Pharmaceutical-grade Ferric Organic Compounds, Uses Thereof And Methods Of Making Same
CHAN; Keith ; et al.
U.S. patent application number 16/352455 was filed with the patent office on 2019-09-05 for pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same. The applicant listed for this patent is Panion & BF Biotech Inc.. Invention is credited to Keith CHAN, David W.K. Kwok, Nikolay Mintchev Stoynov, Winston Town.
| Application Number | 20190269722 16/352455 |
| Document ID | / |
| Family ID | 67767892 |
| Filed Date | 2019-09-05 |









View All Diagrams
| United States Patent Application | 20190269722 |
| Kind Code | A1 |
| CHAN; Keith ; et al. | September 5, 2019 |
PHARMACEUTICAL-GRADE FERRIC ORGANIC COMPOUNDS, USES THEREOF AND METHODS OF MAKING SAME
Abstract
The present invention discloses pharmaceutical-grade ferric organic compounds, including ferric citrate, which are soluble over a wider range of pH, and which have a large active surface area. A manufacturing and quality control process for making a pharmaceutical-grade ferric citrate that consistently complies with the established Manufacture Release Specification is also disclosed. The pharmaceutical-grade ferric organic compounds are suitable for treating disorders characterized by elevated serum phosphate levels.
| Inventors: | CHAN; Keith; (Rockville, MD) ; Town; Winston; (Hong Kong, CN) ; Kwok; David W.K.; (Vancouver, CA) ; Stoynov; Nikolay Mintchev; (Vancouver, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 67767892 | ||||||||||
| Appl. No.: | 16/352455 | ||||||||||
| Filed: | March 13, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15699025 | Sep 8, 2017 | |||
| 16352455 | ||||
| 14701933 | May 1, 2015 | 9757416 | ||
| 15699025 | ||||
| 14306756 | Jun 17, 2014 | 9050316 | ||
| 14701933 | ||||
| 13661558 | Oct 26, 2012 | 8754257 | ||
| 14306756 | ||||
| 13289048 | Nov 4, 2011 | 8299298 | ||
| 13661558 | ||||
| 12064058 | Feb 18, 2008 | 8093423 | ||
| PCT/US2006/032385 | Aug 18, 2006 | |||
| 13289048 | ||||
| 11206981 | Aug 18, 2005 | 7767851 | ||
| 12064058 | ||||
| PCT/US2004/004646 | Feb 18, 2004 | |||
| 11206981 | ||||
| 60709511 | Aug 19, 2005 | |||
| 60462684 | Apr 15, 2003 | |||
| 60447690 | Feb 19, 2003 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07C 51/412 20130101; C07C 51/418 20130101; C07C 59/265 20130101; A61K 31/295 20130101; A61K 33/26 20130101 |
| International Class: | A61K 33/26 20060101 A61K033/26; C07C 59/265 20060101 C07C059/265; A61K 31/295 20060101 A61K031/295; C07C 51/41 20060101 C07C051/41 |
Claims
1-57. (canceled)
58. A method of treating hyperphosphatemia, comprising administering one or more tablets comprising ferric citrate and a pharmaceutically acceptable carrier to an adult subject, wherein: (a) the subject has chronic kidney disease and is undergoing dialysis, (b) the tablets are prepared from a form of ferric citrate having a BET active surface area of at least about 16 m.sup.2/g, and (c) the tablets are administered to provide a dose between 4 and 15 grams of ferric citrate to the subject per day.
59. The method of claim 58, wherein the chronic kidney disease is selected from mid-stage kidney disease and late-stage kidney disease.
60. The method of claim 58, wherein the dialysis is selected from hemodialysis and peritoneal dialysis.
61. The method of claim 58, wherein the tablets are administered to provide a dose of 6 to 12 grams of the ferric citrate to the subject per day.
62. The method of claim 58, wherein the tablets are administered to provide a dose of 6 grams of the ferric citrate to the subject per day.
63. The method of claim 58, wherein the tablets are administered twice, or three times per day.
64. The method of claim 58, wherein the dose of the ferric citrate is adjusted based on serum phosphorus levels of the subject.
Description
[0001] This application is a Continuation of U.S. Ser. No. 14/306,756, filed Jun. 17, 2014, which is a Continuation of U.S. Ser. No. 13/661,558, filed Oct. 26, 2012, which is a Continuation of U.S. Ser. No. 13/289,048, filed Nov. 4, 2011, which is a Continuation of U.S. Ser. No. 12/064,058, filed Feb. 18, 2008, which is a National Stage of International Application No. PCT/US2006/032385, filed Aug. 18, 2006, which claims the benefit of U.S. Ser. No. 60/709,511, filed Aug. 19, 2005, and is a Continuation-in-part of U.S. Ser. No. 11/206,981, filed Aug. 18, 2005, which is a Continuation-in-part of Int'l App'l No. PCT/US2004/004646, filed Feb. 18, 2004, which claims the benefit of U.S. Ser. No. 60/462,684, filed Apr. 15, 2003, and U.S. Ser. No. 60/447,690, filed Feb. 19, 2003. The entire contents and disclosures of the preceding applications are incorporated herein by reference into this application.
[0002] Throughout this application, various publications are referenced. Disclosures of these publications in their entireties are hereby incorporated by reference into this application to more fully describe the state of the art to which this invention pertains.
TECHNICAL FIELD
[0003] This invention relates to the preparation and use of pharmaceutical-grade ferric organic compounds, such as ferric citrate. Uses of the pharmaceutical-grade ferric citrate described herein, include, but are not limited to methods of treating various disorders in humans and non-human subjects or patients.
BACKGROUND OF THE INVENTION
Uses of Iron Compounds
[0004] Ferric iron containing compounds are useful in the treatment of a number of disorders, including, but not limited to, hyperphosphatemia and metabolic acidosis. See Hsu et al., "New Phosphate Binding Agents: Ferric Compounds", J Am Soc Nephrol, Vol. 10, Pages 1274-1280, 1999. Previous studies and inventions have reported the use of ferric compounds in binding with dietary phosphates, and such ferric compounds are potentially useful for the treatment of hyperphosphatemia in renal failure patients (U.S. Pat. No. 5,753,706, 1998; U.S. Pat. No. 6,903,235, 2005; CN 1315174, 2001; Yang W. C., et al., Nephrol. Dial. Transplant 17:265:270 (2002)). Elevated amounts of phosphate in the blood can be removed by administering compounds such as ferric citrate. Once in solution, the ferric iron binds phosphate, and the ferric phosphate compounds precipitate in the gastrointestinal tract, resulting in effective removal of dietary phosphate from the body. It is also believed that the absorbed citrate from ferric citrate is converted to bicarbonate which corrects metabolic acidosis, a condition common in renal failure patients.
[0005] Int'l App. No. PCT/US2004/004646, filed Feb. 18, 2004, published under Int'l Publication No. WO2004/074444 on Sep. 2, 2004, discloses a method of preparing ferric organic compounds, including ferric citrate that remains soluble over a wider range of pH than previously described preparations, and which have a large active surface area. However, commercially scalable manufacturing processes with quality control/analysis measures to ensure and/or to verify the compliance of the pharmaceutical-grade ferric citrate or ferric organic compounds with established standards or specifications were not previously disclosed.
[0006] Accordingly, there exists a need for a scalable process for synthesizing pharmaceutical-grade ferric organic compounds or ferric citrate for human use. The process needs to consistently produce ferric organic compounds or ferric citrate of the required pharmaceutical grade.
[0007] This invention further discloses dosage for ferric citrate for treating of a human or non-human subject or patient in need of the compound. Different routes of administration are explored.
SUMMARY OF THE INVENTION
[0008] In accordance with these and other objects of the invention, a brief summary of the present invention is presented. Some simplifications and omission may be made in the following summary, which is intended to highlight and introduce some aspects of the present invention, but not to limit its scope. Detailed descriptions of a preferred exemplary embodiment adequate to allow those of ordinary skill in the art to make and use the invention concepts will follow in later sections.
[0009] An embodiment of the invention provides a manufacturing and quality control process/analysis for making a pharmaceutical-grade ferric citrate that consistently complies with the established Manufacture Release Specification. The process of the present invention can be adapted to produce multi-kilogram batches of pharmaceutical-grade ferric citrate, and can be readily scaled up to provide additional manufacturing capacity for ferric citrate.
[0010] An illustrative embodiment of the manufacturing method may be exemplified by the following non-limiting sequence of steps for preparing pharmaceutical-grade ferric citrate comprising the steps of: (a) dissolving an appropriate amount of Ferric chloride hexahydrate in water to form a Ferric chloride hexahydrate solution; (b) dissolving an appropriate amount of NaOH in water to form a NaOH solution; (c) mixing the Ferric chloride hexahydrate solution and NaOH solution to form a solution with Fe(OH).sub.3 precipitate; (d) maintaining the pH of the solution with Fe(OH).sub.3 precipitate above 7.0; (e) isolating the Fe(OH).sub.3 precipitate; (f) washing the Fe(OH).sub.3 precipitate three times with water; (g) suspending the Fe(OH).sub.3 precipitate in water; (h) adding citric acid to the Fe(OH).sub.3 precipitate to form a ferric-organic acid solution; (i) stirring and heating the ferric-organic acid solution at 90-100.degree. C. for 30 to 120 minutes; (j) removing solids in the ferric-organic acid solution by adding citric acid; (k) allowing the ferric-organic acid solution to cool to below 30.degree. C.; (l) maintaining the pH of the ferric-organic acid solution to between 0.8-1.5; (m) filtering the ferric-organic acid solution to obtain a liquid filtrate; (n) mixing acetone and liquid filtrate to form ferric citrate; (o) isolating ferric citrate; (p) washing ferric citrate with acetone three times; and (q) drying the ferric citrate.
[0011] A further embodiment provides a large-scale production scheme for pharmaceutical-grade ferric citrate comprising the steps of: (a) mixing an appropriate amount of NaOH and Ferric chloride hexahydrate in a suitable reactor to form a ferric hydroxide slurry with ferric hydroxide precipitate; (b) maintaining the pH of the ferric hydroxide slurry to above 7.0; (c) isolating the ferric hydroxide precipitate from the ferric hydroxide slurry using pressure filtration; (d) washing the ferric hydroxide precipitate three times; (e) maintaining the % Cl in the ferric hydroxide precipitate to below 5%; (f) isolating the washed ferric hydroxide precipitate using pressure filtration; (g) mixing citric acid with washed ferric hydroxide precipitate to form a ferric organic acid solution; (h) stirring and maintaining the temperature of the ferric organic acid solution at 80.+-.5.degree. C. for 2 hours; (i) allowing the ferric organic acid solution to cool to 60.degree. C.; (j) maintaining the pH of the ferric organic acid solution to between 0.8 to 1.5 and the amount of Fe in the ferric organic acid solution to .gtoreq.85% of Fe added in step (a); (k) filtering the ferric organic acid solution using pressure filtration to obtain a liquid filtrate; (l) mixing the liquid filtrate with acetone to obtain ferric citrate; (m) isolating ferric citrate using pressure filtration; (n) washing the ferric citrate with acetone; (o) isolating the washed ferric citrate using pressure filtration; (p) drying the washed ferric citrate in fluidized bed dryer; and (q) maintaining the organic volatile impurities to .ltoreq.1000 ppm acetone.
