meta bisulfate (Na2S2O5), tryptophane, tyrosine, histidine or derivatives thereof, kresol, imidazol, and combinations thereof; wherein the quenching agent is added in an amount to result in a final concentration between about 1 mM and about 100 mM quenching agent, under conditions comprising a time period between about 5 minutes and about 120 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring. In another embodiment, the quenching agent is L-cysteine. In still another embodiment, the L-cysteine is added to result in a final concentration of about 10 mM and the conditions comprise a time period of about 60 minutes, a temperature of about 22.degree. C., the absence of light and with stirring.
[0022] In another embodiment, an aforementioned method is provided comprising: a) a first step comprising adjusting the pH value of a solution comprising the therapeutic protein to a pH value between about 5.0 and about 8.0, wherein the therapeutic protein concentration is between about 0.3 mg/ml and about 3.0 mg/ml; b) a second step comprising oxidizing one or more carbohydrates on the therapeutic protein, wherein the oxidizing agent is added to the solution in the first step to result in a final concentration between about 50 .mu.M and about 1000 .mu.M, under conditions comprising a time period between about 0.1 minutes and about 120 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; c) a third step comprising contacting the therapeutic protein with a desired excess concentration of activated water soluble polymer, wherein the excess concentration is between about 1-molar excess and about 300-molar excess, under conditions comprising a time period between about 0.5 hours and about 24 hours, a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring; d) a fourth step comprising adding a nucleophilic catalyst to the solution of the third step, wherein the nucleophilic catalyst is added to result in a final concentration between about 1 mM and about 50 mM, under conditions comprising a time period between about 0.1 minutes and about 30 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; e) a fifth step wherein the therapeutic protein is incubated with the activated water soluble polymer and nucleophilic catalyst under conditions that allow conjugation of the activated water-soluble polymer to one or more oxidized carbohydrates on the therapeutic protein, said conditions comprising a time period between about 0.5 hours and about 24 hours, a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; and f) a sixth step wherein the conjugating the water soluble polymer to the one or more oxidized carbohydrates of the therapeutic protein in the fifth step is stopped by the addition of a quenching agent selected from the group consisting of L-cysteine, methionine, glutathione, glycerol, Na2S205 (sodium meta bisulfite), tryptophane, tyrosine, histidine or derivatives thereof, kresol, imidazol, and combinations thereof; wherein the quenching agent is added to result in a final concentration of about 1 mM and about 100 mM, under conditions comprising a time period between about 5 minutes and about 120 minutes; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring. In another embodiment, the initial concentration of the therapeutic protein in the first step is about 1 mg/ml and the pH is about 6.0; wherein the final concentration of oxidizing agent in the second step is about 400 .mu.M, and the conditions in the fifth step comprise a time period of about 10 minutes, a temperature of about 22.degree. C., the absence of light and with stirring; wherein the excess concentration in the third step is about 50 molar excess; wherein the conditions in the third step comprise a time period of about 15 minutes, a temperature of about 22.degree. C., the absence of light and with stirring; wherein the final concentration of the nucleophilic catalyst in the fourth step is about 10 mM, and the conditions in the fourth step comprise a time period of about 15 minutes, a temperature of about 22.degree. C., the absence of light and with stirring; wherein the conditions of incubating the therapeutic protein with the activated water soluble polymer and nucleophilic catalyst in the fifth step comprise a time period of about 2 hours; a temperature of about 22.degree. C.; the absence of light; and with stirring; and wherein the quenching agent in the sixth step is L-cysteine; and wherein the L-cysteine is added to result in a final concentration of about 10 mM and the conditions in the sixth step comprise a time period of about 60 minutes, a temperature of about 22.degree. C., the absence of light and with stirring.
[0023] In another embodiment, an aforementioned method is provided wherein the water soluble polymer is PSA. In another embodiment the PSA is comprised of about 10-300 sialic acid units. In another embodiment, the water soluble polymer is PEG. In another embodiment, the water soluble polymer is HES. In still another embodiment, the water soluble polymer is HAS.
[0024] In still another embodiment, an aforementioned method is provided wherein the therapeutic protein is FIX. In another embodiment, the therapeutic protein is FVIIa. In another embodiment, the therapeutic protein is FVIII.
[0025] In yet another embodiment, an aforementioned method is provided wherein the oxidizing agent is sodium periodate (NaIO4).
[0026] In another embodiment, an aforementioned method is provided wherein the oxidized carbohydrate moiety of the therapeutic protein is located in the activation peptide of the blood coagulation protein.
[0027] In one embodiment, an aforementioned method is provided wherein PSA is prepared by reacting an activated aminooxy linker with oxidized PSA; wherein the aminooxy linker is selected from the group consisting of: [0028] a) a 3-oxa-pentane-1,5-dioxyamine linker of the formula:
[0028] ##STR00001## [0029] b) a 3,6,9-trioxa-undecane-1,11-dioxyamine linker of the formula:
##STR00002##
[0030] and [0031] c) a 3,6,9,12,15-penatoxa-heptadecane-1,17-dioxyamine linker of the formula:
##STR00003##
[0032] wherein the PSA is oxidized by incubation with a oxidizing agent to form a terminal aldehyde group at the non-reducing end of the PSA. In a related embodiment, the aminooxy linker is 3-oxa-pentane-1,5-dioxyamine.
[0033] In still another embodiment, an aforementioned method is provided wherein the oxidizing agent is NaIO4.
[0034] In another embodiment, an aforementioned method is provided wherein the nucleophilic catalyst is provided at a concentration between about 1 mM and about 50 mM. In one embodiment, the nucleophilic catalyst is m-toluidine. In still another embodiment, the m-toluidine is present in the conjugation reaction at a concentration of about 10 mM.
[0035] In yet another embodiment, an aforementioned method is provided further comprising the step of reducing an oxime linkage in the conjugated therapeutic protein by incubating the conjugated therapeutic protein in a buffer comprising a reducing compound selected from the group consisting of sodium cyanoborohydride (NaCNBH3), ascorbic acid (vitamin C) and NaBH3. In one embodiment, the reducing compound is sodium cyanoborohydride (NaCNBH3).
[0036] In still another embodiment, an aforementioned method is provided further comprising the step of purifying the conjugated therapeutic protein. In another embodiment, the conjugated therapeutic protein is purified by a method selected from the group consisting of chromatography, filtration and precipitation. In another embodiment, the chromatography is selected from the group consisting of Hydrophobic Interaction Chromatography (HIC), Ion Exchange chromatography (IEC), Size exclusion chromatography (SEC), Affinity chromatography, and Reversed-phase chromatography. In still another embodiment, an anti-chaotropic salt is used in a chromotagraphy loading step and in a chromatography washing step. In yet another embodiment, the chromatography takes place in a column. In another embodiment, the column comprises a chromatography resin selected from the group consisting of Phenyl-Sepharose FF and Butyl-Sepharose FF. In another embodiment, the resin is present in the column at a bed height of between about 5 cm and about 20 cm. In one embodiment, the bed height is about 10 cm.
[0037] In another embodiment, an aforementioned method is provided comprising one or more washing steps wherein flow direction is set to up-flow and wherein the flow rate is between about 0.2 cm/min and about 6.7 cm/min. As used herein, the term "down-flow" refers to a flow direction from the top of the chromatographic column to the bottom of the chromatographic column (normal flow direction/standard mode). As used herein, the term "up-flow" refers to a flow direction from the bottom to the top of the column (reversed flow direction). In one embodiment, the flow rate is about 2 cm/min.
[0038] In another embodiment, an aforementioned method is provided comprising one or more elution steps wherein flow direction is set to down-flow and wherein the flow rate is between about 0.1 cm/min and about 6.7 cm/min. In a related embodiment, the flow rate is about 1 cm/min.
[0039] In still another embodiment, an aforementioned method is provided comprising concentrating the conjugated therapeutic protein by ultra-/diafiltration (UF/DF). In another embodiment, the final concentration of therapeutic protein is between about 0.5 and about 3 mg/ml.
[0040] In another embodiment, an aforementioned method is provided wherein the therapeutic protein comprises between about 5 and about 11 water-soluble polymer moieties. In another embodiment, the therapeutic protein comprises between about 1 and about 3 water-soluble polymers.
[0041] In still another embodiment, an aforementioned method is provided wherein the conjugated therapeutic protein is purified using chromatography; wherein an anti-chaotropic salt is used for a loading step and for a washing step; the method comprising one or more washing steps wherein flow direction is set to up-flow and wherein the flow rate is between about 0.2 cm/min and about 6.7 cm/min and one or more elution steps wherein flow direction is set to down-flow and wherein the flow rate is between about 0.2 cm/min andabout 6.7 cm/min; further comprising concentrating the conjugated therapeutic protein by ultra-/diafiltration (UF/DF). In another embodiment, the chromatography is hydrophobic interaction chromatography (HIC); wherein the one or more washing steps flow rate is about 2 cm/min; and wherein the one or more elution steps flow rate is about 1 cm/min.
[0042] In another embodiment, a modified therapeutic protein produced by any of the aforementioned methods is provided.
[0043] In still another embodiment, a method of forming an oxime linkage between an oxidized carbohydrate moiety on a therapeutic protein and an activated water soluble polymer containing an active aminooxy group is provided comprising the steps of: a) oxidizing a carbohydrate moiety on a therapeutic protein by incubating said protein with an oxidizing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); and b) forming an oxime linkage between the oxidized carbohydrate moiety of the therapeutic protein and the activated water soluble polymer containing an active aminooxy group in the presence of a nuclephilic catalyst under conditions allowing formation of said oxime linkage; wherein said water soluble polymer containing an active aminooxy group is selected from the group consisting polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); wherein the nucleophilic catalyst is selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0044] In yet another embodiment, a method of forming an oxime linkage between an oxidized carbohydrate moiety on a therapeutic protein and an activated water soluble polymer containing an active aminooxy group is provided comprising the steps of: a) oxidizing a carbohydrate moiety on a therapeutic protein by incubating said protein with an oxidinzing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); and b) forming an oxime linkage between the oxidized carbohydrate moiety of the therapeutic protein and the activated water soluble polymer containing an an active aminooxy group in the presence of a nuclephilic catalyst under conditions allowing formation of said oxime linkage; wherein the therapeutic protein is selected from the group consisting of Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF), ADAMTS 13 protease, IL-1 alpha, IL-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-11, colony stimulating factor-1 (CSF-1), M-CSF, SCF, GM-CSF, granulocyte colony stimulating factor (G-CSF), EPO, interferon-alpha (IFN-alpha), consensus interferon, IFN-beta, IFN-gamma, IFN-omega, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-14, IL-15, IL-16, IL-17, IL-18, IL-19, IL-20, IL-21, IL-22, IL-23, IL-24, IL-31, IL-32 alpha, IL-33, thrombopoietin (TPO), Ang-1, Ang-2, Ang-4, Ang-Y, angiopoietin-like polypeptide 1 (ANGPTL1), angiopoietin-like polypeptide 2 (ANGPTL2), angiopoietin-like polypeptide 3 (ANGPTL3), angiopoietin-like polypeptide 4 (ANGPTL4), angiopoietin-like polypeptide 5 (ANGPTL5), angiopoietin-like polypeptide 6 (ANGPTL6), angiopoietin-like polypeptide 7 (ANGPTL7), vitronectin, vascular endothelial growth factor (VEGF), angiogenin, activin A, activin B, activin C, bone morphogenic protein-1, bone morphogenic protein-2, bone morphogenic protein-3, bone morphogenic protein-4, bone morphogenic protein-5, bone morphogenic protein-6, bone morphogenic protein-7, bone morphogenic protein-8, bone morphogenic protein-9, bone morphogenic protein-10, bone morphogenic protein-11, bone morphogenic protein-12, bone morphogenic protein-13, bone morphogenic protein-14, bone morphogenic protein-15, bone morphogenic protein receptor IA, bone morphogenic protein receptor IB, bone morphogenic protein receptor II, brain derived neurotrophic factor, cardiotrophin-1, ciliary neutrophic factor, ciliary neutrophic factor receptor, cripto, cryptic, cytokine-induced neutrophil chemotactic factor 1, cytokine-induced neutrophil, chemotactic factor 2a, cytokine-induced neutrophil chemotactic factor 2.beta.,.beta. endothelial cell growth factor, endothelin 1, epidermal growth factor, epigen, epiregulin, epithelial-derived neutrophil attractant, fibroblast growth factor 4, fibroblast growth factor 5, fibroblast growth factor 6, fibroblast growth factor 7, fibroblast growth factor 8, fibroblast growth factor 8b, fibroblast growth factor 8c, fibroblast growth factor 9, fibroblast growth factor 10, fibroblast growth factor 11, fibroblast growth factor 12, fibroblast growth factor 13, fibroblast growth factor 16, fibroblast growth factor 17, fibroblast growth factor 19, fibroblast growth factor 20, fibroblast growth factor 21, fibroblast growth factor acidic, fibroblast growth factor basic, glial cell line-derived neutrophic factor receptor .alpha.1, glial cell line-derived neutrophic factor receptor .alpha.2, growth related protein, growth related protein .alpha., growth related protein .beta., growth related protein .gamma., heparin binding epidermal growth factor, hepatocyte growth factor, hepatocyte growth factor receptor, hepatoma-derived growth factor, insulin-like growth factor I, insulin-like growth factor receptor, insulin-like growth factor II, insulin-like growth factor binding protein, keratinocyte growth factor, leukemia inhibitory factor, leukemia inhibitory factor receptor .alpha., nerve growth factor nerve growth factor receptor, neuropoietin, neurotrophin-3, neurotrophin-4, oncostatin M (OSM), placenta growth factor, placenta growth factor 2, platelet-derived endothelial cell growth factor, platelet derived growth factor, platelet derived growth factor A chain, platelet derived growth factor AA, platelet derived growth factor AB, platelet derived growth factor B chain, platelet derived growth factor BB, platelet derived growth factor receptor .alpha., platelet derived growth factor receptor .beta., pre-B cell growth stimulating factor, stem cell factor (SCF), stem cell factor receptor, TNF, TNF0, TNF1, TNF2, transforming growth factor .alpha., transforming growth factor .beta., transforming growth factor .beta.1, transforming growth factor .beta.1.2, transforming growth factor .beta.2, transforming growth factor .beta.3, transforming growth factor .beta.5, latent transforming growth factor .beta.1, transforming growth factor .beta. binding protein I, transforming growth factor .beta. binding protein II, transforming growth factor .beta. binding protein III, thymic stromal lymphopoietin (TSLP), tumor necrosis factor receptor type I, tumor necrosis factor receptor type II, urokinase-type plasminogen activator receptor, phospholipase-activating protein (PUP), insulin, lectin ricin, prolactin, chorionic gonadotropin, follicle-stimulating hormone, thyroid-stimulating hormone, tissue plasminogen activator, IgG, IgE, IgM, IgA, and IgD, .alpha.-galactosidase, .beta.-galactosidase, DNAse, fetuin, leutinizing hormone, estrogen, insulin, albumin, lipoproteins, fetoprotein, transferrin, thrombopoietin, urokinase, integrin, thrombin, leptin, Humira (adalimumab), Prolia (denosumab), Enbrel (etanercept), a protein from Table 1, or a biologically active fragment, derivative or variant thereof; wherein said water soluble polymer containing an active aminooxy group is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); wherein the nucleophilic catalyst is selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0045] In yet another embodiment, a method of forming a hydrazone linkage between an oxidized carbohydrate moiety on a therapeutic protein and an activated water soluble polymer containing an active hydrazide group is provided comprising the steps of: a) oxidizing a carbohydrate moiety on a therapeutic protein by incubating said protein with an oxidinzing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); and b) forming a hydrazone linkage between the oxidized carbohydrate moiety of the therapeutic protein and the activated water soluble polymer containing an an active hydrazide group in the presence of a nuclephilic catalyst under conditions allowing formation of said hydrazone linkage; wherein said water soluble polymer containing an active hydrazide group is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); wherein the nucleophilic catalyst is selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0046] In another embodiment, a method of forming a hydrazone linkage between an oxidized carbohydrate moiety on a therapeutic protein and an activated water soluble polymer containing an active hydrazide group comprising the steps of: a) oxidizing a carbohydrate moiety on a therapeutic protein by incubating said protein with an oxidinzing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); and b) forming a hydrazone linkage between the oxidized carbohydrate moiety of the therapeutic protein and the activated water soluble polymer containing an an active hydrazide group in the presence of a nuclephilic catalyst under conditions allowing formation of said hydrazone linkage; wherein the therapeutic protein is selected from the group consisting of Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF), ADAMTS 13 protease, IL-1 alpha, IL-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-11, colony stimulating factor-1 (CSF-1), M-CSF, SCF, GM-CSF, granulocyte colony stimulating factor (G-CSF), EPO, interferon-alpha (IFN-alpha), consensus interferon, IFN-beta, IFN-gamma, IFN-omega, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-14, IL-15, IL-16, IL-17, IL-18, IL-19, IL-20, IL-21, IL-22, IL-23, IL-24, IL-31, IL-32 alpha, IL-33, thrombopoietin (TPO), Ang-1, Ang-2, Ang-4, Ang-Y, angiopoietin-like polypeptide 1 (ANGPTL1), angiopoietin-like polypeptide 2 (ANGPTL2), angiopoietin-like polypeptide 3 (ANGPTL3), angiopoietin-like polypeptide 4 (ANGPTL4), angiopoietin-like polypeptide 5 (ANGPTL5), angiopoietin-like polypeptide 6 (ANGPTL6), angiopoietin-like polypeptide 7 (ANGPTL7), vitronectin, vascular endothelial growth factor (VEGF), angiogenin, activin A, activin B, activin C, bone morphogenic protein-1, bone morphogenic protein-2, bone morphogenic protein-3, bone morphogenic protein-4, bone morphogenic protein-5, bone morphogenic protein-6, bone morphogenic protein-7, bone morphogenic protein-8, bone morphogenic protein-9, bone morphogenic protein-10, bone morphogenic protein-11, bone morphogenic protein-12, bone morphogenic protein-13, bone morphogenic protein-14, bone morphogenic protein-15, bone morphogenic protein receptor IA, bone morphogenic protein receptor IB, bone morphogenic protein receptor II, brain derived neurotrophic factor, cardiotrophin-1, ciliary neutrophic factor, ciliary neutrophic factor receptor, cripto, cryptic, cytokine-induced neutrophil chemotactic factor 1, cytokine-induced neutrophil, chemotactic factor 2a, cytokine-induced neutrophil chemotactic factor 2.beta.,.beta. endothelial cell growth factor, endothelin 1, epidermal growth factor, epigen, epiregulin, epithelial-derived neutrophil attractant, fibroblast growth factor 4, fibroblast growth factor 5, fibroblast growth factor 6, fibroblast growth factor 7, fibroblast growth factor 8, fibroblast growth factor 8b, fibroblast growth factor 8c, fibroblast growth factor 9, fibroblast growth factor 10, fibroblast growth factor 11, fibroblast growth factor 12, fibroblast growth factor 13, fibroblast growth factor 16, fibroblast growth factor 17, fibroblast growth factor 19, fibroblast growth factor 20, fibroblast growth factor 21, fibroblast growth factor acidic, fibroblast growth factor basic, glial cell line-derived neutrophic factor receptor .alpha.1, glial cell line-derived neutrophic factor receptor .alpha.2, growth related protein, growth related protein .alpha., growth related protein .beta., growth related protein .gamma., heparin binding epidermal growth factor, hepatocyte growth factor, hepatocyte growth factor receptor, hepatoma-derived growth factor, insulin-like growth factor I, insulin-like growth factor receptor, insulin-like growth factor II, insulin-like growth factor binding protein, keratinocyte growth factor, leukemia inhibitory factor, leukemia inhibitory factor receptor .alpha., nerve growth factor nerve growth factor receptor, neuropoietin, neurotrophin-3, neurotrophin-4, oncostatin M (OSM), placenta growth factor, placenta growth factor 2, platelet-derived endothelial cell growth factor, platelet derived growth factor, platelet derived growth factor A chain, platelet derived growth factor AA, platelet derived growth factor AB, platelet derived growth factor B chain, platelet derived growth factor BB, platelet derived growth factor receptor .alpha., platelet derived growth factor receptor .beta., pre-B cell growth stimulating factor, stem cell factor (SCF), stem cell factor receptor, TNF, TNF0, TNF1, TNF2, transforming growth factor .alpha., transforming growth factor .beta., transforming growth factor .beta.1, transforming growth factor .beta.1.2, transforming growth factor .beta.2, transforming growth factor .beta.3, transforming growth factor .beta.5, latent transforming growth factor .beta.1, transforming growth factor .beta. binding protein I, transforming growth factor .beta. binding protein II, transforming growth factor .beta. binding protein III, thymic stromal lymphopoietin (TSLP), tumor necrosis factor receptor type I, tumor necrosis factor receptor type II, urokinase-type plasminogen activator receptor, phospholipase-activating protein (PUP), insulin, lectin ricin, prolactin, chorionic gonadotropin, follicle-stimulating hormone, thyroid-stimulating hormone, tissue plasminogen activator, IgG, IgE, IgM, IgA, and IgD, .alpha.-galactosidase, .beta.-galactosidase, DNAse, fetuin, leutinizing hormone, estrogen, insulin, albumin, lipoproteins, fetoprotein, transferrin, thrombopoietin, urokinase, integrin, thrombin, leptin, Humira (adalimumab), Prolia (denosumab), Enbrel (etanercept), a protein from Table 1, or a biologically active fragment, derivative or variant thereof; wherein said water soluble polymer containing an active hydrazide group is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); wherein the nucleophilic catalyst is selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0047] In another embodiment, an aforementioned method is provided wherein the water soluble polymer containing an active aminooxy group is prepared by a method comprising: incubating a solution comprising an oxidized water-soluble polymer with an activated aminooxy linker comprising an active aminooxy group under conditions that allow the formation of a stable oxime linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; thereby forming a water soluble polymer containing an active aminooxy group; and b) purifying the water soluble polymer containing an active aminooxy group by a method selected from the group consisting of chromatography, filtration and precipitation. The term "activated water-soluble polymer" refers, in one embodiment, to a water-soluble polymer containing an aldehyde group.
[0048] In yet another embodiment, an aforementioned method is provided wherein the water soluble polymer containing an active aminooxy group is prepared by a method comprising: a) incubating a solution comprising an oxidized water-soluble polymer with an activated aminooxy linker comprising an active aminooxy group under conditions that allow the formation of a stable oxime linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; thereby forming a water soluble polymer containing an active aminooxy group; b) incubating a solution comprising the water soluble polymer containing an active aminooxy group of step a) with a reducing agent under conditions that allow the formation of a stable alkoxamine linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring; and c) purifying the water soluble polymer containing an active aminooxy group by a method selected from the group consisting of chromatography, filtration and precipitation.
[0049] In still another embodiment, an aforementioned method is provided wherein the water soluble polymer containing an active aminooxy group is prepared by a method comprising: a) incubating a solution comprising an oxidized water-soluble polymer with an activated aminooxy linker comprising an active aminooxy group under conditions that allow the formation of a stable oxime linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; thereby forming a water soluble polymer containing an active aminooxy group; b) incubating a solution comprising the water soluble polymer containing an active aminooxy group of step a) with a nucleophilic catalyst under conditions comprising a time period between 1 minute and 24 hours; a temperature between 2.degree. C. and 37.degree. C.; in the presence or absence of light; and with or without stirring; and c) purifying the water soluble polymer containing an active aminooxy group by a method selected from the group consisting of chromatography, filtration and precipitation.
[0050] In yet another embodiment, an aforementioned method is provided wherein the water soluble polymer containing an active aminooxy group is prepared by a method comprising: a) incubating a solution comprising an oxidized water-soluble polymer with an activated aminooxy linker comprising an active aminooxy group under conditions that allow the formation of a stable oxime linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light, and with or without stirring; thereby forming a water soluble polymer containing an active aminooxy group; b) incubating a solution comprising the water soluble polymer containing an active aminooxy group of step a) with a nucleophilic catalyst under conditions comprising a time period between 1 minute and 24 hours; a temperature between 2.degree. C. and 37.degree. C.; in the presence or absence of light; and with or without stirring; c) incubating a solution comprising the water soluble polymer containing an active aminooxy group of step b) with a reducing agent under conditions that allow the formation of a stable alkoxamine linkage between the oxidized water-soluble polymer and the activated aminooxy linker, said conditions comprising a time period between about 1 minute and about 24 hours; a temperature between about 2.degree. C. and about 37.degree. C.; in the presence or absence of light; and with or without stirring; and d) purifying the water soluble polymer containing an active aminooxy group by a method selected from the group consisting of chromatography, filtration and precipitation.
[0051] In another embodiment, an aforementioned method is provided wherein the oxidized water soluble polymer is selected from the group consisting of polyethylene glycol (PEG), branched PEG, PolyPEG.RTM. (Warwick Effect Polymers; Coventry, UK), polysialic acid (PSA), starch, hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC), and wherein said water-soluble polymer is oxidized by incubation with a oxidizing agent to form a terminal aldehyde group at the non-reducing end of the water-soluble polymer. In one embodiment, the water-soluble polymer is PSA.
[0052] In another embodiment, an aforementioned method is provided wherein the oxidizing agent is NaIO4.
[0053] In still another embodiment, an aforementioned method is provided wherein the aminooxy linker is selected from the group consisting of: [0054] a) a 3-oxa-pentane-1,5-dioxyamine linker of the formula:
[0054] ##STR00004## [0055] b) a 3,6,9-trioxa-undecane-1,11-dioxyamine linker of the formula:
##STR00005##
[0055] and [0056] c) a 3,6,9,12,15-penatoxa-heptadecane-1,17-dioxyamine linker of the formula:
##STR00006##
[0057] In yet another embodiment, an aforementioned method is provided wherein the reducing agent is selected from the group consisting of sodium cyanoborohydride (NaCNBH3), ascorbic acid (vitamin C) and NaBH3. In one embodiment, the reducing agent is sodium cyanoborohydride (NaCNBH3).
[0058] In another embodiment, an aforementioned method is provided wherein the nucleophilic catalyst is selected from the group consisting of o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine. In one embodiment, the nucleophilic catalyst is m-toluidine. In another embodiment, the nucleophilic catalyst is added in an amount to result in a final concentration between about 1.0 mM and about 50 mM nucleophilic catalyst.
[0059] In another embodiment, an aforementioned method is provided further comprising concentrating the conjugated therapeutic protein by ultra-/diafiltration (UF/DF).
[0060] In another embodiment, a method of conjugating a water soluble polymer to an oxidized carbohydrate moiety of a blood coagulation protein is provided comprising contacting the oxidized carbohydrate moiety with an activated water soluble polymer under conditions that allow conjugation;
[0061] said blood coagulation protein selected from the group consisting of Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF) and ADAMTS 13 protease or a biologically active fragment, derivative or variant thereof;
[0062] said water soluble polymer containing an active aminooxy group and is selected from the group consisting of polyethylene glycol (PEG), branched PEG, polysialic acid (PSA), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC); and
[0063] said carbohydrate moiety oxidized by incubation with a buffer comprising an oxidizing agent selected from the group consisting of sodium periodate (NaIO4), lead tetraacetate (Pb(OAc)4) and potassium perruthenate (KRuO4); wherein an oxime linkage is formed between the oxidized carbohydrate moiety and the active aminooxy group on the water soluble polymer.
FIGURES
[0064] FIG. 1 shows the primary structure of coagulation Factor IX (SEQ ID NO: 1).
[0065] FIG. 2 shows the coupling of oxidized rFIX to aminooxy-PSA.
[0066] FIG. 3 shows the synthesis of the water soluble di-aminoxy linkers 3-oxa-pentane-1,5-dioxyamine and 3,6,9-trioxa-undecane-1,11-dioxyamine.
[0067] FIG. 4 shows the preparation of aminooxy-PSA.
[0068] FIG. 5 shows the visualization of PSA-FIX conjugates prepared in the presence of different catalysts by SDS PAGE. a) Comparison of aniline with m-toluidine using different concentrations; b) Comparison of aniline with o-aminobenzoic acid, m-aminobenzoic acid, p-aminobenzoic acid, p-aminobenzamide and sulfanilic acid; c) Comparison of aniline and m-toluidine with o-anisidine and m-anisidine.
[0069] FIG. 6 shows percent of polysialylation with various nucleophilic catalysts.
DETAILED DESCRIPTION OF THE INVENTION
[0070] The pharmacological and immunological properties of therapeutic proteins can be improved by chemical modification and conjugation with polymeric compounds such as polyethylene glycol (PEG), branched PEG, polysialic acid (PSA), hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG), polyoxazoline, polyacryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC). The properties of the resulting conjugates generally strongly depend on the structure and the size of the polymer. Thus, polymers with a defined and narrow size distribution are usually preferred in the art. Synthetic polymers like PEG can be manufactured easily with a narrow size distribution, while PSA can be purified in such a manner that results in a final PSA preparation with a narrow size distribution. In addition PEGylation reagents with defined polymer chains and narrow size distribution are on the market and commercially available for a reasonable price.
[0071] The addition of a soluble polymer, such as through polysialylation, is one approach to improve the properties of therapeutic proteins such as the blood coagulation protein FIX, as well as other coagulation proteins (e.g., VWF, FVIIa (see, e.g., US 2008/0221032A1, incorporated herein by reference) and FVIII).
Therapeutic Proteins
[0072] In certain embodiments of the invention, the aforementioned polypeptides and polynucleotides are exemplified by the following therapeutic proteins: enzymes, antigens, antibodies, receptors, blood coagulation proteins, growth factors, hormones, and ligands. In certain embodiments, the therapeutic protein is a blood coagulation protein such as Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF) or ADAMTS 13 protease. In one embodiment, a therapeutic protein according to the invention is a glycoprotein or, in various embodiments, a protein that is not naturally glycosylated in vivo (i.e., a protein that does not contain a natural glycosylation site or a protein that is not glycosylated in a host cell prior to purification).
[0073] In certain embodiments, the therapeutic protein is immunoglobulins, cytokines such IL-1 alpha, IL-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-11, colony stimulating factor-1 (CSF-1), M-CSF, SCF, GM-CSF, granulocyte colony stimulating factor (G-CSF), EPO, interferon-alpha (IFN-alpha), consensus interferon, IFN-beta, IFN-gamma, IFN-omega, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-14, IL-15, IL-16, IL-17, IL-18, IL-19, IL-20, IL-21, IL-22, IL-23, IL-24, IL-31, IL-32 alpha, IL-33, thrombopoietin (TPO), angiopoietins, for example Ang-1, Ang-2, Ang-4, Ang-Y, the human angiopoietin-like polypeptides ANGPTL1 through 7, vitronectin, vascular endothelial growth factor (VEGF), angiogenin, activin A, activin B, activin C, bone morphogenic protein-1, bone morphogenic protein-2, bone morphogenic protein-3, bone morphogenic protein-4, bone morphogenic protein-5, bone morphogenic protein-6, bone morphogenic protein-7, bone morphogenic protein-8, bone morphogenic protein-9, bone morphogenic protein-10, bone morphogenic protein-11, bone morphogenic protein-12, bone morphogenic protein-13, bone morphogenic protein-14, bone morphogenic protein-15, bone morphogenic protein receptor IA, bone morphogenic protein receptor IB, bone morphogenic protein receptor II, brain derived neurotrophic factor, cardiotrophin-1, ciliary neutrophic factor, ciliary neutrophic factor receptor, cripto, cryptic, cytokine-induced neutrophil chemotactic factor 1, cytokine-induced neutrophil, chemotactic factor 2a, cytokine-induced neutrophil chemotactic factor 2.beta.,.beta. endothelial cell growth factor, endothelin 1, epidermal growth factor, epigen, epiregulin, epithelial-derived neutrophil attractant, fibroblast growth factor 4, fibroblast growth factor 5, fibroblast growth factor 6, fibroblast growth factor 7, fibroblast growth factor 8, fibroblast growth factor 8b, fibroblast growth factor 8c, fibroblast growth factor 9, fibroblast growth factor 10, fibroblast growth factor 11, fibroblast growth factor 12, fibroblast growth factor 13, fibroblast growth factor 16, fibroblast growth factor 17, fibroblast growth factor 19, fibroblast growth factor 20, fibroblast growth factor 21, fibroblast growth factor acidic, fibroblast growth factor basic, glial cell line-derived neutrophic factor receptor .alpha.1, glial cell line-derived neutrophic factor receptor .alpha.2, growth related protein, growth related protein .alpha., growth related protein .beta., growth related protein .gamma., heparin binding epidermal growth factor, hepatocyte growth factor, hepatocyte growth factor receptor, hepatoma-derived growth factor, insulin-like growth factor I, insulin-like growth factor receptor, insulin-like growth factor II, insulin-like growth factor binding protein, keratinocyte growth factor, leukemia inhibitory factor, leukemia inhibitory factor receptor .alpha., nerve growth factor nerve growth factor receptor, neuropoietin, neurotrophin-3, neurotrophin-4, oncostatin M (OSM), placenta growth factor, placenta growth factor 2, platelet-derived endothelial cell growth factor, platelet derived growth factor, platelet derived growth factor A chain, platelet derived growth factor AA, platelet derived growth factor AB, platelet derived growth factor B chain, platelet derived growth factor BB, platelet derived growth factor receptor .alpha., platelet derived growth factor receptor 13, pre-B cell growth stimulating factor, stem cell factor (SCF), stem cell factor receptor, TNF, including TNF0, TNF1, TNF2, transforming growth factor .alpha., transforming growth factor 13, transforming growth factor 131, transforming growth factor 131.2, transforming growth factor .beta.2, transforming growth factor .beta.3, transforming growth factor .beta.5, latent transforming growth factor .beta.1, transforming growth factor .beta. binding protein I, transforming growth factor .beta. binding protein II, transforming growth factor .beta. binding protein III, thymic stromal lymphopoietin (TSLP), tumor necrosis factor receptor type I, tumor necrosis factor receptor type II, urokinase-type plasminogen activator receptor, vascular endothelial growth factor, and chimeric proteins and biologically or immunologically active fragments thereof.
