Ether-Based Lubricant Compositions, Methods and Uses
LAMB; David Gordon ; et al.
U.S. patent application number 16/470242 was filed with the patent office on 2019-10-10 for ether-based lubricant compositions, methods and uses. The applicant listed for this patent is CASTROL LIMITED. Invention is credited to David Gordon LAMB, John Michael REDSHAW, Kevin Richard WEST, Rebecca YATES.
| Application Number | 20190309237 16/470242 |
| Document ID | / |
| Family ID | 60702781 |
| Filed Date | 2019-10-10 |











View All Diagrams
| United States Patent Application | 20190309237 |
| Kind Code | A1 |
| LAMB; David Gordon ; et al. | October 10, 2019 |
Ether-Based Lubricant Compositions, Methods and Uses
Abstract
##STR00001## The present invention provides a lubricant composition for an internal combustion engine comprising a base oil of lubricating viscosity, wherein the base oil comprises an ether base stock of formula (A): where: R.sub.a and R.sub.b are aliphatic hydrocarbyl groups and may be the same or different; wherein at least one of R.sub.a and R.sub.b is branched-chain alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl; the lubricant composition further comprising: i) at least one molybdenum compound as a lubricant additive which is present, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition; or ii) at least one polymethacrylate compound as a lubricant additive which is present in an amount of from 0.1 to 7.5% by weight of the lubricant composition. The lubricant composition may be used for lubricating a surface in an internal combustion engine as well as for improving the fuel economy performance and/or piston cleanliness performance and/or turbocharger cleanliness performance of an engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine.
| Inventors: | LAMB; David Gordon; (Reading Berkshire, GB) ; REDSHAW; John Michael; (Reading, Berkshire, GB) ; WEST; Kevin Richard; (Reading, Berkshire, GB) ; YATES; Rebecca; (Reading, Berkshire, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 60702781 | ||||||||||
| Appl. No.: | 16/470242 | ||||||||||
| Filed: | December 14, 2017 | ||||||||||
| PCT Filed: | December 14, 2017 | ||||||||||
| PCT NO: | PCT/EP2017/082926 | ||||||||||
| 371 Date: | June 16, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 2205/0285 20130101; C10M 2227/066 20130101; C10M 2203/1025 20130101; C10N 2030/10 20130101; C10N 2030/02 20130101; C10N 2030/08 20130101; C10M 2207/04 20130101; C10N 2030/74 20200501; C10N 2030/54 20200501; C10M 2223/045 20130101; C10N 2030/52 20200501; C10M 2215/065 20130101; C10M 169/044 20130101; C10M 2207/028 20130101; C10M 2207/026 20130101; C10N 2030/04 20130101; C10N 2010/12 20130101; C10M 2215/066 20130101; C10M 2219/046 20130101; C10M 2209/084 20130101; C10M 2201/066 20130101; C10N 2040/25 20130101; C10M 2207/0406 20130101; C10M 2203/1025 20130101; C10N 2020/02 20130101; C10M 2203/1025 20130101; C10N 2020/02 20130101 |
| International Class: | C10M 169/04 20060101 C10M169/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 16, 2016 | CN | PCT/CN2016/110333 |
Claims
1. A lubricant composition for an internal combustion engine comprising a base oil of lubricating viscosity, wherein the base oil comprises an ether base stock of formula (A): ##STR00071## where: R.sub.a and R.sub.b are aliphatic hydrocarbyl groups and may be the same or different; wherein at least one of R.sub.a and R.sub.b is branched-chain alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl; the lubricant composition further comprising: i) at least one molybdenum compound as a lubricant additive which is present, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition; or ii) at least one polymethacrylate compound as a lubricant additive which is present in an amount of from 0.1 to 7.5% by weight of the lubricant composition.
2. The lubricant composition of claim 1, wherein R.sub.a and R.sub.b are independently selected from alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl, provided that when R.sub.a and R.sub.b are both alkyl at least one of R.sub.a and R.sub.b is/are branched-chain alkyl.
3. The lubricant composition of claim 1, wherein R.sub.a contains more carbon atoms than R.sub.b.
4. The lubricant composition of claim 1, wherein R.sub.a contains from 12 to 30 carbon atoms, and/or R.sub.b contains from 2 to 20 carbon atoms.
5. The lubricant composition of claim 1, wherein the ether base stock is of formula (1): ##STR00072## where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl; R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; R.sub.6 is alkyl or ##STR00073## where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; R.sub.9 is H or alkyl; X is alkylene or is absent; and p is 0, 1, 2 or 3; and m and n are 0, 1, 2 or 3, wherein m is 0 when R.sub.4 and R.sub.5 are H.
6. The lubricant composition of claim 5, wherein R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl; and/or wherein R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl.
7. The lubricant composition of claim 5, wherein m and n are 0, 1 or 2.
8. The lubricant composition of claim 5, wherein the ether base stock has the formula (4): ##STR00074## where: R.sub.1 and R.sub.4 are alkyl; R.sub.3 and R.sub.5 are H or alkyl.
9. The lubricant composition of claim 5, wherein the ether base stock has the formula (7): ##STR00075## where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon to which they are attached, cycloalkyl; R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; and R.sub.6 is alkyl.
10. The lubricant composition of claim 1, wherein the ether base stock contains a total number of carbons atoms of from 20 to 50.
11. The lubricant composition of claim 1, wherein the ether base stock is prepared from bio-derived feedstock containing greater than 50% by weight of biobased carbon.
12. The lubricant composition of claim 1, wherein the at least one molybdenum compound is present, on a molybdenum element basis, in an amount from 0.06% to 0.25% by weight of the lubricant composition.
13. The lubricant composition of claim 1, wherein the at least one polymethacrylate compound is present in an amount of from 0.25 to 7% by weight of the lubricant composition.
14. The lubricant composition of claim 1, wherein the at least one polymethacrylate compound is a comb-type polymer.
15. The lubricant composition of claim 1, wherein the base oil of the lubricant composition comprises greater than 10% by weight of the ether base stock and/or wherein the lubricant composition comprises greater than 50% by weight of the base oil.
16. The lubricant composition of claim 15, wherein the base oil of the lubricant composition further comprises a base stock selected from Group I, Group II, Group III, Group IV and Group V base stocks and mixtures thereof.
17. The lubricant composition of claim 1, wherein the lubricant composition has at least one of: a kinematic viscosity at 40.degree. C. of less than 60 cSt; a kinematic viscosity at 100.degree. C. of less than 12 cSt; a viscosity index of greater than 100; a viscosity at 150.degree. C. and a shear rate of 10.sup.6 s.sup.-1 of no greater than 3 cP; and a Noack volatility of less than 25% by weight.
18. The lubricant composition of claim 1, wherein the lubricant composition has at least one of: an oxidative stability performance on a CEC-L-088-02 test indicated by an absolute viscosity increase at 40.degree. C. of no more than 45 cSt; an oxidative stability performance on a CEC-L-109-14 test indicated by an increase in kinematic viscosity at 100.degree. C. of less than 200%; a fuel economy performance on a CEC-L-054-96 test of at least 2.5%; a piston cleanliness performance on a CEC-L-088-02 test indicated by an overall piston merit of at least 8.5; and a high temperature stability performance on a KHT test at 280.degree. C. in accordance with JPI-5S-55-99 indicated by an overall deposit merit of at least 7.0.
19. The lubricant composition of claim 1, wherein the composition further comprises a phenate detergent in an amount of 0.1 to 5% by weight actives of the lubricant composition.
20. The lubricant composition of claim 1, wherein the composition further comprises a neutral sulphonate detergent in an amount of 0.01 to 5% by weight of the lubricant composition.
21. A method of preparing a lubricant composition, said method comprising providing a base oil as defined in claim 1 and blending the base oil with: i) at least one molybdenum compound suitable for use as a lubricant additive such that the molybdenum compound is present, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition; or ii) at least one polymethacrylate compound suitable for use as a lubricant additive such that the polymethacrylate compound is present in an amount of from 0.1 to 7.5% by weight of the lubricant composition, and optionally also blending one or more additional lubricant additives, in order to prepare the lubricant composition.
22. A method of lubricating a surface, said method comprising supplying a lubricant composition as defined in claim 1 to said surface, such as wherein the lubricant composition is supplied to a surface in an internal combustion engine.
23-26. (canceled)
27. A method of reducing or preventing i) scuffing in the piston system of an engine and/or ii) deposits in the turbochargers of an engine, comprising the step of providing to the engine a lubricant composition according to claim 1.
28. A method of improving the fuel economy performance and/or piston cleanliness performance and/or turbocharger cleanliness performance of an engine and/or a vehicle, comprising the step of providing to the engine a lubricant composition according to claim 1.
Description
[0001] The present invention relates to lubricant compositions containing base oils comprising certain ether base stocks which are suitable for use in a lubricant composition intended for use in an internal combustion engine. Also provided are methods and uses of the lubricant compositions and of the ether base stocks.
BACKGROUND
[0002] Lubricating compositions generally comprise a base oil of lubricating viscosity together with one or more additives to deliver properties including for example, reduced friction and wear, improved viscosity index, improved dispersancy, detergency, and resistance to oxidation and corrosion. A lubricant base oil may comprise one or more lubricating base stocks.
[0003] Lubricant base stocks used in automotive engine lubricants are generally obtained from petrochemical sources, for example they may be obtained as the higher boiling fractions isolated during the refining of crude oil or as the products of chemical reactions of feedstocks from petrochemical sources. Lubricant base stocks can also be made from Fischer-Tropsch wax.
[0004] Lubricant base stocks may be classified as Group I, II, III, IV and V base stocks according to API standard 1509, "ENGINE OIL LICENSING AND CERTIFICATION SYSTEM", 17.sup.th Edition, Annex E (October 2013 with Errata March 2015), as set out in Table 1.
TABLE-US-00001 TABLE 1 Saturated Sulphur content hydrocarbon content (% by weight) (% by weight) ASTM D2622, D4294, Viscosity Index Group ASTM D2007 D4927, D3120 or D1552 ASTM D2270 I <90 and/or >0.03 and .gtoreq.80 and <120 II .gtoreq.90 and .ltoreq.0.03 and .gtoreq.80 and <120 III .gtoreq.90 and .ltoreq.0.03 and .gtoreq.120 IV Polyalphaolefins V all base stocks not in Groups I, II, III or IV
[0005] Group I base stocks are typically manufactured by known processes including, for example, solvent extraction and solvent dewaxing, or solvent extraction and catalytic dewaxing. Group II and Group III base stocks are typically manufactured by known processes including, for example, catalytic hydrogenation and/or catalytic hydrocracking, and catalytic hydroisomerisation. Group IV base stocks include for example, hydrogenated oligomers of alpha olefins.
[0006] A combination of properties is desirable in a base stock for conferring to a lubricant composition comprising it. In some instances, for example in passenger car engine oils, it may be desirable for a base stock to confer a low viscosity profile on the lubricant composition, since this leads to improved fuel economy, for instance, as a result of a thinner oil film. In particular, it is desirable for base stocks to have a low kinematic viscosity as well as good low-temperature viscosity characteristics, for example a low pour point or low viscosity as measured using a mini-rotary viscometer (MRV). However, the general trend is for an improvement in the viscosity profile (i.e. a reduction in viscosity parameters) of a base oil to be accompanied by an undesirable increase in volatility. A lower viscosity can also give rise to excess wear resulting in shorter engine life as a result of a thinner oil film for lubricating surfaces of the engine.
