Fused Indazole Pyridone Compounds As Antivirals
FU; Jiping ; et al.
U.S. patent application number 16/383382 was filed with the patent office on 2019-10-03 for fused indazole pyridone compounds as antivirals. The applicant listed for this patent is Novartis AG. Invention is credited to Jiping FU, Wooseok HAN, Subramanian KARUR, Peichao LU, Keith Bruce PFISTER, Joseph Michael YOUNG.
| Application Number | 20190300530 16/383382 |
| Document ID | / |
| Family ID | 62148437 |
| Filed Date | 2019-10-03 |

View All Diagrams
| United States Patent Application | 20190300530 |
| Kind Code | A1 |
| FU; Jiping ; et al. | October 3, 2019 |
FUSED INDAZOLE PYRIDONE COMPOUNDS AS ANTIVIRALS
Abstract
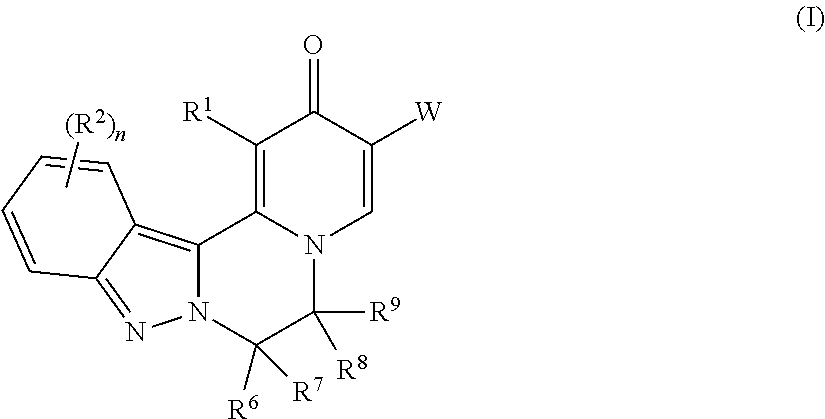
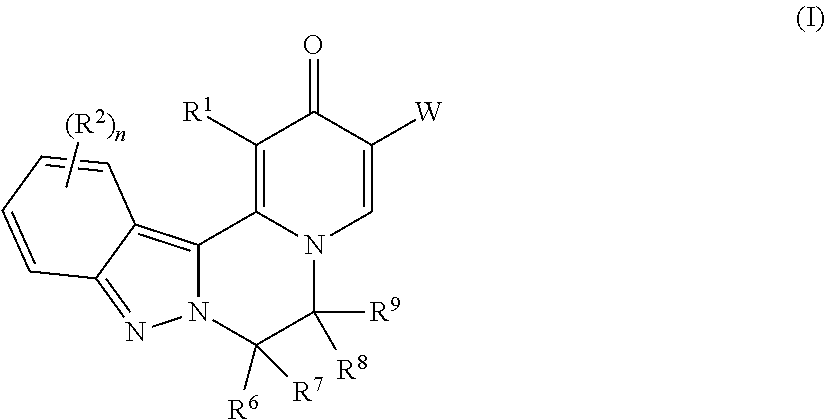
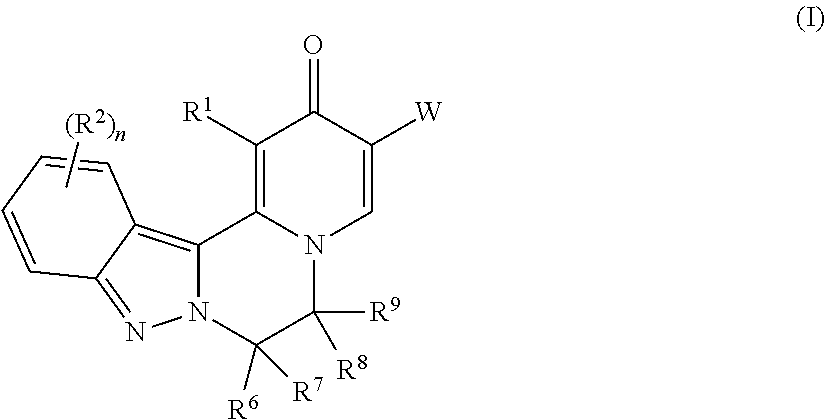
The invention provides compounds of Formula (I) ##STR00001## as described herein, along with pharmaceutically acceptable salts, pharmaceutical compositions containing such compounds, and methods to use these compounds, salts and compositions for treating viral infections, particularly infections caused by hepatitis B virus, and reducing the occurrence of serious conditions associated with HBV.
| Inventors: | FU; Jiping; (Danville, CA) ; HAN; Wooseok; (Cambridge, MA) ; KARUR; Subramanian; (Dublin, CA) ; LU; Peichao; (Pleasant Hill, CA) ; PFISTER; Keith Bruce; (San Ramon, CA) ; YOUNG; Joseph Michael; (Pleasant Hill, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 62148437 | ||||||||||
| Appl. No.: | 16/383382 | ||||||||||
| Filed: | April 12, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15965172 | Apr 27, 2018 | 10301312 | ||
| 16383382 | ||||
| 62490890 | Apr 27, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 31/4985 20130101; C07D 471/14 20130101; C07D 401/12 20130101; C07D 405/12 20130101; A61K 45/06 20130101; A61P 31/12 20180101; C07D 471/22 20130101; C07D 491/16 20130101 |
| International Class: | C07D 471/14 20060101 C07D471/14; C07D 401/12 20060101 C07D401/12; C07D 491/16 20060101 C07D491/16; A61P 31/12 20060101 A61P031/12; A61K 45/06 20060101 A61K045/06; C07D 471/22 20060101 C07D471/22; A61K 31/4985 20060101 A61K031/4985; C07D 405/12 20060101 C07D405/12 |
Claims
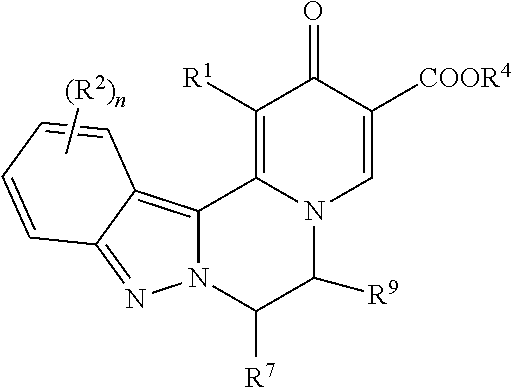
1. A compound of formula (I): ##STR00270## wherein: R.sup.1 is H or halo; R.sup.2 is independently selected at each occurrence from halo, CN, C.sub.1-C.sub.3 haloalkyl, C.sub.1-C.sub.3 haloalkoxy, --OR.sup.3, --N(R)R.sup.3, and C.sub.1-C.sub.3 alkyl optionally substituted with up to three groups selected from R.sup.3,--N(R)R.sup.3, CN, --OH, --CONR.sub.2, --COOR, and --OR.sup.3; R.sup.3 is a C.sub.1-C.sub.4 alkyl that is optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 haloalkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, --COOR, --CONR.sub.2, C.sub.3-C.sub.5 cycloalkyl, and a 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl and 4-6 membered cyclic ether are each optionally substituted with one or two groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, and C.sub.1-C.sub.3 alkyl; or R.sup.3 is a C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether are each optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 alkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, C.sub.1-C.sub.3 haloalkyl, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), and --N(C.sub.1-C.sub.3 alkyl).sub.2; n is 0, 1, or 2; W is --COOR.sup.4, --C(O)NH--SO.sub.2R.sup.5, --C(O)NH--SO.sub.2NR.sub.2, 5-tetrazolyl, or 1,2,4-oxadiazol-3-yl-5 (4H)-one; R.sup.4 is H or C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2; R.sup.5 is C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2; R.sup.6 is H or C.sub.1-C.sub.6 alkyl; R.sup.7 is H or C.sub.1-C.sub.6 alkyl, or R.sup.7 taken together with R.sup.9 and the atoms connecting R.sup.7 with R.sup.9 forms a ring as described below; R.sup.8 is H or C.sub.1-C.sub.6 alkyl; R.sup.9 is selected from: (1) H; (2) C.sub.1-C.sub.6 alkyl optionally substituted with up to three groups selected from C.sub.3-C.sub.6 cycloalkyl, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo; and (3) a ring selected from (a) C.sub.3-C.sub.6 cycloalkyl, (b) phenyl, (c) 5-6 membered heterocyclyl containing one or two heteroatoms selected from N, O and S as ring members, and (d) 5-6 membered heteroaryl containing one or two heteroatoms selected from N, O and S as ring members, wherein each of rings (a) through (d) is optionally substituted with up to three groups selected from C.sub.1-C.sub.2 alkyl, (CH.sub.2).sub.0-2--OR, --NR.sub.2, halo, CN, COOR, and CONR.sub.2; or R.sup.9 taken together with R.sup.7 and the atoms connecting R.sup.9 with R.sup.7 forms a 3-7 membered cycloalkyl ring, or a 3-7 membered heterocyclic ring containing N, O or S as a ring member; wherein the cycloalkyl or heterocyclic ring is optionally substituted with up to three groups selected from R, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo; R is independently selected at each occurrence from H and C.sub.1-C.sub.3 alkyl optionally substituted with one to three groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, and cyclopropyl; and two R groups directly attached to the same atom can optionally be taken together with the atom to which both are attached to form a 3-6 membered ring that can optionally contain a heteroatom selected from N, O and S as a ring member, and can be substituted by up to two groups selected from --OH, oxo, C.sub.1-C.sub.3 alkyl, and C.sub.1-C.sub.3 alkoxy; or a pharmaceutically acceptable salt thereof.
2. A compound according to claim 1, wherein the compound is of the formula: ##STR00271## or a pharmaceutically acceptable salt thereof.
3. A compound according to claim 1, which is of the formula: ##STR00272## or a pharmaceutically acceptable salt thereof.
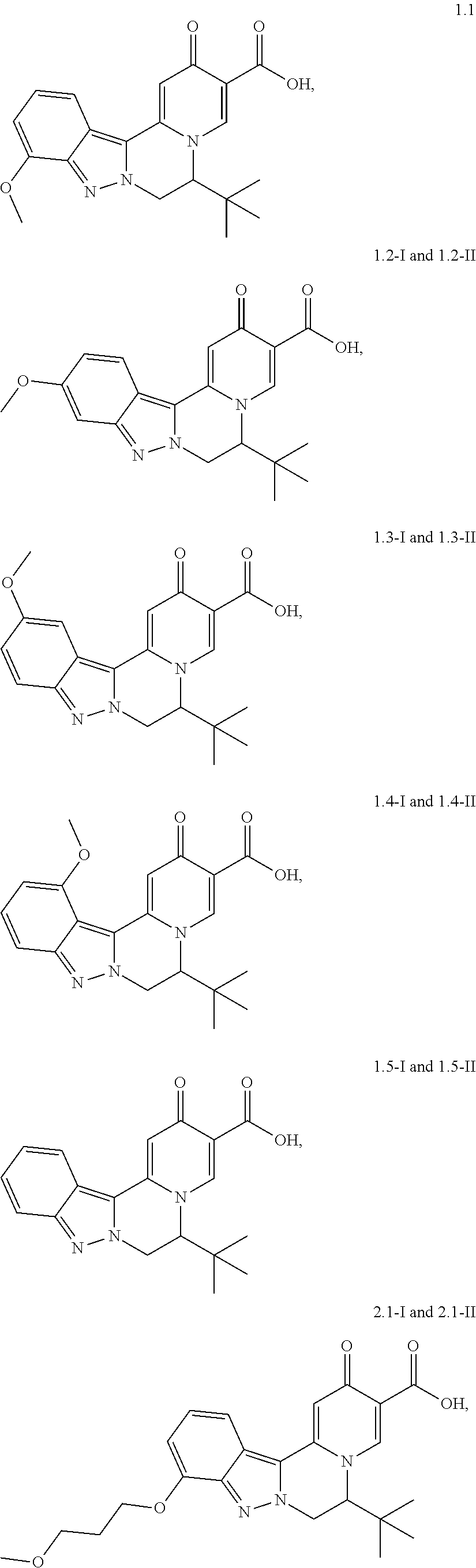
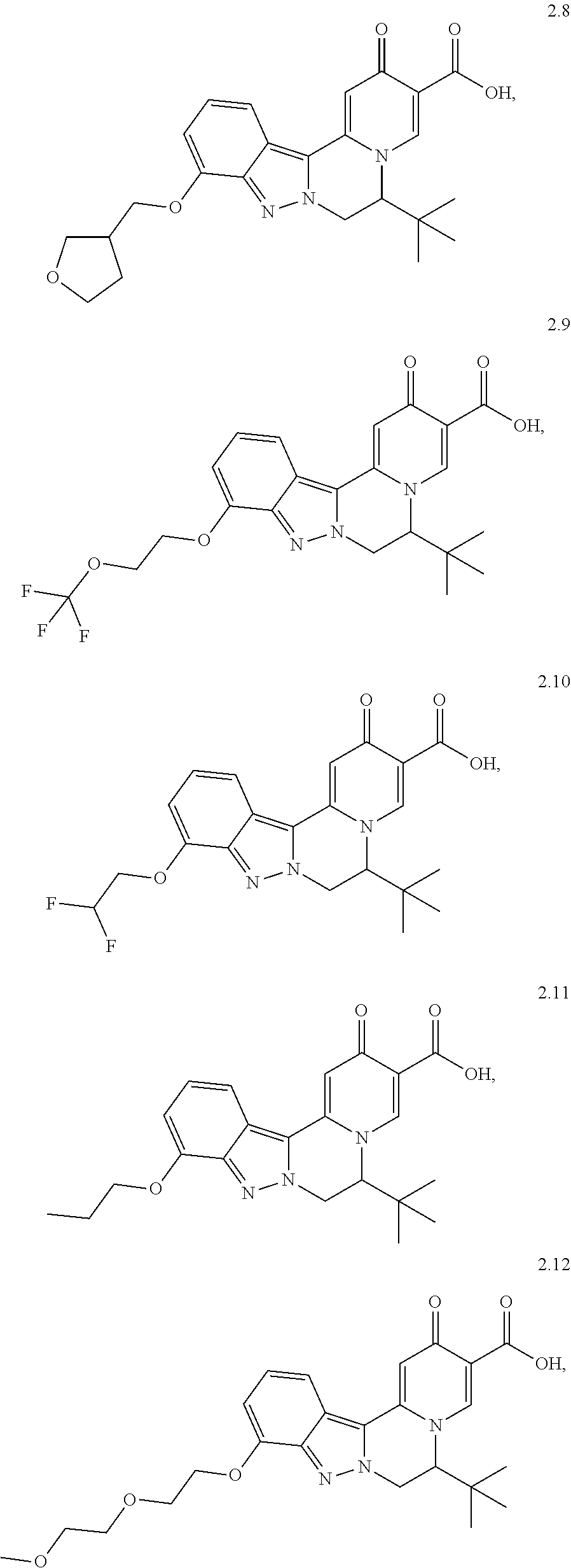
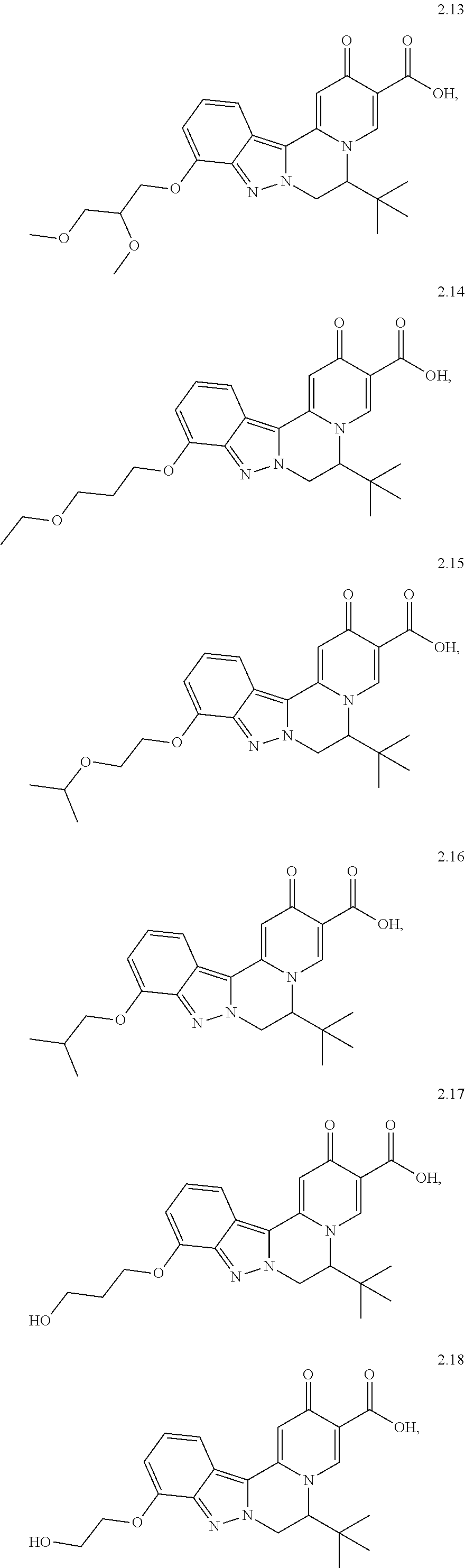
4. The compound of claim 1, which is selected from: ##STR00273## ##STR00274## ##STR00275## ##STR00276## ##STR00277## ##STR00278## ##STR00279## ##STR00280## ##STR00281## ##STR00282## ##STR00283## and the individual enantiomers of any one of these; or a pharmaceutically acceptable salt thereof.
5. A pharmaceutical composition, comprising a compound of claim 1 admixed with at least one pharmaceutically acceptable carrier.
6. A method to treat a subject having a hepatitis B infection, which comprises administering to the subject a compound of claim 1.
7. A method to inhibit replication of hepatitis B virus, which comprises contacting the hepatitis B virus, either in vitro or in vivo, with a compound according to claim 1.
8. A pharmaceutical combination, comprising a compound of claim 1 and at least one additional therapeutic agent.
9. A compound according to claim 1 for use in therapy.
10. Use of a compound according claim 1 in the manufacture of a medicament.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. Ser. No. 15/965,172 filed 27 Apr. 2018, which claims the benefit of priority to U.S. Ser. No. 62/490,890, filed 27 Apr. 2017, the contents of which is incorporated herein by reference.
FIELD OF THE INVENTION
[0002] The present invention relates to novel fused tetracyclic pyridone compounds that are inhibitors of hepatitis virus replication, and are thus useful to treat viral infections, and particularly hepatitis B virus (HBV). The invention provides novel tetracyclic pyridone compounds as disclosed herein, pharmaceutical compositions containing such compounds, and methods of using these compounds and compositions in the treatment and prevention of HBV infections.
BACKGROUND
[0003] Globally, over 400 million people are chronically infected with hepatitis B virus (HBV), and more than 12 million reside in the United States alone. Of those chronically infected patients, up to 40 percent will eventually develop complications of liver failure from cirrhosis or development of hepatocellular carcinoma (HCC). HBV belongs to the family of Hepadnaviridae, a group of small hepatotropic DNA viruses that replicate through the reverse transcription of an RNA intermediate. The 3.2-kb HBV genome in viral particles is in a circular, partially double-stranded DNA conformation (relaxed circular DNA or rcDNA). The HBV genome consists of four overlapping open reading frames (ORFs), which encode for the core, polymerase (Pol), envelope, and X proteins. rcDNA is transcriptionally inert and must be converted into covalently closed circular DNA (cccDNA) in the nucleus of infected cells before viral RNAs can be transcribed. cccDNA is the only template for HBV transcription and, because HBV RNA templates genomic reverse transcription, its persistence is required for persistent infection.
[0004] The envelope of HBV comprises a mixture of surface antigen proteins (HBsAg). The HBsAg coat is a mixture of three overlapping proteins: all three share a common region, which corresponds to the smallest of the three proteins (SHBsAg). The mixture consists mostly of SHBsAg, but also includes Medium HBsAg, which comprises SHBsAg plus an additional polypeptide segment, and Large HBsAg, which comprises M HBsAg plus another added polypeptide segment. In addition to forming the infectious virion particle, the S, M and L HBsAg proteins also assemble into a subviral particle knows as the 22-nm particle, which is not infectious but contains the same proteins that envelope the infectious virus particles. Indeed, these subviral, non-infectious particles have been used as a vaccine, since they contain the same antigenic surface proteins as the infectious HBV virion, and thus elicit antibodies that recognize the infectious agent. Interestingly, these subviral particles greatly outnumber infectious virions, and are believed to protect the infectious virions from the immune system of the infected host. By sheer numbers, they may act as decoys, distracting immune responses from the infectious virus particles, but in addition they are reported to suppress the function of immune cells (monocytes, dendritic cells and natural killer cells) and may thus impair the immune response to HBV. Because these subviral particles protect infectious HBV from the host immune system, reducing the level of subviral particles has been recognized as a viable therapeutic approach. See, e.g., WO2015/113990.
[0005] One of the key diagnostic symptoms of chronic HBV is the high serum levels of the hepatitis B surface antigen (HBsAg). Clinical data in recent years suggest that sustained virologic response is often associated with on-treatment HBsAg decline during the early phase of the treatment as early as week 8, while sustained exposure to HBsAg and other viral antigens may lead to HBV-specific immune-tolerance. Chronic HB patients who experienced larger and faster decreases in serum HBsAg levels achieved significantly higher rate (.about.40%) of sustained virologic response as defined by sustained viral control post treatment.
[0006] Current treatment options for HBV include interferon therapies and nucleoside/nucleotide inhibitors of the viral DNA polymerase, such as entecavir and tenofovir. These focus on reduction in the level of viremia and toleration of hepatic dysfunction, and may have adverse side-effects and also select for drug-resistant virus variants during long term therapy. More importantly, these therapies cannot eradicate the intrahepatic HBV cccDNA pool in chronic hepatitis B patients or limit the transcription of HBsAg from the pre-existing cccDNA, nor do they affect the secretion of synthesized HBsAg into patients' blood to counteract the host innate immune response. As a result, these HBV treatments are in most cases life-long therapies, and discontinuation often leads to virological relapse.
[0007] Accordingly, there remains a need for more effective treatments for HBV, especially for treating chronic HBV infections (cHBV). The invention provides compounds that are believed to operate by suppression of the secretion of the 22 nm subviral particles containing HBsAg. These compounds are useful to treat HBV infections and to reduce the incidence of serious liver disorders caused by HBV infections.
SUMMARY
[0008] The present invention provides novel compounds that inhibit secretion of HBsAg from cells infected with hepatitis B virus and thereby reduce viral load and viral replication in patients having chronic HBV infection. Thus the compounds of the invention are suitable for treatment of patients with HBV, including chronic HBV.
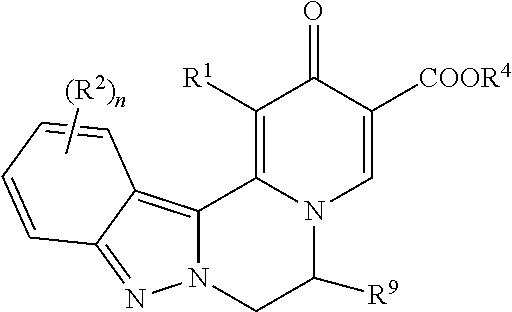
[0009] In one aspect, the invention provides compounds of Formula (I):
##STR00002##
[0010] wherein:
[0011] R.sup.1 is H or halo;
[0012] R.sup.2 is independently selected at each occurrence from halo, CN, C.sub.1-C.sub.3 haloalkyl, C.sub.1-C.sub.3 haloalkoxy, --OR.sup.3, --N(R)R.sup.3, and C.sub.1-C.sub.3 alkyl optionally substituted with up to three groups selected from R.sup.3, --N(R)R.sup.3, CN, --OH, --CONR.sub.2, --COOR, and --OR.sup.3;
[0013] R.sup.3 is a C.sub.1-C.sub.4 alkyl that is optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 haloalkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, --COOR, --CONR.sub.2, C.sub.3-C.sub.5 cycloalkyl, and a 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl and 4-6 membered cyclic ether are each optionally substituted with one or two groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, and C.sub.1-C.sub.3 alkyl;
[0014] or R.sup.3 is a C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether are each optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 alkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, C.sub.1-C.sub.3 haloalkyl, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), and --N(C.sub.1-C.sub.3 alkyl).sub.2;
[0015] n is 0, 1, or 2;
[0016] W is --COOR.sup.4, --C(O)NH--SO.sub.2R.sup.5, --C(O)NH--SO.sub.2NR.sub.2, 5-tetrazolyl, or 1,2,4-oxadiazol-3-yl-5(4H)-one;
[0017] R.sup.4 is H or C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2;
[0018] R.sup.5 is C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2;
[0019] R.sup.6 is H or C.sub.1-C.sub.6 alkyl;
[0020] R.sup.7 is H or C.sub.1-C.sub.6 alkyl,
[0021] or R.sup.7 taken together with R.sup.9 and the atoms connecting R.sup.7 with R.sup.9 forms a ring as described below;
[0022] R.sup.8 is H or C.sub.1-C.sub.6 alkyl;
[0023] R.sup.9 is selected from: H;
[0024] C.sub.1-C.sub.6 alkyl optionally substituted with up to three groups selected from C.sub.3-C.sub.6 cycloalkyl, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo; and
[0025] a ring selected from (a) C.sub.3-C.sub.6 cycloalkyl, (b) phenyl, (c) 5-6 membered heterocyclyl containing one or two heteroatoms selected from N, O and S as ring members, and (d) 5-6 membered heteroaryl containing one or two heteroatoms selected from N, O and S as ring members, wherein each of rings (a) through (d) is optionally substituted with up to three groups selected from C.sub.1-C.sub.2 alkyl, (CH.sub.2).sub.0-2--OR, --NR.sub.2, halo, CN, COOR, and CONR.sub.2;
[0026] or R.sup.9 taken together with R.sup.7 and the atoms connecting R.sup.9 with R.sup.7 forms a 3-7 membered cycloalkyl ring, or a 3-7 membered heterocyclic ring containing N, O or S as a ring member; wherein the cycloalkyl or heterocyclic ring is optionally substituted with up to three groups selected from R, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo;
[0027] R is independently selected at each occurrence from H and C.sub.1-C.sub.3 alkyl optionally substituted with one to three groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, and cyclopropyl;
[0028] and two R groups directly attached to the same atom can optionally be taken together with the atom to which both are attached to form a 3-6 membered ring that can optionally contain a heteroatom selected from N, O and S as a ring member, and can be substituted by up to two groups selected from --OH, oxo, C.sub.1-C.sub.3 alkyl, and C.sub.1-C.sub.3 alkoxy;
[0029] or a pharmaceutically acceptable salt thereof.
[0030] The invention also includes pharmaceutical compositions containing these compounds, methods to use these compounds and compositions to treat viral infections, pharmaceutical combinations comprising these compounds, and methods to use the compounds in the manufacture of a medicament.
DETAILED DESCRIPTION
[0031] For purposes of interpreting this specification, the following definitions will apply, and whenever appropriate, terms used in the singular will also include the plural.
[0032] Terms used in the specification have the following meanings unless the context clearly indicates otherwise:
[0033] As used herein, the term "subject" refers to an animal. In certain aspects, the animal is a mammal. A subject also refers to for example, primates (e.g., humans), cows, sheep, goats, horses, dogs, cats, rabbits, rats, mice, fish, birds and the like. In certain embodiments, the subject is a human. A "patient" as used herein refers to a human subject.
[0034] As used herein, the term "inhibition" or "inhibiting" refers to the reduction or suppression of a given condition, symptom, or disorder, or disease, or a significant decrease in the baseline activity of a biological activity or process.
[0035] As used herein, the term "treating" or "treatment" of any disease or disorder refers in one embodiment, to ameliorating the disease or disorder (i.e., slowing or arresting or reducing the development of the disease or at least one of the clinical symptoms thereof). In another embodiment "treating" or "treatment" refers to alleviating or ameliorating at least one physical parameter including those which may not be discernible by the patient. In yet another embodiment, "treating" or "treatment" refers to modulating the disease or disorder, either physically, (e.g., stabilization of a discernible symptom), physiologically, (e.g., stabilization of a physical parameter), or both. In yet another embodiment, "treating" or "treatment" refers to preventing or delaying the onset or development or progression of the disease or disorder.
[0036] As used herein, the term "a," "an," "the" and similar terms used in the context of the present invention (especially in the context of the claims) are to be construed to cover both the singular and plural unless otherwise indicated herein or clearly contradicted by the context.
[0037] All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g. "such as") provided herein is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention otherwise claimed.
[0038] "Optionally substituted" means the group referred to can be substituted at one or more positions by any one or any combination of the radicals listed thereafter. The number, placement and selection of substituents is understood to encompass only those substitutions that a skilled chemist would expect to be reasonably stable; thus `oxo` would not be a substituent on an aryl or heteroaryl ring, for example, and a single carbon atom would not have three hydroxy or amino substituents. Unless otherwise specified, optional substituents are typically up to four groups selected from halo, oxo, CN, amino, hydroxy, --C.sub.1-3 alkyl, --OR*, --NR*.sub.2, --SR*, --SO.sub.2R*, --COOR*, and --CONR*.sub.2, where each R* is independently H or C.sub.1-3 alkyl.
[0039] "Aryl" as used herein refers to a phenyl or naphthyl group unless otherwise specified. Aryl groups unless otherwise specified may be optionally substituted with up to four groups selected from halo, CN, amino, hydroxy, C.sub.1-3 alkyl, --OR*, --NR*.sub.2, --SR*, --SO.sub.2R*, --COOR*, and --CONR*.sub.2, where each R* is independently H or C.sub.1-3 alkyl.
[0040] "Halo" or "halogen", as used herein, may be fluorine, chlorine, bromine or iodine.
[0041] "C.sub.1-6 alkyl" or "C.sub.1-C.sub.6 alkyl", as used herein, denotes straight chain or branched alkyl having 1-6 carbon atoms. If a different number of carbon atoms is specified, such as C.sub.4 or C.sub.3, then the definition is to be amended accordingly, such as "C.sub.1-4 alkyl" will represent methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl and tert-butyl.
[0042] "C.sub.1-6 alkylene" or "C.sub.1-C.sub.6 alkylene", as used herein, denotes straight chain or branched alkyl having 1-6 carbon atoms and two open valences for connection to two other groups. If a different number of carbon atoms is specified, such as C.sub.4 or C.sub.3, then the definition is to be amended accordingly, such as "C.sub.1-4 alkylene" will represent methylene (--CH.sub.2--), ethylene (--CH.sub.2CH.sub.2--), straight chain or branched propylene (--CH.sub.2CH.sub.2CH.sub.2-- or --CH.sub.2--CHMe-CH.sub.2--), and the like.
[0043] "C.sub.1-6 alkoxy", as used herein, denotes straight chain or branched alkoxy (--O-Alkyl) having 1-6 carbon atoms. If a different number of carbon atoms is specified, such as C.sub.4 or C.sub.3, then the definition is to be amended accordingly, such as "C.sub.1-4 alkoxy" will represent methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, sec-butoxy and tert-butoxy.
[0044] "C.sub.1-4 Haloalkyl" or "C.sub.1-C.sub.4 haloalkyl" as used herein, denotes straight chain or branched alkyl having 1-4 carbon atoms wherein at least one hydrogen has been replaced with a halogen. The number of halogen replacements can be from one up to the number of hydrogen atoms on the unsubstituted alkyl group. If a different number of carbon atoms is specified, such as C.sub.6 or C.sub.3, then the definition is to be amended accordingly. Thus "C.sub.1-4 haloalkyl" will represent methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl and tert-butyl that have at least one hydrogen substituted with halogen, such as where the halogen is fluorine: CF.sub.3CF.sub.2--, (CF.sub.3).sub.2CH--, CH.sub.3--CF.sub.2--, CF.sub.3CF.sub.2--, CF.sub.3, CF.sub.2H--, CF.sub.3CF.sub.2CH(CF.sub.3)-- or CF.sub.3CF.sub.2CF.sub.2CF.sub.2--.
[0045] "C.sub.3-8 cycloalkyl" as used herein refers to a saturated monocyclic hydrocarbon ring of 3 to 8 carbon atoms. Examples of such groups include cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl. If a different number of carbon atoms is specified, such as C.sub.3-C.sub.6, then the definition is to be amended accordingly.
[0046] "4- to 8-Membered heterocyclyl", "5- to 6-membered heterocyclyl", "3- to 10-membered heterocyclyl", "3- to 14-membered heterocyclyl", "4- to 14-membered heterocyclyl" and "5- to 14-membered heterocyclyl", refers, respectively, to 4- to 8-membered, 5- to 6-membered, 3- to 10-membered, 3- to 14-membered, 4- to 14-membered and 5- to 14-membered heterocyclic rings; unless otherwise specified, such rings contain 1 to 7, 1 to 5, or 1 to 3 heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur as ring members, and the rings may be saturated, or partially saturated but not aromatic. The heterocyclic group can be attached to another group at a nitrogen or a carbon atom. The term "heterocyclyl" includes single ring groups, fused ring groups and bridged groups. Examples of such heterocyclyl include, but are not limited to pyrrolidine, piperidine, piperazine, pyrrolidinone, morpholine, tetrahydrofuran, tetrahydrothiophene, tetrahydrothiopyran, tetrahydropyran, 1,4-dioxane, 1,4-oxathiane, 8-aza-bicyclo[3.2.1]octane, 3,8-diazabicyclo[3.2.1]octane, 3-Oxa-8-aza-bicyclo[3.2.1]octane, 8-Oxa-3-aza-bicyclo[3.2.1]octane, 2-Oxa-5-aza-bicyclo[2.2.1]heptane, 2,5-Diaza-bicyclo[2.2.1]heptane, azetidine, ethylenedioxo, oxetane or thiazole. In certain embodiments, if not otherwise specified, heterocyclic groups have 1-2 heteroatoms selected from N, O and S as ring members, and 4-7 ring atoms, and are optionally substituted with up to four groups selected from halo, oxo, CN, amino, hydroxy, C.sub.1-3 alkyl, --OR*, --NR*.sub.2, --SR*, --SO.sub.2R*, --COOR*, and --CONR*.sub.2, where each R* is independently H or C.sub.1-3 alkyl. In particular, heterocyclic groups containing a sulfur atom are optionally substituted with one or two oxo groups on the sulfur.
[0047] "4-6 membered cyclic ether" as used herein refers to a 4 to 6 membered ring comprising one oxygen atom as a ring member. Examples include oxetane, tetrahydrofuran and tetrahydropyran.
[0048] "Heteroaryl" is a completely unsaturated (aromatic) ring. The term "heteroaryl" refers to a 5-14 membered monocyclic- or bicyclic- or tricyclic-aromatic ring system, having 1 to 8 heteroatoms selected from N, O or S. Typically, the heteroaryl is a 5-10 membered ring or ring system (e.g., 5-7 membered monocyclic group or an 8-10 membered bicyclic group), often a 5-6 membered ring containing up to four heteroatoms selected from N, O and S, though often a heteroaryl ring contains no more than one divalent O or S in the ring. Typical heteroaryl groups include furan, isothiazole, thiadiazole, oxadiazole, indazole, indole, quinoline, 2- or 3-thienyl, 2- or 3-furyl, 2- or 3-pyrrolyl, 2-, 4-, or 5-imidazolyl, 3-, 4-, or 5-pyrazolyl, 2-, 4-, or 5-thiazolyl, 3-, 4-, or 5-isothiazolyl, 2-, 4-, or 5-oxazolyl, 3-, 4-, or 5-isoxazolyl, 3- or 5-(1,2,4-triazolyl), 4- or 5-(1,2, 3-triazolyl), tetrazolyl, triazine, pyrimidine, 2-, 3-, or 4-pyridyl, 3- or 4-pyridazinyl, 3-, 4-, or 5-pyrazinyl, 2-pyrazinyl, and 2-, 4-, or 5-pyrimidinyl. Heteroaryl groups are optionally substituted with up to four groups selected from halo, CN, amino, hydroxy, C.sub.1-3 alkyl, --OR*, --NR*.sub.2, --SR*, --SO.sub.2R*, --COOR*, and --CONR*.sub.2, where each R* is independently H or C.sub.1-3 alkyl.
[0049] The term "hydroxy" or "hydroxyl" refers to the group --OH.
[0050] Various embodiments of the invention are described herein. It will be recognized that features specified in each embodiment may be combined with other specified features to provide further embodiments.
[0051] The following enumerated embodiments are representative of the invention:
[0052] A compound of formula (I):
##STR00003##
[0053] wherein:
[0054] R.sup.1 is H or halo;
[0055] R.sup.2 is independently selected at each occurrence from halo, CN, C.sub.1-C.sub.3 haloalkyl, C.sub.1-C.sub.3 haloalkoxy, --OR.sup.3, --N(R)R.sup.3, and C.sub.1-C.sub.3 alkyl optionally substituted with up to three groups selected from R.sup.3, --N(R)R.sup.3, CN, --OH, --CONR.sub.2, --COOR, and --OR.sup.3;
[0056] R.sup.3 is a C.sub.1-C.sub.4 alkyl that is optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 haloalkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, --COOR, --CONR.sub.2, C.sub.3-C.sub.5 cycloalkyl, and a 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl and 4-6 membered cyclic ether are each optionally substituted with one or two groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, and C.sub.1-C.sub.3 alkyl;
[0057] or R.sup.3 is a C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether, wherein the C.sub.3-C.sub.5 cycloalkyl ring or 4-6 membered cyclic ether are each optionally substituted with one to three groups selected from halo, C.sub.1-C.sub.3 alkyl, --OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 haloalkoxy, C.sub.1-C.sub.3 haloalkyl, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), and --N(C.sub.1-C.sub.3 alkyl).sub.2;
[0058] n is 0, 1, or 2;
[0059] W is --COOR.sup.4, --C(O)NH--SO.sub.2R.sup.5, --C(O)NH--SO.sub.2NR.sub.2, 5-tetrazolyl, or 1,2,4-oxadiazol-3-yl-5(4H)-one;
[0060] R.sup.4 is H or C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2;
[0061] R.sup.5 is C.sub.1-C.sub.6 alkyl that is optionally substituted with one to three groups selected from halo, --OR, oxo, CN, and --NR.sub.2;
[0062] R.sup.6 is H or C.sub.1-C.sub.6 alkyl;
[0063] R.sup.7 is H or C.sub.1-C.sub.6 alkyl, or R.sup.7 taken together with R.sup.9 and the atoms connecting R.sup.7 with R.sup.9 forms a ring as described below;
[0064] R.sup.8 is H or C.sub.1-C.sub.6 alkyl;
[0065] R.sup.9 is selected from:
[0066] H;
[0067] C.sub.1-C.sub.6 alkyl optionally substituted with up to three groups selected from C.sub.3-C.sub.6 cycloalkyl, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo; and
[0068] a ring selected from (a) C.sub.3-C.sub.6 cycloalkyl, (b) phenyl, (c) 5-6 membered heterocyclyl containing one or two heteroatoms selected from N, O and S as ring members, and (d) 5-6 membered heteroaryl containing one or two heteroatoms selected from N, O and S as ring members, wherein each of rings (a) through (d) is optionally substituted with up to three groups selected from C.sub.1-C.sub.2 alkyl, (CH.sub.2).sub.0-2--OR, --NR.sub.2, halo, CN, COOR, and CONR.sub.2;
[0069] or R.sup.9 taken together with R.sup.7 and the atoms connecting R.sup.9 with R.sup.7 forms a 3-7 membered cycloalkyl ring, or a 3-7 membered heterocyclic ring containing N, O or S as a ring member; wherein the cycloalkyl or heterocyclic ring is optionally substituted with up to three groups selected from R, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo;
[0070] R is independently selected at each occurrence from H and C.sub.1-C.sub.3 alkyl optionally substituted with one to three groups selected from halo, --OH, C.sub.1-C.sub.3 alkoxy, oxo, CN, --NH.sub.2, --NH(C.sub.1-C.sub.3 alkyl), --N(C.sub.1-C.sub.3 alkyl).sub.2, and cyclopropyl;
[0071] and two R groups directly attached to the same atom can optionally be taken together with the atom to which both are attached to form a 3-6 membered ring that can optionally contain a heteroatom selected from N, O and S as a ring member, and can be substituted by up to two groups selected from --OH, oxo, C.sub.1-C.sub.3 alkyl, and C.sub.1-C.sub.3 alkoxy;
[0072] or a pharmaceutically acceptable salt thereof.
[0073] When R.sup.3 is a cyclic ether in this embodiment, it is typically selected from oxetanyl, tetrahydrofuranyl, and tetrahydropyranyl.
[0074] 2. The compound of embodiment 1, or a pharmaceutically acceptable salt thereof, wherein R.sup.1 is H or F.
[0075] 3. The compound according to any one of the preceding embodiments, or a pharmaceutically acceptable salt thereof, wherein R.sup.2 is --OR.sup.3.
[0076] 4. The compound according to any one of embodiments 1 to 3, wherein W is --COOR.sup.4; or a pharmaceutically acceptable salt thereof. In preferred compounds of this embodiment, W is --COOH.
[0077] 5. The compound of any of embodiments 1-4, wherein R.sup.6 is H and R.sup.8 is H; or a pharmaceutically acceptable salt thereof.
[0078] 6. The compound of any of embodiments 1-5, wherein R.sup.9 is C.sub.1-C.sub.6 alkyl optionally substituted with up to two groups selected from C.sub.3-C.sub.6 cycloalkyl, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo; or R.sup.9 is cyclopropyl, cyclobutyl, or oxetanyl and is optionally substituted by one or two groups selected from methyl, halo, and (CH.sub.2).sub.0-2--OR;
[0079] or a pharmaceutically acceptable salt thereof.
[0080] 7. The compound of any of embodiments 1-5, wherein R.sup.9 is taken together with R.sup.7 and the atoms connecting R.sup.9 to R.sup.7 to form a 5-6 membered cycloalkyl ring or a 5-6 membered heterocyclic ring containing N, O or S as a ring member; wherein the cycloalkyl or heterocyclic ring is optionally substituted with up to two groups selected from R, --OR, --NR.sub.2, halo, CN, COOR, CONR.sub.2, and oxo;
[0081] or a pharmaceutically acceptable salt thereof.
[0082] 8. The compound of any of embodiments 1-7, wherein n is 1; or a pharmaceutically acceptable salt thereof.
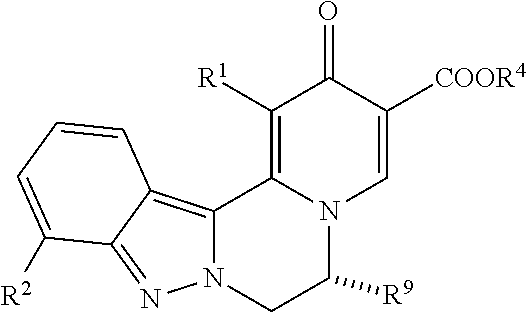
[0083] 9. The compound of any one of embodiments 7-8, which is of the formula:
##STR00004##
[0084] wherein:
[0085] R.sup.9 taken together with R.sup.7 and the atoms connecting R.sup.9 with R.sup.7 forms a 5 or 6 membered cycloalkyl ring or a 5 or 6 membered heterocyclic ring containing O as a ring member; wherein the cycloalkyl or heterocyclic ring is optionally substituted with one or two groups selected from R, --OR, --NR.sub.2, halo, and CN; and
[0086] R.sup.4 is H or C.sub.1-C.sub.4 alkyl;
[0087] or a pharmaceutically acceptable salt thereof.
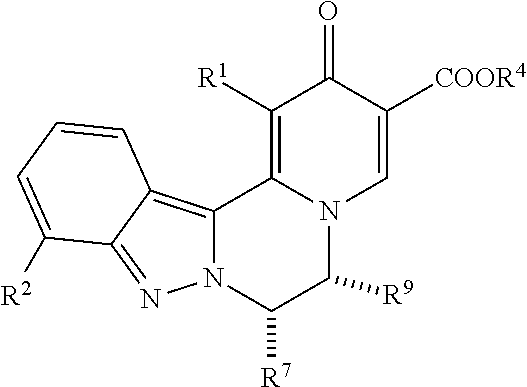
[0088] 10. The compound of any of embodiments 1-6, which is of the formula:
##STR00005##
[0089] wherein R.sup.4 is H or C.sub.1-C.sub.4 alkyl; and
[0090] R.sup.9 is C.sub.1-C.sub.4 alkyl optionally substituted with hydroxy or methoxy, or R.sup.9 is C.sub.3-C.sub.4 cycloalkyl optionally substituted with methyl or (CH.sub.2).sub.0-2--OR;
[0091] or a pharmaceutically acceptable salt thereof.
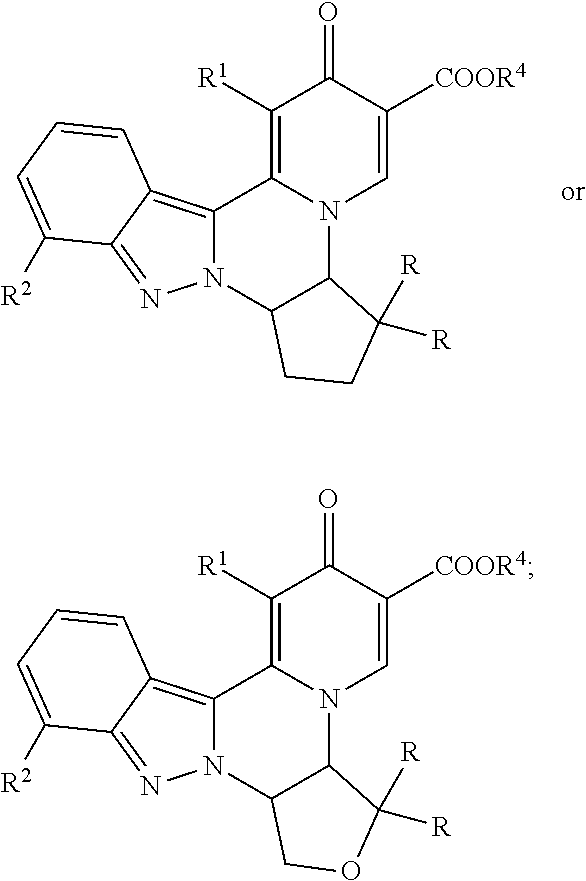
[0092] 11. A compound according to embodiment 10, which is of the formula:
##STR00006##
[0093] or a pharmaceutically acceptable salt thereof.
[0094] 12. A compound according to embodiment 8, wherein the compound is of the formula:
##STR00007##
[0095] or a pharmaceutically acceptable salt thereof.
[0096] 13. A compound according to embodiment 8, which is of the formula:
##STR00008##
[0097] or a pharmaceutically acceptable salt thereof. In compounds of this embodiment, R is preferably methyl, or two R groups on the same carbon taken together form a ring selected from cyclopropyl, cyclobutyl and oxetanyl.
[0098] 14. The compound of any one of embodiments 1-13, wherein R.sup.1 is H; or a pharmaceutically acceptable salt thereof.
[0099] 15. The compound of any one of embodiments 1-13, wherein R.sup.1 is F; or a pharmaceutically acceptable salt thereof.
[0100] 16. The compound of any one of embodiments 1-15, wherein R.sup.4 is H; or a pharmaceutically acceptable salt thereof.
[0101] 17. The compound of embodiment 1, which is selected from:
##STR00009## ##STR00010## ##STR00011## ##STR00012## ##STR00013## ##STR00014## ##STR00015## ##STR00016## ##STR00017## ##STR00018## ##STR00019##
and the individual enantiomers of any one of these; or a pharmaceutically acceptable salt thereof.
[0102] In particular, each of the compounds of the Examples, including each compound listed in Table I, is a specific embodiment of the compounds of the invention.
[0103] 18. A pharmaceutical composition, comprising a compound of any of the preceding embodiments admixed with at least one pharmaceutically acceptable carrier.
[0104] 19. A method to treat a subject having a hepatitis B infection, which comprises administering to the subject a compound of any of embodiments 1-17 or a pharmaceutical composition of claim 17.
[0105] 20. The method of embodiment 19, wherein the compound of any one of embodiments 1-17 or the pharmaceutical composition of embodiment 18 is used in combination with an additional therapeutic agent selected from an interferon or peginterferon, an HBV polymerase inhibitor, a viral entry inhibitor, a viral maturation inhibitor, a capsid assembly inhibitor, an HBV core modulator, a reverse transcriptase inhibitor, a TLR-agonist, or an immunomodulator.
[0106] 21. A method to inhibit replication of hepatitis B virus, which comprises contacting the hepatitis B virus, either in vitro or in vivo, with a compound according to any one of embodiments 1-17.
[0107] 22. A pharmaceutical combination, comprising a compound of any of embodiments 1-17 and at least one additional therapeutic agent.
[0108] 23. A compound according to any of embodiments 1-17 for use in therapy.
[0109] 24. The compound according to embodiment 23 wherein the therapy is treatment of a bacterial infection.
[0110] 25. Use of a compound according to any one of embodiments 1-17 in the manufacture of a medicament.
[0111] Another embodiment of the invention provides a compound as described above, or a pharmaceutically acceptable salt thereof, for use as a medicament. In one aspect, the medicament is for treatment of a subject having an HBV infection. In a particular embodiment, the subject is a human diagnosed with chronic HBV.
[0112] Also within the scope of this invention is the use of a compound of formula (I), or a pharmaceutically acceptable salt thereof, for the manufacture of a medicament; in some embodiments, this medicament is for the treatment or prevention of a viral disease and/or infection in a human being, particularly where the virus involved is HBV.
[0113] Included within the scope of this invention is a pharmaceutical composition, comprising a compound of Formula (I), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier or excipient. Optionally, the composition comprises at least two pharmaceutically acceptable carriers and/or excipients.
[0114] According to a further aspect of this embodiment the pharmaceutical composition according to this invention further comprises a therapeutically effective amount of at least one other antiviral agent.
[0115] The invention also provides the use of a pharmaceutical composition as described hereinabove for the treatment of a HBV infection in a human being having or at risk of having the infection.
[0116] The invention also provides the use of a pharmaceutical composition as described hereinabove for the treatment of HBV infection in a human being having or at risk of having the disease.
[0117] Another aspect of the invention involves a method of treating or preventing a hepatitis B viral disease and/or infection in a human being by administering to the human being an antivirally effective amount of a compound of the invention, a pharmaceutically acceptable salt thereof, or a composition as described above, alone or in combination with at least one other antiviral agent, administered together or separately.
[0118] An additional aspect of this invention refers to an article of manufacture comprising a composition of the invention that is effective to treat a hepatitis B viral disease and/or infection; and packaging material comprising a label which indicates that the composition can be used to treat disease and/or infection by a hepatitis B virus; wherein the composition comprises a compound of formula (I) according to this invention or a pharmaceutically acceptable salt thereof.
[0119] Still another aspect of this invention relates to a method of inhibiting the replication of HBV, comprising exposing the virus to an effective amount of the compound of formula (I), or a salt thereof, under conditions where replication of the virus is inhibited. This method can be practiced in vitro or in vivo.
[0120] Further included in the scope of the invention is the use of a compound of formula (I), or a salt thereof, to inhibit the replication of HBV.
[0121] In all of the embodiment referring to a compound of Formula (I), the compound of Formula (I) can be a compound according to any of embodiments 1-17 described above.
[0122] In some embodiments, the compound of Formula (I) is co-administered with or used in combination with at least one additional therapeutic agent selected from: an interferon or peginterferon, an HBV polymerase inhibitor, a viral entry inhibitor, a viral maturation inhibitor, a capsid assembly inhibitor, an HBV core modulator, a reverse transcriptase inhibitor, a TLR-agonist, or an immunomodulator. Optionally, the compound of Formula (I) may be prepared for simultaneous or sequential use in combination with an additional therapeutic agent; or the compound of Formula (I) may be combined into a pharmaceutical combination comprising a compound of Formula (I) and at least one additional therapeutic agent. Some particular therapeutic agents that may be used in combination with the compounds of the invention include immunomodulators described herein, interferon alfa 2a, interferon alfa-2b, pegylated interferon alfa-2a, pegylated interferon alfa-2b, TLR-7 and TLR-9 agonists, entecavir, tenofovir, cidofovir, telbivudine, didanosine, zalcitabine, stavudine, lamivudine, abacavir, emtricitabine, apricitabine, atevirapine, ribavirin, acyclovir, famciclovir, valacyclovir, ganciclovir, adefovir, efavirenz, nevirapine, delavirdine, and etravirine. Suitable core modulators are disclosed in WO2013/096744; suitable HBV capsid inhibitors are described in US2015/0252057.
[0123] These additional agents may be combined with the compounds of this invention to create a single pharmaceutical dosage form. Alternatively these additional agents may be separately administered to the patient as part of a multiple dosage form, for example, using a kit. Such additional agents may be administered to the patient prior to, concurrently with, or following the administration of a compound of the invention, or a pharmaceutically acceptable salt thereof. Alternatively, these additional therapeutic agents may be administered separately from and optionally by different routes of administration and on different dosing schedules from the compound of the invention, provided the compound of the invention and the additional therapeutic agent are used concurrently for treatment of an HBV infection or a disorder caused or complicated by an HBV infection.
[0124] The dose range of the compounds of the invention applicable per day is usually from 0.01 to 100 mg/kg of body weight, preferably from 0.1 to 50 mg/kg of body weight. Each dosage unit may conveniently contain from 5% to 95% active compound (w/w). Preferably such preparations contain from 20% to 80% active compound.
[0125] The actual pharmaceutically effective amount or therapeutic dosage will of course depend on factors known by those skilled in the art such as age and weight of the patient, route of administration and severity of disease. In any case the combination will be administered at dosages and in a manner which allows a pharmaceutically effective amount to be delivered based upon patient's unique condition.
[0126] When the composition of this invention comprises a combination of a compound of the invention and one or more additional therapeutic or prophylactic agent, both the compound and the additional agent should be present at dosage levels of between about 10 to 100%, and more preferably between about 10 and 80% of the dosage normally administered in a monotherapy regimen.
[0127] Antiviral agents contemplated for use in such combination therapy include agents (compounds or biologicals) that are effective to inhibit the formation and/or replication of a virus in a human being, including but not limited to agents that interfere with either host or viral mechanisms necessary for the formation and/or replication of a virus in a human being. Such agents can be selected from entecavir, tenofovir, cidofovir, telbivudine, didanosine, zalcitabine, stavudine, lamivudine, abacavir, emtricitabine, apricitabine, atevirapine, ribavirin, acyclovir, famciclovir, valacyclovir, ganciclovir, adefovir, efavirenz, nevirapine, delavirdine, and etravirine, and immunomodulators described herein including interferons and pegylated interferons, TLR-7 agonists, and TLR-9 agonists.
[0128] Many compounds of the invention contain one or more chiral centers. These compounds may be made and used as single isomers or as mixtures of isomers. Methods for separating the isomers, including diastereomers and enantiomers, are known in the art, and examples of suitable methods are described herein. In certain embodiments, the compounds of the invention are used as a single substantially pure isomer, meaning at least 90% of a sample of the compound is the specified isomer and less than 10% of the sample is any other isomer or mixture of isomers. Preferably, at least 95% of the sample is a single isomer. Selection of a suitable isomer is within the ordinary level of skill, as one isomer will typically be more active in the in vivo or in vitro assay described herein for measuring HBV activity, and will be the preferred isomer. Where in vitro activity differences between isomers are relatively small, e.g. less than about a factor of 4, a preferred isomer may be selected based on activity level against viral replication in cell culture, using methods such as those described herein: the isomer having a lower MIC (minimum inhibitory concentration) or EC-50 is preferred.
[0129] The compounds of the invention may be synthesized by the general synthetic routes illustrated below, specific examples of which are described in more detail in the Examples. Additional guidance for synthesis of the compounds of Formula (I) and synthetic intermediates useful for these syntheses are disclosed in published PCT applications WO2015/113990 and WO2015/173164.
[0130] Scheme 1 illustrates a general method useful to make compounds of the invention, as demonstrated in the Examples herein. A variety of indazole-3-carboxylate starting materials are known in the art. The carboxylate can be reduced to an alcohol using methods known in the art, and the alcohol can be protected with a protecting group such as known silyl ethers (e.g., TBS). The N-2 nitrogen can be alkylated with a suitable alph-haloketone to introduce the group containing R.sup.9. Reductive amination is one method to introduce nitrogen at the carbonyl center. Once the primary amine is in place, the protected alcohol at C.sub.3 of the indazole can be deprotected and oxidized to an aldehyde oxidation state, at which point it cyclizes with the primary amine to form the new 6-membered ring substituted with R.sup.9.
[0131] The imine of the new ring is then annulated to form an additional fused ring by a method known in the art, using (Z)-ethyl 2-(ethoxymethylene)-3-oxobutanoate. The new ring is then oxidized to provide the pyridone ring shown in Formula (I). Methods useful in preparing these compounds are disclosed in published PCT applications WO2015/113990 and WO2015/173164.
##STR00020##
[0132] Using this general method, other known starting materials, and the examples herein, a person of ordinary skill can synthesize compounds of Formula (I). Enantiomers of these compounds can be separated by chiral HPLC and similar known methods. Alternatively, enantiomers may be synthesized by the general synthetic routes illustrated in Scheme 2, specific examples of which are described in more detail in the Examples.
##STR00021##
[0133] While one enantiomer of compounds of this formula is typically more active than the other enantiomer, both isomers exhibit activity on HBsAg as demonstrated herein.
[0134] The term "an optical isomer" or "a stereoisomer" refers to any of the various stereoisomeric configurations which may exist for a given compound of the present invention and includes geometric isomers. It is understood that a substituent may be attached at a chiral center of a carbon atom. The term "chiral" refers to molecules which have the property of non-superimposability on their mirror image partner, while the term "achiral" refers to molecules which are superimposable on their mirror image partner. Therefore, the invention includes enantiomers, diastereomers or racemates of the compound. "Enantiomers" are a pair of stereoisomers that are non-superimposable mirror images of each other. A 1:1 mixture of a pair of enantiomers is a "racemic" mixture. The term is used to designate a racemic mixture where appropriate. "Diastereoisomers" are stereoisomers that have at least two asymmetric atoms, but which are not mirror-images of each other. The absolute stereochemistry is specified according to the Cahn-lngold- Prelog R-S system. When a compound is a pure enantiomer the stereochemistry at each chiral carbon may be specified by either R or S. Resolved compounds whose absolute configuration is unknown can be designated (+) or (-) depending on the direction (dextro- or levorotatory) which they rotate plane polarized light at the wavelength of the sodium D line. Certain compounds described herein contain one or more asymmetric centers or axes and may thus give rise to enantiomers, diastereomers, and other stereoisomeric forms that may be defined, in terms of absolute stereochemistry, as (R)- or (S)-.
[0135] Depending on the choice of the starting materials and procedures, the compounds can be present in the form of one of the possible isomers or as mixtures thereof, for example as pure optical isomers, or as isomer mixtures, such as racemates and diastereoisomer mixtures, depending on the number of asymmetric carbon atoms. The present invention is meant to include all such possible stereoisomers, including racemic mixtures, diasteriomeric mixtures and optically pure forms. Optically active (R)- and (S)-isomers may be prepared using chiral synthons or chiral reagents, or resolved using conventional techniques. If the compound contains a double bond, the substituent may be E or Z configuration. If the compound contains a disubstituted cycloalkyl, the cycloalkyl substituent may have a cis- or trans-configuration. All tautomeric forms are also intended to be included.
[0136] Any resulting mixtures of isomers can be separated on the basis of the physicochemical differences of the constituents, into the pure or substantially pure geometric or optical isomers or diastereomers, for example, by chromatography and/or fractional crystallization.
[0137] Any resulting racemates of final products or intermediates can be resolved into the optical antipodes by known methods, e.g., by separation of the diastereomeric salts thereof, obtained with an optically active acid or base, and liberating the optically active acidic or basic compound. In particular, a basic moiety may thus be employed to resolve the compounds of the present invention into their optical antipodes, e.g., by fractional crystallization of a salt formed with an optically active acid, e.g., tartaric acid, dibenzoyl tartaric acid, diacetyl tartaric acid, di-O,O'-p-toluoyl tartaric acid, mandelic acid, malic acid or camphor-10-sulfonic acid. Racemic products can also be resolved by chiral chromatography, e.g., high pressure liquid chromatography (HPLC) using a chiral adsorbent.
[0138] Furthermore, the compounds of the present invention, including their salts, can also be obtained in the form of their hydrates, or include other solvents used for their crystallization. The compounds of the present invention may inherently or by design form solvates with pharmaceutically acceptable solvents (including water); therefore, it is intended that the invention embrace both solvated and unsolvated forms. The term "solvate" refers to a molecular complex of a compound of the present invention (including pharmaceutically acceptable salts thereof) with one or more solvent molecules. Such solvent molecules are those commonly used in the pharmaceutical art, which are known to be innocuous to the recipient, e.g., water, ethanol, and the like. The term "hydrate" refers to the complex where the solvent molecule is water.
[0139] The compounds of the present invention, including salts, hydrates and solvates thereof, may inherently or by design form polymorphs.
[0140] As used herein, the terms "salt" or "salts" refers to an acid addition or base addition salt of a compound of the present invention. "Salts" include in particular "pharmaceutically acceptable salts". The term "pharmaceutically acceptable salts" refers to salts that retain the biological effectiveness and properties of the compounds of this invention and, which typically are not biologically or otherwise undesirable. In many cases, the compounds of the present invention are capable of forming acid and/or base salts by virtue of the presence of amino and/or carboxyl groups or groups similar thereto.
[0141] Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids, e.g., acetate, aspartate, benzoate, besylate, bromide/hydrobromide, bicarbonate/carbonate, bisulfate/sulfate, camphorsulfonate, chloride/hydrochloride, chlortheophyllonate, citrate, ethandisulfonate, fumarate, gluceptate, gluconate, glucuronate, hippurate, hydroiodide/iodide, isethionate, lactate, lactobionate, laurylsulfate, malate, maleate, malonate, mandelate, mesylate, methylsulphate, naphthoate, napsylate, nicotinate, nitrate, octadecanoate, oleate, oxalate, palmitate, pamoate, phosphate/hydrogen phosphate/dihydrogen phosphate, polygalacturonate, propionate, stearate, succinate, sulfosalicylate, tartrate, tosylate and trifluoroacetate salts.
[0142] Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like.
[0143] Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, toluenesulfonic acid, sulfosalicylic acid, and the like. Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases.
[0144] Inorganic bases from which salts can be derived include, for example, ammonium salts and metals from columns I to XII of the periodic table. In certain embodiments, the salts are derived from sodium, potassium, ammonium, calcium, magnesium, iron, silver, zinc, and copper; particularly suitable salts include ammonium, potassium, sodium, calcium and magnesium salts.
[0145] Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like. Certain organic amines include isopropylamine, benzathine, cholinate, diethanolamine, diethylamine, lysine, meglumine, piperazine and tromethamine.
[0146] The pharmaceutically acceptable salts of the present invention can be synthesized from a basic or acidic moiety, by conventional chemical methods. Generally, such salts can be prepared by reacting free acid forms of these compounds with a stoichiometric amount of the appropriate base (such as Na, Ca, Mg, or K hydroxide, carbonate, bicarbonate or the like), or by reacting free base forms of these compounds with a stoichiometric amount of the appropriate acid. Such reactions are typically carried out in water or in an organic solvent, or in a mixture of the two. Generally, use of non-aqueous media like ether, ethyl acetate, ethanol, isopropanol, or acetonitrile is desirable, where practicable. Lists of additional suitable salts can be found, e.g., in "Remington's Pharmaceutical Sciences", 20th ed., Mack Publishing Company, Easton, Pa., (1985); and in "Handbook of Pharmaceutical Salts: Properties, Selection, and Use" by Stahl and Wermuth (Wiley-VCH, Weinheim, Germany, 2002).
[0147] Any formula given herein is intended to represent unlabeled forms as well as isotopically labeled forms of the compounds of the present invention having up to three atoms with non-natural isotope distributions, e.g., sites that are enriched in deuterium or .sup.13C or .sup.15N. Isotopically labeled compounds have structures depicted by the formulas given herein except that one or more atoms are replaced by an atom having a selected atomic mass or mass number other than the natural-abundance mass distribution. Examples of isotopes that can be usefully over-incorporated into compounds of the invention include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, fluorine, and chlorine, such as .sup.2H, .sup.3H, .sup.11C, .sup.13C, .sup.14C, .sup.15N, .sup.18F .sup.31P, .sup.32P, .sup.35S, .sup.36Cl, .sup.125I respectively. The invention includes various isotopically labeled compounds of the present invention, for example those into which radioactive isotopes, such as .sup.3H and .sup.14C, or those in which non-radioactive isotopes, such as .sup.2H and .sup.13C are present at levels substantially above normal isotope distribution. Such isotopically labelled compounds are useful in metabolic studies (with .sup.14C, for example), reaction kinetic studies (with, for example .sup.2H or .sup.3H), detection or imaging techniques, such as positron emission tomography (PET) or single-photon emission computed tomography (SPECT) including drug or substrate tissue distribution assays, or in radioactive treatment of patients. In particular, an .sup.18F labeled compound of the present invention may be particularly desirable for PET or SPECT studies. Isotopically-labeled compounds of the present invention can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying Examples and Preparations using an appropriate isotopically-labeled reagent in place of the non-labeled reagent typically employed. Labeled samples may be useful with quite low isotope incorporation, such as where a radiolabel is used to detect trace amounts of the compound.
[0148] Further, site-specific substitution with heavier isotopes, particularly deuterium (i.e., .sup.2H or D), may afford certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements or an improvement in therapeutic index. It is understood that deuterium in this context is regarded as a substituent of a compound of the present invention, and typically a sample of a compound having deuterium as a substituent has at least 50% deuterium incorporation at the labeled position(s). The concentration of such a heavier isotope, specifically deuterium, may be defined by the isotopic enrichment factor. The term "isotopic enrichment factor" as used herein means the ratio between the isotopic abundance and the natural abundance of a specified isotope. If a substituent in a compound of this invention is denoted deuterium, such compound has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium incorporation), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation).
[0149] Pharmaceutically acceptable solvates in accordance with the invention include those wherein the solvent of crystallization may be isotopically substituted, e.g. D.sub.2O, d.sup.6-acetone, d.sup.6-DMSO.
[0150] Compounds of the present invention that contain groups capable of acting as donors and/or acceptors for hydrogen bonds may be capable of forming co-crystals with suitable co-crystal formers. These co-crystals may be prepared from compounds of the present invention by known co-crystal forming procedures. Such procedures include grinding, heating, co-subliming, co-melting, or contacting in solution compounds of the present invention with the co-crystal former under crystallization conditions and isolating co-crystals thereby formed. Suitable co-crystal formers include those described in WO 2004/078163. Hence the invention further provides co-crystals comprising a compound of the present invention.
Methods of Use
[0151] All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g. "such as") provided herein is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention otherwise claimed.
[0152] The compounds of the invention can be administered by known methods, including oral, parenteral, inhalation, and the like. In certain embodiments, the compound of the invention is administered orally, as a pill, lozenge, troche, capsule, solution, or suspension. In other embodiments, a compound of the invention is administered by injection or infusion. Infusion is typically performed intravenously, often over a period of time between about 15 minutes and 4 hours. In other embodiments, a compound of the invention is administered intranasally or by inhalation; inhalation methods are particularly useful for treatment of respiratory infections. Compounds of the present invention exhibit oral bioavailability, so oral administration is sometimes preferred.
[0153] In certain embodiments of the present invention, a compound of the present invention is used in combination with a second antiviral agent, such as those named herein.
[0154] By the term "combination", is meant either a fixed combination in one dosage unit form, as separate dosage forms suitable for use together either simultaneously or sequentially, or as a kit of parts for the combined administration where a compound of the present invention and a combination partner may be administered independently at the same time or separately within time intervals that especially allow that the combination partners show a cooperative, e.g., synergistic, effect, or any combination thereof.
[0155] The second antiviral agent may be administered in combination with the compounds of the present inventions wherein the second antiviral agent is administered prior to, simultaneously, or after the compound or compounds of the present invention. When simultaneous administration of a compound of the invention with a second agent is desired and the route of administration is the same, then a compound of the invention may be formulated with a second agent into the same dosage form. An example of a dosage form containing a compound of the invention and a second agent is a tablet or a capsule.
[0156] In some embodiments, a combination of a compound of the invention and a second antiviral agent may provide synergistic activity. The compound of the invention and second antiviral agent may be administered together, separate but simultaneously, or sequentially.
[0157] An "effective amount" of a compound is that amount necessary or sufficient to treat or prevent a viral infection and/or a disease or condition described herein. In an example, an effective amount of a compound of Formula I is an amount sufficient to treat viral infection in a subject. In another example, an effective amount is an amount sufficient to treat HBV in a subject in need of such treatment. The effective amount can vary depending on such factors as the size and weight of the subject, the type of illness, or the particular compound of the invention. For example, the choice of the compound of the invention can affect what constitutes an "effective amount." One of ordinary skill in the art would be able to study the factors contained herein and make the determination regarding the effective amount of the compounds of the invention without undue experimentation.
[0158] The regimen of administration can affect what constitutes an effective amount. The compound of the invention can be administered to the subject either prior to or after the onset of a viral infection. Further, several divided dosages, as well as staggered dosages, can be administered daily or sequentially, or the dose can be continuously infused, or can be a bolus injection. Further, the dosages of the compound(s) of the invention can be proportionally increased or decreased as indicated by the exigencies of the therapeutic or prophylactic situation.
[0159] Compounds of the invention may be used in the treatment of states, disorders or diseases as described herein, or for the manufacture of pharmaceutical compositions for use in the treatment of these diseases. The invention provides methods of use of compounds of the present invention in the treatment of these diseases or for preparation of pharmaceutical compositions having compounds of the present invention for the treatment of these diseases.
[0160] The language "pharmaceutical composition" includes preparations suitable for administration to mammals, e.g., humans. When the compounds of the present invention are administered as pharmaceuticals to mammals, e.g., humans, they can be given per se or as a pharmaceutical composition containing, for example, 0.1 to 99.5% (more preferably, 0.5 to 90%) of at least one compound of Formula (I) or any subgenus thereof as active ingredient in combination with a pharmaceutically acceptable carrier, or optionally two or more pharmaceutically acceptable carriers.
[0161] The phrase "pharmaceutically acceptable carrier" is art recognized and includes a pharmaceutically acceptable material, composition or vehicle, suitable for administering compounds of the present invention to mammals. The carriers include liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject agent from one organ, or portion of the body, to another organ, or portion of the body. Each carrier must be "acceptable" in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient. Some examples of materials which can serve as pharmaceutically acceptable carriers include: sugars, such as lactose, glucose and sucrose; starches, such as corn starch and potato starch; cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; powdered tragacanth; malt; gelatin; talc; excipients, such as cocoa butter and suppository waxes; oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil and soybean oil; glycols, such as propylene glycol; polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; esters, such as ethyl oleate and ethyl laurate; agar; buffering agents, such as magnesium hydroxide and aluminum hydroxide; alginic acid; pyrogen-free water; isotonic saline; Ringer's solution; ethyl alcohol; phosphate buffer solutions; and other non-toxic compatible substances employed in pharmaceutical formulations. Typically, pharmaceutically acceptable carriers are sterilized and/or substantially pyrogen-free.
[0162] Wetting agents, emulsifiers and lubricants, such as sodium lauryl sulfate and magnesium stearate, as well as coloring agents, release agents, coating agents, sweetening, flavoring and perfuming agents, preservatives and antioxidants can also be present in the compositions.
[0163] Examples of pharmaceutically acceptable antioxidants include: water soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, .alpha.-tocopherol, and the like; and metal chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
[0164] Formulations of the present invention include those suitable for oral, nasal, inhalation, topical, transdermal, buccal, sublingual, rectal, vaginal and/or parenteral administration. The formulations may conveniently be presented in unit dosage form and may be prepared by any methods well known in the art of pharmacy. The amount of active ingredient that can be combined with a carrier material to produce a single dosage form will generally be that amount of the compound that produces a therapeutic effect. Generally, out of one hundred percent, this amount will range from about 1 percent to about ninety-nine percent of active ingredient, preferably from about 5 percent to about 70 percent, most preferably from about 10 percent to about 30 percent.
[0165] Methods of preparing these formulations or compositions include the step of bringing into association a compound of the present invention with the carrier and, optionally, one or more accessory ingredients. In general, the formulations are prepared by uniformly and intimately bringing into association a compound of the present invention with liquid carriers, or finely divided solid carriers, or both, and then, if necessary, shaping the product.
[0166] Formulations of the invention suitable for oral administration may be in the form of capsules, cachets, pills, tablets, lozenges (using a flavored base, for example, usually sucrose and acacia or tragacanth), powders, granules, or as a solution or a suspension in an aqueous or non-aqueous liquid, or as an oil-in-water or water-in-oil liquid emulsion, or as an elixir or syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of a compound of the present invention as an active ingredient. A compound of the present invention may also be administered as a bolus, electuary or paste.
[0167] In solid dosage forms of the invention for oral administration (capsules, tablets, pills, dragees, powders, granules and the like), the active ingredient is mixed with one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; humectants, such as glycerol; disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; solution retarding agents, such as paraffin; absorption accelerators, such as quaternary ammonium compounds; wetting agents, such as, for example, cetyl alcohol and glycerol monostearate; absorbents, such as kaolin and bentonite clay; lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; and coloring agents. In the case of capsules, tablets and pills, the pharmaceutical compositions may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
[0168] A tablet may be made by compression or molding, optionally with one or more accessory ingredients. Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface-active or dispersing agent. Molded tablets may be made by molding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
[0169] The tablets, and other solid dosage forms of the pharmaceutical compositions of the present invention, such as dragees, capsules, pills and granules, may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art. They may also be formulated so as to provide slow or controlled release of the active ingredient therein using, for example, hydroxypropylmethyl cellulose in varying proportions to provide the desired release profile, other polymer matrices, liposomes and/or microspheres. They may be sterilized by, for example, filtration through a bacteria-retaining filter, or by incorporating sterilizing agents in the form of sterile solid compositions that can be dissolved in sterile water, or some other sterile injectable medium immediately before use. These compositions may also optionally contain opacifying agents and may be of a composition that they release the active ingredient(s) only, or preferentially, in a certain portion of the gastrointestinal tract, optionally, in a delayed manner. Examples of embedding compositions that can be used include polymeric substances and waxes. The active ingredient can also be in micro-encapsulated form, if appropriate, with one or more of the above-described excipients.
[0170] Liquid dosage forms for oral administration of the compounds of the invention include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs. In addition to the active ingredient, the liquid dosage forms may contain inert diluent commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof.
[0171] Besides inert diluents, the oral compositions can also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming and preservative agents.
[0172] Suspensions, in addition to the active compounds, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
[0173] Formulations of the pharmaceutical compositions of the invention for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing one or more compounds of the invention with one or more suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active compound.
[0174] Formulations of the present invention which are suitable for vaginal administration also include pessaries, tampons, creams, gels, pastes, foams or spray formulations containing such carriers as are known in the art to be appropriate.
[0175] Dosage forms for the topical or transdermal administration of a compound of this invention include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. The active compound may be mixed under sterile conditions with a pharmaceutically acceptable carrier, and with any preservatives, buffers, or propellants that may be required.
[0176] The ointments, pastes, creams and gels may contain, in addition to an active compound of this invention, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
[0177] Powders and sprays can contain, in addition to a compound of this invention, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances. Sprays can additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
[0178] Transdermal patches have the added advantage of providing controlled delivery of a compound of the present invention to the body. Such dosage forms can be made by dissolving or dispersing the compound in the proper medium. Absorption enhancers can also be used to increase the flux of the compound across the skin. The rate of such flux can be controlled by either providing a rate controlling membrane or dispersing the active compound in a polymer matrix or gel.
[0179] Ophthalmic formulations, eye ointments, powders, solutions and the like, are also contemplated as being within the scope of this invention.
[0180] Pharmaceutical compositions of this invention suitable for parenteral administration may comprise one or more compounds of the invention in combination with one or more pharmaceutically acceptable carriers such as sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
[0181] Examples of suitable aqueous and nonaqueous carriers that may be employed in the pharmaceutical compositions of the invention include water, ethanol, glycol ethers, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
[0182] These compositions may also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents that delay absorption such as aluminum monostearate and gelatin.
[0183] In some cases, in order to prolong the effect of a drug, it is desirable to slow the absorption of the drug from subcutaneous or intramuscular injection. This may be accomplished by the use of a liquid suspension of crystalline or amorphous material having poor water solubility. The rate of absorption of the drug then depends upon its rate of dissolution which, in turn, may depend upon crystal size and crystalline form. Alternatively, delayed absorption of a parenterally-administered drug form is accomplished by dissolving or suspending the drug in an oil vehicle.
[0184] Injectable depot forms are made by forming microencapsule matrices of the subject compounds in biodegradable polymers such as polylactide-polyglycolide. Depending on the ratio of drug to polymer, and the nature of the particular polymer employed, the rate of drug release can be controlled. Examples of other biodegradable polymers include poly(orthoesters) and poly(anhydrides). Depot injectable formulations are also prepared by entrapping the drug in liposomes or microemulsions that are compatible with body tissue.
[0185] The preparations of the present invention may be given orally, parenterally, topically, or rectally. They are of course given by forms suitable for each administration route. For example, they are administered in tablets or capsule form, by injection, inhalation, eye lotion, ointment, suppository, etc., administration by injection, infusion or inhalation; topical by lotion or ointment; and rectal by suppositories.
[0186] The phrases "parenteral administration" and "administered parenterally" as used herein means modes of administration other than enteral and topical administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrasternal injection and infusion. Intravenous infusion is sometimes a preferred method of delivery for compounds of the invention. Infusion may be used to deliver a single daily dose or multiple doses. In some embodiments, a compound of the invention is administered by infusion over an interval between 15 minutes and 4 hours, typically between 0.5 and 3 hours. Such infusion may be used once per day, twice per day or up to three times per day.
[0187] The phrases "systemic administration," "administered systemically," "peripheral administration" and "administered peripherally" as used herein mean the administration of a compound, drug or other material other than directly into the central nervous system, such that it enters the patient's system and, thus, is subject to metabolism and other like processes, for example, subcutaneous administration.
[0188] These compounds may be administered to humans and other animals for therapy by any suitable route of administration, including orally, nasally, as by, for example, a spray, rectally, intravaginally, parenterally, intracisternally and topically, as by powders, ointments or drops, including buccally and sublingually.
[0189] Regardless of the route of administration selected, the compounds of the present invention, which may be used in a suitable hydrated form, and/or the pharmaceutical compositions of the present invention, are formulated into pharmaceutically acceptable dosage forms by conventional methods known to those of skill in the art.
[0190] Actual dosage levels of the active ingredients in the pharmaceutical compositions of this invention may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient.
[0191] The selected dosage level will depend upon a variety of factors including the activity of the particular compound of the present invention employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compound employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
[0192] A physician or veterinarian having ordinary skill in the art can readily determine and prescribe the effective amount of the pharmaceutical composition required. For example, the physician or veterinarian could start doses of the compounds of the invention employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved.
[0193] In general, a suitable daily dose of a compound of the invention will be that amount of the compound that is the lowest dose effective to produce a therapeutic effect. Such an effective dose will generally depend upon the factors described above. Generally, intravenous and subcutaneous doses of the compounds of this invention for a patient, when used for the indicated effects, will range from about 0.0001 to about 100 mg per kilogram of body weight per day, more preferably from about 0.01 to about 50 mg per kg per day, and still more preferably from about 0.1 to about 20 mg per kg per day. An effective amount is that amount which prevents or treats a viral infection, such as HBV.
[0194] Treatment with a compound or composition described herein may be repeated daily for a period sufficient to reduce or substantially eliminate an HBV infection or viral load. For example, treatment may be continued for a week, or two weeks, or 3-4 weeks, or 4-8 weeks, or 8-12 weeks, 2-6 months, or longer, e.g., until viral load or other measure of infection shows a substantial reduction in viral load or viral activity or other signs or symptoms of HBV infection. The skilled treating physician can readily determine a suitable duration of treatment.
[0195] If desired, the effective daily dose of the active compound may be administered as a single dose per day, or as two, three, four, five, six or more sub-doses administered separately at appropriate intervals throughout the day, optionally, in unit dosage forms. Compounds delivered orally or by inhalation, are commonly administered in one to four doses per day. Compounds delivered by injection are typically administered once per day, or once every other day. Compounds delivered by infusion are typically administered in one to three doses per day. When multiple doses are administered within a day, the doses may be administered at intervals of about 4 hours, about 6 hours, about 8 hours or about 12 hours.
[0196] While it is possible for a compound of the present invention to be administered alone, it is preferable to administer the compound as a pharmaceutical composition such as those described herein. Thus methods of using the compounds of the invention include administering the compound as a pharmaceutical composition, wherein at least one compound of the invention is admixed with a pharmaceutically acceptable carrier prior to administration.
Use of Compounds of the Invention in Combination with Immunomodulators
[0197] The compounds and compositions described herein can be used or administered in combination with one or more therapeutic agents that act as immunomodulators, e.g., an activator of a costimulatory molecule, or an inhibitor of an immune-inhibitory molecule, or a vaccine. The Programmed Death 1 (PD-1) protein is an inhibitory member of the extended CD28/CTLA4 family of T cell regulators (Okazaki et al. (2002) Curr Opin Immunol 14: 391779-82; Bennett et al. (2003) J. Immunol. 170:711-8). PD-1 is expressed on activated B cells, T cells, and monocytes. PD-1 is an immune-inhibitory protein that negatively regulates TCR signals (Ishida, Y. et al. (1992) EMBO J. 11:3887-3895; Blank, C. et al. (Epub 2006 Dec. 29) Immunol. Immunother. 56(5):739-745), and is up-regulated in chronic infections. The interaction between PD-1 and PD-L1 can act as an immune checkpoint, which can lead to, e.g., a decrease in infiltrating lymphocytes, a decrease in T-cell receptor mediated proliferation, and/or immune evasion by cancerous or infected cells (Dong et al. (2003) J. Mol. Med. 81:281-7; Blank et al. (2005) Cancer Immunol. Immunother. 54:307-314; Konishi et al. (2004) Clin. Cancer Res. 10:5094-100). Immune suppression can be reversed by inhibiting the local interaction of PD-1 with PD-L1 or PD-L2; the effect is additive when the interaction of PD-1 with PD-L2 is blocked as well (Iwai et al. (2002) Proc. Nat'l. Acad. Sci. USA 99:12293-7; Brown et al. (2003) J. Immunol. 170:1257-66). Immunomodulation can be achieved by binding to either the immune-inhibitory protein (e.g., PD-1) or to binding proteins that modulate the inhibitory protein (e.g., PD-L1, PD-L2).
[0198] In one embodiment, the combination therapies of the invention include an immunomodulator that is an inhibitor or antagonist of an inhibitory molecule of an immune checkpoint molecule. In another embodiment the immunomodulator binds to a protein that naturally inhibits the immuno-inhibitory checkpoint molecule. When used in combination with antiviral compounds, these immunomodulators can enhance the antiviral response, and thus enhance efficacy relative to treatment with the antiviral compound alone.
[0199] The term "immune checkpoints" refers to a group of molecules on the cell surface of CD4 and CD8 T cells. These molecules can effectively serve as "brakes" to down-modulate or inhibit an adaptive immune response. Immune checkpoint molecules include, but are not limited to, Programmed Death 1 (PD-1), Cytotoxic T-Lymphocyte Antigen 4 (CTLA-4), B7H1, B7H4, OX-40, CD137, CD40, and LAG3, which directly inhibit immune cells. Immunotherapeutic agents which can act as immune checkpoint inhibitors useful in the methods of the present invention, include, but are not limited to, inhibitors of PD-L1, PD-L2, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, 2B4 and/or TGFR beta. Inhibition of an inhibitory molecule can be performed by inhibition at the DNA, RNA or protein level. In some embodiments, an inhibitory nucleic acid (e.g., a dsRNA, siRNA or shRNA), can be used to inhibit expression of an inhibitory molecule. In other embodiments, the inhibitor of an inhibitory signal is a polypeptide, e.g., a soluble ligand, or an antibody or antigen-binding fragment thereof, that binds to the inhibitory molecule.
[0200] By "in combination with," it is not intended to imply that the therapy or the therapeutic agents must be administered at the same time and/or formulated for delivery together, although these methods of delivery are within the scope described herein. The immunomodulator can be administered concurrently with, prior to, or subsequent to, one or more compounds of the invention, and optionally one or more additional therapies or therapeutic agents. The therapeutic agents in the combination can be administered in any order. In general, each agent will be administered at a dose and/or on a time schedule determined for that agent. It will further be appreciated that the therapeutic agents utilized in this combination may be administered together in a single composition or administered separately in different compositions. In general, it is expected that each of the therapeutic agents utilized in combination be utilized at levels that do not exceed the levels at which they are utilized individually. In some embodiments, the levels utilized in combination will be lower than those utilized individually.
[0201] In certain embodiments, the antiviral compounds described herein are administered in combination with one or more immunomodulators that are inhibitors of PD-1, PD-L1 and/or PD-L2. Each such inhibitor may be an antibody, an antigen binding fragment thereof, an immunoadhesin, a fusion protein, or an oligopeptide. Examples of such immunomodulators are known in the art.
[0202] In some embodiments, the immunomodulator is an anti-PD-1 antibody chosen from MDX-1106, Merck 3475 or CT-011.
[0203] In some embodiments, the immunomodulator is an immunoadhesin (e.g., an immunoadhesin comprising an extracellular or PD-1 binding portion of PD-L1 or PD-L2 fused to a constant region (e.g., an Fc region of an immunoglobulin sequence).
[0204] In some embodiments, the immunomodulator is a PD-1 inhibitor such as AMP-224.
[0205] In some embodiments, the immunomodulator is a PD-L1 inhibitor such as anti-PD-L1 antibody.
[0206] In some embodiments, the immunomodulator is an anti-PD-L1 binding antagonist chosen from YW243.55.S70, MPDL3280A, MEDI-4736, MSB-0010718C, or MDX-1105. MDX-1105, also known as BMS-936559, is an anti-PD-L1 antibody described in WO2007/005874. Antibody YW243.55.S70 is an anti-PD-L1 described in WO 2010/077634.
[0207] In some embodiments, the immunomodulator is nivolumab (CAS Registry Number: 946414-94-4). Alternative names for nivolumab include MDX-1106, MDX-1106-04, ONO-4538, or BMS-936558. Nivolumab is a fully human IgG4 monoclonal antibody which specifically blocks PD-1. Nivolumab (clone 5C4) and other human monoclonal antibodies that specifically bind to PD-1 are disclosed in U.S. Pat. No. 8,008,449, EP2161336 and WO2006/121168.
[0208] In some embodiments, the immunomodulator is an anti-PD-1 antibody Pembrolizumab. Pembrolizumab (also referred to as Lambrolizumab, MK-3475, MK03475, SCH-900475 or KEYTRUDA.RTM.; Merck) is a humanized IgG4 monoclonal antibody that binds to PD-1. Pembrolizumab and other humanized anti-PD-1 antibodies are disclosed in Hamid, O. et al. (2013) New England Journal of Medicine 369 (2): 134-44, U.S. Pat. No. 8,354,509, WO2009/114335, and WO2013/079174.
[0209] In some embodiments, the immunomodulator is Pidilizumab (CT-011; Cure Tech), a humanized IgG1k monoclonal antibody that binds to PD1. Pidilizumab and other humanized anti-PD-1 monoclonal antibodies are disclosed in WO2009/101611.
