Improved Explosive Composition
ARAOS; Miguel Ulises ; et al.
U.S. patent application number 16/468137 was filed with the patent office on 2020-01-02 for improved explosive composition. The applicant listed for this patent is CMTE Development Limited. Invention is credited to Miguel Ulises ARAOS, Italo ONEDERRA.
| Application Number | 20200002241 16/468137 |
| Document ID | / |
| Family ID | 62557765 |
| Filed Date | 2020-01-02 |


| United States Patent Application | 20200002241 |
| Kind Code | A1 |
| ARAOS; Miguel Ulises ; et al. | January 2, 2020 |
IMPROVED EXPLOSIVE COMPOSITION
Abstract
The present invention provides an explosive composition comprising from about 2 to about 25 w/w hydrogen peroxide, from greater than 0 and up to about 90% w/w of one or more of other oxidisers. The present invention also provides a method of 5 preparing an explosive composition and use of the explosive composition of the invention to break and move ground, such as in mining operations.
| Inventors: | ARAOS; Miguel Ulises; (Jindalee, AU) ; ONEDERRA; Italo; (Pinjarra Hills, AU) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 62557765 | ||||||||||
| Appl. No.: | 16/468137 | ||||||||||
| Filed: | December 12, 2017 | ||||||||||
| PCT Filed: | December 12, 2017 | ||||||||||
| PCT NO: | PCT/AU2017/051368 | ||||||||||
| 371 Date: | June 10, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C06B 47/145 20130101; C06B 21/0008 20130101; C06B 43/00 20130101; C06B 31/285 20130101; C06B 23/003 20130101 |
| International Class: | C06B 47/14 20060101 C06B047/14; C06B 21/00 20060101 C06B021/00; C06B 43/00 20060101 C06B043/00; C06B 23/00 20060101 C06B023/00; C06B 31/28 20060101 C06B031/28 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 12, 2016 | AU | 2016905130 |
Claims
1. An explosive composition comprising: a. from about 2 to about 25% w/w hydrogen peroxide; and b. from greater than 0 and up to about 90% w/w of at least one other oxidiser.
2. The explosive composition according to claim 1, wherein the at least one other oxidiser is selected from the group consisting of nitrate salts, perchlorate salts, sodium peroxide, potassium peroxide and optionally nitric acid, wherein the nitrate salts are selected from the group consisting of ammonium nitrate, calcium ammonium nitrate, calcium nitrate and sodium nitrate, and wherein the perchlorate salts are selected from the group consisting of ammonium perchlorate and sodium perchlorate.
3-6. (canceled)
7. The explosive composition according to claim 1, comprising at most 50% w/w of water.
8. (canceled)
9. The explosive composition according to claim 1, comprising from about 5 to about 25% w/w hydrogen peroxide.
10. The explosive composition according to claim 1, comprising from about 0.1 to about 75% w/w of the at least one other oxidiser.
11. The explosive composition according to claim 1, further comprising at least one of an additive selected from the group consisting of a sensitiser, fuel, secondary fuel, water, thickener, crosslinker, emulsifier, and energy diluent.
12. (canceled)
13. The explosive composition according to claim 11, wherein the sensitiser comprises at least one of a compressible material and bubbles of gas, wherein the bubbles of gas are formed in situ and consist of one of N.sub.2, O.sub.2, CO.sub.2, NO, H.sub.2 bubbles and a mixture thereof, and wherein the compressible material is gas entrapped material selected from the group consisting of glass microballoons, ceramic microballoons, plastic microballoons and EPS with a particle size smaller than 2 mm.
14. (canceled)
15. The explosive composition according to claim 13, further comprising a sufficient amount of the sensitiser such that a density of the explosive composition is controlled to around 0.3 to 1.4 g/cm.sup.3.
16. The explosive composition according to claim 11, wherein said fuel is one of a water soluble fuel and a water insoluble fuel, said water soluble fuel is selected from the group consisting of an amine nitrate, gums, glycerol, ethylene glycol, propylene glycol, sugar molasses, formamide, urea and a mixture thereof, and wherein the water insoluble fuel is selected from the group consisting of diesel, oils, vegetable oils, and mixtures thereof.
17-18. (canceled)
19. The explosive composition according to claim 1, wherein the explosive composition is a watergel composition which comprises at least one of a thickeners and a crosslinker, wherein the thickener is selected from the group consisting of guar gum, xanthan gum, sodium alginate, polyacrylamides and polyvinyl alcohols, and wherein the crosslinker is selected from the group consisting of antimony salts, chromic salts, phosphoric acid and mixtures thereof.
20. (canceled)
21. The explosive composition according to claim 1, wherein the explosive composition is an emulsion composition which comprises at least one emulsifiers selected from the group consisting of PIBSA-amine derivatives, SMO, lecithin and a mixture thereof.
22. The explosive composition according to claim 1, wherein the explosive composition comprises at least one energy diluents selected from the group consisting of EPS, crumb rubber tyre, popcorn, and plastic beads.
23. (canceled)
24. The explosive composition according to claim 1, having an Oxygen balance between +10 and -10.
25. The explosive composition according to claim 1, having a viscosity of from 5 to 50 Pa*s.
26-58. (canceled)
59. A method of breaking and moving ground comprising steps of: applying the explosive composition according to claim 1 to a blasthole; and detonating the explosive composition.
Description
[0001] This application claims priority to and the benefit of Australian provisional patent application no. 2016905130 dated 12 Dec. 2016, which is incorporated herein by cross-reference in its entirety.
FIELD OF THE INVENTION
[0002] The present invention relates to an improved explosive composition. More particularly, the invention relates to an explosive composition comprising hydrogen peroxide (HP) in combination with one or more other oxidisers.
[0003] The invention has been developed primarily for use as a hydrogen peroxide/nitrate salt-based explosive composition for use in mining applications and will be described hereinafter with reference to this application. However, it will be appreciated that the invention is not limited to this particular field of use.
BACKGROUND OF THE INVENTION
[0004] The following discussion of the prior art is provided to place the invention in an appropriate technical context and enable the advantages of it to be more fully understood. It should be appreciated, however, that any discussion of the prior art throughout the specification should not be considered as an express or implied admission that such prior art is widely known or forms part of common general knowledge in the field.
[0005] Nearly all commercial and mining explosives used in the world today are based on ammonium nitrate (AN) or combinations of AN with smaller quantities of other alkaline and/or alkaline earth nitrate salts, e.g. sodium nitrate (SN) or calcium nitrate (CN). AN, which is a strong oxidiser, has been used as the base of commercial explosives for at least the last 50-60 years. Most explosives of this type rely on the energetic reaction of nitrogen compounds incorporated within the explosive to provide the necessary explosive power.
[0006] Initially, mining companies used AN as an explosive on its own. However, they soon realised that the addition of diesel increased the energy output without a large increase on costs (ammonium nitrate--fuel oil, now commonly referred to as `ANFO`). However, the water resistance of ANFO is quite poor, which limited its use in wet blast holes. To ameliorate this issue, slurries and watergels were developed. Slurries typically comprise AN dissolved/dispersed in water, and other salts such as calcium nitrate, sodium nitrate, amine nitrates, perchlorates, etc. and other additives such as guar gum (as thickener) and water soluble or insoluble fuels (glycerol, MMAN, diesel, etc). They can also be blended with ANFO depending on the characteristics of the ground being blasted. Slurries also typically include solid sensitisers (aluminium and high explosives such as TNT, RDX, etc) to enable the slurry to detonate and to minimise misfires. Watergels have similar compositions to slurries, however, crosslinkers can be added to enhance the water resistance of the product.
[0007] One of the drawbacks of watergels and slurries is that there is a limit of AN which can be incorporated into the solution. This drawback was overcome by the development of water-in-oil emulsions. These emulsions can contain AN in high concentration (see U.S. Pat. No. 3,447,978) as emulsions are manufactured at high temperatures. Water-in-oil emulsions are made of a hot aqueous phase (composed of AN, other nitrate salts, perchlorate salts, etc.) dispersed into an organic fuel. The aqueous--organic mixture is stabilised by the use of an emulsifier. Emulsions can also be blended with ANFO in different ratios so suit the ground to be blasted.