[0074] In certain embodiments, the therapeutic protin is alpha-, beta-, and gamma-interferons, colony stimulating factors including granulocyte colony stimulating factors, fibroblast growth factors, platelet derived growth factors, phospholipase-activating protein (PUP), insulin, plant proteins such as lectins and ricins, tumor necrosis factors and related alleles, soluble forms of tumor necrosis factor receptors, interleukin receptors and soluble forms of interleukin receptors, growth factors such as tissue growth factors, such as TGF.alpha.s or TGF.beta.s and epidermal growth factors, hormones, somatomedins, pigmentary hormones, hypothalamic releasing factors, antidiuretic hormones, prolactin, chorionic gonadotropin, follicle-stimulating hormone, thyroid-stimulating hormone, tissue plasminogen activator, and immunoglobulins such as IgG, IgE, IgM, IgA, and IgD, a galactosidase, .alpha.-galactosidase, .beta.-galactosidase, DNAse, fetuin, leutinizing hormone, estrogen, corticosteroids, insulin, albumin, lipoproteins, fetoprotein, transferrin, thrombopoietin, urokinase, DNase, integrins, thrombin, hematopoietic growth actors, leptin, glycosidases, Humira (adalimumab), Prolia (denosumab), Enbrel (etanercept), and fragments thereof, or any fusion proteins comprising any of the above mentioned proteins or fragments thereof. In addition to the aforementioned proteins, the following Table 1 provides therapeutic proteins contemplated by the present invention:
TABLE-US-00001 TABLE 1 Follicular dendritic cell secreted peptide Angiotensin-converting enzyme Dermokine Antithrombin-III Secreted frizzled-related protein 1 Apolipoprotein B-100 Ectodysplasin-A Apolipoprotein D Secreted frizzled-related protein 2 Apolipoprotein E Resistin Beta-1,4-galactosyltransferase 1 Osteopontin Bone morphogenetic protein 7 Secreted frizzled-related protein 5 Complement C1q subcomponent subunit B Secreted frizzled-related protein 4 C4b-binding protein alpha chain Secreted phosphoprotein 24 Calreticulin Glypican-6 Corticosteroid-binding globulin Secreted frizzled-related protein 3 Carboxypeptidase A1 C-C motif chemokine 4 Carboxypeptidase A2 Melanocyte protein Pmel 17 Eotaxin Secreted Ly-6/uPAR-related protein 1 C-C motif chemokine 13 Beta-microseminoprotein C-C motif chemokine 18 Glypican-4 C-C motif chemokine 20 Tumor necrosis factor ligand superfamily Triggering receptor expressed on myeloid member 15 cells 2 Resistin-like beta C-C motif chemokine 2 Tumor necrosis factor ligand superfamily Transforming growth factor-beta-induced member 12 protein ig-h3 SPARC CD40 ligand Glypican-5 Corneodesmosin Anterior gradient protein 2 homolog Complement factor D Protein canopy homolog 2 Chromogranin-A Glypican-1 Collagen alpha-1(I) chain von Willebrand factor A domain-containing Disintegrin and metalloproteinase domain- protein 2 containing protein 18 WNT1-inducible-signaling pathway Cysteine-rich secretory protein LCCL protein 1 domain-containing 1 C-C motif chemokine 1 Collagen alpha-4(IV) chain SPARC-related modular calcium-binding Keratinocyte differentiation-associated protein 2 protein C-type lectin domain family 11 member A Complement C4-B Secreted Ly-6/uPAR-related protein 2 Collagen alpha-2(V) chain Glypican-3 Complement C5 Secreted and transmembrane protein 1 Collagen alpha-1(VII) chain Testis-expressed sequence 264 protein Complement component C7 Glypican-2 Complement component C8 beta chain Serine protease 23 Complement component C8 gamma chain 39S ribosomal protein L55, mitochondrial Collagen alpha-1(XV) chain Protein NipSnap homolog 3A Collagen alpha-1(XVI) chain Fibronectin Collagen alpha-1(XVIII) chain Neudesin Collagen alpha-1(XIX) chain Fibroblast growth factor receptor 2 Cartilage oligomeric matrix protein Carbonic anhydrase 6 C-reactive protein Deleted in malignant brain tumors 1 protein Granulocyte colony-stimulating factor SPARC-related modular calcium-binding Granulocyte-macrophage colony- protein 1 stimulating factor Amyloid beta A4 protein Protein CYR61 Tumor necrosis factor receptor superfamily Complement component receptor 1-like member 6 protein Gamma-aminobutyric acid type B receptor Stem cell growth factor; lymphocyte subunit 1 secreted C-type lectin Pro-neuregulin-1, membrane-bound CMP-N-acetylneuraminate-beta- isoform galactosamide-alpha-2,3-sialyltransferase Glycoprotein hormone alpha-2 Dipeptidyl peptidase 4 Membrane metallo-endopeptidase-like 1 Dentin sialophosphoprotein Fc receptor-like A Endothelin-1 C-C motif chemokine 4-like Ephrin-B1 Epithelial discoidin domain-containing Epidermis-specific serine protease-like receptor 1 protein Mucin-1 EMILIN-1 Vascular endothelial growth factor A Endoplasmin Fibulin-1 Ephrin type-A receptor 3 Prolactin receptor Ephrin type-B receptor 6 Proprotein convertase subtilisin/kexin Glycosyltransferase 1 domain-containing type 6 protein 1 CD209 antigen Coagulation factor X Collagen alpha-2(XI) chain Coagulation factor VIII Granulocyte-macrophage colony- Complement C1q tumor necrosis factor- stimulating factor receptor subunit alpha related protein 7 Elastin Fibrillin-2 Interleukin-15 receptor subunit alpha Alpha-2-HS-glycoprotein Midkine Fibroblast growth factor 10 Integrin alpha-7 Fibrinogen alpha chain Mucin-4 Fibrinogen beta chain Peptidyl-glycine alpha-amidating Long palate, lung and nasal epithelium monooxygenase carcinoma-associated protein 1 Apolipoprotein A-I Gastrin Proteoglycan 4 Glycoprotein hormones alpha chain Tumor necrosis factor receptor superfamily N-acetylglucosamine-1- member 25 phosphotransferase subunits alpha/beta Attractin Granzyme A Prostate-associated microseminoprotein Hepatocyte growth factor-like protein Alpha-amylase 1 Insulin-like growth factor-binding protein 1 Brain-derived neurotrophic factor Insulin-like growth factor-binding protein 2 C-type lectin domain family 4 member M Insulin-like growth factor-binding protein 4 Granulocyte colony-stimulating factor Tumor necrosis factor receptor superfamily receptor member 10D Insulin-like growth factor II Interferon alpha-1/13 Carcinoembryonic antigen-related cell Interferon-induced helicase C domain- adhesion molecule 1 containing protein 1 C-type lectin domain family 7 member A Interferon alpha-2 CMRF35-like molecule 1 Interferon beta Choline transporter-like protein 4 Interferon gamma Pulmonary surfactant-associated protein A1 Insulin-like growth factor IB Spermine oxidase Indian hedgehog protein CMP-N-acetylneuraminate-beta-1,4- Neural cell adhesion molecule galactoside alpha-2,3-sialyltransferase L1-like protein Kallikrein-8 Interleukin-13 Tissue-type plasminogen activator Interleukin-2 Peroxisomal N(1)-acetyl- Chymotrypsin-like elastase family spermine/spermidine oxidase member 2A Probable palmitoyltransferase ZDHHC4 Inhibin beta A chain Cholesteryl ester transfer protein Pancreatic secretory trypsin inhibitor HLA class I histocompatibility antigen, A-2 Tumor necrosis factor receptor superfamily alpha chain member 21 Collagen alpha-1(II) chain Inter-alpha-trypsin inhibitor heavy chain H1 Pro-interleukin-16 Inter-alpha-trypsin inhibitor heavy chain H2 Leptin receptor Inter-alpha-trypsin inhibitor heavy chain H3 Decorin Prostate-specific antigen Stromal cell-derived factor 1 Kallikrein-4 Tenascin Plasma kallikrein Disintegrin and metalloproteinase domain- Calcium-activated chloride channel containing protein 12 regulator 4 A disintegrin and metalloproteinase with Bactericidal/permeability-increasing thrombospondin motifs 13 protein-like 1 T-cell surface glycoprotein CD3 alpha chain Leptin EGFR-coamplified and overexpressed A disintegrin and metalloproteinase with protein thrombospondin motifs 4 Autophagy-related protein 16-1 Hepatic triacylglycerol lipase Breast cancer anti-estrogen resistance Lymphocyte antigen 6 complex locus protein 3 protein G6c Cadherin-23 Eosinophil lysophospholipase Macrophage colony-stimulating factor 1 Lutropin subunit beta Folate receptor alpha Microfibrillar-associated protein 1 Low-density lipoprotein receptor-related Mesencephalic astrocyte-derived protein 8 neurotrophic factor E3 ubiquitin-protein ligase LRSAM1 Matrix Gla protein Neural cell adhesion molecule 1 72 kDa type IV collagenase Neuroligin-4, X-linked Stromelysin-1 Netrin-G1 Neutrophil collagenase GPI transamidase component PIG-T Mesothelin Kit ligand Mucin-5AC Seizure 6-like protein Mucin-6 SLAM family member 7 Norrin Tumor necrosis factor Oxytocin-neurophysin 1 Uromodulin Beta-nerve growth factor Tumor necrosis factor ligand superfamily Tumor necrosis factor ligand superfamily member 13 member 18 Protein CREG1 Neurotrophin-3 EGF-like domain-containing protein 8 Platelet-derived growth factor subunit A Aminoacyl tRNA synthetase complex- Phosphopantothenoylcysteine interacting multifunctional protein 1 decarboxylase ADAMTS-like protein 4 Plasminogen activator inhibitor 1 Coagulation factor XI Plasminogen activator inhibitor 2 Interleukin-22 receptor subunit alpha-2 Procollagen C-endopeptidase enhancer 1 Deformed epidermal autoregulatory factor 1 Transmembrane and ubiquitin-like domain- homolog containing protein 2 Prostaglandin-H2 D-isomerase Protein disulfide-isomerase Alpha-1-antitrypsin Pigment epithelium-derived factor Alpha-1-antichymotrypsin Pepsin A Acyl-CoA-binding protein Gastricsin Complement factor B Sonic hedgehog protein Choriogonadotropin subunit beta Peptidoglycan recognition protein I-alpha Versican core protein Biglycan Epidermal growth factor receptor Prolactin-inducible protein Ecto-NOX disulfide-thiol exchanger 2 Platelet factor 4 Hyaluronidase-1 Plasminogen Interleukin-1 receptor antagonist protein Serum paraoxonase/arylesterase 1 Interleukin-6 receptor subunit beta Alkaline phosphatase, placental type Interleukin-1 receptor-like 1 Peptidyl-prolyl cis-trans isomerase B Insulin Bone marrow proteoglycan Glycodelin Basic salivary proline-rich protein 1 Parathyroid hormone-related protein Pulmonary surfactant-associated protein C Nurim Parathyroid hormone Prolyl 4-hydroxylase subunit alpha-2 Serum amyloid P-component CD276 antigen Secretogranin-1 Cysteine-rich with EGF-like domain Basement membrane-specific heparan protein 1 sulfate proteoglycan core protein CUB and sushi domain-containing protein 1 Antileukoproteinase Ficolin-2 Stabilin-1 Fc receptor-like protein 5 Extracellular superoxide dismutase [Cu--Zn] Protein GPR89 Somatotropin Junctional adhesion molecule A Serpin B5 Leucine-rich repeat-containing protein 8A Spondin-1 Multiple inositol polyphosphate Structural maintenance of chromosomes phosphatase 1 protein 3 Neuropilin-1 Syntaxin-1A Plexin-A4 Tetranectin Plexin-B1 Transforming growth factor beta-1 Periostin Thyroglobulin Protein RIC-3 Metalloproteinase inhibitor 1 SLIT and NTRK-like protein 2 Metalloproteinase inhibitor 2 Sulfatase-modifying factor 1 Metalloproteinase inhibitor 3 Sulfatase-modifying factor 2 Urokinase-type plasminogen activator Transmembrane protease, serine 6 Lactotransferrin Lymphotoxin-alpha Trypsin-1 Tumor necrosis factor receptor superfamily Submaxillary gland androgen-regulated member 10B protein 3B Urokinase plasminogen activator surface Tumor necrosis factor receptor superfamily receptor member 1A V-set domain-containing T-cell activation Vascular endothelial growth factor inhibitor 1 receptor 1 Glucagon Vitamin D-binding protein N-acetylmuramoyl-L-alanine amidase Vitronectin Sulfhydryl oxidase 1 von Willebrand factor Dehydrogenase/reductase SDR family Lymphocyte antigen 6 complex locus member 4 protein G5c Interleukin-18-binding protein Zinc-alpha-2-glycoprotein Kin of IRRE-like protein 2 Uncharacterized protein C14orf93 Myeloid-associated differentiation marker Retinoschisin Chordin Alpha-1,3-mannosyltransferase ALG2 1-acyl-sn-glycerol-3-phosphate C-type lectin domain family 11, member A, acyltransferase gamma isoform CRA_b Advanced glycosylation end product- Major facilitator superfamily domain- specific receptor containing protein 7 NLR family CARD domain-containing Leucine-rich repeat transmembrane protein 4 neuronal protein 1 Pro-neuregulin-2, membrane-bound NADH dehydrogenase [ubiquinone] 1 beta isoform subcomplex subunit 11, mitochondrial Sperm-associated antigen 11A UPF0546 membrane protein C1orf91 Oocyte-secreted protein 1 homolog Carbonic anhydrase-related protein 10 Serum albumin Cholecystokinin Cochlin Codanin-1 Plasma protease C1 inhibitor Uncharacterized protein C6orf89 Interleukin-7 receptor subunit alpha Chondroitin sulfate glucuronyltransferase Inter-alpha-trypsin inhibitor heavy chain H5 Chitinase domain-containing
protein 1 Platelet-derived growth factor D Transmembrane protein C9orf7 Protein S100-A7 CMRF35-like molecule 9 Sialic acid-binding Ig-like lectin 10 Cytochrome P450 2S1 Tubulointerstitial nephritis antigen-like Crumbs protein homolog 3 Tumor necrosis factor ligand superfamily Dehydrogenase/reductase SDR family member 13B member 7 Long-chain-fatty-acid--CoA ligase 5 Protein ENED Claudin-14 Complement factor H-related protein 4 Leucine-rich repeat-containing protein 20 Leucine-rich repeat LGI family member 3 Interleukin-1 family member 7 Gliomedin Lymphocyte antigen 6 complex locus Glycerophosphodiester phosphodiesterase protein G5b domain-containing protein 5 Acetylcholinesterase Probable G-protein coupled receptor 113 Amelogenin, X isoform Probable G-protein coupled receptor 114 Angiogenin Glycerol-3-phosphate acyltransferase 4 Anthrax toxin receptor 2 Gremlin-1 Annexin A2 Potassium channel subfamily K member 17 Apolipoprotein C-III KDEL motif-containing protein 2 Apolipoprotein L1 Layilin Complement C1q subcomponent subunit A Leucine-rich repeat-containing protein 8B Complement C1q subcomponent subunit C Leucine-rich repeat-containing protein 8D Calcitonin Sialic acid-binding Ig-like lectin 6 Soluble calcium-activated nucleotidase 1 Pregnancy-specific beta-1-glycoprotein 2 C-C motif chemokine 15 Ly6/PLAUR domain-containing protein 1 CD97 antigen ( Ly6/PLAUR domain-containing protein 5 Contactin-4 MLN64 N-terminal domain homolog Complement C2 Macrophage migration inhibitory factor Collagen alpha-6(IV) chain 2-acylglycerol O-acyltransferase 3 Collagen alpha-2(VI) chain Mitochondrial carrier homolog 1 Collagen alpha-1(XI) chain Apolipoprotein L6 Crumbs homolog 1 Protocadherin alpha-6 Cystatin-C Protocadherin gamma-A12 Neutrophil defensin 1 Voltage-gated hydrogen channel 1 Endothelin-3 All-trans-retinol 13,14-reductase Low affinity immunoglobulin epsilon Regulator of microtubule dynamics Fc receptor protein 2 Fibroblast growth factor receptor 3 R-spondin-4 Fibroblast growth factor receptor 4 Long-chain fatty acid transport protein 3 Growth arrest-specific protein 6 Vesicle-trafficking protein SEC22c Growth hormone receptor Claudin-1 Bifunctional UDP-N-acetylglucosamine 2- Leucine-rich repeats and immunoglobulin- epimerase/N-acetylmannosamine kinase like domains protein 3 Immunoglobulin superfamily member 8 SLAM family member 9 Interleukin-4 receptor alpha chain Transthyretin Kallikrein-14 Serine/threonine-protein kinase 32B Kallikrein-6 Platelet-derived growth factor subunit B Laminin subunit beta-3 Noggin Leucyl-cystinyl aminopeptidase Tryptase alpha-1 Mannan-binding lectin serine protease 1 Tetratricopeptide repeat protein 14 Mannan-binding lectin serine protease 2 XTP3-transactivated gene B protein Neutrophil gelatinase-associated lipocalin Palmitoyltransferase ZDHHC15 Neuropeptide Y Zona pellucida sperm-binding protein 3 Aggrecan core protein Leucine-rich repeat-containing protein 39 Pulmonary surfactant-associated protein B Pancreatic triacylglycerol lipase Poliovirus receptor-related protein 1 Transmembrane protein 139 Renin Leukemia inhibitory factor Ribonuclease pancreatic Galectin-1 Semenogelin-1 C-C motif chemokine 21 Signaling lymphocytic activation molecule CD5 antigen-like Tissue factor pathway inhibitor Carbohydrate sulfotransferase 9 Usherin Lipopolysaccharide-binding protein Fibroblast growth factor 23 Cysteine-rich motor neuron 1 protein Interleukin-23 subunit alpha Connective tissue growth factor Epididymal secretory protein E1 Protein eyes shut homolog ADAMTS-like protein 1 Mucin-like protein 1 Chemokine-like factor Fibroblast growth factor 19 EGF-like domain-containing protein 7 Follistatin-related protein 3 Tectonic-1 Hedgehog-interacting protein Transmembrane protein 25 Interleukin-17 receptor B UDP-GalNAc: beta-1,3-N- FXYD domain-containing ion transport acetylgalactosaminyltransferase 1 regulator 5 Interleukin-15 (IL-15) Endothelial lipase Multiple epidermal growth factor-like EGF-containing fibulin-like extracellular domains 11 matrix protein 2 Mucin and cadherin-like protein Otoraplin Ribonuclease 4 Group 3 secretory phospholipase A2 SH2 domain-containing protein 3C Group XV phospholipase A2 CMP-N-acetylneuraminate-beta- Tumor necrosis factor ligand superfamily galactosamide-alpha-2,3-sialyltransferase member 14 Transmembrane protein 9 Plexin-A2 WAP four-disulfide core domain protein 2 Papilin Adenosine A3 receptor Prokineticin-1 Gamma-secretase subunit APH-1A Ribonuclease 7 Basigin Kunitz-type protease inhibitor 1 Baculoviral IAP repeat-containing protein 7 Spondin-2 Calumenin Testican-2 Alpha-S1-casein Inactive serine protease PAMR1 Cyclin-L1 Torsin-2A Complement factor H Vasohibin-1 Chorionic somatomammotropin hormone Vasorin Coxsackievirus and adenovirus receptor Xylosyltransferase 1 Ectonucleotide Ectonucleotide pyrophosphatase/phosphodiesterase family pyrophosphatase/phosphodiesterase family member 2 member 6 ERO1-like protein alpha Oncostatin-M Coagulation factor IX Derlin-1 Low affinity immunoglobulin gamma Fc HERV-FRD_6p24.1 provirus ancestral Env region receptor III-B polyprotein Ficolin-3 Prostasin Fc receptor-like protein 2 Transmembrane protease, serine 11E Leucine-rich repeat transmembrane protein HLA class I histocompatibility antigen, FLRT3 Cw-16 alpha chain Gelsolin Wnt inhibitory factor 1 Granulysin C-type natriuretic peptide Transmembrane glycoprotein NMB Angiopoietin-2 Granulins Deoxyribonuclease gamma Heparanase Carboxypeptidase A5 Ig mu chain C region C-C motif chemokine 14 Interleukin-1 alpha Interleukin-5 Interleukin-31 receptor A Interleukin-10 Junctional adhesion molecule B C-X-C motif chemokine 2 Lipocalin-1 C-X-C motif chemokine 5 Leucine-rich repeat-containing G-protein A disintegrin and metalloproteinase with coupled receptor 6 thrombospondin motifs 6 Latent-transforming growth factor beta- Polypeptide binding protein 1 N-acetylgalactosaminyltransferase 1 Matrilin-3 Fibulin-2 Myelin protein zero-like protein 1 Ficolin-1 Neurobeachin-like protein 2 SL cytokine Nicastrin Follistatin ADP-ribose pyrophosphatase, FRAS1-related extracellular matrix mitochondrial protein 1 Protocadherin-15 Enamelin Placenta growth factor Hyaluronan and proteoglycan link protein 1 Protein O-linked-mannose beta-1,2-N- Leukocyte immunoglobulin-like receptor acetylglucosaminyltransferase 1 subfamily A member 3 Probable hydrolase PNKD Interleukin-17F Pleiotrophin Interleukin-1 receptor accessory protein Poliovirus receptor Serine protease inhibitor Kazal-type 5 Reticulon-4 receptor Kallikrein-15 Serum amyloid A protein Interferon alpha-14 Sex hormone-binding globulin Pregnancy-specific beta-1-glycoprotein 4 SLAM family member 6 Collagenase 3 Sarcolemmal membrane-associated protein Matrix metalloproteinase-16 Sushi, von Willebrand factor type A, EGF Pituitary adenylate cyclase-activating and pentraxin domain-containing protein 1 polypeptide Thyroxine-binding globulin Prokineticin-2 Transmembrane and coiled-coil domain- Latent-transforming growth factor beta- containing protein 1 binding protein 3 Transmembrane protease, serine 3 Somatoliberin Tumor necrosis factor receptor superfamily Thrombospondin type-1 domain-containing member 10C protein 1 Tumor necrosis factor receptor superfamily Angiogenic factor with G patch and FHA member 11B domains 1 Serotransferrin TGF-beta receptor type III Tryptase beta-2 Thyrotropin subunit beta Protein YIPF5 Uncharacterized protein C19orf36 Vesicle-associated membrane protein- Complement C1q tumor necrosis associated protein B/C factor-related protein 2 cDNA, FLJ96669, highly similar to Homo Ectonucleotide sapiens secreted protein, acidic, cysteine- pyrophosphatase/phosphodiesterase family rich (osteonectin)(SPARC), mRNA member 5 cDNA FLJ77519, highly similar to Homo Polypeptide N- sapiens secreted frizzled related protein acetylgalactosaminyltransferase-like mRNA protein 2 T-cell differentiation antigen CD6 Slit homolog 1 protein Pikachurin Growth hormone variant Fibrinogen-like protein 1 Angiopoietin-related protein 3 Interleukin-32 Angiopoietin-related protein 7 Matrilin-4 Ecto-ADP-ribosyltransferase 5 Sperm-associated antigen 11B Carbonic anhydrase-related protein 11 Coagulation factor XII Probable ribonuclease 11 Hepcidin Probable carboxypeptidase X1 Klotho Protein FAM3D Serglycin C-X-C motif chemokine 14 Tomoregulin-2 Beta-defensin 127 Chordin-like protein 2 Beta-defensin 129 Tumor necrosis factor receptor superfamily Cysteine-rich secretory protein LCCL member 6B domain-containing 2 UPF0414 transmembrane protein C20orf30 Fibroblast growth factor 21 C-type lectin domain family 4 member C Plasma alpha-L-fucosidase UPF0317 protein C14orf159, mitochondrial Gastrokine-1 Netrin-G2 Gastrokine-2 Metalloreductase STEAP2 Glutathione peroxidase 7 Sushi domain-containing protein 4 HHIP-like protein 1 Protein YIF1B Interferon kappa Apolipoprotein M Apolipoprotein C-I C4b-binding protein beta chain Procollagen C-endopeptidase enhancer 2 T-cell surface glycoprotein CD8 beta chain Left-right determination factor 1 C-C motif chemokine 3-like 1 Leucine-rich repeat LGI family member 4 Fibroblast growth factor 8 BRCA1-A complex subunit Abraxas Sialomucin core protein 24 Leucine zipper protein 2 Programmed cell death 1 ligand 2 Neurexophilin-3 Secreted and transmembrane 1 Osteomodulin Complement C1q tumor necrosis factor- Kazal-type serine protease inhibitor related protein 6 domain-containing protein 1 EGF-like module-containing mucin-like Sperm acrosome membrane-associated hormone receptor-like 3 protein 3 Noelin-3 Secretoglobin family 3A member 1 Odorant-binding protein 2b Tsukushin Urotensin-2 Claudin-2 (SP82) Vitrin Complement factor H-related protein 2 WNT1-inducible-signaling pathway Immunoglobulin superfamily containing protein 3 leucine-rich repeat protein cDNA FLJ75759, highly similar to Homo Leucine-rich repeat and immunoglobulin- sapiens follistatin-like 3 (secreted like domain-containing nogo receptor- glycoprotein) (FSTL3), mRNA interacting protein 1 Angiotensin-converting enzyme 2 Kin of IRRE-like protein 3 Adiponectin Hematopoietic cell signal transducer Angiopoietin-related protein 4 Follitropin subunit beta Apolipoprotein A-V Melanoma inhibitory activity protein 3 Asporin Leucine-rich repeat-containing protein 4 Bactericidal permeability-increasing protein Zinc transporter 5 CUB domain-containing protein 1 Leucine-rich repeat neuronal protein 1 Cartilage intermediate layer protein 1 Apical endosomal glycoprotein Beta-Ala-His dipeptidase Serum amyloid A-4 protein Collagen alpha-1(V) chain Probetacellulin Collagen alpha-1(XXV) chain Beta-1,4-galactosyltransferase 7 Estradiol 17-beta-dehydrogenase 11 3-hydroxybutyrate dehydrogenase type 2 DnaJ homolog subfamily C member 10 C1GALT1-specific chaperone 1 EGF-like domain-containing protein 6 Beta-casein Coagulation factor XIII A chain Kappa-casein Glucose-6-phosphate isomerase Transmembrane protein C2orf18 Appetite-regulating hormone Carboxypeptidase N catalytic chain Interleukin-12 subunit beta CD320 antigen Interleukin-22 Chondroitin sulfate synthase 1 lntelectin-1 Chondroitin sulfate synthase 2 Leucine-rich glioma-inactivated protein 1 CMRF35-like molecule 7 Lymphocyte antigen 96 Protein canopy homolog 3 Matrilysin Short-chain dehydrogenase/reductase 3 Mucin-20 Delta-like protein 4 Proprotein convertase subtilisin/kexin Delta and Notch-like epidermal
growth type 9 factor-related receptor Peptidoglycan recognition protein Dolichol kinase Interferon-induced 17 kDa protein Endothelin-converting enzyme-like 1 Protein Wnt-4 Integral membrane protein 2B Allograft inflammatory factor 1-like Insulin-like growth factor-binding protein 5 Armadillo repeat-containing X-linked Endothelial cell-selective adhesion protein 3 molecule Chondroitin sulfate N- Signal peptide, CUB and EGF-like domain- acetylgalactosaminyltransferase 1 containing protein 1 Chitotriosidase-1 Complement factor H-related protein 3 Claudin domain-containing protein 1 Prorelaxin H1 Erlin-2 Follistatin-related protein 1 Glycosyltransferase 8 domain-containing Globoside alpha-1,3-N- protein 1 acetylgalactosaminyltransferase 1 Golgi membrane protein 1 Gamma-glutamyl hydrolase Probable G-protein coupled receptor 125 Cadherin-24 Interleukin-20 receptor alpha chain Glycerol-3-phosphate acyltransferase 3 Galectin-7 G-protein coupled receptor 56 NKG2D ligand 4 Hyaluronan-binding protein 2 L-amino-acid oxidase Proheparin-binding EGF-like growth factor Prolyl 3-hydroxylase 1 Histidine-rich glycoprotein GPI ethanolamine phosphate transferase 2 Carbohydrate sulfotransferase 14 GPI ethanolamine phosphate transferase 3 Interleukin-20 receptor beta chain Calcium-binding mitochondrial carrier Ectonucleotide protein SCaMC-2 (Small calcium-binding pyrophosphatase/phosphodiesterase mitochondrial carrier protein 2) family member 3 Pulmonary surfactant-associated protein A2 Insulin-like growth factor-binding protein 7 Splicing factor, arginine/serine-rich 16 Kallistatin Alpha-N-acetylgalactosaminide alpha-2,6- Fibronectin type III domain-containing sialyltransferase 6 protein 3B Single Ig IL-1-related receptor Leukemia inhibitory factor receptor Tectonic-3 Lin-7 homolog B Tumor necrosis factor ligand superfamily Thioredoxin-related transmembrane member 11 protein 1 Tumor necrosis factor receptor superfamily Disintegrin and metalloproteinase domain- member 19 containing protein 32 Palmitoyltransferase ZDHHC9 Ly6/PLAUR domain-containing protein 3 Fibulin-5 C-type lectin domain family 14 member A Protein Z-dependent protease inhibitor Protein cornichon homolog Alpha-2-macroglobulin Protein FAM151A Agouti-related protein FK506-binding protein 14 Pancreatic alpha-amylase Neuropilin and tolloid-like protein 2 Natriuretic peptides B Protocadherin beta-13 Atrial natriuretic factor Prenylcysteine oxidase 1 Neutral ceramidase Peflin Beta-2-microglobulin Peptidyl-prolyl cis-trans isomerase-like 1 Bone morphogenetic protein 4 Prostate stem cell antigen Biotinidase Protein patched homolog 2 Scavenger receptor cysteine-rich type 1 Chitobiosyldiphosphodolichol beta- protein M130 mannosyltransferase Carboxypeptidase B2 Protein sel-1 homolog 1 Carboxypeptidase Z ProSAAS C-C motif chemokine 5 Sialic acid-binding Ig-like lectin 9 C-C motif chemokine 7 SLIT and NTRK-like protein 1 C-C motif chemokine 8 Statherin CD59 glycoprotein Testisin Complement factor I Transmembrane channel-like protein 5 Clusterin Transmembrane protease, serine 4 Collagen alpha-2(I) chain Metastasis-suppressor KiSS-1 Collagen alpha-1(III) chain Islet amyloid polypeptide Collagen alpha-1(IV) chain Trem-like transcript 2 protein Collagen alpha-3(IV) chain Thioredoxin domain-containing protein 12 Collagen alpha-5(IV) chain Vascular endothelial growth factor B Collagen alpha-3(VI) chain Vascular endothelial growth factor C Complement component C6 Reticulocalbin-3 Collagen alpha-1(IX) chain Fibrillin-1 Collagen alpha-1(X) chain Protein FAM3A Collagen alpha-1(XVII) chain Protein G7c Collagen alpha-1(XXI) chain Neuropilin and tolloid-like protein 1 Coatomer subunit alpha Pregnancy-specific beta-1-glycoprotein 11 Complement receptor type 1 Serpin B4 Cystatin-SN ADAM DEC1 Deoxyribonuclease-1 ADP-dependent glucokinase Extracellular matrix protein 1 Alpha-amylase 2B Low affinity immunoglobulin gamma UDP-GlcNAc: betaGal beta-1,3-N- Fc region receptor III-A acetylglucosaminyltransferase 3 Alpha-fetoprotein Calcitonin gene-related peptide 2 Heparin-binding growth factor 2 Carboxypeptidase E Fibrinogen gamma chain Cardiotrophin-like cytokine factor 1 Growth/differentiation factor 5 Collagen alpha-2(VIII) chain Glial cell line-derived neurotrophic factor Crumbs homolog 2 Insulin-like growth factor-binding protein 3 Dentin matrix acidic phosphoprotein 1 Insulin-like growth factor IA Down syndrome cell adhesion molecule Ig gamma-1 chain C region Immunoglobulin superfamily member 1 Ig gamma-2 chain C region Interleukin-4 Ig gamma-3 chain C region Interleukin-6 receptor subunit alpha Insulin-like 3 Interleukin-24 Inter-alpha-trypsin inhibitor heavy chain Ladinin-1 UPF0378 protein KIAA0100 Lipase member I Kininogen-1 Pancreatic lipase-related protein 1 Laminin