Quench Hardened Steel
Liimatainen; Tommi
U.S. patent application number 16/480034 was filed with the patent office on 2019-12-05 for quench hardened steel. The applicant listed for this patent is SSAB TECHNOLOGY AB. Invention is credited to Tommi Liimatainen.
| Application Number | 20190368014 16/480034 |
| Document ID | / |
| Family ID | 61007713 |
| Filed Date | 2019-12-05 |





| United States Patent Application | 20190368014 |
| Kind Code | A1 |
| Liimatainen; Tommi | December 5, 2019 |
QUENCH HARDENED STEEL
Abstract
Disclosed is a hardenable steel composition, hardened steel components made from the same, and methods for forming the same. The steel contains relatively high levels of molybdenum in comparison to chromium, silicon and manganese, with the molybdenum providing an excellent combination of hardness and impact strength in the hardened steel. The steel can be cold formed then reheat-quench hardened with water as the quenching agent with no cracking and optionally no tempering. The steels formed have Vickers hardness levels of over 630 Hv10 (e.g. over 700 Hv10), and impact strengths of over 3 J/cm2 (e.g. over 20 J/cm2).
| Inventors: | Liimatainen; Tommi; (Stockholm, SE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 61007713 | ||||||||||
| Appl. No.: | 16/480034 | ||||||||||
| Filed: | January 26, 2018 | ||||||||||
| PCT Filed: | January 26, 2018 | ||||||||||
| PCT NO: | PCT/EP2018/051971 | ||||||||||
| 371 Date: | July 23, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C21D 1/56 20130101; C21D 8/0205 20130101; C22C 38/06 20130101; C22C 38/12 20130101; C21D 8/02 20130101; C21D 8/0263 20130101; C22C 38/04 20130101; C22C 38/02 20130101; C21D 1/18 20130101; C21D 1/60 20130101 |
| International Class: | C22C 38/12 20060101 C22C038/12; C21D 1/18 20060101 C21D001/18; C21D 1/60 20060101 C21D001/60; C21D 8/02 20060101 C21D008/02; C22C 38/04 20060101 C22C038/04; C22C 38/06 20060101 C22C038/06; C22C 38/02 20060101 C22C038/02 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jan 26, 2017 | EP | 17153320.1 |
| Feb 22, 2017 | EP | 17157301.7 |
Claims
1. A hardenable steel composition consisting of the following, in terms of weight percent: TABLE-US-00013 C 0.36-0.55%, Mn 0.1-0.7%, Si 0-0.7%, Mo 0.2-1.9%, Al .ltoreq.0.2% (not including 0%), Cr optionally .ltoreq.1%, Ni optionally .ltoreq.2%, Cu optionally .ltoreq.1%, Nb optionally .ltoreq.0.1%, Ti optionally .ltoreq.0.2%, V optionally .ltoreq.0.2%, B optionally .ltoreq.0.01%, W optionally .ltoreq.1%, and Co optionally .ltoreq.1%,
wherein % Mo.gtoreq.% Cr/2 (when Cr is present), % Mo.gtoreq.% Mn/2, and 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2 the balance being iron, residual contents and unavoidable impurities.
2. The hardenable steel composition of claim 1, wherein C is used at levels of 0.38-0.52%.
3. The hardenable steel composition of claim 1, wherein Si is used at levels of 0.01-0.5%.
4. The hardenable steel composition of claim 1, wherein Mn is used at levels of 0.10-0.50%.
5. The hardenable steel composition of claim 1, wherein Cr is used at levels of 0-0.5%.
6. The hardenable steel composition of claim 1, wherein Mo is used at levels of 0.2-1.5%.
7. The hardenable steel composition of claim 1, wherein % Mo.gtoreq.% Cr (when Cr is present) % Mo.gtoreq.% Mn, and 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2.
8. The hardenable steel composition of claim 1, wherein Ni is optionally present at levels of .ltoreq.1%.
9. The hardenable steel composition of claim 1, having the following composition: TABLE-US-00014 Al 0.01-0.1%, Ti optionally 0.001-0.05%, Nb optionally 0.001-0.05%, V optionally .ltoreq.0.15%, Cu optionally .ltoreq.0.1, Ni optionally .ltoreq.0.1, B 0-0.003%, W optionally .ltoreq.0.2%, Co optionally .ltoreq.0.2%, P .ltoreq.0.020%, S .ltoreq.0.020%, N .ltoreq.0.006%, H .ltoreq.0.0002%, O .ltoreq.0.005%, Ca .ltoreq.0.01, and REM .ltoreq.0.1%.
10. The hardenable steel composition of claim 1, wherein the steel contains Nb at levels of 0.005-0.02%.
11. A hardenable steel that is capable of being reheat-quench hardened to form a steel satisfying the following equation: V.sub.J+(H.sub.X*Hv10)>A.sub.X wherein V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., Hv10 is the Vickers Hardness, A.sub.X=95, and H.sub.X=0.125.
12. The hardenable steel of claim 11, wherein the steel has a composition as defined in claim 1.
13. A component formed from the hardenable steel composition of claim 1, wherein at least a region of the component has been reheat-quench hardened to form a hardened region.
14. The component of claim 13, wherein the Vickers hardness of the hardened region is .gtoreq.630 Hv10, and the Charpy V impact strength of the hardened region is .gtoreq.3 J/cm.sup.2 at 20.degree. C.
15. The component of claim 13, wherein the hardened region satisfies the following: V.sub.J+(H.sub.X*Hv10)>A.sub.X wherein V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., Hv10 is the Vickers Hardness, A.sub.X=95, and H.sub.X=0.125.
16. The component of claim 13, wherein the entire component has been reheat-quench hardened.
17. A method for producing hardened steel, comprising the steps of: a. providing a component formed from a hardenable steel composition as defined in claim 1; b. heating at least a region of the component to a temperature (T.sub.S) above A.sub.c3; c. quenching said region using a quenching fluid to form a hardened region; and d. optionally tempering the hardened region.
18. The method of claim 17, wherein the Vickers hardness of the hardened region is .gtoreq.630 Hv10, and the Charpy V impact strength of the hardened region is .gtoreq.3 J/cm.sup.2 at 20.degree. C.
19. The method of claim 17, wherein the hardened region satisfies the following: V.sub.J+(H.sub.X*Hv10)>A.sub.X wherein V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., Hv10 is the Vickers Hardness, A.sub.X=95, and H.sub.X=0.125.
Description
[0001] The present disclosure relates to a quench-hardenable steel that provides excellent cold formable properties and that can be reheat-quench hardened to provide a steel having excellent impact strength and hardness. The steel has a reduced tendency to hardening induced cracking even when using a very high cooling rate, such as water quenching, and without any tempering. The quench hardening can be carried out over the entire component formed from the steel, or only part of the component such as only at the edge of a blade. The disclosure also relates to a method of quench hardening steel.
BACKGROUND OF THE INVENTION
[0002] Quench hardening is a process in which steel and cast iron alloys are strengthened and hardened. During a typical reheat-quench hardening process, a steel is heated to an austenization temperature (preferably around 900.degree. C.), soaked (i.e. equalized), and then rapidly cooled (quenched) preferably with a liquid such as water or oil. Quenched steels are typically brittle due to an overabundance of martensite. In these cases, the steels can be tempered, i.e. heated to below the critical point (A.sub.c1) to reduce the hardness and increase the toughness. Tempering however also increases the processing steps and costs.
[0003] Quenching may be through the entire steel sample, or localised for example at a blade edge. Targeted heating means such as induction heating may be used if only localised or even surface hardening is required.
[0004] It is commonly understood that the hardness of the resulting martensite is dictated by the carbon content. However, higher carbon contents in turn increase the risk of crack formation during quench hardening. There are a multitude of factors that can give rise to crack formation, such as from thermal stresses arising due to uneven heating or quench cooling. This can particularly be an issue if a thick sample is quench hardened, as the temperature differential between the core and surface can cause significant thermal stresses through the material during the quenching process itself. Transformational stresses can also arise due to volume changes that occur when crystal phase transformations take place within the steel. This can particularly be a problem when the cooling rates are uneven, such as may also occur with thicker samples (e.g. above 6 mm in thickness) or with complex shapes. These localised concentrations of high residual stress may act as points of weakness that give rise to cracks if they are acting in the same direction as an applied load. Often, cracks originate and propagate from certain weak points, such as corners of square shaped holes punched out of a metal piece. This can lead to design restrictions for quench hardened components, as the component piece may need to be modified to avoid any such weak points occurring.
[0005] The quench medium itself can also influence the tendency for crack formation to occur. When water is used as the quench medium, the cooling rate is typically much higher in comparison to oil. While this gives rise to harder steels, the likelihood of stress cracking is higher. Transformational stresses can be reduced during tempering, but this inevitably reduces the hardness of the steel.
[0006] Impact strength is related to the ability of the material to dissipate the energy of an impact through its structure, which is a tendency favoured by softer, more malleable materials. Hardened steels may be extremely resistant to wear at their surface, but often this high hardness leads to a relatively brittle material with low impact strength. Even so, the combination of high hardness and good impact strength is highly desirable in some scenarios. For example, agricultural equipment such as ploughs are required to have high wear resistance and high hardness to allow them to withstand the abrasion they experience when in use. However, ploughs made from hardened materials with low impact strength may be at risk of damage when they encounter stones or other hard objects in the soil.
