Targeting Moiety Peptide Epitope Complexes Having a Plurality of T-Cell Epitopes
Cobbold; Mark ; et al.
U.S. patent application number 16/558467 was filed with the patent office on 2020-01-16 for targeting moiety peptide epitope complexes having a plurality of t-cell epitopes. This patent application is currently assigned to The University of Birmingham. The applicant listed for this patent is The University of Birmingham. Invention is credited to Mark Cobbold, David Millar.
| Application Number | 20200016262 16/558467 |
| Document ID | / |
| Family ID | 55629091 |
| Filed Date | 2020-01-16 |











View All Diagrams
| United States Patent Application | 20200016262 |
| Kind Code | A1 |
| Cobbold; Mark ; et al. | January 16, 2020 |
Targeting Moiety Peptide Epitope Complexes Having a Plurality of T-Cell Epitopes
Abstract
A variety of targeting moiety peptide epitope complexes (TPECs) are described in different embodiments. In each of the embodiments, however, a targeting moiety may be used to deliver the TPEC to an area of unwanted cells, allowing for a therapeutic effect to be delivered locally. The TPEC also contains a plurality of T-cell epitopes. The TPEC further comprises cleavage sites that release the T-cell epitopes from the targeting agent, and in some embodiments from each other, when they are in the microenvironment of the unwanted cells. Although the arrangement and number of T-cell epitopes varies in different embodiments described herein, once cleaved from the targeting agent (and any neighboring T-cell epitopes), the T-cell epitopes function by stimulating an immune response against the unwanted cells.
| Inventors: | Cobbold; Mark; (Winchester, MA) ; Millar; David; (Winchester, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | The University of
Birmingham Birmingham GB |
||||||||||
| Family ID: | 55629091 | ||||||||||
| Appl. No.: | 16/558467 | ||||||||||
| Filed: | September 3, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15012115 | Feb 1, 2016 | 10441649 | ||
| 16558467 | ||||
| 62111069 | Feb 2, 2015 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 47/6811 20170801; A61K 2039/585 20130101; A61K 47/646 20170801; A61K 2039/6056 20130101; A61K 39/245 20130101; A61K 47/6881 20170801; A61P 43/00 20180101; A61K 47/6849 20170801; A61P 35/02 20180101; A61P 31/22 20180101; A61P 37/00 20180101; A61P 37/02 20180101; A61P 35/00 20180101; C12N 2710/16134 20130101 |
| International Class: | A61K 39/245 20060101 A61K039/245; A61K 47/64 20060101 A61K047/64; A61K 47/68 20060101 A61K047/68 |
Claims
1. A method of treating cancer in a patient comprising administering a composition comprising a targeting moiety peptide epitope complex (TPEC) to the patient, wherein: a. T is a targeting moiety that is an antibody or antigen-binding fragment thereof capable of targeting cancer cells; b. L is at least one linker capable of linkage to T where L is a peptide bond or at least one peptide; c. C is at least one cleavage site cleaved by an enzyme outside of the cancer cell and expressed by the cancer cell or cleaved by a protease that is outside of the cancer cell and colocalized to the cancer cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and d. E is at least one viral T-cell epitope that elicits an existing immune response in a human subject and binds to an HLA molecule on the surface of the cancer cells of the human subject and has an HLA matched to the subject, wherein the L, C, and E moieties are arranged in a pattern of at least one of L-(C-E)n, wherein n is an integer of at least 2 and with each C-E attached to the L in series.
2. The method of claim 1, wherein the method comprises retargeting an immune response of a patient to cancer cells.
3. The method of claim 2, wherein the composition comprises a plurality of more than 10 T-cell epitopes conjugated to the targeting moiety with at least one cleavage site, optionally wherein the plurality of T-cell epitopes are not all identical.
4. The method of claim 2, wherein at least one T-cell epitope is an MHC Class I restricted peptide or an MHC Class II restricted peptide.
5. The method of claim 2, wherein the plurality of T-cell epitopes are from about 7 to about 14 amino acids in length.
6. The method of claim 2, wherein the composition comprises at least about 2, 3, 4, 5, 6, 7, 8, 9, or 10 T cell epitopes.
7. The method of claim 2, wherein the T-cell epitopes are chosen from CMV, influenza, EBV, hepatitis, chicken pox, mumps, measles, rubella, polio, rotavirus, vaccinia, and yellow fever T-cell epitopes.
8. The method of claim 2, wherein the composition comprises T-cell epitopes from at least two different viruses.
9. The method of claim 2, wherein the T-cell epitopes are chosen from HLA-A, HLA-B, HLA-C, HLA-E, HLA-F, HLA-G, CD1d, and MR1.
10. The method of claim 2, wherein the T-cell epitopes are chosen from HLA-A*01, HLA-A*02, HLA-A*03, HLA-A*11, HLA-B*44, HLA-B*07, HLA-B*08, HLA-B*15, HLA-B*35, HLA-B*40, HLA-C*07, HLA-C*03, HLA-C*05, HLA-C*04, HLA-C*06, and HLA-E*0101 restricted antigens.
11. The method of claim 2, wherein the composition comprises at least the following T-cell epitopes: HLA-A*02, HLA-A*01, and HLA-A*03.
12. The method of claim 2, wherein the T-cell epitopes comprise at least one of SEQ ID NOS: 1-2, 5-7, 15-57.
13. The method of claim 2, wherein the T-cell epitopes are flanked on one or both ends by at least one human protein domain.
14. The method of claim 2, wherein the enzyme expressed by the cancer cells is a protease.
15. The method of claim 2, wherein the antibody or antigen-binding fragment thereof is an anti-CEA or anti-CEACAM antibody or antigen-binding fragment thereof.
16. The method of claim 2, wherein the patient does not develop an immune response against the composition sufficient to inactivate the composition.
17. The method of claim 2, wherein the patient receives multiple doses of the composition over at least 30, 45, 60, 75, 90, 120, 150, or more days or on an ongoing basis.
18. The method of claim 2, wherein the patient receives multiple doses of the composition over at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 months or on an ongoing basis.
19. The method of claim 2, wherein the composition can be administered to a patient in relapse who received the composition for an earlier round of therapy.
20. The method of claim 2, wherein the cancer is breast cancer, ovarian cancer, endometrial cancer, cervical cancer, bladder cancer, renal cancer, melanoma, lung cancer, prostate cancer, testicular cancer, thyroid cancer, brain cancer, esophageal cancer, gastric cancer, pancreatic cancer, colorectal cancer, liver cancer, leukemia, myeloma, nonHodgkin's lymphoma, Hodgkin's lymphoma, acute myeloid leukemia, acute lymphoblastic leukemia, chronic lymphoblastic leukemia, lymphoproliferative disorder, myelodysplastic disorder, myeloproliferative disease, or premalignant disease
21. The method of claim 2, wherein the composition is administered in conjunction with surgery, radiation, traditional chemotherapy, or an immunostimulatory agent.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a divisional of U.S. application Ser. No. 15/012,115, filed Feb. 1, 2016, which claims the benefit of priority of U.S. Provisional Application No. 62/111,069, filed Feb. 2, 2015.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Apr. 15, 2016, is named 20160415_01131-0005-00US_SeqProject_ST25.txt and is 40,509 bytes in size.
DESCRIPTION
Field
[0003] This application relates to immunotherapeutic agents employing a plurality of T-cell epitopes. In particular, it relates to agents that can be used to treat a condition characterized by the presence of unwanted cells, such as cancer or other disease-causing cells.
Background
[0004] Cancer and other diseases caused by the presence of unwanted cells create significant loss of life, suffering, and economic impact. Immunotherapeutic strategies for targeting cancer have been an active area of translational clinical research.
[0005] WO 2012/123755 discusses the concept of re-directed immunotherapy. In this application, an agent for preventing or treating a condition characterized by the presence of unwanted cells includes a targeting moiety that is capable of targeting to the unwanted cells and a T-cell epitope that can be released from the targeting moiety by selective cleavage of a cleavage site in the agent in the vicinity of the unwanted cells.
[0006] WO 2014/043523 teaches an agent based on an ScFV directed to cancer cells including from 1-10 immunogenic CD8 T-cell epitopes in one of the following two arrangements: T-c-En-c-Fcn or T-c-Fcn-c-En, where T is the ScFv, En is from 1-10 CD8 T-cell epitopes, c: is a protease cleavage site, and Fcn is from 1-10 Fc portions of an IgG antibody. In this reference, the 1-10 immunogenic CD8 T-cells are released from the ScFv and Fc portions of the agent in a single polypeptide chain, still conjugated to each other.
[0007] While some positive test data has been shown with prior approaches, clinically-effective therapeutic strategies must be able to elicit a strong immune response in an individual suffering from a disease such as cancer. Additionally, effective therapies should work well in a wide cross-section of patients from different racial and ethnic groups Maximally-effective therapies would also generate an immune response against the unwanted cells without generating an inhibitory immune response against the therapeutic agent itself so that multiple rounds of treatment could be administered over a period of time. Therefore, additional developments in this field of re-directed immunotherapy are required.
SUMMARY
[0008] In accordance with the description, a variety of targeting moiety peptide epitope complexes (TPECs) are described in different embodiment of this application. In each of the embodiments, however, a targeting moiety may be used to deliver the TPEC to an area of unwanted cells, allowing for a therapeutic effect to be delivered locally. The TPEC also contains a plurality of T-cell epitopes. The TPEC further comprises cleavage sites that release the T-cell epitopes from the targeting agent, and in some embodiments from each other, when they are in the microenvironment of the unwanted cells. Although the arrangement and number of T-cell epitopes varies in different embodiments described herein, once cleaved from the targeting agent (and any neighboring T-cell epitopes), the T-cell epitopes function by stimulating an immune response against the unwanted cells. In some embodiments, maximal benefits may be achieved by releasing all of the T-cell epitopes from both the targeting agent and from each other in the cleavage process, allowing each T-cell epitope the structural freedom to attract an immune response to the unwanted cell.
[0009] Having a plurality of T-cell epitopes, as discussed in detail below, enhances the immune response against the unwanted cells, either by stimulating a stronger immune response in a given patient or by allowing the TPEC to stimulate an immune response across a wide variety of patients in different ethnic and racial groups.
[0010] In one embodiment, a composition for retargeting an immune response to unwanted cells comprises a TPEC wherein:
[0011] a. T is a targeting moiety that is capable of targeting unwanted cells;
[0012] b. L is at least one linker capable of chemical linkage to T where L may be a peptide bond, at least one peptide, or a chemical linker;
[0013] c. C is at least one cleavage site [0014] i. cleaved by an enzyme expressed by the unwanted cells; [0015] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0016] iii. cleaved by a complement-dependent cleavage reaction; or [0017] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and
[0018] d. E is at least one T-cell epitope, wherein the L, C, and E moieties are arranged in at least one of the following patterns: [0019] i. a plurality of L-C-E, each attached separately to T, [0020] ii. at least one of L-(C-E)n, with each C-E attached to the L in parallel, and/or [0021] iii. at least one of L-(C-E)n, with each C-E attached to the L in series.
[0022] In one embodiment, a composition for retargeting an immune response to unwanted cells comprises a TPEC having a plurality of T-cell epitopes separately conjugated to a targeting moiety comprising the formula T-(L-C-E).sub.n or T-(L-C.sub.i-E.sub.j).sub.n, wherein:
[0023] a. T is a targeting moiety that is capable of targeting unwanted cells;
[0024] b. L is a linker capable of chemical linkage to T;
[0025] c. C is a cleavage site [0026] i. cleaved by an enzyme expressed by the unwanted cells; [0027] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0028] iii. cleaved by a complement-dependent cleavage reaction; or [0029] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and
[0030] b. E is a T-cell epitope
wherein n is an integer of at least 2 (optionally from about 2 to 50), i is an integer of at least 2 (optionally from about 1 to 50), and j is an integer of at least 2 (optionally from about 1 to 50). The "n" integer refers to the number of L-C-E moieties on the targeting moiety and the "i" and "j" integers refer to how many different types of cleavage sites and epitopes are within the construct.
[0031] In some embodiments, a composition for retargeting an immune response to unwanted cells comprises a TPEC with either a (i) linear and/or bundled polytope or a (ii) branched polytope. Such a TPEC may comprise the formula T-L-(C.sub.i-E.sub.j).sub.n, wherein:
[0032] a. T is a targeting moiety that is capable of targeting unwanted cells;
[0033] b. L is an optional linker capable of chemical or peptide linkage to T;
[0034] c. C is a cleavage site [0035] i. cleaved by an enzyme expressed by the unwanted cells; [0036] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0037] iii. cleaved by a complement-dependent cleavage reaction; or [0038] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and
[0039] d. E is a T-cell epitope;
wherein n is an integer of at least 2 (optionally from about 2 to 50), i is an integer of at least 2 (optionally from about 1 to 50), and j is an integer of at least 2 (optionally from about 1 to 50). The "n" integer refers to the number of C-E moieties on the targeting moiety and the "i" and "j" integers refer to how many different types of cleavage sites and epitopes are within the construct.
[0040] Further, in some embodiments, a composition for retargeting an immune response to unwanted cells comprises a TPEC having:
[0041] a. a targeting moiety that is capable of targeting unwanted cells;
[0042] b. a plurality of more than 10 T-cell epitopes conjugated to the targeting moiety with at least one cleavage site, wherein the cleavage site is [0043] i. cleaved by an enzyme expressed by the unwanted cells; [0044] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0045] iii. cleaved by a complement-dependent cleavage reaction; or [0046] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC.
[0047] Additional objects and advantages will be set forth in part in the description which follows, and in part will be obvious from the description, or may be learned by practice. The objects and advantages will be realized and attained by means of the elements and combinations particularly pointed out in the appended claims.
[0048] It is to be understood that both the foregoing general description and the following detailed description are exemplary and explanatory only and are not restrictive of the claims.
[0049] The accompanying drawings, which are incorporated in and constitute a part of this specification, illustrate one (several) embodiment(s) and together with the description, serve to explain the principles described herein.
BRIEF DESCRIPTION OF THE DRAWINGS
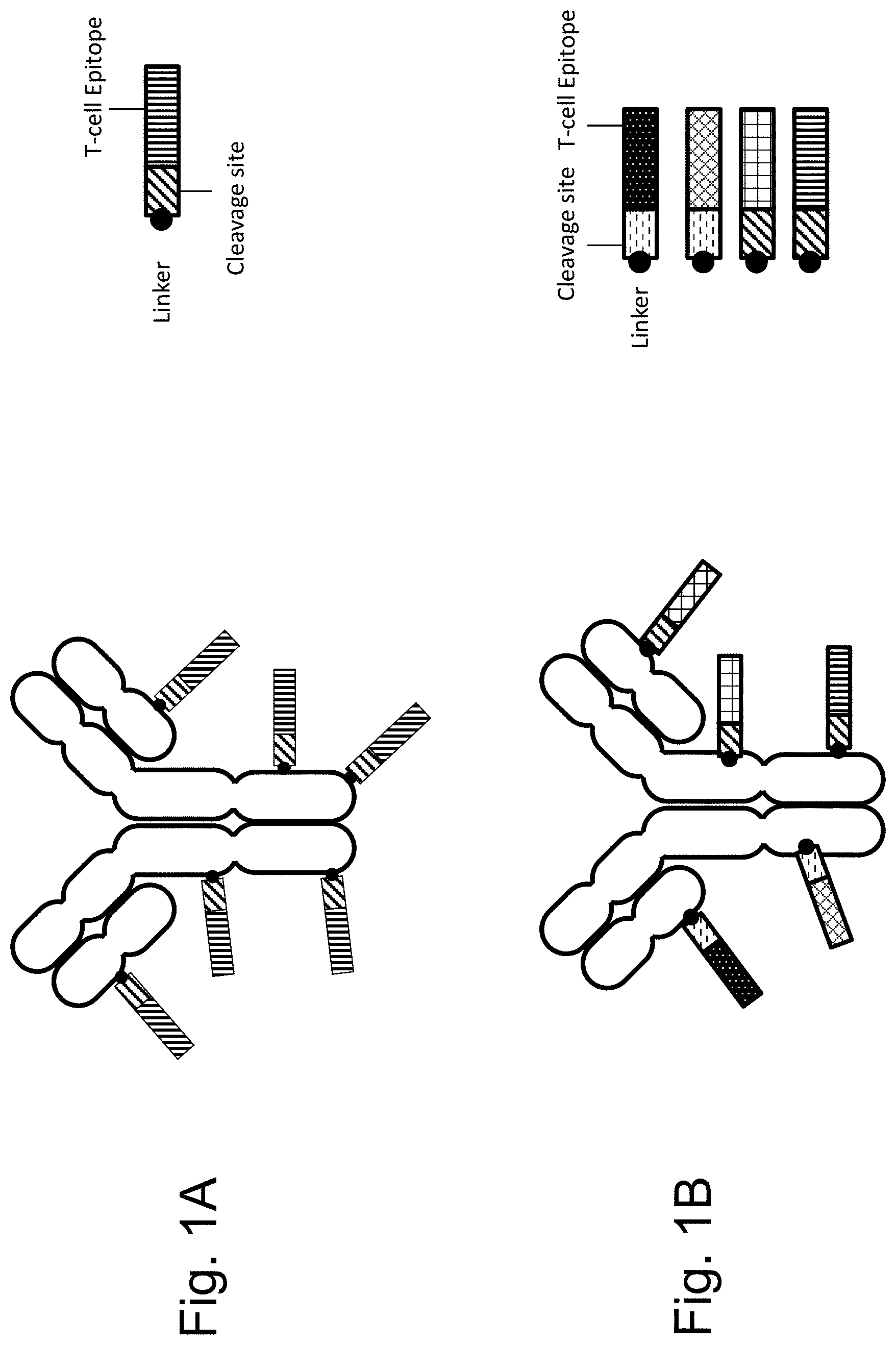
[0050] FIGS. 1A-B show certain embodiments of TPECs. In FIG. 1A, a targeting moiety, optionally an antibody, has a plurality of copies of the same T-cell epitope affixed to it using a chemical linker and a cleavage site. In FIG. 1B, a targeting moiety, optionally an antibody, has a plurality of different T-cell epitopes affixed to it using a chemical linker and a cleavage site. The linker-cleavage site-T-cell-epitopes may be affixed to the targeting moiety in random locations and in random amounts, creating a heterogeneous population of TPECs. In these embodiments, the TPEC comprises a plurality of L-C-E, each attached separately to T, where L is linker, C is cleavage site, E is epitope, and T is targeting moiety.
[0051] FIGS. 2A-C show certain embodiments of TPECs. In FIG. 2A, a targeting moiety, optionally an antibody, has a plurality of different T-cell epitopes affixed to it using a chemical or peptide linker, wherein the plurality of T-cell epitopes is separated by cleavage sites and affixed to the targeting moiety in a bundle. FIG. 2B shows the bundle of T-cell epitopes having immunoglobulin framework regions flanking certain domains. Both FIGS. 2A and 2B include polytopes with a linear configuration. In the embodiments of FIGS. 2A and 2B, the TPEC comprises at least one of L-(C-E)n, with each C-E attached to the L in series (in a bundled and/or linear configuration), and wherein n is an integer of at least 2 (optionally from about 2 to 50) and where L is linker, C is cleavage site, E is epitope, and T is targeting moiety. FIG. 2C shows one embodiment of a TPEC comprising a polytope with a branched configuration. In another embodiment, not shown, the polytope with a branched configuration may have identical T-cell epitopes. In the embodiments of FIG. 2C, the TPEC comprises at least one of L-(C-E)n, with each C-E attached to the L in parallel (in a branched configuration), and wherein n is an integer of at least 2 (optionally from about 2 to 50) and where L is linker, C is cleavage site, E is epitope, and T is targeting moiety.
[0052] FIGS. 3A-C show in vitro activity of re-directed virus specific T cells (Multiple Peptide TPEC approach). Recognition of a lymphoblastoid lymphoma cell line by CD8+ cytomegalovirus-specific cytotoxic T lymphocytes through conjugation of two different viral peptides, RPHERNGFTVL (SEQ ID NO: 2) and NLVPMVATV (SEQ ID NO: 1), to the anti-CD20 antibody Rituximab. Antibody is conjugated using sulfo-SMCC to peptides NLVPMVATVASGV{CIT}GC (SEQ ID NO: 3) and RPHERNGFTVLASGFKGC (SEQ ID NO: 4) at different ratios from 100% NLV:0% RPH through to 0% NLV:100% RPH. Target lymphoma cells labeled with TPEC are recognized by lymphocytes specific towards the NLV peptide (A) at all ratios down to 10% of the conjugated peptide being NLVPMVATVASGV{CIT}GC (SEQ ID NO: 3). Similarly, target lymphoma cells are recognized by lymphocytes specific towards the RPH peptide (B) at all ratios down to 10% of the conjugated peptide being the RPHERNGFTVLASGFKGC (SEQ ID NO: 4). In both (A) and (B), there is no T cell recognition by peptide-specific T cells when the ratio of the conjugated peptide recognized by the T cells is 0%. (C) Overlaid plots of (A) and (B) demonstrating activity of both NLV and RPH specific T cells using Rituximab TPECs conjugated with the two different peptides.