[0007] To meet new tougher fuel economy regulations, there has been a move toward building smaller engines equipped with turbochargers. However, it is known that turbochargers, which operate at high temperatures, promote coking related deposit formation which can, amongst other things, lead to scuffing related engine failure. Thus, it is also desirable for lubricant compositions to exhibit good high temperature stability performance so as to reduce high temperature induced deposit formation.
[0008] Accordingly, there is a need for a lubricant composition having low volatility for a given viscosity profile, but which is also suitable for use in an internal combustion engine. There is also a need for a lubricant composition which offers good fuel economy performance together with high temperature stability.
SUMMARY
[0009] Accordingly, in a first aspect the present invention provides a lubricant composition for an internal combustion engine comprising a base oil of lubricating viscosity, wherein the base oil comprises an ether base stock of formula (A):
##STR00002##
[0010] where: R.sub.a and R.sub.b are aliphatic hydrocarbyl groups and may be the same or different; wherein at least one of R.sub.a and R.sub.b is branched-chain alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl; [0011] the lubricant composition further comprising: [0012] i) at least one molybdenum compound as a lubricant additive which is present, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition; or [0013] ii) at least one polymethacrylate compound as a lubricant additive which is present in an amount of from 0.1 to 7.5% by weight of the lubricant composition.
[0014] In a particularly preferred embodiment, the ether base stock of the lubricant composition is selected from a subset of the compounds of formula (A), namely a compound of formula (1):
##STR00003##
[0015] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0016] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; [0017] R.sub.6 is alkyl or
##STR00004##
[0018] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0019] R.sub.9 is H or alkyl; [0020] X is alkylene or is absent; and [0021] p is 0, 1, 2 or 3; and
[0022] m and n are 0, 1, 2 or 3 provided that m is 0 when R.sub.4 and R.sub.5 are H.
[0023] Also provided are methods of preparing lubricant compositions.
[0024] Also provided is a method for lubricating a surface using a lubricant composition, as well as the use of a lubricant composition for lubricating a surface.
[0025] Also provided are methods and uses of improving the high temperature stability of a lubricant composition, reducing or preventing turbocharger deposits or piston scuffing in an engine as well as improving the fuel economy performance and/or turbocharger cleanliness performance of an engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine.
DETAILED DESCRIPTION
[0026] A lubricant composition for an internal combustion engine is provided comprising a base oil of lubricating viscosity, wherein the base oil comprises an ether base stock of formula (A):
##STR00005##
[0027] where: R.sub.a and R.sub.b are aliphatic hydrocarbyl groups and may be the same or different; wherein at least one of R.sub.a and R.sub.b is branched-chain alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl; [0028] the lubricant composition further comprising: [0029] i) at least one molybdenum compound as a lubricant additive which is present, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition; or [0030] ii) at least one polymethacrylate compound as a lubricant additive which is present in an amount of from 0.1 to 7.5% by weight of the lubricant composition.
[0031] For the purposes of the present invention, the following terms as used herein shall, unless otherwise indicated, be understood to have the following meanings.
[0032] The term "aliphatic hydrocarbyl" as used herein refers to a group comprising hydrogen and carbon atoms, where one or more carbon atoms may optionally be replaced with --O--, which group may be saturated or unsaturated, preferably saturated, and contains from 1 to 40 carbon atoms. Examples of hydrocarbyl groups include hydrocarbyl groups containing from 2 to 28 carbon atoms, such as from 3 to 26 carbon atoms or from 4 to 24 carbon atoms. Where one or more of the carbon atoms is replaced with --O--, from 2% to 35% of the carbon atoms are preferably replaced with --O--, or from 5% to 25%. In other examples, the aliphatic hydrocarbyl group has 1 to 3 carbon atoms replaced with --O--, for example 2 carbon atoms replaced with --O--. In other examples, none of the carbon atoms are replaced with --O--.
[0033] Examples of aliphatic hydrocarbyl groups include acyclic groups, non-aromatic cyclic groups and groups comprising both an acyclic portion and a non-aromatic cyclic portion. The aliphatic hydrocarbyl group may be straight chain or branched chain. The aliphatic hydrocarbyl group includes monovalent groups and polyvalent groups as specified. Examples of monovalent hydrocarbyl groups include alkyl, alkenyl, alkynyl and carbocyclyl (e.g. cycloalkyl or cycloalkenyl).
[0034] The term "alkyl" as used herein refers to a monovalent straight or branched chain alkyl moiety containing from 1 to 40 carbon atoms. Examples of alkyl groups include alkyl groups containing from 1 to 30 carbon atoms, e.g. from 2, 3 or 4 carbon atoms to 24, 25, or 26 carbon atoms, e.g. from 1 to 20 carbon atoms, from 1 to 14 carbon atoms, from 2 to 26 carbon atoms and from 3 to 24 carbon atoms. Particular examples include alkyl groups containing 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29 and 30 carbon atoms. Examples of alkyl groups include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl and the like. Unless specifically indicated otherwise, the term "alkyl" does not include optional substituents.
[0035] The term "cycloalkyl" as used herein refers to a monovalent saturated aliphatic hydrocarbyl moiety containing from 3 to 40 carbon atoms and containing at least one ring, wherein said ring has at least 3 ring carbon atoms. The cycloalkyl groups mentioned herein may optionally have alkyl groups attached thereto. Examples of cycloalkyl groups include cycloalkyl groups containing from 3 to 16 carbon atoms, e.g. from 3 to 10 carbon atoms. Particular examples include cycloalkyl groups containing 3, 4, 5 or 6 ring carbon atoms. Examples of cycloalkyl groups include groups that are monocyclic, polycyclic (e.g. bicyclic) or bridged ring system. Examples of cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and the like.
[0036] The term "alkenyl" as used herein refers to a monovalent straight or branched chain alkyl group containing from 2 to 40 carbon atoms and containing, in addition, at least one carbon-carbon double bond, of either E or Z configuration unless specified. Examples of alkenyl groups include alkenyl groups containing from 2 to 28 carbon atoms, e.g. from 3 to 26 carbon atoms, e.g. from 4 to 24 carbon atoms. Particular examples include alkenyl groups containing 2, 3, 4, 5 or 6 carbon atoms. Examples of alkenyl groups include ethenyl, 2-propenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl and the like.
[0037] The term "alkylene" refers to a divalent straight or branched chain saturated hydrocarbyl group consisting of hydrogen and carbon atoms and containing from 1 to 30 carbon atoms. Examples of alkylene groups include alkylene groups that contain from 1 to 20 carbon atoms, e.g. from 1 to 12 carbon atoms, e.g. from 1 to 10 carbon atoms. Particular examples include alkylene groups that contain 1, 2, 3, 4, 5 or 6 carbon atoms.
[0038] The term "alkoxy" as used herein refers to --O-alkyl, wherein alkyl is as defined herein. In some examples an alkoxy group contains from 1 to 40 carbon atoms, e.g. from 1 to 28 carbon atoms, or from 1 to 26 carbon atoms, or from 1 to 24 carbon atoms e.g. from 1 to 10 carbon atoms. Particular examples include alkoxy groups that contain 1, 2, 3, 4, 5 or 6 carbon atoms. Examples of alkoxy groups include methoxy, ethoxy, propoxy, isopropoxy, butoxy, tert-butoxy, pentoxy, hexoxy and the like.
[0039] The terms "alkoxy-substituted-alkyl" and "cycloalkyl-substituted-alkyl" refer to a straight or branched chain alkyl group in which one of the hydrogens of the alkyl chain is replaced with an alkoxy or cycloalkyl group as described herein, respectively.
[0040] In some embodiments, R.sub.a and R.sub.b of formula (A) are independently selected from alkyl, alkoxy-substituted-alkyl and cycloalkyl-substituted-alkyl, provided that where R.sub.a and R.sub.b are both alkyl at least one of R.sub.a and R.sub.b is branched-chain alkyl. In preferred embodiments, when R.sub.a and R.sub.b are both alkyl, both R.sub.a and R.sub.b are branched-chain alkyl.
[0041] In some embodiments, R.sub.a and R.sub.b of formula (A) are independently selected from C.sub.1-30 alkyl, such as C.sub.2-20 alkyl, C.sub.5-30 cycloalkyl-substituted-alkyl, such as C.sub.5-25 cycloalkyl-substituted-alkyl, or C.sub.2-30 alkoxy-substituted-alkyl, such as C.sub.2-20 alkoxy-substituted-alkyl.
[0042] In some embodiments, R.sub.a of formula (A) contains more carbon atoms than R.sub.b.
[0043] In some embodiments, R.sub.a of formula (A) contains from 12 to 30 carbon atoms, preferably from 12 to 26 carbon atoms, and/or R.sub.b contains from 2 to 20 carbon atoms, preferably from 2 to 12 carbon atoms.
[0044] In particularly preferred embodiments, the ether base stock of the lubricant composition is a compound of formula (1):
##STR00006##
[0045] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl;
[0046] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl;
[0047] R.sub.6 is alkyl or
##STR00007##
[0048] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0049] R.sub.9 is H or alkyl; [0050] X is alkylene or is absent; and [0051] p is 0, 1, 2 or 3; and
[0052] m and n are 0, 1, 2 or 3 provided that m is 0 when R.sub.4 and R.sub.5 are H.
[0053] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl.
[0054] In some embodiments, R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0055] In some embodiments, R.sub.6 is C.sub.1-20 alkyl or
##STR00008##
such as C.sub.1-16 alkyl or
##STR00009##
[0056] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0057] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0058] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0059] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0060] In some embodiments, m and n are 0, 1 or 2, such as 0 or 1.
[0061] R.sub.1 and R.sub.2 are as described as alkyl or, together with the carbon atom to which they are attached, cycloalkyl. It will be understood that, where R.sub.1 and R.sub.2 are both alkyl groups, they may be the same as or different from one another. Similar considerations apply to other substituents which are defined as part of a group of substituents. Thus, the considerations apply, for example, to R.sub.3, R.sub.4 and R.sub.5; to R.sub.7 and R.sub.8; and to the values taken by m and n. For instance, where R.sub.3, R.sub.4 and R.sub.5 are described as being H or alkyl, it will be understood that each of R.sub.3, R.sub.4 and R.sub.5 may be H, each of R.sub.3, R.sub.4 and R.sub.5 may be alkyl, or a subset of R.sub.3, R.sub.4 and R.sub.5 may be H and another subset of R.sub.3, R.sub.4 and R.sub.5 may be alkyl. Where R.sub.3, R.sub.4 and R.sub.5, or a subset thereof, are alkyl, each of R.sub.3, R.sub.4 and R.sub.5 may be the same alkyl group or they may be different alkyl groups. In contrast, where R.sub.1 (or any other notation) is used at a number of locations in a formula, it is used to denote the presence of the same group at each of these locations.
[0062] In each of the embodiments disclosed herein, the ether compounds of the lubricant compositions may contain a total number of carbons atoms of from about 20 to about 50. For instance, the total number of carbons in the ether compounds may be from about 25 to about 45, such as from about 28 to about 40 or from about 28 to about 36.
[0063] As indicated previously, the alkyl and alkylene groups mentioned herein, i.e. those that may be represented by R.sub.a, R.sub.b, R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.5, R.sub.6, R.sub.7, R.sub.8, R.sub.9 and X, may be straight chain alkyl or alkylene groups, though they may also be branched. In some embodiments, each alkyl group and each alkylene group contains a single branch point or is a straight chain alkyl or alkylene group. For example, when R.sub.a and R.sub.b are both alkyl groups, at least one of these alkyl groups is branched, preferably both. In some embodiments, for instance with respect to R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.5, R.sub.6, R.sub.7, R.sub.8, R.sub.9 and X groups, the alkyl and alkylene groups are straight chain alkyl or alkylene groups. It will be understood that, aside from alkyl branching (if present), the alkyl and alkylene groups are unsubstituted unless otherwise indicated and so may not contain any atoms other than carbon or hydrogen.
