Extracranial Implantable Devices, Systems And Methods For The Treatment Of Neurological Disorders
DeGiorgio; Christopher ; et al.
U.S. patent application number 16/291763 was filed with the patent office on 2019-09-05 for extracranial implantable devices, systems and methods for the treatment of neurological disorders. This patent application is currently assigned to THE REGENTS OF THE UNIVERSITY OF CALIFORNIA. The applicant listed for this patent is NEUROSIGMA, INC., THE REGENTS OF THE UNIVERSITY OF CALIFORNIA. Invention is credited to Ian A. Cook, Christopher DeGiorgio, Leon Ekchian.
| Application Number | 20190269922 16/291763 |
| Document ID | / |
| Family ID | 43857099 |
| Filed Date | 2019-09-05 |



| United States Patent Application | 20190269922 |
| Kind Code | A1 |
| DeGiorgio; Christopher ; et al. | September 5, 2019 |
EXTRACRANIAL IMPLANTABLE DEVICES, SYSTEMS AND METHODS FOR THE TREATMENT OF NEUROLOGICAL DISORDERS
Abstract
The present disclosure relates to methods, devices and systems used for the treatment of neurological disorders via stimulation of the superficial elements of the trigeminal nerve ("TNS"). More specifically, minimally invasive methods of stimulation of the superficial branches of the trigeminal nerve located extracranially in the face, namely the supraorbital, supratrochlear, infratrochlear, auriculotemporal, zygomaticotemporal, zygomaticoorbital, zygomaticofacial, nasal, infraorbital, and mentalis nerves (also referred to collectively as the superficial trigeminal nerve) are disclosed herein. Systems and devices configured for therapeutic stimulation of the branches of the trigeminal nerves, such as the superficial trigeminal nerve, and their methods of application are also described.
| Inventors: | DeGiorgio; Christopher; (Valencia, CA) ; Cook; Ian A.; (Los Angeles, CA) ; Ekchian; Leon; (Glendale, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | THE REGENTS OF THE UNIVERSITY OF
CALIFORNIA Oakland CA NEUROSIGMA, INC. Los Angeles CA |
||||||||||
| Family ID: | 43857099 | ||||||||||
| Appl. No.: | 16/291763 | ||||||||||
| Filed: | March 4, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15144499 | May 2, 2016 | |||
| 16291763 | ||||
| 12898696 | Oct 5, 2010 | |||
| 15144499 | ||||
| 61248827 | Oct 5, 2009 | |||
| 61289829 | Dec 23, 2009 | |||
| 61305514 | Feb 17, 2010 | |||
| 61354641 | Jun 14, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61N 1/0476 20130101; A61N 1/3616 20130101; A61N 1/36096 20130101; A61N 1/36175 20130101; A61N 1/36021 20130101; A61N 1/0492 20130101; A61N 1/0551 20130101; A61N 1/36171 20130101; A61N 1/0529 20130101; A61N 1/0456 20130101; A61N 1/36067 20130101; A61N 1/36025 20130101; A61N 1/36064 20130101; A61N 1/36075 20130101; A61N 1/0504 20130101; A61N 1/36082 20130101 |
| International Class: | A61N 1/36 20060101 A61N001/36; A61N 1/04 20060101 A61N001/04; A61N 1/05 20060101 A61N001/05 |
Claims
1-20. (canceled)
21. A method for treating a neurological disorder or condition by trigeminal nerve stimulation, comprising: implanting a subcutaneous electrode assembly in a patient to place a first electrode contact at one side of a supraorbital nerve on the patient's forehead and to place a second electrode contact at an opposing side of the supraorbital nerve on the patient's forehead; and applying electrical signals to the subcutaneous electrode assembly so that current that flows between the first electrode contact and the second electrode contact at specified operational parameters stimulates the supraorbital nerve to treat the neurological disorder or condition, wherein the neurological disorder or condition is epilepsy.
22. The method of claim 21, wherein the electrical signals are applied to minimize current penetration into the patient's brain such that a charge density at a surface of the patient's brain does not exceed 20 .mu.C/cm.sup.2.
23. The method of claim 21, wherein the step of applying electrical signals comprises applying electrical signals at a frequency between approximately 20 and 300 Hz, at a current of 0.1 to 3 mA, and at a pulse duration of less than or equal to 500 .mu.s.
24. The method of claim 21, wherein the step of applying electrical signals comprises applying electrical signals at a frequency between approximately 20 and 300 Hz, at a pulse duration between approximately 50 and 500 .mu.s, at an output current density of not greater than approximately 25 mA/cm.sup.2, and a charge density of not greater than approximately 10 .mu.C/cm.sup.2 at the patient's cerebral cortex.
25. The method of claim 21, wherein the implanting comprises implanting the subcutaneous electrode assembly in the patient to place a third electrode contact at one side of a supratrochlear nerve on the patient's forehead and to place a fourth electrode contact at an opposing side of the supratrochlear nerve on the patient's forehead.
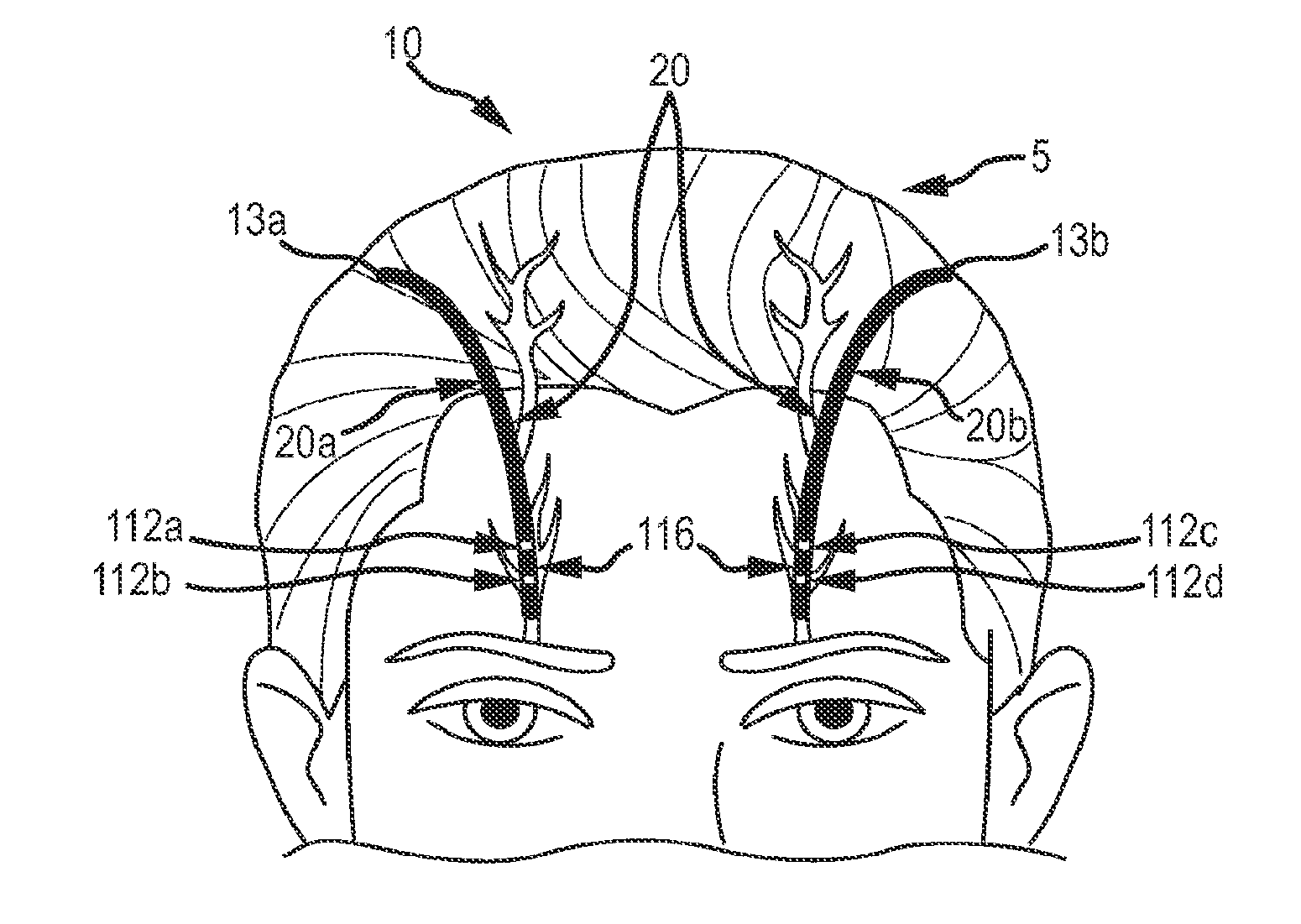
26. The method of claim 25, wherein the method further comprises applying electrical signals to the subcutaneous electrode assembly so that current that flows between the third electrode contact and the fourth electrode contact at specified operational parameters stimulates the supratrochlear nerve to treat the neurological disorder or condition.
27. The method of claim 21, wherein the implanting comprises implanting the subcutaneous electrode assembly in the patient to place a fifth electrode contact at one side of an infraorbital nerve and to place a sixth electrode contact at an opposing side of the infraorbital nerve.
28. The method of claim 27, wherein the method further comprises applying electrical signals to the subcutaneous electrode assembly so that current that flows between the fifth electrode contact and the sixth electrode contact at specified operational parameters stimulates the infraorbital nerve to treat the neurological disorder or condition.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation application of U.S. application Ser. No. 15/144,499 filed on May 2, 2016, which itself is a continuation application of U.S. application Ser. No. 12/898,696, entitled "Extracranial Implantable Devices, Systems and Methods for the Treatment of Neurological Disorders," filed Oct. 5, 2010, which claims the benefit of U.S. Provisional Application No. 61/248,827, entitled "Devices and Methods for Treatment of Psychiatric Disorders," filed Oct. 5, 2009, U.S. Provisional Application No. 61/289,829, entitled "Extracranial Implantable Devices, Systems and Methods for the Treatment of Neuropsychiatric Disorders," filed Dec. 23, 2009, U.S. Provisional Application No. 61/305,514, entitled "Systems, Devices and Methods for the Treatment of Neurological Disorders and Conditions," filed Feb. 17, 2010, and U.S. Provisional Application No. 61/354,641, entitled "Extracranial Implantable Devices, Systems and Methods for the Treatment of Neurological Disorders," filed Jun. 14, 2010, all of which are incorporated herein by reference in their entirety.
[0002] This application is also related to U.S. application Ser. No. 12/898,685, entitled "Extracranial Implantable Devices, Systems and Methods for Treatment of Neuropsychiatric Disorders," filed Oct. 5, 2010, now U.S. Pat. No. 8,958,880, issued Feb. 17, 2015, U.S. application Ser. No. 12/898,675, entitled "Systems, Devices and Methods for the Treatment of Neurological Disorders and Conditions," filed Oct. 5, 2010, now U.S. Pat. No. 8,688,220, issued Apr. 1, 2014, and U.S. application Ser. No. 12/898,686, entitled "Devices, Systems and Methods for Treatment of Neuropsychiatric Disorders," filed Oct. 5, 2010, now U.S. Pat. No. 8,380,315, issued Feb. 19, 2013, all of which are incorporated by reference in their entirety.