[0210] Other anti-PD1 antibodies useful as immunomodulators for use in the methods disclosed herein include AMP 514 (Amplimmune), and anti-PD1 antibodies disclosed in U.S. Pat. No. 8,609,089, US 2010028330, and/or US 20120114649. In some embodiments, the anti-PD-L1 antibody is MSB0010718C. MSB0010718C (also referred to as A09-246-2; Merck Serono) is a monoclonal antibody that binds to PD-L1.
[0211] In some embodiments, the immunomodulator is MDPL3280A (Genentech/Roche), a human Fc optimized IgG1 monoclonal antibody that binds to PD-L1. MDPL3280A and other human monoclonal antibodies to PD-L1 are disclosed in U.S. Pat. No. 7,943,743 and U. S Publication No.: 20120039906. Other anti-PD-L1 binding agents useful as immunomodulators for methods of the invention include YW243.55.S70 (see WO2010/077634), MDX-1105 (also referred to as BMS-936559), and anti-PD-L1 binding agents disclosed in WO2007/005874.
[0212] In some embodiments, the immunomodulator is AMP-224 (B7-DCIg; Amplimmune; e.g., disclosed in WO2010/027827 and WO2011/066342), is a PD-L2 Fc fusion soluble receptor that blocks the interaction between PD1 and B7-H1.
[0213] In some embodiments, the immunomodulator is an anti-LAG-3 antibody such as BMS-986016. BMS-986016 (also referred to as BMS986016) is a monoclonal antibody that binds to LAG-3. BMS-986016 and other humanized anti-LAG-3 antibodies are disclosed in US 2011/0150892, WO2010/019570, and WO2014/008218
[0214] In certain embodiments, the combination therapies disclosed herein include a modulator of a costimulatory molecule or an inhibitory molecule, e.g., a co-inhibitory ligand or receptor.
[0215] In one embodiment, the costimulatory modulator, e.g., agonist, of a costimulatory molecule is chosen from an agonist (e.g., an agonistic antibody or antigen-binding fragment thereof, or soluble fusion) of OX40, CD2, CD27, CDS, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1BB (CD137), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, B7-H3 or CD83 ligand.
[0216] In another embodiment, the combination therapies disclosed herein include an immunomodulator that is a costimulatory molecule, e.g., an agonist associated with a positive signal that includes a costimulatory domain of CD28, CD27, ICOS and/or GITR.
[0217] Exemplary GITR agonists include, e.g., GITR fusion proteins and anti-GITR antibodies (e.g., bivalent anti-GITR antibodies), such as, a GITR fusion protein described in U.S. Pat. No. 6,111,090, European Patent No.: 090505B1, U.S. Pat. No. 8,586,023, PCT Publication Nos.: WO 2010/003118 and 2011/090754, or an anti-GITR antibody described, e.g., in U.S. Pat. No. 7,025,962, European Patent No.: 1947183B1, U.S. Pat. Nos. 7,812,135, 8,388,967, 8,591,886, European Patent No.: EP 1866339, PCT Publication No.: WO 2011/028683, PCT Publication No.: WO 2013/039954, PCT Publication No.: WO2005/007190, PCT Publication No.: WO 2007/133822, PCT Publication No.: WO2005/055808, PCT Publication No.: WO 99/40196, PCT Publication No.: WO 2001/03720, PCT Publication No.: WO99/20758, PCT Publication No.: WO2006/083289, PCT Publication No.: WO 2005/115451, U.S. Pat. No. 7,618,632, and PCT Publication No.: WO 2011/051726.
[0218] In one embodiment, the immunomodulator used is a soluble ligand (e.g., a CTLA-4-Ig), or an antibody or antibody fragment that binds to PD-L1, PD-L2 or CTLA4. For example, the anti-PD-1 antibody molecule can be administered in combination with an anti-CTLA-4 antibody, e.g., ipilimumab, for example. Exemplary anti-CTLA4 antibodies include Tremelimumab (IgG2 monoclonal antibody available from Pfizer, formerly known as ticilimumab, CP-675,206); and Ipilimumab (CTLA-4 antibody, also known as MDX-010, CAS No. 477202-00-9).
[0219] In one embodiment, an anti-PD-1 antibody molecule is administered after treatment with a compound of the invention as described herein.
[0220] In another embodiment, an anti-PD-1 or PD-L1 antibody molecule is administered in combination with an anti-LAG-3 antibody or an antigen-binding fragment thereof. In another embodiment, the anti-PD-1 or PD-L1 antibody molecule is administered in combination with an anti-TIM-3 antibody or antigen-binding fragment thereof. In yet other embodiments, the anti-PD-1 or PD-L1 antibody molecule is administered in combination with an anti-LAG-3 antibody and an anti-TIM-3 antibody, or antigen-binding fragments thereof. The combination of antibodies recited herein can be administered separately, e.g., as separate antibodies, or linked, e.g., as a bispecific or trispecific antibody molecule. In one embodiment, a bispecific antibody that includes an anti-PD-1 or PD-L1 antibody molecule and an anti-TIM-3 or anti-LAG-3 antibody, or antigen-binding fragment thereof, is administered. In certain embodiments, the combination of antibodies recited herein is used to treat a cancer, e.g., a cancer as described herein (e.g., a solid tumor). The efficacy of the aforesaid combinations can be tested in animal models known in the art. For example, the animal models to test the synergistic effect of anti-PD-1 and anti-LAG-3 are described, e.g., in Woo et al. (2012) Cancer Res. 72(4):917-27).
[0221] Exemplary immunomodulators that can be used in the combination therapies include, but are not limited to, e.g., afutuzumab (available from Roche.RTM.); pegfilgrastim (Neulasta.RTM.); lenalidomide (CC-5013, Revlimid.RTM.); thalidomide (Thalomid.RTM.), actimid (CC4047); and cytokines, e.g., IL-21 or IRX-2 (mixture of human cytokines including interleukin 1, interleukin 2, and interferon .gamma., CAS 951209-71-5, available from IRX Therapeutics).
[0222] Exemplary doses of such immunomodulators that can be used in combination with the antiviral compounds of the invention include a dose of anti-PD-1 antibody molecule of about 1 to 10 mg/kg, e.g., 3 mg/kg, and a dose of an anti-CTLA-4 antibody, e.g., ipilimumab, of about 3 mg/kg.
[0223] Examples of embodiments of the methods of using the antiviral compounds of the invention in combination with an immunomodulator include these, which may be used along with a compound of Formula I or any subgenus or species thereof that is disclosed herein:
[0224] i. A method to treat a viral infection in a subject, comprising administering to the subject a compound of Formula (I) as described herein, and an immunomodulator.
[0225] ii. The method of embodiment i, wherein the immunomodulator is an activator of a costimulatory molecule or an inhibitor of an immune checkpoint molecule.
[0226] iii. The method of either of embodiments i and ii, wherein the activator of the costimulatory molecule is an agonist of one or more of OX40, CD2, CD27, CDS, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1BB (CD137), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, B7-H3 and CD83 ligand.
[0227] iv. The method of any of embodiments i-iii above, wherein the inhibitor of the immune checkpoint molecule is chosen from PD-1, PD-L1, PD-L2, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, 2B4 and TGFR beta.
[0228] v. The method of any of embodiments i-iii, wherein the inhibitor of the immune checkpoint molecule is chosen from an inhibitor of PD-1, PD-L1, LAG-3, TIM-3 or CTLA4, or any combination thereof.
[0229] vi. The method of any of embodiments i-v, wherein the inhibitor of the immune checkpoint molecule is a soluble ligand or an antibody or antigen-binding fragment thereof, that binds to the immune checkpoint molecule.
[0230] vii. The method of any of embodiments i-vi, wherein the antibody or antigen-binding fragment thereof is from an IgG1 or IgG4 (e.g., human IgG1 or IgG4).
[0231] viii. The method of any of embodiments i-vii, wherein the antibody or antigen-binding fragment thereof is altered, e.g., mutated, to increase or decrease one or more of: Fc receptor binding, antibody glycosylation, the number of cysteine residues, effector cell function, or complement function.
[0232] ix. The method of any of embodiments i-viii, wherein the antibody molecule is a bispecific or multispecific antibody molecule that has a first binding specificity to PD-1 or PD-L1 and a second binding specificity to TIM-3, LAG-3, or PD-L2.
[0233] x. The method of any of embodiments i-ix, wherein the immunomodulator is an anti-PD-1 antibody chosen from Nivolumab, Pembrolizumab or Pidilizumab.
[0234] xi. The method of any of embodiments i-x, wherein the immunomodulator is an anti-PD-L1 antibody chosen from YW243.55.S70, MPDL3280A, MEDI-4736, MSB-0010718C, or MDX-1105.
[0235] xii. The method of any of embodiments i-x, wherein the immunomodulator is an anti-LAG-3 antibody molecule.
[0236] xiii. The method of embodiment xii, wherein the anti-LAG-3 antibody molecule is BMS-986016.
[0237] xiv. The method of any of embodiments i-x, wherein the immunomodulator is an anti-PD-1 antibody molecule administered by injection (e.g., subcutaneously or intravenously) at a dose of about 1 to 30 mg/kg, e.g., about 5 to 25 mg/kg, about 10 to 20 mg/kg, about 1 to 5 mg/kg, or about 3 mg/kg., e.g., once a week to once every 2, 3, or 4 weeks.
[0238] xv. The method of embodiment xiv, wherein the anti-PD-1 antibody molecule is administered at a dose from about 10 to 20 mg/kg every other week.
[0239] xvi. The method of embodiment xv, wherein the anti-PD-1 antibody molecule, e.g., nivolumab, is administered intravenously at a dose from about 1 mg/kg to 3 mg/kg, e.g., about 1 mg/kg, 2 mg/kg or 3 mg/kg, every two weeks.
[0240] xvii. The method of embodiment xv, wherein the anti-PD-1 antibody molecule, e.g., nivolumab, is administered intravenously at a dose of about 2 mg/kg at 3-week intervals.
[0241] The compounds as described herein may be synthesized by the general synthetic routes below, specific examples of which are described in more detail in the Examples.
General Synthetic Procedures
[0242] All starting materials, building blocks, reagents, acids, bases, dehydrating agents, solvents, and catalysts utilized to synthesize the compounds of the invention are either commercially available or can be produced by organic synthesis methods known to one of ordinary skill in the art (Houben-Weyl 4th Ed. 1952, Methods of Organic Synthesis, Thieme, Volume 21). General methods for synthesis of compounds of the invention are illustrated by the Examples below, the general method in Scheme 1, and by methods disclosed in published PCT applications WO2015/113990 and WO2015/173164.
LIST OF ABBREVIATIONS
[0243] Ac acetyl
ACN Acetonitrile
[0244] AcOEt/EtOAc Ethyl acetate AcOH acetic acid aq aqueous Bn benzyl Bu butyl (nBu=n-butyl, tBu=tert-butyl)
CDI Carbonyldiimidazole
[0245] DBU 1,8-Diazabicyclo[5.4.0]-undec-7-ene Boc20 di-tert-butyl dicarbonate
DCE 1,2-Dichloroethane
DCM Dichloromethane
[0246] DIAD Diisopropyl azodicarboxylate
DiBAl-H Diisobutylaluminum Hydride
DIPEA N-Ethyldiisopropylamine
DMA N,N-dimethylacetamide
DMAP Dimethylaminopyridine
DMF N,N'-Dimethylformamide
DMSO Dimethylsulfoxide
[0247] EDCI 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide EI Electrospray ionisation
Et.sub.2O Diethylether
Et.sub.3N Triethylamine
Ether Diethylether
[0248] EtOAc Ethyl acetate
EtOH Ethanol
[0249] FA Formic acid
FC Flash Chromatography
[0250] h hour(s) HCl Hydrochloric acid
HOBt 1-Hydroxybenzotriazole
HPLC High Performance Liquid Chromatography
H.sub.2O Water
[0251] IPA isopropanol L liter(s)
LC-MS Liquid Chromatography Mass Spectrometry
[0252] LiHMDS Lithium bis(trimethylsilyl)amide Me methyl
MeI Iodomethane
MeOH Methanol
[0253] mg milligram min minute(s) mL milliliter
MS Mass Spectrometry
[0254] Pd/C palladium on charcoal PG protecting group Ph phenyl Ph.sub.3P triphenyl phosphine
Prep Preparative
[0255] Rf ratio of fronts RP reverse phase Rt Retention time rt Room temperature
SFC Supercritical Fluid Chromatography
SiO.sub.2 Silica gel
[0256] T3P.RTM. Propylphosphonic acid anhydride TBAF Tetrabutylammonium fluoride TBDMS t-Butyldimethylsilyl
TEA Triethylamine
[0257] TFA Trifluoroacetic acid
THF Tetrahydrofuran
TLC Thin Layer Chromatography
[0258] TsCl toluene sulfonyl chloride
[0259] Within the scope of this text, a readily removable group that is not a constituent of the particular desired end product of the compounds of the present invention is designated a "protecting group," unless the context indicates otherwise. The protection of functional groups by such protecting groups, the protecting groups themselves, and their cleavage reactions are described for example in standard reference works, such as e.g., Science of Synthesis: Houben-Weyl Methods of Molecular Transformation. Georg Thieme Verlag, Stuttgart, Germany. 2005. 41627 pp. (URL: http://www.science-of-synthesis.com (Electronic Version, 48 Volumes)); J. F. W. McOmie, "Protective Groups in Organic Chemistry", Plenum Press, London and New York 1973, in T. W. Greene and P. G. M. Wuts, "Protective Groups in Organic Synthesis", Third edition, Wiley, New York 1999, in "The Peptides"; Volume 3 (editors: E. Gross and J. Meienhofer), Academic Press, London and New York 1981, in "Methoden der organischen Chemie" (Methods of Organic Chemistry), Houben Weyl, 4th edition, Volume 15/I, Georg Thieme Verlag, Stuttgart 1974, in H.-D. Jakubke and H. Jeschkeit, "Aminosauren, Peptide, Proteine" (Amino acids, Peptides, Proteins), Verlag Chemie, Weinheim, Deerfield Beach, and Basel 1982, and in Jochen Lehmann, "Chemie der Kohlenhydrate: Monosaccharide und Derivate" (Chemistry of Carbohydrates: Monosaccharides and Derivatives), Georg Thieme Verlag, Stuttgart 1974. A characteristic of protecting groups is that they can be removed readily (i.e., without the occurrence of undesired secondary reactions) for example by solvolysis, reduction, photolysis or alternatively under physiological conditions (e.g., by enzymatic cleavage).
[0260] Salts of compounds of the present invention having at least one salt-forming group may be prepared in a manner known per se. For example, salts of compounds of the present invention having acid groups may be formed, for example, by treating the compounds with metal compounds, such as alkali metal salts of suitable organic carboxylic acids, e.g., the sodium salt of 2-ethyl hexanoic acid, with organic alkali metal or alkaline earth metal compounds, such as the corresponding hydroxides, carbonates or hydrogen carbonates, such as sodium or potassium hydroxide, carbonate or hydrogen carbonate, with corresponding calcium compounds or with ammonia or a suitable organic amine, stoichiometric amounts or only a small excess of the salt-forming agent preferably being used. Acid addition salts of compounds of the present invention are obtained in customary manner, e.g., by treating the compounds with an acid or a suitable anion exchange reagent. Internal salts of compounds of the present invention containing acid and basic salt-forming groups, e.g., a free carboxy group and a free amino group, may be formed, e.g., by the neutralization of salts, such as acid addition salts, to the isoelectric point, e.g., with weak bases, or by treatment with ion exchangers.
[0261] Salts can be converted in customary manner into the free compounds; metal and ammonium salts can be converted, for example, by treatment with suitable acids, and acid addition salts, for example, by treatment with a suitable basic agent.
[0262] Mixtures of isomers obtainable according to the invention can be separated in a manner known per se into the individual isomers; diastereoisomers can be separated, for example, by partitioning between polyphasic solvent mixtures, recrystallization and/or chromatographic separation, for example over silica gel or by, e.g., medium pressure liquid chromatography over a reversed phase column, and racemates can be separated, for example, by the formation of salts with optically pure salt-forming reagents and separation of the mixture of diastereoisomers so obtainable, for example by means of fractional crystallization, or by chromatography over optically active column materials.
[0263] Intermediates and final products can be worked up and/or purified according to standard methods, e.g., using chromatographic methods, distribution methods, (re-) crystallization, and the like.
EXAMPLES
[0264] The invention is illustrated by the following examples, which should not be construed as limiting. The assays used to demonstrate the efficacy of compounds of Formula (I) in these assays are generally regarded as predictive of efficacy in subjects.
General Conditions:
[0265] Mass spectra were run on LC-MS systems using electrospray ionization. These were WATERS Acquity Single Quard Detector. [M+H].sup.+ refers to mono-isotopic molecular weights.
[0266] NMR spectra were run on open access Varian 400 or Varian 500 NMR spectrometers. Spectra were measured at 298K and were referenced using the solvent peak. Chemical shifts for .sup.1H NMR are reported in parts per million (ppm).
[0267] Mass spectra were run on LC-MS systems with one of the following conditions:
[0268] Waters Acquity UPLC-H class system equipped with SQD detector.
[0269] Column: ACQUITY UPLC HSS C.sub.18 (50*2.1) mm, 1.8 u.
[0270] Column temperature: Ambient.
[0271] Mobile Phase: A) 0.1% FA+5 mM Ammonium Acetate in Water. [0272] B) 0.1% FA in Acetonitrile.
[0273] Gradient: 5-5% solvent B in 0.40 min, 5-35% solvent B in 0.80 min, 35-55% solvent B in 1.2 min,
[0274] 55-100% solvent B in 2.5 min.
[0275] Flow rate: 0.55 mL/min.
[0276] Compounds were detected by a Waters Photodiode Array Detector.
[0277] Waters LCMS system equipped with ZQ 2000 detector.
[0278] Column: X-BRIDGE C.sub.18 (50*4.6) mm, 3.5 u.
[0279] Column temperature: Ambient.
[0280] Mobile Phase: A) 0.1% NH.sub.3 in Water. [0281] B) 0.1% NH.sub.3 in Acetonitrile.
[0282] Gradient: 5-95% solvent B in 5.00 min.
[0283] Flow rate: 1.0 mL/min.
[0284] Compounds were detected by a Waters Photodiode Array Detector.
[0285] Waters ACQUITY UPLC system and equipped with a ZQ 2000 MS system.
[0286] Column: Kinetex by Phenomenex, 2.6 um, 2.1.times.50 mm
[0287] Column temperature: 50.degree. C.
[0288] Gradient: 2-88% (or 00-45%, or 65-95%) solvent B over a 1.29 min period
[0289] Flow rate: 1.2 mL/min.
[0290] Compounds were detected by a Waters Photodiode Array Detector.
[0291] Chiral separations were done with the following columns:
[0292] AD: ChiralPak AD-H, SFC 21.times.250 mm
[0293] OD: ChiralPak OD-H, SFC 21.times.250 mm
Example 1.1
6-(tert-butyl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1- ,2-b]indazole-3-carboxylic acid [1.1-I] and [1.1-II]
##STR00022##
[0294] Step 1: (7-methoxy-2H-indazol-3-yl)methanol [1.1a]
##STR00023##
[0296] To methyl 7-methoxy-2H-indazole-3-carboxylate (950 mg, 4.61 mmol) was added THF (Volume: 25 mL), cooled to 0.degree. C. then LAH (2M in THF; 3.46 mL, 6.91 mmol) was added. The reaction was allowed to warm to room temperature and stirred for 2 hours or until done by LCMS. The reaction was cooled in ice bath, then quenched carefully by adding excess water dropwise (0.8 ml) and as salts formed magnesium sulfate and then sodium sulfate was added. The reaction was removed from ice bath and stirred for 1 hour, filtered through celite, concentrated to residue to give the desired 1.1a assume in quantitative yield, used as is LC-MS (m/z): 179.1 [M+H]+, 0.48 min.
Step 2: 3-(((tert-butyldimethyl silyl)oxy)methyl)-7-methoxy-2H-indazole [1.1 b]
##STR00024##
[0298] To 1.1a (810 mg, 4.55 mmol) was added DCM (Volume: 25 mL) and imidazole (990 mg, 14.55 mmol) and the mixture was stirred at room temperature for 5 minutes. Then TBDMSCl (2055 mg, 13.64 mmol) was added. The reaction was stirred at room temperature for 2 hours or until done by LCMS. To the reaction was added 10 ml of methanol, stirred for 2-3 minutes and concentrated most of solvent off. Then was added 250 ml of ethyl acetate, and the organic layer was washed with saturated sodium bicarbonate, water, saturated salt solution, dried with anhydrous sodium sulfate, filtered and concentrated to residue to give desired product 1.1b, assumed to be in quantitative yield, which was used as is. LC-MS (m/z): 293.3 [M+H]+, 1.15 min.
Step 3: 1-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2- -yl)-3,3-dimethylbutan-2-one [1.1c]
##STR00025##
[0300] To 1.1b (1300 mg, 4.45 mmol) was added DMF (Volume: 20 mL), and Lithium Carbonate (1314 mg, 17.78 mmol) and the mixture was stirred for 10 minutes at 75-80.degree. C. Then 1-bromo-3,3-dimethylbutan-2-one (1990 mg, 11.11 mmol) was added with the reaction at 75-80.degree. C., and the mixture was stirred at 75-80.degree. C. for 15 hours or until done by LCMS. To the reaction was added 150 ml of ethyl acetate, which was washed with saturated sodium bicarbonate, water, saturated salt solution, dried over sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel column chromatography, using 0 to 30% EtOAc/heptanes. The desired regio-isomer eluted second and was concentrated to constant mass to give 520 mg of the desired product 1.1c in 30% yield. LC-MS (m/z): 391.3 [M+H]+, 1.31 min.
Step 4: (2-(2-amino-3,3-dimethylbutyl)-7-methoxy-2H-indazol-3-yl)methanol [1.1d]
##STR00026##
[0302] To 1.1c (520 mg, 1.331 mmol) was added MeOH (Volume: 8 mL), ammonium acetate (1539 mg, 19.97 mmol) and sodium cyanoborohydride (251 mg, 3.99 mmol). The reaction was then stirred at 55.degree. C. for 40 hours or until done by LCMS. To the crude reaction was added 350 ml of DCM and 25 ml of methanol, extracted with 1:1 solution of (6 M NaOH, saturated salt solution). The aqueous layer was back extracted with DCM. The organics were combined washed with saturated salt solution, dried with sodium sulfate, filtered through 1 cm.times.2 cm celite filter plug, washed with DCM with 10% methanol, concentrated to residue to give the desired product 1.1d assume in quantitative yield, used as is. LC-MS (m/z): 278.3 [M+H]+, 0.51 min.
Step 5: 3-(tert-butyl)-7-methoxy-3,4-dihydropyrazino[1,2-b]indazole [1.1e]
##STR00027##
[0304] To 1.1d (360 mg, 1.298 mmol) was added DCM (Volume: 12 mL), and manganese dioxide (1128 mg, 12.98 mmol). The reaction was then stirred room temperature for 2 hours. Then additional manganese dioxide (564 mg, 6.49 mmol) was added and stirred overnight for a total of 20 hours or until done by LCMS. Additional manganese dioxide can be added if needed. To the crude was added 30 ml of DCM stirred for 30 minutes then filtered through a 1 cm.times.2 cm celite filter plug, flushed with DCM and concentrated to residue. The residue was dissolved in 5 ml of DCM and excess TFA (0.300 mL, 3.89 mmol) was added stirred for 5-10 minutes at room temperature then concentrated to residue give the desired product 1.1e assume in quantitative yield, used as is. LC-MS (m/z): 258.3 [M+H]+, 0.59 min.
Step 6: ethyl 6-(tert-butyl)-10-methoxy-2-oxo-2,6,7,13c-tetrahydro-1H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylate [1.1f]
##STR00028##
[0306] To 1.1e (250 mg, 0.972 mmol) was added Ethanol (Volume: 8 mL), and (Z)-ethyl 2-(ethoxymethylene)-3-oxobutanoate (543 mg, 2.91 mmol). The reaction was then stirred at 85-90.degree. C. for 16 hours or until done by LCMS. The reaction was concentrated to residue to give the desired product 1.1f, assumed to be in quantitative yield, which was used as is in the next step. LC-MS (m/z): 398.3 [M+H]+, 0.77 min.
Step 7: ethyl 6-(tert-butyl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [1.1g-I] and [1.1g-II]
##STR00029##
[0308] To 1.1f (384 mg, 0.966 mmol) was added DME (Volume: 7 mL), and then chloranil (238 mg, 0.966 mmol). The reaction was stirred at 90-95.degree. C. for 90 minutes or until done by LCMS. The crude reaction was concentrated to residue and purified by silica gel chromatography, using 0-100% EtOAc (with 25% ethanol)/heptane. The desired fractions were concentrated to constant mass to give 140 mg of the desired racemic product 1.1g in 37% yield. LC-MS (m/z): 396.4 [M+H]+, 0.77 min.
[0309] The above racemic material 128 mg was separated by chiral chromatography using (OD column, SFC=100 ml/min, CO.sub.2/MeOH=75/25, 276 bar) to give products 1.1g-I (peak 1, tR 4.40 min.) at 58 mg and product 1.1g-II (peak 2, tR 6.69 min.) at 55 mg.
Step 8: 6-(tert-butyl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]py- razino[1,2-b]indazole-3-carboxylic acid [1.1-I] and [1.1-II]
##STR00030##
[0311] To 1.1g-I (58 mg, 0.147 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then NaOH 3M (0.147 ml, 0.440 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated off, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 10.0 mg of the desired product 1.1-I as TFA salt in 14% yield. LC-MS (m/z): 368.3 [M+H]+, 0.80 min. .sup.1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.20-7.32 (m, 2H), 6.84 (d, J=7.6 Hz, 1H), 5.12 (br s, 2H), 4.97 (br s, 1H), 3.96 (s, 3H), 0.70 (s, 9H)
[0312] To 1.1g-II (55 mg, 0.139 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.139 ml, 0.417 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated off, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 23.8 mg of the desired product 1.1-II as TFA salt in 35% yield. LC-MS (m/z): 368.3 [M+H]+, 0.80 min. 1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 7.65 (d, J=8.3 Hz, 1H), 7.21-7.33 (m, 2H), 6.84 (d, J=7.6 Hz, 1H), 5.12 (br s, 2H), 4.96 (br s, 1H), 3.96 (s, 3H), 0.70 (s, 9H)
Example 1.2
6-(tert-butyl)-11-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1- ,2-b]indazole-3-carboxylic acid [1.2-I] and [1.2-II]
##STR00031##
[0313] Step 1 to 7: ethyl 6-(tert-butyl)-11-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [1.2g-I] and [1.2g-II]
##STR00032##
[0315] Compound 1.2g was synthesized from the starting material; methyl 6-methoxy-2H-indazole-3-carboxylate by the process of Example 1.1 following steps 1 through 7 resulting in the desired product 1.2g as a racemate. LC-MS (m/z): 396.3 [M+H]+, 0.79 min.
[0316] The above racemic material (65 mg) was separated by chiral chromatography using AD column, (SFC=100 ml/min, CO.sub.2/EtOH=70/30, 266 bar) to give products 1.2g-I (peak 1, tR 2.11 min.) at 32 mg and product 1.2g-II (peak 2, tR 5.42 min.) at 30 mg.
Step 8: 6-(tert-butyl)-11-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]py- razino[1,2-b]indazole-3-carboxylic acid [1.2-I] and [1.2-II]
##STR00033##
[0318] To 1.2g-I (32 mg, 0.081 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.081 ml, 0.243 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated off, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 8.8 mg of the desired product 1.2-I as TFA salt in 22% yield. LC-MS (m/z): 368.3 [M+H]+, 0.81 min. 1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 8.06 (d, J=9.2 Hz, 1H), 7.32 (s, 1H), 7.16 (s, 1H), 6.95-7.03 (m, 1H), 5.01-5.15 (m, 2H), 4.93 (d, J=4.5 Hz, 1H), 3.85 (s, 3H), 0.70 (s, 9H)
[0319] To 1.2g-II (30 mg, 0.076 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.076 ml, 0.228 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 7.4 mg of the desired product 1.2-II as TFA salt in 20% yield. LC-MS (m/z): 368.3 [M+H]+, 0.81 min. .sup.1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 8.06 (d, J=9.2 Hz, 1H), 7.32 (s, 1H), 7.16 (s, 1H), 6.99 (br d, J=9.2 Hz, 1H), 5.00-5.18 (m, 2H), 4.93 (d, J=4.5 Hz, 1H), 3.85 (s, 3H), 0.70 (s, 9H)
Example 1.3
6-(tert-butyl)-12-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1- ,2-b]indazole-3-carboxylic acid [1.3-] and [1.3-II]
##STR00034##
[0320] Step 1 to 7: ethyl 6-(tert-butyl)-12-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [1.3g-I] and [1.3g-II]
##STR00035##
[0322] Compound 1.3g was synthesized from the starting material; methyl 5-methoxy-2H-indazole-3-carboxylate by the process of Example 1.1 following steps 1 through 7 resulting in the desired product 1.3g as a racemate. LC-MS (m/z): 396.3 [M+H]+, 0.81 min.
[0323] The above racemic material (148 mg) was separated by chiral chromatography using AD column, (SFC=100 ml/min, CO.sub.2/EtOH=85/15, 236 bar) to give products 1.3g-I (peak 1, tR 10.41 min.) at 56 mg and product 1.3g-II (peak 2, tR 12.55 min.) at 68 mg.
Step 8: 6-(tert-butyl)-12-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]py- razino[1,2-b]indazole-3-carboxylic acid [1.3-I] and [1.3-II]
##STR00036##
[0325] To 1.3g-I (55 mg, 0.139 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.139 ml, 0.417 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 18.7 mg of the desired product 1.3-I as TFA salt in 27% yield. LC-MS (m/z): 368.3 [M+H]+, 0.83 min. .sup.1H NMR (DMSO-d6) .delta.: 8.94 (s, 1H), 7.75 (d, J=9.2 Hz, 1H), 7.42 (s, 1H), 7.36 (s, 1H), 7.10 (dd, J=9.5, 1.9 Hz, 1H), 5.02-5.17 (m, 2H), 4.94 (d, J=4.5 Hz, 1H), 3.93 (s, 3H), 0.70 (s, 9H). To 1.3g-II (67 mg, 0.169 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.169 ml, 0.508 mmol). (30 mg, 0.076 mmol) was added THF (Volume: 1.2 ml, Ratio: 12.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.8 ml, Ratio: 8.00) and then 3M NaOH (0.076 ml, 0.228 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated, the residue was dissolved in 4 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 29.8 mg of the desired product 1.3-II as TFA salt in 36% yield. LC-MS (m/z): 368.3 [M+H]+, 0.82 min. .sup.1H NMR (DMSO-d6) .delta.: 8.94 (s, 1H), 7.75 (d, J=9.2 Hz, 1H), 7.42 (s, 1H), 7.36 (s, 1H), 7.10 (dd, J=9.2, 1.9 Hz, 1H), 5.02-5.18 (m, 2H), 4.94 (d, J=4.5 Hz, 1H), 3.93 (s, 3H), 0.70 (s, 9H)
Example 1.4
6-(tert-butyl)-13-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1- ,2-b]indazole-3-carboxylic acid [1.4-I] and [1.4-II]
##STR00037##
[0326] Step 1 to 7: ethyl 6-(tert-butyl)-13-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [1.4g-I] and [1.4g-II]
##STR00038##
[0328] Compound 1.4g was synthesized from the starting material; methyl 4-methoxy-2H-indazole-3-carboxylate by the process of Example 1.1 following steps 1 through 7 resulting in the desired product 1.4g as a racemate. LC-MS (m/z): 396.3 [M+H]+, 0.81 min. The above racemic material (60 mg) was purified by chiral chromatography using OD column, (SFC=100 ml/min, CO.sub.2/MeOH=88/12, 246 bar) to give enriched racemate 1.4g (peak 1, tR 8.74 min., 25% and peak 2, tR 11.37 min., 75%) at 30 mg.
Step 8: 6-(tert-butyl)-13-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]py- razino[1,2-b]indazole-3-carboxylic acid [1.4-I] and [1.4-II]
##STR00039##
[0330] To the above enriched racemate 1.4g (30 mg, 0.076 mmol) was added THF (Volume: 0.5 ml, Ratio: 5.00), MeOH (Volume: 0.1 ml, Ratio: 1.000), Water (Volume: 0.3 ml, Ratio: 3.00) and then 3M NaOH (0.076 ml, 0.228 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated, the residue was dissolved in 2 ml of DMSO with 5% water, purified by reverse phase prep LC, and lyophilized to give 18 mg of enriched racemic product 1.4 as TFA salt in 49% yield. LC-MS (m/z): 368.3 [M+H]+, 0.77 min.
[0331] The above enriched material (18 mg) was further purified by chiral chromatography using (AS column, SFC=100 ml/min, CO.sub.2/EtOH=80/20, 226 bar) to give products 1.4-I (peak 1, tR 4.76 min.) at 2.6 mg in 9% yield and product 1.4-II (peak 2, tR 9.41 min.) at 7.7 mg in 27% yield both as free base.
[0332] 1.4-I LC-MS (m/z): 368.3 [M+H]+, 0.82 min. .sup.1H NMR (DMSO-d6) .delta.: 8.90 (br s, 1H), 7.89 (br s, 1H), 7.30-7.43 (m, 2H), 6.77 (br d, J=5.7 Hz, .sup.1H), 4.99-5.18 (m, 2H), 4.93 (br s, 1H), 3.99 (s, 3H), 0.71 (s, 9H)
[0333] 1.4-II LC-MS (m/z): 368.3 [M+H]+, 0.81 min. .sup.1H NMR (DMSO-d6) .delta.: 8.91 (br s, 1H), 7.90 (br s, 1H), 7.29-7.42 (m, 2H), 6.77 (br d, J=6.1 Hz, .sup.1H), 5.02-5.17 (m, 2H), 4.93 (br s, 1H), 3.99 (s, 3H), 0.71 (s, 9H)
Example 1.5
6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazo- le-3-carboxylic acid [1.5-I] and [1.5-II]
##STR00040##
[0334] Step 1 to 7: ethyl 6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-3-carboxylate [1.5g-I] and [1.5g-II]
##STR00041##
[0336] Compound 1.5g was synthesized from the starting material; methyl 2H-indazole-3-carboxylate by the process of Example 1.1 following steps 1 through 7 resulting in the desired product, 1.5g, as a racemate. LC-MS (m/z): 366.3 [M+H]+, 0.68 min. The above racemic material (60 mg) was purified by chiral chromatography using AD column, (SFC=100 ml/min, CO.sub.2/EtOH=70/30, 256 bar) to give products 1.5g-I (peak 1, tR 2.30 min.) at 13 mg and product 1.5g-II (peak 2, tR 3.97 min.) at 13 mg.
Step 8: 6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-- b]indazole-3-carboxylic acid [1.5-I] and [1.5-II]
##STR00042##
[0338] To 1.5g-I (12 mg, 0.033 mmol) was added THF (Volume: 0.2 ml, Ratio: 1.000), MeOH (Volume: 0.2 ml, Ratio: 1.000) and then 3M NaOH (0.033 ml, 0.099 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated, the residue was dissolved in 1.2 ml of DMSO, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 6.8 mg of the desired product 1.5-I as TFA salt in 45% yield. LC-MS (m/z): 338.3 [M+H]+, 0.68 min. .sup.1H NMR (DMSO-d6) .delta.: 8.99 (s, 1H), 8.20 (d, J=8.5 Hz, 1H), 7.85 (d, J=8.5 Hz, 1H), 7.43-7.52 (m, 1H), 7.33-7.41 (m, 2H), 5.10-5.25 (m, 2H), 4.99 (d, J=4.7 Hz, 1H), 0.71 (s, 9H). To 1.5g-II (12 mg, 0.033 mmol) was added THF (Volume: 0.2 ml, Ratio: 1.000), MeOH (Volume: 0.2 ml, Ratio: 1.000) and then 3M NaOH (0.033 ml, 0.099 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The solvent was concentrated off, the residue was dissolved in 1.2 ml of DMSO, purified by reverse phase prep LC, and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 6.8 mg of the desired product 1.5-II as TFA salt in 45% yield. LC-MS (m/z): 338.3 [M+H]+, 0.68 min. .sup.1H NMR (DMSO-d6) .delta.: 8.99 (s, 1H), 8.20 (d, J=8.5 Hz, 1H), 7.85 (d, J=8.5 Hz, 1H), 7.43-7.50 (m, 1H), 7.31-7.41 (m, 2H), 5.10-5.21 (m, 2H), 4.99 (d, J=4.4 Hz, 1H), 0.71 (s, 9H)
Example 2.1
6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylic acid [2.1-I] and [2.1-II]
##STR00043##
[0339] Step 1: (7-(benzyloxy)-2H-indazol-3-yl)methanol [2.1a]
##STR00044##
[0341] To methyl 7-(benzyloxy)-2H-indazole-3-carboxylate (6000 mg, 21.25 mmol) was added THF (Volume: 100 mL), and stirred to dissolve at room temperature. Then the reaction placed in room temperature water bath and 2M LAH in THF (15.94 mL, 31.9 mmol) was added carefully. The reaction was stirred for 2 hours at room temperature or until done by LCMS. The reaction was cooled in ice bath, then quenched carefully by adding additional THF (Volume: 100 mL), then excess water dropwise (3.0 ml). As the salts formed magnesium sulfate was added followed by sodium sulfate. The reaction was removed from ice bath and stirred for 55 minutes, filtered through celite, concentrated to residue to give 4850 mg of the desired product 2.1a in 90% yield, used as is. LC-MS (m/z): 255.2 [M+H]+, 0.71 min.
Step 2: 7-(benzyloxy)-3-(((tert-butyldimethylsilyl)oxy)methyl)-2H-indazole [2.1b]
##STR00045##
[0343] To 2.1a (4850 mg, 19.07 mmol) was added DCM (Volume: 100 mL) and imidazole (4155 mg, 61.0 mmol) stirred at room temperature for 5 minutes. Then TBDMSCl (8624 mg, 57.2 mmol) was added. The reaction was stirred at room temperature for 15 hours or until done by LCMS. To the reaction was added 10 ml of methanol stirred for 2-3 minutes and concentrated most of solvent off. Then was added 300 ml of ethyl acetate, which was washed with saturated sodium bicarbonate, water, saturated salt solution, dried sodium sulfate, filtered and concentrated to residue to give desired product 2.1b assume in quantitative yield, used as is. LC-MS (m/z): 369.5 [M+H]+, 1.16 min.
Step 3: 1-(7-(benzyloxy)-3-(((tert-butyldimethylsilyl)oxy)methyl)-2H-indaz- ol-2-yl)-3,3-dimethylbutan-2-one [2.1c]
##STR00046##
[0345] To 2.1b (6985, 18.95 mmol) was added DMF (Volume: 60 mL) and lithium carbonate (5602 mg, 76 mmol) and the mixture was stirred for 10 minutes at 90.degree. C. Then 1-bromo-3,3-dimethylbutan-2-one (8484 mg, 47.4 mmol) was added with the reaction at 90.degree. C., and then stirred at 90.degree. C. for 15 hours or until done by LCMS. To the cooled reaction was added 450 ml of ethyl acetate, which was washed with saturated sodium bicarbonate, water, saturated salt solution, dried sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel column chromatography, using 0 to 25% EtOAc/heptanes. The desired regio-isomer eluted second and was concentrated to constant mass to give 3100 mg of the desired product 2.1c in 35% yield. LC-MS (m/z): 467.4 [M+H]+, 1.30 min.
Step 4: (2-(2-amino-3,3-dimethylbutyl)-7-(benzyloxy)-2H-indazol-3-yl)metha- nol [2.1d]
##STR00047##
[0347] To 2.1c (3100 mg, 6.64 mmol) was added MeOH (Volume: 29 mL), ammonium acetate (7680 mg, 100 mmol) and sodium cyanoborohydride (1252 mg, 19.93 mmol). The reaction was then stirred at 55.degree. C. for 40 hours or until done by LCMS. To the crude reaction was added 350 ml of DCM and 25 ml of methanol, extracted with 1:1 solution of (6 M NaOH, saturated salt solution). The aqueous layer was back extracted with DCM. The organics were combined, washed with saturated salt solution, dried with sodium sulfate, filtered through 1 cm.times.2 cm celite filter plug, washed with DCM with 10% methanol, concentrated to residue to give the desired product 2.1d (assume in quantitative yield), which was used as is. LC-MS (m/z): 354.3 [M+H]+, 0.73 min.
Step 5: 7-(benzyloxy)-3-(tert-butyl)-3,4-dihydropyrazino[1,2-b]indazole [2.1e]
##STR00048##
[0349] To 2.1d (2340 mg, 6.62 mmol) was added DCM (Volume: 45 mL), and manganese dioxide (5755 mg, 66.2 mmol). The reaction was then stirred room temperature for 2 hours. Then additional manganese dioxide (2878 mg, 33.1 mmol) was added and the mixture was stirred overnight for a total of 20 hours or until done by LCMS. Additional manganese dioxide can be added if needed. To the crude was added 30 ml of DCM stirred for 30 minutes then filtered through a 1 cm.times.2 cm celite filter plug, flushed with DCM and concentrated to residue. The residue was dissolved in 20 ml of DCM and excess TFA (1.530 mL, 19.86 mmol) was added, and the mixture was stirred for 5-10 minutes at room temperature then concentrated to residue to give the desired product 2.1e (assume quantitative yield), which was used as is. LC-MS (m/z): 334.3 [M+H]+, 0.82 min.
Step 6: ethyl 10-(benzyloxy)-6-(tert-butyl)-2-oxo-2,6,7,13c-tetrahydro-1H-pyrido[2',1':- 3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.1f]
##STR00049##
[0351] To 2.1e (2200 mg, 6.60 mmol) was added Ethanol (Volume: 35 mL), and (Z)-ethyl 2-(ethoxymethylene)-3-oxobutanoate (3686 mg, 19.79 mmol). The reaction was then stirred at 85-90.degree. C. for 16 hours or until done by LCMS. The reaction was concentrated to residue to give the desired product 2.1f assume in quantitative yield, used as is. LC-MS (m/z): 474.6 [M+H]+, 0.92 min.
Step 7: ethyl 10-(benzyloxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyraz- ino[1,2-b]indazole-3-carboxylate [2.1g]
##STR00050##
[0353] To 2.1f (3120 mg, 6.59 mmol) was added DME (Volume: 40 mL), and then chloranil (1620 mg, 6.59 mmol). The reaction was stirred at 90-95.degree. C. for 90 minutes or until done by LCMS. The reaction cooled, 2 ml of water was added; stirred for 5 minutes then diluted with 350 ml of DCM with 2% ethanol. Saturated sodium bicarbonate (100 ml) and 150 ml of water were added and the mixture was stirred for 5 minutes. The reaction was filtered and the layers were separated. The organic layer was washed with water 2.times., saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography, using 0-75% EtOAc (with 25% ethanol)/heptane. The desired fractions were concentrated to constant mass to give 1145 mg of the desired product 2.1g in 37% yield. LC-MS (m/z): 472.5 [M+H]+, 0.98 min. 1H NMR (DMSO-d6) .delta.: 8.51 (s, 1H), 7.48-7.57 (m, 3H), 7.30-7.45 (m, 3H), 7.19 (t, J=8.0 Hz, 1H), 6.90 (d, J=7.6 Hz, 1H), 6.81 (s, 1H), 5.27 (s, 2H), 5.00-5.08 (m, 2H), 4.69 (br d, J=3.0 Hz, 1H), 4.22 (q, J=7.0 Hz, 2H), 1.26 (t, J=7.1 Hz, 3H), 0.68 (s, 9H).
Step 8: ethyl 6-(tert-butyl)-10-hydroxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [2.1h]
##STR00051##
[0355] To 2.1g (1120 mg, 2.375 mmol) was flushed with argon then Pd--C(758 mg, 0.713 mmol) was added. Then under argon with a syringe Ethanol (Volume: 40 mL) was added. To the reaction was added a hydrogen filled balloon. The reaction was evacuated and refilled 5.times. with hydrogen and then stirred at room temperature for 2 hours or until done by LCMS. The reaction was stopped by removing the hydrogen balloon and flushing the reaction with argon. Then 100 ml of THF was added and the mixture stirred for 30 minutes. The reaction was filtered through 1.times.2 cm celite plug, flushed with THF and concentrated to residue to give 900 mg of the desired product 2.1h in 99% yield, which was used as is. LC-MS (m/z): 382.5 [M+H]+, 0.68 min.
Step 9: ethyl 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylate [2.1i-I] and [2.1i-II]
##STR00052##
[0357] To 2.1h (425 mg, 1.114 mmol) was added DMF (Volume: 10 mL) and cesium carbonate (1089 mg, 3.34 mmol). The reaction was stirred at room temperature for 10 minutes, then 1-bromo-3-methoxypropane (341 mg, 2.228 mmol) was added and the reaction was heated to 50-55.degree. C. and stirred for 2 hours or until done by LCMS. The reaction was cooled, diluted with 225 ml of DCM with 2% ethanol, washed with saturated sodium bicarbonate, water, saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography, using 0-100% EtOAc (with 25% ethanol)/heptane. The desired fractions were concentrated to constant mass to give 434 mg of the desired racemic product 2.1i in 86% yield. LC-MS (m/z): 454.5 [M+H]+, 0.82 min. 1H NMR (<dmso>) .delta.: 8.52 (s, 1H), 7.50 (d, J=8.5 Hz, 1H), 7.17 (t, J=8.0 Hz, 1H), 6.74-6.83 (m, 2H), 5.05-5.13 (m, 1H), 4.98-5.05 (m, 1H), 4.69 (d, J=4.5 Hz, 1H), 4.11-4.28 (m, 4H), 3.51 (t, J=6.3 Hz, 2H), 3.25 (s, 3H), 2.04 (quin, J=6.3 Hz, 2H), 1.26 (t, J=7.1 Hz, 3H), 0.68 (s, 9H).
[0358] The above racemic material 434 mg was separated by chiral chromatography using OD column, (SFC=80 ml/min, CO.sub.2/MeOH=70/30, 266 bar) to give products 2.1i-I (peak 1, tR 3.93 min.) at 170 mg and product 2.1i-II (peak 2, tR 6.98 min.) at 172 mg.
Step 10: 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.1-I] and [2.1-II]
##STR00053##
[0360] To 2.1i-I (170 mg, 0.375 mmol) was added acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (1.125 ml, 1.125 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water, acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 154 mg of the desired product 2.1-I as free base in 96% yield. LC-MS (m/z): 426.4 [M+H]+, 0.85 min. 1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.18-7.31 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.03-5.26 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 4.20 (t, J=6.4 Hz, 2H), 3.51 (t, J=6.3 Hz, 2H), 3.25 (s, 3H), 2.04 (quin, J=6.3 Hz, 2H), 0.69 (s, 9H). To 2.1i-II (172 mg, 0.379 mmol) was added acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (1.138 ml, 1.138 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water, acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 154 mg of the desired product 2.1-II as free base in 94% yield. LC-MS (m/z): 426.4 [M+H]+, 0.85 min. .sup.1H NMR (DMSO-d6) .delta.: 8.95 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.17-7.28 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.04-5.23 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.20 (t, J=6.4 Hz, 2H), 3.51 (t, J=6.2 Hz, 2H), 3.25 (s, 3H), 2.04 (quin, J=6.3 Hz, 2H), 0.69 (s, 9H)
Example 2.2
6-(tert-butyl)-10-(2-methoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylic acid [2.2-I] and [2.2-II]
##STR00054##
[0361] Step 1: ethyl 6-(tert-butyl)-10-(2-methoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylate [2.2a-I] and [2.2a-II]
##STR00055##
[0363] To 2.1h (425 mg, 1.114 mmol) was added DMF (Volume: 10 mL) and cesium carbonate (1089 mg, 3.34 mmol). The reaction was stirred at room temperature for 10 minutes then 1-bromo-2-methoxyethane (310 mg, 2.228 mmol) was added and the reaction was heated to 50-55.degree. C. and stirred for 2 hours or until done by LCMS. The reaction was cooled, diluted with 225 ml of DCM with 2% ethanol, washed with saturated sodium bicarbonate, water, saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography, using 0-100% EtOAc (with 25% ethanol)/heptane. The desired fractions were concentrated to constant mass to give 414 mg of the desired racemic product 2.2a in 85% yield. LC-MS (m/z): 440.5 [M+H]+, 0.78 min. The above racemic material 414 mg was separated by chiral chromatography using AD Column, (SFC=100 ml/min, CO.sub.2/EtOH=75/25, 226 bar) to give products 2.2a-I (peak 1, tR 3.55 min.) at 134 mg and product 2.2a-II (peak 2, tR 4.97 min.) at 104 mg.
Step 2: 6-(tert-butyl)-10-(2-methoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2'- ,1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.2-I] and [2.2-II]
##STR00056##
[0365] To 2.2a-I (134 mg, 0.305 mmol) was added acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (0.915 ml, 0.915 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 117 mg of the desired product 2.2-I as free base in 93% yield. LC-MS (m/z): 412.4 [M+H]+, 0.80 min. .sup.1H NMR (DMSO-d6) .delta.: 8.94 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.15-7.30 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.05-5.22 (m, 2H), 4.96 (d, J=4.6 Hz, 1H), 4.24-4.34 (m, 2H), 3.70-3.78 (m, 2H), 3.33 (s, 3H), 0.69 (s, 9H). To 2.2a-II (104 mg, 0.237 mmol) was added acetonitrile (Volume: 4 ml, Ratio: 1.000), Water (Volume: 4 ml, Ratio: 1.000) and then LiOH (0.710 ml, 0.710 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water, acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 93 mg of the desired product 2.2-II as free base in 95% yield. LC-MS (m/z): 412.4 [M+H]+, 0.80 min. .sup.1H NMR (DMSO-d6) .delta.: 8.94 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.18-7.32 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.03-5.29 (m, 2H), 4.95 (d, J=4.6 Hz, 1H), 4.23-4.32 (m, 2H), 3.68-3.79 (m, 2H), 3.33 (s, 3H), 0.69 (s, 9H).
Illustrative Example for Synthesis of Compounds Wherein R.sup.1.dbd.F
Step 1: 5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2-dimethylcyclopentanon- e [1a]
##STR00057##
[0367] A mixture of Pd(OAc).sub.2 (8.16 mg, 0.036 mmol), sodium tert-butoxide (0.454 g, 4.72 mmol), dicyclohexyl(2'-methyl-[1,1'-biphenyl]-2-yl)phosphane (32 mg), 2,2-dimethylcyclopentanone (0.547 ml, 4.36 mmol) and 4-bromo-1-methoxy-2-(3-methoxypropoxy)benzene (1 g, 3.63 mmol) in toluene (4.0 mL) was heated in a sealed vial at 50.degree. C. for 18 hours. The mixture was diluted with EtOAc and filtered. The filtrate was concentrated and the remaining oil was purified by silica gel column chromatography, EtOAc/heptane 5 to 50%, to give product (500 mg, 45% yield). LC-MS (m/z): 307.2 [M+H].sup.+. .sup.1H NMR (400 MHz, CDCl.sub.3): 7.72-7.28 (m, 1H), 6.94-6.61 (m, 2H), 4.10 (m, 2H), 3.91-3.77 (m, 4H), 3.65-3.49 (m, 2H), 3.41-3.27 (m, 3H), 2.36 (d, J=4.2 Hz, 1H), 2.19-1.88 (m, 4H), 1.88-1.71 (m, 1H), 1.21-1.11 (m, 3H), 1.07 (s, 3H).
Step 2: 5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2-dimethylcyclopentanam- ine [1b]
##STR00058##
[0369] To the mixture of 5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2-dimethylcyclopentanone (320 mg, 1.0 mmol) in MeOH (3 mL) was added acetic acid ammonia salt (1.6 g, 20.9 mmol) and sodium cyanoborohydride (656 mg, 10.4 mmol). The mixture was stirred at 80.degree. C. for 8 hours and then was concentrated under reduced pressure. The remaining material was diluted with EtOAc, washed with water and brine, dried over Na.sub.2SO.sub.4 and concentrated. The crude material was used in the next step with no further purification. LCMS (m/z): 308.0 [M+H].sup.+.
Step 3: N-(5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2 dimethylcyclopentyl)formamide [1c]
##STR00059##
[0371] To the mixture of 5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2-dimethylcyclopentanamine (320 mg, 1.041 mmol) in dioxane (3 ml) was added formic acid (0.160 mL, 4.16 mmol). The mixture was stirred at 100.degree. C. for 6 hours. The mixture was concentrated to afford the crude product which was used in the next step with no further purification. LCMS (m/z): 336.2 [M+H].sup.+.
Step 4: 7-methoxy-8-(3-methoxypropoxy)-3,3-dimethyl-2,3,3a,9b-tetrahydro-1- H-cyclopenta[c]isoquinoline [1d-I] and [1d-II]
##STR00060##
[0373] To a mixture of N-(5-(4-methoxy-3-(3-methoxypropoxy)phenyl)-2,2-dimethylcyclopentyl)forma- mide (349 mg, 1.04 mmol) in acetonitrile (1.8 ml) was added POCl.sub.3 (140 .mu.l, 1.50 mmol). The mixture was stirred at 85.degree. C. for 2 hours and then concentrated. The residue was dissolved in EtOAc and basified by adding ammonium hydroxide solution. The phases were separated and the organic layer was washed with brine, dried over Na.sub.2SO.sub.4 and concentrated. The remaining material was purified by silica gel chromatography, acetone/heptane 5 to 50% to give product rac-1d-I and rac-1d-II.
[0374] Trans isomer rac-1d-II: (70 mg, 21% yield). LCMS (m/z): 318.3 [M+H].sup.+. .sup.1H NMR (400 MHz, CDCl.sub.3): 8.22 (s, 1H), 6.93-6.83 (m, 1H), 6.70 (s, 1H), 4.16 (t, J=6.4 Hz, 2H), 3.94-3.83 (m, 3H), 3.57 (q, J=4.7 Hz, 2H), 3.35 (d, J=1.5 Hz, 4H), 2.84 (s, 2H), 2.21-1.98 (m, 4H), 1.84-1.70 (m, 3H), 1.68-1.53 (m, 3H), 1.22 (s, 4H), 1.06 (s, 4H).
[0375] Cis isomer rac-1d-I: (54 mg, 16% yield). LCMS (m/z): 318.3 [M+H].sup.+. 1H NMR (400 MHz, CDCl.sub.3): 8.17 (s, 1H), 6.75 (s, 1H), 6.67 (s, 1H), 4.14 (t, J=6.2 Hz, 3H), 3.92-3.83 (m, 3H), 3.77 (d, J=10.3 Hz, 1H), 3.57 (t, J=5.4 Hz, 3H), 3.35 (d, J=1.6 Hz, 3H), 3.25 (q, J=9.4 Hz, 1H), 2.42-2.21 (m, 2H), 2.19-2.01 (m, 3H), 1.64 (dd, J=20.6, 7.8 Hz, 4H), 1.45 (t, J=8.0 Hz, 2H), 1.24 (d, J=6.1 Hz, 4H), 0.89 (s, 4H). The relative configuration of rac-1d-I and rac-1d-II were confirmed by nuclear Overhauser effect (nOe) experiments.
Step 5. Ethyl 8-fluoro-10-methoxy-11-(3-methoxypropoxy)-3,3-dimethyl-7-oxo-1,2,3,3a,7,1- 2b-hexahydrocyclopenta[c]pyrido[2,1-a]isoquinoline-6-carboxylate [1e-I] and [1e-II]
##STR00061##
[0376] ethyl (Z)-2-(ethoxymethyiene)-4,4-difluoro-3-((trimethylsilyl)oxy)but-3-enoate
[0377] Under an argon atmosphere, a mixture of Mg (3.69 g, 152 mmol) and TMSCl (19.43 mL, 152 mmol) was treated with ultrasound irradiation for 15 min. To the mixture was added DMF (30 mL)), ethyl (Z)-2-(ethoxymethylene)-4,4,4-trifluoro-3-oxobutanoate (4.56 g, 19 mmol) was added dropwise at 50.degree. C. under an argon atmosphere. The reaction mixture was stirred for additional 3 min. at 50.degree. C. After removal of excess TMSCl in vacuo, the crude mixture was filtered and the filtrate (containing DMF) was used in the next step without further purification.
##STR00062##
[0378] To a suspension of ZnI.sub.2 (920 mg, 2.88 mmol)) and 1d-I (915 mg, 2.88 mmol) in dry MeCN (10 mL), a solution of crude ethyl (Z)-2-(ethoxymethylene)-4,4-difluoro-3-((trimethylsilyl)oxy)but-3-enoate (5091 mg, 17.30 mmol) in dry DMF (30 mL) was added dropwise at 50.degree. C., and the reaction mixture was stirred overnight. The reaction mixture was poured into 10% HCl and extracted with DCM. The organic layer was washed with brine, and dried over MgSO.sub.4. After filtration, the organic layer was concentrated and the crude oil purified by silica gel chromatography (0-10% MeOH/EtOAc) to afford rac-1e (1.2 g, 2.53 mmol, 88% yield)) as a pale yellow solid. The material was then purified by chiral SFC (AD column, flow rate 100 ml/min, CO.sub.2/EtOH=70/30, 256 bar) to provide to provide products 1e-1 (tR 2.4 min) and 1e-2 (tR 4.4 min, 280 mg). LC-MS (m/z): 474.2 [M+H].sup.+.
Step 6: (3aS,12bR)-8-fluoro-10-methoxy-11-(3-methoxypropoxy)-3,3-dimethyl-- 7-oxo-1,2,3,3a,7,12b-hexahydrocyclopenta[c]pyrido[2,1-a]isoquinoline-6-car- boxylic acid [3.1]
##STR00063##
[0380] To the solution of 1e-2 (330 mg, 0.697 mmol) in THF (1 mL) was added NaOH (1.394 mL, 1.394 mmol). The reaction mixture was stirred for 2 h, then the reaction was acidified with 1.5 ml 1N HCl and extracted with dichloromethane. The organic layer was washed with brine, and dried over MgSO.sub.4. After filtration and concentration, the resultant solid was recrystallized from hot EtOH/Water (5 ml; 5 ml) and the solids collected by vacuum filtration. The material was further lyophilized from MeCN and water to give product (230 mg, 0.511 mmol, 73.3%) as a tan solid. LC-MS (m/z): 446.4 [M+H].sup.+. .sup.1H NMR (500 MHz, DMSO-d.sup.6) .delta. 8.82 (s, 1H), 7.56 (s, 1H), 7.20 (s, 1H), 4.52 (d, J=5.5 Hz, 1H), 4.20-4.06 (m, 2H), 3.82 (s, 3H), 3.31 (dd, J=15.9, 4.8 Hz, 2H), 3.26 (s, 3H), 3.18 (d, J=15.9 Hz, 1H), 2.01 (p, J=6.3 Hz, 2H), 1.57-1.50 (m, 1H), 0.87 (d, J=6.6 Hz, 3H), 0.73 (d, J=6.7 Hz, 3H). For each compound of Formula (I), the enantiomer having greater potency in the HBsAg assay is preferred. By analogy to other compounds believed to bind in the same target site, the more potent isomer in the HBsAg assay in each case is believed to have an absolute stereochemical configuration corresponding to these structures:
##STR00064##
[0381] where the dashed arc indicates R.sup.7 and R.sup.9 taken together form a ring as described for certain compounds of Formula (I) and substructures thereof that are shown herein.
Example 2.3
6-(tert-butyl)-2-oxo-10-(2-(2,2,2-trifluoroethoxy)ethoxy)-6,7-dihydro-2H-p- yrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.3]
##STR00065##
[0382] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(2-(2,2,2-trifluoroethoxy)ethoxy)-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.3a]
##STR00066##
[0384] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then 2-(2-bromoethoxy)-1,1,1-trifluoroethane (21.71 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.3a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 508.5 [M+H].sup.+, 0.88 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(2-(2,2,2-trifluoroethoxy)ethoxy)-6,7-dihy- dro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.3]
##STR00067##
[0386] To 2.3a (21.32 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 8.2 mg of the desired racemic product 2.3 as TFA salt in 32% yield. LC-MS (m/z): 480.4 [M+H].sup.+, 0.89 min. .sup.1H NMR (DMSO-d6) .delta.: 8.94 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.19-7.30 (m, 2H), 6.84 (d, J=7.6 Hz, 1H), 5.06-5.22 (m, 2H), 4.95 (d, J=4.5 Hz, 1H), 4.30-4.38 (m, 2H), 4.20 (q, J=9.4 Hz, 2H), 3.99-4.06 (m, 2H), 0.69 (s, 9H)
Example 2.4
6-(tert-butyl)-10-((1-(methoxymethyl)cyclobutyl)methoxy)-2-oxo-6,7-dihydro- -2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.4]
##STR00068##
[0387] Step 1: ethyl 6-(tert-butyl)-10-((1-(methoxymethyl)cyclobutyl)methoxy)-2-oxo-6,7-dihydr- o-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.4a]
##STR00069##
[0389] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then 1-(bromomethyl)-1-(methoxymethyl)cyclobutane (20.25 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.4a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 494.5 [M+H].sup.+, 0.96 min.
Step 2: 6-(tert-butyl)-10-((1-(methoxymethyl)cyclobutyl)methoxy)-2-oxo-6,7- -dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.4]
##STR00070##
[0391] To 2.4a (20.73 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 4.0 mg of the desired racemic product 2.4 as TFA salt in 16% yield. LC-MS (m/z): 466.5 [M+H].sup.+, 1.00 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.63 (d, J=8.6 Hz, 1H), 7.19-7.27 (m, 2H), 6.87 (d, J=7.6 Hz, 1H), 5.17-5.25 (m, 1H), 5.05-5.13 (m, 1H), 4.94 (d, J=4.9 Hz, 1H), 4.05-4.18 (m, 2H), 3.47 (s, 2H), 3.25 (s, 3H), 1.85-1.97 (m, 6H), 0.69 (s, 9H)
Example 2.5
6-(tert-butyl)-10-(difluoromethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylic acid [2.5]
##STR00071##
[0392] Step 1: ethyl 6-(tert-butyl)-10-(difluoromethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylate [2.5a]
##STR00072##
[0394] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (77 mg, 0.236 mmol). The reaction was stirred at room temperature for 5 minutes then ethyl 2-bromo-3,3-difluoropropanoate (41.0 mg, 0.189 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 20 hours, or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.5a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 432.5 [M+H].sup.+, 0.91 min.
Step 2: 6-(tert-butyl)-10-(difluoromethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2'- ,1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.5]
##STR00073##
[0396] To 2.5a (20.28 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 0.8 mg of the desired racemic product 2.5 as TFA salt in 3% yield. LC-MS (m/z): 404.4 [M+H].sup.+, 0.88 min. .sup.1H NMR (<cdcl3>) .delta.: 8.58 (s, 1H), 7.74 (d, J=8.5 Hz, 1H), 7.35 (t, J=8.0 Hz, 1H), 7.31 (s, 1H), 7.18 (d, J=1.4 Hz, 1H), 6.81-7.17 (m, 1H), 5.22 (d, J=14.9 Hz, 1H), 4.96 (dd, J=14.9, 5.4 Hz, 1H), 4.26 (d, J=5.2 Hz, 1H), 0.85 (s, 9H)
Example 2.6
6-(tert-butyl)-10-((S)-2-methylbutoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3- ,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.6]
##STR00074##
[0397] Step 1: ethyl 6-(tert-butyl)-10-((S)-2-methylbutoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':- 3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.6a]
##STR00075##
[0399] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then (S)-1-bromo-2-methylbutane (15.84 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.6a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 452.5 [M+H].sup.+, 1.01 min.
Step 2: 6-(tert-butyl)-10-((S)-2-methylbutoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.6]
##STR00076##
[0401] To 2.6a (18.97 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 6.5 mg of the desired racemic product 2.6 as TFA salt in 26% yield. LC-MS (m/z): 424.5 [M+H].sup.+, 1.05 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.18-7.28 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.16-5.24 (m, 1H), 5.05-5.14 (m, 1H), 4.94 (d, J=4.8 Hz, 1H), 3.88-4.10 (m, 2H), 1.83-1.98 (m, 1H), 1.50-1.66 (m, 1H), 1.20-1.33 (m, 1H), 1.01 (t, J=6.6 Hz, 3H), 0.92 (t, J=7.4 Hz, 3H), 0.69 (s, 9H)
Example 2.7
10-(sec-butoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyraz- ino[1,2-b]indazole-3-carboxylic acid [2.7]
##STR00077##
[0402] Step 1: ethyl 10-(sec-butoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyra- zino[1,2-b]indazole-3-carboxylate [2.7a]
##STR00078##
[0404] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then 2-bromobutane (14.37 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.7a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 438.5 [M+H].sup.+, 0.92 min.
Step 2: 10-(sec-butoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3- ,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.7]
##STR00079##
[0406] To 2.7a (18.97 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 6.5 mg of the desired racemic product 2.7 as TFA salt in 27% yield. LC-MS (m/z): 410.5 [M+H].sup.+, 0.95 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.60 (dd, J=8.5, 1.8 Hz, 1H), 7.19-7.26 (m, 2H), 6.82 (dd, J=7.7, 3.0 Hz, 1H), 5.14-5.23 (m, 1H), 5.04-5.13 (m, 1H), 4.95 (d, J=4.8 Hz, 1H), 4.66 (dt, J=12.5, 6.2 Hz, 1H), 1.58-1.83 (m, 2H), 1.31 (dd, J=10.9, 6.0 Hz, 3H), 0.89-0.99 (m, 3H), 0.69 (s, 9H)
Example 2.8
6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)methoxy)-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.8]
##STR00080##
[0407] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)methoxy)-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.8a]
##STR00081##
[0409] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then (tetrahydrofuran-3-yl)methyl 4-methylbenzenesulfonate (24.19 mg, 0.094 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.8a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 466.5 [M+H].sup.+, 0.79 min.
Step 2: 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)methoxy)-6,7-dihydr- o-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.8]
##STR00082##
[0411] To 2.8a (21.88 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 3.7 mg of the desired racemic product 2.8 as TFA salt in 13% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.83 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.18-7.30 (m, 2H), 6.85 (d, J=7.5 Hz, 1H), 5.04-5.24 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 4.00-4.21 (m, 2H), 3.72-3.87 (m, 2H), 3.57-3.71 (m, 2H), 2.69-2.84 (m, 1H), 1.99-2.13 (m, 1H), 1.65-1.77 (m, 1H), 0.69 (s, 9H)
Example 2.9
6-(tert-butyl)-2-oxo-10-(2-(trifluoromethoxy)ethoxy)-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.9]
##STR00083##
[0412] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(2-(trifluoromethoxy)ethoxy)-6,7-dihydro-2H-pyrid- o[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.9a]
##STR00084##
[0414] To 2.1h (15 mg, 0.039 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (44.8 mg, 0.138 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-2-(trifluoromethoxy)ethane (18.97 mg, 0.098 mmol) was added. The reaction was heated to 50.degree. C. and stirred for 15 minutes or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.9a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 494.6 [M+H].sup.+, 0.99 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(2-(trifluoromethoxy)ethoxy)-6,7-dihydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.9]
##STR00085##
[0416] To 2.9a (19.25 mg, 0.039 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (156 .mu.l, 0.156 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.3 mg of the desired racemic product 2.9 as TFA salt in 23% yield. LC-MS (m/z): 466.4 [M+H].sup.+, 0.92 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.69 (d, J=8.5 Hz, 1H), 7.21-7.31 (m, 2H), 6.87 (d, J=7.6 Hz, 1H), 5.05-5.28 (m, 2H), 4.96 (d, J=4.7 Hz, 1H), 4.48 (br dd, J=18.4, 4.9 Hz, 4H), 0.69 (s, 9H)
Example 2.10
6-(tert-butyl)-10-(2,2-difluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3- ,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.10]
##STR00086##
[0417] Step 1: ethyl 6-(tert-butyl)-10-(2,2-difluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':- 3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.10a]
##STR00087##
[0419] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 2,2-difluoroethyl trifluoromethanesulfonate (25.3 mg, 0.118 mmol) was added. The reaction was heated to 50.degree. C. and stirred for 15 minutes or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.10a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 446.4 [M+H].sup.+, 0.81 min.
Step 2: 6-(tert-butyl)-10-(2,2-difluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.10]
##STR00088##
[0421] To 2.10a (20.94 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 3.6 mg of the desired racemic product 2.10 as TFA salt in 14% yield. LC-MS (m/z): 418.4 [M+H].sup.+, 0.84 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.72 (d, J=8.5 Hz, 1H), 7.21-7.32 (m, 2H), 6.92 (d, J=7.6 Hz, 1H), 6.35-6.65 (m, 1H), 5.05-5.27 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.51 (td, J=14.5, 3.4 Hz, 2H), 0.69 (s, 9H).
Example 2.11
[0422] 6-(tert-butyl)-2-oxo-10-propoxy-6,7-dihydro-2H-pyrido[2',1':3,4]pyr- azino[1,2-b]indazole-3-carboxylic acid [2.11]
##STR00089##
Step 1: ethyl 6-(tert-butyl)-2-oxo-10-propoxy-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[- 1,2-b]indazole-3-carboxylate [2.11a]
##STR00090##
[0424] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromopropane (14.51 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.11a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 424.5 [M+H].sup.+, 0.89 min.
Step 2: 6-(tert-butyl)-2-oxo-10-propoxy-6,7-dihydro-2H-pyrido[2',1':3,4]py- razino[1,2-b]indazole-3-carboxylic acid [2.11]
##STR00091##
[0426] To 2.11a (19.90 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.4 mg of the desired racemic product 2.11 as TFA salt in 22% yield. LC-MS (m/z): 396.4 [M+H].sup.+, 0.92 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.18-7.29 (m, 2H), 6.81 (d, J=7.6 Hz, 1H), 5.04-5.22 (m, 2H), 4.94 (d, J=4.8 Hz, 1H), 4.02-4.16 (m, 2H), 1.75-1.88 (m, 2H), 1.02 (t, J=7.4 Hz, 3H), 0.69 (s, 9H)
Example 2.12
6-(tert-butyl)-10-(2-(2-methoxyethoxy)ethoxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.12]
##STR00092##
[0427] Step 1: ethyl 6-(tert-butyl)-10-(2-(2-methoxyethoxy)ethoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.12a]
##STR00093##
[0429] To 2.1h (18 mg, 0.047 mmol) was added DMF (volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-2-(2-methoxyethoxy)ethane (21.60 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.12a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 484.5 [M+H].sup.+, 0.77 min.
Step 2: 6-(tert-butyl)-10-(2-(2-methoxyethoxy)ethoxy)-2-oxo-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.12]
##STR00094##
[0431] To 2.12a (22.73 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 6.6 mg of the desired racemic product 2.12 as TFA salt in 24% yield. LC-MS (m/z): 456.5 [M+H].sup.+, 0.81 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.19-7.30 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.04-5.25 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.23-4.34 (m, 2H), 3.79-3.88 (m, 2H), 3.61 (dd, J=5.6, 3.8 Hz, 2H), 3.47 (dd, J=5.6, 3.8 Hz, 2H), 3.23 (s, 3H), 0.69 (s, 9H)
Example 2.13
6-(tert-butyl)-10-(2,3-dimethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1'- :3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.13]
##STR00095##
[0432] Step 1: ethyl 6-(tert-butyl)-10-(2,3-dimethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1- ':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.13a]
##STR00096##
[0434] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-2,3-dimethoxypropane (21.60 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.13a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 484.5 [M+H].sup.+, 0.78 min.
Step 2: 6-(tert-butyl)-10-(2,3-dimethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.13]
##STR00097##
[0436] To 2.13a (20.94 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.6 mg of the desired racemic product 2.13 as TFA salt in 21% yield. LC-MS (m/z): 456.5 [M+H].sup.+, 0.81 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.14-7.29 (m, 2H), 6.84 (dd, J=7.6, 4.0 Hz, 1H), 5.03-5.25 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.11-4.31 (m, 2H), 3.76 (br t, J=4.5 Hz, 1H), 3.47-3.62 (m, 2H), 3.40 (d, J=3.6 Hz, 3H), 3.28 (s, 3H), 0.69 (s, 9H).
Example 2.14
6-(tert-butyl)-10-(3-ethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylic acid [2.14]
##STR00098##
[0437] Step 1: ethyl 6-(tert-butyl)-10-(3-ethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylate [2.14a]
##STR00099##
[0439] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-ethoxypropane (19.71 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.14a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 468.5 [M+H].sup.+, 0.86 min.
Step 2: 6-(tert-butyl)-10-(3-ethoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2'- ,1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.14]
##STR00100##
[0441] To 2.14a (21.98 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 6.2 mg of the desired racemic product 2.14 as TFA salt in 23% yield. LC-MS (m/z): 440.5 [M+H].sup.+, 0.90 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.19-7.29 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.04-5.23 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.20 (t, J=6.4 Hz, 2H), 3.55 (t, J=6.3 Hz, 2H), 3.42 (q, J=7.0 Hz, 2H), 2.04 (quin, J=6.3 Hz, 2H), 1.09 (t, J=7.0 Hz, 3H), 0.69 (s, 9H)
Example 2.15
6-(tert-butyl)-10-(2-isopropoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3- ,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.15]
##STR00101##
[0442] Step 1: ethyl 6-(tert-butyl)-10-(2-isopropoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':- 3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.15a]
##STR00102##
[0444] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reacting was stirred at room temperature for 5 minutes then 2-(2-bromoethoxy)propane (19.71 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.15a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 468.5 [M+H].sup.+, 0.87 min.
Step 2: 6-(tert-butyl)-10-(2-isopropoxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.15]
##STR00103##
[0446] To 2.15a (20.94 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 4.5 mg of the desired racemic product 2.15 as TFA salt in 17% yield. LC-MS (m/z): 440.5 [M+H].sup.+, 0.91 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.19-7.28 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.06-5.23 (m, 2H), 4.95 (d, J=4.5 Hz, 1H), 4.21-4.30 (m, 2H), 3.74-3.82 (m, 2H), 3.66 (dt, J=12.2, 6.1 Hz, 1H), 1.11 (d, J=6.1 Hz, 6H), 0.69 (s, 9H).
Example 2.16
6-(tert-butyl)-10-isobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino- [1,2-b]indazole-3-carboxylic acid [2.16]
##STR00104##
[0447] Step 1: ethyl 6-(tert-butyl)-10-isobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazin- o[1,2-b]indazole-3-carboxylate [2.16a]
##STR00105##
[0449] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-2-methylpropane (16.17 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.16a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 438.5 [M+H].sup.+, 0.95 min.
Step 2: 6-(tert-butyl)-10-isobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylic acid [2.16]
##STR00106##
[0451] To 2.16a (20.56 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.2 mg of the desired racemic product 2.16 as TFA salt in 21% yield. LC-MS (m/z): 410.5 [M+H].sup.+, 1.00 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.14-7.29 (m, 2H), 6.81 (d, J=7.6 Hz, 1H), 5.05-5.29 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 3.77-4.04 (m, 2H), 2.12 (dt, J=13.3, 6.6 Hz, 1H), 1.02 (dd, J=6.5, 4.9 Hz, 6H), 0.69 (s, 9H).
Example 2.17
6-(tert-butyl)-10-(3-hydroxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylic acid [2.17]
##STR00107##
[0452] Step 1: ethyl 6-(tert-butyl)-10-(3-hydroxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylate [2.17a]
##STR00108##
[0454] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-bromopropan-1-ol (16.40 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added. The crude reaction was purified by reverse phase prep LC, the desired fractions were collected, 0.04 ml of 6M HCl was added and lyophilized to give 9.4 mg of the desired racemic product 2.17a as HCl salt in 42% yield used as is. LC-MS (m/z): 440.5 [M+H].sup.+, 0.70 min.
Step 2: 6-(tert-butyl)-10-(3-hydroxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2- ',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.17]
##STR00109##
[0456] To 2.17a (9.4 mg, 0.021 mmol) was dissolved in 1 ml of DMF, then was added LiOH 1M aq (86 .mu.l, 0.086 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude was purified by reverse phase prep LC, the desired fractions were collected, 0.04 ml of 6M HCl was added and lyophilized. The product was re-dissolved in 1:1 ACN/water, additional 0.02 ml of 6M HCl was added and lyophilized again. Then the sample was again redissolved in 1:1 ACN/water and lyophilized to give 3.2 mg of the desired racemic product 2.17 as HCl salt in 31% yield. LC-MS (m/z): 412.4 [M+H].sup.+, 0.71 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.19-7.28 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.04-5.24 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 4.22 (t, J=6.3 Hz, 2H), 3.60 (t, J=6.2 Hz, 2H), 1.95 (quin, J=6.2 Hz, 2H), 0.69 (s, 9H).
Example 2.18
6-(tert-butyl)-10-(2-hydroxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylic acid [2.18]
##STR00110##
[0457] Step 1: ethyl 6-(tert-butyl)-10-(2-hydroxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylate [2.18a]
##STR00111##
[0459] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 2-bromoethanol (14.74 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added. The crude reaction was purified by reverse phase prep LC, the desired fractions were collected, 0.04 ml of 6M HCl was added and lyophilized to give 7.0 mg of the desired racemic product 2.18a as HCl salt in 32% yield used as is. LC-MS (m/z): 426.4 [M+H].sup.+, 0.68 min.
Step 2: 6-(tert-butyl)-10-(2-hydroxyethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2'- ,1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.18]
##STR00112##
[0461] To 2.18a (7.0 mg, 0.016 mmol) was dissolved in 1 ml of DMF, then was added LiOH 1M aq (0.066 mL, 0.066 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude was purified by reverse phase prep LC, the desired fractions were collected, 0.04 ml of 6M HCl was added and lyophilized. The product was re-dissolved in 1:1 ACN/water, additional 0.02 ml of 6M HCl was added and lyophilized again. Then the sample was again redissolved in 1:1 ACN/water and lyophilized to give 2.3 mg of the desired racemic product 2.18 as HCl salt in 29% yield. LC-MS (m/z): 398.4 [M+H].sup.+, 0.69 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.16-7.29 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.07-5.20 (m, 2H), 4.96 (br d, J=3.8 Hz, 1H), 4.17 (t, J=4.9 Hz, 2H), 3.81 (br t, J=4.9 Hz, 2H), 0.69 (s, 9H)
Example 2.19
6-(tert-butyl)-2-oxo-10-(3-(trifluoromethoxy)propoxy)-6,7-dihydro-2H-pyrid- o[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.19]
##STR00113##
[0462] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(3-(trifluoromethoxy)propoxy)-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.19a]
##STR00114##
[0464] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-(trifluoromethoxy)propane (24.42 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.19a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 508.6 [M+H].sup.+, 1.02 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(3-(trifluoromethoxy)propoxy)-6,7-dihydro-- 2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.19]
##STR00115##
[0466] To 2.19a (23.85 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 10.6 mg of the desired racemic product 2.19 as TFA salt in 37% yield. LC-MS (m/z): 480.4 [M+H].sup.+, 0.97 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.17-7.31 (m, 2H), 6.86 (d, J=7.6 Hz, 1H), 5.04-5.23 (m, 2H), 4.95 (d, J=4.6 Hz, 1H), 4.21-4.35 (m, 4H), 2.22 (br t, J=6.2 Hz, 2H), 0.69 (s, 9H).
Example 2.20
6-(tert-butyl)-2-oxo-10-(2-(2-oxopyrrolidin-1-yl)ethoxy)-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.20]
##STR00116##
[0467] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(2-(2-oxopyrrolidin-1-yl)ethoxy)-6,7-dihydro-2H-p- yrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.20a]
##STR00117##
[0469] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-(2-bromoethyl)pyrrolidin-2-one (22.66 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 7 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.20a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 493.5 [M+H].sup.+, 0.93 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(2-(2-oxopyrrolidin-1-yl)ethoxy)-6,7-dihyd- ro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.20]
##STR00118##
[0471] To 2.20a (23.15 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 7.4 mg of the desired racemic product 2.20 as TFA salt in 27% yield. LC-MS (m/z): 465.4 [M+H].sup.+, 0.73 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.18-7.29 (m, 2H), 6.86 (d, J=7.6 Hz, 1H), 5.07-5.24 (m, 2H), 4.95 (d, J=4.2 Hz, 1H), 4.28 (t, J=5.6 Hz, 2H), 3.58-3.68 (m, 2H), 3.47-3.55 (m, 2H), 2.13-2.26 (m, 2H), 1.90 (quin, J=7.5 Hz, 2H), 0.69 (s, 9H).
Example 2.21
6-(tert-butyl)-10-(3-(2-methoxyethoxy)propoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.21]
##STR00119##
[0472] Step 1: ethyl 6-(tert-butyl)-10-(3-(2-methoxyethoxy)propoxy)-2-oxo-6,7-dihydro-2H-pyrid- o[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.21a]
##STR00120##
[0474] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-(2-methoxyethoxy)propane (23.25 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 7 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.21a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 498.6 [M+H].sup.+, 0.88 min.
Step 2: 6-(tert-butyl)-10-(3-(2-methoxyethoxy)propoxy)-2-oxo-6,7-dihydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.21]
##STR00121##
[0476] To 2.21a (23.39 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 12.5 mg of the desired racemic product 2.21 as TFA salt in 44% yield. LC-MS (m/z): 470.5 [M+H].sup.+, 0.82 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.17-7.28 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.05-5.25 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.20 (t, J=6.3 Hz, 2H), 3.59 (t, J=6.3 Hz, 2H), 3.49-3.54 (m, 2H), 3.40-3.44 (m, 2H), 3.21 (s, 3H), 2.04 (quin, J=6.2 Hz, 2H), 0.69 (s, 9H).
Example 2.22
10-((1,4-dioxan-2-yl)methoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2- ',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.22]
##STR00122##
[0477] Step 1: ethyl 10-((1,4-dioxan-2-yl)methoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.22a]
##STR00123##
[0479] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then (1,4-dioxan-2-yl)methyl methanesulfonate (23.15 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.22a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 482.4 [M+H].sup.+, 0.76 min.
Step 2: 10-((1,4-dioxan-2-yl)methoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.22]
##STR00124##
[0481] To 2.22a (22.63 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized. The crude product was re-dissolved in 1 ml of DMF and re-purified by reverse phase prep LC, and lyophilized to give 6.2 mg of the desired racemic product 2.22 as TFA salt in 22% yield. LC-MS (m/z): 454.6 [M+H].sup.+, 0.86 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.19-7.29 (m, 2H), 6.83 (d, J=6.7 Hz, 1H), 5.16-5.25 (m, 1H), 5.05-5.15 (m, 1H), 4.95 (d, J=4.7 Hz, 1H), 4.11-4.18 (m, 2H), 3.91-4.04 (m, 1H), 3.82-3.91 (m, 1H), 3.75-3.82 (m, 1H), 3.61-3.71 (m, 2H), 0.69 (s, 9H).
Example 2.23
6-(tert-butyl)-10-(carboxymethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]p- yrazino[1,2-b]indazole-3-carboxylic acid [2.23]
##STR00125##
[0482] Step 1: ethyl 6-(tert-butyl)-10-(2-methoxy-2-oxoethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',- 1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.23a]
##STR00126##
[0484] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then methyl 2-bromoacetate (16.04 mg, 0.105 mmol) was added. The reaction was heated to 50.degree. C. and stirred for 1 hour or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.23a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 454.4 [M+H].sup.+, 0.76 min.
Step 2: 6-(tert-butyl)-10-(carboxymethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',- 1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.23]
##STR00127##
[0486] To 2.23a (19.05 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 8.0 mg of the desired racemic product 2.23 as TFA salt in 36% yield. LC-MS (m/z): 412.4 [M+H].sup.+, 0.68 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 13.10 (br s, 1H), 8.95 (s, 1H), 7.68 (d, J=8.5 Hz, 1H), 7.28 (s, 1H), 7.22 (t, J=8.0 Hz, 1H), 6.74 (d, J=7.6 Hz, 1H), 5.13 (d, J=2.3 Hz, 2H), 4.96 (br s, 1H), 4.89 (s, 2H), 0.69 (s, 9H).
Example 2.24
6-(tert-butyl)-10-((1-((methylsulfonyl)methyl)cyclopropyl)methoxy)-2-oxo-6- ,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.24]
##STR00128##
[0487] Step 1: ethyl 6-(tert-butyl)-10-((1-((methylsulfonyl)methyl)cyclopropyl)methoxy)-2-oxo-- 6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.24a]
##STR00129##
[0489] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-(bromomethyl)-1-((methylsulfonyl)methyl)cyclopropane (26.8 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.24a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 528.5 [M+H].sup.+, 0.85 min.
Step 2: 6-(tert-butyl)-10-((1-((methylsulfonyl)methyl)cyclopropyl)methoxy)- -2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxyli- c acid [2.24]
##STR00130##
[0491] To 2.24a (24.80 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 2.4 mg of the desired racemic product 2.24 as TFA salt in 8% yield. LC-MS (m/z): 500.4 [M+H].sup.+, 0.78 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.65 (d, J=8.6 Hz, 1H), 7.17-7.29 (m, 2H), 6.75 (d, J=7.6 Hz, 1H), 5.17-5.28 (m, 1H), 5.05-5.15 (m, 1H), 4.96 (br d, J=4.8 Hz, 1H), 4.26 (d, J=10.0 Hz, 1H), 4.07 (d, J=9.9 Hz, 1H), 3.01 (s, 3H), 0.78-0.93 (m, 4H), 0.70 (s, 9H).
Example 2.25
6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-2-yl)methoxy)-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.25]
##STR00131##
[0492] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-2-yl)methoxy)-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.25a]
##STR00132##
[0494] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 2-(bromomethyl)tetrahydrofuran (19.47 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.25a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 466.5 [M+H].sup.+, 0.90 min.
Step 2: 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-2-yl)methoxy)-6,7-dihydr- o-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.25]
##STR00133##
[0496] To 2.25a (21.88 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 7.6 mg of the desired racemic product 2.25 as TFA salt in 29% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.84 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.14-7.30 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.03-5.23 (m, 2H), 4.95 (d, J=4.5 Hz, 1H), 4.20-4.33 (m, 1H), 4.05-4.19 (m, 2H), 3.80 (qd, J=7.1, 3.5 Hz, 1H), 3.64-3.74 (m, 1H), 1.98-2.11 (m, 1H), 1.78-1.97 (m, 2H), 1.66-1.75 (m, 1H), 0.69 (s, 9H).
Example 2.26
6-(tert-butyl)-10-(oxetan-3-yloxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]p- yrazino[1,2-b]indazole-3-carboxylic acid [2.26]
##STR00134##
[0497] Step 1: ethyl 6-(tert-butyl)-10-(oxetan-3-yloxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylate [2.26a]
##STR00135##
[0499] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-bromooxetane (16.16 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.26a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 438.5 [M+H].sup.+, 0.81 min.
Step 2: 6-(tert-butyl)-10-(oxetan-3-yloxy)-2-oxo-6,7-dihydro-2H-pyrido[2',- 1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.26]
##STR00136##
[0501] To 2.26a (20.56 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.4 mg of the desired racemic product 2.26 as TFA salt in 21% yield. LC-MS (m/z): 410.4 [M+H].sup.+, 0.74 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.70 (d, J=8.5 Hz, 1H), 7.28 (s, 1H), 7.20 (t, J=8.0 Hz, 1H), 6.50 (d, J=7.5 Hz, 1H), 5.48 (br t, J=5.4 Hz, 1H), 5.06-5.24 (m, 2H), 4.99 (dt, J=11.9, 5.9 Hz, 3H), 4.64 (ddd, J=11.6, 7.0, 5.1 Hz, 2H), 0.69 (s, 9H).
Example 2.27
6-(tert-butyl)-2-oxo-10-(piperidin-4-yloxy)-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.27]
##STR00137##
[0502] Step 1: ethyl 10-((1-(tert-butoxycarbonyl)piperidin-4-yl)oxy)-6-(tert-butyl)-2-oxo-6,7-- dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.27a]
##STR00138##
[0504] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then tert-butyl 4-bromopiperidine-1-carboxylate (27.7 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 10 ml of ethyl acetate was added, washed with water 2.times., filtered and concentrated to residue to give the desired product 2.27a used as is for the next step, assume quantitative yield. LC-MS (m/z): 565.6 [M+H].sup.+, 0.98 min.
Step 2: ethyl 6-(tert-butyl)-2-oxo-10-(piperidin-4-yloxy)-6,7-dihydro-2H-pyrido[2',1':3- ,4]pyrazino[1,2-b]indazole-3-carboxylate [2.27b]
##STR00139##
[0506] To 2.27a (23.69 mg, 0.042 mmol) was added DCM (Volume: 1.8 mL, Ratio: 3.60), TFA (0.355 mL, 4.61 mmol) and stirred at room temperature for 1 hour, or until done by LCMS. The reaction was concentrated to residue to give the desired product 2.27b used as is for the next step, assume quantitative yield. LC-MS (m/z): 465.5 [M+H].sup.+, 0.61 min.
Step 3: 6-(tert-butyl)-2-oxo-10-(piperidin-4-yloxy)-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.27]
##STR00140##
[0508] To 2.27b (19.51 mg, 0.042 mmol) that was added 1 ml of DMF and LiOH 1M aq (0.294 mL, 0.294 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 3.4 mg of the desired racemic product 2.27 as TFA salt in 14% yield. LC-MS (m/z): 437.5 [M+H].sup.+, 0.64 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.96 (s, 1H), 8.50 (br s, 2H), 7.69 (d, J=8.5 Hz, 1H), 7.15-7.31 (m, 2H), 6.97 (d, J=7.7 Hz, 1H), 5.12 (br s, 2H), 4.95 (br s, 1H), 4.89-4.93 (m, 1H), 3.12 (br s, 2H), 2.17 (br s, 2H), 1.83-1.97 (m, 2H), 0.69 (s, 9H).
Example 2.28
6-(tert-butyl)-10-(2,3-dihydroxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1'- :3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.28]
##STR00141##
[0509] Step 1: ethyl 6-(tert-butyl)-10-((2,2-dimethyl-1,3-dioxolan-4-yl)methoxy)-2-oxo-6,7-dih- ydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.28a]
##STR00142##
[0511] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 4-(bromomethyl)-2,2-dimethyl-1,3-dioxolane (23.01 mg, 0.118 mmol) added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.28a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 496.4 [M+H].sup.+, 0.84 min.