[0012] A further embodiment encompasses various intermediate compositions that may be useful in the preparation of the pharmaceutical-grade ferric citrate. The intermediate compositions encompassed herein include solids, liquids or multiphase mixtures. A liquid intermediate composition comprising the pharmaceutical-grade ferric citrate may be an aqueous composition or an organic solvent-based composition. A multiphase composition may encompass both aqueous and organic phases.
[0013] An additional embodiment encompasses the methods of storing, packaging and using the various intermediate compositions disclosed herein.
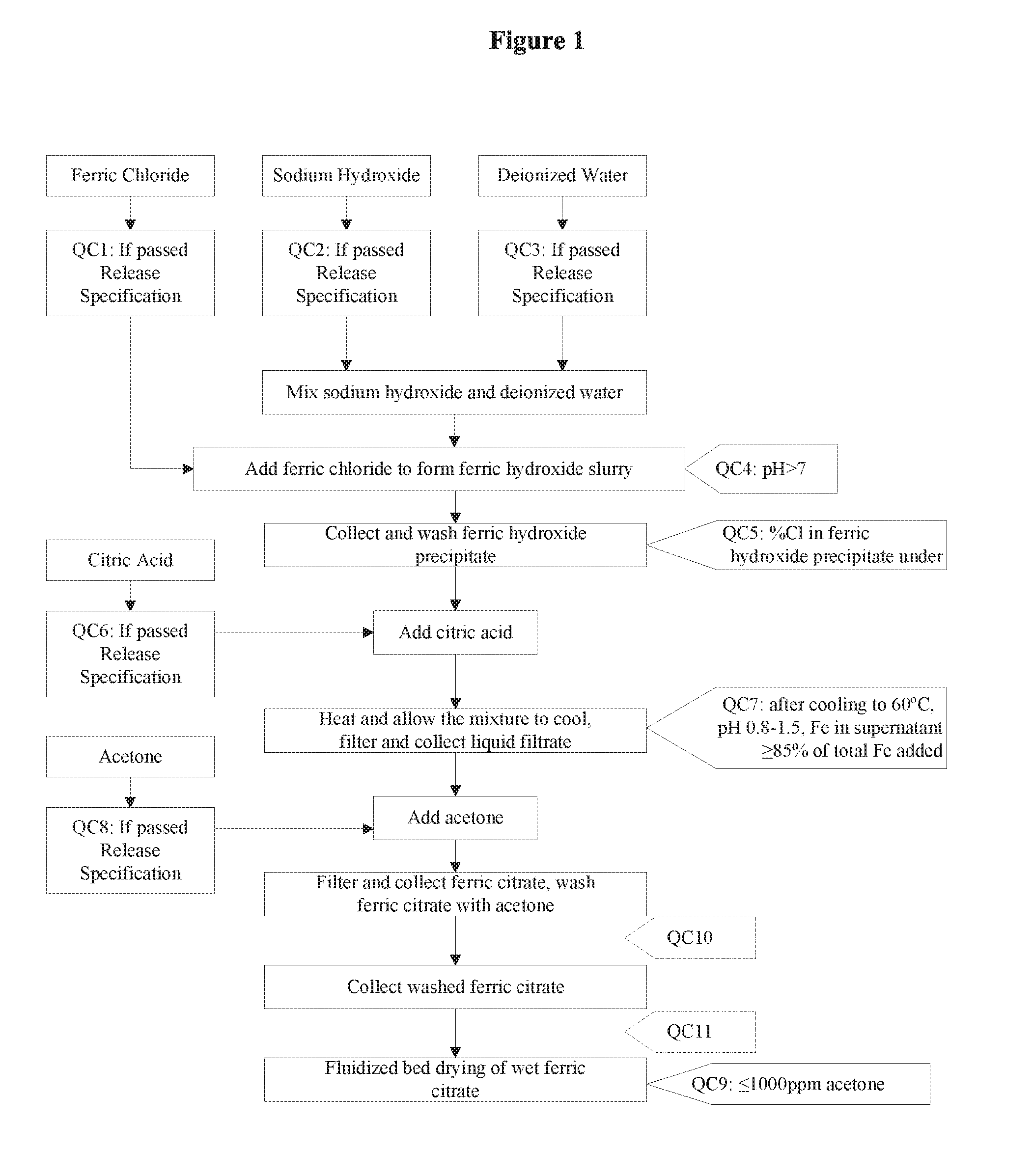
[0014] An additional embodiment provides pharmaceutically useful compositions comprising the pharmaceutical-grade ferric citrate. The pharmaceutically useful composition further comprises any pharmaceutically acceptable carrier, adjuvant, filler or delivery vehicle suitable for administering to a subject or human patient, an effective amount of the pharmaceutical-grade ferric citrate.
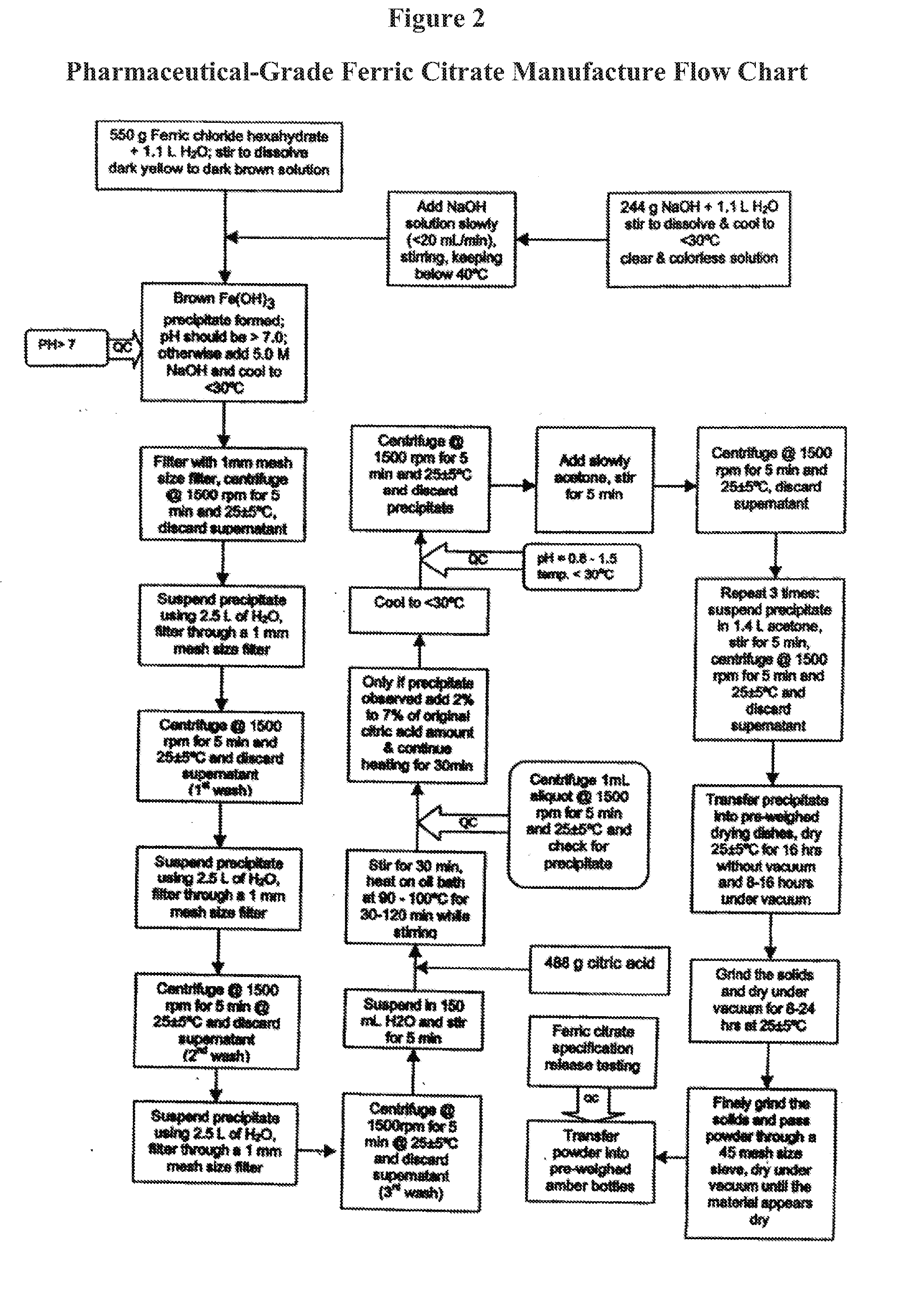
[0015] Further embodiments of the pharmaceutical compositions include, but are not limited to solids, liquids, or semi-solid forms, such as gels, syrups, chewables or pastes.
[0016] Within the scope of the methods of using the pharmaceutically useful compositions disclosed herein are effective doses of the pharmaceutical compositions, in addition to the timing and modes of administering the pharmaceutical compositions.
[0017] A non-limiting example of a method for using the pharmaceutical compositions encompasses treating disorders resulting from elevated blood levels of phosphates, i.e., hyperphosphateimia, in a subject or a human patient. Such disorders are exemplified by, but not limited to, renal failure or the progression of renal failure, mineralization of soft tissues, hyperparathyroidism as well as other complications.
[0018] An embodiment encompassed by the invention includes a pharmaceutical composition comprising:
(a) an amount of a pharmaceutical-grade ferric organic compound effective to achieve a decrease in the serum phosphate level of a subject or patient; (b) a pharmaceutically suitable carrier; and wherein the ferric organic compound complies with at least one of the limits in the manufacture release specification in Table A.
[0019] The pharmaceutical composition described herein may be prepared by a method shown in FIG. 1, 2, 3 or 4. Further to these embodiments are compositions prepared with materials satisfying at least one limit disclosed in each of the release specifications of either of the relevant Tables B to F.