FIELD
[0003] The present disclosure generally relates to neurostimulator systems, devices, and methods of using the same and more particularly relates to neurostimulator systems, devices and methods including at least one implantable electrode for treating neurological disorders by stimulating superficial, cutaneous elements of cranial nerve(s).
BACKGROUND
[0004] Neurological disorders and conditions, such as seizure disorders that are characterized by epileptic seizures, acute or chronic brain injury, coma, chronic headache or migraine, and movement and related disorders may be treated with medication. For example, seizure disorders, frequently referred to as epilepsy, are treated initially with anti-epileptic drugs. In some patients, anti-epileptic drugs fail; for these patients, resective epilepsy surgery or neurostimulation are therapeutic options. Neurostimulation for epilepsy and seizure disorders may include stimulation of the nervous system by vagus nerve stimulation (VNS), which has been shown to be therapeutically useful and has been approved by the U.S. Food and Drug Administration. In this method, stimulating electrodes are surgically implanted on the vagus nerve in the neck. In addition to complications related to anesthesia, potential for infection, cost, and other adverse events with VNS, many of the subjects who undergo VNS treatments do not achieve relief from their seizures, and there is no reliable predictor of good outcomes from the implanted VNS device.
[0005] Other approaches are the focus of on-going research. For example, intracranial implantable approaches, such as deep brain stimulation (DBS) of the anterior thalamus and responsive neurostimulation (RNS) of the epileptic zone may be utilized. RNS employs a device which monitors brain activity and delivers stimuli to terminate seizure discharges. However, these methods are highly surgically invasive because they involve placement of electrodes within the brain, on the surface of the brain, or near sensitive neuro-vascular structures. In addition, these treatments may also have increased costs and side effects compared with other, less invasive approaches. Despite this range of options, a substantial percentage of patients do not recover from or get adequate relief for the neurological disorder despite multiple trials of pharmaceutical or surgical treatment.
SUMMARY
[0006] One aspect of the subject matter of the present disclosure addresses the aforementioned needs by providing systems and devices configured to stimulate cutaneous or superficial branches of the trigeminal nerve, located in the face, including the following nerves: the ophthalmic (supra-orbital), supratrochlear and infratrochlear, auriculotemporal and zygomaticotemporal, zygomaticofacial, zygomaticoorbital, infraorbital, nasal and mentalis nerve(s) and methods of using the same to treat neurological disorders. This disclosure also provides a method of treating neurological disorders using trigeminal nerve stimulation (TNS) with minimally invasive, implantable and easy-to-use devices and systems.
[0007] In another aspect of the present disclosure, an implantable subcutaneous electrode assembly configured for trigeminal nerve stimulation is provided.
[0008] In yet another aspect of the present disclosure, a method of treating neurological disorders using the disclosed implantable or subcutaneous electrode assembly is provided.
[0009] Disclosed herein is a subcutaneous electrode assembly for trigeminal nerve stimulation. In one embodiment, the subcutaneous electrode assembly includes a first electrode comprising a first pair of contacts configured for subcutaneous placement at a first region of a patient's face; and a second electrode comprising a second pair of contacts configured for subcutaneous placement at a second region of a patient's face, wherein the first pair of contacts and the second pair of contacts are configured to be bilaterally implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve for treatment of a neurological disorder by trigeminal nerve stimulation. The at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve and auriculotemporal nerve.
[0010] Disclosed herein is a subcutaneous electrode assembly for trigeminal nerve stimulation. In one embodiment, the subcutaneous electrode assembly includes a first electrode comprising a first plurality of contacts configured for subcutaneous placement at a first region of a patient's face; and a second electrode comprising a second plurality of contacts configured for subcutaneous placement at a second region of a patient's face; wherein the first plurality of contacts and the second plurality of contacts are configured to be unilaterally implanted in proximity to, adjacent to or in contact with at least two different branches of the trigeminal nerve for treatment of a neurological disorder by trigeminal nerve stimulation. The at least two different branches of the trigeminal nerve are selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, nasal nerve, zygomaticoorbital nerve, and auriculotemporal nerve.
[0011] Disclosed herein is a method for treating a neurological disorder or condition by trigeminal nerve stimulation. In one embodiment, the method includes implanting an electrode assembly in a patient, the electrode assembly includes: a first electrode comprising a first plurality of contacts configured for subcutaneous placement at a first region of the patient's face; a second electrode comprising a second plurality of contacts configured for subcutaneous placement at a second region of the patient's face; and wherein the first plurality of contacts and the second plurality of contacts are configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve; and applying electrical signals to the electrode assembly at specified operational parameters to treat a neurological disorder or condition. In one embodiment, the step of applying electrical signals includes applying electrical signals at a frequency between approximately 20 and 300 Hertz, at a current of 0.05 to 5 milliamperes (mA), at a pulse duration of less than or equal to 500 microseconds. In one embodiment, the step of applying electrical signals comprises applying electrical signals at a frequency between approximately 20 and 300 Hertz, at a current of 0.05 to 2 milliamperes (mA) and at a pulse duration not exceeding 500 microseconds. In one embodiment, the neurological disorder or condition is selected from the group consisting of: epilepsy and other seizure related disorders, acute or chronic brain injury, chronic daily headache and migraine and related disorders, and movement disorders.
[0012] Disclosed herein is a system for trigeminal nerve stimulation for treatment of a neurological disorder or condition. In one embodiment, the system includes: a pulse generator; and a subcutaneous electrode assembly including: a first electrode comprising a first plurality of contacts configured for subcutaneous placement at a first region of the patient's face; a second electrode comprising a second plurality of contacts configured for subcutaneous placement at a second region of the patient's face; and wherein the first plurality of contacts and the second plurality of contacts are configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve. The system may further comprise a wire operably connecting the pulse generator and the subcutaneous electrode assembly. In some embodiments, the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, nasal nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, and auriculotemporal nerve.
[0013] Disclosed herein is a subcutaneous electrode assembly for trigeminal nerve stimulation. In one embodiment, the subcutaneous electrode assembly includes: a first electrode comprising a first single contact configured for subcutaneous placement at a first region of a patient's face; and a second electrode comprising a second single contact configured for subcutaneous placement at a second region of a patient's face; wherein the first contact and the second contact are configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve for treatment of a neurological disorder by trigeminal nerve stimulation. In some embodiments, the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, and auriculotemporal nerve.
[0014] Disclosed herein is a system for trigeminal nerve stimulation for treatment of a neurological disorder or condition. In one embodiment, the system includes: a pulse generator; and a subcutaneous electrode assembly in electrical communication with the pulse generator, the assembly comprising: a first electrode comprising at least one contact configured for subcutaneous placement at a first region of the patient's face, wherein the first electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve for treatment of a neurological disorder or condition by trigeminal nerve stimulation, wherein the system is configured for minimal current penetration into a brain of a patient, and wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve. In some embodiments, the assembly further comprises a second electrode comprising at least one contact configured for subcutaneous placement at a second region of the patient's face, wherein the second electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve, wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve. In some embodiments, the first electrode and the second electrode are configured for implantation in proximity to, adjacent to or in contact with a same branch of the trigeminal nerve. In some embodiments, the first electrode and the second electrode are configured for implantation in proximity to, adjacent to or in contact with a different branch of the trigeminal nerve. In some embodiments, the system may further include a wire operably connecting the pulse generator and the subcutaneous electrode assembly. In some embodiments, the system may further include a regulating device configured to regulate the maximum charge balanced output current below approximately 30-50 mA. The neurological disorder or condition is selected from the group consisting of: epilepsy, seizure related disorders, acute brain injury, chronic brain injury, chronic daily headache, migraine, disorders related to migraine and headache and movement disorders. In some embodiments, the pulse generator is configured to apply electrical signals at a frequency between approximately 20 and 300 Hertz, at a pulse duration between approximately 50 and 500 microseconds, at an output current density of not greater than approximately 25 mA/cm.sup.2 and an output charge density of not greater than approximately 10 microCoulomb/cm.sup.2 at the cerebral cortex.
[0015] Disclosed herein is a subcutaneous electrode assembly for trigeminal nerve stimulation for treatment of a neurological disorder or condition. In some embodiments, the assembly includes: a first electrode comprising at least one contact configured for subcutaneous placement at a first region of the patient's face, wherein the first electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve for treatment of a neurological disorder or condition by trigeminal nerve stimulation, wherein the assembly is configured for minimal current penetration into a brain of a patient, and wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve. In some embodiments, the assembly may further include a second electrode comprising at least one contact configured for subcutaneous placement at a second region of the patient's face, wherein the second electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve, wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve. In some embodiments, the first electrode and the second electrode are configured for implantation in proximity to, adjacent to or in contact with a same branch of the trigeminal nerve. In some embodiments, the first electrode and the second electrode are configured for implantation in proximity to, adjacent to or in contact with a different branch of the trigeminal nerve. The neurological disorder or condition is selected from the group consisting of: epilepsy, seizure related disorders, acute brain injury, chronic brain injury, chronic daily headache, migraine, disorders related to migraine and headache, and movement disorders
[0016] Disclosed herein is a method for treating a neurological disorder or condition by trigeminal nerve stimulation. In one embodiment, the method includes implanting an electrode assembly in a patient, the subcutaneous electrode assembly comprising: a first electrode comprising at least one contact configured for subcutaneous placement at a first region of the patient's face, wherein the first electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve for treatment of a neurological disorder or condition by trigeminal nerve stimulation, wherein the assembly is configured for minimal current penetration into a brain of a patient, and wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve; and applying electrical signals to the electrode assembly at specified operational parameters to treat a neurological disorder or condition. In some embodiments, the method further includes an assembly comprising a second electrode comprising at least one contact configured for subcutaneous placement at a second region of the patient's face, wherein the second electrode is configured to be implanted in proximity to, adjacent to or in contact with at least one branch of the trigeminal nerve, wherein the at least one branch of the trigeminal nerve is selected from the group consisting of: ophthalmic nerve, infraorbital nerve, mentalis nerve, supratrochlear nerve, infratrochlear nerve, zygomaticotemporal nerve, zygomaticofacial nerve, zygomaticoorbital nerve, nasal nerve, and auriculotemporal nerve. In some embodiments, the step of applying electrical signals comprises applying electrical signals at a frequency between approximately 20 and 300 Hertz, at a current of 0.05 to 5 milliamperes (mA) and at a pulse duration of less than or equal to 500 microseconds. In some embodiments, the step of applying electrical signals comprises applying electrical signals at a frequency between approximately 20 and 300 Hertz, at a pulse duration between approximately 50 and 500 microseconds, at an output current density of not greater than approximately 25 mA/cm.sup.2 and a charge density of not greater than approximately 10 microCoulomb/cm.sup.2 at the cerebral cortex. The neurological disorder or condition is selected from the group consisting of: epilepsy, seizure related disorders, acute brain injury, chronic brain injury, chronic daily headache, migraine, disorders related to migraine and headache and movement disorders.
