Method Of Separating Metal From Metallic Starting Materials Using A Hydrothermal Reactor System
Bobe; Iulian ; et al.
U.S. patent application number 16/537753 was filed with the patent office on 2019-11-28 for method of separating metal from metallic starting materials using a hydrothermal reactor system. The applicant listed for this patent is Tyton Biosciences, LLC. Invention is credited to Florin G. Barla, Iulian Bobe, Conor Hartman, Jeremy Jones, Todd Showalter, Hsun-Cheng Su.
| Application Number | 20190360070 16/537753 |
| Document ID | / |
| Family ID | 63169638 |
| Filed Date | 2019-11-28 |

| United States Patent Application | 20190360070 |
| Kind Code | A1 |
| Bobe; Iulian ; et al. | November 28, 2019 |
METHOD OF SEPARATING METAL FROM METALLIC STARTING MATERIALS USING A HYDROTHERMAL REACTOR SYSTEM
Abstract
The presently disclosed subject matter is directed to a method of hydrothermally treating metallic packaging, metallic labeling material, and scrap metal items. Particularly, the disclosed method initially comprises providing a metallic starting material. The metallic starting material is introduced to a reactor and processed at elevated temperature and/or pressure for a desired amount of time. As a result, the metallic portion of the starting material separates into a distinct layer that can be removed from the remainder of the starting material.
| Inventors: | Bobe; Iulian; (Danville, VA) ; Barla; Florin G.; (Danville, VA) ; Hartman; Conor; (Durham, NC) ; Su; Hsun-Cheng; (Chapel Hill, NC) ; Jones; Jeremy; (Danville, VA) ; Showalter; Todd; (Danville, VA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 63169638 | ||||||||||
| Appl. No.: | 16/537753 | ||||||||||
| Filed: | August 12, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| PCT/US18/18496 | Feb 16, 2018 | |||
| 16537753 | ||||
| 62459718 | Feb 16, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C22B 21/0023 20130101; C22B 21/0069 20130101; C22B 7/006 20130101 |
| International Class: | C22B 21/00 20060101 C22B021/00; C22B 7/00 20060101 C22B007/00 |
Claims
1. A method of separating metal from a metallic starting material, the method comprising: loading the metallic starting material and a solvent into at least one reactor; optionally adding catalyst, co-solvent, or both to the reactor; applying heat and pressure to the starting material until the metal is separated from the metallic starting material, and physically separating the metal from the starting material.
2. The method of claim 1, wherein the metallic starting material comprises a metal and a substrate.
3. The method of claim 2, wherein the substrate comprises paper, resin, or combinations thereof.
4. The method of claim 3, wherein the resin is selected from polyethylene, polypropylene terephthalate, nylon, polyester, or combinations thereof.
5. The method of claim 1, wherein the starting material has a thickness of about 5-200 micron.
6. The method of claim 1, wherein the metal is tin foil, aluminum foil, tin, copper, gold, silver, a laminate of two metals, a metallized film, or combinations thereof.
7. The method of claim 1, wherein the metal is an insulated wire.
8. The method of claim 7, wherein the insulated wire is selected from insulated copper wire, electric house wire, thermoplastic-sheathed cable, aluminum jacketed Bronx cable with copper interior, and combinations thereof.
9. The method of claim 1, wherein the solvent is selected from water, buffer, or combinations thereof.
10. The method of claim 1, wherein the co-solvent is selected from one or more polar or non-polar organic solvents or oils.
11. The method of claim 1, wherein the reactor is a batch, semi-continuous, or continuous hydrothermal reactor.
12. The method of claim 1, wherein the catalyst is added at a concentration of about 0-1 weight % of the total weight of the input material.
13. The method of claim 1, performed at a temperature of about 170.degree. C. to about 310.degree. C.
14. The method of claim 1, performed at a pressure of about 150 to 1500 psi.
15. The method of claim 1, wherein the ratio of starting material to water in the reactor is about 1:1 to about 1:200.
16. The method of claim 1, wherein the reaction time is about 5 minutes to about 3 hours.
17. The method of claim 1, wherein the physical separation comprises air density separation.
18. The method of claim 1, wherein about 50-100 wt % of the metal in the starting material is separated from the starting material, based upon the total weight of the weight in the starting material.
19. The metal separated by the method of claim 1.
20. The metal of claim 19, selected from aluminum foil, tin, copper, gold, silver, or combinations thereof.
Description
CROSS REFERENCE TO RELATED APPLICATION
[0001] This application is a continuation of International Patent Application No. PCT/US18/18496, filed on Feb. 16, 2018, which claims priority to U.S. Provisional Patent Application No. 62/459,718, filed on Feb. 16, 2017, the entire contents of which are all hereby incorporated herein by reference.
FIELD OF INVENTION
[0002] The presently disclosed subject matter generally relates to methods of separating metal from metallic starting materials, such as packaging material (e.g., metal foil paper, aluminum bag packages, and the like) and scrap metal items (e.g., copper wire, insulated copper cable, power strips, motherboards and the like) using a hydrothermal reactor system.
