Piperidine-2,6-dione Derivatives And Crohn's Disease Treating
ZHANG; Hesheng ; et al.
U.S. patent application number 16/464228 was filed with the patent office on 2019-11-21 for piperidine-2,6-dione derivatives and crohn's disease treating. The applicant listed for this patent is TIANJIN HEMAY BIO-TECH CO., LTD.. Invention is credited to Guanghuai ZENG, Hesheng ZHANG.
| Application Number | 20190352277 16/464228 |
| Document ID | / |
| Family ID | 62194806 |
| Filed Date | 2019-11-21 |












View All Diagrams
| United States Patent Application | 20190352277 |
| Kind Code | A1 |
| ZHANG; Hesheng ; et al. | November 21, 2019 |
PIPERIDINE-2,6-DIONE DERIVATIVES AND CROHN'S DISEASE TREATING
Abstract
Disclosed are piperidine-2,6-dione derivatives and treatment of Crohn's disease.
| Inventors: | ZHANG; Hesheng; (Tianjin, CN) ; ZENG; Guanghuai; (Tianjin, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 62194806 | ||||||||||
| Appl. No.: | 16/464228 | ||||||||||
| Filed: | November 23, 2017 | ||||||||||
| PCT Filed: | November 23, 2017 | ||||||||||
| PCT NO: | PCT/CN2017/112669 | ||||||||||
| 371 Date: | May 24, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 1/00 20180101; A61K 31/454 20130101; A61K 9/2018 20130101; A61P 1/04 20180101; A61K 9/08 20130101; A61K 9/4891 20130101; C07D 209/48 20130101; A61P 37/00 20180101; C07D 211/88 20130101; C07D 401/04 20130101 |
| International Class: | C07D 401/04 20060101 C07D401/04; A61P 1/00 20060101 A61P001/00 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 24, 2016 | CN | 201611041317.5 |
Claims
1. A piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof: ##STR00014## wherein, R.sub.1 represents one or more substituents selected from the group consisting of --H, halogen, --OH, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl; R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and R.sub.3 represents --H or --C.sub.1-4alkyl.
2. The piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof of claim 1: ##STR00015## wherein, R.sub.1 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --CH.sub.2CH.sub.3, --CH.sub.2CH.sub.2CH.sub.3, --NHCH.sub.3, --NH.sub.2, --NHCH.sub.2CH.sub.3, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3; R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and R.sub.3 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
3. The piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof of claim 1: ##STR00016## wherein, R.sub.1 represents one or more substituents selected from the group consisting of --H, --F, --OH, --CH.sub.3, --NHCH.sub.3, --N(CH.sub.3).sub.2, --NHCOCH.sub.3 and --NH.sub.2; R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and R.sub.3 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
4. The piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof of claim 1, which are selected from the group consisting of: ##STR00017## 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; 4-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof, 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-- 1,3-dione and pharmaceutically acceptable salts thereof, 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof, 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof, 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof, 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof, 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, and 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof.
5. A piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof: ##STR00018## wherein, R.sub.4 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl; R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-; and R.sub.7 represents --H or --C.sub.1-4alkyl.
6. The piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof of claim 5: ##STR00019## wherein, R.sub.4 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --NHCH.sub.3, --NHCH.sub.2CH.sub.3, --NH.sub.2, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3; R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--; and R.sub.7 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
7. The piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof of claim 5: ##STR00020## wherein, R.sub.4 represents --NH.sub.2 or --NHCOCH.sub.3; R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--; and R.sub.7 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
8. The piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof of claim 5, which are selected from the group consisting of: ##STR00021## 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, and 4-amino-2-(1-(2-methylsulfonylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof.
9. A piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof: ##STR00022## wherein, R.sub.8 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl; R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-; R.sub.11 represents --H or --C.sub.1-4alkyl; and R.sub.12 represents halogen or --C.sub.1-4alkyl.
10. The piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof of claim 9: ##STR00023## wherein, R.sub.8 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --NHCH.sub.3, --NHCH.sub.2CH.sub.3, --NH.sub.2, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3; R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.10 represents --O--, --S--, --SO--, --SH-- or --N(CH.sub.3)--; R.sub.11 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3; and R.sub.12 represents halogen or --C.sub.1-4alkyl.
11. The piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof of claim 9: ##STR00024## wherein, R.sub.8 represents --NH.sub.2 or --NHCOCH.sub.3; R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--; R.sub.11 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3; and R.sub.12 represents halogen or --C.sub.1-4alkyl.
12. The piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof of claim 9, which are selected from the group consisting of: 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; and 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof.
13. A piperidine-2,6-dione derivative and pharmaceutically acceptable salts thereof, which are selected from the group consisting of: 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; 4-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-- 1,3-dione and pharmaceutically acceptable salts thereof; 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof; 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof; 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methylsulfonylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-dimethylaminoethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof; 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof; 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof; 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof; 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione and pharmaceutically acceptable salts thereof, 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, and 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof.
14. A pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof of claim 1, and a pharmaceutically acceptable carrier, diluent or excipient.
15. A method for treating Crohn's disease, comprising administering a therapeutically effective amount of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof of claim 1.
16. The method of claim 15, wherein the Crohn's disease is mild Crohn's disease, moderate Crohn's disease, severe Crohn's disease or Crohn's disease in remission.
17. The method of claim 15, wherein the subject is human.
18. The method of claim 15, wherein the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof is administered orally.
19. The method of claim 18, wherein the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof is administered orally in a solid or liquid formulation.
20. The method of claim 19, wherein the solid formulation is a tablet, capsule or sugar-coated pill.
21. The method of claim 20, wherein the tablet is a plain tablet, sugar-coated tablet or film-coated tablet.
22. The method of claim 19, wherein the liquid formulation is a solution or suspension.
23-24. (canceled)
Description
RELATED APPLICATION
[0001] The present disclosure claims all the benefits of the patent application No. 201611041317.5 which was filed on Nov. 24, 2016 before the State Intellectual Property Office of the People's Republic of China and was entitled "Use of Piperidine-2,6-dione Derivatives for Treating Ulcerative Colitis", which is incorporated herein by reference in its entirety.
FIELD
[0002] The present disclosure relates to organic chemistry and medicinal chemistry fields.
BACKGROUND
[0003] Crohn's disease is a chronic inflammatory autoimmune disease of the gastrointestinal tract (GI). Crohn's disease can affect any part of the digestive tract from the mouth to the anus. Under the microscope, Crohn's disease affects the entire intestinal wall (transmural injury). Chronic inflammation can cause fibrosis in the subgroup of patients with Crohn's disease, who have the complications including stenosis and fistula, and possibly need repeated surgery. Crohn's disease is associated with increased risk of gastrointestinal malignant tumour. It is found by the study on Crohn's disease that Crohn's disease is associated with TH1 (type 1 T helper cell) and TH17 (type 17 T helper cell) mediated cellular immunity.
[0004] The clinical manifestations of Crohn's disease are various and include gastrointestinal manifestations, systemic manifestations, parenteral manifestations, and complications. Gastrointestinal manifestations mainly include diarrhea, abdominal pain, and bloody stool. Systemic manifestations mainly include weight loss, fever, loss of appetite, fatigue, anemia, and the like. Growth retardation is commonly found in adolescent patients. Common complications include fistula, abdominal abscess, intestinal stenosis, obstruction and perianal lesions. Gastrointestinal bleeding and acute perforation are rare. The long course of disease can lead to cancer. Colonoscopy is generally manifested as segmental and asymmetric various mucosal inflammation, which has the characterized manifestation of non-continuous lesions, longitudinal ulcers and pebble-like appearance. The histopathological changes of Crohn's disease mucosal biopsy samples include: (1) focal discontinuous infiltration of intrinsic membrane inflammatory cells; (2) fissuring ulcers; (3) aphthous ulcers; (4) abnormal crypt structure, glandular hyperplasia, individual crypt abscess, non-obvious reduction of mucus secretion, visible pyloric metaplasia or Paneth cell metaplasia; (5) non-cheese-like necrotic granuloma; (6) chronic inflammatory cell infiltration of lymphocytes and plasma cells, in which the inflammation at the bottom of lamina propria and submucosal layer is severe, and formation of lymph follicle is common; and (7) submucosal lymphatic dilatation; and (8) ganglion cell proliferation and/or ganglion inflammation.
[0005] Mildly active Crohn's disease is treated mainly with 5-aminosalicylic acids and budesonide. If the above treatment is ineffective, Crohn's disease is considered as moderately active. Moderately active Crohn's disease is preferably treated with hormone (such as dexamethasone). If the above treatment is ineffective or becomes dependent, combination with thioglycine or methotrexate will be considered. TNF-.alpha. monoclonal antibody can be used if the above hormone therapy and immunotherapy are ineffective or intolerable. Severely active Crohn's disease is treated mainly with systemic hormones, TNF-.alpha. monoclonal antibodies and surgery. Drugs for maintenance treatment for Crohn's disease in remission include 5-aminosalicylic acids, thioglycines and TNF-.alpha. monoclonal antibodies.
[0006] It is found during the use of 5-aminosalicylic acid drugs (such as sulfasalazine and mesalazine) that about 50% of patients suffer vomiting, anepithymia, liver dysfunction and other digestive disorders, hemolytic anemia and folic acid deficiency anemia and other blood disorders. In addition, since there is a salicylic acid skeleton, there is a possibility that side effects such as diarrhea, abdominal pain, amylase rise, and renal dysfunction occur in cases where the patients have allergic symptoms on salicylic acid agents. Furthermore, because it is discovered that sulfasalazine has side effects of male infertility and colored urine, the patients suffer great mental stress. Due to immunosuppression and short-term efficacy characteristics, budesonide or azathiopurine can also be used, but only in a short term. When the patients are not administered with the biological agents, it will lead to the recurrence of the disease and increase difficulty to treat the disease. Moreover, the biological agents will also give patients a greater financial burden. Monoclonal antibody drugs (such as infliximab and adalimumab) may cause high blood pressure, chills, rash, fever, headache, eczema and so on. As infliximab is a chimeric antibody, it is possible to show antigenicity and sometimes cause acute hypersensitivity.
[0007] Small molecule TNF-.alpha. inhibitors such as lenalidomide and thalidomide have been shown to be not effective in the treatment of Crohn's disease (C. Yang et, Aliment Pharmacol Ther 2015; 41:1079-7093.).
[0008] Moreover, Crohn's disease is a chronic disease that requires a longer duration of drugs. The above data show that there is no drug for long-term effective treatment of Crohn's disease, especially drug by oral administration. Therefore, there is a need for developing better target therapy with optimized chronic use for Crohn's disease, or drugs with better effectiveness or better safety.
SUMMARY
[0009] Dinitrobenzene sulfonic acid (DNBS) or trinitrobenzene sulfonic acid (TNBS)-induced inflammatory models are widely used to study the mechanism of production of Crohn's disease and the development and assessment of drugs for treating Crohn's disease. See the following reference: Establishment of Inflammatory Bowel Disease Models Induced by 2,4,6-Trinitrobenzenesulfonic, J. Med. Res., July 2008, Vol. 37, No. 7, which are incorporated herein by reference in its entirety.
[0010] Small molecular TNF-.alpha. inhibitors such as lenalidomide and thalidomide have been shown to be not effective in the treatment of Crohn's disease (C. Yang et, Aliment Pharmacol Ther 2015; 41:1079-7093.).
[0011] In one aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof:
##STR00001##
[0012] wherein,
[0013] R.sub.1 represents one or more substituents selected from the group consisting of H, halogen, --OH, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0014] R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and
[0015] R.sub.3 represents --H or --C.sub.1-4alkyl.
[0016] In another aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof:
##STR00002##
[0017] wherein,
[0018] R.sub.4 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0019] R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0020] R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-; and
[0021] R.sub.7 represents --H or --C.sub.1-4alkyl.
[0022] In yet another aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof:
##STR00003##
[0023] wherein,
[0024] R.sub.8 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0025] R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0026] R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-;
[0027] R.sub.11 represents --H or --C.sub.1-4alkyl; and
[0028] R.sub.12 represents halogen or --C.sub.1-4alkyl.
[0029] All the compounds of formula (I), formula (II) and formula (III) as mentioned above belong to piperidine-2,6-dione derivatives as mentioned herein.
[0030] In still another aspect, some embodiments disclose a piperidine-2,6-dione derivative and pharmaceutically acceptable salts thereof, which are selected from the group consisting of: [0031] 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; [0032] 4-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; [0033] 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof; [0034] 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof; [0035] 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione and pharmaceutically acceptable salts thereof; [0036] 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; [0037] 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; [0038] 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof; [0039] 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof; [0040] 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof; [0041] 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione and pharmaceutically acceptable salts thereof; [0042] 4-amino-2-(1-(2-methyl sulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof, [0043] 4-amino-2-(1-(2-methylsulfonylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0044] 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof, [0045] 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof, [0046] 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0047] 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0048] 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, [0049] 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof, [0050] 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0051] 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0052] 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0053] 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0054] 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0055] 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione and pharmaceutically acceptable salts thereof, [0056] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, and [0057] 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof.
[0058] In another aspect, some embodiments disclose a pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, and a pharmaceutically acceptable carrier, diluent or excipient.
[0059] In another aspect, some embodiments disclose a method for treating Crohn's disease, comprising administering a therapeutically effective amount of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, or a therapeutically effective amount of the pharmaceutical composition as disclosed herein to a subject in need thereof.
[0060] In another aspect, some embodiments disclose the piperidine-2,6-dione derivative and pharmaceutically acceptable salts thereof of as disclosed herein for treating Crohn's disease.
[0061] In another aspect, some embodiments disclose a pharmaceutical composition for treating Crohn's disease, comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, and a pharmaceutically acceptable carrier, diluent or excipient.
DETAILED DESCRIPTION
[0062] In the following description, certain specific details are included to provide a thorough understanding for various disclosed embodiments. One skilled in the relevant art, however, will recognize that the embodiments may be practiced without one or more these specific details, or with other methods, components, materials, etc.
[0063] Unless the context required otherwise, throughout the specification and claims which follows, the term "comprise" and variations thereof, such as "comprises" and "comprising" are to be construed in an open, inclusive sense, which is as "include, but not limited to".
[0064] Reference throughout this specification to "one embodiment", or "an embodiment", or "in another embodiment", or "in some embodiments" means that a particular referent feature, structure or characteristic described in connection with the embodiments is included in at least one embodiment. Therefore, the appearance of the phrases "in one embodiment", or "in the embodiment", or "in another embodiment", or "in some embodiments" in various places throughout this specification are not necessarily all referring to the same embodiment. Moreover, the particular features, structures or characteristics may be combined in any suitable manner in one or more embodiments.
Definition
[0065] Accordingly, as used in the specification and appended claims, unless specified to the contrary, the following terms have the meanings indicated:
[0066] The term "Crohn's disease" is a nonspecific inflammatory disease, of which the etiology is not yet clear. The more specific definition can be found in the following reference: Consensus on diagnosis and treatment of inflammatory bowel disease (2012, Guangzhou), Chinese Journal of Gastroenterology, 2012, 51 (12):763-781, which is incorporated herein by reference in its entirety.
[0067] According to the severity of disease, Crohn's disease can be mild Crohn's disease, moderate Crohn's disease, or severe Crohn's disease. According to the activity of disease, Crohn's disease can be divided into active Crohn's disease or Crohn's disease in remission.
[0068] The goal of treatment for Crohn's disease is to induce active Crohn's disease to enter the remission (i.e, induction of remission) and/or to maintain the disease in remission (i.e., maintenance of remission).
[0069] In clinical studies, the Montreal CD phenotype is a classification method for Crohn's disease. The details are as follows:
TABLE-US-00001 TABLE 1 Montreal Classification for Crohn's disease Age at diagnosis (A) A1 16 years or younger A2 between 17 to 40 years A3 over 40 years Location (L) L1 Terminal ileum L1 + L4.sup.b L2 Colon L2 + L4.sup.b L3 Ileocolon L2 + L4.sup.b L4 Upper gastrointestinal Behaviour (B) B1.sup.a non-stricturing, non-penetrating B1P.sup.c B2 stricturing B2p.sup.c B3 penetrating B3p.sup.c Note: .sup.aB1 may develop into B2 or B3 when time passes; .sup.bL4 may coexist with L1, L2 or L3; .sup.cp is perianal disease, which may coexist with B1, B2 or B3
[0070] Clinically, the Crohn's disease activity index (CDAI) can also be used to assess the severity of disease activity and evaluate efficacy. Best CDAI calculation can be referred to. The details are as follows:
TABLE-US-00002 TABLE 2 Best CDAI Calculation Variable Multiplier Number of liquid stools (in one week) 2 Abdominal pain ratings (in one week, 0-3 points) 5 General well-being (in one week, 0-4 points) 7 Extraintestinal findings and complications 20 (1 point for each item) Opiods antidiarrhoeal drugs (no = 0; yes = 1) 30 Abdominal mass (questionalbel = 2; definite = 5) 10 Haematocrit reduction (Norm.sup.a: Men: 0.40, Women: 0.37) 6 100 .times. (1-observed body weight/ideal body weight) 1 Note .sup.ahaematocrit norm is based on Chinese standard; total point = the sum of the points in each item, where CDAI is <150, the disease is in remission; where CDAI is .gtoreq.150 the disease is active, where CDAI is 150-220, disease is mild; where CDAI is 221-450, disease is moderate; where CDAI is >450, disease is severe.
[0071] The patient's disease was scored according to the Best CDAI calculation as described above. After scoring, the clinical remission corresponds to Crohn's disease in remission of the present disclosure, mild activity corresponds to mild Crohn's disease of the present disclosure, moderate activity corresponds to moderate Crohn's disease of the present disclosure, and severe activity corresponds to severe Crohn's disease of the present disclosure.