subunit alpha-2 Leucine-rich alpha-2-glycoprotein Laminin subunit alpha-4 Matrix-remodeling-associated protein 5 Laminin subunit beta-1 Netrin-4 Protein-lysine 6-oxidase Hepatocyte growth factor receptor Multimerin-1 C-C motif chemokine 22 Vasopressin-neurophysin 2-copeptin Nyctalopin Nidogen-1 Osteocalcin Phospholipase A2, Basic salivary proline-rich protein 3 Perforin-1 Pregnancy-specific beta-1-glycoprotein 10 Phosphatidylinositol-glycan-specific Leucine-rich repeat transmembrane protein phospholipase D FLRT2 Fibrocystin R-spondin-3 Phospholipid transfer protein Sialoadhesin Prostatic acid phosphatase Trypsin-3 Vitamin K-dependent protein Z Dipeptidase 2 Salivary acidic proline-rich Collagen and calcium-binding EGF phosphoprotein 1/2 domain-containing protein 1 Pregnancy zone protein Germ cell-specific gene 1-like protein Prorelaxin H2 Leucine-rich repeat-containing protein 31 Semaphorin-4D Apolipoprotein O Slit homolog 2 protein Dystroglycan Alpha-tectorin Neutrophil defensin 4 Tenascin-X Amphoterin-induced protein 3 Trefoil factor 3 Gamma-secretase subunit APH-1B Transferrin receptor protein 1 Apolipoprotein C-IV Protransforming growth factor alpha Arylsulfatase G Transforming growth factor beta-2 Glia-activating factor Tumor necrosis factor ligand superfamily Caspase recruitment domain-containing member 6 protein 18 Tumor necrosis factor receptor superfamily Heparan sulfate glucosamine 3-O- member 1B sulfotransferase 3A1 Tumor necrosis factor receptor superfamily Thyrotropin-releasing hormone-degrading member 5 ectoenzyme Thrombopoietin Guanylin VIP peptides Choline transporter-like protein 3 Acidic mammalian chitinase 17-beta-hydroxysteroid dehydrogenase 14 Cysteine-rich secretory protein 2 Immunoglobulin lambda-like polypeptide 1 Haptoglobin-related protein DnaJ homolog subfamily B member 14 C-C motif chemokine 26 F-box only protein 8 Collectin-11 Fibroleukin Cysteine-rich with EGF-like domain Methionine-R-sulfoxide reductase B3, protein 2 mitochondrial C-X-C motif chemokine 16 Leucine-rich repeat LGI family member 2 Fibroblast growth factor-binding protein 1 Vesicle transport protein GOT1B Interleukin-1 family member 5 Integral membrane protein GPR177 Interleukin-1 family member 9 Probable G-protein coupled receptor 78 Kallikrein-5 HEPACAM family member 2 Matrilin-2 Interleukin-27 receptor subunit alpha Cell surface glycoprotein CD200 receptor 1 Proenkephalin-A Lysophosphatidic acid phosphatase type 6 Integrin alpha-10 Nucleotide exchange factor SIL1 KIEL motif-containing protein 1 Thrombospondin type-1 domain-containing Leukocyte immunoglobulin-like receptor protein 4 subfamily A member 5 WNT1-inducible-signaling pathway protein Leucine-rich repeat and fibronectin type-III 2 domain-containing protein 3 Bromodomain-containing protein 9 Uteroglobin CD99 antigen-like protein 2 Netrin-G1 ligand Uncharacterized protein C1orf159 Pannexin-1 Carbohydrate sulfotransferase 12 Protocadherin-12 Probable serine carboxypeptidase CPVL Protocadherin alpha-10 Mucin-3A Protocadherin beta-10 CUB and zona pellucida-like domain- Osteopetrosis-associated transmembrane containing protein 1 protein 1 Polypeptide N- Beta-galactoside alpha-2,6- acetylgalactosaminyltransferase 14 sialyltransferase 1 Galectin-9 GPI transamidase component PIG-S Leucine-rich repeat-containing protein 17 Proline-rich transmembrane protein 3 Leucine-rich repeat neuronal protein 2 Sulfhydryl oxidase 2 Bifunctional heparan sulfate N- A disintegrin and metalloproteinase with deacetylase/N-sulfotransferase 3 thrombospondin motifs 16 Tuftelin SH2 domain-containing protein 3A Brain mitochondrial carrier protein SHC-transforming protein 4 Signal peptide, CUB and EGF-like domain- Disintegrin and metalloproteinase domain- containing protein 3 containing protein 23 14-3-3 protein sigma Transducin beta-like protein 2 Alpha-1-acid glycoprotein 1 Tudor domain-containing protein 10 Alpha-1-acid glycoprotein 2 Transmembrane 9 superfamily member 3 von Willebrand factor A domain-containing Von Willebrand factor D and EGF domain- protein 1 containing protein Disintegrin and metalloproteinase domain- A disintegrin and metalloproteinase with containing protein 9 thrombospondin motifs 17 Angiotensinogen Transmembrane channel-like protein 2 Apolipoprotein A-II (Apo-AII) (ApoA-II) Pregnancy-specific beta-1-glycoprotein 3 Apolipoprotein A-IV (Apo-AIV) (ApoA-IV) Tenomodulin Apolipoprotein C-II (Apo-CII) (ApoC-II) Tetraspanin-6 Beta-2-glycoprotein 1 Thioredoxin domain-containing protein 5 Apoptosis-related protein 3 Vascular endothelial growth factor D Beta-secretase 2 Pregnancy-specific beta-1-glycoprotein 9 Histo-blood group ABO system transferase Semaphorin-3F Cathepsin L2 Acid phosphatase-like protein 2 C-C motif chemokine 3 Apolipoprotein O-like C-type lectin domain family 1 member B Beta-defensin 119 Calcium-activated chloride channel A disintegrin and metalloproteinase with regulator 1 thrombospondin motifs 12 Chymase Protein FAM131A Collagen alpha-1(VI) chain Protein FAM3B Complement component C8 alpha chain Beta-galactosidase-1-like protein Complement component C9 Lysozyme g-like protein 1 Glucose-fructose oxidoreductase domain- Inter-alpha-trypsin inhibitor heavy chain containing protein 2 H5-like protein DnaJ homolog subfamily B member 11 Sperm acrosome-associated protein 5 Ectonucleotide Leucine-rich repeat and immunoglobulin- pyrophosphatase/phosphodiesterase family like domain-containing nogo receptor- member 7 interacting protein 2 Endoplasmic reticulum aminopeptidase 1 Surfactant-associated protein 2 Receptor tyrosine-protein kinase erbB-3 Adiponectin receptor protein 1 Endoplasmic reticulum resident protein Multiple epidermal growth factor-like ERp44 domains 6 IgGFc-binding protein Neuroendocrine protein 7B2 Complement factor H-related protein 1 Alpha-1B-glycoprotein Polypeptide N- WAP, kazal, immunoglobulin, kunitz and acetylgalactosaminyltransferase 2 NTR domain-containing protein 2 Hemopexin Arylacetamide deacetylase-like 1 Hepatocyte growth factor activator Histatin-3 Major histocompatibility complex class I- Pro-neuregulin-3, membrane-bound related gene protein isoform Insulin-like growth factor-binding protein 6 Agouti-signaling protein Ig delta chain C region Claudin-8 Interleukin-1 beta UPF0454 protein C12orf49 Low-density lipoprotein receptor-related von Willebrand factor A domain-containing protein 10 protein 5B1 Junctional adhesion molecule C Cadherin-6 Uncharacterized protein KIAA0319 Cathelicidin antimicrobial peptide Laminin subunit alpha-5 Laminin subunit gamma-1 Fibronectin type III domain-containing Dehydrogenase/reductase SDR family protein 4 member 7B Lipoprotein lipase C-C motif chemokine 16 Interstitial collagenase C-C motif chemokine 24 Matrix metalloproteinase-9 HEAT repeat-containing protein C7orf27 Mucin-16 Collagen alpha-2(IX) chain Mucin-2 Collagen alpha-3(IX) chain
Mucin-5B Colipase Myocilin Collagen alpha-1(XXVII) chain Oxidized low-density lipoprotein receptor 1 Carboxypeptidase N subunit 2 Prostate tumor overexpressed gene 1 Leucine-rich repeat transmembrane protein neuronal protein 4 Receptor-interacting serine/threonine- Collagen triple helix repeat-containing protein kinase 2 protein 1 Equilibrative nucleoside transporter 3 Endothelin-2 Selenoprotein P Fibromodulin Pulmonary surfactant-associated protein D Fc receptor-like B Stimulated by retinoic acid gene 6 protein Zinc finger RAD18 domain-containing homolog protein C1orf124 Trefoil factor 1 Growth/differentiation factor 15 Tissue factor pathway inhibitor 2 Glia-derived nexin Prothrombin Progonadoliberin-1 Toll-like receptor 9 Granzyme K Intercellular adhesion molecule 4 Interferon alpha-17 Interleukin-19 Interferon alpha-21 lsthmin-2 Interferon alpha-8 Kin of IRRE-like protein 1 Interferon omega-1 Kallikrein-10 Early placenta insulin-like peptide Latent-transforming growth factor beta- EGF, latrophilin and seven transmembrane binding protein 4 domain-containing protein 1 Paired immunoglobulin-like type 2 receptor Fibronectin type 3 and ankyrin repeat alpha domains protein 1 Regenerating islet-derived protein 3 alpha Lysyl oxidase homolog 4 E3 ubiquitin-protein ligase RNF5 Lumican Protachykinin-1 Adropin Secreted frizzled-related protein 1, isoform Leucine-rich repeat transmembrane protein CRA_a FLRT1 Plasminogen-related protein B Nucleobindin-2 Probable palmitoyltransferase ZDHHC16 Phospholipase A2 Angiopoietin-related protein 1 Proenkephalin-B UPF0510 protein C19orf63 Peptidoglycan recognition protein I-beta Scavenger receptor cysteine-rich type 1 Immunoglobulin superfamily containing protein M160 leucine-rich repeat protein 2 ER degradation-enhancing alpha- V-set and immunoglobulin domain- mannosidase-like 2 containing protein 2 Beta-galactosidase-1-like protein 2 Peptide YY Interleukin-17 receptor E Retinol-binding protein 3 Interleukin-20 Atherin Interleukin-25 Translocation protein SEC63 homolog PDZ domain-containing protein 11 Transforming growth factor beta-3 Relaxin-3 Protein Wnt-10b Retinoid-inducible serine carboxypeptidase Renalase Short palate, lung and nasal epithelium Proprotein convertase subtilisin/kexin carcinoma-associated protein 2 type 4 WAP four-disulfide core domain protein 5 Carboxypeptidase A4 Platelet-derived growth factor C Olfactomedin-4 Disintegrin and metalloproteinase domain- Insulin-like growth factor-binding protein containing protein 33 complex acid labile chain BSD domain-containing protein 1 Amelogenin, Y isoform Cell adhesion molecule 3 Arylsulfatase F CDC45-related protein Choriogonadotropin subunit beta variant 2 Chondrolectin Beta-defensin 104 Diacylglycerol O-acyltransferase 2 Beta-defensin 105 3-keto-steroid reductase Beta-defensin 107 Interleukin-17 receptor C Protein WFDC11 Interleukin-17 receptor D WAP four-disulfide core domain protein 6 Integrator complex subunit 1 Epigen Junctional adhesion molecule-like Protein FAM19A5 E3 ubiquitin-protein ligase LNX Claudin-6 Leucine-rich repeat transmembrane Carcinoembryonic antigen-related cell neuronal protein 3 adhesion molecule 19 Methionine adenosyltransferase 2 A disintegrin and metalloproteinase with subunit beta thrombospondin motifs 1 Podocalyxin-like protein 2 Protein COQ10 A, mitochondrial Prominin-2 Uncharacterized protein C19orf41 Plexin domain-containing protein 2 Uncharacterized protein C21orf63 Roundabout homolog 4 Protein delta homolog 2 Lactosylceramide alpha-2,3- Cocaine- and amphetamine-regulated sialyltransferase transcript protein SID1 transmembrane family member 2 Lipoma HMGIC fusion partner-like 1 protein Sushi domain-containing protein 1 Leucine-rich repeat-containing protein 18 Serine/threonine-protein kinase TAO2 Leucine-rich repeat-containing protein 25 Transmembrane protease, serine 2 Leucine-rich repeat-containing protein 3B UDP-glucuronic acid decarboxylase 1 Leucine-rich repeat-containing protein 3 Uncharacterized protein C10orf58 Ly6/PLAUR domain-containing protein 4 Thioredoxin-related transmembrane Vitamin K epoxide reductase complex protein 2 subunit 1 CMP-N-acetylneuraminate-beta- A disintegrin and metalloproteinase with galactosamide-alpha-2,3-sialyltransferase thrombospondin motifs 20 Putative uncharacterized protein Putative uncharacterized protein ENSP00000380674 ENSP00000381830 Transmembrane protein 119 Cat eye syndrome critical region protein 1 Transmembrane protein 98 Testis-expressed protein 101 Pre-B lymphocyte protein 3 Xylosyltransferase 2 Putative uncharacterized protein C14orf144 Protein FAM20A Membrane-bound transcription factor site-1 Transmembrane and immunoglobulin protease domain-containing protein 1 Ficolin (Collagen/fibrinogen domain Putative killer cell immunoglobulin-like containing) 3 (Hakata antigen) (NL3) receptor-like protein KIR3DX1 (Leukocyte (Ficolin (Collagen/fibrinogen domain receptor cluster member 12) containing) 3 (Hakata antigen), isoform CRA_b) Interleukin-1 family member 6 Herstatin Prostate and testis expressed protein 2 Leucine-rich repeat-containing protein 28 Group XIIA secretory phospholipase A2 LRRN4 C-terminal-like protein Collagen alpha-3(V) chain Ly6/PLAUR domain-containing protein 2 Alpha-2-macroglobulin-like protein 1 Transmembrane protein 81 Dermatopontin Myelin protein zero-like protein 3 Cartilage-associated protein Protein notum homolog Desert hedgehog protein UDP-glucuronosyltransferase 3A2 Extracellular matrix protein 2 Protocadherin alpha-1 Gastric intrinsic factor Phospholipase D4 Interleukin-33 Retinol dehydrogenase 10 Bone morphogenetic protein 2 Sialic acid-binding Ig-like lectin 14 Bone morphogenetic protein 6 Transmembrane protein 161A Uncharacterized protein KIAA0564 Transmembrane protein 161B Cerberus Transmembrane protein 182 Carbohydrate sulfotransferase 8 Protein FAM24B Contactin-associated protein-like 3 Transmembrane protein 52 Group XIIB secretory phospholipase A2- Major facilitator superfamily domain- like protein containing protein 4 Corticoliberin UDP-glucuronosyltransferase 2A3 A disintegrin and metalloproteinase with Odontogenic ameloblast-associated thrombospondin motifs 19 protein UPF0556 protein C19orf10 Neurosecretory protein VGF C-X-C motif chemokine 3 Secreted phosphoprotein 2, 24 kDa Cystatin-M Protein FAM150B Defensin-5 Growth/differentiation factor 9 Defensin-6 Clusterin-like protein 1 A disintegrin and metalloproteinase with Transmembrane and immunoglobulin thrombospondin motifs 18 domain-containing protein 2 A disintegrin and metalloproteinase with C-type lectin domain-containing protein thrombospondin motifs 3 UNQ5810/PRO19627 Dickkopf-related protein 4 Epididymal-specific lipocalin-10 A disintegrin and metalloproteinase with A disintegrin and metalloproteinase with thrombospondin motifs 5 thrombospondin motifs 8 Mammalian ependymin-related protein 1 Epididymal-specific lipocalin-8 Fibrillin-3 Basic proline-rich peptide P-E Fetuin-B Putative uncharacterized protein C10orf99 Fibroblast growth factor 6 Uncharacterized protein C17orf77 Keratinocyte growth factor Arylacetamide deacetylase-like 2 Growth/differentiation factor 8 Epididymal-specific lipocalin-12 Gastric inhibitory polypeptide B melanoma antigen 2 Glycoprotein hormone beta-5 B melanoma antigen 3 Granzyme M Bovine seminal plasma protein homolog 1 Gastrin-releasing peptide Complement C1q-like protein 3 Serine protease HTRA1 UPF0565 protein C2orf69 Interferon alpha-4 UPF0669 protein C6orf120 Interferon alpha-5 Colipase-like protein C6orf127 Interferon alpha-7 Uncharacterized protein C7orf69 A disintegrin and metalloproteinase with Platelet-derived growth factor receptor-like thrombospondin motifs 7 protein Immunoglobulin superfamily member 10 Chondroadherin-like protein Protease-associated domain-containing Putative uncharacterized protein protein of 21 kDa UNQ6490/PRO21339 Abhydrolase domain-containing protein Putative uncharacterized protein FAM108A1 UNQ6493/PRO21345 A disintegrin and metalloproteinase with Putative uncharacterized protein thrombospondin motifs 9 UNQ5815/PRO19632 Interleukin-9 receptor Cystatin-A Interleukin-9 Peptidase inhibitor R3HDML Inhibin beta B chain Cystatin-9 Serine protease inhibitor Kazal-type 2 DAN domain family member 5 BMP-binding endothelial regulator Insulin-like growth factor-binding protein- protein like 1 Keratinocyte-associated protein 2 Epididymal sperm-binding protein 1 Laminin subunit alpha-1 Elafin Leukocyte cell-derived chemotaxin-2 Protein FAM55A Gastric triacylglycerol lipase Growth/differentiation factor 6 Leucine-rich repeat and calponin Glucose-fructose oxidoreductase domain- homology domain-containing protein 3 containing protein 1 Pancreatic lipase-related protein 2 Erythropoietin Epididymis-specific alpha-mannosidase Glutathione peroxidase 6 Fibronectin type III domain-containing Uncharacterized protein protein 7 UNQ511/PRO1026 Microfibrillar-associated protein 5 Beta-defensin 128 Muellerian-inhibiting factor Interleukin-31 Matrix metalloproteinase-21 Interleukin-34 Matrix metalloproteinase-17 Plasma kallikrein-like protein 4 Matrix metalloproteinase-20 Epididymal-specific lipocalin-9 N-acetylglucosamine-1- cDNA FLJ60957, highly similar to phosphotransferase subunit gamma Secreted frizzled-related protein 4 Multimerin-2 Lipase member M Promotilin CLECSF12 FRAS1-related extracellular matrix Putative inactive group IIC secretory protein 3 phospholipase A2 Protein kinase C-binding protein NELL1 Serine protease MPN2 Protein kinase C-binding protein NELL2 Netrin-5 Neurotrypsin NHL repeat-containing protein 3 Neuroserpin Olfactomedin-like protein 2B Nidogen-2 Ovochymase-2 Abhydrolase domain-containing protein Putative uncharacterized protein FAM108B1 UNQ3029/PRO9830 Neurotrophin-4 Ovochymase-1 Epididymal secretory glutathione Putative pregnancy-specific beta-1- peroxidase glycoprotein 7 Group 10 secretory phospholipase A2 Ovostatin homolog 2 Group IID secretory phospholipase A2 Orexigenic neuropeptide QRFP Lactoperoxidase Lymphocyte antigen 6K p53 apoptosis effector related to PMP-22 Prostate and testis expressed protein 1 Placenta-specific protein 1 Putative phospholipase B-like 1 Tuberoinfundibular peptide of Putative uncharacterized protein 39 residues FLJ42147 Prolargin Otogelin Secretogranin-2 Ribonuclease 8 Endonuclease domain-containing 1 Nuclear pore complex-interacting protein- protein like 2 Semaphorin-3B Proactivator polypeptide-like 1 Somatostatin Protein spinster homolog 2 Dehydrogenase/reductase SDR family von Willebrand factor C domain- member 4-like 2 containing protein 2-like Transcobalamin-1 Urotensin-2B Trefoil factor 2 Tetraspanin-18 Testican-1 UPF0514 membrane protein FAM159A Serum paraoxonase/lactonase 3 Latherin Tolloid-like protein 2 Methyltransferase-like protein 7B Trypsin-2 Protein TEX261 RING finger and SPRY domain- Alkylated DNA repair protein alkB containing protein 1 homolog 7 Calcium-binding and coiled-coil domain- Transmembrane emp24 domain- containing protein 1 containing protein 6 Protein Wnt-2 XK-related protein 5 Ectonucleoside triphosphate Putative V-set and immunoglobulin diphosphohydrolase 8 domain-containing protein 7 Protein Wnt-8b Insulin growth factor-like family member 3 UDP-GlcNAc: betaGal beta-1,3-N- Nuclear pore complex-interacting protein- acetylglucosaminyltransferase 4 like 1 EMI domain-containing protein 1 Secreted phosphoprotein 1 Uncharacterized protein C6orf15 Collagen alpha-5(VI) chain Collectin-10 B melanoma antigen 5 Long-chain-fatty-acid--CoA ligase WAP four-disulfide core domain protein ACSBG2 10A
Oncoprotein-induced transcript 3 protein UPF0369 protein C6orf57 Peptidase inhibitor 15 Putative uncharacterized protein C10orf31 Proline-rich acidic protein 1 Putative uncharacterized protein C11orf45 Urocortin Uncharacterized protein C12orf28 Trypsin-X3 (EC 3.4.21.4) Uncharacterized protein C17orf67 HHIP-like protein 2 Beta-defensin 121 Fractalkine Beta-defensin 130 Protein Wnt-11 Histidine triad nucleotide-binding protein 2 Protein Wnt-7a Apelin FCH and double SH3 domains protein 1 Placenta-specific protein 9 Hepatoma-derived growth factor-related Hepatocellular carcinoma-associated protein 2 protein TD26 Interleukin-12 subunit alpha Persephin UPF0577 protein KIAA1324 Regulated endocrine-specific protein 18 Complement C1q tumor necrosis factor- Complement C1q tumor necrosis factor- related protein 9 related protein 8 Mucin-17 Bone morphogenetic protein 8A Lysosomal protein NCU-G1 Protein WFDC13 Prolyl 4-hydroxylase subunit alpha-3 Protein Wnt-8a Peptidyl-prolyl cis-trans isomerase Ig-like domain-containing protein SDCCAG10 ENSP00000270642 Peptidase inhibitor 16 Abhydrolase domain-containing protein 15 Poliovirus receptor-related protein 4 Ribonuclease-like protein 9 Solute carrier family 22 member 15 Uncharacterized protein C2orf66 GPI inositol-deacylase Uncharacterized protein C17orf99 Transmembrane protein 43 Protein FAM150A Angiopoietin-related protein 2 Placenta-specific 1-like protein Angiopoietin-related protein 6 Uncharacterized protein C18orf20 Arylsulfatase K Beta-defensin 110 Augurin Neuritin-like protein Brain-specific serine protease 4 Histidine-rich carboxyl terminus protein 1 DBH-like monooxygenase protein 1 C-type lectin domain family 2 member A Uncharacterized protein C1orf56 Leucine-rich repeat-containing protein 70 Cerebellin-3 Serpin A13 Cerebellin-4 BTB/POZ domain-containing protein 17 Colipase-like protein C6orf126 Uncharacterized protein C12orf53 Uncharacterized protein C11orf83 C-type lectin domain family 9 member A Uncharacterized protein C16orf89 Complement C1q-like protein 4 Carboxypeptidase-like protein X2 CMRF35-like molecule 4 Cystatin-9-like Protein FAM151B Dehydrogenase/reductase SDR family Abhydrolase domain-containing protein member 13 FAM108A2/A3 Beta-defensin 123 Osteocrin Beta-defensin 132 Transmembrane protease, serine 11E2 Cytokine-like protein 1 Transmembrane protein 14E Dickkopf-related protein 2 Transmembrane protein 207 Dickkopf-like protein 1 TOMM20-like protein 1 Epididymal secretory protein E3-beta Uncharacterized protein C3orf41 EGF-like repeat and discoidin I-like Submaxillary gland androgen-regulated domain-containing protein 3 protein 3A Protein FAM55D B melanoma antigen 1 Fibroblast growth factor 17 Inactive carboxylesterase 4 Fibroblast growth factor 22 Four-jointed box protein 1 Fibroblast growth factor-binding protein 2 Protein HSN2 Growth/differentiation factor 3 Humanin GLIPR1-like protein 1 Kielin/chordin-like protein Serine protease inhibitor Kazal-type 6 UPF0624 protein C6orf186 Interleukin-17B Putative neurofibromin 1-like protein 4/6 Interleukin-17C Peroxidasin-like protein Interleukin-17D SCO-spondin Hyaluronan and proteoglycan link Putative uncharacterized protein protein 3 UNQ9165/PRO28630 Vitelline membrane outer layer protein 1 Calcium-activated chloride channel homolog regulator family member 3 Choriogonadotropin subunit beta Probable serine protease variant 1 UNQ9391/PRO34284 Lysozyme-like protein 1 Uncharacterized protein C4orf26 Matrix metalloproteinase-28 Uncharacterized protein C4orf40 Nephronectin Uncharacterized protein C5orf55 WAP four-disulfide core domain Putative macrophage-stimulating protein protein 12 MSTP9 Olfactomedin-like protein 1 Uncharacterized protein C15orf61 Olfactomedin-like protein 2A Chymotrypsinogen B2 Serine protease 27 Beta-defensin 108A Secretoglobin family 3A member 2 Beta-defensin 111 A disintegrin and metalloproteinase with Putative V-set and immunoglobulin thrombospondin motifs 2 domain-containing protein 6 Disintegrin and metalloproteinase Serine protease inhibitor Kazal-type domain-containing protein 28 5-like 3 Bactericidal/permeability-increasing Putative serine protease inhibitor Kazal- protein-like 2 type 5-like 2 Acid sphingomyelinase-like Dehydrogenase/reductase SDR family phosphodiesterase 3b member 7C Serine protease inhibitor Kazal-type 7 Beta-defensin 131 Neurexophilin-4 Beta-defensin 134 Protein Wnt-9b Beta-defensin 136 Zymogen granule protein 16 homolog B Beta-defensin 116 Semaphorin-3D Protein FAM132A Apolipoprotein L4 Protein FAM132B Transmembrane protease, serine 11D Beta-defensin 115 Scrapie-responsive protein 1 Beta-defensin 114 Putative annexin A2-like protein Serine protease inhibitor Kazal-type 9 Bone morphogenetic protein 10 Lipase member N Secretogranin-3 Pancreatic lipase-related protein 3 Complement C1q tumor necrosis factor- Testis, prostate and placenta-expressed related protein 4 protein Uncharacterized protein C1orf54 Neuromedin-S Carboxypeptidase A6 Neuropeptide S C-C motif chemokine 19 Neuronal pentraxin-like protein C16orf38 C-C motif chemokine 25 Otolin-1 Chymotrypsin-like elastase family Iron/zinc purple acid phosphatase-like member 2B protein Protein CEI Ovostatin homolog 1 Uncharacterized protein C6orf1 Plasminogen-related protein A Uncharacterized protein C7orf34 Polyserase-3 Keratinocyte-associated protein 3 Putative peptide YY-2 Uncharacterized protein C9orf47 Putative peptide YY-3 Collagen alpha-1(VIII) chain Ribonuclease-like protein 10 Uncharacterized protein C18orf54 Ribonuclease-like protein 12 Cystatin-like 1 Ribonuclease-like protein 13 C2 domain-containing protein 2 Serpin A11 DDRGK domain-containing protein 1 Kunitz-type protease inhibitor 4 Protein FAM55C Meteorin-like protein Collagen alpha-1(XXVI) chain Putative testis serine protease 2 Protein FAM19A2 Beta-defensin 112 Protein FAM5B Uncharacterized protein FLJ37543 Fibroblast growth factor 5 Protein FAM24A Probable serine protease HTRA3 Secreted frizzled-related protein 4 Interleukin-1 family member 8 Complement C1q-like protein 2 Serine protease inhibitor Kazal-type 4 Putative uncharacterized protein C17orf69 Otospiralin Putative cystatin-13 Liver-expressed antimicrobial peptide 2 Beta-defensin 109 Lysyl oxidase homolog 1 Beta-defensin 113 Lysyl oxidase homolog 2 Beta-defensin 135 Long palate, lung and nasal epithelium Peptidase S1 domain-containing protein carcinoma-associated protein 4 LOC136242 Lysozyme g-like protein 2 Growth/differentiation factor 7 Endomucin IgA-inducing protein homolog Neuropeptide B Putative lipocalin 1-like protein 1 Kinesin-like protein KIF7 Putative serine protease 29 Leukocyte-associated immunoglobulin- Putative scavenger receptor cysteine-rich like receptor 2 domain-containing protein LOC619207 Calcium-dependent phospholipase A2 Secretoglobin-like protein Proapoptotic caspase adapter protein Putative stereocilin-like protein Integrin beta-like protein 1 Insulin growth factor-like family member 2 Tolloid-like protein 1 KIR2DL4 Kunitz-type protease inhibitor 3 Putative zinc-alpha-2-glycoprotein-like 1 Protein TMEM155 Insulin growth factor-like family member 4 Prosalusin Uncharacterized protein C2orf72 Protein amnionless Replication initiation-like protein Protein WFDC10B Prostate and testis expressed protein 3 WAP four-disulfide core domain protein 8 B melanoma antigen 4 Protein Wnt-5b Putative uncharacterized protein C1orf191 Protein Wnt-7b Beta-defensin 108B-like Zona pellucida-binding protein 2 Uncharacterized protein FLJ90687 SH3 domain-binding protein 5-like Secreted frizzled-related protein 2 Adipocyte adhesion molecule Basic proline-rich peptide IB-1 Uncharacterized protein C12orf59 Fibroblast growth factor 16 Apolipoprotein A-I-binding protein Serine protease inhibitor Kazal-type 8 Claudin-17 Uncharacterized protein KIAA0495 Inactive caspase-12 Platelet basic protein-like 2 Uncharacterized protein C7orf58 Serpin E3 Collagen alpha-1(XXVIII) chain CR1 receptor Dentin matrix protein 4 Secreted phosphoprotein 1 Uncharacterized protein C16orf48 Stress induced secreted protein 1 Carboxylesterase 3 Protein Wnt Protein FAM20B Protein Wnt (Fragment) GPN-loop GTPase 3 Putative serine protease LOC138652 GRAM domain-containing protein 1B TOM1 Phosphatidylinositol glycan anchor Putative uncharacterized protein biosynthesis class U protein FLJ46089 Interleukin-27 subunit alpha Putative uncharacterized protein C1orf134 Pro-neuregulin-4, membrane-bound UDP-GlcNAc: betaGal beta-1,3-N- isoform acetylglucosaminyltransferase 9 Leucine-rich repeat neuronal protein 3 Uncharacterized protein C11orf44 NMDA receptor-regulated protein 2 Uncharacterized protein C12orf73 NADH-cytochrome b5 reductase 1 Putative cystatin-9-like 2 Parkinson disease 7 domain-containing Putative abhydrolase domain-containing protein 1 protein FAM108A5 FK506-binding protein 11 Beta-defensin 133 C-type lectin domain family 12 member B Fibrosin-1 Solute carrier family 35 member F5 Probable folate receptor delta Sialic acid-binding Ig-like lectin 12 RPE-spondin Protein FAM19A3 NPIP-like protein ENSP00000346774 WD repeat-containing protein 82 Putative testis-specific prion protein Adipocyte enhancer-binding protein 1 Proline-rich protein 1 ADAMTS-like protein 3 Putative uncharacterized protein FP248 Coiled-coil domain-containing protein 80 UPF0670 protein C8orf55 Ecto-NOX disulfide-thiol exchanger 1 Putative zinc-alpha-2-glycoprotein-like 2 Neuronal growth regulator 1 SPARC protein Interphotoreceptor matrix proteoglycan 1 Otopetrin-1 cDNA FLJ36603 fis, clone cDNA FLJ55667, highly similar to TRACH2015180, highly similar to Secreted protein acidic and rich in Secreted frizzled-related protein 2 cysteine Lipase member H Lipase member K Mucin-19 (MUC-19) C-type lectin domain family 18 member C Psoriasis susceptibility 1 candidate gene Putative uncharacterized protein 2 protein UNQ6125/PRO20090 Integral membrane protein 2A Complement C3 Vesicle transport protein SFT2B Collagen alpha-2(IV) chain von Willebrand factor A domain- Uncharacterized protein containing protein 3A UNQ6126/PRO20091 Protein shisa-2 homolog Serpin-like protein HMSD Signal peptidase complex subunit 3 Prostate and testis expressed protein 4 CD164 sialomucin-like 2 protein Collagen alpha-1(XXII) chain Cadherin-16 Putative uncharacterized protein C13orf28 Cadherin-19 Cystatin-S Cerebellin-2 R-spondin-1 Transmembrane protein C3orf1 C8orf2 Sperm equatorial segment protein 1 Odorant-binding protein 2a Uncharacterized protein C6orf72 Opiorphin Uncharacterized protein C11orf24 Kidney androgen-regulated protein Acyl-CoA synthetase family member 2, Putative uncharacterized protein mitochondrial UNQ5830/PRO19650/PRO19816 Probable UDP-sugar transporter protein Putative uncharacterized protein SLC35A5 UNQ6975/PRO21958 C-type lectin domain family 1 member A Tachykinin-3 C-type lectin domain family 3 member A Secreted phosphoprotein 1 C-type lectin domain family 4 member E Sclerostin C-type lectin domain family 4 member G ADAMTS-like protein 2 Probable cation-transporting Scavenger receptor cysteine-rich domain- ATPase 13A4 containing protein LOC284297 UPF0480 protein C15orf24 Tryptase beta-1 Zona pellucida sperm-binding protein 4 Tryptase delta Endoplasmic reticulum resident protein Putative cat eye syndrome critical region ERp27 protein 9 Transmembrane protein C16orf54 Plexin domain-containing protein 1 Cytochrome P450 4F12 MC51L-53L-54L homolog (Fragment) Cytochrome P450 4X1 COBW-like placental protein (Fragment) Cytochrome P450 4Z1 Cytokine receptor-like factor 2 Protein CREG2 Beta-defensin 103 DnaJ homolog subfamily B member 9 Beta-defensin 106 Dipeptidase 3 Hyaluronidase-3 Membrane protein FAM174A Interleukin-28 receptor alpha chain Thioredoxin domain-containing Glycosyltransferase 54 domain-containing protein 15 protein Protein FAM19A4 Chordin-like protein 1 Adenosine monophosphate-protein Putative uncharacterized protein transferase FICD UNQ9370/PRO34162 Prenylcysteine oxidase-like Netrin receptor UNC5B Phytanoyl-CoA hydroxylase-interacting Fibroblast growth factor receptor FGFR-1 protein-like secreted form protein (Fragment) FXYD domain-containing ion transport Uncharacterized protein