[0053] FIG. 4 illustrates recognition of a lymphoblastoid lymphoma cell line by CD8+ cytomegalovirus-specific cytotoxic T lymphocytes through conjugation of three different viral peptides, VLEETSVML (SEQ ID NO: 5), CRVLCCYVL (SEQ ID NO: 17) and YILEETSVM (SEQ ID NO: 7), to the anti-CD20 antibody Rituximab. Antibody is conjugated using sulfo-SMCC to equal amount of peptides VLEETSVMLASGFKGC (SEQ ID NO: 8), BRVLBBYVLASGFKGC (SEQ ID NO: 9) where B is amino butyric acid, a homolog for cysteine and YILEETSVMASGFKGC (SEQ ID NO: 10). Target lymphoma cells labeled with the TPEC are recognized by T cells specific for each of the three peptides conjugated to the antibody. Untreated target cells are not recognized by any of the peptide-specific T cells (negative control) and free peptide pulsed target cells are strongly recognized by T cells (positive control).
[0054] FIG. 5 demonstrates recognition of a lymphoblastoid lymphoma cell line by CD8+ cytomegalovirus-specific cytotoxic T lymphocytes through conjugation of polytope peptides. Antibody is conjugated using sulfo-SMCC to peptides
(i) CGVANLVPMVATVAVAVLEETSVML (SEQ ID NO: 11),
(ii) CVARPHERNGFTVLVANLVPMVATV (SEQ ID NO: 12) and
[0055] (iii) CGVANLVPMVATVARPHERNGFTVL (SEQ ID NO: 13). Target lymphoma cells labeled with TPEC are recognized by T cells specific for the NLVPMVATV (SEQ ID NO: 1) peptide. Untreated target cells are not recognized by the peptide-specific T cells (negative control) and free peptide pulsed target cells are strongly recognized by T cells (positive control).
[0056] FIGS. 6A-B demonstrate the protein conjugation chemistry of one embodiment of the branched peptide using a stem peptide that contains four azidonorleucine residues and peptides (branches) that contain propargyl glycine at the amino terminus (FIG. 6A). Incubation of equimolar concentrations of the branches with the stem peptide in DMSO in the presence of 10 mg in 4 ml of the CuSO.sub.45 H2O catalyst mixed with 10 mg in 4 ml ascorbic acid overnight resulted in the formation of the branched peptide used for conjugation. The final concentration of DMSO for the reaction was 50%. The branched peptides were purified using HPLC and verified using mass spectrometry (FIG. 6B). The peptides contain an N-terminal amide group (-nh2) which is useful in peptide stability compared with the usual carboxyl group (--COOH).
[0057] FIGS. 7A-B demonstrate recognition of a lymphoblastoid lymphoma cell line by CD8+ cytomegalovirus-specific cytotoxic T lymphocytes through conjugation of polytope peptides in the branched peptide format. Antibody is conjugated using the embodiment in FIG. 6B using the following peptides:
TABLE-US-00001 Stem: (SEQ ID NO: 168) SEEZSEEZSEEZSEEZ (Z: Azidonorleucine); For Branch 1 TPEC: branch 1-1: (SEQ ID NO: 169) BKPAKFFRLTPRVTGGGAM-nh2 (B: propargyl glycine); Branch 1-2: (SEQ ID NO: 170) BKPAKFFRLRPHERNGFTVL-nh2 (B: propargyl glycine); Branch 1-3: (SEQ ID NO: 171) BKPAKFFRLRELRRKMMYM-nh2 (B: propargyl glycine); and Branch 1-4: (SEQ ID NO: 172) BKPAKFFRLNLVPMVATV-nh2 (B: propargyl glycine); and For Branch 3 TPEC, branch 3-1: (SEQ ID NO: 173) BAIPVSLRTPRVTGGGAM-nh2 (B: propargyl glycine); Branch 3-2: (SEQ ID NO: 174) BAIPVSLRRPHERNGFTVL-nh2 (B: propargyl glycine); Branch 3-3: (SEQ ID NO: 175) BAIPVSLRELRRKMMYM-nh2 (B: propargyl glycine); and Branch 3-4: (SEQ ID NO: 176) BAIPVSLVTEHDTLLY-nh2 (B: propargyl glycine).
[0058] Target lymphoma cells labeled with branched peptide TPEC are recognized by T cells specific for the RPHERNGFTVL (SEQ ID NO: 2) and TPRVTGGGAM (SEQ ID NO: 49) peptides. Untreated target cells are not recognized by the peptide-specific T cells (negative control) and free peptide pulsed target cells are strongly recognized by T cells (positive control).
[0059] FIGS. 8A-B demonstrate recognition of an ovarian carcinoma cell line by CD8+ cytomegalovirus-specific cytotoxic T lymphocytes through conjugation of polytope peptides in the branched peptide format. Antibody is conjugated using the embodiment in FIG. 6B using peptides
TABLE-US-00002 Stem: (SEQ ID NO: 168) SEEZSEEZSEEZSEEZ (Z: Azidonorleucine); For Branch 1 TPEC, branch 1-1: (SEQ ID NO: 169) BKPAKFFRLTPRVTGGGAM-nh2 (B: Propargyl glycine); Branch 1-2: (SEQ ID NO: 170) BKPAKFFRLRPHERNGFTVL-nh2 (B: Propargyl glycine); Branch 1-3: (SEQ ID NO: 171) BKPAKFFRLRELRRIM-nh2 (B: Propargyl glycine); Branch 1-4: (SEQ ID NO: 172) BKPAKFFRLNLVPMVATV-nh2 (B: Propargyl glycine); and For Branch 3 TPEC, branch 3-1: (SEQ ID NO: 173) BAIPVSLRTPRVTGGGAM-nh2 (B: Propargyl glycine); Branch 3-2: (SEQ ID NO: 174) BAIPVSLRRPHERNGFTVL-nh2 (B: Propargyl glycine); Branch 3-3: (SEQ ID NO: 175) BAIPVSLRELRRIM-nh2 (B: Propargyl glycine); and Branch 3-4: (SEQ ID NO: 176) BAIPVSLVTEHDTLLY-nh2 (B: Propargyl glycine).
[0060] Target lymphoma cells labeled with branched peptide TPEC are recognized by T cells specific for the RPHERNGFTVL (SEQ ID NO: 2) and TPRVTGGGAM (SEQ ID NO: 49) peptides. Untreated target cells are not recognized by the peptide-specific T cells (negative control) and free peptide pulsed target cells are strongly recognized by T cells (positive control).
[0061] FIGS. 9A-C provide an embodiment of the bundled domain TPEC. FIG. 9A demonstrates the scaffold (Fibronectin type 3 domain from the fibronectin 1 protein) used for the bundled domain TPEC. The bundled domain sequence (FIG. 9A) demonstrates the mutations made in the outer loops of the domain and contains viral epitopes separated from the fibronectin sequence on both sides by a protease recognition sequence specific for MMP2. The protease cleavage sites are underlined and the viral epitope are shown in bold and italics. FIG. 9B demonstrates the structure of the bundled domain and the "BC", "DE" and "FG" loops that contain the viral epitopes. FIG. 9C demonstrates recognition of an ovarian carcinoma cell line by CD8+ cytomegalovirus specific cytotoxic T lymphocytes. CMV epitopes are contained within an additional protein domain linked to the heavy chain of Cetuximab. The viral epitopes are flanked by an MMP2 protease recognition sequence which will allow release of the viral epitopes in the presence of MMP2, produced by the target cell line. The bundled domain TPEC is produced in Expi293 cells and purified using protein A sepharose beads.
[0062] Target ovarian carcinoma cells labelled with the bundled domain TPEC are recognized by T cells specific for the viral epitope NLVPMVATV (SEQ ID NO: 1) by release of IFN-.gamma.. Four different preparations (two TPECs produced on each of two days using different concentrations of DNA) of the bundled domain were used, all of which produced similar amounts of bundled domain TPEC and produced similar recognition of target cells by the peptide-specific T cells. Untreated target cells are not recognized by the peptide-specific T cells (negative control) and free peptide pulsed target cells are strongly recognized by T cells (positive control).
DESCRIPTION OF THE SEQUENCES
[0063] Table 1 provides a listing of certain sequences referenced herein.
TABLE-US-00003 TABLE 1 Description of the Sequences and SEQ ID NOs Description Sequence # T-cell description from NLVPMVATV 1 cytomegalovirus (CMV TCE) (pp65, position 495-503, HLA-A2) CMV TCE (pp65, position 265-275, RPHERNGFTVL 2 HLA-B7) CMV TCE and cleavage site from NLVPMVATVASGV{Cit}GC 3 capthepsin B cleavage site in bold. {Cit} = citrulline CMV TCE and cleavage site from RPHERNGFTVLASGFKGC 4 capthepsin B cleavage site in bold CMV TCD (IE1, position 316-324, VLEETSVML 5 HLA-A2) CMV TCE BRVLBBYVL, where B is 6 (IE1, 309-317, HLA-C7) alpha-aminobutyric acic, (Peptidomimetic of SEQ ID NO: 17) a homolog for cysteine CMV TCE (IE1, 315-323, HLA-A2) YILEETSVM 7 CMV TCE and cleavage site from VLEETSVMLASGFKGC 8 capthepsin B cleavage site in bold CMV TCE and cleavage site from BRVLBBYVLASGFKGC, where 9 capthepsin B B is alpha-aminobutyric acid cleavage site in bold CMV TCE and cleavage site from YILEETSVMASGFKGC 10 capthepsin B cleavage site in bold Polytope containing two different CGVANLVPMVATVAVAVLEETSVML 11 CMV-derived epitopes separated by cleavage sites in bold cathepsin B cleavage site Polytope containing two different CVARPHERNGFTVLVANLVPMVATV 12 CMV-derived epitopes separated by cleavage site in bold cathepsin B cleavage site Polytope containing two different CGVANLVPMVATVARPHERNGFTVL 13 CMV-derived epitopes separated by cleavage site in bold cathepsin B cleavage site Polytope containing five different CGSFRVTEHDTLLYGSFRRPHERNGFTVLG 14 CMV-derived epitopes separated by SFRELKRKMIYMGSFRNLVPMVATV cathepsin B cleavage sites cleavage sites in bold CMV TCE (IE1, 32-41, HLA-A68 ATTFLQTMLR 15 CMV TCE (pp65, 103-114, HLA-B35) CPSQEPMSIYVY 16 CMV TCE (IE1, 309-317, HLA-C7) CRVLCCYVL 17 CMV TCE (IE1, 279-287, HLA-A1/B18) CVETMCNEY 18 CMV TCE (IE1, 198-206, HLA-A1/B18) DELRRKMMY 19 CMV TCE (pp65, 31-40, HLA-A68) DTPVLPHETR 20 CMV TCE (IE1, 381-389, HLA-B44) EEAIVAYTL 21 CMV TCE (pp65, 512-521, HLA-B44) EFFDANDIY 22 CBV TCE (IE1, 199-207, HLA-B8) ELKRKMIYM 23 CMV TCE (IE1, 199-207, HLA-B8) ELRRKMMYM 24 CMV TCE (IE1, 334-342, HLA-A68) EVISVMKRR 25 CMV TCE (IE2, 381-389, HLA-B41) FEQQPTETPP 26 CMV TCE (IE1, 221-231, HLA-B55) FPKTTNGCSQA 27 CMV TCE (pp65, 188-195, HLA-B35) FPTKDVAL 28 CMV TCE (pp65, 369-379, HLA-A24) FTSQYRIQGKL 29 CMV TCE (pp65, 186-196, HLA-A68) FVFPTKDVALR 30 CMV TCE (IE2, 242-250, HLA-A2) IIYTRNHEVK 31 CMV TCE (pp65, 123-131, HLA-B35) IPSINVHHY 32 CMV TCE (pp150, 101-109, HLA-B7) KARDHLAVL 33 CMV TCE (IE1, 42-50, HLA-B40) KEVNSQLSL 34 CMV TCE (IE1, 201-209, HLA-B27) KRKMIYMCY 35 CMV TCE (pp65, 120-128, HLA-A2) MLNIPSINV 36 CMV TCE (pp150, 212-220, HLA-B7) NVRRSWEEL 37 CMV TCE (IE1, 88-96, HLA-B8) QIKVRVDMV 38 CMV TCE (pp65, 52-61, HLA-B35) QPSLILVSQY 39 CMV TCE (pp150, 792-802, HLA-A68) QTVTSTPVQGR 40 CMV TCE (pp65, 341-349, HLA-A24) QYDVPAALF 41 CMV TCE (pp65, 222-231, HLA-A24) QYVKVYLESF 42 CMV TCE (pp50, 274-282, HLA-A1) RGDPFDKNY 43 CMV TCE (pp65, 522-530, HLA-A2) RIFAELEGV 44 CMV TCE (gB, 4-12, HLA-A2) RIWCLVVCV 45 CMV TCE (IE1, 341-349, HLA-B27) RRIEEICMK 46 CMV TCE (IE1, 201-209, HLA-B27) RRKMMYMCY 47 CMV TCE (pp65, 364-373, HLA-B44) SEHPTFTSQY 48 CMV TCE (pp65, 417-426, HLA-B7) TPRVTGGGAM 49 CMV TCE (pp150, 945-955, HLA-A3) TTVYPPSSTAK 50 CMV TCE (pp50, 52-60, HLA-A3) TVRSHCVSK 51 CMV TCE (IE1, 81-89, HLA-A2) VLAELVKQI 52 CMV TCE (pp50, 245-253, HLA-A1) VTEHDTLLY 53 CMV TCE (pp65, 113-121, HLA-A24) VYALPLKML 54 CMV TCE (pp65, 363-373, HLA-A1) YSEHPTFTSQY 55 CMV TCE (pp65, 61-70, HLA-A68) YTPDSTPCHR 56 CMV TCE (IE1, 316-324, HLA-A2) YVLEETSVM 57 CMV TCE and cleavage site from ADAM28 CKPAKFFRLNLVPMVATV 58 CMV TCE and cleavage site from ADAM28 CKPAKFFRLRPHERNGFTVL 59 CMV TCE and cleavage site from capthepsin D CPRSFFRLGKVLEETSVML 60 CMV TCE and cleavage site from ADAM28 CKPAKFFRLELKRKMIYM 61 CMV TCE and cleavage site from capthepsin D CPRSFFRLGKQIKVRVDMV 62 Polytope comprising four different CGSKPAKFFRLYSEHPTFTSQYGSPRSFFR 63 cytomegalovirus epitopes, each separated by LGKTPRVTGGGAMGSKPAKFFRLQIKVRVD either and ADAM28 or cathepsin D cleavage MVGSPRSFFRLGKELRRKMMYM site T-cell epitope from Epstein Barr Virus RPQKRPSCI 64 (EBV TCE) EBNA1 EBV TCE EBNA1 HPVGEADYF 65 EBV TCE EBNA1 HPVGEADYFEY 66 EBV TCE EBNA1 IPQCRLTPL 67 EBV TCE EBNA1 VLKDAIKDL 68 EBV TCE EBNA2 YHLIVDTDSL 69 EBV TCE EBNA2 DTPLIPLTIF 70 EBV TCE EBNA2 RPTELQPTP 71 EBV TCE EBNA3A QAKWRLQTL 72 EBV TCE EBNA3A AYSSWMYSY 73 EBV TCE EBNA3A RYSIFFDY 74 EBV TCE EBNA3A FLRGRAYGL 75 EBV TCE EBNA3A KRPPIFIRRL 76 EBV TCE EBNA3A RPPIFIRRL 77 EBV TCE EBNA3A LEKARGSTY 78 EBV TCE EBNA3A HLAAQGMAY 79 EBV TCE EBNA3A YPLHEQHGM 80 EBV TCE EBNA3A VFSDGRVAC 81 EBV TCE EBNA3A VPAPAGPIV 82 EBV TCE EBNA3A SVRDRLARL 83 EBV TCE EBNA3A RLRAEAQVK 84 EBV TCE EBNA3A VQPPQLTLQV 85 EBV TCE EBNA3B HRCQAIRKK 86 EBV TCE EBNA3B TYSAGIVQI 87 EBV TCE EBNA3B RRARSLSAERY 88 EBV TCE EBNA3B VSFIEFVGW 89 EBV TCE EBNA3B AVFDRKSDAK 90 EBV TCE EBNA3B IVTDFSVIK 91 EBV TCE EBNA3B AVLLHEESM 92 EBV TCE EBNA3B VEITPYKPTW 93 EBV TCE EBNA3C EGGVGWRHW 94 EBV TCE EBNA3C QNGALAINTF 95 EBV TCE EBNA3C LRGKWQRRYR 96 EBV TCE EBNA3C RRIYDLIEL 97 EBV TCE EBNA3C HHIWQNLL 98 EBV TCE EBNA3C EENLLDFVRF 99 EBV TCE EBNA3C LLDFVRFMGV 100 EBV TCE EBNA3C LDFVRFMGV 101 EBV TCE EBNA3C KEHVIQNAF 102 EBV TCE EBNA3C FRKAQIQGL 103 EBV TCE EBNA3C QPRAPIRPI 104 EBV TCE EBNA-LP SLREWLLRI 105 EBV TCE LMP1 FWLYIVMSD 106 EBV TCE LMP1 FRRDLLCPLGA 107 EBV TCE LMP1 YLLEMLWRL 108 EBV TCE LMP1 YLQQNWWTL 109
EBV TCE LMP1 TLLVDLLWL 110 EBV TCE LMP1 DPHGPVQLSYYD 111 EBV TCE LMP2 MGSLEMVPM 112 EBV TCE LMP2 EDPYWGNGDRHSDYQ 113 EBV TCE LMP2 NPVCLPVIVAPYLF 114 EBV TCE LMP2 LPVIVAPYL 115 EBV TCE LMP2 PYLFWLAAI 116 EBV TCE LMP2 ASCFTASVSTVVTA 117 EBV TCE LMP2 FTASVSTVV 118 EBV TCE LMP2 IEDPPFNSL 119 EBV TCE LMP2 RRRWRRLTV 120 EBV TCE LMP2 RRWRRLTVC 121 EBV TCE LMP2 RRLTVCGGIMF 122 EBV TCE LMP2 TVCGGIMFL 123 EBV TCE LMP2 MFLACVLVLIVDAV 124 EBV TCE LMP2 LIVDAVLQL 125 EBV TCE LMP2 GLGTLGAAI 126 EBV TCE LMP2 LLWTLVVLL 127 EBV TCE LMP2 SSCSSCPLSKI 128 EBV TCE LMP2 ILLARLFLY 129 EBV TCE LMP2 FLYALALLL 130 EBV TCE LMP2 TYGPVFMCL 131 EBV TCE LMP2 CLGGLLTMV 132 EBV TCE LMP2 VMSNTLLSAW 133 EBV TCE LMP2 LTAGFLIFL 134 EBV TCE LMP2 LLSAWILTA 135 EBV TCE BRLF1 LVSDYCNVLNKEFT 136 EBV TCE BRLF1 LVSDYCNVL 137 EBV TCE BRLF1 DYCNVLNKEF 138 EBV TCE BRLF1 AENAGNDAC 139 EBV TCE BRLF1 IACPIVMRYYVLDHLI 140 EBV TCE BRLF1 YVLDHLIVV 141 EBV TCE BRLF1 FFIQAPSNRVMIPAT 142 EBV TCE BRLF1 ATIGTAMYK 143 EBV TCE BRLF1 KHSRVRAYTYSKVLG 144 EBV TCE BRLF1 RALIKTLPRASYSSH 145 EBV TCE BRLF1 ERPIFPHPSKPTFLP 146 EBV TCE BRLF1 QKEEAAICGQMDLS 147 EBV TCE BRLF1 EVCQPKRIRPFHPPG 148 EBV TCE BZLF1 LPEPLPQGQLTAY 149 EBV TCE BZLF1 EPLPQGQLTAY 150 EBV TCE BZLF1 APENAYQAY 151 EBV TCE BZLF1 LQHYREVAA 152 EBV TCE BZLF1 DSELEIKRYKNR 153 EBV TCE BZLF1 RKCCRAKFKQLLQHYR 154 EBV TCE BZLF1 RAKFKQLL 155 EBV TCE BZLF1 SENDRLRLL 156 T-cell epitope from Influenza A CTELKLSDY 157 Virus (IAV TCE) PR8 IAV TCE PB1 VSDGGPNLY 158 IAV TCE MP GILGFVFTL 159 IAV TCE MP ILGFVFTLTV 160 IAV TCE PR8 ILRGSVAHK 161 IAV TCE MP SIIPSGPLK 162 IAV TCE MP1 RMVLASTTAK 163 IAV TCE MP2 KSMREEYRK 164 IAV TCE NP SPIVPSFDM 165 IAV TCE PB1 QPEWFRNVL 166 IAV TCE NP SRYWAIRTR 167 Stem from branched peptide SEEZSEEZSEEZSEEZ, 168 wherein Z is Azidonorleucine Branch 1-1 for branched peptide BKPAKFFRLTPRVTGGGAM-nh2, 169 wherein B is propargyl glycine Branch 1-2 for branched peptide BKPAKFFRLRPHERNGFTVL-nh2, 170 wherein B is propargyl glycine Branch 1-3 for branched peptide BKPAKFFRLRELRRKMMYM-nh2, 171 wherein B is propargyl glycine Branch 1-4 for branched peptide BKPAKFFRLNLVPMVATV-nh2, 172 wherein B is propargyl glycine Branch 3-1 for branched peptide BAIPVSLRTPRVTGGGAM-nh2, 173 wherein B is propargyl glycine Branch 3-2 for branched peptide BAIPVSLRRPHERNGFTVL-nh2, 174 wherein B is propargyl glycine Branch 3-3 for branched peptide BAIPVSLRELRRKMMYM-nh2, 175 wherein B is propargyl glycine Branch 3-4 for branched peptide BAIPVSLVTEHDTLLY-nh2, 176 wherein B is propargyl glycine C1s cleavage site YLGRSYKV 177 C1s cleavage site MQLGRX 178 MASP2 cleavage site SLGRKIQI 179 C2a cleavage site GLARSNLDE 180 Cathepsin D cleavage site PRSFFRLGK 181 ADAM28 cleavage site KPAKFFRL 182 ADAM28 cleavage site DPAKFFRL 183 ADAM28 cleavage site KPMKFFRL 184 ADAM28 cleavage site LPAKFFRL 185 MMP2 cleavage site AIPVSLR 186 187 Bundled domain sequence SASGGGSGGGSVSDVPRDLEVVAATPTSLL 188 ISWDAPAVGGGGSGGGGSTIPVSLRSTPRV TGGAMTIPVSLRSGGGGSGGGGSTVRYYRI TYGETGGNSPVQEFTVPGSKSTATISGLKP GVDYTITVYAVTGGGGGSGGGGSTIPVSLR SNLVPMVATVTIPVSLRSGGGGSGGGGSSP ASSKPISINYRTGGGGSGGGGSTIPVSLRS VLEETSVML MMP2 cleavage site TIPVSLRS 188
DESCRIPTION OF THE EMBODIMENTS
[0064] I. Targeting Moiety Peptide Epitope Complexes Having A Plurality of T-Cell Epitopes
[0065] A variety of targeting moiety peptide epitope complexes (TPECs) are described in different embodiments. In each of the embodiments, however, a targeting moiety may be used to deliver the TPEC to an area of unwanted cells, allowing for a therapeutic effect to be delivered locally. The TPEC also contains a plurality of T-cell epitopes. The TPEC further comprises cleavage sites that release the T-cell epitopes from the targeting agent, and in some embodiments from each other, when they are in the microenvironment of the unwanted cells. Although the arrangement and number of T-cell epitopes varies in different embodiments described herein, once cleaved from the targeting agent (and any neighboring T-cell epitopes), the T-cell epitopes function by stimulating an immune response against the unwanted cells. In some embodiments, maximal benefits may be achieved by releasing all of the T-cell epitopes from both the targeting agent and from each other in the cleavage process, allowing each T-cell epitope to attract an immune response to the unwanted cell.