[0072] Certain chemical groups named herein are preceded by a shorthand notation indicating the total number of carbon atoms that are to be found in the indicated chemical group. For example, C.sub.1-C.sub.4alkyl describes an alkyl group, as defined below, having a total of 1 to 4 carbon atoms, and C.sub.3-C.sub.10cycloalkyl describes a cycloaklyl group, as defined below, having a total of 3 to 10 carbon atoms. The total number of carbon atoms in the shorthand notation does not include the carbons that may exist in the substituents of the groups described.
[0073] The term "mammal" means animals including, for example, dogs, cats, cows, sheep, horses, and humans. In some embodiments, mammals include humans.
[0074] The term "patient" means an animal, such as a human, a companion animal, such as a dog, cat and horse, and livestock, such as cattle, swine and sheep. In some embodiments, patients are mammals, including both males and females. In some embodiments, patients are humans.
[0075] The term "pharmaceutically acceptable" as used herein means the carrier, vehicle, diluent, excipient and/or salt must be compatible with the other ingredients of the formulation, and not deleterious to the recipient thereof.
[0076] "Optional" or "optionally" means that the subsequently described event of circumstances may or may not occur, and that the description includes instances where said event or circumstance occurs and instances in which it does not.
[0077] "Pharmaceutically acceptable carrier, diluent or excipient" includes without limitation any adjuvant, carrier, excipient, glidant, sweetening agent, diluent, preservative, dye/colorant, flavor enhancer, surfactant, wetting agent, dispersing agent, suspending agent, stabilizer, isosmotic agent, solvent, or emulsifier, etc, which has been approved by the United States Food and Drug Administration as being acceptable for use in humans or animals and have no side effects on preparing a pharmaceutical composition.
[0078] "Pharmaceutically acceptable salts" include both "pharmaceutically acceptable acid addition salts" and "pharmaceutically acceptable base addition salts".
[0079] "Pharmaceutically acceptable acid addition salt" refers to those salts which retain the biological effectiveness and properties of the free bases, which are not biologically or otherwise undesirable, and which are formed with inorganic acids such as, but not limited to hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid and the like, and organic acids such as, but not limited to, acetic acid, 2,2-dichloroacetic acid, adipic acid, alginic acid, ascorbic acid, aspartic acid, benzenesulfonic acid, benzoic acid, 4-acetamidobenzoic acid, camphanic acid, camphor-10-sulfonic acid, capric acid, caproic acid, caprylic acid, carbonic acid, cinnamic acid, citric acid, cyclamic acid, dodecylsulfuric acid, ethane-1,2-disulfonic acid, ethanesulfonic acid, 2-hydroxyethanesulfonic acid, formic acid, fumaric acid, galactaric acid, gentisic acid, glucoheptonic acid, gluconic acid, glucuronic acid, glutamic acid, glutaric acid, 2-oxo-glutaric acid, glycerophosphoric acid, glycolic acid, hippuric acid, isobutyric acid, lactic acid, lactobionic acid, lauric acid, maleic acid, malic acid, malonic acid, mandelic acid, methanesulfonic acid, mucic acid, naphthalene-1,5-disulfonic acid, naphthalene-2-sulfonic acid, 1-hydroxy-2-naphthoic acid, nicotinic acid, oleinic acid, orotic acid, oxalic acid, palmitic acid, pamoic acid, propionic acid, pyroglutamic acid, pyruvic acid, salicylic acid, 4-aminosalicylic acid, sebacic acid, stearic acid, succinic acid, tartaric acid, thiocyanic acid, p-toluenesulfonic acid, trifluoroacetic acid, undecylenic acid and the like.
[0080] "Pharmaceutically acceptable base addition salt" refers to those salts which retain the biological effectiveness and properties of the free acids, which are not biologically or otherwise undesirable. These salts are prepared from addition of an inorganic or an organic base to the free acid. Salts derived from inorganic bases include, but are not limited, to sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminium salts and the like. In some embodiments, inorganic salts are the ammonium, sodium, potassium, calcium, and magnesium salts. Salts derived from organic bases include, but are not limited to, salts of primary, secondary and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines and s basic ion exchange resins, such as ammonia, isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, diethanolamine, ethanolamine, 2-dimethylaminoethanol, 2-diethylaminoethanol, dicyclohexylamine, lysine, arginine, histidine, caffeine, procaine, hydrabamine, choline, betaine, benethamine, benzathine, ethylenediamine, glucosamine, methylglucosamine, theobromine, triethanolamine, trometamol, purine, piperazine, piperidine, N-ethyl piperidine, polyamine resins and the like. In some embodiments, organic bases are isopropylamine, diethylamine, ethanolamine, trimethylamine, dicyclohexylamine, choline and caffeine.
[0081] The term "a solvent or a solvent mixture" refers to any and all solvents, In some embodiments, a solvent or a solvent mixture is organic solvents and water, which include, but are not limited to, water, methanol, ethanol, 2-propanol, n-butanol, iso-butanol, acetone, methylethylketone, ethylacetate, 1,4-dioxane, diethylether, MTBE, THF, acetonitrile, dichloromethane, chloroform, DMF, cyclohexane, cyclopentane, n-hexane, n-heptane, n-pentane, toluene, o-xylene, p-xylene, DMSO, pyridine, acetic acid, anisole, butylacetate, cumene, ethylformate, formic acid, iso-butylacetate, iso-propylacetate, methylacetate, 3-methyl-1-butanol, methylisobutylketone, 2-methyl-1-propanol, 1-pentanol, propylacetate, ethylenglycole, and 1-methyl-2-pyrrolidone, as well as any and all mixtures of two or more such solvents. In some embodiments, a solvent or a solvent mixture is single solvent and binary mixtures. In some embodiments, a solvent or a solvent mixture is water and single solvent of organic solvent and binary mixtures of water and organic solvent.
[0082] A "pharmaceutical composition" refers to a formulation of a compound of the present disclosure and a medium generally acceptable in the art for the delivery of the biologically active compound to mammals, e.g., humans. Such a medium includes all pharmaceutically acceptable carriers, diluents or excipients therefor.
[0083] "Therapeutically effective amount" refers to an amount of a compound or combination of compounds that ameliorates, attenuates or eliminates a particular disease or condition or a symptom of a particular disease or condition, or prevents or delays the onset of a particular disease or condition or a symptom of a particular disease or condition. The amount of a compound of the present disclosure which constitutes a "therapeutically effective amount" will vary depending on the compound, the condition and its severity, and the age of the mammal to be treated, but can be determined routinely by one of ordinary skill in the art having regard to his own knowledge and to this disclosure.
[0084] "Treating" or "treatment" as used herein covers the treatment of the disease or condition of interest in a mammal, such as a human, having the disease or disorder of interest, and includes:
[0085] (i) preventing the disease or condition from occurring in a mammal, in particular, when such mammal is predisposed to the condition but has not yet been diagnosed as having it;
[0086] (ii) inhibiting the disease or condition, i.e., arresting its development; or
[0087] (iii) relieving the disease or condition, i.e., causing regression of the disease or condition.
[0088] As used herein, the terms "disease" and "condition" may be used interchangeably or may be different in that the particular malady or condition may not have a known causative agent (so that etiology has not yet been worked out) and it is therefore not yet recognized as a disease but only as an undesirable condition or syndrome, wherein a more or less specific set of symptoms have been identified by clinicians.
[0089] The compounds of the present disclosure or their pharmaceutically acceptable salt may contain one or more asymmetric centers and may thus give rise to enantiomers, diastereoisomers, and other stereoismeric forms that may be defined, in terms of absolute stereochemistry, as (R)- or (S)-, or (D)- or (L)- for amino acids. The present disclosure is meant to include all such possible isomers, as well as their racemic and optically pure forms. Optically active (+) and (-), (R)- and (S)-, or (D)- and (L)-isomers may be prepared using chiral synthons or chiral reagents, or resolved using conventional techniques, such as HPLC using a chiral column. When the compounds described herein contain olefinic double bonds or other centers of geometric asymmetry, and unless specified otherwise, it is intended that the compounds include both E and Z geometric isomers. Likewise, all tautomeric forms are also intended to be included.
[0090] A "stereoisomer" refers to a compound made up of the same atoms bonded by the same bonds but having different three-dimensional structures, which are not interchangeable. The present disclosure contemplates various stereoisomers and mixtures thereof.
[0091] The substituent position of the compounds disclosed in the present disclosure is numbered as follows:
##STR00004##
[0092] The substitution site of R.sub.1 in formula (I) can be one of the sites numbered as 4, 5, 6 and 7 or a combination of several sites numbered as 4, 5, 6 and 7. The substitution site of R.sub.4 in formula (II) can be one of the sites numbered as 4, 5, 6 and 7 or a combination of several sites numbered as 4, 5, 6 and 7. The substitution site of R.sub.8 in formula (III) can be one of the sites numbered as 4, 5, 6 and 7 or a combination of several sites numbered as 4, 5, 6 and 7.
[0093] These and other features, aspects and advantages of the present disclosure will become better understood with reference to the following drawings, description and claims.
Specific Embodiments
[0094] In one aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof:
##STR00005##
[0095] wherein,
[0096] R.sub.1 represents one or more substituents selected from the group consisting of H, halogen, --OH, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0097] R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and
[0098] R.sub.3 represents --H or --C.sub.1-4alkyl.
[0099] Some embodiments disclose a piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof:
##STR00006##
[0100] wherein,
[0101] R.sub.1 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --CH.sub.2CH.sub.3, --CH.sub.2CH.sub.2CH.sub.3, --NHCH.sub.3, --NH.sub.2, --NHCH.sub.2CH.sub.3, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3;
[0102] R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and
[0103] R.sub.3 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
[0104] Some embodiments disclose a piperidine-2,6-dione derivative of formula (I) and pharmaceutically acceptable salts thereof:
##STR00007## [0105] wherein,
[0106] R.sub.1 represents one or more substituents selected from the group consisting of --H, --F, --OH, --CH.sub.3, --NHCH.sub.3, --N(CH.sub.3).sub.2, --NHCOCH.sub.3 and --NH.sub.2;
[0107] R.sub.2 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--; and
[0108] R.sub.3 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
[0109] Some embodiments disclose a piperidine-2,6-dione derivatives of formula (I) and pharmaceutically acceptable salts thereof, which are selected from the group consisting of: [0110] 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; [0111] 4-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof; [0112] 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof; [0113] 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof; [0114] 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione and pharmaceutically acceptable salts thereof, [0115] 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; [0116] 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof; [0117] 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof; [0118] 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof, [0119] 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof, [0120] 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0121] 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0122] 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, [0123] 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof, [0124] 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0125] 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0126] 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0127] 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0128] 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0129] 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione and pharmaceutically acceptable salts thereof, [0130] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, and [0131] 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof.
[0132] In one aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof:
##STR00008##
[0133] wherein,
[0134] R.sub.4 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0135] R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0136] R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-; and
[0137] R.sub.7 represents --H or --C.sub.1-4alkyl.
[0138] Some embodiments disclose a piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof:
##STR00009##
[0139] wherein,
[0140] R.sup.4 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --NHCH.sub.3, --NHCH.sub.2CH.sub.3, --NH.sub.2, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3;
[0141] R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0142] R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--; and
[0143] R.sub.7 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
[0144] Some embodiments disclose a piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof:
##STR00010##
[0145] wherein,
[0146] R.sub.4 represents --NH.sub.2 or --NHCOCH.sub.3;
[0147] R.sub.5 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0148] R.sub.6 represents --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--; and
[0149] R.sub.7 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3.
[0150] Some embodiments disclose a piperidine-2,6-dione derivative of formula (II) and pharmaceutically acceptable salts thereof: [0151] 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione and pharmaceutically acceptable salts thereof; [0152] 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof; and [0153] 4-amino-2-(1-(2-methylsulfonylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof.
[0154] In yet another aspect, some embodiments disclose a piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof:
##STR00011##
[0155] wherein,
[0156] R.sub.8 represents one or more substituents selected from the group consisting of H, halogen, --C.sub.1-4alkyl, --NH.sub.2, --NHC.sub.1-4alkyl, --N(C.sub.1-4alkyl).sub.2 and --NHCOC.sub.1-4alkyl;
[0157] R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0158] R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(C.sub.1-4alkyl)-;
[0159] R.sub.11 represents --H or --C.sub.1-4alkyl; and
[0160] R.sub.12 represents halogen or --C.sub.1-4alkyl.
[0161] Some embodiments disclose a piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof:
##STR00012##
[0162] wherein,
[0163] R.sub.8 represents one or more substituents selected from the group consisting of --H, --F, --Cl, --Br, --OH, --CH.sub.3, --NHCH.sub.3, --NHCH.sub.2CH.sub.3, --NH.sub.2, --N(CH.sub.3).sub.2, --N(CH.sub.2CH.sub.3).sub.2, --NHCOCH.sub.3 and --NHCOCH.sub.2CH.sub.3;
[0164] R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0165] R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--;
[0166] R.sub.11 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3; and
[0167] R.sub.12 represents halogen or --C.sub.1-4alkyl.
[0168] Some embodiments disclose a piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof:
##STR00013##
[0169] wherein,
[0170] R.sub.8 represents --NH.sub.2 or --NHCOCH.sub.3;
[0171] R.sub.9 represents --CH.sub.2CH.sub.2--, --CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2CH.sub.2CH.sub.2CH.sub.2--;
[0172] R.sub.10 represents --O--, --S--, --SO--, --SO.sub.2--, --NH-- or --N(CH.sub.3)--;
[0173] R.sup.11 represents --H, --CH.sub.3 or --CH.sub.2CH.sub.3; and
[0174] R.sub.12 represents halogen or --C.sub.1-4alkyl.
[0175] Some embodiments disclose a piperidine-2,6-dione derivative of formula (III) and pharmaceutically acceptable salts thereof, which are selected from the group consisting of: [0176] 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof; and [0177] 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof.
[0178] All the compounds of formula (I), formula (II) and formula (III) as mentioned above belong to piperidine-2,6-dione derivatives as mentioned herein.
[0179] Some embodiments disclose a piperidine-2,6-dione derivatives and pharmaceutically acceptable salts thereof, which are selected from the group consisting of: [0180] 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof, [0181] 4-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione and pharmaceutically acceptable salts thereof, [0182] 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof, [0183] 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, [0184] 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione and pharmaceutically acceptable salts thereof, [0185] 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0186] 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0187] 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof, [0188] 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof, [0189] 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione and pharmaceutically acceptable salts thereof, [0190] 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione and pharmaceutically acceptable salts thereof, [0191] 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0192] 4-amino-2-(1-(2-methylsulfonylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0193] 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindol in-1,3-dione and pharmaceutically acceptable salts thereof, [0194] 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione and pharmaceutically acceptable salts thereof, [0195] 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione and pharmaceutically acceptable salts thereof, [0196] 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0197] 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof, [0198] 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione and pharmaceutically acceptable salts thereof, [0199] 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0200] 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0201] 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, [0202] 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0203] 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one and pharmaceutically acceptable salts thereof, [0204] 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione and pharmaceutically acceptable salts thereof, [0205] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e and pharmaceutically acceptable salts thereof, and [0206] 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne and pharmaceutically acceptable salts thereof.
[0207] In still another aspect, some embodiments disclose a pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, and a pharmaceutically acceptable carrier, diluent or excipient.
[0208] In yet another aspect, some embodiments disclose a method for treating Crohn's disease, comprising administering a therapeutically effective amount of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, or a therapeutically effective amount of the pharmaceutical composition as disclosed herein to a subject in need thereof.
[0209] Some embodiments disclose a method for treating Crohn's disease, comprising administering 1 mg-10 g of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein to a subject in need thereof.
[0210] Some embodiments disclose a method for treating Crohn's disease, comprising administering 10 mg-3000 mg of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein to a subject in need thereof.
[0211] Some embodiments disclose a method for treating Crohn's disease, comprising administering 100 mg-1000 mg of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein to a subject in need thereof.
[0212] Some embodiments disclose a method for treating Crohn's disease, comprising administering 100 mg, 150 mg, 200 mg, 250 mg, 300 mg, 350 mg, 400 mg, 450 mg, 500 mg, 550 mg, 600 mg, 650 mg, 700 mg, 750 mg, 800 mg, 850 mg, 900 mg or 1000 mg of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein to a subject in need thereof.
[0213] Some embodiments disclose a method for treating Crohn's disease, wherein the Crohn's disease is mild Crohn's disease, moderate Crohn's disease, severe Crohn's disease or Crohn's disease in remission.
[0214] Some embodiments disclose a method for treating Crohn's disease, wherein the subject in need thereof is a mammal.
[0215] Some embodiments disclose a method for treating Crohn's disease, wherein the subject in need thereof is human.
[0216] Some embodiments disclose of a method of maintenance treatment of Crohn's disease, comprising administering a therapeutically effective amount of the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, or a therapeutically effective amount of the pharmaceutical composition as disclosed herein to a subject in need thereof.
[0217] Some embodiments disclose a method for treating Crohn's disease, wherein the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, or the pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein is administered orally.
[0218] Some embodiments disclose a method for treating Crohn's disease, wherein the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, or the pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein is administered orally in a solid or liquid formulation.
[0219] Exemplary examples of the solid formulation that can be used in the method for treating Crohn's disease as disclosed herein comprise, but are not limited to, a tablet, capsule and sugar-coated pill.
[0220] Exemplary examples of the tablet that can be used in the method for treating Crohn's disease as disclosed herein comprise, but are not limited to, a plain tablet, sugar-coated tablet and film-coated tablet.
[0221] Exemplary examples of the liquid formulation that can be used in the method for treating Crohn's disease as disclosed herein comprise, but are not limited to, a solution and suspension.
