Methods For Reducing Hydrogen Sulfide In Crude Oil
Burgazli; Cenk ; et al.
U.S. patent application number 16/431902 was filed with the patent office on 2019-12-05 for methods for reducing hydrogen sulfide in crude oil. This patent application is currently assigned to Innospec Limited. The applicant listed for this patent is Innospec Limited. Invention is credited to Cenk Burgazli, Philip James Maltas.
| Application Number | 20190367819 16/431902 |
| Document ID | / |
| Family ID | 66857939 |
| Filed Date | 2019-12-05 |



| United States Patent Application | 20190367819 |
| Kind Code | A1 |
| Burgazli; Cenk ; et al. | December 5, 2019 |
METHODS FOR REDUCING HYDROGEN SULFIDE IN CRUDE OIL
Abstract
The invention provides a method of removing or lowering amounts of hydrogen sulfide in a crude oil, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
| Inventors: | Burgazli; Cenk; (Middletown, DE) ; Maltas; Philip James; (Willaston, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | ; Innospec Limited Cheshire GB |
||||||||||
| Family ID: | 66857939 | ||||||||||
| Appl. No.: | 16/431902 | ||||||||||
| Filed: | June 5, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62680695 | Jun 5, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 2300/202 20130101; C10G 2300/207 20130101; C10G 2400/08 20130101; C10G 2300/1051 20130101; C10L 2200/043 20130101; C10L 2270/04 20130101; C10L 10/00 20130101; C10G 2300/104 20130101; C10L 1/228 20130101; C10G 2300/1055 20130101; C10L 1/2283 20130101; C10L 2290/543 20130101; C10G 29/20 20130101 |
| International Class: | C10G 29/20 20060101 C10G029/20; C10L 10/00 20060101 C10L010/00; C10L 1/228 20060101 C10L001/228 |
Claims
1. A method of removing or lowering amounts of hydrogen sulfide in a crude oil, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
2. A method of producing a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and/or an amine residue thereof, the method comprising distilling the crude oil to form the distillate fuel component.
3. The method according to claim 1, wherein R.sup.1 represents a methyl group or a 5C alkyl group.
4. The method according to claim 2, wherein R.sup.1 represents a methyl group or a 5C alkyl group.
5. The method according to claim 3, wherein R.sup.1 represents a 5C alkyl group.
6. The method according to claim 4, wherein R.sup.1 represents a 5C alkyl group.
7. The method according to claim 1, wherein the imine compound is tert-butylimine or tert-octylimine.
8. The method according to claim 2, wherein the imine compound is tert-butylimine or tert-octylimine.
9. The method according to claim 3, wherein the imine compound is tert-butylimine or tert-octylimine.
10. The method according to claim 4, wherein the imine compound is tert-butylimine or tert-octylimine.
11. The method according to claim 7, wherein the imine compound is tert-octylimine.
12. The method according to claim 8, wherein the imine compound is tert-octylimine.
13. The method according to claim 9, wherein the imine compound is tert-octylimine.
14. The method according to claim 10, wherein the imine compound is tert-octylimine.
15. The method according to claim 1, wherein the imine compound is prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
16. The method according to claim 2, wherein the imine compound is prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
17. The method according to claim 3, wherein the imine compound is prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
18. The method according to claim 4, wherein the imine compound is prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
19. A composition comprising a crude oil and an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
20. The composition according to claim 19, wherein R.sup.1 represents a methyl group or a 5C alkyl group.
21. The composition according to claim 20, wherein R.sup.1 represents a 5C alkyl group.
22. The composition according to claim 19, wherein the imine compound is tert-butylimine or tert-octylimine.
23. The composition according to claim 22, wherein the imine compound is tert-octylimine.
24. The composition according to claim 19, wherein the imine compound is prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
25. A distillate fuel component boiling in the kerosene range obtainable by a method of producing a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and/or an amine residue thereof, the method comprising distilling the crude oil to form the distillate fuel component.
26. The method of producing a distillate fuel component boiling in the kerosene range according to claim 2, wherein the distillate fuel component has improved thermal stability when compared to a distillate fuel component produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
27. The distillate fuel component boiling in the kerosene range according to claim 25, wherein the distillate fuel component has improved thermal stability when compared to a distillate fuel component produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
28. An aviation turbine fuel composition comprising a distillate fuel component boiling in the kerosene range obtainable by a method of producing a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and/or an amine residue thereof, the method comprising distilling the crude oil to form the distillate fuel component.
29. The aviation turbine fuel composition according to claim 28, wherein the aviation turbine fuel has improved thermal stability as measured by ASTM D3241 when compared to an aviation turbine fuel produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and benefit of U.S. Provisional Application No. 62/680,695, entitled METHODS FOR REDUCING HYDROGEN SULFIDE IN CRUDE OIL, filed on Jun. 5, 2018, which is incorporated by reference herein in its entirety and for all purposes.
TECHNICAL FIELD OF THE INVENTION
[0002] The invention relates to a method of removing or lowering amounts of hydrogen sulfide in a crude oil. The invention also relates to a method of forming a distillate fuel component boiling in the kerosene range from a crude oil. The invention also relates to a composition comprising a crude oil and an imine compound and to a distillate fuel component boiling in the kerosene range.
BACKGROUND TO THE INVENTION
[0003] It is commonly known that crude oil typically contains hydrogen sulfide and other sulfur based species that can liberate hydrogen sulfide (i.e. hydrogen sulfide precursors). Hydrogen sulfide is toxic and corrosive and so it is desirable to remove it from crude oil, or at least reduce the levels of hydrogen sulfide present in crude oil.
[0004] Various methods of removing or reducing the amount of hydrogen sulfide (and optionally other other sulfur based species when present) in crude oil are known. One of the most common methods is to use hydrogen sulfide scavengers, which react with hydrogen sulfide and remove it or reduce its amount, for example when present in crude oils.