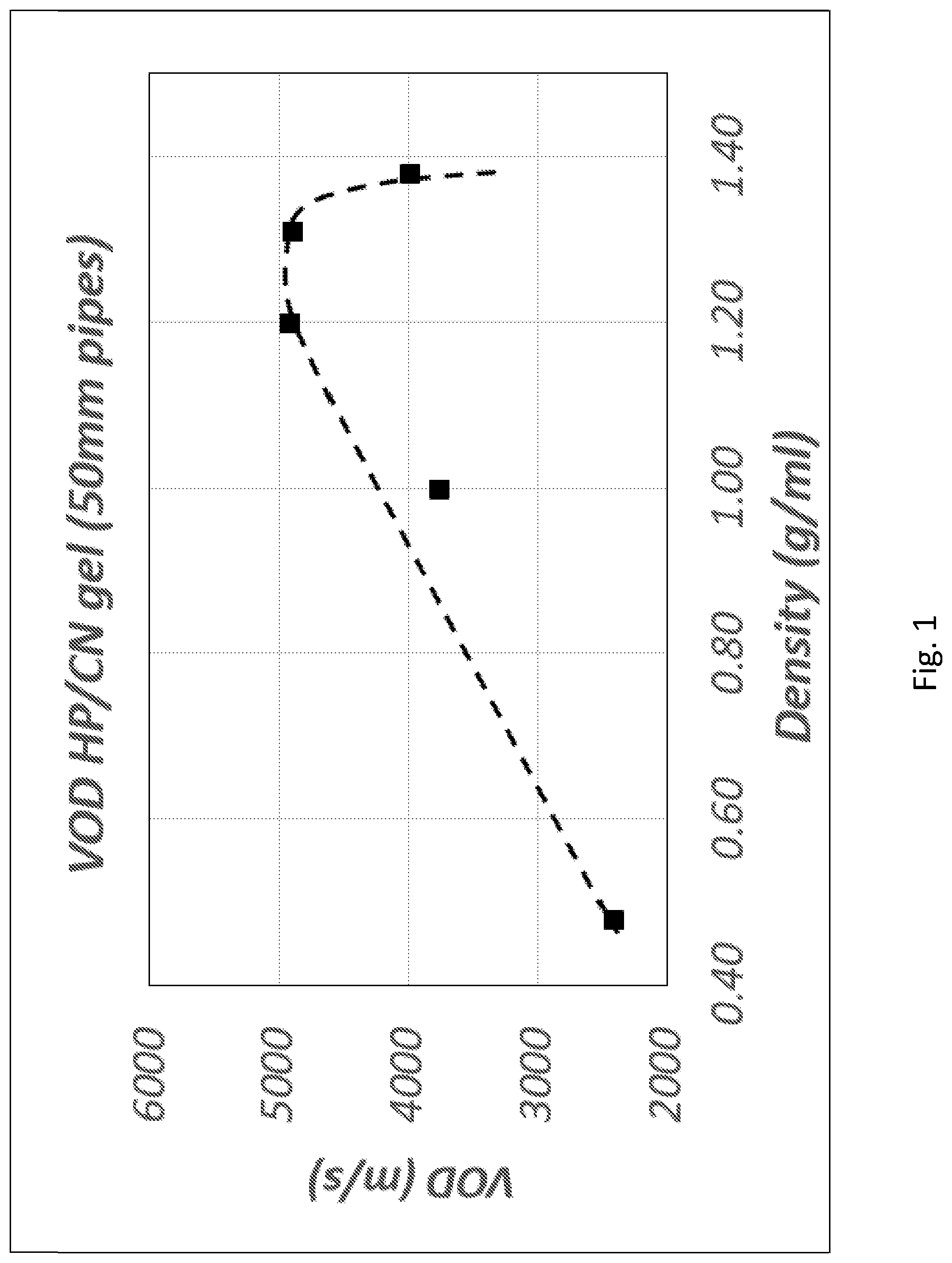
[0008] Despite the development of AN emulsions, AN slurries, and watergels, however, there is still a need to develop improved explosives, which are suitably more cost effective compared to existing explosive compositions and are capable of being produced in large quantities to meet the high demand from industry. It would be advantageous to use less AN in the formulation and instead use other types of nitrates to provide alternatives to the usage of AN. Additionally, such substitutes should preferably be safer, have a relatively low carbon footprint, manufactured nearby the point of use to minimise the transport on public roads, able to be manufactured on an as-needs basis to minimise the need for stockpiling and to increase safety, allow for the use of existing delivery equipment, and/or produce a lower amount of (or no) toxic nitrogen oxide fumes (NO.sub.x) upon detonation, etc. It would also be ideal if there are no onerous regulatory requirements for such a substitute, thereby reducing administrative costs. It would also be preferable for the explosive composition to be crosslinkable in-situ to increase viscosity down the blasthole.
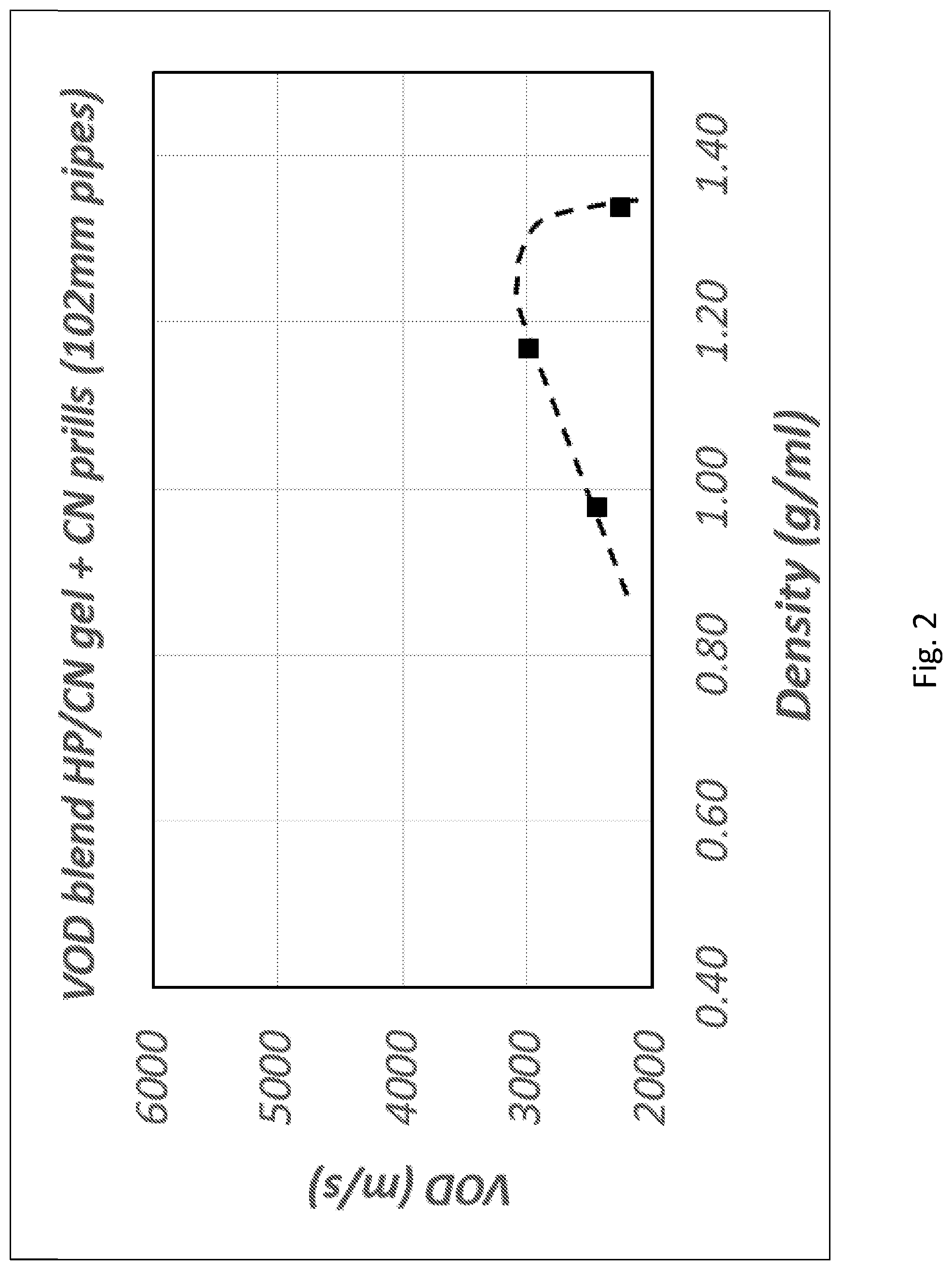
[0009] Despite the advances on the types of compositions that can be manufactured from ammonium nitrate, one of the disadvantages is that during the detonation NO.sub.x fumes can be generated, due to the presence of nitrogen compounds in the explosive composition (from nitrates). These NO.sub.x fumes are toxic and can affect the health of mine site personnel. Therefore the emission of NO.sub.x fume after blasting is a safety issue and, in countries like Australia, there are now strict regulatory controls in place to manage such emissions. See for example "Queensland Guidance Note: Management of oxides of nitrogen in open cut blasting" issued by the regulator in Queensland, Australia, 2011. Likewise, explosive manufacturers in Australia have also issued a code of practice to manage the NO.sub.x fumes after blasting (AEISG Code of Practice, Prevention and Management of Blast Generated NO.sub.x Gases in Surface Blasting, 2011). Therefore, there is a need to find explosive compositions that substantially reduce the production of NO.sub.x.
