Lubricating Composition Comprising Glycerol Monoethers
CHAMPAGNE; Nicolas ; et al.
U.S. patent application number 16/467439 was filed with the patent office on 2019-10-03 for lubricating composition comprising glycerol monoethers. The applicant listed for this patent is CENTRE NATIONAL DE LA RECHERCHE SCIENTIFIQUE, TOTAL MARKETING SERVICES. Invention is credited to Nicolas CHAMPAGNE, Gwendoline GRAVE, Francoise QUIGNARD, Nathalie TANCHOUX.
| Application Number | 20190300809 16/467439 |
| Document ID | / |
| Family ID | 57909744 |
| Filed Date | 2019-10-03 |





| United States Patent Application | 20190300809 |
| Kind Code | A1 |
| CHAMPAGNE; Nicolas ; et al. | October 3, 2019 |
LUBRICATING COMPOSITION COMPRISING GLYCEROL MONOETHERS
Abstract
Disclosed is a lubricating composition including: at least one base oil; and at least one glycerol monoether characterised in that one of the alcohol functions of the glycerol forms an ether function with a linear or branched, alkyl or alkylene R group including 6 or 7 carbon atoms, the composition being an engine lubricant.
| Inventors: | CHAMPAGNE; Nicolas; (Caluire, FR) ; GRAVE; Gwendoline; (Toulouse, FR) ; QUIGNARD; Francoise; (Montpellier, FR) ; TANCHOUX; Nathalie; (Grabels, FR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 57909744 | ||||||||||
| Appl. No.: | 16/467439 | ||||||||||
| Filed: | December 6, 2017 | ||||||||||
| PCT Filed: | December 6, 2017 | ||||||||||
| PCT NO: | PCT/EP2017/081746 | ||||||||||
| 371 Date: | June 6, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10N 2020/071 20200501; C10N 2030/54 20200501; C10M 2207/046 20130101; C10N 2040/25 20130101; C10M 129/16 20130101; C10M 2203/003 20130101; C10M 2209/084 20130101; C10M 2215/064 20130101; C10N 2030/06 20130101; C10M 169/04 20130101; C10M 2205/04 20130101; C10M 2203/1025 20130101; C10N 2020/069 20200501; C10M 2205/0285 20130101; C10M 2203/1025 20130101; C10N 2020/02 20130101; C10M 2205/04 20130101; C10M 2205/06 20130101; C10N 2060/02 20130101; C10M 2203/1025 20130101; C10N 2020/02 20130101; C10M 2205/04 20130101; C10M 2205/06 20130101; C10N 2060/02 20130101 |
| International Class: | C10M 129/16 20060101 C10M129/16; C10M 169/04 20060101 C10M169/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 7, 2016 | FR | 1662075 |
Claims
1-9. (canceled)
10. A lubricating composition comprising: at least one base oil; and at least one glycerol mono-ether, characterized in that one of the alcohol functions of glycerol forms an ether function with a linear or branched alkyl or alkylene group R comprising 6 or 7 carbon atoms, the composition being an engine lubricant.
11. The lubricating composition of claim 10 comprising: at least one base oil; and at least one glycerol mono-ether of formula (I) or (II), taken alone or as a mixture ##STR00008## in which R represents a linear or branched alkyl or alkylene group comprising 6 or 7 carbon atoms.
12. The lubricating composition according to claim 10 wherein R represents a linear or branched alkyl or alkylene group comprising 6 carbon atoms.
13. The lubricating composition according to claim 10 comprising from 0.01% to 5% by weight, of glycerol mono-ether.
14. A method of lubricating an engine comprising the implementation of a lubricating composition according to claim 10.
15. The method according to claim 14 wherein the engine is a motor engine.
16. The method for reducing the friction in an engine comprising the implementation of a lubricating composition according to claim 10.
17. The method for reducing the fuel consumption of an engine comprising the implementation of a lubricating composition according to claim 10.
18. The method for reducing friction in an engine comprising the implementation of a glycerol mono-ether as defined in claim 10 in a lubricating composition of an engine.
19. The method for reducing the fuel consumption of an engine comprising the implementation of a glycerol mono-ether as defined in claim 10 in a lubricating composition of an engine.