[0066] Having a plurality of T-cell epitopes, as discussed in detail below, enhances the immune response against the unwanted cells, either by stimulating a stronger immune response in a given patient or by allowing the TPEC to stimulate an immune response across a wide variety of patients in different ethnic and racial groups. In one embodiment, and while not being bound by theory, the cleavage at the cleavage site allows the T-cell epitopes to be trimmed at either or both ends to the appropriate length to produce a peptide that can fit in the peptide-binding groove of HLA class I and be recognized by T-cells, so as to initiate an immune response.
[0067] Without cleavage of these epitopes, the prior art fails to stimulate a sufficient T-cell response because the T-cell epitopes fused together would not be adequately recognized by the patients T-cells and would, thus, not initiate an immune response against the cancer cells.
[0068] A. TPECs with a Plurality of T-Cell Epitopes Separately Conjugated
[0069] In one embodiment, the TPEC comprises a plurality of T-cell epitopes that are separately conjugated to the targeting moiety. Because the T-cell epitopes will be separately conjugated to the targeting moiety, with two exemplary embodiments shown in FIGS. 1A and 1B, this embodiment may be termed the "hairy" embodiment as the T-cell epitopes protrude from the targeting moiety like individual hairs from a person's skin. In other words, in some embodiments, a composition for retargeting an immune response to unwanted cells may comprise a TPEC comprising the formula T-(L-C-E).sub.n. In such embodiments, a plurality of L-C-E, each attach separately to T, wherein:
[0070] (a) T is a targeting moiety that is capable of targeting unwanted cells;
[0071] (b) L is a linker capable of chemical linkage to T;
[0072] (c) C is a cleavage site that is (i) cleaved by an enzyme expressed by the unwanted cells; (ii) cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; (iii) cleaved by a complement-dependent cleavage reaction; or (iv) cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and
[0073] (d) E is a T-cell epitope.
[0074] N is an integer of at least 2 (optionally from about 2 to 50). The same cleavage site may optionally be used multiple times in the TPEC and the same epitope may optionally be used multiple times in the TPEC.
[0075] The C and E moieties may be the same in the plurality of L-C-E moieties affixed to a single T or a single T may have different C and E moieties in the plurality of L-C-E moieties. Thus, there may be more than one type of cleavage site or only one type of cleavage site. There may also, independently, be more than one type of T-cell epitope or only one type of T-cell epitope.
[0076] In certain aspects of the embodiments discussed in this section, the L-C-E complexes may be affixed to the targeting moiety in a random fashion. Thus, in a preparation of TPECs with a plurality of T-cell epitopes separately conjugated to the targeting moiety, some TPECs may have greater numbers of T-cell epitopes and some may have fewer numbers. Additionally, in a preparation of TPECs with a plurality of T-cell epitopes separately conjugated to the targeting moiety, the location of TPECs may differ throughout the preparation. In this vein, when these compounds are administered to a patient suffering from a disease such as cancer, the patient's body will have a much more difficult time mounting an inhibitory immune response against the preparation of TPECs. This may allow the patient to receive multiple doses of the TPEC preparation over a significant period of time, as discussed further below in Section III.B below.
[0077] B. TPECs with at Least One Polytope
[0078] In another embodiment, the TPEC comprises a plurality of T-cell epitopes that are conjugated to the targeting moiety as a polytope (for poly-epitope), but the conjugation within the polytope and to the targeting moiety occurs through cleavage sites allowing the release of the individual T-cell epitopes in the microenvironment of the unwanted cells. Several embodiments of this arrangement are shown in FIGS. 2A, 2B, and 2C. Thus, the polytope itself can have a linear and/or bundled configuration (like FIGS. 2A and 2B) or a branched configuration like a hairbrush with bristles (like FIG. 2C).
[0079] In FIGS. 2A-B, a composition for retargeting an immune response to unwanted cells comprises a TPEC having at least one of L-(C-E)n with each C-E attached to the L in series and the L attached to the T. It may have one or a plurality of L-(C-E)n attached to the T. In the embodiments of FIGS. 2A-B, the linker may be either a chemical linker, a peptide bond, or at least one peptide.
[0080] In FIG. 2C, a composition for retargeting an immune response to unwanted cells comprises at TPEC having at least one of L-(C-E)n with each C-E attached to the L in parallel and the L attached to the T. It may have one or a plurality of L-(C-E)n attached to the T. In embodiment 2C, the linker may be a chemical linker
[0081] In all of the embodiments of FIG. 2A-C,
[0082] (a) T is a targeting moiety that is capable of targeting unwanted cells;
[0083] (b) L is a linker capable of chemical or peptide linkage to T (including a peptide bond);
[0084] (c) C is a cleavage site (i) cleaved by an enzyme expressed by the unwanted cells; (ii) cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; (iii) cleaved by a complement-dependent cleavage reaction; or (iv) cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and
[0085] (d) E is a T-cell epitope;
[0086] wherein n is an integer of at least 2 (optionally from about 2 to 50).
[0087] The C and E moieties may be the same in the plurality of C-E moieties affixed through L to a single T or a single T may have different C and E moieties in the plurality of C-E moieties. Thus, there may be more than one type of cleavage site or only one type of cleavage site. There may also, independently, be more than one type of T-cell epitope or only one type of T-cell epitope.
[0088] In some embodiments, the plurality of the T-cell epitopes being conjugated in a polytope allow for a uniform product to be produced that contains a standard group, number, and arrangement of T-cell epitopes, while still allowing for the release of all of the T-cell epitopes in the microenvironment of the unwanted cells. Microenvironment means the specific set of physical, chemical, and biological conditions in the vicinity of cells within a distance where these conditions can have an effect on or be sensed by the cells.
[0089] In some embodiments, more than one polytope are conjugated to the targeting moiety. In some embodiments from about 1 to 30, 1 to 20, or 1 to 10 polytopes are conjugated to the targeting moiety. In some embodiments, about 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 polytopes are conjugated to the targeting moiety, each group by one cleavage site. In these embodiments, the polytopes may be the same or different.
[0090] In some embodiments, the TPEC may have both at least one separately conjugated T-cell epitope and at least one polytope, according to sections I.A and I.B, respectively.
[0091] C. TPECs with More Than 10 T-Cell Epitopes
[0092] In another embodiment, the TPEC comprises a plurality of T-cell epitopes that, irrespective of how they are arranged on the targeting moiety, comprise more than 10 T-cell epitopes. In these embodiments, the T-cell epitopes may be separately conjugated or conjugated in a polytope. In other words, a composition for retargeting an immune response to unwanted cells may comprise a TPEC having:
[0093] (a) a targeting moiety that is capable of targeting unwanted cells;
[0094] (b) a plurality of more than 10 T-cell epitopes conjugated to the targeting moiety with at least one cleavage site, wherein the cleavage site is (i) cleaved by an enzyme expressed by the unwanted cells; (ii) cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; (iii) cleaved by a complement-dependent cleavage reaction; or (iv) cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC.
[0095] In some embodiments, including TPECs with more than 10 T-cell epitopes, the immune response against the unwanted cells is notably stronger. In some embodiments, including TPECs with more than 10 T-cell epitopes, the TPEC can be used to treat a greater proportion of patients suffering from the condition characterized by unwanted cells.
[0096] In some compositions with TPECs having more than 10 T-cell epitopes, the T-cell epitopes may be separately conjugated to the targeting moiety, each T-cell epitope by a cleavage site. In some embodiments, with TPECs having more than 10 T-cell epitopes, the T-cell epitopes may be conjugated to the targeting moiety as at least one polytope, each polytope conjugated to the targeting moiety by a cleavage site. In some embodiments, the T-cell epitopes within a polytope have cleavage sites between them.
[0097] D. Compositions Comprising TPECs
[0098] A composition may comprise a plurality of TPECs. In some embodiments, all of the TPECs in the composition are the same. In some embodiments, at least some of the TPECs in the composition are not identical.
[0099] In some embodiments, each TPEC is conjugated to a plurality of identical T-cell epitopes.
[0100] In some embodiments, at least some of the TPECs in the composition are conjugated to a plurality of T-cell epitopes that are not identical.
[0101] In some embodiments, a composition may comprise at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, or more different TPECs.
[0102] E. T-Cell Epitopes
[0103] T-cell epitopes function in the TPECs to attract the patient's own immune response to attack the unwanted cell by labeling it with antigenic T-cell epitopes from infectious agents.
[0104] Depending on desired aspects of the therapy, a variety of optional factors may be considered when choosing T-cell epitopes. Every person has six HLA class-I molecules that can bind peptides to present to CD8 T cells with some HLA types more prevalent in the population than others, such as HLA-A201 which is found in .about.45% of the Caucasian population. In situations where the epitopes chosen for conjugation are able to bind to a limited set of HLA molecules that are only found in a proportion of the population, the TPEC may not have any or as much effectiveness in other segments of the population. Using HLA-A201 as an example, if the targeting moiety is conjugated with a T-cell epitope that binds to HLA-A201 then only patients that express HLA-A201 would bind the epitope and present it to T cells and initiate an immune response. However, in patients who are HLA-A201 negative the epitope may not be able to bind to their HLA molecules and there would be no or a lesser immune response. In situations where the therapy may be provided to a wide segment of the population having different HLA molecules, using different epitopes that bind to more HLA molecules could enhance the effectiveness of the TPEC across the population.
[0105] As an additional factor to consider, in some embodiments, T-cell epitopes may be chosen from those that a particular person has been exposed to, a wide variety of people have been exposed to, or for which vaccines have or can be administered. T cells generally recognize the epitope in complex with an HLA molecule when the patient has been previously infected with the virus from which the epitope is derived (or in instances where the patient has previously received a vaccine containing those epitopes). Vaccines may have been administered for a prior purpose (such as childhood vaccines) or may be administered preceding TPEC treatment with the same epitope.
[0106] In certain instances, T-cell epitopes are chosen from cytomegalovirus (CMV), influenza, Epstein Barr virus (EBV), varicella zoster, mumps, measles, rubella, adenovirus, polio, vaccinia, RSV, rotavirus, tetanus, vaccinia, and yellow fever T-cell epitopes. Epitopes may be chosen from infectious agents that are prevalent across the population in question or for which there are vaccines that are regularly administered or could be administered as part of a combination therapy approach.
[0107] In certain embodiments, the T-cell epitopes are chosen from at least 2, 3, 4, or 5 different infectious agents. In certain embodiments, at least some of the T-cell epitopes are CMV epitopes.
[0108] In any of the various TPECs described herein, in some embodiments, the plurality of T-cell epitopes are not all identical. In some embodiments, the plurality of T-cell epitopes are the same. In certain embodiments, the plurality of T-cell epitopes comprise some that are the same and some that are different.
[0109] The T-cell epitopes may be all MEC Class I restricted peptides, all MEC class II restricted peptides, or a combination of both Class I and Class II.
[0110] In some embodiments, the plurality of T-cell epitopes are from about 7 to 14 amino acids in length, from about 8 to 13, from about 9 to 12, about 9, or about 10 amino acids.
[0111] In some embodiments, at least 2, 3, 4, 5, 6, 7, 8, 9, or 10 different T-cell epitopes are used in the TPECs, whether the TPECs are the same or different. In some embodiments, using a plurality of different T-cell epitopes allows the agent to stimulate a T-cell response in at least about 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 60%, 70%, 80%, or 90% of the human population.
[0112] In some embodiments, the T-cell epitopes are chosen from HLA-A, HLA-B, HLA-C, HLA-E, HLA-F, HLA-G, CD1d, and MR1. In certain embodiments, the T-cell epitopes are chosen from HLA-A*01, HLA-A*02, HLA-A*03, HLA-A*11, HLA-A*24, HLA-B*44, HLA-B*07, HLA-B*08, HLA-B*15, HLA-B*35, HLA-B*40, HLA-C*07, HLA-C*03, HLA-C*05, HLA-C*04, HLA-C*06, and HLA-E*0101 restricted antigens.
[0113] In certain embodiments, the composition comprises at least the following T-cell epitopes HLA-A*02, HLA-A*01, and HLA-A*03.
[0114] When desired, more immunodominant T-cell epitopes may be selected. By immunodominant, we mean those epitopes that elicit the strongest immune response in a given patient and/or those that are known to be immunogenic across a wide cross-section of people.
[0115] In some embodiments, the T-cell epitopes comprise at least one of the epitopes provided in Table 2. In some embodiments, the T-cell epitopes are chosen from the epitopes provided in Table 2.
