Suspensions Of Cyclosporin A Form 2
Gore; Anuradha V. ; et al.
U.S. patent application number 16/277016 was filed with the patent office on 2019-11-14 for suspensions of cyclosporin a form 2. The applicant listed for this patent is Allergan, Inc.. Invention is credited to E. Quinn Farnes, Anuradha V. Gore, Prem Swaroop Mohanty.
| Application Number | 20190343917 16/277016 |
| Document ID | / |
| Family ID | 47278531 |
| Filed Date | 2019-11-14 |








View All Diagrams
| United States Patent Application | 20190343917 |
| Kind Code | A1 |
| Gore; Anuradha V. ; et al. | November 14, 2019 |
SUSPENSIONS OF CYCLOSPORIN A FORM 2
Abstract
Disclosed herein are methods of formulating cyclosporin A Form 2.
| Inventors: | Gore; Anuradha V.; (Irvine, CA) ; Mohanty; Prem Swaroop; (Irvine, CA) ; Farnes; E. Quinn; (Laguna Beach, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 47278531 | ||||||||||
| Appl. No.: | 16/277016 | ||||||||||
| Filed: | February 15, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14995868 | Jan 14, 2016 | 10206971 | ||
| 16277016 | ||||
| 14325167 | Jul 7, 2014 | 9238002 | ||
| 14995868 | ||||
| 13677014 | Nov 14, 2012 | 8796222 | ||
| 14325167 | ||||
| 61559866 | Nov 15, 2011 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 9/0014 20130101; A61P 27/02 20180101; A61K 47/26 20130101; A61K 38/13 20130101; A61K 9/14 20130101; A61K 47/06 20130101; A61P 27/14 20180101; C07K 7/645 20130101; A61K 9/0048 20130101; A61K 47/10 20130101 |
| International Class: | A61K 38/13 20060101 A61K038/13; C07K 7/64 20060101 C07K007/64; A61K 9/00 20060101 A61K009/00; A61K 9/14 20060101 A61K009/14; A61K 47/10 20060101 A61K047/10; A61K 47/26 20060101 A61K047/26; A61K 47/06 20060101 A61K047/06 |
Claims
1. A nanosuspension formulation comprising particles of cyclosporin A form 2 and a vehicle, wherein the average size (d90) of the particles is less than about 10 .mu.m.
2. The formulation of claim 1, wherein the vehicle comprises at least one surfactant and at least one stabilizer.
3. The formulation of claim 2, wherein cyclosporin is at a concentration of about 0.01% to about 10%.
4. The formulation of claim 3, wherein the vehicle comprises one or more surfactants selected from the group consisting of polyoxyethylene (20) sorbitan monooleate, polyethylene glycol 660 hydroxystearate, polyoxyethylene (40) stearate Myrj 52, pluronic F68, polyoxyethylene sorbitan monolaurate, and sodium glycocholate.
5. The formulation of claim 3, wherein the vehicle comprises one or more surfactants selected from the group consisting of polyoxyethylene (20) sorbitan monooleate at a concentration of about 0.1% to about 5% (w/v), polyethylene glycol 660 hydroxystearate at a concentration of about 0.1% to about 5% (w/v), polyoxyethylene (40) stearate Myrj 52 at a concentration of about 0.1% to about 5% (w/v), pluronic F68 at a concentration of about 0.1% to about 5% (w/v), polyoxyethylene sorbitan monolaurate at a concentration of about 0.1% to about 5% (w/v), and sodium glycocholate at a concentration of about 0.1% to about 5% (w/v).
6. The formulation of claim 4, wherein the vehicle comprises at least one stabilizer selected from the group consisting of hydroxy propyl cellulose, hydroxypropylmethyl cellulose, hydroxyethylcellulose, polyvinyl pyrolidone, and carboxymethylcellulose.
7. The formulation of claim 5, wherein the vehicle comprises one or more stabilizers each selected from the group consisting of hydroxypropyl cellulose at a concentration of about 0.1% to about 5% (w/v), hydroxypropylmethyl cellulose at a concentration of about 0.1% to about 5% (w/v), hydroxyethylcellulose at a concentration of about 0.1% to about 5% (w/v), and polyvinyl pyrolidone at a concentration of about 0.1% to about 5% (w/v), and carboxymethylcellulose at a concentration of about 0.1% to about 5% (w/v).
8. The formulation of claim 7, wherein the vehicle further comprises one or more ingredients selected from glycerin, mannitol, Pemulen TR-2, sodium citrate dihydrate, potassium chloride, boric acid, sodium borate decahydrate, and water.
9. The formulation of claim 7, wherein the vehicle further comprises one or more ingredients selected from glycerin at concentration of about 1.0% to about 2.2%, mannitol at concentration of about 0.5%, Pemulen TR-2 at concentration of about 0.01% to about 0.1%, sodium citrate dihydrate at concentration of about 0.4%, potassium chloride at concentration of about 0.14%, boric acid at concentration of about 0.25%, sodium borate decahydrate at concentration of about 0.41%, and water.
10. The formulation of claim 1, wherein the average size of the particles is less than about 1 .mu.m.
Description
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This patent application is a continuation of copending U.S. patent application Ser. No. 14/995,868, filed Jan. 14, 2016, which is a continuation of U.S. patent application Ser. No. 14/325,167, filed Jul. 7, 2014, now issued as U.S. Pat. No. 9,238,002, which is a divisional of U.S. patent application Ser. No. 13/677,014, filed Nov. 14, 2012, now issued as U.S. Pat. No. 8,796,222, which claims priority to U.S. Provisional Patent Application No. 61/559,866, filed Nov. 15, 2011, the entire contents of which are hereby incorporated by reference.
FIELD OF THE INVENTION
[0002] This invention relates to the fields of nanotechnology and drug formulation technology.
BACKGROUND
[0003] Cyclosporine A is the active ingredient in Restasis.RTM., a drug that is used to treat dry eye disease. Cyclosporin A is poorly soluble in water, and so is currently formulated either by dissolving the drug in oil to form an emulsion, or by mixing the drug with high levels of surfactants and/or solubilizers to form an aqueous solution. The inventors have discovered a formulation of cyclosporin A using a new crystalline polymorph of cyclosporin A, to create nanosuspensions comprising particles of cyclosporin A having an average size of around 1 micrometer or less (to put that number in perspective, the average thickness of a human hair is around 100 micrometers).
[0004] A nanosuspension of cyclosporin A, when delivered topically to the eye, may have one or more advantages, including the following: [0005] a higher bioavailability compared to suspensions, due to the higher surface area available for dissolution; [0006] a longer retention on the eye due to smaller particles, leading to further improvement in bioavailability; [0007] a lower potential for foreign body sensation or particle irritation, thus reducing tearing and drainage of formulations from the eye; [0008] a lower level of surfactants or solubilizers in the formulation, improving tolerability and bioavailability of the drug.
BRIEF DESCRIPTION OF THE FIGURES
[0009] FIG. 1 shows the particle size distribution of cyclosporin A Form 2 nanosuspensions prepared using a high pressure homogenizer compared to suspensions prior to milling.