[0017] Disclosed herein is a kit for trigeminal nerve stimulation for treatment of a neurological disorder or condition. In some embodiments, the kit includes a subcutaneous electrode assembly as disclosed herein; and instructions for implanting the electrode assembly in a patient for treatment of a neurological disorder or condition. In some embodiments the kit may further include: a pulse generator; and instructions for applying electrical signals to the electrode assembly for treatment of a neurological disorder or condition.
BRIEF DESCRIPTION OF THE DRAWINGS
[0018] The present disclosure, both as to its organization and manner of operation, may be understood by reference to the following description, taken in connection with the accompanying drawings, in which:
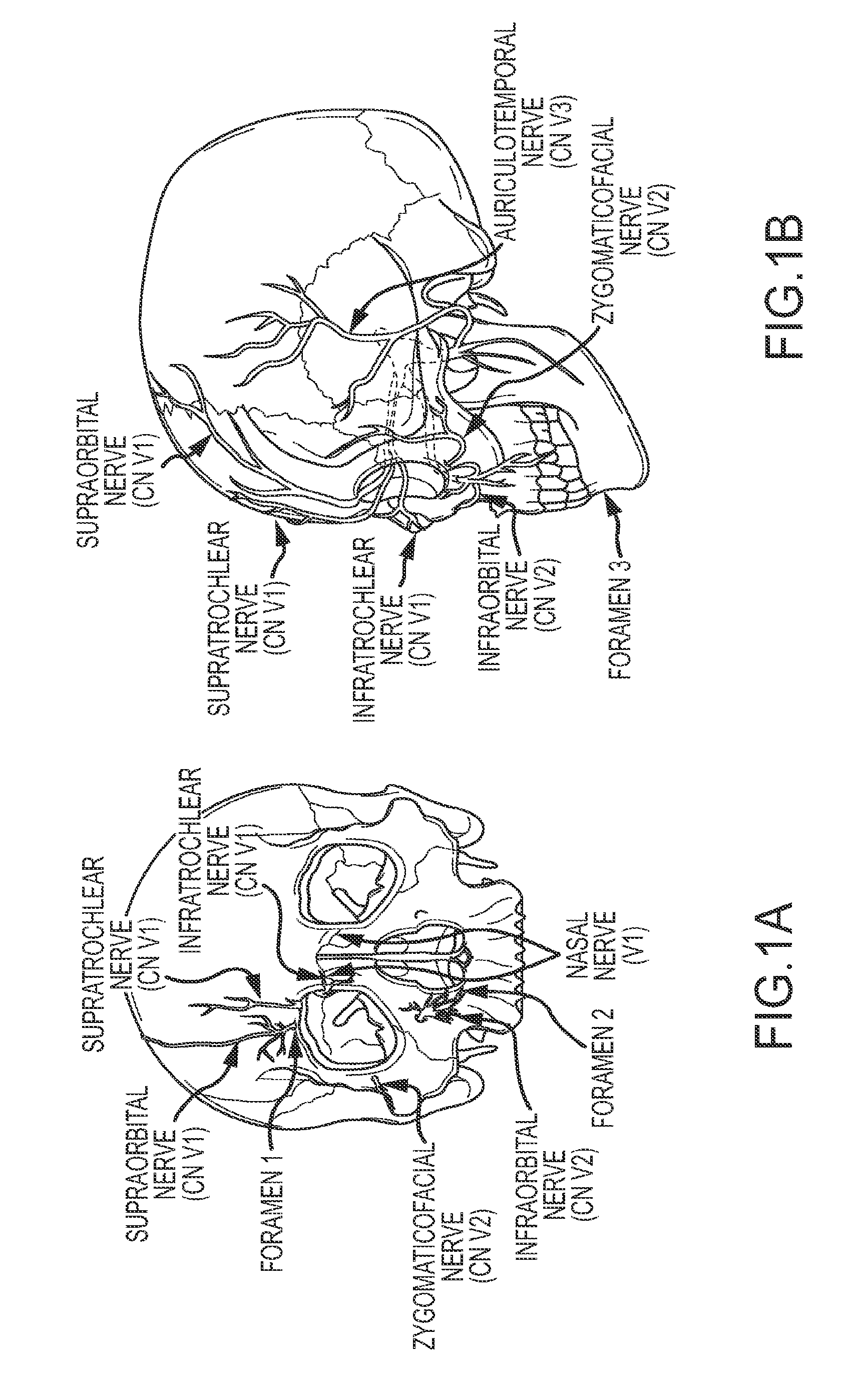
[0019] FIGS. 1A and 1B illustrate the location of several branches (nerves) of the trigeminal nerve and the location of the major foramina for the superficial branches of the trigeminal nerve;
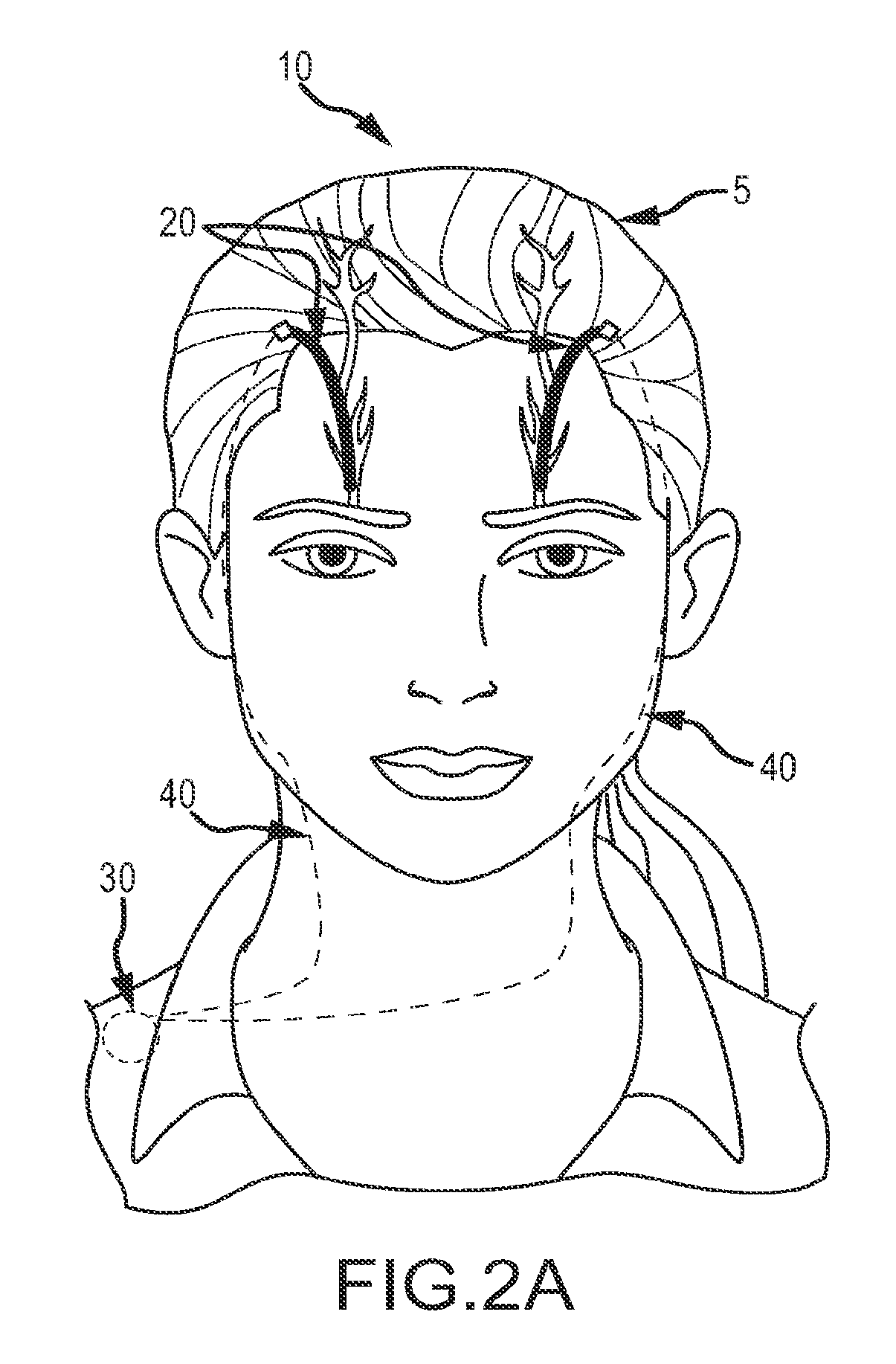
[0020] FIG. 2A shows a subject wearing an embodiment of a system for trigeminal nerve stimulation including a subcutaneous electrode assembly provided according to aspects of the present disclosure;
[0021] FIG. 2B is the subcutaneous electrode assembly of FIG. 2A, wherein a multicontact electrode is shown;
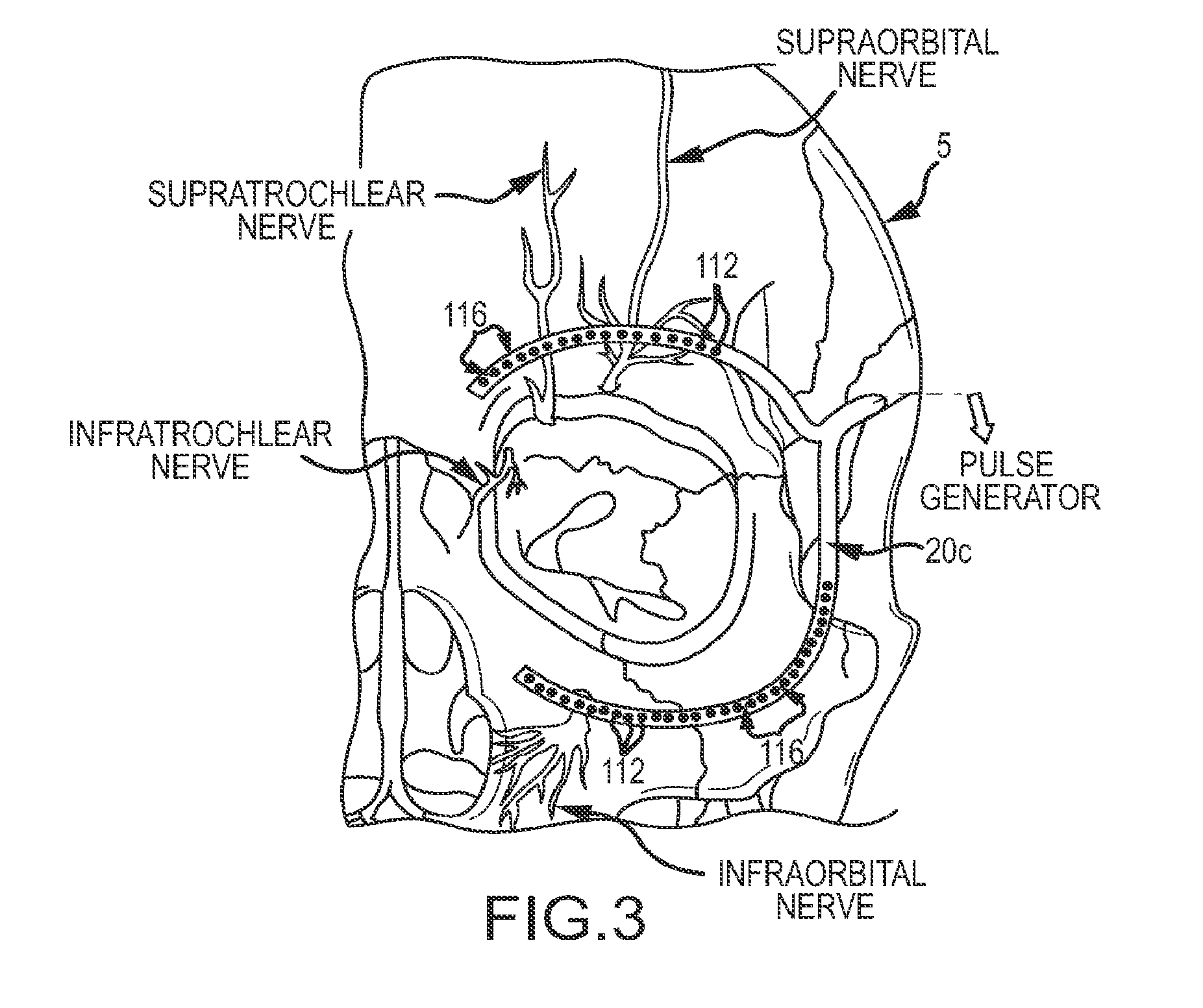
[0022] FIG. 3 depicts another embodiment of a subcutaneous electrode assembly configured for stimulation of a plurality of nerve branches which may be used with the system of FIG. 2A;
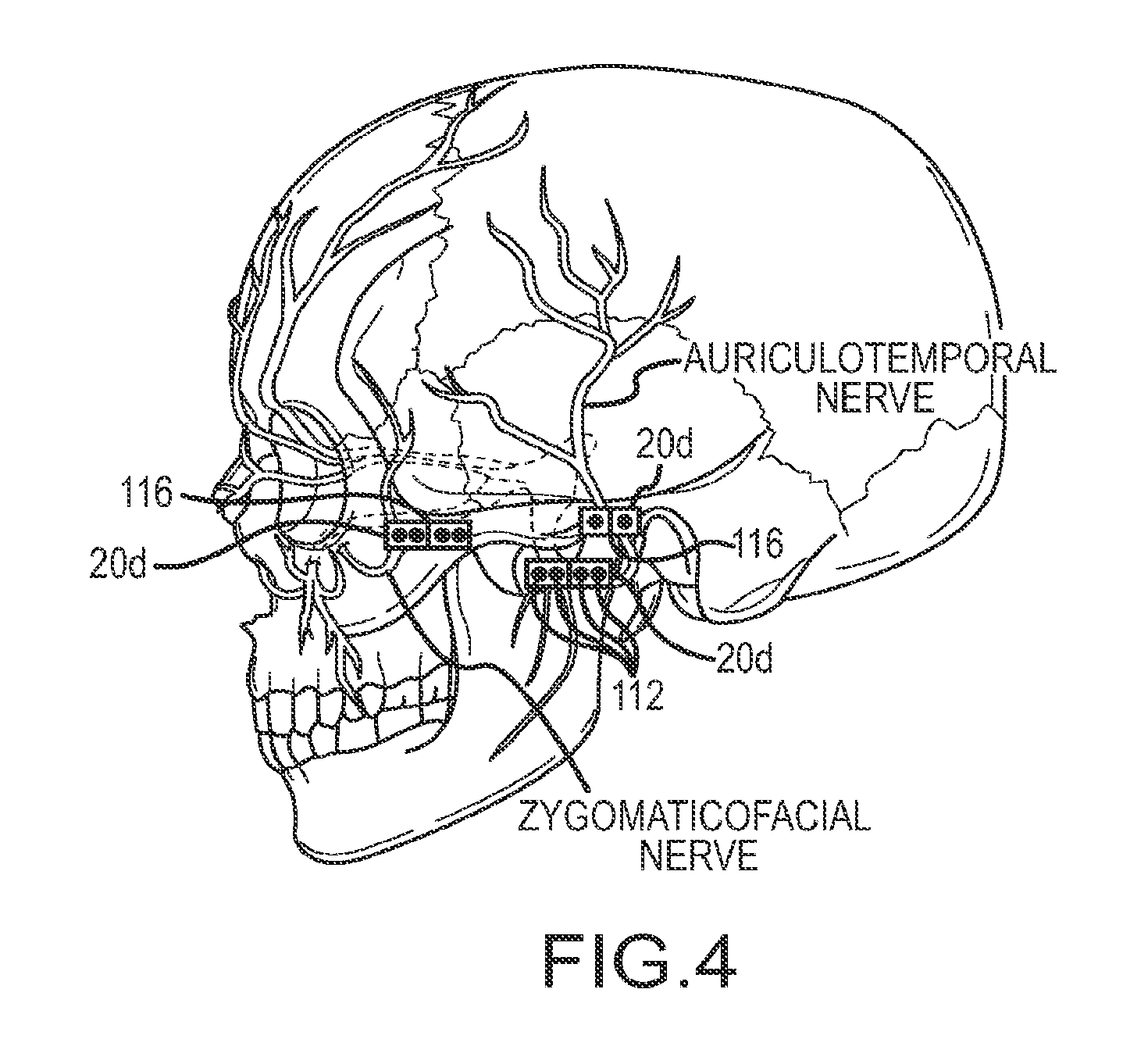
[0023] FIG. 4 depicts another embodiment of a subcutaneous electrode assembly configured for stimulation of the auriculotemporal or zygomaticofacial nerve branches which may be used with the system of FIG. 2A;
[0024] FIG. 5 illustrates the results from a pilot study of external trigeminal nerve stimulation ("TNS"); and
[0025] FIG. 6 summarizes one embodiment of current, charge, current density and charge density parameters for a subject exposed to transcutaneous stimulation of the supraorbital nerve.
DETAILED DESCRIPTION
[0026] The present disclosure relates to methods, devices and systems used for the treatment of neurological disorders via stimulation of the superficial elements of the trigeminal nerve ("TNS"). More specifically, minimally invasive methods of stimulation of the superficial branches of the trigeminal nerve located extracranially in the face, namely the supraorbital, supratrochlear, infratrochlear, auriculotemporal, zygomaticotemporal, zygomaticoorbital, zygomaticofacial, infraorbital, and mentalis nerves (also referred to collectively as the superficial trigeminal nerve) are disclosed herein. Systems and devices configured for therapeutic stimulation of the branches of the trigeminal nerves, such as the superficial trigeminal nerve, and their methods of application are also described. The cutaneous branches of the trigeminal nerve in the face provide an opportunity for a minimally invasive method of stimulating structures of the brain and the brainstem including, but not limited to, the trigeminal nerve nuclei and tracts, locus coeruleus, nucleus tractus solitarius, ventral posterior and ventral medial thalamus, the cerebral cortex, and other structures which may play a role in the disorders listed above.
[0027] The systems, devices and methods disclosed herein provide a less invasive form of neurostimulation to treat a variety of neurological disorders including, but not limited to, seizures, headache, migraine and related disorders, movement disorders, coma, and brain injury. More specifically, an implantable or subcutaneous electrode assembly and a system comprising the same configured for trigeminal nerve stimulation are disclosed herein. As described in more detail below, electrodes are not placed within the brain or near critical structures like the vagus nerve, carotid artery, or jugular vein. The electrodes are also not directly or physically attached or anchored