[0064] The ether compounds described herein may be used for improving the high temperature stability of a lubricant composition, for reducing or preventing scuffing in the piston system of an engine or for reducing or preventing deposits in the turbochargers or other hot surfaces, for example piston undercrowns, of an engine. The presence of a molybdenum compound or polymethacrylate compound as defined herein, as well as phenate and/or neutral sulphonate detergents, in the lubricant compositions of the invention have also been found to enhance high temperature stability in the ether based lubricant compositions and/or be better tolerated in terms of high temperature stability in ether based compositions as defined herein in comparison with conventional non-ether based lubricant compositions.
[0065] Accordingly, the lubricant compositions of the invention may also be used for reducing or preventing scuffing in the pistons of an engine or for reducing or preventing deposits in the turbochargers or other hot surfaces of an engine. There is also provided the use of the lubricant compositions of the invention for improving the fuel economy performance and/or piston cleanliness performance and/or turbocharger cleanliness performance of an engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine.
[0066] Accordingly, there is also provided a method of reducing or preventing i) scuffing in the pistons of an engine and/or ii) deposits in the turbochargers of an engine, comprising the step of providing to the engine a lubricant composition as described herein. There is also provided a method of improving the fuel economy performance and/or piston cleanliness performance and/or turbocharger cleanliness performance of an engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine comprising the step of providing the engine and/or the vehicle with a lubricant composition as described herein.
[0067] The compounds of formula (A) and/or formula (1) may have a kinematic viscosity at 40.degree. C. of less than about 25 cSt, such as less than about 20 cSt, or less than about 17 cSt.
[0068] The compounds may have a kinematic viscosity at 100.degree. C. of less than about 7 cSt, such as less than about 5 cSt, or less than about 4 cSt. The compounds may have a viscosity index of greater than about 100, such as greater than about 110, or greater than about 120. The kinematic viscosity at 40.degree. C. and the kinematic viscosity at 100.degree. C. may be measured according to ASTM D7279. The viscosity index may be measured according to ASTM D2270.
[0069] The compounds may have a Noack volatility of less than about 26%, such as less than about 20%, less than about 16%, or less than about 12% by weight. Noack volatility may be measured according to CEC-L-40-A-93.
[0070] The compounds may have a viscosity at 150.degree. C. and a shear rate of 10.sup.6 s.sup.-1 of no greater than 1.7 cP, such as no greater than 1.5 cP. This high temperature high shear viscosity may be measured according to CEC-L-36-A-90.
[0071] The ether compounds described herein may have a pour point of less than -10.degree. C., such as less than about -25.degree. C., or less than about -35.degree. C. Pour point may be measured according to ASTM D5950.
[0072] The ether compounds may have a cold-crankcase simulator viscosity at -35.degree. C. of less than about 1800 cP, such as less than about 1500 cP, or less than about 1200 cP, for example as measured according to ASTM D5293.
[0073] The ether compounds may have a DSC oxidation onset temperature of greater than about 165.degree. C., such as greater than about 175.degree. C., or greater than about 185.degree. C., for example as measured according to ASTM E2009 (method B).
[0074] In particular embodiments, the ether compounds of formula (A) or formula (1) may have a kinematic viscosity at 100.degree. C. of about 3 to about 4 cSt and a Noack volatility of less than about 20%, such as less than about 16%, or less than about 12%, by weight; or a kinematic viscosity at 100.degree. C. of about 2 to about 3 cSt, and a Noack volatility of less than about 40%, such as less than about 30%, by weight.
[0075] The ether compounds of formula (A) or formula (1) are particularly suited for blending into a lubricant composition. In particular, the compounds are miscible with conventional base stocks, including hydrocarbon base stocks, as well as with conventional lubricant additives. Moreover, the compounds may be used in a lubricant composition in a relatively high amount (for example, in an amount of greater than about 10% by weight, such as greater than about 20% by weight or greater than about 30% by weight) whilst meeting elastomer compatibility requirements for lubricant compositions.
[0076] The compounds of formula (A) and formula (1) may be prepared from a wide range of commercially available feedstocks.
[0077] In some embodiments, the compounds are prepared from bio-derived feedstocks. For instance, the compounds may contain greater than about 50%, such as greater than about 70%, or greater than about 80% by weight of biobased carbon. The biobased carbon content of the compounds may be measured according to ASTM D6866.
Guerbet-Derived Base Stocks
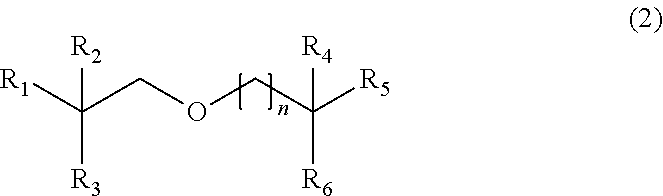
[0078] In preferred embodiments, the compounds of formula (1) are derived from .beta.-alkylated alcohols. In these embodiments, the compound may have the formula (2):
##STR00010##
[0079] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0080] R.sub.3 and R.sub.5 are H or alkyl; [0081] R.sub.4 is alkyl; [0082] R.sub.6 is alkyl or
##STR00011##
[0083] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0084] R.sub.9 is H or alkyl; [0085] X is alkylene or is absent; and [0086] p is 0, 1, 2 or 3; and
[0087] n is 0, 1, 2 or 3.
[0088] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0089] In some embodiments, R.sub.3 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.3 and R.sub.5 are H.
[0090] In some embodiments, R.sub.4 is C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0091] In some embodiments, R.sub.6 is C.sub.1-15 alkyl or
##STR00012##
such as
[0092] C.sub.1-12 alkyl or
##STR00013##
[0093] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0094] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0095] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0096] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0097] In some embodiments, n is 0, 1 or 2, such as 0 or 1.
[0098] Where the compound is derived from a .beta.-alkylated alcohol, it is preferably derived, at least in part, from a Guerbet alcohol. Compounds which are derived, at least in part, from Guerbet alcohols may have the formula (3):
##STR00014##
[0099] where: R.sub.1 is alkyl;
[0100] R.sub.3 and R.sub.5 are H or alkyl;
[0101] R.sub.4 is alkyl;
[0102] R.sub.6 is alkyl or
##STR00015##
[0103] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0104] R.sub.9 is H or alkyl; [0105] X is alkylene or is absent; and [0106] p is 0, 1, 2 or 3; and
[0107] n is 0, 1, 2 or 3.
[0108] In some embodiments, R.sub.1 is C.sub.1-12 alkyl, such as C.sub.2-10 alkyl.
[0109] In some embodiments, R.sub.3 is H or C.sub.1-12 alkyl, such as H or C.sub.2-10 alkyl. Preferably, R.sub.3 is H.
[0110] In some embodiments, R.sub.4 is C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0111] In some embodiments, R.sub.5 is H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0112] In some embodiments, R.sub.6 is C.sub.1-15 alkyl or
##STR00016##
such as C.sub.1-12 alkyl or
##STR00017##
Preferably, R.sub.6 is C.sub.1-15 alkyl, such as C.sub.1-12 alkyl.
[0113] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0114] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0115] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0116] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0117] In some embodiments, n is 0, 1 or 2, such as 0 or 1.
[0118] One portion of the compound of formula (3) has a structure which may be derived from a Guerbet alcohol (i.e. the portion containing R.sub.1 and R.sub.3), whereas the other portion need not be derived from a Guerbet alcohol (i.e. the portion containing R.sub.4, R.sub.5 and R.sub.6). However, in preferred embodiments, the compound may be derived from a combination of two Guerbet alcohols. A compound prepared in this way may have the formula (4):
##STR00018##
[0119] where: R.sub.1 and R.sub.4 are alkyl; [0120] R.sub.3 and R.sub.5 are H or alkyl.
[0121] In some embodiments, R.sub.1 and R.sub.4 are C.sub.1-12 alkyl, such as C.sub.2-10 alkyl.
[0122] In some embodiments, R.sub.3 and R.sub.5 are H or C.sub.1-12 alkyl, such as H or C.sub.2-10 alkyl. Preferably, R.sub.3 and R.sub.5 are H.
[0123] In particular embodiments: R.sub.1 is C.sub.4-12 alkyl, such as C.sub.6-10 alkyl; [0124] R.sub.3 is H; [0125] R.sub.4 is C.sub.1-10 alkyl, such as C.sub.2-8 alkyl; and [0126] R.sub.5 is H.
[0127] Two different Guerbet alcohols may be combined to form compounds of formula (4), in which case R.sub.1 and R.sub.4 may be different. Alternatively, R.sub.3 and R.sub.5 may be different. In some embodiments, R.sub.1 and R.sub.4 are different and R.sub.3 and R.sub.5 are also different.
[0128] However, in some embodiments, the compound may be derived from a reaction in which the same Guerbet alcohols are combined. A compound prepared in this way may have the formula (5):
##STR00019##
[0129] where: R.sub.1 is alkyl; and [0130] R.sub.3 is H or alkyl.
[0131] In some embodiments, R.sub.1 is C.sub.1-10 alkyl, such as C.sub.2-9 alkyl.
[0132] In some embodiments, R.sub.3 is H or C.sub.1-0 alkyl, such as H or C.sub.2-8 alkyl. Preferably, R.sub.3 is H.
[0133] In particular embodiments: R.sub.1 is C.sub.3-10 alkyl, such as C.sub.4-8 alkyl; and R.sub.3 is H.
[0134] Compounds that are derived from Guerbet alcohols include compounds GE1-GE3, GES, GE7-GE9, SE1, SE2 and TE1 as shown in Table 2.
[0135] Guerbet alcohols may be prepared, for example, by dimerising primary alcohols to form a .beta.-alkylated alcohol product in a Guerbet reaction:
##STR00020##
[0136] where R.sub.1 and R.sub.3 are as defined previously;
[0137] and/or:
##STR00021##
[0138] where R.sub.4 and R.sub.5 are as defined previously.
[0139] Guerbet reactions are well-known to the skilled person. The reactions are typically carried out at elevated temperatures in the presence of a catalyst.
[0140] The compound may be prepared from the Guerbet alcohol, for example, according to the following reaction:
##STR00022##
[0141] where: Y is a leaving group; and
[0142] R.sub.1, R.sub.3, R.sub.4, R.sub.5, R.sub.6 and n are as defined previously for the compound of formula (3).
[0143] Where two Guerbet alcohols are combined to form a compound, one of the Guerbet alcohols may first be modified so that it contains a leaving group, Y, and the compound then prepared:
##STR00023##
then:
##STR00024##
or
##STR00025##
then:
##STR00026##
[0144] where: Y is a leaving group; and
[0145] R.sub.1, R.sub.3, R.sub.4 and R.sub.5 are as defined previously for the compound of formula (4).
[0146] Where the same Guerbet alcohols are combined to form a compound, they may be combined, for example, according to the following reactions:
##STR00027##
then:
##STR00028##
[0147] where: Y is a leaving group; and [0148] R.sub.1 and R.sub.3 are as defined previously for the compound of formula (5).
[0149] Methods and reaction conditions for modifying a Guerbet alcohol so that it contains a leaving group, Y, are known to the skilled person. For instance, a mesylate group may be introduced by reacting the Guerbet alcohol with mesyl chloride in the presence of triethylamine. A bromide group may be introduced by reacting the Guerbet alcohol with N-bromosuccinimide and triphenyl phosphine.