[0007] A reduced risk of hardening cracks is extremely important when the cooling rate is higher than standard water quenching, as can occur when using powerful stirring and/or when using salt water (brine) as the quenching bath.
[0008] There remains a need for quench hardenable steels that provide high hardness steels with good impact strength and with a reduced risk of hardening cracks. Furthermore, there remains a need for quench hardenable steels having good structural properties, which allow localised quench hardening particularly using water as the quench medium.
SUMMARY OF THE INVENTION
[0009] The present disclosure relates to a hardenable steel composition consisting of the following, in terms of weight percent:
TABLE-US-00001 C 0.36-0.55%, Mn 0.1-0.7%, Si 0-0.7%, Mo 0.2-1.9%, Al .ltoreq.0.2% (not including 0%), Cr optionally .ltoreq.1%, Ni optionally .ltoreq.2%, Cu optionally .ltoreq.1%, Nb optionally .ltoreq.0.1%, Ti optionally .ltoreq.0.2%, V optionally .ltoreq.0.2%, B optionally .ltoreq.0.01%, W optionally .ltoreq.1%, and Co optionally .ltoreq.1%,
wherein [0010] % Mo>% Cr/2 (when Cr is present) [0011] % Mo>% Mn/2, and 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2 the balance being iron, residual contents and unavoidable impurities.
[0012] Viewed in an alternative way, the present disclosure relates to a hardenable steel that is capable of being reheat-quench hardened to form a steel satisfying the following equation:
V.sub.J+(H.sub.X*Hv10)>A.sub.X [0013] wherein [0014] V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., [0015] Hv10 is the Vickers Hardness, [0016] A.sub.X=95, and [0017] H.sub.X=0.125.
[0018] Preferably, the hardenable steel is capable of being reheat-quench hardened from a temperature above A.sub.c3 to a temperature below M.sub.S at an average cooling rate of .gtoreq.50.degree. C./s (more preferably .gtoreq.80.degree. C./s) to form a crack-free hardened steel.
[0019] Preferably, the hardenable steel consists of the following, in terms of weight percent:
TABLE-US-00002 C 0.36-0.55%, Mn 0.1-0.7%, Si 0-0.7%, Mo 0.2-1.9%, Al .ltoreq.0.2% (not including 0%), Cr optionally .ltoreq.1%, Ni optionally .ltoreq.2%, Cu optionally .ltoreq.1%, Nb optionally .ltoreq.0.1%, Ti optionally .ltoreq.0.2%, V optionally .ltoreq.0.2%, B optionally .ltoreq.0.01%, W optionally .ltoreq.1%, and Co optionally .ltoreq.1%,
the balance being iron, residual contents and unavoidable impurities.
[0020] More preferably, the hardenable steel satisfies the following relationships: [0021] % Mo>% Cr (when present) [0022] % Mo>% Mn, and [0023] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2.
[0024] The present disclosure also relates to a component formed from the hardenable steel (or hardenable steel composition), wherein at least a region of the component has been reheat-quench hardened to form a hardened region.
[0025] Preferably, the entire component has been reheat-quench hardened.
[0026] Preferably, the microstructure of the hardened region of the component is martensitic.
[0027] Preferably, the hardened region of the component has a Vickers hardness of .gtoreq.630 Hv10.
[0028] Preferably, the hardened region of the component has a Charpy V impact strength of .gtoreq.3 J/cm.sup.2 at 20.degree. C.
[0029] Preferably, the hardened region satisfies the following:
V.sub.J+(H.sub.X*Hv10)>A.sub.X [0030] wherein [0031] V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., [0032] Hv10 is the Vickers Hardness, [0033] A.sub.X=95, and [0034] H.sub.X=0.125.
[0035] The present disclosure also relates to a method for producing hardened steel, comprising the steps of: [0036] a. providing a component formed from a hardenable steel composition; [0037] b. heating at least a region of the component to a temperature (T.sub.S) above A.sub.c3; [0038] c. quenching said region using a quenching fluid to form a hardened region; and [0039] d. optionally tempering the hardened region; wherein [0040] said hardenable steel composition consists of the following, in terms of weight percent,
TABLE-US-00003 [0040] C 0.36-0.55%, Mn 0.1-0.7%, Si 0-0.7%, Mo 0.2-1.9%, Al .ltoreq.0.2% (not including 0%), Cr optionally .ltoreq.1%, Ni optionally .ltoreq.2%, Cu optionally .ltoreq.1%, Nb optionally .ltoreq.0.1%, Ti optionally .ltoreq.0.1%, V optionally .ltoreq.0.2%, B optionally .ltoreq.0.01%, W optionally .ltoreq.1%, and Co optionally .ltoreq.1%,
wherein [0041] % Mo>% Cr/2 (when Cr is present) [0042] % Mo>% Mn/2, and [0043] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2 the balance being iron, residual contents and unavoidable impurities.
[0044] Viewed in an alternative way, the present disclosure relates to a method for producing hardened steel, comprising the steps of: [0045] a'. providing a component formed from a hardenable steel; [0046] b'. heating at least a region of the component to a temperature (T.sub.S) above A.sub.c3; [0047] c'. quenching said region using a quenching fluid to form a hardened region; and [0048] d'. optionally tempering the hardened region; wherein [0049] the hardened region satisfies the following:
[0049] V.sub.J+(H.sub.X*Hv10)>A.sub.X [0050] wherein [0051] V.sub.J is the Charpy V impact strength in J/cm.sup.2 at 20.degree. C., [0052] Hv10 is the Vickers Hardness, [0053] A.sub.X=95, and [0054] H.sub.X=0.125.
[0055] Preferably, in step b. (or step b'.) the entire component is heated to a temperature above A.sub.c3, and more preferably in step c. (or step c'.) the entire component is then quenched with the quenching fluid to form a hardened component.
[0056] Preferably, the hardenable steel in step a'. consists of the following, in terms of weight percent:
TABLE-US-00004 C 0.36-0.55%, Mn 0.1-0.7%, Si 0-0.7%, Mo 0.2-1.9%, Al .ltoreq.0.2% (not including 0%), Cr optionally .ltoreq.1%, Ni optionally .ltoreq.2%, Cu optionally .ltoreq.1%, Nb optionally .ltoreq.0.1%, Ti optionally .ltoreq.0.2%, V optionally .ltoreq.0.2%, B optionally .ltoreq.0.01%, W optionally .ltoreq.1%, and Co optionally .ltoreq.1%,
the balance being iron, residual contents and unavoidable impurities.
[0057] More preferably, the hardenable steel in step a'. satisfies the following relationships: [0058] % Mo>% Cr/2 (when Cr is present) [0059] % Mo>% Mn/2, and [0060] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2.
[0061] Preferably, the steel is crack-free after step c. (or step c'.).
[0062] Preferably, the cooling rate in step c. (or c'.) is .gtoreq.50.degree. C./s, more preferably .gtoreq.80.degree. C./s.