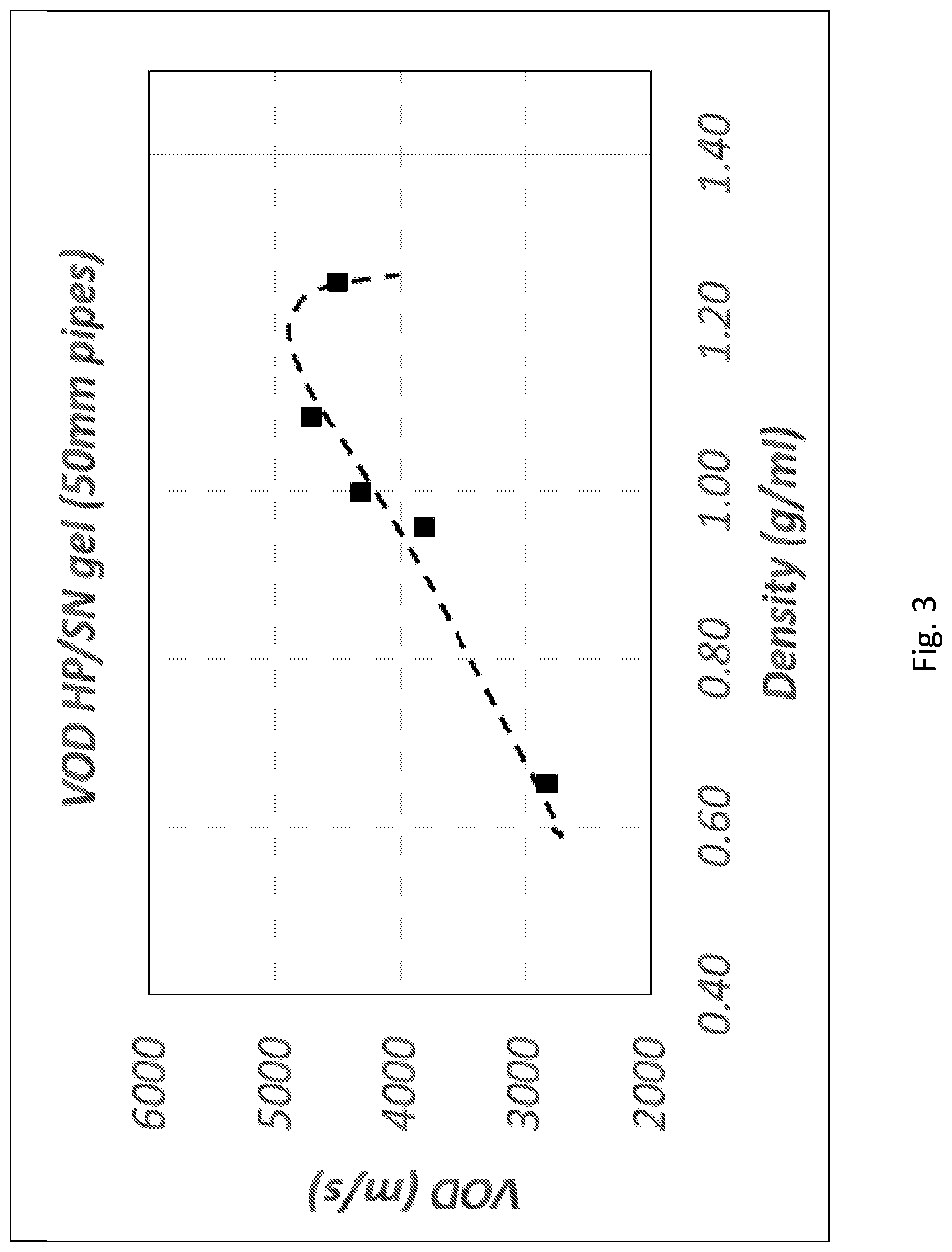
[0010] One material that is also an oxidiser and that has the potential to meet at least some of these needs is hydrogen peroxide (HP).
[0011] International PCT publication WO 2013/013272 describes an explosive composition comprising HP and a sensitiser, wherein the sensitiser comprises a compressible material having one or more gas-filled voids and/or bubbles of gas. WO 2013/013272 also describes a method of preparing an explosive composition comprising combining hydrogen peroxide and a sensitiser, wherein the sensitiser comprises a compressible material and/or bubbles of gas, and use of said explosive composition. The compositions of WO 2013/013272 may contain a very high concentration of HP. Despite prior art examples of HP-based explosives, there is still a need to develop improved explosive compositions. In particular, there is a need to develop improved explosive compositions made from HP and one or more other oxidisers in which the sensitivity, density, velocity of detonation (VOD) and the delivery of the energy can be controlled.
[0012] It is an object of the present invention to overcome or ameliorate one or more of the disadvantages of the prior art, or at least to provide a useful alternative.
SUMMARY OF THE INVENTION
[0013] The present invention relates to explosives for use in commercial, construction, civil, agriculture, mining, and similar fields. However, it will be appreciated that the invention could be utilised in other related fields.
[0014] A preferred objective of an embodiment of the present invention is to provide an explosive composition which meets one or more of the following objectives: is conveniently prepared, has improved stability over time in situ, can use large amounts of sustainable fuels (which lowers the carbon footprint of the explosive), and can use large amount of nitrates other than AN (which lowers the dependency on AN).
[0015] According to a first aspect the present invention provides an explosive composition comprising:
[0016] a. from about 2 to about 25% w/w hydrogen peroxide; and
[0017] b. from greater than 0 and up to about 90% w/w one or more other oxidisers.
[0018] In one embodiment, there is provided an explosive composition comprising: [0019] from about 2 to about 25% w/w hydrogen peroxide; and [0020] from greater than 0 and up to about 90% w/w of one or more of other oxidisers; and [0021] from about 15 to about 25% w/w of fuels, preferably sustainable fuels.
[0022] According to a preferred embodiment, the present invention provides an explosive composition comprising: [0023] from about 2 to about 25% w/w hydrogen peroxide; [0024] from greater than 0 and up to about 90% w/w of one or more other oxidisers; [0025] a fuel phase; [0026] a thickener and/or crosslinker; [0027] a secondary fuel; and [0028] a sensitiser.
[0029] Preferably the composition comprises from about 5 to about 25% w/w hydrogen peroxide. Preferably the one or more other oxidiser(s) is a salt or acid selected from the group consisting of nitrate salts, perchlorate salts, peroxide salts, or nitric acid. For example, the one or more other oxidisers may be selected from the group consisting of nitrate salts, perchlorate salts, sodium peroxide, potassium peroxide and optionally nitric acid. The perchlorate salts may be selected from ammonium perchlorate and sodium perchlorate. Preferably the salts are selected from ammonium nitrate (AN), calcium nitrate (CN), calcium ammonium nitrate (CAN), sodium nitrate (SN), NH.sub.4ClO.sub.4, NaClO.sub.4, Na.sub.2O.sub.2, K.sub.2O.sub.2 or mixtures thereof. For example, the nitrate salts may be selected from ammonium nitrate, calcium nitrate and sodium nitrate. By way of further example, the nitrate salts may be selected from calcium ammonium nitrate, calcium nitrate and sodium nitrate. In one embodiment, the explosive composition is devoid of AN. The one or more other oxidisers in the explosive composition may be selected from calcium nitrate and sodium nitrate. Preferably the explosive composition contains from 0.1 to 75% w/w of one or more other oxidisers. In one embodiment, the explosive composition contains from 0.1 to 75% w/w of dissolved salts. In a preferred embodiment, at least some of at least one of the one or more other oxidisers is not fully dissolved in the explosive composition but is present as a solid oxidiser, e.g., in the form of powder or prills. In such an embodiment, the one or more other oxidisers that is at least partially present as a solid may be selected from the group consisting of AN, SN, CN, CAN, or mixtures thereof. The composition may comprise a solid nitrate oxidiser, for example, in an amount of from contains from 0.1 to 70% w/w. The composition may comprise water. The solid nitrate oxidiser may be selected from the group of AN, SN, CAN or mixtures thereof.
[0030] Preferably the composition comprises 50% w/w or less of water, or 30% w/w or less of water, or 25% w/w or less of water. The explosive composition may further comprise one or more other components selected from the group consisting of sensitisers, fuels, secondary fuels, water, thickeners, crosslinkers, emulsifiers, energy diluents and optionally other additives.
[0031] Preferably the explosive composition comprises a sensitiser. Preferably the sensitiser comprises a compressible material and/or bubbles of gas, or comprises a gas entrapped material. The bubbles of gas may be formed in situ and consist of N.sub.2, O.sub.2, CO.sub.2, NO.sub.x or H.sub.2 bubbles or a mixture thereof. The gas entrapped material may be selected from glass microballoons, ceramic microballoons, plastic microballoons or EPS with a particle size smaller than 2 mm. The explosive composition preferably has a density controlled by adding a sufficient amount of sensitiser such that the composition is detonation-sensitive. The density may be controlled to around 0.3 to 1.4 g/cm.sup.3, or may be formulated to around 0.3 to 1.4 g/cm.sup.3.
[0032] The composition may comprise a fuel, or it may comprise a fuel and a secondary fuel. The fuel may be a water soluble fuel. The water soluble fuel may be selected from an amine nitrate or urea or a mixture thereof. The explosive composition may contain from 0.1 to 30% w/w of water soluble fuel. The composition may contain between 13-25% w/w of the fuel phase. Preferably the fuel phase comprises one or more components selected from the group consisting of gums, glycerol, ethylene glycol, propylene glycol, sugar molasses, formamide or mixtures thereof. For example, the fuel phase may comprise one or more components selected from the group consisting of gums, glycerol, ethylene glycol, propylene glycol, formamide or mixtures thereof. The composition may comprise a sustainable fuel. The sustainable fuel may be present in the composition in an amount of between 15 and 25% w/w.
[0033] Preferably the composition is a watergel composition, in which case the composition may comprise a thickener or crosslinker. The composition may be a watergel composition comprising a thickener and a crosslinker. The thickener may be suspended in the fuel. The thickener may be selected from the group consisting of guar gum, xanthan gum, sodium alginate, polyacrylamides, and polyvinyl alcohols. The composition may comprise a crosslinker selected from the group of antimony salts, chromic salts, phosphoric acid or mixtures thereof. The fuel phase may comprise one or more water insoluble fuels selected from the group consisting of diesel, oils, vegetable oils, or mixtures thereof. Accordingly, the explosive composition may be formulated as an emulsion, in which case it may comprise an emulsifier. The emulsifier may be mixed in the fuel. The emulsifier may be selected from the group consisting of PIBSA-amine derivatives, SMO, lecithin or a mixture thereof.
[0034] Preferably the composition is formulated to have an oxygen balance between +10 to -10, e.g., the composition may have an oxygen balance of between +5 and -5. The explosive composition may contain from 1 to 800% v/v of an energy reducing agent (i.e., diluent material). The energy diluent material may be selected from the group consisting of EPS, crumb rubber tyre, popcorn, and plastic beads. The hydrogen peroxide, one or more other oxidisers and a fuel containing thickeners may be mixed until a thick material is formed, with a viscosity between 5-50 Pa*s. The composition may have a viscosity of from 5 to 50 Pa*s.
