PROCESSES FOR PREPARING MORPHINAN-6-ONE PRODUCTS WITH LOW LEVELS OF a, -UNSATURATED KETONE COMPOUNDS
Buehler; Henry J. ; et al.
U.S. patent application number 16/549718 was filed with the patent office on 2019-12-19 for processes for preparing morphinan-6-one products with low levels of a, -unsaturated ketone compounds. This patent application is currently assigned to SpecGx LLC. The applicant listed for this patent is SpecGx LLC. Invention is credited to Dennis C. Aubuchon, Henry J. Buehler, William E. Dummitt, Hong Gu, Anthony Mannino.
| Application Number | 20190381033 16/549718 |
| Document ID | / |
| Family ID | 38326629 |
| Filed Date | 2019-12-19 |
View All Diagrams
| United States Patent Application | 20190381033 |
| Kind Code | A1 |
| Buehler; Henry J. ; et al. | December 19, 2019 |
PROCESSES FOR PREPARING MORPHINAN-6-ONE PRODUCTS WITH LOW LEVELS OF a, -UNSATURATED KETONE COMPOUNDS
Abstract
The present invention generally relates to processes for preparing highly pure morphinan-6-one products. The processes involve reducing the concentration of alpha, beta unsaturated ketone compounds present as impurities in morphinan 6 one products or reaction mixtures including morphinan 6 one compounds by treatment with a sulfur-containing compound.
| Inventors: | Buehler; Henry J.; (St. Louis, MO) ; Dummitt; William E.; (St. Louis, MO) ; Mannino; Anthony; (Maryland Heights, MO) ; Aubuchon; Dennis C.; (Arnold, MO) ; Gu; Hong; (Oak Park, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | SpecGx LLC Webster Groves MO |
||||||||||
| Family ID: | 38326629 | ||||||||||
| Appl. No.: | 16/549718 | ||||||||||
| Filed: | August 23, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15710990 | Sep 21, 2017 | |||
| 16549718 | ||||
| 15144010 | May 2, 2016 | |||
| 15710990 | ||||
| 14504750 | Oct 2, 2014 | |||
| 15144010 | ||||
| 11915606 | Nov 27, 2007 | 8871779 | ||
| PCT/US2007/005256 | Mar 2, 2007 | |||
| 14504750 | ||||
| 60778258 | Mar 2, 2006 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 489/08 20130101; A61K 31/485 20130101 |
| International Class: | A61K 31/485 20060101 A61K031/485; C07D 489/08 20060101 C07D489/08 |
Claims
1.-50. (canceled)
51. A composition comprising oxymorphone and containing about 0.001% or less, by weight of said oxymorphone, of an .alpha.,.beta. unsaturated ketone.
52. The composition of claim 51 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone or 14-hydroxycodeinone.
53. The composition of claim 51 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone.
54. The composition of claim 51 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone or 14-hydroxycodeinone and no 14-hydroxymorphinone or 14-hydroxycodeinone is detectable by high performance liquid chromatography with mass spectrometry detection.
55. The composition of claim 51 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone and said composition comprises about 0.0005% or less, by weight of said oxymorphone, of 14-hydroxymorphinone.
56. The composition of claim 51 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone or 14-hydroxycodeinone and said composition comprises about 0.0005% or less, by weight of said oxymorphone, of 14-hydroxymorphinone or 14-hydroxycodeinone.
57. The composition of claim 51, wherein the oxymorphone is an oxymorphone salt.
58. The composition of claim 53, wherein the oxymorphone is an oxymorphone salt.
59. The composition of claim 55, wherein the oxymorphone is an oxymorphone salt.
60. The composition of claim 51, wherein the oxymorphone is oxymorphone HCl.
61. The composition of claim 53, wherein the oxymorphone is oxymorphone HCl.
62. The composition of claim 55, wherein the oxymorphone is oxymorphone HCl.
63. A pharmaceutical formulation comprising oxymorphone and containing about 0.001% or less, by weight of said oxymorphone, of an .alpha.,.beta. unsaturated ketone.
64. The pharmaceutical composition of claim 63 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone or 14-hydroxycodeinone.
65. The pharmaceutical composition of claim 63 wherein said .alpha.,.beta. unsaturated ketone is 14-hydroxymorphinone.
66. The pharmaceutical composition of claim 63 comprising about 0.0005% or less, by weight of said oxymorphone, of an .alpha.,.beta. unsaturated ketone.
67. The pharmaceutical composition of claim 63 comprising about 0.0005% or less, by weight of said oxymorphone, of 14-hydroxymorphinone.
68. The pharmaceutical composition of claim 63 wherein said oxymorphone is oxymorphone HCl.
69. The pharmaceutical composition of claim 65 wherein said oxymorphone is oxymorphone HCl.
70. The pharmaceutical composition of claim 67 wherein said oxymorphone is oxymorphone HCl.
Description
FIELD OF THE INVENTION
[0001] The present invention generally relates to processes for preparing morphinan-6-one products. The processes involve reducing the concentration of .alpha.,.beta.-unsaturated ketone compounds from reaction mixtures including morphinan-6-one compounds.
BACKGROUND OF THE INVENTION
[0002] The morphinan alkaloids represent a family of structurally-related products of great medicinal importance. Particular morphinan compounds of pharmaceutical relevance include, for example, codeine, hydrocodone, hydromorphone, morphine, nalbuphine, nalmefene, naloxone, naltrexone, oxycodone, and oxymorphone. Generally, these compounds are analgesics, which are used extensively for pain relief in the field of medicine due to their action as opiate receptor agonists. However, nalmefene, naloxone, naltrexone, and naltrexone methyl bromide are opiate receptor antagonists, and are used for reversal of narcotic/respiratory depression due to opiate receptor agonists, as addiction therapies, and to reverse other undesirable side effects of opiate agonist use, such as severe constipation.
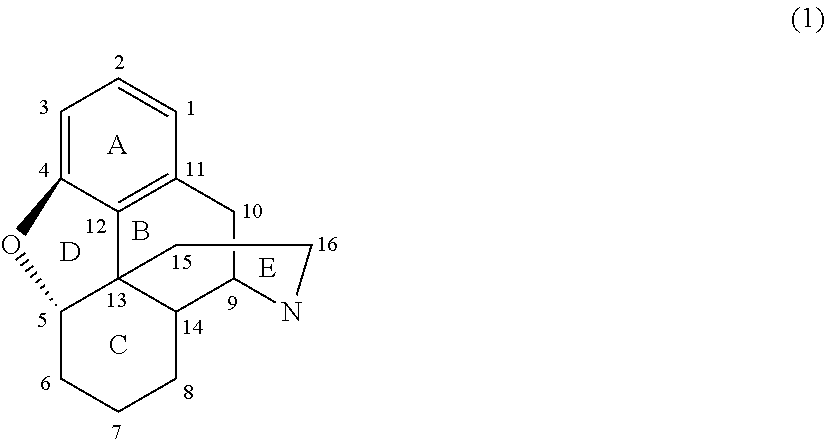
[0003] Morphinan compounds and analogs thereof typically have a ring structure generally corresponding to Formula (1):
##STR00001##
[0004] Various methods are known for the synthesis of morphinan compounds corresponding to Formula (1). Conventional methods used in the commercial production of morphinan compounds typically involve the extraction of opium alkaloids from poppies (Papaver somniferum). Generally speaking, these processes involve the extraction of the alkaloids from opium in a liquid, precipitation of the alkaloids, separation of the raw alkaloids (e.g., morphine and secondary alkaloids such as papaverine, codeine, and thebaine), and purification of the various alkaloids, optionally followed by semi-synthesis steps to produce particular morphinan compounds. See, for example, Barbier, A., "The Extraction of Opium, Twenty-five years of commercial experience in the treatment of opium," Ann. Pharm. Franc., 1947, 5, 121-40; Barbier, A., "The Extraction of Opium Alkaloids," Bull. Narcotics, 1950, vol. 3, 22-29; Neumann, W, "The Manufacture of Alkaloids from Opium," Bull. Narcotics, 1957, vol. 2, 34-40; Lednicer and Mitscher, Organic Chemistry of Drug Synthesis, chapter 15, (Wiley 1977); French Patent No. 1,000,543 to Penau et al.; British Patent No. 713,689 to Wood et al.; and U.S. Pat. No. 2,009,181 to Kabay.
[0005] Synthetic methods for producing various morphinan compounds are also known. These methods commonly utilize 3-methoxy-phenylethylamine as a starting material and include a Grewe cyclization step. For example, in U.S. Pat. No. 4,368,326, Rice discloses a process for preparing a nordihydrothebainone (e.g., 1-bromo-N-formylnordihydrothebainone) from a .beta.,.gamma.-hexahydroisoquinolone (e.g., 1-(2'-bromo-4'-methoxy-5'-hydroxybenzyl)-2formyl-1,3,4,5,7,8-hexahydroqui- nolin-6-one) by Grewe cyclization catalyzed using a super acid catalyst alone or with a combination of an ammonium fluoride complex and trifluoromethanesulfonic acid.
[0006] Many pharmaceutically desirable morphinan compounds and analogs thereof have a ketone group on the C-ring of Formula (1) and a saturated bond between the two carbon atoms positioned .alpha. and .beta. to the ketone on the C-ring of Formula (1). According to the common nomenclature, the ketone is present on the C(6) carbon atom, with the .alpha. and .beta. carbon atoms being the C(7) and C(8) positions (see, e.g., Formula (1)). Thus, these compounds may be referred to as morphinan-6-one compounds. Various processes for producing morphinan-6-one compounds are known, many of which involve some form of catalytic hydrogenation of .alpha.,.beta.-unsaturated ketone intermediate compounds at particular points in the process. Commonly used catalysts include, for example, palladium and platinum. For example, in U.S. Pat. No. 6,177,567 to Chiu et al., 14-hydroxycodeinone (an .alpha.,.beta.-unsaturated ketone compound) is converted to oxycodone by hydrogenating the .alpha.,.beta.-unsaturation using conventional methods such as reduction by diphenylsilane and Pd(Ph.sub.3P)/ZnCl.sub.2, or with sodium hypophosphite in conjunction with a Pd/C catalyst in aqueous acetic acid, or by Pd/C catalytic transfer hydrogenation.
[0007] While these and other methods of reducing or removing the .alpha.,.beta.-unsaturation are generally effective, .alpha.,.beta.-unsaturated ketone compounds may persist as impurities in the final products of desirably .alpha.,.beta.-saturated morphinan-6-one products, such as oxycodone. Additionally, known hydrogenation methods may tend to undesirably reduce the ketone as well as reducing or removing the .alpha.,.beta.-unsaturation. Further, these and other hydrogenation methods are not normally capable of efficiently and economically reducing the levels of 7,8-unsaturation to below 10 to 100 parts per million, or less.
[0008] Some .alpha.,.beta.-unsaturated ketone compounds show mutagenic activity in certain tests. Therefore, a need persists for processes for preparing highly pure morphinan-6-one products having a relatively low concentration of .alpha.,.beta.-unsaturated ketone compounds present as impurities therein.
SUMMARY OF THE INVENTION
[0009] Among the various aspects of the present invention is the provision of a process for the preparation of morphinan-6-one products. The process involves reducing the concentration of .alpha.,.beta.-unsaturated ketone compounds which are present as impurities in reaction mixtures including morphinan-6-one compounds. The process generally involves forming a reaction mixture including a morphinan-6-one compound and an .alpha.,.beta.-unsaturated ketone compound and treating the reaction mixture with a sulfur-containing compound. In one embodiment, the sulfur-containing compound is a sulfur-containing inorganic acid or salt thereof.
[0010] Briefly, therefore, the present invention is directed to a process for the preparation of a morphinan-6-one product, the process comprising:
[0011] forming a reaction mixture comprising a morphinan-6-one compound and an .alpha.,.beta.-unsaturated ketone compound;
[0012] treating the reaction mixture with a sulfur-containing compound to reduce the concentration of the .alpha.,.beta.-unsaturated ketone compound in the reaction mixture; and
[0013] recovering the morphinan-6-one compound to produce the morphinan-6-one product;
wherein
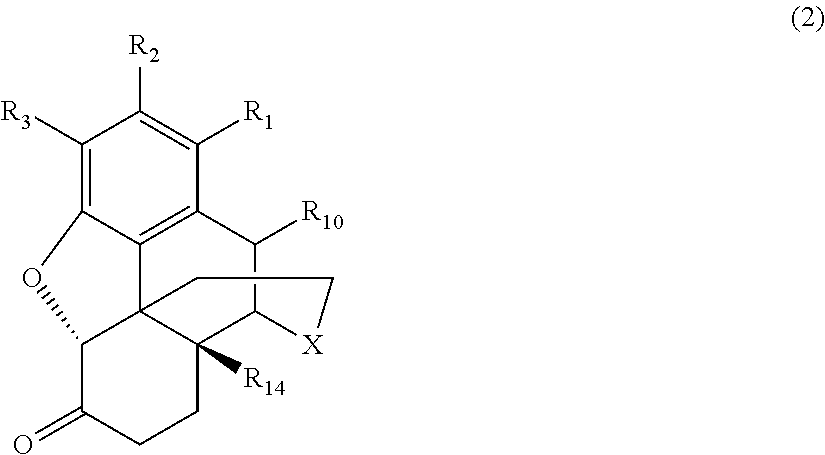
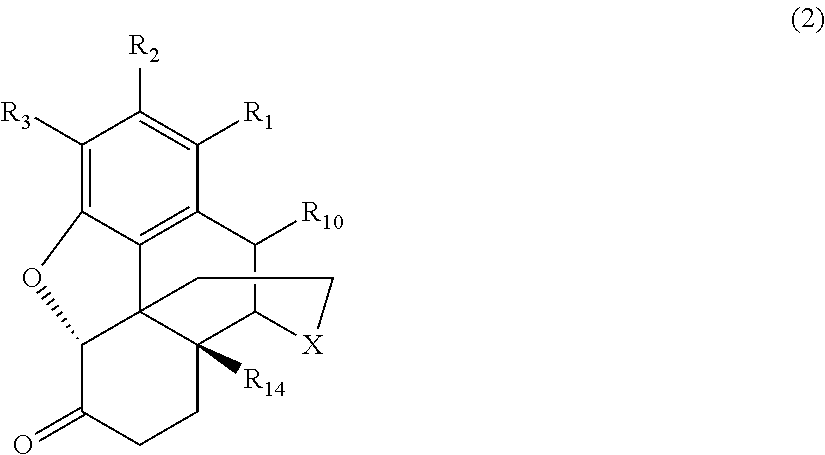
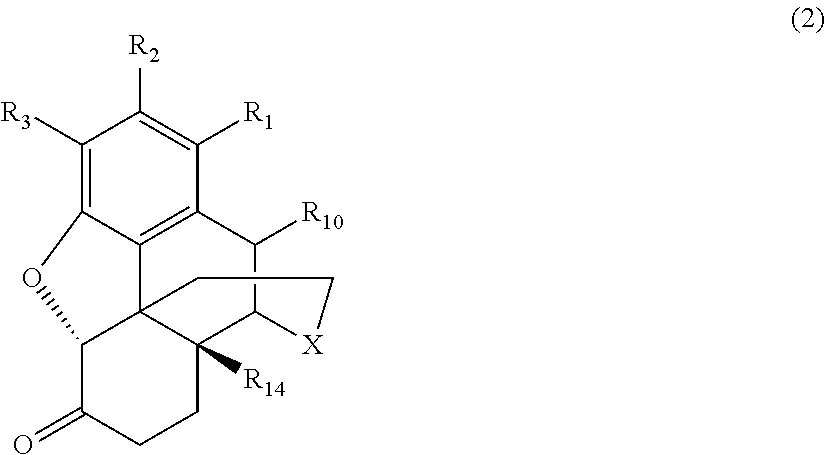
[0014] the morphinan-6-one compound corresponds to Formula (2):
##STR00002##
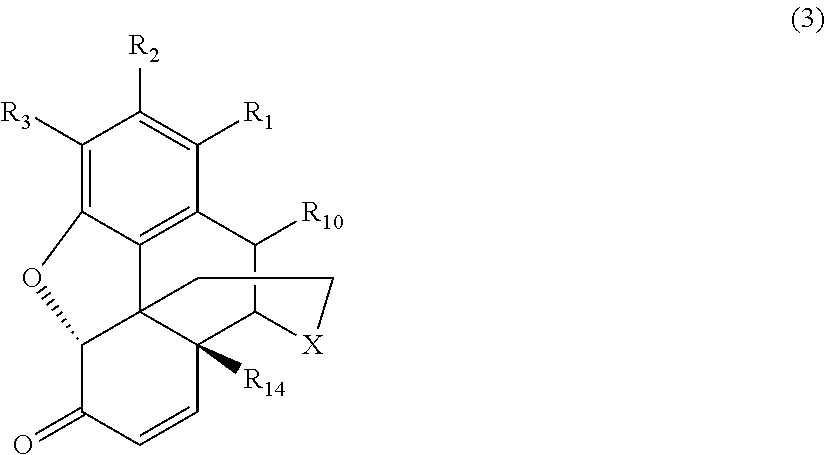
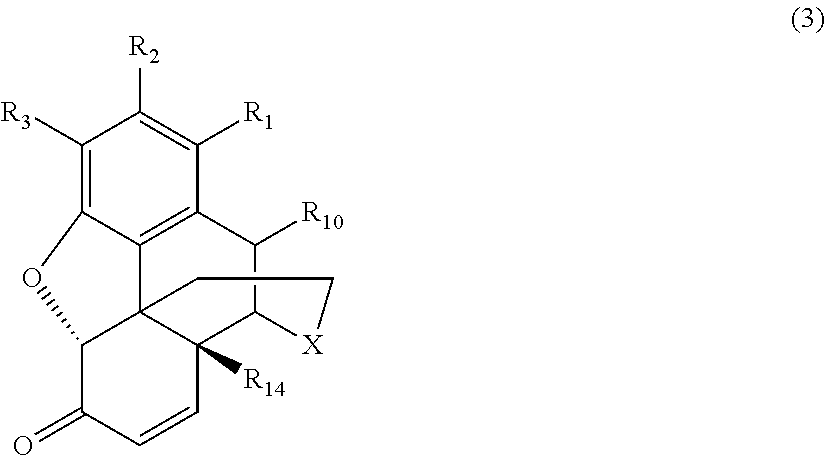
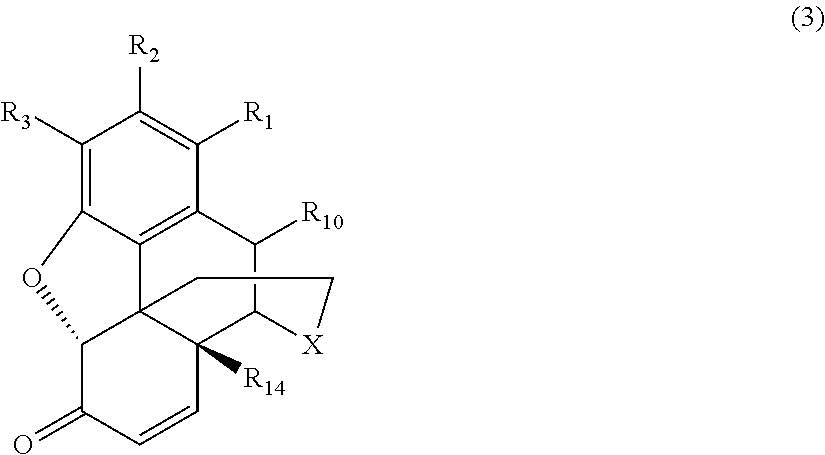
[0015] the .alpha.,.beta.-unsaturated ketone compound corresponds to Formula (3):
##STR00003##
[0016] X is --N(R.sub.17)-- or --N.sup.+(R.sub.17aR.sub.17b)--;
[0017] R.sub.1 and R.sub.2 are independently selected from hydrogen, substituted and unsubstituted acyl, acyloxy, alkenyl, alkoxy, alkoxyaryl, alkyl, alkylamino, alkylthio, alkynyl, amino, aryl, arylalkoxy, carboalkoxy, carbonyl, carboxyalkenyl, carboxyalkyl, carboxyl, cyano, cyanoalkyl, cycloalkyl, cycloalkylalkyl, cycloalkylether, halo, haloalkoxy, haloalkyl, heteroaryl, heterocyclic, hydroxyalkyl, hydroxyl, or nitro;
[0018] R.sub.3 is hydrogen, hydroxy, protected hydroxy, alkoxy, or acyloxy;
[0019] R.sub.10 is hydrogen, hydroxy, protected hydroxy, halo, keto, tosyl, mesyl, or trifluoromesyl;
[0020] R.sub.14 is hydrogen, hydroxy, or protected hydroxy;
[0021] R.sub.17 is hydrogen, alkyl, cycloalkyl, alkylcarboxy, alkylenecycloalkyl, alkoxycarbonyl, allyl, alkenyl, acyl, aryl, formyl, formyl ester, formamide, benzyl, or an amino protecting group; and
[0022] R.sub.17a and R.sub.17b are independently selected from hydrogen, alkyl, alkenyl, allyl, cycloalkyl, aryl, or benzylyl, and
[0023] the morphinan-6-one compound comprises less than about 0.1% (by weight) morphinan-6-one product of the .alpha.,.beta.-unsaturated ketone compound.