BACKGROUND
[0003] Metallic materials have many advantageous properties, including versatility, formability, gas impermeability, relative chemical inertness, easily printability, and recyclability. These properties, together with constant innovation and technological evolution, have enabled metallic materials to extend their packaging applications to serve the demand of modern society and the increasing population. For example, aluminum has very attractive properties when used as a packaging material, including lightness, gloss, and considerable impermeability to gases, water vapor, and light. Further, aluminum is a 100% recyclable and sustainable metal. Aluminum is one of the only materials in the consumer and industrial waste streams capable of saving money when recycled. Namely, recycling aluminum saves more than 90% of the energy required to produce a comparable amount of metal from raw materials. For these reasons, the use of recycled aluminum is an increasing trend in the industry. To this end, nearly 40% of the North American aluminum supply has been created through the recycling process (i.e., as a secondary production). For example, aluminum beverage cans can be easily heated in a furnace to 650.degree. C.-850.degree. C. to produce molten aluminum for further recycling processes.
[0004] However, not all aluminum packaging and/or labels are suitable for use with standard recycling processes. For example, it is common practice to apply aluminum foil labels to containers (e.g., glass bottles) to provide information about the manufacturer and/or the contents of the container. Standard label materials typically include an aluminum foil component laminated to other materials, such as plastics and/or paper. In conventional labeling processes, labels are cut from a foil paper roll and are applied to the surface of a container. However, cutting of the labels generates large amounts of unused scrap metal foil material. The scrap material is typically not recycled because it includes a mixture of paper, foil, plastic, glue, and the like, which decreases the efficiency of aluminum recovery through existing thermolysis methods. Thin metal typically oxidizes quickly and "flashes off", transforming into gas. It would therefore be beneficial to provide a method of separating the metal from the scrap material for recycling and/or repurposing, so that it can be baled into blocks before entering the recycling process thereby enabling the recovery of thin metal waste material.
SUMMARY
[0005] In some embodiments, the presently disclosed subject matter is directed to a method of separating metal from a metallic starting material. The method comprises loading the metallic starting material and a solvent into at least one reactor, and optionally adding catalyst, co-solvent, or both to the reactor. The method further comprises applying heat and pressure to the starting material until the metal is separated from the metallic starting material, and physically separating the metal from the starting material.
[0006] In some embodiments, the presently disclosed subject matter is directed to a method of recycling metal from a metallic starting material. Particularly, the method comprises loading the metallic starting material and a solvent into at least one reactor and optionally adding catalyst, co-solvent, or both to the reactor. The method further comprises applying heat and pressure to the starting material until the metal is separated from the metallic starting material, physically separating the metal from the starting material, and recycling the separated metal.
[0007] In some embodiments, the presently disclosed subject matter is directed to a system comprising a feedstock source comprising a metallic starting material, and a reactor in supply communication with the feedstock source, wherein the reactor is configured to apply heat and pressure to the starting material until metal is separated from the metallic starting material. The system further comprises a computer that controls the reactor and is in communication with at least one sensor, wherein the computer is configured to receive data from the sensor and determine a reaction time when the metal is separated from the starting material based on the received data.
[0008] In some embodiments, the metallic starting material is scrap metallic packaging material. In some embodiments, the starting material comprises a metal and a substrate (i.e., paper, resin, or combinations thereof). In some embodiments, the resin is selected from polyethylene, polypropylene terephthalate, nylon, or combinations thereof. In some embodiments, the starting material has a thickness of about 5-200 micron. In some embodiments, the metal is selected from tin foil, aluminum foil, tin, copper, gold, silver, a laminate of two or more metals (or at least one metal and at least one polymeric material, or two or more polymeric materials), a metallized film, a wire or cord, or combinations thereof. In some embodiments, the metal can be an insulated wire selected from insulated copper wire, electric house wire (PVC wire, coper wire coil), thermoplastic-sheathed cable, aluminum jacketed BX (Bronx cable) with copper interior, and combinations thereof.
[0009] In some embodiments, the solvent is selected from water, buffer, or combinations thereof. In some embodiments, the co-solvent is selected from one or more polar or non-polar organic solvents or oils. In some embodiments, the catalyst is added at a concentration of about 0-1 weight % of the total weight of the input material.
[0010] In some embodiments, the reactor is a batch, semi-continuous, or continuous hydrothermal reactor.
[0011] In some embodiments, the reactor is a hydrothermal reactor. In some embodiments, the reactor is a batch, or semi-continuous, or continuous hydrothermal reactor. In some embodiments, the disclosed method is performed at a temperature of about 170.degree. C. to about 310.degree. C., at a pressure of about 150 to 1500 psi, or both. In some embodiments, the ratio of starting material to water in the reactor is about 1:1 to about 1:200. In some embodiments, a catalyst including (but not limited to) sodium chloride (NaCl) is added to the reactor at a concentration of about 0-1 weight % of the total input material (e.g., starting material and solvent).