[0222] In yet another aspect, some embodiments disclose a piperidine-2,6-dione derivative and pharmaceutically acceptable salts thereof for treating Crohn's disease.
[0223] In still another aspect, some embodiments disclose a pharmaceutical composition for treating Crohn's disease, comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, and a pharmaceutically acceptable carrier, diluent or excipient.
[0224] The piperidine-2,6-dione derivatives as disclosed herein have good therapeutical effects.
[0225] The piperidine-2,6-dione derivatives as disclosed herein have better safety.
[0226] The piperidine-2,6-dione derivatives as disclosed herein have better tolerance.
Pharmaceutical Compositions
[0227] Some embodiments disclose a pharmaceutical composition comprising the piperidine-2,6-dione derivative or pharmaceutically acceptable salts thereof as disclosed herein, and a pharmaceutically acceptable carrier, diluent or excipient.
[0228] In some embodiments, the route of administration of the piperidine-2,6-dione derivatives as disclosed herein to the mammals can be non-parenteral route.
[0229] In some embodiments, the route of administration of the piperidine-2,6-dione derivatives as disclosed herein to the mammals can be oral route.
[0230] In some embodiments, the route of administration of the piperidine-2,6-dione derivatives as disclosed herein to the mammals can be intrarectal route.
[0231] The piperidine-2,6-dione derivatives as described herein may be obtained in any suitable form such as tablet, capsule, powder, oral solution, suspension, rectal gel, rectal foam, rectal enema or suppository and the like. Exemplary examples of tablets comprise, but are not limited to, plain tablets, sugar-coated tablets and film-coated tablets.
[0232] Examples of a pharmaceutically acceptable carrier that can be used in the pharmaceutical composition of the present disclosure include, but are not limited to, any adjuvant, carrier, excipient, glidant, sweetening agent, diluent, preservative, dye/colorant, flavor enchancer, surfactant, wetting agent, dispersing agent, suspending agent, stabilizer, isosmotic agent, solvent or emulsifier, which has been approved by the United States Food and Drug Administration as being acceptable for use in humans or animals. Acceptable carriers or diluents for therapeutic use are well-known in the art, and are described, for example, in Remington's Pharmaceutical Sciences, 18th Ed., Mack Publishing Co., Easton, Pa. (1990), which is incorporated herein by reference in its entirety.
[0233] The pharmaceutical compositions of the present disclosure may be administered by any means that achieve their intended purpose. For example, administration may be by oral, parenteral, topical, enteral, intravenous, intramuscular, inhalant, nasal, intraarticular, intraspinal, transtracheal, transocular, subcutaneous, intraperitoneal, transdermal, or buccal routes. The route of administration can be non-parenteral route, oral route and intrarectal route. The dosage administered will be dependent upon the age, health, and weight of the recipient, kind of concurrent treatment, if any, frequency of treatment, and the nature of the effect desired.
[0234] Suitable dosage forms include, but are not limited to capsules, tablets, pellets, dragees, semi-solids, powders, granules, suppositories, ointments, creams, lotions, inhalants, injections, cataplasms, gels, tapes, eye drops, solution, syrups, aerosols, suspension, emulsion, which can be produced according to methods known in the art.
[0235] Particularly suitable for oral use are ordinary tablets (plain tablets), sugar-coated tablet, film-coated tablets, pills, coated tablets, capsules, powders, granules, syrups, juices or drops, suitable for rectal use are suppositories, suitable for parenteral use are solutions, or oil-based or aqueous solutions, furthermore suspensions, emulsions or implants, and suitable for topical use are ointments, creams or powders. The compounds of the present disclosure may also be lyophilised and the resultant lyophilisates used, for example, for the preparation of injection preparations. The preparations indicated may be sterilised and/or comprise assistants, such as lubricants, preservatives, stabilisers and/or wetting agents, emulsifiers, salts for modifying the osmotic pressure, buffer substances, dyes, flavours and/or a plurality of further active ingredients, for example one or more vitamins.
[0236] In some embodiments, a pharmaceutical composition of the present disclosure is formulated as tablet, solution, granule, patch, ointment, capsule, aerosol or suppository administered via parenteral, transdermal, mucosa, nasal, buccal, sublingual or oral route.
[0237] Preservatives, stabilizers, dyes, sweeteners, flavoring agents, fragrances, and the like, may be provided in the pharmaceutical composition. For example, sodium benzoate, ascorbic acid and esters of p-hydroxybenzoic acid may be added as preservatives. Furthermore, antioxidants and suspending agents may be used.
[0238] In various embodiments, alcohols, esters, sulfating aliphatic alcohols, and the like may be used as surfactants; sucrose, glucose, lactose, starch, crystalline cellulose, mannitol, light anhydrous silicate, magnesium aluminate, methyl magnesium silicate aluminate, synthetic aluminum silicate, calcium carbonate, calcium bicarbonate, calcium hydrogenphosphate, calcium hydroxymethyl cellulose and the like may be used as excipients; magnesium stearate, talc, hardened oil may be used as smoothing agents; coconut oil, olive oil, sesame oil, peanut oil, soybean may be used as suspending agents or lubricants; cellulose acetate phthalate as a derivative of a carbohydrate such as cellulose or sugar, or methylacetate-metharylate copolymer as a derivative of polyethylene may be used as suspending agents; and plasticizers such as ester phthalates and the like may be used as suspending agents.
[0239] Suitable routes of administration may, for example, include oral, rectal, transmucosal, topical, or intestinal administration; parenteral delivery, including intramuscular, subcutaneous, intravenous, intramedullary injections, as well as intrathecal, direct intraventricular, intraperitoneal, intranasal or intraocular injections. The compound can be administered in sustained or controlled release dosage forms, including depot injections, osmotic pumps, pills, transdermal (including electromigrating) patches, and the like for prolonged and/or timed, pulsed administration at a predetermined rate.
[0240] Pharmaceutical compositions of the present disclosure may be manufacture in manner that is itself known, for example, by means of conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping, or tabletting processes.
[0241] Pharmaceutical compositions for use in accordance with the present disclosure thus may be formulated by a conventional manner using one or more physiologically acceptable carriers comprising excipienst and auxiliaries which facilitate processing the active compounds into preparation which can be used pharmaceutically. Proper formulation is dependent on the route of administration chosen. Any of the well-known techniques, carriers and excipients may be used as suitable and as understood in the art.
[0242] Injectables can be prepared in conventional forms, either as liquid solutions or suspensions, solid forms suitable for solution or suspension in liquid prior to injection, or as emulsions. Suitable excipients are, for example, water, saline, glucose, mannitol, lactose, lecithin, albumin, sodium glutamate, cysteine hydrochloride, and the like. Furthermore, if desired, the injectable pharmaceutical compositions may contain minor amounts of nontoxic auxiliary substances, such as wetting agents, pH buffering agents, and the like. Physiologically compatible buffers include, but are not limited to, Hank's solution, Ringer's solution or physiological saline buffer. If desired, absorption enhancing preparations (such as liposomes) may be used.
[0243] For oral administration, the compound can be formulated readily by combining the active compound with pharmaceutically acceptable carriers well known in the art. Such carriers enable the compound of the disclosure to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, ointments, suspensions, and the like, for oral ingestion by a patient to be treated. Pharmaceutical preparation for oral use can be obtained by combining the active compound with solid excipient, optionally grinding a resultant mixture, and processing the mixture of granules, after adding suitable auxiliaries, if desired, to obtain tablets or dragee cores. Suitable excipients are, in particular, fillers such as sugars, including lactose, saccharose, mannitol or sorbitol; cellulose preparations such as, for example, maize starch, wheat starch, rice starch, potato starch, gelatin, gum tragacanth, methylcellulose, hydroxypropyl methylcellulose, sodium carboxymethylcellulose, and/or polyvinylpyrrolidone (PVP). If desired, disintegrating agents may be added, such as the crosslinked polyvinylpyrrolidone, agar, or alginic acid or a salt thereof such as sodium alginate. Dragee cores are provided with suitable coatings. For this purpose, concentrated sugar solutions may be used, which may optionally contain gum arabic, talc, polyvinyl pyrrolidone, carbopol gel, polyethylene glycol, and/or titanium dioxide, lacquer solution, and suitable organic solvents or solvent mixtures. Dyestuffs or pigments may be added into the tablets or dagree coatings for identification or to characterizing different combinations of active compound doses. For this purpose, concentrated sugar solutions may be used, which may optionally contain gum arabic, talc, polyvinyl pyrrolidone, carbopol gel, polyethylene glycol, and/or titanium dioxide, lacquer solution, and suitable organic solvents or solvent mixtures.
[0244] Pharmaceutical preparations which can be used orally include push-fit capsules made of gelatin, as well as soft, sealed capsules made of gelatin and a plasticizer such as glycerol or sorbitol. The push-fit capsules can contain active ingredients in admixture with filler such as sugar, binders such as starches, and/or lubricants such as talc or magnesium stearate and, optionally, stabilizers. In soft capsules, the active ingredients may be dissolved or suspended in suitable liquids, such as fatty oil, liquid paraffin, or liquid polyethylene glycols. Furthermore, stabilizers may be added. All formulations for oral administration should be in dosages suitable for such administration.
[0245] In some embodiments, the pharmaceutical composition of the present disclosure may comprise 0.1%-95% of the piperidine-2,6-dione derivatives as disclosed herein.
[0246] In some embodiments, the pharmaceutical composition of the present disclosure may comprise 1%-70% of the piperidine-2,6-dione derivatives as disclosed herein.
[0247] Under any circumstances, the composition or formulation to be administered may comprise some amount of the piperidine-2,6-dione derivatives as disclosed herein, which is effective to treat the disease/condition of a study subject to be treated.
Methods of Administration
[0248] At least one of the compounds of the present disclosure or the pharmaceutical compositions comprising at least one of the compounds of the present disclosure may be administered to the patient by any suitable means and/or by any means that topically delivers the compounds of the present disclosure. Non-limiting examples of methods of administration include, among others, (a) administration though oral pathways, which administration includes administration in capsule, tablet, granule, spray, syrup, or other such forms; (b) administration through non-oral pathways such as rectal, vaginal, intraurethral, intraocular, intranasal, or intraauricular, which administration includes administration as an aqueous suspension, an oily preparation or the like or as a drip, spray, suppository, salve, ointment or the like; (c) administration via injection, subcutaneously, intraperitoneally, intravenously, intramuscularly, intradermally, intraorbitally, intracapsularly, intraspinally, intrasternally, or the like, including infusion pump delivery; (d) administration locally such as by injection directly in the renal or cardiac area, e.g., by depot implantation; as well as (e) administration topically; as deemed appropriate by those of skill in the art for bringing the compound of the present disclosure into contact with living tissue.
[0249] The most suitable route depends on the nature and severity of the condition to be treated. A person having ordinary skill in the art also knows determination of methods of administration (buccal, intravenous, inhalation subcutaneous, rectal and the like), dosage form, suitable pharmaceutical excipients and other events regarding delivering the compound to a subject in need thereof.
[0250] Pharmaceutical compositions suitable for administration include compositions where the active ingredients are contained in an amount effective to achieve its intended purpose. The therapeutically effective amount of the compounds disclosed herein required as a dose will depend on the route of administration, the type of animal, including human, being treated, and the physical characteristics of the specific animal under consideration. The dose can be tailored to achieve a desired effect, but will depend on such factors as weight, diet, concurrent medication and other factors which those skilled in the medical arts will recognize. More specifically, a therapeutically effective amount means an amount of compound effective to prevent, alleviate or ameliorate symptoms of disease or prolong the survival of the subject being treated. Determination of a therapeutically effective amount is well within the capability of those skilled in the art, especially in light of the detailed disclosure provided herein.
[0251] As will be readily apparent to one skilled in the art, the useful in vivo dosage to be administered and the particular mode of administration will vary depending upon the age, weight and mammalian species treated, the particular compounds employed, and the specific use for which these compounds are employed. The determination of effective dosage levels, that is the dosage levels necessary to achieve the desired result, can be accomplished by one skilled in the art using routine pharmacological methods. Typically, human clinical applications of products are commenced at lower dosage levels, with dosage level being increased until the desired effect is achieved. Alternatively, acceptable in vitro studies can be used to establish useful doses and routes of administration of the compositions identified by the present methods using established pharmacological methods.
[0252] In non-human animal studies, applications of potential products are commenced at higher dosage levels, with dosage being decreased until the desired effect is no longer achieved or adverse side effects disappear. The dosage may range broadly, depending upon the desired affects and the therapeutic indication. Typically, dosages may be between about 10 microgram/kg and 1000 mg/kg body weight, in some embodiments, between about 100 microgram/kg and 300 mg/kg body weight. Alternatively dosages may be based and calculated upon the surface area of the patient, as understood by those of skill in the art.
[0253] The exact formulation, route of administration and dosage for the pharmaceutical compositions of the present disclosure can be chosen by the individual physician in view of the patient's condition. Typically, the dose range of the composition administered to the patient can be from about 0.5 to 1000 mg/kg of the patient's body weight. The dosage may be a single one or a series of two or more given in the course of one or more days, as is needed by the patient. In instances where human dosages for compounds have been established for at least some condition, the present disclosure will use those same dosages, or dosages that are between about 0.1% and 500%, in some embodiments, between about 25% and 250% of the established human dosage. Where no human dosage is established, as will be the case for newly-discovered pharmaceutical compounds, a suitable human dosage can be inferred from ED.sub.50 or ID.sub.50 values, or other appropriate values derived from in vitro or in vivo studies, as qualified by toxicity studies and efficacy studies in animals.
[0254] It should be noted that the attending physician would know how to and when to terminate, interrupt, or adjust administration due to toxicity or organ dysfunctions. Conversely, the attending physician would also know to adjust treatment to higher levels if the clinical response were not adequate (precluding toxicity). The magnitude of an administrated dose in the management of the disorder of interest will vary with the severity of the condition to be treated and to the route of administration. The severity of the condition may, for example, be evaluated, in part, by standard prognostic evaluation methods. Further, the dose and perhaps dose frequency, will also vary according to the age, body weight, and response of the individual patient. A program comparable to that discussed above may be used in veterinary medicine.
[0255] Although the exact dosage will be determined on a drug-by-drug basis, in most cases, some generalizations regarding the dosage can be made. The daily dosage regimen for an adult human patient may be, for example, an oral dose of between 0.1 mg and 2000 mg of each active ingredient, in some embodiments, between 1 mg and 2000 mg, e.g. 5 to 1500 mg. In other embodiments, an intravenous, subcutaneous, or intramuscular dose of each active ingredient of between 0.01 mg and 1000 mg, in some embodiments, between 0.1 mg and 1000 mg, e.g. 1 to 800 mg is used. In cases of administration of a pharmaceutically acceptable salt, dosages may be calculated as the free base. In some embodiments, the composition is administered 1 to 4 times per day. Alternatively the compositions of the disclosure may be administered by continuous intravenous infusion, in some embodiments, at a dose of each active ingredient up to 2000 mg per day. As will be understood by those of skill in the art, in certain situations it may be necessary to administer the compounds disclosed herein in amounts that exceed, or even far exceed, the above-stated dosage range in order to effectively and aggressively treat particularly aggressive diseases or infections. In some embodiments, the compounds will be administered for a period of continuous therapy, for example for a week or more, or for months or years.
[0256] Dosage amount and interval may be adjusted individually to provide plasma levels of the active moiety which are sufficient to maintain the modulating effects, or minimal effective concentration (MEC). The MEC will vary for each compound but can be estimated from in vitro data. Dosages necessary to achieve the MEC will depend on individual characteristics and route of administration. However, HPLC assays or bioassays can be used to determine plasma concentrations.
[0257] Dosage intervals can also be determined using MEC value. Compositions should be administered using a regimen which maintains plasma levels above the MEC for 10-90% of the time, in some embodiments, between 30-90% and in some embodiments, between 50-90%.
[0258] In cases of local administration or selective uptake, the effective local concentration of the drug may not be related to plasma concentration.
[0259] The amount of composition administered will, of course, be dependent on the subject being treated, on the subject's weight, the severity of the affliction, the manner of administration and the judgment of the prescribing physician.
[0260] Compounds disclosed herein can be evaluated for efficacy and toxicity using known methods. For example, the toxicology of a particular compound, or of a subset of the compounds, sharing certain chemical moieties, may be established by determining in vitro toxicity towards a cell line, such as a mammalian, and in some embodiments, human, cell line. The results of such studies are often predictive of toxicity in animals, such as mammals, or more specifically, humans. Alternatively, the toxicity of particular compounds in an animal model, such as mice, rats, rabbits, or monkeys, may be determined using known methods. The efficacy of a particular compound may be established using several recognized methods, such as in vitro methods, animal models, or human clinical trials. Recognized in vitro models exist for nearly every class of condition, including but not limited to cancer, cardiovascular disease, and various immune dysfunction. Similarly, acceptable animal models may be used to establish efficacy of chemicals to treat such conditions. When selecting a model to determine efficacy, the skilled artisan can be guided by the state of the art to choose an appropriate model, dose, and route of administration, and regime. Of course, human clinical trials can also be used to determine the efficacy of a compound in humans.
[0261] The compositions may, if desired, be presented in a pack or dispenser device which may contain one or more unit dosage forms containing the active ingredient. The pack may for example comprise metal or plastic foil, such as a blister pack. The pack or dispenser device may be accompanied by instructions for administration. The pack or dispenser may also be accompanied with a notice associated with the container in form prescribed by a governmental agency regulating the manufacture, use, or sale of pharmaceuticals, which notice is reflective of approval by the agency of the form of the drug for human or veterinary administration. Such notice, for example, may be the labeling approved by the U.S. Food and Drug Administration for prescription drugs, or the approved product insert. Compositions comprising a compound of the disclosure formulated in a compatible pharmaceutical carrier may also be prepared, placed in an appropriate container, and labeled for treatment of an indicated condition.