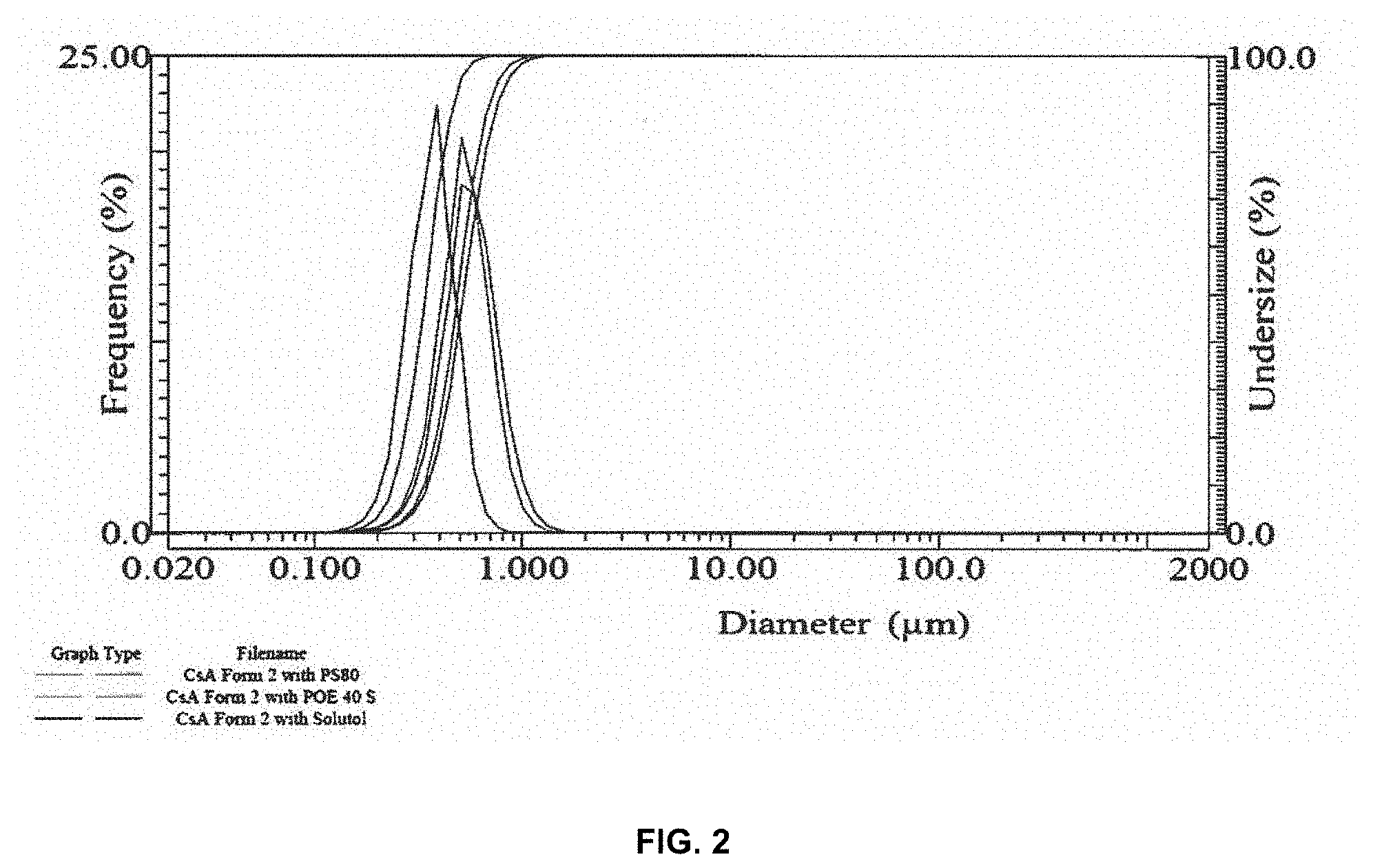
[0010] FIG. 2 compares the particle size distribution of cyclosporin A Form 2 nanosuspensions prepared using different surfactants and stabilizers.
[0011] FIG. 3 shows the particle size distribution of Formulation diluted in vehicle containing Na CMC.
[0012] FIG. 4 shows average particle size of a nanosuspension using cyclosporin A Form 3 compared to a nanosuspensions using Form 2 after milling using a microfluidizer. Form 2 Forms a nanosuspension (particle size <1 .mu.m) while Form 3 does not.
[0013] FIGS. 5-13 show the results when four different formulations according to the invention (summarized in Table 2) and Restasis.RTM. are administered to NZW female rabbits (two rabbits total, one formulation per eye) in a single topical dose:
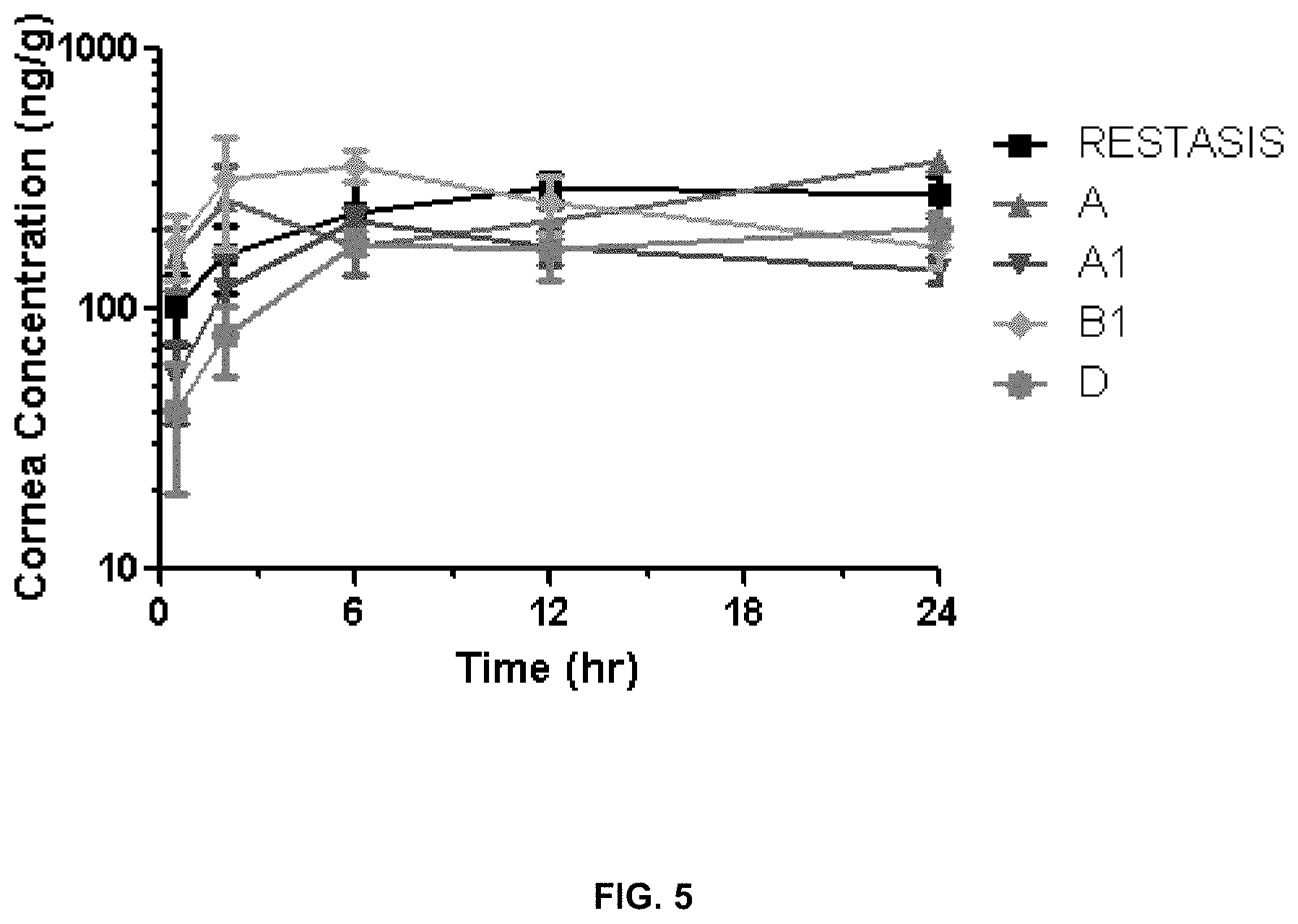
[0014] FIG. 5 shows concentrations in the cornea.
