Peptidomimetic Macrocycles And Uses Thereof
Chen; Hubert ; et al.
U.S. patent application number 16/354873 was filed with the patent office on 2019-09-05 for peptidomimetic macrocycles and uses thereof. The applicant listed for this patent is AILERON THERAPEUTICS, INC.. Invention is credited to Manuel Aivado, David Allen Annis, Yong Chang, Hubert Chen, Karen Olson, Chris Viau.
| Application Number | 20190269753 16/354873 |
| Document ID | / |
| Family ID | 55582021 |
| Filed Date | 2019-09-05 |










View All Diagrams
| United States Patent Application | 20190269753 |
| Kind Code | A1 |
| Chen; Hubert ; et al. | September 5, 2019 |
PEPTIDOMIMETIC MACROCYCLES AND USES THEREOF
Abstract
Methods for treating solid tumor, determined to lack a p53 deactivation mutation, in a subject are provided. Also provided are peptidomimetic macrocycles for use in treatment of a solid tumor, determined to lack a p53 deactivation mutation, in a subject.
| Inventors: | Chen; Hubert; (San Diego, CA) ; Annis; David Allen; (Cambridge, MA) ; Chang; Yong; (Acton, MA) ; Aivado; Manuel; (Chester Springs, PA) ; Olson; Karen; (Waltham, MA) ; Viau; Chris; (Mashpee, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 55582021 | ||||||||||
| Appl. No.: | 16/354873 | ||||||||||
| Filed: | March 15, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14864801 | Sep 24, 2015 | |||
| 16354873 | ||||
| 62054861 | Sep 24, 2014 | |||
| 62213831 | Sep 3, 2015 | |||
| 62216670 | Sep 10, 2015 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 19/08 20180101; C12Q 1/6886 20130101; A61P 17/00 20180101; A61P 35/00 20180101; A61P 21/00 20180101; C12Q 2600/156 20130101; A61P 1/16 20180101; A61P 25/00 20180101; A61K 9/0019 20130101; A61K 38/12 20130101; A61P 15/00 20180101; C12Q 2600/158 20130101 |
| International Class: | A61K 38/12 20060101 A61K038/12; C12Q 1/6886 20060101 C12Q001/6886; A61K 9/00 20060101 A61K009/00 |
Claims
1. A method of treating a cancer in a subject in need thereof, the method comprising: administering to the subject a therapeutically-effective amount of a p53 activator, and a therapeutically-effective amount of another anticancer agent; wherein the p53 activator reduces a side effect associated with the other anticancer agent.
2. The method of claim 1, wherein the therapeutically-effective amount of the p53 activator and the therapeutically-effective amount of the other anticancer agent synergize.
3. The method of claim 1, wherein the therapeutically-effective amount of the other anticancer agent is lower than is a therapeutically-effective amount of the other anticancer agent in absence of the p53 activator.
4. The method of claim 1, wherein the side effect is toxicity.
5. The method of claim 1, wherein the p53 activator and the other anticancer agent are administered in a common pharmaceutical composition.
6. The method of claim 1, wherein the p53 activator and the other anticancer agent are administered in separate pharmaceutical compositions.
7. The method of claim 1, wherein the p53 activator and the other anticancer agent are administered concurrently.
8. The method of claim 1, wherein the p53 activator and the other anticancer agent are administered sequentially.
9. The method of claim 1, wherein the other anticancer agent is a hormonal agent, aromatase inhibitor, selective estrogen receptor modulator (SERM), estrogen receptor antagonist, chemotherapeutic agent, microtubule disassembly blocker, antimetabolite, topoisomerase inhibitor, DNA crosslinker or damaging agent, anti-antigenic agent, VEGF antagonist, receptor antagonist, integrin antagonist, vascular targeting agent (VTA), or vascular disrupting agent (VDA).
10. The method of claim 1, wherein the other anticancer agent is a class I (camptotheca) topoisomerase inhibitor, topotecan, irinotecan, rubitecan, belotecan, a class II (podophyllum) topoisomerase inhibitor, etoposide, VP-16, teniposide, anthracyclines, doxorubicin, epirubicin, Doxil, aclarubicin, amrubicin, daunorubicin, idarubicin, pirarubicin, valrubicin, and zorubicin, anthracenediones, mitoxantrone, or pixantrone.
11. The method of claim 1, wherein the side effect is related to a hematopoietic organ tissue.
12. The method of claim 1, wherein the side effect is hematologic.
13. The method of claim 1, wherein the side effect is thrombocytopenia, neutropenia, or febrile neutropenia.
14. The method of claim 1, wherein the p53 activator binds to MDM2 and/or MDMX.
15. The method of claim 1, wherein the p53 activator disrupts an interaction between p53 and MDM2 and/or MDMX.
16. The method of claim 1, wherein the p53 activator is administered in a dosage of about 0.5-20 mg per kilogram body weight of the subject.
17. The method of claim 1, wherein the therapeutically-effective amount of the p53 activator is about 0.04 mg, 0.08 mg, 0.16 mg, 0.32 mg, 0.64 mg, 1.28 mg, 3.56 mg, 7.12 mg, or 14.24 mg of the p53 activator per kilogram body mass of the subject.
18. The method of claim 1 wherein the administering is over a period of 0.25-2.0 h.
19. The method of claim 1, wherein the p53 activator comprises an amino acid sequence which is at least about 60% identical to an amino acid sequence in any of Table 3, Table 3a, Table 3b, and Table 3c, wherein the peptidomimetic macrocycle has the formula: ##STR00076## wherein: each A, C, D and E is independently an amino acid; each B is independently an amino acid, ##STR00077## [--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each R.sub.3 independently is hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; each L and L' is independently a macrocycle-forming linker of the formula -L.sub.1-L.sub.2-; each L.sub.1, L.sub.2, and L.sub.3 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.E, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v is independently an integer; each w is independently an integer from 3-1000; u is an integer from 1-10; each x, y and z is independently an integer from 0-10; and each n is independently an integer from 1-5.
20. The method of claim 1, wherein the p53 activator induces cell cycle arrest in the subject.
21. The method of claim 1, wherein the cancer is non-small cell lung cancer.
Description
CROSS REFERENCE
[0001] This application is a Continuation Application of U.S. application Ser. No. 14/864,801, filed Sep. 24, 2015, which claims the benefit of U.S. Provisional Application No. 62/054,861, filed Sep. 24, 2014, U.S. Provisional Application No. 62/213,831, filed Sep. 3, 2015 and U.S. Provisional Application No. 62/216,670, filed Sep. 10, 2015, each of which is incorporated herein by reference in its entirety.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Dec. 22, 2015, is named 35224-804-302_SL.txt and is 1,199,023 bytes in size.
BACKGROUND OF THE INVENTION
[0003] The human transcription factor protein p53 induces cell cycle arrest and apoptosis in response to DNA damage and cellular stress, and thereby plays a critical role in protecting cells from malignant transformation. The E3 ubiquitin ligase MDM2 (also known as HDM2 or human double minute 2) negatively regulates p53 function through a direct binding interaction that neutralizes the p53 transactivation activity, leads to export from the nucleus of p53 protein, and targets p53 for degradation via the ubiquitylation-proteasomal pathway. Loss of p53 activity, either by deletion, mutation, or MDM2 overexpression, is the most common defect in human cancers. Tumors that express wild type p53 are vulnerable to pharmacologic agents that stabilize or increase the concentration of active p53. In this context, inhibition of the activities of MDM2 has emerged as a validated approach to restore p53 activity and resensitize cancer cells to apoptosis in vitro and in vivo. MDMX (also known as MDM4, HDM4 or human double minute 4) has more recently been identified as a similar negative regulator of p53, and studies have revealed significant structural homology between the p53 binding interfaces of MDM2 and MDMX.MDMX has also been observed to be overexpressed in human tumors. The p53-MDM2 and p53-MDMX protein-protein interactions are mediated by the same 15-residue alpha-helical transactivation domain of p53, which inserts into hydrophobic clefts on the surface of MDM2 and MDMX. Three residues within this domain of WT p53 (F19, W23, and L26) are essential for binding to MDM2 and MDMX.
[0004] There remains a considerable need for methods for treating solid tumor. Provided herein are compounds capable of binding to and modulating the activity of p53, MDM2 and/or MDMX. Also provided herein are pharmaceutical formulations comprising p53-based peptidomimetic macrocycles that modulate an activity of p53. Also provided herein are pharmaceutical formulations comprising p53-based peptidomimetic macrocycles that inhibit the interactions between p53, MDM2 and/or MDMX proteins.
[0005] Further, provided herein are methods for treating diseases including but not limited to solid tumors and other hyperproliferative diseases.
SUMMARY OF THE INVENTION
[0006] In one aspect, the disclosure provides a method of treating solid tumor determined to lack a p53 deactivating mutation, in a human subject, wherein the method comprises administering to the human subject a therapeutically effective amount of a peptidomimetic macrocycle or a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycle disrupts the interaction between p53 and MDM2 and MDMX.
[0007] In another aspect, the disclosure provides a method of treating a solid tumor that lacks a p53 deactivating mutation, in a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0008] In another aspect, the disclosure provides a method of treating a solid tumor that has a p53 deactivating mutation in a p53 gene, in a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0009] In another aspect, the disclosure provides a method of treating a solid tumor in a human subject in need thereof, wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein the solid tumor is not negative for p53 protein expression (such as solid tumors that express wild-type p53 protein or mutated p53 protein with partial functionality).
[0010] In another aspect, the disclosure provides a method of treating a solid tumor in a human subject in need thereof, wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein the solid tumor expresses a p53 protein with a gain of function mutation (such as a super apoptotic p53).
[0011] In another aspect, the disclosure provides a method of treating a solid tumor in a human subject in need thereof, wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein the solid tumor express a p53 protein with a mutation that causes a partial loss of function.
[0012] In another aspect, the disclosure provides a method of treating a solid tumor a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein cells in the solid tumor express p53 from only a single genomic copy of the p53 gene (for example where the cells have a copy loss mutation, e.g., are haploinsufficient).
[0013] In another aspect, the disclosure provides a method of treating a solid tumor a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein the solid tumor express a p53 protein with one or more silent mutations.
[0014] In another aspect, the disclosure provides a method of treating a solid tumor a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein cells in the solid tumor are negative for p53 expression.
[0015] In another aspect, the disclosure provides a method of treating a solid tumor that has a p53 deactivating mutation in a p53 gene, in a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins and wherein cells in the solid tumor have the p53 deactivating mutation in one copy of the p53 gene. In some embodiments, the cells in the solid tumor have a second p53 deactivating mutation in a second copy of a p53 gene. In some embodiments, the p53 deactivating mutation in one copy of the p53 gene is the same as the second p53 deactivating mutation in the second copy of a p53 gene. In some embodiments, the p53 deactivating mutation in one copy of the p53 gene is different from the second p53 deactivating mutation in the second copy of a p53 gene.
[0016] In some embodiments, the p53 deactivating mutation in the p53 gene results in the lack of p53 protein expression from the p53 gene or in expression of partial a p53 protein with partial loss of function. In some embodiments, the second p53 deactivating mutation in the second copy of a p53 gene results in the lack of p53 protein expression from the p53 gene or in expression of partial a p53 protein with partial loss of function.
[0017] In some embodiments of the methods described herein, the cells of the solid tumor have at least one mutation in a copy of a p53 gene, wherein the mutation eliminates or reduces the or activity of a p53 protein expressed from the copy of the p53 gene, as compared to wild type p53 expressed from a copy of a non-mutated p53 gene.
[0018] In another aspect, the disclosure provides a method of treating a solid tumor in a human subject in need thereof wherein the method comprises administering to the human subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0019] In some embodiments, the peptidomimetic macrocycle used in the various methods described herein is a peptidomimetic macrocycle that disrupts the interaction between p53 and MDM2 and MDMX.
[0020] In some embodiments, the various methods described herein further comprise determining the lack of the p53 deactivating mutation in the solid tumor prior to the administration of the pharmaceutical composition. In some embodiments, the determining the lack of the p53 deactivating mutation comprises confirming the presence of wild type p53 in the solid tumor.
[0021] In some embodiments, the various methods described herein further comprise determining a presence of a p53 gain of function mutation in the solid tumor.
[0022] In some embodiments, the various methods described herein further comprise determining a presence of a deactivating mutation of p53 in the solid tumor.
[0023] In some embodiments, the various methods described herein further comprise determining a presence of a copy loss mutation of p53 in the solid tumor.
[0024] In some embodiments, the various methods described herein further comprise determining a presence of a partial loss of function mutation of P53 in the solid tumor.
[0025] In some embodiments, the methods described herein can further comprise confirming the lack of the p53 deactivating mutation in the solid tumor, prior to the administration of the peptidomimetic macrocycle. For example, confirming the presence of wild type p53 in the solid tumor.
[0026] In some embodiments, the methods described herein can further comprise confirming a presence of a p53 gain of function mutation in the solid tumor.
[0027] In some embodiments, the methods described herein can further comprise confirming a presence of a deactivating mutation of p53 in the solid tumor.
[0028] In some embodiments, the methods described herein can further comprise confirming a presence of a copy loss mutation of p53 in the solid tumor.
[0029] In some embodiments, the methods described herein can further comprise confirming a presence of a partial loss of function mutation of P53 in the solid tumor.
[0030] In various embodiments, the determining or confirming is performed within 3 years, 2 years, within 1 year, within 1-12 months, within 1-3 months, within 1 month, or within 21 days prior to the administration of the peptidomimetic macrocycle.
[0031] In various embodiments, the treatment methods provided herein can result in re-activation of the p53 pathway, decreased tumor cell proliferation, increased p53 protein, increased p21, and/or increased apoptosis in the human subject.
[0032] The peptidomimetic macrocycle can be administered two or three times a week, for example two times a week. In some examples, the peptidomimetic macrocycle is administered once every 2 or 3 weeks. In other examples the peptidomimetic macrocycle is administered once every 1 or 2 weeks. In some embodiments, the peptidomimetic macrocycle is administered on days 1, 8 and 15 of a 28-day cycle. In other examples the peptidomimetic macrocycle is administered once every week. In some examples, a dose of the pharmaceutical composition is administered on days 1, 4, 8 and 11 of a 21-day cycle.
[0033] The amount of the peptidomimetic macrocycle administered is about 0.5-20 mg per kilogram body weight of the human subject, for example 0.5-10 mg per kilogram body weight of the human subject. In some embodiments, the amount of the peptidomimetic macrocycle administered is about 0.04 mg, 0.08 mg, 0.16 mg, 0.32 mg, 0.64 mg, 1.28 mg, 3.56 mg, 7.12 mg, or 14.24 mg per kilogram body weight of the human subject. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered two times a week. In other examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered two times a week. In other examples, 0.32 mg, 0.64 mg, 1.25 mg 2.5 or 5.0 mg per kilogram body weight of the human subject and the pharmaceutical composition is administered two times a week. In some examples, about 0.32 mg, 0.64 mg, 1.25 mg 2.5 or 5.0 mg per kilogram body weight of the human subject and the pharmaceutical composition is administered on day 1, 4, 8, 11 of a 21-day cycle. In some examples, about 0.16 mg, 0.32 mg, 0.64 mg, 1.25 mg, 2.5, 5.0 mg or 10 mg per kilogram body weight of the human subject and the pharmaceutical composition is administered on day 1, 8, and 15 of a 28 day cycle.
[0034] In other examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg, 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once a week.
[0035] In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once daily, three, five or seven times in a seven day period. For example, the peptidomimetic macrocycle is administered once daily intravenously, seven times in a seven day period.
[0036] In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg, or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once daily three, five or seven times in a seven day period. For example, the peptidomimetic macrocycle is administered once daily intravenously, seven times in a seven day period.
[0037] The peptidomimetic macrocycle can be administered gradually over a period of 0.25-12 h, for example over a period of 0.25 h, 0.5 h, 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h, 11 h, or 12 h. In some examples, the peptidomimetic macrocycle is administered over a period of 0.25-2.0 h. In some embodiments, the peptidomimetic macrocycle is gradually administered over a period of 1 h. In other embodiments, the peptidomimetic macrocycle is gradually administered over a period of 2 h.
[0038] The method provided herein can result in reduction of tumor volume. For example, treatment according to methods provided herein can result in about 95%, 90%, 85%, 80%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, or 5% reduction in tumor volume within a period of 1 month after treatment initiation. In some example, the treatment results in at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% reduction in tumor volume within a period of 1 month after treatment initiation. In some examples, the treatment results in about 95%, 90%, 85%, 80%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, or 5% reduction in tumor volume within a period of 1 year after treatment initiation. In some embodiments, the treatment results in at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% reduction in tumor volume within a period of 1 year after treatment initiation. In some examples, the treatment results in about 95%, 90%, 85%, 80%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, or 5% reduction in tumor volume within a period of 6 months after treatment initiation. In some examples, the treatment results in at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% reduction in tumor volume within a period of 6 months after treatment initiation. In some examples, the treatment results in about 95%, 90%, 85%, 80%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, or 5% reduction in tumor volume within a period of 3 months after treatment initiation. In some examples, the treatment results in at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% reduction in tumor volume within a period of 3 months after treatment initiation. In some embodiments, the solid tumor is a stable disease. In some embodiments, the solid tumor is a progressive disease.
[0039] In some embodiments, the methods provided herein can result in an increased survival time of the human subject as compared to the expected survival time of the human subject if the human subject was not treated with the peptidomimetic macrocycle. In some examples, the increase in the survival time of the human subject is at least 30 days, at least 3 months, at least 6 months or at least 1 year.
[0040] The in vivo circulating half-life of the peptidomimetic macrocycle is about 1 h-12 h, for example about 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h or 12 h. In some examples, the in vivo circulating half-life of the peptidomimetic macrocycle is about 4 h, about 6 h.
[0041] The biological tissue half-life of the peptidomimetic macrocycle is about 1 h-12 h, for example about 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h or 12 h. In some examples, the biological tissue half-life of the peptidomimetic macrocycle is about 10 h.
[0042] In some embodiments, the human subject treated in accordance to the methods of the disclosure is refractory and/or intolerant to one or more other treatment of the solid tumor. In some embodiments, the human subject has had at least one unsuccessful prior treatment and/or therapy of the solid tumor.
[0043] In some embodiments, the solid tumor expresses wild-type p53 protein.
[0044] The solid tumor treated by the methods of the disclosure is selected from a group consisting of pancreatic cancer, bladder cancer, colon cancer, liver cancer, colorectal cancer, breast cancer, prostate cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors, bone cancer, skin cancer, ocular tumor, rectal cancer, choriocarcinoma (tumor of the placenta), sarcoma and soft tissue cancer, testicular cancer, gall bladder cancer, and biliary cancer. In some embodiments, the solid tumor is selected from a group consisting of bladder cancer, bone cancer, breast cancer, cervical cancer, CNS cancer, colon cancer, ocular tumor, renal cancer, liver cancer, lung cancer, pancreatic cancer, choriocarcinoma (tumor of the placenta), prostate cancer, sarcoma, skin cancer, soft tissue cancer, gastric cancer, gall bladder cancer, biliary cancer, renal cancer, or neuroendocrine cancer. The ocular tumor can be choroidal nevus, choroidal melanoma, choroidal metastasis, choroidal hemangioma, choroidal osteoma, iris melanoma, uveal melanoma, melanocytoma, metastasis retinal capillary hemangiomas, congenital hypertrophy of the RPE, RPE adenoma or retinoblastoma. In some embodiments, the solid tumor is selected from non-small cell lung cancer, small-cell lung cancer, colon cancer, CNS cancer, melanoma, ovarian cancer, renal cancer, prostate cancer and breast cancer. In some embodiments, the solid tumor is breast cancer. In some embodiments, the solid tumor is gall bladder cancer. In some embodiments, the solid tumor is the solid tumor is biliary cancer. In some embodiments, the solid tumor is neuroendocrine cancer. In some embodiments, the solid tumor is bone cancer. In some embodiments, the solid tumor is osteosarcoma. In some embodiments, the solid tumor is skin cancer. In some embodiments, the solid tumor is melanoma.
[0045] In some examples, the solid tumor treated by the methods of the disclosure is not a HPV positive cancer.
[0046] In some examples, the solid tumor treated by the methods of the disclosure is not HPV positive cervical cancer, HPV positive anal cancer or HPV positive head and neck cancer, such as oropharyngeal cancers.
[0047] In some embodiments, the peptidomimetic macrocycle is administered intravenously.
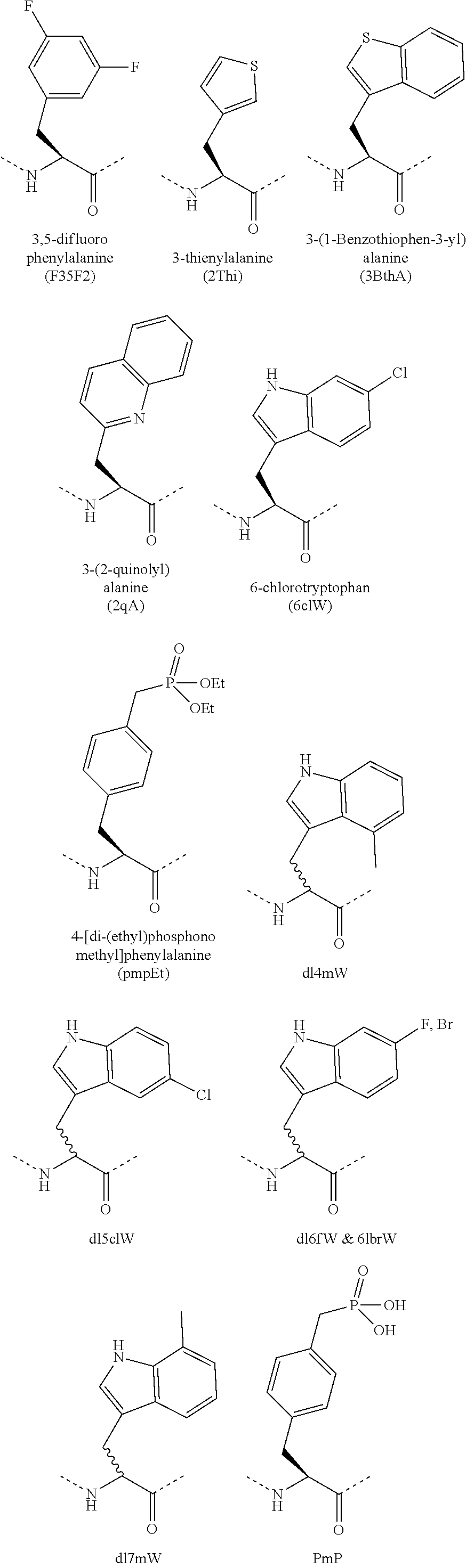
[0048] In some embodiments, the methods of the disclosure further comprise administering in addition to the peptidomimetic macrocycle or pharmaceutically acceptable salt thereof, a therapeutically effective amount of at least one additional therapeutic agent and/or therapeutic procedure to the human subject.
[0049] In some embodiments, the human subject exhibits a complete response to the treatment. In some embodiments, the human subject exhibits a partial response to the treatment.
[0050] In some embodiments, the methods of the disclosure further comprise determining clinical activity of the administered peptidomimetic macrocycle. The clinical activity can be determined by an imaging method selected from a group consisting of computed tomography (CT), magnetic resonance imaging (MRI), and bone scanning.
[0051] The methods of the disclosure can further comprise obtaining a biological sample from the human subject at one or more specific time-points and analyzing the biological sample with an analytical procedure. The biological samples can be used for biomarker assessment, pharmacokinetic assessment, immunogenicity assays and/or pharmacodynamic assessment. The pharmacokinetic assessment can comprise studying the level of the peptidomimetic macrocycle and/or its metabolites in the biological sample at the specific time-points. The pharmacodynamic assessment can comprise studying the level of p53, MDM2, MDMX, p21 and/or caspase in the biological sample at the specific time-points.
[0052] The analytical procedure can be selected from a group comprising blood chemistry analysis, chromosomal translocation analysis, needle biopsy, tissue biopsy, fluorescence in situ hybridization, laboratory biomarker analysis, immunohistochemistry staining method, flow cytometry, or a combination thereof. The methods can further comprise tabulating and/or plotting results of the analytical procedure. The one or more specific time-points can comprise a time-point before the administration of the peptidomimetic macrocycle to the human subject. The one or more specific time-points can comprise a time-point after the administration of the peptidomimetic macrocycle to the human subject. The one or more specific time-points can comprise a time-point before and a time-point after the administration of the peptidomimetic macrocycle to the human subject. The one or more specific time-points comprise multiple time-points before and after the administration of the peptidomimetic macrocycle to the human subject. The method can further comprise comparing the biological samples collected before and after the administration of the peptidomimetic macrocycle to the human subject or comparing the biological samples collected at the multiple time-points. The biological sample can be a blood sample or a tumor specimen.
[0053] The method of the disclosure can further comprise selecting and/or identifying at least one target lesion in the human subject prior to the administration of the peptidomimetic macrocycle to the human subject. The methods can also comprise measuring cumulative diameters at one or more specific time-points, wherein the cumulative diameter is a sum of the diameters of the at least one target lesions at the specific time-point. The one or more specific time-points, can comprise a time-point after the treatment. The methods can also comprise measuring a baseline sum diameter, wherein the baseline sum diameter is a sum of the diameters of the at least one target lesions prior to the administration of the pharmaceutical composition to the human subject. In some examples, the treatment in accordance to the methods of the disclosure results in disappearance of the least one target lesion. In some embodiments, after the treatment all pathological lymph nodes in the human subject exhibit a reduction in short axis to less than 10 mm. In some examples, the cumulative diameter at the time-point after the treatment is at least 30% less than the baseline sum diameter. In some examples, the treatment results in neither a sufficient increase nor a sufficient decrease in the cumulative diameters at the one or more specific time-points, taking as reference the baseline sum diameter.
[0054] In some examples, the peptidomimetic macrocycle is not an inhibitor of cytochrome P450 isoforms. In some examples, the treatment results in essentially no dose-limiting thrombocytopenia. In some examples, the treatment causes essentially no adverse effects in a normal-hematopoietic organ and/or tissue. In some examples, the treatment results in essentially no adverse event in the human subject that can be possibly, probably, or definitely related to the administration of the peptidomimetic macrocycle. In some examples, the treatment results in essentially no serious adverse event in the human subject that can be probably, probably, or definitely related to the administration of the peptidomimetic macrocycle.
[0055] The lack of p53 deactivation mutation can be determined by any known method known in the art. In some examples, the lack of p53 deactivation mutation can be determined by DNA sequencing of the nucleic acid encoding the p53 protein. In some examples, the lack of p53 deactivation mutation can be determined by RNA array based testing. In some examples, the lack of p53 deactivation mutation can be determined by RNA analysis. In some examples, the lack of p53 deactivation mutation can be determined by polymerase chain reaction (PCR).
[0056] In some embodiments, the p53 deactivating mutation can comprise mutations in DNA-binding domain of the protein. In some embodiments, the p53 deactivating mutation can comprise missense mutation. In some embodiments, the p53 deactivating mutation is a dominant deactivating mutation. In some embodiments, the p53 deactivating mutation comprises one or more mutations selected from a groups consisting of V173L, R175H, G245C, R248W, R249S and R273H. In some embodiments, the p53 deactivating mutation comprises one or more of mutations shown in Table 1a. In some embodiments, the p53 gain of function mutation comprises one or more of mutations shown in Table 1b.
[0057] In another aspect, the disclosure provides a method of treating solid tumor in a human subject determined to lack a p53 deactivating mutation, wherein the method comprises administering to the human subject 0.5-20 mg, for example 0.5-10 mg of a peptidomimetic macrocycle per kilogram body weight of the human subject or a pharmaceutically acceptable salt thereof on days 1, 8 and 15 of a 28-day cycle. In some embodiments, the amount of the peptidomimetic macrocycle entered on day 8 and/or day 15 is greater than the amount of the peptidomimetic macrocycle entered on day 1. In some embodiments, the peptidomimetic macrocycle entered on day 8 and/or day 15 is equal than the amount of the peptidomimetic macrocycle entered on day 1. In some embodiments, t the peptidomimetic macrocycle entered on day 1 and/or day 8 is greater than the amount of the peptidomimetic macrocycle entered on day 15. In some embodiments, an equal amount of the peptidomimetic macrocycle is administered on days 1, 8 and 15. In some embodiments, the 28-day cycle is repeated 2 or 3 times.
[0058] In another aspect, the disclosure provides a method of treating solid tumor in a human subject, wherein the method comprises administering to the human subject 0.32-10 mg of a peptidomimetic macrocycle per kilogram body weight of the human subject or a pharmaceutically acceptable salt thereof on days 1, 4, 8 and 11 of a 21-day cycle. In some embodiments, the solid tumor is determined to lack a p53 deactivating mutation. In some embodiments, 0.32 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject or the pharmaceutically acceptable salt thereof is administered on days 1, 4, 8 and 11 respectively of a 21-day cycle. In some embodiments, 0.64 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject or the pharmaceutically acceptable salt thereof is administered on days 1, 4, 8 and 11 respectively of a 21-day cycle. In some embodiments, 1.25 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject or the pharmaceutically acceptable salt thereof is administered on days 1, 4, 8 and 11 respectively of a 21-day cycle. In some embodiments, 2.5 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject or the pharmaceutically acceptable salt thereof is administered on days 1, 4, 8 and 11 respectively of a 21-day cycle. In some embodiments, 5.0 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject or the pharmaceutically acceptable salt thereof is administered on days 1, 4, 8 and 11 respectively of a 21-day cycle.
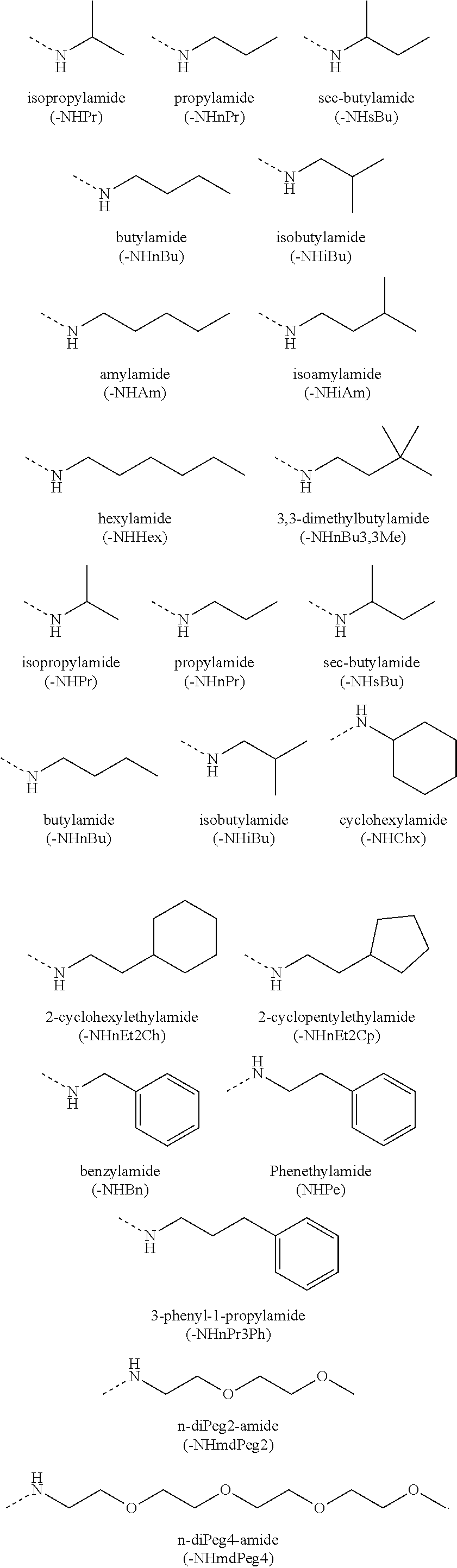
[0059] In various embodiments, the peptidomimetic macrocycle used in the methods described herein comprises an amino acid sequence which is at least about 60%, 70%, 80%, 90%, or 95% identical to an amino acid sequence in any of Table 3, Table 3a, Table 3b, and Table 3c, wherein the peptidomimetic macrocycle has the formula:
##STR00001##
wherein: each A, C, D and E is independently an amino acid; each B is independently an amino acid,
##STR00002##
[--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each R.sub.3 independently is hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; each L and L' is independently a macrocycle-forming linker of the formula -L.sub.1-L.sub.2-; each L.sub.1, L.sub.2, and L.sub.3 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v is independently an integer; each w is independently an integer from 3-1000; u is an integer from 1-10; each x, y and z is independently an integer from 0-10; and each n is independently an integer from 1-5.
[0060] In various embodiments, the peptidomimetic macrocycle used in the methods described herein has formula:
##STR00003##
wherein: each of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 is individually an amino acid, wherein at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8) or Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.12 (SEQ ID NO: 9), where each X.sub.4 and X.sub.11 is independently an amino acid; each D and E is independently an amino acid; each R.sub.1 and R.sub.2 are independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each L or L' is independently a macrocycle-forming linker each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; v is an integer from 1-1000; w is an integer from 0-1000.
[0061] In some embodiments, at least one of the macrocycle-forming linker in the Formulas described herein has a formula -L.sub.1-L.sub.2-, wherein
each L.sub.1 and L.sub.2 are independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.3 is independently hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; and each n is independently an integer from 1-5.
[0062] In some embodiments, at least one of the macrocycle-forming linker in the Formulas described herein each w is independently an integer from 3-1000, for example 3-500, 3-200, 3-100, 3-50, 3-30, 3-20, or 3-10.
[0063] In some embodiments, Xaa5 is Glu or an amino acid analog thereof.
[0064] In some embodiments, each E is independently Ala (alanine), Ser (serine) or an analog thereof.
[0065] In some embodiments, [D]v is -Leu.sub.1-Thr.sub.2.
[0066] In some embodiments, w is 3-10. In some embodiments, w is 3-6. In some embodiments, w is 6-10. In some embodiments, w is 6.
[0067] In some embodiments, v is 1-10. In some embodiments, v is 2-10. In some embodiments, v is 2-5. In some embodiments, v is 2.
[0068] In some embodiments, each L.sub.1, L.sub.2 and L.sub.3 in the Formulas described herein are independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, or heterocycloarylene, each being optionally substituted with R.sub.5.
[0069] In some embodiments, each L.sub.1, L.sub.2 and L.sub.3 are independently alkylene or alkenylene.
[0070] In some embodiments, L is alkylene, alkenylene, or alkynylene. In some embodiments, L is alkylene. In some embodiments, L is C3-C16 alkylene. In some embodiments, L is C10-C14 alkylene.
[0071] In some embodiments, each R.sub.1 and R.sub.2 in the Formulas described herein are independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-. In some embodiments, R.sub.1 and R.sub.2 are H. In some embodiments, each R.sub.1 and R.sub.2 are independently alkyl. In some embodiments, R.sub.1 and R.sub.2 are methyl.
[0072] In some embodiments, x+y+z in the Formulas described herein is 6.
[0073] In some embodiments, u in the Formulas described herein is 1.
[0074] The some embodiments, peptidomimetic macrocycles comprise at least one amino acid which is an amino acid analog. In some embodiments, the peptidomimetic macrocycle is chosen from the peptidomimetic macrocycles shown in Table 3c.
[0075] In one aspect, the disclosure provides a method of identifying one or more solid tumor biomarkers in a human subject lacking a p53 deactivating mutation, comprising administering to the human subject a therapeutically effective amount of a peptidomimetic macrocycle described herein. In some examples, the biomarkers are selected from a group comprising p53 status, MDM2 expression level and MDMX expression level.
[0076] In the various embodiments, the pharmaceutical composition comprises the pharmaceutically acceptable salt of the peptidomimetic macrocycle. In some embodiments, the pharmaceutically acceptable salt is a sodium salt, potassium salt or calcium salt. In some embodiments, the pharmaceutically acceptable salt is a sodium salt.
INCORPORATION BY REFERENCE
[0077] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference.
BRIEF DESCRIPTION OF THE DRAWINGS
[0078] The novel features of the invention are set forth with particularity in the appended claims. A better understanding of the features and advantages of the present invention will be obtained by reference to the following detailed description that sets forth illustrative embodiments, in which the principles of the invention are utilized, and the accompanying drawings of which:
[0079] FIG. 1. Shows human wild type P53 protein sequence (SEQ ID NO: 1501).
[0080] FIG. 2. Shows an exemplary dose level and Dose Regimen.
[0081] FIG. 3. Shows an exemplary dosing overview.
[0082] FIG. 4. Shows the amount of Aileron peptide-1 administered for each dose level (DL) and dose regimen.
[0083] FIG. 5. Shows an exemplary dose escalation strategy of the disclosure.
[0084] FIG. 6. Shows one way Aileron peptide-1 was designed to inhibit both MDMX and MDM2, to reactivate WT p53.
[0085] FIG. 7. Shows potential indications of Aileron peptide-1 (from orphan indications or large market opportunities).
[0086] FIG. 8. Shows the effect of Aileron peptide-1 across a variety of different cancers.
[0087] FIG. 9. Shows the effect of Aileron peptide-1 administered by an intravenous, or IV, injection in an MDMX-driven MCF-7 breast cancer xenograft model
[0088] FIG. 10. Shows dose escalation based on a "3+3" dose escalation design.
[0089] FIGS. 11a and 11b Show drug concentration (measured or projected) in dose levels for Cohorts.
[0090] FIG. 12. Shows the pharmacokinetic model of Aileron peptide-1 which shows a 2-compartment, parallel nonlinear Michaelis-Menten clearance and linear elimination.
[0091] FIG. 13. Shows dose dependent increase of MIC-1.
[0092] FIG. 14. Shows that patients who have completed at least two cycles of treatment have stable disease. Aileron peptide-1 shows a stable disease rate.
[0093] FIG. 15. Shows that Aileron peptide 1 shows on target activation of p21 and p53 in patient blood cells.
DETAILED DESCRIPTION OF THE INVENTION
[0094] While preferred embodiments of the present disclosure have been shown and described herein, it will be obvious to those skilled in the art that such embodiments are provided by way of example only. Numerous variations, changes, and substitutions will now occur to those skilled in the art without departing from the disclosure. It should be understood that various alternatives to the embodiments of the disclosure described herein can be employed in practicing the disclosure. It is intended that the following claims define the scope of the disclosure and that methods and structures within the scope of these claims and their equivalents be covered thereby.
Definitions
[0095] As used herein, the term "macrocycle" refers to a molecule having a chemical structure including a ring or cycle formed by at least 9 covalently bonded atoms.
[0096] As used herein, the term "peptidomimetic macrocycle" or "crosslinked polypeptide" refers to a compound comprising a plurality of amino acid residues joined by a plurality of peptide bonds and at least one macrocycle-forming linker which forms a macrocycle between a first naturally-occurring or non-naturally-occurring amino acid residue (or analog) and a second naturally-occurring or non-naturally-occurring amino acid residue (or analog) within the same molecule. Peptidomimetic macrocycle include embodiments where the macrocycle-forming linker connects the .alpha. carbon of the first amino acid residue (or analog) to the .alpha. carbon of the second amino acid residue (or analog). The peptidomimetic macrocycles optionally include one or more non-peptide bonds between one or more amino acid residues and/or amino acid analog residues, and optionally include one or more non-naturally-occurring amino acid residues or amino acid analog residues in addition to any which form the macrocycle. A "corresponding uncrosslinked polypeptide" when referred to in the context of a peptidomimetic macrocycle is understood to relate to a polypeptide of the same length as the macrocycle and comprising the equivalent natural amino acids of the wild-type sequence corresponding to the macrocycle.
[0097] As used herein, the term "helical stability" refers to the maintenance of a helical structure by a peptidomimetic macrocycle as measured by circular dichroism or NMR. For example, in some embodiments, a peptidomimetic macrocycle exhibits at least a 1.25, 1.5, 1.75 or 2-fold increase in .alpha.-helicity as determined by circular dichroism compared to a corresponding uncrosslinked macrocycle.
[0098] The term "amino acid" refers to a molecule containing both an amino group and a carboxyl group. Suitable amino acids include, without limitation, both the D- and L-isomers of the naturally-occurring amino acids, as well as non-naturally occurring amino acids prepared by organic synthesis or other metabolic routes. The term amino acid, as used herein, includes, without limitation, .alpha.-amino acids, natural amino acids, non-natural amino acids, and amino acid analogs.
[0099] The term ".alpha.-amino acid" refers to a molecule containing both an amino group and a carboxyl group bound to a carbon which is designated the .alpha.-carbon.
[0100] The term ".beta.-amino acid" refers to a molecule containing both an amino group and a carboxyl group in a 3 configuration.
[0101] The term "naturally occurring amino acid" refers to any one of the twenty amino acids commonly found in peptides synthesized in nature, and known by the one letter abbreviations A, R, N, C, D, Q, E, G, H, I, L, K, M, F, P, S, T, W, Y and V.
[0102] The following table shows a summary of the properties of natural amino acids:
TABLE-US-00001 3- 1- Side- Side-chain Letter Letter chain charge Hydropathy Amino Acid Code Code Polarity (pH 7.4) Index Alanine Ala A nonpolar neutral 1.8 Arginine Arg R polar positive -4.5 Asparagine Asn N polar neutral -3.5 Aspartic acid Asp D polar negative -3.5 Cysteine Cys C polar neutral 2.5 Glutamic acid Glu E polar negative -3.5 Glutamine Gln Q polar neutral -3.5 Glycine Gly G nonpolar neutral -0.4 Histidine His H polar positive(10%) -3.2 neutral(90%) Isoleucine Ile I nonpolar neutral 4.5 Leucine Leu L nonpolar neutral 3.8 Lysine Lys K polar positive -3.9 Methionine Met M nonpolar neutral 1.9 Phenylalanine Phe F nonpolar neutral 2.8 Proline Pro P nonpolar neutral -1.6 Serine Ser S polar neutral -0.8 Threonine Thr T polar neutral -0.7 Tryptophan Trp W nonpolar neutral -0.9 Tyrosine Tyr Y polar neutral -1.3 Valine Val V nonpolar neutral 4.2
[0103] "Hydrophobic amino acids" include small hydrophobic amino acids and large hydrophobic amino acids. "Small hydrophobic amino acid" are glycine, alanine, proline, and analogs thereof. "Large hydrophobic amino acids" are valine, leucine, isoleucine, phenylalanine, methionine, tryptophan, and analogs thereof. "Polar amino acids" are serine, threonine, asparagine, glutamine, cysteine, tyrosine, and analogs thereof. "Charged amino acids" are lysine, arginine, histidine, aspartate, glutamate, and analogs thereof.
[0104] The term "amino acid analog" refers to a molecule which is structurally similar to an amino acid and which can be substituted for an amino acid in the formation of a peptidomimetic macrocycle. Amino acid analogs include, without limitation, .beta.-amino acids and amino acids where the amino or carboxy group is substituted by a similarly reactive group (e.g., substitution of the primary amine with a secondary or tertiary amine, or substitution of the carboxy group with an ester).
[0105] The term "non-natural amino acid" refers to an amino acid which is not one of the twenty amino acids commonly found in peptides synthesized in nature, and known by the one letter abbreviations A, R, N, C, D, Q, E, G, H, I, L, K, M, F, P, S, T, W, Y and V. Non-natural amino acids or amino acid analogs include, without limitation, structures according to the following:
##STR00004## ##STR00005## ##STR00006## ##STR00007## ##STR00008## ##STR00009##
[0106] Amino acid analogs include .beta.-amino acid analogs. Examples of .beta.-amino acid analogs include, but are not limited to, the following: cyclic .beta.-amino acid analogs; .beta.-alanine; (R)-.beta.-phenylalanine; (R)-1,2,3,4-tetrahydro-isoquinoline-3-acetic acid; (R)-3-amino-4-(1-naphthyl)-butyric acid; (R)-3-amino-4-(2,4-dichlorophenyl)butyric acid; (R)-3-amino-4-(2-chlorophenyl)-butyric acid; (R)-3-amino-4-(2-cyanophenyl)-butyric acid; (R)-3-amino-4-(2-fluorophenyl)-butyric acid; (R)-3-amino-4-(2-furyl)-butyric acid; (R)-3-amino-4-(2-methylphenyl)-butyric acid; (R)-3-amino-4-(2-naphthyl)-butyric acid; (R)-3-amino-4-(2-thienyl)-butyric acid; (R)-3-amino-4-(2-trifluoromethylphenyl)-butyric acid; (R)-3-amino-4-(3,4-dichlorophenyl)butyric acid; (R)-3-amino-4-(3,4-difluorophenyl)butyric acid; (R)-3-amino-4-(3-benzothienyl)-butyric acid; (R)-3-amino-4-(3-chlorophenyl)-butyric acid; (R)-3-amino-4-(3-cyanophenyl)-butyric acid; (R)-3-amino-4-(3-fluorophenyl)-butyric acid; (R)-3-amino-4-(3-methylphenyl)-butyric acid; (R)-3-amino-4-(3-pyridyl)-butyric acid; (R)-3-amino-4-(3-thienyl)-butyric acid; (R)-3-amino-4-(3-trifluoromethylphenyl)-butyric acid; (R)-3-amino-4-(4-bromophenyl)-butyric acid; (R)-3-amino-4-(4-chlorophenyl)-butyric acid; (R)-3-amino-4-(4-cyanophenyl)-butyric acid; (R)-3-amino-4-(4-fluorophenyl)-butyric acid; (R)-3-amino-4-(4-iodophenyl)-butyric acid; (R)-3-amino-4-(4-methylphenyl)-butyric acid; (R)-3-amino-4-(4-nitrophenyl)-butyric acid; (R)-3-amino-4-(4-pyridyl)-butyric acid; (R)-3-amino-4-(4-trifluoromethylphenyl)-butyric acid; (R)-3-amino-4-pentafluoro-phenylbutyric acid; (R)-3-amino-5-hexenoic acid; (R)-3-amino-5-hexynoic acid; (R)-3-amino-5-phenylpentanoic acid; (R)-3-amino-6-phenyl-5-hexenoic acid; (S)-1,2,3,4-tetrahydro-isoquinoline-3-acetic acid; (S)-3-amino-4-(1-naphthyl)-butyric acid; (S)-3-amino-4-(2,4-dichlorophenyl)butyric acid; (S)-3-amino-4-(2-chlorophenyl)-butyric acid; (S)-3-amino-4-(2-cyanophenyl)-butyric acid; (S)-3-amino-4-(2-fluorophenyl)-butyric acid; (S)-3-amino-4-(2-furyl)-butyric acid; (S)-3-amino-4-(2-methylphenyl)-butyric acid; (S)-3-amino-4-(2-naphthyl)-butyric acid; (S)-3-amino-4-(2-thienyl)-butyric acid; (S)-3-amino-4-(2-trifluoromethylphenyl)-butyric acid; (S)-3-amino-4-(3,4-dichlorophenyl)butyric acid; (S)-3-amino-4-(3,4-difluorophenyl)butyric acid; (S)-3-amino-4-(3-benzothienyl)-butyric acid; (S)-3-amino-4-(3-chlorophenyl)-butyric acid; (S)-3-amino-4-(3-cyanophenyl)-butyric acid; (S)-3-amino-4-(3-fluorophenyl)-butyric acid; (S)-3-amino-4-(3-methylphenyl)-butyric acid; (S)-3-amino-4-(3-pyridyl)-butyric acid; (S)-3-amino-4-(3-thienyl)-butyric acid; (S)-3-amino-4-(3-trifluoromethylphenyl)-butyric acid; (S)-3-amino-4-(4-bromophenyl)-butyric acid; (S)-3-amino-4-(4-chlorophenyl)-butyric acid; (S)-3-amino-4-(4-cyanophenyl)-butyric acid; (S)-3-amino-4-(4-fluorophenyl)-butyric acid; (S)-3-amino-4-(4-iodophenyl)-butyric acid; (S)-3-amino-4-(4-methylphenyl)-butyric acid; (S)-3-amino-4-(4-nitrophenyl)-butyric acid; (S)-3-amino-4-(4-pyridyl)-butyric acid; (S)-3-amino-4-(4-trifluoromethylphenyl)-butyric acid; (S)-3-amino-4-pentafluoro-phenylbutyric acid; (S)-3-amino-5-hexenoic acid; (S)-3-amino-5-hexynoic acid; (S)-3-amino-5-phenylpentanoic acid; (S)-3-amino-6-phenyl-5-hexenoic acid; 1,2,5,6-tetrahydropyridine-3-carboxylic acid; 1,2,5,6-tetrahydropyridine-4-carboxylic acid; 3-amino-3-(2-chlorophenyl)-propionic acid; 3-amino-3-(2-thienyl)-propionic acid; 3-amino-3-(3-bromophenyl)-propionic acid; 3-amino-3-(4-chlorophenyl)-propionic acid; 3-amino-3-(4-methoxyphenyl)-propionic acid; 3-amino-4,4,4-trifluoro-butyric acid; 3-aminoadipic acid; D-.beta.-phenylalanine; .beta.-leucine; L-.beta.-homoalanine; L-.beta.-homoaspartic acid .gamma.-benzyl ester; L-.beta.-homoglutamic acid .delta.-benzyl ester; L-.beta.-homoisoleucine; L-.beta.-homoleucine; L-.beta.-homomethionine; L-.beta.-homophenylalanine; L-.beta.-homoproline; L-.beta.3-homotryptophan; L-.beta.-homovaline; L-N.omega.-benzyloxycarbonyl-.beta.-homolysine; N.omega.-L-.beta.-homoarginine; O-benzyl-L-.beta.-homohydroxyproline; O-benzyl-L-.beta.-homoserine; O-benzyl-L-.beta.-homothreonine; O-benzyl-L-.beta.-homotyrosine; .gamma.-trityl-L-.beta.-homoasparagine; (R)-.beta.-phenylalanine; L-.beta.-homoaspartic acid .gamma.-t-butyl ester; L-.beta.-homoglutamic acid .delta.-t-butyl ester; L-N.omega.-.beta.-homolysine; N.delta.-trityl-L-.beta.-homoglutamine; N.omega.-2,2,4,6,7-pentamethyl-dihydrobenzofuran-5-sulfonyl-L-.beta.-homo- arginine; O-t-butyl-L-.beta.-homohydroxy-proline; O-t-butyl-L-.beta.-homoserine; O-t -butyl-L-.beta.-homothreonine; O-t-butyl-L-.beta.-homotyrosine; 2-aminocyclopentane carboxylic acid; and 2-aminocyclohexane carboxylic acid.
[0107] Amino acid analogs include analogs of alanine, valine, glycine or leucine. Examples of amino acid analogs of alanine, valine, glycine, and leucine include, but are not limited to, the following: .alpha.-methoxyglycine; .alpha.-allyl-L-alanine; .alpha.-aminoisobutyric acid; .alpha.-methyl-leucine; .beta.-(1-naphthyl)-D-alanine; .beta.-(1-naphthyl)-L-alanine; .beta.-(2-naphthyl)-D-alanine; .beta.-(2-naphthyl)-L-alanine; .beta.-(2-pyridyl)-D-alanine; .beta.-(2-pyridyl)-L-alanine; .beta.-(2-thienyl)-D-alanine; .beta.-(2-thienyl)-L-alanine; .beta.-(3-benzothienyl)-D-alanine; .beta.-(3-benzothienyl)-L-alanine; .beta.-(3-pyridyl)-D-alanine; .beta.-(3-pyridyl)-L-alanine; .beta.-(4-pyridyl)-D-alanine; .beta.-(4-pyridyl)-L-alanine; .beta.-chloro-L-alanine; .beta.-cyano-L-alanin; .beta.-cyclohexyl-D-alanine; .beta.-cyclohexyl-L-alanine; .beta.-cyclopenten-1-yl-alanine; .beta.-cyclopentyl-alanine; .beta.-cyclopropyl-L-Ala-OH.dicyclohexylammonium salt; .beta.-t-butyl-D-alanine; .beta.-t-butyl-L-alanine; .gamma.-aminobutyric acid; L-.alpha.,.beta.-diaminopropionic acid; 2,4-dinitro-phenylglycine; 2,5-dihydro-D-phenylglycine; 2-amino-4,4,4-trifluorobutyric acid; 2-fluoro-phenylglycine; 3-amino-4,4,4-trifluoro-butyric acid; 3-fluoro-valine; 4,4,4-trifluoro-valine; 4,5-dehydro-L-leu-OH.dicyclohexylammonium salt; 4-fluoro-D-phenylglycine; 4-fluoro-L-phenylglycine; 4-hydroxy-D-phenylglycine; 5,5,5-trifluoro-leucine; 6-aminohexanoic acid; cyclopentyl-D-Gly-OH.dicyclohexylammonium salt; cyclopentyl-Gly-OH.dicyclohexylammonium salt; D-.alpha.,.beta.-diaminopropionic acid; D-.alpha.-aminobutyric acid; D-.alpha.-t-butylglycine; D-(2-thienyl)glycine; D-(3-thienyl)glycine; D-2-aminocaproic acid; D-2-indanylglycine; D-allylglycine.dicyclohexylammonium salt; D-cyclohexylglycine; D-norvaline; D-phenylglycine; .beta.-aminobutyric acid; .beta.-aminoisobutyric acid; (2-bromophenyl)glycine; (2-methoxyphenyl)glycine; (2-methylphenyl)glycine; (2-thiazoyl)glycine; (2-thienyl)glycine; 2-amino-3-(dimethylamino)-propionic acid; L-.alpha.,.beta.-diaminopropionic acid; L-.alpha.-aminobutyric acid; L-.alpha.-t-butylglycine; L-(3-thienyl)glycine; L-2-amino-3-(dimethylamino)-propionic acid; L-2-aminocaproic acid dicyclohexyl-ammonium salt; L-2-indanylglycine; L-allylglycine.dicyclohexyl ammonium salt; L-cyclohexylglycine; L-phenylglycine; L-propargylglycine; L-norvaline; N-.alpha.-aminomethyl-L-alanine; D-.alpha.,.gamma.-diaminobutyric acid; L-.alpha.,.gamma.-diaminobutyric acid; .beta.-cyclopropyl-L-alanine; (N-.beta.-(2,4-dinitrophenyl))-L-.alpha.,.beta.-diaminopropionic acid; (N-.beta.-1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)ethyl)-D-.alpha.,.b- eta.-diaminopropionic acid; (N-.beta.-1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)ethyl)-L-.alpha.,.b- eta.-diaminopropionic acid; (N-.beta.-4-methyltrityl)-L-.alpha.,.beta.-diaminopropionic acid; (N-.beta.-allyloxycarbonyl)-L-.alpha.,.beta.-diaminopropionic acid; (N-.gamma.-1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)ethyl)-D-.alpha.,.- gamma.-diaminobutyric acid; (N-.gamma.-1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)ethyl)-L-.alpha.,.- gamma.-diaminobutyric acid; (N-.gamma.-4-methyltrityl)-D-.alpha.,.gamma.-diaminobutyric acid; (N-.gamma.-4-methyltrityl)-L-.alpha.,.gamma.-diaminobutyric acid; (N-.gamma.-allyloxycarbonyl)-L-.alpha.,.gamma.-diaminobutyric acid; D-.alpha.,.gamma.-diaminobutyric acid; 4,5-dehydro-L-leucine; cyclopentyl-D-Gly-OH; cyclopentyl-Gly-OH; D-allylglycine; D-homocyclohexylalanine; L-1-pyrenylalanine; L-2-aminocaproic acid; L-allylglycine; L-homocyclohexylalanine; and N-(2-hydroxy-4-methoxy-Bzl)-Gly-OH.
[0108] Amino acid analogs include analogs of arginine or lysine. Examples of amino acid analogs of arginine and lysine include, but are not limited to, the following: citrulline; L-2-amino-3-guanidinopropionic acid; L-2-amino-3-ureidopropionic acid; L-citrulline; Lys(Me).sub.2-OH; Lys(N.sub.3)--OH; N.delta.-benzyloxycarbonyl-L-omithine; No-nitro-D-arginine; No-nitro-L-arginine; .alpha.-methyl-omithine; 2,6-diaminoheptanedioic acid; L-omithine; (N.delta.-1-(4,4-dimethyl-2,6-dioxo-cyclohex-1-ylidene)ethyl)-D-omithine; (N.delta.-1-(4,4-dimethyl-2,6-dioxo-cyclohex-1-ylidene)ethyl)-L-omithine; (N.delta.-4-methyltrityl)-D-omithine; (N.delta.-4-methyltrityl)-L-omithine; D-omithine; L-omithine; Arg(Me)(Pbf)-OH; Arg(Me).sub.2-OH (asymmetrical); Arg(Me).sub.2-OH (symmetrical); Lys(ivDde)-OH; Lys(Me).sub.2-OH.HCl; Lys(Me3)-OH chloride; No-nitro-D-arginine; and N.omega.-nitro-L-arginine.
[0109] Amino acid analogs include analogs of aspartic or glutamic acids. Examples of amino acid analogs of aspartic and glutamic acids include, but are not limited to, the following: .alpha.-methyl-D-aspartic acid; .alpha.-methyl-glutamic acid; .alpha.-methyl-L-aspartic acid; .gamma.-methylene-glutamic acid; (N-.gamma.-ethyl)-L-glutamine; [N-.alpha.-(4-aminobenzoyl)]-L-glutamic acid; 2,6-diaminopimelic acid; L-.alpha.-aminosuberic acid; D-2-aminoadipic acid; D-.alpha.-aminosuberic acid; .alpha.-aminopimelic acid; iminodiacetic acid; L-2-aminoadipic acid; threo-.beta.-methyl-aspartic acid; .gamma.-carboxy-D-glutamic acid .gamma.,.gamma.-di-t-butyl ester; .gamma.-carboxy-L-glutamic acid .gamma.,.gamma.-di-t-butyl ester; Glu(OAll)-OH; L-Asu(OtBu)-OH; and pyroglutamic acid.
[0110] Amino acid analogs include analogs of cysteine and methionine. Examples of amino acid analogs of cysteine and methionine include, but are not limited to, Cys(famesyl)-OH, Cys(famesyl)-OMe, .alpha.-methyl-methionine, Cys(2-hydroxyethyl)-OH, Cys(3-aminopropyl)-OH, 2-amino-4-(ethylthio)butyric acid, buthionine, buthioninesulfoximine, ethionine, methionine methylsulfonium chloride, selenomethionine, cysteic acid, [2-(4-pyridyl)ethyl]-DL-penicillamine, [2-(4-pyridyl)ethyl]-L-cysteine, 4-methoxybenzyl-D-penicillamine, 4-methoxybenzyl-L-penicillamine, 4-methylbenzyl-D-penicillamine, 4-methylbenzyl-L-penicillamine, benzyl-D-cysteine, benzyl-L-cysteine, benzyl-DL-homocysteine, carbamoyl-L-cysteine, carboxyethyl-L-cysteine, carboxymethyl-L-cysteine, diphenylmethyl-L-cysteine, ethyl-L-cysteine, methyl-L-cysteine, t-butyl-D-cysteine, trityl-L-homocysteine, trityl-D-penicillamine, cystathionine, homocystine, L-homocystine, (2-aminoethyl)-L-cysteine, seleno-L-cystine, cystathionine, Cys(StBu)-OH, and acetamidomethyl-D-penicillamine.
[0111] Amino acid analogs include analogs of phenylalanine and tyrosine. Examples of amino acid analogs of phenylalanine and tyrosine include 3-methyl-phenylalanine, 3-hydroxyphenylalanine, .alpha.-methyl-3-methoxy-DL-phenylalanine, .alpha.-methyl-D-phenylalanine, .alpha.-methyl-L-phenylalanine, 1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid, 2,4-dichloro-phenylalanine, 2-(trifluoromethyl)-D-phenylalanine, 2-(trifluoromethyl)-L-phenylalanine, 2-bromo-D-phenylalanine, 2-bromo-L-phenylalanine, 2-chloro-D-phenylalanine, 2-chloro-L-phenylalanine, 2-cyano-D-phenylalanine, 2-cyano-L-phenylalanine, 2-fluoro-D-phenylalanine, 2-fluoro-L-phenylalanine, 2-methyl-D-phenylalanine, 2-methyl-L-phenylalanine, 2-nitro-D-phenylalanine, 2-nitro-L-phenylalanine, 2;4;5-trihydroxy-phenylalanine, 3,4,5-trifluoro-D-phenylalanine, 3,4,5-trifluoro-L-phenylalanine, 3,4-dichloro-D-phenylalanine, 3,4-dichloro-L-phenylalanine, 3,4-difluoro-D-phenylalanine, 3,4-difluoro-L-phenylalanine, 3,4-dihydroxy-L-phenylalanine, 3,4-dimethoxy-L-phenylalanine, 3,5,3'-triiodo-L-thyronine, 3,5-diiodo-D-tyrosine, 3,5-diiodo-L-tyrosine, 3,5-diiodo-L-thyronine, 3-(trifluoromethyl)-D-phenylalanine, 3-(trifluoromethyl)-L-phenylalanine, 3-amino-L-tyrosine, 3-bromo-D-phenylalanine, 3-bromo-L-phenylalanine, 3-chloro-D-phenylalanine, 3-chloro-L-phenylalanine, 3-chloro-L-tyrosine, 3-cyano-D-phenylalanine, 3-cyano-L-phenylalanine, 3-fluoro-D-phenylalanine, 3-fluoro-L-phenylalanine, 3-fluoro-tyrosine, 3-iodo-D-phenylalanine, 3-iodo-L-phenylalanine, 3-iodo-L-tyrosine, 3-methoxy-L-tyrosine, 3-methyl-D-phenylalanine, 3-methyl-L-phenylalanine, 3-nitro-D-phenylalanine, 3-nitro-L-phenylalanine, 3-nitro-L-tyrosine, 4-(trifluoromethyl)-D-phenylalanine, 4-(trifluoromethyl)-L-phenylalanine, 4-amino-D-phenylalanine, 4-amino-L-phenylalanine, 4-benzoyl-D-phenylalanine, 4-benzoyl-L-phenylalanine, 4-bis(2-chloroethyl)amino-L-phenylalanine, 4-bromo-D-phenylalanine, 4-bromo-L-phenylalanine, 4-chloro-D-phenylalanine, 4-chloro-L-phenylalanine, 4-cyano-D-phenylalanine, 4-cyano-L-phenylalanine, 4-fluoro-D-phenylalanine, 4-fluoro-L-phenylalanine, 4-iodo-D-phenylalanine, 4-iodo-L-phenylalanine, homophenylalanine, thyroxine, 3,3-diphenylalanine, thyronine, ethyl-tyrosine, and methyl-tyrosine.
[0112] Amino acid analogs include analogs of proline. Examples of amino acid analogs of proline include, but are not limited to, 3,4-dehydro-proline, 4-fluoro-proline, cis-4-hydroxy-proline, thiazolidine-2-carboxylic acid, and trans-4-fluoro-proline.
[0113] Amino acid analogs include analogs of serine and threonine. Examples of amino acid analogs of serine and threonine include, but are not limited to, 3-amino-2-hydroxy-5-methylhexanoic acid, 2-amino-3-hydroxy-4-methylpentanoic acid, 2-amino-3-ethoxybutanoic acid, 2-amino-3-methoxybutanoic acid, 4-amino-3-hydroxy-6-methylheptanoic acid, 2-amino-3-benzyloxypropionic acid, 2-amino-3-benzyloxypropionic acid, 2-amino-3-ethoxypropionic acid, 4-amino-3-hydroxybutanoic acid, and .alpha.-methylserine.
[0114] Amino acid analogs include analogs of tryptophan. Examples of amino acid analogs of tryptophan include, but are not limited to, the following: .alpha.-methyl-tryptophan; .beta.-(3-benzothienyl)-D-alanine; .beta.-(3-benzothienyl)-L-alanine; 1-methyl-tryptophan; 4-methyl-tryptophan; 5-benzyloxy-tryptophan; 5-bromo-tryptophan; 5-chloro-tryptophan; 5-fluoro-tryptophan; 5-hydroxy-tryptophan; 5-hydroxy-L-tryptophan; 5-methoxy-tryptophan; 5-methoxy-L-tryptophan; 5-methyl-tryptophan; 6-bromo-tryptophan; 6-chloro-D-tryptophan; 6-chloro-tryptophan; 6-fluoro-tryptophan; 6-methyl-tryptophan; 7-benzyloxy-tryptophan; 7-bromo-tryptophan; 7-methyl-tryptophan; D-1,2,3,4-tetrahydro-norharman-3-carboxylic acid; 6-methoxy-1,2,3,4-tetrahydronorharman-1-carboxylic acid; 7-azatryptophan; L-1,2,3,4-tetrahydro-norharman-3-carboxylic acid; 5-methoxy-2-methyl-tryptophan; and 6-chloro-L-tryptophan.
[0115] In some embodiments, amino acid analogs are racemic. In some embodiments, the D isomer of the amino acid analog is used. In some embodiments, the L isomer of the amino acid analog is used. In other embodiments, the amino acid analog comprises chiral centers that are in the R or S configuration. In still other embodiments, the amino group(s) of a .beta.-amino acid analog is substituted with a protecting group, e.g., tert-butyloxycarbonyl (BOC group), 9-fluorenylmethyloxycarbonyl (FMOC), tosyl, and the like. In yet other embodiments, the carboxylic acid functional group of a .beta.-amino acid analog is protected, e.g., as its ester derivative. In some embodiments the salt of the amino acid analog is used.
[0116] A "non-essential" amino acid residue is a residue that can be altered from the wild-type sequence of a polypeptide without abolishing or substantially altering its essential biological or biochemical activity (e.g., receptor binding or activation). An "essential" amino acid residue is a residue that, when altered from the wild-type sequence of the polypeptide, results in abolishing or substantially abolishing the polypeptide's essential biological or biochemical activity.
[0117] A "conservative amino acid substitution" is one in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art. These families include amino acids with basic side chains (e.g., K, R, H), acidic side chains (e.g., D, E), uncharged polar side chains (e.g., G, N, Q, S, T, Y, C), nonpolar side chains (e.g., A, V, L, I, P, F, M, W), beta-branched side chains (e.g., T, V, I) and aromatic side chains (e.g., Y, F, W, H). Thus, a predicted nonessential amino acid residue in a polypeptide, for example, is replaced with another amino acid residue from the same side chain family. Other examples of acceptable substitutions are substitutions based on isosteric considerations (e.g. norleucine for methionine) or other properties (e.g. 2-thienylalanine for phenylalanine, or 6-Cl-tryptophan for tryptophan).