regulator 4 ENSP00000244321 Growth/differentiation factor 11 ECE2 Cerebral dopamine neurotrophic factor EPA6 GPN-loop GTPase 2 Putative soluble interleukin 18 receptor 1 Growth hormone-inducible Putative abhydrolase domain-containing transmembrane protein protein FAM108A6 Glycerophosphodiester Putative V-set and immunoglobulin phosphodiesterase domain-containing domain-containing-like protein protein 2 ENSP00000303034 WAP, kazal, immunoglobulin, kunitz and B cell maturation antigen transcript variant NTR domain-containing protein 1 4 (Tumor necrosis factor receptor superfamily member 17) KDEL motif-containing protein 1 UPF0672 protein C3orf58 Adipophilin Methylthioribose-1-phosphate isomerase Lactase-like protein 17-beta hydroxysteroid dehydrogenase 13 Chondromodulin-1 Aminopeptidase B Collagen alpha-6(VI) chain Dermcidin Leucine-rich repeat-containing protein 33 Meteorin MANSC domain-containing protein 1 Methyltransferase-like protein 7A Lipocalin-15 NL3 Arylsulfatase I N-acetyltransferase 15 Mesoderm development candidate 2 Ephrin-A4 Dickkopf-related protein 1 Protein Plunc Podocan Kallikrein-11 Fibronectin type III domain-containing WNT1 induced secreted protein 1 splice protein 1 variant x (Fragment) Neurotrimin Interleukin-1 family member 10 Olfactory receptor 10W1 PLA2G2D Protein PARM-1 Proteoglycan 3 PDZ domain-containing protein 2 Insulin-like peptide INSL5 Proepiregulin Olfactomedin-like protein 3 Polycystic kidney disease protein 1-like 1 Extracellular glycoprotein lacritin WLPL514 Retinol dehydrogenase 13 Matrix metalloproteinase-26 Neutrophil defensin 3 RELT-like protein 2 GLGQ5807 Solute carrier family 35 member E3 TUFT1 Zinc transporter ZIP9 DRLV8200 Noelin-2 IDLW5808 Seizure 6-like protein 2 UBAP2 Semaphorin-3A C1q/TNF-related protein 8 Semaphorin-4C KIR2DL4 (Fragment) Abhydrolase domain-containing protein Chemokine-like factor super family 2 14A transcript variant 2 Ankyrin repeat domain-containing Keratinocytes associated transmembrane protein 36 protein 1 Protein shisa-4 GKGM353 Neuromedin-U MATL2963 Nodal homolog NINP6167 Synaptogyrin-2 POM121-like Brain-specific angiogenesis inhibitor 1- RTFV9368 (SLE-dependent associated protein 2-like protein 2 upregulation 1) Coiled-coil domain-containing Leucine-rich repeat and immunoglobulin- protein 104 like domain-containing nogo receptor- interacting protein 4 Transmembrane 4 L6 family member 20 KCNQ2 Transmembrane protein 107 ELCV5929 Transmembrane protein 143 KVVM3106 Transmembrane protein 178 ISPF6484 Transmembrane protein 205 LKHP9428 Transmembrane protein 41A VNFT9373 Transmembrane protein 50A ACAH3104 Transmembrane protein 50B RVLA1944 Interleukin-28B Wpep3002 Neuronal pentraxin-2 ZDHHC11 Collectrin AGLW2560 Transmembrane protein 92 TSSP3028 Transmembrane protein 95 RFVG5814 Transmembrane protein 9B SHSS3124 Probable carboxypeptidase PM20D1 MMP19 Tetraspanin-12 GSQS6193 Tetraspanin-13 VGPW2523 Tetraspanin-15 LMNE6487 UPF0513 transmembrane protein ALLA2487 Mitochondrial uncoupling protein 4 GALI1870 Polyserase-2 FRSS1829 Probable palmitoyltransferase ZDHHC24 MRSS6228 Zona pellucida sperm-binding protein 1 GRPR5811 Zona pellucida sperm-binding protein 2 AVLL5809 Conserved oligomeric Golgi complex CR1 C3b/C4b receptor SCR9 (or 16) C- subunit 7 term. exon SCR = short consensus repeat Adiponectin receptor protein 2 PIKR2786 Inhibin beta C chain S100 calcium binding protein A7-like 3 Brorin GTWW5826 (LP5085 protein) Semaphorin-3C KTIS8219 (HCG2020043) Heparan sulfate glucosamine 3-O- Hyaluronan and proteoglycan link sulfotransferase 2 protein 4 Leptin receptor overlapping transcript- Micronovel like 1 SPARC-like protein 1 SAMK3000 Fibulin-7 VFLL3057 Protein HEG homolog 1 CVWG5837 Fibrinogen C domain-containing VGSA5840 protein 1 Phospholipase A1 member A GHPS3125 Basic salivary proline-rich protein 2 GRTR3118 Spermatogenesis-associated protein 6 PAMP6501 Sushi repeat-containing protein SRPX2 LTLL9335 Twisted gastrulation protein homolog 1 VCEW9374 Torsin-1B AHPA9419 Protein Wnt-5a MDHV1887 Acrosin-binding protein HSAL5836 C-type lectin domain family 18 member B LHLC1946 Lysosomal-associated transmembrane Long palate, lung and nasal epithelium protein 4A carcinoma-associated protein 3 (Ligand- binding protein RYA3) Semaphorin-3E LPPA601 Ameloblastin PINK1 Major facilitator superfamily domain- SERH2790 containing protein 5 Angiopoietin-1 FLFF9364 Angiopoietin-4 APELIN Multiple epidermal growth factor-like GLSH6409 domains 9 Acid sphingomyelinase-like SFVP2550 phosphodiesterase 3a ADAMTS-like protein 5 RRLF9220 Spexin PTML5838 Putative trypsin-6 VLGN1945 Proto-oncogene protein Wnt-1 AVPC1948 Bone morphogenetic protein 3b AWQG2491 Bone morphogenetic protein 5 PSVL6168 Bone morphogenetic protein 8B LCII3035 Protein FAM26D PPRR6495 C1q-related factor RLSC6348 WAP four-disulfide core domain protein 1 CSRP2BP Cerebellin-1 GLLV3061 Carboxypeptidase O GWSI6489 Myelin protein zero-like protein 2 cDNA FLJ53955, highly similar to (Epithelial V-like antigen 1) Secreted frizzled-related protein 4 Serine protease 1-like protein 1 PPIF Coiled-coil domain-containing protein 70 VSSW1971 C-C motif chemokine 28 KLIA6249 Uncharacterized protein C4orf29 ALLW1950 CUB domain-containing protein 2 GVEI466 Trem-like transcript 4 protein ESFI5812 Uncharacterized protein C6orf58 GNNC2999 Chondroadherin AAGG6488 Cartilage intermediate layer protein 2 HHSL751 Uncharacterized protein C10orf25 Beta-defensin 108B Isthmin-1 Beta-defensin 118 Cystatin-8 Beta-defensin 124 Cardiotrophin-1 (CT-1) Beta-defensin 125 Chymotrypsinogen B Beta-defensin 126 C-X-C motif chemokine 9 Deoxyribonuclease-1-like 2 C-X-C motif chemokine 13 Stanniocalcin-2 EMILIN-3 Endothelial cell-specific molecule 1 Secretagogin Carboxylesterase 7 Epididymal secretory protein E3-alpha Protein NOV homolog Epiphycan UPF0528 protein FAM172A Protein FAM5C Interleukin-27 subunit beta Fibroblast growth factor 20 Protein FAM3C Fibroblast growth factor-binding protein 3 Stromal cell-derived factor 2-like protein 1 Transmembrane protein 204 Butyrophilin subfamily 1 member A1 Phosphatidylethanolamine-binding Keratinocyte-associated transmembrane protein 4 protein 2 Coagulation factor V Immunoglobulin alpha Fc receptor Coagulation factor VII EMILIN-2 Pro-MCH Ephrin type-A receptor 10 Folate receptor gamma Exostosin-like 2 Mucin-7 Follistatin-related protein 4 Galanin-like peptide Follistatin-related protein 5 Hemicentin-1 Transmembrane protein 66 Interleukin-6 Growth/differentiation factor 2 Embryonic growth/differentiation factor 1 GDNF family receptor alpha-4 Interleukin-8 Ig gamma-4 chain C region Gremlin-2 Lymphocyte antigen 86 Stromelysin-2 Inhibin beta E chain Probable G-protein coupled receptor 171 GRAM domain-containing protein 1C Pappalysin-2 Interferon alpha-10 Microfibril-associated glycoprotein 4 Interferon alpha-16 Neuromedin-B Interferon alpha-6 Mimecan Immunoglobulin superfamily member 21 Matrix metalloproteinase-19 Agrin Interleukin-11 Prolactin Interleukin-17A Kelch-like protein 11 Interleukin-18 Protein Wnt-16 Interleukin-26 Properdin Interleukin-28A Kallikrein-13 Transmembrane emp24 domain- 1-acyl-sn-glycerol-3-phosphate containing protein 3 acyltransferase delta Interleukin-29 Kallikrein-9 Insulin-like peptide INSL6 Vitamin K-dependent protein S Protein Wnt-2b Butyrophilin-like protein 8 Pregnancy-specific beta-1-glycoprotein 1 Laminin subunit beta-4 Sperm acrosome membrane-associated Lymphatic vessel endothelial hyaluronic protein 4 acid receptor 1 Laminin subunit gamma-3 Cystatin-SA Lysyl oxidase homolog 3 Transmembrane protein 59 Neurotensin/neuromedin N Apolipoprotein(a)-like protein 2 MAM domain-containing protein 2 Lysozyme-like protein 2 Microfibrillar-associated protein 2 Lysozyme-like protein 4 Melanoma inhibitory activity protein 2 Reelin Matrix metalloproteinase-24 Retinol-binding protein 4 Matrix metalloproteinase-25 Carbonic anhydrase 14 Netrin-1 Tubulointerstitial nephritis antigen Netrin-3 Neuropeptide W Alpha-N-acetylgalactosaminide alpha- Alpha-1,3-mannosyl-glycoprotein 4-beta- 2,6-sialyltransferase 1 N-acetylglucosaminyltransferase B Alpha-N-acetylgalactosaminide alpha- Transmembrane emp24 domain- 2,6-sialyltransferase 3 containing protein 5 Melanoma-derived growth regulatory Complement C1q tumor necrosis factor- protein related protein 3 FMRFamide-related peptides Podocan-like protein 1 Otoconin-90 Pregnancy-specific beta-1-glycoprotein 5 Neurturin Keratocan Neurexophilin-1 Group IIE secretory phospholipase A2 Neurexophilin-2 Left-right determination factor 2 Platelet factor 4 variant NKG2D ligand 2 Nociceptin Macrophage metalloelastase V-set and transmembrane domain- Triggering receptor expressed on myeloid containing protein 1 cells 1 Proline-rich protein 4 Cytokine receptor-like factor 1 Prolactin-releasing peptide Secretin Serine protease 33 Stromal cell-derived factor 2 Pregnancy-specific beta-1-glycoprotein 8 Lysozyme-like protein 6 Retbindin Serpin A9 FMRFamide-related peptides Sclerostin domain-containing protein 1 Ribonuclease K6 Lysocardiolipin acyltransferase 1 Ribonuclease T2 Plasma glutamate carboxypeptidase Repetin Slit homolog 3 protein Complement C1r subcomponent-like C3 and PZP-like alpha-2-macroglobulin protein domain-containing protein 8 Uncharacterized glycosyltransferase Retinoic acid receptor responder AER61 protein 2 Semaphorin-3G Cartilage acidic protein 1 Secretoglobin family 1C member 1 Stanniocalcin-1 Secretoglobin family 1D member 1 Beta-tectorin Secretoglobin family 1D member 2 Post-GPI attachment to proteins factor 3 Serpin A12 Germ cell-specific gene 1 protein Serpin I2 Interleukin-21 receptor von Willebrand factor C and EGF V-set and immunoglobulin domain- domain-containing protein containing protein 4 A disintegrin and metalloproteinase with Scavenger receptor cysteine-rich domain- thrombospondin motifs 15 containing group B protein Sodium channel subunit beta-2 Prothyroliberin Metalloproteinase inhibitor 4 Semaphorin-4A T-cell immunomodulatory protein A disintegrin and metalloproteinase with Tumor necrosis factor receptor thrombospondin motifs 10 superfamily member 27 Thymic stromal lymphopoietin Toll-like receptor 7 Transmembrane protein 130 Unique cartilage matrix-associated Thioredoxin domain-containing protein protein 16
Urocortin-2 Alpha-2-antiplasmin Urocortin-3 ( WAP four-disulfide core domain protein 3 Protein AMBP Protein WFDC9 Complement C1q tumor necrosis factor- A disintegrin and metalloproteinase with related protein 9-like thrombospondin motifs 14 Growth inhibition and differentiation- Adipocyte plasma membrane-associated related protein 88 protein Protein Wnt-10a Peroxidasin homolog Protein Wnt-3a Progressive ankylosis protein homolog Proto-oncogene protein Wnt-3 Chitinase-3-like protein 1 Protein Wnt-6 UPF0672 protein CXorf36 Protein Wnt-9a Arylsulfatase J Cytokine SCM-1 beta Cortistatin Zymogen granule membrane protein 16 Ceruloplasmin Zona pellucida-binding protein 1 Angiopoietin-related protein 5 Anterior gradient protein 3 homolog Coiled-coil domain-containing protein 126 Amelotin CD177 antigen Uncharacterized protein C5orf46 Protein canopy homolog 4 Uncharacterized aarF domain-containing Fibronectin type-III domain-containing protein kinase 1 protein C4orf31 Draxin Protein FAM180A Fibroblast growth factor 18 Platelet basic protein C-X-C motif chemokine 11 Interferon epsilon Ly6/PLAUR domain-containing protein 6 lntelectin-2 Chymotrypsin-like elastase family Alpha-1,3-mannosyl-glycoprotein 4-beta- member 1 N-acetylglucosaminyltransferase A Erythropoietin receptor Matrix extracellular phosphoglycoprotein MAM domain-containing cDNA FLJ77863, highly similar to Homo glycosylphosphatidylinositol anchor sapiens secreted and transmembrane 1 protein 2 (SECTM1), mRNA Matrix metalloproteinase-27 Epididymal-specific lipocalin-6 Inactive serine protease 35 Afamin Coiled-coil domain-containing Probable cation-transporting ATPase protein 134 13A5 Suprabasin Glutathione peroxidase 3 Secretoglobin family 1D member 4 Claudin-18 V-set and transmembrane domain- Putative killer cell immunoglobulin-like containing protein 2A receptor like protein KIR3DP1 ADM Secretory phospholipase A2 receptor Uncharacterized protein C2orf82 Haptoglobin Insulin growth factor-like family Carcinoembryonic antigen-related cell member 1 adhesion molecule 20 Cadherin-like protein 29 Bone morphogenetic protein 3 Bone morphogenetic protein 15 Bone marrow stromal antigen 2 Plasma serine protease inhibitor Cytochrome P450 20A1 Carcinoembryonic antigen-related cell Bactericidal/permeability-increasing adhesion molecule 21 protein-like 3 Alpha-lactalbumin Protein dpy-19 homolog 2 Sister chromatid cohesion protein DCC1 Group IIF secretory phospholipase A2 Galectin-3-binding protein Carboxypeptidase B Dynein heavy chain domain-containing Glycosyltransferase 8 domain-containing protein 1 protein 2 C-C motif chemokine 17 Protein FAM19A1 Fatty acyl-CoA reductase 1 GDNF family receptor alpha-like Fin bud initiation factor homolog Probable glutathione peroxidase 8 Polymeric immunoglobulin receptor Cystatin-D Prion-like protein doppel Cystatin-F C-X-C motif chemokine 6 Platelet-activating factor acetylhydrolase C-X-C motif chemokine 10 Pappalysin-1 Beta-defensin 1 Solute carrier family 22 member 12 Hyaluronan and proteoglycan link Chorionic somatomammotropin hormone- protein 2 like 1 Disintegrin and metalloproteinase Regulator of microtubule dynamics domain-containing protein 30 protein 3 Suppressor of fused homolog Retinol dehydrogenase 14 Folate receptor beta Galanin Extracellular sulfatase Sulf-2 Transcobalamin-2 Tumor necrosis factor receptor Catechol-O-methyltransferase domain- superfamily member 14 containing protein 1 Artemin Tripeptidyl-peptidase 1 Collagen alpha-1(XII) chain Trem-like transcript 1 protein Collagen alpha-1(XIV) chain Guanylate cyclase activator 2B Beta-defensin 2 Inducible T-cell costimulator Interleukin-21 Interleukin-3 Interleukin-7 Notch homolog 2 N-terminal-like protein Inhibin alpha chain Laminin subunit beta-2 Laminin subunit alpha-3 Neuropilin-2 Dehydrogenase/reductase SDR family EGF-containing fibulin-like extracellular member on chromosome X matrix protein 1 FXYD domain-containing ion transport Receptor-type tyrosine-protein regulator 6 phosphatase kappa Serine incorporator 2 Regenerating islet-derived protein 4 Stromelysin-3 Tachykinin-4 Secreted phosphoprotein 1 Matrix metalloproteinase-23 Serine beta-lactamase-like protein Complement C1q tumor necrosis factor- LACTB, mitochondrial related protein 5 Galectin-3 Opticin Pancreatic prohormone Pre-small/secreted glycoprotein Pregnancy-specific beta-1-glycoprotein 6 Pentraxin-related protein PTX3 Dickkopf-related protein 3 Carboxylesterase 8 Dehydrogenase/reductase SDR family Thioredoxin-related transmembrane member 11 protein 4 Regenerating islet-derived protein 3 Major facilitator superfamily domain- gamma containing protein 2 RING finger protein 43 Kallikrein-12 Semenogelin-2 Brevican core protein Mucin-15 Porimin Bone sialoprotein 2 Torsin-1A Lymphotactin C-C motif chemokine 23 Growth-regulated alpha protein Testican-3 R-spondin-2 Basic salivary proline-rich protein 4 Transmembrane and coiled-coil domain- Tumor necrosis factor receptor containing protein 3 superfamily member 18 VEGF co-regulated chemokine 1 Brother of CDO ADM2 Beta-1,4-galactosyltransferase 4 Hydroxysteroid 11-beta-dehydrogenase Dehydrogenase/reductase SDR family 1-like protein member 9 Delta-like protein 1 Eppin Ephrin-A1 Otoancorin Fibroblast growth factor receptor-like 1 Tenascin-R GDNF family receptor alpha-3 Growth factor Platelet receptor Gi24 Protein TSPEAR Progonadoliberin-2 Hephaestin Kallikrein-7 Butyrophilin-like protein 3 Apolipoprotein F Butyrophilin-like protein 9 Protein CASC4 Laminin subunit gamma-2 VIP36-like protein Protein LMBR1L Magnesium transporter protein 1 Mucin-21 Amiloride-sensitive amine oxidase Endoplasmic reticulum mannosyl- [copper-containing] oligosaccharide 1,2-alpha-mannosidase DNA damage-regulated autophagy Pancreatic secretory granule membrane modulator protein 2 major glycoprotein GP2 Transmembrane protein C17orf87 Semaphorin-4B Complement factor H-related protein 5 Semaphorin-5B FK506-binding protein 7 Epsilon-sarcoglycan Serine incorporator 1 Guanylate-binding protein 5 Transmembrane and ubiquitin-like Ectonucleoside triphosphate domain-containing protein 1 diphosphohydrolase 6 Protein ERGIC-53-like Serpin B3 Toll-like receptor 10 Protein RMD5 homolog B Toll-like receptor 8 Scavenger receptor class A member 5 Selenoprotein T Semaphorin-6B Sialic acid-binding Ig-like lectin 11 Transmembrane protein 108 Sorting nexin-24 Sushi domain-containing protein 3 Complement C1q tumor necrosis factor- Latent-transforming growth factor beta- related protein 1 binding protein 2 Putative uncharacterized protein Putative uncharacterized protein UNQ6494/PRO21346 UNQ6190/PRO20217 Secreted and transmembrane 1 precusor Secreted and transmembrane 1 precusor variant variant C-type lectin domain family 18 member A Collagen alpha-1(XX) chain Cysteine-rich secretory protein 3 Netrin receptor UNC5D Complement C4-A Mucin-13 Putative uncharacterized protein ATP-dependent metalloprotease YME1L1 PRO2829 Calcium-activated chloride channel Proprotein convertase subtilisin/kexin regulator 2 type 5 Neuroblastoma suppressor of tumorigenicity 1
[0075] The therapeutic proteins provided herein should not be considered to be exclusive. Rather, as is apparent from the disclosure provided herein, the methods of the invention are applicable to any protein wherein attachment of a water soluble polymer is desired according to the invention. For example, therapeutic proteins are described in US 2007/0026485, incorporated herein by reference in its entirety.
Blood Coagulation Proteins
[0076] In one aspect, the starting material of the present invention is a blood coagulation protein, which can be derived from human plasma, or produced by recombinant engineering techniques, as described in patents U.S. Pat. Nos. 4,757,006; 5,733,873; 5,198,349; 5,250,421; 5,919,766; and EP 306 968.
[0077] Therapeutic polypeptides such as blood coagulation proteins including Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI (FXI), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF) and ADAMTS 13 protease are rapidly degraded by proteolytic enzymes and neutralized by antibodies. This reduces their half-life and circulation time, thereby limiting their therapeutic effectiveness. Relatively high doses and frequent administration are necessary to reach and sustain the desired therapeutic or prophylactic effect of these coagulation proteins. As a consequence, adequate dose regulation is difficult to obtain and the need of frequent intravenous administrations imposes restrictions on the patient's way of living.
[0078] As described herein, blood coagulation proteins including, but not limited to, Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XI, Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF) and ADAMTS 13 protease are contemplated by the invention. As used herein, the term "blood coagulation protein" refers to any Factor IX (FIX), Factor VIII (FVIII), Factor VIIa (FVIIa), Von Willebrand Factor (VWF), Factor FV (FV), Factor X (FX), Factor XII (FXII), thrombin (FII), protein C, protein S, tPA, PAI-1, tissue factor (TF) and ADAMTS 13 protease which exhibits biological activity that is associated with that particular native blood coagulation protein.
[0079] The blood coagulation cascade is divided into three distinct segments: the intrinsic, extrinsic, and common pathways (Schenone et al., Curr Opin Hematol. 2004; 11:272-7). The cascade involves a series of serine protease enzymes (zymogens) and protein cofactors. When required, an inactive zymogen precursor is converted into the active form, which consequently converts the next enzyme in the cascade.
[0080] The intrinsic pathway requires the clotting factors VIII, IX, X, XI, and XII. Initiation of the intrinsic pathway occurs when prekallikrein, high-molecular-weight kininogen, factor XI (FXI) and factor XII (FXII) are exposed to a negatively charged surface. Also required are calcium ions and phospholipids secreted from platelets.
[0081] The extrinsic pathway is initiated when the vascular lumen of blood vessels is damaged. The membrane glycoprotein tissue factor is exposed and then binds to circulating factor VII (FVII) and to small preexisting amounts of its activated form FVIIa. This binding facilitates full conversion of FVII to FVIIa and subsequently, in the presence of calcium and phospholipids, the conversion of factor IX (FIX) to factor IXa (FIXa) and factor X (FX) to factor Xa (FXa). The association of FVIIa with tissue factor enhances the proteolytic activity by bringing the binding sites of FVII for the substrate (FIX and FX) into closer proximity and by inducing a conformational change, which enhances the enzymatic activity of FVIIa.
[0082] The activation of FX is the common point of the two pathways. Along with phospholipid and calcium, factors Va (FVa) and Xa convert prothrombin to thrombin (prothrombinase complex), which then cleaves fibrinogen to form fibrin monomers. The monomers polymerize to form fibrin strands. Factor XIIIa (FXIIIa) covalently bonds these strands to one another to form a rigid mesh.
[0083] Conversion of FVII to FVIIa is also catalyzed by a number of proteases, including thrombin, FIXa, FXa, factor XIa (FXIa), and factor XIIa (FXIIa). For inhibition of the early phase of the cascade, tissue factor pathway inhibitor targets FVIIa/tissue factor/FXa product complex.
Factor VIIa
[0084] FVII (also known as stable factor or proconvertin) is a vitamin K-dependent serine protease glycoprotein with a pivotal role in hemostasis and coagulation (Eigenbrot, Curr Protein Pept Sci. 2002; 3:287-99).
[0085] FVII is synthesized in the liver and secreted as a single-chain glycoprotein of 48 kD. FVII shares with all vitamin K-dependent serine protease glycoproteins a similar protein domain structure consisting of an amino-terminal gamma-carboxyglutamic acid (Gla) domain with 9-12 residues responsible for the interaction of the protein with lipid membranes, a carboxy-terminal serine protease domain (catalytic domain), and two epidermal growth factor-like domains containing a calcium ion binding site that mediates interaction with tissue factor. Gamma-glutamyl carboxylase catalyzes carboxylation of Gla residues in the amino-terminal portion of the molecule. The carboxylase is dependent on a reduced form of vitamin K for its action, which is oxidized to the epoxide form. Vitamin K epoxide reductase is required to convert the epoxide form of vitamin K back to the reduced form.
[0086] The major proportion of FVII circulates in plasma in zymogen form, and activation of this form results in cleavage of the peptide bond between arginine 152 and isoleucine 153. The resulting activated FVIIa consists of a NH2-derived light chain (20 kD) and a COOH terminal-derived heavy chain (30 kD) linked via a single disulfide bond (Cys 135 to Cys 262). The light chain contains the membrane-binding Gla domain, while the heavy chain contains the catalytic domain.
[0087] The plasma concentration of FVII determined by genetic and environmental factors is about 0.5 mg/mL (Pinotti et al., Blood. 2000; 95:3423-8). Different FVII genotypes can result in several-fold differences in mean FVII levels. Plasma FVII levels are elevated during pregnancy in healthy females and also increase with age and are higher in females and in persons with hypertriglyceridemia. FVII has the shortest half-life of all procoagulant factors (3-6 h). The mean plasma concentration of FVIIa is 3.6 ng/mL in healthy individuals and the circulating half-life of FVIIa is relatively long (2.5 h) compared with other coagulation factors.
[0088] Hereditary FVII deficiency is a rare autosomal recessive bleeding disorder with a prevalence estimated to be 1 case per 500,000 persons in the general population (Acharya et al., J Thromb Haemost. 2004; 2248-56). Acquired FVII deficiency from inhibitors is also very rare. Cases have also been reported with the deficiency occurring in association with drugs such as cephalosporins, penicillins, and oral anticoagulants. Furthermore, acquired FVII deficiency has been reported to occur spontaneously or with other conditions, such as myeloma, sepsis, aplastic anemia, with interleukin-2 and antithymocyte globulin therapy.
[0089] Reference polynucleotide and polypeptide sequences include, e.g., GenBank Accession Nos. J02933 for the genomic sequence, M13232 for the cDNA (Hagen et al. PNAS 1986; 83: 2412-6), and P08709 for the polypeptide sequence (references incorporated herein in their entireties). A variety of polymorphisms of FVII have been described, for example see Sabater-Lleal et al. (Hum Genet. 2006; 118:741-51) (reference incorporated herein in its entirety).
Factor IX
[0090] FIX is a vitamin K-dependent plasma protein that participates in the intrinsic pathway of blood coagulation by converting FX to its active form in the presence of calcium ions, phospholipids and FVIIIa. The predominant catalytic capability of FIX is as a serine protease with specificity for a particular arginine-isoleucine bond within FX. Activation of FIX occurs by FXIa which causes excision of the activation peptide from FIX to produce an activated FIX molecule comprising two chains held by one or more disulphide bonds. Defects in FIX are the cause of recessive X-linked hemophilia B.
[0091] Hemophilia A and B are inherited diseases characterized by deficiencies in FVIII and FIX polypeptides, respectively. The underlying cause of the deficiencies is frequently the result of mutations in FVIII and FIX genes, both of which are located on the X chromosome. Traditional therapy for hemophilias often involves intravenous administration of pooled plasma or semi-purified coagulation proteins from normal individuals. These preparations can be contaminated by pathogenic agents or viruses, such as infectious prions, HIV, parvovirus, hepatitis A, and hepatitis C. Hence, there is an urgent need for therapeutic agents that do not require the use of human serum.
[0092] The level of the decrease in FIX activity is directly proportional to the severity of hemophilia B. The current treatment of hemophilia B consists of the replacement of the missing protein by plasma-derived or recombinant FIX (so-called FIX substitution or replacement treatment or therapy).
[0093] Polynucleotide and polypeptide sequences of FIX can be found for example in the UniProtKB/Swiss-Prot Accession No. P00740, U.S. Pat. No. 6,531,298 and in FIG. 1 (SEQ ID NO: 1).
