Wearable Cardiac Monitor
Golda; George Stefan ; et al.
U.S. patent application number 16/573606 was filed with the patent office on 2020-01-09 for wearable cardiac monitor. This patent application is currently assigned to RHYTHM DIAGNOSTIC SYSTEMS, INC.. The applicant listed for this patent is RHYTHM DIAGNOSTIC SYSTEMS, INC.. Invention is credited to Sam Eletr, George Stefan Golda, Mark P. Marriott, Daniel Van Zandt Moyer, Bruce O'Neil.
| Application Number | 20200008749 16/573606 |
| Document ID | / |
| Family ID | 50433219 |
| Filed Date | 2020-01-09 |









View All Diagrams
| United States Patent Application | 20200008749 |
| Kind Code | A1 |
| Golda; George Stefan ; et al. | January 9, 2020 |
WEARABLE CARDIAC MONITOR
Abstract
Systems, methods and devices for reducing noise in cardiac monitoring including wearable monitoring devices having at least one electrode for cardiac monitoring; in some implementations, the wearable device using a composite adhesive having at least one conductive portion applied adjacent the electrode; and, in some implementations, including circuitry adaptations for the at least one electrode to act as a proxy driven right leg electrode.
| Inventors: | Golda; George Stefan; (El Granada, CA) ; Moyer; Daniel Van Zandt; (Menlo Park, CA) ; Marriott; Mark P.; (Palo Alto, CA) ; Eletr; Sam; (Paris, FR) ; O'Neil; Bruce; (San Francisco, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | RHYTHM DIAGNOSTIC SYSTEMS,
INC. SAN FRANCISCO CA |
||||||||||
| Family ID: | 50433219 | ||||||||||
| Appl. No.: | 16/573606 | ||||||||||
| Filed: | September 17, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 13837748 | Mar 15, 2013 | 10413251 | ||
| 16573606 | ||||
| 61710768 | Oct 7, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61B 5/04085 20130101; A61B 5/0205 20130101; A61B 5/14551 20130101; A61B 2560/0295 20130101; A61B 5/7207 20130101; A61B 5/14552 20130101; A61B 5/0261 20130101; A61B 5/02141 20130101; A61B 5/0402 20130101; A61B 5/0295 20130101; A61B 5/0059 20130101; A61B 5/7214 20130101; A61B 5/04325 20130101; A61B 5/6833 20130101; A61B 5/0002 20130101; A61B 5/02438 20130101; A61B 2560/0412 20130101; A61B 5/72 20130101; A61B 5/0006 20130101; A61B 5/11 20130101; A61B 5/721 20130101; A61B 5/1118 20130101; A61B 5/6828 20130101; A61B 5/04087 20130101; A61B 5/02125 20130101; A61B 5/0456 20130101; A61B 5/7275 20130101; A61B 5/08 20130101 |
| International Class: | A61B 5/00 20060101 A61B005/00; A61B 5/0432 20060101 A61B005/0432; A61B 5/0205 20060101 A61B005/0205; A61B 5/026 20060101 A61B005/026; A61B 5/0295 20060101 A61B005/0295; A61B 5/0408 20060101 A61B005/0408; A61B 5/11 20060101 A61B005/11; A61B 5/0402 20060101 A61B005/0402; A61B 5/08 20060101 A61B005/08; A61B 5/021 20060101 A61B005/021 |
Claims
1. A device for monitoring a physiological parameter, the device being adapted to be adhered to the skin of a subject for the physiological parameter monitoring; the device comprising: a substrate; a conductive sensor connected to the substrate, and a double-sided composite adhesive having: at least one conductive adhesive portion, and at least one non-conductive adhesive portion; the double-sided composite adhesive being attached to the substrate and the conductive sensor; the at least one conductive adhesive portion being disposed in conductive communicative contact with the conductive sensor and being adapted to be conductively adhered to the skin of the subject for conductive signal communication from the subject to the conductive sensor.
2. A device according to claim 1 wherein conductive adhesive portion of the composite adhesive is connected to respective sensor and adapted to be substantially securely connected to the skin to maintain the respective sensor substantially fixed relative to the skin and thereby one or both reduce or eliminate possible sensor movement relative to the skin.
3. A device according to claim 2 wherein the connection of the conductive adhesive portion to the respective sensor and to the skin removes movement of the sensor relative to the skin which removes noise which provides a clean signal.
4. A device according to claim 1 wherein the physiological parameter includes one or more signals for one or more or all of electrocardiography, photoplethysmography, pulse oximetry or subject acceleration.
5. A device according to claim 4 wherein the conductive sensor is an electrode for electrocardiography.
6. A device according to claim 5 wherein the electrode is a proxy driven-right-leg electrode.
7. A device according to claim 1 comprising a plurality of conductive sensors connected to the substrate, the composite adhesive having a plurality of conductive adhesive portions, the plurality of conductive adhesive portions being disposed in conductive communicative contact with respective ones of the plurality of conductive sensors, and being adapted to be conductively adhered to the skin of the subject for conductive signal communication from the subject to the conductive sensor.
8. A device according to claim 7 wherein the plurality of conductive sensors include at least one, two or three electrodes for electrocardiography.
9. A device according to claim 8 wherein one of the electrodes is a proxy driven-right-leg electrode.
10. A device according to claim 1 which is adapted to provide one or both of collected data or analysis to a receiving system component.
11. A device according to claim 10 wherein the one or both of collected data and analysis are transmitted by one or both of wireless or wired connections.
12. A system including a device according to claim 10 wherein the receiving system component is a computing device.
13. A system for collecting and analyzing physiological data corresponding to physiological signals of a subject; the system comprising: a wearable device for collecting physiological data from a subject and one or both of transmitting and analyzing the physiological data; and, a receiving system component for receiving physiological data transmitted by the device.
14. A system according to claim 13 wherein the wearable device comprises on-board memory for storing collected data.
15. A system according to claim 13 wherein the receiving system component is a computing device adapted to analyze data received from the wearable device.
16. A system according to claim 13 wherein the wearable device has at least one electrode and a composite adhesive, the composite adhesive having at least one conductive adhesive portion in communication with the at least one electrode.
17. A system according to claim 13 wherein the wearable device has at least one electrode and circuitry for the at least one electrode to function as a proxy driven-right-leg electrode.
18. A method for reducing noise in cardiac monitoring using a wearable monitoring device having at least one electrode for cardiac monitoring; the method comprising: applying the wearable monitoring device to a subject; including one or both of: using a composite adhesive having at least one conductive portion; or, using circuitry adaptations for the at least one electrode to act as a proxy driven-right-leg electrode; collecting data representative of physiological signals; analyzing the data.
19. A method according to claim 18 further comprising: transmitting the data from the wearable device.
20. A method according to claim 18 further comprising; transmitting the data from the wearable device; and, storing the data prior to one or both of transmitting or analyzing the data.
21. A circuit for mimicking a right leg electrode using a chest reference electrode as proxy therefor; the circuit comprising: a connection to a first electrocardiogram electrode; a connection to a second electrocardiogram electrode; a connection to a reference electrocardiogram electrode; and, an amplifier; the electrode connections all connected to the amplifier; the amplifier maintaining the voltage of the first and second electrodes level, the proxy driven right leg signal being measurable using the reference electrode.
22. A circuit for mimicking a right leg electrode using a chest reference electrode as proxy therefor; the circuit comprising: a connection to a sense electrode; a connection to a drive electrode; a bias voltage input; and, an amplifier; the electrode connections and bias voltage input connected to the amplifier; the amplifier output connected to the drive electrode, the inverting input to the sense electrode, and the non-inverting input to a bias voltage; the amplifier maintaining the voltage of the sense electrode at a level close to the bias voltage; the proxy driven right leg signal being measurable using a third electrode.