to the nerve (e.g. by suturing), which requires intracranial invasion and may cause a spinal fluid leak, infection, nerve damage and/or severe pain. Instead, subcutaneous electrodes (or an electrode assembly) are placed at or near a region of a patient's face or cranium that is in proximity to, adjacent to, in contact with, or distal to the trigeminal nerve (or the relevant branch(es) thereof) by attaching to subcutaneous or connective tissues above the periosteum or pericranium (a membrane that lines the outer surface of the skull) and below the epidermis (the outermost layer of skin). The nerve is stimulated at operational parameters within a defined range to minimize current penetration into the brain and further determined by factors such as patient history, disorder to be treated, or individual sensitivity to the stimulation. The electrode assembly placement as described herein does not require intracranial invasion (i.e. implantation below the skull) thereby reducing the risks of a spinal fluid leak and infection. In some embodiments, the electrode assembly may be placed or otherwise configured to stimulate the smaller branches of the trigeminal nerve. That is, the assembly is placed further away from the brain and the main branch of the nerve. Surprisingly, placement of the assembly further away from the brain and the main branch of the nerve is believed to be as efficacious as direct attachment to the main branch of the nerve and may provide increased safety for the patient.
[0028] In some clinical situations, brain stimulation has been found to be of sufficient clinical use to have been approved by the US Food and Drug Administration, for example, electroconvulsive therapy (ECT) and repetitive transcranial magnetic stimulation (rTMS) for psychiatric conditions. Some brain stimulation methods aim to generate currents in large volumes of the cortex and treat the brain as a bulk conductor, for example, ECT at the whole-lobe level and rTMS at the large regional level (i.e. dorsolateral prefrontal cortex). Additionally, deep brain stimulation is generally predicated on stimulation of small but regional volumes that lead to discharges in a very large number of cells. The systems, devices and methods of the present disclosure send minimal, if any, current into the brain; instead, signals are sent into the brain in order to modulate the activity of relevant neuroanatomical structures. Without wishing to be bound by any particular theory, the electrical pulses generate signals in the cutaneous branches of the trigeminal nerve and the electric fields are generally confined to the skin tissue and there is minimal, if any, leakage into the brain. These electrical pulses trigger a cascade of change in neuronal signaling events that involve very limited and precise recruitment of specific networks of neurons. The neuroanatomic pathways allow targeted modulation of activity in areas involved in depression (locus coeruleus, anterior cingulate, insular cortex). Thus, the systems, devices and methods as disclosed herein utilize the brain's existing infrastructure to transmit signals to the targets of interest. In the context of this disclosure minimal current penetration means (1) a charge density of approximately 0 .mu.C/cm.sup.2 at the cerebral cortex, or (2) calculated, measured, or modeled charge densities below the following thresholds at the cerebral cortex: (a) at currents, charge densities, or charge per phase not likely to cause activation of pyramidal neurons and axons; and (b) to prevent brain injury, a charge density of less than 10 .mu.C/cm.sup.2 in one embodiment, and, in other embodiments, a charge density of less than 0.001 to 0.1 .mu.C/cm.sup.2, and at combinations of charge density and charge per phase not known to cause brain injury. In some embodiments, a lower charge density may be used when the central nervous system of an individual patient is sufficiently sensitive to lower levels of stimulation that the lower level will still permit clinical benefit to accrue.