[0005] Hydrogen sulfide scavengers may be used at various stages of the crude oil production and/or processing operations. For example, the hydrogen sulfide scavenger may be added to the crude oil whilst downhole, during production, during above ground processing, during transportation or storage, i.e. at any point prior to entering the refinery for processing. Hydrogen sulfide scavengers may also be used at the refinery for example, on receipt of the oil, during storage or during the refining process.
[0006] Thus, it is well known to treat crude oil with hydrogen sulfide scavengers to remove or reduce the levels of hydrogen sulfide in the oil. Examples of commonly used organic hydrogen sulfide scavengers include non amine scavengers such as aldehydes and protected aldehydes like acetals, and nitrogen based scavengers such as amines, triazines and imine compounds.
[0007] When the imine compound reacts with hydrogen sulfide and/or sulfur based species, the reaction produces a nitrogen containing compound, for example an amine residue of the imine and a hydrogen sulfide aldehyde adduct.
[0008] U.S. Pat. No. 5,169,411 discloses the use of certain hindered monoimines as hydrogen sulfide scavengers in the sulfur containing complex media of crude oils, petroleum residua and fuels.
[0009] Whilst hydrogen sulfide scavengers are known, it would be desirable to provide further hydrogen sulfide scavengers, for example which are more effective at removing or reducing amounts of hydrogen sulfide in crude oil than current hydrogen sulfide scavengers. In particular it would be desirable to provide imine based hydrogen sulfide scavengers which react more quickly than known scavengers and/or can consistently achieve low levels of hydrogen sulfide at low treat rates.
[0010] At a refinery, the crude oil (for example containing hydrogen sulfide scavenger(s)) is typically treated by a desalting process and then distilled at atmospheric pressure to produce several distillation cuts or fractions including gaseous hydrocarbons, gasoline, naptha, kerosene, light gas oil and heavy gas oil. After this atmospheric first distillation, a high proportion (typically 40 to 60 wt %) remains as a crude oil residue (also known as an atmospheric residue), which residue is then typically distilled in a vacuum distillation unit. After the vacuum distillation, a further crude oil residue remains (also known as a vacuum residue). The distillation fractions and residues may be further processed in the refinery.
[0011] The distillation fractions may be further processed or used as straight run distillates alone or with other blending components to produce fuels meeting appropriate fuel specifications. For example, after further processing, the distillate fuel component boiling in the kerosene range may be suitable for use as a component of an aviation turbine fuel.
[0012] Fuels are required to meet many fuel specification properties such as boiling range, corrosion requirements, lubricity and thermal stability. Of particular importance for the aviation turbine fuel is the thermal stability requirement for example, as listed in ASTM D1655 Standard Specification for Aviation Turbine Fuels for the fuel as measured by ASTM D3241 Test Method for Thermal Oxidation Stability of Aviation Turbine Fuels.
[0013] Starting in 2015 there were several occurrences of thermal stability failures in aviation turbine fuels. The failures were with fuels that were previously qualified by ASTM D3241 as meeting the ASTM D1655 specification thermal stability requirement but upon storage were failing to meet this thermal stability requirement. No other specification parameter tested was found to be outside acceptable limits. The subject fuels were analysed and found to contain trace amounts of atypical amines, specifically amines that were not found in crude oil. The source of these amines have so far not been identified.
[0014] The inventors believe the above mentioned thermal stability issues may result from the presence of the atypical amines in the aviation turbine fuel and that the presence of the atypical amines results from the use of hydrogen sulfide scavengers, where the amines were either directly used as a hydrogen sulfide scavenger to capture hydrogen sulfide or are by products of hydrogen sulfide scavengers. The inventors believe that these amines distil in the kerosene fraction and find their way into aviation turbine fuel, thus adversely impacting aviation turbine fuel thermal stability.
[0015] It would be desirable to overcome the thermal stability issues associated with aviation turbine fuels. It would also be desirable to provide improved methods of removing or reducing levels of hydrogen sulfide in crude oil, and particularly improved methods of removing or reducing levels of hydrogen sulfide in crude oil which do not adversely affect the thermal stability of aviation turbine fuels produced from said crude oils.
SUMMARY OF THE INVENTION
[0016] According to a first aspect of the present invention, there is provided a method of removing or lowering amounts of hydrogen sulfide in a crude oil, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
[0017] According to a second aspect of the present invention, there is provided a method of producing a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and/or an amine residue thereof, the method comprising distilling the crude oil to form the distillate fuel component.
[0018] According to a third aspect of the present invention, there is provided a composition comprising a crude oil and an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
[0019] According to a fourth aspect of the present invention, there is provided a distillate fuel component boiling in the kerosene range obtainable by (or obtained by) a method according to the second aspect of the present invention.
[0020] According to a fifth aspect of the present invention, there is provided the use of a distillate fuel component boiling in the kerosene range according to the fourth aspect of the present invention in an aviation turbine fuel composition.
[0021] According to a sixth aspect of the present invention, there is provided an aviation turbine fuel composition comprising a distillate fuel component boiling in the kerosene range according to the fourth aspect of the present invention.
[0022] According to a seventh aspect of the present invention, there is provided a method of removing or lowering amounts of hydrogen sulfide present during distillation of a crude oil to form a distillate fuel component boiling in the kerosene range, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and distilling the crude oil (including the imine compound) to form the distillate fuel component.
[0023] According to an eighth aspect of the present invention, there is provided the use of an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, to remove or lower the amount of hydrogen sulfide in a crude oil.
[0024] According to a ninth aspect of the present invention, there is provided the use of an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, in the preparation of a distillate fuel component boiling in the kerosene range.
DETAILED DESCRIPTION OF THE INVENTION
[0025] Unless otherwise stated, the following terms used in the specification and claims have the meanings set out below.
[0026] As used herein, the term "crude oil" means unrefined oil. Crude oil is typically composed of a complex mixture of organic (predominantly hydrocarbon) and inorganic compounds.
[0027] As used herein the term "aviation turbine fuel" means any type of fuel suitable for use in aircraft powered by gas-turbine engines. Aviation turbine fuel may also be commonly known as aviation kerosene, aviation turbine kerosene, aviation jet fuel or jet fuel. Aviation turbine fuel typically comprises a petroleum distillate boiling in the kerosene range and is produced to a national or international aviation specification such as ASTM D1655.