[0020] An additional embodiment encompasses compositions of pharmaceutical-grade ferric citrate, prepared according to methods comprising the steps of: [0021] (a) dissolving an appropriate amount of ferric chloride hexahydrate in water to form a ferric chloride hexahydrate solution; [0022] (b) dissolving an appropriate amount of NaOH in water to form a NaOH solution; [0023] (c) mixing the ferric chloride hexahydrate solution and NaOH solution to form a solution with Fe(OH)3 precipitate; [0024] (d) maintaining the pH of the solution with Fe(OH).sub.3 precipitate above 7.0; [0025] (e) isolating the Fe(OH).sub.3 precipitate; [0026] (f) washing the Fe(OH).sub.3 precipitate three times with water; [0027] (g) suspending the Fe(OH).sub.3 precipitate in water; [0028] (h) adding citric acid to the Fe(OH).sub.3 precipitate to form a ferric-organic acid solution; [0029] (i) stirring and heating the ferric-organic acid solution at 90-100.degree. C. for 30 to 120 minutes; [0030] (j) removing solids in the ferric-organic acid solution by adding citric acid; [0031] (k) allowing the ferric-organic acid solution to cool to below 30.degree. C.; [0032] (l) maintaining the pH of the ferric-organic acid solution to between 0.8-1.5; [0033] (m) filtering the ferric-organic acid solution to obtain a liquid filtrate; [0034] (n) mixing acetone and liquid filtrate to form ferric citrate; [0035] (o) isolating ferric citrate; [0036] (p) washing ferric citrate with acetone three times; [0037] (q) drying the ferric citrate; and [0038] (r) performing quality control tests as shown in FIG. 1, 2 or 3.
[0039] It is advantageous to scale-up the method of preparation. Thus, an industrial-scale method is embodied by a method for large-scale production of pharmaceutical-grade ferric citrate comprising the steps of: [0040] (a) mixing an appropriate amount of NaOH and Ferric chloride hexahydrate in a suitable reactor to form a ferric hydroxide slurry with ferric hydroxide precipitate; [0041] (b) maintaining the pH of the ferric hydroxide slurry to above 7.0; [0042] (c) isolating the ferric hydroxide precipitate from the ferric hydroxide slurry using pressure filtration; [0043] (d) washing the ferric hydroxide precipitate three times; [0044] (e) maintaining the % Cl in the ferric hydroxide precipitate to below 5%; [0045] (f) isolating the washed ferric hydroxide precipitate using pressure filtration; [0046] (g) mixing citric acid with washed ferric hydroxide precipitate to form a ferric organic acid solution; [0047] (h) stirring and maintaining the temperature of the ferric organic acid solution at 80.+-.5.degree. C. for 2 hours; [0048] (i) allowing the ferric organic acid solution to cool to 60.degree. C.; [0049] (j) maintaining the pH of the ferric organic acid solution to between 0.8 to 1.5 and the amount of Fe in the ferric organic acid solution to .gtoreq.85% of Fe added in step (a); [0050] (k) filtering the ferric organic acid solution using pressure filtration to obtain a liquid filtrate; [0051] (l) mixing the liquid filtrate with acetone to obtain ferric citrate; [0052] (m) isolating ferric citrate using pressure filtration; [0053] (n) washing the ferric citrate with acetone; [0054] (o) isolating the washed ferric citrate using pressure filtration; [0055] (p) drying the washed ferric citrate in fluidized bed dryer; and [0056] (q) maintaining the organic volatile impurities to .ltoreq.1000 ppm acetone; [0057] and wherein the ferric citrate complies with at least one of the limits in the manufacture release specification in Table A.
[0058] The compositions encompassing pharmaceutical grade ferric citrate are suitable for treating hyperphosphatemia, or other disorders characterized by high serum phosphate levels. Therefore, the invention encompasses treating subjects or patients with various renal diseases; e.g., End Stage Renal Diseases (ESRD), Chronic Kidney Disease (CKD) or other relate kidney diseases, or subjects and patients who are on dialysis but not limited to hemodialysis.
[0059] In another embodiment, the compositions encompassing pharmaceutical grade ferric citrate may used to treat subjects or patients with metabolic acidosis. Other disorders that may be ameliorated by the conversion of citrate to bicarbonate are also encompassed by the invention described.
[0060] An embodiment of a method for using the pharmaceutical composition encompasses treating a human or non-human subject or patient with chronic kidney disease. There are generally five clinical stages of chronic kidney disease, numbered 1 to 5, wherein stage 1 is the least severe and stage 5 the most severe. In the early stages, e.g., stages 1 and 2, dialysis is not required. As chronic kidney disease progresses to stage 5, a patient may require dialysis treatment three times per week. It should be noted that elevated phosphate levels are observed at all stages of chronic kidney disease. Therefore, an embodiment of the invention is a method of treating a subject or person with early or mid-stage chronic kidney disease, with a composition comprising pharmaceutical-grade ferric citrate in order to achieve a lower serum phosphate level.
[0061] It is a further embodiment of the invention to provide a method of treating a human or non-human subject or patient with late-stage chronic kidney disease who undergo hemodialysis, by administering a composition comprising pharmaceutical-grade ferric citrate. It is known that hemodialysis is not sufficiently effective in reducing serum phosphate level. The treatment of a subject or person with late stage kidney disease is applicable whether or not the subject or person is currently undergoing hemodialysis treatment.
[0062] An additional embodiment of the invention is a method of treating a subject or person with chronic kidney disease and undergoing peritoneal dialysis with the pharmaceutical-grade ferric citrate-containing compositions described. It is known that peritoneal dialysis is not sufficiently effective in reducing serum phosphate levels.
[0063] An additional embodiment is a method for using the pharmaceutical composition to inhibit or even reverse soft tissue mineralization, specifically calcification. Hyperphosphatemia may lead to increased calcium phosphate deposition in hard and soft tissues by increasing the likelihood of binding with free calcium to form insoluble calcium phosphate. Therefore, an effective dose of a composition comprising pharmaceutical-grade ferric citrate may decrease serum phosphate levels and result in a corresponding decrease in calcium phosphate deposition.
[0064] It is noteworthy that whereas ferric ion forms insoluble precipitates with phosphate-containing compounds in the lumen of the gastroinstestinal tract, the citrate component is absorbed and functions as a calcium chelator. Because chelated calcium is not available for calcium phosphate formation, administering pharmaceutical-grade ferric citrate decreases may lead to reductions in both serum calcium and phosphate. This may also be expressed as leading to a decrease in the serum calcium-phosphate product. Reducing serum calcium and phosphate would be expected to reduce calcium phosphate deposition. The end result is reversing, i.e., solubilizing or dissolving, the deposited calcium phosphate.
[0065] The decalcifying of a calcified soft tissue, e.g., the sclera of the eye, may be achieved by administering pharmaceutical-grade ferric citrate. It is known among persons of ordinary skill in the relevant medical arts that patients with kidney disease receiving doses of ferric citrate have shown reversal of calcium deposits on the eye. Therefore, an embodiment of the invention is directed to the decalcification of soft tissue such as the eye.
[0066] Kidney stones comprise calcium salts of oxalic acid or phosphates and are formed by mechanisms similar to those described above. Thus, pharmaceutical-grade ferric citrate, in another embodiment of the invention provides a method of treating kidney stones, i.e., renal calculi, by promoting their dissolution.
[0067] The compositions encompassing pharmaceutical grade ferric citrate may operate according to more than one mechanism. A plausible non-limiting mechanism of action may result from the ferric ion binding phosphate in the GI tract, thus forming an insoluble ferric phosphate precipitate. This, in turn, may result in decrease the uptake of phosphate and phosphate-containing compounds from the GI tract.
[0068] In view of such a mechanism, the administering of compositions encompassing pharmaceutical grade ferric citrate via an oral route is encompassed by the inventive methods described.
[0069] This invention provides a method of reversing, preventing or stabilizing soft tissue calcification of a subject, comprising administering to said subject and effective amount of a ferric citrate compound.
[0070] This invention further provides a method of reversing, preventing or stabilizing soft tissue calcification of a subject, comprising administering to said subject and effective amount of a ferric citrate compound, wherein the ferric citrate compound is prepared according to a method as shown in FIGS. 1-4.
[0071] This invention provides a method of reversing, preventing or stabilizing soft tissue calcification of a subject, comprising administering to said subject and effective amount of a ferric citrate compound, wherein the ferric citrate compound is prepared according a method comprising the steps of:
[0072] An embodiment of the invention encompasses tolerable doses of up to 15 grams per day for ferric citrate capsules and 30 grams per day for ferric citrate tablets.
[0073] The compositions encompassing pharmaceutical grade ferric citrate may be administered for varying periods of time. In some embodiments, the tolerability of the compositions encompassing pharmaceutical grade ferric citrate allows for long term administration when necessary.
DETAILED DESCRIPTION OF THE FIGURES
[0074] In drawings which illustrate specific embodiments of the invention, but which should not be construed as restricting the spirit or scope of the invention in any way:
[0075] FIG. 1 is a schematic diagram outlining the general method for synthesis of pharmaceutical-grade ferric citrate including in-process quality control measures to ensure the final ferric citrate product complies with the established Manufacture Release Specification as shown in Table A.
[0076] FIG. 2 is a schematic diagram outlining a method of making pharmaceutical-grade ferric citrate according to the invention.
[0077] FIG. 3 shows an overview of scalable pharmaceutical-grade ferric citrate manufacturing and quality control process according to the invention.
[0078] FIG. 4 is a schematic diagram of scalable pharmaceutical-grade ferric citrate manufacturing and quality control process according to the invention.
[0079] FIG. 5 shows the representative proton NMR spectrum for a pharmaceutical-grade ferric citrate according to the invention.
[0080] FIG. 6 shows the representative FTIR spectrum for pharmaceutical-grade ferric citrate according to the invention.
[0081] FIG. 7 is a comparison of intrinsic dissolution rates at pH 1.0 and 8.0 for pharmaceutical grade and chemical grade ferric citrate.
[0082] FIG. 8 is a schematic diagram outlining the general method for synthesis of pharmaceutical-grade ferric organic compound including in-process quality control measures to ensure the final ferric organic compound complies with established Manufacture Release Specification.