Step 2: 6-(tert-butyl)-10-(2,3-dihydroxypropoxy)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.28]
##STR00143##
[0513] To 2.28a (23.29 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. LC-MS (m/z): 468.4 [M+H].sup.+, 0.89 min. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC using ACN/water with 0.1% TFA. The desired fractions (in ACN/water with 0.1% TFA) were combined and let sit at room temperature for 20 hours to give the desired deprotected product, followed by LCMS. Then 0.2 ml of 3M HCl solution was added and lyophilized. The product was re-dissolved in 1:1 ACN/water and re-lyophilized to give 7.0 mg of the desired racemic product 2.28 as HCl salt in 31% yield. LC-MS (m/z): 428.4 [M+H].sup.+, 0.63 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.18-7.29 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.05-5.24 (m, 2H), 4.96 (br d, J=4.5 Hz, 1H), 4.17 (dt, J=9.8, 3.6 Hz, 1H), 3.99-4.09 (m, 1H), 3.90 (br s, 1H), 3.49 (br d, J=5.6 Hz, 2H), 0.69 (s, 9H).
Example 2.29
6-(tert-butyl)-10-cyclobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazi- no[1,2-b]indazole-3-carboxylic acid [2.29]
##STR00144##
[0514] Step 1: ethyl 6-(tert-butyl)-10-cyclobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyraz- ino[1,2-b]indazole-3-carboxylate [2.29a]
##STR00145##
[0516] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then bromocyclobutane (15.93 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.29a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 436.4 [M+H].sup.+, 0.91 min.
Step 2: 6-(tert-butyl)-10-cyclobutoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.29]
##STR00146##
[0518] To 2.29a (20.47 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 2.0 mg of the desired racemic product 2.29 as TFA salt in 8% yield. LC-MS (m/z): 408.4 [M+H].sup.+, 0.94 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.25 (s, 1H), 7.20 (t, J=8.0 Hz, 1H), 6.66 (d, J=7.5 Hz, 1H), 5.04-5.20 (m, 2H), 4.95 (d, J=4.5 Hz, 1H), 4.89 (quin, J=7.1 Hz, 1H), 2.07-2.20 (m, 2H), 1.83 (q, J=10.2 Hz, 1H), 1.61-1.75 (m, 1H), 0.69 (s, 9H).
Example 2.30
6-(tert-butyl)-10-(3-(methylsulfonyl)propoxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.30]
##STR00147##
[0519] Step 1: ethyl 6-(tert-butyl)-10-(3-(methylsulfonyl)propoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.30a]
##STR00148##
[0521] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-(methylsulfonyl)propane (23.72 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.30a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 502.4 [M+H].sup.+, 0.71 min.
Step 2: 6-(tert-butyl)-10-(3-(methylsulfonyl)propoxy)-2-oxo-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.30]
##STR00149##
[0523] To 2.30a (23.57 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 11.3 mg of the desired racemic product 2.30 as TFA salt in 40% yield. LC-MS (m/z): 474.3 [M+H].sup.+, 0.72 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.18-7.30 (m, 2H), 6.83 (d, J=7.6 Hz, 1H), 5.05-5.25 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.24-4.36 (m, 2H), 3.30-3.35 (m, 2H), 3.04 (s, 3H), 2.18-2.32 (m, 2H), 0.69 (s, 9H).
Example 2.31
6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylic acid [2.31]
##STR00150##
[0524] Step 1: ethyl 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylate [2.31a]
##STR00151##
[0526] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-methoxypropane (18.05 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.31a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 454.5 [M+H].sup.+, 0.82 min.
Step 2: 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2- ',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.31]
##STR00152##
[0528] To 2.31a (21.32 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 9.3 mg of the desired racemic product 2.31 as TFA salt in 36% yield. LC-MS (m/z): 426.4 [M+H].sup.+, 0.83 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.13-7.30 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.04-5.26 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 4.20 (t, J=6.4 Hz, 2H), 3.51 (t, J=6.3 Hz, 2H), 3.25 (s, 3H), 2.04 (t, J=6.3 Hz, 2H), 0.69 (s, 9H).
Example 2.32
10-(azetidin-3-ylmethoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1'- :3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.32]
##STR00153##
[0529] Step 1: ethyl 10-((1-(tert-butoxycarbonyl)azetidin-3-yl)methoxy)-6-(tert-butyl)-2-oxo-6- ,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.32a]
##STR00154##
[0531] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then tert-butyl 3-(bromomethyl)azetidine-1-carboxylate (29.5 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 15 ml of ethyl acetate was added, washed with water 2.times., saturated salt solution, dried sodium sulfate, filtered and concentrated to residue to give the desired product 2.32a used as is for the next step, assume quantitative yield. LC-MS (m/z): 551.7 [M+H].sup.+, 1.01 min.
Step 2: ethyl 10-(azetidin-3-ylmethoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyrido[2',1- ':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.32b]
##STR00155##
[0533] To 2.32a (26.0 mg, 0.047 mmol) was added DCM (Volume: 2 mL, Ratio: 4.00) trifluoroacetic acid (0.4 mL, 5.19 mmol) and stirred at room temperature for 30 minutes or until done by LCMS. The reaction was concentrated to residue. The crude was dissolved in 1 ml of DMF and purified by reverse phase prep LC and lyophilized to give 13.5 mg of the desired product 2.32b in 64% yield, used as is for the next step. LC-MS (m/z): 451.4 [M+H].sup.+, 0.61 min.
Step 3: 10-(azetidin-3-ylmethoxy)-6-(tert-butyl)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.32]
##STR00156##
[0535] To 2.32b (13.5 mg, 0.030 mmol) that was added 1 ml of DMF and LiOH 1M aq LiOH (0.120 mL, 0.120 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 6.6 mg of the desired racemic product 2.32 as TFA salt in 40% yield. LC-MS (m/z): 423.4 [M+H].sup.+, 0.62 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.97 (s, 1H), 8.71 (br d, J=15.6 Hz, 2H), 7.70 (d, J=8.5 Hz, 1H), 7.21-7.33 (m, 2H), 6.88 (d, J=7.6 Hz, 1H), 5.12 (br s, 2H), 4.96 (br s, 1H), 4.37 (br d, J=6.6 Hz, 2H), 4.11 (br s, 2H), 3.91 (br d, J=5.9 Hz, 2H), 0.69 (s, 9H).
Example 2.33
6-(tert-butyl)-2-oxo-10-(((S)-5-oxopyrrolidin-2-yl)methoxy)-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.33-I] and [2.33-II]
##STR00157##
[0536] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(((S)-5-oxopyrrolidin-2-yl)methoxy)-6,7-dihydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.33a]
##STR00158##
[0538] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then (S)-5-(bromomethyl)pyrrolidin-2-one (21.00 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.33a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 479.4 [M+H].sup.+, 0.71 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(((S)-5-oxopyrrolidin-2-yl)methoxy)-6,7-di- hydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.33-I] and [2.33-II]
##STR00159##
[0540] To 2.33a (22.49 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC, using a Sunfire C.sub.18 OBD column, eluting with ACN/water with 0.1% TFA, resulting in desired products 2.33-I (peak 1, tR 5.97 min.) at 3.3 mg in 12% yield and product 2.33-II (peak 2, tR 6.16 min.) at 2.5 mg in 9% yield, both as TFA salts. 2.33-I LC-MS (m/z): 451.4 [M+H]+, 0.70 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.83 (s, 1H), 7.67 (d, J=8.5 Hz, 1H), 7.18-7.30 (m, 2H), 6.85 (d, J=7.6 Hz, 1H), 5.05-5.21 (m, 2H), 4.95 (br d, J=4.2 Hz, 1H), 4.12 (d, J=4.9 Hz, 2H), 3.97 (br dd, J=7.5, 4.5 Hz, 1H), 2.30-2.40 (m, 1H), 2.08-2.26 (m, 2H), 1.90-2.01 (m, 1H), 0.69 (s, 9H). 2.33-II LC-MS (m/z): 451.4 [M+H]+, 0.71 min. .sup.1HNMR (DMSO-d.sub.6) .delta.: 8.96 (s, 1H) 7.84 (s, 1H) 7.67 (d, J=8.51 Hz, 1H) 7.20-7.30 (m, 2H) 6.86 (d, J=7.58 Hz, 1H) 5.07-5.22 (m, 2H) 4.95 (br d, J=4.55 Hz, 1H) 4.06-4.18 (m, 2H) 3.97 (br dd, J=7.51, 4.77 Hz, 1H) 2.28-2.39 (m, 2H) 2.08-2.23 (m, 2H) 1.94 (dt, J=16.81, 4.90 Hz, 1H) 0.69 (s, 9H)
Example 2.34
6-(tert-butyl)-2-oxo-10-(((R)-5-oxopyrrolidin-2-yl)methoxy)-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.34-I] and [2.34-II]
##STR00160##
[0541] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(((R)-5-oxopyrrolidin-2-yl)methoxy)-6,7-dihydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.34a]
##STR00161##
[0543] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then (R)-5-(bromomethyl)pyrrolidin-2-one (21.00 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.34a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 479.4 [M+H].sup.+, 0.70 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(((R)-5-oxopyrrolidin-2-yl)methoxy)-6,7-di- hydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.34-I] and [2.34-II]
##STR00162##
[0545] To 2.34a (22.49 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC, using a Sunfire C18 OBD column, eluting with ACN/water with 0.1% TFA, resulting in desired products 2.34-I (peak 1, tR 5.86 min.) at 4.0 mg in 15% yield and product 2.34-II (peak 2, tR 6.09 min.) at 3.4 mg in 13% yield, both as TFA salts. 2.34-I LC-MS (m/z): 451.4 [M+H]+, 0.70 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.83 (s, 1H), 7.67 (d, J=8.5 Hz, 1H), 7.17-7.28 (m, 2H), 6.85 (d, J=7.6 Hz, 1H), 5.06-5.20 (m, 2H), 4.95 (br d, J=4.2 Hz, 1H), 4.12 (d, J=4.9 Hz, 2H), 3.97 (br dd, J=7.7, 4.5 Hz, 1H), 2.30-2.40 (m, 1H), 2.08-2.26 (m, 2H), 1.89-2.01 (m, 1H), 0.69 (s, 9H). 2.34-II LC-MS (m/z): 451.4 [M+H]+, 0.71 min. .sup.1HNMR (DMSO-d.sub.6) .delta.: 8.96 (s, 1H), 7.84 (s, 1H), 7.67 (d, J=8.5 Hz, 1H), 7.19-7.33 (m, 2H), 6.86 (d, J=7.6 Hz, 1H), 5.03-5.24 (m, 2H), 4.95 (d, J=4.5 Hz, 1H), 4.07-4.23 (m, 2H), 3.97 (br dd, J=7.3, 4.5 Hz, 1H), 2.29-2.39 (m, 1H), 2.08-2.24 (m, 2H), 1.86-2.00 (m, 1H), 0.69 (s, 9H)
Example 2.35
6-(tert-butyl)-10-((1-methoxypropan-2-yl)oxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.35-I] and [2.35-II]
##STR00163##
[0546] Step 1: ethyl 6-(tert-butyl)-10-((1-methoxypropan-2-yl)oxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.35a]
##STR00164##
[0548] To 2.1h (16 mg, 0.042 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (47.8 mg, 0.147 mmol). The reaction was stirred at room temperature for 5 minutes then 2-bromo-1-methoxypropane (16.05 mg, 0.105 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.35a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 454.5 [M+H].sup.+, 0.81 min.
Step 2: 6-(tert-butyl)-10-((1-methoxypropan-2-yl)oxy)-2-oxo-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.35-I] and [2.35-II]
##STR00165##
[0550] To 2.35a (19.05 mg, 0.042 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (168 .mu.l, 0.168 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC, using a Sunfire C.sub.18 OBD column, eluting with ACN/water with 0.1% TFA, resulting in desired products 2.35-I (peak 1, tR 5.50 min.) at 3.0 mg in 13% yield and product 2.35-II (peak 2, tR 5.67 min.) at 3.0 mg in 13% yield, both as TFA salts. 2.35-I LC-MS (m/z): 426.4 [M+H]+, 0.82 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.93 (s, 1H), 7.61 (d, J=8.5 Hz, 1H), 7.18-7.28 (m, 2H), 6.85-6.92 (m, 1H), 5.13-5.22 (m, 1H), 5.03-5.12 (m, 1H), 4.94 (br d, J=4.7 Hz, 1H), 4.88-4.93 (m, 1H), 3.49-3.61 (m, 2H), 3.29 (s, 3H), 1.28 (d, J=6.2 Hz, 3H), 0.69 (s, 9H). 2.35-II LC-MS (m/z): 426.4 [M+H]+, 0.85 min. .sup.1HNMR (DMSO-d.sub.6) .delta.: 8.93 (s, 1H), 7.61 (d, J=8.5 Hz, 1H), 7.18-7.27 (m, 2H), 6.85-6.91 (m, 1H), 5.14-5.20 (m, 1H), 5.05-5.13 (m, 1H), 4.94 (d, J=4.6 Hz, 1H), 4.87-4.93 (m, 1H), 3.49-3.61 (m, 2H), 3.28 (s, 3H), 1.30 (d, J=6.2 Hz, 3H), 0.69 (s, 9H).
Example 2.36
6-(tert-butyl)-10-(2-(dimethylamino)-2-oxoethoxy)-2-oxo-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.36]
##STR00166##
[0551] Step 1: ethyl 6-(tert-butyl)-10-(2-(dimethylamino)-2-oxoethoxy)-2-oxo-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.36a]
##STR00167##
[0553] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 2-bromo-N,N-dimethylacetamide (19.59 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.36a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 467.5 [M+H].sup.+, 0.70 min.
Step 2: 6-(tert-butyl)-10-(2-(dimethylamino)-2-oxoethoxy)-2-oxo-6,7-dihydr- o-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.36]
##STR00168##
[0555] To 2.36a (21.93 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 9.7 mg of the desired racemic product 2.36 as TFA salt in 36% yield. LC-MS (m/z): 439.4 [M+H].sup.+, 0.70 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.27 (s, 1H), 7.21 (t, J=8.0 Hz, 1H), 6.74 (d, J=7.6 Hz, 1H), 5.08-5.19 (m, 2H), 5.00 (d, J=1.7 Hz, 2H), 4.96 (br d, J=2.2 Hz, 1H), 3.03 (s, 3H), 2.85 (s, 3H), 0.69 (s, 9H).
Example 2.37
6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)oxy)-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.37]
##STR00169##
[0557] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)oxy)-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.37a]
##STR00170##
[0558] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-bromotetrahydrofuran (17.81 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.37a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 452.4 [M+H].sup.+, 0.75 min.
Step 2: 6-(tert-butyl)-2-oxo-10-((tetrahydrofuran-3-yl)oxy)-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.37]
##STR00171##
[0560] To 2.37a (21.22 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 7.5 mg of the desired racemic product 2.37 as TFA salt in 29% yield. LC-MS (m/z): 424.4 [M+H].sup.+, 0.77 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.16-7.29 (m, 2H), 6.80 (dd, J=7.5, 4.8 Hz, 1H), 5.25 (br s, 1H), 5.05-5.22 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 3.83-4.00 (m, 3H), 3.78 (tt, J=8.4, 4.3 Hz, 1H), 2.23-2.37 (m, 1H), 1.95-2.16 (m, 1H), 0.69 (s, 9H)
Example 2.38
6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)oxy)-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.38]
##STR00172##
[0561] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)oxy)-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.38a]
##STR00173##
[0563] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 4-bromotetrahydro-2H-pyran (19.47 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 20 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.38a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 466.5 [M+H].sup.+, 0.79 min.
Step 2: 6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)oxy)-6,7-dihydr- o-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.38]
##STR00174##
[0565] To 2.38a (21.88 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 8.1 mg of the desired racemic product 2.38 as TFA salt in 31% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.81 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.64 (d, J=8.5 Hz, 1H), 7.16-7.29 (m, 2H), 6.94 (d, J=7.7 Hz, 1H), 5.05-5.25 (m, 2H), 4.95 (d, J=4.7 Hz, 1H), 4.79-4.91 (m, 1H), 3.89 (ddt, J=11.7, 7.9, 4.0 Hz, 2H), 3.46-3.58 (m, 3H), 2.06 (br dd, J=9.9, 4.9 Hz, 2H), 1.67 (ddd, J=13.1, 9.1, 3.7 Hz, 2H), 0.69 (s, 9H).
Example 2.39
6-(tert-butyl)-10-((3,3-difluorocyclobutyl)methoxy)-2-oxo-6,7-dihydro-2H-p- yrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.39]
##STR00175##
[0566] Step 1: ethyl 6-(tert-butyl)-10-((3,3-difluorocyclobutyl)methoxy)-2-oxo-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.39a]
##STR00176##
[0568] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-(bromomethyl)-1,1-difluorocyclobutane (21.83 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.39a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 486.4 [M+H].sup.+, 0.92 min.
Step 2: 6-(tert-butyl)-10-((3,3-difluorocyclobutyl)methoxy)-2-oxo-6,7-dihy- dro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.39]
##STR00177##
[0570] To 2.39a (22.82 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 9.1 mg of the desired racemic product 2.39 as TFA salt in 33% yield. LC-MS (m/z): 458.4 [M+H].sup.+, 0.94 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.66 (d, J=8.5 Hz, 1H), 7.19-7.32 (m, 2H), 6.85 (d, J=7.6 Hz, 1H), 5.05-5.29 (m, 2H), 4.95 (d, J=4.8 Hz, 1H), 4.14-4.32 (m, 2H), 2.61-2.86 (m, 3H), 0.69 (s, 9H).
Example 2.40
6-(tert-butyl)-2-oxo-10-(3,3,3-trifluoropropoxy)-6,7-dihydro-2H-pyrido[2',- 1'. 3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.40]
##STR00178##
[0571] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-(3,3,3-trifluoropropoxy)-6,7-dihydro-2H-pyrido[2'- ,1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.40a]
##STR00179##
[0573] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-bromo-1,1,1-trifluoropropane (50.1 mg, 0.283 mmol) was added. The reaction was heated to 85.degree. C. and stirred for 40 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.40a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 478.4 [M+H]+, 0.89 min.
Step 2: 6-(tert-butyl)-2-oxo-10-(3,3,3-trifluoropropoxy)-6,7-dihydro-2H-py- rido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.40]
##STR00180##
[0575] To 2.40a (22.44 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 2.0 mg of the desired racemic product 2.40 as TFA salt in 7% yield. LC-MS (m/z): 450.3 [M+H].sup.+, 0.91 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.69 (d, J=8.5 Hz, 1H), 7.19-7.34 (m, 2H), 6.90 (d, J=7.6 Hz, 1H), 5.06-5.24 (m, 2H), 4.95 (br d, J=4.7 Hz, 1H), 4.41 (t, J=6.0 Hz, 2H), 2.90 (tt, J=11.5, 5.6 Hz, 2H), 0.69 (s, 9H).
Example 2.41
6-(tert-butyl)-10-(2-fluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]p- yrazino[1,2-b]indazole-3-carboxylic acid [2.41]
##STR00181##
[0576] Step 1: ethyl 6-(tert-butyl)-10-(2-fluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylate [2.41a]
##STR00182##
[0578] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-2-fluoroethane (14.98 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.41a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 428.4 [M+H].sup.+, 0.77 min.
Step 2: 6-(tert-butyl)-10-(2-fluoroethoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',- 1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.41]
##STR00183##
[0580] To 2.41a (20.09 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 9.5 mg of the desired racemic product 2.41 as TFA salt in 39% yield. LC-MS (m/z): 400.3 [M+H].sup.+, 0.79 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.68 (d, J=8.5 Hz, 1H), 7.12-7.34 (m, 2H), 6.85 (d, J=7.6 Hz, 1H), 5.06-5.25 (m, 2H), 4.96 (d, J=4.7 Hz, 1H), 4.87-4.93 (m, 1H), 4.72-4.81 (m, 1H), 4.43-4.50 (m, 1H), 4.36-4.42 (m, 1H), 0.69 (s, 9H)
Example 2.42
6-(tert-butyl)-10-ethoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1,- 2-b]indazole-3-carboxylic acid [2.42]
##STR00184##
[0581] Step 1: ethyl 6-(tert-butyl)-10-ethoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazino[1- ,2-b]indazole-3-carboxylate [2.42a]
##STR00185##
[0583] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then bromoethane (12.86 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.42a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 410.4 [M+H].sup.+, 0.82 min.
Step 2: 6-(tert-butyl)-10-ethoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyr- azino[1,2-b]indazole-3-carboxylic acid [2.42]
##STR00186##
[0585] To 2.42a (19.25 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 7.2 mg of the desired racemic product 2.42 as TFA salt in 29% yield. LC-MS (m/z): 382.4 [M+H].sup.+, 0.87 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.94 (s, 1H), 7.62 (d, J=8.5 Hz, 1H), 7.16-7.30 (m, 2H), 6.80 (d, J=7.6 Hz, 1H), 5.04-5.23 (m, 2H), 4.95 (d, J=4.6 Hz, 1H), 4.20 (q, J=7.0 Hz, 2H), 1.42 (t, J=6.9 Hz, 3H), 0.69 (s, 9H)
Example 2.43
6-(tert-butyl)-10-(3-(dimethylamino)propoxy)-2-oxo-6,7-dihydro-2H-pyrido[2- ',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.43]
##STR00187##
[0586] Step 1: ethyl 6-(tert-butyl)-10-(3-(dimethylamino)propoxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.43a]
##STR00188##
[0588] To 2.1h (22 mg, 0.058 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (56.4 mg, 0.173 mmol). The reaction was stirred at room temperature for 5 minutes then 3-bromo-N,N-dimethylpropan-1-amine (14.37 mg, 0.087 mmol) was added. The reaction was heated to 50.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The crude reaction was purified by reverse phase prep LC and lyophilized to give 17 mg of the desired racemic product 2.43a as TFA salt in 63% yield. LC-MS (m/z): 467.5 [M+H].sup.+, 0.62 min.
Step 2: 6-(tert-butyl)-10-(3-(dimethylamino)propoxy)-2-oxo-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.43]
##STR00189##
[0590] To 2.43a (17 mg, 0.036 mmol) was added 1 ml of DMF and LiOH 1M aq (0.146 mL, 0.146 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 9.0 mg of the desired racemic product 2.43 as TFA salt in 43% yield. LC-MS (m/z): 439.4 [M+H].sup.+, 0.62 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 9.47 (br s, 1H), 8.97 (s, 1H), 7.68 (d, J=8.5 Hz, 1H), 7.20-7.32 (m, 2H), 6.85 (d, J=7.6 Hz, 1H), 5.11 (br s, 2H), 4.96 (br s, 1H), 4.25 (br t, J=5.4 Hz, 2H), 3.24-3.30 (m, 2H), 2.84 (d, J=4.0 Hz, 6H), 2.21 (br dd, J=9.2, 6.1 Hz, 2H), 0.69 (s, 9H).
Example 2.44
6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)methoxy)-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.44]
##STR00190##
[0591] Step 1: ethyl 6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)methoxy)-6,7-dihydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.44a]
##STR00191##
[0593] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 4-(bromomethyl)tetrahydro-2H-pyran (21.12 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.44a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 480.4 [M+H]+, 0.84 min.
Step 2: 6-(tert-butyl)-2-oxo-10-((tetrahydro-2H-pyran-4-yl)methoxy)-6,7-di- hydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.44]
##STR00192##
[0595] To 2.44a (22.54 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq. (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 8.6 mg of the desired racemic product 2.44 as TFA salt in 32% yield. LC-MS (m/z): 452.4 [M+H].sup.+, 0.86 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.19-7.30 (m, 2H), 6.82 (d, J=7.6 Hz, 1H), 5.16-5.24 (m, 1H), 5.04-5.15 (m, 1H), 4.95 (d, J=4.8 Hz, 1H), 3.97-4.11 (m, 2H), 3.89 (br dd, J=11.2, 2.8 Hz, 2H), 2.07-2.16 (m, 1H), 1.72 (br t, J=14.6 Hz, 2H), 1.31-1.46 (m, 2H), 0.69 (s, 9H)
Example 2.45
6-(tert-butyl)-10-((3-methyloxetan-3-yl)methoxy)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.45]
##STR00193##
[0596] Step 1: ethyl 6-(tert-butyl)-10-((3-methyloxetan-3-yl)methoxy)-2-oxo-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.45a]
##STR00194##
[0598] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 3-(bromomethyl)-3-methyloxetane (19.47 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.45a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 466.4 [M+H].sup.+, 0.79 min.
Step 2: 6-(tert-butyl)-10-((3-methyloxetan-3-yl)methoxy)-2-oxo-6,7-dihydro- -2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.45]
##STR00195##
[0600] To 2.45a (21.88 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 1.0 mg of the desired racemic product 2.45 as TFA salt in 4% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.81 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.96 (s, 1H), 7.67 (d, J=8.5 Hz, 1H), 7.19-7.31 (m, 2H), 6.90 (d, J=7.6 Hz, 1H), 5.24 (br d, J=14.9 Hz, 1H), 5.09 (br dd, J=14.7, 5.2 Hz, 1H), 4.95 (br d, J=5.0 Hz, 1H), 4.54 (t, J=5.5 Hz, 2H), 4.35 (dd, J=5.8, 3.1 Hz, 2H), 4.17-4.31 (m, 2H), 1.42 (s, 3H), 0.70 (s, 9H).
Example 2.46
6-(tert-butyl)-10-((1-cyanocyclopropyl)methoxy)-2-oxo-6,7-dihydro-2H-pyrid- o[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.46]
##STR00196##
[0601] Step 1: ethyl 6-(tert-butyl)-10-((1-cyanocyclopropyl)methoxy)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [2.46a]
##STR00197##
[0603] To 2.1h (18 mg, 0.047 mmol) was added DMF (Volume: 0.5 mL) and cesium carbonate (53.8 mg, 0.165 mmol). The reaction was stirred at room temperature for 5 minutes then 1-(bromomethyl)cyclopropanecarbonitrile (18.88 mg, 0.118 mmol) was added. The reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, 0.5 ml of DMF was added, then filtered through a 0.45 nM in line filter. The DMF solution with the desired product 2.46a was used as is for the next step, assume quantitative yield. LC-MS (m/z): 461.4 [M+H].sup.+, 0.80 min.
Step 2: 6-(tert-butyl)-10-((1-cyanocyclopropyl)methoxy)-2-oxo-6,7-dihydro-- 2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [2.46]
##STR00198##
[0605] To 2.46a (21.64 mg, 0.047 mmol) that was already in 1 ml of DMF was added LiOH 1M aq (188 .mu.l, 0.188 mmol). The reaction was stirred at room temperature for 30 minutes or until done by LCMS. Additional LiOH 1M aq. can be added as needed. The crude reaction was purified by reverse phase prep LC and lyophilized to give 5.8 mg of the desired racemic product 2.46 as TFA salt in 22% yield. LC-MS (m/z): 433.4 [M+H].sup.+, 0.82 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.95 (s, 1H) 7.70 (d, J=8.51 Hz, 1H) 7.17-7.31 (m, 2H) 6.79 (d, J=7.58 Hz, 1H) 5.21-5.32 (m, 1H) 5.06-5.18 (m, 1H) 4.97 (d, J=4.99 Hz, 1H) 4.10-4.33 (m, 2H) 1.36-1.49 (m, 2H) 1.13-1.29 (m, 2H) 0.70 (s, 9H).
Example 2.47
6-(tert-butyl)-10-isopropoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazin- o[1,2-b]indazole-3-carboxylic acid [2.47-] and [2.47-II]
##STR00199##
[0606] Step 1: ethyl 6-(tert-butyl)-10-isopropoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]pyrazi- no[1,2-b]indazole-3-carboxylate [2.47a-I] and [2.47a-II]
##STR00200##
[0608] To 2.1h (250 mg, 0.655 mmol) was added DMF (Volume: 5 mL) and cesium carbonate (641 mg, 1.966 mmol). The reaction was stirred at room temperature for 10 minutes then 2-bromopropane (161 mg, 1.311 mmol) was added and the reaction was heated to 70.degree. C. and stirred for 3 hours or until done by LCMS. The reaction was cooled, diluted with 100 ml of DCM with 3% ethanol, washed with water 3.times., saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography, using 0-85% EtOAc (with 25% ethanol)/heptane. The desired fractions were concentrated to constant mass to give 234 mg of the desired racemic product 2.47a in 84% yield. LC-MS (m/z): 424.6 [M+H]+, 0.94 min. .sup.1H NMR (DMSO-d.sub.6) .delta.: 8.51 (s, 1H), 7.47 (d, J=8.5 Hz, 1H), 7.17 (t, J=8.0 Hz, 1H), 6.79 (t, J=3.7 Hz, 2H), 5.04-5.12 (m, 1H), 4.96-5.04 (m, 1H), 4.87 (dt, J=12.1, 6.0 Hz, 1H), 4.69 (d, J=4.4 Hz, 1H), 4.22 (q, J=7.1 Hz, 2H), 1.34 (t, J=6.4 Hz, 6H), 1.26 (t, J=7.1 Hz, 3H), 0.69 (s, 9H). The racemic material 2.47a, 370 mg was separated by chiral chromatography using (AD column, SFC=100 ml/min, CO.sub.2/MeOH=80/20, 266 bar) to give products 2.47a-I (peak 1, tR 3.72 min.) at 157 mg and product 2.47a-II (peak 2, tR 5.63 min.) at 170 mg.
Step 2: 6-(tert-butyl)-10-isopropoxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4- ]pyrazino[1,2-b]indazole-3-carboxylic acid [2.47-I] and [2.47-II]
##STR00201##
[0610] To 2.47a-I (157 mg, 0.371 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (1.112 ml, 1.112 mmol). The reaction was stirred at room temperature for 16 hours or until done by LCMS. The reaction was diluted with 15 ml of water, acidified with 1M HCl to pH of 2-3, then 150 ml of DCM with 2% ethanol was added and 10 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again 1.times.75 ml DCM with 2% ethanol. The organic layers were combined, washed with water 4.times.75 ml, filtered and concentrated to residue. The residue was dissolved in 10 ml of 1:1 ACN/water, filtered and lyophilized to give 136.8 mg of the desired product 2.47-I as free base in 92% yield. LC-MS (m/z): 396.3 [M+H].sup.+, 0.91 min. .sup.1H NMR (400 MHz, METHANOL-d4) .delta. ppm: 8.82 (s, 1H) 7.52 (d, J=8.46 Hz, 1H) 7.26-7.36 (m, 2H) 6.85 (d, J=7.58 Hz, 1H) 5.09-5.19 (m, 1H) 5.02-5.09 (m, 1H) 4.90-4.96 (m, 1H) 4.76 (d, J=4.50 Hz, 1H) 1.44 (d, J=6.06 Hz, 6H) 0.81 (s, 9H). To 2.47a-II (170 mg, 0.401 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (1.204 ml, 1.204 mmol). The reaction was stirred at room temperature for 16 hours or until done by LCMS. The reaction was diluted with 15 ml of water, acidified with 1M HCl to pH of 2-3, then 150 ml of DCM with 2% ethanol was added and 10 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again 1.times.75 ml DCM with 2% ethanol. The organic layers were combined, washed with water 4.times.75 ml, filtered and concentrated to residue. The residue was dissolved in 10 ml of 1:1 ACN/water, filtered and lyophilized to give 143.3 mg of the desired product 2.47-II as free base in 89% yield. LC-MS (m/z): 396.3 [M+H].sup.+, 0.90 min. .sup.1H NMR (400 MHz, METHANOL-d4) .delta. ppm: 8.82 (s, 1H) 7.51 (d, J=8.46 Hz, 1H) 7.26-7.38 (m, 2H) 6.85 (d, J=7.63 Hz, 1H) 5.10-5.18 (m, 1H) 5.03-5.09 (m, 1H) 4.89-4.96 (m, 1H) 4.76 (d, J=4.45 Hz, 1H) 1.44 (d, J=6.06 Hz, 6H) 0.80 (s, 9H)
Example 3.1
6-(1-hydroxy-2-methylpropan-2-yl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyrido[2- ',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [3.1-I] and [3.1-II]
##STR00202##
[0611] Step 1: 4-(benzyloxy)-1-bromo-3,3-dimethylbutan-2-one [3.1a]
##STR00203##
[0613] To 4-(benzyloxy)-3,3-dimethylbutan-2-one (48.17 g, 234 mmol) in MeOH (Volume: 700 mL) cooled to -78.degree. C., Bromine (13.23 mL, 257 mmol) was added and stirred to rt. Saturated Sodium bicarbonate was added until the solution is basic. The organics were extracted into DCM, dried with Sodium Sulfate and concentrated to give the pdt. The crude was purified by silica gel chromatography using Heptane-EtOAc (0-15% EtOAc, 30 min) to give 33.2g of 3.1a in 49.9% yield. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 7.25-7.38 (m, 5H) 4.47-4.51 (m, 2H) 4.23 (s, 2H) 3.41-3.46 (m, 2H) 1.23 (s, 6H)
Step 2: 4-(benzyloxy)-1-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methox- y-2H-indazol-2-yl)-3,3-dimethylbutan-2-one [3.1b]
##STR00204##
[0615] To 1.1b (3.4 g, 11.63 mmol) and 3.1a (13.26 g, 46.5 mmol) in DMSO (Volume: 124 ml, Ratio: 1.000)-Water (Volume: 6.22 ml, Ratio: 0.05), cesium carbonate (22.73 g, 69.8 mmol) was added and stirred at rt for 4 hrs. The crude was extracted into DCM and concentrated. EtOAc was added and washed with brine to remove any DMSO and purified by silica gel chromatography using Heptane-EtOAc (0-20% EtOAc, 23 min gradient elution) to give 1.24g of 3.1b in 21.5% yield. LC-MS (m/z): 497.6 [M+H]+,1.23 min.
Step 3: (2-(2-amino-4-(benzyloxy)-3,3-dimethylbutyl)-7-methoxy-2H-indazol-- 3-yl)methanol [3.1c]
##STR00205##
[0617] To 3.1b (1.6 g, 3.22 mmol) in Methanol (Volume: 30 ml), Ammonium acetate (4.97 g, 64.4 mmol) and Sodium cyanoborohydride (2.024 g, 32.2 mmol) were added and stirred at 70.degree. C. for 3 days. The crude was diluted with DCM and washed with saturated sodium bicarbonate and the organics concentrated. The organics containing 3.1c were taken to the next step without further purification, assume quantitative yield. LC-MS (m/z): 384.4 [M+H]+,0.74 min.
Step 4: 3-(1-(benzyloxy)-2-methylpropan-2-yl)-7-methoxy-3,4-dihydropyrazin- o[1,2-b]indazole [3.1d]
##STR00206##
[0619] To 3.1c (1.6 g, 4.17 mmol) in DCM (Volume: 40 mL), Manganese dioxide (7.25 g, 83 mmol) was added and stirred at rt. The reaction stalls after stirring at rt for 2 hrs. The crude was filtered and the filtrate was again treated with Manganese dioxide (7.25 g, 83 mmol) and stirred overnight. The crude reaction was again filtered and concentrated to give the crude product 3.1d which was taken to the next step as is, assume quantitative yield. LC-MS (m/z): 364.4 [M+H].sup.+,0.82 min.
Step 5: Ethyl 6-(1-(benzyloxy)-2-methylpropan-2-yl)-10-methoxy-2-oxo-1,6,7,13c-tetrahyd- ro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [3.1e]
##STR00207##
[0621] 3.1d (0.994 g, 2.73 mmol) and ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate (1.528 g, 8.20 mmol) was heated in EtOH (Volume: 17 mL) at 120.degree. C. overnight. The crude containing 3.1e was concentrated and taken to the next step, assume quantitative yield. LC-MS (m/z): 504.5 [M+H].sup.+,0.98 min.
Step 6: Ethyl 6-(1-(benzyloxy)-2-methylpropan-2-yl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [3.1f]
##STR00208##
[0623] To 3.1e (1375 mg, 2.73 mmol) in DME (Volume: 40 ml), Chloramine-T (738 mg, 3.00 mmol) was added and heated at reflux for 2 hrs. The crude was concentrated and redissolved in DCM-Methanol (minimum amount) and purified by silica gel chromatography first using Heptane-EtOAc (0-100%) to remove all non-polar SM/By-products and then purified with DCM-Methanol (0-20% MeOH) to give after concentration 630 mg of 3.1f in 46% yield. LC-MS (m/z): 502.4 [M+H].sup.+,0.96 min.
Step 7: Ethyl 6-(1-hydroxy-2-methylpropan-2-yl)-10-methoxy-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [3.1 g-I] and [3.1 g-II]
##STR00209##
[0625] To 3.1f (630 mg, 1.256 mmol) in EtOH (Volume: 40 mL), 10% palladium on carbon (800 mg, 7.52 mmol) was added and purged with nitrogen followed by hydrogen. A hydrogen balloon was attached and the crude stirred for 4 hrs. The reaction needs to be monitored closely as by-products form when the debenzylation is 70-80% done. The reaction was filtered at this point and the crude was concentrated. The concentrated crude was purified by silica gel chromatography using Heptane-EtOAc with 25% EtOH (0-100%) to give after concentration, 139 mg of 3.1g in 26.9% yield which was given for chiral purification. LC-MS (m/z): 412.4 [M+H].sup.+,0.65 min. The above racemic material 139 mg was separated by chiral chromatography using (OD column, SFC=80 ml/min, CO2/MeOH=75/25, 266 bar) to give products 3.1g-I (peak 1, tR 5.17 min.) at 42 mg and product 3.1g-II (peak 2, tR 9.86 min.) at 43 mg.
Step 8: 6-(1-hydroxy-2-methylpropan-2-yl)-10-methoxy-2-oxo-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [3.1-I] and [3.1-II]
##STR00210##
[0627] To 3.1g-I (42 mg, 0.102 mmol) in Acetonitrile (Volume: 1, Ratio: 1.000)-Water (Volume: 1, Ratio: 1.000), Lithium hydroxide (15 mg, 0.357 mmol) was added and stirred at rt. After 1 hr, the crude was acidified with 1M Sulfuric acid in 100 ml water (until red on pH paper) and extracted into DCM and concentrated. The organics were lyophilized to give 32.5 mg of 3.1-I in 79% yield. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.89 (s, 1H), 7.44 (d, J=8.51 Hz, 1H), 7.23-7.33 (m, 2H), 6.74 (d, J=7.53 Hz, 1H), 5.17 (d, J=14.53 Hz, 1H), 4.85-5.02 (m, 2H), 4.07 (s, 3H), 3.27-3.42 (m, 2H), 1.03 (s, 3H), 0.36 (s, 3H). LC-MS (m/z): 384.3 [M+H].sup.+,0.69 min. To 3.1g-II (42 mg, 0.102 mmol) in Acetonitrile (Volume: 1, Ratio: 1.000)-Water (Volume: 1, Ratio: 1.000), Lithium hydroxide (15 mg, 0.357 mmol) was added and stirred at rt. After 1 hr, the crude was acidified with 1M Sulfuric acid in 100 ml water (until red on pH paper) and extracted into DCM and concentrated. The organics were lyophilized to give 32.5 mg of 3.1-II in 79% yield. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.89 (s, 1H), 7.44 (d, J=8.46 Hz, 1H), 7.21-7.33 (m, 2H), 6.74 (d, J=7.48 Hz, 1H), 5.17 (br d, J=14.48 Hz, 1H), 4.85-5.02 (m, 2H), 4.07 (s, 3H), 3.27-3.41 (m, 2H), 0.99-1.20 (m, 3H), 0.35 (s, 3H). LC-MS (m/z): 384.3 [M+H].sup.+,0.69 min.
Example 4.1: 10-methoxy-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [4.1-I] and [4.1-II]
##STR00211##
[0628] Step 1: 1-bromo-4-methoxy-3,3-dimethylbutan-2-one
##STR00212##
[0630] To 4-methoxy-3,3-dimethylbutan-2-one (9.81 g, 75 mmol) in 200 ml MeOH (Volume: 250 mL) cooled to -78.degree. C., Br2 (4.27 mL, 83 mmol) in 50 ml was added dropwise and stirred to rt overnight. Saturated Sodium bicarbonate was added until the solution is basic. The organics are extracted into DCM, dried with Sodium Sulfate and concentrated to give 4.1a assume in quantitative yield. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 4.24 (s, 2H) 3.35 (s, 2H) 3.31 (s, 3H) 1.21 (s, 6H)
Step 2: 1-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2- -yl)-4-methoxy-3,3-dimethylbutan-2-one [4.1b]
##STR00213##
[0632] To 1.1b (1.5 g, 5.13 mmol) and 4.1a (2.7 g, 12.91 mmol) in DMSO (Volume: 47.9 ml, Ratio: 1.000))-Water (Volume: 3.36 ml, Ratio: 0.07), cesium carbonate (6.68 g, 20.52 mmol) was added and stirred at rt overnight. The crude was extracted into DCM and concentrated. EtOAc was then added to the crude and washed with brine to remove any DMSO and purified by silica gel chromatography using Heptane-EtOAc (0-50% EtOAc) to give after concentration 572 mg of 4.1b (more polar on ISCO) in 26.5% yield. LC-MS (m/z): 421.4 [M+H].sup.+,1.26 min.
Step 3: (2-(2-amino-4-methoxy-3,3-dimethylbutyl)-7-methoxy-2H-indazol-3-yl- )methanol [4.1c]
##STR00214##
[0634] To 4.1b (572 mg, 1.360 mmol) in MeOH (Volume: 7), ammonium acetate (1048 mg, 13.60 mmol) and sodium cyanoborohydride (0.9 g, 14.32 mmol) were added and stirred at 70.degree. C. for 3 days. The crude was diluted with DCM and washed with saturated sodium bicarbonate and the organics concentrated. The organics containing 4.1c were taken to the next step without further purification, assume quantitative yield. LC-MS (m/z): 308.4 [M+H].sup.+,0.53 min.
Step 4: 7-methoxy-3-(1-methoxy-2-methylpropan-2-yl)-3,4-dihydropyrazino[1,- 2-b]indazole [4.1d]
##STR00215##
[0636] To 4.1c (0.418 g, 1.36 mmol) in DCM (Volume: 20 mL), MnO2 (4.3 g, 49.5 mmol) was added and stirred overnight. The crude was filtered through celite and the celite washed with DCM. The organic layers were concentrated and the crude containing 4.1 d was taken to the next step, assume quantitative yield. LC-MS (m/z): 288.3 [M+H].sup.+,0.60 min.
Step 5: Ethyl 10-methoxy-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-1,6,7,13c-tetrahydro-2- H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [4.1e]
##STR00216##
[0638] 4.1d (0.391 g, 1.36 mmol) and ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate (0.760 g, 4.08 mmol) was heated in EtOH (Volume: 6.80 ml) at 100.degree. C. overnight. The crude containing 4.1e was concentrated and taken to the next step, assume quantitative yield. LC-MS (m/z): 428.4 [M+H].sup.+,0.78 min.
Step 6: Ethyl 10-methoxy-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [4.1f-I] and [4.1f-II]
##STR00217##
[0640] To 4.1e (1210 mg, 2.83 mmol) in DME (Volume: 20 mL), Chloramine-T (834 mg, 3.40 mmol) was added and heated at reflux for 2 hrs. The crude was concentrated and redissolved in DCM-Methanol (minimum amount) and purified by silica gel chromatography first using Heptane-EtOAc (0-100%) to remove all non-polar SM/By-products and then purified with DCM-Methanol (0-20% MeOH) to give after concentration 130 mg of 4.1f in 10.8% yield. LC-MS (m/z): 426.4 [M+H].sup.+,0.77 min. The above racemic material 130 mg was separated by chiral chromatography using (OD column, SFC=100 ml/min, CO.sub.2/MeOH=65/35, 266 bar) to give products 4.1f-I (peak 1, tR 2.04 min.) at 38 mg and product 4.1f-II (peak 2, tR 4.47 min.) at 37 mg.
Step 7: 10-methoxy-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-6,7-dihydro-2H-- pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [4.1-I] and [4.1-II]
##STR00218##
[0642] To 4.1f-I (38 mg, 0.089 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (11.24 mg, 0.268 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 29 mg of the desired product 4.1-I as free base in 78% yield. LC-MS (m/z): 398.4 [M+H].sup.+, 0.79 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.69 (s, 1H), 7.45 (d, J=8.46 Hz, 1H), 7.17-7.36 (m, 2H), 6.74 (d, J=7.53 Hz, 1H), 5.12 (d, J=14.92 Hz, 1H), 4.91 (dd, J=5.45, 14.94 Hz, 1H), 4.73 (d, J=5.23 Hz, 1H), 4.07 (s, 3H), 3.37 (s, 3H), 3.02 (d, J=9.93 Hz, 1H), 2.93 (d, J=9.93 Hz, 1H), 1.03 (s, 3H), 0.36 (s, 3H). To 4.1f-II (37 mg, 0.087 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (10.95 mg, 0.261 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 26 mg of the desired product 4.1-II as free base in 71.5% yield. LC-MS (m/z): 398.4 [M+H].sup.+, 0.79 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.69 (s, 1H), 7.45 (d, J=8.46 Hz, 1H), 7.17-7.36 (m, 2H), 6.74 (d, J=7.53 Hz, 1H), 5.12 (d, J=14.92 Hz, 1H), 4.92 (dd, J=5.43, 14.87 Hz, 1H), 4.73 (d, J=5.18 Hz, 1H), 4.07 (s, 3H), 3.37 (s, 3H), 3.02 (d, J=9.98 Hz, 1H), 2.93 (d, J=9.93 Hz, 1H), 1.03 (s, 3H), 0.36 (s, 3H).
Example 5.1: 12-methoxy-3,3-dimethyl-7-oxo-2,3,3a,4a-tetrahydro-1H, 7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [5.1-I] and [5.1-II]
##STR00219##
[0643] Step 1: 5-bromo-2,2-dimethylcyclopentan-1-one [5.1a]
##STR00220##
[0645] To 2,2-dimethylcyclopentan-1-one (25 g, 223 mmol) in MeOH (Volume: 446 ml), Bromine (12.63 ml, 245 mmol) was added at crushed ice temperature and stirred overnight. The crude was washed with sat. Sodium bicarbonate and extracted into DCM, dried with anhydrous sodium sulfate and concentrated to give 5.1a, assume in quantitative yield. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 4.30 (dd, J=6.92, 4.77 Hz, 1H) 2.30-2.52 (m, 1H) 2.05-2.24 (m, 2H) 1.74-1.88 (m, 1H) 1.15-1.28 (s, 3H) 1.07 (s, 3H)
Step 2: 5-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2- -yl)-2,2-dimethylcyclopentan-1-one [5.1b]
##STR00221##
[0647] To 5.1a (5.41 g, 28.3 mmol) and 1.1b (2.07 g, 7.08 mmol) in DMSO (Volume: 25 ml, Ratio: 0.833)-Water (Volume: 1.5 mL, Ratio: 0.05), cesium carbonate (13.84 g, 42.5 mmol) was added and stirred at rt overnight. The crude was extracted into DCM and concentrated. EtOAc was added and washed with brine to remove any DMSO and purified by silica gel chromatography using Heptane-EtOAc (0-50% EtOAc) to give after concentration the desired product, 5.1b (1.14 g, 2.83 mmol, 40.0% yield) is the more polar product eluting around 40-50% EtOAc. LC-MS (m/z): 403.5 [M+H].sup.+,1.30 min.
Step 3: (2-(2-amino-3,3-dimethylcyclopentyl)-7-methoxy-2H-indazol-3-yl)met- hanol [5.1c]
##STR00222##
[0649] To 5.1b (1.14 g, 2.83 mmol) in MeOH (Volume: 15 ml), ammonium acetate (2.183 g, 28.3 mmol) and sodium cyanoborohydride (0.9 g, 14.32 mmol) were added and stirred at 70.degree. C. for 3 days. The crude was diluted with DCM and washed with saturated sodium bicarbonate and the organics concentrated. The organics containing 5.1c were taken to the next step without further purification, assume quantitative yield. LC-MS (m/z): 290.4 [M+H].sup.+,0.61 min.
Step 4: 9-methoxy-3,3-dimethyl-2,3,3a, 11a-tetrahydro-1H-cyclopenta[5,6]pyrazino[1,2-b]indazole [5.1d]
##STR00223##
[0651] To 5.1c (0.819 g, 2.83 mmol) in DCM (Volume: 30 mL), MnO2 (5.5 g, 63.3 mmol) was added and stirred overnight. The crude was filtered through celite and the celite washed with DCM. The organic layers were concentrated and the crude containing 5.1d was taken to the next step, assume quantitative yield. LC-MS (m/z): 270.3 [M+H].sup.+,0.61 min.
Step 5: Ethyl 12-methoxy-3,3-dimethyl-7-oxo-2,3,3a,8,8a,14a-hexahydro-1H,7H-cyclopenta[- 5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylate [5.1e]
##STR00224##
[0653] 5.1d (0.762 g, 2.83 mmol) and ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate (1.581 g, 8.49 mmol) was heated in EtOH (Volume: 14.15 ml) at 120.degree. C. overnight. The crude containing 5.1e was concentrated and taken to the next step, assume quantitative yield. LC-MS (m/z): 410.4 [M+H].sup.+,0.78 min.
Step 6: Ethyl 12-methoxy-3,3-dimethyl-7-oxo-2,3,3a, 14a-tetrahydro-1H,7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-6-carboxylate [5.1f-I] and [5.1f-II]
##STR00225##
[0655] To 5.1e (1159 mg, 2.83 mmol) in DME (Volume: 30 mL), Chloramine-T (834 mg, 3.40 mmol) was added and heated at reflux for 2 hrs. The crude was concentrated and redissolved in DCM-Methanol (minimum amount) and purified by silica gel chromatography first using Heptane-EtOAc (0-100%) to remove all non-polar SM/By-products and then purified with DCM-Methanol (0-20% MeOH) to give after concentration 243 mg of 5.1f in 21% yield. LC-MS (m/z): 408.4 [M+H].sup.+,0.86 min. The above racemic material 243 mg was separated by chiral chromatography using (SFC=100 ml/min, CO.sub.2/MeOH=70/30, 246 bar) to give products 5.1f-I (peak 1, tR 3.58 min.) at 82 mg and product 5.1f-II (peak 2, tR 5.40 min.) at 66 mg.
Step 7: 12-methoxy-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,7H-cyclopen- ta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [5.1-I] and [5.1-II]
##STR00226##
[0657] To 5.1f-I (82 mg, 0.201 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (25.3 mg, 0.604 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 51 mg of the desired product 5.1-I as free base in 63.5% yield. LC-MS (m/z): 380.4 [M+H].sup.+, 0.83 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.51 (s, 1H), 7.50 (br d, J=8.41 Hz, 1H), 7.20-7.41 (m, 2H), 6.75 (br d, J=7.29 Hz, 1H), 5.24 (br s, 1H), 4.43 (br d, J=5.92 Hz, 1H), 4.08 (s, 3H), 3.23-3.44 (m, 1H), 2.53 (br dd, J=6.53, 14.60 Hz, 1H), 1.73-1.90 (m, 1H), 1.51-1.73 (m, 1H), 1.35 (s, 3H), 0.48 (s, 3H). To 5.1f-II (66 mg, 0.162 mmol) was added Acetonitrile (Volume: 5 ml, Ratio: 1.000), Water (Volume: 5 ml, Ratio: 1.000) and then LiOH (20.39 mg, 0.486 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 53 mg of the desired product 5.1-II as free base in 82% yield. LC-MS (m/z): 380.3 [M+H].sup.+, 0.82 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.51 (s, 1H), 7.49 (br d, J=8.51 Hz, 1H), 7.21-7.41 (m, 2H), 6.75 (br d, J=7.48 Hz, 1H), 5.24 (br t, J=5.55 Hz, 1H), 4.43 (br d, J=6.36 Hz, 1H), 4.08 (s, 3H), 3.26-3.41 (m, 1H), 2.53 (br dd, J=6.55, 14.77 Hz, 1H), 1.81 (ddd, J=5.40, 8.36, 13.33 Hz, 1H), 1.51-1.73 (m, 1H), 1.35 (s, 3H), 0.48 (s, 3H).
Example 5.2
12-fluoro-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H, 7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [5.2-1] and [5.2-II]
##STR00227##
[0658] Step 1 to 6: Ethyl 12-fluoro-3,3-dimethyl-7-oxo-2,3,3a, 14a-tetrahydro-1H,7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-6-carboxylate [5.2f-I] and [5.2f-II]
##STR00228##
[0660] Compound 5.2f was synthesized from the starting material; 3-(((tert-butyldimethylsilyl)oxy)methyl)-7-fluoro-2H-indazole (which is from methyl 7-fluoro-2H-indazole-3-carboxylate analogous to method used for preparing 1.1b) by the process of Example 5.1 following steps 1 through 6 resulting in 310 mg of 5.2f as a racemate in 27.7% yield. LC-MS (m/z): 396.5 [M+H].sup.+, 0.91 min. The above racemic material (310 mg) was separated by chiral chromatography using (OD column, SFC=100 ml/min, CO.sub.2/MeOH80/20, 226 bar) to give products 5.2f-I (peak 1, tR 3.95 min.) at 110 mg and product 5.2f-II (peak 2, tR 6.11 min.) at 105 mg.
Step 7: 12-fluoro-3,3-dimethyl-7-oxo-2,3,3a, 14a-tetrahydro-1H,7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-6-carboxylic acid [5.2-I] and [5.2-II]
##STR00229##
[0662] To 5.2f-I (105 mg, 0.266 mmol) was added Acetonitrile (Volume: 4 ml, Ratio: 1.000), Water (Volume: 4 ml, Ratio: 1.000) and then LiOH (33.4 mg, 0.797 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 7 mg of the desired product 5.2-I as free base in 6.8% yield. LC-MS (m/z): 368.3 [M+H].sup.+, 0.83 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 8.52 (s, 1H) 7.73 (d, J=8.51 Hz, 1H) 7.30-7.40 (m, 2H) 7.13 (dd, J=10.78, 7.56 Hz, 1H) 5.16-5.37 (m, 1H) 4.45 (d, J=6.50 Hz, 1H) 3.21-3.41 (m, 1H) 2.54 (dtd, J=14.80, 8.59, 8.59, 6.16 Hz, 1H) 1.84 (ddd, J=13.61, 8.72, 5.06 Hz, 1H) 1.64 (dt, J=13.55, 8.27 Hz, 1H) 1.36 (s, 3H) 0.48 (s, 3H). To 5.2f-II (110 mg, 0.278 mmol) was added Acetonitrile (Volume: 4 ml, Ratio: 1.000), Water (Volume: 4 ml, Ratio: 1.000) and then LiOH (35 mg, 0.835 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 7 mg of the desired product 5.2-II as free base in 6.5% yield. LC-MS (m/z): 368.3 [M+H].sup.+, 0.83 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 8.51 (s, 1H) 7.73 (d, J=8.51 Hz, 1H) 7.27-7.38 (m, 2H) 7.07-7.25 (m, 1H) 5.26 (t, J=5.75 Hz, 1H) 4.43 (d, J=6.50 Hz, 1H) 3.26-3.41 (m, 1H) 2.45-2.64 (m, 1H) 1.84 (ddd, J=13.58, 8.69, 5.06 Hz, 1H) 1.59-1.70 (m, 1H) 1.36 (s, 3H) 0.48 (s, 3H).
Example 6.1
12-(3-methoxypropoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,7H-cyclo- penta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [6.1-I] and [6.1-II]
##STR00230##
[0663] Step 1 to 6: Ethyl 12-(benzyloxy)-3,3-dimethyl-7-oxo-2,3,3a, 14a-tetrahydro-1H,7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-6-carboxylate [6.1f]
##STR00231##
[0665] Compound 6.1f was synthesized from the starting material; 2.1b by the process of Example 5.1 following steps 1 through 6 resulting in the desired product 6.1f as a racemate. LC-MS (m/z): 484.4 [M+H].sup.+, 0.99 min.
Step 7: Ethyl 12-hydroxy-3,3-dimethyl-7-oxo-2,3,3a, 14a-tetrahydro-1H,7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indaz- ole-6-carboxylate [6.1 g]
##STR00232##
[0667] To 6.1 f (315 mg, 0.651 mmol) in EtOH (Volume: 25 ml), nitrogen was purged followed by addition of 10% Pd--C(350 mg, 0.329 mmol) and purged with nitrogen again. A hydrogen balloon was then attached and the reaction mixture purged with hydrogen. The crude was stirred for 1 hour and filtered on celite and purged with DCM-Ethanol. The crude was concentrated to yield crude 262 mg of 6.1g in quantitative yield. LC-MS (m/z): 394.4 [M+H].sup.+, 0.69 min.
Step 8: Ethyl 12-(3-methoxypropoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,7H-cycl- openta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylate [6.1 h-I] and [6.1 h-II]
##STR00233##
[0669] To 6.1g (150 mg, 0.381 mmol) in DMF (Volume: 5 mL), 1-bromo-3-methoxypropane (0.083 mL, 0.763 mmol) and cesium carbonate (373 mg, 1.144 mmol) were added and stirred. The crude was filtered and rinsed with EtOAc and washed with sat. brine to remove any DMF. The crude was purified by silica gel chromatography using Heptane-(EtOAc:EtOH=75:25) (0-100%) to give after concentration 99 mg of the desired product 6.1h in 55.8% yield, which was given for chiral purification. LC-MS (m/z): 466.5 [M+H].sup.+, 0.84 min.
[0670] The above racemic material (99 mg) was separated by chiral chromatography using (OD column, SFC=100 ml/min, CO2/MeOH=75/25, 246 bar) to give products 6.1h-I (peak 1, tR 5.24 min.) at 41 mg and product 6.1h-II (peak 2, tR 7.61 min.) at 38 mg.
Step 9: 12-(3-methoxypropoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,- 7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [6.1-I] and [6.1-II]
##STR00234##
[0672] To 6.1h-I (41 mg, 0.088 mmol) was added Acetonitrile (Volume: 2 ml, Ratio: 1.000), Water (Volume: 1 ml, Ratio: 1.000) and then LiOH (11.09 mg, 0.264 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 19 mg of the desired product 6.1-I as free base in 46.8% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.88 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.48 (s, 1H), 7.40-7.57 (m, 1H), 7.25-7.34 (m, 2H), 6.78 (d, J=7.58 Hz, 1H), 5.23 (t, J=5.70 Hz, 1H), 4.29-4.44 (m, 3H), 3.55-3.71 (m, 2H), 3.29-3.40 (m, 4H), 2.38-2.64 (m, 1H), 2.24 (quin, J=6.32 Hz, 2H), 1.81 (ddd, J=5.14, 8.66, 13.60 Hz, 1H), 1.53-1.68 (m, 1H), 1.34 (s, 3H), 0.48 (s, 3H). To 6.1h-II (38 mg, 0.082 mmol) was added Acetonitrile (Volume: 2 ml, Ratio: 1.000), Water (Volume: 1 ml, Ratio: 1.000) and then LiOH (10.28, 0.245 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 19 mg of the desired product 6.1-II as free base in 50.5% yield. LC-MS (m/z): 438.4 [M+H].sup.+, 0.89 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.49 (s, 1H), 7.47 (d, J=8.56 Hz, 1H), 7.24-7.34 (m, 2H), 6.78 (d, J=7.58 Hz, 1H), 5.23 (t, J=5.89 Hz, 1H), 4.28-4.44 (m, 3H), 3.53-3.74 (m, 2H), 3.29-3.40 (m, 4H), 2.38-2.65 (m, 1H), 2.24 (quin, J=6.32 Hz, 2H), 1.81 (ddd, J=5.23, 8.66, 13.60 Hz, 1H), 1.54-1.68 (m, 1H), 1.34 (s, 3H), 0.48 (s, 3H)
Example 6.2
12-(2-methoxyethoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H, 7H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [6.2-1] and [6.2-II]
##STR00235##
[0673] Step 1: Ethyl 12-(2-methoxyethoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,7H-cyclo- penta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylate [6.2h-1] and [6.2h-II]
##STR00236##
[0675] To 6.1g (150 mg, 0.381 mmol) in DMF (Volume: 3 mL), 1-bromo-2-methoxyethane (0.076 mL, 0.763 mmol) and cesium carbonate (373 mg, 1.144 mmol) were added and stirred. The crude was filtered and rinsed with EtOAc and washed with sat. brine to remove any DMF. The crude was purified by silica gel chromatography using Heptane-(EtOAc:EtOH=75:25) (0-100%) to give after concentration 61 mg of 6.2h in 35.4% yield. LC-MS (m/z): 452.4 [M+H].sup.+, 0.80 min.
[0676] The above racemic material (61 mg) was separated by chiral chromatography using (AD column, SFC=100 ml/min, CO.sub.2/MeOH=70/30, 236 bar) to give products 6.2h-I (peak 1, tR 2.93 min.) at 28 mg and product 6.2h-II (peak 2, tR 4.91 min.) at 29 mg.
Step 2: 12-(2-methoxyethoxy)-3,3-dimethyl-7-oxo-2,3,3a,14a-tetrahydro-1H,7- H-cyclopenta[5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [6.2-I] and [6.2-II]
##STR00237##
[0678] To 6.2h-I (28 mg, 0.062 mmol) was added Acetonitrile (Volume: 2 ml, Ratio: 1.000), Water (Volume: 1 ml, Ratio: 1.000) and then LiOH (7.81 mg, 0.186 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 15 mg of the desired product 6.2-I as free base in 54.3% yield. LC-MS (m/z): 424.4 [M+H].sup.+, 0.82 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.48 (s, 1H), 7.42-7.55 (m, 1H), 7.25-7.33 (m, 2H), 6.77 (d, J=7.63 Hz, 1H), 5.23 (t, J=5.70 Hz, 1H), 4.30-4.48 (m, 3H), 3.86-3.99 (m, 2H), 3.49 (s, 3H), 3.21-3.43 (m, 1H), 2.51 (dt, J=8.46, 14.77 Hz, 1H), 1.80 (ddd, J=5.16, 8.68, 13.60 Hz, 1H), 1.49-1.68 (m, 1H), 1.34 (s, 3H), 0.47 (s, 3H). To 6.2h-II (29 mg, 0.064 mmol) was added Acetonitrile (Volume: 2 ml, Ratio: 1.000), Water (Volume: 1 ml, Ratio: 1.000) and then LiOH (8.09 mg, 0.193 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 15 mg of the desired product 6.2-II as free base in 52.4% yield. LC-MS (m/z): 424.4 [M+H].sup.+, 0.82 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.48 (s, 1H), 7.48 (d, J=8.51 Hz, 1H), 7.25-7.34 (m, 2H), 6.76 (d, J=7.58 Hz, 1H), 5.23 (br t, J=5.77 Hz, 1H), 4.32-4.48 (m, 3H), 3.84-4.01 (m, 2H), 3.49 (s, 3H), 3.24-3.42 (m, 1H), 2.51 (br dd, J=6.24, 14.79 Hz, 1H), 1.80 (ddd, J=5.14, 8.68, 13.57 Hz, 1H), 1.63-1.71 (m, 1H), 1.33 (s, 3H), 0.46 (s, 3H).
Example 7.1
6-(1-methoxy-2-methylpropan-2-yl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-- 2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [7.1-I] and [7.1-II]
##STR00238##
[0679] Step 1: 1-(7-(benzyloxy)-3-(((tert-butyldimethylsilyl)oxy)methyl)-2H-indazol-2-yl- )-4-methoxy-3,3-dimethylbutan-2-one [7.1b]
##STR00239##
[0681] To 2.1b (7.5 g, 20.35 mmol) in DIPEA (35.5 ml, 204 mmol), 4.1a (14.44 g, 69.1 mmol) was added and heated with stirring for 4 hrs. at 110.degree. C. The crude was dissolved in 20 ml DCM and washed with 0.723 ml Sulfuric Acid in water and the organics concentrated. The organics were purified by silica gel chromatography using Heptane-EtOAc (0-50%). The desired regio-isomer eluted second and was concentrated to give 5.503 g of the desired product 7.1b in 57.3% yield. LC-MS (m/z): 497.5 [M+H].sup.+, 1.32 min.
Step 2: (2-(2-amino-4-methoxy-3,3-dimethylbutyl)-7-(benzyloxy)-2H-indazol-- 3-yl)methanol [7.1c]
##STR00240##
[0683] To 7.1b (5.5 g, 11.07 mmol) in MeOH (Volume: 40 ml)ammonium acetate (10.5 g, 136 mmol) and sodium cyanoborohydride (3.48 g, 55.4 mmol) were added and stirred at 70.degree. C. for 3 days. The crude was diluted with DCM and washed with saturated sodium bicarbonate and the organics concentrated. The organics containing 7.1c were taken to the next step without further purification, assume quantitative yield. LC-MS (m/z): 384.5 [M+H].sup.+, 0.70 min.
Step 3: 7-(benzyloxy)-3-(1-methoxy-2-methylpropan-2-yl)-3,4-dihydropyrazin- o[1,2-b]indazole [7.1d]
##STR00241##
[0685] To 7.1c (4.25 g, 11.07 mmol) in DCM (Volume: 50 ml), MnO2 (9.62 g, 111 mmol) was added and stirred at rt overnight. The crude was filtered and the collected MnO2 rinsed with DCM and concentrated to yield crude 7.1 d used as is, assume quantitative yield. LC-MS (m/z): 364.5 [M+H].sup.+, 0.91 min.
Step 4: Ethyl 10-(benzyloxy)-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-1,6,7,13c-tetrahyd- ro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [7.1e]
##STR00242##
[0687] 7.1d (4.02 g, 11.07 mmol) and ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate (8.25 g, 44.3 mmol) was heated in EtOH (Volume: 55 ml) at 120.degree. C. overnight. The crude containing 7.1e was concentrated and taken to the next step as is, assume quantitative yield. LC-MS (m/z): 504.5 [M+H].sup.+, 0.95 min.
Step 5: Ethyl 10-(benzyloxy)-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-6,7-dihydro-2H-pyr- ido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [7.1f]
##STR00243##
[0689] To 7.1e (5.57 g, 11.07 mmol) was added DME (Volume: 80 mL) and then iodine (8.43 g, 33.2 mmol). The reaction was stirred at 35-40.degree. C. overnight. The reaction was diluted with 400 ml of DCM, treated with saturated sodium bicarbonate solution carefully to avoid heat and CO2 evolution, saturated sodium bisulfite to remove residual iodine/HI & saturated salt solution. The organics were separated and dried with anhydrous sodium sulfate, filtered and concentrated to residue. The residue was purified by silica gel chromatography using 0-100% Heptane/EtOAc-EtOH (75:25), concentrated to residue to give 1.51g of 7.1 f in 27.2% yield. LC-MS (m/z): 502.5 [M+H].sup.+, 0.95 min.
Step 6: Ethyl 10-hydroxy-6-(1-methoxy-2-methylpropan-2-yl)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [7.1 g]
##STR00244##
[0691] To 7.1f (1.51 g, 3.01 mmol) in MeOH (Volume: 30.1 ml), nitrogen was purged followed by addition of degussa 10% Pd--C(1.5 g, 1.410 mmol) and purged with nitrogen again. A hydrogen balloon was then attached and the reaction mixture purged with hydrogen. The crude was stirred for 1 hour and filtered on celite and rinsed with DCM-Ethanol. The crude was concentrated to give 1.04g of 7.1 g in 84% yield and taken to the next step without further purification. LC-MS (m/z): 412.4 [M+H].sup.+, 0.66 min.
Step 7: Ethyl 6-(1-methoxy-2-methylpropan-2-yl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro- -2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [7.1 h-I] and [7.1 h-II]
##STR00245##
[0693] To 7.1g (900 mg, 2.187 mmol) in DMF (Volume: 8750 .mu.l), 1-bromo-3-methoxypropane (478 .mu.l, 4.37 mmol) and cesium carbonate (2138 mg, 6.56 mmol) were added and stirred. The crude was purified by silica gel chromatography using 0-100% heptane-(EtOAc/EtOH:75/25) to give after concentration 667 mg of 7.1h in 63% yield. LC-MS (m/z): 484.5 [M+H].sup.+, 0.79 min.
[0694] The above racemic material (667 mg) was separated by chiral chromatography using (AD column, SFC=100 ml/min, CO.sub.2/MeOH=75/25, 236 bar) to give products 7.1h-I (peak 1, tR 2.71 min.) at 279 mg and product 7.1h-II (peak 2, tR 5.07 min.) at 279 mg.
Step 8: 6-(1-methoxy-2-methylpropan-2-yl)-10-(3-methoxypropoxy)-2-oxo-6,7-- dihydro-2H-pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [7.1-I] and [7.1-II]
##STR00246##
[0696] To 7.1h-I (279 mg, 0.577 mmol) was added Acetonitrile (Volume: 20 ml, Ratio: 1.000), Water (Volume: 20 ml, Ratio: 1.000) and then LiOH (72.6 mg, 1.731 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 234 mg of the desired product 7.1-I as free base in 85% yield. LC-MS (m/z): 456.5 [M+H].sup.+, 0.82 min. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. ppm 8.76 (s, 1H) 7.63 (br d, J=8.41 Hz, 1H) 7.12-7.34 (m, 2H) 6.82 (br d, J=7.63 Hz, 1H) 5.12-5.24 (m, 1H) 4.95-5.12 (m, 2H) 4.20 (brt, J=6.38 Hz, 2H) 3.45-3.63 (m, 2H) 3.25 (s, 3H) 2.95-3.09 (m, 4H) 2.81 (br d, J=9.93 Hz, 1H) 1.87-2.13 (m, 2H) 0.88 (s, 3H) 0.36 (s, 3H). To 7.1h-II (279 mg, 0.577 mmol) was added Acetonitrile (Volume: 20 ml, Ratio: 1.000), Water (Volume: 20 ml, Ratio: 1.000) and then LiOH (72.6 mg, 1.731 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 242 mg of the desired product 7.1-II as free base in 87% yield. LC-MS (m/z): 456.5 [M+H].sup.+, 0.82 min; .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. ppm 8.69 (s, 1H) 7.43 (d, J=8.51 Hz, 1H) 7.27-7.33 (m, 2H) 6.77 (d, J=7.58 Hz, 1H) 5.15 (d, J=14.87 Hz, 1H) 4.90 (dd, J=14.89, 5.31 Hz, 1H) 4.72 (d, J=5.38 Hz, 1H) 4.33 (t, J=6.65 Hz, 2H) 3.62 (t, J=5.82 Hz, 2H) 3.37 (s, 6H) 3.02 (d, J=9.93 Hz, 1H) 2.93 (d, J=9.98 Hz, 1H) 2.25 (quin, J=6.32 Hz, 2H) 1.04 (s, 3H) 0.37 (s, 3H).
Example 8.1
6-(tert-butyl)-1-fluoro-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[- 2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [8.1-I] and [8.1-II]
##STR00247##
[0697] Step 1: (Z)-ethyl 2-(ethoxymethylene)-4,4-difluoro-3-((trimethylsilyl)oxy)but-3-enoate [8.1a]
##STR00248##
[0699] Under an argon atmosphere, a mixture of magnesium powder (1.214 g, 50 mmol) and trimethylsilyl chloride (6.34 mL, 50 mmol)) was sonicated prior to the reaction for 15-20 minutes. Then a solution of ethyl (Z)-2-(ethoxymethylene)-4,4,4-trifluoro-3-oxobutanoate (1.5 g, 6.25 mmol) in dry DMF (3 ml) was added dropwise over 5-6 minutes at 50.degree. C. under an argon atmosphere. The reaction mixture was stirred for additional 30 min. at 50.degree. C. The crude mixture was then filtered through a disposable filter funnel with polyethylene frit. The resulting DMF solution with the desired product 8.1a assume in quantitative yield, used as is.
Step 2: Ethyl 10-(benzyloxy)-6-(tert-butyl)-1-fluoro-2-oxo-6,7-dihydro-2H-pyrido[2',1':- 3,4]pyrazino[1,2-b]indazole-3-carboxylate [8.1 b]
##STR00249##
[0701] To 2.1e (420 mg, 1.260 mmol), crude 8.1a (1.513 g, 6.30 mmol) from above was added. Acetonitrile (Volume: 5 mL) and zinc iodide (402 mg, 1.260 mmol) was then added and the crude refluxed overnight at 50-60.degree. C. The crude was purified by silica gel chromatography using 0 to 100% heptane-(EtOAc:EtOH=75:25) and the desired fractions were concentrated to give 210 mg of the desired product 8.1b in 34% yield. LC-MS (m/z): 490.4 [M+H].sup.+, 1.00 min.
Step 3: Ethyl 6-(tert-butyl)-1-fluoro-10-hydroxy-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,4]- pyrazino[1,2-b]indazole-3-carboxylate [8.1c]
##STR00250##
[0703] To 8.1b (210 mg, 0.429 mmol) in Ethanol (Volume: 20 mL), nitrogen was purged followed by addition of 10% Pd--C(210 mg, 1.973 mmol). Hydrogen gas was then bubbled through the solution with venting followed by attaching a balloon of hydrogen and the reaction stirred for 2 hrs. The crude was filtered on celite and concentrated to yield crude 8.1c which was taken to the next step without further purification, assume quantitative yield. LC-MS (m/z): 400.4 [M+H].sup.+, 0.67 min.
Step 4: Ethyl 6-(tert-butyl)-1-fluoro-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [8.1d-I] and [8.1 d-II]
##STR00251##
[0705] To 8.1c (103 mg, 0.258 mmol) in DMF (Volume: 1 mL), 1-bromo-3-methoxypropane (0.056 mL, 0.516 mmol) and cesium carbonate (252 mg, 0.774 mmol) were added and stirred. The crude was purified by silica gel chromatography using 0-100% Heptane-(EtOAc/EtOH:75/25). The desired fractions were concentrated to give 81 mg of the desired racemic product, 8.1d in 66% yield. LC-MS (m/z): 472.5 [M+H].sup.+, 0.83 min. The above racemic material (81 mg) was separated by chiral chromatography using (AD column, SFC=100 ml/min, CO.sub.2/EtOH=70/30, 237 bar) to give products 8.1d-I (peak 1, tR 2.77 min.) at 24 mg and product 8.1d-II (peak 2, tR 5.52 min.) at 24 mg.
Step 5: 6-(tert-butyl)-1-fluoro-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H- -pyrido[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [8.1-I] and [8.1-II]
##STR00252##
[0707] To 8.1d-I (24 mg, 0.051 mmol) was added Acetonitrile (Volume: 3 ml, Ratio: 1.000), Water (Volume: 3 ml, Ratio: 1.000) and then LiOH (6.41 mg, 0.153 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 11 mg of the desired product 8.1-I as free base in 46.3% yield. LC-MS (m/z): 444.4 [M+H].sup.+, 0.87 min. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.58 (s, 1H), 7.55 (dd, J=4.77, 8.63 Hz, 1H), 7.18-7.34 (m, 1H), 6.76 (d, J=7.58 Hz, 1H), 5.26 (d, J=14.87 Hz, 1H), 4.95 (dd, J=5.14, 14.87 Hz, 1H), 4.25-4.42 (m, 3H), 3.63 (t, J=5.99 Hz, 2H), 3.38 (s, 3H), 2.25 (quin, J=6.27 Hz, 2H), 0.78-0.85 (m, 9H). To 8.1d-II (24 mg, 0.051 mmol) was added Acetonitrile (Volume: 3 ml, Ratio: 1.000), Water (Volume: 3 ml, Ratio: 1.000) and then LiOH (6.41 mg, 0.153 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 20 ml of water acidified with 1M HCl to pH of 2-3, then 100 ml of DCM with 5% ethanol was added and 15 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again, 50 ml of DCM with 5% ethanol. The organic layers were combined, washed with water, saturated sodium chloride solution, dried sodium sulfate, filtered through sodium sulfate plug 1 cm.times.1.5 cm and concentrated to residue. The residue was dissolved in 1:1 ACN/water and lyophilized to give 11 mg of the desired product 8.1-II as free base in 46.3% yield. LC-MS (m/z): 444.4 [M+H].sup.+, 0.87 min. .sup.1H NMR (400 MHz, CDCl.sub.3) .delta. 8.59 (s, 1H), 7.55 (dd, J=4.77, 8.68 Hz, 1H), 7.18-7.34 (m, 1H), 6.76 (d, J=7.58 Hz, 1H), 5.25 (br d, J=14.77 Hz, 1H), 4.95 (br dd, J=3.99, 14.75 Hz, 1H), 4.27-4.40 (m, 3H), 3.63 (t, J=5.99 Hz, 2H), 3.38 (s, 3H), 2.25 (quin, J=6.27 Hz, 2H), 0.81 (s, 9H).
Example 9.1
(R)-6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1'- :3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [9.1]
##STR00253##
[0708] Step 1: (R)-methyl 7-(benzyloxy)-2-(2-((tert-butoxycarbonyl)amino)-3,3-dimethylbutyl)-2H-ind- azole-3-carboxylate [9.1a]
##STR00254##
[0710] To methyl 7-(benzyloxy)-2H-indazole-3-carboxylate (18.5 g, 65.5 mmol) was added Triphenylphospine (20.63 g, 79 mmol), (R)-tert-butyl (1-hydroxy-3,3-dimethylbutan-2-yl)carbamate (17.80 g, 82 mmol) and dissolved in THF (Volume: 185 mL). The reaction was placed in a water bath and (E)-di-tert-butyl diazene-1,2-dicarboxylate (18.11 g, 79 mmol) was added portion wise to keep the internal temperature below 22.degree. C. The mixture was stirred at room temperature for 3 hours or until done by LCMS. The reaction was concentrated to residue. The crude residue was dissolved in ethyl acetate with 20% heptane, washed with 0.5M HCl, water, sodium bicarbonate, water, saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude product was purified by silica gel chromatography using 0-25% EtOAc in heptanes with the desired regio-isomer eluting first on silica gel. The desired fractions were concentrated to constant mass to give 27.3 grams of the desired product 9.1a in 87% yield. LC-MS (m/z): 482.4 [M+H].sup.+, 1.15 min.
Step 2: (R)-methyl 2-(2-((tert-butoxycarbonyl)amino)-3,3-dimethylbutyl)-7-hydroxy-2H-indazol- e-3-carboxylate [9.1b]
##STR00255##
[0712] To 9.1a (27.3 g, 56.7 mmol) was added THF (Volume: 350 mL) and flushed with nitrogen. Then Pd--C(3.02 g, 2.83 mmol) was added carefully. Then to the reaction was added a hydrogen balloon. The reaction was evacuated and refilled 7.times. with hydrogen and then stirred at room temperature for 90 min, or until done by LCMS. To the reaction was added 100 ml of DCM, flushed with nitrogen, filtered through celite plug and flushed with THF. The solvent was concentrated off to constant mass to give the desired product 9.1b used as is for the next step, assume in quantitative yield. LC-MS (m/z): 392.3 [M+H].sup.+, 0.91 min.
Step 3: (R)-methyl 2-(2-((tert-butoxycarbonyl)amino)-3,3-dimethylbutyl)-7-(3-methoxypropoxy)- -2H-indazole-3-carboxylate [9.1c]
##STR00256##
[0714] To 9.1b (22.2 g, 56.7 mmol) was added DMF (Volume: 3 mL) and cesium carbonate (37.0 g, 113 mmol). The reaction was stirred at room temperature for 5 minutes then 1-bromo-3-methoxypropane (13.02 g, 85 mmol) was added and the reaction was stirred at room temperature for 3 hours or until done by LCMS. To the reaction was added 800 ml of ethyl acetate with 20% heptane, washed with water 3.times., saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography using 0-45% ethyl acetate and heptane. The desired fractions were concentrated to constant mass to give 23.2 grams of the desired product 9.1c as a free base in 88% yield. LC-MS (m/z): 464.4 [M+H].sup.+, 1.03 min.
Step 4: (R)-tert-butyl (1-(3-(hydroxymethyl)-7-(3-methoxypropoxy)-2H-indazol-2-yl)-3,3-dimethylb- utan-2-yl)carbamate [9.1d]
##STR00257##
[0716] To 9.1c (23.2 g, 50.0 mmol) was added THF (Volume: 140 mL) and then sodium borohydride (5.68 g, 150 mmol). The reaction was heated to 60-65.degree. C. with stirring. Then methanol was added slowly over 30-35 minutes and the reaction was stirred at 70-75.degree. C. for 3 hours or until done by LCMS. The reaction was cooled to room temperature and placed in water bath for cooling. Then carefully add 140 ml of saturated ammonium chloride solution and then stirred for 45 minutes at room temperature. Then 750 ml of ethyl acetate was added and 70 ml of water, extracted, the organic layer was washed with sat. bicarbonate solution, water, saturated salt solution, dried with sodium sulfate, filtered and concentrated to constant mass to give 21.4 grams of the desired product 9.1d as a free base in 98% yield, used as is. LC-MS (m/z): 436.4 [M+H].sup.+, 0.83 min.
Step 5: (R)-tert-butyl (1-(3-formyl-7-(3-methoxypropoxy)-2H-indazol-2-yl)-3,3-dimethylbutan-2-yl- )carbamate [9.1e]
##STR00258##
[0718] To 9.1d (21.4 g, 49.1 mmol) was added DCM (Volume: 150 mL) and then iodobenzene diacetate (22.16 g, 68.8 mmol). Then TEMPO (0.768 g, 4.91 mmol) was added and the reaction was stirred at room temperature for 3 hour or until done by LCMS. Then 700 ml of ethyl acetate was added, washed with sodium thiosulfate, sodium carbonate solution, water, saturated salt solution, dried sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography using 0-50% ethyl acetate and heptane. The desired fractions were concentrated to constant mass to give 14.55 grams of the desired product 9.1e as a free base in 68% yield, used as is. LC-MS (m/z): 434.4 [M+H].sup.+, 0.96 min.
Step 6: (R)-3-(tert-butyl)-7-(3-methoxypropoxy)-3,4-dihydropyrazino[1,2-b]- indazole [9.1f]
##STR00259##
[0720] To 9.1e (14.55 g, 33.6 mmol) was added DCM (Volume: 180 mL) and then TFA (38.8 mL, 503 mmol). The reaction was stirred at room temperature for 1 hour or until done by LCMS. The reaction was concentrated to residue to give the desired product 9.1f assume in quantitative yield, used as is. LC-MS (m/z): 316.3 [M+H].sup.+, 0.66 min.
Step 7: (6R)-ethyl 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-2,6,7,13c-tetrahydro-1H-pyrido- [2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylate [9.1 g]
##STR00260##
[0722] To 9.1f (10.59 g, 33.6 mmol) was added Ethanol (Volume: 110 mL, Ratio: 10.00), Water (Volume: 11 mL, Ratio: 1.000) and (Z)-ethyl 2-(ethoxymethylene)-3-oxobutanoate (21.88 g, 118 mmol). The reaction was then stirred at 90-95.degree. C. for 16 hours or until done by LCMS. The reaction was concentrated to residue to give the desired product 9.1g assume in quantitative yield, used as is. LC-MS (m/z): 456.3 [M+H].sup.+, 0.78 min.
Step 8: (R)-ethyl 6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyrido[2',1':3,- 4]pyrazino[1,2-b]indazole-3-carboxylate [9.1 h]
##STR00261##
[0724] To 9.1g (15.3 g, 33.6 mmol) was added DME (Volume: 190 mL), and then iodine (36.7 g, 144 mmol). The reaction was stirred at 35-40.degree. C. for 5 hours or until done by LCMS. The reaction was diluted with 800 ml of DCM, washed with sodium bisulfite solution 2.times., water, sodium carbonate solution, water, saturated salt solution, dried with sodium sulfate, filtered and concentrated to residue. The crude material was purified by silica gel chromatography using 0-95% (ethyl acetate with 25% ethanol) and heptane. The desired fractions were concentrated to constant mass to give the desired crude product. To the residue was added 100 ml of ethyl ether with 1% ethanol and heated to reflux with stirring for 5 minutes, then was cooled to room temperature for 1 hour. The solid was collected by filtration, washed with ethyl ether and dried to constant mass. The crude product needed additional purification by silica gel chromatography using 0-60% (DCM with 10% ethanol) and DCM, the desired fractions were concentrated to constant mass to give 7.71 grams of the desired product 9.1h as free base in 51% yield, used as is. LC-MS (m/z): 454.3 [M+H].sup.+, 0.80 min. .sup.1H NMR (400 MHz, DMSO-d6) .delta. ppm 8.52 (s, 1H) 7.50 (d, J=8.51 Hz, 1H) 7.17 (t, J=8.00 Hz, 1H) 6.72-6.85 (m, 2H) 5.06-5.12 (m, 1H) 4.98-5.05 (m, 1H) 4.69 (d, J=4.50 Hz, 1H) 4.14-4.28 (m, 4H) 3.51 (t, J=6.28 Hz, 2H) 3.25 (s, 3H) 2.04 (quin, J=6.36 Hz, 2H) 1.26 (t, J=7.09 Hz, 3H) 0.68 (s, 9H).
Step 9: (R)-6-(tert-butyl)-10-(3-methoxypropoxy)-2-oxo-6,7-dihydro-2H-pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-3-carboxylic acid [9.1]
##STR00262##
[0726] To 9.1h (7.71 g, 17.00 mmol) was added Acetonitrile (Volume: 120 ml, Ratio: 1.000), Water (Volume: 120 ml, Ratio: 1.000) and then LiOH (51.0 ml, 51.0 mmol). The reaction was stirred at room temperature for 2 hours or until done by LCMS. The reaction was diluted with 400 ml of water acidified with 1M HCl to pH of 2-3, then 800 ml of DCM with 3% ethanol was added and 200 ml of saturated sodium chloride solution. The organic layer was separated, the aqueous layer was extracted again 1.times.150 ml DCM with 2% ethanol. The organic layers were combined, washed with water 4.times.150 ml, filtered and concentrated to constant mass to give 7.1 grams of the desired product 9.1 as free base in 99% yield. The enantiomeric excess (e.e.) was determine by chiral chromatography using AD column, SFC=5 mL/min, CO.sub.2/EtOH=75/25, (with an enantiomer mixture separating at Rt 1.53 min. and Rt 2.21 min.) giving the desired product 9.1 at Rt 2.20 min., in 99% e.e. LC-MS (m/z): 426.3 [M+H].sup.+, 0.81 min. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. ppm 8.95 (s, 1H) 7.63 (d, J=8.46 Hz, 1H) 7.15-7.32 (m, 2H) 6.82 (d, J=7.58 Hz, 1H) 5.14-5.24 (m, 1H) 5.04-5.13 (m, 1H) 4.95 (d, J=4.70 Hz, 1H) 4.20 (t, J=6.41 Hz, 2H) 3.51 (t, J=6.26 Hz, 2H) 3.25 (s, 3H) 2.01-2.08 (m, 2H) 0.69 (s, 9H). The product 9.1 can be converted to a tris(hydroxymethyl)aminomethane (TRIS) salt by the following procedure. To 9.1 (11.05 g, 26.0 mmol) was added Acetonitrile (Volume: 185 ml, Ratio: 9.25) stirred at 50.degree. C. for 1 hour, until full dissolved. Then 2-amino-2-(hydroxymethyl)propane-1,3-diol (3.15 g, 26.0 mmol) was added and stirred for 3 hours at 50.degree. C. After 1 hour, additional Acetonitrile (Volume: 20 ml, Ratio: 1.000) was added to help stirring and continued at 50.degree. C. for a full 3 hours. Then the solution was cooled to room temperature and stirred for 18 hours at room temperature. The solid was collected by filtration, washed with ACN, dried under vacuum at 40.degree. C. for 24 hours to give 12.82 grams of the desired product 9.1 as the TRIS salt in 89% yield. LC-MS (m/z): 426.3 [M+H].sup.+, 0.80 min. .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. ppm 8.93 (s, 1H), 7.63 (d, J=8.5 Hz, 1H), 7.25 (s, 1H), 7.22 (d, J=8.1 Hz, 1H), 6.82 (d, J=7.6 Hz, 1H), 5.15-5.22 (m, 1H), 5.05-5.13 (m, 1H), 4.94 (d, J=4.7 Hz, 1H), 4.34 (br s, 3H), 4.20 (t, J=6.4 Hz, 2H), 3.51 (t, J=6.3 Hz, 2H), 3.25 (s, 3H), 3.21 (s, 6H), 2.04 (quin, J=6.3 Hz, 2H), 0.69 (s, 9H).
Example 10.1
12-methoxy-3,3-dimethyl-7-oxo-1,3,3a,14a-tetrahydro-7H-furo[3',4':5,6]pyri- do[2',1':3,4]pyrazino[1,2-b]indazole-6-carboxylic acid [10.1-I] and [10.1-II]
##STR00263##
[0727] Step 1: 1-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2-yl)-3-- hydroxy-3-methylbutan-2-one [10.1a]
##STR00264##
[0729] To a mixture of 1.1b (1.65 g, 5.64 mmol) and cesium carbonate (7.35 g, 22.57 mmol) in DMSO (Volume: 19.89 ml, Ratio: 16) was added water (Volume: 1.243 ml, Ratio: 1.000). The mixture was stirred 10 min, then 1-bromo-3-hydroxy-3-methylbutan-2-one (2.043 g, 11.28 mmol) was added. After 30 min, 100 ml EtOAc was added, and the organics washed with water, brine, dried over Na.sub.2SO.sub.4, filtered, and concentrated. The material was purified on SiO.sub.2 (0-60% EtOAc/Hep) to give after concentration desired product 10.1a (0.8 g, 2.038 mmol, 36.1% yield) from the more polar peak with the correct m/z by LC-MS. LC-MS (m/z): 393.4 [M+H].sup.+, 1.09 min.
Step 2: 4-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2- -yl)-2,2-dimethylfuran-3(2H)-one [10.1b]
##STR00265##
[0731] 10.1a (800 mg, 2.038 mmol) was dissolved in toluene (Volume: 2 mL). Bredereck's reagent (0.514 mL, 2.242 mmol) was added. The flask was fitted with a reflux condenser, and heated to 100.degree. C. At 1 h, the reaction was judged complete by LC-MS. The mixture was cooled to rt and concentrated onto diatomaceous earth. The crude material was purified on a 24 g SiO.sub.2 column (0->50% EtOAc in heptanes) to provide after concentration desired product 10.1b (600 mg, 1.490 mmol, 73.1% yield) as a yellow oil from the major peak. LC-MS (m/z): 403.5 [M+H].sup.+, 1.28 min.
Step 3: 4-(3-(((tert-butyldimethylsilyl)oxy)methyl)-7-methoxy-2H-indazol-2- -yl)-2,2-dimethyldihydrofuran-3(2H)-one [10.1c]
##STR00266##
[0733] A 4 mL vial was charged with 10.1b (600 mg, 1.49 mmol) and THF (Volume: 7450 .mu.l). The vial was cooled to -10.degree. C. in an ice/brine bath. L-Selectride (1639 .mu.l, 1.639 mmol) was added dropwise. The reaction was deemed complete by LC-MS at 20 min and quenched with ammonium chloride, extracted with EtOAc, dried over Na.sub.2SO.sub.4, filtered and concentrated. The crude material was purified on SiO.sub.2 (0-60% EtOAc/Hep) to provide after concentration desired product 10.1c (500 mg, 1.236 mmol, 83% yield) as an oil. LC-MS (m/z): 405.4 [M+H].sup.+, 1.21 min.
Step 4: 9-methoxy-3,3-dimethyl-1,3,3a,11a-tetrahydrofuro[3',4':5,6]pyrazin- o[1,2-b]indazole [10.1d]
##STR00267##
[0735] To 10.1c (230 mg, 0.568 mmol) in ethanol (Volume: 3 mL), ammonium acetate (657 mg, 8.53 mmol) was added. The vial was sealed and the mixture stirred at 60.degree. C. for 1 h. Sodium cyanoborohydride (35.7 mg, 0.568 mmol) was added and stirring continued for 1 h. Two more portions of sodium cyanoborohydride were added over the next two hours. The crude mixture was diluted with DCM and washed with 6 N NaOH to provide crude amine (420 mg, 50% pure). This material was taken up in THF (6 mL) and TBAF in THF (1.2 mL, 1.2 mmol) was added. At 30 min, the reaction was diluted with 1 N NaOH and extracted with DCM. The combined organics were dried over Na.sub.2SO.sub.4, filtered and concentrated to provide the hydroxyamine as a brown oil. LC-MS (m/z): 292.3 [M+H].sup.+, 0.66 min. The hydroxyamine was taken up in DCM (8.5 mL) and MnO2 (950 mg, 10.8 mmol) was added. The mixture was stirred for 20 h, diluted with DCM, and passed through a plug of diatomaceous earth. The solution was concentrated and the material was purified on 12 g SiO.sub.2 (0->60% EtOAc/heptane). The peak starting at 30% EtOAc was isolated as desired product 10.1d (120 mg, 51% yield) as a white solid. LC-MS (m/z): 272.3 [M+H].sup.+, 0.68 min.
Step 5: ethyl 12-methoxy-3,3-dimethyl-7-oxo-1,3,3a, 14a-tetrahydro-7H-furo[3',4':5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole- -6-carboxylate [10.1e]
##STR00268##
[0737] A 4 mL vial was charged with 10.1d (120 mg, 0.44 mmol) in water (1.8 ml). Ethyl (E)-2-(ethoxymethylene)-3-oxobutanoate (165 mg, 0.89 mmol) was added. The vial was sealed, purged with N2, and heated to 80.degree. C. overnight. The mixture was extracted with DCM, the combined organics dried over Na.sub.2SO.sub.4, filtered and concentrated to provide an oil. The oil was taken up in DME (1.8 mL) and p-chloroanil (217 mg) was added. The mixture was heated at 100.degree. C. for 90 min. After cooling at rt, the solvent was removed under streaming nitrogen to provide the desired product 10.1e as a brown solid that was used without further purification, assume quantitative yield. LC-MS (m/z): 410.5 [M+H].sup.+, 0.77 min.
Step 6: 12-methoxy-3,3-dimethyl-7-oxo-1,3,3a, 14a-tetrahydro-7H-furo[3',4':5,6]pyrido[2',1':3,4]pyrazino[1,2-b]indazole- -6-carboxylic acid [10.1-I] and [10.1-II]
##STR00269##
[0739] To a solution of crude 10.1e (120 mg, 0.29 mmol) in THF (0.9 mL) was added aqueous LiOH (1 mL, 1.0 mmol) and stirred 1 hour. The solution was acidified to pH 1 by adding 4.0 N HCl (aq.) and extracted with DCM. The combined organic layers were dried over Na.sub.2SO.sub.4 and concentrated to provide a brown solid, which was purified first by achiral chromatography (Sunfire column B-9m-1050, 0.1% TFA in H.sub.2O/MeCN, 75 mL/min), to give 59.9 mg of racemic product 10.1 in 51% yield. 382.5 [M+H].sup.+, 0.88 min. The above racemic product was purified by chiral chromatography (AS column, SFC-UV15=5 mL/min, CO.sub.2/EtOH=75/25, crude in MeOH+diethyl amine) to provide 10.1-I (17.7 mg, at 1.79 min retention time) as a white solid and 10.1-II (13.6 mg, at 3.24 min. retention time) as a white solid. .sup.1H NMR (500 MHz, DMSO-d.sub.6) .delta. ppm 8.87 (s, 1H), 7.74 (d, J=8.5 Hz, 1H), 7.38 (s, 1H), 7.32 (dd, J=8.3, 7.6 Hz, 1H), 6.89 (d, J=7.6 Hz, 1H), 5.55 (dd, J=6.9, 3.3 Hz, 1H), 5.50 (d, J=6.6 Hz, 1H), 4.88 (d, J=10.2 Hz, 1H), 4.42 (dd, J=10.9, 3.8 Hz), 3.98 (s, 3H), 1.47 (s, 3H), 0.56 (s, 3H). LC-MS (m/z): 382.5 [M+H].sup.+, 0.88 min.
Biological Examples
HBV Cell Line
[0740] HepG2-Clone42, a Tet-inducible HBV-expressing cell line with a stably integrated 1.3mer copy of the HBV ayw strain, was generated based on the Tet-inducible HepAD38 cell line with slight modifications. Ladner S K, et al., AntimicrobialAgents and Chemotherapy. 41(8):1715-1720 (1997). HepG2-Clone42 cells were cultured in DMEM/F-12+Glutamax.TM. (Life Technologies, Carlsbad, Calif., USA), supplemented with 10% fetal bovine serum (Life Technologies), G-418 (Corning, Manassas, Va., USA) at a final concentration of 0.5 mg/mL, and 5 .mu.g/mL Doxycycline (Sigma, St. Louis, Mo., USA) and maintained in 5% CO2 at 37.degree. C.
HBsAg Assay
[0741] HepG2-Clone42 cells were seeded into black clear-bottom 96-well plates at a concentration of 6.0.times.10.sup.4 cells/well. 24 hours post-seeding, the cells were treated with 200 .mu.l/well of media containing five-fold serial dilutions of compounds in DMSO. DMSO alone was used as the no drug control. The final DMSO concentration in all wells was 0.5%.
[0742] The HBsAg ELISA kit (Alpha Diagnostic International, San Antonio, Tex., USE, Catalog #4110) was used to determine the level (semi-quantitative) of secreted HBV sAg. The HBSAg ELISA assay was performed following the manufacturer's protocol as described.
[0743] Step 1. Pipet 100 .mu.L each of compound or DMSO treated samples into HBsAg ELISA plates. Seal plates and incubate at room temp for 60 minutes.
[0744] Step 2. Aspirate samples and wash three times with Wash Buffer. Dispense 100 .mu.L of antibody-HRP conjugate to each well. Incubate at room temp for 30 minutes.
[0745] Step 3. Aspirate samples and wash three times with Wash Buffer. Add 100 .mu.L of TMB Substrate to all wells and incubate 15 minutes at room temp.
[0746] Step 4. Dispense 100 .mu.L of Stop Solution to each well. Measure absorbance of ELISA plate at 450 nm.
Dose Response Curves
[0747] Dose-response curves were generated and the EC.sub.50 value was defined as the compound concentration at which HBsAg secretion was reduced 50% compared to the DMSO control.
[0748] EC.sub.50 values were determined as follows:
[0749] Determine the percent of HBsAg secretion inhibition. Calculate the percent inhibition on of HBsAg secretion inhibition using the following equation:
100.times.(X.sub.C-M.sub.B)/(M.sub.D-M.sub.B)
[0750] where X.sub.C is the absorbance signal from compound-treated well; M.sub.B is average absorbance signal (background signal) for column 12 (no cells+HBsAg ELISA sample buffer) and M.sub.D is average absorbance signal from DMSO-treated wells. Then calculate EC.sub.50 values by non-linear regression using a four parameter curve logistic equation.
[0751] The curve fit model employed is XLFit Dose Response One Site Model 204: y=(A+((B-A)/(1+(10{circumflex over ( )}((C-x)*D))))) where A is the minimum y value, B is the maximum y value, C is the log EC50 value, and D is the slope factor. In the following table+.gtoreq.100 nM; 100 nM>++.gtoreq.1 nM; 1 nM>+++
TABLE-US-00001 TABLE 1 In Vitro activity of selected compounds of Formula (I). Compound HBsAg (nM) 1.1-I +++ 1.1-II + 1.2-I ++ 1.2-II ++ 1.3-I ++ 1.3-II ++ 1.4-I + 1.4-II ++ 1.5-I ++ 1.5-II + 2.1-I +++ 2.1-II ++ 2.2-I + 2.2-II +++ 2.3 +++ 2.4 + 2.5 ++ 2.6 +++ 2.7 +++ 2.8 +++ 2.9 +++ 2.10 +++ 2.11 +++ 2.12 ++ 2.13 ++ 2.14 +++ 2.15 +++ 2.16 +++ 2.17 +++ 2.18 ++ 2.19 +++ 2.20 ++ 2.21 +++ 2.22 ++ 2.23 ++ 2.24 + 2.25 ++ 2.26 ++ 2.27 + 2.28 + 2.29 +++ 2.30 ++ 2.31 +++ 2.32 + 2.33-I + 2.33-II + 2.34-I + 2.34-II + 2.35-I ++ 2.35-II ++ 2.36 +++ 2.37 ++ 2.38 ++ 2.39 +++ 2.40 +++ 2.41 +++ 2.42 +++ 2.43 + 2.44 ++ 2.45 ++ 2.46 ++ 2.47-I + 2.47-II +++ 3.1-I + 3.1-II ++ 4.1-I +++ 4.1-II + 5.1-I +++ 5.1-II + 5.2-I ++ 5.2-II + 6.1-I + 6.1-II +++ 6.2-I ++ 6.2-II +++ 7.1-I + 7.1-II +++ 8.1-I + 8.1-II ++ 9.1 +++ 10.1-I + 10.1-II ++ 2.1i ++
* * * * *
References