[0024] The present invention is also directed to a process for preparing a morphinan-6-one product, the process comprising:
[0025] forming a reaction mixture comprising an .alpha.,.beta.-unsaturated ketone compound;
[0026] treating the reaction mixture with a sulfur-containing compound to reduce the .alpha.,.beta.-unsaturated ketone compound to form a morphinan-6-one compound; and
[0027] recovering the morphinan-6-one compound to form the morphinan-6-one product,
wherein
[0028] the morphinan-6-one compound corresponds to Formula (2):
##STR00004##
[0029] the .alpha.,.beta.-unsaturated ketone compound corresponds to Formula (3):
##STR00005##
[0030] X is --N(R.sub.17)-- or --N.sup.+(R.sub.17aR.sub.17b)--;
[0031] R.sub.1 and R.sub.2 are independently selected from hydrogen, substituted and unsubstituted acyl, acyloxy, alkenyl, alkoxy, alkoxyaryl, alkyl, alkylamino, alkylthio, alkynyl, amino, aryl, arylalkoxy, carboalkoxy, carbonyl, carboxyalkenyl, carboxyalkyl, carboxyl, cyano, cyanoalkyl, cycloalkyl, cycloalkylalkyl, cycloalkylether, halo, haloalkoxy, haloalkyl, heteroaryl, heterocyclic, hydroxyalkyl, hydroxyl, or nitro;
[0032] R.sub.3 is hydrogen, hydroxy, protected hydroxy, alkoxy, or acyloxy;
[0033] R.sub.10 is hydrogen, hydroxy, protected hydroxy, halo, keto, tosyl, mesyl, or trifluoromesyl;
[0034] R.sub.14 is hydrogen, hydroxy, or protected hydroxy;
[0035] R.sub.17 is hydrogen, alkyl, cycloalkyl, alkylcarboxy, alkylenecycloalkyl, alkoxycarbonyl, allyl, alkenyl, acyl, aryl, formyl, formyl ester, formamide, benzyl, or an amino protecting group; and
[0036] R.sub.17a and R.sub.17b are independently selected from hydrogen, alkyl, alkenyl, allyl, cycloalkyl, aryl, or benzylyl.
[0037] Other objects and features will be in part apparent and in part pointed out hereinafter.
DETAILED DESCRIPTION OF THE INVENTION
[0038] The present invention is generally directed to processes for preparing highly pure morphinan-6-one products. The processes generally involve treating a reaction mixture including a morphinan-6-one compound and an .alpha.,.beta.-unsaturated ketone compound with a sulfur-containing compound. Advantageously, the process effectively reduces the concentration of undesirable .alpha.,.beta.-unsaturated ketone compounds to acceptable levels without removing or otherwise affecting other more desirable compounds or substituent groups or unsaturation thereon. Moreover, the sulfur-containing compound may be utilized to reduce the concentration of .alpha.,.beta.-unsaturated ketone compounds present in the reaction mixture from levels of about 0.5% (by weight) or more to levels of not more than about 0.1% (by weight), or lower (e.g., about 0.01% (by weight), about 0.001% (by weight), or lower), with minimal side reactions, ketone reduction, and/or any other undesirable effects.
Morphinan Products and Processes for Preparing the Same
[0039] Generally speaking, the morphinan-6-one products of interest in the process of the present invention include morphinan compounds having a keto group at the C(6) carbon atom on the C-ring and a saturated bond between the C(7) and C(8) carbon atoms on the C-ring (i.e., morphinan-6-one compounds). More specifically, the morphinan-6-one compounds are opiate receptor agonists or antagonists generally corresponding to Formula (2):
##STR00006##
wherein
[0040] X is --N(R.sub.17)-- or --N.sup.+(R.sub.17aR.sub.17b)--;
[0041] R.sub.1 and R.sub.2 are independently selected from hydrogen, substituted and unsubstituted acyl, acyloxy, alkenyl, alkoxy, alkoxyaryl, alkyl, alkylamino, alkylthio, alkynyl, amino, aryl, arylalkoxy, carboalkoxy, carbonyl, carboxyalkenyl, carboxyalkyl, carboxyl, cyano, cyanoalkyl, cycloalkyl, cycloalkylalkyl, cycloalkylether, halo, haloalkoxy, haloalkyl, heteroaryl, heterocyclic, hydroxyalkyl, hydroxyl, or nitro;
[0042] R.sub.3 is hydrogen, hydroxy, protected hydroxy, alkoxy, or acyloxy;
[0043] R.sub.10 is hydrogen, hydroxy, protected hydroxy, halo, keto, tosyl, mesyl, or trifluoromesyl;
[0044] R.sub.14 is hydrogen, hydroxy, or protected hydroxy;
[0045] R.sub.17 is hydrogen, alkyl, cycloalkyl, alkylcarboxy, alkylenecycloalkyl, alkoxycarbonyl, allyl, alkenyl, acyl, aryl, formyl, formyl ester, formamide, benzyl, or an amino protecting group; and
[0046] R.sub.17a and R.sub.17b are independently selected from hydrogen, alkyl, alkenyl, allyl, cycloalkyl, aryl, or benzyl.
[0047] When R.sub.17 is hydrogen, alkyl, alkenyl, cycloalkyl, aryl, or benzyl, salts of the secondary or tertiary amine can be formed wherein the anion is chloride, bromide, acetate, formate, sulfate, bisulfate, bisulfite, oxalate, citrate, malate, tartrate, triflate, trifluoroacetate, methane sulfonate, and the like. When X is --N.sup.+(R.sub.17aR.sub.17b)--, the counter-ion can be chloride, bromide, iodide, trifluoroacetate, trifluoromethanesulfonate, methane sulfonate, acetate, p-toluenesulfonate, sulfate, bisulfate, bisulfite, phosphate, hydrogen phosphate, dihydrogen phosphate, fumarate, oxalate, formate, tartrate, benzoate, and the like.
[0048] In one preferred embodiment, R.sub.14 is hydroxy or protected hydroxy. In another preferred embodiment, R.sub.14 is hydrogen.
[0049] In either of the embodiments described above (i.e., when R.sub.14 is hydroxy or protected hydroxy or R.sub.14 is hydrogen), R.sub.3 is either alkoxy, hydroxy, or protected hydroxy. In one particular embodiment, R.sub.3 is methoxy.
[0050] In any one of the embodiments described above, X is --N(R.sub.17)-- or --N.sup.+(R.sub.17aR.sub.17b)--, wherein R.sub.17, R.sub.17a, and R.sub.17b are defined as above. Where X is --N(R.sub.17)--, in one particularly preferred embodiment R.sub.17 is hydrogen, alkyl, alkenyl, alkylcarboxy, or cycloalkyl. Where X is --N.sup.+(R.sub.17aR.sub.17b)--, in one particularly preferred embodiment R.sub.17a and R.sub.17b are independently hydrogen, alkyl, alkenyl, or cycloalkyl.
[0051] Representative morphinan-6-one compounds corresponding to Formula (2) (and the various preferred substituent group definitions described above) which can be treated according to the process described herein include, for example, oxymorphone, naloxone, naltrexone, naltrexone methylbromide, nalbuphone, noroxymorphone, hydromorphone, hydrocodone, oxycodone, diethoxycarbonyl-noroxymorphone, salts thereof, and the like. Additionally, derivatives of the above morphinan-6-one compounds which can be treated according to the process described herein include, for example, N-demethylated-, 10-hydroxy-, 10-halo, and 10-keto-morphinan-6-one derivatives, their protected analogs, and the like.
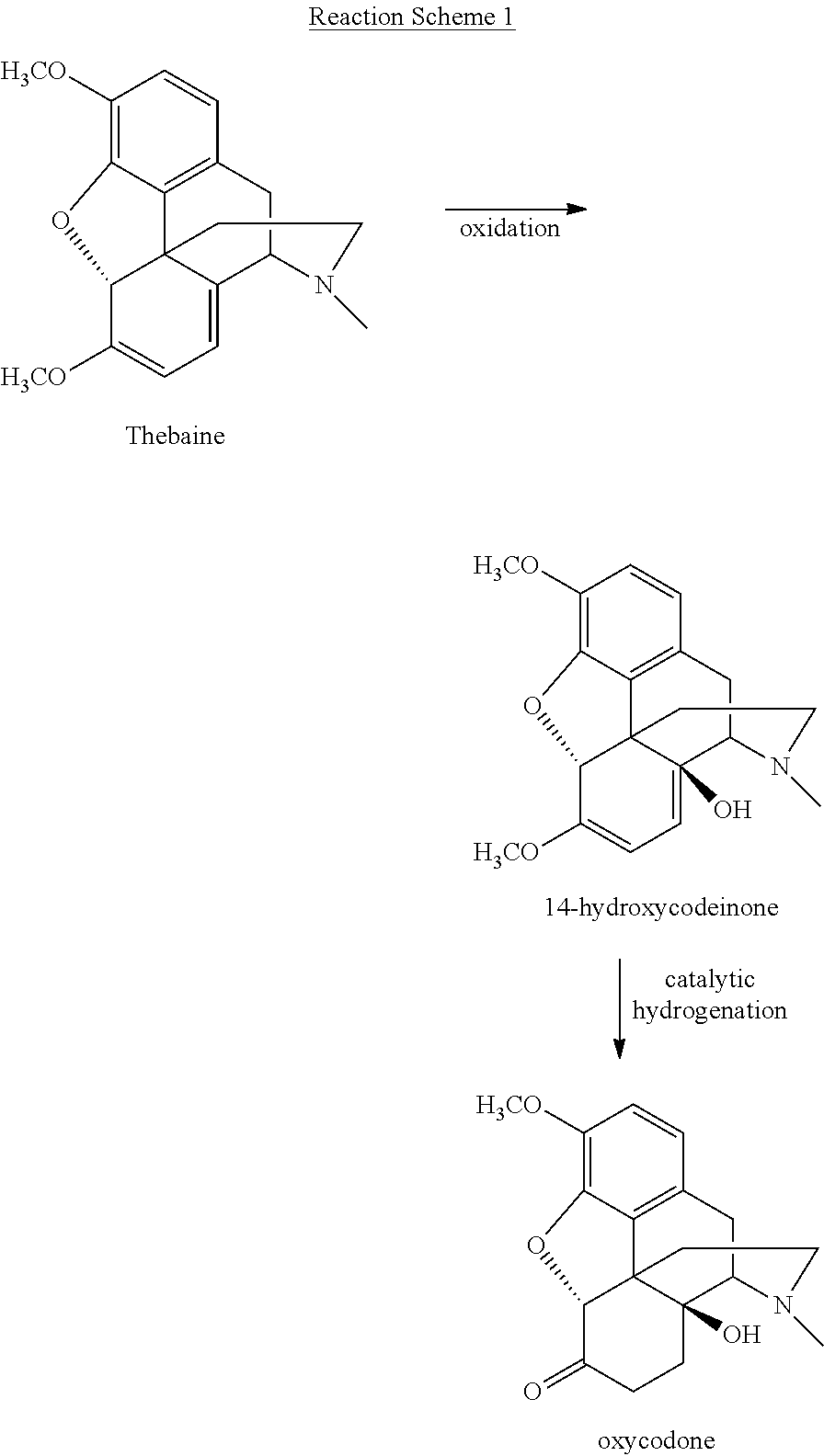
[0052] The method of producing the above-described morphinan-6-one compounds for use in the present invention is not narrowly critical, and various methods for producing morphinan-6-one compounds are well known in the art. For example, commercial processing methods for producing morphinan compounds typically involve the extraction of an opium alkaloid (e.g., thebaine) from poppies, followed by various conventional precipitation and purification steps known to those of skill in the art. By way of further example, the morphinan-6-one compound oxycodone may be produced from thebaine in a substantially two-step process, as illustrated in Reaction Scheme 1:
##STR00007##
[0053] Alternatively, various synthetic methods for producing the above-described morphinan-6-one compounds are also known. In these synthetic methods, a Grewe cyclization reaction is commonly used to form nordihydrothebainone products such as by the processes described in U.S. Pat. Nos. 4,368,326, 4,410,700, 4,521,601, 4,556,712, 4,613,668, 4,727,146, the entire disclosures of which are hereby incorporated by reference herein. Additionally, various methods useful for the semi-synthesis of morphinan compounds and intermediates are known. For example, U.S. Pat. No. 6,177,567 to Chiu et al. and U.S. Pat. No. 6,008,355 to Huang et al. (each of which is hereby incorporated by reference herein) describe methods for the synthesis of oxycodone from codeine. These and other conventional practices are generally applicable in carrying out the preparation of morphinan-6-one compounds and .alpha.,.beta.-unsaturated ketone compounds that may be treated according to the processes described herein.
