Producing High-purity Chlorine Dioxide Gas
WANG; Shuangfei ; et al.
U.S. patent application number 16/182074 was filed with the patent office on 2019-11-14 for producing high-purity chlorine dioxide gas. This patent application is currently assigned to GuangXi University. The applicant listed for this patent is GuangXi University. Invention is credited to Chen LIANG, Xinliang LIU, Shuangxi NIE, Chengrong QIN, Xueping SONG, Shuangfei WANG, Zhiwei WANG, HongXiang ZHU.
| Application Number | 20190345033 16/182074 |
| Document ID | / |
| Family ID | 63204980 |
| Filed Date | 2019-11-14 |
| United States Patent Application | 20190345033 |
| Kind Code | A1 |
| WANG; Shuangfei ; et al. | November 14, 2019 |
PRODUCING HIGH-PURITY CHLORINE DIOXIDE GAS
Abstract
A high-purity chlorine dioxide gas may use hydrogen peroxide as a reducing agent and may use horizontal generator, evaporation crystallizer, dryer and other devices to produce chlorine dioxide gas (product) and sodium sulfate (by-product). Compared to the conventional chlorine dioxide preparation system, the chlorine dioxide reaction and the sodium sulfate crystallization are performed in two processes. These processes are relatively separate and independent, and continuously produce chlorine dioxide gas with high purity and low moisture content while the by-product salt cake is evaporated, crystallized, filtered and dried, thereby producing sodium sulfate, without generating solid and liquid waste.
| Inventors: | WANG; Shuangfei; (Nanning, CN) ; QIN; Chengrong; (Nanning, CN) ; NIE; Shuangxi; (Nanning, CN) ; SONG; Xueping; (Nanning, CN) ; LIANG; Chen; (Nanning, CN) ; LIU; Xinliang; (Nanning, CN) ; WANG; Zhiwei; (Nanning, CN) ; ZHU; HongXiang; (Nanning, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | GuangXi University |
||||||||||
| Family ID: | 63204980 | ||||||||||
| Appl. No.: | 16/182074 | ||||||||||
| Filed: | November 6, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16011684 | Jun 19, 2018 | 10266406 | ||
| 16182074 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B01D 9/0022 20130101; B01J 2219/182 20130101; B01D 2257/2025 20130101; B01J 7/02 20130101; C01B 11/026 20130101; B01D 2256/26 20130101; B01J 19/24 20130101; B01D 9/0059 20130101 |
| International Class: | C01B 11/02 20060101 C01B011/02; B01D 9/00 20060101 B01D009/00 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| May 8, 2018 | CN | 201804829036 |
Claims
1. The method for producing high-purity chlorine dioxide gas, comprising: using hydrogen peroxide as a reducing agent; and using a horizontal generator, an evaporation crystallizer, a dryer and other devices to produce a product comprising chlorine dioxide gas and a by-product comprising sodium sulfate, wherein chlorine dioxide reaction is produced by using the horizontal generator to produce chlorine dioxide, adding air into the horizontal generator, and stirring a reaction liquid and diluting the chlorine dioxide gas continuously to produce the chlorine dioxide gas with high purity and low moisture content, and sodium sulfate crystallization is produced by evaporating, crystallizing, filtering, and drying a salt cake in the evaporation crystallizer and dryer to produce sodium sulfate powder.
2. A method for producing high-purity chlorine dioxide gas in that hydrogen peroxide, sulfuric acid and sodium chlorate are used as raw materials to produce high-purity and low-moisture chlorine dioxide gas while salt cake is dried to produce sodium sulfate, the method comprising: entering sulfuric acid in a horizontal generator via a pipe; delivering a reducing agent through another pipe such that the reducing agent enters the horizontal generator after being mixed with sodium chlorate in a tertiary pipe; generating a chlorine dioxide gas when a REDOX reaction occurs; entering heat source into the horizontal generator by way of a pipe to step heat the reaction liquid, to force the temperature of the reaction liquid to increase from 20-30.degree. C. to 80-85.degree.; compressing the air by an air compressor such that the compressed air enters into the horizontal generator and stirring the reaction liquid to make the reaction uniform while releasing and diluting the chlorine dioxide gas; blowing air, by a blower, into the horizontal generator for further diluting the chlorine dioxide gas concentration to 4-9% (V/V); sending the diluted chlorine dioxide gas to use areas by an induced draft fan; while the raw material enters the horizontal generator from one end, discharging the reacted reaction liquid from another end of the horizontal generator and into the mother liquid discharge tank for storage; sending the discharged reaction liquid into the mother liquid lower circulating pipe by a crystallizer feed pump, and circulation, heating, evaporation and concentration between evaporation crystallizer and heater under a pumping action of a circulating pump, wherein the temperature in the evaporation crystallizer is controlled at 70-73.degree. C., the vacuum pressure is controlled at -79 to -81 KPa(g); concentrating and crystalizing sodium sulfate in the mother liquid and depositing the sodium sulfate at the bottom of the evaporation crystallizer, wherein the sodium sulfate is sent to a filtration device through a feed pump for filtration, washing and decrease water percent; introducing salt cake solid into a dryer to produce sodium sulfate with dryness up to 99.5%; returning a filtrate liquid to the mother liquid circulating pipe via the filtrate pipe for recycling, wherein neither solid nor liquid wastes are produced throughout the production process; sending the clarified mother liquid from the upper part of the evaporation crystallizer to the mother liquid tank by a mother liquid extraction pump for buffering; and sending clarified mother liquid to the horizontal generator by the mother liquid return pump.