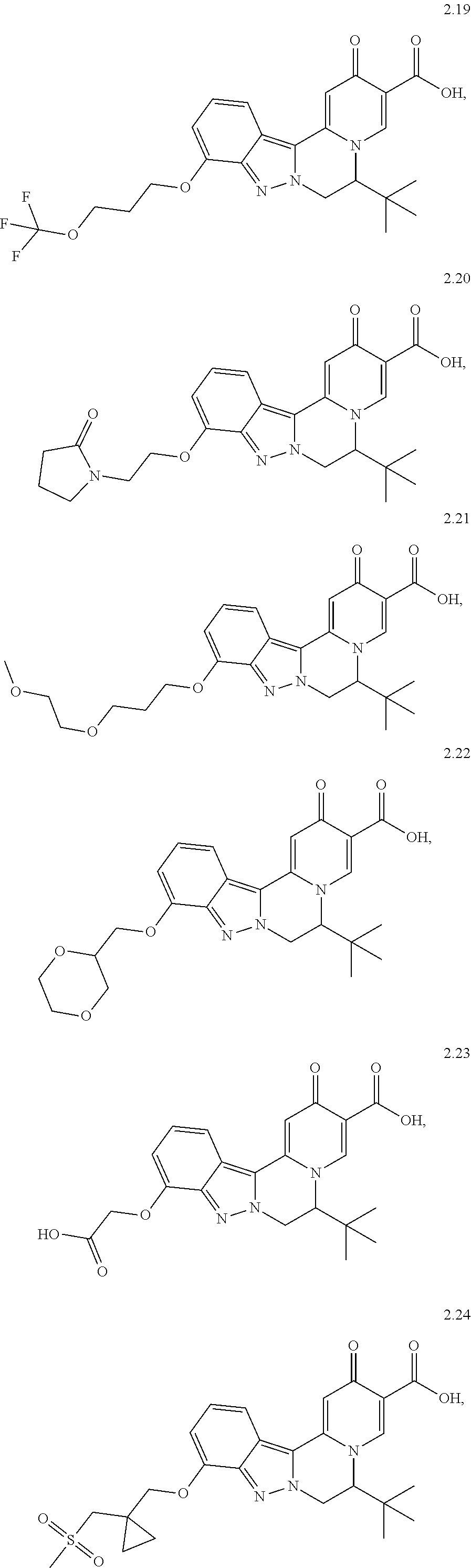

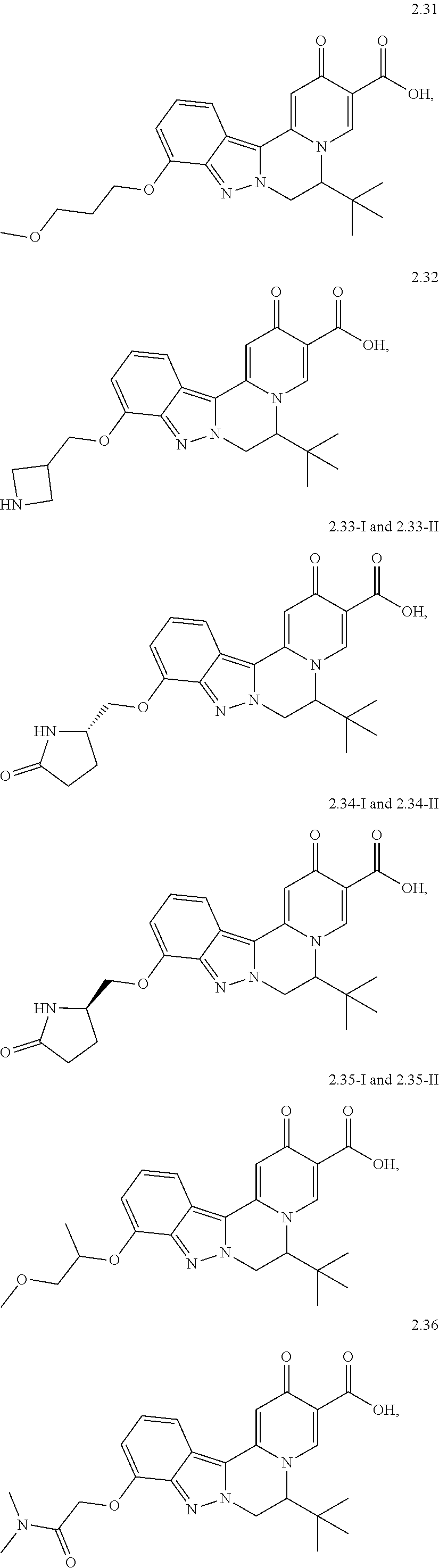
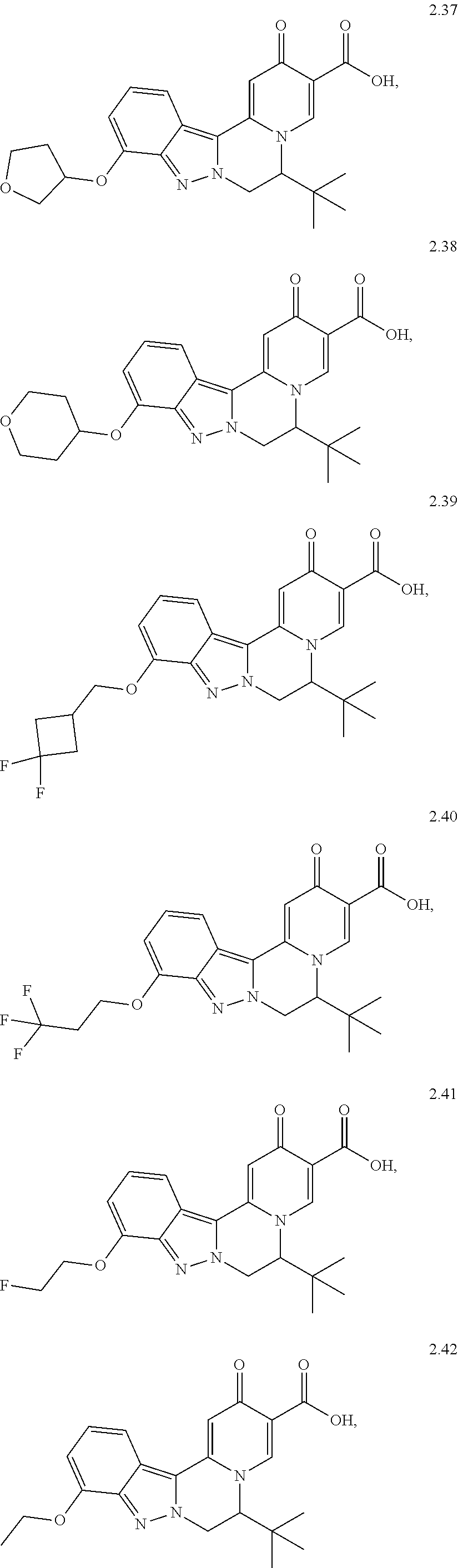
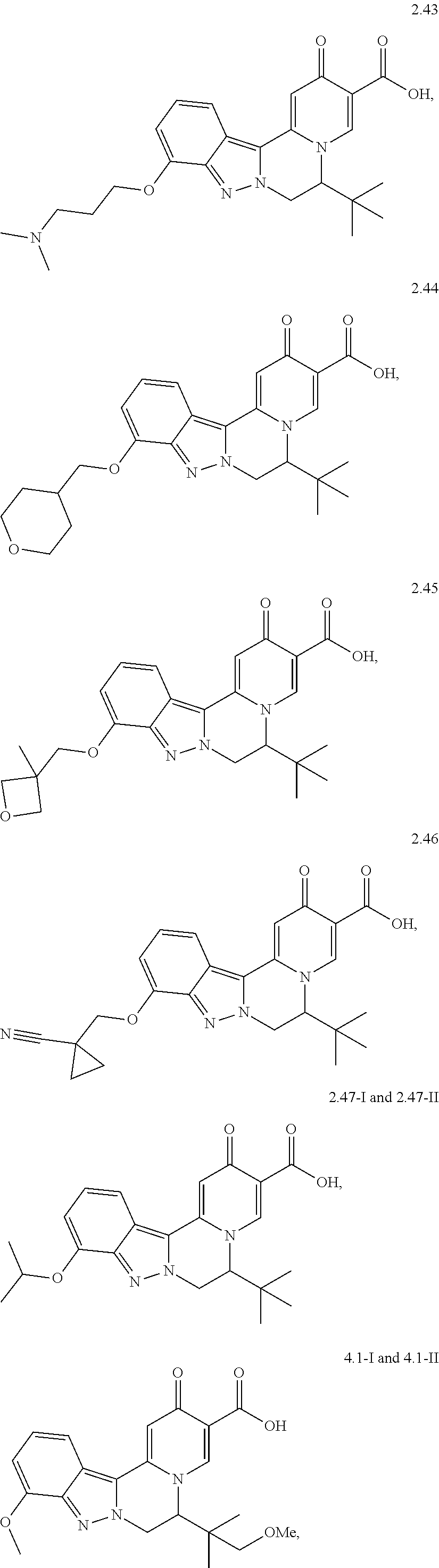
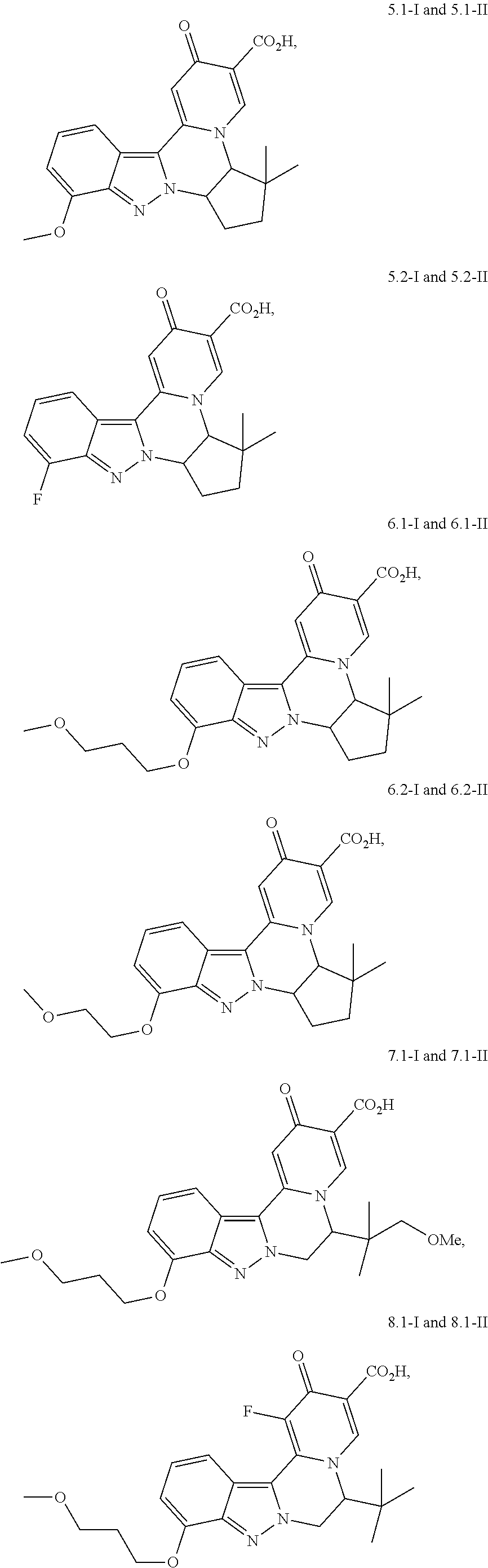
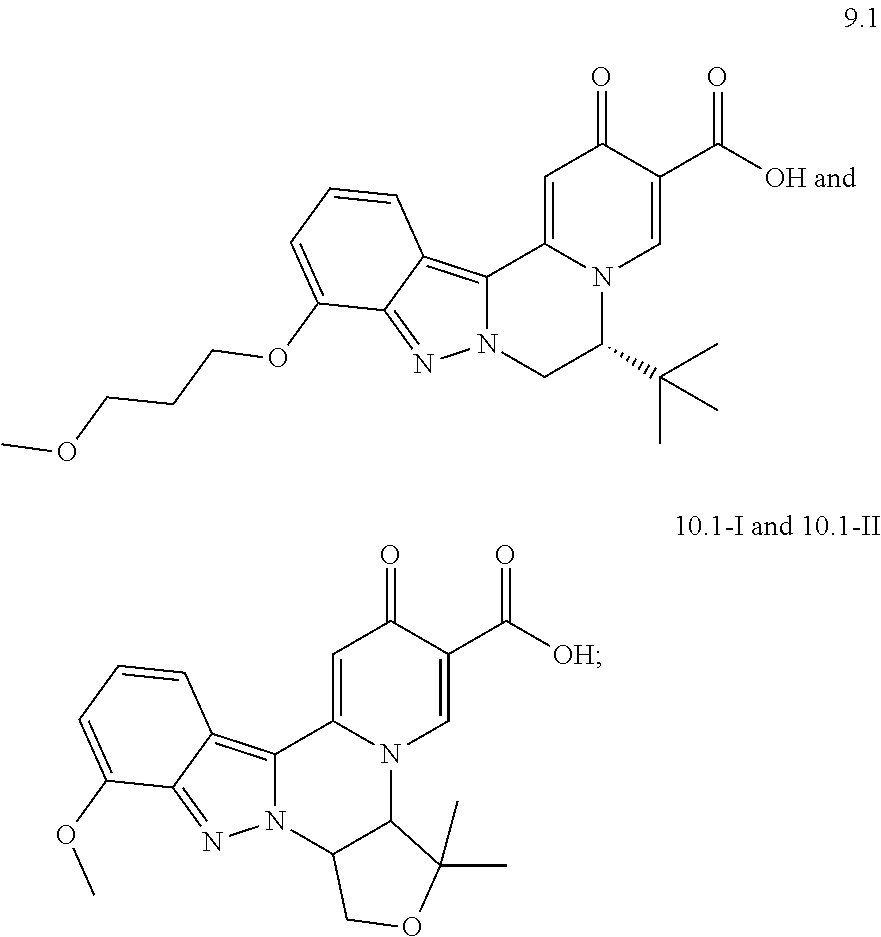
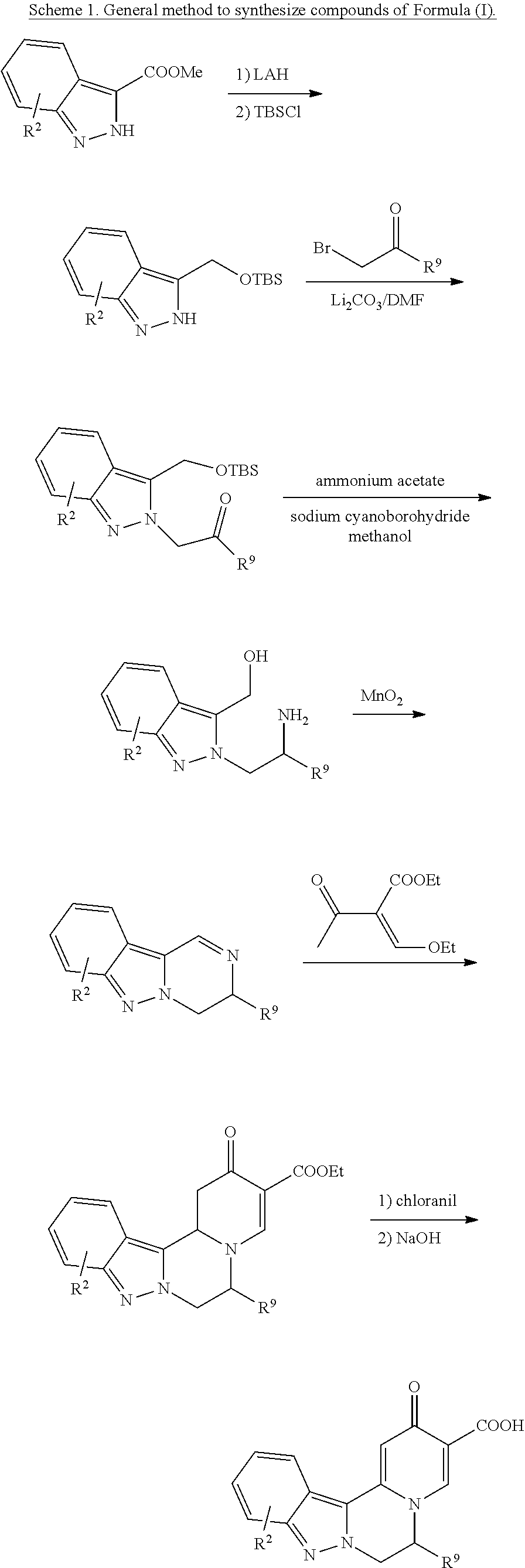
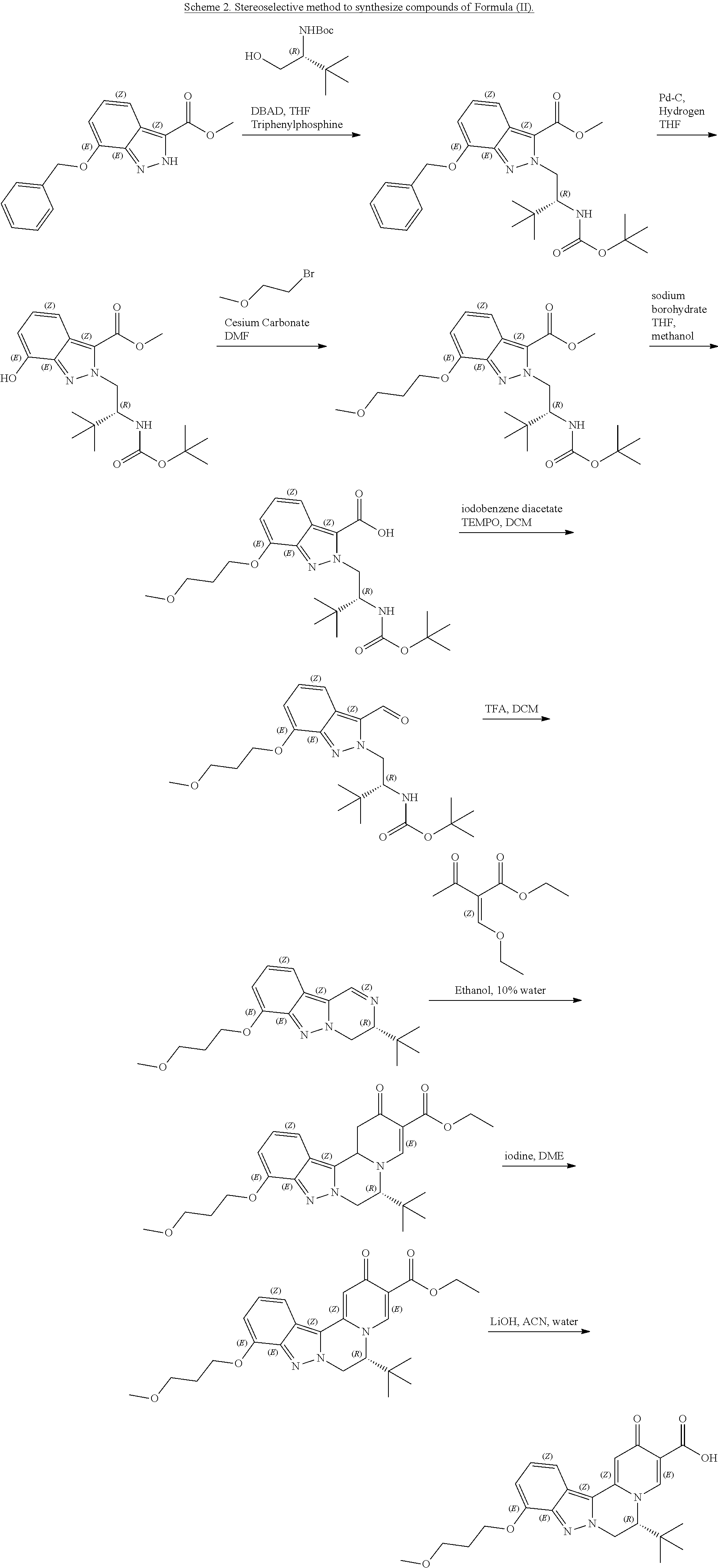
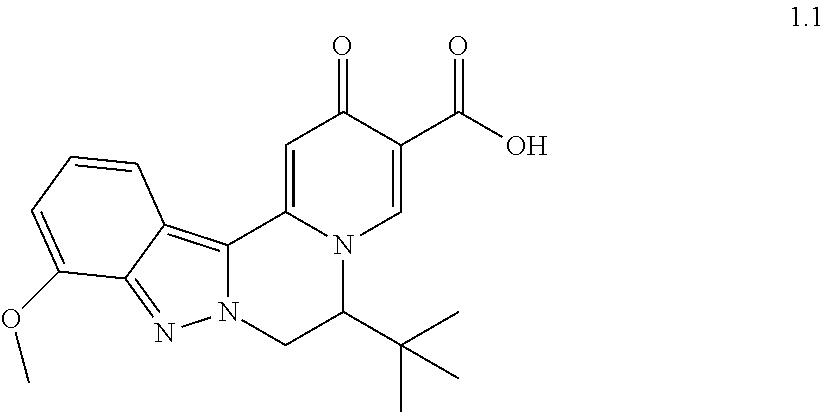
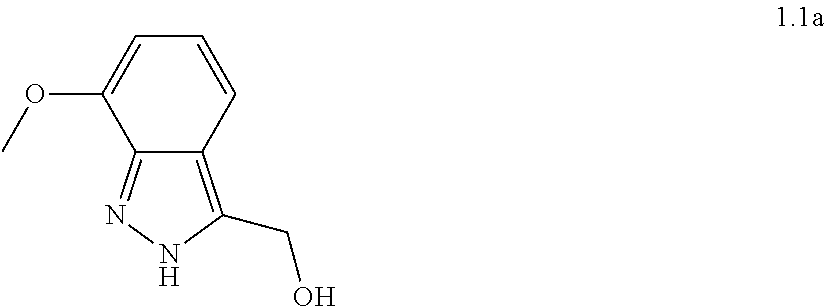
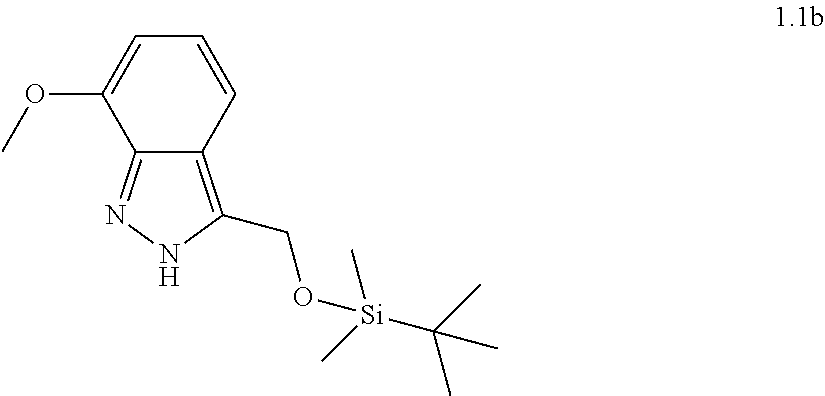