[0118] The term "capping group" refers to the chemical moiety occurring at either the carboxy or amino terminus of the polypeptide chain of the subject peptidomimetic macrocycle. The capping group of a carboxy terminus includes an unmodified carboxylic acid (i.e. --COOH) or a carboxylic acid with a substituent.
[0119] For example, the carboxy terminus can be substituted with an amino group to yield a carboxamide at the C-terminus. Various substituents include but are not limited to primary and secondary amines, including pegylated secondary amines. Representative secondary amine capping groups for the C-terminus include:
##STR00010##
[0120] The capping group of an amino terminus includes an unmodified amine (ie --NH.sub.2) or an amine with a substituent. For example, the amino terminus can be substituted with an acyl group to yield a carboxamide at the N-terminus. Various substituents include but are not limited to substituted acyl groups, including C.sub.1-C.sub.6 carbonyls, C.sub.7-C.sub.30 carbonyls, and pegylated carbamates. Representative capping groups for the N-terminus include, but are not limited to, 4-FBzl (4-fluoro-benzyl) and the following:
##STR00011##
[0121] The term "member" as used herein in conjunction with macrocycles or macrocycle-forming linkers refers to the atoms that form or can form the macrocycle, and excludes substituent or side chain atoms. By analogy, cyclodecane, 1,2-difluoro-decane and 1,3-dimethyl cyclodecane are all considered ten-membered macrocycles as the hydrogen or fluoro substituents or methyl side chains do not participate in forming the macrocycle.
[0122] The symbol "" when used as part of a molecular structure refers to a single bond or a trans or cis double bond.
[0123] The term "amino acid side chain" refers to a moiety attached to the .alpha.-carbon (or another backbone atom) in an amino acid. For example, the amino acid side chain for alanine is methyl, the amino acid side chain for phenylalanine is phenylmethyl, the amino acid side chain for cysteine is thiomethyl, the amino acid side chain for aspartate is carboxymethyl, the amino acid side chain for tyrosine is 4-hydroxyphenylmethyl, etc. Other non-naturally occurring amino acid side chains are also included, for example, those that occur in nature (e.g., an amino acid metabolite) or those that are made synthetically (e.g., an .alpha.,.alpha. di-substituted amino acid).
[0124] The term ".alpha.,.alpha. di-substituted amino" acid refers to a molecule or moiety containing both an amino group and a carboxyl group bound to a carbon (the .alpha.-carbon) that is attached to two natural or non-natural amino acid side chains.
[0125] The term "polypeptide" encompasses two or more naturally or non-naturally-occurring amino acids joined by a covalent bond (e.g., an amide bond). Polypeptides as described herein include full length proteins (e.g., fully processed proteins) as well as shorter amino acid sequences (e.g., fragments of naturally-occurring proteins or synthetic polypeptide fragments).
[0126] The term "first C-terminal amino acid" refers to the amino acid which is closest to the C-terminus. The term "second C-terminal amino acid" refers to the amino acid attached at the N-terminus of the first C-terminal amino acid.
[0127] The term "macrocyclization reagent" or "macrocycle-forming reagent" as used herein refers to any reagent which can be used to prepare a peptidomimetic macrocycle by mediating the reaction between two reactive groups. Reactive groups can be, for example, an azide and alkyne, in which case macrocyclization reagents include, without limitation, Cu reagents such as reagents which provide a reactive Cu(I) species, such as CuBr, CuI or CuOTf, as well as Cu(II) salts such as Cu(CO.sub.2CH.sub.3).sub.2, CuSO.sub.4, and CuCl.sub.2 that can be converted in situ to an active Cu(I) reagent by the addition of a reducing agent such as ascorbic acid or sodium ascorbate. Macrocyclization reagents can additionally include, for example, Ru reagents known in the art such as Cp*RuCl(PPh.sub.3).sub.2, [Cp*RuCl].sub.4 or other Ru reagents which can provide a reactive Ru(II) species. In other cases, the reactive groups are terminal olefins. In such embodiments, the macrocyclization reagents or macrocycle-forming reagents are metathesis catalysts including, but not limited to, stabilized, late transition metal carbene complex catalysts such as Group VIII transition metal carbene catalysts. For example, such catalysts are Ru and Os metal centers having a +2 oxidation state, an electron count of 16 and pentacoordinated. In other examples, catalysts have W or Mo centers. Various catalysts are disclosed in Grubbs et al., "Ring Closing Metathesis and Related Processes in Organic Synthesis" Acc. Chem. Res. 1995, 28, 446-452, U.S. Pat. Nos. 5,811,515; 7,932,397; U.S. Application No. 2011/0065915; U.S. Application No. 2011/0245477; Yu et al., "Synthesis of Macrocyclic Natural Products by Catalyst-Controlled Stereoselective Ring-Closing Metathesis," Nature 2011, 479, 88; and Peryshkov et al., "Z-Selective Olefin Metathesis Reactions Promoted by Tungsten Oxo Alkylidene Complexes," J. Am. Chem. Soc. 2011, 133, 20754. In yet other cases, the reactive groups are thiol groups. In such embodiments, the macrocyclization reagent is, for example, a linker functionalized with two thiol-reactive groups such as halogen groups. In some examples, the macrocyclization reagent include palladium reagents, for example Pd(PPh.sub.3).sub.4, Pd(PPh.sub.3).sub.2Cl.sub.2, Pd(dppe)Cl, Pd(dppp)Cl.sub.2, and Pd(dppf)Cl.sub.2.
[0128] The term "halo" or "halogen" refers to fluorine, chlorine, bromine or iodine or a radical thereof.
[0129] The term "alkyl" refers to a hydrocarbon chain that is a straight chain or branched chain, containing the indicated number of carbon atoms. For example, C.sub.1-C.sub.10 indicates that the group has from 1 to 10 (inclusive) carbon atoms in it. In the absence of any numerical designation, "alkyl" is a chain (straight or branched) having 1 to 20 (inclusive) carbon atoms in it.
[0130] The term "alkylene" refers to a divalent alkyl (i.e., --R--).
[0131] The term "alkenyl" refers to a hydrocarbon chain that is a straight chain or branched chain having one or more carbon-carbon double bonds. The alkenyl moiety contains the indicated number of carbon atoms. For example, C.sub.2-C.sub.10 indicates that the group has from 2 to 10 (inclusive) carbon atoms in it. The term "lower alkenyl" refers to a C.sub.2-C.sub.6 alkenyl chain. In the absence of any numerical designation, "alkenyl" is a chain (straight or branched) having 2 to 20 (inclusive) carbon atoms in it.
[0132] The term "alkynyl" refers to a hydrocarbon chain that is a straight chain or branched chain having one or more carbon-carbon triple bonds. The alkynyl moiety contains the indicated number of carbon atoms. For example, C.sub.2-C.sub.10 indicates that the group has from 2 to 10 (inclusive) carbon atoms in it. The term "lower alkynyl" refers to a C.sub.2-C.sub.6 alkynyl chain. In the absence of any numerical designation, "alkynyl" is a chain (straight or branched) having 2 to 20 (inclusive) carbon atoms in it.
[0133] The term "aryl" refers to a 6-carbon monocyclic or 10-carbon bicyclic aromatic ring system wherein 0, 1, 2, 3, or 4 atoms of each ring are substituted by a substituent. Examples of aryl groups include phenyl, naphthyl and the like. The term "arylalkoxy" refers to an alkoxy substituted with aryl.
[0134] "Arylalkyl" refers to an aryl group, as defined above, wherein one of the aryl group's hydrogen atoms has been replaced with a C.sub.1-C.sub.5 alkyl group, as defined above. Representative examples of an arylalkyl group include, but are not limited to, 2-methylphenyl, 3-methylphenyl, 4-methylphenyl, 2-ethylphenyl, 3-ethylphenyl, 4-ethylphenyl, 2-propylphenyl, 3-propylphenyl, 4-propylphenyl, 2-butylphenyl, 3-butylphenyl, 4-butylphenyl, 2-pentylphenyl, 3-pentylphenyl, 4-pentylphenyl, 2-isopropylphenyl, 3-isopropylphenyl, 4-isopropylphenyl, 2-isobutylphenyl, 3-isobutylphenyl, 4-isobutylphenyl, 2-sec-butylphenyl, 3-sec-butylphenyl, 4-sec-butylphenyl, 2-t-butylphenyl, 3-t-butylphenyl and 4-t-butylphenyl.
[0135] "Arylamido" refers to an aryl group, as defined above, wherein one of the aryl group's hydrogen atoms has been replaced with one or more --C(O)NH.sub.2 groups. Representative examples of an arylamido group include 2-C(O)NH.sub.2-phenyl, 3-C(O)NH.sub.2-phenyl, 4-C(O)NH.sub.2-phenyl, 2-C(O)NH.sub.2-pyridyl, 3-C(O)NH.sub.2-pyridyl, and 4-C(O)NH.sub.2-pyridyl,
[0136] "Alkylheterocycle" refers to a C.sub.1-C.sub.5 alkyl group, as defined above, wherein one of the C.sub.1-C.sub.5 alkyl group's hydrogen atoms has been replaced with a heterocycle. Representative examples of an alkylheterocycle group include, but are not limited to, --CH.sub.2CH.sub.2-morpholine, --CH.sub.2CH.sub.2-piperidine, --CH.sub.2CH.sub.2CH.sub.2-morpholine, and --CH.sub.2CH.sub.2CH.sub.2-imidazole.
[0137] "Alkylamido" refers to a C.sub.1-C.sub.5 alkyl group, as defined above, wherein one of the C.sub.1-C.sub.5 alkyl group's hydrogen atoms has been replaced with a --C(O)NH.sub.2 group. Representative examples of an alkylamido group include, but are not limited to, --CH.sub.2--C(O)NH.sub.2, --CH.sub.2CH.sub.2--C(O)NH.sub.2, --CH.sub.2CH.sub.2CH.sub.2C(O)NH.sub.2, --CH.sub.2CH.sub.2CH.sub.2CH.sub.2C(O)NH.sub.2, --CH.sub.2CH.sub.2CH.sub.2CH.sub.2CH.sub.2C(O)NH.sub.2, --CH.sub.2CH(C(O)NH.sub.2)CH.sub.3, --CH.sub.2CH(C(O)NH.sub.2)CH.sub.2CH.sub.3, --CH(C(O)NH.sub.2)CH.sub.2CH.sub.3, --C(CH.sub.3).sub.2CH.sub.2C(O)NH.sub.2, --CH.sub.2--CH.sub.2--NH--C(O)--CH.sub.3, --CH.sub.2--CH.sub.2--NH--C(O)--CH.sub.3--CH3, and --CH.sub.2--CH.sub.2--NH--C(O)--CH.dbd.CH.sub.2.
[0138] "Alkanol" refers to a C.sub.1-C.sub.5 alkyl group, as defined above, wherein one of the C.sub.1-C.sub.5 alkyl group's hydrogen atoms has been replaced with a hydroxyl group. Representative examples of an alkanol group include, but are not limited to, --CH.sub.2OH, --CH.sub.2CH.sub.2OH, --CH.sub.2CH.sub.2CH.sub.2OH, --CH.sub.2CH.sub.2CH.sub.2CH.sub.2OH, --CH.sub.2CH.sub.2CH.sub.2 CH.sub.2CH.sub.2OH, --CH.sub.2CH(OH)CH.sub.3, --CH.sub.2CH(OH)CH.sub.2CH.sub.3, --CH(OH)CH.sub.3 and --C(CH.sub.3).sub.2CH.sub.2OH.
[0139] "Alkylcarboxy" refers to a C.sub.1-C.sub.5 alkyl group, as defined above, wherein one of the C.sub.1-C.sub.5 alkyl group's hydrogen atoms has been replaced with a --COOH group. Representative examples of an alkylcarboxy group include, but are not limited to, --CH.sub.2COOH, --CH.sub.2CH.sub.2COOH, --CH.sub.2CH.sub.2CH.sub.2COOH, --CH.sub.2CH.sub.2CH.sub.2CH.sub.2COOH, --CH.sub.2CH(COOH)CH.sub.3, --CH.sub.2CH.sub.2CH.sub.2CH.sub.2CH.sub.2COOH, --CH.sub.2CH(COOH)CH.sub.2CH.sub.3, --CH(COOH)CH.sub.2CH.sub.3 and --C(CH.sub.3).sub.2CH.sub.2COOH.
[0140] The term "cycloalkyl" as employed herein includes saturated and partially unsaturated cyclic hydrocarbon groups having 3 to 12 carbons, preferably 3 to 8 carbons, and more preferably 3 to 6 carbons, wherein the cycloalkyl group additionally is optionally substituted. Some cycloalkyl groups include, without limitation, cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cycloheptyl, and cyclooctyl.
[0141] The term "heteroaryl" refers to an aromatic 5-8 membered monocyclic, 8-12 membered bicyclic, or 11-14 membered tricyclic ring system having 1-3 heteroatoms if monocyclic, 1-6 heteroatoms if bicyclic, or 1-9 heteroatoms if tricyclic, said heteroatoms selected from O, N, or S (e.g., carbon atoms and 1-3, 1-6, or 1-9 heteroatoms of O, N, or S if monocyclic, bicyclic, or tricyclic, respectively), wherein 0, 1, 2, 3, or 4 atoms of each ring are substituted by a substituent. Examples of heteroaryl groups include pyridyl, furyl or furanyl, imidazolyl, benzimidazolyl, pyrimidinyl, thiophenyl or thienyl, quinolinyl, indolyl, thiazolyl, and the like.
[0142] The term "heteroarylalkyl" or the term "heteroaralkyl" refers to an alkyl substituted with a heteroaryl. The term "heteroarylalkoxy" refers to an alkoxy substituted with heteroaryl.
[0143] The term "heteroarylalkyl" or the term "heteroaralkyl" refers to an alkyl substituted with a heteroaryl. The term "heteroarylalkoxy" refers to an alkoxy substituted with heteroaryl.
[0144] The term "heterocyclyl" refers to a nonaromatic 5-8 membered monocyclic, 8-12 membered bicyclic, or 11-14 membered tricyclic ring system having 1-3 heteroatoms if monocyclic, 1-6 heteroatoms if bicyclic, or 1-9 heteroatoms if tricyclic, said heteroatoms selected from O, N, or S (e.g., carbon atoms and 1-3, 1-6, or 1-9 heteroatoms of O, N, or S if monocyclic, bicyclic, or tricyclic, respectively), wherein 0, 1, 2 or 3 atoms of each ring are substituted by a substituent. Examples of heterocyclyl groups include piperazinyl, pyrrolidinyl, dioxanyl, morpholinyl, tetrahydrofuranyl, and the like.
[0145] The term "substituent" refers to a group replacing a second atom or group such as a hydrogen atom on any molecule, compound or moiety. Suitable substituents include, without limitation, halo, hydroxy, mercapto, oxo, nitro, haloalkyl, alkyl, alkaryl, aryl, aralkyl, alkoxy, thioalkoxy, aryloxy, amino, alkoxycarbonyl, amido, carboxy, alkanesulfonyl, alkylcarbonyl, and cyano groups.
[0146] In some embodiments, the compounds disclosed herein contain one or more asymmetric centers and thus occur as racemates and racemic mixtures, single enantiomers, individual diastereomers and diastereomeric mixtures. All such isomeric forms of these compounds are included unless expressly provided otherwise. In some embodiments, the compounds disclosed herein are also represented in multiple tautomeric forms, in such instances, the compounds include all tautomeric forms of the compounds described herein (e.g., if alkylation of a ring system results in alkylation at multiple sites, the disclosure includes all such reaction products). All such isomeric forms of such compounds are included unless expressly provided otherwise. All crystal forms of the compounds described herein are included unless expressly provided otherwise.
[0147] As used herein, the terms "increase" and "decrease" mean, respectively, to cause a statistically significantly (i.e., p<0.1) increase or decrease of at least 5%.
[0148] As used herein, the recitation of a numerical range for a variable is intended to convey that the variable is equal to any of the values within that range. Thus, for a variable which is inherently discrete, the variable is equal to any integer value within the numerical range, including the end-points of the range. Similarly, for a variable which is inherently continuous, the variable is equal to any real value within the numerical range, including the end-points of the range. As an example, and without limitation, a variable which is described as having values between 0 and 2 takes the values 0, 1 or 2 if the variable is inherently discrete, and takes the values 0.0, 0.1, 0.01, 0.001, or any other real values .gtoreq.0 and .ltoreq.2 if the variable is inherently continuous.
[0149] As used herein, unless specifically indicated otherwise, the word "or" is used in the inclusive sense of "and/or" and not the exclusive sense of "either/or."
[0150] The term "on average" represents the mean value derived from performing at least three independent replicates for each data point.
[0151] The term "biological activity" encompasses structural and functional properties of a macrocycle. Biological activity is, for example, structural stability, alpha-helicity, affinity for a target, resistance to proteolytic degradation, cell penetrability, intracellular stability, in vivo stability, or any combination thereof.
[0152] The term "binding affinity" refers to the strength of a binding interaction, for example between a peptidomimetic macrocycle and a target. Binding affinity can be expressed, for example, as an equilibrium dissociation constant ("K.sub.D"), which is expressed in units which are a measure of concentration (e.g. M, mM, .mu.M, nM etc). Numerically, binding affinity and K.sub.D values vary inversely, such that a lower binding affinity corresponds to a higher K.sub.D value, while a higher binding affinity corresponds to a lower K.sub.D value. Where high binding affinity is desirable, "improved" binding affinity refers to higher binding affinity and therefore lower K.sub.D values.
[0153] The term "in vitro efficacy" refers to the extent to which a test compound, such as a peptidomimetic macrocycle, produces a beneficial result in an in vitro test system or assay. In vitro efficacy can be measured, for example, as an "IC.sub.50" or "EC.sub.50" value, which represents the concentration of the test compound which produces 50% of the maximal effect in the test system.
[0154] The term "ratio of in vitro efficacies" or "in vitro efficacy ratio" refers to the ratio of IC.sub.50 or EC.sub.50 values from a first assay (the numerator) versus a second assay (the denominator). Consequently, an improved in vitro efficacy ratio for Assay 1 versus Assay 2 refers to a lower value for the ratio expressed as IC.sub.50(Assay 1)/IC.sub.50(Assay 2) or alternatively as EC.sub.50(Assay 1)/EC.sub.50(Assay 2). This concept can also be characterized as "improved selectivity" in Assay 1 versus Assay 2, which can be due either to a decrease in the IC.sub.50 or EC.sub.50 value for Target 1 or an increase in the value for the IC.sub.50 or EC.sub.50 value for Target 2.
[0155] The term "solid tumor" or "solid cancer" as used herein refers to tumors that usually do not contain cysts or liquid areas. Solid tumors as used herein include sarcomas, carcinomas and lymphomas. In various embodiments leukemia (cancer of blood) is not solid tumor.
[0156] The term "adverse event" (AE) as used herein includes any noxious, pathological, or unintended change in anatomical, physiological, or metabolic functions as indicated by physical signs, symptoms, and/or laboratory changes occurring in any phase of the clinical study whether or not temporally associated with the administration of study medication and whether or not considered related to the study medication. This definition includes an exacerbation of pre-existing medical conditions or events, intercurrent illnesses, hypersensitivity reactions, drug interactions, or clinically significant laboratory findings. An AE does not include the following: (i) medical or surgical procedures, e.g., tooth extraction, transfusion, surgery (The medical condition that leads to the procedure is to be recorded as an AE); (ii) pre-existing conditions or procedures present or detected at the start of the study that do not worsen; (iii) hospitalization for elective surgeries or for other situations in which an untoward medical event has not occurred; (iv) abnormal laboratory value, unless it is clinically significant according to the Investigator, requires intervention, or results in a delay, discontinuation or change in the dose of study drug; (v) overdose of study drug or concomitant medication unaccompanied by signs/symptoms; if sign/symptoms occur, the final diagnosis should be recorded as an AE; (vi) pregnancy by itself, unless a complication occurs during pregnancy leading to hospitalization; in this case, the medical condition that leads to the hospitalization is to be recorded as the AE; and (vii) significant worsening of the disease under investigation which is captured as an efficacy parameter in this study and, thus, is not recorded as an AE.
[0157] The term serious adverse event (SAE) as used herein refers to an adverse event that results in any of the following outcomes: (i) death; (ii) life-threatening adverse experience (i.e., immediate risk of death from the event as it occurred; this does not include an adverse event that, had it occurred in a more serious form, might have caused death); (iii) persistent or significant disability/incapacitation; (iv) hospitalization or prolongation of existing hospitalization; and (v) congenital anomaly/birth defect. Important medical events that may not result in death, be life-threatening, or require hospitalization can be considered serious when, based on medical judgment, they can jeopardize the patient or may require medical or surgical intervention to prevent one of the outcomes listed in this definition. Hospitalizations due to the underlying disease will not be reported as an SAE unless there is reason to suspect a causal relationship with the study drug.
[0158] An AE or suspected adverse reaction is considered "unexpected" (referred to as Unexpected Adverse Event (UAE) if it is not listed in the peptidomimetic macrocycle Investigator's Brochure or is not listed at the specificity or severity that has been observed; or, is not consistent with the risk information described in the protocol or elsewhere. For example, under this definition, hepatic necrosis would be unexpected (by virtue of greater severity) if the Investigator's Brochure referred only to elevated hepatic enzymes or hepatitis. Similarly, cerebral thromboembolism and cerebral vasculitis would be unexpected (by virtue of greater specificity) if the Investigator's Brochure listed only cerebral vascular accidents. "Unexpected," as used in this definition, also refers to AEs or suspected adverse reactions that are mentioned in the Investigator's Brochure as occurring with a class of drugs or as anticipated from the pharmacological properties of the peptidomimetic macrocycle but are not specifically mentioned as occurring with the peptidomimetic macrocycle.
[0159] A "Dose-Limiting Toxicity" (DLT) as used herein is defined as any Grade .gtoreq.3 AE that is considered to be possibly, probably, or definitely related to the study drug, with the following exceptions: (1) for nausea, emesis, diarrhea, rash, or mucositis, only Grade .gtoreq.3 AE that do not respond within 48 hours to standard supportive/pharmacological treatment will be considered DLT; (2) for electrolyte imbalances, only Grade .gtoreq.3 AE that do not respond to correction within 24 hours will be considered DLT. In addition, specific hematologic DLTs are defined as: [0160] (i) Thrombocytopenia--Grade 4 of any duration, Grade 3 for .gtoreq.7 days, or Grade 3 associated with clinically significant bleeding; [0161] (ii) Neutropenia--Grade 4 for .gtoreq.3 days, or any Grade .gtoreq.3 febrile neutropenia
[0162] The above criteria can be used to make individual patient determinations regarding dose reductions, interruptions or discontinuation throughout the course of the trial, but DLTs occurring during Cycle 1 will be used to inform safety and tolerability assessments for dose escalation decisions.
[0163] The "Maximum Tolerated Dose" (MTD) as used herein is defined as the dose at which .ltoreq.1 of 6 patients experiences a treatment-related toxicity that qualifies as a DLT, with the next higher dose having .gtoreq.2 of up to 6 patients experiencing a DLT. The MTD may not be established until all patients enrolled in the cohort have completed Cycle 1, discontinued treatment or had a dose reduction. Previously established tolerability of a dose level will be reevaluated if DLTs are observed in later cycles.
[0164] "Measurable disease" (MD) as used herein is defined by the presence of at least one measurable lesion.
[0165] Measurable lesions are defined as those that can be accurately measured in at least one dimension [longest diameter (LD) in the plane of measurement to be recorded] with a minimum size of: 10 mm by CT scan (CT scan slice thickness no greater than 5 mm), 10 mm caliper measurement by clinical exam (lesions which cannot be accurately measured with calipers can be recorded as non-measurable), or 20 mm by chest x-ray.
[0166] "Malignant lymph nodes" is to be considered pathologically enlarged and measurable if the lymph node is .gtoreq.15 mm in short axis when assessed by CT scan (CT scan slice thickness no greater than 5 mm).
[0167] "Non-measurable Disease" as used herein include all other lesions (or sites of disease) that are not measurable, including small lesions (longest diameter <10 mm or pathological lymph nodes with .gtoreq.10 to <15 mm short axis) are considered non-measurable disease. Lesions considered truly non-measurable include: leptomeningeal disease, ascites, pleural/pericardial effusions, lymphangitis cutis/pulmonis, inflammatory breast disease, abdominal masses/abdominal organomegaly identified by physical exam and not followed by CT or MRI.
[0168] "Target lesions" as used herein include all measurable lesions up to a maximum of two lesions per organ and five lesions in total, representative of all involved organs identified as target lesions and recorded and measured at baseline. Target lesions can be selected on the basis of their size (lesions with the longest diameter) and their suitability for accurate repeated measurements (either by imaging techniques or clinically). A sum of the diameters (longest for non-nodal lesions, short axis for nodal lesions) for all target lesions can be calculated and reported as the baseline sum diameters. The baseline sum diameters can be used as reference by which to characterize the objective tumor response.
[0169] "Non-target lesions" as used herein include all other lesions (or sites of disease) including pathological lymph nodes that are not target lesions. Non-target lesions can be identified as non-target lesions and can also be recorded at baseline. Measurements of these lesions may not be required and these lesions can be followed as "present," "absent," or in rare cases "unequivocal progression." In addition, it can be possible to record multiple non-target lesions involving the same organ as a single item on the case report form (e.g., `multiple enlarged pelvic lymph nodes` or `multiple liver metastases`).
[0170] "Complete response" (CR) as used herein is defined as the disappearance of all target lesions. Any pathological lymph nodes (whether target or non-target) must have reduction in short axis to <10 mm.
[0171] "Partial response (PR)" as used herein is defined as at least a 30% decrease in the sum of diameters of target lesions, taking as reference the baseline sum diameters.
[0172] "Progressive disease (PD)" as used herein is defined as at least a 20% increase in the sum of diameters of target lesions, taking as reference the smallest sum on study (this includes the baseline sum if that is the smallest). In addition to the relative increase of 20%, the sum must also demonstrate an absolute increase of at least 5 mm. The appearance of one or more new lesions can also be considered progression.
[0173] "Stable disease" (SD) as used herein is defined as neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as reference the smallest sum diameters while on study.
[0174] The term "subject" or "patient" encompasses mammals and non-mammals. Examples of mammals include, but are not limited to, humans; non-human primates such as chimpanzees, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like. Examples of non-mammals include, but are not limited to, birds, fish and the like. In one embodiment of the methods and compositions provided herein, the mammal is a human.
[0175] The term "haploinsufficient" means a condition that occurs when a diploid organism has only a single functional copy of a gene (with the other copy inactivated by mutation) and the single functional copy does not produce enough of a gene product (typically a protein) to bring about a wild-type condition, leading to an abnormal or diseased state.
[0176] The term "silent mutation" as used herein silent mutation is a type of mutation in the coding region of a gene that doesn't actually change the amino acid sequence of the protein that is made.
[0177] The details of one or more particular embodiments of the invention are set forth in the accompanying drawings and the description below. Other features, objects, and advantages of the invention will be apparent from the description and drawings, and from the claims.
Overview
[0178] In one aspect, the disclosure provides a method of treating solid tumor in a subject. For example, the methods disclosed herein can be used for treating solid tumors that are not p53 negative. In some cases the methods disclosed herein can be used for treating solid tumor which has been determined to lack a p53 deactivating mutation. The methods of the disclosure can also be used to treat solid tumors that express gain of function mutant p53 i.e. a super apoptotic p53. In other examples, the methods of the disclosure are useful in treating solid tumors, wherein the solid tumor expresses p53 with a partial loss of function mutation, p53 with a copy loss mutation, or p53 with one or more silent mutations. In some example, the solid tumor expresses p53 with a copy loss mutation and a deactivating mutation.
[0179] The method comprises administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycle disrupts the interaction between p53 and MDM2 and MDMX.
[0180] In another aspect, the disclosure provides a method of treating solid tumor in a subject expressing wild type p53. The method comprises administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycle disrupts the interaction between p53 and MDM2 and MDMX.
[0181] In some embodiments the subject treated by the methods disclosed herein is a human. In some embodiments, a subject treated in accordance with the methods provided herein is a human who has or is diagnosed with solid tumor lacking p53 deactivating mutation and/or expressing wild type p53. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human predisposed or susceptible to solid tumor lacking p53 deactivating mutation and/or expressing wild type p53. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human at risk of developing solid tumor lacking p53 deactivating mutation and/or expressing wild type p53. A p53 deactivating mutation in some examples can be a mutation in DNA-binding domain of the p53 protein. In some examples the p53 deactivating mutation can be a missense mutation. In various examples, the solid tumor can be determined to lack one or more p53 deactivating mutations selected from mutations at one or more of residues R175, G245, R248, R249, R273, and R282. The lack of p53 deactivating mutation and/or the presence of wild type p53 in the solid tumor can be determined by any suitable method known in art, for example by sequencing, array based testing, RNA analysis and amplifications methods like PCR.
[0182] In certain embodiments, the human subject is refractory and/or intolerant to one or more other standard treatment of the solid tumor known in art. In some embodiments, the human subject has had at least one unsuccessful prior treatment and/or therapy of the solid tumor.
[0183] In some embodiments, the subject treated in accordance with the methods of the disclosure is a human who has or is diagnosed with solid tumor that is not p53 negative. In some embodiments, the subject treated in accordance with the methods of the disclosure is a human who has or is diagnosed with solid tumor that express gain of function mutant p53 i.e. a super apoptotic p53. In some embodiments, the subject treated in accordance with the methods of the disclosure is a human who has or is diagnosed with solid tumor that expresses p53 with a partial loss of function mutation. In some embodiments, the subject treated in accordance with the methods provided herein is a human who has or is diagnosed with solid tumor that expresses p53 with a copy loss mutation. In some embodiments, the subject treated in accordance with the methods provided herein is a human who has or is diagnosed with solid tumor that expresses p53 with one or more silent mutations. In some embodiments, the subject treated in accordance with the methods provided herein is a human who has or is diagnosed with solid tumor that expresses p53 with a copy loss mutation and a deactivating mutation.
[0184] In some embodiments, the methods for treating solid tumor provided herein inhibit, reduce, diminish, arrest, or stabilize a tumor associated with the solid tumor. In some embodiments, the methods for treating solid tumor provided herein inhibit, reduce, diminish, arrest, or stabilize the blood flow, metabolism, or edema in a tumor associated with the solid tumor or one or more symptoms thereof. In some embodiments, the methods for treating solid tumor provided herein cause the regression of a tumor, tumor blood flow, tumor metabolism, or peritumor edema, and/or one or more symptoms associated with the solid tumor. In some embodiments, the methods for treating solid tumor provided herein maintain the size of the tumor so that it does not increase, or so that it increases by less than the increase of a tumor after administration of a standard therapy as measured by conventional methods available to one of skill in the art, such as ultrasound, CT Scan, MRI, dynamic contrast-enhanced MRI, or PET Scan. In specific embodiments, the methods for treating solid tumor provided herein decrease tumor size. In some embodiments, the methods for treating solid tumor provided herein reduce the formation of a tumor. In certain embodiments, the methods for treating solid tumor provided herein eradicate, remove, or control primary, regional and/or metastatic tumors associated with the solid tumor. In some embodiments, the methods for treating solid tumor provided herein decrease the number or size of metastases associated with the solid tumor. In some embodiments, the methods for treating solid tumor provided herein result in complete response to the treatment. In some embodiments, the methods for treating solid tumor provided herein result in partial response to the treatment. In some embodiments, the solid tumor treated by the methods disclosed herein is a stable disease. In some embodiments, the solid tumor treated by the methods disclosed herein is a progressive disease.
[0185] Solid tumor cancers that can be treated by the methods provided herein include, but are not limited to, sarcomas, carcinomas, and lymphomas. In specific embodiments, solid tumors that can be treated in accordance with the methods described include, but are not limited to, cancer of the breast, liver, neuroblastoma, head, neck, eye, mouth, throat, esophagus, esophagus, chest, bone, lung, kidney, colon, rectum or other gastrointestinal tract organs, stomach, spleen, skeletal muscle, subcutaneous tissue, prostate, breast, ovaries, testicles or other reproductive organs, skin, thyroid, blood, lymph nodes, kidney, liver, pancreas, and brain or central nervous system.
[0186] The peptidomimetic macrocycle can be any cross-linked peptide, i.e. any peptide that comprises at least one macrocycle-forming linker which forms a macrocycle between a first amino acid residue (or analog) and a second amino acid residue. For example, the peptidomimetic macrocycle can be a peptidomimetic macrocycle capable of binding to the MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycles can be a peptidomimetic macrocycle of Formula I:
##STR00012##
wherein: each of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 is individually an amino acid, wherein at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8) or Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.2 (SEQ ID NO: 9), where each X is an amino acid; each D and E is independently an amino acid; each R.sub.1 and R.sub.2 are independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or at least one of R.sub.1 and R.sub.2 forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each L or L' is independently a macrocycle-forming each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v is an integer from 1-1000, for example 1-500, 1-200, 1-100, 1-50, 1-30, 1-20, or 1-10; and each w is an integer from 0-1000, for example 1-500, 1-200, 1-100, 1-50, 1-30, 1-20, or 1-10.
[0187] Administration of the a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof can be achieved by any suitable means. For example the pharmaceutical composition can be administered parenterally. For example, administration can be intravenous, intra-arterial, intraosseous infusion, intra-muscular, intracerebral, intracerebroventricular, intrathecal, or subcutaneous. In some embodiments administration is performed intravenously.
[0188] In some embodiments, the methods disclosed herein additionally or optionally comprise evaluating the safety and/or tolerability of a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, of the disclosure in subjects with solid tumors determined to lack a p53 deactivating mutation or with solid tumors expressing wild-type (WT) p53 protein.
[0189] Also provided here in are methods to determine the dose limiting toxicities (DLT) and the maximum tolerated dose (MTD) of the peptidomimetic macrocycles disclosed herein in subjects with solid tumors determined to lack a p53 deactivating mutation or with solid tumors expressing wild-type (WT) p53 protein.
[0190] In some embodiments, the methods disclosed herein additionally or optionally comprise the pharmacokinetic(PK) analysis of the peptidomimetic macrocycles and/or its metabolites in blood following single and/or multiple administration of the peptidomimetic macrocycles to the subject.
[0191] In some embodiments, the methods disclosed herein additionally or optionally comprise studying the effect of the peptidomimetic macrocycles on pharmacodynamic biomarkers in tumor biopsy samples (e.g., p21, caspase, MDM2) and blood samples (e.g., macrophage inhibitory cytokine-1 [MIC-1]), and assessing possible correlation between these biomarkers and clinical response.
[0192] In some embodiments, the methods disclosed herein additionally or optionally include steps to assess potential patient biomarkers (e.g., p53 status, MDM2 and MDMX expression levels), the effect of the peptidomimetic macrocycles treatment on these biomarkers, and possible correlation between these biomarkers and clinical response of the peptidomimetic macrocycles.
[0193] Also provided herein are methods to evaluate clinical activity of the peptidomimetic macrocycles in subjects with specific tumor types lacking a p53 deactivating mutation and/or expressing WT p53 in the dose expansion phase.
Compound and Compositions
Peptidomimetic Macrocycles
[0194] In some embodiments, a peptidomimetic macrocycle has the Formula (I):
##STR00013##
wherein: each A, C, and D is independently an amino acid; each B is independently an amino acid,
##STR00014##
[--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each E is independently an amino acid selected from the group consisting of Ala (alanine), D-Ala (D-alanine), Aib (.alpha.-aminoisobutyric acid), Sar (N-methyl glycine), and Ser (serine); each R.sub.3 is independently hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each L and L' is independently a macrocycle-forming linker; each L.sub.3 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v is independently an integer; each w is independently an integer from 3-1000; u is an integer from 1-10; each x, y and z is independently an integer from 0-10; and each n is independently an integer from 1-5.
[0195] In some embodiments, each v and w is independently integers between 1-30. In some embodiments, w is an integer from 3-1000, for example 3-500, 3-200, 3-100, 3-50, 3-30, 3-20, or 3-10. In some embodiments, the sum of x+y+z is 3 or 6. In some embodiments, the sum of x+y+z is 3. In other embodiments, the sum of x+y+z is 6.
[0196] In some embodiments, peptidomimetic macrocycles are also provided of the formula:
##STR00015##
wherein: each of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 is individually an amino acid, wherein at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8), where each X is an amino acid; each D and E is independently an amino acid; each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or at least one of R.sub.1 and R.sub.2 forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each L or L' is independently a macrocycle-forming linker; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; v is an integer from 1-1000, for example 1-500, 1-200, 1-100, 1-50, 1-30, 1-20 or 1-10; and w is an integer from 3-1000, for example 3-500, 3-200, 3-100, 3-50, 3-30, 3-20, or 3-10.
[0197] In some embodiments, each v and w is independently an integer between 1-30. In some embodiments, w is an integer from 3-1000, for example 3-500, 3-200, 3-100, 3-50, 3-30, 3-20, or 3-10. In some embodiments, the sum of x+y+z is 3 or 6. In some embodiments, the sum of x+y+z is 3. In other embodiments, the sum of x+y+z is 6.
[0198] In some embodiments of any of the Formulas described herein, at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8). In other embodiments, at least four of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8). In other embodiments, at least five of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8). In other embodiments, at least six of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8). In other embodiments, at least seven of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Hiss-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.sub.10- -X.sub.11-Ser.sub.12 (SEQ ID NO: 8).
[0199] In some embodiments, a peptidomimetic macrocycle has the Formula:
##STR00016##
wherein: each of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 is individually an amino acid, wherein at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.12 (SEQ ID NO: 9), where each X is an amino acid; each D is independently an amino acid; each E is independently an amino acid, for example an amino acid selected from Ala (alanine), D-Ala (D-alanine), Aib (.alpha.-aminoisobutyric acid), Sar (N-methyl glycine), and Ser (serine); each R.sub.1 and R.sub.2 are independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or at least one of R.sub.1 and R.sub.2 forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each L or L' is independently a macrocycle-forming linker; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; v is an integer from 1-1000, for example 1-500, 1-200, 1-100, 1-50, 1-30, 1-20, or 1-10; w is an integer from 3-1000, for example 3-500, 3-200, 3-100, 3-50, 3-30, 3-20, or 3-10; and.
[0200] In some embodiments of the above Formula, at least three of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp-Ala.sub.8-Gln.sub.9-Leu.sub.10/- Cba.sub.10-X.sub.1-Ala.sub.2 (SEQ ID NO: 9). In other embodiments of the above Formula, at least four of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.12 (SEQ ID NO: 9). In other embodiments of the above Formula, at least five of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.1-Ala.sub.2 (SEQ ID NO: 9). In other embodiments of the above Formula, at least six of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.12 (SEQ ID NO: 9). In other embodiments of the above Formula, at least seven of Xaa.sub.3, Xaa.sub.5, Xaa.sub.6, Xaa.sub.7, Xaa.sub.8, Xaa.sub.9, and Xaa.sub.10 are the same amino acid as the amino acid at the corresponding position of the sequence Phe.sub.3-X.sub.4-Glu.sub.5-Tyr.sub.6-Trp.sub.7-Ala.sub.8-Gln.sub.9-Leu.s- ub.10/Cba.sub.10-X.sub.11-Ala.sub.12 (SEQ ID NO: 9).
[0201] In some embodiments, w is an integer from 3-10, for example 3-6, 3-8, 6-8, or 6-10. In some embodiments, w is 3. In other embodiments, w is 6. In some embodiments, v is an integer from 1-10, for example 2-5. In some embodiments, v is 2.
[0202] In some embodiments, a peptidomimetic macrocycle of Formula (I) has Formula (Ic):
##STR00017##
wherein: each A, C, D, and E is independently a natural or non-natural amino acid; each B is independently a natural or non-natural amino acid, amino acid analog,
##STR00018##
[--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each L is independently a macrocycle-forming linker; each L' is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene, each being optionally substituted with R.sub.5, or a bond, or together with R.sub.1 and the atom to which both R.sub.1 and L' are bound forms a ring; each L'' is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene, each being optionally substituted with R.sub.5, or a bond, or together with R.sub.2 and the atom to which both R.sub.2 and L'' are bound forms a ring; each R.sub.1 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-, or together with L' and the atom to which both R, and L' are bound forms a ring; each R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-, or together with L'' and the atom to which both R.sub.2 and L'' are bound forms a ring; R.sub.3 is hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5; each L.sub.3 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, heteroarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each n is an integer from 1-5; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v and w is independently an integer from 1-1000, for example 1-500, 1-200, 1-100, 1-50, 1-40, 1-25, 1-20, 1-15, or 1-10; and each u, x, y and z is independently an integer from 0-10.
[0203] In some embodiments, the peptidomimetic macrocycles have the Formula (I):
##STR00019##
wherein: each A, C, D, and E is independently a natural or non-natural amino acid; each B is independently a natural or non-natural amino acid, amino acid analog,
##STR00020##
[--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; each R.sub.3 is independently hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5; each L is independently a macrocycle-forming linker of the formula
##STR00021##
each L.sub.1, L.sub.2 and L.sub.3 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, heteroarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.5 is independently halogen, alkyl, --OR.sub.6, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5, or part of a cyclic structure with a D residue; each R.sub.8 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, aryl, or heteroaryl, optionally substituted with R.sub.5, or part of a cyclic structure with an E residue; each v and w is independently an integer from 1-1000; each u, x, y and z is independently integers from 0-10; and n is an integer from 1-5.
[0204] In an embodiment of any of the Formulas described herein, of the macrocycle-forming linker (L) has a formula -L.sub.1-L.sub.2-, wherein
each L.sub.1 and L.sub.2 are independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is independently O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.3 is independently hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R; and n is an integer from 1-5.
[0205] In some embodiments in the Formulas described herein, L (or L') is a macrocycle-forming linker of the formula
##STR00022##
[0206] Exemplary embodiments of such macrocycle-forming linkers L are shown below.
##STR00023## ##STR00024## ##STR00025## ##STR00026## ##STR00027## ##STR00028## ##STR00029## ##STR00030## ##STR00031##
[0207] In an embodiment of any of the Formulas described herein, L.sub.1 and L.sub.2, either alone or in combination, form a triazole or a thioether.
[0208] In an embodiment of any of the Formulas described herein, L.sub.1 and L.sub.2, either alone or in combination, do not form a triazole or a thioether.
[0209] In one example, at least one of R.sub.1 and R.sub.2 is alkyl, unsubstituted or substituted with halo-. In another example, each R, and R.sub.2 are independently alkyl, unsubstituted or substituted with halo-. In some embodiments, at least one of R, and R.sub.2 is methyl. In other embodiments, R.sub.1 and R.sub.2 are methyl.
[0210] In some embodiments, x+y+z is at least 3. In other embodiments, x+y+z is 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. In some embodiments, the sum of x+y+z is 3 or 6. In some embodiments, the sum of x+y+z is 3. In other embodiments, the sum of x+y+z is 6. Each occurrence of A, B, C, D or E in a macrocycle or macrocycle precursor is independently selected. For example, a sequence represented by the formula [A].sub.x, when x is 3, encompasses embodiments where the amino acids are not identical, e.g. Gln-Asp-Ala as well as embodiments where the amino acids are identical, e.g. Gln-Gln-Gln. This applies for any value of x, y, or z in the indicated ranges. Similarly, when u is greater than 1, each compound can encompass peptidomimetic macrocycles which are the same or different. For example, a compound can comprise peptidomimetic macrocycles comprising different linker lengths or chemical compositions.
[0211] In some embodiments, the peptidomimetic macrocycle comprises a secondary structure which is an .alpha.-helix and R.sub.5 is --H, allowing intrahelical hydrogen bonding. In some embodiments, at least one of A, B, C, D or E is an .alpha.,.alpha.-disubstituted amino acid. In one example, B is an .alpha.,.alpha.-disubstituted amino acid. For instance, at least one of A, B, C, D or E is 2-aminoisobutyric acid. In other embodiments, at least one of A, B, C, D or E is
##STR00032##
[0212] In other embodiments, the length of the macrocycle-forming linker L as measured from a first C.alpha. to a second C.alpha. is selected to stabilize a desired secondary peptide structure, such as an .alpha.-helix formed by residues of the peptidomimetic macrocycle including, but not necessarily limited to, those between the first C.alpha. to a second C.alpha..
[0213] In one embodiment, the peptidomimetic macrocycle of Formula (I) is:
##STR00033##
wherein each R.sub.1 and R.sub.2 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-.
[0214] In related embodiments, the peptidomimetic macrocycle of Formula (I) is:
##STR00034##
wherein each R.sub.1' and R.sub.2' is independently an amino acid.
[0215] In other embodiments, the peptidomimetic macrocycle of Formula (I) is a compound of any of the formulas shown below:
##STR00035## ##STR00036##
wherein "AA" represents any natural or non-natural amino acid side chain and "" is [D].sub.v, [E].sub.w as defined above, and n is an integer between 0 and 20, 50, 100, 200, 300, 400 or 500. In some embodiments, n is 0. In other embodiments, n is less than 50.
[0216] Exemplary embodiments of the macrocycle-forming linker L are shown below.
##STR00037##
[0217] In other embodiments, D and/or E in the compound of Formula I are further modified in order to facilitate cellular uptake. In some embodiments, lipidating or PEGylating a peptidomimetic macrocycle facilitates cellular uptake, increases bioavailability, increases blood circulation, alters pharmacokinetics, decreases immunogenicity and/or decreases the needed frequency of administration.
[0218] In other embodiments, at least one of [D] and [E] in the compound of Formula I represents a moiety comprising an additional macrocycle-forming linker such that the peptidomimetic macrocycle comprises at least two macrocycle-forming linkers. In a specific embodiment, a peptidomimetic macrocycle comprises two macrocycle-forming linkers. In an embodiment, u is 2.
[0219] In some embodiments, any of the macrocycle-forming linkers described herein can be used in any combination with any of the sequences shown in Table 3, Table 3a, Table 3b, or Table 3c and also with any of the R-substituents indicated herein.
[0220] In some embodiments, the peptidomimetic macrocycle comprises at least one .alpha.-helix motif. For example, A, B and/or C in the compound of Formula I include one or more .alpha.-helices. As a general matter, .alpha.-helices include between 3 and 4 amino acid residues per turn. In some embodiments, the .alpha.-helix of the peptidomimetic macrocycle includes 1 to 5 turns and, therefore, 3 to 20 amino acid residues. In specific embodiments, the .alpha.-helix includes 1 turn, 2 turns, 3 turns, 4 turns, or 5 turns. In some embodiments, the macrocycle-forming linker stabilizes an .alpha.-helix motif included within the peptidomimetic macrocycle. Thus, in some embodiments, the length of the macrocycle-forming linker L from a first C.alpha. to a second C.alpha. is selected to increase the stability of an .alpha.-helix. In some embodiments, the macrocycle-forming linker spans from 1 turn to 5 turns of the .alpha.-helix. In some embodiments, the macrocycle-forming linker spans approximately 1 turn, 2 turns, 3 turns, 4 turns, or 5 turns of the .alpha.-helix. In some embodiments, the length of the macrocycle-forming linker is approximately 5 .ANG. to 9 .ANG. per turn of the .alpha.-helix, or approximately 6 .ANG. to 8 .ANG. per turn of the .alpha.-helix. Where the macrocycle-forming linker spans approximately 1 turn of an .alpha.-helix, the length is equal to approximately 5 carbon-carbon bonds to 13 carbon-carbon bonds, approximately 7 carbon-carbon bonds to 11 carbon-carbon bonds, or approximately 9 carbon-carbon bonds. Where the macrocycle-forming linker spans approximately 2 turns of an .alpha.-helix, the length is equal to approximately 8 carbon-carbon bonds to 16 carbon-carbon bonds, approximately 10 carbon-carbon bonds to 14 carbon-carbon bonds, or approximately 12 carbon-carbon bonds. Where the macrocycle-forming linker spans approximately 3 turns of an .alpha.-helix, the length is equal to approximately 14 carbon-carbon bonds to 22 carbon-carbon bonds, approximately 16 carbon-carbon bonds to 20 carbon-carbon bonds, or approximately 18 carbon-carbon bonds. Where the macrocycle-forming linker spans approximately 4 turns of an .alpha.-helix, the length is equal to approximately 20 carbon-carbon bonds to 28 carbon-carbon bonds, approximately 22 carbon-carbon bonds to 26 carbon-carbon bonds, or approximately 24 carbon-carbon bonds. Where the macrocycle-forming linker spans approximately 5 turns of an .alpha.-helix, the length is equal to approximately 26 carbon-carbon bonds to 34 carbon-carbon bonds, approximately 28 carbon-carbon bonds to 32 carbon-carbon bonds, or approximately 30 carbon-carbon bonds. Where the macrocycle-forming linker spans approximately 1 turn of an .alpha.-helix, the linkage contains approximately 4 atoms to 12 atoms, approximately 6 atoms to 10 atoms, or approximately 8 atoms. Where the macrocycle-forming linker spans approximately 2 turns of the .alpha.-helix, the linkage contains approximately 7 atoms to 15 atoms, approximately 9 atoms to 13 atoms, or approximately 11 atoms. Where the macrocycle-forming linker spans approximately 3 turns of the .alpha.-helix, the linkage contains approximately 13 atoms to 21 atoms, approximately 15 atoms to 19 atoms, or approximately 17 atoms. Where the macrocycle-forming linker spans approximately 4 turns of the .alpha.-helix, the linkage contains approximately 19 atoms to 27 atoms, approximately 21 atoms to 25 atoms, or approximately 23 atoms. Where the macrocycle-forming linker spans approximately 5 turns of the .alpha.-helix, the linkage contains approximately 25 atoms to 33 atoms, approximately 27 atoms to 31 atoms, or approximately 29 atoms. Where the macrocycle-forming linker spans approximately 1 turn of the .alpha.-helix, the resulting macrocycle forms a ring containing approximately 17 members to 25 members, approximately 19 members to 23 members, or approximately 21 members. Where the macrocycle-forming linker spans approximately 2 turns of the .alpha.-helix, the resulting macrocycle forms a ring containing approximately 29 members to 37 members, approximately 31 members to 35 members, or approximately 33 members. Where the macrocycle-forming linker spans approximately 3 turns of the .alpha.-helix, the resulting macrocycle forms a ring containing approximately 44 members to 52 members, approximately 46 members to 50 members, or approximately 48 members. Where the macrocycle-forming linker spans approximately 4 turns of the .alpha.-helix, the resulting macrocycle forms a ring containing approximately 59 members to 67 members, approximately 61 members to 65 members, or approximately 63 members. Where the macrocycle-forming linker spans approximately 5 turns of the .alpha.-helix, the resulting macrocycle forms a ring containing approximately 74 members to 82 members, approximately 76 members to 80 members, or approximately 78 members.
[0221] In other embodiments, provided are peptidomimetic macrocycles of Formula (IV) or (IVa):
##STR00038##
wherein: each A, C, D, and E is independently a natural or non-natural amino acid, and the terminal D and E independently optionally include a capping group; each B is a natural or non-natural amino acid, amino acid analog,
##STR00039##
[--NH-L.sub.3-CO--], [--NH-L.sub.3-SO.sub.2--], or [--NH-L.sub.3-]; each R, and R.sub.2 are independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, cycloalkylalkyl, heteroalkyl, or heterocycloalkyl, unsubstituted or substituted with halo-; or at least one of R, and R.sub.2 forms a macrocycle-forming linker L' connected to the alpha position of one of said D or E amino acids; each R.sub.3 is hydrogen, alkyl, alkenyl, alkynyl, arylalkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, cycloalkylalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; each L is a macrocycle-forming linker of the formula -L.sub.1-L.sub.2-; each L.sub.1, L.sub.2 and L.sub.3 are independently alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, cycloarylene, heterocycloarylene, or [--R.sub.4--K--R.sub.4--].sub.n, each being optionally substituted with R.sub.5; each R.sub.4 is alkylene, alkenylene, alkynylene, heteroalkylene, cycloalkylene, heterocycloalkylene, arylene, or heteroarylene; each K is O, S, SO, SO.sub.2, CO, CO.sub.2, or CONR.sub.3; each R.sub.5 is independently halogen, alkyl, --OR, --N(R.sub.6).sub.2, --SR.sub.6, --SOR.sub.6, --SO.sub.2R.sub.6, --CO.sub.2R.sub.6, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.6 is independently --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkylalkyl, heterocycloalkyl, a fluorescent moiety, a radioisotope or a therapeutic agent; each R.sub.7 is --H, alkyl, alkenyl, alkynyl, arylalkyl, cycloalkyl, heteroalkyl, cycloalkylalkyl, heterocycloalkyl, cycloaryl, or heterocycloaryl, optionally substituted with R.sub.5; each v and w are independently integers from 1-1000; u is an integer from 1-10; each x, y and z are independently integers from 0-10; and each n is an integer from 1-5.
[0222] In one example, L.sub.1 and L.sub.2, either alone or in combination, do not form a triazole or a thioether.
[0223] In one example, at least one of R.sub.1 and R.sub.2 is alkyl, unsubstituted or substituted with halo-. In another example, both R.sub.1 and R.sub.2 are independently alkyl, unsubstituted or substituted with halo-. In some embodiments, at least one of R.sub.1 and R.sub.2 is methyl. In other embodiments, R.sub.1 and R.sub.2 are methyl.
[0224] In some embodiments, x+y+z is at least 1. In other embodiments, x+y+z is at least 2. In other embodiments, x+y+z is 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. Each occurrence of A, B, C, D or E in a macrocycle or macrocycle precursor is independently selected. For example, a sequence represented by the formula [A].sub.x, when x is 3, encompasses embodiments where the amino acids are not identical, e.g. Gln-Asp-Ala as well as embodiments where the amino acids are identical, e.g. Gln-Gln-Gln. This applies for any value of x, y, or z in the indicated ranges.
[0225] In some embodiments, the peptidomimetic macrocycle comprises a secondary structure which is an .alpha.-helix and R.sub.8 is --H, allowing intrahelical hydrogen bonding. In some embodiments, at least one of A, B, C, D or E is an .alpha.,.alpha.-disubstituted amino acid. In one example, B is an .alpha.,.alpha.-disubstituted amino acid. For instance, at least one of A, B, C, D or E is 2-aminoisobutyric acid. In other embodiments, at least one of A, B, C, D or E is
##STR00040##
[0226] In other embodiments, the length of the macrocycle-forming linker L as measured from a first C.alpha. to a second C.alpha. is selected to stabilize a desired secondary peptide structure, such as an .alpha.-helix formed by residues of the peptidomimetic macrocycle including, but not necessarily limited to, those between the first C.alpha. to a second C.alpha..
[0227] Exemplary embodiments of the macrocycle-forming linker -L.sub.1-L.sub.2-are shown below.
##STR00041##
[0228] Unless otherwise stated, any compounds (including peptidomimetic macrocycles, macrocycle precursors, and other compositions) are also meant to encompass compounds which differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the described structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by .sup.13C- or .sup.14C-enriched carbon are within the scope of this disclosure.
[0229] In some embodiments, the compounds disclosed herein can contain unnatural proportions of atomic isotopes at one or more of atoms that constitute such compounds. For example, the compounds can be radiolabeled with radioactive isotopes, such as for example tritium (.sup.3H), iodine-125 (.sup.125I) or carbon-14 (.sup.14C). In other embodiments, one or more carbon atoms is replaced with a silicon atom. All isotopic variations of the compounds disclosed herein, whether radioactive or not, are contemplated herein.
[0230] The circulating half-life of the peptidomimetic macrocycles in human blood can be about 1-24 h. For example the circulating half-life of the peptidomimetic macrocycles in human blood can be about 2-24 h, 4-24 h, 6-24 h, 8-24 h, 10-24 h, 12-24 h, 14-24 h, 16-24 h, 18-24 h, 20-24 h, 22-24 h, 1-20 h, 4-20 h, 6-20 h, 8-20 h, 10-20 h, 12-20 h, 14-20 h, 16-20 h, 18-20 h, 1-16 h, 4-16 h, 6-16 h, 8-16 h, 10-16 h, 12-16 h, 14-16 h, 1-12 h, 4-12 h, 6-12 h, 8-12 h, 10-12 h, 1-8 h, 4-8 h, 6-8 h, or 1-4 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood can be bout 1-12 h, for example about 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h, 11 h, or 12 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 2 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 4 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 6 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 8 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 10 h.