20. The lubricating composition according to claim 11 comprising from 0.01 to 2% by weight of glycerol mono-ether of formula (I) or (II) taken alone or as a mixture.
21. The lubricating composition according to claim 10, comprising from 0.1 to 1.5% by weight of glycerol mono-ether characterized in that one of the alcohol functions of glycerol forms an ether function with a linear or branched alkyl or alkylene group R, wherein R represents a linear or branched alkyl or alkylene group comprising 6 carbon atoms.
Description
[0001] The invention relates to the field of friction modifiers, and, in particular, organic friction modifiers. More particularly, the invention relates to a lubricating composition comprising a friction modifier selected from glycerol monoethers. The invention also relates to the use of this lubricating composition for lubricating an engine.
[0002] One of the major objectives of the present automotive industry is the reduction of the fuel consumption of engines, especially motor vehicle engines, and thus the improvement of fuel economy vehicles. Reducing friction in an engine is an effective way to achieve fuel economy. Thus, much research has been done on friction modifiers.
[0003] Four major groups stand out among friction modifiers: nanoparticles, polymers, organomolybdenum compounds and organic molecules.
[0004] Although nanoparticles and polymers are little used today, this is not the case for organomolybdenum compounds, which represent the most important family of friction modifiers. The best-known and most widely used inorganic friction modifier is molybdenum dithiocarbamate (MoDTC). These inorganic friction modifiers, although very effective, have certain drawbacks. In fact, they may induce some fouling and corrosion of engine parts. In addition, they are only active at high temperatures and may damage some type of amorphous carbon (DLC) surface.
[0005] In addition, from an ecological point of view, it is necessary to reduce the content of sulfur or phosphorus elements of the lubricating compositions used.
[0006] Thus, organic friction modifiers are studied and conventionally used. It has been found that glycerol esters are effective, and, in particular, glycerol mono-oleate is the most commonly used commercially. It has the advantage of not containing ash, phosphorus or sulfur and being produced from renewable raw materials. However, its properties as a friction modifier are lower than those of molybdenum dithiocarbamate.
[0007] The use of glycerol ethers as a friction modifier is also known. Thus, the application JPS5925890 describes the use of glycerol ethers comprising an alkyl chain comprising from 4 to 28 carbon atoms. JP2000273481 also discloses the use of glycerol ethers comprising an alkyl chain comprising more than 14 carbons as friction modifiers.
[0008] There is therefore an interest in proposing new friction modifiers to improve efficiency.
[0009] Thus, an object of the present invention is to provide a friction modifier and a lubricating composition comprising this friction modifier that overcome all or some of the aforementioned drawbacks.
[0010] Another object of the present invention is to provide an organic friction modifier derived from renewable raw materials and which does not have the drawbacks of MoDTC and which is effective at low temperature.
[0011] Yet another object of the present invention is to provide a lubricating composition for reducing friction and thus reducing the fuel consumption of the engine, preferably of a motor vehicle.
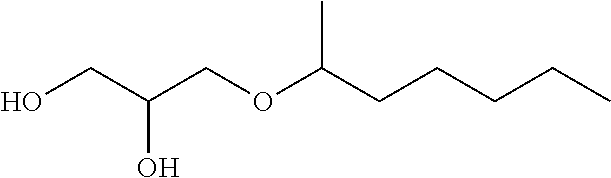
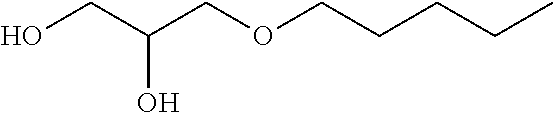
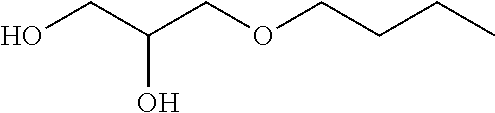
[0012] The present invention thus relates to a lubricating composition comprising: [0013] at least one base oil; and [0014] at least one glycerol mono-ether, characterized in that one of the alcohol functions of glycerol forms an ether function with a linear or branched alkyl or alkylene group R comprising from 5 to 8 carbon atoms, preferably from 6 to 7 carbon atoms, preferably 6 carbon atoms.
[0015] Preferably, in the composition according to the invention, the group R comprises 6 or 7 carbon atoms.
[0016] Preferably, the composition according to the invention is an engine lubricant.
