Acrylic Polymers And Compositions Containing Such Polymers
Gibanel; Sebastien ; et al.
U.S. patent application number 16/342873 was filed with the patent office on 2019-08-15 for acrylic polymers and compositions containing such polymers. This patent application is currently assigned to SWIMC LLC. The applicant listed for this patent is SWIMC LLC. Invention is credited to Marie Braillon, Joseph DeSousa, Sebastien Gibanel, Nusrah Hussain, Jason S. Ness, Robert M. O'Brien, Samuel Puaud, Kailas Sawant.
| Application Number | 20190249029 16/342873 |
| Document ID | / |
| Family ID | 62018867 |
| Filed Date | 2019-08-15 |
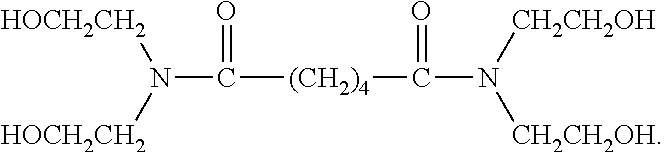
| United States Patent Application | 20190249029 |
| Kind Code | A1 |
| Gibanel; Sebastien ; et al. | August 15, 2019 |
ACRYLIC POLYMERS AND COMPOSITIONS CONTAINING SUCH POLYMERS
Abstract
A coating composition is provided that is preferably substantially free of bisphenol A. The coating composition is useful in coating metal substrates such as, for example, interior and/or exterior surfaces of food or beverage cans. In some embodiments, the coating composition is formulated using an acrylic polymer that is formed form ingredients that do not include styrene.
| Inventors: | Gibanel; Sebastien; (Givry, FR) ; Braillon; Marie; (Tournus, FR) ; Puaud; Samuel; (Tournus, FR) ; Hussain; Nusrah; (Gibsonia, PA) ; Sawant; Kailas; (Mars, PA) ; DeSousa; Joseph; (Pittsburgh, PA) ; O'Brien; Robert M.; (Monongahela, PA) ; Ness; Jason S.; (Woodbury, MN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | SWIMC LLC Cleveland OH |
||||||||||
| Family ID: | 62018867 | ||||||||||
| Appl. No.: | 16/342873 | ||||||||||
| Filed: | October 19, 2017 | ||||||||||
| PCT Filed: | October 19, 2017 | ||||||||||
| PCT NO: | PCT/US2017/057432 | ||||||||||
| 371 Date: | April 17, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62410255 | Oct 19, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B65D 51/00 20130101; B65D 23/02 20130101; C09D 133/08 20130101; B05D 7/227 20130101; B05D 7/14 20130101; C08F 220/18 20130101; B05D 1/02 20130101; B65D 25/14 20130101; C09D 133/02 20130101; B05D 2520/05 20130101; C08F 2/22 20130101; C09D 125/14 20130101; C09D 7/65 20180101; C09D 133/10 20130101; C08L 33/06 20130101; C08F 220/1804 20200201; B05D 2202/25 20130101; C09D 5/024 20130101 |
| International Class: | C09D 133/08 20060101 C09D133/08; B05D 7/22 20060101 B05D007/22; B05D 7/14 20060101 B05D007/14; C08F 220/18 20060101 C08F220/18 |
Claims
1. An inside spray coating composition comprising: an acid- or anhydride-functional acrylic polymer comprising an acid- or anhydride-functional latex that is substantially free of styrene and has a glass transition temperature of greater than 40.degree. C.; and a nitrogen-containing carboxyl-reactive crosslinker; wherein the coating composition is an aqueous coating composition that is suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A.
2-40. (canceled)
41. The coating composition of claim 1, wherein the nitrogen-containing carboxyl-reactive crosslinker includes hydroxyl groups and at least one amide group.
42. The coating composition of claim 41, wherein the nitrogen-containing carboxyl-reactive crosslinker includes a hydroxyl group that is located beta relative to the nitrogen atom of at least one amide group.
43. The coating composition of claim 1, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises one or more groups capable of forming an intermediate having an oxazolinium structure.
44. The coating composition of claim 1, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises: ##STR00004##
45. The coating composition claim 1, wherein the nitrogen-containing carboxyl-reactive crosslinker includes one or more aziridine, diimide, or oxazoline groups.
46. The coating composition of claim 1, wherein the coating composition includes at least one weight percent, based on total resin solids, of the nitrogen-containing carboxyl-reactive crosslinker, and wherein the coating composition includes at least 50 weight percent, based on total resin solids, of the acid- or anhydride-functional latex.
47. The coating composition of claim 1, wherein the acid- or anhydride-functional latex has an acid number of at least 20 mg KOH/g resin.
48. The coating composition of claim 1, wherein at least a portion of the acid- or anhydride-functional latex is formed from an emulsion polymerized ethylenically unsaturated monomer component including at least one monomer having (i) a Tg of more than 40.degree. C. and (ii) one or more groups selected from cyclic groups, branched organic groups, or a combination thereof.
49. The coating composition of claim 48, wherein at least one branched organic group is present, and the at least one monomer having (i) and (ii) has the following structure: (R.sup.3).sub.2--C.dbd.C(R.sup.4)--W.sub.n--Y, wherein: R3 is independently selected from hydrogen or an organic group; R4 is selected from hydrogen or an alkyl group; W, if present, is a divalent linking group; n is 0 or 1; and Y comprises a branched organic group including one or more branching atoms.
50. The coating composition of claim 49, wherein Y is a branched organic group of the following structure: --C(CH.sub.3).sub.t(R.sup.5).sub.3-t wherein: t is 1; each R.sup.5 comprises an alkyl group that may optionally be itself branched; two or more R.sup.5 may optionally form a cyclic group with one another; and the total number of carbon atoms in both R.sup.5 groups is 6, 7, or 8.
51. The coating composition of claim 48, wherein the emulsion polymerized ethylenically unsaturated monomer component includes at least 10 weight percent of one or more branched or cyclic monomers.
52. The coating composition of claim 1, wherein one or both of: (i) the acid- or anhydride-functional latex polymer and (ii) the coating composition are substantially free of each of bisphenols and halogenated monomers.
53. The coating composition of claim 1, wherein the acid- or anhydride functional latex is formed from ingredients including an emulsion polymerized ethylenically unsaturated monomer component that includes a multi-ethylenically unsaturated monomer.
54. The coating composition of claim 1, wherein the emulsion polymerized ethylenically unsaturated monomer component includes at least 20 weight percent of methyl methacrylate.
55. The coating composition of claim 1, wherein the coating composition includes, based on total resin solids, from 1 to 20 weight percent of the nitrogen-containing carboxyl reactive crosslinker and from 50 to 99 weight percent of the acid- or anhydride-functional latex.
56. The coating composition of claim 1, wherein the coating composition includes both the nitrogen-containing carboxyl reactive crosslinker and a resole phenolic crosslinker.
57. The coating composition of claim 17, wherein the coating composition includes, based on total resin solids, 2 to 10 weight percent of a beta-hydroxyalkylamide crosslinker and 1 to 10 weight percent of a resole phenolic crosslinker.
58. The coating composition of claim 1, wherein the acid- or anhydride-functional latex is a reaction product of a multi-ethylenically unsaturated monomer component emulsion polymerized in the presence of a non-polymeric surfactant.
59. The coating composition of claim 1, wherein the coating composition exhibits an elongation at break of at least 1% when tested pursuant to the Elongation at Break test disclosed herein.
60. A method of coating a food or beverage can, comprising the steps of: spray applying the coating composition of claim 1 on an interior surface of a food or beverage can, or a portion thereof, and curing the coating composition on the metal substrate to form a continuous cured coating having an average film thickness of from about 2 to about 15 micrometers and a metal exposure value after drop damage of less than 10 mA when tested pursuant to the Metal Exposure after Drop Damage test disclosed herein.
61. A food or beverage can, or a portion thereof, having an interior food-contact coating having an overall average dry coating thickness of from 2 to 15 micrometers, wherein: the interior food-contact coating is formed from a spray applied aqueous coating composition of claim 1; and the interior food-contact coating has a metal exposure value after drop damage of less than 10 mA when tested pursuant to the Metal Exposure after Drop Damage test disclosed herein.
Description
CROSS REFERENCE TO RELATED APPLICATION
[0001] This application claims the benefit of U.S. Provisional Application No. 62/410,255 filed on Oct. 19, 2016 and entitled "STYRENE-FREE ACRYLIC POLYMERS AND COMPOSITION CONAINING SUCH POLYMERS," which is incorporated herein by reference in its entirety.
BACKGROUND OF THE INVENTION
[0002] There is a desire to reduce or eliminate the use of certain bisphenol A-derived polymers in food or beverage container coatings. Although a number of replacement coating compositions made without bisphenol A have been proposed, some replacement compositions have exhibited insufficient coating properties such as insufficient corrosion resistance on metal substrates, insufficient flexibility or insufficient toughness.
[0003] In recent years styrene has also come under greater scrutiny. Although the balance of scientific evidence indicates that coatings containing polymerized styrene are safe for food-contact end uses, there is a desire by some to eliminate styrene from such end uses. Styrene, however, brings advantageous properties that contribute to the overall performance of food or beverage can coatings and can be difficult to replicate using other materials.
[0004] Coatings for use on food or beverage containers should avoid unsuitably altering the taste of the packaged food or beverage products, and should also avoid flaking or chipping into the packaged products. The coatings should also resist chemically aggressive food or beverage products (which can have a complex chemical profile, including salts, acids, sugars, fats, etc.) for extended periods of time (e.g., years). Food or beverage container coatings should also have good adhesion to the underlying substrate and remain sufficiently flexible after curing to survive subsequent fabrication and/or denting during transportation, storage or use that causes the metal substrate to deform and the coating to flex. Some brittle coatings have been observed to crack during flexure, thereby exposing the container metal to the packaged products, which can cause contamination of the packaged product and even cause a leak in the container. Even a low probability of coating failure may cause a significant number of containers to leak, given the large number of manufactured food and beverage containers.
[0005] Accordingly, it will be appreciated that what is needed in the art are improved coating compositions that are made without intentionally using bisphenol A and/or styrene, but which exhibit the stringent balance of coating properties to permit the use of such coating compositions on food or beverage containers.
SUMMARY
[0006] In one aspect, the invention provides a free-radical polymerized polymer that is preferably an acrylic polymer, more preferably an acrylic polymer that is substantially free of styrene. In preferred embodiments, the polymer preferably: (i) has a glass transition temperature of greater than 40.degree. C., more preferably from greater than 40.degree. C. to less than 100.degree. C., and more preferably from greater than 50.degree. C. to less than 80.degree. C. and/or (ii) is formed from ingredients including an ethylenically unsaturated monomer component that includes one or both of a monomer having a cyclic group or a monomer having a branched organic group. An acid- or anhydride-functional acrylic latex is preferred in some embodiments.
[0007] In another aspect, the invention provides an acrylic coating composition that is preferably substantially free of each of styrene (and preferably also substantially free of halogenated monomers) and bisphenol A (and preferably substantially free of each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof), and exhibits an enhanced elongation at break that is preferably comparable to a conventional styrene-containing acrylic coating. In preferred embodiments, the coating composition exhibits a sufficient amount of flexibility when cured to be suitable for use as an interior or exterior coating on an aluminum beverage can.
[0008] In one embodiment, the invention provides a coating composition that exhibits an elongation at break of at least 1% when suitably cured and tested as a free film. The coating composition preferably includes an emulsion polymerized latex polymer that is substantially free of each of styrene and halogenated monomers and preferably has a glass transition temperature of greater than 40.degree. C. The coating composition is preferably an aqueous coating composition that is suitable for use in forming a food-contact coating of a metal food or beverage can (e.g., an inside spray coating of an aluminum beverage can) and is substantially free of bisphenol A (and preferably substantially free of each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof).
[0009] In yet another aspect, the invention provides a coating composition that includes an acid- or anhydride-functional acrylic polymer (more preferably an acid or anhydride-functional acrylic latex) that is preferably substantially free of styrene and preferably has a glass transition temperature of greater than 40.degree. C. The coating composition preferably includes a carboxyl-reactive crosslinker, and more preferably a nitrogen-containing carboxyl-reactive crosslinker. The coating composition preferably includes a liquid carrier that includes one or both of water and an organic solvent. In preferred embodiments, the coating composition is a coating composition suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A (and preferably substantially free of each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof).
[0010] In yet another aspect, the invention provides a coating composition that includes an acid- or anhydride-functional acrylic polymer that is optionally substantially free of styrene (some embodiments may include styrene). The acid- or anhydride-functional acrylic polymer is preferably an acid- or anhydride-functional latex formed by emulsion polymerizing ethylenically unsaturated monomers comprising more than 5 wt- %, more than 6 wt- %, more than 7 wt- %, more than 8 wt- %, more than 9 wt- %, more than 10 wt- %, more than 11 wt- %, more than 12 wt- %, more than 13 wt- %, or more than 14 wt- % of multi-ethylenically unsaturated monomer. The coating composition preferably includes a carboxyl-reactive crosslinker (e.g., a nitrogen-containing carboxyl-reactive crosslinker). In preferred embodiments, the coating composition is an aqueous coating composition that is suitable for use in forming a food-contact coating (e.g., inside spray beverage can coating) of a metal food or beverage can and is substantially free of bisphenol A (and preferably substantially free of each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof).
[0011] In yet another aspect, the invention provides a method of coating a food or beverage can, or a portion thereof, including receiving a coating composition described herein and applying the coating composition on a metal substrate prior to, or after, forming the metal substrate into a food or beverage can or a portion thereof In some embodiments, the method includes spray applying the coating composition to an interior portion of a food or beverage can.
[0012] In yet another aspect, the invention provides an inside spray beverage can coating composition that comprises an aqueous coating composition that is preferably substantially free of each of styrene and halogenated monomers and is also preferably substantially free of bisphenol A (and more preferably substantially free of each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof). The coating composition preferably includes, based on total resin solids, at least 50 wt- % of an emulsion polymerized latex. In preferred embodiments, the inside spray beverage can coating composition, when spray applied onto an interior of a standard 12-ounce two-piece drawn and ironed aluminum 211 diameter beverage can at a dry film weight of 120 milligrams per can and cured at an oven temperature of at least 188.degree. C. to achieve a dome peak temperature of at least 199.degree. C., gives a metal exposure of less than 20 mA, less than 10 mA, or less than 3.5 mA when tested pursuant to the Metal Exposure after Drop Damage test disclosed herein.
[0013] In yet another aspect, the invention provides an article having a metal substrate, wherein at least a portion of the metal substrate has a coating disposed thereon formed from a coating composition of the present invention. In some embodiments, the article is a food or beverage can or a portion thereof. In certain preferred embodiments, the article is an aluminum beverage can having an inside spray coating disclosed herein on an interior surface.
[0014] The above summary of the present invention is not intended to describe each disclosed embodiment or every implementation of the present invention. The description that follows more particularly exemplifies illustrative embodiments. In several places throughout the application, guidance is provided through lists of examples, which can be used in various combinations. In each instance, the recited list serves only as a representative group and should not be interpreted as limiting or as an exclusive list.
[0015] The details of one or more embodiments of the invention are set forth in the description below. Other features, objects, and advantages of the invention will be apparent from the description and from the claims.
Selected Definitions
[0016] Unless otherwise specified, the following terms as used herein have the meanings as provided below.
[0017] As used herein, the term "organic group" means a hydrocarbon group (with optional elements other than carbon and hydrogen, such as oxygen, nitrogen, sulfur, and silicon) that is classified as an aliphatic group, a cyclic group, or combination of aliphatic and cyclic groups (e.g., alkaryl and aralkyl groups).
[0018] A group that may be the same or different is referred to as being "independently" something. Substitution on the organic groups of the compounds of the present invention is contemplated. As used throughout this application, the terms "group" and "moiety" are used to differentiate between chemical species that allow for substitution or that may be substituted and those that do not allow or may not be so substituted. Thus, when the term "group" is used to describe a chemical substituent, the described chemical material includes the unsubstituted group and that group with O, N, Si, or S atoms, for example, in the chain (as in an alkoxy group) as well as carbonyl groups or other conventional substitution. Where the term "moiety" is used to describe a chemical compound or substituent, only an unsubstituted chemical material is intended to be included. For example, the phrase "alkyl group" is intended to include not only pure open chain saturated hydrocarbon alkyl substituents, such as methyl, ethyl, propyl, t-butyl, and the like, but also alkyl substituents bearing further substituents known in the art, such as hydroxy, alkoxy, alkylsulfonyl, halogen atoms, cyano, nitro, amino, carboxyl, etc. Thus, "alkyl group" includes ether groups, haloalkyls, nitroalkyls, carboxyalkyls, hydroxyalkyls, sulfoalkyls, etc. On the other hand, the phrase "alkyl moiety" is limited to the inclusion of only pure open chain saturated hydrocarbon alkyl substituents, such as methyl, ethyl, propyl, t-butyl, and the like. As used herein, the term "group" is intended to be a recitation of both the particular moiety, as well as a recitation of the broader class of substituted and unsubstituted structures that includes the moiety.
[0019] The term "ethylenically unsaturated" refers to a carbon-carbon double or triple bond capable of participating in a free-radical initiated polymerization reaction, and is not intended to encompass the carbon-carbon double bonds present in aryl groups such as, for example, the phenyl group of styrene. Thus, for example, dodecyl benzene sulfonic acid is not considered to include an ethylenically unsaturated group.
[0020] The term "branched organic group" refers to a carbon-containing group that has a branching atom (e.g., carbon, nitrogen, silicon, or phosphorus) that is attached to at least three other atoms other than hydrogen, more typically at least three carbon-containing groups (e.g., --CR.sub.3, --OCR.sub.3, --NH--C(O)--O--CR.sub.3, and the like, where each R is independently any suitable atom or group such as a halogen, a hydrogen, an organic group, or a non-carbon-containing group (e.g., --OH, --NH.sub.2, etc.)), and even more typically at least three carbon atoms of at least three carbon-containing groups.
[0021] The term "branched alkyl group" refers to an alkyl group, which optionally includes one or more heteroatoms (e.g., O, N, P, Si, etc.), that includes at least one carbon-containing substituent group in place of a hydrogen (e.g., --CR.sub.3, --O--CR.sub.3, --NH--C(O)--O--CR.sub.3 and the like where each R is as described above). The term "branched alkyl moiety" refers to a branched alkyl group that does not include any heteroatoms.
[0022] The term "on" when used in the context of a coating applied on a surface or substrate, includes both coatings applied directly or indirectly to the surface or substrate. Thus, for example, a coating applied to a primer layer overlying a substrate constitutes a coating applied on the substrate.
[0023] Unless otherwise indicated, the term "polymer" includes both homopolymers and copolymers (e.g., polymers of two or more different monomers). Similarly, unless otherwise indicated, the use of a term designating a polymer class such as, for example, "acrylic" is intended to include both homopolymers and copolymers (e.g., polyether-acrylate copolymers).
[0024] The term "monomer" includes any reactant molecule used to produce a polymer, and encompasses both single-unit molecules (e.g., an acrylic molecule) and multi-unit molecules (e.g., an acrylic oligomer).
[0025] A group that may be the same or different is referred to as being "independently" something. The term "group" also encompasses single atom moieties. Thus, for example, a halogen atom can be a group.
[0026] The terms "acrylate" and "acrylic" are used broadly (and interchangeably) herein and encompass materials prepared from, for example, one or more of acrylic acid, methacrylic acid, or any acrylate or methacrylate compound. Thus, for example, a homopolymer consisting entirely of polymerized (meth)acrylic acid would still be an "acrylate" polymer even though no (meth)acrylate monomer was employed.
[0027] The term "(meth)" as used in "(meth)acrylate", "(meth)acrylic acid", and the like is intended to indicate that either a hydrogen or methyl group may be attached to the pertinent carbon atom of the monomer. For example "ethyl (meth)acrylate" encompasses both ethyl acrylate and ethyl methacrylate, as well as mixtures thereof.
[0028] The term "substantially free" when used with respect to a coating composition, or polymer or other composition, that may contain a particular compound means that the referenced composition contains less than 1,000 parts per million (ppm) of the recited compound whether the compound is mobile in the composition or bound to a constituent of the composition (e.g., as a structural unit of a polymer). The term "essentially free" when used with respect to a coating composition, or polymer or other composition, that may contain a particular compound means that the referenced composition contains less than 100 parts per million (ppm) of the recited compound. The term "essentially completely free" when used with respect to a coating composition, or polymer or other composition, that may contain a particular compound means that the referenced composition contains less than 5 parts per million (ppm) of the recited compound. The term "completely free" when used with respect to a coating composition, or a polymer or other composition, that may contain a particular compound means that the referenced composition contains less than 20 parts per billion (ppb) of the recited compound. When the phrases "free of" (outside the context of the aforementioned phrases), "does not include any" and the like are used herein, such phrases are not intended to preclude the presence of trace amounts of the pertinent structure or compound which may be present, e.g., as environmental contaminants.