[0015] FIG. 6 summarizes pharmacokinetic data.
[0016] FIG. 7 shows concentrations in the bulbar conjunctiva.
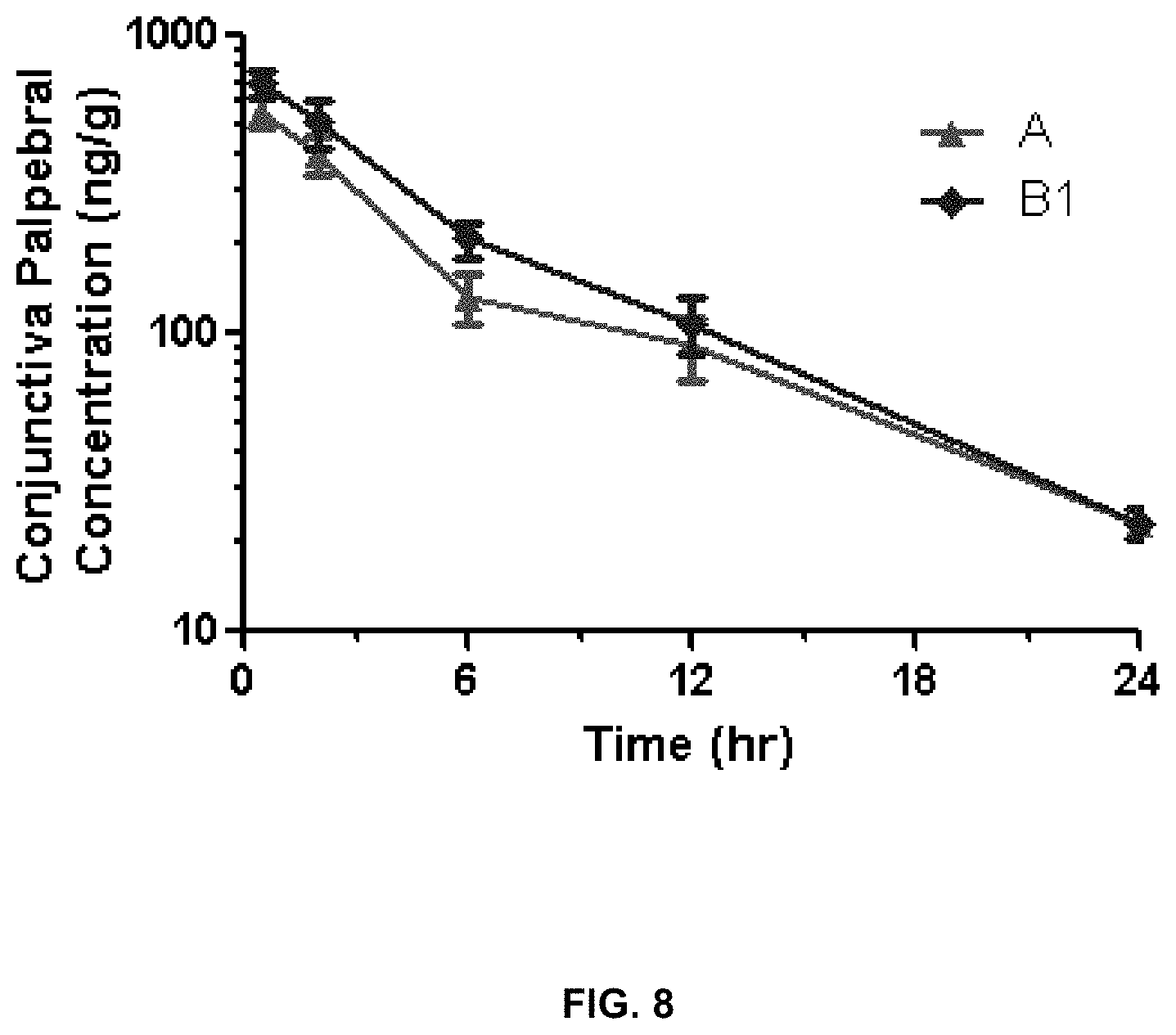
[0017] FIG. 8 shows concentrations in the palpebral conjunctiva.