[0054] As noted above, in the various conventional processes for producing morphinan-6-one compounds described above, the resulting morphinan product typically also includes some amount of an .alpha.,.beta.-unsaturated ketone compound present as an impurity in addition to the desired morphinan-6-one compound. The .alpha.,.beta.-unsaturated ketone compounds present as impurities generally correspond to Formula (3):
##STR00008##
wherein X, R.sub.1, R.sub.2, R.sub.3, R.sub.10, and R.sub.14 are defined as above.
[0055] Reaction Conditions
[0056] As noted above, the morphinan products produced from conventional processes for preparing morphinan-6-one compounds also yield some amount of an .alpha.,.beta.-unsaturated ketone present as an impurity; that is, both the morphinan-6-one compound corresponding to Formula (2) and the .alpha.,.beta.-unsaturated ketone compound corresponding to Formula (3) are present in the morphinan product.
[0057] The morphinan products produced from conventional morphinan processing methods typically comprise less than about 2% by weight of an .alpha.,.beta.-unsaturated ketone compound. Preferably, the morphinan products comprise less than about 1% by weight of an .alpha.,.beta.-unsaturated ketone compound. More preferably, the morphinan products comprise less than about 0.8% by weight of an .alpha.,.beta.-unsaturated ketone compound. Still more preferably, the morphinan products comprise less than about 0.5% by weight of an .alpha.,.beta.-unsaturated ketone compound. As noted above, however, it is desirable to minimize or further minimize the concentration of .alpha.,.beta.-unsaturated ketone compounds present in such products.
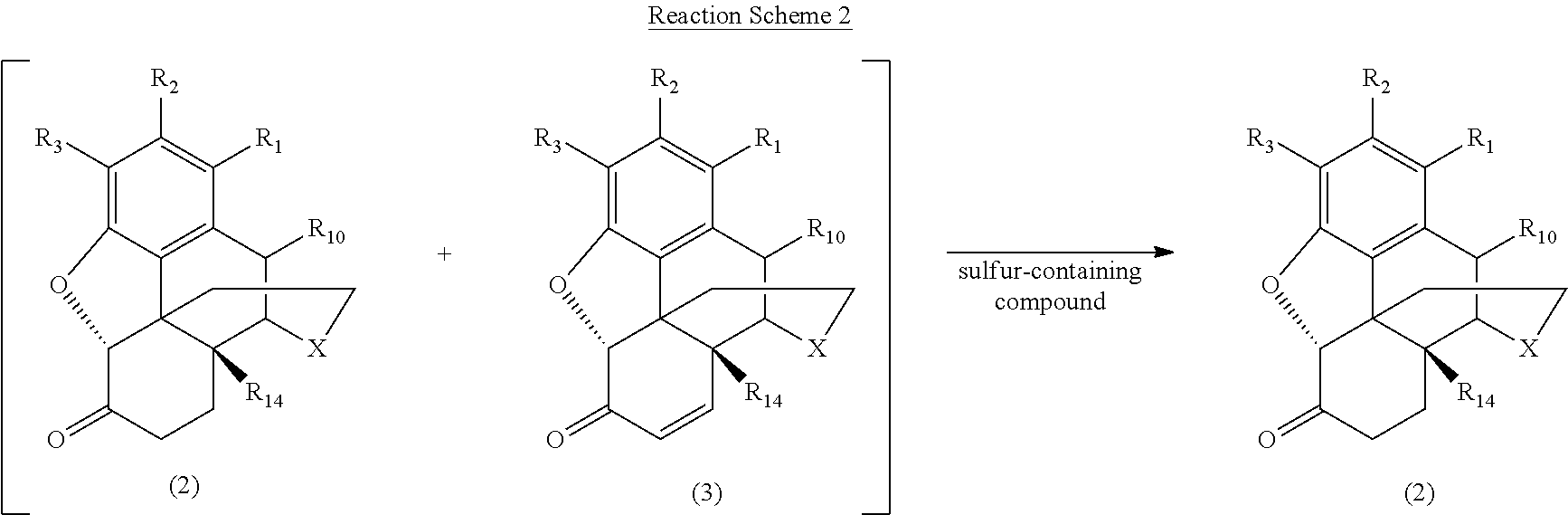
[0058] According to the present invention, a reaction mixture is formed including a morphinan-6-one compound of Formula (2) and an .alpha.,.beta.-unsaturated ketone compound of Formula (3). The morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound may be produced by any conventional method (such as those described above), and the morphinan-6-one compound may exist as the free base or as a salt, such as the hydrochloride salt. The reaction mixture is treated with a sulfur-containing compound to reduce the concentration of the .alpha.,.beta.-unsaturated ketone compound (either by forming additional morphinan-6-one compound or by facilitating the removal of the .alpha.,.beta.-unsaturated ketone compound), and the morphinan-6-one compound is recovered to produce the desired morphinan-6-one product. This process is generically illustrated in Reaction Scheme 2, wherein the reaction mixture including the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound is shown in brackets, and X, R.sub.1, R.sub.2, R.sub.3, R.sub.10, and R.sub.14 are defined as above.
##STR00009##
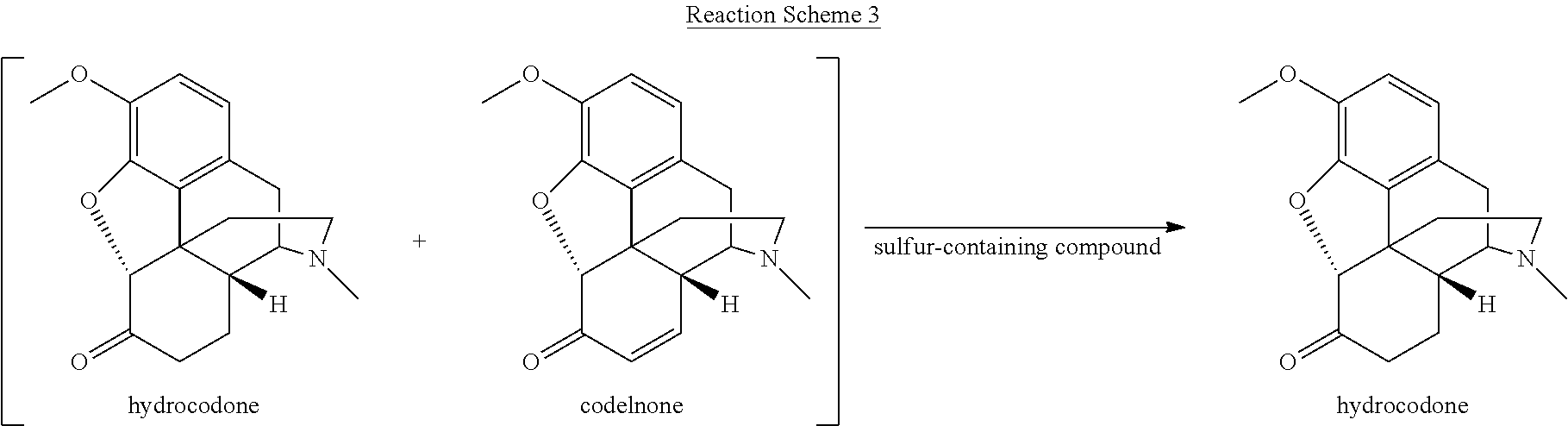
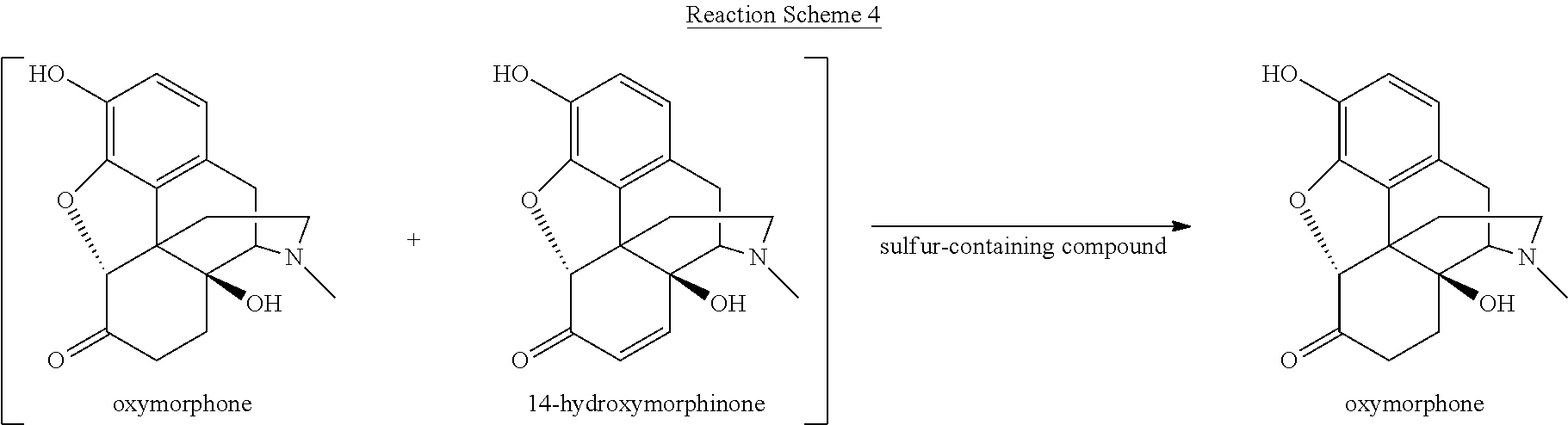
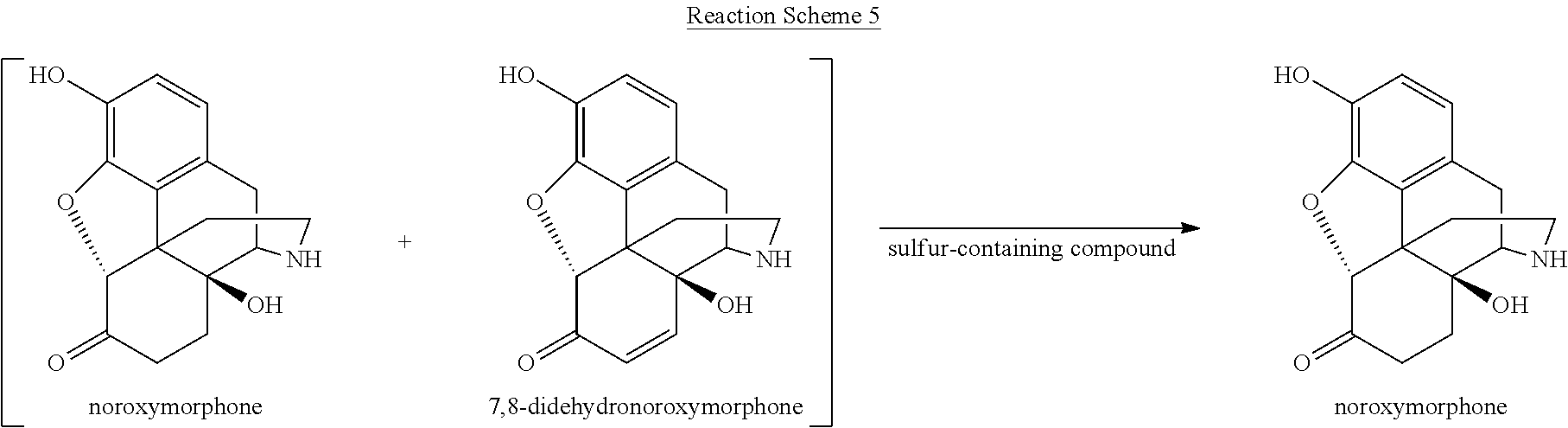
[0059] Various reaction mixtures (bracketed) including a morphinan-6-one compound and an .alpha.,.beta.-unsaturated ketone compound may be treated according to the processes described herein to yield various highly pure morphinan-6-one products, as illustrated in Reaction Schemes 3-10.
##STR00010##
##STR00011##
##STR00012##
##STR00013##
##STR00014##
##STR00015##
##STR00016##
##STR00017##
[0060] According to various embodiments, the reaction mixture is formed by dissolving or otherwise dispersing the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound in a media material (i.e., a morphinan product including the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound is dispersed in the media material). The reaction mixture is then treated with a sulfur-containing compound. Ideally, the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound are in solution, but a heterogeneous mixture may also be treated according to the processes described herein.
[0061] The media material is desirably an aqueous media or an aqueous/organic solvent biphasic media. Exemplary aqueous media for use in the process of the present invention includes, for example, water, water/alcohol mixtures, dilute inorganic solvents such as dilute sulfuric acid, ethereal solvents such as dioxane or tetrahydrofuran, combinations thereof, and the like. Exemplary organic solvents for use in aqueous/organic solvent biphasic media includes, for example, butanone, ethyl acetate, butanol, diethyl ether, benzene, chloroform, tetrachloroethylene, toluene, 1,1,1-trichloroethane, carbon tetrachloride, dibutyl ether, cyclohexane, hexane, dipentyl ether, heptane, hexadecane, combinations thereof, and the like.
[0062] Generally, a sufficient amount of media material to substantially solubilize the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound in the reaction mixture is desired, Higher amounts of media material may increase the costs of manufacturing, as the more dilute reaction mixture may require additional process cycle time, or require the removal or excess media material during subsequent processing steps.
[0063] The weight ratio of media material to morphinan-6-one compound in the reaction mixture is preferably from about 1:1 to about 50:1. More preferably, the weight ratio of media material to morphinan-6-one compound in the reaction mixture is from about 1:1 to about 25:1. For example, the weight ratio of media material to morphinan-6-one compound in the reaction mixture may be from about 1:1 to about 5:1, from about 1:1 to about 10:1, from about 1:1 to about 15:1, or from about 1:1 to about 20:1. Still more preferably, the weight ratio of media material to morphinan-6-one compound in the reaction mixture is from about 5:1 to about 25:1. For example, the weight ratio of media material to morphinan-6-one compound in the reaction mixture may be from about 5:1 to about 10:1, from about 5:1 to about 15:1, or from about 5:1 to about 20:1. Still more preferably, the weight ratio of media material to morphinan-6-one compound in the reaction mixture is from about 5:1 to about 15:1. For example, the weight ratio of media material to morphinan-6-one compound in the reaction mixture may be from about 5:1 to about 6:1, from about 5:1 to about 7:1, from about 5:1 to about 8:1, from about 5:1 to about 9:1, from about 5:1 to about 10:1, from about 5:1 to about 11:1, from about 5:1 to about 12:1, from about 5:1 to about 13:1, or from about 5:1 to about 14:1. Most preferably, the weight ratio of media material to morphinan-6-one compound in the reaction mixture is from about 5:1 to about 11:1. It will be understood that some portion of the media material may be derived from the sulfur-containing compound itself (e.g., as water of hydration).
[0064] Optionally, a phase transfer catalyst may also be added to the aqueous/organic solvent biphasic media. The phase transfer catalyst is preferably any suitable composition for use in the transfer of reactants (i.e., morphinan-6-one compounds, .alpha.,.beta.-unsaturated ketone compounds, and/or sulfur-containing compounds) between the aqueous and organic solvent interface. Typically, the phase transfer catalyst is an ammonium-based compound, such as a quaternary ammonium salt. Suitable quaternary ammonium salts for use as phase transfer catalysts include tetraalkylammonium salts such as, for example, tetramethyl-, tetraethyl-, tetrabutyl-, tetrahexyl-, tetraoctyl-, methyltriphenyl-, methyltrioctyl-, benzyltrimethyl-, benzyltriethyl-, benzyltributyl-, hexadecyltrimethyl-ammonium salts, and the like. Suitable salts include, for example, halide, hydroxide, bicarbonate, bisulfate, thiocyanate, tetrafluoroborate, and the like. Other phase transfer catalysts such as phosphonium salts may be suitable as well.
[0065] A variety of sulfur-containing compounds may be utilized to treat the reaction mixture and reduce the concentration of the .alpha.,.beta.-unsaturated ketone compound according to the processes described herein. In various embodiments, the sulfur-containing compound is a sulfur-containing nucleophile. As utilized herein, "nucleophile" refers to an ion or molecule that donates a pair of electrons to an atomic nucleus to form a covalent bond. In other embodiments, the sulfur-containing compound is a sulfur-containing reducing agent. As utilized herein, "reducing agent" refers to an agent having the ability to add one or more electrons to an atom, ion or molecule. In either of the two embodiments described above (i.e., when the sulfur-containing compound is a sulfur-containing nucleophile or a sulfur-containing reducing agent), the sulfur-containing compound is a compound having the ability to effect the reduction of and/or a 1,4 addition across the .alpha.,.beta.-unsaturated bond of the .alpha.,.beta.-unsaturated ketone compound.