[0231] The half-life of the peptidomimetic macrocycles in biological tissue can be about 1-24 h. For example the circulating half-life of the peptidomimetic macrocycles in human blood can be about 1-24 h, 5-24 h, 10-24 h, 15-24 h, 20-24 h, 1-22 h, 5-22 h, 10-22 h, 15-22 h, 20-22 h, 1-20 h, 5-20 h, 15-20 h, 1-18 h, 5-18 h, 10-18 h, 15-18 h, 1-16 h, 5-16 h, 10-16 h, 15-16 h, 1-14 h, 5-14 h, 10-14 h, 1-12 h, 5-12 h, 10-12 h, 1-10 h, 5-10 h, 1-8 h, 5-8 h, 1-6 h, 5-6 h, or 1-4 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood can be bout 5-20 h, for example about 5 h, 6 h, 7 h, 8 h, 9 h, 10 h, 11 h, 12 h, 13 h, 14 h, 15 h, 16 h, 17 h, 18 h, 19 h or 20 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 2 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 4 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 6 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 8 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 10 h.
[0232] The circulating half-life of the peptidomimetic macrocycles in human blood can be greater than, equal to, or less than the half-life of the peptidomimetic macrocycles in biological tissue. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood can be greater than the half-life of the peptidomimetic macrocycles in biological tissue. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood can be equal to the half-life of the peptidomimetic macrocycles in biological tissue. In some examples, the half-life of the peptidomimetic macrocycles in biological tissue is greater than the circulating half-life of the peptidomimetic macrocycles in human blood. This can facilitate administration of the peptidomimetic macrocycles at a lower dose and/or at lower frequency. In some embodiments, the half-life of the peptidomimetic macrocycles in biological tissue is at least 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h, 11 h, or 12 h greater than the than the circulating half-life of the peptidomimetic macrocycles in human blood. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 4 h and the half-life of the in biological tissue is about 10 h. In some examples, the circulating half-life of the peptidomimetic macrocycles in human blood is about 6 h and the half-life of the in biological tissue is about 10 h.
Preparation of Peptidomimetic Macrocycles
[0233] Peptidomimetic macrocycles can be prepared by any of a variety of methods known in the art. For example, any of the residues indicated by "$" or "$r8" in Table 3, Table 3a, Table 3b, or Table 3c can be substituted with a residue capable of forming a crosslinker with a second residue in the same molecule or a precursor of such a residue.
[0234] Various methods to effect formation of peptidomimetic macrocycles are known in the art. For example, the preparation of peptidomimetic macrocycles of Formula I is described in Schafmeister et al., J. Am. Chem. Soc. 122:5891-5892 (2000); Schafmeister & Verdine, J. Am. Chem. Soc. 122:5891 (2005); Walensky et al., Science 305:1466-1470 (2004); U.S. Pat. No. 7,192,713 and PCT application WO 2008/121767. The .alpha.,.alpha.-disubstituted amino acids and amino acid precursors disclosed in the cited references can be employed in synthesis of the peptidomimetic macrocycle precursor polypeptides. For example, the "S5-olefin amino acid" is (S)-.alpha.-(2'-pentenyl) alanine and the "R8 olefin amino acid" is (R)-.alpha.-(2'-octenyl) alanine. Following incorporation of such amino acids into precursor polypeptides, the terminal olefins are reacted with a metathesis catalyst, leading to the formation of the peptidomimetic macrocycle. In various embodiments, the following amino acids can be employed in the synthesis of the peptidomimetic macrocycle:
##STR00042## ##STR00043##
[0235] In other embodiments, the peptidomimetic macrocycles are of Formula IV or IVa. Methods for the preparation of such macrocycles are described, for example, in U.S. Pat. No. 7,202,332.
[0236] Additional methods of forming peptidomimetic macrocycles which are envisioned as suitable include those disclosed by Mustapa, M. Firouz Mohd et al., J. Org. Chem (2003), 68, pp. 8193-8198; Yang, Bin et al. Bioorg Med. Chem. Lett. (2004), 14, pp. 1403-1406; U.S. Pat. Nos. 5,364,851; 5,446,128; 5,824,483; 6,713,280; and 7,202,332. In such embodiments, amino acid precursors are used containing an additional substituent R-- at the alpha position. Such amino acids are incorporated into the macrocycle precursor at the desired positions, which can be at the positions where the crosslinker is substituted or, alternatively, elsewhere in the sequence of the macrocycle precursor. Cyclization of the precursor is then effected according to the indicated method.
[0237] The peptidomimetic macrocycles described herein can be at least 1% pure, at least 2% pure, at least 3% pure, at least 4% pure, at least 5% pure, at least 6% pure, at least 7% pure, at least 8% pure, at least 9% pure, at least 10% pure, at least 11% pure, at least 12% pure, at least 13% pure, at least 14% pure, at least 15% pure, at least 16% pure, at least 17% pure, at least 18% pure, at least 19% pure, at least 20% pure, at least 21% pure, at least 22% pure, at least 23% pure, at least 24% pure, at least 25% pure, at least 26% pure, at least 27% pure, at least 28% pure, at least 29% pure, at least 30% pure, at least 31% pure, at least 32% pure, at least 33% pure, at least 34% pure, at least 35% pure, at least 36% pure, at least 37% pure, at least 38% pure, at least 39% pure, at least 40% pure, at least 41% pure, at least 42% pure, at least 43% pure, at least 44% pure, at least 45% pure, at least 46% pure, at least 47% pure, at least 48% pure, at least 49% pure, at least 50% pure, at least 51% pure, at least 52% pure, at least 53% pure, at least 54% pure, at least 55% pure, at least 56% pure, at least 57% pure, at least 58% pure, at least 59% pure, at least 60% pure, at least 61% pure, at least 62% pure, at least 63% pure, at least 64% pure, at least 65% pure, at least 66% pure, at least 67% pure, at least 68% pure, at least 69% pure, at least 70% pure, at least 71% pure, at least 72% pure, at least 73% pure, at least 74% pure, at least 75% pure, at least 76% pure, at least 77% pure, at least 78% pure, at least 79% pure, at least 80% pure, at least 81% pure, at least 82% pure, at least 83% pure, at least 84% pure, at least 85% pure, at least 86% pure, at least 87% pure, at least 88% pure, at least 89% pure, at least 90% pure, at least 91% pure, at least 92% pure, at least 93% pure, at least 94% pure, at least 95% pure, at least 96% pure, at least 97% pure, at least 98% pure, at least 99% pure, at least 99.1% pure, at least 99.2% pure, at least 99.3% pure, at least 99.4% pure, at least 99.5% pure, at least 99.6% pure, at least 99.7% pure, at least 99.8% pure, or at least 99.9% pure on a chemical, optical, isomeric, enantiomeric, or diastereomeric basis. Purity can be assessed, for example, by HPLC, MS, LC/MS, melting point, or NMR.
[0238] Two or more peptides/peptidomimetic macrocycles can share a degree of homology. A pair of peptides/peptidomimetic macrocycles can have, for example, up to about 20% pairwise homology, up to about 25% pairwise homology, up to about 30% pairwise homology, up to about 35% pairwise homology, up to about 40% pairwise homology, up to about 45% pairwise homology, up to about 50% pairwise homology, up to about 55% pairwise homology, up to about 60% pairwise homology, up to about 65% pairwise homology, up to about 70% pairwise homology, up to about 75% pairwise homology, up to about 80% pairwise homology, up to about 85% pairwise homology, up to about 90% pairwise homology, up to about 95% pairwise homology, up to about 96% pairwise homology, up to about 97% pairwise homology, up to about 98% pairwise homology, up to about 99% pairwise homology, up to about 99.5% pairwise homology, or up to about 99.9% pairwise homology. A pair of peptides can have, for example, at least about 20% pairwise homology, at least about 25% pairwise homology, at least about 30% pairwise homology, at least about 35% pairwise homology, at least about 40% pairwise homology, at least about 45% pairwise homology, at least about 50% pairwise homology, at least about 55% pairwise homology, at least about 60% pairwise homology, at least about 65% pairwise homology, at least about 70% pairwise homology, at least about 75% pairwise homology, at least about 80% pairwise homology, at least about 85% pairwise homology, at least about 90% pairwise homology, at least about 95% pairwise homology, at least about 96% pairwise homology, at least about 97% pairwise homology, at least about 98% pairwise homology, at least about 99% pairwise homology, at least about 99.5% pairwise homology, at least about 99.9% pairwise homology.
[0239] Various methods and software programs can be used to determine the homology between two or more peptides, such as NCBI BLAST, Clustal W, MAFFT, Clustal Omega, AlignMe, Praline, or another suitable method or algorithm.
Assays
[0240] The properties of peptidomimetic macrocycles are assayed, for example, by using the methods described below. In some embodiments, a peptidomimetic macrocycle has improved biological properties relative to a corresponding polypeptide lacking the substituents described herein.
Assay to Determine .alpha.-Helicity
[0241] In solution, the secondary structure of polypeptides with .alpha.-helical domains will reach a dynamic equilibrium between random coil structures and .alpha.-helical structures, often expressed as a "percent helicity". Thus, for example, alpha-helical domains are predominantly random coils in solution, with .alpha.-helical content usually under 25%. Peptidomimetic macrocycles with optimized linkers, on the other hand, possess, for example, an alpha-helicity that is at least two-fold greater than that of a corresponding uncrosslinked polypeptide. In some embodiments, macrocycles will possess an alpha-helicity of greater than 50%. To assay the helicity of peptidomimetic macrocycles, the compounds are dissolved in an aqueous solution (e.g. 50 mM potassium phosphate solution at pH 7, or distilled H.sub.2O, to concentrations of 25-50 .mu.M). Circular dichroism (CD) spectra are obtained on a spectropolarimeter (e.g., Jasco J-710) using standard measurement parameters (e.g. temperature, 20.degree. C.; wavelength, 190-260 nm; step resolution, 0.5 nm; speed, 20 nm/sec; accumulations, 10; response, 1 sec; bandwidth, 1 nm; path length, 0.1 cm). The .alpha.-helical content of each peptide is calculated by dividing the mean residue ellipticity (e.g. [.PHI.]222obs) by the reported value for a model helical decapeptide (Yang et al. (1986), Methods Enzymol. 130:208)).
Assay to Determine Melting Temperature (Tm)
[0242] A peptidomimetic macrocycle comprising a secondary structure such as an .alpha.-helix exhibits, for example, a higher melting temperature than a corresponding uncrosslinked polypeptide. Typically peptidomimetic macrocycles exhibit Tm of >60.degree. C. representing a highly stable structure in aqueous solutions. To assay the effect of macrocycle formation on melting temperature, peptidomimetic macrocycles or unmodified peptides are dissolved in distilled H.sub.2O (e.g. at a final concentration of 50 .mu.M) and the Tm is determined by measuring the change in ellipticity over a temperature range (e.g. 4 to 95.degree. C.) on a spectropolarimeter (e.g., Jasco J-710) using standard parameters (e.g. wavelength 222 nm; step resolution, 0.5 nm; speed, 20 nm/sec; accumulations, 10; response, 1 sec; bandwidth, 1 nm; temperature increase rate: 1.degree. C./min; path length, 0.1 cm).
Protease Resistance Assay
[0243] The amide bond of the peptide backbone is susceptible to hydrolysis by proteases, thereby rendering peptidic compounds vulnerable to rapid degradation in vivo. Peptide helix formation, however, typically buries the amide backbone and therefore can shield it from proteolytic cleavage. The peptidomimetic macrocycles can be subjected to in vitro trypsin proteolysis to assess for any change in degradation rate compared to a corresponding uncrosslinked polypeptide. For example, the peptidomimetic macrocycle and a corresponding uncrosslinked polypeptide are incubated with trypsin agarose and the reactions quenched at various time points by centrifugation and subsequent HPLC injection to quantitate the residual substrate by ultraviolet absorption at 280 nm. Briefly, the peptidomimetic macrocycle and peptidomimetic precursor (5 mcg) are incubated with trypsin agarose (Pierce) (S/E .about.125) for 0, 10, 20, 90, and 180 minutes. Reactions are quenched by tabletop centrifugation at high speed; remaining substrate in the isolated supernatant is quantified by HPLC-based peak detection at 280 nm. The proteolytic reaction displays first order kinetics and the rate constant, k, is determined from a plot of In[S] versus time (k=-1.times.slope).
Ex Vivo Stability Assay
[0244] Peptidomimetic macrocycles with optimized linkers possess, for example, an ex vivo half-life that is at least two-fold greater than that of a corresponding uncrosslinked polypeptide, and possess an ex vivo half-life of 12 hours or more. For ex vivo serum stability studies, a variety of assays can be used. For example, a peptidomimetic macrocycle and a corresponding uncrosslinked polypeptide (2 mcg) are incubated with fresh mouse, rat and/or human serum (2 mL) at 37.degree. C. for 0, 1, 2, 4, 8, and 24 hours. To determine the level of intact compound, the following procedure can be used: The samples are extracted by transferring 100 .mu.l of serato 2 ml centrifuge tubes followed by the addition of 10 .mu.L of 50% formic acid and 500 .mu.L acetonitrile and centrifugation at 14,000 RPM for 10 min at 4.+-.2.degree. C. The supernatants are then transferred to fresh 2 ml tubes and evaporated on Turbovap under N.sub.2<10 psi, 37.degree. C. The samples are reconstituted in 100 .mu.L of 50:50 acetonitrile:water and submitted to LC-MS/MS analysis.
In vitro Binding Assays
[0245] To assess the binding and affinity of peptidomimetic macrocycles and peptidomimetic precursors to acceptor proteins, a fluorescence polarization assay (FPA) is used, for example. The FPA technique measures the molecular orientation and mobility using polarized light and fluorescent tracer. When excited with polarized light, fluorescent tracers (e.g., FITC) attached to molecules with high apparent molecular weights (e.g. FITC-labeled peptides bound to a large protein) emit higher levels of polarized fluorescence due to their slower rates of rotation as compared to fluorescent tracers attached to smaller molecules (e.g. FITC-labeled peptides that are free in solution).
[0246] For example, fluoresceinated peptidomimetic macrocycles (25 nM) are incubated with the acceptor protein (25-1000 nM) in binding buffer (140 mM NaCl, 50 mM Tris-HCL, pH 7.4) for 30 minutes at room temperature. Binding activity is measured, for example, by fluorescence polarization on a luminescence spectrophotometer (e.g. Perkin-Elmer LS50B). Kd values can be determined by nonlinear regression analysis using, for example, Graphpad Prism software (GraphPad Software, Inc., San Diego, Calif.). A peptidomimetic macrocycle shows, In some embodiments, similar or lower Kd than a corresponding uncrosslinked polypeptide.
In Vitro Displacement Assays to Characterize Antagonists of Peptide-Protein Interactions
[0247] To assess the binding and affinity of compounds that antagonize the interaction between a peptide and an acceptor protein, a fluorescence polarization assay (FPA) utilizing a fluoresceinated peptidomimetic macrocycle derived from a peptidomimetic precursor sequence is used, for example. The FPA technique measures the molecular orientation and mobility using polarized light and fluorescent tracer. When excited with polarized light, fluorescent tracers (e.g., FITC) attached to molecules with high apparent molecular weights (e.g. FITC-labeled peptides bound to a large protein) emit higher levels of polarized fluorescence due to their slower rates of rotation as compared to fluorescent tracers attached to smaller molecules (e.g. FITC-labeled peptides that are free in solution). A compound that antagonizes the interaction between the fluoresceinated peptidomimetic macrocycle and an acceptor protein will be detected in a competitive binding FPA experiment.
[0248] For example, putative antagonist compounds (1 nM to 1 mM) and a fluoresceinated peptidomimetic macrocycle (25 nM) are incubated with the acceptor protein (50 nM) in binding buffer (140 mM NaCl, 50 mM Tris-HCL, pH 7.4) for 30 minutes at room temperature. Antagonist binding activity is measured, for example, by fluorescence polarization on a luminescence spectrophotometer (e.g. Perkin-Elmer LS50B). Kd values can be determined by nonlinear regression analysis using, for example, Graphpad Prism software (GraphPad Software, Inc., San Diego, Calif.).
[0249] Any class of molecule, such as small organic molecules, peptides, oligonucleotides or proteins can be examined as putative antagonists in this assay.
Assay for Protein-Ligand Binding by Affinity Selection-Mass Spectrometry
[0250] To assess the binding and affinity of test compounds for proteins, an affinity-selection mass spectrometry assay is used, for example. Protein-ligand binding experiments are conducted according to the following representative procedure outlined for a system-wide control experiment using 1 .mu.M peptidomimetic macrocycle plus 5 .mu.M hMDM2. A 1 .mu.L DMSO aliquot of a 40 .mu.M stock solution of peptidomimetic macrocycle is dissolved in 19 .mu.L of PBS (Phosphate-buffered saline: 50 mM, pH 7.5 Phosphate buffer containing 150 mM NaCl). The resulting solution is mixed by repeated pipetting and clarified by centrifugation at 10 000 g for 10 min. To a 4 .mu.L aliquot of the resulting supernatant is added 4 .mu.L of 10 M hMDM2 in PBS. Each 8.0 .mu.L experimental sample thus contains 40 pmol (1.5 .mu.g) of protein at 5.0 M concentration in PBS plus 1 .mu.M peptidomimetic macrocycle and 2.5% DMSO. Duplicate samples thus prepared for each concentration point are incubated for 60 min at room temperature, and then chilled to 4.degree. C. prior to size-exclusion chromatography-LC-MS analysis of 5.0 .mu.L injections. Samples containing a target protein, protein-ligand complexes, and unbound compounds are injected onto an SEC column, where the complexes are separated from non-binding component by a rapid SEC step. The SEC column eluate is monitored using UV detectors to confirm that the early-eluting protein fraction, which elutes in the void volume of the SEC column, is well resolved from unbound components that are retained on the column. After the peak containing the protein and protein-ligand complexes elutes from the primary UV detector, it enters a sample loop where it is excised from the flow stream of the SEC stage and transferred directly to the LC-MS via a valving mechanism. The (M+3H).sup.3+ ion of the peptidomimetic macrocycle is observed by ESI-MS at the expected m/z, confirming the detection of the protein-ligand complex.
Assay for Protein-Ligand Kd Titration Experiments
[0251] To assess the binding and affinity of test compounds for proteins, a protein-ligand Kd titration experiment is performed, for example. Protein-ligand K.sub.d titrations experiments are conducted as follows: 2 .mu.L DMSO aliquots of a serially diluted stock solution of titrant peptidomimetic macrocycle (5, 2.5, . . . , 0.098 mM) are prepared then dissolved in 38 .mu.L of PBS. The resulting solutions are mixed by repeated pipetting and clarified by centrifugation at 10 000 g for 10 min. To 4.0 .mu.L aliquots of the resulting supernatants is added 4.0 .mu.L of 10 M hMDM2 in PBS. Each 8.0 .mu.L experimental sample thus contains 40 pmol (1.5 .mu.g) of protein at 5.0 .mu.M concentration in PBS, varying concentrations (125, 62.5, . . . , 0.24 .mu.M) of the titrant peptide, and 2.5% DMSO. Duplicate samples thus prepared for each concentration point are incubated at room temperature for 30 min, then chilled to 4.degree. C. prior to SEC-LC-MS analysis of 2.0 .mu.L injections. The (M+H).sup.1+, (M+2H).sup.2+, (M+3H).sup.3+, and/or (M+Na).sup.1+ ion is observed by ESI-MS; extracted ion chromatograms are quantified, then fit to equations to derive the binding affinity K.sub.d as described in "A General Technique to Rank Protein-Ligand Binding Affinities and Determine Allosteric vs. Direct Binding Site Competition in Compound Mixtures." Annis, D. A.; Nazef, N.; Chuang, C. C.; Scott, M. P.; Nash, H. M. J Am. Chem. Soc. 2004, 126, 15495-15503; also in "ALIS: An Affinity Selection-Mass Spectrometry System for the Discovery and Characterization of Protein-Ligand Interactions" D. A. Annis, C.-C. Chuang, and N. Nazef. In Mass Spectrometry in Medicinal Chemistry. Edited by Wanner K, Hofner G: Wiley-VCH; 2007:121-184. Mannhold R, Kubinyi H, Folkers G (Series Editors): Methods and Principles in Medicinal Chemistry.
Assay for Competitive Binding Experiments by Affinity Selection-Mass Spectrometry
[0252] To determine the ability of test compounds to bind competitively to proteins, an affinity selection mass spectrometry assay is performed, for example. A mixture of ligands at 40 .mu.M per component is prepared by combining 2 .mu.L aliquots of 400 .mu.M stocks of each of the three compounds with 14 .mu.L of DMSO. Then, 1 .mu.L aliquots of this 40 .mu.M per component mixture are combined with 1 .mu.L DMSO aliquots of a serially diluted stock solution of titrant peptidomimetic macrocycle (10, 5, 2.5, . . . , 0.078 mM). These 2 L samples are dissolved in 38 .mu.L of PBS. The resulting solutions were mixed by repeated pipetting and clarified by centrifugation at 10 000 g for 10 min. To 4.0 .mu.L aliquots of the resulting supernatants is added 4.0 .mu.L of 10 .mu.M hMDM2 protein in PBS. Each 8.0 .mu.L experimental sample thus contains 40 pmol (1.5 .mu.g) of protein at 5.0 .mu.M concentration in PBS plus 0.5 .mu.M ligand, 2.5% DMSO, and varying concentrations (125, 62.5, . . . , 0.98 .mu.M) of the titrant peptidomimetic macrocycle. Duplicate samples thus prepared for each concentration point are incubated at room temperature for 60 min, then chilled to 4.degree. C. prior to SEC-LC-MS analysis of 2.0 .mu.L injections. Additional details on these and other methods are provided in "A General Technique to Rank Protein-Ligand Binding Affinities and Determine Allosteric vs. Direct Binding Site Competition in Compound Mixtures." Annis, D. A.; Nazef, N.; Chuang, C. C.; Scott, M. P.; Nash, H. M. J Am. Chem. Soc. 2004, 126, 15495-15503; also in "ALIS: An Affinity Selection-Mass Spectrometry System for the Discovery and Characterization of Protein-Ligand Interactions" D. A. Annis, C.-C. Chuang, and N. Nazef. In Mass Spectrometry in Medicinal Chemistry. Edited by Wanner K, Hofner G: Wiley-VCH; 2007:121-184. Mannhold R, Kubinyi H, Folkers G (Series Editors): Methods and Principles in Medicinal Chemistry.
Binding Assays in Intact Cells
[0253] It is possible to measure binding of peptides or peptidomimetic macrocycles to their natural acceptors in intact cells by immunoprecipitation experiments. For example, intact cells are incubated with fluoresceinated (FITC-labeled) compounds for 4 hrs in the absence of serum, followed by serum replacement and further incubation that ranges from 4-18 hrs. Cells are then pelleted and incubated in lysis buffer (50 mM Tris [pH 7.6], 150 mM NaCl, 1% CHAPS and protease inhibitor cocktail) for 10 minutes at 4.degree. C. Extracts are centrifuged at 14,000 rpm for 15 minutes and supernatants collected and incubated with 10 .mu.l goat anti-FITC antibody for 2 hrs, rotating at 4.degree. C. followed by further 2 hrs incubation at 4.degree. C. with protein A/G Sepharose (50 .mu.l of 50% bead slurry). After quick centrifugation, the pellets are washed in lysis buffer containing increasing salt concentration (e.g., 150, 300, 500 mM). The beads are then re-equilibrated at 150 mM NaCl before addition of SDS-containing sample buffer and boiling. After centrifugation, the supernatants are optionally electrophoresed using 4%-12% gradient Bis-Tris gels followed by transfer into Immobilon-P membranes. After blocking, blots are optionally incubated with an antibody that detects FITC and also with one or more antibodies that detect proteins that bind to the peptidomimetic macrocycle.
Cellular Penetrability Assays
[0254] A peptidomimetic macrocycle is, for example, more cell penetrable compared to a corresponding uncrosslinked macrocycle. Peptidomimetic macrocycles with optimized linkers possess, for example, cell penetrability that is at least two-fold greater than a corresponding uncrosslinked macrocycle, and often 20% or more of the applied peptidomimetic macrocycle will be observed to have penetrated the cell after 4 hours. To measure the cell penetrability of peptidomimetic macrocycles and corresponding uncrosslinked macrocycle, intact cells are incubated with fluorescently-labeled (e.g. fluoresceinated) peptidomimetic macrocycles or corresponding uncrosslinked macrocycle (10 .mu.M) for 4 hrs in serum free media at 37.degree. C., washed twice with media and incubated with trypsin (0.25%) for 10 min at 37.degree. C. The cells are washed again and resuspended in PBS. Cellular fluorescence is analyzed, for example, by using either a FACSCalibur flow cytometer or Cellomics' KineticScan.RTM. HCS Reader.
Cellular Efficacy Assays
[0255] The efficacy of certain peptidomimetic macrocycles is determined, for example, in cell-based killing assays using a variety of tumorigenic and non-tumorigenic cell lines and primary cells derived from human or mouse cell populations. Cell viability is monitored, for example, over 24-96 hrs of incubation with peptidomimetic macrocycles (0.5 to 50 .mu.M) to identify those that kill at EC.sub.50<10 .mu.M. Several standard assays that measure cell viability are commercially available and are optionally used to assess the efficacy of the peptidomimetic macrocycles. In addition, assays that measure Annexin V and caspase activation are optionally used to assess whether the peptidomimetic macrocycles kill cells by activating the apoptotic machinery. For example, the Cell Titer-glo assay is used which determines cell viability as a function of intracellular ATP concentration.
In Vivo Stability Assay
[0256] To investigate the in vivo stability of the peptidomimetic macrocycles, the compounds are, for example, administered to mice and/or rats by IV, IP, PO or inhalation routes at concentrations ranging from 0.1 to 50 mg/kg and blood specimens withdrawn at 0', 5', 15', 30', 1 hr, 4 hrs, 8 hrs and 24 hours post-injection. Levels of intact compound in 25 .mu.L of fresh serum are then measured by LC-MS/MS as above.
In Vivo Efficacy in Animal Models
[0257] To determine the anti-oncogenic activity of peptidomimetic macrocycles in vivo, the compounds are, for example, given alone (IP, IV, PO, by inhalation or nasal routes) or in combination with sub-optimal doses of relevant chemotherapy (e.g., cyclophosphamide, doxorubicin, etoposide). In one example, 5.times.10.sup.6 RS4;11 cells (established from the bone marrow of a patient with acute lymphoblastic leukemia) that stably express luciferase are injected by tail vein in NOD-SCID mice 3 hrs after they have been subjected to total body irradiation. If left untreated, this form of leukemia is fatal in 3 weeks in this model. The leukemia is readily monitored, for example, by injecting the mice with D-luciferin (60 mg/kg) and imaging the anesthetized animals (e.g., Xenogen In Vivo Imaging System, Caliper Life Sciences, Hopkinton, Mass.). Total body bioluminescence is quantified by integration of photonic flux (photons/sec) by Living Image Software (Caliper Life Sciences, Hopkinton, Mass.). Peptidomimetic macrocycles alone or in combination with sub-optimal doses of relevant chemotherapeutics agents are, for example, administered to leukemic mice (10 days after injection/day 1 of experiment, in bioluminescence range of 14-16) by tail vein or IP routes at doses ranging from 0.1 mg/kg to 50 mg/kg for 7 to 21 days. Optionally, the mice are imaged throughout the experiment every other day and survival monitored daily for the duration of the experiment. Expired mice are optionally subjected to necropsy at the end of the experiment. Another animal model is implantation into NOD-SCID mice of DoHH2, a cell line derived from human follicular lymphoma, that stably expresses luciferase. These in vivo tests optionally generate preliminary pharmacokinetic, pharmacodynamic and toxicology data.
Clinical Trials
[0258] To determine the suitability of the peptidomimetic macrocycles for treatment of humans, clinical trials are performed. For example, patients diagnosed with solid tumor and in need of treatment can be selected and separated in treatment and one or more control groups, wherein the treatment group is administered a peptidomimetic macrocycle, while the control groups receive a placebo or a known anti-cancer drug. The treatment safety and efficacy of the peptidomimetic macrocycles can thus be evaluated by performing comparisons of the patient groups with respect to factors such as survival and quality-of-life. In this example, the patient group treated with a peptidomimetic macrocycle can show improved long-term survival compared to a patient control group treated with a placebo.
Formulation and Administration
Mode of Administration
[0259] An effective amount of a peptidomimetic macrocycles or a pharmaceutically acceptable salt thereof of the disclosure can be administered in a pharmaceutical composition, as either single or multiple doses by an accepted mode of administration. In some embodiments, a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, of the disclosure are administered parenterally, for example, by subcutaneous, intramuscular, intrathecal, intravenous or epidural injection. For example, a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof is administered intravenously, intraarterially, subcutaneously or by infusion. In some examples, a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof is administered intravenously. In some examples, a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof is administered intraarterially.
[0260] Regardless of the route of administration selected, the peptidomimetic macrocycles of the present disclosure, and/or the pharmaceutical compositions of the present disclosure, are formulated into pharmaceutically-acceptable dosage forms by conventional methods known to those of skill in the art. The peptidomimetic macrocycles according to the disclosure can be formulated for administration in any convenient way for use in human or veterinary medicine, by analogy with other pharmaceuticals.
[0261] In one aspect, the disclosure provides pharmaceutical composition comprising a therapeutically-effective amount of one or more of the peptidomimetic macrocycles described above, formulated together with one or more pharmaceutically acceptable carriers (additives) and/or diluents. In one embodiment, one or more of the peptidomimetic macrocycles described herein are formulated for parenteral administration for parenteral administration, one or more peptidomimetic macrocycles disclosed herein can be formulated as aqueous or nonaqueous solutions, dispersions, suspensions or emulsions or sterile powders which can be reconstituted into sterile injectable solutions or dispersions just prior to use. Such a pharmaceutical composition can comprise sugars, alcohols, antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents, A pharmaceutical composition can also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms upon the subject compounds can be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It can also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into a pharmaceutical composition. In addition, prolonged absorption of an injectable pharmaceutical composition can be brought about by the inclusion of agents which delay absorption such as aluminum monostearate and gelatin. If desired a pharmaceutical composition can be diluted prior to use with, for example, an isotonic saline solution or a dextrose solution. In some examples, the peptidomimetic macrocycle is formulated as an aqueous solution and is administered intravenously.
Amount and Frequency of Administration
[0262] Dosing can be determined using techniques known to one skilled in the art. The selected dosage level can depend upon a variety of factors including the activity of the particular peptidomimetic macrocycle employed, the route of administration, the time of administration, the rate of excretion or metabolism of the particular peptidomimetic macrocycle being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular peptidomimetic macrocycle employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts. The dosage values can also vary with the severity of the condition to be alleviated. For any particular subject, specific dosage regimens can be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the compositions.
[0263] A physician or veterinarian having ordinary skill in the art can readily determine and prescribe the effective amount of the pharmaceutical composition required. For example, the physician or veterinarian could start doses of the compounds of the disclosure employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved.
[0264] In some embodiments, a suitable daily dose of a peptidomimetic macrocycle of the disclosure can be that amount of the peptidomimetic macrocycle which is the lowest dose effective to produce a therapeutic effect. Such an effective dose will generally depend upon the factors described above. The precise time of administration and amount of any particular peptidomimetic macrocycle that will yield the most effective treatment in a given patient will depend upon the activity, pharmacokinetics, and bioavailability of a particular peptidomimetic macrocycle, physiological condition of the patient (including age, sex, disease type and stage, general physical condition, responsiveness to a given dosage and type of medication), route of administration, and the like.
[0265] Dosage can be based on the amount of the peptidomimetic macrocycle per kg body weight of the patient. Other amounts are known to those of skill in the art and readily determined. Alternatively, the dosage of the subject disclosure can be determined by reference to the plasma concentrations of the peptidomimetic macrocycle. For example, the maximum plasma concentration (Cmax) and the area under the plasma concentration-time curve from time 0 to infinity (AUC) can be used.
[0266] In some embodiment, the subject is a human subject and the amount of the peptidomimetic macrocycle administered is 0.01-100 mg per kilogram body weight of the human subject. For example, in various examples, the amount of the peptidomimetic macrocycle administered is about 0.01-50 mg/kg, about 0.01-20 mg/kg, about 0.01-10 mg/kg, about 0.1-100 mg/kg, about 0.1-50 mg/kg, about 0.1-20 mg/kg, about 0.1-10 mg/kg, about 0.5-100 mg/kg, about 0.5-50 mg/kg, about 0.5-20 mg/kg, about 0.5-10 mg/kg, about 1-100 mg/kg, about 1-50 mg/kg, about 1-20 mg/kg, about 1-10 mg/kg body weight of the human subject. In one embodiment, about 0.5 mg-10 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered. In some examples the amount of the peptidomimetic macrocycle administered is about 0.16 mg, 0.32 mg, 0.64 mg, 1.28 mg, 3.56 mg, 7.12 mg, 14.24, or 20 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is about 0.16 mg, 0.32 mg, 0.64 mg, 1.28 mg, 3.56 mg, 7.12 mg, or 14.24 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 0.16 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 0.32 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 0.64 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 1.28 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 3.56 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 7.12 mg per kilogram body weight of the human subject. In some examples the amount of the peptidomimetic macrocycle administered is 14.24 mg per kilogram body weight of the human subject.
[0267] In some embodiments about 0.5-20 mg or 0.5-10 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered two times a week. For example about 0.5-1.0 mg, 0.5-5.0 mg, 0.5-10.0 mg, 0.5-15 mg, or 1-5 mg, 1-10 mg, 1-15 mg, 1-20 mg, 5-10 mg, 1-15 mg, 5-20 mg, 10-15 mg, 10-20 mg, 15-20 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administrated about twice a week. In some examples about 1.0 mg, 1.25 mg, 1.5 mg, 1.75 mg, 2.0 mg, 2.25 mg, 2.5 mg, 2.75 mg, 3.0 mg, 3.25 mg, 3.5 mg, 3.75 mg, 4.0 mg, 4.25 mg, 4.5 mg, 4.75 mg, 5.0 mg, 5.25 mg, 5.5 mg, 5.75 mg, 6.0 mg, 6.25 mg, 6.5 mg, 6.75 mg, 7.0 mg, 7.25 mg, 7.5 mg, 7.75 mg, 8.0 mg, 8.25 mg, 8.5 mg, 8.75 mg, 9.0 mg, 9.25 mg, 9.5 mg, 9.75 mg, 10.0 mg, 10.25 mg, 10.5 mg, 10.75 mg, 11.0 mg, 11.25 mg, 11.5 mg, 11.75 mg, 12.0 mg, 12.25 mg, 12.5 mg, 12.75 mg, 13.0 mg, 13.25 mg, 13.5 mg, 13.75 mg, 14.0 mg, 14.25 mg, 14.5 mg, 14.75 mg, 15.0 mg, 15.25 mg, 15.5 mg, 15.75 mg, 16.0 mg, 16.5 mg, 17.0 mg, 17.5 mg, 18.0 mg, 18.5 mg, 19.0 mg, 19.5 mg, or 20.0 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered two times a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg, 10.0, or 20 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered two times a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered two times a week.
[0268] In some embodiments about 0.5-20 mg or 0.5-10 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered once a week. For example about 0.5-1.0 mg, 0.5-5.0 mg, 0.5-10.0 mg, 0.5-15 mg, or 1-5 mg, 1-10 mg, 1-15 mg, 1-20 mg, 5-10 mg, 1-15 mg, 5-20 mg, 10-15 mg, 10-20 mg, 15-20 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administrated once a week. In some examples about 1.0 mg, 1.25 mg, 1.5 mg, 1.75 mg, 2.0 mg, 2.25 mg, 2.5 mg, 2.75 mg, 3.0 mg, 3.25 mg, 3.5 mg, 3.75 mg, 4.0 mg, 4.25 mg, 4.5 mg, 4.75 mg, 5.0 mg, 5.25 mg, 5.5 mg, 5.75 mg, 6.0 mg, 6.25 mg, 6.5 mg, 6.75 mg, 7.0 mg, 7.25 mg, 7.5 mg, 7.75 mg, 8.0 mg, 8.25 mg, 8.5 mg, 8.75 mg, 9.0 mg, 9.25 mg, 9.5 mg, 9.75 mg, 10.0 mg, 10.25 mg, 10.5 mg, 10.75 mg, 11.0 mg, 11.25 mg, 11.5 mg, 11.75 mg, 12.0 mg, 12.25 mg, 12.5 mg, 12.75 mg, 13.0 mg, 13.25 mg, 13.5 mg, 13.75 mg, 14.0 mg, 14.25 mg, 14.5 mg, 14.75 mg, 15.0 mg, 15.25 mg, 15.5 mg, 15.75 mg, 16.0 mg, 16.5 mg, 17.0 mg, 17.5 mg, 18.0 mg, 18.5 mg, 19.0 mg, 19.5 mg, or 20.0 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered once a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once a week
[0269] In some embodiments about 0.5-20 mg or 0.5-10 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered 3, 4, 5, 6 or 7 times a week. For example about 0.5-1.0 mg, 0.5-5.0 mg, 0.5-10.0 mg, 0.5-15 mg, or 1-5 mg, 1-10 mg, 1-15 mg, 1-20 mg, 5-10 mg, 1-15 mg, 5-20 mg, 10-15 mg, 10-20 mg, 15-20 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administrated 3, 4, 5, 6, or 7 times a week. In some examples about 1.0 mg, 1.25 mg, 1.5 mg, 1.75 mg, 2.0 mg, 2.25 mg, 2.5 mg, 2.75 mg, 3.0 mg, 3.25 mg, 3.5 mg, 3.75 mg, 4.0 mg, 4.25 mg, 4.5 mg, 4.75 mg, 5.0 mg, 5.25 mg, 5.5 mg, 5.75 mg, 6.0 mg, 6.25 mg, 6.5 mg, 6.75 mg, 7.0 mg, 7.25 mg, 7.5 mg, 7.75 mg, 8.0 mg, 8.25 mg, 8.5 mg, 8.75 mg, 9.0 mg, 9.25 mg, 9.5 mg, 9.75 mg, 10.0 mg, 10.25 mg, 10.5 mg, 10.75 mg, 11.0 mg, 11.25 mg, 11.5 mg, 11.75 mg, 12.0 mg, 12.25 mg, 12.5 mg, 12.75 mg, 13.0 mg, 13.25 mg, 13.5 mg, 13.75 mg, 14.0 mg, 14.25 mg, 14.5 mg, 14.75 mg, 15.0 mg, 15.25 mg, 15.5 mg, 15.75 mg, 16.0 mg, 16.5 mg, 17.0 mg, 17.5 mg, 18.0 mg, 18.5 mg, 19.0 mg, 19.5 mg, or 20.0 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered 3, 4, 5, 6, or 7 times a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered 3, 4, 5, 6, or 7 times a week. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered 3, 4, 5, 6, or 7 times a week.
[0270] In some embodiments about 0.5-20 mg or 0.5-10 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered once every 2, 3, or 4 weeks. For example about 0.5-1.0 mg, 0.5-5.0 mg, 0.5-10.0 mg, 0.5-15 mg, or 1-5 mg, 1-10 mg, 1-15 mg, 1-20 mg, 5-10 mg, 1-15 mg, 5-20 mg, 10-15 mg, 10-20 mg, 15-20 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administrated 3, 4, 5, 6, or 7 once every 2 or 3 week. In some examples about 1.0 mg, 1.25 mg, 1.5 mg, 1.75 mg, 2.0 mg, 2.25 mg, 2.5 mg, 2.75 mg, 3.0 mg, 3.25 mg, 3.5 mg, 3.75 mg, 4.0 mg, 4.25 mg, 4.5 mg, 4.75 mg, 5.0 mg, 5.25 mg, 5.5 mg, 5.75 mg, 6.0 mg, 6.25 mg, 6.5 mg, 6.75 mg, 7.0 mg, 7.25 mg, 7.5 mg, 7.75 mg, 8.0 mg, 8.25 mg, 8.5 mg, 8.75 mg, 9.0 mg, 9.25 mg, 9.5 mg, 9.75 mg, 10.0 mg, 10.25 mg, 10.5 mg, 10.75 mg, 11.0 mg, 11.25 mg, 11.5 mg, 11.75 mg, 12.0 mg, 12.25 mg, 12.5 mg, 12.75 mg, 13.0 mg, 13.25 mg, 13.5 mg, 13.75 mg, 14.0 mg, 14.25 mg, 14.5 mg, 14.75 mg, 15.0 mg, 15.25 mg, 15.5 mg, 15.75 mg, 16.0 mg, 16.5 mg, 17.0 mg, 17.5 mg, 18.0 mg, 18.5 mg, 19.0 mg, 19.5 mg, or 20.0 mg of the peptidomimetic macrocycle per kilogram body weight of the human subject is administered once every 2 or 3 weeks. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once every 2 weeks. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once every 2 weeks. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg 10.0 mg, or 20.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once every 3 weeks. In some examples, the amount of the peptidomimetic macrocycle administered is about 1.25 mg, 2.5 mg, 5.0 mg or 10.0 mg per kilogram body weight of the human subject and the peptidomimetic macrocycle is administered once every 3 weeks.
[0271] In some embodiments, the peptidomimetic macrocycle is administered gradually over a period of time. A desired amount of peptidomimetic macrocycle can be administered gradually over a period of from about 0.1 h-24 h. In some cases a desired amount of peptidomimetic macrocycle is administered gradually over a period of 0.1 h, 0.5 h, 1 h, 1.5 h, 2 h, 2.5 h, 3 h, 3.5 h, 4 h, 4.5 h, 5 h, 6 h, 7 h, 8 h, 9 h, 10 h, 11 h, 12 h, 13 h, 14 h, 15 h, 16 h, 17 h, 18 h, 19 h, 20 h, 21 h, 22 h, 23 h, or 24 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 0.25-12 h, for example over a period of 0.25-1 h, 0.25-2 h, 0.25-3 h, 0.25-4 h, 0.25-6 h, 0.25-8 h, 0.25-10 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 0.25-2 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 0.25-1 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 0.25 h, 0.3 h, 0.4 h, 0.5 h, 0.6 h, 0.7 h, 0.8 h, 0.9 h, 1.0 h, 1.1 h, 1.2 h, 1.3 h, 1.4 h, 1.5 h, 1.6 h, 1.7 h, 1.8 h, 1.9 h, or 2.0 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 1 h. In some examples, a desired amount of peptidomimetic macrocycle is administered gradually over a period of 2 h.
[0272] Administration of the peptidomimetic macrocycles can continue as long as necessary to treat a solid tumor in a subject in need thereof. In some embodiments, one or more peptidomimetic macrocycle of the disclosure is administered for more than 1 day, 1 week, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 13 months, 14 months, 15 months, 16 months, 17 months, 18 months, 19 months, 20 months, 21 months, 22 months, 23 months, or 24 months. In some embodiments, one or more peptidomimetic macrocycle of the disclosure is administered for less than 1 week, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 13 months, 14 months, 15 months, 16 months, 17 months, 18 months, 19 months, 20 months, 21 months, 22 months, 23 months, or 24 months.
[0273] In some embodiments, the peptidomimetic macrocycle is administered on day 1, 8, 15 and 28 of a 28 day cycle. In some embodiments, the peptidomimetic macrocycle is administered on day 1, 8, 15 and 28 of a 28 day cycle and administration is continued for two cycles. In some embodiments, the peptidomimetic macrocycle is administered on day 1, 8, 15 and 28 of a 28 day cycle and administration is continued for three cycles. In some embodiments, the peptidomimetic macrocycle is administered on day 1, 8, 15 and 28 of a 28 day cycle and administration is continued for 4, 5, 6, 7, 8, 9, 10, or more cycles.
[0274] In some embodiments, one or more peptidomimetic macrocycle of the disclosure is administered chronically on an ongoing basis. In some embodiments administration of one or more peptidomimetic macrocycle of the disclosure is continued until documentation of disease progression, unacceptable toxicity, or patient or physician decision to discontinue administration.
Method and Uses
[0275] In one aspect, the disclosure provides a method of treating solid tumor in a subject, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycle can disrupt the interaction between p53 and MDM2 and MDMX. In some embodiments, treatment according to the method disclosed herein can result in re-activation of the p53 pathway, decreased tumor cell proliferation, increased p53 protein, increased p21, and/or increased apoptosis in the human subject.
[0276] In one aspect, the disclosure provides a method of treating solid tumor, that lacks a p53 deactivating mutation, in a subject the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the solid tumor is determined to lack a p53 mutation prior to treatment In some embodiments, the peptidomimetic macrocycle can disrupt the interaction between p53 and MDM2 and MDMX. The method further can comprise confirming the lack of the p53 deactivating mutation in the subject prior to the administration of the peptidomimetic macrocycle. In some embodiments, treatment according to the method disclosed herein can result in re-activation of the p53 pathway, decreased tumor cell proliferation, increased p53 protein, increased p21, and/or increased apoptosis in the human subject.
[0277] In one aspect, the disclosure provides a method of treating solid tumor in a subject expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some embodiments, the peptidomimetic macrocycle can disrupt the interaction between p53 and MDM2 and MDMX. The method further can comprise confirming the wild type p53 status of the subject prior to the administration of the peptidomimetic macrocycle. In some embodiments, treatment according to the method disclosed herein can result in re-activation of the p53 pathway, decreased tumor cell proliferation, increased p53 protein, increased p21, and/or increased apoptosis in the human subject.
[0278] In some embodiments, the methods for treating solid tumor provided herein inhibit, reduce, diminish, arrest, or stabilize a tumor associated with the solid tumor. In other embodiments, the methods for treating solid tumor provided herein inhibit, reduce, diminish, arrest, or stabilize the blood flow, metabolism, or edema in a tumor associated with the solid tumor or one or more symptoms thereof. In some examples, the methods for treating solid tumor provided herein cause the regression of a tumor, tumor blood flow, tumor metabolism, or peritumor edema, and/or one or more symptoms associated with the solid tumor. In other examples, the methods for treating solid tumor provided herein maintain the size of the tumor so that it does not increase, or so that it increases by less than the increase of a tumor after administration of a standard therapy as measured by conventional methods available to one of skill in the art, such as ultrasound, CT Scan, MRI, dynamic contrast-enhanced MRI, or PET Scan. In some examples, the methods for treating solid tumor provided herein decrease tumor size. In some examples, the methods for treating solid tumor provided herein reduce the formation of a tumor. In some examples, the methods for treating solid tumor provided herein eradicate, remove, or control primary, regional and/or metastatic tumors associated with the solid tumor. In some examples, the methods for treating solid tumor provided herein decrease the number or size of metastases associated with the solid tumor. In some examples, the methods for treating solid tumor provided herein reduce the tumor volume or tumor size (e.g., diameter) in a subject by an amount in the range of about 5-10%, 5-20%, 10-20%, 15-20%, 10-30%, 20-30%, 20-40%, 30-40%, 10-50%, 20-50%, 30-50%, 40-50%, 10-60%, 20-60%, 30-60%, 40-60%, 50-60%, 10-70%, 20-70%, 30-70%, 40-70%, 50-70%, 60-70%, 10-80%, 20-80%, 30-80%, 40-80%, 50-80%, 60-80%, 70- 80%, 10-90%, 20-90%, 30-90%, 40-90%, 50-90%, 60-90%, 70-90%, 80-90%, 10-100%, 20%-100%, 30-100%, 40-100%, 50-100%, 60-100%, 70-100%, 80-100%, 90-100%, 95-100%, or any range in between, relative to tumor size (e.g., volume or diameter) in a subject prior to administration of the peptidomimetic macrocycles as assessed by methods well known in the art, e.g., CT Scan, MRI, DCE-MRI, or PET Scan. In certain embodiments, the methods herein reduce the tumor size (e.g., volume or diameter) in a subject by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 80%, 85%, 90%, 95%, 99%, or 100%, relative to tumor volume or tumor size (e.g., diameter) prior to administration of the peptidomimetic macrocycle as assessed by methods well known in the art, e.g., CT Scan, MRI, DCE-MRI, or PET Scan.
[0279] In some embodiments, the methods provided herein reduce the tumor perfusion in a subject by an amount in the range of about 5-10%, 5-20%, 10-20%, 15-20%, 10-30%, 20-30%, 20-40%, 30-40%, 10-50%, 20-50%, 30-50%, 40-50%, 10-60%, 20-60%, 30-60%, 40-60%, 50-60%, 10-70%, 20-70%, 30-70%, 40-70%, 50-70%, 60-70%, 10-80%, 20-80%, 30-80%, 40-80%, 50-80%, 60-80%, 70-80%, 10-90%, 20-90%, 30- 90%, 40-90%, 50-90%, 60-90%, 70-90%, 80-90%, 10-100%, 20%-100%, 30-100%, 40-100%, 50-100%, 60-100%, 70-100%, 80-100%, 90-100%, 95-100%, or any range in between, relative to tumor perfusion prior to administration of the peptidomimetic macrocycle, as assessed by methods well known in the art, e.g., MRI, DCE-MRI, or PET Scan. In certain embodiments, the methods provided herein reduce the tumor perfusion in a subject by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 80%, 85%, 90%, 95%, 99%, or 100%, relative to tumor perfusion prior to administration of the peptidomimetic macrocycle as assessed by methods well known in the art, e.g., MRI, DCE-MRI, or PET Scan.
[0280] In some embodiments, the methods provided herein inhibit or decrease tumor metabolism in a subject in the range of about 5-10%, 5-20%, 10-20%, 15-20%, 10-30%, 20-30%, 20-40%, 30-40%, 10-50%, 20-50%, 30-50%, 40-50%, 10-60%, 20-60%, 30-60%, 40-60%, 50-60%, 10-70%, 20-70%, 30-70%, 40-70%, 50-70%, 60-70%, 10-80%, 20-80%, 30-80%, 40-80%, 50-80%, 60-80%, 70-80%, 10-90%, 20-90%, 30- 90%, 40-90%, 50-90%, 60-90%, 70-90%, 80-90%, 10-100%, 20%-100%, 30-100%, 40-100%, 50-100%, 60-100%, 70-100%, 80-100%, 90-100%, 95-100%, or any range in between, relative to tumor metabolism prior to administration of the peptidomimetic macrocycle, as assessed by methods well known in the art. In certain embodiments, the methods provided herein inhibit or decrease tumor metabolism in a subject as assessed by methods well known in the art, e.g., PET scanning. In specific embodiments, the methods provided herein inhibit or decrease tumor metabolism in a subject by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 80%, 85%, 90%, 95%, or 100%, relative to tumor metabolism prior to administration of the peptidomimetic macrocycle, as assessed by methods well known in the art.
[0281] In other aspect, the disclosure provides a method for increasing the survival time of a subject with solid tumor determined to lack a p53 deactivating mutation and/or with solid tumor expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins. In some examples, the survival time of the subject is at least 30 days longer than the expected survival time of the subject if the subject was not treated according to the methods provided herein. In some examples, the survival time of the subject is at 1 month-about 5 years longer than the expected survival time of the subject if the subject was not treated according to the methods provided herein. For example, the survival time of the subject is at least 3 months, at least 6 months, at least 9 months, at least 12 months, at least 15 months, at least 18 months, at least 21 months, or at least 24 months longer than the expected survival time of the subject if the subject was not treated according to the methods disclosed herein disclosure.
[0282] In one aspect, the disclosure provides a method to access presence, absence or amount of the biomarker biomarkers in a subject suffering with solid tumor, the method comprising. In some examples, the biomarkers include patient biomarkers for example the p53 status of the subject and the MDM2 and MDMX expression levels in the subject.
[0283] The method of the disclosure can also optionally include studying and/or evaluating the safety and/or tolerability of the peptidomimetic macrocycles disclosed herein in the subject.
[0284] Also provided herein is a method to re-activate the p53 pathway in a subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0285] Also provided herein is a method to decrease tumor cell proliferation in a human subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0286] Also provided herein is a method to increased p53 protein in a subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0287] Also provided herein is a method to increased p21 in a subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0288] Also provided herein is a method to increased apoptosis in a subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53, the method comprising administering to the subject a pharmaceutical composition comprising a therapeutically effective amount of a peptidomimetic macrocycle or a therapeutically equivalent amount of a pharmaceutically acceptable salt thereof, wherein the peptidomimetic macrocycle binds to MDM2 and/or MDMX proteins.
[0289] In some embodiments, the disclosure also provides a method to determine the dose limiting toxicities (DLTs) and/or maximum tolerated dose (MTD) of the peptidomimetic macrocycles disclosed herein in subject with a solid tumor lacking a p53 deactivating mutation and/or expressing wild type p53.
[0290] The methods of the disclosure can optionally include pharmacokinetic analysis of the peptidomimetic macrocycles disclosed herein. Accordingly, the methods can further comprise collecting one or more biological sample from the subject at one or more specific time point and analyzing the one or more biological sample for levels of the peptidomimetic macrocycles and/or it metabolites. The biological sample can be a blood sample from the subject, for example blood sample from a human subject. The one or more specific time point can include time points before, after and/or during the administration of the peptidomimetic macrocycle to the subject. In some embodiments one or more biological sample includes biological samples collected before and after each administration of the peptidomimetic macrocycle to the subject. In some embodiments a biological sample for pharmacokinetic analysis is collected before the first administration of the peptidomimetic macrocycle to the subject and at one or more time points after each administration of the peptidomimetic macrocycles to the subject. The biological sample collected before the administration of the peptidomimetic macrocycle to the subject can be done within 0-24 hour before the start of administration of the peptidomimetic macrocycle to the subject. For example, the biological sample can be collected within 24 h, 23 h, 22 h, 21 h, 20 h, 19 h, 18 h, 17 h, 16 h, 15 h, 14 h, 13 h, 12 h, 11 h, 10 h, 9 h, 8 h, 7 h, 6 h, 5 h, 4 h, 3 h, 2 h, 1 h, 30 min, 15 min, or immediately before the administration of the peptidomimetic macrocycle to the subject. One or more biological samples collected after the administration of the peptidomimetic macrocycle to the subject can be collected from 0-about 72 h, for example after 0 min, 5 min, 10 min, 20 min, 30 min, 45 min, 60 min, 1.25 h, 1.5 h, 1.75 h, 2.0 h, 2.25 h, 2.5 h, 2.75 h, 3.0 h, 3.25 h, 3.5 h, 3.75 h, 4.0 h, 4.25 h, 4.5 h, 4.75 h, 5.0 h, 5.25 h, 5.5 h, 5.75 h, 6.0 h, 6.25 h, 6.5 h, 6.75 h, 7.0 h, 7.25 h, 7.5 h, 7.75 h, 8.0 h, 8.25 h, 8.5 h, 8.75 h, 9.0 h, 9.25 h, 9.5 h, 9.75 h, 10.0 h, 10.25 h, 10.5 h, 10.75 h, 11.0 h, 11.25 h, 11.5 h, 11.75 h, 12.0 h, 20 h, 24 h, 28 h, 32 h, 36 h, 40 h, 44 h, 48 h, 52 h, 56 h, 60 h, 64 h, 68 h, or 72 h after the administration of the peptidomimetic macrocycle to the subject. In some embodiments, the peptidomimetic macrocycle is administered on day 1, day 8, day 15 of a 28 day cycle and biological sample is collected before administration on day 1, after the administration on day 1 (multiple biological samples can be collected, for example after about 0 min, about 30 min, about 1 h, about 2 h, about 4 h, about 8 h, about 24 h, and 48 hour after administration), before administration on day 8, after administration on day 8 (multiple biological samples can be collected, for example after about 0 min, about 30 min, about 1 h, about 2 h, and about 4 h after administration), before administration on day 15 and after administration on day 15(multiple biological samples can be collected, for example after about 0 min, about 30 min, about 1 h, about 2 h, about 4 h, about 8 h, and about 24 h after administration).
[0291] The method of the disclosure can optionally include pharmacodynamic analysis of the peptidomimetic macrocycles disclosed herein. Accordingly, the methods can comprise collecting one or more biological samples from the subject at one or more specific time points for pharmacodynamic analysis. Pharmacodynamic analysis can include analyzing the levels of biomarkers including MIC-1, p53, MDM2, MDMX, p21 and/or cases in the biological sample. Detection of biomarkers in a biological sample can be performed by any conventional method for detecting the type of biomarker, e.g., direct measurement, immunohistochemistry, immunoblotting, immunoflourescense, immunoabsorbence, immunoprecipitations, protein array, flourescence in situ hybridization, FACS analysis, hybridization, in situ hybridization, Northern blots, Southern blots, Western blots, ELISA, radioimmunoassay, gene array/chip, PCR, RT-PCR, or cytogenetic analysis. The biological sample for pharmacodynamic analysis can be a blood sample or a tumor specimen from the subject, for example biological sample for pharmacodynamic analysis can be a blood sample or a tumor specimen from the human subject. The biological samples for pharmacodynamic analysis of the peptidomimetic macrocycles can be collected any time before, during, or after the administration of the peptidomimetic macrocycle to the subject. In some embodiments a blood sample for pharmacokinetic analysis is collected before the first administration of the peptidomimetic macrocycle to the subject and at one or more time points after each administration of the peptidomimetic macrocycles to the subject. The blood sample collected before the administration of the peptidomimetic macrocycle to the subject can be done within 0-24 hour before the start of administration of the peptidomimetic macrocycle to the subject. For example, the biological sample can be collected within 24 h, 23 h, 22 h, 21 h, 20 h, 19 h, 18 h, 17 h, 16 h, 15 h, 14 h, 13 h, 12 h, 11 h, 10 h, 9 h, 8 h, 7 h, 6 h, 5 h, 4 h, 3 h, 2 h, 1 h, 30 min, 15 min, or immediately before the administration of the peptidomimetic macrocycle to the subject. One or more blood samples for pharmacodynamic analysis collected after the administration of the peptidomimetic macrocycle to the subject can be collected from 0-about 72 h, for example after about 12 h, 24 h, 36 h or 48 h after the administration of the peptidomimetic macrocycle to the subject. In some embodiments, the peptidomimetic macrocycle is administered on day 1, day 8, day 15 of a 28 day cycle and blood samples for pharmacodynamic analysis are collected before administration on day 1, after the administration on day 1 (multiple samples can be collected, for example after about 24 h and 48 hour after administration), before administration on day 8, after administration on day 8 (multiple samples can be collected, for example with about 1 h administration), before administration on day 15 and after administration on day 15(multiple samples can be collected, for example with about 1 h and about 48 h after administration), and day 22. Tumor specimens for pharmacodynamic analysis can be collected at any time before, after or during the administration of the peptidomimetic macrocycle to the subject. For example the peptidomimetic macrocycle can be administered on day 1, day 8, day 15 of a 28 day cycle and tumor samples for pharmacodynamic analysis are collected before administration on day 1 and between day 14-day 18, for example of day 16.
[0292] The method of the disclosure can optionally include clinical activity analysis of the peptidomimetic macrocycles disclosed herein. Accordingly, the methods can comprise analyzing one or more biological samples collected from the subject at one or more specific time points. Any appropriate analytical procedure can be used for the analysis of the biological samples. For example, imaging techniques like radiographs, ultrasound, CT scan, PET scan, MRI scan, chest x-ray, laparoscopy, complete blood count (CBC) test, bone scanning and fecal occult blood test can be used. Further analytical procedures that can be used include blood chemistry analysis, chromosomal translocation analysis, needle biopsy, tissue biopsy, fluorescence in situ hybridization, laboratory biomarker analysis, immunohistochemistry staining method, flow cytometry, or a combination thereof. The method can further comprise tabulating and/or plotting results of the analytical procedure.
Biological Samples
[0293] As used in the present application, "biological sample" means any fluid or other material derived from the body of a normal or diseased subject, such as blood, serum, plasma, lymph, urine, saliva, tears, cerebrospinal fluid, milk, amniotic fluid, bile, ascites fluid, pus, and the like. Also included within the meaning of the term "biological sample" is an organ or tissue extract and culture fluid in which any cells or tissue preparation from a subject has been incubated. Biological samples also include tumor samples or specimens. Tumor sample can be a tumor tissue sample. Methods of obtaining tumor tissue samples are well known in the art and can vary according to the type and location of a tumor and preferences of the physician. In some embodiments, the tumor tissue sample can obtained from surgically excised tissue. Tissue samples and cellular samples can also be obtained without invasive surgery, for example by punctuating the chest wall or the abdominal wall or from masses of breast, thyroid or other sites with a fine needle and withdrawing cellular material (fine needle aspiration biopsy).