EXAMPLES
[0262] The following abbreviations were used in the discussion, examples and preparations.
[0263] LPS: lipopolysaccharide;
[0264] PBS: phosphate buffered saline;
[0265] Tween-20
[0266] PBST: 0.05% of Tween-20 in phosphate buffered saline;
[0267] HRP: horseradish peroxidase;
[0268] TMB: 3,3',5,5'-tetramethylbenzidine;
[0269] DSS: dextra sulfate sodium;
[0270] 5-ASA: 5-aminosalicylic acid;
[0271] CMC-Na: sodium carboxymethyl cellulose;
[0272] DNBS: dinitrobenzenesulfonic acid;
[0273] 1% CMC-Na: 1% (weight/volume) of CMC-Na in water;
[0274] 1.6% DSS: 1.6% (weight/volume) of DSS in water;
[0275] 0.9% DSS: 0.9% (weight/volume) of DSS in water;
[0276] mg/Kg: milligram/kilogram;
[0277] mg/Kg/d: milligram/kilogram/day;
[0278] ml/Kg: milliliter/kilogram;
[0279] cm: centimeter;
[0280] Mean.+-.SD: Mean.+-.standard deviation;
[0281] 30% ethanol: 30% (volume/volume) of ethanol in water;
[0282] cm.sup.2: square centimeter;
[0283] U/mg: unit/milligram;
[0284] g: gram;
[0285] mmol: millimole;
[0286] mol/L: mole/liter;
[0287] i.g: intragastric administration;
[0288] ml: milliliter;
[0289] MPO: myeloperoxidase;
[0290] .mu.L: microliter;
[0291] nm: nanometer;
[0292] s: second;
[0293] OD value: optical density value;
[0294] .mu.m: micron;
[0295] Qd, QD, Q.D or qd: once a day;
[0296] The following instruments or devices may be used in the experiments:
TABLE-US-00003 Instruments Type Company Animal Endoscopy VET-6011 Shanghai Aohua Endoscopy Co., Ltd. Fully Automated Cobas c 311 Roche Biochemical Analyzer ELIASA Multiskan GO Thermo Scientific ELIASA Model 680 Biorad Microplate Washer Model 1575 Biorad Animal Fixator Chinese Suzhou Monkey Animal Patent No. Experimental Equipment ZL200800089997.7 Technology Co., Ltd. High-speed Centrifuge TGL-16B Shanghai Anting Scientific Instrument Factory Triple Quadrupole Mass API3000 Applied Biosystems Spectrometer (TQMS) High Performance Liquid LC-20A Shimadzu Corporation Chromatography (HPLC) Vortex Mixer QL901 Haimen Kylin-Bell Lab Instruments Co., Ltd. Low-temperature Heraeus Thermo Scientific High-speed Centrifuge Multifuge X3R Fully Automated Leica ASP200S Leica Instruments Vacuum Processor Germany GmbH Embedding Machine Leica Leica Instruments EG1150H + C Germany GmbH Manual Microtome Leica RM2235 Leica Instruments Germany GmbH Water Bath for Paraffin Leica-HI1210 Leica Instruments Sections Germany GmbH Fully Automated Stainer Leica-ST5020 Leica Instruments Germany GmbH Fully Automated Glass Leica CV5030 Leica Instruments Coverslipper Germany GmbH
[0297] The Chinese patent No. ZL200510013292.3 discloses the preparation process of 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e, which is incorporated herein by reference in its entirety.
Example 1
Preparation of 4-acetylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione
[0298] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione was prepared in accordance with the process disclosed in the Chinese patent No. ZL200510013292.3.
[0299] In dichloromethane (DCM) (30 mL) was dissolved 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e (3.0 g). Triethylamine (TEA) (2.75 g, 3.77 mL) and 4-dimethylaminopyridine (DMAP) (0.126 g) were added into the solution. The air was replaced with argon. The temperature of the reaction system was lowered to 0.degree. C. To the reaction system was added acetyl chloride (4.59 g, 4.16 mL) with stirring. The temperature of the reaction system was slowly heated to the room temperature until the reaction was complete. To the reaction solution was added water (60 mL) to quench the reaction. The solution was extracted once with dichloromethane. The organic phase was washed once with saturated aqueous solution of sodium chloride, dried over anhydrous magnesium sulfate and filtered. The solvent was evaporated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give a solid product (1.8 g). The solid product was dissolved with dichloromethane. To the solution was added dropwise diethyl ether to separate out a product. The resultant product was dissolved with dichloromethane. To the solution was added diethyl ether to separate out a product. The resultant product was dried under reduced pressure to give the title compound of Example 1 as a white solid (1.3 g) (HPLC purity: 98.06%). Yield: 38.3%.
[0300] .sup.1HNMR (deuterated chloroform (CDCl.sub.3), 400 MHz) .delta. 9.411 (bs, 1H), 8.823-8.802 (d, 1H), 7.730-7.690 (dd, 1H), 7.557-7.537 (dd, 1H), 4.995-4.950 (m, 1H), 4.146-4.001 (m, 2H), 3.555-3.521 (m, 2H), 3.345 (s, 3H), 3.048-2.956 (m, 1H), 2.848-2.719 (m, 2H), 2.267 (s, 3H), 2.186-2.086 (m, 1H). MS(m/e): 374.21 (M+H+).
Example 2
Preparation of 4-methyl amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione
[0301] To a reaction flask were added 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e (4.0 g), di-tert-butyl dicarbonate (10.528 g) and 4-dimethylaminopyridine (0.147 g). To the reaction flask was added tetrahydrofuran (40 mL). The solution was stirred at the room temperature overnight. The reaction solution was concentrated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give 4-di-BOC-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-- 1,3-dione (3.3 g) as a white solid. Yield: 74.1%. MS(m/e): 554.40 (M+Na.sup.+).
[0302] 4-di-BOC-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoin- dolin-1,3-dione (2.05 g) was dissolved in dichloromethane (600 mL). To the solution was added tetrahydrofuran (3 mL). The reaction solution was stirred at the room temperature until the reaction was complete. To the resultant solution was added saturated aqueous solution of sodium carbonate to quench the reaction. The solution was separated. The organic phase was dried over anhydrous magnesium sulfate and filtered to give a filtrate. The filtrate was concentrated under reduced pressure to give 4-BOC-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3- -dione (1.66 g) (HPLC purity: 98.62%). Yield: 100%. MS(m/e): 454.29 (M+Na.sup.+).
[0303] To a reaction flask were added 4-Boc-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3- -dione (3.0 g), methyl iodide (2.96 g) and potassium carbonate (2.88 g). To the reaction flask was added N,N-dimethylformamide (30 mL). The reaction solution was stirred at the room temperature overnight. The reaction solution was diluted with dichloromethane (100 mL). The organic phase was washed with water. The aqueous phase was extracted with dichloromethane. The organic phases were combined. The combined organic phase was washed with saturated aqueous solution of sodium chloride, dried over anhydrous magnesium sulfate and filtered to give a filtrate. The filtrate was concentrated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give 4-BOC-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione as a yellow oil (2.91 g) (HPLC purity: 96.43%). Yield: 94.0%. MS(m/e): 468.31 (M+Na.sup.+).
[0304] 4-BOC-methylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-is- o indolin-1,3-dione (2.9 g) was dissolved in dichloromethane (30 mL). To the solution was added tetrahydrofuran (6 mL). The reaction solution was stirred at the room temperature over 2 hours. The resultant solution was concentrated under reduced pressure to give a crude product. The crude product was dissolved in methyl tert-butyl ether (35 mL). The solution was stirred overnight to separate out a yellow solid product and filtered. The product was dried under reduced pressure to give the title compound as a yellow solid (1.83 g) (HPLC purity: 98.62%). Yield: 81.6%.
[0305] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.538-7.499 (dd, 1H), 7.112-7.094 (d, 1H), 6.887-6.866 (d, 1H), 4.949-4.904 (m, 1H), 4.122-3.999 (m, 2H), 3.543-3.510 (m, 2H), 3.341 (s, 3H), 2.989-2.950 (m, 1H), 2.981 (s, 3H), 2.820-2.700 (m, 2H), 2.115-2.060 (m, 1H). MS(m/e): 346.24 (M+H+).
Example 3
Preparation of 4-dimethylamino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-- 1,3-dione
[0306] To a pressure-resistant reaction tube were added 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e (3.0 g), methyl iodide (14.2 g) and potassium carbonate (6.25 g). To the pressure-resistant reaction tube was added N,N-dimethylformamide (20 mL). The reaction tube was sealed and heated to 80.degree. C. in an oil bath. The reaction solution was stirred over 80 hours. The resultant solution was diluted with dichloromethane (200 mL) and filtered to give a filtrate. The filtrate was concentrated under reduced pressure to give a black oily crude product. The crude product was purified with silica gel column chromatography to give a brown solid (3.2 g). The brown solid was dissolved in ethyl acetate (20 mL) and stirred overnight. The resultant solution was filtered to give a solid. The solid was dried under reduced pressure to give the title compound as a yellow powdery solid (1.67 g) (HPLC purity: 97.22%). Yield: 51.4%.
[0307] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.543-7.504 (dd, 1H), 7.308-7.287 (d, 1H), 7.121-7.100 (d, 1H), 5.004-4.959 (m, 1H), 4.115-3.999 (m, 2H), 3.537-3.507 (m, 2H), 3.337 (s, 3H), 3.108 (s, 6H), 2.982-2.944 (m, 1H), 2.803-2.751 (m, 2H), 2.094-2.079 (m, 1H). MS(m/e): 360.27 (M+H+).
Example 4
Preparation of 4-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne
[0308] To a reaction flask were added 1-(2-methoxyethyl)-3-benzyloxyamido-2,6-piperidinedione acetate (35 g), palladium on carbon (3.5 g, content of palladium: 10%) and acetic acid (13 mL). The air in the flask was replaced with hydrogen three times. The reaction solution was stirred at the room temperature over 72 hours. The resultant solution was filtered to give a filtrate, which was directly used in the next step.
[0309] To a reaction flask were added the above filtrate (comprising 2.0 g (calculated) of 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate), 3-fluorophthalic anhydride (1.35 g), sodium acetate (0.67 g) and acetic acid (43 mL). The solution was heated to 140.degree. C. in an oil bath and stirred to react over 5 hours. The resultant solution was concentrated under reduced pressure to give a black crude product. The crude product was purified with silica gel column chromatography to give the title compound as a white solid (1.56 g) (HPLC purity: 99.88%). Yield: 57.4%.
[0310] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.796-7.746 (m, 1H), 7.717-7.700 (d, 1H), 7.447-7.403 (m, 1H), 5.028-4.982 (m, 1H), 4.127-3.998 (m, 2H), 3.541-3.510 (m, 2H), 3.340 (s, 3H), 3.019-2.951 (m, 1H), 2.895-2.739 (m, 2H), 2.171-2.085 (m, 1H). MS(m/e): 357.19 (M+Na.sup.+).
Example 5
Preparation of 4,5,6,7-tetrafluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindo- lin-1,3-dione
[0311] Using the preparation process in Example 4, 2,3,4,5-tetrafluorophthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to give the title compound as a white solid (2.93 g) (HPLC purity: 97.58%). Yield: 78.6%.
[0312] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 4.999-4.953 (m, 1H), 4.118-3.984 (m, 2H), 3.530-3.500 (m, 2H), 3.333 (s, 3H), 3.029-2.991 (m, 1H), 2.803-2.764 (m, 2H), 2.136-2.117 (m, 1H). MS(m/e): 389.22 (M+H.sup.+).
Example 6
Preparation of 4-amino-2-(1-(2-hydroxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e
[0313] To a reaction flask were added 3-amino-N-(2,6-dioxo-3-piperidinyl) phthalimide (3.5 g), 2-bromoethanol (4.8 g), potassium carbonate (1.77 g) and N,N-dimethylformamide (35 mL). The reaction flask was heated to 40.degree. C. in an oil bath. The reaction solution was stirred over 80 hours. The reaction system was cooled to the room temperature. To the reaction system was added water (80 mL) to quench the reaction. To the solution was added ethyl acetate. The resultant solution was stirred to separate out a solid. The resultant mixture was filtered under reduced pressure. The filter cake was discarded and the filtrate was collected and separated. The aqueous phase was back-extracted with ethyl acetate four times. The organic phases were combined. The combined organic phase was washed with saturated aqueous solution of sodium chloride, dried over anhydrous magnesium sulfate and filtered. The organic phase was concentrated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give the title compound as a yellow solid (2.0 g) (HPLC purity: 95.53%). Yield: 49.2%.
[0314] .sup.1HNMR(CDCl.sub.3, 400 MHzz) .delta. 7.457-7.419 (t, 1H), 7.176-7.158 (d, 1H), 6.886-6.865 (d, 1H), 4.990-4.945 (m, 1H), 4.135-4.065 (m, 2H), 3.813-3.785 (m, 2H), 3.006-2.966 (m, 1H), 2.829-2.714 (m, 2H), 2.153-2.114 (m, 1H). MS(m/e): 340.18 (M+Na.sup.+).
Example 7
Preparation of 5-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e
[0315] Using the preparation process in Example 4, 4-nitrophthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to give 5-nitro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-dio- ne as a white solid (2.8 g) (HPLC purity: 99.62%). Yield: 54.5. MS(m/e): 384.18 (M+Na.sup.+).
[0316] To a reaction flask were added 5-nitro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-dio- ne (2.6 g), palladium on carbon (0.26 g) and tetrahydrofuran (30 mL). The air in the flask was replaced with hydrogen three times. The reaction solution was stirred at the room temperature over 20 hours until the reaction was complete. The resultant solution was filtered to give a filtrate. The filtrate was concentrated under reduced pressure with water pump to give a crude product. The crude product was dissolved and purified with ethyl acetate to give the title compound as a yellow solid (1.56 g) (HPLC purity: 95.74%). Yield: 65.4%.
[0317] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.607-7.587 (d, 1H), 7.012-7.007 (d, 1H), 6.846-6.820 (dd, 1H), 4.970-4.925 (m, 1H), 4.135-3.982 (m, 2H), 3.540-3.509 (m, 2H), 3.340 (s, 3H), 3.024-2.903 (m, 1H), 2.829-2.710 (m, 2H), 2.117-2.062 (m, 1H). MS(m/e): 354.22 (M+Na.sup.+).
Example 8
Preparation of 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindoli- n-1,3-dione
[0318] To a reaction flask were added 4-hydroxyphthalate dimethyl (125 g) and concentrated sulfuric acid (600 mL). The solution was stirred and cooled to 0.degree. C. To the reaction system was slowly added dropwise fuming nitric acid (39.44 g). The temperature of the reaction system was controlled to be below 5.degree. C. The addition of fuming nitric acid was complete in about 1 hour. The ice bath was removed. The solution was heated to the room temperature and stirred over 22 hours until the reaction was complete. The resultant solution was slowly poured into ice water to quench the reaction. The aqueous phase was extracted with ethyl acetate five times. The organic phases were combined. The combined organic phase was dried over anhydrous magnesium sulfate and filtered. The filtrate was collected and concentrated under reduced pressure with water pump and oil pump successively to give 4-hydroxy-3-nitrophthalate dimethyl (169 g) (HPLC purity: 36.54%), which was directly used in the next step. MS(m/e): 254.07 (M-H).sup.-.
[0319] To a reaction flask were added 4-hydroxy-3-nitrophthalate dimethyl (164 g), benzyl bromide (135.9 g), potassium carbonate (400 g) and acetone (1730 mL). The reaction flask was heated to 70.degree. C. in an oil bath. The solution was stirred over 23 hours until the reaction was complete. The reaction system was cooled to the room temperature and concentrated under reduced pressure. The filter cake was washed with dichloromethane. The filtrate was collected and concentrated under reduced pressure. To the concentrate were added dichloromethane (2000 mL) and saturated aqueous solution of sodium chloride (1000 mL). The resultant solution was extracted. The organic phase was dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to give a yellow solid crude product. The crude product was recrystallized and purified with ethyl acetate to give 4-benzyloxy-3-nitrophthalate dimethyl as a white solid (40 g) (HPLC purity: 96.96%). Yield: 18.1%. MS(m/e): 346.14 (M+H.sup.+).
[0320] To a reaction flask were added 4-benzyloxy-3-nitrophthalate dimethyl (30 g) and ethanol (300 mL). To the reaction flask was added preformulated aqueous solution of sodium hydroxide (24 g of sodium hydroxide in 300 mL of water). The reaction flask was heated to 70.degree. C. in an oil bath. The solution was stirred over 7 hours until the reaction was complete. The reaction system was cooled to the room temperature and concentrated under reduced pressure to evaporate most of ethanol. To the concentrate was added 4 N hydrochloric acid at the room temperature until the pH of the solution was adjusted to 2 such that a lot of white solids were separated out. The resultant mixture was filtered under reduced pressure. The filter cake was collected and dried under reduced pressure with oil pump to give 4-benzyloxy-3-nitrophthalic acid as a white solid (27.5 g) (HPLC purity: 99.55%). Yield: 100%. MS(m/e): 316.15 (M-H).sup.-.
[0321] To a reaction flask were added 4-benzyloxy-3-nitrophthalic acid (19.1 g) and acetic anhydride (120 mL). The flask was heated to 140.degree. C. in an oil bath. The solution was stirred over 6 hours. The reaction system was cooled to the room temperature. The resultant solution was concentrated under reduced pressure to give a crude product. The crude product was dissolved and purified with methyl tert-butyl ether and n-hexane to give 4-benzyloxy-3-nitrophthalic anhydride as a brown solid (16.8 g). Yield: 93.3%.
[0322] Using the preparation process in Example 4, 4-benzyloxy-3-nitrophthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to prepare 4-nitro-5-benzyloxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl- )-isoindolin-1,3-dione as a white solid (22.3 g) (HPLC purity: 99.69%). Yield: 85.0%. MS(m/e): 468.22 (M+H.sup.+).
