TGFBeta Antibodies, Methods and Uses
Carven; Gregory J. ; et al.
U.S. patent application number 16/317635 was filed with the patent office on 2019-09-26 for tgfbeta antibodies, methods and uses. The applicant listed for this patent is Scholar Rock, Inc.. Invention is credited to Gregory J. Carven, Thomas Schurpf, Katherine Turner.
| Application Number | 20190292254 16/317635 |
| Document ID | / |
| Family ID | 60953402 |
| Filed Date | 2019-09-26 |


| United States Patent Application | 20190292254 |
| Kind Code | A1 |
| Carven; Gregory J. ; et al. | September 26, 2019 |
TGFBeta Antibodies, Methods and Uses
Abstract
ProTGFB1-GARP complex-selective antibodies, polynucleotides capable of encoding the proTGFB1-GARP complex-selective antibodies or antigen-binding fragments, cells expressing proTGFB1-GARP complex-selective antibodies or antigen-binding fragments, as well as associated vectors and detectably labeled proTGFB1-GARP complex-selective antibodies or antigen-binding fragments may be used to enhance an immune response in a subject, for example, against a cancer.
| Inventors: | Carven; Gregory J.; (Maynard, MA) ; Schurpf; Thomas; (Cambridge, MA) ; Turner; Katherine; (Acton, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 60953402 | ||||||||||
| Appl. No.: | 16/317635 | ||||||||||
| Filed: | July 14, 2017 | ||||||||||
| PCT Filed: | July 14, 2017 | ||||||||||
| PCT NO: | PCT/US2017/042162 | ||||||||||
| 371 Date: | January 14, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62362393 | Jul 14, 2016 | |||
| 62371355 | Aug 5, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 39/39 20130101; C07K 16/22 20130101; A61K 2039/505 20130101; C12N 9/00 20130101; C07K 2317/76 20130101; A61P 35/00 20180101; A61K 39/3955 20130101; C07K 16/28 20130101; A61P 37/02 20180101; C07K 2317/32 20130101; C07K 2317/92 20130101; A61P 31/00 20180101; A61K 38/18 20130101 |
| International Class: | C07K 16/22 20060101 C07K016/22; A61K 38/18 20060101 A61K038/18; C07K 16/28 20060101 C07K016/28; A61K 39/395 20060101 A61K039/395; A61K 39/39 20060101 A61K039/39 |
Claims
1. An isolated antibody, or an antigen-binding fragment thereof, wherein the antibody or antigen binding fragment specifically binds to human proTGF.beta.1 in a complex with human glycoprotein A repetitions predominant (proTGF.beta.1-GARP complex) while said complex is in solution and the antibody or antigen binding fragment thereof has no detectable binding according to the biolayer interferometry assay using the experimental design described in Example 4 to any of the following agents: a TGF.beta.1 growth factor domain, a TGF.beta.2 growth factor domain, TGF.beta.3 growth factor domain, proTGF.beta.1 covalently associated with LTBP1, proTGF.beta.1 covalently associated with LTBP3, proTGF.beta.1 covalently associated with LRRC33, and proTGF.beta.1 that is unassociated with human GARP, a. as measured by OctetRed 384 under the conditions shown in Examples 4 and 6 and further wherein the antibody or antigen-binding fragment thereof has an inhibitory concentration (IC.sub.50) of less than or equal to 10 nM for inhibition of TGF.beta.1 growth factor release from a cell-associated proTGF.beta.1-GARP complex under the conditions shown in Example 5, wherein the antibody or antigen binding fragment comprises: b. a heavy chain CDR1 having the amino acid sequence of SEQ ID NO: 4, a heavy chain CDR2 having the amino acid sequence of SEQ ID NO: 5, and a heavy chain CDR3 having the amino acid sequence of SEQ ID NO: 6, a light chain CDR1 having the amino acid sequence of SEQ ID NO: 7, a light chain CDR2 having the amino acid sequence of SEQ ID NO: 8, and a light chain CDR3 having the amino acid sequence of SEQ ID NO: 9; or c. a heavy chain CDR1 having the amino acid sequence of SEQ ID NO: 10, a heavy chain CDR2 having the amino acid sequence of SEQ ID NO: 11, and a heavy chain CDR3 having the amino acid sequence of SEQ ID NO: 12, a light chain CDR1 having the amino acid sequence of SEQ ID NO: 13, a light chain CDR2 having the amino acid sequence of SEQ ID NO: 14, and a light chain CDR3 having the amino acid sequence of SEQ ID NO: 15.
2. The antibody or antigen-binding fragment of claim 1 wherein the antibody or antigen-binding fragment inhibits Treg function in vitro.
3. The antibody or antigen-binding fragment of claim 1, wherein the antibody or antigen binding fragment thereof inhibits the activation of TGF.beta.1.
4. The antibody or antigen-binding fragment of claim 1, wherein the antibody or antigen binding fragment thereof binds to an epitope of human proTGF.beta.1 modified as a result of complex formation with human GARP.
5. The antibody or antigen-binding fragment of claim 1, wherein the antibody or antigen binding fragment thereof binds to a polypeptide having the amino acid sequence of SEQ ID NO: 2 in the presence of a polypeptide having the amino acid sequence of SEQ ID NO: 1.
6. The antibody or antigen-binding fragment of claim 1, wherein the antibody or antigen-binding fragment thereof specifically binds to human proTGF.beta.1 with a binding affinity of at least 880 pM as measured by biolayer interferometry assay using the experimental design described in Example 4.
7. The antibody or antigen-binding fragment of claim 1, wherein the antibody or antigen-binding fragment thereof binds to human proTGF.beta.1 with a dissociation constant (Kd) of less than or equal to 1 nM for human proTGF.beta.1 in a complex with human glycoprotein A repetitions predominant (proTGF.beta.1-GARP complex) under the conditions shown in Example 7 and wherein said proTGF.beta.1-GARP complex is in solution.
8. (canceled)
9. An isolated antibody, or an antigen-binding fragment thereof, comprising a heavy chain variable region selected from the group consisting of the amino acid sequences of SEQ ID NOs: 16 and 18 and a light chain variable region selected from the group consisting of the amino acid sequences of SEQ ID NOs: 17 and 19.
10. (canceled)
11. The antibody or antigen-binding fragment of claim 9, wherein a. the heavy chain variable region comprises the amino acid sequence of SEQ ID NO: 16 paired with a light chain variable region comprising the amino acid sequence of SEQ ID NO: 17; or b. the heavy chain variable region comprises the amino acid sequence of SEQ ID NO: 18 paired with a light chain variable region comprising the amino acid sequence of SEQ ID NO: 19.
12. The antigen-binding fragment of claim 11 wherein the antigen binding fragment is a Fab fragment, a Fab2 fragment, or a single chain antibody.
13. The antibody or antigen-binding fragment of claim 11 wherein the antibody or antigen-binding fragment is recombinant.
14. The antibody or antigen-binding fragment of claim 13 wherein the antibody or antigen-binding fragment thereof are of IgG1, IgG2, IgG3, or IgG4 isotype.
15. The antibody or antigen-binding fragment of claim 14, wherein the antibody is an IgG4 isotype.
16. An isolated antibody, or an antigen-binding fragment thereof, comprising: a. a heavy chain CDR1 having at least 80% identity to the amino acid sequence of SEQ ID NO: 4, a heavy chain CDR2 having at least 80% identity to the amino acid sequence of SEQ ID NO: 5, and a heavy chain CDR3 having at least 80% identity to the amino acid sequence of SEQ ID NO: 6, a light chain CDR1 having at least 80% identity to the amino acid sequence of SEQ ID NO: 7, a light chain CDR2 having at least 80% identity to the amino acid sequence of SEQ ID NO: 8, and a light chain CDR3 having at least 80% identity to the amino acid sequence of SEQ ID NO: 9; or b. a heavy chain CDR1 having at least 80% identity to the amino acid sequence of SEQ ID NO: 10, a heavy chain CDR2 having at least 80% identity to the amino acid sequence of SEQ ID NO: 11, and a heavy chain CDR3 having at least 80% identity to the amino acid sequence of SEQ ID NO: 12, a light chain CDR1 having at least 80% identity to the amino acid sequence of SEQ ID NO: 13, a light chain CDR2 having at least 80% identity to the amino acid sequence of SEQ ID NO: 14, and a light chain CDR3 having at least 80% identity to the amino acid sequence of SEQ ID NO: 15.
17. An isolated antibody, or an antigen-binding fragment thereof, comprising: a. a heavy chain variable region having at least 80% identity to the amino acid sequence of SEQ ID NO: 16 paired with a light chain variable region having at least 80% identity to the amino acid sequence of SEQ ID NO: 17; or b. a heavy chain variable region having at least 80% identity to the amino acid sequence of SEQ ID NO: 18 paired with a light chain variable region having at least 80% identity to the amino acid sequence of SEQ ID NO: 19.
18. A polynucleotide encoding the antibody or antigen binding fragment of claim 11.
19. A vector comprising the polynucleotide of claim 18.
20. A host cell comprising the vector of claim 19.
21. A process for the production of an antibody or antigen-binding fragment, comprising: culturing the host cell as defined in claim 20 under the conditions allowing the expression of the antibody or antigen-binding fragment, and recovering the antibody or antigen-binding molecule from the culture.
22. A pharmaceutical composition comprising the antibody, or antigen binding fragment thereof, of claim 11 and a pharmaceutically acceptable carrier.
23. A kit comprising the antibody, or antigen binding fragment thereof, of claim 11 and packaging for the same.
Description
SEQUENCE LISTING
[0001] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Jul. 10, 2017, is named JBI5093_SL.txt and is 27,577 bytes in size.
TECHNICAL FIELD
[0002] The present invention relates to monoclonal antibodies that inhibit growth factor activity and methods of producing and using the described antibodies.
BACKGROUND
[0003] Regulatory T cells, or Tregs, are a subset of CD4+ T lymphocytes specialized in the inhibition of immune responses. Insufficient Treg function results in autoimmune pathology, while excessive Treg function may inhibit anti-tumor immune responses in cancer patients. The exact mechanisms by which Tregs inhibit immune responses are not fully understood. Due to their immunosuppressive functions, Tregs represent potential inhibitors of spontaneous or vaccine-induced anti-tumor immune responses. In murine models, the depletion of Tregs can improve immune responses against experimental tumors (Colombo et al. Nat. Rev. Cancer 2007, 7:880-887). Thus, targeting Tregs in humans could improve the efficacy of immunotherapy against cancer.
[0004] TGF-.beta.1, which is instrumental in activating human Tregs but not other types of human T lymphocytes (Stockis, J. et al. Eur. J. Immunol. 2009, 39:869-882), could be a target of interest. However, antibodies against hTGF-.beta.1 were not found promising. Phase 1 clinical trials have been conducted in focal segmental glomerulosclerosis (FSGS), idiopathic pulmonary fibrosis (IPF) and advanced malignant melanoma or renal cell carcinoma (RCC) (Lonning S et al. Current Pharmaceutical Biotechnology 2011, 12:2176-2189). Depending on the trial, adverse events were observed in some patients. The main adverse reactions reported consisted in the development of keratoacanthoma (KA) and squamous cell carcinoma (SCC) in melanoma patients. It is possible that KA or SCC lesions in melanoma patients evolved from pre-cancerous cells whose proliferation was being inhibited by endogenous TGF-.beta.1 (Lonning S et al. Current Pharmaceutical Biotechnology 2011, 12:2176-2189). Therefore, a major concern regarding the use of anti-TGF-.beta.1 antibodies in the context of cancer is that they may favor the appearance of new neoplastic lesions, due to the inhibition of the tumor-suppressive effect exerted by endogenous TGF-.beta.1 on pre-cancerous cells. Thus, new strategies for improving cancer treatment by preventing TGF-.beta.1 release from Tregs are desirable.
SUMMARY OF THE PRESENT INVENTION
[0005] The present invention includes proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments thereof. Also described are related polynucleotides capable of encoding the provided proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments, cells expressing the provided antibodies and antigen-binding fragments, as well as associated vectors and detectably labeled proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments. The antibody or antigen binding fragment thereof does not selectively bind to a TGF.beta.1 growth factor domain, a TGF.beta.2 growth factor domain, a TGF.beta.3 growth factor domain, proTGF.beta.1 covalently associated with LTBP1, proTGF.beta.1 covalently associated with LTBP3, proTGF.beta.1 covalently associated with LRRC33, and proTGF.beta.1 that is unassociated with human GARP, as measured by OctetRed_384 under the conditions shown in Examples 4-6.
[0006] In some embodiments, the antibodies and antigen-binding fragments of the invention may have: (1) a dissociation constant (Kd) of less than or equal to 1 nM for human proTGF.beta.1 in a complex with human glycoprotein A repetitions predominant (proTGF.beta.1-GARP complex) in solution; (2) an inhibitory concentration (IC50) of less than or equal to 10 nM for inhibition of TGF.beta.1 growth factor release from cell-associated proTGF.beta.1-GARP complex; and (3) a greater than 100-fold selectivity for proTGF.beta.1-GARP complex over TGF.beta.1 growth factor domain, TGF.beta.2 growth factor domain, TGF.beta.3 growth factor domain, proTGF.beta.1 covalently associated with LTBP1, proTGF.beta.1 covalently associated with LTBP3, and proTGF.beta.1 covalently associated with LRRC33, wherein the isolated antibodies, or antigen binding fragments thereof, do not bind to proTGF.beta.1 that is unassociated with human GARP.
[0007] In addition, methods of using the provided proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments are described. The described proTGF.beta.1-GARP complex-selective antibodies can be used in methods of treating a variety of TGF.beta.1-related diseases or disorders in which it is desirable to modulate an immune response, such as a variety of immunotherapy applications, e.g., cancers, vaccines and infectious disease.
[0008] In some embodiments, the present invention comprises isolated antibodies and antigen-binding fragments wherein the antibody or antigen binding fragment specifically binds to human proTGF.beta.1 in a complex with human glycoprotein A repetitions predominant (proTGF.beta.1-GARP complex) while said complex is in solution. These proTGF.beta.1-GARP complex-selective antibodies, or antigen-binding fragments thereof may inhibit Treg function in vitro. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments inhibit activation of TGF.beta.1. In some embodiments the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments bind to an epitope of human proTGF.beta.1 modified as a result of complex formation with human GARP. This proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment may bind to proTGF.beta.1 of a proTGF.beta.1-GARP complex with a binding affinity of 880 pM or less.
TABLE-US-00001 TABLE 1 CDR sequences of human proTGF.beta.-GARP complex-selective mAbs (SEQ ID NO:) ID HC-CDR1 HC-CDR2 HC-CDR3 LC-CDR1 LC-CDR2 LC-CDR3 4B1C1 DYTMH LISWDGGSTYYADSVKG DADDSTFDI (6) RASQSVSRNLA (7) WASTRES QQYYSVPYT (4) (5) (8) (9) 4B16B9 SYAIS GIIPMFGTTNYAQKFQG DREWEPAYGMDV IGTSSDVGGYNYVS DVSNRPS SAYTVSSTWV (10) (11) (12) (13) (14) (15)
[0009] In some embodiments, the proTGF.beta.1-GARP complex-selective antibody, or an antigen-binding fragment thereof, comprises a heavy chain comprising a CDR1, a CDR2, and a CDR3 of any one of the amino acid sequences described in Table 1 and a light chain comprising a CDR1, a CDR2, and a CDR3 of any one of the amino acid sequences described in Table 1. The proTGF.beta.1-GARP complex-selective antibodies of the invention may comprise the heavy chain variable regions sequences of SEQ ID NOs: 16 and 18 and may comprise the light chain variable region sequences of SEQ ID NOs 17 and 19.
[0010] The proTGF.beta.1-GARP complex-selective antibodies described herein include antibodies with the described features of the CDRs and variable domains in combination with any of the IgG isotypes, including modified versions in which the Fc sequence has been modified to effect different effector functions.
[0011] In addition to the described proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments, also provided are polynucleotide sequences capable of encoding the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments. Vectors comprising the described polynucleotides are also provided, as are cells expressing the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments provided herein. Also described are cells capable of expressing the disclosed vectors. These cells may be mammalian cells (such as 293F cells, CHO cells), insect cells (such as Sf9 cells), yeast cells, plant cells, or bacteria cells (such as E. coli). A process for the production of the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments is also provided.
[0012] The present invention also comprises methods of using the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments. ProTGF.beta.1-GARP complex-selective antibodies for use in the methods discussed in this section include those with the set of CDRs described for antibodies in Table 1. For example, the key role that TGF.beta.1 plays in an immune response makes it an attractive target for immunotherapy, including inducing or enhancing an immune response against any weakly immunogenic antigen including tumor antigens. As such, the proTGF.beta.1-GARP complex-selective antibodies have utility in the treatment of various cancers and infectious disease.
[0013] In one embodiment, the proTGF.beta.1-GARP complex-selective antibodies are administered to block the release of TGF.beta.1 from Tregs and thereby, prevent the inhibition of effector T cell activity by regulatory T cells. Such inhibition can be assayed by a variety of methods known in the art, including, for example, by monitoring T cell proliferation, expression of known markers of activation, or cytokine secretion. In another embodiment, a proTGF.beta.1-GARP complex-selective antibody is administered to a subject to decrease the level of regulatory T cells, for instance the level of tumor regulatory T cells. In yet another embodiment, the activity of effector T cells is induced or enhanced by administering a proTGF.beta.1-GARP complex-selective antibody as provided herein.