[0066] In one embodiment, the sulfur-containing compound is a sulfur-containing inorganic acid or salt thereof. Suitable sulfur-containing inorganic acids include, for example, hydrosulfuric acid (H.sub.2S); sulfurous acid (H.sub.2SO.sub.3); persulfuric acid (H.sub.2SO.sub.5); thiosulfurous acid (H.sub.2S.sub.2O.sub.2); dithionous acid (H.sub.2S.sub.2O.sub.4); disulfurous acid (H.sub.2S.sub.2O.sub.5); dithionic acid (H.sub.2S.sub.2O.sub.6); pyrosulfuric acid (H.sub.2S.sub.2O.sub.7); peroxydisulfuric acid (H.sub.2S.sub.2O.sub.5); trithionic acid (H.sub.2S.sub.3O.sub.6); tetrathionic acid (H.sub.2S.sub.4O.sub.6); pentathionic acid (H.sub.2S.sub.5O.sub.6); chiorosulfonic acid (HSO.sub.3Cl); furosulfonic acid (HSO.sub.3F); sulfamic acid (HSO.sub.3NH.sub.2); salts thereof; and the like.
[0067] Generally, the sulfur-containing inorganic acid salt may be an alkali metal salt or an alkaline earth metal salt. For example, the salt may be a monovalent or divalent cation selected from Li.sup.+, Na.sup.+, K.sup.+, Rb.sup.+, Cs.sup.+, Fr.sup.+, Be.sup.2+, Mg.sup.2+, Ca.sup.2+, Sr.sup.2+, Ba.sup.2+, or Ra.sup.2+. Preferably, the salt is selected from the group consisting of Li.sup.+, Na.sup.+, K.sup.+, Mg.sup.2+, Ca.sup.2+, and combinations thereof.
[0068] Alternatively, the sulfur-containing inorganic acid salt may be an ammonium salt (NH.sub.4.sup.+) or a quaternary ammonium salt. For example, the sulfur-containing inorganic acid salt may be a tetraalkylated ammonium salt; that is, a quaternary ammonium salt substituted with four alkyl groups preferably having from 1 to about 18 carbon atoms. Suitable tetraalkylated ammonium salts include, for example, tetramethylammonium salts, tetraethylammonium salts, tetrapropylammonium salts, tetrabutylammonium salts, and the like.
[0069] In one particular embodiment, the sulfur-containing inorganic acid is dithionous acid (H.sub.2S.sub.2O.sub.4) or salts thereof. By way of example, salts of dithionous acid include MHS.sub.2O.sub.4 and M.sub.2S.sub.2O.sub.4, wherein M is selected from alkali metal salts, alkaline earth metal salts, ammonium salt (NH.sub.4.sup.+), and quaternary ammonium salts. According to this embodiment, the .alpha.,.beta.-unsaturated ketone compound is chemically reduced to form the morphinan-6-one compound upon treatment with the sulfur-containing compound, discussed in further detail below.
[0070] In another particular embodiment, the sulfur-containing inorganic acid is selected from the group consisting of sulfurous acid (H.sub.2SO.sub.3); disulfurous acid (H.sub.2S.sub.2O.sub.5); and salts thereof. By way of example, salts of sulfurous acid and disulfurous acid include MHSO.sub.3, M.sub.2SO.sub.3, MHS.sub.2O.sub.5, and M.sub.2S.sub.2O.sub.5 wherein M is selected from alkali metal salts, alkaline earth metal salts, ammonium salt (NH.sub.4.sup.+), and quaternary ammonium salts. According to this embodiment, the sulfur-containing inorganic acid or salt thereof is one which dissociates into the bisulfite ion (HSO.sub.3.sup.-) and/or the sulfite ion (SO.sub.3.sup.2-) in the reaction mixture. It will be understood by one of ordinary skill in the art that sulfurous acid (H.sub.2SO.sub.3) generally exists as a solution of SO.sub.2 (commonly about 6%) in water. The pKa of sulfurous acid (H.sub.2SO.sub.3) is about 1.78 and its ionization expression is:
H.sub.2O+SO.sub.2H.sub.2SO.sub.3H.sup.++HSO.sub.3.sup.-H.sup.++SO.sub.3.- sup.2-
According to this embodiment, various 1,2- and 1,4-sulfonated addition products are formed from the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound by reaction with the bisulfite ion and/or the sulfite ion, discussed in further detail below.
[0071] In another particular embodiment, the sulfur-containing compound is a thiol having the formula: R--SH, wherein R is hydrocarbyl, substituted hydrocarbyl, or heterocyclo. For example, R may be substituted or unsubstituted alkyl, alkenyl, alkynyl, or aryl. Exemplary thiols having the formula R--SH, wherein R is defined as above, include alkyl or aryl thiols such as methanethiol, ethanethiol, benzenethiol, and the like. Other exemplary thiols include thiocarboxylic acids and salts thereof (e.g., thiobenzoic acid) and thiol-terminated carboxylic acids and salts thereof (e.g., thioglycolic acid (mercaptoacetic acid), mercaptopropionic acid, and the like). Still other exemplary thiols include amino acids (e.g., L- or D,L-cysteine), other thiol-containing amines and/or quaternary salts thereof (e.g., cysteamine HCl, thiocholine, and the like), or polymer-bound thiols (e.g., polycysteine, polyvinylarylthiol, and the like). In one preferred embodiment, the thiol is benzenethiol. Without being bound to one theory, it is believed that the thiol forms various 1,2- and 1,4-sulfonated addition products from the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound.
[0072] The amount of sulfur-containing compound utilized to treat the reaction mixture may vary considerably according to the various reaction mixture components (such as the particular morphinan-6-one compound, the .alpha.,.beta.-unsaturated ketone compound, and/or the media material) and concentrations thereof, time of reaction, temperature, pressure, and the like. Relatively high usage rates of sulfur-containing compound generally offer no significant advantages and tend to waste chemicals and/or reactor volume.
[0073] The molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture is typically greater than about 0.5:1. Preferably, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture is from about 0.5:1 to about 3.0:1. For example, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture may be from about 0.5:1 to about 0.8:1, from about 0.5:1 to about 1.0:1, from about 0.5:1 to about 1.5:1, from about 0.5:1 to about 2.0:1, or from about 0.5:1 to about 2.5:1. More preferably, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture is from about 0.6:1 to about 2.8:1. For example, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture may be from about 0.6:1 to about 0.8:1, from about 0.6:1 to about 1.0:1, from about 0.6:1 to about 1.5:1, from about 0.6:1 to about 2.0:1, or from about 0.6:1 to about 2.5:1. Most preferably, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture is from about 0.8:1 to about 2.5:1. For example, the molar ratio of sulfur-containing compound to morphinan-6-one compound in the reaction mixture may be from about 0.8:1 to about 1.0:1, from about 0.8:1 to about 1.2:1, from about 0.8:1 to about 1.4:1, from about 0.8:1 to about 1.6:1, from about 0.8:1 to about 1.8:1, from about 0.8:1 to about 2.0:1, from about 0.8:1 to about 2.2:1, or from about 0.8:1 to about 2.4:1.
[0074] The treatment of the reaction mixture with the sulfur-containing compound may be carried out in ambient air or in an oxygen-free environment. Preferably, the treatment is carried out in an inert atmosphere such as, for example, argon or nitrogen gas. The treatment is preferably carried out at a pressure of from about 0.5 atm to about 2.0 atm. More preferably, the treatment is carried out at a pressure of from about 0.75 atm to about 1.5 atm; most preferably from about 0.9 atm to about 1.25 atm.
[0075] In various embodiments, the pH of the reaction mixture during treatment with the sulfur-containing compound is greater than about 3. Typically, the pH of the reaction mixture during treatment is less than about 10, although the upper pH limit may depend on the treatment time and/or solubility of the various reaction mixture components. Preferably, the pH of the reaction mixture during treatment with the sulfur-containing compound is from about 3 to about 9; more preferably from about 6 to about 9. For example, the pH of the reaction mixture during treatment with the sulfur-containing compound may be about 3, about 4, about 5, about 6, about 7, about 8, or about 9. Most preferably, the treatment occurs at a pH of from about 6 to about 7.25. Upon the addition of the sulfur-containing compound to the reaction mixture including the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound, the pH may be adjusted to the desired level (e.g. using a base such as ammonium hydroxide). Other suitable bases include, for example, sodium hydroxide, potassium hydroxide, and the like.
[0076] The time of reaction is generally a function of the other variables in the reaction, such as pH, ratio of media material to morphinan-6-one compound, amount of sulfur-containing compound, and the like. Typically, some reduction of the concentration of .alpha.,.beta.-unsaturated ketone compound in the reaction mixture can be observed after about 1 hour. Preferably, the reaction mixture is treated with the sulfur-containing compound for at least about 1 hour. In some embodiments, the time of reaction is less than about 24 hours. In other embodiments, the time of reaction is from about 1 hour to about 18 hours; in still other embodiments from about 1 hour to about 15 hours; in still other embodiments from about 1 hour to about 10 hours. More preferably, the reaction mixture is treated with the sulfur-containing compound for about 1 hour to about 5 hours. For example, the reaction mixture may be treated with the sulfur-containing compound for about 1 hour, for about 2 hours, for about 3 hours, for about 4 hours, or for about 5 hours.
[0077] The temperature of the reaction mixture during treatment with the sulfur-containing compound is generally from about 0.degree. C. to about 100.degree. C. For example, the temperature of the reaction mixture during treatment with the sulfur-containing compound may be from about 10.degree. C. to about 90.degree. C., from about 20.degree. C. to about 80.degree. C., or from about 30.degree. C. to about 70.degree. C. Preferably, the temperature of the reaction mixture during treatment with the sulfur-containing compound is above room temperature. The preferred reaction temperature may vary for each morphinan-6-one. More preferably, the temperature of the reaction mixture during treatment with the sulfur-containing compound is from about 30.degree. C. to about 50.degree. C. For example, the temperature of the reaction mixture during treatment with the sulfur-containing compound may be about 30.degree. C., about 35.degree. C., about 40.degree. C., about 45.degree. C., or about 50.degree. C.
[0078] Once the treatment is complete or has proceeded as long as desired, the treated morphinan-6-one compound is recovered to produce the morphinan-6-one product. Advantageously, the morphinan-6-one compound may be recovered from the reaction mixture without the use of an organic solvent. The absence of the need for organic solvents in the recovery process not only provides various environmental and material handling benefits, but also results in a more efficient process suitable for industrial scale applications. Typically, the morphinan-6-one compound is precipitated from the reaction mixture as a base (or salt if desirable) and may then be readily converted into a generally more pharmaceutically acceptable form, if so desired. For example, the pH of the reaction mixture is typically adjusted to about 9-10 or greater with a suitable base such as ammonium hydroxide, and the (desired) precipitated compound recovered. Generally speaking, this pH is at the point wherein opium alkaloids are not ionized. The morphinan-6-one compounds can then be optionally converted into a form more physiologically tolerable, such as the hydrochloride salt, e.g., oxycodone HCl, using conventional methods known to those of skill in the art. For example, the morphinan-6-one base can be dissolved or otherwise dispersed in water, reacted with an acid such as HCl, heated, and cooled to precipitate the morphinan-6-one salt. By way of an alternative example, the morphinan-6-one base can be dissolved or otherwise dispersed in an alcohol solvent (e.g., methanol, ethanol, etc.) or a solvent system (i.e., a mixture of solvents), reacted with concentrated HCl or an HCl/alcohol mixture, and cooled to precipitate the morphinan-6-one hydrochloride salt. By way of another example, the morphinan-6-one base can be dissolved or otherwise dispersed in water, alcohol solvent, or a solvent system, reacted with gaseous HCl, heated, and cooled to precipitate the morphinan-6-one hydrochloride salt.
[0079] Treatment Reaction Mechanisms
[0080] Without being bound to one theory, it is believed that the reduction of the concentration of .alpha.,.beta.-unsaturated ketone compounds in the reaction mixture is performed via different mechanisms, depending on the particular sulfur-containing compound selected to treat the reaction mixture.
[0081] In one embodiment, the .alpha.,.beta.-unsaturated ketone compound is reduced by the sulfur-containing compound to form the desired .alpha.,.beta.-saturated morphinan-6-one compound. See, e.g., Camps et al., Tetrahedron Letters, Vol. 29, No. 45, 1988, 5811-5814; Louis-Andre et al., Tetrahedron Letters, Vol. 26, No. 7, 1985, 831-832). By way of example, dithionous acid (H.sub.2S.sub.2O.sub.4) and salts thereof (e.g., MHS.sub.2O.sub.4 or M.sub.2S.sub.2O.sub.4, wherein M is defined as above) operate according to this mechanism; other sulfur-containing compounds, however, may also operate according to the same or a similar mechanism. Reaction Scheme 11 generally illustrates the reduction of the .alpha.,.beta.-unsaturated ketone compound (3) to form the desired morphinan-6-one compound (2) according to this embodiment, wherein X, R.sub.1, R.sub.2, R.sub.3, R.sub.10, and R.sub.14 are defined as above.
##STR00018##
[0082] In an alternative embodiment, various 1,2- and 1,4-sulfonated addition products are formed during treatment that assist in the removal of the .alpha.,.beta.-unsaturated ketone compounds from the reaction mixture. As noted above, several sulfur-containing compounds dissociate into various sulfur-containing species. In particular, sulfurous acid (H.sub.2SO.sub.3), disulfurous acid (H.sub.2S.sub.2O.sub.5), and their salts dissociate into, among other things, bisulfite (HSO.sub.3.sup.-) and sulfite (SO.sub.3.sup.2-).
[0083] Bisulfite has been shown to add via radical initiation across isolated double bonds (see, e.g., March, J., Advanced Organic Chemistry, p. 688, J. Wiley & Sons, 1985, 3d. ed.) and/or add via an ionic mechanism (see, e.g., Gilbert, E.; Sulfonation and Related Reactions, p. 152, Interscience, N.Y. 1965; Patai et al, The Chemistry of Alkenes, p. 478, Interscience, London 1965). Without being bound to one theory, it is believed that when the reaction mixture is treated with sulfurous acid, disulfurous acid, or salts thereof and the pH is adjusted to between about 3 and about 9, certain 1,2- and 1,4-addition products and adducts are stably and/or reversibly formed from the .alpha.,.beta.-unsaturated ketone compound and the morphinan-6-one compound. It is further believed that the products are generally stable within the pH range of from about 3 to about 9, and adjusting the pH outside of this range after their formation from the .alpha.,.beta.-unsaturated ketone compounds and the morphinan-6-one compounds facilitates the removal of the .alpha.,.beta.-unsaturated ketone compound from the reaction mixture, resulting in a highly pure morphinan-6-one product.
[0084] One preferred embodiment of the present invention is illustrated in Reaction Schemes 12A and 12B, wherein X, R.sub.1, R.sub.2, R.sub.3, R.sub.10, and R.sub.14 are defined as above and M is a monovalent or divalent cation. For example, M may be one or more alkali metal or alkaline earth metal monovalent or divalent cations from the sulfur-containing compound. Alternatively, M may be one or more monovalent or divalent cations from the alkaline compound (e.g., NaOH, KOH, NH.sub.4OH, etc.) used to adjust the pH of the reaction mixture to between about 3 and about 9 after the addition of the sulfur-containing compound to the reaction mixture.
##STR00019##
##STR00020##
[0085] As shown in Reaction Schemes 12A and 12B, various 1,2- and 1,4-sulfonated compounds are formed from the morphinan-6-one compound (2) (scheme 12A) and the .alpha.,.beta.-unsaturated ketone compound (3) (scheme 12B) upon treatment of a reaction mixture including these compounds with a sulfur-containing compound at a pH of between about 3 and about 9. While it is understood that sulfurous acid, disulfurous acid, and salts thereof operate according to the mechanism illustrated in Reaction Schemes 12A, 12B, and 12C, other sulfur-containing compounds may also operate according to the same or a similar mechanism. For example, thiols (e.g., benzenethiol) may also operate according to the mechanism described in connection with Reaction Schemes 12A, 12B, and 12C.
[0086] Particularly, when the reaction mixture is treated with a sulfur-containing compound and the pH of the reaction mixture is adjusted to between about 3 and about 9, the morphinan-6-one compound (2) forms the reversible, water-soluble 1,2-bisulfite adduct (2A). Once the reaction mixture is sufficiently in solution in the media material and/or the sulfur-containing compound, dissociated sulfur specie (such as sulfite and bisulfite) react more readily with the .alpha.,.beta.-unsaturated ketone compound (3) also present in the reaction mixture.