[0029] As used herein, the term "styrene-free" indicates that styrene was not intentionally used, although trace amounts of contaminating styrene may be present (e.g., due to environmental contamination).
[0030] The terms "preferred" and "preferably" refer to embodiments that may afford certain benefits, under certain circumstances. However, other embodiments may also be preferred, under the same or other circumstances. Furthermore, the recitation of one or more preferred embodiments does not imply that other embodiments are not useful, and is not intended to exclude other embodiments from the scope of the invention.
[0031] The terms "comprises" and variations thereof do not have a limiting meaning where these terms appear in the description and claims.
[0032] As used herein, "a," "an," "the," "at least one," and "one or more" are used interchangeably. Thus, for example, a coating composition that comprises "a" surfactant can be interpreted to mean that the coating composition includes "one or more" surfactants.
[0033] Also herein, the recitations of numerical ranges by endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.). Furthermore, disclosure of a range includes disclosure of all subranges included within the broader range (e.g., 1 to 5 discloses 1 to 4, 1.5 to 4.5, 4 to 5, etc.).
DETAILED DESCRIPTION
[0034] The present invention provides an acrylic coating composition that provides a good balance of coating properties for food and beverage container coatings. Such properties include, for example, good adhesion, good flexibility, and good corrosion resistance. In preferred embodiments, the coating composition is formulated using an acrylic polymer that is styrene-free. In such preferred embodiments, the coating composition is also preferably styrene-free. In some embodiments, the acrylic resin system, and preferably also the coating composition, is also substantially free of substituted styrene compounds (e.g., alpha-methyl styrene, methyl styrenes (e.g., 2-methyl styrene, 4-methyl styrene, vinyl toluene, and the like), dimethyl styrenes (e.g., 2,4-dimethyl styrene), trans-beta-styrene, divinylbenzene, and the like).
[0035] Nonetheless, the various embodiments disclosed herein may optionally also, or alternatively, include styrene, although it is not presently preferred.
[0036] The use of styrene in can coating compositions, including inside spray beverage can coating compositions, has been advantageous for a variety of reasons, including, for example, because styrene possesses both a high level of hydrophobicity and a relatively high glass transition temperature ("Tg") and can contribute to substrate adhesion, which can positively affect coating flexibility. Prior attempts to replace styrene in acrylic food or beverage can coatings have often resulted in coating systems that either exhibit an unsuitable balance of coating properties for such end uses or exhibit one or more coating properties that are substantially diminished relative to conventional styrene-containing acrylic coating systems.
[0037] Acrylic can coatings have often suffered from flexibility problems and have generally been regarded as relatively inflexible as compared to other can coatings such as bisphenol-A-based epoxy can coatings. The relative inflexibility of acrylic resin systems tends to worsen with increasing Tg and, therefore, most conventional "high" Tg acrylics are unsuitable for use in can coatings, including inside spray beverage can coatings, due to insufficient flexibility for the end use. Moreover, omitting styrene from such "high" Tg acrylics further exacerbates the flexibility problems. The use of "high" Tg acrylic polymers, however, can be beneficial for purposes of achieving one or both of (i) decreased flavor scalping by the cured coating and (ii) enhanced chemical resistance by the cured coating. Surprisingly, preferred acrylic coating compositions of the present invention are capable of simultaneously exhibiting good adhesion, good flexibility (e.g., sufficient flexibility for use as an inside spray coating of a drawn and ironed aluminum beverage can), good corrosion resistance, and reduced flavor scalping without the use of any styrene and at "high" Tg's (e.g., acrylic polymers having a Tg>40.degree. C. or >60.degree. C.).
[0038] To achieve such improved coating properties without the use of styrene, in preferred embodiments, the acrylic coating composition of the present invention includes one or both of: (i) a carboxyl reactive crosslinker, more preferably a nitrogen-containing carboxyl-reactive crosslinker and (ii) an ethylenically unsaturated monomer having a cyclic group or a branched organic group.
[0039] In the discussion that follows, emphasis is placed on acrylic latexes and latex-based coatings, and particularly styrene-free acrylic latexes and latex-based coatings. It should be understood, however, that the teachings and disclosure contained herein may also be applied to acrylic polymers, and especially styrene-free acrylic polymers, that are not latexes and acrylic coatings that are not latex-based. Examples of such acrylic polymers that may not be latexes include organic-solution polymerized acrylics, which may or may not be water-dispersible.
[0040] While not intending to be bound by any theory, it is believed that the use of one, and more preferably both of, the above (i) and (ii) can result in a styrene-free acrylic coating, including a "high" Tg styrene-free acrylic latex based coating, capable of exhibiting enhanced elongation at break properties that can correlate to improved flexibility in food or can coating end uses, and particularly improved flexibility for inside spray beverage can coatings. Assuming that other requisite coating properties are present (e.g., suitable coating adhesion), it is believed that a suitably high elongation at break can correlate to suitable coating flexibility in end use specific food or beverage can coating tests such as the "Metal Exposure after Drop Damage" test described herein. Thus, in some embodiments, the styrene-free acrylic latex based coatings of the present invention preferably exhibit an elongation at break value that is comparable to that of a reference styrene-containing acrylic latex based coating. An example of such a reference styrene-containing acrylic latex based coating is Comparative Example 5 in the Examples section below.
[0041] Moreover, it is contemplated that the use of, for example, one or more preferred crosslinkers provided herein may even result in a styrene-free coating having an improved elongation at break relative to such a reference styrene-containing coating.
[0042] While it is contemplated that the styrene-free coatings of the present invention can exhibit any suitable elongation at break values, in some embodiments, the coatings, when evaluated as suitable cured free films, preferably exhibit an elongation at break percent of at least 1%, more preferably at least 1.5%, even more preferably at least 2%, and even more preferably at least 3%. In some embodiments, the elongation at break of the coating compositions, when evaluated as suitable cured free films, are greater than 5%, greater than 10%, and in some instances even 15% or more (e.g., .gtoreq.15%, >20%, or .gtoreq.30%). The elongation at break is not restricted on the upper end, but may be, for example, less than 100%, 80%, 50%, 40%, 30%, or 20%. A suitable methodology for assessing elongation at break is disclosed later herein.
[0043] While not wishing to be bound by any theory, it is believed that the beneficial effects of the above (i) (e.g., nitrogen-containing carboxyl-reactive crosslinker) may alternatively be achieved using certain internal crosslinking moieties within the polymer itself to achieve suitable flexibility improvements at high "Tg" without requiring the use of styrene. Thus, for example, it is believed that a coating composition that exhibits the above minimum elongation break values can be achieved without using a nitrogen-containing carboxyl-reactive crosslinker described herein if suitable internal cross-link density is built into the polymer. For example, monomers such as allyl methacrylate, glycidyl methacrylate, multi-ethylenically unsaturated monomers (e.g., di(meth)acrylates), etc. may be useful in creating such internal crosslinks.
[0044] The polymer is preferably a free-radical polymerized polymer formed from ingredients including an ethylenically unsaturated monomer component, which may be polymerized in one or more stages, which may be of a same or different monomer composition. More preferably, the polymer is an acrylic polymer such as, for example, an organic solution polymerized acrylic polymer (which may optionally be water-dispersible) or an acrylic latex polymer, and more preferably an acrylic latex polymer that may optionally include a water-dispersible polymer such as, for example, a water-dispersible organic solution polymerized acrylic polymer. In a preferred embodiment, the polymer is a latex polymer formed by emulsion polymerizing an ethylenically unsaturated monomer component in the presence of a water-dispersible polymer (e.g., a polymeric surfactant).
[0045] The inventors have surprisingly discovered that the use of certain nitrogen-containing carboxyl-reactive ("NCCR") crosslinkers can substantially improve the flexibility of acrylic food or beverage can coatings, including acrylic coatings having a relatively "high" Tg (e.g., Tg greater than 60.degree. C.). This was surprising because conventional crosslinkers used in can coatings are generally not capable of providing large improvements in coating flexibility when used to formulate acrylic coatings. Consistent with this, the substantial improvement in coating flexibility was not observed for other more conventional crosslinkers. For example, resole phenolic crosslinkers, which are often used to formulate acrylic can coatings, were not capable of yielding a comparable flexibility improvement. In addition, the use of NCCR crosslinkers such as, e.g., hydroxyalkylamide crosslinkers allows for production of a formaldehyde-free acrylic coating composition having sufficient flexibility for use as an interior or exterior coating of a food or beverage can.
[0046] The NCCR crosslinker can have any suitable combination of one or more carboxyl-reactive functional groups, and more preferably includes two or more such groups. Hydroxyl groups are preferred carboxyl-reactive groups. Other suitable carboxyl-reactive groups may include thiol groups. In some embodiments, the NCCR includes two or more, three or more, or four or more hydroxyl groups.
[0047] The NCCR crosslinker can include any suitable number of nitrogen atoms, although it will typically include two or more nitrogen atoms, and, in some embodiments, two total nitrogen atoms. In some embodiments, one or more (and more preferably two or more) nitrogen atoms are present in an amide group, an aziridine group, an imide group, a diimide group, an oxazoline group, a urethane group, or a combination thereof. In a preferred embodiment, the NCCR crosslinker includes two or more amide groups. It is contemplated, however, that the NCCR crosslinker may contain a single amide group such as, for example, a poly-substituted amide group having two or more hydroxyl groups.
[0048] In certain preferred embodiments, the NCCR crosslinker includes one or more, and more preferably two or more, groups having the structure of the below Formula (I):
HO--R.sup.1--N(R.sup.2)--C(.dbd.O)--
wherein each R.sub.1 is independently an organic group, and each R.sub.2 is independently hydrogen or an organic group.
[0049] As shown in Formula (I), the depicted hydroxyl group can be a primary hydroxyl group, secondary hydroxyl group, or tertiary hydroxyl group depending on the structure of R.sup.1. In some embodiments, the hydroxyl group is a primary hydroxyl group.
[0050] R.sup.1 can include any suitable number of carbon atoms, but will typically include from 2 to 10 carbons atoms, more typically from 2 to 8 carbon atoms, more typically from 2 to 6 carbons atoms, and even more typically from 2 to 4 carbon atoms. le will typically include at least two carbon atoms in a chain connected on one end to the depicted nitrogen atom and on the other end to the depicted hydroxyl group. In an embodiment, the depicted hydroxyl group is attached directly to a first carbon atom, which is attached directly to a second carbon, which is in-turn attached directly to the depicted nitrogen atom. In some embodiments R.sup.1, is --(CH.sub.2).sub.2--.
[0051] In some embodiments, R.sup.1 is an alkylene group preferably containing from 1 to 5 carbon atoms (e.g., methylene, ethylene, n-propylene, sec-propylene, n-butyl, sec-butylene, tert-butylene, pentylene, etc.).
[0052] In some embodiments R.sup.2 is an organic group that includes a hydroxyl group. In some such embodiments, R.sup.2 is of the formula HO--R.sup.1--, wherein le is as described above. Examples of such R.sup.2 groups include hydroxyl alkyl groups preferably having from 1 to 5 carbon atoms (e.g., hydroxy-ethyl, 3-hydroxy-propyl, 2-hydroxy-propyl, 4-hydroxy-butyl, 3-hydroxy-butyl, 2-hydroxy-2-propyl-methyl, 5-hydroxy-pentyl, 4-hydroxy-pentyl, 3-hydroxy-pentyl, 2-hydroxy-pentyl and the pentyl isomers). An example of an NCCR crosslinker including such an R.sup.2 group is provided below (which is believed to be the structure of the PRIMID XL-552 product commercially available from EMS):
##STR00001##
[0053] In some embodiments, the NCCR crosslinker is a compound having the structure of the below Formula (II):
(HO--R.sup.1--N(R.sup.2)--C(.dbd.O)).sub.n--X,
wherein:
[0054] R.sup.1 and R.sup.2 are as described above,
[0055] n is 2 or more, and
[0056] X is a polyvalent organic group.
[0057] In some embodiments, X is an alkylene group. In some embodiments, X is a --(CH.sub.2).sub.m-- group wherein (i) m is 1 or more, 2 or more, 3 or more, 4 or more, and more typically from 2 to 10 and (ii) one or more hydrogens may be replaced with substituent groups (e.g., organic substituent groups). In an embodiment, X is --(CH.sub.2).sub.4--.
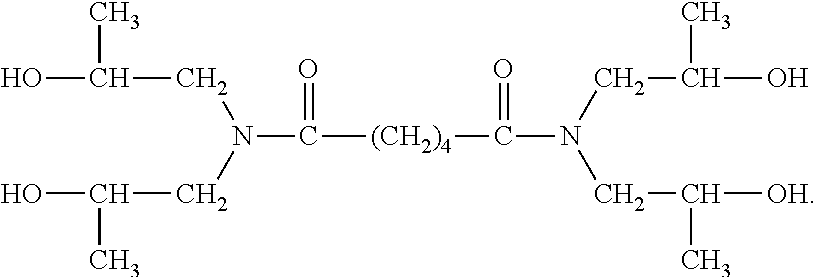
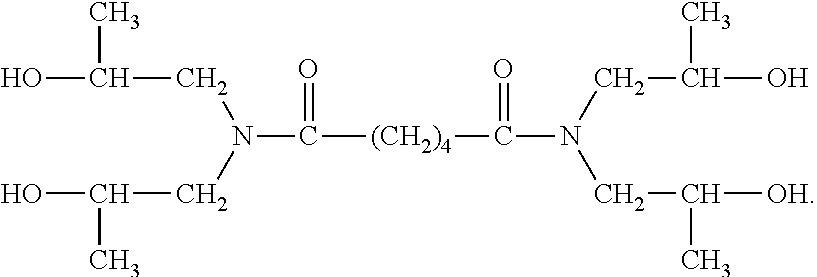
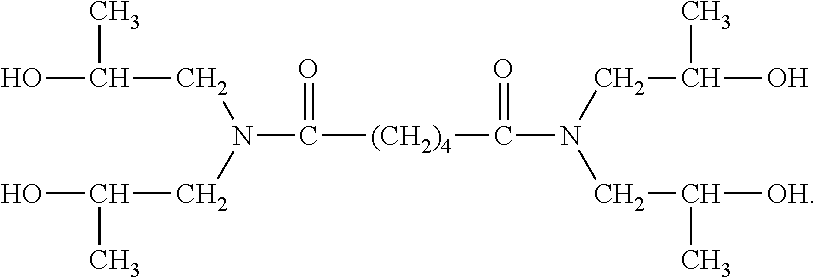
[0058] In certain preferred embodiments, the hydroxyl group is located "beta" relative to a nitrogen atom, more preferably a nitrogen atom of an amide bond. Thus, for example, in certain preferred embodiments the NCCR crosslinker is a beta-hydroxyalkylamide compound. Some examples of such compounds include: bis[N,N-di(.beta.-hydroxy-ethyl)]adipamide, bis[N,N-di(.beta.-hydroxy-propyl)]succinamide, bis[N,N-di(.beta.-hydroxy-ethyl)]azelamide, bis[N,N-di(.beta.-hydroxy-propyl)]adipamide, bis[N-metil-N-(.beta.-hydroxy-ethyl)]oxamide, and mixtures thereof. The PRIMID QM-1260 product commercially available from EMS is an example of a preferred beta-hydroxyalkylamide crosslinker. The structure believed to correspond to the PRIMID QM-1260 product is provided below:
##STR00002##
[0059] Without intending to be bound by theory, the use of beta-hydroxyalkylamides is preferred in certain embodiments due to the formation of an oxazolinium intermediate that is believed to occur and result in enhanced reactivity of the crosslinker with carboxyl groups. Thus, in some embodiments, the NCCR crosslinker is preferably capable of forming an oxazolinium intermediate or other carbon-nitrogen heterocyclic intermediate having enhanced reactivity with carboxyl groups. Preferably, such reactive intermediates are formed under typical food or beverage can coating thermal cure conditions. For example, for beverage inside spray coatings, such reactive intermediates are preferably formed at oven bake conditions of from 188 to 199.degree. C. during an oven residence time of 30 to 85 seconds.
[0060] The NCCR crosslinker is preferably formed from reactants that do not include formaldehyde.
[0061] Although in presently preferred embodiments the NCCR crosslinkers described herein are used in combination with a styrene-free acrylic resin system (e.g., a styrene-free acrylic latex), it is also contemplated that the NCCR may be used in conjunction with styrene-containing acrylic resin systems (not presently preferred) to improve one or more coating properties of coatings formulating using such styrene-containing resin systems.
[0062] As previously discussed, in some embodiments, one or more branched or cyclic monomers are used in place of styrene, alone or in combination with one or more other monomers (e.g., one or more non-branched or non-cyclic (meth)acrylates) to provide a styrene-free acrylic polymer that, when suitably formulated, provides comparable coating properties in food or beverage can coatings to conventional styrene-containing acrylic formulations. For purposes of convenience, hereinafter an ethylenically unsaturated monomer having a branched organic group is referred to as a "branched monomer" and an ethylenically unsaturated monomer having a cyclic group is referred to as a "cyclic monomer." An ethylenically unsaturated monomer that, when incorporated into the acrylic polymer, does not provide a pendant branched group is not considered a branched monomer. Thus, as used herein, methyl methacrylate is not considered to be a branched monomer because it does not provide a pendant branched group when incorporated into an acrylic polymer. Stated otherwise, methyl methacrylate is not considered herein to be a branched monomer because it does not provide a pendant group having at least one branching atom that is not present in the polymer backbone.
[0063] In some embodiments, an ethylenically unsaturated monomer component used to form the acrylic polymer includes both one or more branched monomers and one or more cyclic monomers. Similarly, in some embodiments, the ethylenically unsaturated monomer component includes one or more monomers that include both a branched group and a cyclic group.
[0064] The branched and/or cyclic monomer can be any suitable monomer. Preferably, the monomer is capable of being incorporated into a polymer, such as, for example, an acrylic polymer, via a free-radical polymerization process.
[0065] In some embodiments, the branched and/or cyclic monomer is a vinyl ester monomer.
[0066] A branched and/or cyclic monomer having any suitable Tg may be used. The selection of a branched or cyclic monomer having a particular Tg value may be influenced by a variety of factors including the end use of the coating composition (e.g., whether the coating is intended for an exterior or interior of a can) and the Tg of the other monomers selected. Typically, the branched and/or cyclic monomer will have a Tg greater than -10.degree. C., more typically greater than 0.degree. C.
[0067] In some embodiments, branched monomers having Tg's as low as about -3.degree. C. (e.g., the VeoVa 10 monomer product commercially available from Hexion) or even as low as about -40.degree. C. (e.g., the VeoVa 11 monomer product commercially available from Hexion) may be used. If used, such "low" Tg monomers will typically be used in combination with one or more "high" Tg monomers, such as one or more "high" Tg branched or cyclic monomers (e.g., Tg>40.degree. C.).
[0068] In embodiments in which the branched and/or cyclic monomer is intended as at least a partial replacement for styrene, such as for an interior food-contact can coating, the branched and/or cyclic monomer preferably has a glass transition temperature ("Tg") that is sufficiently high to offset the replaced styrene. Thus, in some embodiments, the branched and/or cyclic monomer preferably has a Tg >40.degree. C., more preferably >50.degree. C., even more preferably >60.degree. C. , and optimally >70.degree. C. Although the upper Tg is not restricted, in some embodiments, the branched and/or cyclic monomer has a Tg <110.degree. C., <95.degree. C., <85.degree. C., or <75.degree. C. For branched and/or cyclic monomers specifically referenced herein, any Tg values provided herein for such monomers should be used for comparison relative to the above Tg thresholds. For a branched and/or cyclic monomers not having a reported Tg value herein, in the absence of a reliable Tg value reported by a manufacturer of the monomer, the Tg of the monomer may be determined by making a homopolymer having a number average molecular weight of at least about 4,000 and a suitable polydispersity index (e.g., preferably less than 3 and ideally as low as possible) and measuring the Tg of the homopolymer using a suitable procedure such as the procedure included in the test methods section below.
[0069] Any suitable cyclic monomer or combination of cyclic monomers may be used including, for example, vinyl aromatics compounds, vinyl alicyclic compounds, and combinations thereof. If a vinyl aromatic monomer is used, it preferably is not styrene or a substituted styrene. In some embodiments, the coating composition is substantially free of vinyl aromatic compounds. In some embodiments, the acrylic polymer (e.g., emulsion polymerized acrylic latex) is substantially free of cyclic-group-containing vinyl monomers (e.g., certain embodiments such as, e.g., certain embodiments when methyl (meth)acrylate is employed).