Factor VIII
[0094] Coagulation factor VIII (FVIII) circulates in plasma at a very low concentration and is bound non-covalently to Von Willebrand factor (VWF). During hemostasis, FVIII is separated from VWF and acts as a cofactor for activated factor IX (FIXa)-mediated FX activation by enhancing the rate of activation in the presence of calcium and phospholipids or cellular membranes.
[0095] FVIII is synthesized as a single-chain precursor of approximately 270-330 kD with the domain structure A1-A2-B-A3-C1-C2. When purified from plasma (e.g., "plasma-derived" or "plasmatic"), FVIII is composed of a heavy chain (A1-A2-B) and a light chain (A3-C1-C2). The molecular mass of the light chain is 80 kD whereas, due to proteolysis within the B domain, the heavy chain is in the range of 90-220 kD.
[0096] FVIII is also synthesized as a recombinant protein for therapeutic use in bleeding disorders. Various in vitro assays have been devised to determine the potential efficacy of recombinant FVIII (rFVIII) as a therapeutic medicine. These assays mimic the in vivo effects of endogenous FVIII. In vitro thrombin treatment of FVIII results in a rapid increase and subsequent decrease in its procoagulant activity, as measured by in vitro assays. This activation and inactivation coincides with specific limited proteolysis both in the heavy and the light chains, which alter the availability of different binding epitopes in FVIII, e.g. allowing FVIII to dissociate from VWF and bind to a phospholipid surface or altering the binding ability to certain monoclonal antibodies.
[0097] The lack or dysfunction of FVIII is associated with the most frequent bleeding disorder, hemophilia A. The treatment of choice for the management of hemophilia A is replacement therapy with plasma derived or rFVIII concentrates. Patients with severe hemophilia A with FVIII levels below 1%, are generally on prophylactic therapy with the aim of keeping FVIII above 1% between doses. Taking into account the average half-lives of the various FVIII products in the circulation, this result can usually be achieved by giving FVIII two to three times a week.
[0098] Reference polynucleotide and polypeptide sequences include, e.g., UniProtKB/Swiss-Prot P00451 (FA8 HUMAN); Gitschier J et al., Characterization of the human Factor VIII gene, Nature, 312(5992): 326-30 (1984); Vehar G H et al., Structure of human Factor VIII, Nature, 312(5992):337-42 (1984); Thompson A R. Structure and Function of the Factor VIII gene and protein, Semin Thromb Hemost, 2003:29; 11-29 (2002).
Von Willebrand Factor
[0099] Von Willebrand factor (VWF) is a glycoprotein circulating in plasma as a series of multimers ranging in size from about 500 to 20,000 kD. Multimeric forms of VWF are composed of 250 kD polypeptide subunits linked together by disulfide bonds. VWF mediates initial platelet adhesion to the sub-endothelium of the damaged vessel wall. Only the larger multimers exhibit hemostatic activity. It is assumed that endothelial cells secrete large polymeric forms of VWF and those forms of VWF which have a low molecular weight (low molecular weight VWF) arise from proteolytic cleavage. The multimers having large molecular masses are stored in the Weibel-Pallade bodies of endothelial cells and liberated upon stimulation.
[0100] VWF is synthesized by endothelial cells and megakaryocytes as prepro-VWF that consists to a large extent of repeated domains. Upon cleavage of the signal peptide, pro-VWF dimerizes through disulfide linkages at its C-terminal region. The dimers serve as protomers for multimerization, which is governed by disulfide linkages between the free end termini. The assembly to multimers is followed by the proteolytic removal of the propeptide sequence (Leyte et al., Biochem. J. 274 (1991), 257-261).
[0101] The primary translation product predicted from the cloned cDNA of VWF is a 2813-residue precursor polypeptide (prepro-VWF). The prepro-VWF consists of a 22 amino acid signal peptide and a 741 amino acid propeptide, with the mature VWF comprising 2050 amino acids (Ruggeri Z. A., and Ware, J., FASEB J., 308-316 (1993).
[0102] Defects in VWF are causal to Von Willebrand disease (VWD), which is characterized by a more or less pronounced bleeding phenotype. VWD type 3 is the most severe form, in which VWF is completely missing, and VWD type 1 relates to a quantitative loss of VWF and its phenotype can be very mild. VWD type 2 relates to qualitative defects of VWF and can be as severe as VWD type 3. VWD type 2 has many sub forms, some being associated with the loss or the decrease of high molecular weight multimers. Von Willebrand disease type 2a (VWD-2A) is characterized by a loss of both intermediate and large multimers. VWD-2B is characterized by a loss of highest-molecular-weight multimers. Other diseases and disorders related to VWF are known in the art.
[0103] The polynucleotide and amino acid sequences of prepro-VWF are available at GenBank Accession Nos. NM_000552 and NP_000543, respectively.
[0104] Other blood coagulation proteins according to the present invention are described in the art, e.g. Mann K G, Thromb Haemost, 1999; 82:165-74.
A. Polypeptides
[0105] In one aspect, the starting material of the present invention is a protein or polypeptide. As described herein, the term therapeutic protein refers to any therapeutic protein molecule which exhibits biological activity that is associated with the therapeutic protein. In one embodiment of the invention, the therapeutic protein molecule is a full-length protein.
[0106] Therapeutic protein molecules contemplated include full-length proteins, precursors of full length proteins, biologically active subunits or fragments of full length proteins, as well as biologically active derivatives and variants of any of these forms of therapeutic proteins. Thus, therapeutic protein include those that (1) have an amino acid sequence that has greater than about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98% or about 99% or greater amino acid sequence identity, over a region of at least about 25, about 50, about 100, about 200, about 300, about 400, or more amino acids, to a polypeptide encoded by a referenced nucleic acid or an amino acid sequence described herein; and/or (2) specifically bind to antibodies, e.g., polyclonal or monoclonal antibodies, generated against an immunogen comprising a referenced amino acid sequence as described herein, an immunogenic fragment thereof, and/or a conservatively modified variant thereof.
[0107] According to the present invention, the term "recombinant therapeutic protein" includes any therapeutic protein obtained via recombinant DNA technology. In certain embodiments, the term encompasses proteins as described herein.
[0108] As used herein, "endogenous therapeutic protein" includes a therapeutic protein which originates from the mammal intended to receive treatment. The term also includes therapeutic protein transcribed from a transgene or any other foreign DNA present in said mammal. As used herein, "exogenous therapeutic protein" includes a blood coagulation protein which does not originate from the mammal intended to receive treatment.
[0109] As used herein, "plasma-derived blood coagulation protein" or "plasmatic" includes all forms of the protein found in blood obtained from a mammal having the property participating in the coagulation pathway.
[0110] As used herein "biologically active derivative" or "biologically active variant" includes any derivative or variant of a molecule having substantially the same functional and/or biological properties of said molecule, such as binding properties, and/or the same structural basis, such as a peptidic backbone or a basic polymeric unit.
[0111] An "analog," such as a "variant" or a "derivative," is a compound substantially similar in structure and having the same biological activity, albeit in certain instances to a differing degree, to a naturally-occurring molecule. For example, a polypeptide variant refers to a polypeptide sharing substantially similar structure and having the same biological activity as a reference polypeptide. Variants or analogs differ in the composition of their amino acid sequences compared to the naturally-occurring polypeptide from which the analog is derived, based on one or more mutations involving (i) deletion of one or more amino acid residues at one or more termini of the polypeptide and/or one or more internal regions of the naturally-occurring polypeptide sequence (e.g., fragments), (ii) insertion or addition of one or more amino acids at one or more termini (typically an "addition" or "fusion") of the polypeptide and/or one or more internal regions (typically an "insertion") of the naturally-occurring polypeptide sequence or (iii) substitution of one or more amino acids for other amino acids in the naturally-occurring polypeptide sequence. By way of example, a "derivative" is a type of analog and refers to a polypeptide sharing the same or substantially similar structure as a reference polypeptide that has been modified, e.g., chemically.
[0112] A variant polypeptide is a type of analog polypeptide and includes insertion variants, wherein one or more amino acid residues are added to a therapeutic protein amino acid sequence of the invention. Insertions may be located at either or both termini of the protein, and/or may be positioned within internal regions of the therapeutic protein amino acid sequence. Insertion variants, with additional residues at either or both termini, include for example, fusion proteins and proteins including amino acid tags or other amino acid labels. In one aspect, the blood coagulation protein molecule optionally contains an N-terminal Met, especially when the molecule is expressed recombinantly in a bacterial cell such as E. coli.
[0113] In deletion variants, one or more amino acid residues in a therapeutic protein polypeptide as described herein are removed. Deletions can be effected at one or both termini of the therapeutic protein polypeptide, and/or with removal of one or more residues within the therapeutic protein amino acid sequence. Deletion variants, therefore, include fragments of a therapeutic protein polypeptide sequence.
[0114] In substitution variants, one or more amino acid residues of a therapeutic protein polypeptide are removed and replaced with alternative residues. In one aspect, the substitutions are conservative in nature and conservative substitutions of this type are well known in the art. Alternatively, the invention embraces substitutions that are also non-conservative. Exemplary conservative substitutions are described in Lehninger, [Biochemistry, 2nd Edition; Worth Publishers, Inc., New York (1975), pp. 71-77] and are set out immediately below.
Conservative Substitutions
TABLE-US-00002 [0115] SIDE CHAIN CHARACTERISTIC AMINO ACID Non-polar (hydrophobic): A. Aliphatic A L I V P B. Aromatic F W C. Sulfur-containing M D. Borderline G Uncharged-polar: A. Hydroxyl S T Y B. Amides N Q C. Sulfhydryl C D. Borderline G Positively charged (basic) K R H Negatively charged (acidic) D E
[0116] Alternatively, exemplary conservative substitutions are set out immediately below.
Conservative Substitutions II
TABLE-US-00003 [0117] EXEMPLARY ORIGINAL RESIDUE SUBSTITUTION Ala (A) Val, Leu, Ile Arg (R) Lys, Gln, Asn Asn (N) Gln, His, Lys, Arg Asp (D) Glu Cys (C) Ser Gln (Q) Asn Glu (E) Asp His (H) Asn, Gln, Lys, Arg Ile (I) Leu, Val, Met, Ala, Phe, Leu (L) Ile, Val, Met, Ala, Phe Lys (K) Arg, Gln, Asn Met (M) Leu, Phe, Ile Phe (F) Leu, Val, Ile, Ala Pro (P) Gly Ser (S) Thr Thr (T) Ser Trp (W) Tyr Tyr (Y) Trp, Phe, Thr, Ser Val (V) Ile, Leu, Met, Phe, Ala
B. Polynucleotides
[0118] Nucleic acids encoding a therapeutic protein of the invention include, for example and without limitation, genes, pre-mRNAs, mRNAs, cDNAs, polymorphic variants, alleles, synthetic and naturally-occurring mutants.
[0119] Polynucleotides encoding a therapeutic protein of the invention also include, without limitation, those that (1) specifically hybridize under stringent hybridization conditions to a nucleic acid encoding a referenced amino acid sequence as described herein, and conservatively modified variants thereof; (2) have a nucleic acid sequence that has greater than about 95%, about 96%, about 97%, about 98%, about 99%, or higher nucleotide sequence identity, over a region of at least about 25, about 50, about 100, about 150, about 200, about 250, about 500, about 1000, or more nucleotides (up to the full length sequence of 1218 nucleotides of the mature protein), to a reference nucleic acid sequence as described herein. Exemplary "stringent hybridization" conditions include hybridization at 42.degree. C. in 50% formamide, 5.times.SSC, 20 mM Na.PO4, pH 6.8; and washing in 1.times.SSC at 55.degree. C. for 30 minutes. It is understood that variation in these exemplary conditions can be made based on the length and GC nucleotide content of the sequences to be hybridized. Formulas standard in the art are appropriate for determining appropriate hybridization conditions. See Sambrook et al., Molecular Cloning: A Laboratory Manual (Second ed., Cold Spring Harbor Laboratory Press, 1989) .sctn..sctn. 9.47-9.51.
[0120] A "naturally-occurring" polynucleotide or polypeptide sequence is typically derived from a mammal including, but not limited to, primate, e.g., human; rodent, e.g., rat, mouse, hamster; cow, pig, horse, sheep, or any mammal. The nucleic acids and proteins of the invention can be recombinant molecules (e.g., heterologous and encoding the wild type sequence or a variant thereof, or non-naturally occurring).
C. Production of Therapeutic Proteins
[0121] Production of a therapeutic protein includes any method known in the art for (i) the production of recombinant DNA by genetic engineering, (ii) introducing recombinant DNA into prokaryotic or eukaryotic cells by, for example and without limitation, transfection, electroporation or microinjection, (iii) cultivating said transformed cells, (iv) expressing therapeutic protein, e.g. constitutively or upon induction, and (v) isolating said blood coagulation protein, e.g. from the culture medium or by harvesting the transformed cells, in order to obtain purified therapeutic protein.
[0122] In other aspects, the therapeutic protein is produced by expression in a suitable prokaryotic or eukaryotic host system characterized by producing a pharmacologically acceptable blood coagulation protein molecule. Examples of eukaryotic cells are mammalian cells, such as CHO, COS, HEK 293, BHK, SK-Hep, and HepG2.
[0123] A wide variety of vectors are used for the preparation of the therapeutic protein and are selected from eukaryotic and prokaryotic expression vectors. Examples of vectors for prokaryotic expression include plasmids such as, and without limitation, pRSET, pET, and pBAD, wherein the promoters used in prokaryotic expression vectors include one or more of, and without limitation, lac, trc, trp, recA, or araBAD. Examples of vectors for eukaryotic expression include: (i) for expression in yeast, vectors such as, and without limitation, pAO, pPIC, pYES, or pMET, using promoters such as, and without limitation, AOX1, GAP, GAL1, or AUG1; (ii) for expression in insect cells, vectors such as and without limitation, pMT, pAc5, pIB, pMIB, or pBAC, using promoters such as and without limitation PH, p10, MT, Ac5, OpIE2, gp64, or polh, and (iii) for expression in mammalian cells, vectors such as and without limitation pSVL, pCMV, pRc/RSV, pcDNA3, or pBPV, and vectors derived from, in one aspect, viral systems such as and without limitation vaccinia virus, adeno-associated viruses, herpes viruses, or retroviruses, using promoters such as and without limitation CMV, SV40, EF-1, UbC, RSV, ADV, BPV, and .beta.-actin.
D. Administration
[0124] In one embodiment a conjugated therapeutic protein of the present invention may be administered by injection, such as intravenous, intramuscular, or intraperitoneal injection.
[0125] To administer compositions comprising a conjugated therapeutic protein of the present invention to human or test animals, in one aspect, the compositions comprise one or more pharmaceutically acceptable carriers. The terms "pharmaceutically" or "pharmacologically acceptable" refer to molecular entities and compositions that are stable, inhibit protein degradation such as aggregation and cleavage products, and in addition do not produce allergic, or other adverse reactions when administered using routes well-known in the art, as described below. "Pharmaceutically acceptable carriers" include any and all clinically useful solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents and the like, including those agents disclosed above.
[0126] As used herein, "effective amount" includes a dose suitable for treating a disease or disorder or ameliorating a symptom of a disease or disorder. In one embodiment, "effective amount" includes a dose suitable for treating a mammal having a bleeding disorder as described herein.
[0127] The compositions may be administered orally, topically, transdermally, parenterally, by inhalation spray, vaginally, rectally, or by intracranial injection. The term parenteral as used herein includes subcutaneous injections, intravenous, intramuscular, intracisternal injection, or infusion techniques. Administration by intravenous, intradermal, intramuscular, intramammary, intraperitoneal, intrathecal, retrobulbar, intrapulmonary injection and or surgical implantation at a particular site is contemplated as well. Generally, compositions are essentially free of pyrogens, as well as other impurities that could be harmful to the recipient.
[0128] Single or multiple administrations of the compositions can be carried out with the dose levels and pattern being selected by the treating physician. For the prevention or treatment of disease, the appropriate dosage will depend on the type of disease to be treated, as described above, the severity and course of the disease, whether drug is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the drug, and the discretion of the attending physician.
[0129] The present invention also relates to a pharmaceutical composition comprising an effective amount of a conjugated therapeutic protein as defined herein. The pharmaceutical composition may further comprise a pharmaceutically acceptable carrier, diluent, salt, buffer, or excipient. The pharmaceutical composition can be used for treating the above-defined bleeding disorders. The pharmaceutical composition of the invention may be a solution or a lyophilized product. Solutions of the pharmaceutical composition may be subjected to any suitable lyophilization process.
[0130] As an additional aspect, the invention includes kits which comprise a composition of the invention packaged in a manner which facilitates its use for administration to subjects. In one embodiment, such a kit includes a compound or composition described herein (e.g., a composition comprising a conjugated therapeutic protein), packaged in a container such as a sealed bottle or vessel, with a label affixed to the container or included in the package that describes use of the compound or composition in practicing the method. In one embodiment, the kit contains a first container having a composition comprising a conjugated therapeutic protein and a second container having a physiologically acceptable reconstitution solution for the composition in the first container. In one aspect, the compound or composition is packaged in a unit dosage form. The kit may further include a device suitable for administering the composition according to a specific route of administration. Preferably, the kit contains a label that describes use of the therapeutic protein or peptide composition.
Water Soluble Polymers
[0131] In one aspect, a therapeutic protein derivative (i.e., a conjugated therapeutic protein) molecule provided is bound to a water-soluble polymer including, but not limited to, polyethylene glycol (PEG), branched PEG, polysialic acid (PSA), hydroxyalkyl starch (HAS), hydroxylethyl starch (HES), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG) polyoxazoline, poly acryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC). In one embodiment of the invention, the water soluble polymer is consisting of sialic acid molecule having a molecular weight range of 350 to 120,000, 500 to 100,000, 1000 to 80,000, 1500 to 60,000, 2,000 to 45,000 Da, 3,000 to 35,000 Da, and 5,000 to 25,000 Da. The coupling of the water soluble polymer can be carried out by direct coupling to the protein or via linker molecules. One example of a chemical linker is MBPH (4-[4-N-Maleimidophenyl]butyric acid hydrazide) containing a carbohydrate-selective hydrazide and a sulfhydryl-reactive maleimide group (Chamow et al., J Biol Chem 1992; 267:15916-22). Other exemplary and preferred linkers are described below.
[0132] In one embodiment, the derivative retains the full functional activity of native therapeutic protein products, and provides an extended half-life in vivo, as compared to native therapeutic protein products. In another embodiment, the derivative retains at least 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44. 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 110, 120, 130, 140, or 150 percent (%) biological activity relative to native blood coagulation protein. In a related aspect, the biological activities of the derivative and native blood coagulation protein are determined by the ratios of chromogenic activity to blood coagulation factor antigen value (blood coagulation factor:Chr:blood coagulation factor:Ag). In still another embodiment of the invention, the half-life of the construct is decreased or increased 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1.1, 1.2, 1.3, 1.4, 1.5, 2, 3, 4, 5, 6, 7, 8, 9, or 10-fold relative to the in vivo half-life of native therapeutic protein.
A. Sialic Acid and PSA
[0133] PSAs consist of polymers (generally homopolymers) of N-acetylneuraminic acid. The secondary amino group normally bears an acetyl group, but it may instead bear a glycolyl group. Possible substituents on the hydroxyl groups include acetyl, lactyl, ethyl, sulfate, and phosphate groups.
##STR00007##
[0134] Structure of Sialic Acid (N-Acetylneuraminic Acid)
[0135] PSAs and mPSAs generally comprise linear polymers consisting essentially of N-acetylneuraminic acid moieties linked by 2,8- or 2,9-glycosidic linkages or combinations of these (e.g. alternating 2,8- and 2,9-linkages). In particularly preferred PSAs and mPSAs, the glycosidic linkages are .alpha.-2,8. Such PSAs and mPSAs are conveniently derived from colominic acids, and are referred to herein as "CAs" and "mCAs". Typical PSAs and mPSAs comprise at least 2, preferably at least 5, more preferably at least 10 and most preferably at least 20 N-acetylneuraminic acid moieties. Thus, they may comprise from 2 to 300 N-acetylneuraminic acid moieties, preferably from 5 to 200 N-acetylneuraminic acid moieties, or most preferably from 10 to 100 N-acetylneuraminic acid moieties. PSAs and CAs preferably are essentially free of sugar moieties other than N-acetylneuraminic acid. Thus PSAs and CAs preferably comprise at least 90%, more preferably at least 95% and most preferably at least 98% N-acetylneuraminic acid moieties.
[0136] Where PSAs and CAs comprise moieties other than N-acetylneuraminic acid (as, for example in mPSAS and mCAs) these are preferably located at one or both of the ends of the polymer chain. Such "other" moieties may, for example, be moieties derived from terminal N-acetylneuraminic acid moieties by oxidation or reduction.
[0137] For example, WO-A-0187922 describes such mPSAs and mCAs in which the non-reducing terminal N-acetylneuraminic acid unit is converted to an aldehyde group by reaction with sodium periodate. Additionally, WO 2005/016974 describes such mPSAs and mCAs in which the reducing terminal N-acetylneuraminic acid unit is subjected to reduction to reductively open the ring at the reducing terminal N-acetylneuraminic acid unit, whereby a vicinal diol group is formed, followed by oxidation to convert the vicinal diol group to an aldehyde group.
[0138] Sialic acid rich glycoproteins bind selectin in humans and other organisms. They play an important role in human influenza infections. E.g., sialic acid can hide mannose antigens on the surface of host cells or bacteria from mannose-binding lectin. This prevents activation of complement. Sialic acids also hide the penultimate galactose residue thus preventing rapid clearance of the glycoprotein by the galactose receptor on the hepatic parenchymal cells.
##STR00008##
[0139] Structure of Colominic Acid (Homopolymer of N-Acetylneuraminic Acid)
[0140] Colominic acids (a sub-class of PSAs) are homopolymers of N-acetylneuraminic acid (NANA) with .alpha. (2.fwdarw.8) ketosidic linkage, and are produced, inter alia, by particular strains of Escherichia coli possessing K1 antigen. Colominic acids have many physiological functions. They are important as a raw material for drugs and cosmetics.
[0141] Comparative studies in vivo with polysialylated and unmodified asparaginase revealed that polysialylation increased the half-life of the enzyme (Fernandes and Gregoriadis, Biochimica Biophysica Acta 1341: 26-34, 1997).
[0142] As used herein, "sialic acid moieties" includes sialic acid monomers or polymers ("polysaccharides") which are soluble in an aqueous solution or suspension and have little or no negative impact, such as side effects, to mammals upon administration of the PSA-blood coagulation protein conjugate in a pharmaceutically effective amount. The polymers are characterized, in one aspect, as having 1, 2, 3, 4, 5, 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, or 500 sialic acid units. In certain aspects, different sialic acid units are combined in a chain.
[0143] In one embodiment of the invention, the sialic acid portion of the polysaccharide compound is highly hydrophilic, and in another embodiment the entire compound is highly hydrophilic. Hydrophilicity is conferred primarily by the pendant carboxyl groups of the sialic acid units, as well as the hydroxyl groups. The saccharide unit may contain other functional groups, such as, amine, hydroxyl or sulphate groups, or combinations thereof. These groups may be present on naturally-occurring saccharide compounds, or introduced into derivative polysaccharide compounds.
[0144] The naturally occurring polymer PSA is available as a polydisperse preparation showing a broad size distribution (e.g. Sigma C-5762) and high polydispersity (PD). Because the polysaccharides are usually produced in bacteria carrying the inherent risk of copurifying endotoxins, the purification of long sialic acid polymer chains may raise the probability of increased endotoxin content. Short PSA molecules with 1-4 sialic acid units can also be synthetically prepared (Kang S H et al., Chem Commun. 2000; 227-8; Ress D K and Linhardt R J, Current Organic Synthesis. 2004; 1:31-46), thus minimizing the risk of high endotoxin levels. However PSA preparations with a narrow size distribution and low polydispersity, which are also endotoxin-free, can now be manufactured. Polysaccharide compounds of particular use for the invention are, in one aspect, those produced by bacteria. Some of these naturally-occurring polysaccharides are known as glycolipids. In one embodiment, the polysaccharide compounds are substantially free of terminal galactose units.
B. Polyethylene Glycol (PEG) and Pegylation
[0145] In certain aspects, therapeutic proteins are conjugated to a water soluble polymer by any of a variety of chemical methods (Roberts J M et al., Advan Drug Delivery Rev 2002; 54:459-76). For example, in one embodiment a therapeutic protein is modified by the conjugation of PEG to free amino groups of the protein using N-hydroxysuccinimide (NHS) esters. In another embodiment the water soluble polymer, for example PEG, is coupled to free SH groups using maleimide chemistry or the coupling of PEG hydrazides or PEG amines to carbohydrate moieties of the therapeutic protein after prior oxidation.
[0146] The conjugation is in one aspect performed by direct coupling (or coupling via linker systems) of the water soluble polymer to a therapeutic protein under formation of stable bonds. In addition degradable, releasable or hydrolysable linker systems are used in certain aspects the present invention (Tsubery et al. J Biol Chem 2004; 279:38118-24/Greenwald et al., J Med Chem 1999; 42:3657-67/Zhao et al., Bioconj Chem 2006; 17:341-51/WO2006/138572A2/U.S. Pat. No. 7,259,224B2/U.S. Pat. No. 7,060,259B2).
[0147] In one embodiment of the invention, a therapeutic protein is modified via lysine residues by use of polyethylene glycol derivatives containing an active N-hydroxysuccinimide ester (NHS) such as succinimidyl succinate, succinimidyl glutarate or succinimidyl propionate. These derivatives react with the lysine residues of the therapeutic protein under mild conditions by forming a stable amide bond. In one embodiment of the invention, the chain length of the PEG derivative is 5,000 Da. Other PEG derivatives with chain lengths of 500 to 2,000 Da, 2,000 to 5,000 Da, greater than 5,000 up to 10,000 Da or greater than 10,000 up to 20,000 Da, or greater than 20,000 up to 150,000 Da are used in various embodiments, including linear and branched structures.
[0148] Alternative methods for the PEGylation of amino groups are, without limitation, the chemical conjugation with PEG carbonates by forming urethane bonds, or the reaction with aldehydes or ketones by reductive amination forming secondary amide bonds.
[0149] In one embodiment of the present invention a therapeutic protein molecule is chemically modified using PEG derivatives that are commercially available. These PEG derivatives in alternative aspects have linear or branched structures. Examples of PEG-derivatives containing NHS groups are listed below.
[0150] The following PEG derivatives are non-limiting examples of those commercially available from Nektar Therapeutics (Huntsville, Ala.; see www.nektar.com/PEG reagent catalog; Nektar Advanced PEGylation, price list 2005-2006):
##STR00009##
##STR00010##
[0151] This reagent with branched structure is described in more detail by Kozlowski et al. (BioDrugs 2001; 5:419-29).
[0152] Other non-limiting examples of PEG derivatives are commercially available from NOF Corporation (Tokyo, Japan; see www.nof.co.jp/english: Catalogue 2005)
##STR00011##
[0153] Structures of Branched PEG-Derivatives (NOF Corp.):
##STR00012##
[0154] These propane derivatives show a glycerol backbone with a 1,2 substitution pattern. In the present invention branched PEG derivatives based on glycerol structures with 1,3 substitution or other branched structures described in US2003/0143596A1 are also contemplated.
[0155] PEG derivatives with degradable (for example, hydrolysable) linkers as described by Tsubery et al. (J Biol Chem 2004; 279:38118-24) and Shechter et al. (WO04089280A3) are also contemplated.
[0156] Surprisingly, the PEGylated therapeutic protein of this invention exhibits functional activity, combined with an extended half-life in vivo. In addition the PEGylated rFVIII, FVIIa, FIX, or other blood coagulation factor seems to be more resistant against thrombin inactivation.
C. Hydroxyalkyl Starch (HAS) and Hydroxylethyl Starch (HES)
[0157] In various embodiments of the present invention, a therapeutic protein molecule is chemically modified using hydroxyalkyl starch (HAS) or hydroxylethyl starch (HES) or derivatives thereof.
[0158] HES is a derivative of naturally occurring amylopectin and is degraded by alpha-amylase in the body. HES is a substituted derivative of the carbohydrate polymer amylopectin, which is present in corn starch at a concentration of up to 95% by weight. HES exhibits advantageous biological properties and is used as a blood volume replacement agent and in hemodilution therapy in the clinics (Sommermeyer et al., 1987, Krankenhauspharmazie, 8 (8), 271-278; and Weidler et al., 1991, Arzneim.-Forschung/Drug Res. g 419 494-498).
[0159] Amylopectin consists of glucose moieties, wherein in the main chain alpha-1,4-glycosidic bonds are present and at the branching sites alpha-1, 6-glycosidic bonds are found. The physical-chemical properties of this molecule are mainly determined by the type of glycosidic bonds. Due to the nicked alpha-1,4-glycosidic bond, helical structures with about six glucose-monomers per turn are produced. The physico-chemical as well as the biochemical properties of the polymer can be modified via substitution. The introduction of a hydroxyethyl group can be achieved via alkaline hydroxyethylation. By adapting the reaction conditions it is possible to exploit the different reactivity of the respective hydroxy group in the unsubstituted glucose monomer with respect to a hydroxyethylation. Owing to this fact, the skilled person is able to influence the substitution pattern to a limited extent.
[0160] HAS refers to a starch derivative which has been substituted by at least one hydroxyalkyl group. Therefore, the term hydroxyalkyl starch is not limited to compounds where the terminal carbohydrate moiety comprises hydroxyalkyl groups R1, R2, and/or R3, but also refers to compounds in which at least one hydroxy group present anywhere, either in the terminal carbohydrate moiety and/or in the remaining part of the starch molecule, HAS', is substituted by a hydroxyalkyl group R1, R2, or R3.
##STR00013##
[0161] The alkyl group may be a linear or branched alkyl group which may be suitably substituted. Preferably, the hydroxyalkyl group contains 1 to 10 carbon atoms, more preferably from 1 to 6 carbon atoms, more preferably from 1 to 4 carbon atoms, and even more preferably 2-4 carbon atoms. "Hydroxyalkyl starch" therefore preferably comprises hydroxyethyl starch, hydroxypropyl starch and hydroxybutyl starch, wherein hydroxyethyl starch and hydroxypropyl starch are particularly preferred.
[0162] Hydroxyalkyl starch comprising two or more different hydroxyalkyl groups is also comprised in the present invention. The at least one hydroxyalkyl group comprised in HAS may contain two or more hydroxy groups. According to one embodiment, the at least one hydroxyalkyl group comprised HAS contains one hydroxy group.
[0163] The term HAS also includes derivatives wherein the alkyl group is mono- or polysubstituted. In one embodiment, the alkyl group is substituted with a halogen, especially fluorine, or with an aryl group, provided that the HAS remains soluble in water. Furthermore, the terminal hydroxy group a of hydroxyalkyl group may be esterified or etherified. HAS derivatives are described in WO/2004/024776, which is incorporated by reference in its entirety.
D. Methods of Attachment
[0164] A therapeutic protein may be covalently linked to the polysaccharide compounds by any of various techniques known to those of skill in the art. In various aspects of the invention, sialic acid moieties are bound to a therapeutic protein, e.g., FIX, FVIII, FVIIa or VWF, for example by the method described in U.S. Pat. No. 4,356,170, which is herein incorporated by reference.
[0165] Other techniques for coupling PSA to polypeptides are also known and contemplated by the invention. For example, US Publication No. 2007/0282096 describes conjugating an amine or hydrazide derivative of, e.g., PSA, to proteins. In addition, US Publication No. 2007/0191597 describes PSA derivatives containing an aldehyde group for reaction with substrates (e.g., proteins) at the reducing end. These references are incorporated by reference in their entireties.
[0166] Various methods are disclosed at column 7, line 15, through column 8, line 5 of U.S. Pat. No. 5,846,951 (incorporated by reference in its entirety). Exemplary techniques include linkage through a peptide bond between a carboxyl group on one of either the blood coagulation protein or polysaccharide and an amine group of the blood coagulation protein or polysaccharide, or an ester linkage between a carboxyl group of the blood coagulation protein or polysaccharide and a hydroxyl group of the therapeutic protein or polysaccharide. Another linkage by which the therapeutic protein is covalently bonded to the polysaccharide compound is via a Schiff base, between a free amino group on the blood coagulation protein being reacted with an aldehyde group formed at the non-reducing end of the polysaccharide by periodate oxidation (Jennings H J and Lugowski C, J Immunol. 1981; 127:1011-8; Fernandes A I and Gregoriadis G, Biochim Biophys Acta. 1997; 1341; 26-34). The generated Schiff base is in one aspect stabilized by specific reduction with NaCNBH3 to form a secondary amine. An alternative approach is the generation of terminal free amino groups in the PSA by reductive amination with NH4C1 after prior oxidation. Bifunctional reagents can be used for linking two amino or two hydroxyl groups. For example, PSA containing an amino group is coupled to amino groups of the protein with reagents like BS3 (Bis(sulfosuccinimidyl)suberate/Pierce, Rockford, Ill.). In addition heterobifunctional cross linking reagents like Sulfo-EMCS (N-.epsilon.-Maleimidocaproyloxy) sulfosuccinimide ester/Pierce) is used for instance to link amine and thiol groups.