[0028] As used herein, the term "alkyl" includes both straight and branched chain alkyl groups. The term "alkyl" does not include aryl groups. References to individual alkyl groups such as "propyl" are specific for the straight chain version only and references to individual branched chain alkyl groups such as "isopropyl" are specific for the branched chain version only. For example, "(1-5C)alkyl" includes (1-2C)alkyl, propyl, isopropyl and tert-butyl.
[0029] Throughout this specification, the term "comprising" or "comprises" means including the component(s) specified but not to the exclusion of the presence of other components. The term "consisting essentially of" or "consists essentially of" means including the components specified but excluding other components except for components added for a purpose other than achieving the technical effect of the invention. The term "consisting of" or "consists of" means including the components specified but excluding other components.
[0030] Whenever appropriate, depending upon the context, the use of the term "comprises" or "comprising" may also be taken to include the meaning "consists essentially of" or "consisting essentially of", and may also be taken to include the meaning "consists of" or "consisting of".
[0031] The optional features set out herein may be used either individually or in combination with each other where appropriate and particularly in the combinations as set out in the accompanying claims. The optional features for each exemplary embodiment of the invention, as set out herein are also applicable to any other aspects or exemplary embodiments of the invention, where appropriate. In other words, the skilled person reading this specification should consider the optional features for each aspect or embodiment of the invention as interchangeable and combinable between different aspects of the invention.
Method
[0032] In a first aspect, the present invention provides a method of removing or lowering amounts of hydrogen sulfide in a crude oil, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
[0033] In the method of the first aspect, the crude oil (prior to addition of the imine compound) may, for example, contain up to 5000 mg of hydrogen sulfide per L (litre) of crude oil, for example up to 1000 mg of hydrogen sulfide per L (litre) of crude oil, or for example up to 500 mg/L. Typically, crude oils may contain up to 200 mg, or up 150 mg, of hydrogen sulfide per L of crude oil. For example, the crude oil may contain 1 to 150 mg, such as 1 to 100 mg, of hydrogen sulfide per L of crude oil. Typically, the crude oil may contain 10 to 50 mg of hydrogen sulfide per L of crude oil. Such amounts refer to the amount of hydrogen sulfide in the crude oil in the liquid phase.
[0034] It is common for levels of hydrogen sulfide to be measured in the gas phase such as in the head space of a tank. Such measurements in the gas phase are commonly quoted as parts per million (ppm). There is no direct correlation between the amount of hydrogen sulfide present in the gas phase (as measured in ppm) and the amount of hydrogen sulfide present in the crude oil liquid phase (as measured in mg of hydrogen sulfide per L of (liquid) crude oil). The ratio of these two measures, or partition coefficients, is affected by many factors, such as the temperature of the crude oil, the viscosity of the crude oil etc. As a very rough guide, 1 mg of hydrogen sulfide in 1 L of in crude oil in the liquid phase may typically result in a level of 1 to 500 ppm of hydrogen sulfide in the gas phase. Thus, the method of the first aspect may be used to treat crude oils which have hydrogen sulfide present in the gas phase in amounts of up to 100,000 ppm or more.
[0035] The method of the first aspect of the invention comprises adding an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, to a crude oil. The imine compound is added in an amount that is effective to remove or lower the amount of hydrogen sulfide present.
[0036] The particular treat rate of imine compound (i.e. amount of imine compound added to the crude oil) will depend on several factors including, for example, the amount of hydrogen sulfide present in the crude oil; the desired final level of hydrogen sulfide to be achieved; the exact imine compound being used; the desired reaction time to achieve the desired level of hydrogen sulfide; the temperature, pressure, water content, or pH of the crude oil; the mixing efficiency of the crude oil and imine compound; and the variability in any of these parameters. For example, one mole of imine compound may react with one mole of hydrogen sulfide. For example, 141 mg of tert-octylimine per L of crude oil may be required to treat 34 mg of hydrogen sulfide per L of crude oil. Higher treat ratios may be desirable in some circumstances, for example to achieve faster reaction times, or to provide a safety margin ensuring very low levels of hydrogen sulfide in the treated fuel. Lower treat ratios may be desirable in some circumstances for example to achieve a reduction in hydrogen sulfide levels without completely removing the hydrogen sulfide. For example, in some circumstances 0.1 to 100 or 0.5 to 20 or 0.8 to 10 molar equivalents of imine compound may be used.
[0037] Suitably, in the method of the first aspect some or all of the hydrogen sulfide present in the crude oil is converted to a less corrosive, reactive and/or toxic form, for example through chemical reaction with the imine compound.
[0038] The treat rate of imine compound may be from 1 to 20000 mg, for example from 1 to 5000 mg, for example from 1 to 2500 mg, such as from 1 to 1000 mg, of imine compound per L of crude oil. Typically, the treat rate of imine compound may be from 1 to 500 mg of imine compound per L of crude oil.
[0039] The treat rate/ratio of imine compound:hydrogen sulfide present in the crude oil may be from 40:1 to 1:10, preferably from 10:1 to 1:2, more preferably from 6:1 to 1:1 and most preferably from 5:1 to 2:1. Such treat rate ratios are on a weight basis. For example, a treat rate of 40 mg imine compound per L of crude oil when used to treat an amount of 1 mg of hydrogen sulfide per L of crude oil would be a ratio of 40:1.
[0040] Suitably, the method of the first aspect of the invention lowers the amount of hydrogen sulfide present in a crude oil to an amount of less than 20 mg of hydrogen sulfide per L of crude oil, preferably less than 15 mg of hydrogen sulfide per L of crude oil, such as less than 10 mg or less than 5 mg of hydrogen sulfide per L of crude oil.
[0041] By "removing hydrogen sulfide" we mean that the hydrogen sulfide is substantially removed, for example such that 1 L of the treated crude oil would contain less than 5 mg, such as less than 4 mg, preferably less than 3 mg, less than 2 mg or less than 1 mg, of hydrogen sulfide.
[0042] The hydrogen sulfide may be present in the crude oil naturally or may be liberated from sulfur based species naturally present in the crude oil.