[0087] As illustrated in Reaction Scheme 12B, one reaction between the .alpha.,.beta.-unsaturated ketone compound (3) and the sulfur-containing compound involves the rapid and reversible 1,2-addition of the bisulfite to the carbonyl (similar to the reaction of the sulfur-containing compound with the morphinan-6-one compound illustrated in Reaction Scheme 12A) to form the reversible 1,2-adduct (3A) from the .alpha.,.beta.-unsaturated ketone compound (3). Another reaction between the sulfur-containing compound and the .alpha.,.beta.-unsaturated ketone compound (3) is the slower 1,4-addition, forming the more stable 1,4-addition product (3B). The introduction of the sulfonate group in the .beta.-position generally enhances the reactivity of the carbonyl group by destroying its conjugation with the double bond, such that the reversible product is a 1,2- and 1,4-bis adduct (3C) (see Patai et al., The Chemistry of Alkenes, p. 478, Interscience, London 1965).
[0088] Reaction Scheme 12C illustrates the removal of certain addition products formed in the reaction mixture according to Reaction Schemes 12A and 12B and the resulting highly pure morphinan-6-one product, wherein X, R.sub.1, R.sub.2, R.sub.3, R.sub.10, R.sub.14, and M are defined as above.
##STR00021##
[0089] As illustrated in Reaction Scheme 12C, the removal of the .alpha.,.beta.-unsaturated ketone addition products is generally based in the differences in solubility of the 1,4-addition product (3B) generated from the .alpha.,.beta.-unsaturated ketone compound and the desired morphinan-6-one compound (2). Adjusting the pH outside of the range between about 3 and about 9 (i.e., the pH is adjusted to less than about 3 or the pH is adjusted to greater than about 9) with an acid (e.g., sulfuric acid (H.sub.2SO.sub.4)) or a base (e.g., ammonium hydroxide (NH.sub.4OH)) results in the decomposition of the 1,2-addition products of each compound, rendering the desired morphinan-6-one compound (2) insoluble in water. The relatively more stable 1,4-addition product (3B) formed from the .alpha.,.beta.-unsaturated ketone compound remains and is water-soluble in the final mixture at an alkaline pH (e.g., pH .about.9 or greater). The 1,4-addition product (38) may thus be removed from the mixture with the mother liquor, leaving the insoluble morphinan-6-one base (2). The desired morphinan-6-one base may then be converted into a more physiologically-tolerable salt form, such as the hydrochloride salt, using methods known to those of skill in the art.
[0090] One particularly preferred embodiment of the present invention is illustrated in Reaction Schemes 13A and 13B, wherein M is defined as above.
##STR00022##
##STR00023##
[0091] As shown in Reaction Schemes 13A and 13B, various sulfonated compounds are formed from oxycodone (20) (scheme 13A) and the .alpha.,.beta.-unsaturated ketone compound 14-hydroxycodeinone (30) (scheme 13B) upon treatment of a reaction mixture including these compounds with a sulfur-containing compound at a pH of between about 3 and about 9. As discussed above, while it is generally understood that sulfurous acid, disulfurous acid, and salts thereof operate according to the mechanism described in Reaction Schemes 13A and 13B, other sulfur-containing compounds may also operate according to the same or a similar mechanism.
[0092] Particularly, when the reaction mixture is treated with a sulfur-containing compound and the pH of the reaction mixture is adjusted to between about 3 and about 9, oxycodone (20) forms the reversible, water-soluble 1,2-bisulfite adduct (20A). Once the reaction mixture is sufficiently in solution in the media material and the sulfur-containing compound, dissociated sulfur specie (such as sulfite and bisulfite) react more readily with the 14-hydroxycodeinone (30) also present in the reaction mixture.
[0093] As illustrated in Reaction Scheme 13B, one reaction between 14-hydroxycodeinone (30) and the sulfur-containing compound involves the rapid and reversible 1,2-addition of the sulfite to the carbonyl (similar to the reaction of the sulfur-containing compound with oxycodone illustrated in Reaction Scheme 13A) to form the reversible 1,2-adduct (30A) from 14-hydroxycodeinone. Another reaction between the sulfur-containing compound and 14-hydroxycodeinone (30) is the slower 1,4-addition, forming the more stable 1,4-addition product (30B). The introduction of the sulfonate group in the n-position generally enhances the reactivity of the carbonyl group by destroying its conjugation with the double bond, such that the reversible product is a 1,2- and 1,4-bis adduct (30C) (see Patai et al., The Chemistry of Alkenes, p. 478, Interscience, London 1965).
[0094] Reaction Scheme 13C illustrates the removal of certain addition products formed in the reaction mixture according to Reaction Schemes 13A and 13B and the resulting highly pure oxycodone, wherein M is defined as above.
##STR00024##
[0095] As illustrated in Reaction Scheme 13C, the removal of the 14-hydroxycodeinone addition products is generally based on the differences in solubility of the 1,4-addition product (30B) generated from 14-hydroxycodeinone and the desired oxycodone (20). Adjusting the pH outside of the range between about 3 and about 9 (i.e., the pH is adjusted to less than about 3 or greater than about 9) with an acid (e.g., sulfuric acid (H.sub.2SO.sub.4)) or a base (e.g., ammonium hydroxide (NH.sub.4OH)) results in the decomposition of the 1,2-addition products of each compound, rendering the desired oxycodone (20) insoluble in water. The relatively more stable 1,4-addition product (30B) formed from 14-hydroxycodeinone remains and is water soluble in the final mixture at an alkaline pH (e.g., pH .about.9 or greater). The 1,4-addition product (30B) may thus be removed from the mixture with the mother liquor, leaving the insoluble oxycodone base (20). The oxycodone base may then be converted into a more physiologically-tolerable salt form, such as the hydrochloride salt, using methods known to those of skill in the art.
[0096] Removal of Residual Sulfur-Containing Species from the Reaction Mixture
[0097] Using the process described herein to reduce the concentration of .alpha.,.beta.-unsaturated ketone compounds from a reaction mixture by treating the reaction mixture with a sulfur-containing compound may result in the undesirable accumulation of residual sulfur-containing species (such as sulfites and bisulfites) in the reaction mixture and/or final morphinan-6-one product. Accordingly, the residual sulfur-containing species may be optionally substantially removed from the reaction mixture following the treatment with the sulfur-containing compound using a variety of methods known to those of skill in the art.
[0098] As described above, in various embodiments 1,2- and 1,4-sulfonated addition products may be formed by the reaction of a sulfur-containing compound with the morphinan-6-one compound and the .alpha.,.beta.-unsaturated ketone compound at a pH of between about 3 to about 9. The adjustment of the pH outside of this range eliminates the 1,2-addition products, renders the morphinan-6-one compound insoluble in water, and the remaining water soluble 1,4-addition product can be removed in the waste stream.
[0099] To optionally substantially remove the residual sulfur-containing species upon completion of the reaction with the sulfur-containing compound, the pH of the reaction mixture may be adjusted to less than about 3 (instead of adjusting the pH to greater than 9) with an acid (e.g., sulfuric acid (H.sub.2SO.sub.4)) and manipulated prior to the precipitation of the morph inan-6-one compound as described in detail above. More preferably, the pH is adjusted to less than about 2. The reduction in pH converts any residual sulfur species that may be present in the reaction mixture into SO.sub.2 gas, which typically has a limited solubility in water. In one embodiment, the SO.sub.2 gas may then be optionally heat refluxed out of the reaction mixture by conventional means known to those of skill in the art. Typically, the reaction mixture is heat refluxed for about 2 hours to about 5 hours. The temperature and pressure during reflux are also generally variable. For example, the temperature of the reaction mixture during reflux is typically from about 20.degree. C. to about 100.degree. C., and the reflux may be performed at a pressure of from about 0.003 atm to about 1.0 atm. Alternatively, substantially all of the water (and the SO.sub.2 gas) may be optionally distilled off to a receiver tank and discarded. This procedure is also generally known to those of skill in the art.
[0100] As discussed above, after treatment of the reaction mixture with the sulfur-containing compound to reduce the concentration of the .alpha.,.beta.-unsaturated ketone compound in the reaction mixture, the morphinan-6-one compound is recovered to produce the desired morphinan-6-one product. Generally speaking, recovery refers to one or more of the precipitation, filtration and drying of the morphinan-6-one base, the formation of the physiologically acceptable morphinan-6-one salt (e.g., the hydrochloride salt), the removal of the residual sulfur-containing species, and/or combinations thereof, to produce a morphinan-6-one product.
[0101] The treatment of the reaction mixture with a sulfur-containing compound according to the various processes and embodiments described herein significantly reduces the concentration of .alpha.,.beta.-unsaturated ketone compounds in the reaction mixture, and a highly pure morphinan-6-one product may be produced therefrom. Typically, the morphinan-6-one product comprises less than about 0.1% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound. For example, the morphinan-6-one product may comprise less than about 0.05% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound Preferably, the morphinan-6-one product comprises less than about 0.01% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound. For example, the morphinan-6-one product may comprise less than about 0.005% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound. More preferably, the morphinan-6-one product comprises less than about 0.001% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound. For example, the morphinan-6-one product may comprise less than about 0.0005% (by weight morphinan-6-one product) of an .alpha.,.beta.-unsaturated ketone compound. Still more preferably, no detectable amount of an .alpha.,.beta.-unsaturated ketone compound is present in the morphinan-6-one product.
Abbreviations and Definitions
[0102] The following definitions and methods are provided to better define the present invention and to guide those of ordinary skill in the art in the practice of the present invention. Unless otherwise noted, terms are to be understood according to conventional usage by those of ordinary skill in the relevant art.
[0103] The term "alkyl" as used herein describes groups which are preferably lower alkyl containing from one to eight carbon atoms in the principal chain and up to 20 carbon atoms. They may be straight or branched chain or cyclic and include methyl, ethyl, propyl, isopropyl, allyl, benzyl, hexyl and the like.
[0104] The term "alkenyl" as used herein describes groups which are preferably lower alkenyl containing from two to eight carbon atoms in the principal chain and up to 20 carbon atoms. They may be straight or branched chain or cyclic and include ethenyl, propenyl, isopropenyl, butenyl, isobutenyl, hexenyl, and the like.
[0105] The term "alkynyl" as used herein describes groups which are preferably lower alkynyl containing from two to eight carbon atoms in the principal chain and up to 20 carbon atoms. They may be straight or branched chain and include ethynyl, propynyl, butynyl, isobutynyl, hexynyl, and the like.
[0106] The term "aromatic" as used herein alone or as part of another group denotes optionally substituted homo- or heterocyclic aromatic groups. These aromatic groups are preferably monocyclic, bicyclic, or tricyclic groups containing from 6 to 14 atoms in the ring portion. The term "aromatic" encompasses the "aryl" and "heteroaryl" groups defined below.
[0107] The term "aryl" as used herein alone or as part of another group denote optionally substituted homocyclic aromatic groups, preferably monocyclic or bicyclic groups containing from 6 to 12 carbons in the ring portion, such as phenyl, biphenyl, naphthyl, substituted phenyl, substituted biphenyl or substituted naphthyl. Phenyl and substituted phenyl are the more preferred aryl.
[0108] The terms "halogen," "halide" or "halo" as used herein alone or as part of another group refer to chlorine, bromine, fluorine, and iodine.
[0109] The term "heteroatom" shall mean atoms other than carbon and hydrogen.
[0110] The terms "heterocyclo" or "heterocyclic" as used herein alone or as part of another group denote optionally substituted, fully saturated or unsaturated, monocyclic or bicyclic, aromatic or non-aromatic groups having at least one heteroatom in at least one ring, and preferably 5 or 6 atoms in each ring. The heterocyclo group preferably has 1 or 2 oxygen atoms and/or 1 to 4 nitrogen atoms in the ring, and is bonded to the remainder of the molecule through a carbon or heteroatom. Exemplary heterocyclo groups include heteroaromatics such as furyl, pyridyl, oxazolyl, pyrrolyl, indolyl, quinolinyl, or isoquinolinyl and the like. Exemplary substituents include one or more of the following groups: hydrocarbyl, substituted hydrocarbyl, hydroxy, protected hydroxy, acyl, acyloxy, alkoxy, alkenoxy, alkynoxy, aryloxy, halogen, amido, amino, cyano, ketals, acetals, esters and ethers.
[0111] The term "heteroaromatic" as used herein alone or as part of another group denote optionally substituted aromatic groups having at least one heteroatom in at least one ring, and preferably 5 or 6 atoms in each ring. The heteroaromatic group preferably has 1 or 2 oxygen atoms, 1 or 2 sulfur atoms, and/or 1 to 4 nitrogen atoms in the ring, and may be bonded to the remainder of the molecule through a carbon or heteroatom. Exemplary heteroaromatics include furyl, thienyl, pyridyl, oxazolyl, pyrrolyl, indolyl, quinolinyl, or isoquinolinyl and the like. Exemplary substituents include one or more of the following groups: hydrocarbyl, substituted hydrocarbyl, keto, hydroxy, protected hydroxy, acyl, acyloxy, alkoxy, alkenoxy, alkynoxy, aryloxy, halogen, amido, amino, nitro, cyano, thiol, ketals, acetals, esters and ethers.
[0112] The term "acyl," as used herein alone or as part of another group, denotes the moiety formed by removal of the hydroxy group from the group --COOH of an organic carboxylic acid, e.g., RC(O)--, wherein R is R.sup.1, R.sup.1O--, R.sup.1R.sup.2N--, or R.sup.1S--, R.sup.1 is hydrocarbyl, heterosubstituted hydrocarbyl, or heterocyclo, and R.sup.2 is hydrogen, hydrocarbyl or substituted hydrocarbyl.
[0113] The term "acyloxy," as used herein alone or as part of another group, denotes an acyl group as described above bonded through an oxygen linkage (--O--), e.g., RC(O)O-- wherein R is as defined in connection with the term "acyl."
[0114] The term "heteroaryl" as used herein alone or as part of another group denote optionally substituted aromatic groups having at least one heteroatom in at least one ring, and preferably 5 or 6 atoms in each ring. The heteroaryl group preferably has 1 or 2 oxygen atoms and/or 1 to 4 nitrogen atoms in the ring, and is bonded to the remainder of the molecule through a carbon. Exemplary heteroaryls include furyl, benzofuryl, oxazolyl, isoxazolyl, oxadiazolyl, benzoxazolyl, benzoxadiazolyl, pyrrolyl, pyrazolyl, imidazolyl, triazolyl, tetrazolyl, pyridyl, pyrimidyl, pyrazinyl, pyridazinyl, indolyl, isoindolyl, indolizinyl, benzimidazolyl, indazolyl, benzotriazolyl, tetrazolopyridazinyl, carbazolyl, purinyl, quinolinyl, isoquinolinyl, imidazopyridyl and the like. Exemplary substituents include one or more of the following groups: hydrocarbyl, substituted hydrocarbyl, hydroxy, protected hydroxy, acyl, acyloxy, alkoxy, alkenoxy, alkynoxy, aryloxy, halogen, amido, amino, cyano, ketals, acetals, esters and ethers.
[0115] The terms "hydrocarbon" and "hydrocarbyl" as used herein describe organic compounds or radicals consisting exclusively of the elements carbon and hydrogen. These moieties include alkyl, alkenyl, alkynyl, and aryl moieties. These moieties also include alkyl, alkenyl, alkynyl, and aryl moieties substituted with other aliphatic or cyclic hydrocarbon groups, such as alkaryl, alkenaryl and alkynaryl. Unless otherwise indicated, these moieties preferably comprise 1 to 20 carbon atoms.
[0116] The "substituted hydrocarbyl" moieties described herein are hydrocarbyl moieties which are substituted with at least one atom other than carbon, including moieties in which a carbon chain atom is substituted with a hetero atom such as nitrogen, oxygen, silicon, phosphorous, boron, sulfur, or a halogen atom. These substituents include halogen, heterocyclo, alkoxy, alkenoxy, aryloxy, hydroxy, protected hydroxy, acyl, acyloxy, nitro, amino, amido, nitro, cyano, ketals, acetals, esters and ethers.