[0035] Many advantages result from the inventive explosive compositions taught herein. For example, certain formulations of the compositions of the invention may be more convenient to prepare, more cost effective compared to existing explosive compositions, safer to produce and to store, and/or capable of being produced in large quantities to meet the demand from the mining industry. Added safety is provided by the use of, for example, relatively high concentrations of CAN, which is known to be less sensitive to unintended detonation than AN. The same applies the use of SN in the formula. The present invention is therefore a significant advance in the art. The explosive compositions of the invention utilise HP, which is a sustainably-produced material that has a relatively low carbon footprint compared to other types oxidisers used in the art. The composition may also use sustainable fuels, as opposed to current technology used in the mining industry. To explain, current explosive compositions use a low concentration of fuel, which is typically sourced from the petrochemical industry. In contrast, the inventive explosive compositions disclosed herein are able to incorporate relatively higher amounts of sustainable fuel than commercially available or prior art explosive compositions. Accordingly, the lower reliance on fuels from the petrochemical industry is a significant advance in the art. Additionally the increase of sustainable fuels in the formulation means that the amount of oxidiser material in the formulation can be decreased without affecting the detonation properties.
[0036] The present invention is counterintuitive to the common knowledge in the art. To explain, it is currently believed that it is impossible or very difficult to detonate a composition that has a relatively high concentration of nitrates, such as CAN. However, surprisingly, the present invention provides the ability to detonate compositions that contain up to 30% w/w of CAN, and even up to 65% w/w. This aspect of the present invention is a significant advance in the art. The present invention also provides the ability to incorporate a relatively high amount of nitrates by making a watergel or emulsion, which already comprises HP/nitrate in the aqueous phase, with a further solid nitrate phase in the form of prills. Use of oxidiser in solid form enables some control over the density of the overall composition, and therefore provides some control over the VOD, as will be discussed below.
[0037] The explosive compositions of the invention may also be formulated into emulsion form. It will also be appreciated that the inventive compositions of the invention may produce low amounts of NO.sub.x, and in some forms of the invention no NO.sub.x at all.
[0038] The compositions of the invention are contemplated to provide several advantages over the prior art, such as better stability over time than explosive compositions comprising a higher percentage of HP. This is advantageous in the context of both safety and storage. More specifically, the "sleeping time" (i.e. the time over which an explosive deteriorates in situ such that its velocity of detonation decreases below a defined useful limit of such an explosive composition when it is in contact with rocks) is expected to be greater than an explosive comprising a higher percentage of HP. By way of example, a CAN/HP composition according to the invention has been made and found to have a sleeping time that is comparable/compatible for application in a commercially viable product, for example, a sleeping time beyond 24 hours or more. It is therefore contemplated that larger blasts are possible because there is a longer time (e.g. several days) over which explosives can be loaded into many holes before the first-loaded explosive becomes unstable in its hole. More holes can therefore be loaded before detonation.
[0039] Another advantage is that these compositions detonate when CAN is used. This is unexpected because CAN is not easy to detonate. Accordingly, given the relatively reduced propensity for CAN to detonate (compared to AN), the inventors contemplate that there are safety advantages in using combinations of CAN and CAN/SN compared to AN-based explosives with CN and SN additions.
[0040] According to a second aspect the present invention provides a method of preparing an explosive composition according to the first aspect, the method comprising combining hydrogen peroxide and one or more other oxidisers, optionally further comprising sensitisation of the composition. Preferably the hydrogen peroxide, other oxidiser(s) and fuel are mixed until a thick material is formed, with a viscosity between about 5 to 50 Pa*s. Preferably the density of the composition is controlled by adding a sufficient amount of sensitiser such that the composition is detonation-sensitive.
[0041] According to a third aspect the present invention provides use of an explosive composition according to the first aspect to break and move ground, e.g. in mining operations.
Definitions
[0042] In describing and claiming the present invention, the following terminology will be used in accordance with the definitions set out below. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments of the invention only and is not intended to be limiting. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one having ordinary skill in the art to which the invention pertains.
[0043] Unless the context clearly requires otherwise, throughout the description and the claims, the words `comprise`, `comprising`, and the like are to be construed in an inclusive sense as opposed to an exclusive or exhaustive sense; that is to say, in the sense of `including, but not limited to`.
[0044] Other than in the operating examples, or where otherwise indicated, all numbers expressing quantities of ingredients or reaction conditions used herein are to be understood as modified in all instances by the term `about`. The examples are not intended to limit the scope of the invention. In what follows, or where otherwise indicated, `%` will mean `weight %`, `ratio` will mean `weight ratio` and `parts` will mean `weight parts`.
[0045] Unless the context clearly indicates otherwise, all references to a component being present at a certain % w/w are with respect to the entire explosive composition. For example, an explosive composition comprising 2-25% w/w hydrogen peroxide refers to an explosive composition comprising 2-25 g hydrogen peroxide per 100 g of the explosive composition.
[0046] The term HP is an abbreviation for hydrogen peroxide.
[0047] The term AN means ammonium nitrate.
[0048] CN means calcium nitrate tetra hydrate.
[0049] CAN means calcium ammonium nitrate
[0050] SN is an abbreviation for sodium nitrate.
[0051] ANFO is an abbreviation for ammonium nitrate fuel oil.
[0052] Amine nitrates is an abbreviation for monomethylamine or ethyl amine or propyl amine nitrate.
[0053] Sensitiser means an additive that introduces voids in the composition. Sensitisers can increase the sensitivity to detonation of energetic materials. The sensitiser can be chemically generated voids (gas bubbles) or can enclose or entrap a gas (examples of which include ceramic/glass microballoons, EPS and polyurethane foams).
[0054] GMB is an abbreviation for glass micro balloons.
[0055] EPS is an abbreviation for expanded polystyrene.
[0056] TNT means trinitrotoluene.
[0057] HMX refers to octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine.
[0058] RDX refers to 1,3,5-trinitroperhydro-1,3,5-triazine.
[0059] VOD refers to velocity of detonation in m/sec.
[0060] OB means oxygen balance.
[0061] The term g/cm.sup.3 is has the same meaning as g/ml.
[0062] The terms `preferred`, `preferably` and `suitably` refer to embodiments of the invention that may afford certain benefits, under certain circumstances. However, other embodiments may also be preferred, under the same or other circumstances. Furthermore, the recitation of one or more preferred embodiments does not imply that other embodiments are not useful, and is not intended to exclude other embodiments from the scope of the invention.
[0063] The terms `a`, `an` and `the` mean `one or more`, unless expressly specified otherwise. The terms `an embodiment`, `embodiment`, `embodiments`, `the embodiment`, `the embodiments`, `an embodiment`, `some embodiments`, `an example embodiment`, `at least one embodiment`, `one or more embodiments` and `one embodiment` mean `one or more (but not necessarily all) embodiments of the present invention(s)` unless expressly specified otherwise.
[0064] The prior art referred to in this specification is incorporated herein by reference.
BRIEF DESCRIPTION OF THE DRAWINGS
[0065] FIG. 1 is a graph plotting VOD against density for an approximate 20% HP and approximate 40% CAN/fuel-based gel in a 50 mm diameter pipe. Sensitization was achieved by the addition of a gassing agent that decomposed HP to O.sub.2 bubbles.
[0066] FIG. 2 is a graph plotting VOD against density for a blend made with approximately 6% HP and approximate 65% CAN/fuel-based gel and CAN prill in a ratio 45:55 (gel:prill) in a 102 mm diameter pipe. Sensitization was achieved by the addition of a gassing agent that decomposed HP to O.sub.2 bubbles.
[0067] FIG. 3 is a graph plotting VOD against density for an approximate 30% HP and approximate 24% SN/fuel-based gel in a 50 mm diameter pipe. Sensitization was achieved by the addition of a gassing agent that decomposed HP to O.sub.2 bubbles.
[0068] FIG. 4 is a graph plotting VOD against density for a blend made with approximately 16% HP and approximate 44% SN/fuel-based gel and SN prill in a ratio 70:30 (gel:prill) and in a 102 mm diameter pipe. Sensitization was achieved by the addition of a gassing agent that decomposed HP to O.sub.2 bubbles.
DETAILED DESCRIPTION OF THE INVENTION
[0069] According to a first aspect, the present invention provides an explosive composition comprising: [0070] from about 2 to about 25% w/w hydrogen peroxide; and [0071] from greater than 0 and up to about 90% w/w of one or more other oxidisers.
[0072] Suitably the composition further comprises one or more other components selected from the group consisting of sensitisers, fuels, other fuels, water, thickeners, crosslinkers, emulsifiers, energy diluents and optionally other additives as discussed further below.
Hydrogen Peroxide (HP)
[0073] The concentration of HP in the composition of the invention is between about 2% to 25% w/w.
[0074] Suitably the HP concentration in the composition is about 0.5, 0.8, 1, 2, 5, 6, 10, 12, 13, 15, 20 or 25% w/w. For example, the HP concentration in the composition may be between about 0.5, 0.8, 1, 2, 5, 6, 10, 12, 13, 15, or 20 and 25% w/w. For example, the HP concentration in the composition may be greater than 1% but less than 25%, or may be greater than 2% but less than 25%, or may be greater than 5% but less than 25% w/w. Suitably the HP concentration in the composition is thus around between about 1 to 5, 2 to 5, 1 to 10, 5 to 10, 5 to 6, 6 to 10, 5 to 15, 10 to 12, 12 to 13, 13 to 15, 2 to 20, 5 to 20, 15 to 25, 15 to 20 or 2 to 25, 5 to 25, or 20 to 25% w/w. For example, the HP concentration in the composition may be from 2 to 10% w/w, or from 12 to 25% w/w, or from 10 to 20% w/w.
[0075] It will be understood that the % w/w of hydrogen peroxide present in the composition refers the amount of pure hydrogen peroxide. As hydrogen peroxide is provided in the form of an aqueous solution having an HP concentration of less than 100%, for example, having an HP concentration of 50% w/w, or 35% w/w, or 30% w/w, the skilled person will readily understand the need and manner by which they can adjust the amount of diluted HP solution required to ensure the explosive compositions of the invention comprise 2 to 25% w/w HP. To take an example for the avoidance of doubt, if a composition of the invention contains 20% of a 50% w/w solution of HP, the composition contains 10% w/w HP. The skilled person will also appreciate that the 2 to 25% w/w concentration of HP is the final HP concentration in the explosive composition, and thus account must be taken of the diluting effects of any other components (e.g., fuels, oxidisers, thickeners, etc.) added to the composition during formulation.