[0294] The biological samples obtained can be used in fresh, frozen, or fixed (e.g., paraffin-embedded) form, depending on the nature of the sample, the assay used, and the convenience of the practitioner. Although fresh, frozen and fixed materials are suitable for various RNA and protein assays, generally, fresh tissues can be preferred for ex vivo measurements of activity.
[0295] Fixed tissue samples can also be employed. Tissue obtained by biopsy is often fixed, usually by formalin, formaldehyde, or gluteraldehyde, for example, or by alcohol immersion. Fixed biological samples are often dehydrated and embedded in paraffin or other solid supports, as is known in the art. See the reference Plenat et al., 2001, Ann. Pathol. 21:29-47. Non-embedded, fixed tissue, as well as fixed and embedded tissue, can be used in the present methods. Solid supports for embedding fixed tissue can be removed with organic solvents to enable subsequent rehydration of preserved tissue.
[0296] In some cases, the assay includes a step of cell or tissue culture. Culture methods are well known in the art. For example, cells from a biopsy can be disaggregated using enzymes (such as collagenase and hyaluronidase) and or physical disruption (e.g., repeated passage through a 25-gauge needle) to dissociate the cells, collected by centrifugation, and resuspended in desired buffer or culture medium for culture, immediate analysis, or further processing.
Subject/Patient Population
[0297] In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor.
[0298] In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor, determined to lack a p53 deactivating mutation and/or expressing wild type p53. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor, determined to lack a p53 deactivating mutation and/or expressing wild type p53. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor, determined to lack a p53 deactivating mutation and/or expressing wild type p53. A p53 deactivating mutation, as used herein is any mutation that leads to loss of (or a decrease in) the in vitro apoptotic activity of p53. Non limiting examples of p53 deactivating mutations are included in Table 1a. Accordingly, in some embodiments, a subject with a solid tumor in accordance with the composition as provided herein is a human who has or is diagnosed with a solid tumor that is determined to lack a p53 deactivation mutation, such as those shown in Table 1a.
[0299] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor, determined to have a p53 gain of function mutation. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor, determined to have a p53 gain of function mutation. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor, determined to have a p53 gain of function mutation. A p53 gain of function mutation, as used herein is any mutation such that the mutant p53 exerts oncogenic functions beyond their negative domination over the wild-type p53 tumor suppressor functions. The p53 gain of function mutant protein mat exhibit new activities that can contribute actively to various stages of tumor progression and to increased resistance to anticancer treatments. Non limiting examples of p53 gain of function mutation mutations are included in Table 1b. Accordingly, in some embodiments, a subject with a solid tumor in accordance with the composition as provided herein is a human who has or is diagnosed with a solid tumor that is determined to have a p53 gain of function mutation, such as those shown in Table 1b.
[0300] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor that is not p53 negative. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor that is not p53 negative. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor that is not p53 negative.
[0301] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor that expresses p53 with partial loss of function mutation. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor that expresses p53 with partial loss of function mutation. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor that expresses p53 with partial loss of function mutation. As used herein "a partial loss of p53 function" mutation means that the mutant p53 exhibits some level of function of normal p53, but to a lesser or slower extent. For example, a partial loss of p53 function can mean that the cells become arrested in cell division to a lesser or slower extent.
[0302] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor that expresses p53 with a copy loss mutation and a deactivating mutation. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor that expresses p53 with a copy loss mutation and a deactivating mutation. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor that expresses p53 with a copy loss mutation and a deactivating mutation.
[0303] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor that expresses p53 with a copy loss mutation. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor that expresses p53 with a copy loss mutation. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor that expresses p53 with a copy loss mutation.
[0304] In some embodiments, the subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor that expresses p53 with one or more silent mutations. In other embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, predisposed or susceptible to a solid tumor that expresses p53 with one or more silent mutations. In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, at risk of developing a solid tumor that expresses p53 with one or more silent mutations. Silent mutations as used herein are mutations which cause no change in the encoded p53 amino acid sequence.
[0305] In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human, who has or is diagnosed with a solid tumor, determined to lack a dominant p53 deactivating mutation. Dominant p53 deactivating mutation or dominant negative mutation, as used herein, is a mutation wherein the mutated p53 inhibits or disrupt the activity of the wild-type p53 gene.
TABLE-US-00002 TABLE 1a Exemplary of p53 deactivating mutations Mutation at position Amino acid change 62 E62_W91del 122 V122X 135 C135S 143 V143A 144 Q144P 146 W146X 157 V157F 158 R158H 163 Y163N 168 H168Y 173 V173L 175 R175H 175 R175P 175 R175Q 175 R175S 219 P219H 234 Y234C 234 Y234H 237 M237I 240 S240R 245 G245C 245 G245S 246 M246I 248 R248Q 248 R248W 249 R249S 272 V272M 273 R273H 274 V274F 279 G279E 280 R280K 281 D281H 282 R282W 306 R306P 308 P300_L308del 327 P300_Y327del 332 D324_I332del 337 R337C 344 L344P
TABLE-US-00003 TABLE 1b Exemplary p53 activating mutations AA position AAchange 46 S46F 121 S121F 123 T123A 288 N288K
[0306] Table 1a and 1b refer to the sequence of the wild-type human TP53 tumor protein p53 shown in FIG. 1. Amino acid changes are reported as: the amino acid being substituted followed by the position of the amino acid being substituted in the wild type p53 sequence, followed by the amino acid used for substitution. For example L344P, indicates that the lysine (K) at the 344 position in the wild type sequence is replaced by a proline (P).
[0307] In some embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a refractory patient. In a certain embodiment, a refractory patient is a patient refractory to a standard therapy (e.g., surgery, radiation, anti-androgen therapy and/or drug therapy such as chemotherapy). In certain embodiments, a patient with the solid tumor is refractory to a therapy when the solid tumor has not significantly been eradicated and/or the one or more symptoms have not been significantly alleviated. The determination of whether a patient is refractory can be made either in vivo or in vitro by any method known in the art for assaying the effectiveness of a treatment of solid tumor. In various embodiments, a patient with solid tumor is refractory when one or more tumors associated with the solid tumor, have not decreased or have increased. In various embodiments, a patient with solid tumor is refractory when one or more tumors metastasize and/or spread to another organ.
[0308] In some embodiments, a subject treated for solid tumor accordance with the methods provided herein is a human that has proven refractory to therapies other than treatment with the peptidomimetic macrocycles of the disclosure, but is no longer on these therapies. In certain embodiments, a subject treated for solid tumor in accordance with the methods provided herein is a human already receiving one or more conventional anti-cancer therapies, such as surgery, drug therapy such as chemotherapy, anti-androgen therapy or radiation. Among these patients are refractory patients, patients who are too young for conventional therapies, and patients with recurring tumors despite treatment with existing therapies.
[0309] In some embodiments, the subject is a human who has had at least one unsuccessful prior treatment and/or therapy of the solid tumor.
Methods of Detecting Wild Type p53 and/or p53 Mutations
[0310] The tumor samples from a subject can be assayed in order to determine the lack of a p53 deactivating mutation and/or expression of wild type p53.
[0311] In order to detect the p53 wild-type gene and/or lack of p53 deactivation mutation in a tissue, it can be helpful to isolate the tissue free from surrounding normal tissues. Means for enriching a tissue preparation for tumor cells are known in the art. For example, the tissue can be isolated from paraffin or cryostat sections. Cancer cells can also be separated from normal cells by flow cytometry. These as well as other techniques for separating tumor from normal cells are well known in the art. If the tumor tissue is highly contaminated with normal cells, detection of mutations can be more difficult.
[0312] Detection of point mutations can be accomplished by molecular cloning of the p53 allele (or alleles) present in the tumor tissue and sequencing that allele(s) using techniques well known in the art. Alternatively, the polymerase chain reaction can be used to amplify p53 gene sequences directly from a genomic DNA preparation from the tumor tissue. The DNA sequence of the amplified sequences can then be determined. The polymerase chain reaction itself is well known in the art. See e.g., Saiki et al., Science, Vol. 239, p. 487, 1988; U.S. Pat. Nos. 4,683,202; and 4,683,195.
[0313] Specific deletions of p53 genes can also be detected. For example, restriction fragment length polymorphism (RFLP) probes for the p53 gene or surrounding marker genes can be used to score loss of a p53 allele. Other techniques for detecting deletions, as are known in the art can be used.
[0314] Loss of wild-type p53 genes can also be detected on the basis of the loss of a wild-type expression product of the p53 gene. Such expression products include both the mRNA as well as the p53 protein product itself. Point mutations can be detected by sequencing the mRNA directly or via molecular cloning of cDNA made from the mRNA. The sequence of the cloned cDNA can be determined using DNA sequencing techniques which are well known in the art. The cDNA can also be sequenced via the polymerase chain reaction (PCR).
[0315] Alternatively, mismatch detection can be used to detect point mutations in the p53 gene or its mRNA product. The method can involve the use of a labeled riboprobe which is complementary to the human wild-type p53 gene. The riboprobe and either mRNA or DNA isolated from the tumor tissue are annealed (hybridized) together and subsequently digested with the enzyme RNase A which is able to detect some mismatches in a duplex RNA structure. If a mismatch is detected by RNase A, it cleaves at the site of the mismatch. Thus, when the annealed RNA preparation is separated on an electrophoretic gel matrix, if a mismatch has been detected and cleaved by RNase A, an RNA product will be seen which is smaller than the full-length duplex RNA for the riboprobe and the p53 mRNA or DNA. The riboprobe need not be the full length of the p53 mRNA or gene but can be a segment of either. If the riboprobe comprises only a segment of the p53 mRNA or gene it will be desirable to use a number of these probes to screen the whole mRNA sequence for mismatches.
[0316] In similar fashion, DNA probes can be used to detect mismatches, through enzymatic or chemical cleavage. See, e.g., Cotton et al., Proc. Natl. Acad. Sci. USA, vol. 85, 4397, 1988; and Shenk et al., Proc. Natl. Acad. Sci. USA, vol. 72, p. 989, 1975. Alternatively, mismatches can be detected by shifts in the electrophoretic mobility of mismatched duplexes relative to matched duplexes. See, e.g., Cariello, Human Genetics, vol. 42, p. 726, 1988. With either riboprobes or DNA probes, the cellular mRNA or DNA which might contain a mutation can be amplified using PCR (see below) before hybridization.
[0317] DNA sequences of the p53 gene from the tumor tissue which have been amplified by use of polymerase chain reaction can also be screened using allele-specific probes. These probes are nucleic acid oligomers, each of which contains a region of the p53 gene sequence harboring a known mutation. For example, one oligomer can be about 30 nucleotides in length, corresponding to a portion of the p53 gene sequence. At the position coding for the 175th codon of p53 gene the oligomer encodes an alanine, rather than the wild-type codon valine. By use of a battery of such allele-specific probes, the PCR amplification products can be screened to identify the presence of a previously identified mutation in the p53 gene. Hybridization of allele-specific probes with amplified p53 sequences can be performed, for example, on a nylon filter. Hybridization to a particular probe indicates the presence of the same mutation in the tumor tissue as in the allele-specific probe.
[0318] The identification of p53 gene structural changes in tumor cells has been facilitated through the development and application of a diverse series of high resolution, high throughput microarray platforms. Essentially there are two types of array; those that carry PCR products from cloned nucleic acids {e.g. cDNA, BACs, cosmids) and those that use oligonucleotides. Each has advantages and disadvantages but it is now possible to survey genome wide DNA copy number abnormalities and expression levels to allow correlations between losses, gains and amplifications in tumor cells with genes that are over- and under-expressed in the same samples. The gene expression arrays that provide estimates of mRNA levels in tumors have given rise to exon-specific arrays that can identify both gene expression levels, alternative splicing events and mRNA processing alterations. Oligonucleotide arrays are also being used to interrogate single nucleotide polymorphisms (SNPs) throughout the genome for linkage and association studies and these have been adapted to quantify copy number abnormalities and loss of heterozygosity events. Ultimately DNA sequencing arrays will allow resequencing of chromosome regions and whole genomes.
[0319] SNP-based arrays or other gene arrays or chips are also contemplated to determine the presence of wild-type p53 allele and the structure of mutations. A single nucleotide polymorphism (SNP), a variation at a single site in DNA, is the most frequent type of variation in the genome. For example, there are an estimated 5-10 million SNPs in the human genome. As SNPs are highly conserved throughout evolution and within a population, the map of SNPs serves as an excellent genotypic marker for research. An SNP array is a useful tool to study the whole genome.
[0320] In addition, SNP array can be used for studying the Loss Of Heterozygosity (LOH). LOH is a form of allelic imbalance that can result from the complete loss of an allele or from an increase in copy number of one allele relative to the other. While other chip-based methods (e.g., comparative genomic hybridization can detect only genomic gains or deletions), SNP array has the additional advantage of detecting copy number neutral LOH due to uniparental disomy (UPD). In UPD, one allele or whole chromosome from one parent are missing leading to reduplication of the other parental allele (uni-parental=from one parent, disomy=duplicated). In a disease setting this occurrence can be pathologic when the wild-type allele (e.g., from the mother) is missing and instead two copies of the heterozygous allele (e.g., from the father) are present. This usage of SNP array has a huge potential in cancer diagnostics as LOH is a prominent characteristic of most human cancers. Recent studies based on the SNP array technology have shown that not only solid tumors (e.g. gastric cancer, liver cancer etc) but also hematologic malignancies (ALL, MDS, CML etc) have a high rate of LOH due to genomic deletions or UPD and genomic gains. In the present disclosure, using high density SNP array to detect LOH allows identification of pattern of allelic imbalance to determine the presence of wild-type p53 allele (Lips et ah, 2005; Lai et al, 2007).
[0321] Examples for current p53 gene sequence and single nucleotide polymorphism arrays include p53 Gene Chip (Affymetrix, Santa Clara, Calif.), Roche p53 Ampli-Chip (Roche Molecular Systems, Pleasanton, Calif.), GeneChip Mapping arrays (Affymetrix, Santa Clara, Calif.), SNP Array 6.0 (Affymetrix, Santa Clara, Calif.), BeadArrays (Illumina, San Diego, Calif.), etc.
[0322] Mutations of wild-type p53 genes can also be detected on the basis of the mutation of a wild-type expression product of the p53 gene. Such expression products include both the mRNA as well as the p53 protein product itself. Point mutations can be detected by sequencing the mRNA directly or via molecular cloning of cDNA made from the mRNA. The sequence of the cloned cDNA can be determined using DNA sequencing techniques which are well known in the art. The cDNA can also be sequenced via the polymerase chain reaction (PCR). A panel of monoclonal antibodies could be used in which each of the epitopes involved in p53 functions are represented by a monoclonal antibody. Loss or perturbation of binding of a monoclonal antibody in the panel would indicate mutational alteration of the p53 protein and thus of the p53 gene itself. Mutant p53 genes or gene products can also be detected in body samples, such as, serum, stool, or other body fluids, such as urine and sputum. The same techniques discussed above for detection of mutant p53 genes or gene products in tissues can be applied to other body samples. 2. Assessment of p53 Protein Level
[0323] Loss of wild-type p53 genes can also be detected by screening for loss of wild-type p53 protein function. Although all of the functions which the p53 protein undoubtedly possesses have yet to be elucidated, at least two specific functions are known. Protein p53 binds to the SV40 large T antigen as well as to the adenovirus E1B antigen. Loss of the ability of the p53 protein to bind to either or both of these antigens indicates a mutational alteration in the protein which reflects a mutational alteration of the gene itself. Alternatively, a panel of monoclonal antibodies could be used in which each of the epitopes involved in p53 functions are represented by a monoclonal antibody. Loss or perturbation of binding of a monoclonal antibody in the panel would indicate mutational alteration of the p53 protein and thus of the p53 gene itself. Any means for detecting an altered p53 protein can be used to detect loss of wild-type p53 genes.
[0324] Mutant p53 genes or gene products can also be detected in body samples, such as, serum, stool, or other body fluids, such as urine and sputum. The same techniques discussed above for detection of mutant p53 genes or gene products in tissues can be applied to other body samples.
[0325] Determination of the lack of p53 deactivating mutation and/or expression of wild type p53 in the subject with solid tumor can be performed any time before, during or after the administration of the peptidomimetic macrocycles. In some embodiments, the determination of the lack of a p53 deactivating mutation and/or expression of wild type p53 is performed before the first administration of the peptidomimetic macrocycle to the subject, for example about 5 years-1 month, 4 years-1 month, 3 years-1 month, 2 years-1 month, 1 years-1 month, 5 years-1 week, 4 years-1 week, 3 years-1 month, 2 years-1 week, 1 year-1 week, 5 years-1 day, 4 years-1 day, 3 years-1 days, 2 years-1 day, 1 year-1 day, 15 months-1 month, 15 months-1 week, 15 months-1 day, 12 months-1 month, 12 months-1 week, 12 months-1 day, 6 months-1 month, 6 months-1 week, 6 months-1 day, 3 months-1 month, 3 months-1 week, or 3 months-1 day prior to the first administration of the peptidomimetic macrocycle to the subject. In some examples, the confirmation of the lack of the p53 deactivating mutation and/or expression of wild type p53 is performed up to 6 years, 5 years, 4 years, 3 years, 24 months, 23 months, 22 months, 21 months, 20 months, 19 months, 18 months, 17 months, 16 months, 15 months, 14 months, 13 months, 12 months, 11 months, 10 months, 9 months, 8 months, 7 months, 6 months, 5 months, 4 months, 3 months, 2 months, 1 months, 4 weeks (28 days), 3 weeks (21 days), 2 weeks (14 days), 1 week (7 days), 6 days, 5 days, 4 days, 3 days, 2 days or 1 day before the first administration of the peptidomimetic macrocycle to the subject. In some examples the confirmation of the lack of the p53 deactivating mutation is performed within 1 month of the first administration of the peptidomimetic macrocycle to the subject. In some examples the confirmation of the lack of the p53 deactivating mutation is performed within 21 days of the first administration of the peptidomimetic macrocycle to the subject.
Solid Tumors
[0326] Solid tumors that can be treated by the instant methods include tumors and/or metastasis (wherever located) other than lymphatic cancer, for example brain and other central nervous system tumors (including but not limited to tumors of the meninges, brain, spinal cord, cranial nerves and other parts of central nervous system, e.g. glioblastomas or medulla blastomas); head and/or neck cancer; breast tumors; circulatory system tumors (including but not limited to heart, mediastinum and pleura, and other intrathoracic organs, vascular tumors and tumor-associated vascular tissue); excretory system tumors (including but not limited to tumors of kidney, renal pelvis, ureter, bladder, other and unspecified urinary organs); gastrointestinal tract tumors (including but not limited to tumors of oesophagus, stomach, small intestine, colon, colorectal, rectosigmoid junction, rectum, anus and anal canal, tumors involving the liver and intrahepatic bile ducts, gall bladder, other and unspecified parts of biliary tract, pancreas, other and digestive organs); oral cavity tumors (including but not limited to tumors of lip, tongue, gum, floor of mouth, palate, and other parts of mouth, parotid gland, and other parts of the salivary glands, tonsil, oropharynx, nasopharynx, pyriform sinus, hypopharynx, and other sites in the lip, oral cavity and pharynx); reproductive system tumors (including but not limited to tumors of vulva, vagina, Cervix uteri, Corpus uteri, uterus, ovary, and other sites associated with female genital organs, placenta, penis, prostate, testis, and other sites associated with male genital organs); respiratory tract tumors (including but not limited to tumors of nasal cavity and middle ear, accessory sinuses, larynx, trachea, bronchus and lung, e.g. small cell lung cancer or non-small cell lung cancer); skeletal system tumors (including but not limited to tumors of bone and articular cartilage of limbs, bone articular cartilage and other sites); skin tumors (including but not limited to malignant melanoma of the skin, non-melanoma skin cancer, basal cell carcinoma of skin, squamous cell carcinoma of skin, mesothelioma, Kaposi's sarcoma); and tumors involving other tissues including peripheral nerves and autonomic nervous system, connective and soft tissue, retroperitoneum and peritoneum, eye and adnexa, thyroid, adrenal gland and other endocrine glands and related structures, secondary and unspecified malignant neoplasm of lymph nodes, secondary malignant neoplasm of respiratory and digestive systems and secondary malignant neoplasm of other sites.
[0327] In some examples, the solid tumor treated by the methods of the instant disclosure is pancreatic cancer, bladder cancer, colon cancer, liver cancer, colorectal cancer (colon cancer or rectal cancer), breast cancer, prostate cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors, bone cancer, skin cancer, ocular tumor, choriocarcinoma (tumor of the placenta), sarcoma or soft tissue cancer.
[0328] In some examples, the solid tumor to be treated by the methods of the instant disclosure is selected bladder cancer, bone cancer, breast cancer, cervical cancer, CNS cancer, colon cancer, ocular tumor, renal cancer, liver cancer, lung cancer, pancreatic cancer, choriocarcinoma (tumor of the placenta), prostate cancer, sarcoma, skin cancer, soft tissue cancer or gastric cancer.
[0329] In some examples, the solid tumor treated by the methods of the instant disclosure is breast cancer. Non limiting examples of breast cancer that can be treated by the instant methods include ductal carcinoma in situ (DCIS or intraductal carcinoma), lobular carcinoma in situ (LCIS), invasive (or infiltrating) ductal carcinoma, invasive (or infiltrating) lobular carcinoma, inflammatory breast cancer, triple-negative breast cancer, paget disease of the nipple, phyllodes tumor (phylloides tumor or cystosarcoma phyllodes), angiosarcoma, adenoid cystic (or adenocystic) carcinoma, low-grade adenosquamous carcinoma, medullary carcinoma, papillary carcinoma, tubular carcinoma, metaplastic carcinoma, micropapillary carcinoma, and mixed carcinoma.
[0330] In some examples, the solid tumor treated by the methods of the instant disclosure is bone cancer. Non limiting examples of bone cancer that can be treated by the instant methods include osteosarcoma, chondrosarcoma, the Ewing Sarcoma Family of Tumors (ESFTs).
[0331] In some examples, the solid tumor treated by the methods of the instant disclosure is skin cancer. Non limiting examples of skin cancer that can be treated by the instant methods include melanoma, basal cell skin cancer, and squamous cell skin cancer.
[0332] In some examples, the solid tumor treated by the methods of the instant disclosure is ocular tumor. Non limiting examples of ocular tumor that can be treated by the methods of the instant disclosure include ocular tumor is choroidal nevus, choroidal melanoma, choroidal metastasis, choroidal hemangioma, choroidal osteoma, iris melanoma, uveal melanoma, intraocular lymphoma, melanocytoma, metastasis retinal capillary hemangiomas, congenital hypertrophy of the RPE, RPE adenoma or retinoblastoma.
[0333] In some embodiments solid tumors treated by the methods disclosed herein exclude cancers that are known to be associated with HPV (Human papillomavirus). The excluded group includes HPV positive cervical cancer, HPV positive anal cancer, and HPV head and neck cancers, such as oropharyngeal cancers.
[0334] The effectiveness and/or response of cancer treatment by the methods disclosed herein can be determined by any method known in art. The response can be a complete response, and which can be an objective response, a clinical response, or a pathological response to treatment. For example, the response can be determined based upon the techniques for evaluating response to treatment of solid tumors as described in Therese et al., New Guidelines to Evaluate the Response to Treatment in Solid Tumors, J. of the National Cancer Institute 92(3):205-207 (2000), which is hereby incorporated by reference in its entirety. The response can be a duration of survival (or probability of such duration) or progression-free interval. The timing or duration of such events can be determined from about the time of diagnosis, or from about the time treatment is initiated or from about the time treatment is finished (like the final administration of the peptidomimetic macrocycle). Alternatively, the response can be based upon a reduction in tumor size, tumor volume, or tumor metabolism, or based upon overall tumor burden, or based upon levels of serum markers especially where elevated in the disease state.
[0335] The response in individual patients can be characterized as a complete response, a partial response, stable disease, and progressive disease, as these terms are understood in the art. In some embodiments, the response is complete response (CR). Complete response, in some examples can be defined as disappearance of all target lesions i.e. any pathological lymph nodes (whether target or non-target) must have reduction in short axis to <10 mm. In certain embodiments, the response is a partial response (PR). Partial response can be defined to mean at least 30% decrease in the sum of diameters of target lesions, taking as reference the baseline sum diameters. In some embodiments, the response is progressive disease (PD). Progressive disease can be defined as at least a 20% increase in the sum of diameters of target lesions, taking as reference the smallest sum on study (this includes the baseline sum if that is the smallest) and an absolute increase of at least 5 mm in the sum of diameters of target lesions. The appearance of one or more new lesions can also be considered as progression. In some embodiments, the disease can be stable disease (SD). Stable disease can be characterized by neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as reference the smallest sum diameters while on study. In certain embodiments, the response is a pathological complete response. A pathological complete response, e.g., as determined by a pathologist following examination of tissue removed at the time of surgery or biopsy, generally refers to an absence of histological evidence of invasive tumor cells in the surgical specimen.
Combination Treatment
[0336] Also provided herein are combination therapies for the treatment of a solid tumor which involve the administration of the peptidomimetic macrocycles disclosed herein in combination with one or more additional therapies to a subject with solid tumor determined to lack a p53 deactivating mutation and/or express wild type p53. In a specific embodiment, presented herein are combination therapies for the treatment of solid tumor which involve the administration of an effective amount of the peptidomimetic macrocycles in combination with an effective amount of another therapy to a subject with a solid tumor determined to lack a p53 deactivating mutation and/or with a solid tumor expressing wild type p53.
[0337] As used herein, the term "in combination," refers, in the context of the administration of the peptidomimetic macrocycles, to the administration of the peptidomimetic macrocycles prior to, concurrently with, or subsequent to the administration of one or more additional therapies (e.g., agents, surgery, or radiation) for use in treating solid tumor. The use of the term "in combination" does not restrict the order in which the peptidomimetic macrocycles and one or more additional therapies are administered to a subject. In specific embodiments, the interval of time between the administration of the peptidomimetic macrocycles and the administration of one or more additional therapies can be about 1-5 minutes, 1-30 minutes, 30 minutes to 60 minutes, 1 hour, 1-2 hours, 2-6 hours, 2-12 hours, 12-24 hours, 1-2 days, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8 weeks, 9 weeks, 10 weeks, 15 weeks, 20 weeks, 26 weeks, 52 weeks, 11-15 weeks, 15-20 weeks, 20-30 weeks, 30-40 weeks, 40-50 weeks, 1 month, 2 months, 3 months, 4 months 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 1 year, 2 years, or any period of time in between. In certain embodiments, the peptidomimetic macrocycles and one or more additional therapies are administered less than 1 day, 1 week, 2 weeks, 3 weeks, 4 weeks, one month, 2 months, 3 months, 6 months, 1 year, 2 years, or 5 years apart.
[0338] In some embodiments, the combination therapies provided herein involve administering of the peptidomimetic macrocycles 1-2 times a week, once every week, once every 2 weeks, once every 3 weeks, once every 4 weeks, once every 5 weeks, once every 6 weeks, once every 7 weeks or once every 8 weeks and administering one or more additional therapies once a week, once every 2 weeks, once every 3 weeks, once every 4 weeks, once every month, once every 2 months (e.g., approximately 8 weeks), once every 3 months (e.g., approximately 12 weeks), or once every 4 months (e.g., approximately 16 weeks). In certain embodiments, the peptidomimetic macrocycles and one or more additional therapies are cyclically administered to a subject. Cycling therapy involves the administration of the peptidomimetic macrocycles compounds for a period of time, followed by the administration of one or more additional therapies for a period of time, and repeating this sequential administration. In certain embodiments, cycling therapy can also include a period of rest where the peptidomimetic macrocycles or the additional therapy is not administered for a period of time (e.g., 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 10 weeks, 20 weeks, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 2 years, or 3 years). In an embodiment, the number of cycles administered is from 1 to 12 cycles, from 2 to 10 cycles, or from 2 to 8 cycles.
[0339] In some embodiments, the methods for treating solid tumor provided herein comprise administering the peptidomimetic macrocycles as a single agent for a period of time prior to administering the peptidomimetic macrocycles in combination with an additional therapy. In certain embodiments, the methods for treating cancer provided herein comprise administering an additional therapy alone for a period of time prior to administering the peptidomimetic macrocycles in combination with the additional therapy.
[0340] In some embodiments, the administration of the peptidomimetic macrocycles and one or more additional therapies in accordance with the methods presented herein have an additive effect relative the administration of the peptidomimetic macrocycles or said one or more additional therapies alone. In some embodiments, the administration of the peptidomimetic macrocycles and one or more additional therapies in accordance with the methods presented herein have a synergistic effect relative to the administration of the peptidomimetic macrocycles or said one or more additional therapies alone.
[0341] As used herein, the term "synergistic," refers to the effect of the administration of the peptidomimetic macrocycles in combination with one or more additional therapies (e.g., agents), which combination is more effective than the additive effects of any two or more single therapies (e.g., agents). In a specific embodiment, a synergistic effect of a combination therapy permits the use of lower dosages (e.g., sub-optimal doses) of the peptidomimetic macrocycles or an additional therapy and/or less frequent administration of the peptidomimetic macrocycles or an additional therapy to a subject. In certain embodiments, the ability to utilize lower dosages of the peptidomimetic macrocycles or of an additional therapy and/or to administer the peptidomimetic macrocycles or said additional therapy less frequently reduces the toxicity associated with the administration of the peptidomimetic macrocycles or of said additional therapy, respectively, to a subject without reducing the efficacy of the peptidomimetic macrocycles or of said additional therapy, respectively, in the treatment of solid tumor. In some embodiments, a synergistic effect results in improved efficacy of the peptidomimetic macrocycles and each of said additional therapies in treating cancer. In some embodiments, a synergistic effect of a combination of the peptidomimetic macrocycles and one or more additional therapies avoids or reduces adverse or unwanted side effects associated with the use of any single therapy.
[0342] The combination of the peptidomimetic macrocycles and one or more additional therapies can be administered to a subject in the same pharmaceutical composition. Alternatively, the peptidomimetic macrocycles and one or more additional therapies can be administered concurrently to a subject in separate pharmaceutical compositions. The peptidomimetic macrocycles and one or more additional therapies can be administered sequentially to a subject in separate pharmaceutical compositions. The peptidomimetic macrocycles compounds and one or more additional therapies can also be administered to a subject by the same or different routes of administration.
[0343] The combination therapies provided herein involve administering to a subject to in need thereof the peptidomimetic macrocycles in combination with conventional, or known, therapies for treating cancer. Other therapies for cancer or a condition associated therewith are aimed at controlling or relieving one or more symptoms. Accordingly, in some embodiments, the combination therapies provided herein involve administering to a subject to in need thereof a pain reliever, or other therapies aimed at alleviating or controlling one or more symptoms associated with or a condition associated therewith.
[0344] Non-limiting specific examples of anti-cancer agents that can be used in combination with the peptidomimetic macrocycles include: a hormonal agent (e.g., aromatase inhibitor, selective estrogen receptor modulator (SERM), and estrogen receptor antagonist), chemotherapeutic agent (e.g., microtubule disassembly blocker, antimetabolite, topoisomerase inhibitor, and DNA crosslinker or damaging agent), anti-antigenic agent (e.g., VEGF antagonist, receptor antagonist, integrin antagonist, vascular targeting agent (VTA)/vascular disrupting agent (VDA)), radiation therapy, and conventional surgery.
[0345] Non-limiting examples of hormonal agents that can be used in combination with the peptidomimetic macrocycles include aromatase inhibitors, SERMs, and estrogen receptor antagonists. Hormonal agents that are aromatase inhibitors can be steroidal or no steroidal. Non-limiting examples of no steroidal hormonal agents include letrozole, anastrozole, aminoglutethimide, fadrozole, and vorozole. Non-limiting examples of steroidal hormonal agents include aromasin (exemestane), formestane, and testolactone. Non-limiting examples of hormonal agents that are SERMs include tamoxifen (branded/marketed as Nolvadex.RTM.), afimoxifene, arzoxifene, bazedoxifene, clomifene, femarelle, lasofoxifene, ormeloxifene, raloxifene, and toremifene. Non-limiting examples of hormonal agents that are estrogen receptor antagonists include fulvestrant. Other hormonal agents include but are not limited to abiraterone and lonaprisan.
[0346] Non-limiting examples of chemotherapeutic agents that can be used in combination with of peptidomimetic macrocycles include microtubule disassembly blocker, antimetabolite, topoisomerase inhibitor, and DNA crosslinker or damaging agent. Chemotherapeutic agents that are microtubule disassembly blockers include, but are not limited to, taxanes (e.g., paclitaxel (branded/marketed as TAXOL.RTM.), docetaxel, abraxane, larotaxel, ortataxel, and tesetaxel); epothilones (e.g., ixabepilone); and vinca alkaloids (e.g., vinorelbine, vinblastine, vindesine, and vincristine (branded/marketed as ONCOVIN.RTM.)).
[0347] Chemotherapeutic agents that are antimetabolites include, but are not limited to, folate anitmetabolites (e.g., methotrexate, aminopterin, pemetrexed, raltitrexed); purine antimetabolites (e.g., cladribine, clofarabine, fludarabine, mercaptopurine, pentostatin, thioguanine); pyrimidine antimetabolites (e.g., 5-fluorouracil, capcitabine, gemcitabine (GEMZAR.RTM.), cytarabine, decitabine, floxuridine, tegafur); and deoxyribonucleotide antimetabolites (e.g., hydroxyurea).
[0348] Chemotherapeutic agents that are topoisomerase inhibitors include, but are not limited to, class I (camptotheca) topoisomerase inhibitors (e.g., topotecan (branded/marketed as HYCAMTIN.RTM.) irinotecan, rubitecan, and belotecan); class II (podophyllum) topoisomerase inhibitors (e.g., etoposide or VP-16, and teniposide); anthracyclines (e.g., doxorubicin, epirubicin, Doxil, aclarubicin, amrubicin, daunorubicin, idarubicin, pirarubicin, valrubicin, and zorubicin); and anthracenediones (e.g., mitoxantrone, and pixantrone).
[0349] Chemotherapeutic agents that are DNA crosslinkers (or DNA damaging agents) include, but are not limited to, alkylating agents (e.g., cyclophosphamide, mechlorethamine, ifosfamide (branded/marketed as IFEX.RTM.), trofosfamide, chlorambucil, melphalan, prednimustine, bendamustine, uramustine, estramustine, carmustine (branded/marketed as BiCNU.RTM.), lomustine, semustine, fotemustine, nimustine, ranimustine, streptozocin, busulfan, mannosulfan, treosulfan, carboquone, N,N'N'-triethylenethiophosphoramide, triaziquone, triethylenemelamine); alkylating-like agents (e.g., carboplatin (branded/marketed as PARAPLATIN.RTM.), cisplatin, oxaliplatin, nedaplatin, triplatin tetranitrate, satraplatin, picoplatin); nonclassical DNA crosslinkers (e.g., procarbazine, dacarbazine, temozolomide (branded/marketed as TEMODAR.RTM.), altretamine, mitobronitol); and intercalating agents (e.g., actinomycin, bleomycin, mitomycin, and plicamycin).
[0350] Non-limiting examples of other therapies that can be administered to a subject in combination with the peptidomimetic macrocycles include: (1) a statin such as lovastatin (e.g., branded/marketed as MEVACOR.RTM.); (2) an mTOR inhibitor such as sirolimus which is also known as Rapamycin (e.g., branded/marketed as RAPAMUNE.RTM.), temsirolimus (e.g., branded/marketed as TORISEL.RTM.), evorolimus (e.g., branded/marketed as AFINITOR.RTM.), and deforolimus; (3) a famesyltransferase inhibitor agent such as tipifarnib; (4) an antifibrotic agent such as pirfenidone; (5) a pegylated interferon such as PEG-interferon alfa-2b; (6) a CNS stimulant such as methylphenidate (branded/marketed as RITALIN.RTM.); (7) a HER-2 antagonist such as anti-HER-2 antibody (e.g., trastuzumab) and kinase inhibitor (e.g., lapatinib); (8) an IGF-1 antagonist such as an anti-IGF-1 antibody (e.g., AVE1642 and IMC-A11) or an IGF-1 kinase inhibitor; (9) EGFR/HER-1 antagonist such as an anti-EGFR antibody (e.g., cetuximab, panitumamab) or EGFR kinase inhibitor (e.g., erlotinib; gefitinib); (10) SRC antagonist such as bosutinib; (11) cyclin dependent kinase (CDK) inhibitor such as seliciclib; (12) Janus kinase 2 inhibitor such as lestaurtinib; (13) proteasome inhibitor such as bortezomib; (14) phosphodiesterase inhibitor such as anagrelide; (15) inosine monophosphate dehydrogenase inhibitor such as tiazofurine; (16) lipoxygenase inhibitor such as masoprocol; (17) endothelin antagonist; (18) retinoid receptor antagonist such as tretinoin or alitretinoin; (19) immune modulator such as lenalidomide, pomalidomide, or thalidomide; (20) kinase (e.g., tyrosine kinase) inhibitor such as imatinib, dasatinib, erlotinib, nilotinib, gefitinib, sorafenib, sunitinib, lapatinib, or TG100801; (21) non-steroidal anti-inflammatory agent such as celecoxib (branded/marketed as CELEBREX.RTM.); (22) human granulocyte colony-stimulating factor (G-CSF) such as filgrastim (branded/marketed as NEUPOGEN.RTM.); (23) folinic acid or leucovorin calcium; (24) integrin antagonist such as an integrin .alpha.5.beta.1-antagonist (e.g., JSM6427); (25) nuclear factor kappa beta (NF-.kappa..beta.) antagonist such as OT-551, which is also an anti-oxidant. (26) hedgehog inhibitor such as CUR61414, cyclopamine, GDC-0449, and anti-hedgehog antibody; (27) histone deacetylase (HDAC) inhibitor such as SAHA (also known as vorinostat (branded/marketed as ZOLINZA)), PCI-24781, SB939, CHR-3996, CRA-024781, ITF2357, JNJ-26481585, or PCI-24781; (28) retinoid such as isotretinoin (e.g., branded/marketed as ACCUTANE.RTM.); (29) hepatocyte growth factor/scatter factor (HGF/SF) antagonist such as HGF/SF monoclonal antibody (e.g., AMG 102); (30) synthetic chemical such as antineoplaston; (31) anti-diabetic such as rosaiglitazone (e.g., branded/marketed as AVANDIA.RTM.); (32) antimalarial and amebicidal drug such as chloroquine (e.g., branded/marketed as ARALEN.RTM.); (33) synthetic bradykinin such as RMP-7; (34) platelet-derived growth factor receptor inhibitor such as SU-101; (35) receptor tyrosine kinase inhibitorsof Flk-1/KDR/VEGFR2, FGFR1 and PDGFR beta such as SU5416 and SU6668; (36) anti-inflammatory agent such as sulfasalazine (e.g., branded/marketed as AZULFIDINE.RTM.); and (37) TGF-beta antisense therapy.
[0351] In some embodiments a peptidomimetic macrocycles disclosed herein can inhibit one or more transporter enzymes (e.g., OATP1B1, OATP1B3, BSEP) at concentrations that can be clinically relevant. Therefore such a peptidomimetic macrocycles disclosed herein can interact with medications that are predominantly cleared by hepatobiliary transporters. In particular, methotrexate and statins (e.g., atorvastatin, fluvastatin lovastatin, pitavastatin pravastatin, rosuvastatin and simvastatin) may not be dosed within 48 h, 36 h, 24 h, or 12 h ((for example within 24 h) of the administration of such a peptidomimetic macrocycle. Exemplary medications that can be affected by co-administration of such a peptidomimetic macrocycles are listed below. In various embodiments one or more of the medications selected from Table 2 may not be dosed within 48 h, 36 h, 24 h, or 12 h (for example within 24 h) of the administration of such a peptidomimetic macrocycle.
TABLE-US-00004 TABLE 2 Exemplary medications that can be affected by co-administration with a peptidomimetic macrocycles disclosed herein. Medication Therapeutic Area Irinotecan Oncology Bosentan Pulmonary artery hypertension Caspofungin Antifungal Methotrexate Oncology & rheumatology Repaglinide Diabetes mellitus Atorvastatin Hypercholesterolemia Cerivastatin Hypercholesterolemia Fluvastatin Hypercholesterolemia Lovastatin Hypercholesterolemia Pitavastatin Hypercholesterolemia Pravastatin Hypercholesterolemia Rosuvastatin Hypercholesterolemia Simvastatin Hypercholesterolemia
EXAMPLES
Example 1: Peptidomimetic Macrocycles
[0352] Peptidomimetic macrocycles were synthesized, purified and analyzed as previously described and as described below (Schafmeister et al., J. Am. Chem. Soc. 122:5891-5892 (2000); Schafmeister & Verdine, J. Am. Chem. Soc. 122:5891 (2005); Walensky et al., Science 305:1466-1470 (2004); and U.S. Pat. No. 7,192,713). Peptidomimetic macrocycles were designed by replacing two or more naturally occurring amino acids with the corresponding synthetic amino acids. Substitutions were made at i and i+4, and i and i+7 positions. Peptide synthesis was performed either manually or on an automated peptide synthesizer (Applied Biosystems, model 433A), using solid phase conditions, rink amide AM resin (Novabiochem), and Fmoc main-chain protecting group chemistry. For the coupling of natural Fmoc-protected amino acids (Novabiochem), 10 equivalents of amino acid and a 1:1:2 molar ratio of coupling reagents HBTU/HOBt (Novabiochem)/DIEA were employed. Non-natural amino acids (4 equiv) were coupled with a 1:1:2 molar ratio of HATU (Applied Biosystems)/HOBt/DIEA. The N-termini of the synthetic peptides were acetylated, while the C-termini were amidated.
[0353] Purification of cross-linked compounds was achieved by high performance liquid chromatography (HPLC) (Varian ProStar) on a reverse phase C18 column (Varian) to yield the pure compounds. Chemical composition of the pure products was confirmed by LC/MS mass spectrometry (Micromass LCT interfaced with Agilent 1100 HPLC system) and amino acid analysis (Applied Biosystems, model 420A).
[0354] The following protocol was used in the synthesis of dialkyne-crosslinked peptidomimetic macrocycles, including SP662, SP663 and SP664. Fully protected resin-bound peptides were synthesized on a PEG-PS resin (loading 0.45 mmol/g) on a 0.2 mmol scale. Deprotection of the temporary Fmoc group was achieved by 3.times.10 min treatments of the resin bound peptide with 20% (v/v) piperidine in DMF. After washing with NMP (3.times.), dichloromethane (3.times.) and NMP (3.times.), coupling of each successive amino acid was achieved with 1.times.60 min incubation with the appropriate preactivated Fmoc-amino acid derivative. All protected amino acids (0.4 mmol) were dissolved in NMP and activated with HCTU (0.4 mmol) and DIEA (0.8 mmol) prior to transfer of the coupling solution to the deprotected resin-bound peptide. After coupling was completed, the resin was washed in preparation for the next deprotection/coupling cycle. Acetylation of the amino terminus was carried out in the presence of acetic anhydride/DIEA in NMP. The LC-MS analysis of a cleaved and deprotected sample obtained from an aliquot of the fully assembled resin-bound peptide was accomplished in order to verifying the completion of each coupling. In a typical example, tetrahydrofuran (4 ml) and triethylamine (2 ml) were added to the peptide resin (0.2 mmol) in a 40 ml glass vial and shaken for 10 minutes. Pd(PPh.sub.3).sub.2Cl.sub.2 (0.014 g, 0.02 mmol) and copper iodide (0.008 g, 0.04 mmol) were then added and the resulting reaction mixture was mechanically shaken 16 hours while open to atmosphere. The diyne-cyclized resin-bound peptides were deprotected and cleaved from the solid support by treatment with TFA/H.sub.2O/TIS (95/5/5 v/v) for 2.5 h at room temperature. After filtration of the resin the TFA solution was precipitated in cold diethyl ether and centrifuged to yield the desired product as a solid. The crude product was purified by preparative HPLC.
[0355] The following protocol was used in the synthesis of single alkyne-crosslinked peptidomimetic macrocycles, including SP665. Fully protected resin-bound peptides were synthesized on a Rink amide MBHA resin (loading 0.62 mmol/g) on a 0.1 mmol scale. Deprotection of the temporary Fmoc group was achieved by 2.times.20 min treatments of the resin bound peptide with 25% (v/v) piperidine in NMP. After extensive flow washing with NMP and dichloromethane, coupling of each successive amino acid was achieved with 1.times.60 min incubation with the appropriate preactivated Fmoc-amino acid derivative. All protected amino acids (1 mmol) were dissolved in NMP and activated with HCTU (1 mmol) and DIEA (1 mmol) prior to transfer of the coupling solution to the deprotected resin-bound peptide. After coupling was completed, the resin was extensively flow washed in preparation for the next deprotection/coupling cycle. Acetylation of the amino terminus was carried out in the presence of acetic anhydride/DIEA in NMP/NMM. The LC-MS analysis of a cleaved and deprotected sample obtained from an aliquot of the fully assembled resin-bound peptide was accomplished in order to verifying the completion of each coupling. In a typical example, the peptide resin (0.1 mmol) was washed with DCM. Resin was loaded into a microwave vial. The vessel was evacuated and purged with nitrogen. Molybdenumhexacarbonyl (0.01 eq, Sigma Aldrich 199959) was added. Anhydrous chlorobenzene was added to the reaction vessel. Then 2-fluorophenol (1 eq, Sigma Aldrich F 12804) was added. The reaction was then loaded into the microwave and held at 130.degree. C. for 10 minutes. Reaction may need to be pushed a subsequent time for completion. The alkyne metathesized resin-bound peptides were deprotected and cleaved from the solid support by treatment with TFA/H.sub.2O/TIS (94/3/3 v/v) for 3 h at room temperature. After filtration of the resin the TFA solution was precipitated in cold diethyl ether and centrifuged to yield the desired product as a solid. The crude product was purified by preparative HPLC.
[0356] Table 3 shows a list of peptidomimetic macrocycles prepared.
TABLE-US-00005 TABLE 3 SEQ ID Exact Found Calc Calc Calc Sequence NO: Isomer Mass Mass (M + 1)/1 (M + 2)/2 (M + 3)/3 1 Ac-F$r8AYWEAc3cL$AAA-NH.sub.2 10 1456.78 729.44 1457.79 729.4 486.6 2 Ac-F$r8AYWEAc3cL$AAibA-NH.sub.2 11 1470.79 736.4 1471.8 736.4 491.27 3 Ac-LTF$r8AYWAQL$SANle-NH.sub.2 12 1715.97 859.02 1716.98 858.99 573 4 Ac-LTF$r8AYWAQL$SAL-NH.sub.2 13 1715.97 859.02 1716.98 858.99 573 5 Ac-LTF$r8AYWAQL$SAM-NH.sub.2 14 1733.92 868.48 1734.93 867.97 578.98 6 Ac-LTF$r8AYWAQL$SAhL-NH.sub.2 15 1729.98 865.98 1730.99 866 577.67 7 Ac-LTF$r8AYWAQL$SAF-NH.sub.2 16 1749.95 876.36 1750.96 875.98 584.32 8 Ac-LTF$r8AYWAQL$SAI-NH.sub.2 17 1715.97 859.02 1716.98 858.99 573 9 Ac-LTF$r8AYWAQL$SAChg-NH.sub.2 18 1741.98 871.98 1742.99 872 581.67 10 Ac-LTF$r8AYWAQL$SAAib-NH.sub.2 19 1687.93 845.36 1688.94 844.97 563.65 11 Ac-LTF$r8AYWAQL$SAA-NH.sub.2 20 1673.92 838.01 1674.93 837.97 558.98 12 Ac-LTF$r8AYWA$L$S$Nle-NH.sub.2 21 1767.04 884.77 1768.05 884.53 590.02 13 Ac-LTF$r8AYWA$L$S$A-NH.sub.2 22 1724.99 864.23 1726 863.5 576 14 Ac-F$r8AYWEAc3cL$AANle-NH.sub.2 23 1498.82 750.46 1499.83 750.42 500.61 15 Ac-F$r8AYWEAc3cL$AAL-NH.sub.2 24 1498.82 750.46 1499.83 750.42 500.61 16 Ac-F$r8AYWEAc3cL$AAM-NH.sub.2 25 1516.78 759.41 1517.79 759.4 506.6 17 Ac-F$r8AYWEAc3cL$AAhL-NH.sub.2 26 1512.84 757.49 1513.85 757.43 505.29 18 Ac-F$r8AYWEAc3cL$AAF-NH.sub.2 27 1532.81 767.48 1533.82 767.41 511.94 19 Ac-F$r8AYWEAc3cL$AAI-NH.sub.2 28 1498.82 750.39 1499.83 750.42 500.61 20 Ac-F$r8AYWEAc3cL$AAChg-NH.sub.2 29 1524.84 763.48 1525.85 763.43 509.29 21 Ac-F$r8AYWEAc3cL$AACha-NH.sub.2 30 1538.85 770.44 1539.86 770.43 513.96 22 Ac-F$r8AYWEAc3cL$AAAib-NH.sub.2 31 1470.79 736.84 1471.8 736.4 491.27 23 Ac-LTF$r8AYWAQL$AAAibV-NH.sub.2 32 1771.01 885.81 1772.02 886.51 591.34 24 Ac-LTF$r8AYWAQL$AAAibV-NH.sub.2 33 iso2 1771.01 886.26 1772.02 886.51 591.34 25 Ac-LTF$r8AYWAQL$SAibAA-NH.sub.2 34 1758.97 879.89 1759.98 880.49 587.33 26 Ac-LTF$r8AYWAQL$SAibAA-NH.sub.2 35 iso2 1758.97 880.34 1759.98 880.49 587.33 27 Ac-HLTF$r8HHWHQL$AANleNle-NH.sub.2 36 2056.15 1028.86 2057.16 1029.08 686.39 28 Ac-DLTF$r8HHWHQL$RRLV-NH.sub.2 37 2190.23 731.15 2191.24 1096.12 731.08 29 Ac-HHTF$r8HHWHQL$AAML-NH.sub.2 38 2098.08 700.43 2099.09 1050.05 700.37 30 Ac-F$r8HHWHQL$RRDCha-NH.sub.2 39 1917.06 959.96 1918.07 959.54 640.03 31 Ac-F$r8HHWHQL$HRFV-NH.sub.2 40 1876.02 938.65 1877.03 939.02 626.35 32 Ac-HLTF$r8HHWHQL$AAhLA-NH.sub.2 41 2028.12 677.2 2029.13 1015.07 677.05 33 Ac-DLTF$r8HHWHQL$RRChgl-NH.sub.2 42 2230.26 1115.89 2231.27 1116.14 744.43 34 Ac-DLTF$r8HHWHQL$RRChgl-NH.sub.2 43 iso2 2230.26 1115.96 2231.27 1116.14 744.43 35 Ac-HHTF$r8HHWHQL$AAChav-NH.sub.2 44 2106.14 1053.95 2107.15 1054.08 703.05 36 Ac-F$r8HHWHQL$RRDa-NH.sub.2 45 1834.99 918.3 1836 918.5 612.67 37 Ac-F$r8HHWHQL$HRAibG-NH.sub.2 46 1771.95 886.77 1772.96 886.98 591.66 38 Ac-F$r8AYWAQL$HHNleL-NH.sub.2 47 1730.97 866.57 1731.98 866.49 578 39 Ac-F$r8AYWSAL$HQANle-NH.sub.2 48 1638.89 820.54 1639.9 820.45 547.3 40 Ac-F$r8AYWVQL$QHChgl-NH.sub.2 49 1776.01 889.44 1777.02 889.01 593.01 41 Ac-F$r8AYWTAL$QQNlev-NH.sub.2 50 1671.94 836.97 1672.95 836.98 558.32 42 Ac-F$r8AYWYQL$HAibAa-NH.sub.2 51 1686.89 844.52 1687.9 844.45 563.3 43 Ac-LTF$r8AYWAQL$HHLa-NH.sub.2 52 1903.05 952.27 1904.06 952.53 635.36 44 Ac-LTF$r8AYWAQL$HHLa-NH.sub.2 53 iso2 1903.05 952.27 1904.06 952.53 635.36 45 Ac-LTF$r8AYWAQL$HQNlev-NH.sub.2 54 1922.08 962.48 1923.09 962.05 641.7 46 Ac-LTF$r8AYWAQL$HQNlev-NH.sub.2 55 iso2 1922.08 962.4 1923.09 962.05 641.7 47 Ac-LTF$r8AYWAQL$QQMl-NH.sub.2 56 1945.05 973.95 1946.06 973.53 649.36 48 Ac-LTF$r8AYWAQL$QQMl-NH.sub.2 57 iso2 1945.05 973.88 1946.06 973.53 649.36 49 Ac-LTF$r8AYWAQL$HAibhLV-NH.sub.2 58 1893.09 948.31 1894.1 947.55 632.04 50 Ac-LTF$r8AYWAQL$AHFA-NH.sub.2 59 1871.01 937.4 1872.02 936.51 624.68 51 Ac-HLTF$r8HHWHQL$AANlel-NH.sub.2 60 2056.15 1028.79 2057.16 1029.08 686.39 52 Ac-DLTF$r8HHWHQL$RRLa-NH.sub.2 61 2162.2 721.82 2163.21 1082.11 721.74 53 Ac-HHTF$r8HHWHQL$AAMv-NH.sub.2 62 2084.07 1042.92 2085.08 1043.04 695.7 54 Ac-F$r8HHWHQL$RRDA-NH.sub.2 63 1834.99 612.74 1836 918.5 612.67 55 Ac-F$r8HHWHQL$HRFCha-NH.sub.2 64 1930.06 966.47 1931.07 966.04 644.36 56 Ac-F$r8AYWEAL$AA-NHAm 65 1443.82 1445.71 1444.83 722.92 482.28 57 Ac-F$r8AYWEAL$AA-NHiAm 66 1443.82 723.13 1444.83 722.92 482.28 58 Ac-F$r8AYWEAL$AA-NHnPr3Ph 67 1491.82 747.3 1492.83 746.92 498.28 59 Ac-F$r8AYWEAL$AA-NHnBu33Me 68 1457.83 1458.94 1458.84 729.92 486.95 60 Ac-F$r8AYWEAL$AA-NHnPr 69 1415.79 709.28 1416.8 708.9 472.94 61 Ac-F$r8AYWEAL$AA-NHnEt2Ch 70 1483.85 1485.77 1484.86 742.93 495.62 62 Ac-F$r8AYWEAL$AA-NHnEt2Cp 71 1469.83 1470.78 1470.84 735.92 490.95 63 Ac-F$r8AYWEAL$AA-NHHex 72 1457.83 730.19 1458.84 729.92 486.95 64 Ac-LTF$r8AYWAQL$AAIA-NH.sub.2 73 1771.01 885.81 1772.02 886.51 591.34 65 Ac-LTF$r8AYWAQL$AAIA-NH.sub.2 74 iso2 1771.01 866.8 1772.02 886.51 591.34 66 Ac-LTF$r8AYWAAL$AAMA-NH.sub.2 75 1731.94 867.08 1732.95 866.98 578.32 67 Ac-LTF$r8AYWAAL$AAMA-NH.sub.2 76 iso2 1731.94 867.28 1732.95 866.98 578.32 68 Ac-LTF$r8AYWAQL$AANleA-NH.sub.2 77 1771.01 867.1 1772.02 886.51 591.34 69 Ac-LTF$r8AYWAQL$AANleA-NH.sub.2 78 iso2 1771.01 886.89 1772.02 886.51 591.34 70 Ac-LTF$r8AYWAQL$AAIa-NH.sub.2 79 1771.01 886.8 1772.02 886.51 591.34 71 Ac-LTF$r8AYWAQL$AAIa-NH.sub.2 80 iso2 1771.01 887.09 1772.02 886.51 591.34 72 Ac-LTF$r8AYWAAL$AAMa-NH.sub.2 81 1731.94 867.17 1732.95 866.98 578.32 73 Ac-LTF$r8AYWAAL$AAMa-NH.sub.2 82 iso2 1731.94 867.37 1732.95 866.98 578.32 74 Ac-LTF$r8AYWAQL$AANlea-NH.sub.2 83 1771.01 887.08 1772.02 886.51 591.34 75 Ac-LTF$r8AYWAQL$AANlea-NH.sub.2 84 iso2 1771.01 887.08 1772.02 886.51 591.34 76 Ac-LTF$r8AYWAAL$AAIv-NH.sub.2 85 1742.02 872.37 1743.03 872.02 581.68 77 Ac-LTF$r8AYWAAL$AAIv-NH.sub.2 86 iso2 1742.02 872.74 1743.03 872.02 581.68 78 Ac-LTF$r8AYWAQL$AAMv-NH.sub.2 87 1817 910.02 1818.01 909.51 606.67 79 Ac-LTF$r8AYWAAL$AANlev-NH.sub.2 88 1742.02 872.37 1743.03 872.02 581.68 80 Ac-LTF$r8AYWAAL$AANlev-NH.sub.2 89 iso2 1742.02 872.28 1743.03 872.02 581.68 81 Ac-LTF$r8AYWAQL$AAIl-NH.sub.2 90 1813.05 907.81 1814.06 907.53 605.36 82 Ac-LTF$r8AYWAQL$AAIl-NH.sub.2 91 iso2 1813.05 907.81 1814.06 907.53 605.36 83 Ac-LTF$r8AYWAAL$AAMl-NH.sub.2 92 1773.99 887.37 1775 888 592.34 84 Ac-LTF$r8AYWAQL$AANlel-NH.sub.2 93 1813.05 907.61 1814.06 907.53 605.36 85 Ac-LTF$r8AYWAQL$AANlel-NH.sub.2 94 iso2 1813.05 907.71 1814.06 907.53 605.36 86 Ac-F$r8AYWEAL$AAMA-NH.sub.2 95 1575.82 789.02 1576.83 788.92 526.28 87 Ac-F$r8AYWEAL$AANleA-NH.sub.2 96 1557.86 780.14 1558.87 779.94 520.29 88 Ac-F$r8AYWEAL$AAIa-NH.sub.2 97 1557.86 780.33 1558.87 779.94 520.29 89 Ac-F$r8AYWEAL$AAMa-NH.sub.2 98 1575.82 789.3 1576.83 788.92 526.28 90 Ac-F$r8AYWEAL$AANlea-NH.sub.2 99 1557.86 779.4 1558.87 779.94 520.29 91 Ac-F$r8AYWEAL$AAIv-NH.sub.2 100 1585.89 794.29 1586.9 793.95 529.64 92 Ac-F$r8AYWEAL$AAMv-NH.sub.2 101 1603.85 803.08 1604.86 802.93 535.62 93 Ac-F$r8AYWEAL$AANlev-NH.sub.2 102 1585.89 793.46 1586.9 793.95 529.64 94 Ac-F$r8AYWEAL$AAIl-NH.sub.2 103 1599.91 800.49 1600.92 800.96 534.31 95 Ac-F$r8AYWEAL$AAMl-NH.sub.2 104 1617.86 809.44 1618.87 809.94 540.29 96 Ac-F$r8AYWEAL$AANlel-NH.sub.2 105 1599.91 801.7 1600.92 800.96 534.31 97 Ac-F$r8AYWEAL$AANlel-NH.sub.2 106 iso2 1599.91 801.42 1600.92 800.96 534.31 98 Ac-LTF$r8AY6clWAQL$SAA-NH.sub.2 107 1707.88 855.72 1708.89 854.95 570.3 99 Ac-LTF$r8AY6clWAQL$SAA-NH.sub.2 108 iso2 1707.88 855.35 1708.89 854.95 570.3 100 Ac-WTF$r8FYWSQL$AVAa-NH.sub.2 109 1922.01 962.21 1923.02 962.01 641.68 101 Ac-WTF$r8FYWSQL$AVAa-NH.sub.2 110 iso2 1922.01 962.49 1923.02 962.01 641.68 102 Ac-WTF$r8VYWSQL$AVA-NH.sub.2 111 1802.98 902.72 1803.99 902.5 602 103 Ac-WTF$r8VYWSQL$AVA-NH.sub.2 112 iso2 1802.98 903 1803.99 902.5 602 104 Ac-WTF$r8FYWSQL$SAAa-NH.sub.2 113 1909.98 956.47 1910.99 956 637.67 105 Ac-WTF$r8FYWSQL$SAAa-NH.sub.2 114 iso2 1909.98 956.47 1910.99 956 637.67 106 Ac-WTF$r8VYWSQL$AVAaa-NH.sub.2 115 1945.05 974.15 1946.06 973.53 649.36 107 Ac-WTF$r8VYWSQL$AVAaa-NH.sub.2 116 iso2 1945.05 973.78 1946.06 973.53 649.36 108 Ac-LTF$r8AYWAQL$AVG-NH.sub.2 117 1671.94 837.52 1672.95 836.98 558.32 109 Ac-LTF$r8AYWAQL$AVG-NH.sub.2 118 iso2 1671.94 837.21 1672.95 836.98 558.32 110 Ac-LTF$r8AYWAQL$AVQ-NH.sub.2 119 1742.98 872.74 1743.99 872.5 582 111 Ac-LTF$r8AYWAQL$AVQ-NH.sub.2 120 iso2 1742.98 872.74 1743.99 872.5 582 112 Ac-LTF$r8AYWAQL$SAa-NH.sub.2 121 1673.92 838.23 1674.93 837.97 558.98 113 Ac-LTF$r8AYWAQL$SAa-NH.sub.2 122 iso2 1673.92 838.32 1674.93 837.97 558.98 114 Ac-LTF$r8AYWAQhL$SAA-NH.sub.2 123 1687.93 844.37 1688.94 844.97 563.65 115 Ac-LTF$r8AYWAQhL$SAA-NH.sub.2 124 iso2 1687.93 844.81 1688.94 844.97 563.65 116 Ac-LTF$r8AYWEQLStSA$-NH.sub.2 125 1826 905.27 1827.01 914.01 609.67 117 Ac-LTF$r8AYWAQL$SLA-NH.sub.2 126 1715.97 858.48 1716.98 858.99 573 118 Ac-LTF$r8AYWAQL$SLA-NH.sub.2 127 iso2 1715.97 858.87 1716.98 858.99 573 119 Ac-LTF$r8AYWAQL$SWA-NH.sub.2 128 1788.96 895.21 1789.97 895.49 597.33 120 Ac-LTF$r8AYWAQL$SWA-NH.sub.2 129 iso2 1788.96 895.28 1789.97 895.49 597.33 121 Ac-LTF$r8AYWAQL$SVS-NH.sub.2 130 1717.94 859.84 1718.95 859.98 573.65 122 Ac-LTF$r8AYWAQL$SAS-NH.sub.2 131 1689.91 845.85 1690.92 845.96 564.31 123 Ac-LTF$r8AYWAQL$SVG-NH.sub.2 132 1687.93 844.81 1688.94 844.97 563.65 124 Ac-ETF$r8VYWAQL$SAa-NH.sub.2 133 1717.91 859.76 1718.92 859.96 573.64 125 Ac-ETF$r8VYWAQL$SAA-NH.sub.2 134 1717.91 859.84 1718.92 859.96 573.64 126 Ac-ETF$r8VYWAQL$SVA-NH.sub.2 135 1745.94 873.82 1746.95 873.98 582.99 127 Ac-ETF$r8VYWAQL$SLA-NH.sub.2 136 1759.96 880.85 1760.97 880.99 587.66 128 Ac-ETF$r8VYWAQL$SWA-NH.sub.2 137 1832.95 917.34 1833.96 917.48 611.99 129 Ac-ETF$r8KYWAQL$SWA-NH.sub.2 138 1861.98 931.92 1862.99 932 621.67 130 Ac-ETF$r8VYWAQL$SVS-NH.sub.2 139 1761.93 881.89 1762.94 881.97 588.32 131 Ac-ETF$r8VYWAQL$SAS-NH.sub.2 140 1733.9 867.83 1734.91 867.96 578.97 132 Ac-ETF$r8VYWAQL$SVG-NH.sub.2 141 1731.92 866.87 1732.93 866.97 578.31 133 Ac-LTF$r8VYWAQL$SSa-NH.sub.2 142 1717.94 859.47 1718.95 859.98 573.65 134 Ac-ETF$r8VYWAQL$SSa-NH.sub.2 143 1733.9 867.83 1734.91 867.96 578.97 135 Ac-LTF$r8VYWAQL$SNa-NH.sub.2 144 1744.96 873.38 1745.97 873.49 582.66 136 Ac-ETF$r8VYWAQL$SNa-NH.sub.2 145 1760.91 881.3 1761.92 881.46 587.98 137 Ac-LTF$r8VYWAQL$SAa-NH.sub.2 146 1701.95 851.84 1702.96 851.98 568.32 138 Ac-LTF$r8VYWAQL$SVA-NH.sub.2 147 1729.98 865.53 1730.99 866 577.67 139 Ac-LTF$r8VYWAQL$SVA-NH.sub.2 148 iso2 1729.98 865.9 1730.99 866 577.67 140 Ac-LTF$r8VYWAQL$SWA-NH.sub.2 149 1816.99 909.42 1818 909.5 606.67 141 Ac-LTF$r8VYWAQL$SVS-NH.sub.2 150 1745.98 873.9 1746.99 874 583 142 Ac-LTF$r8VYWAQL$SVS-NH.sub.2 151 iso2 1745.98 873.9 1746.99 874 583 143 Ac-LTF$r8VYWAQL$SAS-NH.sub.2 152 1717.94 859.84 1718.95 859.98 573.65 144 Ac-LTF$r8VYWAQL$SAS-NH.sub.2 153 iso2 1717.94 859.91 1718.95 859.98 573.65 145 Ac-LTF$r8VYWAQL$SVG-NH.sub.2 154 1715.97 858.87 1716.98 858.99 573 146 Ac-LTF$r8VYWAQL$SVG-NH.sub.2 155 iso2 1715.97 858.87 1716.98 858.99 573 147 Ac-LTF$r8EYWAQCha$SAA-NH.sub.2 156 1771.96 886.85 1772.97 886.99 591.66 148 Ac-LTF$r8EYWAQCha$SAA-NH.sub.2 157 iso2 1771.96 886.85 1772.97 886.99 591.66 149 Ac-LTF$r8EYWAQCpg$SAA-NH.sub.2 158 1743.92 872.86 1744.93 872.97 582.31 150 Ac-LTF$r8EYWAQCpg$SAA-NH.sub.2 159 iso2 1743.92 872.86 1744.93 872.97 582.31 151 Ac-LTF$r8EYWAQF$SAA-NH.sub.2 160 1765.91 883.44 1766.92 883.96 589.64 152 Ac-LTF$r8EYWAQF$SAA-NH.sub.2 161 iso2 1765.91 883.89 1766.92 883.96 589.64 153 Ac-LTF$r8EYWAQCba$SAA-NH.sub.2 162 1743.92 872.42 1744.93 872.97 582.31 154 Ac-LTF$r8EYWAQCba$SAA-NH.sub.2 163 iso2 1743.92 873.39 1744.93 872.97 582.31 155 Ac-LTF3Cl$r8EYWAQL$SAA-NH.sub.2 164 1765.89 883.89 1766.9 883.95 589.64 156 Ac-LTF3Cl$r8EYWAQL$SAA-NH.sub.2 165 iso2 1765.89 883.96 1766.9 883.95 589.64 157 Ac-LTF34F2$r8EYWAQL$SAA-NH.sub.2 166 1767.91 884.48 1768.92 884.96 590.31 158 Ac-LTF34F2$r8EYWAQL$SAA-NH.sub.2 167 iso2 1767.91 884.48 1768.92 884.96 590.31