23. A device for monitoring physiological parameters of a patient comprising: a flexible circuit embedded in a flat elastic substrate having a top surface and a bottom surface, the flexible circuit having (i) at least one sensor mounted in the bottom surface of the flat elastic substrate, the at least one sensor being capable of electrical or optical communication with the patient, (ii) at least one signal processing module for accepting signals from the at least one sensor and transforming such signals for storage as patient data; (iii) at least one memory module for accepting and storing patient data, (iv) at least one data communication module for transferring stored patient data to an external device, and (v) a control module for controlling the timing and operation of the at least one sensor, the at least one signal processing module, the at least one memory module, and the at least one data communication module, the control module capable of receiving commands to implement transfer of patient data by the at least one data communication module and to erase and/or wipe patient data from the at least one memory module; and an anisotropically conductive adhesive removably attached to the bottom surface of the flat elastic substrate, the anisotropically conductive adhesive capable of adhering to skin of the patient and of conducting an electrical signal substantially only in a direction perpendicular to the bottom surface of the flat elastic substrate.
24. The device of 23 wherein said at least one sensor includes an electrode for measuring electrical signals from said patient's heart and an optical blood oxygen sensor.
25. The device of 23 wherein said at least one data communication module is capable of transmitting said patient data by wireless signals to an external device.
26. The device of 25 wherein said external device receiving said patient data is a signal relay device.
27. The device of 23 wherein said flexible circuit is capable of being powered wirelessly.
28. The device of 23 wherein said at least one signal processing module receives a signal from at least one electrode from measuring electrical signals from said patient's heart and a signal from a driven right leg circuit for reducing common mode noise from said patient data.
29. A system for monitoring physiological parameters of a patient comprising: a flexible circuit embedded in a flat elastic substrate having a top surface and a bottom surface, the flexible circuit having (i) at least one sensor mounted in the bottom surface of the flat elastic substrate, the at least one sensor being capable of electrical or optical communication with the patient, (ii) at least one signal processing module for accepting signals from the at least one sensor and transforming such signals for storage as patient data; (iii) at least one memory module for accepting and storing patient data, (iv) at least one data communication module for transferring stored patient data to an external device, and (v) a control module for controlling the timing and operation of the at least one sensor, the at least one signal processing module, the at least one memory module, and the at least one data communication module, the control module capable of receiving commands to implement transfer of patient data by the at least one data communication module and to erase and/or wipe patient data from the at least one memory module; an anisotropically conductive adhesive removably attached to the bottom surface of the flat elastic substrate, the anisotropically conductive adhesive capable of adhering to skin of the patient and of conducting an electrical signal substantially only in a direction perpendicular to the bottom surface of the flat elastic substrate; and a computer for accepting patient data from said data communication module and for displaying the patient data and/or for generating an alert if values of patient data exceed predetermined limits.
Description
CROSS-REFERENCE TO RELATED APPLICATION(S)
[0001] This is a nonprovisional application claiming the benefit of and priority to the provisional application, U.S. 61/710,768 filed Oct. 7, 2012, the entire contents, teachings and suggestions thereof being incorporated herein by this reference as if fully set forth here.
BACKGROUND
[0002] Advances in electronics, sensor technology and materials science have revolutionized patient monitoring technologies. In particular, many light and wearable devices are becoming available for a variety of cardiac monitoring applications. However, improvements may yet be desired for robust wearable devices that provide effective data collection, in some cases also with increased patient convenience and comfort. Other alternatives may include developments in one or more of device attachment, size, flexibility, data transfer, among others.
[0003] Further alternatives for cardiac patients and their physicians may then include robust and convenient personal cardiac monitors that in some instances may collect and transfer long-term data as well as monitor events in real-time.
SUMMARY
[0004] Described herein are several medical monitoring devices and systems, in some instances for long-term sensing and/or recording of cardiac patient data. A number of alternative implementations and applications are summarized and/or exemplified herein below and throughout this specification.
[0005] These as well as other aspects are exemplified in a number of illustrated alternative implementations and applications, some of which are shown in the figures and characterized in the claims section that follows. However, as will be understood by the ordinarily skilled artisan, the above summary and the detailed description below do not describe the entire scope of the inventions hereof and are indeed not intended to describe each illustrated embodiment or every implementation of the present inventions nor provide any limitation on the claims or scope of protection herein set forth below.
BRIEF DESCRIPTION OF THE DRAWINGS
[0006] The drawings include:
[0007] FIG. 1, which includes sub-part FIGS. 1A-1G, illustrates several alternatives of the present inventions, including various of isometric, top and bottom plan and elevational views of devices and alternative conductive adhesive structures.
[0008] FIG. 2, which includes sub-part FIGS. 2A-2C, provides circuit diagrams of alternatives to a driven right leg circuit.
[0009] FIG. 3 is a flow chart including alternative methods of use.
[0010] FIG. 4 illustrates an exemplary computer system or computing resources with which implementations hereof may be utilized.
[0011] FIG. 5, which includes sub-part FIGS. 5A-5D, provides alternative screenshots of alternative software implementations according hereto.
DETAILED DESCRIPTION
[0012] While the inventions hereof amenable to various modifications and alternative forms, specifics thereof have been shown by way of example in the drawings and the following description. It should be understood, however, that the intention is not to limit the invention to the particular embodiments described. The intention is to cover all modifications, equivalents, and alternatives falling within the spirit and scope of the invention whether described here or otherwise being sufficiently appreciable as included herewithin even if beyond the literal words hereof.
[0013] In one aspect, a system hereof may include a device for monitoring physiological parameters such as one or more or all of electrocardiogram (aka ECG or EKG), photoplethysmogram (aka PPG), pulse oximetry and/or patient acceleration or movement signals. Systems hereof may be established to measure and process such signals of a patient using or including one or more of the following elements: (a) a circuit, sometimes flexible, embedded in a flat elastic substrate having a top surface and a bottom surface, the circuit having (i) at least one sensor mounted in or adjacent the bottom surface of the flat elastic substrate, the at least one sensor being capable of electrical or optical communication with the patient, (ii) at least one signal processing module for receiving and/or accepting signals from the at least one sensor in some implementations also providing for transforming such signals for storage as patient data; (iii) at least one memory module for receiving and/or accepting and storing patient data, (iv) at least one data communication module for transferring stored patient data to an external device, and (v) a control module for controlling the timing and operation of the at least one sensor, one or more of the at least one signal processing module, the at least one memory module, the at least one data communication module, and/or the control module capable of receiving commands to implement transfer of patient data by the at least one data communication module and to erase and/or wipe patient data from the at least one memory module; and (b) a anisotropically conductive adhesive removably attached to the bottom surface of the flat elastic substrate, the anisotropically conductive adhesive capable of adhering to skin of the patient and of conducting an electrical signal substantially only in a direction perpendicular to the bottom surface of the flat elastic substrate, and/or in some implementations including a conductive portion adjacent the sensor or sensors and a non-conductive portion.
[0014] In some implementations, devices hereof will be for comprehensive long-term cardiac monitoring. Features of such may include one or more of a Lead 1 ECG, PPG, pulse oximeter, accelerometer, and a button or other indicator for manual patient event marking Such a device may be adapted to store up to, for example, about two weeks of continuous data (though more will also be feasible in alternative implementations), which may in some implementations be downloaded to a clinic or other computer in a short time period, as for one example, in only about 90 seconds (though less time will be viable in alternative implementations) via computer connection, whether wireless or wired as in one example by USB or other acceptable data connection. A companion software data analysis package may be adapted to provide automated event capture and/or allow immediate, local data interpretation.
[0015] Intermittent cardiac anomalies are often difficult for physicians to detect and/or diagnose, as they would typically have to occur during a physical examination of the patient. A device hereof may address this problem with what in some implementations may be a continuous or substantially continuous monitoring of a number of vital signs.
[0016] Some alternative features may include (i) a driven "Right Leg" circuit with electrodes located only on the chest, (ii) a "z-Axis" or anisotropic conductive adhesive electrode interface that may permit electrical communication only between an electrode and a patient's skin immediately beneath the electrode, (iii) data transmission to and interpretation by a local computer accessible to CCU/ICU personnel, (iv) a unique combination of hardware allows correlation of multiple data sources in time concordance to aid in diagnosis.