[0083] FIG. 9 is a summary of clinical results for pharmaceutical-grade ferric citrate
[0084] FIG. 10 is a summary of the efficacy data for pharmaceutical-grade ferric citrate
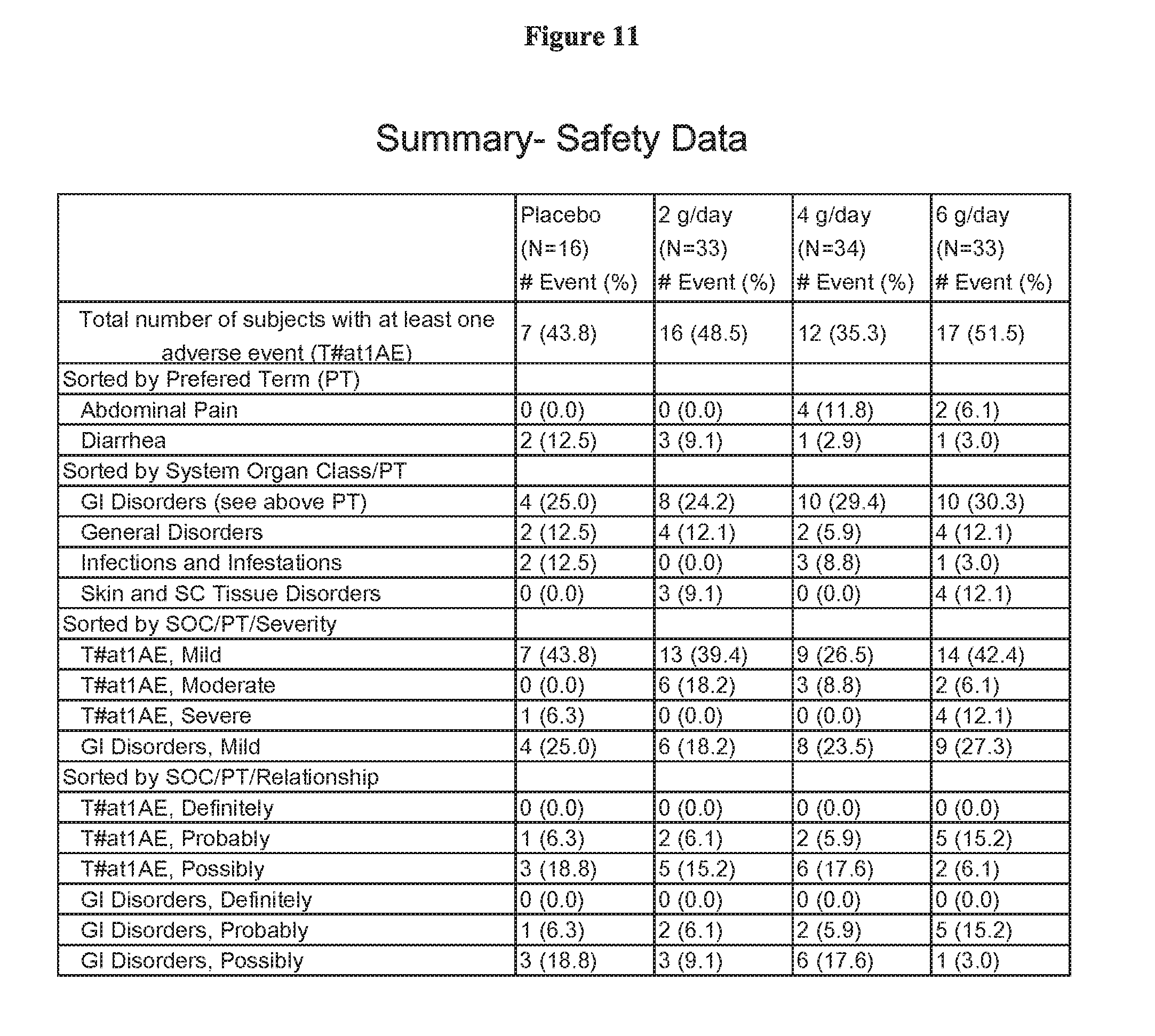
[0085] FIG. 11 is a summary of the safety data from a clinical study
[0086] FIG. 12 is a comparison of the safety profiles of chemical grade and pharmaceutical grade ferric citrates
[0087] FIG. 13 is a comparison of the efficacy profiles of chemical grade and pharmaceutical grade ferric citrates
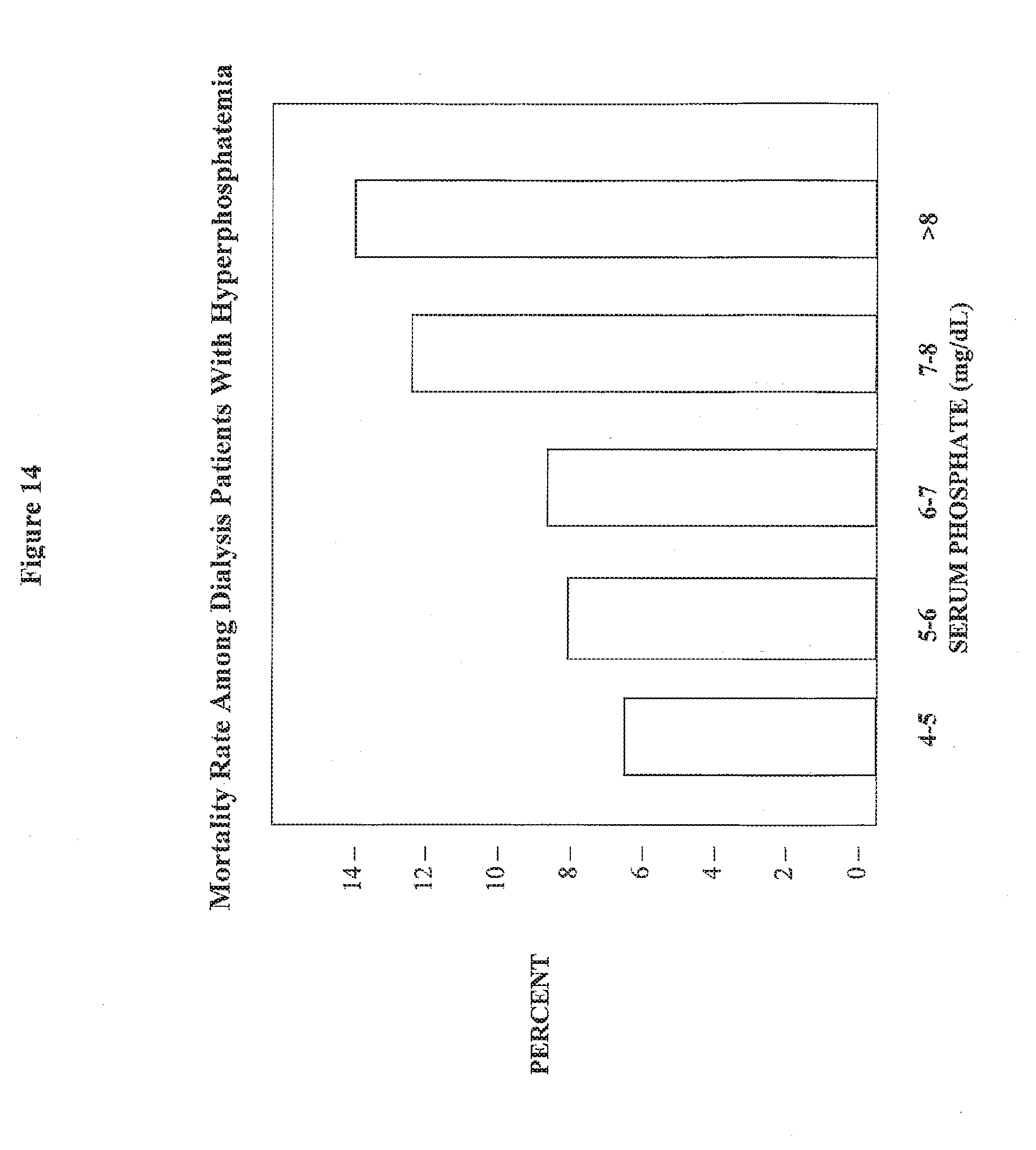
[0088] FIG. 14 shows a bar graph of the relationship between the rate of dialysis patient mortality and hyperphosphatemia
DETAILED DESCRIPTION OF THE INVENTION
[0089] In drawings which illustrate specific embodiments of the invention, but which should not be construed as restricting the spirit or described in detail to avoid unnecessarily obscuring the invention. Accordingly, the specification and drawings are to be regarded in an illustrative, rather than a restrictive, sense.
[0090] Throughout this application, references are made to the United States Pharmacopeia (USP), and the latest edition of the USP, USP 28, is hereby incorporated by reference into this application in its entirety.
[0091] This invention provides a method of preparing pharmaceutical-grade ferric citrate, comprising the steps of: (a) dissolving an appropriate amount of Ferric chloride hexahydrate in water to form a Ferric chloride hexahydrate solution; (b) dissolving an appropriate amount of NaOH in water to form a NaOH solution; (c) mixing the Ferric chloride hexahydrate solution and NaOH solution to form a solution with Fe(OH).sub.3 precipitate; (d) maintaining the pH of the solution with Fe(OH).sub.3 precipitate above 7.0; (e) isolating the Fe(OH).sub.3 precipitate; (f) washing the Fe(OH).sub.3 precipitate three times with water; (g) suspending the Fe(OH).sub.3 precipitate in water; (h) adding citric acid to the Fe(OH).sub.3 precipitate to form a ferric-organic acid solution; (i) stirring and heating the ferric-organic acid solution at 90-100.degree. C. for 30 to 120 minutes; (j) removing solids in the ferric-organic acid solution by adding citric acid; (k) allowing the ferric-organic acid solution to cool to below 30.degree. C.; (l) maintaining the pH of the ferric-organic acid solution to between 0.8-1.5; (m) filtering the ferric-organic acid solution to obtain a liquid filtrate; (n) mixing acetone and liquid filtrate to form ferric citrate; (o) isolating ferric citrate; (p) washing ferric citrate with acetone three times; and (q) drying the ferric citrate.
[0092] This invention provides a method for scalable or large-scale production of pharmaceutical-grade ferric citrate comprising the steps of: (a) mixing an appropriate amount of NaOH and Ferric chloride hexahydrate in a suitable reactor to form a ferric hydroxide slurry with ferric hydroxide precipitate; (b) maintaining the pH of the ferric hydroxide slurry to above 7.0; (c) isolating the ferric hydroxide precipitate from the ferric hydroxide slurry using pressure filtration; (d) washing the ferric hydroxide precipitate three times; (e) maintaining the % Cl in the ferric hydroxide precipitate to below 5%; (f) isolating the washed ferric hydroxide precipitate using pressure filtration; (g) mixing citric acid with washed ferric hydroxide precipitate to form a ferric organic acid solution; (h) stirring and maintaining the temperature of the ferric organic acid solution at 80.+-.5.degree. C. for 2 hours; (i) allowing the ferric organic acid solution to cool to 60.degree. C.; (j) maintaining the pH of the ferric organic acid solution to between 0.8 to 1.5 and the amount of Fe in the ferric organic acid solution to .gtoreq.85% of Fe added in step (a); (k) filtering the ferric organic acid solution using pressure filtration to obtain a liquid filtrate; (l) mixing the liquid filtrate with acetone to obtain ferric citrate; (m) isolating ferric citrate using pressure filtration; (n) washing the ferric citrate with acetone; (o) isolating the washed ferric citrate using pressure filtration; (p) drying the washed ferric citrate in fluidized bed dryer; and (q) maintaining the organic volatile impurities to .ltoreq.1000 ppm acetone.
[0093] In an embodiment, the ferric chloride hexahydrate complies with the release specification as shown in Table B; the citric acid complies with the release specification as shown in Table F; the water complies with the release specification as shown in Table D; the acetone complies with the release specification as shown in Table E; and the sodium hydroxide complies with the release specification as shown in Table C.