[0017] Advantageously, the R group of the glycerol monoether according to the invention forms an ether bond with any oxygen atom of glycerol, whether it is bonded to one of the two primary carbons or to the secondary carbon of glycerol.
[0018] Advantageously according to the invention, the glycerol mono-ether is chosen from glycerol monoethers of formula (I) or (II), taken alone or as a mixture:
##STR00001##
[0019] in which R represents a linear or branched alkyl or alkylene group comprising from 5 to 8 carbon atoms, preferably from 6 to 7 carbon atoms, preferably 6 carbon atoms.
[0020] Preferably, in the compounds of formula (I) or (II), R represents a linear or branched alkyl or alkylene group comprising 6 or 7 carbon atoms.
[0021] Preferably, the glycerol mono-ether according to the invention is chosen from the compounds of formula (I).
[0022] Preferably, the glycerol mono-ether according to the invention is chosen from the compounds of formula (II).
[0023] Preferably, the glycerol mono-ether according to the invention is a mixture of at least one compound of formula (I) and at least one compound of formula (II).
[0024] Advantageously, the lubricating composition according to the invention comprises from 0.01% to 5% by weight of glycerol mono-ether according to the invention relative to the total weight of the lubricating composition. Preferably, the lubricating composition comprises from 0.01% to 2% by weight, preferably from 0.1% to 1.5% by weight of glycerol mono-ether as defined above, relative to the total weight of the lubricating composition.
[0025] The glycerol monoethers according to the invention may be obtained by any technique known to those skilled in the art. In particular, a preferred synthetic route for the preparation of glycerol monoethers of the invention consists in the reaction between glycerol and an alcohol ROH, wherein R has the definition given above.
[0026] The etherification reaction between glycerol and an alcohol is preferably carried out in the presence of an acid catalyst, preferably an acid heterogeneous catalyst or a homogeneous acid catalyst.
[0027] Preferably, the acidic heterogeneous catalysts are chosen from zeolites, resins or oxides of alumina.
[0028] Among the resins, may be mentioned perfluorinated polymer resins such as Nafion.RTM. NR50, cation exchange resins such as Dowex 50wx8, or among ion exchange resins with acidic properties such as Amberlyst.RTM. 15 and 36.
[0029] Among the zeolites, particular mention may be made of mordenites, ZMS-5 zeolites of the MFI type, BEA zeolites or faujasites. Preferably, the acidic heterogeneous catalyst is a zeolite, preferably a mordenite type zeolite, preferably a mordenite with a Si/Al ratio of 11.
[0030] Preferably, the homogeneous acidic catalysts are chosen from phosphoric acid, para-toluenesulfonic acid (APTS) and triflic acid. Preferably, the acidic homogeneous catalyst is para-toluenesulfonic acid (APTS).
[0031] Advantageously according to the invention and in order to favor the synthesis of the glycerol mono-ether, the amount of acidic homogeneous catalyst involved in the reaction is between 1% and 10 mol% relative to the glycerol content, preferably it is between 1% and 2.5 mol%. Advantageously, the amount of heterogeneous catalyst involved in the reaction is between 1 and 5 mol% relative to the glycerol content, preferably it is 3.5 mol%.
[0032] The alcohol/glycerol molar ratio is optimized in order to obtain the best possible yield of glycerol mono-ether while limiting the secondary reactions of glycerol oligomer formation. Preferably, the alcohol/glycerol molar ratio is between 1/6 and 3/1, preferably it is 1/1.
[0033] The etherification reaction may also, in another embodiment, be carried out from the di-ether resulting from the reaction of the alcohol on itself.
[0034] The etherification reaction of glycerol may be carried out in any type of reactor known to those skilled in the art. Advantageously, it is conducted in an autoclave.
[0035] The conditions of temperature, pressure and the etherification reaction time of the glycerol can be determined in the usual manner by those skilled in the art. Advantageously, the reaction is carried out at a temperature of between 80 and 200.degree. C., preferably between 130 and 160.degree. C., for example it may be carried out at 150.degree. C.
[0036] Advantageously according to the invention, the reaction is carried out for at least 5 hours, preferably for 5 to 48 hours. The reaction time may be 24 hours or 48 hours.