[0029] The following description is provided to enable any person skilled in the art to make and use the subject matter of this disclosure, and it sets forth the best modes contemplated by the inventors of carrying out the various aspects of the disclosure. Various modifications, however, will remain readily apparent to those skilled in the art, since the principles of the disclosed subject matter have been defined herein specifically to describe: (1) methods of treating neurological disorders by trigeminal nerve stimulation, (2) a system and an implantable electrode assembly configured for trigeminal nerve stimulation; and (3) methods of treating neurological disorders using such system and electrode assembly.
[0030] For a discussion related to the trigeminal nerve, reference is first made to FIGS. 1A-1B, which illustrate the location of several branches of the trigeminal nerve and the location of the major foramina for the superficial branches of the trigeminal nerve. The trigeminal nerve is the largest of the 12 paired cranial nerves, and has extensive connections with the brainstem and other brain structures. The trigeminal nerve (frequently identified as the fifth cranial nerve, cranial nerve V, or CN V) has three major divisions, the cutaneous branches of which are all bilateral and highly accessible. The supraorbital nerve, or ophthalmic nerve, is frequently referred to as the V1 division and this division also includes the supratrochlear nerve and the infratrochlear nerve. The infraorbital branch or maxillary nerve is commonly referred to as the V2 division and this division also includes the zygomaticofacial nerve and the infraorbital nerve. The mentalis branch of the mandibular nerve is referred to as the V3 division and this division also includes the auriculotemporal nerve. The supraorbital nerve supplies sensory information about the forehead, the upper eyelid, the anterior part of the nose, and the eye. The infraorbital branch supplies sensory information about the lower eyelid, cheek, and upper lip. The mentalis branch supplies sensory information about the skin of the lower face (e.g. jaw and tongue) and lips. These branches exit the skull through three foramina, as shown in FIGS. 1A-B. The supraorbital nerve or ophthalmic nerve exits at foramen 1, approximately 2.1-2.6 cm from the nasal midline (in adults), and is located immediately above the orbital ridge that is located below the eyebrow. The infraorbital branch or maxillary nerve exits at foramen 2, approximately 2.4-3.0 cm from the nasal midline (in adults) and the mentalis nerve exits at foramen 3, approximately 2.0-2.3 cm from the nasal midline (in adults). The nasal nerve is a division of the ophthalmic nerve. Other sensory branches, including the zygomaticofacial, zygomaticoorbital, zygomaticotemporal, and auriculotemporal, arise from other foramina.
[0031] Fibers from the three major branches join together to form the trigeminal ganglion. From there, fibers ascend into the brainstem at the level of the pons, to synapse with the main sensory nucleus of the pons and the spinal nucleus and tract of CN V. Pain fibers descend in the spinal nucleus and tract of V, and then ascend to the ventral posterior medial nucleus (VPM) of the thalamus, and then project to the cerebral cortex. Light touch sensory fibers are large myelinated fibers, which ascend to the ventral posterior lateral (VPL) nucleus of the thalamus, and also project to the cerebral cortex.
[0032] The trigeminal nuclei have projections to other cranial nerve structures, including the nucleus tractus solitarius (NTS), and the locus coeruleus, among others. The NTS receives afferents from the vagus nerve and trigeminal nerve. NTS integrates input from multiple sources, and projects to structures in the brainstem and forebrain, including the locus coeruleus. The locus coeruleus is a paired nuclear structure in the dorsal pons, and is located just beneath the floor of the fourth ventricle. The locus coeruleus has extensive axonal projections to a large number of brainstem, sub-cortical and cortical structures, and is an important part of the reticular activating system. The locus coeruleus is a core part of the brainstem noradrenergic pathway, and produces the neurotransmitter norepinephrine. Norepinephrine may play a role in attention, alertness, blood pressure and heart rate regulation, anxiety, and mood.
[0033] While not wishing to be bound by any particular theory, in certain embodiments, the connections between the trigeminal nerve, locus coeruleus, nucleus and tractus solitarius, thalamus, and cerebral cortex, may be relevant to a potential role of the trigeminal nerve in numerous neurological disorders, including coma and brain injury, seizure disorders, headache, migraine, and movement disorders, as may be apparent to one skilled in the art. Thus, subcutaneous stimulation of the trigeminal nerve at custom tailored settings and parameters could be effective in the treatment of multiple neurological disorders.
[0034] Neurological Disorders
[0035] Coma and Vegetative State. Subcutaneous neurostimulation may improve consciousness in persons in coma and vegetative state. Without wishing to be bound by a particular theory, the brainstem reticular activating system (including locus coeruleus) and thalamus may play a role in alerting, awakening, and activating higher cortical structures. Stimulation of these and other brain structures, to which the trigeminal nerve and nuclei project, could assist in promoting awakening in coma, as well as recovery of cognition and motor function after various forms of brain injury. Given the projections of the trigeminal nerve to key brainstem, thalamic, and cortical structures involved in wakefulness and consciousness, the trigeminal nerve represents one method to activate these key structures.
[0036] Headache and Migraine. Without wishing to be bound by a particular theory, headache and migraine involve pathways linked to the trigeminal nerve. Activation of specific trigeminal structures and pathways may play a role in headache. (Nature Medicine 2002; 8:136-142). Afferent trigeminal nerve fibers from vascular structures in the pia covering the cerebral cortex are activated, and activate or sensitize the trigeminal ganglion and the caudal trigeminal nuclei, which in turn activate the superior salvitory nucleus and the sphenopalatine ganglia. (Nature Medicine 2002; 8:136-142). Projections from these structures to vessels in the dura mater (the outer protective lining of the brain) lead to the release of vasoactive peptides, protein extravasation, and activation of nitric acid pathways, all of which result in dilatation of dural vessels, which may lead to headache. This is frequently referred to as the trigeminal-vascular reflex, and may be a mechanism in the genesis of migraine. (Nature Medicine 2002; 8:136-142). Without wishing to be bound by a particular theory, surgically lesioning or blocking the trigeminal nerve may inhibit this response, leading to a reduction in the cascade of events involved with migraine and other headache syndromes. As disclosed herein, acute or chronic electrical stimulation of the trigeminal nerve via its cutaneous or superficial braches in the face, at frequencies which inhibit the circuit described above, is one method to modulate this trigeminal-vascular reflex response, and reduce or inhibit headaches or migraines in which the trigeminal nuclei and nerves play a role.
[0037] Movement Disorders. Movement disorders are characterized by involuntary movements of the body, and include, but are not limited to, tremors, twitches, and spasms, involuntary increases in tone of muscles, such as dystonias, and complex movements, such as dyskinesias and choreas. Without wishing to be bound by any particular theory, we hypothesize that TNS may modulate activity in key structures involved in movement disorders, including but not limited to the thalamus, basal ganglia, brain stem, and cerebral cortex, and may inhibit, by afferent stimulation, abnormal neuronal activity in motor systems which give rise to these involuntary phenomena.
[0038] Tardive and other Dyskinesias. Many medications which act on the dopaminergic neurons in the brain have a liability for inducing involuntary movements. This has been reported for treatment of Parkinson's disease with levodopa, for the use of neuroleptic medications in psychosis, bipolar disorder, and other conditions (Damier, Curr Opin Neurol 22:394-399, 2009), and for dopaminergic medications used to address gastrointestinal symptoms (Rao and Camilleri, Ailment Pharmacol Ther 31:11-19. 2010). Other individuals may suffer from dyskinesia on a genetic-related basis (Coubes et al., Lancet 355:2220-1, 2000). These dyskinesia syndromes consist of involuntary movements that usually start oro-facially, with the muscles of the tongue, lips, mouth or face, but can increase in severity and come to involve other parts of body. The exact mechanisms by which these dyskinesias arise is not clear, but surgical treatment approaches have implicated the thalamus and the globus pallidum as locations where deep brain stimulation can lead to improvement (Kupsch et al., J Neurol 250 Suppl 1:147-152 2003). While not wishing to be bound by any particular theory, the connections between the trigeminal nerve, nucleus and tractus solitarius, and thalamus may provide a mechanism by which trigeminal nerve stimulation can ameliorate symptoms of dyskinesia by activating these key structures.
[0039] Seizure Disorders. Without wishing to be bound by any particular theory, trigeminal nerve stimulation may modulate activity in the locus coeruleus, brainstem, thalamus, and cerebral cortex, and may activate inhibitory mechanisms and pathways which affect neuronal excitability. Trigeminal nerve stimulation may also inhibit excitatory mechanisms and pathways, resulting in inhibition of epileptic discharges and their spread in cortex, and subcortical structures. These processes may have a direct or indirect effect on activity in the epileptic focus itself.
[0040] Accordingly, stimulation of the superficial or cutaneous branches of the trigeminal nerve as disclosed herein provide a minimally invasive neuromodulation option. Further, stimulation parameters can be tailored for the individual condition, such that the brainstem, thalamic, or cortical structures involved in the individual condition can be activated or inhibited depending on the pathophysiology of the condition being treated.
[0041] For a discussion of certain embodiments of methods, systems and devices using implantable electrodes according to aspects of the present disclosure, reference is now made to FIGS. 2A-4 which show various embodiments of the systems and devices that may be used for the subcutaneous stimulation of the superficial branches of the trigeminal nerve and methods of using the same.
[0042] According to one aspect of the present disclosure, a method of treating neurological disorders using trigeminal nerve stimulation ("TNS") is provided. In some embodiments, the method of treating these disorders by stimulating superficial branches of the trigeminal nerve comprises implanting electrodes adjacent to, in proximity to, in contact with, or distal to at least one of the three paired foramina or superficial branches of the trigeminal nerves in the face (FIGS. 1A-1B), and stimulating the electrodes using a pulse generator for a period of time at specified operational parameters. The electrode assembly placement does not require intracranial invasion (i.e. implantation below the skull) because the electrode assembly is attached or otherwise anchored to subcutaneous or connective tissues located above the periosteum or pericranium and below the epidermis in order to place the electrode assembly in proximity to, adjacent to, in contact with or distal to the target nerve branch. In some embodiments, the electrode assembly may be configured to stimulate the smaller branches of the trigeminal nerve. Surprisingly, placement of the assembly further away from the brain and the main branch of the nerve is believed to be as efficacious as direct attachment or other contact with the main branch of the nerve and may provide increased safety for the patient.