[0167] In another approach, a PSA hydrazide is prepared and coupled to the carbohydrate moiety of the protein after prior oxidation and generation of aldehyde functions.
[0168] As described above, a free amine group of the therapeutic protein reacts with the 1-carboxyl group of the sialic acid residue to form a peptidyl bond or an ester linkage is formed between the 1-carboxylic acid group and a hydroxyl or other suitable active group on a blood coagulation protein. Alternatively, a carboxyl group forms a peptide linkage with deacetylated 5-amino group, or an aldehyde group of a molecule of a therapeutic protein forms a Schiff base with the N-deacetylated 5-amino group of a sialic acid residue.
[0169] Alternatively, the polysaccharide compound is associated in a non-covalent manner with a therapeutic protein. For example, the polysaccharide compound and the pharmaceutically active compound are in one aspect linked via hydrophobic interactions. Other non-covalent associations include electrostatic interactions, with oppositely charged ions attracting each other.
[0170] In various embodiments, the therapeutic protein is linked to or associated with the polysaccharide compound in stoichiometric amounts (e.g., 1:1, 1:2, 1:3, 1:4, 1:5, 1:6, 1:7, 1:7, 1:8, 1:9, or 1:10, etc.). In various embodiments, 1-6, 7-12 or 13-20 polysaccharides are linked to the blood coagulation protein. In still other embodiments, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or more polysaccharides are linked to the blood coagulation protein.
[0171] In various embodiments, the therapeutic protein is modified to introduce glycosylation sites (i.e., sites other than the native glycosylation sites). Such modification may be accomplished using standard molecular biological techniques known in the art. Moreover, the therapeutic protein, prior to conjugation to a water soluble polymer via one or more carbohydrate moieties, may be glycosylated in vivo or in vitro. These glycosylated sites can serve as targets for conjugation of the proteins with water soluble polymers (US Patent Application No. 20090028822, US Patent Application No. 2009/0093399, US Patent Application No. 2009/0081188, US Patent Application No. 2007/0254836, US Patent Application No. 2006/0111279, and DeFrees S. et al., Glycobiology, 2006, 16, 9, 833-43). For example, a protein that is not naturally glycoslyated in vivo (e.g., a protein that is not a glycoprotein) may be modified as described above.
E. Aminooxy Linkage
[0172] In one embodiment of the invention, the reaction of hydroxylamine or hydroxylamine derivatives with aldehydes (e.g., on a carbohydrate moiety following oxidation by sodium periodate) to form an oxime group is applied to the preparation of conjugates of blood coagulation protein. For example, a glycoprotein (e.g., a therapeutic protein according to the present invention) is first oxidized with a oxidizing agent such as sodium periodate (NaIO4) (Rothfus J A et Smith E L., J Biol Chem 1963, 238, 1402-10; and Van Lenten L and Ashwell G., J Biol Chem 1971, 246, 1889-94). The periodate oxidation of glycoproteins is based on the classical Malaprade reaction described in 1928, the oxidation of vicinal diols with periodate to form an active aldehyde group (Malaprade L., Analytical application, Bull Soc Chim France, 1928, 43, 683-96). Additional examples for such an oxidizing agent are lead tetraacetate (Pb(OAc)4), manganese acetate (MnO(Ac)3), cobalt acetate (Co(OAc)2), thallium acetate (TlOAc), cerium sulfate (Ce(SO4)2) (U.S. Pat. No. 4,367,309) or potassium perruthenate (KRuO4) (Marko et al., J Am Chem Soc 1997, 119, 12661-2). By "oxidizing agent" a mild oxidizing compound which is capable of oxidizing vicinal diols in carbohydrates, thereby generating active aldehyde groups under physiological reaction conditions is meant.
[0173] The second step is the coupling of the polymer containing an aminooxy group to the oxidized carbohydrate moiety to form an oxime linkage. In one embodiment of the invention, this step can be carried out in the presence of catalytic amounts of the nucleophilic catalyst aniline or aniline derivatives (Dirksen A et Dawson P E, Bioconjugate Chem. 2008; Zeng Y et al., Nature Methods 2009; 6:207-9). The aniline catalysis dramatically accelerates the oxime ligation allowing the use of very low concentrations of the reagents. In another embodiment of the invention the oxime linkage is stabilized by reduction with NaCNBH3 to form an alkoxyamine linkage (FIG. 2). Additional catalysts are described below.
[0174] Additional information on aminooxy technology can be found in the following references, each of which is incorporated in their entireties: EP 1681303A1 (HASylated erythropoietin); WO 2005/014024 (conjugates of a polymer and a protein linked by an oxime linking group); WO96/40662 (aminooxy-containing linker compounds and their application in conjugates); WO 2008/025856 (Modified proteins); Peri F et al., Tetrahedron 1998, 54, 12269-78; Kubler-Kielb J et. Pozsgay V., J Org Chem 2005, 70, 6887-90; Lees A et al., Vaccine 2006, 24(6), 716-29; and Heredia K L et al., Macromoecules 2007, 40(14), 4772-9.
[0175] In various embodiments of the invention, the water soluble polymer which is linked according to the aminooxy technology described herein to an oxidized carbohydrate moiety of a therapeutic protein (e.g., FVIII, FVIIa, or FIX) include, but are not limited to polyethylene glycol (PEG), branched PEG, polysialic acid (PSA), carbohydrate, polysaccharides, pullulane, chitosan, hyaluronic acid, chondroitin sulfate, dermatan sulfate, starch, dextran, carboxymethyl-dextran, polyalkylene oxide (PAO), polyalkylene glycol (PAG), polypropylene glycol (PPG) polyoxazoline, poly acryloylmorpholine, polyvinyl alcohol (PVA), polycarboxylate, polyvinylpyrrolidone, polyphosphazene, polyoxazoline, polyethylene-co-maleic acid anhydride, polystyrene-co-maleic acid anhydride, poly(l-hydroxymethylethylene hydroxymethylformal) (PHF), 2-methacryloyloxy-2'-ethyltrimethylammoniumphosphate (MPC).
Nucleophilic Catalysts
[0176] As described herein, the conjugation of water soluble polymers to therapeutic proteins can be catalyzed by aniline. Aniline strongly catalyzes aqueous reactions of aldehydes and ketones with amines to form stable imines such as hydrazones and oximes. The following diagram compares an uncatalyzed versus the aniline-catalyzed oxime ligation reaction (Kohler J J, ChemBioChem 2009; 10:2147-50):
##STR00014##
[0177] However, considering the numerous health risks associated with aniline, alternative catalysts are desirable. The present invention provides aniline derivatives as alternative oxime ligation catalysts. Such aniline derivatives include, but are not limited to, o-amino benzoic acid, m-amino benzoic acid, p-amino benzoic acid, sulfanilic acid, o-aminobenzamide, o-toluidine, m-toluidine, p-toluidine, o-anisidine, m-anisidine, and p-anisidine.
[0178] In one embodiment of the invention, m-toluidine (aka meta-toluidine, m-methylaniline, 3-methylaniline, or 3-amino-1-methylbenzene) is used to catalyze the conjugation reactions described herein. M-toluidine and aniline have similar physical properties and essentially the same pKa value (m-toluidine: pKa 4.73, aniline: pKa 4.63).
[0179] The nucleophilic catalysts of the invention are useful for oxime ligation (e.g, using aminooxy linkage) or hydrazone formation (e.g., using hydrazide chemistry). In various embodiments of the invention, the nucleophilic catalyst is provided in the conjugation reaction at a concentration of of 0.1, 0.2, 0.3, 0.5, 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, 9.5, 10.0, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, or 50 mM. In one embodiment, the nucleophilic catalyst is provided between 1 to 10 mM. In various embodiments of the invention, the pH range of conjugation reaction is 4.5, 5.0, 5.5, 6.0, 6.5, 7.0 and 7.5. In one embodiment, the pH is between 5.5 to 6.5.
Purification of Conjugated Proteins
[0180] In various embodiments, purification of a protein that has been incubated with an oxidizing agent and/or a therapeutic protein that has been conjugated with a water soluble polymer according to the present disclosure, is desired. Numerous purification techniques are known in the art and include, without limitation, chromatographic methods such as ion-exchange chromatography, hydrophobic interaction chromatography, size exclusion chromatography and affinity chromatography or combinations thereof, filtration methods, and precipitation methods (Guide to Protein Purification, Meth. Enzymology Vol 463 (edited by Burgess R R and Deutscher M P), 2.sup.nd edition, Academic Press 2009).
[0181] The following examples are not intended to be limiting but only exemplary of specific embodiments of the invention.
EXAMPLES
Example 1
Preparation of the Homobifunctional Linker NH.sub.2[OCH.sub.2CH.sub.2].sub.2ONH.sub.2
[0182] The homobifunctional linker NH.sub.2[OCH.sub.2CH.sub.2].sub.2ONH.sub.2
##STR00015##
[0183] (3-oxa-pentane-1,5-dioxyamine) containing two active aminooxy groups was synthesized according to Boturyn et al. (Tetrahedron 1997; 53:5485-92) in a two step organic reaction employing a modified Gabriel-Synthesis of primary amines (FIG. 3). In the first step, one molecule of 2,2-chlorodiethylether was reacted with two molecules of Endo-N-hydroxy-5-norbornene-2,3-dicarboximide in dimethylformamide (DMF). The desired homobifunctional product was prepared from the resulting intermediate by hydrazinolysis in ethanol.
Example 2
Preparation of the Homobifunctional Linker NH.sub.2[OCH.sub.2CH.sub.2].sub.4ONH.sub.2
[0184] The Homobifunctional Linker NH.sub.2[OCH.sub.2CH.sub.2].sub.4ONH.sub.2
##STR00016##
[0185] (3,6,9-trioxa-undecane-1,11-dioxyamine) containing two active aminooxy groups was synthesized according to Boturyn et al. (Tetrahedron 1997; 53:5485-92) in a two step organic reaction employing a modified Gabriel-Synthesis of primary amines (FIG. 3). In the first step one molecule of Bis-(2-(2-chlorethoxy)-ethyl)-ether was reacted with two molecules of Endo-N-hydroxy-5-norbornene-2,3-dicarboximide in DMF. The desired homobifunctional product was prepared from the resulting intermediate by hydrazinolysis in ethanol.
Example 3
Preparation of the Homobifunctional Linker NH.sub.2[OCH.sub.2CH.sub.2].sub.6ONH.sub.2
[0186] The Homobifunctional Linker NH.sub.2[OCH.sub.2CH.sub.2].sub.6ONH.sub.2
##STR00017##
[0187] (3,6,9,12,15-penatoxa-heptadecane-1,17-dioxyamine) containing two active aminooxy groups was synthesized according to Boturyn et al. (Tetrahedron 1997; 53:5485-92) in a two step organic reaction employing a modified Gabriel-Synthesis of primary amines. In the first step one molecule of hexaethylene glycol dichloride was reacted with two molecules of Endo-N-hydroxy-5-norbornene-2,3-dicarboximide in DMF. The desired homobifunctional product was prepared from the resulting intermediate by hydrazinolysis in ethanol.
Example 4
Detailed Synthesis of the Aminooxy-PSA Reagent
[0188] 3-oxa-pentane-1,5 dioxyamine was synthesized according to Botyryn et al (Tetrahedron 1997; 53:5485-92) in a two step organic synthesis as outlined in Example 1.
Step 1:
[0189] To a solution of Endo-N-hydroxy-5-norbonene-2,3-dicarboxiimide (59.0 g; 1.00 eq) in 700 ml anhydrous N,N-dimetylformamide anhydrous K.sub.2CO.sub.3 (45.51 g; 1.00 eq) and 2,2-dichlorodiethylether (15.84 ml; 0.41 eq) were added. The reaction mixture was stirred for 22 h at 50.degree. C. The mixture was evaporated to dryness under reduced pressure. The residue was suspended in 2 L dichloromethane and extracted two times with saturated aqueous NaCl-solution (each 1 L). The Dichloromethane layer was dried over Na.sub.2SO.sub.4 and then evaporated to dryness under reduced pressure and dried in high vacuum to give 64.5 g of 3-oxapentane-1,5-dioxy-endo-2',3'-dicarboxydiimidenorbornene as a white-yellow solid (intermediate 1).
Step 2:
[0190] To a solution of intermediate 1 (64.25 g; 1.00 eq) in 800 ml anhydrous Ethanol, 31.0 ml Hydrazine hydrate (4.26 eq) were added. The reaction mixture was then refluxed for 2 hrs. The mixture was concentrated to the half of the starting volume by evaporating the solvent under reduced pressure. The occurring precipitate was filtered off. The remaining ethanol layer was evaporated to dryness under reduced pressure. The residue containing the crude product 3-oxa-pentane-1,5-dioxyamine was dried in vacuum to yield 46.3 g. The crude product was further purified by column chromatography (Silicagel 60; isocratic elution with Dichloromethane/Methanol mixture, 9/1) to yield 11.7 g of the pure final product 3-oxa-pentane-1,5-dioxyamine.
Example 5
Preparation of Aminooxy-PSA
[0191] 1000 mg of oxidized PSA (MW=20 kD) obtained from the Serum Institute of India (Pune, India) was dissolved in 16 ml 50 mM phospate buffer pH 6.0. Then 170 mg 3-oxa-pentane-1,5-dioxyamine was given to the reaction mixture. After shaking for 2 hrs at RT 78.5 mg sodium cyanoborohydride was added and the reaction was performed for 18 hours over night. The reaction mixture was then subjected to a ultrafiltration/diafiltration procedure (UF/DF) using a membrane with a 5 kD cut-off made of regenerated cellulose (50 cm.sup.2, Millipore).
Example 6
Preparation of Aminooxy-PSA Employing a Chromatographic Purification Step
[0192] 1290 mg of oxidized PSA (MW=20 kD) obtained from the Serum Institute of India (Pune, India) was dissolved in 25 ml 50 mM phosphate buffer pH 6.0 (Buffer A). Then 209 mg 3-oxa-pentane-1,5-dioxyamine was given to the reaction mixture. After shaking for 1 h at RT 101 mg sodium cyanoborohydride was added and the reaction was performed for 3 hours. Then the mixture was then subjected to a weak anion exchange chromatography step employing a Fractogel EMD DEAE 650-M chromatography gel (column dimension: XK26/135). The reaction mixture was diluted with 110 ml Buffer A and loaded onto the DEAE column pre-equilibrated with Buffer A at a flow rate of 1 cm/min. Then the column was washed with 20 CV Buffer B (20 mM Hepes, pH 6.0) to remove free 3-oxa-pentane-1,5-dioxyamine and cyanide at a flow rate of 2 cm/min. The aminooxy-PSA reagent was then eluted with a step gradient consisting of 67% Buffer B and 43% Buffer C (20 mM Hepes, 1M NaCl, pH 7.5). The eluate was concentrated by UF/DF using a 5 kD membrane made of polyether sulfone (50 cm.sup.2, Millipore). The final diafiltration step was performed against Buffer D (20 mM Hepes, 90 mM NaCl, pH 7.4). The preparation was analytically characterized by measuring total PSA (Resorcinol assay) and total aminooxy groups (TNBS assay) to determine the degree of modification. Furthermore the polydispersity as well as free 3-oxa-pentane-1,5-dioxyamine and cyanide was determined.
Example 7
Preparation of Aminooxy-PSA without a Reduction Step
[0193] 573 mg of oxidized PSA (MW=20 kD) obtained from the Serum Institute of India (Pune, India) was dissolved in 11.3 ml 50 mM phosphate buffer pH 6.0 (Buffer A). Then 94 mg 3-oxa-pentane-1,5-dioxyamine was given to the reaction mixture. After shaking for 5 h at RT the mixture was then subjected to a weak anion exchange chromatography step employing a Fractogel EMD DEAE 650-M chromatography gel (column dimension: XK16/105). The reaction mixture was diluted with 50 ml Buffer A and loaded onto the DEAE column pre-equilibrated with Buffer A at a flow rate of 1 cm/min. Then the column was washed with 20 CV Buffer B (20 mM Hepes, pH 6.0) to remove free 3-oxa-pentane-1,5-dioxyamine and cyanide at a flow rate of 2 cm/min. The aminooxy-PSA reagent was the eluted with a step gradient consisting of 67% Buffer B and 43% Buffer C (20 mM Hepes, 1 M NaCl, pH 7.5). The eluate was concentrated by UF/DF using a 5 kD membrane made of polyether sulfone (50 cm.sup.2, Millipore). The final diafiltration step was performed against Buffer D (20 mM Hepes, 90 mM NaCl, pH 7.4). The preparation was analytically characterized by measuring total PSA (Resorcinol assay) and total aminooxy groups (TNBS assay) to determine the degree of modification. Furthermore the polydispersity as well as free 3-oxa-pentane-1,5-dioxyamine was determined.
Example 8
Preparation of Aminooxy-PSA without a Reduction Step in the Presence of the Nucleophilic Catalyst m-Toluidine
[0194] 573 mg of oxidized PSA (MW=20 kD) obtained from the Serum Institute of India (Pune, India) is dissolved in 9 ml 50 mM phosphate buffer pH 6.0 (Buffer A). Then 94 mg 3-oxa-pentane-1,5-dioxyamine is given to this solution. Subsequently 2.3 ml of a 50 mM m-toluidine stock solution are added to this reaction mixture. After shaking for 2 h at RT the mixture is then subjected to a weak anion exchange chromatography step employing a Fractogel EMD DEAE 650-M chromatography gel (column dimension: XK16/105). The reaction mixture is diluted with 50 ml Buffer A and loaded onto the DEAE column pre-equilibrated with Buffer A at a flow rate of 1 cm/min. Then the column is washed with 20CV Buffer B (20 mM Hepes, pH 6.0) to remove free 3-oxa-pentane-1,5-dioxyamine and cyanide at a flow rate of 2 cm/min. The aminooxy-PSA reagent is the eluted with a step gradient consisting of 67% Buffer B and 43% Buffer C (20 mM Hepes, 1 M NaCl, pH 7.5). The eluate is concentrated by UF/DF using a 5 kD membrane made of polyether sulfone (50 cm.sup.2, Millipore). The final diafiltration step is performed against Buffer D (20 mM Hepes, 90 mM NaCl, pH 7.4). The preparation is analytically characterized by measuring total PSA (Resorcinol assay) and total aminooxy groups (TNBS assay) to determine the degree of modification. Furthermore the polydispersity as well as free 3-oxa-pentane-1,5-dioxyamine is determined.
Example 9
Preparation of Aminooxy-PSA Reagent
[0195] An Aminooxy-PSA reagent was prepared according to the Examples 4-8. After diafiltration, the product was frozen at -80.degree. C. and lyophilized. After lyophilization the reagent was dissolved in the appropriate volume of water and used for preparation of PSA-protein conjugates via carbohydrate modification.
Example 10
Evaluation of the Efficacy of Different Alternative Nucleophilic Catalysts
[0196] rFIX was incubated with sodium periodate, aminooxy-PSA reagent under standardized conditions (1 mg/ml rFIX in 20 mM L-histidine, 150 mM NaCl, 5 mM CaCl.sub.2), pH 6.0, 5-fold molar aminooxy-PSA reagent excess, 100 .mu.M NaIO.sub.4) using different nucleophilic catalysts (aniline, m-toluidine, o-anisidine, m-anisidine, o-aminobenzoic acid, m-aminobenzoic acid, p-aminobenzoic acid, p-aminobenzamide, sulfanilic acid/standard concentration: 10 mM) The reaction was carried out for 2 hrs in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of aqueous cysteine solution with a final concentration of 1 mM.
[0197] The coupling efficiency was determined by SDS-PAGE using an Invitrogen X-cell mini system. Samples were spiked with lithium dodecyl sulfate (LDS) buffer and denatured for 10 min at 70.degree. C. Then the samples were applied on 3-8% TRIS-acetate gels and ran at 150 V for 60 min. Subsequently the gels were stained with Coomassie.
[0198] In addition the samples were characterized by use of a SEC-HPLC system using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77).
[0199] 50 .mu.l of samples were injected undiluted and eluted isocratically with a 0.22 .mu.m filtered solution of 20 mM NaH2PO4, 50 mM Na2SO4, pH 6.1 at a flow rate of 0.5 ml/min. The elution pattern was recorded at 280 nm.
[0200] The results are summarized in FIGS. 5A-C and 6 (SDS PAGE) and Table 2 (SEC-HPLC results). The catalytic effect of the different preparations is demonstrated. It is shown that the use of m-toluidine leads to equivalent results as obtained with aniline.
TABLE-US-00004 TABLE 2 di-PSAylated mono- free nucleophilic catalysts rFIX PSAylated rFIX rFIX no catalyst 4.5% 24.9% 70.6% 10 mM aniline 47.7% 33.6% 18.7% 10 mM m-toluidine 31.4% 40.8% 27.8% 10 mM o-aminobenzioc acid 30.9% 38.5% 30.6% 10 mM m-aminobenzioc acid 27.6% 38.0% 34.4% 10 mM p-aminobenzioc acid 18.1% 39.3% 42.6% 10 mM o-aminobenzamide 15.9% 38.4% 45.7% 10 mM sulfanilic acid 11.8% 35.8% 52.4%
Example 11
Polysialylation of rFIX Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0201] 12.3 mg rFIX was dissolved in 6.1 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). 254 .mu.l of an aqueous sodium periodate solution (5 mM) was then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 6.5 .mu.l of a 1 M aqueous cysteine solution. The mixture was subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0202] The retentate (8.8 ml), containing oxidized rFIX was mixed with 2.46 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) was added to give a 5-fold molar reagent excess. This mixture was incubated for 2.5 h at RT in the dark under gentle stirring.
[0203] The free rFIX was removed by means of anion exchange chromatography (AEC). The reaction mixture was diluted with 15 ml Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. The column was then eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFIX elutes at a conductivity between 12-25 mS/cm and the conjugate between 27-45 mS/cm. The conductivity of the conjugate containing fractions was subsequently raised to 190 mS/cm with Buffer C (50 mM Hepes, 5M NaCl, 5 mM CaCl2, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free aminooxy-PSA reagent was washed out within 5 CV Buffer D. Subsequently the conjugate is eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions were concentrated by UF/DF using Vivaspin 15R 10 kD centrifugal filtrator. The final diafiltration step was performed against histidine buffer, pH 7.2 containing 150 mM NaCl and 5 mM CaCl2. The preparation was analytically characterized by measuring total protein (Bradford) and FIX chromogenic activity. The PSA-rFIX conjugate showed a specific activity of >50% in comparison to native rFIX is determined.
Method 2:
[0204] 12.3 mg rFIX is dissolved in in L-histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2) to get a final protein concentration of 1 mg rFIX/ml. A 5 mM aqueous sodium periodate solution is added to get a final concentration of 100 .mu.M and the reaction mixture is incubated for 1 hour in the dark at 4.degree. C. under gentle stirring at pH 6.0 and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution (or other quenching reagents) to get a final concentration of 10 mM. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0205] The obtained retentate (8.8 ml), containing oxidized rFIX, is mixed with an aqueous m-toluidine solution (50 mM) to give a final concentration of 10 mM and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture was incubated at pH 6.0 for 2.5 hours at room temperature; 0.5 hours to 18 hours at +4.degree. C.) in the dark under gentle stirring.
[0206] The free rFIX is removed by means of anion exchange chromatography (AEC). The reaction mixture is diluted with appropriate amounts of Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) to correct the solutions conductivity and pH prior to load onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFIX is eluted by a step gradient using 25% of Buffer B, which results in a conductivity between 12-25 mS/cm in the obtained fraction and the conjugate using a step gradient of 50% Buffer B, which results in a conductivity between between 27-45 mS/cm in the conjugate fraction. The conductivity of the conjugate containing fraction is subsequently raised to 190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9 or by use of anti-chaotropic salts e.g. ammonium sulphate, ammonium acetate etc.) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn. or comparable HIC media) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free aminooxy-PSA reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against L-histidine buffer, pH 7.2 containing 150 mM NaCl and 5 mM CaCl2. The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and FIX chromogenic- and clotting activity. For the PSA-rFIX conjugate a specific activity of >50% in comparison to native rFIX is determined.
Method 3:
[0207] 25.4 mg rFIX was dissolved in 18.7 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). 531 .mu.l of an aqueous sodium periodate solution (5 mM) and 5.07 ml of an aqueous m-toluidine solution (50 mM) were then added. Subsequently, the aminooxy-PSA reagent with a MW of 20 kD (described above) was added to give a 5-fold molar reagent excess. The mixture was incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 25 .mu.l of 1 M aqueous cysteine solution.
[0208] The free rFIX was removed by means of anion exchange chromatography (AEC). The reaction mixture was diluted with 20 ml Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column was eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFIX eluted at a conductivity between 12-25 mS/cm and the conjugate between 27-45 mS/cm. The conductivity of the conjugate containing fractions was subsequently raised to 190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free aminooxy-PSA reagent was washed out within 5 CV Buffer D. Subsequently, the conjugate was eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions were concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step was performed against histidine buffer, pH 7.2 containing 150 mM NaCl and 5 mM CaCl2. The preparation was analytically characterized by measuring total protein (Bradford) and FIX chromogenic activity. For the PSA-rFIX conjugate a specific activity of >50% in comparison to native rFIX was determined. The conjugate was additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It was shown that the preparation contains no free FIX. The conjugate consisted of 57% mono-polysialylated and 31% di-polysialylated and 12% tri-polysialyated product.
Method 4:
[0209] 25.4 mg rFIX was dissolved in L-histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2) to get a final protein concentration of 2 mg rFIX/ml. Subsequently an 5 mM aqueous sodium periodate solution was added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PSA reagent with a MW of 20 kD (described above) was added to give a 5-fold molar reagent excess. After correction of the pH to 6.0 the mixture was incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0210] The free rFIX was removed by means of ion exchange chromatography (IEC). The reaction mixture was diluted with appropriate amounts of Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) to correct the solutions conductivity and pH value prior to load onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column was eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFIX was eluted by a step gradient using 25% of Buffer B, which results in a conductivity between 12-25 mS/cm in the obtained fraction and the conjugate using a step gradient of 50% Buffer B, which results in a conductivity between 27-45 mS/cm in the conjugate fraction. The conductivity of the conjugate containing fraction was subsequently raised to 190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9; by use of anti-chaotropic salts e.g. ammonium acetate) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.; or comparable HIC media) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free aminooxy-PSA reagent was washed out within 5 CV Buffer D. Subsequently the conjugate was eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions were concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step was performed against L-histidine buffer, pH 7.2 containing 150 mM NaCl and 5 mM CaCl2. The preparation was analytically characterized by measuring total protein (Bradford and BCA procedure) and FIX chromogenic- and clotting activity. For the PSA-rFIX conjugate a specific activity of >50% in comparison to native rFIX was determined. The conjugate was additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It was shown that the preparation contains no free FIX. The conjugate consisted of 57% mono-polysialylated and 31% di-polysialylated and 12% tri-polysialyated product.
Example 12
Polysialylation of rFVIII Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0211] 50 mg rFVIII was transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 was added to give a final concentration of 200 .mu.M. The oxidation was carried at RT for 30 min in the dark under gentle shaking. Then the reaction was quenched with cysteine (final concentration: 10 mM) for 60 min at RT. The solution was subjected to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which was equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl.sub.2), pH 7.0). The column was equilibrated with 5 CV Buffer A. Then the oxidized rFVIII was eluted with Buffer B (20 mM Hepes, 5 mM CaCl.sub.2), 1M NaCl, pH 7.0). The rFVIII containing fractions were collected. The protein content was determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl. Then a 50-fold molar excess of a aminooxy-PSA reagent with a MW of 20 kD (described above) was added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction was performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy-PSA reagent was removed by means of HIC. The conductivity of the reaction mixture was raised to 130 mS/cm by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate was eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl.sub.2). Finally, the PSA-rFVIII containing fractions were collected and subjected to UF/DF by use of a 30 kD membrane made of regenerated cellulose (88 cm.sup.2, Millipore). The preparation was analytically characterized by measuring total protein (Coomassie, Bradford) and FVIII chromogenic activity. The PSA-rFVIII conjugate showed a specific activity of >70% in comparison to native rFVIII was determined.
Method 2:
[0212] 58 mg of recombinant factor VIII (rFVIII) derived from the ADVATE process in Hepes buffer (50 mM HEPES, .about.350 mM sodium chloride, 5 mM calcium chloride, 0.1% Polysorbate 80, pH 7.4) is dissolved in reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0213] The oxidized rFVIII is further purified by anion exchange chromatography on EMD TMAE (M) (Merck). The mixture is diluted with Buffer A (20 mM Hepes, 5 mM CaCl.sub.2), pH 6.5) to give a conductivity of 5 ms/cm. This solution is loaded onto the IEX column (bed height: 5.4 cm) with a column volume of 10 ml using a flow rate of 1.5 cm/min. This column is subsequently washed (flow rate: 1.5 cm/min) with 5 CV of a 92:8 mixture (w/w) of Buffer A and Buffer B (20 mM Hepes, 5 mM CaCl.sub.2), 1.0 M NaCl, pH 7.0). Then the oxidized rFVIII is eluted with a 50:50 (w/w) mixture of Buffer A and Buffer B followed by a postelution step with 5 CV of Buffer B. The elution steps are carried out by use of a flow rate of 1.0 cm/min.
[0214] Subsequently, the aminooxy-polysialic acid (PSA-ONH.sub.2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized rFVIII within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0215] The obtained PSA-rFVIII conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0216] The reaction mixture is spiked with ammonium acetate by addition of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture are mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of a 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column at flow rate of 1 cm/min followed by a washing step using >3 CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0217] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified PSA-rFVIII conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PSA-rFVIII conjugate is monitored at UV 280 nm and the eluate containing the conjugate is collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIII from the main product.
[0218] Finally the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with a molecular weight cut off 30 kD (88 cm.sup.2, Millipore).
[0219] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein, FVIII chromogenic activity and determination of the polysialyation degree by measuring the PSA content (resorcinol assay). For the conjugate obtained a specific activity >50% and a PSA degree >5.0 is calculated.
Method 3:
[0220] 50 mg rFVIII was transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) was added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction was performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction was quenched with cysteine for 60 min at RT (final concentration: 10 mM). Then the conductivity of the reaction mixture was raised to 130 mS/cm by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently, the conjugate was eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-rFVIII containing fractions were collected and subjected to UF/DF by use of a 30 kD membrane made of regenerated cellulose (88 cm.sup.2, Millipore). The preparation was analytically characterized by measuring total protein (Bradford) and FVIII chromogenic activity. For the PSA-rFVIII conjugate a specific activity of >70% in comparison to native rFVIII was determined.
Method 4:
[0221] 50 mg recombinant factor VIII (rFVIII) derived from the ADVATE process in 50 mM Hepes buffer (50 mM HEPES, .about.350 mM sodium chloride, 5 mM calcium chloride, 0.1% Polysorbate 80, pH 7.4) was dissolved in reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution was corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0222] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent was added in a 50-fold molar excess to this rFVIII solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) was added within 15 minutes to get a final concentration of 10 mM. Finally, a 40 mM aqueous sodium periodate solution was added to give a concentration of 400 .mu.M.
[0223] The reaction mixture was incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction was stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0224] The obtained PSA-rFVIII conjugate was purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0225] The reaction mixture was spiked with ammonium acetate by addition of of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture was mixed with 1 volume of the ammonium acetate containing buffer system and the pH value was corrected to pH 6.9 by drop wise addition of an 0.5 N aqueous NaOH solution. This mixture was loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0226] For removal of reaction by-products and anti-chaotropic salt a second washing step was performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFVIII conjugate was performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PSA-rFVIII conjugate was monitored at UV 280 nm and the eluate containing the conjugate was collected within <4 CV. The post elution step was performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIII from the main product.