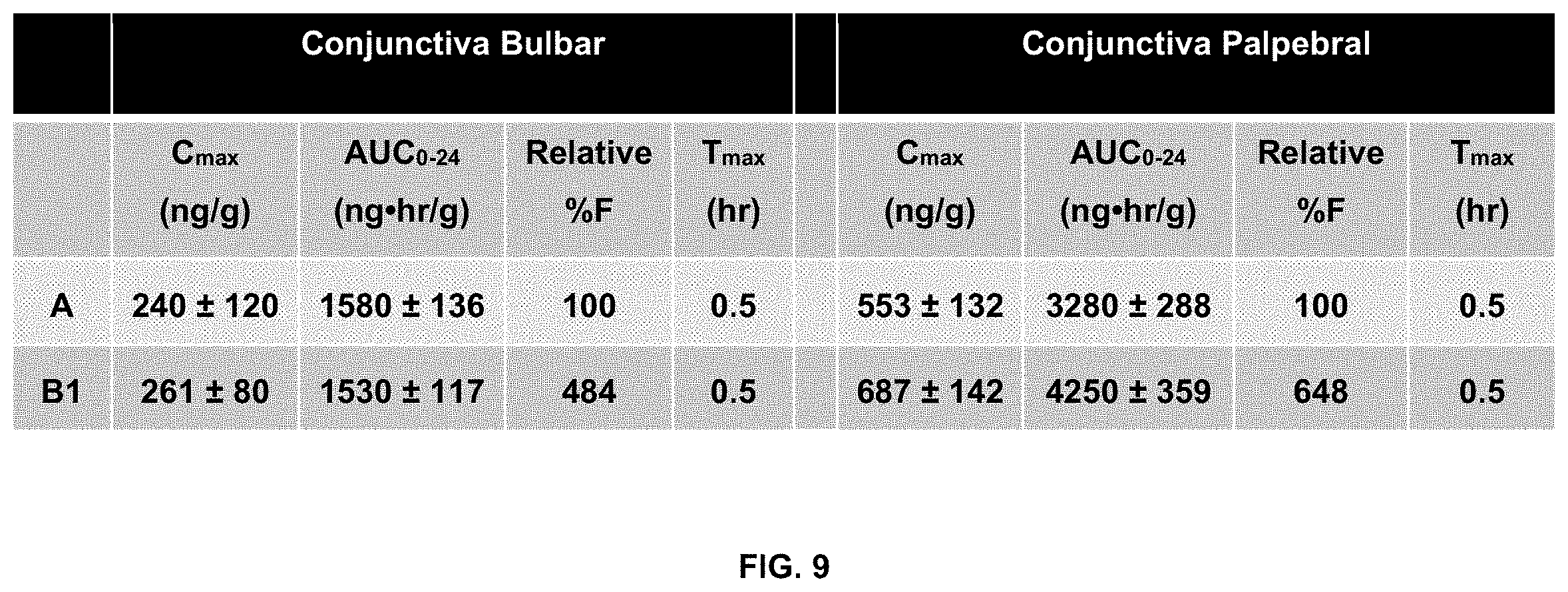
[0018] FIG. 9 summarizes pharmacokinetic data for the bulbar conjunctiva and palpebral conjunctiva.
[0019] FIG. 10 shows concentrations in the bulbar conjunctiva.
[0020] FIG. 11 shows concentrations in the palpebral conjunctiva.
[0021] FIG. 12 shows concentrations in the lacrimal gland.
[0022] FIG. 13 summarizes pharmacokinetic data for lacrimal gland.
[0023] FIG. 14 depicts characteristic X-ray powder diffraction (XRPD) patterns of CsA in a new crystalline form (designated as Form 2 herein), tetragonal form (designated as Form 1 herein), and orthorhombic form (designated as Form 3 herein).
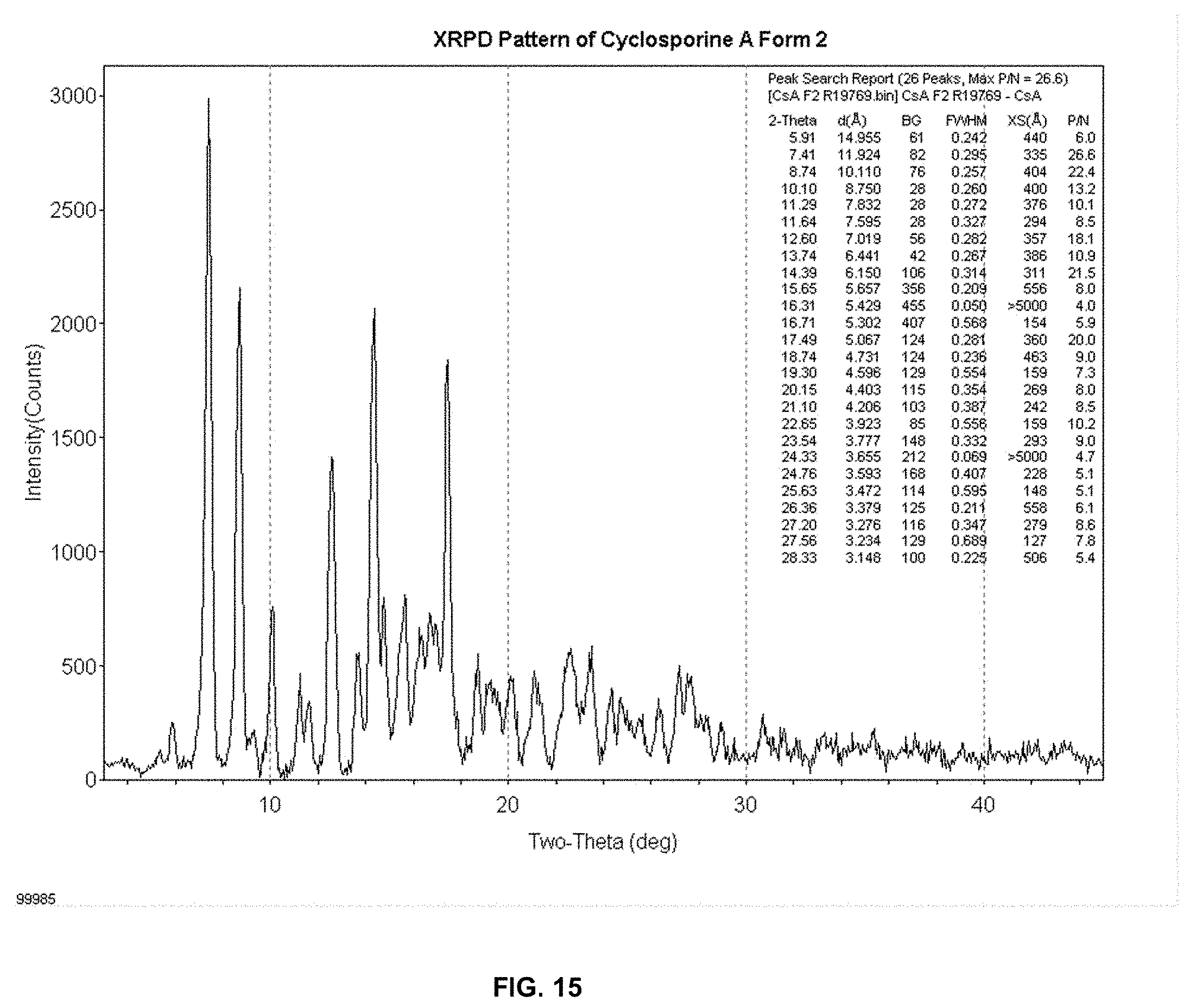
[0024] FIG. 15 depicts the XRPD diffractogram of CsA crystalline Form 2.
[0025] FIG. 16 depicts the water sorption/desorption profile of CsA Form 2.