[0037] Advantageously according to the invention, the method comprises a purification step. This purification step may comprise the following steps: liquid-liquid extraction and/or fractional distillation under reduced pressure. The liquid-liquid extraction is conducted in the presence of a pair of solvents, which may be determined in the usual manner by those skilled in the art.
[0038] Preferably, the acetonitrile/heptane pair may be used.
[0039] Advantageously, at least one liquid-liquid extraction is conducted, preferably at least two. Even more preferably, three liquid-liquid extractions are carried out.
[0040] In a preferred manner according to the invention, during the distillation step, the initial alcohol and the glycerol mono-ether are separated.
[0041] Advantageously, the process according to the invention makes it possible to selectively obtain glycerol monoethers.
[0042] The lubricating composition according to the invention comprises at least one base oil.
[0043] The base oil(s) used in the lubricating compositions according to the invention may be chosen from a wide range. The base oil of the lubricating composition used according to the invention may, in particular, be chosen from mineral, synthetic or natural oils, bio-sourced, animal, plant, known to those skilled in the art.
[0044] The base oil(s) according to the invention may be oils of mineral or synthetic origin, chosen from oils of groups I to V according to the classes defined in the API classification (or their equivalents according to the ATIEL classification) (Table A), alone or in mixtures.
TABLE-US-00001 TABLE A Saturated Viscosity index content Sulfur content (VI) Group I Mineral oils <90% >0.03% 80 .ltoreq. VI < 120 Group II Hydrocracked .gtoreq.90% .ltoreq.0.03% 80 .ltoreq. VI < 120 oils Group III Hydrocracked .gtoreq.90% .ltoreq.0.03% .gtoreq.120 or hydro-isomerized oils Group IV Polyalphaolefines (PAO) Group V Esters and other bases not included in groups I to IV
[0045] The mineral base oils useful according to the invention include all types of bases obtained by atmospheric and vacuum distillation of crude oil, followed by refining operations such as solvent extraction, desalphating, solvent dewaxing, hydrotreatment, hydrocracking, hydroisomerization and hydrofinishing.
[0046] Mixtures of synthetic and mineral oils may also be used.
[0047] There is generally no limitation on the use of different lubricating bases for producing the lubricating compositions used according to the invention, except that they must have properties, in particular viscosity, viscosity index, sulfur, oxidation resistance, adapted for use for engines or for vehicle transmissions.
[0048] The lubricating composition according to the invention may comprise from 50 to 99% of at least one base oil, preferably from 60 to 99%, advantageously from 70 to 99% by weight, relative to the total weight of lubricating composition.
[0049] The lubricating composition according to the invention may also comprise numerous additives.
[0050] The preferred additives for the lubricating composition used according to the invention are chosen from detergents, antiwear additives, extreme pressure additives, viscosity index improvers, dispersants, antioxidants, pour point improvers, anti-foam agents, and inorganic friction modifiers and mixtures thereof.
[0051] The lubricating composition according to the invention may also comprise at least one detergent additive.
[0052] The detergent additives generally make it possible to reduce the formation of deposits on the surface of the metal parts by dissolving the secondary oxidation and combustion products.
[0053] The detergent additives that may be used in the lubricating composition according to the invention are generally known to those skilled in the art. The detergent additives may be anionic compounds comprising a long lipophilic hydrocarbon chain and a hydrophilic head. The associated cation may be a metal cation of an alkali metal or alkaline earth metal.
[0054] The detergent additives are preferably chosen from the alkali metal or alkaline earth metal salts of carboxylic acids, the sulphonates, the salicylates, the naphthenates and the phenate salts. The alkali and alkaline earth metals are preferably calcium, magnesium, sodium or barium.
[0055] These metal salts generally comprise the metal in stoichiometric amount or in excess, therefore in an amount greater than the stoichiometric amount. These are then overbased detergent additives; the excess metal bringing the overbased character to the detergent additive is then generally in the form of an oil-insoluble metal salt, for example a carbonate, a hydroxide, an oxalate, an acetate, a glutamate, preferably a carbonate.
[0056] Advantageously, the lubricating composition according to the invention may comprise from 2 to 4% by weight of detergent additive relative to the total mass of the lubricating composition.
[0057] Anti-wear additives and extreme pressure additives protect friction surfaces by forming a protective film adsorbed on these surfaces.