[0094] In another embodiment, the ferric citrate is dried using a fluidized bed dryer or is dried under vacuum.
[0095] In a further embodiment, the process as described above further comprises testing the ferric citrate for compliance with the release specification as shown in Table A. In a further embodiment, testing comprises performing at least one test selected from the group consisting of: assay content purity of ferric citrate and ferric citrate monohydrate; assay content of citric acid; assay content of detectable ferric citrate related substances; assay content of ferric ion; elemental iron impurity test; limit of ferrous iron test; loss on drying test; hydrate test (water content by differential scanning calorimetry); hydrate test (water content by karl Fischer Titration); trace or heavy metals test (As, Ca, Cd, Cu, Fe, Hg, Na, Pb, Sr, Zn); limit of oxalic acid test; identification A for ferric salts test; identification B by FTIR test; insoluble substances test; limit of ammonium test; limit of chloride test; limit of nitrate test; limit of tartrate test; residue on ignition test; organic volatile impurities test; and microbial, mold and yeast test.
[0096] This invention provides a pharmaceutical-grade ferric citrate prepared according to the methods described above.
[0097] This invention provides a composition comprising the ferric citrate prepared according to the methods described above for treating hyperphosphatemia or metabolic acidosis.
[0098] This invention provides a composition prepared according to the methods described above for treating disorders responsive to ferric organic compound therapy.
[0099] This invention provides a pharmaceutical-grade ferric citrate prepared according to the methods described above, wherein the ferric citrate produces the peak as shown in FIG. 6 when subjected to NMR spectroscopy analysis.
[0100] This invention provides a pharmaceutical-grade ferric citrate prepared according to the methods described above, wherein the ferric citrate possesses the dissolution rates as shown in FIG. 8.
[0101] This invention provides a pharmaceutical-grade ferric citrate prepared according to the methods described above, wherein the ferric citrate produces the spectral data as shown in FIG. 7 when subjected to Fourier Transform Infrared Spectrometry (FTIR) spectrum analysis.
[0102] This invention provides a composition comprising the ferric citrate as described above for treating hyperphosphatemia or metabolic acidosis.
[0103] This invention provides a composition comprising the ferric acid as describe above for treating disorders responsive to ferric organic compound therapy.
[0104] This invention provides a pharmaceutical-grade ferric citrate prepared by the process comprising the steps as shown in FIG. 1-4.
[0105] The pharmaceutical-grade ferric citrate according to the invention is useful for treating a subject suffering from hyperphosphatemia, metabolic acidosis or a disorder responsive to ferric organic compound therapy. In an embodiment, the subject is a human being.
[0106] The invention also provide a use for a composition comprising pharmaceutical-grade ferric citrate effective for one or more of the following uses, [0107] (a) treating hyperphosphatemia; [0108] (b) decreasing mortality rate in dialysis patients; [0109] (c) treating metabolic acidosis; [0110] (d) inhibiting calcium phosphate deposition; [0111] (e) decreasing serum calcium-phosphate product ([Ca].times.[P]) [0112] (f) decreasing serum calcium levels; [0113] (g) reversing calcification of soft tissue; and [0114] (h) aiding to dissolve a kidney stone.
[0115] The use may further encompass at least partly relying on reducing serum levels of calcium and phosphate ions.
[0116] The uses as stated above may apply to soft tissue such as a blood vessel or an eye.
[0117] The uses as stated may be carried out by administering over a long-term.
[0118] The invention allows for the above-stated uses to be achieved wherein the use is accompanied by less gastronintestinal adverse side effects than if the composition comprised chemical grade ferric citrate.
[0119] In accordance with the above-stated uses, the invention provides for a method for treating a disorder characterized by a high serum phosphate level comprising administering an effective amount of a composition comprising pharmaceutical-grade ferric citrate.
[0120] The invention also provides for the method wherein an effective amount of pharmaceutical-grade ferric citrate is administered in the form of a tablet, a powder, a suspension, an emulsion, a capsule, a lozenge, a granule, a troche, a pill, a liquid, a spirit, or a syrup.
[0121] The invention also provides for the method wherein the effective amount of pharmaceutical-grade ferric citrate is from 2 to 100 grams per day, preferably between 4 and 60 grams per day.
[0122] In some embodiments, the method may provide a daily effective amount of 2, 4, 6, or 8 grams.
[0123] In general, hyperphosphatemia is prevalent in patients with chronic renal failure and in patients on dialysis. There is also evidence that indicate elevated serum phosphorus, calcium-phosphorus product (Ca.times.P) and parathyroid hormone (PTH) levels contribute to increased incidence of vascular, visceral, peripheral vascular and soft tissue calcification in renal disease patients
[0124] Thus, it is an additional novel feature of the claimed invention to prevent or reverse calcification in renal disease patient or in a normal person. For example, dissolving kidney stones that may accompany renal failure.
[0125] Phosphorous exerts a negative impact on vascular calcification by direct participation in the Ca.times.P and indirectly in the pathogenesis and progression of hyperthyroidism. Serum calcium and phosphorous are metastable under normal circumstances, which means that their concentrations are not sufficient to produce spontaneous precipitation. However, once the calcification process begins, the concentrations are sufficient to support crystal proliferation.
[0126] Available evidence confirms a high prevalence of underlying vascular disease and structural heart disease in patients with severe chronic renal failure. These structural lesions are then exposed to elevated serum phosphorus, Ca.times.P, and PTH(1).
[0127] Factors which are considered likely to contribute to elevated serum phosphorus and Ca.times.P include administration of calcium-containing phosphorus binders. The calcium-containing phosphorus binders, such as calcium acetate are prescribed to many new hemodialysis (HD) and peritoneal dialysis (PD) patients, thus providing a large source of exogenous calcium to the GI tract.
[0128] Calcification also extends beyond renal disease patients and can include anyone who is over the age of 40. While the leading cause of death in the United States is acute myocardial infarction and stroke, hypercholesteromia contribute to only 15% of the deaths in this category and 85% is caused by ventricular calcification.
[0129] It has been shown that abnormalities in serum phosphorous, Ca.times.P and PTH levels can result in vascular, visceral and/or soft tissue calcification. For example, calcifications of myocardium, coronary arteries, cornea can lead to the development of a number of clinically significant complications including myocardial ischemia, myocardial infarction, impaired myocardial function, congestive heart failure, cardiac valve insufficiency and blindness.
[0130] Accordingly, there exists a need for methods of managing or reducing serum phosphorous as a means of treating numerous medical disorders. The method includes administering a phosphate binder which does not adversely affect serum calcium levels and does not cause toxic side effects in the patient.
EXAMPLES
[0131] In examples which are intended to illustrate embodiments of the invention but which are not intended to limit the scope of the invention:
Example 1. General Method for Synthesis of a Pharmaceutical-Grade Ferric Citrate
[0132] Referring to FIG. 1, which shows a general method of synthesis of the pharmaceutical-grade ferric citrate according to the invention, the flow chart shows implementation of quality control measures at selected stages of the synthesis process to ensure the final ferric citrate product complies with the Manufacture Release Specification as shown in Table A or any established Manufacture Release Specification for pharmaceutical-grade ferric citrate which have been approved or are suitable for human use. Other quality control measures or procedures, which are readily apparent to one of ordinary skill in the art, can be used or incorporated into the pharmaceutical-grade synthesis process to maintain the quality and purity of the final product and to increase the efficiency and yield of the synthesis process. See, for example, QC10 and QC11 in FIG. 1.
[0133] Examples of quality control measures employed in the synthesis process include: (QC4) maintaining pH of the ferric hydroxide slurry above 7.0; (QC5) maintaining the % Cl in ferric hydroxide precipitate below 5%; (QC7) maintaining the pH of the mixture between 0.8-1.5 and the Fe in mixture .gtoreq.85% of total Fe added after adding citric acid to ferric hydroxide precipitate; and (QC9) maintaining the level of acetone to .ltoreq.1000 ppm during the drying stage.
[0134] In an embodiment, the raw materials, i.e., ferric chloride, deionized water, citric acid, acetone, sodium hydroxide, must pass release specifications, such as those provided on Table B-F, before they can be used in the synthesis process. See FIG. 1, QC1-QC3, QC6 and QC8.
Example 1A. General Method for Synthesis of a Pharmaceutical-Grade Ferric Organic Compounds
[0135] Referring to FIG. 10, which shows a general method of synthesis of the pharmaceutical-grade ferric organic compounds according to the invention, the flow chart shows implementation of quality control measures at selected stages of the synthesis process to ensure the final ferric citrate product complies with established Manufacture Release Specification for pharmaceutical-grade ferric organic compounds which have been approved or are suitable for human use. Other quality control measures or procedures, which are readily apparent to one of ordinary skill in the art, can be used or incorporated into the pharmaceutical-grade synthesis process to maintain the quality and purity of the final product and to increase the efficiency and yield of the synthesis process. See, for example, QC10A and QC11A in FIG. 10.
[0136] Examples of quality control measures employed in the synthesis process include: (QC4A) maintaining pH of the ferric hydroxide slurry above 7.0; (QC5A) maintaining the % Cl in ferric hydroxide precipitate below 5%; (QC7A) maintaining the pH of the mixture between 0.8-1.5 and the Fe in mixture .gtoreq.85% of total Fe added after adding organic acid to ferric hydroxide precipitate; and (QC9A) maintaining the level of organic solvent to .ltoreq.1000 ppm during the drying stage.
[0137] In an embodiment, the raw materials, i.e., ferric iron salt, deionized water, organic acid, organic solvent, alkaline metal hydroxide, must pass release specifications before they can be used in the synthesis process. See FIG. 1, QC1A-QC3A, QC6A and QC8A. The organic acid can comprise citric acid, acetic acid, isocitric acid, succinic acid, fumaric acid, tartaric acid, or any other suitable organic acid. The organic solvent can comprise ethanol, methanol, butanol, acetone, isopropyl alcohol, tetrahydrofuran, or any other suitable organic solvent.
Example 2. Solubility Profile of Ferric Organic Compounds According to the Invention
[0138] The ferric organic compounds produced according to the methods described above are more soluble than commercially available ferric organic compounds, over a wider range of pH levels. This increase in solubility of the ferric organic compounds of the present invention is believed to be a result of the unique significantly large active surface area of the ferric organic compounds of the present invention. For example, at pH 8.0, the intrinsic dissolution rate of ferric citrate of the present invention is 3.08 times greater than the commercially available ferric citrate. See Table 1.
[0139] The intrinsic dissolution rates of commercially available ferric citrate were compared with the ferric citrate of the present invention. The intrinsic dissolution rate is defined as the dissolution rate of pure substances under the condition of constant surface area. The dissolution rate and bioavailability of a drug substance is influence by its solid state properties: crystallinity, amorphism, polymorphism, hydration, solvation, particle size and particle surface area. The measured intrinsic dissolution rate is dependent on these solid-state properties and is typically determined by exposing a constant surface area of a material to an appropriate dissolution medium while maintaining constant temperature, stirring rate, and pH. The intrinsic dissolution rates are presented in Table 1.