Description
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application is a divisional of, and claim priority to, U.S. patent application Ser. No. 16/011,684, filed on Jun. 19, 2018, which claims the benefit of, and priority to, Chinese Patent Application No. 2018104829036, filed on May 8, 2018. The subject matter thereof is hereby incorporated herein by reference in its entirety.
FIELD
[0002] The present invention generally relates to chlorine dioxide preparation, and more particularly, to a process, system, and apparatus for producing high-purity chlorine dioxide gas.
BACKGROUND
[0003] Chlorine dioxide (ClO2) is a yellow orange gas at normal temperature and pressure and has an irritating and spicy taste similar to a mixture of chlorine and ozone. The boiling point is 11.degree. C., the freezing point is -59.degree. C., and the gaseous density is 3.09 g/m.sup.3 at 11.degree. C. Gaseous ClO.sub.2 is unstable. Exposure to light or organic matter at high concentration will decompose violently and production of oxygen and chlorine. Normally it must be produced at the mill near its point of application. It is more stable when diluted to lower than 12% volume content with air or steam at atmospheric temperature or in the state of a low-temperature water solution. ClO.sub.2 is a strong oxidizer and can be used as a bleaching agent for pulp and textiles, and as water treatment agent, new air purifying freshener, and a disinfection, sterilizing, deodorizing agent for food, epidemic prevention, hygiene, etc.
[0004] At present, the main methods of producing chlorine dioxide include methanol method and integrated method. Methanol method uses methanol, sulfuric acid, and sodium chlorate as raw materials, and the vertical generator is used to produce chlorine dioxide. The product of chlorine dioxide has a large moisture content up to 80% (w/w) or more, and contains a small amount of chlorine; The integrated method uses hydrochloric acid and sodium chlorate as raw materials and both vertical generator and horizontal generator can be used, but chlorine dioxide gas contains a lot of chlorine, and the molar ratio of chlorine dioxide to chlorine is 2:1. For the use areas with high purity requirements (such as sodium chlorite preparation, food and medicine area, etc.), the purity of chlorine dioxide produced by the above two preparation methods cannot meet the use requirements.
[0005] In addition, chlorine dioxide product produced by common industrial chlorine dioxide preparation device are pumped to the final use areas in the form of low-temperature chlorine dioxide solution, not supplied in the form of gas.
[0006] Accordingly, an improved process and apparatus for producing high-purity chlorine dioxide gas may be beneficial.
SUMMARY
[0007] Certain embodiments of the present invention may provide solutions to the problems and needs in the art that have not yet been fully identified, appreciated, or solved by conventional chlorine dioxide preparation technologies. For example, some embodiments pertain to a process and apparatus for producing high-purity chlorine dioxide gas.
BRIEF DESCRIPTION OF THE DRAWINGS
[0008] In order that the advantages of certain embodiments of the invention will be readily understood, a more particular description of the invention briefly described above will be rendered by reference to specific embodiments that are illustrated in the appended drawings. While it should be understood that these drawings depict only typical embodiments of the invention and are not therefore to be considered to be limiting of its scope, the invention will be described and explained with additional specificity and detail through the use of the accompanying drawings, in which:
[0009] FIG. 1 is a schematic diagram illustrating a high-purity chlorine dioxide gas production device, according to an embodiment of the present invention.