[0014] Within the scope of the invention are kits including the disclosed proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments thereof. The described kits may be used to carry out the methods of using the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments provided herein, or other methods known to those skilled in the art. In some embodiments the described kits may include the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein and reagents for use in detecting the presence of proTGF.beta.1-GARP complex in a biological sample and, optionally, a vessel for containing the proTGF.beta.1-GARP complex-selective antibody or fragment when not in use, instructions for use of the proTGF.beta.1-GARP complex-selective antibody or fragment, the proTGF.beta.1-GARP complex-selective antibody or fragment affixed to a solid support, and/or detectably labeled forms of the proTGF.beta.1-GARP complex-selective antibody or fragment.
BRIEF DESCRIPTION OF THE DRAWINGS
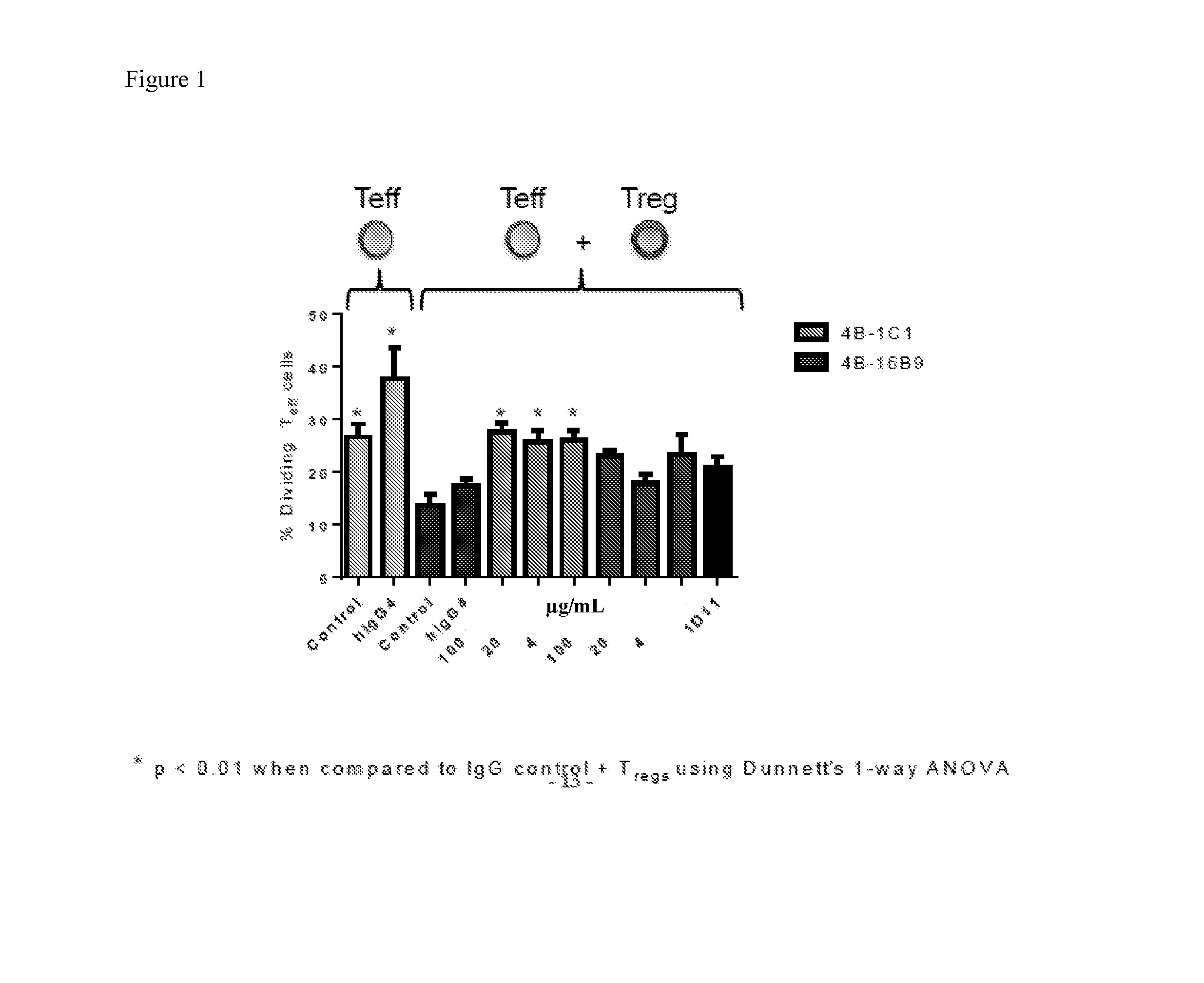
[0015] FIG. 1 shows that addition of 4B1C1 and 4B16B9 to T cell co-cultures inhibit T regulatory cell activity through the enhanced growth of T effector cells.
[0016] FIG. 2 shows 4B1C1 and 4B16B9 inhibit TGF.beta.1 activation as assessed by SMAD signaling.
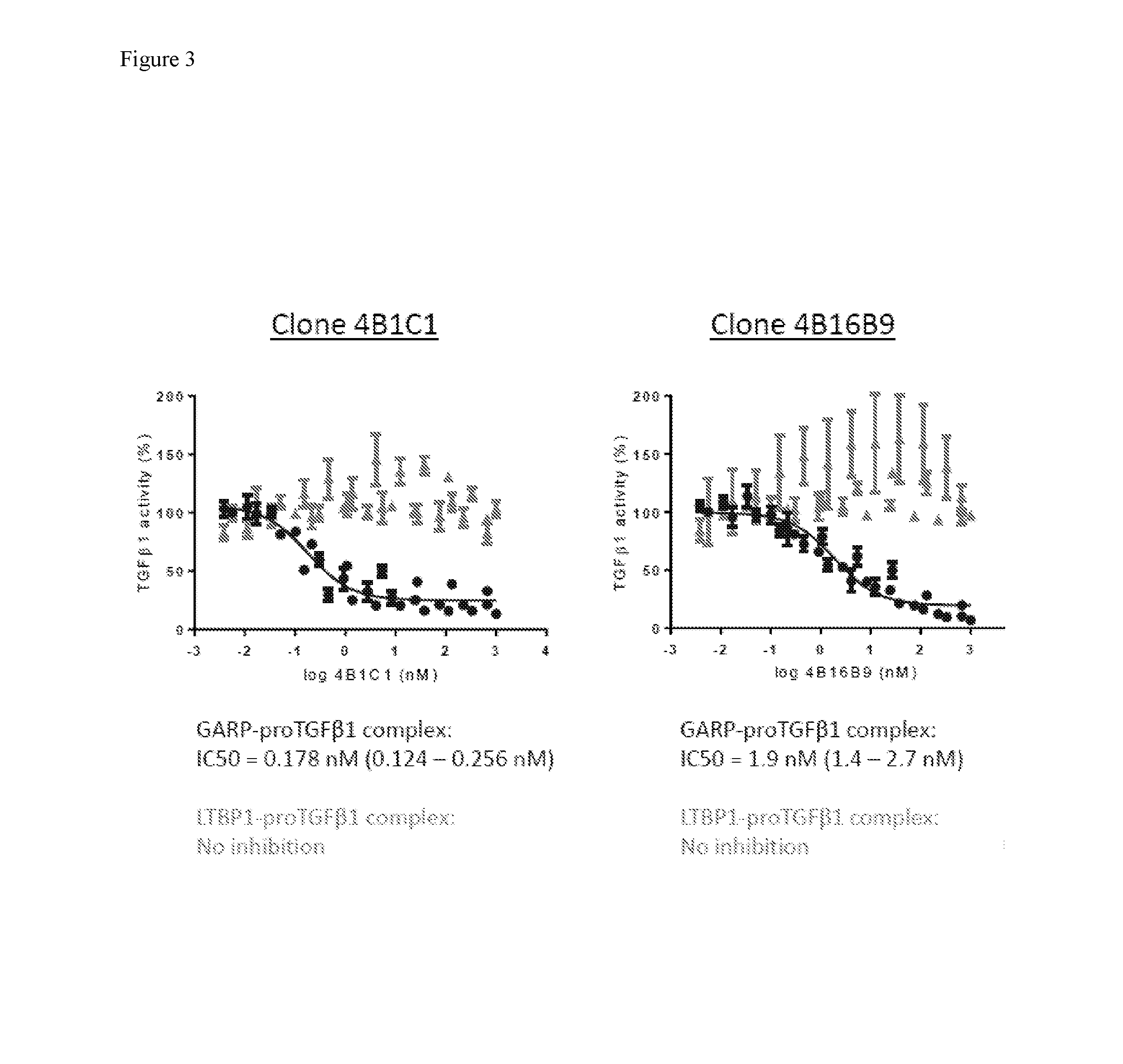
[0017] FIG. 3 shows the dose-dependent inhibition of TGF.beta.1 activity by 4B1C1 and 4B16B9.
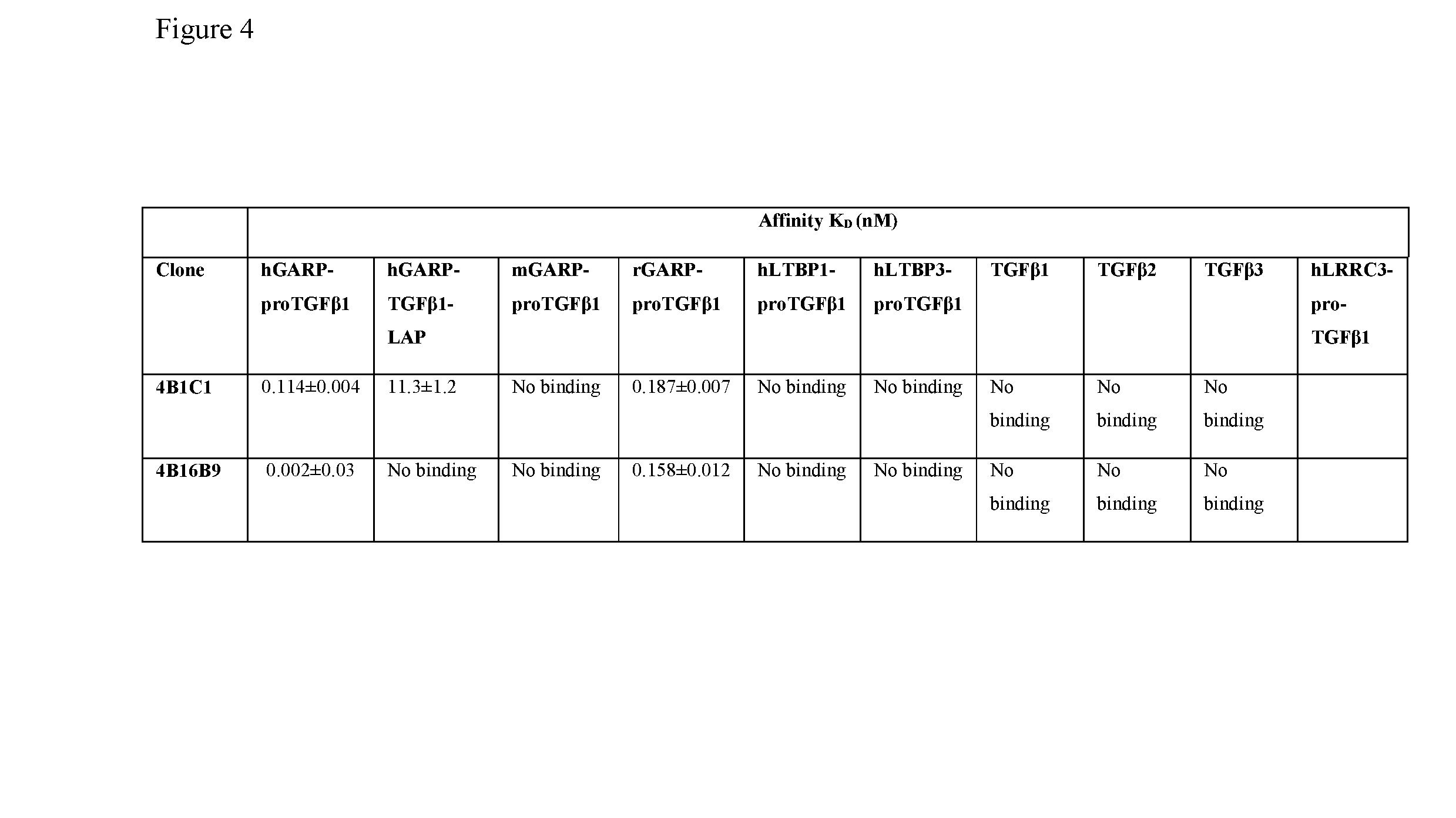
[0018] FIG. 4. Octet affinity results for proTGF.beta.1-GARP complex-selective antibody candidates demonstrate specificity by binding to the human proTGF.beta.1-GARP complex but no other proTGFb1-complexes or soluble forms of TGFb1, 2 or 3.
DETAILED DESCRIPTION OF ILLUSTRATIVE EMBODIMENTS
Definitions
[0019] Various terms relating to aspects of the description are used throughout the specification and claims. Such terms are to be given their ordinary meaning in the art unless otherwise indicated. Other specifically defined terms are to be construed in a manner consistent with the definitions provided herein.
[0020] As used in this specification and the appended claims, the singular forms "a," "an," and "the" include plural referents unless the content clearly dictates otherwise. Thus, for example, reference to "a cell" includes a combination of two or more cells, and the like.
[0021] The term "about" as used herein when referring to a measurable value such as an amount, a temporal duration, and the like, is meant to encompass variations of up to .+-.10% from the specified value, as such variations are appropriate to perform the disclosed methods. Unless otherwise indicated, all numbers expressing quantities of ingredients, properties such as molecular weight, reaction conditions, and so forth used in the specification and claims are to be understood as being modified in all instances by the term "about." Accordingly, unless indicated to the contrary, the numerical parameters set forth in the following specification and attached claims are approximations that may vary depending upon the desired properties sought to be obtained by the present invention. At the very least, and not as an attempt to limit the application of the doctrine of equivalents to the scope of the claims, each numerical parameter should at least be construed in light of the number of reported significant digits and by applying ordinary rounding techniques.
[0022] Notwithstanding that the numerical ranges and parameters setting forth the broad scope of the invention are approximations, the numerical values set forth in the specific examples are reported as precisely as possible. Any numerical value, however, inherently contains certain errors necessarily resulting from the standard deviation found in their respective testing measurements.
[0023] "Isolated" means a biological component (such as a nucleic acid, peptide or protein) has been substantially separated, produced apart from, or purified away from other biological components of the organism in which the component naturally occurs, i.e., other chromosomal and extrachromosomal DNA and RNA, and proteins. Nucleic acids, peptides and proteins that have been "isolated" thus include nucleic acids and proteins purified by standard purification methods. "Isolated" nucleic acids, peptides and proteins can be part of a composition and still be isolated if such composition is not part of the native environment of the nucleic acid, peptide, or protein. The term also embraces nucleic acids, peptides and proteins prepared by recombinant expression in a host cell as well as chemically synthesized nucleic acids. An "isolated" antibody or antigen-binding fragment, as used herein, is intended to refer to an antibody or antigen-binding fragment which is substantially free of other antibodies or antigen-binding fragments having different antigenic specificities (for instance, an isolated antibody that is a proTGF.beta.1-GARP complex-selective antibody is substantially free of antibodies that are not proTGF.beta.1-GARP complex-selective antibodies).
[0024] As used herein, the terms "transforming growth factor beta-1" and "TGF.beta.1" specifically include the human TGF.beta.1 protein. TGF.beta.1 is also known in the scientific literature as TGFbeta1 and TGFB1. TGF.beta.1 growth factor is synthesized in conjunction with a prodomain, for example as described in GenBank.TM. Accession No. AK291907, NCBI Reference Sequence: NP_000651.3.1 and UniProtKB/Swiss-Prot Accession No. P01137.2 (see also Derynck et al. 1985, Nature 316, 701-705). In a particular embodiment, the TGF.beta.1 translated protein is a human protein having the amino acid sequence of SEQ ID NO: 2. TGF.beta.1 that includes both prodomain and growth factor elements is referred to herein as "proTGF.beta.1." In some embodiments, proTGF.beta.1 includes prodomain and growth factor components that have been proteolytically separated, but that remain associated through one or more chemical interactions. Such chemical interactions may include, but are not limited to, hydrophobic bonds, interactions influenced by van der Waals forces, polar and ionic interactions, hydrogen bonds, and noncovalent bonds.
[0025] As used herein, the terms "glycoprotein A repetitions predominant" and "GARP" refer to human GARP. GARP is otherwise known as leucine-rich repeat-containing protein 32 (LRRC32) and garpin. NCBI Reference Sequence NP_001122394.1 and NP_005503.1 provide exemplary human GARP amino acid sequences. In a particular embodiment, the GARP is a human GARP of SEQ ID NO: 1.
[0026] "Antibody" refers to all isotypes of immunoglobulins (IgG, IgA, IgE, IgM, IgD, and IgY) including various monomeric, polymeric and chimeric forms, unless otherwise specified. Specifically encompassed by the term "antibody" are polyclonal antibodies, monoclonal antibodies (mAbs), and antibody-like polypeptides, such as chimeric antibodies and humanized antibodies.