[0017] In some alternative implementations, devices and systems hereof may provide 1) reusability (in some cases near or greater than about 1000 patients) allows recouping cost of the device in just about 10-15 patient tests, 2) one or more of ecg waveform data, inertial exertion sensing, manual event marking, and/or pulse oximeter, any or all of which in time concordance to better detect and analyze arrhythmic events, 3) efficient watertightness or waterproofing (the patient can even swim while wearing the device), and 4) a comprehensive analysis package for immediate, local data interpretation. An alternative device may be adapted to take advantage of flex-circuit technology, to provide a device that is light-weight, thin, durable, and flexible to conform to the patient's skin.
[0018] FIGS. 1 and 2 illustrate examples of alternative implementations of devices that may be so adapted.
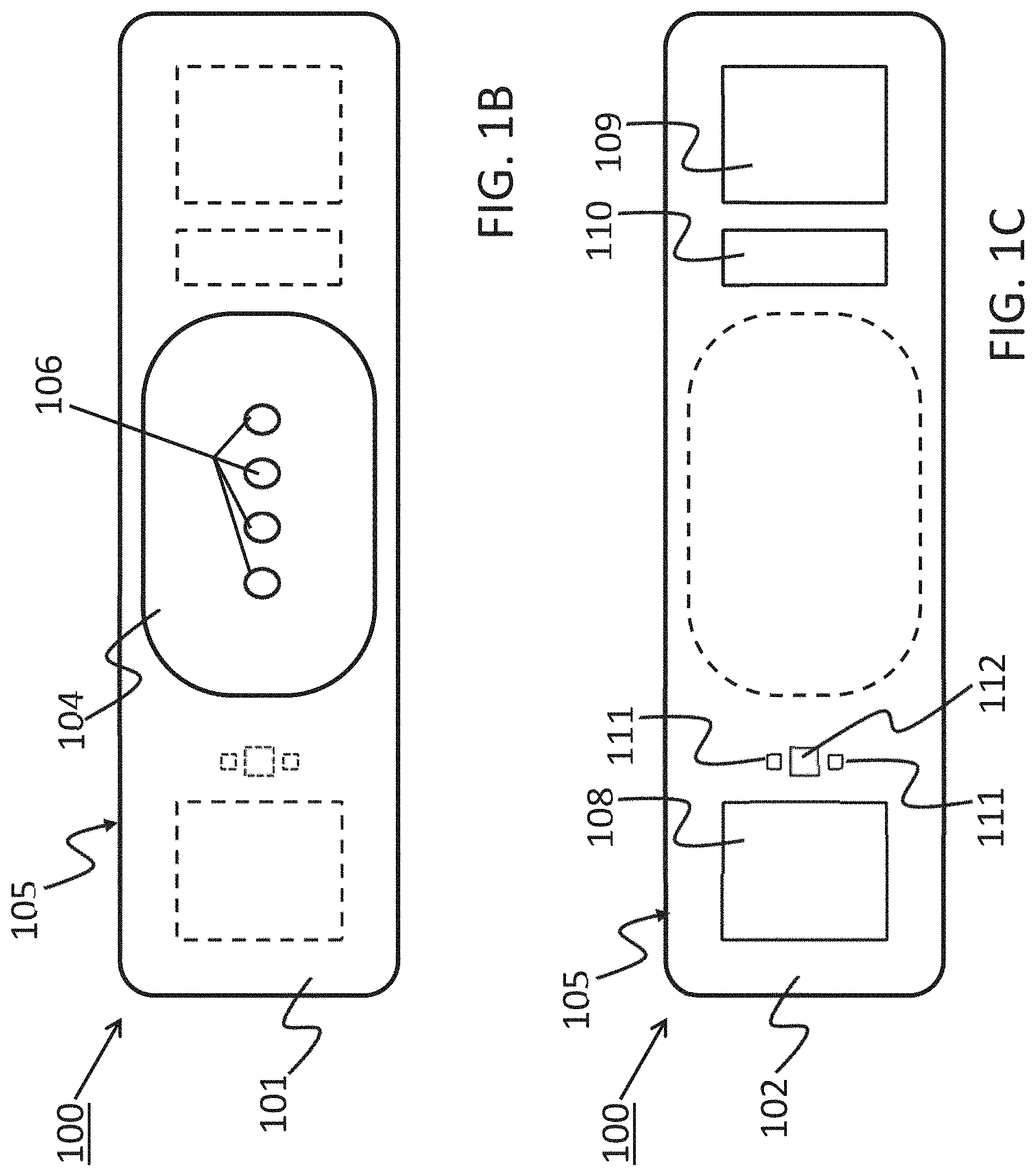
[0019] FIG. 1 shows a device 100 that has a component side or top side 101, patient side or circuit side 102, and one or more inner electrical layer(s), generally identified by the reference 103 and an elongated strip layer 105. The strip layer 105 may have electronics thereon and/or therewithin. FIG. 1A shows isometrically these together with some other elements that may be used herewith. FIG. 1B is more specifically directed to a top side 101 plan view and FIG. 1C to an underside, patient side 102 plan view and FIG. 1D a first elevational, side view.
[0020] Many of the electronics hereof may be disposed in the electronics layer or layers 103, and as generally indicated here, the electronics may be encapsulated in a material 104 (see FIGS. 1A, 1B and 1D for some examples), plastic or the like, or potting material, to fix them in operative position on or in or otherwise functionally disposed relative to the elongated strip layer 105. The potting or other material may in many implementations also or alternatively provide a waterproof or watertight or water resistant coverage of the electronics to keep them operative even in water or sweat usage environments. One or more access points, junctions or other functional units 106 may be provided on and/or through any side of the encapsulation material 104 for exterior access and/or communication with the electronics disposed therewithin, or thereunder. FIGS. 1A, 1B and 1D show four such accesses 106 on the top side. These may include high Z data communication ports and/or charging contacts, inter alia. This upper or component side 101 of device 100 may be coated in a silicone compound for protection and/or waterproofing, with only, in some examples, a HS USB connector exposed via one or more ports 106, e.g., for data communication or transfer and/or for charging.
[0021] The elongated strip layer 105 may be or may include a circuit or circuit portions such as electrical leads or other inner layer conductors, e.g., leads 107 shown in FIG. 1D, for communication between the electronics 103 and the electrically conductive pads or contacts 108, 109 and 110 described further below (108 and 109 being in some examples, high impedance/high Z silver or copper/silver electrodes for electrocardiograph, ECG, and 110 at times being a reference electrode). In many implementations, the strip layer 105 may be or may include flex circuitry understood to provide acceptable deformation, twisting, bending and the like, and yet retain robust electrical circuitry connections therewithin. Note, though the electronics 103 and electrodes 108, 109, 110 are shown attached to layer 105; on top for electronics 103, and to the bottom or patient side for electrodes 108, 109, 110; it may be that such elements may be formed in or otherwise disposed within the layer 105, or at least be relatively indistinguishably disposed in relative operational positions in one or more layers with or adjacent layer 105 in practice. Similarly, the leads or traces 107 are shown embedded (by dashed line representation in FIG. 1D); however, these may be on the top or bottom side, though more likely top side to insulate from other skin side electrical communications. If initially top side (or bottom), the traces may be subsequently covered with an insulative encapsulant or like protective cover (not separately shown), in many implementations, a flexible material to maintain a flexible alternative for the entire, or majority of layer 105.
[0022] On the patient side 102, the ECG electrodes 108, 109 and 110 may be left exposed for substantially direct patient skin contact (though likely with at least a conductive gel applied therebetween); and/or, in many implementations, the patient side electrodes 108, 109 and/or 110 may be covered by a conductive adhesive material as will be described below. The electrodes may be or may be may be plated with a robust high conductive material, as for example, silver/silver chloride for biocompatibility and high signal quality, and in some implementations may be highly robust and, for one non-limiting example, be adapted to withstand over about 1000 alcohol cleaning cycles between patients. Windows or other communication channels or openings 111, 112 may be provided for a pulse oximeter, for example, for LEDs and a sensor. Such openings 111, 112 would typically be disposed for optimum light communication to and from the patient skin. An alternative disposition of one or more light conduits 111a/112a is shown in a non-limiting example in FIG. 1D more nearly disposed and/or connected to the electronics 103. A variety of alternative placements may be usable herein/herewith.