[0323] Using the preparation process in Example 7, 4-nitro-5-benzyloxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoind- olin-1,3-dione was used to give the title compound as a yellow solid (18.4 g) (HPLC purity: 99.03%). Yield: 86.8%.
[0324] .sup.1HNMR (dimethyl sulfoxide (DMSO), 400 MHz) .delta. 10.820 (s, 1H), 6.974-6.955 (d, 1H), 6.915-6.896 (d, 1H), 5.936-5.935 (bs, 2H), 5.099-5.053 (q, 1H), 3.917-3.730 (m, 2H), 3.363-3.331 (t, 2H), 3.217 (s, 3H), 3.002-2.911 (m, 1H), 2.762-2.702 (m, 1H), 2.560-2.450 (m, 1H), 2.027-1.963 (m, 1H). MS(m/e): 348.17 (M+H+).
Example 9
Preparation of 4-amino-5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione
[0325] To a reaction flask were added 4-fluorophthalic acid (1.0 g) and concentrated sulfuric acid (5 mL). The solution was stirred and cooled to 0.degree. C. To the reaction system was slowly added dropwise concentrated nitric acid (1.05 g, content: 65%). After the addition was complete, the resultant solution was heated to 80.degree. C. in an oil bath. The solution was stirred over 7 hours. HPLC was used to monitor the reaction until the reaction was complete. The resultant solution was slowly poured into ice water (20 g) to quench the reaction. The aqueous phase was extracted with ethyl acetate. The organic phases were combined. The combined organic phase was washed with saturated aqueous solution of sodium chloride, dried over anhydrous magnesium sulfate and filtered. The filtrate was collected and concentrated under reduced pressure with water pump and oil pump successively to give a yellow solid (1.3 g), which was directly used in the next step.
[0326] To a reaction flask was added thionyl chloride (5 mL). The reaction system was heated to 90.degree. C. in an oil bath and stirred over 11 hours. The reaction system was then cooled to the room temperature. The reaction solution was concentrated under reduced pressure with water pump to give a crude product as a yellow oil (0.56 g), which was directly used in the next step.
[0327] Using the preparation process in Example 4, 4-fluoro-3-nitrophthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to prepare 5-fluoro-4-nitro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-i- soindolin-1,3-dione as a black solid (0.3 g) (HPLC purity: 90.63%). MS(m/e): 380.21 (M+H.sup.+).
[0328] Using the preparation process in Example 7, 5-fluoro-4-nitro-2-(1-(2-methoxyethyl)-2,6-di oxopiperidin-3-yl)-isoindolin-1,3-dione was used to give the title compound as a yellow solid (0.137 g) (HPLC purity: 97.59%). Yield: 57.2%.
[0329] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.256-7.208 (dd, 1H), 7.158-7.129 (dd, 1H), 5.269 (bs, 2H), 4.959-4.914 (m, 1H), 4.140-3.988 (m, 2H), 3.544-3.512 (m, 2H), 3.341 (s, 3H), 3.000-2.961 (m, 1H), 2.795-2.745 (m, 2H), 2.121-2.081 (m, 1H). MS(m/e): 350.2 (M+H+).
Example 10
Preparation of 4-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindol-- 1,3-dione
[0330] 4-BOC-amino-7-hydroxyphthalate dimethyl was prepared in accordance with Journal of Organic Chemistry, 62(12), 4088-4096; 1997.
[0331] To a reaction flask were added 3-BOC-amino-6-hydroxyphthalate dimethyl (3.0 g), 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate (4.5 g) and pyridine (60 mL). The reaction flask was heated to 100.degree. C. in an oil bath. The reaction solution was stirred over 40 hours and then cooled to the room temperature. The resultant solution was concentrated to give a crude product. The crude product was purified with silica gel column chromatography to give 4-BOC-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoi- ndolin-1,3-dione as a yellow solid (2.35 g) (HPLC purity: 95.96%). Yield: 48.8%. MS(m/e): 470.28 (M+Na.sup.+).
[0332] 4-BOC-amino-7-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl- )-isoindolin-1,3-dione (2.25 g) was dissolved in dichloromethane (20 mL). To the solution was added tetrahydrofuran (4 mL). The resultant solution was stirred at the room temperature over 6 hours until the reaction was complete. The solution was concentrated under reduced pressure to give a crude product. The crude product was dissolved and purified with methyl tert-butyl ether to give the title compound as brown solid (1.413 g) (HPLC purity: 95.35%). Yield: 77.4%.
[0333] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.020-6.998 (d, 1H), 6.858-6.835 (d, 1H), 4.934-4.889 (m, 1H), 4.145-3.995 (m, 2H), 3.555-3.521 (m, 2H), 3.346 (s, 3H), 3.031-2.926 (m, 1H), 2.827-2.742 (m, 2H), 2.140-2.059 (m, 1H). MS(m/e): 348.22 (M+H.sup.+).
Example 11
Preparation of 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-d- ione
[0334] To a reaction flask were added 3-amino-N-(2,6-dioxo-3-piperidinyl) phthalimide (2.5 g), 2-chloroethyl methyl sulfide (2.55 g), potassium carbonate (3.8 g), sodium iodide (0.28 g) and N,N-dimethylformamide (35 mL). The reaction flask was heated to 50.degree. C. in an oil bath. The reaction solution was stirred over 20 hours until the reaction was complete. To the reaction solution were added dichloromethane and water to extract. The aqueous phase was back-extracted with dichloromethane twice. The organic phases were combined. The combined organic phase was dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give the title compound as a yellow solid (2.111 g) (HPLC purity: 96.68%). Yield: 55.3%.
[0335] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.457-7.418 (dd, 1H), 7.180-7.161 (dd, 1H), 6.885-6.863 (dd, 1H), 4.966-4.921 (m, 1H), 4.110-3.973 (m, 2H), 2.994-2.926 (m, 1H), 2.820-2.753 (m, 2H), 2.683-2.646 (t, 2H), 2.144 (s, 3H), 2.125-2.079 (m, 1H). MS(m/e): 348.19 (M+H+).
Example 12
Preparation of 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione
[0336] To a reaction flask were added 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-- dione (4.85 g), m-chloroperoxybenzoic acid (3.13 g) and dichloromethane (170 mL). The solution was stirred at the room temperature over 18 hours. To the reaction system was added saturated aqueous solution of sodium bicarbonate (100 mL) to extract. The aqueous phase was back-extracted with dichloromethane (150 mL.times.3). The organic phases were combined. The combined organic phase was dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to give a crude product. The crude product was purified with silica gel column chromatography to give the title compound 4-amino-2-(1-(2-methylsulfinylethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-- 1,3-dione (0.846 g) (HPLC purity: 95.09%). Yield: 16.7%.
[0337] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.462-7.423 (dd, 1H), 7.172-7.155 (d, 1H), 6.895-6.874 (d, 1H), 4.974-4.931 (m, 1H), 4.371-4.192 (m, 2H), 3.073-2.909 (m, 3H), 2.827-2.696 (m, 2H), 2.650-2.647 (d, 3H), 2.144-2.104 (m, 1H). MS(m/e): 364.22 (M+H.sup.+).
Example 13
Preparation of 4-amino-2-(1-(2-methyl sulfonylethyl)-2,6-dixopiperidin-3-yl)isoindolin-1,3-dione
[0338] In accordance with the preparation process in Example 12, 4-amino-2-(1-(2-methylthioethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-- dione was used to give the title compound as a yellow solid (1.78 g) (HPLC purity: 99.47%). Yield: 33.6%.
[0339] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.470-7.431 (dd, 1H), 7.184-7.167 (d, 1H), 6.896-6.876 (d, 1H), 4.988-4.943 (m, 1H), 4.400-4.231 (m, 2H), 3.311-3.277 (t, 2H), 2.999 (s, 3H), 3.012-2.932 (m, 1H), 2.834-2.697 (m, 2H), 2.153-2.110 (m, 1H). MS(m/e): 380.23 (M+H.sup.+).
Example 14
Preparation of 4-amino-2-(1-(2-methoxyethyl)-3-fluoro-2,6-dioxopiperidin-3-yl)isoindolin- -1,3-dione
[0340] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1- ,3-dione was prepared in accordance with PCT Int. Appl., 2006105697, 12 Oct. 2006.
[0341] 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1- ,3-dione (5 g) was dissolved in tetrahydrofuran (100 mLl) under argon atmosphere. The solution was cooled to -78.degree. C. To the solution was slowly added dropwise lithium hexamethyldisilazide (40 mL). After addition, the resultant solution was stirred over 1 hour. To the solution was added N-fluorobenzenesulfonimide (11.67 g in 30 mL of tetrahydrofuran). After addition, the resultant solution was stirred over 1 hour. The resultant solution was slowly heated to the room temperature and reacted overnight. To the reaction mixture were added saturated aqueous solution of ammonium chloride (150 mL) and ethyl acetate (150 mL). The solution was separated. The aqueous phase was extracted with ethyl acetate (100 mL). The organic phases were combined. The combined organic phase was washed with saturated aqueous solution of sodium chloride (200 mL), dried over anhydrous sodium sulfate and filtered. The organic phase was concentrated. The residue was purified with column chromatography (ethyl acetate/petroleum ether=1/3) to give the title compound as a yellow solid (968 mg) (HPLC purity: 97.20%). Yield: 18.4%.
[0342] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.461-7.499 (dd, 1H), 7.171-7.153 (d, 1H), 6.931-6.899 (d, 1H), 4.193-4.024 (m, 2H), 3.670-3.640 (t, 2H), 3.606-3.5558 (m, 1H), 3.020 (s, 3H), 2.371-2.960 (m, 1H), 2.656-2.555 (m, 1H), 2.433-2.342 (m, 1H). MS(m/e): 372.22 (M+Na.sup.+).
Example 15
Preparation of 4-amino-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)isoindolin- -1)isoindolin-1,3-dione
[0343] 3-amino-3-methylpiperidin-2,6-dione hydrochloride was prepared in accordance with PCT Int. Appl., 2006081251, 3 Aug. 2006.
[0344] Using the preparation process in Example 4, 3-amino-3-methylpiperidin-2,6-dione hydrochloride and 3-nitrophthalic anhydride were reacted to give 2-(3-methyl-2,6-di oxopiperidin-3-yl)-4-nitroisoindolin-1,3-dione as a white solid (2.7 g) (HPLC purity: 66.03%). Yield: 69.1%. MS(m/e): 318.12 (M+H+).
[0345] Using the preparation process in Example 11, 2-(3-methyl-2,6-dioxopiperidin-3-yl)-4-nitroisoindolin-1,3-dione was used to prepare 4-nitro-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)-isoindoli- n-1,3-dion e as a yellow solid (0.736 g) (HPLC purity: 96.45%). Yield: 28.3%. MS(m/e): 376.26 (M+H+).
[0346] Using the preparation process in Example 7, 4-nitro-2-(1-(2-methoxyethyl)-3-methyl-2,6-dioxopiperidin-3-yl)-isoindoli- n-1,3-dion e was used to give the title compound as yellow solid (0.572 g) (HPLC purity: 94.51%). Yield: 84.1%.
[0347] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.430-7.391 (t, 1H), 7.104-7.086 (d, 1H), 6.854-6.833 (d, 1H), 4.112-4.074 (m, 2H), 3.615-3.580 (m, 2H), 3.373 (s, 3H), 2.844-2.793 (m, 1H), 2.734-2.690 (m, 2H), 2.046-1.993 (m, 1H), 1.969 (s, 3H). MS(m/e): 368.22 (M+Na.sup.+). MS(m/e): 368.22 (M+Na.sup.+).
Example 16
Preparation of 4-acetylamino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1- ,3-dione
[0348] N-((benzyloxy)carbonyl)-glutamic anhydride was prepared in accordance with Archives of Pharmacal Research, 31(7), 834-837; 2008.
[0349] To a reaction flask were added N-((benzyloxy)carbonyl)-glutamic anhydride (19 g), 3-methoxypropylamine (6.4 g), triethylamine (7.3 g), 4-dimethylaminopyridine (0.88 g) and tetrahydrofuran (270 mL). The reaction solution was stirred at the room temperature over 40 hours until the reaction was complete. The resultant reaction was the tetrahydrofuran solution of the product, which was directly used in the next step.
[0350] N,N'-carbonyldiiazole (23.4 g) was added into the above reaction solution in batch. The resultant solution was stirred at the room temperature over 72 hours until the reaction was complete. The solution was concentrated under reduced pressure. To the concentrate was added dichloromethane (500 mL). The resultant solution was washed with 1 N hydrochloric acid (250 mL), saturated aqueous solution of sodium bicarbonate (250 mL) and saturated aqueous solution of sodium chloride (250 mL). The organic phase was dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to give 1-(2-methoxypropyl)-3-benzyloxyamido-2,6-piperidinedione as a brown oil (23.7 g), which was directly used in the next step. MS(m/e): 357.24 (M+Na+).
[0351] Using the preparation process in Example 4, 1-(2-methoxypropyl)-3-benzyloxyamido-2,6-pi peridinedione was used to prepare the title compound as a white solid (0.434 g) (HPLC purity: 97.71%). Yield: 12.5%.
[0352] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 9.411 (bs, 1H), 8.822-8.801 (d, 1H), 7.729-7.689 (dd, 1H), 7.555-7.535 (dd, 1H), 4.943-4.987 (m, 1H), 3.993-3.879 (m, 2H), 3.428-3.397 (t, 2H), 3.304 (s, 3H), 3.005-2.936 (m, 1H), 2.825-2.705 (m, 2H), 2.266 (s, 3H), 2.146-2.092 (m, 1H), 1.880-1.802 (m, 2H). MS(m/e): 410.31 (M+Na.sup.+).
Example 17
Preparation of 4-fluoro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one
[0353] Using the preparation process in Example 4, 1-(2-methoxypropyl)-3-amino-2,6-piperidinedione acetate and 3-fluorophthalic anhydride were reacted to give the title compound as a yellowish sticky product (0.631 g) (HPLC purity: 97.42%). Yield: 31.1%.
[0354] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.792-7.743 (m, 1H), 7.715-7.697 (d, 1H), 7.444-7.400 (m, 1H), 4.978-4.932 (m, 1H), 3.988-3.871 (m, 2H), 3.421-3.390 (t, 2H), 3.300 (s, 3H), 3.000-2.938 (m, 1H), 2.851-2.691 (m, 2H), 2.142-2.091 (m, 1H), 1.872-1.797 (m, 2H). MS(m/e): 349.22 (M+H+).
Example 18
Preparation of 5-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne
[0355] Using the preparation process in Example 4, 5-nitrophthalic anhydride and 1-(2-methoxypropyl)-3-amino-2,6-piperidinedione acetate were reacted to prepare 5-nitro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one as a yellowish oily product (1.118 g) (HPLC purity: 98.44%). Yield: 20%. MS(m/e): 376.25 (M+H.sup.+).
[0356] Using the preparation process in Example 7, 5-nitro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one was used to give the title compound as a yellowish solid (0.697 g) (HPLC purity: 97.79%). Yield: 67.7%.
[0357] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.624-7.603 (d, 1H), 7.031-7.027 (d, 1H), 6.861-6.835 (dd, 1H), 4.925-4.879 (m, 1H), 3.977-3.863 (m, 2H), 3.421-3.390 (t, 2H), 3.301 (s, 3H), 2.969-2.922 (m, 1H), 2.820-2.676 (m, 2H), 2.108-2.056 (m, 1H), 1.869-1.794 (m, 2H). MS(m/e): 346.24 (M+H+).
Example 19
Preparation of 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dione
[0358] Using the preparation process in Example 16, N-((benzyloxy)carbonyl)-glutamic anhydride was used to prepare 1-(2-ethoxyethyl)-3-benzyloxyamido-2,6-piperidinedione as a brown oily crude product (17.2 g).
[0359] Using the preparation process in Example 4, 1-(2-ethoxyethyl)-3-amino-2,6-piperidinedione acetate and 3-nitrophthalic anhydride were reacted to prepare 4-nitro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-dion- e as an off-white solid (1.326 g) (HPLC purity: 95.24%). Yield: 39.4%. MS(m/e): 376.23 (M+H+).
[0360] Using the preparation process in Example 7, 4-nitro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-dion- e was used to prepare 4-amino-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-dion- e as a yellow solid (0.631 g) (HPLC purity: 94.95%). Yield: 51.7%.
[0361] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.450-7.411 (dd, 1H), 7.174-7.156 (d, 1H), 6.881-6.860 (d, 1H), 4.963-4.918 (m, 1H), 4.119-3.994 (m, 2H), 3.575-3.480 (m, 4H), 3.023-2.921 (m, 1H), 2.839-2.714 (m, 2H), 2.140-2.035 (m, 1H), 1.180-1.145 (t, 3H). MS(m/e): 346.24 (M+H.sup.+).
Example 20
Preparation of 4-fluoro-2-(1-(2-ethoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e
[0362] Using the preparation process in Example 4, 1-(2-ethoxyethyl)-3-amino-2.6-piperidinedione acetate and 3-fluorophthalic anhydride were reacted to give the title compound as a yellowish sticky product (1.566 g) (HPLC purity: 98.63%). Yield: 74.7%.
[0363] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.792-7.743 (m, 1H), 7.715-7.698 (d, 1H), 7.444-7.400 (m, 1H), 5.019-4.974 (m, 1H), 4.147-3.984 (m, 2H), 3.570-3.475 (m, 4H), 3.045-2.941 (m, 1H), 2.862-2.733 (m, 2H), 2.167-2.087 (m, 1H), 1.178-1.143 (t, 3H). MS(m/e): 349.23 (M+H.sup.+).