[0070] The cyclic monomers may include any suitable number of cyclic groups, which may be monocyclic groups or polycyclic groups and may be saturated or unsaturated. The atoms in the ring(s) of the one or more cyclic groups may be all carbon atoms or may include one or more heteroatoms (e.g., N, O, P, Si, etc.). Similarly, the rings may be of any suitable size and may, for example, include 3 to 13 atoms in the ring, more typically 4 to 9 atoms in the ring, and even more typically 4 to 6 atoms in the ring. In some embodiments, the ring of the cyclic group is a C4 ring (e.g., cyclobutane), a C5 ring (e.g., cyclopentane), or a C6 ring (e.g., cyclohexane). Cyclohexane groups, which may optionally include one or more substituents in place of hydrogen, are preferred cyclic groups in some embodiments.
[0071] Examples of suitable cyclic monomers may include benzyl (meth)acrylate, cyclohexyl (meth)acrylate, isobornyl (meth)acrylate, phenyl (meth)acrylate, substituted variants thereof (e.g., 3,3,5-trimethylcyc1ohexyl (meth)acrylate and 4-tert-butylcyclohexyl (meth)acrylate), and mixtures thereof. Cyclohexyl methacrylate is a preferred cyclic monomer, and may be used to replace styrene in some embodiments.
[0072] Any suitable branched monomer or combination of branched monomers may be used. Preferred branched monomers include branched organic groups such as, for example, branched hydrocarbon groups, with branched alkyl groups being preferred in certain embodiments. The branched organic group may optionally include one or more heteroatoms (e.g., O, N, P, Si, etc.). In certain preferred embodiments, the branched organic group includes one or more, two or more, or even three or more branching atoms (preferably tertiary or quaternary carbon atoms). Although the branched organic group (inclusive of any carbon branching atoms) can include any suitable number of carbon atoms, typically it will include 3 or more, 4 or more, 5 or more, or 6 or more total carbon atoms. While the upper number of carbon atoms is not restricted, typically the branched organic group will include 18 or less, 13 or less, or 10 or less carbon atoms (see, e.g., branched organic group "Y" of Formula (III)) described herein). If the branched organic group is connected to the ethylenically unsaturated group via a heteroatom-containing linkage (e.g., linkages including at least one or more heteroatoms such as N, O, P, S, etc.) the carbon atoms of the heteroatom-containing linkage are not counted as being part of the branched organic group. Examples of suitable heteroatom-containing linkages include, for example, those formed by reacting two complimentary reactive functional groups (e.g., --OH and --COOH) such as are used, for example, to produce condensation linkages and the like. Example of suitable heteroatom containing-linkages include amide, carbonate ester, ester, ether, urea, and urethane linkages.
[0073] In some embodiments, the branched organic group is a branched C3 to C13 alkyl group or moiety, more preferably a branched C4 to C10 alkyl group or moiety.
[0074] The branched organic group may optionally include one or more cyclic groups. In some embodiments, the branched organic group includes one or more branching atoms (e.g., tertiary or quaternary carbon atoms) in a ring of the cyclic group or in a location other than the ring. Some examples of such compounds include 3,3,5-trimethylcyclohexyl (meth)acrylate (branching atom, in the form of a quaternary carbon atom, included in an aliphatic ring) and 4-tert-butylcyclohexyl (meth)acrylate (branching atom, in the form of quaternary carbon atom, attached to an aliphatic ring).
[0075] In some embodiments, the branched organic group (and optionally the branched monomer) does not include any cyclic groups. Thus, in some embodiments, the branched organic group is a branched, open-chain alkyl group (e.g., isopropyl, sec-butyl, isobutyl, tert-butyl, etc).
[0076] In some embodiments, the ethylenically unsaturated monomer component includes one or more branched and/or cyclic monomers of the below Formula (III):
(R.sup.3).sub.2--C.dbd.C(R.sup.4)--W.sub.n--Y,
wherein: [0077] R.sup.3 is independently selected from hydrogen or an organic group; [0078] R.sup.4 is selected from hydrogen or an alkyl group; [0079] W, if present, is a divalent linking group; [0080] n is 0 or 1, more typically 1; and [0081] Y is: (i) a branched organic group including one or more branching atoms, more typically one or more branching carbon atoms, (ii) a cyclic group (e.g., any of the cyclic groups disclosed herein), or (iii) a combination of (i) and (ii) (e.g., such as present in 4-tert-butylcyclohexyl (meth)acrylate).
[0082] The branched and/or cyclic monomer of Formula (III) can be either a vinyl monomer or an olefin monomer depending upon the R.sup.3 and R.sup.4 selections. In preferred embodiments, the monomer of Formula (III) is a vinyl monomer (e.g., a (meth)acrylate or vinyl ester) and both R.sup.3 are hydrogen.
[0083] In preferred embodiments, R.sup.4 is hydrogen, a methyl moiety (--CH.sub.3), or an ethyl moiety, more preferably hydrogen or a methyl moiety.
[0084] When present, W is typically a heteroatom-containing linkage such as, for example, any of those previously discussed. Examples of suitable such linkages include amide, carbonate, ester, ether, urea, and urethane. Ester linkages of either directionality (--C(O)--O-- or --O--C(O)--) are preferred such linkages. In some embodiments, W is an ester linkage and the carbonyl carbon of the ester is attached to a carbon atom of Y.
[0085] In some embodiments, Y is a branched organic group having the structure of the below Formula (IV):
--C(CH.sub.3).sub.t(R.sup.5).sub.3-t
wherein: [0086] t is 0 to 3; [0087] each R.sup.5, if present, is independently an organic group that may optionally be itself branched, more typically an alkyl group that may optionally include one or more heteroatoms (e.g., N, O, P, Si, etc.); and [0088] two or more R.sup.5 may optionally form a cyclic group with one another.
[0089] In some embodiments, t is 3. Tert-butyl acrylate and tert-butyl methacrylate are two examples of a branched monomer of Formula (III) in which "Y" has a structure of Formula (IV) and t is 3.
[0090] In some embodiments, t is 1 and the total number of carbon atoms present in both R.sup.5 groups is 6, 7, or 8. Examples of such branched monomers include the VEOVA 9 (Tg 70.degree. C.), VEOVA 10 (Tg -3.degree. C.), and VEOVA 11 (Tg -40.degree. C.) monomers commercially available from Hexion.
[0091] In some embodiments in which the monomer of Formula (III) includes a Y group of Formula (IV), t is 0, 1, or 2, and Y includes at least one R.sup.5 group that is itself a branched organic group, more typically a branched alkyl group. Thus, for example, in some embodiments, at least one R.sup.5 is present that includes a tertiary or quaternary carbon atom. The VEOVA 9 monomer is an example of such a branched monomer.
[0092] Additional examples of suitable branched monomers may include isopropyl methacrylate, isobutyl methacrylate, sec-butyl methacrylate, and mixtures thereof.
[0093] In certain preferred embodiments, Y includes at least one carbon atom that functions as a branching point (e.g., the leftmost carbon atom depicted in Formula (IV)). Nonetheless, it is contemplated that the branched organic group may include a branching atom(s) other than carbon such as, for example, P, Si, or N. Such a branching heteroatom may be used in place of, or in addition to, a branching carbon atom in Y.
[0094] In some embodiments, the branched organic group is provided by neodecanoic acid and/or neononanoic acid. In one embodiment, the branched monomer is a vinyl ester of neodecanoic acid or neononanoic acid.
[0095] The ethylenically unsaturated monomer component (e.g., an ethylenically unsaturated monomer component polymerized in the presence of a polymeric and/or non-polymeric surfactant(s) to form a latex) can include any suitable amount of one or more branched and/or cyclic monomers. The amount of branched and/or cyclic monomer employed can vary depending upon a variety of factors such as, for example, the other monomer(s) present, the desired Tg, and the desired end use including, e.g., the desired amount of coating flexibility and/or corrosion resistance. In some embodiments, one or both of: (i) the ethylenically unsaturated monomer component or (ii) an acrylic-containing resin system formed, at least in part, from the ethylenically unsaturated monomer component (e.g., a latex formed by emulsion polymerizing the ethylenically unsaturated component in the presence of a water-dispersible polymer) includes at least 10 weight percent ("wt- %"), more preferably at least 20 wt- %, even more preferably at least 30 wt- %, and in some embodiments 40 wt- % or more of one or more branched and/or cyclic monomers. Although the upper amount of branched and/or cyclic monomers is not restricted, typically the ethylenically unsaturated monomer component will include less than 90 wt- %, more typically less than 70 wt- %, more typically less than 60 wt- %, and even more typically less than 50 wt- % of one or more branched and/or cyclic monomers. In some such embodiments, one or more branched monomers are present in an above recited amount, while either no cyclic monomers are present or one or more cyclic monomers are present in an additional amount. In some such embodiments, one or more cyclic monomers are present in an above recited amount, while either no branched monomers are present or one or more branched monomers are present in an additional amount.
[0096] In some embodiments in which the polymer is an emulsion polymerized latex, the overall latex particle or polymer preferably includes an amount of one or more branched and/or cyclic monomers pursuant to the amounts disclosed in the preceding paragraph. For example, in certain embodiments where the ethylenically unsaturated monomer component is polymerized in the presence of a water-dispersible acrylic polymer (e.g., an acid- or anhydride-functional organic solution polymerized acrylic polymer), the overall latex particle or polymer includes an amount of one or more branched and/or cyclic monomers in such amounts.
[0097] It is contemplated that, in some embodiments, the ethylenically unsaturated monomer component may not include one or more branched and/or cyclic monomers, but rather, may include a reactive functional group through which a branched and/or cyclic organic group may be subsequently grafted. For example, a polymer having pendant reactive groups could be post-modified with a branched and/or cyclic reactive compound (e.g., a compound including a "Y" branched and/or cyclic group and a reactive functional group capable of forming a "W" divalent linkage) to include one or more branched organic groups disclosed herein. For example, the hydroxyl groups of a latex polymer could be reacted with neodecanoic acid and/or neononanoic acid to provide branched organic groups thereon.
[0098] As previously discussed, in certain preferred embodiments, the polymer is a latex polymer. The latex polymer is preferably formed by emulsion polymerizing the ethylenically unsaturated monomer component in an aqueous medium. The latex can be a single-stage latex or a multi-stage latex. The ethylenically unsaturated monomer component is typically emulsion polymerized in the aqueous medium in the presence of at least one surfactant (or emulsifier), which can be polymeric, non-polymeric, or a blend thereof.
[0099] The latex polymer may be of any suitable molecular weight. In some embodiments, the latex polymer has a number average molecular weight (M.sub.n) of at least about 30,000, at least about 100,000, at least about 200,000, or at least about 300,000. The upper range of the M.sub.n of the latex polymer is not restricted and may be 1,000,000 or more. In certain embodiments, however, the M.sub.n of the latex polymer is less than about 1,000,000, less than about 600,000, or less than about 400,000. Because in some embodiments the molecular weight may be too high to measure (e.g., via GPC analysis using polystyrene standards), it may be necessary to determine the number average molecular weight via theoretical calculation.
[0100] Preferably, at least 5 wt- %, more preferably at least 25 wt- %, even more preferably at least 40 wt- %, even more preferably at least 50 wt- %, and even more preferably at least 55 wt- % or at least 60 wt- %, of the ethylenically unsaturated monomer component is used in making the latex polymer. In some embodiments, the ethylenically unsaturated monomer component may comprise up to about 100 wt- %, up to 98 wt- %, up to 95 wt- %, up to 80 wt- %, or up to 70 wt- % of the latex polymer. Such percentages are based on the total combined weight of the ethylenically unsaturated component and any polymeric or polymerizable surfactant used to make the latex.
[0101] In embodiments in which a water-dispersible polymer is used to facilitate emulsion polymerization of the latex polymer (e.g., as a "polymeric surfactant"), preferably no greater than 95 wt- %, more preferably no greater than 90 wt- %, and even more preferably no greater than 85 wt- % of the ethylenically unsaturated monomer component is used in making the latex polymer. Such percentages are based on the total weight of ethylenically unsaturated monomer component and water-dispersible polymer used to make the latex.
[0102] In preferred embodiments, the ethylenically unsaturated monomer component is a mixture of monomers, more preferably a mixture of monomers that that includes at least one (meth)acrylate monomer. Any combination of one or more (meth)acrylates may be included in the ethylenically unsaturated monomer component, including, for example, combinations of one or more branched and/or cyclic (meth)acrylate monomers and one or more other (meth)acylate monomers (e.g., "linear" alkyl (meth)acrylates) optionally in combination with one or more non-(meth)acrylate monomers.
[0103] Suitable (meth)acrylates include any of those referenced herein, as well as those having the structure: CH.sub.2.dbd.C(R.sup.6)--CO--OR.sup.7 wherein R.sup.6 is hydrogen or methyl, and R.sup.7 is an alkyl group preferably containing one to sixteen carbon atoms, a cycloaliphatic group, an aryl group, a silane group, or a combination thereof. If desired, R.sup.7 may optionally be substituted with one or more (e.g., one to three) moieties such as hydroxy, halo, phenyl, and alkoxy, for example. Examples of suitable (meth)acrylates (including, e.g., suitable alkyl (meth)acrylates) include methyl (meth)acrylate, ethyl (meth)acrylate, propyl (meth)acrylate, isopropyl (meth)acrylate, butyl (meth)acrylate, isobutyl (meth)acrylate, pentyl (meth)acrylate, isoamyl (meth)acrylate, hexyl (meth)acrylate, 2-ethylhexyl (meth)acrylate, cyclohexyl (meth)acrylate, decyl (meth)acrylate, isodecyl (meth)acrylate, benzyl (meth)acrylate, phenyl (meth)acrylate, lauryl (meth)acrylate, isobornyl (meth)acrylate, octyl (meth)acrylate, nonyl (meth)acrylate, hydroxyethyl (meth)acrylate, hydroxypropyl (meth)acrylate, and the like, substituted variants thereof (e.g., ring substituted variants of benzyl (meth)acrylate or phenyl (meth)acrylate), and isomers and mixtures thereof.
[0104] Typically, (meth)acrylates will constitute a substantial portion of the ethylenically unsaturated monomer component. In some embodiments, (meth)acrylates may constitute at least 20 wt- %, at least 30 wt- %, at least 50 wt- %, at least 70 wt- %, at least 95 wt- %, or even 99 wt- % or more of the ethylenically unsaturated monomer component. The aforementioned weight percentages include all (meth)acrylates monomers present in the ethylenically unsaturated monomer component, regardless of whether one or more of the monomers may also qualify as a branched monomer, a cyclic monomer, or both.
[0105] In preferred embodiments, the ethylenically unsaturated monomer component includes at least one "linear" alkyl (meth)acrylate having a linear (e.g., non-branched) alkyl group. Examples of such linear groups include the following moieties: methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, etc. Preferred such monomers include one or more of methyl (meth)acrylate, ethyl (meth)acrylate, n-propyl (meth)acrylate, and n-butyl (meth)acrylate, with acrylate forms thereof being particularly preferred in certain embodiments. In some embodiments, the ethylenically unsaturated monomer component includes at least 10 wt- %, more preferably at least 15 wt- %, and even more preferably at least 20 wt- % of one or more linear alkyl (meth)acrylates. When present, linear alkyl (meth)acrylates preferably constitute less than 90 wt- %, more preferably less than 80 wt- %, and even more preferably less than 70 wt- % of the ethylenically unsaturated monomer component.
[0106] One or more multi-functional monomers may optionally be included in the ethylenically unsaturated monomer component. Examples of preferred multi-functional monomers include multi-ethylenically unsaturated monomers such as multi-ethylenically-unsaturated (meth)acrylates. Examples of multi-ethylenically unsaturated (meth)acrylates include polyhydric alcohol esters of acrylic acid or methacrylic acid, such as ethanediol di(meth)acrylate, propanediol di(meth)acrylate (e.g. 1,2-propanediol di(meth)acrylate and 1,3-propanediol di(meth)acrylate), butanediol di(meth)acrylate (e.g., 1,3-butanediol di(meth)acrylate and 1,4-butanediol di(meth)acrylate), heptanediol di(meth)acrylate, hexanediol di(meth)acrylate, trimethylolethane tri(meth)acrylate trimethylolpropane tri(meth)acrylate, trimethylolbutane tri(meth)acrylate, trimethylolheptane tri(meth)acrylate, trimethylolhexane tri(meth)acrylate, tetramethylol methane tetra(meth)acrylate, dipropylene glycol di(meth)acrylate, trimethylol hexane tri(meth)acrylate, pentaerythritol tetra(meth)acrylate, isosorbide di(meth)acrylate, allyl (meth)acrylate, glycerol dimethacrylate, isomers thereof, and mixtures thereof. Examples of multi-ethylenically-unsaturated monomers other than (meth)acrylates include diallyl phthalate, divinylbenzene, divinyltoluene, divinylnaphthalene, and mixtures thereof
[0107] In one embodiment, 1,4-butane diol di(meth)acrylate is included in the ethylenically unsaturated monomer component.
[0108] In some embodiments, the ethylenically unsaturated monomer component includes at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, at least 10%, at least 11%, at least 12%, at least 13%, or at least 14% by weight of multi-ethylenically unsaturated monomer. If used, such multi-ethylenically unsaturated monomers will typically be included in the ethylenically unsaturated monomer component in an amount of less than about 25 wt- %, more typically less than about 20 wt- %, and even more typically less than about 17.5 wt- %. In some embodiments, di(meth)acrylates are preferred multi-ethylenically unsaturated monomers.
[0109] In some embodiments, the ethylenically unsaturated monomer component includes at least one oxirane-functional monomer, more preferably at least one oxirane-functional alpha, beta-unsaturated monomer. The ethylenically unsaturated monomer component preferably contains no greater than 30 wt- %, more preferably no greater than 20 wt- %, even more preferably no greater than 10 wt- %, and optimally no greater than 9 wt- %, of the oxirane group-containing monomer. In some embodiments, the ethylenically unsaturated monomer component includes more than 1 wt- %, more than 2 wt- %, more than 3 wt- %, or 5 or more wt- % of oxirane-group containing monomer.
[0110] Suitable oxirane-functional monomers include monomers having a reactive carbon-carbon double bond and an oxirane (viz., a glycidyl) group. Typically, the monomer is a glycidyl ester of an alpha, beta-unsaturated acid, or anhydride thereof (viz., an oxirane group-containing alpha, beta-ethylenically unsaturated monomer). Suitable alpha, beta-unsaturated acids include monocarboxylic acids or dicarboxylic acids. Examples of such carboxylic acids include, but are not limited to, acrylic acid, methacrylic acid, alpha-chloroacrylic acid, alpha-cyanoacrylic acid, beta-methylacrylic acid (crotonic acid), alpha-phenylacrylic acid, beta-acryloxypropionic acid, sorbic acid, alpha-chlorosorbic acid, angelic acid, cinnamic acid, p-chlorocinnamic acid, beta-stearylacrylic acid, itaconic acid, citraconic acid, mesaconic acid, glutaconic acid, aconitic acid, maleic acid, fumaric acid, tricarboxyethylene, maleic anhydride, and mixtures thereof.
[0111] Specific examples of suitable monomers containing a glycidyl group are glycidyl (meth)acrylate (viz., glycidyl methacrylate and glycidyl acrylate), mono- and di-glycidyl itaconate, mono- and di-glycidyl maleate, and mono- and di-glycidyl formate. It also is envisioned that allyl glycidyl ether and vinyl glycidyl ether can be used as the oxirane-functional monomer. Preferred monomers are glycidyl acrylate and glycidyl methacrylate ("GMA"), with GMA being particularly preferred in some embodiments.
[0112] In embodiments in which the ethylenically unsaturated monomer component includes an oxirane-functional monomer, it may be advantageous to employ an amine, more preferably a tertiary amine, in preparing the latex or other acrylic polymer. Without intending to be bound by theory, under preferred conditions an acid group (e.g., present in the ethylenically unsaturated monomer component and/or a water-dispersible polymer), an oxirane group, and an amine (particularly a tertiary amine) are believed to form a quaternary salt linkage. This linkage is favored, as it not only links the polymers but promotes water dispersibility of the joined polymer and can enhance the mechanical properties of a coating composition including the latex polymer. It should be noted that an acid group and an oxirane group may also form an ester. Some of this reaction is possible, though this linkage is less desirable when water dispersibility is sought.
[0113] In some embodiments, the ethylenically unsaturated monomer component does not include any monomers having oxirane groups.
[0114] In some embodiments (e.g., when the ethylenically unsaturated monomer component is emulsion polymerized), the ethylenically unsaturated monomer component may include a minority amount (e.g., less than 30 wt- %, less than 25 wt- %, less than 20 wt- %, less than 15 wt- %, less than 10 wt- %, less than 5 wt- %, less than 2 wt- %, or less than 1 wt- %) of acid- or anhydride-functional ethylenically unsaturated monomer. Examples of suitable such acid- or anhydride-functional monomers may include any of those disclosed herein. When present, the acid- or anhydride-functional monomer are typically present in an amount of more than 1 wt- %, more than 2 wt- %, more than 3 wt- %, more than 5 wt- %, or even more than 10 wt- %, based on the total weight of the ethylenically unsaturated monomer component.