[0012] In some embodiments, a co-solvent is added in a concentration of about 50-100 wt % of the total input material. The co-solvent can include common organic solvents, such as polar or non-polar organic solvents including (but not limited to) methanol and/or DMSO or oils (e.g., vegetable cooking oil and/or waste cooking oils). In some embodiments, the disclosed method is performed for a reaction time of about 5 minutes to about 3 hours.
[0013] In some embodiments, the physical separation comprises the use of an air density separation. In some embodiments, about 50-100 weight % of the metal in the starting material is extracted (e.g., separated from the starting material), based on the total weight of the starting material.
[0014] In some embodiments, the metallic starting material comprises a metal and a substrate.
[0015] In some embodiments, the presently disclosed subject matter includes the metal separated by the disclosed method (e.g., aluminum foil, tin, copper, gold, silver, or combinations thereof).
BRIEF DESCRIPTION OF THE DRAWINGS
[0016] The previous summary and the following detailed description are to be read in view of the drawings, which illustrates particular exemplary embodiments and features as briefly described below. The summary and detailed descriptions, however, are not limited to only those embodiments and features explicitly illustrated.
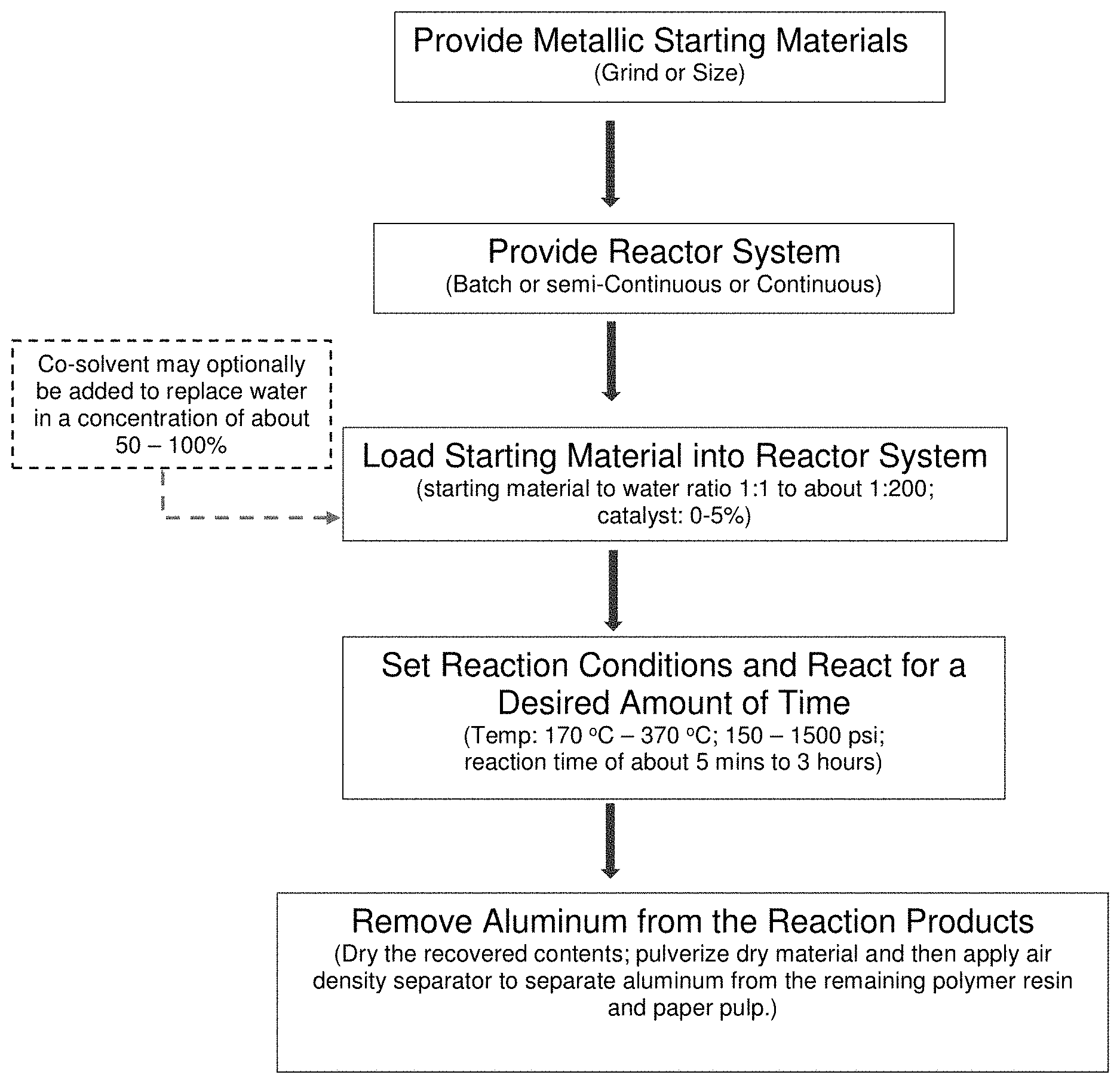
[0017] FIG. 1 is a schematic illustrating one method of performing the disclosed method in accordance with some embodiments of the presently disclosed subject matter.