[0117] The term "hydroxy protecting group" refers to hydrocarbyl and substituted hydrocarbyl moieties which bond to an hydroxy oxygen atom in a molecule so as to protect that oxygen atom from further reaction during synthesis. This protection allows reactions to occur selectively at another reaction site on the same molecule. Examples of hydroxy protecting groups include, but are not limited to, ethers such as methyl, t-butyl, benzyl, p-methoxybenzyl, p-nitrobenzyl, allyl, trityl, methoxymethyl, methoxyethoxymethyl, ethoxyethyl, tetra hydropyranyl, tetrahydrothiopyranyl, and trialkylsilyl ethers such as trimethylsilyl ether, triethylsilyl ether, dimethylarylsilyl ether, triisopropylsilyl ether and t-butyldimethylsilyl ether; esters such as benzoyl, acetyl, phenylacetyl, formyl, mono-, di-, and trihaloacetyl such as chloroacetyl, dichloroacetyl, trichloroacetyl, trifluoroacetyl; and carbonates including but not limited to alkyl carbonates having from one to six carbon atoms such as methyl, ethyl, n-propyl, isopropyl, n-butyl, t-butyl; isobutyl, and n-pentyl; alkyl carbonates having from one to six carbon atoms and substituted with one or more halogen atoms such as 2,2,2-trichloroethoxymethyl and 2,2,2-trichloroethyl; alkenyl carbonates having from two to six carbon atoms such as vinyl and allyl; cycloalkyl carbonates have from three to six carbon atoms such as cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl; and phenyl or benzyl carbonates optionally substituted on the ring with one or more C.sub.1-8 alkoxy, or nitro.
[0118] Having described the invention in detail, it will be apparent that modifications and variations are possible without departing the scope of the invention defined in the appended claims. Furthermore, it should be appreciated that all examples in the present disclosure are provided as non-limiting examples.
Example 1
[0119] In this Example, an oxycodone HCl sample was treated with a sulfur-containing compound according to the processes described herein.
[0120] To a 250 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 10 g of oxycodone HCl (0.028 moles; >0.3% by weight 14-hydroxycodeinone (14-OHC) impurity). Next, with mixing 100 g of deoxygenated water (10 minute N.sub.2 purge) was added. The solution pH was adjusted to about 6 with ammonium hydroxide. Next, 5.0 g of sodium dithionite (Na.sub.2S.sub.2O.sub.4) was added. The pH was then adjusted to about 7 with concentrated ammonium hydroxide. The resulting mixture was stirred at 70.degree. C. for about 16 hours.
[0121] After about 16 hours, the pH was adjusted to about 9 with ammonium hydroxide, precipitating the oxycodone base. The mixture was stirred for about 1 hour, and the precipitated oxycodone base was filtered, washed with water, and dried overnight at 40.degree. C. under reduced pressure.
[0122] The oxycodone base sample was converted to the oxycodone HCl salt by dissolving about 14.5 g of the oxycodone base in a 100 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control. Next, with mixing about 29 g of H.sub.2O and about 12.6 g of concentrated HCl was added. The resulting mixture was heated to about 65.degree. C.-75.degree. C. until substantially all was in solution. The heat was then removed, resulting in the precipitation of the oxycodone HCl salt. The precipitated mixture was stirred for about 1-3 hours at less than about 10.degree. C. and filtered to collect the precipitated oxycodone HCl.
[0123] The 14-hydroxycodeinone (14-OHC) content was analyzed in the oxycodone base sample and the oxycodone HCl sample using an Agilent HPLC with MS interface capability. The results are illustrated in Table 1.
TABLE-US-00001 TABLE 1 Initial 14- Final 14-OHC content OHC content Oxycodone base (% Oxycodone HCl (% by wt.) by wt.) (% by wt.) 0.3 0.0005 0.0005
Examples 2A-2G
[0124] In Examples 2A-2G, an oxycodone HCl sample was treated with a sulfur-containing compound according to the processes described herein. The treatment was performed at various temperatures, times of reaction, concentration of reactants, and pH.
Example 2A
[0125] To a 100 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 9.2 g of wet oxycodone HCl (0.02 moles; 0.13% by weight 14-hydroxycodeinone (14-OHC) impurity). Next, with mixing 36.2 g of H.sub.2O and 40.3 g of 6 wt. % SO.sub.2/H.sub.2O solution was added. The resulting mixture was heated to about 30.degree. C. and the solution pH was adjusted to about 6 with ammonium hydroxide. The mixture was stirred for about 3 hours. The pH of the mixture was then adjusted to about 8.8-9.8 with concentrated ammonium hydroxide and stirred for about 30 minutes. The precipitated oxycodone base was then filtered from the mother liquor, washed with about 25.73 g of H.sub.2O, and dried. The 14-hydroxycodeinone content (14-OHC) in the oxycodone base was then measured as described in the preceding Example.
[0126] The experiment was repeated using identical reagents, amounts thereof, and conditions to form the oxycodone base sample. This oxycodone base sample was converted to the oxycodone HCl salt as described in the preceding example. The 14-hydroxycodeinone content (14-OHC) in the oxycodone base sample and the oxycodone HCl sample were then measured.
[0127] Results and reaction conditions for this experiment are illustrated in Table 2.
TABLE-US-00002 TABLE 2 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 1 30 3 6 10.2 1.8:1 0.13 0.0007 Not tested 2 30 3 6 10.2 1.8:1 0.13 0.0007 0.0007
Example 2B
[0128] This Example was performed according to the process described in Example 2A. However, in this Example 9.4 g of wet oxycodone HCl (0.02 moles; 0.13% by weight 14-hydroxycodeinone (14-OHC) impurity) was mixed with about 34.6 g of H.sub.2O and about 27.4 g of 6 wt. % SO.sub.2/H.sub.2O solution. The mixture was heated to about 50.degree. C. Next, the pH was adjusted to about 7 using ammonium hydroxide.
[0129] The resulting mixture was allowed to react for either 1 hour or 5 hours. At the end of the desired reaction time, the solution was adjusted to a pH of 8.8-9.8 with about 2.0 g of concentrated ammonium hydroxide and stirred for about 30 minutes. The solids were filtered and washed with about 28.0 g of H.sub.2O and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed according to the method described in the preceding example. The results and reaction conditions in the various trials are illustrated in Table 3.
TABLE-US-00003 TABLE 3 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 3 50 1 7 8.2 1.2:1 0.13 None detected None detected 4 50 5 7 8.2 1.2:1 0.13 0.00005 0.0005
Example 2C
[0130] This Example was performed according to the process described in Example 2A. However, in this Example 9.1 g of wet oxycodone HCl (0.02 moles; 0.13-0.14% by weight 14-hydroxycodeinone (14-OHC) impurity) was mixed with about 7.0 g of H.sub.2O and about 52.8 g of 6 wt. % SO.sub.2/H.sub.2O solution. The mixture was heated to either 10.degree. C. or 50.degree. C. Next, the pH was adjusted to 7 using ammonium hydroxide.
[0131] The resulting mixture was allowed to react for either 1 hour or 5 hours. At the end of the desired reaction time, the solution was adjusted to a pH of 8.8-9.8 with about 2.0-2.5 g of concentrated ammonium hydroxide and stirred for about 30 minutes. The solids were filtered and washed with about 28.0 g of H.sub.2O and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed by the method described in the preceding example. The results and reaction conditions in the various trials are illustrated in Table 4.
TABLE-US-00004 TABLE 4 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 5 50 1 7 8.2 2.4:1 0.13 None detected 0.0006 6 50 5 7 8.2 2.4:1 0.13 0.00015 0.0004 7 10 5 7 8.2 2.4:1 0.14 0.001 Not tested
Example 2D
[0132] This Example was performed according to the process described in Example 2A. However, in this Example 9.52 g of wet oxycodone HCl (0.02 moles; 0.13% by weight 14-hydroxycodeinone (14-OHC) impurity) was mixed with about 72.24 g of H.sub.2O and about 27.76 g of 6 wt. % SO.sub.2/H.sub.2O solution. The mixture was heated to about 50.degree. C. Next, the pH was adjusted to about 7 using ammonium hydroxide.
[0133] The resulting mixture was allowed to react for either 1 hour or 5 hours. At the end of the desired reaction time, the solution was adjusted to a pH of 8.8-9.8 with about 2.0-2.5 g of concentrated ammonium hydroxide and stirred for about 30 minutes. The solids were filtered and washed with about 28.0 g of H.sub.2O and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed by the method described in the preceding example. The results and reaction conditions in the various trials are illustrated in Table 5.
TABLE-US-00005 TABLE 5 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 8 50 1 7 13.1 1.2:1 0.13 0.0002 0.0003 9 50 5 7 13.1 1.2:1 0.13 None detected 0.0004
Example 2E
[0134] This Example was performed according to the process described in Example 2A. However, in this Example 9.5 g of wet oxycodone HCl (0.02 moles; 0.13-0.14% by weight 14-hydroxycodeinone (14-OHC) impurity) was mixed with about 39.7 g of H.sub.2O and about 55.6 g of 6 wt. % SO.sub.2/H.sub.2O solution. The mixture was heated to either 10.degree. C. or 50.degree. C. Next, the pH was adjusted to about 7 using ammonium hydroxide.
[0135] The resulting mixture was allowed to react for either 1 hour or 5 hours. At the end of the desired reaction time, the solution was adjusted to a pH of 8.8-9.8 with about 2.0-2.5 g of concentrated ammonium hydroxide and stirred for about 30 minutes. The solids were filtered and washed with about 30.6 g of H.sub.2O and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed by the method described in the preceding example. The results and reaction conditions in the various trials are illustrated in Table 6.
TABLE-US-00006 TABLE 6 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 10 50 1 7 12.3 2.4:1 0.13 None detected 0.0004 11 50 5 7 12.3 2.4:1 0.13 None detected 0.0004 12 10 5 7 12.3 2.4:1 0.13 0.0008 Not tested
Example 2F
[0136] To a 22 L, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 1840 g of wet oxycodone HCl (4.27 moles: 0.13% by weight 14-hydroxycodeinone (14-OHC) impurity). Next, with mixing 2706 g of H.sub.2O and 7717 g of 6.4 wt. % SO.sub.2/H.sub.2O solution was added. The resulting mixture was heated to about 40.degree. C. and the solution pH was adjusted to about 7 using concentrated ammonium hydroxide. The mixture was stirred for about 5 hours.
[0137] After about 5 hours, the solution was adjusted to a pH of about 1.7 with the addition of 293.0 g concentrated sulfuric acid (96-98%). The pressure was slowly reduced to about 0.26 atm to facilitate the distillation/removal of unreacted SO.sub.2. As the distillation progressed, 23.4 g of concentrated sulfuric acid was added as the pressure was decreased to about 0.11 atm and the solution temperature was increased to about 50-55.degree. C.
[0138] The solution was then cooled to about 30.degree. C. and the solution pH adjusted to about 8.5-10 with concentrated ammonium hydroxide. The solution was stirred for about 30 minutes and filtered. The solids were filtered and washed with about 2000 g of H.sub.2O and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed by the method described in the preceding example. The results and reaction conditions are illustrated in Table 7.
TABLE-US-00007 TABLE 7 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 13 40 5 7 6.6 1.8:1 0.13 0.0001 0.0005
Example 2G
[0139] To a 50 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 3.33 g of oxycodone HCl (0.0095 moles; 0.2% by weight 14-hydroxycodeinone (14-OHC) impurity). Next, with mixing 33.3 g of H.sub.2O and 0.83 g of sodium bisulfite was added. The resulting mixture was heated to about 30.degree. C. and the solution pH was adjusted to about 7 with ammonium hydroxide. The mixture was stirred for about 15 hours. The pH of the mixture was then adjusted to about 8.8-9.8 with concentrated ammonium hydroxide and stirred for about 60 minutes. The precipitated oxycodone base was then filtered from the mother liquor, washed with about 10.0 g of H.sub.2O, and dried. The 14-hydroxycodeinone (14-OHC) content in the resulting oxycodone base was measured, as was the 14-hydroxycodeinone (14-OHC) content in the oxycodone HCl salt formed by the method described in the preceding example. The results and reaction conditions in the various trials are illustrated in Table 8.
TABLE-US-00008 TABLE 8 Concentration Molar Ratio Initial 14- Final 14-OHC content Temperature Time (g H.sub.2O per g of SO.sub.2 to OHC content Oxycodone Oxycodone Trial (.degree. C.) (hr.) pH Oxycodone HCl) Oxycodone HCl (% by wt.) base (% by wt.) HCl (% by wt.) 14 30 15 7 10 0.84:1 0.2 0.0004 0.0004
Example 3
[0140] In this Example, an oxymorphone HCl sample was treated with a sulfur-containing compound according to the processes described herein.
[0141] To a 250 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 150 g H.sub.2O and 15 g oxymorphone HCl sample (0.044 moles; 0.3-0.5% by weight 14-hydroxymorphinone (14-OHM) impurity). Next, 7.5 g of sodium bisulfite (NaHSO.sub.3) was added. The pH was then adjusted to about 7 with concentrated ammonium hydroxide, and the resulting mixture was stirred at 23.degree. C. for about 16 hours.
[0142] After about 16 hours, the pH was adjusted to about 8.8-9.8 with ammonium hydroxide and the solution was cooled to about 20.degree. C. The precipitated oxymorphone base was filtered, washed with water (about 45 g), and dried for 4 hours at 65.degree. C.
[0143] The oxymorphone base sample was analyzed using the methods described above, and the sample contained no detectable amount of 14-hydroxymorphinone or 14-hydroxycodeinone. This experiment was repeated using a 6 wt. % SO.sub.2/H.sub.2O solution in place of sodium bisulfite and similar results were obtained.
Example 4
[0144] In this Example, oxycodone base was treated with a thiol according to the processes described herein.
[0145] To a 25 ml, 3 neck round bottom flask equipped with a mechanical stirrer, N.sub.2 inlet, and thermocouple for temperature control was added 3.0 g oxycodone base (0.01 moles; 0.3-0.5% by weight 14-hydroxycodeinone (14-OHC) impurity). Next, 18 g of chloroform was added, and the mixture was stirred at 70.degree. C. until the oxycodone base was dissolved. After the mixture was substantially homogenous, 1.5 g of benzenethiol was added to the mixture with stirring.
[0146] After about 16 hours, a sample was analyzed using the methods described in the preceding examples. HPLC area percent analysis indicated a 14-hydroxycodeinone level of less than about 0.0022%.