[0150] Methods and reaction conditions for carrying out etherification reactions are known to the skilled person. A base (for example potassium hydroxide or potassium tert-butoxide), a catalyst (for example Starks' catalyst: N-Methyl-N,N,N-trioctyloctan-1-ammonium chloride) or both may be used in the abovementioned compound forming reactions, i.e. the etherification reactions.
[0151] In the abovementioned compound forming reactions, Y may be any suitable leaving group, such as a halogen (for example bromine, chlorine or iodine) or a sulfonate ester (for example mesylate or tosylate).
Secondary and Tertiary Ether Base Stocks
[0152] In some preferred embodiments, the compounds of formula (1) are secondary or tertiary ether compounds. In these embodiments, the compound may have the formula (6):
##STR00029##
[0153] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon to which they are attached, cycloalkyl; [0154] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl;
[0155] R.sub.6 is alkyl or
##STR00030##
[0156] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0157] R.sub.9 is H or alkyl; [0158] X is alkylene or is absent; and [0159] p is 0, 1, 2 or 3; and
[0160] n is 0, 1, 2 or 3.
[0161] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0162] In some embodiments, R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0163] In some embodiments, R.sub.6 is C.sub.1-20 alkyl or
##STR00031##
such as C.sub.1-16 alkyl or
##STR00032##
[0164] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0165] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0166] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0167] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0168] In some embodiments, n is 0, 1 or 2, such as 0 or 1.
[0169] Secondary and tertiary ether compounds may have the formula (7):
##STR00033##
[0170] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon to which they are attached, cycloalkyl; [0171] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; and [0172] R.sub.6 is alkyl.
[0173] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon to which they are attached, C.sub.5-25 cycloalkyl.
[0174] In some embodiments, R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0175] In some embodiments, R.sub.6 is C.sub.1-20 alkyl, such as C.sub.1-16 alkyl.
[0176] The compounds may be secondary ether compounds of formula (8):
##STR00034##
[0177] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon to which they are attached, cycloalkyl; [0178] R.sub.4 and R.sub.5 are H or alkyl; and [0179] R.sub.6 is alkyl.
[0180] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0181] In other embodiments, the secondary ether may be obtained from a cyclic compound. In this case, R.sub.1 and R.sub.2, together with the carbon to which they are attached, form a cycloalkyl group, such as a C.sub.5-30 cycloalkyl or a C.sub.5-25 cycloalkyl. The cycloalkyl group may contain a cyclopentyl, cyclohexyl or cycloheptyl group optionally having one or more alkyl groups, such as C.sub.1-12 alkyl or C.sub.1-8 alkyl, attached thereto.
[0182] In some embodiments, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0183] In some embodiments, R.sub.6 is C.sub.1-20 alkyl, such as C.sub.1-16 alkyl.
[0184] In particular embodiments: R.sub.1 and R.sub.2 are C.sub.3-12 alkyl, such as C.sub.5-10 alkyl; [0185] R.sub.4 and R.sub.5 are H; and [0186] R.sub.6 is C.sub.4-20 alkyl, such as C.sub.6-15 alkyl.
[0187] In other particular embodiments: R.sub.1 and R.sub.2 are C.sub.3-12 alkyl, such as C.sub.5-10 alkyl; [0188] R.sub.4 is C.sub.3-12 alkyl, such as C.sub.5-10 alkyl; [0189] R.sub.5 is H; and [0190] R.sub.6 is C.sub.3-12 alkyl, such as C.sub.5-10 alkyl.
[0191] The compounds may be tertiary ether compounds of formula (9):
##STR00035##
[0192] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon to which they are attached, cycloalkyl; [0193] R.sub.3 is alkyl; [0194] R.sub.4 and R.sub.5 are H or alkyl; and [0195] R.sub.6 is alkyl.
[0196] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0197] In some embodiments, R.sub.3 is C.sub.1-12 alkyl, such as C.sub.1-10 alkyl.
[0198] In some embodiments, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl.
[0199] In some embodiments, R.sub.6 is C.sub.1-20 alkyl, such as C.sub.1-16 alkyl.
[0200] In particular embodiments: R.sub.1 and R.sub.2 are C.sub.2-12 alkyl, such as C.sub.4-10 alkyl; [0201] R.sub.3 is C.sub.1-10 alkyl, such as C.sub.1-8 alkyl; [0202] R.sub.4 and R.sub.5 are H; and [0203] R.sub.6 is C.sub.4-20 alkyl, such as C.sub.6-15 alkyl.
[0204] In other particular embodiments: R.sub.1, R.sub.2 and R.sub.3 are C.sub.2-12 alkyl, such as C.sub.4-10 alkyl; [0205] R.sub.3 is C.sub.1-10 alkyl, such as C.sub.1-8 alkyl; [0206] R.sub.4 is C.sub.3-12 alkyl, such as C.sub.5-10 alkyl; [0207] R.sub.5 is H; and [0208] R.sub.6 is C.sub.3-12 alkyl, such as C.sub.5-10 alkyl.
[0209] Examples of secondary and tertiary ether compounds include SE1, SE2 and TE1 as shown in Table 2.
[0210] The secondary and tertiary ether compounds may be prepared according to the following reactions:
##STR00036##
or:
##STR00037##
[0211] where: Y is a leaving group; and [0212] R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.5, R.sub.6 and n are as defined previously for the compound of formula (6).
[0213] Similarly:
##STR00038##
or:
##STR00039##
[0214] where: Y is a leaving group; and [0215] R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.5 and R.sub.6 are as defined previously for the compound of formula (7).
[0216] The skilled person will be aware of methods and reaction conditions for carrying out these etherification reactions. For instance, the reaction may be carried out in the presence of magnesium sulfate, sulfuric acid and dichloromethane.
[0217] Secondary and tertiary alcohol starting materials for use in etherification reactions will generally be commercially available, or they may be obtained from commercially available ketones.
[0218] The groups
##STR00040##
may be prepared by introducing a leaving group, Y, into the alcohol starting materials. Methods and reaction conditions for introducing the leaving group into alcohol are known to the skilled person.
[0219] In the abovementioned secondary and tertiary ether compound forming reactions, Y may be any suitable leaving group, such as a halogen (for example bromine, chlorine or iodine) or a sulfonate ester (for example mesylate or tosylate).
Secondary or Tertiary Ethers Derived from a Guerbet Alcohol
[0220] In some embodiments, the compound may comprise an ether which is derived on one side from a secondary or tertiary alcohol and is derived on the other side from a Guerbet alcohol. In these embodiments, the compound may have the formula (10):
##STR00041##
[0221] where: R.sub.1 and R.sub.4 are alkyl; [0222] R.sub.3 and R.sub.5 are H or alkyl;
[0223] R.sub.6 is alkyl or
##STR00042##
[0224] where: R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0225] R.sub.9 is H or alkyl; [0226] X is alkylene or is absent; and [0227] and p is 0, 1, 2 or 3.
[0228] In some embodiments, R.sub.1 is C.sub.1-12 alkyl, such as C.sub.2-10 alkyl.
[0229] In some embodiments, R.sub.3 is H or C.sub.1-12 alkyl, such as H or C.sub.2-10 alkyl. Preferably, R.sub.3 is H.
[0230] In some embodiments, R.sub.4 is C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0231] In some embodiments, R.sub.5 is H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.5 is H.
[0232] In some embodiments, R.sub.6 is C.sub.1-15 alkyl or
##STR00043##
such as C.sub.1-12 alkyl or
##STR00044##
[0233] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0234] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0235] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0236] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0237] Examples of secondary and tertiary ether compounds derived from a Guerbet-alcohol include compounds SE1, SE2 and TE1 as shown in Table 2.
Di-Ether Base Stocks
[0238] It is generally preferred that the compounds of formula (1) are monoethers. However, in some embodiments, the compound is a diether compound. Such compounds may have the formula (11):
##STR00045##
[0239] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0240] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; [0241] R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0242] R.sub.9 is H or alkyl; [0243] X is alkylene or is absent; [0244] p is 0, 1, 2 or 3; and [0245] m and n are 0, 1, 2 or 3.
[0246] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0247] In some embodiments, R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.3 and R.sub.5 are H.
[0248] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0249] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0250] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0251] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0252] In some embodiments, m and n are 0, 1 or 2, such as 0 or 1.
[0253] In some embodiments, the diether compound may contain two ether groups, at least one of which is derived from a .beta.-alkylated alcohol. In such embodiments, the compound may have the formula (12):
##STR00046##
[0254] where: R.sub.1 and R.sub.2 are alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0255] R.sub.3, R.sub.4 and R.sub.5 are H or alkyl; [0256] R.sub.7 and R.sub.8 are H, alkyl or, together with the carbon atom to which they are attached, cycloalkyl; [0257] R.sub.9 is H or alkyl; [0258] X is alkylene or is absent; [0259] p is 0, 1, 2 or 3; and [0260] n is 0, 1, 2 or 3.
[0261] In some embodiments, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.1 and R.sub.2 are C.sub.1-15 alkyl, such as C.sub.2-12 alkyl.
[0262] In some embodiments, R.sub.3, R.sub.4 and R.sub.5 are H or C.sub.1-15 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.3 and R.sub.5 are H. Preferably, R.sub.4 is C.sub.1-15 alkyl, such as C.sub.2-12 alkyl
[0263] In some embodiments, R.sub.7 and R.sub.8 are H, C.sub.1-20 alkyl or, together with the carbon atom to which they are attached, C.sub.5-30 cycloalkyl, such as H, C.sub.2-12 alkyl or, together with the carbon atom to which they are attached, C.sub.5-25 cycloalkyl. Preferably, R.sub.7 and R.sub.8 are C.sub.1-20 alkyl, such as C.sub.2-12 alkyl.
[0264] In some embodiments, R.sub.9 is H or C.sub.1-20 alkyl, such as H or C.sub.2-12 alkyl. Preferably, R.sub.9 is H.
[0265] In some embodiments, X is C.sub.1-20 alkylene, such as C.sub.3-15 alkylene.
[0266] In some embodiments, p is 0, 1 or 2, such as 0 or 1.
[0267] In some embodiments, n is 0, 1 or 2, such as 0 or 1.
[0268] Examples of Guerbet-derived base stocks GE1-GE9, secondary ether base stocks SE1 and SE2, and tertiary ether base stock TE1 of formula (1), which may preferably be used in connection with the present application, are shown in Table 2.
TABLE-US-00002 TABLE 2 Molecular Chemical Weight Formula Structure GE1 466.87 C.sub.32H.sub.66O ##STR00047## GE2 466.87 C.sub.32H.sub.66O ##STR00048## GE3 522.97 C.sub.36H.sub.74O ##STR00049## GE4 466.87 C.sub.32H.sub.66O ##STR00050## GE5 410.76 C.sub.28H.sub.58O ##STR00051## GE6 466.87 C.sub.32H.sub.66O ##STR00052## GE7 522.57 C.sub.36H.sub.74O ##STR00053## GE8 382.42 C.sub.26H.sub.54O ##STR00054## GE9 466.51 C.sub.32H.sub.66O ##STR00055## GE10 410.76 C.sub.28H.sub.58O ##STR00056## GE12 382.71 C.sub.26H.sub.54O ##STR00057## GE14 410.76 C.sub.28H.sub.58O ##STR00058## GEIS 354.65 C.sub.24H.sub.50O ##STR00059## GE16 424.79 C.sub.29H.sub.60O ##STR00060## GE18 438.81 C.sub.30H.sub.62O ##STR00061## GE20 354.65 C.sub.24H.sub.50O ##STR00062## GE21 382.71 C.sub.26H.sub.54O ##STR00063## GE22 410.76 C.sub.28H.sub.58O ##STR00064## GE23 382.71 C.sub.26H.sub.54O ##STR00065## SE1 452.84 C.sub.31H.sub.64O ##STR00066## SE2 396.43 C.sub.27H.sub.56O ##STR00067## TE1 466.87 C.sub.32H.sub.66O ##STR00068##
Base Oils and Lubricant Compositions
[0269] The ether compounds of formula (A), or the subset thereof of formula (1), are used as part of a base oil in accordance with the present invention.