TABLE-US-00004 TABLE 2 Exemplary T-Cell Epitopes Peptide from Sequence Position HLA Type Cytomegalovirus Epitopes IE1 ATTFLQTMLR (SEQ ID NO: 15) 32-41 HLA-A68 IE1 BRVLBBYVL (SEQ ID NO: 6) 309-317 HLA-C7 (variant of SEQ ID NO: 17, where the C residues are changed to B residues, wherein B is alpha- aminobutyric acid) pp65 CPSQEPMSIYVY (SEQ ID NO: 16) 103-114 HLA-B35 IE1 CRVLCCYVL (SEQ ID NO: 17) 309-317 HLA-C7 IE1 CVETMCNEY (SEQ ID NO: 18) 279-287 HLA-A1/B18 IE1 DELRRKMMY (SEQ ID NO: 19) 198-206 HLA-A1/B18 pp65 DTPVLPHETR (SEQ ID NO: 20) 31-40 HLA-A68 IE1 EEAIVAYTL (SEQ ID NO: 21) 381-389 HLA-B44 pp65 EFFDANDIY (SEQ ID NO: 22) 512-521 HLA-B44 IE1 ELKRKMIYM (SEQ ID NO: 23) 199-207 HLA-B8 IE1 ELRRKMMYM (SEQ ID NO: 24) 199-207 HLA-B8 IE1 EVISVMKRR (SEQ ID NO: 25) 334-342 HLA-A68 1E2 FEQPTETPP (SEQ ID NO: 26) 381-389 HLA-B41 IE1 FPKTTNGCSQA (SEQ ID NO: 27) 221-231 HLA-B55 pp65 FPTKDVAL (SEQ ID NO: 28) 188-195 HLA-B35 pp65 FTSQYRIQGKL (SEQ ID NO: 29) 369-379 HLA-A24 pp65 FVFPTKDVALR (SEQ ID NO: 30) 186-196 HLA-A68 IE2 IIYTRNHEVK (SEQ ID NO: 31) 242-250 HLA-A2 pp65 IPSINVHHY (SEQ ID NO: 32) 123-131 HLA-B35 pp150 KARDHLAVL (SEQ ID NO: 33) 101-109 FRA-B7 IE1 KEVNSQLSL (SEQ ID NO: 34) 42-50 FRA-B40 IE1 KRKMIYMCY (SEQ ID NO: 35) 201-209 FRA-B27 pp65 MLNIPSINV (SEQ ID NO: 36) 120-128 HLA-A2 pp65 NLVPMVATV (SEQ ID NO: 1) 495-503 HLA-A2 pp150 NVRRSWEEL (SEQ ID NO: 37) 212-220 HLA-B7 IE1 QIKVRVDMV (SEQ ID NO: 38) 88-96 HLA-B8 pp65 QPSLILVSQY (SEQ ID NO: 39) 52-61 HLA-B35 pp150 QTVTSTPVQGR (SEQ ID NO: 40) 792-802 HLA-A68 pp65 QYDVPAALF (SEQ ID NO: 41) 341-349 HLA-A24 pp65 QYVKVYLESF (SEQ ID NO: 42) 222-231 HLA-A24 pp50 RGDPFDKNY (SEQ ID NO: 43) 274-282 HLA-A1 pp65 RIFAELEGV (SEQ ID NO: 44) 522-530 HLA-A2 gB RIWCLVVCV (SEQ ID NO: 45) 4-12 HLA-A2 pp65 RPHERNGFTVL (SEQ ID NO: 2) 265-275 HLA-B7 IE1 RRIEEICMK (SEQ ID NO: 46) 341-349 HLA-B27 IE1 RRKMMYMCY (SEQ ID NO: 47) 201-209 FRA-B27 pp65 SEHPTFTSQY (SEQ ID NO: 48) 364-373 HLA-B44 pp65 TPRVTGGGAM (SEQ ID NO: 49) 417-426 HLA-B7 pp150 TTVYPPSSTAK (SEQ ID NO: 50) 945-955 HLA-A3 pp50 TVRSHCVSK (SEQ ID NO: 51) 52-60 HLA-A3 IE1 VLAELVKQI (SEQ ID NO: 52) 81-89 HLA-A2 IE1 VLEETSVML (SEQ ID NO: 5) 316-324 HLA-A2 pp50 VTEHDTLLY (SEQ ID NO: 53) 245-253 HLA-A1 pp65 VYALPLKML (SEQ ID NO: 54) 113-121 HLA-A24 IE1 YILEETSVM (SEQ ID NO: 7) 315-323 HLA-A2 pp65 YSEHPTFTSQY (SEQ ID NO: 55) 363-373 HLA-A1 pp65 YTPDSTPCHR (SEQ ID NO: 56) 61-70 HLA-A68 IE1 YVLEETSVM (SEQ ID NO: 57) 316-324 HLA-A2 Epstein Barr Virus EBNA1 RPQKRPSCI (SEQ ID NO: 64) 72-80 HLA-B7 EBNA1 HPVGEADYF (SEQ ID NO: 65) 407-415 HLA-B53 EBNA1 HPVGEADYFEY (SEQ ID NO: 66) 407-417 HLA-B35 EBNA1 IPQCRLTPL (SEQ ID NO: 67) 528-536 HLA-B7 EBNA1 VLKDAIKDL (SEQ ID NO: 68) 574-582 HLA-A2 EBNA2 YHLIVDTDSL (SEQ ID NO: 69) 14-23 HLA-B38 EBNA2 DTPLIPLTIF (SEQ ID NO: 70) 42-51 HLA-A2/B51 EBNA2 RPTELQPTP (SEQ ID NO: 71) 234 242 HLA-B55 EBNA3A QAKWRLQTL (SEQ ID NO: 72) 158 166 HLA-B8 EBNA3A AYSSWMYSY (SEQ ID NO: 73) 176 184 HLA-A30 EBNA3A RYSIFFDY (SEQ ID NO: 74) 246-253 HLA-A24 EBNA3A FLRGRAYGL (SEQ ID NO: 75) 325-333 HLA-B8 EBNA3A KRPPIFIRRL (SEQ ID NO: 76) 378 387 HLA-B27 EBNA3A RPPIFIRRL (SEQ ID NO: 77) 379-387 HLA-B7 EBNA3A LEKARGSTY (SEQ ID NO: 78) 406-414 HLA-B62 EBNA3A HLAAQGMAY (SEQ ID NO: 79) 450-458 HLA-A1 EBNA3A YPLHEQHGM (SEQ ID NO: 80) 458-466 HLA-B35 EBNA3A VFSDGRVAC (SEQ ID NO: 81) 491-499 HLA-A29 EBNA3A VPAPAGPIV (SEQ ID NO: 82) 502-510 HLA-B7 EBNA3A SVRDRLARL (SEQ ID NO: 83) 596-604 HLA-A2 EBNA3A RLRAEAQVK (SEQ ID NO: 84) 603-611 HLA-A3 EBNA3A VQPPQLTLQV (SEQ ID NO: 85) 617 625 HLA-B46 EBNA3B HRCQAIRKK (SEQ ID NO: 86) 149-457 HLA-B27 EBNA3B TYSAGIVQI (SEQ ID NO: 87) 217-225 HLA-A24 EBNA3B RRARSLSAERY (SEQ ID NO: 88) 244-254 HLA-B27 EBNA3B VSFIEFVGW (SEQ ID NO: 89) 279-287 HLA-B58 EBNA3B AVFDRKSDAK (SEQ ID NO: 90) 399-408 HLA-A11 EBNA3B IVTDFSVIK (SEQ ID NO: 91) 416-424 HLA-A11 EBNA3B AVLLHEESM (SEQ ID NO: 92) 488-496 HLA-B35.01 EBNA3B VEITPYKPTW (SEQ ID NO: 93) 657-666 HLA-B44 EBNA3C EGGVGWRHW (SEQ ID NO: 94) 163-171 HLA-B44 EBNA3C QNGALAINTF (SEQ ID NO: 95) 213-222 HLA-B62 EBNA3C LRGKWQRRYR (SEQ ID NO: 96) 249-258 HLA-B27 EBNA3C RRIYDLIEL (SEQ ID NO: 97) 258-266 HLA-B27 EBNA3C HHIWQNLL (SEQ ID NO: 98) 271-278 HLA-B39 EBNA3C EENLLDFVRF (SEQ ID NO: 99) 281-290 HLA-B44 EBNA3C LLDFVRFMGV (SEQ ID NO: 100) 284-293 HLA-A2 EBNA3C LDFVRFMGV (SEQ ID NO: 101) 285-293 HLA-B37 EBNA3C KEHVIQNAF (SEQ ID NO: 102) 335-343 HLA-B44 EBNA3C FRKAQIQGL (SEQ ID NO: 103) 343-351 HLA-B27 EBNA3C QPRAPIRPI (SEQ ID NO: 104) 881-889 HLA-B7 EBNA-LP SLREWLLRI (SEQ ID NO: 105) 284-292 HLA-A2 (A*0203) LMP1 FWLYIVMSD (SEQ ID NO: 106) 38-46 MHC Class I LMP1 FRRDLLCPLGA (SEQ ID NO: 107) 72-82 HLA-B40 LMP1 YLLEMLWRL (SEQ ID NO: 108) 125-133 HLA-A2 LMP1 YLQQNWWTL (SEQ ID NO: 109) 159-167 HLA-A2 LMP1 TLLVDLLWL (SEQ ID NO: 110) 166-174 HLA-A2 LMP1 DPHGPVQLSYYD (SEQ ID NO: 111) 375-386 HLA-B51 LMP2 MGSLEMVPM (SEQ ID NO: 112) 1-9 HLA-B35 LMP2 EDPYWGNGDRHSDYQ (SEQ ID NO: 113) 61-75 MHC Class I LMP2 MPVCLPVIVAPYLF (SEQ ID NO: 114) 121-134 MHC Class I LMP2 LPVIVAPYL (SEQ ID NO: 115) 125-133 HLA-B53 LMP2 PYLFWLAAI (SEQ ID NO: 116) 131-139 HLA-A23 LMP2 ASCFTASVSTVVTA (SEQ ID NO: 117) 141-154 MHC Class I LMP2 FTASVSTVV (SEQ ID NO: 118) 144-152 HLA-A68 LMP2 IEDPPFNSL (SEQ ID NO: 119) 200-208 HLA-B40 LMP2 RRRWRRLTV (SEQ ID NO: 120) 236-244 HLA-B27 LMP2 RRWRRLTVC (SEQ ID NO: 121) 237-245 HLA-B14 LMP2 RRLTVCGGIMF (SEQ ID NO: 122) 240-250 HLA-B27 LMP2 TVCGGIMFL (SEQ ID NO: 123) 243-251 HLA-A1 LMP2 MFLACVLVLIVDAV (SEQ ID NO: 124) 249-262 MHC Class I LMP2 LIVDAVLQL (SEQ ID NO: 125) 257-265 HLA-A2 LMP2 GLGTLGAAI (SEQ ID NO: 126) 293-301 HLA-A2 LMP2 LLWTLVVLL (SEQ ID NO: 127) 329-337 HLA-A2 LMP2 SSCSSCPLSKI (SEQ ID NO: 128) 340-350 HLA-A11 LMP2 ILLARLFLY (SEQ ID NO: 129) 349-358 HLA-A29 LMP2 FLYALALLL (SEQ ID NO: 130) 356-364 HLA-A2 LMP2 TYGPVFMCL (SEQ ID NO: 131) 419-427 HLA-A24 LMP2 CLGGLLTMV (SEQ ID NO: 132) 426-434 HLA-A2 LMP2 MVSNTLLSAW (SEQ ID NO: 133) 442-451 HLA-A25 LMP2 LTAGFLIFL (SEQ ID NO: 134) 453-461 HLA-A2 LMP2 LLSAWILTA (SEQ ID NO: 135) 447-455 HLA-A2 BRLF1 LVSDYCNVLNKEFT (SEQ ID NO: 136) 25-39 MHC Class I BRLF1 LVSDYCNVL (SEQ ID NO: 137) 25-33 HLA-A2 BRLF1 DYCNVLNKEF (SEQ ID NO: 138) 28-37 HLA-A24 BRLF1 AENAGNDAC (SEQ ID NO: 139) 91-99 HLA-B45 BRLF1 IACPIVMRYYVLDHLI (SEQ ID NO: 140) 101-115 HLA-A24/C2 BRLF1 YVLDHLIVV (SEQ ID NO: 141) 109-117 HLA-A2 BRLF1 FFIQAPSNRVMIPAT (SEQ ID NO: 142) 121-135 MHC Class I BRLF1 ATIGTAMYK (SEQ ID NO: 143) 134-142 HLA-A11 BRLF1 KHSRVRAYTYSKVLG (SEQ ID NO: 144) 145-159 HLA-A3 BRLF1 RALIKTLPRASYSSH (SEQ ID NO: 145) 225-239 HLA-A2 BRLF1 ERPIFPHPSKPTFLP (SEQ ID NO: 146) 393-407 HLA-C4 BRLF1 QKEEAAICGQMDLS (SEQ ID NO: 147) 529-543 HLA-B61 BRLF1 EVCQPKRIRPFHPPG (SEQ ID NO: 148) 441-455 MHC Class I BZLF1 LPEPLPQGQLTAY (SEQ ID NO: 149) 52-64 MHC Class I BZLF1 EPLPQGQLTAY (SEQ ID NO: 150) 54-63 HLA-B35 BZLF1 APENAYQAY (SEQ ID NO: 151) 81-89 HLA-B35 BZLF1 LQHYREVAA (SEQ ID NO: 152) 101-115 HLA-C8 BZLF1 DSELEIKRYKNR (SEQ ID NO: 153) 172-183 HLA-B18 BZLF1 RKCCRAKFKQLLQHYR (SEQ ID NO: 154) 186-201 HLA-C6 BZLF1 RAKFKQLL (SEQ ID NO: 155) 190-197 HLA-B8 BZLF1 SENDRLRLL (SEQ ID NO: 156) 209-217 HLA-B60 Influenza A Virus PR8 CTELKLSDY (SEQ ID NO: 157) 44-52 HLA-A1 PB1 VSDGGPNLY (SEQ ID NO: 158) 591-599 HLA-A1 MP GILGFVFTL (SEQ ID NO: 159) 58-66 HLA-A2 MP ILGFVFTLTV (SEQ ID NO: 160) 59-68 HLA-A2 PR8 ILRGSVAHK (SEQ ID NO: 161) 265-274 HLA-A3 MP SIIPSGPLK (SEQ ID NO: 162) 13-21 HLA-A11 MP1 RMVLASTTAK (SEQ ID NO: 163) 178-187 HLA-A11 MP2 KSMREEYRK (SEQ ID NO: 164) 70-78 HLA-A11 NP SPIVPSFDM (SEQ ID NO: 165) 473-481 HLA-B7 PB1 QPEWFRNVL (SEQ ID NO: 166) 329-337 HLA-B7 NP SRYWAIRTR (SEQ ID NO: 167) 383-391 HLA-B27
[0116] F. Targeting Moiety
[0117] The targeting moiety functions in the TPEC by delivering the TPEC to the local environment of the unwanted cells, enabling a localized treatment strategy. In certain embodiments, the targeting moiety targets the unwanted cells by specifically binding to the unwanted cells. In some instances, the targeting moiety specifically binds the unwanted cells even while the plurality of T-cell epitopes are conjugated to the targeting moiety.
[0118] In certain embodiments, the targeting moiety is an antibody or functional part thereof.
[0119] Certain antibody targets (with examples of unwanted cell types in parentheses) may include: Her2/Neu (Epithelial malignancies); CD22 (B cells, autoimmune or malignant); EpCAM (CD326) (Epithelial malignancies); EGFR (epithelial malignancies); PMSA (Prostate Carcinoma); CD30 (B cell malignancies); CD20 (B cells, autoimmune, allergic or malignant); CD33 (Myeloid malignancies); membrane lgE (Allergic B cells); lgE Receptor (CD23) (Mast cells or B cells in allergic disease), CD80 (B cells, autoimmune, allergic or malignant); CD86 (B cells, autoimmune, allergic or malignant); CD2 (T cell or NK cell lymphomas); CA125 (multiple cancers including Ovarian carcinoma); Carbonic Anhydrase IX (multiple cancers including Renal Cell Carcinoma); CD70 (B cells, autoimmune, allergic or malignant); CD74 (B cells, autoimmune, allergic or malignant); CD56 (T cell or NK cell lymphomas); CD40 (B cells, autoimmune, allergic or malignant); CD19 (B cells, autoimmune, allergic or malignant); c-met/HGFR (Gastrointestinal tract and hepatic malignancies; TRAIL-R1 (multiple malignancies including ovarian and colorectal carcinoma); DRS (multiple malignancies including ovarian and colorectal carcinoma); PD-1 (B cells, autoimmune, allergic or malignant); PD1L (Multiple malignancies including epithelial adenocarcinoma); IGF-1R (Most malignancies including epithelial adenocarcinoma); VEGF-R2 (The vasculature associated with the majority of malignancies including epithelial adenocarcinomas; Prostate stem cell antigen (PSCA) (Prostate Adenocarcinoma); MUC1 (Epithelial malignancies); CanAg (tumors such as carcinomas of the colon and pancreas); Mesothelin (many tumors including mesothelioma and ovarian and pancreatic adenocarcinoma); P-cadherin (Epithelial malignancies, including breast adenocarcinoma); Myostatin (GDF8) (many tumors including sarcoma and ovarian and pancreatic adenocarcinoma); Cripto (TDGF1) (Epithelial malignancies including colon, breast, lung, ovarian, and pancreatic cancers); ACVRL 1/ALK1 (multiple malignancies including leukaemias and lymphomas); MUC5AC (Epithelial malignancies, including breast adenocarcinoma); CEACAM (Epithelial malignancies, including breast adenocarcinoma); CD137 (B cells or T cells, autoimmune, allergic or malignant); CXCR4 (B cells or T cells, autoimmune, allergic or malignant); Neuropilin 1 (Epithelial malignancies, including lung cancer); Glypicans (multiple cancers including liver, brain and breast cancers); HERS/EGFR (Epithelial malignancies); PDGFRa (Epithelial malignancies); EphA2 (multiple cancers including neuroblastoma, melanoma, breast cancer, and small cell lung carcinoma); CD38 (Myeloma); CD138 (Myeloma); a4-integrin (AML, myeloma, CLL, and most lymphomas).
[0120] In certain modes, antibodies include an anti-epidermal growth factor receptor antibody such as Cetuximab, an anti-Her2 antibody, an anti-CD20 antibody such as Rituximab, an anti-CD22 antibody such as Inotuzumab, G544 or BU59, an anti-CD70 antibody, an antiCD-33 antibody such as hp67.6 or Gemtuzumab, an anti-MUC1 antibody such as GP1.4 and SM3, an anti-CD40 antibody, an anti-CD74 antibody, an anti-P-cadherin antibody, an anti-EpCAM antibody, an anti-CD138 antibody, an anti-E-cadherin antibody, an anti-CEA antibody, an anti-FGFR3 antibody, and an anti-.alpha.4-integrin antibody such as natalizumab.
[0121] G. Cleavage Sites
[0122] The cleavage sites function to release the T-cell epitope from the targeting moiety and, in some embodiments, to release the T-cell epitopes from each other. Releasing the T-cell epitopes into single epitopes allows them to most effectively label the unwanted cell for immune attack.
[0123] In some instances, the cleavage site may be a separate sequence and in other instances the cleavage site may be integrated into the T-cell epitope such that one sequence serves the function of both elements. This may apply to any of the embodiments described herein and is determined by the selection of the epitope sequences.
[0124] The cleavage sites can function in different ways to release the T-cell epitopes in the microenvironment of the unwanted cells. The cleavage may occur inside the unwanted cell or outside the unwanted cell, depending on the strategy employed. If cleavage occurs outside the unwanted cell, the T-cell epitope peptides can be presented without first being internalized into a cell and being engaged in the classical antigen-processing pathways. If cleavage occurs outside the unwanted cell, it may occur in the microenvironment surrounding the cell, including at the cell surface. For example, when the unwanted cell is a cancer cell, the cleavage may occur in the tumor microenvironment (outside of an in the vicinity of the cancer cell), including at the surface of the cancer cell.
[0125] In certain embodiments, at least one cleavage site may be cleaved by an enzyme expressed by the unwanted cells. Cancer cells, for instance, are known to express certain enzymes, such as proteases, and these may be employed in the TPEC strategy to cleave the TPEC's cleavage site. By way of nonlimiting example, cathepsin B cleaves FR, FK, VA and VR amongst others; cathepsin D cleaves PRSFFRLGK (SEQ ID NO: 181), ADAM28 cleaves KPAKFFRL (SEQ ID NO: 182), DPAKFFRL (SEQ ID NO: 183), KPMKFFRL (SEQ ID NO: 184) and LPAKFFRL (SEQ ID NO: 185); and MMP2 cleaves AIPVSLR (SEQ ID NO: 186).
[0126] In some embodiments, at least one cleavage site may be cleaved through a pH-sensitive cleavage reaction inside the unwanted cell. If the TPEC is internalized into the cell, the cleavage reaction may occur inside the cell and may be triggered by a change in pH between the microenvironment outside the unwanted cell and the interior of the cell. Specifically, some cancer types are known to have acidic environments in the interior of the cancer cells. Such an approach may be employed when the interior unwanted cell type has a characteristically different pH from the extracellular microenvironment, such as particularly the glycocalyx. Because pH cleavage can occur in all cells in the lysozymes, selection of a targeting agent when using a pH-sensitive cleavage site may require, when desired, more specificity. For example, when a pH-sensitive cleavage site is used, a targeting agent that binds only or highly preferably to cancer cells may be desired (such as, for example, an antibody binding to mesothelin for treatment of lung cancer).
[0127] In certain embodiments, at least one cleavage site may be cleaved by a complement-dependent cleavage reaction. Once TPECs bind to the unwanted cell, the patient's complement cascade may be triggered. In such a case, the complement cascade may also be used to cleave the T-cell epitope from the targeting agent by using a cleavage site sensitive to a complement protease. For example, C1r and C1s and the C3 convertases (C4B,2a and C3b,Bb) are serine proteases. C3/C5 and C5 are also complement proteases Mannose-associated binding proteins (MASP), serine proteases also involved in the complement cascade and responsible for cleaving C4 and C2 into C4b2b (a C3 convertase) may also be used. For example, and without limitation, C1s cleaves YLGRSYKV (SEQ ID NO: 177) and MQLGRX (SEQ ID NO: 178). MASP2 is believed to cleave SLGRKIQI (SEQ ID NO: 179). Complement component C2a and complement factor Bb are believed to cleave GLARSNLDE (SEQ ID NO: 180).
[0128] In some embodiments, at least one cleavage site may be cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC. For example, any protease may be simultaneously directed to the microenvironment of the unwanted cells by conjugating the protease to a targeting agent that delivers the protease to that location. The targeting agent may be any targeting agent described herein. The protease may be affixed to the targeting agent through a peptide or chemical linker and may maintain sufficient enzymatic activity when bound to the targeting agent.
[0129] In some embodiments, the TPEC has a plurality of cleavage sites that are the same. In other embodiments, the TPEC has a plurality of cleavage sites that are not all identical, either in sequence and/or in type, as described above.
[0130] H. Preparation of the T-Cell Epitopes and Cleavage Sites
[0131] 1. Preparation of Individual T-Cell Epitopes and Cleavage Sites to be Separately Conjugated to Targeting Agent
[0132] Individual T-cell epitopes conjugated to cleavage sites may be prepared through standard peptide synthesis chemistry, such as by coupling the carboxyl group of the incoming amino acid to the N-terminus of the growing peptide chain using a N-terminal protecting group addition strategy, such as tert-butoxycaronyl (Boc) and 9-fluorenylmethoxycarbonyl (Fmoc), with their respective deprotection agents TFA and piperidine. Peptide synthesis may occur by hand or in an automated machine. Alternatively, services may be employed that prepare peptides upon order.
[0133] 2. Preparation of Linear Polytopes
[0134] Polytopes up to -50 amino acids could be made by standard peptide synthesis. This would allow up to about 5 epitopes to be incorporated into the polytope. In instances where more epitopes in the polytope are desired, or when human protein domains flank the epitopes in the polytope, then recombinant production of the polytope may occur. The DNA sequence for the polytope would be incorporated into a vector that would allow a cell line such as Chinese hamster ovary (CHO) cells to express the protein. The protein would be secreted by the cell line and could be purified from the cell culture supernatant. The purified polytope could then be conjugated to a targeting moiety through a chemical linker, as described herein. In another embodiment, nucleic acids encoding the polytope could be added on to an end of the DNA sequence encoding the targeting moiety to make one continuous polypeptide chain incorporating the targeting moiety and polytope. This could then be expressed in a cell line in the same way as the previous embodiment.