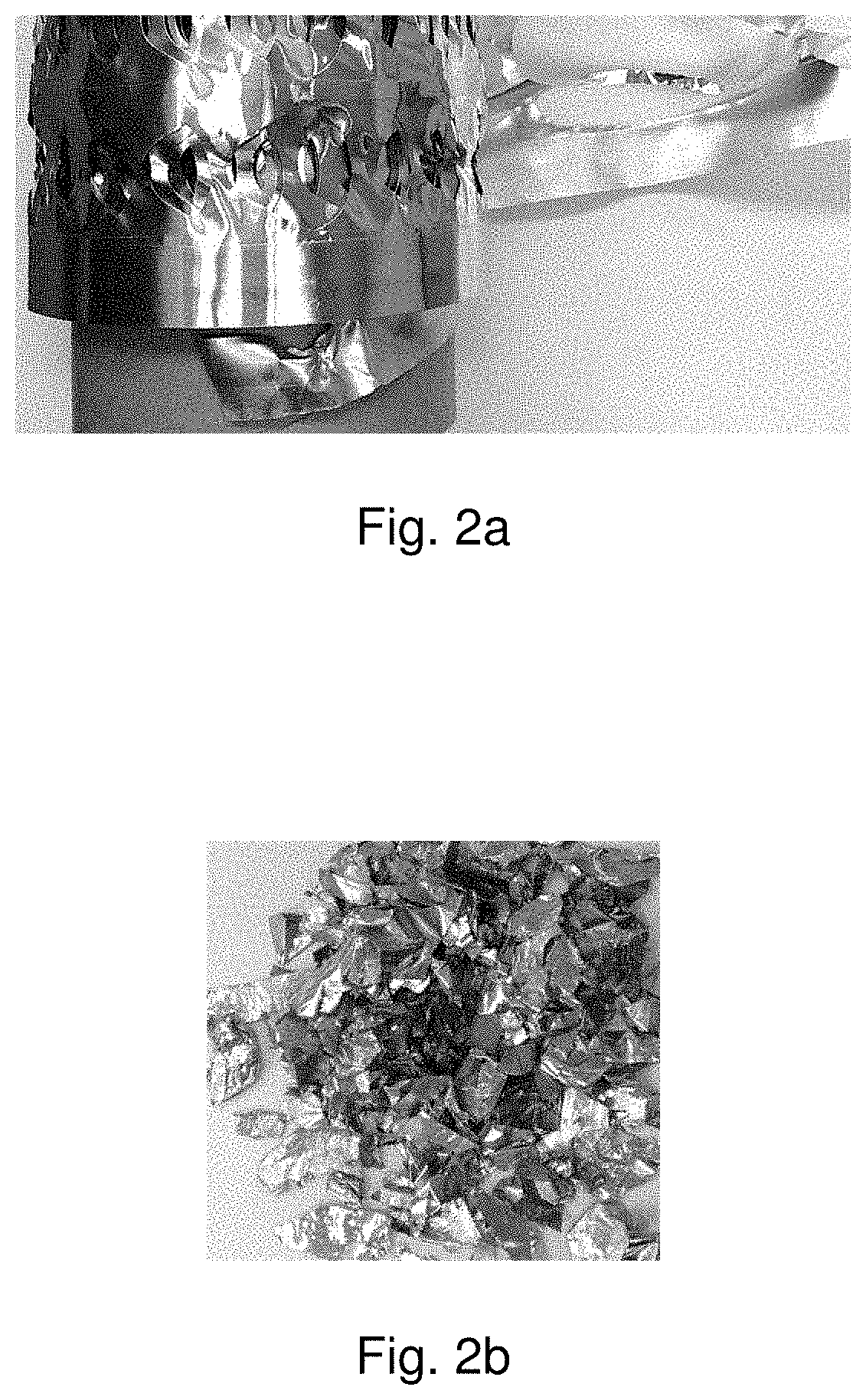
[0018] FIG. 2a is a photograph of scrap aluminum foil paper material that can be treated according to some embodiments of the presently disclosed subject matter.
[0019] FIG. 2b is a photograph of recovered aluminum after treatment according to some embodiments of the presently disclosed subject matter.