[0043] In one embodiment, the implanted electrodes are positioned adjacent to the foramina of the supraorbital or ophthalmic nerves (FIGS. 1A-1B, Foramen 1) since unilateral stimulation or bilateral stimulation of the trigeminal nerve is achievable by placing single or separate electrodes on the right and/or left sides. In one embodiment, the electrode assembly is configured for unilateral stimulation. In one embodiment, the electrode assembly is configured for bilateral stimulation. In some embodiments, bilateral stimulation may offer similar or better efficacy than unilateral stimulation because the function of different brain structures may not be the same on right and left (e.g. verbal expression is most commonly localized to speech centers in the left hemisphere, and injury there produces catastrophic loss of the ability to speak, while damage to the corresponding region on the right does not produce this profound loss of function, but may alter subtle functions). There may also be synergistic effects that arise with bilateral stimulation.
[0044] In some embodiments, a patient may be implanted with two separate electrodes in the soft tissues of the forehead, with each electrode near the foramen or branches of the ophthalmic nerve. In alternative embodiments, the implanted/implantable electrode(s) can also be positioned adjacent to, in proximity to, or in contact with the infraorbital foramen (infraorbital nerves) (FIGS. 1A-1B, Foramen 2) or the mentalis foramen (mentalis nerves) (FIGS. 1A-1B, Foramen 3). In other embodiments, electrodes may be placed adjacent to, in proximity to, or in contact with the supratrochlear nerve, infratrochlear nerve, zygomaticotemporal, zygomaticofacial, zygomaticoorbital, nasal, and/or auriculotemporal nerves and their respective foramina. Unilateral stimulation or bilateral stimulation of the trigeminal nerve is achievable by placing single or separate electrodes on the right and/or left sides of the face to unilaterally apply stimulation near one superficial foramen of the trigeminal nerves. In still other embodiments, the electrodes may be implanted over a plurality of superficial foramina in the face to simultaneously or asynchronously stimulate different trigeminal nerves. In other embodiments, the stimulation may take place in the cutaneous territories of branches of the trigeminal nerves, without attachment to the nerves.
[0045] As can be understood from FIGS. 2A-2B, and with reference to FIGS. 3-4, in one embodiment, a system 10 for the treatment of neurological disorders and conditions via subcutaneous TNS includes an implantable or subcutaneous electrode assembly 20, a pulse generator 30 and electrical cable or wire 40 which may be placed under the skin.
[0046] The pulse generator may be any of a variety of appropriate stimulating, signal generating devices. In some embodiments, the pulse generator 30 may include electronic circuitry for receiving data and/or power from outside the body by inductive, radio-frequency (RF), or other electromagnetic coupling. In some embodiments, electronic circuitry includes an inductive coil for receiving and transmitting RF data and/or power, an integrated circuit (IC) chip for decoding and storing stimulation parameters and generating stimulation pulses, and additional discrete electronic components required to complete the electronic circuit functions, e.g. capacitor(s), resistor(s), transistor(s), coil(s), and the like.
[0047] In other embodiments, pulse generator 30 may include a programmable memory for storing a set(s) of data, stimulation, and control parameters. Among other things, memory may allow stimulation and control parameters to be adjusted to settings that are safe and efficacious with minimal discomfort for each individual. Specific parameters may provide therapeutic advantages for various neurological disorders. For instance, some patients may respond favorably to intermittent stimulation, while others may require continuous stimulation to treat their symptoms.
[0048] In some embodiments, the implantable pulse generator 30 may include a power source and/or power storage device. Possible options for providing power to the system include but are not limited to: an external power source coupled to pulse generator 30, e.g., via an RF link, a self-contained power source utilizing any suitable means of generation or storage of energy (e.g., a primary battery, a replenishable or rechargeable battery such as a lithium ion battery, an electrolytic capacitor, a super-capacitor, a kinetic generator, or the like), and if the self-contained power source is replenishable or rechargeable, means of replenishing or recharging the power source (e.g., an RF link, an optical link, a thermal link, an inductive link, or other energy-coupling link).
[0049] In some embodiments, pulse generator 30 operates independently. In other embodiments, pulse generator 30 operates in coordination with other implanted device(s) or other device(s) external to the patient's body. For example, a pulse generator may communicate with other implanted pulse generators or neurostimulators, other implanted devices, and/or devices external to a patient's body via, e.g., an RF link, an ultrasonic link, a thermal link, an optical link, or the like. Specifically, a pulse generator may communicate with an external remote control (e.g., patient and/or physician programmer) that is capable of sending commands and/or data to a pulse generator and that may also be capable of receiving commands and/or data from a pulse generator.
[0050] In some embodiments, the system may include a regulation device. The regulation device is configured to be attached to the pulse generator 15 and is configured to govern the maximum charge balanced output current below approximately 30-50 mA to minimize current penetration to the brain and increase patient tolerance. The regulation device may be internally programmed to range from 0.25-5.0, 0-10, 0, 15 (all in mA), depending on the surface area, placement, and orientation of the electrode, and whether the electrode is stimulating near or adjacent to the skull, or away from the skull, (e.g. mentalis nerve), where current ranges may be higher or lower. Current TENS units stimulate with maximum output currents of up to 100 mA, which result in currents which may penetrate the skull and which may not be well tolerated.
[0051] In one embodiment, the electrical cable or wire 40 is configured to provide a physical and electrical link between the pulse generator 30 and the electrode assembly 20. In other embodiments, the pulse generator 30 and the electrode assembly 20 communicate wirelessly (i.e. the wire 40 is not used). The system 10 and/or the electrode assembly 20 may be part of a kit. In some embodiments, the kit may also include instructions for treatment of a neurological disorder or condition according to a method disclosed herein.
[0052] In one embodiment, as shown in FIG. 2A, the electrode assembly 20 shown in the illustrated embodiment is also referred to as a bilateral supraorbital electrode. The electrode assembly 20 is connectable to an implanted/implantable pulse generator by electrical cables 40. Alternatively, the electrodes may be connectable to an external pulse generator wirelessly, with transfer of energy across the skin by inductive coupling between a coil implanted in the patient and a coil in the external pulse generator (not shown).
[0053] As shown in FIG. 2B, in one embodiment, the implantable or subcutaneous electrode assembly 20 may include a set of multicontact electrodes 20a, 20b configured for the bilateral simultaneous and asynchronous stimulation of the ophthalmic nerves. The multicontact electrodes 20a, 20b of the electrode assembly 20 comprise an electrode including a first pair of contacts 112a, 112b for implantation at a first region of the patient's face, such as the patient's right forehead or the right side of the patient's face, and an electrode including a second pair of contacts 112c, 112d for implantation at a second region of the patient's face, such as in the patient's left forehead or the left side of the patient's face. In other embodiments, the first and second region of the patient's face may be on the same side of the face but each region may correspond to a different nerve branch, foramina or etc. For example, the first region may correspond to the supraorbital nerve and the second region may correspond to the infraorbital nerve. The electrode assembly 20 may also include an insulated region 116 or a plurality of insulated regions 116 configured to separate the individual electrode contacts. The first pair of contacts comprises a first upper contact 112a and a first lower contact 112b, while the second pair of contacts comprises a second upper contact 112c and a second lower contact 112d. The electrode assembly 20 comprises four electrodes that deliver the stimulation pulses to the nerves bilaterally. While the electrode assembly 20 is shown in FIG. 2B with only pairs of electrical contacts (112a/b, 112c/d), in other embodiments, there may be a greater or lesser number of contacts on each of the electrodes 20a and 20b.
[0054] In some embodiments, as shown in FIG. 3, the electrode assembly 20 may comprise a multicontact electrode 20c with a plurality of contacts 112 and a plurality of insulated regions 116. The electrode assembly of FIG. 3 is configured to unilaterally stimulate both the supraorbital nerve and the infraorbital nerve. In other embodiments, the electrode assembly may comprise a plurality of multicontact electrodes which may include a plurality of contacts and a plurality of insulated regions. In various embodiments, the geometry or layout of the electrode assembly may be a linear electrode with a single contact or a series or plurality of conductive contacts and insulating spaces, or a flatter, "ribbon" or "strip" electrode, also with the possibility of one or more conductive area(s) and insulated area(s) on the surface(s). Those of skill in the art will recognize that other related geometries are also contemplated to be within the scope of the present disclosure.
[0055] FIG. 4 depicts still another embodiment of an electrode assembly 20 that may be used in the system 200. In some embodiments, as shown in FIG. 4, the electrode assembly 20 may comprise a multicontact electrode 20d with a plurality of contacts 112 and a plurality of insulated regions 116. The electrode assembly of FIG. 4 is configured to unilaterally stimulate at least one of the auriculotemporal nerve or the zygomaticofacial nerve. In other embodiments, the electrode assembly 20d may be configured to stimulate both the auriculotemporal nerve and the zygomaticofacial nerve. As can be understood from FIG. 4, in one embodiment, the electrode assembly may be implanted unilaterally. The electrode assembly is configured to be placed at, near or over a superficial foramina in the face and simultaneously or asynchronously stimulate one or more different trigeminal nerves (e.g. the auriculotemporal nerve and/or the zygomaticofacial nerve). In other embodiments, the electrode assembly may be implanted bilaterally to stimulate the target nerve on both sides of a patient's face.
[0056] Those skilled in the art will appreciate that various adaptations and modifications of the above-described embodiments of the electrode assembly 20 are within the scope and spirit of the present disclosure. For example, one embodiment of the present device comprises a unilateral electrode assembly configured for the unilateral stimulation of ophthalmic nerves (see FIG. 3). In other embodiments, the implantable electrode assembly may be configured for the stimulation of the infraorbital nerves or the mentalis nerves. In other embodiments, an electrode assembly may be configured for the simultaneous or asynchronous stimulation of a plurality of elements of the trigeminal nerves, either unilaterally or bilaterally. In other embodiments, both external, transcutaneous electrodes and implanted subcutaneous electrodes are used to simultaneously or asynchronously stimulate one or more branches of the trigeminal nerves. One example of the external, transcutaneous electrode assemblies are described in U.S. patent application Ser. No. 12/898,675, entitled "Systems, Devices and Methods for the Treatment of Neurological Disorders and Conditions," now U.S. Pat. No. 8,688,220, referenced herein above.
[0057] For ease of the reader, the remaining discussion is made with respect to FIG. 2B. However, it is understood that the disclosure also applies to embodiments which include a single multicontact electrode with a plurality of contacts, a single contact electrode, and embodiments which include a plurality of multicontact electrodes with a plurality of contacts and embodiments configured for unilateral or bilateral stimulation and other embodiments within the spirit and scope of the present disclosure.