[0270] The base oils may contain an amount of compound of formula (A), or a compound of the subset thereof of formula (1), which is sufficient to impart beneficial properties of the compound onto the base oil.
[0271] In some embodiments, the base oil comprises greater than about 5%, such as greater than about 25%, greater than about 40%, or greater than 50% by weight of ether compound of formula (A), or the subset thereof of formula (1). The base oil may comprise up to about 100%, such as up to about 90% of compound of formula (A), or of the subset thereof of formula (1). The compound of formula (A), or of the subset thereof of formula (1), in the base oil may be composed of a single compound or a combination of compounds of formula (A), or of the subset thereof of formula (1).
[0272] The remainder of the base oil may be made up with base stocks which are not compounds of formula (A) and formula (1). Base stocks other than those of formula (A) and formula (1) which are suitable for use in the base oil include non-aqueous base stocks, such as Group I, Group II, Group III, Group IV and Group V base stocks. The remainder of the base oil may comprise a single base stock or a combination of base stocks other than those of formula (A) and formula (1).
The Base Oils are Used as Part of the Lubricant Composition in Accordance with the Present Invention.
[0273] The lubricant compositions may contain an amount of base oil which is sufficient to impart beneficial properties of the compound of formula (A), or a compound of the subset thereof of formula (1), onto the lubricating composition.
[0274] In some embodiments, the lubricant composition comprises greater than about 50%, such as greater than about 65%, or greater than about 80% by weight of base oil. The base oil may be composed of a single base oil or a combination of base oils comprising compound of formula (A), or of the subset thereof of formula (1).
[0275] A particular advantage of the present invention relates to the high temperature stability conferred to the lubricant composition by the presence of ether compounds of formula (A), or of the subset thereof of formula (1). The presence of at least one molybdenum compound or at least one polymethacrylate compound is better tolerated in the ether based compositions of the invention compared to conventional non-ether compositions from a high temperature stability perspective. Thus, the at least one molybdenum compound or at least one polymethacrylate compound may be used to enhance high temperature stability in the lubricant compositions defined herein, or provide desirable properties to the composition without impacting the high temperature stability of the lubricant composition. For instance, molybdenum compounds are well known to those skilled in the art of oil formulation to function as friction modifiers to lower engine friction and promote fuel economy. However, too high a level of molybdenum in conventional non-ether compositions can contribute to deposits which can lead to excess wear and shorten engine life. Similarly, polymethacrylates are known for use as viscosity index improvers as well as pour point depressants, but their use beyond a certain amount can lead to unwanted deposit formation on hot surfaces of the engine, for example turbochargers. The present invention allows the benefits of molybdenum or polymethacrylate compounds to be taken advantage of without lessening high temperature stability of the lubricant composition to the same extent as in conventional non-ether based compositions and the presence of molybdenum or polymethacrylate compounds has been found to enhance high temperature stability in some cases.
[0276] Particular problems associated with a lack of high temperature stability is engine scuffing, in particular, piston scuffing and deposits in high temperature regions of the engine, particularly in the turbochargers. ASTM Terminology standard G40 defines scuffing as a form of wear occurring in inadequately-lubricated tribosystems that is characterized by macroscopically observable changes in texture, with features related to the direction of motion. Engine scuffing is intrinsically linked with the presence of deposits in high temperature regions of the engine and therefore scuffing can be impacted by the choice of lubricant oil that is used for lubricating the engine. Where reference is made herein to piston scuffing or piston system scuffing, it will be appreciated that this refers to scuffing on the piston ring, skirt or cylinder liner.
[0277] Whether or not an engine oil is likely to give rise to engine scuffing may be determined by means of the Komatsu hot-tube test (KHT), which corresponds to standard method, JPI-5S-55-99. The KHT test evaluates the high temperature stability of a lubricant and is described in detail in: Ohkawa, S., Seto, K., Nakashima, T., and Takase, K., ""Hot Tube Test"-Analysis of Lubricant Effect on Diesel Engine Scuffing," SAE Technical Paper 840262, 1984, doi:10.4271/840262. In the KHT test, droplets of a candidate oil are forced by air up inside a heated narrow glass capillary tube and the thin film oxidative stability of the lubricant is measured by the degree of lacquer formation on the glass tube, the resulting colour of the tube being rated on a scale of 0 to 10.
[0278] The results of the KHT test correspond to deposit merit ratings which can be related to engine scuffing as described in the above paper. A deposit merit rating of 0 refers to heavy deposit formation whilst a deposit merit rating of 10 means a clean glass tube at the end of the test. The level of lacquer formation in the tube reflects the high temperature stability of the oil and its tendency during service to form deposits in high temperature regions of the engine, thereby causing scuffing. Engine oils which pass the KHT test will have lower propensity to cause scuffing engine failure in Heavy Duty applications from scuffing as a result of deposits.
[0279] The KHT test also evaluates the `hot surface deposit control`, which includes surfaces associated with turbochargers, for JASO engine oil specifications (DH-1-05, DH-2-08, DH-1-08, available in document JASO M 355:2008). `Engine Failure Analysis: Internal Combustion Engine Failures and Their Causes` by Ernst Greuter and Stefan Zima, published by SAE International, 2012, page 493, ISBN 978-0-7860-0885-2, also describes carbon deposit formation on turbochargers as a result of hot surface deposit formation and supports the connection between hot surface deposits and engine scuffing which underlies the KHT test.
[0280] The Thermo-oxidation Engine oil Simulation Test (TEOST) 33C, which corresponds to standard method ASTM D6335, is a bench test which simulates the oxidation and carbonaceous deposit-forming characteristics of engine oils in the turbochargers of modern high-performance engines. The TEOST 33C test generates physical measurements of deposits and represents another test through which to determine an oil's susceptibility to deposit formation in the turbochargers specifically. Hot Liquid Process Simulator (HLPS) testing may also be used as a means for characterising the propensity of an oil to create deposits in an engine by running oil over a hot surface.
[0281] Lubricant compositions according to the present invention have been found to outperform corresponding conventional non-ether based compositions in KHT and HLPS testing without adversely affecting the TEOST 33C, indicating that the ether compositions of the invention have greater high temperature stability than conventional lubricant compositions differing only in the absence of ether base stock. Moreover, the presence of at least one molybdenum compound or at least one polymethacrylate compound in accordance with the present invention is better tolerated in the ether compositions of the present invention than in corresponding conventional non-ether based compositions.
[0282] The lubricant composition according to the invention comprises the at least one molybdenum compound, on a molybdenum element basis, in an amount of at least 0.06% by weight of the lubricant composition, when present. Alternatively, the lubricant composition according to the invention comprises the at least one polymethacrylate compound in an amount of from 0.1 to 7.5% by weight of the lubricant composition, when present.
[0283] In preferred embodiments, when the lubricant composition comprises at least one molybdenum compound as a lubricant additive, the at least one molybdenum compound is present, on a molybdenum element basis, in an amount from 0.06% to 0.25%, from 0.075% to 0.175%, or from 0.075% to 0.125%, by weight of the lubricant composition. The amount of molybdenum element present in the lubricant compositions of the invention may, for example, be determined according to method ASTM D5185.
[0284] In preferred embodiments, when the lubricant composition comprises at least one polymethacrylate compound as a lubricant additive, the at least one polymethacrylate compound is present in an amount of from 0.25 to 7%, from 1 to 6%, from 2 to 4% by weight of the lubricant composition.
[0285] In some embodiments, the lubricant composition comprises at least one molybdenum compound. Any molybdenum compound may be used which is suitable for use as an additive for a lubricant composition intended for use in an internal combustion engine. As will be appreciated, the term "molybdenum compound" used herein refers to a molybdenum-containing compound or complex, which has oil-solubility or oil-dispersibility properties. Molybdenum compounds for use in the lubricant compositions of the present invention include organo molybdenum compounds, molybdenum dialkyldithiocarbamates, molybdenum dialkylthiophosphates, molybdenum disulphide, tri-molybdenum cluster dialkyldithiocarbamates, non-sulphur molybdenum compounds and the like. Suitable molybdenum-containing compounds are described for example, in EP 1533362 A1, for example, in paragraphs [0101] to [0117]. Certain molybdenum compounds of these classes are well known to have friction modifier properties in lubricant compositions.
[0286] Particularly preferred molybdenum compounds for use in the present invention are molybdenum-sulfur compounds, particularly tri-nuclear molybdenum-sulfur cluster compounds as, for instance, described in EP 1 040 115 and WO 99/31113. Further, examples of trinuclear molybdenum-sulfur compounds are also disclosed in WO98/26030, WO99/31113, WO99/66013, EP 1 138 752 and EP 1 138 686. As the skilled person will be aware, such compounds may be added to a base oil fully formed or such compounds may be formed in situ as a result of the presence of sulfur-containing compounds or complexes (e.g. ZDDP), for example by means of ligand exchange.
[0287] In some embodiments, the lubricant composition comprises a polymethacrylate compound. The term "polymethacrylate compound" used herein refers to poly(methylacrylate) homopolymers of various chain lengths as well as homo- and co-polymers of various chain length alkyl methacrylates. Such compounds are in some cases known for their viscosity index improver and/or pour point depressant properties. Suitable number average molecular weights for the at least one polymethacrylate compound are from about 15,000 to about 1,000,000, for example about 20,000 to about 600,000, as determined by gel permeation chromatography or light scattering methods. As the skilled person will appreciate, polymethacrylate compounds may be added to a base oil composition in preparation of the lubricant composition in the form of a solid or as a solution with the polymethacyrlate dissolved in suitable solvent. Reference herein to an amount of at least one polymethacrylate compound included in the lubricant composition is to be understood as referring to the weight of polymethacrylate compound employed itself, without any dilution.
[0288] In some embodiments, the at least one polymethacrlyate compound may be a comb-type polymer. As the skilled person is aware, a comb-type polymer refers to a polymer having a linear main chain (back bone) with a number of branches along the chain.
[0289] In some embodiments, the at least one polymethacrylate compound may be functionalized. As the skilled person is aware, "functionalized" in this context refers to a polymethacrylate compound with modified side chains for the purpose of imparting dispersancy to the polymer or for conferring properties of a pour point depressant. For example, preferred functionalized polymethacrylate compounds contain amine functionality (e.g. N,N-dialkylaminoalkyl(meth)acrylamide units).
[0290] Hot Liquid Process Simulator (HLPS) testing has also indicated that particular detergents, specifically phenate and neutral sulphonate detergents, can also enhance the high temperature stability of the ether compositions to a far greater degree than the enhancement which is observed in conventional non-ether based compositions. Thus, in a further aspect, the present invention also provides a lubricant composition for an internal combustion engine comprising a base oil of lubricating viscosity, wherein the base oil comprises an ether base stock of formula (A):
##STR00069##
[0291] where: R.sub.a and R.sub.b are aliphatic hydrocarbyl groups and may be the same or different; wherein at least one of R.sub.a and R.sub.b is branched-chain alkyl, alkoxy-substituted-alkyl or cycloalkyl-substituted-alkyl;
[0292] the lubricant composition further comprising: [0293] a) at least one phenate detergent; and/or [0294] b) at least one neutral sulphonate detergent.