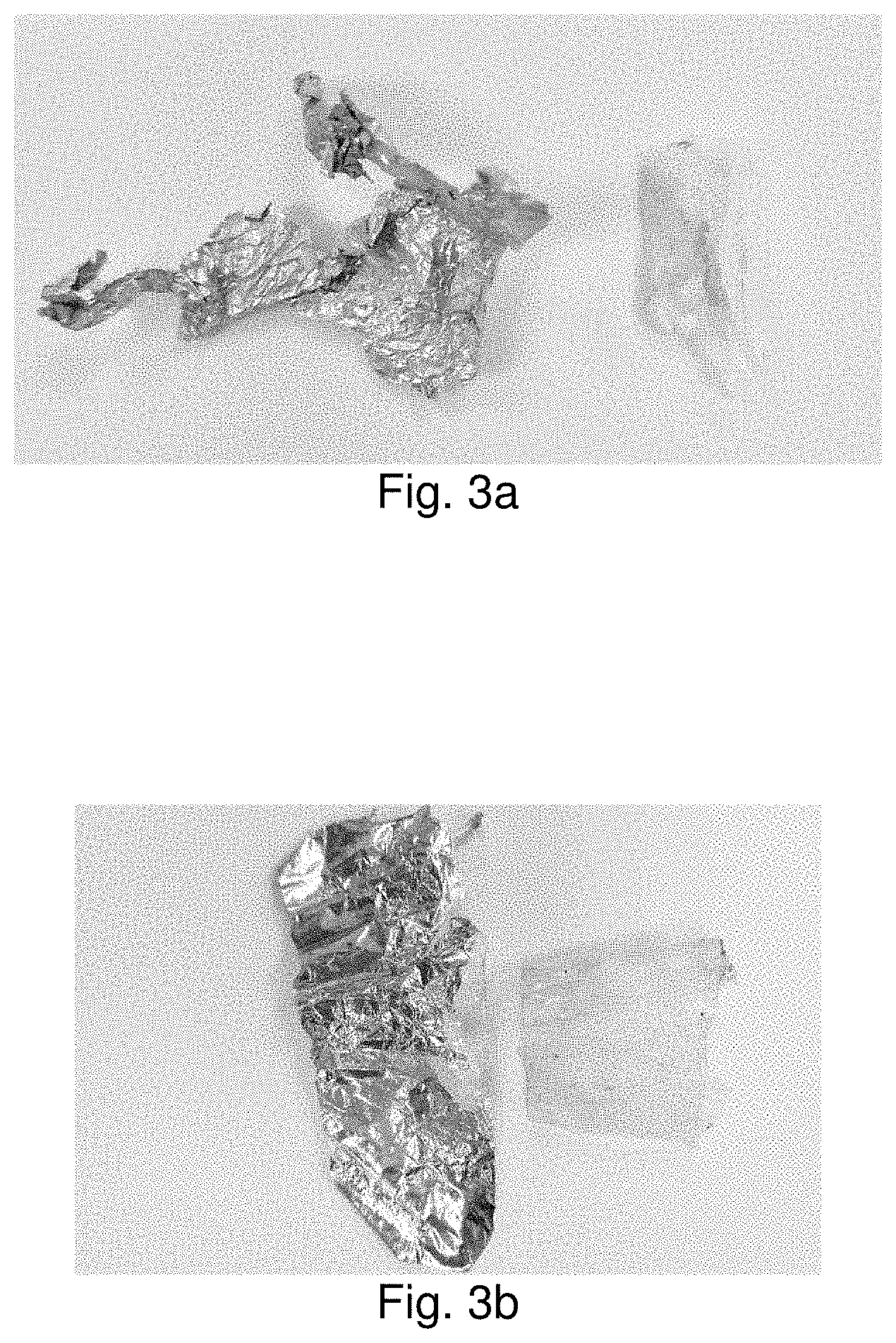
[0020] FIGS. 3a and 3b are photographs of recovered aluminum when co-solvent conditions were used.
DETAILED DESCRIPTION
[0021] Unless otherwise defined, all technical terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure belongs.
[0022] Following long-standing patent law convention, the terms "a," "an," and "the" refer to one or more when used in this application, including the claims. Thus, for example, reference to "a metal" includes a plurality of metals, unless the context clearly is to the contrary.
[0023] For the purposes of this specification and appended claims, the term "about" when used in connection with one or more numbers or numerical ranges, should be understood to refer to all such numbers, including all numbers in a range and modifies that range by extending the boundaries above and below the numerical values set forth. The recitation of numerical ranges by endpoints includes all numbers, e.g., whole integers, including fractions thereof, subsumed within that range (for example, the recitation of 1 to 5 includes 1, 2, 3, 4, and 5, as well as fractions thereof, e.g., 1.5, 2.25, 3.75, 4.1, and the like) and any range within that range. In some embodiments, the term "about" includes variations of up to +/-20%, +/-15%, +/-10%, +/-5%, +/-1%, or +/-0.1% of a recited value.
[0024] Throughout this specification and the claims, the terms "comprise," "comprises," and "comprising" are used in a non-exclusive sense, except where the context requires otherwise. Likewise, the term "include" and its grammatical variants are intended to be non-limiting, such that recitation of items in a list is not to the exclusion of other like items that can be substituted or added to the listed items.
[0025] The descriptions herein are presented with sufficient details to provide an understanding of one or more particular embodiments of broader inventive subject matters. These descriptions expound upon and exemplify particular features of those particular embodiments without limiting the inventive subject matters to the explicitly described embodiments and features. Considerations in view of these descriptions will likely give rise to additional and similar embodiments and features without departing from the scope of the inventive subject matters. Although the term "step" may be expressly used or implied relating to features of processes or methods, no implication is made of any particular order or sequence among such expressed or implied steps unless an order or sequence is explicitly stated.