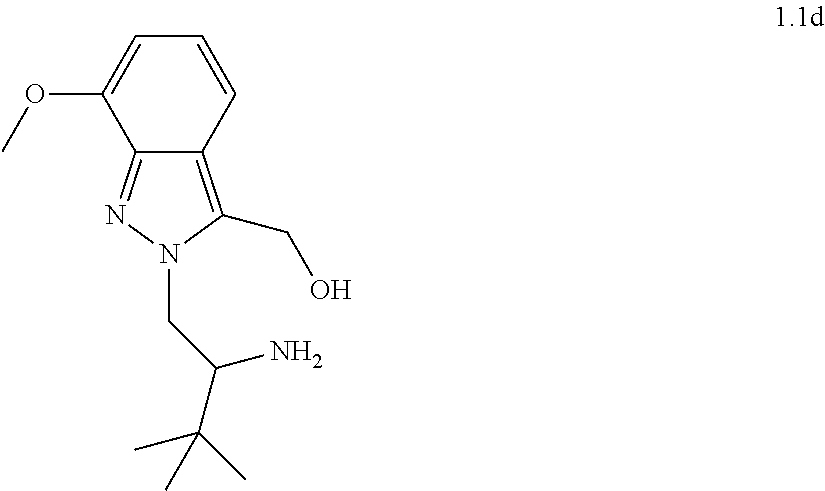
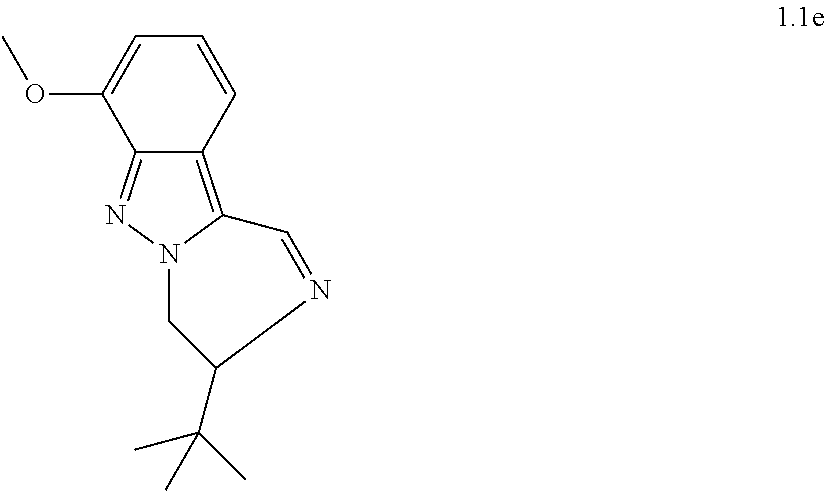
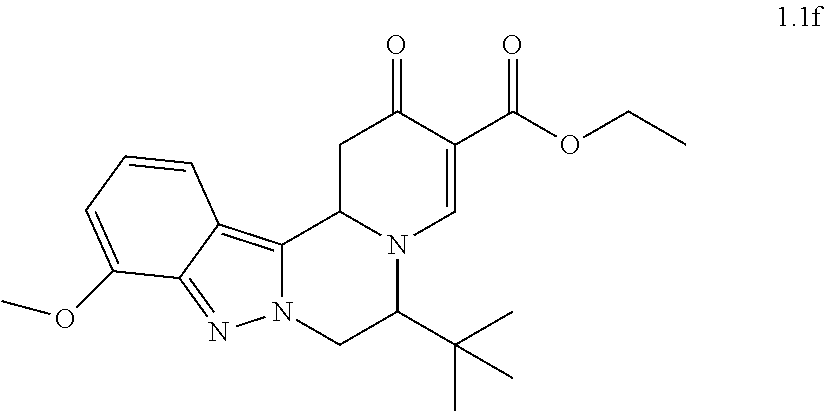
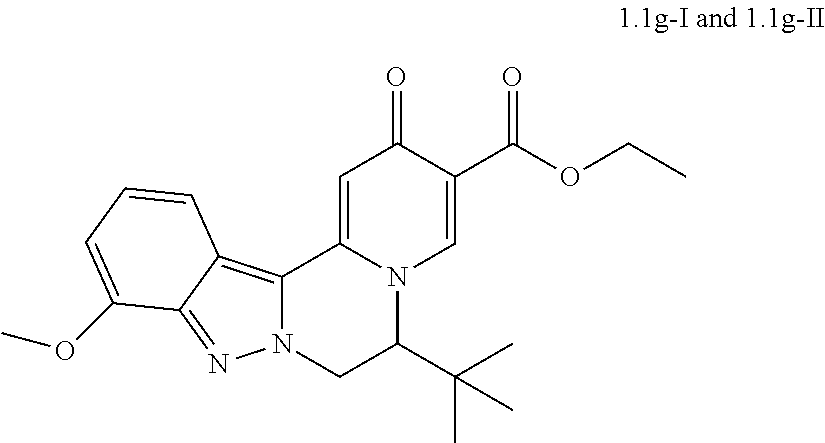
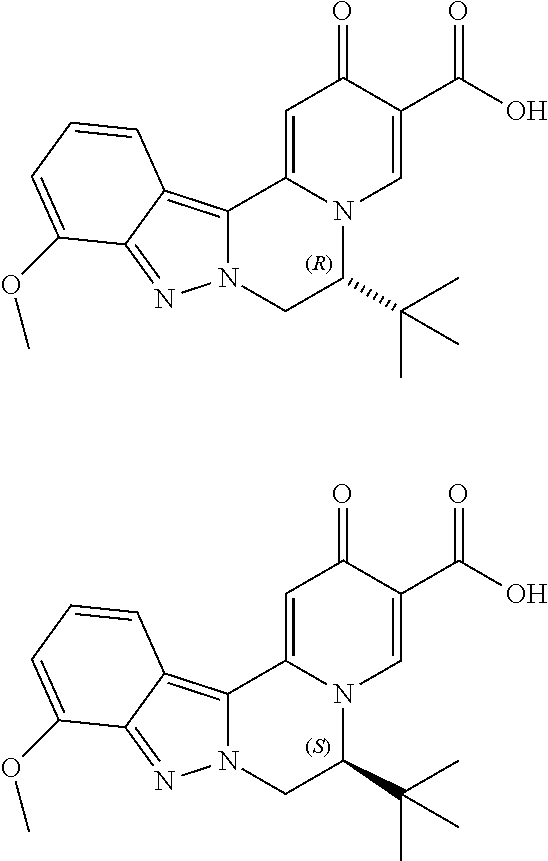
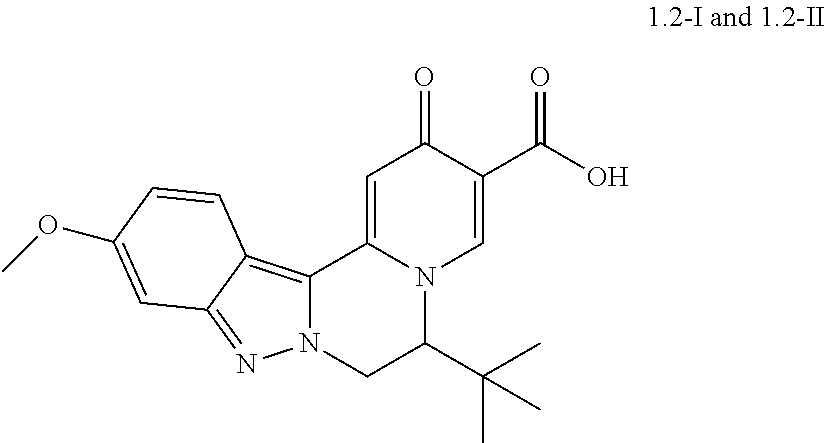
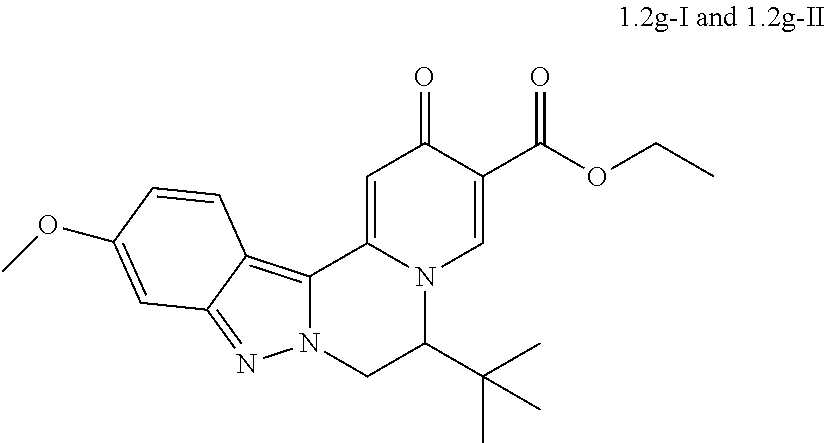
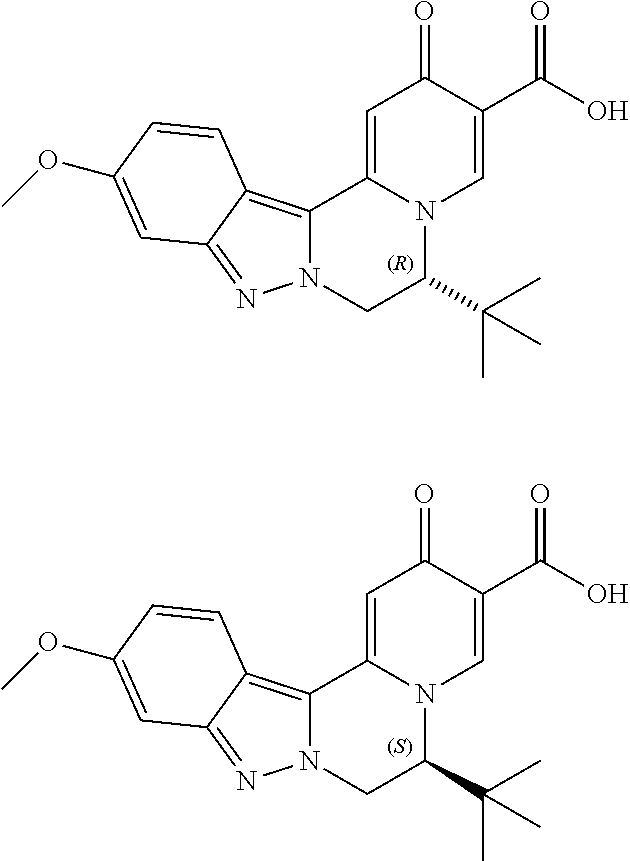
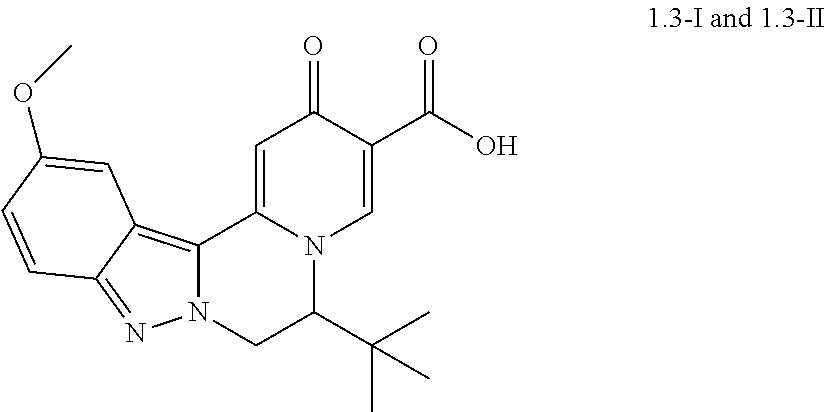
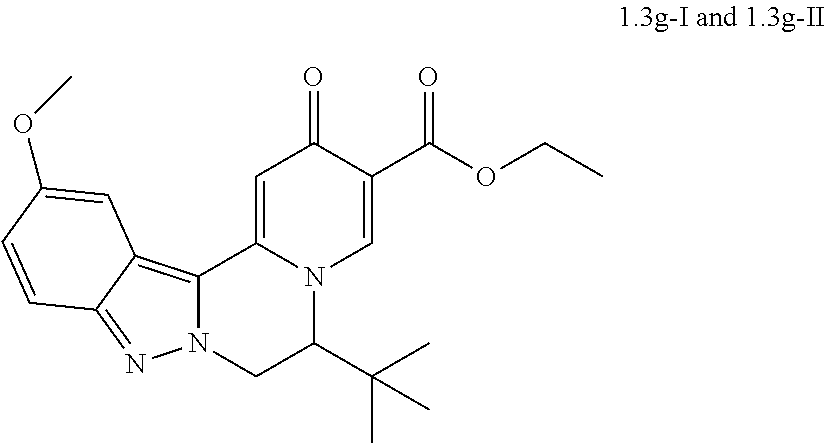
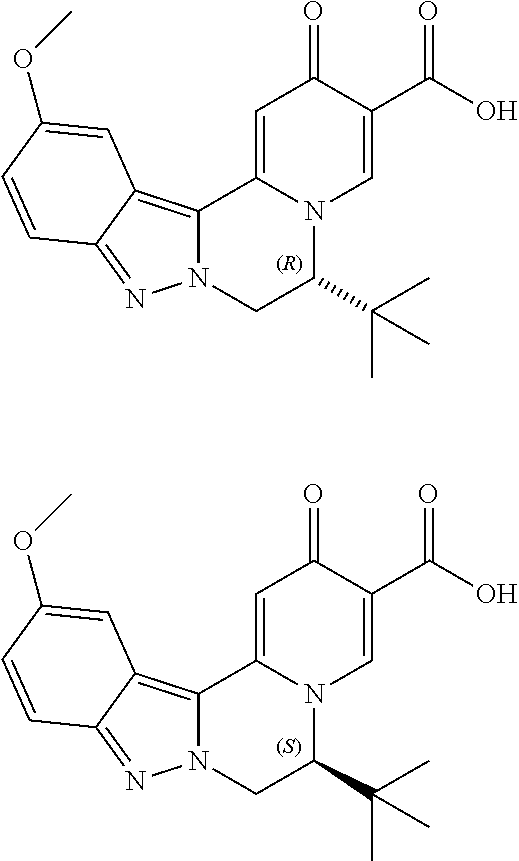
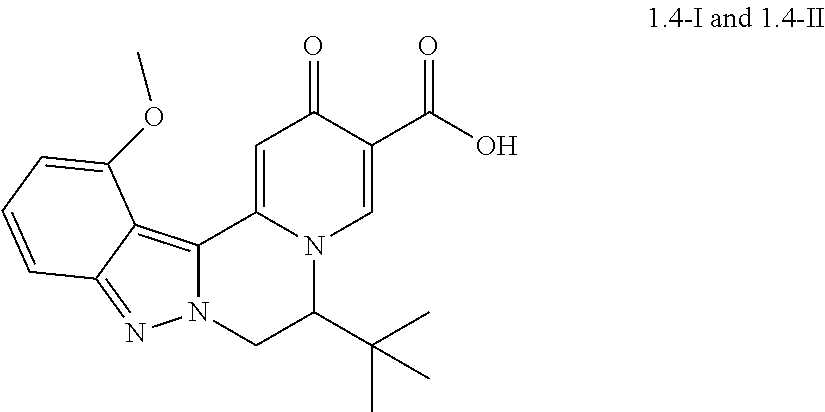

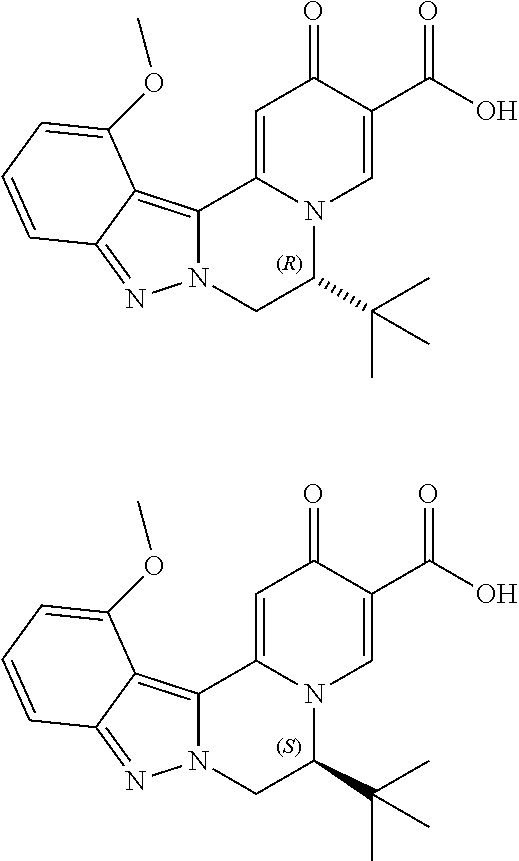
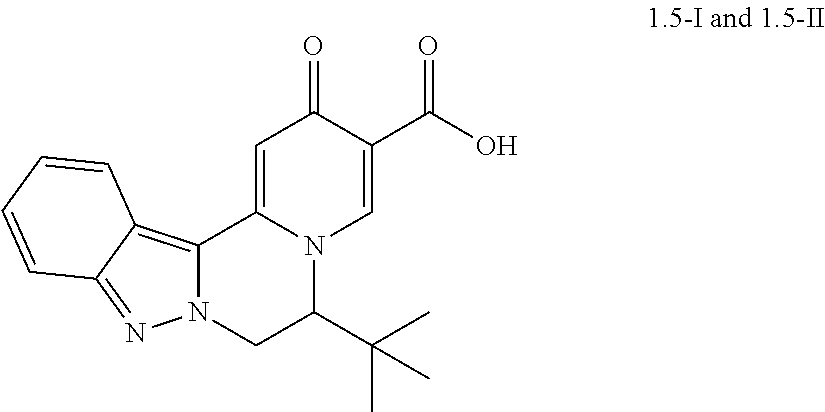
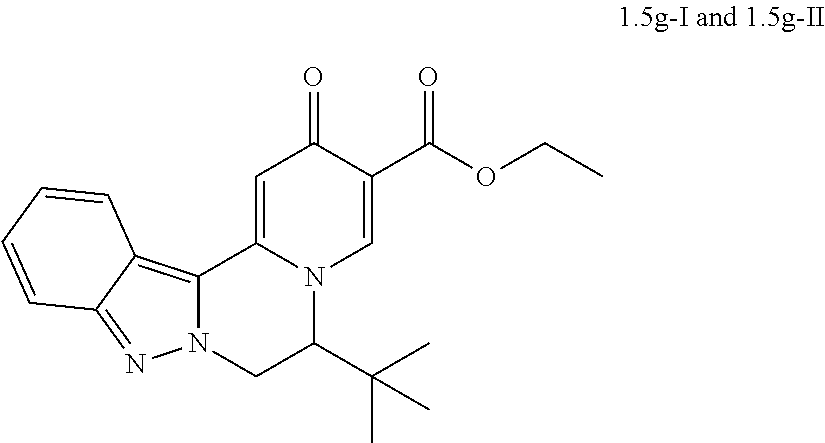
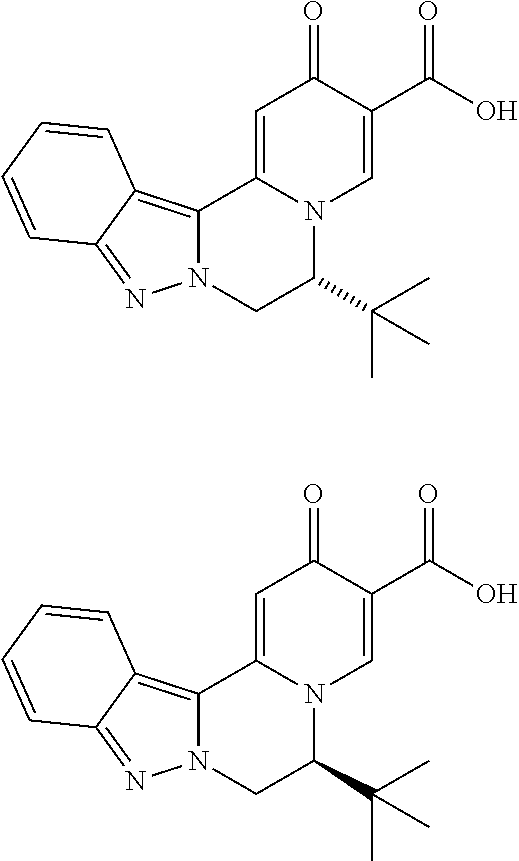
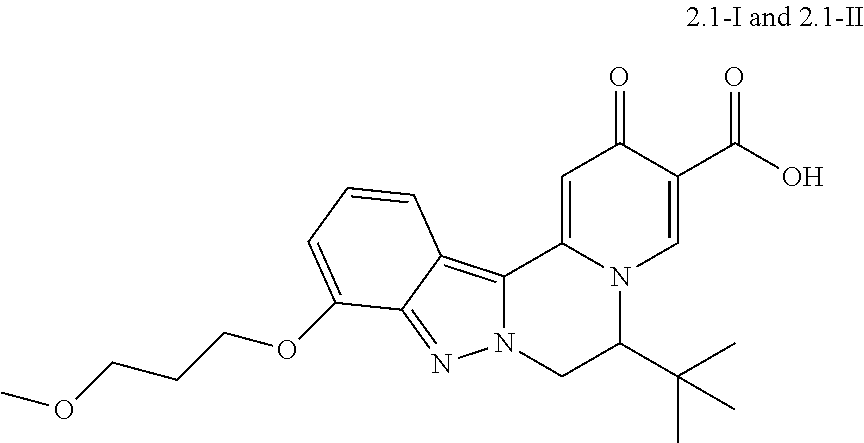
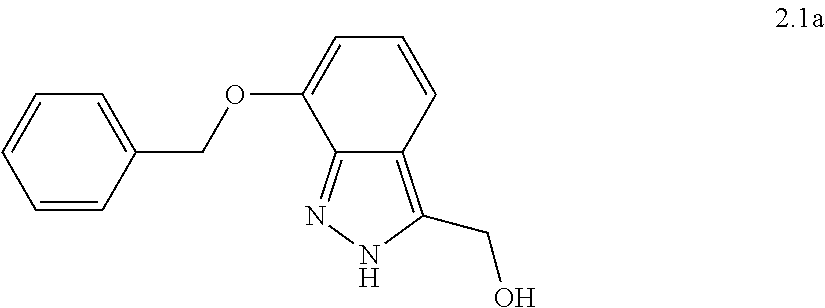
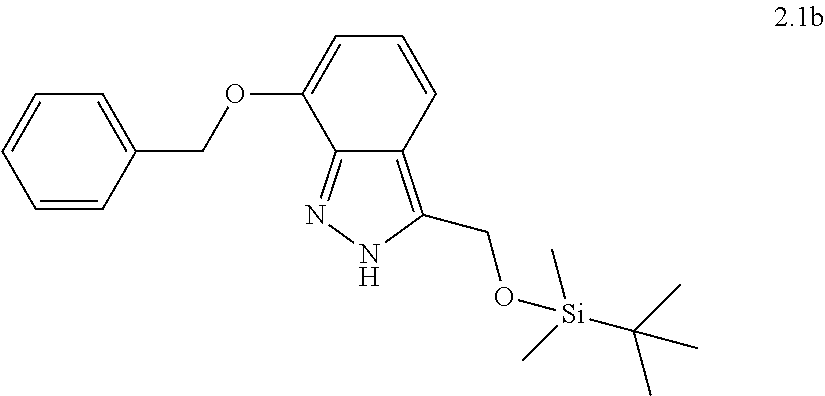
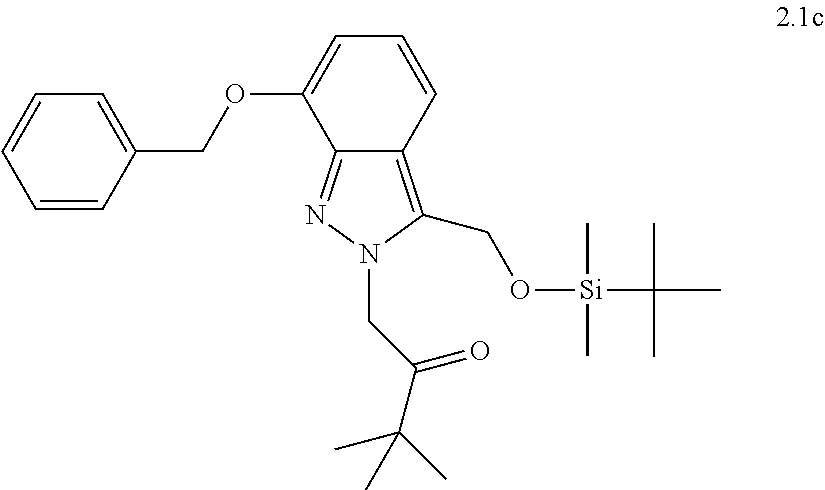
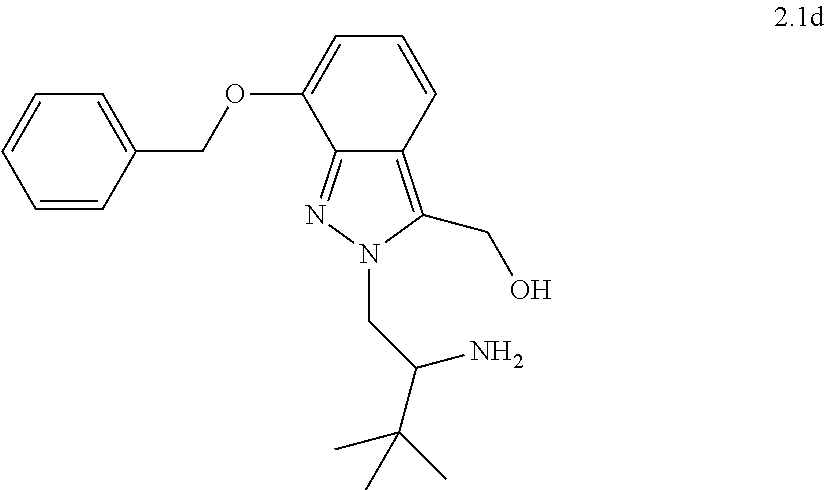
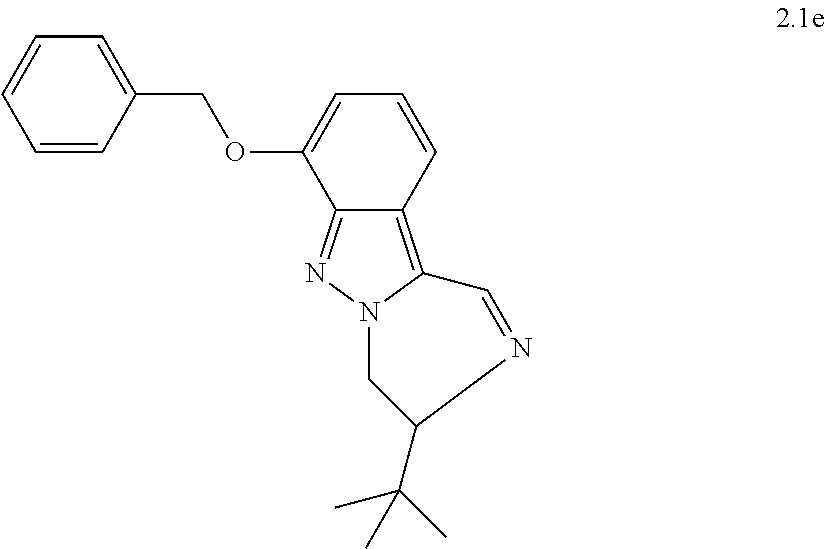
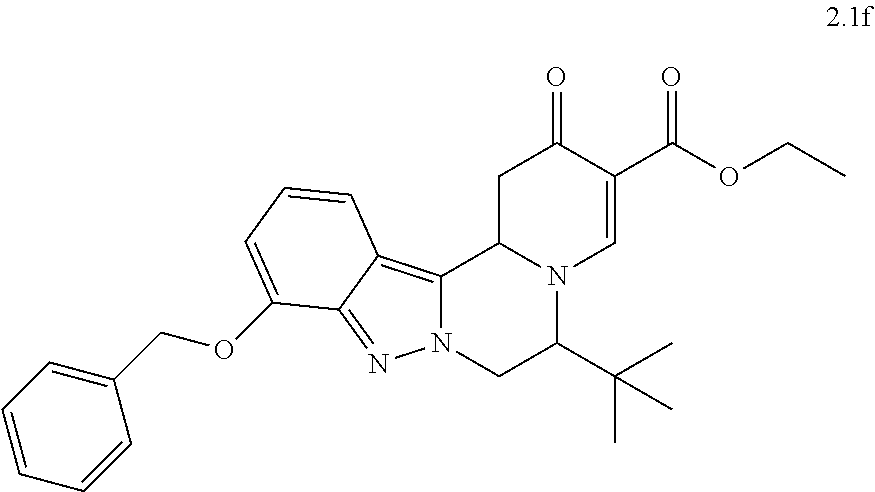
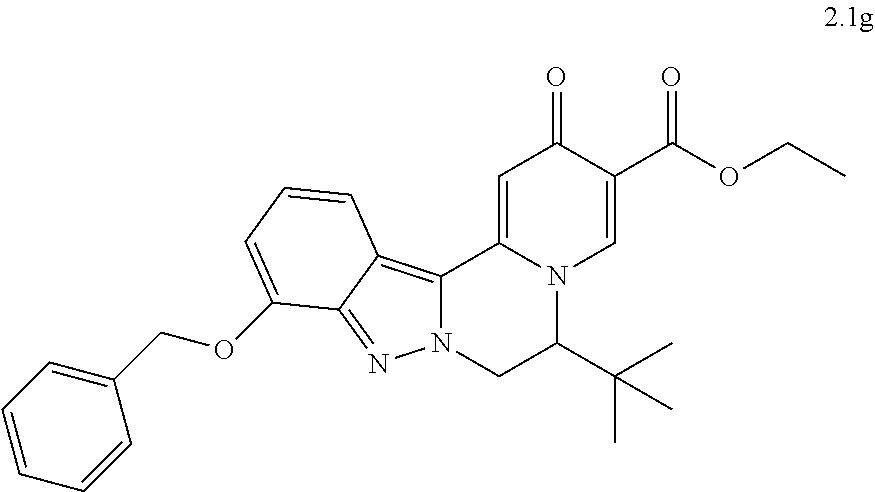
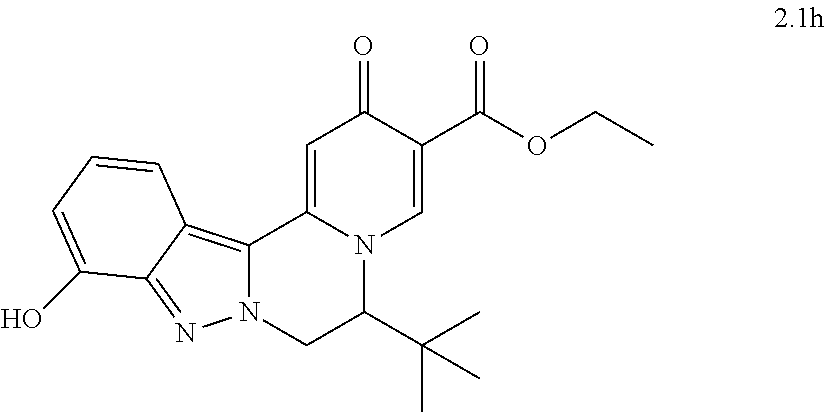
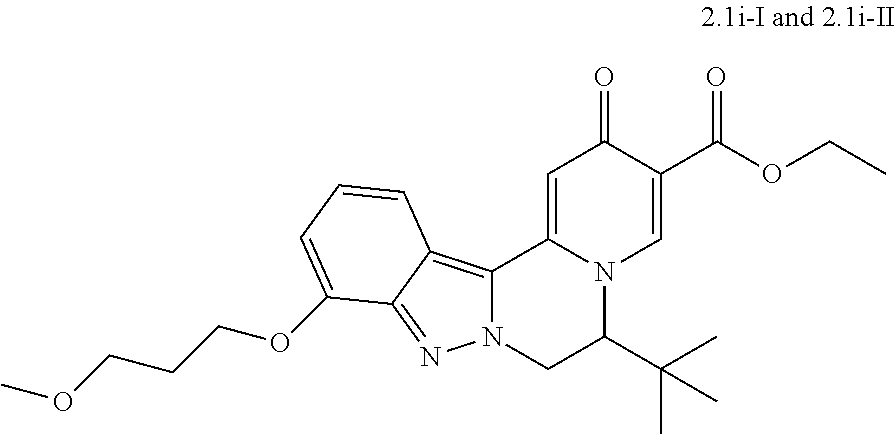
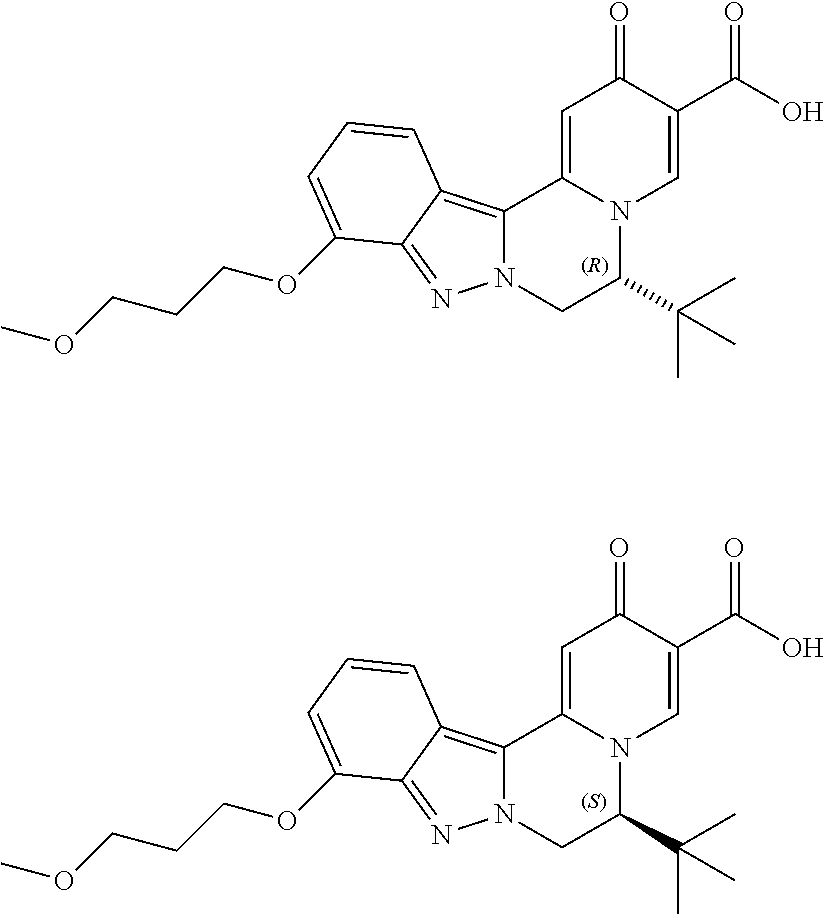
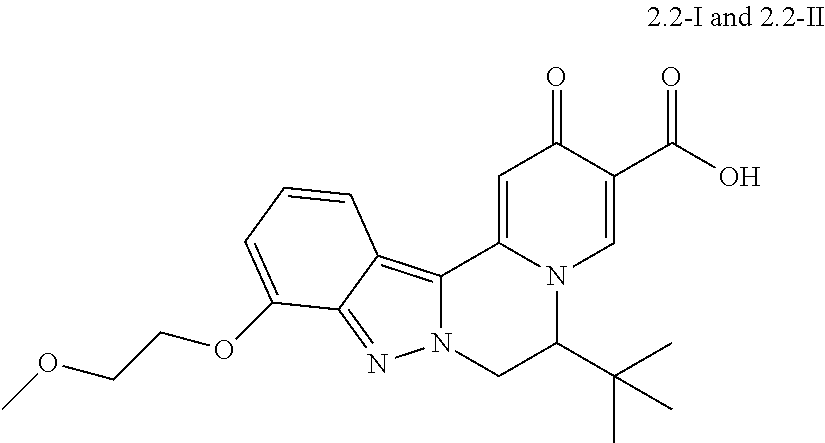
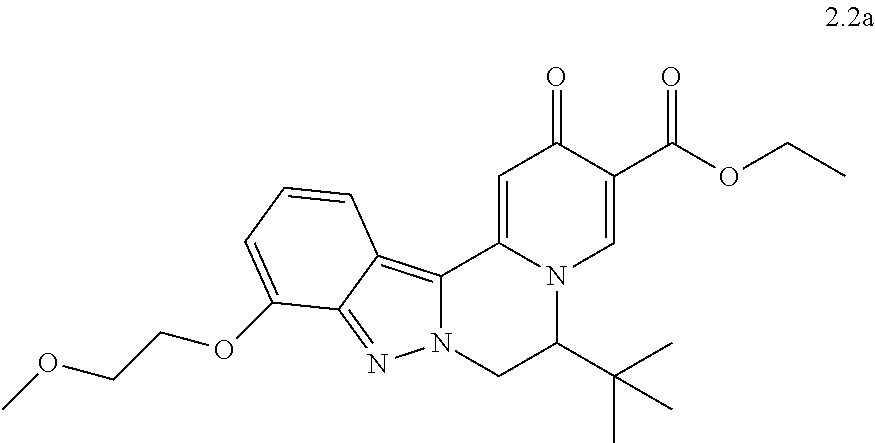

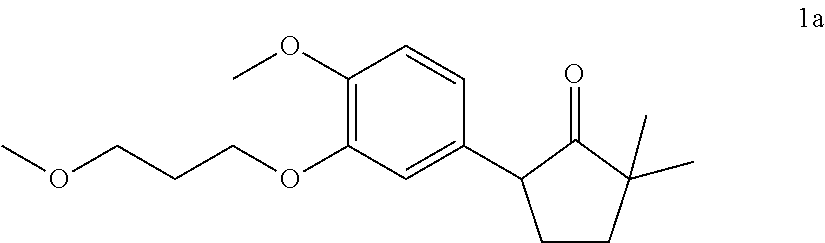
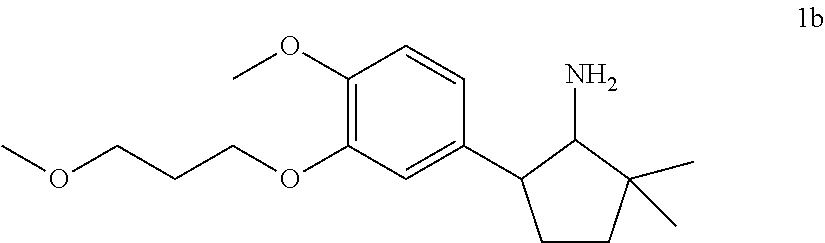
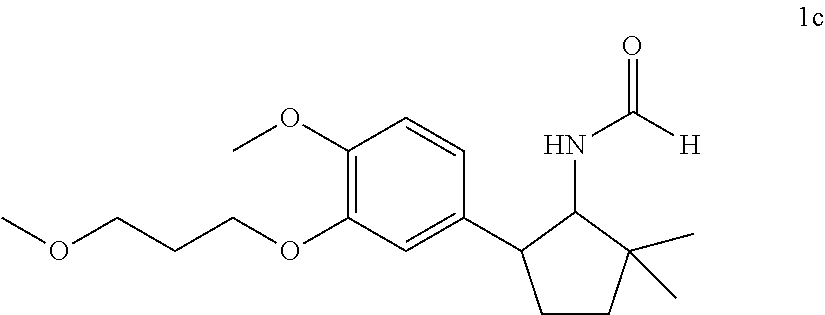
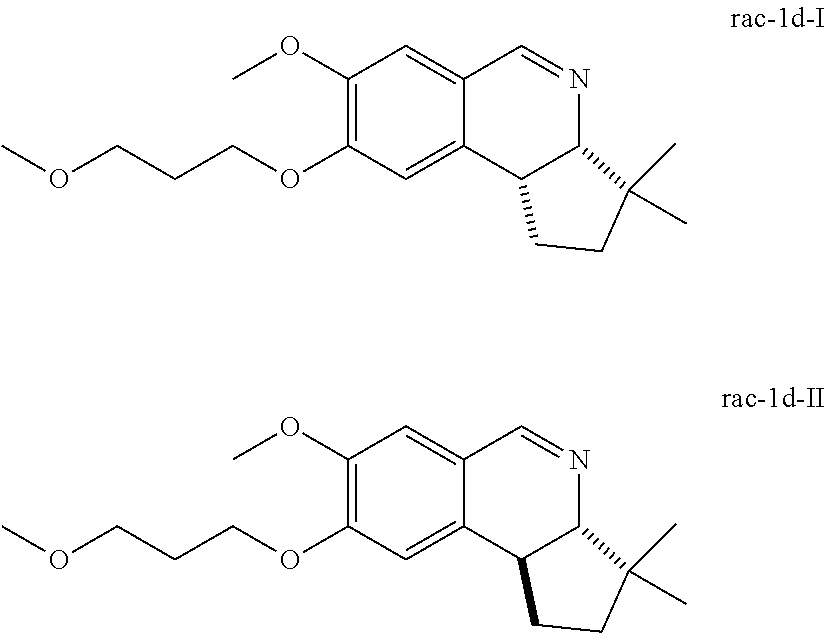
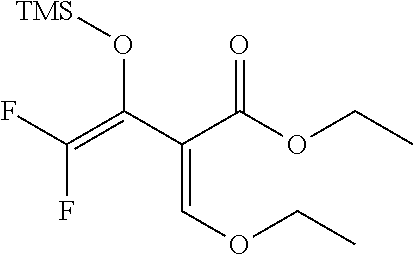
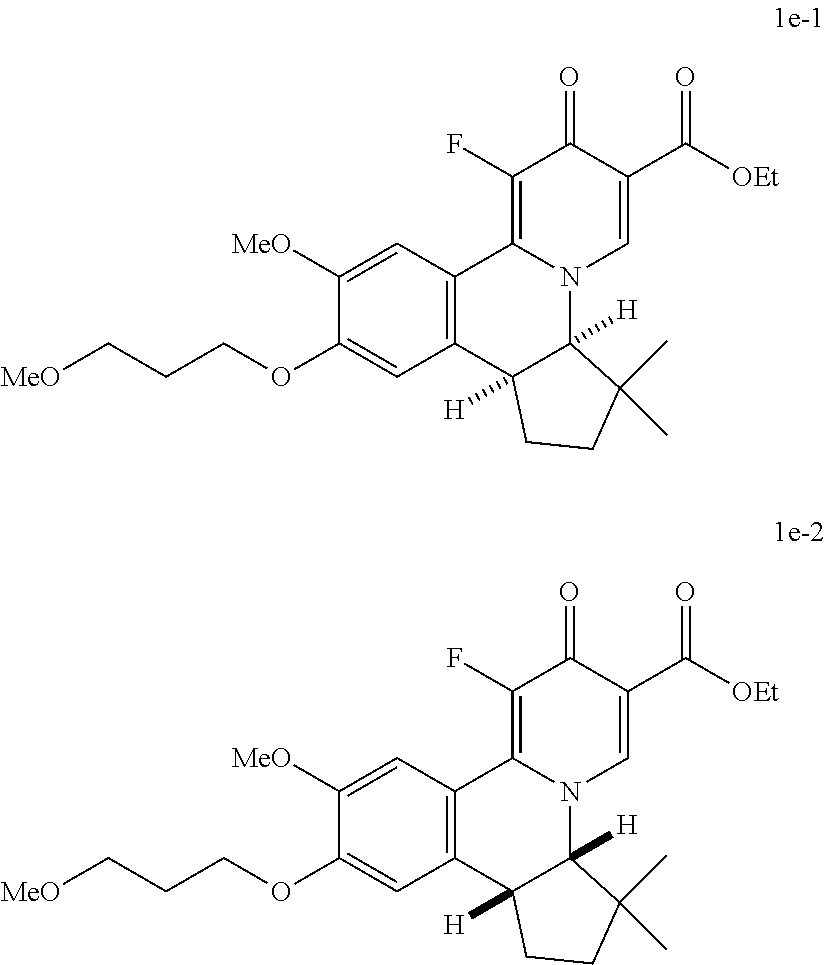
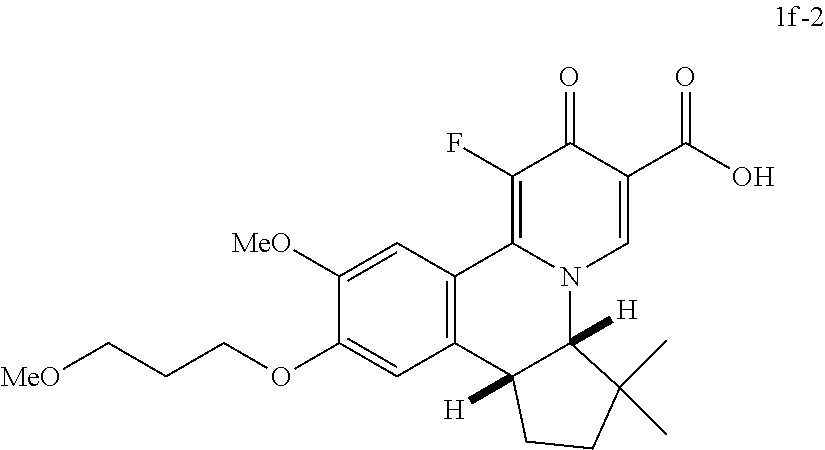
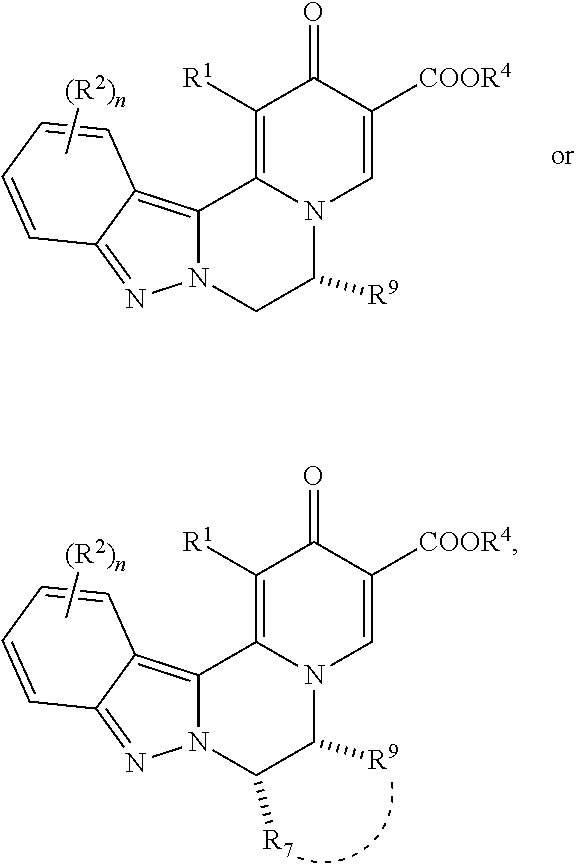
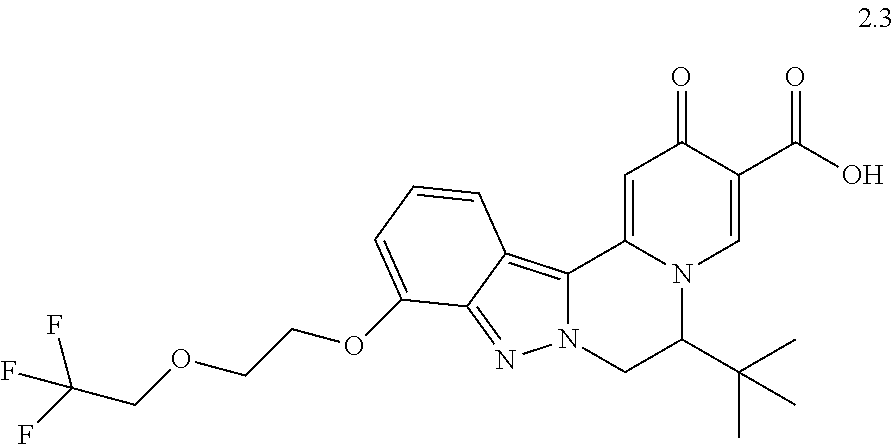
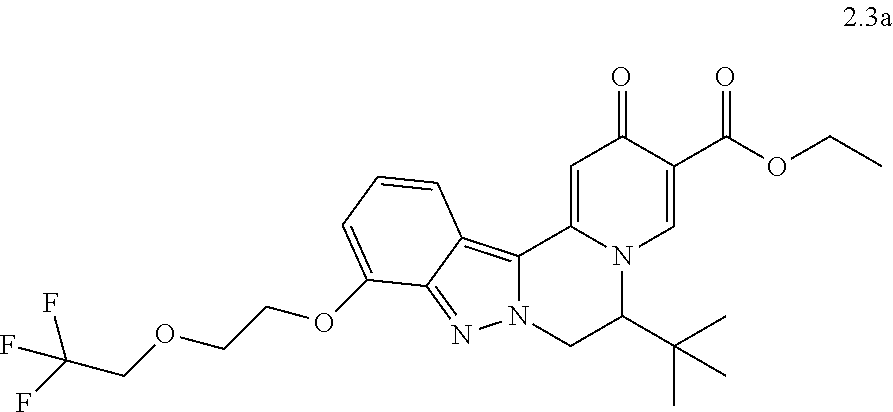
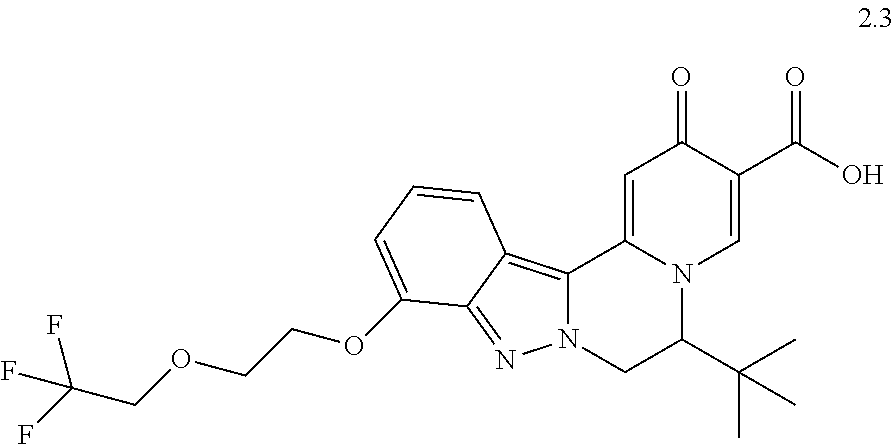
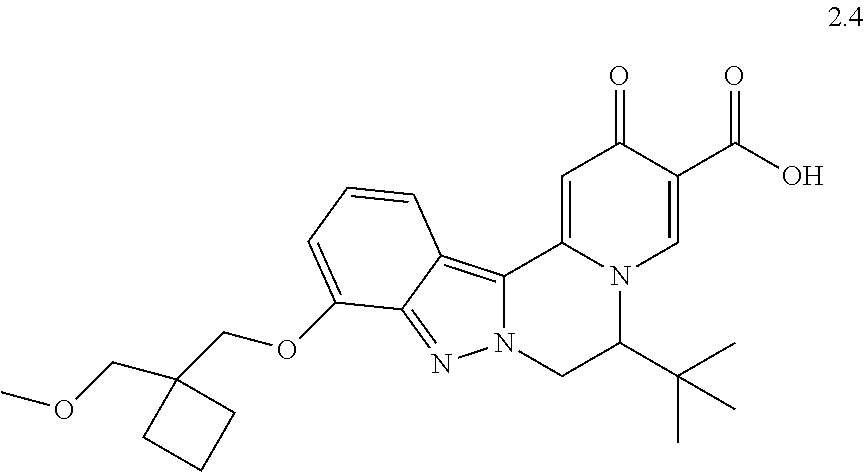
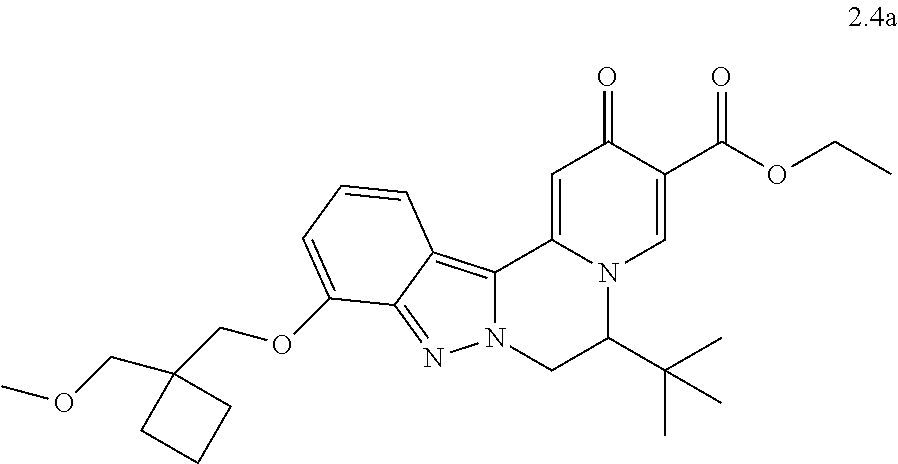

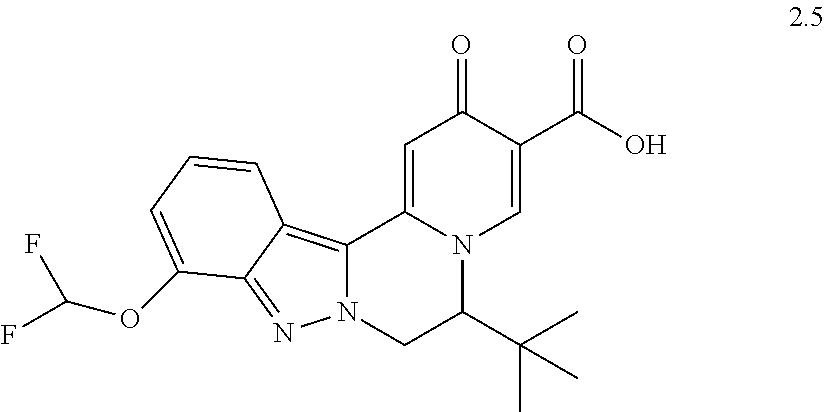
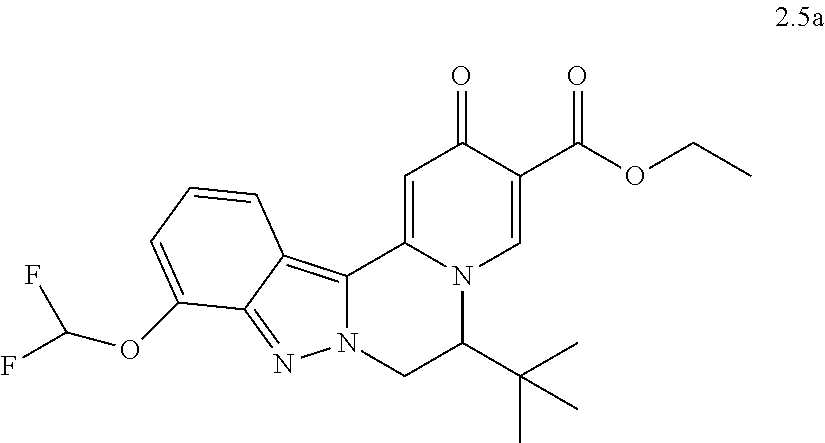
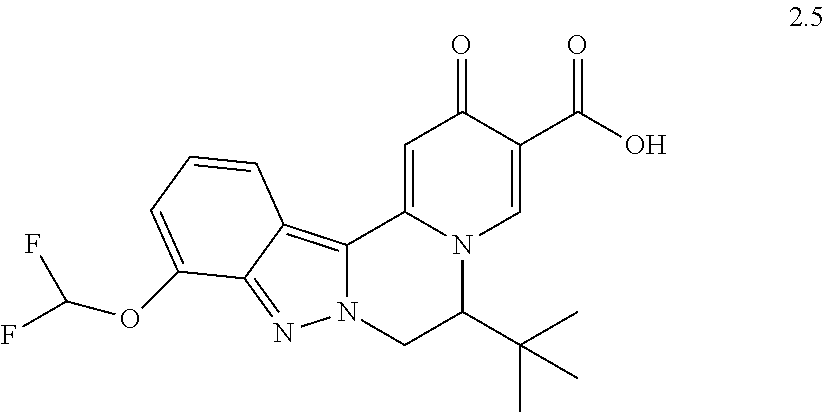

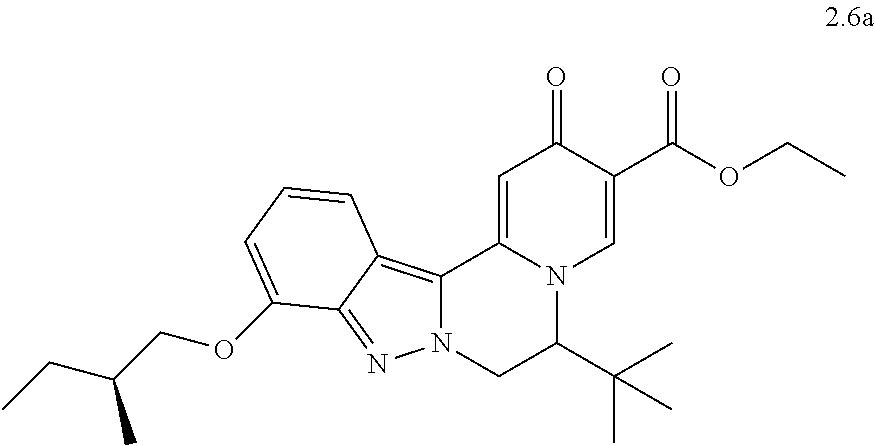
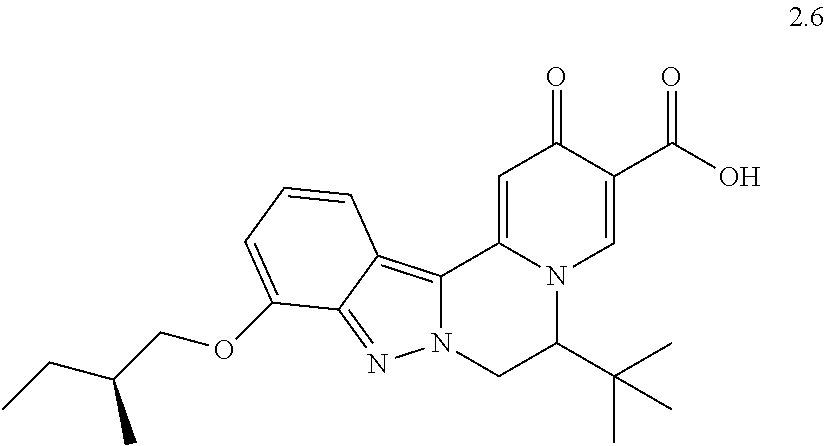
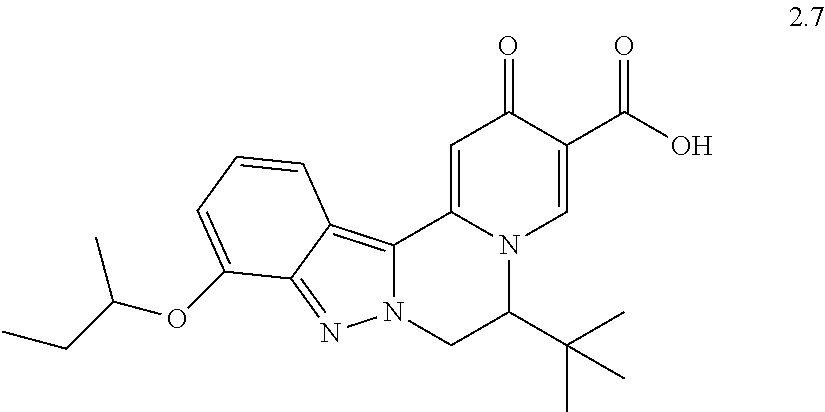
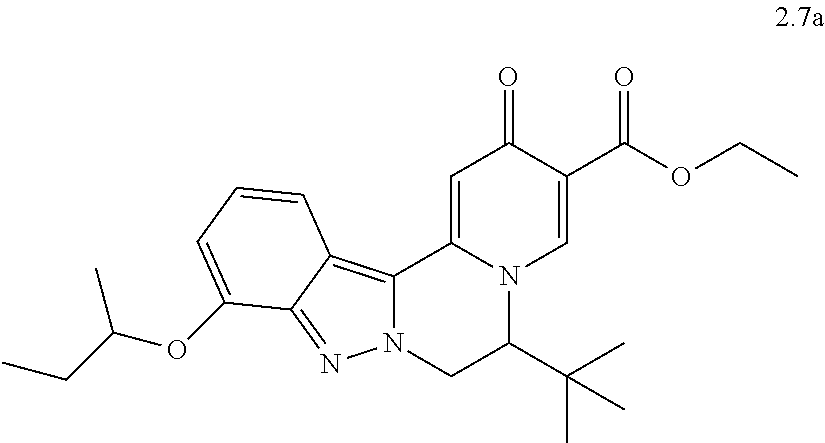

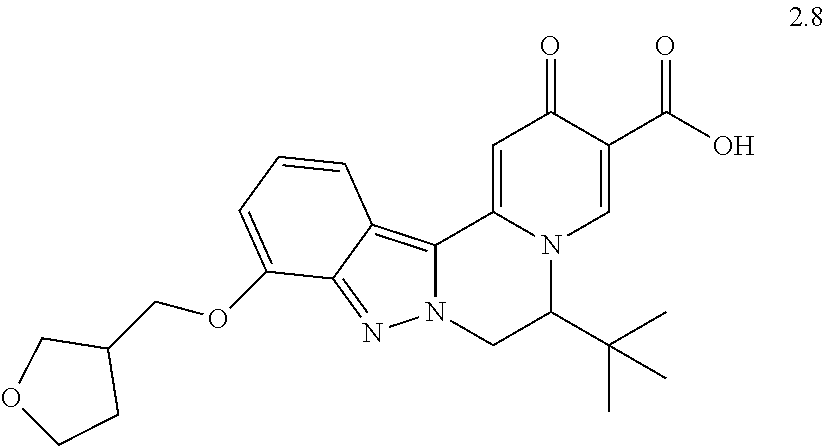

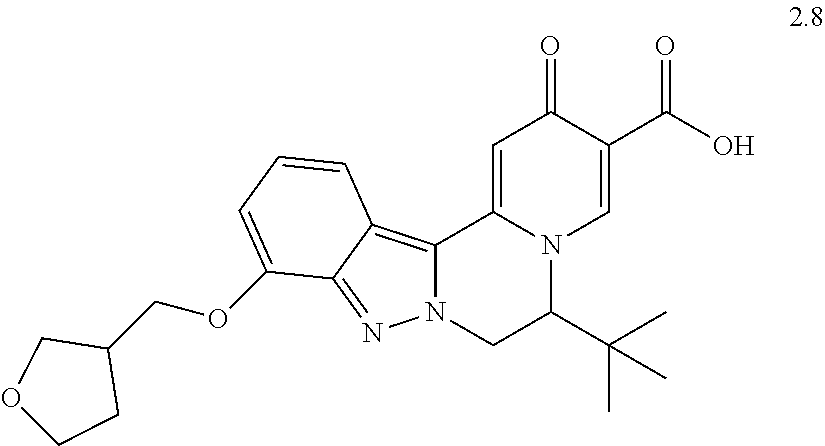
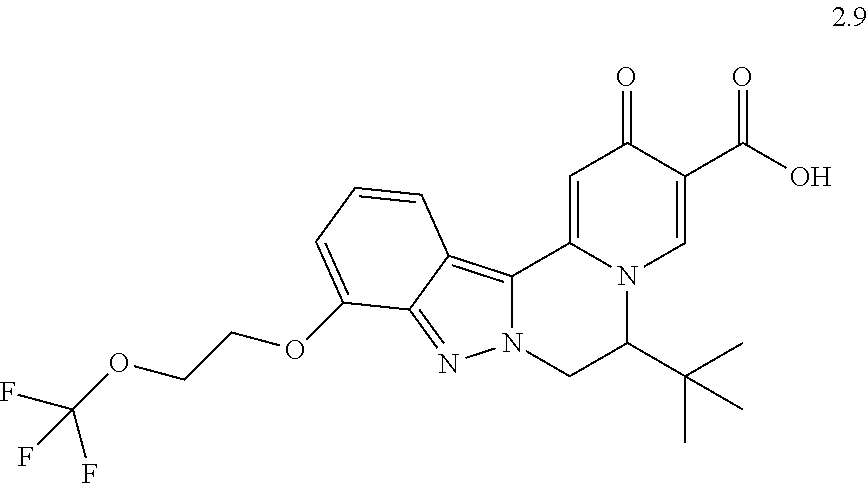
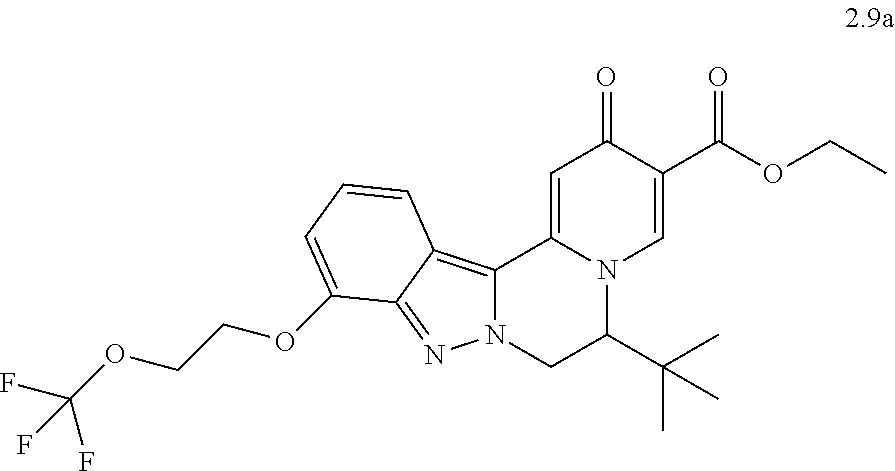
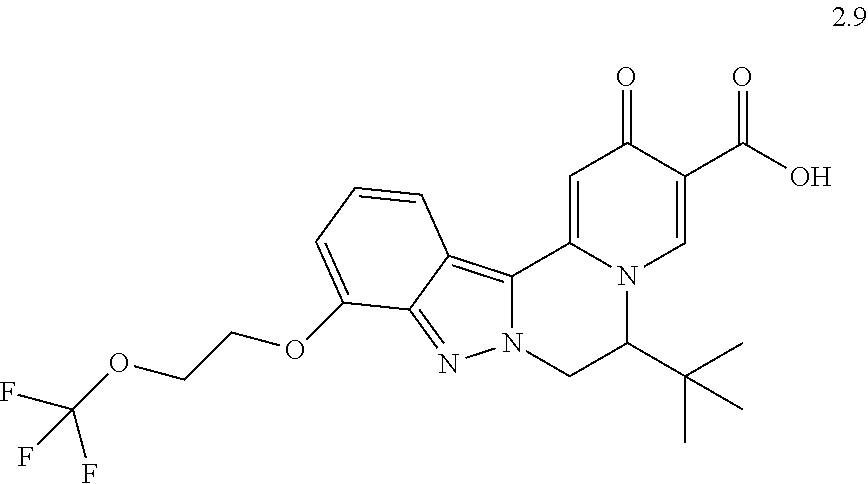
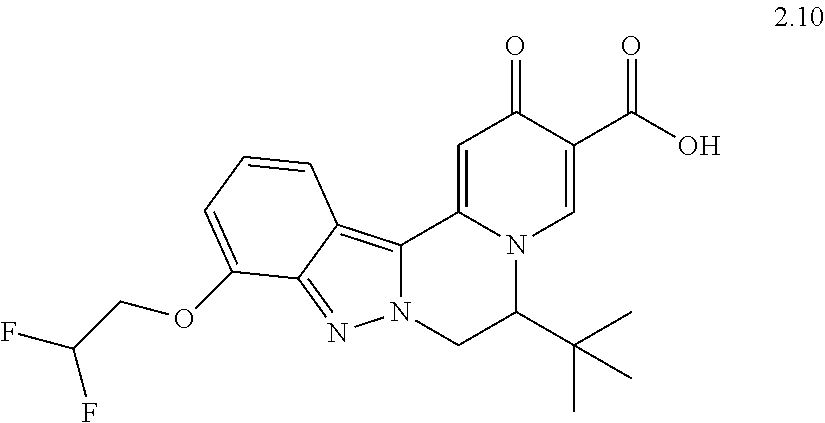

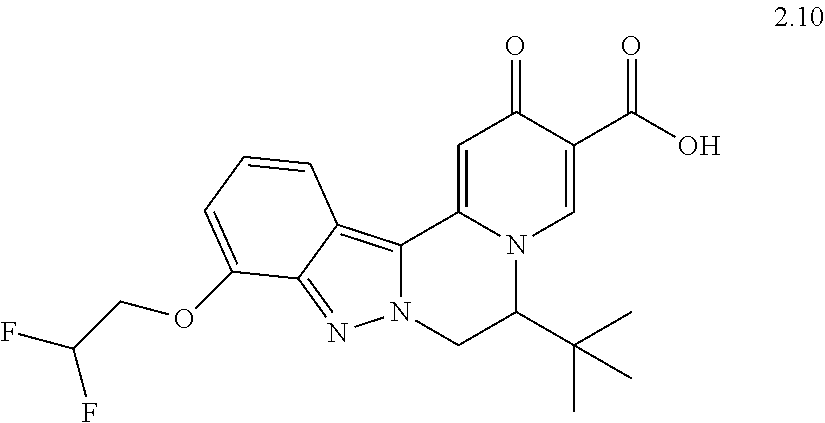
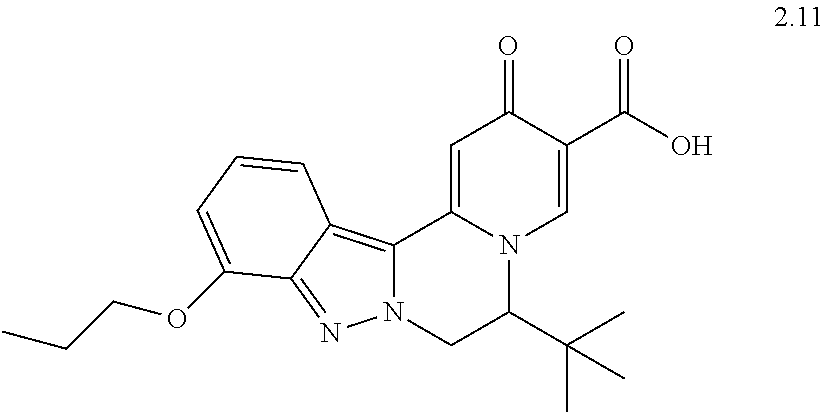