[0135] 3. Preparation of Branched Polytope
[0136] Branched polytopes, such as shown in FIG. 2C, may also be prepared. Individual T-cell epitopes to be applied to a branched polytope may be prepared according to Section I.H.1 before being conjugated to a connecting stem. In some embodiments, the connecting stem may be comprised of amino acids and may be a peptide stem.
[0137] The peptide stem may comprise amino acids that have a reactive property that can be targeted by crosslinking reagents. In some embodiments, cysteine and lysine may be used as there are a large number of crosslinking reagents that may be used at these amino acids. In one embodiment, a connecting stem may be a peptide comprising a large number of lysine or cysteine residues to facilitate linking reactions by sulfo-SMCC, for example. In another embodiment, the connecting stem may comprise serine or threonine, so as to partner with a crosslinking agent that would link to hydroxyl groups on these amino acids.
[0138] In another embodiment, spacer amino acids may be used in between those that have a reactive property to the crosslinking reagents in order to allow for protease attack without being blocked by other peptides bound to the stem. In certain aspects, a basic spacer may include glycine and serine. In some embodiments, the incorporation of proline between reactive amino acids may be useful as proline induces a slight rotation of the peptide which may help to keep the protease cleavable peptides bound to the stem further apart.
[0139] In certain embodiments, the peptide stem may comprise from about 10 to 80, 20 to 80, or 40 to 80 amino acids. In one embodiment the peptide stem comprises from about 2 to 20 amino acids that are bound to a T-cell epitope and cleavage site.
[0140] The individual T-cell epitopes may be conjugated to the peptide stem using the chemical linker technology discussed in Section I.I below. The connecting stem comprising the individual T-cell epitopes may also be conjugated to the targeting moiety using the chemical linker technology discussed in Section LI below. In some embodiments, a different chemical linker may be chosen for these two conjugations so as to have additional control over the conjugation process. In one embodiment, T-cell epitopes may be affixed to the connecting stem by sulfo-SMCC and the connecting stem comprising the T-cell epitopes to the targeting moiety by 3-MPA, for example.
[0141] I. Method of Conjugation to the Targeting Moiety
[0142] Different approaches may be employed to conjugate the cleavage site(s) and plurality of T-cell epitopes (whether as separate T-cell epitope and cleavage site pairs or whether as a polytope) to the targeting agent. In some aspects, a polytope comprising a plurality of cleavage sites and a plurality of T-cell epitopes are conjugated to the targeting agent using at least one peptide bond. In certain aspects, the conjugation occurs through at least one bond other than a peptide bond. Such a conjugation may be through a chemical linker A nonpeptide bond, such as using a chemical linker, may be employed for separately conjugated pairs of T-cell epitopes and cleavage sites or for polytopes.
[0143] In some instances, a chemical linker may be a heterobifunctional crosslinking reagent. Heterobifunctional crosslinking reagents covalently bind two proteins together by targeting different functional groups on separate proteins. The amine-to-sulphydryl crosslinking reagents contain an NHS-ester at one end of the reagent, which binds to free amine groups on a first protein/peptide, predominantly on the amino acid lysine. At the other end of the reagent, a maleimide group binds to sulphydryl group of a second protein/peptide, which can be found on the amino acid cysteine. Examples of amine-to-sulphydryl crosslinking reagents may include (a) sulfosuccinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (Sulfo-SMCC, which is soluble in water) or succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC, which has poor water solubility), (b) succinimidyl 4-(p-maleimidophenyl)butyrate (SMPB), (c) succinimidyl 6-[(beta-maleimidopropionamido)hexanoate] (SMPH); and (d) Sulfosuccinimidyl 6-(3'-[2-pyridyldithio]-propionamido) hexanoate (Sulfo-LC-SPDP); or (e) maleimide such as 3-maleimidopropionic acid.
[0144] Alternatively, another class of cross-linking reagents are the sulphydryl-to-carbohydrate reagents, which contain a maleimide group at one end and a hydrazide group at the other end. The maleimide group binds to a free sulphydryl on a first protein/peptide, such as that found on the amino acid cysteine, and the hydrazide group binds to an aldehyde group formed from an oxidized carbohydrate on a second protein/peptide. This family of reagents includes (a) 3,3'-N-[.epsilon.-Maleimidocaproic acid] hydrazide, trifluoroacetic acid salt (EMCH) and (b)N-beta-Maleimidopropionic acid hydrazide-trifluoroacetic acid (BMPH).
[0145] A third group of heterobifunctional cross-linking reagents are the photocleavable reagents including sulfosuccinimidyl 2-([4,4'-azipentanamido]ethyl)-1,3'-dithiopropionate (SDAD). These reagents have an NHS-ester at one end linking to free amines (lysine) and a diazrine group at the other end which can react with any amino acid side chain or peptide backbone upon activation with long-wave ultra violet light (330-370 nm). There are many other reagents from these groups and from other cross-linking groups that can be used in this technology.
[0146] J. Inclusion of Human Protein Domains
[0147] In certain embodiments, the T-cell epitopes are flanked on one or both ends by at least one human protein domain. In some embodiments, the cleavage sites reside between the T-cell epitopes and the human protein domains
[0148] Various human protein domains may be used in this manner. For example, at least one human protein domain may be a beta barrel or a coiled coil. In some embodiments, the human protein may be a FN3 (Fibronectin type III) domain. In some embodiments, the human protein may be beta-sandwich, Lipocalin, EETI-II/AGRP, Kunitz domain (BPTI), Thioredoxin, Protein A, Ankyrin, gamma-B-crystallin/ubiquitin, CTLD3/Tetranexin, or and LDLR-A In some embodiments, the human protein may be human serum albumin, immunoglobulin CH2 domain, or camelid VHH domain.
[0149] In certain instances, a human protein domain may be a CL, CH2, and/or CH3 domain. For example, a T-cell epitope or plurality of T-cell epitopes may be flanked by CH2 sequences. Such CH2 sequences may be within the CH2 domain of an antibody or functional part serving as the targeting moiety. Such CH2 sequences may also be an additional CH2 domain distinct from the targeting moiety. In some embodiments, such as shown in FIG. 2B, framework regions may be employed in a polytope to give additional three dimensional organization to the polytope.
[0150] In some aspects, the human protein domain is not a human immunoglobulin protein domain
[0151] In some aspects, the human protein domain displays the T-cell epitopes on its three-dimensional surface.
[0152] II. Pharmaceutical Compositions
[0153] The TPECs may be employed as pharmaceutical compositions. As such, they may be prepared along with a pharmaceutically acceptable carrier. If parenteral administration is desired, for instance, the TPECs may be provided in sterile, pyrogen-free water for injection or sterile, pyrogen-free saline. Alternatively, the TPECs may be provided in lyophilized form for resuspension with the addition of a sterile liquid carrier.
[0154] III. Methods of Treatment
[0155] A. Reduction of Unwanted Cells, Retargeting of Immune Response, and Treatment of Cancer
[0156] The TPECs described herein may be used in a method of treating a disease in a patient characterized by the presence of unwanted cells comprising administering a TPEC composition to the patient. This may include both treating and preventing a disorder. Additionally, the TPECs described herein may also be used in a method of retargeting (i.e., redirecting) a patient's own immune response to unwanted cells comprising administering a TPEC composition to the patient.
[0157] The amount of the agent administered to the patient may be chosen by the patient's physician so as to provide an effective amount to treat the condition in question.
[0158] The patient receiving treatment may be a human. The patient may be a primate or any mammal. Alternatively, the patient may be an animal, such as a domesticated animal (for example, a dog or cat), a laboratory animal (for example, a laboratory rodent, such as a mouse, rat, or rabbit), or an animal important in agriculture (such as horses, cattle, sheep, or goats).
[0159] The condition characterized by unwanted cells may include cancer. The cancer may be a solid or non-solid malignancy. The cancer may be any cancer such as breast cancer, ovarian cancer, endometrial cancer, cervical cancer, bladder cancer, renal cancer, melanoma, lung cancer, prostate cancer, testicular cancer, thyroid cancer, brain cancer, oesophageal cancer, gastric cancer, pancreatic cancer, colorectal cancer, liver cancer, leukaemia, myeloma, nonHodgkin's lymphoma, Hodgkin's lymphoma, acute myeloid leukaemia, acute lymphoblastic leukaemia, chronic lymphoblastic leukaemia, lymphoproliferative disorder, myelodysplastic disorder, myeloproliferative disease and premalignant disease.
[0160] The condition characterized by unwanted cells may also include an allergic or autoimmune disease. For instance, autoimmune diseases may include Addison's disease, celiac disease, dermatomyositis, Graves' disease, Hashimoto's thyroiditis, multiple sclerosis, myasthenia gravis, pernicious anemia, reactive arthritis, rheumatoid arthritis, Sjogren syndrome, and systemic lupus erythematosus.
[0161] The TPECs may be administered alone or in conjunction with other forms of therapy, including surgery, radiation, or traditional chemotherapy. In some embodiments, the activity of the TPECs may also be enhanced by boosting the patient's immune response against one or more of the T-cell epitopes used, such as by vaccinating the subject with the T-cell epitope or by administering immunostimulatory agents.
[0162] In some embodiments, the patient receives multiple doses of the composition over at least 30, 45, 60, 75, 90, 120, 150, or more days, or on an ongoing basis. In certain modes the patient receives multiple doses of the composition over at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 months, or on an ongoing basis. If approaches are employed to reduce the patient's immune response against the TPEC itself, additional benefits may be achieved by administering the composition to a patient in relapse who received the composition for an earlier round of therapy.
[0163] B. Reduction of Immune Response Against TPEC Itself
[0164] In certain aspects described herein, having multiple copies of the same T-cell epitope increases an immune response against the unwanted cell when the agent is administered to a patient and having a diversity of T-cell epitopes assures effectiveness across a wide variety of patients from different racial and ethnic groups. Nevertheless, if a physician desires to use a TPEC therapy over a continued period of time, optional steps may be taken to reduce the patient's own immune response against the TPEC itself.
[0165] In certain embodiments, the arrangement or composition of T-cell epitopes on the TPEC are random in nature. For example, if a mixture of T-cell epitopes are conjugated using a chemical linker to the targeting moiety, different combinations of T-cell epitopes may affix to the targeting moieties. This makes it significantly more difficult for the patient to mount an immune response against the TPECs themselves. Even in instances where the plurality of T-cell epitopes are the same, affixing them separately to the targeting moiety through a chemical linker may also result in a random placement of the T-cell epitopes along the targeting moiety, likewise damping the patient's own immune response against the TPEC.
[0166] Such approaches may ensure that the patient does not develop an immune response against the TPEC composition sufficient to inactivate the TPEC composition upon subsequent administrations, meaning that no more than about 0%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, or 90% of the composition is inactivated by the patient's own antibodies to the TPEC.
[0167] Immunogenicity of a composition may be measured in an ELISA assay using human serum. For example, test compositions may be immobilized on a 96-well plate, blocked and diluted serum samples incubated for 1 hour. Antibodies against the test composition present in the human sera may bind to the test compositions on the plate. Biotinylated test agent or a biotinylated secondary antibody such as an anti-human IgG/IgA/IgM in combination with streptavidin-HRP and TMB could be used to generate a signal.
[0168] The present compositions are expected to be at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, or 90% less immunogenic than prior compositions.
EXAMPLES
Example 1. Preparation of T-cell Epitopes and Cleavage Sites
[0169] Peptides comprising T-cell epitopes and cleavage sites were ordered from external companies who synthesized them in standard solid phase peptide synthesis. The peptides were dissolved in DMSO to a final concentration of 10 mg/ml to be used for conjugation.
Example 2. Standard Operating Protocol for Conjugation of Polytope Using Sulfo-SMCC
[0170] The following procedure was used as a standard-operating protocol for conjugation of polytope peptide (comprising a plurality of T-cell epitopes and cleavage sites) to an antibody serving as a targeting moiety by using sulfo-SMCC.
[0171] 1. Cysteinylated peptides dissolved in DMSO to final concentration of 10 mg/ml.
[0172] 2. Weigh 1 mg Sulfosuccinimidyl 4-[N-maleimidomethyl]cyclohexane-1-carboxylate (Sulfo-SMCC) and dissolve in 200 .mu.l phosphate buffered saline (PBS) (final concentration 5 mg/ml).
[0173] 3. Add 100 .mu.l antibody (10 mg/ml, 1 mg antibody) to 10 .mu.l dissolved Sulfo-SMCC (5 mg/ml) and incubate at room temperature for 30 minutes.
[0174] 4. Wash a ZebaSpin desalting column (Pierce) by firstly spinning the column at 1500 g for 60 seconds to remove the ethanol (storage buffer).
[0175] 5. Add 300 .mu.l PBS and spin at 1500 g for 60 seconds. Remove eluate and repeat a further three times.
[0176] 6. Add up to 125 .mu.l antibody-SMCC to each column, and centrifuge at 1500 g for 120 seconds, collecting the eluate.
[0177] 7. To 100 .mu.l antibody-SMCC conjugate, add 3 .mu.l cysteinylated peptide (10 mg/ml) and incubate at room temperature for 30 minutes.
[0178] 8. Wash a Protein A column (GE Healthcare) by firstly spinning the column at 100 g for 30 seconds to remove the ethanol (storage buffer).
[0179] 9. Add 500 .mu.l PBS and spin at 100 g for 30 seconds. Remove eluate and repeat a further two times.
[0180] 10. Dilute antibody-peptide conjugate to 500 .mu.l in PBS and add it to the protein A column, mixing the antibody with the beads. Leave at room temperature for 20 minutes, shaking
[0181] 11. Spin the column at 100 g and remove the eluate, the antibody-peptide complex should still be coupled to the beads.
[0182] 12. Wash the column by adding 500 .mu.l PBS and mixing the beads well before spinning at 100 g for 30 seconds and removing eluate. Repeat this step a further three times.
[0183] 13. To elute the bound antibody, add 200 .mu.l 0.1M citric acid to the beads and incubate for 5 minutes at room temperature. Place column in a 2 ml eppendorf containing 25 .mu.l 1M Tris pH9 and spin at 100 g for 30 seconds; collect eluate.
[0184] 14. Optionally repeat step 13.
[0185] 15. Store antibody-peptide complex at 4.degree. C.
Example 3. Standard Operating Protocol for Production of a TPEC Comprising Separately-Conjugated Epitopes Using Sulfo-SMCC
[0186] The following procedure was used as a standard-operating protocol for separately conjugating a plurality of T-cell epitopes and their corresponding cleavage sites to an antibody serving as a targeting moiety by using sulfo-SMCC.
[0187] 1. Cysteinylated peptides dissolved in DMSO to final concentration of 10 mg/ml.
[0188] 2. Weigh 1 mg Sulfosuccinimidyl 4-[N-maleimidomethyl]cyclohexane-1-carboxylate (Sulfo-SMCC) and dissolve in 200 .mu.l phosphate buffered saline (PBS) (final concentration 5 mg/ml).
[0189] 3. Add 100 .mu.l antibody (10 mg/ml, 1 mg antibody) to 10 .mu.l dissolved Sulfo-SMCC (5 mg/ml) and incubate at room temperature for 30 minutes.
[0190] 4. Wash a ZebaSpin desalting column (Pierce) by firstly spinning the column at 1500 g for 60 seconds to remove the ethanol (storage buffer).
[0191] 5. Add 300 .mu.l PBS and spin at 1500 g for 60 seconds. Remove eluate and repeat a further three times.
[0192] 6. Add up to 125 .mu.l antibody-SMCC to each column, and centrifuge at 1500 g for 120 seconds, collecting the eluate.
[0193] 7. Generate a mixture or required peptides in a tube so that there is a total of 30 .mu.g of total peptide. Add the peptide mixture to 100 .mu.l of antibody-SMCC complex and leave at room temperature for 30 minutes.
[0194] 8. Wash a Protein A column (GE Healthcare) by firstly spinning the column at 100 g for 30 seconds to remove the ethanol (storage buffer).
[0195] 9. Add 500 .mu.l PBS and spin at 100 g for 30 seconds. Remove eluate and repeat a further two times.
[0196] 10. Dilute antibody-peptide conjugate to 500 .mu.l in PBS and add it to the protein A column, mixing the antibody with the beads. Leave at room temperature for 20 minutes, shaking.
[0197] 11. Spin the column at 100 g and remove the eluate, the antibody-peptide complex should still be coupled to the beads.
[0198] 12. Wash the column by adding 500 .mu.l PBS and mixing the beads well before spinning at 100 g for 30 seconds and removing eluate. Repeat this step a further three times.
[0199] 13. To elute the bound antibody, add 200 .mu.l 0.1M citric acid to the beads and incubate for 5 minutes at room temperature. Place column in a 2 ml eppendorf containing 25 .mu.l 1M Tris pH9 and spin at 100 g for 30 seconds; collect eluate.
[0200] 14. Optionally repeat step 13.
[0201] 15. Store antibody-peptide complex at 4.degree. C.
Example 4. Standard Operating Protocol for Production of Polytope TPEC Using 3-Maleimidopropionic Acid Linked Peptide
[0202] The following procedure was used as a standard-operating protocol for separately conjugating a polytope to an antibody serving as a targeting moiety by using 3-MPA.
[0203] 1. 3-maleimidopropioinc acid linked peptides dissolved in DMSO to final concentration of 10 mg/ml.
[0204] 2. To 100 .mu.l antibody (diluted in PBS if required), add 3 .mu.l polytope peptide (10 mg/ml) and incubate at room temperature for 30 minutes.
[0205] 3. Wash a Protein A column (GE Healthcare) by firstly spinning the column at 100 g for 30 seconds to remove the ethanol (storage buffer).
[0206] 4. Add 500 .mu.l PBS and spin at 100 g for 30 seconds. Remove eluate and repeat a further two times.
[0207] 5. Dilute antibody-peptide conjugate to 500 .mu.l in PBS and add it to the protein A column, mixing the antibody with the beads. Leave at room temperature for 20 minutes, shaking
[0208] 6. Spin the column at 100 g and remove the eluate, the antibody-peptide complex should still be coupled to the beads.
[0209] 7. Wash the column by adding 500 .mu.l PBS and mixing the beads well before spinning at 100 g for 30 seconds and removing eluate. Repeat this step a further three times.
[0210] 8. To elute the bound antibody, add 200 .mu.l 0.1M citric acid to the beads and incubate for 5 minutes at room temperature. Place column in a 2 ml eppendorf containing 25 .mu.l 1M Tris pH9 and spin at 100 g for 30 seconds; collect eluate.
[0211] 9. Optionally repeat step 8.
[0212] 10. Store antibody-peptide complex at 4.degree. C.
Example 5. Standard Operating Protocol for Production of a TPEC Comprising Separately-Conjugated Epitopes Using 3-Maleimidopropionic Acid Linked Peptide
[0213] The following procedure was used as a standard-operating protocol for separately conjugating a plurality of T-cell epitopes and their corresponding cleavage sites to an antibody serving as a targeting moiety by using 3-MPA.
[0214] 1. 3-maleimidopropioinc acid linked peptides dissolved in DMSO to final concentration of 10 mg/ml.
[0215] 2. Generate a mixture or required peptides in a tube so that there is a total of 30 .mu.g of total peptide, add to 100 .mu.l of antibody (diluted in PBS if required) and incubate at room temperature for 30 minutes.
[0216] 3. Wash a Protein A column (GE Healthcare) by firstly spinning the column at 100 g for 30 seconds to remove the ethanol (storage buffer).
[0217] 4. Add 500 .mu.l PBS and spin at 100 g for 30 seconds. Remove eluate and repeat a further two times.
[0218] 5. Dilute antibody-peptide conjugate to 500 .mu.l in PBS and add it to the protein A column, mixing the antibody with the beads. Leave at room temperature for 20 minutes, shaking
[0219] 6. Spin the column at 100 g and remove the eluate, the antibody-peptide complex should still be coupled to the beads.
[0220] 7. Wash the column by adding 500 .mu.l PBS and mixing the beads well before spinning at 100 g for 30 seconds and removing eluate. Repeat this step a further three times.
[0221] 8. To elute the bound antibody, add 200 .mu.l 0.1M citric acid to the beads and incubate for 5 minutes at room temperature. Place column in a 2 ml eppendorf containing 25 .mu.l 1M Tris pH9 and spin at 100 g for 30 seconds; collect eluate.
[0222] Optionally repeat step 8.
[0223] Store antibody-peptide complex at 4.degree. C.
Example 6. TPEC Compositions Comprising Two Cytomegalovirus Peptides and Evaluation in B-Lymphoblastoid Cells
[0224] We contacted B-lymphoblastoid cells (B-LCL) with an agent comprising Rituximab conjugated to two different cytomegalovirus peptides NLVPMVATV (SEQ ID NO: 1) and RPHERNGFTVL (SEQ ID NO: 2) each with a cleavage site for cathepsin B. Subsequent exposure to peptide-specific T cells resulted in the generation of a T cell response to the B-LCL.