BRIEF DESCRIPTION OF THE DRAWINGS
[0063] The present disclosure will hereinafter be further explained by means of non-limiting examples with reference to the appended figures where;
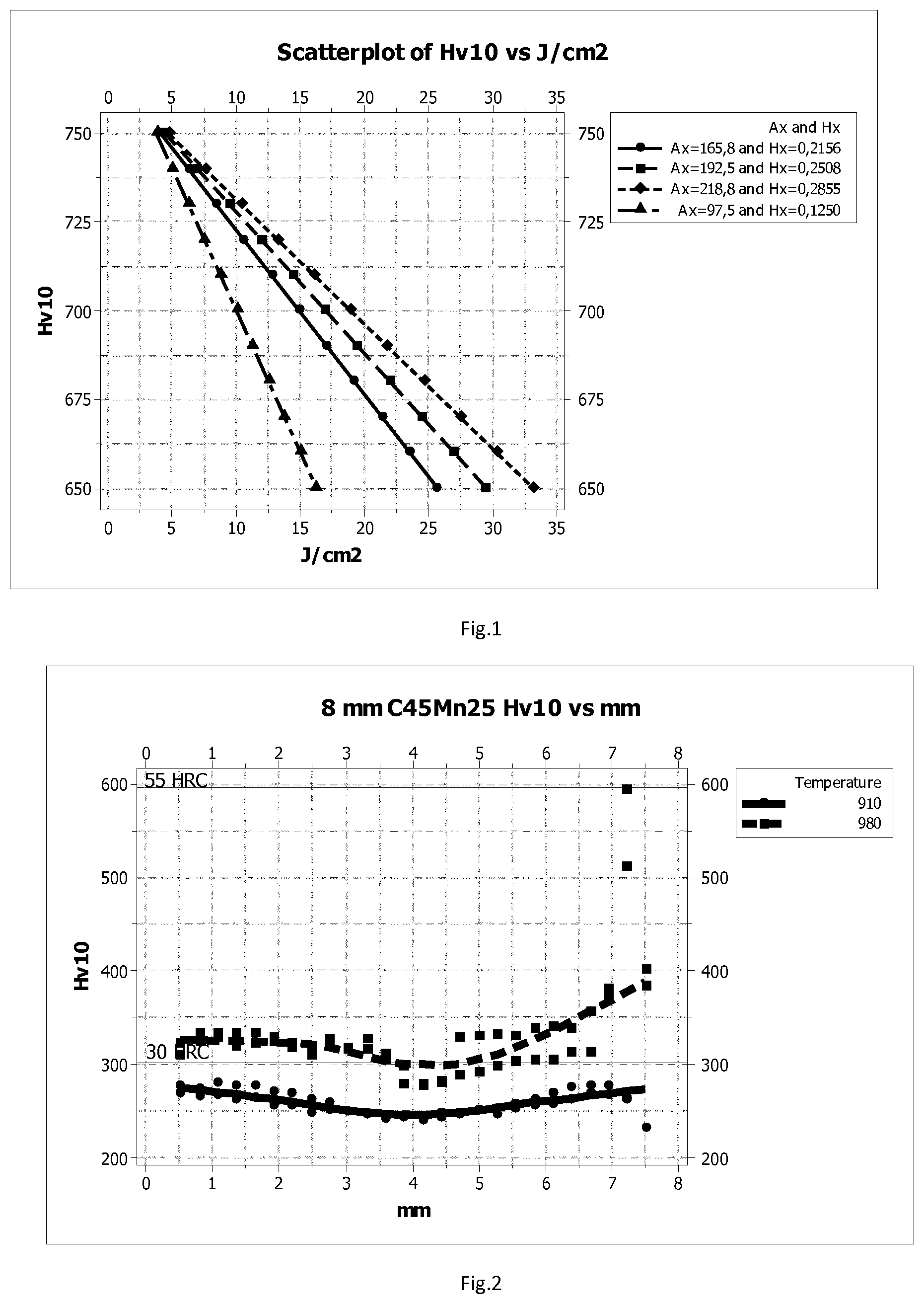
[0064] FIG. 1 shows a scatter plot of Vickers hardness and impact strength for varying A.sub.X values;
[0065] FIG. 2 shows the hardness vs depth profile of steel 1-A;
[0066] FIG. 3 shows the hardness vs depth profile of steel 1-B;
[0067] FIG. 4 shows the hardness vs depth profile of steel 1-C;
[0068] FIG. 5 shows the hardness vs depth profile of steel 1-D;
[0069] FIG. 6 shows the hardness vs depth profile of steel 1-D, 12 mm sample;
[0070] FIG. 7 shows the effect of tempering on hardness for steel 1-D;
[0071] FIG. 8 shows the effect of tempering on impact strength for steel 1-D; and
[0072] FIG. 9 shows the blank used in the crack resistance testing.
[0073] FIG. 10 shows the median hardness values of the test steels of Example 5.
DETAILED DESCRIPTION OF THE INVENTION
[0074] The hardenable steel (composition) of the present disclosure provides excellent quench hardening characteristics with reduced risk for quench cracking compared to standard hardenable steels such as 51CrV4, 38MnB5, 42MnV7, and the like. The steel is able to provide hardness levels of >630 Hv10 when quench hardened using water, and can typically be used even without tempering (i.e. the quench hardened steel is crack-free without tempering). The excellent formability and structural properties of the steel prior to quench hardening allows it to be hot or cold formed into a range of components, quench hardened (optionally locally) to form a martensitic steel using a simple and cost effective process, and then used without any additional tempering steps.
[0075] By "hardenable steel (composition)" is meant the hardenable steel composition set out above, as well as the hardenable steel capable of being reheat-quench hardened to form a steel satisfying the equation V.sub.J+(H.sub.X*Hv10)>A.sub.X as set out above. The preferred aspects described herein, such as the preferred structural limitations or properties of the steel as well as the preferred processing conditions, apply to both the hardenable steel as well as the hardenable steel composition.
[0076] By "crack-free" is meant that the steel meets ISO 23278:2015, Class 2X. The ISO23278:2015 standard specifies acceptance levels for indication from imperfections in ferromagnetic steel welds detected by magnetic particle testing. This tolerance level means that, when tested using magnetic particle detection according to the ISO23278:2015 standard, any linear imperfections such as cracks/defects are below 1.5 mm in length, and non-linear imperfections such as cracks/defects are below 3 mm in maximum dimension. In this context, a linear imperfection is defined as having a length greater than three times its width, while a non-linear imperfection is defined as having a length less than or equal to three times its width.
[0077] Preferably, by "crack-free" is meant that the steel meets ISO23278:2015, Class 2X when held at room temperature for 24 hours, more preferably for 1 week, after quench hardening.
[0078] As used herein, the terms "martensite" and "martensitic" include martensite, self-tempered (or auto-tempered) martensite and tempered martensite. Thus, within the meaning of this disclosure a steel containing .gtoreq.95% martensite has .gtoreq.95% of is structure being martensite, self-tempered martensite and tempered martensite.
[0079] As used herein, A.sub.c1 is the temperature at which austenite (.gamma.) begins to form during heating.
[0080] As used herein, A.sub.r1 is the temperature at which austenite (.gamma.) to ferrite (.alpha.) transformation is completed during cooling.
[0081] As used herein, A.sub.c3 is the temperature at which transformation of ferrite (.alpha.) to austenite (.gamma.) is completed during heating.
[0082] As used herein, A.sub.r3 is the temperature at which austenite (.gamma.) begins to transform to ferrite (.alpha.) during cooling.
[0083] A.sub.c1, A.sub.c3, A.sub.r1, A.sub.r3 are well known parameters that would be known to the skilled person. They may be estimated experimentally using standard methodology. Alternatively, the parameters may be estimated theoretically, for example using the formulae from Brandis (Rechnerische Bestimmung der Umwandlungstemperaturen von niedriglegierten Stahlen. TEW--Technische Berichte, Band 1, Heft 1, 1975, 8-10) or Lutsenko (The Definition and Use of Technological Reserves--An Effective Way to Improve the Production Technology of Rolled Metal. Abschlu bericht, Kommission der Europaischen Gemeinschaften, Luxembourg, 1991, 136 p.), as set out below:
A.sub.c1=739-22*[% C]-7*[% Mn]+2*[% Si]+14*[% Cr]+13*[% Mo]-13*[% Ni]+20*[% V]
A.sub.c3=902-255*[% C]-11*[% Mn]+19*[% Si]-5*[% Cr]+13*[% Mo]-20*[% Ni]+55*[% V]
A.sub.r1=741.7-7.13*[% C]-14.09*[% Mn]+16.26*[% Si]+11.54*[% Cr]-49.69*[% Ni]
A.sub.r3=913.7-207.13*[% C]-46.6*[% Mn]+110.54*[% Cr]+108.1*[% N]
[0084] Where [% X] is the weight percent of element X.
[0085] As used herein, the term "hardenable" preferably means that a sample of the steel with a thickness of 3 mm, when heated to and soaked at 950.degree. C. for a 1 hour and then quenched with still water, has at least 90% martensitic microstructure at the centre of the sample.
[0086] The key characterising features of the hardenable steel composition of the disclosure is the relatively low Mn, Si and Cr content in combination with the relatively high Mo content. The combination of these elements in these ratios provides a steel having good structural properties that displays exceptional impact strength and quench hardenability combined with the low risk of quench induced cracking.
[0087] The chemical composition of the hardenable steel (composition) is now described in more detail. In this disclosure, all percentages in the compositions are percentages by weight.
[0088] Moreover, each of the preferred ranges for the various components mentioned below may be combined with any the preferred ranges of the remaining components.
[0089] Carbon
[0090] Carbon is primarily present to ensure the hardenable steel (composition) is capable of quench hardening to form martensite. A higher carbon content will provide a harder steel. However, if the carbon content is too high, the resultant quenched steel can become too brittle and the risk of quench induced cracking increases. A maximum carbon content of 0.55% is therefore preferably used.
[0091] Preferably, carbon is used at levels of 0.38-0.52%, more preferably 0.40-0.50%, even more preferably 0.41-0.48%.
[0092] Silicon
[0093] Silicon is included in steels to improve cleanliness during the smelt processing and it can have a positive effect on hardenability. However, too much silicon can reduce the amount of self-tempering that occurs during quenching, as well as have an impact on the surface quality of the finished steel. A maximum silicon content of 0.7 is therefore preferably used.
[0094] Preferably, silicon is used at levels of 0.01-0.5%, more preferably 0.05-0.35%, most preferably 0.10-0.25%.
[0095] Manganese, Chromium and Molybdenum
[0096] Manganese, chromium and molybdenum play an important role in controlling the quench-hardening properties of the hardenable steel (composition). The importance of these three elements is set out below.
[0097] Manganese has a significant effect on hardenability. It is also effective at sequestering any oxygen and sulphur that may be present in the steel. However, manganese has a strong tendency to segregate and also significantly reduces the martensite start temperature, M.sub.S. The overall manganese content is therefore quite low, with a preferable maximum amount of 0.7%, preferably a maximum of 0.65%.
[0098] Preferably, manganese is used at levels of 0.10-0.50%, more preferably 0.15-0.40%, most preferably 0.20-0.30%.