[0043] The imine compound may also react with other sulfur species, such as HS.sup.- and S.sup.2- when present.
[0044] The imine compound used in the method of the first aspect of the invention is represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, also shown as:
##STR00001##
wherein R.sup.1 represents a (1-5C)alkyl group.
[0045] References to "an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group" are intended to refer to one or more of such imine compounds selected from this formula. Thus, mixtures of two or more imine compounds having the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2 may be used.
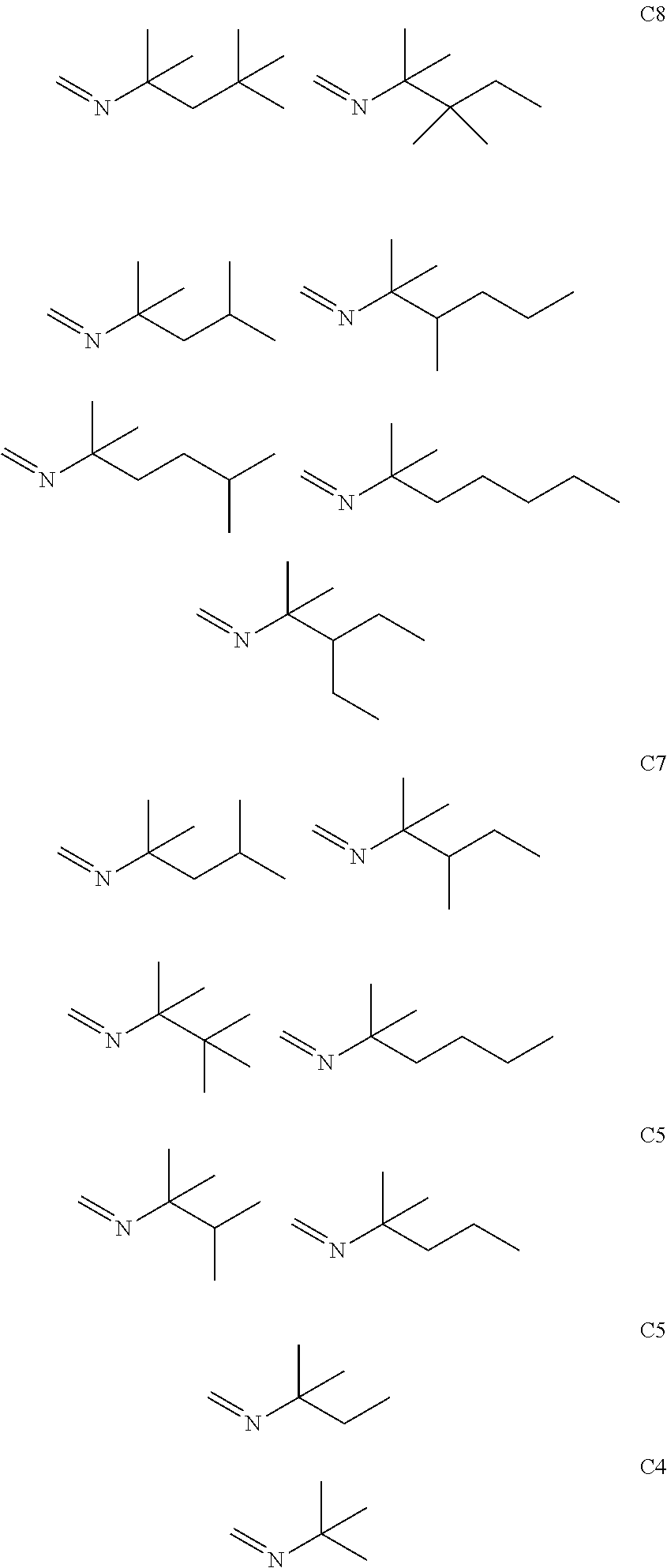
[0046] Examples of suitable imine compounds for use in the method of the first aspect of the present invention are as follows:
##STR00002##
[0047] Mixtures of imine compounds having the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2 may also be used.
[0048] Suitably, in the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, R.sup.1 represents a methyl group or a 5C alkyl group, more particularly R.sup.1 represents a 5C alkyl group. For example, the imine compound that is used in the method of the first aspect of the invention may be tert-butylimine or tert-octylimine or mixtures thereof. More particularly the imine compound that is used in the method of the first aspect of the invention may be tert-octylimine.
[0049] Suitably, the imine compound used in the method of the first aspect of the invention may be an imine compound prepared from tert-butyl amine and formaldehyde or paraformaldehyde.
[0050] Suitably, the imine compound used in the method of the first aspect of the invention may be an imine compound prepared from tert-octyl amine and formaldehyde or paraformaldehyde. Preparation of imines from amines and aldehydes is well known to those skilled in the art.
[0051] When the aforementioned imine compounds are used to remove or reduce levels of hydrogen sulfide in a crude oil, the imine compound reacts with the hydrogen sulfide, i.e. the imine compound acts as a hydrogen sulfide scavenger. The imine compounds may also react with other sulfur based species such as HS.sup.- and S.sup.2- when present (which species can liberate hydrogen sulfide). The scavenger does not remove sulfur from the crude oil, but it changes the hydrogen sulfide into a different form of sulfur compound (which form is more readily tolerated). The reaction of the imine compound with hydrogen sulfide produces at least an amine compound, i.e. an amine residue of the imine compound. For example, when the imine compound is tert-octylimine, the amine produced by reaction with hydrogen sulfide is tert-octylamine. When the imine compound is tert-butylimine, the amine produced by reaction with hydrogen sulfide is tert-butylamine.
[0052] Suitably, the method of the first aspect of the invention is for removing or lowering amounts of hydrogen sulfide in a crude oil. It has advantageously been found that when an imine compound as defined herein is used in this method, both the imine compound and the amine residue (i.e. the amine that is produced by the reaction of the imine compound with hydrogen sulfide and/or a sulfur based species) are removed during the distillation of the crude oil at temperatures below the boiling range of the kerosene cut. This means that little or none of the imine and/or amine residue thereof enters the kerosene cut. In other words, the method treats the crude oil so that the hydrogen sulfide scavenger and residues thereof do not carry over into the kerosene distillate. The inventors believe that this provides a kerosene distillate that may be used as a component of aviation turbine fuel, in which little or no atypical amines are present such that aviation turbine fuels including this distillate as a component thereof do not exhibit undesirable thermal instability as discussed above. Thus, the method of the first aspect of the invention overcomes the thermal stability issues associated with aviation turbine fuels.