Example 21
Preparation of 5-fluoro-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one
[0364] Using the preparation process in Example 4, 4-fluorophthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to give the title compound as a white solid (2.905 g) (HPLC purity: 99.64%). Yield: 77.9%.
[0365] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.912-7.880 (dd, 1H), 7.572-7.549 (dd, 1H), 7.457-7.408 (dt, 1H), 5.023-4.978 (m, 1H), 4.144-3.988 (m, 2H), 3.544-3.512 (m, 2H), 3.342 (s, 3H), 3.056-2.933 (m, 1H), 2.859-2.738 (m, 2H), 2.175-2.080 (m, 1H). MS(m/e): 357.20 (M+Na.sup.+).
Example 22
Preparation of 4-amino-2-(1-(2-methoxybutyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e
[0366] Using the preparation process in Example 11, 3-amino-N-(2,6-dioxo-3-piperidinyl)-phthalimide and 1-bromo-4-methoxybutane were reacted to give the title compound as a yellow solid (2.625 g) (HPLC purity: 99.02%). Yield: 79.9%.
[0367] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.455-7.416 (dd, 1H), 7.178-7.160 (dd, 1H), 6.884-6.863 (dd, 1H), 4.927-4.882 (m, 1H), 3.858-3.816 (m, 2H), 3.402-3.371 (t, 2H), 3.317 (s, 3H), 3.007-2.916 (m, 1H), 2.819-2.685 (m, 2H), 2.117-2.067 (m, 1H), 1.651-1.571 (m, 4H). MS(m/e): 360.28 (M+H+).
Example 23
Preparation of 4-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-di- one
[0368] Using the preparation process in Example 4, 3-hydroxyphthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to give the title compound as a white solid (2.833 g) (HPLC purity: 99.54%). Yield: 80.1%.
[0369] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.649-7.609 (dd, 1H), 7.539 (bs, 1H), 7.430-7.412 (d, 1H), 7.214-7.193 (d, 1H), 4.980-4.934 (m, 1H), 4.146-3.991 (m, 2H), 3.549-3.516 (m, 2H), 3.342 (s, 3H), 3.048-2.941 (m, 1H), 2.843-2.724 (m, 2H), 2.165-2.083 (m, 1H). MS(m/e): 333.19 (M+H.sup.+).
Example 24
Preparation of 4-methyl-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindolin-1,3-di- one
[0370] Using the preparation process in Example 4, 3-methylphthalic anhydride and 1-(2-methoxyethyl)-3-amino-2,6-piperidinedione acetate were reacted to give the title compound as a white solid (2.348 g) (HPLC purity: 98.36%). Yield: 68.1%.
[0371] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.716-7.697 (d, 1H), 7.623-7.586 (t, 1H), 7.508-7.489 (d, 1H), 5.017-4.972 (m, 1H), 4.143-3.996 (m, 2H), 3.547-3.516 (t, 2H), 3.344 (s, 3H), 3.006-2.944 (m, 1H), 2.855-2.732 (m, 2H), 2.698 (s, 3H), 2.153-2.084 (m, 1H). MS(m/e): 353.23 (M+Na.sup.+).
Example 25
Preparation of 4-amino-5-methoxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione
[0372] To a reaction flask were added 4-amino-5-hydroxy-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)-isoindol- in-1,3-dione (2.5 g), methyl iodide (1.02 g), potassium carbonate (1.99 g) and N,N-dimethylformamide (25 mL). The solution was stirred at the room temperature over 16 hours until the reaction was complete. The solution was added into water at the room temperature. The resultant solution was stirred and filtered. The filter cake was collected as crude product. The crude product was dissolved and purified with methyl tert-butyl ether/ethyl acetate to give the title compound as a yellow solid (1.94 g) (HPLC purity: 96.08%). Yield: 74.6%.
[0373] .sup.1HNMR(CDCl.sub.3, 400 MHz) .delta. 7.180-7.160 (d, 1H), 6.891-6.871 (d, 1H), 5.388 (bs, 2H), 4.954-4.909 (m, 1H), 4.136-3.987 (m, 2H), 3.943 (s, 3H), 3.541-3.511 (t, 2H), 3.340 (s, 3H), 2.983-2.944 (m, 1H), 2.793-2.744 (m, 2H), 2.130-2.035 (m, 1H). MS(m/e): 362.22 (M+H.sup.+).
Example 26
Preparation of 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne
[0374] To a three-neck flask (100 mL) were added 3-nitrophthalic anhydride (5 g, 25.9 mmol), L-glutamine (3.782 g, 25.9 mmol), and acetonitrile (45 mL). The reaction solution was magnetically stirred under argon atmosphere. The reaction solution was refluxed at 90.degree. C. over 14 hours. After the reaction was complete, the solution was cooled. To the solution was added N,N'-carbonyldiiazole (12.591 g, 77.7 mmol) at the room temperature. The solution was stirred at the room temperature until no bubble was released. The resultant solution was reacted over 1 hour in an oil bath of 84.degree. C. The solution was cooled to the room temperature. The solution was added into hydrochloric acid (225 mL, 1 mol/L) with vigorously stirring. The resultant solution was stirred and filtered. The filter cake was dissolved in absolute ethanol (25 mL). The solution was filtered. The resultant solid was dried in vacuo to give 2-(2,6-dioxopiperidin-3-yl)-4-nitroisoindolin-1,3-dione (5.125 g) as a yellow product. Yield: 65.3%.
[0375] To a single-neck flask (100 mL) were added 2-(2,6-dioxopiperidin-3-yl)-4-nitroisoindolin-1,3-dione (3 g, 9.9 mmol) and N,N-dimethylformamide (45 mL). Sodium hydride (content: 60%, 0.872 g, 21.8 mmol) was added in one portion. The solution was stirred at the room temperature over 15 minutes. To the solution was slowly added dropwise 3-bromopropyl methyl ether (1.8 g, 12 mmol) in N,N-dimethylformamide (4 mL). After addition, the reaction continued over 2.5 hours. The reaction solution was poured into saturated aqueous solution of ammonium chloride (400 mL). The resultant solution was extracted with ethyl acetate (200 mL.times.3). The organic phases were combined. The combined organic phase was dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated. The residue was separated with column chromatography to give 4-nitro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,- 3-dione (1.211 g). Yield: 32.3%.
[0376] To a single-neck flask were added 4-nitro-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne (1.875 g, 5 mmol), palladium on carbon (10%, 0.4 g), methanol (10 mL), tetrahydrofuran (10 mL). The air was replaced with hydrogen. The solution was stirred at the room temperature and under atmospheric pressure overnight. After the reaction was complete, the solution was filtered with diatomite. The filtrate was concentrated. The residue was separated with column chromatography to give the title compound 4-amino-2-(1-(2-methoxypropyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dio- ne (1.135 g). HPLC purity: 96.51%. Yield: 65.8%.
[0377] MS(m/e): 346 (M+H.sup.+)
Example 27
Preparation of 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e
[0378] The Chinese patent No. ZL200510013292.3 discloses the preparation process of 4-amino-2-(1-(2-methoxyethyl)-2,6-dioxopiperidin-3-yl)isoindolin-1,3-dion- e, which is incorporated herein by reference in its entirety.
[0379] Alternatively, in accordance with the preparation process in Example 27, 3-bromopropyl methyl ether was replaced with 3-bromoethyl methyl ether to prepare the title compound.
[0380] .sup.1HNMR(CDCl.sub.3, 600 MHz) .delta. 7.38 (t, 1H), 7.11 (d, 1H), 6.85 (d, 1H), 5.30 (bs, 2H), 4.95-4.98 (m, 1H), 4.09-4.13 (m, 1H), 4.00-4.04 (m, 1H), 3.52-3.54 (m, 2H), 3.34 (s, 3H), 2.96-2.99 (m, 1H), 2.76-2.75 (m, 1H), 2.78-2.80 (m, 1H), 2.08-2.11 (m, 1H). MS(m/e): 332 (M+H+).
Biological Example 1
Effects of Piperidine-2,6-Dione Derivatives on Treatment of DNBS-Induced Inflammatory Bowel Disease Models in Rats
Experimental Animals:
[0381] Wistar rats, male, body weight of 100-120 g.
Methods:
[0382] 90 male Wistar rats were fasted for 40 hours. A 5% glucose injection was administered subcutaneously during fasting. 60 rats with normal conditions and moderate body weight were selected from the fasting rats. The animals were randomly divided into 6 groups according to the body weight: normal control group, model control group, sulfasalazine group (300 mg/Kg), and the compound in Example 27 groups consisting of 5, 15 and 30 mg/Kg dose groups. There were 10 rats in each group. The animals were anesthetized with ether. A specially made gavage device (made by a rat gavage device having the diameter of 2 mm, which was nested on a mouse gavage device having the diameter of 1 mm) was slowly inserted into the enteric cavity at 8 cm from the anus. The model control group, the sulfasalazine group and the compound in Example 27 groups were administered with 0.5 mL of 50 mg/mL DNBS in 30% ethanol solution. The normal control group was administered with 0.5 mL of 30% ethanol solution. The animals were inverted in anesthesia for 15 minutes after the gavage device was removed. The day when the model was prepared was the first day of the experiment. After 4 hours of DNBS induction, the corresponding drug or the corresponding volume of 1% CMC-Na was administered once per day. The body weights were recorded and the stool characteristics were scored. On the sixth day of the experiment colon tissues were taken from the animals to observe the results of each index.
[0383] The compound in Example 27 and sulfasalazine: Preparation Method: Suspended with 1% CMC-Na, and prepared when it was needed.
TABLE-US-00004 TABLE 3 Grouped Table DNBS Clysis Administration DNBS Concentration Volume Administration Dose Administration Groups Solvent (mg/mL) (mL/rat) Route (mg/Kg) Regimen Normal Control 30% 0 0.5 i.g 0 qd .times. 6 Group ethanol Model Control 30% 50 0.5 i.g 0 qd .times. 6 Group ethanol Sulfasalazine 30% 50 0.5 i.g 300 qd .times. 6 Group ethanol Compound in 30% 50 0.5 i.g 5 qd .times. 6 Example 27 ethanol Group 30% 50 0.5 i.g 15 qd .times. 6 ethanol 30% 50 0.5 i.g 30 qd .times. 6 ethanol Note: i.g: intragastric administration; qd .times. 6: once per day, a total of 6 times.
[0384] Animals were euthanized on the sixth day of the experiment to observe the results of each index. After the animals were sacrificed, colons were obtained. The adhesion degree of colon was observed and the length of the colon was measured. The ulcer area was calculated after removing the contents in the colon and the weight of the colon tissue was weighed. The colon tissue was longitudinally divided into two parts. One part of the colon tissue was fixed in 10% formalin solution for later use. The other part of the colon tissue was put into the liquid nitrogen to quickly frozen, and then was reserved at -80.degree. C. for the detection of MPO.
TABLE-US-00005 TABLE 4 Criteria for Adhesion Degree of Colon in Rats Adhesion Degree Point None 0 Mild 1 Severe 2
[0385] Reference: Videla S, Vilaseca J, Medina C, et al. Selective inhibition of phosphodiesterase-4 ameliorates chronic colitis and prevents intestinal fibrosis [J]. Journal of Pharmacology and Experimental Therapeutics, 2006, 316(2): 940-945.
[0386] Calculation method for ulcer area: length of ulcer point (cm).times.width of ulcer point (cm).
[0387] The frozen tissue was ground into powder in liquid nitrogen. An appropriate amount of the powder was weighed and was added into 50 mM potassium phosphate solution (containing 0.5% cetyltrimethylammonium bromide) to give a 25 mg/mL solution. The resultant solution was homogenized by an electric homogenizer. 1 mL of suspension was centrifuged to give a supernatant. 7 .mu.L of the supernatant was added into a 96-well plate and 200 .mu.L of o-dianisidine mixture (containing 0.167 mg/mL o-dianisidine and 0.0006% H.sub.2O.sub.2 potassium phosphate buffer). OD values of the resultant solution at 0, 30, 60, and 90 s were detected at 450 nm of the microplate reader.
Statistical Analysis of Data:
[0388] Test for homogeneity of variance was performed with Spss13.0. Where variance of data was homogeneous (P>0.05), one-way ANOVA LSD test was performed. Where the analysis of variance was significant (P.ltoreq.0.05), then Dunnett's multiple comparison (parameter method) or Kruskal-Wallis non-parametric test was performed. Where the results of Kruskal-Wallis non-parametric test were significant (P.ltoreq.0.05), the Mann-Whitney U test was performed for pairwise comparison.
Experimental Results:
TABLE-US-00006 [0389] TABLE 5 Results of Animal Colon Indexes in Each Group (Mean .+-. SD) Adhesion Degree Colon Weight Groups (Point) (mg) Normal Control Group 0.0 .+-. 0.0 1454.3 .+-. 177.9 Model Control Group 0.7 .+-. 0.8 2026.1 .+-. 781.0 Sulfasalazine Group- 0.2 .+-. 0.4 1794.9 .+-. 242.3 300 mg/Kg Compound in Example 0.4 .+-. 0.7 1775.7 .+-. 321.0 27 Group-5 mg/Kg Compound in Example 0.1 .+-. 0.3 1668.7 .+-. 245.3 27 Group-15 mg/Kg Compound in Example 0.1 .+-. 0.3 1534.7 .+-. 156.2 27 Group-30 mg/Kg Note: * compared with normal control group, P < 0.01; #: compared with model control group, P < 0.05.
TABLE-US-00007 TABLE 6 Results of Animal Colon Indexes in Each Group (Mean .+-. SD) Colon weight to Colon MPO length ratio Ulcer Area Activity Groups (mg/cm) (cm.sup.2) (U/mg) Normal Control Group 90.0 .+-. 9.5 0.0 .+-. 0.0 1.7 .+-. 1.2 Model Control Group 145.4 .+-. 50.1 1.8 .+-. 1.9 33.4 .+-. 19.6* Sulfasalazine Group- 140.6 .+-. 27.9 0.7 .+-. 0.6 26.0 .+-. 14.8 300 mg/Kg Compound in Example 144.2 .+-. 30.5 0.8 .+-. 0.9 20.3 .+-. 19.8 27 Group-5 mg/Kg Compound in Example 127.1 .+-. 13.7 0.7 .+-. 0.9 16.7 .+-. 9.8# 27 Group-15 mg/Kg Compound in Example 117.7 .+-. 19.4 0.3 .+-. 0.5 15.1 .+-. 13.0# 27 Group-30 mg/Kg Note: *compared with normal control group, P < 0.05; #compared with model control group, P < 0.05.
[0390] Results of colonic tissue-related indexes showed that compared with the normal control group, the colon of the model control group was significantly shortened, the colon tissue was abnormally proliferated and the colon weight was increased. At the same time, the MPO activity of colon tissue was significantly increased, the colon had a larger area of ulcer injury. Sulfasalazine as positive drug at the dose of 300 mg/Kg reduced the colon abnormal proliferation and reduced the colon weight, and had some improvement on colonic ulcers with the reduction of the ulcer area by 60.degree. %. The compound in Example 27 at the dose of 5 mg/Kg, 15 mg/Kg and 30 mg/Kg reduced the adhesion degree of colons, reduced the weight of colon tissue and the ulcer degree, and reduced the MPO activity of colon tissue, and thus showed a certain dose effect and a good therapeutic effect without the weight loss of animals. The compound in Example 27 at the dose of 30 mg/Kg reduced the ulcer degree of colon with a reduction of the ulcer area by about 80%, while the MPO activity of colon tissue was significantly reduced (P<0.05), of which the therapeutic effects are better than those of sulfasalazine as positive drug at the dose of 300 mg/Kg.
Biological Example 2
Effects of Piperidine-2,6-Dione Derivatives on Treatment of DNBS-Induced Inflammatory Bowel Disease Models in Rats
Experimental Grouping and Modeling:
[0391] 110 male Wistar rats were fasted for 40 hours. A 5% glucose injection (10 mL/Kg) was administered subcutaneously during fasting. 48 rats with normal conditions and moderate body weight were selected from the fasting rats. The animals were randomly divided into 5 groups according to the body weight: normal control group, model control group, sulfasalazine group (300 mg/Kg), dexamethasone group (0.1 mg/Kg), the compound in Example 26 group (30 mg/kg) and the compound in Example 27 group (30 mg/kg), with 8 rats in each group. The animals were anesthetized with isoflurane. A specially made gavage device (made by a rat gavage device having the diameter of 2 mm, which was nested on a mouse gavage device having the diameter of 1 mm) was slowly inserted into the enteric cavity at 8 cm from the anus. The model control group, the compound in Example 26 group and the compound in Example 27 group were administered with 0.5 mL of 50 mg/mL DNBS in 30% ethanol solution. The normal control group was administered with 0.5 mL of 30% ethanol solution. The animals were inverted in anesthesia for 15 minutes after the gavage device was removed. The day when the model was prepared was the first day of the experiment. After 4 hours of DNBS induction, the corresponding drug or the corresponding volume of 1% CMC-Na was administered once per day. On the sixth day of the experiment, the animals were euthanized to observe the results of each index so as to score the colon adhesion degree, measure the length of colon and ulcer area, weigh colon and calculate the colon weight to length ratio.
[0392] The compound in Example 26 and Example 27, sulfasalazine and dexamethasone: Preparation Method: Suspended with 1% CMC-Na, and prepared when it was needed.
TABLE-US-00008 TABLE 7 Grouped Table DNBS Clysis Administration Number Concentration Volume Administration Dose Administration of Groups (mg/mL) (mL/rat) Route (mg/Kg) Regimen Animals Normal Control 0 0.5 i.g 0 qd .times. 6 8 Group Model Control 50 0.5 i.g 0 qd .times. 6 8 Group Sulfasalazine 50 0.5 i.g 300 qd .times. 6 8 Group Dexamethasone 50 0.5 i.g 0.1 qd .times. 6 8 Group Compound in 50 0.5 i.g 30 qd .times. 6 8 Example 27 Group Compound in 50 0.5 i.g 30 qd .times. 6 8 Example 26 Group Note: qd .times. 6: once per day, a total of 6 times.