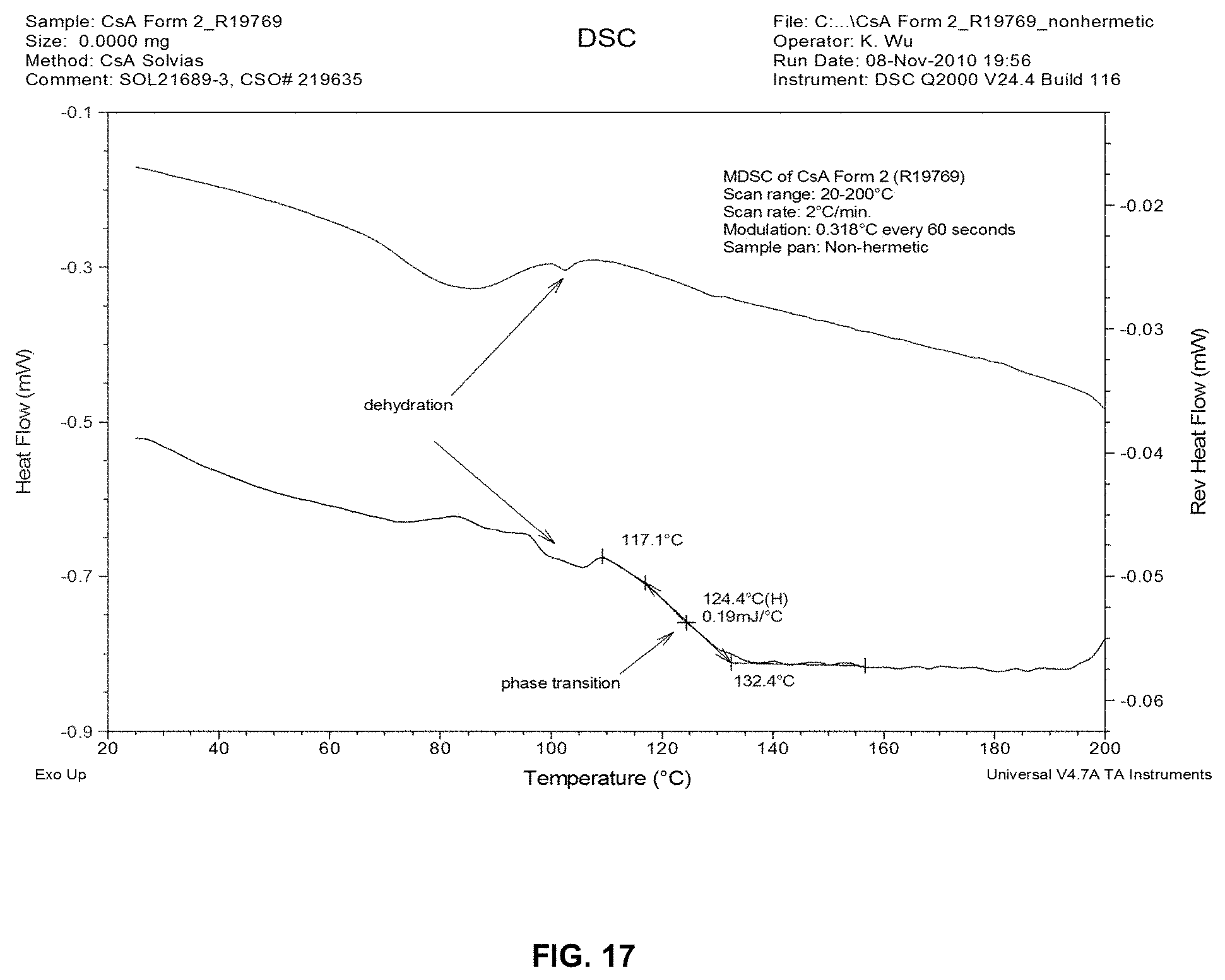
[0026] FIG. 17 depicts MDSC analysis of CsA Form 2 recovered from 0.04% formulation with 1% PS80.
[0027] FIG. 18 shows the simulated XRPD pattern of cyclosporine A forms.
DETAILED DESCRIPTION
Cyclosporin A
[0028] Cyclosporin A (CsA) is a cyclic peptide having the following chemical structure:
##STR00001##
Its chemical name is cyclo[[(E)-(2S,3R,4R)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl]-L-2-- aminobutyryl-N-methylglycyl-N-methyl-Lleucyl-L-valyl-N-methyl-L-leucyl-L-a- lanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-Nmethyl-L-valyl]. It is also known by the names cyclosporine, cyclosporine A, ciclosporin, and ciclosporin A. It is the active ingredient in Restasis.RTM. (Allergan, Inc., Irvine, Calif.), an emulsion comprising 0.05% (w/v) cyclosporin. Restasis.RTM. is approved in the United States to increase tear production in patients whose tear production is presumed to be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca.
Cyclosporin a Form 2
[0029] Cyclosporin A is known to exist in an amorphous form, liquid crystal form, tetragonal crystalline form (form 1), and an orthorhombic form (form 3). A new crystalline form, cyclosporin A Form 2, has recently been discovered.
[0030] The XRPD pattern of CsA Form 2 differs significantly from the tetragonal form and orthorhombic form (FIG. 14). The major crystalline peaks for CsA form 2 appear at (2.theta.) when scanned by an X-ray diffractometer with X-ray source as Cu K.alpha. radiation, .lamda.=1.54 .ANG., at 30 kV/15 mA: 7.5, 8.8, 10.2, 11.3, 12.7, 13.8, 14.5, 15.6 and 17.5 (d-spacing in crystal lattice at about 11.8, 10.0, 8.7, 7.8, 7.0, 6.4, 6.1, 5.6 and 5.1 .ANG., respectively, FIG. 15). These major peaks are defined as those being unique to Form 2 relative to the orthorhombic or tetragonal forms; as well as, peaks having an intensity greater than 5 times the background.
[0031] In one embodiment, the new crystalline form (Form 2) of CsA is a nonstoichiometric hydrate of Cyclosporin A. In another embodiment, the crystalline Form 2 is represented by the formula:
##STR00002##
wherein X is the number of molecules of water and varies from 0 to 3. In one embodiment, X in the above formula is 2.
[0032] Form 2 appears to be a kinetically stable form of CsA in aqueous suspensions. Suspensions containing Form 2 show no conversion to other known polymorphic or pseudomorphic forms upon storage. It has been found that Form 1 and the amorphous form convert to Form 2 in the presence of water.
[0033] The single crystal structure of the hydrate form of CsA Form 2 has been determined and the crystal structure parameters are listed in Table 2. These results indicate that Form 2 is unique compared to other known crystalline forms of cyclosporine A.