[0295] As the skilled person will appreciate, preferred embodiments relating to components of the lubricant compositions according to the first aspect of the invention apply equally to the lubricant compositions according to this further aspect of the invention.
[0296] Metallic and non-metallic phenate and neutral sulphonate detergents may be used in accordance with this aspect of the invention.
[0297] Neutral and overbased metal phenate detergents are well-known for their use as lubricant additives (overbased compounds containing more than the stoichiometric amount of metal required to react with the phenol in order to prepare the metal phenate). Metal phenates include alkali or alkaline earth metal phenates, preferably wherein the metal is selected from barium, sodium, potassium, lithium, calcium, and magnesium, most preferably calcium and magnesium. Phenols employed in the preparation of phenate detergents include hydrocarbyl substituted phenols, such as para-substituted phenols, phenols with more than one hydroxyl group, phenols with fused aromatic rings and/or alkylene bridged biphenols, any of which may be sulphurised (for example, mono- and di-sulphide bridged biphenols). Suitable phenate detergents for use in the present invention include those described, for example, in U.S. Pat. Nos. 4,221,673, 4,104,180 and 4,973,411.
[0298] The phenate detergent may have a base number (BN) of from 0.1 to 400 mg KOH/g, or from 50 to 200 mg KOH/g, for example 150 mg KOH/g, as measured in accordance with ASTM D2896. In preferred embodiments, an overbased phenate detergent is employed having a base number (BN) of from 150 to 400 mg KOH/g, preferably 200 to 300 mg KOH/g, for example from 240 to 260 mg KOH/g, as measured in accordance with ASTM D2896.
[0299] Neutral metal sulphonate detergents are well-known for their use as lubricant additives and include alkali or alkaline earth metal sulphonates, preferably wherein the metal is selected from barium, sodium, potassium, lithium, calcium, and magnesium, most preferably calcium and magnesium. Neutral sulphonates for use in the present invention may have a TBN of less than 60 mg KOH/g, preferably less than 40 mg KOH/g, as measured in accordance with ASTM D2896. Suitably sulphonates may be prepared from sulfonic acids which are typically obtained by the sulphonation of alkyl substituted aromatic hydrocarbons, such as those obtained by alkylating benzene, toluene, xylene, naphthalene, diphenyl or their halogen derivatives such as chlorobenzene, chlorotoluene and chloronaphthalene. The alkyl substituted aryl sulphonates typically contain from about 9 to about 80 or more carbon atoms, preferably from about 16 to about 60 carbon atoms.
[0300] Where the lubricant composition according to the different aspects of the invention comprises a phenate detergent, preferably the phenate detergent is present in an amount of from 0.1 to 5%, more preferably in an amount of from 0.25 to 2.5%, most preferably in an amount of from 0.5 to 1.5%, by weight of the lubricant composition.
[0301] Where the lubricant composition according to the different aspects of the invention comprises a neutral sulphonate detergent, preferably the neutral sulphonate detergent is present in an amount of from 0.01 to 5%, more preferably in an amount of from 0.1 to 2.5%, most preferably in an amount of from 0.25 to 1.5%, by weight of the lubricant composition.
[0302] The lubricant compositions according to the present invention may also comprise additional lubricant additives, in addition to those referred to herein (i.e. the at least one molybdenum compound/polymethacrylate compound or neutral sulphonate/phenate detergent). The additional lubricant additives will typically be present in the lubricant composition in an amount of from about 2% to about 40% by weight, such as about 5% to about 30% by weight.
[0303] Suitable additional lubricant additives include detergents (including metallic and non-metallic detergents), friction modifiers, viscosity modifiers, dispersants (including metallic and non-metallic dispersants), dispersant viscosity modifiers, viscosity index improvers, pour point depressants, anti-wear additives, rust inhibitors, corrosion inhibitors, antioxidants (sometimes also called oxidation inhibitors), anti-foams (sometimes also called anti-foaming agents), seal swell agents (sometimes also called seal compatibility agents), extreme pressure additives (including metallic, non-metallic, phosphorus containing, non-phosphorus containing, sulphur containing and non-sulphur containing extreme pressure additives), surfactants, demulsifiers, anti-seizure agents, wax modifiers, lubricity agents, anti-staining agents, chromophoric agents, metal deactivators, and mixtures of two or more thereof.
[0304] In some embodiments, the lubricant composition comprises a detergent. Examples of detergents include ashless detergents (that is, non-metal containing detergents) and metal-containing detergents. Suitable non-metallic detergents are described for example in U.S. Pat. No. 7,622,431. Metal-containing detergents comprise at least one metal salt of at least one organic acid, which is called soap or surfactant. Suitable organic acids include for example, sulphonic acids, phenols (suitably sulphurised and including for example, phenols with more than one hydroxyl group, phenols with fused aromatic rings, phenols which have been modified for example, alkylene bridged phenols, and Mannich base-condensed phenols and saligenin-type phenols, produced for example by reaction of phenol and an aldehyde under basic conditions) and sulphurised derivatives thereof, and carboxylic acids including for example, aromatic carboxylic acids (for example hydrocarbyl-substituted salicylic acids and derivatives thereof, for example hydrocarbyl substituted salicylic acids and sulphurised derivatives thereof).
[0305] In some embodiments, the lubricant composition comprises a friction modifier. Suitable friction modifiers include for example, ash-producing additives and ashless additives. Examples of suitable friction modifiers include fatty acid derivatives including for example, fatty acid esters, amides, amines, and ethoxylated amines. Examples of suitable ester friction modifiers include esters of glycerol for example, mono-, di-, and tri-oleates, mono-palmitates and mono-myristates. A particularly suitable fatty acid ester friction modifier is glycerol monooleate. Examples of suitable friction modifiers also include molybdenum compounds for example, organo molybdenum compounds, molybdenum dialkyldithiocarbamates, molybdenum dialkylthiophosphates, molybdenum disulphide, tri-molybdenum cluster dialkyldithiocarbamates, non-sulphur molybdenum compounds and the like. Suitable molybdenum-containing compounds are described for example, in EP 1533362 A1 for example in paragraphs [0101] to [0117]. As the skilled person will appreciate, where the lubricant composition comprises the at least one molybdenum compound, additional molybdenum-containing compounds may be present which are added, in particular, for their friction modifier properties. Alternatively, the presence of additional molybdenum compounds may be unnecessary.
[0306] In some embodiments, the lubricant composition comprises a dispersant. Examples of suitable ashless dispersants include oil soluble salts, esters, amino-esters, amides, imides and oxazolines of long chain hydrocarbon-substituted mono- and polycarboxylic acids or anhydrides thereof; thiocarboxylate derivatives of long chain hydrocarbons; long chain aliphatic hydrocarbons containing polyamine moieties attached directly thereto; Mannich condensation products formed by condensing a long chain substituted phenol with formaldehyde and polyalkylene polyamine; Koch reaction products and the like. Particularly preferred dispersants for use in the present invention are long chain aliphatic hydrocarbons containing polyamine moieties attached directly thereto such as polyisobutylene succinyl anhydride-polyamines (PIBSA-PAM).
[0307] Advantageously, borated dispersants may also be used in the lubricant compositions of the present invention without negatively impacting oxidative stability. In some embodiments, the lubricant composition may contain boron in an amount from 0.005 wt. % to 0.05 wt. %, preferably from 0.015 wt. % to 0.035 wt. %. This level of elemental boron may be derived from the use of a borated dispersants and/or boron-containing anti-wear additives or otherwise.
[0308] In some embodiments, the lubricant composition comprises a dispersant viscosity modifier. Examples of suitable dispersant viscosity modifiers and methods of making them are described in WO 99/21902, WO 2003/099890 and WO 2006/099250.
[0309] In some embodiments, the lubricant composition comprises a viscosity index improver. Examples of suitable viscosity modifiers include high molecular weight hydrocarbon polymers (for example polyisobutylene, copolymers of ethylene and propylene and higher alpha-olefins); polyesters (for example polymethacrylates); hydrogenated poly(styrene-co-butadiene or isoprene) polymers and modifications (for example star polymers); and esterified poly(styrene-co-maleic anhydride) polymers. Oil-soluble viscosity modifying polymers generally exhibit number average molecular weights of at least about 15,000 to about 1,000,000, such as about 20,000 to about 600,000 as determined by gel permeation chromatography or light scattering methods. As the skilled person will appreciate, where the lubricant composition comprises the at least one polymethacrylate compound, additional methacrylate polymers may be present which are added, in particular, for their viscosity index improver properties. Alternatively, the presence of additional methacrylate polymers may be unnecessary.
[0310] In some embodiments, the lubricant composition comprises a pour point depressant. Examples of suitable pour point depressants include C.sub.8 to C.sub.18 dialkyl fumarate/vinyl acetate copolymers, methacrylates, polyacrylates, polyarylamides, polymethacrylates, polyalkyl methacrylates, vinyl fumarates, styrene esters, condensation products of haloparaffin waxes and aromatic compounds, vinyl carboxylate polymers, terpolymers of dialkyfumarates, vinyl esters of fatty acids and allyl vinyl ethers, wax naphthalene and the like. As the skilled person will appreciate, where the lubricant composition comprises the at least one polymethacrylate compound, additional methacrylate polymers may be present which are added, in particular, for their pour point depressant properties. Alternatively, the presence of additional methacrylate polymers may be unnecessary.
[0311] In some embodiments, the lubricant composition comprises at least one anti-wear additive. Examples of suitable anti-wear additives include non-phosphorus containing additives for example, sulphurised olefins. Examples of suitable anti-wear additives also include phosphorus-containing anti-wear additives. Examples of suitable ashless phosphorus-containing anti-wear additives include trilauryl phosphite and triphenylphosphorothionate and those disclosed in paragraph [0036] of US 2005/0198894. Examples of suitable ash-forming, phosphorus-containing anti-wear additives include dihydrocarbyl dithiophosphate metal salts. Examples of suitable metals of the dihydrocarbyl dithiophosphate metal salts include alkali and alkaline earth metals, aluminium, lead, tin, molybdenum, manganese, nickel, copper and zinc. Particularly suitable dihydrocarbyl dithiophosphate metal salts are zinc dihydrocarbyl dithiophosphates (ZDDP). As the skilled person will appreciate, where the lubricant composition comprises the at least one molybdenum compound, additional molybdenum-containing compounds may be present which are added, in particular, for their anti-wear properties. Alternatively, the presence of additional molybdenum compounds may be unnecessary.
[0312] In some embodiments, the amount of phosphorus contained in the lubricant composition is less than 0.5 wt. %, preferably from 0.001 to 0.3 wt. %, more preferably from 0.025 to 0.2 wt. %, based on the total weight of the lubricant composition.
[0313] In some embodiments, the lubricant composition comprises a rust inhibitor. Examples of suitable rust inhibitors include non-ionic polyoxyalkylene polyols and esters thereof, polyoxyalkylene phenols, polyoxyalkylene polyols, anionic alky sulphonic acids, zinc dithiophosphates, metal phenolates, basic metal sulphonates, fatty acids and amines.