[0023] FIG. 1D provides a first example of an adhesive 113 that may be used herewith. The adhesive layer 113 is here a double-sided adhesive for application to the bottom side 102 of the device 100, and a second side, perhaps with a different type of adhesive for adhering to the skin of the human patient (not shown). Different types of materials for adhesion might be used in that the material of choice to which the adhesive layer is to be attached are different; typically, circuit or circuit board material for connection to the device 100, and patient skin (not separately shown) on the patient side. A protective backing 114 may be employed on the patient side until application to the patient is desired. Note, in many applications, the adhesive 113 is anisotropic in that it may preferably be only conductive in a single or substantially a single direction, e.g., the axis perpendicular to the surface of adhesive contact. Thus, good electrically conductive contact for signal communication can be had through such adhesive to/through the adhesive to the electrical contacts or electrodes, 108, 109 and 110. Note, a corresponding one or more light apertures 111b/112b are shown in the adhesive of 113 of the example of FIG. 1D to communicate light therethrough in cooperation with the light conduit(s) 111a/112a in/through layer 105 for communication of light data typically involved in pulse oximetry.
[0024] The adhesive may thus be placed or disposed on the device 100, in some implementations substantially permanently, or with some replaceability. In some implementations, the device as shown in FIGS. 1A-1D and/or 1G without (or with in some implementations) the adhesive may be reusable. In many such cases, the adhesive layer 113 may be removed and replaced before each subsequent use, though subsequent re-use of and with a layer 113 is not foreclosed. In a first or subsequent use with a replaceable adhesive layer 113, it may be that the user applying the device to the patient, e.g., the physician or technician or even the patient, him/herself, applies the conductive transfer adhesive 113 to the patient side 102 of the device 100. The protective backing 114 may then be removed, and the device adhered to the patient and activated. Activation may occur in a number of ways; in some, it may be pre-set that an affirmative activation interaction may not be necessary from the doctor or patient or like due to either an inertial and/or a pulse oximeter activation which may be substantially automatically activating, e.g., upon receiving sufficient minimum input (movement in case of inertial system or light reflection of blood flow for pulse oximetry); however, a button may be provided at access 106 or in some other location adjacent the electronics to allow the patient to start or stop the device or otherwise mark an event if desired. In one exemplar implementation the device may be worn for a period such as two weeks for collection of data substantially continuously, or at intervals as may be preferred and established in or by the systems hereof.
[0025] After a monitoring period is over a physician, technician, patient or other person may then remove the device from the patient body, remove the adhesive, in some instances with alcohol, and may establish a data communication connection for data transfer, e.g., by wireless communication or by insertion/connection of a USB or like data connector to download the data. The data may then be processed and/or interpreted and in many instances, interpreted immediately if desired. A power source on board may include a battery and this can then also be re-charged between uses, in some implementations, fully recharged quickly as within about 24 hours, after which the device could then be considered ready for the next patient.
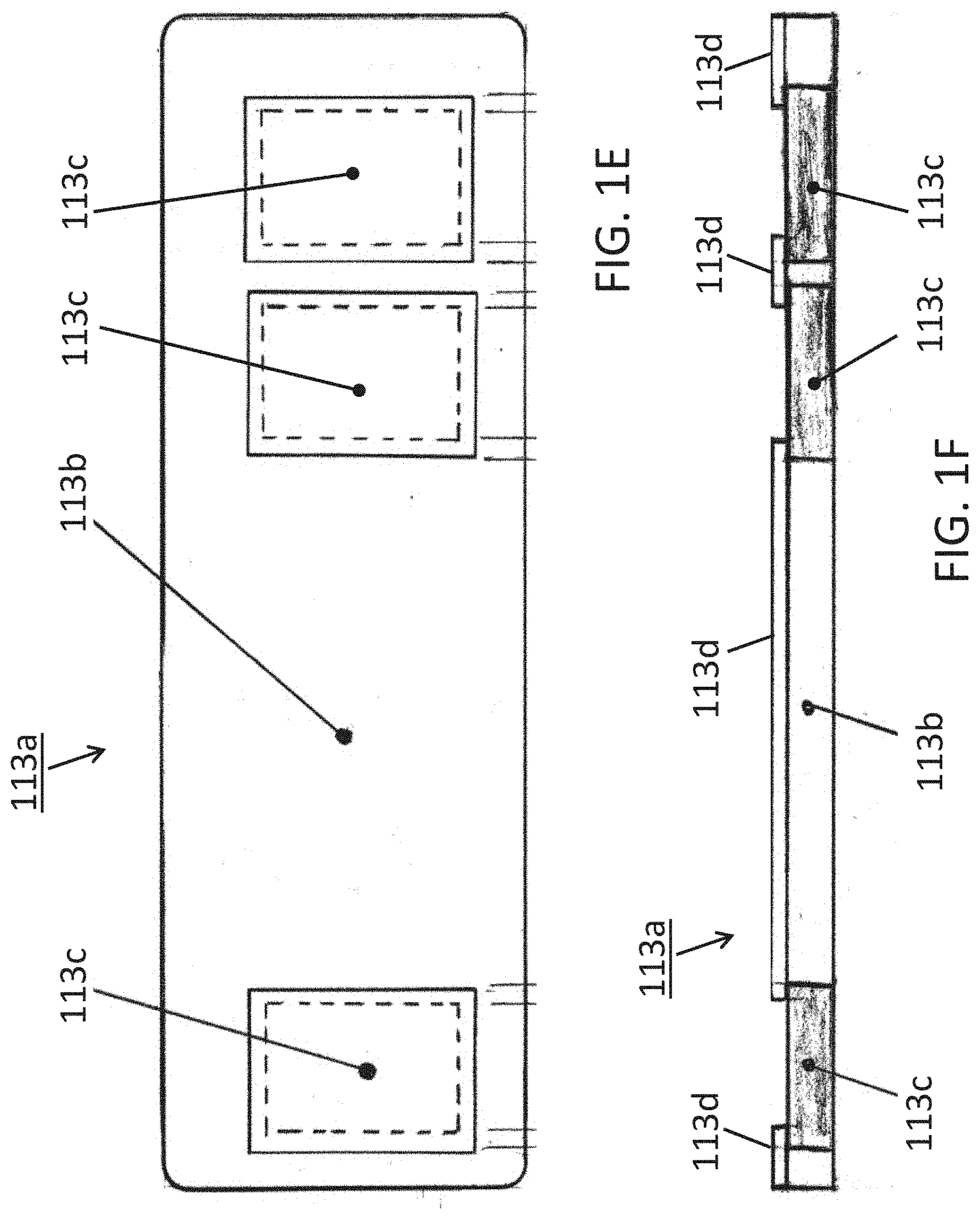
[0026] Some alternative conductive adhesives may be used herewith. FIGS. 1E, 1F and 1G show one such alternative conductive adhesive 113a; a bottom plan view in FIG. 1E and elevational side views thereof in FIGS. 1F and 1G (as being connected to a device 100 in FIG. 1G). In some implementations, the conductivity may be anisotropic as introduced above; in some conductive primarily if not entirely in the direction of the Z-Axis; perpendicular to the page (into and/or out of the page) in FIG. 1E, and/or vertically or transversally relative to the long horizontal shown axis of device 100 in the implementation view of FIG. 1F.
[0027] The implementation of this particular example includes a composite adhesive 113a which itself may include some non-conductive portion(s) 113b and some one or more conductive portions 113c. The adhesive composite 113a may, as described for adhesive 113 above be double sided such that one side adheres to the patient while the other side would adhere to the underside 102 of the device 100 (see FIG. 1G) so that one or more conductive portions 113c may be disposed or placed in electrically communicative and/or conductive contact with the integrated electrodes on the electronic monitoring device 100. Since the electrodes would operate better where they may be electrically isolated or insulated from each other, yet each making electrical contact or communication with the patient's skin, the adhesive may further be more specifically disposed in some implementations as follows.