DETAILED DESCRIPTION OF THE EMBODIMENTS
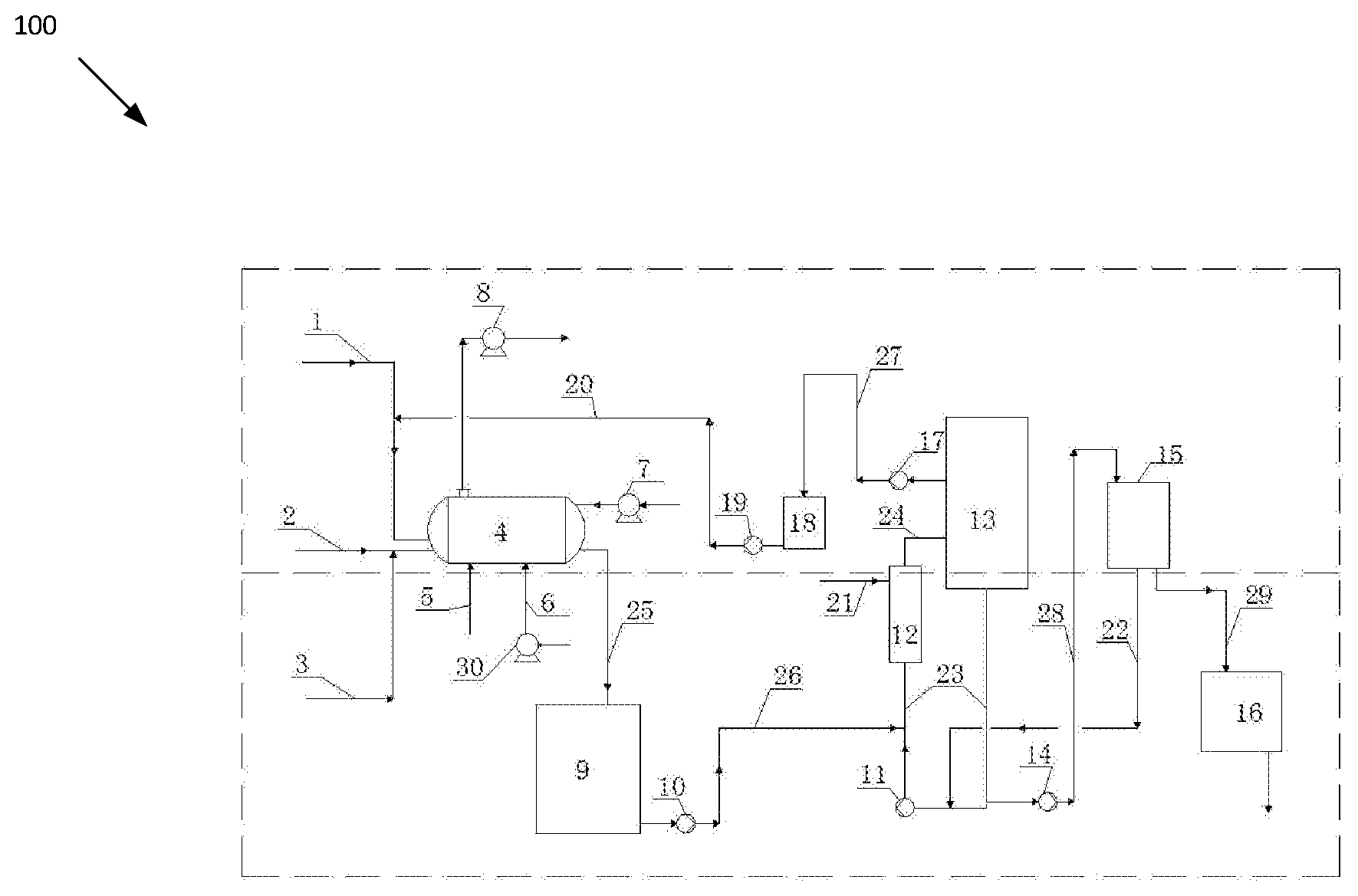
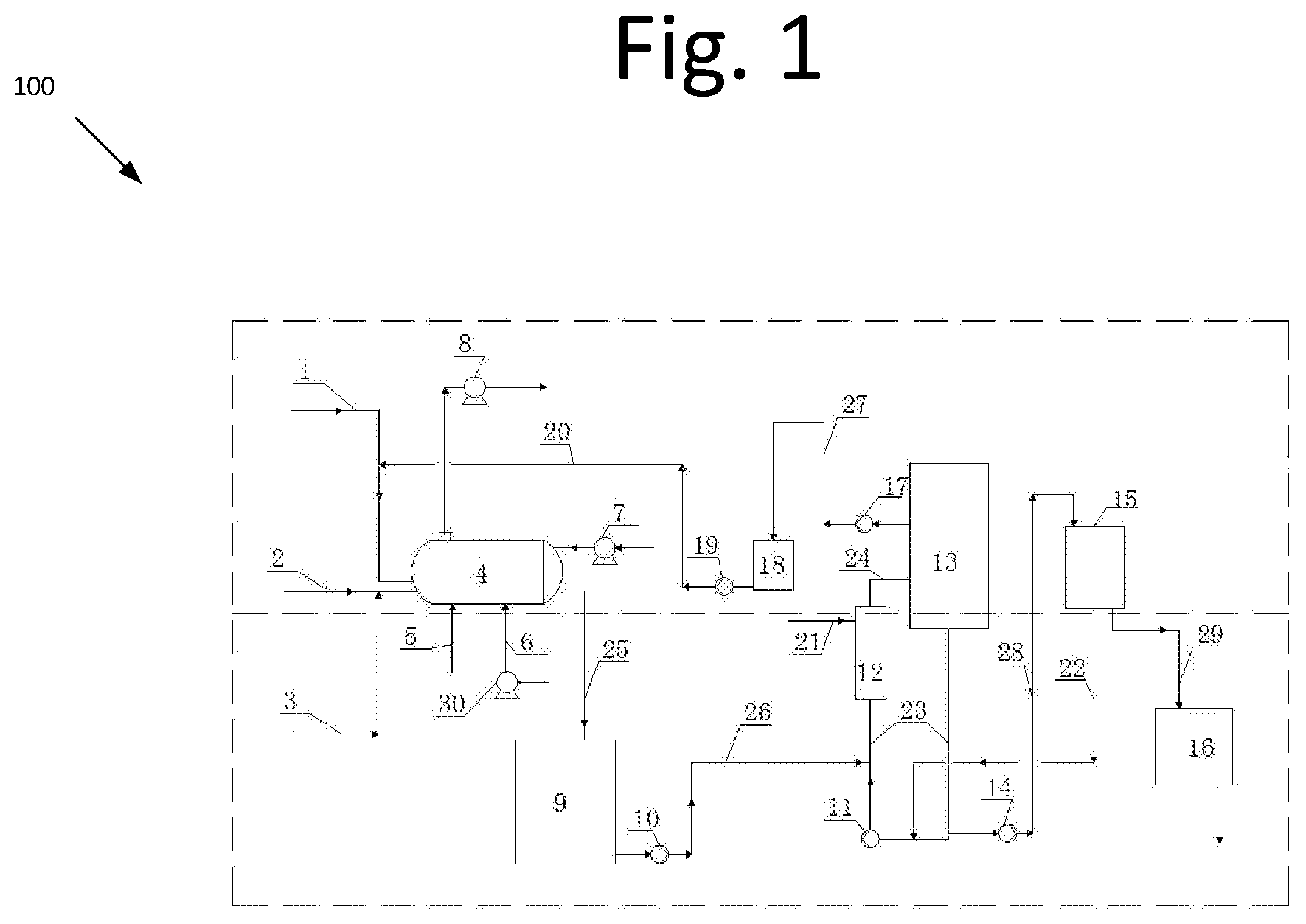
[0010] FIG. 1 is a schematic diagram illustrating a high-purity chlorine dioxide gas production device 100, according to an embodiment of the present invention. The devices for producing high-purity chlorine dioxide gas including a horizontal generator (4), blower (7), induced draft fan (8), mother liquid discharge tank (9), crystallizer feed pump (10), circulating pump (11), heater (12), evaporation crystallizer (13), filtration unit feed pump (14), filtration unit (15), dryer (16), mother liquid extraction pump (17), mother liquid tank (18), mother liquid return pump (19), air compressor (30). The sulfuric acid pipe (1) is connected with the horizontal generator (4); the hydrogen peroxide pipe (2) joins the sodium chlorate pipe (3) and are connected to the horizontal generator (4); the heat source pipe (5) is connected to the inlet of the horizontal generator (4); the compressed air pipe (6) is connected to the air compressor (30) and the horizontal generator (4); the horizontal generator (4) is connected to the mother liquid discharge tank (9) through a reaction liquid (mother liquid) discharge pipe (25); the mother liquid discharge tank (9) is connected with the mother liquid lower circulating pipe (23) through the inlet/outlet pipe (26) of the crystallizer feed pump; mother liquid lower circulating pipe (23) and mother liquid upper circulating pipe (24) are connected with the evaporation crystallizer (13), heater (12) and the circulating pump (11); the mother liquid tank (18) is connected to the evaporation crystallizer (13) and the horizontal generator (4) respectively through the inlet/outlet pipe of the mother liquid extraction pump (27) and the inlet/outlet pipe of the mother liquid