[0026] FIG. 1 is a schematic illustrating one embodiment of a method for hydrothermally treating a metallic starting material (such as metal foil paper). Particularly, the disclosed method initially comprises providing a metallic starting material. The term "metallic starting material" includes any material that includes one or more metals and at least one substrate. For example, in some embodiments, the starting material comprises a scrap metallic material, such as (but not limited to) a metal foil, copper wire, insulated copper cable with rubber jacket, silver products, and e-waste (e.g., computer motherboards and the like). The term "metal" or "metallic" as used herein refers to alkali metals, alkali earth metals, transition metals, or mixtures thereof. For example, in some embodiments, the term "metal" can include aluminum, tin, copper, gold, silver, and the like.
[0027] In some embodiments, the metallic starting material can include a metal foil. The term "metal foil" as used herein refers to a thin, flexible sheet of any suitable metal. In some embodiments, the metal material can comprise a packaging foil, such as tin foil, aluminum foil, laminate, and/or metallized film. In some embodiments, aluminum foil is preferred. Suitable input waste recycling materials can include (but are not limited to) aluminum food packages, blister packaging, cap seal liners, tetra packages (polyethylene-polyethylene-aluminum-polyethylene-paper-polyethylene), aluminum containers, aluminum beverage packages, wine caps (polylaminate capsules), aluminum pump sprays (for lotion, perfume, essential oils), aluminum haute couture, aluminum cosmetic packages, and the like.
[0028] The starting material further comprises a substrate, such as paper, plastic, and/or resin. The term "substrate" as used herein refers to any suitable material that can be used to support the metallic component of the starting material. For example, suitable substrates can include paper made from wood pulp, cotton, or synthetic wood-free fibers and board. The paper can be coated, uncoated, calendared, or machine glazed. In some embodiments, the resin can be polyethylene, polypropylene, polyethylene terephthalate, nylon, or combinations thereof.
[0029] The metallic component and the substrate component of the starting material can be adhered together using any method known or used in the art. For example, in some embodiments, pressure sensitive adhesives can be used. The starting material can further include a variety of other materials, including (but not limited to) inks, dyes, binders, polyethylene, polypropylene, or the like to be used as adhesives.
[0030] One example of a suitable starting material is aluminum foil paper rolls produced by Avery Dennison Corporation (Glendale, Calif., United States of America). Thus, in some embodiments, the starting material can be a metallic foil-paper laminate or a metallized paper product, such as the type used to produce bottle labels.
[0031] The starting material can have any desired thickness. For example, in some embodiments, the starting material can have a thickness of about 5-200 microns, such as about 10-20, 10-30, 10-40, 10-50, 10-75, 10-100, 10-150 or 10-200 microns. The starting material can be in any of a wide variety of forms, such as rolls, stacks of sheets, and the like.
[0032] In some embodiments, the starting material is scrap, used for label making. The labels or other items are cut from the starting material and the remainder (scrap) material is processed using the disclosed method. In some embodiments, the starting material can comprise labels that have been removed from bottles after use. In some embodiments, the starting material can comprise discarded packaging for various consumer products.
[0033] As shown in FIG. 1, at least one reactor is provided in the disclosed system and method. The term "reactor" as used herein refers to a device that can be used for any number of chemical processes involving a starting material. In some embodiments, the reactor comprises a hydrothermal reactor. The term "hydrothermal" as used herein refers to an aqueous system under pressure and increased temperature, typically near or above the critical point of water (374.degree. C., 22.1 MPa). Thus, the reactor can provide hydrothermal conditions, such as (but not limited to) a batch reactor, semi-continuous, or continuous reactor. In some embodiments, a batch reactor is preferred.
[0034] The disclosed method comprises transferring the starting material and one or more solvents to the reactor and processing for a desired amount of time. In some embodiments, the solvent can be water, buffer, and the like. Suitable buffers can include any buffer known or used in the art, including (but not limited to) TBS, PBS, BIS-TRIS, HEPES, HEPES sodium salt, MES, MES sodium salt, MOPS, MOPS sodium salt, sodium chloride, ammonium acetate, ammonium formate, ammonium phosphate monobasic, ammonium tartrate dibasic, bicarbonate, citrate, formic acid, imidazole, magnesium acetate, magnesium formate, potassium acetate, potassium acetate, potassium acetate, potassium citrate tribasic, potassium formate, potassium phosphate, potassium sodium tartrate, propionic acid, STE, STET, sodium acetate, sodium formate, sodium phosphate, sodium tartrate, TNT, TRIS glycine, TRIS acetate-EDTA, triethylammonium phosphate, trimethylammonium acetate, trimethylammonium phosphate, and/or TRIS-EDTA.
[0035] In some embodiments, a catalyst can be added to the starting material. The term "catalyst" as used herein refers to any substance that increases the rate of a chemical reaction without itself being consumed in the reaction. Suitable catalysts can include (but are not limited) sodium chloride (NaCl). The catalyst can be added at a concentration of about 0-1 weight % of the total input material (starting material, catalyst, solvent, and optionally co-solvent).