[0115] The ethylenically unsaturated monomer component may also include any other suitable monomers. For example, suitable other ethylenically unsaturated monomers (e.g., olefinic or vinyl monomers other than (meth)acrylates) may include isoprene, diallylphthalate, conjugated butadiene, vinyl naphthalene, acrylonitrile, (meth)acrylamides (e.g., acrylamide, methacrylamide, N-isobutoxymethyl acrylamide, N-butoxymethyl acrylamide, etc.), methacrylonitrile, vinyl acetate, vinyl propionate, vinyl butyrate, vinyl stearate, and the like, and variants and mixtures thereof
[0116] The ethylenically unsaturated monomer component may optionally include one or more vinyl aromatic compounds, including styrene. In preferred embodiments, however, the ethylenically unsaturated monomer component is not intentionally formulated to include styrene. Suitable vinyl aromatic compounds may include styrene (not preferred), substituted styrene compounds (not preferred in some embodiments), and/or other types of vinyl aromatic compounds (e.g., any of the aryl-group-containing ethylenically unsaturated monomers described herein, including aryl (meth)acrylates such as, e.g., benzyl (meth)acrylate). In some embodiments, the ethylenically unsaturated monomer component includes, if any, less than 20 wt- %, less than 10 wt- %, less than 5 wt- % or less than 1 wt- % of vinyl aromatic compounds. In some embodiments, the ethylenically unsaturated monomer component, and preferably the final polymer, is substantially free of such compounds.
[0117] In presently preferred embodiments, the ethylenically unsaturated monomer component, and preferably the final polymer, does not include any acrylamide-type monomers (e.g., acrylamides or methacrylamides).
[0118] In embodiments in which one or more surfactants are used to prepare a latex polymer, the surfactant can be an anionic, a cationic or a zwitterionic surfactant, or a mixture thereof, and also preferably includes one or more salt groups. In preferred embodiments, the surfactant includes one or more neutralized acid or anhydride groups. Examples of suitable neutralized acid groups may include carboxylate groups (--COO.sup.-), sulfate groups (--OSO.sub.3.sup.-), sulfinate groups (--SOO.sup.-), sulfonate groups (--SO.sub.2O.sup.-), phosphate groups (--OPO.sub.3.sup.-), phosphinate groups (-POO.sup.-), phosphonate groups (--PO.sub.3.sup.-), and combinations thereof.
[0119] Anionic surfactants are preferred in some embodiments.
[0120] Examples of suitable anionic surfactants include any of the following surfactants, which preferably have been at least partially neutralized with a suitable base (e.g., any of the bases disclosed herein): any of the acid- or anhydride-functional polymeric surfactants disclosed herein, dodecylbenzene sulfonic acid, dinonylnaphthalene sulfonic acid, dinonylnaphthylenedisulfonic acid, bis(2-ethylhexyl)sulfosuccinic acid, dioctyl sulfosuccinic acid, sodium lauryl sulfate, sodium dodecyl sulfate, sodium laureth sulfate, fatty acid (ester) sulfonate, polyaryl ether phosphate acid or sulfonate acid, and the like, including mixtures thereof.
[0121] In some embodiments, it may be useful to use a surfactant that is a "strong acid" surfactant prior to neutralization. Examples of "strong acid" surfactants include surfactants having a pK.sub.a of less than 4 prior to neutralization.
[0122] Although any suitable base may be used to neutralize or partially neutralize polymeric or non-polymeric surfactants to form anionic salt groups, amines are preferred bases, with tertiary amines being particularly preferred. Some examples of suitable tertiary amines are trimethyl amine, dimethylethanol amine (also known as dimethylamino ethanol), methyldiethanol amine, triethanol amine, ethyl methyl ethanol amine, dimethyl ethyl amine, dimethyl propyl amine, dimethyl 3-hydroxy-1-propyl amine, dimethylbenzyl amine, dimethyl 2-hydroxy-1-propyl amine, diethyl methyl amine, dimethyl 1-hydroxy-2-propyl amine, triethyl amine, tributyl amine, N-methyl morpholine, and mixtures thereof. Most preferably triethyl amine or dimethyl ethanol amine is used as the tertiary amine.
[0123] Some additional examples of neutralizing bases for forming anionic salt groups include inorganic and organic bases such as sodium hydroxide, potassium hydroxide, lithium hydroxide, ammonium hydroxide, and mixtures thereof.
[0124] Some examples of neutralizing compounds for neutralizing base groups present on the surfactant and forming cationic salt groups include organic and inorganic acids such as formic acid, acetic acid, hydrochloric acid, sulfuric acid, and combinations thereof.
[0125] Although the surfactant may optionally include one or more ethylenically unsaturated groups (e.g., if the surfactant is a polymerizable non-polymeric surfactant), in some embodiments, the surfactant is a saturated surfactant. By way of example, amine-neutralized dodecylbenzenesulfonic acid is considered to be a saturated surfactant. Although amine-neutralized dodecylbenzenesulfonic acid includes an aryl group that includes carbon-carbon double bonds, it does not include any ethylenically unsaturated groups.
[0126] The surfactant can be any suitable type of surfactant and may, for example, be a "lower" molecular-weight surfactant (e.g., a surfactant that is non-polymeric and/or has a number average molecular weight of less than about 1,000 Daltons, more typically less than about 750 Daltons, and even more typically less than about 500 Daltons).
[0127] In certain preferred embodiments, a polymeric surfactant is used which has, for example, a number average molecular weight greater than about 2,000 Daltons or even greater than about 4,000 Daltons. It is generally preferable to use a polymeric surfactant and/or a polymerizable surfactant to, for example, minimize or eliminate the possibility of surfactant migrating out of the cured coating and into the packaged product. Examples of suitable polymeric surfactants may include water-dispersible polymers of the acrylic, alkyd, polyester, polyether, polyolefin, or polyurethane type, including copolymers thereof (e.g., polyether-acrylic copolymers), and mixtures thereof. Typically, such water-dispersible polymers include one or more salt groups to facilitate stable dispersion into water. Examples of suitable such polymer salts are disclosed in U.S. Pat. No. 8,092,876; U.S. Application Ser. No. 62/362,729 filed on Jul. 15, 2016 and entitled "Latex Coating Composition Having Reduced Flavor Scalping Properties" (corresponds to International Application No. PCT/US2017/041858) and U.S. Pub. No. 2016/024325 (which describes the use of certain (poly)ethylene (meth)acrylic acid copolymers).
[0128] An example of a specific water-dispersible polymer for use as a "polymeric surfactant" is a "higher" acid number acid-functional polymer (e.g., acid number greater than about 40, more preferably greater than about 100 milligrams KOH per gram polymer). In a preferred embodiment, an acrylic polymer having such an acid number is solution polymerized in organic solvent and then inverted into water (e.g., via at least partial neutralization with a suitable base such as, e.g., an amine or any of the other bases disclosed herein) and used to support emulsion polymerization of the ethylenically unsaturated monomer component. In some embodiments, the acid- or anhydride-functional organic solution polymerized acrylic polymer is formed from an ethylenically unsaturated monomer component that includes an acid- or anhydride functional monomer, a branched and/or cyclic monomer, and optionally any other suitable ethylenically unsaturated monomer. In some such embodiments, the acrylic polymer is styrene-free.
[0129] A variety of acid- or anhydride-functional monomers, or salts thereof, can be used; their selection is dependent on the desired final polymer properties. Preferably, such monomers are ethylenically unsaturated, more preferably, alpha, beta-ethylenically unsaturated. Suitable ethylenically unsaturated acid- or anhydride-functional monomers include monomers having a reactive carbon-carbon double bond and an acidic or anhydride group, or salts thereof. Preferred such monomers have from 3 to 20 carbons, at least 1 site of unsaturation, and at least 1 acid or anhydride group, or salt thereof
[0130] Suitable acid-functional monomers include ethylenically unsaturated monobasic and dibasic acids, as well as anhydrides and monoesters of dibasic acids. The selected monomers preferably are readily copolymerizable with any other monomer(s) used to prepare the water-dispersible polymer. Illustrative monobasic acids include those represented by the formula CH.sub.2.dbd.C(R.sup.8)--COOH, where R.sup.8 is hydrogen or an alkyl group of 1 to 6 carbon atoms, more typically hydrogen or methyl (--CH.sub.3).
[0131] Examples of useful ethylenically unsaturated acid-functional monomers include acids such as, for example, acrylic acid, methacrylic acid, alpha-chloroacrylic acid, alpha-cyanoacrylic acid, crotonic acid, alpha-phenylacrylic acid, beta-acryloxypropionic acid, fumaric acid, maleic acid, sorbic acid, alpha-chlorosorbic acid, angelic acid, cinnamic acid, p-chlorocinnamic acid, beta-stearylacrylic acid, citraconic acid, mesaconic acid, glutaconic acid, aconitic acid, tricarboxyethylene, 2-methyl maleic acid, itaconic acid, 2-methyl itaconic acid, methyleneglutaric acid, and the like, or mixtures thereof. Preferred unsaturated acid-functional monomers include acrylic acid, methacrylic acid, crotonic acid, fumaric acid, maleic acid, 2-methyl maleic acid, itaconic acid, 2-methyl itaconic acid, and mixtures thereof. More preferred unsaturated acid-functional monomers include acrylic acid, methacrylic acid, crotonic acid, fumaric acid, maleic acid, itaconic acid, and mixtures thereof. Most preferred unsaturated acid-functional monomers include acrylic acid, methacrylic acid, maleic acid, crotonic acid, and mixtures thereof If desired, aqueous salts of the above acids may also be employed.
[0132] Examples of suitable ethylenically unsaturated anhydride monomers include compounds derived from the above acids (e.g., as a pure anhydride or mixtures of such). Preferred anhydrides include acrylic anhydride, methacrylic anhydride, and maleic anhydride.
[0133] Examples of suitable polymerizable surfactants include those disclosed in U.S. Publication No. 2002/0155235; and those commercially available under the tradename "REASOAP" from Adeka Corporation, Tokyo, Japan; under the tradenames "NOIGEN" and "HITENOL" from Da-Ichi Kogyo Siyyaku Co., Ltd., Tokyo, Japan; and under the tradename "SIPOMER" from Solvay Rhodia, Brussels, Belgium.
[0134] In some embodiments, a non-ionic surfactant is included in the reaction mixture used to make the latex polymer. Any suitable non-ionic surfactant may be employed. Examples of suitable non-ionic surfactants include ethoxylated compounds. In some embodiments, the non-ionic compound is a sucrose ester, sorbitan ester, alkyl glycoside, glycerol ester, or mixture thereof. In some embodiments, a non-ionic surfactant is used that includes hydroxyl groups. Non-ionic surfactants that comprise, or are derived from, polysorbate compounds may be used in some embodiments.
[0135] In some embodiments, a surfactant or mixture of surfactants as described in U.S. App. No. 62/387,129 entitled "Latex Polymer Made Using Metallic-Base-Neutralized Surfactant and Blush-Resistant Coating Compositions Containing Such Polymers" may be used. For example, one or more anionic or zwitterionic surfactant (e.g., non-polymeric surfactant) having an acid group neutralized with a metallic base may be used (e.g., a metallic base including aluminum, calcium, lithium, magnesium, sodium, or potassium). An example of such a surfactant is dioctyl sodium sulfosuccinate.
[0136] The latex polymer or other acrylic polymer of the present invention may exhibit any suitable glass transition (Tg) value(s). In this context, the Tg value refers to the Tg value of the latex polymer (or other acrylic polymer) alone (e.g., prior to formulating a coating composition with additional optional ingredients such as co-resins, crosslinkers, etc.). In some embodiments, it may be desirable that the latex polymer (or other acrylic polymer) has a relatively "high" Tg, such as interior can coatings that will be exposed to sensitive flavor products (e.g., certain colas in which certain flavorants are present at very low concentrations) and/or chemically aggressive food or beverage products (e.g., high acid, high salt, and/or high fat). While not intending to be bound by any theory, such a "high" Tg can be beneficial from one or more of the following perspectives: (i) decreased flavor scalping by the cured coating and/or (ii) enhanced chemical resistance exhibited by the cured coating. In such embodiments, preferred glass transition temperatures for the latex polymer (or other acrylic polymer) include those greater than 40.degree. C., more preferably greater than 50.degree. C., even more preferably greater than 60.degree. C., and in some embodiments greater than 70.degree. C. or greater than 80.degree. C. Preferred glass transition temperatures for the latex polymer (or other acrylic polymer) include those less than 120.degree. C., more preferably less than 115.degree. C., even more preferably less than 110.degree. C., and in some embodiments, less than 100.degree. C. or less than 80.degree. C. The term "latex polymer" is used broadly in the above Tg discussion and is also intended to include latex particles that include, for example, two polymers that are not covalently attached. An example of such a latex particle is one that includes a polymeric surfactant and a polymer resulting from emulsion polymerization of the ethylenically unsaturated component, wherein the two polymers are not covalently attached to one another.
[0137] In some embodiments, the Tg of the latex polymer or other acrylic polymer may be less than that described above, such as, for example, less than 40.degree. C., 0 to 40.degree. C., or 20 to 40.degree. C.
[0138] Differential scanning calorimetry (DSC) is an example of a useful method for determining the Tg of the latex polymer/particle, with a representative DSC methodology provided in the tests method section described below.
[0139] It should be noted that it may not be possible to measure a discrete Tg for certain latex polymers. For example, this may be the case for a gradient Tg latex polymer, which can contain an almost infinite number of Tg stages. For example, one may start with a high Tg monomer composition and then at a certain point in the polymerization start to feed a lower Tg stage monomer composition into the high Tg stage monomer feed. The resulting multistage latex polymer will have a gradient Tg from high to low. A "power feed" process may be used to prepare such compositions. A gradient Tg polymer may also be used in conjunction with multiple multistage Tg polymers. As an example, one may prepare a high Tg monomer feed (F1) and a low Tg monomer feed (F2). One would begin to feed F1 into the latex reactor vessel and initiate polymerization. At a certain period during the F1 feed, one would then feed F2 into F1 wherein the F2 feed rate is faster than the overall feed rate of F1 +F2 into the reactor vessel. Consequently, once the F2 feed into F1 is complete, the overall Tg of the F1+F2 monomer feed blend will be a lower Tg "soft stage" monomer composition. For such gradient Tg latex polymers, the Fox equation may be used instead of DSC to calculate Tg. If the monomers used to produce such a latex polymer include one or more monomers not having a homopolymer Tg (e.g., because the monomer does not homopolymerize), then a non-gradient reference latex can be made, in a non-power feed method, using the same overall monomer composition and used to measure Tg.
[0140] The overall latex polymer (or other acrylic polymer) may have any suitable acid number so long as the polymer is preferably capable of being stably dispersed into water. While not intending to be bound by any theory, it is believed that the presence of at least some acid groups in the latex polymer is desirable, for example, to enhance the liquid stability of the varnish and crosslinking of the resin system. In embodiments in which a carboxyl-reactive crosslinker is used (e.g., a beta-hydroxyalkylamide compound), the latex polymer (or other acrylic polymer) preferably has an acid number of at least 8 more preferably at least 15, even more preferably at least 20, and optimally at least 30 mg KOH per gram of the polymer. Preferably, the acid number is less than 200, more preferably less than 120, even more preferably less than 100, and optimally less than 80 mg KOH per gram of the polymer. Acid numbers can be measured pursuant to BS EN ISO 3682-1998 standard. The above acid numbers are inclusive of any acid- or anhydride-functional polymeric surfactant(s) incorporated into the latex polymer/particle, regardless of whether the polymeric surfactant(s) are covalently attached to the emulsion polymerized ethylenically unsaturated monomer component. The above acid numbers do not include any non-polymeric and non-polymerizable surfactant that may have been used to produce the polymer. Neutralized dodecyl benzene sulfonic acid is an example of such a non-polymeric and non-polymerizable surfactant.
[0141] When hydroxyl-functional monomer is used, the latex polymer may have any suitable hydroxyl number to achieve the desired result.
[0142] Any suitable process or materials may be employed in making the latex polymer (or other acrylic polymer). In preferred embodiments, the latex polymer is prepared using a single-stage or multi-stage emulsion polymerization process. The emulsion polymerization process may be conducted in a variety of manners. For example, the emulsion polymerization reaction of the ethylenically unsaturated monomer component can occur as a batch, intermittent, or continuous operation.
[0143] In some embodiments, the emulsion polymerization process uses an optional pre-emulsion monomer mixture in which some or all of the reactant components and one or more surfactants are dispersed in the aqueous carrier under agitation to form a stable pre-emulsion.
[0144] In other embodiments, the ethylenically unsaturated monomer component is polymerized without the use of a pre-emulsion step.
[0145] A portion of the surfactant(s) and a portion of the aqueous carrier may also be introduced into a reactor, and are preferably heated, agitated, and held under nitrogen sparge to assist in the subsequent polymerization reactions. Preferred temperatures for heating the surfactant dispersion include temperatures greater than about 65.degree. C., and more preferably from about 70.degree. C. to about 90.degree. C.
[0146] The monomer pre-emulsion or non-pre-emulsified ethylenically unsaturated monomer component may be fed to the heated aqueous dispersion in the reactor incrementally or continuously over time. Alternatively, in certain embodiments a batch or semi-batch process may be used to polymerize the reactant monomers in the aqueous dispersion, as described in, for example, U.S. Pat. No. 8,092,876. In further embodiments, the polymerization process can occur in a classic two-stage (or multiple stage) "core-shell" arrangement. Alternatively, the polymerization process can occur in a multiple stage "inverse core-shell" arrangement as discussed in International Publication No.WO2015/002958.
[0147] With regard to the conditions of the emulsion polymerization, the ethylenically unsaturated monomer component is preferably polymerized in aqueous medium with a water-soluble free radical initiator.
[0148] The temperature of polymerization is typically from 0.degree. C. to 100.degree. C., preferably from 50.degree. C. to 90.degree. C., more preferably from 70.degree. C. to 90.degree. C., and even more preferably from about 80.degree. C. to about 85.degree. C. The pH of the aqueous medium is usually maintained at a pH of 5 to 12.
[0149] In embodiments in which the acrylic polymer is a latex, the free radical initiator can be selected from one or more water-soluble peroxides which are known to act as free radical initiators. Examples include hydrogen peroxide and t-butyl hydroperoxide. Redox initiator systems well known in the art (e.g., t-butyl hydroperoxide, erythorbic acid, and ferrous complexes) can also be employed.
[0150] Further examples of polymerization initiators which can be employed include polymerization initiators which thermally decompose at the polymerization temperature to generate free radicals. Examples include both water-soluble and water-insoluble species. Further examples of free radical initiators that can be used include persulfates, such as ammonium or alkali metal (potassium, sodium or lithium) persulfate; azo compounds such as 2,2'-azo-bis(isobutyronitrile), 2,2'-azo-bis(2,4-dimethylvaleronitrile), and 1-t-butyl-azocyanocyclohexane; hydroperoxides such as t-butyl hydroperoxide, hydrogen peroxide, t-amyl hydroperoxide, methyl hydroperoxide, and cumene hydroperoxide; peroxides such as benzoyl peroxide, caprylyl peroxide, di-t-butyl peroxide, ethyl 3,3'-di(t-butylperoxy) butyrate, ethyl 3,3'-di(t-amylperoxy) butyrate, t-amylperoxy-2-ethyl hexanoate, and t-butylperoxy pivilate; peresters such as t-butyl peracetate, t-butyl perphthalate, and t-butyl perbenzoate; as well as percarbonates, such as di(1-cyano-1-methylethyl)peroxy dicarbonate; perphosphates, and the like; and combinations thereof.
[0151] Polymerization initiators can be used alone or as the oxidizing component of a redox system, which also preferably includes a reducing component such as ascorbic acid, malic acid, glycolic acid, oxalic acid, lactic acid, thiogycolic acid, or an alkali metal sulfite, more specifically a hydrosulfite, hyposulfite or metabisulfite, such as sodium hydrosulfite, potassium hyposulfite and potassium metabisulfite, or sodium formaldehyde sulfoxylate, benzoin and combinations thereof. The reducing component is frequently referred to as an accelerator or a catalyst activator.
[0152] The initiator and accelerator (if any) are preferably used in proportion from about 0.001% to 5% each, based on the weight of monomers to be copolymerized. Promoters such as chloride and sulfate salts of cobalt, iron, nickel or copper can be used in small amounts, if desired. Examples of redox catalyst systems include tert-butyl hydroperoxide/sodium formaldehyde sulfoxylate/Fe(II), and ammonium persulfate/sodium bisulfate/sodium hydrosulfite/Fe(II).
[0153] Chain transfer agents can be used to control polymer molecular weight, if desired.
[0154] After the polymerization is completed, at least a portion of the carboxylic acid groups and/or anhydride groups of the latex polymer (or other salt-forming groups such as, e.g., other neutralizable acid groups and/or neutralizable base groups) may be neutralized or partially neutralized with a suitable basic compound (or other suitable neutralizing compound) to produce water-dispersing groups. The basic compound used for neutralization can be a metallic base, a fugitive base (e.g., ammonia and primary, secondary, and/or tertiary amines), or a mixture thereof In preferred embodiments, the base is a fugitive base, more preferably an amine. The degree of neutralization may vary considerably depending upon the amount of acid or base groups included in the latex polymer, and the degree of dispersibility that is desired.