[0053] It is also believed that the particular imine compounds used in the method of the first aspect of the present invention are especially effective at removing or lowering amounts of hydrogen sulfide (and optionally other sulfur based species when present) from crude oil.
[0054] A second aspect of the invention provides a method of forming a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and/or an amine residue thereof, the method comprising distilling the crude oil to form the distillate fuel component.
[0055] By the "distillate fuel component boiling in the kerosene range" we mean a distillate with a boiling range that is suitable for use as an aviation turbine fuel component. Typically, such a component may boil in the range of 150 to 300.degree. C., for example in the range of 190 to 270.degree. C.
[0056] Suitable and preferred aspects of the second aspect of the invention, for example in relation to the imine compound and amine residue thereof, are as set out above in relation to the first aspect.
[0057] In the second aspect of the invention, the crude oil contains the imine compound and/or amine residue thereof. The imine compound will typically have been added to the crude oil prior to distillation, as discussed herein. For example, the imine compound may have been added to the crude oil whilst it is being removed (for example pumped) from the ground and/or transported and/or stored and/or processed prior to distillation.
[0058] The imine compound will have been added to the crude oil in an amount that is effective to remove or reduce levels of hydrogen sulfide therein. Such treat rates are as described in relation to the first aspect.
[0059] The amine residue that may be contained in the crude oil will be present as a product of the reaction between the imine compound and the hydrogen sulfide (and optionally other sulfur containing species when present) contained in the crude oil, as discussed herein. The amine residue may be present at between 0 mol % and 100 mol % of the amount of imine compound originally added. Typically the amine residue may be present in an amount of 1 to 2500 mg, such as 1 to 1000 mg, of amine per L of crude oil. More typically, the amine residue may be present in an amount of 1 to 500 mg of amine per L of crude oil.
[0060] The method of the second aspect of the invention is advantageous as it allows the formation of a distillate fuel component boiling in the kerosene range that is substantially free of the imine compound and amine residue thereof. This is believed to provide a distillate fuel component boiling in the kerosene range that may be used as a component of aviation turbine fuel, in which little or no atypical amines are present such that aviation turbine fuels including this distillate fuel component do not exhibit undesirable thermal instability as discussed above. Thus, the method of the second aspect of the invention overcomes the observed thermal stability issues associated with atypical amines being present in aviation turbine fuels.
[0061] The methods of the invention may comprise any further suitable steps, such as mixing (for example by stirring) the crude oil and the imine compound together and optionally heating.
[0062] The present invention further provides a method of producing a distillate fuel component boiling in the kerosene range from a crude oil, wherein the crude oil contains an imine compound selected from tert-butylimine and/or tert-octylimine (preferably the imine compound is tert-octylimine), and/or an amine residue thereof selected from tert-butylamine and/or tert-octylamine (preferably the amine residue is tert-octylamine), the method comprising distilling the crude oil to form the distillate fuel component.
Composition
[0063] A third aspect of the present invention provides a composition comprising a crude oil and an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group.
[0064] As discussed above in relation to the first and second aspects, examples of suitable imine compounds for including in the composition of the third aspect of the present invention are as follows:
##STR00003##
[0065] Mixtures of imine compounds having the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2 may also be used.
[0066] Suitably, in the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, R.sup.1 represents a methyl group or a 5C alkyl group, more particularly R.sup.1 represents a 5C alkyl group. For example, the imine compound that is included in the composition of the third aspect of the invention may be tert-butylimine or tert-octylimine or mixtures thereof. More particularly, the imine compound may be tert-octylimine.
[0067] Suitably, the imine compound that is included in the composition of the third aspect of the invention may be an imine compound prepared from tert-butyl amine and formaldehyde or paraformaldehyde.
[0068] Suitably, the imine compound that is included in the composition of the third aspect of the invention may be an imine compound prepared from tert-octyl amine and formaldehyde or paraformaldehyde.
[0069] Immediately after addition of the imine compound, the composition of the third aspect comprises the imine compound in an amount that is effective to remove or lower the amount of hydrogen sulfide present. Such treat rates are as described in relation to the first aspect.
[0070] It will be appreciated that on reaction with hydrogen sulfide, the imine compound will form an amine residue as discussed herein. Thus, the composition of the third aspect may additionally comprise an amine residue of the imine compound.
[0071] Suitably, the composition of the third aspect may comprise a crude oil and from 1 to 20000 mg, for example from 1 to 5000 mg, such as from 1 to 1000 mg, preferably from 1 to 500 mg of an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and from 5000 to 1 mg, such as from 1000 to 1 mg, preferably from 500 to 1 mg of an amine residue thereof, per L of crude oil.
[0072] The particular imine compound(s) included in the composition of the third aspect of the present invention are especially effective at removing or lowering amounts of hydrogen sulfide (and optionally other sulfur based species when present) from crude oil, for example compared to other known hydrogen sulfide scavengers.
[0073] The composition of the third aspect of the invention is also considered to be advantageous because the particular imine compounds included in the composition are removed during the distillation of the crude oil at temperatures below the boiling range of the kerosene cut. This means that little or none of the imine and/or amine residue thereof enters the kerosene cut. The inventors believe that this provides a kerosene distillate that may be used as a component of aviation turbine fuel, in which little or no atypical amines are present such that aviation turbine fuels including this component do not exhibit undesirable thermal instability as discussed above. Thus, the composition of the third aspect of the invention overcomes the observed thermal stability issues associated with atypical amines being present in aviation turbine fuels.
[0074] The composition of the third aspect of the invention may comprise further additional components. Such additional components are typical crude oil additives arising from for example drilling, fracturing, completion, production, storage and transport. Such additives are known to those skilled in the art.