[0028] As shown in FIGS. 1E and 1F, three isolated conductive portions 113c may be disposed separated from each other by a body portion 113b which may be non-conductive. These could then correspond to the electrodes 108, 109, 110 from the above-described examples, and as more particularly shown schematically in FIG. 1G (note the scale is exaggerated for the adhesive 113a and thus, exact matching to the electrodes of device 100 is not necessarily shown). In some examples, the electrode areas 113c may be a conductive hydrogel that may or may not be adhesive, and in some examples, may be made of a conductive an adhesive conductive material such as 3M Corporation 9880 Hydrogel adhesive (3M Company, St. Paul, Minn.). These areas 113c may then be isolated from each other by a non-conductive material 113b such as 3M Corporation 9836 tape or 3M double-sided Transfer Adhesive 9917 (3M, St. Paul, Minn.) or equivalent. The additional layer 113d, if used, might be a 3M 9917 adhesive together with the 113b of a 9836 material. These constructs may provide the effect of creating a low electrical impedance path in the Z-axis direction (perpendicular to page for FIG. 1E and vertically/transversally for FIGS. 1F and 1G) for the electrode areas 113c, and high electrical impedance path between the electrodes in the X/Y directions. (See FIGS. 1E, 1F and 1G; coplanar with the page in FIG. 1E and horizontal and perpendicular to the page in FIGS. 1F and 1G). Thus, a composite adhesive strip can ensure not only device adhering to the patient, but also that the electrodes whether two or as shown three electrodes are conductively connected by conductive portions of the adhesive strip, where the combination of conductive and non-conductive portions can then reduce signal noise and/or enhance noise free characteristics. Electrodes that move relative to skin can introduce noise. I.e., electrodes electrically communicative/connected to the skin via a gel may move relative to the skin and thus introduce noise. However, with one or more conductive adhesive portions in a composite adhesive connected to respective electrodes and then substantially securely connected to the skin will keep the respective electrodes substantially fixed relative to the skin and thereby reduce or even eliminate electrode movement relative to the skin. Removal of such movement would then remove noise which would thereby provide a clean signal that can allow for monitoring cardiac P waves which enhances the possibility to detect arrythmias that couldn't otherwise be detected. Further description is set forth below.
[0029] In some implementations, a further optional connective and/or insulative structure 113d may be implemented as shown in FIG. 113d to provide further structural and insulative separation between electrodes with connected to a device 100 on the underside 102 thereof (see FIG. 1G). Though shown separate in FIGS. 1F and 1G, it may be contiguous with the insulative adhesive 113b of these views.
[0030] Some alternative implementations hereof may include a driven right leg ECG circuit with one or more chest only electrodes ("Driven Chest Electrode"). In addition to the electrodes used to measure a single or multiple lead electrocardiogram signal, a device 100 may use an additional electrode, as for example the reference electrode 110 (see FIGS. 1A, 1C, 1D and 1G, e.g.) to reduce common mode noise. Such an electrode may function in a manner similar to the commonly-used driven right leg electrode, but may here be located on the patient's chest rather than on the patient's right leg but nevertheless this third/reference electrode may play the role of the leg electrode. This chest electrode may thus mimic a right leg electrode and/or be considered a proxy driven right leg electrode. A circuit, or portion of an overall circuit, adapted to operate in this fashion may include a number of amplifier stages to provide gain, as well as filtering to ensure circuit stability and to shape the overall frequency response. Such a circuit may be biased to control the common mode bias of the electrocardiogram signal. This driven chest electrode implementation may be used in conjunction with a differential or instrumentation amplifier to reduce common mode noise. In this case, the sense electrode may be used as one of the electrocardiogram electrodes. Alternatively, a single-ended electrocardiogram amplifier may be used where the differential electrocardiogram signal is referenced to ground or to some other known voltage.
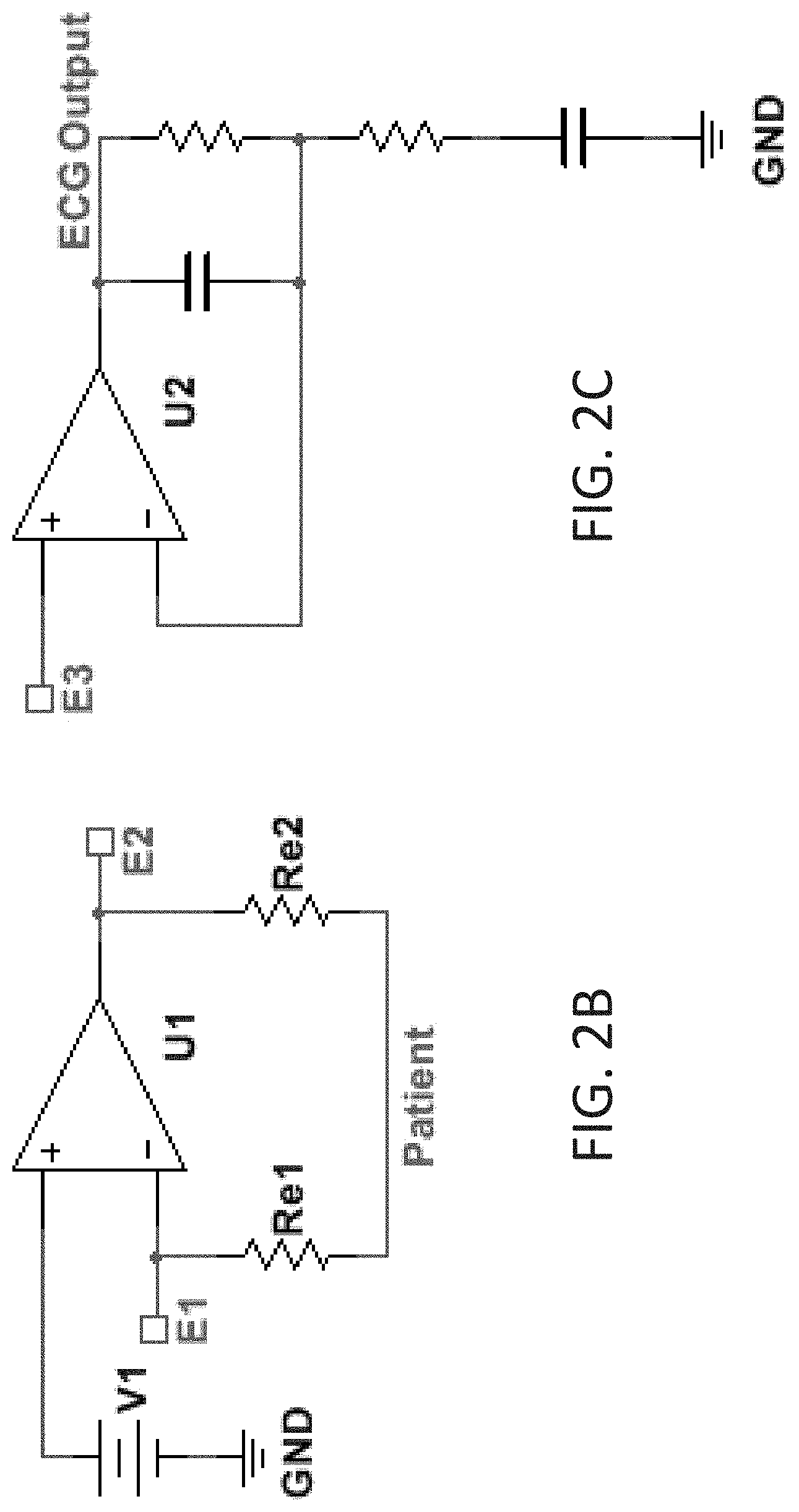
[0031] A circuit or sub-circuit 200 using a transistor 201 as shown in FIG. 2 may be such a circuit (aka module) and may thus include as further shown in FIG. 2A, a sense electrode 202, a drive electrode 203, and an amplifier 204. Both the sense and drive electrodes 202, 203 are placed on the patient's chest such that they provide an electrical connection to the patient. The amplifier 204 may include gain and filtering. The amplifier output is connected to the drive electrode, the inverting input to the sense electrode, and the non-inverting input to a bias voltage 205. The amplifier maintains the voltage of the sense electrode at a level close to the bias voltage. An electrocardiogram signal may then be measured using additional electrodes. Indeed, as was the case for the improved conductivity through use of anisotropic adhesive portions above, here also or alternatively, the use of this third electrode as a proxy for a right leg electrode (i.e., proxy driven right leg electrode) can provide signal reception otherwise unavailable. Clean signals may thus allow for receiving cardiac P waves which enhances the possibility to detect arrythmias that couldn't otherwise be detected.