return pump (20); the heat source pipe (21) is connected to the inlet pipe of the heater (12); the filtration unit (15) is connected to the mother liquid lower circulating pipe (23) through the inlet/outlet pipe of the feed pump (28) and the filtrate pipe (22) to form a circulation loop; the filtration unit (15) is connected to the dryer (16) via the salt cake discharge pipe (29).
[0011] In some embodiments, to make the chlorine dioxide preparation system run smoothly and continuously, a mother liquid discharge tank (9) with buffer effect and a mother liquid tank (18) are equipped between the horizontal generator (4) and the evaporation crystallizer (13). The chlorine dioxide reaction device and the evaporation crystallization device are relatively separated and independent.
[0012] In some embodiments, to make the reaction liquid of the horizontal generator (4) enter to the evaporation crystallizer (13), the reaction liquid discharge pipe (25) and inlet/out pipe (26) of the crystallizer feed pump are equipped before and after the mother liquid discharge tank (9). to respectively connect with the horizontal generator (4) and mother liquid lower circulating pipe (23). To make the supernatant of the evaporation crystallizer (13) return to the horizontal generator (4), the mother liquid return pump (19), extraction pump (17) and corresponding pipeline are equipped before and after the mother liquid tank (18) and are respectively connected to the evaporation crystallizer (13) and the horizontal generator (4).
[0013] In certain embodiments, to facilitate the circulation, evaporation and concentration of the mother liquid, the evaporation crystallizer (13), heater (12) and circulation pump (11) are connected in series by the mother liquid lower circulating pipe (23) and upper circulating pipe (24) to form a circulation loop.