[0155] Coating compositions of the present invention preferably include at least a film-forming amount of the latex polymer or other acrylic polymer described herein. In preferred embodiments, the coating composition includes at least about 50 wt- %, more preferably at least about 65 wt- %, and even more preferably at least about 80 wt- % or at least about 90 wt- % of the latex polymer (or other acrylic polymer described herein), based on the total resin solids weight of the coating composition. The coating composition includes 100 wt- % or less, more typically less than about 99 wt- %, and even more typically less than about 95 wt- % of the latex polymer (or other acrylic polymer described herein), based on the total resin solids weight of the coating composition. The above weight percentages of latex polymer are inclusive of any surfactant(s) (e.g., polymeric and/or non-polymeric surfactant) used to make the latex polymer, regardless of whether the surfactant(s) are covalently attached to the emulsion polymerized ethylenically unsaturated monomer component.
[0156] The coating composition may be formulated from the latex polymer and/or other acrylic polymer described herein, optionally with the inclusion of one or more additives. In embodiments in which the coating composition includes one or more additives, the additives preferably do not adversely affect the latex emulsion or other polymer described herein, or a cured coating formed from the coating composition. For example, such optional additives may be included in the coating composition to enhance composition aesthetics, to facilitate manufacturing, processing, handling, and application of the composition, and to further improve a particular functional property of the coating composition or a cured coating resulting therefrom.
[0157] Such optional additives include, for example, catalysts, dyes, pigments, toners, extenders, fillers, lubricants, anticorrosion agents, flow-control agents, thixotropic agents, dispersing agents, antioxidants, adhesion promoters, light stabilizers, curing agents, co-resins, organosilicon materials, and mixtures thereof. Each optional additives is preferably included in a sufficient amount to serve its intended purpose, but not in such an amount to adversely affect the coating composition or a cured coating resulting therefrom.
[0158] One preferred optional additive is a catalyst to increase the rate of cure. Examples of catalysts, include, but are not limited to, strong acids (e.g., dodecylbenzene sulfonic acid (DDBSA, available as CYCAT 600 from Cytec), methane sulfonic acid (MSA), p-toluene sulfonic acid (pTSA), dinonylnaphthalene disulfonic acid (DNNDSA), and triflic acid), quaternary ammonium compounds, phosphorous compounds, and tin, titanium, and zinc compounds. Specific examples include, but are not limited to, a tetraalkyl ammonium halide, a tetraalkyl or tetraaryl phosphonium iodide or acetate, tin octoate, zinc octoate, triphenylphosphine, and similar catalysts known to persons skilled in the art.
[0159] If used, the catalyst is preferably present in an amount of at least about 0.01% by weight, and more preferably at least about 0.1% by weight, based on the total solids weight of the coating composition. Furthermore, if used, the catalyst is also preferably present in a non-volatile amount of no greater than about 3% by weight, and more preferably no greater than about 1% by weight, based on the total solids weight of the coating composition.
[0160] Another useful optional ingredient is a lubricant (e.g., a wax), which facilitates manufacture of metal closures and other fabricated coated articles by imparting lubricity to coated metal substrate. Preferred lubricants include, for example, Carnauba wax and polyethylene-type lubricants. If used, a lubricant is preferably present in the coating composition in an amount of at least about 0.1% by weight, and preferably no greater than about 2% by weight, and more preferably no greater than about 1% by weight, based on the total solids weight of the coating composition.
[0161] Another useful optional ingredient is an organosilicon material, such as a siloxane-based and/or polysilicone-based materials. Representative examples of suitable such materials are disclosed in International Publication Nos. WO/2014/089410 and WO/2014/186285.
[0162] Another useful optional ingredient is a pigment, such as titanium dioxide. If used, a pigment is present in the coating composition in an amount of no greater than about 70% by weight, more preferably no greater than about 50% by weight, and even more preferably no greater than about 40% by weight, based on the total solids weight of the coating composition.
[0163] As previously discussed, preferred embodiments of the coating composition include a carboxyl-reactive crosslinker, more preferably a NCCR crosslinker, and even more preferably a beta-hydroxyalkylamide crosslinker. In addition, or alternatively, the coating may include one or more additional curing agents such as, for example, any of the crosslinkers described below. Preferred crosslinkers are substantially free of each of BPA, BPF, BPS, including glycidyl ether compounds thereof (e.g., BADGE), and epoxy novolacs.
[0164] Any of the well-known, hydroxyl-reactive curing resins can be used. For example, phenoplast, blocked isocyanates, and aminoplast curing agents may be used, as well as combinations thereof.
[0165] Phenoplast resins include the condensation products of aldehydes with phenols. Formaldehyde and acetaldehyde are preferred aldehydes. Various phenols can be employed such as phenol, cresol, p-phenylphenol, p-tert-butylphenol, p-tert-amylphenol, and cyclopentylphenol. Resole phenolics, particularly resole phenolics not made using BPA, BPF, or BPS, are preferred phenoplasts.
[0166] Aminoplast resins are the condensation products of aldehydes such as formaldehyde, acetaldehyde, crotonaldehyde, and benzaldehyde with amino or amido group-containing substances such as urea, melamine, and benzoguanamine. Examples of suitable aminoplast crosslinking resins include benzoguanamine-formaldehyde resins, melamine-formaldehyde resins, esterified melamine-formaldehyde, and urea-formaldehyde resins.
[0167] As examples of other generally suitable crosslinkers are the blocked or non-blocked aliphatic, cycloaliphatic or aromatic di-, tri-, or poly-valent isocyanates, such as hexamethylene diisocyanate (HMDI), cyclohexyl-1,4-diisocyanate, and the like. Further examples of generally suitable blocked isocyanates include isomers of isophorone diisocyanate, dicyclohexylmethane diisocyanate, toluene diisocyanate, diphenylmethane diisocyanate, phenylene diisocyanate, tetramethyl xylene diisocyanate, xylylene diisocyanate, and mixtures thereof.
[0168] The total amount of one or more crosslinkers included in the coating composition may depend on the type of crosslinker, the time and temperature of the bake, and molecular weight. If used, the one or more crosslinkers are typically present in an amount of up to about 50 wt- %, preferably up to about 30 wt- %, more preferably up to about 15wt- %. If used, the crosslinker is typically present in an amount of at least about 0.1 wt- %, more preferably at least about 1 wt- %, and even more preferably at least about 2 wt- %. These weight percentages are based on the total resin solids weight of the coating composition.
[0169] In preferred embodiments, the coating composition includes at least 1 wt- %, more preferably at least 2 wt- %, and even more preferably at least 3 wt- % of one or more NCCR crosslinkers, based on total resin solids of the coating composition. Although the upper amount is not restricted, the coating composition preferably includes less than 20 wt- %, more preferably less than 15 wt- %, and even more preferably less than 10 wt- % of one or more NCCR crosslinkers, based on total resin solids of the coating composition. In certain preferred embodiments, the coating composition includes from 4 to 8.5 wt- % of one or more NCCR crosslinkers (e.g., Primid QM1260 crosslinker), more preferably from 5 to 7.5 wt- % of one or more NCCR crosslinker, based on the total resin solids of the coating composition. In some embodiments, the coating composition includes at least the above amount of one or more beta-hydroxyalkylamide crosslinkers.
[0170] In embodiments in which excellent "hard-to-hold" food-contact coating performance is desired (e.g., the ability to withstand the corrosive properties of packaging food or products having particularly challenging chemical profiles such as, e.g., certain alcoholic cider beverages), it has been discovered that it can be beneficial to use both an NCCR crosslinker and a phenoplast crosslinker. The use of certain such crosslinker combinations has been observed to result in interior can coatings that have increased chemical resistance when challenged with certain "hard-to-hold" products (e.g., alcoholic ciders), as indicated, for example, by reduced instances of coating blush and reduced aluminum pickup. A preferred combination of such crosslinkers is the combination of one or more beta-hydroxyalkylamides (e.g., the PRIMID QM 1260 crosslinker) with one or more resole phenolic crosslinkers (more preferably one or more resole phenolics that are not intentionally made with BPA, BPF, or BPS as a starting ingredient). Thus, in some embodiments, the coating composition includes, based on total resin solids: (i) 2 to 10 wt- %, 4 to 8.5 wt- %, or 5 to 7.5 wt- % of NCCR crosslinker (more preferably a beta-hydroxyalkylamide crosslinker); and (ii) 1 to 10 wt- %, 3 to 8 wt- %, or 4 to 6 wt- % of a phenoplast crosslinker (more preferably a resole phenolic crosslinker).
[0171] In some embodiments, the coating composition may be cured without the use of an external crosslinker. Additionally, the coating composition may be substantially free of formaldehyde and formaldehyde-containing materials, essentially free of these compounds, essentially completely free of these compounds, or even completely free of these compounds. In preferred embodiments, the coating composition is also substantially free, essentially free, essentially completely free, or completely free of each of BPA, BPF, and BPS, including any diepoxides thereof (e.g., diglycidyl ethers thereof such as the diglycidyl ether of BPA ("BADGE")). In some embodiments, the coating composition is substantially free or completely free of bisphenol compounds.
[0172] In preferred embodiments, the coating composition is also substantially free, essentially free, essentially completely free, or completely free of one or both of styrene and substituted styrene compounds.
[0173] In preferred embodiments, the coating composition is substantially free or completely free of halogenated monomers (whether free or polymerized), such as chlorinated vinyl monomers.
[0174] In some embodiments, such as for certain spray coating applications (e.g., aqueous inside spray for food or beverage cans including, e.g., aluminum beverage cans), the coating composition may have a total solids weight greater than about 5%, more preferably greater than about 10%, and even more preferably greater than about 15%. In these embodiments, the coating composition may also have a total solids weight less than about 40%, more preferably less than about 30%, and even more preferably less than about 25%. In some of these embodiments, the coating composition may have a total solids weight ranging from about 18% to about 22%. The liquid carrier (e.g., aqueous carrier) may constitute the remainder of the weight of the coating composition.
[0175] If desired, the coating composition may also include one or more other optional polymers, such as, for example, one or more acrylic polymers, alkyd polymers, epoxy polymers, polyolefin polymers, polyurethane polymers, polysilicone polymers, polyester polymers, and copolymers and mixtures thereof.
[0176] In aqueous embodiments, the aqueous carrier of the coating composition preferably includes water and may further include one or more optional organic solvents (e.g., one or more water-miscible solvents). In some embodiments, water constitutes greater than about 20% by weight, more preferably greater than about 35% by weight, and even more preferably greater than about 50% by weight of the total weight of the aqueous carrier. In some embodiments, water constitutes 100% or less, less than about 95% by weight, or less than about 90% by weight of the total weight of the aqueous carrier.
[0177] While not intending to be bound by theory, the inclusion of a suitable amount of an organic solvent in the aqueous carrier can be advantageous in some embodiments. Accordingly, in certain embodiments, the one or more organic solvents may constitute greater than 0%, more preferably greater than about 5%, and even more preferably greater than about 10% by weight of the aqueous carrier. In these embodiments, the organic solvents may also constitute less than about 80%, more preferably less than about 65%, and even more preferably less than about 50% or less than about 40% by weight of the aqueous carrier.
[0178] In some embodiments, the coating composition is a solvent-based coating composition that preferably includes no more than a de minimus amount (e.g., 0 to 2 wt- %) of water. For example, in some embodiments, the coating composition is a styrene-free, organic-solvent-based food or beverage can coating composition that includes a styrene-free organic solution polymerized acid- or anhydride-functional acrylic polymer (e.g., acid number>20 mg KOH/g resin) preferably in combination with one or more NCCR described herein. Such organic solution polymerized acid- or anhydride acrylic polymers can have any suitable number molecular weight (Mn) such as, for example, greater than 3,000, greater than 4,000, greater than 5,000, or even as high as or greater than 30,000 Mn if viscosity can be suitably controlled.
[0179] The coating composition preferably has a viscosity suitable for a given coating application. In some embodiments (e.g., aqueous inside spray for food or beverage cans), the coating composition may have an average viscosity greater than about 5 seconds, more preferably greater than 10 seconds, and even more preferably greater than about 15 seconds, based on the Viscosity Test described below. In some embodiments e.g., aqueous inside spray for food or beverage cans), the coating composition may also have an average viscosity less than about 40 seconds, more preferably less than 30 seconds, and even more preferably less than about 25, based on the Viscosity Test described below.
[0180] The coating composition of the present invention may be applied to a variety of different substrates using a variety of different coating techniques (e.g., spray coating, roll coating, wash coating, dipping, etc.). In certain preferred embodiments, the coating composition is applied as an inside spray coating. As briefly described above, cured coatings formed from the coating composition are particularly suitable for use on metal food and beverage cans (e.g., two-piece cans, three-piece cans, and the like). Two-piece cans (e.g., two-piece beer or soda cans and certain food cans) are typically manufactured by a drawn and ironing ("D&I") process. The cured coatings are also suitable for use in food or beverage contact situations (collectively referred to herein as "food-contact"), and may be used on the inside or outside of such cans.
[0181] The disclosed coating compositions may be present as a layer of a mono-layer coating system or as one or more layers of a multi-layer coating system. The coating composition can be used as a primer coat, an intermediate coat, a top coat, or a combination thereof. The coating thickness of a particular layer, and of the overall coating system, will vary depending upon the coating material used, the substrate, the coating application method, and the end use for the coated article. Mono-layer or multi-layer coating systems including one or more layers formed from the disclosed coating composition may have any suitable overall coating thickness, but will typically have an overall average dry coating thickness of from about 1 to about 60 micrometers and more typically from about 2 to about 15 micrometers. Typically, the overall average dry coating thickness for rigid metal food or beverage can applications will be about 3 to about 10 micrometers. Coating systems for use on closures (e.g., twist-off metal closures) for food or beverage containers may have an overall average dry coating thickness up to about 15 micrometers. In certain embodiments in which the coating composition is used as an interior coating on a drum (e.g., a drum for use with food or beverage products), the overall average dry coating thickness may be approximately 25 micrometers.
[0182] The metal substrate used in forming rigid food or beverage cans, or portions thereof, typically has an average thickness in the range of about 125 micrometers to about 635 micrometers. Electro-tinplated steel, cold-rolled steel and aluminum are commonly used as metal substrates for food or beverage cans, or portions thereof. In embodiments in which a metal foil substrate is employed in forming, e.g., a packaging article, the thickness of the metal foil substrate may be even thinner that that described above.
[0183] The disclosed coating compositions may be applied to a substrate either prior to, or after, the substrate is formed into an article such as, for example, a food or beverage container or a portion thereof In one embodiment, a method of forming food or beverage cans is provided that includes: applying a coating composition described herein to a metal substrate (e.g., applying the composition to the metal substrate in the form of a planar coil or sheet), hardening the composition, and forming (e.g., via stamping or other deformation process) the substrate into a packaging container or a portion thereof (e.g., a food or beverage can or a portion thereof). For example, two-piece or three-piece cans or portions thereof such as riveted beverage can ends (e.g., soda or beer cans) having a cured coating of the disclosed coating composition on a surface thereof can be formed in such a method. In another embodiment, a method of forming food or beverage cans is provided that includes: providing a packaging container or a portion thereof (e.g., a food or beverage can or a portion thereof), applying a coating composition described herein to the inside, outside or both inside and outside portions of such packaging container or a portion thereof (e.g., via spray application, dipping, etc.), and hardening the composition.
[0184] After applying the coating composition onto a substrate, the composition can be cured using a variety of processes, including, for example, oven baking by either conventional or convectional methods, or any other method that provides an elevated temperature suitable for curing the coating. The curing process may be performed in either discrete or combined steps. For example, substrates can be dried at ambient temperature to leave the coating compositions in a largely uncrosslinked state. The coated substrates can then be heated to fully cure the compositions. In certain instances, the disclosed coating compositions may be dried and cured in one step.
[0185] The cure conditions will vary depending upon the method of application and the intended end use. The curing process may be performed at any suitable temperature, including, for example, oven temperatures in the range of from about 100.degree. C. to about 300.degree. C., and more typically from about 177.degree. C. to about 250.degree. C. If a metal coil is the substrate to be coated, curing of the applied coating composition may be conducted, for example, by heating the coated metal substrate over a suitable time period to a peak metal temperature ("PMT") of preferably greater than about 177.degree. C. More preferably, the coated metal coil is heated for a suitable time period (e.g., about 5 to 900 seconds) to a PMT of at least about 218.degree. C.
[0186] In some embodiments, the coating composition is an inside spray coating composition capable of being spray applied on an interior of a food or beverage can (e.g., a 2-piece steel or aluminum food or beverage can) to effectively, and evenly, coat the substrate and form a continuous cured coating (e.g., a coating that exhibits a suitable initial metal exposure value, thereby indicating that the substrate has been effectively coated and is free of unsuitable pores or gaps in the coating).
[0187] For interior food-contact coatings, and interior coatings for soda beverage cans in particular, preferred Tg for the cured coating include those greater than about 50.degree. C., more preferably greater than about 60.degree. C., even more preferably greater than about 70.degree. C., and in some embodiments, greater than about 80.degree. C. Preferred Tg for the cured coating include those less than about 120.degree. C., more preferably less than about 115.degree. C., even more preferably less than about 110.degree. C., and in some embodiments, less than about 100.degree. C. An example of a suitable DSC methodology is provided below.
[0188] In some embodiments, the cured coating preferably exhibits desired properties for use as an interior food-contact coating (e.g., inside spray coating) for food and beverage cans. For example, the cured coating preferably gives a global extraction of less than about 25 parts-per-million (ppm), and more preferably less than about 10 ppm, and even more preferably less than about 1 ppm, pursuant to the Global Extraction test below. Additionally, the cured coating preferably exhibits a metal exposure less than about 5 milliamps (mA), more preferably less than about 2 mA, and even more preferably less than about 1 mA, pursuant to the Initial Metal Exposure test below. In addition, the cured coating is preferably free of or substantially free of blush (e.g., exhibits a blush rating of at least 8, more preferably at least 9, and optimally 10) pursuant to the Blush Resistance test described below. For inside spray beverage can coating compositions, preferred cured coatings give metal exposure values after drop damage of less than 30 mA, more preferred values of less than 20 mA, even more preferred values of less than 10 mA, even more preferred values of less than 3.5 mA, even more preferred values of less than 2.5 mA, and even more preferred values of less than 1.5 mA pursuant to the Metal Exposure After Drop Damage test below
[0189] The coating composition of the present disclosure may also offers utility in other coating applications. These additional applications include, but are not limited to, wash coating, sheet coating, and side seam coatings (e.g., food can side seam coatings). Other commercial coating application and curing methods are also envisioned, for example, electrocoating, extrusion coating, laminating, powder coating, and the like. The coating composition may also be useful in medical or cosmetic packaging applications, including, for example, on surfaces of metered-dose inhalers ("MDIs"), including on drug-contact surfaces.
[0190] Because the balance of coating requirements for food or beverage can coatings are particularly stringent and difficult to achieve, it is believed the coating compositions of the present invention may also be suitable for a multitude of other coatings areas, which generally have less stringent coating requirements. For example, the coating compositions of the present invention may be particularly suitable for non-packaging-related coil coating operations (e.g., "industrial" coil coatings). The coating composition may be applied to planar metal stock such as is used, for example, for lighting fixtures; architectural metal skins (e.g., gutter stock, window blinds, siding and window frames); interior or exterior steel building products; HVAC applications; agricultural metal products; industrial coating applications (e.g., appliance coatings); packaging coating applications (e.g., food or beverage cans, drug cans, etc.) and the like. The coating composition may be particularly suited for a coil coating operation where the composition is applied on rapidly moving planar metal coil substrate and then baked at elevated temperatures (e.g., >100.degree. C.) as the coated substrate travels toward an uptake coil winder.
Exemplary Embodiments
[0191] Embodiment 1 is a coating composition comprising: [0192] an acid- or anhydride-functional acrylic polymer that is substantially free of styrene; [0193] a nitrogen-containing carboxyl-reactive crosslinker; and [0194] liquid carrier that includes one or both of water and an organic solvent; [0195] wherein the coating composition is a food or beverage can coating composition suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A, including epoxides thereof.
[0196] Embodiment 1' is the coating composition of Embodiment 1 wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof.
[0197] Embodiment 2 is the coating composition of any preceding embodiment, wherein the coating composition is an aqueous coating composition.
[0198] Embodiment 3 is a coating composition comprising: [0199] an acid- or anhydride-functional acrylic polymer comprising an acid- or anhydride-functional latex that is substantially free of styrene and has a glass transition temperature of greater than 40.degree. C., greater than 50.degree. C., or greater than 60.degree. C.; and [0200] a nitrogen-containing carboxyl-reactive crosslinker; [0201] wherein the coating composition is an aqueous coating composition that is suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A.