Other Oxidisers
[0076] The compositions described herein comprise one or more other substances or mixtures of substances which are themselves suitable as explosive materials.
[0077] In particular, the explosive compositions of the invention comprise one or more other oxidisers (e.g. one other oxidiser, e.g. two other oxidisers). Any suitable oxidiser can be used. For example, the one or more other oxidiser(s) are suitably selected from the group consisting of nitrate salts, perchlorate salts, sodium peroxide and potassium peroxide and optionally nitric acid.
[0078] The nitrate salts may be selected from the group consisting of ammonium nitrate, sodium nitrate, calcium ammonium nitrate, calcium nitrate, potassium nitrate, barium nitrate and magnesium nitrate.
[0079] The perchlorate salts may be selected from the group consisting of ammonium perchlorate, sodium perchlorate, potassium perchlorate, barium perchlorate, magnesium perchlorate and calcium perchlorate (e.g. ammonium perchlorate and sodium perchlorate).
[0080] In one embodiment the one or more other oxidiser(s) are selected from the group consisting of nitrate salts and perchlorate salts. In one embodiment the one or more other oxidiser(s) are selected from nitrate salts. In one embodiment the one or more other oxidiser(s) are selected from the group consisting of AN, CAN and SN. In one embodiment the one or more other oxidiser(s) are selected from the group consisting of CAN, CN and SN. In one embodiment the one or more other oxidiser(s) are selected from the group consisting of CAN and SN. In one embodiment the other oxidiser is CAN. In one embodiment the other oxidiser is SN. In one embodiment the other oxidiser is CN. In one embodiment, the one or more other oxidiser(s) do not include AN. In other words, in one embodiment, the explosive composition is devoid of AN.
[0081] The compositions of the invention comprise from greater than 0 and up to about 90% w/w of one or more other oxidisers, such as from about 0.1% to about 75% w/w. For example, compositions of the invention may comprise from greater than 0, from 0.1%, from 1%, from 10%, from 20%, from 30%, from 40%, from 50%, or from 60% w/w up to 90% w/w of one or more other oxidisers, e.g., compositions of the invention may comprise from 1 to 20%, from 20 to 40%, from 15 to 35%, from 35 to 55%, from 30 to 70%, from 40 to 70%, or from 50 to 80% w/w of the one or more other oxidisers. For example, compositions of the invention may comprise up to 90%, 80%, 75%, 70%, 65%, 60%, 50%, 40%, 30%, 20% w/w, etc of one or more other oxidisers, or may comprise about 90%, 80%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 30%, or 20% w/w of one or more other oxidisers. It will be understood that the explosive compositions herein comprise one or more oxidisers according to the foregoing amounts or ranges in total, and as such, where more than one oxidiser is used, each oxidiser may be present in any suitable amount within the foregoing amounts or ranges such that the total mass of the oxidisers adds up to the specified amount or range.
[0082] It will be appreciated that the oxidiser can be in the form of a mixture of solid and liquids. To explain, typically the oxidiser will be solubilised in water when used at a relatively low concentration, and if present at higher concentrations beyond the solubility of the oxidiser, then the oxidiser will be solubilised and in a solid form. In some embodiments, the oxidiser is fully solubilised (or substantially fully solubilised) in the composition. In such embodiments, excess solid oxidiser, e.g., in the form of prills, may be added. In other embodiments, the oxidiser is only partially solubilised in the composition, in which case solid oxidiser (e.g., in the form of solid prills) may be added just prior to detonation such that there is insufficient time for the prills to solubilise substantially. The oxidiser can be in a liquid:solid ratio of between 100:0 to 20:80, and any ratio in between. For example, the liquid:solid ratio may be between 100:0 and 70:30, or between 80:20 and 60:40, or between 70:30 and 40:60, or between 5:50 and 30:70, or of 100:0, 70:30, 60:40, 50:50, 45:55, 40:60; or 20:80.
[0083] It also lies within the invention to have as a further explosive component of the composition well known explosive materials comprising one or more of for example trinitrotoluene, nitroglycerine or pentaerythritol tetranitrate.
Water
[0084] The explosive compositions described herein may comprise water. In one embodiment, the explosive composition may comprise less than 50% w/w of water, or 40% w/w or less or water, or 30% w/w or less of water, for example 25% w/w or less, 20% w/w or less, 15% w/w or less or 10% w/w or less. In one embodiment, the explosive composition may comprise 5% w/w or more of water, for example 10% w/w or more. The composition may thus comprise between 5 and 50% w/w water, or between 5 and 20% w/w water, or between 15 and 30% w/w water, or between 10 and 40% w/w water, or 50, 45, 40, 35, 30, 25, 20, 15, 10, 5 or 1% w/w water.
Sensitisers
[0085] The explosive composition according to the invention may comprise one or more sensitisers dispersed in the composition to produce voids which improve sensitivity to detonation. In addition, HP may itself act as both a sensitiser and an oxidiser. Alternatively, HP itself may act as the sensitiser and no other sensitisers may be used.
[0086] Suitably the explosive composition of the invention is adapted to retain the sensitiser in a substantially homogenous dispersion (e.g. by a thickener or an emulsifier in the case of a watergel or an emulsion, respectively). It will be appreciated that a variety of techniques can be utilised to achieve this property, as discussed further below.
[0087] Suitably a minimum concentration of sensitiser is included into the composition to cause it to be explosive. Suitably the sensitiser is included in a detonation-sensitive concentration or amount. The sensitiser is also suitably maintained in a detonation-sensitive dispersion/distribution throughout the composition. Suitably the final density of the composition is controlled into a preferred pre-determined explosive range. Suitably the final density is controlled with sensitiser to around 0.3 to 1.4 g/cm.sup.3. Suitably the density of the composition is formulated to be around 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 1.1, 1.2, 1.3, or 1.4 g/cm.sup.3. Suitably the final density of the composition is formulated to be between around 0.3 to 0.4, 0.4 to 0.5, 0.5 to 0.6, 0.6 to 0.7, 0.7 to 0.8, 0.8 to 0.9, 0.9 to 1.0, 1.0 to 1.1, 1.1 to 1.2, 1.2 to 1.3, or 1.3 to 1.4 g/cm.sup.3.
[0088] The skilled person will appreciate that a mathematical conversion will be required to convert the weight of ceramic/glass/plastic micro balloons or the amount of chemical to be decomposed into bubbles to yield a certain density, to volume (for gas bubbles). However, irrespective of the type of sensitisation, it will be appreciated that the final density is controlled to a predetermined value to yield an explosive composition and to thereby control the parameters discussed above.
[0089] Once the explosive is sensitised, it can be easily initiated by a primer, which as the skilled person will be aware is an explosive which generates a high detonation pressure which then initiates detonation of the sensitised explosive.
[0090] The introduction of voids into the composition can be provided by a variety of techniques (by entrapping gas bubbles when mixing, by using gas bubbles chemically generated in situ, by injecting gas bubbles, or mixing the composition with gas entrapped material), which are all applicable to the present invention.
[0091] In one embodiment the bubbles may be `trapped` during the preparation of the explosive composition by the mechanical mixing. In U.S. Pat. No. 3,400,026 a formulation which uses protein in solution (albumin, collagen, soy protein, etc.) in order to favour the formation of bubbles and their stabilization is described. U.S. Pat. No. 3,582,411 describes a watergel explosive formulation which contains a foaming agent of the guar gum type modified by hydroxy groups. In U.S. Pat. No. 3,678,140 a process for the incorporation of air by means of the use of protein solution is described, by passing the composition through a series of openings at pressures from 40 to 200 psi and simultaneously introducing air through eductors.
[0092] Wherein in situ generation of gas bubbles is provided by the decomposition of chemicals compounds, the decomposition suitably produces O.sub.2, CO.sub.2, N.sub.2, H.sub.2, NO or combinations thereof.
[0093] Various gases in bubble form have been used to sensitise blasting agents, for example nitrogen (see U.S. Pat. Nos. 3,713,919, 3,886,010, 4,875,951; 4,305,766; and 3,390,030), carbon dioxide (see U.S. Pat. Nos. 3,288,658 and 3,390,032), oxygen (see U.S. Pat. Nos. 3,706,607; 3,790,415, 5,397,399; 4,081,299 and 3,617,401), and hydrogen (see U.S. Pat. No. 3,711,345).
[0094] Suitable chemicals for the in situ generation of gas bubbles include HP itself which can be decomposed with manganese (Mn) salts, yeast, iodide salts, etc; nitrogen-based compounds such as nitrosoamines, such as, for example, N,N'-dinitrosopentamethylenetetramine; boron-based compounds such as, for example, sodium borohydride; carbonates such as, for example, sodium carbonate.
[0095] The discontinuous phase of fine gas bubbles may be incorporated into the compositions of the present invention by injection/bubbling the gas through the composition. It is known to directly inject air or gas into the explosive mixture (see for example U.S. Pat. Nos. 6,537,399; 3,582,411 and 3,642,547).
[0096] Suitable gases for injection include air, oxygen, nitrogen, carbon dioxide, hydrogen, NO and noble gases (such as Argon).
[0097] Alternatively, hollow gas-filled compressible particles such as micro balloons, or porous particles, or expanded polystyrene (EPS) or mixtures thereof are included. In related embodiments the compressible material is any low density material which has a specific gravity <1.0 g/cm.sup.3. In brief summary, examples of glass balloons can be seen in U.S. Pat. Nos. 4,326,900 and 3,447,978, and plastic micro balloons in U.S. Pat. Nos. 4,820,361 and 4,547,234. These balloons are typically 0.05 mm in diameter and have a bulk density of 100 g/L. Use of expanded polystyrene can be seen for example in U.S. Pat. Nos. 5,470,407 and 5,271,779.