[0099] Chromium is optionally used to increase hardenability. However, it also significantly reduces the martensite start temperature, which can negatively affect the amount of self-tempering that occurs during quenching. The total chromium content is therefore preferably less than 1%, preferably less than 0.75%.
[0100] Preferably, chromium is used at levels of 0-0.5%, more preferably 0-0.3%, most preferably 0-0.20%. Molybdenum is included to provide hardenability and improve tempering resistance.
[0101] Advantageously, molybdenum does not have a significant impact on the martensite start temperature, meaning that the use of molybdenum as the primary hardening agent ensures the martensite start temperature remains high and self-tempering is promoted during quenching.
[0102] Molybdenum also helps improve the impact strength of the hardened steel composition. It is well known that impact strength increases with increasing temperature, with lower temperature materials being more brittle and higher temperature materials being more ductile. The plot of impact strength against temperature for steels is therefore typically a sigmoidal shape, with a relatively steep transition region where the steel changes from brittle to ductile behaviour and impact strength increases significantly.
[0103] For martensitic steels, the presence of alloying elements typically lowers the impact strength. However, the applicant has found that molybdenum and nickel frequently increase the impact strength of the steel, effectively lowering the temperature at which the steel transitions from brittle to ductile behaviour, with the effect from molybdenum being particularly significant. Molybdenum also helps retain hardness during tempering at relatively high temperatures, such as from 500-650 or more preferably from 500 to 600.degree. C.
[0104] Preferably, molybdenum is used at levels of 0.2-1.9%. Preferably, molybdenum is used at levels of 0.2-1.5%, more preferably 0.30-1.2%, more preferably 0.40-1.1%, even more preferably 0.50-1.0%.
[0105] The combination of manganese, chromium and molybdenum provide the hardenability in the hardenable steel (composition). However, molybdenum is used in favour of the other two to provide a steel with high hardenability and high impact strength, that retains a high martensite start temperature, and which is more able to retain hardness and impact strength during tempering after hardening.
[0106] Manganese, chromium and molybdenum respectively have decreasing influence on the hardenability, with comparably more chromium and often even more molybdenum being needed as compared to manganese to achieve the same hardenability. Consequently, the hardenable steel (composition) disclosed herein preferably contains a relatively higher level of molybdenum in comparison to chromium and manganese, and preferably satisfies the following conditions: [0107] % Mo.gtoreq.% Cr/2 (when Cr is present) [0108] % Mo.gtoreq.% Mn/2, and [0109] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2
[0110] Preferably: [0111] Mo.gtoreq.% Cr (when Cr is present) [0112] % Mo.gtoreq.% Mn, and [0113] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2
[0114] Preferably: [0115] % Mo.gtoreq.% Cr+% Mn, and [0116] 0.4.ltoreq.% Mn+% Cr+% Mo.ltoreq.2
[0117] Preferably: [0118] % Mo.gtoreq.% Cr+2*% Mn.
[0119] Moreover, preferably: [0120] 0.5.ltoreq.% Mn+% Cr+% Mo.ltoreq.1.5,
[0121] More preferably: [0122] 0.85.ltoreq.% Mn+% Cr+% Mo.ltoreq.1.35.
[0123] The steel preferably contains enough manganese, chromium and molybdenum to ensure a good level of hardenability. Typically, the hardenability is sufficient if the carbon equivalent, C.sub.eq, is >0.60, preferably >0.62, wherein:
C.sub.eq=% C+% Mn/6+(% Cu+% Ni)/15+(% Cr+% Mo+% V)/5+% W/10
[0124] If the steel contains niobium, then the hardenable steel (composition) preferably displays sufficient hardenability if the following relationship is satisfied:
X.sub.H.gtoreq.3.5, preferably
X.sub.H.gtoreq.4,
wherein
X.sub.H=(1+(2.13*% Mo))*(1+(2.4*% Cr))*(1+(3.3*% Mn))
[0125] Aluminium
[0126] Aluminium is used as a deoxidation (killing) agent. However, too high levels of aluminium should be avoided, as it can impact on the hardenability/self-tempering properties of the steel. Consequently, the preferable maximum aluminium levels are 0.2%.
[0127] Preferably, aluminium is used in the range 0.01-0.1%, more preferably from 0.01-0.06%, most preferably from 0.015-0.045%. These low aluminium levels are particularly preferred when good electric resistance welding properties are desired.
[0128] Niobium, Titanium and Vanadium
[0129] Niobium and titanium control the texture of the steel after hot-rolling, and prevent grain growth during the heating step prior to quenching. They are therefore helpful in controlling the retention of properties of the hot-rolled steel during quench hardening. However, excessive amounts can lead to precipitates forming, such as large titanium nitrides which can negatively affect impact strength.
[0130] Generally, steels with a larger grain size have better hardenability. However, larger grain sizes will increase the tendency for cracking during quench hardening. Titanium and niobium reduce the rate of grain size growth at the austenising temperature, which can reduce hardenability. This effect can be compensated for by ensuring that sufficient alloying elements to obtain good hardenability are included in the steel, for instance by ensuring that X.sub.H is suitably high. When suitably formulated, control over grain coarsening provided by titanium and niobium (particularly niobium) during austenisation can be advantageous, allowing longer austenisation soak times (T.sub.S) and/or higher temperature without significantly increasing the risk of hardening induced cracking.
[0131] This makes it possible to combine, for example, hot forming, stamping and hardening in one process step (i.e. hot press-forming or die quench method), making production more cost effective. When hot press-forming, the steel can cool down rapidly during transfer to the die and the hot forming die, for instance at rates of around 20.degree. C./s. Due to this, it is usually necessary to use a very high start temperature, to ensure the steel remains at a suitable temperature (e.g. above the austenite temperature A.sub.13) until quenching.
[0132] Preferably, titanium is present in the range of 0-0.1%, more preferably 0.001-0.05%, more preferably 0.005-0.02%.
[0133] Preferably, Ti/N<3.42.
[0134] If boron is not alloyed in the steel composition, it is desirable to ensure that Ti/N.ltoreq.3.42. This will ensure a higher amount of small TiN precipitates, so as to ensure the advantages of titanium may be obtained without a significant deterioration of the impact strength.
[0135] Preferably, niobium is present in the range of 0.001-0.05%, more preferably 0.005-0.02%, most preferably 0.008-0.015%.
[0136] Preferably, the hardenable steel (composition) contains at least one of titanium or niobium.
[0137] Preferably, the hardenable steel (composition) contains niobium.
[0138] Niobium can affect the ductility of the steel, reducing its cold-formability. If very demanding cold forming is needed prior to reheat-quenching, the hardenable steel (composition) preferably has a niobium content of <0.005%.
[0139] Vanadium increases hardenability, but vanadium carbides can be difficult to dissolve during austenisation prior to quench hardening. These carbides can act as nuclei for ferrite crystals to grow during quenching. Consequently, vanadium content should preferably be limited to .ltoreq.0.2%. Preferably, vanadium is optionally present at levels of .ltoreq.0.15%, more preferably .ltoreq.0.10%, more preferably .ltoreq.0.06%, most preferably .ltoreq.0.04%.
[0140] Due to the difficulty of dissolving vanadium carbides, these lower vanadium contents are particularly preferred when the soak temperature (T.sub.S) is low and soak time (t.sub.S) is relatively short.
[0141] Copper and Nickel
[0142] As noted above, nickel has a positive influence on the impact strength of the martensitic steel. However, too much nickel can have an impact on the temper resistance of the steel, i.e. steels with too much nickel have been found to lose hardness during tempering. Nickel also reduces the M.sub.S, which has an impact on the self-tempering properties of the steel.
[0143] In view of this, nickel is typically optionally present at .ltoreq.1%.
[0144] Nickel is preferably optionally present at .ltoreq.0.4%, more preferably .ltoreq.0.1%.
[0145] Copper is preferably optionally present at .ltoreq.0.4%, more preferably .ltoreq.0.1%.
[0146] Preferably, Cu+Ni.ltoreq.0.4%, more preferably .ltoreq.0.1%.
[0147] Copper has a tendency to phase separate and migrate to the surface (under the scale) during processing above the melt temperature of copper, which can be harmful to the surface quality when the material is hot formed. Nickel mitigates this effect, and as such the Ni content is desirably at least 0.33*% Cu, preferably at least 0.50*% Cu, when the % Cu.gtoreq.0.2.
[0148] Boron, Tungsten and Cobalt
[0149] Boron and tungsten can improve hardenability. However, they are typically not needed as the hardenability of the steel is primarily provided by other elements. Moreover, boron has a smaller effect on the hardenability of steels with higher carbon levels, such as those disclosed herein. In order for boron to have an effect on higher carbon steels, typically the nitrogen level must be low and/or the titanium content must be high enough, which increases the likelihood of TiN precipitates forming. Boron is therefore not harmful to the steel, it is simply not essential to provide the desired balance of properties if there are not any coarse TiN precipitates present. Cobalt is very expensive and typically unnecessary.
[0150] Preferably, boron is present at levels of 0-0.003%, more preferably 0-0.0005%, even more preferably 0-0.0002%.
[0151] Preferably, tungsten is present a .ltoreq.0.2%, more preferably .ltoreq.0.1%.