[0032] Further alternative descriptions of circuitry include that which is shown in FIGS. 2B and 2C; in which are shown non-limiting alternatives in which three adjacent electrodes E1, E2, and E3 may be used to pick up the ECG signal, one of which electrodes playing the role of the distant limb electrode of traditional ECG monitors. Because the electrode-patient interface has an associated impedance (Re1 and Re2), current flowing through this interface will cause a difference in voltage between the patient and the electrode. The circuit may use a sense electrode (E1) to detect the patient voltage. Because this exemplar circuit node has a high impedance to circuit ground (GND), very little current flows through the electrode interface, so that the voltage drop between the patient and this node is minimized. The first of these alternative, non-limiting circuits (FIG. 2B) also contains an amplifier (U1) whose low-impedance output is connected to a separate drive electrode (E2). The amplifier uses negative feedback to control the drive electrode such that the patient voltage (as measured by the sense electrode E1) is equal to the bias voltage (V1). This may effectively maintain the patient voltage equal to the bias voltage despite any voltage difference between the driven electrode (E2) and the patient. This can include voltage differences caused by power line-induced current flowing between the drive electrode and the patient (through Re2). This arrangement differs from a traditional `driven-right-leg` circuit in at least two ways: the driven electrode is placed on the patient's chest (rather than the right leg), and the ECG signal is a single-ended (not differential) measurement taken from a third electrode (E3). Because all electrodes are located on the patient's chest, a small device placed there may contain all the necessary electrodes for ECG measurement. One possible benefit of the single-ended measurement is that gain and filtering circuitry (U2 and associated components (FIG. 2C)) necessary to condition the ECG signal prior to recording (ECG Output) requires fewer components and may be less sensitive to component tolerance matching. The examples of FIGS. 2A, 2B and 2C are non-limiting examples and not intended to limit the scope of the claims hereto as other circuits with other circuit elements can be formed by skilled artisans in view hereof and yet remain within the spirit and scope of claims hereof.
[0033] In many implementations, a system hereof may include other circuitry operative together with the ECG electrodes, which may thus be accompanied by other sensors to provide time concordant traces of: i) ECG p-, qrs-, and t-waves; ii) O2 Saturation, as measured by Pulse Oxymetry; and/or iii) xyz acceleration, to provide an index of physical activity. Such circuitry may be implemented to one or more of the following electrical specifications. The overall system might in some implementations include as much as two weeks (or more) of continuous run time; gathering data during such time. Some implementations may be adapted to provide as many or even greater than 1000 uses. Alternatives may include operability even after or during exposure to fluids or wetness; in some such examples being water resistant, or waterproof, or watertight, in some cases continuing to be fully operable when fully submerged (in low saline water). Other implementations may include fast data transfer, as for an example where using an HS USB for full data transfer in less than about 90 seconds. A rechargeable battery may typically be used.
[0034] A further alternative implementation may include an electronic "ground": In a device hereof, mounted entirely on a flexible circuit board, the ground plane function may be provided by coaxial ground leads adjacent to the signal leads. The main contribution of this type of grounding system may be hat it may allow the device the flexibility required to conform and adhere to the skin.
[0035] For electrocardiograph; EKG or ECG, some implementations may include greater than about 10 Meg Ohms input impedance; some implementations may operate with a 0.1-48 Hz bandwidth; and some with an approximate 256 Hz Sampling Rate; and may be implementing 12 Bit Resolution. For PPG and Pulse Oximeter, operation may be with 660 and 940 nm Wavelength; about 80-100 SpO2 Range; a 0.05-4.8 Hz Bandwidth; a 16 Hz Sampling Rate; and 12 bit resolution. For an accelerometer: a 3-Axis Measurement may be employed, and in some implementations using a .+-.2 G Range; with a 16 Hz Sampling Rate; and a 12 Bit Resolution.
[0036] Some summary methodologies may now be understood with relation to FIG. 3, though others may be understood through and as parts of the remainder of the disclosure hereof. A flow chart 300 as in FIG. 3 may demonstrate some of the alternatives; where an initial maneuver 301 might be the application of the device 100 to the patient. Indeed, this might include some one or more of the alternatives for adhesive application as described here above, whether by/through use of an adhesive such as that 113 of FIG. 1D, or that of FIGS. 1E, 1F and/or 1G. Then, as shown, in moving by flow line 311, a data collection operation 302 may be implemented. Note, this might include a continuous or substantially continuous collection or an interval or periodic collection or perhaps even a one time event collection. This may depend upon the type of data to be collected and/or be dependent upon other features or alternatives, as for example whether a long term quantity of data is desired, for ECG for example, or whether for example a relative single data point might be useful, as in some cases of pulse oximetry (sometimes a single saturation point might be of interest, as for example, if clearly too low, though comparison data showing trending over time, may indeed be more typical).
[0037] Several alternatives then present in FIG. 3, flow chart 300; a first such might be the following of flowline 312 to the transmission of data operation 303, which could then involve either wireless or wired (e.g., USB or other) data communication from the device 100 to data analysis and/or storage devices and/or systems (not separately shown in FIG. 3; could include computing devices, see e.g., FIG. 4 described below, or the like). Options from this point also appear; however, a first such might include following flow line 313 to the data analysis operation 304 for analyzing the data for determination of the relative health and/or for condition diagnosis of a patient. Computing systems, e.g., a computer (could be of many types, whether hand-held, personal or mainframe or other; see FIG. 4 and description below) could be used for this analysis; however, it could be that sufficient intelligence might be incorporated within the electronics 103 of device 100 such that some analysis might be operable on or within device 100 itself. A non-limiting example, might be a threshold comparison, as for example relative to pulse oximetry where when a low (or in some examples, perhaps a high) threshold level is reached an indicator or alarm might be activated all on/by the electronics 103 of the device 100.
[0038] A similar such example, might be considered by the optional alternative flow path 312a which itself branches into parts 312b and 312c. Following flow path 312a, and then, in a first example path 312b, a skip of the transmit data operation 303 can be understood whereby analysis 304 might be achieved without substantial data transfer. This could explain on board analysis, whether as for example according to the threshold example above, or might in some instances include more detailed analysis depending upon how much intelligence is incorporated on/in the electronics 103. Another view, is relative to how much transmission may be involved even if the transmission operation 303 is used; inasmuch as this could include at one level the transmission of data from the patient skin through the conductors 108, 109 and/or 110 through the traces 107 to the electronics 103 for analysis there. In other examples, of course, the transmission may include off-board downloading to other computing resources (e.g., FIG. 4). In some cases, such off-loading of the data may allow or provide for more sophisticated analysis using higher computing power resources.
[0039] Further alternatives primarily may involve data storage, both when and where, if used. As with intelligence, it may be that either some or no storage or memory may be made available in/by the electronics 103 on-board device 100. If some storage, whether a little or a lot, is made available on device 100, then, flow path 312a to and through path 312c may be used to achieve some storing of data 305. This may in many cases then, though not necessarily be before transmission or analysis (note, for some types of data multiple paths may be taken simultaneously, in parallel though perhaps not at the same time or serially (eg., paths 312b and 312c need not be taken totally to the exclusion of the other), so that storage and transmission or storage and analysis may occur without necessarily requiring a completion of any particular operation before beginning or otherwise implementing another). Thus, after (or during) storage 305, flow path 315a may be followed for stored data which may then be transmitted, by path 315b to operation 303, and/or analyzed, by path 315c to operation 304. In such a storage example, which in many cases may also be an on-board storage example, data can be collected then stored in local memory and later off-loaded/transmitted to one or more robust computing resources (e.g., FIG. 4) for analysis. Frequently, this can include long term data collection, e.g., in the manner of days or weeks or even longer, and may thus include remote collection when a patient is away from a doctor's office or other medical facilities. Thus, data can be collected from the patient in the patient's real world circumstances. Then, after collection, the data can be transmitted from its storage on device 100 back to the desired computing resource (FIG. 4, e.g.), and such transmission might be wireless or wired or come combination of both, as for example a blue tooth or WiFi connection to a personal computer (FIG. 4 for one example) which might then communicate the data over the internet to the designated computer for final analysis. Another example might include a USB connection to a computer, either to a PC or a mainframe (FIG. 4), and may be to the patient computer or to the doctor computer for analysis.