159 Ac-LTF34F2$r8EYWAQhL$SAA-NH.sub.2 168 1781.92 891.44 1782.93 891.97 594.98 160 Ac-LTF34F2$r8EYWAQhL$SAA-NH.sub.2 169 iso2 1781.92 891.88 1782.93 891.97 594.98 161 Ac-ETF$r8EYWAQL$SAA-NH.sub.2 170 1747.88 874.34 1748.89 874.95 583.63 162 Ac-LTF$r8AYWVQL$SAA-NH.sub.2 171 1701.95 851.4 1702.96 851.98 568.32 163 Ac-LTF$r8AHWAQL$SAA-NH.sub.2 172 1647.91 824.83 1648.92 824.96 550.31 164 Ac-LTF$r8AEWAQL$SAA-NH.sub.2 173 1639.9 820.39 1640.91 820.96 547.64 165 Ac-LTF$r8ASWAQL$SAA-NH.sub.2 174 1597.89 799.38 1598.9 799.95 533.64 166 Ac-LTF$r8AEWAQL$SAA-NH.sub.2 175 iso2 1639.9 820.39 1640.91 820.96 547.64 167 Ac-LTF$r8ASWAQL$SAA-NH.sub.2 176 iso2 1597.89 800.31 1598.9 799.95 533.64 168 Ac-LTF$r8AF4coohWAQL$SAA-NH.sub.2 177 1701.91 851.4 1702.92 851.96 568.31 169 Ac-LTF$r8AF4coohWAQL$SAA-NH.sub.2 178 iso2 1701.91 851.4 1702.92 851.96 568.31 170 Ac-LTF$r8AHWAQL$AAIa-NH.sub.2 179 1745 874.13 1746.01 873.51 582.67 171 Ac-ITF$r8FYWAQL$AAIa-NH.sub.2 180 1847.04 923.92 1848.05 924.53 616.69 172 Ac-ITF$r8EHWAQL$AAIa-NH.sub.2 181 1803.01 903.17 1804.02 902.51 602.01 173 Ac-ITF$r8EHWAQL$AAIa-NH.sub.2 182 iso2 1803.01 903.17 1804.02 902.51 602.01 174 Ac-ETF$r8EHWAQL$AAIa-NH.sub.2 183 1818.97 910.76 1819.98 910.49 607.33 175 Ac-ETF$r8EHWAQL$AAIa-NH.sub.2 184 iso2 1818.97 910.85 1819.98 910.49 607.33 176 Ac-LTF$r8AHWVQL$AAIa-NH.sub.2 185 1773.03 888.09 1774.04 887.52 592.02 177 Ac-ITF$r8FYWVQL$AAIa-NH.sub.2 186 1875.07 939.16 1876.08 938.54 626.03 178 Ac-ITF$r8EYWVQL$AAIa-NH.sub.2 187 1857.04 929.83 1858.05 929.53 620.02 179 Ac-ITF$r8EHWVQL$AAIa-NH.sub.2 188 1831.04 916.86 1832.05 916.53 611.35 180 Ac-LTF$r8AEWAQL$AAIa-NH.sub.2 189 1736.99 869.87 1738 869.5 580 181 Ac-LTF$r8AF4coohWAQL$AAIa-NH.sub.2 190 1799 900.17 1800.01 900.51 600.67 182 Ac-LTF$r8AF4coohWAQL$AAIa-NH.sub.2 191 iso2 1799 900.24 1800.01 900.51 600.67 183 Ac-LTF$r8AHWAQL$AHFA-NH.sub.2 192 1845.01 923.89 1846.02 923.51 616.01 184 Ac-ITF$r8FYWAQL$AHFA-NH.sub.2 193 1947.05 975.05 1948.06 974.53 650.02 185 Ac-ITF$r8FYWAQL$AHFA-NH.sub.2 194 iso2 1947.05 976.07 1948.06 974.53 650.02 186 Ac-ITF$r8FHWAQL$AEFA-NH.sub.2 195 1913.02 958.12 1914.03 957.52 638.68 187 Ac-ITF$r8FHWAQL$AEFA-NH.sub.2 196 iso2 1913.02 957.86 1914.03 957.52 638.68 188 Ac-ITF$r8EHWAQL$AHFA-NH.sub.2 197 1903.01 952.94 1904.02 952.51 635.34 189 Ac-ITF$r8EHWAQL$AHFA-NH.sub.2 198 iso2 1903.01 953.87 1904.02 952.51 635.34 190 Ac-LTF$r8AHWVQL$AHFA-NH.sub.2 199 1873.04 937.86 1874.05 937.53 625.35 191 Ac-ITF$r8FYWVQL$AHFA-NH.sub.2 200 1975.08 988.83 1976.09 988.55 659.37 192 Ac-ITF$r8EYWVQL$AHFA-NH.sub.2 201 1957.05 979.35 1958.06 979.53 653.36 193 Ac-ITF$r8EHWVQL$AHFA-NH.sub.2 202 1931.05 967 1932.06 966.53 644.69 194 Ac-ITF$r8EHWVQL$AHFA-NH.sub.2 203 iso2 1931.05 967.93 1932.06 966.53 644.69 195 Ac-ETF$r8EYWAAL$SAA-NH.sub.2 204 1690.86 845.85 1691.87 846.44 564.63 196 Ac-LTF$r8AYWVAL$SAA-NH.sub.2 205 1644.93 824.08 1645.94 823.47 549.32 197 Ac-LTF$r8AHWAAL$SAA-NH.sub.2 206 1590.89 796.88 1591.9 796.45 531.3 198 Ac-LTF$r8AEWAAL$SAA-NH.sub.2 207 1582.88 791.9 1583.89 792.45 528.63 199 Ac-LTF$r8AEWAAL$SAA-NH.sub.2 208 iso2 1582.88 791.9 1583.89 792.45 528.63 200 Ac-LTF$r8ASWAAL$SAA-NH.sub.2 209 1540.87 770.74 1541.88 771.44 514.63 201 Ac-LTF$r8ASWAAL$SAA-NH.sub.2 210 iso2 1540.87 770.88 1541.88 771.44 514.63 202 Ac-LTF$r8AYWAAL$AAIa-NH.sub.2 211 1713.99 857.39 1715 858 572.34 203 Ac-LTF$r8AYWAAL$AAIa-NH.sub.2 212 iso2 1713.99 857.84 1715 858 572.34 204 Ac-LTF$r8AYWAAL$AHFA-NH.sub.2 213 1813.99 907.86 1815 908 605.67 205 Ac-LTF$r8EHWAQL$AHIa-NH.sub.2 214 1869.03 936.1 1870.04 935.52 624.02 206 Ac-LTF$r8EHWAQL$AHIa-NH.sub.2 215 iso2 1869.03 937.03 1870.04 935.52 624.02 207 Ac-LTF$r8AHWAQL$AHIa-NH.sub.2 216 1811.03 906.87 1812.04 906.52 604.68 208 Ac-LTF$r8EYWAQL$AHIa-NH.sub.2 217 1895.04 949.15 1896.05 948.53 632.69 209 Ac-LTF$r8AYWAQL$AAFa-NH.sub.2 218 1804.99 903.2 1806 903.5 602.67 210 Ac-LTF$r8AYWAQL$AAFa-NH.sub.2 219 iso2 1804.99 903.28 1806 903.5 602.67 211 Ac-LTF$r8AYWAQL$AAWa-NH.sub.2 220 1844 922.81 1845.01 923.01 615.67 212 Ac-LTF$r8AYWAQL$AAVa-NH.sub.2 221 1756.99 878.86 1758 879.5 586.67 213 Ac-LTF$r8AYWAQL$AAVa-NH.sub.2 222 iso2 1756.99 879.3 1758 879.5 586.67 214 Ac-LTF$r8AYWAQL$AALa-NH.sub.2 223 1771.01 886.26 1772.02 886.51 591.34 215 Ac-LTF$r8AYWAQL$AALa-NH.sub.2 224 iso2 1771.01 886.33 1772.02 886.51 591.34 216 Ac-LTF$r8EYWAQL$AAIa-NH.sub.2 225 1829.01 914.89 1830.02 915.51 610.68 217 Ac-LTF$r8EYWAQL$AAIa-NH.sub.2 226 iso2 1829.01 915.34 1830.02 915.51 610.68 218 Ac-LTF$r8EYWAQL$AAFa-NH.sub.2 227 1863 932.87 1864.01 932.51 622.01 219 Ac-LTF$r8EYWAQL$AAFa-NH.sub.2 228 iso2 1863 932.87 1864.01 932.51 622.01 220 Ac-LTF$r8EYWAQL$AAVa-NH.sub.2 229 1815 908.23 1816.01 908.51 606.01 221 Ac-LTF$r8EYWAQL$AAVa-NH.sub.2 230 iso2 1815 908.31 1816.01 908.51 606.01 222 Ac-LTF$r8EHWAQL$AAIa-NH.sub.2 231 1803.01 903.17 1804.02 902.51 602.01 223 Ac-LTF$r8EHWAQL$AAIa-NH.sub.2 232 iso2 1803.01 902.8 1804.02 902.51 602.01 224 Ac-LTF$r8EHWAQL$AAWa-NH.sub.2 233 1876 939.34 1877.01 939.01 626.34 225 Ac-LTF$r8EHWAQL$AAWa-NH.sub.2 234 iso2 1876 939.62 1877.01 939.01 626.34 226 Ac-LTF$r8EHWAQL$AALa-NH.sub.2 235 1803.01 902.8 1804.02 902.51 602.01 227 Ac-LTF$r8EHWAQL$AALa-NH.sub.2 236 iso2 1803.01 902.9 1804.02 902.51 602.01 228 Ac-ETF$r8EHWVQL$AALa-NH.sub.2 237 1847 924.82 1848.01 924.51 616.67 229 Ac-LTF$r8AYWAQL$AAAa-NH.sub.2 238 1728.96 865.89 1729.97 865.49 577.33 230 Ac-LTF$r8AYWAQL$AAAa-NH.sub.2 239 iso2 1728.96 865.89 1729.97 865.49 577.33 231 Ac-LTF$r8AYWAQL$AAAibA-NH.sub.2 240 1742.98 872.83 1743.99 872.5 582 232 Ac-LTF$r8AYWAQL$AAAibA-NH.sub.2 241 iso2 1742.98 872.92 1743.99 872.5 582 233 Ac-LTF$r8AYWAQL$AAAAa-NH.sub.2 242 1800 901.42 1801.01 901.01 601.01 234 Ac-LTF$r5AYWAQL$s8AAIa-NH.sub.2 243 1771.01 887.17 1772.02 886.51 591.34 235 Ac-LTF$r5AYWAQL$s8SAA-NH.sub.2 244 1673.92 838.33 1674.93 837.97 558.98 236 Ac-LTF$r8AYWAQCba$AANleA-NH.sub.2 245 1783.01 892.64 1784.02 892.51 595.34 237 Ac-ETF$r8AYWAQCba$AANleA-NH.sub.2 246 1798.97 900.59 1799.98 900.49 600.66 238 Ac-LTF$r8EYWAQCba$AANleA-NH.sub.2 247 1841.01 922.05 1842.02 921.51 614.68 239 Ac-LTF$r8AYWAQCba$AWNleA-NH.sub.2 248 1898.05 950.46 1899.06 950.03 633.69 240 Ac-ETF$r8AYWAQCba$AWNleA-NH.sub.2 249 1914.01 958.11 1915.02 958.01 639.01 241 Ac-LTF$r8EYWAQCba$AWNleA-NH.sub.2 250 1956.06 950.62 1957.07 979.04 653.03 242 Ac-LTF$r8EYWAQCba$SAFA-NH.sub.2 251 1890.99 946.55 1892 946.5 631.34 243 Ac-LTF34F2$r8EYWAQCba$SANleA-NH.sub.2 252 1892.99 947.57 1894 947.5 632 244 Ac-LTF$r8EF4coohWAQCba$SANleA-NH.sub.2 253 1885 943.59 1886.01 943.51 629.34 245 Ac-LTF$r8EYWSQCba$SANleA-NH.sub.2 254 1873 937.58 1874.01 937.51 625.34 246 Ac-LTF$r8EYWWQCba$SANleA-NH.sub.2 255 1972.05 987.61 1973.06 987.03 658.36 247 Ac-LTF$r8EYWAQCba$AAIa-NH.sub.2 256 1841.01 922.05 1842.02 921.51 614.68 248 Ac-LTF34F2$r8EYWAQCba$AAIa-NH.sub.2 257 1876.99 939.99 1878 939.5 626.67 249 Ac-LTF$r8EF4coohWAQCba$AAIa-NH.sub.2 258 1869.01 935.64 1870.02 935.51 624.01 250 Pam-ETF$r8EYWAQCba$SAA-NH.sub.2 259 1956.1 979.57 1957.11 979.06 653.04 251 Ac-LThF$r8EFWAQCba$SAA-NH.sub.2 260 1741.94 872.11 1742.95 871.98 581.65 252 Ac-LTA$r8EYWAQCba$SAA-NH.sub.2 261 1667.89 835.4 1668.9 834.95 556.97 253 Ac-LTF$r8EYAAQCba$SAA-NH.sub.2 262 1628.88 815.61 1629.89 815.45 543.97 254 Ac-LTF$r8EY2NalAQCba$SAA-NH.sub.2 263 1754.93 879.04 1755.94 878.47 585.98 255 Ac-LTF$r8AYWAQCba$SAA-NH.sub.2 264 1685.92 844.71 1686.93 843.97 562.98 256 Ac-LTF$r8EYWAQCba$SAF-NH.sub.2 265 1819.96 911.41 1820.97 910.99 607.66 257 Ac-LTF$r8EYWAQCba$SAFa-NH.sub.2 266 1890.99 947.41 1892 946.5 631.34 258 Ac-LTF$r8AYWAQCba$SAF-NH.sub.2 267 1761.95 882.73 1762.96 881.98 588.32 259 Ac-LTF34F2$r8AYWAQCba$SAF-NH.sub.2 268 1797.93 900.87 1798.94 899.97 600.32 260 Ac-LTF$r8AF4coohWAQCba$SAF-NH.sub.2 269 1789.94 896.43 1790.95 895.98 597.65 261 Ac-LTF$r8EY6clWAQCba$SAF-NH.sub.2 270 1853.92 929.27 1854.93 927.97 618.98 262 Ac-LTF$r8AYWSQCba$SAF-NH.sub.2 271 1777.94 890.87 1778.95 889.98 593.65 263 Ac-LTF$r8AYWWQCba$SAF-NH.sub.2 272 1876.99 939.91 1878 939.5 626.67 264 Ac-LTF$r8AYWAQCba$AAIa-NH.sub.2 273 1783.01 893.19 1784.02 892.51 595.34 265 Ac-LTF34F2$r8AYWAQCba$AAIa-NH.sub.2 274 1818.99 911.23 1820 910.5 607.34 266 Ac-LTF$r8AY6clWAQCba$AAIa-NH.sub.2 275 1816.97 909.84 1817.98 909.49 606.66 267 Ac-LTF$r8AF4coohWAQCba$AAIa-NH.sub.2 276 1811 906.88 1812.01 906.51 604.67 268 Ac-LTF$r8EYWAQCba$AAFa-NH.sub.2 277 1875 938.6 1876.01 938.51 626.01 269 Ac-LTF$r8EYWAQCba$AAFa-NH.sub.2 278 iso2 1875 938.6 1876.01 938.51 626.01 270 Ac-ETF$r8AYWAQCba$AWNlea-NH.sub.2 279 1914.01 958.42 1915.02 958.01 639.01 271 Ac-LTF$r8EYWAQCba$AWNlea-NH.sub.2 280 1956.06 979.42 1957.07 979.04 653.03 272 Ac-ETF$r8EYWAQCba$AWNlea-NH.sub.2 281 1972.01 987.06 1973.02 987.01 658.34 273 Ac-ETF$r8EYWAQCba$AWNlea-NH.sub.2 282 iso2 1972.01 987.06 1973.02 987.01 658.34 274 Ac-LTF$r8AYWAQCba$SAFa-NH.sub.2 283 1832.99 917.89 1834 917.5 612 275 Ac-LTF$r8AYWAQCba$SAFa-NH.sub.2 284 iso2 1832.99 918.07 1834 917.5 612 276 Ac-ETF$r8AYWAQL$AWNlea-NH.sub.2 285 1902.01 952.22 1903.02 952.01 635.01 277 Ac-LTF$r8EYWAQL$AWNlea-NH.sub.2 286 1944.06 973.5 1945.07 973.04 649.03 278 Ac-ETF$r8EYWAQL$AWNlea-NH.sub.2 287 1960.01 981.46 1961.02 981.01 654.34 279 Dmaac-LTF$r8EYWAQhL$SAA-NH.sub.2 288 1788.98 896.06 1789.99 895.5 597.33 280 Hexac-LTF$r8EYWAQhL$SAA-NH.sub.2 289 1802 902.9 1803.01 902.01 601.67 281 Napac-LTF$r8EYWAQhL$SAA-NH.sub.2 290 1871.99 937.58 1873 937 625 282 Decac-LTF$r8EYWAQhL$SAA-NH.sub.2 291 1858.06 930.55 1859.07 930.04 620.36 283 Admac-LTF$r8EYWAQhL$SAA-NH.sub.2 292 1866.03 934.07 1867.04 934.02 623.02 284 Tmac-LTF$r8EYWAQhL$SAA-NH.sub.2 293 1787.99 895.41 1789 895 597 285 Pam-LTF$r8EYWAQhL$SAA-NH.sub.2 294 1942.16 972.08 1943.17 972.09 648.39 286 Ac-LTF$r8AYWAQCba$AANleA-NH.sub.2 295 iso2 1783.01 892.64 1784.02 892.51 595.34 287 Ac-LTF34F2$r8EYWAQCba$AAIa-NH.sub.2 296 iso2 1876.99 939.62 1878 939.5 626.67 288 Ac-LTF34F2$r8EYWAQCba$SAA-NH.sub.2 297 1779.91 892.07 1780.92 890.96 594.31 289 Ac-LTF34F2$r8EYWAQCba$SAA-NH.sub.2 298 iso2 1779.91 891.61 1780.92 890.96 594.31 290 Ac-LTF$r8EF4coohWAQCba$SAA-NH.sub.2 299 1771.92 887.54 1772.93 886.97 591.65 291 Ac-LTF$r8EF4coohWAQCba$SAA-NH.sub.2 300 iso2 1771.92 887.63 1772.93 886.97 591.65 292 Ac-LTF$r8EYWSQCba$SAA-NH.sub.2 301 1759.92 881.9 1760.93 880.97 587.65 293 Ac-LTF$r8EYWSQCba$SAA-NH.sub.2 302 iso2 1759.92 881.9 1760.93 880.97 587.65 294 Ac-LTF$r8EYWAQhL$SAA-NH.sub.2 303 1745.94 875.05 1746.95 873.98 582.99 295 Ac-LTF$r8AYWAQhL$SAF-NH.sub.2 304 1763.97 884.02 1764.98 882.99 589 296 Ac-LTF$r8AYWAQhL$SAF-NH.sub.2 305 iso2 1763.97 883.56 1764.98 882.99 589 297 Ac-LTF34F2$r8AYWAQhL$SAA-NH.sub.2 306 1723.92 863.67 1724.93 862.97 575.65 298 Ac-LTF34F2$r8AYWAQhL$SAA-NH.sub.2 307 iso2 1723.92 864.04 1724.93
862.97 575.65 299 Ac-LTF$r8AF4coohWAQhL$SAA-NH.sub.2 308 1715.93 859.44 1716.94 858.97 572.98 300 Ac-LTF$r8AF4coohWAQhL$SAA-NH.sub.2 309 iso2 1715.93 859.6 1716.94 858.97 572.98 301 Ac-LTF$r8AYWSQhL$SAA-NH.sub.2 310 1703.93 853.96 1704.94 852.97 568.98 302 Ac-LTF$r8AYWSQhL$SAA-NH.sub.2 311 iso2 1703.93 853.59 1704.94 852.97 568.98 303 Ac-LTF$r8EYWAQL$AANleA-NH.sub.2 312 1829.01 915.45 1830.02 915.51 610.68 304 Ac-LTF34F2$r8AYWAQL$AANleA-NH.sub.2 313 1806.99 904.58 1808 904.5 603.34 305 Ac-LTF$r8AF4coohWAQL$AANleA-NH.sub.2 314 1799 901.6 1800.01 900.51 600.67 306 Ac-LTF$r8AYWSQL$AANleA-NH.sub.2 315 1787 894.75 1788.01 894.51 596.67 307 Ac-LTF34F2$r8AYWAQhL$AANleA-NH.sub.2 316 1821 911.79 1822.01 911.51 608.01 308 Ac-LTF34F2$r8AYWAQhL$AANleA-NH.sub.2 317 iso2 1821 912.61 1822.01 911.51 608.01 309 Ac-LTF$r8AF4coohWAQhL$AANleA-NH.sub.2 318 1813.02 907.95 1814.03 907.52 605.35 310 Ac-LTF$r8AF4coohWAQhL$AANleA-NH.sub.2 319 iso2 1813.02 908.54 1814.03 907.52 605.35 311 Ac-LTF$r8AYWSQhL$AANleA-NH.sub.2 320 1801.02 901.84 1802.03 901.52 601.35 312 Ac-LTF$r8AYWSQhL$AANleA-NH.sub.2 321 iso2 1801.02 902.62 1802.03 901.52 601.35 313 Ac-LTF$r8AYWAQhL$AAAAa-NH.sub.2 322 1814.01 908.63 1815.02 908.01 605.68 314 Ac-LTF$r8AYWAQhL$AAAAa-NH.sub.2 323 iso2 1814.01 908.34 1815.02 908.01 605.68 315 Ac-LTF$r8AYWAQL$AAAAAa-NH.sub.2 324 1871.04 936.94 1872.05 936.53 624.69 316 Ac-LTF$r8AYWAQL$AAAAAAa-NH.sub.2 325 iso2 1942.07 972.5 1943.08 972.04 648.37 317 Ac-LTF$r8AYWAQL$AAAAAAa-NH.sub.2 326 isol 1942.07 972.5 1943.08 972.04 648.37 318 Ac-LTF$r8EYWAQhL$AANleA-NH.sub.2 327 1843.03 922.54 1844.04 922.52 615.35 319 Ac-AATF$r8AYWAQL$AANleA-NH.sub.2 328 1800 901.39 1801.01 901.01 601.01 320 Ac-LTF$r8AYWAQL$AANleAA-NH.sub.2 329 1842.04 922.45 1843.05 922.03 615.02 321 Ac-ALTF$r8AYWAQL$AANleAA-NH.sub.2 330 1913.08 957.94 1914.09 957.55 638.7 322 Ac-LTF$r8AYWAQCba$AANleAA-NH.sub.2 331 1854.04 928.43 1855.05 928.03 619.02 323 Ac-LTF$r8AYWAQhL$AANleAA-NH.sub.2 332 1856.06 929.4 1857.07 929.04 619.69 324 Ac-LTF$r8EYWAQCba$SAAA-NH.sub.2 333 1814.96 909.37 1815.97 908.49 605.99 325 Ac-LTF$r8EYWAQCba$SAAA-NH.sub.2 334 iso2 1814.96 909.37 1815.97 908.49 605.99 326 Ac-LTF$r8EYWAQCba$SAAAA-NH.sub.2 335 1886 944.61 1887.01 944.01 629.67 327 Ac-LTF$r8EYWAQCba$SAAAA-NH.sub.2 336 iso2 1886 944.61 1887.01 944.01 629.67 328 Ac-ALTF$r8EYWAQCba$SAA-NH.sub.2 337 1814.96 909.09 1815.97 908.49 605.99 329 Ac-ALTF$r8EYWAQCba$SAAA-NH.sub.2 338 1886 944.61 1887.01 944.01 629.67 330 Ac-ALTF$r8EYWAQCba$SAA-NH.sub.2 339 iso2 1814.96 909.09 1815.97 908.49 605.99 331 Ac-LTF$r8EYWAQL$AAAAAa-NH.sub.2 340 iso2 1929.04 966.08 1930.05 965.53 644.02 332 Ac-LTF$r8EY6clWAQCba$SAA-NH.sub.2 341 1777.89 890.78 1778.9 889.95 593.64 333 Ac-LTF$r8EF4cooh6clWAQCba$SANleA-NH.sub.2 342 1918.96 961.27 1919.97 960.49 640.66 334 Ac-LTF$r8EF4cooh6clWAQCba$SANleA-NH.sub.2 343 iso2 1918.96 961.27 1919.97 960.49 640.66 335 Ac-LTF$r8EF4cooh6clWAQCba$AAIa-NH.sub.2 344 1902.97 953.03 1903.98 952.49 635.33 336 Ac-LTF$r8EF4cooh6clWAQCba$AAIa-NH.sub.2 345 iso2 1902.97 953.13 1903.98 952.49 635.33 337 Ac-LTF$r8AY6clWAQL$AAAAAa-NH.sub.2 346 1905 954.61 1906.01 953.51 636.01 338 Ac-LTF$r8AY6clWAQL$AAAAAa-NH.sub.2 347 iso2 1905 954.9 1906.01 953.51 636.01 339 Ac-F$r8AY6clWEAL$AAAAAAa-NH.sub.2 348 1762.89 883.01 1763.9 882.45 588.64 340 Ac-ETF$r8EYWAQL$AAAAAa-NH.sub.2 349 1945 974.31 1946.01 973.51 649.34 341 Ac-ETF$r8EYWAQL$AAAAAa-NH.sub.2 350 iso2 1945 974.49 1946.01 973.51 649.34 342 Ac-LTF$r8EYWAQL$AAAAAAa-NH.sub.2 351 2000.08 1001.6 2001.09 1001.05 667.7 343 Ac-LTF$r8EYWAQL$AAAAAAa-NH.sub.2 352 iso2 2000.08 1001.6 2001.09 1001.05 667.7 344 Ac-LTF$r8AYWAQL$AANleAAa-NH.sub.2 353 1913.08 958.58 1914.09 957.55 638.7 345 Ac-LTF$r8AYWAQL$AANleAAa-NH.sub.2 354 iso2 1913.08 958.58 1914.09 957.55 638.7 346 Ac-LTF$r8EYWAQCba$AAAAAa-NH.sub.2 355 1941.04 972.55 1942.05 971.53 648.02 347 Ac-LTF$r8EYWAQCba$AAAAAa-NH.sub.2 356 iso2 1941.04 972.55 1942.05 971.53 648.02 348 Ac-LTF$r8EF4coohWAQCba$AAAAAa-NH.sub.2 357 1969.04 986.33 1970.05 985.53 657.35 349 Ac-LTF$r8EF4coohWAQCba$AAAAAa-NH.sub.2 358 iso2 1969.04 986.06 1970.05 985.53 657.35 350 Ac-LTF$r8EYWSQCba$AAAAAa-NH.sub.2 359 1957.04 980.04 1958.05 979.53 653.35 351 Ac-LTF$r8EYWSQCba$AAAAAa-NH.sub.2 360 iso2 1957.04 980.04 1958.05 979.53 653.35 352 Ac-LTF$r8EYWAQCba$SAAa-NH.sub.2 361 1814.96 909 1815.97 908.49 605.99 353 Ac-LTF$r8EYWAQCba$SAAa-NH.sub.2 362 iso2 1814.96 909 1815.97 908.49 605.99 354 Ac-ALTF$r8EYWAQCba$SAAa-NH.sub.2 363 1886 944.52 1887.01 944.01 629.67 355 Ac-ALTF$r8EYWAQCba$SAAa-NH.sub.2 364 iso2 1886 944.98 1887.01 944.01 629.67 356 Ac-ALTF$r8EYWAQCba$SAAAa-NH.sub.2 365 1957.04 980.04 1958.05 979.53 653.35 357 Ac-ALTF$r8EYWAQCba$SAAAa-NH.sub.2 366 iso2 1957.04 980.04 1958.05 979.53 653.35 358 Ac-AALTF$r8EYWAQCba$SAAAa-NH.sub.2 367 2028.07 1016.1 2029.08 1015.04 677.03 359 Ac-AALTF$r8EYWAQCba$SAAAa-NH.sub.2 368 iso2 2028.07 1015.57 2029.08 1015.04 677.03 360 Ac-RTF$r8EYWAQCba$SAA-NH.sub.2 369 1786.94 895.03 1787.95 894.48 596.65 361 Ac-LRF$r8EYWAQCba$SAA-NH.sub.2 370 1798.98 901.51 1799.99 900.5 600.67 362 Ac-LTF$r8EYWRQCba$SAA-NH.sub.2 371 1828.99 916.4 1830 915.5 610.67 363 Ac-LTF$r8EYWARCba$SAA-NH.sub.2 372 1771.97 887.63 1772.98 886.99 591.66 364 Ac-LTF$r8EYWAQCba$RAA-NH.sub.2 373 1812.99 908.08 1814 907.5 605.34 365 Ac-LTF$r8EYWAQCba$SRA-NH.sub.2 374 1828.99 916.12 1830 915.5 610.67 366 Ac-LTF$r8EYWAQCba$SAR-NH.sub.2 375 1828.99 916.12 1830 915.5 610.67 367 5-FAM-BaLTF$r8EYWAQCba$SAA-NH.sub.2 376 2131 1067.09 2132.01 1066.51 711.34 368 5-FAM-BaLTF$r8AYWAQL$AANleA-NH.sub.2 377 2158.08 1080.6 2159.09 1080.05 720.37 369 Ac-LAF$r8EYWAQL$AANleA-NH.sub.2 378 1799 901.05 1800.01 900.51 600.67 370 Ac-ATF$r8EYWAQL$AANleA-NH.sub.2 379 1786.97 895.03 1787.98 894.49 596.66 371 Ac-AAF$r8EYWAQL$AANleA-NH.sub.2 380 1756.96 880.05 1757.97 879.49 586.66 372 Ac-AAAF$r8EYWAQL$AANleA-NH.sub.2 381 1827.99 915.57 1829 915 610.34 373 Ac-AAAAF$r8EYWAQL$AANleA-NH.sub.2 382 1899.03 951.09 1900.04 950.52 634.02 374 Ac-AATF$r8EYWAQL$AANleA-NH.sub.2 383 1858 930.92 1859.01 930.01 620.34 375 Ac-AALTF$r8EYWAQL$AANleA-NH.sub.2 384 1971.09 987.17 1972.1 986.55 658.04 376 Ac-AAALTF$r8EYWAQL$AANleA-NH.sub.2 385 2042.12 1023.15 2043.13 1022.07 681.71 377 Ac-LTF$r8EYWAQL$AANleAA-NH.sub.2 386 1900.05 952.02 1901.06 951.03 634.36 378 Ac-ALTF$r8EYWAQL$AANleAA-NH.sub.2 387 1971.09 987.63 1972.1 986.55 658.04 379 Ac-AALTF$r8EYWAQL$AANleAA-NH.sub.2 388 2042.12 1022.69 2043.13 1022.07 681.71 380 Ac-LTF$r8EYWAQCba$AANleAA-NH.sub.2 389 1912.05 958.03 1913.06 957.03 638.36 381 Ac-LTF$r8EYWAQhL$AANleAA-NH.sub.2 390 1914.07 958.68 1915.08 958.04 639.03 382 Ac-ALTF$r8EYWAQhL$AANleAA-NH.sub.2 391 1985.1 994.1 1986.11 993.56 662.71 383 Ac-LTF$r8ANmYWAQL$AANleA-NH.sub.2 392 1785.02 894.11 1786.03 893.52 596.01 384 Ac-LTF$r8ANmYWAQL$AANleA-NH.sub.2 393 iso2 1785.02 894.11 1786.03 893.52 596.01 385 Ac-LTF$r8AYNmWAQL$AANleA-NH.sub.2 394 1785.02 894.11 1786.03 893.52 596.01 386 Ac-LTF$r8AYNmWAQL$AANleA-NH.sub.2 395 iso2 1785.02 894.11 1786.03 893.52 596.01 387 Ac-LTF$r8AYAmwAQL$AANleA-NH.sub.2 396 1785.02 894.01 1786.03 893.52 596.01 388 Ac-LTF$r8AYAmwAQL$AANleA-NH.sub.2 397 iso2 1785.02 894.01 1786.03 893.52 596.01 389 Ac-LTF$r8AYWAibQL$AANleA-NH.sub.2 398 1785.02 894.01 1786.03 893.52 596.01 390 Ac-LTF$r8AYWAibQL$AANleA-NH.sub.2 399 iso2 1785.02 894.01 1786.03 893.52 596.01 391 Ac-LTF$r8AYWAQL$AAibNleA-NH.sub.2 400 1785.02 894.38 1786.03 893.52 596.01 392 Ac-LTF$r8AYWAQL$AAibNleA-NH.sub.2 401 iso2 1785.02 894.38 1786.03 893.52 596.01 393 Ac-LTF$r8AYWAQL$AaNleA-NH.sub.2 402 1771.01 887.54 1772.02 886.51 591.34 394 Ac-LTF$r8AYWAQL$AaNleA-NH.sub.2 403 iso2 1771.01 887.54 1772.02 886.51 591.34 395 Ac-LTF$r8AYWAQL$ASarNleA-NH.sub.2 404 1771.01 887.35 1772.02 886.51 591.34 396 Ac-LTF$r8AYWAQL$ASarNleA-NH.sub.2 405 iso2 1771.01 887.35 1772.02 886.51 591.34 397 Ac-LTF$r8AYWAQL$AANleAib-NH.sub.2 406 1785.02 894.75 1786.03 893.52 596.01 398 Ac-LTF$r8AYWAQL$AANleAib-NH.sub.2 407 iso2 1785.02 894.75 1786.03 893.52 596.01 399 Ac-LTF$r8AYWAQL$AANleNmA-NH.sub.2 408 1785.02 894.6 1786.03 893.52 596.01 400 Ac-LTF$r8AYWAQL$AANleNmA-NH.sub.2 409 iso2 1785.02 894.6 1786.03 893.52 596.01 401 Ac-LTF$r8AYWAQL$AANleSar-NH.sub.2 410 1771.01 886.98 1772.02 886.51 591.34 402 Ac-LTF$r8AYWAQL$AANleSar-NH.sub.2 411 iso2 1771.01 886.98 1772.02 886.51 591.34 403 Ac-LTF$r8AYWAQL$AANleAAib-NH.sub.2 412 1856.06 1857.07 929.04 619.69 404 Ac-LTF$r8AYWAQL$AANleAAib-NH.sub.2 413 iso2 1856.06 1857.07 929.04 619.69 405 Ac-LTF$r8AYWAQL$AANleANmA-NH.sub.2 414 1856.06 930.37 1857.07 929.04 619.69 406 Ac-LTF$r8AYWAQL$AANleANmA-NH.sub.2 415 iso2 1856.06 930.37 1857.07 929.04 619.69 407 Ac-LTF$r8AYWAQL$AANleAa-NH.sub.2 416 1842.04 922.69 1843.05 922.03 615.02 408 Ac-LTF$r8AYWAQL$AANleAa-NH.sub.2 417 iso2 1842.04 922.69 1843.05 922.03 615.02 409 Ac-LTF$r8AYWAQL$AANleASar-NH.sub.2 418 1842.04 922.6 1843.05 922.03 615.02 410 Ac-LTF$r8AYWAQL$AANleASar-NH.sub.2 419 iso2 1842.04 922.6 1843.05 922.03 615.02 411 Ac-LTF$/r8AYWAQL$/AANleA-NH.sub.2 420 1799.04 901.14 1800.05 900.53 600.69 412 Ac-LTFAibAYWAQLAibAANleA-NH.sub.2 421 1648.9 826.02 1649.91 825.46 550.64 413 Ac-LTF$r8Cou4YWAQL$AANleA-NH.sub.2 422 1975.05 989.11 1976.06 988.53 659.36 414 Ac-LTF$r8Cou4YWAQL$AANleA-NH.sub.2 423 iso2 1975.05 989.11 1976.06 988.53 659.36 415 Ac-LTF$r8AYWCou4QL$AANleA-NH.sub.2 424 1975.05 989.11 1976.06 988.53 659.36 416 Ac-LTF$r8AYWAQL$Cou4ANleA-NH.sub.2 425 1975.05 989.57 1976.06 988.53 659.36 417 Ac-LTF$r8AYWAQL$Cou4ANleA-NH.sub.2 426 iso2 1975.05 989.57 1976.06 988.53 659.36 418 Ac-LTF$r8AYWAQL$ACou4NleA-NH.sub.2 427 1975.05 989.57 1976.06 988.53 659.36 419 Ac-LTF$r8AYWAQL$ACou4NleA-NH.sub.2 428 iso2 1975.05 989.57 1976.06 988.53 659.36 420 Ac-LTF$r8AYWAQL$AANleA-OH 429 1771.99 887.63 1773 887 591.67 421 Ac-LTF$r8AYWAQL$AANleA-OH 430 iso2 1771.99 887.63 1773 887 591.67 422 Ac-LTF$r8AYWAQL$AANleA-NHnPr 431 1813.05 908.08 1814.06 907.53 605.36 423 Ac-LTF$r8AYWAQL$AANleA-NHnPr 432 iso2 1813.05 908.08 1814.06 907.53 605.36 424 Ac-LTF$r8AYWAQL$AANleA-NHnBu33Me 433 1855.1 929.17 1856.11 928.56 619.37 425 Ac-LTF$r8AYWAQL$AANleA-NHnBu33Me 434 iso2 1855.1 929.17 1856.11 928.56 619.37 426 Ac-LTF$r8AYWAQL$AANleA-NHHex 435 1855.1 929.17 1856.11 928.56 619.37 427 Ac-LTF$r8AYWAQL$AANleA-NHHex 436 iso2 1855.1 929.17 1856.11 928.56 619.37
428 Ac-LTA$r8AYWAQL$AANleA-NH.sub.2 437 1694.98 849.33 1695.99 848.5 566 429 Ac-LThL$r8AYWAQL$AANleA-NH.sub.2 438 1751.04 877.09 1752.05 876.53 584.69 430 Ac-LTF$r8AYAAQL$AANleA-NH.sub.2 439 1655.97 829.54 1656.98 828.99 553 431 Ac-LTF$r8AY2NalAQL$AANleA-NH.sub.2 440 1782.01 892.63 1783.02 892.01 595.01 432 Ac-LTF$r8EYWCou4QCba$SAA-NH.sub.2 441 1947.97 975.8 1948.98 974.99 650.33 433 Ac-LTF$r8EYWCou7QCba$SAA-NH.sub.2 442 16.03 974.9 17.04 9.02 6.35 434 Ac-LTF%r8EYWAQCba%SAA-NH.sub.2 443 1745.94 874.8 1746.95 873.98 582.99 435 Dmaac-LTF$r8EYWAQCba$SAA-NH.sub.2 444 1786.97 894.8 1787.98 894.49 596.66 436 Dmaac-LTF$r8AYWAQL$AAAAAa-NH.sub.2 445 1914.08 958.2 1915.09 958.05 639.03 437 Dmaac-LTF$r8AYWAQL$AAAAAa-NH.sub.2 446 iso2 1914.08 958.2 1915.09 958.05 639.03 438 Dmaac-LTF$r8EYWAQL$AAAAAa-NH.sub.2 447 1972.08 987.3 1973.09 987.05 658.37 439 Dmaac-LTF$r8EYWAQL$AAAAAa-NH.sub.2 448 iso2 1972.08 987.3 1973.09 987.05 658.37 440 Dmaac-LTF$r8EF4coohWAQCba$AAIa-NH.sub.2 449 1912.05 957.4 1913.06 957.03 638.36 441 Dmaac-LTF$r8EF4coohWAQCba$AAIa-NH.sub.2 450 iso2 1912.05 957.4 1913.06 957.03 638.36 442 Dmaac-LTF$r8AYWAQL$AANleA-NH.sub.2 451 1814.05 908.3 1815.06 908.03 605.69 443 Dmaac-LTF$r8AYWAQL$AANleA-NH.sub.2 452 iso2 1814.05 908.3 1815.06 908.03 605.69 444 Ac-LTF%r8AYWAQL%AANleA-NH.sub.2 453 1773.02 888.37 1774.03 887.52 592.01 445 Ac-LTF%r8EYWAQL%AAAAAa-NH.sub.2 454 1931.06 966.4 1932.07 966.54 644.69 446 Cou6BaLTF$r8EYWAQhL$SAA-NH.sub.2 455 2018.05 1009.9 2019.06 1010.03 673.69 447 Cou8BaLTF$r8EYWAQhL$SAA-NH.sub.2 456 1962.96 982.34 1963.97 982.49 655.32 448 Ac-LTF4I$r8EYWAQL$AAAAAa-NH.sub.2 457 2054.93 1028.68 2055.94 1028.47 685.98 449 Ac-LTF$r8EYWAQL$AAAAAa-NH.sub.2 458 1929.04 966.17 1930.05 965.53 644.02 550 Ac-LTF$r8EYWAQL$AAAAAa-OH 459 1930.02 966.54 1931.03 966.02 644.35 551 Ac-LTF$r8EYWAQL$AAAAAa-OH 460 iso2 1930.02 965.89 1931.03 966.02 644.35 552 Ac-LTF$r8EYWAEL$AAAAAa-NH.sub.2 461 1930.02 966.82 1931.03 966.02 644.35 553 Ac-LTF$r8EYWAEL$AAAAAa-NH.sub.2 462 iso2 1930.02 966.91 1931.03 966.02 644.35 554 Ac-LTF$r8EYWAEL$AAAAAa-OH 463 1931.01 967.28 1932.02 966.51 644.68 555 Ac-LTF$r8EY6clWAQL$AAAAAa-NH.sub.2 464 1963 983.28 1964.01 982.51 655.34 556 Ac-LTF$r8EF4bOH2WAQL$AAAAAa-NH.sub.2 465 1957.05 980.04 1958.06 979.53 653.36 557 Ac-AAALTF$r8EYWAQL$AAAAAa-NH.sub.2 466 2142.15 1072.83 2143.16 1072.08 715.06 558 Ac-LTF34F2$r8EYWAQL$AAAAAa-NH.sub.2 467 1965.02 984.3 1966.03 983.52 656.01 559 Ac-RTF$r8EYWAQL$AAAAAa-NH.sub.2 468 1972.06 987.81 1973.07 987.04 658.36 560 Ac-LTA$r8EYWAQL$AAAAAa-NH.sub.2 469 1853.01 928.33 1854.02 927.51 618.68 561 Ac-LTF$r8EYWAibQL$AAAAAa-NH.sub.2 470 1943.06 973.48 1944.07 972.54 648.69 562 Ac-LTF$r8EYWAQL$AAibAAAa-NH.sub.2 471 1943.06 973.11 1944.07 972.54 648.69 563 Ac-LTF$r8EYWAQL$AAAibAAa-NH.sub.2 472 1943.06 973.48 1944.07 972.54 648.69 564 Ac-LTF$r8EYWAQL$AAAAibAa-NH.sub.2 473 1943.06 973.48 1944.07 972.54 648.69 565 Ac-LTF$r8EYWAQL$AAAAAiba-NH.sub.2 474 1943.06 973.38 1944.07 972.54 648.69 566 Ac-LTF$r8EYWAQL$AAAAAiba-NH.sub.2 475 iso2 1943.06 973.38 1944.07 972.54 648.69 567 Ac-LTF$r8EYWAQL$AAAAAAib-NH.sub.2 476 1943.06 973.01 1944.07 972.54 648.69 568 Ac-LTF$r8EYWAQL$AaAAAa-NH.sub.2 477 1929.04 966.54 1930.05 965.53 644.02 569 Ac-LTF$r8EYWAQL$AAaAAa-NH.sub.2 478 1929.04 966.35 1930.05 965.53 644.02 570 Ac-LTF$r8EYWAQL$AAAaAa-NH.sub.2 479 1929.04 966.54 1930.05 965.53 644.02 571 Ac-LTF$r8EYWAQL$AAAaAa-NH.sub.2 480 iso2 1929.04 966.35 1930.05 965.53 644.02 572 Ac-LTF$r8EYWAQL$AAAAaa-NH.sub.2 481 1929.04 966.35 1930.05 965.53 644.02 573 Ac-LTF$r8EYWAQL$AAAAAA-NH.sub.2 482 1929.04 966.35 1930.05 965.53 644.02 574 Ac-LTF$r8EYWAQL$ASarAAAa-NH.sub.2 483 1929.04 966.54 1930.05 965.53 644.02 575 Ac-LTF$r8EYWAQL$AASarAAa-NH.sub.2 484 1929.04 966.35 1930.05 965.53 644.02 576 Ac-LTF$r8EYWAQL$AAASarAa-NH.sub.2 485 1929.04 966.35 1930.05 965.53 644.02 577 Ac-LTF$r8EYWAQL$AAAASara-NH.sub.2 486 1929.04 966.35 1930.05 965.53 644.02 578 Ac-LTF$r8EYWAQL$AAAAASar-NH.sub.2 487 1929.04 966.08 1930.05 965.53 644.02 579 Ac-7LTF$r8EYWAQL$AAAAAa-NH.sub.2 488 1918.07 951.99 1919.08 960.04 640.37 581 Ac-TF$r8EYWAQL$AAAAAa-NH.sub.2 489 1815.96 929.85 1816.97 908.99 606.33 582 Ac-F$r8EYWAQL$AAAAAa-NH.sub.2 490 1714.91 930.92 1715.92 858.46 572.64 583 Ac-LVF$r8EYWAQL$AAAAAa-NH.sub.2 491 1927.06 895.12 1928.07 964.54 643.36 584 Ac-AAF$r8EYWAQL$AAAAAa-NH.sub.2 492 1856.98 859.51 1857.99 929.5 620 585 Ac-LTF$r8EYWAQL$AAAAa-NH.sub.2 493 1858 824.08 1859.01 930.01 620.34 586 Ac-LTF$r8EYWAQL$AAAa-NH.sub.2 494 1786.97 788.56 1787.98 894.49 596.66 587 Ac-LTF$r8EYWAQL$AAa-NH.sub.2 495 1715.93 1138.57 1716.94 858.97 572.98 588 Ac-LTF$r8EYWAQL$Aa-NH.sub.2 496 1644.89 1144.98 1645.9 823.45 549.3 589 Ac-LTF$r8EYWAQL$a-NH.sub.2 497 1573.85 1113.71 1574.86 787.93 525.62 590 Ac-LTF$r8EYWAQL$AAA-OH 498 1716.91 859.55 1717.92 859.46 573.31 591 Ac-LTF$r8EYWAQL$A-OH 499 1574.84 975.14 1575.85 788.43 525.95 592 Ac-LTF$r8EYWAQL$AAA-NH.sub.2 500 1715.93 904.75 1716.94 858.97 572.98 593 Ac-LTF$r8EYWAQCba$SAA-OH 501 1744.91 802.49 1745.92 873.46 582.64 594 Ac-LTF$r8EYWAQCba$S-OH 502 1602.83 913.53 1603.84 802.42 535.28 595 Ac-LTF$r8EYWAQCba$S-NH.sub.2 503 1601.85 979.58 1602.86 801.93 534.96 596 4-FBzl-LTF$r8EYWAQL$AAAAAa-NH.sub.2 504 2009.05 970.52 2010.06 1005.53 670.69 597 4-FBzl-LTF$r8EYWAQCba$SAA-NH.sub.2 505 1823.93 965.8 1824.94 912.97 608.98 598 Ac-LTF$r8RYWAQL$AAAAAa-NH.sub.2 506 1956.1 988.28 1957.11 979.06 653.04 599 Ac-LTF$r8HYWAQL$AAAAAa-NH.sub.2 507 1937.06 1003.54 1938.07 969.54 646.69 600 Ac-LTF$r8QYWAQL$AAAAAa-NH.sub.2 508 1928.06 993.92 1929.07 965.04 643.69 601 Ac-LTF$r8CitYWAQL$AAAAAa-NH.sub.2 509 1957.08 987 1958.09 979.55 653.37 602 Ac-LTF$r8GlaYWAQL$AAAAAa-NH.sub.2 510 1973.03 983 1974.04 987.52 658.68 603 Ac-LTF$r8F4gYWAQL$AAAAAa-NH.sub.2 511 2004.1 937.86 2005.11 1003.06 669.04 604 Ac-LTF$r82mRYWAQL$AAAAAa-NH.sub.2 512 1984.13 958.58 1985.14 993.07 662.38 605 Ac-LTF$r8ipKYWAQL$AAAAAa-NH.sub.2 513 1970.14 944.52 1971.15 986.08 657.72 606 Ac-LTF$r8F4NH.sub.2YWAQL$AAAAAa-NH.sub.2 514 1962.08 946 1963.09 982.05 655.03 607 Ac-LTF$r8EYWAAL$AAAAAa-NH.sub.2 515 1872.02 959.32 1873.03 937.02 625.01 608 Ac-LTF$r8EYWALL$AAAAAa-NH.sub.2 516 1914.07 980.88 1915.08 958.04 639.03 609 Ac-LTF$r8EYWAAibL$AAAAAa-NH.sub.2 517 1886.03 970.61 1887.04 944.02 629.68 610 Ac-LTF$r8EYWASL$AAAAAa-NH.sub.2 518 1888.01 980.51 1889.02 945.01 630.34 611 Ac-LTF$r8EYWANL$AAAAAa-NH.sub.2 519 1915.02 1006.41 1916.03 958.52 639.35 612 Ac-LTF$r8EYWACitL$AAAAAa-NH.sub.2 520 1958.07 1959.08 980.04 653.7 613 Ac-LTF$r8EYWAHL$AAAAAa-NH.sub.2 521 1938.04 966.24 1939.05 970.03 647.02 614 Ac-LTF$r8EYWARL$AAAAAa-NH.sub.2 522 1957.08 1958.09 979.55 653.37 615 Ac-LTF$r8EpYWAQL$AAAAAa-NH.sub.2 523 2009.01 2010.02 1005.51 670.68 616 Cbm-LTF$r8EYWAQCba$SAA-NH.sub.2 524 1590.85 1591.86 796.43 531.29 617 Cbm-LTF$r8EYWAQL$AAAAAa-NH.sub.2 525 1930.04 1931.05 966.03 644.35 618 Ac-LTF$r8EYWAQL$SAAAAa-NH.sub.2 526 1945.04 1005.11 1946.05 973.53 649.35 619 Ac-LTF$r8EYWAQL$AAAASa-NH.sub.2 527 1945.04 986.52 1946.05 973.53 649.35 620 Ac-LTF$r8EYWAQL$SAAASa-NH.sub.2 528 1961.03 993.27 1962.04 981.52 654.68 621 Ac-LTF$r8EYWAQTba$AAAAAa-NH.sub.2 529 1943.06 983.1 1944.07 972.54 648.69 622 Ac-LTF$r8EYWAQAdm$AAAAAa-NH.sub.2 530 2007.09 990.31 2008.1 1004.55 670.04 623 Ac-LTF$r8EYWAQCha$AAAAAa-NH.sub.2 531 1969.07 987.17 1970.08 985.54 657.36 624 Ac-LTF$r8EYWAQhCha$AAAAAa-NH.sub.2 532 1983.09 1026.11 1984.1 992.55 662.04 625 Ac-LTF$r8EYWAQF$AAAAAa-NH.sub.2 533 1963.02 957.01 1964.03 982.52 655.35 626 Ac-LTF$r8EYWAQhF$AAAAAa-NH.sub.2 534 1977.04 1087.81 1978.05 989.53 660.02 627 Ac-LTF$r8EYWAQL$AANleAAa-NH.sub.2 535 1971.09 933.45 1972.1 986.55 658.04 628 Ac-LTF$r8EYWAQAdm$AANleAAa-NH.sub.2 536 2049.13 1017.97 2050.14 1025.57 684.05 629 4-FBz-BaLTF$r8EYWAQL$AAAAAa-NH.sub.2 537 2080.08 2081.09 1041.05 694.37 630 4-FBz-BaLTF$r8EYWAQCba$SAA-NH.sub.2 538 1894.97 1895.98 948.49 632.66 631 Ac-LTF$r5EYWAQL$s8AAAAAa-NH.sub.2 539 1929.04 1072.68 1930.05 965.53 644.02 632 Ac-LTF$r5EYWAQCba$s8SAA-NH.sub.2 540 1743.92 1107.79 1744.93 872.97 582.31 633 Ac-LTF$r8EYWAQL$AAhhLAAa-NH.sub.2 541 1999.12 2000.13 1000.57 667.38 634 Ac-LTF$r8EYWAQL$AAAAAAAa-NH.sub.2 542 2071.11 2072.12 1036.56 691.38 635 Ac-LTF$r8EYWAQL$AAAAAAAAa-NH.sub.2 543 2142.15 778.1 2143.16 1072.08 715.06 636 Ac-LTF$r8EYWAQL$AAAAAAAAAa-NH.sub.2 544 2213.19 870.53 2214.2 1107.6 738.74 637 Ac-LTA$r8EYAAQCba$SAA-NH.sub.2 545 1552.85 1553.86 777.43 518.62 638 Ac-LTA$r8EYAAQL$AAAAAa-NH.sub.2 546 1737.97 779.45 1738.98 869.99 580.33 639 Ac-LTF$r8EPmpWAQL$AAAAAa-NH.sub.2 547 2007.03 779.54 2008.04 1004.52 670.02 640 Ac-LTF$r8EPmpWAQCba$SAA-NH.sub.2 548 1821.91 838.04 1822.92 911.96 608.31 641 Ac-ATF$r8HYWAQL$S-NH.sub.2 549 1555.82 867.83 1556.83 778.92 519.61 642 Ac-LTF$r8HAWAQL$S-NH.sub.2 550 1505.84 877.91 1506.85 753.93 502.95 643 Ac-LTF$r8HYWAQA$S-NH.sub.2 551 1555.82 852.52 1556.83 778.92 519.61 644 Ac-LTF$r8EYWAQCba$SA-NH.sub.2 552 1672.89 887.18 1673.9 837.45 558.64 645 Ac-LTF$r8EYWAQL$SAA-NH.sub.2 553 1731.92 873.32 1732.93 866.97 578.31 646 Ac-LTF$r8HYWAQCba$SAA-NH.sub.2 554 1751.94 873.05 1752.95 876.98 584.99 647 Ac-LTF$r8SYWAQCba$SAA-NH.sub.2 555 1701.91 844.88 1702.92 851.96 568.31 648 Ac-LTF$r8RYWAQCba$SAA-NH.sub.2 556 1770.98 865.58 1771.99 886.5 591.33 649 Ac-LTF$r8KYWAQCba$SAA-NH.sub.2 557 1742.98 936.57 1743.99 872.5 582 650 Ac-LTF$r8QYWAQCba$SAA-NH.sub.2 558 1742.94 930.93 1743.95 872.48 581.99 651 Ac-LTF$r8EYWAACba$SAA-NH.sub.2 559 1686.9 1032.45 1687.91 844.46 563.31 652 Ac-LTF$r8EYWAQCba$AAA-NH.sub.2 560 1727.93 895.46 1728.94 864.97 576.98 653 Ac-LTF$r8EYWAQL$AAAAA-OH 561 1858.99 824.54 1860 930.5 620.67 654 Ac-LTF$r8EYWAQL$AAAA-OH 562 1787.95 894.48 1788.96 894.98 596.99 655 Ac-LTF$r8EYWAQL$AA-OH 563 1645.88 856 1646.89 823.95 549.63 656 Ac-LTF$r8AF4bOH2WAQL$AAAAAa-NH.sub.2 564 657 Ac-LTF$r8AF4bOH2WAAL$AAAAAa-NH.sub.2 565 658 Ac-LTF$r8EF4bOH2WAQCba$SAA-NH.sub.2 566 659 Ac-LTF$r8ApYWAQL$AAAAAa-NH.sub.2 567 660 Ac-LTF$r8ApYWAAL$AAAAAa-NH.sub.2 568 661 Ac-LTF$r8EpYWAQCba$SAA-NH.sub.2 569 662 Ac-LTF$rda6AYWAQL$da5AAAAAa-NH.sub.2 570 1974.06 934.44 663 Ac-LTF$rda6EYWAQCba$da5SAA-NH.sub.2 571 1846.95 870.52 869.94 664 Ac-LTF$rda6EYWAQL$da5AAAAAa-NH.sub.2 572 665 Ac-LTF$ra9EYWAQL$a6AAAAAa-NH.sub.2 573 936.57 935.51 666 Ac-LTF$ra9EYWAQL$a6AAAAAa-NH.sub.2 574 667 Ac-LTF$ra9EYWAQCba$a6SAA-NH.sub.2 575 668 Ac-LTA$ra9EYWAQCba$a6SAA-NH.sub.2 576 669 5-FAM-BaLTF$ra9EYWAQCba$a6SAA-NH.sub.2 577 670 5-FAM-BaLTF$r8EYWAQL$AAAAAa-NH.sub.2 578 2316.11 671 5-FAM-BaLTF$/r8EYWAQL$/AAAAAa-NH.sub.2 579 2344.15 672 5-FAM-BaLTA$r8EYWAQL$AAAAAa-NH.sub.2 580 2240.08 673 5-FAM-BaLTF$r8AYWAQL$AAAAAa-NH.sub.2 581 2258.11 674 5-FAM-BaATF$r8EYWAQL$AAAAAa-NH.sub.2 582 2274.07 675 5-FAM-BaLAF$r8EYWAQL$AAAAAa-NH.sub.2 583 2286.1 676 5-FAM-BaLTF$r8EAWAQL$AAAAAa-NH.sub.2 584 2224.09 677 5-FAM-BaLTF$r8EYAAQL$AAAAAa-NH.sub.2 585 2201.07
678 5-FAM-BaLTA$r8EYAAQL$AAAAAa-NH.sub.2 586 2125.04 679 5-FAM-BaLTF$r8EYWAAL$AAAAAa-NH.sub.2 587 2259.09 680 5-FAM-BaLTF$r8EYWAQA$AAAAAa-NH.sub.2 588 2274.07 681 5-FAM-BaLTF$/r8EYWAQCba$/SAA-NH.sub.2 589 2159.03 682 5-FAM-BaLTA$r8EYWAQCba$SAA-NH.sub.2 590 2054.97 683 5-FAM-BaLTF$r8EYAAQCba$SAA-NH.sub.2 591 2015.96 684 5-FAM-BaLTA$r8EYAAQCba$SAA-NH.sub.2 592 1939.92 685 5-FAM-BaQSQQTF$r8NLWRLL$QN-NH.sub.2 593 2495.23 686 5-TAMRA-BaLTF$r8EYWAQCba$SAA-NH.sub.2 594 2186.1 687 5-TAMRA-BaLTA$r8EYWAQCba$SAA-NH.sub.2 595 2110.07 688 5-TAMRA-BaLTF$r8EYAAQCba$SAA-NH.sub.2 596 2071.06 689 5-TAMRA-BaLTA$r8EYAAQCba$SAA-NH.sub.2 597 1995.03 690 5-TAMRA-BaLTF$/r8EYWAQCba$/SAA-NH.sub.2 598 2214.13 691 5-TAMRA-BaLTF$r8EYWAQL$AAAAAa-NH.sub.2 599 2371.22 692 5-TAMRA-BaLTA$r8EYWAQL$AAAAAa-NH.sub.2 600 2295.19 693 5-TAMRA-BaLTF$/r8EYWAQL$/AAAAAa-NH.sub.2 601 2399.25 694 Ac-LTF$r8EYWCou7QCba$SAA-OH 602 1947.93 695 Ac-LTF$r8EYWCou7QCba$S-OH 603 1805.86 696 Ac-LTA$r8EYWCou7QCba$SAA-NH.sub.2 604 1870.91 697 Ac-LTF$r8EYACou7QCba$SAA-NH.sub.2 605 1831.9 698 Ac-LTA$r8EYACou7QCba$SAA-NH.sub.2 606 1755.87 699 Ac-LTF$/r8EYWCou7QCba$/SAA-NH.sub.2 607 1974.98 700 Ac-LTF$r8EYWCou7QL$AAAAAa-NH.sub.2 608 2132.06 701 Ac-LTF$/r8EYWCou7QL$/AAAAAa-NH.sub.2 609 2160.09 702 Ac-LTF$r8EYWCou7QL$AAAAA-OH 610 2062.01 703 Ac-LTF$r8EYWCou7QL$AAAA-OH 611 1990.97 704 Ac-LTF$r8EYWCou7QL$AAA-OH 612 1919.94 705 Ac-LTF$r8EYWCou7QL$AA-OH 613 1848.9 706 Ac-LTF$r8EYWCou7QL$A-OH 614 1777.86 707 Ac-LTF$r8EYWAQL$AAAASa-NH.sub.2 615 iso2 974.4 973.53 708 Ac-LTF$r8AYWAAL$AAAAAa-NH.sub.2 616 iso2 1814.01 908.82 1815.02 908.01 605.68 709 Biotin-BaLTF$r8EYWAQL$AAAAAa-NH.sub.2 617 2184.14 1093.64 2185.15 1093.08 729.05 710 Ac-LTF$r8HAWAQL$S-NH.sub.2 618 iso2 1505.84 754.43 1506.85 753.93 502.95 711 Ac-LTF$r8EYWAQCba$SA-NH.sub.2 619 iso2 1672.89 838.05 1673.9 837.45 558.64 712 Ac-LTF$r8HYWAQCba$SAA-NH.sub.2 620 iso2 1751.94 877.55 1752.95 876.98 584.99 713 Ac-LTF$r8SYWAQCba$SAA-NH.sub.2 621 iso2 1701.91 852.48 1702.92 851.96 568.31 714 Ac-LTF$r8RYWAQCba$SAA-NH.sub.2 622 iso2 1770.98 887.45 1771.99 886.5 591.33 715 Ac-LTF$r8KYWAQCba$SAA-NH.sub.2 623 iso2 1742.98 872.92 1743.99 872.5 582 716 Ac-LTF$r8EYWAQCba$AAA-NH.sub.2 624 iso2 1727.93 865.71 1728.94 864.97 576.98 717 Ac-LTF$r8EYWAQL$AAAAAaBaC-NH.sub.2 625 2103.09 1053.12 2104.1 1052.55 702.04 718 Ac-LTF$r8EYWAQL$AAAAAadPeg4C-NH.sub.2 626 2279.19 1141.46 2280.2 1140.6 760.74 719 Ac-LTA$r8AYWAAL$AAAAAa-NH.sub.2 627 1737.98 870.43 1738.99 870 580.33 720 Ac-LTF$r8AYAAAL$AAAAAa-NH.sub.2 628 1698.97 851 1699.98 850.49 567.33 721 5-FAM-BaLTF$r8AYWAAL$AAAAAa-NH.sub.2 629 2201.09 1101.87 2202.1 1101.55 734.7 722 Ac-LTA$r8AYWAQL$AAAAAa-NH.sub.2 630 1795 898.92 1796.01 898.51 599.34 723 Ac-LTF$r8AYAAQL$AAAAAa-NH.sub.2 631 1755.99 879.49 1757 879 586.34 724 Ac-LTF$rda6AYWAAL$da5AAAAAa-NH.sub.2 632 1807.97 1808.98 904.99 603.66 725 FITC-BaLTF$r8EYWAQL$AAAAAa-NH.sub.2 633 2347.1 1174.49 2348.11 1174.56 783.37 726 FITC-BaLTF$r8EYWAQCba$SAA-NH.sub.2 634 2161.99 1082.35 2163 1082 721.67 733 Ac-LTF$r8EYWAQL$EAAAAa-NH.sub.2 635 1987.05 995.03 1988.06 994.53 663.36 734 Ac-LTF$r8AYWAQL$EAAAAa-NH.sub.2 636 1929.04 966.35 1930.05 965.53 644.02 735 Ac-LTF$r8EYWAQL$AAAAAaBaKbio-NH.sub.2 637 2354.25 1178.47 2355.26 1178.13 785.76 736 Ac-LTF$r8AYWAAL$AAAAAa-NH.sub.2 638 1814.01 908.45 1815.02 908.01 605.68 737 Ac-LTF$r8AYAAAL$AAAAAa-NH.sub.2 639 iso2 1698.97 850.91 1699.98 850.49 567.33 738 Ac-LTF$r8AYAAQL$AAAAAa-NH.sub.2 640 iso2 1755.99 879.4 1757 879 586.34 739 Ac-LTF$r8EYWAQL$EAAAAa-NH.sub.2 641 iso2 1987.05 995.21 1988.06 994.53 663.36 740 Ac-LTF$r8AYWAQL$EAAAAa-NH.sub.2 642 iso2 1929.04 966.08 1930.05 965.53 644.02 741 Ac-LTF$r8EYWAQCba$SAAAAa-NH.sub.2 643 1957.04 980.04 1958.05 979.53 653.35 742 Ac-LTF$r8EYWAQLStAAA$r5AA-NH.sub.2 644 2023.12 1012.83 2024.13 1012.57 675.38 743 Ac-LTF$r8EYWAQL$A$AAA$A-NH.sub.2 645 2108.17 1055.44 2109.18 1055.09 703.73 744 Ac-LTF$r8EYWAQL$AA$AAA$A-NH.sub.2 646 2179.21 1090.77 2180.22 1090.61 727.41 745 Ac-LTF$r8EYWAQL$AAA$AAA$A-NH.sub.2 647 2250.25 1126.69 2251.26 1126.13 751.09 746 Ac-AAALTF$r8EYWAQL$AAA-OH 648 1930.02 1931.03 966.02 644.35 747 Ac-AAALTF$r8EYWAQL$AAA-NH.sub.2 649 1929.04 965.85 1930.05 965.53 644.02 748 Ac-AAAALTF$r8EYWAQL$AAA-NH.sub.2 650 2000.08 1001.4 2001.09 1001.05 667.7 749 Ac-AAAAALTF$r8EYWAQL$AAA-NH.sub.2 651 2071.11 1037.13 2072.12 1036.56 691.38 750 Ac-AAAAAALTF$r8EYWAQL$AAA-NH.sub.2 652 2142.15 2143.16 1072.08 715.06 751 Ac-LTF$rda6EYWAQCba$da6SAA-NH.sub.2 653 iso2 1751.89 877.36 1752.9 876.95 584.97 752 Ac-t$r5wya$r5f4CF3ekllr-NH.sub.2 654 844.25 753 Ac-tawy$r5nf4CF3e$r5llr-NH.sub.2 655 837.03 754 Ac-tawya$r5f4CF3ek$r5lr-NH.sub.2 656 822.97 755 Ac-tawyanf4CF3e$r5llr$r5a-NH.sub.2 657 908.35 756 Ac-t$s8wyanf4CF3e$r5llr-NH.sub.2 658 858.03 757 Ac-tawy$s8nf4CF3ekll$r5a-NH.sub.2 659 879.86 758 Ac-tawya$s8f4CF3ekllr$r5a-NH.sub.2 660 936.38 759 Ac-tawy$s8naekll$r5a-NH.sub.2 661 844.25 760 5-FAM-Batawy$s8nf4CF3ekll$r5a-NH.sub.2 662 761 5-FAM-Batawy$s8naekll$r5a-NH.sub.2 663 762 Ac-tawy$s8nf4CF3eall$r5a-NH.sub.2 664 763 Ac-tawy$s8nf4CF3ekll$r5aaaaa-NH.sub.2 665 764 Ac-tawy$s8nf4CF3eall$r5aaaaa-NH.sub.2 666
[0357] Table 3a shows a selection of peptidomimetic macrocycles.
TABLE-US-00006 TABLE 3a SEQ ID Exact Found Calc Calc Calc Sequence NO: Iso Mass Mass (M + 1)/1 (M + 2)/2 (M + 3)/3 244 Ac-LTF$r8EF4coohWAQCba$SANleA-NH.sub.2 667 1885 943.59 1886.01 943.51 629.34 331 Ac-LTF$r8EYWAQL$AAAAAa-NH.sub.2 668 iso2 1929.04 966.08 1930.05 965.53 644.02 555 Ac-LTF$r8EY6clWAQL$AAAAAa-NH.sub.2 669 1963 983.28 1964.01 982.51 655.34 557 Ac-AAALTF$r8EYWAQL$AAAAAa-NH.sub.2 670 2142.15 1072.83 2143.16 1072.08 715.06 558 Ac-LTF34F2$r8EYWAQL$AAAAAa-NH.sub.2 671 1965.02 984.3 1966.03 983.52 656.01 562 Ac-LTF$r8EYWAQL$AAibAAAa-NH.sub.2 672 1943.06 973.11 1944.07 972.54 648.69 564 Ac-LTF$r8EYWAQL$AAAAibAa-NH.sub.2 673 1943.06 973.48 1944.07 972.54 648.69 566 Ac-LTF$r8EYWAQL$AAAAAiba-NH.sub.2 674 iso2 1943.06 973.38 1944.07 972.54 648.69 567 Ac-LTF$r8EYWAQL$AAAAAAib-NH.sub.2 675 1943.06 973.01 1944.07 972.54 648.69 572 Ac-LTF$r8EYWAQL$AAAAaa-NH.sub.2 676 1929.04 966.35 1930.05 965.53 644.02 573 Ac-LTF$r8EYWAQL$AAAAAA-NH.sub.2 677 1929.04 966.35 1930.05 965.53 644.02 578 Ac-LTF$r8EYWAQL$AAAAASar-NH.sub.2 678 1929.04 966.08 1930.05 965.53 644.02 551 Ac-LTF$r8EYWAQL$AAAAAa-OH 679 iso2 1930.02 965.89 1931.03 966.02 644.35 662 Ac-LTF$rda6AYWAQL$da5AAAAAa-NH.sub.2 680 1974.06 934.44 933.49 367 5-FAM-BaLTF$r8EYWAQCba$SAA-NH.sub.2 681 2131 1067.09 2132.01 1066.51 711.34 349 Ac-LTF$r8EF4coohWAQCba$AAAAAa-NH.sub.2 682 iso2 1969.04 986.06 1970.05 985.53 657.35 347 Ac-LTF$r8EYWAQCba$AAAAAa-NH.sub.2 683 iso2 1941.04 972.55 1942.05 971.53 648.02
[0358] Table 3b shows a further selection of peptidomimetic macrocycles.
TABLE-US-00007 TABLE 3b SEQ ID Exact Found Calc Calc Calc SP Sequence NO: Iso Mass Mass (M + 1)/1 (M + 2)/2 (M + 3)/3 581 Ac-TF$r8EYWAQL$AAAAAa-NH.sub.2 684 1815.96 929.85 1816.97 908.99 606.33 582 Ac-F$r8EYWAQL$AAAAAa-NH.sub.2 685 1714.91 930.92 1715.92 858.46 572.64 583 Ac-LVF$r8EYWAQL$AAAAAa-NH.sub.2 686 1927.06 895.12 1928.07 964.54 643.36 584 Ac-AAF$r8EYWAQL$AAAAAa-NH.sub.2 687 1856.98 859.51 1857.99 929.5 620 585 Ac-LTF$r8EYWAQL$AAAAa-NH.sub.2 688 1858 824.08 1859.01 930.01 620.34 586 Ac-LTF$r8EYWAQL$AAAa-NH.sub.2 689 1786.97 788.56 1787.98 894.49 596.66 587 Ac-LTF$r8EYWAQL$AAa-NH.sub.2 690 1715.93 1138.57 1716.94 858.97 572.98 588 Ac-LTF$r8EYWAQL$Aa-NH.sub.2 691 1644.89 1144.98 1645.9 823.45 549.3 589 Ac-LTF$r8EYWAQL$a-NH.sub.2 692 1573.85 1113.71 1574.86 787.93 525.62
[0359] In the sequences shown above and elsewhere, the following abbreviations are used: "Nle" represents norleucine, "Aib" represents 2-aminoisobutyric acid, "Ac" represents acetyl, and "Pr" represents propionyl. Amino acids represented as "$" are alpha-Me S5-pentenyl-alanine olefin amino acids connected by an all-carbon crosslinker comprising one double bond. Amino acids represented as "$r5" are alpha-Me R5-pentenyl-alanine olefin amino acids connected by an all-carbon comprising one double bond. Amino acids represented as "$s8" are alpha-Me S8-octenyl-alanine olefin amino acids connected by an all-carbon crosslinker comprising one double bond. Amino acids represented as "$r8" are alpha-Me R8-octenyl-alanine olefin amino acids connected by an all-carbon crosslinker comprising one double bond. "Ahx" represents an aminocyclohexyl linker. The crosslinkers are linear all-carbon crosslinker comprising eight or eleven carbon atoms between the alpha carbons of each amino acid. Amino acids represented as "$/" are alpha-Me S5-pentenyl-alanine olefin amino acids that are not connected by any crosslinker. Amino acids represented as "$/r5" are alpha-Me R5-pentenyl-alanine olefin amino acids that are not connected by any crosslinker. Amino acids represented as "$/s8" are alpha-Me S8-octenyl-alanine olefin amino acids that are not connected by any crosslinker. Amino acids represented as "$/r8" are alpha-Me R8-octenyl-alanine olefin amino acids that are not connected by any crosslinker. Amino acids represented as "Amw" are alpha-Me tryptophan amino acids. Amino acids represented as "Aml" are alpha-Me leucine amino acids. Amino acids represented as "Amf" are alpha-Me phenylalanine amino acids. Amino acids represented as "2ff" are 2-fluoro-phenylalanine amino acids. Amino acids represented as "3ff" are 3-fluoro-phenylalanine amino acids. Amino acids represented as "St" are amino acids comprising two pentenyl-alanine olefin side chains, each of which is crosslinked to another amino acid as indicated. Amino acids represented as "St//" are amino acids comprising two pentenyl-alanine olefin side chains that are not crosslinked. Amino acids represented as "% St" are amino acids comprising two pentenyl-alanine olefin side chains, each of which is crosslinked to another amino acid as indicated via fully saturated hydrocarbon crosslinks. Amino acids represented as "Ba" are beta-alanine. The lower-case character "e" or "z" within the designation of a crosslinked amino acid (e.g. "$er8" or "$zr8") represents the configuration of the double bond (E or Z, respectively). In other contexts, lower-case letters such as "a" or "f" represent D amino acids (e.g. D-alanine, or D-phenylalanine, respectively). Amino acids designated as "NmW" represent N-methyltryptophan. Amino acids designated as "NmY" represent N-methyltyrosine. Amino acids designated as "NmA" represent N-methylalanine. "Kbio" represents a biotin group attached to the side chain amino group of a lysine residue. Amino acids designated as "Sar" represent sarcosine. Amino acids designated as "Cha" represent cyclohexyl alanine. Amino acids designated as "Cpg" represent cyclopentyl glycine. Amino acids designated as "Chg" represent cyclohexyl glycine. Amino acids designated as "Cba" represent cyclobutyl alanine. Amino acids designated as "F4I" represent 4-iodo phenylalanine. "7L" represents N15 isotopic leucine. Amino acids designated as "F3Cl" represent 3-chloro phenylalanine. Amino acids designated as "F4cooh" represent 4-carboxy phenylalanine. Amino acids designated as "F34F2" represent 3,4-difluoro phenylalanine. Amino acids designated as "6clW" represent 6-chloro tryptophan. Amino acids designated as "$rda6" represent alpha-Me R6-hexynyl-alanine alkynyl amino acids, crosslinked via a dialkyne bond to a second alkynyl amino acid. Amino acids designated as "$da5" represent alpha-Me S5-pentynyl-alanine alkynyl amino acids, wherein the alkyne forms one half of a dialkyne bond with a second alkynyl amino acid. Amino acids designated as "$ra9" represent alpha-Me R9-nonynyl-alanine alkynyl amino acids, crosslinked via an alkyne metathesis reaction with a second alkynyl amino acid. Amino acids designated as "$a6" represent alpha-Me S6-hexynyl-alanine alkynyl amino acids, crosslinked via an alkyne metathesis reaction with a second alkynyl amino acid. The designation "iso1" or "iso2" indicates that the peptidomimetic macrocycle is a single isomer.
[0360] Amino acids designated as "Cit" represent citrulline. Amino acids designated as "Cou4", "Cou6", "Cou7" and "Cou8", respectively, represent the following structures:
##STR00044## ##STR00045##
[0361] In some embodiments, a peptidomimetic macrocycle is obtained in more than one isomer, for example due to the configuration of a double bond within the structure of the crosslinker (E vs Z). Such isomers can or cannot be separable by conventional chromatographic methods. In some embodiments, one isomer has improved biological properties relative to the other isomer. In one embodiment, an E crosslinker olefin isomer of a peptidomimetic macrocycle has better solubility, better target affinity, better in vivo or in vitro efficacy, higher helicity, or improved cell permeability relative to its Z counterpart. In another embodiment, a Z crosslinker olefin isomer of a peptidomimetic macrocycle has better solubility, better target affinity, better in vivo or in vitro efficacy, higher helicity, or improved cell permeability relative to its E counterpart.
[0362] Table 3c shows exemplary peptidomimetic macrocycle:
TABLE-US-00008 TABLE 3c Structure SP154 (SEQ ID NO: 163) ##STR00046## Chemical Formula: C.sub.87H.sub.125N.sub.17O.sub.21 Exact Mass: 1743.92 Molecular Weight: 1745.02 SP115 (SEQ ID NO: 124) ##STR00047## Chemical Formula: C.sub.85H.sub.125N.sub.17O.sub.19 Exact Mass: 1687.93 Molecular Weight: 1689.00 SP114 (SEQ ID NO: 123) ##STR00048## Chemical Formula: C.sub.85H.sub.125N.sub.17O.sub.19 Exact Mass: 1687.93 Molecular Weight: 1689.00 SP99 (SEQ ID NO: 108) ##STR00049## Chemical Formula: C.sub.84H.sub.122ClN.sub.17O.sub.19 Exact Mass: 1707.88 Molecular Weight: 1709.42 SP388 (SEQ ID NO: 397) ##STR00050## Chemical Formula: C.sub.91H.sub.135N.sub.18O.sub.19 Exact Mass: 1785.02 Molecular Weight: 1786.16 SP331 (SEQ ID NO: 340) ##STR00051## Chemical Formula: C.sub.95H.sub.140N.sub.20O.sub.23 Exact Mass: 1929.04 Molecular Weight: 1930.25 SP445 (SEQ ID NO: 454) ##STR00052## Chemical Formula: C.sub.95H.sub.142N.sub.20O.sub.23 Exact Mass: 1931.06 Molecular Weight: 1932.26 SP351 (SEQ ID NO: 360) ##STR00053## Chemical Formula: C.sub.96H.sub.140N.sub.20O.sub.24 Exact Mass: 1957.03 Molecular Weight: 1958.26 SP71 (SEQ ID NO: 80) ##STR00054## Chemical Formula: C.sub.90H.sub.134N.sub.18O.sub.19 Exact Mass: 1771.01 Molecular Weight: 1772.14 SP69 (SEQ ID NO: 78) ##STR00055## Chemical Formula: C.sub.90H.sub.134N.sub.18O.sub.19 Exact Mass: 1771.01 Molecular Weight: 1772.14 SP7 (SEQ ID NO: 16) ##STR00056## Chemical Formula: C.sub.90H.sub.127N.sub.17O.sub.19 Exact Mass: 1749.95 Molecular Weight: 1751.07 SP160 (SEQ ID NO: 169) ##STR00057## Chemical Formula: C.sub.87H.sub.125F.sub.2N.sub.17O.sub.21 Exact Mass: 1781.92 Molecular Weight: 1783.02 SP315 (SEQ ID NO: 324) ##STR00058## Chemical Formula: C.sub.93H.sub.138N.sub.20O.sub.21 Exact Mass: 1871.03 Molecular Weight: 1872.21 SP249 (SEQ ID NO: 258) ##STR00059## Chemical Formula: C.sub.94H.sub.136N.sub.18O.sub.22 Exact Mass: 1869.01 Molecular Weight: 1870.19 SP437 (SEQ ID NO: 446) ##STR00060## Chemical Formula: C.sub.95H.sub.143N.sub.21O.sub.21 Exact Mass: 1914.08 Molecular Weight: 1915.28 SP349 (SEQ ID NO: 358) ##STR00061## Chemical Formula: C.sub.97H.sub.140N.sub.20O.sub.24 Exact Mass: 1969.03 Molecular Weight: 1970.27 SP555 (SEQ ID NO: 464) ##STR00062## Chemical Formula: C.sub.95H.sub.139ClN.sub.20O.sub.23 Exact Mass: 1963.00 Molecular Weight: 1964.69 SP557 (SEQ ID NO: 466) ##STR00063## Chemical Formula: C.sub.104H.sub.155N.sub.23O.sub.26 Exact Mass: 2142.15 Molecular Weight: 2143.48 SP558 (SEQ ID NO: 467) ##STR00064## Chemical Formula: C.sub.95H.sub.138F.sub.2N.sub.20O.sub.23 Exact Mass: 1965.02 Molecular Weight: 1966.23 SP367 (SEQ ID NO: 376) ##STR00065## SP562 (SEQ ID NO: 471) ##STR00066## Chemical Formula: C.sub.96H.sub.142N.sub.20O.sub.23 Exact Mass: 1943.06 Molecular Weight: 1944.27 SP564 (SEQ ID NO: 473) ##STR00067## Chemical Formula: C.sub.96H.sub.142N.sub.20O.sub.23 Exact Mass: 1943.06 Molecular Weight: 1944.27 SP566 (SEQ ID NO: 475) ##STR00068## SP567 (SEQ ID NO: 476) ##STR00069## Chemical Formula: C.sub.96H.sub.142N.sub.20O.sub.23 Exact Mass: 1943.06 Molecular Weight: 1944.27 SP572 (SEQ ID NO: 481) ##STR00070## Chemical Formula: C.sub.95H.sub.140N.sub.20O.sub.23 Exact Mass: 1929.04 Molecular Weight: 1930.25 SP573 (SEQ ID NO: 482) ##STR00071## Chemical Formula: C.sub.95H.sub.140N.sub.20O.sub.23 Exact Mass: 1929.04 Molecular Weight: 1930.25 SP578 (SEQ ID NO: 487) ##STR00072## Chemical Formula: C.sub.95H.sub.140N.sub.20O.sub.23 Exact Mass: 1929.04 Molecular Weight: 1930.25 SP664 (SEQ ID NO: 572) ##STR00073## Chemical Formula: C.sub.95H.sub.134N.sub.20O.sub.23 Exact Mass: 1922.99 Molecular Weight: 1924.20 SP664 (SEQ ID NO: 572) ##STR00074## Chemical Formula: C.sub.95H.sub.134N.sub.20O.sub.23 Exact Mass: 1922.99 Molecular Weight: 1924.20 (SEQ ID NO: 1500) ##STR00075## Chemical Formula: C.sub.96H.sub.136N.sub.20O.sub.23 Exact Mass: 1937.01 Molecular Weight: 1938.23
[0363] In some embodiments, peptidomimetic macrocycles exclude peptidomimetic macrocycles shown in Table 4a:
TABLE-US-00009 TABLE 4a Number Sequence SEQ ID NO: 765 L$r5QETFSD$s8WKLLPEN 693 766 LSQ$r5TFSDLW$s8LLPEN 694 767 LSQE$r5FSDLWK$s8LPEN 695 768 LSQET$r5SDLWKL$s8PEN 696 769 LSQETF$r5DLWKLL$s8EN 697 770 LXQETFS$r5LWKLLP$s8N 698 771 LSQETFSD$r5WKLLPE$s8 699 772 LSQQTF$r5DLWKLL$s8EN 700 773 LSQETF$r5DLWKLL$s8QN 701 774 LSQQTF$r5DLWKLL$s8QN 702 775 LSQETF$r5NLWKLL$s8QN 703 776 LSQQTF$r5NLWKLL$s8QN 704 777 LSQQTF$r5NLWRLL$s8QN 705 778 QSQQTF$r5NLWKLL$s8QN 706 779 QSQQTF$r5NLWRLL$s8QN 707 780 QSQQTA$r5NLWRLL$s8QN 708 781 L$r8QETFSD$WKLLPEN 709 782 LSQ$r8TFSDLW$LLPEN 710 783 LSQE$r8FSDLWK$LPEN 711 784 LSQET$r8SDLWKL$PEN 712 785 LSQETF$r8DLWKLL$EN 713 786 LXQETFS$r8LWKLLP$N 714 787 LSQETFSD$r8WKLLPE$ 715 788 LSQQTF$r8DLWKLL$EN 716 789 LSQETF$r8DLWKLL$QN 717 790 LSQQTF$r8DLWKLL$QN 718 791 LSQETF$r8NLWKLL$QN 719 792 LSQQTF$r8NLWKLL$QN 720 793 LSQQTF$r8NLWRLL$QN 721 794 QSQQTF$r8NLWKLL$QN 722 795 QSQQTF$r8NLWRLL$QN 723 796 QSQQTA$r8NLWRLL$QN 724 797 QSQQTF$r8NLWRKK$QN 725 798 QQTF$r8DLWRLL$EN 726 799 QQTF$r8DLWRLL$ 727 800 LSQQTF$DLW$LL 728 801 QQTF$DLW$LL 729 802 QQTA$r8DLWRLL$EN 730 803 QSQQTF$r5NLWRLL$s8QN 731 (dihydroxylated olefin) 804 QSQQTA$r5NLWRLL$s8QN 732 (dihydroxylated olefin) 805 QSQQTF$r8DLWRLL$QN 733 806 QTF$r8NLWRLL$ 734 807 QSQQTF$NLW$LLPQN 735 808 QS$QTF$NLWRLLPQN 736 809 $TFS$LWKLL 737 810 ETF$DLW$LL 738 811 QTF$NLW$LL 739 812 $SQE$FSNLWKLL 740
In Table 4a, X represents S or any amino acid. Peptides shown can comprise an N-terminal capping group such as acetyl or an additional linker such as beta-alanine between the capping group and the start of the peptide sequence.
[0364] In some embodiments, peptidomimetic macrocycles do not comprise a peptidomimetic macrocycle structure as shown in Table 4a.
[0365] In other embodiments, peptidomimetic macrocycles exclude peptidomimetic macrocycles shown in Table 4b.
TABLE-US-00010 TABLE 4b Observed SEQ Exact mass SP# Sequence ID NO: Mass M + 2 (m/e) 813 Ac-LSQETF$r8DLWKLL$EN-NH.sub.2 741 2068.13 1035.07 1035.36 814 Ac-LSQETF$r8NLWKLL$QN-NH.sub.2 742 2066.16 1034.08 1034.31 815 Ac-LSQQTF$r8NLWRLL$QN-NH.sub.2 743 2093.18 1047.59 1047.73 816 Ac-QSQQTF$r8NLWKLL$QN-NH.sub.2 744 2080.15 1041.08 1041.31 817 Ac-QSQQTF$r8NLWRLL$QN-NH.sub.2 745 2108.15 1055.08 1055.32 818 Ac-QSQQTA$r8NLWRLL$QN-NH.sub.2 746 2032.12 1017.06 1017.24 819 Ac-QAibQQTF$r8NLWRLL$QN-NH.sub.2 747 2106.17 1054.09 1054.34 820 Ac-QSQQTFSNLWRLLPQN-NH.sub.2 748 2000.02 1001.01 1001.26 821 Ac-QSQQTF$/r8NLWRLL$/QN-NH.sub.2 749 2136.18 1069.09 1069.37 822 Ac-QSQAibTF$r8NLWRLL$QN-NH.sub.2 750 2065.15 1033.58 1033.71 823 Ac-QSQQTF$r8NLWRLL$AN-NH.sub.2 751 2051.13 1026.57 1026.70 824 Ac-ASQQTF$r8NLWRLL$QN-NH.sub.2 752 2051.13 1026.57 1026.90 825 Ac-QSQQTF$r8ALWRLL$QN-NH.sub.2 753 2065.15 1033.58 1033.41 826 Ac-QSQETF$r8NLWRLL$QN-NH.sub.2 754 2109.14 1055.57 1055.70 827 Ac-RSQQTF$r8NLWRLL$QN-NH.sub.2 755 2136.20 1069.10 1069.17 828 Ac-RSQQTF$r8NLWRLL$EN-NH.sub.2 756 2137.18 1069.59 1069.75 829 Ac-LSQETFSDLWKLLPEN-NH.sub.2 757 1959.99 981.00 981.24 830 Ac-QSQ$TFS$LWRLLPQN-NH.sub.2 758 2008.09 1005.05 1004.97 831 Ac-QSQQ$FSN$WRLLPQN-NH.sub.2 759 2036.06 1019.03 1018.86 832 Ac-QSQQT$SNL$RLLPQN-NH.sub.2 760 1917.04 959.52 959.32 833 Ac-QSQQTF$NLW$LLPQN-NH.sub.2 761 2007.06 1004.53 1004.97 834 Ac-RTQATF$r8NQWAibANle$TNAibTR-NH.sub.2 762 2310.26 1156.13 1156.52 835 Ac-QSQQTF$r8NLWRLL$RN-NH.sub.2 763 2136.20 1069.10 1068.94 836 Ac-QSQRTF$r8NLWRLL$QN-NH.sub.2 764 2136.20 1069.10 1068.94 837 Ac-QSQQTF$r8NNleWRLL$QN-NH.sub.2 765 2108.15 1055.08 1055.44 838 Ac-QSQQTF$r8NLWRNleL$QN-NH.sub.2 766 2108.15 1055.08 1055.84 839 Ac-QSQQTF$r8NLWRLNle$QN-NH.sub.2 767 2108.15 1055.08 1055.12 840 Ac-QSQQTY$r8NLWRLL$QN-NH.sub.2 768 2124.15 1063.08 1062.92 841 Ac-RAibQQTF$r8NLWRLL$QN-NH.sub.2 769 2134.22 1068.11 1068.65 842 Ac-MPRFMDYWEGLN-NH.sub.2 770 1598.70 800.35 800.45 843 Ac-RSQQRF$r8NLWRLL$QN-NH.sub.2 771 2191.25 1096.63 1096.83 844 Ac-QSQQRF$r8NLWRLL$QN-NH.sub.2 772 2163.21 1082.61 1082.87 845 Ac-RAibQQRF$r8NLWRLL$QN-NH.sub.2 773 2189.27 1095.64 1096.37 846 Ac-RSQQRF$r8NFWRLL$QN-NH.sub.2 774 2225.23 1113.62 1114.37 847 Ac-RSQQRF$r8NYWRLL$QN-NH.sub.2 775 2241.23 1121.62 1122.37 848 Ac-RSQQTF$r8NLWQLL$QN-NH.sub.2 776 2108.15 1055.08 1055.29 849 Ac-QSQQTF$r8NLWQAmlL$QN-NH.sub.2 777 2094.13 1048.07 1048.32 850 Ac-QSQQTF$r8NAmlWRLL$QN-NH.sub.2 778 2122.17 1062.09 1062.35 851 Ac-NlePRF$r8DYWEGL$QN-NH.sub.2 779 1869.98 935.99 936.20 852 Ac-NlePRF$r8NYWRLL$QN-NH.sub.2 780 1952.12 977.06 977.35 853 Ac-RF$r8NLWRLL$Q-NH.sub.2 781 1577.96 789.98 790.18 854 Ac-QSQQTF$r8N2ffWRLL$QN-NH.sub.2 782 2160.13 1081.07 1081.40 855 Ac-QSQQTF$r8N3ffWRLL$QN-NH.sub.2 783 2160.13 1081.07 1081.34 856 Ac-QSQQTF#r8NLWRLL#QN-NH.sub.2 784 2080.12 1041.06 1041.34 857 Ac-RSQQTA$r8NLWRLL$QN-NH.sub.2 785 2060.16 1031.08 1031.38 858 Ac-QSQQTF%r8NLWRLL%QN-NH.sub.2 786 2110.17 1056.09 1056.55 859 HepQSQ$TFSNLWRLLPQN-NH.sub.2 787 2051.10 1026.55 1026.82 860 HepQSQ$TF$r8NLWRLL$QN-NH.sub.2 788 2159.23 1080.62 1080.89 861 Ac-QSQQTF$r8NL6clWRLL$QN-NH.sub.2 789 2142.11 1072.06 1072.35 862 Ac-QSQQTF$r8NLMe6clwRLL$QN-NH.sub.2 790 2156.13 1079.07 1079.27 863 Ac-LTFEHYWAQLTS-NH.sub.2 791 1535.74 768.87 768.91 864 Ac-LTF$HYW$QLTS-NH.sub.2 792 1585.83 793.92 794.17 865 Ac-LTFE$YWA$LTS-NH.sub.2 793 1520.79 761.40 761.67 866 Ac-LTF$zr8HYWAQL$zS-NH.sub.2 794 1597.87 799.94 800.06 867 Ac-LTF$r8HYWRQL$S-NH.sub.2 795 1682.93 842.47 842.72 868 Ac-QS$QTFStNLWRLL$s8QN-NH.sub.2 796 2145.21 1073.61 1073.90 869 Ac-QSQQTASNLWRLLPQN-NH.sub.2 797 1923.99 963.00 963.26 870 Ac-QSQQTA$/r8NLWRLL$/QN-NH.sub.2 798 2060.15 1031.08 1031.24 871 Ac-ASQQTF$/r8NLWRLL$/QN-NH.sub.2 799 2079.16 1040.58 1040.89 872 Ac-$SQQ$FSNLWRLLAibQN-NH.sub.2 800 2009.09 1005.55 1005.86 873 Ac-QS$QTF$NLWRLLAibQN-NH.sub.2 801 2023.10 1012.55 1012.79 874 Ac-QSQQ$FSN$WRLLAibQN-NH.sub.2 802 2024.06 1013.03 1013.31 875 Ac-QSQQTF$NLW$LLAibQN-NH.sub.2 803 1995.06 998.53 998.87 876 Ac-QSQQTFS$LWR$LAibQN-NH.sub.2 804 2011.06 1006.53 1006.83 877 Ac-QSQQTFSNLW$LLA$N-NH.sub.2 805 1940.02 971.01 971.29 878 Ac-$/SQQ$/FSNLWRLLAibQN-NH.sub.2 806 2037.12 1019.56 1019.78 879 Ac-QS$/QTF$/NLWRLLAibQN-NH.sub.2 807 2051.13 1026.57 1026.90 880 Ac-QSQQ$/FSN$/WRLLAibQN-NH.sub.2 808 2052.09 1027.05 1027.36 881 Ac-QSQQTF$/NLW$/LLAibQN-NH.sub.2 809 2023.09 1012.55 1013.82 882 Ac-QSQ$TFS$LWRLLAibQN-NH.sub.2 810 1996.09 999.05 999.39 883 Ac-QSQ$/TFS$/LWRLLAibQN-NH.sub.2 811 2024.12 1013.06 1013.37 884 Ac-QS$/QTFSt//NLWRLL$/s8QN-NH.sub.2 812 2201.27 1101.64 1102.00 885 Ac-$r8SQQTFS$LWRLLAibQN-NH.sub.2 813 2038.14 1020.07 1020.23 886 Ac-QSQ$r8TFSNLW$LLAibQN-NH.sub.2 814 1996.08 999.04 999.32 887 Ac-QSQQTFS$r8LWRLLA$N-NH.sub.2 815 2024.12 1013.06 1013.37 888 Ac-QS$r5QTFStNLW$LLAibQN-NH.sub.2 816 2032.12 1017.06 1017.39 889 Ac-$/r8SQQTFS$/LWRLLAibQN-NH.sub.2 817 2066.17 1034.09 1034.80 890 Ac-QSQ$/r8TFSNLW$/LLAibQN-NH.sub.2 818 2024.11 1013.06 1014.34 891 Ac-QSQQTFS$/r8LWRLLA$/N-NH.sub.2 819 2052.15 1027.08 1027.16 892 Ac-QS$/r5QTFSt//NLW$/LLAibQN-NH.sub.2 820 2088.18 1045.09 1047.10 893 Ac-QSQQTFSNLWRLLAibQN-NH.sub.2 821 1988.02 995.01 995.31 894 Hep/QSQ$/TF$/r8NLWRLL$/QN-NH.sub.2 822 2215.29 1108.65 1108.93 895 Ac-ASQQTF$r8NLRWLL$QN-NH.sub.2 823 2051.13 1026.57 1026.90 896 Ac-QSQQTF$/r8NLWRLL$/Q-NH.sub.2 824 2022.14 1012.07 1012.66 897 Ac-QSQQTF$r8NLWRLL$Q-NH.sub.2 825 1994.11 998.06 998.42 898 Ac-AAARAA$r8AAARAA$AA-NH.sub.2 826 1515.90 758.95 759.21 899 Ac-LTFEHYWAQLTSA-NH.sub.2 827 1606.78 804.39 804.59 900 Ac-LTF$r8HYWAQL$SA-NH.sub.2 828 1668.90 835.45 835.67 901 Ac-ASQQTFSNLWRLLPQN-NH.sub.2 829 1943.00 972.50 973.27 902 Ac-QS$QTFStNLW$r5LLAibQN-NH.sub.2 830 2032.12 1017.06 1017.30 903 Ac-QSQQTFAibNLWRLLAibQN-NH.sub.2 831 1986.04 994.02 994.19 904 Ac-QSQQTFNleNLWRLLNleQN-NH.sub.2 832 2042.11 1022.06 1022.23 905 Ac-QSQQTF$/r8NLWRLLAibQN-NH.sub.2 833 2082.14 1042.07 1042.23 906 Ac-QSQQTF$/r8NLWRLLNleQN-NH.sub.2 834 2110.17 1056.09 1056.29 907 Ac-QSQQTFAibNLWRLL$/QN-NH.sub.2 835 2040.09 1021.05 1021.25 908 Ac-QSQQTFNleNLWRLL$/QN-NH.sub.2 836 2068.12 1035.06 1035.31 909 Ac-QSQQTF%r8NL6clWRNleL%QN-NH.sub.2 837 2144.13 1073.07 1073.32 910 Ac-QSQQTF%r8NLMe6clWRLL%QN-NH.sub.2 838 2158.15 1080.08 1080.31 911 Ac-FNle$YWE$L-NH.sub.2 839 1160.63 -- 1161.70 912 Ac-F$r8AYWELL$A-NH.sub.2 840 1344.75 -- 1345.90 913 Ac-F$r8AYWQLL$A-NH.sub.2 841 1343.76 -- 1344.83 914 Ac-NlePRF$r8NYWELL$QN-NH.sub.2 842 1925.06 963.53 963.69 915 Ac-NlePRF$r8DYWRLL$QN-NH.sub.2 843 1953.10 977.55 977.68 916 Ac-NlePRF$r8NYWRLL$Q-NH.sub.2 844 1838.07 920.04 920.18 917 Ac-NlePRF$r8NYWRLL$-NH.sub.2 845 1710.01 856.01 856.13 918 Ac-QSQQTF$r8DLWRLL$QN-NH.sub.2 846 2109.14 1055.57 1055.64 919 Ac-QSQQTF$r8NLWRLL$EN-NH.sub.2 847 2109.14 1055.57 1055.70 920 Ac-QSQQTF$r8NLWRLL$QD-NH.sub.2 848 2109.14 1055.57 1055.64 921 Ac-QSQQTF$r8NLWRLL$S-NH.sub.2 849 1953.08 977.54 977.60 922 Ac-ESQQTF$r8NLWRLL$QN-NH.sub.2 850 2109.14 1055.57 1055.70 923 Ac-LTF$r8NLWRNleL$Q-NH.sub.2 851 1635.99 819.00 819.10 924 Ac-LRF$r8NLWRNleL$Q-NH.sub.2 852 1691.04 846.52 846.68 925 Ac-QSQQTF$r8NWWRNleL$QN-NH.sub.2 853 2181.15 1091.58 1091.64 926 Ac-QSQQTF$r8NLWRNleL$Q-NH.sub.2 854 1994.11 998.06 998.07 927 Ac-QTF$r8NLWRNleL$QN-NH.sub.2 855 1765.00 883.50 883.59 928 Ac-NlePRF$r8NWWRLL$QN-NH.sub.2 856 1975.13 988.57 988.75 929 Ac-NlePRF$r8NWWRLL$A-NH.sub.2 857 1804.07 903.04 903.08 930 Ac-TSFAEYWNLLNH.sub.2 858 1467.70 734.85 734.90 931 Ac-QTF$r8HWWSQL$S-NH.sub.2 859 1651.85 826.93 827.12 932 Ac-FM$YWE$L-NH.sub.2 860 1178.58 -- 1179.64 933 Ac-QTFEHWWSQLLS-NH.sub.2 861 1601.76 801.88 801.94 934 Ac-QSQQTF$r8NLAmwRLNle$QN-NH.sub.2 862 2122.17 1062.09 1062.24 935 Ac-FMAibY6clWEAc3cL-NH.sub.2 863 1130.47 -- 1131.53 936 Ac-FNle$Y6clWE$L-NH.sub.2 864 1194.59 -- 1195.64 937 Ac-F$zr8AY6clWEAc3cL$z-NH.sub.2 865 1277.63 639.82 1278.71 938 Ac-F$r8AY6clWEAc3cL$A-NH.sub.2 866 1348.66 -- 1350.72 939 Ac-NlePRF$r8NY6clWRLL$QN-NH.sub.2 867 1986.08 994.04 994.64 940 Ac-AF$r8AAWALA$A-NH.sub.2 868 1223.71 -- 1224.71 941 Ac-TF$r8AAWRLA$Q-NH.sub.2 869 1395.80 698.90 399.04 942 Pr-TF$r8AAWRLA$Q-NH.sub.2 870 1409.82 705.91 706.04 943 Ac-QSQQTF%r8NLWRNleL%QN-NH.sub.2 871 2110.17 1056.09 1056.22 944 Ac-LTF%r8HYWAQL%SA-NH.sub.2 872 1670.92 836.46 836.58 945 Ac-NlePRF%r8NYWRLL%QN-NH.sub.2 873 1954.13 978.07 978.19 946 Ac-NlePRF%r8NY6clWRLL%QN-NH.sub.2 874 1988.09 995.05 995.68 947 Ac-LTF%r8HY6clWAQL%S-NH.sub.2 875 1633.84 817.92 817.93 948 Ac-QS%QTF%StNLWRLL%s8QN-NH.sub.2 876 2149.24 1075.62 1075.65 949 Ac-LTF%r8HY6clWRQL%S-NH.sub.2 877 1718.91 860.46 860.54 950 Ac-QSQQTF%r8NL6clWRLL%QN-NH.sub.2 878 2144.13 1073.07 1073.64 951 Ac-%r8SQQTFS%LWRLLAibQN-NH.sub.2 879 2040.15 1021.08 1021.13 952 Ac-LTF%r8HYWAQL%S-NH.sub.2 880 1599.88 800.94 801.09 953 Ac-TSF%r8QYWNLL%P-NH.sub.2 881 1602.88 802.44 802.58 954 Ac-LTFEHYWAQLTS-NH.sub.2 882 1535.74 768.87 769.5 955 Ac-F$er8AY6clWEAc3cL$e-NH.sub.2 883 1277.63 639.82 1278.71 956 Ac-AF$r8AAWALA$A-NH.sub.2 884 1277.63 639.82 1277.84 957 Ac-TF$r8AAWRLA$Q-NH.sub.2 885 1395.80 698.90 699.04 958 Pr-TF$r8AAWRLA$Q-NH.sub.2 886 1409.82 705.91 706.04 959 Ac-LTF$er8HYWAQL$eS-NH.sub.2 887 1597.87 799.94 800.44 960 Ac-CCPGCCBaQSQQTF$r8NLWRLL$QN-NH.sub.2 888 2745.30 1373.65 1372.99 961 Ac-CCPGCCBaQSQQTA$r8NLWRLL$QN-NH.sub.2 889 2669.27 1335.64 1336.09 962 Ac-CCPGCCBaNlePRF$r8NYWRLL$QN-NH.sub.2 890 2589.26 1295.63 1296.2 963 Ac-LTF$/r8HYWAQLS/S-NH.sub.2 891 1625.90 813.95 814.18 964 Ac-F%r8HY6clWRAc3cL%-NH.sub.2 892 1372.72 687.36 687.59 965 Ac-QTF%r8HWWSQL%S-NH.sub.2 893 1653.87 827.94 827.94 966 Ac-LTA$r8HYWRQL$S-NH.sub.2 894 1606.90 804.45 804.66 967 Ac-Q$r8QQTFSN$WRLLAibQN-NH.sub.2 895 2080.12 1041.06 1041.61 968 Ac-QSQQ$r8FSNLWR$LAibQN-NH.sub.2 896 2066.11 1034.06 1034.58 969 Ac-F$r8AYWEAc3cL$A-NH.sub.2 897 1314.70 658.35 1315.88 970 Ac-F$r8AYWEAc3cL$S-NH.sub.2 898 1330.70 666.35 1331.87 971 Ac-F$r8AYWEAc3cL$Q-NH.sub.2 899 1371.72 686.86 1372.72 972 Ac-F$r8AYWEAibL$S-NH.sub.2 900 1332.71 667.36 1334.83 973 Ac-F$r8AYWEAL$S-NH.sub.2 901 1318.70 660.35 1319.73 974 Ac-F$r8AYWEQL$S-NH.sub.2 902 1375.72 688.86 1377.53 975 Ac-F$r8HYWEQL$S-NH.sub.2 903 1441.74 721.87 1443.48 976 Ac-F$r8HYWAQL$S-NH.sub.2 904 1383.73 692.87 1385.38 977 Ac-F$r8HYWAAc3cL$S-NH.sub.2 905 1338.71 670.36 1340.82 978 Ac-F$r8HYWRAc3cL$S-NH.sub.2 906 1423.78 712.89 713.04 979 Ac-F$r8AYWEAc3cL#A-NH.sub.2 907 1300.69 651.35 1302.78 980 Ac-NlePTF%r8NYWRLL%QN-NH.sub.2 908 1899.08 950.54 950.56 981 Ac-TF$r8AAWRAL$Q-NH.sub.2 909 1395.80 698.90 699.13 982 Ac-TSF%r8HYWAQL%S-NH.sub.2 910 1573.83 787.92 787.98 983 Ac-F%r8AY6clWEAc3cL%A-NH.sub.2 911 1350.68 676.34 676.91 984 Ac-LTF$r8HYWAQI$S-NH.sub.2 912 1597.87 799.94 800.07 985 Ac-LTF$r8HYWAQNle$S-NH.sub.2 913 1597.87 799.94 800.07 986 Ac-LTF$r8HYWAQL$A-NH.sub.2 914 1581.87 791.94 792.45 987 Ac-LTF$r8HYWAQL$Abu-NH.sub.2 915 1595.89 798.95 799.03 988 Ac-LTF$r8HYWAbuQL$S-NH.sub.2 916 1611.88 806.94 807.47 989 Ac-LTF$er8AYWAQL$eS-NH.sub.2 917 1531.84 766.92 766.96 990 Ac-LAF$r8HYWAQL$S-NH.sub.2 918 1567.86 784.93 785.49 991 Ac-LAF$r8AYWAQL$S-NH.sub.2 919 1501.83 751.92 752.01 992 Ac-LTF$er8AYWAQL$eA-NH.sub.2 920 1515.85 758.93 758.97 993 Ac-LAF$r8AYWAQL$A-NH.sub.2 921 1485.84 743.92 744.05 994 Ac-LTF$r8NLWANleL$Q-NH.sub.2 922 1550.92 776.46 776.61 995 Ac-LTF$r8NLWANleL$A-NH.sub.2 923 1493.90 747.95 1495.6 996 Ac-LTF$r8ALWANleL$Q-NH.sub.2 924 1507.92 754.96 755 997 Ac-LAF$r8NLWANleL$Q-NH.sub.2 925 1520.91 761.46 761.96 998 Ac-LAF$r8ALWANleL$A-NH.sub.2 926 1420.89 711.45 1421.74 999 Ac-A$r8AYWEAc3cL$A-NH.sub.2 927 1238.67 620.34 1239.65 1000 Ac-F$r8AYWEAc3cL$AA-NH.sub.2 928 1385.74 693.87 1386.64 1001 Ac-F$r8AYWEAc3cL$Abu-NH.sub.2 929 1328.72 665.36 1330.17 1013 Ac-F$r8AYWEAc3cL$Nle-NH.sub.2 930 1356.75 679.38 1358.22 1014 Ac-F$r5AYWEAc3cL$s8A-NH.sub.2 931 1314.70 658.35 1315.51 1015 Ac-F$AYWEAc3cL$r8A-NH.sub.2 932 1314.70 658.35 1315.66 1016 Ac-F$r8AYWEAc3cl$A-NH.sub.2 933 1314.70 658.35 1316.18 1017 Ac-F$r8AYWEAc3cNle$A-NH.sub.2 934 1314.70 658.35 1315.66 1018 Ac-F$r8AYWEAmlL$A-NH.sub.2 935 1358.76 680.38 1360.21 1019 Ac-F$r8AYWENleL$A-NH.sub.2 936 1344.75 673.38 1345.71 1020 Ac-F$r8AYWQAc3cL$A-NH.sub.2 937 1313.72 657.86 1314.7 1021 Ac-F$r8AYWAAc3cL$A-NH.sub.2 938 1256.70 629.35 1257.56 1022 Ac-F$r8AYWAbuAc3cL$A-NH.sub.2 939 1270.71 636.36 1272.14 1023 Ac-F$r8AYWNleAc3cL$A-NH.sub.2 940 1298.74 650.37 1299.67 1024 Ac-F$r8AbuYWEAc3cL$A-NH.sub.2 941 1328.72 665.36 1329.65 1025 Ac-F$r8NleYWEAc3cL$A-NH.sub.2 942 1356.75 679.38 1358.66 1026 5-FAM-BaLTFEHYWAQLTS-NH.sub.2 943 1922.82 962.41 962.87 1027 5-FAM-BaLTF%r8HYWAQL%S-NH.sub.2 944 1986.96 994.48 994.97 1028 Ac-LTF$r8HYWAQhL$S-NH.sub.2 945 1611.88 806.94 807 1029 Ac-LTF$r8HYWAQTle$S-NH.sub.2 946 1597.87 799.94 799.97 1030 Ac-LTF$r8HYWAQAdm$S-NH.sub.2 947 1675.91 838.96 839.09 1031 Ac-LTF$r8HYWAQhCha$S-NH.sub.2 948 1651.91 826.96 826.98 1032 Ac-LTF$r8HYWAQCha$S-NH.sub.2 949 1637.90 819.95 820.02 1033 Ac-LTF$r8HYWAc6cQL$S-NH.sub.2 950 1651.91 826.96 826.98 1034 Ac-LTF$r8HYWAc5cQL$S-NH.sub.2 951 1637.90 819.95 820.02 1035 Ac-LThF$r8HYWAQL$S-NH.sub.2 952 1611.88 806.94 807 1036 Ac-LTIgl$r8HYWAQL$S-NH.sub.2 953 1625.90 813.95 812.99 1037 Ac-LTF$r8HYWAQChg$S-NH.sub.2 954 1623.88 812.94 812.99 1038 Ac-LTF$r8HYWAQF$S-NH.sub.2 955 1631.85 816.93 816.99 1039 Ac-LTF$r8HYWAQIgl$S-NH.sub.2 956 1659.88 830.94 829.94 1040 Ac-LTF$r8HYWAQCba$S-NH.sub.2 957 1609.87 805.94 805.96 1041 Ac-LTF$r8HYWAQCpg$S-NH.sub.2 958 1609.87 805.94 805.96 1042 Ac-LTF$r8HhYWAQL$S-NH.sub.2 959 1611.88 806.94 807 1043 Ac-F$r8AYWEAc3chL$A-NH.sub.2 960 1328.72 665.36 665.43 1044 Ac-F$r8AYWEAc3cTle$A-NH.sub.2 961 1314.70 658.35 1315.62 1045 Ac-F$r8AYWEAc3cAdm$A-NH.sub.2 962 1392.75 697.38 697.47 1046 Ac-F$r8AYWEAc3chCha$A-NH.sub.2 963 1368.75 685.38 685.34 1047 Ac-F$r8AYWEAc3cCha$A-NH.sub.2 964 1354.73 678.37 678.38 1048 Ac-F$r8AYWEAc6cL$A-NH.sub.2 965 1356.75 679.38 679.42 1049 Ac-F$r8AYWEAc5cL$A-NH.sub.2 966 1342.73 672.37 672.46 1050 Ac-hF$r8AYWEAc3cL$A-NH.sub.2 967 1328.72 665.36 665.43 1051 Ac-Igl$r8AYWEAc3cL$A-NH.sub.2 968 1342.73 672.37 671.5 1052 Ac-F$r8AYWEAc3cF$A-NH.sub.2 969 1348.69 675.35 675.35 1053 Ac-F$r8AYWEAc3cIgl$A-NH.sub.2 970 1376.72 689.36 688.37 1054 Ac-F$r8AYWEAc3cCba$A-NH.sub.2 971 1326.70 664.35 664.47 1055 Ac-F$r8AYWEAc3cCpg$A-NH.sub.2 972 1326.70 664.35 664.39 1056 Ac-F$r8AhYWEAc3cL$A-NH.sub.2 973 1328.72 665.36 665.43 1057 Ac-F$r8AYWEAc3cL$Q-NH.sub.2 974 1371.72 686.86 1372.87 1058 Ac-F$r8AYWEAibL$A-NH.sub.2 975 1316.72 659.36 1318.18 1059 Ac-F$r8AYWEAL$A-NH.sub.2 976 1302.70 652.35 1303.75 1060 Ac-LAF$r8AYWAAL$A-NH.sub.2 977 1428.82 715.41 715.49 1061 Ac-LTF$r8HYWAAc3cL$S-NH.sub.2 978 1552.84 777.42 777.5 1062 Ac-NleTF$r8HYWAQL$S-NH.sub.2 979 1597.87 799.94 800.04 1063 Ac-VTF$r8HYWAQL$S-NH.sub.2 980 1583.85 792.93 793.04 1064 Ac-FTF$r8HYWAQL$S-NH.sub.2 981 1631.85 816.93 817.02 1065 Ac-WTF$r8HYWAQL$S-NH.sub.2 982 1670.86 836.43 836.85 1066 Ac-RTF$r8HYWAQL$S-NH.sub.2 983 1640.88 821.44 821.9