[0058] There is a wide variety of anti-wear additives. In a preferred manner for the lubricating composition according to the invention, the anti-wear additives are chosen from phosphosulfur additives such as metal alkylthiophosphates, in particular zinc alkylthiophosphates, and more specifically zinc dialkyldithiophosphates or ZnDTPs. The preferred compounds have the formula Zn((SP(S)(OR.sup.11)(OR.sup.12.sub.2)), in which R.sup.11 and R.sup.12, which may be identical or different, independently represent an alkyl group, preferably an alkyl group comprising from 1 to 18 carbon atoms.
[0059] Amine phosphates are also anti-wear additives which may be used in the lubricating composition according to the invention. However, the phosphorus provided by these additives may act as a poison of the catalytic systems of automobiles because these additives are ash generators. These effects may be minimized by partially substituting amine phosphates with non-phosphorus additives, such as, for example, polysulfides, especially sulfur-containing olefins. Advantageously, the lubricating composition according to the invention may comprise from 0.01 to 6% by weight, preferably from 0.05 to 4% by weight, more preferably from 0.1 to 2% by weight relative to the total weight of the lubricating composition, anti-wear additives and extreme pressure additives.
[0060] The lubricating composition of the present invention may also comprise at least one viscosity index improving additive. As examples of additives improving the viscosity index, mention may be made of polymeric esters, homopolymers or copolymers, hydrogenated or non-hydrogenated, of styrene, butadiene and isoprene, in particular polyacrylates, polymethacrylates (PMA) or alternatively olefin copolymers, especially ethylene/propylene copolymers.
[0061] Advantageously, the lubricating composition according to the invention may also comprise at least one dispersing agent.
[0062] The dispersing agent may be chosen from Mannich bases, succinimides and their derivatives.
[0063] Also advantageously, the lubricating composition according to the invention may comprise from 0.2 to 10% by weight of dispersing agent relative to the total mass of the lubricating composition.
[0064] Advantageously, the lubricating composition according to the invention may comprise at least one antioxidant additive.
[0065] The antioxidant additive generally serves to retard the degradation of the lubricating composition in service. This degradation may notably result in the formation of deposits, the presence of sludge or an increase in the viscosity of the lubricating composition.
[0066] Antioxidant additives act in particular as radical inhibitors or destroyers of hydroperoxides. Among the antioxidant additives commonly used, mention may be made of antioxidant additives of the phenolic type, antioxidant additives of the amine type, antioxidant phosphosulfur additives. Some of these antioxidant additives, for example phosphosulfur antioxidant additives, may be ash generators. Phenolic antioxidant additives may be ash-free or may be in the form of neutral or basic metal salts. The antioxidant additives may especially be chosen from sterically hindered phenols, sterically hindered phenol esters, and sterically hindered phenols comprising a thioether bridge, diphenylamines, diphenylamines substituted with at least one C.sub.1-C.sub.12 alkyl group, and N,N'-dialkyl-aryl diamines and mixtures thereof.
[0067] Preferably, according to the invention, the sterically hindered phenols are chosen from compounds comprising a phenol group in which at least one vicinal carbon of the carbon bearing the alcohol function is substituted by at least one C.sub.1-C.sub.10 alkyl group, preferably a C.sub.1-C.sub.6alkyl group, preferably a C.sub.4 alkyl group, preferably by the ter-butyl group.
[0068] Amino compounds are another class of antioxidant additives that may be used, optionally in combination with phenolic antioxidant additives. Examples of amine compounds are aromatic amines, for example aromatic amines of formula NR.sup.13R.sup.14R.sup.15 in which R.sup.13 represents an optionally substituted aliphatic or aromatic group, R.sup.14 represents an optionally substituted aromatic group, R.sup.15 represents a hydrogen atom, an alkyl group, an aryl group or a group of formula R.sup.16S(O).sub.2R.sup.17 in which R.sup.16 represents an alkylene group or an alkenylene group, R.sup.17 represents an alkyl group, an alkenyl group or an aryl group and z represents 0, 1 or 2.
[0069] Sulfurized alkyl phenols or their alkali and alkaline earth metal salts may also be used as antioxidant additives.
[0070] Another class of antioxidant additives is copper compounds, for example copper thio- or dithio-phosphates, copper and carboxylic acid salts, dithiocarbamates, sulphonates, phenates, copper acetylacetonates. Copper salts I and II, succinic acid or anhydride salts may also be used.