[0040] If little or no storage or memory is resident on device 100 (or in some examples even where there may be a large amount of resident memory available), then, relatively soon after collection, the data would need to or otherwise might desirably either or both be transmitted and then stored, see path 313a after operation 303, and/or transmitted and analyzed, paths 312 and 313. If path 313a is used, then, more typically, the data storage may be in/on computing resources (not shown in FIG. 3, but see FIG. 4 described below) off-board (though on-board memory could be used as well), and then, any of paths 315a, 315b and 315c may be used.
[0041] A feature hereof may include an overall system including one or more devices 100 and computing resources (see FIG. 4, for example) whether on-board device(s) 100, or separate, as for example in personal or mobile or hand-held computing devices (generally by FIG. 4), the overall system then providing the ability for the physician or doctor to have immediate, in-office analysis and presentation of collected test data. This would in some implementations allow for on-site data analysis from the device without utilization of a third party for data extraction and analysis.
[0042] Alternative implementations hereof may thus include one or more hardware and software combinations for multiple alternative data source interpretations. As noted above, a device 100 hereof includes hardware that monitors one or more of various physiologic parameters, then generates and stores the associated data representative of the monitored parameters. Then, a system which includes hardware such as device 100 and/or the parts thereof, and software and computing resources (FIG. 4, generally) for the processing thereof. The system then includes not only the collection of data but also interpretation and correlation of the data.
[0043] For example, an electrocardiogram trace that reveals a ventricular arrhythmia during intense exercise may be interpreted differently than the same arrhythmia during a period of rest. Blood oxygen saturation levels that vary greatly with movement can indicate conditions that may be more serious than when at rest, inter alia. Many more combinations of the four physiologic parameters are possible, and the ability of software hereof to display and highlight possible problems will greatly aid the physician in diagnosis. Thus, a system as described hereof can provide beneficial data interpretation.
[0044] Some of the features which can assist toward this end may be subsumed within one or more of operations 303 and 304 of FIG. 3, wherein data collected on a device 100 can rather simply be communicated/transmitted to computing resources (again, whether on-board device 100 or discrete therefrom as e.g., FIG. 4). For an example, when a patient having had a device applied (operation 301) may return to a physician's office after a test period wherein data was collected (operation 302) the device is connected via one or more data transmission alternatives, as for example, USB to a computer (Windows or Mac) (generally with reference to FIG. 4 and description thereof) in the office, allowing immediate analysis by the physician while the patient waits (note, the device 100 may first have been removed from the patient or might remain thereon pending transmission and analysis for determination of whether more data may be desired). In some implementations, data analysis time may be relatively quick, at approximately 15 minutes in some implementations, and might be achieved with a user-friendly GUI (Graphic User Interface) to guide the physician through the analysis software.
[0045] The analysis/software package may be disposed to present the physician with results in a variety of formats. In some implementations, an overview of the test results may be presented, either together with or in lieu of more detailed results. In either case, a summary of detected anomalies and/or patient-triggered events may be provided, either as part of an overview and/or as part of the more detailed presentation. Selecting individual anomalies or patient-triggered events may provide desirable flexibility to allow a physician to view additional detail, including raw data from the ECG and/or from other sensors. The package may also allow data to be printed and saved with annotations in industry-standard EHR formats.
[0046] In one implementation, patient data may be analyzed with software having the one or more of the following specifications. Some alternative capabilities may include: 1. Data Acquisition; i.e., loading of data files from device; 2. Data Formatting; i.e., formatting raw data to industry standard file formats (whether, e.g., aECG (xml); DICOM; or SCP-ECG) (note, such data formatting may be a part of Acquisition, Storage or Analysis, or may have translation from one to another (e.g., data might be better stored in a compact format that may need translation or other un-packing to analyze)); 3. Data Storage (whether local, at a clinic/medical facility level or e.g., in the Cloud (optional and allows offline portable browser based presentation/analysis); 4. Analysis which inter alia, may include, e.g., noise filtering (High pass/Low pass digital filtering); and/or QRS (Beat) detection (in some cases, may include Continuous Wave Transform (CWT) for speed and accuracy); and/or 5. Data/Results Presentation, whether including one or more graphical user interface(s) (GUIs) perhaps more particularly with an overall Summary and/or General Statistics and/or Anomaly Summary of Patient triggered event(s); presentation of additional levels of detail whether of Strip view(s) of anomaly data by incident (previous, next) Blood Oxygen saturation, stress correlation or the like; and/or allowing care provider bookmarking/annotations/notes by incident and/or Print capability.
[0047] Further, on alternative combinations of hardware with proprietary software packages: I) One on-device software package may be adapted to store the measurements from the data signals acquired from one or more of EKG/ECG (whether right leg and/or p-, qrs- and/or t-waves), or O2 saturation, or xyz acceleration, in a time concordant manner, so that a physician may access a temporal history of the measurements (say, in some examples, over a 1-2 week interval), which would provide useful information on what the patient's activity level was prior to, during, and after the occurrence of a cardiac event. ii) an alternative to alternately manage the real-time transmission of the real-time measured parameters to a nearby station or relay. And/or; iii) an off-device ECG analysis software aimed at recognizing arrhythmias.
[0048] The software mentioned above may be industry understood software provided by a 3rd party, or specially adapted for the data developed and transmitted by and/or received from a wearable device 100 hereof. Thorough testing using standard (MIT-BIH/AHA/NST) arrhythmia databases, FDA 510(k) approvals preferred. Such software may be adapted to allow one or more of automated ECG analysis and interpretation by providing callable functions for ECG signal processing, QRS detection and measurement, QRS feature extraction, classification of normal and ventricular ectopic beats, heart rate measurement, measurement of PR and QT intervals, and rhythm interpretation.
[0049] In many implementations, the software may be adapted to provide and/or may be made capable of supplying one or more of the following measurements: [0050] 1. Heart Rate Min, Max and Average [0051] 2. QRS duration average [0052] 3. PR interval average [0053] 4. QT interval average [0054] 5. ST deviation average and, may be adapted to recognize a broad range of arrhythmias such as those set forth here: [0055] 1. SINUS RHYTHM [0056] 2. SINUS RHYTHM+IVCD [0057] 3. SINUS BRADYCARDIA [0058] 4. SINUS BRADYCARDIA+IVCD [0059] 5. SINUS TACHYCARDIA [0060] 6. PAUSE [0061] 7. UNCLASSIFIED RHYTHM [0062] 8. ARTIFACT
[0063] This first group of 8 given above are arrhythmia types that may be recognizable even if there is no discernible P wave. They are the ones typically recognized by existing products in the outpatient monitoring market that we propose to address.
[0064] A second set or group of arrhythmias; below, may require a discernible and measurable P wave. Some implementations hereof may be adapted to be able to detect and recognize them, as device 100 may be able as described above to detect P waves, depending of course, and for example, on whether the strength of the P wave is affected by device 100 placement or patient physiology. [0065] 9. ATRIAL FIBRILLATION/FLUTTER SVR (slow) [0066] 10. ATRIAL FIBRILLATION/FLUTTER CVR (normal rate) [0067] 11. ATRIAL FIBRILLATION/FLUTTER RVR (rapid [0068] 12. FIRST DEGREE AV BLOCK+SINUS RHYTHM [0069] 13. FIRST DEGREE AV BLOCK+SINUS TACHYCARDIA [0070] 14. FIRST DEGREE AV BLOCK+SINUS BRADYCARDIA [0071] 15. SECOND DEGREE AV BLOCK [0072] 16. THIRD DEGREE AV BLOCK [0073] 17. PREMATURE ATRIAL CONTRACTION [0074] 18. SUPRAVENTRICULAR TACHYCARDIA [0075] 19. PREMATURE VENTRICULAR CONTRACTION [0076] 20. VENTRICULAR COUPLET [0077] 21. VENTRICULAR BIGEMINY [0078] 22. VENTRICULAR TRIGEMINY [0079] 23. IDIOVENTRICULAR RHYTHM [0080] 24. VENTRICULAR TACHYCARDIA [0081] 25. SLOW VENTRICULAR TACHYCARDIA
[0082] Further in alternative software implementations; some sample screenshots are shown in FIG. 5. A first such alternative is shown in FIG. 5A, which is an example screenshot showing ECG and Oxygen Saturation data taken by using a patch device such as a device 100 hereof. An extremely clean signal is shown (no filtering or smoothing has been done on this data). Distinct p-waves are also shown (3 of which are shown as an example with arrows). P wave detection can be extremely important for ECG anomaly detection. Oxygen Saturation, as measured by Pulse Oxymetry, is shown on the bottom plot. This is data taken by the a device on the chest, and is taken in time concordance with the ECG data.