1067 Ac-KTF$r8HYWAQL$S-NH.sub.2 984 1612.88 807.44 807.91 1068 Ac-LNleF$r8HYWAQL$S-NH.sub.2 985 1609.90 805.95 806.43 1069 Ac-LVF$r8HYWAQL$S-NH.sub.2 986 1595.89 798.95 798.93 1070 Ac-LFF$r8HYWAQL$S-NH.sub.2 987 1643.89 822.95 823.38 1071 Ac-LWF$r8HYWAQL$S-NH.sub.2 988 1682.90 842.45 842.55 1072 Ac-LRF$r8HYWAQL$S-NH.sub.2 989 1652.92 827.46 827.52 1073 Ac-LKF$r8HYWAQL$S-NH.sub.2 990 1624.91 813.46 813.51 1074 Ac-LTF$r8NleYWAQL$S-NH.sub.2 991 1573.89 787.95 788.05 1075 Ac-LTF$r8VYWAQL$S-NH.sub.2 992 1559.88 780.94 780.98 1076 Ac-LTF$r8FYWAQL$S-NH.sub.2 993 1607.88 804.94 805.32 1077 Ac-LTF$r8WYWAQL$S-NH.sub.2 994 1646.89 824.45 824.86 1078 Ac-LTF$r8RYWAQL$S-NH.sub.2 995 1616.91 809.46 809.51 1079 Ac-LTF$r8KYWAQL$S-NH.sub.2 996 1588.90 795.45 795.48 1080 Ac-LTF$r8HNleWAQL$S-NH.sub.2 997 1547.89 774.95 774.98 1081 Ac-LTF$r8HVWAQL$S-NH.sub.2 998 1533.87 767.94 767.95 1082 Ac-LTF$r8HFWAQL$S-NH.sub.2 999 1581.87 791.94 792.3 1083 Ac-LTF$r8HWWAQL$S-NH.sub.2 1000 1620.88 811.44 811.54 1084 Ac-LTF$r8HRWAQL$S-NH.sub.2 1001 1590.90 796.45 796.52 1085 Ac-LTF$r8HKWAQL$S-NH.sub.2 1002 1562.90 782.45 782.53 1086 Ac-LTF$r8HYWNleQL$S-NH.sub.2 1003 1639.91 820.96 820.98 1087 Ac-LTF$r8HYWVQL$S-NH.sub.2 1004 1625.90 813.92 814.03 1088 Ac-LTF$r8HYWFQL$S-NH.sub.2 1005 1673.90 837.95 838.03 1089 Ac-LTF$r8HYWWQL$S-NH.sub.2 1006 1712.91 857.46 857.5 1090 Ac-LTF$r8HYWKQL$S-NH.sub.2 1007 1654.92 828.46 828.49 1091 Ac-LTF$r8HYWANleL$S-NH.sub.2 1008 1582.89 792.45 792.52 1092 Ac-LTF$r8HYWAVL$S-NH.sub.2 1009 1568.88 785.44 785.49 1093 Ac-LTF$r8HYWAFL$S-NH.sub.2 1010 1616.88 809.44 809.47 1094 Ac-LTF$r8HYWAWL$S-NH.sub.2 1011 1655.89 828.95 829 1095 Ac-LTF$r8HYWARL$S-NH.sub.2 1012 1625.91 813.96 813.98 1096 Ac-LTF$r8HYWAQL$Nle-NH.sub.2 1013 1623.92 812.96 813.39 1097 Ac-LTF$r8HYWAQL$V-NH.sub.2 1014 1609.90 805.95 805.99 1098 Ac-LTF$r8HYWAQL$F-NH.sub.2 1015 1657.90 829.95 830.26 1099 Ac-LTF$r8HYWAQL$W-NH.sub.2 1016 1696.91 849.46 849.5 1100 Ac-LTF$r8HYWAQL$R-NH.sub.2 1017 1666.94 834.47 834.56 1101 Ac-LTF$r8HYWAQL$K-NH.sub.2 1018 1638.93 820.47 820.49 1102 Ac-Q$r8QQTFSN$WRLLAibQN-NH.sub.2 1019 2080.12 1041.06 1041.54 1103 Ac-QSQQ$r8FSNLWR$LAibQN-NH.sub.2 1020 2066.11 1034.06 1034.58 1104 Ac-LT2Pal$r8HYWAQL$S-NH.sub.2 1021 1598.86 800.43 800.49 1105 Ac-LT3Pal$r8HYWAQL$S-NH.sub.2 1022 1598.86 800.43 800.49 1106 Ac-LT4Pal$r8HYWAQL$S-NH.sub.2 1023 1598.86 800.43 800.49 1107 Ac-LTF2CF3$r8HYWAQL$S-NH.sub.2 1024 1665.85 833.93 834.01 1108 Ac-LTF2CN$r8HYWAQL$S-NH.sub.2 1025 1622.86 812.43 812.47 1109 Ac-LTF2Me$r8HYWAQL$S-NH.sub.2 1026 1611.88 806.94 807 1110 Ac-LTF3Cl$r8HYWAQL$S-NH.sub.2 1027 1631.83 816.92 816.99 1111 Ac-LTF4CF3$r8HYWAQL$S-NH.sub.2 1028 1665.85 833.93 833.94 1113 Ac-LTF4tBu$r8HYWAQL$S-NH.sub.2 1029 1653.93 827.97 828.02 1114 Ac-LTF5F$r8HYWAQL$S-NH.sub.2 1030 1687.82 844.91 844.96 1115 Ac-LTF$r8HY3BthAAQL$S-NH.sub.2 1031 1614.83 808.42 808.48 1116 Ac-LTF2Br$r8HYWAQL$S-NH.sub.2 1032 1675.78 838.89 838.97 1117 Ac-LTF4Br$r8HYWAQL$S-NH.sub.2 1033 1675.78 838.89 839.86 1118 Ac-LTF2Cl$r8HYWAQL$S-NH.sub.2 1034 1631.83 816.92 816.99 1119 Ac-LTF4Cl$r8HYWAQL$S-NH.sub.2 1035 1631.83 816.92 817.36 1120 Ac-LTF3CN$r8HYWAQL$S-NH.sub.2 1036 1622.86 812.43 812.47 1121 Ac-LTF4CN$r8HYWAQL$S-NH.sub.2 1037 1622.86 812.43 812.47 1122 Ac-LTF34Cl2$r8HYWAQL$S-NH.sub.2 1038 1665.79 833.90 833.94 1123 Ac-LTF34F2$r8HYWAQL$S-NH.sub.2 1039 1633.85 817.93 817.95 1124 Ac-LTF35F2$r8HYWAQL$S-NH.sub.2 1040 1633.85 817.93 817.95 1125 Ac-LTDip$r8HYWAQL$S-NH.sub.2 1041 1673.90 837.95 838.01 1126 Ac-LTF2F$r8HYWAQL$S-NH.sub.2 1042 1615.86 808.93 809 1127 Ac-LTF3F$r8HYWAQL$S-NH.sub.2 1043 1615.86 808.93 809 1128 Ac-LTF4F$r8HYWAQL$S-NH.sub.2 1044 1615.86 808.93 809 1129 Ac-LTF4I$r8HYWAQL$S-NH.sub.2 1045 1723.76 862.88 862.94 1130 Ac-LTF3Me$r8HYWAQL$S-NH.sub.2 1046 1611.88 806.94 807.07 1131 Ac-LTF4Me$r8HYWAQL$S-NH.sub.2 1047 1611.88 806.94 807 1132 Ac-LT1Nal$r8HYWAQL$S-NH.sub.2 1048 1647.88 824.94 824.98 1133 Ac-LT2Nal$r8HYWAQL$S-NH.sub.2 1049 1647.88 824.94 825.06 1134 Ac-LTF3CF3$r8HYWAQL$S-NH.sub.2 1050 1665.85 833.93 834.01 1135 Ac-LTF4NO2$r8HYWAQL$S-NH.sub.2 1051 1642.85 822.43 822.46 1136 Ac-LTF3NO2$r8HYWAQL$S-NH.sub.2 1052 1642.85 822.43 822.46 1137 Ac-LTF$r82ThiYWAQL$S-NH.sub.2 1053 1613.83 807.92 807.96 1138 Ac-LTF$r8HBipWAQL$S-NH.sub.2 1054 1657.90 829.95 830.01 1139 Ac-LTF$r8HF4tBuWAQL$S-NH.sub.2 1055 1637.93 819.97 820.02 1140 Ac-LTF$r8HF4CF3WAQL$S-NH.sub.2 1056 1649.86 825.93 826.02 1141 Ac-LTF$r8HF4ClWAQL$S-NH.sub.2 1057 1615.83 808.92 809.37 1142 Ac-LTF$r8HF4MeWAQL$S-NH.sub.2 1058 1595.89 798.95 799.01 1143 Ac-LTF$r8HF4BrWAQL$S-NH.sub.2 1059 1659.78 830.89 830.98 1144 Ac-LTF$r8HF4CNWAQL$S-NH.sub.2 1060 1606.87 804.44 804.56 1145 Ac-LTF$r8HF4NO2WAQL$S-NH.sub.2 1061 1626.86 814.43 814.55 1146 Ac-LTF$r8H1NalWAQL$S-NH.sub.2 1062 1631.89 816.95 817.06 1147 Ac-LTF$r8H2NalWAQL$S-NH.sub.2 1063 1631.89 816.95 816.99 1148 Ac-LTF$r8HWAQL$S-NH.sub.2 1064 1434.80 718.40 718.49 1149 Ac-LTF$r8HY1NalAQL$S-NH.sub.2 1065 1608.87 805.44 805.52 1150 Ac-LTF$r8HY2NalAQL$S-NH.sub.2 1066 1608.87 805.44 805.52 1151 Ac-LTF$r8HYWAQI$S-NH.sub.2 1067 1597.87 799.94 800.07 1152 Ac-LTF$r8HYWAQNle$S-NH.sub.2 1068 1597.87 799.94 800.44 1153 Ac-LTF$er8HYWAQL$eA-NH.sub.2 1069 1581.87 791.94 791.98 1154 Ac-LTF$r8HYWAQL$Abu-NH.sub.2 1070 1595.89 798.95 799.03 1155 Ac-LTF$r8HYWAbuQL$S-NH.sub.2 1071 1611.88 806.94 804.47 1156 Ac-LAF$r8HYWAQL$S-NH.sub.2 1072 1567.86 784.93 785.49 1157 Ac-LTF$r8NLWANleL$Q-NH.sub.2 1073 1550.92 776.46 777.5 1158 Ac-LTF$r8ALWANleL$Q-NH.sub.2 1074 1507.92 754.96 755.52 1159 Ac-LAF$r8NLWANleL$Q-NH.sub.2 1075 1520.91 761.46 762.48 1160 Ac-F$r8AYWAAc3cL$A-NH.sub.2 1076 1256.70 629.35 1257.56 1161 Ac-LTF$r8AYWAAL$S-NH.sub.2 1077 1474.82 738.41 738.55 1162 Ac-LVF$r8AYWAQL$S-NH.sub.2 1078 1529.87 765.94 766 1163 Ac-LTF$r8AYWAbuQL$S-NH.sub.2 1079 1545.86 773.93 773.92 1164 Ac-LTF$r8AYWNleQL$S-NH.sub.2 1080 1573.89 787.95 788.17 1165 Ac-LTF$r8AbuYWAQL$S-NH.sub.2 1081 1545.86 773.93 773.99 1166 Ac-LTF$r8AYWHQL$S-NH.sub.2 1082 1597.87 799.94 799.97 1167 Ac-LTF$r8AYWKQL$S-NH.sub.2 1083 1588.90 795.45 795.53 1168 Ac-LTF$r8AYWOQL$S-NH.sub.2 1084 1574.89 788.45 788.5 1169 Ac-LTF$r8AYWRQL$S-NH.sub.2 1085 1616.91 809.46 809.51 1170 Ac-LTF$r8AYWSQL$S-NH.sub.2 1086 1547.84 774.92 774.96 1171 Ac-LTF$r8AYWRAL$S-NH.sub.2 1087 1559.89 780.95 780.95 1172 Ac-LTF$r8AYWRQL$A-NH.sub.2 1088 1600.91 801.46 801.52 1173 Ac-LTF$r8AYWRAL$A-NH.sub.2 1089 1543.89 772.95 773.03 1174 Ac-LTF$r5HYWAQL$s8S-NH.sub.2 1090 1597.87 799.94 799.97 1175 Ac-LTF$HYWAQL$r8S-NH.sub.2 1091 1597.87 799.94 799.97 1176 Ac-LTF$r8HYWAAL$S-NH.sub.2 1092 1540.84 771.42 771.48 1177 Ac-LTF$r8HYWAAbuL$S-NH.sub.2 1093 1554.86 778.43 778.51 1178 Ac-LTF$r8HYWALL$S-NH.sub.2 1094 1582.89 792.45 792.49 1179 Ac-F$r8AYWHAL$A-NH.sub.2 1095 1310.72 656.36 656.4 1180 Ac-F$r8AYWAAL$A-NH.sub.2 1096 1244.70 623.35 1245.61 1181 Ac-F$r8AYWSAL$A-NH.sub.2 1097 1260.69 631.35 1261.6 1182 Ac-F$r8AYWRAL$A-NH.sub.2 1098 1329.76 665.88 1330.72 1183 Ac-F$r8AYWKAL$A-NH.sub.2 1099 1301.75 651.88 1302.67 1184 Ac-F$r8AYWOAL$A-NH.sub.2 1100 1287.74 644.87 1289.13 1185 Ac-F$r8VYWEAc3cL$A-NH.sub.2 1101 1342.73 672.37 1343.67 1186 Ac-F$r8FYWEAc3cL$A-NH.sub.2 1102 1390.73 696.37 1392.14 1187 Ac-F$r8WYWEAc3cL$A-NH.sub.2 1103 1429.74 715.87 1431.44 1188 Ac-F$r8RYWEAc3cL$A-NH.sub.2 1104 1399.77 700.89 700.95 1189 Ac-F$r8KYWEAc3cL$A-NH.sub.2 1105 1371.76 686.88 686.97 1190 Ac-F$r8ANleWEAc3cL$A-NH.sub.2 1106 1264.72 633.36 1265.59 1191 Ac-F$r8AVWEAc3cL$A-NH.sub.2 1107 1250.71 626.36 1252.2 1192 Ac-F$r8AFWEAc3cL$A-NH.sub.2 1108 1298.71 650.36 1299.64 1193 Ac-F$r8AWWEAc3cL$A-NH.sub.2 1109 1337.72 669.86 1338.64 1194 Ac-F$r8ARWEAc3cL$A-NH.sub.2 1110 1307.74 654.87 655 1195 Ac-F$r8AKWEAc3cL$A-NH.sub.2 1111 1279.73 640.87 641.01 1196 Ac-F$r8AYWVAc3cL$A-NH.sub.2 1112 1284.73 643.37 643.38 1197 Ac-F$r8AYWFAc3cL$A-NH.sub.2 1113 1332.73 667.37 667.43 1198 Ac-F$r8AYWWAc3cL$A-NH.sub.2 1114 1371.74 686.87 686.97 1199 Ac-F$r8AYWRAc3cL$A-NH.sub.2 1115 1341.76 671.88 671.94 1200 Ac-F$r8AYWKAc3cL$A-NH.sub.2 1116 1313.75 657.88 657.88 1201 Ac-F$r8AYWEVL$A-NH.sub.2 1117 1330.73 666.37 666.47 1202 Ac-F$r8AYWEFL$A-NH.sub.2 1118 1378.73 690.37 690.44 1203 Ac-F$r8AYWEWL$A-NH.sub.2 1119 1417.74 709.87 709.91 1204 Ac-F$r8AYWERL$A-NH.sub.2 1120 1387.77 694.89 1388.66 1205 Ac-F$r8AYWEKL$A-NH.sub.2 1121 1359.76 680.88 1361.21 1206 Ac-F$r8AYWEAc3cL$V-NH.sub.2 1122 1342.73 672.37 1343.59 1207 Ac-F$r8AYWEAc3cL$F-NH.sub.2 1123 1390.73 696.37 1392.58 1208 Ac-F$r8AYWEAc3cL$W-NH.sub.2 1124 1429.74 715.87 1431.29 1209 Ac-F$r8AYWEAc3cL$R-NH.sub.2 1125 1399.77 700.89 700.95 1210 Ac-F$r8AYWEAc3cL$K-NH.sub.2 1126 1371.76 686.88 686.97 1211 Ac-F$r8AYWEAc3cL$AV-NH.sub.2 1127 1413.77 707.89 707.91 1212 Ac-F$r8AYWEAc3cL$AF-NH.sub.2 1128 1461.77 731.89 731.96 1213 Ac-F$r8AYWEAc3cL$AW-NH.sub.2 1129 1500.78 751.39 751.5 1214 Ac-F$r8AYWEAc3cL$AR-NH.sub.2 1130 1470.80 736.40 736.47 1215 Ac-F$r8AYWEAc3cL$AK-NH.sub.2 1131 1442.80 722.40 722.41 1216 Ac-F$r8AYWEAc3cL$AH-NH.sub.2 1132 1451.76 726.88 726.93 1217 Ac-LTF2NO2$r8HYWAQL$S-NH.sub.2 1133 1642.85 822.43 822.54 1218 Ac-LTA$r8HYAAQL$S-NH.sub.2 1134 1406.79 704.40 704.5 1219 Ac-LTF$r8HYAAQL$S-NH.sub.2 1135 1482.82 742.41 742.47 1220 Ac-QSQQTF$r8NLWALL$AN-NH.sub.2 1136 1966.07 984.04 984.38 1221 Ac-QAibQQTF$r8NLWALL$AN-NH.sub.2 1137 1964.09 983.05 983.42 1222 Ac-QAibQQTF$r8ALWALL$AN-NH.sub.2 1138 1921.08 961.54 961.59 1223 Ac-AAAATF$r8AAWAAL$AA-NH.sub.2 1139 1608.90 805.45 805.52 1224 Ac-F$r8AAWRAL$Q-NH.sub.2 1140 1294.76 648.38 648.48 1225 Ac-TF$r8AAWAAL$Q-NH.sub.2 1141 1310.74 656.37 1311.62 1226 Ac-TF$r8AAWRAL$A-NH.sub.2 1142 1338.78 670.39 670.46 1227 Ac-VF$r8AAWRAL$Q-NH.sub.2 1143 1393.82 697.91 697.99 1228 Ac-AF$r8AAWAAL$A-NH.sub.2 1144 1223.71 612.86 1224.67 1229 Ac-TF$r8AAWKAL$Q-NH.sub.2 1145 1367.80 684.90 684.97 1230 Ac-TF$r8AAWOAL$Q-NH.sub.2 1146 1353.78 677.89 678.01 1231 Ac-TF$r8AAWSAL$Q-NH.sub.2 1147 1326.73 664.37 664.47 1232 Ac-LTF$r8AAWRAL$Q-NH.sub.2 1148 1508.89 755.45 755.49 1233 Ac-F$r8AYWAQL$A-NH.sub.2 1149 1301.72 651.86 651.96 1234 Ac-F$r8AWWAAL$A-NH.sub.2 1150 1267.71 634.86 634.87 1235 Ac-F$r8AWWAQL$A-NH.sub.2 1151 1324.73 663.37 663.43 1236 Ac-F$r8AYWEAL$-NH.sub.2 1152 1231.66 616.83 1232.93 1237 Ac-F$r8AYWAAL$-NH.sub.2 1153 1173.66 587.83 1175.09 1238 Ac-F$r8AYWKAL$-NH.sub.2 1154 1230.72 616.36 616.44 1239 Ac-F$r8AYWOAL$-NH.sub.2 1155 1216.70 609.35 609.48 1240 Ac-F$r8AYWQAL$-NH.sub.2 1156 1230.68 616.34 616.44 1241 Ac-F$r8AYWAQL$-NH.sub.2 1157 1230.68 616.34 616.37 1242 Ac-F$r8HYWDQL$S-NH.sub.2 1158 1427.72 714.86 714.86 1243 Ac-F$r8HFWEQL$S-NH.sub.2 1159 1425.74 713.87 713.98 1244 Ac-F$r8AYWHQL$S-NH.sub.2 1160 1383.73 692.87 692.96 1245 Ac-F$r8AYWKQL$S-NH.sub.2 1161 1374.77 688.39 688.45 1246 Ac-F$r8AYWOQL$S-NH.sub.2 1162 1360.75 681.38 681.49 1247 Ac-F$r8HYWSQL$S-NH.sub.2 1163 1399.73 700.87 700.95 1248 Ac-F$r8HWWEQL$S-NH.sub.2 1164 1464.76 733.38 733.44 1249 Ac-F$r8HWWAQL$S-NH.sub.2 1165 1406.75 704.38 704.43 1250 Ac-F$r8AWWHQL$S-NH.sub.2 1166 1406.75 704.38 704.43 1251 Ac-F$r8AWWKQL$S-NH.sub.2 1167 1397.79 699.90 699.92 1252 Ac-F$r8AWWOQL$S-NH.sub.2 1168 1383.77 692.89 692.96 1253 Ac-F$r8HWWSQL$S-NH.sub.2 1169 1422.75 712.38 712.42 1254 Ac-LTF$r8NYWANleL$Q-NH.sub.2 1170 1600.90 801.45 801.52 1255 Ac-LTF$r8NLWAQL$Q-NH.sub.2 1171 1565.90 783.95 784.06 1256 Ac-LTF$r8NYWANleL$A-NH.sub.2 1172 1543.88 772.94 773.03 1257 Ac-LTF$r8NLWAQL$A-NH.sub.2 1173 1508.88 755.44 755.49 1258 Ac-LTF$r8AYWANleL$Q-NH.sub.2 1174 1557.90 779.95 780.06 1259 Ac-LTF$r8ALWAQL$Q-NH.sub.2 1175 1522.89 762.45 762.45 1260 Ac-LAF$r8NYWANleL$Q-NH.sub.2 1176 1570.89 786.45 786.5 1261 Ac-LAF$r8NLWAQL$Q-NH.sub.2 1177 1535.89 768.95 769.03 1262 Ac-LAF$r8AYWANleL$A-NH.sub.2 1178 1470.86 736.43 736.47 1263 Ac-LAF$r8ALWAQL$A-NH.sub.2 1179 1435.86 718.93 719.01 1264 Ac-LAF$r8AYWAAL$A-NH.sub.2 1180 1428.82 715.41 715.41 1265 Ac-F$r8AYWEAc3cL$AAib-NH.sub.2 1181 1399.75 700.88 700.95 1266 Ac-F$r8AYWAQL$AA-NH.sub.2 1182 1372.75 687.38 687.78 1267 Ac-F$r8AYWAAc3cL$AA-NH.sub.2 1183 1327.73 664.87 664.84 1268 Ac-F$r8AYWSAc3cL$AA-NH.sub.2 1184 1343.73 672.87 672.9 1269 Ac-F$r8AYWEAc3cL$AS-NH.sub.2 1185 1401.73 701.87 701.84 1270 Ac-F$r8AYWEAc3cL$AT-NH.sub.2 1186 1415.75 708.88 708.87 1271 Ac-F$r8AYWEAc3cL$AL-NH.sub.2 1187 1427.79 714.90 714.94 1272 Ac-F$r8AYWEAc3cL$AQ-NH.sub.2 1188 1442.76 722.38 722.41 1273 Ac-F$r8AFWEAc3cL$AA-NH.sub.2 1189 1369.74 685.87 685.93 1274 Ac-F$r8AWWEAc3cL$AA-NH.sub.2 1190 1408.75 705.38 705.39 1275 Ac-F$r8AYWEAc3cL$SA-NH.sub.2 1191 1401.73 701.87 701.99 1276 Ac-F$r8AYWEAL$AA-NH.sub.2 1192 1373.74 687.87 687.93 1277 Ac-F$r8AYWENleL$AA-NH.sub.2 1193 1415.79 708.90 708.94 1278 Ac-F$r8AYWEAc3cL$AbuA-NH.sub.2 1194 1399.75 700.88 700.95 1279 Ac-F$r8AYWEAc3cL$NleA-NH.sub.2 1195 1427.79 714.90 714.86 1280 Ac-F$r8AYWEAibL$NleA-NH.sub.2 1196 1429.80 715.90 715.97 1281 Ac-F$r8AYWEAL$NleA-NH.sub.2 1197 1415.79 708.90 708.94 1282 Ac-F$r8AYWENleL$NleA-NH.sub.2 1198 1457.83 729.92 729.96 1283 Ac-F$r8AYWEAibL$Abu-NH.sub.2 1199 1330.73 666.37 666.39 1284 Ac-F$r8AYWENleL$Abu-NH.sub.2 1200 1358.76 680.38 680.39 1285 Ac-F$r8AYWEAL$Abu-NH.sub.2 1201 1316.72 659.36 659.36 1286 Ac-LTF$r8AFWAQL$S-NH.sub.2 1202 1515.85 758.93 759.12 1287 Ac-LTF$r8AWWAQL$S-NH.sub.2 1203 1554.86 778.43 778.51 1288 Ac-LTF$r8AYWAQI$S-NH.sub.2 1204 1531.84 766.92 766.96 1289 Ac-LTF$r8AYWAQNle$S-NH.sub.2 1205 1531.84 766.92 766.96 1290 Ac-LTF$r8AYWAQL$SA-NH.sub.2 1206 1602.88 802.44 802.48 1291 Ac-LTF$r8AWWAQL$A-NH.sub.2 1207 1538.87 770.44 770.89 1292 Ac-LTF$r8AYWAQI$A-NH.sub.2 1208 1515.85 758.93 759.42 1293 Ac-LTF$r8AYWAQNle$A-NH.sub.2 1209 1515.85 758.93 759.42 1294 Ac-LTF$r8AYWAQL$AA-NH.sub.2 1210 1586.89 794.45 794.94 1295 Ac-LTF$r8HWWAQL$S-NH.sub.2 1211 1620.88 811.44 811.47 1296 Ac-LTF$r8HRWAQL$S-NH.sub.2 1212 1590.90 796.45 796.52 1297 Ac-LTF$r8HKWAQL$S-NH.sub.2 1213 1562.90 782.45 782.53 1298 Ac-LTF$r8HYWAQL$W-NH.sub.2 1214 1696.91 849.46 849.5 1299 Ac-F$r8AYWAbuAL$A-NH.sub.2 1215 1258.71 630.36 630.5 1300 Ac-F$r8AbuYWEAL$A-NH.sub.2 1216 1316.72 659.36 659.51 1301 Ac-NlePRF%r8NYWRLL%QN-NH.sub.2 1217 1954.13 978.07 978.54 1302 Ac-TSF%r8HYWAQL%S-NH.sub.2 1218 1573.83 787.92 787.98 1303 Ac-LTF%r8AYWAQL%S-NH.sub.2 1219 1533.86 767.93 768 1304 Ac-HTF$r8HYWAQL$S-NH.sub.2 1220 1621.84 811.92 811.96 1305 Ac-LHF$r8HYWAQL$S-NH.sub.2 1221 1633.88 817.94 818.02 1306 Ac-LTF$r8HHWAQL$S-NH.sub.2 1222 1571.86 786.93 786.94 1307 Ac-LTF$r8HYWHQL$S-NH.sub.2 1223 1663.89 832.95 832.38 1308 Ac-LTF$r8HYWAHL$S-NH.sub.2 1224 1606.87 804.44 804.48 1309 Ac-LTF$r8HYWAQL$H-NH.sub.2 1225 1647.89 824.95 824.98 1310 Ac-LTF$r8HYWAQL$S-NHPr 1226 1639.91 820.96 820.98 1311 Ac-LTF$r8HYWAQL$S-NHsBu 1227 1653.93 827.97 828.02 1312 Ac-LTF$r8HYWAQL$S-NHiBu 1228 1653.93 827.97 828.02 1313 Ac-LTF$r8HYWAQL$S-NHBn 1229 1687.91 844.96 844.44 1314 Ac-LTF$r8HYWAQL$S-NHPe 1230 1700.92 851.46 851.99 1315 Ac-LTF$r8HYWAQL$S-NHChx 1231 1679.94 840.97 841.04 1316 Ac-ETF$r8AYWAQL$S-NH.sub.2 1232 1547.80 774.90 774.96 1317 Ac-STF$r8AYWAQL$S-NH.sub.2 1233 1505.79 753.90 753.94 1318 Ac-LEF$r8AYWAQL$S-NH.sub.2 1234 1559.84 780.92 781.25
1319 Ac-LSF$r8AYWAQL$S-NH.sub.2 1235 1517.83 759.92 759.93 1320 Ac-LTF$r8EYWAQL$S-NH.sub.2 1236 1589.85 795.93 795.97 1321 Ac-LTF$r8SYWAQL$S-NH.sub.2 1237 1547.84 774.92 774.96 1322 Ac-LTF$r8AYWEQL$S-NH.sub.2 1238 1589.85 795.93 795.9 1323 Ac-LTF$r8AYWAEL$S-NH.sub.2 1239 1532.83 767.42 766.96 1324 Ac-LTF$r8AYWASL$S-NH.sub.2 1240 1490.82 746.41 746.46 1325 Ac-LTF$r8AYWAQL$E-NH.sub.2 1241 1573.85 787.93 787.98 1326 Ac-LTF2CN$r8HYWAQL$S-NH.sub.2 1242 1622.86 812.43 812.47 1327 Ac-LTF3Cl$r8HYWAQL$S-NH.sub.2 1243 1631.83 816.92 816.99 1328 Ac-LTDip$r8HYWAQL$S-NH.sub.2 1244 1673.90 837.95 838.01 1329 Ac-LTF$r8HYWAQTle$S-NH.sub.2 1245 1597.87 799.94 800.04 1330 Ac-F$r8AY6clWEAL$A-NH.sub.2 1246 1336.66 669.33 1338.56 1331 Ac-F$r8AYdl6brWEAL$A-NH.sub.2 1247 1380.61 691.31 692.2 1332 Ac-F$r8AYdl6fWEAL$A-NH.sub.2 1248 1320.69 661.35 1321.61 1333 Ac-F$r8AYdl4mWEAL$A-NH.sub.2 1249 1316.72 659.36 659.36 1334 Ac-F$r8AYdl5clWEAL$A-NH.sub.2 1250 1336.66 669.33 669.35 1335 Ac-F$r8AYdl7mWEAL$A-NH.sub.2 1251 1316.72 659.36 659.36 1336 Ac-LTF%r8HYWAQL%A-NH.sub.2 1252 1583.89 792.95 793.01 1337 Ac-LTF$r8HCouWAQL$S-NH.sub.2 1253 1679.87 840.94 841.38 1338 Ac-LTFEHCouWAQLTS-NH.sub.2 1254 1617.75 809.88 809.96 1339 Ac-LTA$r8HCouWAQL$S-NH.sub.2 1255 1603.84 802.92 803.36 1340 Ac-F$r8AYWEAL$AbuA-NH.sub.2 1256 1387.75 694.88 694.88 1341 Ac-F$r8AYWEAI$AA-NH.sub.2 1257 1373.74 687.87 687.93 1342 Ac-F$r8AYWEANle$AA-NH.sub.2 1258 1373.74 687.87 687.93 1343 Ac-F$r8AYWEAmlL$AA-NH.sub.2 1259 1429.80 715.90 715.97 1344 Ac-F$r8AYWQAL$AA-NH.sub.2 1260 1372.75 687.38 687.48 1345 Ac-F$r8AYWAAL$AA-NH.sub.2 1261 1315.73 658.87 658.92 1346 Ac-F$r8AYWAbuAL$AA-NH.sub.2 1262 1329.75 665.88 665.95 1347 Ac-F$r8AYWNleAL$AA-NH.sub.2 1263 1357.78 679.89 679.94 1348 Ac-F$r8AbuYWEAL$AA-NH.sub.2 1264 1387.75 694.88 694.96 1349 Ac-F$r8NleYWEAL$AA-NH.sub.2 1265 1415.79 708.90 708.94 1350 Ac-F$r8FYWEAL$AA-NH.sub.2 1266 1449.77 725.89 725.97 1351 Ac-LTF$r8HYWAQhL$S-NH.sub.2 1267 1611.88 806.94 807 1352 Ac-LTF$r8HYWAQAdm$S-NH.sub.2 1268 1675.91 838.96 839.04 1353 Ac-LTF$r8HYWAQIgl$S-NH.sub.2 1269 1659.88 830.94 829.94 1354 Ac-F$r8AYWAQL$AA-NH.sub.2 1270 1372.75 687.38 687.48 1355 Ac-LTF$r8ALWAQL$Q-NH.sub.2 1271 1522.89 762.45 762.52 1356 Ac-F$r8AYWEAL$AA-NH.sub.2 1272 1373.74 687.87 687.93 1357 Ac-F$r8AYWENleL$AA-NH.sub.2 1273 1415.79 708.90 708.94 1358 Ac-F$r8AYWEAibL$Abu-NH.sub.2 1274 1330.73 666.37 666.39 1359 Ac-F$r8AYWENleL$Abu-NH.sub.2 1275 1358.76 680.38 680.38 1360 Ac-F$r8AYWEAL$Abu-NH.sub.2 1276 1316.72 659.36 659.36 1361 Ac-F$r8AYWEAc3cL$AbuA-NH.sub.2 1277 1399.75 700.88 700.95 1362 Ac-F$r8AYWEAc3cL$NleA-NH.sub.2 1278 1427.79 714.90 715.01 1363 H-LTF$r8AYWAQL$S-NH.sub.2 1279 1489.83 745.92 745.95 1364 mdPEG3-LTF$r8AYWAQL$S-NH.sub.2 1280 1679.92 840.96 840.97 1365 mdPEG7-LTF$r8AYWAQL$S-NH.sub.2 1281 1856.02 929.01 929.03 1366 Ac-F$r8ApmpEt6clWEAL$A-NH.sub.2 1282 1470.71 736.36 788.17 1367 Ac-LTF3Cl$r8AYWAQL$S-NH.sub.2 1283 1565.81 783.91 809.18 1368 Ac-LTF3Cl$r8HYWAQL$A-NH.sub.2 1284 1615.83 808.92 875.24 1369 Ac-LTF3Cl$r8HYWWQL$S-NH.sub.2 1285 1746.87 874.44 841.65 1370 Ac-LTF3Cl$r8AYWWQL$S-NH.sub.2 1286 1680.85 841.43 824.63 1371 Ac-LTF$r8AYWWQL$S-NH.sub.2 1287 1646.89 824.45 849.98 1372 Ac-LTF$r8HYWWQL$A-NH.sub.2 1288 1696.91 849.46 816.67 1373 Ac-LTF$r8AYWWQL$A-NH.sub.2 1289 1630.89 816.45 776.15 1374 Ac-LTF4F$r8AYWAQL$S-NH.sub.2 1290 1549.83 775.92 776.15 1375 Ac-LTF2F$r8AYWAQL$S-NH.sub.2 1291 1549.83 775.92 776.15 1376 Ac-LTF3F$r8AYWAQL$S-NH.sub.2 1292 1549.83 775.92 785.12 1377 Ac-LTF34F2$r8AYWAQL$S-NH.sub.2 1293 1567.83 784.92 785.12 1378 Ac-LTF35F2$r8AYWAQL$S-NH.sub.2 1294 1567.83 784.92 1338.74 1379 Ac-F3Cl$r8AYWEAL$A-NH.sub.2 1295 1336.66 669.33 705.28 1380 Ac-F3Cl$r8AYWEAL$AA-NH.sub.2 1296 1407.70 704.85 680.11 1381 Ac-F$r8AY6clWEAL$AA-NH.sub.2 1297 1407.70 704.85 736.83 1382 Ac-F$r8AY6clWEAL$-NH.sub.2 1298 1265.63 633.82 784.1 1383 Ac-LTF$r8HYWAQLSt/S-NH.sub.2 1299 16.03 9.02 826.98 1384 Ac-LTF$r8HYWAQL$S-NHsBu 1300 1653.93 827.97 828.02 1385 Ac-STF$r8AYWAQL$S-NH.sub.2 1301 1505.79 753.90 753.94 1386 Ac-LTF$r8AYWAEL$S-NH.sub.2 1302 1532.83 767.42 767.41 1387 Ac-LTF$r8AYWAQL$E-NH.sub.2 1303 1573.85 787.93 787.98 1388 mdPEG3-LTF$r8AYWAQL$S-NH.sub.2 1304 1679.92 840.96 840.97 1389 Ac-LTF$r8AYWAQhL$S-NH.sub.2 1305 1545.86 773.93 774.31 1390 Ac-LTF$r8AYWAQCha$S-NH.sub.2 1306 1571.88 786.94 787.3 1391 Ac-LTF$r8AYWAQChg$S-NH.sub.2 1307 1557.86 779.93 780.4 1392 Ac-LTF$r8AYWAQCba$S-NH.sub.2 1308 1543.84 772.92 780.13 1393 Ac-LTF$r8AYWAQF$S-NH.sub.2 1309 1565.83 783.92 784.2 1394 Ac-LTF4F$r8HYWAQhL$S-NH.sub.2 1310 1629.87 815.94 815.36 1395 Ac-LTF4F$r8HYWAQCha$S-NH.sub.2 1311 1655.89 828.95 828.39 1396 Ac-LTF4F$r8HYWAQChg$S-NH.sub.2 1312 1641.87 821.94 821.35 1397 Ac-LTF4F$r8HYWAQCba$S-NH.sub.2 1313 1627.86 814.93 814.32 1398 Ac-LTF4F$r8AYWAQhL$S-NH.sub.2 1314 1563.85 782.93 782.36 1399 Ac-LTF4F$r8AYWAQCha$S-NH.sub.2 1315 1589.87 795.94 795.38 1400 Ac-LTF4F$r8AYWAQChg$S-NH.sub.2 1316 1575.85 788.93 788.35 1401 Ac-LTF4F$r8AYWAQCba$S-NH.sub.2 1317 1561.83 781.92 781.39 1402 Ac-LTF3Cl$r8AYWAQhL$S-NH.sub.2 1318 1579.82 790.91 790.35 1403 Ac-LTF3Cl$r8AYWAQCha$S-NH.sub.2 1319 1605.84 803.92 803.67 1404 Ac-LTF3Cl$r8AYWAQChg$S-NH.sub.2 1320 1591.82 796.91 796.34 1405 Ac-LTF3Cl$r8AYWAQCba$S-NH.sub.2 1321 1577.81 789.91 789.39 1406 Ac-LTF$r8AYWAQhF$S-NH.sub.2 1322 1579.84 790.92 791.14 1407 Ac-LTF$r8AYWAQF3CF3$S-NH.sub.2 1323 1633.82 817.91 818.15 1408 Ac-LTF$r8AYWAQF3Me$S-NH.sub.2 1324 1581.86 791.93 791.32 1409 Ac-LTF$r8AYWAQ1Nal$S-NH.sub.2 1325 1615.84 808.92 809.18 1413 Ac-LTF$r8AYWAQBip$S-NH.sub.2 1326 1641.86 821.93 822.13 1414 Ac-LTF$r8FYWAQL$A-NH.sub.2 1327 1591.88 796.94 797.33 1415 Ac-LTF$r8HYWAQL$S-NHAm 1328 1667.94 834.97 835.92 1416 Ac-LTF$r8HYWAQL$S-NHiAm 1329 1667.94 834.97 835.55 1417 Ac-LTF$r8HYWAQL$S-NHnPr3Ph 1330 1715.94 858.97 859.79 1418 Ac-LTF$r8HYWAQL$S-NHnBu3,3Me 1331 1681.96 841.98 842.49 1419 Ac-LTF$r8HYWAQL$S-NHnPr 1332 1639.91 820.96 821.58 1420 Ac-LTF$r8HYWAQL$S-NHnEt2Ch 1333 1707.98 854.99 855.35 1421 Ac-LTF$r8HYWAQL$S-NHHex 1334 1681.96 841.98 842.4 1422 Ac-LTF$r8AYWAQL$S-NHmdPeg2 1335 1633.91 817.96 818.35 1423 Ac-LTF$r8AYWAQL$A-NHmdPeg2 1336 1617.92 809.96 810.3 1424 Ac-LTF$r8AYWAQL$A-NHmdPeg4 1337 1705.97 853.99 854.33 1425 Ac-F$r8AYdl4mWEAL$A-NH.sub.2 1338 1316.72 659.36 659.44 1426 Ac-F$r8AYdl5clWEAL$A-NH.sub.2 1339 1336.66 669.33 669.43 1427 Ac-LThF$r8AYWAQL$S-NH.sub.2 1340 1545.86 773.93 774.11 1428 Ac-LT2Nal$r8AYWAQL$S-NH.sub.2 1341 1581.86 791.93 792.43 1429 Ac-LTA$r8AYWAQL$S-NH.sub.2 1342 1455.81 728.91 729.15 1430 Ac-LTF$r8AYWVQL$S-NH.sub.2 1343 1559.88 780.94 781.24 1431 Ac-LTF$r8HYWAAL$A-NH.sub.2 1344 1524.85 763.43 763.86 1432 Ac-LTF$r8VYWAQL$A-NH.sub.2 1345 1543.88 772.94 773.37 1433 Ac-LTF$r8IYWAQL$S-NH.sub.2 1346 1573.89 787.95 788.17 1434 Ac-FTF$r8VYWSQL$S-NH.sub.2 1347 1609.85 805.93 806.22 1435 Ac-ITF$r8FYWAQL$S-NH.sub.2 1348 1607.88 804.94 805.2 1436 Ac-2NalTF$r8VYWSQL$S-NH.sub.2 1349 1659.87 830.94 831.2 1437 Ac-ITF$r8LYWSQL$S-NH.sub.2 1350 1589.89 795.95 796.13 1438 Ac-FTF$r8FYWAQL$S-NH.sub.2 1351 1641.86 821.93 822.13 1439 Ac-WTF$r8VYWAQL$S-NH.sub.2 1352 1632.87 817.44 817.69 1440 Ac-WTF$r8WYWAQL$S-NH.sub.2 1353 1719.88 860.94 861.36 1441 Ac-VTF$r8AYWSQL$S-NH.sub.2 1354 1533.82 767.91 768.19 1442 Ac-WTF$r8FYWSQL$S-NH.sub.2 1355 1696.87 849.44 849.7 1443 Ac-FTF$r8IYWAQL$S-NH.sub.2 1356 1607.88 804.94 805.2 1444 Ac-WTF$r8VYWSQL$S-NH.sub.2 1357 1648.87 825.44 824.8 1445 Ac-FTF$r8LYWSQL$S-NH.sub.2 1358 1623.87 812.94 812.8 1446 Ac-YTF$r8FYWSQL$S-NH.sub.2 1359 1673.85 837.93 837.8 1447 Ac-LTF$r8AY6clWEAL$A-NH.sub.2 1360 1550.79 776.40 776.14 1448 Ac-LTF$r8AY6clWSQL$S-NH.sub.2 1361 1581.80 791.90 791.68 1449 Ac-F$r8AY6clWSAL$A-NH.sub.2 1362 1294.65 648.33 647.67 1450 Ac-F$r8AY6clWQAL$AA-NH.sub.2 1363 1406.72 704.36 703.84 1451 Ac-LHF$r8AYWAQL$S-NH.sub.2 1364 1567.86 784.93 785.21 1452 Ac-LTF$r8AYWAQL$S-NH.sub.2 1365 1531.84 766.92 767.17 1453 Ac-LTF$r8AHWAQL$S-NH.sub.2 1366 1505.84 753.92 754.13 1454 Ac-LTF$r8AYWAHL$S-NH.sub.2 1367 1540.84 771.42 771.61 1455 Ac-LTF$r8AYWAQL$H-NH.sub.2 1368 1581.87 791.94 792.15 1456 H-LTF$r8AYWAQL$A-NH.sub.2 1369 1473.84 737.92 737.29 1457 Ac-HHF$r8AYWAQL$S-NH.sub.2 1370 1591.83 796.92 797.35 1458 Ac-aAibWTF$r8VYWSQL$S-NH.sub.2 1371 1804.96 903.48 903.64 1459 Ac-AibWTF$r8HYWAQL$S-NH.sub.2 1372 1755.91 878.96 879.4 1460 Ac-AibAWTF$r8HYWAQL$S-NH.sub.2 1373 1826.95 914.48 914.7 1461 Ac-fWTF$r8HYWAQL$S-NH.sub.2 1374 1817.93 909.97 910.1 1462 Ac-AibWWTF$r8HYWAQL$S-NH.sub.2 1375 1941.99 972.00 972.2 1463 Ac-WTF$r8LYWSQL$S-NH.sub.2 1376 1662.88 832.44 832.8 1464 Ac-WTF$r8NleYWSQL$S-NH.sub.2 1377 1662.88 832.44 832.6 1465 Ac-LTF$r8AYWSQL$a-NH.sub.2 1378 1531.84 766.92 767.2 1466 Ac-LTF$r8EYWARL$A-NH.sub.2 1379 1601.90 801.95 802.1 1467 Ac-LTF$r8EYWAHL$A-NH.sub.2 1380 1582.86 792.43 792.6 1468 Ac-aTF$r8AYWAQL$S-NH.sub.2 1381 1489.80 745.90 746.08 1469 Ac-AibTF$r8AYWAQL$S-NH.sub.2 1382 1503.81 752.91 753.11 1470 Ac-AmfTF$r8AYWAQL$S-NH.sub.2 1383 1579.84 790.92 791.14 1471 Ac-AmwTF$r8AYWAQL$S-NH.sub.2 1384 1618.86 810.43 810.66 1472 Ac-NmLTF$r8AYWAQL$S-NH.sub.2 1385 1545.86 773.93 774.11 1473 Ac-LNmTF$r8AYWAQL$S-NH.sub.2 1386 1545.86 773.93 774.11 1474 Ac-LSarF$r8AYWAQL$S-NH.sub.2 1387 1501.83 751.92 752.18 1475 Ac-LGF$r8AYWAQL$S-NH.sub.2 1388 1487.82 744.91 745.15 1476 Ac-LTNmF$r8AYWAQL$S-NH.sub.2 1389 1545.86 773.93 774.2 1477 Ac-TF$r8AYWAQL$S-NH.sub.2 1390 1418.76 710.38 710.64 1478 Ac-ETF$r8AYWAQL$A-NH.sub.2 1391 1531.81 766.91 767.2 1479 Ac-LTF$r8EYWAQL$A-NH.sub.2 1392 1573.85 787.93 788.1 1480 Ac-LT2Nal$r8AYWSQL$S-NH.sub.2 1393 1597.85 799.93 800.4 1481 Ac-LTF$r8AYWAAL$S-NH.sub.2 1394 1474.82 738.41 738.68 1482 Ac-LTF$r8AYWAQhCha$S-NH.sub.2 1395 1585.89 793.95 794.19 1483 Ac-LTF$r8AYWAQChg$S-NH.sub.2 1396 1557.86 779.93 780.97 1484 Ac-LTF$r8AYWAQCba$S-NH.sub.2 1397 1543.84 772.92 773.19 1485 Ac-LTF$r8AYWAQF3CF3$S-NH.sub.2 1398 1633.82 817.91 818.15 1486 Ac-LTF$r8AYWAQ1Nal$S-NH.sub.2 1399 1615.84 808.92 809.18 1487 Ac-LTF$r8AYWAQBip$S-NH.sub.2 1400 1641.86 821.93 822.32 1488 Ac-LT2Nal$r8AYWAQL$S-NH.sub.2 1401 1581.86 791.93 792.15 1489 Ac-LTF$r8AYWVQL$S-NH.sub.2 1402 1559.88 780.94 781.62 1490 Ac-LTF$r8AWWAQL$S-NH.sub.2 1403 1554.86 778.43 778.65 1491 Ac-FTF$r8VYWSQL$S-NH.sub.2 1404 1609.85 805.93 806.12 1492 Ac-ITF$r8FYWAQL$S-NH.sub.2 1405 1607.88 804.94 805.2 1493 Ac-ITF$r8LYWSQL$S-NH.sub.2 1406 1589.89 795.95 796.22 1494 Ac-FTF$r8FYWAQL$S-NH.sub.2 1407 1641.86 821.93 822.41 1495 Ac-VTF$r8AYWSQL$S-NH.sub.2 1408 1533.82 767.91 768.19 1496 Ac-LTF$r8AHWAQL$S-NH.sub.2 1409 1505.84 753.92 754.31 1497 Ac-LTF$r8AYWAQL$H-NH.sub.2 1410 1581.87 791.94 791.94 1498 Ac-LTF$r8AYWAHL$S-NH.sub.2 1411 1540.84 771.42 771.61 1499 Ac-aAibWTF$r8VYWSQL$S-NH.sub.2 1412 1804.96 903.48 903.9 1500 Ac-AibWTF$r8HYWAQL$S-NH.sub.2 1413 1755.91 878.96 879.5 1501 Ac-AibAWTF$r8HYWAQL$S-NH.sub.2 1414 1826.95 914.48 914.7 1502 Ac-fWTF$r8HYWAQL$S-NH.sub.2 1415 1817.93 909.97 910.2 1503 Ac-AibWWTF$r8HYWAQL$S-NH.sub.2 1416 1941.99 972.00 972.7 1504 Ac-WTF$r8LYWSQL$S-NH.sub.2 1417 1662.88 832.44 832.7 1505 Ac-WTF$r8NleYWSQL$S-NH.sub.2 1418 1662.88 832.44 832.7 1506 Ac-LTF$r8AYWSQL$a-NH.sub.2 1419 1531.84 766.92 767.2 1507 Ac-LTF$r8EYWARL$A-NH.sub.2 1420 1601.90 801.95 802.2 1508 Ac-LTF$r8EYWAHL$A-NH.sub.2 1421 1582.86 792.43 792.6 1509 Ac-aTF$r8AYWAQL$S-NH.sub.2 1422 1489.80 745.90 746.1 1510 Ac-AibTF$r8AYWAQL$S-NH.sub.2 1423 1503.81 752.91 753.2 1513 Ac-AmfTF$r8AYWAQL$S-NH.sub.2 1424 1579.84 790.92 791.2 1514 Ac-AmwTF$r8AYWAQL$S-NH.sub.2 1425 1618.86 810.43 810.7 1515 Ac-NmLTF$r8AYWAQL$S-NH.sub.2 1426 1545.86 773.93 774.1 1516 Ac-LNmTF$r8AYWAQL$S-NH.sub.2 1427 1545.86 773.93 774.4 1517 Ac-LSarF$r8AYWAQL$S-NH.sub.2 1428 1501.83 751.92 752.1 1518 Ac-TF$r8AYWAQL$S-NH.sub.2 1429 1418.76 710.38 710.8 1519 Ac-ETF$r8AYWAQL$A-NH.sub.2 1430 1531.81 766.91 767.4 1520 Ac-LTF$r8EYWAQL$A-NH.sub.2 1431 1573.85 787.93 788.2 1521 Ac-WTF$r8VYWSQL$S-NH.sub.2 1432 1648.87 825.44 825.2 1522 Ac-YTF$r8FYWSQL$S-NH.sub.2 1433 1673.85 837.93 837.3 1523 Ac-F$r8AY6clWSAL$A-NH.sub.2 1434 1294.65 648.33 647.74 1524 Ac-ETF$r8EYWVQL$S-NH.sub.2 1435 1633.84 817.92 817.36 1525 Ac-ETF$r8EHWAQL$A-NH.sub.2 1436 1563.81 782.91 782.36 1526 Ac-ITF$r8EYWAQL$S-NH.sub.2 1437 1589.85 795.93 795.38 1527 Ac-ITF$r8EHWVQL$A-NH.sub.2 1438 1575.88 788.94 788.42 1528 Ac-ITF$r8EHWAQL$S-NH.sub.2 1439 1563.85 782.93 782.43 1529 Ac-LTF4F$r8AYWAQCba$S-NH.sub.2 1440 1561.83 781.92 781.32 1530 Ac-LTF3Cl$r8AYWAQhL$S-NH.sub.2 1441 1579.82 790.91 790.64 1531 Ac-LTF3Cl$r8AYWAQCha$S-NH.sub.2 1442 1605.84 803.92 803.37 1532 Ac-LTF3Cl$r8AYWAQChg$S-NH.sub.2 1443 1591.82 796.91 796.27 1533 Ac-LTF3Cl$r8AYWAQCba$S-NH.sub.2 1444 1577.81 789.91 789.83 1534 Ac-LTF$r8AY6clWSQL$S-NH.sub.2 1445 1581.80 791.90 791.75 1535 Ac-LTF4F$r8HYWAQhL$S-NH.sub.2 1446 1629.87 815.94 815.36 1536 Ac-LTF4F$r8HYWAQCba$S-NH.sub.2 1447 1627.86 814.93 814.32 1537 Ac-LTF4F$r8AYWAQhL$S-NH.sub.2 1448 1563.85 782.93 782.36 1538 Ac-LTF4F$r8AYWAQChg$S-NH.sub.2 1449 1575.85 788.93 788.35 1539 Ac-ETF$r8EYWVAL$S-NH.sub.2 1450 1576.82 789.41 788.79 1540 Ac-ETF$r8EHWAAL$A-NH.sub.2 1451 1506.79 754.40 754.8 1541 Ac-ITF$r8EYWAAL$S-NH.sub.2 1452 1532.83 767.42 767.75 1542 Ac-ITF$r8EHWVAL$A-NH.sub.2 1453 1518.86 760.43 760.81 1543 Ac-ITF$r8EHWAAL$S-NH.sub.2 1454 1506.82 754.41 754.8 1544 Pam-LTF$r8EYWAQL$S-NH.sub.2 1455 1786.07 894.04 894.48 1545 Pam-ETF$r8EYWAQL$S-NH.sub.2 1456 1802.03 902.02 902.34 1546 Ac-LTF$r8AYWLQL$S-NH.sub.2 1457 1573.89 787.95 787.39 1547 Ac-LTF$r8EYWLQL$S-NH.sub.2 1458 1631.90 816.95 817.33 1548 Ac-LTF$r8EHWLQL$S-NH.sub.2 1459 1605.89 803.95 804.29 1549 Ac-LTF$r8VYWAQL$S-NH.sub.2 1460 1559.88 780.94 781.34 1550 Ac-LTF$r8AYWSQL$S-NH.sub.2 1461 1547.84 774.92 775.33 1551 Ac-ETF$r8AYWAQL$S-NH.sub.2 1462 1547.80 774.90 775.7 1552 Ac-LTF$r8EYWAQL$S-NH.sub.2 1463 1589.85 795.93 796.33 1553 Ac-LTF$r8HYWAQL$S-NHAm 1464 1667.94 834.97 835.37 1554 Ac-LTF$r8HYWAQL$S-NHiAm 1465 1667.94 834.97 835.27 1555 Ac-LTF$r8HYWAQL$S-NHnPr3Ph 1466 1715.94 858.97 859.42 1556 Ac-LTF$r8HYWAQL$S-NHnBu3,3Me 1467 1681.96 841.98 842.67 1557 Ac-LTF$r8HYWAQL$S-NHnBu 1468 1653.93 827.97 828.24 1558 Ac-LTF$r8HYWAQL$S-NHnPr 1469 1639.91 820.96 821.31 1559 Ac-LTF$r8HYWAQL$S-NHnEt2Ch 1470 1707.98 854.99 855.35 1560 Ac-LTF$r8HYWAQL$S-NHHex 1471 1681.96 841.98 842.4 1561 Ac-LTF$r8AYWAQL$S-NHmdPeg2 1472 1633.91 817.96 855.35 1562 Ac-LTF$r8AYWAQL$A-NHmdPeg2 1473 1617.92 809.96 810.58 1563 Ac-LTF$r5AYWAAL$s8S-NH.sub.2 1474 1474.82 738.41 738.79 1564 Ac-LTF$r8AYWCouQL$S-NH.sub.2 1475 1705.88 853.94 854.61 1565 Ac-LTF$r8CouYWAQL$S-NH.sub.2 1476 1705.88 853.94 854.7 1566 Ac-CouTF$r8AYWAQL$S-NH.sub.2 1477 1663.83 832.92 833.33 1567 H-LTF$r8AYWAQL$A-NH.sub.2 1478 1473.84 737.92 737.29 1568 Ac-HHF$r8AYWAQL$S-NH.sub.2 1479 1591.83 796.92 797.72 1569 Ac-LT2Nal$r8AYWSQL$S-NH.sub.2 1480 1597.85 799.93 800.68 1570 Ac-LTF$r8HCouWAQL$S-NH.sub.2 1481 1679.87 840.94 841.38 1571 Ac-LTF$r8AYWCou2QL$S-NH.sub.2 1482 1789.94 895.97 896.51 1572 Ac-LTF$r8Cou2YWAQL$S-NH.sub.2 1483 1789.94 895.97 896.5 1573 Ac-Cou2TF$r8AYWAQL$S-NH.sub.2 1484 1747.90 874.95 875.42 1574 Ac-LTF$r8ACou2WAQL$S-NH.sub.2 1485 1697.92 849.96 850.82
1575 Dmaac-LTF$r8AYWAQL$S-NH.sub.2 1486 1574.89 788.45 788.82 1576 Hexac-LTF$r8AYWAQL$S-NH.sub.2 1487 1587.91 794.96 795.11 1577 Napac-LTF$r8AYWAQL$S-NH.sub.2 1488 1657.89 829.95 830.36 1578 Pam-LTF$r8AYWAQL$S-NH.sub.2 1489 1728.06 865.03 865.45 1579 Ac-LT2Nal$r8HYAAQL$S-NH.sub.2 1490 1532.84 767.42 767.61 1580 Ac-LT2Nal$/r8HYWAQL$/S-NH.sub.2 1491 1675.91 838.96 839.1 1581 Ac-LT2Nal$r8HYFAQL$S-NH.sub.2 1492 1608.87 805.44 805.9 1582 Ac-LT2Nal$r8HWAAQL$S-NH.sub.2 1493 1555.86 778.93 779.08 1583 Ac-LT2Nal$r8HYAWQL$S-NH.sub.2 1494 1647.88 824.94 825.04 1584 Ac-LT2Nal$r8HYAAQW$S-NH.sub.2 1495 1605.83 803.92 804.05 1585 Ac-LTW$r8HYWAQL$S-NH.sub.2 1496 1636.88 819.44 819.95 1586 Ac-LT1Nal$r8HYWAQL$S-NH.sub.2 1497 1647.88 824.94 825.41
[0366] Observed mass was measured by electrospray ionization-mass spectrometry.
[0367] In some embodiments, a peptidomimetic macrocycles disclosed herein do not comprise a peptidomimetic macrocycle structure as shown in Table 4b.
[0368] Table 4c shows examples of non-crosslinked polypeptides comprising D-amino acids.
TABLE-US-00011 TABLE 4c SEQ ID Exact Found Calc Calc Calc SP Sequence NO: Isomer Mass Mass (M + 1)/1 (M + 2)/2 (M + 3)/3 1587 Ac-tawyanfekllr-NH.sub.2 1498 777.46 1588 Ac-tawyanf4CF3ekllr-NH.sub.2 1499 811.41
Example 2: Cell Viability Assay
[0369] Cells were thawed from a liquid nitrogen preserved state. Once cells were expanded and divide at their expected doubling times, screening began. Cells were seeded in growth media in black 384-well tissue culture treated plates at 500 cells/well. Cells were equilibrated in assay plates via centrifugation and placed in incubators attached to the Dosing Modules at 37.degree. C. for 24 hr before treatment, resulting in cell densities of approximately 500 cells/plate. At the time of treatment, a set of assay plates (which did not receive treatment) were collected and ATP levels were measured by adding ATPLite (Perkin Elmer). These T-zero (To) plates were read using ultra-sensitive luminescence on Envision Plate Readers. Using an automated acoustic dispensing system, assay plates were treated with compound or peptide from a 1000.times.DMSO stock, achieving a 1:1000 working dilution. Final treatment concentrations in the plates were 0 (vehicle), 0.001, 0.003, 0.01, 0.03, 0.1, 0.3, 1, 3, 10 and 30 M. The assay plates (4 replicates per treatment) were incubated with compound or peptide for 72 hr. After 72 hr, plates were developed for endpoint analysis using ATPLite. All data points were collected via automated processes; quality controlled; and analyzed using Zalicus proprietary software. Assay plates were accepted if they passed the following quality control standards: relative luciferase values were consistent throughout the entire experiment, Z-factor scores were greater than 0.6, and untreated/vehicle controls behaved consistently on the plate.
[0370] Growth Inhibition (GI), used as a measure of cell viability, was measured at the time of dosing (To) and after 72 hr (T.sub.72). A GI reading of 0% represents no growth inhibition; a GI 100% represents complete growth inhibition, a cytostatic effect. A GI 200% represents complete death of all cells in the culture well. Compounds reaching an activity plateau of GI 200% were considered cytotoxic. GI was calculated by the following test and equation:
If T < V 0 : 100 * ( 1 - T - V 0 V 0 ) ##EQU00001## If T .gtoreq. V 0 : 100 * ( 1 - T - V 0 V - V 0 ) ##EQU00001.2##
[0371] Where T is the signal measure for a test article, V is the vehicle-treated control measure, and Vo is the vehicle control measure at time zero. This formula was derived from the Growth Inhibition calculation used in the National Cancer Institute's NCI-60 high throughput screen
[0372] Cell lines were assigned as p53 wild-type, mutant, or null from information available from the Cancer Cell Line Encyclopedia. Results for exemplary p53 peptidomimetic macrocycles are shown in Table 5 below
TABLE-US-00012 TABLE 5 Cell Viability in p53.sup.WT/p53.sup.MUT cancer cell lines SP-154 SP-763 EC.sub.50 EC.sub.50 Cell lines Cancer Type p53.sup.WT/p53.sup.MUT (.mu.M) (.mu.M) A2058 skin p53.sup.MUT 18.6 30 AsPC-1 Pancreas p53.sup.MUT 30 30 Caov-3 ovary p53.sup.MUT 12.9 30 Caov-4 ovary p53.sup.MUT 30 30 COLO-679 skin p53.sup.MUT 13.5 30 COLO-684 endometrium p53.sup.MUT 10.1 30 COLO-741 skin p53.sup.MUT 18.5 30 EBC-1 Lung-NSCLC p53.sup.MUT 30 30 ECC10 gastric p53.sup.MUT 15.2 30 KM12 colorectal p53.sup.MUT 30 30 LS-123 colorectal p53.sup.MUT 30 30 MIA PaCa-2 pancreas p53.sup.MUT 30 30 NCI-H508 colorectal p53.sup.MUT 30 30 OVCAR-3 ovary p53.sup.MUT 30 30 RPMI-7951 skin p53.sup.MUT 10.8 30 SF126 brain p53.sup.MUT 11 30 SK-OV-3 ovary p53.sup.MUT 14.3 30 SW480 colorectal p53.sup.MUT 30 30 HCT-116 colorectal p53.sup.WT 0.464 1.02 HEC-151 endometrium p53.sup.WT 1.19 30 HEC-265 endometrium p53.sup.WT 1.78 30 HUH-6-clone5 liver p53.sup.WT 0.865 0.791 IST-MES1 lung p53.sup.WT 1.43 30 KP-N-RT-BM-1 nerve p53.sup.WT 0.285 0.255 KP-N-S19s nerve p53.sup.WT 0.176 0.0168 LoVo colorectal p53.sup.WT 0.431 0.134 LS-174T colorectal p53.sup.WT 0.402 0.205 MSTO-211H lung p53.sup.WT 0.288 0.209 MV-4-11 AML p53.sup.WT 0.159 0.307 NCI-H929 multiple myeloma p53.sup.WT 0.858 0.24 PA-1 ovary p53.sup.WT 0.339 0.592 WM-115 skin p53.sup.WT 0.425 0.429 COLO-205 colorectal p53.sup.WT 0.646 0.429 COLO-849 skin p53.sup.WT 1.66 0.501 NCI-H28 lung p53.sup.WT 3.42 30
Example 3: Safety and/or Tolerability Study--I
Study Objectives
[0373] This study was designed to (i) evaluate the safety and/or tolerability of Aileron peptide 1, and to (ii) determine the DLTs and the MTD of the Aileron peptide 1 in patients with advanced solid tumors, including patients with tumors expressing WT p53 protein. Aileron peptide 1 is an alpha helical hydrocarbon cross-linked polypeptide macrocycle, with an amino acid sequence less than 20 amino acids long that is derived from the transactivation domain of wild type human P53 protein and that contains a phenylalanine, a tryptophan and a leucine amino acid in the same positions relative to each other as in the transactivation domain of wild type human P53 protein. Aileron peptide 1 has a single cross link spanning amino acids in the i to the i+7 position of the amino acid sequence and has more than three amino acids between the i+7 position and the carboxyl terminus. Aileron peptide 1 binds to human MDM2 and MDM4 and has an observed mass of 950-975 m/e as measured by electrospray ionization-mass spectrometry.
Investigational Plan
Study Design
[0374] The study consisted of a Dose Escalation Phase (DEP) and an Expansion Phase (EXP). The DEP was a "3+3" dose escalation design to establish the MTD of Aileron peptide-1. The EXP enrolled patients with specific solid tumors at the MTD to further investigate the clinical safety profile and potential efficacy of the dose level. The selection of patients for the EXP is finalized based on results of the DEP, as well as data from additional nonclinical pharmacology studies. The later includes the investigation of multiple solid cancer cell lines (e.g., breast, bladder, head/neck, gastrointestinal, liver, lung, pancreas, prostate, sarcoma) that facilitates the comparison of cell line sensitivity to Aileron peptide-1 across and within tumor types.
[0375] After completing the screening, eligible patients received a single IV dose of Aileron peptide-1 on Days 1, 8, and 15, and remained in the clinic for approximately 8 hours after dosing completion for clinical evaluation, laboratory testing and pharmacokinetic assessments. In addition, a tumor biopsy was performed within 48 hours of Dose 3 (last dose) in Cycle 1 or 2 for pharmacodynamic assessment, unless the biopsy posed a significant risk to the patient. The selection of Cycle 1 or 2 was made at the discretion of the Investigator. Patients returned to the clinic for additional observation and laboratory assessments on Day 22 and for end-of-cycle assessments on Day 29.
[0376] Treatment of patients in the dose escalation and the dose expansion phases of the study continued until documentation of disease progression, unacceptable toxicity, or patient or physician decision to discontinue therapy.
p53 Status Determination and Tumor Sampling Requirement Prior to Enrollment:
[0377] A central laboratory tested both archived tissue samples or fresh biopsy samples from all patients enrolled in the study for p53 status using Next-Generation Sequencing (NGS).
[0378] For the First 3 Dose Levels of Stage 1:
[0379] Patients were enrolled irrespective of p53 status. Nevertheless, patients were still tested for p53 status at the central laboratory. To this end, archived tissue was used (sample were not older than 3 years), or alternatively, a fresh biopsy was considered, unless the biopsy posed a significant risk to the patient.
[0380] Starting at Dose Level 4 of Stage 1 (and for patients enrolled in Stage 2 of the DEP):
[0381] Only patients with tumors expressing WT p53 protein were enrolled. This key inclusion criterion was based on the proposed mechanism of action of Aileron peptide-1, which requires WT p53 protein to be pharmacologically active. The inclusion criterion is also supported by results of in vitro tumor growth assays, in which Aileron peptide-1 demonstrated activity in tumor cells expressing WT p53 protein, but not in cells with null-mutations of p53. Patients met the p53 requirement through one of the following scenarios: [0382] Patients were eligible based on a previous p53 gene test result done at a local lab. These patients were still tested for p53 status using NGS at the central laboratory. To this end, archived tissue was used (sample was not be older than 3 years), or alternatively, a fresh biopsy was considered, unless the biopsy posed a significant risk to the patient. Patients who did not have archived tissue and for whom a biopsy posed a significant risk, were not enrolled. [0383] Patients were eligible based on archived tissue tested for p53 (sample was not be older than 3 years) at the central lab, or alternatively, a fresh biopsy was considered, unless the biopsy posed a significant risk to the patient. Patients who didn't have archived tissue and for whom a biopsy posed a significant risk, were enrolled.
[0384] For patients enrolling into the EXP: [0385] Only patients with tumors expressing p53 WT were enrolled, and all patients were tested for p53 status using NGS at the central laboratory PRIOR to enrollment. Archived tissue were used (if sample were not older than 1 year), or alternatively, a fresh biopsy was considered, unless the biopsy posed a significant risk to the patient. Patients who didn't have archived tissue and for whom a biopsy posed a significant risk, were not enrolled.
[0386] Only patients with tumors expressing WT p53 protein were enrolled. The determination of p53 status was performed on tumor samples obtained during the screening period. The assay was performed by study sites with required capabilities; otherwise it was performed at a central laboratory. Results from archival tissue samples, if available, could also be used to determine patient eligibility in the DEP. The total number of patients enrolled in the study depended on the number of dose levels and the number of patients in each cohort before MTD is established. Approximately 27 adult patients, exclusive of replacements for patients who discontinue for non-safety reasons, were enrolled in the DEP, and approximately 30 additional patients were enrolled in the EXP. Enrollment of a total of up to 60 patients was planned for the study. Up to 6 clinical sites in the US were planned. The expected accrual phase is approximately 24 months. The expected follow-up phase is approximately 9 months after the last patient is enrolled, for a total study duration of approximately 33 months.
[0387] Patients who satisfy all inclusion and exclusion criteria, including documentation of WT p53 status, were enrolled in cohorts of 3 to 6 patients to receive Aileron peptide 1. Aileron peptide 1 was administered by IV infusion over 1 hour (.+-.15 min) on Days 1, 8 and 15 of each 28-day cycle. Treatment continued until disease progression, unacceptable toxicity or patient or physician withdrawal of consent. After the MTD was established, approximately 30 additional patients were enrolled in an expansion cohort to gain further experience at this dose level and in particular patient or tumor types.
[0388] Safety was evaluated based on the incidence, severity, duration, causality, seriousness, and type of AE, and changes in the patient's physical examination, vital signs and clinical laboratory results. Investigators used the NCI CTCAE version 4.0 to assess the severity of AEs.
[0389] Because the primary objectives of this study were based on safety and pharmacokinetic, statistical analyses were descriptive in nature and accounted for all doses studied and all observed responses, including patients who achieve a complete response (CR) or partial response (PR) or who maintain stable disease (SD) based on RECIST 1.1. Patients who received at least one dose of Aileron peptide 1 constituted the safety population and were be included in all safety analyses. Patients who completed at least one cycle of Aileron peptide 1 and underwent a post-treatment objective disease assessment constituted the efficacy-evaluable patient population.
Patient Population
Inclusion Criterias
[0390] All patients were required to meet the following inclusion criterias: (i) Male or female patients age 18 years and older, inclusive, at the time of informed consent (ii) Histologically- or cytologically-confirmed malignancy that is metastatic or unresectable and for which standard curative measures do not exist or are no longer effective; (iii) WT p53 status for the relapsing or treatment-refractory solid neoplasm is mandatory for patients enrolling at dose level 4 and higher in Stage 1 of the DEP, as well as for all patients enrolled in Stage 2 of the DEP or in the EXP; (iv) at least one target lesion that is measurable by RECIST 1.1; (v) ECOG performance status 0-1; (vi) predicted life expectancy of .gtoreq.3 months; (vii) adequate hematologic function, measured within 7 days prior to the first dose of Aileron peptide 1 (defined as: ANC .gtoreq.1.5.times.10.sup.9/L, Hemoglobin .gtoreq.9.0 g/d, and Platelets .gtoreq.100.times.10.sup.9/L); (viii) adequate hepatic function, measured within 7 days prior to the first dose of Aileron peptide 1 (defined as: in the absence of disease involvement in the liver:bilirubin .ltoreq.1.5 times institutional ULN:AST and ALT .ltoreq.2.5 times ULN; in the presence of disease involvement in the liver:bilirubin .ltoreq.2 times ULN:AST and ALT .ltoreq.5 times ULN, (ix) adequate renal function, measured within 7 days prior to the first dose of Aileron peptide 1, (defined as: urinalysis with no evidence of +2 or higher proteinuria, serum creatinine .ltoreq.1.5 times institutional ULN or calculated creatinine clearance .gtoreq.50 mL/min (Cockcroft-Gault formula)); (x) acceptable coagulation profile, measured within 7 days prior to the first dose of Aileron peptide 1 (defined as: PT or INR .ltoreq.1.5 times ULN; aPTT .ltoreq.1.5 times ULN); (Xi) at least 4 weeks since prior chemotherapy or biologic therapy, radiotherapy or surgery (intra-thoracic, intra-abdominal or intra-pelvic) with recovery to Grade 1 or baseline of significant toxicities, excluding alopecia, from previous therapies. Palliative radiotherapy for bone lesions .ltoreq.2 weeks prior to the first dose of Aileron peptide 1 is acceptable if acute toxicity has resolved; (xii) negative serum pregnancy test within 14 days prior to the first dose of Aileron peptide 1 for women of child-bearing potential, defined as a sexually mature woman who has not undergone a hysterectomy or who has not been naturally postmenopausal for .gtoreq.24 consecutive months (i.e., who has had menses any time in the preceding 24 consecutive months); (xiii) all patients (males and females) of child-bearing potential agree to use an effective method of birth control (i.e., latex condom, diaphragm, cervical cap, IUD, birth control pill, etc.) beginning two weeks before the first dose of Aileron peptide 1 and for 30 days after the last dose of Aileron peptide 1; (xiv) ability to understand and willingness to sign a written informed consent document; and patients with prostate cancer must continue androgen deprivation therapy, unless such therapy was discontinued 6 months prior to first dose of Aileron peptide 1.
Exclusion Criterias
[0391] Patients who meet any of the following criteria at screening or Day -1 were excluded: (i) previous treatment with investigational agents that affect MDM2 or MDMX activity; known hypersensitivity to any study drug component; (iii) known and untreated brain metastases. Patients with brain metastases that have been treated and demonstrated to be clinically stable for .gtoreq.30 days can be enrolled onto the dose escalation portion of the study; (iv) history of coagulopathy, platelet disorder or history of non-drug induced thrombocytopenia; (v) history of pulmonary embolism within 6 months prior to the first dose of Aileron peptide 1 or untreated DVT; (vi) required concurrent use of anti-coagulants or anti-platelet medication, with the exception of aspirin doses .ltoreq.81 mg/day, low-dose SC heparin or SC low-molecular-weight heparin for DVT prophylaxis, or heparin flushes to maintain IV catheter patency; (vii) patients with pre-existing history of or known cardiovascular risk (for example: history of acute coronary syndromes including myocardial infarction, unstable angina, coronary artery bypass graft, angioplasty, or stenting within 6 months prior to the first dose of Aileron peptide 1; uncontrolled hypertension defined as a systolic BP .gtoreq.160 mmHg and/or diastolic BP .gtoreq.100 mmHg; pre-existing cardiac failure (New York Heart Association class III-IV); atrial fibrillation on anti-coagulants; clinically significant uncontrolled arrhythmias or arrhythmia requiring treatment, with the exceptions of atrial fibrillation and paroxysmal supraventricular tachycardia; severe valvulopathy; corrected QTc interval on screening ECG .gtoreq.450 msec for males and .gtoreq.470 msec for females); (viii) clinically significant gastrointestinal bleeding within 6 months prior to the first dose of Aileron peptide 1; (ix) clinically significant third-space fluid accumulation (e.g., ascites requiring tapping despite the use of diuretics, or pleural effusion that requires tapping or is associated with shortness of breath); (x) pregnant or lactating females; (xi) evidence of serious and/or unstable pre-existing medical, psychiatric or other condition (including laboratory abnormalities) that could interfere with patient safety or provision of informed consent to participate in this study; (xii) active uncontrolled infection, a history of HIV/AIDS, or a history of hepatitis B or C in the absence of hepatocellular carcinoma. Patients with primary liver cancer that have positive hepatitis serology but are not demonstrating active viral hepatitis can be considered for enrollment if they meet all other inclusion and no other exclusion criteria; (xiii) starting at dose level 4 and higher in Stage 1 of the DEP (as well as for all patients enrolling in Stage 2 of the DEP or in the EXP): Cancers with known Human Papilloma Virus (HPV)-association such as HPV-positive cervical cancers, HPV-positive oropharyngeal cancers or HPV-positive anal cancers; (xiv)known history of another primary malignancy that has not been in remission for .gtoreq.2 years. Non-melanoma skin cancer and cervical carcinoma in situ or squamous intraepithelial lesions (e.g., CIN or PIN) are allowed; (xv) any psychological, sociological, or geographical condition that could potentially interfere with compliance with the study protocol and follow-up schedule; (xvi) the required use of any concomitant medications that are predominantly cleared by hepatobiliary transporters (e.g., OATP members OATP1B1 and OATP1B3) within 24 hours of Aileron peptide 1 infusion; (xvii) the use of any investigational agents within 4 weeks or 5 circulating half-lives prior to the first dose of Aileron peptide 1.
Patient Removal/Replacement from Study Therapy
[0392] A patient was removed from the study therapy for a variety of reasons, including: (i) disease progression; (ii) unacceptable adverse event(s); (iii) intercurrent illness that prevents further participation; (iv) clinically significant toxicity despite a 2-week dosing delay or after two dose reductions; (v) patient refusal to continue treatment through the study and/or consent withdrawal for study participation; (vi) patient unable or unwilling to comply with study requirements; (vii) pregnancy or failure to use adequate birth control; (viii) general or specific changes in the patient's condition that render the patient unacceptable for further treatment in this study in the judgment of the investigator
[0393] Any patient who completed enrollment and did not receive a dose of Aileron peptide 1 was replaced. A patient in the dose escalation portion of the study who discontinued the study prior to completion of the first cycle for reasons other than safety was replaced. A patient in the dose expansion portion of the study who discontinued the study participation prior to the completion of the first cycle of treatment for any reason was replaced.
Treatment Plan
Drug Administration Study--1
[0394] The study drug was the investigational agent Aileron peptide 1. This investigational agent was be distributed to clinical sites. Patients began treatment with Aileron peptide 1 within 21 days following the start of screening. Aileron peptide 1 drug was be a frozen liquid product supplied in single-use glass vials. The peptidomimetic macrocycle for injection was stored frozen at .ltoreq.-15.degree. C. Aileron peptide 1 was introduced into an IV infusion bag containing D5W; known as Aileron peptide 1 dosing solution, and was provided by the site pharmacy for administration to the patient. Aileron peptide 1 dosing solution was labeled with a patient identification number. An investigative staff confirmed this information and its relevancy to the intended patient.
[0395] Aileron peptide 1 was administered by IV infusion in D5W over 1 hour (.+-.15 min) on Days 1, 8 and 15 of each 28-day treatment cycle. The pre-defined dose were calculated for each patient based on body weight at the start of each cycle. Aileron peptide 1 was not administered outside of the planned schedule (i.e., infusions on Days 1, 8 and 15 of a 28-day cycle). Deviations if any were noted on the eCRF. Treatment of patients in the dose escalation and the dose expansion phases of the study continued until documentation of disease progression, unacceptable toxicity, or patient or physician decision to discontinue therapy.
[0396] In case of infusion-related reactions, Aileron peptide 1 infusion were temporarily discontinued. Pharmacologic agents and other therapeutic interventions were administered per institutional guidelines. The decision to re-start Aileron peptide 1 infusion was made after a careful assessment of the patient.
Starting Dose, Dose Escalation and Dose Reduction
Dose Levels for the Dose Escalation Portion of Study
[0397] In the Dose Escalation portion of the study, increasing dose levels of Aileron peptide 1 was evaluated in cohorts of 3-6 patients. Aileron peptide 1 was administered by IV infusion over 1 hour (.+-.15 min) on Days 1, 8 and 15 of each 28-day cycle. Patients enrolled in Cohort 1 received Aileron peptide 1 at Dose Level 1 (0.16 mg/kg). Based on allometric scaling, the projected AUC in humans at 0.16 mg/kg (50 .mu.ghr/mL) is approximately 9% of the rat AUC at STD.sub.10 and approximately 6% of the AUC at the monkey HNSTD.
[0398] In the absence of DLT, subsequent cohorts of 3 to 6 patients received escalated doses until the MTD was established.
[0399] A 2-stage dose escalation design was employed. During the initial Stage 1 Escalation Phase (Table 6), 100% dose increments was utilized until .gtoreq.1 of 3 patients in a cohort experiences any Grade .gtoreq.2 AE that is at least possibly related to study drug. Subsequent dose escalation continued using 3-patient cohorts and the modified Fibonacci sequence (i.e., Stage 2 Escalation Phase; Table 7), until the MTD was established.
TABLE-US-00013 TABLE 6 Stage 1 Dose escalation schedule Dose Level Aileron peptide 1 Dose (mg/kg) Level -2 0.04 Level -1 0.08 Level 1 0.16 Level 2 0.32 Level 3 0.64 Level 4 1.25 Level 5 2.5 Level 6 5 Continue 100% dose increase in subsequent cohorts until the MTD is established or Stage 2 escalation schedule is initiated
TABLE-US-00014 TABLE 7 Stage 2 Dose escalation Schedule Dose Level Aileron peptide 1 Dose (mg/kg) Level 1 Last Stage 1 Dose x 1.67 Level 2 Previous Dose x 1.5 Level 3 Previous Dose x 1.4 Level 4 Previous Dose x 1.33 In the absence of DLT, continue dose escalation at 33% increments after Stage 2 Level 4 until the MTD is established
[0400] The escalation scheme were switched to the Stage 2 Escalation Schedule at any point that the Investigators, Sponsor's Medical Monitor and Safety Physician representative agreed on a more conservative progression.
[0401] The observation of DLT(s) was used to make individual patient determinations regarding dose reductions, interruptions or discontinuation throughout the course of the trial, but DLTs occurring during Cycle 1 were used to inform safety and tolerability assessments for dose escalation decisions.
[0402] If DLTs was observed in the first cohort, the dose was de-escalated to Dose Level -1. If DLTs were observed at Dose Level -1, the dose was de-escalated to Dose Level -2. If DLTs was observed at Dose Level -2, other dose levels were considered and implemented after discussions among the Investigators, Sponsor's Medical Monitor and Safety Physician representative.
[0403] At least three patients were treated at each dose level. If no patients experience a DLT, then the subsequent 3 patients were treated at the next planned dose level.
[0404] If DLT was observed in .gtoreq.2 of 3 patients in a cohort, then no further dose escalation occurred, and the current dose was defined as the MAD.
[0405] If DLT was observed in 1 of 3 patients, then up to 3 additional patients were enrolled at that same dose level. If DLT was observed in .gtoreq.2 patients in the expanded cohort, then no further dose escalation will occur, and the current dose was defined as the MAD.
[0406] After the MAD was defined, either the previously administered lower dose was expanded to a total of 6 patients, or an intermediate (between the MAD and the next lower dose level) was investigated in up to six patients. The highest dose tolerated without DLT in at least 5 of 6 patients in a cohort was defined as the MTD.
Dose Level for the Expansion Portion of Study
[0407] After the MTD was defined, approximately 30 additional patients were enrolled in an expansion study to gain further experience at this dose level and investigate the effect of Aileron peptide 1 in specific patient or tumor types. Two disease types were selected for evaluation, and 15 patients of each disease type were enrolled in each of two cohorts in the expansion study. The dose of Aileron peptide 1 administered to patients in the expansion cohort was derived from evaluation of available safety and other information from patients in the dose escalation portion of the study.
Intra-Patient Dose Escalation
[0408] Intra-patient dose escalation was not permitted.
Dose and Schedule Adjustments for Toxicity