TABLE-US-00001 TABLE 1 Intrinsic dissolution rates of ferric citrate at 37.degree. C. in solutions of pH 8 Mean Rate of Intrinsic Intrinsic Acetone Dissolution Dissolution Addition Rates Rates Sample (ml/min) (mg/cm2/min) (mg/cm2/min) RFS-12 (sigma/ 10.0 0.83 0.83 commercially available) STM-134 10.0 1.88 3.08 (reference material) PAN031203A 10.0 3.82 (experimental batch 1) PAN031203B 10.0 4.00 (experimental batch 2) PAN031203C 9.5 2.68 (experimental batch 3) PAN031203D 40 2.95 (experimental batch 4) PAN031203E 4.4 3.13 (experimental batch 5)
[0140] For example, the BET active surface area of the ferric citrate of the present invention is at least 16 times larger than the commercially available ferric citrate. See Table 2.
[0141] The analysis of active surface area is based on BET theory which describes the phenomenon of mass and energy interaction and phase changes during gas adsorption onto solid surfaces and in pore spaces. In BET active surface area measurement, the volume of a monolayer of gas is determined which allows the surface area of the sample to be determined using the area occupied by a single layer of adsorbed gas molecule. Table 4 is a comparison of the active surface area of the ferric citrate of the present invention compared to the active surface area of commercially available ferric citrate compounds.
TABLE-US-00002 TABLE 2 BET active surface areas of various forms of ferric citrate Mean BET Dissolution Active Rates Surface Sample (mg/cm2/min) Area RFS-12-1 (sigma/commercially 0.76 0.61 available) RFS-12-2 (sigma/commercially available) STM-134-1 (reference material 1) 2.47 16.17 STM-134-2 (reference material 2) STM-182-1 (lab-scale 500 g batch 1) 2.61 19.85 STM-182-2 (lab-scale 500 g batch 2)
Example 3. Use of Ferric Organic Compounds According to the Invention in the Treatment of Disorders
[0142] The ferric organic compounds produced according to the methods described above are useful in the treatment of hyperphosphatemia, metabolic acidosis, and any other disorders responsive to ferric organic compound therapy. Because the ferric organic compounds of the present invention are more soluble than commercially available ferric organic compounds, smaller amounts of the ferric organic compounds of the present invention can be used to effectively treat patients suffering from such disorders.
[0143] Improved aqueous solubility is particularly relevant to the use of the ferric organic compounds of the present invention in the treatment of disorders responsive to ferric organic compound therapy. Because the ferric organic compounds of the present invention are more soluble, they will be more effective when taken orally, and therefore can be taken in lower doses. The ferric organic compounds of the present invention are more soluble over a wider pH range than commercially available ferric organic compounds; therefore, the ferric organic compounds of the present invention can be more effective by being soluble in the small intestine.
[0144] For example, in an experiment simulating the alkaline condition in the small intestine, the ferric citrate of the present invention showed better dissolution rate than the commercially available ferric citrate. It is suggested that the ferric citrate of the present invention can be more effective by being more soluble in the small intestine. See Table 1. As a result, patients can take lower doses of medication with lower incidences of side effects.
[0145] In one embodiment of the invention, the ferric citrate of the present invention has a significantly higher rate of aqueous solubility under physiological conditions than commercially available forms of ferric citrate, and therefore the ferric citrate of the present invention is believed to provide a significant improvement in the orally effective use of ferric citrate at a reduced dosage. By reducing the orally effective dose of ferric citrate, it is believed that the ferric citrate of the present invention will provide a lower incidence of ulcerative gastrointestinal adverse effects associated with commercially available ferric citrate compounds. In addition, it is believed that the increased rate of dissolution of the ferric citrate of the present invention will provide a more rapid onset of action in binding to dietary phosphate.
[0146] The ferric organic compounds of the present invention can be administered in a number of forms, including orally administrable forms, which can comprise the ferric organic compounds of the present invention alone or in combination with a pharmaceutically acceptable carrier. The orally administrable form includes, but is not limited to, a tablet, a powder, a suspension, an emulsion, a capsule, a granule, a troche, a pill, a liquid, a spirit, or a syrup. The composition can be administered to human beings or other animals suffering from illnesses responsive to ferric organic compound therapy.
Example 4. A Method of Making a Pharmaceutical-Grade Ferric Citrate
[0147] The present invention describes a process for manufacturing pharmaceutical-grade ferric citrate suitable as an active pharmaceutical ingredient for human use. An overview of the Ferric Citrate Manufacture Flow Chart is shown in FIG. 1. For a specific example, pharmaceutical-grade ferric citrate was produced using the procedure described below. Also see FIG. 2.
4.1. Preparation of Ferric Chloride Solution
[0148] Weigh 550 g of ferric chloride hexahydrate (correct for CoA purity) into a 1 L beaker.
[0149] Transfer the ferric chloride hexahydrate into a 4 L Erlenmeyer flask.
[0150] Measure 1.1 L of deionized water using a graduated cylinder. Use a small portion of the deionized water to rinse the beaker and transfer the water into the 4 L Erlenmeyer flask. Transfer the remaining water into the Erlenmeyer flask.
[0151] Stir solution using a magnetic stirring bar until completely dissolved. The solution is a dark yellow to dark brown color.
4.2. Preparation of Sodium Hydroxide Solution
[0152] Weigh 244 g of sodium hydroxide (correct for CoA purity) into a 500 mL beaker.
[0153] Transfer the sodium hydroxide into a 2 L Erlenmeyer flask.
[0154] Measure 1.1 L of deionized water using a graduated cylinder. Use a small portion of the deionized water to rinse the beaker and transfer the water into a 4 L Erlenmeyer flask. Transfer the remaining water into the Erlenmeyer flask slowly.
[0155] In a fumehood, stir the solution using a stirring bar while adding the water and stir until completely dissolved. The solution is clear and colorless.
[0156] Cool solution to below 30.degree. C. using a water bath.
4.3. Preparation of Ferric Hydroxide Intermediate
[0157] Place a magnetic stirring bar into the ferric chloride solution and place the flask in a water bath. Set up on a stirring plate and start the stirring plate at a low speed.
[0158] Add slowly the sodium hydroxide solution to the ferric chloride solution (at a rate of less than 20 mL/min) using an addition funnel and control the temperature of the reaction mixture below 40.degree. C. using the water bath and the rate of addition of sodium hydroxide.
[0159] Continue to cool the brown viscous mixture to below 30.degree. C. using the water bath.
[0160] The final pH should be above 7. Use a suitable volume of 5 M aqueous sodium hydroxide solution to correct the pH if not above 7. Measure and record the final pH. A dark brown precipitate of ferric hydroxide is formed.
[0161] If required, cool the brown viscous mixture to below 30.degree. C. using the cold water bath and filter the ferric hydroxide suspension through 1 mm size stainless steel filter to break up large precipitates.
[0162] Transfer equal amounts of ferric hydroxide suspension into four 500 mL centrifuge containers. Balance the weight of each centrifuge container using a top-loading balance before centrifugation.
[0163] Centrifuge the ferric hydroxide suspension at 1500 rpm and 25.+-.5.degree. C. for 5 minutes. Discard the supernatant.
[0164] Measure 2.5 L of deionized water using a graduated cylinder and use approximately 1 L of water to re-suspend the ferric hydroxide precipitate from the centrifuge containers.
[0165] Transfer the ferric hydroxide suspension into a 4 L Erlenmeyer flask fitted with a 1 mm size stainless steel filter over a glass funnel. Use the remaining 1.5 L of deionized water to rinse the containers and wash the precipitate retained on the stainless steel filter.
[0166] Wash the precipitate two more times by repeating the steps beginning with "Transfer equal amounts . . . ."
[0167] After the third wash, recover the precipitate by repeating the steps beginning with "Transfer equal amounts . . . " and ending with "Centrifuge the ferric hydroxide suspension . . . ."
[0168] Re-suspend the precipitate in 150 mL of deionized water.
4.4. Preparation of Ferric Citrate
[0169] Homogenize the ferric hydroxide precipitate using a mechanical stirrer for 5 min in a 2 L Erlenmeyer flask.
[0170] Weigh 490 g of citric acid (correct for CoA purity) into a 500 mL beaker.
[0171] Place a stir bar in the 2 L Erlenmeyer flask in an oil bath and stir at high speed.
[0172] Add the citric acid into the ferric hydroxide suspension.
[0173] Stir the solution for 30 minutes.
[0174] Heat the mixture at 90 to 100.degree. C. (in oil bath) until the color changes from orange-brown to a clear black-brown (for 30 to 120 min) or until ferric hydroxide precipitate is dissolved.
[0175] Take 1 mL aliquot of the reaction mixture in a 6 mL glass test tube and centrifuge at 1500 rpm and 25.+-.5.degree. C. for 5 minutes. Proceed to the next step if no precipitate is observed. If some precipitate is observed add 10 to 34 g citric acid to the mixture and continue heating for 10 to 30 min.
[0176] Terminate the heating and cool the mixture to below 30.degree. C. Measure the pH of the reaction mixture; it should be pH 0.8 to 1.5.
[0177] Transfer equal amounts of the reaction mixture into four 500 mL centrifuge containers and balance the weight of each container using a top-loading balance.
[0178] Centrifuge the reaction mixture at 1500 rpm and 25.+-.5.degree. C. for 5 minutes. Transfer and pool all the ferric citrate supernatant to a clean 4 L Erlenmeyer flask.
[0179] Repeat the above 2 steps for all of the ferric citrate reaction mixture.
[0180] Place one-half of the ferric citrate supernatant in a 4 L Erlenmeyer flask and stir with a magnetic stir bar at high speed.
[0181] Add slowly (over 20 min) 3.5 L of acetone (accurate volume acetone calculated as five fold the supernatant volume) and stir for an additional 10 min. A light-beige color precipitate forms.
[0182] Transfer the suspension into four 500 mL centrifuge containers and balance the weight of each container using a top-loading balance.
[0183] Centrifuge the ferric citrate suspension at 1500 rpm and 25.+-.5.degree. C. for 5 minutes.
[0184] Transfer and pool all the ferric citrate precipitate to a clean 4 L Erlenmeyer flask.
[0185] Repeat the above 4 steps with the second half of the ferric citrate supernatant.
[0186] Pool all ferric citrate precipitate, add 1.4 L of acetone and stir for 5 min.
[0187] Transfer the suspension into four 500 mL centrifuge containers and balance the weight of each container using a top-loading balance.
[0188] Centrifuge the suspension at 1500 rpm and 25.+-.5.degree. C. for 5 minutes.
[0189] Repeat the above 2 steps until all suspension is centrifuged.