[0202] Embodiment 3' is the coating composition of Embodiment 3 wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof.
[0203] Embodiment 4 is a coating composition comprising: [0204] an acid- or anhydride-functional acrylic polymer comprising an acid- or anhydride-functional latex that is optionally substantially free of styrene (i.e., some embodiments may include styrene), the acid- or anhydride-functional latex formed by emulsion polymerizing ethylenically unsaturated monomers comprising more than 5 wt- %, more than 6 wt- %, more than 7 wt- %, more than 8 wt- %, more than 9 wt- %, more than 10 wt- %, more than 11 wt- %, more than 12 wt- %, more than 13 wt- %, or more than 14 wt- % of multi-ethylenically unsaturated monomer; [0205] a carboxyl-reactive crosslinker (e.g., a nitrogen-containing carboxyl-reactive crosslinker); [0206] wherein the coating composition is an aqueous coating composition that is suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A.
[0207] Embodiment 4' is the coating composition of Embodiment 4 wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof.
[0208] Embodiment 5 is the coating composition of any of embodiments 1, 1', 2, 2', 4 or 4', wherein the acid- or anhydride-functional acrylic polymer has a glass transition temperature of greater than 40.degree. C., greater than 50.degree. C., or greater than 60.degree. C.
[0209] Embodiment 6 is the coating composition of any preceding embodiment, wherein the carboxyl-reactive crosslinker (e.g., nitrogen-containing carboxyl-reactive crosslinker) is not formed from reactants including formaldehyde.
[0210] Embodiment 7 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker (e.g., nitrogen-containing carboxyl-reactive crosslinker) includes hydroxyl groups.
[0211] Embodiment 8 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker includes two or more nitrogen atoms.
[0212] Embodiment 9 is the coating composition of any preceding embodiment, wherein the carboxyl-reactive crosslinker includes at least one amide group, and optionally two or more amide groups.
[0213] Embodiment 10 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker includes a hydroxyl group that is located beta relative to the nitrogen atom of an amide bond.
[0214] Embodiment 11 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker includes two or more groups of the following structure:
HO--R.sup.1--N(R.sup.2)--C(.dbd.O)-- [0215] wherein each R.sub.1 is independently an organic group, and each R.sub.2 is independently hydrogen or an organic group.
[0216] Embodiment 12 is the coating composition of embodiment 11, wherein each R independently includes from 2 to 10 carbon atoms.
[0217] Embodiment 13 is the coating composition of embodiment 12, wherein the depicted hydroxyl group is attached directly to a first carbon atom that is attached directly to a second carbon, and wherein the depicted nitrogen atom is attached directly to the second carbon atom.
[0218] Embodiment 14 is the coating composition of any of embodiment 11 to 13, wherein each R.sup.2 is independently an organic group that includes a hydroxyl group.
[0219] Embodiment 15 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises one or more groups capable of forming an intermediate having an oxazolinium structure.
[0220] Embodiment 16 is the coating composition of any of embodiment 11 to 15, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises a compound having the following structure:
(HO--R.sup.1--N(R.sup.2)--C(.dbd.O)).sub.n--X [0221] wherein: [0222] n is 2 or more, and [0223] X is a polyvalent organic group.
[0224] Embodiment 17 is the coating composition of embodiment 16, wherein Xis an alkylene group.
[0225] Embodiment 18 is the coating composition of embodiment 17, wherein the alkylene group is --(CH.sub.2).sub.4--.
[0226] Embodiment 19 is the coating composition of any preceding embodiment, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises:
##STR00003##
[0227] Embodiment 20 is the coating composition of any of embodiments 1 to 18, wherein the nitrogen-containing carboxyl-reactive crosslinker is selected from bis[N,N-di(.beta.-hydroxy-ethyl)]adipamide, bis[N,N-di(.beta.-hydroxy-propyl)]succinamide, bis[N,N-di(.beta.-hydroxy-ethyl)]azelamide, bis[N,N-di(.beta.-hydroxy-propyl)]adipamide, bis[N-metil-N-(.beta.-hydroxy-ethyl)]oxamide, or a mixture thereof.
[0228] Embodiment 21 is the coating composition of any of embodiments 1 to 18, wherein the nitrogen-containing carboxyl-reactive crosslinker comprises a beta-hydroxyalkylamide crosslinker.
[0229] Embodiment 22 is the coating composition of any of embodiments 1 to 8, wherein the carboxyl-reactive crosslinker includes one or more aziridine, diimide, or oxazoline groups.
[0230] Embodiment 23 is the coating composition of any preceding embodiment, wherein the coating composition includes at least I wt- %, at least 2 wt- %, at least 3 wt- %, at least 4 wt- %, or at least 5 wt- %, based on total resin solids, of the carboxyl-reactive crosslinker (e.g., nitrogen-containing carboxyl-reactive crosslinker).
[0231] Embodiment 24 is the coating composition of any preceding embodiment, wherein the coating composition includes at least 50 weight percent, based on total esin solids, of the acid- or anhydride-functional acrylic polymer.
[0232] Embodiment 25 is the coating composition of any preceding embodiment, wherein the coating composition includes one or more water-miscible organic solvents.
[0233] Embodiment 26 is the coating composition of any preceding embodiment, wherein the coating composition, when thermally cured, has a glass transition temperature of at least 40.degree. C., at least 50.degree. C., or at least 60.degree. C.
[0234] Embodiment 27 is the coating composition of any preceding embodiment, wherein the acid- or anhydride-functional acrylic polymer has a glass transition temperature of from 50 to 80.degree. C.
[0235] Embodiment 28 is the coating composition of any preceding embodiment, wherein the acid- or anhydride-functional acrylic polymer has a number average molecular weight of at least 3,000.
[0236] Embodiment 29 is the coating composition of any preceding embodiment, wherein the acid- or anhydride-functional acrylic polymer has a number average molecular weight of more than 30,000.
[0237] Embodiment 30 is the coating composition of any preceding embodiment, wherein the acid- or anhydride-functional acrylic polymer has an acid number of at least 20 mg KOH/g resin.
[0238] Embodiment 31 is the coating composition of any of embodiments 3 to 30, wherein the acid- or anhydride-functional latex is a reaction product of an ethylenically unsaturated monomer component emulsion polymerized in the presence of an aqueous dispersion of a water-dispersible polymer.
[0239] Embodiment 32 is the coating composition of embodiment 31, wherein the water-dispersible polymer comprises an acrylic polymer, a polyether polymer, a polyolefin polymer, a polyester polymer, or a mixture or copolymer thereof.
[0240] Embodiment 33 is the coating composition of embodiment 32, wherein the water-dispersible polymer comprises an at least partially neutralized acid- or anhydride-functional acrylic polymer.
[0241] Embodiment 34 is the coating composition of embodiment 32 or 33, wherein the acrylic polymer is an organic solution polymerized acrylic polymer.
[0242] Embodiment 35 is the coating composition of any preceding embodiment, wherein at least a portion of the acid- or anhydride-functional acrylic polymer is formed an ethylenically unsaturated monomer component including at least one monomer having (i) a Tg of more than 40.degree. C. and (ii) one or more groups selected from cyclic groups, branched organic groups, or a combination thereof.
[0243] Embodiment 36 is the coating composition of embodiment 35, wherein at least one cyclic group is present.
[0244] Embodiment 37 is the coating composition of embodiment 36, wherein the at least one cyclic group is selected from one or more of substituted or unsubstituted: cyclobutane groups, cyclopentane groups, cyclohexane groups, phenylene groups, norbornene groups, norbornane groups, tricyclodecane groups, or a combination thereof.
[0245] Embodiment 38 is the coating composition of embodiment 35, wherein at least one branched organic group is present.
[0246] Embodiment 39 is the coating composition of embodiment 38, wherein the at least one branched organic group is present in a monomer selected from isopropyl methacrylate, isobutyl methacrylate, sec-butyl methacrylate, or a mixture thereof.
[0247] Embodiment 40 is the coating composition of embodiment 35, wherein the at least one monomer having (i) and (ii) has the following structure:
(R.sup.3).sub.2--C.dbd.C(R.sup.4)--W.sub.n--Y, [0248] wherein: [0249] R.sup.3 is independently selected from hydrogen or an organic group; [0250] R.sup.4 is selected from hydrogen or an alkyl group; [0251] W, if present, is a divalent linking group; [0252] n is 0 or 1; and [0253] Y is: (a) a branched organic group including one or more branching atoms, (b) a cyclic group, or (c) a combination of (a) and (b).
[0254] Embodiment 41 is the coating composition of embodiment 40, wherein Y is a branched organic group of the following structure:
--C(CH.sub.3).sub.t(R.sup.5).sub.3-t [0255] wherein: [0256] t is 0 to 3; [0257] each R.sup.5, if present, is independently an organic group that may optionally be itself branched and may optionally include one or more heteroatoms; and [0258] two or more R.sup.5 may optionally form a cyclic group with one another.
[0259] Embodiment 42 is the coating composition of embodiment 41, wherein t is 1, each R.sup.5 comprises an alkyl group, and the total number of carbon atoms in both R.sup.5 groups is 6, 7, or 8.
[0260] Embodiment 43 is the coating composition of embodiment 41, wherein t is 0, 1, or 2 and at least one R.sup.5 is a branched organic group.
[0261] Embodiment 44 is the coating composition of any one of embodiments 41 to 43, wherein at least one R.sup.5 includes a tertiary or quaternary carbon atom.
[0262] Embodiment 45 is the coating composition of any preceding embodiment, wherein the acid- or anhydride-functional polymer (e.g., acid- or anhydride-functional latex) is formed by polymerizing (e.g., emulsion polymerizing in an aqueous media) an ethylenically unsaturated monomer component comprising at least 10 wt- %, at least 20 wt- %, at least 30 wt- %, and in some embodiments 40 wt- % or more of one or more branched and/or cyclic monomers.
[0263] Embodiment 46 is the coating composition of any preceding embodiment, wherein one or both of: (i) the acid- or anhydride-functional acrylic polymer and (ii) the coating composition are substantially free of each of bisphenols and/or halogenated monomers.
[0264] Embodiment 47 is the coating composition of any of embodiments 3 to 46, wherein the acid- or anhydride functional latex is formed from ingredients including an emulsion polymerized ethylenically unsaturated monomer component that includes a multi-ethylenically unsaturated monomer, and wherein the emulsion polymerized ethylenically unsaturated monomer component is optionally substantially free of oxirane-group-containing monomers.
[0265] Embodiment 48 is the coating composition of Embodiment 47, wherein multi-ethylenically unsaturated monomer comprise more than 5 wt- %, more than 6 wt- %, more than 7 wt- %, more than 8 wt- %, more than 9 wt- %, more than 10 wt- %, more than 11 wt- %, more than 12 wt- %, more than 13 wt- %, or more than 14 wt- % of the total weight of emulsion polymerized ethylenically unsaturated monomers used to form the acid- or anhydride-functional latex.
[0266] Embodiment 49 is the coating composition of any of embodiments 4 to 48, wherein the multi-ethylenically unsaturated monomer comprises 1,4-butandiol di(meth)acrylate.
[0267] Embodiment 50 is the coating composition of any of embodiments 3 to 49, wherein methyl (meth)acrylate comprises at least 20 wt- %, at least 25 wt- %, at least 30 wt- %, or at least 40 wt- % of the ethylenically unsaturated monomers used to form the acid- or anhydride-functional latex.
[0268] Embodiment 51 is the coating composition of any of embodiments 3 to 50, wherein methyl methacrylate comprises at least 20 wt- %, at least 25 wt- %, at least 30 wt- %, or at least 40 wt- % of the emulsion polymerized ethylenically unsaturated monomers used to form the acid- or anhydride-functional latex.
[0269] Embodiment 52 is the coating composition of any of embodiments 3 to 51, wherein the ethylenically unsaturated monomer component used to form the acid or anhydride-functional latex includes methyl methacrylate and ethyl acrylate.
[0270] Embodiment 53 is the coating composition of any of embodiment 52, wherein the ethylenically unsaturated monomer component include a hydroxyl-functional (meth)acrylate (e.g., hydroxyethyl methacrylate).
[0271] Embodiment 54 is the coating composition of any of embodiments 47 to 53, wherein an acid- or anhydride-functional acrylic latex has a glass transition temperature of greater than 40.degree. C., greater than 50.degree. C., or greater than 60.degree. C.
[0272] Embodiment 55 is the coating composition of any preceding embodiment, wherein the coating composition includes, based on total resin solids, from 1 to 20 wt- % (e.g., 2 to 10 wt- %, 4 to 8.5 wt- %, or 5 to 7.5 wt- %) of the nitrogen-containing carboxyl reactive crosslinker and from 50 to 99 wt- % of the acid- or anhydride-functional acrylic polymer.
[0273] Embodiment 56 is the coating composition of any preceding embodiment, wherein the coating composition is an inside spray beverage can coating composition.
[0274] Embodiment 57 is a method of coating a food or beverage can, comprising: [0275] applying the coating composition of any preceding embodiment on a metal substrate prior to, or after, forming the metal substrate into a food or beverage container or a portion thereof.
[0276] Embodiment 58 is the method of embodiment 57, wherein the coating composition is spray applied on the metal substrate.
[0277] Embodiment 59 is the method of embodiment 57, wherein the coating composition is spray applied on an interior surface of an aluminum beverage can.
[0278] Embodiment 60 is the method of any one of embodiments 57 to 59, and further comprising curing the coating composition on the metal substrate to form a continuous cured coating having an average film thickness of from 2 to 15 microns.
[0279] Embodiment 61 is a food or beverage can, or a portion thereof, resulting from the method of any of embodiments 57 to 60.
[0280] Embodiment 62 is food or beverage can, or a portion thereof, having a metal substrate with a cured coating formed from the coating composition of any of embodiments 1 to 56 applied on an interior surface, an exterior surface, or both.
[0281] Embodiment 63 is the food or beverage can, or portion thereof, of embodiments 61 or 62, wherein the cured coating has a Tg of at least 50.degree. C., at least 60.degree. C., or at least 70.degree. C.
[0282] Embodiment 64 is a food or beverage can, or a portion thereof, having an interior food-contact coating having an overall average dry coating thickness of from 2 to 15 micrometers, wherein: [0283] the interior food-contact coating is formed from a spray applied aqueous coating composition that is substantially free of each of styrene and halogenated monomers and is also substantially free of bisphenol A, and wherein the coating composition includes, based on total resin solids, at least 50 wt- % of an emulsion polymerized latex; and [0284] the interior food-contact coating has a metal exposure value after drop damage of less than 10 mA when tested pursuant to the Metal Exposure after Drop Damage test disclosed herein.
[0285] Embodiment 64' is the s a food or beverage can, or a portion thereof, of embodiment 64, wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof.
[0286] Embodiment 65 is the food or beverage can, or a portion thereof, of embodiment 64 or 64', wherein the can comprises an aluminum beverage can and the interior food-contact coating has a metal exposure value after drop damage of less than 3.5 mA.
[0287] Embodiment 66 is the food or beverage can, or portion thereof, of any of embodiments 64 to 65, wherein the emulsion polymerized latex comprises the emulsion polymerized latex of any one of embodiments 3 to 56.
[0288] Embodiment 67 is the food or beverage can, or portion thereof, of any of embodiments 64 to 66, wherein the coating composition includes any of the features recited in embodiments 3 to 56.
[0289] Embodiment 68 is an inside spray beverage can coating composition, wherein the coating composition comprises an aqueous coating composition that is substantially free of each of styrene and halogenated monomers and is also substantially free of bisphenol A; and wherein the coating composition includes, based on total resin solids, at least 50 wt- % of an emulsion polymerized latex; and wherein the inside spray beverage can coating composition, when spray applied onto an interior of a standard 12 ounce two-piece drawn and ironed aluminum 211 diameter beverage can at a dry film weight of 120 milligrams per can and baked for 50 seconds at an oven temperature of at least 188.degree. C. to achieve a dome peak metal temperature of at least 199.degree. C., gives a metal exposure of less than 20 mA, less than 10 mA, or less than 3.5 mA when tested pursuant to the Metal Exposure after Drop Damage test disclosed herein.
[0290] Embodiment 68' is the inside spray beverage can coating composition of embodiment 68, wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof.
[0291] Embodiment 69 is the inside spray beverage can coating composition of embodiment 68 or 68', wherein the emulsion polymerized latex comprises the emulsion polymerized latex of any one of embodiments 3 to 56.
[0292] Embodiment 70 is the inside spray beverage can coating composition of any of embodiments 68 to 70, wherein the coating composition includes any of the features recited in embodiments 3 to 56.
[0293] Embodiment 71 is the inside spray beverage can coating composition of any of embodiments 68 to 70, wherein the emulsion polymerized latex has a Tg of greater than 40.degree. C., greater than 50.degree. C., or greater than 60.degree. C.
[0294] Embodiment 72 is the inside spray beverage can coating composition of any of embodiments 64 to 71, wherein the emulsion polymerized latex polymer is formed from ingredients including an ethylenically unsaturated monomer component including at least one monomer having (i) a Tg of more than 40.degree. C. and (ii) one or more groups selected from cyclic groups, branched organic groups, or a combination thereof.
[0295] Embodiment 73 is a coating composition comprising: [0296] an emulsion polymerized latex polymer that is substantially free of each of styrene and halogenated monomers and preferably has a glass transition temperature of greater than 40.degree. C., greater than 50.degree. C., or greater than 60.degree. C.; [0297] wherein the coating composition is an aqueous coating composition that is suitable for use in forming a food-contact coating of a metal food or beverage can and is substantially free of bisphenol A; and [0298] wherein the coating composition exhibits an elongation at break of at least 1%, when tested as described herein.
[0299] Embodiment 73' is the coating composition of embodiment 73, wherein the coating composition is substantially free of, more preferably essentially free of, each of bisphenol A, bisphenol F, and bisphenol S, including epoxides thereof
[0300] Embodiment 74 is the coating composition of embodiment 73 or 73' comprising any of the features recited in embodiments 3 to 56.
[0301] Embodiment 75 is a food or beverage can, or a portion thereof, having an interior food-contact coating formed from the coating composition of any of embodiments 73 to 74.
[0302] Embodiment 76 is the food or beverage can, or portion thereof, of embodiment 75 wherein the can comprises an aluminum beverage can, and wherein the interior food-contact coating is an inside spray coating.
[0303] Embodiment 77 is the coating composition, method, or can of any preceding embodiment, wherein the coating composition is substantially free of styrene, and optionally substantially free of substituted styrene compounds.
[0304] Embodiment 78 is the coating composition, method, or can of any preceding embodiment, wherein the coating composition is made without using any polyolefin polymer.
[0305] Embodiment 79 is the coating composition, method, or can of any of embodiments 3 to 78, wherein the acid- or anhydride-functional latex is made without using any non-polymeric surfactant.
[0306] Embodiment 80 is the coating composition, method, or can of any preceding embodiment, wherein the coating composition is made without using a phosphorus acid compound.
[0307] Embodiment 81 is the coating composition, method, or can of any of embodiments 3 to 80, wherein the acid- or anhydride-functional latex is made without using a surfactant that is a polymerizable with at least one ethylenically unsaturated monomer.
[0308] Embodiment 82 is the coating composition, method, or can of any preceding embodiment, wherein the coating composition includes both the NCCR crosslinker (more preferably a beta-hydroxyalkylamide crosslinker) and a phenoplast crosslinker (more preferably a resole phenolic crosslinker).
[0309] Embodiment 83 the coating composition, method, or can of any preceding embodiment, wherein the coating composition includes, based on total resin solids: [0310] 2 to 10 wt- %, 4 to 8.5 wt- %, or 5 to 7.5 wt- % of NCCR crosslinker (more preferably a beta-hydroxyalkylamide crosslinker); and [0311] 1 to 10 wt- %, 3 to 8 wt- %, or 4 to 6 wt- % of phenoplast crosslinker (more preferably a resole phenolic crosslinker).
[0312] Polymers and coating compositions such as those described in the Examples may be evaluated using a variety of tests including:
1. Viscosity Test
[0313] This test measures the viscosity of a latex emulsion or coating composition for rheological purposes, such as for sprayability and other coating application properties. The test is performed pursuant to ASTM D1200-88 using a Ford Viscosity Cup #4 at 25.degree. C. The results are measured in the units of seconds.
2. Curing Conditions
[0314] For beverage inside spray bakes, the curing conditions typically involve maintaining the temperature measured at the can dome at 188.degree. C. to 199.degree. C. for at least 30 seconds.
3. Initial Metal Exposure
[0315] This test method determines the amount of the inside surface of the can that has not been effectively coated by the sprayed coating. This determination is made through the use of an electrically conductive solution (1% NaC1 in deionized water). The interior "inside spray" coating is typically applied using a high pressure airless spray. The following film weights are typically used: 1.6 grams per square meter ("gsm") for a beer can, 2.3 gsm for a soda can, and 3.4 gsm for a can intended for use in packaging a "hard-to-hold" product.