[0036] In some embodiments, a co-solvent can be added to the starting material. Suitable co-solvents can include organic solvents, such as polar or non-polar organic solvents, including (but not limited to) methanol and/or DMSO, oils. Suitable organic solvents can include (but are not limited to) alcohols, alkanes, glycol ethers, ketones, hydrocarbons, amines, organic acids, and the like.
[0037] In some embodiments, a co-solvent is added in a concentration of about 50-100 wt %, based on the total weight of the input materials (starting material, solvent, co-solvent, and optionally catalyst).
[0038] Thus, in some embodiments, the reactor input materials can comprise the starting materials, solvent, co-solvent, and/or catalyst.
[0039] In some embodiments, the starting material can be treated in the reactor at a temperature of about 170.degree. C. to 310.degree. C. Thus, the temperature can range from about 170-310.degree. C., 180-300.degree. C., 190-290.degree. C., 200-280.degree. C., 210-270.degree. C., 220-260.degree. C. or 230-250.degree. C. The temperature can therefore be at least about (or no more than about) 170, 180, 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300 or 310.degree. C.
[0040] In some embodiments, the starting material can be treated in the reactor at a pressure of about 150 to 1500 psi, such as about 200-1400, 300-1300, 400-1200, 500-1100, 600-1000, or 700-900 psi. Thus, the pressure can be at least about (or no more than about) 150, 200, 250, 300, 350, 400, 450, 500, 550, 600, 650, 700, 750, 800, 850, 900, 950, 1000, 1050, 1100, 1150, 1200, 1250, 1300, 1350, or 1400 psi.
[0041] In some embodiments, the ratio of input materials to solvent (e.g., water) is about 1:1 to 1:200, such as about 1:5 to 1:175, 1:10 to 1:150, 1:25 to 1:100, or 1:50 to 1:100. Thus, the ratio of input materials to solvent can be at least about (or no more than about) 1:1, 1:5, 1:10, 1:20, 1:30, 1:40, 1:50, 1:60, 1:70, 1:80, 1:90, 1:100, 1:110, 1:120, 1:130, 1:140, 1:150, 1:160, 1:170, 1:180, 1:190, or 1:200.
[0042] In some embodiments, the reactor reaction time is about 5 minutes to about 3 hours, such as about 10 minutes to 2.5 hours, 15 minutes to 2 hours, 30 minutes to 1.5 hours, or 35 minutes to 1 hour. Thus, the reaction time can be at least about (or no more than about) 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, or 60 minutes. In some embodiments, the reaction time can be at least about (or no more than about) 1, 1.25, 1.5, 1.75, 2, 2.25, 2.5, 2.75, or 3 hours.
[0043] In the reactor, the metallic portion of the starting material is separated from the remainder of the starting material as a result of the temperature and/or pressure received from the reactor. For example, in some embodiments, the metallic packaging materials form a liquid/solid mixture after hydrothermal treatment that can be easily separated from the remainder of the starting material using standard separation methods, including (but not limited to) air density separation processes (as indicated in the FIG. 1). The term "air density separation" refers to the separation of dense materials from less-dense materials. In some embodiments, the air density separator includes an air stream that carries material through a separator, where heavier materials fall into a trap under the weight of gravity.
[0044] As a result, each solid constituent is separated and metallic material (e.g., aluminum flakes) can be recovered from the remaining materials (e.g., polymer resin (such as polyester), paper pulp). In some embodiments, the pure metal can be baled into block for further recovery processing, as would be known to those of ordinary skill in the art.
[0045] After removal of the solid constituent portions (metal, polyester, paper pulp, etc), the remainder of the starting material (paper, adhesive, dye, plastic, etc.) can in some embodiments be flushed with water down a standard drain for disposal.
[0046] In some embodiments, the starting material can be pre-processed prior to treating in the reactor to reduce particle size. Particularly, the starting material can be reduced to uniform particle size by shredding, cutting, or any other method known or used in the art. In these embodiments, the mixture of glue, plastic, and/or fiber can function to protect the metal material during hydrothermal processing. As a result, the metal component will not react as quickly and become lost in the liquid phase.
[0047] Thus, the presently disclosed subject matter advantageously provides a method of extracting metal from a metallic substrate starting material. In some embodiments, about 40-100 wt % of the metal in the starting material can be extracted, based on the total weight of the starting material. For example, at least about (or no more than about) 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 99, or 100 weight percent of the metal can be extracted, based on the total weight of the metal in the starting material.
EXAMPLES
[0048] The following Examples have been included to provide guidance to one of ordinary skill in the art for practicing representative embodiments of the presently disclosed subject matter. In light of the present disclosure and the general level of skill in the art, those of skill can appreciate that the following Examples are intended to be exemplary only and that numerous changes, modifications, and alterations can be employed without departing from the scope of the presently disclosed subject matter.
Example 1
Separation of Aluminum from Aluminum Paper Rolls
[0049] A sample of leftover metallic aluminum foil paper roll (after labels had been removed) was obtained. The aluminum foil paper starting material is shown in FIG. 2a and was produced by Avery Dennison Corporation (Glendale, Calif., United States of America).