[0083] Another alternative is shown in FIG. 5B, which is an example screenshot of Analysis Software. This is a sample of ECG data taken from the MIT-BIH Arrhythmia Database, Record 205. As analyzed by the Analysis system hereof, we see in the Event Occurrences Summary list (top, left) five (5) anomaly types (plus normal sinus rhythm). This list also shows the number of occurrences of each anomaly, total duration of the anomaly in the complete ECG, and the percent time this anomaly occurs in the complete ECG. To view specific instances of each anomaly, the user double clicks the specific row in the Event Occurrences Summary list, as shown in FIG. 5C.
[0084] As introduced, FIG. 5C is an example screenshot showing specific instance of Ventricular Tachycardia. The ECG plot automatically navigates to the specific time in the ECG waveform, and marks the beginning and end of the event. More detailed data about this specific event is now shown in the Occurrence Details: HR Average, HR Max, etc. for the duration of this event. To show the instances of another anomaly in this ECT, the user can click on the Premature Ventricular Contraction (PVC) row of the Event Occurrences Summary, as shown FIG. 5D.
[0085] As introduced, FIG. 5D is an example screenshot showing specific instance of Premature Ventricular Contraction. This shows occurrences of the PVC. The Start Times list (middle top) shows all instances of PVC occurrences in this ECG, and lists the start time for each occurrence. In this case, the user can click on the PVC that starts at 00:15:27 (the 11.sup.th occurrence). The ECG plot is automatically taken to this point in time to show and indicate the PVC instances in the waveform. Since there are 3 instances of a PVC in this timeslot, all 3 occurrences are marked.
[0086] Some further alternatives may include data transmission and/or interpretation by local medical facilities, whether physician or doctor offices or e.g., ICU/CCU (Intensive Care/Coronary Care Units). Accordingly, a device 100 hereof that will measure one or more of a variety of physiologic signals, possibly including electrocardiogram, photoplethysmogram, pulse oximetry and/or patient acceleration signals will be placed on the patient's chest and held with an adhesive as described herein. The device transmits the physiologic signals wirelessly or by wire (e.g., USB) to a nearby base station for interpretation and further transmission, if desired. The wireless transmission may use Bluetooth, WiFi, Infrared, RFID (Radio Frequency IDentification) or another wireless protocol. The device may be powered by wireless induction, battery, or a combination of the two. The device 100 monitors physiological signals and/or collects data representative thereof. The collected data may then be transmitted wirelessly or by wire connection, in real time, to the nearby base station. The device may be wirelessly powered by the base station or by battery, removing the need for wires between the patient and the station.
[0087] Thus, some of the alternative combinations hereof may include one or more of: 1) medical grade adhesives (from many possible sources) selected for their ability to maintain in intimate contact with the skin without damaging it, for several days (up to, say 10 days or two weeks in some examples), as well as operability with different types of sensors; 2) conductive electrodes or photo-sensitive detectors able to supply electrical signals from the skin or from the photo-response of cutaneous or subcutaneous tissues to photo-excitation; 3) amplifiers, microprocessors and memories, capable of treating these signals and storing them; 4) power supply for the electronics hereof with stored or with wirelessly accessible re-chargeability; 5) flex circuits capable of tying the above elements together within a flexible strip capable of conforming to a cutaneous region of interest.
[0088] Examples of physiological parameters that may be subject to monitoring, recordation/collection and/or analyzing may include one or more of: electrocardiograms, photo responses of photo-excited tissues for e.g., oxygen saturation of blood; pulse rates and associated fluctuations; indications of physical activity/acceleration. One or more of these may be used in monitoring ambulatory cardiac outpatients over several days and nights, which could thereby provide for recording, for post-test analysis, several days' worth of continuous ECG signals together with simultaneous recording of O2 saturation and an index of physical exertion. Similarly, one or more of these may be used in monitoring ambulatory pulmonary outpatients over several days and nights for recording, for post-test analysis, O2 saturation together with simultaneous recording of an index of physical activity. Alternatively and/or additionally, one or more of these could be used for monitoring in-patients or other patients of interest, as for example neonatals, wirelessly (or in some cases wired), whether in clinics, emergency rooms, or ICUs, in some instances detecting the parameters of EKG, O2 and/or physical exertion, but instead of storing them would transmit them wirelessly to either a bedside monitor or a central station monitor, thus freeing the patient from attachment to physical wires.
[0089] An exemplary computer system or computing resources which may be used herewith will now be described, though it should be noted that many alternatives in computing systems and resources may be available and operable within the reasonably foreseeable scope hereof so that the following is intended in no way to be limiting of the myriad possible computational alternatives properly intended within both the spirit and scope hereof.
[0090] Some of the implementations of the present invention include various steps. A variety of these steps may be performed by hardware components or may be embodied in machine-executable instructions, which may be used to cause a general-purpose or special-purpose processor programmed with the instructions to perform the steps. Alternatively, the steps may be performed by a combination of hardware, software, and/or firmware. As such, FIG. 4 is an example of computing resources or a computer system 400 with which implementations hereof may be utilized. According to the present example, a sample such computer system 400 may include a bus 401, at least one processor 402, at least one communication port 403, a main memory 404, a removable storage media 405, a read only memory 406, and a mass storage 407. More or fewer of these elements may be used in a particular implementation hereof.
[0091] Processor(s) 402 can be any known processor, such as, but not limited to, an Intel.RTM. Itanium.RTM. or Itanium 2.RTM. processor(s), or AMD.RTM. Opteron.RTM. or Athlon MP.RTM. processor(s), or Motorola.RTM. lines of processors. Communication port(s) 403 can be any of an RS-232 port for use with a modem based dialup connection, a 10/100 Ethernet port, a Universal Serial Bus (USB) port, or a Gigabit port using copper or fiber. Communication port(s) 403 may be chosen depending on a network such a Local Area Network (LAN), Wide Area Network (WAN), or any network to which the computer system 400 connects or may be adapted to connect.
[0092] Main memory 404 can be Random Access Memory (RAM), or any other dynamic storage device(s) commonly known in the art. Read only memory 406 can be any static storage device(s) such as Programmable Read Only Memory (PROM) chips for storing static information such as instructions for processor 402.
[0093] Mass storage 407 can be used to store information and instructions. For example, hard disks such as the Adaptec.RTM. family of SCSI drives, an optical disc, an array of disks such as RAID, such as the Adaptec family of RAID drives, or any other mass storage devices may be used.
[0094] Bus 401 communicatively couples processor(s) 402 with the other memory, storage and communication blocks. Bus 401 can be a PCI/PCI-X or SCSI based system bus depending on the storage devices used.
[0095] Removable storage media 405 can be any kind of external hard-drives, floppy drives, IOMEGA.RTM. Zip Drives, Compact Disc-Read Only Memory (CD-ROM), Compact Disc-Re-Writable (CD-RW), Digital Video Dis-Read Only Memory (DVD-ROM).
[0096] The components described above are meant to exemplify some types of possibilities. In no way should the aforementioned examples limit the scope of the invention, as they are only exemplary embodiments.
[0097] Embodiments of the present invention relate to devices, systems, methods, media, and arrangements for monitoring and processing cardiac parameters and data, inter alia. While detailed descriptions of one or more embodiments of the invention have been given above, various alternatives, modifications, and equivalents will be apparent to those skilled in the art without varying from the spirit of the invention. Therefore, the above description should not be taken as limiting the scope of the invention, which is defined by the appended claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.