[0152] Preferably, cobalt is present a .ltoreq.0.2%, more preferably .ltoreq.0.1%.
[0153] Preferably, Co+W.ltoreq.0.5%, more preferably .ltoreq.0.3%, more preferably .ltoreq.0.1%.
[0154] Residual Contents and Unavoidable Impurities
[0155] Residual contents include contents that may unavoidably exist in the steel, i.e. alloying elements having residual contents are not purposefully added.
[0156] Unavoidable impurities can be phosphorus (P), sulphur (S), nitrogen (N), hydrogen (H), oxygen (O), calcium (Ca), and rare earth metals (REM) or the like. Their contents are preferably limited as follows in order to ensure the properties of the hardenable steel (composition): [0157] P.ltoreq.0.020%, preferably .ltoreq.0.015%, more preferably .ltoreq.0.012%, most preferably [0158] .ltoreq.0.008% (lower levels are particularly required when the steel is tempered) [0159] S.ltoreq.0.020%, preferably .ltoreq.0.010%, more preferably .ltoreq.0.005% [0160] N.ltoreq.0.012%, preferably .ltoreq.0.006%, [0161] H.ltoreq.0.0002%, [0162] O.ltoreq.0.005%, [0163] Ca.ltoreq.0.01 (typically used to control sulphur and oxygen levels) [0164] REM.ltoreq.0.1%.
[0165] The difference between residual contents and unavoidable impurities is that residual contents are controlled quantities of alloying elements, which are not considered to be impurities. A residual content is normally controlled by an industrial process does not have an essential effect upon the alloy. The levels of residual contents of hardenable steels are typically low.
[0166] The martensitic start temperature (M.sub.S) of the hardenable steel (composition) is preferably 340.degree. C., more preferably .gtoreq.350.degree. C., more preferably .gtoreq.360.degree. C., more preferably .gtoreq.370.degree. C., most preferably .gtoreq.375.degree. C.
[0167] For the purposes of this disclosure, the martensite start temperature M.sub.S may be calculated as:
M.sub.S=530-350*% C-40*% Mn-20*% Cr-10*% Mo-17*% Ni-8*% W-35*% V-10*% Cu+15*% Co+30*% Al
[0168] Method for Producing the Hardenable Steel
[0169] The hardenable steel (composition) disclosed herein is formed by hot-rolling (e.g. the above composition) and cooling to form a hardenable steel product. The actual process steps used to form the steel product can vary, as can the microstructure of the resultant steel prior to quench hardening. The steel product is preferably hot rolled, though cold rolling (for instance skin rolling) can be carried out prior to hardening.
[0170] Preferably, the steel product is hot rolled (i.e. no cold rolling is carried out).
[0171] Preferably, the hardenable steel (composition) is hot rolled.
[0172] Typical process steps for forming the hardenable steel product comprise the following steps in the given sequence: [0173] i. providing a steel slab (e.g. consisting of the chemical composition disclosed herein) at a temperature in the range of 950-1350.degree. C.; [0174] ii. hot-rolling in a temperature range of A.sub.r1 to 1300.degree. C. (preferably A.sub.r3 to 1300.degree. C.) to obtain a hot-rolled steel; [0175] iii. cooling and optionally coiling the hot-rolled steel to provide a hardenable steel product; [0176] iv. optionally processing the hardenable steel product to form a component therefrom.
[0177] The step of providing a steel slab may comprise forming a melt (e.g. from suitable components that combine to make the hardenable steel composition), and extruding the melt directly into the hot rolling. Alternatively, the step may comprise providing a preformed slab (or billet) of the hardenable steel composition and heating it to the required temperature prior to hot rolling.
[0178] The conditions used in the hot rolling step may be adjusted accordingly to ensure that the resultant steel has the desired balance of strength and flexibility. Likewise, the cooling steps following hot rolling are not critical, and may be suitably adjusted to provide the desired microstructure of the hot-rolled product.
[0179] The hardenability properties (hardness after quenching, tendency to form cracks) are influenced by the grain size of the hardenable steel. Typical average austenite grain size for the hardened steel is .ltoreq.25 .mu.m, preferably around 5-20 .mu.m, more preferably around 5-15 .mu.m. A suitable hot rolling protocol to form a product with this grain size is as follows: [0180] Rough rolling the heated steel slab e.g. for seven passes, the optional first pass being a calibration pass then followed by (preferably) six rough rolling passes each having about 20-40% (preferably 30%) size reduction. The slab start temperature during rough rolling is preferably from 1100-1300.degree. C., preferably 1230-1280.degree. C. [0181] Finish rolling through (e.g.) six rollers, the size reduction on the first roller being about 25-60% (preferably 30-50%), which gradually reduces to about 5-20% (preferably 10-15%) on the final roller. The finish rolling temperature is preferably 800-950.degree. C., preferably 860-930.degree. C. [0182] Coiling at 580-750.degree. C., preferably 650-720.degree. C., then air cooling
[0183] The purpose of the rough rolling is to compress the slab and remove any porosity that may still be present following the slab formation. The finish rolling refines the grain sizes in the steel.
[0184] The coiling temperature influences the predominant phase in the final steel. Coiling at 650-750.degree. C. (e.g. about 720.degree. C.) will promote ferrite and pearlite and provide a softer, more malleable material. However, these higher coiling temperatures will typically lead to more scale at the surface of the steel. Coiling at 580-650.degree. C. (e.g. about 630.degree. C.) reduces the likelihood of scale formation and promotes some bainite growth giving a favourable balance of properties.
[0185] Coiling at temperatures below 580.degree. C. (particularly below 500.degree. C.) promotes growth of high levels of bainite, which will make the resultant product less formable and potentially more difficult to process into a component prior to quench hardening. A high cooling rate and coiling at a temperature below M.sub.S promotes high levels of martensite, which increases the risk of cracks forming in the coiled strip.
[0186] Typically, the hot-rolling comprises hot-rolling to form a steel strip having a thickness of 2-15 mm, preferably 2-12 mm. Cold rolling may optionally be carried out, particularly if a strip with thickness <2 mm is desired. Alternatively, the hot rolling may form a plate having thickness of from 3-80 mm, preferably from 4-50 mm, more preferably 5-15 mm.
[0187] The issue of crack hardening is most problematic for thicker samples. Consequently, the steel of the disclosure shows most significant advantages for thicker samples. Preferably, the thickness of the steel is at least 5 mm, more preferably at least 6 mm, more preferably at least 6.5 mm, and even more preferably at least 7 mm.
[0188] Preferred thicknesses therefore include 5-15 mm, more preferably 6-12 mm.
[0189] These preferred thicknesses also relate to components formed from the steel. However, in the case of the component, the thickness may vary across the component, for instance if part of the component has been machined to form an edge. Preferably therefore the component has a maximum thickness of at least 5 mm, more preferably at least 6 mm, more preferably at least 6.5 mm, and even more preferably at least 7 mm.
[0190] Preferably the component has a minimum thickness of at least 5 mm, more preferably at least 6 mm, more preferably at least 6.5 mm, and even more preferably at least 7 mm.
[0191] By "maximum thickness" is meant the thickness of the thickest part of the component.
[0192] By "minimum thickness" is meant the thickness of the thinnest part of the component.
[0193] If a strip product is formed, coiling the strip at around 700.degree. C. will typically form a two-phase composition containing ferrite and pearlite. Coiling the strip at lower temperatures, such as around 600.degree. C., will typically promote bainite to form in addition to ferrite and pearlite. Lower coiling temperatures also promote a finer grain size.
[0194] The microstructure of the hot-rolled product may therefore vary depending on the process conditions used. The microstructure is typically tailored to provide the right balance of properties to enable the material to be formed into the desired product prior to quench hardening. The resultant steel is usually relatively soft (typical Vickers hardness levels range from 200-300 Hv10, preferably from 200-250 Hv10), typically with good flexibility (such as a bend radius of 2t when edges of the steel are machined), and medium-high strength (tensile strength 600-1000 MPa) so as to allow easy processing before quench hardening.
[0195] The properties of the hot rolled product may also be tailored by adjusting the microalloying elements. For instance, if very good fold forming properties are required, then niobium levels should be minimised, or preferably niobium should be avoided.
[0196] Processing the Steel
[0197] The optional processing steps can vary and may include machining, cutting (e.g. by oxy-fuel, plasma, waterjet or laser cutting), grinding (e.g. sharpening of a tool edge to form a blade), and cold forming (e.g. bending, flanging or the like).
[0198] Once formed into the final component, the material may be (reheat) quench hardened to form the final product. Quench hardening is carried out using a process comprising: [0199] a. providing a component formed from a hardenable steel composition; [0200] b. heating at least a region of the component to a temperature (T.sub.S) above A.sub.c3; [0201] c. quenching said region using a quenching fluid; and [0202] d. optionally tempering the quenched region.
[0203] An alternative process comprises the steps of: [0204] a'. providing a component formed from a hardenable steel; [0205] b'. heating at least a region of the component to a temperature (T.sub.S) above A.sub.c3; [0206] c'. quenching said region using a quenching fluid to form a hardened region; and [0207] d'. optionally tempering the hardened region;
[0208] The step a. (or a'.) of providing a component can comprise any or all of the steps i.-iv. set out above, and particularly step iv. set out above.