TABLE-US-00001 TABLE 1 Crystal data and data collection parameters of crystal structure solution of CsA Form 2. formula C.sub.52H.sub.115N.sub.11O.sub.14 formula weight 1238.67 space group P 2.sub.1 2.sub.1 2.sub.1 (No. 19) a (.ANG.) 12.6390(5) b (.ANG.) 19.7562(8) c (.ANG.) 29.266(2) volume (.ANG..sup.3) 7383.6(7) Z 4 d.sub.calc (g cm.sup.-3) 1.114 crystal dimensions (mm) 0.27 .times. 0.16 .times. 0.12 temperature (K) 150 radiation (wavelength in .ANG.) Cu K.sub.3 (1.54184) monochromator confocal optics linear abs coef (mm.sup.-1) 0.640 absorption correction applied empirical.sup.a transmission factors (min, max) 0.80, 0.93 diffractometer Rigaku RAPID-II h, k, l range -13 to 13 -21 to 21 -32 to 21 2.theta. range (deg) 5.38-115.00 mosaicity (deg) 1.31 programs used SHELXTL F.sub.0oo 2704.0 weighting 1/[.sigma..sup.2(Fo.sup.2) + (0.0645P).sup.2 + 0.0000P] where P = (Fo.sup.2 + 2Fc.sup.2)/3 data collected 37360 unique data 9964 R.sub.int 0.077 data used in refinement 9964 cutoff used in R-factor calculations F.sub.o.sup.2 > 2.0 s(F.sub.o.sup.2) data with I > 2.0 s(l) 6597 number of variables 834 largest shift/esd in final cycle 0.00 R(F.sub.o) 0.061 R.sub.w(F.sub.o.sup.2) 0.145 goodness of fit 1.037 absolute structure determination Flack parameter.sup.b (0.0(3))
[0034] The asymmetric unit of this CsA Form 2 contains one cyclosporine A molecule and two water molecules. It is possible that any small molecule that can hydrogen bond to water could play the role of space filler, which would give a range of potential structures running from the orthorhombic dihydrate to distorted monoclinic dihydrate The XRPD pattern calculated from the single-crystal structure is shown in FIG. 8 and it matches the experimental pattern shown in FIG. 2. These matching patterns further corroborate that Form 2 is a unique and pure crystalline form of cyclosporine A.
[0035] Without wishing to be bound by theory, thermogravimetric analysis combined with KF titration and vapor sorption desorption analysis (VSA) suggest that CsA Form 2 is a non-stoichiometric hydrate of CsA. The vapor sorption analysis of Cyclosporine Form 2 indicates that water content in the new crystal form reversibly varies with relative humidity as shown in FIG. 16. Similar to the tetragonal form, the new CsA form undergoes a phase transition to a liquid crystal or amorphous form at 124.4.degree. C. prior to melting as indicated by the modulated differential calorimetric (MDSC) analysis (FIG. 17).
[0036] Cyclosporin A Form 2 may be obtained by suspending amorphous 0.05% cyclosporin A (w/v) in 1% Polysorbate 80, heating the solution to 65.degree. C., holding it at that temperature for 24 hours, and then recovering the precipitate by vacuum filtration. One can then use the cyclosporin A Form 2 thus obtained to generate additional amounts, using Cyclosporin A Form 2 as a seed crystal; in this method, one suspends about 30 g cyclosporin A in a solution of 900 ml water containing 1% (w/v) Polysorbate 80, heats the solution to 65.degree. C., and then seeds it with 0.2 g of cyclosporin A Form 2 at a temperature of 52.degree. C. The solution is then stirred for about 22 hours at a temperature of between about 61.degree. C. and 65.degree. C., and then recovers the precipitate that results.
[0037] Further details regarding CsA Form 2 may be found in U.S. patent application Ser. No. 13/480,710, the entire contents of which are incorporated by reference herein.
Suspensions of Cyclosporin a Form 2
[0038] Compositions of the invention are ophthalmically acceptable suspensions of Cyclosporin A form 2. By "ophthalmically acceptable," the inventors mean that the suspensions are formulated in such a way as to be non-irritating when administered to the eye of a mammal, such as a human. In one embodiment, the compositions are suspensions; that is, they comprise particles of cyclosporin A form 2, having an average particle size greater than about 1 .mu.m, dispersed throughout a liquid vehicle. In another embodiment, the compositions are nanosuspensions; that is, they comprise particles of cyclosporin A form 2, having an average particle size of less than about 1 .mu.m, that are dispersed throughout a liquid vehicle.
[0039] In one embodiment, the suspension comprises cyclosporin A form 2 at a concentration of about 0.001% to about 10% (w/v). In one embodiment, the suspension comprises cyclosporin A form 2 at a concentration of about 0.001% (w/v) to about 0.01%, about 0.001% (w/v) to about 0.04% (w/v), about 0.001% (w/v) to about 0.03% (w/v), about 0.001% (w/v) to about 0.02% (w/v), or about 0.001% (w/v) to about 0.01% (w/v). In another embodiment, the suspension comprises cyclosporin A form 2 at a concentration of about 0.01% (w/v) to about 0.05%, about 0.01% (w/v) to about 0.04% (w/v), about 0.01% (w/v) to about 0.03% (w/v), about 0.01% (w/v) to about 0.02% (w/v), or about 0.01% (w/v) to about 0.01% (w/v). In another embodiment, the suspension comprises cyclosporin A form 2 at a concentration of about 0.01% (w/v) to about 0.1%, about 0.1% (w/v) to about 0.5% (w/v), about 0.01% (w/v) to about 1% (w/v), or about 1% (w/v) to about 10%.
[0040] For example, the suspensions may comprise about 0.001% (w/v), about 0.002% (w/v), about 0.003% (w/v), about 0.004% (w/v), about 0.005% (w/v), about 0.006% (w/v), about 0.007% (w/v), about 0.008% (w/v), about 0.009% (w/v), about 0.01% (w/v), about 0.015% (w/v), about 0.02% (w/v), about 0.025% (w/v), about 0.03% (w/v), about 0.035% (w/v), about 0.04% (w/v), about 0.045% (w/v), about 0.05% (w/v), about 0.055% (w/v), about 0.06% (w/v), about 0.065% (w/v), about 0.07% (w/v), about 0.075% (w/v), about 0.08% (w/v), about 0.085% (w/v), about 0.09% (w/v), about 0.095% (w/v), about 0.1% (w/v), about 0.15% (w/v), about 0.2% (w/v), about 0.25% (w/v), about 0.3% (w/v), about 0.35% (w/v), about 0.4% (w/v), about 0.45% (w/v), about 0.5% (w/v), about 0.55% (w/v), about 0.6% (w/v), about 0.65% (w/v), about 0.7% (w/v), about 0.75% (w/v), about 0.8% (w/v), about 0.85% (w/v), about 0.9% (w/v), about 0.95% (w/v), or about 1.0% (w/v) cyclosporin A form 2.
[0041] In one embodiment, the suspension comprises a surfactant. In one embodiment, the surfactant is selected from polyoxyethylene (20) sorbitan monooleate (Polysorbate 80), polyethylene glycol 660 hydroxystearate (Solutol), polyoxyethylene (40) stearate Myrj 52 (POE-40-Stearate), pluronic F68 (Polaxamer 188), polyoxyethylene sorbitan monolaurate (Polysorbate 20), and sodium glycocholate (NaGC). The vehicle may contain all of these surfactants, or one, two, three, four, or five of them.