[0225] Rituximab was conjugated with the peptides NLVPMVATVASGV{CIT}GC (SEQ ID NO: 3) and RPHERNGFTVLASGFKGC (SEQ ID NO: 4) at the following ratios from Table 3, wherein CIT represents citrulline, a type of amino acid that is similar to arginine
TABLE-US-00005 TABLE 3 Peptide Ratios NLVPMVATVASGV RPHERNGFTVLAS {CIT}GC GFKGC TPEC (SEQ ID NO: 3) (SEQ ID NO: 4) 1 0% 100% 2 10% 90% 3 20% 80% 4 30% 70% 5 40% 60% 6 50% 50% 7 60% 40% 8 70% 30% 9 80% 20% 10 90% 10% 11 100% 0%
[0226] After staining the cells with Rituximab conjugated to the mixture of peptides, labeled cells were washed and cultured overnight at 37.degree. C. in the presence of peptide-specific T cells. Supernatant was harvested and assayed for the presence of IFN-.gamma..
[0227] Using NLV-specific T cells, TPEC #11 was recognized strongly as seen by a large production of IFN-.gamma. whereas TPEC #1 is not recognized at all as there was no production of IFN-.gamma.. See FIG. 3A. Conversely, using RPH-specific T cells TPEC #1 was recognized strongly as seen by a large production of IFN-.gamma. whereas TPEC #11 is not recognized at all as there was no production of IFN-.gamma.. See FIG. 3B. There is production of IFN-.gamma. by both NLV- and RPH-specific T cells towards TPEC #2-10, demonstrating the notion that conjugating more than one peptide to the antibody can allow one to engage with more than one specificity of peptide-specific T cell.
Example 7. TPEC Compositions Comprising Three Cytomegalovirus Peptides and Evaluation in B-Lymphoblastoid Cells
[0228] We contacted B-lymphoblastoid cells (B-LCL) with an agent comprising Rituximab conjugated to three different cytomegalovirus peptides VLEETSVML (SEQ ID NO: 5), BRVLBBYVL (SEQ ID NO: 6) and YILEETSVM (SEQ ID NO: 7) each with a cleavage site for cathepsin B. Subsequent exposure to peptide-specific T cells resulted in the generation of a T cell response to the B-LCL.
[0229] Rituximab was conjugated with VLEETSVMLASGFKGC (SEQ ID NO: 8), BRVLBBYVLASGFKGC (SEQ ID NO: 9) where B is amino butyric acid, a homolog for cysteine, and YILEETSVMASGFKGC (SEQ ID NO: 10) at equal amounts of peptide. B-LCL cells were labeled with Rituximab TPEC and after incubation, washed to remove excess TPEC. Labeled target cells were incubated overnight at 37.degree. C. in the presence of VLE-specific, CRV-specific or YIL-specific T cells. Supernatant from the cultures was used to assess the presence of IFN-.gamma..
[0230] Results are shown in FIG. 4. There was IFN-.gamma. released by all of the different peptide-specific T cells in response to the TPEC-labeled target cells suggesting that each of the three conjugated peptides could be released from the antibody and used to stimulate peptide-specific T cells.
Example 8. TPEC Compositions Comprising a Single Peptide Having Two Different Cytomegalovirus Peptides and Evaluation in B-Lymphoblastoid Cells
[0231] We contacted B-lymphoblastoid cells (B-LCL) with an agent comprising Rituximab conjugated to a single peptide which comprised two different cytomegalovirus peptides: (i) NLVPMVATV (SEQ ID NO: 1) and RPHERNGFTVL (SEQ ID NO: 2) or (ii) NLVPMVATV (SEQ ID NO: 1) and VLEETSVML (SEQ ID NO: 5) each separated from the other by a cleavage site for cathepsin B. Subsequent exposure to NLV peptide-specific T cells resulted in the generation of a T cell response to the B-LCL.
[0232] Rituximab was conjugated with the peptides (i) CGVANLVPMVATVAVAVLEETSVML (SEQ ID NO: 11), (ii) CVARPHERNGFTVLVANLVPMVATV (SEQ ID NO: 12) and (iii) CGVANLVPMVATVARPHERNGFTVL (SEQ ID NO: 13). Target cells were labeled with TPEC and washed after incubation to remove excess TPEC. Target cells were cultured overnight at 37.degree. C. in the presence of NLV-specific T cells and the supernatant assayed for the presence of IFN-.gamma. the following day.
[0233] NLV-specific T cells were able to recognize target cells labeled with each of the three TPECs tested as determined by IFN-.gamma. release. This demonstrates the ability of a peptide containing more than one cytomegalovirus derived peptide, separated by a protease cleavage site, to stimulate peptide-specific T cells. Furthermore, the position of the NLVPMVATV (SEQ ID NO: 1) peptide in the polytope appears to affect the magnitude of the response with the NLV peptide being furthest from the antibody CVARPHERNGFTVLVANLVPMVATV (SEQ ID NO: 12) producing the largest response. However, both polytope peptides containing the NLV peptide closest to the antibody CGVANLVPMVATVAVAVLEETSVML (SEQ ID NO: 11) and CGVANLVPMVATVARPHERNGFTVL (SEQ ID NO: 13) also demonstrate a T cell response above background.
[0234] Results are shown in FIG. 5.
Example 9. Preparation of a TPEC Comprising Polytope Branched Peptide
[0235] A TPEC comprising polytope branched peptide was prepared. A stem peptide that contains four azidonorleucine residues was provided along with peptides (branches) that contain propargyl glycine at the amino terminus (FIG. 6A). Each branched peptide has a NH.sub.2 cap on the C-terminal end which can aid in peptide serum stability. The sequence of the stem peptide was SEEZSEEZSEEZSEEZ, wherein Z is Azidonorleucine (SEQ ID NO: 168). For Branched TPEC 1, the sequences of the branched peptides were BKPAKFFRLTPRVTGGGAM-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 169), BKPAKFFRLRPHERNGFTVL-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 170), BKPAKFFRLRELRRI M-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 171), and BKPAKFFRLNLVPMVATV-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 172).
[0236] And for Branched TPEC 3, the sequences of the branched peptides were BAIPVSLRTPRVTGGGAM-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 173), BAIPVSLRRPHERNGFTVL-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 174), BAIPVSLRELRRI M-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 175), and BAIPVSLVTEHDTLLY-nh.sub.2, wherein B is propargyl glycine (SEQ ID NO: 176).
[0237] Incubation of equimolar concentrations of the branches with the stem peptide in DMSO in the presence of 10 mg in 4 ml of the CuSO.sub.4.5H.sub.2O catalyst mixed with 10 mg in 4 ml ascorbic acid overnight resulted in the formation of the branched peptide used for conjugation. The final concentration of DMSO for the reaction was 50%. The branched peptides were purified using HPLC and verified using mass spectrometry (FIG. 6B).
[0238] The branched peptide was conjugated to either Rituximab or Cetuximab by firstly reducing the antibody in the presence of approximately 0.1 mM (tris(2-carboxyethyl)phosphine (TCEP) for 90 minutes. The branched peptide is then added at 3.times. the concentration as TCEP for 60 minutes and N-acetyl cysteine is added to quench the free peptide using the same concentration as the branched peptide for 30 minutes. The free peptide is removed using protein A sepahrose beads to bind the TPEC whilst the free peptide is washed off. The TPEC is then collected from the protein A beads by eluting using 0.1M glycine and buffered using 1 m Tris pH8.
Example 10. In Vitro Targeting of a Transformed B Cell Line Using TPEC Comprising Polytope Branched Peptide
[0239] Two agents comprised of Rituximab conjugated with a branched peptide consisting of four different viral epitopes, each linked to a single stem peptide. In Branched TPEC 1, each of the four peptides was separated from the stem peptide by an ADAM28 protease cleavage sequence. In Branched TPEC 3, each of the four peptides was separated from the stem peptide by an MMP2 cleavage sequence. The target cells were labelled with the Rituximab-TPEC before being incubated with CD8 T cells specific for one of two different viral epitopes. Specifically, TPR T cells were specific for the viral epitope TPRVTGGGAM (SEQ ID NO: 49) and this epitope was present in branches 1-1 and 3-1. RPH T cells were specific for the viral epitope RPHERNGFTVL (SEQ ID NO: 2) and this epitope was present in branches 1-2 and 3-2.
[0240] Upon binding malignant cells (transformed B cell line), viral epitopes were released from the stem peptide by proteolytic cleavage releasing the viral epitopes TPRVTGGGAM (SEQ ID NO: 49), RPHERNGFTVL (SEQ ID NO: 2), ELRRKMMYM (SEQ ID NO: 24) and VTEHDTLLY (SEQ ID NO: 53). Upon release, the peptides were presented on MHC class I molecules on the malignant cells where they could be recognized by the CD8 T cells. Recognition of malignant cells by the CD8 T cells was measured by release of IFN-.gamma. by the T cells which can be used as a proxy for T cell killing
[0241] Results are shown in FIGS. 7A and 7B. Target lymphoma cells labeled with branched peptide TPEC are recognized by T cells specific for the RPHERNGFTVL (SEQ ID NO: 2) and TPRVTGGGAM (SEQ ID NO: 49) peptides, especially using the Branch 1 TPEC. Untreated target cells were not recognized by the peptide-specific T cells (negative control) whereas free peptide pulsed target cells were strongly recognized by T cells (positive control).
[0242] These results demonstrate that branched TPECs can obtain positive results in a widely-recognized model for retargeting a T cell response.
Example 11. In Vitro Targeting of Ovarian Carcinoma Cell Line Using TPEC Comprising Polytope Branched Peptide
[0243] Two agents comprised of Cetuximab conjugated with a branched peptide consisting of four different viral epitopes, all linked to a single stem peptide was provided. In Branched TPEC 1, each of the four peptides was separated from the stem peptide by an ADAM28 protease cleavage sequence. In Branched TPEC 3, each of the four peptides was separated from the stem peptide by an MMP2 cleavage sequence. The target cells were labelled with the Cetuximab-TPEC before being incubated with CD8 T cells specific for one of two different viral epitopes. Specifically, TPR T cells were specific for the viral epitope TPRVTGGGAM (SEQ ID NO: 49) and this epitope was present in branches 1-1 and 3-1. RPH T cells were specific for the viral epitope RPHERNGFTVL (SEQ ID NO: 2) and this epitope was present in branches 1-2 and 3-2.
[0244] Upon binding malignant cells (ovarian carcinoma cell line), viral epitopes were released from the stem peptide by proteolytic cleavage releasing the viral epitopes TPRVTGGGAM (SEQ ID NO: 49), RPHERNGFTVL (SEQ ID NO: 2), ELRRKMMYM (SEQ ID NO: 24) and VTEHDTLLY (SEQ ID NO: 53). Upon release, the peptides were presented on MHC class I molecules on the malignant cells where they could be recognized by the CD8 T cells. Recognition of malignant cells by the CD8 T cells was measured by release of IFN-.gamma. by the T cells.
[0245] Results are shown in FIGS. 8A and 8B. Target lymphoma cells labeled with branched peptide TPEC are recognized by T cells specific for the RPHERNGFTVL (SEQ ID NO: 2) and TPRVTGGGAM (SEQ ID NO: 49) peptides, especially using the Branch 1 TPEC. Untreated target cells were not recognized by the peptide-specific T cells (negative control) whereas free peptide pulsed target cells were strongly recognized by T cells (positive control).
[0246] These results demonstrate that branched TPECs can obtain positive results in a widely-recognized model for retargeting a T cell response.
Example 12. In Vitro Targeting of Ovarian Carcinoma Cell Line Using TPEC Containing a Bundled Polytope Domain
[0247] DNA for an agent comprising Cetuximab containing an additional fibronectin type 3 domain (from fibronectin 1 protein) attached to each heavy chain (depicted in FIG. 2B) was synthesized (FIG. 9A). The fibronectin type 3 domain was mutated at the "BC", "DE" and "FG" loops to contain the viral epitopes TPRVTGGGAM (SEQ ID NO: 49), NLVPMVATV (SEQ ID NO: 1), and VLEETSVML (SEQ ID NO: 5). The viral epitopes were separated from the fibronectin type 3 domain by MMP2 protease cleavage sequences on both sides of the CMV epitope as depicted: [0248] Cetuximab heavy chain--Domain--MMP2--Epitope--MMP2--Domain--MMP2--Epitope--MMP2--Domain--- MMP2--Epitope
[0249] This would allow release of the viral epitopes from the bundled domain and targeting agent (Cetuximab) after proteolytic cleavage by MMP2. The DNA for the bundled domain TPEC was transfected into Expi293 cells and the supernatant harvested 10 days post transfection. The bundled domain TPEC was purified from the supernatant using protein A sepharose beads. The target cells, ovarian carcinoma cell line, were labelled with the purified Cetuximab-TPEC with excess, unbound TPEC washed off. The labelled target cells were then incubated overnight in the presence of NLV-specific T cells.
[0250] Upon binding malignant cells (ovarian carcinoma cell line), viral epitopes were released from the additional protein domain by proteolytic cleavage. The released peptides were presented on MHC class I molecules on the malignant cells where they could be recognized by the CD8+ T cells. Recognition of malignant cells by the CD8+ T cells was measured by release of IFN-.gamma. by the T cells.
[0251] Results are shown in FIG. 9. Target ovarian carcinoma cells labeled with bundled domain TPEC were recognized by T cells specific for the NLVPMVATV (SEQ ID NO: 1) peptide from four different bundled domain TPEC preparations. Untreated target cells were not recognized by the peptide-specific T cells (negative control) whereas free peptide pulsed target cells were strongly recognized by T cells (positive control).
[0252] These results demonstrate that bundled domain TPECs can obtain positive results in a widely-recognized model for retargeting a T cell response.
Example 13. In Vivo Targeting of B Cell Lymphoma Using the Separately Conjugated T-Cell Epitopes (i.e., "Hairbrush") (e.g., Chronic Lymphocytic Leukaemia)
[0253] An agent comprising Rituximab or an anti-CD22 antibody is conjugated with a peptide mixture containing equal parts of five different cytomegalovirus-derived peptides each containing a protease cleavage site specific for cathepsin B (i) NLVPMVATVASGV{CIT}GC (SEQ ID NO: 3), (ii) VLEETSVMLASGFKGC (SEQ ID NO: 8), (iii) RPHERNGFTVLASGFKGC (SEQ ID NO: 4), (iv) YILEETSVMASGFKGC (SEQ ID NO: 10) and (v) BRVLBBYVLASGFKGC (SEQ ID NO: 9). The patient is infused with the agent, which targets all B cells, healthy and malignant. Upon binding malignant cells, the agent comes into contact with proteases whereby cleavage of the protease recognition domain releases the T-cell epitopes (i) NLVPMVATV (SEQ ID NO: 1), (ii) VLEETSVML (SEQ ID NO: 5), (iii) RPHERNGFTVL (SEQ ID NO: 2), (iv) YILEETSVM (SEQ ID NO: 7) and (v) BRVLBBYVL (SEQ ID NO: 6), which subsequently bind to HLA molecules on the surface of the malignant B cell. The malignant B cells expressing the T-cell epitopes are targeted by the host immune system for cytolysis by T cells.
Example 14. In Vivo Targeting of B-Cell Lymphoma Using a TPEC Comprising Polytope (e.g., Chronic Lymphocytic Leukaemia)
[0254] An agent comprising Rituximab or an anti-CD22 antibody is conjugated with a peptide containing four different cytomegalovirus-derived epitopes (i) CGSFRVTEHDTLLYGSFRRPHERNGFTVLGSFRELKRKMIYMGSFRN LVPMVATV (SEQ ID NO: 14). Each of the four epitopes is separated from the next one by a protease cleavage site specific for cathepsin B in the conformation targeting agent--C-E.sub.1-C-E.sub.2-C-E.sub.3-C-E.sub.4 where C is protease cleavage and E is epitope. The patient is infused with the agent, which targets all B cells, healthy and malignant. Upon binding malignant cells, the agent comes into contact with proteases whereby cleavage of the protease recognition domain releases the T-cell epitopes (i) NLVPMVATV (SEQ ID NO: 1), (ii) VTEHDTLLY (SEQ ID NO: 53), (iii) RPHERNGFTVL (SEQ ID NO: 2), and (iv) ELKRKMIYM (SEQ ID NO: 23), which subsequently bind to HLA molecules on the surface of the malignant B cell. The cells expressing the T-cell epitopes are then targeted by the host immune system for cytolysis by T cells.
Example 15. In Vivo Targeting of B Cell Lymphoma Using the Separately Conjugated T-Cell Epitopes (I.E., "Hairbrush") (e.g., Chronic Lymphocytic Leukaemia)
[0255] An agent comprising Rituximab or an anti-CD22 antibody is conjugated with a peptide mixture containing equal parts of five different cytomegalovirus-derived peptides each containing a protease cleavage site specific for either ADAM28 or cathepsin D (i) CKPAKFFRLNLVPMVATV (SEQ ID NO: 58), (ii) CKPAKFFRLRPHERNGFTVL (SEQ ID NO: 59), (iii) CPRSFFRLGKVLEETSVML (SEQ ID NO: 60), (iv) CKPAKFFRLELKRKMIYM (SEQ ID NO: 61) and (v) CPRSFFRLGKQIKVRVDMV (SEQ ID NO: 62). The patient is infused with the agent, which targets all B cells, healthy and malignant. Upon binding malignant cells, the agent comes into contact with proteases whereby cleavage of the protease recognition domain releases the T-cell epitopes (i) NLVPMVATV (SEQ ID NO: 1), (ii) RPHERNGFTVL (SEQ ID NO: 2), (iii) VLEETSVML (SEQ ID NO: 5), (iv) ELKRKMIYM (SEQ ID NO: 23) and (v) QIKVRVDMV (SEQ ID NO: 38), which subsequently bind to HLA molecules on the surface of the malignant B cell. The malignant B cells expressing the T-cell epitopes on their HLA molecules at the cell surface are targeted by the host immune system for cytolysis by T cells.
Example 16. In Vivo Targeting of B-Cell Lymphoma Using a TPEC Comprising Polytope (e.g., Chronic Lymphocytic Leukaemia)
[0256] An agent comprising Rituximab or an anti-CD22 antibody is conjugated with a peptide containing four different cytomegalovirus-derived epitopes (i) CGSKPAKFFRLYSEHPTFTSQYGSPRSFFRLGKTPRVTGGGAMG SKPAKFFRLQIKVRVDMVGSPRSFFRLGKELRRKMMYM (SEQ ID NO: 63). Each of the four epitopes is separated from the next one by a protease cleavage site specific for either ADAM28 or cathepsin D in a potential conformation, but not limited to the conformation, Targeting agent--C.sub.i-E.sub.1-C.sub.ii-E.sub.2-C.sub.iE.sub.3 C.sub.ii-E.sub.4 where C is protease cleavage and E is epitope. The patient is infused with the agent, which targets all B cells, healthy and malignant. Upon binding malignant cells, the agent comes into contact with proteases whereby cleavage of the protease recognition domain releases the T-cell epitopes (i) YSEHPTFTSQY (SEQ ID NO: 55), (ii) TPRVTGGGAM (SEQ ID NO: 49), (iii) QIKVRVDMV (SEQ ID NO: 38), and (iv) ELRRKMMYM (SEQ ID NO: 24), which subsequently bind to HLA molecules on the surface of the malignant B cell. The cells expressing the T-cell epitopes are then targeted by the host immune system for cytolysis by T cells.
Example 17. Embodiments Described Herein
[0257] Various embodiments are described in the following nonlimiting items.
[0258] Item 1. A composition for retargeting an immune response to unwanted cells comprising a TPEC wherein: [0259] a. T is a targeting moiety that is capable of targeting unwanted cells; [0260] b. L is at least one linker capable of linkage to T, where L may be a peptide bond, at least one peptide, or a chemical linker; [0261] c. C is at least one cleavage site [0262] i. cleaved by an enzyme expressed by the unwanted cells; [0263] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0264] iii. cleaved by a complement-dependent cleavage reaction; or [0265] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and [0266] d. E is at least one T-cell epitope, wherein the L, C, and E moieties are arranged in at least one of the following patterns: [0267] i. a plurality of L-C-E, each attached separately to T, [0268] ii. at least one of L-(C-E)n, n is an integer of at least 2 (optionally from about 2 to 50) and with each C-E attached to the L in parallel, and/or [0269] iii. at least one of L-(C-E)n, n is an integer of at least 2 (optionally from about 2 to 50) and with each C-E attached to the L in series.