* * * * *












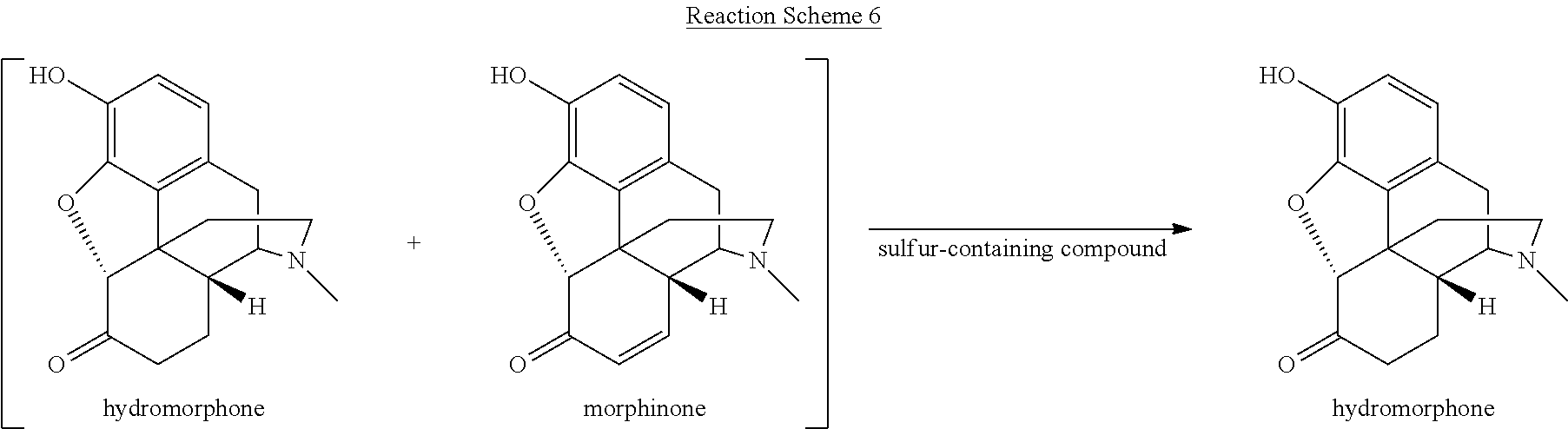
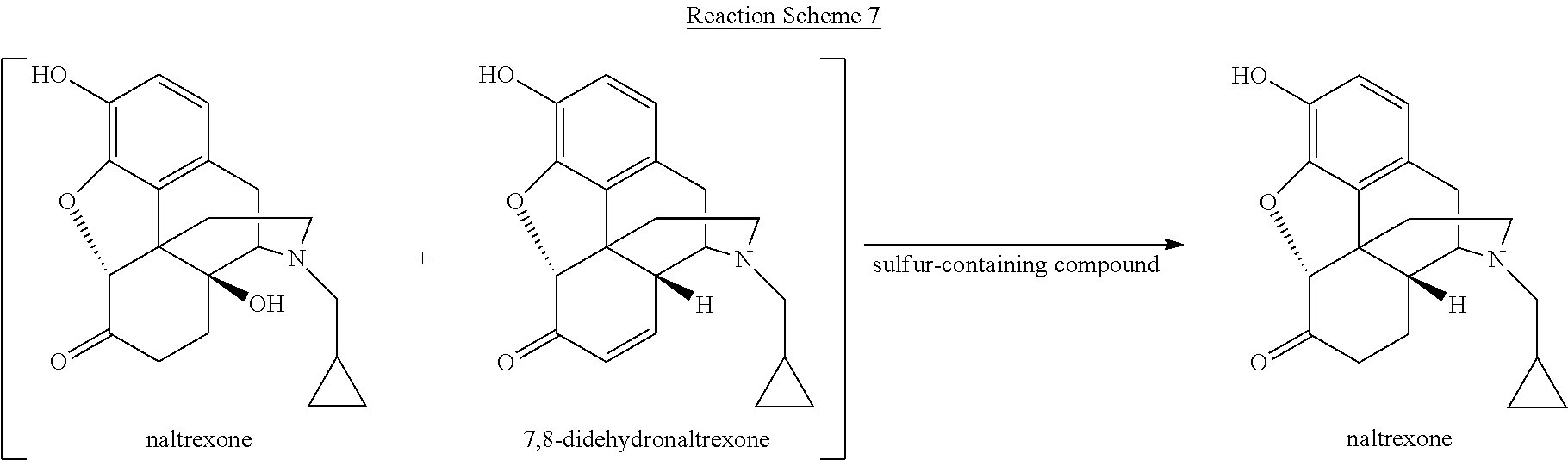
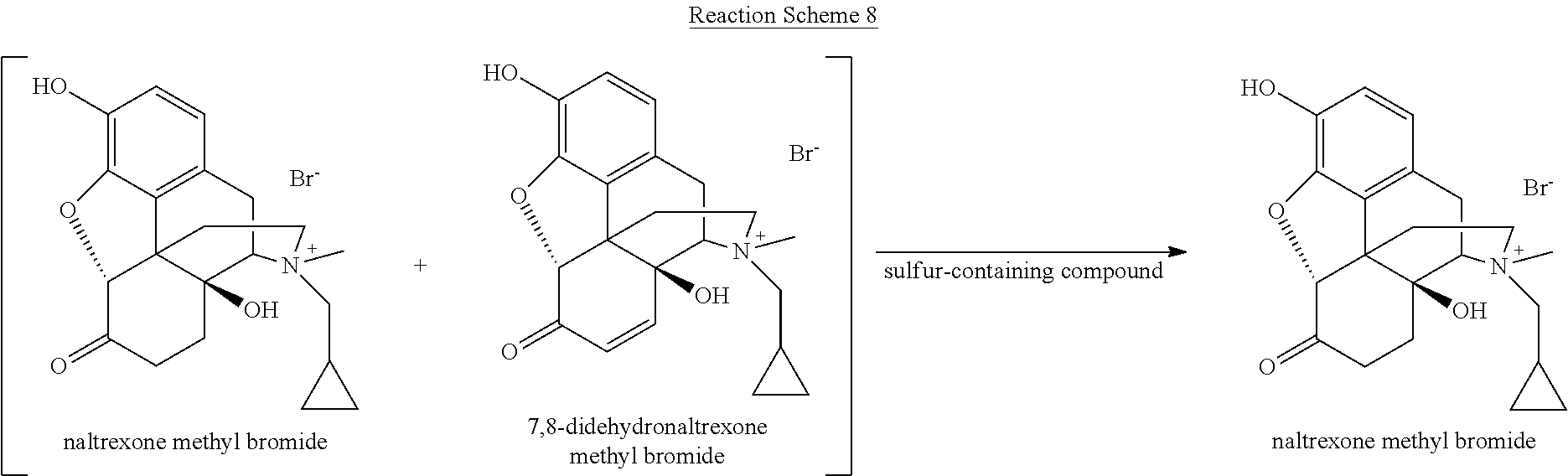
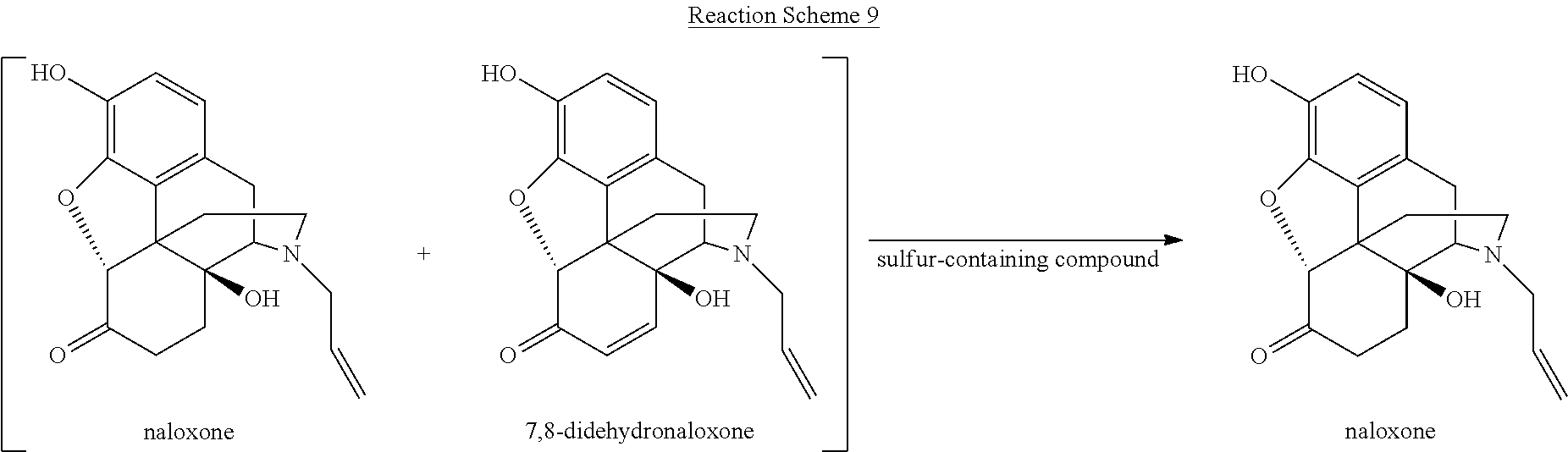
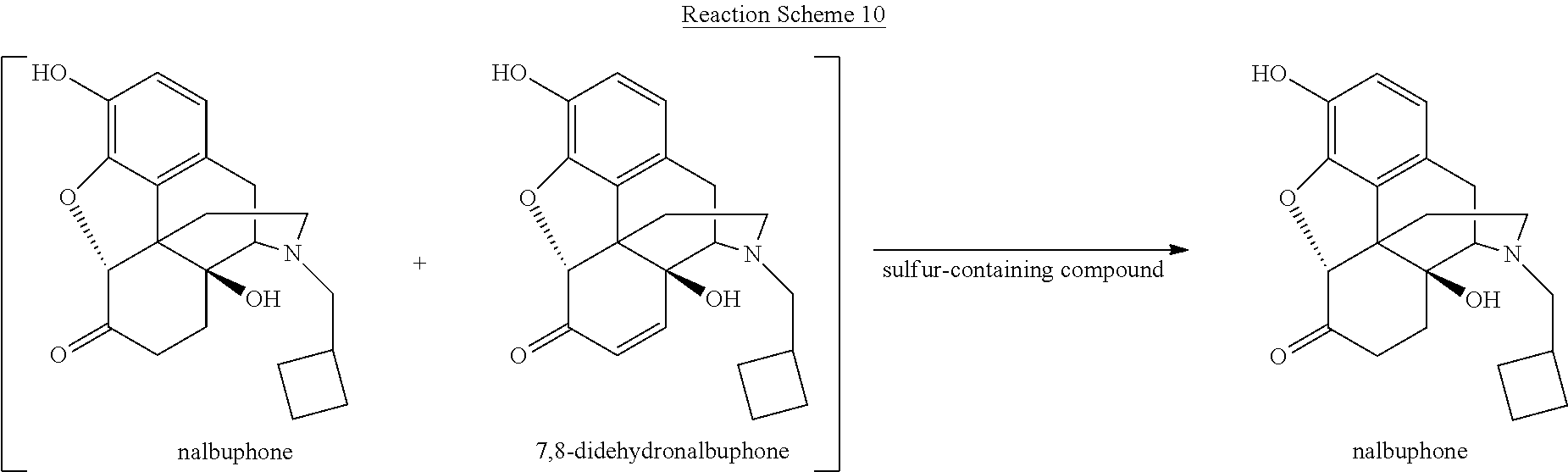
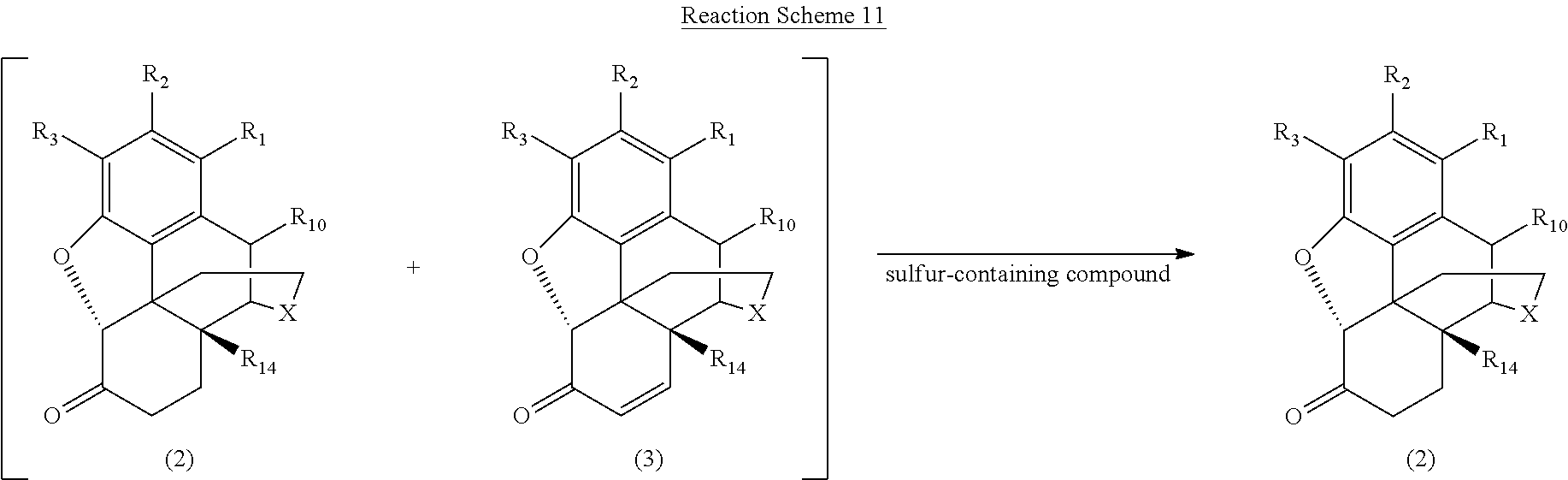