[0014] In an embodiment, to make full use of the salt cake and filtrate and avoid waste generated, the filtration unit (15) is provided with filtrate pipe (22) and salt cake discharge pipe (29) to respectively connect with the mother liquid lower circulating pipe (23) and dryer (16) to form a filtrate liquid return channel and salt cake treatment channel.
[0015] The invention provides a method for producing high-purity chlorine dioxide gas by using hydrogen peroxide, sulfuric acid and sodium chlorate as raw materials to produce chlorine dioxide and drying the salt cake to produce salt cake powder. The specific steps are as follows:
[0016] Step 1, sulfuric acid enters the horizontal generator (4) through pipe (1); the reducing agent--hydrogen peroxide is transferred through pipe (2) and enters the horizontal generator (4) after being mixed with the sodium chlorate in the pipe (3), chlorine dioxide gas is generated after the redox reaction.
[0017] Step 2, while the raw material enters the horizontal generator (4) from one end, the reaction liquid after reacted is discharged from the other end into the mother liquid discharge tank (9) for storage, and then sent to the mother liquid into the lower circulating pipe (23) by the crystallizer feed pump (10). circulation, heating, evaporation and concentration between the evaporation crystallizer (13) and the heater (12) under the pumping action of the circulating pump (11); the salt cake in the mother liquid is concentrated and crystallized and is deposited at the bottom of the evaporation crystallizer (13) and the lower circulating pipe (23) and sent to the filtration unit (15) through the feed pump (14) for filtration, washing and concentration. Concentrated solid salt cake enters the dryer (16) to produce sodium sulfate powder with a dryness up to 99.5% (which can be sold as a byproduct); the filtrate liquid is returned to the mother liquid circulating pipe (23) via the filtrate liquid pipe (22).
[0018] Step 3, the clarified mother liquid in the upper of the evaporation crystallizer (13) is sent to the mother liquid tank (18) by the mother liquid extraction pump (17) for buffering, and then sent to the horizontal generator (4) by the mother liquid return pump (19).
[0019] In some embodiments, the heat source adds to the horizontal generator (4) through pipe (5) to heat the reaction solution indirectly, under the small negative pressure condition, the temperature of the reaction liquid is step increased from 20-30.degree. C. to 80-85.degree. C.; the air is compressed by the air compressor (30) and then enters the horizontal generator (4) and stirs the reaction liquid to make the reaction uniform while releasing and diluting the chlorine dioxide gas; in addition, the blower (7) blows the air into the horizontal generator (4) for further diluting the chlorine dioxide gas concentration to 4-9% (V/V); the diluted chlorine dioxide gas is sent out to the end use areas by the induced draft fan (8). The above-mentioned process can avoid the violent reaction during the preparation process and high concentration of chlorine dioxide, thus to avoid ClO.sub.2 decomposition, as well as avoid the salt cake crystallization in the horizontal generator (4) due to the high reaction temperature and excess water evaporation. The achieved chlorine dioxide gas has a moisture content of less than 8% (w/w) and is substantially free of chlorine.
[0020] In some embodiments, to increase the evaporation efficiency and save heat source, the temperature in the evaporation crystallizer is controlled at 70-73.degree. C. And the vacuum is controlled at -79 to -81 KPa (g).
[0021] The invention provides a method and devices for producing high-purity chlorine dioxide gas, which have the following advantages and effects:
[0022] The invention describes a device for producing high-purity chlorine dioxide gas, a mother liquid discharge tank (9) and a mother liquid tank (18) are equipped between the horizontal generator (4) and the evaporation crystallizer (13), make the chlorine dioxide reaction device and the evaporation crystallization device relatively separated and independent. Meanwhile, the mother liquid discharge tank (9) and the mother liquid tank (18) play a role in buffering, so that the chlorine dioxide preparation system can be continuously and smoothly running.
[0023] A method described in the invention, by which the chlorine dioxide gas is produced with high purity, is using the hydrogen peroxide, sulfuric acid and sodium chlorate as raw materials and adopting a horizontal generator, the reaction liquid is heated step by step from 25.degree. C. to 85.degree. C. under a small negative pressure condition, which can effectively avoid salt cake crystallization in the generator due to excessive evaporation of the reaction liquid and blocking the generator, as well as to avoid chlorine dioxide decomposition due to the violent reaction, and the prepared chlorine dioxide gas has a moisture content less than 8% (w/w) and essentially free of chlorine; the blower (7) blows the air into the horizontal generator (4) to dilute the chlorine dioxide gas concentration to 4-9% (V/V) to ensure the chlorine dioxide is stable and not decomposed easily. The by-product salt cake can be concentrated and crystallized by controlling the temperature in the evaporation crystallizer at 70-73.degree. C. and the vacuum degree at -79 to -81 KPa (g) to produce sodium sulfate powder after being filtered and dried. The filtrate liquid returns to the mother liquid lower circulating pipe (23) through pipe (22), no solid and liquid wastes produced.