[0270] Item 2. A composition for retargeting an immune response to unwanted cells comprising a TPEC comprising the formula T-(L-C-E).sub.n or T-(L-C.sub.i-E.sub.i).sub.n, wherein: [0271] a. T is a targeting moiety that is capable of targeting unwanted cells; [0272] b. L is a linker capable of chemical linkage to T; [0273] c. C is a cleavage site [0274] i. cleaved by an enzyme expressed by the unwanted cells; [0275] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0276] iii. cleaved by a complement-dependent cleavage reaction; or [0277] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and [0278] d. E is a T-cell epitope wherein n is an integer of at least 2 (optionally from about 2 to 50), i is an integer of at least 1 (optionally from about 1 to 50), and j is an integer of at least 2 (optionally from about 1 to 50).
[0279] Item 3. A composition for retargeting an immune response to unwanted cells comprising a TPEC comprising the formula T-L-(C.sub.i-E.sub.i).sub.n, wherein: [0280] a. T is a targeting moiety that is capable of targeting unwanted cells; [0281] b. L is an optional linker capable of chemical or peptide linkage to T; [0282] c. C is a cleavage site [0283] i. cleaved by an enzyme expressed by the unwanted cells; [0284] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0285] iii. cleaved by a complement-dependent cleavage reaction; or [0286] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC; and [0287] d. E is a T-cell epitope; wherein n is an integer of at least 2 (optionally from about 2 to 50), i is an integer of at least 1 (optionally from about 1 to 50), and j is an integer of at least 1 (optionally from about 1 to 50).
[0288] Item 4. A composition for retargeting an immune response to unwanted cells comprising a TPEC having:
[0289] a. a targeting moiety that is capable of targeting unwanted cells;
[0290] b. a plurality of more than 10 T-cell epitopes conjugated to the targeting moiety with at least one cleavage site, wherein the cleavage site is [0291] i. cleaved by an enzyme expressed by the unwanted cells; [0292] ii. cleaved through a pH-sensitive cleavage reaction inside the unwanted cell; [0293] iii. cleaved by a complement-dependent cleavage reaction; or [0294] iv. cleaved by a protease that is colocalized to the unwanted cell by a targeting moiety that is the same or different from the targeting moiety in the TPEC.
[0295] Item 5. The composition of any one of items 1, 2, or 4, wherein the plurality of T-cell epitopes are individually conjugated to the targeting moiety, each T-cell epitope by a cleavage site.
[0296] Item 6. The composition of any one of items 1, 3, or 4, wherein the plurality of T-cell epitopes are conjugated as at least one bundle to the targeting moiety by one cleavage site.
[0297] Item 7. The composition of item 6, wherein the T-cell epitopes within a bundle have cleavage sites between them.
[0298] Item 8. The composition of any one of items 1, 3-4, 6-7, wherein at least two groups of a plurality of T-cell epitopes are conjugated to the targeting moiety, each group by one cleavage site.
[0299] Item 9. The composition of any one of items 1-8, wherein the plurality of T-cell epitopes are not all identical.
[0300] Item 10. The composition of any one of items 1-8, wherein the plurality of T-cell epitopes are the same.
[0301] Item 11. The composition of any one of items 1-8, wherein some the T-cell epitopes are the same and further wherein some are different.
[0302] Item 12. The composition of any one of items 1-11, wherein the agent has a plurality of cleavage sites that are the same.
[0303] Item 13. The composition of any one of items 1-11, wherein the agent has a plurality of cleavage sites that are not all identical.
[0304] Item 14. The composition of any one of items 1, 3-4, and 6-13, wherein the at least one cleavage site and the plurality of T-cell epitopes are conjugated to the targeting agent using at least one peptide bond.
[0305] Item 15. The composition of any one of items 1-13, wherein the at least one cleavage site and the plurality of T-cell epitopes are conjugated to the targeting agent through at least one bond other than a peptide bond.
[0306] Item 16. The composition of item 15, wherein the at least one cleavage site and the plurality of T-cell epitopes are conjugated to the targeting agent through a Sulfo-SMCC, SMCC, or maleimide linkage.
[0307] Item 17. The composition of any one of items 1-16 wherein at least one T-cell epitope is an MHC Class I restricted peptide.
[0308] Item 18. The composition of any one of items 1-17, wherein at least one T-cell epitope is an MHC Class II restricted peptide.
[0309] Item 19. The composition of any one of items 1-18, wherein the plurality of T-cell epitopes are from about 7 to about 14 amino acids in length.
[0310] Item 20. The composition of any one of items 1-19, wherein the plurality of T cell epitopes are about 8 to 13, about 9 to 12, about 9, or about 10 amino acids in length.
[0311] Item 21. The composition of any one of items 1-20, wherein the agent comprises about or at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 20, 30, 40, 50, 60, 70, 80, 90, or 100 T cell epitopes, optionally from 2 to 100 T cell epitopes.
[0312] Item 22. The composition of any one of items 1-21, wherein the T-cell epitopes are chosen from CMV, influenza, EBV, hepatitis, chicken pox, mumps, measles, diphtheria, Haemophilus influenzae, rubella, pertussis, polio, pneumococcus, rotavirus, tetanus, vaccinia, and yellow fever T-cell epitopes.
[0313] Item 23. The composition of any one of items 1-22, wherein the composition comprises T-cell epitopes from at least two different infectious agents.
[0314] Item 24. The composition of any one of items 1-23, wherein the TPEC comprises at least one CMV T-cell epitope.
[0315] Item 25. The composition of any one of items 1-9 and 11-24, wherein using a plurality of different T-cell epitopes allows the agent to stimulate a T-cell response in at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, or 90% of the human population.
[0316] Item 26. The composition of any one of items 1-25, wherein the T-cell epitopes are chosen from HLA-A, HLA-B, HLA-C, HLA-E, HLA-F, HLA-G, CD1d, and MR1.
[0317] Item 27. The composition of any one of items 1-26, wherein the T-cell epitopes are chosen from HLA-A*01, HLA-A*02, HLA-A*03, HLA-A*11, HLA-B*44, HLA-B*07, HLA-B*08, HLA-B*15, HLA-B*35, HLA-B*40, HLA-C*07, HLA-C*03, HLA-C*05, HLA-C*04, HLA-C*06, and HLA-E*0101 restricted antigens.
[0318] Item 28. The composition of any one of items 1-9 and 11-27, wherein the composition comprises at least the following T-cell epitopes: HLA-A*02, HLA-A*01, and HLA-A*03.
[0319] Item 29. The composition of any one of items 1-28, wherein having multiple copies of the same T-cell epitope increases an immune response against the unwanted cell when the agent is administered to a subject.
[0320] Item 30. The composition of any one of items 1-29, wherein the T-cell epitopes comprise at least one of SEQ ID NOS: 1-2, 5-7, 15-57.
[0321] Item 31. The composition of any one of items 1-30, wherein the targeting moiety targets the unwanted cells by specifically binding to the unwanted cells.
[0322] Item 32. The composition of any one of items 1-32, wherein the targeting moiety specifically binds the unwanted cells even while the plurality of T-cell epitopes are conjugated to the targeting moiety.
[0323] Item 33. The composition of any one of items 1-32, wherein the targeting moiety is an antibody or functional part thereof.
[0324] Item 34. The composition of any one of items 1-33, wherein the T-cell epitopes are flanked on one or both ends by at least one human protein domain.
[0325] Item 35. The composition of items 34, wherein at least one human protein domain is a beta barrel or a coiled coil.
[0326] Item 36. The composition of any one of items 34-35, wherein the human protein domain is a CL, CH2, and/or CH3 domain.
[0327] Item 37. The composition of any one of items 34-35, wherein the human protein domain is a FN3 domain, beta-sandwich, Lipocalin, EETI-II/AGRP, Kunitz domain (BPTI), Thioredoxin, Protein A, Ankyrin, gamma-B-crystallin/ubiquitin, CTLD3/Tetranexin LDLR-A, human serum albumin, immunoglobulin CH2 domain, or camelid VHH domain
[0328] Item 38. The composition of any one of item 34-35 or 37, wherein the human protein domain is not a human immunoglobulin protein domain.
[0329] Item 39. The composition of any one of items 34-38, wherein the human protein domain displays the T-cell epitopes on its three-dimensional surface.
[0330] Item 40. The composition of any one of items 1-39, wherein the cleavage site is capable of being cleaved outside the unwanted cells.
[0331] Item 41. The composition of any one of items 1-40, wherein the enzyme expressed by the unwanted cells is a protease.
[0332] Item 42. The composition of any one of items 1-41, wherein the unwanted cells are cancer cells.
[0333] Item 43. A composition comprising a plurality of different TPECs of any one of items 1-42, wherein each targeting agent is conjugated to a plurality of identical T-cell epitopes.
[0334] Item 44. A composition comprising a plurality of compositions of any one of items 1-42, wherein at least some of the targeting agents in the composition are conjugated to a plurality of T-cell epitopes that are not identical.
[0335] Item 45. A composition comprising a plurality of compositions of item 44, wherein at least some of the compositions are not identical.
[0336] Item 46. A composition comprising a plurality of compositions of any one of items 43-45, wherein the composition comprises at least about 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, or more different TPECs.
[0337] Item 47. A pharmaceutical composition comprising a composition of any one of items 1-46 and a pharmaceutically acceptable carrier.
[0338] Item 48. A method of treating a disease in a patient characterized by the presence of unwanted cells comprising administering a composition of any one of items 1-47 to the patient.
[0339] Item 49. A method of retargeting an immune response of a patient to unwanted cells comprising administering a composition of any one of items 1-47 to the patient.
[0340] Item 50. The method of any of items 48-49, wherein the patient does not develop an immune response against the composition sufficient to inactivate the composition.
[0341] Item 51. The method of any one of items 48-50, wherein the patient receives multiple doses of the composition over at least 30, 45, 60, 75, 90, 120, 150, or more days or on an ongoing basis.
[0342] Item 52. The method of any one of items 48-50, wherein the patient receives multiple doses of the composition over at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 months or on an ongoing basis.
[0343] Item 53. The method of any one of items 48-52, wherein the composition can be administered to a patient in relapse who received the composition for an earlier round of therapy.
[0344] Item 54. The method of any one of items 48-53, wherein the patient has cancer.
EQUIVALENTS
[0345] The foregoing written specification is considered to be sufficient to enable one skilled in the art to practice the embodiments. The foregoing description and Examples detail certain embodiments and describes the best mode contemplated by the inventors. It will be appreciated, however, that no matter how detailed the foregoing may appear in text, the embodiment may be practiced in many ways and should be construed in accordance with the appended claims and any equivalents thereof.
[0346] As used herein, the term about refers to a numeric value, including, for example, whole numbers, fractions, and percentages, whether or not explicitly indicated. The term about generally refers to a range of numerical values (e.g., +/-5-10% of the recited range) that one of ordinary skill in the art would consider equivalent to the recited value (e.g., having the same function or result). When terms such as at least and about precede a list of numerical values or ranges, the terms modify all of the values or ranges provided in the list. In some instances, the term about may include numerical values that are rounded to the nearest significant figure.
Sequence CWU 1
1
18819PRTHuman cytomegalovirus 1Asn Leu Val Pro Met Val Ala Thr Val1
5211PRTHuman cytomegalovirus 2Arg Pro His Glu Arg Asn Gly Phe Thr
Val Leu1 5 10316PRTArtificial SequenceCMV TCE and cleavage site
from capthepsin BMISC_FEATURE(13)..(14)Cleavage
siteMISC_FEATURE(14)..(14)X = citrulline 3Asn Leu Val Pro Met Val
Ala Thr Val Ala Ser Gly Val Xaa Gly Cys1 5 10 15418PRTArtificial
SequenceCMV TCE and cleavage site from capthepsin
BMISC_FEATURE(12)..(18)Cleavage site 4Arg Pro His Glu Arg Asn Gly
Phe Thr Val Leu Ala Ser Gly Phe Lys1 5 10 15Gly Cys59PRTHuman
cytomegalovirus 5Val Leu Glu Glu Thr Ser Val Met Leu1
569PRTArtificial SequenceCMV TCE, (IE1, 309-317, HLA-C7),
(Peptidomimetic of SEQ ID NO 17)MISC_FEATURE(1)..(9)X =
alpha-aminobutyric acid 6Xaa Arg Val Leu Xaa Xaa Tyr Val Leu1
579PRTHuman cytomegalovirus 7Tyr Ile Leu Glu Glu Thr Ser Val Met1
5816PRTArtificial SequenceCMV TCE and cleavage site from capthepsin
BMISC_FEATURE(10)..(16)Cleavage site 8Val Leu Glu Glu Thr Ser Val
Met Leu Ala Ser Gly Phe Lys Gly Cys1 5 10 15916PRTArtificial
SequenceCMV TCE and cleavage site from capthepsin
BMISC_FEATURE(1)..(16)X = alpha-aminobutyric
acidMISC_FEATURE(10)..(16)Cleavage site 9Xaa Arg Val Leu Xaa Xaa
Tyr Val Leu Ala Ser Gly Phe Lys Gly Cys1 5 10 151016PRTArtificial
SequenceCMV TCE and cleavage site from capthepsin
BMISC_FEATURE(10)..(16)Cleavage site 10Tyr Ile Leu Glu Glu Thr Ser
Val Met Ala Ser Gly Phe Lys Gly Cys1 5 10 151125PRTArtificial
SequencePolytope containing two different CMV-derived epitopes
separated by cathepsin B cleavage siteMISC_FEATURE(1)..(4)Cleavage
siteMISC_FEATURE(15)..(16)Cleavage site 11Cys Gly Val Ala Asn Leu
Val Pro Met Val Ala Thr Val Ala Val Ala1 5 10 15Val Leu Glu Glu Thr
Ser Val Met Leu 20 251225PRTArtificial SequencePolytope containing
two different CMV-derived epitopes separated by cathepsin B
cleavage siteMISC_FEATURE(1)..(3)Cleavage
siteMISC_FEATURE(15)..(16)Cleavage site 12Cys Val Ala Arg Pro His
Glu Arg Asn Gly Phe Thr Val Leu Val Ala1 5 10 15Asn Leu Val Pro Met
Val Ala Thr Val 20 251325PRTArtificial SequencePolytope containing
two different CMV-derived epitopes separated by cathepsin B
cleavage siteMISC_FEATURE(1)..(4)Cleavage
siteMISC_FEATURE(14)..(14)Cleavage site 13Cys Gly Val Ala Asn Leu
Val Pro Met Val Ala Thr Val Ala Arg Pro1 5 10 15His Glu Arg Asn Gly
Phe Thr Val Leu 20 251455PRTArtificial SequencePolytope containing
five different CMV-derived epitopes separated by cathepsin B
cleavage sitesMISC_FEATURE(2)..(5)Cleavage
siteMISC_FEATURE(15)..(18)Cleavage
siteMISC_FEATURE(30)..(33)Cleavage
siteMISC_FEATURE(43)..(46)Cleavage site 14Cys Gly Ser Phe Arg Val
Thr Glu His Asp Thr Leu Leu Tyr Gly Ser1 5 10 15Phe Arg Arg Pro His
Glu Arg Asn Gly Phe Thr Val Leu Gly Ser Phe 20 25 30Arg Glu Leu Lys
Arg Lys Met Ile Tyr Met Gly Ser Phe Arg Asn Leu 35 40 45Val Pro Met
Val Ala Thr Val 50 551510PRTHuman cytomegalovirus 15Ala Thr Thr Phe
Leu Gln Thr Met Leu Arg1 5 101612PRTHuman cytomegalovirus 16Cys Pro
Ser Gln Glu Pro Met Ser Ile Tyr Val Tyr1 5 10179PRTHuman
cytomegalovirus 17Cys Arg Val Leu Cys Cys Tyr Val Leu1 5189PRTHuman
cytomegalovirus 18Cys Val Glu Thr Met Cys Asn Glu Tyr1 5199PRTHuman
cytomegalovirus 19Asp Glu Leu Arg Arg Lys Met Met Tyr1
52010PRTHuman cytomegalovirus 20Asp Thr Pro Val Leu Pro His Glu Thr