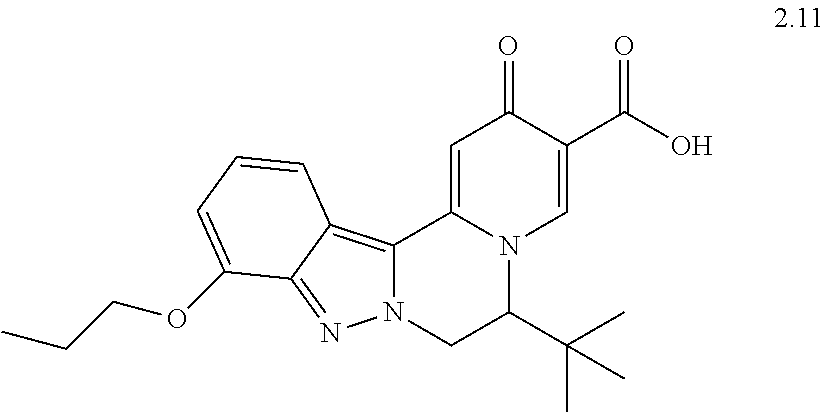
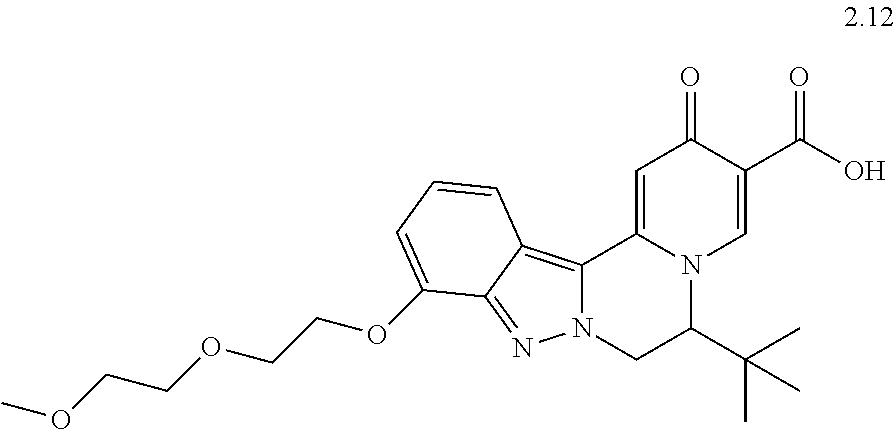
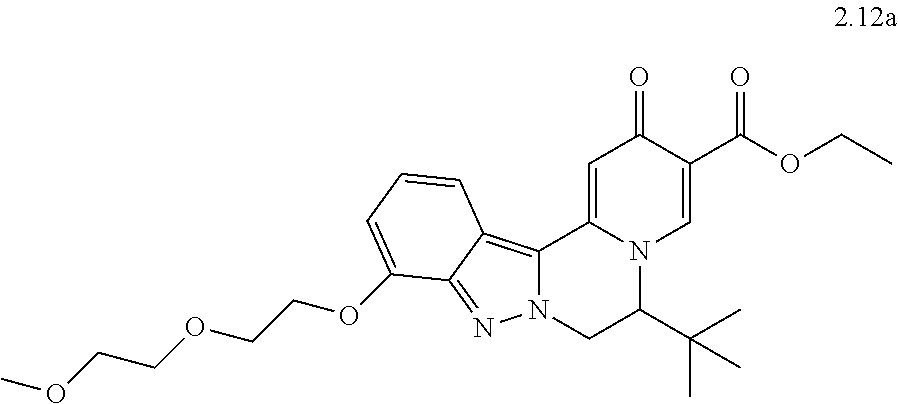
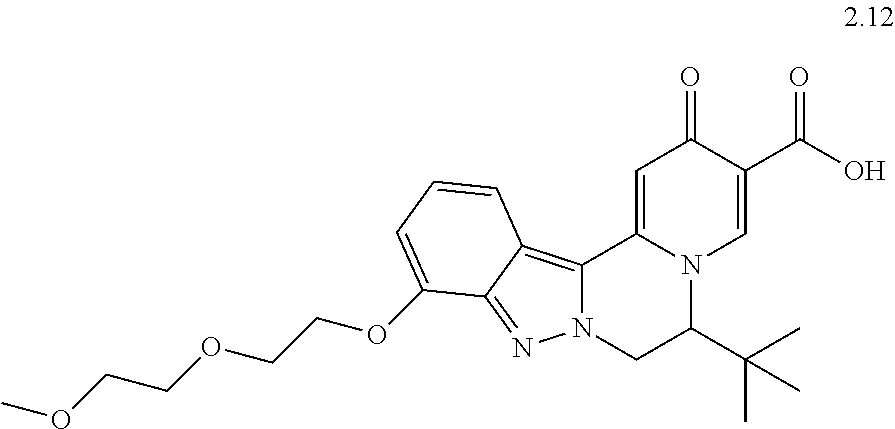
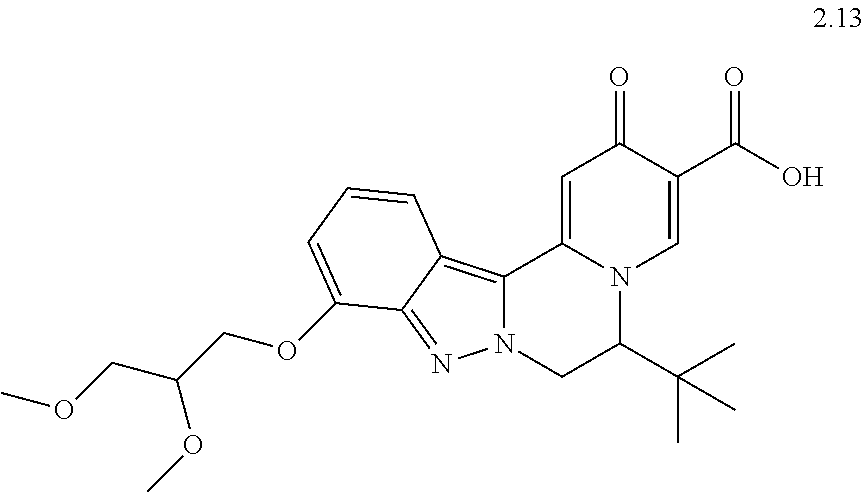
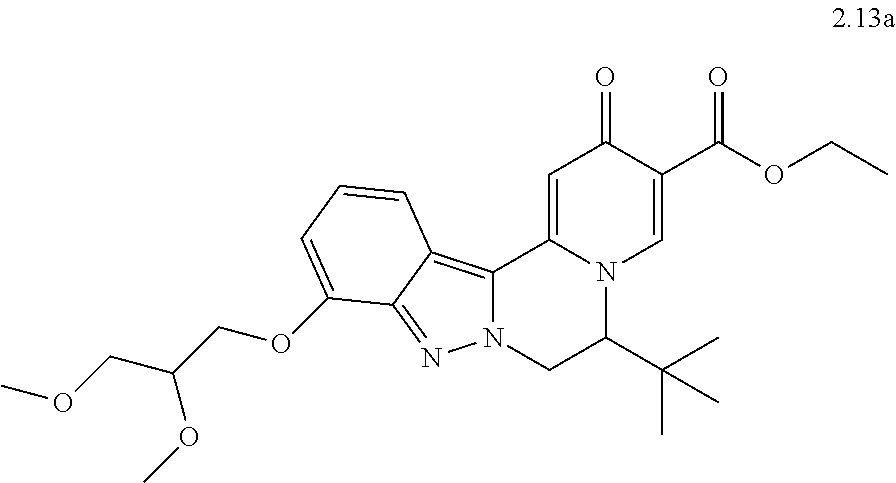
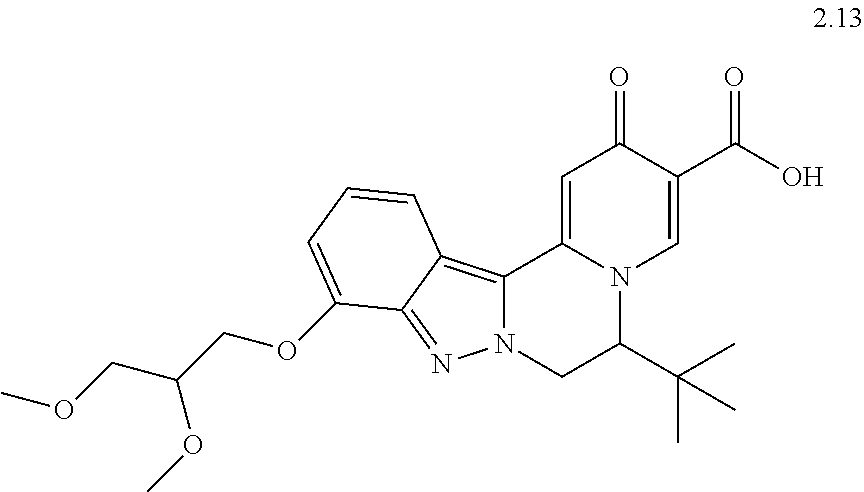
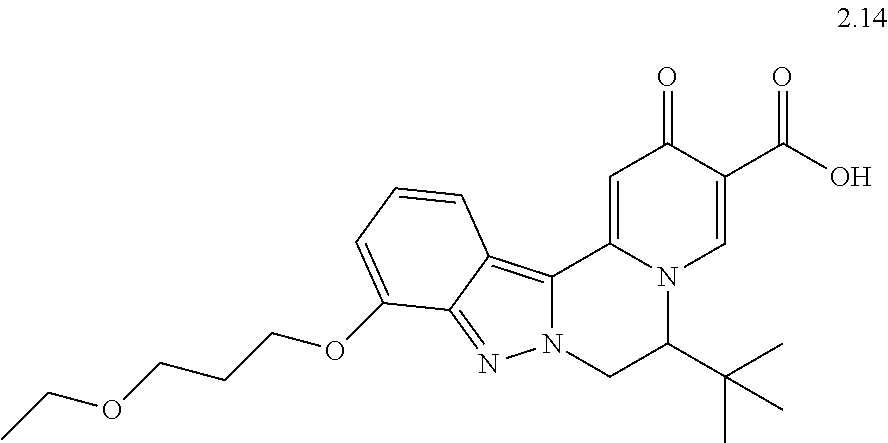
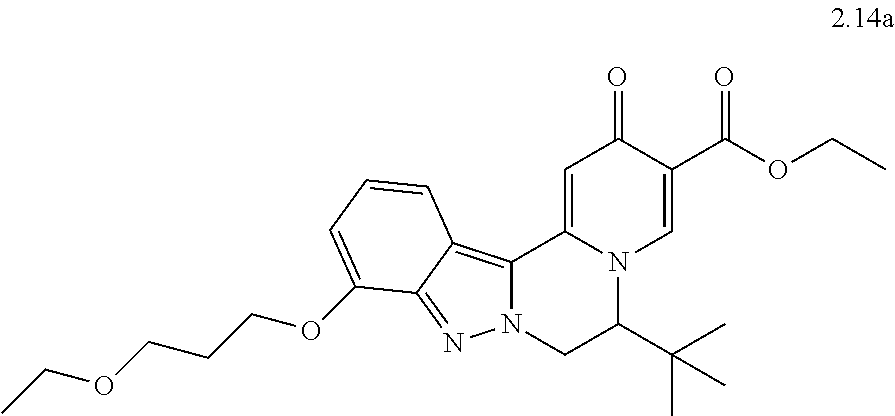
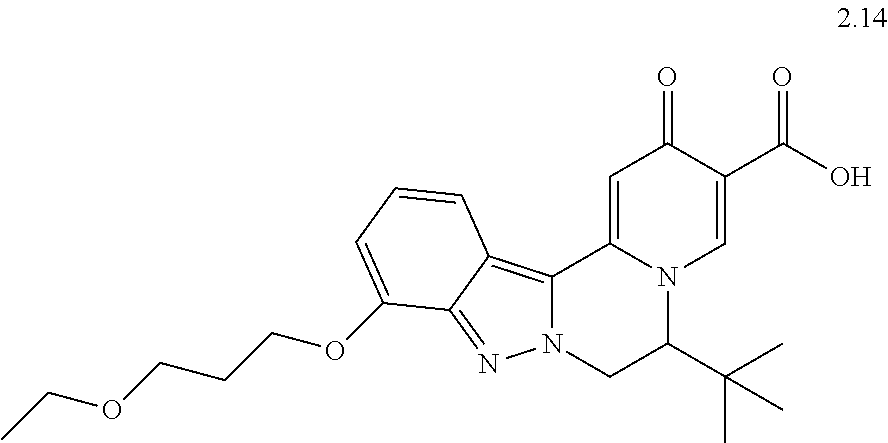
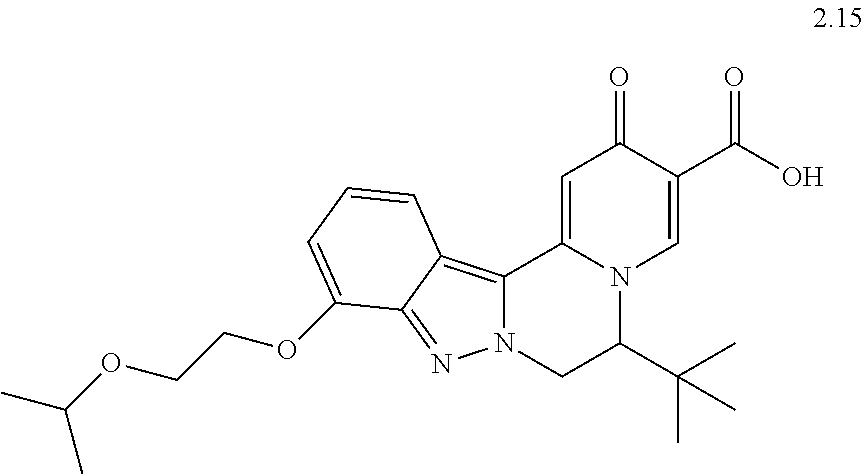
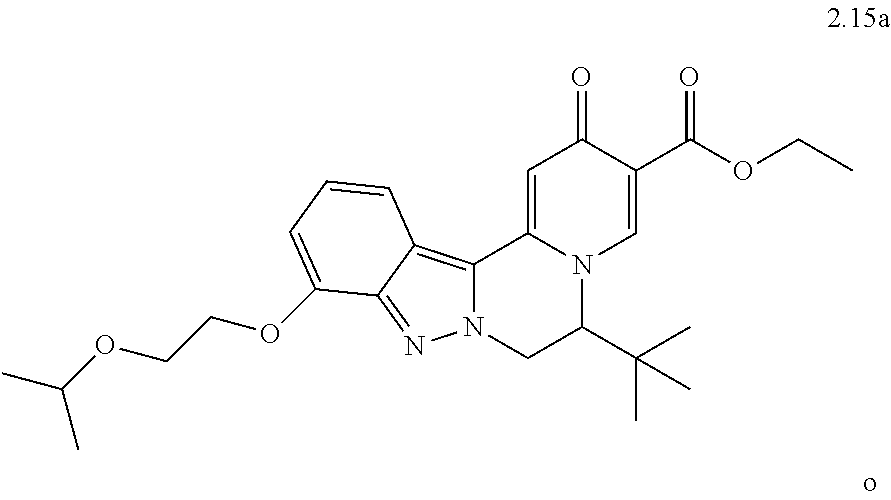
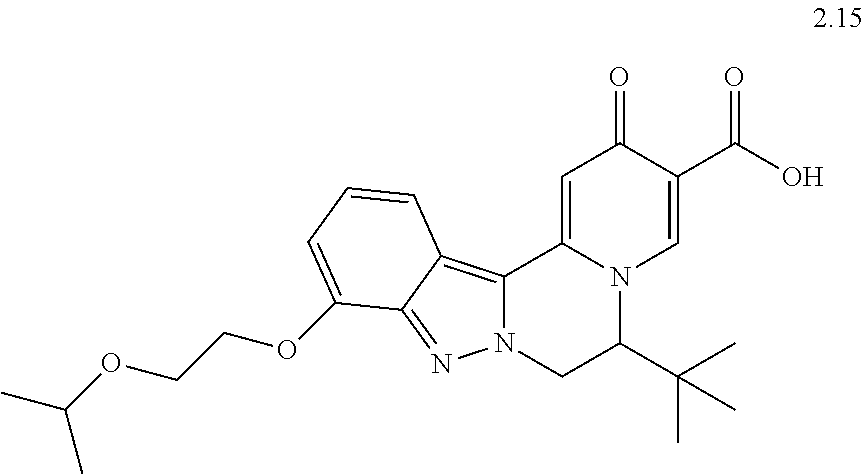
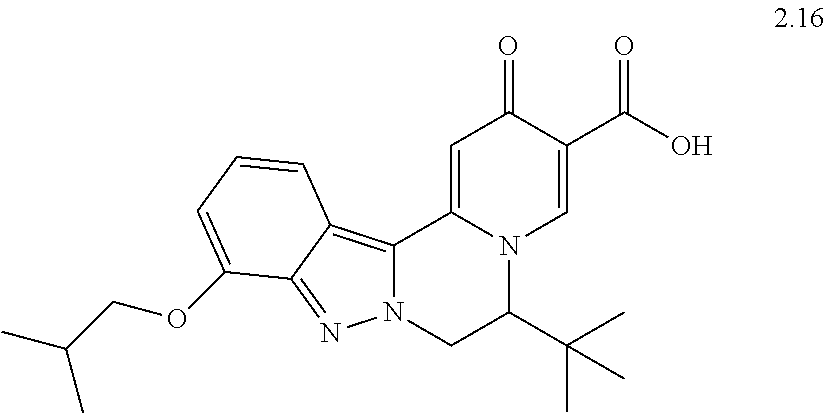
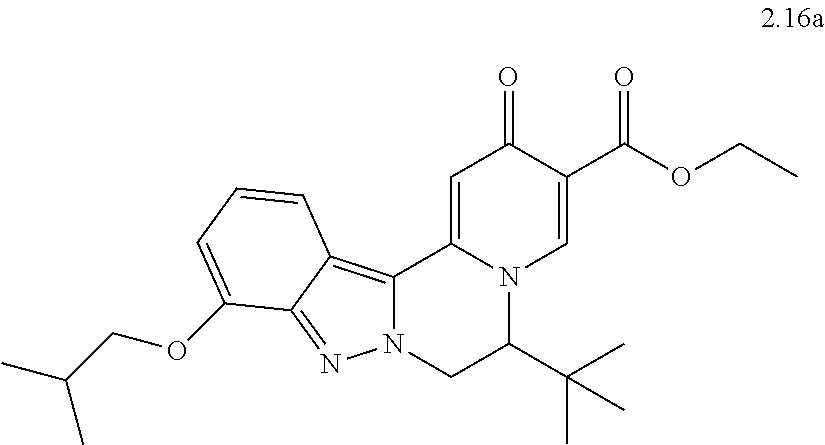

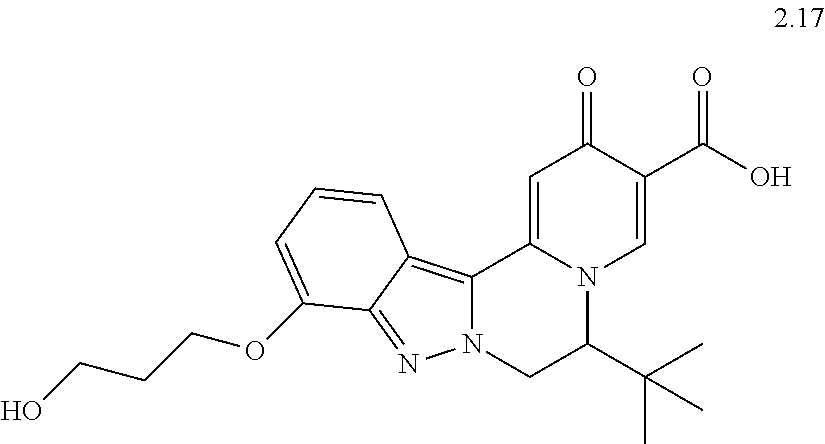
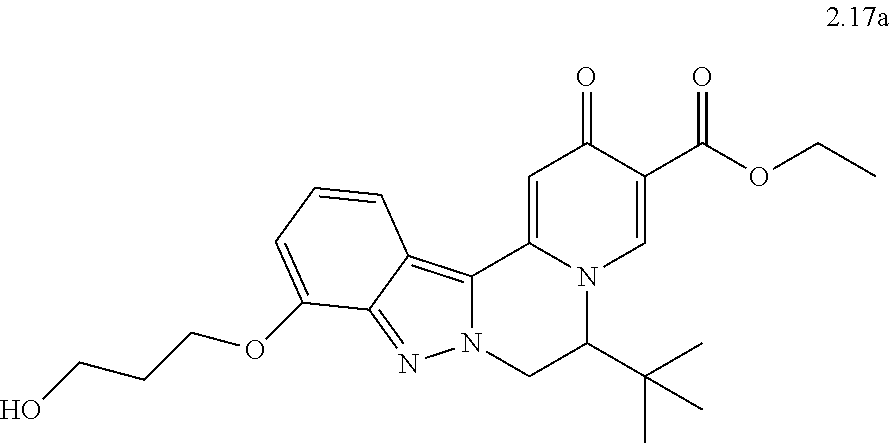
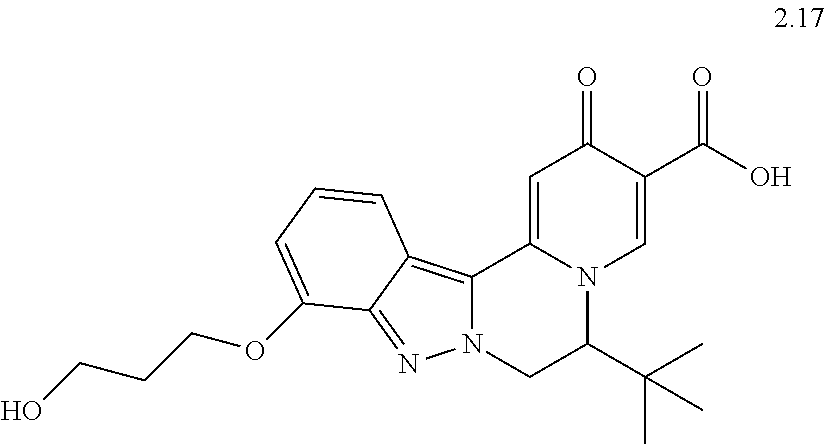
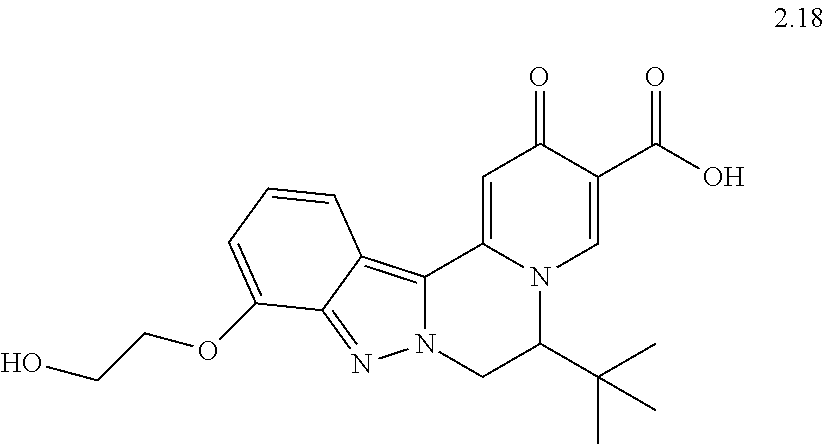
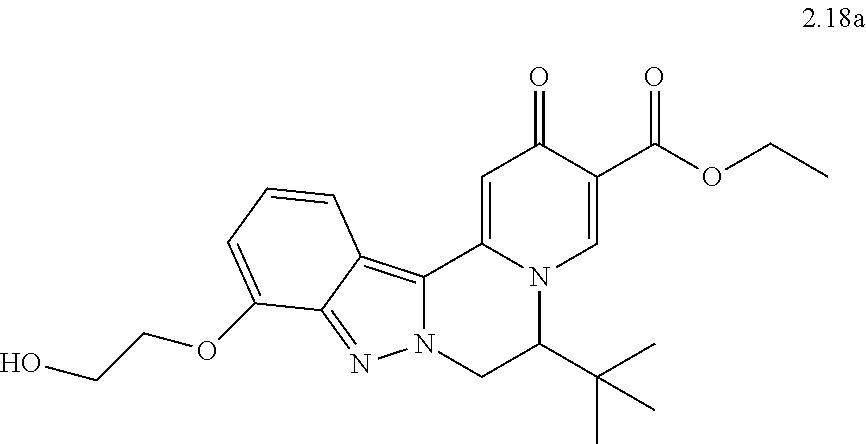
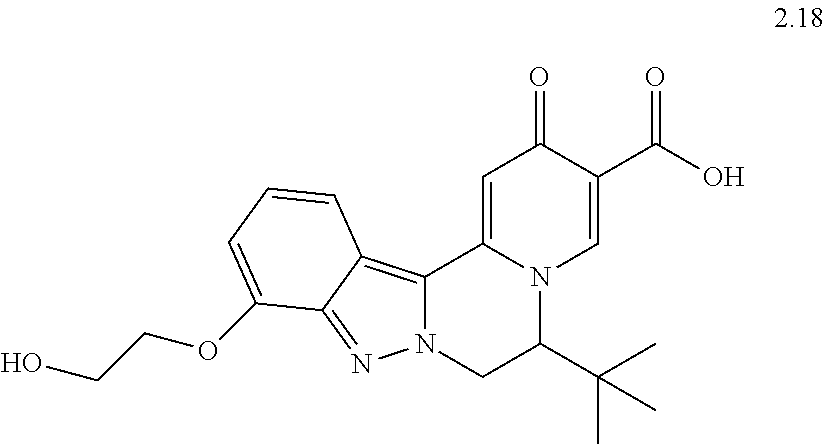
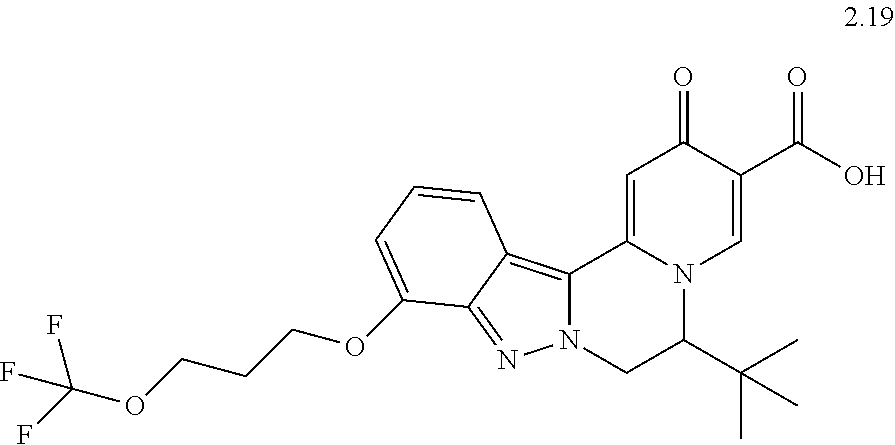
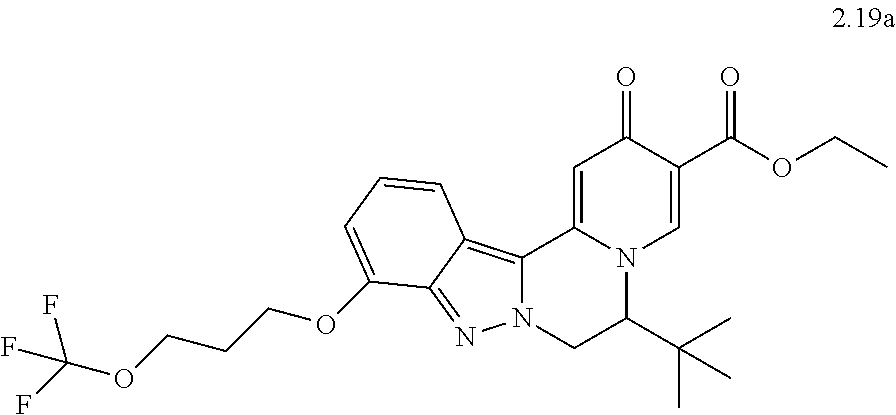
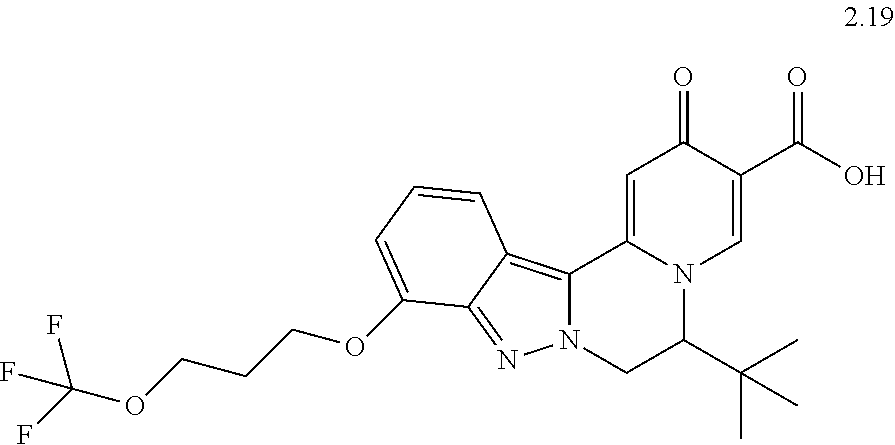

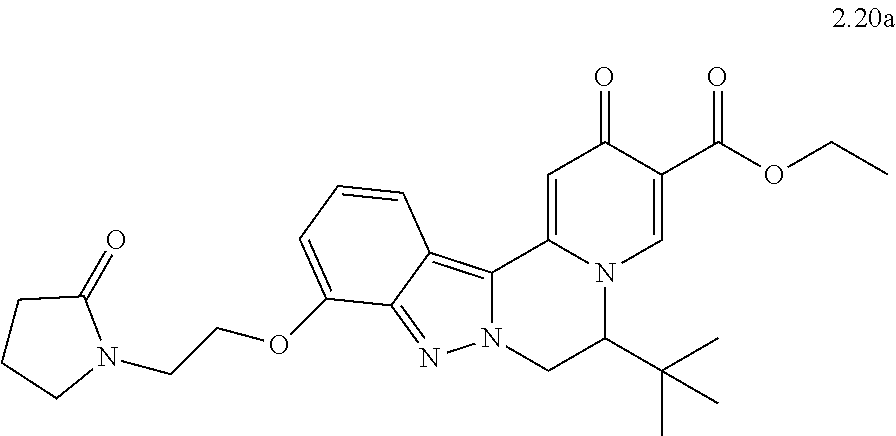
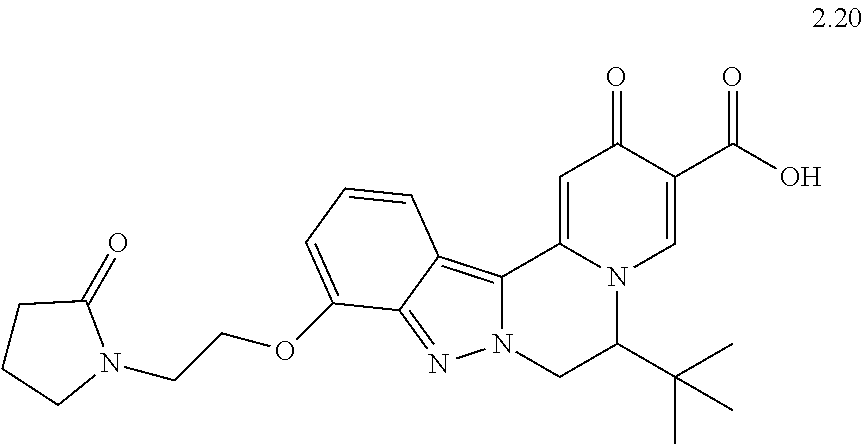
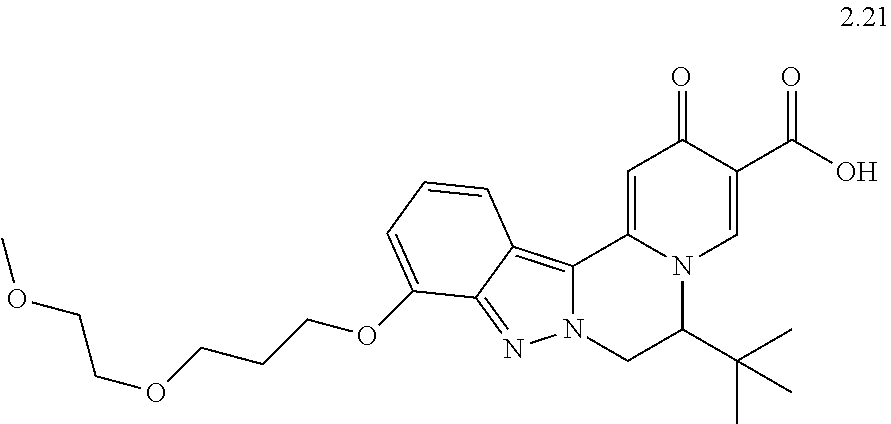
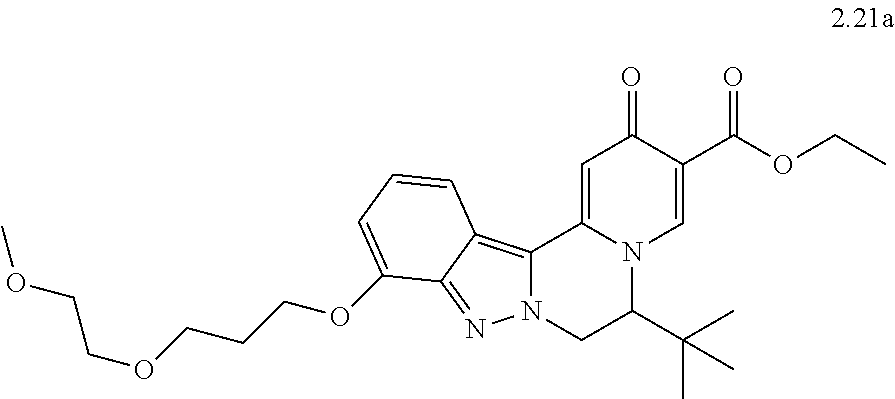
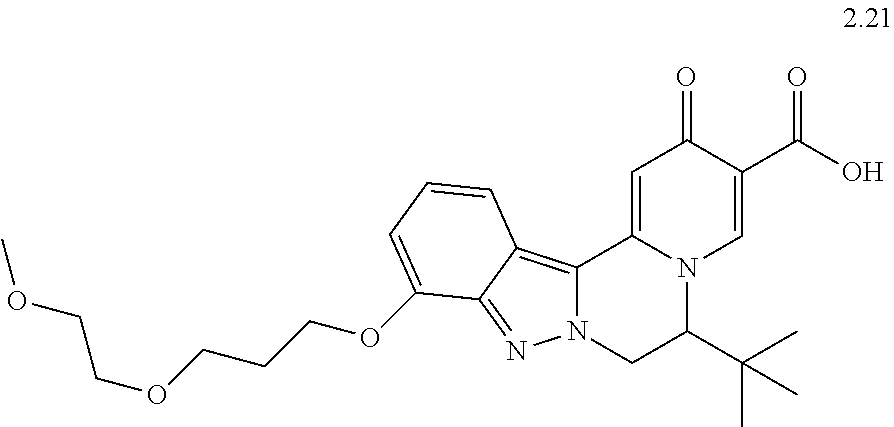
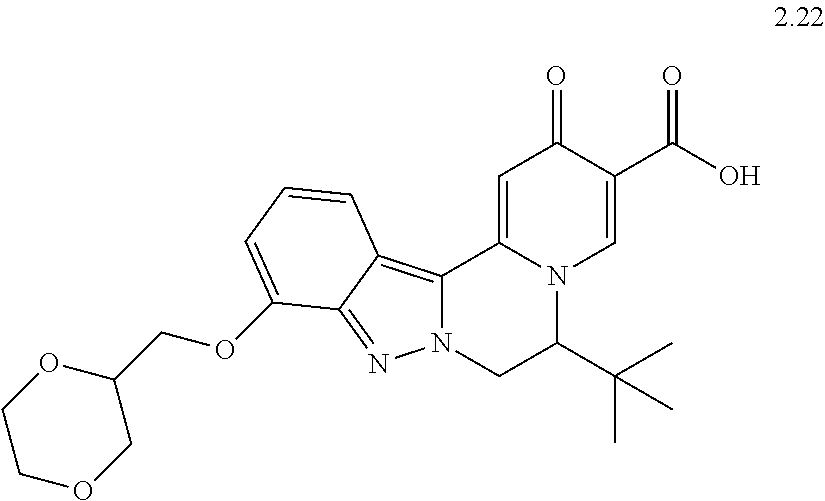
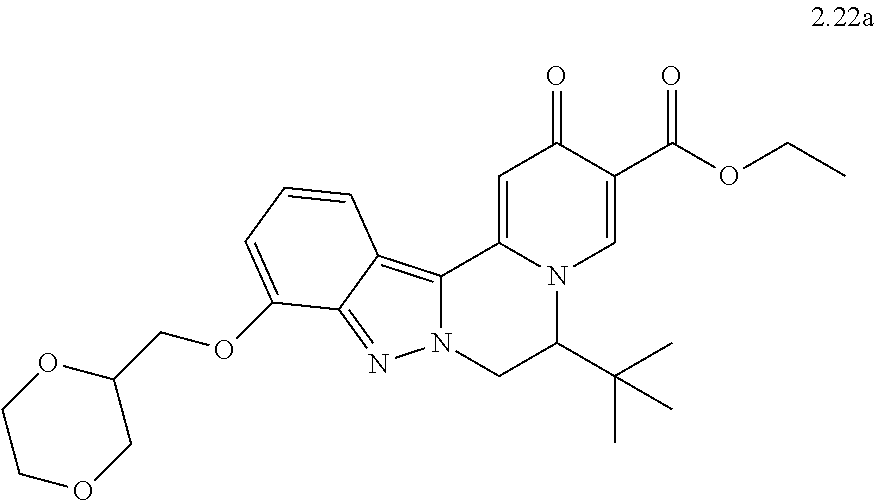
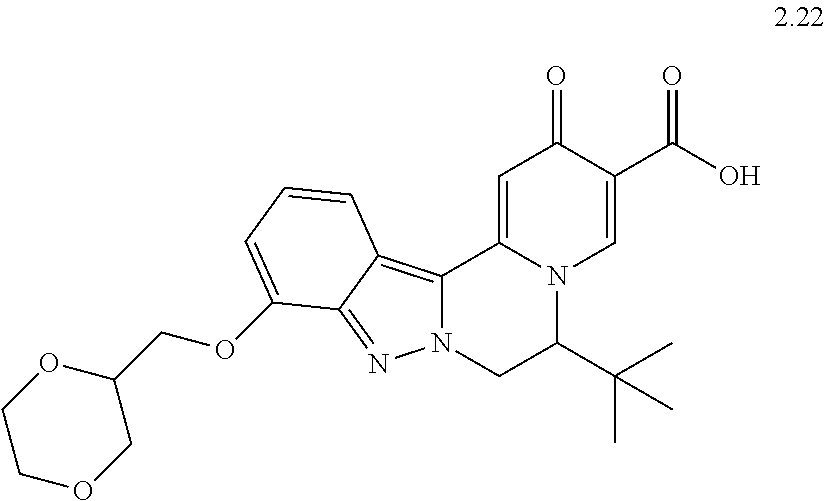
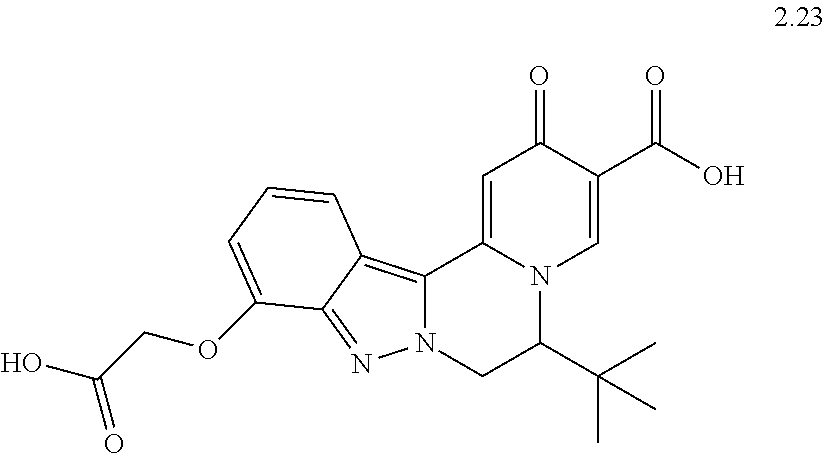
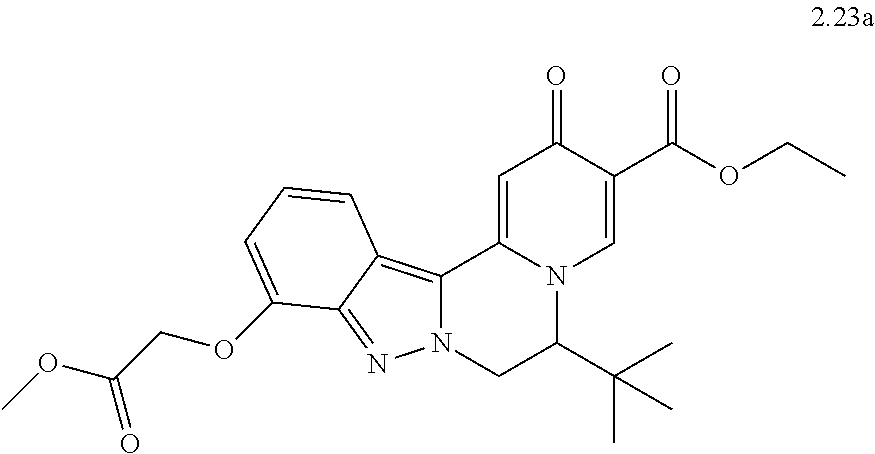
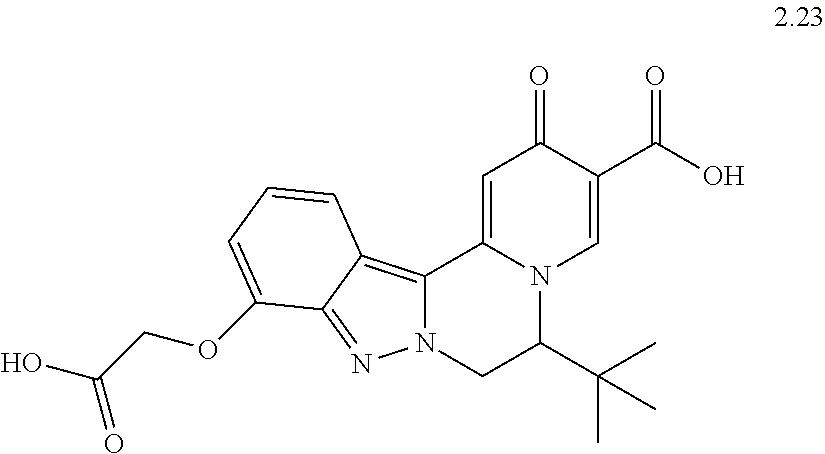
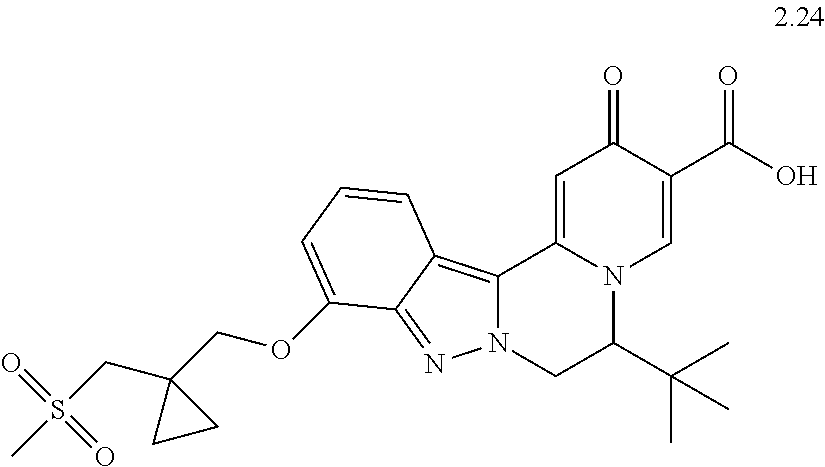
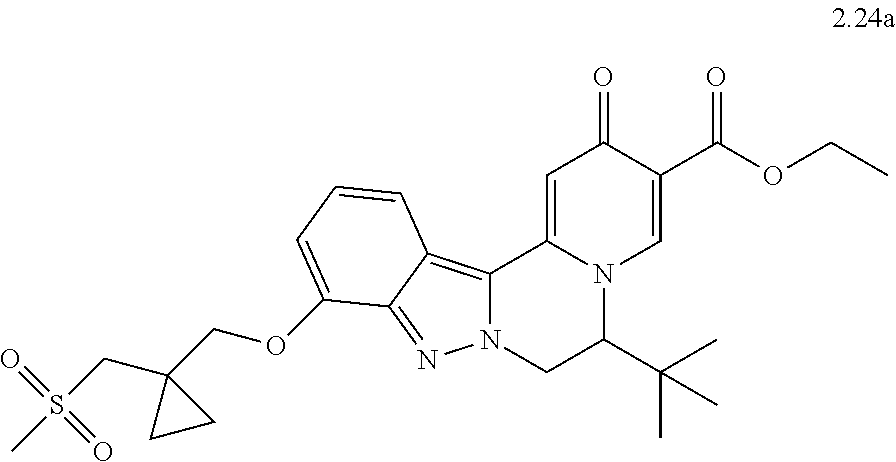
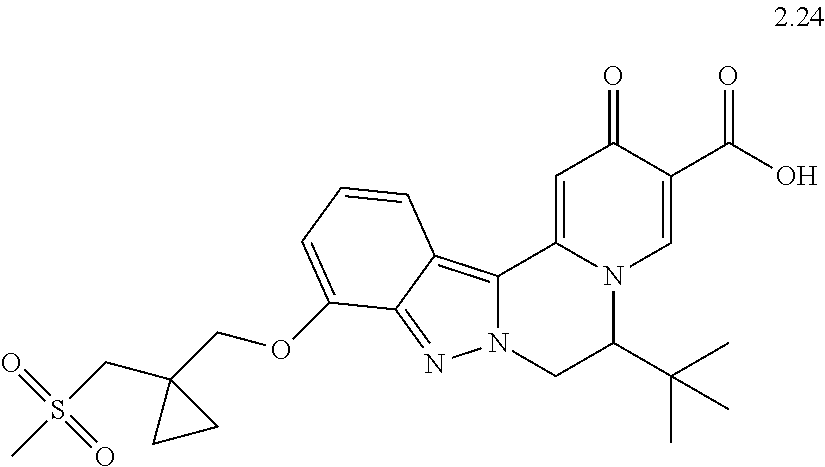
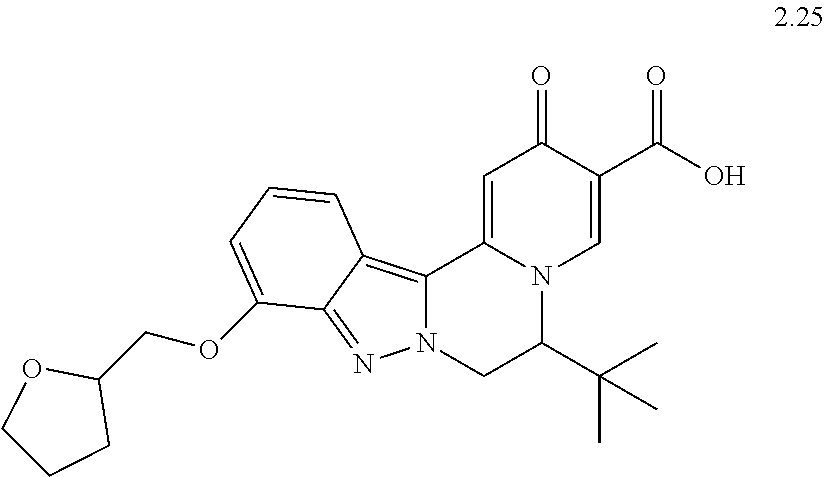
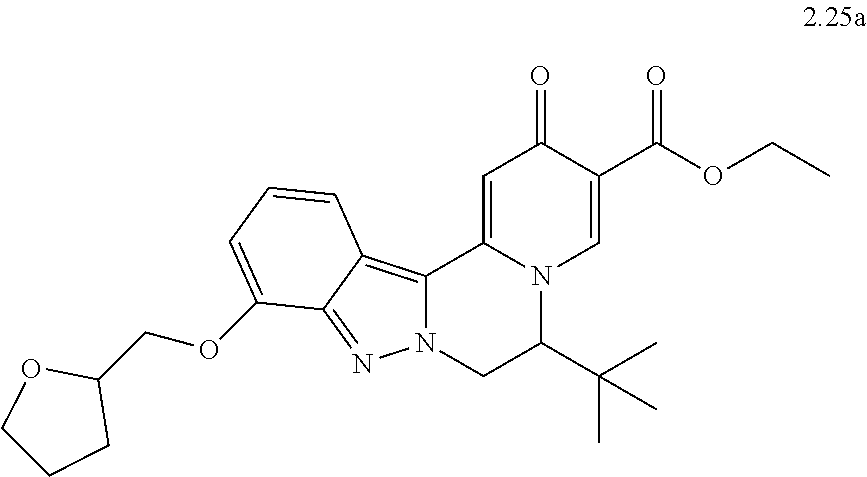
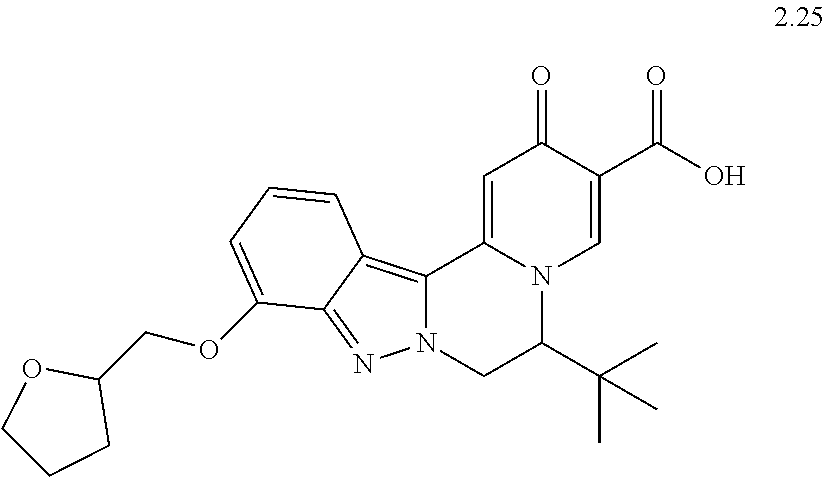

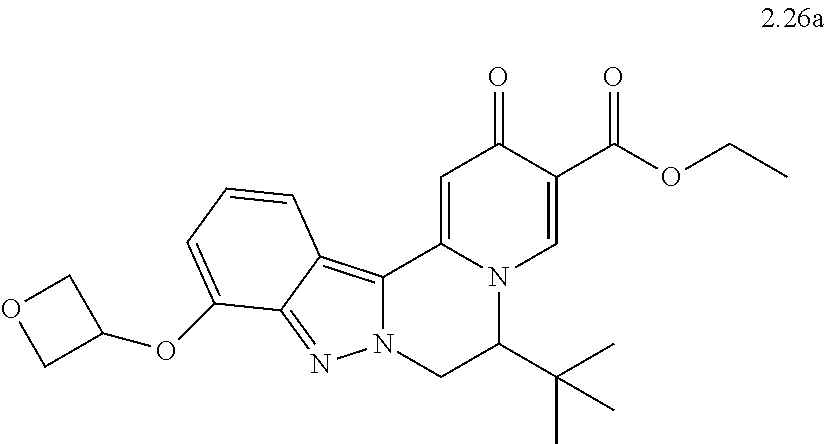
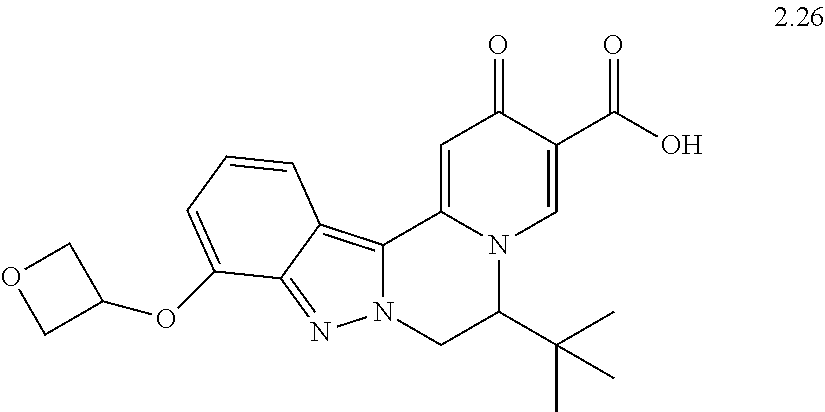
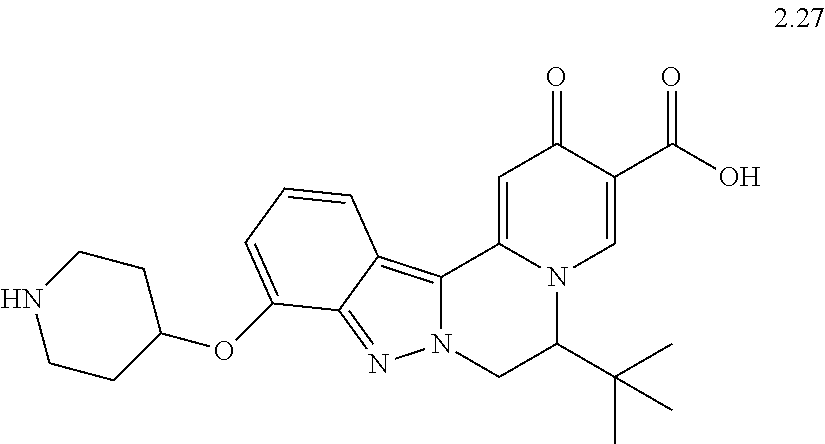
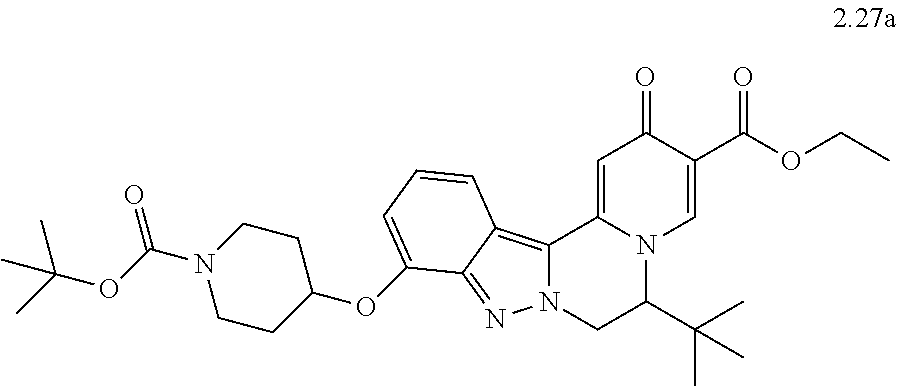
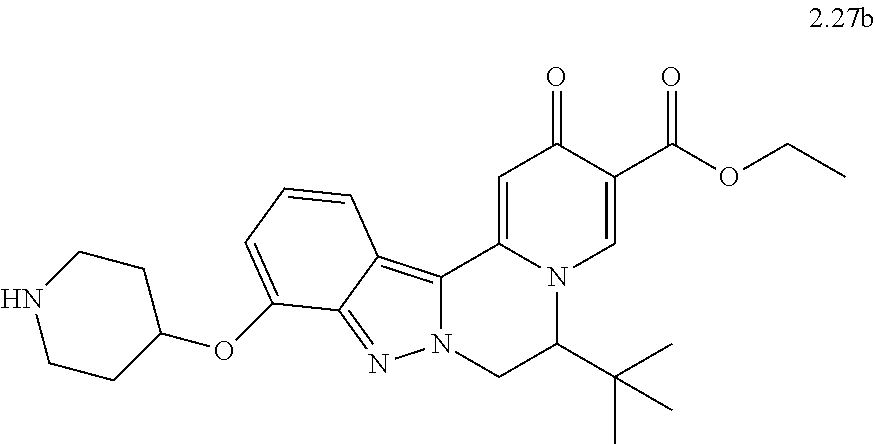
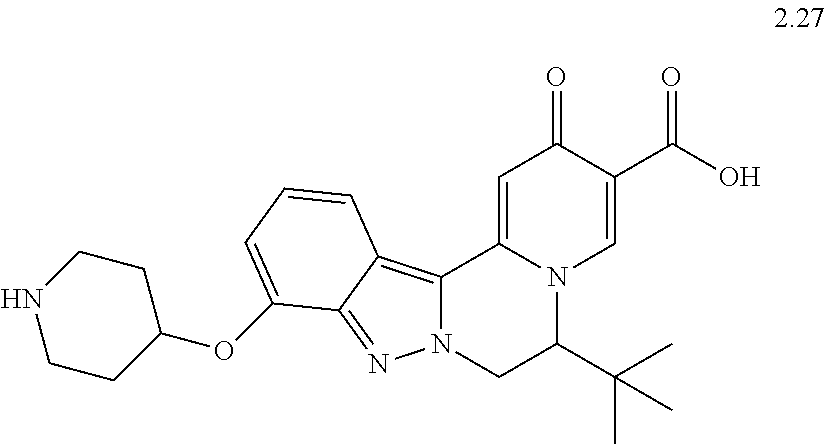
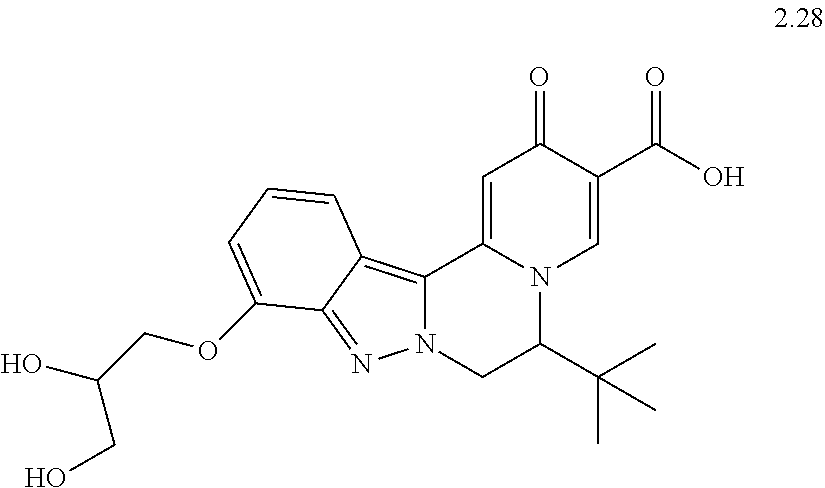
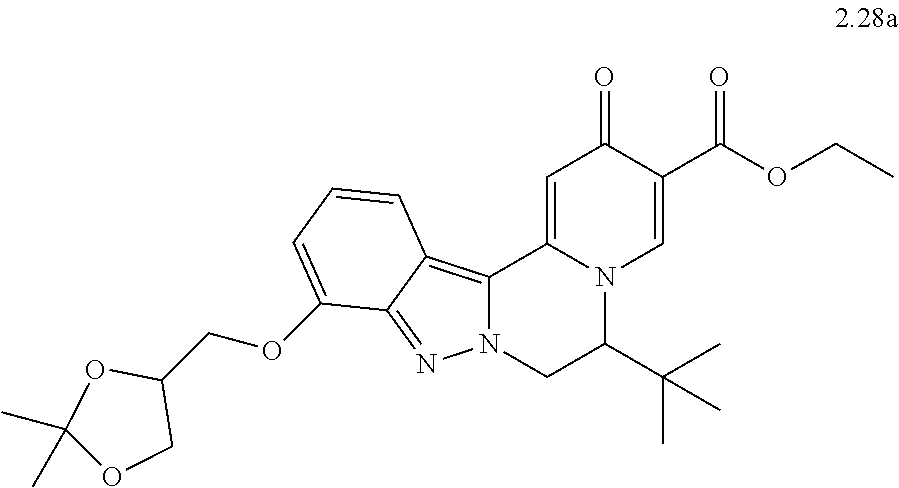
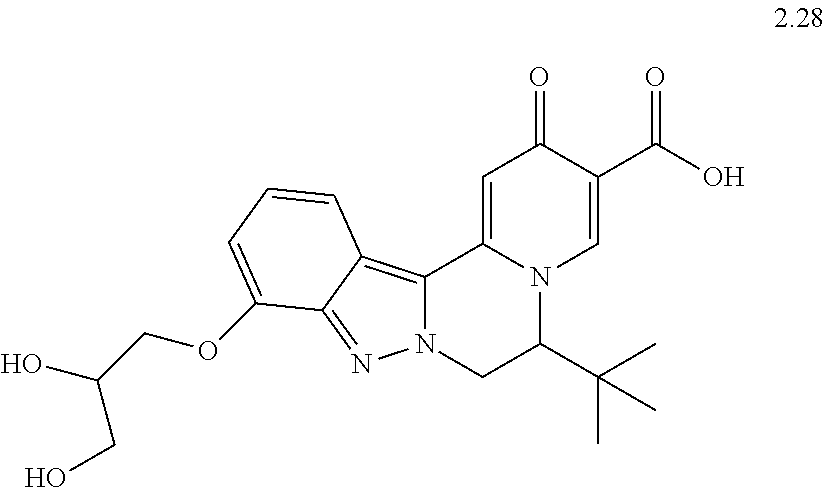
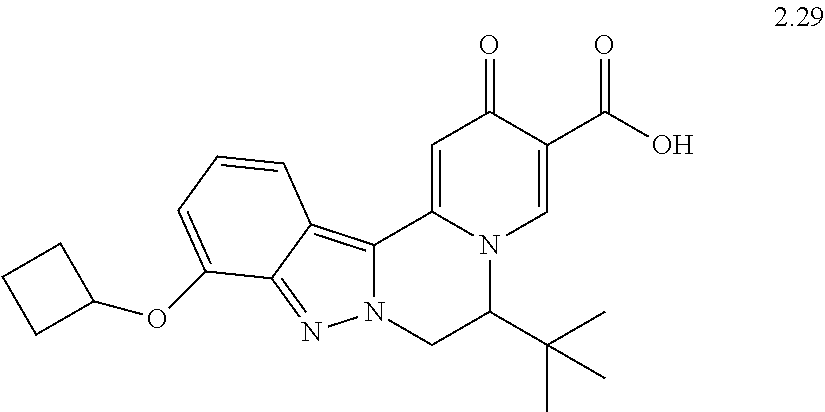
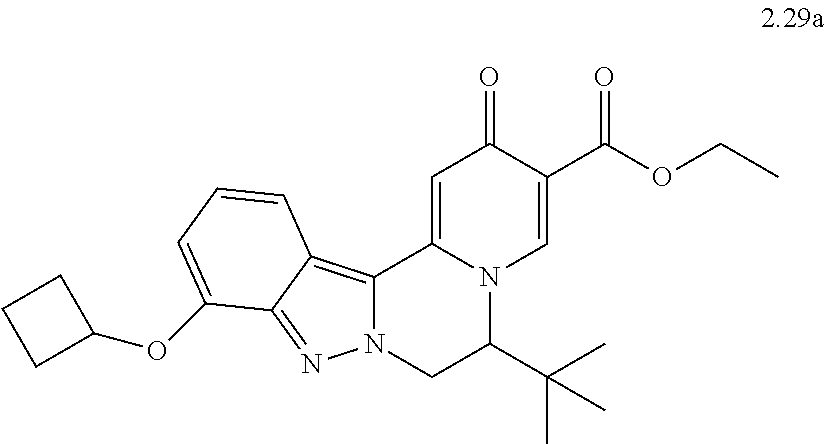
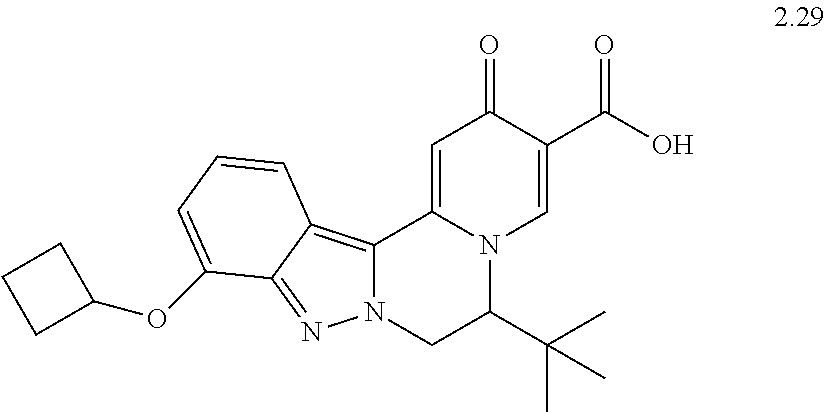
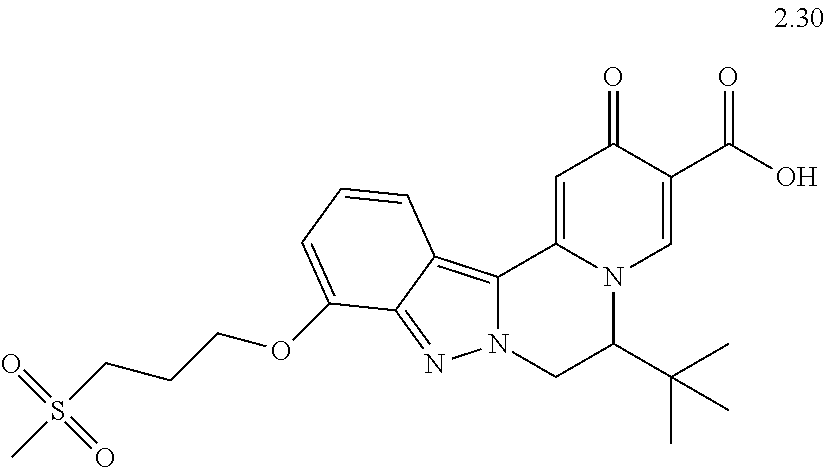
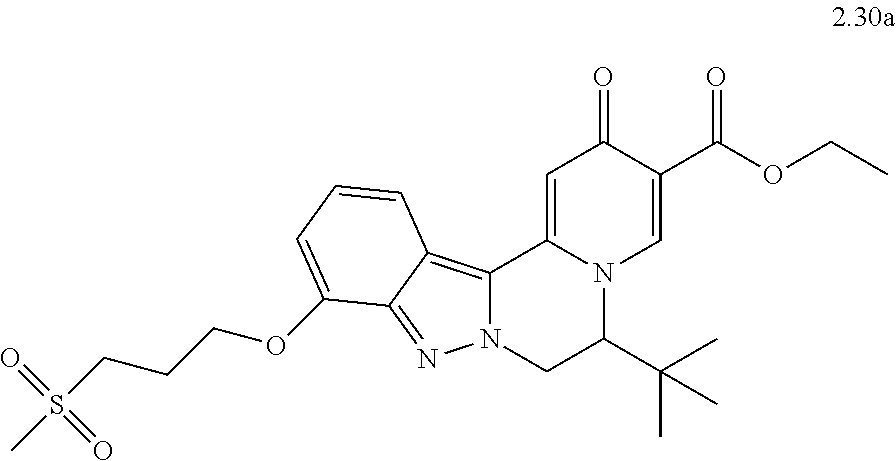
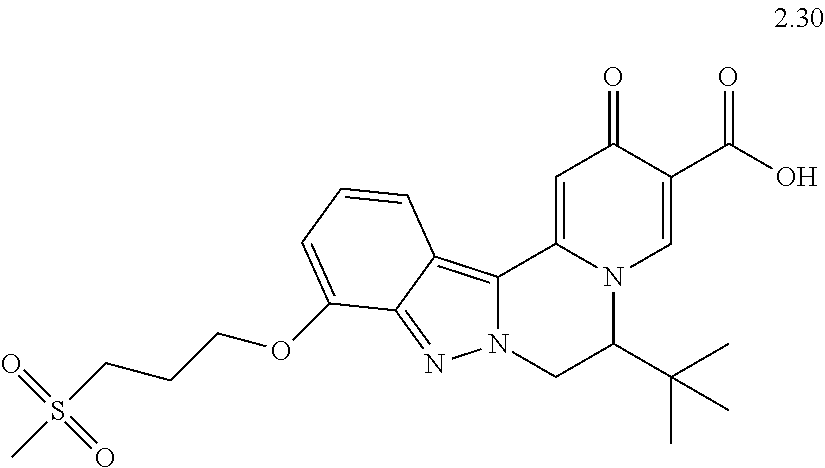
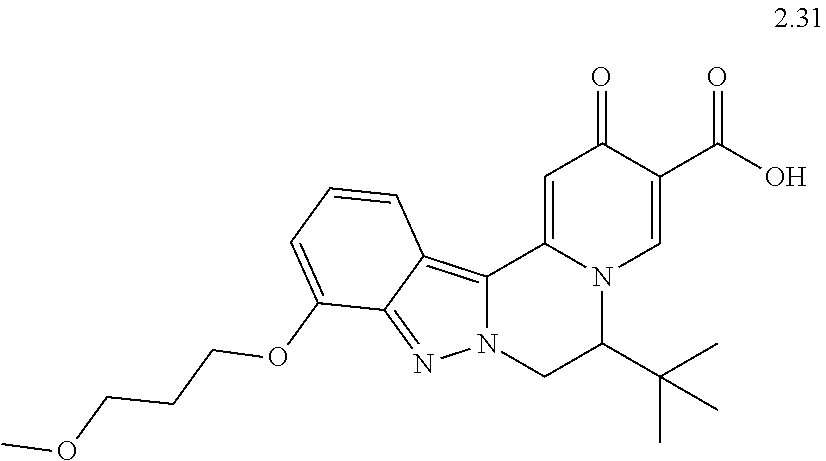
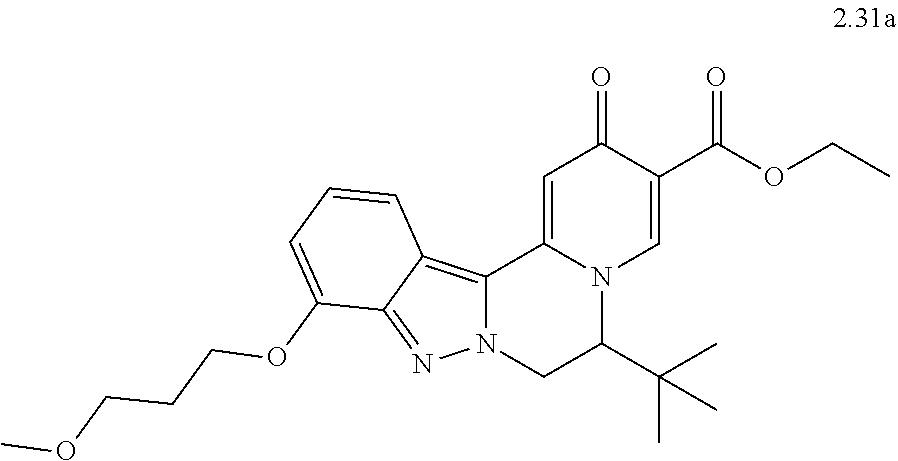
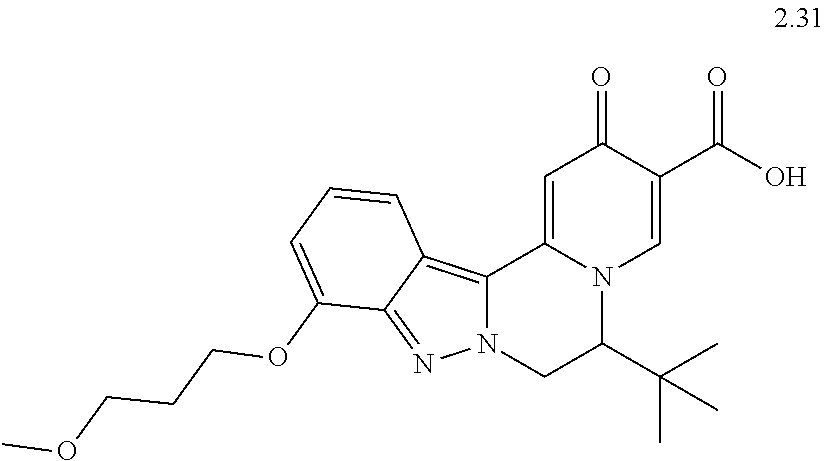
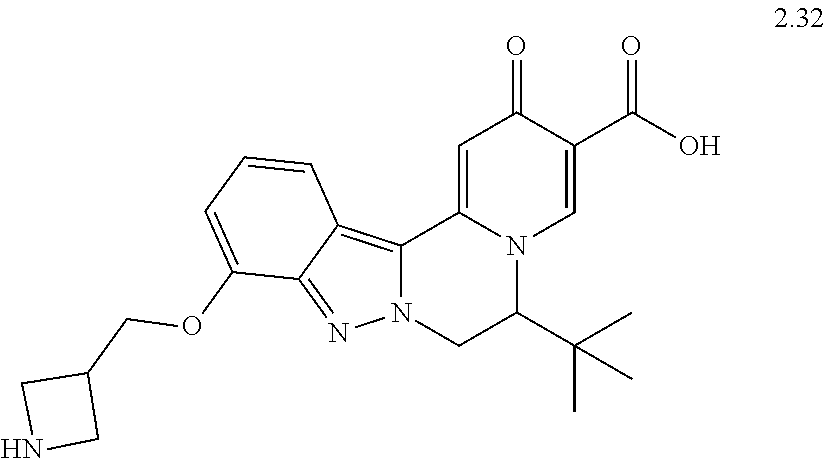
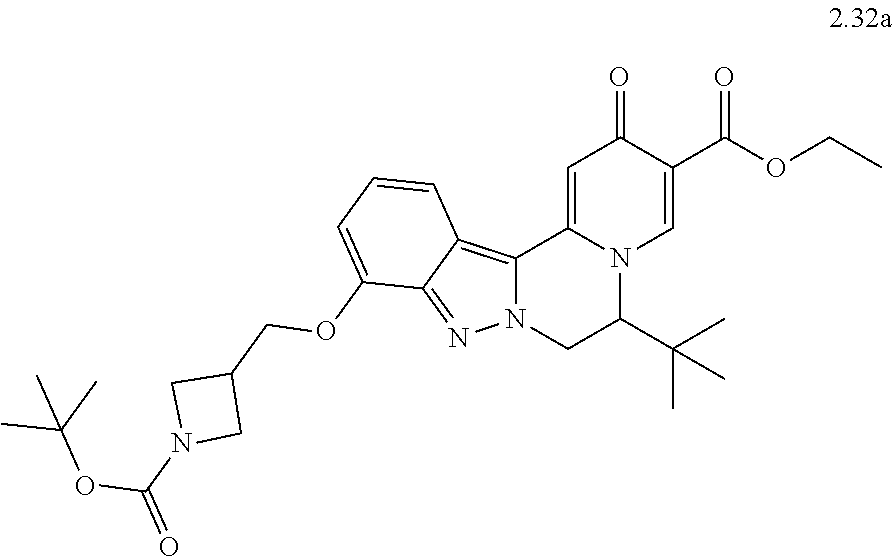
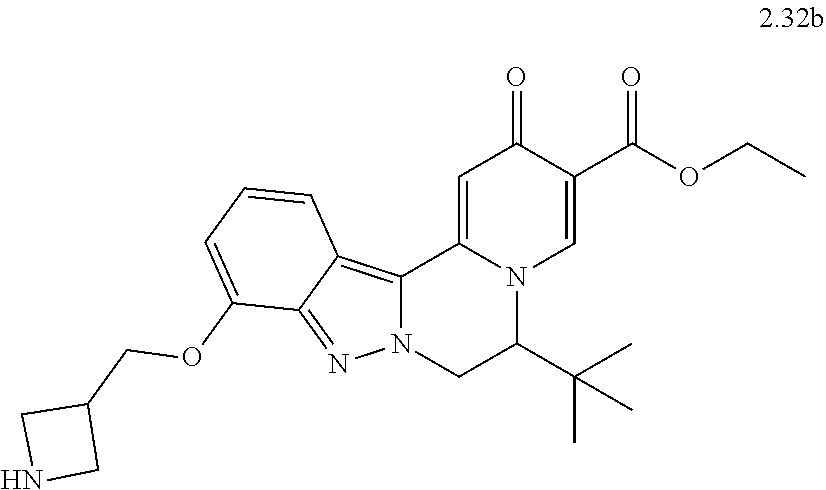
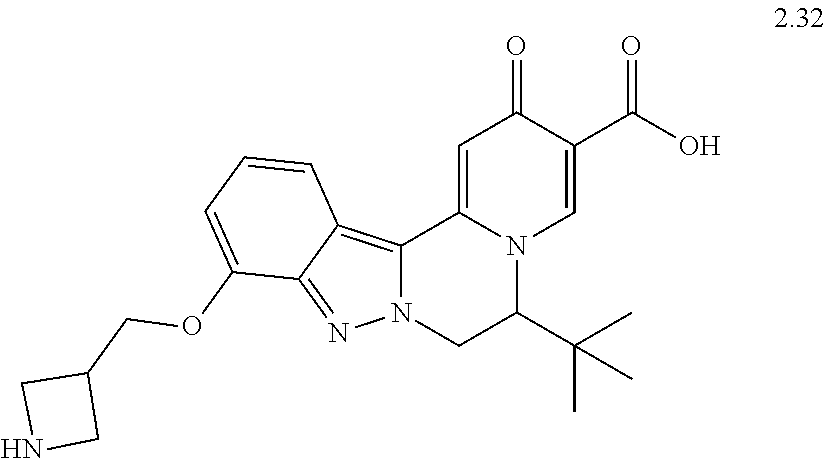
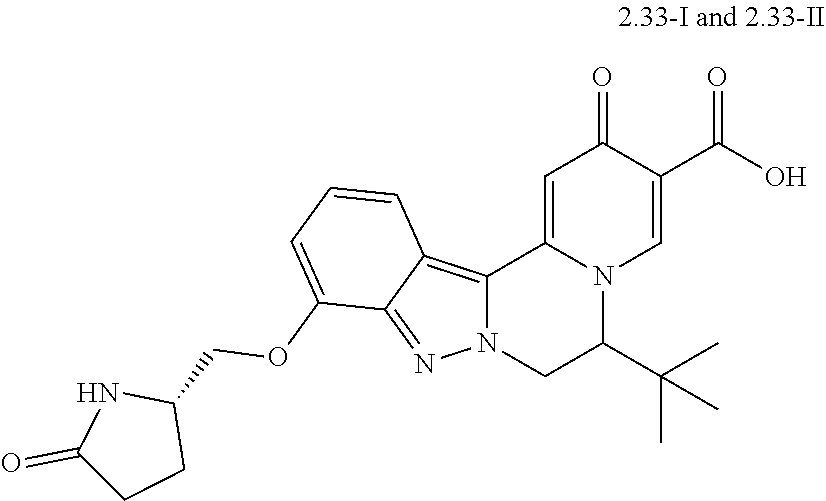
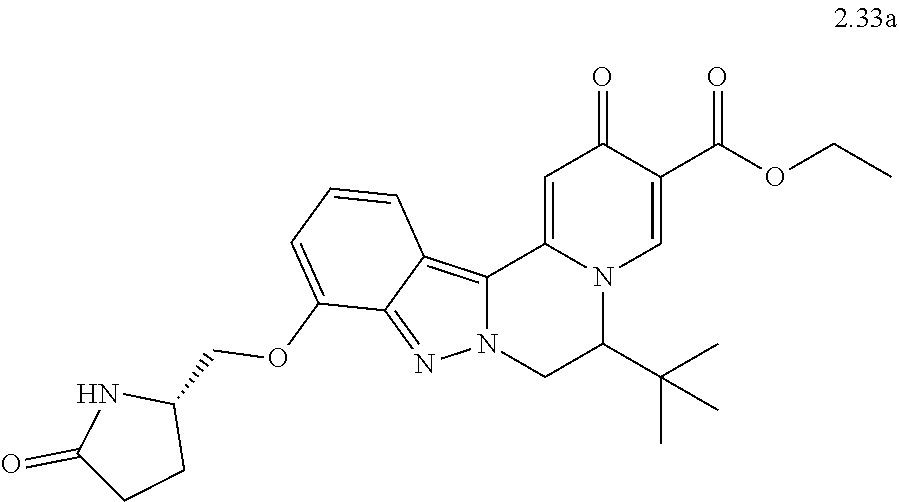