Distillate Fuels
[0075] A fourth aspect of the present invention provides a distillate fuel component boiling in the kerosene range obtainable by (or obtained by) a method according to the second aspect of the present invention.
[0076] Suitably, the distillate fuel component of the fourth aspect of the invention is substantially free of imine compound(s) represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group and amine residues thereof. Thus, the distillate fuel component of the fourth aspect of the present invention is particularly suitable for use as a component of an aviation turbine fuel composition, as discussed herein.
[0077] A fifth aspect of the present invention provides the use of a distillate fuel component boiling in the kerosene range according to the fourth aspect of the present invention in an aviation turbine fuel composition.
[0078] A sixth aspect of the present invention provides an aviation turbine fuel composition comprising a distillate fuel component boiling in the kerosene range according to the fourth aspect of the present invention.
[0079] The present invention further provides a method of producing a distillate fuel component boiling in the kerosene range according to the second aspect of the invention, wherein the distillate fuel component has improved thermal stability when compared to a distillate fuel component produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
[0080] The present invention further provides a distillate fuel component boiling in the kerosene range according to the fourth aspect of the invention, wherein the distillate fuel component has improved thermal stability when compared to a distillate fuel component produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
[0081] The present invention further provides an aviation turbine fuel composition according to the sixth aspect of the invention, wherein the aviation turbine fuel has improved thermal stability as measured by ASTM D3241 when compared to an aviation turbine fuel produced in the same way but from a crude oil containing an imine compound represented by the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 10 or more carbon atoms, and/or an amine residue thereof.
[0082] A seventh aspect of the present invention provides a method of removing or lowering amounts of hydrogen sulfide present during distillation of a crude oil to form a distillate fuel component boiling in the kerosene range, the method comprising adding to the crude oil an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, and distilling the crude oil to form the distillate fuel component.