Arg1 5 10219PRTHuman cytomegalovirus 21Glu Glu Ala Ile Val Ala Tyr
Thr Leu1 5229PRTHuman cytomegalovirus 22Glu Phe Phe Asp Ala Asn Asp
Ile Tyr1 5239PRTHuman cytomegalovirus 23Glu Leu Lys Arg Lys Met Ile
Tyr Met1 5249PRTHuman cytomegalovirus 24Glu Leu Arg Arg Lys Met Met
Tyr Met1 5259PRTHuman cytomegalovirus 25Glu Val Ile Ser Val Met Lys
Arg Arg1 5269PRTHuman cytomegalovirus 26Phe Glu Gln Pro Thr Glu Thr
Pro Pro1 52711PRTHuman cytomegalovirus 27Phe Pro Lys Thr Thr Asn
Gly Cys Ser Gln Ala1 5 10288PRTHuman cytomegalovirus 28Phe Pro Thr
Lys Asp Val Ala Leu1 52911PRTHuman cytomegalovirus 29Phe Thr Ser
Gln Tyr Arg Ile Gln Gly Lys Leu1 5 103011PRTHuman cytomegalovirus
30Phe Val Phe Pro Thr Lys Asp Val Ala Leu Arg1 5 103110PRTHuman
cytomegalovirus 31Ile Ile Tyr Thr Arg Asn His Glu Val Lys1 5
10329PRTHuman cytomegalovirus 32Ile Pro Ser Ile Asn Val His His
Tyr1 5339PRTHuman cytomegalovirus 33Lys Ala Arg Asp His Leu Ala Val
Leu1 5349PRTHuman cytomegalovirus 34Lys Glu Val Asn Ser Gln Leu Ser
Leu1 5359PRTHuman cytomegalovirus 35Lys Arg Lys Met Ile Tyr Met Cys
Tyr1 5369PRTHuman cytomegalovirus 36Met Leu Asn Ile Pro Ser Ile Asn
Val1 5379PRTHuman cytomegalovirus 37Asn Val Arg Arg Ser Trp Glu Glu
Leu1 5389PRTHuman cytomegalovirus 38Gln Ile Lys Val Arg Val Asp Met
Val1 53910PRTHuman cytomegalovirus 39Gln Pro Ser Leu Ile Leu Val
Ser Gln Tyr1 5 104011PRTHuman cytomegalovirus 40Gln Thr Val Thr Ser
Thr Pro Val Gln Gly Arg1 5 10419PRTHuman cytomegalovirus 41Gln Tyr
Asp Val Pro Ala Ala Leu Phe1 54210PRTHuman cytomegalovirus 42Gln
Tyr Val Lys Val Tyr Leu Glu Ser Phe1 5 10439PRTHuman
cytomegalovirus 43Arg Gly Asp Pro Phe Asp Lys Asn Tyr1 5449PRTHuman
cytomegalovirus 44Arg Ile Phe Ala Glu Leu Glu Gly Val1 5459PRTHuman
cytomegalovirus 45Arg Ile Trp Cys Leu Val Val Cys Val1 5469PRTHuman
cytomegalovirus 46Arg Arg Ile Glu Glu Ile Cys Met Lys1 5479PRTHuman
cytomegalovirus 47Arg Arg Lys Met Met Tyr Met Cys Tyr1
54810PRTHuman cytomegalovirus 48Ser Glu His Pro Thr Phe Thr Ser Gln
Tyr1 5 104910PRTHuman cytomegalovirus 49Thr Pro Arg Val Thr Gly Gly
Gly Ala Met1 5 105011PRTHuman cytomegalovirus 50Thr Thr Val Tyr Pro
Pro Ser Ser Thr Ala Lys1 5 10519PRTHuman cytomegalovirus 51Thr Val
Arg Ser His Cys Val Ser Lys1 5529PRTHuman cytomegalovirus 52Val Leu
Ala Glu Leu Val Lys Gln Ile1 5539PRTHuman cytomegalovirus 53Val Thr
Glu His Asp Thr Leu Leu Tyr1 5549PRTHuman cytomegalovirus 54Val Tyr
Ala Leu Pro Leu Lys Met Leu1 55511PRTHuman cytomegalovirus 55Tyr
Ser Glu His Pro Thr Phe Thr Ser Gln Tyr1 5 105610PRTHuman
cytomegalovirus 56Tyr Thr Pro Asp Ser Thr Pro Cys His Arg1 5
10579PRTHuman cytomegalovirus 57Tyr Val Leu Glu Glu Thr Ser Val
Met1 55818PRTArtificial SequenceCMV TCE and cleavage site from
ADAM28MISC_FEATURE(2)..(9)Cleavage site 58Cys Lys Pro Ala Lys Phe
Phe Arg Leu Asn Leu Val Pro Met Val Ala1 5 10 15Thr
Val5920PRTArtificial SequenceCMV TCE and cleavage site from
ADAM28MISC_FEATURE(2)..(9)Cleavage site 59Cys Lys Pro Ala Lys Phe
Phe Arg Leu Arg Pro His Glu Arg Asn Gly1 5 10 15Phe Thr Val Leu
206019PRTArtificial SequenceCMV TCE and cleavage site from
capthepsin DMISC_FEATURE(2)..(10)Cleavage site 60Cys Pro Arg Ser
Phe Phe Arg Leu Gly Lys Val Leu Glu Glu Thr Ser1 5 10 15Val Met
Leu6118PRTArtificial SequenceCMV TCE and cleavage site from
ADAM28MISC_FEATURE(2)..(9)Cleavage site 61Cys Lys Pro Ala Lys Phe
Phe Arg Leu Glu Leu Lys Arg Lys Met Ile1 5 10 15Tyr
Met6219PRTArtificial SequenceCMV TCE and cleavage site from
capthepsin DMISC_FEATURE(2)..(10)Cleavage site 62Cys Pro Arg Ser
Phe Phe Arg Leu Gly Lys Gln Ile Lys Val Arg Val1 5 10 15Asp Met
Val6382PRTArtificial SequencePolytope comprising four different
cytomegalovirus epitopes, each separated by either an ADAM28 or
cathepsin D cleavage siteMISC_FEATURE(4)..(11)Cleavage
siteMISC_FEATURE(25)..(33)Cleavage
siteMISC_FEATURE(46)..(53)Cleavage
siteMISC_FEATURE(65)..(73)Cleavage site 63Cys Gly Ser Lys Pro Ala
Lys Phe Phe Arg Leu Tyr Ser Glu His Pro1 5 10 15Thr Phe Thr Ser Gln
Tyr Gly Ser Pro Arg Ser Phe Phe Arg Leu Gly 20 25 30Lys Thr Pro Arg
Val Thr Gly Gly Gly Ala Met Gly Ser Lys Pro Ala 35 40 45Lys Phe Phe
Arg Leu Gln Ile Lys Val Arg Val Asp Met Val Gly Ser 50 55 60Pro Arg
Ser Phe Phe Arg Leu Gly Lys Glu Leu Arg Arg Lys Met Met65 70 75
80Tyr Met649PRTEpstein Barr Virus 64Arg Pro Gln Lys Arg Pro Ser Cys
Ile1 5659PRTEpstein Barr Virus 65His Pro Val Gly Glu Ala Asp Tyr
Phe1 56611PRTEpstein Barr Virus 66His Pro Val Gly Glu Ala Asp Tyr
Phe Glu Tyr1 5 10679PRTEpstein Barr Virus 67Ile Pro Gln Cys Arg Leu
Thr Pro Leu1 5689PRTEpstein Barr Virus 68Val Leu Lys Asp Ala Ile
Lys Asp Leu1 56910PRTEpstein Barr Virus 69Tyr His Leu Ile Val Asp
Thr Asp Ser Leu1 5 107010PRTEpstein Barr Virus 70Asp Thr Pro Leu
Ile Pro Leu Thr Ile Phe1 5 10719PRTEpstein Barr Virus 71Arg Pro Thr
Glu Leu Gln Pro Thr Pro1 5729PRTEpstein Barr Virus 72Gln Ala Lys
Trp Arg Leu Gln Thr Leu1 5739PRTEpstein Barr Virus 73Ala Tyr Ser
Ser Trp Met Tyr Ser Tyr1 5748PRTEpstein Barr Virus 74Arg Tyr Ser
Ile Phe Phe Asp Tyr1 5759PRTEpstein Barr Virus 75Phe Leu Arg Gly
Arg Ala Tyr Gly Leu1 57610PRTEpstein Barr Virus 76Lys Arg Pro Pro
Ile Phe Ile Arg Arg Leu1 5 10779PRTEpstein Barr Virus 77Arg Pro Pro
Ile Phe Ile Arg Arg Leu1 5789PRTEpstein Barr Virus 78Leu Glu Lys
Ala Arg Gly Ser Thr Tyr1 5799PRTEpstein Barr Virus 79His Leu Ala
Ala Gln Gly Met Ala Tyr1 5809PRTEpstein Barr Virus 80Tyr Pro Leu
His Glu Gln His Gly Met1 5819PRTEpstein Barr Virus 81Val Phe Ser
Asp Gly Arg Val Ala Cys1 5829PRTEpstein Barr Virus 82Val Pro Ala
Pro Ala Gly Pro Ile Val1 5839PRTEpstein Barr Virus 83Ser Val Arg
Asp Arg Leu Ala Arg Leu1 5849PRTEpstein Barr Virus 84Arg Leu Arg
Ala Glu Ala Gln Val Lys1 58510PRTEpstein Barr Virus 85Val Gln Pro
Pro Gln Leu Thr Leu Gln Val1 5 10869PRTEpstein Barr Virus 86His Arg
Cys Gln Ala Ile Arg Lys Lys1 5879PRTEpstein Barr Virus 87Thr Tyr
Ser Ala Gly Ile Val Gln Ile1 58811PRTEpstein Barr Virus 88Arg Arg
Ala Arg Ser Leu Ser Ala Glu Arg Tyr1 5 10899PRTEpstein Barr Virus
89Val Ser Phe Ile Glu Phe Val Gly Trp1 59010PRTEpstein Barr Virus
90Ala Val Phe Asp Arg Lys Ser Asp Ala Lys1 5 10919PRTEpstein Barr
Virus 91Ile Val Thr Asp Phe Ser Val Ile Lys1 5929PRTEpstein Barr
Virus 92Ala Val Leu Leu His Glu Glu Ser Met1 59310PRTEpstein Barr
Virus 93Val Glu Ile Thr Pro Tyr Lys Pro Thr Trp1 5 10949PRTEpstein
Barr Virus 94Glu Gly Gly Val Gly Trp Arg His Trp1 59510PRTEpstein
Barr Virus 95Gln Asn Gly Ala Leu Ala Ile Asn Thr Phe1 5
109610PRTEpstein Barr Virus 96Leu Arg Gly Lys Trp Gln Arg Arg Tyr
Arg1 5 10979PRTEpstein Barr Virus 97Arg Arg Ile Tyr Asp Leu Ile Glu
Leu1 5988PRTEpstein Barr Virus 98His His Ile Trp Gln Asn Leu Leu1
59910PRTEpstein Barr Virus 99Glu Glu Asn Leu Leu Asp Phe Val Arg
Phe1 5 1010010PRTEpstein Barr Virus 100Leu Leu Asp Phe Val Arg Phe
Met Gly Val1 5 101019PRTEpstein Barr Virus 101Leu Asp Phe Val Arg
Phe Met Gly Val1 51029PRTEpstein Barr Virus 102Lys Glu His Val Ile
Gln Asn Ala Phe1 51039PRTEpstein Barr Virus 103Phe Arg Lys Ala Gln
Ile Gln Gly Leu1 51049PRTEpstein Barr Virus 104Gln Pro Arg Ala Pro
Ile Arg Pro Ile1 51059PRTEpstein Barr Virus 105Ser Leu Arg Glu Trp
Leu Leu Arg Ile1 51069PRTEpstein Barr Virus 106Phe Trp Leu Tyr Ile
Val Met Ser Asp1 510711PRTEpstein Barr Virus 107Phe Arg Arg Asp Leu
Leu Cys Pro Leu Gly Ala1 5 101089PRTEpstein Barr Virus 108Tyr Leu
Leu Glu Met Leu Trp Arg Leu1 51099PRTEpstein Barr Virus 109Tyr Leu
Gln Gln Asn Trp Trp Thr Leu1 51109PRTEpstein Barr Virus 110Thr Leu
Leu Val Asp Leu Leu Trp Leu1 511112PRTEpstein Barr Virus 111Asp Pro
His Gly Pro Val Gln Leu Ser Tyr Tyr Asp1 5 101129PRTEpstein Barr
Virus 112Met Gly Ser Leu Glu Met Val Pro Met1 511315PRTEpstein Barr
Virus 113Glu Asp Pro Tyr Trp Gly Asn Gly Asp Arg His Ser Asp Tyr
Gln1 5 10 1511414PRTEpstein Barr Virus 114Asn Pro Val Cys Leu Pro
Val Ile Val Ala Pro Tyr Leu Phe1 5 101159PRTEpstein Barr Virus
115Leu Pro Val Ile Val Ala Pro Tyr Leu1 51169PRTEpstein Barr Virus
116Pro Tyr Leu Phe Trp Leu Ala Ala Ile1 511714PRTEpstein Barr Virus
117Ala Ser Cys Phe Thr Ala Ser Val Ser Thr Val Val Thr Ala1 5
101189PRTEpstein Barr Virus 118Phe Thr Ala Ser Val Ser Thr Val Val1
51199PRTEpstein Barr Virus 119Ile Glu Asp Pro Pro Phe Asn Ser Leu1
51209PRTEpstein Barr Virus 120Arg Arg Arg Trp Arg Arg Leu Thr Val1
51219PRTEpstein Barr Virus 121Arg Arg Trp Arg Arg Leu Thr Val Cys1
512211PRTEpstein Barr Virus 122Arg Arg Leu Thr Val Cys Gly Gly Ile
Met Phe1 5 101239PRTEpstein Barr Virus 123Thr Val Cys Gly Gly Ile
Met Phe Leu1 512414PRTEpstein Barr Virus 124Met Phe Leu Ala Cys Val
Leu Val Leu Ile Val Asp Ala Val1 5 101259PRTEpstein Barr Virus
125Leu Ile Val Asp Ala Val Leu Gln Leu1 51269PRTEpstein Barr Virus
126Gly Leu Gly Thr Leu Gly Ala Ala Ile1 51279PRTEpstein Barr Virus
127Leu Leu Trp Thr Leu Val Val Leu Leu1 512811PRTEpstein Barr Virus
128Ser Ser Cys Ser Ser Cys Pro Leu Ser Lys Ile1 5 101299PRTEpstein
Barr Virus 129Ile Leu Leu Ala Arg Leu Phe Leu Tyr1 51309PRTEpstein
Barr Virus 130Phe Leu Tyr Ala Leu Ala Leu Leu Leu1 51319PRTEpstein
Barr Virus 131Thr Tyr Gly Pro Val Phe Met Cys Leu1 51329PRTEpstein
Barr Virus 132Cys Leu Gly Gly Leu Leu Thr Met Val1 513310PRTEpstein
Barr Virus 133Val Met Ser Asn Thr Leu Leu Ser Ala Trp1 5
101349PRTEpstein Barr Virus 134Leu Thr Ala Gly Phe Leu Ile Phe Leu1
51359PRTEpstein Barr Virus 135Leu Leu Ser Ala Trp Ile Leu Thr Ala1
513614PRTEpstein Barr Virus 136Leu Val Ser Asp Tyr Cys Asn Val Leu
Asn Lys Glu Phe Thr1 5 101379PRTEpstein Barr Virus 137Leu Val Ser
Asp Tyr Cys Asn Val Leu1 513810PRTEpstein Barr Virus 138Asp Tyr Cys
Asn Val Leu Asn Lys Glu Phe1 5 101399PRTEpstein Barr Virus 139Ala
Glu Asn Ala Gly Asn Asp Ala Cys1 514016PRTEpstein Barr Virus 140Ile
Ala Cys Pro Ile Val Met Arg Tyr Tyr Val Leu Asp His Leu Ile1 5 10
151419PRTEpstein Barr Virus 141Tyr Val Leu Asp His Leu Ile Val Val1
514215PRTEpstein Barr Virus 142Phe Phe Ile Gln Ala Pro Ser Asn Arg
Val Met Ile Pro Ala Thr1 5 10 151439PRTEpstein Barr Virus 143Ala
Thr Ile Gly Thr Ala Met Tyr Lys1 514415PRTEpstein Barr Virus 144Lys
His Ser Arg Val Arg Ala Tyr Thr Tyr Ser Lys Val Leu Gly1 5 10
1514515PRTEpstein Barr Virus 145Arg Ala Leu Ile Lys Thr Leu Pro Arg
Ala Ser Tyr Ser Ser His1 5 10 1514615PRTEpstein Barr Virus 146Glu
Arg Pro Ile Phe Pro His Pro Ser Lys Pro Thr Phe Leu Pro1 5 10
1514714PRTEpstein Barr Virus 147Gln Lys Glu Glu Ala Ala Ile Cys Gly
Gln Met Asp Leu Ser1 5 1014815PRTEpstein Barr Virus 148Glu Val Cys
Gln Pro Lys Arg Ile Arg Pro Phe His Pro Pro Gly1 5 10
1514913PRTEpstein Barr Virus 149Leu Pro Glu Pro Leu Pro Gln Gly Gln
Leu Thr Ala Tyr1 5 1015011PRTEpstein Barr Virus 150Glu Pro Leu Pro
Gln Gly Gln Leu Thr Ala Tyr1 5 101519PRTEpstein Barr Virus 151Ala
Pro Glu Asn Ala Tyr Gln Ala Tyr1 51529PRTEpstein Barr Virus 152Leu
Gln His Tyr Arg
Glu Val Ala Ala1 515312PRTEpstein Barr Virus 153Asp Ser Glu Leu Glu
Ile Lys Arg Tyr Lys Asn Arg1 5 1015416PRTEpstein Barr Virus 154Arg
Lys Cys Cys Arg Ala Lys Phe Lys Gln Leu Leu Gln His Tyr Arg1 5 10
151558PRTEpstein Barr Virus 155Arg Ala Lys Phe Lys Gln Leu Leu1
51569PRTEpstein Barr Virus 156Ser Glu Asn Asp Arg Leu Arg Leu Leu1
51579PRTInfluenza A Virus 157Cys Thr Glu Leu Lys Leu Ser Asp Tyr1
51589PRTInfluenza A Virus 158Val Ser Asp Gly Gly Pro Asn Leu Tyr1
51599PRTInfluenza A Virus 159Gly Ile Leu Gly Phe Val Phe Thr Leu1
516010PRTInfluenza A Virus 160Ile Leu Gly Phe Val Phe Thr Leu Thr
Val1 5 101619PRTInfluenza A Virus 161Ile Leu Arg Gly Ser Val Ala
His Lys1 51629PRTInfluenza A Virus 162Ser Ile Ile Pro Ser Gly Pro
Leu Lys1 516310PRTInfluenza A Virus 163Arg Met Val Leu Ala Ser Thr
Thr Ala Lys1 5 101649PRTInfluenza A Virus 164Lys Ser Met Arg Glu
Glu Tyr Arg Lys1 51659PRTInfluenza A Virus 165Ser Pro Ile Val Pro
Ser Phe Asp Met1 51669PRTInfluenza A Virus 166Gln Pro Glu Trp Phe
Arg Asn Val Leu1 51679PRTInfluenza A Virus 167Ser Arg Tyr Trp Ala
Ile Arg Thr Arg1 516816PRTArtificial SequenceStem for branched
peptideMISC_FEATURE(1)..(16)X = Azidonorleucine 168Ser Glu Glu Xaa
Ser Glu Glu Xaa Ser Glu Glu Xaa Ser Glu Glu Xaa1 5 10
1516919PRTArtificial SequenceBranch 1-1 for branched
peptideMISC_FEATURE(1)..(19)X = propargyl glycine 169Xaa Lys Pro
Ala Lys Phe Phe Arg Leu Thr Pro Arg Val Thr Gly Gly1 5 10 15Gly Ala
Met17020PRTArtificial SequenceBranch 1-2 for branched
peptideMISC_FEATURE(1)..(20)X = propargyl glycine 170Xaa Lys Pro
Ala Lys Phe Phe Arg Leu Arg Pro His Glu Arg Asn Gly1 5 10 15Phe Thr
Val Leu 2017119PRTArtificial SequenceBranch 1-3 for branched
peptideMISC_FEATURE(1)..(19)X = propargyl glycine 171Xaa Lys Pro
Ala Lys Phe Phe Arg Leu Arg Glu Leu Arg Arg Lys Met1 5 10 15Met Tyr
Met17218PRTArtificial SequenceBranch 1-4 for branched
peptideMISC_FEATURE(1)..(18)X = propargyl glycine 172Xaa Lys Pro
Ala Lys Phe Phe Arg Leu Asn Leu Val Pro Met Val Ala1 5 10 15Thr
Val17318PRTArtificial SequenceBranch 3-1 for branched
peptideMISC_FEATURE(1)..(18)X = propargyl glycine 173Xaa Ala Ile
Pro Val Ser Leu Arg Thr Pro Arg Val Thr Gly Gly Gly1 5 10 15Ala
Met17419PRTArtificial SequenceBranch 3-2 for branched
peptidemisc_feature(1)..(1)Xaa can be any naturally occurring amino
acid 174Xaa Ala Ile Pro Val Ser Leu Arg Arg Pro His Glu Arg Asn Gly
Phe1 5 10 15Thr Val Leu17517PRTArtificial SequenceBranch 3-3 for
branched peptideMISC_FEATURE(1)..(17)X = propargyl glycine 175Xaa
Ala Ile Pro Val Ser Leu Arg Glu Leu Arg Arg Lys Met Met Tyr1 5 10
15Met17616PRTArtificial SequenceBranch 3-4 for branched
peptideMISC_FEATURE(1)..(16)X = propargyl glycine 176Xaa Ala Ile
Pro Val Ser Leu Val Thr Glu His Asp Thr Leu Leu Tyr1 5 10
151778PRTArtificial SequenceC1s cleavage site 177Tyr Leu Gly Arg
Ser Tyr Lys Val1 51786PRTArtificial SequenceC1s cleavage
sitemisc_feature(6)..(6)Xaa can be any naturally occurring amino
acid 178Met Gln Leu Gly Arg Xaa1 51798PRTArtificial SequenceMASP2
cleavage site 179Ser Leu Gly Arg Lys Ile Gln Ile1
51809PRTArtificial SequenceC2a cleavage site 180Gly Leu Ala Arg Ser
Asn Leu Asp Glu1 51819PRTArtificial SequenceCathepsin D cleavage
site 181Pro Arg Ser Phe Phe Arg Leu Gly Lys1 51828PRTArtificial
SequenceADAM28 cleavage site 182Lys Pro Ala Lys Phe Phe Arg Leu1
51838PRTArtificial SequenceADAM28 cleavage site 183Asp Pro Ala Lys
Phe Phe Arg Leu1 51848PRTArtificial SequenceADAM28 cleavage site
184Lys Pro Met Lys Phe Phe Arg Leu1 51858PRTArtificial
SequenceADAM28 cleavage site 185Leu Pro Ala Lys Phe Phe Arg Leu1
51867PRTArtificial SequenceMMP2 cleavage site 186Ala Ile Pro Val
Ser Leu Arg1 5187220PRTArtificial SequenceBundled domain sequence
187Ser Ala Ser Gly Gly Gly Ser Gly Gly Gly Ser Val Ser Asp Val Pro1
5 10 15Arg Asp Leu Glu Val Val Ala Ala Thr Pro Thr Ser Leu Leu Ile
Ser 20 25 30Trp Asp Ala Pro Ala Val Gly Gly Gly Gly Ser Gly Gly Gly
Gly Ser 35 40 45Thr Ile Pro Val Ser Leu Arg Ser Thr Pro Arg Val Thr
Gly Gly Gly 50 55 60Ala Met Thr Ile Pro Val Ser Leu Arg Ser Gly Gly
Gly Gly Ser Gly65 70 75 80Gly Gly Gly Ser Thr Val Arg Tyr Tyr Arg
Ile Thr Tyr Gly Glu Thr 85 90 95Gly Gly Asn Ser Pro Val Gln Glu Phe
Thr Val Pro Gly Ser Lys Ser 100 105 110Thr Ala Thr Ile Ser Gly Leu
Lys Pro Gly Val Asp Tyr Thr Ile Thr 115 120 125Val Tyr Ala Val Thr
Gly Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser 130 135 140Thr Ile Pro
Val Ser Leu Arg Ser Asn Leu Val Pro Met Val Ala Thr145 150 155
160Val Thr Ile Pro Val Ser Leu Arg Ser Gly Gly Gly Gly Ser Gly Gly
165 170 175Gly Gly Ser Ser Pro Ala Ser Ser Lys Pro Ile Ser Ile Asn
Tyr Arg 180 185 190Thr Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Thr
Ile Pro Val Ser 195 200 205Leu Arg Ser Val Leu Glu Glu Thr Ser Val
Met Leu 210 215 2201888PRTArtificial SequenceMMP2 cleavage site
188Thr Ile Pro Val Ser Leu Arg Ser1 5
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.