[0024] This invention will be described in detail below with reference to the accompanying drawings and embodiments. The described embodiments are merely some but not all of the embodiments of this invention. All the other embodiments obtained by the technical person in this field based on the embodiments of this invention without creative efforts shall fall within the protection scope of the present invention.
EXAMPLE 1
[0025] As shown in FIG. 1, devices for producing high-purity chlorine dioxide gas include a horizontal generator (4), blower (7), induced draft fan (8), mother liquid discharge tank (9), crystallizer feed pump (10), circulating pump (11), heater (12), evaporation crystallizer (13), filtration unit feed pump (14), filtration unit (15), dryer (16), mother liquid extraction pump (17), mother liquid tank (18), mother liquid return pump (19), air compressor (30). The sulfuric acid pipe (1) is connected with the horizontal generator (4); the hydrogen peroxide pipe (2) joins the sodium chlorate pipe (3) and are connected to the horizontal generator (4); the heat source pipe (5) is connected to the inlet of the horizontal generator (4); the compressed air pipe (6) is connected to the air compressor (30) and the horizontal generator (4); the horizontal generator (4) is connected to the mother liquid discharge tank (9) through a reaction liquid (mother liquid) discharge pipe (25); the mother liquid discharge tank (9) is connected with the mother liquid lower circulating pipe (23) through the inlet/outlet pipe (26) of the crystallizer feed pump; mother liquid lower circulating pipe (23) and upper circulating pipe (24) are connected with the evaporation crystallizer (13), heater (12) and the circulating pump (11); the mother liquid tank (18) is connected to the evaporation crystallizer (13) and the horizontal generator (4) through the inlet/outlet pipe of the mother liquid extraction pump (27) and the inlet/outlet pipe of the mother liquid return pump (20); the heat source pipe (21) is connected to the inlet pipe of the heater (12); the filtration unit (15) is connected to the mother liquid lower circulating pipe (23) through the inlet/outlet pipe of the feed pump (28) and the filtrate pipe (22) to form a circulation loop; the filtration unit (15) is connected to the dryer (16) via the salt cake discharge pipe (29).
[0026] Step 1, sulfuric acid enters into the horizontal generator (4) through pipe (1); the reducing agent--hydrogen peroxide is transferred through pipe (2) and enters into the horizontal generator (4) after being mixed with the sodium chlorate in pipe (3), chlorine dioxide gas is generated after the redox reaction. The heat source enters the horizontal generator (4) through pipe (5) to gradually heat the reaction liquid, so that the temperature of the reaction liquid is increased from 20.degree. C. to 80.degree. C. The air is compressed by the air compressor (30) and then enters into the horizontal generator (4) and stirs the reaction liquid to make the reaction uniform while releasing and diluting the chlorine dioxide gas; in addition, the blower (7) blows the air into the horizontal generator (4) for further diluting the chlorine dioxide gas concentration to 4% (V/V); the diluted chlorine dioxide gas is sent out to the end use areas by the induced draft fan (8). The product has a moisture content of 7.8% (w/w) and is essentially free of chlorine.
[0027] Step 2, while the raw material enters into the horizontal generator (4) from one end, the reacted reaction liquid is discharged from the other end of the horizontal generator to the mother liquid discharge tank (9) for storage, and then sent to the mother liquid lower circulating pipe (23) by the crystallizer feed pump (10), and circulation, heating, evaporation and concentration between the evaporation crystallizer (13) and the heater (12) under the pumping action of the circulating pump (11); the temperature in the evaporation crystallizer is controlled at 73.degree. C., the vacuum degree is controlled at -79 KPa(g). The salt cake in the mother liquid is concentrated and crystallized and is deposited at the bottom of the evaporation crystallizer (13) and the lower circulating pipe (23) and sent to the filtration device (15) through the feed pump (14) for filtration, washing and concentration. The concentrated solid salt cake is introduced into the dryer (16) to produce sodium sulfate powder with dryness up to 99.6% (which can be sold as a byproduct); the filtrate liquid is returned to the mother liquid circulating pipe (23) via the filtrate pipe (22) for recycling. No solid and liquid wastes produced throughout the production process.
[0028] Step 3, the clarified mother liquid in the upper of the evaporation crystallizer (13) is sent to the mother liquid tank (18) by the mother liquid extraction pump (17) for buffering, and then sent to the horizontal generator (4) by the mother liquid return pump (19).