[0098] In one embodiment, the compressible material is gas-filled and selected from small hollow microspheres of ceramic, glass or resinous materials or porous materials, and combinations thereof, such as perlite or fly ash.
[0099] Suitably the microspheres/micro balloons contain gas such as pentane, etc. Suitably the microspheres are sized between about 20 to 2000 micron and have a bulk density of less than 1000 g/L.
[0100] In alternative embodiments, the compressible material is a cellular material, such as expanded polystyrene (EPS), polyurethane foam, cotton seeds, expanded pop corn, husks, and combinations thereof.
[0101] Examples of suitable hollow particles include small hollow microspheres of ceramic, glass and resinous materials such as phenol-formaldehyde, poly(vinylidene chloride)/poly(acrylonitrile) copolymers and ureaformaldehyde. Examples of suitable hollow particles include Q-Cel, Envirospheres.RTM., Cenospheres.RTM., Expancel.RTM., 3M, Extendospheres.RTM., etc. Examples of porous materials include expanded minerals such as perlite, fly ash. A further example of a porous material is hollow particles that are a by-product of coal fired power stations.
[0102] Typically, sufficient bubbles and/or hollow particles and/or porous particles are used in the compositions of the present invention to give an explosive composition having a density in the range of from 0.3 to 1.4 g/cm.sup.3.
[0103] For example, an explosive composition of the invention may have a density of up to 1.4 g/cm.sup.3, up to 1.3 g/cm.sup.3, up to 1.2 g/cm.sup.3, up to 1.1 g/cm.sup.3, up to 1.0 g/cm.sup.3, etc. An explosive composition of the invention may have a density of from 0.3 g/cm.sup.3, from 0.4 g/cm.sup.3, from 0.5 g/cm.sup.3, etc. Using conventional mixing techniques to provide bubbles in emulsion explosive compositions often produce bubbles with a range of bubble sizes. For example, the bubbles often have diameters up to 2000 microns and average bubble diameters of less than 300 microns are also common. By choice of suitable surfactants bubbles of smaller or larger diameters can be produced. Thus by choice of an appropriate surfactant at a desired concentration the mean gas bubble diameter in the discontinuous gas phase may be controlled, and bubbles of 50 to 300 microns are possible. For emulsified explosives the density range is suitably around 0.60-1.30 g/cm.sup.3, and for watergels the density range is suitably between 0.2-1.40 g/cm.sup.3. In an emulsified system the gas bubbles are suitably 10-100 times larger than the disperse phase droplets. The oily phase is likely to be in contact the gas bubble, whereas the oxidiser (or discontinuous phase) does not.
[0104] Other types of sensitising materials can be used in the compositions of the invention, e.g. TNT, HMX, RDX, aluminium powder and silicon powder and combinations thereof (e.g. TNT, HMX, RDX and aluminium powder and combinations thereof).
Fuels for Watergels
[0105] The explosive compositions of the invention may comprise one or more fuels.
[0106] HP-based watergels can be prepared with either water-miscible or water immiscible fuels.
[0107] The skilled person will appreciate that there are many options available for use as a fuel. For example, depending on their origin, the fuel may be a product of vegetable origin, such as sugars, molasses, vegetable oils or alcohols. Such fuels may be regarded as sustainable fuels. Other fuels can be sourced from the petrochemical industry, as for example diesel, paraffinic oils or mineral oil, organic acids, ethers, esters, amine nitrates, urea, hexamine, etc. Other fuels may be silicone oils, etc. Suitable fuels for use in the compositions of the invention are glycerol, sugar, syrup, alcohol, carbon, ground coal, waxes, oils such as corn, cottonseed, olive, peanut, or fatty acid oils. Suitable sustainable fuels for use in the compositions of the invention may include, sugar molasses, vegetable oil, alcohol, oils such as corn, cottonseed, olive, peanut, fatty acid oils, or gums. Other fuels may be selected from ethylene glycol, glycerol, propylene glycol, and/or formamide Preferably, the sustainable fuel is glycerol. The composition may comprise between 15 and 25% w/w sustainable fuel, e.g., between 15 and 20%, or between 20 and 25% w/w. The composition may alternatively comprise less than 40% w/w sustainable fuel, less than 30%, less than 25%, or less than 20% w/w sustainable fuel, e.g., 5%, 10%, 15%, 20%, 25%, 30%, 35% or 40% w/w sustainable fuel. Alternatively, the above fuels can also split into water-soluble and water-insoluble fuels. Water-miscible fuels which can be used with the present invention can be selected from the group consisting of: glycerol, sugar, amine nitrates (e.g. mono methyl ammonium nitrate or ethanol amine nitrate), hexamine and urea.
[0108] Water-immiscible fuels which can be used with the present invention can be selected from the group consisting of: include aliphatic, alicyclic and aromatic compounds and mixtures thereof which are in the liquid state at the formulation temperature. Suitable organic fuels may be chosen from fuel oil, diesel oil, distillate, kerosene, naphtha, waxes, (e.g. microcrystalline wax, paraffin wax and slack wax) paraffin oils, benzene, toluene, xylenes, asphaltic materials, polymeric oils such as the low molecular weight polymers of olefins, vegetable oils, animal oils, fish oils, and other mineral, hydrocarbon or fatty oils, and mixtures thereof. Suitable organic fuels are liquid hydrocarbons generally referred to as petroleum distillates such as gasoline, kerosene, fuel oils, paraffin oils and vegetable oils or mixture thereof.
[0109] Typically, the water miscible or water-immiscible fuel of the watergel composition of the present invention comprises from 5 to 30% w/w and suitably 10 to 25% w/w of the total composition. Suitably the fuel is included in a concentration of about 5, 7, 8, 10, 12, 15, 20, 25, 30, 35, 40, 45, or 50% w/w. Suitably the fuel is included in a concentration of between about 5 to 10, 10 to 15, 5 to 20, 15 to 20, 15 to 25, 20 to 25, 25 to 30, 20 to 40, 30 to 35, 35 to 40, 40 to 45, or 45 to 50% w/w.
[0110] In one embodiment, the water-immiscible fuel is included at 7 to 25% w/w of the total composition.
[0111] In one embodiment, the water-miscible fuel is included at 8 to 25% w/w of the total composition.
Fuels for Emulsions
[0112] The explosive compositions of the invention may comprise one or more fuels.
[0113] HP-based emulsions can be prepared with water-immiscible fuels.
[0114] The fuel can be any fuel such as diesel fuel. Alternatively, it can be paraffinic, mineral, olefinic, naphthenic, animal, vegetable, fish and silicone oils. Other types of fuels are benzene, toluene, xylenes, asphaltic materials and the likes. The fuel may be a sustainable fuel. Suitable sustainable fuels for use in emulsions may include vegetable oil, oils such as corn, cottonseed, olive, peanut, or fatty acid oils. The composition may comprise between 15 and 25% w/w sustainable fuel, e.g., between 15 and 20%, or between 20 and 25% w/w. The composition may alternatively comprise less than 40% w/w sustainable fuel, less than 30%, less than 25%, or less than 20% w/w sustainable fuel, e.g., 5%, 10%, 15%, 20%, 25%, 30%, 35% or 40% w/w sustainable fuel.
[0115] The water-immiscible organic phase component of the composition of the present invention comprises the continuous "oil" phase of the water-in-oil emulsion and is the fuel. Suitable organic fuels include aliphatic, alicyclic and aromatic compounds and mixtures thereof which are in the liquid state at the formulation temperature. Suitable organic fuels may be chosen from fuel oil, diesel oil, distillate, kerosene, naphtha, waxes, (e.g. microcrystalline wax, paraffin wax, and slack wax) paraffin oils, benzene, toluene, xylenes, asphaltic materials, polymeric oils such as the low molecular weight polymers of olefins, vegetable oils, animal oils, fish oils, and other mineral, hydrocarbon or fatty oils, and mixtures thereof. Suitable organic fuels are liquid hydrocarbons generally referred to as petroleum distillates such as gasoline, kerosene, fuel oils, paraffin oils and vegetable oils or mixture thereof.
[0116] Typically, the organic fuel or continuous phase of the HP-based emulsion composition of the present invention comprises from 2 to 20% w/w and suitably 3 to 20% w/w of the total composition, e.g. from 7 to 12% w/w of the total composition. Suitably the organic fuel is included in a concentration of about 2, 4, 6, 8, 10, 12, 14, 16, 18, or 20% w/w. Suitably the organic fuel is included in a concentration of between about 2 to 4, 4 to 6, 6 to 8, 8 to 10, 10 to 12, 12 to 14, 14 to 16, 16 to 18, or 18 to 20% w/w.
Secondary Fuels for Watergels and Emulsions
[0117] If desired, other optional fuel materials, hereinafter referred to as secondary fuels, may be incorporated into the compositions of the present invention.
[0118] Examples of such secondary fuels include finely divided solids. Examples of secondary fuels include sulphur, aluminium, gilsonite, comminuted coke or charcoal, carbon black, abietic acid, glucose or dextrose, starch, nut meal, grain meal, wood pulp, etc., and combinations thereof. Examples of secondary fuels also include recycled plastic waste.
[0119] Typically, the optional secondary fuel component of the compositions of the present invention comprise from 0 to 20% w/w of the total composition, e.g. at 0.1 to 12% w/w.
Ratios of Components
[0120] In one embodiment, the explosive composition may comprise a ratio of HP:one or more other oxidisers in the range between 100:1 to 30:70.