Statistical Analysis of Data:
[0393] Levene's Test for homogeneity of data was performed with Spss. Where the data were homogeneous (P>0.05), one-way ANOVA LSD test was performed. Where the analysis of variance was significant (P.ltoreq.0.05), Dunnett's multiple comparison (parameter method) was performed. Where the results of Levene's Test were significant (P.ltoreq.0.05), Kruskal-Wallis nonparametric test was performed. Where the results of Kruskal-Wallis nonparametric test were significant (P.ltoreq.0.05), Mann-Whitney U test was performed for pairwise comparison.
Experimental Results:
TABLE-US-00009 [0394] TABLE 8 Results of Animal Colon Indexes in Each Group (Mean .+-. SD) Colon Adhesion Length Colon Weight Degree Ulcer Area Groups (cm) (mg) (Points) (cm.sup.2) Normal Control Group 15.1 .+-. 0.9 1406.3 .+-. 83.7 0.0 .+-. 0.0 0.0 .+-. 0.0 Model Control Group 12.6 .+-. 1.3** 1863.3 .+-. 406.0* 0.9 .+-. 0.6 3.3 .+-. 1.4** Sulfasalazine Group 13.2 .+-. 2.5 1776.1 .+-. 280.7 0.8 .+-. 0.7 1.6 .+-. 1.2# Dexamethasone Group 14.2 .+-. 0.9 1496.0 .+-. 450.0# 0.3 .+-. 0.5 1.4 .+-. 1.5# Compound in Example 13.7 .+-. 1.4 1628.3 .+-. 185.5 0.3 .+-. 0.5 1.1 .+-. 0.9## 26 Group Compound in Example 12.9 .+-. 2.3 1737.9 .+-. 410.6 0.9 .+-. 0.8 2.0 .+-. 1.9 27 Group Note: *compared with normal control group, P < 0.05; **compared with normal control group, P < 0.01; #compared with model control group, P < 0.05, ##compared with model control group, P < 0.01.
[0395] The results of this experiment showed that after the animals were induced by DNBS in ethanol solution, the growth rate of the body weight was lower than that of the normal control group. On the fourth day of the experiment, the growth rate of the body weight of the animals in the sulfasalazine group and the compound in Example 26 group and the compound in Example 27 group was higher than that in the model control group, while the growth of the body weight of the animals in the dexamethasone group were slower, which indirectly implied the improvement effects of the drugs on the colonic injury.
[0396] The results of fecal characteristics showed that diarrhea symptoms were observed in all groups of DNBS-induced animals. On the fifth day of experiment, symptoms of diarrhea and liquid stools of animals in each administration group were slightly improved compared with the model control group.
[0397] Colon indexes showed that the area of colonic ulcer and the weight of colon of model control group were significantly higher than those of normal control group (P<0.05), and the colon was significantly shortened. Compared with model control group, there was significant reduction in the area of colonic ulcer in each administration group (P<0.05).
[0398] In view of the above results, the compound in Example 26 and the compound in Example 27 have good therapeutic effects on the improved model.
Biological Example 3
Effects of Piperidine-2,6-Dione Derivatives on Treatment of DNBS-Induced Inflammatory Bowel Disease Models in Rats
Experimental Grouping and Modeling:
[0399] 102 male Wistar rats were fasted for 40 hours. A 5% glucose injection (10 mL/Kg) was administered subcutaneously during fasting. 72 rats with normal conditions and moderate body weight were selected from the fasting rats. The animals were randomly divided into 9 groups according to the body weight (see Table 9. Grouped Table).
[0400] The animals were anesthetized with ether after 40 hours fasting. A specially made gavage device was slowly inserted into the enteric cavity at 8 cm from the anus. The model control group and each test group were administered with 0.5 mL of 50 mg/mL DNBS in 30% ethanol solution. The normal control group was administered with 0.5 mL of 30% ethanol solution. The animals were inverted in anesthesia for 15 minutes. The day when the model was prepared was day 0 of the experiment. After 4 hours of DNBS induction, the corresponding drug or the corresponding volume of 1% CMC-Na was administered once per day. On day 7 of the experiment, the animals were euthanized to score the colon adhesion degree, measure the length of colon and ulcer area, weigh colon and calculate the colon weight to length ratio.
TABLE-US-00010 TABLE 9 Grouped Table DNBS Clysis Administration Number DNBS Concentration Volume Administration Dose Administration of Groups Solvent (mg/ml) (ml/rat) Route (mg/Kg) Regimen Animals Normal Control 30% 0 0.5 -- -- -- 8 Group ethanol Model Control 50 0.5 ig -- qd .times. 7 Group Example 27 30 Example 7 Example 4 Example 21 Example 22 Example 6 Example 16 Note: The compound in each Example was suspended with 1% CMC-Na to prepare a 3 mg/mL of homogenous suspension. The administration volume was 10 mL/Kg. qd .times. 7: once per day, a total of 7 times. i.g: intragastric administration.
[0401] One-way ANOVA analysis of the length of colon and colon weight per length of rats was performed by SPSS22. Statistical analyses between groups were performed. The adhesion degree was scored. Mann-Whitney U non-parametric test was performed on ulcer area. Statistical analyses between groups were performed.
TABLE-US-00011 TABLE 10 Criteria for Adhesion Degree of Colon in Rats Adhesion Degree Point None 0 Mild 1 Severe 2
Experimental Results:
TABLE-US-00012 [0402] TABLE 11 Table of Body Weights Changes of Experimental Rats over 7 Days (g, Mean .+-. SD) Day 0 Day 1 Day 2 Day 3 Day 4 Day 5 Day 6 Day 7 Blank Control 135.7 .+-. 4.1 150.4 .+-. 7.4 162.7 .+-. 12.0 168.1 .+-. 15.5 172.3 .+-. 12.8 180.5 .+-. 15.7 182.7 .+-. 12.9 197.0 .+-. 13.1 Group Model Control 139.9 .+-. 6.6 141.6 .+-. 10.4 140.8 .+-. 11.0 137.9 .+-. 6.6 143.8 .+-. 7.9 154.0 .+-. 4.6 158.3 .+-. 7.7 168.2 .+-. 14.3 Group Example 27 135.4 .+-. 4.9 135.2 .+-. 4.2 136.7 .+-. 5.0 141.7 .+-. 7.5 150.1 .+-. 9.2 159.1 .+-. 10.3 165.2 .+-. 10.0 175.8 .+-. 8.2 Example 7 136.1 .+-. 2.5 136.7 .+-. 3.7 137.0 .+-. 8.7 141.6 .+-. 11.9 148.2 .+-. 12.7 159.2 .+-. 13.2 168.0 .+-. 15.7 173.5 .+-. 18.5 Example 4 137.1 .+-. 6.2 136.2 .+-. 4.1 138.2 .+-. 6.9 141.1 .+-. 8.8 151.8 .+-. 13.4 162.5 .+-. 13.0 170.8 .+-. 13.0 181.0 .+-. 12.2 Example 21 135.6 .+-. 6.9 134.4 .+-. 6.8 133.9 .+-. 6.8 138.7 .+-. 6.8 146.4 .+-. 6.9 157.9 .+-. 8.9 168.3 .+-. 4.6 173.5 .+-. 7.4 Example 22 137.5 .+-. 7.2 137.0 .+-. 9.0 139.7 .+-. 11.2 144.1 .+-. 12.7 152.6 .+-. 20.0 162.7 .+-. 19.0 170.5 .+-. 17.7 178.3 .+-. 20.7 Example 6 137.5 .+-. 5.6 138.1 .+-. 8.0 141.8 .+-. 13.1 147.4 .+-. 16.5 154.5 .+-. 17.7 164.2 .+-. 16.2 169.3 .+-. 18.4 180.9 .+-. 15.5 Example 16 137.3 .+-. 1.7 135.9 .+-. 3.7 137.6 .+-. 7.0 145.0 .+-. 9.2 155.2 .+-. 10.9 166.8 .+-. 10.2 171.5 .+-. 15.1 184.3 .+-. 13.8
TABLE-US-00013 TABLE 12 Colonic Data of Rats in Each Group Inhibitory Adhesion Inhibitory rate of weight Inhibitory Degree of rate of colon Colon weight increase Ulcer area rate of ulcer Colon adhesion to length ratio per length of colon area of colon (unit: point) (%) (unit: mg/cm) (%) (unit: cm.sup.2) (%) Blank Control 0.0 .+-. 0.0 76 .+-. 13 0.00 .+-. 0.00 Group Model Control 1.7 .+-. 0.5*** 155 .+-. 56*** 3.09 .+-. 2.23*** Group Example 27 0.9 .+-. 0.4# 47.1# 109 .+-. 12## 58.2## 0.01 .+-. 0.02## 99.7## Example 22 0.8 .+-. 0.9# 52.9# 112 .+-. 40# 54.4# 1.84 .+-. 2.59 40.5 Example 21 0.9 .+-. 0.7# 47.1# 104 .+-. 17## 64.6## 0.93 .+-. 1.52 70.0 Example 16 1.1 .+-. 0.8 35.3 114 .+-. 23# 51.9# 0.84 .+-. 1.25# 72.8# Example 7 0.9 .+-. 0.8# 47.1# 127 .+-. 52 35.4 0.77 .+-. 1.05 75.1 Example 6 0.6 .+-. 0.5## 64.7# 99 .+-. 21## 70.9## 0.34 .+-. 0.54# 89.0# Example 4 0.6 .+-. 0.7## 64.7## 125 .+-. 34 38.0 0.58 .+-. 0.78# 81.2# Note: ***compared with blank control group, P < 0.001; #compared with model control group, P < 0.05; ##compared with model control group, 0.001 .ltoreq. P < 0.01.
[0403] Calculation Formula of Inhibitory rate of weight increase per length: (Colon weight to length ratio in model group-Colon weight to length ratio in administration group)/(Colon weight to length ratio in model group-Colon weight to length ratio in blank group)
[0404] The pathological changes of the colon (adhesion, hyperplasia and ulcer) in the animals can be effectively relieved by the treatment in each experimental group.
Biological Example 4
Effects of Piperidine-2,6-Dione Derivatives on Treatment of DNBS-Induced Inflammatory Bowel Disease Models in Rats
Experimental Grouping and Modeling:
[0405] 102 male Wistar rats were fasted for 72 hours. A 5% glucose injection was administered subcutaneously twice during fasting. 88 rats with normal conditions and moderate body weight were selected from the fasting rats. The animals were randomly divided into 11 groups according to the body weight (see Table 11. Grouped Table).
[0406] The animals were anesthetized with ether after 72 hours fasting. A specially made gavage device was slowly inserted into the enteric cavity at 8 cm from the anus. The model control group and each test group were administered with 0.5 mL of 50 mg/mL DNBS in 30% ethanol solution. The normal control group was administered with 0.5 mL of 30% ethanol solution. The animals were inverted in anesthesia for 15 minutes. The day when the model was prepared was day 0 of the experiment. After 4 hours of DNBS induction, the corresponding drug or the corresponding volume of 1% CMC-Na was administered once per day. On day 7 of the experiment, the animals were euthanized to score the colon adhesion degree, measure the length of colon and ulcer area, weigh colon and calculate the colon weight to length ratio.
TABLE-US-00014 TABLE 13 Grouped Table DNBS Clysis Administration Number DNBS Concentration Volume Administration Dose Administration of Groups Solvent (mg/ml) (ml/rat) Route (mg/Kg) Regimen Animals Normal Control 30% 0 0.5 -- -- -- 8 Group ethanol Model Control 50 0.5 i.g -- qd .times. 7 Group Example 27 30 Example 8 Example 14 Example 24 Example 5 Example 23 Example 10 Example 2 Example 12 Note: The drug in the experiment was suspended with 1% CMC-Na to prepare a 3 mg/mL of homogenous suspension. The administration volume was 10 mL/Kg. i.g: intragastric administration. qd .times. 7: once per day, a total of 7 times.
TABLE-US-00015 TABLE 14 Criteria for Adhesion Degree of Colon in Rats Adhesion Degree Point None 0 Mild 1 Severe 2
[0407] One-way ANOVA analysis of the length of colon and colon weight per length of rats was performed by SPSS22. Statistical analyses between groups were performed. The adhesion degree was scored. Mann-Whitney U non-parametric test was performed on ulcer area. Statistical analyses between groups were performed.
Experimental Results:
TABLE-US-00016 [0408] TABLE 15 Table of Body Weights Changes of Experimental Rats over 7 Days (g, Mean .+-. SD) Day 0 Day 1 Day 2 Day 3 Day 4 Day 5 Day 6 Day 7 Blank Control 117.4 .+-. 1.6 132.2 .+-. 3.7 143.1 .+-. 7.7 153.6 .+-. 7.7 160.7 .+-. 7.2 172.3 .+-. 5.4 170.3 .+-. 4.3 185.0 .+-. 5.6 Group Model Control 121.1 .+-. 4.9 125.7 .+-. 6.3 121.4 .+-. 11.6 123.0 .+-. 8.1 125.3 .+-. 9.4 129.2 .+-. 12.5 134.7 .+-. 9.7 153.9 .+-. 12.7 Group Example 27 121.7 .+-. 7.0 125.5 .+-. 8.4 124.4 .+-. 8.1 125.8 .+-. 8.2 133.1 .+-. 9.2 141.6 .+-. 12.0 146.7 .+-. 11.8 156.3 .+-. 11.0 Example 8 125.3 .+-. 5.4 128.6 .+-. 5.3 128.2 .+-. 9.2 131.6 .+-. 13.5 136.8 .+-. 17.4 149.1 .+-. 16.7 149.7 .+-. 16.8 164.1 .+-. 17.9 Example 14 119.3 .+-. 6.4 126.2 .+-. 8.7 125.1 .+-. 10.5 129.5 .+-. 17.8 136.5 .+-. 19.7 146.4 .+-. 19.4 147.6 .+-. 19.9 165.7 .+-. 20.8 Example 24 123.5 .+-. 4.8 129.1 .+-. 5.5 130.9 .+-. 10.4 133.2 .+-. 14.5 135.3 .+-. 13.6 144.9 .+-. 12.6 145.9 .+-. 13.3 165.6 .+-. 12.1 Example 5 123.4 .+-. 5.5 126.7 .+-. 8.9 125.0 .+-. 11.0 126.9 .+-. 14.0 133.9 .+-. 13.3 141.9 .+-. 14.9 146.9 .+-. 13.9 159.7 .+-. 14.3 Example 23 121.9 .+-. 7.0 126.6 .+-. 8.5 123.2 .+-. 9.0 123.9 .+-. 7.6 136.5 .+-. 11.8 148.1 .+-. 14.5 152.1 .+-. 15.9 166.8 .+-. 12.8 Example 10 119.7 .+-. 4.2 122.4 .+-. 6.7 122.1 .+-. 8.0 126.6 .+-. 9.8 136.9 .+-. 12.5 143.7 .+-. 18.2 144.9 .+-. 19.7 160.8 .+-. 25.7 Example 2 117.9 .+-. 4.6 122.1 .+-. 4.6 123.0 .+-. 8.4 127.1 .+-. 11.3 132.0 .+-. 12.4 143.9 .+-. 11.9 141.5 .+-. 11.5 162.8 .+-. 15.8 Example 12 123.7 .+-. 3.3 129.1 .+-. 5.1 128.7 .+-. 6.6 133.9 .+-. 9.8 141.8 .+-. 12.0 149.2 .+-. 11.0 149.5 .+-. 11.2 167.3 .+-. 11.3
TABLE-US-00017 TABLE 16 Colonic Data of Animals in Each Experimental Group Inhibitory Adhesion Inhibitory rate of weight Inhibitory Degree of rate of colon Colon weight increase Ulcer area rate of ulcer Colon adhesion to length ratio per length of colon area of colon (unit: point) (%) (unit: mg/cm) (%) (unit: cm.sup.2) (%) Blank Control 0.3 .+-. 0.5 77 .+-. 8 0 .+-. 0 Group Model Control 1.6 .+-. 0.5*** 162 .+-. 84*** 2.4 .+-. 3.2** Group Example 27 -- -- 106 .+-. 14 65.9 0.4 .+-. 0.6 83.3 Example 24 0.8 .+-. 0.7# 62.2# 99 .+-. 18 74.1 0.3 .+-. 0.3 87.5 Example 23 0.8 .+-. 0.7# 62.2# 105 .+-. 12 67.1 0.2 .+-. 0.3# 91.7# Example 14 0.8 .+-. 0.5# 62.2 102 .+-. 18 70.6 0.1 .+-. 0.4# 95.8# Example 12 0.8 .+-. 0.7# 62.2# 92 .+-. 16# 82.4# 0.4 .+-. 0.9 83.3 Example 10 0.5 .+-. 0.5## 81.1## 93 .+-. 12# 81.2# 0.2 .+-. 0.5# 91.7## Example 8 0.9 .+-. 0.6 52.7 106 .+-. 13 65.9 0.2 .+-. 0.4 91.7 Example 5 0.8 .+-. 0.5# 62.2# 99 .+-. 14 74.1 0.2 .+-. 0.5 91.7 Example 2 0.8 .+-. 0.9 66.9# 120 .+-. 25 57.6 1.0 .+-. 1.6 83.3 Note: ***compared with blank control group, P < 0.001; **compared with blank control group, 0.001 .ltoreq. P < 0.01; #compared with model control group, P < 0.05; ##compared with model control group, 0.001 .ltoreq. P < 0.01.
[0409] Calculation Formula of Inhibitory rate of weight increase per length: (Colon weight to length ratio in model group-Colon weight to length ratio in administration group)/(Colon weight to length ratio in model group-Colon weight to length ratio in blank group)
[0410] The pathological changes of the colon (adhesion, hyperplasia and ulcer) in the animals can be effectively relieved by the treatment in each experimental group.