EXAMPLE 2
[0029] As shown in FIG. 1, devices for producing high-purity chlorine dioxide gas includes a horizontal generator (4), blower (7), induced draft fan (8), mother liquid discharge tank (9), crystallizer feed pump (10), circulating pump (11), heater (12), evaporation crystallizer (13), filtration unit feed pump (14), filtration unit (15), dryer (16), mother liquid extraction pump (17), mother liquid tank (18), mother liquid return pump (19), air compressor (30). The sulfuric acid pipe (1) is connected with the horizontal generator (4); the hydrogen peroxide pipe (2) joins the sodium chlorate pipe (3) and are connected to the horizontal generator (4); the heat source pipe (5) is connected to the inlet of the horizontal generator (4); the compressed air pipe (6) is connected to the air compressor (30) and the horizontal generator (4); the horizontal generator (4) is connected to the mother liquid discharge tank (9) through a reaction liquid (mother liquid) discharge pipe (25); the mother liquid discharge tank (9) is connected with the mother liquid lower circulating pipe (23) through the inlet/outlet pipe (26) of the crystallizer feed pump; mother liquid lower circulating pipe (23) and upper circulating pipe (24) are connected with the evaporation crystallizer (13), heater (12) and the circulating pump (11); the mother liquid tank (18) is connected to the evaporation crystallizer (13) and the horizontal generator (4) through the inlet/outlet pipe of the mother liquid extraction pump (27) and the inlet/outlet pipe of the mother liquid return pump (20); the heat source pipe (21) is connected to the inlet pipe of the heater (12); the filtration unit (15) is connected to the mother liquid lower circulating pipe (23) through the inlet/outlet pipe of the feed pump (28) and the filtrate pipe (22) to form a circulation loop; the filtration unit (15) is connected to the dryer (16) via the salt cake discharge pipe (29).
[0030] The method for preparing 8 t/d chlorine dioxide gas using the above mentioned device is as follows:
[0031] Step 1, sulfuric acid enters the horizontal generator (4) through pipe (1); the reducing agent--hydrogen peroxide is transferred through pipe (2) and enters the horizontal generator (4) after being mixed with the sodium chlorate in pipe (3), chlorine dioxide gas is generated after the redox reaction. The heat source enters into the horizontal generator (4) through pipe (5) to gradually heat the reaction liquid, so that the temperature of the reaction liquid is increased from 25.degree. C. to 83.degree. C. The air is compressed by the air compressor (30) and then enters the horizontal generator (4) and stirs the reaction liquid to make the reaction uniform while releasing and diluting the chlorine dioxide gas; in addition, the blower (7) blows the air into the horizontal generator (4) for further diluting the chlorine dioxide gas concentration to 6% (V/V); the diluted chlorine dioxide gas is sent out to the end use areas by the induced draft fan (8). The product has a moisture content of 7.8% (w/w) and is essentially free of chlorine.
[0032] Step 2, while the raw material enters into the horizontal generator (4) from one end, the reacted reaction liquid is discharged from the other end of the horizontal generator (4) to the mother liquid discharge tank (9) for storage, and then sent to the mother liquid lower circulating pipe (23) by the crystallizer feed pump (10), and circulation, heating, evaporation and concentration between the evaporation crystallizer (13) and the heater (12) under the pumping action of the circulating pump (11); the temperature in the evaporation crystallizer is controlled at 72.degree. C., the vacuum degree is controlled at -80 KPa(g). The salt cake in the mother liquid is concentrated and crystallized and is deposited at the bottom of the evaporation crystallizer (13) and the lower circulating pipe (23) and sent to the filtration device (15) through the feed pump (14) for filtration, washing and concentration. The concentrated solid salt cake is introduced into the dryer (16) to produce sodium sulfate powder with dryness up to 99.65% (which can be sold as a byproduct); the filtrate liquid is returned to the mother liquid circulating pipe (23) via the filtrate pipe (22) for recycling. No solid and liquid wastes produced throughout the production process.
[0033] Step 3, the clarified mother liquid in the upper of the evaporation crystallizer (13) is sent to the mother liquid tank (18) by the mother liquid extraction pump (17) for buffering, and then sent to the horizontal generator (4) by the mother liquid return pump (19).
EXAMPLE 3
[0034] As shown in FIG. 1, a device for producing high-purity chlorine dioxide gas includes a horizontal generator (4), blower (7), induced draft fan (8), mother liquid discharge tank (9), crystallizer feed pump (10), circulating pump (11), heater (12), evaporation crystallizer (13), filtration unit feed pump (14), filtration unit (15), dryer (16), mother liquid extraction pump (17), mother liquid tank (18), mother liquid return pump (19), air compressor (30). The sulfuric acid pipe (1) is connected with the horizontal generator (4); the hydrogen peroxide pipe (2) joins the sodium chlorate pipe (3) and are connected to the horizontal generator (4); the heat source pipe (5) is connected to the inlet of the horizontal generator (4); the compressed air pipe (6) is connected to the air compressor (30) and the horizontal generator (4); the horizontal generator (4) is connected to the mother liquid discharge tank (9) through a reaction liquid (mother liquid) discharge pipe (25); the mother liquid discharge tank (9) is connected with the mother liquid lower circulating pipe (23) through the inlet/outlet pipe (26) of the crystallizer feed pump; mother liquid lower circulating pipe (23) and upper circulating pipe (24) are connected with the evaporation crystallizer (13), heater (12) and the circulating pump (11); the mother liquid tank (18) is connected to the evaporation crystallizer (13) and the horizontal generator (4) through the inlet/outlet pipe of the mother liquid extraction pump (27) and the inlet/outlet pipe of the mother liquid return pump (20); the heat source pipe (21) is connected to the inlet pipe of the heater (12); the filtration unit (15) is connected to the mother liquid lower circulating pipe (23) through the inlet/outlet pipe of the feed pump (28) and the filtrate pipe (22) to form a circulation loop; the filtration unit (15) is connected to the dryer (16) via the salt cake discharge pipe (29).