[0121] In one embodiment, the explosive composition may comprise a ratio of HP (or HP+one or more oxidisers):fuel in the range between 87:13 to 64:36.
[0122] In one embodiment, the explosive composition may comprise a ratio of HP (or HP+one or more oxidisers):fuel:water in the range between 60:20:20 to 72:24:4.
Thickeners
[0123] The explosive compositions of the invention may comprise one or more thickeners. More particularly, the watergel explosive compositions of the invention may comprise one or more thickeners.
[0124] Because bubbles of gas and materials enclosing gas have a relatively low density, they will tend to migrate towards the surface of the column of explosive if the viscosity of the HP-based explosive composition is not capable of maintaining the sensitising material homogeneously dispersed throughout. Migration of the sensitising material towards the surface is undesirable as it may render the explosive too insensitive to initiation, and therefore the explosive composition may not deliver the energy and gases needed to break and move the rock as required or even worse, the explosive may undergo a misfire. One way to ameliorate this issue is to formulate the explosive composition into a watergel. These types of compositions can be formulated with different levels of viscosity by using a thickener. Viscosities can be selected to generally retain the sensitising material in a homogeneously dispersed state throughout the composition.
[0125] If desired the aqueous solution of the compositions of the present invention may comprise thickeners which optionally may be crosslinked. Any conventional thickener may be used with the present invention. The thickeners, when used in the compositions of the present invention, are suitably polymeric materials, especially gum materials typified by the galactomannan gums such as locust bean gum or xantham gum or guar gum or derivatives thereof such as hydroxypropyl guar gum. The thickener may be selected from gums including natural gums, such guar gum, xanthan gum, sodium alginate, carboxymethylcellullose, methylcellulose and the like. Other useful, but less preferred, gums are the so-called biopolymeric gums such as the heteropolysaccharides prepared by the microbial transformation of carbohydrate material, for example the treatment of glucose with a plant pathogen of the genus Xanthomonas typified by Xanthomonas campestris. Other useful thickeners include synthetic polymeric materials and in particular synthetic polymeric materials which are derived, at least in part, from the monomer acrylamide. An example of a synthetic thickener is polyacrylamide. Inorganic thickeners, such as fumed silica, clays and carbosil, may also be used, or a combination thereof. Suitably the thickener is selected from locust bean gum, guar gum, hydroxypropyl guar gum, sodium alginate and heteropolysaccharides, and combinations thereof.
[0126] Typically, the thickener component of the compositions of the present invention comprises from 0 to 5% w/w of the total composition, e.g. from 0.5 to 5% w/w, e.g. from 0 to 2% w/w of the total composition, e.g. from 0.1 to 2% by weight of the total composition.
Crosslinkers
[0127] Crosslinkers can also be used with the present invention.
[0128] Thickeners in combination with crosslinkers can improve the water resistance and mechanical strength of the explosive. It is convenient for this purpose to use conventional crosslinkers such as zinc chromate or a dichromate either as a separate entity or as a component of a redox system such as, for example, a mixture of potassium dichromate and potassium antimony tartrate. Salts of Ca, Ti, Sb can also be used as crosslinkers. In one embodiment the crosslinker is selected from salts containing zinc, calcium, titanium, antimony, chromium, borate and dichromate and combinations thereof.
[0129] Typically, the crosslinker component of the compositions of the present invention comprises from 0 to 3% w/w, e.g. from 0 to 0.1% w/w of the total composition, e.g. from 0.1 to 1% w/w of the total composition, e.g. from 1 to 2% w/w of the total composition, e.g. from 2 to 3% w/w of the total composition.
Emulsifier/Stabiliser
[0130] The explosive compositions of the invention, when prepared as emulsion form, may comprise one or more emulsifiers.
[0131] HP-based emulsion compositions are made of a discontinuous phase of oxidising material that is dispersed in a continuous phase of an organic fuel in the presence of one or more emulsifiers. The emulsifier is adapted or chosen to maintain phase separation.
[0132] The emulsifier component of the composition of the present invention may be chosen from the wide range of emulsifiers known in the art for the preparation of water-in-oil emulsion explosive compositions. Examples of such emulsifiers include polyisobutylene succinic anhydride (PIBSA) reacted with amines; examples of other emulsifiers are PIB-lactone and its amino derivatives, alcohol alkoxylates, phenol alkoxylates, poly(oxyalkylene) glycols, poly(oxyalkylene) fatty acid esters, amine alkoxylates, fatty acid esters of sorbitol and glycerol, fatty acid salts, sorbitan esters, poly(oxyalkylene) sorbitan esters, fatty amine alkoxylates, poly(oxyalkylene) glycol esters, fatty acid amides, fatty acid amide alkoxylates, fatty amines, quaternary amines, alkyloxazolines, alkenyloxazolines, imidazolines, alkyl-sulfonates, alkylarylsulfonates, alkylsulfosuccinates, alkylphosphates, alkenylphosphates, phosphate esters, lecithin, copolymers of poly(oxyalkylene) glycols and poly(12-hydroxystearic acid), and mixtures thereof.
[0133] Among the preferred emulsifiers are the 2-alkyl- and 2-alkenyl-4,4'-bis (hydroxymethyl) oxazoline, the fatty acid esters of sorbitol, lecithin, copolymers of poly(oxyalkylene) glycols and poly(l2-hydroxystearic acid), and mixtures thereof, and particularly sorbitan mono-oleate, sorbitan sesquioleate, 2-oleyl-4,4'-bis (hydroxymethyl) oxazoline, mixture of sorbitan sesquioleate, lecithin and a copolymer of poly(oxyalkylene glycol and poly (12-hydroxystearic acid), and mixtures thereof.
[0134] Typically, the emulsifier component of the composition of the present invention comprises up to 5% w/w of the total composition. Higher proportions of the emulsifier may be used and may serve as a supplemental fuel for the composition but in general it is not necessary to add more than 5% w/w of emulsifier to achieve the desired effect. One of the advantages of the compositions of the present invention is that stable emulsions can be formed using relatively low levels of emulsifier and for reasons of economy it is preferable to keep the amount of emulsifier used to the minimum required to have the desired effect. A suitable level of emulsifier is in the range from 0.1 to 2.0% w/w of the total composition.
Energy Diluents
[0135] The explosive compositions of the invention may optionally comprise one or more energy diluents.
[0136] In the context of this invention, energy diluting agents are inert materials that have minimal contribution to the detonation process as sensitising means, however they can be used to replace part of the energetic material in the composition and therefore reduce the energy output of the hydrogen peroxide-based explosive.
[0137] In some cases these energy-diluting agents may increase, decrease or not alter the density of the HP-based composition. In some cases, these energy diluting agents are able to reduce the density of the HP-based composition without increasing the sensitivity.
[0138] Examples of these diluents materials are EPS (with particle size larger than 2 mm in diameter), granulated/shredded rubber (from tyres), cotton seeds, saw dust, husk, expanded popcorn, plastic beads, wool meal, bagasse, peanut and oat husks, peanut shells etc. U.S. Pat. No. 5,409,556 describes some examples of these energy reducing agents. In one embodiment the energy diluting agent is selected from granulated/shredded tyres, rubber, expanded rice, expanded popcorn, expanded wheat, and combinations thereof. These materials could also be used in combination with sensitising agents to offer more flexibility (as shown in U.S. Pat. No. 5,470,407) as far as the performance properties of the hydrogen peroxide-based explosive is concerned. Therefore, another advantage of the HP-based explosive is that the performance properties of the explosive can be altered to suit the characteristics of the blasting site.
[0139] Watergel or water-in-oil HP-based explosive compositions made according to the present invention include energy-diluting agents in concentration between 0-800% by volume. As a result, the use of the additives (sensitising agent and energy-diluting agents), provides a better control of the density, VOD and energy delivery in the ground being blasted. Therefore, an additional advantage of the HP-based explosive is that it could be used in a range of density between 0.3 g/cm.sup.3 to 1.4 g/cm.sup.3.
Density of the Explosive Compositions
[0140] Suitably the final density is controlled with sensitiser to around 0.3 to 1.4 g/cm.sup.3. Suitably the density of the composition is formulated to be around 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 1.1, 1.2, 1.3, or 1.4 g/cm.sup.3. Suitably the final density of the composition is formulated to be between around 0.3 to 0.4, 0.4 to 0.5, 0.5 to 0.6, 0.6 to 0.7, 0.7 to 0.8, 0.8 to 0.9, 0.9 to 1.0, 1.0 to 1.1, 1.1 to 1.2, 1.2 to 1.3, or 1.3 to 1.4 g/cm.sup.3. In some embodiments the density is controlled to a predetermined target value by selection of the ratios of the components of the composition. For example, by balancing the concentration of a component which reduces the density, such as hollow microspheres, and one that has a relatively high density, such as nitrate prills.
pH of the Explosive Compositions
[0141] The pH of the emulsion explosive compositions of the present invention is not narrowly critical. However, in general the pH is between 0 and 8 and suitably the pH is between 1 and 6, and may be controlled by suitable addition of conventional additives, for example inorganic or organic acids and salts.
Viscosity of the Explosive Compositions
[0142] The viscosity of the explosive compositions (watergel or emulsion type) will be discussed in terms of apparent viscosity. Where used herein the term "apparent viscosity" refers to viscosity measure using a Brookfield RVT viscometer, #7 spindle at 50 r.p.m.
[0143] It is preferred in the process of the present invention that the explosive composition of the water-in-oil emulsion explosive particles have an apparent viscosity greater than 10 Pa*s (Pascal*second) prior to the entrainment of gas bubbles. Apparent viscosity is more suitably in the range 5 to 50 Pa*s. A more preferred viscosity range for the entrainment of gas bubbles by mechanical mixing is from 10 to 35 Pa*s. The range 10 to 25 Pa*s provides the most efficient entrainment of gas bubbles by mechanical mixing.