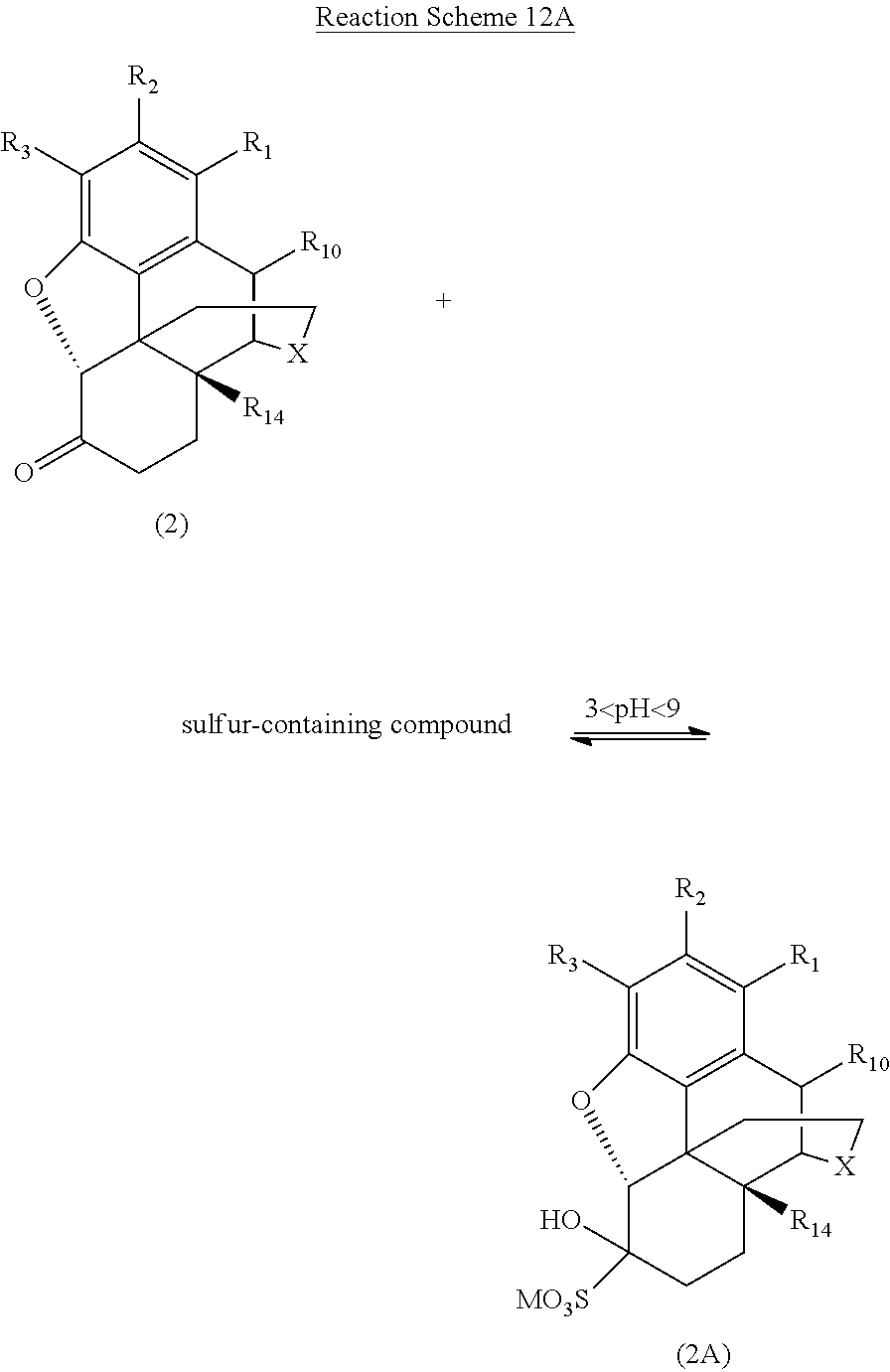
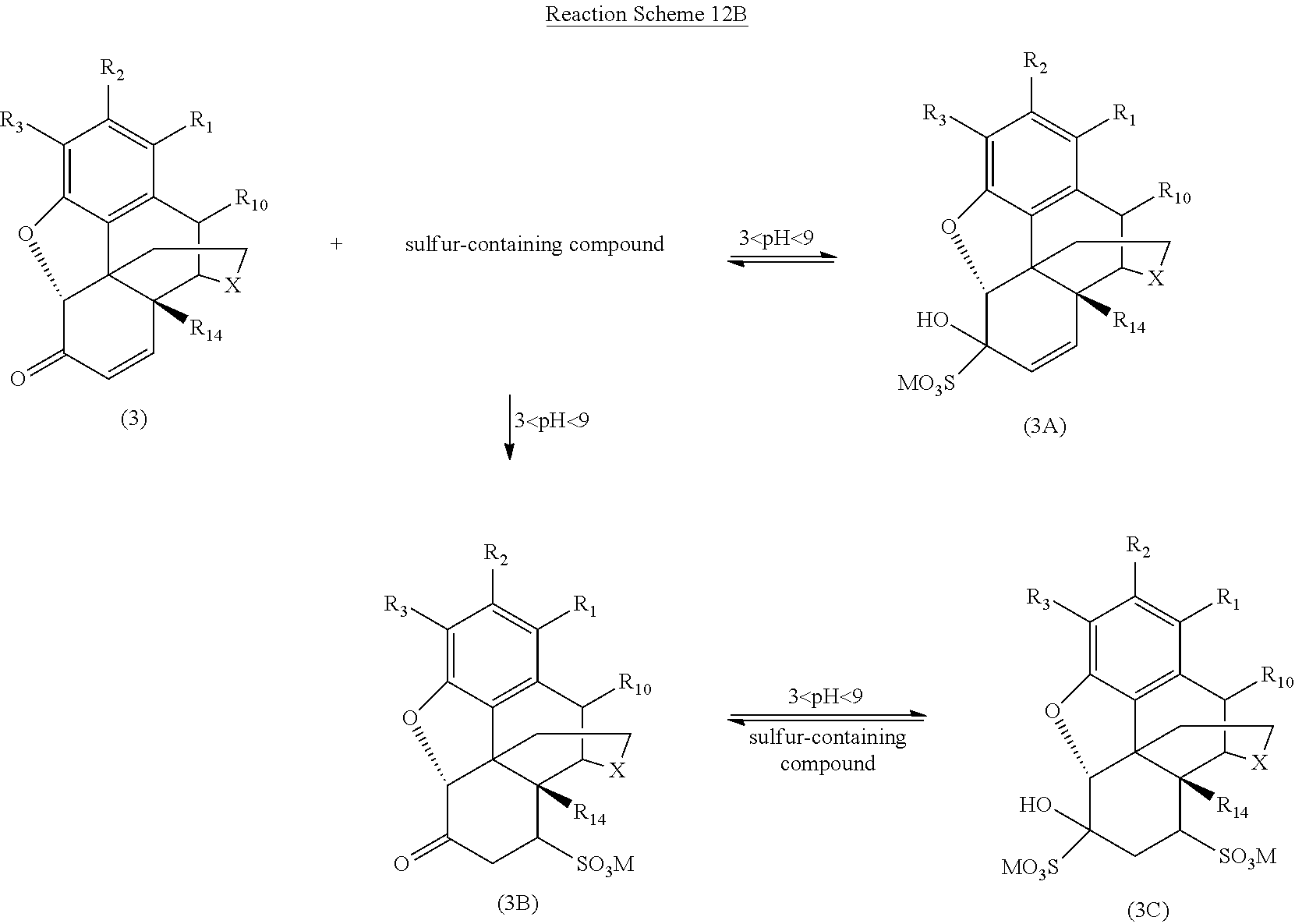
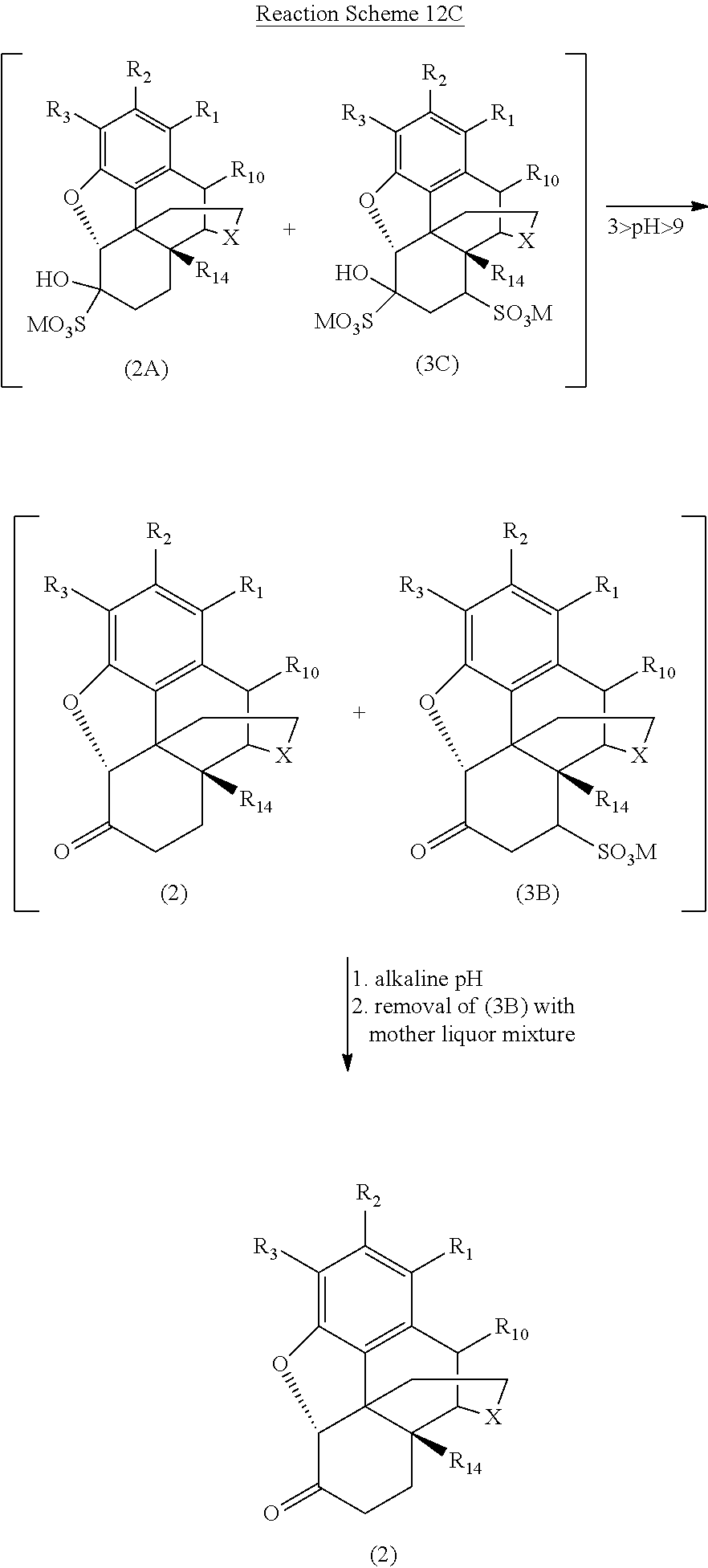
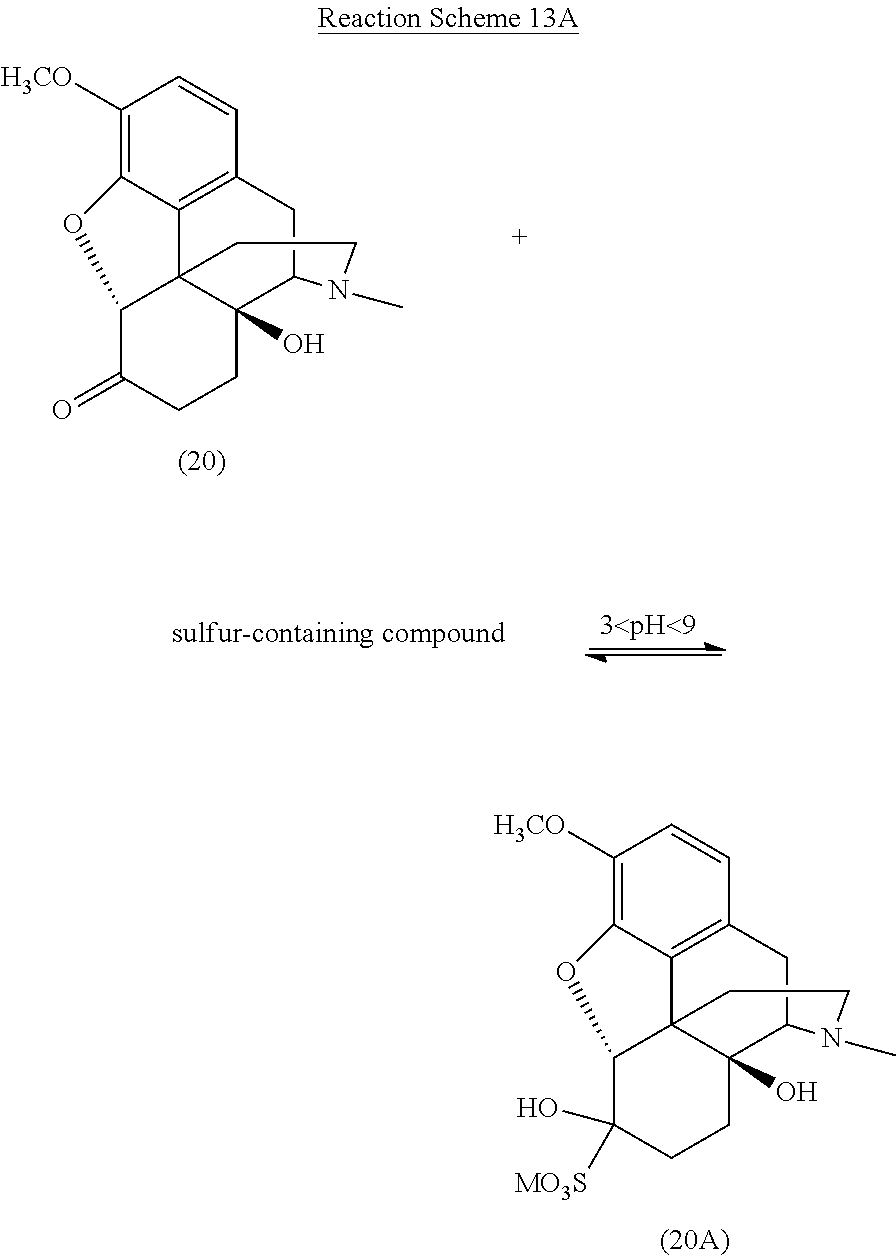
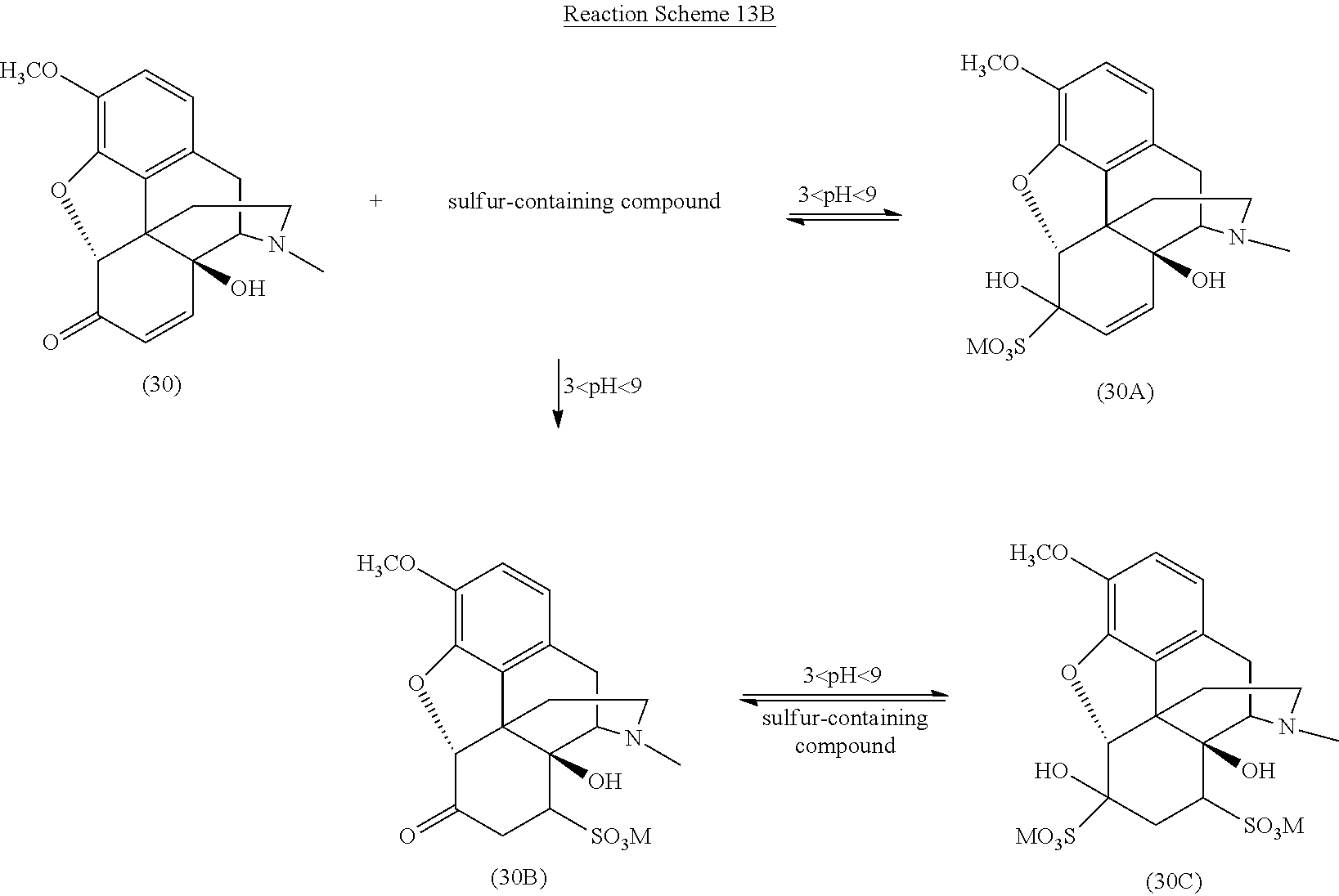
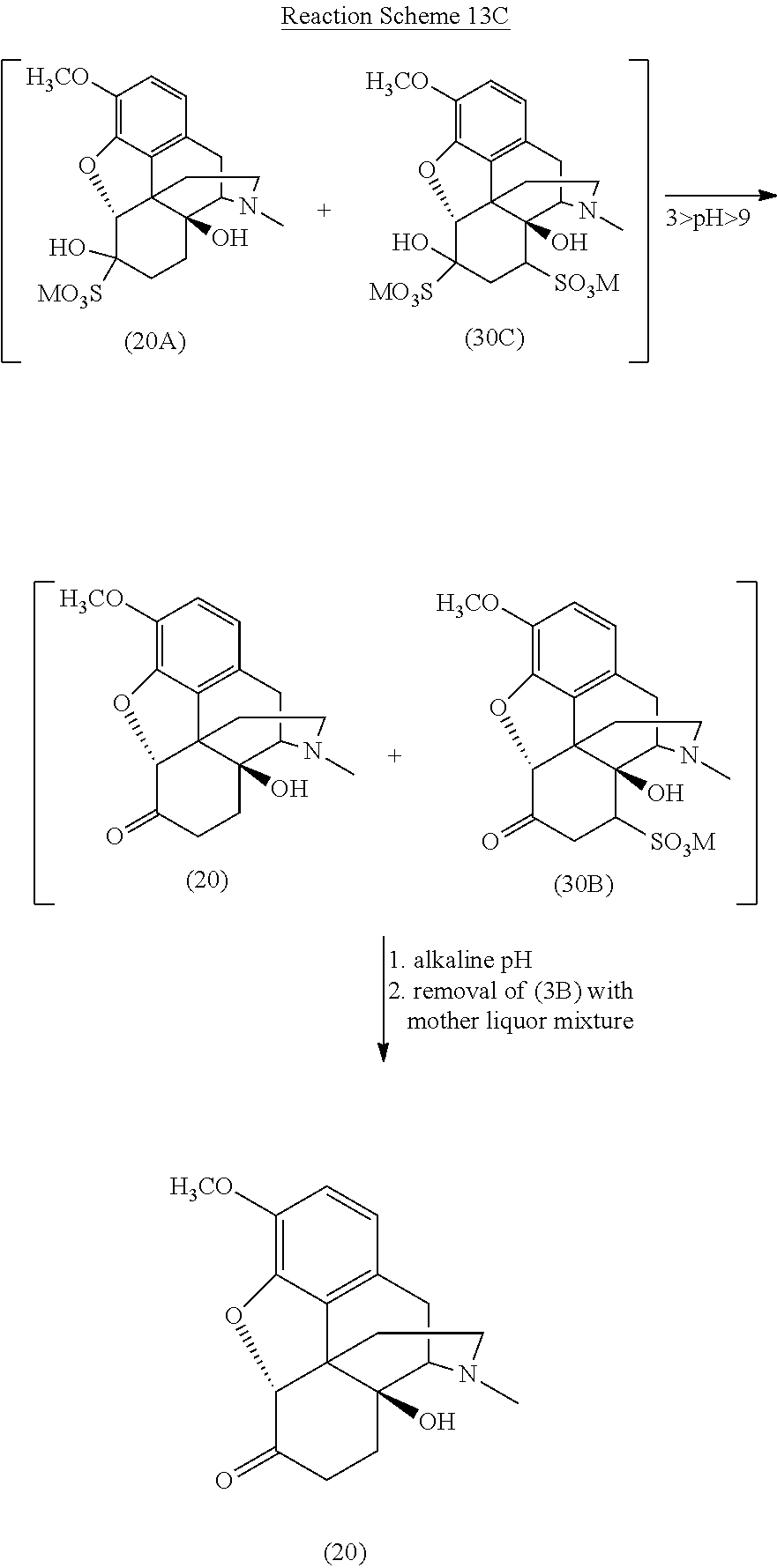
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.