[0027] "Antigen-binding fragments" are any proteinaceous structure that may exhibit binding affinity for a particular antigen. Antigen-binding fragments include those provided by any known technique, such as enzymatic cleavage, peptide synthesis, and recombinant techniques. Some antigen-binding fragments are composed of portions of intact antibodies that retain antigen-binding specificity of the parent antibody molecule. For example, antigen-binding fragments may comprise at least one variable region (either a heavy chain or light chain variable region) or one or more CDRs of an antibody known to bind a particular antigen. Examples of suitable antigen-binding fragments include, without limitation diabodies and single-chain molecules as well as Fab, F(ab')2, Fc, Fabc, and Fv molecules, single chain (Sc) antibodies, individual antibody light chains, individual antibody heavy chains, chimeric fusions between antibody chains or CDRs and other proteins, protein scaffolds, heavy chain monomers or dimers, light chain monomers or dimers, dimers consisting of one heavy and one light chain, a monovalent fragment consisting of the VL, VH, CL and CH1 domains, or a monovalent antibody as described in WO2007059782, bivalent fragments comprising two Fab fragments linked by a disulfide bridge at the hinge region, a Fd fragment consisting essentially of the V.sub.H and C.sub.H1 domains; a Fv fragment consisting essentially of the VL and VH domains of a single arm of an antibody, a dAb fragment (Ward et al., Nature 341, 544-546 (1989)), which consists essentially of a VH domain and also called domain antibodies (Holt et al; Trends Biotechnol. 2003 November; 21(11):484-90); camelid or nanobodies (Revets et al; Expert Opin Biol Ther. 2005 January; 5(1):111-24); an isolated complementarity determining region (CDR), and the like. All antibody isotypes may be used to produce antigen-binding fragments. Additionally, antigen-binding fragments may include non-antibody proteinaceous frameworks that may successfully incorporate polypeptide segments in an orientation that confers affinity for a given antigen of interest, such as protein scaffolds. Antigen-binding fragments may be recombinantly produced or produced by enzymatic or chemical cleavage of intact antibodies. The phrase "an antibody or antigen-binding fragment thereof" may be used to denote that a given antigen-binding fragment incorporates one or more amino acid segments of the antibody referred to in the phrase.
[0028] The terms "CDR", and its plural "CDRs", refer to a complementarity determining region (CDR) of which three make up the binding character of a light chain variable region (CDRL1, CDRL2 and CDRL3) and three make up the binding character of a heavy chain variable region (CDRH1, CDRH2 and CDRH3). CDRs contribute to the functional activity of an antibody molecule and are separated by amino acid sequences that comprise scaffolding or framework regions. The exact definitional CDR boundaries and lengths are subject to different classification and numbering systems. CDRs may therefore be referred to by Kabat, Chothia, contact or any other boundary definitions. Despite differing boundaries, each of these systems has some degree of overlap in what constitutes the so called "hypervariable regions" within the variable sequences. CDR definitions according to these systems may therefore differ in length and boundary areas with respect to the adjacent framework region. See for example Kabat et al., Sequences of Proteins of Immunological Interest, 5th ed. NIH Publication No. 91-3242 (1991); Chothia et al., "Canonical Structures For the Hypervariable Regions of Immunoglobulins," J. Mol. Biol. 196:901 (1987); and MacCallum et al., "Antibody-Antigen Interactions: Contact Analysis and Binding Site Topography," J. Mol. Biol. 262:732 (1996)), each of which is hereby incorporated by reference in its entirety.
[0029] Typically, CDRs form a loop structure that can be classified as a canonical structure. The term "canonical structure" refers to the main chain conformation that is adopted by the antigen binding (CDR) loops. From comparative structural studies, it has been found that five of the six antigen binding loops have only a limited repertoire of available conformations. Each canonical structure can be characterized by the torsion angles of the polypeptide backbone. Correspondent loops between antibodies may, therefore, have very similar three dimensional structures, despite high amino acid sequence variability in most parts of the loops (Chothia et al., "Canonical Structures For the Hypervariable Regions of Immunoglobulins," J. Mol. Biol. 196:901 (1987); Chothia et al., "Conformations of Immunoglobulin Hypervariable Regions," I 342:877 (1989); Martin and Thornton, "Structural Families in Loops of Homologous Proteins: Automatic Classification, Modelling and Application to Antibodies," J. Mol. Biol. 263:800 (1996), each of which is incorporated by reference in its entirety). Furthermore, there is a relationship between the adopted loop structure and the amino acid sequences surrounding it. The conformation of a particular canonical class is determined by the length of the loop and the amino acid residues residing at key positions within the loop, as well as within the conserved framework (i.e., outside of the loop). Assignment to a particular canonical class can therefore be made based on the presence of these key amino acid residues.
[0030] The term "polypeptide" is used interchangeably with the term "protein" and in its broadest sense refers to a compound of two or more subunit amino acids, amino acid analogs or peptidomimetics. The subunits may be linked by peptide bonds. In another embodiment, the subunit may be linked by other bonds, e.g., ester, ether, etc. As used herein the term "amino acid" refers to either natural and/or unnatural or synthetic amino acids, including glycine and both the D and L optical isomers, amino acid analogs and peptidomimetics. A peptide of three or more amino acids is commonly called an oligopeptide if the peptide chain is short. If the peptide chain is long, the peptide is commonly called a polypeptide or a protein.
[0031] "Specifically binds" or "binds specifically" or derivatives thereof when used in the context of antibodies, or antibody fragments, represents binding via domains encoded by immunoglobulin genes or fragments of immunoglobulin genes to one or more epitopes of a protein of interest, without preferentially binding other molecules in a sample containing a mixed population of molecules. Typically, an antibody binds to a cognate antigen with a K.sub.d of less than about 1.times.10.sup.-8 M, as measured by a surface plasmon resonance assay, or a cell-binding assay. In a preferred embodiment, binding specificity is measure using biolayer interferometry. Phrases such as "[antigen]-specific" antibody are meant to convey that the recited antibody specifically binds the recited antigen.
[0032] "Polynucleotide," synonymously referred to as "nucleic acid molecule," "nucleotides" or "nucleic acids," refers to any polyribonucleotide or polydeoxyribonucleotide, which may be unmodified RNA or DNA or modified RNA or DNA. "Polynucleotides" include, without limitation single- and double-stranded DNA, DNA that is a mixture of single- and double-stranded regions, single- and double-stranded RNA, and RNA that is mixture of single- and double-stranded regions, hybrid molecules comprising DNA and RNA that may be single-stranded or, more typically, double-stranded or a mixture of single- and double-stranded regions. In addition, "polynucleotide" refers to triple-stranded regions comprising RNA or DNA or both RNA and DNA. The term polynucleotide also includes DNAs or RNAs containing one or more modified bases and DNAs or RNAs with backbones modified for stability or for other reasons. "Modified" bases include, for example, tritylated bases and unusual bases such as inosine. A variety of modifications may be made to DNA and RNA; thus, "polynucleotide" embraces chemically, enzymatically or metabolically modified forms of polynucleotides as typically found in nature, as well as the chemical forms of DNA and RNA characteristic of viruses and cells. "Polynucleotide" also embraces relatively short nucleic acid chains, often referred to as oligonucleotides.
[0033] A "vector" is a replicon, such as plasmid, phage, cosmid, or virus in which another nucleic acid segment may be operably inserted so as to bring about the replication or expression of the segment.
[0034] As used herein, the term "host cell" can be any type of cell, e.g., a primary cell, a cell in culture, or a cell from a cell line. In specific embodiments, the term "host cell" refers to a cell transfected with a nucleic acid molecule and the progeny or potential progeny of such a cell. Progeny of such a cell may not be identical to the parent cell transfected with the nucleic acid molecule, e.g., due to mutations or environmental influences that may occur in succeeding generations or integration of the nucleic acid molecule into the host cell genome. The terms "expression" and "production" are used synonymously herein, and refer to the biosynthesis of a gene product. These terms encompass the transcription of a gene into RNA. These terms also encompass translation of RNA into one or more polypeptides, and further encompass all naturally occurring post-transcriptional and post-translational modifications. The expression or production of an antibody or antigen-binding fragment thereof may be within the cytoplasm of the cell, or into the extracellular milieu such as the growth medium of a cell culture. The meaning of "substantially the same" can differ depending on the context in which the term is used. Because of the natural sequence variation likely to exist among heavy and light chains and the genes encoding them, one would expect to find some level of variation within the amino acid sequences or the genes encoding the antibodies or antigen-binding fragments described herein, with little or no impact on their unique binding properties (e.g., specificity and affinity). Such an expectation is due in part to the degeneracy of the genetic code, as well as to the evolutionary success of conservative amino acid sequence variations, which do not appreciably alter the nature of the encoded protein. Accordingly, in the context of nucleic acid sequences, "substantially the same" means at least 65% identity between two or more sequences. Preferably, the term refers to at least 70% identity between two or more sequences, more preferably at least 75% identity, more preferably at least 80% identity, more preferably at least 85% identity, more preferably at least 90% identity, more preferably at least 91% identity, more preferably at least 92% identity, more preferably at least 93% identity, more preferably at least 94% identity, more preferably at least 95% identity, more preferably at least 96% identity, more preferably at least 97% identity, more preferably at least 98% identity, and more preferably at least 99% or greater identity. The percent identity between two sequences is a function of the number of identical positions shared by the sequences (i.e., % homology=# of identical positions/total # of positions.times.100), taking into account the number of gaps, and the length of each gap, which need to be introduced for optimal alignment of the two sequences. The percent identity between two nucleotide or amino acid sequences may e.g. be determined using the algorithm of E. Meyers and W. Miller, Comput. Appl. Biosci 4, 11-17 (1988) which has been incorporated into the ALIGN program (version 2.0), using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4. In addition, the percent identity between two amino acid sequences may be determined using the Needleman and Wunsch, J. Mol. Biol. 48, 444-453 (1970) algorithm.
[0035] The degree of variation that may occur within the amino acid sequence of a protein without having a substantial effect on protein function is much lower than that of a nucleic acid sequence, since the same degeneracy principles do not apply to amino acid sequences. Accordingly, in the context of an antibody or antigen-binding fragment, "substantially the same" means antibodies or antigen-binding fragments having 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to the antibodies or antigen-binding fragments described. Other embodiments include proTGF.beta.1-GARP complex-selective antibodies, or antigen-binding fragments, that have framework, scaffold, or other non-binding regions that do not share significant identity with the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments described herein, but do incorporate one or more CDRs or other sequences needed to confer binding that are 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identical to such sequences described herein.
[0036] "Binding affinity" generally refers to the strength of the sum total of non-covalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, "binding affinity" refers to intrinsic binding affinity which reflects a 1:1 interaction between members of a binding pair (e.g., antibody and antigen). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (K.sub.D). Affinity can be measured and/or expressed in a number of ways known in the art, including, but not limited to, equilibrium dissociation constant (K.sub.D), and equilibrium association constant (K.sub.A). The K.sub.D is calculated from the quotient of k.sub.off/k.sub.on, whereas K.sub.A is calculated from the quotient of k.sub.on/k.sub.off. k.sub.m refers to the association rate constant of, e.g., an antibody to an antigen, and k.sub.off refers to the dissociation of, e.g., an antibody to an antigen. The k.sub.on and k.sub.off can be determined by techniques known to one of ordinary skill in the art, such as biolayer interferometry.
[0037] The term "subject" refers to human and non-human animals, including all vertebrates, e.g., mammals and non-mammals, such as non-human primates, mice, rabbits, sheep, dogs, cats, horses, cows, chickens, amphibians, and reptiles. In many embodiments of the described methods, the subject is a human.
proTGF.beta.1-GARP Complex-Selective Antibodies and Antigen-Binding Fragments
[0038] Described herein are isolated monoclonal antibodies or antigen-binding fragments that are proTGF.beta.1-GARP complex-selective antibodies. As used herein, the term "proTGF.beta.1-GARP complex-selective antibody" refers to an antibody with distinct affinity, specificity, and activity. proTGF.beta.1-GARP complex-selective antibodies may have: (1) a dissociation constant (Kd) of less than or equal to 1 nM for human proTGF.beta.1 when the proTGF.beta.1 is in a complex with human GARP in solution (e.g., as measured using a cell-free assay); (2) an inhibitory concentration (IC50) of less than or equal to 10 nM for inhibition of TGF.beta.1 growth factor release from cell-associated proTGF.beta.1-GARP complexes; (3) a greater than 100 fold selectivity (as measured by binding affinity, i.e., Kd value) for proTGF.beta.1-GARP complex over each of a TGF.beta.1 growth factor domain, a TGF.beta.2 growth factor domain, a TGF.beta.3 growth factor domain, proTGF.beta.1 covalently associated with LTBP1, proTGF.beta.1 covalently associated with LTBP3, and proTGF.beta.1 covalently associated with LRRC33; and (4) a lack of affinity for proTGF.beta.1 when not in a complex with GARP. In some cases, proTGF.beta.1-GARP complex-selective antibodies also have a greater than 100 fold selectivity for proTGF.beta.1-GARP complex over proTGF.beta.1 covalently associated with LTBP2 and/or LTBP4. The general structure of an antibody molecule comprises an antigen binding domain, which includes heavy and light chains, and the Fc domain, which serves a variety of functions, including complement fixation and binding antibody receptors.
[0039] The described proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments include all isotypes, IgA, IgD, IgE, IgG and IgM, and synthetic multimers of the four-chain immunoglobulin structure. The described antibodies or antigen-binding fragments also include the IgY isotype generally found in hen or turkey serum and hen or turkey egg yolk.
[0040] The proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments may be derived from any species by recombinant means. For example, the antibodies or antigen-binding fragments may be mouse, rat, goat, horse, swine, bovine, chicken, rabbit, camelid, donkey, human, or chimeric versions thereof. For use in administration to humans, non-human derived antibodies or antigen-binding fragments may be genetically or structurally altered to be less antigenic upon administration to a human patient.
[0041] In some embodiments, the antibodies or antigen-binding fragments are chimeric. As used herein, the term "chimeric" refers to an antibody, or antigen-binding fragment thereof, having at least some portion of at least one variable domain derived from the antibody amino acid sequence of a non-human mammal, a rodent, or a reptile, while the remaining portions of the antibody, or antigen-binding fragment thereof, are derived from a human.
[0042] In some embodiments, the antibodies are humanized antibodies. Humanized antibodies may be chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab', F(ab')2 or other antigen-binding subsequences of antibodies) that contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a complementary-determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity, and capacity. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or substantially all of the framework regions are those of a human immunoglobulin sequence. The humanized antibody may include at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin.
[0043] The antibodies or antigen-binding fragments described herein can occur in a variety of forms, but will include one or more of the antibody CDRs shown in Table 1.
[0044] In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments are human IgG, or derivatives thereof. While the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments exemplified herein are human, the antibodies or antigen-binding fragments exemplified may be chimerized.
[0045] In some embodiments are provided proTGF.beta.1-GARP complex-selective antibodies comprising a heavy chain comprising a CDR1, a CDR2, and a CDR3 of any one of the antibodies described in Table 1 and a light chain comprising a CDR1, a CDR2, and a CDR3 of any one of the antibodies described in Table 1.
[0046] In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain CDR1 comprising SEQ ID NO: 4, a heavy chain CDR2 comprising SEQ ID NO: 5, a heavy chain CDR3 comprising SEQ ID NO: 6, a light chain CDR1 comprising SEQ ID NO: 7, a light chain CDR2 comprising SEQ ID NO: 8, and a light chain CDR3 comprising SEQ ID NO: 9. This proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment may comprise human framework sequences. This proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment may bind to the proTGF.beta.1 of the proTGF.beta.1-GARP complex with an affinity of 880 pM or less, may inhibit Treg function in vitro and may inhibit the activation of TGF.beta.1. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain substantially the same as, or identical to, SEQ ID NO: 16 and a light chain substantially the same as, or identical to, SEQ ID NO: 17. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain variable region substantially the same as, or identical to, amino acid sequence 1-118 of SEQ ID NO: 16 and a light chain variable region substantially the same as, or identical to, amino sequence 1-107 of SEQ ID NO: 17. The heavy chain and light chain variable regions of antibodies discussed in this paragraph are suitable for inclusion in bispecific constructs in which one arm is a proTGF.beta.1-GARP complex-selective antibody arm.
[0047] In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain CDR1 comprising SEQ ID NO: 10, a heavy chain CDR2 comprising SEQ ID NO: 11, a heavy chain CDR3 comprising SEQ ID NO: 12, a light chain CDR1 comprising SEQ ID NO: 13, a light chain CDR2 comprising SEQ ID NO: 14, and a light chain CDR3 comprising SEQ ID NO: 15. This proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment may comprise human framework sequences. This proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment may bind to proTGF.beta.1 of a proTGF.beta.1-GARP complex with an affinity of 880 pM or less, may inhibit Treg function in vitro and may and may inhibit the activation of TGF.beta.1. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain substantially the same as, or identical to, SEQ ID NO: 18 and a light chain substantially the same as, or identical to, SEQ ID NO: 19. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments comprise a heavy chain variable region substantially the same as, or identical to, amino acid sequence 1-121 of SEQ ID NO: 18 and a light chain variable region substantially the same as, or identical to, amino sequence 1-110 of SEQ ID NO: 19. The heavy chain and light chain variable regions of antibodies discussed in this paragraph are suitable for inclusion in bispecific constructs in which one arm is a proTGF.beta.1-GARP complex-selective antibody arm.
[0048] The proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments may have amino acid sequences having at least 70% identity, at least 75% identity, at least 80% identity, at least 85% identity, at least 90% identity, at least 91% identity, at least 92% identity, at least 93% identity, at least 94% identity, at least 95% identity, at least 96% identity, at least 97% identity, at least 98% identity, and at least 99% or greater identity to the CDR amino acid sequences of SEQ ID NOS: 4-15 and variable region amino acid sequences of SEQ ID NOS: 16-19.