[0071] The lubricating composition according to the invention may contain all types of antioxidant additives known to those skilled in the art.
[0072] Advantageously, the lubricating composition comprises at least one ash-free antioxidant additive.
[0073] Also advantageously, the lubricating composition according to the invention comprises from 0.5 to 2% by weight relative to the total weight of the composition, of at least one antioxidant additive.
[0074] Also advantageously, the lubricating composition according to the invention may also comprise at least one pour point depressant additive.
[0075] By slowing the formation of paraffin crystals, pour point depressant additives generally improve the cold behavior of the lubricating composition according to the invention.
[0076] As examples of pour point depressant additives, mention may be made of alkyl polymethacrylates, polyacrylates, polyarylamides, polyalkylphenols, polyalkylnaphthalenes and alkylated polystyrenes.
[0077] The lubricating composition may also comprise an anti-foam additive chosen from silicones and their derivatives, such as polysiloxanes and their derivatives. Such an anti-foam additive may be Bluesil 47V12500.RTM. marketed by Bluestar Silicones. The antifoam additive of the lubricating composition according to the invention may also be chosen from acrylics, such as PC1244.RTM. marketed by Mosanto.
[0078] Advantageously, the lubricating composition according to the invention may comprise at least one additional friction-modifying additive, such as an inorganic friction modifier. The inorganic friction modifier additive may be selected from a compound providing metal elements and an ash free compound. Among the compounds providing metal elements, mention may be made of transition metal complexes such as Mo, Sb, Sn, Fe, Cu and Zn, the ligands of which may be hydrocarbon compounds comprising oxygen, nitrogen, sulfur or phosphorus.
[0079] In particular, mention may be made of organomolybdenum compounds which represent the most important family of friction modifiers. Molybdenum dithiocarbamate (MoDTC) is the best known compound in this family.
[0080] Advantageously, the lubricating composition according to the invention may comprise from 0.01 to 5% by weight or from 0.01 to 2% by weight, preferably from 0.1 to 1.5% by weight or 0.1 at 2% by weight relative to the total weight of the lubricating composition, of the additional friction modifier additive.
[0081] The invention also relates to the use of a lubricating composition according to the invention for lubricating an engine, preferably a motor vehicle engine.
[0082] The invention also relates to the use of a lubricating composition according to the invention for reducing the friction in an engine, preferably a motor vehicle engine.
[0083] The invention also relates to the use of the lubricating composition according to the invention for reducing the fuel consumption of an engine, preferably of a motor vehicle engine.
[0084] Preferably, the reduction of friction in an engine or the reduction of the fuel consumption of an engine is measured with respect to the friction or fuel consumption measured with respect to a reference composition.
[0085] Advantageously, the reference composition does not comprise glycerol mono-ether, characterized in that one of the alcohol functions of glycerol forms an ether function with an alkyl or alkylene R group comprising 6 or 7 carbon atoms.
[0086] Advantageously, the invention also relates to the use of the lubricating composition according to the invention for reducing the fuel consumption at the start and during the operating phase of the engine.
[0087] The invention also relates to the use of a glycerol mono-ether as defined above in a lubricating composition for reducing the friction in an engine or for reducing the fuel consumption of an engine, preferably a motor vehicle engine.
[0088] Preferably, the use of a glycerol mono-ether of formula according to the invention as defined above allows a reduction in fuel consumption at startup and during the operating phase of the engine.
[0089] The invention also relates to a method of lubricating an engine, preferably a motor vehicle engine comprising the implementation of a lubricating composition according to the invention.
[0090] The invention also relates to a method for reducing the fuel consumption of an engine, preferably a motor vehicle engine comprising the implementation in the engine of a lubricating composition according to the invention.
[0091] The various aspects of the invention are illustrated by the following examples, but are not limited thereto.
EXAMPLES
[0092] Compounds Used:
[0093] The different glycerol monoethers tested are described in Table I below. The synthetic route requires that the glycerol and fatty alcohol used is soluble one in the other. However, any fatty alcohol comprising 8 or more carbon atoms is not soluble in glycerol, wherein the corresponding glycerol ethers can not be synthesized according to the described synthetic route.