[0144] Suitably the explosive composition of the invention can be easily pumped.
Oxygen Balance of the Explosive Compositions
[0145] "Oxygen balance" (OB) is a term of the art which is used to indicate the degree to which an explosive can be oxidised. An OB close to zero is preferred when formulating mining explosives, such that no reactant is in excess during the detonation process, and therefore the expected products are nitrogen, water and carbon dioxide. If the oxygen balance is far from zero, some part of the reactant materials will not react and instead, those unreacted material absorb/sink heat from the detonation reaction, which in turn will cause the explosive to underperform. For example, some prior art compositions are unsuitable for combustion, as they lack fuel (and therefore the OB is too positive) and the composition cannot burn.
[0146] Suitably the amount of fuels materials in the explosive composition can be adjusted so the composition has a final oxygen balance between +10 and -10, e.g. between +5 and -5.
Preparation of Explosive Compositions
[0147] According to a second aspect the present invention provides a method of preparing an explosive composition according to the first aspect, the method comprising combining hydrogen peroxide and one or more other oxidisers.
[0148] The explosive compositions of the present invention may be prepared by a number of methods. Possible variations of this general procedure will be evident to those skilled in the art of the preparation of emulsion explosive compositions.
[0149] The method may comprise the step of formulating said composition as a watergel or an emulsion.
[0150] The method may further comprise the step of adding a water-immiscible or water-miscible fuel.
[0151] The method may further comprise the step of adding a thickener or an emulsifier.
[0152] The method may further comprise the step of adding a secondary fuel.
[0153] The method may further comprise the step of adding an energy diluting agent.
[0154] The method may further include a sensitisation step.
[0155] The method may further comprise the step of adding a crosslinker.
[0156] In one suitable method of manufacture the explosive watergel type compositions may be prepared by: [0157] making an oxidiser phase by combining hydrogen peroxide with one or more other oxidisers and one or more water soluble fuels, and then [0158] making a fuel phase by adding one or more water-miscible fuels (e.g. glycerol) and one or more thickeners (e.g. gum such as guar gum) and mixing (e.g. stirring) to suspend the thickener; [0159] mixing both the fuel phase and the oxidiser phase; [0160] optionally one or more other oxidisers in a solid form (which are the same as or different from the one or more other oxidisers of the oxidiser phase) can be added, as a powder or prills, and mixing; [0161] adding and mixing secondary fuels; [0162] adding and mixing energy diluting agents; [0163] sensitising agents are mixed into said composition in an amount capable to sensitise said composition (e.g. after 2 to 5 minutes of mixing, a gassing agent is added); [0164] crosslinking agents are mixed to convert the composition into a gel.
[0165] In one suitable method of manufacture the explosive emulsion type compositions may be prepared by: [0166] making an oxidiser phase by dissolving one or more oxidisers with hydrogen peroxide; and then [0167] making a fuel phase by adding one or more water insoluble fuels (e.g. diesel) and one or more emulsifiers (e.g. PIBSA-based or SMO) and mixing (e.g. stirring) to suspend the thickener; [0168] adding the fuel phase to the oxidiser phase whilst mixing; [0169] optionally one or more other oxidisers in a solid form (which are the same as or different from the one or more other oxidisers of the oxidiser phase) can be added, as a powder or prills, and mixing; and [0170] sensitising agents are mixed into said composition in an amount capable to sensitise said emulsion.
Use of the Compositions
[0171] It will be appreciated that the composition of the invention can be used for many purposes, but in particular to break and move ground in mining operations.
[0172] Thus according to a third aspect the present invention provides use of an explosive composition according to the first aspect to break and move ground, e.g. in mining operations.
[0173] Suitably the explosive composition is introduced (e.g. by pumping) into a blasthole that already has a detonator placed therein. Detonation of blastholes in a mining operation is well known to the person skilled in the art.
Preferred Embodiment of the Invention
[0174] Numerous embodiments are described in this patent application, and are presented for illustrative purposes only. The described embodiments are not intended to be limiting in any sense. The invention is widely applicable to numerous embodiments, as is readily apparent from the disclosure herein.
[0175] In one embodiment, the present invention comprises or consists essentially of hydrogen peroxide, one or more other oxidisers, wherein at least some of at least one of the one or more other oxidisers may be present in the composition in solid form (e.g., powder, prills) a fuel, a thickener, a sensitiser, a crosslinker, a secondary fuel and an energy diluting agent. In other embodiments the present invention comprises or consists essentially of hydrogen peroxide, one or more other oxidisers, wherein at least some of at least one of the one or more other oxidisers is present in the composition in solid form (e.g., powder, prills), a fuel, a surfactant/emulsifier, a sensitiser, a secondary fuel and an energy diluting agent. In such embodiments, the final oxygen balance of the mixture is preferably between -5 to +5.
[0176] The present invention relates to a peroxide-nitrate/fuel-based explosive composition that is suitably prepared as watergel or water-in-oil emulsion, which may or may not contain solid oxidiser and is sensitised. Typical components for each type of explosive technology are listed in Table 1.
TABLE-US-00001 TABLE 1 Typical components of the present invention for each type of explosive technology. Explosive technology Component Watergel Water-in-oil emulsion Oxidiser(s) hydrogen peroxide and one or hydrogen peroxide and one or more other oxidisers selected more other oxidisers selected from the group consisting of from the group consisting of nitrate salts and/or perchlorate nitrate salts and/or perchlorate salts and/or sodium/potassium salts and/or sodium/potassium peroxide and/or nitric acid. peroxide and/or nitric acid. Optionally water miscible fuels Optionally water miscible fuels can be added to this phase can be added to this phase (amine nitrates, urea, etc) (amine nitrates, urea, etc) Sensitiser gas bubbles (chemically gas bubbles (chemically generated or injected bubbles) generated or injected bubbles) and/or gas entrapped and/or gas entrapped compressible materials compressible materials Fuel Water miscible fuels, water Water miscible fuels, water soluble fuels or water-insoluble fuels immiscible fuels, water soluble fuels or water-insoluble fuels Stabilisers thickeners emulsifiers Additives crosslinkers, catalysts for catalysts for gassing, pH adjusters gassing, pH adjusters Energy Granulated/shredded rubber, Granulated/shredded rubber, diluents expanded popcorn, expanded expanded popcorn, expanded (optional) rice, plastic beads, EPS >5 mm rice, plastic beads, EPS >5 mm
Error! Reference source not found. lists the components of explosive systems discussed herein and provides typical ranges for each.
TABLE-US-00002 TABLE 2 Components for explosive systems discussed herein with typical ranges for each Component (in % by weight of total composition except where Explosive technology indicated otherwise) Watergel Emulsion HP From about 2 to 25 From about 2 to 25 One or more other From greater From greater oxidisers than 0 to 90 than 0 to 90 Sensitiser (% by volume) From 1 to 800* From 1 to 800* Fuels From 8 to 25 From 7 to 25 Secondary fuels From 0.1 to 11 From 0.1 to 11 Water From 5 to 20 From 5 to 20 Thickeners From 0.5 to 5 N/A Emulsifiers N/A From 0.5 to 5 Additives 0.1 to 5 0.1 to 5 Energy diluting agents (% From 1 to 300** From 1 to 300** by volume) Oxygen Balance From 5 to -5 From 5 to -5 Final densities (g/cm.sup.3) 0.30 to 1.40 0.30 to 1.40 NOTE: it will be appreciated that the volume can be increased by 8x(*), and 3x(**), respectively.
Examples
[0177] The present invention can be used for a variety of forms of explosives provided of course that the principles of the invention as described herein are observed. The invention is further illustrated with reference to the following examples detailed in Table 3.
TABLE-US-00003 TABLE 3 Examples of the invention Composition (%-w/w) 1 2 3 4 HP-CAN/ HP-CAN/Fuel- HP- HP-SN/Fuel- Fuel- based gel blend + SN/Fuel- based gel blend + Chemical based gel CAN prills based gel SN prills HP (50% 39.50* 10.8** 55.3*** 32.34**** w/w) CAN 39.50 65.8 SN 23.7 43.86 Glycerol 19.32 22.7 18.9 21.42 Xanthan 1.68 0.7 2.1 2.38 gum *39.5 .times. (50/100)% = 19.75 w/w % HP **10.8 .times. (50/100)% = 5.4 w/w % HP ***55.3 .times. (50/100)% = 27.65 w/w % HP ****32.34 .times. (50/100)% = 16.17 w/w % HP
Detonation Testing
[0178] Selected compositions were tested to determine detonation. PVC pipes 50 mm in diameter by 600 mm in length and sealed on one end were used for gels. PVC pipes 102 mm in diameter by 600 mm in length, sealed on one end were used for composition made of gel and prills.
[0179] VOD was measured using a Time Domain Reflectometry (TRD)-based VOD instrument.
[0180] The examples 1-4 above in Table 3 (corresponding to FIGS. 1-4, respectively) show that combination of HP and nitrates are able to be detonated over a range of densities and providing a range of velocities of detonation.
[0181] From the Figures, it can be seen that explosives compositions, such as HP-CAN/fuel based gel blend and CAN prills, as described herein are able to be detonated with low concentrations of HP, e.g., 5.4% w/w, or lower. The examples also contain a relatively large amount of nitrates in the composition. There is no evidence in the previous art that high levels of CAN or SN can be used for water-based explosive compositions at different densities.
[0182] Although the invention has been described with reference to specific examples, it will be appreciated by those skilled in the art that the invention may be embodied in many other forms. In particular features of any one of the various described examples may be provided in any combination in any of the other described examples.
[0183] The skilled addressee will understand that the invention comprises the embodiments and features disclosed herein as well as all combinations and/or permutations of the disclosed embodiments and features.
* * * * *
D00000

D00001

D00002

D00003

D00004

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.