[0316] The coated can is filled with this room-temperature conductive solution, and an electrical probe is attached in contact to the outside of the can (uncoated, electrically conducting). A second probe is immersed in the salt solution in the middle of the inside of the can.
[0317] If any uncoated metal is present on the inside of the can, a current is passed between these two probes and registers as a value on an LED display of a suitable measurement apparatus. The LED displays the conveyed currents in milliamps (mA). The current that is passed is directly proportional to the amount of metal that has not been effectively covered with coating. The goal is to achieve 100% coating coverage on the inside of the can, which would result in an LED reading of 0.0 mA. Preferred coatings give metal exposure values of less than 3 mA, more preferred values of less than 2 mA, and even more preferred values of less than 1 mA. Commercially acceptable metal exposure values are typically less than 2.0 mA on average.
4. Can Formation
[0318] This is a flexibility test for a coating, and correlates to how an inside-spray coating will withstand a can formation process (e.g., necking steps). In this test, the coated can undergoes a can formation process, including a necking step and bottom dome reformation. The formed can is then tested in the electrically conductive solution following the same steps discussed above in the Initial Metal Exposure test.
5. Metal Exposure After Drop Damage
[0319] Drop damage resistance measures the ability of the coated container to resist cracks after being in conditions simulating dropping of a filled can. The ability of a coating to withstand drop damage without rupturing can also be indicative of the ability of the coating to withstand post-coating fabrication steps such as dome reformation and necking. The presence of cracks is measured by passing electrical current via an electrolyte solution, as previously described in the Initial Metal Exposure section. A coated container is filled with the electrolyte solution (1% NaCl in deionized water) and the initial metal exposure is recorded. The electrolyte solution is removed and the can is then filled with room-temperature tap water. For two-piece "inside spray" beverage cans, the film weights described in the Initial Metal Exposure test can be used.
[0320] The water-filled can, which does not include a "top" can end, is dropped through a vertical cylindrical tube having a 2 and 7/8 inch (7.3 centimeter) internal diameter, can bottom down, onto two opposing impact wedges (each wedge provides an inclined plane angled upwards at 33 degrees relative to a horizontal plane orthogonal to the vertical cylindrical tube, with the inclined planes angled outward relative to one another). The impact wedges are positioned relative to the cylindrical tube such that two dents are formed opposite one another in the rim area where the can bottom end meets the sidewall (typically referred to as the "chime" of a beverage can). The water-filled can is dropped through the tube from a 24-inch (61 centimeter) height (as measured between the can bottom and the point of impact on the impact wedges) onto the inclined planes.
[0321] Water is then removed from the can and metal exposure is again measured as described above. If there is no damage, no change in current (mA) will be observed relative to the Initial Metal Exposure value. Typically, an average of 6 or 12 container runs is recorded. The metal exposures results for before and after the drop are reported as absolute values. The lower the milliamp value, the better the resistance of the coating to drop damage. Preferred coatings give metal exposure values after drop damage of less than 3.5 mA, more preferred values of less than 2.5 mA, and even more preferred values of less than 1.5 mA.
6. Adhesion
[0322] Adhesion testing is performed to assess whether the coating adheres to the coated substrate. The adhesion test is performed according to ASTM D 3359 - Test Method B, using SCOTCH 610 tape, available from 3M Company of Saint Paul, Minn. Adhesion is generally rated on a scale of 0-10 where a rating of "10" indicates no adhesion failure (best), a rating of "9" indicates 90% of the coating remains adhered, a rating of "8" indicates 80% of the coating remains adhered, and so on. Adhesion ratings of 10 are typically desired for commercially viable coatings.
7. Blush Resistance
[0323] Blush resistance measures the ability of a coating to resist attack by various solutions. Typically, blush is measured by the amount of solution (e.g., water) absorbed into a coated film. When the film absorbs water, it generally becomes cloudy or looks white. Blush is generally measured visually using a scale of 0-10 where a rating of "10" indicates no blush (best) and a rating of "0" indicates complete whitening of the film (worst). Blush ratings of 7 or higher are typically desired for commercially viable coatings, and optimally 9-10.
[0324] To assess blush, the coating composition to be assessed is spray applied using an airless sprayer to a standard aluminum beverage can.
8. Corrosion Resistance
[0325] These tests measure the ability of a coating to resist attack by solutions of different levels of aggressiveness. Briefly, a given coating is subjected to a particular solution, as described below, and then measured for adhesion and blush resistance (or whitening), each also described below. For each test, a result is given using a scale of 0-10, based on the Adhesion Resistance and/or Blush Resistance, where a rating of "10" is best and a rating of "0" is worst.
A. Acetic Acid Solution
[0326] A 3% solution of acetic acid (C.sub.2H.sub.4O.sub.2) in deionized water is prepared and heated to 100.degree. C. Coated panels are immersed in the heated solution for 30 minutes and then removed, rinsed, and dried. Samples are then evaluated for adhesion and blush, as previously described.
B. Citric Acid Solution
[0327] A 2% solution of citric acid (C.sub.6H.sub.8O.sub.7) in deionized water is prepared and heated while subjected to a pressure sufficient to achieve a solution temperature of 121.degree. C. Coated panels are immersed in the heated solution for 30 minutes and then removed, rinsed, and dried. Samples are then evaluated for adhesion and blush, as previously described.
[0328] 9. Pasteurization
[0329] The Sterilization or pasteurization test determines how a coating withstands the processing conditions for different types of food products packaged in a container. Typically, a coated substrate is immersed in a water bath and heated for 5-60 minutes at temperatures ranging from 65.degree. C. to 100.degree. C. For the present evaluation, the coated substrate was immersed in a deionized water bath for 45 minutes at 85.degree. C. The coated substrate was then removed from the water bath and tested for coating adhesion and blush as described above. Commercially viable coatings preferably provide adequate pasteurization resistance with perfect adhesion (rating of 10) and blush ratings of 5 or more, optimally 9-10.
10. Glass Transition Temperature ("Tg")
[0330] Samples for differential scanning calorimetry ("DSC") testing may be prepared by first applying the liquid resin composition onto aluminum sheet panels. The panels are then baked in a Fisher Isotemp electric oven for 20 minutes at 300.degree. F. (149.degree. C.) to remove volatile materials. After cooling to room temperature, the samples are scraped from the panels, weighed into standard sample pans and analyzed using the standard DSC heat-cool-heat method. The samples are equilibrated at -60.degree. C., then heated at 20.degree. C. per minute to 200.degree. C., cooled to -60.degree. C., and then heated again at 20.degree. C. per minute to 200.degree. C. Glass transitions are calculated from the thermogram of the last heat cycle. The glass transition is measured at the inflection point of the transition.
11. Global Extraction
[0331] The global extraction test is designed to estimate the total amount of mobile material that can potentially migrate out of a coating and into food packed in a coated can. Typically coated substrate is subjected to water or solvent blends under a variety of conditions to simulate a given end use. Acceptable extraction conditions and media can be found in 21 CFR .sctn. 175.300 paragraphs (d) and (e). The allowable global extraction limit as defined by the FDA regulation is 50 parts per million (ppm).
[0332] The extraction procedure used in the current invention is described in 21 CFR .sctn. 175.300 paragraph (e)(4)(xv) with the following modifications to ensure worst-case scenario performance: (1) the alcohol (ethanol) content is increased to 10% by weight, and (2) the filled containers are held for a 10-day equilibrium period at 37.8.degree. C. (100.degree. F.). These conditions are per the FDA publication "Guidelines for Industry" for preparation of Food Contact Notifications.
[0333] The coated beverage can is filled with 10% by weight aqueous ethanol and subjected to pasteurization conditions (65.6.degree. C., 150.degree. F.) for 2 hours, followed by a 10-day equilibrium period at 37.8.degree. C. (100.degree. F.). Determination of the amount of extractives is determined as described in 21 CFR .sctn. 175.300 paragraph (e) (5), and ppm values are calculated based on surface area of the can (no end) of 44 square inches with a volume of 355 milliliters. Preferred coatings give global extraction results of less than 50 ppm, more preferred results of less than 10 ppm, even more preferred results of less than 1 ppm. Most preferably, the global extraction results are optimally non-detectable.
12. Elongation at Break
[0334] Elongation at break can be an indicator of flexibility for a cured coating. A cured coating sample suitable for testing can be prepared using a #12 bar to apply liquid coating to release paper, which is then baked in an oven so that the temperature of the coating reaches 380.degree. F. for 45 seconds with a maximum temperature between 390-400.degree. F. An initial indicator of sample flexibility can be obtained if one is able to remove the sample from the free film. Samples that are too brittle to be tested or removed from the release paper to be tested are noted as too brittle. A sample is then cut from the free film using an ASTM-D638 Type V die using a manual die press. The elongation at break of the cured free-film coating sample can be assessed using a test procedure similar to ASTM-D638-10 "Standard Test Method for Tensile Properties of Plastics". A TA Instruments RSA-G2 with free-film geometry can be used in conjunction with TA Instruments TRIOS software package to analyze experimental measurements for measured behaviors. A sample width of 3.18 millimeters ("mm") and gage length of 7.62 mm can be used. Sample dimensions are entered into the software and the sample is loaded into the film-fiber tension clamp of the instrument. The sample is tested at room temperature and elongated at a Hencky strain rate of 10% per minute to measure the tensile properties of the cured film and determine the elongation at break.
13. Necking Test
[0335] This test measures the flexibility and adhesion of the film following commercial necking process. Necking is done to facilitate the application of a container end that allows sealing the container. The test involves applying the coating to the container at a recommended film thickness and subjecting the container to a recommended bake (see above can, coating, and bake specifications for items 2-4). Prior to the necking process, sample cans typically will have a metal exposure value of <1.0 mA (average of 12 cans) when evaluated using an electrolyte solution as described above. After the necking process, the cans should display no increase in metal exposure compared to the average for 12 non-necked cans. Elevated mA values indicate a fracture in the film which constitutes film failure.
EXAMPLES
[0336] The following examples are offered to aid in understanding of the present invention and are not to be construed as limiting the scope thereof. It is to be understood that the particular examples, materials, amounts, and procedures are to be interpreted broadly in accordance with the scope and spirit of the inventions as set forth herein. Unless otherwise indicated, all parts and percentages are by weight.
Example 1: Styrene-Free Acid-Functional Acrylic Emulsifier
[0337] A premix of 336.35 parts glacial methacrylic acid, 723.15 parts ethyl acrylate (EA), 622.25 parts cyclohexyl methacrylate ("CHMA"), 20.22 parts n-butanol, and 36.99 parts Luperox 26 initiator was prepared in a monomer premix vessel. To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 737.64 parts n-butanol and 42.89 parts deionized water were added. With agitation and an inert blanket, the reaction vessel was heated to 97.degree. C. to 102.degree. C. with reflux occurring. Once within the temperature range, 5.74 parts Luperox 26 initiator was added. Five minutes after the Luperox 26 initiator addition, the monomer-initiator premix was added to the reaction vessel over two and a half hours maintaining the temperature range of 97.degree. C. to 102.degree. C. with reflux and cooling as needed. After the premix additions, the monomer-initiator premix vessel was rinsed with 83.33 parts n-butanol going into the reaction vessel. Immediately after rinsing, a second initiator premix of 7.33 parts Luperox 26 initiator and 60.67 parts n-butanol was added to the reaction vessel over thirty minutes maintaining the temperature range of 97.degree. C. to 102.degree. C. At the end of the addition, the premix vessel was rinsed with 15.5 parts n-butanol and the rinse was added to the reaction vessel. Thirty minutes after rinsing the initiator premix vessel, 1.43 parts Luperox 26 initiator was added to the reaction vessel and rinsed with 40.44 parts n-butanol. The ingredients where allowed to react an additional two hours, at which time 202.22 parts n-butanol and 6.74 parts deionized water were added and the reaction vessel was cooled to less than 60.degree. C. This process gave an acrylic polymeric emulsifier with solids (i.e. non-volatile or "NV") of .about.58.0%, with an acid number of .about.125 mg KOH/g resin, a Brookfield viscosity of .about.25,000 centipoise at 80.degree. F., an Mn of 10,680, a Mw of 37,240, and polydispersity index (PDI) of 3.5. The Tg using DSC was 55.degree. C.
Example 2: Styrene-Free Latex
[0338] To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 111.96 parts of deionized water and 483.35 parts of the acid-functional acrylic polymeric emulsifier of Example 1 were added to the reaction vessel. Next, 32.79 parts dimethyl ethanol amine ("DMEOA") was added over 5-10 minutes while the temperature of the reaction mixture was allowed to increase. The DMEOA addition vessel was rinsed with 6.32 parts deionized water, and the rinse was added to the reaction vessel. Next, 850.30 parts deionized water was added over 30-45 minutes while heating the reaction vessel to 50.degree. C. In a separate vessel, 287.76 parts CHMA, 94.20 parts butyl acrylate, and 38.56 parts glycidyl methacrylate were premixed and stirred until uniform. This monomer premix was then added over 20-25 minutes. When the premix vessel was empty, it was rinsed with 412.61 parts deionized water and the rinse was added to the reaction vessel. The reaction vessel was stirred for 15 minutes to make the contents uniform. Next, 0.811 parts Trigonox TAHP-W85 initiator was added and rinsed with 5.69 parts deionized water. The reaction mixture was stirred for five minutes after which a premix of 0.60 parts erythorbic acid, 51.36 parts deionized water, 0.60 parts DMEOA, and 0.058 parts iron complex was added over one hour. The reaction vessel was allowed to increase in temperature to a maximum of 84.degree. C. When the premix addition was complete, the premix vessel was rinsed with 14.86 parts deionized water and allowed to react for 60 minutes while the temperature allowed to drift down to 55.degree. C. After the 60 minute time, 0.09 parts Trigonox TAHP-W85 initiator was added and rinsed with 0.63 parts deionized water followed by a premix of 0.07 parts erythorbic acid, 5.71 parts deionized water, and 0.07 parts DMEOA rinsed with 1.38 parts deionized water and allowed to react for 60 minutes. The reaction mixture was held for one hour at 55.degree. C. before cooling to below 38.degree. C. This process yielded a latex material containing .about.28% solids, a #4 Ford viscosity of 21 seconds at 80.degree. F., an acid number of 53 mg KOH/g resin, a pH of 7.4, and a particle size of 0.12 microns.
Example 3: Styrene-Free Acid-Functional Acrylic Emulsifier
[0339] A premix of 132.24 parts glacial methacrylic acid, 165.3 parts butyl acrylate, 130.5 parts VeoVa 9 vinyl ester monomer (commercially available from Hexion), 115.7 parts of methyl methacrylate, 12.76 parts Luperox 26 initiator, 54.62 parts butanol, and 4.65 parts deionized water was prepared in a monomer premix vessel. To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 206.71 parts butanol and 10.10 parts deionized water were added. With agitation and an inert blanket, the reaction vessel was heated to 97.degree. C. to 102.degree. C. with reflux occurring. Once within the temperature range, 2.00 parts Luperox 26 initiator was added. Five minutes after the Luperox 26 initiator addition, 14.5 parts VeoVa 9, 6.96 parts methacrylic acid, 6.09 parts methyl methacrylate, and 8.70 parts butyl acrylate were added. After the addition, the monomer-initiator premix was added to the reaction vessel over two and a half hours maintaining the temperature range of 97.degree. C. to 102.degree. C. with reflux and cooling as needed. After the premix additions, the monomer-initiator premix vessel was rinsed with 13.92 parts butanol going into the reaction vessel. Immediately after rinsing, a second initiator premix of 2.53 parts Luperox 26 initiator and 20.92 parts butanol was added to the reaction vessel over thirty minutes maintaining the temperature range of 97.degree. C. to 102.degree. C. At the end of the addition, the premix vessel was rinsed into the reaction vessel with 5.35 parts butanol. Thirty minutes after rinsing the initiator premix vessel, 0.49 parts Luperox 26 initiator was added to the reaction vessel and rinsed with 13.95 parts butanol. The ingredients were allowed to react an additional two hours. After the two hour time, 0.49 parts Luperox 26 initiator was added and allowed to react for 60 minutes. After the 60 minute time, 2.32 parts deionized water and 69.73 parts butanol was added and the reaction vessel cooled to less than 60.degree. C. This process yielded acrylic polymeric emulsifier with solids of .about.56.0% NV, an acid number of 163 mg KOH/g resin, a Brookfield viscosity of 52,000 centipoise at 26.7.degree. C., an Mn of 9,100, an Mw of 30,070, and PDI of about 3.3. The Tg using DSC was 88.degree. C.
Example 4: Styrene-Free Latex
[0340] To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 93.30 parts of deionized water and 402.79 parts of the acid-functional acrylic polymeric emulsifier of Example 3 were added to the reaction vessel. Next, 27.32 parts DMEOA was added over 5-10 minutes while the temperature of the reaction mixture was allowed to increase. The DMEOA addition vessel was rinsed with 5.26 parts deionized water and the rinse was added to the reaction vessel. Next, 708.58 parts deionized water was added over 30-45 minutes while heating the reaction vessel to 50.degree. C. In a separate vessel, 239.80 parts of VeoVa 9 vinyl ester monomer, 78.50 parts butyl acrylate, and 32.13 parts glycidyl methacrylate were premixed and stirred until uniform. This monomer premix was added to the reaction vessel over 20 minutes. When the premix vessel was empty it was rinsed with 343.84 parts deionized water and the rinse was added to the reaction vessel. The reaction vessel was stirred for 15 minutes to make the contents uniform. Next, 0.680 parts Trigonox TAHP-W85 initiator was added and rinsed with 2.36 parts deionized water. The reaction vessel was stirred for five minutes after which a premix of 0.50 parts erythorbic acid, 42.80 parts deionized water, 0.50 parts DMEOA, and 0.05 parts iron complex was added over one hour. The reaction vessel was allowed to increase in temperature to a maximum of 62.degree. C. When the premix addition was complete, the premix vessel was rinsed with 12.38 parts deionized water and allowed to react for 60 minutes while the temperature allowed to drift down to 55.degree. C. After the 60 minute time, 0.08 parts Trigonox TAHP-W85 initiator was added and rinsed with 0.53 parts deionized water followed by a premix of 0.06 parts erythorbic acid, 4.76 parts deionized water, and 0.06 parts DMEOA and allowed to react for 60 minutes. After the 60 minute time, 0.08 parts Trigonox TAHP-W85 initiator was added and rinsed with 0.53 parts deionized water followed by a premix of 0.06 parts erythorbic acid, 4.76 parts deionized water, and 0.06 parts DMEOA rinsed with 1.38 parts deionized water. The material was held for one hour at 55.degree. C. before cooling to below 38.degree. C. This process yielded a latex material with .about.28% solids, a #4 Ford viscosity of 29 seconds at 26.7.degree. C., an acid number of .about.67 mg KOH/g resin, a pH of 7.3, and a particle size of 0.2 microns.
Comparative Example A: Styrene-Containing Acid-Functional Acrylic Emulsifier
[0341] A premix of 115.982 parts glacial methacrylic acid, 249.361 parts ethyl acrylate, 214.567parts styrene, 47.649 parts butanol, and 4.649 parts deionized water was prepared in a monomer premix vessel. In a separate vessel, an initiator premix of 12.756 parts Luperox 26 initiator and 6.973 parts butanol was prepared. To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 206.71 parts butanol and 10.14 parts deionized water were added. With agitation and an inert blanket, the reaction vessel was heated to 97.degree. C. to 102.degree. C. with reflux occurring. Once within the temperature range, 1.979 parts Luperox 26 initiator was added. Five minutes after the Luperox 26 initiator addition, the monomer premix and the initiator premix was added simultaneously to the reaction vessel over two and a half hours maintaining the temperature range of 97.degree. C. to 102.degree. C. with reflux and cooling as needed. After the premix additions, the monomer premix vessel was rinsed with 10.46 parts butanol and the initiator premix vessel was rinsed with 3.487 parts butanol, and both rinses were added to the reaction vessel. Immediately after rinsing, a second initiator premix of 2.528 parts Luperox 26 initiator and 20.919 parts butanol was added to the reaction vessel over thirty minutes maintaining the temperature range of 97.degree. C. to 102.degree. C. At the end of the addition, the premix vessel was rinsed with 5.346 parts butanol and the rinse was added to the reaction vessel. Thirty minutes after rinsing the initiator premix vessel, 0.494 parts Luperox 26 initiator was added to the reaction vessel and rinsed with 13.946 parts butanol. The ingredients were allowed to react an additional two hours when 69.73 parts butanol and 2.324 parts deionized water were added and the reaction vessel was cooled to less than 60.degree. C. This process yielded an acrylic polymeric emulsifier with solids of 58.0% NV, an acid number of .about.130 mg KOH/g resin, a Brookfield viscosity of about 22,000 centipoise at 26.7.degree. C., an Mn of 12,000, a Mw of 29,500, and PDI of about 2.5. The Tg using DSC was 68.degree. C.