[0227] Finally, the purified conjugate was concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with a molecular weight cut off 30 kD (88 cm2, Millipore).
[0228] The conjugates prepared by use of this procedure were analytically characterized by measuring total protein, FVIII chromogenic activity and determination of the polysialyation degree by measuring the PSA content (resorcinol assay). [0229] Analytical data (mean of 6 consecutive batches): [0230] Process yield (Bradford): 58.9% [0231] Process yield (FVIII chrom.): 46.4% [0232] Specific activity: (FVIII chrom./mg protein): 4148 IU/mg [0233] Specific activity (% of starting material): 79.9% [0234] PSA degree (mol/mol): 8.1
Example 13
PEGylation of r FVIII Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0235] rFVIII is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 14.7 mg rFVIII is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). Then 296 .mu.l of an aqueous sodium periodate solution (5 mM) is added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture was subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0236] The retentate (10.9 ml), containing oxidized rFVIII, is mixed with 2.94 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PEG reagent with a MW of 20 kD is added to give a 5-fold molar reagent excess. This mixture was incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0237] Finally, the PEG-rFVIII conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a 30 kD membrane (50 cm2, Millipore). The preparation is analytically characterized by measuring total protein (Coomassie, Bradford) and FVIII chromogenic activity. It is expected that the PEG-rFVIII conjugate will demonstrate a specific activity of >70% in comparison to native rFVIII was determined.
Method 2:
[0238] rFVIII is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). A starting weight or concentration of rFVIII is dissolved in or transferred to a reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0239] The oxidized rFVIII is further purified by anion exchange chromatography on EMD TMAE (M) (Merck). The mixture is diluted with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 6.5) to give a conductivity of 5 ms/cm. This solution is loaded onto the IEX column (bed height: 5.4 cm) with a column volume of 10 ml using a flow rate of 1.5 cm/min. This column is subsequently washed (flow rate: 1.5 cm/min) with 5 CV of a 92:8 mixture (w/w) of Buffer A and Buffer B (20 mM Hepes, 5 mM CaCl2, 1.0 M NaCl, pH 7.0). Then the oxidized rFVIII is eluted with a 50:50 (w/w) mixture of Buffer A and Buffer B followed by a postelution step with 5 CV of Buffer B. The elution steps are carried out by use of a flow rate of 1.0 cm/min.
[0240] Subsequently, the aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized rFVIII within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0241] The obtained PEG-rFVIII conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0242] The reaction mixture is spiked with ammonium acetate by addition of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture are mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of a 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3 CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0243] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFVIII conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PEG-rFVIII conjugate is monitored at UV 280 nm and the eluate containing the conjugate is collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIII from the main product.
[0244] Finally, the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with a molecular weight cut off 30 kD (Millipore).
[0245] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0246] rFVIII is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 7.84 mg rFVIII, dissolved in 6 ml Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) are mixed with 314 .mu.l of an aqueous sodium periodate solution (10 mM), and 1.57 ml of an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0247] Finally the PEG-rFVIII conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl.sub.2). The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl.sub.2) and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a 30 kD membrane (88 cm.sup.2, Millipore). The analytical characterization of the conjugate by FVIII chromogenic assay and determination of total protein (Bradford) shows a specific activity of >60% compared to the rFVIII starting material.
Method 4:
[0248] rFVIII is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of rFVIII is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg rFVIII/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0249] The free rFVIII is removed by means of ion exchange chromatography (IEC). The reaction mixture was diluted with appropriate amounts of Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) to correct the solutions conductivity and pH value prior to load onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column was eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFVIII was eluted by a step gradient using 25% of Buffer B, which results in a conductivity between 12-25 mS/cm in the obtained fraction and the conjugate using a step gradient of 50% Buffer B, which results in a conductivity between 27-45 mS/cm in the conjugate fraction. The conductivity of the conjugate containing fraction is subsequently raised with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9; by use of anti-chaotropic salts e.g. ammonium acetate, ammonium sulphate etc.) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.; or comparable HIC media) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free PEG-reagent was washed out within 5 CV Buffer D. Subsequently, the conjugate was eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0250] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 14
Polysialylation of rFVIIa Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0251] A starting concentration or weight of recombinant factor VIIa (rFVIIa) is transferred or dissolved in reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous NaOH solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 50 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0252] The oxidized rFVIIa is further purified by anion exchange chromatography on EMD TMAE (M) (Merck). The mixture is diluted with Buffer A (20 mM Hepes, 5 mM CaCl.sub.2), pH 6.5) to give a conductivity of 5 ms/cm. This solution is loaded onto the IEX column (bed height: 5.4 cm) with a column volume of 10 ml using a flow rate of 1.5 cm/min. This column is subsequently washed (flow rate: 1.5 cm/min) with 5 CV of a 92:8 mixture (w/w) of Buffer A and Buffer B (20 mM Hepes, 5 mM CaCl.sub.2), 1.0 M NaCl, pH 7.0). Then the oxidized rFVIIa is eluted with a 50:50 (w/w) mixture of Buffer A and Buffer B followed by a postelution step with 5 CV of Buffer B. The elution steps are carried out by use of a flow rate of 1.0 cm/min.
[0253] Subsequently, the aminooxy-polysialic acid (PSA-ONH.sub.2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized rFVIIa within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0254] The obtained PSA-rFVIIa conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0255] The reaction mixture is spiked with ammonium acetate by addition of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture are mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of a 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3 CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0256] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFVIIa conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PSA-rFVIIa conjugate is monitored at UV 280 nm and the eluate containing the conjugate is collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIIa from the main product.
[0257] Finally, the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (e.g. 10 kD MWCO, 88 cm.sup.2, Millipore).
[0258] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 2:
[0259] A starting weight or concentration of rFVIIa is dissolved in or transferred to a reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous NaOH solution.
[0260] Subsequently, the aminooxy-polysialic acid (PSA-ONH.sub.2) reagent is added in a 50-fold molar excess to this rFVIIa solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 150 .mu.M.
[0261] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0262] The obtained PSA-rFVIIa conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0263] The reaction mixture is spiked with ammonium acetate by addition of of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture is mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of an 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0264] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFVIIa conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PSA-rFVIIa conjugate is monitored at UV 280 nm and the eluate containing the conjugate was collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIII from the main product.
[0265] Finally, the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0266] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 15
PEGylation of rFIX Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0267] rFIX is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). A starting weight or concentration of rFIX is dissolved in or transferred to a reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0268] The oxidized rFVIII is further purified by anion exchange chromatography on EMD TMAE (M) (Merck). The mixture is diluted with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 6.5) to give a conductivity of 5 mS/cm. This solution is loaded onto the IEX column (bed height: 5.4 cm) with a column volume of 10 ml using a flow rate of 1.5 cm/min. This column is subsequently washed (flow rate: 1.5 cm/min) with 5 CV of a 92:8 mixture (w/w) of Buffer A and Buffer B (20 mM Hepes, 5 mM CaCl2, 1.0 M NaCl, pH 7.0). Then the oxidized rFIX is eluted with a 50:50 (w/w) mixture of Buffer A and Buffer B followed by a postelution step with 5 CV of Buffer B. The elution steps are carried out by use of a flow rate of 1.0 cm/min.
[0269] Subsequently, the aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized rFIX within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0270] The obtained PEG-rFIX conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0271] The reaction mixture is spiked with ammonium acetate by addition of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture are mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of a 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3 CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0272] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFIX conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PEG-rFIX conjugate is monitored at UV 280 nm and the eluate containing the conjugate is collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFIX from the main product.
[0273] Finally, the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with a molecular weight cut off 10 kD (88 cm2, Millipore).
[0274] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 2:
[0275] rFIX is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of rFIX is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg rFIX/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0276] The free rFIX is removed by means of ion exchange chromatography (IEC). The reaction mixture was diluted with appropriate amounts of Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) to correct the solutions conductivity and pH value prior to load onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column was eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFIX was eluted by a step gradient using 25% of Buffer B, which results in a conductivity between 12-25 mS/cm in the obtained fraction and the conjugate using a step gradient of 50% Buffer B, which results in a conductivity between 27-45 mS/cm in the conjugate fraction. The conductivity of the conjugate containing fraction is subsequently raised with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9; by use of anti-chaotropic salts e.g. ammonium acetate, etc) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.; or comparable HIC media) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free aminooxy-PEG reagent was washed out within 5 CV Buffer D. Subsequently, the conjugate was eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0277] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 16
PEGylation of rFVIIa Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0278] rFVIIa is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). A starting weight or concentration of rFVIIa is dissolved in or transferred to a reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous NaOH solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 50 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0279] The oxidized rFVIIa is further purified by anion exchange chromatography on EMD TMAE (M) (Merck). The mixture is diluted with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 6.5) to give a conductivity of 5 mS/cm. This solution is loaded onto the IEX column (bed height: 5.4 cm) with a column volume of 10 ml using a flow rate of 1.5 cm/min. This column is subsequently washed (flow rate: 1.5 cm/min) with 5 CV of a 92:8 mixture (w/w) of Buffer A and Buffer B (20 mM Hepes, 5 mM CaCl2, 1.0 M NaCl, pH 7.0). Then the oxidized rFVIIa is eluted with a 50:50 (w/w) mixture of Buffer A and Buffer B followed by a postelution step with 5 CV of Buffer B. The elution steps are carried out by use of a flow rate of 1.0 cm/min.
[0280] Subsequently, the aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized rFVIIa within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0281] The obtained PEG-rFVIIa conjugate is purified by Hydrophobic Interaction Chromatography (HIC) using a Phenyl Sepharose FF low sub resin (GE Healthcare) packed into a column manufactured by GE Healthcare with a bed height (h) of 15 cm and a resulting column volume (CV) of 81 ml.
[0282] The reaction mixture is spiked with ammonium acetate by addition of 50 mM Hepes buffer, containing 350 mM sodium chloride, 8 M ammonium acetate, 5 mM calcium chloride, pH 6.9. Two volumes of the reaction mixture are mixed with 1 volume of the ammonium acetate containing buffer system and the pH value is corrected to pH 6.9 by drop wise addition of a 0.5 N aqueous NaOH solution. This mixture is loaded onto the HIC column using a flow rate of 1 cm/min followed by a washing step using >3 CV equilibration buffer (50 mM Hepes, 350 mM sodium chloride, 2.5 M ammonium acetate, 5 mM calcium chloride, pH 6.9).
[0283] For removal of reaction by-products and anti-chaotropic salt a second washing step is performed with >5CV washing buffer 1 (50 mM Hepes, 3 M sodium chloride, 5 mM calcium chloride, pH 6.9) in upflow mode at a flow rate of 2 cm/min. Then elution of purified rFVIIa conjugate is performed in down flow mode using a step gradient of 40% washing buffer 2 (50 mM Hepes, 1.5 M sodium chloride, 5 mM calcium chloride, pH 6.9) and 60% elution buffer (20 mM Hepes, 5 mM calcium chloride, pH 7.5) at a flow rate of 1 cm/min. The elution of the PEG-rFVIIa conjugate is monitored at UV 280 nm and the eluate containing the conjugate is collected within <4 CV. The post elution step is performed with >3 CV elution buffer under the same conditions to separate minor and/or non modified rFVIIa from the main product.
[0284] Finally, the purified conjugate is concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with a molecular weight cut off 10 kD (Millipore).
[0285] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 2:
[0286] rFVIIa is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of rFVIIa is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg rFVIIa/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0287] The free rFVIIa is removed by means of ion exchange chromatography (IEC). The reaction mixture was diluted with appropriate amounts of Buffer A (50 mM Hepes, 5 mM CaCl2, pH 7.5) to correct the solutions conductivity and pH value prior to load onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column was eluted with Buffer B (50 mM Hepes, 1 M NaCl, 5 mM CaCl2, pH 7.5). Free rFVIIa was eluted by a step gradient using 25% of Buffer B, which results in a conductivity between 12-25 mS/cm in the obtained fraction and the conjugate using a step gradient of 50% Buffer B, which results in a conductivity between 27-45 mS/cm in the conjugate fraction. The conductivity of the conjugate containing fraction is subsequently raised with Buffer C (50 mM Hepes, 5 M NaCl, 5 mM CaCl2, pH 6.9; by use of anti-chaotropic salts e.g. ammonium acetate) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.; or comparable HIC media) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, 5 mM CaCl2, pH 6.9). Free PEG-reagent was washed out within 5 CV Buffer D. Subsequently the conjugate was eluted with 100% Buffer E (50 mM Hepes, 5 mM CaCl2, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0288] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 17
Polysialylation of rFIX in the Presence of o-Amino Benzoic Acid
Method 1:
[0289] 8.2 mg rFIX is dissolved in 4.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). Then 82 .mu.l of an aqueous sodium periodate solution (5 mM) is added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 4 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 6 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0290] The retentate (6.5 ml), containing oxidized rFIX, is mixed with 1.64 ml of an aqueous o-amino benzoic acid (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture was incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0291] The further purification of the conjugate is carried out as described herein.
Method 2:
[0292] A solution of 1 mg rFIX in 0.65 ml sodium phosphate buffer, pH 6.0 containing a 5-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) was prepared. Then 333 .mu.l of an aqueous o-amino benzoic acid solution (30 mM) was added as nucleophilic catalyst to give a final concentration of 10 mM. Subsequently 20 .mu.l of an aqueous solution of NaIO4 (5 mM) was added yielding in a final concentration of 100 .mu.M. The coupling process was performed for 2 hours in the dark under gentle shaking at room temperature and quenched for 15 min at room temperature by the addition of 1 .mu.l of aqueous cysteine solution (1 M). The further purification of the conjugate is carried out as described herein.
Example 18
Polysialylation of EPO Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0293] A starting concentration of erythropoietin (EPO) is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0294] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl.sub.2), pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized EPO is eluted with Buffer B (20 mM Hepes, 5 mM CaCl.sub.2), 1M NaCl, pH 7.0). The EPO containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M HCl.
[0295] A 50-fold molar excess of a aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy-PSA reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl.sub.2). Finally the PSA-EPO containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (MWCO 10 kD, 50 cm.sup.2, Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0296] In an alternative embodiment, Method 1 is carried out as follows.
[0297] 10 mg EPO is dissolved in 5 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0298] The retentate (approx. 7 ml), containing oxidized EPO, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0299] The free EPO is removed by means of anion exchange chromatography (AEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 7.5). Free EPO is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 7.2 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSA-EPO conjugate a specific activity of >50% in comparison to native EPO is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free EPO.
Method 2:
[0300] EPO is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0301] The oxidized EPO is further purified by ion exchange chromatography. The oxidized EPO containing fractions of the eluate are collected and used for the conjugation reaction.
[0302] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized EPO within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0303] The obtained PSA-EPO conjugate is further purified by ion exchange chromatography. The PSA-EPO conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0304] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0305] Erythropoietin (EPO) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50 fold molar excess of a aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-EPO containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (MWCO 10 kD, 88 cm2, Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0306] In an alternative embodiment, Method 3 is carried out as follows. 10 mg EPO is dissolved in 8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently, the aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0307] The free EPO is removed by means of anion exchange chromatography (AEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 7.5). Free EPO is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 7.4). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against histidine buffer, pH 7.2 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSA-EPO conjugate a specific activity of >50% in comparison to native EPO is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free EPO.
Method 4:
[0308] EPO is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0309] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this EPO solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0310] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0311] The obtained PSA-EPO conjugate is purified by ion-exchange chromatography. The PSA-EPO containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (MWCO 10 kD, 88 cm2, Millipore).
[0312] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 19
Polysialylation of Ang-2 Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0313] A starting concentration of angiopoietin-2 (Ang-2) is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0314] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts, or, in the alternative, subjected to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized Ang-2 is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1 M NaCl, pH 7.0). The Ang-2 containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0315] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-Ang-2-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0316] In an alternative embodiment, Method 1 is carried out as follows. Angiopoietin-2 (Ang-2) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at R.T.
[0317] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0318] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-Ang-2 conjugate-containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0319] Ang-2 is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0320] The oxidized Ang-2 is further purified by ion exchange chromatography. The oxidized Ang-2 containing fractions of the eluate are collected and used for the conjugation reaction.
[0321] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Ang-2 within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0322] The obtained PSA-Ang-2 conjugate is further purified by ion-exchange chromatography
[0323] The PSA-Ang-2 conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0324] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0325] Angiopoietin-2 (Ang-2) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50 fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA Ang-2-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0326] In an alternative embodiment, Method 3 is carried out as follows. Angiopoietin-2 (Ang-2) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. PSA Ang-2-containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0327] Ang-2 is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0328] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this Ang-2 solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0329] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0330] The obtained PSA-Ang-2 conjugate is purified by ion-exchange chromatography. The PSA-Ang-2 containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0331] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 20
Polysialylation of VEGF Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0332] A starting concentration of vascular endothelial growth factor (VEGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0333] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized VEGF is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1 M NaCl, pH 7.0). The VEGF containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M NaOH.
[0334] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally the PSA-VEGF-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0335] In an alternative embodiment, Method 1 is carried out as follows. Vascular endothelial growth factor (VEGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0336] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0337] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-VEGF-containing fractions of the eluate are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0338] VEGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0339] The oxidized VEGF is further purified by ion exchange chromatography. The oxidized VEGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0340] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized VEGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0341] The obtained PSA-VEGF conjugate is further purified by ion exchange chromatography. The PSA-VEGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0342] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0343] Vascular endothelial growth factor (VEGF) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-VEGF containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0344] In an alternative embodiment, Method 3 is carried out as follows. Vascular endothelial growth factor (VEGF) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. The PSA-VEGF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0345] VEGF is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0346] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this VEGF solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0347] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0348] The obtained VEGF-conjugate is purified by ion-exchange chromatography. The PSA-VEGF containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0349] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 21
Polysialylation of EGF Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0350] A starting concentration of epidermal growth factor (EGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at R.T.
[0351] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized EGF is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The EGF containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M HCl.
[0352] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-EGF containing fractions are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0353] In an alternative embodiment, Method 1 is carried out as follows. Epidermal growth factor (EGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at R.T.
[0354] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0355] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-EGF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0356] EGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0357] The oxidized EGF is further purified by ion exchange chromatography. The oxidized EGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0358] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized EGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0359] The obtained PSA-EGF conjugate is further purified by ion exchange chromatography. The PSA-EGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0360] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0361] Epidermal growth factor (EGF) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally the PSA-EGF containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0362] In an alternative embodiment, Method 3 is carried out as follows. Epidermal growth factor (EGF) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. The conjugate containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0363] EGF is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0364] Subsequently the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this EGF-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0365] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0366] The obtained EGF-conjugate is purified by ion-exchange chromatography. The PSA-EGF containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0367] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 22
Polysialylation of NGF Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0368] A starting concentration of nerve growth factor (NGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0369] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized NGF is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The NGF containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M HCl.
[0370] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-NGF containing fractions are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0371] In an alternative embodiment, Method 1 is carried out as follows. Nerve growth factor (NGF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0372] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0373] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-NGF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0374] NGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0375] The oxidized NGF is further purified by ion exchange chromatography. The oxidized NGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0376] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized NGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0377] The obtained PSA-NGF conjugate is further purified by ion exchange chromatography. The PSA-NGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0378] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0379] Nerve growth factor (NGF) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA NGF-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0380] In an alternative embodiment, Method 3 is carried out as follows. Nerve growth factor (NGF) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. Then the PSA-NGF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0381] NGF is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0382] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this NGF-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0383] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0384] The obtained NGF-conjugate is purified by ion-exchange chromatography. The PSA-NGF containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0385] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 23
Polysialylation of HGH Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0386] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0387] A starting concentration of human growth hormone (HGH) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0388] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized HGH is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1 M NaCl, pH 7.0). The HGH containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0389] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-HGH containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0390] In an alternative embodiment, Method 1 is carried out as follows. As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art. HGH is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0391] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0392] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-HGH containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0393] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0394] HGH is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0395] The oxidized HGH is further purified by ion exchange chromatography. The oxidized HGH containing fractions of the eluate are collected and used for the conjugation reaction.
[0396] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized HGH within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0397] The obtained PSA-HGH conjugate is further purified by ion exchange chromatography. The PSA-HGH conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0398] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0399] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0400] Human growth hormone (HGH) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50 fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA HGH-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0401] In an alternative embodiment, Method 3 is carried out as follows. As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art. HGH is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50 fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. Then the PSA-HGH-containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0402] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0403] HGH is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0404] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this HGH-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0405] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0406] The obtained HGH-conjugate is purified by ion-exchange chromatography. The PSA-HGH containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0407] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 24
Polysialylation of TNF-Alpha Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
[0408] A starting concentration of tumor necrosis factor-alpha (TNF-alpha) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0409] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized TNF-alpha is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The TNF-alpha containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M HCl.
[0410] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally the PSA-TNF-alpha-containing fractions are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0411] In an alternative embodiment, Method 1 is carried out as follows. Tumor necrosis factor-alpha (TNF-alpha) is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0412] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-TNF-alpha containing fractions of the eluate are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0413] TNF-alpha is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0414] The oxidized TNF-alpha is further purified by ion exchange chromatography. The oxidized TNF-alpha containing fractions of the eluate are collected and used for the conjugation reaction.
[0415] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized TNF-alpha within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0416] The obtained PSA-TNF-alpha conjugate is further purified by ion exchange chromatography. The PSA-TNF-alpha conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0417] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0418] Tumor necrosis factor-alpha (TNF-alpha) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally the PSA-TNF-alpha-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0419] In an alternative embodiment, Method 3 is carried out as follows. Tumor necrosis factor-alpha (TNF-alpha) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). and the conjugate is purified by ion exchange chromatography. The PSA-TNF-alpha containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0420] TNF-alpha is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0421] Subsequently the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this TNF-alpha-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0422] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0423] The obtained TNF-alpha conjugate is purified by ion-exchange chromatography. The PSA-TNF-alpha containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0424] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 25
Polysialylation of Insulin Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0425] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art. A starting concentration of insulin is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0426] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized insulin is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1 M NaCl, pH 7.0). The insulin containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0427] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally the PSA-insulin containing fractions are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0428] In an alternative embodiment, Method 1 is carried out as follows. As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art. Insulin is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0429] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0430] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-insulin containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0431] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0432] Insulin is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0433] The oxidized insulin is further purified by ion exchange chromatography. The oxidized insulin containing fractions of the eluate are collected and used for the conjugation reaction.
[0434] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized insulin within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0435] The obtained PSA-insulin conjugate is further purified by ion exchange chromatography. The PSA-insulin conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0436] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0437] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0438] Insulin is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-insulin containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0439] In an alternative embodiment, Method 3 is carried out as follows. As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0440] Insulin is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. PSA-insulin containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0441] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0442] Insulin is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0443] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this insulin-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0444] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0445] The obtained insulin conjugate is purified by ion-exchange chromatography. The PSA-insulin containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0446] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 26
Polysialylation of Interferon-Alpha Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0447] A starting concentration of interferon-alpha is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0448] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized interferon-alpha is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The interferon-alpha containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0449] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally the PSA-interferon-alpha containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0450] In an alternative embodiment, Method 1 is carried out as follows. Interferon-alpha is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0451] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0452] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion-exchange chromatography. The PSA-interferon-alpha containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0453] Interferon-alpha is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0454] The oxidized interferon-alpha is further purified by ion exchange chromatography. The oxidized interferon-alpha containing fractions of the eluate are collected and used for the conjugation reaction.
[0455] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized interferon-gamma within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0456] The obtained PSA-interferon-alpha conjugate is further purified by ion exchange chromatography. The PSA-interferon-alpha conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
Method 3:
[0457] Interferon-alpha is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of a PSA aminooxy reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-interferon-alpha containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0458] In an alternative embodiment, Method 3 is carried out as follows. Interferon-alpha is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. The PSA-interferon-alpha containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0459] Interferon-alpha is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0460] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this interferon-alpha solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally, a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0461] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0462] The obtained interferon-alpha conjugate is purified by ion-exchange chromatography. The PSA-interferon-alpha containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0463] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 27
Polysialylation of Interferon-Gamma Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0464] 10 mg interferon-gamma is dissolved in 5 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0465] The retentate (approx. 7 ml), containing oxidized interferon-gamma, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0466] The free Interferon-gamma is removed by means of cation exchange chromatography (CEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free interferon-gamma is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 6.9). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSA-Interferon-gamma conjugate a specific activity of >50% in comparison to native Interferon-gamma is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Interferon gamma.
Method 2:
[0467] 10 mg interferon-gamma is dissolved in 8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently the aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0468] The free interferon gamma is removed by means of cation exchange chromatography (CEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free interferon-gamma is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 6.9). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSAinterferon-gamma conjugate a specific activity of >50% in comparison to native interferon-gamma is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free interferon-gamma.
Method 3:
[0469] 10 mg interferon-gamma is dissolved in 8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently the aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0470] The free interferon gamma is removed by means of cation exchange chromatography (CEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free interferon-gamma is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 6.9). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSAinterferon-gamma conjugate a specific activity of >50% in comparison to native interferon-gamma is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free interferon-gamma.
Method 4:
[0471] Interferon-gamma is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0472] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this interferon-gamma solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally, a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0473] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0474] The obtained interferon-gamma conjugate is purified by ion-exchange chromatography. The PSA-interferon-gamma containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0475] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 28
Polysialylation of G-CSF Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0476] A starting concentration of granulocyte-colony stimulating factor (G-CSF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0477] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized G-CSF is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1 M NaCl, pH 7.0). The G-CSF containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0478] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently, the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally the PSA-G-CSF-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0479] In an alternative embodiment, Method 1 is carried out as follows. Granulocyte-colony stimulating factor (G-CSF) is transferred into a reaction buffer (e.g., 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0480] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography. The PSA-G-CSF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0481] G-CSF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0482] The oxidized G-CSF is further purified by ion exchange chromatography. The oxidized G-CSF containing fractions of the eluate are collected and used for the conjugation reaction.
[0483] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized G-CSF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0484] The obtained PSA-G-CSF conjugate is further purified by ion exchange chromatography. The PSA-G-CSF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0485] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0486] Granulocyte-colony stimulating factor (G-CSF) is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally, the PSA-G-CSF-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0487] In an alternative embodiment, Method 3 is carried out as follows. Granulocyte-colony stimulating factor (G-CSF) is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. The PSA-G-CSF containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0488] G-CSF is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0489] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this G-CSF solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally, a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0490] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0491] The obtained G-CSF conjugate is purified by ion-exchange chromatography. The PSA-G-CSF containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0492] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 29
Polysialylation of Humira Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0493] A starting concentration of Humira is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0494] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized Humira is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The Humira containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5M HCl.
[0495] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-Humira containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0496] In an alternative embodiment, Method 1 is carried out as follows. Humira is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0497] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of ion exchange chromatography The PSA-Humira containing fractions of the elutae are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0498] Humira is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0499] The oxidized Humira is further purified by ion exchange chromatography. The oxidized Humira containing fractions of the eluate are collected and used for the conjugation reaction.
[0500] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Humira within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0501] The obtained PSA-Humira conjugate is further purified by ion exchange chromatography. The PSA-Humira conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0502] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0503] Humira is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally the PSA-Humira containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0504] In an alternative embodiment, Method 3 is carried out as follows. Humira is transferred into reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM) and the conjugate is purified by ion exchange chromatography. The PSA-Humira containing fractions of the eluate are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0505] Humira is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0506] Subsequently, the aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to this Humira solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0507] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0508] The obtained Humira-conjugate is purified by ion-exchange chromatography. The PSA-Humira containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0509] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 30
Polysialylation of Prolia Using Aminooxy-PSA and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0510] A starting concentration of Prolia is transferred into a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. To this solution, NaIO4 is added to give a final concentration of 200 .mu.M. The oxidation is carried at RT for 30 min in the dark under gentle shaking. The reaction is then quenched with cysteine (final concentration: 10 mM) for 60 min at RT.
[0511] The solution is next subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof or, in the alternative, to an IEX column with a volume of 20 ml (Merck EMD TMAE (M)) which is equilibrated with Buffer A (20 mM Hepes, 5 mM CaCl2, pH 7.0). The column is equilibrated with 5 CV Buffer A. The oxidized Prolia is eluted with Buffer B (20 mM Hepes, 5 mM CaCl2, 1M NaCl, pH 7.0). The Prolia containing fractions are collected. The protein content is determined (Coomassie, Bradford) and adjusted to 1 mg/ml with reaction buffer and adjusted to pH 6.0 by dropwise addition of 0.5 M HCl.
[0512] A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (final concentration: 10 mM). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. The excess of aminooxy reagent is removed by means of HIC. The conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with 80 ml Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes buffer pH 7.5 containing 5 mM CaCl2. Finally, the PSA-Prolia containing fractions are collected and subjected to UF/DF by use of a a membrane made of regenerated cellulose (Millipore). The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0513] In an alternative embodiment, Method 1 is carried out as follows. 10 mg Prolia is dissolved in 5 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0514] The retentate (approx. 7 ml), containing oxidized Prolia, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0515] The free Prolia is removed by means of cation exchange chromatography (CEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free Prolia is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently, the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 6.9). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD, Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSA-Prolia conjugate a specific activity of >50% in comparison to native Prolia is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Prolia.
Method 2:
[0516] Prolia is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0517] The oxidized Prolia is further purified by ion exchange chromatography. The oxidized Prolia containing fractions of the eluate are collected and used for the conjugation reaction.
[0518] The aminooxy-polysialic acid (PSA-ONH2) reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Prolia within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. at pH 6.0 in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking (protein concentration: 1 mg/ml).
[0519] The obtained Prolia conjugate is further purified by ion exchange chromatography. The Prolia conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0520] The conjugate prepared by use of this procedure is analytically characterized by measuring total protein, biological activity, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Method 3:
[0521] Prolia is transferred into reaction buffer (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and diluted to obtain a protein concentration of 1 mg/ml. A 50-fold molar excess of aminooxy-PSA reagent with a MW of 20 kD (described above) is added followed by m-toluidine as a nucleophilic catalyst (10 mM final concentration) and NaIO4 (final concentration: 400 .mu.M). The coupling reaction is performed for 2 hours in the dark under gentle shaking at room temperature. Subsequently, the reaction is quenched with cysteine for 60 min at RT (cysteine concentration: 10 mM). Then the conductivity of the reaction mixture is adjusted by adding a buffer containing ammonium acetate (50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, 8 M ammonium acetate, pH 6.9) and loaded onto a column filled with Phenyl Sepharose FF (GE Healthcare, Fairfield, Conn.) pre-equilibrated with 50 mM Hepes, 2.5 M ammonium acetate, 350 mM sodium chloride, 5 mM calcium chloride, 0.01% Tween 80, pH 6.9. Subsequently the conjugate is eluted with 50 mM Hepes, 5 mM calcium chloride, pH 7.5. Finally the PSA Prolia-containing fractions are collected and subjected to UF/DF by use of a membrane made of regenerated cellulose (Millipore). The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0522] In an alternative embodiment, Method 3 is carried out as follows. 10 mg Prolia is dissolved in 8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently the aminooxy-PSA reagent with a MW of 20 kD (described above) is added to give a 5 fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0523] The free Prolia is removed by means of cation exchange chromatography (CEC). The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free Prolia is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conductivity of the conjugate containing fractions is subsequently raised to .about.190 mS/cm with Buffer C (50 mM Hepes, 5 M NaCl, pH 6.9) and loaded onto a 20 ml HiPrep Butyl FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer D (50 mM Hepes, 3 M NaCl, pH 6.9). Free PSA-reagent is washed out within 5 CV Buffer D. Subsequently the conjugate is eluted with 100% Buffer E (50 mM Hepes, pH 6.9). The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm.sup.2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PSA-Prolia conjugate a specific activity of >50% in comparison to native Prolia is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Prolia.
Method 4:
[0524] Prolia is dissolved in or transferred to a reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution.
[0525] Subsequently the aminooxy-polysialic acid (PSA-ONH.sub.2) reagent is added in a 50-fold molar excess to this Prolia-solution within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. Finally a 40 mM aqueous sodium periodate solution is added to give a concentration of 400 .mu.M.
[0526] The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking. Then the reaction is stopped by the addition of an aqueous L-cysteine solution (1 M) to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0527] The obtained Prolia conjugate is purified by ion-exchange chromatography. The PSA-Prolia containing fractions of the eluate are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose (Millipore).