[0209] Reheat-Quench Hardening
[0210] In step b. (or b'.), the component or a region thereof is then heated to a temperature (T.sub.S) above A.sub.c3, i.e. the austenisation temperature. At this temperature, the microstructure of the steel becomes 100% austenite, although some carbides and/or nitrides such as VC, NbC, MoC, and TiN may remain undissolved. To enable this reaction to take place, the process preferably includes a soaking step (b.sub.s. or b.sub.s'.) after heating and before quenching, in which the component is held at a temperature above A.sub.r3 for a soak time t.sub.S.
[0211] The heat temperature (T.sub.S) will depend primarily on the composition of the hardenable steel. Generally speaking, a lower Mo content allows a lower T.sub.S to be used. Preferably, T.sub.S is above 850.degree. C., preferably above 900.degree. C., more preferably above 950.degree. C.
[0212] If T.sub.S is .gtoreq.950.degree. C., the hardenable steel (composition) preferably contains Ti and/or Nb.
[0213] If T.sub.S is too high, the rate of decarburisation at the surface of the component can become problematic, particularly if the soaking atmosphere is air. Likewise, crystal grain growth can also occur, so excessively high T.sub.S levels are to be avoided for steels that do not contain Nb and/or Ti.
[0214] Preferably, an upper value for T.sub.S is 1050.degree. C., preferably 1000.degree. C. If higher temperatures are used, the soaking time should be kept as short as possible and optionally a protective atmosphere (i.e. an inert gas or vacuum) should be used.
[0215] The optional soak time t.sub.S will vary depending on the shape size of the component. The soak time will typically be long enough to ensure complete austenisation, but not too long so as to avoid excessive grain growth. Excessive soak times beyond those needed to ensure complete austenisation moreover increase the costs unnecessarily.
[0216] Nevertheless, by way of guidance, a soak time for a component having a maximum thickness of 6 mm would typically be around 12 minutes when the temperature of the soak furnace is about 950.degree. C. Most components are therefore completely austenised when the soak time is 30 minutes or less, preferably 20 minutes or less.
[0217] The soak media is not critical, and air, inert gas or a vacuum may be used, preferably air. For higher soak temperatures or very long soak times, an inert gas or vacuum should be used as the soak media, to mitigate the risk of decarburisation.
[0218] Where only a portion of the component is being heated (for example only the surface region or a particular part such as a blade edge), the soak time will be correspondingly less, such as 2 minutes or less, preferably 1 minute or less. This ensures that only the area of interest reaches the austenising temperature, and excessive heat transfer to other regions is avoided.
[0219] The sample may be heated by any suitable means. When the entire component is being heated, a furnace is typically used. When only part of the component is being heated, only that part may be placed in the furnace. More typically, localised heating may be used, such as induction heating or a flame.
[0220] Preferably, the furnace temperature and the temperature of the soak medium will be above T.sub.S. For instance, a component being heated to around 930.degree. C. will typically be heated/soaked in a furnace set to 950.degree. C.
[0221] Higher soak temperatures are typically used when the product is to be processed during soaking to form the final component. For instance, the steel product may be in the form of a blank which is heated then processed (e.g. by hot stamping) to form the final component, which is then quench cooled. Since it is typically not possible to carry out these processing steps in a heating furnace, higher soak temperatures are required to ensure that the residual heat in the product keeps the temperature of the final component above A.sub.r3 throughout the processing steps prior to quenching. If multiple process steps are to be carried out, the steel product may be placed in the furnace between processing steps to ensure its temperature is maintained suitable high. In any event, the temperature should be maintained above A.sub.r3 prior to quenching.
[0222] Preferably, the steel is 100% austenite after step b. (or b'.) and optional step c. (or c'.).
[0223] During soaking, any molybdenum carbide in the austenite dissolves to form free molybdenum atoms. Preferably, after soaking, the heated region of the steel does not contain any molybdenum carbide.
[0224] Other undissolved carbides can be desirable as they prevent grain ground and lower the risk of hardening cracks forming. However, the can reduce hardness and also impact strength. For instance, if the vanadium content is high, there can be a risk of having a high amount of VC in the austenite, which can serve as a nucleation site for ferrite when the steel is cooled. Consequently, after soaking the heated region of the steel preferable does not contain any vanadium carbide.
[0225] After heating an optional soaking, the heated region is then quenched using a quenching fluid. The purpose of quenching is to rapidly cool the austenised steel down to below M.sub.S, the martensite start temperature. The quenching (i.e. step c. or c'.) occurs directly after steps b. and b.sub.s. (or b'. and b.sub.s'.), such that the temperature of the region does not fall below A.sub.r3 until the region is quenched.
[0226] Thus, in step c. (or c'.), the steel is quenched to below M.sub.S. Preferably, the steel is quenched to below 100.degree. C., more preferably the steel is quenched to room temperature.
[0227] Without wishing to be bound by theory, the improved resistance to quench cracking is believed to arise in part due to the influence the molybdenum has in promoting self-tempering during the quenching. Once the steel is cooled below M.sub.S, the majority of the austenite transforms into martensite very rapidly. The quenching causes the steel to continue cooling, however there is a period where the steel is at a temperature above room temperature but below M.sub.S. During this time, the martensite undergoes self-tempering, that is tempering caused by the residual heat retained in the steel rather than heat which is externally applied.
[0228] A higher M.sub.S temperature will promote self-tempering at an earlier stage as the steel cools. Mn, Cr and Ni have a bigger impact on M.sub.S than Mo has, so the steel of the disclosure provides good hardenability in combination with a relatively high M.sub.S. Due to the relatively high M.sub.S temperature in the steel of the disclosure, the steel undergoes more extensive self-tempering during quenching. Moreover, the lower carbon levels at the surface due to decarburisation raise the M.sub.S in these regions, which further helps to lengthen the time that the surface undergoes self-tempering.
[0229] The self-tempering promoted by molybdenum helps relieve internal stresses as the steel is cooled. Moreover, molybdenum also helps improve the impact strength of the quenched steel, decreasing the brittle to ductile transition temperature in a Charpy-V impact test.
[0230] The quench media may be any suitable media to ensure rapid cooling of the component, i.e. a cooling rate of >20.degree. C./s, preferably >50.degree. C./s, more preferably >80.degree. C./s. Suitable quench media include oil or water, with water being preferred.
[0231] Agitating (or mixing/stirring) the water will increase the cooling rate even further. Suitable means for agitating the quench water include a propeller.
[0232] By "water" as the quench media is meant water or salt water (i.e. brine).
[0233] After quenching, the region of the component subjected to quench hardening has a martensitic microstructure. Preferably, the microstructure of the region comprises .gtoreq.90 martensite, preferably .gtoreq.95% martensite, more preferably .gtoreq.98% martensite, more preferably .gtoreq.99% martensite, more preferably .gtoreq.99.5% martensite, most preferably 100% martensite.
[0234] The martensitic region can contain precipitates such as carbides including (for example) MoC, NbC, TiC, NbTiC, and VC, and nitrides including (for example) TiN and VN, as well as carbonitrides.
[0235] The hardened region is preferably crack-free, and advantageously crack-free without the need for any tempering steps.
[0236] The Vickers hardness of the region is preferably .gtoreq.630 Hv10, preferably .gtoreq.650 Hv10, more preferably .gtoreq.670 Hv10, even more preferably .gtoreq.700 Hv10.
[0237] Vickers hardness may be measured using standard SFS EN ISO 6507-1:2006, for instance using a DuraScan 80 as a hardness meter.
[0238] The hardness of a steel can vary according to the depth from the surface. Typically, the surface hardness is slightly lower, as the surface may decarburize during austenisation.
[0239] Likewise, the centre of the steel cools more slowly, so the hardness at the centre is typically lower.
[0240] As used herein, the hardness of the steel is preferably the hardness at 1/4 the sample thickness or 4 mm from the surface, whichever is less.
[0241] Preferably, for samples <12 mm in thickness, the average hardness of the sample is preferably .gtoreq.630 Hv10, preferably .gtoreq.650 Hv10, more preferably .gtoreq.670 Hv10, even more preferably .gtoreq.700 Hv10.
[0242] As used herein, the "average hardness" corresponds to the mean of several hardness measurements taken at points evenly spaced through the samples thickness, for instance every 1 mm starting 0.5 mm from the surface, preferably starting 1 mm from the surface.
[0243] More preferably, for samples <12 mm in thickness, the entire thickness of the sample is preferably .gtoreq.630 Hv10, preferably .gtoreq.650 Hv10, more preferably .gtoreq.670 Hv10, even more preferably .gtoreq.700 Hv10.
[0244] To measure the hardness at a given depth, the steel sample is preferably cut in two across its thickness (i.e. perpendicular to a surface), then the hardness is measured on the exposed face which transverses the steel.
[0245] The hardened region (e.g. a .gtoreq.5 mm sample quenched with water) preferably has a median Charpy V impact strength of .gtoreq.3 J/cm.sup.2 at 20.degree. C., preferably .gtoreq.6 J/cm.sup.2 at 20.degree. C., preferably .gtoreq.12 J/cm.sup.2 at 20.degree. C. and more preferably .gtoreq.18 J/cm.sup.2 at 20.degree. C.