[0049] Also disclosed are isolated polynucleotides that encode the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments of the present disclosure. The isolated polynucleotides capable of encoding the variable domain segments provided herein may be included on the same, or different, vectors to produce antibodies or antigen-binding fragments.
[0050] Polynucleotides encoding recombinant antigen-binding proteins also are within the scope of the disclosure. In some embodiments, the polynucleotides described (and the peptides they encode) include a leader sequence. Any leader sequence known in the art may be employed. The leader sequence may include, but is not limited to, a restriction site or a translation start site.
[0051] The proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein include variants having single or multiple amino acid substitutions, deletions, or additions that retain the biological properties (e.g., binding affinity or immune effector activity) of the described proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments. These variants may include: (a) variants in which one or more amino acid residues are substituted with conservative or nonconservative amino acids, (b) variants in which one or more amino acids are added to or deleted from the polypeptide, (c) variants in which one or more amino acids include a substituent group, and (d) variants in which the polypeptide is fused with another peptide or polypeptide such as a fusion partner, a protein tag or other chemical moiety, that may confer useful properties to the polypeptide, such as, for example, an epitope for an antibody, a polyhistidine sequence, a biotin moiety and the like. Antibodies or antigen-binding fragments described herein may include variants in which amino acid residues from one species are substituted for the corresponding residue in another species, either at the conserved or nonconserved positions. In other embodiments, amino acid residues at nonconserved positions are substituted with conservative or nonconservative residues. The techniques for obtaining these variants, including genetic (deletions, mutations, etc.), chemical, and enzymatic techniques, are known to persons having ordinary skill in the art.
[0052] The proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein may embody several antibody isotypes, such as IgM, IgD, IgG, IgA and IgE. In some embodiments the antibody isotype is IgG1, IgG2, IgG3, or IgG4 isotype, preferably IgG1 isotype. Antibody or antigen-binding fragment thereof specificity is largely determined by the amino acid sequence, and arrangement, of the CDRs. Therefore, the CDRs of one isotype may be transferred to another isotype without altering antigen specificity. Alternatively, techniques have been established to cause hybridomas to switch from producing one antibody isotype to another (isotype switching) without altering antigen specificity. Accordingly, such antibody isotypes are within the scope of the described antibodies or antigen-binding fragments.
[0053] The proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein have binding affinities for proTGF.beta.1 of a proTGF.beta.1-GARP complex that include a dissociation constant (K.sub.D) of less than about 880 pM. The affinity of the described proTGF.beta.1-GARP complex-selective antibodies, or antigen-binding fragments, may be determined by a variety of methods known in the art, such as biolayer interferometry, surface plasmon resonance or ELISA-based methods. Assays for measuring affinity by biolayer interferometry include assays performed using an OctetRed 384 where the assay is performed at room temperature (e.g. at or near 25.degree. C.), wherein the antibody capable of binding to proTGF.beta.1 of a proTGF.beta.1-GARP complex is captured on the streptavidin biosensors loaded with biotinylated proTGF.beta.1-GARP complex.
[0054] Also provided are vectors comprising the polynucleotides described herein. The vectors can be expression vectors. Recombinant expression vectors containing a sequence encoding a polypeptide of interest are thus contemplated as within the scope of this disclosure. The expression vector may contain one or more additional sequences such as but not limited to regulatory sequences (e.g., promoter, enhancer), a selection marker, and a polyadenylation signal. Vectors for transforming a wide variety of host cells are well known and include, but are not limited to, plasmids, phagemids, cosmids, baculoviruses, bacmids, bacterial artificial chromosomes (BACs), yeast artificial chromosomes (YACs), as well as other bacterial, yeast and viral vectors.
[0055] Recombinant expression vectors within the scope of the description include synthetic, genomic, or cDNA-derived nucleic acid fragments that encode at least one recombinant protein which may be operably linked to suitable regulatory elements. Such regulatory elements may include a transcriptional promoter, sequences encoding suitable mRNA ribosomal binding sites, and sequences that control the termination of transcription and translation. Expression vectors, especially mammalian expression vectors, may also include one or more nontranscribed elements such as an origin of replication, a suitable promoter and enhancer linked to the gene to be expressed, other 5' or 3' flanking nontranscribed sequences, 5' or 3' nontranslated sequences (such as necessary ribosome binding sites), a polyadenylation site, splice donor and acceptor sites, or transcriptional termination sequences. An origin of replication that confers the ability to replicate in a host may also be incorporated.
[0056] The transcriptional and translational control sequences in expression vectors to be used in transforming vertebrate cells may be provided by viral sources. Exemplary vectors may be constructed as described by Okayama and Berg, 3 Mol. Cell. Biol. 280 (1983).
[0057] In some embodiments, the antibody- or antigen-binding fragment-coding sequence is placed under control of a powerful constitutive promoter, such as the promoters for the following genes: hypoxanthine phosphoribosyl transferase (HPRT), adenosine deaminase, pyruvate kinase, beta-actin, human myosin, human hemoglobin, human muscle creatine, and others. In addition, many viral promoters function constitutively in eukaryotic cells and are suitable for use with the described embodiments. Such viral promoters include without limitation, Cytomegalovirus (CMV) immediate early promoter, the early and late promoters of SV40, the Mouse Mammary Tumor Virus (MMTV) promoter, the long terminal repeats (LTRs) of Maloney leukemia virus, Human Immunodeficiency Virus (HIV), Epstein Barr Virus (EBV), Rous Sarcoma Virus (RSV), and other retroviruses, and the thymidine kinase promoter of Herpes Simplex Virus. In one embodiment, the proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment thereof coding sequence is placed under control of an inducible promoter such as the metallothionein promoter, tetracycline-inducible promoter, doxycycline-inducible promoter, promoters that contain one or more interferon-stimulated response elements (ISRE) such as protein kinase R 2',5'-oligoadenylate synthetases, Mx genes, ADAR1, and the like.
[0058] Vectors described herein may contain one or more Internal Ribosome Entry Site(s) (IRES). Inclusion of an IRES sequence into fusion vectors may be beneficial for enhancing expression of some proteins. In some embodiments the vector system will include one or more polyadenylation sites (e.g., SV40), which may be upstream or downstream of any of the aforementioned nucleic acid sequences. Vector components may be contiguously linked, or arranged in a manner that provides optimal spacing for expressing the gene products (i.e., by the introduction of "spacer" nucleotides between the ORFs), or positioned in another way. Regulatory elements, such as the IRES motif, may also be arranged to provide optimal spacing for expression.
[0059] The vectors may comprise selection markers, which are well known in the art. Selection markers include positive and negative selection markers, for example, antibiotic resistance genes (e.g., neomycin resistance gene, a hygromycin resistance gene, a kanamycin resistance gene, a tetracycline resistance gene, a penicillin resistance gene), glutamate synthase genes, HSV-TK, HSV-TK derivatives for ganciclovir selection, or bacterial purine nucleoside phosphorylase gene for 6-methylpurine selection (Gadi et al., 7 Gene Ther. 1738-1743 (2000)). A nucleic acid sequence encoding a selection marker or the cloning site may be upstream or downstream of a nucleic acid sequence encoding a polypeptide of interest or cloning site.
[0060] The vectors described herein may be used to transform various cells with the genes encoding the described antibodies or antigen-binding fragments. For example, the vectors may be used to generate proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment-producing cells. Thus, another aspect features host cells transformed with vectors comprising a nucleic acid sequence encoding an antibody or antigen-binding fragment thereof that specifically binds proTGF.beta.1 of a proTGF.beta.1-GARP complex, such as the antibodies or antigen-binding fragments described and exemplified herein.
[0061] Numerous techniques are known in the art for the introduction of foreign genes into cells and may be used to construct the recombinant cells for purposes of carrying out the described methods, in accordance with the various embodiments described and exemplified herein. The technique used should provide for the stable transfer of the heterologous gene sequence to the host cell, such that the heterologous gene sequence is heritable and expressible by the cell progeny, and so that the necessary development and physiological functions of the recipient cells are not disrupted. Techniques which may be used include but are not limited to chromosome transfer (e.g., cell fusion, chromosome mediated gene transfer, micro cell mediated gene transfer), physical methods (e.g., transfection, spheroplast fusion, microinjection, electroporation, liposome carrier), viral vector transfer (e.g., recombinant DNA viruses, recombinant RNA viruses) and the like (described in Cline, 29 Pharmac. Ther. 69-92 (1985)). Calcium phosphate precipitation and polyethylene glycol (PEG)-induced fusion of bacterial protoplasts with mammalian cells may also be used to transform cells.
[0062] Cells suitable for use in the expression of the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein are preferably eukaryotic cells, more preferably cells of plant, rodent, or human origin, for example but not limited to NSO, CHO, CHOK1, perC.6, Tk-ts13, BHK, HEK293 cells, COS-7, T98G, CV-1/EBNA, L cells, C127, 3T3, HeLa, NS1, Sp2/0 myeloma cells, and BHK cell lines, among others. In addition, expression of antibodies may be accomplished using hybridoma cells. Methods for producing hybridomas are well established in the art.
[0063] Cells transformed with expression vectors described herein may be selected or screened for recombinant expression of the antibodies or antigen-binding fragments described herein. Recombinant-positive cells are expanded and screened for subclones exhibiting a desired phenotype, such as high level expression, enhanced growth properties, or the ability to yield proteins with desired biochemical characteristics, for example, due to protein modification or altered post-translational modifications. These phenotypes may be due to inherent properties of a given subclone or to mutation. Mutations may be effected through the use of chemicals, UV-wavelength light, radiation, viruses, insertional mutagens, inhibition of DNA mismatch repair, or a combination of such methods.
Methods of using proTGF.beta.1-GARP Complex-Selective Antibodies for Treatment
[0064] Provided herein are proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments thereof for use in therapy. In particular, these antibodies or antigen-binding fragments may be useful in treating cancer. As described above, active TGF.beta.1 released from Tregs inhibit the actions of other T cells. Thus, inhibiting the TGF.beta.1-mediated immunosuppressive function represents an attractive approach for boosting an immune response against a variety of cancers. The proTGF.beta.1-GARP complex-selective antibodies can be used to treat both solid tumors, as well as hematological cancers, including leukemia.
[0065] The antibodies for use in these methods include those described herein above, for example a proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment with the features set out in Table 1, for example the CDRs or variable domain sequences, and in the further discussion of these antibodies.
[0066] In some embodiments described herein, immune effector properties of the proTGF.beta.1-GARP complex-selective antibodies may be modulated through Fc modifications by techniques known to those skilled in the art. For example, Fc effector functions such as Clq binding, complement dependent cytotoxicity (CDC), antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cell-mediated phagocytosis (ADCP), down regulation of cell surface receptors (e.g., B cell receptor; BCR), etc. may be provided and/or controlled by modifying residues in the Fc responsible for these activities.
[0067] "Antibody-dependent cell-mediated cytotoxicity" or "ADCC" refers to a cell-mediated reaction in which non-specific cytotoxic cells that express Fc receptors (FcRs) (e.g. Natural Killer (NK) cells, neutrophils, and macrophages) recognize bound antibody on a target cell and subsequently cause lysis of the target cell.
[0068] The ability of monoclonal antibodies to induce ADCC can be enhanced by engineering their oligosaccharide component. Human IgG1 or IgG3 are N-glycosylated at Asn297 with the majority of the glycans in the well-known biantennary G0, G0F, G1, G1F, G2 or G2F forms. Antibodies produced by non-engineered CHO cells typically have a glycan fucose content of about at least 85%. The removal of the core fucose from the biantennary complex-type oligosaccharides attached to the Fc regions enhances the ADCC of antibodies via improved Fc.gamma.RTIIa binding without altering antigen binding or CDC activity. Such mAbs can be achieved using different methods reported to lead to the successful expression of relatively high defucosylated antibodies bearing the biantennary complex-type of Fc oligosaccharides such as control of culture osmolality (Konno et al., Cytotechnology 64:249-65, 2012), application of a variant CHO line Lec13 as the host cell line (Shields et al., J Biol Chem 277:26733-26740, 2002), application of a variant CHO line EB66 as the host cell line (Olivier et al., MAbs; 2(4), 2010; Epub ahead of print; PMID:20562582), application of a rat hybridoma cell line YB2/0 as the host cell line (Shinkawa et al., J Biol Chem 278:3466-3473, 2003), introduction of small interfering RNA specifically against the .alpha. 1,6-fucosyltrasferase (FUT8) gene (Mori et al., Biotechnol Bioeng 88:901-908, 2004), or coexpression of beta-1,4-N-acetylglucosaminyltransferase III and Golgi alpha-mannosidase II or a potent alpha-mannosidase I inhibitor, kifunensine (Ferrara et al., J Biol Chem 281:5032-5036, 2006, Ferrara et al., Biotechnol Bioeng 93:851-861, 2006; Xhou et al., Biotechnol Bioeng 99:652-65, 2008).
[0069] In some embodiments described herein, ADCC elicited by the proTGF.beta.1-GARP complex-selective antibodies may also be enhanced by certain substitutions in the antibody Fc. Exemplary substitutions are for example substitutions at amino acid positions 256, 290, 298, 312, 356, 330, 333, 334, 360, 378 or 430 (residue numbering according to the EU index) as described in U.S. Pat. No. 6,737,056.
Pharmaceutical Compositions and Administration
[0070] The pharmaceutical compositions provided herein comprise: a) an effective amount of a proTGF.beta.1-GARP complex-selective antibody or antibody fragment of the present invention, and b) a pharmaceutically acceptable carrier, which may be inert or physiologically active. In preferred embodiments, the proTGF.beta.1-GARP complex-selective antibody is a proTGF.beta.1-GARP complex-selective antibody as described herein, or an antigen-binding fragment thereof. As used herein, the term "pharmaceutically acceptable carriers" includes any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, and the like that are physiologically compatible. Examples of suitable carriers, diluents and/or excipients include one or more of water, saline, phosphate buffered saline, dextrose, glycerol, ethanol, and the like, as well as any combination thereof. In many cases, it will be preferable to include isotonic agents, such as sugars, polyalcohols, or sodium chloride in the composition. In particular, relevant examples of suitable carrier include: (1) Dulbecco's phosphate buffered saline, pH.about.7.4, containing or not containing about 1 mg/mL to 25 mg/mL human serum albumin, (2) 0.9% saline (0.9% w/v sodium chloride (NaCl)), and (3) 5% (w/v) dextrose; and may also contain an antioxidant such as tryptamine and a stabilizing agent such as Tween 20.RTM..
[0071] The compositions herein may also contain a further therapeutic agent, as necessary for the particular disorder being treated. Preferably, the proTGF.beta.1-GARP complex-selective antibodies or antibody fragment and the supplementary active compound will have complementary activities that do not adversely affect each other. In a preferred embodiment, the further therapeutic agent is cytarabine, an anthracycline, histamine dihydrochloride, or interleukin 2. In a preferred embodiment, the further therapeutic agent is a chemotherapeutic agent.
[0072] The compositions of the invention may be in a variety of forms. These include for example liquid, semi-solid, and solid dosage forms, but the preferred form depends on the intended mode of administration and therapeutic application. Typical preferred compositions are in the form of injectable or infusible solutions. The preferred mode of administration is parenteral (e.g. intravenous, intramuscular, intraperitoneal, subcutaneous). In a preferred embodiment, the compositions of the invention are administered intravenously as a bolus or by continuous infusion over a period of time. In another preferred embodiment, they are injected by intramuscular, subcutaneous, intra-articular, intrasynovial, intratumoral, peritumoral, intralesional, or perilesional routes, to exert local as well as systemic therapeutic effects.
[0073] Sterile compositions for parenteral administration can be prepared by incorporating the antibody, antibody fragment or antibody conjugate of the present invention in the required amount in the appropriate solvent, followed by sterilization by microfiltration. As solvent or vehicle, there may be used water, saline, phosphate buffered saline, dextrose, glycerol, ethanol, and the like, as well as combination thereof. In many cases, it will be preferable to include isotonic agents, such as sugars, polyalcohols, or sodium chloride in the composition. These compositions may also contain adjuvants, in particular wetting, isotonizing, emulsifying, dispersing and stabilizing agents. Sterile compositions for parenteral administration may also be prepared in the form of sterile solid compositions which may be dissolved at the time of use in sterile water or any other injectable sterile medium.
[0074] The proTGF.beta.1-GARP complex-selective antibodies or antibody fragment may also be orally administered. As solid compositions for oral administration, tablets, pills, powders (gelatine capsules, sachets) or granules may be used. In these compositions, the active ingredient according to the invention is mixed with one or more inert diluents, such as starch, cellulose, sucrose, lactose or silica, under an argon stream. These compositions may also comprise substances other than diluents, for example one or more lubricants such as magnesium stearate or talc, a coloring, a coating (sugar-coated tablet) or a glaze.
[0075] As liquid compositions for oral administration, there may be used pharmaceutically acceptable solutions, suspensions, emulsions, syrups and elixirs containing inert diluents such as water, ethanol, glycerol, vegetable oils or paraffin oil. These compositions may comprise substances other than diluents, for example wetting, sweetening, thickening, flavoring or stabilizing products.
[0076] The doses depend on the desired effect, the duration of the treatment and the route of administration used; they are generally between 5 mg and 1000 mg per day orally for an adult with unit doses ranging from 1 mg to 250 mg of active substance. In general, the doctor will determine the appropriate dosage depending on the age, weight and any other factors specific to the subject to be treated.