TABLE-US-00002 TABLE I Name Formula MEG C7P ##STR00002## MEG C7S ##STR00003## MEG C6P ##STR00004## MEG C5P ##STR00005## MEG C4P ##STR00006## MEG C3P ##STR00007##
[0094] The operating conditions used for the preparation of each of the above glycerol monoethers are presented in the following Table II:
TABLE-US-00003 TABLE II Efficiency in Name Reactor Catalyst Temperature Period MEG (%) MEG B 2.5% mol 160.degree. C. 24 h 15 C7P APTS MEG ROR 2.5% mol 150.degree. C. 14 h 10 C7S APTS MEG R2L 1% mol 150.degree. C. 24 h 25 C6P APTS MEG ROR 2.5% mol 150.degree. C. 7 h 33 C5P APTS MEG MP 1% mol 150.degree. C. 24 h 15 C4P APTS MEG ROR 2.5% mol 140.degree. C. 5 h 8 C3P APTS
[0095] The reactors used are the following: [0096] B: Buchi glass reactor immersed in an oil bath, [0097] ROR: Fast opening reactor with a stirring propeller of 220 mL, [0098] MP: Multi-reactors consisting of six autoclaves of 75 mL, [0099] R2L: 2L stainless steel reactor with stirring blade.
[0100] The amount of catalyst is a molar amount relative to the amount of glycerol used.
[0101] For the synthesis of each of the above glycerol monoethers: [0102] the glycerol, the catalyst and the alcohol are mixed and react together; [0103] then the mixture thus obtained is first purified by decantation without solvent, then subjected to a liquid-liquid extraction and finally subjected to fractional distillation to recover the glycerol mono-ether.
[0104] The amount of glycerol monoethers obtained at the end of the reaction is measured by gas chromatography.
[0105] TGA Measurements
[0106] Thermogravimetric analysis (TGA) gives the mass loss of a sample as a function of temperature. The sample is heated according to programmed temperature ramps. The mass losses are determined using a scale that continuously measures the mass of the sample during its rise in temperature. The results of these TGA measurements on the various glycerol monoethers above are described in Table III.
TABLE-US-00004 TABLE III % of weight loss Temperature (.degree. C.) MEG C3P 100% 169.5.degree. C. MEG C4P 100% 186.6.degree. C. MEG C5P 100% 192.4.degree. C. MEG C6P 100% 216.1.degree. C. MEG C7P 100% 222.9.degree. C. MEG C7S 100% 195.1.degree. C.
[0107] Thus short glycerol monoethers (MEG C3P and MEG C4P) have a loss of mass of 100% for lower temperatures, and especially less than 190.degree. C. which makes them difficult to use in an engine application or even incompatible with such an application.
[0108] Conversely, the glycerol monoethers according to the invention (MEG CSP, MEG C6P, MEG C7P and MEG C7S) have a loss of mass of 100% for higher temperatures, and, in particular, may be greater than 200.degree. C. which makes them fully compatible with use in a lubricating composition for a engine application.
[0109] Characterization Tests
[0110] In order to characterize the behavior of the compositions according to the invention, two series of tests were performed.
[0111] The HFRR (High Frequency Reciprocating Rig) test is used to evaluate the performance of fuels and lubricants in terms of friction in the mixed/limit regime.
[0112] This test involves fixing a steel plane, on which the lubricating composition to be tested is placed, in a heating block. A steel ball subjected to a certain load is then brought into contact with the lubricating composition and the steel plane before being vibrated. The vibration frequency and the load applied to the ball as well as the temperature at which the lubricating composition is subjected are adjustable. This test makes it possible to obtain a coefficient of friction curve as a function of the duration of the test.
[0113] In practice, the test lasts 30 min, the ball travels back and forth 2 mm at a frequency of 20 Hz, an average shear rate of 40 mm.s.sup.-1 (limit regime). At the stop point of the ball, the speed is zero which places this test well in a limited and mixed lubrication regime. A load of 200 g is applied on the ball, which corresponds to a pressure of 800 MPa. The oil is heated to 100.degree. C.
[0114] The MTM test (Traction Machine or Mini Traction Machine) makes it possible to evaluate the performance of lubricants in terms of friction in the mixed/hydrodynamic regime. This test consists in setting in relative motion a steel ball and a steel plane, at different speeds, allowing definition of the % SSR (ratio of the speed of slip/drive speed or Slide-to-Roll Ratio) which corresponds at the slip speed/drive speed.