[0409] Toxicity that occurred during a cycle was required to recover as outlined below for treatment to continue.
[0410] Hemoglobin .gtoreq.8.5 g/dL; ANC .gtoreq.1.0 10.sup.9/L; platelet count .gtoreq.75.times.10.sup.9/L; liver function tests back to grade prior to previous cycle (includes PT/INR); other toxicities must return to Grade .ltoreq.1 or to baseline level if Grade >1 was acceptable for inclusion in the trial.
[0411] If a clinically significant AE was observed in a patient during a treatment cycle, further dosing was be delayed until the toxicity has resolved to an acceptable level. Treatment can be delayed by up to 2 weeks to allow for the resolution of AEs, and a dose reduction to the preceding level can be made at the discretion of the Investigator in consultation with Sponsor's Medical Monitor and Safety Physician representative. If a patient experiences multiple AEs, decisions on dosing delay or dose reduction was based on the most severe AE. Any patient who experienced recurrent, clinically significant AE after one dose reduction underwent one additional dose reduction. Patients who continued to experience clinically significant toxicity after a 2-week delay or two dose reductions were discontinued from the study.
[0412] Adverse events considered for dose reduction did not include the events assessed by the investigator as exclusively related to underlying disease or other medical condition or concomitant treatment. A patient who experienced an AE considered related to Aileron peptide 1 continued on study if the patient was receiving clinical benefit and/or the Investigator felt continued participation was in the best interest of the patient. In such cases, at the Investigator's discretion and in agreement with Sponsor's Medical Monitor and Safety Physician representative, the dose for a patient was reduced to the preceding lower level.
[0413] Up to two dose reductions for a patient were permitted, after which the patient were discontinued from the study.
[0414] A patient who experienced a DLT continued treatment at the preceding lower level at the discretion of the Investigator and in agreement with Sponsor's Medical Monitor and Safety Physician representative until disease progression or unacceptable toxicity. Once the dose had been reduced for a patient, it was not re-escalated.
[0415] Toxicity grading was based on NCI CTCAE v4.0.
Statistical Methods
[0416] Statistical analyses of safety and efficacy for DEP and EXP were primarily descriptive in nature because the objectives of the study were to determine the DLTs and MTD. These objectives were achieved by the results of a deterministic algorithm; thus, statistical hypothesis testing was neither intended nor appropriate within this context. Continuous variables were summarized using descriptive statistics [n, mean, standard deviation, median, minimum, and maximum]. Categorical variables were summarized showing the number and percentage (n, %) of patients within each classification.
Study Procedures
Schedule of Study Events
[0417] The schedule of study activities, including assessments, tests, exams, disease assessments, submission of tissue specimens, and study drug administration) that will be conducted, beginning with screening and continuing through Cycle 1 [day 1, day 8, and day 15 of a 28 day cycle] are outlined in Table 8. Study that will be conducted beginning with Cycle 2 [day 29 of cycle 1=day 1 of cycle 2] are listed on Table 9.
TABLE-US-00015 TABLE 8 Schedule of study activities through Cycle 1 Day 29/ Day 1, Cycle Day 8 .+-. Day 15 .+-. 2 .+-. Clinical Day 1 1 d 1 d 3 d Molecular Screen -21 pre- post- Day 2 .+-. Day 3 .+-. pre- post- pre- post- Day 16 .+-. Day 22 .+-. Refer to Screen days Day -1 dose dose 4 h 4 h dose dose dose dose 2 d 1 d Table 9 Written informed X X consent Medical history X Demographics X Tumor biopsy or X X archive tissue sample for p53 WT confirmation and biomarker assessment Confirm X X eligibility Blood test for X HIV, hepatitis B and C Serum or urine X pregnancy Vital signs: X X X X X X X X X X X X Blood pressure, pulse, respiration rate, body temperature Physical exam X X X X 12-lead ECG X X X Laboratory X X X X X X X X assessments: Clinical chemistry (glucose, calcium, albumin, total protein, sodium, potassium, CO.sub.2, chloride, BUN [blood urea nitrogen], serum creatinine, ALP, ALT, AST, total and direct bilirubin), hematology (complete blood count, platelets and differential), urinalysis (dipstick measurement [pH, specific gravity, protein, glucose, ketones, nitrite, leukocyte esterase] with microscopic analysis, if results of the dipstick indicate additional testing required), coagulation (PT, INR, aPTT). Collection of X blood for immunogenicity Collection of X X X X X X X X X blood for biomarker assessments Collection of X X X X X X X X X blood for pharmacokinetic assessments ECOG X X X X Performance Status Needle biopsy X X for biomarker assessments Tumor X Assessment Aileron peptide 1 X X X dosing Concomitant X X X X X X X X X X X X medications AE assessment X X X X X X X X X
TABLE-US-00016 TABLE 9 Schedule of study activities through Cycle 2 Day 29 of prior cycle/ Day 1 of next cycle for End-of-Study patients continuing Day 8 .+-. Day 15 .+-. 30 .+-. 3 d treatment .+-. 3 d 1 d 1 d After even after last Pre- post- pre- Post- pre- post- Day 16 .+-. numbered dose or study dose dose dose dose dose dose 2 d cycles withdrawal Serum pregnancy X Vital signs: X X X X X X X X Blood pressure, pulse, respiration rate, body temperature. Physical exam X X X X 12-lead ECG X X At X pre-dose pre-dose and and EOI (+10 EOI (+10 min) min) Laboratory X X X X X assessments: (Hematology Clinical chemistry, only) hematology, urinalysis, coagulation (PT, INR, aPTT). Collection of blood X X X for immunogenicity Collection of blood X X X X X for biomarker assessments (each cycle) Collection of blood X X X X X X for pharmacokinetic assessments (Cycle 2 and End-of-Study only) ECOG Performance X X X X status Needle Biopsy for X biomarker assessments Tumor assessment X At X end of even- numbered cycles. Prior to start of the next treatment cycle Aileron peptide 1 X X X dosing Concomitant X X X X X X X X medications AE assessment X X X X X X X X (begins at the point of the first Aileron peptide 1 infusion and continues until 30 days after last infusion)
Pharmacokinetic Analysis
[0418] Levels of Aileron peptide 1 and its metabolites will be measured in blood samples collected at specific time points described below. Pharmacokinetic data will be tabulated and summarized by individual patient and collectively by dose level. Graphical displays will be provided where useful in the interpretation of results.
[0419] Blood samples for pharmacokinetic assessment will be collected at the following time points:
TABLE-US-00017 TABLE 10 Time points for collection of blood samples for pharmacokinetic assessment Cycle 1 Day 1 within one hour before SOI EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) 8 hr after EOI (.+-.10 min) Day 2 24 hours (.+-.4 hr) after the start of Day 1 infusion Day 3 48 hours (.+-.4 hr) after the start of Day 1 infusion Day 8 within one hour before SOI EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Day 15 within one hour before SOI EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) 8 hr after EOI (.+-.10 min) Day 16 24 hours (.+-.4 hrs) after the start of Day 15 infusion Cycle 2 Cycle 1 Day 29/ within one hour before SOI Cycle 2 Day 1 EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Day 15 within one hour SOI EOI (.+-.5 min) 30 min after EOI (.+-.5 min) Day 15 (cont.) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Day 16 24 hours (.+-.4 hrs) after the start of Day 15 infusion End of study visit During end of study visit
[0420] SOI stands for start of infusion of the Aileron peptide 1; EOI stands for the end of infusion of the Aileron peptide 1.
Pharmacodynamic Analysis
[0421] Levels of p53, MDM2, MDMX, p21 and caspase will be measured in tumor specimens collected before beginning treatment and at the end of Cycle 1 or Cycle 2. MIC-1 will be measured in blood samples. The specific time points for blood and tissue collection for pharmacodynamic assessments are described below. Pharmacodynamic data will be tabulated and summarized by individual patient and collectively by dose level. Graphical displays will be provided where useful in the interpretation of results.
[0422] Results available from previous genetic and biomarker tests, and additional tests of the blood and tumor samples for biomarkers relevant to the safety and efficacy of Aileron peptide-1 can be investigated for possible correlation with patient outcome.
[0423] Blood samples for pharmacodynamic assessments will be collected at the following time points:
TABLE-US-00018 TABLE 11 Time points for collection of blood samples for pharmacodynamic assessments Cycle 1 Day 1 within 1 hour before the start of infusion (SOI) Day 2 24 hours (.+-.4 hr) after the start of Day 1 infusion Day 3 48 hours (.+-.4 hr) after the start of Day 1 infusion Day 8 within 1 hour before SOI within 1 hour after the end of infusion (EOI) Day 15 within 1 hour before SOI within 1 hour after EOI Day 16 24 hours (.+-.4 hrs) after the start of Day 15 infusion Day 22 During Day 22 visit Each Subsequent Cycle Starting in Cycle 2 Cycle 1 Day 29/ within 1 hour before SOI Cycle 2 Day 1 within 1 hour after EOI Day 15 within 1 hour before SOI Day 16 24 hours (.+-.4 hrs) after the start of Day 15 infusion End of study visit During end of study visit
Assessment of Clinical Activity of the Peptidomimetic Macrocycle
[0424] To evaluate clinical activity, response rates and duration of response based on RECIST 1.1 or other appropriate criteria will be provided with a case-by-case description of all patients who exhibit CR, PR or SD. A descriptive analysis of other evidence of anti-tumor activity or other clinical benefit will be provided based on clinical, radiographic or other appropriate assessment of efficacy or clinical anti-tumor activity. Analysis of clinical activity will be conducted on two patient populations: (1) the subset of patients who receive at least one cycle of therapy and have at least one post-baseline disease assessment (the efficacy-evaluable population) and (2) a larger group of patients that includes the efficacy-evaluable population as well as patients who exhibit objective disease progression or experience a DLT and/or unacceptable toxicity prior to the end of Cycle 1.
[0425] Imaging scans, physical examination, and/or laboratory-based assays (e.g., prostate specific antigen) for patients with relevant disease indications will be obtained at baseline (within 21 days of Cycle 1 Day 1) and for objective tumor assessment after the second cycle of treatment and after every second treatment cycle thereafter (Cycle 4, Cycle 6, etc.). The same type of imaging, physical examination, or laboratory-based assay procedure will be used for each assessment for a patient. RECIST 1.1 will be used to assess tumor response and duration of response. Scheduled scans (and/or other laboratory-based assay) will be interpreted prior to the start of the next treatment cycle. If the criteria for a CR or PR are met, then the scan will be repeated no earlier than within 4 weeks to confirm the response. A responding patient (CR, PR or SD) will continue on study, with disease assessment after every second cycle, until disease progression, withdrawal of informed consent, or unacceptable toxicity.
[0426] Films or other records from imaging procedures, including those procedures performed at a regional or other facility outside of the primary institutions, will be read and reviewed by the radiology staff at the corresponding primary study institution for the patient.
Drug Administration Study--II
Study Objectives
[0427] This Phase I open-label, multi-center, dose-escalation, 2-arm study was designed to evaluate the safety, tolerability, pharmacokinetic, pharmacodynamic, and anti-tumor effects of Aileron peptide-1 administered by IV infusion using 2 different dosing regimens of a 28- or 21-day cycle, in patients with advanced solid tumors or lymphomas expressing WT p53 protein (see p53 Status Determination below). Patients received Aileron peptide-1 either once weekly for three consecutive weeks for a 28-day cycle or twice weekly for two consecutive weeks for a 21-day cycle. Many patients with a solid tumor or lymphoma present circulating tumor cells (CTC) in peripheral blood, which can be detected and analyzed using flow cytometry. This enabled the detection of study drug-specific target engagement in these cells.
[0428] This study consisted of a DEP and a EXP. The DEP was a "3+3" dose escalation designed to establish the MTD or the OBD of Aileron peptide-1. The EXP enrolled up to 2 distinct groups of patients with specific solid tumors to further investigate the clinical safety profile and potential efficacy of Aileron peptide-1 at the MTD or OBD.
Starting Dose, Dose Escalation, and Dose Reduction
[0429] All subjects were dosed at a pre-defined level based on body weight. Starting at Dose Level (DL) 3, patients was sequentially assigned to one of two treatment arms: Dose Regimen (DR) A testing administration of Aileron peptide-1 once per week, or Dose Regimen (DR) B testing administration of Aileron peptide-1 twice per week. For Dose Level 3, DR-A were enrolled first, DR-B were enrolled second. The starting dose (DL-1) in DEP, based on results from nonclinical toxicology assessments, was 0.16 mg/kg.
[0430] During the first 2 dose levels, patients received Aileron peptide-1 on Days 1, 8, and 15 of a 28-day cycle. Starting with DL 3, patients in DR-A continued being treated once a week on Days 1, 8, and 15 of a 28-day cycle, whereas patients in DR-B were treated twice a week, on Days 1 and 4, 8 and 11 of a 21-day cycle. This dosing schedule is summarized in FIG. 2.
[0431] Doses were doubled in subsequent dose levels until .gtoreq.1 of 3 patients in a cohort experienced any drug-related Grade .gtoreq.2 adverse event (AE). A drug-related AE is an event that is possibly, probably or definitely attributed to Aileron peptide-1. Grading of AEs was defined by the NCI Common Terminology Criteria for Adverse Events (CTCAE) version 4.03. Subsequent dose escalation continued using modified Fibonacci sequence (i.e., 67%, 50%, 40%, and 33%; FIGS. 3 and 4).
[0432] Escalation to the next dose level within each DR proceeded in the absence of DLT at the completion of Cycle 1 (treatment cycle=28 days for DR-A and 21 days for DR-B). Escalation to the next dose level within each DR was decided by a Safety Review Committee (SRC), consisting of the Principal Investigators, Sponsor's Medical Monitor, and Safety Physician representative, which reviewed all available safety information from all patients.
[0433] Within each Dose Regimen cohort, if no DLT was observed in a cohort, the subsequent patient group was enrolled at the next planned dose level of that dose regimen. If a DLT was observed in .gtoreq.2 of 3 patients at any dose level no further dose escalation occurred in that DR, and the current dose was defined as the maximum administered dose (MAD). If a DLT was observed in 1 of 3 patients in a cohort at any dose level, then up to 3 additional patients were enrolled in the same DR at that dose level. If a DLT was observed in 2 or more patients in the expanded cohort, then no further dose escalation occurred, and the current dose was defined as the MAD. After the MAD was defined, either the previously administered lower dose was expanded to a total of 6 patients, or an intermediate dose (between the MAD and the previous dose level) was investigated in a total of 6 patients. The highest dose tolerated in at least 5 of 6 patients was defined as the MTD or OBD.
[0434] The selection of dose regimen and dose level for up to 2 EXP cohorts was based on the MTD determination in Cycle 1, as well as the cumulative safety, efficacy and pharmacokinetic/pharmacodynamic profile of Aileron peptide-1 in subsequent treatment cycles in DEP.
[0435] Dose levels were not increased between cycles within each cohort, and patients were assigned only one dose level (i.e., no intra-patient dose escalation).
Statistical Methods
[0436] Results from DR-A and DR-B will be compared for all dose levels and patient groups.
Screening Assessments and Other Requirements Prior to Day 1 of Cycle 1
[0437] Molecular Screening Prior to Day 1 of Cycle 1: Molecular screening encompassed the following prior to the first administration of Aileron peptide-1 (Day 1 of Cycle 1): (i) collection of signed informed consent for molecular screening; (ii) collection of an archived tumor sample or a fresh tumor biopsy (unless a biopsy poses significant clinical risk) for p53 testing; (i) if confirmed to be p53 WT, the remainder of the tissue sample from enrolled patients was used to test for pharmacodynamic biomarkers. Confirmation of p53 WT status before administration of the first dose of Aileron peptide-1 was mandatory for enrollment in stage 1 of DEP for patients starting at Dose Level 4 and higher and stage 2 (if necessary) of DEP and EXP for all patients.
[0438] Molecular Screening Prior At Dose Level 4 and higher in Stage 1 of the DEP (as well as for all patients enrolled in Stage 2 of the DEP), molecular screening in patients with unknown p53 status was done prior to initiating the clinical screening. If the p53 status was known to be WT, these patients proceeded to clinical screening and were enrolled and receive Aileron peptide-1 before confirmation of p53 WT by the central laboratory.
[0439] In the EXP, patients had completed molecular screening at the central laboratory prior to proceeding to enrolment. These patients were only enrolled and received Aileron peptide-1 after confirmation of p53 WT by the central laboratory.
Clinical Screening within 21 Calendar Days Prior to Day 1 of Cycle 1 for DR-A and DR-B, all Dose Levels
[0440] The screening assessments and procedures performed within 21 calendar days (or as noted) prior to the first administration of Aileron peptide-1 (Day 1 of Cycle 1) included collection of signed informed consent, medical history (evaluation of baseline signs and symptoms), demographics, eligibility assessment, blood test for HIV, hepatitis B and C, vital signs (includes blood pressure, pulse, respiration rate, body temperature), physical examination, ECG, laboratory assessments including clinical chemistry (glucose, calcium, albumin, total protein, sodium, potassium, CO.sub.2, chloride, phosphate, BUN [blood urea nitrogen], serum creatinine, uric acid, ALP, ALT, AST, total and direct bilirubin), hematology (complete blood count, platelets and differential), urinalysis (dipstick measurement [pH, specific gravity, protein, glucose, ketones, nitrite, leukocyte esterase] with microscopic analysis, if results of the dipstick indicate additional testing required), coagulation (PT, INR, aPTT), ECOG performance status, RECIST--(for solid tumor patients) or IWG--(for lymphoma patients) compliant imaging for disease assessment and tumor measurements as well as laboratory-based assays (e.g., prostate specific antigen) for patients with relevant disease indications, including a baseline PET-FDG and possibly FLT-PET scan(s), concomitant medications (current medications and those taken within 28 days of Cycle 1, Day 1).
Within 7 Calendar Days Prior to Day 1 of Cycle 1 for DR-A and DR-B, all Dose Levels
[0441] The screening assessments that was completed within 7 calendar days prior to the first administration of Aileron peptide-1 (Day 1 of Cycle 1) included serum or urine pregnancy test (.beta.-hCG) for women of child-bearing potential: performed within 2 days prior to first dose of Aileron peptide-1, confirm eligibility, vital signs, laboratory assessments--can be omitted if screening tests were performed within 7 days prior, ECOG performance status, and concomitant medications.
Requirements During Cycle 1
Day 1 of Cycle 1, for DR-A and DR-B, all Dose Levels
[0442] The study procedures that were performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI, physical exam, ECG: within 30 minutes prior to SOI (performed in triplicate (5-10 min between readings)), collection of blood for immunogenicity within 1 hr prior to SOI, collection of blood for all biomarker assessments within 1 hr prior to SOI, collection of blood for pharmacokinetic assessments: within 1 hr prior to SOI, and concomitant medications.
[0443] The study procedures that were performed after administration of Aileron peptide-1 included vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) At EOI (.+-.5 min), and 1 (.+-.5 min) and 2 hrs (.+-.10 min) following EOI; ECG: At EOI (.+-.5 min) and 1 hr (.+-.5 min) and 2 hrs (.+-.10 min) following EOI. Perform in triplicate (5-10 min between readings) only if patient has a QTc that is a) >500 msec; b) increased by 60 msec over pre-dose; or c) decreased by 50 msec below pre-dose recording; collection of blood for pharmacokinetic assessments: At EOI (.+-.5 min), 30 min (.+-.5 min) and 1 hr (.+-.5 min), 2 (.+-.10 min), 4 (.+-.10 min) and 8 hrs (.+-.10 min) following EOI; collection of blood for all biomarker assessments, EOI (.+-.5 min) and 1 hr (.+-.5 min) and 2, 4, and 8 hrs (.+-.10 min) following EOI; concomitant medications; and adverse event (AE) assessment
Day 2 of Cycle 1, for DR-A and DR-B, all Dose Levels
[0444] The study procedures that were performed included vital signs, laboratory assessments, collection of blood for all biomarker assessments at 24 hr (.+-.4 hr) after Day 1 SOI, collection of blood for pharmacokinetic assessments at 24 hr (.+-.4 hr) after Day 1 SOI, concomitant medications, AE assessment, and TLS monitoring (via routine laboratory assessment sample)
Day 3 of Cycle 1, for DR-A and DR-B, all Dose Levels
[0445] The study procedures performed included vital signs, laboratory assessments (collection of blood for all biomarker assessments at 48 hr (.+-.4 hr) after Day 1 SOI), collection of blood for pharmacokinetic assessments at 48 hr (.+-.4 hr) after Day 1 SOI, concomitant medications and AE assessment.
Day 4 of Cycle 1, for DR-B ONLY, all Dose Levels
[0446] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI, physical exam, ECG: within 30 minutes prior to SOI. Perform in triplicate (5-10 min between readings), laboratory assessments, collection of blood for immunogenicity within 1 hr prior to SOI, collection of blood for all biomarker assessments within 1 hr prior to SOI, collection of blood for pharmacokinetic assessments: within 1 hr prior to SOI, concomitant medications, and dverse Event (AE) Assessments
[0447] The study procedures performed after administration of Aileron peptide-1 included vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) At EOI (.+-.5 min), and 1 and 2 hrs (.+-.10 min) following EOI; ECG: At EOI (.+-.5 min) and 1 hr (.+-.5 min) and 2 hr (.+-.10 min) following EOI. Perform in triplicate (5-10 min between readings) only if patient has a QTc that is a) >500 msec; b) increased by 60 msec over pre-dose; or c) decreased by 50 msec below pre-dose recording; laboratory assessments; collection of blood for all biomarker assessments within 1 hr after EOI; ollection of blood for pharmacokinetic assessments at EOI (.+-.5 min), 30 min (.+-.5 min) and 1 hr (.+-.5 min), 2 (.+-.10 min), 4 (.+-.10 min) after EOI; concomitant medications and adverse event (AE) assessment
Day 8 of Cycle 1 for DR-A and DR-B, all Dose Levels
[0448] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI; physical exam; laboratory assessments, collection of blood for all biomarker assessments within 1 hr prior to SOI, collection of blood for pharmacokinetic assessments within 1 hr prior to SOI, ECOG performance status, concomitant medications and AE assessment.
[0449] The study procedures performed after administration of Aileron peptide-1 include vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) At EOI (.+-.5 min), and 1 hr (.+-.5 min) and 2 hrs (.+-.10 min) following EOI; collection of blood for all biomarker assessments within 1 hr after EOI; collection of blood for pharmacokinetic assessments at EOI (.+-.5 min) and 30 min (.+-.5 min), 1 hr (.+-.5 min), 2 and 4 hrs (.+-.10 min) after EOI; concomitant medications and AE assessment.
Day 15 for DR-A and Day 11 for DR-B of Cycle 1
[0450] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI; physical exam; laboratory assessments; collection of blood for all biomarker assessments within 1 hr prior to SOI; collection of blood for pharmacokinetic assessments within 1 hr prior to SOI; ECOG performance status; concomitant medication and AE assessment.
[0451] The study procedures performed after administration of Aileron peptide-1 included vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) At EOI (.+-.5 min), and 1 hr (.+-.5 min) and 2 hrs (.+-.10 min) following EOI; collection of blood for pharmacokinetic assessments At EOI (.+-.5 min) and 30 min (.+-.5 min), 1 hr (.+-.5 min); 2, 4, and 8 hrs (.+-.10 min) after EOI; collection of blood for all biomarker assessments within 1 hr at EOI (.+-.5 min); EOI plus 1 hr (.+-.5 min); 4, and 8 hr (.+-.10 min) after EOI; concomitant medications; and AE assessment.
Day 16 DR-A and Day 12 DR-B for Cycle 1
[0452] The study procedures performed included vital signs; laboratory assessments; collection of blood for biomarker assessments: at 24 hr (.+-.4 hr) after SOI on the day prior; only for patients with a successful study biopsy performed prior to start of study medication: Needle biopsy for biomarker assessments- to be performed within 48 hours of Cycle 1, Day 15 (DR-A) or Day 11 (DR-B) infusion OR Cycle 2, Day 15 (DR-A) or Day 11 (DR-B) infusion, at the discretion of the Investigator (unless the biopsy poses significant risk to the patient); collection of blood for pharmacokinetic assessments at 24 hr (.+-.4 hr) after Day 15 (DR-A) or Day 11 (DR-B) SOI; collection of blood for all pharmacodynamic assessments at 24 hr (.+-.4 hr) after SOI on the day prior; concomitant medications; cAE assessment; and FLT-PET for patients who received FLT-PET at screen and have SUV .gtoreq.5.
Day 22 for DR-A and Day 18 for DR-B or of Cycle 1
[0453] The study procedures performed included vital signs; laboratory assessments--hematology only; collection of blood for all biomarker assessments; concomitant medications; and AE assessment.
Day 29 for DR-A and Day 22 for DR-B of Cycle 1 (-1 Day Up to +3 Days)/Cycle 2 Day 1
[0454] Procedures listed below under Requirements During Subsequent Cycles Beginning with Cycle 2 were performed. Note: "Day 22 or 29"=Day 1 of next cycle for patients continuing treatment. Cycle 1 Day 22 or 29/Cycle 2 Day 1 pre-dose evaluations were to be done within 3 days prior to next cycle drug administration.
[0455] If the patient was not continuing treatment beyond Cycle 1 the procedures listed below under End-of-Study Visit section were performed.
Requirements During Subsequent Cycles Beginning with Cycle 2
Day 29 for DR-A and Day 22 for DR-B of Prior Cycle/Day 1 of Cycle 2 and Subsequent Cycles
[0456] Note: "Day 22 or 29"=Day 1 of next cycle for patients continuing treatment. Day 22 or 29 of prior cycle/Day 1 of current cycle pre-dose evaluations were done within 3 days prior to drug administration.
[0457] Note: Blood samples to evaluate CTC were not collected in Cycle 2 or subsequent cycles.
[0458] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI; physical exam; ECG: within 30 minutes prior to SOI. Perform in triplicate (5-10 min between readings); laboratory assessments; collection of blood for immunogenicity: within 1 hr prior to SOI; collection of blood for biomarker assessments (MIC-1 only): within 1 hr prior to SOI; collection of blood for pharmacokinetic assessments (Cycle 2 Only): within 1 hr prior to SOI; ECOG performance status; concomitant medications; and AE assessment.
[0459] The study procedures performed after administration of Aileron peptide-1 included vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) at EOI (.+-.5 min) and as clinically indicated following EOI; ECG: at EOI (.+-.5 min). Perform in triplicate (5-10 min between readings) only if patient has a QTc that is a) >500 msec; b) increased by 60 msec over pre-dose; or c) decreased by 50 msec below pre-dose recording; collection of blood for biomarker assessments (MIC-1 only): within 1 hr after EOI; collection of blood for pharmacokinetic assessments (Cycle 2 Only): At EOI (.+-.5 min) and 30 min ((.+-.5 min), 1 hr (.+-.5 min), 2 and 4 hrs (.+-.10 min) after EOI; concomitant medications; and AE assessment.
Day 8 of DR-A and Days 4 and 8 of DR-B of Cycle 2 and Beyond
[0460] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 minutes prior to SOI; physical exam; laboratory assessments--hematology only; ECOG performance status; concomitant medications; and AE assessment.
[0461] The study procedures performed after administration of Aileron peptide-1 included vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) at EOI (.+-.5 min) and as clinically indicated following EOI; concomitant medications; and AE assessment.
Day 15 of DR-A and Day 11 of DR-B of Cycle 2 and Beyond
[0462] The study procedures performed prior to administration of Aileron peptide-1 included vital signs: within 30 min prior to SOI; physical exam; laboratory assessments; collection of blood for biomarker assessments (MIC-1 only): within 1 hr prior to SOI; collection of blood for pharmacokinetic assessments (Cycle 2 Only): within 1 hr prior to SOI; ECOG performance status; concomitant medications; and AE assessment.
[0463] The study procedures performed after administration of Aileron peptide-1 included: vital signs: (During infusion) 30 min (.+-.3 min); (Post-infusion) at EOI (.+-.5 min) and as clinically indicated following EOI; collection of blood for biomarker assessments (MIC-1 only): within 1 hr after EOI; collection of blood for pharmacokinetic assessments (Cycle 2 Only) at EOI (.+-.5 min) and 30 min (5 min), 1 hr (.+-.5 min), 2 and 4 hrs (.+-.10 min) after EOI; concomitant medications and AE assessment.
Day 16 DR-A and Day 12 of DR-B of Cycle 2 and Beyond
[0464] The study procedures performed included vital signs, laboratory assessments, collection of blood for biomarker assessments: at 24 hr (.+-.4 hr) after Day 15 or 11 SOI, collection of blood for pharmacokinetic assessments (Cycle 2 Only): at 24 hr (.+-.4 hr) after Day 15 or 11 SOI, concomitant medications, and AE assessment.
After Even Numbered Cycles
[0465] Blood was collected for immunogenicity. Tumor assessment was performed following same procedure used for baseline measurement, for example imaging, physical exam as well as laboratory-based assays (e.g., prostate specific antigen) for patients with relevant disease indications.
[0466] For patients who achieved "Stable Disease" as defined by RECIST or IWG criteria, a FDG-PET scan was indicated, provided that an evaluable FDG-PET-scan was performed prior to starting treatment with study drug.
CT Imaging
[0467] All patients receive a CT image prior to the first dose. After dosing commences in Dosing Regimen--A (DR-A), CT images will be obtained at the end of Cycle 2 and every other cycle thereafter in DR-A, e.g., Cycles 4, 6, and 8. In Dosing Regimen-B (DR-B) CT images will be obtained after the last infusion in Cycle 3 and every third cycle thereafter in DR-B, e.g., Cycles 6, 9, and 12. Images will be obtained after the last dose is administered in those cycles but prior to the Day 18 visit.
End-of-Study Visit
[0468] The end-of-study visit was to be conducted 30 (.+-.2) calendar days after the last administration of Aileron peptide-1 or withdrawal from the study. The study procedures performed included serum or urine pregnancy, vital signs, physical exam, ECG, laboratory assessments, collection of blood for immunogenicity, collection of blood for biomarker assessment, collection of blood for pharmacokinetic assessment, ECOG performance status, tumor assessment following same procedure used for baseline measurement, for example imaging, physical exam as well as laboratory-based assays (e.g., prostate specific antigen) for patients with relevant disease indications, concomitant medication and AE assessment.
Pharmacodynamic Assessment
[0469] Blood samples for pharmacodynamic assessments were collected at the following timepoints:
TABLE-US-00019 TABLE 12 Cycle 1 and Cycle 2 Dose Regimen Pharmacodynamic Assessments Dose Regimens Assessment Blood Sample Collection Schedule Cycle 1 DR-A, DR-B, or Both: Day 1- Both (pre) MIC-1 and within 1 hour before the start of CTC infusion (SOI) Day 1- Both (post) Samples EOI (+5 min) & EOI + 1 hr (.+-.5 min), 2, 4, and 8 hr (.+-.10 min) Day 2- Both 24 hours (.+-.4 hr) after SOI on Day 1 Day 3- Both 48 hours (.+-.4 hr) after SOI on Day 1 Day 8 DR-A within 1 hour before SOI and Day 4 & 8 DR-B within 1 hour after the end of infusion (EOI) Day 15 DR-A within 1 hour before SOI and Day 11 DR-B within 1 hour after EOI Day 15 DR-A within 1 hour before SOI and Day 11 DR-B EOI (+5 min) & EOI + 1 hr (.+-.5 min), 2, 4, and 8 hr (.+-.10 min) Day 16 DR-A 24 hours (.+-.4 hrs) after SOI day Day 12 DR-B prior Day 22 DR-A During Day visit Day 18 DR-B Each Subsequent Cycle Starting in Cycle (Cy) 2 Cy 1 Day 29 DR-A MIC-1 Only within 1 hour before SOI and Cy 1 Day 23 DR-B = within 1 hour after EOI Cycle 2 Day 1 Day 15 DR-A within 1 hour before SOI and Day 11 DR-B within 1 hour after EOI Day 16 DR-A 24 hours (.+-.4 hrs) after SOI day Day 12 DR-B prior End of study visit During end of study visit NOTE: no pharmacodynamic assessments for solid tumors or lymphoma on Day 8 DR-A or Days 4 and 8 DR-B
Pharmacokinetic (PK) Assessments
[0470] Blood samples for pharmacokinetic assessment were collected at the following timepoints:
TABLE-US-00020 TABLE 13 Cycle 1 and Cycle 2 Dose Regimen Pharmacokinetic Assessments Cycle 1 Day 1 within one hour before SOI DR-A and DR-B EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) 8 hr after EOI (.+-.10 min) Day 2 24 hours (.+-.4 hr) after SOI day DR-A and DR-B prior Day 3 48 hours (.+-.4 hr) after SOI Day 1 DR-A and DR-B Day 8, DR-A within one hour before SOI Days 4 & 8, DR-B EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Day 15, DR-A within one hour before SOI Day 11, DR-B EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) 8 hr after EOI (.+-.10 min) Day 16, DR-A 24 hours (.+-.4 hrs) after SOI day Day 12, DR-B prior Cycle 2 Cycle 1 Day 29/ within one hour before SOI Cycle 2 Day 1, DR-A EOI (+5 min) Cycle 1 Day 23/ 30 min after EOI (.+-.5 min) Cycle 2 Day 1, DR-B 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Cycle 2 Day 15, DR-A within one hour SOI Day 11, DR-B EOI (+5 min) 30 min after EOI (.+-.5 min) 1 hr after EOI (.+-.5 min) 2 hr after EOI (.+-.10 min) 4 hr after EOI (.+-.10 min) Day 16, DR-A {close oversize brace} 24 hours (.+-.4 hrs) after SOI day Cycle 2 Only prior Day 12, DR-B NOTE: no PK assessments on Day 8 in DR-A or Days 4 and 8 in DR-B End of study visit During end of study visit
Example 4: Further Studies
[0471] Aileron peptide-1 was evaluated for safety, tolerability, pharmacokinetics and pharmacodynamics in adult patients with advanced solid tumors or lymphomas expressing WT p53 that are refractory to or intolerant of standard therapy, or for which no standard therapy exists. FIG. 6 shows one way Aileron peptide-1 was designed to inhibit MDMX and/or MDM2, which leads to reactivation of WT p53.
[0472] Aileron peptide-1 was able to penetrate the cell membrane and localize within the nucleus. Further Aileron peptide-1 can disrupt protein-protein interactions within the cell such as the interactions between p53 and MDM2 and MDMX.
[0473] Several in-vivo and in-vitro studies of Aileron peptide-1 were conducted. In these studies, Aileron peptide-1 binded to both MDM2 and MDMX with nanomolar affinities and demonstrated evidence of specific on-target mechanism in vitro by gene expression profiling. In addition, Aileron peptide-1 demonstrated tumor growth suppression, p53-dependent cell cycle arrest, apoptosis and anti-tumor activity in an MDM2/MDMX-overexpressing xenograft cancer model with clear correlation to on-target pharmacokinetic and pharmacodynamic, or pharmacokinetic/pharmacodynamic, activity.
[0474] Dose escalation phase were designed to evaluate Aileron peptide-1 in patients with solid tumors or lymphoma. The dose escalation phase was not limited by type of tumor or lymphoma. Aileron peptide-1 was administered to patients with sarcoma, gastric cancer, non-small cell lung cancer, ovarian cancer and thymoma. In some cases Aileron peptide-1 was used to treat tumors and lymphomas in which WT p53 is prevalent in greater than 50% of patients. p53 wild-type status is prevalent in greater than 50% of patients suffering from at least 19 different tumor types. Thus, indication potential can vary from orphan indications or large market opportunities. See e.g., FIG. 7.
[0475] p53 signal activation study was conducted to determine if Aileron peptide-1 had a differential effect on cancer cell lines with mutant p53 compared to WT p53. In the study, we measured the effect of Aileron peptide-1 in 312 cell lines across a variety of different cancers to compare the effect of Aileron peptide-1 in cell lines with mutant p53 and cell lines with WT p53. See FIG. 8. In the 207 mutant p53 cell lines, Aileron peptide-1 had no discernable effect, but in the 105 WT p53 cell lines, nearly all showed tumor cell death. See FIG. 8. The WT p53 cell lines that did not show tumor cell death included WT p53 cell lines associated with Human Papilloma Virus, or HPV, related cancers, such as cervical and head and neck. By concentrating on WT p53 and responsive tumors, we are able to predict patient populations that can have a better chance of response from our product candidate.
[0476] In another study, the binding affinity of Aileron peptide-1 for MDM2 or MDMX relative to the binding affinity for MDM2 and MDMX of WT p53 and of an MDM2 small molecule inhibitor was measured. The affinity of a drug to a receptor is the measure of how effectively that drug binds to its target and can provide insight on the potential for on-target effect and off-target toxicity. Aileron peptide-1 was designed to bind to MDM2 and/or MDMX with a higher affinity than WT p53 so that Aileron peptide-1 disrupts the binding of MDM2 and/or MDMX to WT p53 by binding to MDM2 and/or MDMX in lieu of p53. Such binding can enable p53 to be released and activated. In this study, we also measured a small molecule MDM2 inhibitor's binding affinity to MDMX, which showed no binding to this target. Table 14 below shows Aileron peptide-1's ability to bind to MDM2 and MDMX relative to WT p53 and the small molecule MDM2 inhibitor.
TABLE-US-00021 TABLE 14 Aileron peptide-l's ability to bind to MDM2 and MDMX relative to WT p53 and the small molecule MDM2 inhibitor. Kd, nM WT p53 Aileron peptide-l MDM2 Inhibitor MDM2 770 13.7 9.8 MDMX 480 8.9 >3000
In Vivo
[0477] We studied the effects of Aileron peptide-1 in both solid tumors. In the study depicted FIGS. 9a and 9b, we evaluated the effect of Aileron peptide-1 administered by an intravenous, or IV, injection in an MDMX-driven MCF-7 breast cancer xenograft model. In this study, we evaluated different doses, schedules and durations of treatment with Aileron peptide-1 and vehicle to determine effect on tumor volume growth. Aileron peptide-1 showed statistically significant tumor growth inhibition at doses ranging from 2.5 mg/kg to 5 mg/kg to 10 mg/kg and 20 mg/kg when these doses were administered twice a week for a 28 day period. See FIGS. 11a and 11b.
Toxicology and Nonclinical Safety Experiments
[0478] The pivotal 4-week multiple-dose GLP studies in rats and monkeys utilized twice-weekly IV dosing rather than the once-weekly IV dosing planned as the initial clinical regimen. The studies provided dose- and exposure-related assessments during both dosing and recovery periods, and results were utilized to define the maximum tolerated doses (MTD) and estimate the severely toxic dose for 10% (STD.sub.10) of rats and the highest non-severely toxic dose (HNSTD) in monkeys. All gross and microscopic signs of intolerance (e.g., reduced organ weights, sporadic findings of multi-tissue hemorrhage and hepatic necrosis) and changes in serum chemistry parameters were considered as secondary to red blood cell (RBC), platelet and/or white blood cell (WBC) depletions or anorexia and dehydration in both species. Recovery assessments revealed regenerative and compensatory changes consistent with marrow cell survival and reversibility of all related hematologic and secondary toxicities.
[0479] The DLT in both animal species appears to be related to the suppression of hematopoietic cells in the bone marrow, in particular cells of the megakaryocyte lineage, resulting in significant decreases in peripheral blood platelets that demonstrated recovery upon the cessation of dosing. See FIG. 7.
[0480] The STD.sub.10 in rats was defined at 10 mg/kg based on the mortality of one animal in a satellite group for hematology sampling during recovery. The HNSTD in monkeys was defined at 5 mg/kg, based on a complete lack of significant thrombocytopenia at this lowest dose level. However, almost all of the monkeys at the mid- and high-dose levels tolerated Aileron peptide-1 administration well; only one animal at each of these dose levels developed significant thrombocytopenia (<100,000.times.10.sup.6/ml).
[0481] Rats are more sensitive to the bone marrow and hematologic effects of Aileron peptide-1 than monkeys on the basis of exposures at maximally tolerated doses. Exposure at rat STD.sub.10 (AUC.sub.0-.infin.=562 .mu.ghr/mL at 10 mg/kg) was below that of HNSTD in monkeys (AUC.sub.0-.infin.=813 .mu.ghr/mL at 5 mg/kg). These in vivo results correlate with those obtained from in vitro hemotoxicity assays via luminescence output (HALO). In these investigations, Aileron peptide-1 in general inhibited the induced proliferation of bone marrow precursor cells from rats to a greater extent than those from monkeys or humans. IC.sub.50 values were .about.2- to 8-fold higher for rat cells than for monkey or human cells, with the largest difference noted for megakaryocyte colony forming cells, the platelet precursors. These results correlate with in vivo findings indicating that rats are more sensitive to the bone marrow and hematologic effects of Aileron peptide-1 than monkeys on the basis of dose and exposures at maximally tolerated doses. These results also suggest that, in terms of projecting potential bone marrow and hematological toxicity levels in humans, the monkey pharmacokinetic--pharmacodynamic data can be more clinically relevant than the rat data.
[0482] Aileron peptide-1 was negative in genetic toxicology studies, including bacterial mutagenicity (Ames), chromosomal aberrations (human peripheral blood lymphocyte) and in vivo micronucleus (rat bone marrow) assays. Safety pharmacology studies were performed to assess the effects of Aileron peptide-1 on hERG potassium channels in vitro and on cardiac function in cynomolgus monkeys. There were no significant adverse findings in these studies.
[0483] Compared to the twice-weekly IV dosing schedule utilized in the 4-week GLP toxicity studies, the first-in-human clinical trial of Aileron peptide-1 will initially assess once-weekly IV dosing for three weeks. In addition, the demonstrated reversibility of Aileron test peptide-1-induced hematologic effects, the ability to detect such findings with routine laboratory measurements, and the availability of effective supportive therapies, all provide additional safety margin in the clinic.
Pharmacokinetics and Absorption, Distribution, Metabolism and Excretion
[0484] In rats, Aileron peptide-1 generally showed linear, dose-proportional increases in C.sub.max and AUC. In the 4-week rat GLP toxicity study, C.sub.max of Aileron peptide-1 ranged from 49.9 to 186 .mu.g/mL, AUC.sub.0-.infin. ranged from 90.5 to 562 .mu.ghr/mL, and clearance ranged from 19.2 to 28.3 mL/hr/kg. Half-life (t.sub.1/2) values could not be calculated due to variable coefficients of determination (r.sup.2<0.9).
[0485] In non-human primates, Aileron peptide-1 generally showed exposures that increased proportionally with dose, although an apparent plateau in exposure was observed at the high-dose group (20 mg/kg) in the 4-week monkey GLP toxicity study. In the study, C.sub.max of Aileron peptide-1 ranged from 133 to 562 .mu.g/mL, t.sub.1/2 ranged from 3.7 to 6.0 hrs, AUC.sub.0-.infin. ranged from 813 to 1,600 .mu.ghr/mL, and clearance ranged from 6.5 to 13.8 mL/hr/kg.
[0486] No significant sex-based differences in pharmacokinetic parameters were observed in either rats or monkeys, and no accumulation was observed following repeated doses on a twice-weekly schedule in the GLP toxicity studies.
[0487] Proteolysis is the expected major biotransformation pathway of Aileron peptide-1. The predominant metabolite, Aileron peptide metabolite-1, is a 3-amino acid truncation with the cyclic peptide portion intact, and the same metabolite profile was noted in in vitro stability studies with monkey, rat, mouse and human cryopreserved hepatocytes. In a single-dose rat study, hepatobiliary metabolism and elimination represented the predominant clearance pathway for Aileron peptide-1, with Aileron peptide metabolite-1 as the major excretion product observed in the bile.
[0488] In vitro studies revealed that Aileron peptide-1 is not an inhibitor of any cytochrome P450 (CYP) isoforms tested. In vitro assays for CYP induction also did not indicate any significant treatment-related effects with Aileron peptide-1. Based on these findings, the potential of clinically relevant drug-drug interactions for concomitant medications that are cleared through CYP-mediated mechanisms is deemed to be low.
[0489] Aileron peptide-1 was tested in vitro against common transporters, and >90% inhibition of organic anion transporter polypeptide (OATP) members OATP1B1 and OATP1B3 and bile salt export pump (BSEP) at concentrations that can be clinically relevant (e.g., at C.sub.max of high-dose levels) was observed. Based on these findings, the potential of clinically relevant drug-drug interactions by Aileron peptide-1 with medications (e.g., methotrexate, statins) that are significantly cleared by hepatobiliary transporters should be considered.
In Vivo
[0490] An open-label, multi-center, dose-escalation, two-arm study was used to design to evaluate the safety, tolerability, pharmacokinetic, pharmacodynamic and anti-tumor effects of Aileron peptide-1 administered by intravenous (IV) infusion in patients with advanced solid tumors or lymphomas expressing WT p53 that are refractory to or intolerant of standard therapy, or for which no standard therapy exists. The study included a dose escalation phase to establish the maximum tolerated dose, or MTD, or the optimum biologic dose, or OBD, of Aileron peptide-1, and a dose expansion phase to investigate the clinical safety profile and potential efficacy of Aileron peptide-1 at the MTD or OBD. In the expansion phase of the study, Aileron peptide-1 was studied in distinct groups of patients with specific solid tumors or lymphomas. The selection of the solid tumors or lymphomas was finalized based on results of the dose escalation phase, as well as data from additional nonclinical pharmacology studies. The latter included the investigation of multiple solid cancer cell lines such as breast, bladder, head/neck, gastrointestinal, or GI, liver, lung, pancreas, prostate and sarcoma to facilitate the comparison of cell line sensitivity to Aileron peptide-1 across and within tumor types. Treatment of patients in the dose escalation and the dose expansion phases of the trial continued until documentation of disease progression, unacceptable toxicity, or patient or physician decision to discontinue therapy.
[0491] The dose escalation phase is based on a "3+3" dose escalation design. In the dose escalation phase, patients in the first two dose levels received Aileron peptide-1 once a week for three weeks every 28 days. Patients in the higher dose levels received Aileron peptide-1 either once weekly for three consecutive weeks for a 28-day cycle or twice weekly for two consecutive weeks for a 21-day cycle. See FIG. 10.
[0492] Patients with solid tumors or lymphoma were enrolled in the dose escalation phase who had exhausted standard therapies or for whom standard therapies are not available, completed enrollment up to dose group 4b and are enrolled patients in dose group 5a. Patients suffering from cancers with known HPV-association were excluded from enrollment, because HPV is known to deactivate WT p53. The tumor types included as of such date are non-small cell lung cancer, various types of sarcoma, cholangiocarcinoma, cystic adenoid carcinoma, follicular non-hodgkin lymphoma, thymoma, prostate cancer, endometrial cancer, and ovarian cancer. Since our trial was primarily safety and tolerability focused, we started dosing at relatively low dose levels and the protocol did not require patients in the first three dose levels to be p53-wildtype or HPV-negative.
[0493] To identify specific p53 patients for our trial, we employed a central laboratory to test both archived tumor tissue samples and fresh biopsy samples from patients enrolled in the trial for p53 status using Next-Generation Sequencing. 12 of 13 patients enrolled in those dose levels were confirmed to have WT status. Starting in dose level 4, WT p53 status was a mandatory eligibility criterion.
[0494] In this trial, clinical activity or response to Aileron peptide-1 was assessed through the use of both pharmacodynamic biomarkers and imaging assessment. Pharmacodynamic biomarkers provided us with information as to on-target activity, specific patient type response and early insight as to effect on tumor. As part of the trial, we were also assessing the effect of Aileron peptide-1 on potential Pharmacodynamic biomarkers in various different sources of biological samples such as tumor biopsies, circulating tumor cells where detectable, mononuclear blood cells and blood samples. Dependent on the sample type, those pharmacodynamic biomarkers include measurements of MDMX, MDM2, p21, p53, apoptosis and macrophage inhibitory cytokine-1, or MIC-1. In addition, we received standard imaging assessments, such as computed tomography, or CT, magnetic resonance imaging, bone scans and PET scans from patients, depending on number cycles administered. CT-imaging was performed at the end of cycle 2, and every two cycles thereafter in the 28-day cycle group and at the end of cycle 3 and every three cycles thereafter in the 21-day cycle group. We were measuring anti-tumor activity using RECIST for patients with solid tumors and 2014 International Working Group, or IWG, criteria for patients with lymphomas, enabling us to objectively evaluate whether a tumor has progressed, stabilized or shrunk. In addition, anti-tumor effects can be determined by physical examination or clinically validated serum tumor markers.
Pharmacokinetic Profile
[0495] Aileron peptide-1 was delivered systemically in an IV administration given potential advantages of avoiding metabolic impact from hepatic and gastrointestinal enzymes as well as ability for reproducible systemic bioavailability with dose escalation. As shown in the FIG. 11a, drug concentration was measured in dose levels for Cohorts 1 (0.16 mg/kg), 2 (0.32 mg/kg), 3a (0.64 mg/kg), 3b (0.32 mg/kg), 4a (1.25 mg/kg). In patients, Aileron peptide-1 has consistently produced a dose dependent increase in maximum drug serum concentration observed, or Cmax, as well as longer corresponding half-life of between eight and 10 hours. This half-life is adequate to re-activate WT p53 and begin the process of initiating the regulation of the transcription of genes.
[0496] Aileron peptide-1 shows reproducible profiles from patient to patient and dose-to dose enabling exposure projections for higher dose levels to predict efficacy and safety. FIG. 11b shows the measured drug concentration in dose levels 1 (0.16 mg/kg), 2 (0.32 mg/kg) and 3 (0.64 mg/kg); and projected for dose levels 4 (1.25 mg/kg), 5 (2.5 mg/kg), and 6 (5.0 mg/kg).
[0497] FIG. 12 shows the pharmacokinetic model of Aileron peptide-1. The peptide shows nonlinear Michaelis-Menten clearance and linear elimination.
Safety Results
[0498] Aileron peptide-1 was considered to be well tolerated at all dose levels by the investigators. There were no reported dose-limiting toxicities nor study-related serious adverse events. Looking at non-hematologic safety, the most common related adverse events are nausea and fatigue. Looking at hematologic safety, the first two dose levels 1 and 2 showed no cytopenias during cycles 1 and 2, whereas at dose levels 3A, 3B and 4A, patients showed drug-related events of mild to moderate anemia, mild thrombocytopenia and mild neutropenia. One patient at dose level 3B experienced a grade 4 neutropenia which Investigators reported as probably related to study medication. The patient's complete blood count presented trough values of grade 2 leukocytopenia, grade 1 anemia and grade 1 thrombocytopenia. Two concomitant medications were initiated around the same time that treatment with Aileron peptide-1 was started, both of which have been suspected to be associated with the occurrence of neutropenia. There was no association between the patient's neutropenia and drug exposure, the patient's last complete blood count showed an improvement to grade 3 neutropenia, no treatment was administered for neutropenia and no infectious complications were reported.]
[0499] 4 formal Safety Review Meetings with Investigators confirm no DLTs. For DL1, 2 and 3A there was an unanimous decision to escalate at double the dose. For DL3B, there was an unanimous decision to escalate via Fibonacci in DL4B. New Dose may be 0.53 mg/Kg instead of 0.64 mg/Kg.
[0500] Hematologic and non-hematologic adverse events were generally consistent with our preclinical toxicology profile: [0501] No genotoxicity [0502] No immunogenicity [0503] No relevant findings in cardiovascular safety [0504] No relevant findings suggestive of GI toxicity [0505] No Myelosuppression as the dose-limiting toxicity
Biomarker Assessments
[0506] In the dose escalation phase, we used several exploratory biomarkers to confirm Aileron peptide-1's pharmacological or on-target biological activity, aid patient recruitment and help inform dose selection.
[0507] Pharmacodynamic biomarkers were received on MDMX, MDM2, p21, p53, apoptosis and MIC-1. The first biomarker for which we received data is MIC-1. MIC-1 is a secreted p53-regulated cytokine that is readily measured in blood if p53 is activated, and can serve as a biomarker for p53 activation. Under normal conditions, p53 expression remains low, resulting in corresponding negligible levels of MIC-1. However, when WT p53 activation occurs in response to a tumor, this also leads to increased levels of MIC-1. We measured MIC-1 one hour before initial infusion and again 24 hours after initial infusion. In patients at dose levels ranging from dose level 1 to dose level 4A, we observed a statistically significant dose dependent response in the increase of MIC-1. See FIG. 13.
[0508] Further, mononuclear blood cells from 4 patients confirmed that Aileron peptide-1 penetrates cell membrane and activates p53-signalling. We measured the amount of intracellular p53 and p21 in mononuclear blood cells from 4 patients at: (a) end of infusion of Aileron peptide-1, (b) 1 hour after the end of infusion of Aileron peptide-1 and (c) 4 hour after the end of infusion of Aileron peptide-1. As seen in FIG. 14, a 1.8 times increase in the levels of intracellular p53 and about 3 times increase in the levels of intracellular p21 was observed.
[0509] Thus, we conclude that Aileron peptide-1 is penetrating the cell membrane, localizing within the nucleus and releasing WT p53. An increase of at least eight fold in MIC-1 levels from baseline serves as guidance for the minimum dose needed for p53 re-activation.
[0510] Overall, at least two independent biomarker studies support Aileron peptide-1-mediated activation of intracellular p53 signaling: (i) MIC-1 serum-protein (as measured by ELISA): dose-response relationship, and (ii) p53 and p21 increase (as measured by flow-cytometry) in blood cells.
Efficacy
[0511] Objective tumor response is an endpoint for efficacy in trials. Patients in the 28-day cycle group are measured at baseline and again after two cycles of therapy, or approximately within 56 days following initial dosing. Patients in the 21-day cycle group are measured at baseline and again after three cycles of therapy, or approximately within 63 days following initial dosing. RECIST criteria definitions are as follows: [0512] Stable Disease, or SD: Neither sufficient shrinkage to qualify for partial response nor sufficient increase to qualify for progression, taking as reference the smallest sum diameters while on study. [0513] Partial Response, or PR: At least a 30% decrease in the sum of the diameters of target lesions, taking as reference the baseline sum diameters. [0514] Complete Response, or CR: Disappearance of all target lesions. Any pathological lymph nodes, whether target or non-target, must have reduction in short axis to less than 10 millimeters.
[0515] Tests show that patients who have completed at least two cycles of treatment, some patients have stable disease. Aileron peptide-1 has shown a stable disease rate. See FIG. 15.
[0516] Table 15 below shows exemplary patients who were treated with Aileron peptide-1. These patients encompassed a range of solid tumors with either wild type or mutant p53. As seen in Table 16, after 2/3 cycle treatment each of the patient 4, 5, 7, 8, 10, 11 and 15 have stable disease, while only patients 2, 6 and 12 showed a progressive disease. After completing 3/4 treatment cycles, patient 11 continued to show stable disease. As used here, stable disease refers to the situation where there is neither a sufficient shrinkage of the tumor to qualify for partial response nor sufficient increase to qualify for progression, taking as reference the smallest sum diameters while on study.
TABLE-US-00022 TABLE 15 Patient information Tumor Tumor Patient P53 NGS Tissue(s) Size Burden No. 1.degree. Cancer Histopath 2.degree. Cancer WT Tested Organ mm mm 1 Salivary gland squamous cell UNK liver Lymph node 30 55 Right Hepatic 25 Lobe Posterior 2 liposarcoma liposarcoma Yes retroperitoneal: soft tissue 48 48 mass 3 Gall bladder adenocarcinoma Yes Gall bladder liner 16 36 endoperitoneal 20 4 Submandibular Adenoid cystic Yes adenoid oropharynx 43 65 adenocarcinoma Carcinoma lung 22 5 liposarcoma Liposarcoma Yes liposarcoma Retroperitoneal 175 301 retroperitoneal 126 6 GIST (sarcoma) GASTROINTESTINAL UNK N/A MESENTRIC 25 135 STROMAL TUMOR MASS lung 20 lung 16 liver 84 7 NSCLC invasive Brain/head UNK N/A lung 10 71 andenocarcinoma Lymph node 20 lung 20 Lymph node 21 8 liposarcoma Basal Cell Yes tumor Stomach 147 443 resection- Stomach 127 retroperitoneal Bone 155 lymph node Liver 14 9 prostate Adenocarcinoma UNK N/A Lymph node 48 86 liver 15 Lymph node 23 10 NSCLS Stage IV Yes right lung Lymph node 15 99 adenocarcinoma nodule Lymph node 21 the left lung Lymph node 21 Lung 25 Lung 17 11 ovarian met. ov cancer No Tumor Liver 17 64 stage iv ca with resection- Liver 30 liver mets uterus Other 17 12 endometrial ENDOMETRIAL No Liver 13 78 SEROUS Liver 17 CARCINOMA Lymph node 18 Lymph node 20 13 breast Carcinoma osteosarcoma Yes Tumor lung 64 181 resection- lung 117 osteosarcoma 14 leiomyosarcoma Unknown UNK Lung 54 195 Liver 12 Liver 99 Liver 30 15 leiomyosarcoma Uterine Yes Lymph node 44 76 Lymph node 32 16 lymphona b-cell lymphoma- Lymph node 300 follicular (CT & PET) 17 thymoma
TABLE-US-00023 TABLE 16 Patient response after treatment with Aileron Peptide -1 for 2/3 cycle Tumor Tumor % change Patient Size Burder from New No. Organ mm mm bsln Lesions? Target Non-Target Overall 1 Lymph node Off Study: Right Hepatic Lobe Non- Posterior compliance 2 soft tissue mass 62 62 29% No Progression Not Done Progressive Disease 3 liner Off Study: endoperitoneal CDP 4 oropharynx 45 69 6% Yes Stable Progressive Stable lung 24 Disease Disease Disease 5 retroperitoneal 186 336 11.2% NO Stable Not done Stable retroperitoneal 150 Disease Disease 6 MESENTRIC 29 159 17.8% Yes Stable Non-complete Progressive MASS (SUPERIOR Disease Response/Non- Disease lung 11 HEPATIC progression lung 19 LOBE UP liver 100 TO 1.5 CM) 7 lung 13 77 8% No Stable Non-complete Stable Lymph node 20 Disease Response/Non- Disease lung 20 progression Lymph node 24 8 Stomach 157 518 16.9% No Stable Progressive Stable Stomach 145 Disease Disease Disease Bone 180 Liver 36 9 Lymph node 53 122 41.86% No Progressive Non-Complete Off Study: liver 35 Disease Response/Non- Objective Lymph node 34 Progression Disease Progression 10 Lymph node 21 116 17.20% No Stable Not done Stable Lymph node 24 Disease Disease Lymph node 24 Lung 28 Lung 19 11 Liver 17 71 10.90% No Stable Not done Stable Liver 24 Disease Disease Other 30 12 Liver 15 100 28.21% Yes (LIVER Progression Not all Progressive Liver 19 LESION) Evaluated or Disease Lymph node 32 Not Assessable Lymph node 32 13 lung -- -- -- lung -- 14 Lung 69 233 19.48% Yes (left Not entered Not entered yet Not entered Liver 19 subhepatic yet yet Liver 105 mass) Liver 40 15 Lymph node 44 77 1.30% No Stable Non-complete Stable Lymph node 33 Disease Response/Non- Disease progression
Sequence CWU 0 SQTB SEQUENCE LISTING The patent application
contains a lengthy "Sequence Listing" section. A copy of the
"Sequence Listing" is available in electronic form from the USPTO
web site
(http://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20190269753A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
0 SQTB SEQUENCE LISTING The patent application contains a lengthy
"Sequence Listing" section. A copy of the "Sequence Listing" is
available in electronic form from the USPTO web site
(http://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20190269753A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
References
