[0083] An eighth aspect of the present invention provides the use of an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, to remove or lower the amount of hydrogen sulfide in a crude oil.
[0084] A ninth aspect of the present invention provides the use of an imine compound represented by the formula R.sup.1--C(CH.sub.3).sub.2--N.dbd.CH.sub.2, wherein R.sup.1 represents a (1-5C)alkyl group, in the preparation of a distillate fuel component boiling in the kerosene range.
[0085] For example, R.sup.1 may represent a methyl group or a 5C alkyl group, more particularly R.sup.1 may represent a 5C alkyl group.
[0086] The preferred features of the fourth, fifth, sixth, seventh, eighth and ninth aspects are as defined in relation to the first, second and third aspects.
BRIEF DESCRIPTION OF DRAWINGS
[0087] For a better understanding of the invention, and to show how exemplary embodiments of the same may be carried into effect, reference will be made, by way of example only, to the accompanying diagrammatic Figures, in which:
[0088] FIG. 1 shows the amine content found in distillation fractions treated with Scavenger C in Example 1, with % w/w of the Scavenger C on the y axis and the distillation fraction (in .degree. C.) on the x axis;
[0089] FIG. 2 shows the amine content found in distillation fractions treated with tert-octylamine in Example 1, with % w/w of the tert-octylamine on the y axis and the distillation fraction (in .degree. C.) on the x axis;
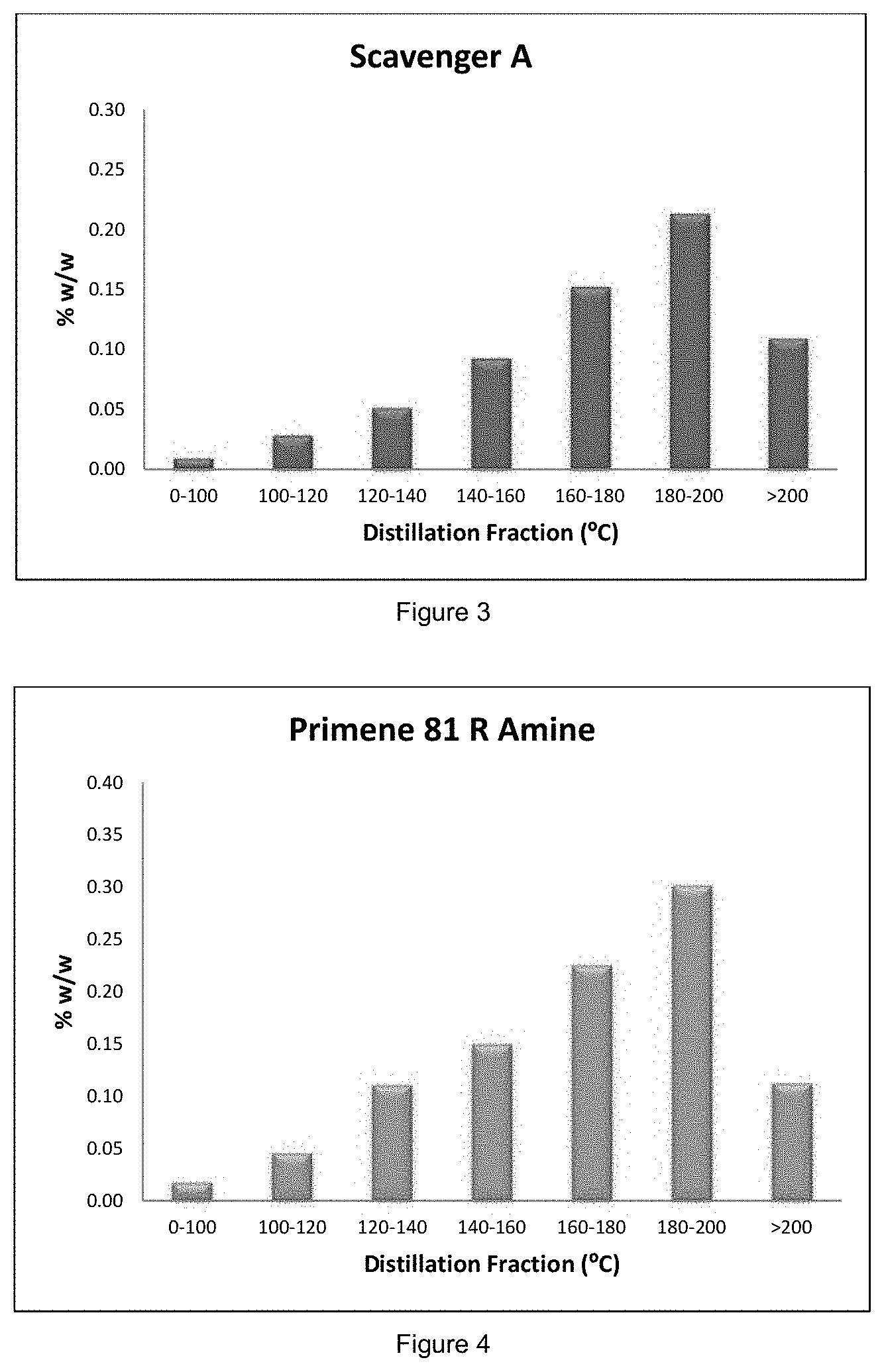
[0090] FIG. 3 shows the amine content found in distillation fractions treated with Scavenger A in Example 1, with % w/w of the Scavenger A on the y axis and the distillation fraction (in .degree. C.) on the x axis;
[0091] FIG. 4 shows the amine content found in distillation fractions treated with Primene 81R Amine in Example 1, with % w/w of the Primene 81R on the y axis and the distillation fraction (in .degree. C.) on the x axis;
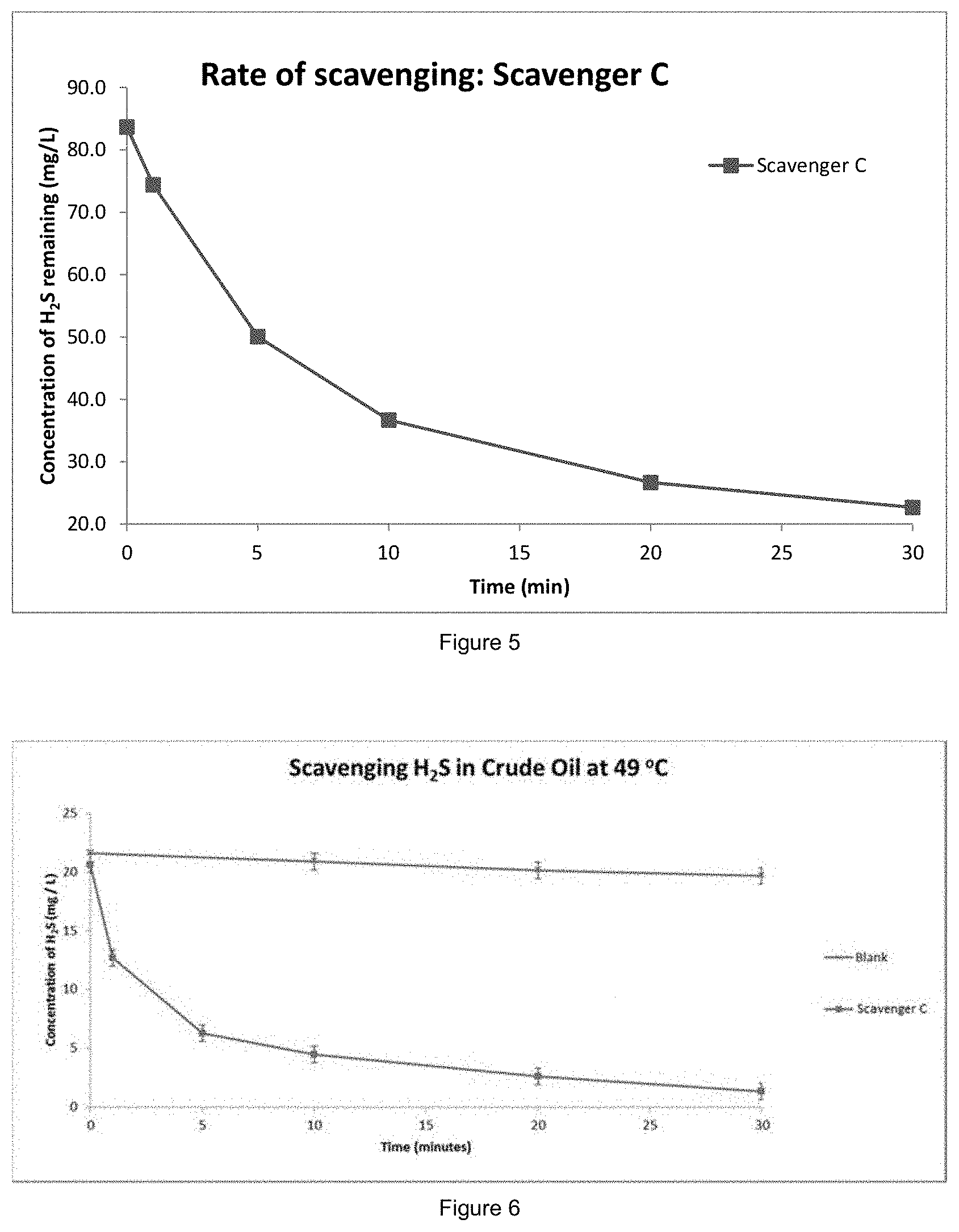
[0092] FIG. 5 shows hydrogen sulfide scavenging efficiency for Scavenger C according to Example 2, with the concentration of hydrogen sulfide remaining (in mg/L) on the y axis and time (in minutes) on the x axis; and
[0093] FIG. 6 shows hydrogen sulfide scavenging efficiency for Scavenger C according to Example 3, with the concentration of hydrogen sulfide remaining (in mg/L) on the y axis and time (in minutes) on the x axis.
EXAMPLES
[0094] The invention will now be described with reference to the following non-limiting examples.