[0115] The tests consist of a 120 min accumulation period where the ball rotates at 100 mm.s.sup.-1 (hydrodynamic regime) with a % SSR of 50%, for a load of 1.1 GPa and an oil temperature 100.degree. C.
[0116] A friction modifier is considered effective if it lowers the coefficient of friction. The different glycerol monoethers are tested in a reference lubricating composition whose composition is given in Table IV below.
TABLE-US-00005 TABLE IV Description Content (%) Base oil 1 Group III (kinematic 59.4 viscosity measured at 100.degree. C. according to ASTM D556 = 4 mm.sup.2/s), Base oil 2 Group IV (kinematic 20 viscosity measured at 100.degree. C. according to ASTM D556 = 6 mm.sup.2/s) Viscosity modifier Poly(isoprene-styrene- 6.6 hydrogenated) Pour point depressant polymer poly (alkylmethacrylate) 0.2 Antioxidant Diphenylamine 1.5 Additive package P6003 .RTM. marketed by the 12.3 company Infineum
[0117] Each glycerol mono-ether is added at a content of 1% by weight relative to the total weight of the reference lubricating composition. The various lubricating compositions tested are described in Table V below.
TABLE-US-00006 TABLE V Composition 1 Composition 2 Composition 3 Composition 4 Composition 5 Composition 6 Composition 7 Reference 99 99 99 99 99 99 99 composition MEG C3P 1 MEG C4P 1 MEG C5P 1 MEG C6P 1 MEG C7P 1 MEG C7S 1 Glycerol 1 mono- oleate
Example 1
HFRR Tests
[0118] Table VI below represents the coefficients of friction of the reference composition and compositions 3, 4, 5 and 6; the values were taken after 900 seconds of testing.
TABLE-US-00007 TABLE VI Reference lubricating composition Composition 3 Composition 4 Composition 5 Composition 6 Coefficient 0.131 0.112 0.119 0.119 0.119 of friction at 900 seconds
[0119] Thus, all of the lubricating compositions according to the invention (compositions 3 to 6) significantly lower the coefficient of friction on this HFRR test under severe conditions.
Example 2
MTM Tests
[0120] As previously described, the MTM tests will probe the performance of the friction modifiers under less severe conditions than the HFRR test but are nevertheless representative of operating points of certain driving members. Table VII below indicates the values of the coefficients of friction of compositions 1, 2, 4, 5, 6 and 7; the values were taken after 6120 seconds of testing.
TABLE-US-00008 TABLE VII Coefficient of friction Composition 1 0.063 Composition 2 0.045 Composition 4 0.041 Composition 5 0.039 Composition 6 0.040 Composition 7 0.045
[0121] Thus, the lubricating compositions 4 to 6 according to the invention significantly lower the coefficient of friction on this MTM test in comparison with the comparative compositions 1 and 2 (comprising respectively a C.sub.3 and C.sub.4 glycerol mono-ether) or comparative composition 7 (comprising glycerol mono-oleate)).
Example 3
Engine Tests
[0122] In order to confirm the good results expressed on tribological tests, the composition 3 and the composition 6 were evaluated comparatively by the implementation of the motor test described below:
[0123] During the test, the fluid temperature (water/oil) is regulated. The engine is then positioned at a selected operating point (rpm/torque) and the fuel consumption is then measured at this point. A consumption map is thus produced by this means. The test engine is a Renault R9M engine.
[0124] Table VIII shows the gains in terms of % fuel consumption compared to the reference composition at a temperature (water/oil) set at 90.degree. C. The threshold of significance of the test is equal to 0.15%.
TABLE-US-00009 TABLE VIII Regime Couple Composition 6 Composition 3 2500 160 0.14 0.31 2000 160 0.24 0.44 1500 160 0.19 0.35 2500 90 0.37 0.61 2000 90 0.06 0.50 1500 90 0.28 0.60 2500 40 0.42 0.88 2000 40 0.58 0.82 1500 40 0.71 0.79 849 2 1.96 0.95 Average heat 0.49 0.62 gain
[0125] Thus the lubricating compositions according to the invention significantly improve the fuel consumption, especially when hot.
* * * * *








XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.