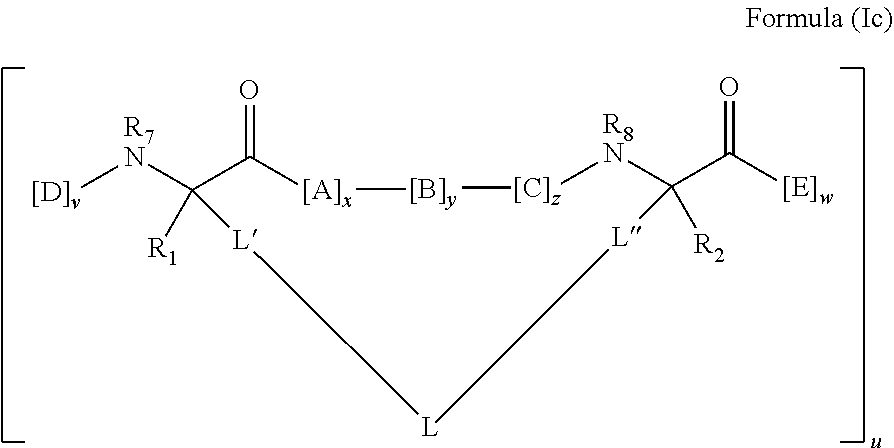





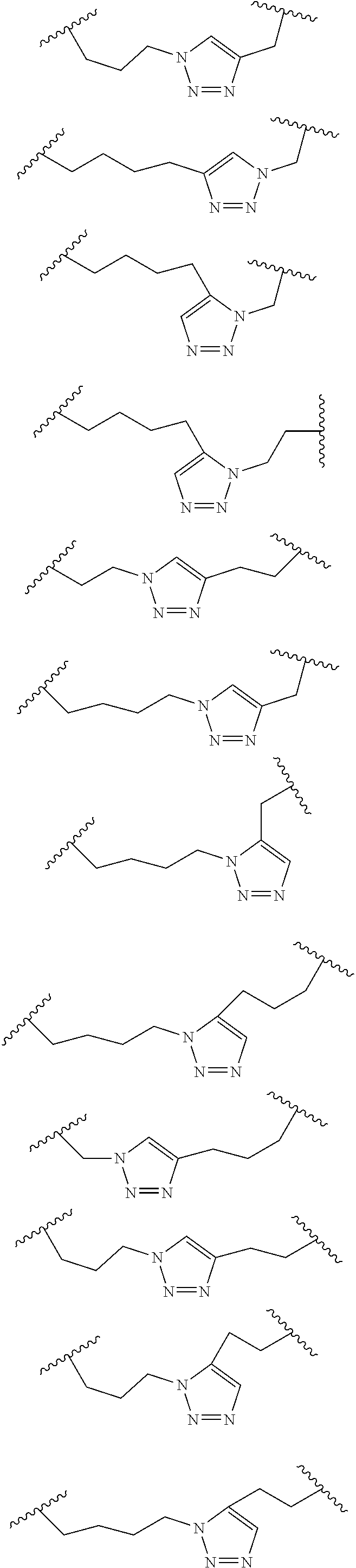



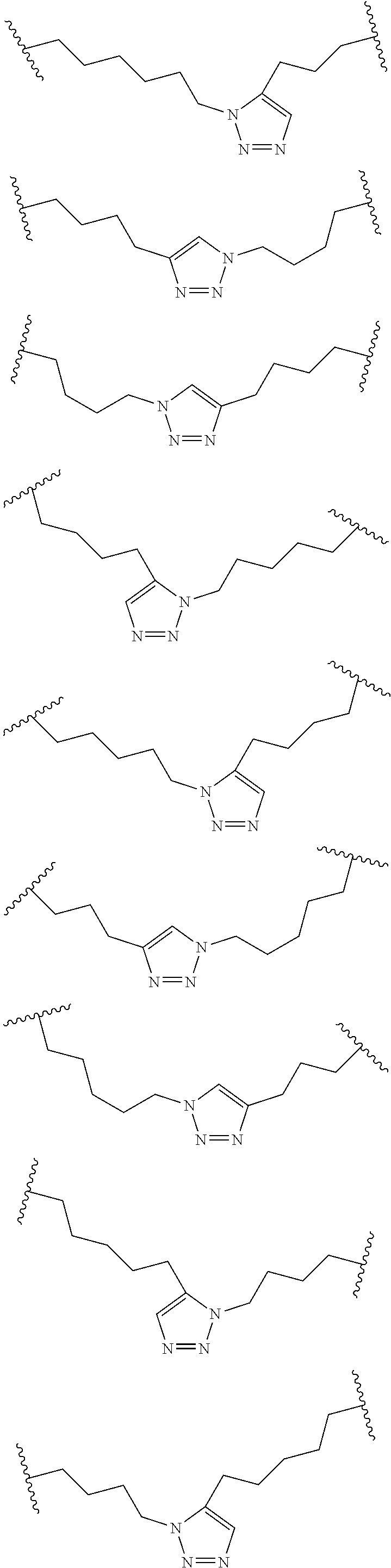

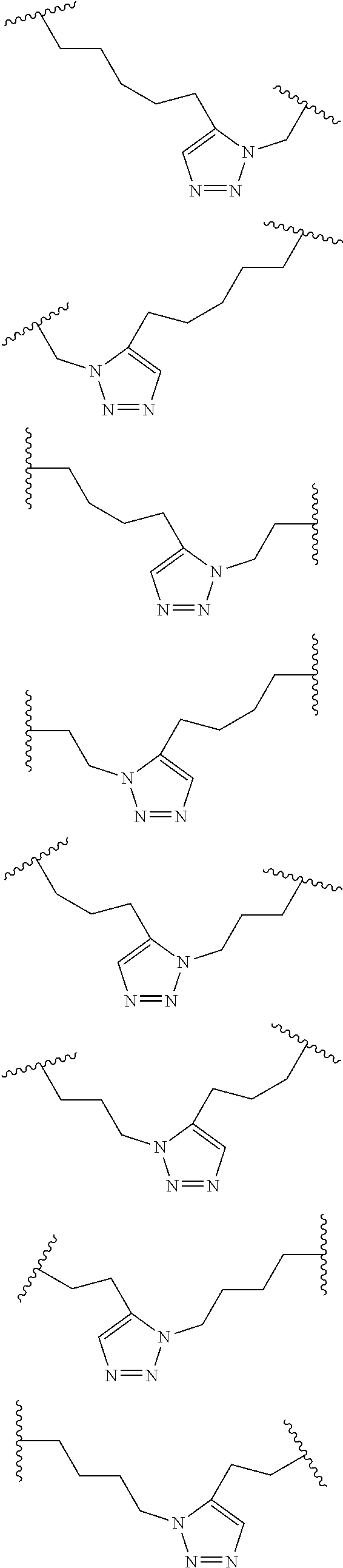
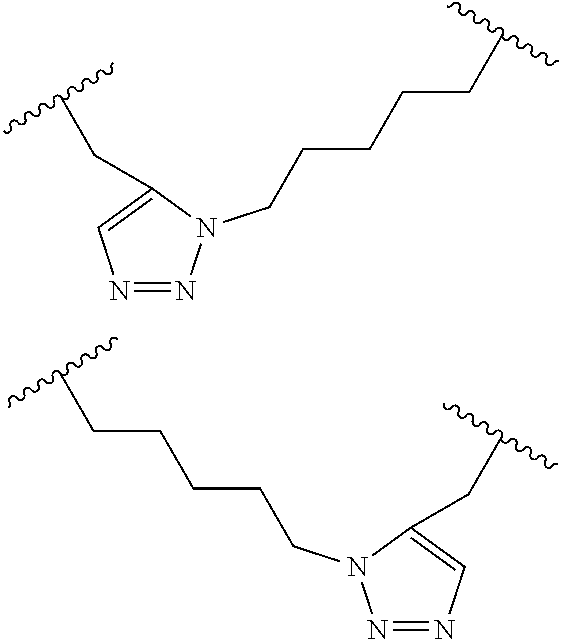








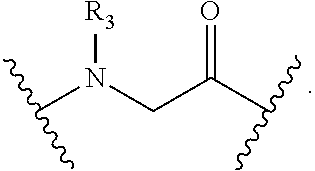


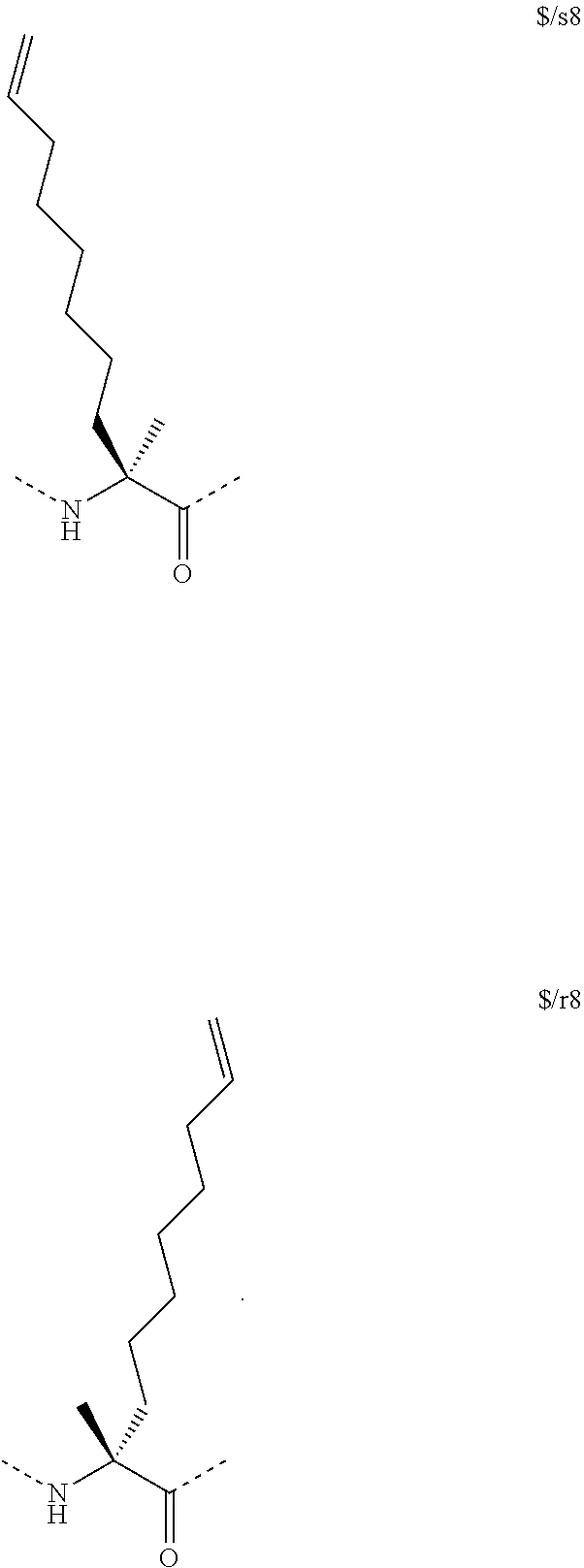


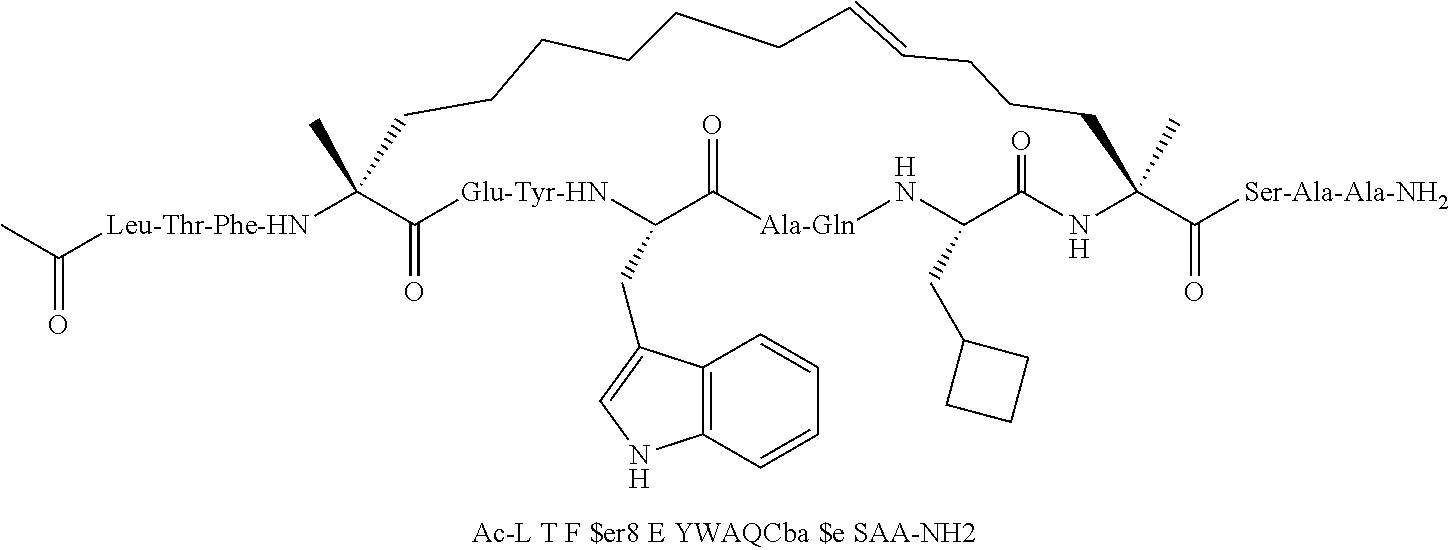
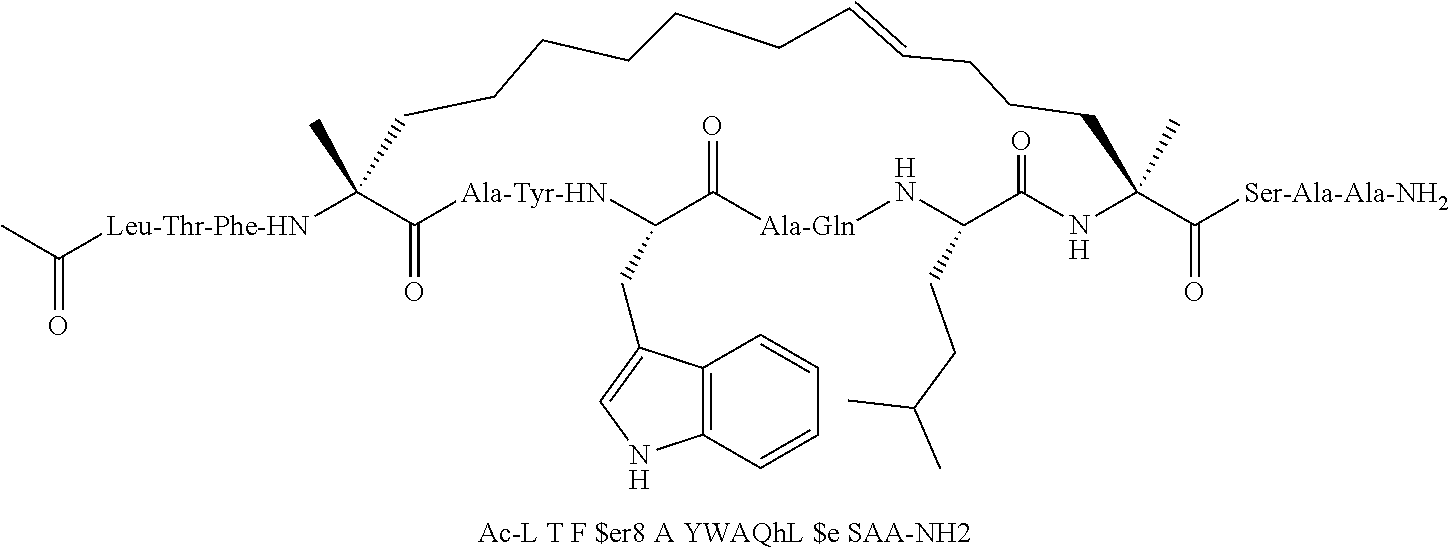




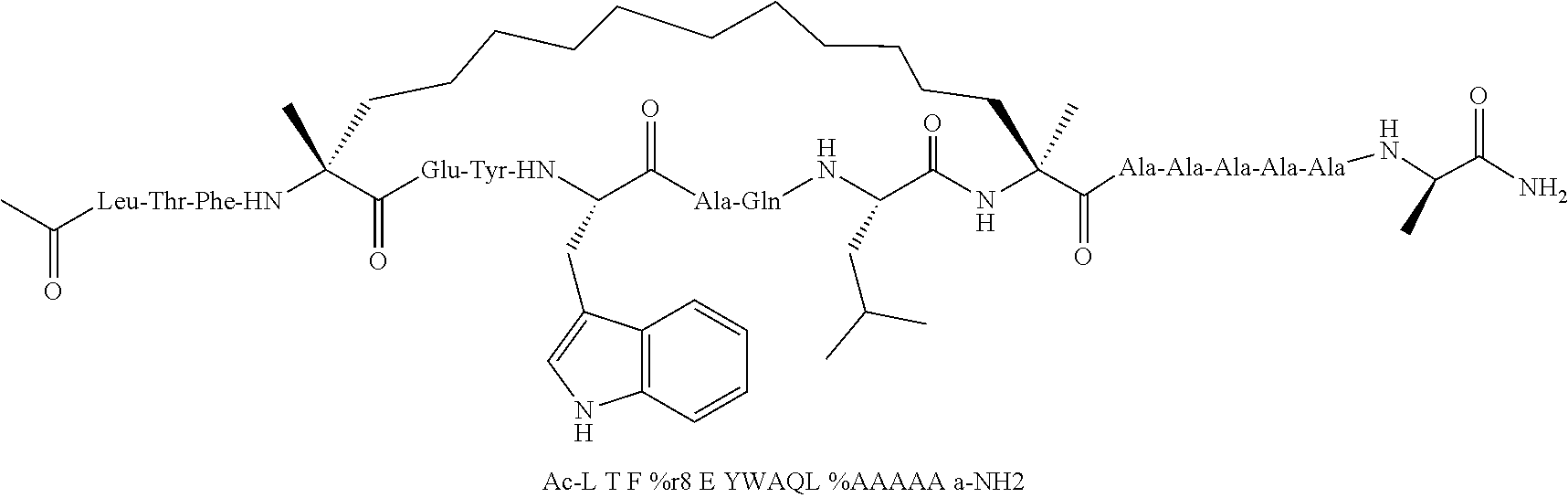


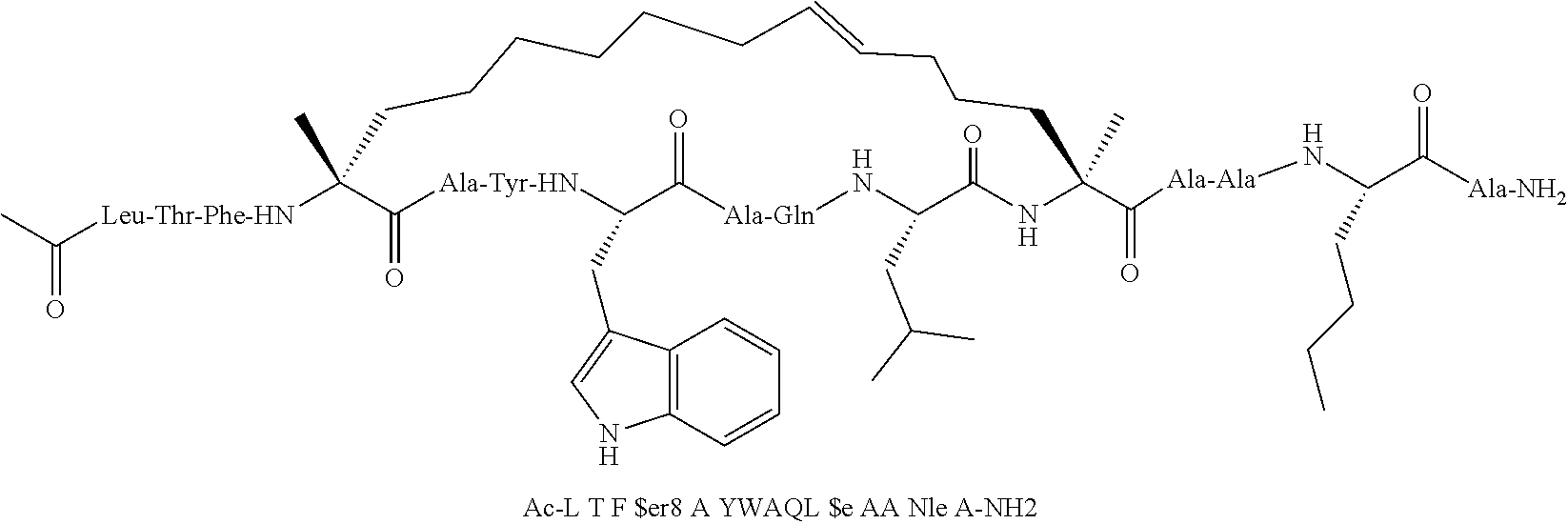






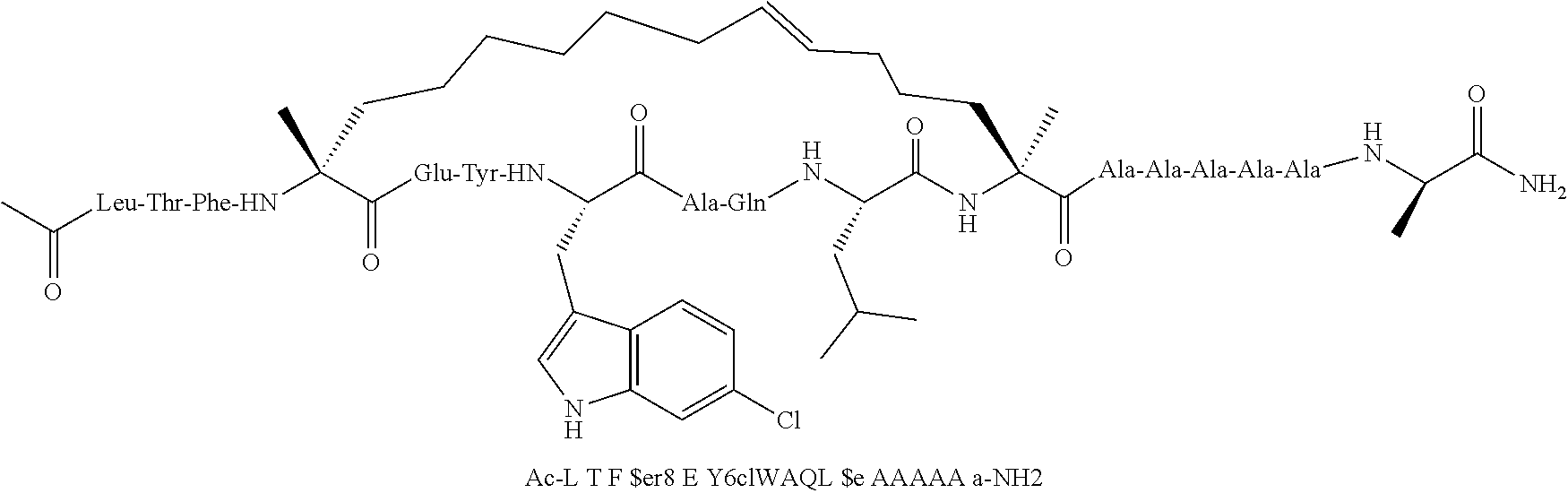
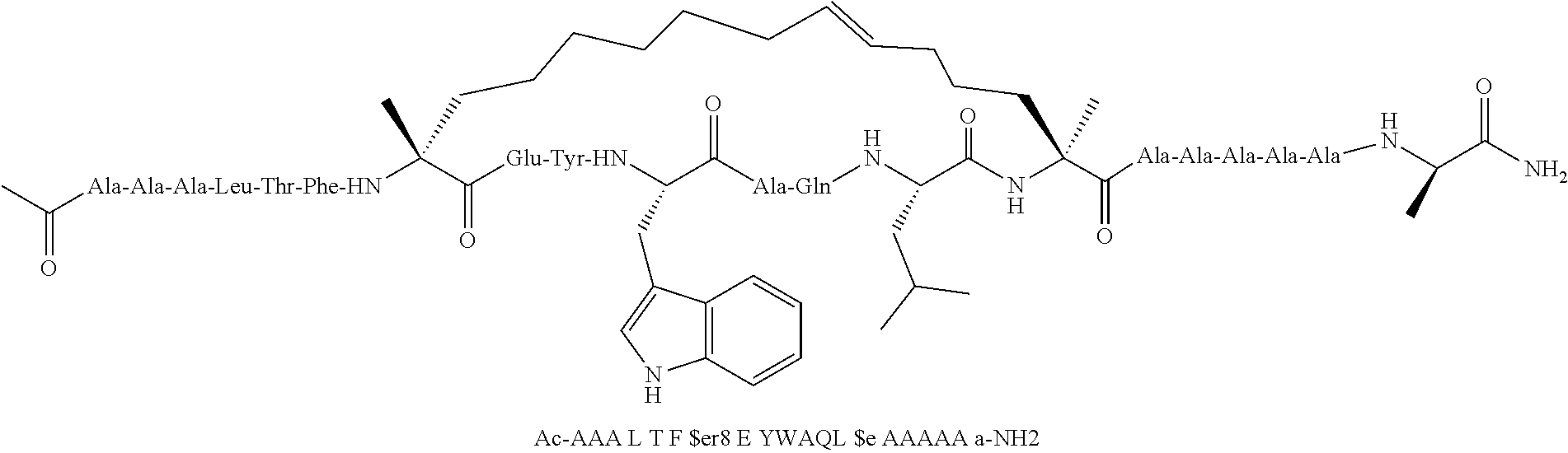






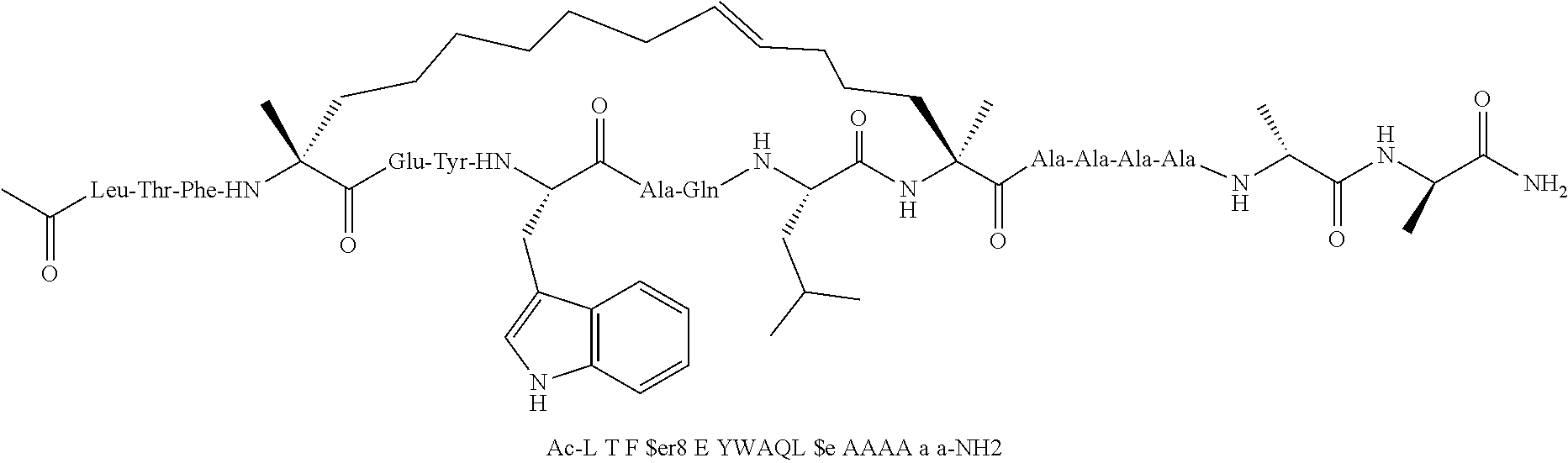

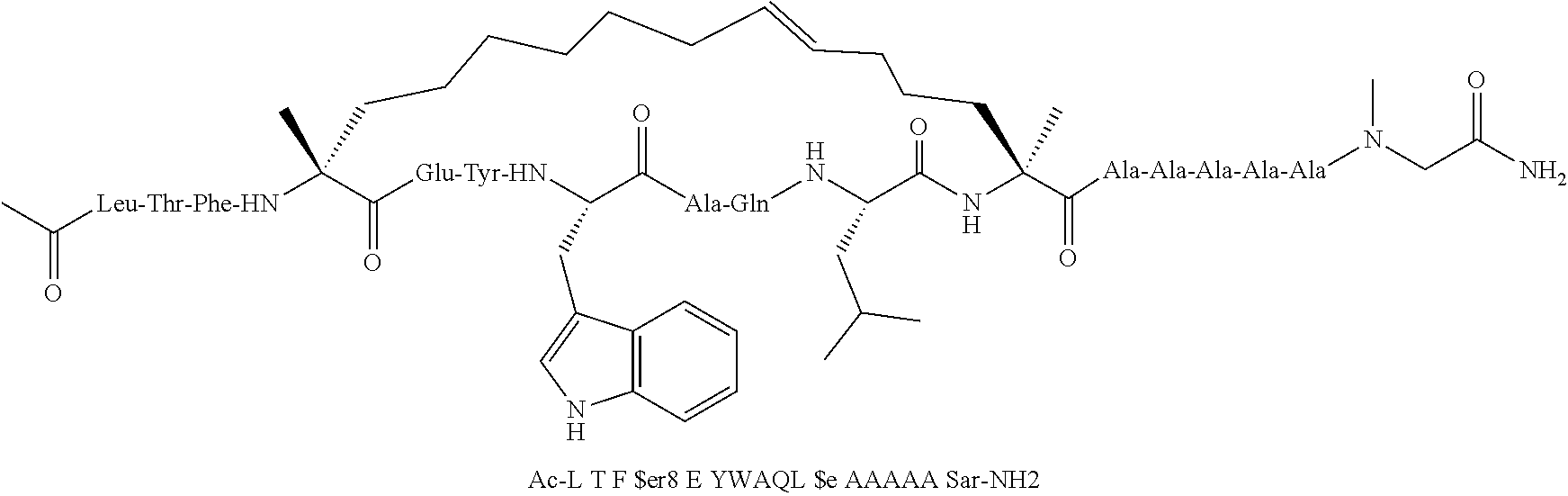





D00001

D00002
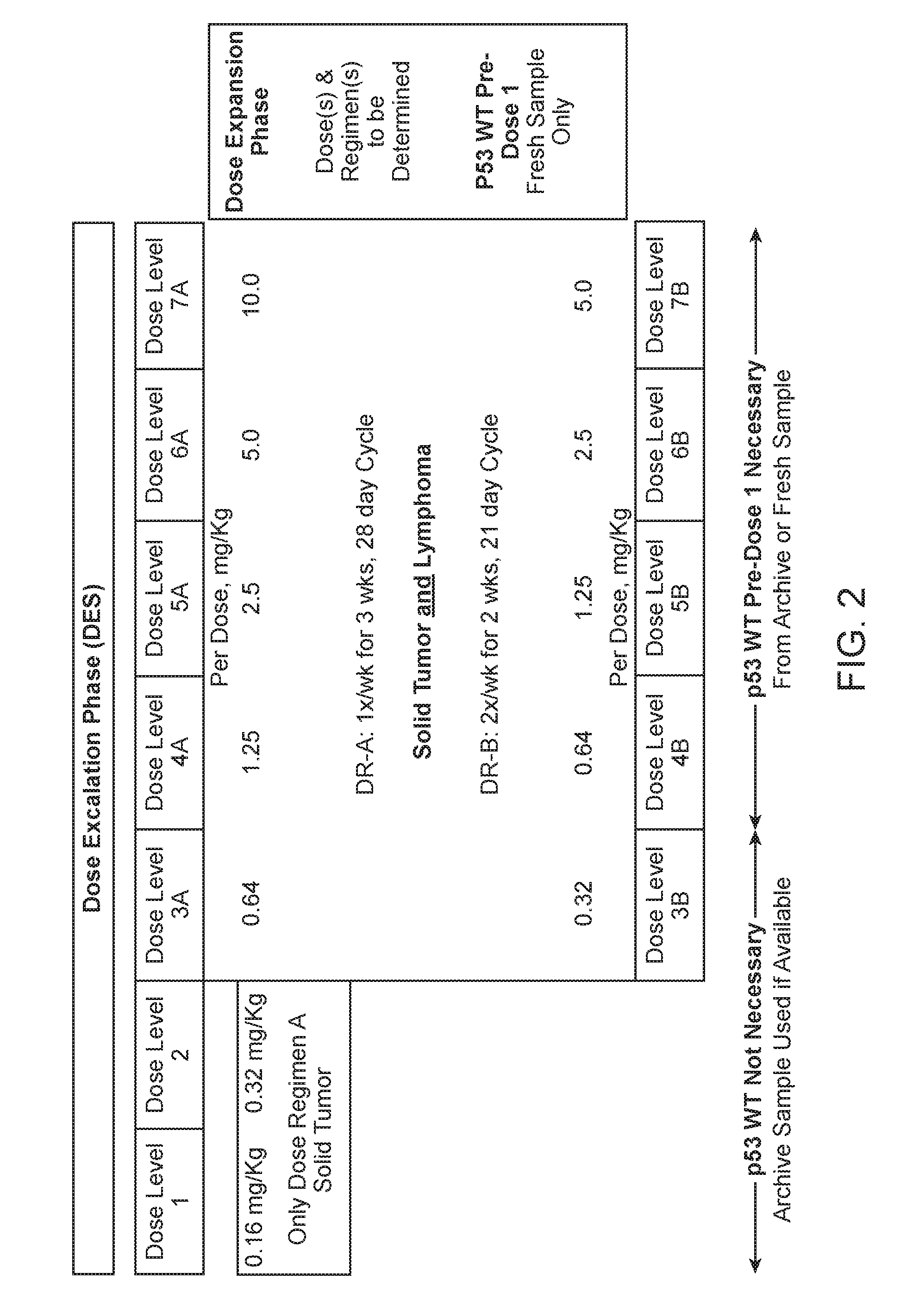
D00003
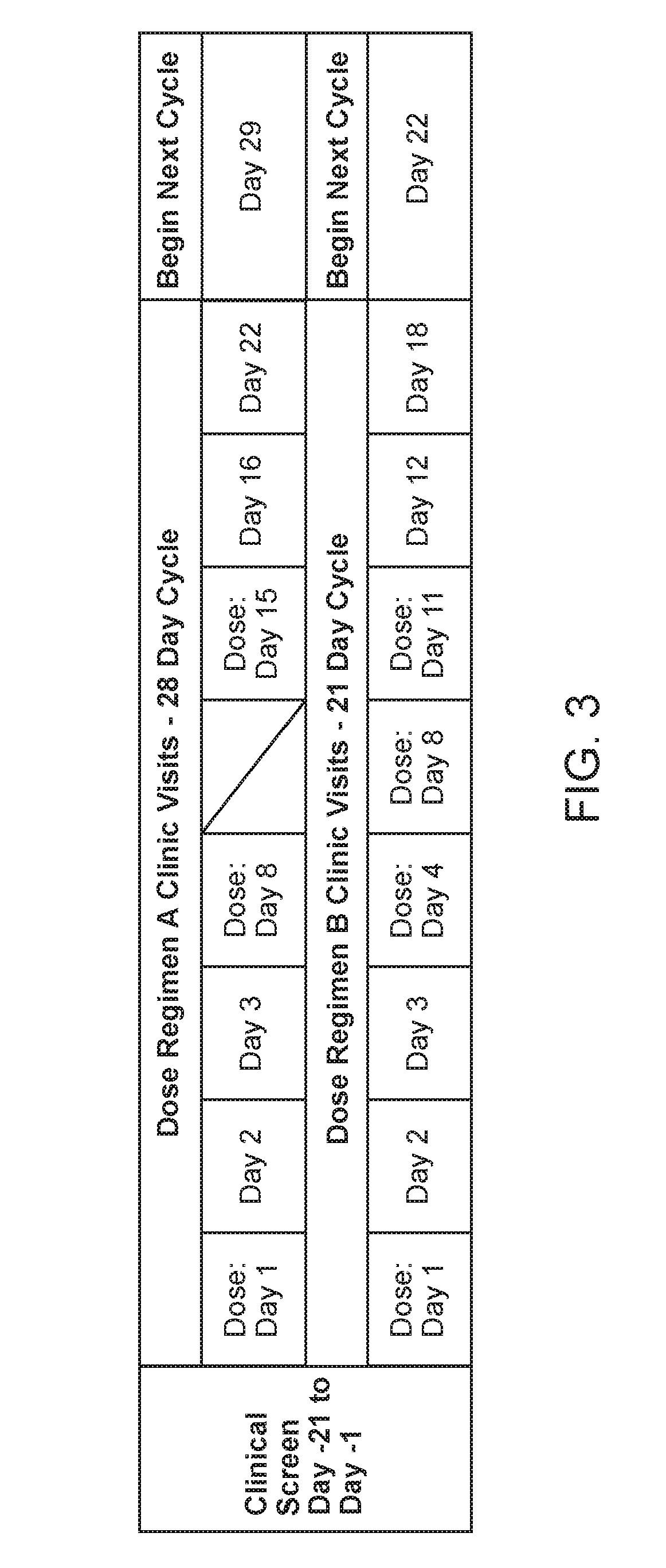
D00004
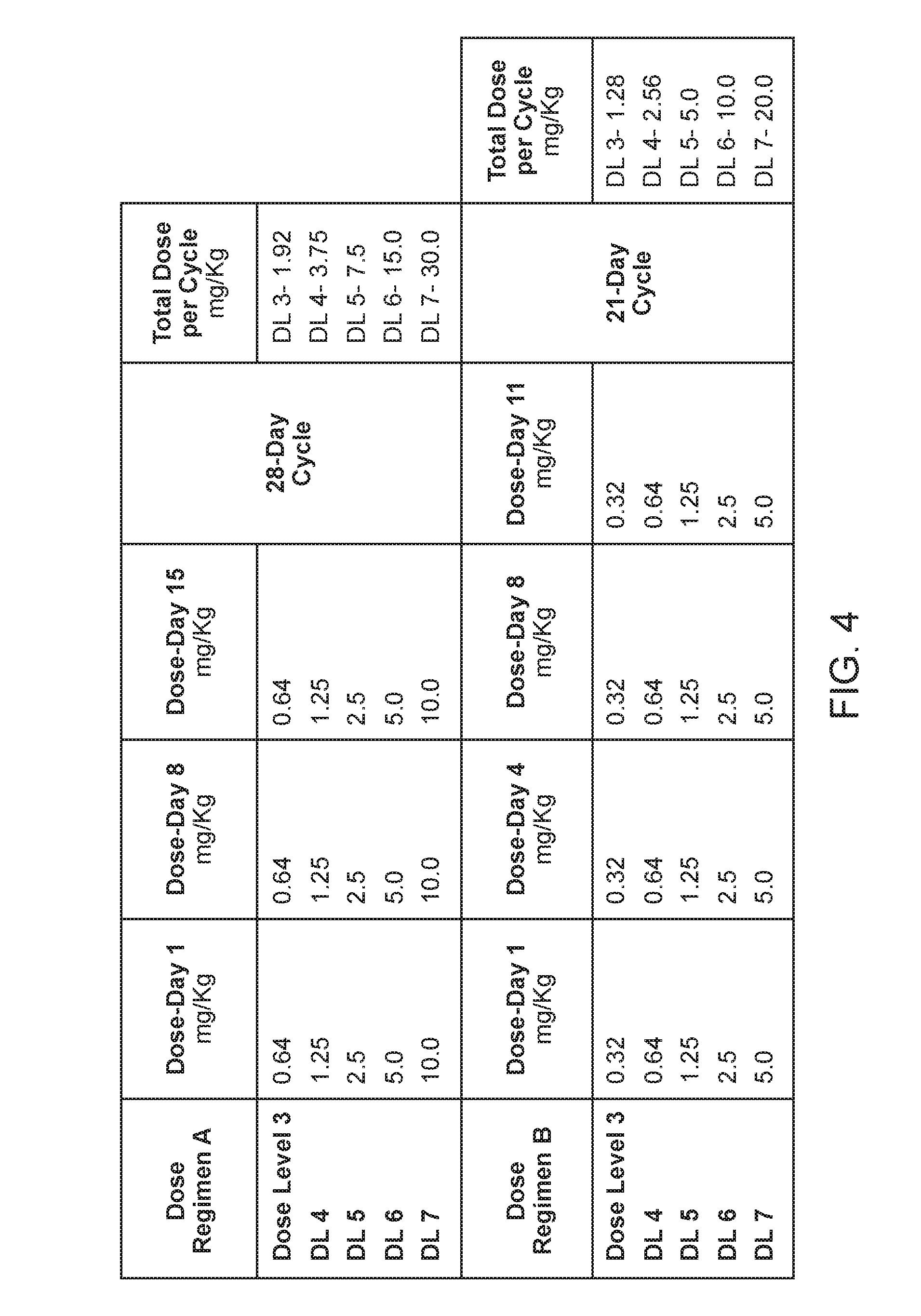
D00005

D00006
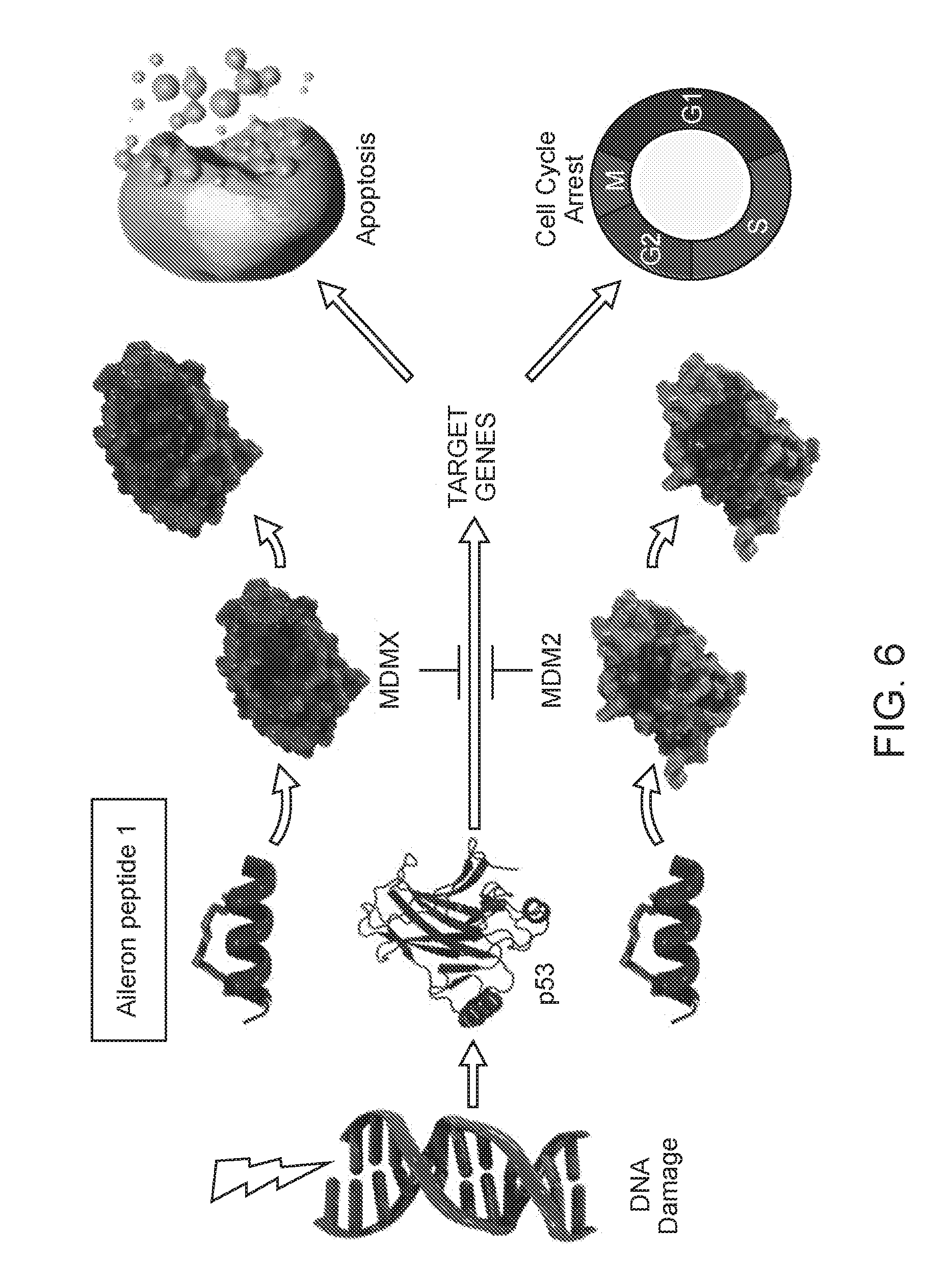
D00007
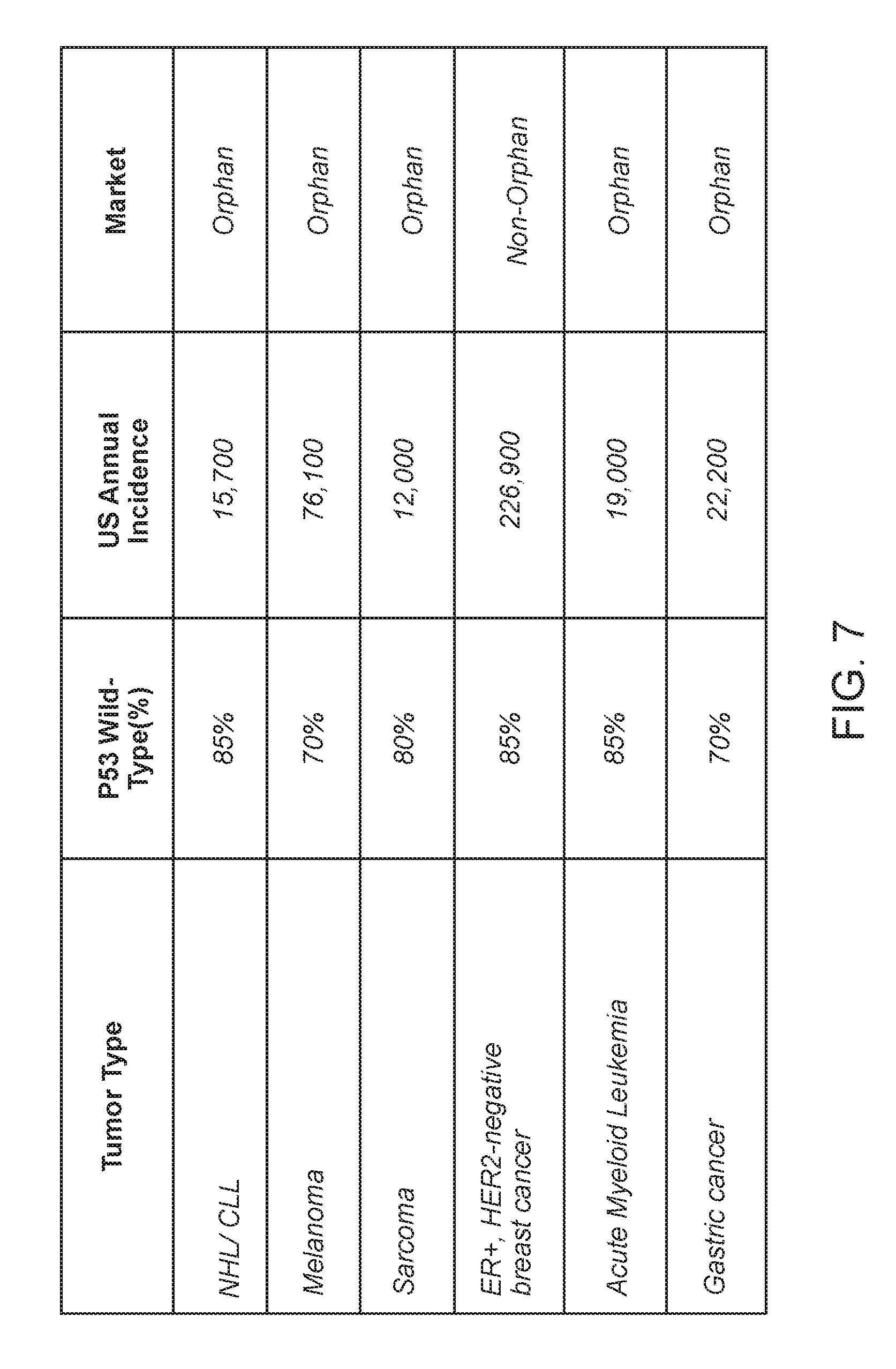
D00008

D00009
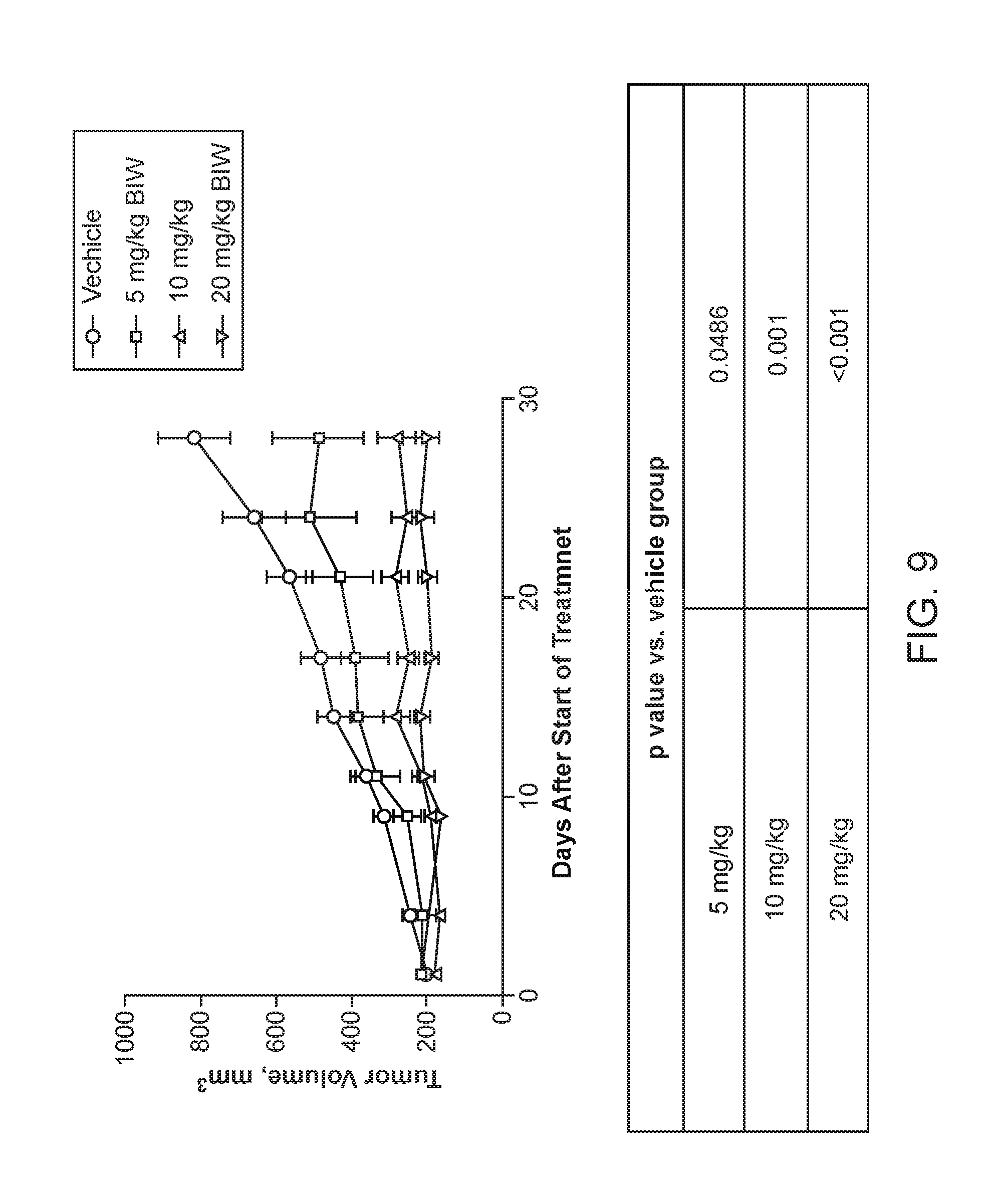
D00010

D00011

D00012

D00013
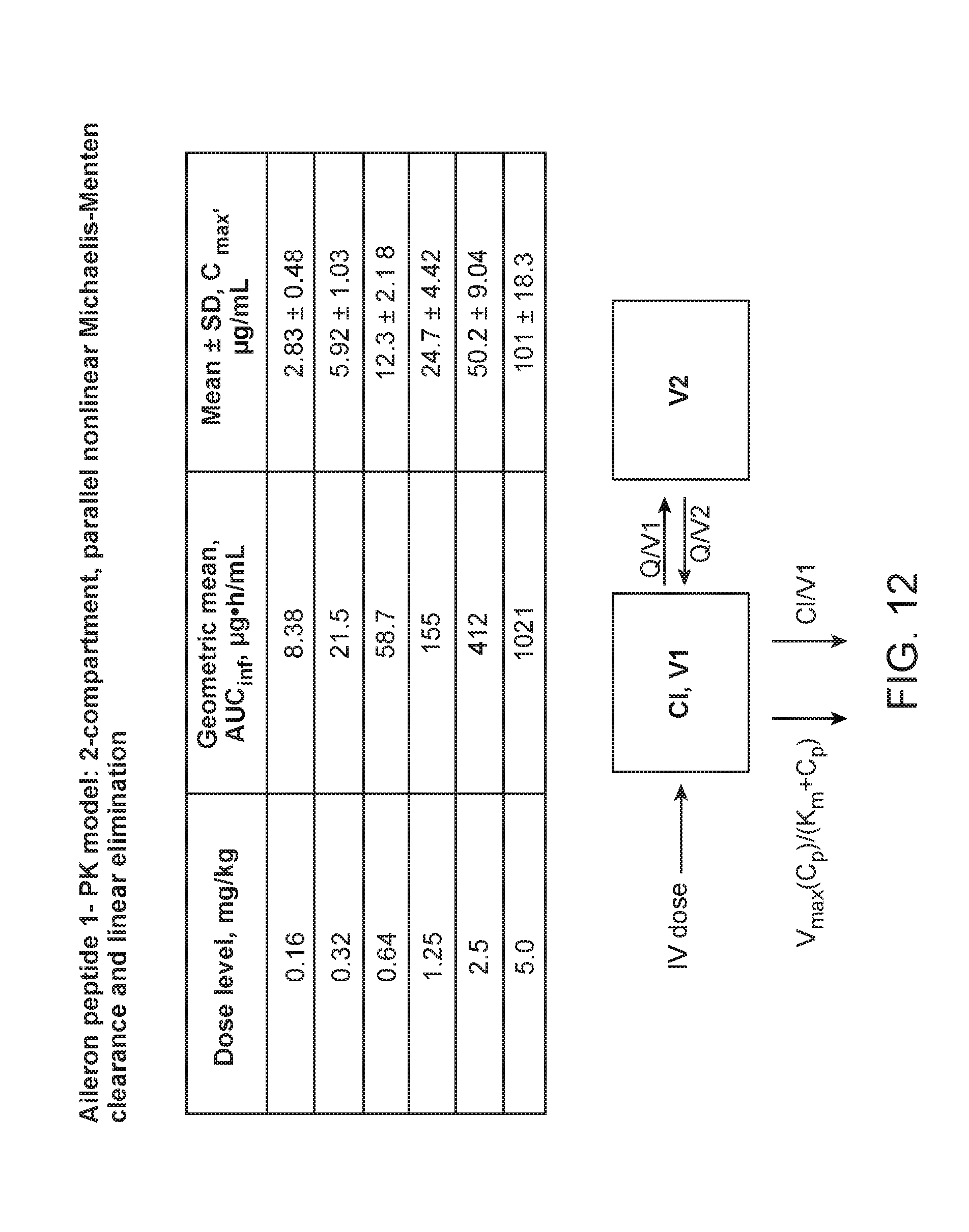
D00014

D00015

D00016


P00001
P00002
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.