[0042] One can use between about 0.001% (w/v) and about 5% (w/v) of the surfactant. In one embodiment, the suspensions contain about 0.001% (w/v) to about 1% (w/v), about 0.001% (w/v) to about 0.1% (w/v), about 0.01% (w/v) to about 0.1% (w/v), or about 0.1% (w/v) to about 1% (w/v) of the surfactant. For example, the suspensions may contain about 0.001% (w/v), about 0.002% (w/v), about 0.003% (w/v), about 0.004% (w/v), about 0.005% (w/v), about 0.006% (w/v), about 0.007% (w/v), about 0.008% (w/v), about 0.009% (w/v), about 0.01% (w/v), about 0.02% (w/v), about 0.03% (w/v), about 0.04% (w/v), about 0.05% (w/v), about 0.06% (w/v), about 0.07% (w/v), about 0.08% (w/v), about 0.09% (w/v), about 0.1% (w/v), about 0.2% (w/v), about 0.3% (w/v), about 0.4% (w/v), about 0.5% (w/v), about 0.6% (w/v), about 0.7% (w/v), about 0.8% (w/v), about 0.9% (w/v), about 1% (w/v), about 2% (w/v), about 3% (w/v), about 4% (w/v), or about 5% (w/v) of the surfactant.
[0043] When using more than one surfactant, the suspension may contain the same or different amounts of each.
[0044] In addition to a surfactant, the suspensions may comprise a stabilizer. In one embodiment, the stabilizer is selected from hydroxy propyl cellulose, hydroxypropylmethyl cellulose, hydroxyethylcellulose, polyvinyl pyrolidone, carboxymethylcellulose, Pemulen.RTM., and Pemulen.RTM. TR-2. Pemulen.RTM. is the trade name for high molecular weight, crosslinked copolymers of acrylic acid and C10-C30 alkyl acrylate produced by Lubrizol Corp. Pemulen.RTM. TR-2 is a C10-30 alkyl acrylate crosspolymer containing a higher level of hydrophobic groups than other Pemulen.RTM. polymers. The vehicle may contain all of these stabilizers, or none of them, or it may contain one, two, three, four, or five of them.
[0045] One can use between about 0.01% (w/v) and about 10% (w/v) of the stabilizer. In one embodiment, the suspensions contain about 0.01% (w/v) to about 1% (w/v), or about 0.01% (w/v) to about 0.1% (w/v), or about 0.1% (w/v) to about 1% (w/v) of the stabilizer. For example, the suspensions may contain about 0.01% (w/v), about 0.02% (w/v), about 0.03% (w/v), about 0.04% (w/v), about 0.05% (w/v), about 0.06% (w/v), about 0.07% (w/v), about 0.08% (w/v), about 0.09% (w/v), about 0.1% (w/v), about 0.2% (w/v), about 0.3% (w/v), about 0.4% (w/v), about 0.5% (w/v), about 0.6% (w/v), about 0.7% (w/v), about 0.8% (w/v), about 0.9% (w/v), about 1% (w/v), about 2% (w/v), about 3% (w/v), about 4% (w/v), about 5% (w/v), about 6% (w/v), about 7% (w/v), about 8% (w/v), about 9% (w/v), or about 10% of the stabilizer.
[0046] When using more than one surfactant, the suspension may contain the same or different amounts of each.
[0047] In addition to a surfactant, the vehicle may also comprise a tonicity adjustor selected from glycerin, mannitol, sodium citrate dihydrate, potassium chloride, boric acid, and sodium borate decahydrate. The tonicity adjustor is added as needed to achieve the desired tonicity; the vehicle may contain all of these tonicity adjusters, or none of them, or it may contain one, two, three, four, or five of them. In one embodiment, the tonicity adjustors are present in an amount of between about 0.1 (w/v) and about 10% (w/v). When using more than one tonicity adjustor, the suspension may contain the same or different amounts of each.
[0048] The suspension usually contains water, in an amount sufficient to provide a desired pH, tonicity, and other characteristics that would make the suspension appropriate for administration to the eye.
Methods of Preparing Suspension of Cyclosporin a Form 2
[0049] The formulations of the invention may be made by mixing cyclosporin A form 2 with the appropriate surfactants, stabilizers, and tonicity adjustors, as described above, to form a suspension If fine particles of cyclosporin A are desired, the suspension is then milled using a high pressure homogenizer, such as those commercially available from Microfluidics Intl Corp. of Newton, Mass. A unique and surprising property of cyclosporin form A 2 is that it may be milled, if desired, to obtain a suspension with an average particle size (d90) of less than 1 .mu.m. The cyclosporin A in such a nanosuspension has higher bioavailability compared to other (macro) suspensions of cyclosporin A, due to the higher surface area available for dissolution; bioavailability is further enhanced because the smaller particles enable the Cyclosporin A to be retained on the eye longer. The smaller particles of the nanosuspensions result in a formulation with a lower potential to produce a foreign body sensation or other irritation that a subject perceives when the formulation is instilled in the eye. Also, because the particles are smaller, they associate more readily with surfactants and stabilizers, thereby permitting one to use lower concentrations of them.
[0050] After the cyclosporin A form 2 suspension is milled, it is diluted to obtain the final product.
Methods of Treatment
[0051] Compositions of the invention may be used to treat any condition of the eye which is known to be amenable to topical treatment with cyclosporin A (such as with Restasis.RTM.) at the concentrations stated here. For example, compositions of the invention may be used to treat patients suffering from dry eye, to treat blepharitis and meibomian gland disease, to restore corneal sensitivity that has been impaired due to refractive surgery on the eye, to treat allergic conjunctivitis and atopic and vernal keratoconjunctivitis, and to treat ptyregia, conjunctival and corneal inflammation, keratoconjuntivitis, graft versus host disease, post-transplant glaucoma, corneal transplants, mycotic keratitis, Thygeson's superficial punctate keratitis, uveitis, and Theodore's superior limbic keratoconjunctivitis, among other conditions.
[0052] The International Dry Eye Workshop (DEWS) defines dry eye as "a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface, accompanied by increased osmolarity of the tear film and inflammation of the ocular surface." It includes those conditions, such as keratoconjunctivitis sicca, that are caused by tear deficiency or excessive evaporation of tears.
[0053] Blepharitis is a chronic disorder producing inflammation of the anterior and posterior lid margin, with involvement of skin and its related structures (hairs and sebaceous glands), the mucocutaneous junction, and the meibomian glands. It can also affect the conjunctiva, tear film, and the corneal surface in advanced stages and may be associated with dry eye. Blepharitis is commonly classified into anterior or posterior blepharitis, with anterior affecting the lash bearing region of the lids, and posterior primarily affecting the meibomian gland orifices.
[0054] Meibomian gland disease most often occurs as one of three forms: primary meibomitis, secondary meibomitis, and meibomian seborrhea. Meibomian seborrhea is characterized by excessive meibomian secretion in the absence of inflammation (hypersecretory meibomian gland disease). Primary meibomitis, by contrast, is distinguished by stagnant and inspissated meibomian secretions (obstructive hypersecretory meibomian gland disease). Secondary meibomitis represents a localized inflammatory response in which the meibomian glands are secondarily inflamed in a spotty fashion from an anterior lid margin blepharitis.
[0055] Impaired corneal sensitivity often occurs after refractive surgery, such as photorefractive keratectomy, laser assisted sub-epithelium keratomileusis (LASEK), EPI-LASEK, customized transepithelial non-contact ablation, or other procedures in which the corneal nerves are severed. Impaired corneal sensitivity may also occur after viral infection, such as by HSV-1, HSV-2, and VZV viruses. Patients with impaired corneal sensitivity often complain that their eyes feel dry, even though tear production and evaporation may be normal, suggesting that "dryness" in such patients is actually a form of corneal neuropathy that results when corneal nerves are severed by surgery or inflamed after viral infection.