[0190] Transfer and pool all the ferric citrate precipitate to a clean 4 L Erlenmeyer flask.
[0191] Repeat the above 5 steps two additional times (total of 3 washes).
[0192] Label and weigh drying trays, and record their weight.
[0193] Transfer the ferric citrate precipitate onto the drying dishes and dry at ambient temperature (25.+-.5.degree. C.) for 16 hours.
[0194] Place the drying trays with precipitate into a vacuum oven and dry at ambient temperature (25.+-.5.degree. C.) and under vacuum (about 20 mm Hg) for 8 to 16 hours (until the material appears ready for grinding).
[0195] Reduce the particle size of the ferric citrate in a porcelain mortar and pestle.
[0196] Place the ferric citrate powder into a vacuum oven and dry at ambient temperature (25.+-.5.degree. C.) and under vacuum (about 20 mm Hg) for 8 to 24 hours, until the material appears ready for sieving.
[0197] Finely reduce the ferric citrate particle size in a porcelain mortar and pestle. Screen the ferric citrate powder through a 45 mesh size (355 micron) sieve.
[0198] Transfer the ferric citrate powder into drying trays and place the trays in an oven to dry at 25.+-.5.degree. C. and under high vacuum until the material appears dry (20 to 48 hours).
[0199] Transfer the powder into pre-weighed plastic amber containers.
[0200] Label and store the containers at ambient temperature and protected from light.
Example 5. Method for Scalable Manufacture of Pharmaceutical-Grade Ferric Citrate
[0201] The present invention provides a scalable manufacturing of pharmaceutical-grade ferric citrate. Preferably, the ferric citrate manufacturing process is capable of producing at least 125 kg batches of pharmaceutical-grade ferric citrate. An overview of the ferric citrate manufacturing is shown in FIG. 3. Details of the synthesis of ferric citrate are shown in FIG. 4.
[0202] The scalable manufacturing process further employs fluidized bed dryer for drying wet ferric citrate and for attaining release specifications for organic volatile impurities. See Table A for the Manufacture Release Specifications for Pharmaceutical-Grade Ferric Citrate.
Example 6. A Pharmaceutical-Grade Ferric Citrate
[0203] This invention provides a pharmaceutical-grade ferric citrate which complies with the Manufacture Release Specifications as shown below in Table A. The pharmaceutical-grade ferric citrate of consistent purity and quality can be prepared using the manufacturing process of the present invention. See FIGS. 1-4 for the schematic diagram of the ferric citrate manufacturing and quality control process. The ferric citrate manufacturing and quality control process can be readily scaled to produce multi-kilogram batch sizes or scaled up to a manufacturing scale.
TABLE-US-00003 TABLE A Manufacture Release Specification for Pharmaceutical-Grade Ferric Citrate Test Item Method Limit of Specification Appearance Visual Light brown to beige powder Purity of solid Calculate based on LC/MS - NLT 90% w/w state ferric citrate flow injection anhydrous basis quantitation and profile, based on USP 25 <621>, <731>, <1086>, <736> Assay content of LC/MS - flow injection Run and report ferric citrate non- profile, based on USP 25 as % w/w related substances <621>, <731>, <1086>, anhydrous basis in solution state <736> (attach table summary) Assay content purity LC/MS - flow injection NLT 70% w/w of ferric citrate quantitation, based on anhydrous basis and water adduct in USP 25 <621>, <731>, solution state <1086>, <736> Assay content of LC/MS - flow injection NLT 10% w/w citric acid related quantitation, based on anhydrous basis substance in USP 25 <621>, <731>, solution state <1086>, <736> Assay content of LC/MS - flow injection Run and report other ferric citrate profile, based on USP 25 as % w/w related substances <621>, <731>, <1086>, anhydrous basis in solution state <736> (attach table (excluding citric summary) acid) Assay content of Based on USP 25 ferric NLT 16% w/w fresh ferric iron sulfate assay weight basis Limit of ferrous Gravimetric method using NMT 1% w/w fresh iron potassium ferricynide, weight basis based on USP 25 <191> Loss on drying USP 25 <731> NMT 20% Hydrate (water Karl Fischer Titration NMT 20% content) USP 25 <921> water determination, Method Ia (Direct Titration) Identification Method A: based on USP 25 Dark blue <191> ferric salt precipitate with K4Fe(CN)6 TS; reddish brown ppt with excess 1N NaOH; deep red color not destroyed by mineral acids with NH4SCN TS Sample solution gives light red color with pyridine and acetic anhydride Limit of chloride Based on USP 25 Ferric NMT 5% sulfate procedures Reside on ignition Modified USP 25 <281> Run and report Organic Volatile Based on USP 25 <467> Acetone not more Impurities than 1000 ppm Limit of acid Gravimetric Not more than insoluble determination, based on 0.02% w/w fresh substances USP 25, ferric sulfate weight basis Trace/heavy USP 25 method <231> or As <0.3 ppm metals equivalent Others: run and ICP for Zn, Cu, Sr, Ca, report Na GFAAS for As Lead, Cadmium ICP-MS Pb < 5 ppm Cd < 2 ppm Mercury Cold vapor/AA Hg < 0.3 ppm Total iron content ICP NLT 16% w/w fresh weight basis Microbial/Mold USP Method <61> and Yeast Salmonella Salmonella = negative E. Coli E. Coli = negative Coliforms Total Coliforms < 3 cfu/g Total aerobic count Total aerobic count < 10 cfu/g Total combined mold and Total mold and yeast < 20 cfu/g yeast
TABLE-US-00004 TABLE B Ferric chloride hexahydrate Release Specifications Test Item Method Limit of Specification Appearance Visual Yellow to yellowish brown powder, crystals or chunks Identification - USP 25 <191> Yield dark blue precipitate with Ferric salts potassium ferrocyanide TS Form reddish brown precipitate with excess 1N NaOH Red deep color with ammonium thiocyanate TS and color not destroyed by dilute mineral acids Identification - USP 25 <191> Aqueous solution of ferric chloride Chloride yields with 0.1N silver nitrate TS a white, cruddy precipitate, which is insoluble in nitric acid but is soluble in a slight excess of 6N ammonium hydroxide Heavy metals ICP-MS As < 0.1 ppm Cd < 0.1 ppm Hg < 0.1 ppm Pb < 0.1 ppm Assay - Ferric USP 25, Assay >18% iron ferric sulfate, p. 2303
TABLE-US-00005 TABLE C Sodium Hydroxide Release Specifications Test Item Method Limit of Specification Appearance Visual White pellets, odorless Identification - USP25, <191> No precipitate is formed with Sodium potassium carbonate A dense precipitate is formed with potassium pyroantimonate An intense yellow color to a non- luminous flame
TABLE-US-00006 TABLE D Deionized Water Release Specification Test Item Method Limit of Specification Appearance Visual Clear, colorless and odorless Mineral scan ICP-MS As < 0.001 ml/L Cd < 0.0002 ml/L Pb < 0.001 ml/L Hg < 0.02 .mu.g/L Total Organic Standard <1 mg/L Carbon Method for the Examination of Water and Wastewater, 20.sup.th ed. Total Hardness Standard <4 mg/L Method for the Examination of Water and Wastewater, 20.sup.th ed. Total Plate USP 25 method <10 cfu Count <61> Total Coliform USP 25 method <3 cfu Count <61>
TABLE-US-00007 TABLE E Acetone Release Specification Test Item Method Limit of Specification Appearance Visual Clear colorless liquid Identification - USP 25, The IR absorption of a thin film Acetone p. 2502, between KBr plates exhibits a FTIR strong band at about 5.8 .mu.m; a strong region of absorption between 6.8 and 7.5 .mu.m, with maxima at about 7.0 and 7.3 .mu.m; a strong maximum at about 8.2 .mu.m; and a weak maxima at about 9.2 and 11.0 .mu.m. Assay From NLT 99.5% manufacturer's Certificate of Analysis (result from GC method preferred) Aldehyde From NMT 0.002% (as HCHO) manufacturer's Certificate of Analysis Isopropyl From NMT 0.05% alcohol manufacturer's Certificate of Analysis Methanol From NMT 0.05% manufacturer's Certificate of Analysis Residue after From NMT 5 ppm evaporation manufacturer's Certificate of Analysis Acids From NMT 0.0003 meq manufacturer's Certificate of Analysis (result from titrimetric test) Bases From NMT 0.0006 meq manufacturer's Certificate of Analysis (result from titrimetric test) Water From NMT 2% manufacturer's Certificate of Analysis Insoluble From filtered through .ltoreq.0.45 .mu.m substances manufacturer's filter Certificate of Analysis or filtered through .ltoreq.0.45 .mu.m filter
TABLE-US-00008 TABLE F Citric Acid Release Specification Test Item Method Limit of Specification Appearance Visual White or colorless crystals or powder Identification - USP 25 <191> A light red color is produced Citrate
Example 8. Final Product Manufacture Release Testing Methods
[0204] The following tests are performed to ensure the final ferric citrate product prepared according to the method or process of the present invention complies with the established Manufacture Release Specification as shown in Table A. The Manufacture Release Specification may be readily modified or revised by one of ordinary skill in the art following the teaching of this invention to enhance the purity and safety of the pharmaceutical-grade ferric citrate for human use. [0205] (a) Based on USP 25 <191> Gravimetric method using potassium ferricyanide, p. 1918--Limit of Ferrous Iron in Ferric Citrate [0206] (b) Based on USP 25, ferric sulfate assay, p. 728--Limit of Chloride in Ferric Citrate [0207] (c) Based on USP 25, ferric sulfate assay, p. 728--Limit of Acid Insoluble Substances in Ferric Citrate [0208] (d) Based on USP 25, ferric sulfate assay, p. 728--Assay Content of Ferric Iron in Ferric Citrate [0209] (e) Based on USP 25 <467>--Determination of Acetone in Ferric Citrate Samples by GClFID Headspace [0210] (f) Based on USP 25 <191> Ferric Salts; Citrate, p. 1918--Identification of Ferric Citrate [0211] (g) Based on USP 25 <621>, p. 1988-1995, <731>, <1086>, p. 2157-2159, <736>, p. 2029-2033--LCMS Flow-Injection Quantitation and Profile of Ferric Citrate and Related Substances [0212] (h) USP 25 <731> Loss on Drying [0213] (i) USP 25 <921> Water Determination, Method Ia (Direct Titration) [0214] (j) ICP for Zn, Cu, Sr, Ca, Na and total iron [0215] (k) GFAAS for As [0216] (l) ICP-MS for lead and cadmium [0217] (m) Cold vapor/AA for mercury [0218] (n) Residue on ignition [0219] (o) Microbial/mold and yeast
Example 9 Methods of Using and Testing the Pharmaceutical-Grade Ferric Citrate in Patients In Vivo
A. Handling and Forms of Test Compositions
[0220] Ferric citrate is supplied in 500 mg capsules, whereas the placebo will be provided in identical-looking capsules (indistinguishable from those containing the active drug); the placebo capsules will contain sorbitol and colorant to match the powder color of the active capsules. The placebo capsule shells will be identical to the active capsule shells.