[0077] In a preferred embodiment, proTGF.beta.1-GARP complex-selective antibodies or antibody fragments of the invention are used for the treatment of a hyperproliferative disorder in a mammal. In a more preferred embodiment, one of the pharmaceutical compositions disclosed above, and which contains a proTGF.beta.1-GARP complex-selective antibody or antibody fragment of the invention, is used for the treatment of a hyperproliferative disorder in a mammal. In one embodiment, the disorder is a cancer. A variety of different cancerous tumors such as for an adrenocortical carcinoma, anal cancer, bladder cancer, brain tumor, glioma, breast carcinoma, carcinoid tumor, cervical cancer, colon carcinoma, endometrial cancer, esophageal cancer, extrahepatic bile duct cancer, Ewings tumor, extracranial germ cell tumor, eye cancer, gall bladder cancer, gastric cancer, germ cell tumor, gestational trophoblastic tumor, head and neck cancer, hypopharyngeal cancer, islet cell carcinoma, kidney cancer, laryngeal cancer, leukemia, lip and oral cavity cancer, liver cancer, lung cancer, lymphoma, melanoma, mesothelioma, merkel cell carcinoma, metastatic squamous head and neck cancer, myeloma, neoplasm, nasopharyngeal cancer, neuroblastoma, oral cancer, oropharyngeal cancer, osteosarcoma, ovarian cancer, pancreatic cancer, sinus and nasal cancer, parathyroid cancer, penile cancer, pheochromocytoma cancer, pituitary cancer, plasma cell neoplasm, prostate cancer, rhabdomyosarcoma, rectal cancer, renal cell carcinoma, salivary gland cancer, skin cancer, Kaposi's sarcoma, T-cell lymphoma, soft tissue sarcoma, stomach cancer, testicular cancer, thymoma, thyroid cancer, urethral cancer, uterine cancer, vaginal cancer, vulvar cancer, or Wilms' tumor can be treated with the antibodies described herein.
[0078] In treating any of the foregoing cancers, the treatment methods that are provided can be utilized to inhibit further tumor growth, induce tumor regression, increase progression-free survival and/or extend overall survival in an individual that has a tumor. In some embodiments, the proTGF.beta.1-GARP complex-selective antibodies can also delay or prevent the onset of metastasis. Progress in treatment can be monitored using various methods. For instance, inhibition can result in reduced tumor size and/or a decrease in metabolic activity within the tumor. Both of these parameters can be measured by MRI or PET scans for example. Inhibition can also be monitored by biopsy to ascertain the level of necrosis, tumor cell death and the level of vascularity within the tumor. The extent of metastasis can be monitored using known methods. Accordingly, the pharmaceutical compositions of the invention are useful in the treatment or prevention of metastasis of a variety of cancers, including (but not limited to) the following: melanoma, lung, head and neck, renal cell, colorectal, breast, prostate, endometrial, bladder, kidney, esophageal, testicular, ovarian, squamous cell carcinoma (e.g., squamous cell carcinoma of the head and neck--SCCHN), uveal melanoma, follicular lymphoma, cervical, brain, pancreatic, liver, lymphoma, Hodgkin's disease, multiple myeloma, gastric, and astrocyctic.
[0079] Similarly, further provided herein is a method for inhibiting the growth of selected cell populations comprising contacting TGF.beta.1-expressing immune cells with an effective amount of a proTGF.beta.1-GARP complex-selective antibody or antibody fragment of the present disclosure, either alone or in combination with other therapeutic agents. In preferred embodiments, the proTGF.beta.1-GARP complex-selective antibody is a proTGF.beta.1-GARP complex-selective antibody as described herein, or an antigen-binding fragment thereof. In a preferred embodiment, the further therapeutic agent is an immunotherapy i.e., an immunostimulatory agent that induces or enhances an immune response. Such agents can include, for example: 1) activators of dendritic cells, 2) vaccine adjuvants, 3) T cell stimulators, 4) inhibitors of immune checkpoints, and 5) inhibitors of suppressive cells, cytokines and/or enzymes. Thus, in one embodiment, an antibody is administered with a vaccine.
[0080] For clinical use, a therapeutically effective amount of the proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment is administered to a subject in need thereof. For example, the proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments thereof may be useful in the treatment of cancerous tumors that contain TGF.beta.1-positive immune cells. In preferred embodiments the proTGF.beta.1-GARP complex-selective antibody is a proTGF.beta.1-GARP complex-selective antibody as described herein, or an antigen-binding fragment thereof. In some embodiments, the subject is a mammal, preferably a human. In some embodiments, the proTGF.beta.1-GARP complex-selective antibody or antigen-binding fragment will be administered as a solution that has been tested for sterility.
[0081] Dosage regimens in the above methods of treatment and uses are adjusted to provide the optimum desired response (e.g., a therapeutic response). For example, a single bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. Parenteral compositions may be formulated in dosage unit form for ease of administration and uniformity of dosage.
[0082] The efficient dosages and the dosage regimens for the proTGF.beta.1-GARP complex-selective antibodies and fragments depend on the disease or condition to be treated and may be determined by one skilled in the art. An exemplary, non-limiting range for a therapeutically effective amount of a compound of the present invention is about 0.001-10 mg/kg, such as about 0.001-5 mg/kg, for example about 0.001-2 mg/kg, such as about 0.001-1 mg/kg, for instance about 0.001, about 0.01, about 0.1, about 1 or about 10 mg/kg.
[0083] A physician or veterinarian having ordinary skill in the art may readily determine and prescribe the effective amount of the pharmaceutical composition required. For example, the physician or veterinarian could start doses of the proTGF.beta.1-GARP complex-selective antibody or fragment employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved. In general, a suitable daily dose of a proTGF.beta.1-GARP complex-selective antibody of the present invention will be that amount of the compound which is the lowest dose effective to produce a therapeutic effect. Administration may e.g. be parenteral, such as intravenous, intramuscular or subcutaneous. In one embodiment, the proTGF.beta.1-GARP complex-selective antibody or fragment may be administered by infusion in a weekly dosage of calculated by mg/m.sup.2. Such dosages can, for example, be based on the mg/kg dosages provided above according to the following: dose (mg/kg).times.70. Such administration may be repeated, e.g., 1 to 8 times, such as 3 to 5 times. The administration may be performed by continuous infusion over a period of from 2 to 24 hr, such as of from 2 to 12 hr. In one embodiment, the proTGF.beta.1-GARP complex-selective antibody or fragment may be administered by slow continuous infusion over a long period, such as more than 24 hours, in order to reduce toxic side effects.
[0084] In one embodiment, the proTGF.beta.1-GARP complex-selective antibody or fragment may be administered in a weekly dosage calculated as a fixed dose for up to eight times, such as from four to six times when given once a week. Such regimen may be repeated one or more times as necessary, for example, after six months or twelve months. Such fixed dosages can, for example, be based on the mg/kg dosages provided above, with a body weight estimate of 70 kg. The dosage may be determined or adjusted by measuring the amount of proTGF.beta.1-GARP complex-selective antibody of the present invention in the blood upon administration by for instance taking out a biological sample and using anti-idiotypic antibodies which target the antigen binding region of the proTGF.beta.1-GARP complex-selective antibodies of the present invention.
[0085] In one embodiment, the proTGF.beta.1-GARP complex-selective antibody or fragment may be administered by maintenance therapy, such as, e.g., once a week for a period of six months or more.
[0086] A proTGF.beta.1-GARP complex-selective antibody or fragment may also be administered prophylactically in order to reduce the risk of developing cancer, delay the onset of the occurrence of an event in cancer progression, and/or reduce the risk of recurrence when a cancer is in remission.
[0087] The proTGF.beta.1-GARP complex-selective antibodies and fragments thereof as described herein may also be administered in combination therapy, i.e., combined with other therapeutic agents relevant for the disease or condition to be treated. Accordingly, in one embodiment, the antibody-containing medicament is for combination with one or more further therapeutic agent, such as a chemotherapeutic agent. In some embodiments, the other therapeutic agents include, but are not limited to, anti-neoplastic agents including alkylating agents including: nitrogen mustards, such as mechlorethamine, cyclophosphamide, ifosfamide, melphalan and chlorambucil; nitrosoureas, such as carmustine (BCNU), lomustine (CCNU), and semustine (methyl-CCNU); Temodal.TM. (temozolamide), ethylenimines/methylmelamine such as thriethylenemelamine (TEM), triethylene, thiophosphoramide (thiotepa), hexamethylmelamine (HMM, altretamine); alkyl sulfonates such as busulfan; triazines such as dacarbazine (DTIC); antimetabolites including folic acid analogs such as methotrexate and trimetrexate, pyrimidine analogs such as 5-fluorouracil (5FU), fluorodeoxyuridine, gemcitabine, cytosine arabinoside (AraC, cytarabine), 5-azacytidine, 2,2'-difluorodeoxycytidine, purine analogs such as 6-mercaptopurine, 6-thioguanine, azathioprine, 2'-deoxycoformycin (pentostatin), erythrohydroxynonyladenine (EHNA), fludarabine phosphate, and 2-chlorodeoxyadenosine (cladribine, 2-CdA); natural products including antimitotic drugs such as paclitaxel, vinca alkaloids including vinblastine (VLB), vincristine, and vinorelbine, taxotere, estramustine, and estramustine phosphate; pipodophylotoxins such as etoposide and teniposide; antibiotics such as actimomycin D, daunomycin (rubidomycin), doxorubicin, mitoxantrone, idarubicin, bleomycins, plicamycin (mithramycin), mitomycinC, and actinomycin; enzymes such as L-asparaginase; biological response modifiers such as interferon-alpha, IL-2, G-CSF and GM-CSF; miscellaneous agents including platinum coordination complexes such as cisplatin and carboplatin, anthracenediones such as mitoxantrone, substituted urea such as hydroxyurea, methylhydrazine derivatives including N-methylhydrazine (MIH) and procarbazine, adrenocortical suppressants such as mitotane (o,p-DDD) and aminoglutethimide; hormones and antagonists including adrenocorticosteroid antagonists such as prednisone and equivalents, dexamethasone and aminoglutethimide; Gemzar.TM. (gemcitabine), progestin such as hydroxyprogesterone caproate, medroxyprogesterone acetate and megestrol acetate; estrogen such as diethylstilbestrol and ethinyl estradiol equivalents; antiestrogen such as tamoxifen; androgens including testosterone propionate and fluoxymesterone/equivalents; antiandrogens such as flutamide, gonadotropin-releasing hormone analogs and leuprolide; and non-steroidal antiandrogens such as flutamide. Therapies targeting epigenetic mechanism including, but not limited to, histone deacetylase inhibitors, demethylating agents (e.g., Vidaza) and release of transcriptional repression (ATRA) therapies can also be combined with the proTGF.beta.1-GARP complex-selective antibodies.
[0088] Additional specific examples of chemotherapeutic agents include, taxol, taxenes (e.g., docetaxel and Taxotere), modified paclitaxel (e.g., Abraxane and Opaxio) doxorubicin, Avastin.RTM., Sutent, Nexavar, and other multikinase inhibitors, cisplatin and carboplatin, etoposide, gemcitabine, and vinblastine. Specific inhibitors of other kinases can also be used in combination with the proTGF.beta.1-GARP complex-selective antibodies, including but not limited to, MAPK pathway inhibitors (e.g., inhibitors of ERK, JNK and p38), Pl3kinase/AKT inhibitors and Pim inhibitors. Other inhibitors include Hsp90 inhibitors, proteasome inhibitors (e.g., Velcade) and multiple mechanism of action inhibitors such as Trisenox.
[0089] Such combined administration may be simultaneous, separate or sequential, in any order. For simultaneous administration the agents may be administered as one composition or as separate compositions, as appropriate.
[0090] In one embodiment, a proTGF.beta.1-GARP complex-selective antibody or fragment thereof is combined with an agent that stimulates antigen-presenting cells. Examples of such agents include various CD40 agonists, such as an agonist anti-CD40 antibody or CD40L.
[0091] Some methods involve administering a proTGF.beta.1-GARP complex-selective antibody or fragment thereof with a vaccine adjuvant. Such adjuvants include, for instance, IL-12, and various Toll Like Receptor (TLR) agonists, including CpG (a TLR 9 agonist), monophosphoryl lipid A (MPL--a TLR4 agonist), PolyI:C or PolyICLC (TLR3 agonist), and resiquimod and 852A (TLR 7/8 agonists).
[0092] In other therapeutic approaches, a proTGF.beta.1-GARP complex-selective antibody is administered in combination with T cell growth factors such as IL-15 and/or IL-17, or activators of these molecules. In related methods, a T cell stimulator is combined with a proTGF.beta.1-GARP complex-selective antibody. Such stimulators include agonists of 4-1BB, such as agonist anti-4-1BB antibodies and 4-1BBL.
[0093] In one embodiment, a proTGF.beta.1-GARP complex-selective antibody or fragment thereof is administered with a T cell checkpoint inhibitor, e.g., molecules that send an inhibitory signal to the immune system. Examples of such agents include inhibitors of PD-1 or PD-L1 (B7-H1), such as anti-PD-1 antibodies, including nivolumab (Bristol-Myers Squibb) and pembrolizumab, also known as MK-3475 (Merck), pidilizumab (Curetech), AMP-224 (Amplimmune), and anti-PD-L1 antibodies, including MPDL3280A (Roche), MDX-1105 (Bristol Myer Squibb), MEDI-4736 (AstraZeneca) and MSB-0010718C (Merck). Other checkpoint inhibitors include antagonists of CTLA-4, such as anti-CTLA-4 antibodies. An exemplary anti-CTLA4 antibody is Yervoy.RTM. (ipilimumab) marketed by Bristol-Myers Squibb. Other exemplary CTLA-4 antibodies include tremelimumab (Pfizer), Ticilimumab (AstraZeneca) and AMGP-224 (Glaxo Smith Kline).
[0094] In yet other methods, a proTGF.beta.1-GARP complex-selective antibody or fragment thereof is administered in combination with an inhibitor of an enzyme that has an immunosuppressive effect. An example is 1-methyl tryptophan (1MT), which is a small molecule inhibitor of indoleamine 2,3-dioxygenase.
[0095] The proTGF.beta.1-GARP complex-selective antibody or fragment thereof can also be used in combination with T-VEC (talimogene laherparepvec) by Amgen.
[0096] In certain embodiments, the proTGF.beta.1-GARP complex-selective antibody or fragment thereof is administered in combination with a bispecific antibody. The bispecific antibody can direct the immune system of a host, in particular the cyotoxic activity of T-cells, against cancer cells.
[0097] A proTGF.beta.1-GARP complex-selective antibody or fragment thereof can also be administered in combination with a variety of targeted therapies. Examples of targeted therapies include, but are not limited to, use of therapeutic antibodies. Exemplary antibodies include, but are not limited to, those which bind to cell surface proteins Her2, CDC20, CDC33, mucin-like glycoprotein, and epidermal growth factor receptor (EGFR) present on tumor cells, OX40, PD-1, CTLA-4, and optionally induce a cytostatic and/or cytotoxic effect on tumor cells displaying these proteins. Exemplary antibodies also include HERCEPTIN.RTM. (trastuzumab), which may be used to treat breast cancer and other forms of cancer, and RITUXAN.RTM. (rituximab), ZEVALIN.TM. (ibritumomab tiuxetan), and LYMPHOCIDE.TM. (epratuzumab), which may be used to treat non-Hodgkin's lymphoma and other forms of cancer. Certain exemplary antibodies also include panitumumab (VECTIBIX.RTM.), ERBITUX.RTM. (IMC-C225); BEXXAR.TM. (iodine 131 tositumomab); KDR (kinase domain receptor) inhibitors; anti VEGF antibodies and antagonists (e.g., Avastin.RTM. and VEGAF-TRAP); anti VEGF receptor antibodies and antigen binding regions; anti-Ang-1 and Ang-2 antibodies and antigen binding regions; antibodies to Tie-2 and other Ang-1 and Ang-2 receptors; Tie-2 ligands; antibodies against Tie-2 kinase inhibitors; inhibitors of Hif-1a, and Campath.TM. (Alemtuzumab). In certain embodiments, cancer therapy agents are polypeptides which selectively induce apoptosis in tumor cells, including, but not limited to, the TNF-related polypeptide TRAIL.
[0098] In one embodiment, a proTGF.beta.1-GARP complex-selective antibody or fragment thereof, as provided herein is used in combination with one or more anti-angiogenic agents that decrease angiogenesis. Certain such agents include, but are not limited to, IL-8 antagonists; Campath, B-FGF; FGF antagonists; Tek antagonists (Cerretti et al., U.S. Publication No. 2003/0162712; Cerretti et al., U.S. Pat. No. 6,413,932, and Cerretti et al., U.S. Pat. No. 6,521,424); anti-TWEAK agents (which include, but are not limited to, antibodies and antigen binding regions); soluble TWEAK receptor antagonists (Wiley, U.S. Pat. No. 6,727,225); an ADAM distintegrin domain to antagonize the binding of integrin to its ligands (Fanslow et al., U.S. Publication No. 2002/0042368); anti-eph receptor and anti-ephrin antibodies; antigen binding regions, or antagonists (U.S. Pat. Nos. 5,981,245; 5,728,813; 5,969,110; 6,596,852; 6,232,447; 6,057,124); anti-VEGF agents (e.g., antibodies or antigen binding regions that specifically bind VEGF, or soluble VEGF receptors or a ligand binding regions thereof) such as Avastin.RTM. or VEGF-TRAP.TM., and anti-VEGF receptor agents (e.g., antibodies or antigen binding regions that specifically bind thereto), EGFR inhibitory agents (e.g., antibodies or antigen binding regions that specifically bind thereto) such as panitumumab, IRESSA.TM. (gefitinib), TARCEVA.TM. (erlotinib), anti-Ang-1 and anti-Ang-2 agents (e.g., antibodies or antigen binding regions specifically binding thereto or to their receptors, e.g., Tie-2/TEK), and anti-Tie-2 kinase inhibitory agents (e.g., antibodies or antigen binding regions that specifically bind and inhibit the activity of growth factors, such as antagonists of hepatocyte growth factor (HGF, also known as Scatter Factor), and antibodies or antigen binding regions that specifically bind its receptor "c-met" (e.g., rilotumumab and AMG 337, Amgen); anti-PDGF-BB antagonists; antibodies and antigen binding regions to PDGF-BB ligands; and PDGFR kinase inhibitors.