[0050] The metallic paper was introduced to a hydrothermal reactor (Parr, 4553M, 2 Gal) and exposed to the following conditions: 270.degree. C., 1488 psi, reaction time of 2.5 hours, with about 100 grams aluminum foil and 6 L water. The paper and glue portion of the sample was degraded during the reaction, the plastic portion of the sample melted and floated at the surface of the water, and the aluminum portion of the sample settled at the bottom. The recovered aluminum is shown in FIG. 2b. The results from the recovered aluminum quality test are shown in Table 1. Testing was performed in accordance with ASTM E1479, incorporated by reference herein. The aluminum content was calculated by difference. The results indicate that the sample comprises pure aluminum, and the recycled aluminum can be classified as the UNS grade designations A91050, A91110, and A91145.
TABLE-US-00001 TABLE 1 Recycled Aluminum Sample Chemical Testing Results Silicon .08% Iron .34 Copper .034 Manganese .003 Magnesium .003 Zinc .004 Nickel .007 Chromium .003 Lead <.003 Tin <.003 Titanium .015 Vanadium .010 Boron .003 Others Each <.03 Aluminum 99.50**
Example 2
Wastewater Results of Separation of Aluminum from Aluminum Paper Rolls
[0051] After the aluminum was removed, the remainder of the reaction products were analyzed to determine whether they could be discarded by flushing with water down a standard drain (i.e., a sink). Analytical results were provided by a 3.sup.rd party analytical service, and the results are given below in Table 2. Certification IDs: Florida/NELAP Certification No. E87648; Massachusetts Certification No. M-NC030; North Carolina Drinking Water Certification No. 37712; North Carolina Wastewater Certification No. 40; South Carolina Certification No. 99030001; Virginia/VELAP Certification No. 460222.
TABLE-US-00002 TABLE 2 Wastewater Testing Analytical Preparation Report Regulation Parameter Method Method Results Limit Limit.sup.1 6010 MET Arsenic EPA EPA 3010A ND.sup.2 10.0 .mu.g/L 100 .mu.g/L ICP 6010 Cadmium EPA EPA 3010A 3.9 .mu.g/L 1.0 .mu.g/L 50 .mu.g/L 6010 Lead EPA EPA 3010A 6.1 .mu.g/L 5.0 .mu.g/L 350 .mu.g/L 6010 7470 Mercury Mercury EPA EPA 7470 ND.sup.2 0.20 .mu.g/L 40 .mu.g/L 7470 351.2 Total Nitrogen, EPA -- 13.3 .mu.g/L 0.50 .mu.g/L -- Kjeldahl Kjeldahl, 351.2 Nitrogen total 365.1 Phosphorous EPA -- ND.sup.2 0.050 mg/L -- Phosphorous, total 365.1 total.sup.3 5220D COD Chemical SM -- 4200 mg/L 125 mg/L -- Oxygen 5220D Demand (COD) .sup.1Regulation Limit based on local regulation recommendation (Wastewater Division of Danville Utilities, City of Danville, Virginia, United States). .sup.2ND = Not detected at or above adjusted reporting limit. .sup.3Total phosphorous: No established limit, prefer <5 mg/L; COD: There is no limit for this parameter.
[0052] According to the wastewater chemical analysis results (Table 2), no harmful metal toxic materials were released in the post-treatment wastewater. The post-treatment wastewater was also within the local wastewater regulation limit. The above table indicate that the disclosed method is an environmentally-friendly process that could efficiently recover aluminum without producing harmful by-products.
Example 3
Separation of Aluminum from Tetra Pak package in Batch Reactor Using Co-Solvent
[0053] Samples of waste Tetra Pak (Pully, Switzerland) packages were obtained. The collected packages were introduced to a hydrothermal reactor (Parr, 4553M, 2 Gal) and a co-solvent (mixture of methanol and ddH.sub.2O; mixture of vegetable cooking oil and ddH.sub.2O) was added. The packages were exposed to the reaction conditions set forth in Table 3. Specifically, the operation temperature ranged from 170.degree. C. to 180.degree. C., pressure ranged from 115 psi to 1100 psi, residence time ranged from 30 mins to 50 mins, with about 30 grams Tetra Pak package and 6 L water.
TABLE-US-00003 TABLE 3 Co-solvent reaction conditions of Tetra Pak package in batch reactor Temperature (.degree. C.) Pressure (psi) MeOH:ddH.sub.2O Oil:ddH2O 170 1100 50:50 -- 175 220 50:50 -- 180 325 48:52 -- 180 320 .sup. 33:67 *.sup.1 -- 170 115 -- 90:10 *.sup.1 *.sup.1 Include 0.05% (w/v) sodium chloride (NaCl) in the reaction solvent
[0054] It was observed that when co-solvent conditions had been incorporated, the final recovered aluminum (as shown in FIG. 3a, 3b) was less contaminated with the other materials, such as polyester and paper.
* * * * *
D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.