Comparative Example B: Styrene-Containing Latex
[0342] To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 201.394 parts acid-functional acrylic polymeric emulsifier of Comparative Example A and 46.65 parts deionized water were added to the reaction vessel. Next, 13.661 parts DMEOA was added over 5-10 minutes with the temperature of the material allowed to increase. The DMEOA was rinsed with 2.632 parts deionized water and the rinse was added to the reaction vessel. Next, 354.29 parts deionized water was added over 30-45 minutes while heating the reaction vessel to 50.degree. C. In a separate vessel, 119.898 parts styrene, 39.248 parts butyl acrylate, and 16.067 parts glycidyl methacrylate were premixed and stirred until uniform. This monomer premix was then added to the reaction vessel over 20-25 minutes. When the premix vessel was empty it was rinsed with 171.92 parts deionized water and the rinse was added to the reaction vessel. The reaction vessel was stirred for 15 minutes to make the contents uniform. Next, 0.338 parts Trigonox TAHP-W85 initiator was added and rinsed with 2.369 parts deionized water. The reaction mixture was stirred for five minutes after which a premix of 0.248 parts erythorbic acid, 21.398 parts deionized water 0.248 parts DMEOA, and 0.024 parts iron complex were added over one hour. The reaction vessel was allowed to increase in temperature to a maximum of 84.degree. C. When the premix addition was complete, the premix vessel was rinsed with 6.19 parts deionized water and allowed to react for 60 minutes while the temperature was allowed to drift down to 55.degree. C. After the 60 minute time, 0.038 parts Trigonox TAHP-W85 initiator was added and rinsed with 0.263 parts deionized water followed by a premix of 0.028 parts erythorbic acid, 2.378 parts deionized water and 0.028 parts DMEOA rinsed with 1.69 parts deionized water. The material was held for 60 minutes at 55.degree. C. before cooling to below 38.degree. C. This process yielded latex materials containing 28.2-30.2% solids, a #4 Ford viscosity of 15-100 seconds, an acid number of 40-60 mg KOH/g resin, a pH of 7.2-8.2, and a particle size of about 0.07-0.14 microns.
Example 5: Inside Spray Coating Compositions
[0343] Inside spray coating compositions were prepared using the ingredients provided below in Table 1, with the ingredients added under agitation in order as provided below in Table 1. DMEOA was used as needed to adjust for final viscosity.
TABLE-US-00001 TABLE 1 Composition Ex. 5, Ex. 5, Ex. 5, Ex. 5, Comparative (Weight Parts) Run 1 Run 2 Run 3 Run 4 Example 5 Example 2 Latex 67.8 65.8 0 0 0 Example 4 Latex 0 0 72.0 69.8 0 Comparative 0 0 0 0 68.1 Example B Latex PRIMID QM1260 0 2.0 0 2.0 0 Crosslinker Deionized Water 18.2 18.1 14.7 14.8 17.6 Water-Miscible 10.7 10.9 10 10 10.5 Organic Solvents Resole Phenolic 0 0 0 0 0.5 Crosslinker Deionized Water 3.0 3.0 3.0 3.0 3.0 DMEOA As Needed As Needed As Needed As Needed As Needed Formulation Solids 20% 20% 20% 20% 20% Viscosity #2 Ford 51 52 65 60 50 Cup (seconds)
[0344] The formulations in Table 1 were sprayed at typical laboratory conditions at 110 mg/can to 130 mg/can (per 12-ounce can) coating weight into the interior of industry standard 12-ounce aluminum beverage cans. The inside-spray coated cans were cured at 188.degree. C. to 199.degree. C. (measured at the can dome) for 30 to 60 seconds through a gas oven conveyor at typical heat schedules for this application. Pertinent application and film coating properties in a beverage can inside spray end use are shown below in Table 2.
TABLE-US-00002 TABLE 2 Coating Properties Ex. 5, Ex. 5, Ex. 5, Ex. 5, Comparative Coating Run 1 Run 2 Run 3 Run 4 Example 5 Initial 1.2 mA 1.2 mA 2.7 mA 1.7 mA 0.9 mA Metal Exposure Metal 223.2 mA 208.9 mA 166.5 mA 5.6 mA 1.3 mA Exposure After Drop Damage Blister Commercially Commercially Commercially Commercially Commercially Resistance Acceptable Acceptable Acceptable Acceptable Acceptable Wetting Pass Pass Pass Pass Pass Adhesion Pass Pass Pass Pass Pass Boiling Pass Pass Pass Pass Pass Water Resistance 2% Citric Pass Pass Pass Pass Pass Acid Resistance
[0345] As shown in the data of Table 2, the styrene-free inside spray coatings of Example 5, Runs 1 and 3 formulated without Primid QM1260 crosslinker exhibited poor flexibility upon drop can challenge. In contrast, the styrene-free inside spray coating of Example 5, Run 4 exhibited both a good initial metal exposure after spray application and a satisfactory metal exposure after drop damage. In addition, the Example 5, Run 4 coating exhibited good blister resistance, substrate wetting, substrate adhesion, boiling water resistance, and 2% citric acid resistance. Thus, the coating of Example 5, Run 4 exhibited a good balance of coating properties for an inside spray beverage can end use. It is believed the drop can damage resistance could be improved by further optimization of the coating formulation such that it is comparable to that of the styrene-containing control (Comparative Example 5).
[0346] As for Example 5, Run 2, the metal exposure after drop can damage was not suitable for an inside spray application. It is unclear why the inside spray coating of Example 5, Run 2 exhibited poor flexibility even when using PRIMID QM1260 crosslinker. Notably, styrene-free latexes having good flexibility, including sufficient flexibility for interior beverage can coatings, were successfully developed using CHMA (see, e.g., Table 6 below). Thus, it is clear that suitable styrene-free latexes may be successfully synthesized using CHMA.
Example 6: Preparation of a Styrene-Free Latex
[0347] A styrene-free latex emulsion was prepared using the ingredients provided in the below Table 3.
TABLE-US-00003 TABLE 3 Weight Weight % Reactor A1 Water 1280.00 37.21 Polyethylene glycol sorbitan 1.92 0.0558 monolaurate (Glycosperse L-20 KFG surfactant) Dioctyl sodium sulfosuccinate 2.40 0.0698 (AEROSOL OT 70 surfactant) Iron sulfate heptahydrate 0.0045 0.0001 Water 45.00 1.31 Part B1 Cyclohexyl methacrylate (CHMA) 406.55 11.82 Ethyl Acrylate 363.64 10.57 Acrylic Acid 116.36 3.38 Hydroxy Ethyl Methacrylate 105.45 3.07 1,4-Butanediol dimethacrylate 176.00 5.12 Part B2 Polyethylene glycol sorbitan 9.28 0.27 monolaurate (Glycosperse L-20 KFG surfactant) Dioctyl sodium sulfosuccinate 12.80 0.37 (AEROSOL OT 70 surfactant) Water 544.00 15.81 Tertioamyl Hydroperoxide 1.60 0.0465 (LUPEROX TAH 85) Initiator C Isoascorbic acid 0.90 0.0262 DMEAO (dimethyl ethanol amine) 0.45 0.0131 Water 147.20 4.28 Part D WATER FLUSH 160.00 4.65 Spike redox (E) Tertioamyl Hydroperoxide 0.64 0.0186 (LUPEROX TAH 85) Isoascorbic acid 0.3200 0.0093 DMEAO 0.16 0.0047 Iron sulfate heptahydrate 0.0032 0.0001 Water 32.00 0.9303 Spike redox (E2) Tertioamyl Hydroperoxide 0.64 0.0186 (LUPEROX TAH 85) Isoascorbic acid 0.3200 0.0093 DMEAO 0.16 0.0047 Iron sulfate heptahydrate 0.0032 0.0001 Water 32.00 0.9303 Total 3439.80 100.00
Process
[0348] 1. Monomer Pre-Emulsion Preparation:
[0349] First, a premix was prepared from all the constituents of part B2. Slow agitation was required at this stage to avoid formation of foam. Once homogeneous, the monomers (part B1) were added under vigorous agitation at room temperature and stirred for 20 minutes. The medium turned white and liquid.
[0350] 2. Latex preparation:
[0351] The ingredients of Part A1 were loaded in the 6L reactor equipped with a reflux condenser, thermometer, mechanical stirred, two metering pumps and nitrogen sparge and the reactor was heated up to 80.degree. C., under moderate agitation.
[0352] The stable monomer pre-emulsion (resulting from Parts B1 and B2) and the initiator solution (premix part C) were then added in the reactor with two separate lines at a constant rate over 180 minutes at 80.degree. C. and under agitation (120 -150 revolutions per minute). Once the monomer addition was completed, Part D1 was added and the mixture held for one hour at 80.degree. C. to reach complete conversion.
[0353] The redox package (Part E) was then added in the reactor to reduce as much as possible the level of free monomers in the resin and then the mixture was held for an additional hour. At this stage, a post-neutralization of the final latex can be envisaged to improve the stability and/or increase the viscosity of the latex.
[0354] The reactor was then slowly cooled down to 40.degree. C. and filtered to collect the resulting latex emulsion. The final latex had a non-volatile content ("NVC") of 34 to-35% (1 g/30 min/ 180.degree. C.). The resulting latex emulsion is referred to as Example 6, Run 1. Additional latexes were prepared in a similar manner using methyl methacrylate in place of CHMA (Example 6, Run 2).
Example 7: Preparation of Styrene-Free Latexes
[0355] Additional latex emulsions were prepared using the process and ingredients of Example 6, with the composition of the monomer premix part B1 employed for each of Runs 1-4 noted below in Table 4. Otherwise the process and materials used were the same as in Example 6. The measured Tg value for each latex is also provided in Table 4. All of the ingredient amounts indicated in the below Table 4 are weight parts.
TABLE-US-00004 TABLE 4 Example 7 Latexes Run 1 Run 2 Run 3 Run 4 Monomers premix part B1 VeoVa 9 MMA (methyl methacrylate) 30 CHMA 34.83 41.7 VeoVa 9 34.83 Ethyl acrylate (EA) 31.13 34.7 22 31.13 Acrylic acid (AA) 9.96 15 11.11 9.96 Hydroxy ethyl methacrylate 9.02 10.3 10.19 9.02 (HEMA) 1,4-Butanediol dimethacrylate 15.06 10 15 15.06 (BDDMA) Tg range (DSC) .degree. C. 60-65 60-65 85-90 55-60
Example 8: Preparation of Styrene-Free, Acid-Functional Acrylic
[0356] This preparation was used in some of the coating compositions as an additive to improve substrate wetting performance.
[0357] A premix of 647.22 parts glacial acrylic acid (GAA), 359.5 parts ethyl acrylate (EA), 431.28 parts methyl methacrylate (MMA), 436.26 parts Butyl CELLOSOLVE, and 48.29 parts deionized water was prepared in a monomer premix vessel. In a separate vessel, an initiator premix of 86.34 parts LUPEROXTM 26 initiator from Arkema and 240 parts butyl CELLOSOLVE was prepared. To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 512.75 parts butyl CELLOSOLVE and 25.15 parts deionized water were added. With agitation and an inert blanket, the reaction vessel was heated to 97 to 102.degree. C. with reflux occurring. Once within the temperature range, 13.40 parts LUPEROX 26 initiator was added. Five minutes after the initiator addition, the monomer premix and the initiator premix were added simultaneously to the reaction vessel over three hours while maintaining the temperature range at 97 to 102.degree. C. with reflux and cooling as needed. The ingredients were allowed to react an additional two hours. If the monomer conversion is not achieved at this stage, an additional initiator premix can be added to the vessel over one hour maintaining the temperature range of 97.degree. C. to 102.degree. C. Sixty minutes after the addition of the second initiator premix, the reaction vessel was cooled to less than 60.degree. C. under agitation. This process yielded an acrylic emulsifying polymer (viz., an acrylic polymeric emulsifier) with solids of .about.55.0% NVC, an acid number of .about.300 mg KOH/g resin.
Example 9: Preparation of Styrene-Free, Acid-Functional Acrylic
[0358] This preparation was used in some of the coating compositions as an additive to improve substrate wetting performance.
[0359] A premix of 647.22 parts glacial acrylic acid (GAA), 359.5 parts ethyl acrylate (EA), 431.28 parts cyclohexyl methacrylate (CHMA), 436.26 parts Butyl CELLOSOLVE, and 48.29 parts deionized water was prepared in a monomer premix vessel. In a separate vessel, an initiator premix of 86.34parts LUPEROX.TM. 26 initiator from Arkema and 240 parts butyl CELLOSOLVE was prepared. To a reaction vessel equipped with a stirrer, reflux condenser, thermocouple, heating and cooling capability, and inert gas blanket, 512.75 parts butyl CELLOSOLVE and 25.15 parts deionized water were added. With agitation and an inert blanket, the reaction vessel was heated to 97 to 102.degree. C. with reflux occurring. Once within the temperature range, 13.40 parts LUPEROX 26 initiator was added. Five minutes after the initiator addition, the monomer premix and the initiator premix were added simultaneously to the reaction vessel over three hours while maintaining the temperature range at 97 to 102.degree. C. with reflux and cooling as needed. The ingredients were allowed to react an additional two hours. If the monomer conversion is not achieved at this stage, an additional initiator premix can be added to the vessel over one hour maintaining the temperature range of 97.degree. C. to 102.degree. C. Sixty minutes after the addition of the second initiator premix, the reaction vessel was cooled to less than 60.degree. C. under agitation.
[0360] This process yielded an acrylic emulsifying polymer (viz., an acrylic polymeric emulsifier) with solids of .about.55.0% NVC, an acid number of .about.300 mg KOH/g resin.
Examples 10-18: Inside Spray Coating Compositions
[0361] The coating compositions of Examples 10-18 were prepared from the latex emulsions of Example 7, Runs 1-4 using the ingredients and amounts indicated in the below Table 5. The acrylic polymeric emulsifiers of Examples 8 and 9 were added in additive levels to improve application of the coating to substrate. Coating compositions were spray applied to the inside of aluminum beverage containers, cured, and evaluated. The coating composition ingredients were added in the order shown in Table 5 with agitation. Ingredients 2 and 3 were premixed before addition. Ingredient 7 was added as needed to obtain a desired final viscosity. All of the ingredient amounts indicated in the below Table 5 are weight parts.
TABLE-US-00005 TABLE 5 Inside Spray Coating Compositions Spray Coating Ex. 10 Ex. 11 Ex. 12 Ex. 13 Ex. 14 Ex. 15 Ex. 16 Ex. 17 Ex. 18 Notable "High" Tg CHMA CHMA CHMA MMA MMA CHMA CHMA VeoVa 9 VeoVa 9 Monomer Latex Tg (.degree. C.) range, DSC 60-65 60-65 60-65 60-65 60-65 80-90 80-90 55-60 55-60 Crosslinker None Primid Phenolic None Primid None Primid None Primid Ingredient 1 Ex. 7, Run 1500.0 1500.0 1500.0 1 Latex Ex. 7, Run 2 1500.0 1500.0 Latex Ex. 7, Run 1500.0 1500.0 3 Latex Ex. 7, Run 1540.0 1540.0 4 Latex Ingredient 2 Deionized 900.0 900.0 940 1140.0 1140.0 930.0 930.0 1065.0 1065.0 water Ingredient 3 DMEOA 10.0 10.0 7.0 6.0 6.0 10.3 10.3 4.6 4.6 Ingredient 4 Water- 349 349 149 3500 350 349 349 356.4 356.4 Miscible Organic Solvents Ingredient 5 Resole 66.0 Phenolic Crosslinker Primid QM 92.0 92.0 91.0 92 1260 Crosslinker (EMS) Ingredient 6 Example 8 41.0 41.0 33.0 33.0 33.0 33.0 Acrylic Emulsifier Example 9 33.0 33.0 33.0 Acrylic Emulsifier Ingredient 7 DMEOA As As As As As As As As As Viscosity in needed needed needed needed needed needed needed needed needed seconds 40 40 37 48 48 40 40 38 38 using ASTM #2 cup at ambient temperature
[0362] The coating compositions of Table 5 were sprayed into the interior of 33 cl (330 milliliter) aluminum beverage cans using typical laboratory conditions and a 100 to 140 mg/can dry coating weight (120 mg/can dry coating weight target), and cured at 180 to 200.degree. C. (as measured at the can dome) for 30 to 60 seconds through a gas oven conveyor from Greenbank Technology Ltd. at typical heat schedules for this application. The application and film properties are shown in Table 6 below.
TABLE-US-00006 TABLE 6 Inside Spray Coating Properties Comp. Ex. 10 Ex. 11 Ex. 12 Ex. 13 Ex. 14 Ex. 15 Ex. 16 Ex. 17 Ex. 18 Ex. C Notable CHMA CHMA CHMA MMA MMA CHMA CHMA VeoVa 9 VeoVa 9 CHMA "High" Tg high Tg high Tg Monomer Latex Tg .degree. C. 60-65 60-65 60-65 60-65 60-65 80-90 80-90 55-60 55-60 80-90 range, DSC Crosslinker None Primid Phenolic None Primid None Primid None Primid None Initial Metal <1 mA <1 mA <1 mA <1 mA <1 mA <1 mA <1 mA <1 mA <1 mA 1.3 mA Exposure Metal >200 mA 0.5 mA 39.6 mA >200 mA 0.3 mA >200 mA 20.5 mA >200 mA 2.6 mA >200 mA Exposure after Drop Damage (from initial) Necking Fail Pass Fail No Data No data Fail Fail No Data No data No data Dome Fail Pass Fail No Data No Data Fail Fail No Data No Data No data Reforming Water Pass Pass Pass Fail Pass Pass Pass Pass Pass Pass Pasteurization 3% Boiling Pass Pass Pass Fail Pass Pass Pass Fail Pass Fail Acetic acid
[0363] The data in Table 6 illustrates that the use of Primid QM 1260 crosslinker allows for production of a styrene-free latex coating composition yielding sufficient flexibility for use as an interior coating of an aluminum beverage can. As illustrated by Example 12, while the use of resole phenolic crosslinker helped to improve drop can damage resistance relative to Example 10 which lacked crosslinker, the use of resole phenolic crosslinker was not capable of yielding a styrene-free latex coating composition having acceptable flexibility for an inside spray aluminum beverage can coating.
[0364] Table 6 also includes Comparative Example C, which is a reproduction of Example 6 of U.S. Publication No. 2016/0009941 formulated for beverage can inside spray application using the same solvent package as the other Examples. Although Comparative Example 6 demonstrated a satisfactory initial metal exposure value after spray application, the coating was inflexible and did not exhibit any meaningful drop can damage resistance. As such, the coating is unsuitable for use as a beverage can coating.
[0365] A coating composition similar to that of Example 14 was tested for elongation at break pursuant to the Elongation at Break test method. (The latex in the sample tested was prepared using all of the same monomers as the Example 7, Run 2 latex employed in the coating formulation of Example 14, with the main difference being that it included 15 wt- % of BDDMA, as opposed to 10 wt- % BDDMA, and had a lower acid number.) When the sample was tested in tensile elongation it exhibited a linear region of deformation before yielding and then further stretched before breaking, as defined in ASTM-D638-10. The latex by itself (i.e., without formulation) could not be tested in tensile elongation without further formulation. For example, when a formulation similar to Example 14 was tested without any Primid QM1260 crosslinker, it was too brittle to be tested and could not be removed from the release paper. This representation of the sample, without the Primid QM1260 crosslinker, was deemed to be highly inflexible with a low elongation to break. It is believed that the addition of the Primid crosslinker to the sample gives the coating mechanical strength and flexibility in reacting with the latex to form a strong film that can be removed from the release paper in addition to be tested in tensile elongation. The tensile elongation at break of the aforementioned fully formulated coating composition similar to Example 14 exhibited an elongated at break ranging from between 5 and 10%, which met or exceeded the tensile elongation at break of a commercial styrene-containing latex coating standard.
[0366] The complete disclosure of all patents, patent applications, and publications (including material safety data sheets, technical data sheets and product brochures for the raw materials and ingredients used in the Examples), and electronically available material cited herein are incorporated herein by reference as if individually incorporated. The foregoing detailed description and examples have been given for clarity of understanding only. No unnecessary limitations are to be understood therefrom. The invention is not limited to the exact details shown and described, for variations obvious to one skilled in the art will be included within the invention defined by the claims. The invention illustratively disclosed herein suitably may be practiced, in some embodiments, in the absence of any element which is not specifically disclosed herein.
* * * * *
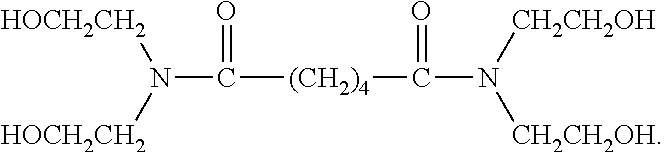



XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.