[0528] The conjugates prepared by use of this procedure are analytically characterized by measuring total protein, biological activity according to methods known in the art, and determination of the polysialyation degree by measuring the PSA content (resorcinol assay).
Example 31
Polysialylation of Other Therapeutic Proteins
[0529] Polysialylation reactions performed in the presence of alternative nucleophilic catalysts like m-toluidine or o-aminobenzoic acid as described herein may be extended to other therapeutic proteins. For example, in various aspects of the invention, the above polysialylation or PEGylation reactions as described herein with PSA aminooxy or PEG aminooxy reagents is repeated with therapeutic proteins such as those proteins described herein.
Example 32
PEGylation of EPO Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0530] Erythropoietin (EPO) is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EPO is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0531] The retentate containing oxidized EPO is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0532] Finally, the PEG-EPO conjugate is purified by ion-exchange chromatography (e.g. on Q Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl.sub.2). The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl.sub.2) and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0533] In an alternative embodiment, Method 1 is carried out as follows. EPO is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg EPO is dissolved in 5 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0534] The retentate (approx. 7 ml), containing oxidized EPO, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0535] Finally, the PEG-EPO conjugate is purified by ion-exchange chromatography on Q Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 7.5). Free EPO is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 7.2 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity biological activity according to methods known in the art. For the PEG-EPO conjugate a specific activity of >50% in comparison to native EPO is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free EPO.
Method 2:
[0536] EPO is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan).
[0537] EPO is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0538] The oxidized EPO is further purified by ion exchange chromatography. The oxidized EPO containing fractions of the eluate are collected and used for the conjugation reaction.
[0539] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized EPO within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0540] The obtained PEG-EPO conjugate is further purified by ion exchange chromatography. The PEG-EPO conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0541] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0542] EPO is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EPO is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0543] Finally, the PEG-EPO conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0544] In an alternative embodiment, Method 3 is carried out as follows. EPO is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg EPO is dissolved in .about.8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently, the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0545] Finally, the PEG-EPO conjugate is purified by ion-exchange chromatography on Q Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 7.5) and loaded onto a 20 ml HiPrep QFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 7.5). Free EPO is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 7.2 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PEG-EPO conjugate a specific activity of >50% in comparison to native EPO is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free EPO.
Method 4:
[0546] EPO is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of EPO is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg EPO/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0547] The PEG-EPO conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm.sup.2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl.sub.2), pH 7.5).
[0548] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 33
PEGylation of Ang-2 Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0549] Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Ang-2 is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0550] The retentate containing oxidized Ang-2 is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0551] Finally, the PEG-Ang-2 conjugate is purified by ion-exchange chromatography (e.g. on Q Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0552] In an alternative embodiment, Method 1 is carried out as follows. Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Ang-2 is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0553] The retentate containing oxidized Ang-2 is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0554] Finally, the PEG-Ang-2 conjugate is purified by ion-exchange chromatography. The conjugate containing fraction of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0555] Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan).
[0556] Ang-2 is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0557] The oxidized Ang-2 is further purified by ion exchange chromatography. The oxidized Ang-2 containing fractions of the eluate are collected and used for the conjugation reaction.
[0558] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Ang-2 within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0559] The obtained PEG-Ang-2 conjugate is further purified by ion exchange chromatography. The PEG-Ang-2 conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0560] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0561] Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Ang-2 is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0562] Finally, the PEG-Ang-2 conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0563] In an alternative embodiment, Method 3 is carried out as follows. Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Ang-2 is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0564] Finally the PEG-Ang-2 conjugate is purified by ion-exchange chromatography The conjugate containing reactions of the eluate are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0565] Ang-2 is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of Ang-2 is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg Ang-2/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0566] The PEG-Ang-2 conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0567] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
[0568] Subsequently, the free Ang-2 is removed by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF.
Example 34
PEGylation of VEGF Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0569] VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). VEGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0570] The retentate containing oxidized VEGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0571] Finally, the PEG-VEGF conjugate is purified by ion-exchange chromatography (e.g., on Q Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0572] In an alternative embodiment, Method 1 is carried out as follows. VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). VEGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0573] The retentate containing oxidized VEGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0574] Finally, the PEG-VEGF conjugate is purified by ion-exchange chromatography The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0575] VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). VEGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0576] The oxidized VEGF is further purified by ion exchange chromatography. The oxidized VEGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0577] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized VEGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0578] The obtained PEG-VEGF conjugate is further purified by ion exchange chromatography. The PEG-VEGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0579] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0580] VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). VEGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0581] Finally, the PEG-VEGF conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0582] In an alternative embodiment, Method 3 is carried out as follows. VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). VEGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0583] Finally, the PEG-VEGF conjugate is purified by ion-exchange chromatography. The conjugate conjugate fractions of the eluate are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0584] VEGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of VEGF is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg VEGF/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0585] The PEG-VEGF conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0586] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 35
PEGylation of EGF Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0587] EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0588] The retentate containing oxidized EGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0589] Finally, the PEG-EGF conjugate is purified by ion-exchange chromatography (e.g., on Q Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0590] In an alternative embodiment, Method 1 is carried out as follows. EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0591] The retentate containing oxidized EGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0592] Finally, the PEG-EGF conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0593] EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0594] The oxidized EGF is further purified by ion exchange chromatography. The oxidized EGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0595] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized NGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0596] The obtained PEG-EGF conjugate is further purified by ion exchange chromatography. The PEG-EGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0597] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0598] EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0599] Finally, the PEG-EGF conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0600] In an alternative embodiment, Method 3 is carried out as follows. EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0601] Finally, the PEG-EGF conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0602] EGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of EGF is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg EGF/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0603] The PEG-EGF conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0604] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 36
PEGylation of NGF Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0605] NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). NGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0606] The retentate containing oxidized NGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0607] Finally, the PEG-NGF conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0608] In an alternative embodiment, Method 1 is carried out as follows. NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). NGF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0609] The retentate containing oxidized NGF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0610] Finally, the PEG-NGF conjugate is purified by ion-exchange chromatography (The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0611] NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). NGF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0612] The oxidized NGF is further purified by ion exchange chromatography. The oxidized NGF containing fractions of the eluate are collected and used for the conjugation reaction.
[0613] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized NGF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0614] The obtained PEG-NGF conjugate is further purified by ion exchange chromatography. The PEG-NGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0615] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0616] NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). NGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0617] Finally, the PEG-NGF conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0618] In an alternative embodiment, Method 3 is carried out as follows. NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). NGF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0619] Finally, the PEG-NGF conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0620] NGF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of NGF is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg NGF/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0621] The PEG-NGF conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0622] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 37
PEGylation of HGH Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0623] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0624] HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). HGH is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0625] The retentate containing oxidized HGH is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0626] Finally, the PEG-HGH conjugate is purified by ion-exchange chromatography (e.g., on Q Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0627] In an alternative embodiment, Method 1 is carried out as follows. As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0628] HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). HGH is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0629] The retentate containing oxidized HGH is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0630] Finally, the PEG-HGH conjugate is purified by ion-exchange chromatography (The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0631] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0632] HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). HGH is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0633] The oxidized HGH is further purified by ion exchange chromatography. The oxidized HGH containing fractions of the eluate are collected and used for the conjugation reaction.
[0634] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized HGH within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0635] The obtained PEG-HGH conjugate is further purified by ion exchange chromatography. The PEG-NGF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0636] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0637] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0638] HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). HGH is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0639] Finally, the PEG-HGH conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0640] In an alternative embodiment, Method 3 is carried out as follows. As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art. HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). HGH is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0641] Finally, the PEG-HGH conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0642] As described herein, the amino acid sequence of human growth hormone (HGH) is first modified to incorporate at least one glycosylation site. Following purification, HGH is glycosylated in vitro according to methods known in the art.
[0643] HGH is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of HGH is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg HGH/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0644] The PEG-HGH conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0645] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 38
PEGylation of TNF-Alpha Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0646] TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). TNF-alpha is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0647] The retentate containing oxidized TNF-alpha is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0648] Finally, the PEG-TNF-alpha conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0649] In an alternative embodiment, Method 1 is carried out as follows. TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). TNF-alpha is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0650] The retentate containing oxidized TNF-alpha is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 mm at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0651] Finally, the PEG-TNF-alpha conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0652] TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). TNF-alpha is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0653] The oxidized TNF-alpha is further purified by ion exchange chromatography. The oxidized TNF-alpha containing fractions of the eluate are collected and used for the conjugation reaction.
[0654] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized TNF alpha within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0655] The obtained PEG-TNF-alpha conjugate is further purified by ion exchange chromatography. The PEG-TNF-alpha conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0656] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0657] TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). TNF-alpha is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0658] Finally, the PEG-TNF-alpha conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0659] In an alternative embodiment, Method 3 is carried out as follows. TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). TNF-alpha is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0660] Finally, the PEG-TNF-alpha conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0661] TNF-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of TNF-alpha is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg TNF-alpha/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0662] The PEG-TNF-alpha conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0663] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 39
PEGylation of Insulin Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0664] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art. Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Insulin is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0665] The retentate containing oxidized insulin is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0666] Finally, the PEG-insulin conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0667] In an alternative embodiment, Method 1 is carried out as follows. As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art. Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Insulin is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0668] The retentate containing oxidized insulin is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0669] Finally, the PEG-insulin conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0670] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0671] Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Insulin is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0672] The oxidized insulin is further purified by ion exchange chromatography. The oxidized insulin containing fractions of the eluate are collected and used for the conjugation reaction.
[0673] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized insulin within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0674] The obtained PEG-insulin conjugate is further purified by ion exchange chromatography. The PEG-insulin conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0675] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0676] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0677] Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Insulin is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0678] Finally, the PEG-insulin conjugate is purified by ion-exchange chromatography on Q Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0679] In an alternative embodiment, Method 3 is carried out as follows. As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art. Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Insulin is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0680] Finally, the insulin-conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0681] As described herein, the amino acid sequence of insulin is first modified to incorporate at least one glycosylation site. Following purification, insulin is glycosylated in vitro according to methods known in the art.
[0682] Insulin is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of insulin is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg insulin/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0683] The PEG-insulin conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0684] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 40
PEGylation of Interferon-Alpha Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0685] Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-alpha is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0686] The retentate containing oxidized interferon-alpha is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0687] Finally, the PEG-interferon-alpha conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0688] In an alternative embodiment, Method 1 is carried out as follows. Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-alpha is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0689] The retentate containing oxidized interferon-alpha is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0690] Finally, the PEG-interferon-alpha conjugate is purified by ion-exchange chromatography The conjugate containing freactions are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0691] Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-alpha is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0692] The oxidized interferon-alpha is further purified by ion exchange chromatography. The oxidized interferon-alpha containing fractions of the eluate are collected and used for the conjugation reaction.
[0693] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized interferon-alpha within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0694] The obtained PEG-interferon-alpha conjugate is further purified by ion exchange chromatography. The PEG-interferon alpha conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0695] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0696] Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-alpha is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0697] Finally, the PEG-interferon-alpha conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0698] In an alternative embodiment, Method 3 is carried out as follows. Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-alpha is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0699] Finally, the PEG-interferon-alpha conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0700] Interferon-alpha is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of interferon-alpha is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg interferon-alpha/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0701] The PEG-interferon-alpha conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0702] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 41
PEGylation of Interferon-Gamma Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0703] Interferon-gamma is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg Interferon-gamma is dissolved in 5 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0704] The retentate (approx. 7 ml), containing oxidized interferon-gamma, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0705] Finally, the PEG-interferon-gamma conjugate is purified by ion-exchange chromatography on SP Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free interferon-gamma is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PEG-interferon-gamma conjugate a specific activity of >50% in comparison to native Interferon gamma is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Interferon-gamma.
Method 2:
[0706] Interferon-gamma is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Interferon-gamma is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5 N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0707] The oxidized interferon-gamma is further purified by ion exchange chromatography. The oxidized interferon-gamma containing fractions of the eluate are collected and used for the conjugation reaction.
[0708] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized interferon-gamma within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0709] The obtained PEG-interferon-gamma conjugate is further purified by ion exchange chromatography. The PEG-interferon-gamma conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
[0710] The conjugate prepared by use of this procedure are analytically characterized by measuring total protein and biological activity according to methods known in the art.
Method 3:
[0711] Interferon-gamma is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg interferon-gamma is dissolved in .about.8 ml histidine-buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0712] Finally the PEG-interferon-gamma conjugate is purified by ion-exchange chromatography on SP-Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SP FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free intergferon-gamma is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according according to methods known in the art. For the PEG-interferon-gamma conjugate a specific activity of >50% in comparison to native interferon-gamma is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free interferon-gamma.
Method 4:
[0713] Interferon-gamma is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of interferon-gamma is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg interferon-gamma/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0714] The PEG-interferon-gamma conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0715] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 42
PEGylation of G-CSF Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0716] G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). G-CSF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0717] The retentate containing oxidized G-CSF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0718] Finally, the PEG-G-CSF conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0719] In an alternative embodiment, Method 1 is carried out as follows. G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). G-CSF is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0720] The retentate containing oxidized G-CSF is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0721] Finally, the PEG-G-CSF conjugate is purified by ion-exchange chromatography (The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0722] G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). G-CSF is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0723] The oxidized G-CSF is further purified by ion exchange chromatography. The oxidized G-CSF containing fractions of the eluate are collected and used for the conjugation reaction.
[0724] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized G-CSF within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0725] The obtained PEG-G-CSF conjugate is further purified by ion exchange chromatography. The PEG-G-CSF conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
Method 3:
[0726] G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). G-CSF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0727] Finally, the PEG-G-CSF conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0728] In an alternative embodiment, Method 3 is carried out as follows. G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). G-CSF is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0729] Finally, the PEG-G-CSF conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0730] G-CSF is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of G-CSF is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg G-CSF/ml. Subsequently, an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0731] The G-CSF conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0732] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 43
PEGylation of Humira Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0733] Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Humira is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0734] The retentate containing oxidized Humira is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0735] Finally, the PEG-Humira conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0736] In an alternative embodiment, Method 1 is carried out as follows. Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Humira is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0737] The retentate containing oxidized Humira is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0738] Finally, the PEG-Humira conjugate is purified by ion-exchange chromatography. The conjugate containing fractions of the eluate are collected and then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
Method 2:
[0739] Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Humira is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0740] The oxidized Humira is further purified by ion exchange chromatography. The oxidized Humira containing fractions of the eluate are collected and used for the conjugation reaction.
[0741] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Humira within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0742] The obtained PEG-Humira conjugate is further purified by ion exchange chromatography. The PEG-Humira conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
Method 3:
[0743] Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Humira is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently, the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0744] Finally, the PEG-Humira conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0745] In an alternative embodiment, Method 3 is carried out as follows. Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Humira is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0746] Finally, the PEG-Humira conjugate is purified by ion-exchange chromatography. The conjugate containing fractions are collected and then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
Method 4:
[0747] Humira is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of Humira is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg Humira/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of a 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0748] The Humira conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0749] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 44
PEGylation of Prolia Using an Aminooxy-PEG Reagent and m-Toluidine as a Nucleophilic Catalyst
Method 1:
[0750] Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Prolia is dissolved in 7.0 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl, 5 mM CaCl2). An aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 7.5 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0751] The retentate containing oxidized Prolia is next mixed with an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Aminooxy-PEG reagent with a MW of 20 kD is then added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at room temperature in the dark under gentle stirring.
[0752] Finally, the PEG-Prolia conjugate is purified by ion-exchange chromatography (e.g., on Q-Sepharose FF). For example, 1.5 mg protein/ml gel is loaded on the column equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using an appropriate MW cutoff membrane. The preparation is next analytically characterized by measuring total protein (Coomassie, Bradford) and biological activity according to methods known in the art.
[0753] In an alternative embodiment, Method 1 is carried out as follows. Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg rFIX is dissolved in 5 ml histidine-buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 100 .mu.l of an aqueous sodium periodate solution (5 mM) is then added and the reaction mixture is incubated for 1 h in the dark at 4.degree. C. under gentle stirring and quenched for 15 min at room temperature by the addition of 50 .mu.l of a 1 M aqueous cysteine solution. The mixture is subsequently subjected to UF/DF employing Vivaspin 15R 10 kD centrifugal filtrators to remove excess periodate, quencher and the byproducts thereof.
[0754] The retentate (approx. 7 ml), containing oxidized Prolia, is mixed with 2 ml of an aqueous m-toluidine solution (50 mM) and incubated for 30 min at room temperature. Then aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. This mixture is incubated for 2.5 h at RT in the dark under gentle stirring.
[0755] Finally the PEG-Prolia conjugate is purified by ion-exchange chromatography on SP Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SP FF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free Prolia is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PEG-Prolia conjugate a specific activity of >50% in comparison to native Prolia is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Prolia.
Method 2:
[0756] Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). Prolia is transferred or dissolved in reaction buffer (e.g. 50 mM Hepes, 350 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 1.0+/-0.25 mg/ml. Then the pH of the solution is corrected to 6.0 by drop wise addition of a 0.5N aqueous HCl solution. Subsequently, a 40 mM aqueous sodium periodate solution is added within 10 minutes to give a concentration of 200 .mu.M. The oxidation reaction is carried out for 30+/-5 min at a temperature (T) of T=+22+/-2.degree. C. Then the reaction is stopped by addition of an aqueous L-cysteine solution (1 M) within 15 minutes at T=+22+/-2.degree. C. to give a final concentration of 10 mM in the reaction mixture and incubation for 60+/-5 min.
[0757] The oxidized Prolia is further purified by ion exchange chromatography. The oxidized Humira containing fractions of the eluate are collected and used for the conjugation reaction.
[0758] The aminooxy-PEG reagent with a MW of 20 kD reagent is added in a 50-fold molar excess to the eluate containing the purified oxidized Prolia within a maximum time period (t) of 15 minutes under gentle stirring. Then an aqueous m-toluidine solution (50 mM) is added within 15 minutes to get a final concentration of 10 mM. The reaction mixture is incubated for 120+/-10 min. in the dark at a temperature (T) of T=+22+/-2.degree. C. under gentle shaking.
[0759] The obtained PEG-Prolia conjugate is further purified by ion exchange chromatography. The PEG-Prolia conjugate containing fractions are collected and concentrated by ultra-/diafiltration (UF/DF) using a membrane made of regenerated cellulose with an appropriate molecular weight cut off (Millipore).
Method 3:
[0760] Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). EPO is dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) and mixed with an aqueous sodium periodate solution (10 mM), and an aqueous m-toluidine solution (50 mM). Subsequently the aminooxy reagent is added to give a 20-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 8 .mu.l of aqueous cysteine solution (1 M).
[0761] Finally, the PEG-Prolia conjugate is purified by ion-exchange chromatography on Q-Sepharose FF. 1.5 mg protein/ml gel is loaded on the column pre equilibrated with 50 mM Hepes buffer, pH 7.4 containing 5 mM CaCl2. The conjugate is eluted with 50 mM Hepes buffer containing 5 mM CaCl2 and 500 mM sodium chloride, pH 7.4 and is then subjected to UF/DF using a membrane. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art.
[0762] In an alternative embodiment, Method 3 is carried out as follows.
[0763] Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). 10 mg Prolia is dissolved in .about.8 ml histidine buffer, pH 6.0 (20 mM L-histidine, 150 mM NaCl). 200 .mu.l of an aqueous sodium periodate solution (5 mM) and 2 ml of an aqueous m-toluidine solution (50 mM) are then added. Subsequently, the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 5-fold molar reagent excess. The mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of 100 .mu.l of 1 M aqueous cysteine solution.
[0764] Finally the PEG-Prolia conjugate is purified by ion-exchange chromatography on SP-Sepharose FF. The reaction mixture is diluted with 20 ml Buffer A (50 mM Hepes, pH 6.5) and loaded onto a 20 ml HiPrep SPFF 16/10 column (GE Healthcare, Fairfield, Conn.) pre-equilibrated with Buffer A. Then the column is eluted with Buffer B (50 mM Hepes, 1 M NaCl, pH 6.5). Free Prolia is eluted by washing the column with 25% Buffer B and the conjugate at 50% Buffer B. The conjugate containing fractions are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against histidine buffer, pH 6.9 containing 150 mM NaCl. The preparation is analytically characterized by measuring total protein (Bradford) and biological activity according to methods known in the art. For the PEG-Prolia conjugate a specific activity of >50% in comparison to native Prolia is determined. The conjugate is additionally analytically characterized by Size Exclusion HPLC using a Agilent 1200 HPLC system equipped with a Shodex KW 803 column under conditions as previously described (Kolarich et al, Transfusion 2006; 46:1959-77). It is shown that the preparation contains no free Prolia.
Method 4:
[0765] Prolia is PEGylated by use of a linear 20 kD PEGylation reagent containing an aminooxy group. An example of this type of reagent is the Sunbright.RTM. CA series from NOF (NOF Corp., Tokyo, Japan). An initial concentration or weight of Humira is transferred or dissolved in Hepes buffer (50 mM Hepes, 150 mM sodium chloride, 5 mM calcium chloride, pH 6.0) to get a final protein concentration of 2 mg Prolia/ml. Subsequently an 5 mM aqueous sodium periodate solution is added within 15 minutes to give a final concentration of 100 .mu.M, followed by addition of an 50 mM aqueous m-toluidine solution to get a final concentration of 10 mM within a time period of 30 minutes. Then the aminooxy-PEG reagent with a MW of 20 kD (described above) is added to give a 20-fold molar reagent excess. After correction of the pH to 6.0 the mixture is incubated for 2 h in the dark at room temperature under gentle stirring and quenched for 15 min at room temperature by the addition of an 1 M aqueous L-cysteine solution to give a final concentration of 10 mM.
[0766] The Prolia conjugate is purified by means of ion exchange chromatography (IEC). The conjugate containing fractions of the eluate are concentrated by UF/DF using a 10 kD membrane made of regenerated cellulose (88 cm2, cut-off 10 kD/Millipore). The final diafiltration step is performed against Hepes buffer (50 mM Hepes, 5 mM CaCl2, pH 7.5).
[0767] The preparation is analytically characterized by measuring total protein (Bradford and BCA procedure) and biological activity according to known methods.
Example 45
PEGylation of a Therapeutic Protein Using Branched PEG
[0768] PEGylation of a therapeutic protein of the invention may be extended to a branched or linear PEGylation reagent, which is made of an aldehyde and a suitable linker containing an active aminooxy group.
Sequence CWU 1
1
11422PRTHomo sapiens 1Leu Asn Arg Pro Lys Arg Tyr Asn Ser Gly Lys
Leu Glu Glu Phe Val1 5 10 15Gln Gly Asn Leu Glu Arg Glu Cys Met Glu
Glu Lys Cys Ser Phe Glu 20 25 30Glu Pro Arg Glu Val Phe Glu Asn Thr
Glu Lys Thr Thr Glu Phe Trp 35 40 45Lys Gln Tyr Val Asp Gly Asp Gln
Cys Glu Ser Asn Pro Cys Leu Asn 50 55 60Gly Gly Ser Cys Lys Asp Asp
Ile Asn Ser Tyr Glu Cys Trp Cys Pro65 70 75 80Phe Gly Phe Glu Gly
Lys Asn Cys Glu Leu Asp Val Thr Cys Asn Ile 85 90 95Lys Asn Gly Arg
Cys Glu Gln Phe Cys Lys Asn Ser Ala Asp Asn Lys 100 105 110Val Val
Cys Ser Cys Thr Glu Gly Tyr Arg Leu Ala Glu Asn Gln Lys 115 120
125Ser Cys Glu Pro Ala Val Pro Phe Pro Cys Gly Arg Val Ser Val Ser
130 135 140Gln Thr Ser Lys Leu Thr Arg Ala Glu Ala Val Phe Pro Asp
Val Asp145 150 155 160Tyr Val Asn Pro Thr Glu Ala Glu Thr Ile Leu
Asp Asn Ile Thr Gln 165 170 175Gly Thr Gln Ser Phe Asn Asp Phe Thr
Arg Val Val Gly Gly Glu Asp 180 185 190Ala Lys Pro Gly Gln Phe Pro
Trp Gln Val Val Leu Asn Gly Lys Val 195 200 205Asp Ala Phe Cys Gly
Gly Ser Ile Val Asn Glu Lys Trp Ile Val Thr 210 215 220Ala Ala His
Cys Val Glu Thr Gly Val Lys Ile Thr Val Val Ala Gly225 230 235
240Glu His Asn Ile Glu Glu Thr Glu His Thr Glu Gln Lys Arg Asn Val
245 250 255Ile Arg Ala Ile Ile Pro His His Asn Tyr Asn Ala Ala Ile
Asn Lys 260 265 270Tyr Asn His Asp Ile Ala Leu Leu Glu Leu Asp Glu
Pro Leu Val Leu 275 280 285Asn Ser Tyr Val Thr Pro Ile Cys Ile Ala
Asp Lys Glu Tyr Thr Asn 290 295 300Ile Phe Leu Lys Phe Gly Ser Gly
Tyr Val Ser Gly Trp Ala Arg Val305 310 315 320Phe His Lys Gly Arg
Ser Ala Leu Val Leu Gln Tyr Leu Arg Val Pro 325 330 335Leu Val Asp
Arg Ala Thr Cys Leu Arg Ser Thr Lys Phe Thr Ile Tyr 340 345 350Asn
Asn Met Phe Cys Ala Gly Phe His Glu Gly Gly Arg Asp Ser Cys 355 360
365Gln Gly Asp Ser Gly Gly Pro His Val Thr Glu Val Glu Gly Thr Ser
370 375 380Phe Leu Thr Gly Ile Ile Ser Trp Gly Glu Glu Cys Ala Met
Lys Gly385 390 395 400Lys Tyr Gly Ile Tyr Thr Lys Val Ser Arg Tyr
Val Asn Trp Ile Lys 405 410 415Glu Lys Thr Lys Leu Thr 420
References











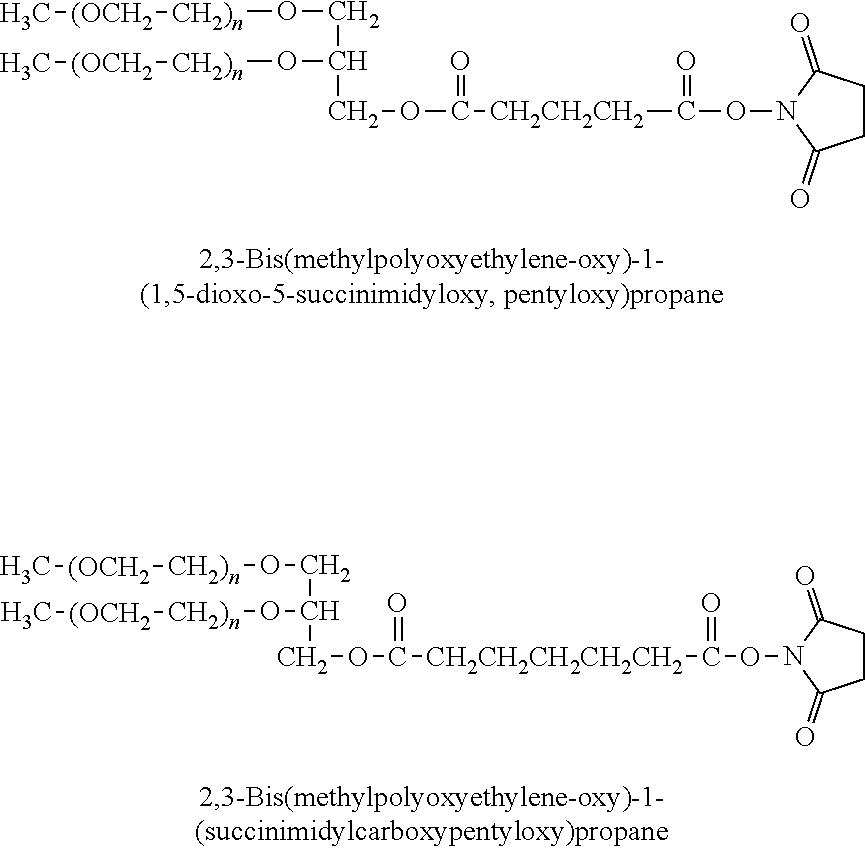
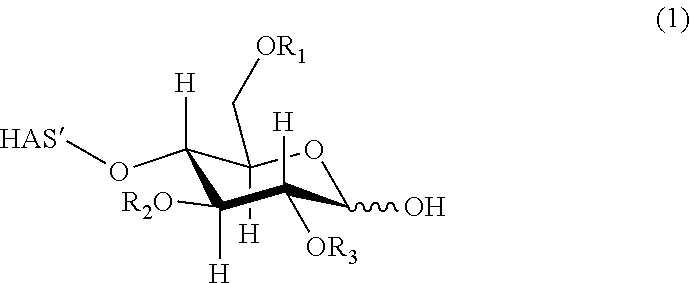




D00001

D00002

D00003
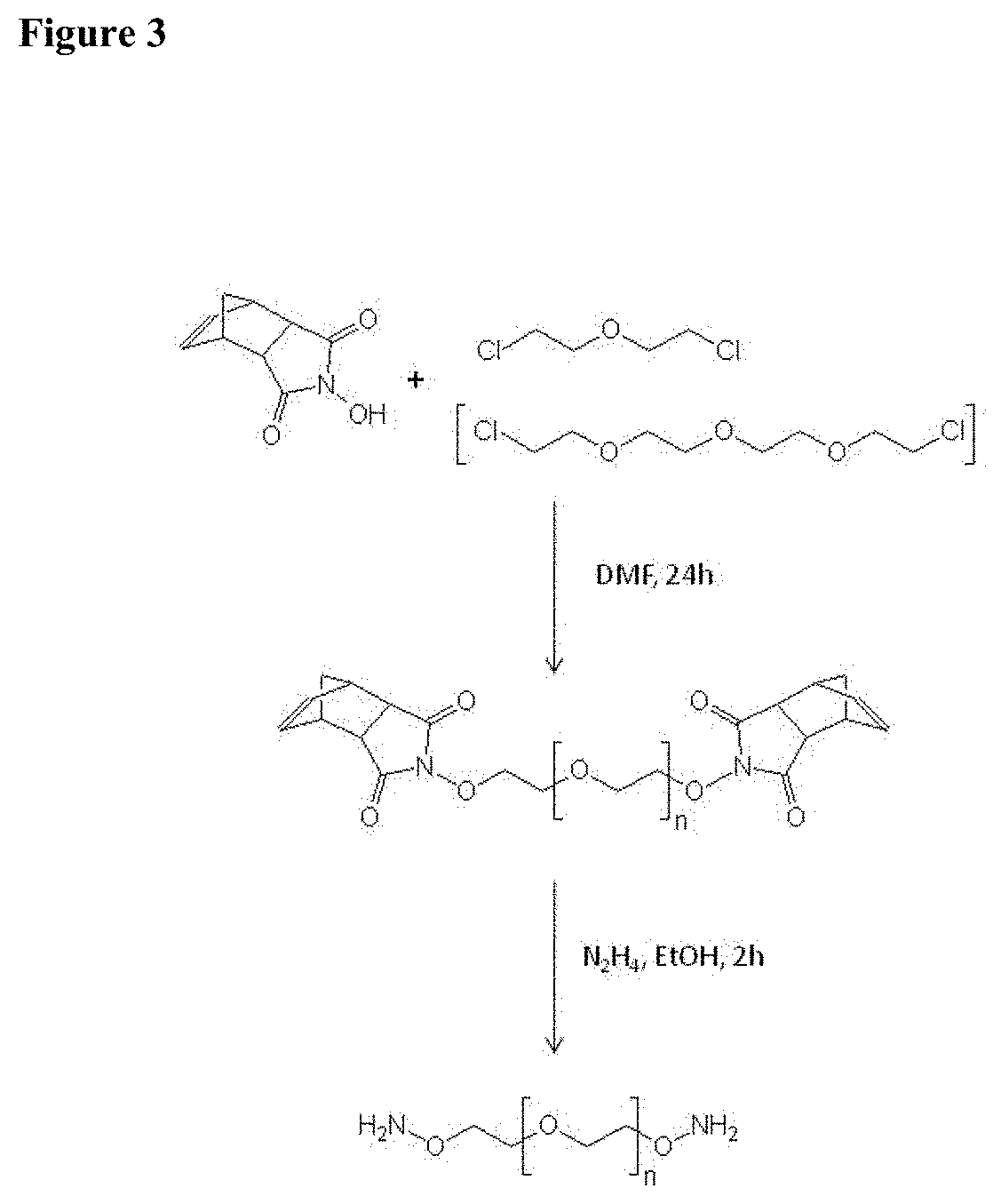
D00004

D00005

D00006

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.