[0246] The Charpy V impact strength may be measured using standard ISO 148 at 20.degree. C. (e.g. ISO148:2010). Any suitable sample size in accordance with the standard can in principle be used (e.g. 5 mm, 7.5 mm or 10 mm), although a 5 mm sample is typical. Typically at least three (preferably at least five) measurements are taken, with the test specimen taken being longitudinal to the main hot rolling direction.
[0247] Tempering of the quenched steel reduces the hardness, but increases the impact strength. Likewise, quenching with oil will produce a less hard steel with a higher impact strength. The advantage of the steel of the disclosure is not merely the high hardness or high impact strength, but the combination of both properties which may be optimised relative to one another by tempering.
[0248] The applicant has found that the relationship between the hardness and impact strength may preferably be characterised as follows:
V.sub.J+(H.sub.X*Hv10)>A.sub.X
where [0249] V.sub.J is the Charpy V impact strength in J/cm.sup.2 [0250] Hv10 is the Vickers Hardness [0251] A.sub.X=95, and [0252] H.sub.X=0.125.
[0253] Preferably, A.sub.X is 97.5 and H.sub.X is 0.125.
[0254] Preferably, A.sub.X is 100 and H.sub.X is 0.125.
[0255] More preferably, A.sub.X is 165.8 and H.sub.X is 0.2156.
[0256] More preferably, A.sub.X is 192.5 and H.sub.X is 0.2508.
[0257] More preferably, A.sub.X is 218.8 and H.sub.X is 0.2855.
[0258] Plots of these relationships are shown in FIG. 1. Thus, the hardened steels according to the disclosure have a combination of impact strength and hardness that falls to the right (or above) the various plotted lines. It should be noted that the higher values of A.sub.X are typically only achievable when the steel is quenched with water and tempered following hardening.
[0259] Preferably, the entire component is quenched in step d. (or d'.).
[0260] Preferably, the entire component is tempered in step e. (or e'.). Preferably, the entire component is reheat-quench hardened.
[0261] Preferably, the entire component has a hardness of .gtoreq.630 Hv10, preferably .gtoreq.650 Hv10, more preferably .gtoreq.670 Hv10, even more preferably .gtoreq.700 Hv10.
[0262] If just a region of the product such as the surface or a blade edge, the product may need to be processed differently during the hot rolling stages to ensure the final product has the desired properties. For instance, if the hot rolled product is air cooled following hot rolling, the overall properties vary depending on the balance of ferrite, pearlite and bainite. However, typically speaking the steel does not possess suitable structural properties for end use, as the resultant steel is generally quite brittle. One possible option is therefore to quench the steel following finish rolling to provide a hardened product which may be further processed then reheat-quench hardened to form the final product. In these embodiments, the resultant quenched hot rolled steel is not coiled, since the coiling process would likely fracture the quenched steel.
[0263] If this methodology is used, the quench hardening following hot rolling is preferably carried out with a cooling rate of 20-50.degree. C./s, for example by quenching with oil. This will form a product having a hardness preferably in the region of 475-560 Hv10 with good impact strength. The material is therefore hard enough for most final uses, but not so hard that it cannot be processed (for example cut or sharpened) into a final component. Tempering may optionally be carried out at this stage to lower the hardness and improve impact strength. A region (such as a blade edge) may then be reheat-quench hardened with a cooling rate of >50.degree. C./s (for example quenching with water) to form a region having higher hardness.
[0264] Optionally, the component may be tempered following quench hardening. Tempering may be carried out by heating the component to a tempering temperature T.sub.Q (preferably at a temperature of from 150.degree. C. to 700.degree. C.), holding the component for a tempering time t.sub.Q, then cooling the component down to room temperature. Preferably, the cooling following tempering is done in air, preferably still air.
[0265] Tempering of the component reduces the hardness, but increases tensile strength, ductility and toughness of the component. The molybdenum provides tempering resistance (i.e. it helps to retain the hardness during tempering), especially at temperatures well above M.sub.S.
[0266] An advantage of the steel of the disclosure is that tempering is not essential to prevent hardening induced cracking. Likewise, the impact strength is typically sufficiently high even without tempering. Preferably, the only tempering done may be the heat treatment to fuse any powder paint coatings on the component following quench hardening. Such treatments are preferably carried out at around 175-225.degree. C. (such as 175-200.degree. C. or 200-225.degree. C.), which is typically well below M.sub.S. Following these treatments, the steel retains its martensitic structure and typically has a hardness of .gtoreq.575 Hv10 (more preferably .gtoreq.600 Hv10, more preferably .gtoreq.625 Hv10 and with a high carbon content even .gtoreq.650 Hv10) together with acceptable other properties like high impact strength, low brittleness and good tensile strength.
[0267] The balance of hardness and impact strength achievable using the steels disclosed herein is often very desirable. However, there may be a need to provide an extremely hard region which has lower impact strength, for instance at the edge of a blade. Such components may be formed using the double quenching method set out above.
[0268] The disclosure consequently provides a method comprising the following steps: [0269] 1) providing a hardenable steel (composition); [0270] 2) heating the hardenable steel (composition) to a temperature (T.sub.S) above A.sub.c3; [0271] 3) quenching hardenable steel (composition) in a first quenching step using a first quenching fluid to form a component; [0272] 4) optionally processing the component; [0273] 5) heating a region of the component to a temperature (T.sub.S) above A.sub.c3; [0274] 6) quenching the region in a second quenching step using a second quenching fluid; and [0275] 7) optionally tempering the component; [0276] wherein [0277] the region does not encompass the entire component, and [0278] the cooling rate in the second quenching step is >50.degree. C./s, preferably >80.degree. C./s.
[0279] In this method, steps 1)-3) may form part of the hot rolling process used to form the steel. In which case, step 4) preferably comprises the mechanical processing steps such as stamping, cutting etc. to form a final component, followed by optional tempering. Alternatively, steps 1)-3) may be carried out on a preformed component, in which case the method is essentially a double reheat-quench method. Even so, optional step 4) may still include tempering, as well as some mechanical processing such as sharpening of an edge.
[0280] The cooling rate in the first quenching step is preferably >20.degree. C./s, though higher rates such as >50.degree. C. or even >80.degree. C./s are possible. If higher rates are used (>50.degree. C./s), preferably step 4) will involve tempering, preferably tempering at >300.degree. C.
[0281] Preferably, the hardness of the component after step 4) will be from 350 to 600 Hv10, preferably from 475 to 560 Hv10.
[0282] Step 5) involves heating a localised region of the component, such as by induction heating or flame heating. This localised region is then quench cooled in a second quenching step with a very fast quenching rate (>50.degree. C./s, preferably >80.degree. C./s). The hardness of the region is preferably >630 Hv10, preferably .gtoreq.650 Hv10, more preferably 670 Hv10, even more preferably .gtoreq.700 Hv10.
[0283] The first and second quenching fluid may be the same. However, in this case, step 4) will preferably comprise tempering so as to reduce the hardness of the overall component before the second quenching step.
[0284] Preferably, the second quench fluid is water. Preferably, the first quench fluid is oil.
EXAMPLES
Example 1--Laboratory Scale, Plate Rolled
[0285] Steels were made having compositions as set out in the following table:
TABLE-US-00005 1-A 1-B 1-C 1-D C 0.453 0.463 0.387 0.433 Si 0.183 0.190 0.191 0.205 Mn 0.245 0.246 0.468 0.251 Mo 0.002 0.001 0.001 0.769 Cr 0.063 0.218 0.213 0.079 Ni 0.021 0.022 0.021 0.022 Al 0.036 0.036 0.038 0.045 S 0.0039 0.0038 0.0034 0.0043 P 0.005 0.005 0.005 0.005 Ti 0.012 0.012 0.011 0.012 Nb 0.013 0.014 0.013 0.014 V 0.001 0.001 0.001 0.031 Cu 0.008 0.008 0.008 0.011 N 0.0036 0.0032 0.0032 0.0044 T.sub.s.sup.1/.degree. C. 910 910 910 930 T.sub.s.sup.2/.degree. C. 980 980 980 980
[0286] Four billets of the steel were direct quenched after hot rolling in a lab scale process. The billets were then reheat-quench hardened (using water) at the soak temperatures (T.sub.S.sup.1 or T.sub.S.sup.2) shown in the table, two billets at each temperature. All billets of steel 1-D were defect and crack free after quench hardening. Three of the four billets of steel 1-C were defect and crack free. However one small crack occurred in one of the billets of steel 1-C quench hardened from a soak temperature of 910.degree. C. The remaining steels all formed cracks during quench hardening.
[0287] Hardness tests to determine Vickers hardness were carried out according to standard SFS EN ISO 6507-1:2006 using a DuraScan 80 as a hardness meter.
[0288] FIG. 2 shows the plot of hardness vs. depth for an 8 mm thick sample of steel 1-A, with the hardness plotted as Vickers (Hv10) hardness, with the corresponding Rockwell C hardness levels being shown. The measurements were taken by cutting the steel into two and measuring the hardness on the exposed cut face which transverses the steel. FIG. 3 shows a similar plot for steel 1-B, while FIG. 4 shows a similar plot for steel 1-C. These plots show the steels have relatively low hardness when quench hardened at 910.degree. C. Steel 1-B is able to obtain reasonable hardness at the surface when quench hardened at 980.degree. C., although the hardness is not retained through the entire thickness of the sample. Steel 1-C obtains a similar hardness throughout the entire thickness of the sample, but the hardness obtained is approximately 600 Hv10 (55 HRC).