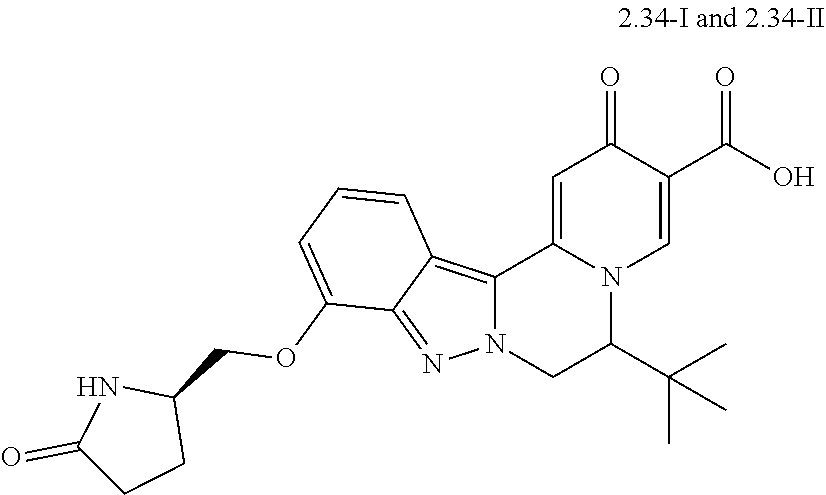
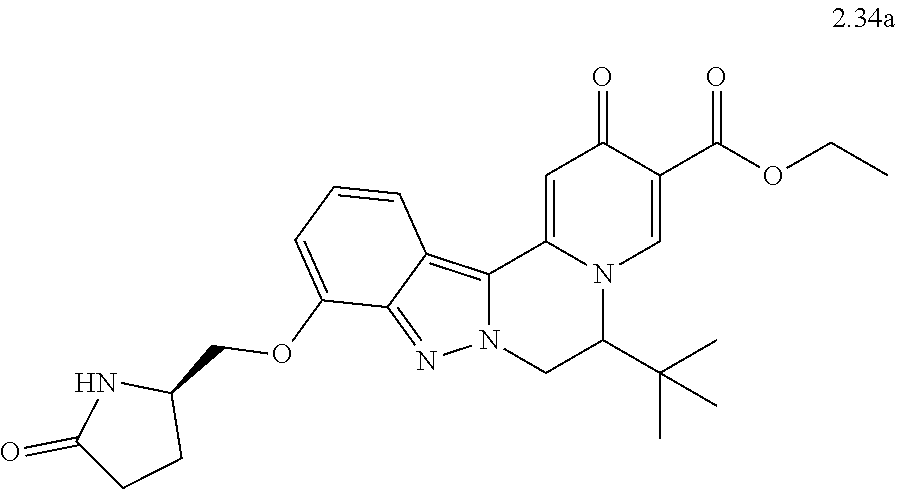


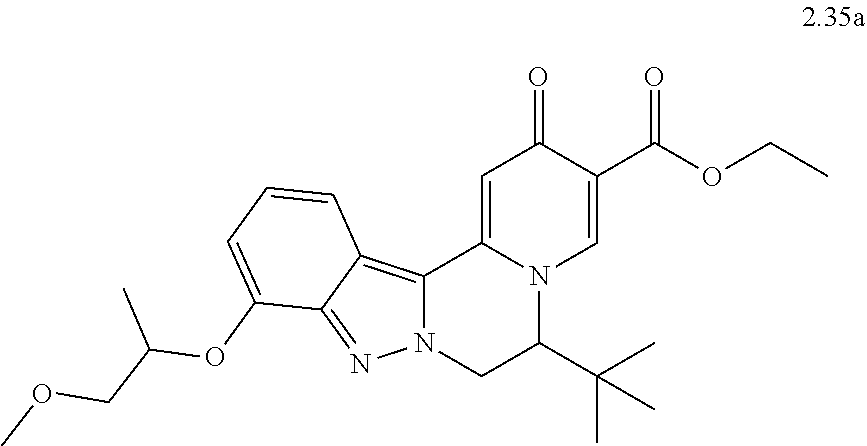
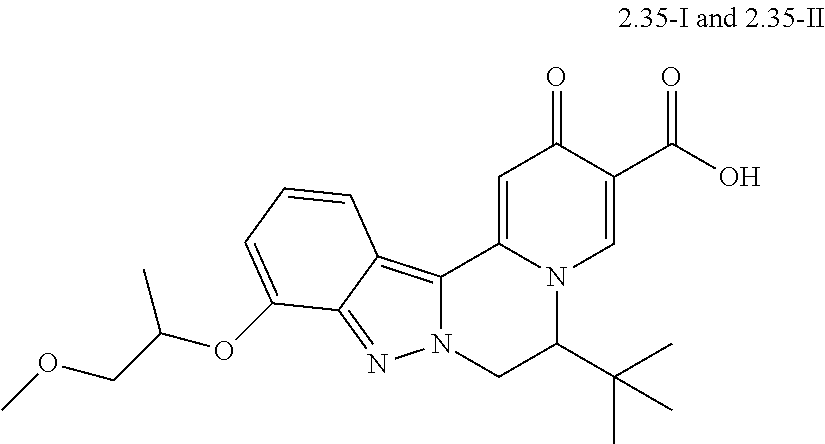
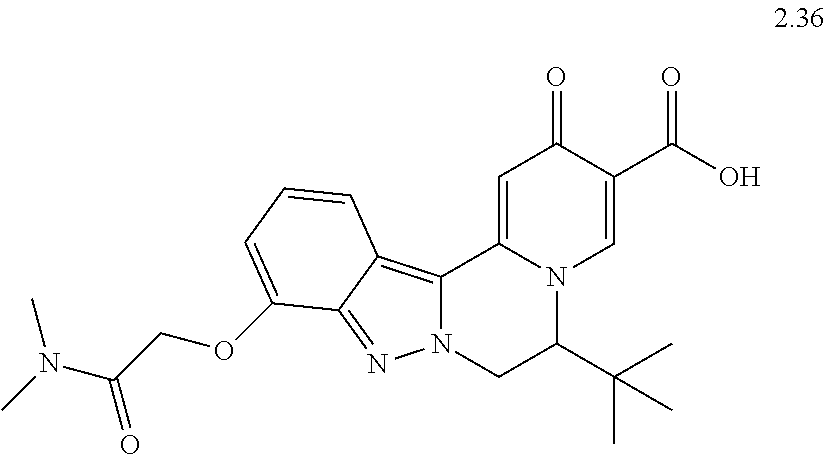
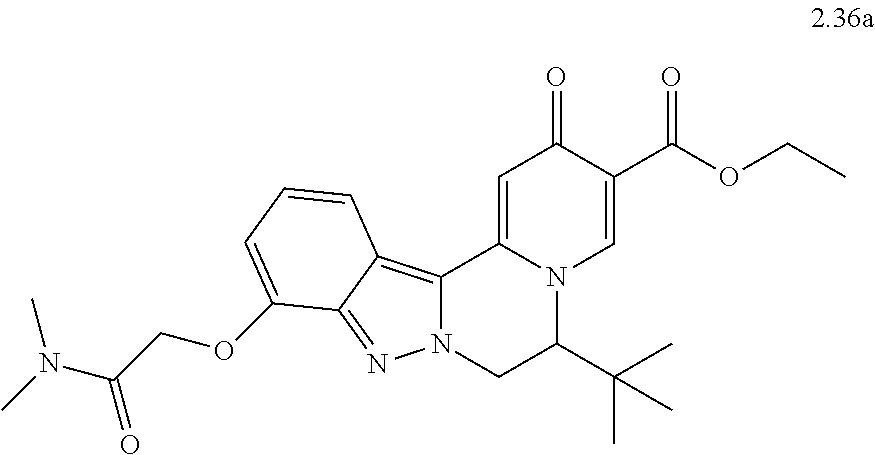
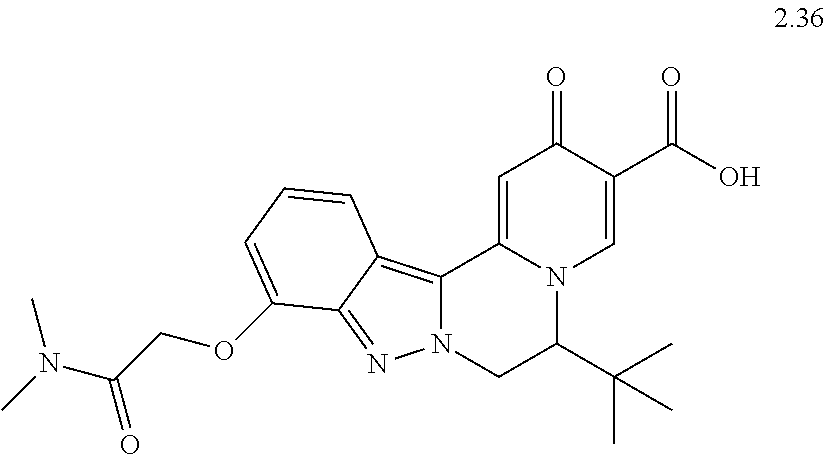
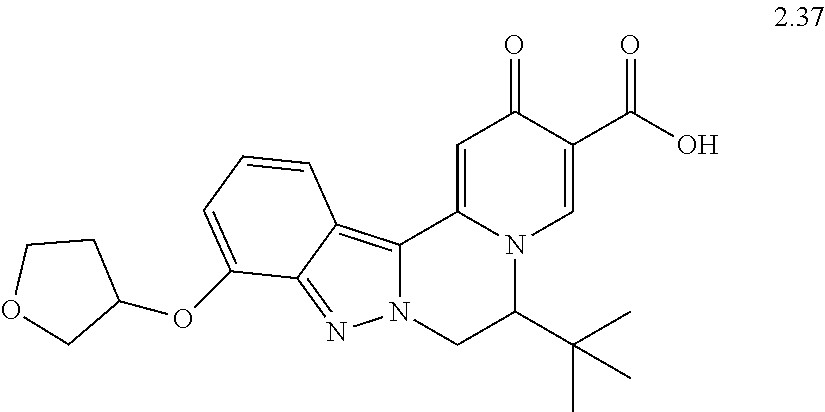
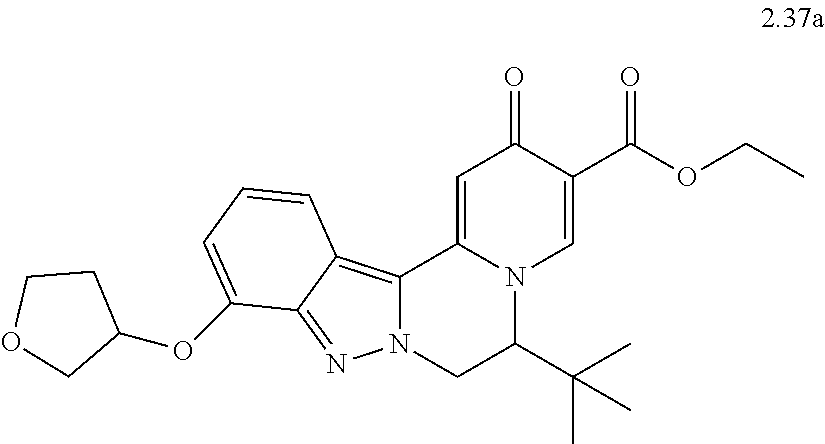
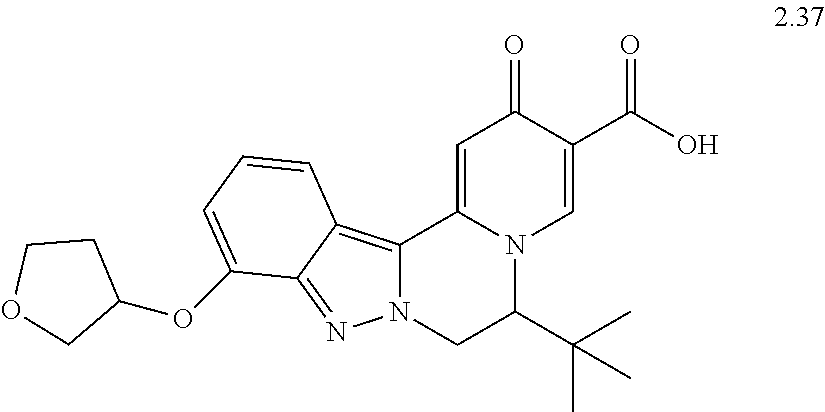
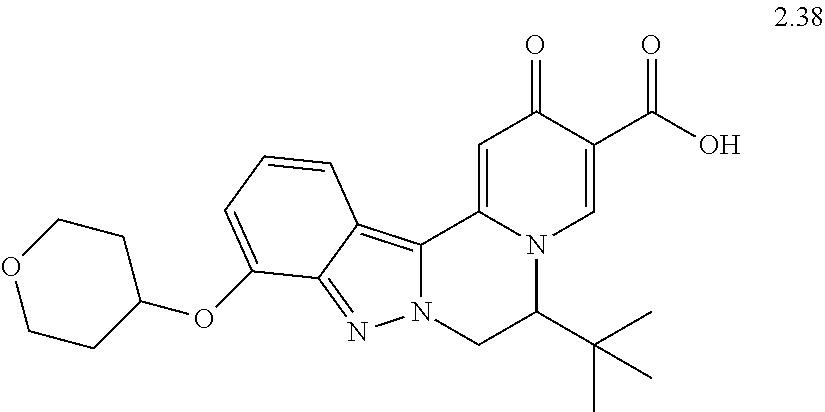
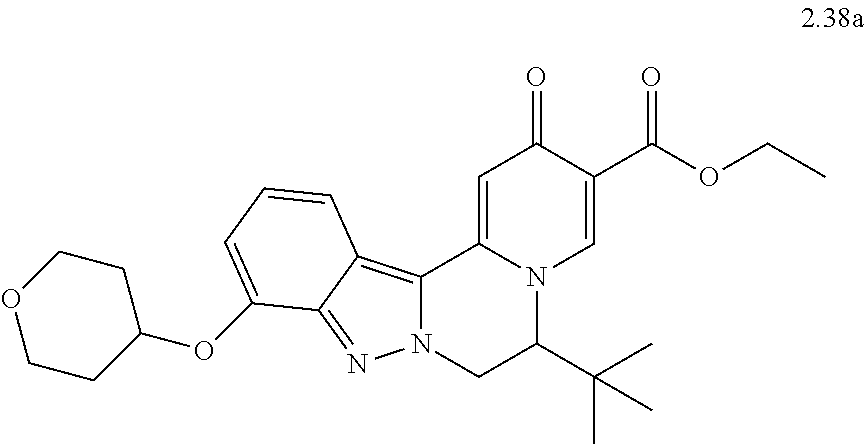
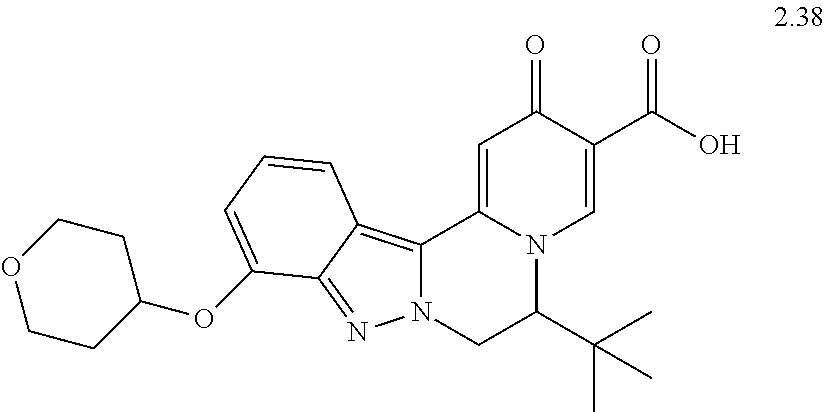
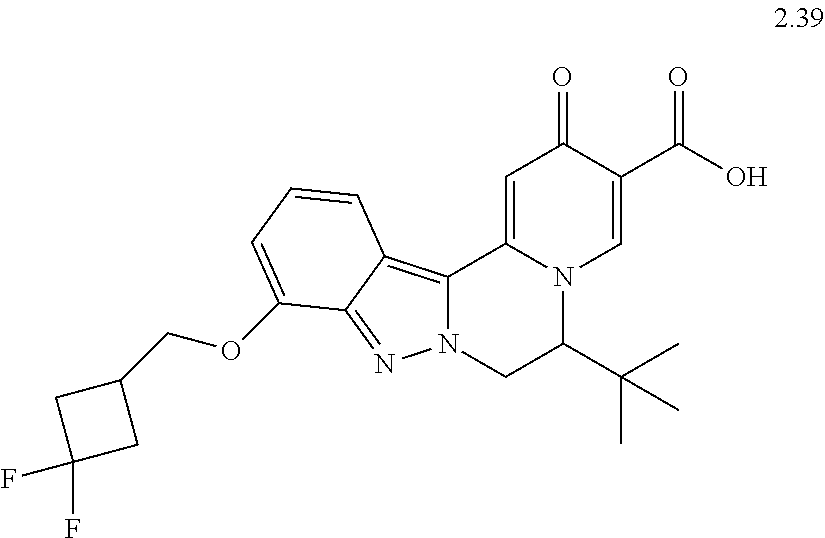
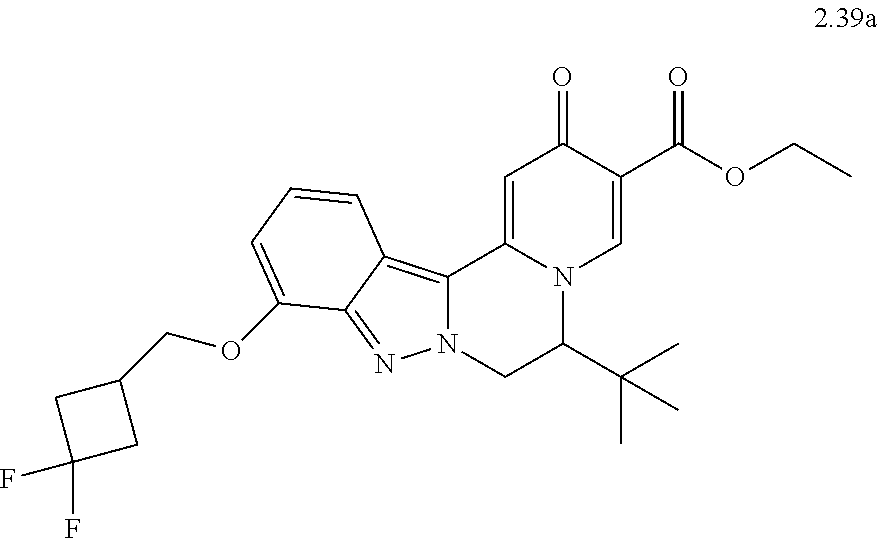
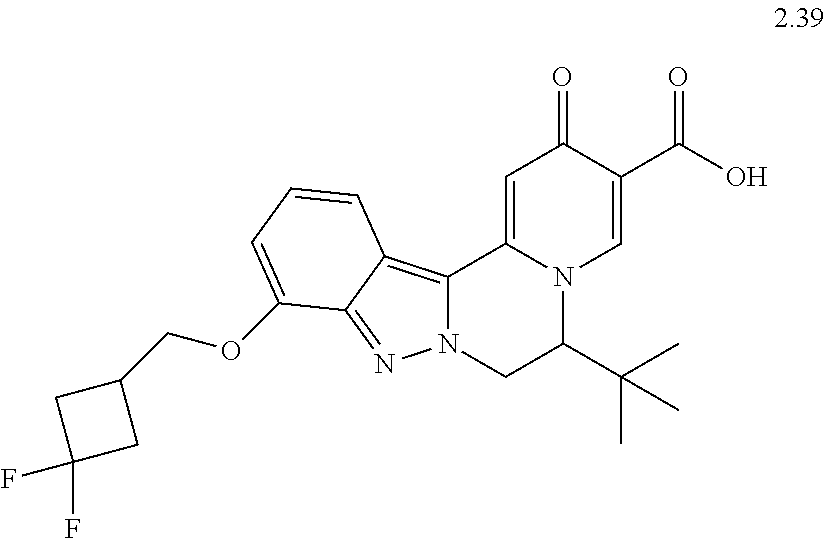
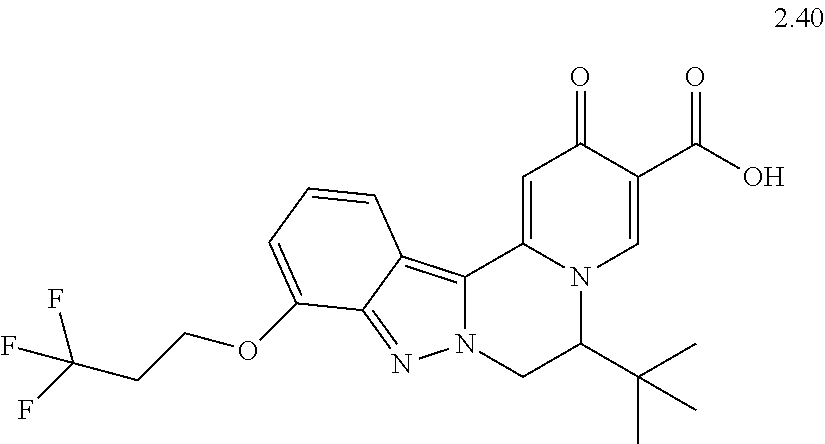
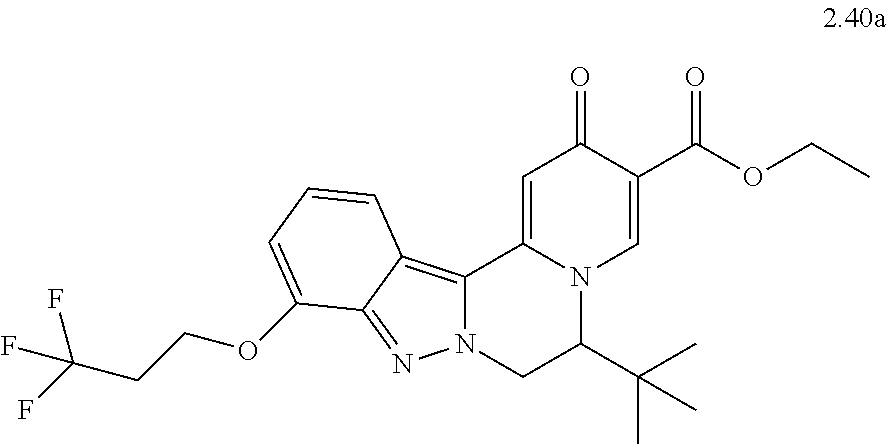
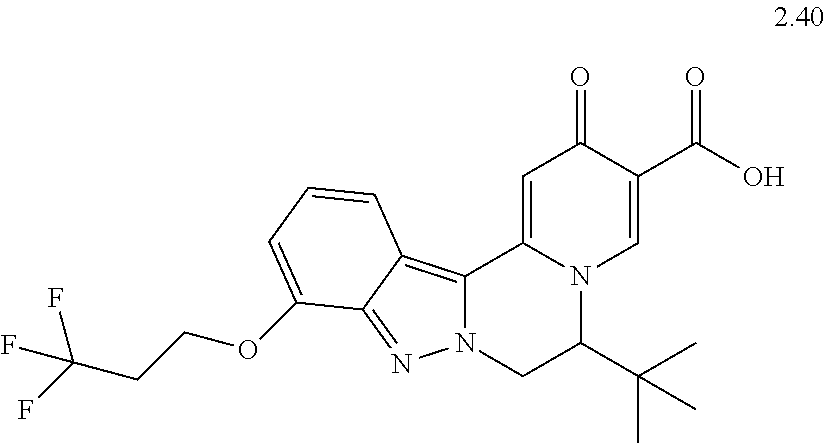
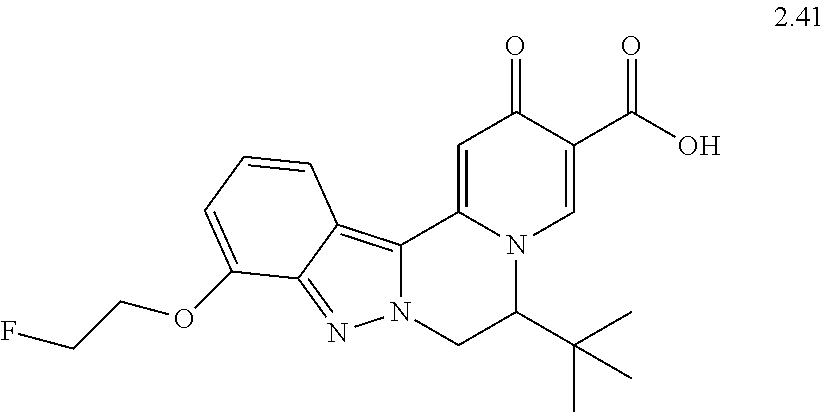
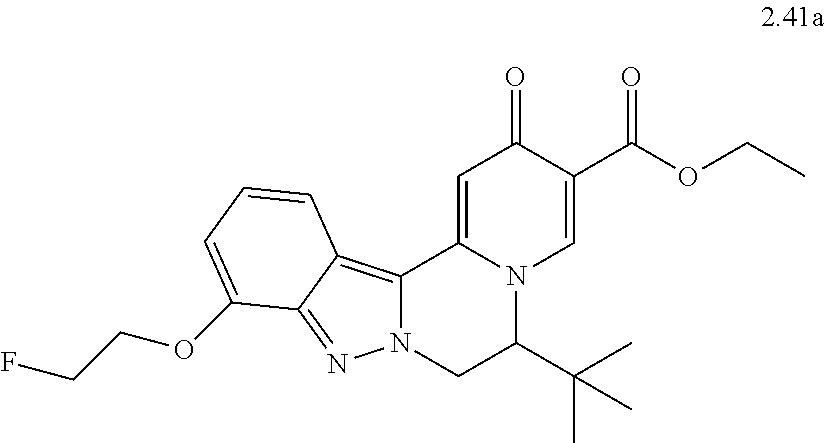

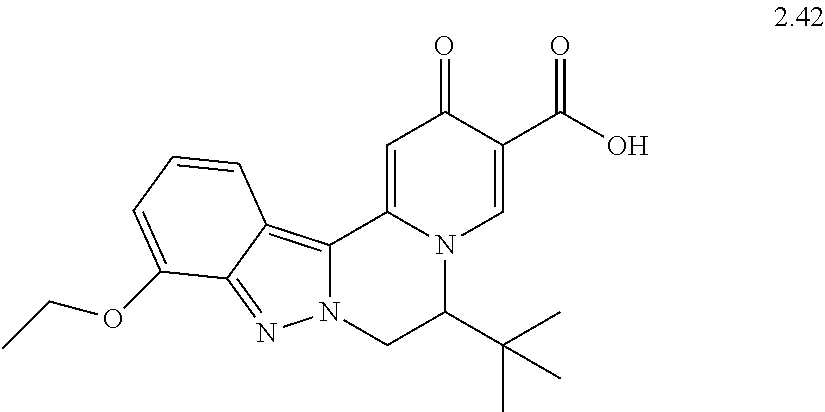
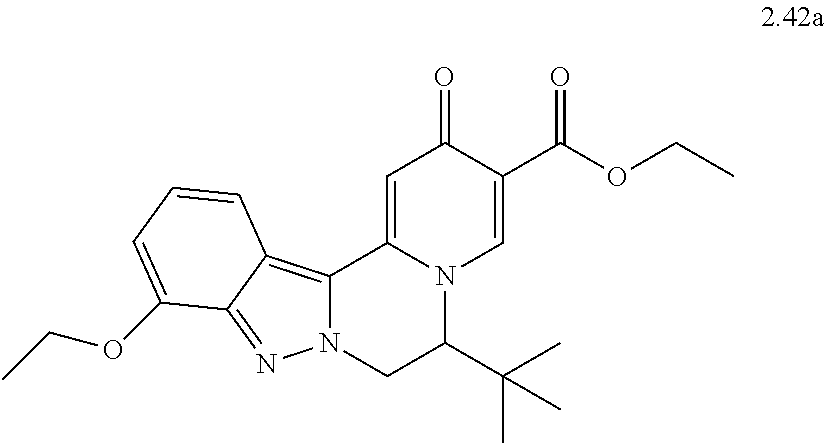
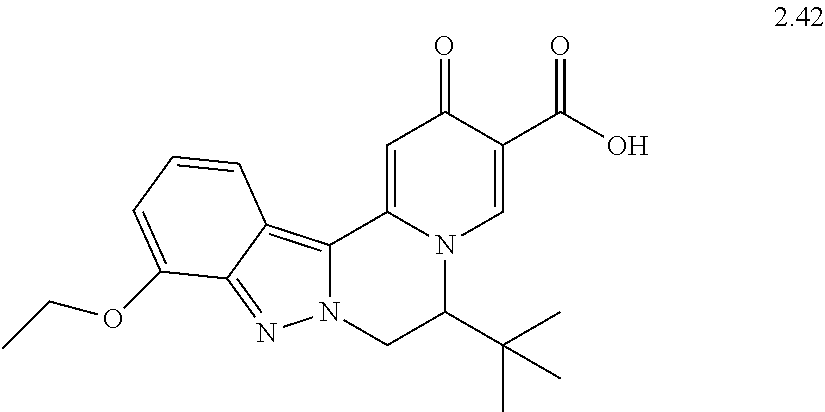
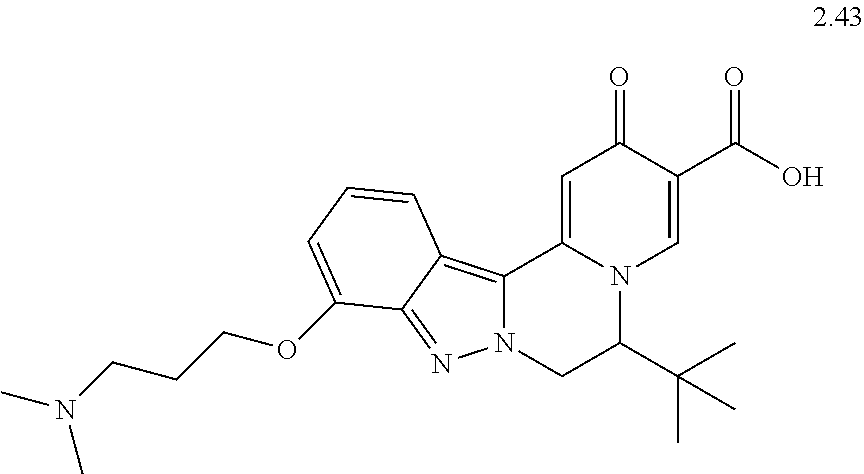
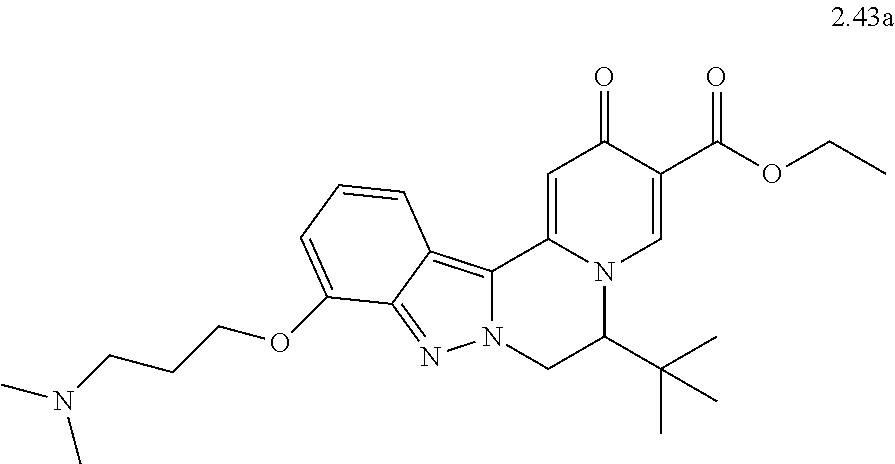
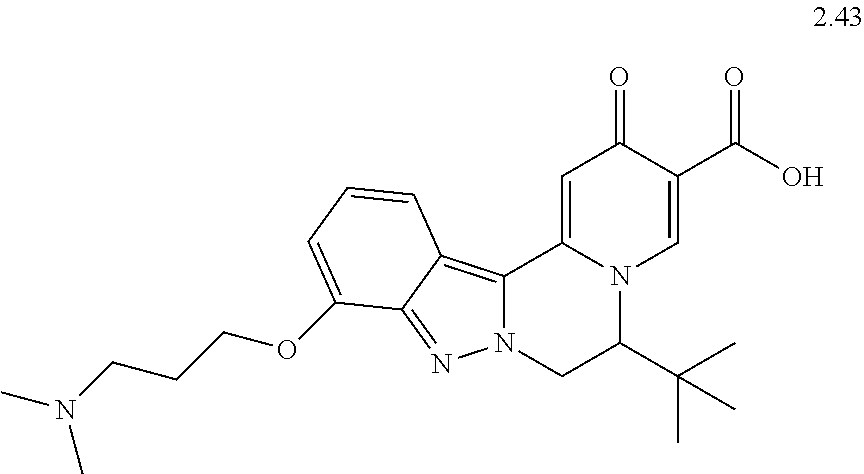
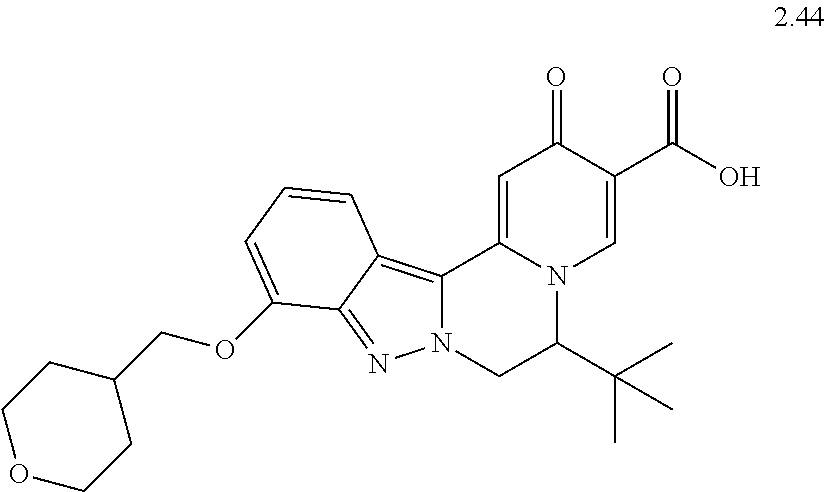
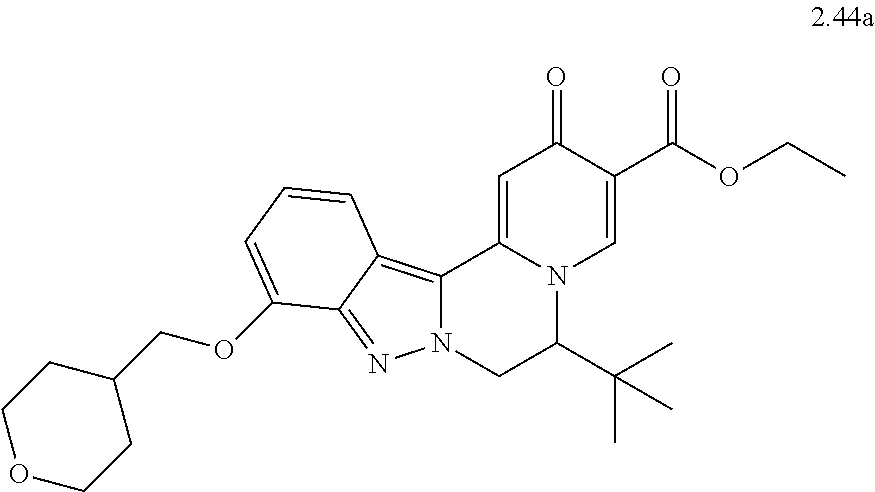
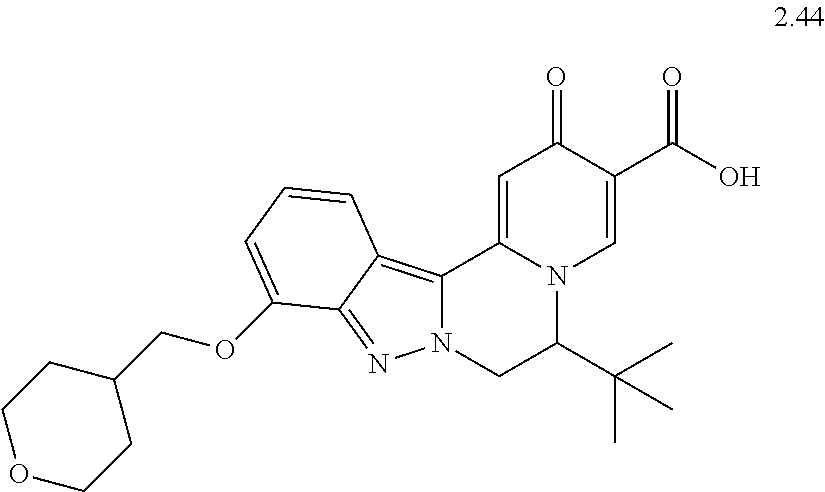
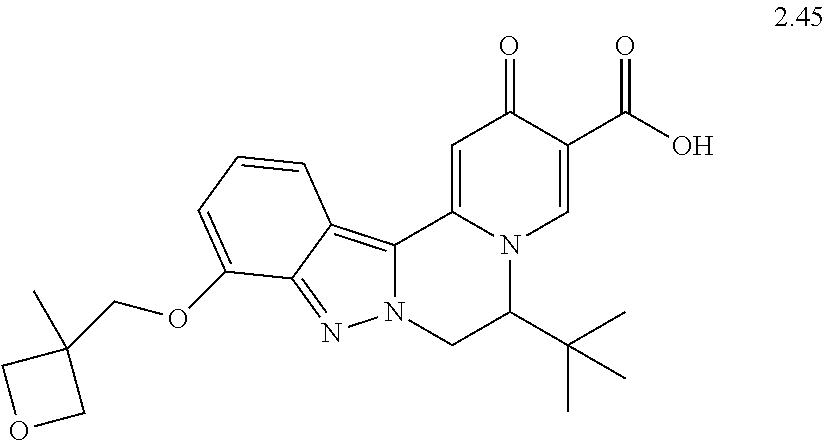
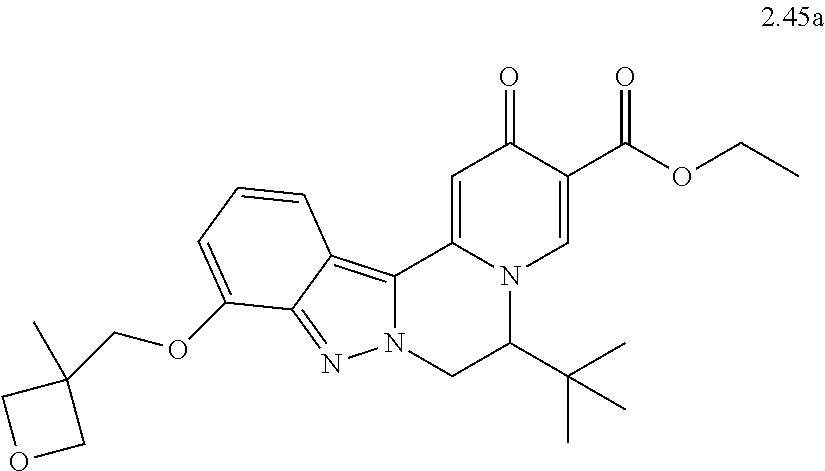
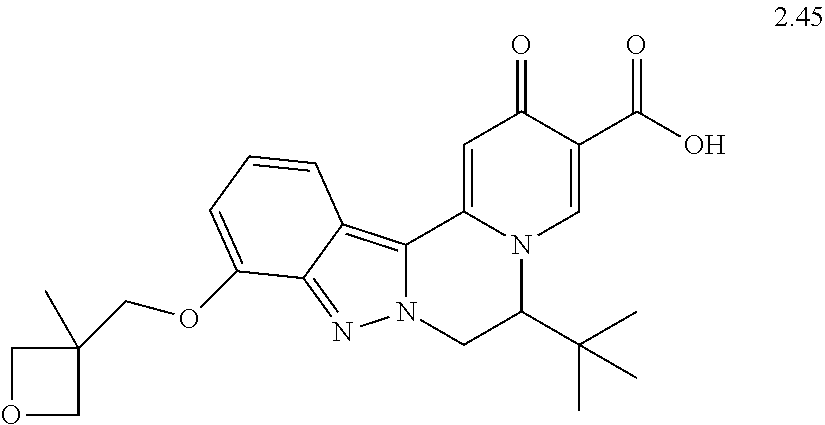
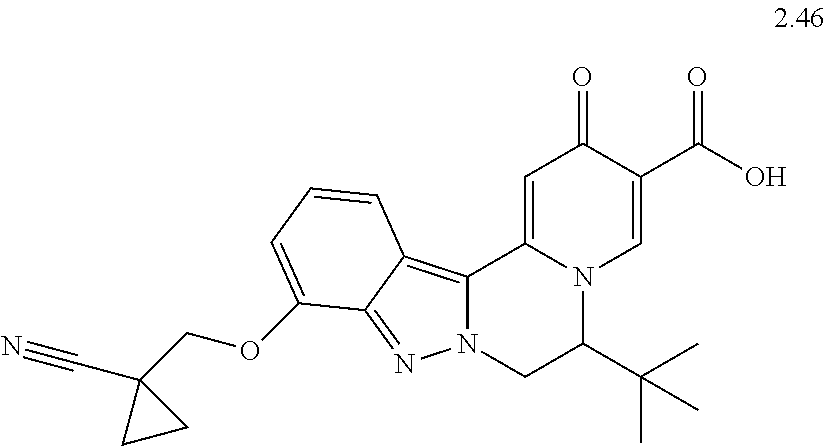
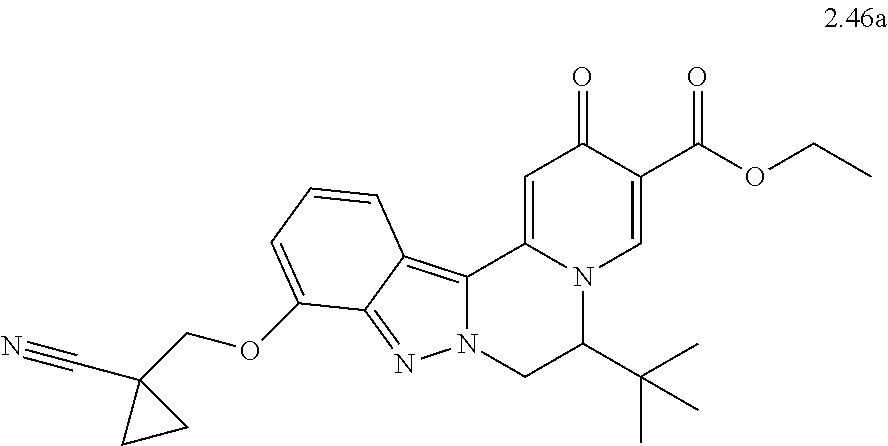
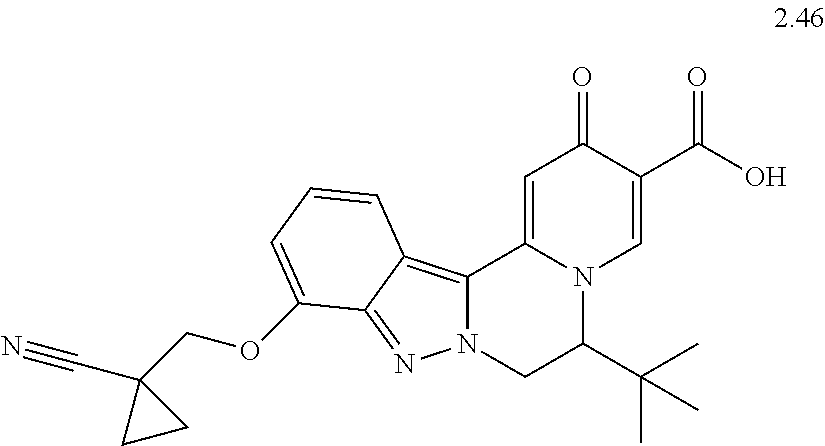
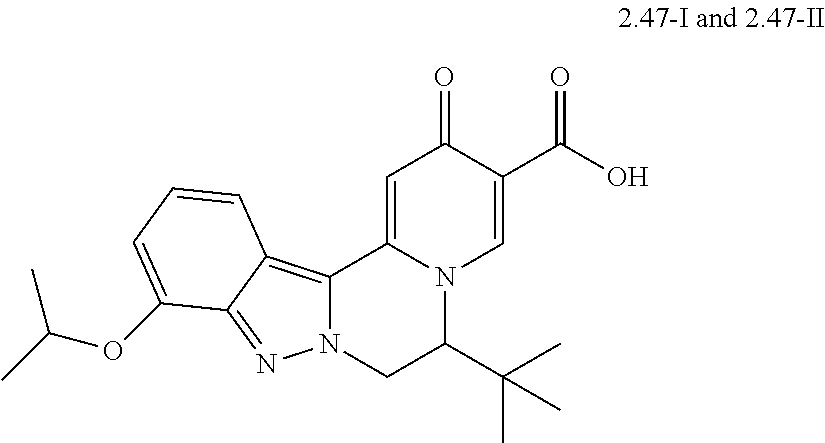
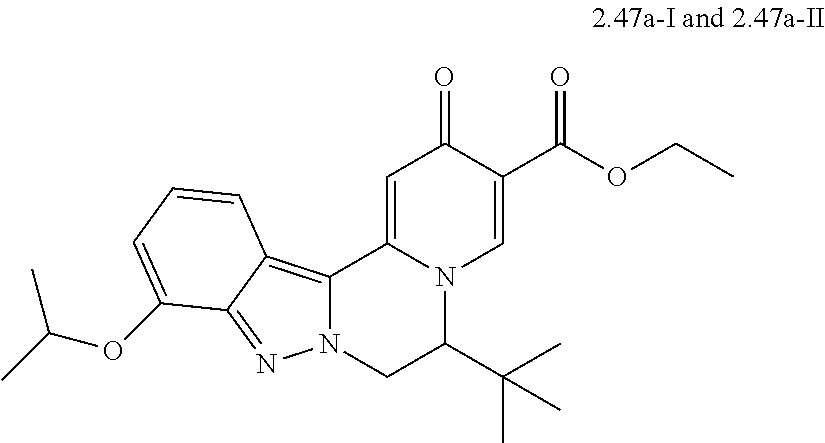
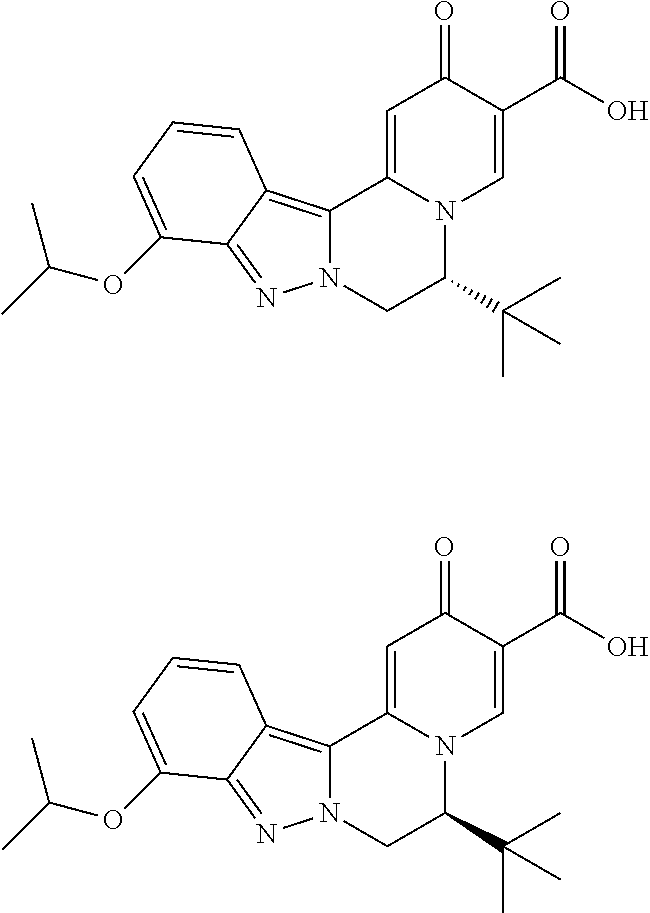
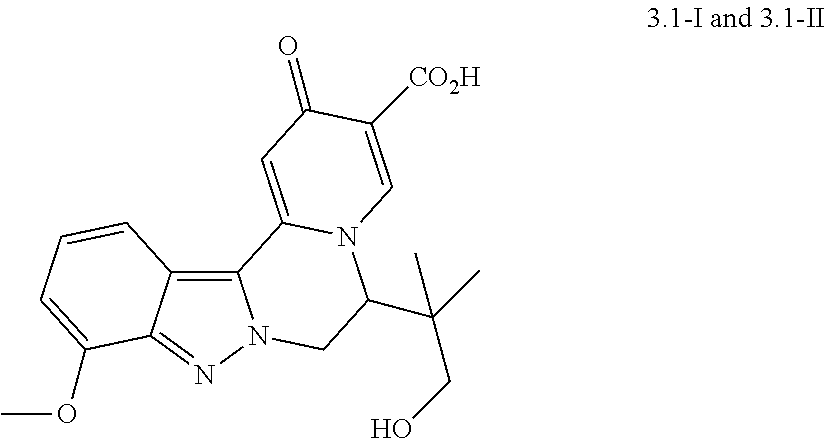

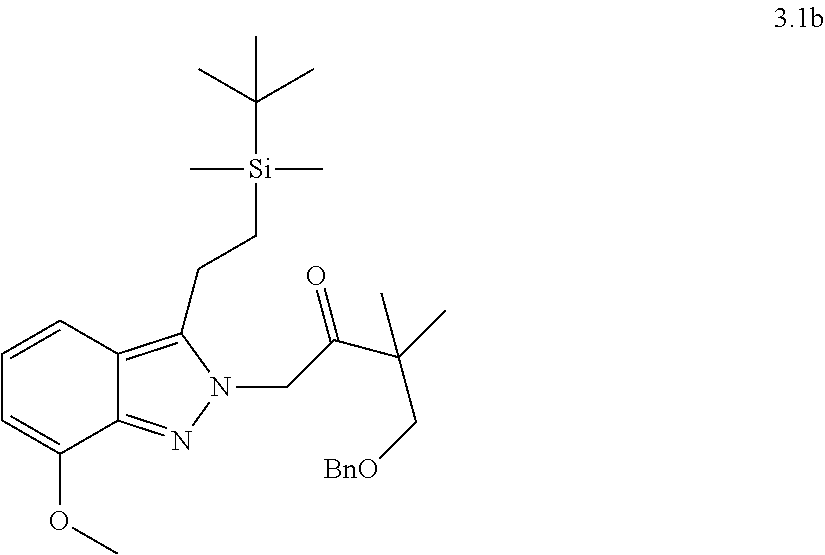

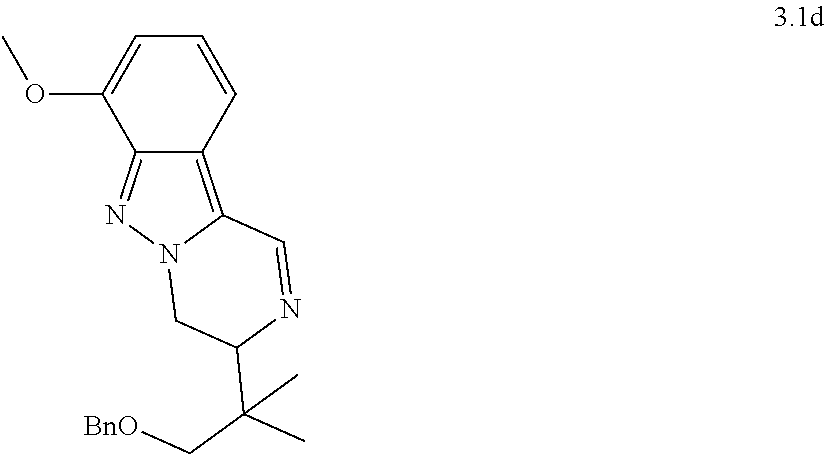
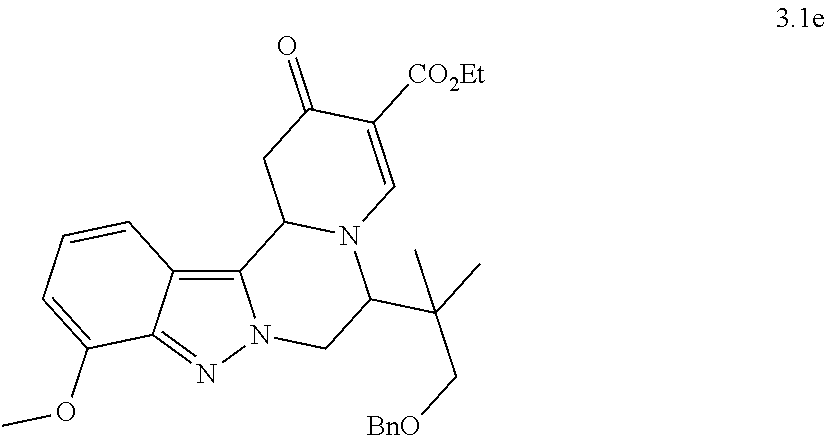
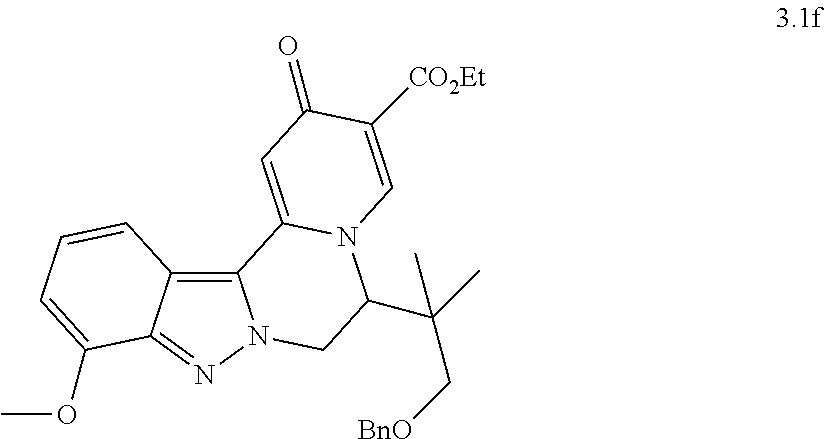
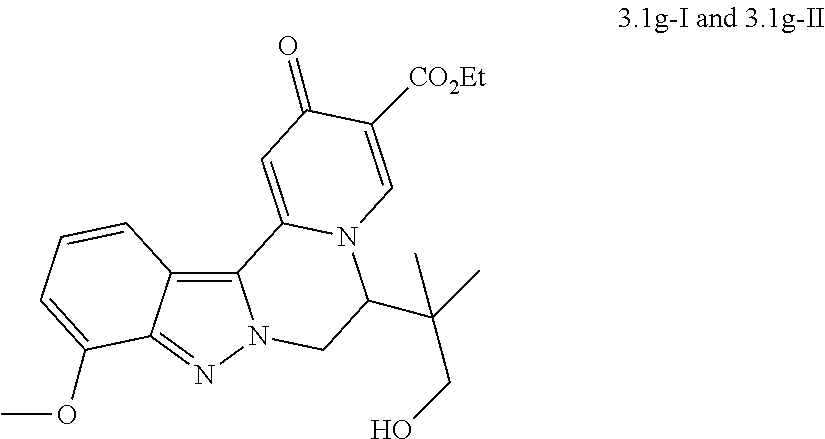
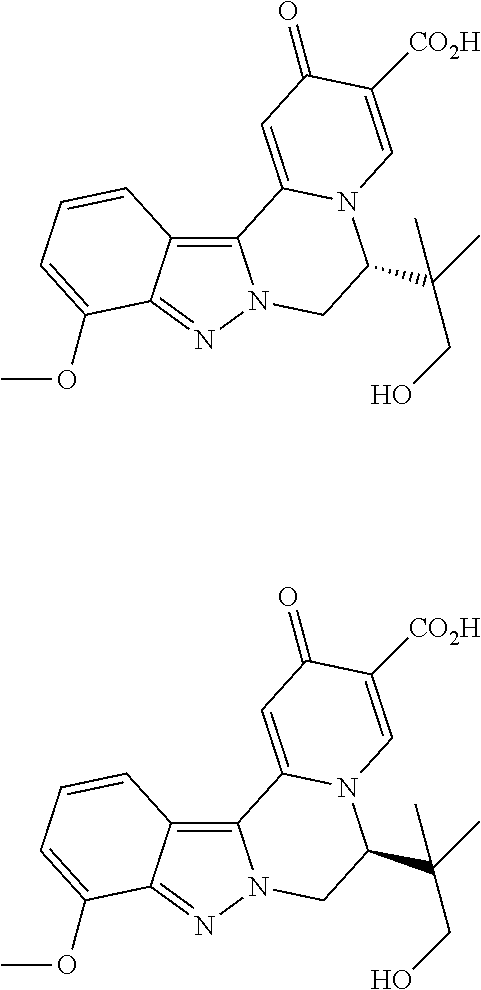
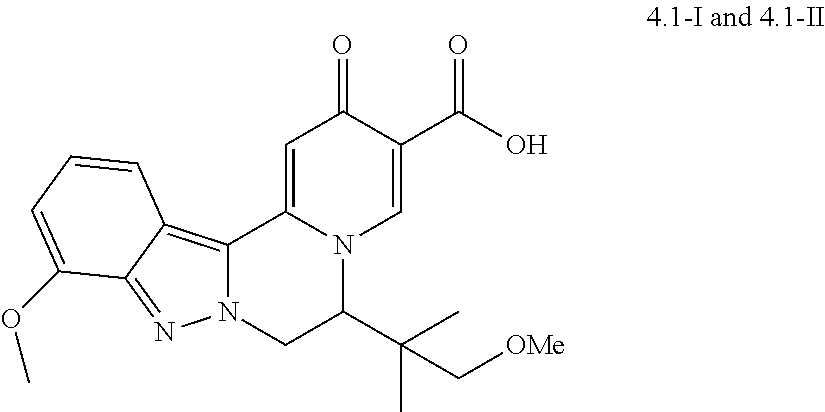
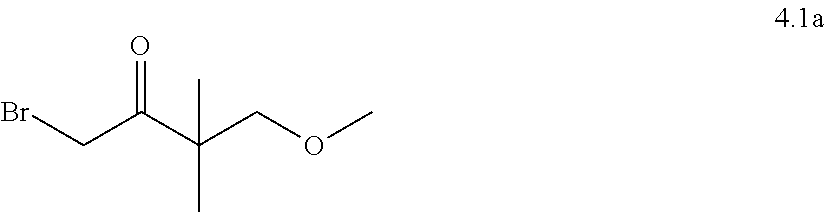

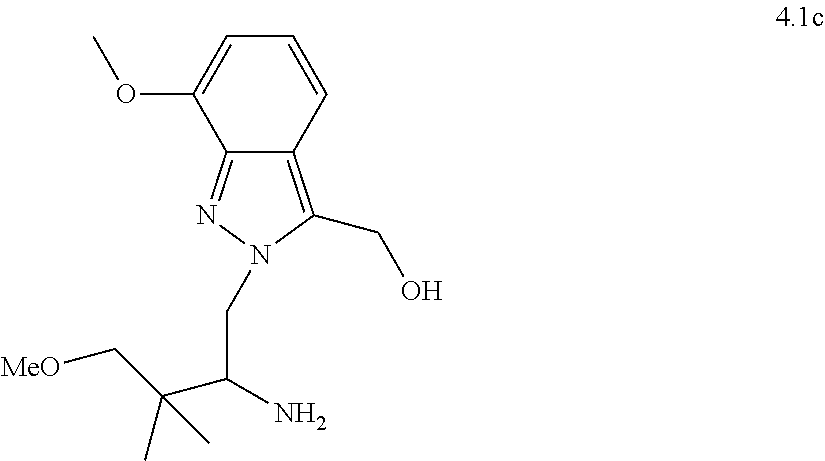

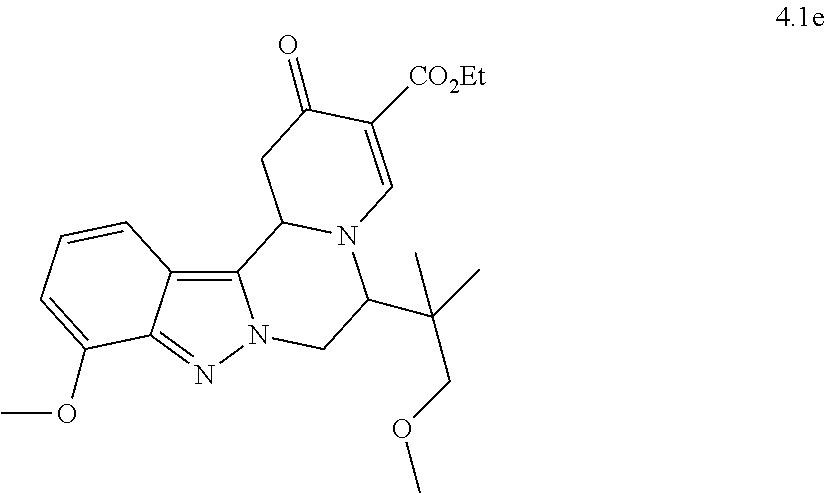
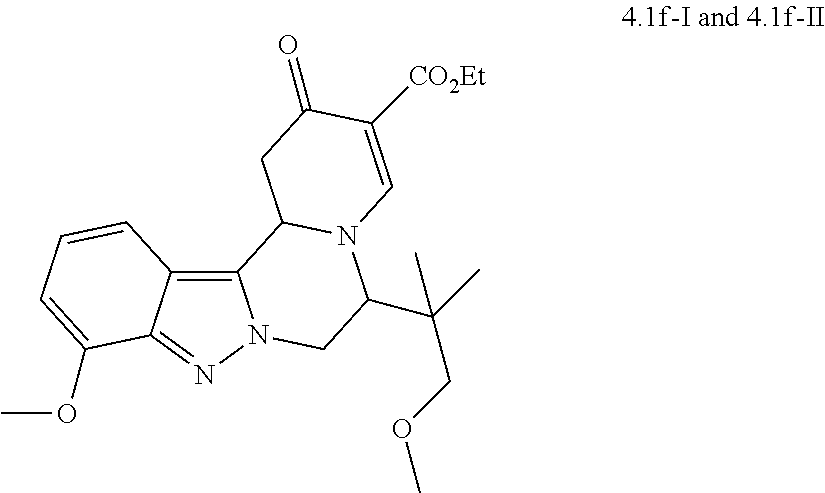
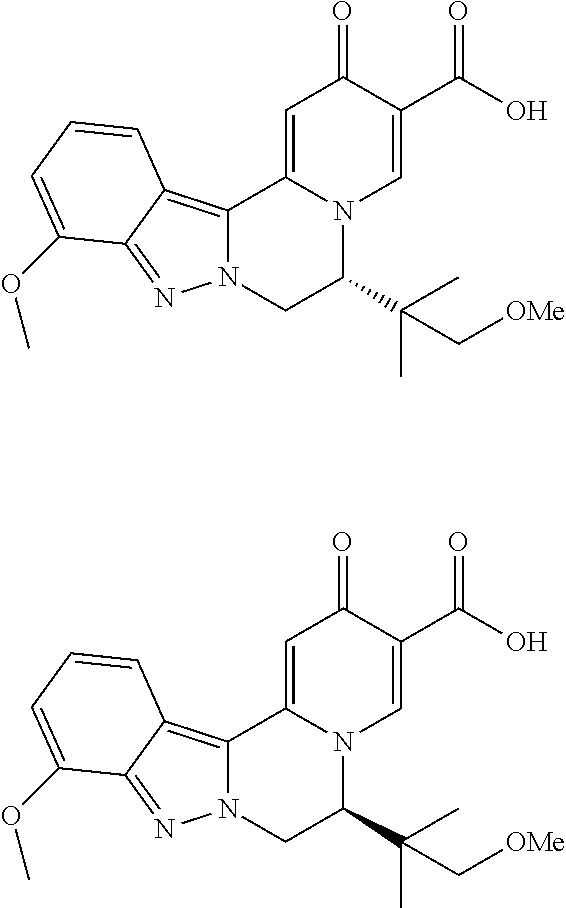
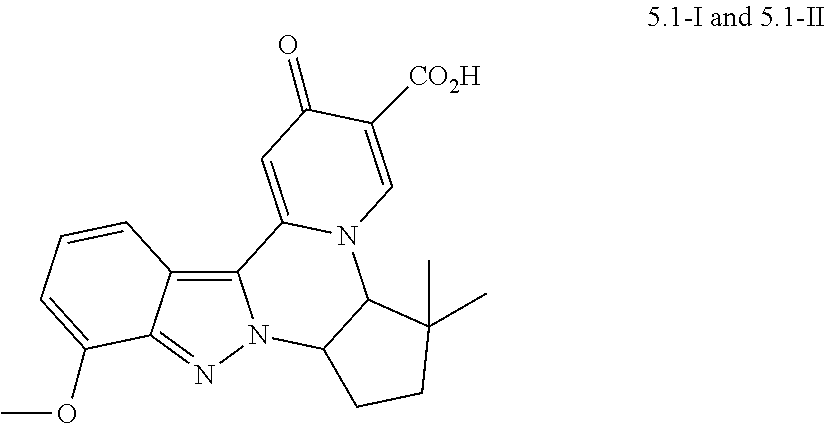

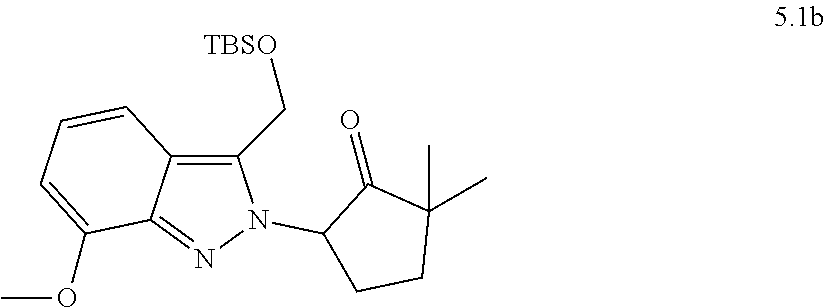
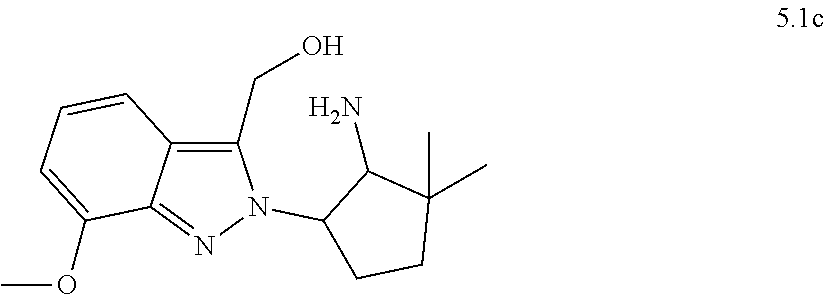
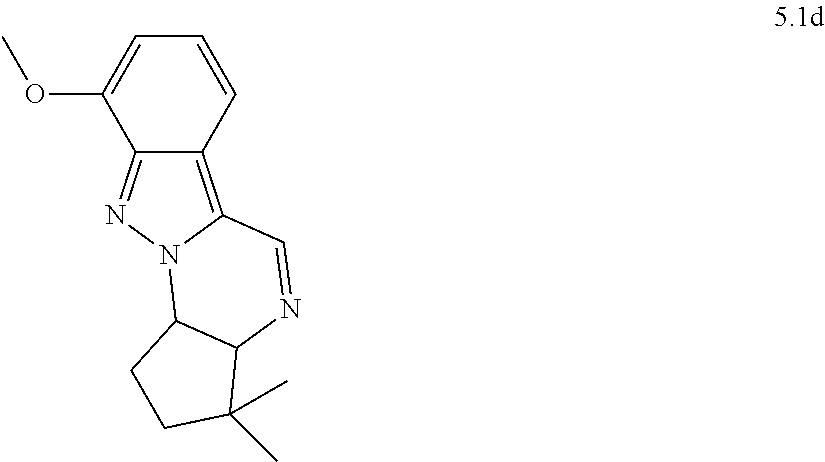
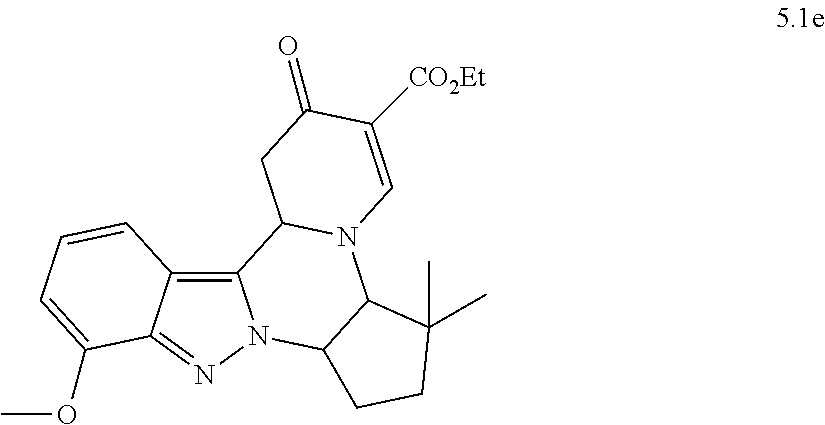
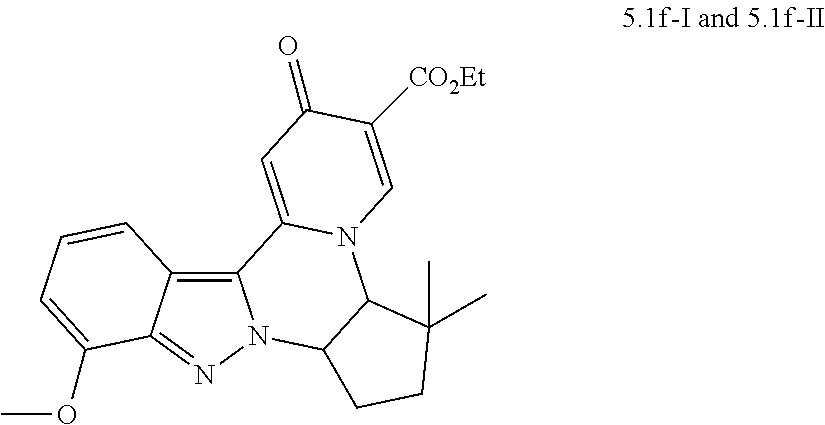
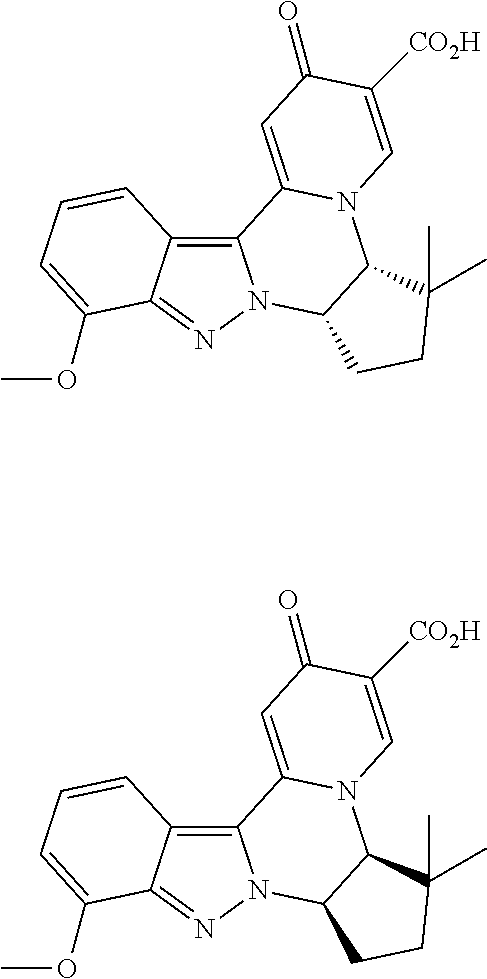
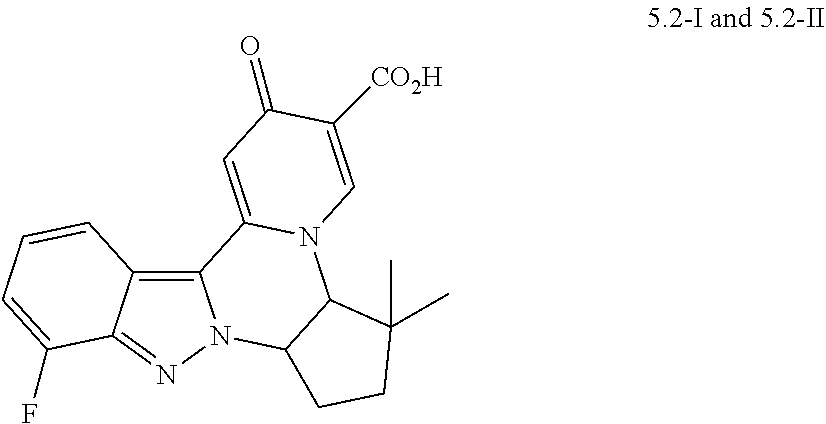
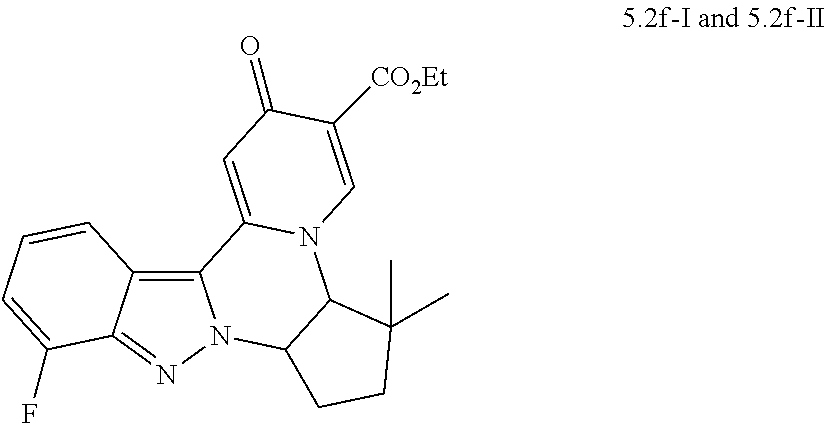