[0314] In some embodiments, the lubricant composition comprises a corrosion inhibitor. Examples of suitable corrosion inhibitors include phosphosulphurised hydrocarbons and the products obtained by the reaction of phosphosulphurised hydrocarbon with an alkaline earth metal oxide or hydroxide, non-ionic polyoxyalkylene polyols and esters thereof, polyoxyalkylene phenols, thiadiazoles, triazoles and anionic alkyl sulphonic acids. Examples of suitable epoxidised ester corrosion inhibitors are described in US 2006/0090393.
[0315] In some embodiments, the lubricant composition comprises an antioxidant. Examples of suitable antioxidants include alkylated diphenylamines, N-alkylated phenylenediamines, phenyl-a-naphthylamine, alkylated phenyl-a-naphthylamines, dimethylquinolines, trimethyldihydroquinolines and oligomeric compositions derived therefrom, hindered phenolics (including ashless (metal-free) phenolic compounds and neutral and basic metal salts of certain phenolic compounds), aromatic amines (including alkylated and non-alkylated aromatic amines), sulphurised alkyl phenols and alkali and alkaline earth metal salts thereof, alkylated hydroquinones, hydroxylated thiodiphenyl ethers, alkylidenebisphenols, thiopropionates, metallic dithiocarbamates, 1,3,4-dimercaptothiadiazole and derivatives, oil soluble copper compounds (for example, copper dihydrocarbyl thio- or thio-phosphate, copper salts of a synthetic or natural carboxylic acids, for example a C.sub.8 to C.sub.18 fatty acid, an unsaturated acid or a branched carboxylic acid, for example basic, neutral or acidic Cu(I) and/or Cu(II) salts derived from alkenyl succinic acids or anhydrides), alkaline earth metal salts of alkylphenolthioesters, suitably containing C.sub.5 to C.sub.12 alkyl side chains, calcium nonylphenol sulphide, barium t-octylphenyl sulphide, dioctylphenylamine, phosphosulphised or sulphurised hydrocarbons, oil soluble phenates, oil soluble sulphurised phenates, calcium dodecylphenol sulphide, phosphosulphurised hydrocarbons, sulphurised hydrocarbons, phosphorus esters, low sulphur peroxide decomposers and the like.
[0316] In some embodiments, the lubricant composition comprises an antifoam agent. Examples of suitable anti-foam agents include silicones, organic polymers, siloxanes (including poly siloxanes and (poly) dimethyl siloxanes, phenyl methyl siloxanes), acrylates and the like.
[0317] In some embodiments, the lubricant composition comprises a seal swell agent. Examples of suitable seal swell agents include long chain organic acids, organic phosphates, aromatic esters, aromatic hydrocarbons, esters (for example butylbenzyl phthalate) and polybutenyl succinic anhydride.
[0318] The lubricant composition may comprise lubricant additives in the amounts shown in Table 3.
TABLE-US-00003 TABLE 3 Lubricant composition Suitable amount (actives) if Preferred amount (actives) if Additive type present by weight present by weight Phosphorus-containing Corresponding to about 10 to Corresponding to about 10 to anti-wear additives about 6000 ppm P about 1000 ppm P Molybdenum-containing Corresponding to about 10 to Corresponding to about 40 to anti-wear additives about 1000 ppm Mo about 600 ppm Mo Boron-containing anti- Corresponding to about 10 to Corresponding to about 50 to wear additives about 500 ppm B about 100 ppm B Friction modifiers About 0.01 to about 5% About 0.01 to about 1.5% Molybdenum-containing Corresponding to about 10 to Corresponding to about 400 friction modifiers about 1000 ppm Mo to about 600 ppm Mo Molybdenum-containing Corresponding to about 10 to Corresponding to about 40 to additives (e.g. both anti- about 2000 ppm Mo about 1200 ppm Mo wear additives and friction modifiers) Dispersants About 0.1 to about 20% About 0.1 to about 8% Detergents About 0.01 to about 6% About 0.01 to about 4% Viscosity index improvers About 0.01 to about 20% About 0.01 to about 15% Pour point depressants About 0.01 to about 5% About 0.01 to about 1.5% Corrosion and/or rust About 0.01 to about 5% About 0.01 to about 1.5% inhibitors Anti-oxidants About 0.01 to about 10% About 0.5 to 5 about % Antifoams containing Corresponding to about 1 to Corresponding to about 1 to silicon about 20 ppm Si about 10 ppm Si
[0319] The lubricant compositions may have a kinematic viscosity at 40.degree. C. of less than about 60 cSt, such as less than about 55 cSt, or less than about 50 cSt. The lubricant compositions may have a kinematic viscosity at 100.degree. C. of less than about 12 cSt, such as less than about 10 cSt, or less than about 9.5 cSt. The lubricant compositions may have a viscosity index of greater than about 100, such as greater than about 110, or greater than about 120. The kinematic viscosity at 40.degree. C. and the kinematic viscosity at 100.degree. C. may be measured according to ASTM D445. The viscosity index may be calculated according to ASTM D2270.
[0320] The lubricant compositions may have a Noack volatility of less than about 25%, such as less than about 15%, or less than about 10% by weight. Noack volatility may be measured according to CEC-L-40-A-93.
[0321] The lubricant compositions may have a viscosity at 150.degree. C. and a shear rate of 10.sup.6 s.sup.-1 of no greater than 3 cP, such as no greater than 2.8 cP. This high temperature high shear viscosity may be measured according to CEC-L-36-A-90.
[0322] The lubricant composition may have at least one of: [0323] an oxidative stability performance on a CEC-L-088-02 test indicated by an absolute viscosity increase at 40.degree. C. of no more than 45 cSt, such as no more than 35 cSt or no more than 25 cSt; an oxidative stability performance on a CEC-L-109-14 test indicated by an increase in kinematic viscosity at 100.degree. C. of less than 200%, preferably less than 150%; a fuel economy performance on a CEC-L-054-96 test of at least 2.5%, such as at least 3%; a piston cleanliness performance on a CEC-L-088-02 test indicated by an overall piston merit of at least 8.5, such as 9; and a high temperature stability performance on a KHT test at 280.degree. C. in accordance with JPI-5S-55-99 indicated by an overall deposit merit of at least 7.0
[0324] The lubricant compositions may have a cold-crankcase simulator performance at -30.degree. C. of less than about 3000, such as less than about 2800, or less than about 2750, for example as measured according to ASTM D5293.
[0325] Preferred lubricant compositions meet the requirements set out in SAE J300.
[0326] The lubricant compositions may be used in a method of lubricating a surface.
[0327] Suitable surfaces include those in power transmission systems for example drive lines and gear boxes for example for vehicles including for example passenger vehicles and heavy duty vehicles; and those in internal combustion engines, for example the crankcases of internal combustion engines. Suitable surfaces also include those in turbine bearings for example in water turbine bearings.
[0328] Suitable internal combustion engines include, for example, engines used in automotive applications, engines used in marine applications and engines used in land-based power generation plants. The lubricant compositions are particularly suited to use in an automotive internal combustion engine.
[0329] The lubricant compositions may be used to improve the fuel economy and/or piston cleanliness performance and/or turbocharger cleanliness performance of an internal combustion engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine. Accordingly, there are provided methods of improving the fuel economy and/or piston cleanliness performance and/or turbocharger cleanliness performance of an internal combustion engine and/or a vehicle, such as an automotive vehicle associated with an internal combustion engine, comprising the step of providing or supplying to the engine and/or vehicle at least one of the lubricant compositions.
[0330] The invention will now be described with reference to the accompanying figures and examples, which are not limiting in nature, in which:
[0331] FIG. 1 is a graph of Maximum Deposit Thickness (MDT) observed in HLPS analysis of blended compositions containing Guerbet-derived base stock (GE3) and/or a Group III base stock (Yubase 4) together with different detergents; and
[0332] FIG. 2 is a graph of deposit volume (cm.sup.3.times.10.sup.-17) observed in HLPS analysis of blended compositions containing Guerbet-derived base stock (GE3) and/or a Group III base stock (Yubase 4) together with different detergents.
EXAMPLES
Example 1
Properties of Ether Base Stocks
[0333] Guerbet-derived base stock GE3 of formula (1) was prepared, the structure of which is shown in Table 4.
TABLE-US-00004 TABLE 4 Molecular Chemical Weight Formula Structure GE3 522.97 C.sub.36H.sub.74O ##STR00070##
The Following Properties of the Base Stock were Tested:
[0334] Kinematic viscosity at 100.degree. C. (KV100) and kinematic viscosity at 40.degree. C. (KV40) were tested according to ASTM D7279.
[0335] Viscosity index (VI) was calculated according to ASTM D2270.
[0336] Pour point was determined according to ASTM D7346.
[0337] Differential scanning calorimetry (DSC) oxidation onset temperature was tested using a method which was based on ASTM E2009 (method B). According to the method, the base stocks were heated from 50.degree. C. to 300.degree. C., at a rate of 50.degree. C./minute, under a pressure of 500 psi in an aluminium SFI pan. The temperature at which an exotherm was observed was recorded.
[0338] Noack volatility was measured using a method which was based on IP 393 and was considered similar to CEC-L-40-A-93. According to the method, reference oils of known Noack volatility were heated from 40.degree. C. to 550.degree. C. to determine the temperature at which the Noack volatility weight loss of each of the reference oils was reached. The base stocks were subjected to the same process as the reference oils. The Noack weight of the base stocks could be determined based on the results obtained from the reference oils.
[0339] The results of the tests are summarized in Table 5, together with results obtained from a conventional base stock (Yubase 4, a Group III base stock).
TABLE-US-00005 TABLE 5 DSC Noack Pour Oxidation volatility KV100 KV40 Point Onset T (% by (cSt) (cSt) VI (.degree. C.) (.degree. C.) weight) GE3 3.9 16.0 143 -42 202.89 2.4 Yubase 4 4.2 19.2 126 -12 220.00 11.7
[0340] It can be seen that the Guerbet-derived base stock ether has a lower volatility, lower pour point and lower kinematic viscosity as compared to the conventional base oil, although the DSC oxidation onset temperature is lower in Guerbet-derived base stock than the conventional based oil.
Example 2
Properties of Lubricant Compositions Containing Ether Base Stocks
[0341] Guerbet-derived ether base stock GE3 was blended with conventional base oil additives (additive A, a commercially available additive package providing a dispersant level representative of high performance engine oil between 7 and 10 wt % based on the total weight of the lubricant composition; additive B, a cold-flow improver; additive C, an oxidation inhibitor; and additive D, a viscosity index improver) and conventional base oils (Yubase 4, a Group III base oil; and Yubase 6, a Group III base oil) to form a lubricant blend. A Baseline blend was also prepared. Yubase 4 was chosen as the main component of the Baseline blend, since it exhibits a similar KV100 to Guerbet-derived ether base stock, GE3. The Baseline blend was believed to be a stringent baseline for comparison, since it is a 5W-30 formulation which meets certain specifications (ACEA A5/B5, API-SN/GF-4). The details of the blended compositions are shown in Table 6 in % by weight.
TABLE-US-00006 TABLE 6 Baseline blend GE3 blend Additive A 16.4 16.4 Additive B 0.15 0.15 Additive C 0.1 0.1 Additive D 4 4 Yubase 4 67.45 17.45 Yubase 6 11.9 11.9 GE3 0 50
[0342] No problems with miscibility were encountered during preparation of the blended compositions.
[0343] The blended compositions were tested to see whether the advantageous properties of the base stocks would be reflected in a fully formulated lubricant composition. The following properties were tested:
[0344] Kinematic viscosity at 100.degree. C. (KV100) and kinematic viscosity at 40.degree. C. (KV40) were tested according to ASTM D445 (part of SAE J300).
[0345] Viscosity index (VI) was calculated according to ASTM D2270.