[0099] Other anti-angiogenic agents that can be used in combination with a proTGF.beta.1-GARP complex-selective antibody or fragment thereof include agents such as MMP-2 (matrix-metalloproteinase 2) inhibitors, MMP-9 (matrix-metalloproteinase 9) inhibitors, and COX-II (cyclooxygenase II) inhibitors. Examples of useful COX-II inhibitors include CELEBREX.TM. (celecoxib), valdecoxib, and rofecoxib.
[0100] A proTGF.beta.1-GARP complex-selective antibody or fragment thereof as provided herein can also be used in combination with a growth factor inhibitor. Examples of such agents, include, but are not limited to, agents that can inhibit EGF-R (epidermal growth factor receptor) responses, such as EGF-R antibodies (e.g., panitumumab (VECTIBIX.RTM.)), EGF antibodies, and molecules that are EGF-R inhibitors; VEGF (vascular endothelial growth factor) inhibitors, such as VEGF receptors and molecules that can inhibit VEGF; and erbB2 receptor inhibitors, such as organic molecules or antibodies that bind to the erbB2 receptor, for example, HERCEPTIN.RTM. (Genentech, Inc.). EGF-R inhibitors are described in, for example in U.S. Pat. No. 5,747,498, WO 98/14451, WO 95/19970, and WO 98/02434.
[0101] In some treatment applications, particularly when the cancer has metastasized to the bone such that the bone is negatively impacted, it can be useful to administer a proTGF.beta.1-GARP complex-selective antibody or fragment thereof with a therapeutic agent that inhibits further bone loss or aids in restoring bone that has been lost. Accordingly, the proTGF.beta.1-GARP complex-selective antibody or fragment thereof can be administered with a therapeutically effective amount of a bone growth promoting (anabolic) agent or a bone anti-resorptive agent including but not limited to: bone morphogenic factors designated BMP-1 to BMP-12; transforming growth factor-.beta. and TGF-.beta. family members; fibroblast growth factors FGF-1 to FGF-10; interleukin-1 inhibitors (including IL-1ra, antibodies to IL-1 and antibodies to IL-1 receptors); TNF.alpha. inhibitors (including etanercept, adalibumab and infliximab); RANK ligand inhibitors (including soluble RANK, osteoprotegerin and antagonistic antibodies that specifically bind RANK or RANK ligand, such as denosumab (XGEVA.RTM.)), Dkk-1 inhibitors (e.g., anti-Dkk-1 antibodies), parathyroid hormone, E series prostaglandins, bisphosphonates and bone-enhancing minerals such as fluoride and calcium. Anabolic agents that can be used in combination with the proTGF.beta.1-GARP complex-selective antibodies and functional fragments thereof include parathyroid hormone and insulin-like growth factor (IGF), wherein the latter agent is preferably complexed with an IGF binding protein. An IL-1 receptor antagonist suitable for such combination treatment is described in WO89/11540 and a suitable soluble TNF receptor-1 is described in WO98/01555. Exemplary RANK ligand antagonists are disclosed, for example, in WO 03/086289, WO 03/002713, U.S. Pat. Nos. 6,740,511 and 6,479,635.
[0102] In one embodiment, a method for treating a cancer includes administration of a therapeutically effective amount of a proTGF.beta.1-GARP complex-selective antibody as described herein, along with radiotherapy to a subject in need thereof. Radiotherapy may comprise radiation or associated administration of radiopharmaceuticals to a patient. The source of radiation may be either external or internal to the patient being treated (radiation treatment may, for example, be in the form of external beam radiation therapy (EBRT) or brachytherapy (BT)). Radioactive elements that may be used in practicing such methods include, e.g., radium, cesium-137, iridium-192, americium-241, gold-198, cobalt-57, copper-67, technetium-99, iodide-123, iodide-131, and indium-111.
Methods of Detecting proTGF.beta.1-GARP Complex
[0103] Provided herein are methods for detecting proTGF.beta.1-GARP complex in a biological sample by contacting the sample with an antibody, or antigen-binding fragment thereof, described herein. As described herein, the sample may be derived from urine, blood, serum, plasma, saliva, ascites, circulating cells, circulating tumor cells, cells that are not tissue associated (i.e., free cells), tissues (e.g., surgically resected tumor tissue, biopsies, including fine needle aspiration), histological preparations, and the like. In some embodiments the described methods include detecting proTGF.beta.1-GARP complex in a biological sample by contacting the sample with any of the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments thereof described herein.
[0104] In some embodiments the sample may be contacted with more than one of the proTGF.beta.1-GARP complex-selective antibodies or antigen-binding fragments described herein. For example, a sample may be contacted with a first proTGF.beta.1-GARP complex-selective antibody, or antigen-binding fragment thereof, and then contacted with a second proTGF.beta.1-GARP complex-selective antibody, or antigen-binding fragment thereof, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are not the same antibody or antigen-binding fragment. In some embodiments, the first antibody, or antigen-binding fragment thereof, may be affixed to a surface, such as a multiwell plate, chip, or similar substrate prior to contacting the sample. In other embodiments the first antibody, or antigen-binding fragment thereof, may not be affixed, or attached, to anything at all prior to contacting the sample.
[0105] The described proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments may be detectably labeled. In some embodiments labeled antibodies and antigen-binding fragments may facilitate the detection of proTGF.beta.1-GARP complex via the methods described herein. Many such labels are readily known to those skilled in the art. For example, suitable labels include, but should not be considered limited to, radiolabels, fluorescent labels, epitope tags, biotin, chromophore labels, ECL labels, or enzymes. More specifically, the described labels include ruthenium, .sup.111In-DOTA, .sup.111In-diethylenetriaminepentaacetic acid (DTPA), horseradish peroxidase, alkaline phosphatase and beta-galactosidase, poly-histidine (HIS tag), acridine dyes, cyanine dyes, fluorone dyes, oxazin dyes, phenanthridine dyes, rhodamine dyes, Alexafluor.RTM. dyes, and the like.
[0106] The described proTGF.beta.1-GARP complex-selective antibodies and antigen-binding fragments may be used in a variety of assays to detect proTGF.beta.1-GARP complex in a biological sample. Some suitable assays include, but should not be considered limited to, western blot analysis, radioimmunoassay, surface plasmon resonance, immunofluorimetry, immunoprecipitation, equilibrium dialysis, immunodiffusion, electrochemiluminescence (ECL) immunoassay, immunohistochemistry, fluorescence-activated cell sorting (FACS) or ELISA assay.
Kits for Detecting proTGF.beta.1-GARP Complex
[0107] Provided herein are kits for detecting proTGF.beta.1-GARP complex in a biological sample. These kits include one or more of the proTGF.beta.1-GARP complex-selective antibodies described herein, or an antigen-binding fragment thereof, and instructions for use of the kit.
[0108] The provided proTGF.beta.1-GARP complex-selective antibody, or antigen-binding fragment, may be in solution; lyophilized; affixed to a substrate, carrier, or plate; or detectably labeled.
[0109] The described kits may also include additional components useful for performing the methods described herein. By way of example, the kits may comprise means for obtaining a sample from a subject, a control or reference sample, e.g., a sample from a subject having slowly progressing cancer and/or a subject not having cancer, one or more sample compartments, and/or instructional material which describes performance of a method of the invention and tissue specific controls or standards.
[0110] The means for determining the level of proTGF.beta.1-GARP complex can further include, for example, buffers or other reagents for use in an assay for determining the level of proTGF.beta.1-GARP complex. The instructions can be, for example, printed instructions for performing the assay and/or instructions for evaluating the level of expression of proTGF.beta.1-GARP complex.
[0111] The described kits may also include means for isolating a sample from a subject. These means can comprise one or more items of equipment or reagents that can be used to obtain a fluid or tissue from a subject. The means for obtaining a sample from a subject may also comprise means for isolating blood components, such as serum, from a blood sample. Preferably, the kit is designed for use with a human subject.
EXAMPLES
[0112] The following examples are provided to supplement the prior disclosure and to provide a better understanding of the subject matter described herein. These examples should not be considered to limit the described subject matter. It is understood that the examples and embodiments described herein are for illustrative purposes only and that various modifications or changes in light thereof will be apparent to persons skilled in the art and are to be included within, and can be made without departing from, the true scope of the invention.
Example 1
Discovery of proTGF.beta.1-GARP Complex-Selective Antibodies Using Phage Display Technology
[0113] Antibody development campaigns were undertaken to develop proTGF.beta.1-GARP complex-selective antibodies. The ChemPartner proprietary fully human naive phage display library (Chempartner, Shanghai, China) was used as a source of human antibody fragments. The library was first negatively panned against a combined mixture of biotinylated sGARP (SEQ ID NO:1) and LTBP1-proTGF.beta.1 (SEQ ID NOs 2 and 3) to remove scFv fragments that bind the undesired LTBP1-proTGFb1 complex or the uncomplexed sGARP protein. The output of the deselected library was then panned against sGARP-proTGF.beta.1 for several rounds. Twenty-five scFvs that bound specifically to sGARP-proTGF.beta.1 were selected based on unique HCDR3 sequences, desired complex selectivity, and sequence liabilities. The unique heavy chain V-regions were cloned into human IgG4 expression vectors, the unique light chains were cloned into human kappa expression vectors, and the resultant proTGF.beta.1-GARP complex-selective antibody candidates were tested again for binding activity in an ELISA. The top binders from this assay were selected for further characterization.
Example 2
Inhibition Of Human Treg Function by 4B1C1 AND 4B16B9 In Vitro
[0114] The proTGF.beta.1-GARP complex-selective antibody candidates produced in the previous example were tested for inhibition of human Treg function in an in vitro suppression assay. Activated CD4.sup.|CD25Hi suppressor cells were used as a source of Tregs and CFSE-labeled CD4+CD25- effector T cells (Teff cells) were used as targets for suppression of proliferation. Cells were incubated at a 1 Treg/1 Teff ratio with plate-bound anti-CD3, soluble anti-CD28 and in the presence or absence of proTGF.beta.1-GARP complex-selective antibody candidates. The Tregs inhibited the proliferation of Teff cells by .about.50% as compared to T effectors only. Proliferation levels of Teff (incubated with Tregs) were restored in the presence of 4B1C1, and partially restored by 4B16B9, when compared to the Treg/Teff control or hIgG4 groups (FIG. 1). These results confirm the activity of TGF-.beta.1 in the immunosuppression by human Tregs and indicate that 4B1C1 and 4B16B9 can partially block this activity in vitro, similar to the positive control neutralizing TGF.beta. antibody 1D11 (R&D Systems catalog number MAB-1835).
TABLE-US-00002 TABLE 2 CDR sequences of the two proTGF.beta.1-GARP complex-selective antibody candidates that showed binding against proTGF.beta.1-GARP complex and inhibition of human Treg function in vitro (SEQ ID NO:) ID HC-CDR1 HC-CDR2 HC-CDR3 LC-CDR1 LC-CDR2 LC-CDR3 4B1C1 DYTMH (4) LISWDGGSTYYADSVKG DADDSTFDI RASQSVSRNLA (7) WASTRES QQYYSVPYT (5) (6) (8) (9) 4B16B9 SYAIS (10) GIIPMFGTTNYAQKFQG DREWEPAYG IGTSSDVGGYNYVS DVSNRPS SAYTVSSTW (11) MDV (12) (13) (14) V (15)
VH and VL of the Two proTGF.beta.1-GARP Complex-Selective Antibody Candidates are Shown Below in Table 3.
TABLE-US-00003 TABLE 3 Heavy chain and light chain sequences of the two proTGF.beta.1-GARP complex- selective antibody candidates that showed binding against proTGF.beta.1-GARP complex and inhibition of human Treg function in vitro. Variable regions are underlined. mAb Heavy Chain Amino Acid SEQ ID Light Chain Amino Acid SEQ ID ID Sequence NO: Sequence NO: 4B1C1 EVQLVQSGGVVVQPG 16 ETTLTQSPATLSVSPGE 17 GSLRLSCAASGFTFDD RVTLSCRASQSVSRNL YTMHWVRQAPGKGLE AWYQQKPGQPPKLLIY WVSLISWDGGSTYYA WASTRESGVPDRFSGS DSVKGRFTISRDNSKN GSGTDFTLTISSLQAED SLYLQMNSLRTEDTAL VAVYYCQQYYSVPYT YYCAKDADDSTFDIW FGQGTKLEIKRTVAAP GQGTMVTVSSASTKGP SVFIFPPSDEQLKSGTA SVFPLAPCSRSTSESTA SVVCLLNNFYPREAKV ALGCLVKDYFPEPVTV QWKVDNALQSGNSQE SWNSGALTSGVHTFPA SVTEQDSKDSTYSLSST VLQSSGLYSLSSVVTV LTLSKADYEKHKVYA PSSSLGTKTYTCNVDH CEVTHQGLSSPVTKSF KPSNTKVDKRVESKY NRGEC GPPCPPCPAPEFLGGPS VFLFPPKPKDTLMISRT PEVTCVVVDVSQEDPE VQFNWYVDGVEVHN AKTKPREEQFNSTYRV VSVLTVLHQDWLNGK EYKCKVSNKGLPSSIE KTISKAKGQPREPQVY TLPPSQEEMTKNQVSL TCLVKGFYPSDIAVEW ESNGQPENNYKTTPPV LDSDGSFFLYSRLTVD KSRWQEGNVFSCSVM HEALHNHYTQKSLSLS LG 4B16B9 QMQLVQSGAEVKKPG 18 QSALTQPASVSGSPGQ 19 SSVKVSCKASGGTFSS SITISCIGTSSDVGGYN YAISWVRQAPGQGLE YVSWYQQHPGKAPKL WMGGIIPMFGTTNYA MIYDVSNRPSGVSNRF QKFQGRVTIIADESTST SGSKSGNTASLTISGLQ AYMELRSLRSDDTAV AEDEAMYYCSAYTVS YYCARDREWEPAYGM STWVFGGGTKVTVLGG DVWGQGTTVTVSSASAS QPKAAPSVTLFPPSSEE TKGPSVFPLAPCSRSTS LQANKATLVCLISDFY ESTAALGCLVKDYFPE PGAVTVAWKADSSPV PVTVSWNSGALTSGV KAGVETTTPSKQSNNK HTFPAVLQSSGLYSLSS YAASSYLSLTPEQWKS VVTVPSSSLGTKTYTC HRSYSCQVTHEGSTVE NVDHKPSNTKVDKRV KTVAPTECS ESKYGPPCPPCPAPEFL GGPSVFLFPPKPKDTL MISRTPEVTCVVVDVS QEDPEVQFNWYVDGV EVHNAKTKPREEQFNS TYRVVSVLTVLHQDW LNGKEYKCKVSNKGL PSSIEKTISKAKGQPRE PQVYTLPPSQEEMTKN QVSLTCLVKGFYPSDI AVEWESNGQPENNYK TTPPVLDSDGSFFLYSR LTVDKSRWQEGNVFS CSVMHEALHNHYTQK SLSLSLG
Example 3
TGF.beta.1 Bioassay
[0115] The ability of the proTGF.beta.1-GARP complex-selective antibody candidates to regulate the levels of active TGF.beta.1 was measured using TMLC reporter cells with an integrated TGF.beta./Smad3-responsive luciferase expression unit. Briefly, HEK293 or Sw480 cells were transiently transfected with either human proTGF.beta.1 and LTBP1 or human proTGF.beta.1 and GARP expression plasmids. The cells were allowed to recover from the transfection and to express proTGF.beta.1 in complex with either LTBP1 or GARP for 24 hours at 37.degree. C., at which point the assay could be performed. To set up the assay, the transient transfectants were co-cultured with SW480(36 cells, which stably express the TGF.beta.1-activating integrin .degree.V.beta.6. To confirm that the assay worked as intended, media samples with known concentrations of TGF-.beta.1 growth factor were added to TMLC reporter cell cultures to generate a standard curve.
[0116] ProTGF.beta.1-GARP complex-selective antibody candidates (10 .mu.g/mL) were combined with transfected cells and added to TMLC reporter cell cultures. The plates were then incubated at 37.degree. C. for 16 hours. Successful TGF.beta.1 signaling was expected to activate the SMAD2/3 pathway, followed by luciferase expression, which could be detected by adding Bright-Glo, as indicated by the manufacturer (Promega), and measuring the resultant luminescence in a Biotek Synergy H1 plate reader (Biotek).
[0117] 4B1C1 and 4B16B9 antibodies induced significantly decreased luciferase expression compared to treatment with the control group, indicating that these antibodies regulate the levels of active TGF.beta.1 growth factor and reduce TGF.beta.1-mediated signaling in the cells. This effect is similar to the impact of the positive control neutralizing TGF.beta. antibody 1D11.