Biological Example 5
Acute Toxicity Experiment of Single Oral Administration of Piperidine-2,6-Dione Derivatives in Mice
Experimental Process
[0411] The mice were fasted one day before the experiment. On the day of the experiment, 6 mice (ICR mice, body weight of 19-21 g, three females and three males) were selected in each experiment group. First of all, one female and one male were selected to orally give 1000 mg/Kg, and observed for 5-10 minutes. If there were no obvious side effects, the remaining 4 animals were orally given the same dose, and observed for 10-15 minutes. If no animal died, the animals were placed in a box to balancedly feed for 7 days. The state was observed and the body weight was recorded daily. The animal grouping plan and the initial dose of the drug were shown in the table below:
TABLE-US-00018 TABLE 17 Information Table of Animal Groups Administration Administration Drug Number Dose Volume Concentration of Schedule Groups mg/Kg ml/Kg mg/mL Solvent Animals Day 1 Blank -- 15 -- 1% CMC-Na Three Control females Example 1 1000 66.67 and three Example 8 males Example 3 Example 2 Example 4 Example 24 Day 2 Example 23 Example 10 Example 2 Example 11 Example 20 Example 17 Example 13 Example 12 Day 3 Example 19 Example 26 Example 6 Example 3 Example 9
Observation Indexes within 0-15 Minutes after Administration
[0412] Activity: restlessness, hyperactivity
[0413] Nervous system response: tremor, spasm, convulsion, ataxia, posture abnormality, etc.
[0414] Autonomic nerve: exophthalmos, salivation, tears, urination (hematuria), diarrhea, piloerection, breathing, etc.
Observation Indexes Over 7 Days
[0415] Body weight
[0416] State of Animal: lethargy, hyperactivity, excitement, hair loss, diarrhea, abnormal behavior, death, etc.
Experimental Results
TABLE-US-00019 [0417] TABLE 18 Summary Table of Toxic Response of Tested Animals in 7 Days Group Sex Summary of toxic response of tested animals in 7 Days Example 23 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 10 Male Decrease of activity was observed in all the animals within 30 minutes after administration but activity was recovered within this period. Abnormality was not observed in all the animals from day 2 to 7. Female Decrease of activity was observed in all the animals within 30 minutes after administration but activity was recovered within this period. Abnormality was not observed in all the animals from day 2 to 7. Example 2 Male Slight abnormality of activity was occasionally observed in two animals but activity was recovered within in 30 minutes. Abnormality was not observed in all the animals from day 2 to 7. Female Abnormality was not observed in 7 days of the test Example 11 Male Abnormality was not observed in 7 days of the test Decrease of activity was observed in one animal after administration but activity was recovered within 3 minutes. Female Abnormality was not observed in all the animals from day 2 to 7. Example 20 Male Decrease of activity was observed in one animal 11 minutes after administration but activity was recovered later. Abnormality was not observed in all the animals from day 2 to 7. Female Decrease of activity was observed in two animals in 10 minutes after administration but activity was recovered later. Abnormality was not observed in all the animals from day 2 to 7. Example 17 Male Motor dysfunction in two animals was observed 30 minutes after administration and function was recovered later. Abnormality was not observed in all the animals from day 2 to 7. Female Motor dysfunction in one animal was observed 10 minutes after administration and function was recovered later. Abnormality was not observed in all the animals from day 2 to 7. Example 13 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 12 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 19 Male Severe motor dysfunction in two animals was observed 30 minutes after administration and function was recovered in about 2 hours. Abnormality was not observed in all the animals from day 2 to 7. Female Severe motor dysfunction in three animals was observed 30 minutes after administration and function was recovered in about 2 hours. Abnormality was not observed in all the animals from day 2 to 7. Example 26 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 6 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 3 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Example 9 Male Abnormality was not observed in 7 days of the test Female Abnormality was not observed in 7 days of the test Note: The dosage is 1000 mg/Kg in Table 18 unless indicated otherwise. "n = 1" indicate the number of the tested animal in the group is one. The number of the tested animal is 3 where "n = 1" is not indicated in the group. Abnormality is not observed where the observation index is not mentioned.
TABLE-US-00020 TABLE 19 Table of Daily Body Weight of Tested Male Mice (g, Mean .+-. SD) Day 1 Day 2 Day 3 Day 4 Day 5 Day 6 Day 7 Blank Control 17.4 .+-. 1.4 23.1 .+-. 1.5 24.6 .+-. 2.0 25.9 .+-. 1.5 26.9 .+-. 0.7 27.9 .+-. 1.5 28.7 .+-. 1.7 Group Example 1 20.1 .+-. 1.4 23.8 .+-. 1.4 24.9 .+-. 1.0 26.3 .+-. 1.2 27.7 .+-. 1.3 29.2 .+-. 1.4 29.4 .+-. 1.4 Example 8 18.0 .+-. 1.3 23.1 .+-. 0.9 24.6 .+-. 1.0 25.6 .+-. 1.7 27.2 .+-. 2.2 28.2 .+-. 2.2 29.2 .+-. 2.6 Example 3 18.0 .+-. 0.2 22.9 .+-. 0.3 24.3 .+-. 0.9 25.2 .+-. 1.1 27.0 .+-. 1.3 28.2 .+-. 1.9 28.5 .+-. 2.7 Example 4 17.6 .+-. 0.2 22.2 .+-. 0.3 23.6 .+-. 0.6 24.7 .+-. 0.6 26.7 .+-. 0.4 27.6 .+-. 0.8 28.2 .+-. 0.1 Example 22 17.1 .+-. 0.3 22.9 .+-. 0.8 23.5 .+-. 0.9 25.2 .+-. 0.5 27.2 .+-. 1.9 27.1 .+-. 1.8 27.4 .+-. 1.9 Example 18 18.2 .+-. 0.4 23.0 .+-. 0.7 24.1 .+-. 0.4 25.6 .+-. 0.8 28.1 .+-. 1.1 29.0 .+-. 1.4 29.0 .+-. 1.0 Example 24 17.7 .+-. 0.2 22.3 .+-. 0.7 24.5 .+-. 1.0 25.2 .+-. 1.0 27.2 .+-. 1.3 28.1 .+-. 2.0 28.9 .+-. 1.9 Example 23 19.4 .+-. 0.8 24.6 .+-. 1.2 24.5 .+-. 0.6 24.5 .+-. 0.6 25.4 .+-. 1.2 28.1 .+-. 0.8 29.0 .+-. 0.8 Example 10 19.4 .+-. 0.3 24.4 .+-. 0.6 25.5 .+-. 0.9 25.5 .+-. 0.9 26.8 .+-. 0.9 29.2 .+-. 0.7 30.3 .+-. 0.7 Example 2 18.6 .+-. 0.5 24.1 .+-. 0.4 24.7 .+-. 0.2 24.7 .+-. 0.2 26.2 .+-. 0.6 28.8 .+-. 0.5 29.2 .+-. 0.8 Example 11 19.5 .+-. 0.6 23.7 .+-. 0.8 25.4 .+-. 0.4 25.4 .+-. 0.4 27.0 .+-. 0.5 29.0 .+-. 0.7 29.8 .+-. 1.0 Example 20 18.8 .+-. 1.0 24.0 .+-. 1.1 25.3 .+-. 1.2 25.3 .+-. 1.2 26.7 .+-. 1.2 29.0 .+-. 1.1 29.3 .+-. 0.7 Example 17 19.0 .+-. 0.7 24.0 .+-. 0.7 24.6 .+-. 0.8 24.6 .+-. 0.8 26.1 .+-. 0.7 28.1 .+-. 0.6 29.3 .+-. 1.0 Example 13 19.6 .+-. 0.7 23.1 .+-. 0.6 24.4 .+-. 0.9 24.4 .+-. 0.9 26.5 .+-. 0.5 28.3 .+-. 1.3 28.4 .+-. 0.8 Example 12 18.5 .+-. 0.2 24.3 .+-. 0.6 24.9 .+-. 0.1 24.9 .+-. 0.1 26.1 .+-. 0.7 27.8 .+-. 0.7 28.5 .+-. 0.7 Example 19 18.9 .+-. 1.2 20.7 .+-. 1.1 21.3 .+-. 0.6 25.0 .+-. 2.0 24.8 .+-. 1.9 26.5 .+-. 1.9 27.7 .+-. 2.2 Example 26 18.7 .+-. 0.8 23.4 .+-. 0.5 25.0 .+-. 0.6 26.6 .+-. 1.4 27.3 .+-. 1.3 28.3 .+-. 1.3 28.8 .+-. 1.4 Example 6 18.4 .+-. 0.6 21.5 .+-. 0.9 21.6 .+-. 1.3 25.5 .+-. 0.5 25.9 .+-. 0.9 26.9 .+-. 1.1 27.1 .+-. 1.1 Example 3 18.8 .+-. 0.7 23.3 .+-. 0.1 24.3 .+-. 0.1 25.6 .+-. 0.7 26.9 .+-. 0.8 27.9 .+-. 0.9 28.6 .+-. 0.4 Example 9 18.3 .+-. 0.7 23.6 .+-. 0.7 24.1 .+-. 1.0 25.9 .+-. 1.1 27.2 .+-. 0.5 26.9 .+-. 1.4 27.9 .+-. 1.5
TABLE-US-00021 TABLE 20 Table of Daily Body Weight of Tested Female Mice (g, Mean .+-. SD) Day 1 Day 2 Day 3 Day 4 Day 5 Day 6 Day 7 Blank Control 17.4 .+-. 1.4 22.2 .+-. 0.3 23.4 .+-. 0.7 23.0 .+-. 0.2 24.3 .+-. 1.6 24.7 .+-. 1.6 24.1 .+-. 1.1 Group Example 1 18.5 .+-. 0.1 22.8 .+-. 0.3 23.2 .+-. 0.7 24.0 .+-. 0.2 25.3 .+-. 1.6 25.7 .+-. 1.6 27.1 .+-. 1.1 Example 8 18.9 .+-. 0.9 22.9 .+-. 0.9 24.3 .+-. 0.7 24.3 .+-. 1.1 25.3 .+-. 1.4 25.8 .+-. 1.5 26.0 .+-. 2.5 Example 3 18.8 .+-. 0.4 22.1 .+-. 1.2 22.7 .+-. 0.8 23.0 .+-. 1.4 23.7 .+-. 1.9 24.1 .+-. 1.2 24.4 .+-. 1.8 Example 4 18.4 .+-. 0.4 21.8 .+-. 0.5 23.2 .+-. 0.6 23.5 .+-. 1.2 24.7 .+-. 0.9 24.2 .+-. 1.2 24.0 .+-. 0.8 Example 22 18.5 .+-. 0.6 21.8 .+-. 0.3 22.9 .+-. 0.4 24.0 .+-. 1.0 25.0 .+-. 0.4 25.3 .+-. 0.6 25.6 .+-. 0.2 Example 18 18.4 .+-. 0.7 22.5 .+-. 0.8 23.8 .+-. 1.1 24.8 .+-. 1.5 24.8 .+-. 0.6 25.4 .+-. 0.7 25.2 .+-. 1.1 Example 24 18.4 .+-. 0.7 22.4 .+-. 1.0 23.4 .+-. 1.0 23.6 .+-. 0.9 24.3 .+-. 0.6 24.7 .+-. 0.6 24.8 .+-. 1.5 Example 23 19.1 .+-. 0.6 22.4 .+-. 0.4 23.0 .+-. 0.1 23.0 .+-. 0.1 24.6 .+-. 0.4 24.8 .+-. 0.7 24.6 .+-. 0.4 Example 10 19.2 .+-. 0.5 21.9 .+-. 0.4 22.4 .+-. 0.2 22.4 .+-. 0.2 22.9 .+-. 0.6 24.1 .+-. 1.5 24.2 .+-. 1.8 Example 2 19.2 .+-. 0.5 22.7 .+-. 0.6 23.2 .+-. 0.5 23.2 .+-. 0.5 24.4 .+-. 0.2 24.6 .+-. 1.1 24.5 .+-. 1.9 Example 11 18.1 .+-. 0.2 21.7 .+-. 0.2 22.4 .+-. 0.1 22.4 .+-. 0.1 23.5 .+-. 0.4 23.2 .+-. 0.8 23.2 .+-. 0.9 Example 20 18.7 .+-. 0.4 21.6 .+-. 1.0 22.1 .+-. 1.0 22.1 .+-. 1.0 23.0 .+-. 1.4 23.3 .+-. 0.2 23.7 .+-. 0.8 Example 17 18.6 .+-. 1.0 21.9 .+-. 0.4 22.7 .+-. 0.4 22.7 .+-. 0.4 23.0 .+-. 0.4 23.1 .+-. 1.0 22.7 .+-. 1.2 Example 13 18.8 .+-. 1.1 21.3 .+-. 0.3 21.8 .+-. 0.4 21.8 .+-. 0.4 23.0 .+-. 0.5 23.2 .+-. 0.9 23.6 .+-. 0.9 Example 12 18.7 .+-. 0.7 21.8 .+-. 0.6 22.6 .+-. 0.1 22.6 .+-. 0.0 23.7 .+-. 0.6 25.3 .+-. 1.0 25.7 .+-. 1.6 Example 19 18.0 .+-. 0.6 20.7 .+-. 1.1 21.3 .+-. 0.6 22.8 .+-. 0.3 23.2 .+-. 0.7 23.3 .+-. 1.2 23.0 .+-. 1.7 Example 26 18.0 .+-. 0.7 21.9 .+-. 0.3 22.1 .+-. 0.5 22.4 .+-. 0.2 22.7 .+-. 0.3 22.6 .+-. 0.3 22.4 .+-. 0.2 Example 6 17.5 .+-. 0.9 21.5 .+-. 0.9 21.6 .+-. 1.3 22.0 .+-. 1.2 22.3 .+-. 1.9 22.4 .+-. 2.3 22.9 .+-. 2.0 Example 3 18.3 .+-. 0.4 21.7 .+-. 0.2 22.3 .+-. 0.2 22.6 .+-. 0.8 23.0 .+-. 1.4 22.6 .+-. 1.3 22.7 .+-. 0.4 Example 9 17.5 .+-. 0.6 22.1 .+-. 0.9 22.1 .+-. 0.7 22.8 .+-. 0.8 22.8 .+-. 1.0 22.7 .+-. 1.6 23.6 .+-. 1.7
[0418] The body weight of the animals in each group had a trend of increase with slight fluctuations. Each compound had no side effects on the change of the body weights of the mice at the dosage of 1000 mg/Kg.
[0419] All the patents, patent application publications, patent applications and non-patent literatures as cited in the present disclosure are incorporated herein by reference in their entirety.
[0420] It is to be understood that the foregoing description relates to exemplary embodiments of the present application and that modifications may be made without departing from the spirit and scope of the present application as set forth in the claims.
* * * * *













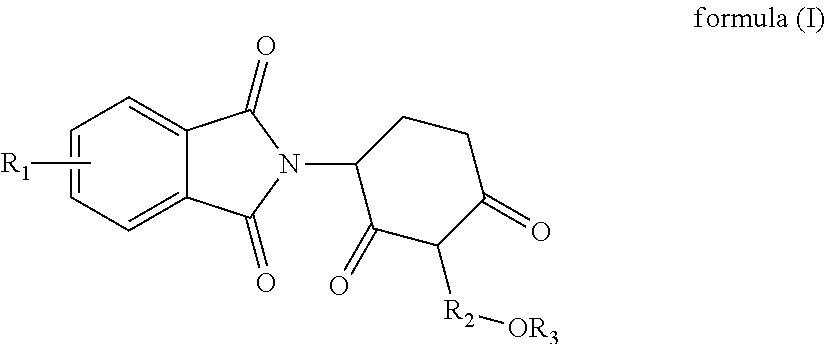





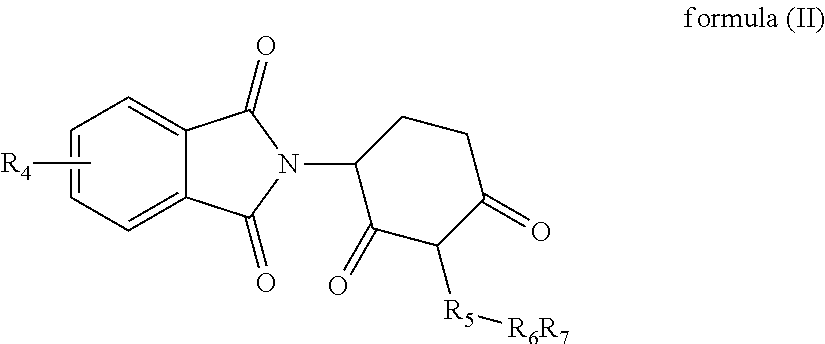


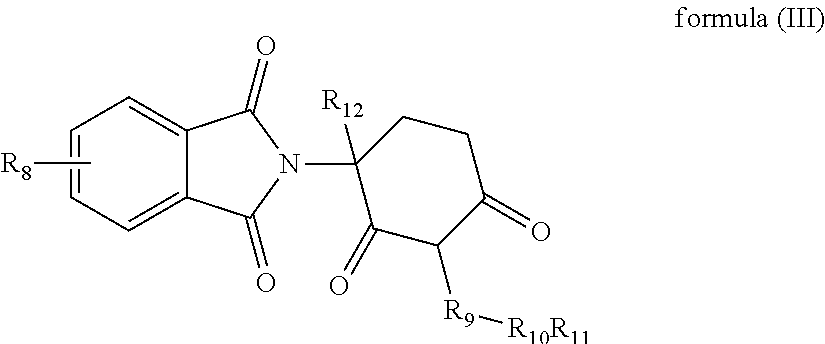

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.