[0056] Allergic conjunctivitis is an inflammation of the conjunctiva resulting from hypersensitivity to one or more allergens. It may be acute, intermittent, or chronic. It occurs seasonally, that is, at only certain time of the year, or it occurs perennially, that is, chronically throughout the year. Symptoms of seasonal and perennial allergic conjunctivitis include, in addition to inflammation of the conjunctiva, lacrimation, tearing, conjunctival vascular dilation, itching, papillary hyperlasia, chemosis, eyelid edema, and discharge from the eye. The discharge may form a crust over the eyes after a night's sleep.
[0057] Atopic keratoconjunctivitis is a chronic, severe form of allergic conjunctivitis that often leads to visual impairment. Symptoms include itching, burning, pain, redness, foreign body sensation, light sensitivity and blurry vision. There is often a discharge, especially on awakening from a night's sleep; the discharge may be stringy, ropy, and mucoid. The lower conjunctiva is often more prominently affected than the upper conjunctiva. The conjunctiva may range from pale, edematous, and featureless to having the characteristics of advanced disease, including papillary hypertrophy, subepithelial fibrosis, formix foreshortening, trichiasis, entropion, and madurosis. In some patients the disease progresses to punctate epithelial erosions, corneal neovascularization, and other features of keratopathy which may impair vision. There is typically goblet cell proliferation in the conjunctiva, epithelial pseudotubular formation, and an increased number of degranulating eosinophils and mast cells in the epithelium. CD25+ T lymphocytes, macrophages, and dendritic cells (HLA-DR.sup.+, HLA-CD1+) are significantly elevated in the substantia propria.
[0058] Like atopic keratoconjunctivitis, vernal keratoconjunctivitis is a severe form of allergic conjunctivitis, but it tends to affect the upper conjunctiva more prominently than the lower. It occurs in two forms. In the palpebral form, square, hard, flattened, closely packed papillae are present; in the bulbar (limbal) form, the circumcorneal conjunctiva becomes hypertrophied and grayish. Both forms are often accompanied by a mucoid discharge. Corneal epithelium loss may occur, accompanied by pain and photophobia, as may central corneal plaques and Trantas' dots.
EXAMPLES
[0059] The invention is further illustrated by the following examples.
Example 1
[0060] The inventors prepared the following compositions:
TABLE-US-00002 TABLE 1 Formulations of cyclosporin A prepared in Example 1 A B C D E F Ingredient Concentration % (w/v) CsA 0.01 to 0.05 Polysorbate 80 0.001 to 0.05 Glycerin 1 1.2 1.2 1 1.2 2.2 Mannitol 0.5 CMC 0.5 HEC 0.1-0.5 Pemulen TR-2 0.01-0.1 0.05-0.1 HPMC 0.1-1.0 PVP 0.1-10.0 Sodium Citrate 0.4 0.4 0.4 0.4 0.4 Dihydrate Potassium 0.14 Chloride Boric Acid 0.25 0.25 0.25 0.25 0.25 Sodium Borate 0.41 0.41 0.41 0.41 0.41 Decahydrate Purified Water qs Qs qs qs qs Qs
[0061] The formulations of Table 1 were prepared by the following process:
[0062] 1. Preparation of Concentrated Cyclosporin a Nanosuspensions [0063] a. Cyclosporin A was mixed with an appropriate vehicle to form a suspension. The concentration of Cyclosporin A in this suspension is in the range of 1-10%. [0064] b. The cyclosporin A suspension was milled using a high pressure homogenizer (a Microfluidizer.RTM., manufactured by Microfluidics, Newton, Mass.) or a ball mill to get nanosuspensions such that d90<11 Jm. [0065] c. Vehicles used for preparation of nanosuspension concentrate are as listed in Table 1
[0066] 2. Preparation of Final Product [0067] a. The concentrated nanosuspensions prepared in Step-1 were diluted in vehicles suitable for ophthalmic dosing to obtain final product at required dose strength of cyclosporin A. Vehicles suitable for dilution may contain buffers, stabilizers, gelling agents and/or dilution to obtain final formulations with desired concentration of CsA. [0068] b. Compositions of nanosuspension formulations prepared after dilution of the nanosuspension concentrates are listed in Table 2,
[0069] Particle-size distribution of the different Formulations are shown in FIGS. 1, 2, and 3.
[0070] FIG. 1 shows the particle size distribution of cyclosporin A Form 2 nanosuspensions prepared using a high pressure homogenizer compared to suspensions prior to milling.
[0071] FIG. 2 compares the particle size distribution of cyclosporin A Form 2 nanosuspensions prepared using different surfactants and stabilizers.
[0072] FIG. 3 shows the particle size distribution of Formulation diluted in vehicle containing Na CMC (Table-2, vehicle A). Note that no change in particle size is seen in the diluted Formulations over 2 weeks as compared to the nanosuspension concentrate.
[0073] Average particle size of cyclosporin A Form 3 are shown in FIG. 4. Cyclosporin A Form 2 Forms a nanosuspensions (particle size <1 .mu.m) while Form 3 does not.
[0074] These experiments show that Form 2 consistently produced nanosuspensions with lower particle size than any other crystalline form of CsA. The smaller particle size of Form 2 nanosuspensions is an advantage over other forms; among other reasons, it is expected to show higher bioavailability due to larger surface area for dissolution and longer retention in the eye, as well as improved physical stability.
Example 2
[0075] The inventors prepared the cyclosporin A nanosuspensions listed in Table 2, below:
TABLE-US-00003 TABLE 2 Formulations of cyclosporin A prepared in Example 2 Restasis .RTM. A A1 B1 D Ingredient Concentration (% w/w) Nano-suspension CsA -- 0.05 0.05 0.01 0.05 Polysorbate 80 -- 0.005 0.005 0.001 -- Na glychocholate -- -- -- -- from suspension Oil Phase CsA 0.05 -- -- -- -- Castor oil 1.25 -- -- -- -- Aqueous Vehicle PS80 1 0.045 0.045 0.049 0.05 Glycerin 2.2 2.2 2.2 2.2 2.2 Citric acid*H2O -- -- 0.007 -- -- Na2HPO4*7H2O -- -- 0.134 -- -- Pemulen TR-2 0.05 0.05 -- 0.05 0.05 Gellan Gum -- -- 0.6 -- -- Purified water QS QS QS QS QS
[0076] The inventors administered the above formulations to NZW female rabbits (two rabbits total, one formulation per eye) in a single topical dose. Results are summarized in FIGS. 5-13. They show that formulation B1 delivers a 5-fold lower dose of cyclosporin A (0.01% versus 0.05%) but maintains comparable cornea exposure to 0.05% Restasis.RTM. as well as improves delivery to the bulbar and palpebral conjunctiva.
* * * * *


D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015
D00016

D00017
D00018

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.