[0035] The method for preparing 12 t/d chlorine dioxide gas using the above mentioned device is as follows:
[0036] Step 1, sulfuric acid enters into the horizontal generator (4) through pipe (1); the reducing agent--hydrogen peroxide is transferred through pipe (2) and enters into the horizontal generator (4) after being mixed with the sodium chlorate in pipe (3), chlorine dioxide gas is generated after the redox reaction. The heat source enters the horizontal generator (4) through pipe (5) to gradually heat the reaction liquid, so that the temperature of the reaction liquid is increased from 30.degree. C. to 85.degree. C. The air is compressed by the air compressor (30) and then enters the horizontal generator (4) and stirs the reaction liquid to make the reaction uniform while releasing and diluting the chlorine dioxide gas; in addition, the blower (7) blows the air into the horizontal generator (4) for further diluting the chlorine dioxide gas concentration to 9% (V/V); the diluted chlorine dioxide gas is sent out to the end use areas by the induced draft fan (8). The product has a moisture content of 7.5% (w/w) and is essentially free of chlorine.
[0037] Step 2, while the raw material enters into the horizontal generator (4) from one end, the reacted reaction liquid is discharged from the other end of the horizontal generator (4) to the mother liquid discharge tank (9) for storage, and then sent to the mother liquid lower circulating pipe (23) by the crystallizer feed pump (10), and circulation, heating, evaporation and concentration between the evaporation crystallizer (13) and the heater (12) under the pumping action of the circulating pump (11); the temperature in the evaporation crystallizer is controlled at 70.degree. C., the vacuum degree is controlled at -81 KPa(g). The salt cake in the mother liquid is concentrated and crystallized and is deposited at the bottom of the evaporation crystallizer (13) and the lower circulating pipe (23) and sent to the filtration device (15) through the feed pump (14) for filtration, washing and concentration. The concentrated solid salt cake is introduced into the dryer (16) to produce sodium sulfate with dryness up to 99.6% (which can be sold as a product); the filtrate is returned to the mother liquid circulating pipe (23) via the filtrate pipe (22) for recycling. No solid and liquid wastes produced throughout the production process.
[0038] Step 3, the clarified mother liquid in the upper of the evaporation crystallizer (13) is sent to the mother liquid tank (18) by the mother liquid extraction pump (17) for buffering, and then sent to the horizontal generator (4) by the mother liquid return pump (19).
[0039] It will be readily understood that the components of various embodiments of the present invention, as generally described and illustrated in the figures herein, may be arranged and designed in a wide variety of different configurations. Thus, the detailed description of the embodiments of the present invention, as represented in the attached figures, is not intended to limit the scope of the invention as claimed but is merely representative of selected embodiments of the invention.
[0040] The features, structures, or characteristics of the invention described throughout this specification may be combined in any suitable manner in one or more embodiments. For example, reference throughout this specification to "certain embodiments," "some embodiments," or similar language means that a particular feature, structure, or characteristic described in connection with the embodiment is included in at least one embodiment of the present invention. Thus, appearances of the phrases "in certain embodiments," "in some embodiment," "in other embodiments," or similar language throughout this specification do not necessarily all refer to the same group of embodiments and the described features, structures, or characteristics may be combined in any suitable manner in one or more embodiments.
[0041] It should be noted that reference throughout this specification to features, advantages, or similar language does not imply that all of the features and advantages that may be realized with the present invention should be or are in any single embodiment of the invention. Rather, language referring to the features and advantages is understood to mean that a specific feature, advantage, or characteristic described in connection with an embodiment is included in at least one embodiment of the present invention. Thus, discussion of the features and advantages, and similar language, throughout this specification may, but do not necessarily, refer to the same embodiment.
[0042] Furthermore, the described features, advantages, and characteristics of the invention may be combined in any suitable manner in one or more embodiments. One skilled in the relevant art will recognize that the invention can be practiced without one or more of the specific features or advantages of a particular embodiment. In other instances, additional features and advantages may be recognized in certain embodiments that may not be present in all embodiments of the invention.
[0043] One having ordinary skill in the art will readily understand that the invention as discussed above may be practiced with steps in a different order, and/or with hardware elements in configurations which are different than those which are disclosed. Therefore, although the invention has been described based upon these preferred embodiments, it would be apparent to those of skill in the art that certain modifications, variations, and alternative constructions would be apparent, while remaining within the spirit and scope of the invention. In order to determine the metes and bounds of the invention, therefore, reference should be made to the appended claims.
* * * * *
D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.