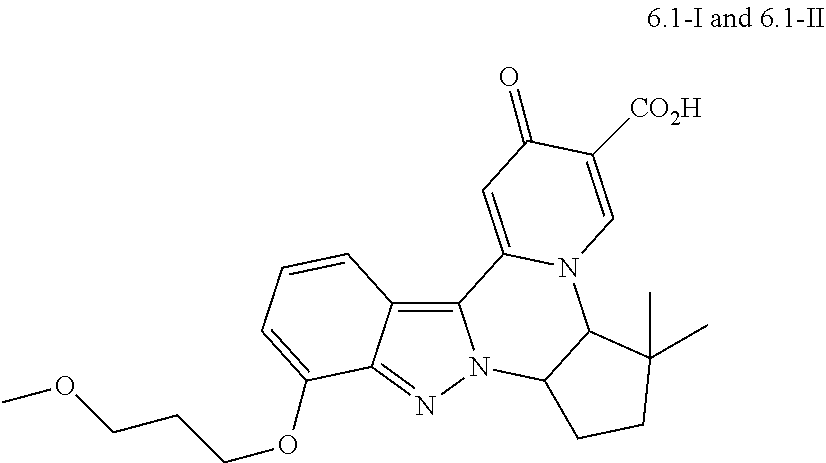
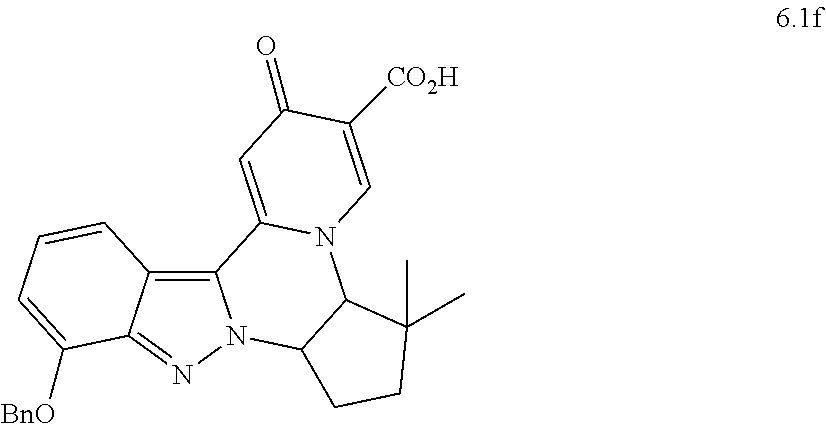
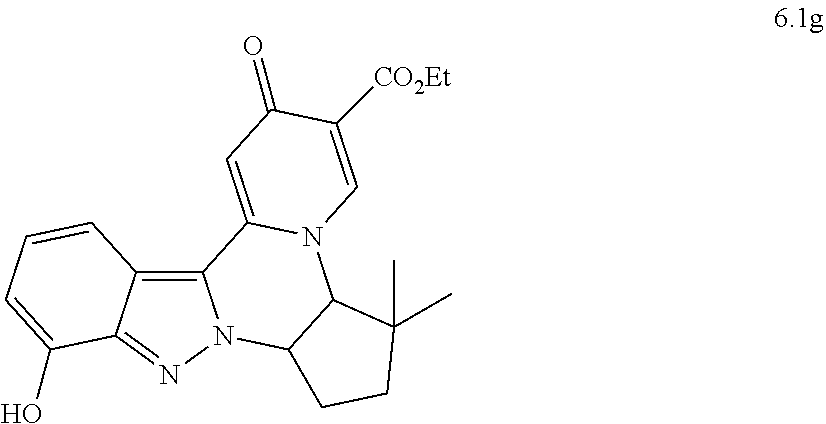
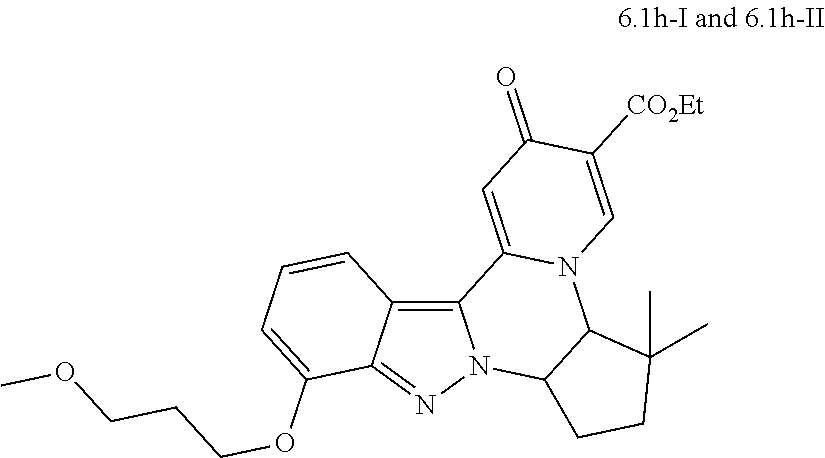
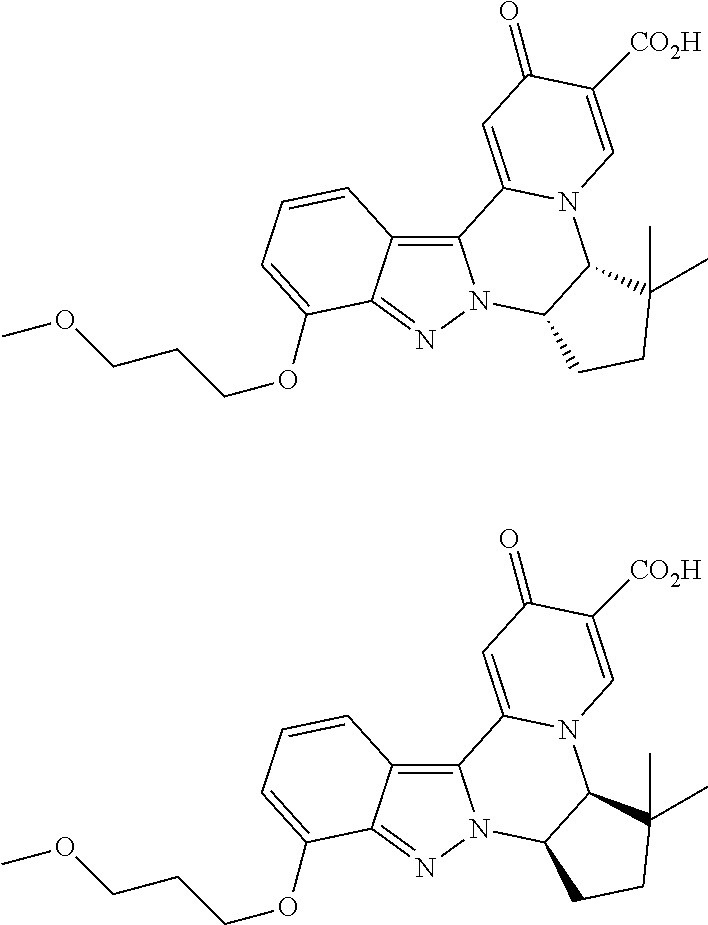
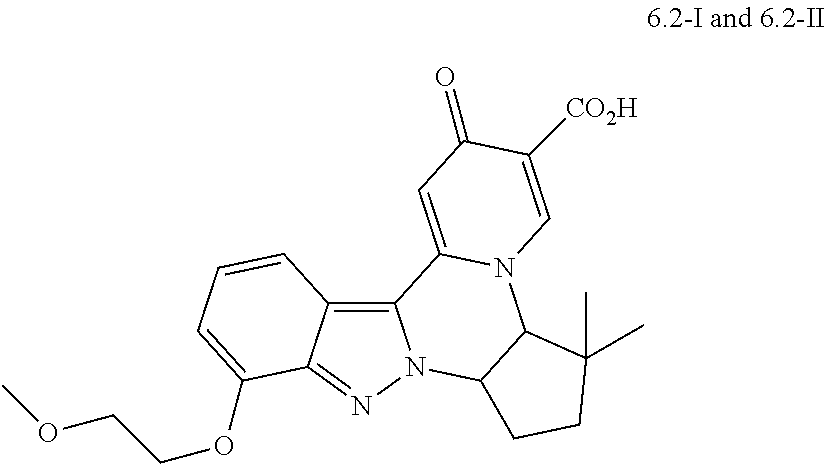
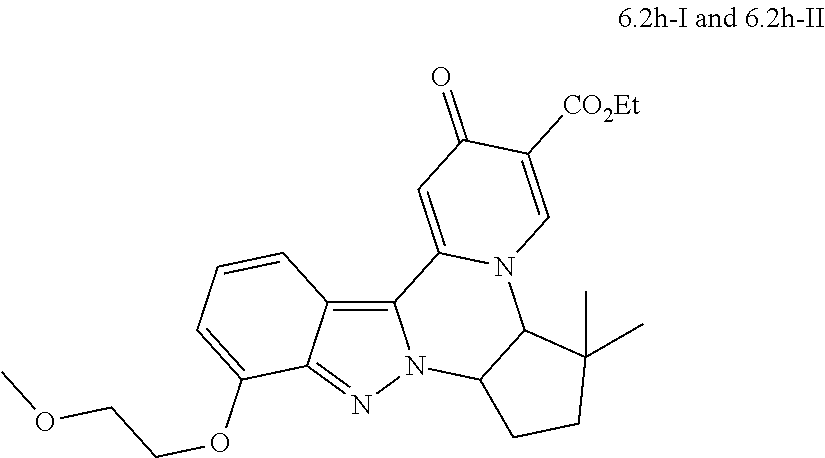
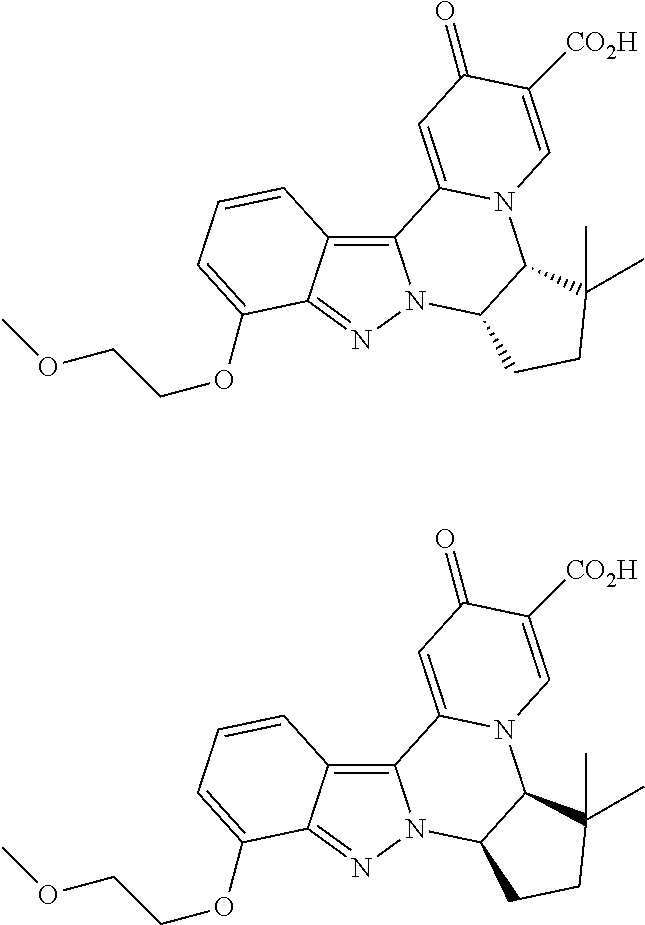
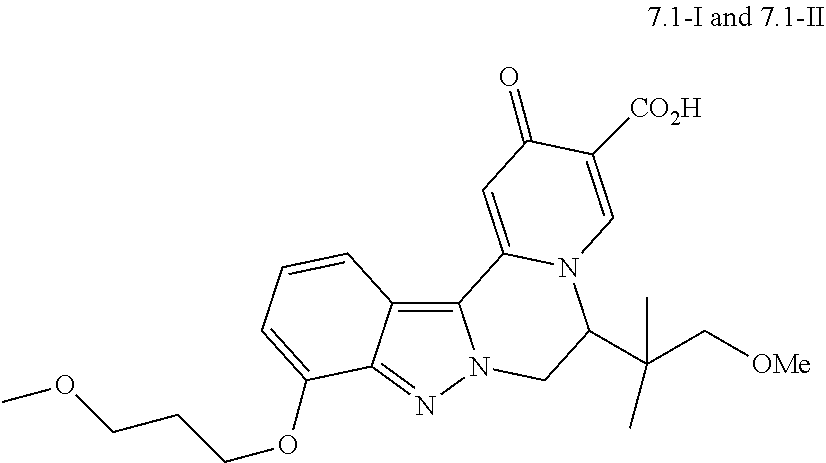
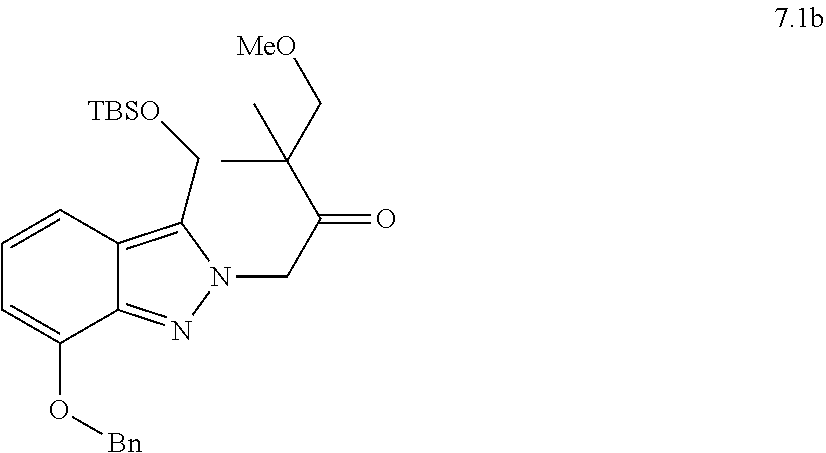
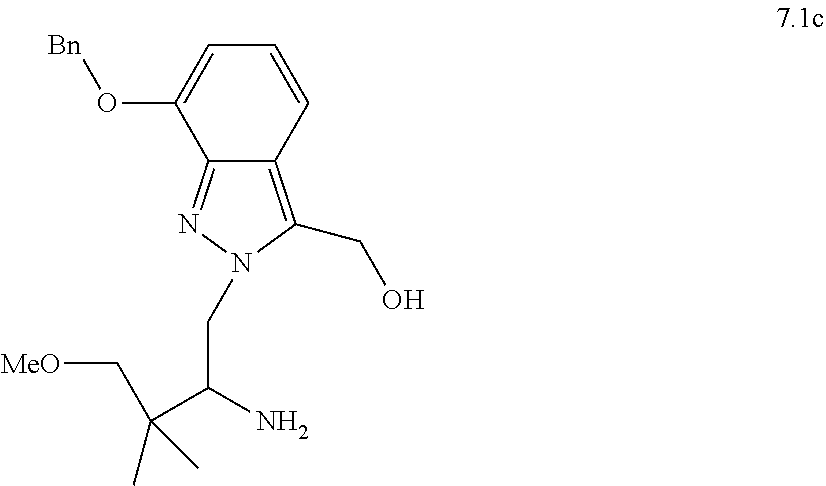
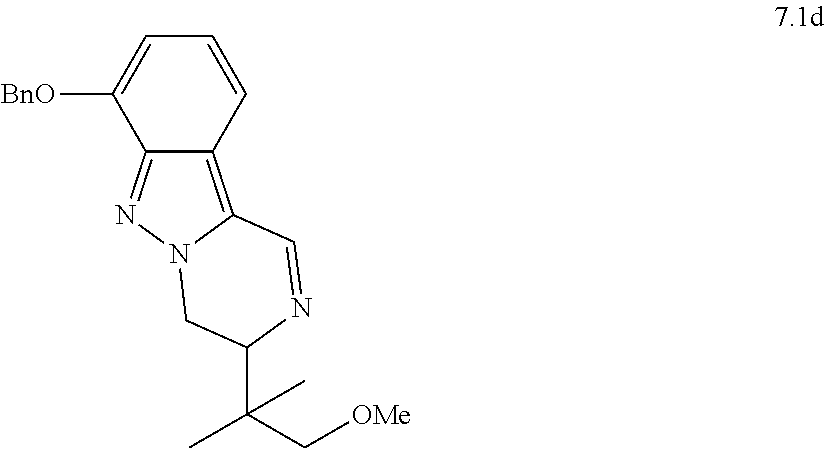
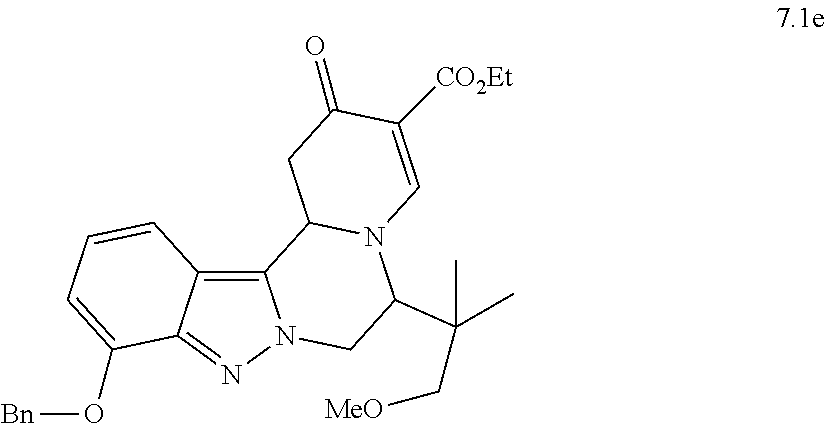
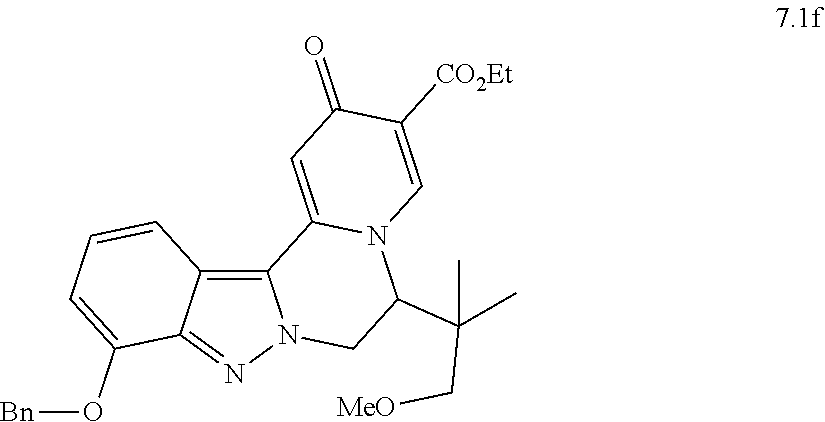
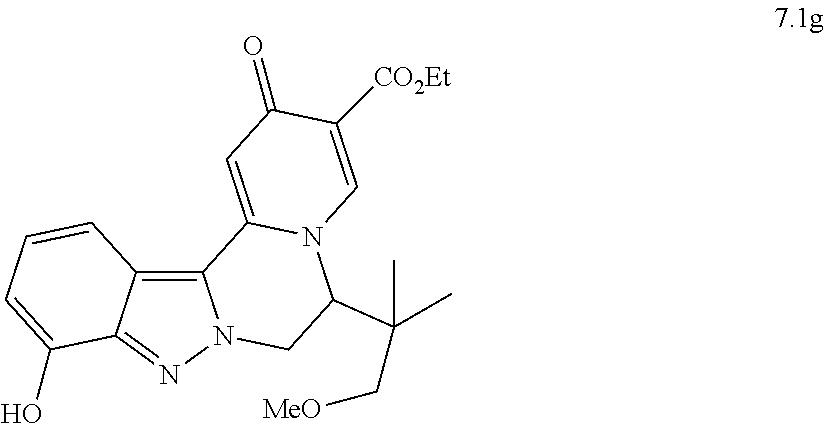
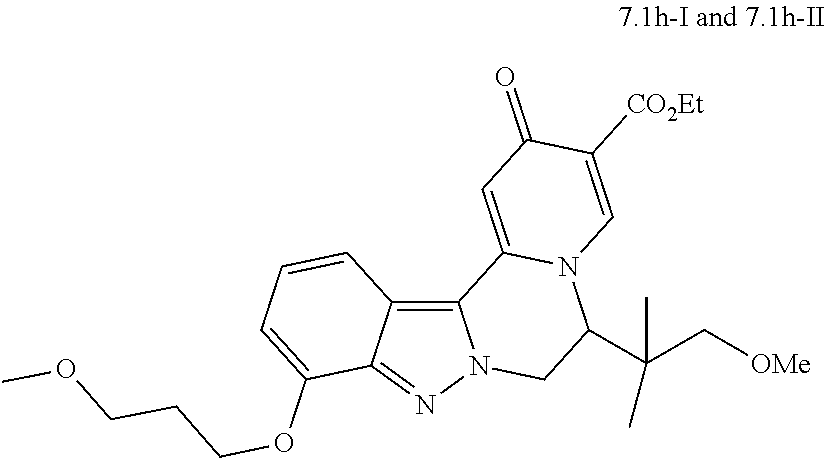
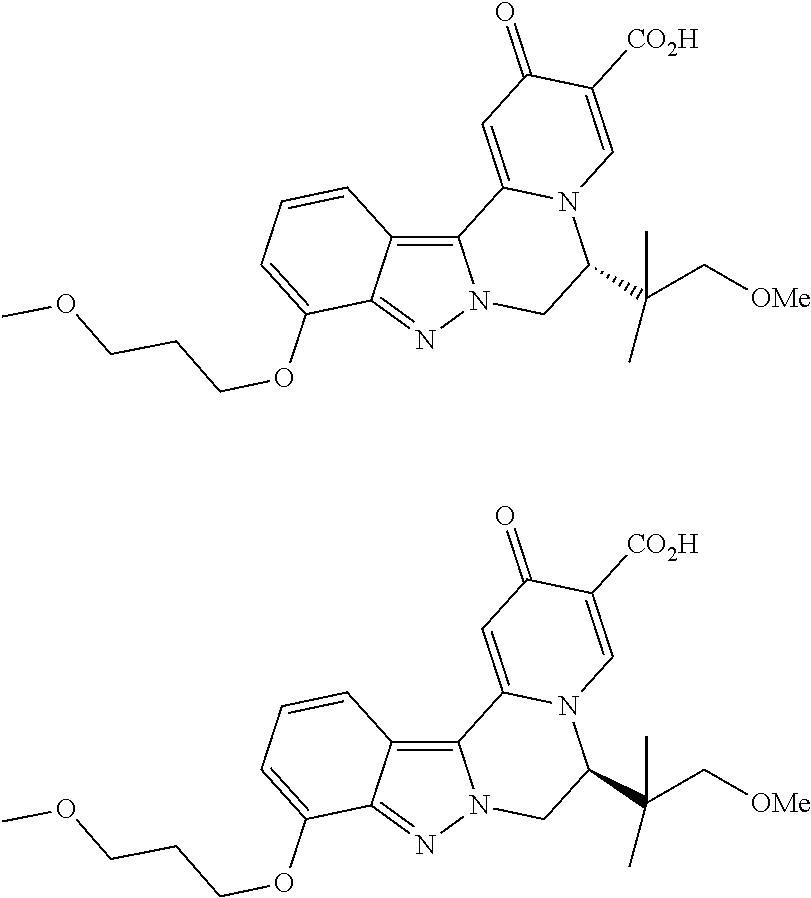
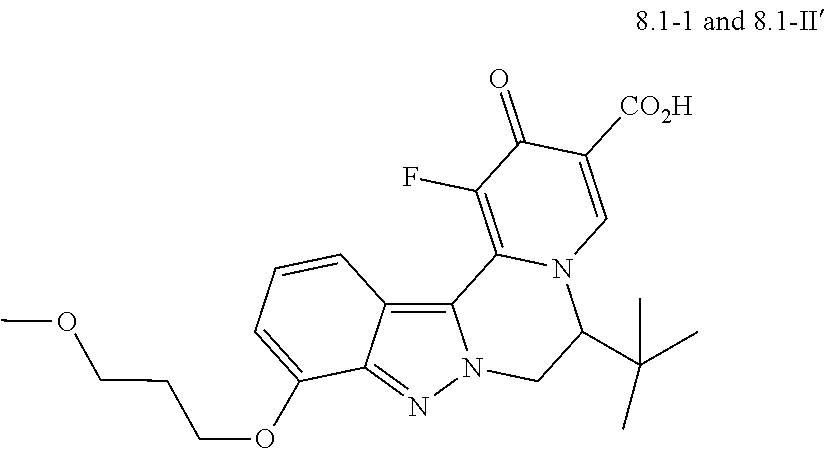

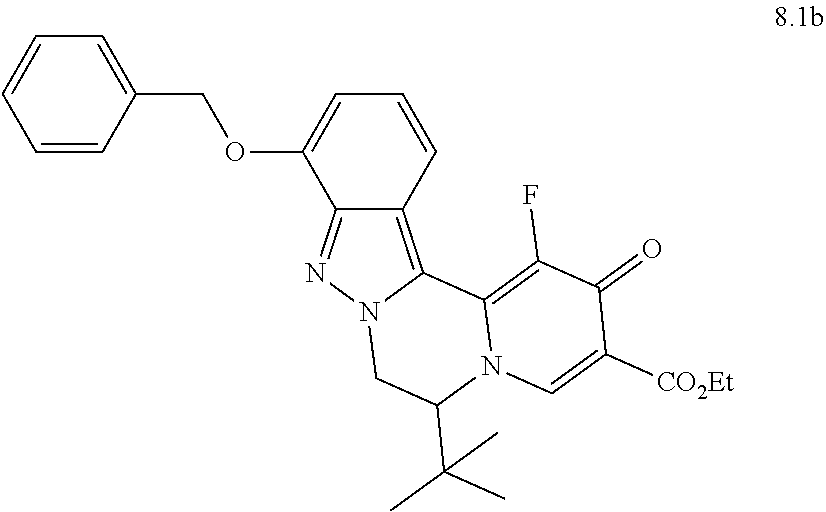

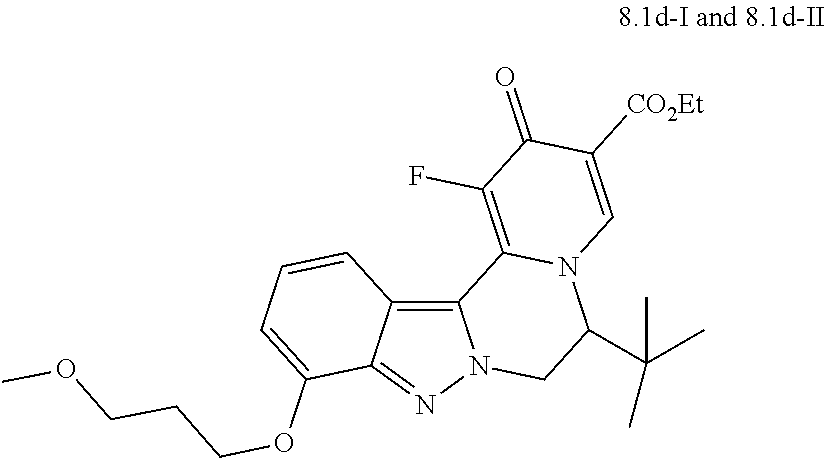
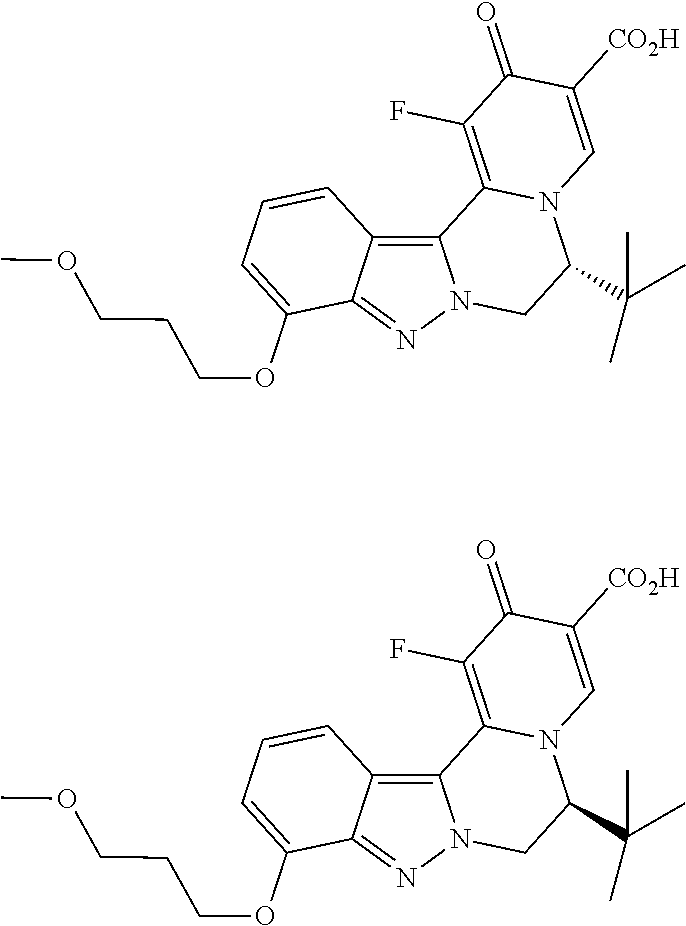
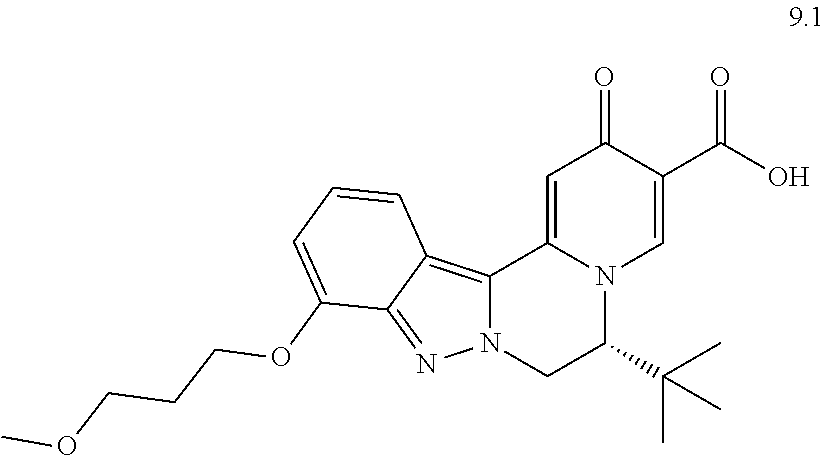
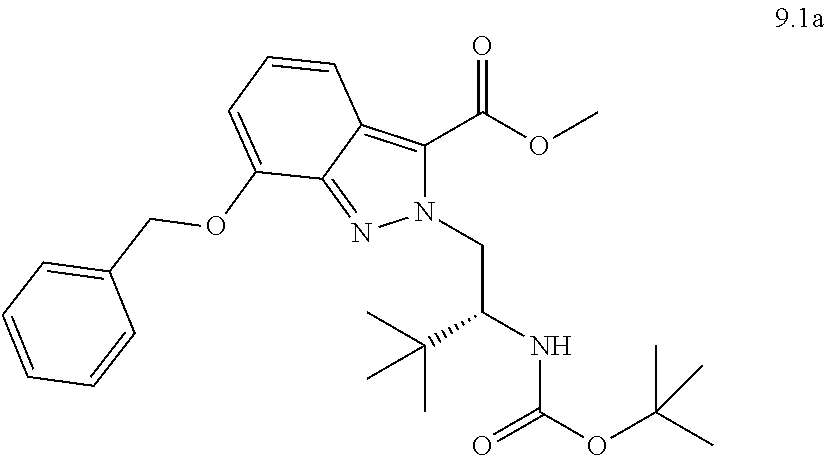
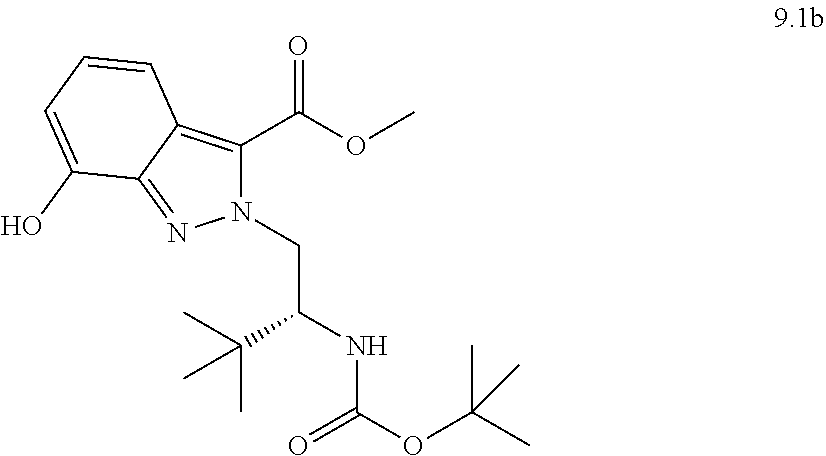
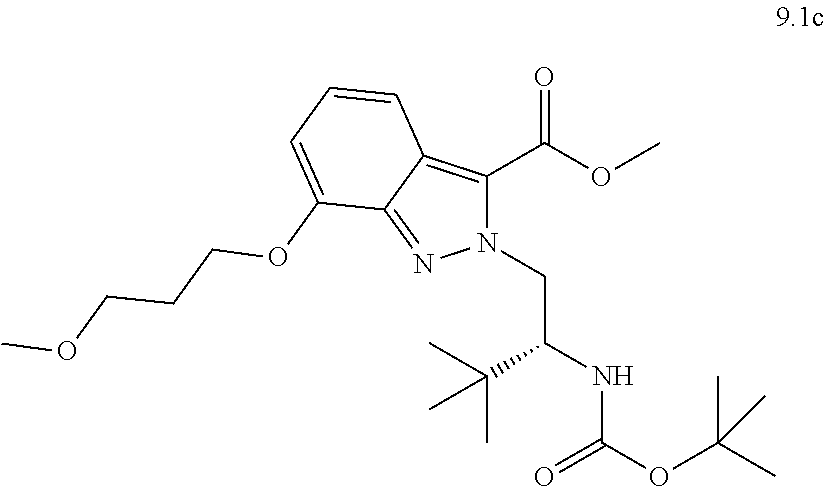
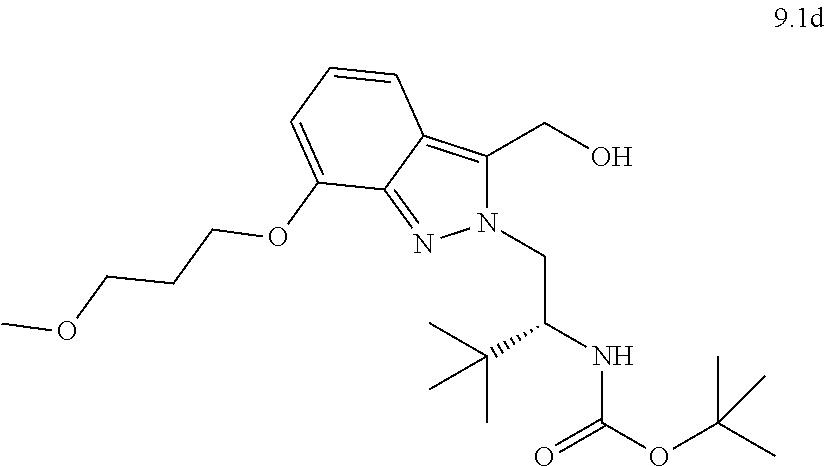
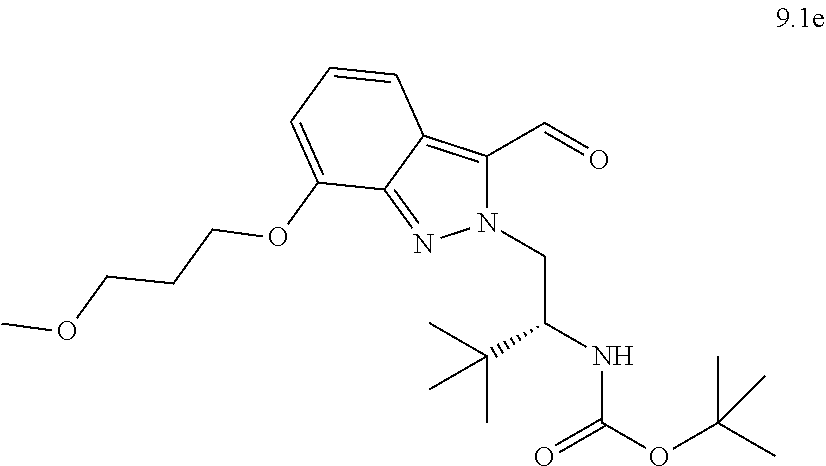

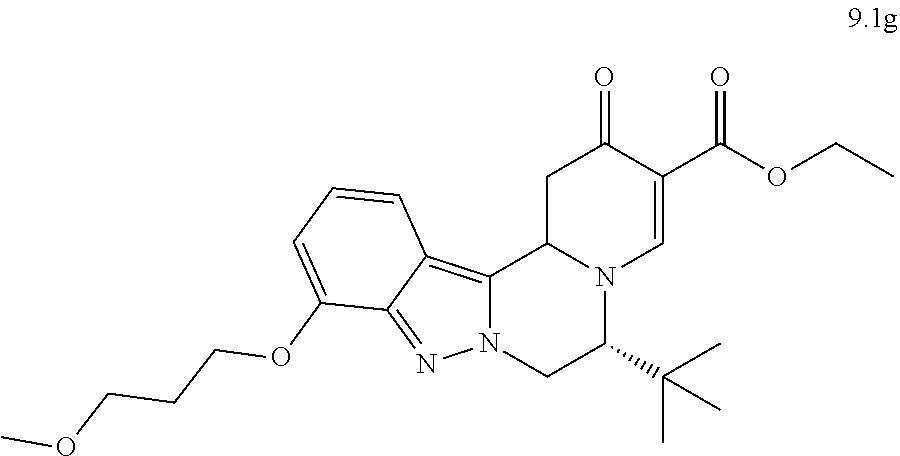
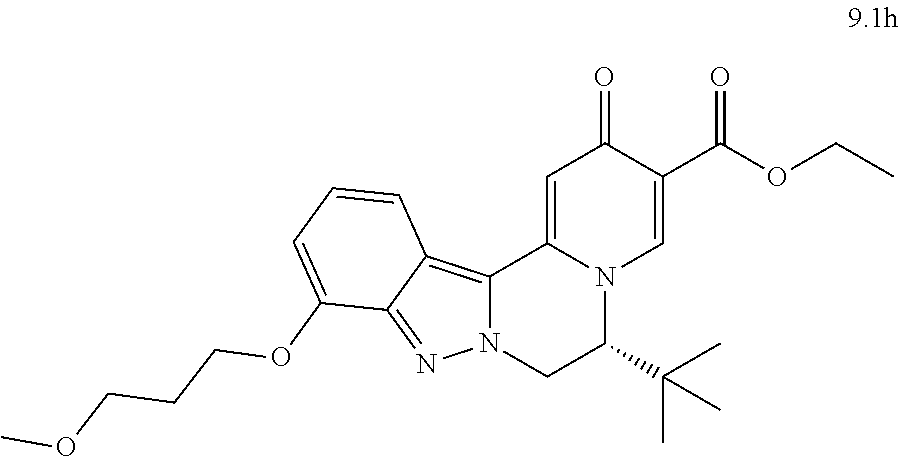
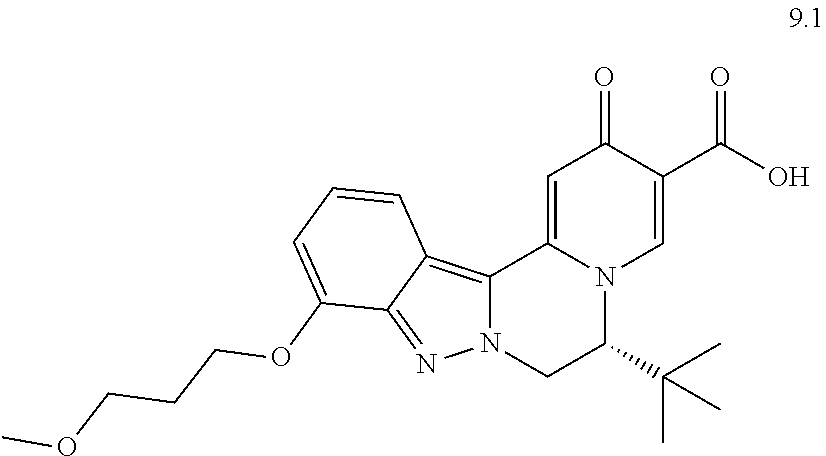
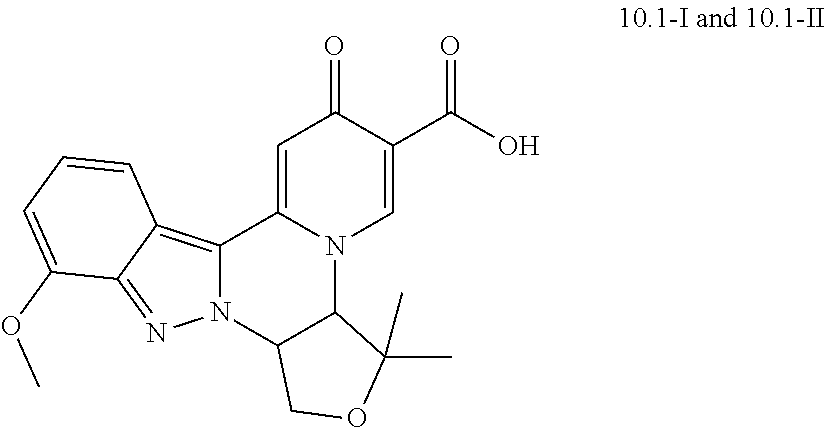
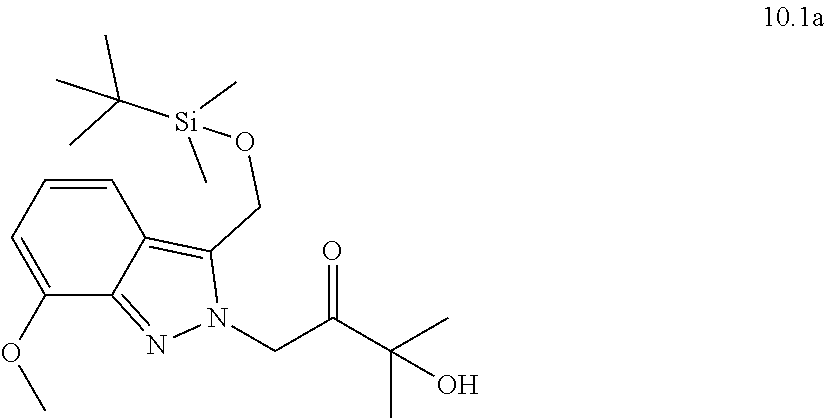
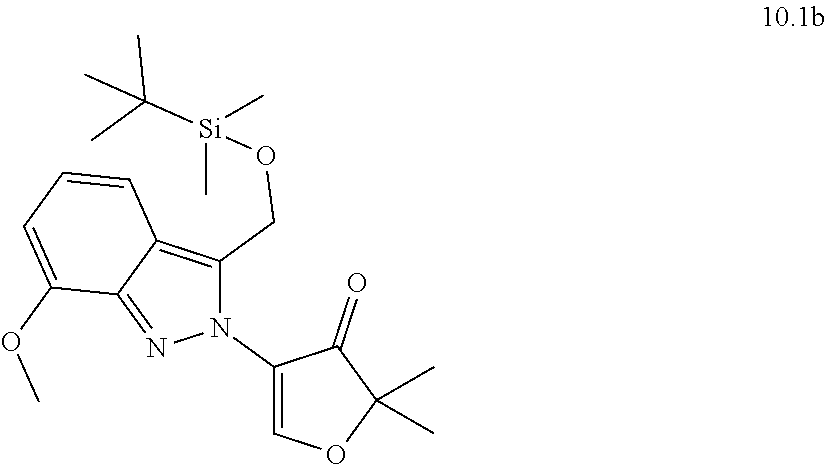
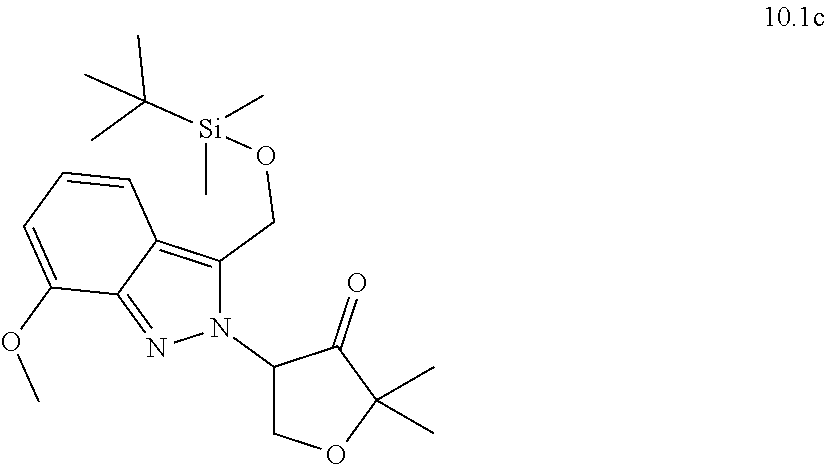
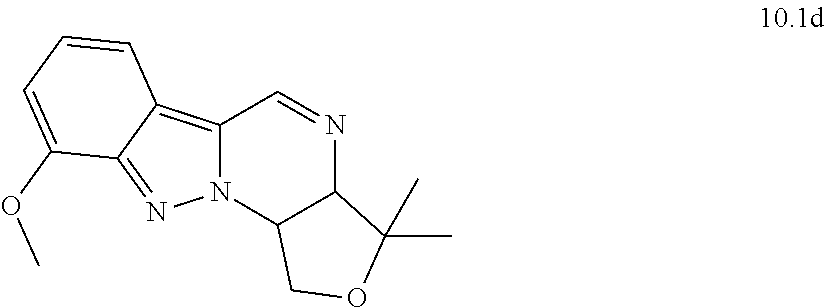
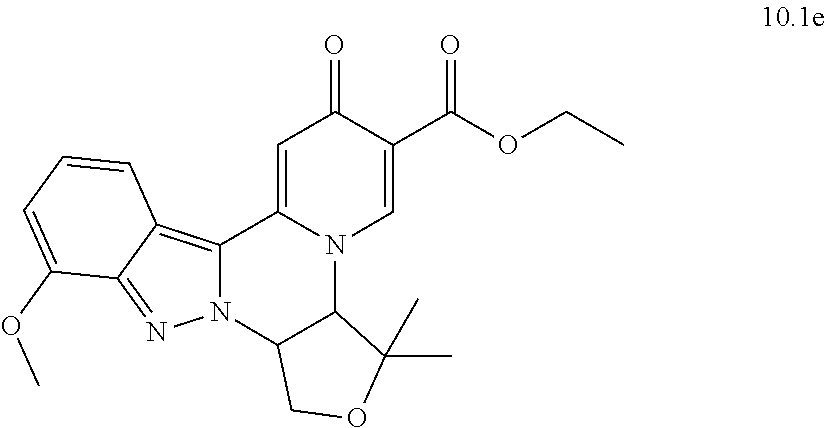
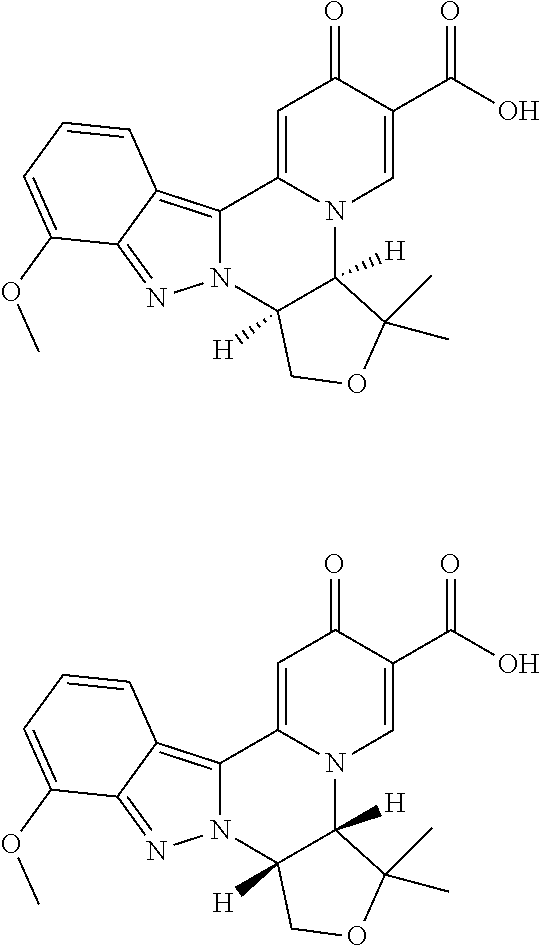
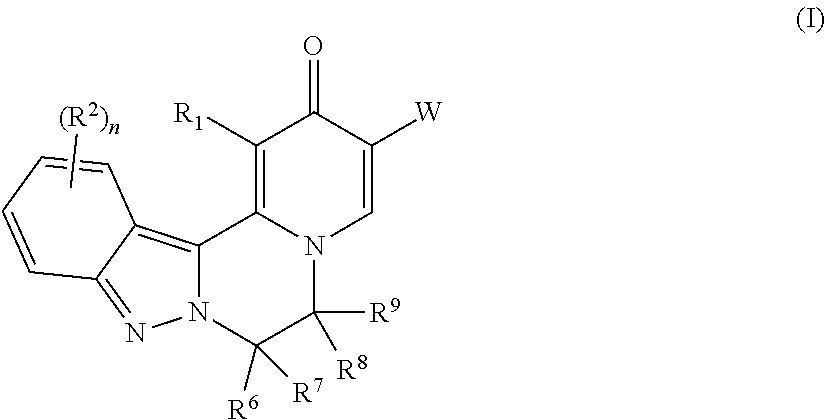

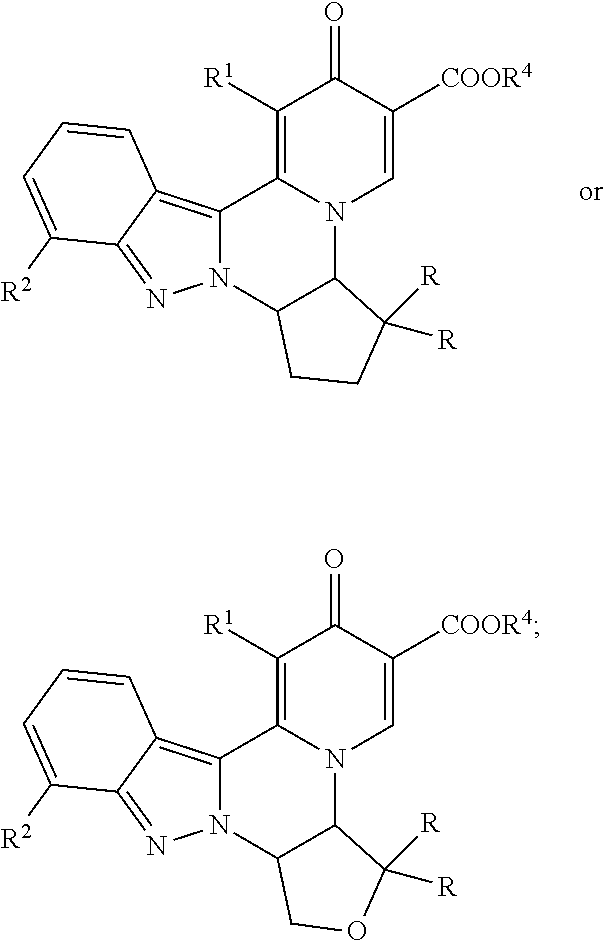
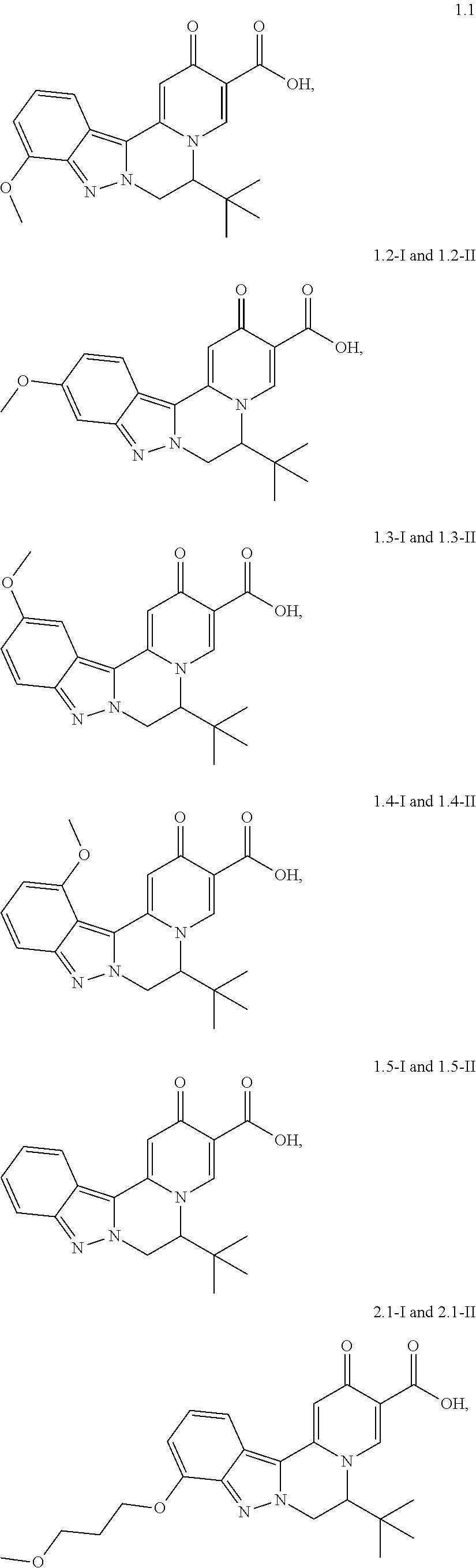
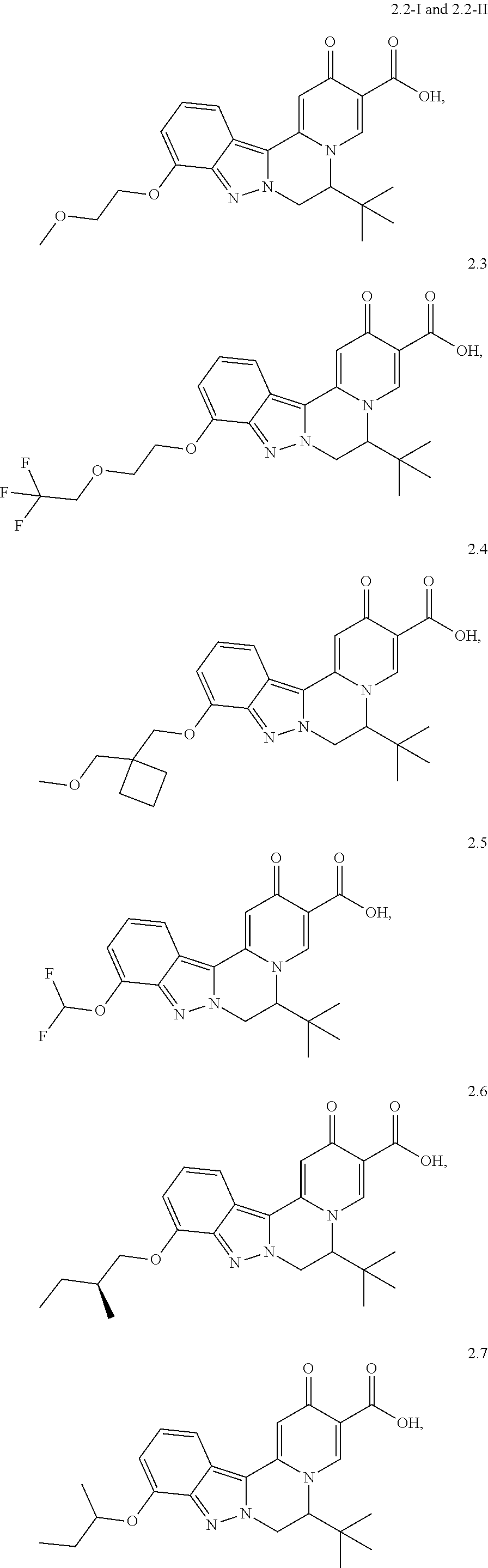
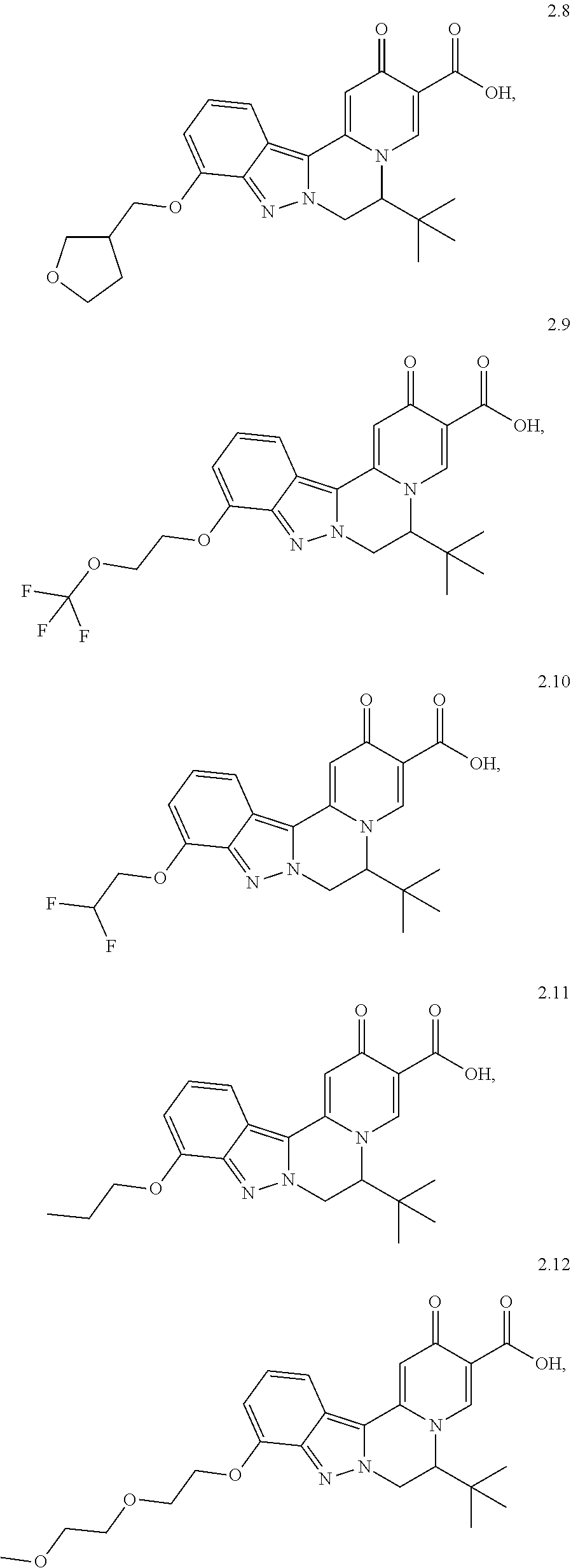
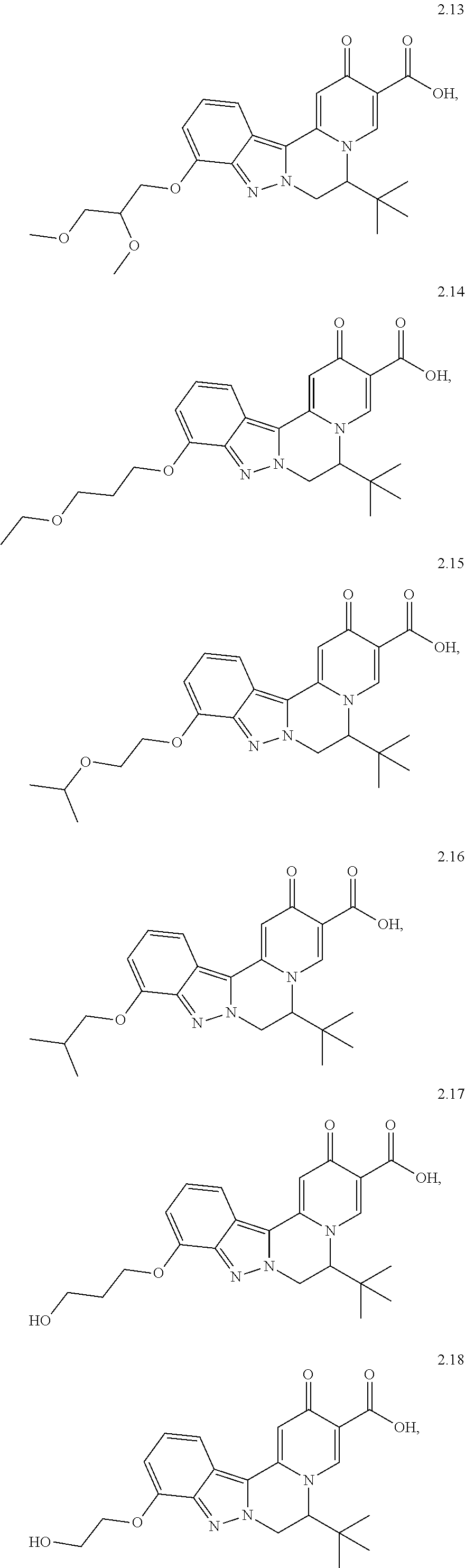
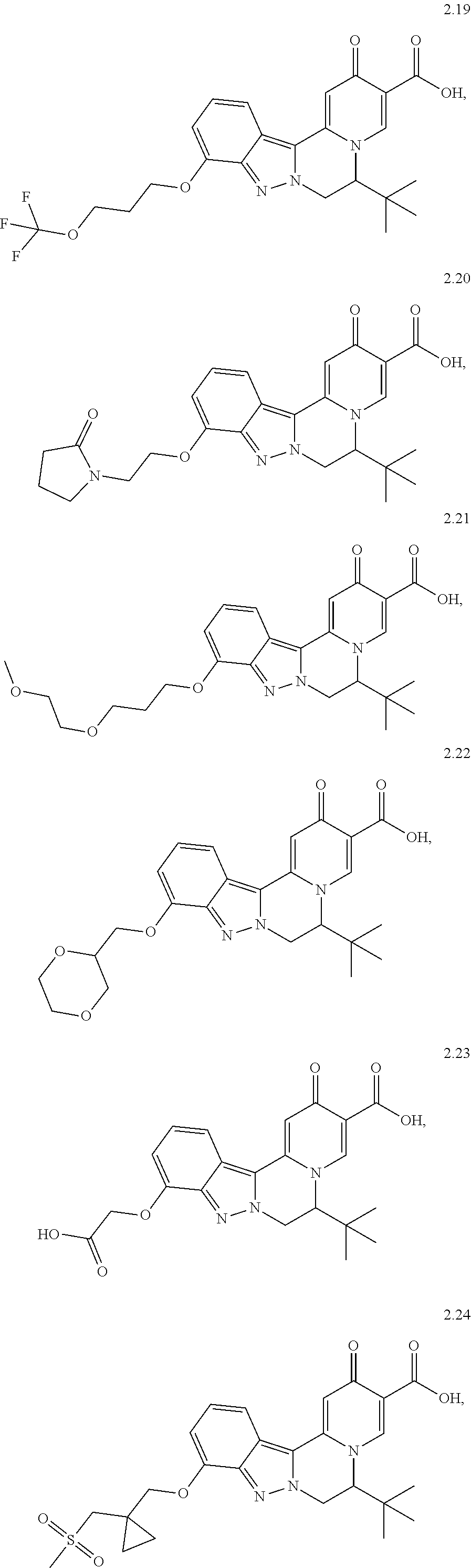
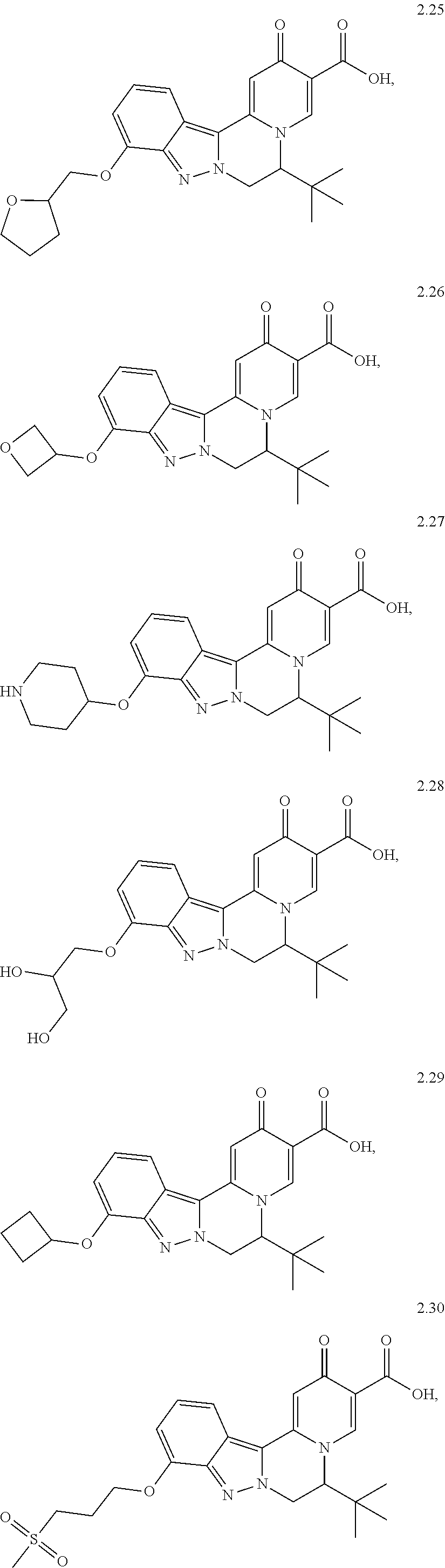
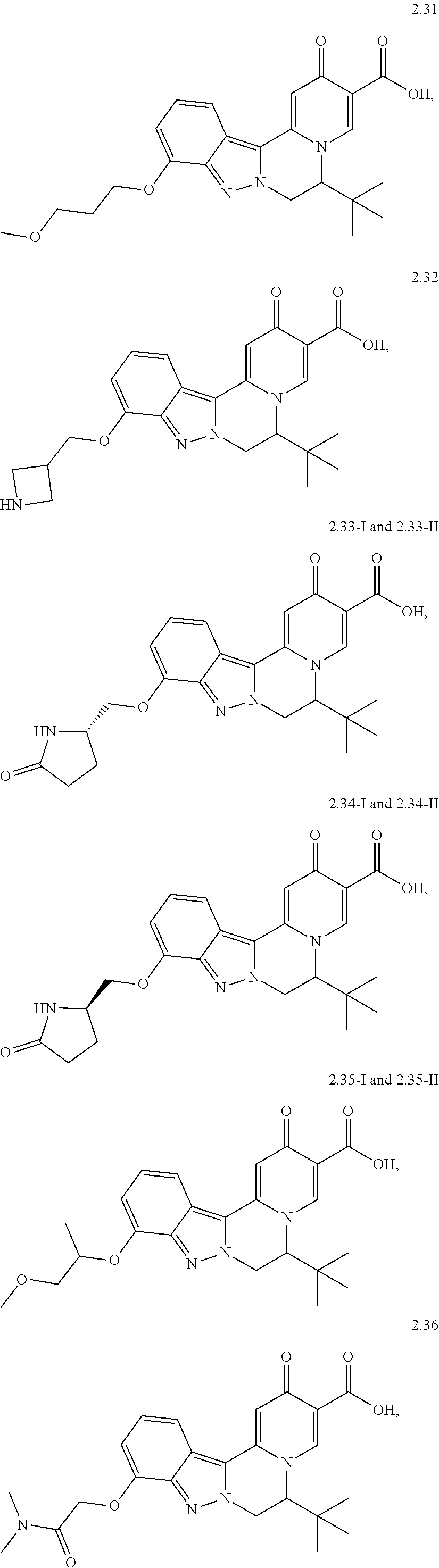
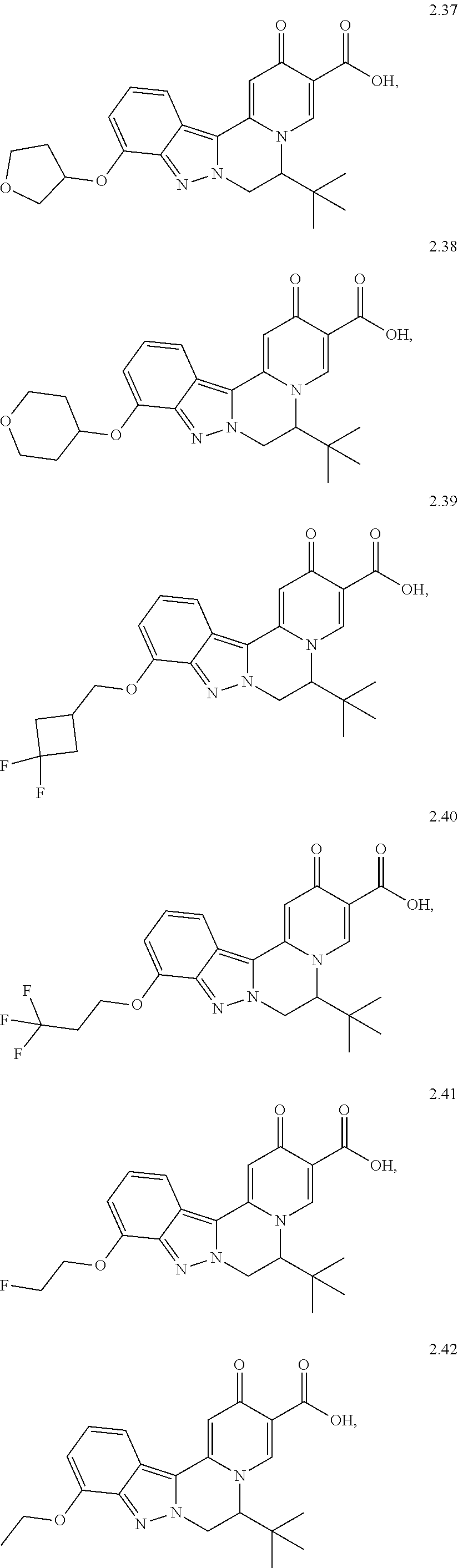
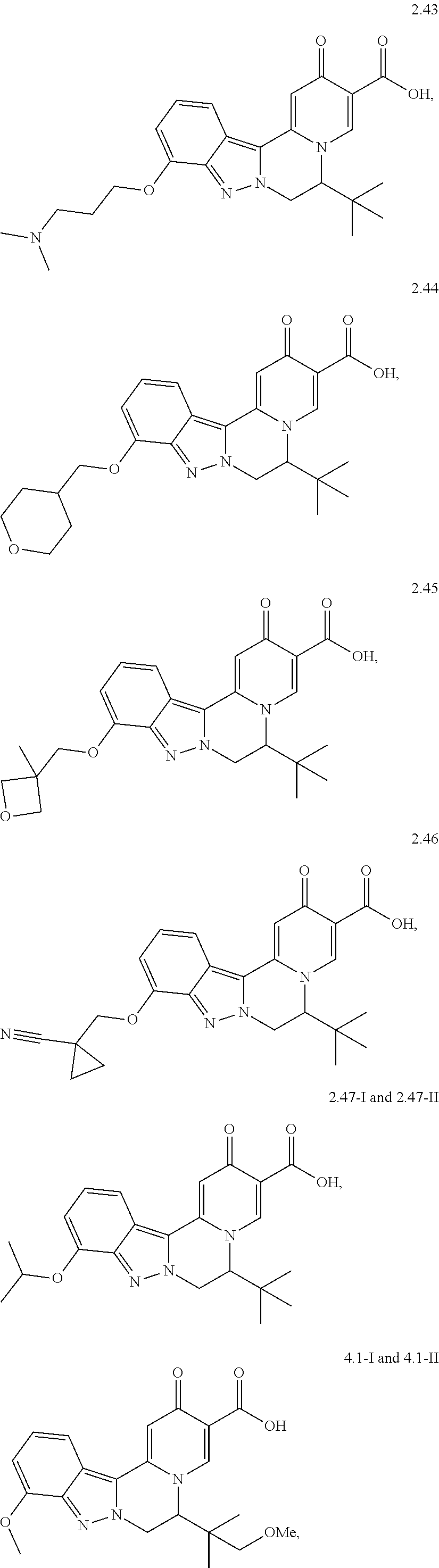
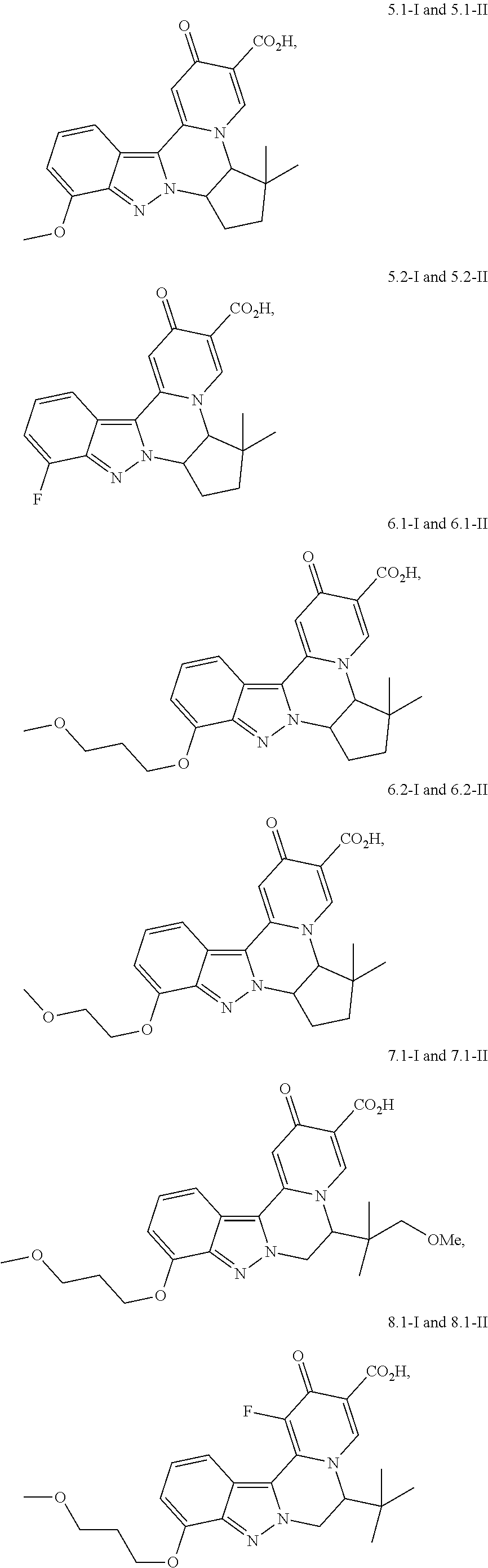
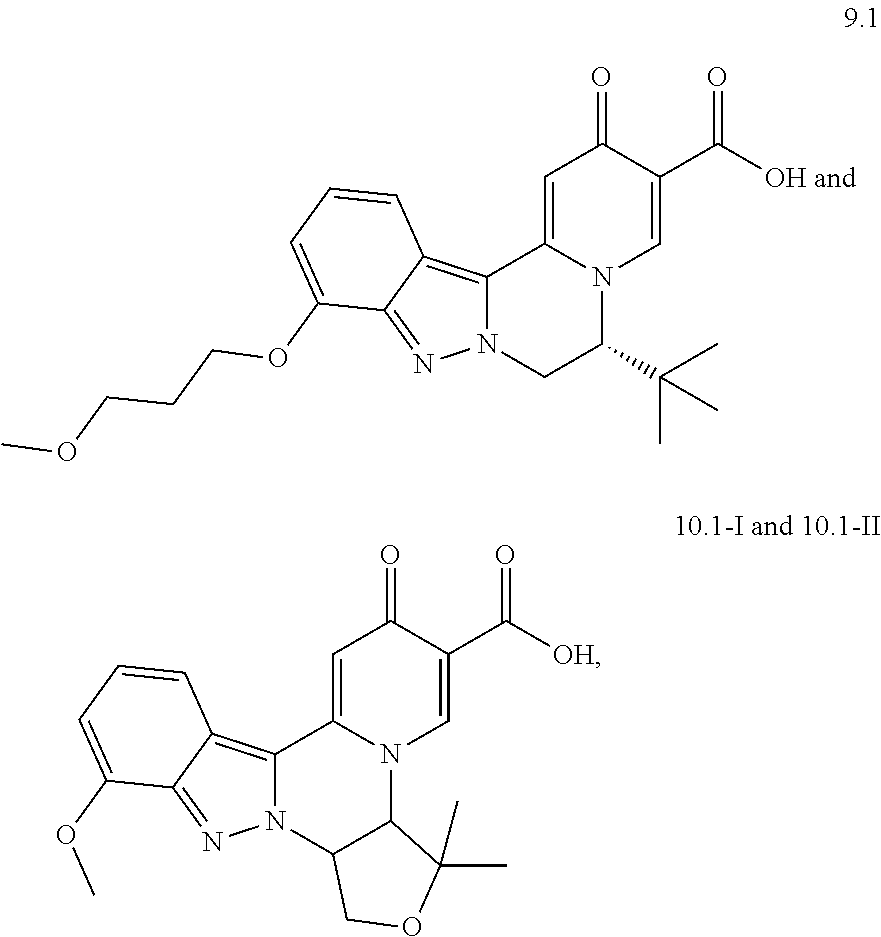
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.