[0346] Cold-cranking simulator (CCS) analysis was carried out at -30.degree. C. according to ASTM D5293 (part of SAE J300).
[0347] High temperature high shear (HTHS) analysis was carried out according to CEC-L-36-A-90.
[0348] Total base number (TBN) was determined according to ASTM D2896.
[0349] Noack volatility was tested according to CEC-L-40-A-93.
[0350] Sulphated ash content was measured according to IP 163.
[0351] The results of the tests are summarized in Table 7.
TABLE-US-00007 TABLE 7 Baseline blend GE3 blend KV40 (cSt) 53.59 44.63 KV100 (cSt) 9.542 8.688 VI 164 177 CCS -30.degree. C. (cP) 4656 2702 HTHS (cP) 2.98 2.75 TBN (mg KOH/g) 11.66 11.44 NOACK (% by weight) 11.2 9.7 Sulphated ash (%) 1.22 1.27
[0352] It can be seen that the properties of the Guerbet-derived base stock are also exhibited in the blended composition. In particular, beneficial viscosity, volatility and cold-flow properties are observed. The Guerbet-derived base stock also exhibited similar HTHS measurements, TBNs and sulphated ash contents to the Baseline blend.
Example 3
Komatsu Hot-Tube (KHT) Test
[0353] Fully formulated compositions comprising Guerbet-derived base stock (GE3) and/or a Group III base stock (Yubase 4) together with varying amounts of a molybdenum-sulfur compound and/or a polymethacrylate compound (a comb-type copolymer of alkyl methacrylates in solution--approximately 1:1 dilution ratio) as well as additional lubricant additives including (non-borated) dispersant, ZDDP, detergents, antioxidants and viscosity modifier (VM) were subjected to the KHT test in accordance with JPI-5S-55-99 and SAE Technical Paper 840262. Results obtained from the KHT testing in the form of deposit merits are shown in Table 8 (compositional data shown in % by weight).
TABLE-US-00008 TABLE 8 Lubricant Composition 1 2 3 4 5 6 Yubase 4 83.683 33.683 81.925 31.925 83.685 33.685 GE3 ether 50 50 50 Phenolic AO 0.5 0.5 0.5 0.5 0.5 0.5 Aminic AO 0.1 0.1 0.1 0.1 0.1 0.1 Detergents 2.18 2.18 2.18 2.18 2.18 2.18 Dispersant 6 6 6 6 6 6 ZDDP 0.535 0.535 0.535 0.535 0.535 0.535 VM 7 7 7 7 Antifoam 0.002 0.002 PMA 7 7 Mo--S 1.76 1.76 compound Mo (elemental, 0.08 0.08 wt. %) KHT 3 7.5 6.5 6.5 6.5 7.5 rating
[0354] According to the results of the KHT testing, the ether containing lubricant composition 2 exhibits a significantly higher KHT merit rating (7.5) compared to that of the non-ether containing lubricant composition 1. These results indicate that the presence of the ether confers a benefit in reducing high temperature induced deposit formation which would otherwise lead to scuffing. In addition, the ether-containing lubricant composition 4, containing an amount of 0.08 wt. % of molybdenum, exhibits the same KHT deposit merit rating (6.5) as the corresponding non-ether containing composition 3. This is particularly advantageous since the ether composition exhibits a lower viscosity profile than the non-ether composition and yet exhibits equivalent high temperature stability. This means that a benefit may be seen in terms of fuel economy through the use of the ether composition of the invention over a conventional non-ether containing composition without any accompanying increase in turbocharger deposits and piston scuffing. As discussed in more detail below, the results in the TEOST 33C testing also demonstrate that the presence of molybdenum impacts deposit formation in ether-containing compositions to no more of a degree than the non-ether containing compositions despite the DSC oxidation onset temperature of the ether base stock being lower than that of the conventional Group III base stock as demonstrated in Example 1.
[0355] The results of the KHT testing also demonstrate a significant advantage in the presence of the at least one polymethacrylate compound, particular in the case of the ether-containing compositions. For instance, the ether-containing lubricant composition 6 also containing a polymethacrylate compound exhibits a substantially higher deposit merit rating (7.5) in the KHT test compared to the non-ether containing composition 5. Thus, when the at least one polymethacrylate compound is present, the ether composition is able to out-perform the corresponding non-ether composition in terms of high temperature stability. This is of particular benefit since the ether composition exhibits a lower viscosity profile than the non-ether composition and so the present invention is able to benefit from increased fuel economy without increasing deposits in hot regions of the engine, such as in turbochargers, or causing piston scuffing which would otherwise shorten engine lifetime.
Example 4
TEOST 33C Test
[0356] Fully formulated compositions comprising Guerbet-derived base stock (GE3) and/or a Group III base stock (Yubase 4) together with varying amounts of a molybdenum-sulfur compound and/or a polymethacrylate (a comb-type copolymer of alkyl methacrylates in solution--approximately 1:1 dilution ratio) as well as additional lubricant additives including (non-borated) dispersant, ZDDP, detergents, antioxidants and viscosity index modifier (VIM) were subjected to the TEOST 33C test in accordance with standard method ASTM D6335. Results obtained from the TEOST 33C testing in the form of total deposits (in turbochargers) are shown in Table 9 (compositional data shown in % by weight).
TABLE-US-00009 TABLE 9 Lubricant Composition A B C D E F G H I J K Yubase 4 83.593 89.273 87.793 84.193 84.873 86.533 85.823 83.783 82.993 33.87 32.99 GE3 ether 50 50 Phenolic AO 0.5 0 0 0 0 0 0 0.5 0.5 0.5 0.5 Aminic AO 0.2 0.1 0.5 0.1 0.5 0.3 1 1 1 1 1 Sulphonate (400BN) 0.86 0.43 0.86 0.86 0.43 0.64 0.65 0.43 0.43 0.43 0.43 Sulphonate (neutral) 0.34 0.17 0.34 0.34 0.17 0.26 0.26 0.17 0.17 0.17 0.17 Phenate (150BN) 0.97 0.49 0.97 0.97 0.49 0.73 0.73 0.49 0.49 0.49 0.49 Dispersant 6 2 2 6 6 4 4 6 6 6 6 ZDDP 0.535 0.535 0.535 0.535 0.535 0.535 0.535 0.535 0.535 0.535 0.535 VIM 7 7 7 7 7 7 7 Antifoam 0.002 0.002 0.002 0.002 0.002 0.002 0.002 0.002 0.002 0.002 0.002 PMA 7 7 7 7 Mo--S compound 0.88 0.88 Mo (elemental, wt. %) 0.04 0.04 Total deposits (mg) 17.3 14.2 15.5 7.4 18.2 12.9 15.7 36.6 49.3 33.8 47.4 TEOST 33C
The results of the TEOST 33C generally demonstrate the benefit of increasing dispersant and detergent levels while decreasing antioxidant levels, particularly aminic antioxidant, for reducing deposit formation in turbochargers in non-ether containing compositions, in line with expectations (for instance, compare results for compositions F and G or compare composition D with A-C and E to G). In addition, the results demonstrate that the presence of the at least one polymethacrylate compound, alone or also in combination with the at least one molybdenum compound, is tolerated better by the ether composition than the corresponding non-ether composition (compare results of compositions H and J as well as I and K) in the TEOST 33C test. This is surprising when the DSC oxidation onset stability of the ether base stock is lower than that of the conventional Group III base stock as demonstrated in Example 1 (on the basis of which poorer TEOST 33C performance would be expected). This is particularly advantageous since the ether composition exhibits a lower viscosity profile than the corresponding non-ether composition whilst exhibiting greater high temperature stability in the TEOST 33C test than the corresponding non-ether composition. This means that a benefit may be seen in terms of fuel economy through the use of the ether composition of the invention in comparison to a conventional Group III base oil composition containing no ether base stock, yet without any accompanying increase in turbocharger deposits and piston scuffing.
Example 5
Hot Liquid Process Simulator (HLPS) Testing
[0357] Blended compositions comprising Guerbet-derived base stock (GE3) and/or a
[0358] Group III base stock (Yubase 4) together with varying amounts of sulphonate (400BN), neutral sulphonate, and phenate (150BN) detergents were subjected to HLPS testing. The HLPS testing corresponds to a hot-tube test in which all oil compositions were subjected to the same heating stress, for the same period of time. HLPS testing is used as a means for characterising the propensity of an oil to create deposits in a hotregion of the engine by simulating pressurized oil lines. Results of the HLPS testing in the form of Maximum Deposit Thickness ("MDT") (nm), which corresponds to the maximum thickness of deposit measured along the HLPS tube, and total deposit volume (cm.sup.3.times.10.sup.-17) are provided in Table 10 (compositional data shown in % by weight).
TABLE-US-00010 TABLE 10 Lubricant Composition a b c d e f g h i j k l Yubase 4 100 99.35 97.9 98.57 99.14 99.403 50 49.35 47.9 48.57 49.14 49.403 GE3 ether 50 50 50 50 50 50 Sulphonate (400BN) 0.65 1.43 0.65 1.43 Sulphonate (neutral) 0.86 0.86 Phenate 2.1 0.597 2.1 0.597 HLPS deposit vol. 4.08 5.97 6.76 No result 5.13 2.95 4.28 3.90 2.73 3.98 3.86 3.42 (cm.sup.3, .times. 10.sup.-17) MDT nm 252 178 249 232 98 192 249 110 250 199 75
[0359] The results of the HLPS tests demonstrate that, for a given TBN value, the ether based compositions comprising a phenate detergent exhibit significantly lower deposit levels than the corresponding non-ether based compositions. The ether compositions comprising a typical level of neutral sulphonate also perform markedly better than the corresponding non-ether based compositions. The results shown in Table 10 are also shown in the graphs of FIGS. 1 and 2 which show Maximum Deposit Thickness and deposit volume for each of the compositions tested respectively. These data show the advantages that may be derived by employing particular detergents, for instance phenate or neutral sulphonate, in an ether-based lubricant composition for reducing the propensity for deposit formation. This means that other lubricant additives that are known to increase deposits, for example antioxidants, may be used at higher levels without resulting in unacceptable level of deposit formation.
[0360] The dimensions and values disclosed herein are not to be understood as being strictly limited to the exact numerical values recited. Instead, unless otherwise specified, each such dimension is intended to mean both the recited value and a functionally equivalent range surrounding that value. For example, a dimension disclosed as "40 mm" is intended to mean "about 40 mm."
[0361] Every document cited herein, including any cross referenced or related patent or application, is hereby incorporated herein by reference in its entirety unless expressly excluded or otherwise limited. The citation of any document is not an admission that it is prior art with respect to any invention disclosed or claimed herein or that it alone, or in any combination with any other reference or references, teaches, suggests or discloses any such invention. Further, to the extent that any meaning or definition of a term in this document conflicts with any meaning or definition of the same term in a document incorporated by reference, the meaning or definition assigned to that term in this document shall govern.
[0362] While particular embodiments of the present invention have been illustrated and described, it would be obvious to those skilled in the art that various other changes and modifications can be made without departing from the spirit and scope of the invention. It is therefore intended to cover in the appended claims all such changes and modifications that are within the scope and spirit of this invention.
* * * * *










































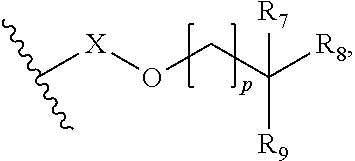






























D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.