[0095] Scavenger A is a terminal imine formed by the reaction of Primene 81R Amine and formaldehyde (or paraformaldehyde) and is a known commercial hydrogen sulfide scavenger. Primene 81R is a primary aliphatic amine in which the amino nitrogen is linked to a tertiary carbon and the total number of carbon atoms is in the C12-C14 range. Thus Scavenger A is a compound of the formula R.sup.2--N.dbd.CH.sub.2, wherein R.sup.2 represents an alkyl group having 12-14 carbon atoms.
Preparative Example Scavenger A (Comparative)
[0096] Primene 81R amine (10 g, 50.3 mmol) was dissolved in toluene (50 ml). Paraformaldehyde (1.81 g, 60.3 mmol) was added and the mixture heated to reflux for 1 hour with a Dean Stark trap in place to capture the water produced. After cooling to room temperature, the organic phase was concentrated in vacuo to afford Scavenger A (9.52 g, 45.1 mmol, 90%) as a yellow oil.
Preparative Example Scavenger C (Inventive; Tert-Octylimine)
[0097] Paraformaldehyde (0.94 g, 31.1 mmol) was added to in EtOAc (20 ml) to form a suspension. tert-Octylamine (4.03 g, 31.1 mmol) was added and the mixture stirred at room temperature for 1 hour, then at 60.degree. C. for 1 hour. After cooling to room temperature, the aqueous phase was separated and the organic phase dried over MgSO.sub.4 and concentrated in vacuo to afford tert-octylimine, Scavenger C (3.43 g, 24.3 mmol, 78%) as a colourless oil.
Example 1: Distillation Profile of Nitrogen-Containing Oil Additives
[0098] A mixture of diesel (100 ml), aviation turbine fuel (100 ml) and gasoline (50 ml) was placed in a 500 ml round bottom flask. A stirrer bar was added followed by an additive to be tested (0.3 ml). Distillation equipment, comprising a thermometer, water-cooled condensing tube and receiver vessel were then attached to the round bottom flask. The system was purged with nitrogen and heat was applied to the round bottom flask while the contents were stirred. Separate fuel distillation fractions were then collected based on the temperature reading of the thermometer. Fractions collected corresponded to the following temperature ranges 0-100.degree. C., 100-120.degree. C., 120-140.degree. C., 140-160.degree. C., 160-180.degree. C., 180-200.degree. C. The non-distilled residue was collected and classed as the >200.degree. C. fraction. Each fraction was then tested to determine the amine/nitrogen content. Results are shown in FIGS. 1 to 4.
[0099] The Additives tested were: Scavenger C, tert-octylamine, Scavenger A, and Primene 81R Amine.
[0100] FIGS. 1 and 2 show that Scavenger C and tert-octylamine (the potential nitrogen-containing constituents present when treating oil-products with Scavenger C) are found solely in the low temperature (i.e. <200.degree. C.) distillation fractions. Consequently, only a small amount of these materials would be expected within higher temperature cuts. In contrast, Scavenger A and Primene 81 R Amine (the potential nitrogen-containing constituents present when treating oil-products with Scavenger A) were found in all distillation fractions (see FIGS. 3 and 4). Amines from Scavenger A and Primene 81 R Amine would therefore be considerably more likely to be present within higher temperature cuts, including those corresponding to aviation turbine fuel.
Example 2: Hydrogen Sulfide Scavenging Efficiency
[0101] Caromax 20 (20 ml) was placed into a reaction vessel containing a stirrer bar and the system sealed. A stock solution of Caromax 20/H.sub.2S (4 ml, about 525 mg/L H.sub.2S) was added to the reaction vessel and the mixture heated to 75.degree. C. with stirring. Once the temperature had stabilised the time=0 liquid H.sub.2S content was determined (in mg H.sub.2S/L). Hydrogen sulfide Scavenger C (415 mg/L) was then injected into the system and the liquid H.sub.2S content determined at the following time points after injection; 1, 5, 10, 20 and 30 minutes. The results are shown in FIG. 5.
[0102] FIG. 5 shows that Scavenger C reduces the amount of hydrogen sulfide in the system.
Example 3: Hydrogen Sulfide Scavenging Efficiency
[0103] Crude oil (20 ml) was placed into a reaction vessel containing a stirrer bar and the system sealed. A stock solution of Caromax 20/H2S (1.3 ml, about 525 mg/L H2S) was added to the reaction vessel and the mixture heated to 49.degree. C. with stirring. Once the temperature had stabilised the time=0 liquid H2S content was determined (in mg H2S/L). Hydrogen sulfide Scavenger C (525 mg/L) was then injected into the system and the liquid H2S content determined at the following time points after injection; 1, 5, 10, 20 and 30 minutes. The results are shown in FIG. 6.
[0104] FIG. 6 shows that Scavenger C reduces the amount of hydrogen sulfide in the system.
Example 4: Hydrogen Sulfide Scavenging Efficiency
[0105] A series of tests were performed to study the effectiveness of Scavenger C. In these tests, 2 ml of a stock solution containing 0.6 mg/ml of Na.sub.2S in MeOH was added to 8 ml of Caromax and the vessel sealed. Hydrogen sulfide was generated in situ by injecting 0.07 ml of 0.5M HCl (2 molar equivalents). Thus, the solution contained approximately 50 mg/L of hydrogen sulfide. Scavenger C was then injected in an amount as shown in Table 1. The mixture was heated to 75.degree. C. for 30 minutes and then allowed to cool. The presence of hydrogen sulfide remaining was assessed using a colourimetric test which tests positive if >1 mg/L of hydrogen sulfide is present. Thus, a positive test indicates a failure to reduce the hydrogen sulfide concentration to <1 mg/L.
[0106] Multiple tests were performed over an extended period of time (several weeks) using different batches of Scavenger C and the results are summarised in Table 1.
TABLE-US-00001 TABLE 1 Amount of Total Scavenger C added number of Molar Equivalents tests Passes Fails % pass 0.5 31 24 7 77 1.0 40 40 0 100 2.0 36 36 0 100 5.0 8 8 0 100 10.0 2 2 0 100 15.0 2 2 0 100
[0107] The results show that at treat rates at or above 1 mole of Scavenger C per mole of hydrogen sulfide, all tests resulted in hydrogen sulfide levels <1 mg/L.
* * * * *
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.