Example 4
Affinity Measurements by Biolayer Interferometry
[0118] The binding affinities of the proTGF.beta.1-GARP complex-selective antibody candidates to proTGF.beta.1-GARP complexes were measured by biolayer interferometry on an OctetRed 384 (Fortebio, Menlo Park, Calif.). Strepavidin biosensors (Fortebio, Cat. No. 18-5020) were loaded with biotinylated sGARP-proTGF.beta.1 complex at 20 .mu.g/ml in sodium acetate buffer, pH 5, washed in the same buffer and transferred to wells containing 10 .mu.g/mL proTGF.beta.1-GARP complex-selective antibody candidates in the same buffer. The dissociation constant was obtained by non-linear fitting of the responses to a steady state algorithm using Octet software (Table 4). Similar affinities were obtained by kinetic fitting.
TABLE-US-00004 TABLE 4 Octet affinity results for proTGF.beta.1-GARP complex-selective antibody candidates binding to human proTGF.beta.1-GARP complex. mAb proTGF.beta.1-GARP complex K.sub.D (nM) 4B1C1 human 0.114 +/- 0.004 4B16B9 human 0.880 +/- 0.036
[0119] To ascertain binding specificity, 4B1C1 and 4B16B9 were screened as above for binding to TGF.beta.1, TGF.beta.2, TGF.beta.3, proTGF.beta.1-LTBP1, proTGF.beta.1-LTBP3 and proTGF.beta.1-LRRC33. As described above, antibodies were tested at 10 .mu.g/ml and antigens at 20 .mu.g/ml and tested under the following conditions. These studies demonstrated no discernible binding of antibodies to any antigen other than the proTGF.beta.1-GARP complex and shown in FIG. 4.
TABLE-US-00005 TABLE 5 Assay conditions for determination of antibody binding specificity Assay Step Number Step Type Assay Time in Seconds 1 Baseline 60 2 Antigen Loading 180 3 Baseline 60 4 Association 300 5 Dissociation 600
Example 5
Dose Response Assay
[0120] The concentration dependence of proTGF.beta.1-GARP complex-selective antibody candidate regulation of levels of active TGF.beta.1 was measured using TMLC reporter cells with an integrated TGF.beta./Smad3-responsive luciferase expression unit as described in EXAMPLE 3 with the only difference being that proTGF.beta.1-GARP complex-selective antibody candidates were added to experimental wells at various concentrations.
[0121] 4B1C1 and 4B16B9 demonstrated they could inhibit TGF.beta.1 activation in a dose-dependent manner with IC50s of 0.178 nM and 1.9 nM, respectively (FIG. 3).
Example 6
Antibody Characterization
[0122] proTGF.beta.1-GARP complex-selective antibody candidates were measured by biolayer interferometry on an OctetRed 384 (Fortebio, Menlo Park, Calif.) for selectivity for proTGF.beta.1-GARP complex over other protein complexes (FIG. 4). 4B1C1 and 4B16B9 exhibited a dissociation constant (Kd) of less than 1 nM for proTGF.beta.1-GARP complex, while exhibiting no detectable binding to proTGF.beta.1-LTBP1 or proTGF.beta.1-LTBP3 complexes. 4B1C1 and 4B16B9 also did not exhibit binding to TGF.beta.1, TGF.beta.2, or TGF.beta.3 growth factors.
[0123] Antibody binding to proTGF.beta.1-LRRC33 complexes was also tested. proTGF.beta.1-LRRC33 complex was expressed and purified by size-exclusion chromatography (SEC) and the formation of non-aggregated complexes was confirmed by analytical SEC. Binding of 4B1C1 and 4B16B9 to purified proTGF.beta.1-LRRC33 complex was then tested by OctetRed 384 (Fortebio, Menlo Park, Calif.) analysis as described in Example 4. For that, 4B1C1 or 4B16B9 was captured on anti-human Fc tips, and binding of either proTGF.beta.1-GARP or proTGF.beta.1-LRRC33 was detected by Octet. In contrast to proTGF.beta.1-GARP, no binding of 4B1C1 or 4B16B9 to proTGF.beta.1-LRRC33 complex was detected.
BRIEF DESCRIPTION OF THE SEQUENCE LISTING
TABLE-US-00006 [0124] SEQ ID NO: Type Species Description Sequence 1 PRT human sGARP HQDKVPCKMVDKKVSCQVLGLLQV PSVLPPDTETLDLSGNQLRSILASPLG FYTALRHLDLSTNEISFLQ PGAFQALTHLEHLSLAHNRLAMATA LSAGGLGPLPRVTSLDLSGNSLYSGL LERLLGEAPSLHTLSLAEN SLTRLTRHTFRDMPALEQLDLHSNVL MDIEDGAFEGLPRLTHLNLSRNSLTCI SDFSLQQLRVLDLSCNS IEAFQTASQPQAEFQLTWLDLRENKL LHFPDLAALPRLIYLNLSNNLIRLPTG PPQDSKGIHAPSEGWSA LPLSAPSGNASGRPLSQLLNLDLSYN EIELIPDSFLEHLTSLCFLNLSRNCLRT FEARRLGSLPCLMLLD LSHNALETLELGARALGSLRTLLLQG NALRDLPPYTFANLASLQRLNLQGN RVSPCGGPDEPGPSGCVAF SGITSLRSLSLVDNEIELLRAGAFLHT PLTELDLSSNPGLEVATGALGGLEAS LEVLALQGNGLMVLQVD LPCFICLKRLNLAENRLSHLPAWTQA VSLEVLDLRNNSFSLLPGSAMGGLET SLRRLYLQGNPLSCCGNG WLAAQLHQGRVDVDATQDLICRFSS QEEVSLSHVRPEDCEKGGLKNINHH HHHH 2 PRT human proTGF.beta.1 LSTCKTIDMELVKRKRIEAIRGQILSK LRLASPPSQGEVPPGPLPEAVLALYN STRDRVAGESAEPEPEP EADYYAKEVTRVLMVETHNEIYDKF KQSTHSIYMFFNTSELREAVPEPVLLS RAELRLLRLKLKVEQHVE LYQKYSNNSWRYLSNRLLAPSDSPE WLSFDVTGVVRQWLSRGGEIEGFRL SAHCSCDSRDNTLQVDINGF TTGRRGDLATIHGMNRPFLLLMATPL ERAQHLQSSRHRRALDTNYCFSSTEK NCCVRQLYIDFRKDLGWK WIHEPKGYHANFCLGPCPYIWSLDTQ YSKVLALYNQHNPGASAAPCCVPQA LEPLPIVYYVGRKPKVEQL SNMIVRSCKCS 3 PRT human LTBP1 EINECTVNPDICGAGHCINLPVRYTCI fragment CYEGYRFSEQQRKCVDIDECTQVQH LCSQGRCENTEGSFLCIC PAGFMASEEGTNCIDVDECLRPDVC GEGHCVNTVGAFRCEYCDSGYRMT QRGRCEDIDECLNPSTCPDEQ CVNSPGSYQCVPCTEGFRGWNGQCL DVDECLEPNVCANGDCSNLEGSYMC SCHKGYTRTPDHKHCRDIDE CQQGNLCVNGQCKNTEGSFRCTCGQ GYQLSAAKDQCEDIDECQHRHLCAH GQCRNTEGSFQCVCDQGYRA SGLGDHCEDINECLEDKSVCQRGDCI NTAGSYDCTCPDGFQLDDNKTCQDI NECEHPGLCGPQGECLNTE GSFHCVCQQGFSISADGRTCEDIDEC VNNTVCDSHGFCDNTAGSFRCLCYQ GFQAPQDGQGCVDVNECEL LSGVCGEAFCENVEGSFLCVCADEN QEYSPMTGQCRSRTSTDLDVDVDQP KEEKKECYYNLNDASLCDNV LAPNVTKQECCCTSGVGWGDNCEIF PCPVLGTAEFTEMCPKGKGFVPAGES SSEAGGENYKDADECLLFG QEICKNGFCLNTRPGYECYCKQGTY YDPVKLQCFDMDECQDPSSCIDGQC VNTEGSYNCFCTHPMVLDAS EKRCIHHHHH 4 PRT human 4B1C1- DYTMH HCDR1 5 PRT human 4B1C1- LISWDGGSTYYADSVKG HCDR2 6 PRT human 4B1C1- DADDSTFDI HCDR3 7 PRT human 4B1C1- RASQSVSRNLA LCDR1 8 PRT human 4B1C1- WASTRES LCDR2 9 PRT human 4B1C1- QQYYSVPYT LCDR3 10 PRT human 4B16B9- SYAIS HCDR1 11 PRT human 4B16B9- GIIPMFGTTNYAQKFQG HCDR2 12 PRT human 4B16B9- DREWEPAYGMDV HCDR3 13 PRT human 4B16B9- IGTSSDVGGYNYVS LCDR1 14 PRT human 4B16B9- DVSNRPS LCDR2 15 PRT human 4B16B9- SAYTVSSTWV LCDR3 16 PRT human 4B1C1- EVQLVQSGGVVVQPGGSLRLSCAAS Heavy GFTFDDYTMHWVRQAPGKGLEWVS Chain LISWDGGSTYYADSVKGRFTISRDNS KNSLYLQMNSLRTEDTALYYCAKDA DDSTFDIWGQGTMVTVSSASTKGPS VFPLAPCSRSTSESTAALGCLVKDYF PEPVTVSWNSGALTSGVHTFPAVLQS SGLYSLSSVVTVPSSSLGTKTYTCNV DHKPSNTKVDKRVESKYGPPCPPCPA PEFLGGPSVFLFPPKPKDTLMISRTPE VTCVVVDVSQEDPEVQFNWYVDGV EVHNAKTKPREEQFNSTYRVVSVLT VLHQDWLNGKEYKCKVSNKGLPSSI EKTISKAKGQPREPQVYTLPPSQEEM TKNQVSLTCLVKGFYPSDIAVEWESN GQPENNYKTTPPVLDSDGSFFLYSRL TVDKSRWQEGNVFSCSVMHEALHN HYTQKSLSLSLG 17 PRT human 4B1C1- ETTLTQSPATLSVSPGERVTLSCRASQ Light SVSRNLAWYQQKPGQPPKLLIYVVAS Chain TRESGVPDRFSGSGSGTDFTLTISSLQ AEDVAVYYCQQYYSVPYTFGQGTKL EIKRTVAAPSVFIFPPSDEQLKSGTAS VVCLLNNFYPREAKVQWKVDNALQ SGNSQESVTEQDSKDSTYSLSSTLTLS KADYEKHKVYACEVTHQGLSSPVTK SFNRGEC 18 PRT human 4B16B9- QMQLVQSGAEVKKPGSSVKVSCKAS Heavy GGTFSSYAISWVRQAPGQGLEWMGG Chain IIPMFGTTNYAQKFQGRVTIIADESTS TAYMELRSLRSDDTAVYYCARDRE WEPAYGMDVWGQGTTVTVSSASTK GPSVFPLAPCSRSTSESTAALGCLVK DYFPEPVTVSWNSGALTSGVHTFPA VLQSSGLYSLSSVVTVPSSSLGTKTY TCNVDHKPSNTKVDKRVESKYGPPC PPCPAPEFLGGPSVFLFPPKPKDTLMI SRTPEVTCVVVDVSQEDPEVQFNWY VDGVEVHNAKTKPREEQFNSTYRVV SVLTVLHQDWLNGKEYKCKVSNKG LPSSIEKTISKAKGQPREPQVYTLPPS QEEMTKNQVSLTCLVKGFYPSDIAVE WESNGQPENNYKTTPPVLDSDGSFFL YSRLTVDKSRWQEGNVFSCSVMHEA LHNHYTQKSLSLSLG 19 PRT human 4b16B9- QSALTQPASVSGSPGQSITISCIGTSSD Light VGGYNYVSWYQQHPGKAPKLMIYD Chain VSNRPSGVSNRFSGSKSGNTASLTISG LQAEDEAMYYCSAYTVSSTWVFGG GTKVTVLGQPKAAPSVTLFPPSSEEL QANKATLVCLISDFYPGAVTVAWKA DSSPVKAGVETTTPSKQSNNKYAASS YLSLTPEQWKSHRSYSCQVTHEGST VEKTVAPTECS
Sequence CWU 1
1
191614PRTHomo sapiens 1His Gln Asp Lys Val Pro Cys Lys Met Val Asp
Lys Lys Val Ser Cys1 5 10 15Gln Val Leu Gly Leu Leu Gln Val Pro Ser
Val Leu Pro Pro Asp Thr 20 25 30Glu Thr Leu Asp Leu Ser Gly Asn Gln
Leu Arg Ser Ile Leu Ala Ser 35 40 45Pro Leu Gly Phe Tyr Thr Ala Leu
Arg His Leu Asp Leu Ser Thr Asn 50 55 60Glu Ile Ser Phe Leu Gln Pro
Gly Ala Phe Gln Ala Leu Thr His Leu65 70 75 80Glu His Leu Ser Leu
Ala His Asn Arg Leu Ala Met Ala Thr Ala Leu 85 90 95Ser Ala Gly Gly
Leu Gly Pro Leu Pro Arg Val Thr Ser Leu Asp Leu 100 105 110Ser Gly
Asn Ser Leu Tyr Ser Gly Leu Leu Glu Arg Leu Leu Gly Glu 115 120
125Ala Pro Ser Leu His Thr Leu Ser Leu Ala Glu Asn Ser Leu Thr Arg
130 135 140Leu Thr Arg His Thr Phe Arg Asp Met Pro Ala Leu Glu Gln
Leu Asp145 150 155 160Leu His Ser Asn Val Leu Met Asp Ile Glu Asp
Gly Ala Phe Glu Gly 165 170 175Leu Pro Arg Leu Thr His Leu Asn Leu
Ser Arg Asn Ser Leu Thr Cys 180 185 190Ile Ser Asp Phe Ser Leu Gln
Gln Leu Arg Val Leu Asp Leu Ser Cys 195 200 205Asn Ser Ile Glu Ala
Phe Gln Thr Ala Ser Gln Pro Gln Ala Glu Phe 210 215 220Gln Leu Thr
Trp Leu Asp Leu Arg Glu Asn Lys Leu Leu His Phe Pro225 230 235
240Asp Leu Ala Ala Leu Pro Arg Leu Ile Tyr Leu Asn Leu Ser Asn Asn
245 250 255Leu Ile Arg Leu Pro Thr Gly Pro Pro Gln Asp Ser Lys Gly
Ile His 260 265 270Ala Pro Ser Glu Gly Trp Ser Ala Leu Pro Leu Ser
Ala Pro Ser Gly 275 280 285Asn Ala Ser Gly Arg Pro Leu Ser Gln Leu
Leu Asn Leu Asp Leu Ser 290 295 300Tyr Asn Glu Ile Glu Leu Ile Pro
Asp Ser Phe Leu Glu His Leu Thr305 310 315 320Ser Leu Cys Phe Leu
Asn Leu Ser Arg Asn Cys Leu Arg Thr Phe Glu 325 330 335Ala Arg Arg
Leu Gly Ser Leu Pro Cys Leu Met Leu Leu Asp Leu Ser 340 345 350His
Asn Ala Leu Glu Thr Leu Glu Leu Gly Ala Arg Ala Leu Gly Ser 355 360
365Leu Arg Thr Leu Leu Leu Gln Gly Asn Ala Leu Arg Asp Leu Pro Pro
370 375 380Tyr Thr Phe Ala Asn Leu Ala Ser Leu Gln Arg Leu Asn Leu
Gln Gly385 390 395 400Asn Arg Val Ser Pro Cys Gly Gly Pro Asp Glu
Pro Gly Pro Ser Gly 405 410 415Cys Val Ala Phe Ser Gly Ile Thr Ser
Leu Arg Ser Leu Ser Leu Val 420 425 430Asp Asn Glu Ile Glu Leu Leu
Arg Ala Gly Ala Phe Leu His Thr Pro 435 440 445Leu Thr Glu Leu Asp
Leu Ser Ser Asn Pro Gly Leu Glu Val Ala Thr 450 455 460Gly Ala Leu
Gly Gly Leu Glu Ala Ser Leu Glu Val Leu Ala Leu Gln465 470 475
480Gly Asn Gly Leu Met Val Leu Gln Val Asp Leu Pro Cys Phe Ile Cys
485 490 495Leu Lys Arg Leu Asn Leu Ala Glu Asn Arg Leu Ser His Leu
Pro Ala 500 505 510Trp Thr Gln Ala Val Ser Leu Glu Val Leu Asp Leu
Arg Asn Asn Ser 515 520 525Phe Ser Leu Leu Pro Gly Ser Ala Met Gly
Gly Leu Glu Thr Ser Leu 530 535 540Arg Arg Leu Tyr Leu Gln Gly Asn
Pro Leu Ser Cys Cys Gly Asn Gly545 550 555 560Trp Leu Ala Ala Gln
Leu His Gln Gly Arg Val Asp Val Asp Ala Thr 565 570 575Gln Asp Leu
Ile Cys Arg Phe Ser Ser Gln Glu Glu Val Ser Leu Ser 580 585 590His
Val Arg Pro Glu Asp Cys Glu Lys Gly Gly Leu Lys Asn Ile Asn 595 600
605His His His His His His 6102361PRTHomo sapiens 2Leu Ser Thr Cys
Lys Thr Ile Asp Met Glu Leu Val Lys Arg Lys Arg1 5 10 15Ile Glu Ala
Ile Arg Gly Gln Ile Leu Ser Lys Leu Arg Leu Ala Ser 20 25 30Pro Pro
Ser Gln Gly Glu Val Pro Pro Gly Pro Leu Pro Glu Ala Val 35 40 45Leu
Ala Leu Tyr Asn Ser Thr Arg Asp Arg Val Ala Gly Glu Ser Ala 50 55
60Glu Pro Glu Pro Glu Pro Glu Ala Asp Tyr Tyr Ala Lys Glu Val Thr65
70 75 80Arg Val Leu Met Val Glu Thr His Asn Glu Ile Tyr Asp Lys Phe
Lys 85 90 95Gln Ser Thr His Ser Ile Tyr Met Phe Phe Asn Thr Ser Glu
Leu Arg 100 105 110Glu Ala Val Pro Glu Pro Val Leu Leu Ser Arg Ala
Glu Leu Arg Leu 115 120 125Leu Arg Leu Lys Leu Lys Val Glu Gln His
Val Glu Leu Tyr Gln Lys 130 135 140Tyr Ser Asn Asn Ser Trp Arg Tyr
Leu Ser Asn Arg Leu Leu Ala Pro145 150 155 160Ser Asp Ser Pro Glu