[0058] As can be understood from FIG. 2B, the electrode assembly 20 is configured to stimulate both the right and left ophthalmic nerves either simultaneously or asynchronously. The placement of the first implanted electrode contact pair 112a, 112b and the second electrode contact pair 112c, 112d on opposite sides of the nasal midline assures that stimulation current moves orthodromically or in the direction of the afferent ophthalmic or supraorbital nerve. Furthermore, this configuration of the electrode assembly 20 allows the electrode contact points 112a/112b and 112c/112d to be stimulated independently and/or unilaterally, as the response to stimulus may be localized and thus varied from one side of the midline to the other side. Depending on the location of the pulse generator, in some embodiments, the electrodes and/or their connectors (e.g. the wires 40) are longer than 150 mm where the supraorbital, infraorbital and/or the mentalis branch is the desired target. For other branches, a shorter electrode/connector length may be desired depending on the placement of the pulse generator.
[0059] For stimulations where electrical pulses of a single polarity are generated, the upper electrode contact points 112a, 112c and lower contact points 112b, 112d have fixed polarities. For stimulations where electrical pulses of alternating polarities are generated, the upper contact points 112a, 112c and lower contact points 112b, 112d have alternating polarities.
[0060] Each of the contacts 112a, 112b, 112c, and 112d .mu.s configured to deliver an electrical pulse with minimal risk of scalp tissue injury due to excess charge accumulation, and with minimal potential for current penetration beyond the inner surface of the skull bone. The distance between the first implanted electrode pair 112a, 112b and the second electrode pair 112c, 112d .mu.s configured to stimulate the ophthalmic nerves while minimizing any current delivery to the surface of the brain. The electrode size and the inter-electrode distance of electrode placement may vary for children and adults, males and females, depending upon the dimensions of an individual person's anatomy.
[0061] Electrode assembly 20, and in particular the contact points 112a, 112b, 112c, 112d, may be made of a noble or refractory metal or compound, such as titanium, titanium nitride, platinum, iridium, tantalum, niobium, rhenium, palladium, gold, nichrome, stainless steel, or alloys of any of these, in order to avoid corrosion or electrolysis which could damage the surrounding tissues and the device. Other compounds for implantable electrodes will be apparent to one skilled in the art.
[0062] In various embodiments, the distance between contacts 112a and 112b and the distance between contacts 112c and 112d can be in a range greater than, equal to, and/or less than one or more of 0.1 cm, 0.5 cm, 1 cm, 2 cm, 3 cm, 4 cm, 5 cm, 6 cm, 7 cm, 8 cm, 9 cm, or 10 cm. Those of skill in the art will recognize that one or more of the above distances can be used as a border of a range of distances.
[0063] In some embodiments, sensing electrodes may be included in the electrode assembly to monitor physiological parameters, such as electroencephalographic data, and permit a feedback system that can adaptively adjust the stimulation parameters to optimize therapeutic benefit and safety. In some embodiments, the sensing electrode is one of the stimulating electrodes and is used for sensing during the `off` part of the duty cycle. In some embodiments, the sensing electrode is an additional electrode and is dedicated to sensing only.
[0064] As shown in FIG. 2B, the electrode assembly may comprise two implanted electrodes 20a, 20b which are placed adjacent to the supraorbital foramina of a patient 5, which is located over the orbital ridge approximately 2.1 to 2.6 cm lateral to the nasal midline. The superior ends 13a, 13b of the electrodes 20a, 20b .mu.ndicate the place at which the electrodes 20a, 20b connect to leads (not shown) for conveying the electrical stimuli from the pulse generator (not shown). The pulse generator itself may be placed in a variety of locations under the skin, such as pectorally, on the back, in the tissues of the neck, or under the scalp, and the leads placed under the skin of the patient to connect them. In other embodiments, the pulse generator may be located externally, such as attached to the patient's clothing.
[0065] In some embodiments, such as the embodiment shown in FIGS. 2A-2B, the neurostimulation is provided using an electrical pulse generator at the following exemplary settings: frequency between approximately 20-150 Hz, current between approximately 0.05-20 mA, pulse duration of between approximately 50-250 microseconds, a duty cycle of 10% to 50%, for at least one hour per day. For optimal patient comfort and low power consumption, stimulation parameters at the lower end of these ranges may be preferred. In other embodiments, different values of the operational parameters may be used as described in more detail below. In alternative embodiments, a single implanted electrode may be used.
[0066] As can be understood from FIG. 3, in one embodiment, the electrode assembly may be implanted unilaterally. The electrode assembly may also be configured to stimulate more than one nerve. For example, as shown in FIG. 3, the electrode assembly is configured to be placed at, near or over a plurality of superficial foramina in the face and simultaneously or asynchronously stimulate different trigeminal nerves (e.g. the supraorbital nerve and the infraorbital nerve).
[0067] As can be best understood from FIGS. 2A-2B, in one embodiment, the electrode assembly 20 is implanted in the soft tissues of the forehead of the patient 5. The electrode assembly 20 is then connected to an implanted pulse generator 30 via the implanted electrical cables 40, which are placed under or in the patient's skin. In the illustrated embodiment, the stimulation via the pulse generator 30 is via electrical cables 40. In alternative embodiments, the electrical stimulation can be performed wirelessly, with an external, non-implanted pulse generator, which uses inductive coupling to deliver energy to the implanted electrode assembly 20. In other embodiments, the electrode assembly may be connected to an external pulse generator via wires or wirelessly.
[0068] The stimulation is carried out at the operational parameters as described herein. In some embodiments, the values of the operational parameters are within a range that produces minimal current penetration into the brain and may further be selected such that a patient will experience a stimulation sensation, such as mild tingling over the forehead, scalp, or teeth, without causing the patient significant discomfort or pain. These values may vary according to the treatment of interest.
[0069] According to one aspect of the present disclosure, there is provided a method of treatment of neurological disorders using the electrode assembly 20, as described above. In one embodiment, the method of treating neurological disorders comprises implanting the electrode assembly 20 subcutaneously (e.g. in the forehead of a patient), connecting the electrode assembly 20 to an implanted pulse generator 30, and stimulating the electrode assembly 20 at defined values of the operational parameters. In one embodiment, the bilateral supraorbital electrode 20 illustrated in FIGS. 2A-2B is stimulated at a stimulus frequency between about 20 and about 300 Hz, at a pulse-duration between 50 microseconds (.mu.sec) to 250 .mu.sec, at an output current of less than 10 mA at the cerebral cortex for at least one-half to one hour per day. In some cases, stimulation can be provided for less than one-half hour per day or may be provided for up to 24 hours per day.
[0070] Accepted standards of safe stimulation may be incorporated for chronic stimulation. Parameters may be selected or calculated to deliver no stimulation or negligible stimulation to the surface of the brain. The currently accepted safe parameters for chronic stimulation are less than a charge per phase of <20 .mu.C/cm.sup.2/phase at the surface of the brain (Exp Neurol 1983; 79:397-41). In general, for any region of the surface of the brain, the cumulative charge per phase resulting from all the electrode contacts should not exceed this threshold. It is recognized that these guidelines are subject to change, and that parameters should be selected which deliver no current or negligible current to the surface of the brain, while still being sufficient to stimulate the nerves disclosed herein.
[0071] According to one aspect of the present disclosure, the method of treating neurological disorders by TNS comprises selecting optimal values for the operational parameters for the stimulation of each individual patient. In one embodiment, the values of the operational parameters are selected such that a patient will experience a stimulation sensation, such as a mild tingling over the forehead, scalp, or face, without being in discomfort or in pain. In some embodiments, lower currents (e.g. 0.05-5 mA) and careful electrode placement may be selected to avoid recruitment of nerves supplying pain sensation to the teeth. In some embodiments, lower currents (e.g. 0.05-5 mA) may also be selected to avoid penetration of the current into the skull and brain, especially in supraorbital locations.
[0072] In one embodiment, the method of selecting operational parameters comprises evaluating variables such as the pulse duration, the electrode current, the duty cycle and the stimulation frequency; the parameters are selected to ensure that the total charge, the charge density, and charge per phase are well within accepted safety limits for the scalp or facial tissue, nerve and brain while preventing or minimizing current penetration beneath the bone tissue of the skull. Additionally, in some embodiments, selection of the electrical stimulation parameters, electrode design, and inter-electrode distance is made such that the electrical stimulation zone includes the superficial elements of the trigeminal nerves (approximately 3-4 mm deep), while preventing or minimizing current penetration beneath the bone tissue of the skull.
[0073] In various embodiments, the stimulation parameters delivered by the implanted pulse generator may be determined (programmed) at the time the device is surgically implanted. In other embodiments, these parameters may be modified, controlled, or otherwise programmed by an external device. This external programming element communicates with the implanted components wirelessly. This may take place, for example, by radiofrequency signals, by inductive coupling, or other means apparent to one skilled in the art.
[0074] In various embodiments, the stimulation is delivered at a specific pulse width or range of pulse widths. The stimulation can be set to deliver pulse widths in the range greater than, equal to, and/or less than one or more of 50 .mu.s, 60 .mu.s, 70 .mu.s, 80 .mu.s, 90 .mu.s, 100 .mu.s, 125 .mu.s, 150 .mu.s, 175 .mu.s, 200 .mu.s, 225 .mu.s, 250 .mu.s, up to 500 .mu.s. Those of skill in the art will recognized that one or more of the above times can be used as a border of a range of pulse widths.
[0075] In some embodiments, the stimulation amplitude is delivered as a voltage or current controlled stimulation. In other embodiments it can be delivered as a capacitive discharge. In various embodiments, the current amplitude can be in any range within a lower limit of about 300 .mu.A and an upper limit of about 30 mA-35 mA, depending on the surface area of the electrodes, inter-electrode distance, the branch(es) stimulated, and the modeling data as described above. In some embodiments, the current used will range from 0.1 mA to 10 mA. In other embodiments, the current used will range from 0.1-3 mA. In various embodiments, the amplitude can be in a range greater than, equal to, and/or less than one or more of 50 .mu.A, 75 .mu.A, 100 .mu.A, 125 .mu.A, 150 .mu.A, 175 .mu.A, 200 .mu.A, 225 .mu.A, 250 .mu.A, 275 .mu.A, 300 .mu.A, 325 .mu.A, 350 .mu.A, 375 .mu.A, 400 .mu.A, 425 .mu.A, 450 .mu.A, 475 .mu.A, 500 .mu.A, 525 .mu.A, 550 .mu.A, 575 .mu.A, 600 .mu.A, 625 .mu.A, 650 .mu.A, 675 .mu.A, 700 .mu.A, 725 .mu.A, 850 .mu.A, 875 .mu.A, 900 .mu.A, 925 .mu.A, 950 .mu.A, 975 .mu.A, 1 mA, 2 mA, 3 mA, 4 mA, 5 mA, 6 mA, 7 mA, 8 mA, 9 mA, 10 mA, 20 mA. Those of skill in the art will recognize that one or more of the above amplitudes can be used as a border of a range of amplitudes. The current may be delivered constantly or intermittently.