[0289] FIG. 5 shows the equivalent plot for steel 1-D according to the disclosure. The plot clearly shows significantly higher hardness values which are retained through the entire sample thickness. FIG. 6 shows an equivalent result is obtained for a 12 mm thick sample.
[0290] FIG. 7 shows the Vickers hardness at 2 mm depth for samples of steel 1-D quenched from T.sub.S 980.degree. C. after tempering at temperatures of 150-600.degree. C. The plot shows the steel shows good hardness retention during tempering, retaining Rockwell hardness values of >55 HRC when tempering at 200.degree. C.
[0291] Impact tests were carried out according to standard ISO 148-1:2010 at a temperature of +20.degree. C. The thickness of the specimens was 8 mm (6 mm thickness accounting for the notch), and the values reported at then converted to J/cm.sup.2. The results are shown in the table below:
TABLE-US-00006 A.sub.x Steel T.sub.s s/mm V-Notched Initial J Ave. Hv10 (H.sub.x = 0.125) 1-A 910 8 Yes 300 47 260 80 1-A 980 8 Yes 300 9 325 50 1-B 910 8 Yes 300 52 280 87 1-B 980 8 Yes 300 9 560 79 1-C 910 8 Yes 300 23 310 62 1-C 980 8 Yes 300 11 600 86 1-D 930 8 Yes 300 7.3 740 100 1-D 980 8 Yes 300 7.3 740 100
[0292] The results show that steel 1-D has excellent impact toughness at both quench temperatures, whereas the remaining steels only achieve good impact toughness in the notched test at the higher quench temperature. Steel 1-D according to the disclosure achieves A.sub.X values in line with the preferred ranges above for each value of H.sub.X. The impact strength of steel 1-D is relatively low, but it is still very good considering the hardness. Tempering the steel reduces the hardness and increases the impact strength
Example 2--Full Scale, Strip Rolled
[0293] Steels with similar compositions to 1-D were produced in a large scale (commercial) facility. The steel compositions and their corresponding rolling conditions are summarised in the following tables:
TABLE-US-00007 2-D 3-D C 0.4300 0.4140 Si 0.200 0.186 Mn 0.255 0.250 Mo 0.699 0.664 Cr 0.059 0.055 Ni 0.050 0.061 Al 0.034 0.034 S 0.006 0.0011 P 0.0070 0.0070 Ti 0.0130 0.0120 Nb 0.0130 0.0120 V 0.0400 0.0400 Cu 0.0140 0.0160 N 0.0028 0.0046 Ca 0.0018 0.0016
TABLE-US-00008 Rough Finish Coiling R.sub.p0.2 R.sub.M Rolling Rolling Temp Roll./Trav. Roll./Trav. Steel Temp (.degree. C.) (.degree. C.) Temp (.degree. C.) (MPa) (MPa) 2-D 1085 890 725 436/437 680/654 2-D 1095 875 630 528/552 737/730 2-D 1085 890 720 431/447 682/673 3-D 1090 870 630 489/532 696/699 3-D 1090 890 720 407/451 644/660
[0294] To show the relationship between the hardness and impact strength of the reheat-quench hardened steels, steel billets (8 mm.times.65 mm.times.150 mm) of steel 2-D were heated in an air furnace at 980.degree. C. for 40 minutes then quenched with water. The resultant steels were then tempered for 120 minutes at various temperatures. The hardness and impact strength of the resultant hardened and tempered steels are shown in FIGS. 7 and 8. These values are summarised in the following table:
TABLE-US-00009 A.sub.x Tempering Temp. (.degree. C.) Hv10 Impact Strength (J/cm.sup.2) (H.sub.x = 0.125) -- 650 23 104 175 615 37 114 300 510 39 103 400 445 59 115 500 405 74 125 600 400 77 127
[0295] As shown in the table, the steel retains the high A.sub.X value for all tempering temperatures, with a particularly good combination of values being obtained for tempering at 175.degree. C.
Example 3
[0296] Several quench hardenable steels with the following formula were compared to steel 2-D:
TABLE-US-00010 51CrV4 38MnB5 42MnV7 2-D C 0.52 0.396 0.395 0.4300 Si 0.233 0.183 0.159 0.200 Mn 0.828 1.27 1.67 0.255 Mo 0.010 0.001 0.007 0.699 Cr 0.911 0.207 0.050 0.059 Ni 0.044 0.034 0.046 0.050 Al 0.028 0.039 0.037 0.034 S 0.0025 0.0029 0.0069 0.006 P 0.010 0.013 0.010 0.0070 Ti 0.002 0.034 0.017 0.0130 Nb 0.002 0.001 0.001 0.0130 V 0.111 0.010 0.091 0.0400 Cu 0.055 0.006 0.023 0.0140 N 0.031 0.0014 0.0060 0.0028
[0297] Samples of these steels (5 mm thick, 8 mm thick for steel 2-D water quenched) were reheat-quench hardened under various conditions, and the resulting hardness and impact strengths are summarised in the table below:
TABLE-US-00011 Impact Quench Quench Hardness* Strength A.sub.x Steel Temp (.degree. C.) medium (HV10) (J/cm.sup.2) (H.sub.x = 0.125) 51CrV4 1000 Oil 704 5 93 38MnB5 1000 Oil 603 13 88 42MnV7 1000 Oil 621 10 88 42MnV7 1000 Water 658 5 87 2-D* 980 Oil 523 43 108 2-D** 980 Water 650 23 104 2-D*** 980 Water 615 37 115 *Average measured hardness Hv10 through the thickness starting from 0.5 mm from the surface (1 mm from the surface for 42MnV7) **8 mm thick sample ***8 mm thick sample, tempered for two hours at 175.degree. C.
[0298] The 51CrV4 and occasionally 38MnB5 steels could not be successfully water quenched due to quench cracks forming. When oil quenched, both steels provided good hardness, but relatively poor impact strengths.
[0299] 42MnV7 can be quenched with either water or oil, but the resultant steel has relatively poor impact strength. Also, the water quenched steel has a high tendency to form cracks.
[0300] Steel 2-D can be quenched with either water or oil to give a steel with a better combination of hardness and impact strength. Tempering the quenched steel reduces the hardness, but increases the impact strength. No hardening induced cracks were observed.
Example 4
[0301] Several 6 mm thick blanks were made from steel 2-D having dimensions of 310 mm in the rolling direction (1010), and 300 mm in the longitudinal direction. To joining edges were cut by flame cutting (1020), and two edges by mechanical cutting (1030). Multiple 20 mm diameter holes were stamped in the test pieces, four holes 20 mm apart in the centre forming a square, and two holes 10 mm from the edges at the corners of the mechanically cut edges and flame cut edges, as shown in FIG. 9.
18 blanks were heated for 30 minutes at 980.degree. C. and quenched using still water to room temperature (approx. 20-30.degree. C.). 9 samples were tempered within 5 minutes at 175.degree. C. for 2 hours. The samples were visually checked for cracks following quenching and optional tempering, with no cracks being detected.
[0302] After five weeks, the samples were visually inspected again, with both sides of the samples also being checked by magnetic-particle testing. Again, no cracks were detected when assessed to standard ISO 23278:2015, Class 2X.
Example 5--Laboratory Scale, Plate Rolled
[0303] Steels were made having compositions as set out in the following table:
TABLE-US-00012 1605 1606 1607 1608 1609 1610 C 0.452 0.451 0.530 0.525 0.444 0.451 Si 0.206 0.203 0.206 0.205 0.204 0.200 Mn 0.473 0.478 0.245 0.239 0.238 0.238 Mo 0.383 0.400 0.681 0.300 0.299 0.297 Cr 0.208 0.209 0.014 0.012 0.254 0.014 Ni 0.011 0.011 0.011 0.010 0.010 0.010 Al 0.053 0.049 0.047 0.042 0.046 0.043 S 0.003 0.003 0.003 0.004 0.003 0.004 P 0.007 0.006 0.006 0.006 0.005 0.006 Ti 0.012 0.012 0.013 0.012 0.012 0.029 Nb 0.014 0 0.014 0.015 0.015 0.016 V 0.001 0.001 0.029 0.029 0.029 0.029 Cu 0.008 0.008 0.009 0.007 0.007 0.007 B 0 0 0 0 0 0.002 N 0.004 0.004 0.004 0.004 0.004 0.005 T.sub.s.sup.1/.degree. C. 850 850 850 850 850 850 T.sub.s.sup.2/.degree. C. 980 980 980 980 980 980
[0304] Several billets of the steels were direct quenched after hot rolling in a lab scale process. The billets were then reheat-quenched (using water) at the soak temperatures (T.sub.S.sup.1 or T.sub.S.sup.2) shown in the table. All the reheat-quenched billets were crack-free after quenching. The hardness of the reheat-quenched steels are shown in FIG. 10 (median value of five measurements).
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.