Storage
[0221] All study drug supplies must be stored under secure conditions and are not to be used after their expiration date, which is imprinted on the study drug container. The study drugs should be kept under controlled conditions (15 to 30.degree. C.; 59 to 86.degree. F.) in a tightly closed container, protected from light.
Dosage
[0222] A recent pilot study compared ferric citrate (3 g daily) to calcium carbonate (3 g daily) for reducing serum P0.sub.4 in patients with End Stage Renal Disease (ESRD). Although ferric citrate caused a significant decrease in serum P04, it was not as effective as calcium carbonate. This dose of ferric citrate was associated with mild, but tolerable GI symptoms.
[0223] As shown in FIGS. 12 and 13, treatments using pharmaceutical-grade ferric citrate provide several advantages over chemical grade ferric citrate. In general, while pharmaceutical-grade ferric citrate demonstrates an efficacy approximately equal to that of chemical grade ferric citrate, it achieves this result with less adverse side effects than chemical grade ferric citrate.
[0224] FIG. 12 also indicates that adverse side effects associated with administering pharmaceutical-grade ferric citrate were not statistically different from those associated with the placebo. An advantage of this safety profile is that an individual patient may have his dosing of pharmaceutical-grade ferric citrate titrated over a broad range of doses with less concern about side effect. In this way, a patient's individual treatment may be tailored to suit his specific needs and tolerances.
[0225] The doses of ferric citrate chosen for study or treatment may be from 1 to 30 grams of ferric citrate per day. In part, this may depend on the nature of the formulation provided. For example, ferric citrate capsules may be administered up to a daily dose of about 15 grams/day, whereas the tablet form may be administered up to 30 grams/day. Thus, there is a very broad range of dosing regimens encompassed by the invention.
Titration of Optimal Dosage for a Subject
[0226] In the context of this invention, the term "subject" refers to either a human or non-human animal.
[0227] The optimal dosage of an individual subject or groups may be determined as follows. A dose of approximately one or two grams per day is merely suggested as an illustrative starting dose. The daily dose may be increased as needed until the desired result is observed.
[0228] The intent of the invention is to not limit the dose range employed. Therefore, depending on the subject(s) the daily dose administered may approximate thirty, forty, fifty, sixty, seventy, eighty, ninety or one hundred grams per day. The safety profile of the pharmaceutical-grade ferric citrate allows the implementation of a broad range of doses.
[0229] Further, it is the intent of the invention to not be limited to capsules and tablets as oral formulations. It is known in the art that a wide variety of oral formulations may be adapted for use with the invention.
Illustrative Example of a Dosage Regimen
[0230] An non-limiting example of a dosing regimen is provided below. This is not meant to limit the invention as to how an effective amount of ferric citrate is selected, or the form in which it is provided or the frequency of administering the composition per day. The following merely illustrates how ferric citrate and placebo may be administered; e.g., as 500 mg capsules of identical appearance. All patients may receive (in a blinded fashion) 4 capsules with each of three meals, on a daily basis, for 28 days. Patients will be instructed to take the study medication within 10 minutes of finishing their meals (breakfast, lunch, and dinner).
[0231] The number of placebo, and active capsules to be taken at each meal, are as follows:
Placebo Arm of the Study
[0232] 4 placebo capsules, with breakfast 4 placebo capsules, with lunch 4 placebo capsules with dinner
Ferric Citrate 2 g Per Day Arm of the Study
[0233] 1 ferric citrate capsule and 3 placebo capsules with breakfast 1 ferric citrate capsule and 3 placebo capsules with lunch 2 ferric citrate capsules and 2 placebo capsules with dinner
Ferric Citrate 4 g Per Day Arm
[0234] 2 ferric citrate capsules and 2 placebo capsules with breakfast 3 ferric citrate capsule and 1 placebo capsule with lunch 3 ferric citrate capsules and 1 placebo capsule with dinner
Ferric Citrate 6 g Per Day Arm
[0235] 4 ferric citrate capsules with breakfast 4 ferric citrate capsules with lunch 4 ferric citrate capsules with dinner
B. Clinical Schedule and Assessments
Duration of Study Treatment
[0236] Each patient's participation in the trial lasts for up to 8 weeks: the screening period (approximately 1-2 weeks), a 1-2 week washout, and 4 weeks of treatment with study medication.
Screening Visit 1 (Study Days -30 to -15)
[0237] The following procedures will be performed at the first screening visit:
1. Medical history, including concomitant medications. 2. Demographic data. 3. Physical examination, including height, weight, and vital signs. 4. Dietary interview, using 24 hour recall method, to assess dietary intake of calcium and phosphorous, three times during screening period, to include one dialysis day, one non-dialysis day, and one weekend day. Note: Dietary interview may be also performed, in part or in whole, during the washout period. 5. Laboratory assessment: [0238] Hematology: complete blood count (CBC) with differential, platelets. [0239] Chemistries: sodium, potassium, chloride, bicarbonate, blood urea nitrogen (BUN), creatinine, glucose (random), aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), total bilirubin, total protein, albumin, serum calcium, serum phosphate, magnesium [0240] Total and LDL cholesterol levels [0241] Serum (3-HCG for women of childbearing potential [0242] Iron panel: serum iron, ferritin, transferrin saturation percentage, and total iron binding capacity. 6. 12-lead ECG. 7. Patients will be given instructions for the Washout
Period (Study Days -14 to -1):
[0243] a. All phosphate-binding agents will be discontinued at Day -14 b. Any iron therapy (oral or intravenous) will be discontinued at Day -14 c. Patients who have been receiving a stable dose of vitamin D or calcitriol for I month prior to enrollment will be instructed to maintain their current dose throughout the study d. Patients will be advised to avoid changes in diet, calcium or magnesium containing antacids (other medications).
Screening Visit 2 (Study Days -7+/-1 Day)
1. Laboratory Assessment:
Serum P0.sub.4
[0244] Note: Patients with a Day -7 serum P0.sub.4 .gtoreq.5.5 mg/dL and .ltoreq.10 mg/dL may be randomized before the 2-week washout is complete. The day of randomization will automatically become Day 0. Note: Patients with a Day -7 phosphate level of .gtoreq.10 mg/dL will be removed from the study and instructed to resume their pre-study medications.
Study Day 0 (Randomization and Dosing)
[0245] 1. Physical examination, including weight and vital signs. 2. Adverse event query. 3. Concomitant medication query. 4. Baseline Laboratory assessments: [0246] Serum P04 [0247] Serum Ca [0248] Iron panel: serum iron, ferritin, transferrin saturation percentage, and total iron binding capacity Note: The Baseline Laboratory Assessments may be done up to 3 days prior to Day 0 5. Patients with a P0.sub.4 level 0.5 mg/dL and 5 10 mg/dL will be randomized and a 15-day supply of study medication will be dispensed. Note: Patients should be instructed to begin taking study medication within 10 minutes of completing their next meal on Day 0.
Study Day 14 (Midpoint Evaluation)
[0249] The following procedures will be performed at Study Day 14+/-1 day.
1. Physical examination including weight and vital signs. 2. Adverse event query. 3. Concomitant medication query. 4. Dispense an additional 15-day supply of study medication. All returned capsules should be counted and recorded in the Case Report Form. 5. Laboratory assessment: [0250] Hematology: CBC with differential, platelets. [0251] Chemistries: sodium, potassium, chloride, bicarbonate, BUN, creatinine, glucose (random), AST, ALT, ALP, total bilirubin, total protein, albumin, calcium, phosphate, magnesium. [0252] Iron panel: serum iron, ferritin, transferrin saturation percentage, and total iron binding capacity. [0253] Total and LDL cholesterol levels. Note: Patients with a Day 14 phosphate level of >10 mg/dL will be removed from the study and instructed to resume their pre-study medications.
Study Day 28 (End of Study Evaluation)
[0254] The following procedures will be performed at Study Day 28+/-1 day or on the day of early termination.
1. Physical examination including weight and vital signs 2. Adverse event query. 3. Concomitant medication query. 4. Laboratory assessment: a. Hematology: CBC with differential, platelets b. Chemistries: sodium, potassium, chloride, bicarbonate, BUN, creatinine, glucose (random), AST, ALT, ALP, total bilirubin, total protein, albumin, calcium, phosphate, magnesium. c. Total and LDL cholesterol levels [0255] d. Iron panel: serum iron, ferritin, transferrin saturation percentage, and total iron binding capacity. 5. 12-lead ECG 6. Patients will be instructed to resume their pre-study medications after completing the study.
C. Data Management and Analysis
[0256] GloboMax will be the primary data management, monitoring, and coordinating center. Case report forms (CRF) will be provided for each subject. Subjects will not be identified by name or initials on CRFs. The CRF will be monitored at the clinical sites and collected by GloboMax's study monitor. Audited CRFs will be used to create electronic data files.
[0257] Three major categories of endpoints reflect biochemical and clinical issues being addressed at the outset. Additional clinical and biochemical issues are addressed as they arise. Therefore, the endpoints are not meant to limit the totality of relevant findings and measurements collected in these, or future studies.
Primary Endpoints (see FIGS. 9-10)
[0258] The change in serum P0.sub.4 concentration at Days 14 and 28 from baseline.
Secondary Endpoints (see FIGS. 9-10)
[0258] [0259] The change in serum calcium concentration at Days 14 and 28 from baseline. [0260] The change in iron, ferritin, transferrin saturation percentage, and total iron binding capacity at Days 14 and 28 from baseline. [0261] The change in the Ca'PO.sub.4 product at Days 14 and 28 from baseline.
[0262] It is further noted that in comparison to chemical grade ferric citrate, the pharmaceutical grade ferric citrate demonstrates similar efficacy. See FIG. 13. However, the safety profiles indicate that the pharmaceutical grade generally results in less adverse clinical effects. See FIG. 13.
Safety Endpoints (See FIG. 11)
[0263] Safety will be monitored by recording adverse events (FIG. 11) and the results of physical examinations, vital signs and laboratory tests at each study visit.
[0264] Specific rules for withdrawal from the study, based on laboratory data, have also been set up to ensure patient safety. A nonlimiting example of such criteria is illustrated by the following:
[0265] If a patient's serum phosphate level increases to .gtoreq.10 mg/dL at any time during the study period, the patient will be withdrawn from the study.
[0266] Specific studies have also shown that pharmaceutical grade ferric citrate possesses similar efficacy to chemical grade ferric citrate. (See FIG. 13). However, the pharmaceutical grade generally affords a significantly more desirable safety profile as shown in FIG. 12. This indicates an important advance in regulating serum phosphate levels.
* * * * *
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.