Trp Leu Ser Phe Asp Val Thr Gly Val Val Arg 165 170 175Gln Trp Leu
Ser Arg Gly Gly Glu Ile Glu Gly Phe Arg Leu Ser Ala 180 185 190His
Cys Ser Cys Asp Ser Arg Asp Asn Thr Leu Gln Val Asp Ile Asn 195 200
205Gly Phe Thr Thr Gly Arg Arg Gly Asp Leu Ala Thr Ile His Gly Met
210 215 220Asn Arg Pro Phe Leu Leu Leu Met Ala Thr Pro Leu Glu Arg
Ala Gln225 230 235 240His Leu Gln Ser Ser Arg His Arg Arg Ala Leu
Asp Thr Asn Tyr Cys 245 250 255Phe Ser Ser Thr Glu Lys Asn Cys Cys
Val Arg Gln Leu Tyr Ile Asp 260 265 270Phe Arg Lys Asp Leu Gly Trp
Lys Trp Ile His Glu Pro Lys Gly Tyr 275 280 285His Ala Asn Phe Cys
Leu Gly Pro Cys Pro Tyr Ile Trp Ser Leu Asp 290 295 300Thr Gln Tyr
Ser Lys Val Leu Ala Leu Tyr Asn Gln His Asn Pro Gly305 310 315
320Ala Ser Ala Ala Pro Cys Cys Val Pro Gln Ala Leu Glu Pro Leu Pro
325 330 335Ile Val Tyr Tyr Val Gly Arg Lys Pro Lys Val Glu Gln Leu
Ser Asn 340 345 350Met Ile Val Arg Ser Cys Lys Cys Ser 355
3603640PRTHomo sapiens 3Glu Ile Asn Glu Cys Thr Val Asn Pro Asp Ile
Cys Gly Ala Gly His1 5 10 15Cys Ile Asn Leu Pro Val Arg Tyr Thr Cys
Ile Cys Tyr Glu Gly Tyr 20 25 30Arg Phe Ser Glu Gln Gln Arg Lys Cys
Val Asp Ile Asp Glu Cys Thr 35 40 45Gln Val Gln His Leu Cys Ser Gln
Gly Arg Cys Glu Asn Thr Glu Gly 50 55 60Ser Phe Leu Cys Ile Cys Pro
Ala Gly Phe Met Ala Ser Glu Glu Gly65 70 75 80Thr Asn Cys Ile Asp
Val Asp Glu Cys Leu Arg Pro Asp Val Cys Gly 85 90 95Glu Gly His Cys
Val Asn Thr Val Gly Ala Phe Arg Cys Glu Tyr Cys 100 105 110Asp Ser
Gly Tyr Arg Met Thr Gln Arg Gly Arg Cys Glu Asp Ile Asp 115 120
125Glu Cys Leu Asn Pro Ser Thr Cys Pro Asp Glu Gln Cys Val Asn Ser
130 135 140Pro Gly Ser Tyr Gln Cys Val Pro Cys Thr Glu Gly Phe Arg
Gly Trp145 150 155 160Asn Gly Gln Cys Leu Asp Val Asp Glu Cys Leu
Glu Pro Asn Val Cys 165 170 175Ala Asn Gly Asp Cys Ser Asn Leu Glu
Gly Ser Tyr Met Cys Ser Cys 180 185 190His Lys Gly Tyr Thr Arg Thr
Pro Asp His Lys His Cys Arg Asp Ile 195 200 205Asp Glu Cys Gln Gln
Gly Asn Leu Cys Val Asn Gly Gln Cys Lys Asn 210 215 220Thr Glu Gly
Ser Phe Arg Cys Thr Cys Gly Gln Gly Tyr Gln Leu Ser225 230 235
240Ala Ala Lys Asp Gln Cys Glu Asp Ile Asp Glu Cys Gln His Arg His
245 250 255Leu Cys Ala His Gly Gln Cys Arg Asn Thr Glu Gly Ser Phe
Gln Cys 260 265 270Val Cys Asp Gln Gly Tyr Arg Ala Ser Gly Leu Gly
Asp His Cys Glu 275 280 285Asp Ile Asn Glu Cys Leu Glu Asp Lys Ser
Val Cys Gln Arg Gly Asp 290 295 300Cys Ile Asn Thr Ala Gly Ser Tyr
Asp Cys Thr Cys Pro Asp Gly Phe305 310 315 320Gln Leu Asp Asp Asn
Lys Thr Cys Gln Asp Ile Asn Glu Cys Glu His 325 330 335Pro Gly Leu
Cys Gly Pro Gln Gly Glu Cys Leu Asn Thr Glu Gly Ser 340 345 350Phe
His Cys Val Cys Gln Gln Gly Phe Ser Ile Ser Ala Asp Gly Arg 355 360
365Thr Cys Glu Asp Ile Asp Glu Cys Val Asn Asn Thr Val Cys Asp Ser
370 375 380His Gly Phe Cys Asp Asn Thr Ala Gly Ser Phe Arg Cys Leu
Cys Tyr385 390 395 400Gln Gly Phe Gln Ala Pro Gln Asp Gly Gln Gly
Cys Val Asp Val Asn 405 410 415Glu Cys Glu Leu Leu Ser Gly Val Cys
Gly Glu Ala Phe Cys Glu Asn 420 425 430Val Glu Gly Ser Phe Leu Cys
Val Cys Ala Asp Glu Asn Gln Glu Tyr 435 440 445Ser Pro Met Thr Gly
Gln Cys Arg Ser Arg Thr Ser Thr Asp Leu Asp 450 455 460Val Asp Val
Asp Gln Pro Lys Glu Glu Lys Lys Glu Cys Tyr Tyr Asn465 470 475
480Leu Asn Asp Ala Ser Leu Cys Asp Asn Val Leu Ala Pro Asn Val Thr
485 490 495Lys Gln Glu Cys Cys Cys Thr Ser Gly Val Gly Trp Gly Asp
Asn Cys 500 505 510Glu Ile Phe Pro Cys Pro Val Leu Gly Thr Ala Glu
Phe Thr Glu Met 515 520 525Cys Pro Lys Gly Lys Gly Phe Val Pro Ala
Gly Glu Ser Ser Ser Glu 530 535 540Ala Gly Gly Glu Asn Tyr Lys Asp
Ala Asp Glu Cys Leu Leu Phe Gly545 550 555 560Gln Glu Ile Cys Lys
Asn Gly Phe Cys Leu Asn Thr Arg Pro Gly Tyr 565 570 575Glu Cys Tyr
Cys Lys Gln Gly Thr Tyr Tyr Asp Pro Val Lys Leu Gln 580 585 590Cys
Phe Asp Met Asp Glu Cys Gln Asp Pro Ser Ser Cys Ile Asp Gly 595 600
605Gln Cys Val Asn Thr Glu Gly Ser Tyr Asn Cys Phe Cys Thr His Pro
610 615 620Met Val Leu Asp Ala Ser Glu Lys Arg Cys Ile His His His
His His625 630 635 64045PRTHomo sapiens 4Asp Tyr Thr Met His1
5517PRTHomo sapiens 5Leu Ile Ser Trp Asp Gly Gly Ser Thr Tyr Tyr
Ala Asp Ser Val Lys1 5 10 15Gly69PRTHomo sapiens 6Asp Ala Asp Asp
Ser Thr Phe Asp Ile1 5711PRTHomo sapiens 7Arg Ala Ser Gln Ser Val
Ser Arg Asn Leu Ala1 5 1087PRTHomo sapiens 8Trp Ala Ser Thr Arg Glu
Ser1 599PRTHomo sapiens 9Gln Gln Tyr Tyr Ser Val Pro Tyr Thr1
5105PRTHomo sapiens 10Ser Tyr Ala Ile Ser1 51117PRTHomo sapiens
11Gly Ile Ile Pro Met Phe Gly Thr Thr Asn Tyr Ala Gln Lys Phe Gln1
5 10 15Gly1212PRTHomo sapiens 12Asp Arg Glu Trp Glu Pro Ala Tyr Gly
Met Asp Val1 5 101314PRTHomo sapiens 13Ile Gly Thr Ser Ser Asp Val
Gly Gly Tyr Asn Tyr Val Ser1 5 10147PRTHomo sapiens 14Asp Val Ser
Asn Arg Pro Ser1 51510PRTHomo sapiens 15Ser Ala Tyr Thr Val Ser Ser
Thr Trp Val1 5 1016444PRTHomo sapiens 16Glu Val Gln Leu Val Gln Ser
Gly Gly Val Val Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys
Ala Ala Ser Gly Phe Thr Phe Asp Asp Tyr 20 25 30Thr Met His Trp Val
Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Ser Leu Ile Ser
Trp Asp Gly Gly Ser Thr Tyr Tyr Ala Asp Ser Val 50 55 60Lys Gly Arg
Phe Thr Ile Ser Arg Asp Asn Ser Lys Asn Ser Leu Tyr65 70 75 80Leu
Gln Met Asn Ser Leu Arg Thr Glu Asp Thr Ala Leu Tyr Tyr Cys 85 90
95Ala Lys Asp Ala Asp Asp Ser Thr Phe Asp Ile Trp Gly Gln Gly Thr
100 105 110Met Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser Val
Phe Pro 115 120 125Leu Ala Pro Cys Ser Arg Ser Thr Ser Glu Ser Thr
Ala Ala Leu Gly 130 135 140Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro
Val Thr Val Ser Trp Asn145 150 155 160Ser Gly Ala Leu Thr Ser Gly
Val His Thr Phe Pro Ala Val Leu Gln 165 170 175Ser Ser Gly Leu Tyr
Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser 180 185 190Ser Leu Gly
Thr Lys Thr Tyr Thr Cys Asn Val Asp His Lys Pro Ser 195 200 205Asn
Thr Lys Val Asp Lys Arg Val Glu Ser Lys Tyr Gly Pro Pro Cys 210 215
220Pro Pro Cys Pro Ala Pro Glu Phe Leu Gly Gly Pro Ser Val Phe
Leu225 230 235 240Phe Pro Pro Lys Pro Lys Asp Thr Leu Met Ile Ser
Arg Thr Pro Glu 245 250 255Val Thr Cys Val Val Val Asp Val Ser Gln
Glu Asp Pro Glu Val Gln 260 265 270Phe Asn Trp Tyr Val Asp Gly Val
Glu Val His Asn Ala Lys Thr Lys 275 280 285Pro Arg Glu Glu Gln Phe
Asn Ser Thr Tyr Arg Val Val Ser Val Leu 290 295 300Thr Val Leu His
Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys305 310 315 320Val
Ser Asn Lys Gly Leu Pro Ser Ser Ile Glu Lys Thr Ile Ser Lys 325 330
335Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser
340 345 350Gln Glu Glu Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu
Val Lys 355 360 365Gly Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu
Ser Asn Gly Gln 370 375 380Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp Gly385 390 395 400Ser Phe Phe Leu Tyr Ser Arg
Leu Thr Val Asp Lys Ser Arg Trp Gln 405 410 415Glu Gly Asn Val Phe
Ser Cys Ser Val Met His Glu Ala Leu His Asn 420 425 430His Tyr Thr
Gln Lys Ser Leu Ser Leu Ser Leu Gly 435 44017214PRTHomo sapiens
17Glu Thr Thr Leu Thr Gln Ser Pro Ala Thr Leu Ser Val Ser Pro Gly1
5 10 15Glu Arg Val Thr Leu Ser Cys Arg Ala Ser Gln Ser Val Ser Arg
Asn 20 25 30Leu Ala Trp Tyr Gln Gln Lys Pro Gly Gln Pro Pro Lys Leu
Leu Ile 35 40 45Tyr Trp Ala Ser Thr Arg Glu Ser Gly Val Pro Asp Arg
Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser
Ser Leu Gln Ala65 70 75 80Glu Asp Val Ala Val Tyr Tyr Cys Gln Gln
Tyr Tyr Ser Val Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Leu Glu
Ile Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe Ile Phe Pro
Pro Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala Ser Val Val
Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln
Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln145 150 155
160Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser
165 170 175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys
Val Tyr 180 185 190Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro
Val Thr Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys 21018447PRTHomo
sapiens 18Gln Met Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ser1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Gly Thr Phe
Ser Ser Tyr 20 25 30Ala Ile Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Met 35
40 45Gly Gly Ile Ile Pro Met Phe Gly Thr Thr Asn Tyr Ala Gln Lys
Phe 50 55 60Gln Gly Arg Val Thr Ile Ile Ala Asp Glu Ser Thr Ser Thr
Ala Tyr65 70 75 80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Glu Trp Glu Pro Ala Tyr Gly
Met Asp Val Trp Gly 100 105 110Gln Gly Thr Thr Val Thr Val Ser Ser
Ala Ser Thr Lys Gly Pro Ser 115 120 125Val Phe Pro Leu Ala Pro Cys
Ser Arg Ser Thr Ser Glu Ser Thr Ala 130 135 140Ala Leu Gly Cys Leu
Val Lys Asp Tyr Phe Pro Glu Pro Val Thr Val145 150 155 160Ser Trp
Asn Ser Gly Ala Leu Thr Ser Gly Val His Thr Phe Pro Ala 165 170
175Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val Thr Val
180 185 190Pro Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr Cys Asn Val
Asp His 195 200 205Lys Pro Ser Asn Thr Lys Val Asp Lys Arg Val Glu
Ser Lys Tyr Gly 210 215 220Pro Pro Cys Pro Pro Cys Pro Ala Pro Glu
Phe Leu Gly Gly Pro Ser225 230 235 240Val Phe Leu Phe Pro Pro Lys
Pro Lys Asp Thr Leu Met Ile Ser Arg 245 250 255Thr Pro Glu Val Thr
Cys Val Val Val Asp Val Ser Gln Glu Asp Pro 260 265 270Glu Val Gln
Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn Ala 275 280 285Lys
Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr Tyr Arg Val Val 290 295
300Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu
Tyr305 310 315 320Lys Cys Lys Val Ser Asn Lys Gly Leu Pro Ser Ser
Ile Glu Lys Thr 325 330 335Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu
Pro Gln Val Tyr Thr Leu 340 345 350Pro Pro Ser Gln Glu Glu Met Thr
Lys Asn Gln Val Ser Leu Thr Cys 355 360 365Leu Val Lys Gly Phe Tyr
Pro Ser Asp Ile Ala Val Glu Trp Glu Ser 370 375 380Asn Gly Gln Pro
Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp385 390 395 400Ser
Asp Gly Ser Phe Phe Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser 405 410
415Arg Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala
420 425 430Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Leu
Gly 435 440 44519216PRTHomo sapiens 19Gln Ser Ala Leu Thr Gln Pro
Ala Ser Val Ser Gly Ser Pro Gly Gln1 5 10 15Ser Ile Thr Ile Ser Cys
Ile Gly Thr Ser Ser Asp Val Gly Gly Tyr 20 25 30Asn Tyr Val Ser Trp
Tyr Gln Gln His Pro Gly Lys Ala Pro Lys Leu 35 40 45Met Ile Tyr Asp
Val Ser Asn Arg Pro Ser Gly Val Ser Asn Arg Phe 50 55 60Ser Gly Ser
Lys Ser Gly Asn Thr Ala Ser Leu Thr Ile Ser Gly Leu65 70 75 80Gln
Ala Glu Asp Glu Ala Met Tyr Tyr Cys Ser Ala Tyr Thr Val Ser 85 90
95Ser Thr Trp Val Phe Gly Gly Gly Thr Lys Val Thr Val Leu Gly Gln
100 105 110Pro Lys Ala Ala Pro Ser Val Thr Leu Phe Pro Pro Ser Ser
Glu Glu 115 120 125Leu Gln Ala Asn Lys Ala Thr Leu Val Cys Leu Ile
Ser Asp Phe Tyr 130 135 140Pro Gly Ala Val Thr Val Ala Trp Lys Ala
Asp Ser Ser Pro Val Lys145 150 155 160Ala Gly Val Glu Thr Thr Thr
Pro Ser Lys Gln Ser Asn Asn Lys Tyr 165 170 175Ala Ala Ser Ser Tyr
Leu Ser Leu Thr Pro Glu Gln Trp Lys Ser His 180 185 190Arg Ser Tyr
Ser Cys Gln Val Thr His Glu Gly Ser Thr Val Glu Lys 195 200 205Thr
Val Ala Pro Thr Glu Cys Ser 210 215
D00000

D00001

D00002

D00003

D00004

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.