[0076] In some embodiments, treatment at a given current amplitude is delivered so as to minimize or eliminate any spread of current to the cerebral cortex, while ensuring that accepted limits of charge density and charge per phase at the brain surface (e.g., generally <20 .mu.C/cm.sup.2/phase, Exp Neurol 1983; 79:397-411) are adhered to, for the safety of the patient. Without wishing to be bound by any particular theory, it is believed that with the use of multicontact electrodes as described herein, even lower charge densities may be employed because more fibers within the nerves may be engaged in the neurostimulation process.
[0077] In various embodiments, the stimulation can be delivered at one or more frequencies, or within a range of frequencies. The stimulation can be set to be delivered at frequencies less than, equal to, and/or greater than one or more of 50 Hz, 45 Hz, 40 Hz, 35 Hz, 30 Hz, 25 Hz, 20 Hz, 15 Hz, or 10 Hz. In various embodiments, the stimulation can be set to be delivered at frequencies greater than, equal to, and/or less than, one or more of 20 Hz, 30 Hz, 40 Hz, 50 Hz, 60 Hz, 70 Hz, 80 Hz, 90 Hz, 100 Hz, 125 Hz, 150 Hz, up to 300 Hz. Those of skill in the art will recognize that one or more of the above frequencies can be used as a border of a range of frequencies.
[0078] In various embodiments, the stimulation is delivered at a specific duty cycle or range of duty cycles. The stimulation can be set to be delivered at a duty cycle in the range greater than, equal to, and/or less than one or more of 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 100%. In some embodiments, to ensure preservation of the nerve, a duty cycle of 10% to 50% may be preferable. In some embodiments, duty cycles up to 100% may be useful in particular circumstances. Those of skill in the art will recognize that one or more of the above percentages can be used as a border of a range of duty cycles.
[0079] In some embodiments, an external device may be used to identify the location of the branch or branches of the trigeminal nerve that will be targeted in an individual patient for stimulation by the implanted electrode assembly disclosed herein. The external device may be used for mapping and targeting the desired branch or branches of the trigeminal nerve and for identifying the individual stimulation parameters that are optimal for efficacy and safety. In one embodiment, the device may include a plurality of external (transcutaneous) TNS electrodes. The practitioner approximates the location of the target branch and affixes the electrodes to the patient's skin above the target location. Stimulation may be applied and the actual location or preferred (optimal) stimulation location of the target branch or branches may be determined. Stimulation parameters may also be established. Once the location and/or stimulation parameters have been established via the external device, that data may be used to help guide the placement of the implanted electrodes for an individual patient and to establish the customized stimulation parameters for that patient.
[0080] In addition, the use of external electrodes for stimulation of the trigeminal nerve may identify individuals who are likely to derive therapeutic benefit from this minimally invasive system in addition to the optimal specific locations and parameters of stimulation based on person-to-person variability. Various neurodiagnostic, imaging, or cutaneous nerve mapping methods may be able to delineate differences in individual anatomy to optimize stimulation for efficacy and/or safety. Furthermore, the use of this minimally invasive system may allow screening and identification of those individuals who are likely to derive benefit from other implantable systems, such as deep brain stimulation. This can be conceptualized as linking the three approaches as stage I (external TNS of the trigeminal nerve), stage II (implanted TNS of the superficial trigeminal nerve), and stage III (deep brain stimulation), such that stage I can screen for stage II, and stage II for stage III. By monitoring a patient for evidence of useful therapeutic effect, such as by reduction in the severity of symptoms, the results of treatment at one stage may be used to judge the likely effect of treatment with a more invasive treatment from a higher stage.
[0081] A method of evaluating the use of trigeminal nerve stimulation for treatment of a neurological disorder in a patient is disclosed herein. The method may include applying a transcutaneous system for stimulation of the trigeminal nerve to the patient and monitoring the patient for at least one of evidence of a useful therapeutic response or evidence of tolerability of TNS treatment, providing a subcutaneous electrode assembly or system as disclosed herein, and implanting the subcutaneous electrode assembly or system as disclosed herein in the patient for treatment of a neurological disorder.
[0082] A method of evaluating the use of deep brain stimulation for treatment of a neurological disorder in a patient is disclosed herein. The method may include applying a transcutaneous system for stimulation of the trigeminal nerve to the patient and monitoring the patient for at least one of evidence of a useful therapeutic response or evidence of tolerability of TNS treatment thereby generating external measurement criteria, providing a subcutaneous electrode assembly or system as disclosed herein, implanting the subcutaneous electrode assembly or system as disclosed herein in the patient for treatment of a neurological disorder, monitoring the patient for at least one of a useful therapeutic response or tolerability of the implanted device, thereby generating extracranial measurement criteria, and analyzing the external measurement criteria and extracranial measurement criteria to determine whether the patient will benefit from deep brain stimulation.
[0083] The following examples are presented to set forth more clearly the subject matter of this disclosure without imposing any limits on the scope thereof and to illustrate the clinical benefits of trigeminal nerve stimulation. In Example 1, patients with epilepsy were treated by TNS with external transcutaneous electrodes. In the second example, a patient was treated using transcutaneous electrodes for bilateral supraorbital stimulation.
Example 1
[0084] FIG. 5 illustrates the results from a pilot study of external trigeminal nerve stimulation. Research subjects with epilepsy who met inclusion and exclusion criteria for a pilot feasibility study of external trigeminal nerve stimulation were enrolled in this study. Subjects initially participated in a 1-month baseline period where seizures were counted, followed by active stimulation of the infraorbital or ophthalmic branch of the trigeminal nerve. Inclusion criteria were: subjects with poorly controlled epilepsy; ages 18-65 years; at least three complex-partial or generalized tonic-clonic seizures per month; no serious or progressive medical or psychiatric conditions; and exposure to at least 2 antiepileptic drugs (AED's). Subjects with a vagus nerve stimulator were excluded from the study. All subjects received unblinded TNS augmentation (adjunctive) treatment for at least 8-12 hours each day. Assessments were made at study intake and at monthly periodic visits for three months following the one-month baseline. These initial assessments were then followed-up with visits to a neurologist skilled in epilepsy for three to six month intervals for up to three years or as approved by the local Institutional Research Committee.
[0085] Subjects underwent stimulation using an electrical stimulator, such as the EMS Model 7500 commercially available from TENS Products, Inc. (Grand Lake, Colo.) at a frequency of 120 Hertz, a current less than 20 mA, pulse duration of 250 .mu.pec, and a duty cycle at 15 to 30 seconds on and 15 to 30 seconds off, for a minimum of 8 hours per day.
[0086] FIG. 5 illustrates the clinical results from this pilot study showing the effectiveness of external trigeminal nerve stimulation. Five of twelve subjects experienced greater than 50% reduction in adjusted mean daily seizure rate at 6 and 12 months of treatment. Mean reduction at 3 months was 66% and was 59% at 12 months. (DeGiorgio et al., Neurology 2009; 72:936-938). Overall, the data from the table of FIG. 5 show that the trigeminal nerve stimulation using the described operational parameter values was effective in reducing seizures and was well tolerated by the subjects tested. No serious adverse events were reported. Importantly, the therapeutic effect of the device was observed in several standard measures, indicating the broad-reaching benefits of this treatment on a variety of outcome measures.
Example 2
[0087] FIG. 6 summarizes current, charge, current density and charge density in a subject exposed to transcutaneous stimulation of the supraorbital nerve. FIG. 6 illustrates representative parameters for bilateral supraorbital stimulation recorded in a subject using an EMS 7500 stimulator, 120 HZ, 150-250 .mu.sec, Tyco superior silver electrodes 1.25'', one inch from the midline above the eyebrows. Data recorded with Fluke Oscilloscope, 50 mV/div, resistor=10.1.OMEGA.. In general, these findings show that as the pulse width increased, the maximum tolerable current decreased.
[0088] Transcutaneous electrical stimulation of the supraorbital branch of the trigeminal nerve with round 1.25-inch TENS patch electrodes results in current densities and charge density/phase that are well within the limits of safety. In general, the maximum current comfortably tolerated by TNS patients studied previously is approximately 25 mA's, and patients typically are stimulated at an amplitude setting well below 25 mA's (6-10 mA's).
[0089] The 1.25-inch TENS electrodes are circular electrodes with a radius of 1.59 cm. The surface area can be calculated as A=.pi..sup.2=[.pi.].times.[1.59 cm].sup.2=7.92 cm.sup.2. Using these electrodes, typical stimulation current ranges from 6-10 mA at pulse durations of 150-250 .mu.sec.
[0090] Current Density: In a typical subject, stimulation currents of 6-10 mA result in current densities ranging from 0.76 to 1.3 mA/cm.sup.2. McCreery et al have established a maximum safe current density of 25 mA/cm at the stimulating electrode for transcranial electrical stimulation. Assuming even higher currents of up to 25 mA with electrodes of surface area 7.92 cm.sup.2, current densities may range to a maximum of 3.16 mA/cm.sup.2. From 0.76 mA/cm.sup.2 to 3.16 mA/cm.sup.2, TNS delivers a current density 8-33 times less than the maximum safe allowable current density. Charge Density (Charge density/phase): Yuen et al. have identified a safe limit for charge density/phase delivered at the cerebral cortex of 40 .mu.C/cm.sup.2 [Yuen et al., 1981] and more recently McCreery et al. (McCreery et al., 1990) have identified 10 .mu.C/cm.sup.2 as the safe limit. Assuming 10 mA at 250 .mu.sec, the charge density/phase is [0.010 A].times.[250 .mu.sec]/7.92=0.32 .mu.C/cm.sup.2 at the stimulating electrode. Assuming even higher levels of stimulation, 25 mA at 250 .mu.sec, the maximum charge density per phase is 0.79 .mu.C/cm.sup.2. At these levels, the charge density is generally 12 to 120 fold less at the stimulating electrode than the maximum allowed at the cerebral cortex. Since the cortex is a minimum of 10-13 mm from the stimulating electrodes, and given the interposed layers of skin, fat, bone, dura, and CSF, the actual charge densities will be significantly lower. This is of importance in avoiding the undesired passage of current directly through brain tissue as a bulk conductor.
[0091] As shown in FIG. 6, stimulation intensity responses in a subject with electrodes of surface area 7.92 cm2, at pulse durations between 150-250 .mu.sec, results in current densities at the scalp well below currently recommended current densities for transcranial stimulation, which are 25 mA/cm.sup.2, and charge densities at the scalp significantly lower than safe charge densities at the cerebral cortex (0.15-0.18 .mu.C/cm.sup.2).
[0092] Those skilled in the art will appreciate that various adaptations and modifications of the above described preferred embodiments may be configured without departing from the scope and spirit of this disclosure. Stimulation of the target nerve may be accomplished by application of energy in many forms, such as magnetic or ultrasonic. Therefore, it is to be understood that the subject matter of this disclosure may be practiced other than as specifically described herein.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.